WO2025010424A1 - Antibodies against staphylococcus antigens and methods of using the same - Google Patents
Antibodies against staphylococcus antigens and methods of using the same Download PDFInfo
- Publication number
- WO2025010424A1 WO2025010424A1 PCT/US2024/036949 US2024036949W WO2025010424A1 WO 2025010424 A1 WO2025010424 A1 WO 2025010424A1 US 2024036949 W US2024036949 W US 2024036949W WO 2025010424 A1 WO2025010424 A1 WO 2025010424A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- antigen
- antibody
- binding
- binding fragment
- fragment
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/12—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from bacteria
- C07K16/1267—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from bacteria from Gram-positive bacteria
- C07K16/1271—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from bacteria from Gram-positive bacteria from Micrococcaceae (F), e.g. Staphylococcus
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/72—Increased effector function due to an Fc-modification
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Definitions
- Staphylococcus aureus causes diverse infections that often overlap with Enterococci and coagulase-negative staphylococci (CoNS), including skin/soft tissue infections, bacteremia/sepsis, hospital -associated pneumonia, endocarditis, neutropenia, hemodialysis associated/line associated bacteremia, osteomyelitis/joint infections, menstrual toxic shock syndrome, and peritonitis.
- CoNS coagulase-negative staphylococci
- antibiotics are the only option for treatment, which has been limited by antibiotic resistance. Attempted vaccines and monoclonal antibodies to-date have not been protective.
- S. aureus Protein A can prevent serum antibodies and IgGl opsonizing monoclonal antibodies from harnessing Fc-mediated immunity. Protein A immune evasion may thwart development of a humoral-based vaccine for S. aureus.
- the lack of an effective vaccine for S. aureus and related Gram-positive bacteria leads to significant morbidity and mortality. There is an unmet need for modalities to prevent and/or treat infection by Staphylococcus and related Gram-positive bacteria.
- Figures 1A-1D show a workflow for discovery, recombinant expression, and characterization of antigen-specific monoclonal antibodies from human donors who recovered from S. aureus infection, and examples of results from the workflow.
- Figures 2A-2P relate to antibody targeting of lipoteichoic acid (LTA).
- Figure 2A shows (left) a table summarizing types of LTA and bacterial species expressing the indicated LTA is found, and (right) a schematic illustration of LTA types (I-V) at a cell membrane).
- Gro glycerol
- Glc glucose
- Gal galactose
- P-Glu P-glucosamine
- AATGal 2-acetamido-4-amino-2,4,6-trideoxy-D-galactose
- GalNAc N-acetylgalactosamine
- Rto ribitol
- GlcNAc Nacetylglucosamine
- P phosphate
- Figure 2B provides a table showing binding (quantified as EC50 ng/ml) of certain anti-LTA antibodies of the present disclosure (SSI18-SSA8) to bacteria expressing Type I, Type II, or Type IV LTA (E coli was included as a reference), and variable domain gene usage of these antibodies.
- the humanized anti-LTA antibody pagibaximab e.g., Patel and Kaufman, Expert Opin Biol Ther 75(4):595-600 (2015); doi: 10.1517/14712598.2015.1019857
- pagibaximab did not reduce sepsis or death.
- Figure 2C shows sensorgram curves showing that antibody SSA12 of the present disclosure does not compete with pagibaximab for LTA binding.
- Figure 2D shows that SSA12 has more consistent binding across Type I LTA bacteria than pagibaximab.
- Figure 2E shows binding (ELISA on LTA) of certain anti-LTA antibodies of the present disclosure, with pagibaximab as a comparator.
- Figure 2F shows binding (EC50 ng/mL on LTA), gene usage, and IMGT CDRH3 amino acid sequence and length of the anti-LTA antibodies.
- VH3-23 is used by 4 monoclonal antibodies produced by 3 donors.
- VH3-7/VK1-6 are used by 10 clonally related monoclonal antibodies produced by 3 donors.
- Figure 2G shows binding (ELISA on fixed bacteria) by certain anti-LTA antibodies of the present disclosure, with pagibaximab as a comparator.
- Figure 2H shows quantified binding by the antibodies to bacteria expressing Type I, Type II, or Type IV LTA, or E. coli.
- Figure 21 shows (left) FACS data showing binding by SSA12 to the indicated bacteria and (right) affinity for LTA, as compared to pagibaximab.
- Figure 2J shows neutralization of infection on TLR2- expressing HEK293 cells. The foregoing data show that SSA12 is a broadly reactive anti-LTA antibody that binds with high affinity to a site on LTA that is not recognized by pagibaximab and can neutralize LTA activity.
- Figure 2K shows a schematic of signaling initiated by binding of LTA to TLR2.
- Figure 2L shows neutralization of LTA activity on TLR2-expressing HEK293 cells by certain anti-LTA antibodies of the present disclosure, and by pagibaximab.
- HEK-Blue hTLR2 cells were co-transfected with hTLR2 and SEAP (secreted embryonic alkaline phosphatase) reporter genes under the control of the IFN-b minimal promoter fused to NF-kB and AP-l-binding sites. Stimulation of TLR2 activates NF-kB and AP-1 which induces the production of SEAP.
- SEAP secreted embryonic alkaline phosphatase
- Figure 2M shows (left) a schematic for a Cl -binding assay and (right) binding to Cl by certain opsonized Fc engineered anti-LTA antibodies of the present disclosure.
- LS M428L/N434S mutations in Fc (increasing affinity for FcRn and in vivo half-life).
- RF H435R/Y436F mutations in Fc (reducing binding to Protein A).
- LS-RF M428L/N434S/H435R/Y436F, wherein Fc amino acid numbering is according to the EU numbering system, with reference to human IgGl . Pagibaximab does not avoid Protein A binding.
- Figure 2N shows (left) a schematic for a Cl -binding assay and (right) binding to Cl by opsonized SSA12 bearing either LS or LS/RF mutations in the Fc.
- FIG. 2P shows (left) Cl binding by SSA12 variant antibodies (pagibaximab and S2X303-LS-RF included as comparators) on S. epidermidis and (right) Cl binding AUC with S. epidermidis versus with S. aureus.
- RPYL R292P/Y300L.
- GARPYL G236A/R292P/Y300L.
- GAYL G236A/Y300L.
- Fc amino acid numbering is according to the EU numbering system, with reference to human IgGl.
- KAEA KAEA
- EFTAE EFTAE mutations
- Figure 3 relates to antibody targeting of glucosaminidase.
- glucosaminidase is a peptidoglycan-modifying enzyme that is highly conserved among Staphylococci and is necessary for cell division.
- Glucosaminidase (Gmd or GlcA) is a domain of the autolysin (Atl) protein, the most predominant peptidoglycan hydrolase in S. aureus. Gmd is located on the cell surface and is highly conserved among S. aureus and S. epidermidis. Gmd catalyzes the hydrolysis of glucosidic linkages and plays a role in cell wall turnover, division, and separation.
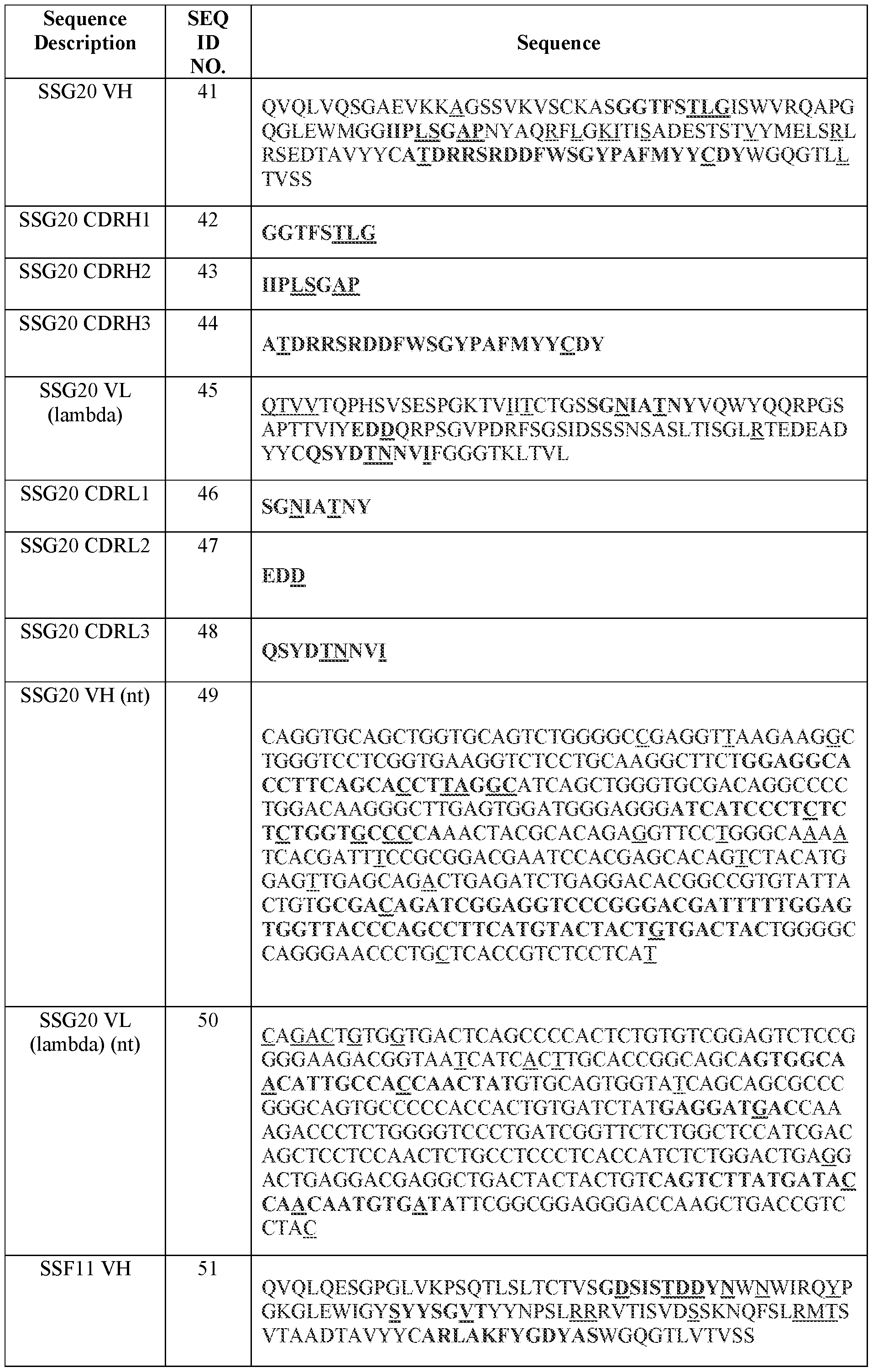
- FIG. 3 shows (right) a schematic illustration of a Gmd neutralization assay and (left, middle) results from ELISA binding studies using antibody SSG20 of the present disclosure (with 1C11 as comparator) against Gmd (left) and fixed bacteria (middle).
- Figures 4A-4F relate to antibody targeting of alphatoxin or bicomponent toxins.
- Staphylococcus secretes two types of P-barrel pore-forming toxins: 1. Alpha toxin; 2. Bicomponent (LukSF, LukED, LukAB, HlgAB and HlgCB). These toxins cause disruption of the plasma membrane, leading to osmotic imbalance and cell death, and are highly inflammatory.
- the vast majority of S. aureus clinical isolates express Hla, HlgABC, and Luk.
- the Luk S- and F-components are highly related structurally and share up to 80% amino acid identity.
- Alpha toxin is a significant contributor to pathogenesis in some S.
- Figures 4C-4F show: (Figure 4C) a schematic activity of bicomponent pore-forming cytotoxins (PFTs) at a cell membrane; (Figure 4D) binding (ELISA, SPR (Octet)) by certain antibodies of the present disclosure against Luk and HIg antigens; (Figure 4E) toxin neutralization on human monocytes; and (Figure 4F) binding competition assays.
- PFTs bicomponent pore-forming cytotoxins
- a variant of SpA Protein A contains substitution mutations in each of the five Ig-binding domains (Q9K, Q10K, D36A, D37A), abolishing the ability to bind Fey or Fab VH3 and promote B cell apoptosis (see Kim et al. J Exp Med, 207(9): 1863-1870 (2010); doi: 10.1084/jem.20092514).
- Figure 5B shows (top) baiting of SPAKKAA memory B cells sorting and (bottom) binding (ELISA; SPR (Octet)) by certain antibodies of the present disclosure.
- Figures 6A-6C relate to targeting of Clumping factor A (ClfA) by cross-reactive antibodies.
- Clumping factor A Clumping factor A
- S. aureus and S. epidermidis express sortase-attached SD repeat (SDR) proteins.
- Clumping factor (Clf) promotes binding of fibrinogen to the bacterial cell and facilitates bacterial clumping.
- Serine aspartate repeat proteins (Sdr) also promote bacterial adhesion.
- Figure 6A shows a schematic of domains of Clf and Sdr proteins of S. aureus and S. epidermidis.
- Figure 6B shows binding (ELISA) by certain antibodies of the present disclosure to ClfA.
- Figures 7A-7C relate to antibody targeting of alpha-toxin.
- Figure 7A shows (left graph) binding and (two graphs at right) neutralization by anti-alpha-toxin antibodies SSE1 and SSE158 of the present disclosure, with MEDI4893 as a comparator.
- MEDI4893 also known as AR-320
- Figure 7B shows results from a synergy/antagonism neutralization study combining anti-alpha-toxin antibodies SSE1 and SSE158.
- Figure 7C shows survival of BALB/c mice prophylactically administered SSE1, SSE158, and/or comparator MEDI4893 at the indicated dose followed by infection (i.n.) with S. aureus. The combination study is repeated and in silica analysis of SSE1 and SSE158 is performed.
- Figures 8A-8C relate to targeting of Gmd by certain antibodies.
- Figures 8A-8B show results from ELISA binding studies using certain anti-Gmd antibodies of the present disclosure against Gmd from S. aureus and S. epidermidis.
- Figure 8C provides a table showing quantified binding of certain anti-Gmd antibodies of the present disclosure to Gmd (S. aureus and S. epidermidis), quantified binding to bacterial strains, and variable domain gene usage of these antibodies. 1C11 was included as a comparator.
- Figures 9A and 9B relate to antibody targeting of LTA.
- Figure 9A LTA neutralization (reported as absolute EC50 ng/mL) for the antibodies shown in Figure 2F.
- Figures 10A-10F relate to targeting of ClfA by certain antibodies.
- Figure 10A shows binding by certain anti -ClfA antibodies of the present disclosure across strains of S. aureus with different ClfA genotypes, with E. coli included as a negative control.
- the humanized anti-ClfA antibody tefibazumab e.g., Weems JJ Jr, Steinberg JP, Filler S, et al. Antimicrob Agents Chemother. 2006;50(8):2751-2755. doi: 10.1128/AAC.00096-06
- Tefibazumab has been tested in a Phase 2 study for treatment of S. aureus in cystic fibrosis patients and treatment of S.
- Figure 13 shows a graph comparing half-life of antibodies “FYl-rlgGl-LS” and “FY1- rlgGl-LS-RF” in Tg32 SCID mice expressing human FcRn. FYI recognizes influenza HA.
- FIG. 14B shows (left) a schematic of FcyR binding measured by SPR and (right) a table showing FcyR binding by the indicated SSA12-LS-RF (or variant) antibodies, quantified by SPR. The values provided show a Log2 fold change compared to SSA12-LS-RF binding.
- FIG 141 shows Staphylococcus survival in a whole blood assay after exposure to isotype control (S2X303-LS-RF) and SSA12-LS-RF (or variant) antibodies, tested by donor. In each graph, Staphylococcal survival for isotype control and the tested mAb is shown for each donor. Each line represents the change in survival with addition of the tested mAb.
- Figure 14J shows (left) percent survival of S.
- Figure 19 shows binding to Cl by anti-LTA antibodies SSA8 and SSA12 of the present disclosure.
- Pagibaximab was included as a comparator.
- Figure 20 shows neutralization, by anti-Gmd antibodies SSG20 and SSF11 of the present disclosure, in a cell wall digestion assay. 1C11 was included as a comparator.
- Figure 21 shows quantified binding values (EC50 ng/mL to ClfA_001, ClfA_002, ClfA_004, and S. aureus strains). Tefibazumab was included as a comparator.
- Figure 22 shows survival of BALB/c mice prophylactically administered SSE1, SSE158, MEDI4893, or an isotype control at the indicated dose followed by infection (i.n.) with S. aureus, as indicated.
- Figure 23 relates to antibody targeting of Hla.
- Figure 24 provides a table showing results from Hla-binding (quantified as EC50 ng/mL) and Hla neutralization (on THP-1 cells, quantified as IC50 ng/mL) assays by certain anti-Hla antibodies of the present disclosure.
- Figure 25 relates to study design for testing efficacy of monoclonal antibodies against Staphylococcus targets in FPR3757 bacteremia model in BL6 mice.
- Figure 26 provides a schematic (top) of the study design and a table showing the descriptions of the various study groups, antibody dose, antibody delivery route, antibody dosing time, group size, Staphylococcus aureus strain and dosage used to challenge, route of infection, take down day and read outs, and days for clinical observation.
- Figures 27A-27B relate to promotion of clearance of Staphylococcus aureus infection by select n -Staphylococcus antibodies within a subset of treated mice in a disseminated infection model.
- Figure 27A provides a graph showing effects of antibody treatment on bacterial kidney burden as measured by kidney CFU.
- Figure 27B provides a graph showing effects of antibody treatment on weight loss.
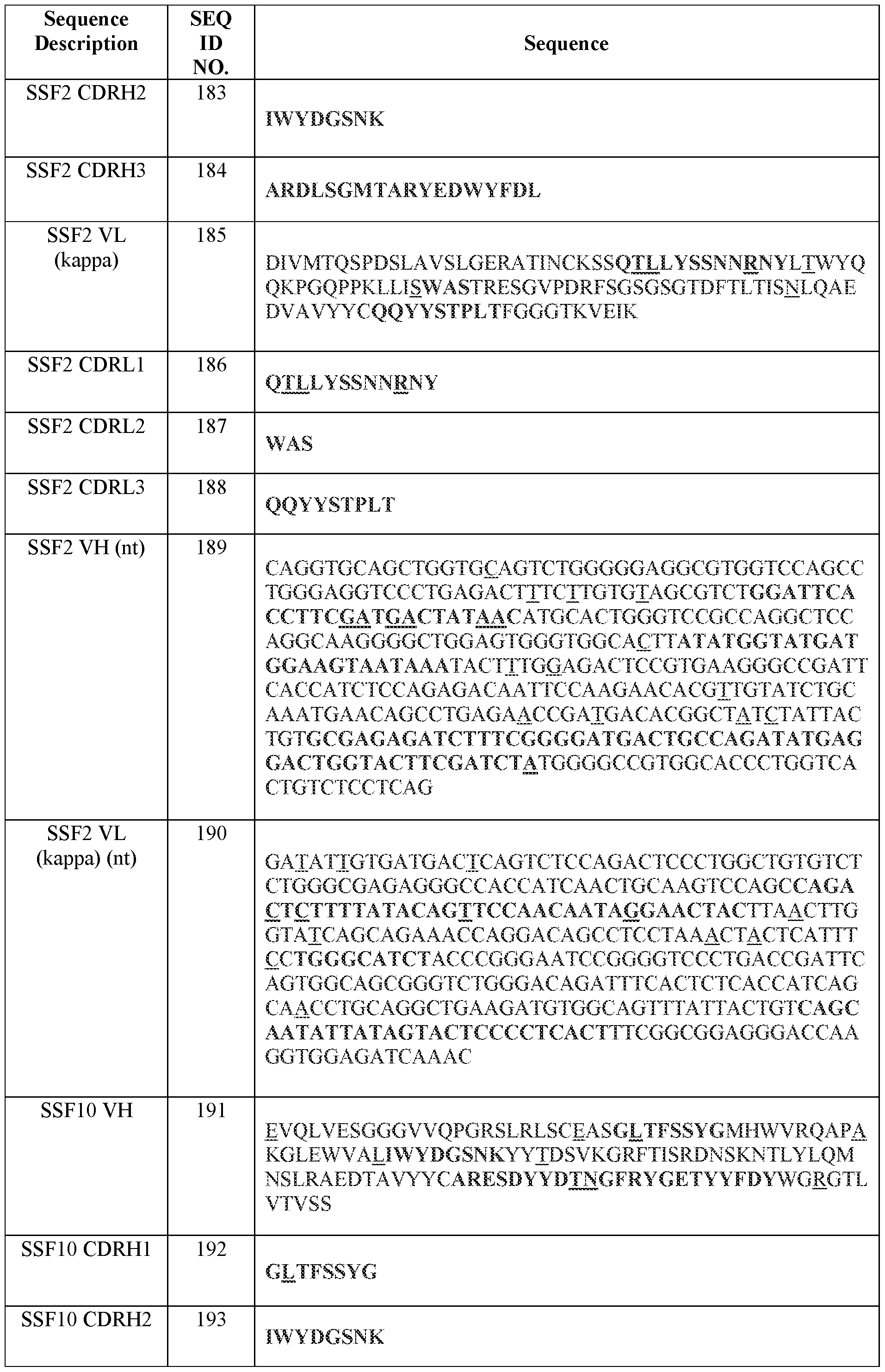
- Figure 28 provides graphs showing individual mouse weight loss within each antibody study group.
- the antigen is an antigen that is also expressed by one or more (e.g., gram-positive) bacteria that is not of genus Staphylococcus, and the antibody or antigenbinding fragment is capable of binding thereto.
- a "Staphylococcus antigen” is not limited to antigens expressed solely by bacteria of genus Staphylococcus and includes antigens that are expressed by Staphylococcus and by bacteria of one or more other genus.
- the antigen is also expressed by a bacteria of genus Enterococcus (e.g., E. faecalis), a bacteria of genus Lactococcus (e.g., L.
- the antibody or antigen-binding fragment is capable of binding to S. aureus e.g., MRS A), Coagulase-negative staphylococci (CoNS) such as S. epidermidis, S. pneumoniae, S. lugdunensis, S. hominis, or any combination thereof.
- the antibody or antigen-binding fragment is capable of binding to: S. aureus FPR3757, S. aureus NE284 TE2 mutant, S. epidermidis RP62A, S. epidermidis 1200, S. epidermidis NIH04008, S. lugdunensis e.g., N860297), S. pyogenes, E. faecalis, S. agalactiae, S. hominis, L. garvieae, S. pneumoniae, E. coli, or any combination of thereof.
- the antibody or antigen-binding fragment binds to LTA with higher affinity than does pagibaximab.
- pagibaximab binds to an LTA with a Kd of 1.75E-1 IM and an antibody or antigen-binding fragment of the present disclosure binds to the LTA with a Kd of 3.48E-12M, as determined by surface plasmon resonance (SPR, e.g. using an Octet instrument).
- the antibody or antigen-binding fragment binds to Hla with a Kd of 1.96E-10 M or of 6.14E-11 M, and MEDI4893 binds to the Hla with a Kd of 5.1E-10 by the same assay.
- the antibody or antigen-binding fragment neutralizes Hla (e.g., in an assay using rabbit RBCs).
- an antibody or antigen-binding fragment of the present disclosure is capable of binding to S. aureus HIgB.
- an antibody or antigen-binding fragment of the present disclosure is capable of binding to a SpA.
- the antibody or antigen-binding fragment is capable of binding to SpA with a Kd of about 2.46E-09 M, about 1.1 IE-08 M, about 7.633E- 10 M, 1.77E-09 M, or about 1.44E-09 M, as determined by surface plasmon resonance using an Octet instrument.
- an antibody or antigen-binding fragment of the present disclosure is capable of binding to a ClfA.
- the antibody or antigen-binding fragment is capable of binding to ClfA OOl, to ClfA_002 to ClfA_004, or to any combination thereof.
- an antibody or antigen-binding fragment of the present disclosure is capable of binding to a Sbi.
- an antibody or antigen-binding fragment is human, humanized, or chimeric.
- the antibody or antigen-binding fragment comprises human amino acid sequences, e.g. : one, two, three, four, five, or six human CDR sequences; one, two, three, four, five, six, seven, or eight human variable domain framework region sequences.
- the antibody or antigen-binding fragment comprises an Fc polypeptide, which can be, for example, a human Fc polypeptide or an engineered variant of a human Fc polypeptide.
- any concentration range, percentage range, ratio range, or integer range is to be understood to include the value of any integer within the recited range and, when appropriate, fractions thereof (such as one tenth and one hundredth of an integer), unless otherwise indicated.
- any number range recited herein relating to any physical feature, such as polymer subunits, size or thickness are to be understood to include any integer within the recited range, unless otherwise indicated.
- the term “about” means ⁇ 20% of the indicated range, value, or structure, unless otherwise indicated. It should be understood that the terms “a” and “an” as used herein refer to “one or more” of the enumerated components.
- a “conservative substitution” refers to amino acid substitutions that do not significantly affect or alter binding characteristics of a particular protein. Generally, conservative substitutions are ones in which a substituted amino acid residue is replaced with an amino acid residue having a similar side chain. Conservative substitutions include a substitution found in one of the following groups: Group 1 : Alanine (Ala or A), Glycine (Gly or G), Serine (Ser or S), Threonine (Thr or T); Group 2: Aspartic acid (Asp or D), Glutamic acid (Glu or Z); Group 3: Asparagine (Asn or N), Glutamine (Gin or Q); Group 4: Arginine (Arg or R), Lysine (Lys or K), Histidine (His or H); Group 5: Isoleucine (He or I), Leucine (Leu or L), Methionine (Met or M), Valine (Vai or V); and Group 6: Phenylalanine (Phe or F), Tyrosine (Tyr or
- Modified nucleosides may include pseudouridine, such as N 1 - methylpseudouridine, 5-methylcytidine, 2-thiouridine, N6-methyladenonsine.
- methylation of a naturally occurring (unmodified) nucleotide may be used alone or in combination with a modified nucleotide to achieve any of the above effects achievable using a modified nucleotide.
- the proportion of nucleotides having a specific base or located in a specific sequence that are methylated may be used to achieve such effects.
- methylation may be used to reduce recognition of the mRNA (or, in the case of DNA therapeutics, the DNA) as foreign in a mammalian host cell.
- the mRNA, particularly saRNA or taRNA may include a cap or a cap analog, more specifically a 7-methylguanosine moiety linked via a phosphate, particularly a trisphosphate to an end nucleotide.
- the including of a cap or cap analog may help prevent exonuclease cleavage of the mRNA, particularly saRNA or taRNA, and/or initiate translation of the mRNA, particularly saRNA or taRNA, in a mammalian host cell.
- the cap or cap analog may initiate translation of the replication protein or peptide.
- circRNA may lack a suitable location for a cap or cap analog because of the absence of an otherwise unbound 5’ end.
- linear mRNA, prior to circularization to form circRNA may contain a cap or cap analog to, for example, increase stability and/or amplification of the linear mRNA prior to circularization.
- UTRs may be 5’ and 3’ of the sequence encoding at least one, typically each, protein or peptide, or 5’ and 3’ of the sequence encoding at least two proteins or peptides, all proteins or peptides that function together once expressed (e.g. theVH and VL-containing sequences), or all proteins and peptides encoded by the mRNA.
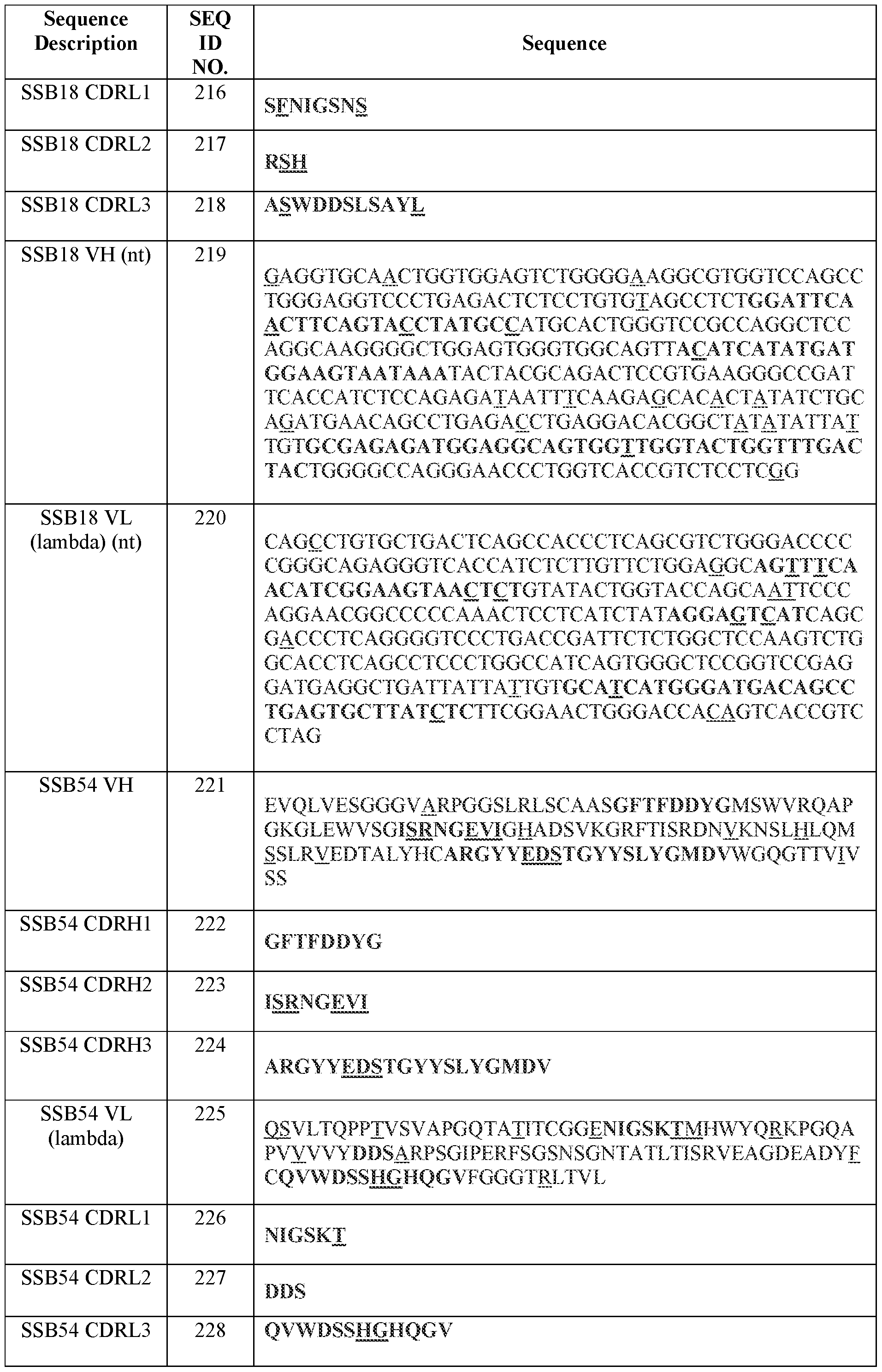
- the polyA tail prior to initial translation of mRNA, particularly saRNA or taRNA, particularly taRNA sequences encoding amplification proteins, containing the polyA tail, may have a length of at least or approximately 250, 200, 100, 50, 20, or 10 nucleotides, or a length in a range between 10 and 250, 10 and 200, 10 and 100, 10 and 50, 10 and 20, 20 and 250, 20 and 200, 20 and 100, 20 and 50, 50 and 250, 50 and 200, 50 and 100, 100 and 250, 100 and 200, or 200 and 250 nucleotides.
- the mRNA, including circRNA, taRNA, or saRNA may be produced in a production host cell, which is a type of host cell.
- the mRNA, including circRNA, taRNA, or saRNA may be produced in a cell-free system, such as a system using a DNA template and enzymes.
- isolated can, in some embodiments, also describe an antibody, antigen-binding fragment, polynucleotide, vector, host cell, or composition that is outside of a human body. In certain embodiments, an isolated antibody, antigen-binding fragment, polynucleotide, vector, host cell, or composition is provided.
- gene means the segment of DNA or RNA involved in producing a polypeptide chain; in certain contexts, it includes regions preceding and following the coding region (e.g., 5’ untranslated region (UTR) and 3’ UTR) as well as intervening sequences (introns) between individual coding segments (exons).
- regions preceding and following the coding region e.g., 5’ untranslated region (UTR) and 3’ UTR
- intervening sequences introns between individual coding segments (exons).
- a “functional variant” refers to a polypeptide or polynucleotide that is structurally similar or substantially structurally similar to a parent or reference compound of this disclosure, but differs slightly in composition (e.g., one base, atom or functional group is different, added, or removed), such that the polypeptide or encoded polypeptide is capable of performing at least one function of the parent polypeptide with at least 50% efficiency, preferably at least 55%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, at least 99.9%, or 100% level of activity of the parent polypeptide.
- a functional variant of a polypeptide or encoded polypeptide of this disclosure has “similar binding,” “similar affinity” or “similar activity” when the functional variant displays no more than a 50% reduction in performance in a selected assay as compared to the parent or reference polypeptide, such as an assay for measuring binding affinity (e.g., Biacore® or tetramer staining measuring an association (Ka) or a dissociation (KD) constant).
- binding affinity e.g., Biacore® or tetramer staining measuring an association (Ka) or a dissociation (KD) constant.
- a “functional portion” or “functional fragment” refers to a polypeptide or polynucleotide that comprises only a domain, portion or fragment of a parent or reference compound, and the polypeptide or encoded polypeptide retains at least 50% activity associated with the domain, portion or fragment of the parent or reference compound, preferably at least 55%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, at least or 99.9%, or 100% level of activity of the parent polypeptide, or provides a biological benefit (e.g., effector function).
- a biological benefit e.g., effector function
- a “functional portion” or “functional fragment” of a polypeptide or encoded polypeptide of this disclosure has “similar binding” or “similar activity” when the functional portion or fragment displays no more than a 50% reduction in performance in a selected assay as compared to the parent or reference polypeptide (preferably no more than 20% or 10%, or no more than a log difference as compared to the parent or reference with regard to affinity).
- homologous refers to a gene, protein, compound, nucleic acid molecule, or activity found in or derived from a host cell, species, or strain.
- a heterologous or exogenous polynucleotide or gene encoding a polypeptide may be homologous to a native polynucleotide or gene and encode a homologous polypeptide or activity, but the polynucleotide or polypeptide may have an altered structure, sequence, expression level, or any combination thereof.
- a non-endogenous polynucleotide or gene, as well as the encoded polypeptide or activity may be from the same species, a different species, or a combination thereof.
- a nucleic acid molecule or portion thereof native to a host cell will be considered heterologous to the host cell if it has been altered or mutated, or a nucleic acid molecule native to a host cell may be considered heterologous if it has been altered with a heterologous expression control sequence or has been altered with an endogenous expression control sequence not normally associated with the nucleic acid molecule native to a host cell.
- heterologous can refer to a biological activity that is different, altered, or not endogenous to a host cell.
- endogenous or “native” refers to a polynucleotide, gene, protein, compound, molecule, or activity that is normally present in a host cell or a subject.
- expression refers to the process by which a polypeptide is produced based on the encoding sequence of a nucleic acid molecule, such as a gene.
- the process may include transcription, post-transcriptional control, post-transcriptional modification, translation, post-translational control, post-translational modification, or any combination thereof.
- An expressed nucleic acid molecule is typically operably linked to an expression control sequence (e.g., a promoter).
- operably linked refers to the association of two or more nucleic acid molecules on a single nucleic acid fragment so that the function of one is affected by the other.
- a promoter is operably linked with a coding sequence when it is capable of affecting the expression of that coding sequence (i.e., the coding sequence is under the transcriptional control of the promoter).
- Unlinked means that the associated genetic elements are not closely associated with one another and the function of one does not affect the other.
- construct refers to any polynucleotide that contains a recombinant nucleic acid molecule (or, when the context clearly indicates, a fusion protein of the present disclosure).
- a (polynucleotide) construct may be present in a vector (e.g., a bacterial vector, a viral vector) or may be integrated into a genome.
- a “vector” is a nucleic acid molecule that is capable of transporting another nucleic acid molecule.
- Vectors may be, for example, plasmids, cosmids, viruses, a RNA vector or a linear or circular DNA or RNA molecule that may include chromosomal, non-chromosomal, semi -synthetic or synthetic nucleic acid molecules.
- Vectors of the present disclosure also include transposon systems (e.g., Sleeping Beauty, see, e.g., Geurts et al., Mol. Ther. 5: 108, 2003: Mates et al., Nat. Genet. 41'.753, 2009).
- the vector may replicate and function independently of the host genome, or may, in some instances, integrate into the genome itself or deliver the polynucleotide contained in the vector into the genome without the vector sequence.
- plasmid “expression plasmid,” “virus,” and “vector” are often used interchangeably.
- the “vector” may comprise or consist of mRNA, such as saRNA, taRNA, or circRNA.
- the term “introduced” in the context of inserting a nucleic acid molecule into a cell means “transfection”, “transformation,” or “transduction” and includes reference to the incorporation of a nucleic acid molecule into a eukaryotic or prokaryotic cell wherein the nucleic acid molecule may be incorporated into the genome of a cell (e.g., chromosome, plasmid, plastid, or mitochondrial DNA), converted into an autonomous replicon (e.g. a replicon formed by saRNA), or transiently expressed e.g., transfected mRNA, such as circRNA, taRNA, or saRNA).
- a cell e.g., chromosome, plasmid, plastid, or mitochondrial DNA
- an autonomous replicon e.g. a replicon formed by saRNA
- transiently expressed e.g., transfected mRNA, such as
- “Retroviruses” are viruses having an RNA genome, which is reverse-transcribed into DNA using a reverse transcriptase enzyme, the reverse-transcribed DNA is then incorporated into the host cell genome.
- “Gammaretrovirus” refers to a genus of the retroviridae family. Examples of gammaretroviruses include mouse stem cell virus, murine leukemia virus, feline leukemia virus, feline sarcoma virus, and avian reticuloendotheliosis viruses.
- Lentiviral vectors include HIV-based lentiviral vectors for gene delivery, which can be integrative or non-integrative, have relatively large packaging capacity, and can transduce a range of different cell types. Lentiviral vectors are usually generated following transient transfection of three (packaging, envelope, and transfer) or more plasmids into producer cells. Like HIV, lentiviral vectors enter the target cell through the interaction of viral surface glycoproteins with receptors on the cell surface. On entry, the viral RNA undergoes reverse transcription, which is mediated by the viral reverse transcriptase complex. The product of reverse transcription is a double-stranded linear viral DNA, which is the substrate for viral integration into the DNA of infected cells.
- a host cell may include any individual cell or cell culture which may receive a vector or the incorporation of nucleic acids or express proteins. The term also encompasses progeny of the host cell, whether genetically or phenotypically the same or different. Suitable host cells may depend on the vector and may include mammalian cells, animal cells, human cells, simian cells, insect cells, yeast cells, and bacterial cells. These cells may be induced to incorporate the vector or other material by use of a viral vector, transformation via calcium phosphate precipitation, DEAE-dextran, electroporation, microinjection, or other methods. See, for example, Sambrook el al., Molecular Cloning: A Laboratory Manual 2d ed. (Cold Spring Harbor Laboratory, 1989).
- epitope includes any molecule, structure, amino acid sequence, or protein determinant that is recognized and specifically bound by a cognate binding molecule, such as an immunoglobulin, or other binding molecule, domain, or protein.
- Epitopic determinants generally contain chemically active surface groupings of molecules, such as amino acids or sugar side chains, and can have specific three-dimensional structural characteristics, as well as specific charge characteristics.
- a bacteria of genus Streptococcus e.g., S. pyogenes, S. agalacliae
- a bacteria of genus Listeria e.g., L. monocytogenes
- a bacteria of genus Clostridium e.g., C. inocuum, C. difficile
- a Type I LTA can be found on Staphylococcus aureus and other Staphylococci, as well as on Enterococci (e.g., E. faecalis), L.
- binding can be determined by recombinantly expressing an antigen in a host cell (e.g, by transfection) and immunostaining the (e.g, fixed, or fixed and permeabilized) host cell with antibody and analyzing binding by flow cytometry (e.g., using a ZE5 Cell Analyzer (BioRad®) and FlowJo software (TreeStar).
- positive binding can be defined by differential staining by antibody of antigen-expressing cells versus control (e.g., mock) cells.
- the antibody or antigen-binding fragment is capable of binding to the antigen with a KD in a range from about 1.0E-9 M to about 3.5E-12 M, such as, for example, about 1.0E-9 M, about 1.5E-9 M, about 2.0E-9 M, about 2.5E-9 M, about 3.0E-9 M, about 3.5E-9 M, about 4.0E-9 M, about 4.5E-9 M, about 5.0E-9 M, about 5.5E-9 M, about 6.0E-
- the binding is as assessed by biolayer interferometry (BLI). In other embodiments, the binding is as assessed by surface plasmon resonance (SPR).
- the antibody or antigen-binding fragment is capable of activating a human FcyRIIIa.
- activation is as determined using a host cell (optionally, a Jurkat cell) comprising: (i) the human FcyRIIIa (optionally, a F158 allele); and (ii) a NF AT expression control sequence operably linked to a sequence encoding a reporter, such as a luciferase reporter, following incubation (e.g., of 23 hours) of the antibody or antigen-binding fragment with a target cell (e.g., a A549 cell) infected with an antigen-expressing bacteria.
- the antibody or antigen-binding fragment is capable of treating and/or preventing an infection by an antigen-expressing bacteria in a subject.
- antibody refers to an intact antibody comprising at least two heavy (H) chains and two light (L) chains inter-connected by disulfide bonds, as well as any antigen-binding portion or fragment of an intact antibody that has or retains the ability to bind to the antigen target molecule recognized by the intact antibody, such as an scFv, Fab, or Fab’2 fragment.
- antibody herein is used in the broadest sense and includes polyclonal and monoclonal antibodies, including intact antibodies and functional (antigen-binding) antibody fragments thereof, including fragment antigen binding (Fab) fragments, F(ab’)2 fragments, Fab’ fragments, Fv fragments, recombinant IgG (rlgG) fragments, single chain antibody fragments, including single chain variable fragments (scFv), and single domain antibodies (e.g., sdAb, sdFv, nanobody) fragments.
- Fab fragment antigen binding
- rlgG recombinant IgG
- scFv single chain variable fragments
- single domain antibodies e.g., sdAb, sdFv, nanobody
- the term encompasses genetically engineered and/or otherwise modified forms of immunoglobulins, such as intrabodies, peptibodies, chimeric antibodies, fully human antibodies, humanized antibodies, and heteroconjugate antibodies, multispecific, e.g., bispecific antibodies, diabodies, triabodies, tetrabodies, tandem di-scFv, and tandem tri-scFv.
- antibody should be understood to encompass functional antibody fragments thereof.
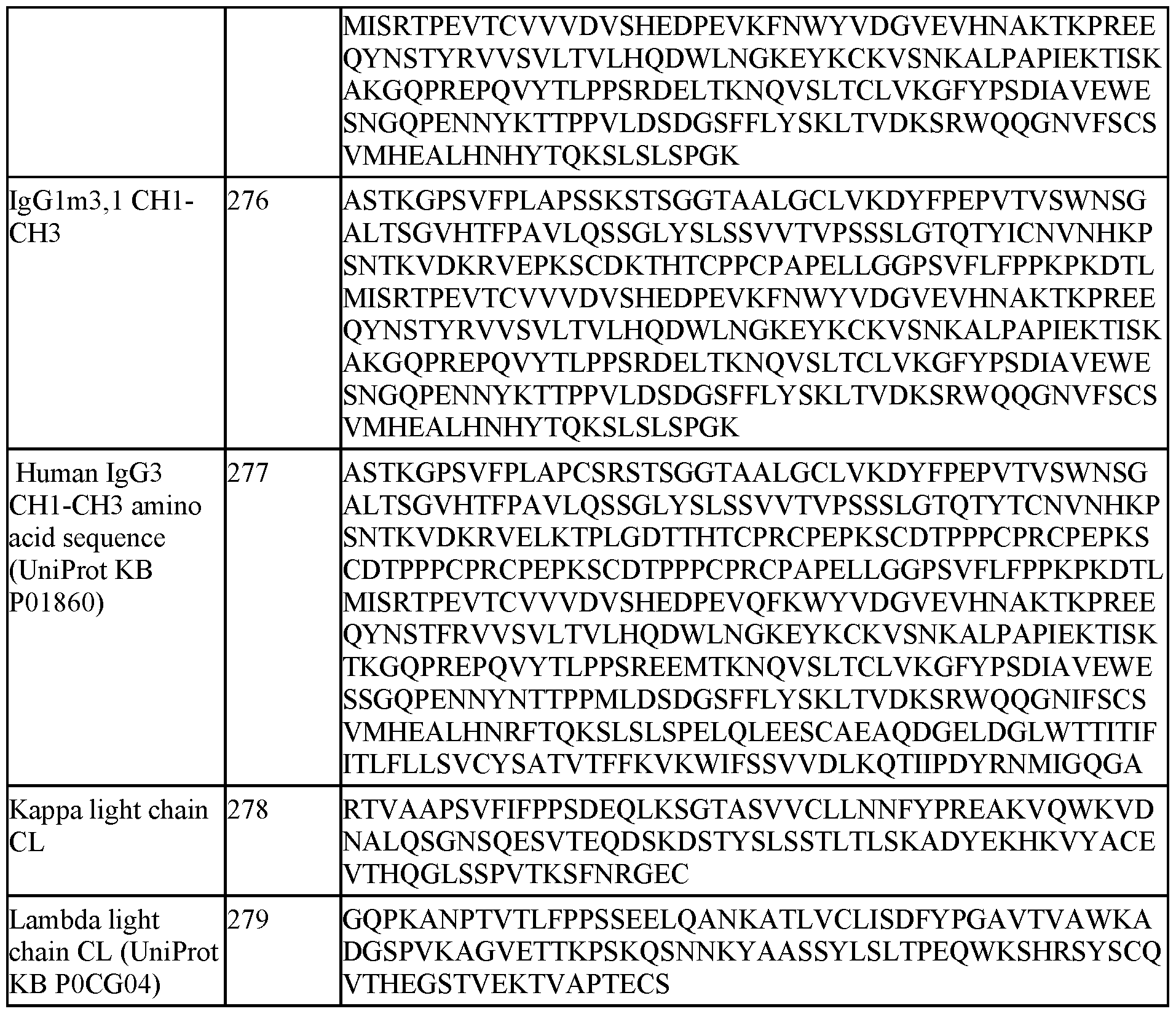
- the term also encompasses intact or full-length antibodies, including antibodies of any class or sub-class, including IgG and sub-classes thereof (IgGl, IgG2, IgG3, IgG4), IgM, IgE, IgA, and IgD.
- variable binding region also called variable region or variable binding domain or variable domain
- a VL is a kappa (K) class (also “VK” herein).
- a VL is a lambda ( ) class.
- the variable binding regions comprise discrete, well-defined sub-regions known as “complementarity determining regions” (CDRs) and “framework regions” (FRs).
- CDR complementarity determining region
- HVR hypervariable region
- an antibody VH comprises four FRs and three CDRs as follows: FR1- HCDR1-FR2-HCDR2-FR3-HCDR3-FR4; and an antibody VL comprises four FRs and three CDRs as follows: FR1-LCDR1-FR2-LCDR2-FR3-LCDR3-FR4.
- the VH and the VL together form the antigen-binding site (or domain) through their respective CDRs.
- one or more CDRs do not contact antigen and/or do not contribute energetically to antigen binding (but at least one CDR does contact antigen).
- a “variant” of a CDR refers to a functional variant of a CDR sequence having up to 1-3 amino acid substitutions (e.g., conservative or non-conservative substitutions), deletions, or combinations thereof.
- Numbering of CDR and framework regions may be according to any known method or scheme, such as the Kabat, Chothia, EU, IMGT, Contact, North, Martin, AbM, and Aho numbering schemes see, e.g., Kabat et al., “Sequences of Proteins of Immunological Interest, US Dept. Health and Human Services, Public Health Service National Institutes of Health, 1991, 5 th ed.; Chothia and Lesk, J. Mol. Biol. 796:901-917 (1987)); Lefranc etal., Dev. Comp. Immunol. 27:55, 2003; Honegger and Pliickthun, J. Mol. Bio. 309:657-670 (2001); North et al. J Mol Biol.
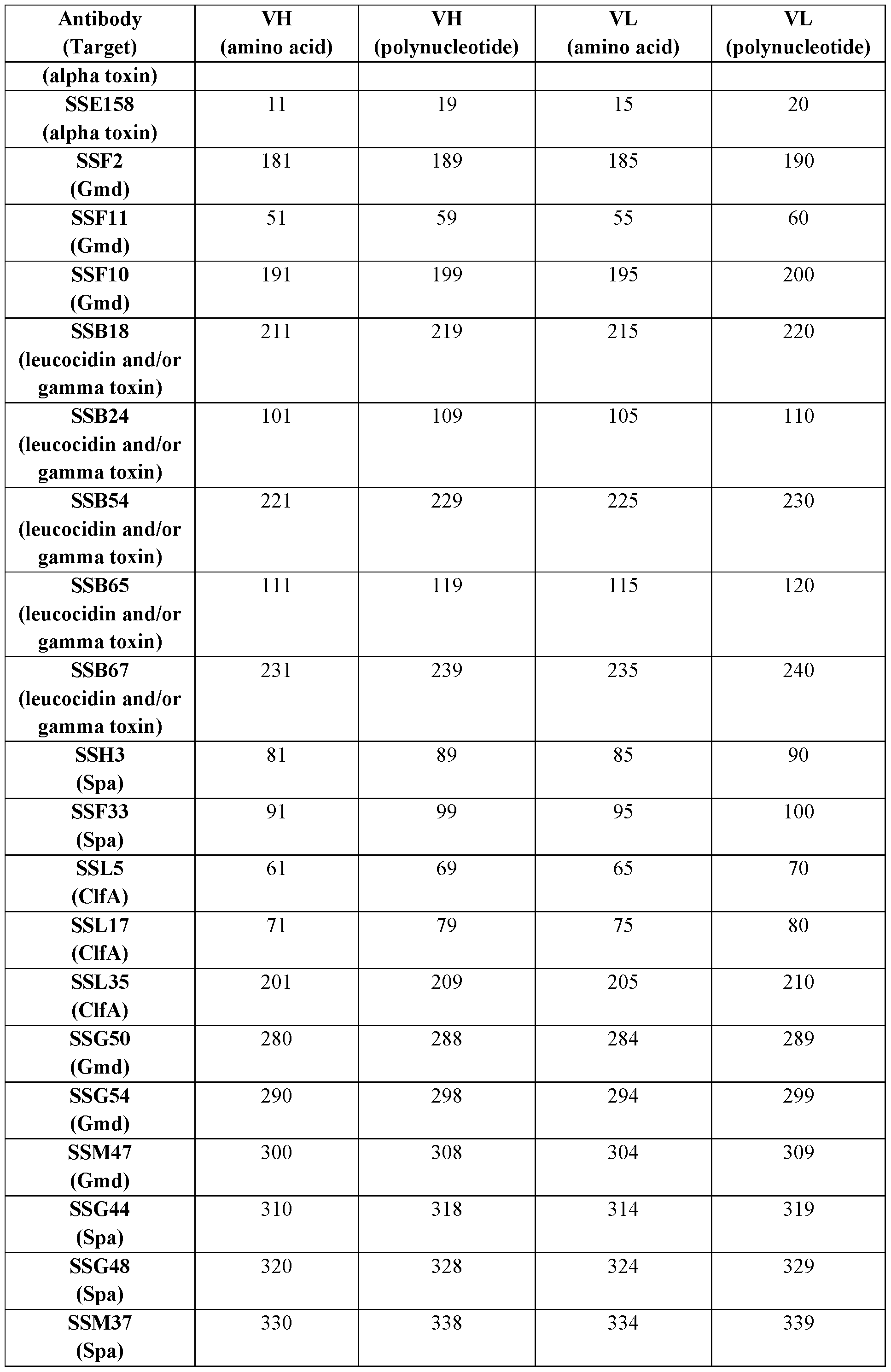
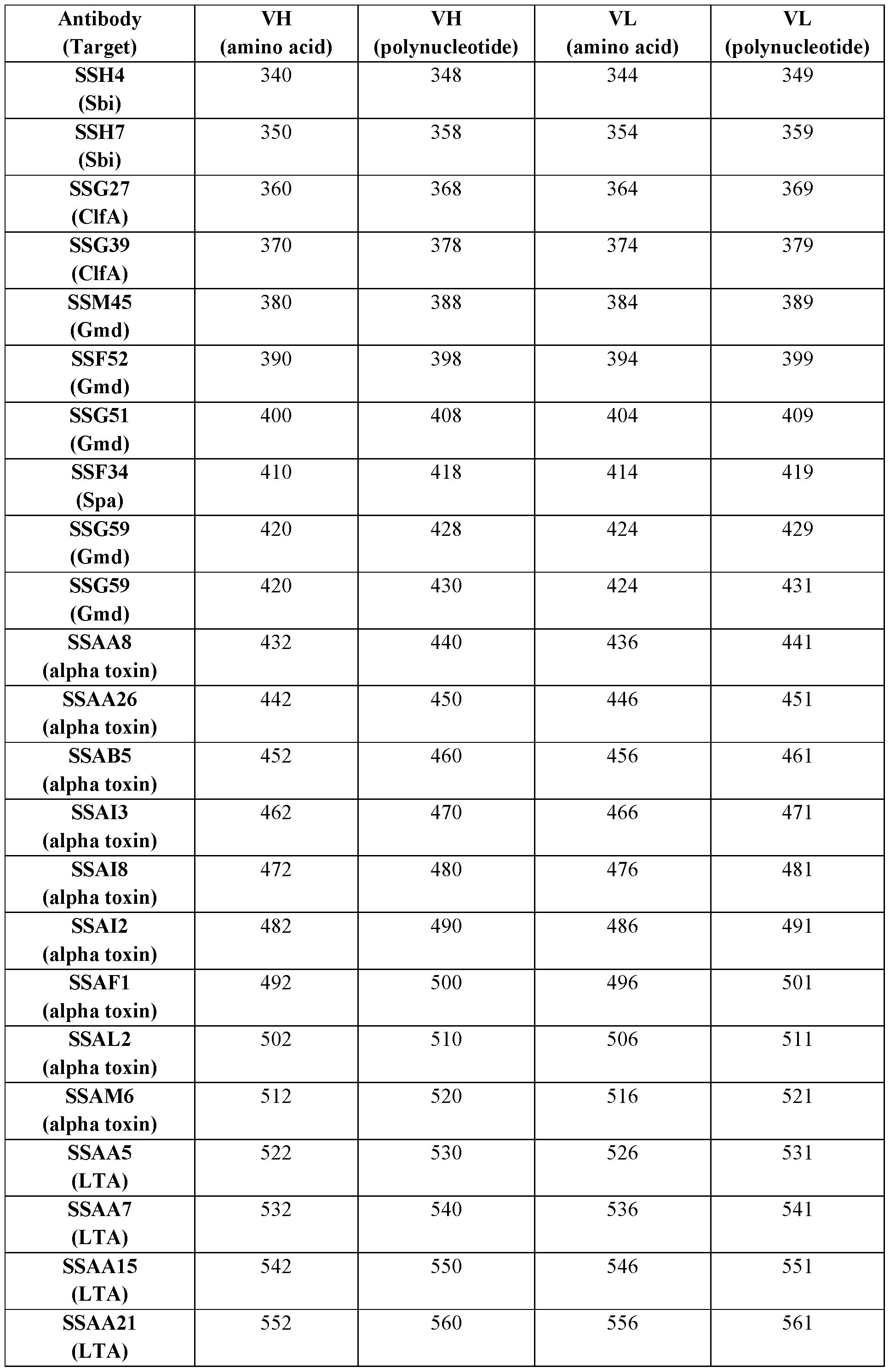
- an antibody or an antigen-binding fragment of the present disclosure comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein each CDR is independently selected from a corresponding CDR of an antigen-specific antibody as provided in Table 1, Table 2, Table 3, or Table 4. That is, all combinations of CDRs from antigen-specific antibodies provided in Table 1 and/or Table 2 and/or Table 3 and/or Table 4 are contemplated.
- an antibody or an antigen-binding fragment of the present disclosure comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3 of any one of the antibodies shown in Table 1 or 2.
- CDRs are in accordance with the IMGT numbering method.
- Table 2 summarizes IMGT CDR amino acid sequences (SEQ ID NOs.) of certain antibodies of the present disclosure.
- Table 3 summarizes variable domain amino acid sequences and examples of corresponding codon-optimized polynucleotide sequences (SEQ ID NOS.) of certain antibodies of the present disclosure.
- Table 3 summarizes variable domain amino acid sequences and examples of corresponding codon-optimized polynucleotide sequences (SEQ ID NOS.) of certain antibodies of the present disclosure.
- Table 3 Amino acid and certain polynucleotide sequences (SEQ ID NOs.) of variable domains of certain antibodies
- an antibody or antigen-binding fragment comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and/or a CDRL3, or functional variants thereof (and in certain embodiments, comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, or functional variants thereof) of the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii)
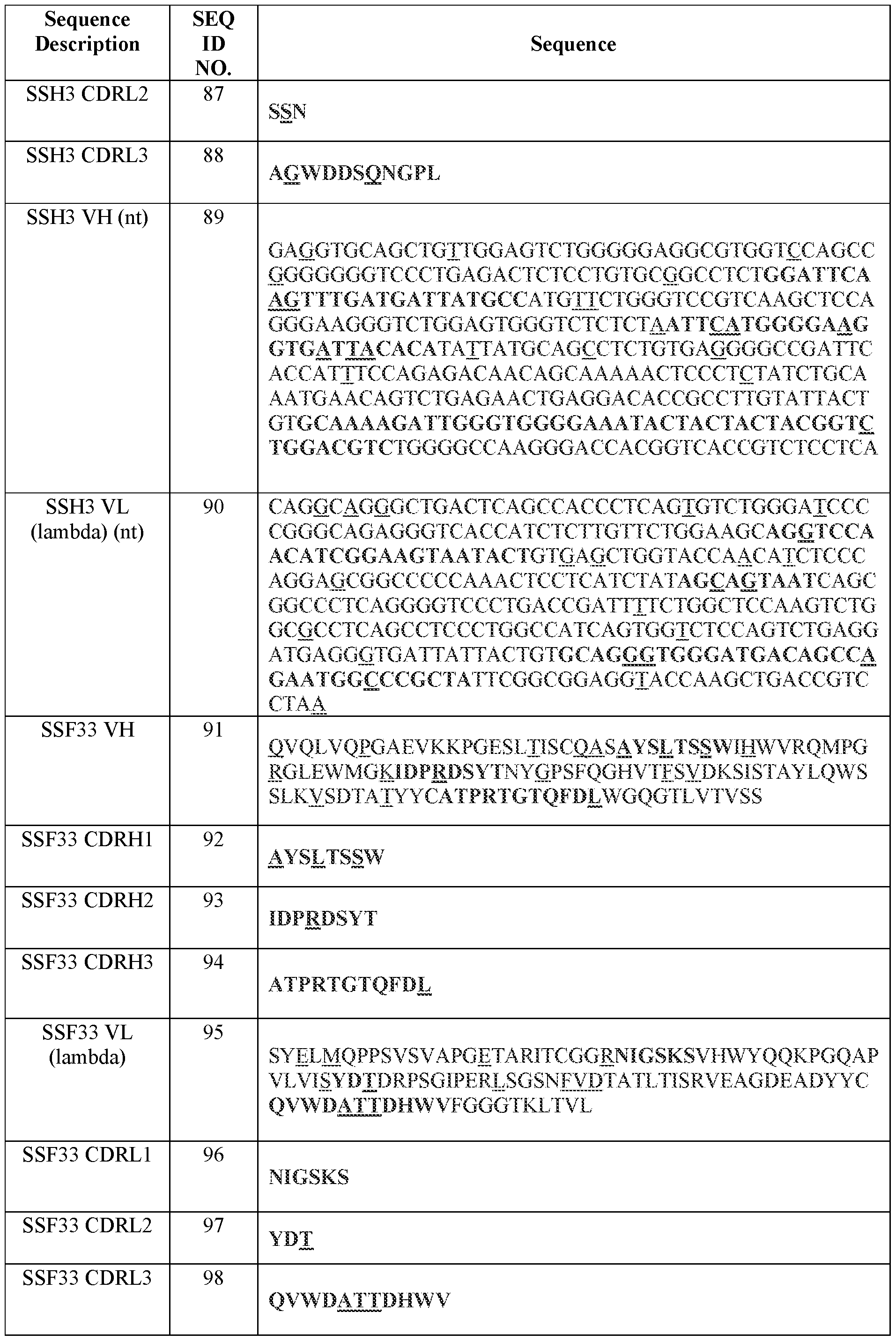
- an antibody or antigen-binding fragment comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, and optionally a VH and a VL, of one of the following antibodies as set forth in Table 2 and Table 3: SSG20; SSC35; SSE73; SSA12; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSE1; SSE158; SSF2; SSF11; SSF10; SSB18; SSB24; SSB54; SSB65; SSB67; SSH3; SSF33; SSL5; SSL17; SSL35; SSG50; SSG54; SSM47; SSG44; SSG48; SSM37; SSH4; SSH7; SSG27; SSG39; SSM45;
- the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the IMGT numbering system (optionally using the junction definitions for CDR3 sequences). In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Kabat numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Chothia numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Enhanced Chothia (also referred to as “Martin”) numbering system.
- Enhanced Chothia also referred to as “Martin”
- the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the AHo numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the North numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Contact numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the EU numbering system.
- the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the AbM numbering system (AbM antibody modelling software from Oxford Molecular). In some embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to a combination of any two or more of IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact. In some embodiments, the two or more numbering systems combined produce the CDR definition having the greatest length of the CDR according to any numbering system or combination of numbering systems.
- an antibody or antigen-binding fragment comprises:
- a CDRH1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.: 42, 32, 122, 22, 132, 142, 152, 162, 242, 172, 2, 12, 182, 52, 192, 212, 102, 222, 112, 232, 82, 92, 62, 72, 202, 281, 291, 301, 311, 321, 331, 341, 351, 361, 371, 381, 391, 401, 411, 421, 433, 443, 453, 463, 473, 483, 493, 503, 513, 523, 533, 543, 553, 563, 573, 583, 593, 603, 613, 623, 633, 643, 653, 663, 673, 683, 693, 703, 713, 723, 733, 743, and 753, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/
- a CDRH2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs. :43, 33, 123, 23, 133, 143, 153, 163, 243, 173, 3, 13, 183, 53, 193, 213, 103, 223, 113, 233, 83, 93, 63, 73, 203, 282, 292, 302, 312, 322, 332, 342,
- a CDRH3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.: 44, 34, 124, 24, 134, 144, 154, 164, 244, 174, 4,
- a CDRL1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:46, 36, 126, 26, 136, 146, 156, 166, 246, 176, 6,
- a CDRL2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:47, 37, 127, 27, 137, 147, 157, 167, 247, 177, 7,
- a CDRL3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs. :48, 38, 128, 28, 138, 148, 158, 168, 248, 178, 8,
- the antibody or antigen-binding fragment comprises a CDRH3 and a CDRL3, wherein the CDRH3 and the CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (i) 44 and 48, respectively; (ii) 34 and 38, respectively; (iii) 124 and 128, respectively; (iv) 24 and 28, respectively; (v) 134 and 138, respectively; (vi) 144 and 148, respectively; (vii) 154 and 158, respectively; (viii) 164 and 168, respectively; (ix) 244 and 248, respectively; (x) 174 and 178, respectively; (xi) 4 and 8, respectively; (xii) 14 and 18, respectively; (xiii) 184 and 188, respectively; (xiv) 54 and 58, respectively; (xv) 194 and 198, respectively; (xvi) 214 and 218, respectively; (xvii) 104 and 108, respectively; (xviii)
- the antibody or antigen-binding fragment comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (i) 42-44 and 46-48, respectively; (ii) 32-34 and 36- 38, respectively; (iii) 122-124 and 126-128, respectively; (iv) 22-24 and 26-28, respectively; (v) 132-134 and 136-138, respectively; (vi) 142-144 and 146-148, respectively; (vii) 152-154 and 156-158, respectively; (viii) 162-164 and 166-168, respectively; (ix) 242-244 and 246-248, respectively; (x) 172-174 and 176-178, respectively; (xi) 2-4 and 6-8, respectively; (xii)
- an antibody or antigen-binding fragment comprises an amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1- 6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40;
- an antibody or antigen-binding fragment comprises amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGHJ3, I
- an antibody or antigen-binding fragment comprises a framework amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting of, an amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1- 27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6;
- an antibody or antigen-binding fragment comprises framework amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the framework amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGHJ3,
- Framework amino acid sequences can be identified by a same numbering scheme that is used to identify CDRs; e.g., IMGT, Kabat, Chothia, Enhanced Chothia, AbM, AHo, North, Martin, Contact, or any combination thereof.
- an antibody or antigen-binding fragment comprises a VH framework region (VHFR)1, a VHFR2, a VHFR3, a VL framework region (VLFR)1, a VLFR2, a VLFR3, and/or a VLFR4 (or a variant of the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4 comprising one, two, three, four, or five substitutions, insertions, and/or deletions, or a variant having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4, respectively) of the VH and VL amino
- the framework region or regions are according to the IMGT numbering scheme. In some embodiments, the framework region or regions are according to the Kabat numbering scheme. In some embodiments, the framework region or regions are according to the Chothia numbering scheme. In some embodiments, the framework region or regions are according to the Enhanced Chothia numbering scheme. In some embodiments, the framework region or regions are according to the AbM numbering scheme. In some embodiments, the framework region or regions are according to the EU numbering scheme. In some embodiments, the framework region or regions are according to the North numbering scheme. In some embodiments, the framework region or regions are according to the Contact numbering scheme. In some embodiments, the framework region or regions are according to the AHo numbering scheme. In some embodiments, the framework region or regions are according to a combination of any two or more of the following numbering schemes: IMGT, Kabat Chothia, Enhanced Chothia, AbM, AHo, EU, North, Contact.
- the antibody or antigen-binding fragment comprises a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and
- DNA in the germline variable (V), joining (J), and diversity (D) gene loci may be rearranged and insertions and/or deletions of nucleotides in the coding sequence may occur. Somatic mutations may be encoded by the resultant sequence, and can be identified by reference to a corresponding known germline sequence.
- somatic mutations that are not critical to a desired property of the antibody e.g., binding to a herein-disclosed antigen
- that confer an undesirable property upon the antibody e.g., an increased risk of immunogenicity in a subject administered the antibody
- the antibody or antigenbinding fragment of the present disclosure comprises at least one more germline-encoded amino acid in a variable region as compared to a parent antibody or antigen-binding fragment, provided that the parent antibody or antigen binding fragment comprises one or more somatic mutations.
- Variable region and CDR amino acid sequences (SEQ ID NOs.) of certain antibodies of the present disclosure are provided in Tables 2-4 herein.
- CL refers to an “immunoglobulin light chain constant region” or a “light chain constant region,” z.e., a constant region from an antibody light chain.
- CH refers to an “immunoglobulin heavy chain constant region” or a “heavy chain constant region,” which is further divisible, depending on the antibody isotype, into CHI, CH2, and CH3 (IgA, IgD, IgG), or CHI, CH2, CH3, and CH4 domains (IgE, IgM).
- CHI immunoglobulin heavy chain constant region
- an antibody or antigen-binding fragment of the present disclosure comprises any one or more of CL, a CHI, a CH2, and a CH3. In any of the presently disclosed embodiments, an antibody or antigen-binding fragment of the present disclosure may comprise any one or more of CL, a CHI, a CH2, and a CH3.
- a CL comprises an amino acid sequence having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity (or similarity) to the amino acid sequence of a human IgG kappa constant domain (e.g., to SEQ ID NO.:278) or to a human IgG lambda constant domain (e.g., to SEQ ID NO.:279).
- a CH1-CH2-CH3 comprises an amino acid sequence having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity (or similarity) to the amino acid sequence of a human IgGl isotype, an engineered human IgGl isotype, a human IgG3 isotype, or an engineered human IgG3 isotype.
- a CH1-CH2-CH3 comprises an amino acid sequence having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity (or similarity) to the amino acid sequence of any one of SEQ ID NOs.:251-277 and 765. It will be understood that, for example, production in a mammalian cell line can remove one or more C-terminal lysine of an antibody heavy chain (see, e.g., Liu et al. mAbs 6(5): 1145-1154 (2014)).
- an antibody or antigen-binding fragment of the present disclosure can comprise a heavy chain, a CH1-CH3, a CH3, or an Fc polypeptide wherein a C-terminal lysine residue is present or is absent; in other words, encompassed are embodiments where the C-terminal residue of a heavy chain, a CH1-CH3, or an Fc polypeptide is not a lysine, and embodiments where a lysine is the C-terminal residue.
- a composition comprises a plurality of an antibody and/or an antigenbinding fragment of the present disclosure, wherein one or more antibody or antigen-binding fragment does not comprise a lysine residue at the C-terminal end of the heavy chain, CH1-CH3, or Fc polypeptide, and wherein one or more antibody or antigen-binding fragment comprises a lysine residue at the C-terminal end of the heavy chain, CH1-CH3, or Fc polypeptide.
- an antibody or antigen-binding fragment of the present disclosure can comprise a heavy chain, a CH1-CH3, a CH3, or an Fc polypeptide wherein a C-terminal glycine-lysine sequence e.g., the last two amino acids of SEQ ID NO.:251) is present or is absent.
- a “Fab” fragment antigen binding is the part of an antibody that binds to antigens and includes the variable region and CHI of the heavy chain linked to the light chain via an interchain disulfide bond. Each Fab fragment is monovalent with respect to antigen binding, i.e., it has a single antigen-binding site (or domain). Pepsin treatment of an antibody yields a single large F(ab’)2 fragment that roughly corresponds to two disulfide linked Fab fragments having divalent antigen-binding activity and is still capable of cross-linking antigen.
- Fab fragments differ from Fab fragments by having additional few residues at the carboxy terminus of the CHI domain including one or more cysteines from the antibody hinge region.
- Fab’-SH is the designation herein for Fab’ in which the cysteine residue(s) of the constant domains bear a free thiol group.
- F(ab’)2 antibody fragments originally were produced as pairs of Fab’ fragments that have hinge cysteines between them. Other chemical couplings of antibody fragments are also known.
- Fv is a small antibody fragment that contains a complete antigen-recognition and antigen-binding site (or domain). This fragment generally consists of a dimer of one heavy- and one light-chain variable region domain in tight, non-covalent association. However, even a single variable domain (or half of an Fv comprising only three CDRs specific for an antigen) has the ability to recognize and bind antigen, although typically at a lower affinity than the entire binding site (or domain).
- Single-chain Fv also abbreviated as “sFv” or “scFv”, are antibody fragments that comprise the VH and VL antibody domains connected into a single polypeptide chain.
- the scFv polypeptide comprises a polypeptide linker disposed between and linking the VH and VL domains that enables the scFv to retain or form the desired structure for antigen binding.
- a peptide linker can be incorporated into a fusion polypeptide using standard techniques well known in the art.
- the antibody or antigenbinding fragment comprises a scFv comprising a VH domain, a VL domain, and a peptide linker linking the VH domain to the VL domain.
- a scFv comprises a VH domain linked to a VL domain by a peptide linker, which can be in a VH-linker-VL orientation or in a VL-linker-VH orientation.
- Peptide linker sequences may be chosen, for example, based on: (1) their ability to adopt a flexible extended conformation; (2) their inability or lack of ability to adopt a secondary structure that could interact with functional epitopes on the first and second polypeptides and/or on a target molecule; and/or (3) the lack or relative lack of hydrophobic or charged residues that might react with the polypeptides and/or target molecule.
- linker design e.g., length
- linker design can include the conformation or range of conformations in which the VH and VL can form a functional antigen-binding site (or domain).
- peptide linker sequences contain, for example, Gly, Asn and Ser residues.
- linker sequence may also be included in a linker sequence.
- Other amino acid sequences which may be usefully employed as linker include those disclosed in Maratea et al., Gene 40:39 46 (1985); Murphy et al., Proc. Natl. Acad. Sci. USA 83:8258 8262 (1986); U.S. Pat. No. 4,935,233, and U.S. Pat. No. 4,751,180.
- linkers may include, for example, Glu-Gly-Lys-Ser-Ser-Gly-Ser-Gly-Ser-Glu-Ser-Lys-Val-Asp (Chaudhary et al., Proc. Natl. Acad. Sci.
- Any suitable linker may be used, and in general can be about 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 15 23, 24, 25, 26, 27, 28, 29, 30, 40, 50, 60, 70, 80, 90, 100 amino acids in length, or less than about 200 amino acids in length, and will preferably comprise a flexible structure (can provide flexibility and room for conformational movement between two regions, domains, motifs, fragments, or modules connected by the linker), and will preferably be biologically inert and/or have a low risk of immunogenicity in a human.
- ScFvs can be constructed using any combination of the VH and VL sequences or any combination of the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 sequences disclosed herein.
- linker sequences are not required; for example, when the first and second polypeptides have non-essential N-terminal amino acid regions that can be used to separate the functional domains and prevent steric interference.
- an antibody or antigen-binding fragment of the present disclosure is monospecific (e.g., binds to a single epitope) or is multispecific (e.g., binds to multiple epitopes and/or target molecules).
- Antibodies and antigen binding fragments may be constructed in various formats. Exemplary antibody formats disclosed in Spiess et al., Mol. Immunol.
- FIT-Ig e.g., PCT Publication No.
- the antibody or antigen-binding fragment comprises two or more of VH domains, two or more VL domains, or both (i.e., two or more VH domains and two or more VL domains).
- an antigen-binding fragment comprises the format (N-terminal to C-terminal direction) VH-linker-VL-linker-VH-linker-VL, wherein the two VH sequences can be the same or different and the two VL sequences can be the same or different.
- Such linked scFvs can include any combination of VH and VL domains arranged to bind to a given target, and in formats comprising two or more VH and/or two or more VL, one, two, or more different epitopes or antigens may be bound. It will be appreciated that formats incorporating multiple antigen-binding domains may include VH and/or VL sequences in any combination or orientation.
- the antigen-binding fragment can comprise the format VL-linker-VH-linker-VL-linker-VH, VH-linker-VL-linker-VL-linker-VH, or VL-linker-VH- linker-VH-linker-VL.
- Monospecific or multispecific antibodies or antigen-binding fragments of the present disclosure can comprise any combination of the VH and VL sequences and/or any combination of the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 sequences disclosed herein.
- a bispecific or multispecific antibody or antigen-binding fragment may, in some embodiments, comprise one, two, or more antigen-binding domains (e.g., a VH and a VL) of the instant disclosure.
- Two or more binding domains may be present that bind to the same or a different antigen epitope, and a bispecific or multispecific antibody or antigen-binding fragment as provided herein can, in some embodiments, comprise a further antigen-specific binding domain, and/or can comprise a binding domain that binds to a different antigen or pathogen altogether.
- the antibody or antigen-binding fragment can be multispecific; e.g., bispecific, trispecific, or the like.
- an antibody, or an antigen-binding fragment thereof which is a multi-specific antibody or antigen-binding fragment thereof, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a Sp
- each antigen-binding domain of a multispecific antibody or antigen-binding fragment is capable of targeting a different binding site of the same target selected from: an LTA, an alpha toxin, a gamma toxin, a SpA, a ClfA, a Gmd, a Sbi, and a leucocidin.
- a multispecific antibody or antigen-binding fragment comprises at least two antigen-binding sites, wherein at least one antigen-binding site is capable of targeting a first binding site of a target selected from: an LTA, an alpha toxin, a gamma toxin, a SpA, a ClfA, a Gmd, a Sbi, and a leucocidin; and wherein at least one antigen-binding site is capable of targeting a second binding site of the target.
- a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to an opsonizing target and a second antigen-binding domain of the multispecific antibody or antigen-binding fragment binds to a neutralizing target.
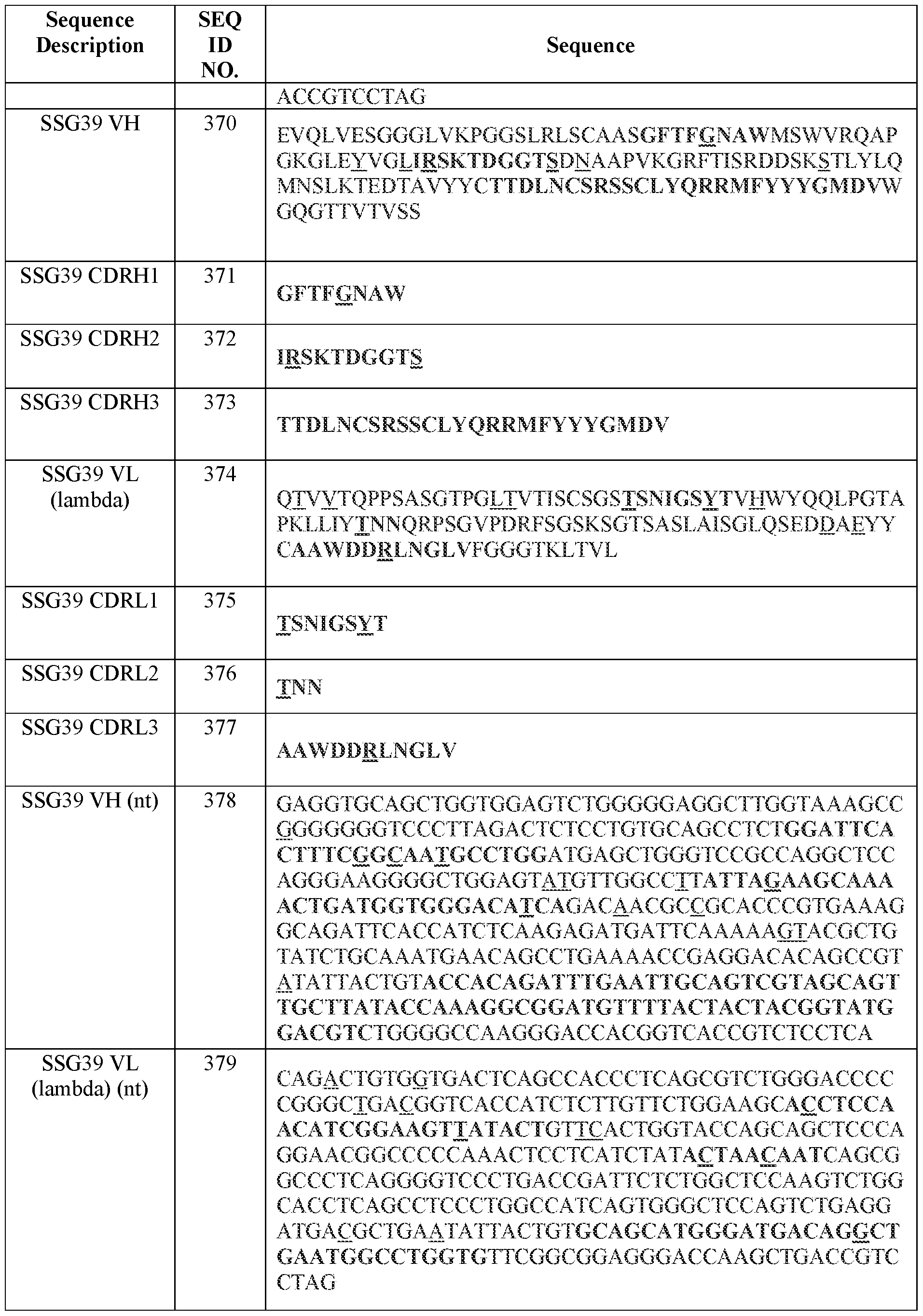
- a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to a target selected from LTA, ClfA, Protein A, and Gmd, and a second antigenbinding domain of the multispecific antibody or antigen-binding fragment binds to an alpha toxin.
- a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to an opsonizing target and a second antigen-binding domain of the multispecific antibody or antigen-binding fragment binds to an alpha toxin.
- a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to a target selected from LTA, ClfA, Protein A, and Gmd and a second antigen-binding domain of the multispecific antibody or antigen-binding fragment binds to a neutralizing target.
- fusion proteins that comprise an antibody or antigen-binding fragment of the present disclosure.
- a fusion protein comprises (i) an extracellular component comprising the antibody or antigen-binding fragment, (ii) a transmembrane component, and (iii) an intracellular component comprising one or more signaling domains (e.g., from CD3( ⁇ , CD28, 4-1BB, and/or TLR8).
- a fusion protein comprises a chimeric antigen receptor.
- a fusion protein comprises a chimeric engulfment receptor (see, e.g., Corey el al. , Molecular Therapy Methods & Clinical Development 28: 1-10 (2023); doi . org/ 10.1016/j . omtim .2022.11.004).
- the antibody or antigen-binding fragment comprises a Fc polypeptide, or a fragment thereof.
- the “Fc” fragment or Fc polypeptide comprises the carboxyterminal portions i.e., the CH2 and CH3 domains of IgG) of both antibody H chains held together by disulfides.
- An Fc may comprise a dimer comprised of two Fc polypeptides (i.e., two CH2-CH3 polypeptides).
- Antibody “effector functions” refer to those biological activities attributable to the Fc region (a native sequence Fc region or amino acid sequence variant Fc region) of an antibody, and vary with the antibody isotype.
- antibody effector functions include: Clq binding and complement dependent cytotoxicity; Fc receptor binding; antibody-dependent cell-mediated cytotoxicity (ADCC); phagocytosis; down regulation of cell surface receptors (e.g., B cell receptor); and B cell activation.
- modifications e.g., amino acid substitutions
- Fc domain in order to modify (e.g., improve, reduce, or ablate) one or more functionality of an Fc-containing polypeptide (e.g., an antibody of the present disclosure).
- Such functions include, for example, Fc receptor (FcR) binding, antibody half-life modulation (e.g., by binding to FcRn), ADCC function, protein A binding, protein G binding, and complement binding.
- Amino acid modifications that modify (e.g., improve, reduce, or ablate) Fc functionalities include, for example, the T250Q/M428L, M252Y/S254T/T256E, H433K/N434F, M428L/N434S, M428L/434A, E233P/L234V/L235A/G236 + A327G/A330S/P331S, E333A, S239D/A330L/I332E, P257VQ311, K326W/E333S, S239D/I332E/G236A, N297Q, K322A, S228P, L235E + E318A/K320A/K322A, L234A/L235A (also referred to herein as “LALA”), and L234A/L235A/P329G mutations, and other mutations described herein, certain of which mutations are summarized and annotated in
- the Clq protein complex can bind to at least two molecules of IgGl or one molecule of IgM when the immunoglobulin molecule(s) is attached to the antigenic target (Ward, E. S., and Ghetie, V., Ther. Immunol. 2 (1995) 77-94).
- Burton, D. R. described (Mol. Immunol. 22 (1985) 161-206) that the heavy chain region comprising amino acid residues 318 to 337 is involved in complement fixation.
- FcR binding can be mediated by the interaction of the Fc moiety (of an antibody) with Fc receptors (FcRs), which are specialized cell surface receptors on cells including hematopoietic cells.
- Fc receptors belong to the immunoglobulin superfamily, and shown to mediate both the removal of antibody-coated pathogens by phagocytosis of immune complexes, and the lysis of erythrocytes and various other cellular targets (e.g. tumor cells) coated with the corresponding antibody, via antibody dependent cell mediated cytotoxicity (ADCC; Van de Winkel, J. G., and Anderson, C. L., J. Leukoc. Biol. 49 (1991) 511-524).
- ADCC antibody dependent cell mediated cytotoxicity
- FcRs are defined by their specificity for immunoglobulin classes; Fc receptors for IgG antibodies are referred to as FcyR, for IgE as FcsR, for IgA as FcaR and so on and neonatal Fc receptors are referred to as FcRn.
- Fc receptor binding is described for example in Ravetch, J. V., and Kinet, J. P., Annu. Rev. Immunol. 9 (1991) 457-492; Capel, P. J., et al., Immunomethods 4 (1994) 25-34; de Haas, M., et al., J Lab. Clin. Med. 126 (1995) 330-341; and Gessner, J. E., et al., Ann. Hematol. 76 (1998) 231-248.
- FcyR Fc domain of native IgG antibodies
- FcyR In humans, three classes of FcyR have been characterized to-date, which are: (i) FcyRI (CD64), which binds monomeric IgG with high affinity and is expressed on macrophages, monocytes, neutrophils and eosinophils; (ii) FcyRII (CD32), which binds complexed IgG with medium to low affinity, is widely expressed, in particular on leukocytes, is believed to be a central player in antibody-mediated immunity, and which can be divided into FcyRIIA, FcyRIIB and FcyRIIC, which perform different functions in the immune system, but bind with similar low affinity to the IgG-Fc, and the ectodomains of these receptors are highly homologuous; and (iii) FcyRIII (CD 16), which binds IgG with medium to low affinity and has been found in two forms: FcyRIIIA, which has been found on NK cells, macrophages,
- FcyRIIA is found on many cells involved in killing (e.g. macrophages, monocytes, neutrophils) and seems able to activate the killing process.
- FcyRIIB seems to play a role in inhibitory processes and is found on B-cells, macrophages and on mast cells and eosinophils. Importantly, it has been shown that 75% of all FcyRIIB is found in the liver (Ganesan, L. P. et al., 2012: “FcyRIIb on liver sinusoidal endothelium clears small immune complexes,” loumal of Immunology 189: 4981-4988).
- FcyRIIB is abundantly expressed on Liver Sinusoidal Endothelium, called LSEC, and in Kupffer cells in the liver and LSEC are the major site of small immune complexes clearance (Ganesan, L. P. et al., 2012: FcyRIIb on liver sinusoidal endothelium clears small immune complexes. lournal of Immunology 189: 4981-4988).
- the antibodies disclosed herein and the antigen-binding fragments thereof comprise an Fc polypeptide or fragment thereof for binding to FcyRIIb, in particular an Fc region, such as, for example IgG-type antibodies.
- FcyRIIb an Fc region
- it is possible to engineer the Fc moiety to enhance FcyRIIB binding by introducing the mutations S267E and L328F as described by Chu, S. Y. et al., 2008: Inhibition of B cell receptor-mediated activation of primary human B cells by co-engagement of CD19 and FcgammaRIIb with Fc-engineered antibodies.
- Molecular Immunology 45, 3926-3933 are examples of the FcyRIIb.
- the antibodies of the present disclosure comprise an engineered Fc moiety with the mutations S267E and L328F, in particular as described by Chu, S. Y. et al., 2008: Inhibition of b cell receptor-mediated activation of primary human B cells by co-engagement of CD19 and FcgammaRIIb with Fc-engineered antibodies.
- FcyRIIB may function to suppress further immunoglobulin production and isotype switching to, for example, the IgE class.
- FcyRIIB On macrophages, FcyRIIB is thought to inhibit phagocytosis as mediated through FcyRIIA.
- the B form On eosinophils and mast cells, the B form may help to suppress activation of these cells through IgE binding to its separate receptor.
- modification in native IgG of at least one of E233-G236, P238, D265, N297, A327 and P329 reduces binding to FcyRI.
- IgG2 residues at positions 233-236, substituted into corresponding positions IgGl and IgG4, reduces binding of IgGl and IgG4 to FcyRI by 10 3 -fold and eliminated the human monocyte response to antibody-sensitized red blood cells (Armour, K. L., et al. Eur. J. Immunol. 29 (1999) 2613-2624).
- FcyRIIA reduced binding for FcyRIIA is found, e.g., for IgG mutation of at least one of E233-G236, P238, D265, N297, A327, P329, D270, Q295, A327, R292 and K414.
- FcyRIII binding reduced binding to FcyRIIIA is found, e.g., for mutation of at least one of E233-G236, P238, D265, N297, A327, P329, D270, Q295, A327, S239, E269, E293, Y296, V303, A327, K338 and D376. Mapping of the binding sites on human IgGl for Fc receptors, the above-mentioned mutation sites, and methods for measuring binding to FcyRI and FcyRIIA, are described in Shields, R. L., et al., J. Biol. Chem. 276 (2001) 6591-6604.
- FcyRIIIA Two allelic forms of human FcyRIIIA are the “Fl 58” variant, which binds to IgGl Fc with lower affinity, and the “V158” variant, which binds to IgGl Fc with higher affinity. See, e.g., Bruhns et al., Blood 11331X6-3125 (2009).
- two regions of native IgG Fc appear to be involved in interactions between FcyRIIs and IgGs, namely (i) the lower hinge site of IgG Fc, in particular amino acid residues L, L, G, G (234 - 237, EU numbering), and (ii) the adjacent region of the CH2 domain of IgG Fc, in particular a loop and strands in the upper CH2 domain adjacent to the lower hinge region, e.g. in a region of P331 (Wines, B.D., et al., I. Immunol. 2000; 164: 5313 - 5318).
- FcyRI appears to bind to the same site on IgG Fc
- FcRn and Protein A bind to a different site on IgG Fc, which appears to be at the CH2-CH3 interface
- mutations that increase binding affinity of an Fc polypeptide or fragment thereof of the present disclosure to a (i.e., one or more) Fey receptor (e.g., as compared to a reference Fc polypeptide or fragment thereof or containing the same that does not comprise the mutation(s)). See, e.g., Delillo and Ravetch, Cell 161(5): 1035-1045 (2015) and Ahmed et al., J. Struc. Biol. 194(1):78 (2016), the Fc mutations and techniques of which are incorporated herein by reference.
- an antibody or antigen-binding fragment can comprise a Fc polypeptide or fragment thereof comprising a mutation selected from G236A; S239D; A330L; and I332E; or a combination comprising any two or more of the same; e.g., S239D/I332E; S239D/A330L/I332E; G236A/S239D/I332E; G236A/A330L/I332E (also referred to herein as “GAALIE”); or G236A/S239D/A330L/I332E.
- the Fc polypeptide or fragment thereof does not comprise S239D.
- the Fc polypeptide or fragment thereof comprises S at position 239 (EU numbering).
- the Fc polypeptide or fragment thereof may comprise or consist of at least a portion of an Fc polypeptide or fragment thereof that is involved in FcRn binding.
- the Fc polypeptide or fragment thereof comprises one or more amino acid modifications that improve binding affinity for (e.g., enhance binding to) FcRn (e.g., at a pH of about 6.0) and, in some embodiments, thereby extend in vivo half-life of a molecule comprising the Fc polypeptide or fragment thereof (e.g., as compared to a reference Fc polypeptide or fragment thereof or antibody that is otherwise the same but does not comprise the modification(s)).
- the Fc polypeptide or fragment thereof comprises or is derived from a IgG Fc and a half-life-extending mutation comprises any one or more of: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q311I; D376V; T307A; E380A (EU numbering).
- a half-life-extending mutation comprises M428L/N434S (also referred to herein as “MLNS”, “LS”, “_LS”, and “-LS”).
- a half-life-extending mutation comprises M252Y/S254T/T256E.
- a half-life-extending mutation comprises T250Q/M428L. In certain embodiments, a half-life-extending mutation comprises P257EQ311I. In certain embodiments, a half-life-extending mutation comprises P257I/N434H. In certain embodiments, a half-life- extending mutation comprises D376V/N434H. In certain embodiments, a half-life-extending mutation comprises T307A/E380A/N434A. In certain embodiments, a half-life-extending mutation comprises M428L/N434A.
- an antibody or antigen-binding fragment includes a Fc moiety that comprises the substitution mutations M428L/N434S. In some embodiments, an antibody or antigen-binding fragment includes a Fc moiety that comprises the substitution mutations M428L/N434A. In some embodiments, an antibody or antigen-binding fragment includes a Fc polypeptide or fragment thereof that comprises the substitution mutations G236A/A330L/I332E.
- an antibody or antigen-binding fragment includes a (e.g., IgG) Fc moiety that comprises a G236A mutation, an A330L mutation, and a I332E mutation (GAALIE), and does not comprise a S239D mutation (e.g., comprises a native S at position 239).
- an antibody or antigen-binding fragment includes an Fc polypeptide or fragment thereof that comprises the substitution mutations: M428L/N434S and G236A/A330L/I332E, and optionally does not comprise S239D (e.g., comprises S at 239).
- an antibody or antigen-binding fragment includes a Fc polypeptide or fragment thereof that comprises the substitution mutations: M428L/N434S and G236 A/S239D/A330L/I332E.
- an antibody or antigen-binding fragment comprises an IgGl Fc polypeptide (or an engineered variant thereof) comprising an H435R mutation, a Y436F mutation, or an H435R mutation and a Y436F mutation. See, e.g., Jendeberg et al. J Immunol Methods 207(l):25-34 (1997); doi: 10.1016/s0022-1759(96)00215-3.
- an antibody or antigen-binding fragment includes an (e.g., IgGl) Fc polypeptide that comprises the following mutations: (i) M428L, N434S, H435R; (ii) M428L, N434S, Y436F; or (iii) (ii) M428L, N434S, H435R, and Y436F.
- an antibody or antigen-binding fragment may be expressed as (e.g., recombinant) human IgG3 (IgG3 contains an arginine “R” residue at EU position 435).
- an antibody has a native IgGl isotype and is expressed recombinantly as an IgG3 isotype.
- the antibody or antigen-binding fragment comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or the antibody or antigen-binding fragment is partially or fully aglycosylated and/or is partially or fully afucosylated.
- Host cell lines and methods of making partially or fully aglycosylated or partially or fully afucosylated antibodies and antigen-binding fragments are known (see, e.g., PCT Publication No. WO 2016/181357; Suzuki et al. Clin. Cancer Res. 73(6): 1875-82 (2007); Huang et al.
- an antibody or antigen-binding fragment comprises a heavy chain that comprises one or more mutations in the hinge, CH2, and/or CH3 (or in the Fc), wherein the antibody or antigen-binding fragment has one or more improved characteristics over, for example, the antibody or antigenbinding fragment comprising reference wild-type Fc polypeptide and/or comprising a known variant Fc polypeptide).
- Fc variants possess, for example: increased binding to one or more human FcyRA (e.g., a FcyRIIA and/or a FcyRIIIA; decreased/reduced binding to a human FcyRIIB; increased binding to one or more human FcyRA as compared to binding to a human FcyRIIB; increased thermostability as compared to known Fc polypeptides; increased binding to human Clq; increased human FcyRIIIA signaling in a host cell expressing the FcyRIIIA, increased human FcyRIIIA signaling in a host cell expressing the FcyRIIA, decreased human FcyRIIB signaling in a host cell expressing the FcyRIIB, a relative increase in binding to FcyRA as compared to FcyRIIB, improved qualities for production as compared to known Fc polypeptides; and combinations of such features.
- human FcyRA e.g., a FcyRIIA
- antibodies comprising a variant Fc polypeptide of the present disclosure provide surprising advantages, such as any one or more of the following: increased binding affinity (e.g. as determined by surface plasmon resonance, e.g. using a Biacore instrument and/or as determined by a electrochemiluminescence assay, such as a meso scale discovery (MSD) assay) for and/or inducing increased signaling (e.g.
- increased binding affinity e.g. as determined by surface plasmon resonance, e.g. using a Biacore instrument and/or as determined by a electrochemiluminescence assay, such as a meso scale discovery (MSD) assay
- MSD meso scale discovery
- an Fc variant antibody (2) antigen-expressing target cells and (3) reporter cells expressing one or more human FcyRA, optionally driving expression of a reporter gene such as, for example, GFP or luciferase) by one or more human FcyRA, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; decreased binding affinity for and/or inducing decreased signaling of human FcyRIIB, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; a unique and optionally improved binding profile across human FcyRIIA-H, human FcyRIIA-R, human FcyRIIB, human FcyRIIIA-F, and human FcyRIIIA-V, wherein improved binding comprises an overall increase in binding to and/or activation of FcyRA signaling relative to binding to and/or activation of inhibitory FcyR signaling, as
- moDCs and/or increasing expression of CD83 by moDCs in a sample when provided in combination with the antigen, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state, when provided in combination with the antigen; increasing production of one or more cytokine (optionally selected from the group consisting of IL-ip, IFN-y, IL-6, and TNF-a) by moDCs in a sample when provided in combination with the antigen, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state, when provided in combination with the antigen; and/or increasing the ability of moDCs to stimulate antigenspecific CD4+ T cells when provided to the moDCs in combination with the antigen, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state, when provided to the moDCs in combination with the antigen, wherein, optionally,
- an engineered Fc or Fc fragment of the present disclosure (or a polypeptide comprising the same) comprises two or more substitution mutations as compared to a reference wild-type Fc or Fc fragment, and the combined effect of the two or more substitutions is different than, and is optionally greater than, would be expected based on the effects of the individual component substitution mutations and/or based on the effects of a subset of the two or more substitution mutations.
- combination mutations comprise a non-additive or synergistic effect with reference to the individual component mutations and/or to a subset thereof.
- antibodies or antigen-binding fragments comprising an Fc variant possess characteristics such as effector functions, ability to bind human Clq, ability to induce FcyRA-mediated cell signaling, ability to bind to human FcRn, ability to promote ADCP, ability to promote ADCC, ability to promote activation of CD4+ T cells, and the like.
- polypeptides include those that comprise a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises one or more modifications as compared to the IgG Fc polypeptide or fragment thereof.
- a “reference” polypeptide or antibody e.g., reference IgG Fc polypeptide or fragment thereof, reference antibody, reference CH2 polypeptide, reference IgG hinge-CH2, reference IgG hinge-Fc polypeptide, reference CH3 polypeptide
- is preferably identical to the recited molecule e.g., variant of an Fc polypeptide or fragment thereof; polypeptide comprising such a variant; antibody comprising a variant of an Fc polypeptide except for the recited difference or differences.
- a reference Fc polypeptide includes an IgGl Fc polypeptide that is otherwise identical to the variant except that a native glycine (G) amino acid is found at EU position 236.
- a reference Fc polypeptide fragment is preferably of an identical length to the variant and preferably differs from the variant only by the recited features (e.g., amino acid mutation or mutations present in the variant).
- a reference Fc polypeptide, Fc polypeptide fragment, or antibody comprises a wild-type amino acid sequence (e.g., wild-type human IgGl). Excepting the recited differences present in the variant, a reference Fc polypeptide, Fc polypeptide fragment, or antibody will be of the same isotype, and, preferably, of the same allotype, as the variant. In the case of a reference antibody, the Fabs or other antigen-binding domains will preferably be identical to those present in the specified antibody comprising a variant Fc polypeptide or fragment thereof.
- variants of IgG Fc polypeptides or fragments thereof include one or more amino acid substitution as compared to a reference (e.g.
- the position of an amino acid in a variant IgG Fc polypeptide or fragment may be described by referencing the “EU position”; it will be understood that “the EU position” follows the EU numbering system as set forth in Kabat.
- EU position follows the EU numbering system as set forth in Kabat.
- two or more amino acid substitutions present in a variant can be expressed in a variety of ways, for example, as G236A_Y300L, or as G236A/Y300L.
- a mutation or combination mutation may be referenced using a short form including the original amino acid(s) and the amino acid(s) resulting from the substitution(s).
- G236A may be described as “GA” or “236A”; G236A_Y300L may be described as “GAYL”; G236A_L328V_Q295E may be described as “GALVQE”; G236A_R292P_Y300L may be described as “GARPYL”, G236A_R292P_I377N may be described as “GARPIN”, or the like.
- a variant of an Fc polypeptide or fragment thereof can be derived from or comprise a human Fc polypeptide or fragment thereof, and/or can be derived from or comprise a human IgGl, a human IgG2, a human IgG3, or a human IgG4 isotype.
- the expression “derived from” means that the variant is the same as the referenced polypeptide or isotype, with the exception of the specified modification(s) (e.g., amino acid substitution(s)).
- a variant Fc polypeptide which comprises a wild-type human IgGl Fc amino acid sequence with the exception of the amino acid substitution mutations G236A L328V Q295E (and, optionally, other amino acid substitutions) can be said to be “derived from” wild-type human IgGl Fc.
- a polypeptide, CH2, Fc, Fc fragment, or antibody may comprise human Ig sequence, such as human IgGl sequence.
- the polypeptide, CH2, Fc, Fc fragment, or antibody can comprise a native or wild-type human Ig sequence with the exception of the described mutation(s), or can comprise a human Ig (e.g. IgG) sequence that contains one or more additional mutations.
- An antibody or antigen-binding fragment may be of any allotype or combination of allotypes.
- Allotype refers to the allelic variation found among the IgG subclasses.
- an allotype may comprise Glml (or Glm(a)), Glm2 (or Glm(x)), Glm3 (or Glm(f)), Glml7 (or Gm(z))m), Glm27, and/or Glm28 (Glm27 and Glm28 have been described as “alloallotypes”).
- the Glm3 and Glml7 allotypes are located at the same position in the CHI domain (position 214 according to EU numbering).
- Glm3 comprises R214 (EU)
- Glml7 comprises K214 (EU).
- the Glml allotype is located in the CH3 domain (at positions 356 and 358 (EU)) and refers to the replacements E356D and M358L.
- the Glm2 allotype refers to a replacement of the alanine in position 431 (EU) by a glycine.
- Glm allotypes, alloallotypes, and features thereof are known in the art and described at, for example, www.imgt.org/IMGTrepertoire/Proteins/allotypes/human/IGH/IGHC/Glm_allotypes.html and Lefranc, M.-P. and Lefranc, G. Human Gm, Km and Am allotypes and their molecular characterization: a remarkable demonstration of polymorphism In: B. Tait, F. Christiansen (Eds.), Immunogenetics, chap. 34, Humana Press, Springer, New York, USA. Methods Mol. Biol. 2012; 882, 635-680. PMID: 22665258, LIGM: 406, the contents and allotypes and allotype information of which are incorporated herein by reference.
- the Glml allotype may be combined, for example, with the Glm3, Glm 17, Glm27, Glm2, and/or Glm28 allotype.
- an allotype is Glm3 with no Glml (Glm3,-1).
- an allotype is Glml7,l allotype.
- an allotype is Glm3,l.
- an allotype is Glml7 with no Glml (Glml7,-1).
- these allotypes may be combined (or not combined) with the Glm2, Glm27 or Glm28 allotype.
- an allotype may be Glml7,l,2.
- an antibody or antigen-binding fragment of the present disclosure comprises a Glm3 allotype or a Glm3,l allotype. In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a Glm3 allotype and comprises M428L and N434S or M428L and N434A mutations or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- an antibody or antigen-binding fragment of the present disclosure comprises a Glm3,l allotype and comprises M428L and N434S or M428L and N434A mutations or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- an antibody or antigen-binding fragment of the present disclosure comprises a Glml7, 1 allotype.
- an antibody or antigen-binding fragment of the present disclosure comprises a Glml7, 1 allotype and comprises M428L and N434S or M428L and N434A mutations or any other mutation(s) that enhance binding to a human FcRn, as described further herein.
- the antibody or antigen-binding fragment comprises an IgG3 isotype. In some embodiments, the antibody or antigen-binding fragment comprises an IgG3m(b*) allotype, an IgG3m(b**) allotype, an IgG3m(c3*) allotype, an IgG3m(c3c5*) allotype, an IgG3m(g*) allotype, an IgG3m(s*) allotype, an IgG3m(st*) allotype, an IgG3 GM5 allotype, an IgG3 G4m5 allotype, or any combination thereof.
- an antibody or antigen-binding fragment comprises a CH3 sequence engineered to reduce potential immunogenicity of the antibody or antigen-binding fragment by replacing specific amino acids of one allotype with those of another allotype (these may be referred to as isoallotype mutations), as described in more detail in Stickler et al. (Genes Immun. 2011 Apr; 12(3): 213-221), which is herein incorporated by reference in its entirety, including, for example, the isoallotype mutations disclosed therein.
- specific amino acids of the Glml allotype are replaced.
- isoallotype mutations D356E and L358M are made in the CH3 sequence.
- a variant of an Fc polypeptide comprises only the specified or recited amino acid mutations (e.g. substitutions), and does not comprise any further amino acid substitutions or mutations; e.g., relative to the reference polypeptide (e.g., a wild-type Fc polypeptide or fragment thereof).
- a variant Fc polypeptide comprising the amino acid substitutions G236A Y300L does not comprise any other amino acid substitutions; i.e., comprises an amino acid sequence that is wild-type except for G236A and Y300L.
- a variant of an Fc polypeptide may comprise one or more additional amino acid mutations (e.g. substitutions), which can be specified (e.g., M428L_N434S; M428L_N434A).
- additional amino acid mutations e.g. substitutions
- a further amino acid mutation or mutations is physically remote to the recited amino acid positions in tertiary structure, and/or is of such nature (e.g.
- variant of an Fc polypeptide comprises the mutations M428L and N434S or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, including those described herein.
- an antibody or antigen-binding fragment (described further herein) is provided that comprises, in a(n e.g.
- the antibody or antigen-binding fragment is afucosylated.
- the antibody or antigen-binding fragment further comprises one or more mutation that enhances binding to a human FcRn, such as M428L and N434S mutations or M428L and N434A mutations (EU numbering) or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- the IgGl heavy chain comprises a CH1-CH3 or a CH2-CH3 or a hinge-CH2-CH3, wherein the CH1-CH3 or CH2-CH3 or hinge-CH2-CH3 has at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity (or similarity) to a wild-type human IgGl CH1-CH3 or CH2-CH3 or hinge-CH2-CH3, respectively.
- an antibody or antigen-binding fragment of the present disclosure comprises an Fc variant comprising an amino acid sequence having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity (or similarity) to the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765.
- the antibody or antigen-binding fragment comprises the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765, or a variant thereof, e.g. that further comprises one or more mutation that enhances binding to a human FcRn, such as M428L and N434S mutations or M428L and N434A mutations (EU numbering) or any other mutation(s) that enhance binding to a human FcRn, including those described herein.
- the antibody or antigen-binding fragment comprises an amino acid sequence that differs from the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765 only by one or more IgGl allotype-specific mutations and/or by the presence of M428L and N434S mutations or M428L and N434A mutations or other mutation(s) that enhance binding to a human FcRn.
- an antibody or antigen-binding fragment comprises an Fc polypeptide comprising one or more of the following mutations to, e.g., in a human IgGl background, can promote binding to Cl : K236WZE333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L.
- an Fc polypeptide can comprise any mutation or mutations described in Table A to increase binding to Cl (or Clq).
- an antibody or antigen-binding fragment comprises an Fc polypeptide comprising E345R/E430G/S440Y mutations to, e.g., in a human IgGl background, can promote oligomerization.
- An antibody or antigen-binding fragment of the present disclosure can be fucosylated (e.g., comprising one or more fucosyl moiety, and typically comprising a native (wild-type) fucosylation pattern or a fucosylation pattern that includes one or more additional, or fewer, fucosyl moieties as compared to native), or can be afucosylated.
- native IgGl antibodies carry a glycan site at N297, and this is typically the only site where a core fucose moiety may be found in the antibody, though some glycan sites may arise through mutation (e.g. in the variable domains) during antibody development.
- Fucosylation of an antibody or antigenbinding fragment can be affected by introducing amino acid mutations to introduce or disrupt a fucosylation site (e.g. a mutation at N297, such as N297Q or N297A, to disrupt formation of a glycan that can include a core fucose moiety), though typically it is preferred to maintain N297 and the glycan thereof, such as by expressing the antibody or antigen-binding fragment in a host cell which has been genetically engineered to lack the ability (or have an inhibited or compromised ability) to fucosylate the antibody or antigen-binding fragment; by expressing the antibody or antigen-binding fragment under conditions in which a host cell is impaired in its ability to fucosylate the polypeptide (e.g, in the presence of 2-fluoro-L-fucose (2FF)), or the like.
- a fucosylation site e.g. a mutation at N297, such as N297Q or N297A, to disrupt formation of a
- An afucosylated antibody or antigen-binding fragment can comprise no fucose moieties, or substantially no fucose moieties, and/or can be expressed by a host cell that is genetically engineered to lack the ability (or have an inhibited or compromised ability) to fucosylate the antibody or antigen-binding fragment and/or can be expressed under conditions in which a host cell is impaired in its ability to fucosylate the antibody or antigen-binding fragment (e.g, in the presence of 2-fluoro-L-fucose (2FF)).
- an antibody or antigen-binding fragment does not comprise a core fucose moiety at Asn297.
- afucosylated antibodies or antigen-binding fragments have increased binding to FcyRIIIA.
- addition of 2FF to a culture media comprising host cells expressing an antibody results in about 85% or more of the antibodies or antigen-binding fragments not carrying a fucose moiety. Accordingly, a plurality of antibodies or antigen-binding fragments may be described as “afucosylated” when the plurality was produced in the presence of 2FF or like reagent.
- a plurality of antibodies or antigen-binding fragments may be described as, for example, afucosylated, meaning that about 85% or more of the single antibody or antigen-binding fragment molecules of the plurality do not comprise a fucose moiety.
- an afucosylated antibody or antigen-binding fragment or a population or a plurality thereof comprises an asparagine (N) at EU position 297. Fucosylation or lack thereof can be assessed using, for example, mass spectrometry (e.g. Electrospray mass spectrometry (ESI-MS)).
- compositions are provided that comprise a plurality of any one or more of the presently disclosed antibodies or antigen-binding fragments, wherein the composition comprises afucosylated antibodies or antigen-binding fragments.
- IgG Fc polypeptides or fragments thereof possess one or more function that is distinct from (e.g. improved as compared to) the corresponding function of a reference Fc polypeptide that comprises the following mutation or mutations: G236A; G236S; G236A_A330L_I332E; G236A_A330L_I332E_M428L_N434S; A330L I332E; or G236A_S239D_A330L_I332E.
- a presently disclosed variant of an IgG Fc polypeptide or fragment thereof possesses one or more of the following properties, as compared to a reference Fc polypeptide that comprises the following mutation or mutations: G236A; G236S; G236A_A330L_I332E; G236A_A330L_I332E_M428L_N434S; A330LJ332E; or G236A_S239D_A330L_I332E: increased binding (e.g. affinity) to and/or signaling via a human FcyRIIa H131; increased binding (e.g.
- affinity to and/or signaling via a human FcyRIIa R131
- decreased binding to (e.g. affinity) and/or signaling via human FcyRIIb an increased ratio of binding to (e.g. affinity) and/or signaling via a human FcyRIIa (H131, R131, or both) versus the ratio of binding to or signaling via (respectively) a human FcyRIIb
- increased binding e.g.
- binding of a variant Fc polypeptide or fragment may be described as increased (or “greater than”, or the like) or decreased (or “reduced” or “less than”, or the like) as compared to the binding of a comparator (e.g., to a reference wild-type IgGl Fc, or to a reference IgGl Fc that is wild-type except for M428L and N434S mutations or except for M428L and N434A mutations or to a variant IgGl Fc comprising G236A_A330L_I332E mutations) to a same binding partner.
- a comparator e.g., to a reference wild-type IgGl Fc, or to a reference IgGl Fc that is wild-type except for M428L and N434S mutations or except for M428L and N434A mutations or to a variant IgGl Fc comprising G236A_A330L_I332E mutations
- Binding interactions between a variant Fc polypeptide or fragment (or an antibody or polypeptide comprising the same) and a binding partner can preferably be determined using an electrochemiluminescence assay, more preferably using the Meso Scale Discovery (“MSD”; mesoscale.com) platform.
- MSD binding assay is similar to ELISA though MSD uses electrochemiluminescence, as opposed to colorimetry, as a detection technique.
- Other techniques for measuring binding interactions are known and include, for example, ELISA, surface plasmon resonance (SPR), biolayer interferometry (BLI), and the like.
- binding includes affinity, avidity, or both.
- Affinity refers to the strength of a bond between a binding molecule and its binding partner.
- binding can include affinity and/or avidity.
- avidity refers to the total binding strength of a molecule to a binding partner, and reflects binding affinity, valency of binding sites (or domains) (e.g., whether an Fc polypeptide comprises one, two, or more binding sites (or domains)), and, for example, whether another agent is present that can affect the binding (e.g., a non-competitive inhibitor of the Fc polypeptide).
- a binding interaction between a variant molecule of the present disclosure and a binding partner can be expressed in terms of fold-change relative to the binding interaction between a reference molecule and the binding partner.
- binding of a presently disclosed antibody comprising a variant Fc to a human FcyRIIa may be stronger than the binding of the antibody comprising a wild-type Fc to the human FcyRIIa, and the relative increased strength of the variant can be expressed in terms of fold-change e.g., linear scale of area-under-the-curve) relative to the reference molecule binding using the same assay.
- a variant Fc polypeptide or fragment may bind to a FcyRIIa with a 2-fold, 3-fold, 4-fold, or 5-fold greater binding strength than a reference Fc polypeptide or fragment binds to the FcyRIIa.
- a variant Fc polypeptide or fragment thereof may bind less strongly to a FcyRIIb as compared to a reference Fc or fragment thereof; e.g., may have a 0.9-fold binding, 0.8-fold binding, 0.7-fold binding, 0.6-fold binding, or the like, as compared to the reference Fc polypeptide or fragment thereof.
- the expression “2-fold greater binding as compared to the binding of a reference” means a 2-fold increase in binding as compared to the reference.
- binding of a variant Fc molecule of the present disclosure to two different partner molecules can be described in terms of a ratio, and this ratio can be compared to a like ratio obtained using a reference molecule with the same assay.
- a variant Fc polypeptide may bind to a human FcyRIIa H131 five times more strongly than it binds to a human FcyRIIb, while a reference wild-type Fc polypeptide binds to FcyRIIa H131 as strongly as it binds to a human FcyRIIb.
- the variant Fc polypeptide can be said to have a 5: 1 (binding FcyRIIIa H131 :binding FcyRIIb) binding ratio, which can be compared to the 1 : 1 (binding FcyRIIIa H131 :binding FcyRIIb) binding ratio of the reference wild-type Fc polypeptide.
- Variant Fc molecules of the present disclosure may also be described in terms of ability to induce signaling in a host cell, wherein the host cell expresses or over-expresses one or more FcyR (e.g., FcyRIIa H131, FcyRIIa R131, FcyRIIb, FcyRIIIa F158, or FcyRIIIa V158) and the signaling is induced by binding of the variant molecule to the FcyR.
- Reporter cells useful for determining signaling include, for example, cells in which NF AT drives expression of a luciferase reporter (e.g., available from Promega®).
- FcyRs, FcRn, and Cl e.g., Clq
- FcyRs, FcRn, and Cl e.g., Clq
- an antibody or antigen-binding fragment comprising a variant Fc polypeptide or fragment is preferably capable of inducing one or more of: antibody-dependent cell cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP); and complementdependent cytotoxicity. Assays for measuring these functions are known.
- a variant Fc polypeptide or fragment preferably has comparable binding to a human FcRn (e.g., at pH 6.0) and/or a comparable in vivo half-life in a mammal as compared to a reference Fc polypeptide, fragment, antibody, or antigen-binding fragment, respectively.
- a variant Fc polypeptide or fragment preferably has increased binding to a human FcRn (e.g., at pH 6.0) and/or increased in vivo half-life in a mammal as compared to a reference Fc polypeptide, fragment, antibody, or antigen-binding fragment, respectively.
- a variant Fc polypeptide or fragment preferably has a melting temperature (Tm) that is less than 12°C, less than 11°C, less than 10°C, less than 9°C, less than 8°C, less than 7°C, less than 6°C, less than 5°C, less than 4°C, less than 3°C, less than 2°C, or less than 1°C below the Tm of a reference Fc polypeptide (or antibody or antigen-binding fragment comprising the same), or has a Tm that is higher than the Tm of the reference Fc polypeptide or fragment (or polypeptide or antibody comprising the same).
- the reference polypeptide or fragment is or comprises a wild-type human Fc polypeptide (or antibody comprising the same).
- a variant Fc polypeptide or fragment (or antibody or antigenbinding fragment comprising the same) has a melting temperature that is higher than the melting temperature of a reference Fc polypeptide or fragment (or antibody or antigen-binding fragment comprising the same) that comprises the mutations G236A, A330L, I332E, and, optionally, M428L and N434S,
- a variant Fc polypeptide or fragment is preferably capable of being produced in a host cell line (e.g., a CHO cell line) at least about as efficiently (e.g., produces at least about the same titer and/or within less than O. l-fold, less than 0.09-fold, less than 0.08-fold, less than 0.07-fold, less than 0.06-fold, less than 0.05-fold, less than 0.04-fold, less than 0.03-fold, less than 0.02-fold, or less than 0.02 -fold less) as compared to a reference Fc polypeptide or fragment (or antibody or antigen-binding fragment comprising the same).
- a host cell line e.g., a CHO cell line
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a valine (V) at EU position 328, and a glutamic acid (E) at EU position 295.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GALVQE”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG hinge-CH2 polypeptide; or (ii) an IgG hinge-Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an alanine (A) at EU position 230, and a glutamic acid (E) at EU position 295.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wildtype) IgGl Fc polypeptide or fragment thereof (“GAPAQE”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a proline (P) at EU position 292, and an asparagine (N) at EU position 377.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARPIN”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an alanine (A) at EU position 334, and a glutamic acid (E) at EU position 295.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GAKAQE”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a serine (S) at EU position 236, a proline (P) at EU position 292, and a leucine (L) at EU position 300.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GSRPYL”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a proline (P) at EU position 292, and a leucine (L) at EU position 300.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARPYL”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236 and a leucine (L) at EU position 300.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GAYL”).
- the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide; or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an aspartic acid (D) at EU position 239, and a glutamic acid (E) at EU position 268.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GASDHE”).
- the antibody or antigenbinding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein.
- the antibody or antigen-binding fragment is afucosylated.
- the antibody or antigen-binding fragment has increased binding to a human FcyRIIa and/or has decreased binding to a human FcyRIIb, as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa or the human FcyRIIb, respectively, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the increased binding to a human FcyRIIa comprises more than 1-fold, at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG e.g. IgGl
- the human FcyRIIa comprises H131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold greater binding to the human FcyRIIa H131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa H131.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof
- the human FcyRIIa comprises R131 and, optionally, the increased binding to the human FcyRIIa R131 comprises more than 1-fold, at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold greater binding to the human FcyRIIa R131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa R131.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof
- the decreased binding to a human FcyRIIb comprises less than 0.9-fold, less than 0.8-fold, less than 0.7-fold, less than 0.6-fold, or between 0.5-fold and 0.9- fold, of the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIb.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof
- a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a reference polypeptide to the human FcyRIIa to (iv) the binding of the reference antibody or antigen-binding fragment to the human FcyRIIb, wherein the reference antibody or antigen-binding fragment optionally comprises a wild-type human IgG (e.g.
- the human FcyRIIa comprises H131, R131, or both.
- the ratio in (1) is more than 1-fold, at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11 -fold, at least 12-fold, at least 13-fold, or at least 14-fold greater than the ratio in (2).
- an antibody or antigen-binding fragment of the present disclosure comprising a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236 and a leucine (L) at EU position 300.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GAYL”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- the antibody or antigen-binding fragment has increased binding to a human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the increased binding to a human FcyRIIa comprises at least 4- fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13-fold, at least 14-fold, at least 15-fold, at least 16-fold, at least 17-fold, or at least 18-fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof
- the human FcyRIIa comprises H131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 4-fold, at least 5-fold, at least 6- fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold, at least 11-fold, at least 12- fold, at least 13-fold, at least 14-fold, at least 15-fold, at least 16-fold, at least 17-fold, or at least 18-fold greater binding to the human FcyRIIa H131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa H131.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof
- the human FcyRIIa comprises R131 and, optionally, the increased binding to the human FcyRIIa R131 comprises at least 4-fold greater binding to the human FcyRIIa R131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa R131.
- a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG (e.g IgGl) Fc polypeptide or a fragment thereof
- (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa to (iv) the binding of the reference polypeptide to the human FcyRIIb, wherein the reference antibody or antigen-binding fragment comprises a wild-type human IgG Fc polypeptide or a fragment thereof.
- the human FcyRIIa comprises H131, R131, or both.
- the ratio in (1) is at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13-fold, at least 14-fold, at least 15-fold, at least 16-fold, or at least 17-fold greater than the ratio in (2).
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a proline (P) at EU position 292, and a leucine (L) at EU position 300.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARPYL”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- the antibody or antigen-binding fragment has increased binding to a human FcyRIIIa, as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the increased binding to a human FcyRIIa comprises at least 2- fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11 -fold, at least 12-fold, at least 13 -fold, or at least 14-fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa.
- the human FcyRIIa comprises H131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 2-fold, at least 3-fold, at least 4- fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13-fold, or at least 14-fold greater binding to the human FcyRIIa H131 as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa H131.
- the human FcyRIIa comprises R131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 2-fold greater binding to the human FcyRIIa R131 as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa R131.
- (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa to (iv) the binding of the reference antibody or antigen-binding fragment to the human FcyRIIb, wherein the reference antibody or antigen-binding fragment optionally comprises a wild-type human IgG Fc polypeptide or a fragment thereof, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the human FcyRIIa comprises H131, R131, or both.
- the ratio in (1) is at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13- fold, at least 14-fold, or at least 15-fold greater than the ratio in (2).
- the antibody or antigen-binding fragment has increased binding to a human FcyRIIIa, as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the human FcyRIII comprises V158, F158, or both.
- the increased binding to a human FcyRIIIa comprises greater than 2-fold, at least 2.1-fold, at least 2.2-fold, at least 2.3-fold, at least 2.4-fold, at least 2.5-fold, at least 2.6-fold, at least 2.7-fold, at least 2.8-fold, at least 2.9-fold, at least 3.0 fold, at least 3.1-fold, at least 3.2- fold, at least 3.3-fold, at least 3.4-fold, at least 3.5-fold, at least 3.6-fold, or at least 3.7-fold greater binding to the human FcyRIIIa as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIIa.
- the antibody or antigen-binding fragment is capable of binding to a human complement component Iq (Clq), wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of an IgG Fc polypeptide, wherein the variant comprises a serine (S) at EU position 236, a valine (V) at EU position 420, a glutamic acid (E) at EU position 446, and a threonine (T) at EU position 309.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GSGVGELT”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigenbinding fragment is afucoyslated.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide, wherein the variant comprises an alanine (A) at EU position 236 and a proline (P) at EU position 292.
- the antibody or antigen-binding fragment comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARP”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- the antibody or antigen-binding fragment has decreased binding to a human FcyRIIb as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIb, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the decreased binding to a human FcyRIIb comprises less than 0.9-fold, less than 0.8-fold, less than 0.7-fold, less than 0.6-fold, less than 0.5-fold, or less than 0.4-fold as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIb.
- the antibody or antigen-binding fragment has increased binding to a human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the increased binding to the human FcyRIIa comprises greater than 1-fold, at least 2-fold, at least 3 -fold, at least 4-fold, or at least 5 -fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa.
- the human FcyRIIa comprises H131, R131, or both.
- (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a antibody or antigen-binding fragment to the human FcyRIIa to (iv) the binding of the reference antibody or antigen-binding fragment to the human FcyRIIb, wherein the reference antibody or antigenbinding fragment optionally comprises a wild-type human IgG Fc polypeptide or a fragment thereof, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the human FcyRIIa comprises H131, R131, or both.
- the ratio in (1) is at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 10-fold, at least 11-fold, or at least 12-fold greater than the ratio in (2).
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide, wherein the variant comprises a proline (P) at EU position 292 and a leucine (L) at EU position 300, and wherein, optionally, variant and, further optionally, the antibody or antigen-binding fragment has increased binding to a human FcyRIIIa with as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIIa, wherein, optionally, the binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
- the antibody or antigen-binding fragment comprises an (e.g., otherwise wild-type) IgGl CH2 polypeptide or IgG Fc polypeptide (“RPYL”).
- RYL IgG Fc polypeptide
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- the human FcyRIIIa comprises V158, F158, or both, and wherein the increased binding to the human FcyRIIIa comprises at least 4-fold, at least 4.5-fold, at least 5-fold, at least 5.1-fold, or at least 5.2-fold greater binding as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a leucine (L) at EU position 300.
- the IgG CH2 polypeptide or IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“YL”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a lysine (K) at EU position 345, a serine (S) at EU position 236, tyrosine (Y) at EU position 235, and a glutamic acid (E) at EU position 267.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GSEKLYSE”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG hinge-CH2 polypeptide or (ii) an IgG hinge-Fc polypeptide or a fragment thereof, wherein the variant comprises an arginine (R) at EU position 272, a threonine (T) at EU position 309, a tyrosine (Y) at EU position 219, and a glutamic acid (E) at EU position 267.
- the IgG hinge-CH2 polypeptide or an IgG hinge-Fc polypeptide or a fragment thereof comprises an (e.g.
- IgGl hinge-CH2 polypeptide or IgG hinge-Fc polypeptide or a fragment thereof (“SYSEERLT”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a tyrosine (Y) at EU position 236.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GY”).
- GY tyrosine
- the antibody or antigen-binding fragment is afucoyslated.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a tryptophan (W) at EU position 236.
- the IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof comprises an (e.g., otherwise wild-type) IgGl CH2 polypeptide or Fc polypeptide or fragment thereof (“GW”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigenbinding fragment is afucoyslated.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, wherein the IgG Fc polypeptide or fragment thereof, and optionally the polypeptide, is afucosylated, and wherein, further optionally, the variant comprises a leucine (L) at EU position 330 and a glutamic acid (E) at EU postion 332, wherein, still further optionally, the variant does not comprise an aspartic acid (D) at EU position 239, and, even further optionally, comprises a serine (S) at EU position 239.
- the IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof comprises an (e.g., otherwise wild-type) IgGl CH2 polypeptide or Fc polypeptide or fragment thereof (“GA-afuc” or “GAALIE-afuc”, respectively).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a leucine (L) at EU position 243, a glutamic acid (E) at EU position 446, a leucine (L) at EU position 396, and a glutamic acid (E) at EU position 267.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“FLSEPLGE”).
- the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
- the antibody or antigen-binding fragment is afucoyslated.
- an antibody or antigen-binding fragment that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an aspartic acid (D) at EU position 239, a glutamic acid (E) and EU position 332, a leucine (L) at EU position 428, and a serine (S) or an alanine (A) at EU position 434.
- the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GASDIEMLNS” or “GASDIEMLNA”).
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of an (e.g. IgGl) IgG Fc polypeptide, wherein the variant comprises the following mutations, according to EU numbering: (i) M428L, N434S, G236A, L328V, and Q295E; (ii) M428L, N434S, G236A, R292P, and I377N; (iii) M428L, N434S, G236A, and Y300L; (iv) M428L, N434S, G236A, R292P, and Y300L; (v) M428L, N434S, G236A, L328V, and Q295E, wherein the antibody or antigen-binding fragment is afucosylated; (vi) M428L, N434S, G236A, R292P, and I377N, wherein the antibody or
- the variant of an (e.g. IgGl) IgG Fc polypeptide comprises amino acid substitutions that consist essentially of the substitution mutations in (i), (ii), (iii), (iv), (v), (vi), (vii), or (viii) above.
- the antibody or antigen-binding fragment comprises a kappa light chain.
- an antibody or antigen-binding fragment of the present disclosure comprises a variant of an (e.g. IgGl) IgG Fc polypeptide, wherein the variant comprises the following mutations, according to EU numbering: (i) M428L, N434A, G236A, L328V, and Q295E; (ii) M428L, N434A, G236A, R292P, and I377N; (iii) M428L, N434A, G236A, and Y300L; (iv) M428L, N434A, G236A, R292P, and Y300L; (v) M428L, N434A, G236A, L328V, and Q295E, wherein the antibody or antigen-binding fragment is afucosylated; (vi) M428L, N434A, G236A, R292P, and I377N, wherein the antibody or
- the variant of an IgG Fc polypeptide comprises amino acid substitutions that consist essentially of the substitution mutations in (i), (ii), (iii), (iv), (v), (vi), (vii), or (viii) above.
- the antibody comprises a kappa light chain.
- the antibody or antigenbinding fragment is capable of eliciting continued protection in vivo in a subject even once no detectable levels of the antibody or antigen-binding fragment can be found in the subject (z.e., when the antibody or antigen-binding fragment has been cleared from the subject following administration). Such protection is referred to herein as a vaccinal effect.
- an antibody or antigen-binding fragment comprises one or more modifications, such as, for example, mutations in the Fc comprising G236A, A330L, and I332E, that are capable of activating dendritic cells that may induce, e.g., T cell immunity to the antigen.
- the antibody or antigen-binding fragment comprises a Fc polypeptide or a fragment thereof, including a CH2 (or a fragment thereof, a CH3 (or a fragment thereof), or a CH2 and a CH3, wherein the CH2, the CH3, or both can be of any isotype and may contain amino acid substitutions or other modifications as compared to a corresponding wild-type CH2 or CH3, respectively.
- a Fc of the present disclosure comprises two CH2-CH3 polypeptides that associate to form a dimer.
- an antibody or antigen-binding fragment of the present disclosure comprises a heavy chain comprising an amino acid sequence having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity (or similarity) to, or comprising or consisting of, the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765.
- the antibody or antigen-binding fragment can be monoclonal.
- the term “monoclonal antibody” (mAb) as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, z.e., individual antibodies comprising the population are identical except for possible naturally occurring mutations that may be present, in some cases in minor amounts. Monoclonal antibodies are highly specific, being directed against a single antigenic site. Furthermore, in contrast to polyclonal antibody preparations that include different antibodies directed against different epitopes, each monoclonal antibody is directed against a single epitope of the antigen. In addition to their specificity, the monoclonal antibodies are advantageous in that they may be synthesized uncontaminated by other antibodies.
- monoclonal antibodies useful in the present invention may be prepared by the hybridoma methodology first described by Kohler et al., Nature 256 :495 (1975), or may be made using recombinant DNA methods in bacterial, eukaryotic animal, or plant cells (see, e.g., U.S. Pat. No. 4,816,567).
- Monoclonal antibodies may also be isolated from phage antibody libraries using the techniques described in Clackson et al., Nature, 352:624-628 (1991) and Marks et al., J. Mol. Biol., 222:581-597 (1991), for example.
- Monoclonal antibodies may also be obtained using methods disclosed in PCT Publication No. WO 2004/076677A2.
- Antibodies and antigen-binding fragments of the present disclosure include “chimeric antibodies” in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (see, U.S. Pat. Nos. 4,816,567; 5,530,101 and 7,498,415; and Morrison et al., Proc. Natl. Acad. Sci. USA, 57:6851-6855 (1984)).
- chimeric antibodies may comprise human and non-human residues.
- chimeric antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance. For further details, see Jones etal., Nature 321 :522-525 (1986); Riechmann etal., Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol. 2:593-596 (1992). Chimeric antibodies also include primatized and humanized antibodies.
- a “humanized antibody” is generally considered to be a human antibody that has one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues are typically taken from a variable domain. Humanization may be performed following the method of Winter and co-workers (Jones et al., Nature, 321 : 522-525 (1986); Reichmann et al., Nature, 332:323-327 (1988); Verhoeyen et al., Science, 239: 1534- 1536 (1988)), by substituting non-human variable sequences for the corresponding sequences of a human antibody. Accordingly, such “humanized” antibodies are chimeric antibodies (U.S. Pat. Nos.
- a “humanized” antibody is one which is produced by a non-human cell or animal and comprises human sequences, e.g., He domains.
- human antibody is an antibody containing only sequences that are present in an antibody that is produced by a human (i.e., sequences that are encoded by human antibodyencoding genes).
- human antibodies may comprise residues or modifications not found in a naturally occurring human antibody (e.g., an antibody that is isolated from a human), including those modifications and variant sequences described herein. These are typically made to further refine or enhance antibody performance.
- human antibodies are produced by transgenic animals. For example, see U.S. Pat. Nos. 5,770,429; 6,596,541 and 7,049,426.
- an antibody or antigen-binding fragment of the present disclosure is chimeric, humanized, or human.
- PK pharmacokinetic
- ti/2 or “half-life” refers to the elimination half-life of the antibody or antigen-binding fragment included in the pharmaceutical composition administered to a subject.
- Clast generally refers to the last measurable plasma concentration (z.e., subsequent thereto, the substance is not present at a measurable concentration in plasma).
- the antibody, or the antigen-binding fragment comprises a human antibody, a monoclonal antibody, a purified antibody, a single chain antibody, a Fab, a Fab’, a F(ab’)2, or Fv.
- the antibody or antigen-binding fragment is a multi-specific antibody or antigen-binding fragment, wherein, optionally, the antibody or antigen-binding fragment is a bi-specific antibody or antigen-binding fragment.
- the antibody or antigen-binding fragment comprises an (e.g., IgGl) Fc polypeptide or a fragment thereof.
- the antibody or antigenbinding fragment comprises an IgG, IgA, IgM, IgE, or IgD isotype.
- the antibody or antigen-binding fragment comprises an IgG isotype selected from IgGl, IgG2, IgG3, and IgG4. In certain embodiments, the antibody or antigen-binding fragment comprises an IgGl isotype.
- the antibody or antigen-binding fragment comprises an IgGlm3 allotype, an IgGlml7 allotype, an IgGl ml allotype, or any combination thereof. In some embodiments, the antibody or antigen-binding fragment comprises an IgG3 isotype.
- the antibody or antigen-binding fragment comprises an IgG3m(b*) allotype, an IgG3m(b**) allotype, an IgG3m(c3*) allotype, an IgG3m(c3c5*) allotype, an IgG3m(g*) allotype, an IgG3m(s*) allotype, an IgG3m(st*) allotype, an IgG3 GM5 allotype, an IgG3 G4m5 allotype, or any combination thereof.
- the Fc polypeptide or fragment thereof comprises: (i) a mutation that increases binding affinity to a human FcRn (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols)), as compared to a reference Fc polypeptide that does not comprise the mutation; and/or (ii) a mutation that increases binding affinity to a human FcyR (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols, and/or as measured using mesoscale discovery (MSD))) as compared to a reference Fc polypeptide that does not comprise the mutation.
- SPR surface plasmon resonance
- MSD mesoscale discovery
- the mutation that increases binding affinity to a human FcRn comprises: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q31 II; D376V; T307A; E380A; or any combination thereof.
- the mutation that enhances binding to a FcyR comprises S239D; I332E; A330L; G236A; or any combination thereof.
- the mutation that enhances binding to a FcyR comprises: (i) S239D/I332E; (ii) S239D/A330L/I332E; (iii) G236A/S239D/I332E; or (iv) G236A/A330L/I332E, wherein the Fc polypeptide or fragment thereof optionally comprises Ser at position 239.
- the antibody or antigen-binding fragment comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or which is aglycosylated, and/or which is afucosylated.
- antibody conjugates that comprise an antibody or antigen-binding fragment of the present disclosure conjugated to a payload molecule or moiety, such as, for example, a drug compound.
- Antibody or antigen-binding fragment structures amenable to conjugation include, for example, lysine amino acid residues, cysteine amino acid residues, nonnatural amino acids (e.g., amino acid analogs), carbohydrate moi eties, and N-termini of heavy and light chains (or of VH and VL).
- Such structures may be native to the antibody or antigenbinding fragment, or may be introduced (e.g., a native non-lysine or non-cysteine amino acid of the antibody may be replaced by a lysine or cysteine, and/or a lysine or cysteine may be inserted into the amino acid sequence of the antibody or antigen-binding fragment, as an additional amino acid).
- conjugation techniques in which a protein with a strong interaction with an antibody or antigen-binding fragment (e.g., FcRn, Protein A, Protein G, or fragments of these) is conjugated to a compound and non-covalent interaction between the protein and the antibody or antigen-binding fragment effectively couples the compound to the antibody or antigen-binding fragment, as well as modification in nucleotide-binding sites (e.g., for modification by photoaffinity labeling), or in catalytic sites (if present).
- an antibody or antigen-binding fragment e.g., FcRn, Protein A, Protein G, or fragments of these
- modification in nucleotide-binding sites e.g., for modification by photoaffinity labeling
- catalytic sites if present.
- WO 2006/034488 WO 2011/005481, WO 2011/156328, WO 2016/040856, WO 2015/157595, WO 2013/093809, WO 2005/018572, WO 1996/014339, WO 2008/070593, WO 2009/092011, WO 2020/191306, WO 2006/074397, WO 2008/038024, WO 2010/141902, and WO 2004/042017, as well as in Sang et al., Methods Mol. Biol. 2078:235-250 (2020), Haque et al. Chem. Commun. 57:10689-10702 (2021), Brun and Gauzy -Lazo, Methods Mol. Biol.
- Non-limiting examples of mutations include the following (all residue positions according to EU numbering of human IgGl): Al 18C, S239C, T289C, A330C, and S442C in the heavy chain, and K149C in the light chain.
- An antibody or antigen-binding fragment of a conjugate can comprise one or more native lysine, one or more native cysteine, one or more introduced lysine, one or more introduced cysteine, or any combination thereof.
- an antibody or antigen-binding fragment comprises its native lysines, its native cysteines, or both.
- an antibody or antigen-binding fragment does not comprise modifications in its amino acid sequence for conjugation.
- Conjugation may comprise forming conjugates at all, or at fewer than all, of the available conjugation sites.
- a payload molecule is covalently linked by a linker to the antibody or antigen-binding fragment thereof.
- the linker is selected from a cleavable linker and a non-cleavable linker.
- the cleavable linker is a protease-sensitive linker, a pH-sensitive linker, or a glutathione-sensitive linker.
- the cleavable linker is a protease-sensitive linker comprising a valine-citrulline dipeptide.
- the linker comprises a maleimide group.
- the herein disclosed antibody or antigen-binding fragment thereof comprises a reduced disulfide bridge in a hinge region and the reduced disulfide bridge is coupled to the maleimide group.
- the linker further comprises a self-demolishing group, such as, for example, para-amino benzyl alcohol (PABC).
- PABC para-amino benzyl alcohol
- an antibody antigen-binding fragment conjugate comprises payload molecule that is covalently linked by a linker to the antibody or antigenbinding fragment thereof.
- Linkers used in antibody antigen-binding fragment conjugates may be organic compounds that fall into one of two groups, organized according to the mechanism by which the payload molecule is released from the carrier molecule.
- Cleavable linkers are designed to be selectively degraded or cleaved according to an inherent property of the target cell: three types of cleavable linkers are protease-sensitive linkers (whereby cleavage of the linker, e.g., a linker comprising a valine-citrulline or phenylalanine-lysine dipeptide or a tetrapeptide (e.g., GFLG or ALAL), by proteases present in a tumor cell lysosome releases the payload molecule); pH-sensitive linkers, containing an acid labile group that is selectively hydrolyzed by the lower pH of endosomal and lysosomal compartments, relative to cytosolic pH; and glutathione-sensitive linkers, which comprise a disulfide bridge that is reduced by intracellular glutathione.
- Non-cleavable linkers rely on non-specific degradation of the antibody conjugate to release the payload molecule.
- an antibody or antigen-binding fragment conjugate comprises a linker is selected from a cleavable linker and a non-cleavable linker.
- the linker is a cleavable linker selected from a protease-sensitive linker, a pH-sensitive linker, or a glutathionesensitive linker.
- a cleavable linker is a protease-sensitive linker comprising a valine-citrulline dipeptide.
- a linker may be connected or coupled to the antibody or antigen-binding fragment thereof using any appropriate technique or mechanism.
- a linker comprises a maleimide group (optionally PEGylated) capable of reacting with a reduced disulfide bridge in a hinge region of the antibody or antigen-binding fragment thereof.
- Other sites on the carrier molecule (i.e., the antibody or antigen-binding fragment thereof) suitable for conjugation to a linker may be introduced or engineered using recombinant techniques, such as introducing cysteine residues or non-natural amino acids for site-specific conjugation. Methods for introducing such modifications include, for example, the method described in Examples 6.3-7 of PCT Publication No. WO 2012/032181.
- a linker further comprises a self-demoli shing group, also referred to as a self-immolative group or a self-immolative spacer, to assist in a selective cleavage reaction.
- the self-demoli shing group is para-amino benzyl alcohol (PABC).
- Click chemistries useful for generating antibody conjugates include those described in Meyer et al.. Bioconjug. Chem. 27(12):2791-2807 (2016), and are herein incorporated by reference in their entirety.
- the payload molecule may be selected from a therapeutic agent and a detectable indicator.
- the present disclosure provides isolated polynucleotides that encode any of the presently disclosed antibodies or an antigen-binding fragment thereof, or a portion thereof (e.g., a CDR, a VH, a VL, a Fab, a Fd, a heavy chain, or a light chain, or a heavy chain and a light chain), or that encode a presently disclosed polypeptide.
- the polynucleotide comprises deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), wherein the RNA optionally comprises messenger RNA (mRNA).
- DNA deoxyribonucleic acid
- RNA ribonucleic acid
- mRNA messenger RNA
- the polynucleotide comprises a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof.
- the polynucleotide comprises a pseudouridine, a N6-methyladenonsine, a 5-methylcytidine, a 2- thiouridine, or any combination thereof.
- the pseudouridine comprises N1 -methylpseudouridine.
- the polynucleotide is codon-optimized for expression in a host cell (e.g., a human cell, or a CHO cell).
- a host cell e.g., a human cell, or a CHO cell.
- codon optimization can be performed using known techniques and tools, e.g., using the GenScript® OptimumGeneTM tool, or the like).
- Codon-optimized sequences include sequences that are partially codon-optimized (z.e., one or more codon is optimized for expression in the host cell) and those that are fully codon-optimized.
- polynucleotides encoding antibodies and antigen-binding fragments of the present disclosure may possess different nucleotide sequences while still encoding a same antibody or antigen-binding fragment due to, for example, the degeneracy of the genetic code, splicing, and the like.
- the polynucleotide can comprise deoxyribonucleic acid (DNA) or ribonucleic acid (RNA).
- the RNA comprises messenger RNA (mRNA), more specifically circRNA, taRNA, or saRNA.
- mRNA messenger RNA
- the polynucleotide may be optimized for expression in a host cell.
- the polynucleotide comprises taRNA or saRNA, one or more sequences encoding a replication protein or peptide, one or more sequences encoding an antibody or antigen-binding fragment, or combinations thereof may be optimized for expression in human cells.
- a polynucleotide comprises (i) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the VH polynucleotide sequence of an antibody as set forth in Table 3, and (ii) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the corresponding VL polynucleotide sequence of the antibody as set
- a polynucleotide comprises (i) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to SEQ ID NO.:29, and (ii) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to SEQ ID NO.:30.
- the polynucleotide can comprise deoxyribonucleic acid (DNA) or ribonucleic acid (RNA).
- the RNA comprises messenger RNA (mRNA).
- the first polynucleotide may further comprise a nucleic acid sequence encoding a replication protein or peptide.
- a second polynucleotide is provided that comprises a nucleic acid sequence encoding a replication protein or peptide suitable to cause replication of the first polynucleotide.
- the first polynucleotide comprises or consists of a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the VH polynucleotide sequence of an antibody as set forth in Table 3 and the second polynucleotide comprises or consists of a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at
- the polynucleotides are mRNAs at least one and optionally both of the first and second polynucleotide is a saRNA, and further comprise a nucleic acid sequence encoding a replication protein or peptide.
- a third polynucleotide is provided that comprises a nucleic acid sequence encoding a replication protein or peptide suitable to cause replication of the first polynucleotide and second polynucleotide.
- Vectors are also provided, wherein the vectors comprise or contain a polynucleotide as disclosed herein (e.g., a polynucleotide that encodes an antibody or antigen-binding fragment).
- a vector can comprise any one or more of the vectors disclosed herein.
- a vector is provided that comprises a DNA plasmid construct encoding the antibody or antigen-binding fragment, or a portion thereof (e.g., so-called “DMAb”; see, e.g., Muthumani et al., J Infect Dis. 2/-/(3):369-378 (2016); Muthumani etal., Hum Vaccin Immunother 9:2253-2262 (2013)); Flingai et al., Sci Rep.
- a DNA plasmid construct comprises a single open reading frame encoding a heavy chain and a light chain (or a VH and a VL) of the antibody or antigen-binding fragment, wherein the sequence encoding the heavy chain and the sequence encoding the light chain are optionally separated by polynucleotide encoding a protease cleavage site and/or by a polynucleotide encoding a self-cleaving peptide.
- the substituent components of the antibody or antigen-binding fragment are encoded by a polynucleotide comprised in a single plasmid. In other embodiments, the substituent components of the antibody or antigen-binding fragment are encoded by a polynucleotide comprised in two or more plasmids (e.g, a first plasmid comprises a polynucleotide encoding a heavy chain, VH, or VH+CH1, and a second plasmid comprises a polynucleotide encoding the cognate light chain, or VL).
- a single plasmid comprises a polynucleotide encoding a heavy chain and/or a light chain from two or more antibodies or antigen-binding fragments of the present disclosure.
- An exemplary expression vector is pVaxl, available from Invitrogen®.
- a DNA plasmid of the present disclosure can be delivered to a subject by, for example, electroporation (e.g., intramuscular electroporation), or with an appropriate formulation (e.g., hyaluronidase).
- the vector may comprise or consist of circRNA, taRNA, or saRNA.
- the RNA therapeutic consists of a naked circRNA, taRNA, or saRNA vector
- the cirRNA, taRNA, or saRNA may, in particular, contain modified nucleosides or other modifications disclosed herein to facilitate delivery to and expression in a human host cell in vivo.
- naked DNA vectors or other naked DNA therapeutics may contain modifications to facilitate delivery to and expression in a human host cell in vivo.
- DNA therapeutics and RNA therapeutics including circRNA therapeutics, taRNA therapeutics, or saRNA therapeutics, are also provided, wherein the DNA therapeutics or RNA therapeutics comprise or contain a polynucleotide as disclosed herein (e.g., a polynucleotide that encodes an antibody or antigen-binding fragment that binds to two or more bacterial antigens as described herein).
- a polynucleotide as disclosed herein e.g., a polynucleotide that encodes an antibody or antigen-binding fragment that binds to two or more bacterial antigens as described herein.
- the present disclosure also provides a host cell expressing an antibody or antigen-binding fragment according to the present disclosure; or comprising or containing a DNA therapeutic, RNA therapeutic, vector, or polynucleotide according the present disclosure.
- a method comprises administering to a subject a first polynucleotide (e.g., mRNA) encoding an antibody heavy chain, a VH, or a Fd (VH + CHI), and administering to the subject a second polynucleotide (e.g., mRNA) encoding the cognate antibody light chain, or VL.
- a first polynucleotide e.g., mRNA
- VH + CHI Fd
- a method comprises administering to a subject a first plasmid or vector that comprises a polynucleotide (e.g. mRNA) encoding a VH, a VH + CHI, or a heavy chain, and a second plasmid or vector is provided that comprises a polynucleotide (e.g. mRNA) encoding the cognate VL or light chain.
- a first plasmid or vector and/or the second plasmid or vector can be comprised in a composition that further comprises a pharmaceutically acceptable carrier, excipient, or diluent.
- a polynucleotide e.g., mRNA
- a polynucleotide that encodes a heavy chain and a light chain of an antibody or antigen-binding fragment thereof.
- a polynucleotide e.g., mRNA
- a polynucleotide is provided that encodes two heavy chains and two light chains of an antibody or antigen-binding fragment thereof. See, e.g. Li, JQ., Zhang, ZR., Zhang, HQ. et al. Intranasal delivery of replicating mRNA encoding neutralizing antibody against SARS-CoV-2 infection in mice. Sig Transduct Target Ther 6, 369 (2021).
- a polynucleotide is delivered to a subject via an alphavirus replicon particle (VRP) delivery system.
- VRP alphavirus replicon particle
- a replicon comprises a modified VEEV replicon comprising two subgenomic promoters.
- a polynucleotide or replicon can translate simultaneously the heavy chain (or VH, or VH+1) and the light chain (or VL) of an antibody or antigen-binding fragment thereof.
- a method is provided that comprises delivering to a subject such a polynucleotide or replicon.
- the present disclosure also provides a host cell expressing an antibody or antigen-binding fragment according to the present disclosure; or comprising or containing a vector or polynucleotide according the present disclosure.
- the cells include but are not limited to, eukaryotic cells, e.g., yeast cells, animal cells, insect cells, plant cells; and prokaryotic cells, including A. coli.
- the cells are mammalian cells, such as human B cells.
- the cells are a mammalian cell line such as CHO cells (e.g., DHFR- CHO cells (Urlaub et al., PNAS 77:4216 (1980)), human embryonic kidney cells (e.g., HEK293T cells), PER.C6 cells, Y0 cells, Sp2/0 cells.
- NS0 cells human liver cells, e.g.
- Hepa RG cells myeloma cells or hybridoma cells.
- mammalian host cell lines include mouse sertoli cells (e.g., TM4 cells); monkey kidney CV1 line transformed by SV40 (COS-7); baby hamster kidney cells (BHK); African green monkey kidney cells (VERO-76); monkey kidney cells (CV1); human cervical carcinoma cells (HELA); human lung cells (W138); human liver cells (Hep G2); canine kidney cells (MDCK; buffalo rat liver cells (BRL 3 A); mouse mammary tumor (MMT 060562); TRI cells; MRC 5 cells; and FS4 cells.
- mouse sertoli cells e.g., TM4 cells
- COS-7 monkey kidney CV1 line transformed by SV40
- BHK baby hamster kidney cells
- VERO-76 African green monkey kidney cells
- CV1 monkey kidney cells
- HELA human cervical carcinoma cells
- W138 human lung cells
- Hep G2 human liver cells
- canine kidney cells MDCK; buffalo
- Mammalian host cell lines suitable for antibody production also include those described in, for example, Yazaki and Wu, Methods in Molecular Biology, Vol. 248 (B. K. C. Lo, ed., Humana Press, Totowa, N.J.), pp. 255-268 (2003).
- a host cell is a prokaryotic cell, such as an E. coli.
- a prokaryotic cell such as an E. coli.
- the expression of peptides in prokaryotic cells such as A. coli is well established (see, e.g., Pluckthun, A. Bio/Technology 9:545-551 (1991).
- antibodies may be produced in bacteria, in particular when glycosylation and Fc effector function are not needed.
- For expression of antibody fragments and polypeptides in bacteria see, e.g., U.S. Pat. Nos. 5,648,237; 5,789,199; and 5,840,523.
- the cell may be transfected with a vector, DNA therapeutic, or RNA therapeutic according to the present description with an expression vector.
- transfection refers to the introduction of nucleic acid molecules, such as DNA or RNA (e.g. mRNA) molecules, into cells, such as into eukaryotic cells.
- RNA e.g. mRNA
- transfection encompasses any method known to the skilled person for introducing nucleic acid molecules into cells, such as into eukaryotic cells, including into mammalian cells.
- the present disclosure also provides recombinant host cells that heterologously express an antibody or antigen-binding fragment of the present disclosure.
- the cell may be of a species that is different to the species from which the antibody was fully or partially obtained (e.g., CHO cells expressing a human antibody or an engineered human antibody).
- the cell type of the host cell does not express the antibody or antigen-binding fragment in nature.
- the present disclosure provides methods for producing an antibody, or antigen-binding fragment, wherein the methods comprise culturing a host cell of the present disclosure under conditions and for a time sufficient to produce the antibody, or the antigenbinding fragment.
- Methods useful for isolating and purifying recombinantly produced antibodies may include obtaining supernatants from suitable host cell/vector systems that secrete the recombinant antibody into culture media and then concentrating the media using a commercially available filter. Following concentration, the concentrate may be applied to a single suitable purification matrix or to a series of suitable matrices, such as an affinity matrix or an ion exchange resin.
- One or more reverse phase HPLC steps may be employed to further purify a recombinant polypeptide. These purification methods may also be employed when isolating an immunogen from its natural environment. Methods for large scale production of one or more of the isolated/recombinant antibody described herein include batch cell culture, which is monitored and controlled to maintain appropriate culture conditions. Purification of soluble antibodies may be performed according to methods described herein and known in the art and that comport with laws and guidelines of domestic and foreign regulatory agencies.
- compositions that comprise any one or more of the presently disclosed antibodies, antigen-binding fragments, polypeptides, polynucleotides, vectors, DNA therapeutics, RNA therapeutics) (including mRNA therapeutics, circ RNA therapeutics, taRNA therapeutics, and saRNA therapeutics), or host cells, singly or in any combination, and can further comprise a pharmaceutically acceptable carrier, excipient, or diluent.
- RNA therapeutics including mRNA therapeutics, circ RNA therapeutics, taRNA therapeutics, and saRNA therapeutics
- a composition comprises a first vector comprising a first plasmid, and a second vector comprising a second plasmid, wherein the first plasmid comprises a polynucleotide encoding a heavy chain, VH, or VH+CH1, and a second plasmid comprises a polynucleotide encoding the cognate light chain or VL of the antibody or antigen-binding fragment thereof.
- a composition comprises a polynucleotide (e.g., mRNA) coupled to a suitable delivery vehicle or carrier.
- a composition comprises a first polynucleotide (e.g., mRNA) encoding an antibody heavy chain, a VH, or a Fd (VH + CHI), and a second polynucleotide (e.g., mRNA) encoding the cognate antibody light chain or VL.
- a first polynucleotide e.g., mRNA
- VH + CHI a Fd
- VH + CHI Fd
- Exemplary vehicles or carriers for administration to a human subject include a lipid or lipid-derived delivery vehicle, such as a liposome, solid lipid nanoparticle, oily suspension, submicron lipid emulsion, lipid microbubble, inverse lipid micelle, cochlear liposome, lipid microtubule, lipid microcylinder, or lipid nanoparticle (LNP) or a nanoscale platform see, e.g., Li et al. Wilery Interdiscip Rev. Nanomed Nanobiotechnol. 77(2):el530 (2019)). Principles, reagents, and techniques for designing appropriate mRNA and formulating mRNA-LNP and delivering the same are described in, for example, Pardi et al.
- lipid nanoparticles e.g., ionizable cationic lipid/phosphatidylcholine/cholesterol/PEG-lipid; ionizable lipid:distearoyl PC:cholesterol:polyethylene glycol lipid
- subcutaneous, intramuscular, intradermal, intravenous, intraperitoneal, and intratracheal administration of the same, are incorporated herein by reference.
- a composition comprises a first antibody or antigen-binding fragment of the present disclosure and a second antibody or antigen-binding fragment of the present disclosure, wherein of the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are different.
- the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment bind to the following, respectively: an LTA and a Gmd; an LTA and a PFT (e.g., an alpha toxin, a gamma toxin, or a leukotoxin); an LTA and a SpA; an LTA and a ClfA; a Gmd and a PFT; a Gmd and a SpA; a Gmd and a ClfA; a PFT and a SpA; a PFT and a ClfA; a SpA and a ClfA; a Sbi and an LTA; a Sbi and a Gmd; a Sbi and a PFT; a Sbi and a SpA; or a Sbi and a ClfA.
- an LTA and a Gmd an LTA and a PFT (e.g., an alpha toxin,
- a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to an opsonizing target and a second antibody or antigen-binding fragment thereof binds to a neutralizing target.
- a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to a target selected from LTA, ClfA, Protein A, and Gmd and a second antibody or antigen-binding fragment thereof binds to an alpha toxin.
- a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to a target selected from LTA, ClfA, Protein A, and Gmd and a second antibody or antigen-binding fragment thereof binds to a neutralizing target.
- a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to an opsonizing target and a second antibody or antigen-binding fragment thereof binds to an alpha toxin.
- a combination or composition comprises a first antibody or antigen-binding fragment thereof and a second antigen-binding fragment thereof, wherein the first antibody or antigen-binding fragment thereof or the second antibody or antigen-binding fragment thereof comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) S
- a combination or composition comprises a first antibody or antigen-binding fragment thereof and a second antigen-binding fragment thereof, wherein the first antibody or antigen-binding fragment thereof and the second antibody or antigen-binding fragment thereof comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively.
- the six CDR sequences e.g., according to IMGT
- VH and VL of (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and S
- the first antibody or antigen-binding fragment and/or the second antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S/H435R/Y436F mutations.
- a composition comprises polynucleotide, a vector, a RNA therapeutic, or an DNA therapeutic.
- the composition comprises a single polynucleotide encoding i) a heavy chain, VH, or VH+CH; and ii) a cognate light chain, VL, or VL+CL, of the antibody or antigen-binding fragment.
- the polynucleotide is an saRNA and further encodes a replication protein or peptide.
- the polynucleotide is a circRNA.
- the composition comprises a first taRNA encoding i) a heavy chain, VH, or VH+CH; and ii) a cognate light chain, VL, or VL+CL, of the antibody or antigenbinding fragment, and a second taRNA that encodes a replication protein or peptide.
- at least one, or both the first and second taRNA is a circRNA.
- the composition comprises a first polynucleotide encoding a heavy chain, VH, or VH+CH, and a second polynucleotide encoding a cognate light chain, VL, or VL+CL, of the antibody or antigen-binding fragment.
- the polynucleotides are mRNAs
- the first polynucleotide, the second polynucleotide, or both are saRNAs or taRNAs an further encode a replication protein or peptide.
- at least one, at least two, or all of the polynucleotides is a circRNA.
- the composition comprises a first mRNA encoding a heavy chain, VH, or VH+CH, and a second mRNA encoding a cognate light chain, VL, or VL+CL, of the antibody or antigen-binding fragment.
- the composition in which the polynucleotides are mRNAs, is a taRNA composition that comprises a third taRNA that encodes a replication protein or peptide.
- at least one, at least two, or all of the polynucleotides is a circRNA.
- LTA laminol
- Gmd glucosaminidase
- PFT Pore Forming Toxin
- a leukocidin e.g., LukED, a LukSF, or both
- an alpha toxin e.g., Hla
- a gamma toxin or gamma hemolysin e.g., HIgAB
- SpA Staphylococcal Protein A
- ClfA clumping factor A
- an infection comprises infection by bacteria of any or more of: genus Staphylococcus,' genus Enterococcus,' genus Streptococcus,' and genus Lactococcus.
- an infection comprises infection by: S. aureus (e.g., MRS A); Coagulase-negative staphylococci (CoNS); S. epidermidis,' S. pneumoniae,' S. lugdunensis,' or S. hominis,' S. aureus FPR3757; S. az//vz/.s NE284 TE2 mutant; S. epidermidis RP62A; S. epidermidis 1200; S.
- S. aureus e.g., MRS A
- CoNS Coagulase-negative staphylococci
- S. epidermidis S. pneumoniae,' S. lugdunensis,' or S. hominis,' S. aureus FPR3757
- an infection comprises infection by a gram-positive bacteria.
- Methods of diagnosis may include contacting an antibody, antibody fragment (e.g., antigen binding fragment) with a sample.
- samples may be isolated from a subject, for example an isolated tissue sample taken from, for example, nasal passages, sinus cavities, salivary glands, lung, liver, pancreas, kidney, ear, eye, placenta, alimentary tract, heart, ovaries, pituitary, adrenals, thyroid, brain, skin, or blood.
- the methods of diagnosis may also include the detection of an antigen/antibody complex, in particular following the contacting of an antibody or antibody fragment with a sample.
- a detection step can be performed at the bench, i.e. without any contact to the human or animal body. Examples of detection methods are well-known to the person skilled in the art and include, e.g., ELISA (enzyme-linked immunosorbent assay), including direct, indirect, and sandwich ELISA.
- “Treat,” “treatment,” or “ameliorate” refers to medical management of a disease, disorder, or condition of a subject e.g., a human or non-human mammal, such as a primate, horse, cat, dog, goat, mouse, or rat).
- an appropriate dose or treatment regimen comprising an antibody or composition of the present disclosure is administered in an amount sufficient to elicit a therapeutic or prophylactic benefit.
- Therapeutic or prophylactic/preventive benefit includes improved clinical outcome; lessening or alleviation of symptoms associated with a disease; decreased occurrence of symptoms; improved quality of life; longer disease-free status; diminishment of extent of disease, stabilization of disease state; delay or prevention of disease progression; remission; survival; prolonged survival; or any combination thereof.
- therapeutic or prophylactic/preventive benefit includes reduction or prevention of hospitalization for treatment of a bacterial infection i.e., in a statistically significant manner).
- therapeutic or prophylactic/preventive benefit includes a reduced duration of hospitalization for treatment of a bacterial infection (z.e., in a statistically significant manner).
- a “therapeutically effective amount” or “effective amount” of an antibody, antigenbinding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition of this disclosure refers to an amount of the composition or molecule sufficient to result in a therapeutic effect, including improved clinical outcome; lessening or alleviation of symptoms associated with a disease; decreased occurrence of symptoms; improved quality of life; longer disease-free status; diminishment of extent of disease, stabilization of disease state; delay of disease progression; remission; survival; or prolonged survival in a statistically significant manner.
- a therapeutically effective amount refers to the effects of that ingredient or cell expressing that ingredient alone.
- a therapeutically effective amount refers to the combined amounts of active ingredients or combined adjunctive active ingredient with a cell expressing an active ingredient that results in a therapeutic effect, whether administered serially, sequentially, or simultaneously.
- a human subject treated according to the present disclosure is an infant, a child, a young adult, an adult of middle age, or an elderly person. In certain embodiments, a human subject treated according to the present disclosure is less than 1 year old, or is 1 to 5 years old, or is between 5 and 125 years old (e.g., 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, or 125 years old, including any and all ages therein or therebetween).
- a human subject treated according to the present disclosure is 0-19 years old, 20-44 years old, 45-54 years old, 55-64 years old, 65-74 years old, 75-84 years old, or 85 years old, or older. Persons of middle, and especially of elderly age are can be at particular risk.
- the human subject is 45-54 years old, 55-64 years old, 65-74 years old, 75-84 years old, or 85 years old, or older.
- the human subject is male. In some embodiments, the human subject is female.
- a subject treated according to the present disclosure has received a vaccine for a bacterial infection and the vaccine is determined to be ineffective, e.g., by postvaccine infection or symptoms in the subject, by clinical diagnosis or scientific or regulatory consensus.
- Prophylaxis of a bacterial infection refers in particular to prophylactic settings, wherein the subject was not diagnosed with infection by the bacteria (either no diagnosis was performed or diagnosis results were negative) and/or the subject does not show or experience symptoms of bacterial infection.
- Prophylaxis of bacterial infection is particularly useful in subjects at greater risk of severe disease or complications when infected, such as pregnant women, children (such as children under 59 months), the elderly, subjects with chronic medical conditions (such as chronic cardiac, pulmonary, renal, metabolic, neurodevelopmental, liver or hematologic diseases) and individuals with immunosuppressive conditions (such as HIV/AIDS, receiving chemotherapy or steroids, or malignancy), subjects using a ventilator, subjects undergoing or having recently undergone surgery, subjects with a blood stream infection or history of blood stream infections, subjects receiving or having received a prosthetic joint or other implant, subjects undergoing or having undergone dialysis, subjects, subjects with bacteremia or a history of bacteremia, subjects having wound, or the like.
- prophylaxis of bacterial infection is also particularly useful in subjects at greater risk acquiring bacterial infection, e.g., due to increased exposure, for example subjects working or staying in unsanitary areas.
- treatment is administered as peri-exposure or pre-exposure prophylaxis.
- treatment is administered as post-exposure prophylaxis.
- the subject has a bacterial infection, is diagnosed with a bacterial infection, and/or shows symptoms of a bacterial infection.
- treatment and “therapy”/” therapeutic” of bacterial infection can refer to (complete) cure as well as attenuation/reduction of bacterial infection and/or related symptoms (e.g., attenuation/reduction of severity of infection and/or symptoms, number of symptoms, duration of infection and/or symptoms, or any combination thereof).
- a reference subject can be, for example, (i) the same subject during an earlier period of time, (ii) a subject of a same or a similar: age or age group; gender; pregnancy status; chronic medical condition (such as chronic cardiac, pulmonary, renal, metabolic, neurodevelopmental, liver or hematologic diseases) or lack thereof; and/or immunosuppressive condition or lack thereof; or (iii) a typical subject within a population (e.g., local, regional, or national, including of a same or similar age or age range and/or general state of health) during a period of time.
- Prophylaxis can be determined by, for example, the failure to develop a diagnosed bacterial infection and/or the lack of symptoms associated with a bacterial infection over a period of time.
- treatment and/or prevention comprises post-exposure prophylaxis.
- the subject has received, is receiving, or will receive an antibiotic agent.
- the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti- infectives, or any combination thereof.
- the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin- clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof.
- a subject has received is receiving, or will receive, wound drainage, removal of a medical device, or both.
- Typical routes of administering the presently disclosed compositions include, without limitation, oral, topical, transdermal, inhalation, parenteral, sublingual, buccal, rectal, vaginal, and intranasal.
- parenteral includes subcutaneous injections, intravenous, intramuscular, intrasternal injection or infusion techniques.
- administering comprises administering by a route that is selected from oral, intravenous, parenteral, intragastric, intrapleural, intrapulmonary, intrarectal, intradermal, intraperitoneal, intratumoral, subcutaneous, topical, transdermal, intracisternal, intrathecal, intranasal, and intramuscular.
- a method comprises orally administering the antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
- composition to be administered will, in any event, contain an effective amount of an antibody or antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition of the present disclosure, for treatment of a disease or condition of interest in accordance with teachings herein.
- a composition may be in the form of a solid or liquid.
- the carrier(s) are particulate, so that the compositions are, for example, in tablet or powder form.
- the carrier(s) may be liquid, with the compositions being, for example, an oral oil, injectable liquid or an aerosol, which is useful in, for example, inhalatory administration.
- the pharmaceutical composition is preferably in either solid or liquid form, where semi solid, semi liquid, suspension and gel forms are included within the forms considered herein as either solid or liquid.
- the pharmaceutical composition may be formulated into a powder, granule, compressed tablet, pill, capsule, chewing gum, wafer or the like.
- a solid composition will typically contain one or more inert diluents or edible carriers.
- binders such as carboxymethylcellulose, ethyl cellulose, microcrystalline cellulose, gum tragacanth or gelatin; excipients such as starch, lactose or dextrins, disintegrating agents such as alginic acid, sodium alginate, Primogel, com starch and the like; lubricants such as magnesium stearate or Sterotex; glidants such as colloidal silicon dioxide; sweetening agents such as sucrose or saccharin; a flavoring agent such as peppermint, methyl salicylate or orange flavoring; and a coloring agent.
- a liquid carrier such as polyethylene glycol or oil.
- the composition may be in the form of a liquid, for example, an elixir, syrup, solution, emulsion or suspension.
- the liquid may be for oral administration or for delivery by injection, as two examples.
- preferred compositions contain, in addition to the present compounds, one or more of a sweetening agent, preservatives, dye/colorant and flavor enhancer.
- a surfactant, preservative, wetting agent, dispersing agent, suspending agent, buffer, stabilizer and isotonic agent may be included.
- Liquid pharmaceutical compositions may include one or more of the following adjuvants: sterile diluents such as water for injection, saline solution, preferably physiological saline, Ringer’s solution, isotonic sodium chloride, fixed oils such as synthetic mono or diglycerides which may serve as the solvent or suspending medium, polyethylene glycols, glycerin, propylene glycol or other solvents; antibacterial agents such as benzyl alcohol or methyl paraben; antioxidants such as ascorbic acid or sodium bisulfite; chelating agents such as ethylenediaminetetraacetic acid; buffers such as acetates, citrates or phosphates and agents for the adjustment of tonicity such as sodium chloride or dextrose.
- the parenteral preparation can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic.
- Physiological saline is a preferred adjuvant.
- a liquid composition intended for either parenteral or oral administration should contain an amount of an antibody or antigen-binding fragment as herein disclosed such that a suitable dosage will be obtained. Typically, this amount is at least 0.01% of the antibody or antigenbinding fragment in the composition. When intended for oral administration, this amount may be varied to be between 0.1 and about 70% of the weight of the composition. Certain oral pharmaceutical compositions contain between about 4% and about 75% of the antibody or antigen-binding fragment. In certain embodiments, pharmaceutical compositions and preparations according to the present invention are prepared so that a parenteral dosage unit contains between 0.01 to 10% by weight of antibody or antigen-binding fragment prior to dilution.
- the composition may be intended for topical administration, in which case the carrier may suitably comprise a solution, emulsion, ointment or gel base.
- the base may comprise one or more of the following: petrolatum, lanolin, polyethylene glycols, bee wax, mineral oil, diluents such as water and alcohol, and emulsifiers and stabilizers. Thickening agents may be present in a composition for topical administration. If intended for transdermal administration, the composition may include a transdermal patch or iontophoresis device.
- the pharmaceutical composition may be intended for rectal administration, in the form, for example, of a suppository, which will melt in the rectum and release the drug.
- the composition for rectal administration may contain an oleaginous base as a suitable nonirritating excipient.
- bases include, without limitation, lanolin, cocoa butter and polyethylene glycol.
- a composition may include various materials which modify the physical form of a solid or liquid dosage unit.
- the composition may include materials that form a coating shell around the active ingredients.
- the materials that form the coating shell are typically inert, and may be selected from, for example, sugar, shellac, and other enteric coating agents.
- the active ingredients may be encased in a gelatin capsule.
- the composition in solid or liquid form may include an agent that binds to the antibody or antigen-binding fragment of the disclosure and thereby assists in the delivery of the compound. Suitable agents that may act in this capacity include monoclonal or polyclonal antibodies, one or more proteins or a liposome.
- the composition may consist essentially of dosage units that can be administered as an aerosol.
- aerosol is used to denote a variety of systems ranging from those of colloidal nature to systems consisting of pressurized packages. Delivery may be by a liquefied or compressed gas or by a suitable pump system that dispenses the active ingredients. Aerosols may be delivered in single phase, bi phasic, or tri phasic systems in order to deliver the active ingredient(s). Delivery of the aerosol includes the necessary container, activators, valves, subcontainers, and the like, which together may form a kit. One of ordinary skill in the art, without undue experimentation, may determine preferred aerosols.
- compositions of the present disclosure also encompass carrier molecules for polynucleotides, as described herein (e.g., lipid nanoparticles, nanoscale delivery platforms, and the like).
- compositions may be prepared by methodology well known in the pharmaceutical art.
- a composition intended to be administered by injection can be prepared by combining a composition that comprises an antibody, antigen-binding fragment thereof, or antibody conjugate as described herein and optionally, one or more of salts, buffers and/or stabilizers, with sterile, distilled water so as to form a solution.
- a surfactant may be added to facilitate the formation of a homogeneous solution or suspension.
- Surfactants are compounds that non-covalently interact with the peptide composition so as to facilitate dissolution or homogeneous suspension of the antibody or antigen-binding fragment thereof in the aqueous delivery system.
- an appropriate dose and treatment regimen provide the composition(s) in an amount sufficient to provide therapeutic and/or prophylactic benefit (such as described herein, including an improved clinical outcome (e.g., a decrease in frequency, duration, or severity of diarrhea or associated dehydration, or inflammation, or longer disease-free and/or overall survival, or a lessening of symptom severity).
- a dose should be sufficient to prevent, delay the onset of, or diminish the severity of a disease associated with disease or disorder.
- Prophylactic benefit of the compositions administered according to the methods described herein can be determined by performing pre-clinical (including in vitro and in vivo animal studies) and clinical studies and analyzing data obtained therefrom by appropriate statistical, biological, and clinical methods and techniques, all of which can readily be practiced by a person skilled in the art.
- compositions are administered in an effective amount (e.g., to treat a bacterial (e.g., Staphylococcus infection), which will vary depending upon a variety of factors including the activity of the specific compound employed; the metabolic stability and length of action of the compound; the age, body weight, general health, sex, and diet of the subject; the mode and time of administration; the rate of excretion; the drug combination; the severity of the particular disorder or condition; and the subject undergoing therapy.
- a bacterial e.g., Staphylococcus infection
- test subjects will exhibit about a 10% up to about a 99% reduction in one or more symptoms associated with the disease or disorder being treated as compared to placebo-treated or other suitable control subjects.
- a therapeutically effective dose of an antibody or antigen binding fragment is (for a 70 kg mammal) from about 0.001 mg/kg (z.e., 0.07 mg) to about 100 mg/kg (i.e., 7.0 g); preferably a therapeutically effective dose is (for a 70 kg mammal) from about 0.01 mg/kg (i.e., 0.7 mg) to about 50 mg/kg (i.e., 3.5 g); more preferably a therapeutically effective dose is (for a 70 kg mammal) from about 1 mg/kg (i.e., 70 mg) to about 25 mg/kg (i.e., 1.75 g).
- a therapeutically effective dose may be different than for an antibody or antigen-binding fragment.
- the dose may be sufficient to cause endogenous production of the antibody or antigen-binding fragment in a human patient that results, within a specified period of time, in serum concentrations equivalent to that of a therapeutically effective daily dose of the antibody or antigen-binding fragment.
- the specified period of time may be 1, 2, 5, 7, 14, or 30 days from the time of administration.
- the serum concentration equivalent to that of a therapeutically effective daily dose of the antibody or antigen-binding fragment may be maintained for at least 2 weeks, 1 month, 2 months, 3 months, or 6 months, or for at least a time between 2 weeks and 6 months, 2 weeks and 3 months, 2 weeks and 2 months, 2 weeks and 1 month, 1 and 6 months, 1 and 3 months, 1 and 2 months, 2 and 6 months, 2 and 3 months, or 3 and 6 months from the time of administration.
- the polynucleotide may not integrate into the human host cell genome or persist in the human host, such that the antibody or antigen-binding fragment may be undetectable in serum by at least 1, 2, 3, 6, or 12 months from the time of administration.
- Embodiment 70 The polynucleotide of any one of Embodiments 67-69, comprising a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof, wherein, optionally, the polynucleotide comprises a pseudouridine, aN6-methyladenonsine, a 5 -methylcytidine, a 2-thiouridine, or any combination thereof, wherein, further optionally, the pseudouridine comprises Nl- methylpseudouridine .
- Embodiment 82 The composition or combination of Embodiment 81, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are capable of binding to: an LTA and a Gmd, respectively; an LTA and an alpha toxin, respectively; an LTA and a gamma toxin, respectively; an LTA and a leucocidin, respectively; an LTA and a SpA, respectively; an LTA and a ClfA, respectively; an alpha toxin and a Gmd, respectively; an alpha toxin and a gamma toxin, respectively; an alpha toxin and a leucocidin, respectively; an alpha toxin and a SpA, respectively; an alpha toxin and a ClfA, respectively; a gamma toxin and a Gmd, respectively; a gamma toxin and a leucocidin, respectively; a gamma toxin and
- Embodiment 83 The composition or combination of any one of Embodiments 80-82, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment or the second antibody or antigen-binding fragment comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45
- Embodiment 88 The method of making an antibody or antigen-binding fragment of Embodiment 87, wherein the host cell of Embodiment 78 or the human B cell of Embodiment 79 comprises the polynucleotide of any one of Embodiments 67-74 and/or recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77.
- Embodiment 89 The method of Embodiment 87 or 88, further comprising isolating the antibody or antigen-binding fragment.
- Embodiment 90 A method of treating or preventing a bacterial infection in a(n, e.g., human) subject, wherein the bacterial infection is optionally bacteremia, wherein further optionally the bacteremia is hemodialysis associated/line associated bacteremia, the method comprising administering to the subject an effective amount of: (i) the antibody or antigen -binding fragment of any one of Embodiments 1-66; (ii) the polynucleotide of any one of Embodiments 67-74; (iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77; (iv) the host cell of Embodiment 78; (v) the human B cell of Embodiment 79; and/or (vi) the composition or combination of any one of Embodiments 80-86, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma to
- Embodiment 91 The method of Embodiment 90, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'. Aureus (MRSA); (2) a Coagulase-negative Staphylococcus (CoNS); (3) .S', epidermidis,' (4) E.
- MRSA methicillin-resistant .S'. Aureus
- CoNS Coagulase-negative Staphylococcus
- Embodiment 92 The method of Embodiment 90 or 91, comprising administering a single dose of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
- Embodiment 93 The method of any one of Embodiments 90-92, comprising administering two or more doses of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
- Embodiment 94 The method of any one of Embodiments 90-93, comprising administering the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition intramuscularly, subcutaneously, or intravenously.
- Embodiment 95 The method of any one of Embodiments 90-94, wherein the treatment and/or prevention comprises pre-exposure or post-exposure prophylaxis.
- Embodiment 96 The method of any one of Embodiments 90-95, wherein the subject: is using a ventilator; is undergoing, will undergo, or has recently undergone surgery; has a blood stream infection or a history of blood stream infections; is receiving, will receive, or has received a prosthetic joint or other implant; is undergoing, will undergo, or has undergone dialysis; has bacteremia or a history of bacteremia; has neutropenia or a history of neutropenia; has hemodialysis associated/line associated bacteremia or a history of hemodialysis associated/line associated bacteremia; has a wound; or any combination of the foregoing.
- Embodiment 97 The method of any one of Embodiments 90-96, further comprising administering to the subject or more antibiotic agent, or wherein the subject has received one or more antibiotic agent.
- Embodiment 98 The method of Embodiment 97, wherein the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof.
- the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or
- Embodiment 99 The method of Embodiment 97 or 98, wherein the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin-clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof.
- the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levoflox
- Embodiment 100 The antibody or antigen-binding fragment of any one of Embodiments 1- 61a, the polynucleotide of any one of Embodiments 62-66b, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 67-67b, the host cell of Embodiment 68, the human B cell of Embodiment 69, and/or the composition or combination of any one of Embodiments 70-71, for use in a method of treating or preventing a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
- Embodiment 101 The antibody or antigen-binding fragment of any one of Embodiments 1- 66, the polynucleotide of any one of Embodiments 67-74, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77, the host cell of Embodiment 78, the human B cell of Embodiment 79, and/or the composition or combination of any one of Embodiments 80-86, for use in the preparation of a medicament for the treatment or prevention of a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
- Embodiment 102 A method for in vitro diagnosis of a bacterial infection, the method comprising:
- Aureus MRSA
- CoNS Coagulase -negative Staphylococcus
- CoNS Coagulase -negative Staphylococcus
- .S' epidermidis
- E.faecalis E.faecalis
- (5) .S', pyogenes (6) .S', agalactiae,' (7) L. garviae
- Embodiment 104 A library comprising a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprising a plurality of polynucleotides encoding amino acid sequences encoded by any one or more of the following human immunoglobulin genes: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGL
- Embodiment 106 The library of Embodiment 104 or 105, wherein the library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences from any one or more of (i)-(xxiii): (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3- 23,
- Embodiment 107 The library of Embodiment 106, wherein the plurality of antibodies or antigenbinding fragments, or the plurality of polynucleotides encoding the antibodies or antigen-binding fragments, further comprises or encodes additional amino acid sequences, wherein the additional amino acid sequences are optionally randomized and/or are limited to one or more CDR regions of an antibody or antigen-binding fragment.
- Embodiment 108 A method comprising screening one or more antibodies of the library of any one of Embodiments 104-107 for binding to a bacterial (e.g., .S', aureus) antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi.
- LTA lipoteichoic acid
- Gmd glucosaminidase
- ClfA clumping factor A
- SpA Staphylococcus Protein A
- Embodiment 109 A kit comprising a liquid composition comprising and antibody or antigenbinding fragment of any one of Embodiments 1-66, 100, 101, or 103, the polynucleotide of any one of Embodiments 67-74, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77, the host cell of Embodiment 78, the human B cell of Embodiment 79, and/or the composition of any one of Embodiments 80-86, and instructions for use thereof in treating a bacterial (e.g., Staphylococcus) infection in a subject.
- a bacterial e.g., Staphylococcus
- Embodiment 201 An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and/or CDRL3 amino acid sequence(s), or functional variants thereof (and optionally comprising the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, or functional variants thereof), of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: (a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532
- Embodiment 202 An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, and optionally the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences, of one of the following antibodies: (a) SSC35; SSE73; SSA12; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; or SSF69, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA); (b) SSG20; SSF
- Embodiment 203 The antibody or antigen-binding fragment of embodiment 201 or 202, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences are according to: (i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system; (vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs
- Embodiment 204 An antibody or antigen-binding fragment thereof comprising a CDRH3 and a CDRL3, wherein the CDRH3 and the CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (a) (i) 34 and 38, respectively; (ii) 124 and 128, respectively; (iii) 24 and 28, respectively; (iv) 134 and 138, respectively; (v) 144 and 148, respectively; (vi) 154 and 158, respectively; (vii) 164 and 168, respectively; (viii) 244 and 248, respectively; (ix) 174 and 178, respectively; (x) 525 and 529, respectively; (xi) 535 and 539, respectively; (xii) 545 and 549, respectively; (xiii) 555 and 559, respectively; (xiv) 565 and 569, respectively; (xv) 575 and 579, respectively; (xvi) 585 and 589, respectively;
- Staphylococcus Protein A (SpA); (e) (i) 4 and 8, respectively; (ii) 14 and 18, respectively; (iii) 435 and 439, respectively; (iv) 445 and 449, respectively; (v) 455 and 459, respectively; (vi) 465 and 469, respectively; (vii) 475 and 479, respectively; (viii) 485 and 489, respectively; (ix) 495 and 499, respectively; (x) 505 and 509, respectively; or (xi) 515 and 519, respectively; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) (i) 214 and 218, respectively; (ii) 104 and 108, respectively; (iii) 224 and 228, respectively; (iv) 114 and 118, respectively; or (v) 234 and 238, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin
- Embodiment 205 An antibody, or an antigen-binding fragment thereof, comprising a complementarity determining region (CDR)H1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (a) (i) 32-34 and 36-38, respectively; (ii) 122-124 and 126-128, respectively; (iii) 22-24 and 26-28, respectively; (iv) 132-134 and 136-138, respectively; (v) 142-144 and 146-148, respectively; (vi) 152-154 and 156-158, respectively; (vii) 162-164 and 166-168, respectively; (viii) 242-244 and 246-248, respectively; (ix) 172-174 and 176- 178, respectively; (x) 523-525 and 527-529
- Embodiment 206 The antibody or antigen-binding fragment of any one of embodiments 201- 205, comprising a heavy chain variable domain framework region (VHFR)1, a VHFR2, a VHFR3, a light chain variable domain framework region (VLFR)1, a VLFR2, a VLFR3, and/or a VLFR4 (or a variant of the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4 comprising one, two, three, four, or five substitutions, insertions, and/or deletions, or a variant having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2,
- Embodiment 207 The antibody or antigen-binding fragment of embodiment 206, wherein the framework region or regions are according to: (i)the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system; (vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
- the two or more numbering systems combined produce C
- Embodiment 208 The antibody or antigen-binding fragment of any one of embodiments 201- 207, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: (a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and
- Staphylococcus Protein A (SpA); (e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively; (iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
- Embodiment 209 The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a LTA, wherein, optionally, the antibody or antigen-binding binding fragment is capable of binding to: (i) a Type I LTA; (ii) a Type II LTA; (iii) a Type IV LTA; or (iv) any combination of (i)-(iii).
- Embodiment 210 The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding binding fragment is capable of binding to a Gmd, wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a .S'. aureus, to .S'. epide rmidis. or to .S' aureus and to .S' epidermidis .
- Embodiment 211 The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a ClfA.
- Embodiment 212 The antibody or antigen-binding fragment of embodiment 211, wherein the antibody or antigen-binding binding is capable of binding to a ClfA OOl, a ClfA_002, and/or a ClfA_004.
- Embodiment 213 The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA), wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a SPAKKAA-
- SpA Staphylococcus Protein A
- Embodiment 214 The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin, wherein, optionally, the alpha toxin comprises a-hemolysin (also called Hla).
- a-hemolysin also called Hla
- Embodiment 215. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a gamma toxin.
- Embodiment 216 The antibody or antigen-binding fragment of any one of embodiments 201-208 and 215, wherein the antibody or antigen-binding fragment is capable of binding to a leukocidin, wherein, optionally, the leukocidin comprises a LukD, a LukE, a LukF, a LukS, or any combination thereof.
- Embodiment 217 The antibody or antigen-binding fragment of embodiment 216, wherein the antibody or antigen-binding fragment is capable of binding to a LukED, a LukSF, or to a LukED and a LukSF.
- Embodiment 218 The antibody or antigen-binding fragment of any one of embodiments 201-208 and 215-217, wherein the antibody or antigen-binding fragment is capable of binding to a HIgA, a HIgB, or both.
- Embodiment 219. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin.).
- a second immunoglobulin-binding protein also called a Staphylococcal binder of immunoglobulin.
- Embodiment 22 An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
- LTA complementarity determining region
- VH heavy chain variable domain
- VL light chain variable domain
- first antigen-binding domain comprising a first antigen-binding domain and second antigen-binding domain, wherein the first antigen-binding domain or the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; or (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSE52; (xxi) SSG51; (xxii) SSE34
- Embodiment 232 An antibody, or an antigen-binding fragment thereof, which is a multi-specific antibody or antigen-binding fragment thereof, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a
- 232 which comprises or is of a format selected from: a Bispecific T cell Engager (BiTE); a DART; a Knobs-Into-Holes (KIH) assembly; a scFv-CH3-KIH assembly; a KIH Common Light-Chain antibody; a TandAb; a Triple Body; a TriBi Minibody; a Fab-scFv; a scFv-CH-CL-scFv; a F(ab’)2-scFv2; a tetravalent Hcab; an intrabody; a CrossMab; a Dual Action Fab (DAF) (two-in-one or four-in-one); a DutaMab; a DT-IgG, a charge paired antibody; a Fab-arm Exchange antibody, a SEEDbody; a Triomab; a LUZ-Y assembly, an Fcab; a K/.-body: an orthogonal
- the mutation that increases binding affinity to a human FcRn comprises M428L/N434S or M428L/N434A.
- Embodiment 246 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 247 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 248 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 249. The antibody or antigen-binding fragment of any one of embodiments 201-
- amino acid mutation that promotes complement activation comprises or consists of: K236W/E333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L; G236A/Y300L; or G236A/R292P/Y300L.
- Embodiment 250 The antibody or antigen-binding fragment of any one of embodiments 201-
- amino acid mutation that reduces binding by Staphylococcus Protein A comprises or consists of: H435R; Y436F; or H435R/Y436F.
- Embodiment 251 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 252 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 251 which is capable of neutralizing an infection (in vitro, ex vivo, and/or in vivo) by a bacteria expressing the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi.
- Embodiment 253. The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 254 The antibody or antigen-binding fragment of any one of embodiments 201-
- the bacteria comprises .S', aureus.
- Embodiment 255 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 256 The antibody or antigen-binding fragment of any one of embodiments 201-
- Embodiment 257 An antibody, or an antigen-binding fragment, that is capable of binding to an .S', aureus antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi, wherein, optionally:
- LTA lipoteichoic acid
- Gmd glucosaminidase
- ClfA clumping factor A
- SpA Staphylococcus Protein A
- the antibody or antigen-binding fragment binds to a LTA and does not complete with pagibaximab for binding to the LTA;
- the antibody or antigen-binding fragment comprises a VH and a VL that comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.:
- Embodiment 259 The polynucleotide of embodiment 258, wherein the polynucleotide comprises deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), wherein the RNA optionally comprises messenger RNA (mRNA).
- DNA deoxyribonucleic acid
- RNA ribonucleic acid
- mRNA messenger RNA
- Embodiment 260 The polynucleotide of embodiment 259, wherein the polynucleotide comprises self-amplifying RNA (saRNA), trans-amplifying RNA (taRNA), or circular RNA (circRNA).
- saRNA self-amplifying RNA
- taRNA trans-amplifying RNA
- circRNA circular RNA
- Embodiment 261 The polynucleotide of any one of embodiments 258-260, comprising a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof, wherein, optionally, the polynucleotide comprises a pseudouridine, a N6-methyladenonsine, a 5 -methylcytidine, a 2- thiouridine, or any combination thereof, wherein, further optionally, the pseudouridine comprises Nl- methylpseudouridine .
- Embodiment 264 The polynucleotide of embodiment 262, wherein the host cell is a production host cell.
- Embodiment 265. The polynucleotide of embodiment 262, wherein the host cell is an in vivo human host cell.
- Embodiment 266 A recombinant vector, DNA therapeutic, or RNA therapeutic comprising the polynucleotide of any one of embodiments 258-265.
- Embodiment 267 A DNA therapeutic construct or RNA therapeutic construct comprising the polynucleotide of any one of embodiments 258-265 encapsulated in a carrier, wherein the carrier optionally comprises a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
- a lipid-derived delivery vehicle such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or
- Embodiment 268 A recombinant vector comprising the polynucleotide of any one of embodiments 258-265.
- Embodiment 269. A host cell comprising the polynucleotide of any one of embodiments 258-265 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, wherein the polynucleotide is optionally heterologous to the host cell and/or wherein the host cell is capable of expressing the encoded antibody or antigen-binding fragment or polypeptide.
- Embodiment 270 An isolated human B cell comprising the polynucleotide of any one of embodiments 258-265 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, wherein polynucleotide is optionally heterologous to the human B cell and/or wherein the human B cell is immortalized.
- Embodiment 271 A composition or combination comprising: (i) the antibody or antigen-binding fragment of any one of embodiments 201-257; (ii) the polynucleotide of any one of embodiments 258- 265; (iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266- 268; (iv) the host cell of embodiment 269; and/or (v) the human B cell of embodiment 270, and, optionally, a pharmaceutically acceptable excipient, carrier, or diluent.
- Embodiment 272 The composition or combination of embodiment 271, which comprises (1) a first antibody or antigen-binding fragment of any one of embodiments 201-257 and (2) a second, different antibody or antigen-binding fragment of any one of embodiments 201-257.
- Embodiment 273 The composition or combination of embodiment 272, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are capable of binding to: an LTA and a Gmd, respectively; an LTA and an alpha toxin, respectively; an LTA and a gamma toxin, respectively; an LTA and a leucocidin, respectively; an LTA and a SpA, respectively; an LTA and a ClfA, respectively; an alpha toxin and a Gmd, respectively; an alpha toxin and a gamma toxin, respectively; an alpha toxin and a leucocidin, respectively; an alpha toxin and a SpA, respectively; an alpha toxin and a ClfA, respectively; a gamma toxin and a Gmd, respectively; a gamma toxin and a leucocidin, respectively; a gamma toxin and a SpA,
- Embodiment 274 The composition or combination of any one of embodiments 271-273, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment or the second antibody or antigenbinding fragment comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx
- Embodiment 275 The composition or combination of any one of embodiments 271-274, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment and the second antibody or antigenbinding fragment comprise the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S/H435R/Y436F
- Embodiment 276 The composition or combination of embodiment 271, comprising a first polynucleotide and a second polynucleotide, each according to any one of embodiments 258-265, or a recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, encoding two or more antibodies or antigen-binding fragments, wherein the first antibody or antigen-binding fragment encoded by the first polypeptide and second antibody or antigen-binding fragment encoded by the second polypeptide, respectively, or the multispecific antibody or antigen-binding fragment, comprise(s) CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3, and optionally VH and VL, according to any two antibodies of the present disclosure, or any antigen-binding fragments thereof.
- Embodiment 277 A composition comprising the polynucleotide of any one of embodiments 258- 265 or the vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268 encapsulated in a carrier molecule, wherein the carrier molecule optionally comprises a lipid, a lipidderived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
- Embodiment 278 A(n, e.g.
- in vitro or ex vivo method of making an antibody or antigen-binding fragment of any one of embodiments 201-257, comprising culturing the host cell of embodiment 269 or the human B cell of embodiment 270 for a time and under conditions sufficient for the host cell or human B cell, respectively, to express the antibody or antigen-binding fragment.
- Embodiment 279. The method of making an antibody or antigen-binding fragment of embodiment 278, wherein the host cell of embodiment 269 or the human B cell of embodiment 270 comprises the polynucleotide of any one of embodiment 258-265 and/or recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268.
- Embodiment 280 The method of embodiment 278 or 279, further comprising isolating the antibody or antigen-binding fragment.
- Embodiment 28 A method of treating or preventing a bacterial infection in a(n, e.g. , human) subject, wherein the bacterial infection is optionally bacteremia, wherein further optionally the bacteremia is hemodialysis associated/line associated bacteremia, the method comprising administering to the subject an effective amount of: (i) the antibody or antigen-binding fragment of any one of embodiments 201-257;
- Embodiment 282 The method of embodiment 281, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'. Aureus (MRSA); (2) a Coagulase-negative Staphylococcus (CoNS); (3) .S', epidermidis,' (4) E.
- MRSA methicillin-resistant .S'. Aureus
- CoNS Coagulase-negative Staphylococcus
- Embodiment 283 The method of embodiment 281 or 282, comprising administering a single dose of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
- Embodiment 284 The method of any one of embodiments 281-283, comprising administering two or more doses of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
- Embodiment 285. The method of any one of embodiments 281-284, comprising administering the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition intramuscularly, subcutaneously, or intravenously.
- Embodiment 286 The method of any one of embodiments 281-285, wherein the treatment and/or prevention comprises pre-exposure or post-exposure prophylaxis.
- Embodiment 287 The method of any one of embodiments 281-286, wherein the subject: is using a ventilator; is undergoing, will undergo, or has recently undergone surgery; has a blood stream infection or a history of blood stream infections; is receiving, will receive, or has received a prosthetic joint or other implant; is undergoing, will undergo, or has undergone dialysis; has bacteremia or a history of bacteremia; has neutropenia or a history of neutropenia; has hemodialysis associated/line associated bacteremia or a history of hemodialysis associated/line associated bacteremia; has a wound; or any combination of the foregoing.
- Embodiment 288 The method of any one of embodiments 281-287, further comprising administering to the subject or more antibiotic agent, or wherein the subject has received one or more antibiotic agent.
- Embodiment 289. The method of embodiment 288, wherein the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof.
- the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof.
- Embodiment 290 The method of embodiment 288 or 289, wherein the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin-clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof.
- the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin
- Embodiment 291 The antibody or antigen-binding fragment of any one of embodiments 201- 257, the polynucleotide of any one of embodiments 258-265, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, the host cell of embodiment 269, the human B cell of embodiment 270, and/or the composition or combination of any one of embodiments 271-277, for use in a method of treating or preventing a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S', aureus.
- Embodiment 292 The antibody or antigen-binding fragment of any one of embodiments 201- 257, the polynucleotide of any one of embodiments 258-265, the recombinant vector, DNA therapeutic, RNA or therapeutic of any one of embodiments 266-268, the host cell of embodiment 269, the human B cell of embodiment 270, and/or the composition or combination of any one of embodiments 271-277, for use in the preparation of a medicament for the treatment or prevention of a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
- Embodiment 293. A method for in vitro diagnosis of a bacterial infection, the method comprising: (i) contacting a sample from a subject with an antibody or antigen-binding fragment of any one of embodiments 201-257; and (ii) detecting a complex comprising an antigen and the antibody, or comprising an antigen and the antigen-binding fragment, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
- Embodiment 294 The antibody, antigen-binding fragment, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, human B cell, or composition for use of embodiment 291 or 292, or the method of embodiment 293, wherein wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'.
- Aureus MRSA
- CoNS Coagulase -negative Staphylococcus
- CoNS Coagulase -negative Staphylococcus
- .S' epidermidis
- E. faecalis E. faecalis
- (5) .S', pyogenes E. faecalis
- (6) .S' pyogenes
- (6) .S' agalactiae
- L. garviae E.
- Embodiment 295. A library comprising a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprising a plurality of polynucleotides encoding amino acid sequences encoded by any one or more of the following human immunoglobulin genes: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGL
- Embodiment 296 The library of embodiment 295, wherein the human immunoglobulin genes in the library consist essentially of, or consist of, any one or more of the human immunoglobulin genes of embodiment 295.
- Embodiment 297 The library of embodiment 295 or 296, wherein the library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences from any one or more of (i)-(xxiii): (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3- 23, IGHJ4,
- Embodiment 298 The library of embodiment 297, wherein the plurality of antibodies or antigenbinding fragments, or the plurality of polynucleotides encoding the antibodies or antigen-binding fragments, further comprises or encodes additional amino acid sequences, wherein the additional amino acid sequences are optionally randomized and/or are limited to one or more CDR regions of an antibody or antigen-binding fragment.
- Embodiment 299. A method comprising screening one or more antibodies of the library of any one of embodiments 104-107 for binding to a bacterial (e.g. , .S', aureus) antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi.
- LTA lipoteichoic acid
- Gmd glucosaminidase
- ClfA clumping factor A
- SpA Staphylococcus Protein A
- Embodiment 300 A kit comprising a liquid composition comprising and antibody or antigenbinding fragment of any one of embodiments 201-257, 291, 292, or 294, the polynucleotide of any one of embodiments 258-265, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, the host cell of embodiment 269, the human B cell of embodiment 270, and/or the composition of any one of embodiment 271-277, and instructions for use thereof in treating a bacterial (e.g., Staphylococcus) infection in a subject.
- a bacterial e.g., Staphylococcus
- Monoclonal antibodies (mAbs) reactive against one or more Staphylococcus antigen were identified from a human tonsil bank and from PBMCs from human donors recovered from S. aureus, as shown and described for Figures 1A-1D.
- MAbs were cloned, recombinantly expressed (in some contexts, carrying one or more Fc mutation), and characterized for binding specificity, neutralization, and other functions, as shown and described for Figures 2A-6C.
- opsonizing antibodies include anti-LTA antibodies, anti-Gmd antibodies, anti-ClfA antibodies, and anti-SpA antibodies.
- Neutralizing antibodies include anti- Hla antibodies and anti-bicomponent (Luk, HIgAB) antibodies.
- anti-LTA antibodies further experiments assess breadth of binding to live bacteria (FACS) and epitope-binning.
- anti-Gmd antibodies further experiments assess breadth of binding to different strains of Staphylococcus, epitope-binning, and effect on Gmd enzymatic activity.
- anti-ClfA antibodies opsonophagocytic killing and fibrinogen inhibition of binding are assessed.
- anti-Hla antibodies synergism of Hla neutralization and neutralization on THP1 cells are assessed.
- anti-bicomponent toxins neutralization studies are performed.
- mice are used to study bacteremia, pneumonia, dermonecrosis, a prosthetic joint infection model, a foreign body model, and a surgical site infection model.
- a foreign body model beads coated with 500 CFU of S. aureus are injected into the skin, representative of a real-life infection dose.
- S. aureus introduced on a foreign object is not cleared as easily as suspension cells and presents as biofilm-like.
- a surgically placed stainless steel K-wire implant can be inserted into the knee joints of mice and inoculated with S. aureus to model a prosthetic joint infection.
- Figure 7A shows results from SSE1 and SSE158 binding and neutralization studies, with MEDI4893 included as a comparator.
- SSE1 and SSE158 bind to H1A and neutralize H1A activity on rabbit red blood cells (“RBCs”) and THP-1 cells.
- Figure 7B shows results (neutralization of infection) from a synergy/antagonism study with SSE1 and SSE158. SSE1 and SSE158 did not show synergistic neutralization in this assay.
- Figure 7C shows that SSE1 and SSE158 protect BALB/c mice in an in vivo model of S. aureus pneumonia.
- the experimental set-up was as shown in the schematic in the bottom of Figure 7C. Percent survival was measured post-infection.
- Figures 8A-8B show binding (ELISA) to Gmd S. aureus and Gmd S. epidermis, with 1C11 included as a comparator.
- Figure 8C shows quantified binding EC50 values to Gmd S. aureus and Gmd S. epidermis,' to strains of S. aureus, S. epidermidis, and S. lugdunensis,' and V- domain gene usage of the indicated antibodies.
- Figures 9A-9B relate to neutralization of LTA activity on TLR-2-expressing cells by certain antibodies of the present disclosure.
- HEK-Blue hTLR2 cells were co-transfected with hTLR2 and SEAP (secreted embryonic alkaline phosphatase) reporter genes under the control of the IFN-b minimal promoter fused to NF-kB and AP-l-binding sites. Stimulation of TLR2 activates NF-kB and AP-1 which induces the production of SEAP. The hydrolysis of the substrate in the medium by SEAP produces an easily detected purple/blue color.
- Figure 10A shows binding (ELISA) by certain antibodies of the present disclosure to strains of S. aureus with different ClfA genotypes (ClfA_001, ClfA_002, and ClfA_004), with E. coli included as a negative control.
- Figure 10B shows quantified binding EC50 values to ClfA_001, ClfA_002, ClfA_004, and strains of S. aureus,' binding affinity values (Kato ClfA OOl); inhibition of ClfA binding to fibrinogen (IC50); and V-domain gene usage of certain antibodies of the present disclosure. Tefibazumab was included as a comparator.
- Figure 10C shows inhibition of binding between ClfA and fibrinogen (Fg) by certain antibodies of the present disclosure, with tefibazumab included as a comparator.
- Figure 10D shows sequence alignments between ClfA_001, ClfA_002, and ClfA_004 genotypes and a schematic of a representative recombinant ClfA expression construct.
- Figure 10E shows inhibition of binding between ClfA and Fg by certain antibodies of the present disclosure.
- Figure 11A shows binding (EC50 as determined by ELISA) to SpA-KKAA, percent inhibition of SpA binding to IgG, binding affinity values of antibodies of the present disclosure to Spa-KKAA (Ka), V-domain gene usage, and epitope binning of certain antibodies of the present disclosure.
- Figure 11B shows neutralization of binding between SpA and IgGl by the indicated antibodies of the present disclosure (Octet).
- Figure 11B shows antibody neutralization of Protein A binding of IgGs assessed using BLI (Octet).
- FIG. 18A shows a schematic of Sbi structure.
- Figure 18B shows (top) binding quantified by ELISA and (bottom) quantified binding values (EC50), percent IgG-binding inhibition, binding affinity (KD), and V-domain gene usage of SSH4 and SSH7.
- Figure 18C shows sensorgram curves showing that antibodies SSH4 and SSH7 of the present disclosure do not compete with one another for Sbi binding.
- anti-LTA, anti-ClfA, anti-SpA, and anti-Gmd mAbs were administered to 8-week-old female C57BL/6 mice at 10 mg/kg intravenously 2 days prior to challenge with 2xlO A 7 cfu of S. aureus strain FPR3757 via retroorbital injection (Figure 25). Mice were then monitored for weight loss and kidney burden was determined 4 days post infection by organ homogenization and serial dilution.
- mice As a control, one group was treated with daptomycin (50 mg/kg subcutaneously) at days 2 and 3 post infection to compare the relative weight reduction and kidney burden of antibiotics in a treatment model. Additional control groups included mice dosed with non-binding isotype control mAbs and a histidine buffer group. Mouse serum Ig level was measured at day 1 post-dosing using meso scale discovery (goat antihuman IgG as capture antibody, sulfo tag anti-human CH2 as detection antibody; Figure 26). 514G3 RF antibody had 2.3-fold lower serum Ig level compared to isotype control, similar to results for 514G3 IgG3 antibody format. SSF11 antibody had 3-folder lower serum Ig level compared to isotype control.
- FIGS 27A-27B show kidney burden at 4 days post infection for individual mice (Figure 27 A), weight loss over time and the weight loss calculated from the area under the curve (Figure 27B). Mice treated with isotype control antibodies had uniformly high kidney burden with pronounced weight loss, while daptomycin treated mice showed a 1-3 log reduction in S. aureus kidney burden associated with a minor reduction in weight loss. Individual mice treated with anti-Staphylococcal antibodies varied from no reduction to 6 log reduction in kidney burden ( Figure 28).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Oncology (AREA)
- Animal Behavior & Ethology (AREA)
- Communicable Diseases (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
The instant disclosure provides antibodies and antigen-binding fragments thereof that can bind to a bacterial antigen selected from a lipoteichoic acid (LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; or a Sbi. The antigen can be an antigen that is expressed by Staphylococci (e.g., S. aureus) and, in certain embodiments, by one or more other bacteria. Also provided are polynucleotides that encode an antibody or antigen-binding fragment, vectors that comprise such polynucleotides, host cells that can express the antibodies, related compositions, and methods of using the herein disclosed compositions to, for example, treat or prevent a bacterial infection.
Description
ANTIBODIES AGAINST STAPHYLOCOCCUS ANTIGENS AND METHODS OF USING THE SAME
REFERENCE TO AN ELECTRONIC SEQUENCE LISTING
The contents of the electronic sequence listing (368564_44802_SL,xml; Size 692,224 bytes; Date of Creation: June 28, 2024) is herein incorporated by reference in its entirety.
BACKGROUND
Gram-positive bacteria, such as of the genus Staphylococcus, cause infections leading to significant morbidity and mortality. Staphylococcus aureus (S. aureus), for example, causes diverse infections that often overlap with Enterococci and coagulase-negative staphylococci (CoNS), including skin/soft tissue infections, bacteremia/sepsis, hospital -associated pneumonia, endocarditis, neutropenia, hemodialysis associated/line associated bacteremia, osteomyelitis/joint infections, menstrual toxic shock syndrome, and peritonitis. Currently, antibiotics are the only option for treatment, which has been limited by antibiotic resistance. Attempted vaccines and monoclonal antibodies to-date have not been protective.
For example, S. aureus Protein A can prevent serum antibodies and IgGl opsonizing monoclonal antibodies from harnessing Fc-mediated immunity. Protein A immune evasion may thwart development of a humoral-based vaccine for S. aureus. The lack of an effective vaccine for S. aureus and related Gram-positive bacteria leads to significant morbidity and mortality. There is an unmet need for modalities to prevent and/or treat infection by Staphylococcus and related Gram-positive bacteria.
BRIEF DESCRIPTION OF THE DRAWINGS
In the drawings, the following Figures are identified as “Fig.”
Figures 1A-1D show a workflow for discovery, recombinant expression, and characterization of antigen-specific monoclonal antibodies from human donors who recovered from S. aureus infection, and examples of results from the workflow.
Figures 2A-2P relate to antibody targeting of lipoteichoic acid (LTA). Figure 2A shows (left) a table summarizing types of LTA and bacterial species expressing the indicated LTA is found, and (right) a schematic illustration of LTA types (I-V) at a cell membrane). Other abbreviations: Gro = glycerol, Glc = glucose, Gal = galactose, P-Glu = P-glucosamine, AATGal = 2-acetamido-4-amino-2,4,6-trideoxy-D-galactose, GalNAc = N-acetylgalactosamine, Rto =
ribitol, GlcNAc = Nacetylglucosamine, P = phosphate. Figure 2B provides a table showing binding (quantified as EC50 ng/ml) of certain anti-LTA antibodies of the present disclosure (SSI18-SSA8) to bacteria expressing Type I, Type II, or Type IV LTA (E coli was included as a reference), and variable domain gene usage of these antibodies. The humanized anti-LTA antibody pagibaximab (e.g., Patel and Kaufman, Expert Opin Biol Ther 75(4):595-600 (2015); doi: 10.1517/14712598.2015.1019857) was included as a comparator. In a Phase III clinical study, pagibaximab did not reduce sepsis or death. Figure 2C shows sensorgram curves showing that antibody SSA12 of the present disclosure does not compete with pagibaximab for LTA binding. Figure 2D shows that SSA12 has more consistent binding across Type I LTA bacteria than pagibaximab. Figure 2E shows binding (ELISA on LTA) of certain anti-LTA antibodies of the present disclosure, with pagibaximab as a comparator. Figure 2F shows binding (EC50 ng/mL on LTA), gene usage, and IMGT CDRH3 amino acid sequence and length of the anti-LTA antibodies. VH3-23 is used by 4 monoclonal antibodies produced by 3 donors. VH3-7/VK1-6 are used by 10 clonally related monoclonal antibodies produced by 3 donors. Figure 2G shows binding (ELISA on fixed bacteria) by certain anti-LTA antibodies of the present disclosure, with pagibaximab as a comparator. Figure 2H shows quantified binding by the antibodies to bacteria expressing Type I, Type II, or Type IV LTA, or E. coli. Figure 21 shows (left) FACS data showing binding by SSA12 to the indicated bacteria and (right) affinity for LTA, as compared to pagibaximab. Figure 2J shows neutralization of infection on TLR2- expressing HEK293 cells. The foregoing data show that SSA12 is a broadly reactive anti-LTA antibody that binds with high affinity to a site on LTA that is not recognized by pagibaximab and can neutralize LTA activity. Figure 2K shows a schematic of signaling initiated by binding of LTA to TLR2. Figure 2L shows neutralization of LTA activity on TLR2-expressing HEK293 cells by certain anti-LTA antibodies of the present disclosure, and by pagibaximab. Briefly, HEK-Blue hTLR2 cells were co-transfected with hTLR2 and SEAP (secreted embryonic alkaline phosphatase) reporter genes under the control of the IFN-b minimal promoter fused to NF-kB and AP-l-binding sites. Stimulation of TLR2 activates NF-kB and AP-1 which induces the production of SEAP. The hydrolysis of the substrate in the medium by SEAP produces an easily detected purple/blue color. Figure 2M shows (left) a schematic for a Cl -binding assay and (right) binding to Cl by certain opsonized Fc engineered anti-LTA antibodies of the present disclosure. Pagibaximab and “S2X303-LS”, a non-targeting negative control, were used as comparators. “LS” = M428L/N434S mutations in Fc (increasing affinity for FcRn and in vivo half-life). “RF” = H435R/Y436F mutations in Fc (reducing binding to Protein A). Accordingly,
“LS-RF” = M428L/N434S/H435R/Y436F, wherein Fc amino acid numbering is according to the EU numbering system, with reference to human IgGl . Pagibaximab does not avoid Protein A binding. These data show that Fc-engineered anti-LTA antibodies of the present disclosure, opsonized on Staphylococci, demonstrate increased binding to complement as compared to pagibaximab. Figure 2N shows (left) a schematic for a Cl -binding assay and (right) binding to Cl by opsonized SSA12 bearing either LS or LS/RF mutations in the Fc. Pagibaximab and “S2X303 -LS-RF”, a non-targeting negative control, were used as comparators. Antibodies were opsonized on S. aureus (which expresses Protein A) or S. epidermidis (which is Protein A- deficient). These data show that Protein A blocks Cl binding to IgGl, and introduction of IgG3 “RF” residues (via H435R/Y436F mutations) to IgGl Fc restores Cl binding. Figure 20 shows (left) a schematic for a Cl-binding assay and (right) binding to Cl by opsonized SSA12 expressed as IgG3, or as IgGl with LS + RF mutations in Fc. Pagibaximab and S2X303-LS-RF were used as comparators. Antibodies were opsonized on S. aureus (which expresses Protein A) or S. epidermidis (which is Protein A-deficient). These data show that Protein A interferes with Cl binding and IgG3 overcomes impact on Cl binding when Protein A is present. Figure 2P shows (left) Cl binding by SSA12 variant antibodies (pagibaximab and S2X303-LS-RF included as comparators) on S. epidermidis and (right) Cl binding AUC with S. epidermidis versus with S. aureus. Abbreviations for Fc mutations (EU numbering per human IgGl): KWES = K236W/E333S; KAEA = K326A/E333A; EFTAE = S267E/H268F/S324T/G236A/I332E.
RPYL = R292P/Y300L. GARPYL = G236A/R292P/Y300L. GAYL = G236A/Y300L. Fc amino acid numbering is according to the EU numbering system, with reference to human IgGl. For KWES, KAEA, and EFTAE mutations, see, e.g., Moore et al.,MAbs 2(2): 181-189 (2010); doi: 10.4161/mabs.2.2.11158 ; PMID: 20150767. For RPYL, GARPYL, and GAYL mutations, see PCT Publication WO 2022/251119.
Figure 3 relates to antibody targeting of glucosaminidase. By way of background, glucosaminidase is a peptidoglycan-modifying enzyme that is highly conserved among Staphylococci and is necessary for cell division. Glucosaminidase (Gmd or GlcA) is a domain of the autolysin (Atl) protein, the most predominant peptidoglycan hydrolase in S. aureus. Gmd is located on the cell surface and is highly conserved among S. aureus and S. epidermidis. Gmd catalyzes the hydrolysis of glucosidic linkages and plays a role in cell wall turnover, division, and separation. Gmd KO bacterial cells divide along an asymmetric plane, do not fully separate and form clusters instead, and cannot form biofilms. See, e.g., Nega et al. Commun Biol 3(1): 695 (2020); doi: 10.1038/s42003-020-01405-2. Both human patients and mice have been
found to produce anti-Gmd antibodies in response to S. aureus infection. Clinical studies of patients with S. aureus osteomyelitis found that for each 10X increase in anti-Gmd antibody concentration there is a 60% reduction in adverse event risk. Anti-Gmd antibody treatments have been shown to synergize with the standard of care antibiotic vancomycin in vivo. In an S. aureus-m cted implant model in mice, treatment with the anti-Gmd antibody 1C11 led to a significant decrease in osteolysis and abscess formation. Treatment also facilitated macrophage entry into abscesses and eventual opsonophagocytosis and internalization of S. aureus megaclusters. Figure 3 shows (right) a schematic illustration of a Gmd neutralization assay and (left, middle) results from ELISA binding studies using antibody SSG20 of the present disclosure (with 1C11 as comparator) against Gmd (left) and fixed bacteria (middle).
Figures 4A-4F relate to antibody targeting of alphatoxin or bicomponent toxins. By way of background, Staphylococcus secretes two types of P-barrel pore-forming toxins: 1. Alpha toxin; 2. Bicomponent (LukSF, LukED, LukAB, HlgAB and HlgCB). These toxins cause disruption of the plasma membrane, leading to osmotic imbalance and cell death, and are highly inflammatory. The vast majority of S. aureus clinical isolates express Hla, HlgABC, and Luk. The Luk S- and F-components are highly related structurally and share up to 80% amino acid identity. Alpha toxin is a significant contributor to pathogenesis in some S. aureus infections, including skin and soft tissues infections and pneumonia. Studies have shown that in vitro Hla activity of MRS A strains obtained from patients with bacteremia is significantly associated with increased risk for thrombocytopenia and death. An anti-alpha toxin antibody “MEDI4893” aka “AR-302” recently completed a Phase II clinical trial for prevention of ventilator-associated pneumonia (VAP). Figures 4A and 4B show binding and neutralization by anti-alpha-toxin antibodies “SSE1” and “SSE158” of the present disclosure, with MEDI4893 as a comparator. For the neutralization studies, a negative control antibody was also included. Figures 4C-4F show: (Figure 4C) a schematic activity of bicomponent pore-forming cytotoxins (PFTs) at a cell membrane; (Figure 4D) binding (ELISA, SPR (Octet)) by certain antibodies of the present disclosure against Luk and HIg antigens; (Figure 4E) toxin neutralization on human monocytes; and (Figure 4F) binding competition assays. For the experiments shown in Figures 4E and 4F, AB28-10 (also known as AB-28-10, see U.S. Pre-Grant Publication No. US 2018/0179267 Al) was used as a comparator.
Figures 5A and 5B relate to antibody targeting of Protein A. By way of background, Protein A is a highly conserved surface protein expressed by approximately 90% of clinical isolates. Protein A is able to bind human IgGl, IgG2, and IgG4, but not IgG3. Binding to
immunoglobulin mediates immune evasion, preventing normal phagocytosis and inducing B cell death. Figure 5A shows a schematic of Protein A, which includes 5 IgG binding domains (E, D, A, B, C). A variant of SpA Protein A (SPAKKAA) contains substitution mutations in each of the five Ig-binding domains (Q9K, Q10K, D36A, D37A), abolishing the ability to bind Fey or Fab VH3 and promote B cell apoptosis (see Kim et al. J Exp Med, 207(9): 1863-1870 (2010); doi: 10.1084/jem.20092514). Figure 5B shows (top) baiting of SPAKKAA memory B cells sorting and (bottom) binding (ELISA; SPR (Octet)) by certain antibodies of the present disclosure.
Figures 6A-6C relate to targeting of Clumping factor A (ClfA) by cross-reactive antibodies. By way of background, S. aureus and S. epidermidis express sortase-attached SD repeat (SDR) proteins. Clumping factor (Clf) promotes binding of fibrinogen to the bacterial cell and facilitates bacterial clumping. Serine aspartate repeat proteins (Sdr) also promote bacterial adhesion. Figure 6A shows a schematic of domains of Clf and Sdr proteins of S. aureus and S. epidermidis. Figure 6B shows binding (ELISA) by certain antibodies of the present disclosure to ClfA. Figure 6C shows quantified binding EC50 values, V-domain gene usage, and CDRH3 amino acid sequence and length of certain antibodies. For the experiments shown in Figures 6B and 6C, “SaHul2-9” (also called “humanized 12-9”; see U.S. Patent No. 7,364,738 B2) was used as a comparator.
Figures 7A-7C relate to antibody targeting of alpha-toxin. Figure 7A shows (left graph) binding and (two graphs at right) neutralization by anti-alpha-toxin antibodies SSE1 and SSE158 of the present disclosure, with MEDI4893 as a comparator. MEDI4893 (also known as AR-320) is an anti-Hla mAb that has been tested in a Phase 2 study for prevention of ventilator-associated pneumonia. Figure 7B shows results from a synergy/antagonism neutralization study combining anti-alpha-toxin antibodies SSE1 and SSE158. Figure 7C shows survival of BALB/c mice prophylactically administered SSE1, SSE158, and/or comparator MEDI4893 at the indicated dose followed by infection (i.n.) with S. aureus. The combination study is repeated and in silica analysis of SSE1 and SSE158 is performed.
Figures 8A-8C relate to targeting of Gmd by certain antibodies. Figures 8A-8B show results from ELISA binding studies using certain anti-Gmd antibodies of the present disclosure against Gmd from S. aureus and S. epidermidis. Figure 8C provides a table showing quantified binding of certain anti-Gmd antibodies of the present disclosure to Gmd (S. aureus and S. epidermidis), quantified binding to bacterial strains, and variable domain gene usage of these antibodies. 1C11 was included as a comparator.
Figures 9A and 9B relate to antibody targeting of LTA. (Figure 9A) LTA neutralization (reported as absolute EC50 ng/mL) for the antibodies shown in Figure 2F. (Figure 9B) Neutralization of LTA activity on TLR-2-expressing HEK293 cells by certain anti -LTA antibodies of the present disclosure, and by pagibaximab, was assessed using the assay discussed in the brief description of Figure 2L, above.
Figures 10A-10F relate to targeting of ClfA by certain antibodies. Figure 10A shows binding by certain anti -ClfA antibodies of the present disclosure across strains of S. aureus with different ClfA genotypes, with E. coli included as a negative control. The humanized anti-ClfA antibody tefibazumab (e.g., Weems JJ Jr, Steinberg JP, Filler S, et al. Antimicrob Agents Chemother. 2006;50(8):2751-2755. doi: 10.1128/AAC.00096-06) was included as a comparator. Tefibazumab has been tested in a Phase 2 study for treatment of S. aureus in cystic fibrosis patients and treatment of S. aureus bacteremia. Figure 10B shows (top) quantified binding values (EC50 ng/mL to ClfA_001, ClfA_002, ClfA_004, and S. aureus strains), quantified affinity values (Kdfor ClfA OOl), quantified inhibition of binding to fibrinogen values (absolute IC50 ng/mL), (bottom left) neutralization against E. coli and (bottom right) V-domain gene usage of certain antibodies, with tefibazumab included as a comparator. Figure 10C shows (leftmost) a schematic of ClfA binding fibrinogen (Fg), (second-left) antibody-mediated disruption of the binding interaction, and (right) inhibition of fibrinogen binding by certain antibodies of the present disclosure, with tefibazumab included as a comparator. Figure 10D shows (top) amino acid sequence alignments of ClfA_001, ClfA_004, and ClfA_002 genotypes and (bottom) a schematic of a recombinant ClfA expression construct. The ClfA genotypes were selected to represent the most divergent genotypes and ClfAs were expressed, using the schematized expression construct, for binding inhibition studies. Briefly, experimental conditions were: coating fibrinogen (10 ug/ml; Dunn Labortechnik) in PBS; blocking with 1% casein in PBS; mix of ClfA (7.5 ug/mL) + serial dilutions of anti-ClfA antibody (from 150 ug/mL); development (Strep-Tacting AP). Figure 10E shows that certain antibodies of the present disclosure inhibit binding between ClfA and fibrinogen, quantified by percent inhibition. Figure 10F shows antibodies grouped according to like binding profiles as in Figure 10B (top).
Figures 11A and 11B relate to antibody binding against Protein A. Figure 11A provides a table showing binding (quantified as EC50 ng/mL to SpA-KKAA), percent inhibition of Protein A binding to IgG, binding affinity (quantified as Kd), V-domain gene usage, and epitope data for certain antibodies of the present disclosure. Monoclonal antibody “514G3” (e.g., Varshney AK, Kuzmicheva GA, Lin J, et al. PLoS One. 2018;13(l):e0190537. Published 2018
Jan 24. doi: 10.1371/journal. pone.0190537) was included as a comparator. 514G3 is an anti-SpA antibody that has been tested in a Phase 2 study for treatment of bacteremia. Figure 11B shows antibody neutralization (SPR(Octet)) of Protein A binding to IgGl by certain antibodies of the present disclosure, with a negative control antibody, “KB”, included. These studies identified several antibodies that bind to SpA-KKAA with EC50 ~5-15ng/mL, are clonally related, and recognize different epitopes on SpA-KKAA. Eight (8) antibodies showed inhibition of >75%) of Protein A binding to Fc of IgG.
Figures 12A and 12B relate to human whole blood assays. Figure 12A shows a schematic for a human whole blood assay of bacterial survival when exposed to antibody. Figure 12B shows percent survival of S. aureus in whole blood from donors after exposure to certain antibodies of the present disclosure.
Figure 13 shows a graph comparing half-life of antibodies “FYl-rlgGl-LS” and “FY1- rlgGl-LS-RF” in Tg32 SCID mice expressing human FcRn. FYI recognizes influenza HA.
Figures 14A-14K relate to Fc effector function modifications made in antibody “SSA12- LS-RF”. SSA12 is an antibody of the present disclosure. “LS” = M428L/N434S mutations in Fc (increasing affinity for FcRn and in vivo half-life). “RF” = H435R/Y436F mutations in Fc (reducing binding to Protein A). Accordingly, “LS-RF” = M428L/N434S/H435R/Y436F, wherein Fc amino acid numbering is according to the EU numbering system, with reference to human IgGl. The Fc modifications indicated in Figures 14A-14K are afucosylation or are additional Fc mutations made to the SSA12-LS-RF heavy chain sequence according to the EU numbering system with reference to human IgGl. Abbreviations for Fc mutations (EU numbering per human IgGl): EFTAE = S267E/H268F/S324T/G236A/I332E; GRLR = G236R/L328R. “Wild-type” indicates the SSA12-LS-RF antibody, “afuc” indicates an antibody expressed under conditions that inhibit fucosylation. “IgG3” indicates a recombinant SSA12 antibody expressed as an IgG3 isotype. “1” = FcyRI; “2AH” = FcyRIIa H131; “2AR” = FcyRIIa R131; “2B” = FcyRIIb; “3 AV” = FcyRIIIa V158; “3AF” = FcyRIIIa F158; “3B” = FcyRIIIb. “S. epi” = S. epidermidis and “S. aureus” = S. aureus. Figure 14A provides (a) FcyR binding quantified by SPR and (b) Cl binding on S. epidermidis and S. aureus by SSA12-LS-RF (or variant) antibodies. The values provided show a Log2 fold change compared to SSA12-LS-RF binding. SPR values for antibodies bearing G236A-Y300L or G236A-R292P-Y300L Fc mutations were taken from an assay wherein those mutations were present in an anti-hepatitis B virus antibody. For cells with an “X”, no values were available. Figure 14B shows (left) a schematic of FcyR binding measured by SPR and (right) a table showing FcyR binding by the
indicated SSA12-LS-RF (or variant) antibodies, quantified by SPR. The values provided show a Log2 fold change compared to SSA12-LS-RF binding. Figure 14C shows (left) a schematic of FcyR binding measured by Meso Scale Discovery (MSD; Meso Scale Diagnostics, LLC) and (right) a table showing FcyR binding by SSA12-LS-RF (or variant) antibodies quantified by MSD. The values provided show a Log2 fold change compared to SSA12-LS-RF binding. Figure 14D shows correlation between SPR (Figure 14B) and MSD (Figure 14C) datasets. Figure 14E shows (left) a schematic of Clq binding measured by BLI and (right) a table showing Clq binding by SSA12-LS-RF (or variant) antibodies quantified by BLI. The values provided show a Log2 fold change compared to SSA12-LS-RF binding. Figure 14F shows (left) a schematic of Clq binding measured by ELISA and (right) a table showing Clq binding on S. epidermidis and S. aureus by SSA12-LS-RF (or variant) antibodies quantified by ELISA. The values provided show a Log2 fold change compared to SSA12-LS-RF binding. Figure 14G shows correlation between BLI (Figure 14E) and ELISA (Figure 14F) datasets. Figure 14H shows (left) a schematic of a bacterial survival assay wherein antibody and bacteria were combined in whole blood and then bacteria were plated, and (right) percent survival of S. epidermidis in whole blood assays after exposure to SSA12-LS-RF (or variant) antibodies. Each dot represents the median value of an experiment with one donor performed in triplicate. Mann- Whitney t-test was used to determine significance. Figure 141 shows Staphylococcus survival in a whole blood assay after exposure to isotype control (S2X303-LS-RF) and SSA12-LS-RF (or variant) antibodies, tested by donor. In each graph, Staphylococcal survival for isotype control and the tested mAb is shown for each donor. Each line represents the change in survival with addition of the tested mAb. Figure 14J shows (left) percent survival of S. aureus, normalized to isotype control (anti-SARS-CoV-2 antibody S2X303 with LS-RF Fc) in whole blood assays after exposure to SSA12-LS-RF (or variant) antibodies of the present disclosure and (right) S. aureus survival in whole blood assays after exposure to SSA12-LS-RF (or variant) antibodies, tested by donor. In each graph, survival for isotype control and the tested mAb is shown for each donor. Each line represents the change in survival with addition of the tested mAb. Figure 14K shows (left) a principal component analysis (PCA) of FcyR binding quantified by SPR and Clq binding quantified by BLI and (right) a table showing antibodies of the present disclosure which decreased survival of S. epidermidis in a whole blood assay.
Figure 15A (top) shows a schematic of Clq binding measured by ELISA, (bottom) a table showing fold-change in human Cl (hCl) and mouse Clq binding (mClq) binding by SSA12-LS-RF (or variant) antibodies quantified by ELISA.
Figure 15B (left) human Cl binding by the indicated antibodies with S. aureus, and (right) mouse Clq binding by the indicated antibodies with S. aureus. S2X303-LS-RF was included as an isotype control. “LS-RF” = SSA12-LS-RF. “SEST” = S267E/S324T. The Fc modifications indicated in Figure 16 are additional Fc mutations made to the SSA12-LS-RF heavy chain sequence according to the EU numbering system with reference to human IgGl .
Figures 16A and 16B relate to disseminated S. aureus infection models. Figure 16A shows a disseminated S. aureus infection model in BL/6 mice with isotype control (S2X303 9MPK IgGl LS-RF) and histidine buffer conditions. Figure 16B shows a disseminated S. aureus infection model in mice engineered to express human FcyRs with isotype control (S2X303-rIgGlm3-LS-RF) and histidine buffer conditions.
Figures 17A and 17B provide information further to the data shown in Figures 8B and 2B, respectively. In Figure 17B, values > 10,000 are expressed as a quantified value (e.g., 39147.0) and in Figure 2B, these values are expressed as “10000”.
Figures 18A-18C relate to antibody targeting of Sbi. Sbi is a surface protein that is highly conserved among Staphylococci. Sbi includes two immunoglobulin-binding domains and mediates immune evasion. A variant of Sbi (Sbixx) lacking IgG-Fc binding was used for selection and characterization of certain antibodies of the present disclosure. Figure 18A shows a schematic of Sbi. Figure 18B shows (top) binding quantified by ELISA and (bottom) quantified binding values (EC50), percent IgG-binding inhibition, affinity (KD), and V-domain gene usage of certain antibodies of the present disclosure. Figure 18C shows sensorgram curves showing that antibodies SSH4 and SSH7 of the present disclosure do not compete with one another for Sbi binding.
Figure 19 shows binding to Cl by anti-LTA antibodies SSA8 and SSA12 of the present disclosure. Pagibaximab was included as a comparator.
Figure 20 shows neutralization, by anti-Gmd antibodies SSG20 and SSF11 of the present disclosure, in a cell wall digestion assay. 1C11 was included as a comparator.
Figure 21 shows quantified binding values (EC50 ng/mL to ClfA_001, ClfA_002, ClfA_004, and S. aureus strains). Tefibazumab was included as a comparator.
Figure 22 shows survival of BALB/c mice prophylactically administered SSE1, SSE158, MEDI4893, or an isotype control at the indicated dose followed by infection (i.n.) with S. aureus, as indicated.
Figure 23 relates to antibody targeting of Hla. Figure 24 provides a table showing results from Hla-binding (quantified as EC50 ng/mL) and Hla neutralization (on THP-1 cells, quantified as IC50 ng/mL) assays by certain anti-Hla antibodies of the present disclosure.
Figure 24 relates to antibody targeting of LTA. Figure 24 provides a table showing results from assays measuring binding (quantified as EC50 ng/mL) by certain anti-LTA antibodies of the present disclosure to LTA and to bacteria expressing Type I, Type II, or Type IV LTA (with E. Colt included as a negative control), and neutralization of LTA activity on TLR2-expressing HEK293 cells by certain antibodies of the present disclosure (quantified by absolute IC50 ng/mL).
Figure 25 relates to study design for testing efficacy of monoclonal antibodies against Staphylococcus targets in FPR3757 bacteremia model in BL6 mice. Figure 26 provides a schematic (top) of the study design and a table showing the descriptions of the various study groups, antibody dose, antibody delivery route, antibody dosing time, group size, Staphylococcus aureus strain and dosage used to challenge, route of infection, take down day and read outs, and days for clinical observation.
Figure 26 relates to mouse serum Ig level for each study group at 24 hours post-dosing of Staphylococcus aureus as measured using mesoscale discovery (MSD).
Figures 27A-27B relate to promotion of clearance of Staphylococcus aureus infection by select n -Staphylococcus antibodies within a subset of treated mice in a disseminated infection model. Figure 27A provides a graph showing effects of antibody treatment on bacterial kidney burden as measured by kidney CFU. Figure 27B provides a graph showing effects of antibody treatment on weight loss.
Figure 28 provides graphs showing individual mouse weight loss within each antibody study group.
DETAILED DESCRIPTION
Provided herein are antibodies and antigen-binding fragments that are capable of binding to one or more bacterial (e.g, Staphylococcus) antigen. In some embodiments, the one or more bacterial (e.g, Staphylococcus) antigen is selected from: a lipoteichoic acid (LTA), e.g., a Type I LTA, a Type II LTA, a Type IV LTA, or any combination thereof; a glucosaminidase (Gmd); a Pore Forming Toxin (PFT, also referred to as a Pore Forming Cytotoxin) such as a leukocidin (e.g., LukD, LukF, LukE, or LukS, or any combination thereof, such as LukED, LukSF, or both; “leukocidin” may alternatively be spelled as “leucocidin”), an alpha toxin (e.g., Hla; an alpha
toxin may also be referred to as an alpha hemolysin), or a gamma hemolysin (also referred to as a gamma toxin; e.g., HIgA and/or HIgB); a Staphylococcus Protein A (SpA); a Sbi (also referred to as Staphylococcal binder of immunoglobulin or second immunoglobulin binding protein); and a clumping factor A (ClfA).
Briefly, antibodies were identified from samples from human subjects that recovered from S. aureus infection (see, e.g., Table 1). In any of the presently disclosed embodiments, the one or more bacterial antigen is an antigen that is expressed by Staphylococci and, in some cases, one or more other gram-positive bacteria. In some embodiments, the antigen is expressed by S. aureus. Presently disclosed antigens may be described herein as “Staphylococcus antigens”.
In certain embodiments, the antigen is an antigen that is also expressed by one or more (e.g., gram-positive) bacteria that is not of genus Staphylococcus, and the antibody or antigenbinding fragment is capable of binding thereto. Accordingly, a "Staphylococcus antigen” is not limited to antigens expressed solely by bacteria of genus Staphylococcus and includes antigens that are expressed by Staphylococcus and by bacteria of one or more other genus. In certain embodiments, the antigen is also expressed by a bacteria of genus Enterococcus (e.g., E. faecalis), a bacteria of genus Lactococcus (e.g., L. garviae), a bacteria of genus Streptococcus (e.g., S. pyogenes, S. agalactiae) a bacteria of genus Listeria e.g., L. monocytogenes), a bacteria of genus Clostridium e.g., C. inocuum, C. difficile), or any combination thereof.
In some embodiments, the antibody or antigen-binding fragment is capable of binding to S. aureus e.g., MRS A), Coagulase-negative staphylococci (CoNS) such as S. epidermidis, S. pneumoniae, S. lugdunensis, S. hominis, or any combination thereof. In certain embodiments, the antibody or antigen-binding fragment is capable of binding to: S. aureus FPR3757, S. aureus NE284 TE2 mutant, S. epidermidis RP62A, S. epidermidis 1200, S. epidermidis NIH04008, S. lugdunensis e.g., N860297), S. pyogenes, E. faecalis, S. agalactiae, S. hominis, L. garvieae, S. pneumoniae, E. coli, or any combination of thereof.
In certain embodiments, an antibody or antigen-binding fragment of the present disclosure binds to an LTA. In certain embodiments, the antibody or antigen-binding fragment binds to a greater breadth of LTA-expressing bacteria e.g., Type I LTA-expressing bacteria) than the antibody pagibaximab. In certain embodiments, the antibody or antigen-binding fragment has improved binding to an isolated LTA, and/or to one or more LTA-expressing bacteria, as compared to the antibody pagibaximab e.g., as determined by EC50 value in an ELISA binding assay). In some embodiments, the antibody or antigen-binding fragment binds to an LTA with a non-zero EC50 (ng/mL) of 200 or less, 150 or less, 100 or less, 60 or less, 50 or
less, 40 or less, 30 or less, 20 or less, 15 or less, 14 or less, 13 or less, 12 or less, 11 or less, or 10 or less. In some embodiments, the antibody or antigen-binding fragment binds to an LTA with an EC50 (ng/mL) of about 9, about 10, about 11, about 12, about 13, about 14, about 15, about 16, about 17, about 18, about 19, about 20, about 21, about 22, about 23, about 24, about 25, about 30, about 35, about 40, about 45, about 50, or about 55. In some embodiments, the antibody or antigen-binding fragment binds to an LTA with an EC50 (ng/mL) of between 9 and 55, of between 9 and 25, of between 9 and 10, of between 9 and 12, of between 11 and 12, of between 11 and 14, of between 14 and 25, or of between 20 and 55.
In some embodiments, the antibody or antigen-binding fragment does not compete with pagibaximab for binding to LTA.
In some embodiments, the antibody or antigen-binding fragment binds to LTA with higher affinity than does pagibaximab. In certain embodiments, pagibaximab binds to an LTA with a Kd of 1.75E-1 IM and an antibody or antigen-binding fragment of the present disclosure binds to the LTA with a Kd of 3.48E-12M, as determined by surface plasmon resonance (SPR, e.g. using an Octet instrument).
In some embodiments, the antibody or antigen-binding fragment is capable of neutralizing LTA activity, e.g., as determined in an assay using TLR2-expressing HEK293 cells (e.g., HEK293 cells co-transfected with hTLR2 and SEAP (secreted embryonic alkaline phosphatase) reporter genes under the control of a minimal promoter fused to NF-kb and AP-1- binding sides) and determining whether TLR2 signaling initiated by LTA binding is occurring, e.g. by detection of a purple/blue color produced by hydrolysis of a substrate in the medium by SEAP. In certain embodiments, the antibody or antigen-binding fragment neutralizes LTA activity more potently than does pagibaximab.
In some embodiments, an anti-LTA antibody or antigen-binding fragment strongly binds Streptococcus pneumoniae while not displaying non-specific binding to Lactococcus or E. coli. Such specificity may provide utility of the antibody or antigen-binding fragment across a range of gram-positive pathogens that can cause serious disease. In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to a Gmd. In certain embodiments, the antibody or antigen-binding fragment is capable of binding to a Gmd of S. aureus, to a Gmd of S. epidermidis, or to both. Binding can be determined using, for example, an ELISA assay. In some embodiments, the antibody or antigen-binding fragment binds to one or more of the following bacteria in a fixed setting: S. aureus TE2 mutant; S. epidermidis 1200; S. epidermidis RP62A, and S. lugdunensis. In some embodiments, the antibody or antigen-
binding fragment binds to a greater breadth of Gmd-expressing bacteria as compared to reference antibody “1C11” (see, e.g., Varrone et al., J Orthop Res. 32(10): 1389-96 (2014); doi: 10.1002/jor.22672). In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of neutralizing Gmd activity (e.g., cell wall lysis) by measuring Gmd effect on Micrococcus luteus viability. In certain embodiments, the antibody or antigenbinding fragment neutralizes Gmd activity more potently than does 1C11.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to one or more Pore-Forming Cytotoxin (PFT). In certain embodiments, a PFT comprises an alpha toxin (e.g., Hla), a leucocidin (also referred to as a leukocidin or a leukotoxin), or gamma toxin (e.g., HIgA and/or HIgB; a gamma toxin may be referred to as a gamma hemolysin). In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to LukD, to LukF, to LukE, to LukS, to HIgA, to HIgB, or to any combination thereof. In certain embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to LukD, to LukF, and to HIgB. In certain embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to LukE, to LukS, and to HIgA. In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of neutralizing LukED, LukSF, or both (e.g., as determined by assessing viability of human monocytes exposed to Staphylococcus expressing the LukED and/or LukSF). In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to a S. aureus alpha toxin (e.g., Hla). In certain embodiments, the antibody or antigen-binding fragment is capable of binding to Hla with greater potency as compared to the anti-Hla antibody MEDI4893 (see, e.g., Tabor et al., Antimicrob Agents Chemother. 60(9):5312-21 (2016); doi: 10.1128/AAC.00357-16). In some embodiments, the antibody or antigen-binding fragment binds to Hla with a Kd of 1.96E-10 M or of 6.14E-11 M, and MEDI4893 binds to the Hla with a Kd of 5.1E-10 by the same assay. In some embodiments, the antibody or antigen-binding fragment neutralizes Hla (e.g., in an assay using rabbit RBCs). In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to S. aureus HIgB.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to a SpA. In some embodiments, the antibody or antigen-binding fragment is capable of binding to SpA with a Kd of about 2.46E-09 M, about 1.1 IE-08 M, about 7.633E- 10 M, 1.77E-09 M, or about 1.44E-09 M, as determined by surface plasmon resonance using an Octet instrument.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to a ClfA. In some embodiments, the antibody or antigen-binding fragment is capable of binding to ClfA OOl, to ClfA_002 to ClfA_004, or to any combination thereof.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of binding to a Sbi.
In certain embodiments, an antibody or antigen-binding fragment is human, humanized, or chimeric. In some embodiments, the antibody or antigen-binding fragment comprises human amino acid sequences, e.g. : one, two, three, four, five, or six human CDR sequences; one, two, three, four, five, six, seven, or eight human variable domain framework region sequences. In some embodiments, the antibody or antigen-binding fragment comprises an Fc polypeptide, which can be, for example, a human Fc polypeptide or an engineered variant of a human Fc polypeptide.
In certain embodiments, an antibody or antigen-binding fragment comprises one or more CDRs (or functional variants thereof) of an antibody or antigen-binding fragment as set forth in any one of Tables 1-4. In certain embodiments, an antibody or antigen-binding fragment comprises the six CDRs (according to any known numbering scheme, e.g., IMGT, Kabat, North, Chothia, Martin (known as Enhanced Chothia), AHo, AbM, Contact, or the like, or any combination thereof) of an antibody or antigen-binding fragment as set forth in any one of Tables 1-4. In certain embodiments, an antibody or antigen-binding fragment comprises (i) a VH comprising, consisting essentially of, or consisting of an amino acid sequence having at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% to the VH amino acid sequence of an antibody or antigen-binding fragment as set forth in any one of Tables 1-4; and (ii) a VL comprising, consisting essentially of, or consisting of an amino acid sequence having at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% to the VL amino acid sequence of an antibody or antigen-binding fragment as set forth in any one of Tables 1-4.
In particular embodiments, an antibody or antigen-binding fragment of the present disclosure comprises the six CDRs, and optionally the VH and VL amino acid sequences, of any one of the following antibodies: SSA12; SSG20; SSH3; SSL17; SSE1; SSE158; SSB24; SSB65; SSC35; SSE73; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSF2; SSF11; SSF10; SSB18; SSB54; SSB67; SSF33; SSL5; SSL35; SSG50; SSG54; SSM47; SSG44; SSG48; SSM37; SSH4; SSH7; SSG27; SSG39; SSM45; SSF52; SSG51; SSF34; SSG59; SSAA8; SSAA26; SSAB5;
SSAI3; SSAI8; SSAI2; SSAF1; SSAL2; SSAM6; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; SSF69; SSN2; SSN6; SSH8; SSM35; SSM36; and SSM40.
In some embodiments, an antibody or antigen-binding fragment comprises a VH and a VL, wherein the VH and VL comprise or consist of amino acid sequences having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity to, or comprising or consisting of, the VH and VL amino acid sequences of any one of the following antibodies: SSA12; SSG20; SSH3; SSL17; SSE1; SSE158; SSB24; SSB65; SSC35; SSE73; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSF2; SSF11; SSF10; SSB18; SSB54; SSB67; SSF33; SSL5; SSL35; SSG50; SSG54; SSM47; SSG44; SSG48; SSM37; SSH4; SSH7; SSG27; SSG39; SSM45; SSF52; SSG51; SSF34; SSG59; SSAA8; SSAA26; SSAB5;
SSAI3; SSAI8; SSAI2; SSAF1; SSAL2; SSAM6; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; SSF69; SSN2; SSN6; SSH8; SSM35; SSM36; and SSM40. In certain embodiments, the antibody or antigen-binding fragment comprises the six CDRs of: SSA12; SSG20; SSH3; SSL17; SSE1; SSE158; SSB24; SSB65; SSC35; SSE73; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSF2; SSF11; SSF10; SSB18; SSB54; SSB67; SSF33; SSL5; SSL35; SSG50; SSG54; SSM47; SSG44; SSG48; SSM37; SSH4; SSH7; SSG27; SSG39;
SSM45; SSF52; SSG51; SSF34; SSG59; SSAA8; SSAA26; SSAB5; SSAI3; SSAI8; SSAI2; SSAF1; SSAL2; SSAM6; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; SSF69; SSN2; SSN6; SSH8; SSM35; SSM36; or SSM40.
In any of the presently disclosed embodiments, an antibody or antigen-binding fragment can comprise one or more features selected to reduce binding by SpA. In some embodiments, an antibody or antigen-binding fragment that is natively encoded/expressed as a human isotype other than IgG3 (e.g., human IgGl) is provided in a human IgG3 isotype. In some embodiments, an antibody or antigen-binding fragment comprises an engineered variant of a human IgGl Fc polypeptide, comprising an H435R mutation, a Y436F mutation, or H435R/Y436F mutations. In some embodiments, an antibody or antigen-binding fragment comprises an engineered variant of a human IgGl Fc polypeptide, comprising E345R/E340G/S440Y mutations.
In any of the presently disclosed embodiments, an antibody or antigen-binding fragment can comprise an Fc polypeptide comprising one or more mutations to: increase binding affinity
to human FcRn and/or increase in vivo half-life (e.g, M428L/N434S); and/or to increase one or more effector function (e.g., increased binding to and/or activation of a FcyRa, decreased binding to and/or activation of an FcyRb, binding to complement (Cl), ADCP, ADCC, or the like). In certain embodiments, an Fc polypeptide comprises the following mutation(s): K236WZE333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L; S267E; S324T; G236A/Y300L; G236A/R292P/I377N; R292P/Y300L; E340G; G236D (optionally wherein the antibody or antigen-binding fragment is afucosylated); E345K; S239D;
S267E/S324T; F243L; or G236A/R292P/Y300L. In some embodiments, an antibody or antigenbinding fragment comprises a heavy chain comprising M428L/N434S/H435R/Y436F mutations. In some embodiments, an antibody or antigen-binding fragment comprises an IgGl heavy chain comprising any of the mutations, or comprises an Fc variant, as shown in any one of Figures 14A-14C and 14E-14K. Other suitable mutations are known in the art and/or are discussed herein.
In certain embodiments, an antibody or antigen-binding fragment of the present disclosure is capable of neutralizing an infection by one or more antigen-expressing bacteria, e.g., Staphylococcus, such as S. aureus.
In some embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment is provided that comprises the six CDRS, and optionally the VH and VL, of an antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1). In certain embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment is provided that comprises (1) the six CDRS, and optionally the VH and VL, of a first antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1) and (2) the six CDRS, and optionally the VH and VL, of a second, different antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1). In some embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment can bind to two or more different antigens; for example, in some embodiments, the multispecific (e.g., bispecific) antibody or antigen-binding fragment binds to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a SpA and a ClfA; or a Sbi and: a LTA, a Gmd, an alpha toxin, a gamma toxin, a leucocidin, a SpA, or a Clfa. A multispecific (e.g., bispecific)
antibody or antigen-binding fragment can comprise, for example, the six CDRs, and optionally the VH and VL, of any one or more or any two or more two or more antibodies described in Table 1.
In some embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment can bind to two or more different antigens, wherein a first antigen is a bacterial (e.g., Staphylococcus) antigen and a second antigen is a non-bacterial or non-Staphylococcus antigen.
In some embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment comprises a first antigen -binding domain and second antigen-binding domain, wherein the first antigen-binding domain or the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC10; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF10; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE150; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
In some embodiments, an antibody or antigen-binding fragment is capable of binding: a LTA and a Gmd; a LTA and a ClfA; a LTA and a SpA; a Gmd and a ClfA; a Gmd and a SpA; a ClfA and a SpA; a LTA and a LTA (e.g., different epitopes on an LTA and/or comprising a nonnative format comprising two binding domains for a same epitope on an LTA); a Gmd and a Gmd (e.g., different epitopes on an Gmd and/or comprising a non-native format comprising two binding domains for a same epitope on a Gmd); a SpA and a SpA (e.g., different epitopes on an SpA and/or comprising a non-native format comprising two binding domains for a same epitope on an SpA); a ClfA and a ClfA (e.g., different epitopes on an ClfA and/or comprising a non- native format comprising two binding domains for a same epitope on an ClfA); a LTA and a non-Slaphylococctis antigen; a Gmd and a non-Staphylococcus antigen; a SpA and a non- Staphylococcus antigen; a ClfA and a non-Staphylococcus antigen; a first alpha toxin and a
second alpha toxin; two epitopes on an alpha toxin; an alpha toxin and a non-Staphylococcus antigen; a first leukotoxin and a second leukotoxin (also called first leucocidin and second leucocidin, respectively); two epitopes on a leukotoxin; or a leukotoxin and a non- Staphylococcus antigen. In certain embodiments, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S mutations or M428L/N434S/H435R/Y436F mutations. In certain embodiments, the human IgGl Fc polypeptide further comprises two CH2-CH3 -containing polypeptides wherein the two CH3 domains, together, comprise mutations to promote associate between the two CH3 domains, e.g., knob-into-hole mutations, modifications to introduce a disulfide bond (e.g., introduced cysteines), charge-pair mutations, or any combination thereof.
In some embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment comprises a first antigen -binding domain and second antigen-binding domain, wherein the first antigen-binding domain and the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively. In certain embodiments, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S mutations or M428L/N434S/H435R/Y436F mutations. In certain embodiments, the human IgGl Fc polypeptide further comprises two CH2-CH3- containing polypeptides wherein the two CH3 domains, together, comprise mutations to promote associate between the two CH3 domains, e.g., knob-into-hole mutations, modifications to introduce a disulfide bond (e.g., introduced cysteines), charge-pair mutations, or any combination thereof.
In some embodiments, a combination therapy or composition comprises two antibodies or antigen-binding fragments, wherein each of the two antibodies or antigen-binding fragments comprises the six CDRS, and optionally the VH and VL, of a different antibody or antigenbinding fragment of the present disclosure (e.g., as shown in Table 1). In some embodiments, a first antibody or antigen-binding fragment binds to a first antigen and a second antibody or antigen-binding fragment binds to a second, different, antigen; for example, in some embodiments, first and the second antibodies or antigen-binding fragments respectively bind to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin
and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a SpA and a ClfA; a Sbi and a LT A; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a SpA; or a Sbi and a ClfA.
In some embodiments, a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment are respectively capable of binding: a LTA and a Gmd; a LTA and a ClfA; a LTA and a SpA; a Gmd and a ClfA; a Gmd and a SpA; a ClfA and a SpA; a LTA and a LTA (e.g., different epitopes on an LTA); a Gmd and a Gmd (e.g., different epitopes on an Gmd); a SpA and a SpA (e.g., different epitopes on an SpA); a ClfA and a ClfA (e.g., different epitopes on an ClfA); a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a SpA; a Sbi and a ClfA; a Sbi and a Sbi (e.g. , different epitopes on an SpA); a LTA and a non-Staphylococcus antigen; a Gmd and a non- Staphylococcus antigen; a SpA and a non-Staphylococcus antigen; a ClfA and a non- Staphylococcus antigen; a first alpha toxin and a second alpha toxin; two epitopes on an alpha toxin; an alpha toxin and a non-Staphylococcus antigen; a first leukotoxin and a second leukotoxin (also called first leucocidin and second leucocidin, respectively); two epitopes on a leukotoxin; a leukotoxin and a non-Staphylococcus antigen; two epitopes on a Sbi; or a Sbi and a non-Staphylococcus antigen. In certain embodiments, each of the first the antibody or antigenbinding fragment and the second antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S mutations or M428L/N434S/H435R/Y436F mutations. In certain embodiments, the human IgGl Fc polypeptide further comprises two CH2- CH3 -containing polypeptides wherein the two CH3 domains, together, comprise mutations to promote associate between the two CH3 domains, e.g, knob-into-hole mutations, modifications to introduce a disulfide bond (e.g, introduced cysteines), charge-pair mutations, or any combination thereof.
In particular embodiments, a multispecific (e.g., bispecific) antibody or antigen-binding fragment is provided that binds to an alpha toxin and to a LTA, a ClfA, a Protein A, a Gmd, or a Sbi. In some embodiments, the multispecific (e.g., bispecific) antibody or antigen-binding fragment comprises the six CDRs, and optionally the VH and VL (or amino acid sequences having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity thereto), of SSE1 or SSE158. In some embodiments, the
multispecific (e.g., bispecific) antibody or antigen-binding fragment comprises the six CDRs, and optionally the VH and VL (or amino acid sequences having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity thereto), an anti-LTA, anti-ClfA, anti-Protein A, anti-Gmd, or anti-Sbi antibody or antigen-binding fragment of the present disclosure. In some embodiments, the anti-LTA, anti-ClfA, anti-Protein A, or anti-Gmd antibody or antigen-binding fragment is SSA12, SSF11, SSG27, or SSH3.In particular embodiments, a combination therapy or composition is provided that comprises a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the a first antibody or antigen-binding fragment binds to an alpha toxin and the second antibody or antigen-binding fragment to a LTA, a ClfA, a Protein A, a Gmd, or a Sbi. In some embodiments, the first antibody or antigen-binding fragment comprises the six CDRs, and optionally the VH and VL (or amino acid sequences having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least
94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity thereto), of
SSE1 or SSE158. In some embodiments, the second antibody or antigen-binding fragment comprises the six CDRs, and optionally the VH and VL (or amino acid sequences having at least
85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least
92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least
99% identity thereto), an anti-LTA, anti-ClfA, anti-Protein A, anti-Gmd, or anti-Sbi antibody or antigen-binding fragment of the present disclosure. In some embodiments, the anti-LTA, anti- ClfA, anti-Protein A, or anti-Gmd antibody or antigen-binding fragment is SSA12, SSF11, SSG27, or SSH3.
Also provided are polynucleotides that encode the antibodies and antigen-binding fragments, vectors, DNA therapeutics, RNA therapeutics, host cells, and related compositions, as well as methods of using the antibodies, antigen-binding fragments, polynucleotides, vectors, DNA therapeutics, RNA therapeutics, host cells, and related compositions to treat (e.g., reduce, delay, eliminate, or prevent) a bacterial (e.g., Staphylococcus, such as S. aureus) infection in a subject and/or in the manufacture of a medicament for treating bacterial infection in a subject.
Also provided is a library comprising a plurality of antibodies or antigen-binding fragments, or comprising polynucleotides encoding the same, wherein the library comprises or encodes certain human immunoglobulin variable domain alleles. Briefly, the present disclosure teaches that certain alleles and combinations of alleles are used by clonally related antibodies
that can bind to a herein disclosed antigen. A library can, for example, comprise polynucleotides having a genomic allele sequence, and/or can contain alternative polynucleotide sequences that encode a same amino acid sequence encoded by the genomic allele. The library can be enriched for these sequences. Alternatively or additionally, a library can comprise antibodies or antigenbinding fragments. The antibodies or antigen-binding fragments can be expressed (e.g., the polynucleotides can be expressed by a plurality of host cell, such as, for example, yeast, human B cells, or human plasma cells, and antibody variable domain genes may undergo recombination, somatic hypermutation, or both) and the library can be screened for antibodies or antigen-binding fragments that bind to a Staphylococcus antigen as provided herein.
Prior to setting forth this disclosure in more detail, it may be helpful to an understanding thereof to provide definitions of certain terms to be used herein. Additional definitions are set forth throughout this disclosure.
In the present description, any concentration range, percentage range, ratio range, or integer range is to be understood to include the value of any integer within the recited range and, when appropriate, fractions thereof (such as one tenth and one hundredth of an integer), unless otherwise indicated. Also, any number range recited herein relating to any physical feature, such as polymer subunits, size or thickness, are to be understood to include any integer within the recited range, unless otherwise indicated. As used herein, the term “about” means ± 20% of the indicated range, value, or structure, unless otherwise indicated. It should be understood that the terms “a” and “an” as used herein refer to “one or more” of the enumerated components. The use of the alternative (e.g., “or”) should be understood to mean either one, both, or any combination thereof of the alternatives. As used herein, the terms “include,” “have,” and “comprise” are used synonymously, which terms and variants thereof are intended to be construed as non-limiting.
“Optional” or “optionally” means that the subsequently described element, component, event, or circumstance may or may not occur, and that the description includes instances in which the element, component, event, or circumstance occurs and instances in which they do not.
In addition, it should be understood that the individual constructs, or groups of constructs, derived from the various combinations of the structures and subunits described herein, are disclosed by the present application to the same extent as if each construct or group of constructs was set forth individually. Thus, selection of particular structures or particular subunits is within the scope of the present disclosure.
The term “consisting essentially of’ is not equivalent to “comprising” and refers to the specified materials or steps of a claim, or to those that do not materially affect the basic characteristics of a claimed subject matter. For example, a protein domain, region, or module (e.g., a binding domain) or a protein “consists essentially of’ a particular amino acid sequence when the amino acid sequence of a domain, region, module, or protein includes extensions, deletions, mutations, or a combination thereof (e.g., amino acids at the amino- or carboxyterminus or between domains) that, in combination, contribute to at most 20% (e.g., at most 15%, at most 10%, at most 8%, at most 6%, at most 5%, at most 4%, at most 3%, at most 2% or at most 1%) of the length of a domain, region, module, or protein and do not substantially affect (i.e., do not reduce the activity by more than 50%, such as no more than 40%, no more than 30%, no more than 25%, no more than 20%, no more than 15%, no more than 10%, no more than 5%, or no more than 1%) the activity of the domain(s), region(s), module(s), or protein (e.g., the target binding affinity of a binding protein).
As used herein, “amino acid” refers to naturally occurring and synthetic amino acids, as well as amino acid analogs and amino acid mimetics that function in a manner similar to the naturally occurring amino acids. Naturally occurring amino acids are those encoded by the genetic code, as well as those amino acids that are later modified, e.g., hydroxyproline, y- carboxyglutamate, and O-phosphoserine. Amino acid analogs refer to compounds that have the same basic chemical structure as a naturally occurring amino acid, i.e., an a-carbon that is bound to a hydrogen, a carboxyl group, an amino group, and an R group, e.g., homoserine, norleucine, methionine sulfoxide, methionine methyl sulfonium. Such analogs have modified R groups (e.g., norleucine) or modified peptide backbones, but retain the same basic chemical structure as a naturally occurring amino acid. Amino acid mimetics refer to chemical compounds that have a structure that is different from the general chemical structure of an amino acid, but that functions in a manner similar to a naturally occurring amino acid.
As used herein, “mutation” refers to a change in the sequence of a nucleic acid molecule or polypeptide molecule as compared to a reference or wild-type nucleic acid molecule or polypeptide molecule, respectively. A mutation can result in several different types of change in sequence, including substitution, insertion or deletion of nucleotide(s) or amino acid(s).
A “conservative substitution” refers to amino acid substitutions that do not significantly affect or alter binding characteristics of a particular protein. Generally, conservative substitutions are ones in which a substituted amino acid residue is replaced with an amino acid residue having a similar side chain. Conservative substitutions include a substitution found in
one of the following groups: Group 1 : Alanine (Ala or A), Glycine (Gly or G), Serine (Ser or S), Threonine (Thr or T); Group 2: Aspartic acid (Asp or D), Glutamic acid (Glu or Z); Group 3: Asparagine (Asn or N), Glutamine (Gin or Q); Group 4: Arginine (Arg or R), Lysine (Lys or K), Histidine (His or H); Group 5: Isoleucine (He or I), Leucine (Leu or L), Methionine (Met or M), Valine (Vai or V); and Group 6: Phenylalanine (Phe or F), Tyrosine (Tyr or Y), Tryptophan (Trp or W). Additionally or alternatively, amino acids can be grouped into conservative substitution groups by similar function, chemical structure, or composition (e.g., acidic, basic, aliphatic, aromatic, or sulfur-containing). For example, an aliphatic grouping may include, for purposes of substitution, Gly, Ala, Vai, Leu, and He. Other conservative substitutions groups include: sulfur- containing: Met and Cysteine (Cys or C); acidic: Asp, Glu, Asn, and Gin; small aliphatic, nonpolar or slightly polar residues: Ala, Ser, Thr, Pro, and Gly; polar, negatively charged residues and their amides: Asp, Asn, Glu, and Gin; polar, positively charged residues: His, Arg, and Lys; large aliphatic, nonpolar residues: Met, Leu, He, Vai, and Cys; and large aromatic residues: Phe, Tyr, and Trp. Additional information can be found in Creighton (1984) Proteins, W.H. Freeman and Company.
As used herein, “protein” or “polypeptide” refers to a polymer of amino acid residues. Proteins apply to naturally occurring amino acid polymers, as well as to amino acid polymers in which one or more amino acid residue is an artificial chemical mimetic of a corresponding naturally occurring amino acid, and non-naturally occurring amino acid polymers. Variants of proteins, peptides, and polypeptides of this disclosure are also contemplated. In certain embodiments, variant proteins, peptides, and polypeptides comprise or consist of an amino acid sequence that is at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.9% identical to an amino acid sequence of a defined or reference amino acid sequence as described herein.
Any polypeptide of this disclosure (e.g., VH, VL, scFv, scFab, Fab, Fd, antibody heavy chain, antibody light chain) can, as encoded by a polynucleotide sequence, comprise a “signal peptide” (also known as a leader sequence, leader peptide, or transit peptide). Signal peptides target newly synthesized polypeptides to their appropriate location inside or outside the cell. A signal peptide may be removed in whole or in part from the polypeptide during or once localization or secretion is completed. Polypeptides that have a signal peptide can be referred to as a “pre-protein” and polypeptides having their signal peptide removed can be referred to as
“mature” proteins or polypeptides. In any of the herein disclosed embodiments, an antibody or antigen-binding fragment comprises, or is, a mature protein, or is or comprises a pre-protein.
“Nucleic acid molecule” or “polynucleotide” or “polynucleic acid” refers to a polymeric compound including covalently linked nucleotides, which can be made up of natural subunits (e.g., purine or pyrimidine bases) or non-natural subunits (e.g., morpholine ring). Purine bases include adenine, guanine, hypoxanthine, and xanthine, and pyrimidine bases include uracil, thymine, and cytosine. Nucleic acid molecules include polyribonucleic acid (RNA), which includes mRNA, microRNA, siRNA, self-assembling RNA, self-amplifying RNA (saRNA), trans-amplifying RNA (taRNA), viral genomic RNA, circular RNA (circRNA), and synthetic RNA, and polydeoxyribonucleic acid (DNA, also referred to as deoxyribonucleic acid), which includes cDNA, genomic DNA, and synthetic DNA, either of which may be single or double stranded. If single-stranded, the nucleic acid molecule may be the coding strand or non-coding (anti-sense) strand. A nucleic acid molecule encoding an amino acid sequence includes all nucleotide sequences that encode the same amino acid sequence. Some versions of the nucleotide sequences may also include intron(s) to the extent that the intron(s) would be removed through co- or post-transcriptional mechanisms. In other words, different nucleotide sequences may encode the same amino acid sequence as the result of the redundancy or degeneracy of the genetic code, or by splicing.
In some embodiments, the polynucleotide comprises a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof. In certain embodiments, the polynucleotide comprises a pseudouridine, a N6-methyladenonsine, a 5-methylcytidine, a 2- thiouridine, or any combination thereof. In some embodiments, the pseudouridine comprises N1 -methylpseudouridine. These features are known in the art and are discussed in, for example, Zhang et al. Front. Immunol., DOI=10.3389/fimmu.2019.00594 (2019); Eyler et al. PNAS 116(46): 23068-23071; DOI: 10.1073/pnas. l821754116 (2019); Nance and Meier, ACS Cent. Set. 2021, 7, 5, 748-756; doi.org/10.1021/acscentsci.lc00197 (2021), and van Hoecke and Roose, J. Translational Med 17:54 (2019); https://doi.org/10.1186/sl2967-019-1804-8, which modified nucleosides and mRNA features are incorporated herein by reference. Variants of nucleic acid molecules of this disclosure are also contemplated. Variant nucleic acid molecules include those that are at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, and are preferably at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or at least 99.9% identical to a nucleic acid molecule of a defined or reference polynucleotide (or polynucleotide sequence) as described herein, or that hybridize to a polynucleotide under stringent hybridization
conditions of 0.015M sodium chloride, 0.0015M sodium citrate at about 65-68°C or 0.015M sodium chloride, 0.0015M sodium citrate, and 50% formamide at about 42°C. Nucleic acid molecule variants retain the capacity to encode a binding domain thereof having a functionality described herein, such as binding a target molecule.
“mRNA” refers to any form of messenger RNA having a protein-coding region able to be translated in a host cell to produce the protein coded, in particular an antibody or antigen-binding fragment.
In some embodiments, each distinct mRNA may encode only one protein, such as a HC or a VH-containing fragment thereof, or a LC or a VL-containing fragment thereof. In such embodiments, a RNA therapeutic containing such mRNA may include at least two distinct mRNAs, one encoding a HC or a VH-containing fragment thereof, and the other encoding a LC or a VL-containing fragment thereof, so that, upon expression of the mRNAs in the cell, the resulting VH and VL proteins may combine to form an antigen-binding domain.
In other embodiments, all proteins, or at least a protein comprising a VH and a protein comprising a VL, may be encoded in a single mRNA. In some such embodiments, the VH- comprising protein and the VL-comprising protein may be expressed as separate proteins due to other regulatory elements within the coding sequence or encoded amino acid sequence, such as signal peptides, spacers, and translation-regulatory sequences, such as internal ribosome entry site (IRES) sequences. In some embodiments, a mRNA encodes a self-cleaving (e.g., 2A, such as a F2A, T2A, P2A, or E2A) peptide. A 2A peptide, optionally with a short linker or spacer sequence at the N- and/or C-terminal end, may be disposed between a VH-comprising protein and a VL-comprising protein. In certain embodiments, the order in which the VH-comprising protein and VL-comprising protein and/or other expressed proteins occur in the mRNA may be optimized. In particular, RNA therapeutics containing saRNA or circRNA may be optimized in this manner, in some embodiments by placing a sequence encoding a VH-comprising protein 5’ to a sequence encoding a VL-comprising protein.
In some embodiments, the antibody or antigen-binding fragment may include a signal peptide that causes secretion of the antibody or antigen-binding fragment from the host cell. mRNA of the present disclosure, in particular circRNA, taRNA, or saRNA, may further contain one or more modified nucleosides. In particular embodiments, the modified nucleosides may stabilize the mRNA and reduce degradation of the mRNA in vitro, such as during production, storage, or after reconstitution and prior to administration of the mRNA, a vector comprising or consisting of the mRNA, an mRNA therapeutic construct, or a composition
containing any of the preceding. In particular embodiments, the modified nucleosides may stabilize and reduce degradation of the mRNA in vivo, such as in the skin, muscle, blood, interstitial fluid, other extracellular environment, or intracellular environment after administration of the mRNA, a vector or construct containing the mRNA, an mRNA therapeutic construct, or a composition containing any of the preceding. In particular embodiments, the modified nucleosides may reduce or avoid a cellular immune response to the mRNA. In particular embodiments, the modified nucleosides may enhance amplification of the mRNA (particularly in the case of taRNA or saRNA) in the host cell and/or expression of a protein or peptide encoded by the mRNA. Modified nucleosides may include pseudouridine, such as N1- methylpseudouridine, 5-methylcytidine, 2-thiouridine, N6-methyladenonsine. Nucleoside modifications suitable for use with RNA are further described in Zhang et al. Front. Immunol., DOI=10.3389/fimmu.2019.00594 (2019); Eyler et al. PNAS 116(46): 23068-23071; DOI: 10.1073/pnas.1821754116 (2019); Nance and Meier, ACS Cent. Sci. 2021, 7, 5, 748-756; doi.org/10.1021/acscentsci. lc00197 (2021), and van Hoecke and Roose, J. Translational Med 17:54 (2019); https://doi.org/10.1186/sl2967-019-1804-8, which modified nucleosides and mRNA features are incorporated herein by reference. mRNA may include more than two or more types of modified nucleosides.
In some embodiments, methylation of a naturally occurring (unmodified) nucleotide may be used alone or in combination with a modified nucleotide to achieve any of the above effects achievable using a modified nucleotide. In some embodiments, the proportion of nucleotides having a specific base or located in a specific sequence that are methylated may be used to achieve such effects. For example, methylation may be used to reduce recognition of the mRNA (or, in the case of DNA therapeutics, the DNA) as foreign in a mammalian host cell. Types of RNA methylation and their effects are further described in Yujia Z., et al., “Principles of RNA methylation and their implications for biology and medicine,” Biomedicine & Pharmacotherapy (131) 110731 (2020), which is incorporated by reference in its entirety herein.
In some embodiments, the mRNA, particularly saRNA or taRNA, may include a cap or a cap analog, more specifically a 7-methylguanosine moiety linked via a phosphate, particularly a trisphosphate to an end nucleotide. The including of a cap or cap analog may help prevent exonuclease cleavage of the mRNA, particularly saRNA or taRNA, and/or initiate translation of the mRNA, particularly saRNA or taRNA, in a mammalian host cell. In specific embodiments, the cap or cap analog may initiate translation of the replication protein or peptide. circRNA may lack a suitable location for a cap or cap analog because of the absence of an otherwise unbound
5’ end. However, linear mRNA, prior to circularization to form circRNA, may contain a cap or cap analog to, for example, increase stability and/or amplification of the linear mRNA prior to circularization.
In some embodiments, the mRNA may include at least one untranslated region (UTR) sequence that regulates expression of an encoded protein or peptide. In some embodiments, the UTR may be endogenous to the host cell. In some embodiments, the UTR may be exogenous to the host cell or artificially designed. In some embodiments, the mRNA may include two UTRs, one 5’ and one 3’ of the nucleic acid sequence encoding the protein or peptide. For mRNAs encoding more than one protein or peptide, UTRs may be 5’ and 3’ of the sequence encoding at least one, typically each, protein or peptide, or 5’ and 3’ of the sequence encoding at least two proteins or peptides, all proteins or peptides that function together once expressed (e.g. theVH and VL-containing sequences), or all proteins and peptides encoded by the mRNA.
In some embodiments, the mRNA, particularly saRNA or taRNA, may include a polyA tail or a sequence that results in a polyA tail in mRNA produced from saRNA or taRNA (e.g. by reverse transcription) in a mammalian host cell. In some embodiments, the polyA tail, prior to initial translation of mRNA, particularly saRNA or taRNA, particularly taRNA sequences encoding amplification proteins, containing the polyA tail, may have a length of at least or approximately 250, 200, 100, 50, 20, or 10 nucleotides, or a length in a range between 10 and 250, 10 and 200, 10 and 100, 10 and 50, 10 and 20, 20 and 250, 20 and 200, 20 and 100, 20 and 50, 50 and 250, 50 and 200, 50 and 100, 100 and 250, 100 and 200, or 200 and 250 nucleotides. The length of the polyA tail may be varied to affect the lifespan of the mRNA in the host cell, with a longer polyA tail resulting in a longer lifespan and typically also a greater number of times the mRNA is translated prior to degradation. circRNA may lack a suitable location for a polyA tail because of the absence of an unbound end. However, linear mRNA, prior to circularization to form circRNA, may contain a polyA tail, for example, increase stability and/or amplification of the linear mRNA prior to circularization.
In certain embodiments, the mRNA, including circRNA, taRNA, or saRNA, may be produced in a production host cell, which is a type of host cell. In other embodiments, the mRNA, including circRNA, taRNA, or saRNA, may be produced in a cell-free system, such as a system using a DNA template and enzymes.
“Circular RNA” (“circRNA”) is a type of single-stranded mRNA that forms a covalently- closed continuous loop. In some embodiments, the 3’ and 5’ ends of a corresponding linear protein-coding mRNA are covalently bound in circRNA. In some embodiments, circRNA may
be resistant to exonuclease degradation as compared to corresponding linear mRNA. In specific embodiments, the circRNA includes one or more nucleotide sequences encoding one or more antibodies or antigen-binding fragments that are expressed when the circRNA is present in the host cell. In a specific embodiment of the present disclosure, circRNA may include one or more nucleotide sequences encoding an antibody or antigen-binding fragment according to the present disclosure that are expressed and produce the antibody or antigen-binding fragment in a human host cell in vivo.
In some embodiments circRNA may be formed from mature linear mRNA, particularly by self-splicing. In some embodiments, circRNA may self-splice by back-splicing, in which the 3’ end, after a protein coding region, binds to a 5’ end before the protein-coding region. In some embodiments, circRNA may self-splice by intron-pairing driven circularization, in which introns in the protein-coding region bind to one another, for example using Alu repeats. In some embodiments, circRNA may form a debranching-resistant lariat in an intron of the proteincoding region. In some embodiments, circRNA may form an exon-skipping lariat within the protein-coding region.
In all of these embodiments, the circRNA or linear mRNA from which it is formed may contain circularization sequences, which facilitate circularization of linear mRNA. In some embodiments, circRNA may be formed from linear mRNA via action of a spliceosome in an in vitro production host cell.
In some embodiments, circRNA may be introduced to the human host cell via a circRNA therapeutic construct. This introduction may occur in vivo in a human subject and, in particular in an in vivo human host cell. Once in the cytosol, the circRNA is translated, thereby expressing the encoded protein(s).
“Self-amplifying RNA” (“sa RNA” or “saRNA”) is also sometimes referred to as “selfreplicating RNA” or a “replicon.” saRNA is a type of mRNA that contains one or more nucleotide sequences that, when the saRNA is present in a host cell, cause replication of the saRNA and that also includes one or more nucleotide sequences encoding one or more antibodies or antigen-binding fragments that are expressed when the saRNA is present in the host cell. In a specific embodiment of the present disclosure, a saRNA may include i) one or more nucleotide sequences encoding an antibody or antigen-binding fragment according to the present disclosure that are expressed and produce the antibody or antigen-binding fragment in a human host cell in vivo, and ii) one or more nucleotide sequences that cause replication of the saRNA in the human
host cell in vivo. In some embodiments, the one or more nucleotide sequences that cause replication of the saRNA in a host cell may encode one or more replication proteins or peptides.
In some embodiments, the saRNA includes one or more promoters that cause expression of the replication protein or peptide and the polypeptide of the present disclosure in the host cell. In some embodiments, the saRNA further includes one or more promoters that cause expression of the replication protein or peptide and the antibody or antigen-binding fragment in the host cell. In specific embodiments, the promoter has a sequence comprising the following sequence TAACCTGAATGGACTACGACATAGTCTAGTCCGCCAAGTCTAGCATATGGCCACCAT G (SEQ ID NO.: 764)
In specific embodiments, the one or more nucleotide sequences that cause replication of the saRNA in a human host cell include sequences encoding one or more replication proteins or peptides that replicate the saRNA. In a more specific embodiment, the replication protein or peptide may be an RNA polymerase, such as an RNA-dependent RNA polymerase (RDRP). In some embodiments, the RDRP maybe an alphavirus RDRP or a variant thereof, more particularly a Venezuelan Equine Encephalitis Virus (VEEV), particularly the VEE TC-83 virus, Semliki Forest Virus (SFV), or Snibidis Virus RDRP or variant thereof. In more particular embodiments, the replication proteins may comprise or consist of non- structural proteins 1-4 (nsPl-4), in which nsPl is a protein that causes mRNA capping, nsP2 is a NTPase/helicase/protease, nsP3 is a macrodomain that mediates interactions between viral and host proteins, and nsP4 is RDRP.
In some embodiments, SEQ ID NO:762 includes the amino acid sequence for nsPl-4.
The X in SEQ ID NO.: 762 may be an amino acid, and may be any amino acid. In some embodiments, X may be R. The X in SEQ ID NO.: 763 may be an amino acid, and may be any amino acid. In some embodiments, X is preferably R.
In some embodiments, the saRNA encodes at least one replication protein having a sequence with at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity to SEQ ID NO: 187.
Modifications and production methods described above for saRNA may, in some embodiments, be present or used with other types of mRNA.
In some embodiments, the saRNA may be introduced to a human (or other) host cell via an saRNA therapeutic construct. This introduction may occur in vivo in a human subject and, in particular in an in vivo human host cell. Once in the cytosol, ribosomes may translate the
saRNA, thereby expressing the one or more replication proteins or peptides. The replication proteins or peptides may then produce a negative saRNA strand (a strand complementary to the saRNA as delivered to the host cell), which is then used as a template by the replication proteins or peptides to produce replicated positive saRNA strands. In some embodiments, the replicated positive saRNA strands may be identical in sequence to the saRNA delivered to the host cell or may otherwise comprise both the sequence encoding the replication proteins or peptides and the sequence encoding the antibody or antigen-binding fragment, and may be referred to as “complete replicated saRNA.” In other embodiments, the replicated positive saRNA strands may comprise the sequence encoding the antibody or antigen-binding fragment, but lack the sequence encoding the replication proteins or peptides, and may be referred to as “antibody- restricted replicated saRNA.” In some embodiments, both complete replicated saRNA and antibody-restricted replicated saRNA are produced in the human host cell.
The complete replicated saRNA or the antibody-restricted replicated saRNA, or both, may then be translated by cellular ribosomes to express the antibody or antigen-binding fragment.
“Trans-amplifying RNA” or “taRNA” refers to a collection of mRNAs in a taRNA therapeutic construct, that may function similarly to an saRNA therapeutic construct. Such a taRNA therapeutic construct may comprise multiple distinct mRNA molecules that collectively encode the replication proteins or peptides and the antibody or antigen-binding fragment that may be found in an saRNA therapeutic construct. For example, the taRNA therapeutic construct may comprise a first mRNA encoding the antibody or antigen-binding fragment and a second or additional mRNA encoding one or more replication proteins or peptides. For example, the taRNA therapeutic construct may comprise a distinct mRNA for each antibody or antigenbinding fragment and replication protein or peptide that might otherwise be found in an saRNA therapeutic construct.
In some embodiments, at least one taRNA in a taRNA therapeutic construct encodes at least one replication protein having a sequence with at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity to SEQ ID NO.: 762 or SEQ ID NO.: 763.
In further specific embodiments, the saRNA or the taRNA may be incapable of genomic insertion in the host cell, particularly in a human host cell in vivo.
In some embodiments, saRNA or taRNA may also be circRNA.
“Percent sequence identity” refers to a relationship between two or more sequences, as determined by comparing the sequences. Preferred methods to determine sequence identity are designed to give the best match between the sequences being compared. For example, the sequences are aligned for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second amino acid or nucleic acid sequence for optimal alignment). Further, non-homologous sequences may be disregarded for comparison purposes. The percent sequence identity referenced herein is calculated over the length of the reference sequence, unless indicated otherwise. Methods to determine sequence identity and similarity can be found in publicly available computer programs. Sequence alignments and percent identity calculations may be performed using a BLAST program (e.g., BLAST 2.0, BLASTP, BLASTN, or BLASTX). The mathematical algorithm used in the BLAST programs can be found in Altschul et al., Nucleic Acids Res. 25:3389-3402, 1997. Other examples include Clustal W, MAFFT, Clustal Omega, AlignMe, Praline, GAP, BESTFIT, Needle (EMBOSS), Stretcher (EMBOSS), GGEARCH2SEQ, Water (EMBOSS), Matcher (EMBOSS), LALIGN, and SSEARCH2SEQ. A global alignment algorithm, such as a Needleman and Wunsch algorithm, can be used to align two sequences over their entire length, maximizing the number of matches and minimizes the number of gaps. Default values can be used.
In certain embodiments, a BLAST program using default values is preferred.
To generate similarity scores for two amino acid sequences, scoring matrices can be used that assign positive scores for some non-identical amino acids (e.g., conservative amino acid substitutions, amino acids with similar physio-chemical properties, and/or amino acids that exhibit frequent substitutions in orthologs, homologs, or paralogs). Non-limiting examples of scoring matrices include PAM30, PAM70, PAM250, BLOSUM45, BLOSUM50, BLOUM62, BLOSUM80, and BLOSUM90.
Within the context of this disclosure, it will be understood that where sequence analysis software is used for analysis, the results of the analysis are based on the “default values” of the program referenced. “Default values” mean any set of values or parameters which originally load with the software when first initialized.
The term “isolated” means that the material is removed from its original environment (e.g., the natural environment if it is naturally occurring). For example, a naturally occurring nucleic acid or polypeptide present in a living animal is not isolated, but the same nucleic acid or polypeptide, separated from some or all of the co-existing materials in the natural system, is isolated. Such nucleic acid could be part of a vector and/or such nucleic acid or polypeptide
could be part of a composition (e.g., a cell lysate), and still be isolated in that such vector or composition is not part of the natural environment for the nucleic acid or polypeptide.
“Isolated” can, in some embodiments, also describe an antibody, antigen-binding fragment, polynucleotide, vector, host cell, or composition that is outside of a human body. In certain embodiments, an isolated antibody, antigen-binding fragment, polynucleotide, vector, host cell, or composition is provided.
The term “gene” means the segment of DNA or RNA involved in producing a polypeptide chain; in certain contexts, it includes regions preceding and following the coding region (e.g., 5’ untranslated region (UTR) and 3’ UTR) as well as intervening sequences (introns) between individual coding segments (exons).
A “functional variant” refers to a polypeptide or polynucleotide that is structurally similar or substantially structurally similar to a parent or reference compound of this disclosure, but differs slightly in composition (e.g., one base, atom or functional group is different, added, or removed), such that the polypeptide or encoded polypeptide is capable of performing at least one function of the parent polypeptide with at least 50% efficiency, preferably at least 55%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, at least 99.9%, or 100% level of activity of the parent polypeptide. In other words, a functional variant of a polypeptide or encoded polypeptide of this disclosure has “similar binding,” “similar affinity” or “similar activity” when the functional variant displays no more than a 50% reduction in performance in a selected assay as compared to the parent or reference polypeptide, such as an assay for measuring binding affinity (e.g., Biacore® or tetramer staining measuring an association (Ka) or a dissociation (KD) constant).
As used herein, a “functional portion” or “functional fragment” refers to a polypeptide or polynucleotide that comprises only a domain, portion or fragment of a parent or reference compound, and the polypeptide or encoded polypeptide retains at least 50% activity associated with the domain, portion or fragment of the parent or reference compound, preferably at least 55%, at least 60%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, at least or 99.9%, or 100% level of activity of the parent polypeptide, or provides a biological benefit (e.g., effector function). A “functional portion” or “functional fragment” of a polypeptide or encoded polypeptide of this disclosure has “similar binding” or “similar activity” when the functional portion or fragment displays no more than a 50% reduction in performance in a selected assay as compared to the
parent or reference polypeptide (preferably no more than 20% or 10%, or no more than a log difference as compared to the parent or reference with regard to affinity).
As used herein, the term “engineered,” “recombinant,” or “non-natural” refers to an organism, microorganism, cell, nucleic acid molecule, vector, DNA therapeutic, or RNA therapeutic that includes at least one genetic alteration or has been modified by introduction of an exogenous or heterologous nucleic acid molecule, wherein such alterations or modifications are introduced by genetic engineering (z.e., human intervention). Genetic alterations include, for example, modifications introducing expressible nucleic acid molecules encoding functional RNA, proteins, fusion proteins or enzymes, or other nucleic acid molecule additions, deletions, substitutions, or other functional disruption of a cell’s genetic material. Additional modifications include, for example, non-coding regulatory regions in which the modifications alter expression of a polynucleotide, gene, or operon.
As used herein, “heterologous” or “non-endogenous” or “exogenous” refers to any gene, protein, compound, nucleic acid molecule, or activity that is not native to a host cell or a subject, or any gene, protein, compound, nucleic acid molecule, or activity native to a host cell or a subject that has been altered. Heterologous, non-endogenous, or exogenous includes genes, proteins, compounds, or nucleic acid molecules that have been mutated or otherwise altered such that the structure, activity, or both is different as between the native and altered genes, proteins, compounds, or nucleic acid molecules. In certain embodiments, heterologous, non-endogenous, or exogenous genes, proteins, or nucleic acid molecules (e.g., receptors, ligands, etc.) may not be endogenous to a host cell or a subject, but instead nucleic acids encoding such genes, proteins, or nucleic acid molecules may have been added to a host cell by conjugation, transformation, transfection, electroporation, or the like, wherein the added nucleic acid molecule may integrate into a host cell genome or can exist as extra-chromosomal genetic material (e.g., as a plasmid or other self-replicating vector). The term “homologous” or “homolog” refers to a gene, protein, compound, nucleic acid molecule, or activity found in or derived from a host cell, species, or strain. For example, a heterologous or exogenous polynucleotide or gene encoding a polypeptide may be homologous to a native polynucleotide or gene and encode a homologous polypeptide or activity, but the polynucleotide or polypeptide may have an altered structure, sequence, expression level, or any combination thereof. A non-endogenous polynucleotide or gene, as well as the encoded polypeptide or activity, may be from the same species, a different species, or a combination thereof.
In certain embodiments, a nucleic acid molecule or portion thereof native to a host cell will be considered heterologous to the host cell if it has been altered or mutated, or a nucleic acid molecule native to a host cell may be considered heterologous if it has been altered with a heterologous expression control sequence or has been altered with an endogenous expression control sequence not normally associated with the nucleic acid molecule native to a host cell. In addition, the term “heterologous” can refer to a biological activity that is different, altered, or not endogenous to a host cell. As described herein, more than one heterologous nucleic acid molecule can be introduced into a host cell as separate nucleic acid molecules, as a plurality of individually controlled genes, as a polycistronic nucleic acid molecule, as a single nucleic acid molecule encoding a fusion protein, or any combination thereof.
As used herein, the term “endogenous” or “native” refers to a polynucleotide, gene, protein, compound, molecule, or activity that is normally present in a host cell or a subject.
The term “expression”, as used herein, refers to the process by which a polypeptide is produced based on the encoding sequence of a nucleic acid molecule, such as a gene. The process may include transcription, post-transcriptional control, post-transcriptional modification, translation, post-translational control, post-translational modification, or any combination thereof. An expressed nucleic acid molecule is typically operably linked to an expression control sequence (e.g., a promoter).
The term “operably linked” refers to the association of two or more nucleic acid molecules on a single nucleic acid fragment so that the function of one is affected by the other. For example, a promoter is operably linked with a coding sequence when it is capable of affecting the expression of that coding sequence (i.e., the coding sequence is under the transcriptional control of the promoter). “Unlinked” means that the associated genetic elements are not closely associated with one another and the function of one does not affect the other.
As described herein, more than one heterologous nucleic acid molecule can be introduced into a host cell as separate nucleic acid molecules, as a plurality of individually controlled genes, as a polycistronic nucleic acid molecule, as a single nucleic acid molecule encoding a protein (e.g., a heavy chain of an antibody), or any combination thereof. When two or more heterologous nucleic acid molecules are introduced into a host cell, it is understood that the two or more heterologous nucleic acid molecules can be introduced as a single nucleic acid molecule (e.g., on a single vector), on separate vectors, integrated into the host chromosome at a single site or multiple sites, or any combination thereof. The number of referenced heterologous nucleic acid molecules or protein activities refers to the number of encoding nucleic acid molecules or
the number of protein activities, not the number of separate nucleic acid molecules introduced into a host cell.
The term “construct” refers to any polynucleotide that contains a recombinant nucleic acid molecule (or, when the context clearly indicates, a fusion protein of the present disclosure). A (polynucleotide) construct may be present in a vector (e.g., a bacterial vector, a viral vector) or may be integrated into a genome. A “vector” is a nucleic acid molecule that is capable of transporting another nucleic acid molecule. Vectors may be, for example, plasmids, cosmids, viruses, a RNA vector or a linear or circular DNA or RNA molecule that may include chromosomal, non-chromosomal, semi -synthetic or synthetic nucleic acid molecules. Vectors of the present disclosure also include transposon systems (e.g., Sleeping Beauty, see, e.g., Geurts et al., Mol. Ther. 5: 108, 2003: Mates et al., Nat. Genet. 41'.753, 2009). Exemplary vectors are those capable of autonomous replication (episomal vector), capable of delivering a polynucleotide to a cell genome (e.g., viral vector), or capable of expressing nucleic acid molecules to which they are linked (expression vectors).
As used herein, “expression vector” or “vector” refers to a DNA or RNA construct containing a nucleic acid molecule that is operably linked to a suitable control sequence capable of effecting the expression of the nucleic acid molecule in a suitable host. Such control sequences include a promoter to effect transcription, an optional operator sequence to control such transcription, a sequence encoding suitable mRNA ribosome binding sites, and sequences which control termination of transcription and translation. The vector may be a plasmid, a phage particle, a virus, or simply a potential genomic insert. Once transformed into a suitable host, the vector may replicate and function independently of the host genome, or may, in some instances, integrate into the genome itself or deliver the polynucleotide contained in the vector into the genome without the vector sequence. In the present specification, “plasmid,” “expression plasmid,” “virus,” and “vector” are often used interchangeably.
In some embodiments, the “vector” may comprise or consist of mRNA, such as saRNA, taRNA, or circRNA.
The term “introduced” in the context of inserting a nucleic acid molecule into a cell, means “transfection”, “transformation,” or “transduction” and includes reference to the incorporation of a nucleic acid molecule into a eukaryotic or prokaryotic cell wherein the nucleic acid molecule may be incorporated into the genome of a cell (e.g., chromosome, plasmid, plastid, or mitochondrial DNA), converted into an autonomous replicon (e.g. a replicon formed by
saRNA), or transiently expressed e.g., transfected mRNA, such as circRNA, taRNA, or saRNA).
In certain embodiments, polynucleotides of the present disclosure may be operatively linked to certain elements of a vector. For example, polynucleotide sequences that are needed to effect the expression and processing of coding sequences to which they are ligated may be operatively linked. Expression control sequences may include appropriate transcription initiation, termination, promoter, and enhancer sequences; efficient RNA processing signals such as splicing and polyadenylation signals; sequences that stabilize cytoplasmic mRNA; sequences that enhance translation efficiency (z.e., Kozak consensus sequences); sequences that enhance protein stability; and possibly sequences that enhance protein secretion. Expression control sequences may be operatively linked if they are contiguous with the gene of interest and expression control sequences that act in trans or at a distance to control the gene of interest.
In certain embodiments, the vector comprises a plasmid vector or a viral vector (e.g., a lentiviral vector or a y-retroviral vector). Viral vectors include retrovirus, adenovirus, parvovirus (e.g., adeno-associated viruses), coronavirus, negative strand RNA viruses such as orthomyxovirus (e.g., influenza virus), rhabdovirus (e.g., rabies and vesicular stomatitis virus), paramyxovirus (e.g., measles and Sendai), positive strand RNA viruses such as picornavirus and alphavirus, and double-stranded DNA viruses including adenovirus, herpesvirus (e.g., Herpes Simplex virus types 1 and 2, Epstein-Barr virus, cytomegalovirus), and poxvirus (e.g., vaccinia, fowlpox, and canarypox). Other viruses include, for example, Norwalk virus, togavirus, flavivirus, reoviruses, papovavirus, hepadnavirus, and hepatitis virus. Examples of retroviruses include avian leukosis-sarcoma, mammalian C-type, B-type viruses, D type viruses, HTLV-BLV group, lentivirus, spumavirus (Coffin, J. M., Retroviridae: The viruses and their replication, In Fundamental Virology, Third Edition, B. N. Fields et al., Eds., Lippincott-Raven Publishers, Philadelphia, 1996).
“Retroviruses” are viruses having an RNA genome, which is reverse-transcribed into DNA using a reverse transcriptase enzyme, the reverse-transcribed DNA is then incorporated into the host cell genome. “Gammaretrovirus” refers to a genus of the retroviridae family. Examples of gammaretroviruses include mouse stem cell virus, murine leukemia virus, feline leukemia virus, feline sarcoma virus, and avian reticuloendotheliosis viruses.
“Lentiviral vectors” include HIV-based lentiviral vectors for gene delivery, which can be integrative or non-integrative, have relatively large packaging capacity, and can transduce a range of different cell types. Lentiviral vectors are usually generated following transient
transfection of three (packaging, envelope, and transfer) or more plasmids into producer cells. Like HIV, lentiviral vectors enter the target cell through the interaction of viral surface glycoproteins with receptors on the cell surface. On entry, the viral RNA undergoes reverse transcription, which is mediated by the viral reverse transcriptase complex. The product of reverse transcription is a double-stranded linear viral DNA, which is the substrate for viral integration into the DNA of infected cells.
In certain embodiments, the viral vector can be a gammaretrovirus, e.g., Moloney murine leukemia virus (MLV)-derived vectors. In other embodiments, the viral vector can be a more complex retrovirus-derived vector, e.g., a lentivirus-derived vector. HIV-l-derived vectors belong to this category. Other examples include lentivirus vectors derived from HIV-2, FIV, equine infectious anemia virus, SIV, and Maedi-Visna virus (ovine lentivirus). Methods of using retroviral and lentiviral viral vectors and packaging cells for transducing mammalian host cells with viral particles containing transgenes are known in the art and have been previous described, for example, in: U.S. Patent 8,119,772; Walchli et al., PLoS One 6:327930, 2011; Zhao et al., J. Immunol. 174 AM5, 2005; Engels etal., Hum. Gene Ther. 74: 1155, 2003; Frecha et al., Mol. Ther. 18: 1748, 2010; and Verhoeyen et al ., Methods Mol. Biol. 506:97 , 2009. Retroviral and lentiviral vector constructs and expression systems are also commercially available. Other viral vectors also can be used for polynucleotide delivery including DNA viral vectors, including, for example adenovirus-based vectors and adeno-associated virus (AAV)-based vectors; vectors derived from herpes simplex viruses (HSVs), including amplicon vectors, replication-defective HSV and attenuated HSV (Krisky et al., Gene Ther. 5: 1517, 1998).
Other vectors that can be used with the compositions and methods of this disclosure include those derived from baculoviruses and a-viruses. (Jolly, D J. 1999. Emerging Viral Vectors, pp 209-40 in Friedmann T. ed. The Development of Human Gene Therapy. New York: Cold Spring Harbor Lab), or plasmid vectors (such as sleeping beauty or other transposon vectors).
When a viral vector genome comprises a plurality of polynucleotides to be expressed in a host cell as separate transcripts, the viral vector may also comprise additional sequences between the two (or more) transcripts allowing for bicistronic or multi ci stronic expression. Examples of such sequences used in viral vectors include internal ribosome entry sites (IRES), furin cleavage sites, viral 2A peptide, or any combination thereof.
Plasmid vectors, including DNA-based antibody or antigen-binding fragment-encoding plasmid vectors for direct administration to a subject, are described further herein.
As used herein, a “carrier” or “vehicle” can refer to a molecule able to introduce a polynucleotide into a host cell. In specific embodiments, the carrier may comprise a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, a lipid nanoparticle (LNP), a liopolyplex (LPP), a cationic polypeptide, a polymeric nanoparticle, or a nanoscale platform, such as a nanoemulsion. (see, e.g., Li et al. Wilery Interdiscip Rev. Nanomed Nanobiotechnol. 77(2):el530 (2019), incorporated by reference herein with respect to its description of carriers and vehicles and methods of making and using such carriers and vehicles to form therapeutics).
Principles, reagents, and techniques for designing appropriate mRNA and formulating mRNA-LNP and delivering the same are described in, for example, Pardi et al. (J Control Release 277345-351 (2015)); Thess et al. (Mol Ther 23: 1456-1464 (2015)); Thran et al. (EMBO Mol Med 9(10): 1434-1448 (2017); Kose et al. (Set. Immunol. 4 eaaw6647 (2019); and Sabnis et al. (Mol. Ther. 26: 1509-1519 (2018)), which techniques, including capping, codon optimization, nucleoside modification, purification of mRNA, incorporation of the mRNA into stable lipid nanoparticles (e.g., ionizable cationic lipid/phosphatidylcholine/cholesterol/PEG-lipid; ionizable lipid:distearoyl PC:cholesterol:polyethylene glycol lipid), and subcutaneous, intramuscular, intradermal, intravenous, intraperitoneal, and intratracheal administration of the same, are incorporated herein by reference.
In some embodiments, LNPs may be formed from a cationic lipid, in particular an ionizable lipid. In other embodiments, LNPs may be formed from lipidoids, which are molecules derived from ionizable lipids and dendrimers. LNPs, in some embodiments, may further include structural lipids, such as a phospholipid, cholesterol, or both. In some embodiments, the LNPs may comprise i) an ionizable lipid having a positive charge able to bind to the negatively charged RNA backbone, ii) a PEGylated lipid, iii) a cholesterol, and iv) a phospholipid.
As used herein, an “RNA therapeutic” is an RNA therapeutic construct or naked RNA, including naked circRNA, taRNA, or saRNA. An “RNA therapeutic construct” comprises an RNA molecule and a carrier.
In some embodiments, the RNA therapeutic construct may “self assemble” from a mixture comprising the RNA and the carrier or carrier precursors, such as smaller lipid vesicles. Self-assembly may particularly occur with LNP carriers.
A “circRNA therapeutic construct” comprises circRNA and a carrier. circRNA may present different behaviors in the formation and function of a circRNA therapeutic construct than are observed with respect to corresponding linear mRNA in the presence of the same carrier. For example, due to the circular nature of circRNA, the number of copies that can be contained in a carrier may be different than the number of copies possible using corresponding linear mRNA. As another example, the conditions that result in encapsulation of circRNA in a carrier, or its release in vivo from the carrier, may also be different than the conditions applicable to corresponding linear mRNA.
A “taRNA therapeutic construct” comprises at least two distinct taRNAs, a first taRNA encoding a replication protein and a second taRNA encoding a protein or peptide of interest, typically an antibody or antigen-binding fragment thereof. In some embodiments, the second taRNA may encode a VH-containing protein and the taRNA therapeutic construct may further include a third taRNA that encodes a VL-containing protein.
An “saRNA therapeutic construct” comprises an saRNA and a carrier. saRNA, due to the inclusion of sequences encoding replication proteins or peptides, is often larger than mRNA encoding only a protein or peptide of interest, and saRNA therapeutic constructs may therefore benefit from the use of carriers functional with longer RNA molecules, such as carriers, particularly LNPs, comprising polyethylenimine (PEI).
In some embodiments, the saRNA therapeutic construct may include at least 10, 50, or 100, or between 10 and 50, between 10 and 100, or between 50 and 100 copies of the saRNA per carrier.
As used herein a “DNA therapeutic” is a DNA therapeutic construct, or naked DNA, including naked DNA vectors. A “DNA therapeutic construct” comprises a DNA molecule and a carrier.
As used herein, the term “host” refers to a cell or microorganism targeted for genetic modification with a heterologous nucleic acid molecule to produce a polypeptide of interest (e.g., an antibody of the present disclosure).
A host cell may include any individual cell or cell culture which may receive a vector or the incorporation of nucleic acids or express proteins. The term also encompasses progeny of the host cell, whether genetically or phenotypically the same or different. Suitable host cells may depend on the vector and may include mammalian cells, animal cells, human cells, simian cells, insect cells, yeast cells, and bacterial cells. These cells may be induced to incorporate the vector or other material by use of a viral vector, transformation via calcium phosphate precipitation,
DEAE-dextran, electroporation, microinjection, or other methods. See, for example, Sambrook el al., Molecular Cloning: A Laboratory Manual 2d ed. (Cold Spring Harbor Laboratory, 1989).
In embodiments relating to RNA therapeutics, including circRNA therapeutics, taRNA therapeutics, or saRNA therapeutics, and DNA therapeutics the host cell may be a human cell. In some more specific embodiments, the host cell may be a cell able to produce antibodies prior to introduction of the RNA or DNA, such as a B cell. In other more specific embodiments, the host cell may be a cell not otherwise able to produce antibodies prior to introduction of the RNA or DNA, such as a muscle cell.
In embodiments relating to RNA therapeutics or DNA therapeutics that are not produced using a cell-free system, at least two types of host cells may exist, a “production host cell” used to produce RNA or DNA for the DNA therapeutic, and an “/// vivo human host cell” into which the RNA or DNA is introduced.
In the context of a bacterial (e.g., Staphylococcus or other bacteria expressing an antigen as described herein) infection, a “host” refers to a cell or a subject infected with the bacteria.
“Antigen” or “ Ag”, as used herein, refers to an immunogenic molecule that provokes an immune response. This immune response may involve antibody production, activation of specific immunologically competent cells, activation of complement, antibody dependent cytotoxicicity, or any combination thereof. An antigen (immunogenic molecule) may be, for example, a peptide, glycopeptide, polypeptide, glycopolypeptide, polynucleotide, polysaccharide, lipid, or the like. It is readily apparent that an antigen can be synthesized, produced recombinantly, or derived from a biological sample. Exemplary biological samples that can contain one or more antigens include tissue samples, stool samples, cells, biological fluids, or combinations thereof. Antigens can be produced by cells that have been modified or genetically engineered to express an antigen. Antigens can also be expressed or presented on the surface of a cell infected by a bacteria.
The term “epitope” or “antigenic epitope” includes any molecule, structure, amino acid sequence, or protein determinant that is recognized and specifically bound by a cognate binding molecule, such as an immunoglobulin, or other binding molecule, domain, or protein. Epitopic determinants generally contain chemically active surface groupings of molecules, such as amino acids or sugar side chains, and can have specific three-dimensional structural characteristics, as well as specific charge characteristics. Where an antigen is or comprises a peptide or protein, the epitope can be comprised of consecutive amino acids (e.g., a linear epitope), or can be comprised of amino acids from different parts or regions of the protein that are brought into proximity by
protein folding (e.g., a discontinuous or conformational epitope), or non-contiguous amino acids that are in close proximity irrespective of protein folding.
Antibodies, Antigen-Binding Fragments, and Compositions
In certain embodiments, the present disclosure provides an (e.g, isolated) antibody, or an antigen-binding fragment thereof, that is capable of binding to an antigen selected from: a lipoteichoic acid (LTA), e.g., a Type I LT A, a Type II LT A, a Type IV LT A, or any combination thereof; a glucosaminidase (Gmd); a Pore Forming Toxin (PFT) such as a leukocidin (e.g., LukD, LukF, LukE, or LukS, or any combination thereof, such as LukED, LukSF, or both), an alpha toxin (e.g., Hla; an alpha toxin may also be referred to as an alpha hemolysin), or a gamma hemolysin (also referred to as a gamma toxin; e.g., HIgA and/or HIgB); a Staphylococcal Protein A (SpA); a clumping factor A (ClfA); a Sbi; or any combination of two or more of the foregoing. The antigen can be referred to as a “ Staphylococcus antigen” and the antibody or antigen-binding fragment can be referred to as an “ n -Staphylococcus” antibody or antigen-binding fragment (or “ r\ -Staphylococcus aureus _antibody or antigen-binding fragment”), and/or as an “anti- Staphylococcus antigen” antibody or antigen-binding fragment. Any antibody or antigenbinding fragment of the present disclosure (including a multispecific antibody or antigen-binding fragment) can be described as an “ n -Staphylococcus” antibody or antigen-binding fragment, and/or as an ''&n A-Staphylococcus antigen” antibody or antigen-binding fragment.
In certain embodiments, the Staphylococcus antigen is an antigen that is also expressed by one or more (e.g., gram-positive) bacteria that is not of genus Staphylococcus, and the antibody or antigen-binding fragment is capable of binding thereto. Accordingly, the term “ Staphylococcus antigen” is not limited to antigens expressed solely by bacteria of genus Staphylococcus and includes antigens that are expressed by Staphylococcus and by bacteria of one or more other genus. In certain embodiments, the antigen is also expressed by a bacteria of genus Enterococcus (e.g., E. faecalis), a bacteria of genus Lactococcus (e.g., L. garvieae), a bacteria of genus Streptococcus (e.g., S. pyogenes, S. agalacliae) a bacteria of genus Listeria (e.g., L. monocytogenes), a bacteria of genus Clostridium (e.g., C. inocuum, C. difficile), or any combination thereof. For example, a Type I LTA can be found on Staphylococcus aureus and other Staphylococci, as well as on Enterococci (e.g., E. faecalis), L. monocytogenes, Streptococcus pyogenes, and Streptococcus agalactiae,' a Type I LTA of any of these bacteria can be referred to as a “ Staphylococcus antigen” or a “ Staphylococcus LTA”.
Accordingly, an ^nii-Staphylococcus antibody or antigen-binding fragment can, in certain embodiments, bind to the antigen (such as: a lipoteichoic acid (LTA), e.g., a Type I LTA, a Type II LTA, a Type IV LTA, or any combination thereof; a glucosaminidase (Gmd); a Pore Forming Toxin (PFT) such as a leukocidin (e.g., LukD, LukF, LukE, or LukS, or any combination thereof, such as LukED, LukSF, or both), an alpha toxin (e.g., Hla; an alpha toxin may also be referred to as an alpha hemolysin), a gamma hemolysin (also referred to as a gamma toxin; e.g., HIgA and/or HIgB); a Staphylococcal Protein A (SpA); a clumping factor A (ClfA); or a Sbi) when the antigen is from a bacteria that is not of the Staphylococcus genus. For example, an anti- Staphylococcus antibody or antigen-binding fragment can, in certain embodiments, bind to an antigen-expressing bacteria of genus Enterococcus (e.g., E. faecalis), of genus Lactococcus (e.g., L. garviae), of genus Streptococcus (e.g., S. pyogenes, S. agalactiae) of genus Listeria (e.g., L. monocytogenes), of genus Clostridium (e.g., C. inocuum, C. difficile), or any combination thereof.
An antibody or antigen-binding fragment may also be described with reference to the target(s) to which it binds. For example, the present disclosure provides, in certain embodiments, an “anti-LTA” antibody or antigen-binding fragment, an “anti-alpha toxin” antibody or antigen-binding fragment, an “anti-Gmd” antibody or antigen-binding fragment, an “anti-PFT” (or anti -alpha toxin or anti-alpha hemolysin or anti-leukotoxin or anti-leukocidin or anti-gamma toxin or anti-gamma hemolysin) antibody or antigen-binding fragment, an “anti- SpA” antibody or antigen-binding fragment, an “anti-ClfA” antibody or antigen-binding fragment, and an “anti-Sbi” antibody or antigen-binding fragment. In certain embodiments, an antibody or antigen-binding fragment of the present disclosure associates with or unites with an antigen (lipoteichoic acid (LTA), e.g., a Type I LTA, a Type II LTA, a Type IV LTA, or any combination thereof;; a glucosaminidase (Gmd); Pore Forming Toxin (PFT) such as a leukocidin (e.g., LukED, a LukSF, or both), an alpha toxin (e.g., Hla), or a gamma toxin or gamma hemolysin (e.g., HIgAB); a Staphylococcal Protein A (SpA); a clumping factor A (ClfA); or a Sbi) while not significantly associating or uniting with any other molecules or components in a sample.
In certain embodiments, an antibody or antigen-binding fragment of the present disclosure specifically binds to an antigen selected from: lipoteichoic acid (LTA), e.g., a Type I LTA, a Type II LTA, a Type IV LTA, or any combination thereof; a glucosaminidase (Gmd); Pore Forming Toxin (PFT) such as a leukocidin (e.g., LukED, a LukSF, or both), an alpha toxin (e.g., Hla), or a gamma toxin or gamma hemolysin (e.g., HIgAB); a Staphylococcal Protein A
(SpA); a clumping factor A (ClfA); and a Sbi. As used herein, “specifically binds” refers to an association or union of an antibody or antigen-binding fragment to an antigen with an affinity or Ka (z.e., an equilibrium association constant of a particular binding interaction with units of 1/M) equal to or greater than 105 M'1 (which equals the ratio of the on-rate [Kon] to the off rate [Koir] for this association reaction), while not significantly associating or uniting with any other molecules or components in a sample. Alternatively, affinity may be defined as an equilibrium dissociation constant (Ka) of a particular binding interaction with units of M (e.g., 10'5 M to 10'13 M). Antibodies may be classified as “high-affinity” antibodies or as “low-affinity” antibodies. “High-affinity” antibodies refer to those antibodies having a Ka of at least 107M-1, at least 108 M’ at least 109 M’1, at least IO10 M’1, at least 1011 M’1, at least 1012M-1, or at least 1013 M’1. “Low- affinity” antibodies refer to those antibodies having a Ka of up to 107M-1, up to 106 M’1, up to 105 M’1. Alternatively, affinity may be defined as an equilibrium dissociation constant (Ka) of a particular binding interaction with units of M (e.g., 10'5 M to 10'13 M).
A variety of assays are known for identifying antibodies of the present disclosure that bind a particular target, as well as determining binding domain or binding protein affinities, such as Western blot, ELISA (e.g., direct, indirect, or sandwich), analytical ultracentrifugation, spectroscopy, biolayer interferometry, and surface plasmon resonance (Biacore®) analysis (see, e.g., Scatchard et al., Ann. N.Y. Acad. Sci. 57:660, 1949; Wilson, Science 295:2103, 2002; Wolff etal., Cancer Res. 53:2560, 1993; and U.S. Patent Nos. 5,283,173, 5,468,614, or the equivalent). Assays for assessing affinity or apparent affinity or relative affinity are also known.
In certain examples, binding can be determined by recombinantly expressing an antigen in a host cell (e.g, by transfection) and immunostaining the (e.g, fixed, or fixed and permeabilized) host cell with antibody and analyzing binding by flow cytometry (e.g., using a ZE5 Cell Analyzer (BioRad®) and FlowJo software (TreeStar). In some embodiments, positive binding can be defined by differential staining by antibody of antigen-expressing cells versus control (e.g., mock) cells.
In some embodiments an antibody or antigen-binding fragment of the present disclosure binds to a herein disclosed antigen, as measured using biolayer interferometry, or by surface plasmon resonance.
Certain characteristics of presently disclosed antibodies or antigen-binding fragments may be described using IC50 or EC50 values. In certain embodiments, the IC50 is the concentration of a composition (e.g., antibody) that results in half-maximal inhibition of the indicated biological or biochemical function, activity, or response. In certain embodiments, the
EC50 is the concentration of a composition that provides the half-maximal response in the assay. In some embodiments, e.g., for describing the ability of a presently disclosed antibody or antigen-binding fragment to neutralize infection by a bacterial (e.g., Staphylococcus), IC50 and EC 50 are used interchangeably.
In certain embodiments, an antibody of the present disclosure is capable of neutralizing infection by, and/or of neutralizing one or more activity of, an antigen-expressing bacteria. As used herein, a “neutralizing antibody” is one that can neutralize, i.e., prevent, inhibit, reduce, impede, or interfere with, the ability of a pathogen to initiate and/or perpetuate an infection in a host, and/or prevent, inhibit, reduce, impede, or interfere with, the ability of a pathogen to perform one or more function, such as virulence, synthesis of a cell wall, cell wall turnover, cell division, cell separation, immune evasion, binding to fibrinogen, bacterial clumping, forming pores on a host cell surface, disrupting a host cell plasma membrane, and/or causing or initiating cell death. The terms “neutralizing antibody” and “an antibody that neutralizes” or “antibodies that neutralize” are used interchangeably herein. In any of the presently disclosed embodiments, the antibody or antigen-binding fragment can be capable of preventing and/or neutralizing infection and/or activity of an antigen-expressing bacteria in an in vitro model and/or in an in vivo animal model and/or in a human.
In certain embodiments, the antibody, or antigen-binding fragment thereof, is human, humanized, or chimeric.
In certain embodiments, the antigen is expressed on the surface of a host cell (e.g., a CHO cell) and binding to antigen is according to flow cytometry.
In certain embodiments, the antibody or antigen-binding fragment is capable of binding to the antigen with a KD in a range from about 1.0E-9 M to about 3.5E-12 M, such as, for example, about 1.0E-9 M, about 1.5E-9 M, about 2.0E-9 M, about 2.5E-9 M, about 3.0E-9 M, about 3.5E-9 M, about 4.0E-9 M, about 4.5E-9 M, about 5.0E-9 M, about 5.5E-9 M, about 6.0E-
9 M, about 6.5E-9 M, about 7.0E-9 M, about 7.5E-9 M, about 8.0E-9 M, about 9.5E-9 M, about 10.0E-9M, about 1.0E-10 M, about 1.5E-10 M, about 2.0E-10 M, about 2.5E-10 M, about 3.0E-
10 M, about 3.5E-10 M, about 4.0E-10 M, about 4.5E-10 M, about 5.0E-10 M, about 5.5E-10 M, about 6.0E-10 M, about 6.5E-10 M, about 7.0E-10 M, about 7.5E-10 M, about 8.0E-10 M, about 9.5E-10 M, about 10.0E-10 M, about 1.0E-11 M, about 1.5E-11 M, about 2.0E-11 M, about 2.5E-11 M, about 3.0E-11 M, about 3.5E-11 M, about 4.0E-11 M, about 4.5E-11 M, about 5.0E-
11 M, about 5.5E-11 M, about 6.0E-11 M, about 6.5E-11 M, about 7.0E-11 M, about 7.5E-11 M, about 8.0E-11 M, about 9.5E-11 M, about 10.0E-11 M, about 1.0E-12 M, about 1.5E-12 M,
about 2.0E-12 M, about 2.5E-12 M, about 3.0E-12 M, or about 3.5E-12 M. In some embodiments, the binding is as assessed by biolayer interferometry (BLI). In other embodiments, the binding is as assessed by surface plasmon resonance (SPR).
In certain embodiments, the antibody or antigen-binding fragment is capable of activating a human FcyRIIIa. In further embodiments, activation is as determined using a host cell (optionally, a Jurkat cell) comprising: (i) the human FcyRIIIa (optionally, a F158 allele); and (ii) a NF AT expression control sequence operably linked to a sequence encoding a reporter, such as a luciferase reporter, following incubation (e.g., of 23 hours) of the antibody or antigen-binding fragment with a target cell (e.g., a A549 cell) infected with an antigen-expressing bacteria. In certain embodiments, the antibody or antigen-binding fragment is capable of treating and/or preventing an infection by an antigen-expressing bacteria in a subject.
Table 1 lists certain antibodies of the present disclosure, along with target(s) to which the antibodies bind.
Terms understood by those in the art of antibody technology are each given the meaning acquired in the art, unless expressly defined differently herein. For example, the term “antibody” refers to an intact antibody comprising at least two heavy (H) chains and two light (L) chains inter-connected by disulfide bonds, as well as any antigen-binding portion or fragment of an intact antibody that has or retains the ability to bind to the antigen target molecule recognized by the intact antibody, such as an scFv, Fab, or Fab’2 fragment. Thus, the term “antibody” herein is used in the broadest sense and includes polyclonal and monoclonal antibodies, including intact antibodies and functional (antigen-binding) antibody fragments thereof, including fragment antigen binding (Fab) fragments, F(ab’)2 fragments, Fab’ fragments, Fv fragments, recombinant IgG (rlgG) fragments, single chain antibody fragments, including single chain variable fragments (scFv), and single domain antibodies (e.g., sdAb, sdFv, nanobody) fragments. The term encompasses genetically engineered and/or otherwise modified forms of immunoglobulins, such as intrabodies, peptibodies, chimeric antibodies, fully human antibodies, humanized antibodies, and heteroconjugate antibodies, multispecific, e.g., bispecific antibodies, diabodies, triabodies, tetrabodies, tandem di-scFv, and tandem tri-scFv. Unless otherwise stated, the term “antibody” should be understood to encompass functional antibody fragments thereof. The term also encompasses intact or full-length antibodies, including antibodies of any class or sub-class, including IgG and sub-classes thereof (IgGl, IgG2, IgG3, IgG4), IgM, IgE, IgA, and IgD.
The terms “VL” or “VL” and “VH” or “VH” refer to the variable binding region (also called variable region or variable binding domain or variable domain) from an antibody light chain and an antibody heavy chain, respectively. In certain embodiments, a VL is a kappa (K) class (also “VK” herein). In certain embodiments, a VL is a lambda ( ) class. The variable binding regions comprise discrete, well-defined sub-regions known as “complementarity determining regions” (CDRs) and “framework regions” (FRs). The terms “complementarity determining region,” and “CDR,” are synonymous with “hypervariable region” or “HVR,” and refer to sequences of amino acids within antibody variable regions, which, in general, together confer the antigen specificity and/or binding affinity of the antibody, wherein consecutive CDRs (z.e., CDR1 and CDR2, CDR2 and CDR3) are separated from one another in primary structure by a framework region. There are three CDRs in each variable region (HCDR1, HCDR2, HCDR3; LCDR1, LCDR2, LCDR3; also referred to as CDRHs and CDRLs, respectively). In certain embodiments, an antibody VH comprises four FRs and three CDRs as follows: FR1- HCDR1-FR2-HCDR2-FR3-HCDR3-FR4; and an antibody VL comprises four FRs and three CDRs as follows: FR1-LCDR1-FR2-LCDR2-FR3-LCDR3-FR4. In general, the VH and the VL
together form the antigen-binding site (or domain) through their respective CDRs. In certain embodiments, one or more CDRs do not contact antigen and/or do not contribute energetically to antigen binding (but at least one CDR does contact antigen).
As used herein, a “variant” of a CDR refers to a functional variant of a CDR sequence having up to 1-3 amino acid substitutions (e.g., conservative or non-conservative substitutions), deletions, or combinations thereof.
Numbering of CDR and framework regions may be according to any known method or scheme, such as the Kabat, Chothia, EU, IMGT, Contact, North, Martin, AbM, and Aho numbering schemes see, e.g., Kabat et al., “Sequences of Proteins of Immunological Interest, US Dept. Health and Human Services, Public Health Service National Institutes of Health, 1991, 5th ed.; Chothia and Lesk, J. Mol. Biol. 796:901-917 (1987)); Lefranc etal., Dev. Comp. Immunol. 27:55, 2003; Honegger and Pliickthun, J. Mol. Bio. 309:657-670 (2001); North et al. J Mol Biol. (2011) 706:228-56; doi: 10.1016/j.jmb.2010.10.030; Abhinandan and Martin, Mol Immunol. (2008) 45:3832-9. 10.1016/j.molimm.2008.05.022). The antibody and CDR numbering systems of these references are incorporated herein by reference. Equivalent residue positions can be annotated and for different molecules to be compared using Antigen receptor Numbering And Receptor Classification (ANARCI) software tool (2016, Bioinformatics 15:298- 300). Accordingly, identification of CDRs of an exemplary variable domain (VH or VL) sequence as provided herein according to one numbering scheme is not exclusive of an antibody comprising CDRs of the same variable domain as determined using a different numbering scheme.
In certain embodiments, an antibody or an antigen-binding fragment of the present disclosure comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein each CDR is independently selected from a corresponding CDR of an antigen-specific antibody as provided in Table 1, Table 2, Table 3, or Table 4. That is, all combinations of CDRs from antigen-specific antibodies provided in Table 1 and/or Table 2 and/or Table 3 and/or Table 4 are contemplated. In certain embodiments, an antibody or an antigen-binding fragment of the present disclosure comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3 of any one of the antibodies shown in Table 1 or 2.
In some embodiments, CDRs are in accordance with the IMGT numbering method. Table 2 summarizes IMGT CDR amino acid sequences (SEQ ID NOs.) of certain antibodies of the present disclosure.
Table 3 summarizes variable domain amino acid sequences and examples of corresponding codon-optimized polynucleotide sequences (SEQ ID NOS.) of certain antibodies of the present disclosure. Table 3. Amino acid and certain polynucleotide sequences (SEQ ID NOs.) of variable domains of certain antibodies
In some embodiments, an antibody or antigen-binding fragment comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and/or a CDRL3, or functional variants thereof (and in certain embodiments, comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a
CDRL3, or functional variants thereof) of the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively. In some embodiments, an antibody or antigen-binding fragment comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, and optionally a VH and a VL, of one of the following antibodies as set forth in Table 2 and Table 3: SSG20; SSC35; SSE73; SSA12; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSE1; SSE158; SSF2; SSF11; SSF10; SSB18; SSB24; SSB54; SSB65; SSB67; SSH3; SSF33; SSL5; SSL17; SSL35; SSG50; SSG54; SSM47; SSG44; SSG48; SSM37; SSH4; SSH7; SSG27; SSG39; SSM45;
SSF52; SSG51; SSF34; SSG59; SSAA8; SSAA26; SSAB5; SSAI3; SSAI8; SSAI2; SSAF1;
SSAL2; SSAM6; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; SSF69; SSN2; SSN6; SSH8; SSM35; SSM36; and SSM40. In some embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the IMGT numbering system (optionally using the junction definitions for CDR3 sequences). In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Kabat numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Chothia numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Enhanced Chothia (also referred to as “Martin”) numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the AHo numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the North numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the Contact numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the EU numbering system. In other embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to the AbM numbering system (AbM antibody modelling software from Oxford Molecular). In some embodiments, the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 are according to a combination of any two or more of IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact. In some embodiments, the two or more numbering systems combined produce the CDR definition having the greatest length of the CDR according to any numbering system or combination of numbering systems.
In some embodiments, an antibody or antigen-binding fragment comprises:
(i) a CDRH1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.: 42, 32, 122, 22, 132, 142, 152, 162, 242, 172, 2, 12, 182, 52, 192, 212, 102, 222, 112, 232, 82, 92, 62, 72, 202, 281, 291, 301, 311, 321, 331, 341, 351, 361, 371, 381, 391, 401, 411, 421, 433, 443, 453, 463, 473, 483, 493, 503, 513, 523, 533, 543, 553, 563, 573, 583, 593, 603, 613, 623, 633, 643, 653, 663, 673, 683, 693, 703, 713, 723, 733, 743, and 753, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid;
(ii) a CDRH2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs. :43, 33, 123, 23, 133, 143, 153, 163, 243, 173, 3,
13, 183, 53, 193, 213, 103, 223, 113, 233, 83, 93, 63, 73, 203, 282, 292, 302, 312, 322, 332, 342,
352, 362, 372, 382, 392, 402, 412, 422, 434, 444, 454, 464, 474, 484, 494, 504, 514, 524, 534,
544, 554, 564, 574, 584, 594, 604, 614, 624, 634, 644, 654, 664, 674, 684, 694, 704, 714, 724,
734, 744, and 754, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid;
(iii) a CDRH3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.: 44, 34, 124, 24, 134, 144, 154, 164, 244, 174, 4,
14, 184, 54, 194, 214, 104, 224, 114, 234, 84, 94, 64, 74, 204, 283, 293, 303, 313, 323, 333, 343,
353, 363, 373, 383, 393, 403, 413, 423, 435, 445, 455, 465, 475, 485, 495, 505, 515, 525, 535,
545, 555, 565, 575, 585, 595, 605, 615, 625, 635, 645, 655, 665, 675, 685, 695, 705, 715, 725,
735, 745, and 755, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid;
(iv) a CDRL1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:46, 36, 126, 26, 136, 146, 156, 166, 246, 176, 6,
16, 186, 56, 196, 216, 106, 226, 116, 236, 86, 96, 66, 76, 206, 285, 295, 305, 315, 325, 335, 345,
355, 365, 375, 385, 395, 405, 415, 425, 437, 447, 457, 467, 477, 487, 497, 507, 517, 527, 537,
547, 557, 567, 577, 587, 597, 607, 617, 627, 637, 647, 657, 667, 677, 687, 697, 707, 717, 727,
737, 747, and 757, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid;
(v) a CDRL2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:47, 37, 127, 27, 137, 147, 157, 167, 247, 177, 7,
17, 187, 57, 197, 217, 107, 227, 117, 237, 87, 97, 67, 77, 207, 286, 296, 306, 316, 326, 336, 346,
356, 366, 376, 386, 396, 406, 416, 426, 438, 448, 458, 468, 478, 488, 498, 508, 518, 528, 538,
548, 558, 568, 578, 588, 598, 608, 618, 628, 638, 648, 658, 668, 678, 688, 698, 708, 718, 728,
738, 748, and 758, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid; and/or
(vi) a CDRL3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs. :48, 38, 128, 28, 138, 148, 158, 168, 248, 178, 8,
18, 188, 58, 198, 218, 108, 228, 118, 238, 88, 98, 68, 78, 208, 287, 297, 307, 317, 327, 337, 347,
357, 367, 377, 387, 397, 407, 417, 427, 439, 449, 459, 469, 479, 489, 499, 509, 519, 529, 539, 549, 559, 569, 579, 589, 599, 609, 619, 629, 639, 649, 659, 669, 679, 689, 699, 709, 719, 729, 739, 749, and 759, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid.
In certain embodiments, the antibody or antigen-binding fragment comprises a CDRH3 and a CDRL3, wherein the CDRH3 and the CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (i) 44 and 48, respectively; (ii) 34 and 38, respectively; (iii) 124 and 128, respectively; (iv) 24 and 28, respectively; (v) 134 and 138, respectively; (vi) 144 and 148, respectively; (vii) 154 and 158, respectively; (viii) 164 and 168, respectively; (ix) 244 and 248, respectively; (x) 174 and 178, respectively; (xi) 4 and 8, respectively; (xii) 14 and 18, respectively; (xiii) 184 and 188, respectively; (xiv) 54 and 58, respectively; (xv) 194 and 198, respectively; (xvi) 214 and 218, respectively; (xvii) 104 and 108, respectively; (xviii) 224 and 228, respectively; (xix) 114 and 118, respectively; (xx) 234 and 238, respectively; (xxi) 84 and 88, respectively; (xxii) 94 and 98, respectively; (xxiii) 64 and 68, respectively; (xxiv) 74 and 78, respectively; (xxv) 204 and 208, respectively; (xxvi) 283 and 287, respectively; (xxvii) 293 and 297, respectively; (xxviii) 303 and 307, respectively; (xxix) 313 and 317, respectively; (xxx) 323 and 327, respectively; (xxxi) 333 and 337, respectively; (xxxii) 343 and 347, respectively; (xxxiii) 353 and 357, respectively; (xxxiv) 363 and 367, respectively; (xxxv) 373 and 377, respectively, (xxxvi) 383 and 387, respectively; (xxxvii) 393 and 397, respectively; (xxxviii) 403 and 407, respectively; (xxxix) 413 and 417, respectively; (xl) 423 and 427, respectively; (xli) 435 and 439, respectively; (xlii) 445 and 449, respectively; (xliii) 455 and 459, respectively; (xliv) 465 and 469, respectively; (xlv) 475 and 479, respectively; (xlvi) 485 and 489, respectively; (xlvii) 495 and 499, respectively; (xlviii) 505 and 509, respectively; (xlix) 515 and 519, respectively; (1) 525 and 529, respectively; (li) 535 and 539, respectively; (lii) 545 and 549, respectively; (liii) 555 and 559, respectively; (liv) 565 and 569, respectively; (Iv) 575 and 579, respectively; (Ivi) 585 and 589, respectively; (Ivii) 595 and 599, respectively; (Iviii) 605 and 609, respectively; (lix) 615 and 619, respectively; (lx) 625 and 629, respectively; (Ixi) 635 and 639, respectively; (Ixii) 645 and 649, respectively; (Ixiii) 655 and 659, respectively; (Ixiv) 665 and 669, respectively; (Ixv) 675 and 679, respectively; (Ixvi) 685 and 689, respectively; (Ixvii) 695 and 699, respectively; (Ixviii) 705 and 709, respectively; (Ixix) 715 and 719, respectively; (Ixx) 725 and 729, respectively; (Ixxi) 735 and 739, respectively; (Ixxii) 745 and 749, respectively; or (Ixxiii) 755 and 759, respectively.
In certain embodiments, the antibody or antigen-binding fragment comprises a CDRH1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (i) 42-44 and 46-48, respectively; (ii) 32-34 and 36- 38, respectively; (iii) 122-124 and 126-128, respectively; (iv) 22-24 and 26-28, respectively; (v) 132-134 and 136-138, respectively; (vi) 142-144 and 146-148, respectively; (vii) 152-154 and 156-158, respectively; (viii) 162-164 and 166-168, respectively; (ix) 242-244 and 246-248, respectively; (x) 172-174 and 176-178, respectively; (xi) 2-4 and 6-8, respectively; (xii) 12-14 and 16-18, respectively; (xiii) 182-184 and 186-188, respectively; (xiv) 52-54 and 56-58, respectively; (xv) 192-194 and 196-198, respectively; (xvi) 212-214 and 216-218, respectively; (xvii) 102-104 and 106-108, respectively; (xviii) 222-224 and 226-228, respectively; (xix) 112- 114 and 116-118, respectively; (xx) 232-234 and 236-238, respectively; (xxi) 82-84 and 86-88, respectively; (xxii) 92-94 and 96-98, respectively; (xxiii) 62-64 and 66-68; (xxiv) 72-74 and 76- 78, respectively; (xxv) 202-204 and 206-208, respectively; (xxvi) 281-283 and 285-287, respectively; (xxvii) 291-293 and 295-297, respectively; (xxviii) 301-303 and 305-307, respectively; (xxix) 311-313 and 315-317, respectively; (xxx) 321-323 and 325-327, respectively; (xxxi) 331-333 and 335-337, respectively; (xxxii) 341-343 and 345-347, respectively; (xxxiii) 351-353 and 355-357, respectively; (xxxiv) 361-363 and 365-367, respectively; (xxxv) 371-373 and 375-377, respectively; (xxxvi) 381-383 and 385-387, respectively; (xxxvii) 391-393 and 395-397, respectively; (xxxviii) 401-403 and 405-407, respectively; (xxxix) 411-413 and 415-417, respectively; (xl) 421-423 and 425-427, respectively; (xli) 433-435 and 437-439, respectively; (xlii) 443-445 and 447-449, respectively; (xliii) 453- 455 and 457-459, respectively; (xliv) 463-465 and 467-469, respectively; (xlv) 473-475 and 477- 479, respectively; (xlvi) 483-485 and 487-489, respectively; (xlvii) 493-495 and 497-499, respectively; (xlviii) 503-505 and 507-509, respectively; (xlix) 513-515 and 517-519, respectively; (1) 523-525 and 527-529, respectively; (li) 533-535 and 537-539, respectively; (lii) 543-545 and 547-549, respectively; (liii) 553-555 and 557-559, respectively; (liv) 563-565 and 567-569, respectively; (Iv) 573-575 and 577-579, respectively; (Ivi) 583-585 and 587-589, respectively; (Ivii) 593-595 and 597-599, respectively; (Iviii) 603-605 and 607-609, respectively; (lix) 613-615 and 617-619, respectively; (lx) 623-625 and 627-629, respectively; (Ixi) 633-635 and 637-639, respectively; (Ixii) 643-645 and 647-649, respectively; (Ixiii) 653- 655 and 657-659, respectively; (Ixiv) 663-665 and 667-669, respectively; (Ixv) 673-675 and 677- 679, respectively; (Ixvi) 683-685 and 687-689, respectively; (Ixvii) 693-695 and 697-699,
respectively; (Ixviii) 703-705 and 707-709, respectively; (Ixix) 713-715 and 717-719, respectively; (Ixx) 723-725 and 727-729, respectively; (Ixxi) 733-735 and 737-739, respectively; (Ixxii) 743-745 and 747-749, respectively; or (Ixxiii) 753-755 and 757-759, respectively.
In certain embodiments, an antibody or antigen-binding fragment comprises an amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1- 6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3- 64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3- 11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1-16; IGKV3-20; IGLV3- 21; IGHV1-69; IGHV3-43, or any combination thereof. In some embodiments, an antibody or antigen-binding fragment comprises amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5- 51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3- 21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4,
IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3- 49, IGHJ4, IGLV1-40, and IGLJ1 ; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6- 57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3- 49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
In certain embodiments, an antibody or antigen-binding fragment comprises a framework amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting of, an amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1- 27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1- 39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1-16; IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof. In some embodiments, an antibody or antigen-binding fragment comprises framework amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the framework amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6,
and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1- 16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3- 30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3- 21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2. Framework amino acid sequences can be identified by a same numbering scheme that is used to identify CDRs; e.g., IMGT, Kabat, Chothia, Enhanced Chothia, AbM, AHo, North, Martin, Contact, or any combination thereof.
In some embodiments, an antibody or antigen-binding fragment comprises a VH framework region (VHFR)1, a VHFR2, a VHFR3, a VL framework region (VLFR)1, a VLFR2, a VLFR3, and/or a VLFR4 (or a variant of the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4 comprising one, two, three, four, or five substitutions, insertions, and/or deletions, or a variant having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4, respectively) of the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125,
respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively. In some embodiments, the framework region or regions are according to the IMGT numbering scheme. In some embodiments, the framework region or regions are according to the Kabat numbering scheme. In some embodiments, the framework region or regions are according to the Chothia numbering scheme. In some embodiments, the framework region or regions are according to the Enhanced Chothia numbering scheme. In some embodiments, the framework region or regions are according to the AbM numbering scheme. In some embodiments, the framework region or regions are according to the EU numbering scheme. In some embodiments, the framework region or regions are according to the North numbering scheme. In some embodiments, the framework region or regions are according
to the Contact numbering scheme. In some embodiments, the framework region or regions are according to the AHo numbering scheme. In some embodiments, the framework region or regions are according to a combination of any two or more of the following numbering schemes: IMGT, Kabat Chothia, Enhanced Chothia, AbM, AHo, EU, North, Contact.
In some embodiments, the antibody or antigen-binding fragment comprises a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716,
respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively.
During antibody development, DNA in the germline variable (V), joining (J), and diversity (D) gene loci may be rearranged and insertions and/or deletions of nucleotides in the coding sequence may occur. Somatic mutations may be encoded by the resultant sequence, and can be identified by reference to a corresponding known germline sequence. In some contexts, somatic mutations that are not critical to a desired property of the antibody (e.g., binding to a herein-disclosed antigen), or that confer an undesirable property upon the antibody (e.g., an increased risk of immunogenicity in a subject administered the antibody), or both, may be replaced by the corresponding germline-encoded amino acid, or by a different amino acid, so that a desirable property of the antibody is improved or maintained and the undesirable property of the antibody is reduced or abrogated. Thus, in some embodiments, the antibody or antigenbinding fragment of the present disclosure comprises at least one more germline-encoded amino acid in a variable region as compared to a parent antibody or antigen-binding fragment, provided that the parent antibody or antigen binding fragment comprises one or more somatic mutations. Variable region and CDR amino acid sequences (SEQ ID NOs.) of certain antibodies of the present disclosure are provided in Tables 2-4 herein.
The term “CL” refers to an “immunoglobulin light chain constant region” or a “light chain constant region,” z.e., a constant region from an antibody light chain. The term “CH” refers to an “immunoglobulin heavy chain constant region” or a “heavy chain constant region,” which is further divisible, depending on the antibody isotype, into CHI, CH2, and CH3 (IgA, IgD, IgG), or CHI, CH2, CH3, and CH4 domains (IgE, IgM). The Fc region of an antibody heavy chain is described further herein. In any of the presently disclosed embodiments, an antibody or antigen-binding fragment of the present disclosure comprises any one or more of CL, a CHI, a CH2, and a CH3. In any of the presently disclosed embodiments, an antibody or antigen-binding fragment of the present disclosure may comprise any one or more of CL, a CHI, a CH2, and a CH3. In certain embodiments, a CL comprises an amino acid sequence having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity (or similarity) to the amino acid sequence of a human IgG kappa constant domain (e.g., to SEQ ID NO.:278) or to a human IgG lambda constant domain (e.g., to SEQ ID NO.:279). In certain embodiments, a CH1-CH2-CH3 comprises an amino acid sequence having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100%
identity (or similarity) to the amino acid sequence of a human IgGl isotype, an engineered human IgGl isotype, a human IgG3 isotype, or an engineered human IgG3 isotype. In certain embodiments, a CH1-CH2-CH3 comprises an amino acid sequence having at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity (or similarity) to the amino acid sequence of any one of SEQ ID NOs.:251-277 and 765. It will be understood that, for example, production in a mammalian cell line can remove one or more C-terminal lysine of an antibody heavy chain (see, e.g., Liu et al. mAbs 6(5): 1145-1154 (2014)). Accordingly, an antibody or antigen-binding fragment of the present disclosure can comprise a heavy chain, a CH1-CH3, a CH3, or an Fc polypeptide wherein a C-terminal lysine residue is present or is absent; in other words, encompassed are embodiments where the C-terminal residue of a heavy chain, a CH1-CH3, or an Fc polypeptide is not a lysine, and embodiments where a lysine is the C-terminal residue. In certain embodiments, a composition comprises a plurality of an antibody and/or an antigenbinding fragment of the present disclosure, wherein one or more antibody or antigen-binding fragment does not comprise a lysine residue at the C-terminal end of the heavy chain, CH1-CH3, or Fc polypeptide, and wherein one or more antibody or antigen-binding fragment comprises a lysine residue at the C-terminal end of the heavy chain, CH1-CH3, or Fc polypeptide.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure can comprise a heavy chain, a CH1-CH3, a CH3, or an Fc polypeptide wherein a C-terminal glycine-lysine sequence e.g., the last two amino acids of SEQ ID NO.:251) is present or is absent.
A “Fab” (fragment antigen binding) is the part of an antibody that binds to antigens and includes the variable region and CHI of the heavy chain linked to the light chain via an interchain disulfide bond. Each Fab fragment is monovalent with respect to antigen binding, i.e., it has a single antigen-binding site (or domain). Pepsin treatment of an antibody yields a single large F(ab’)2 fragment that roughly corresponds to two disulfide linked Fab fragments having divalent antigen-binding activity and is still capable of cross-linking antigen. Both the Fab and F(ab’)2 are examples of “antigen-binding fragments.” Fab’ fragments differ from Fab fragments by having additional few residues at the carboxy terminus of the CHI domain including one or more cysteines from the antibody hinge region. Fab’-SH is the designation herein for Fab’ in which the cysteine residue(s) of the constant domains bear a free thiol group. F(ab’)2 antibody fragments originally were produced as pairs of Fab’ fragments that have hinge cysteines between them. Other chemical couplings of antibody fragments are also known.
Fab fragments may be joined, e.g., by a peptide linker, to form a single chain Fab, also referred to herein as “scFab.” In these embodiments, an inter-chain disulfide bond that is present in a native Fab may not be present, and the linker serves in full or in part to link or connect the Fab fragments in a single polypeptide chain. A heavy chain-derived Fab fragment (e.g., comprising, consisting of, or consisting essentially of VH + CHI, or “Fd”) and a light chain- derived Fab fragment (e.g., comprising, consisting of, or consisting essentially of VL + CL) may be linked in any arrangement to form a scFab. For example, a scFab may be arranged, in N- terminal to C-terminal direction, according to (heavy chain Fab fragment - linker - light chain Fab fragment) or (light chain Fab fragment - linker - heavy chain Fab fragment). Peptide linkers and exemplary linker sequences for use in scFabs are discussed in further detail herein.
“Fv” is a small antibody fragment that contains a complete antigen-recognition and antigen-binding site (or domain). This fragment generally consists of a dimer of one heavy- and one light-chain variable region domain in tight, non-covalent association. However, even a single variable domain (or half of an Fv comprising only three CDRs specific for an antigen) has the ability to recognize and bind antigen, although typically at a lower affinity than the entire binding site (or domain).
“Single-chain Fv” also abbreviated as “sFv” or “scFv”, are antibody fragments that comprise the VH and VL antibody domains connected into a single polypeptide chain. In some embodiments, the scFv polypeptide comprises a polypeptide linker disposed between and linking the VH and VL domains that enables the scFv to retain or form the desired structure for antigen binding. Such a peptide linker can be incorporated into a fusion polypeptide using standard techniques well known in the art. For a review of scFv, see Pluckthun in The Pharmacology of Monoclonal Antibodies, vol. 113, Rosenburg and Moore eds., Springer-Verlag, New York, pp. 269-315 (1994); Borrebaeck 1995, infra. In certain embodiments, the antibody or antigenbinding fragment comprises a scFv comprising a VH domain, a VL domain, and a peptide linker linking the VH domain to the VL domain. In particular embodiments, a scFv comprises a VH domain linked to a VL domain by a peptide linker, which can be in a VH-linker-VL orientation or in a VL-linker-VH orientation. Any scFv of the present disclosure may be engineered so that the C-terminal end of the VL domain is linked by a short peptide sequence to the N-terminal end of the VH domain, or vice versa (i.e., (N)VL(C)-linker-(N)VH(C) or (N)VH(C)-linker- (N)VL(C). Alternatively, in some embodiments, a linker may be linked to an N-terminal portion or end of the VH domain, the VL domain, or both.
Peptide linker sequences may be chosen, for example, based on: (1) their ability to adopt a flexible extended conformation; (2) their inability or lack of ability to adopt a secondary structure that could interact with functional epitopes on the first and second polypeptides and/or on a target molecule; and/or (3) the lack or relative lack of hydrophobic or charged residues that might react with the polypeptides and/or target molecule. Other considerations regarding linker design (e.g., length) can include the conformation or range of conformations in which the VH and VL can form a functional antigen-binding site (or domain). In certain embodiments, peptide linker sequences contain, for example, Gly, Asn and Ser residues. Other near neutral amino acids, such as Thr and Ala, may also be included in a linker sequence. Other amino acid sequences which may be usefully employed as linker include those disclosed in Maratea et al., Gene 40:39 46 (1985); Murphy et al., Proc. Natl. Acad. Sci. USA 83:8258 8262 (1986); U.S. Pat. No. 4,935,233, and U.S. Pat. No. 4,751,180. Other illustrative and non-limiting examples of linkers may include, for example, Glu-Gly-Lys-Ser-Ser-Gly-Ser-Gly-Ser-Glu-Ser-Lys-Val-Asp (Chaudhary et al., Proc. Natl. Acad. Sci. USA 87: 1066-1070 (1990)) and Lys-Glu-Ser-Gly-Ser- Val-Ser-Ser-Glu-Gln-Leu-Ala-Gln-Phe-Arg-Ser-Leu-Asp (Bird et al., Science 242:423-426 (1988)) and the pentamer Gly-Gly-Gly-Gly-Ser when present in a single iteration or repeated 1 to 5 or more times, or more. Any suitable linker may be used, and in general can be about 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 15 23, 24, 25, 26, 27, 28, 29, 30, 40, 50, 60, 70, 80, 90, 100 amino acids in length, or less than about 200 amino acids in length, and will preferably comprise a flexible structure (can provide flexibility and room for conformational movement between two regions, domains, motifs, fragments, or modules connected by the linker), and will preferably be biologically inert and/or have a low risk of immunogenicity in a human. ScFvs can be constructed using any combination of the VH and VL sequences or any combination of the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 sequences disclosed herein. In some embodiments, linker sequences are not required; for example, when the first and second polypeptides have non-essential N-terminal amino acid regions that can be used to separate the functional domains and prevent steric interference.
In certain embodiments, an antibody or antigen-binding fragment of the present disclosure is monospecific (e.g., binds to a single epitope) or is multispecific (e.g., binds to multiple epitopes and/or target molecules). Antibodies and antigen binding fragments may be constructed in various formats. Exemplary antibody formats disclosed in Spiess et al., Mol. Immunol. 67(2):95 (2015), and in Brinkmann and Kontermann, mAbs 9(2): 182-212 (2017), which formats and methods of making the same are incorporated herein by reference and
include, for example, Bispecific T cell Engagers (BiTEs), DARTs, Knobs-Into-Holes (KIH) assemblies, scFv-CH3-KIH assemblies, KIH Common Light-Chain antibodies, TandAbs, Triple Bodies, TriBi Minibodies, Fab-scFv, scFv-CH-CL-scFv, F(ab’)2-scFv2, tetravalent Hcabs, Intrabodies, CrossMabs, Dual Action Fabs (DAFs) (two-in-one or four-in-one), DutaMabs, DT- IgG, Charge Pairs, Fab-arm Exchange, SEEDbodies, Triomabs, LUZ-Y assemblies, Fcabs, KX- bodies, orthogonal Fabs, DVD-Igs (e.g., US Patent No. 8,258,268, which formats are incorporated herein by reference in their entirety), IgG(H)-scFv, scFv-(H)IgG, IgG(L)-scFv, scFv-(L)IgG, IgG(L,H)-Fv, IgG(H)-V, V(H)-IgG, IgG(L)-V, V(L)-IgG, KIH IgG-scFab, 2scFv- IgG, IgG-2scFv, scFv4-Ig, Zybody, and DVLIgG (four-in-one), as well as so-called FIT-Ig (e.g., PCT Publication No. WO 2015/103072, which formats are incorporated herein by reference in their entirety), so-called WuxiBody formats (e.g., PCT Publication No. WO 2019/057122, which formats are incorporated herein by reference in their entirety), and so-called In-Elbow-Insert Ig formats (lELIg; e.g., PCT Publication Nos. WO 2019/024979 and WO 2019/025391, which formats are incorporated herein by reference in their entirety).
In certain embodiments, the antibody or antigen-binding fragment comprises two or more of VH domains, two or more VL domains, or both (i.e., two or more VH domains and two or more VL domains). In particular embodiments, an antigen-binding fragment comprises the format (N-terminal to C-terminal direction) VH-linker-VL-linker-VH-linker-VL, wherein the two VH sequences can be the same or different and the two VL sequences can be the same or different. Such linked scFvs can include any combination of VH and VL domains arranged to bind to a given target, and in formats comprising two or more VH and/or two or more VL, one, two, or more different epitopes or antigens may be bound. It will be appreciated that formats incorporating multiple antigen-binding domains may include VH and/or VL sequences in any combination or orientation. For example, the antigen-binding fragment can comprise the format VL-linker-VH-linker-VL-linker-VH, VH-linker-VL-linker-VL-linker-VH, or VL-linker-VH- linker-VH-linker-VL.
Monospecific or multispecific antibodies or antigen-binding fragments of the present disclosure can comprise any combination of the VH and VL sequences and/or any combination of the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 sequences disclosed herein. A bispecific or multispecific antibody or antigen-binding fragment may, in some embodiments, comprise one, two, or more antigen-binding domains (e.g., a VH and a VL) of the instant disclosure. Two or more binding domains may be present that bind to the same or a different antigen epitope, and a bispecific or multispecific antibody or antigen-binding fragment as
provided herein can, in some embodiments, comprise a further antigen-specific binding domain, and/or can comprise a binding domain that binds to a different antigen or pathogen altogether.
In any of the presently disclosed embodiments, the antibody or antigen-binding fragment can be multispecific; e.g., bispecific, trispecific, or the like.
In some embodiments, an antibody, or an antigen-binding fragment thereof is provided which is a multi-specific antibody or antigen-binding fragment thereof, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and a ClfA; a SpA and a ClfA; an LTA and a Sbi; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; a Sbi and a ClfA; or a Sbi and a Gmd. In some embodiments, each antigen-binding domain of a multispecific antibody or antigen-binding fragment is capable of targeting a different binding site of the same target selected from: an LTA, an alpha toxin, a gamma toxin, a SpA, a ClfA, a Gmd, a Sbi, and a leucocidin. In some embodiments, a multispecific antibody or antigen-binding fragment comprises at least two antigen-binding sites, wherein at least one antigen-binding site is capable of targeting a first binding site of a target selected from: an LTA, an alpha toxin, a gamma toxin, a SpA, a ClfA, a Gmd, a Sbi, and a leucocidin; and wherein at least one antigen-binding site is capable of targeting a second binding site of the target.
In some embodiments, a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to an opsonizing target and a second antigen-binding domain of the multispecific antibody or antigen-binding fragment binds to a neutralizing target. In some embodiments, a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to a target selected from LTA, ClfA, Protein A, and Gmd, and a second antigenbinding domain of the multispecific antibody or antigen-binding fragment binds to an alpha toxin. In some embodiments, a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to an opsonizing target and a second antigen-binding domain of the multispecific antibody or antigen-binding fragment binds to an alpha toxin.
In some embodiments, a first antigen-binding domain of a multispecific antibody or antigen-binding fragment binds to a target selected from LTA, ClfA, Protein A, and Gmd and a
second antigen-binding domain of the multispecific antibody or antigen-binding fragment binds to a neutralizing target.
Also provided are fusion proteins that comprise an antibody or antigen-binding fragment of the present disclosure. In some embodiments, a fusion protein comprises (i) an extracellular component comprising the antibody or antigen-binding fragment, (ii) a transmembrane component, and (iii) an intracellular component comprising one or more signaling domains (e.g., from CD3(^, CD28, 4-1BB, and/or TLR8). In some embodiments, a fusion protein comprises a chimeric antigen receptor. In some embodiments, a fusion protein comprises a chimeric engulfment receptor (see, e.g., Corey el al. , Molecular Therapy Methods & Clinical Development 28: 1-10 (2023); doi . org/ 10.1016/j . omtim .2022.11.004).
In certain embodiments, the antibody or antigen-binding fragment comprises a Fc polypeptide, or a fragment thereof. The “Fc” fragment or Fc polypeptide comprises the carboxyterminal portions i.e., the CH2 and CH3 domains of IgG) of both antibody H chains held together by disulfides. An Fc may comprise a dimer comprised of two Fc polypeptides (i.e., two CH2-CH3 polypeptides). Antibody “effector functions” refer to those biological activities attributable to the Fc region (a native sequence Fc region or amino acid sequence variant Fc region) of an antibody, and vary with the antibody isotype. Examples of antibody effector functions include: Clq binding and complement dependent cytotoxicity; Fc receptor binding; antibody-dependent cell-mediated cytotoxicity (ADCC); phagocytosis; down regulation of cell surface receptors (e.g., B cell receptor); and B cell activation. As discussed herein, modifications (e.g., amino acid substitutions) may be made to an Fc domain in order to modify (e.g., improve, reduce, or ablate) one or more functionality of an Fc-containing polypeptide (e.g., an antibody of the present disclosure). Such functions include, for example, Fc receptor (FcR) binding, antibody half-life modulation (e.g., by binding to FcRn), ADCC function, protein A binding, protein G binding, and complement binding. Amino acid modifications that modify (e.g., improve, reduce, or ablate) Fc functionalities include, for example, the T250Q/M428L, M252Y/S254T/T256E, H433K/N434F, M428L/N434S, M428L/434A, E233P/L234V/L235A/G236 + A327G/A330S/P331S, E333A, S239D/A330L/I332E, P257VQ311, K326W/E333S, S239D/I332E/G236A, N297Q, K322A, S228P, L235E + E318A/K320A/K322A, L234A/L235A (also referred to herein as “LALA”), and L234A/L235A/P329G mutations, and other mutations described herein, certain of which mutations are summarized and annotated in “Engineered Fc Regions”, published by InvivoGen (2011) and available online at invivogen.com/PDF/review/review-Engineered-Fc-Regions-
invivogen.pdf?utm_source=review&utm_medium=pdf&utm_ campaign=review&utm_content=Engineered-Fc-Regions, and are incorporated herein by reference.
For example, to activate the complement cascade, the Clq protein complex can bind to at least two molecules of IgGl or one molecule of IgM when the immunoglobulin molecule(s) is attached to the antigenic target (Ward, E. S., and Ghetie, V., Ther. Immunol. 2 (1995) 77-94). Burton, D. R., described (Mol. Immunol. 22 (1985) 161-206) that the heavy chain region comprising amino acid residues 318 to 337 is involved in complement fixation. Duncan, A. R., and Winter, G. (Nature 332 (1988) 738-740), using site directed mutagenesis, reported that Glu318, Lys320 and Lys322 form the binding site to Clq. The role of Glu318, Lys320 and Lys 322 residues in the binding of Clq was confirmed by the ability of a short synthetic peptide containing these residues to inhibit complement mediated lysis.
For example, FcR binding can be mediated by the interaction of the Fc moiety (of an antibody) with Fc receptors (FcRs), which are specialized cell surface receptors on cells including hematopoietic cells. Fc receptors belong to the immunoglobulin superfamily, and shown to mediate both the removal of antibody-coated pathogens by phagocytosis of immune complexes, and the lysis of erythrocytes and various other cellular targets (e.g. tumor cells) coated with the corresponding antibody, via antibody dependent cell mediated cytotoxicity (ADCC; Van de Winkel, J. G., and Anderson, C. L., J. Leukoc. Biol. 49 (1991) 511-524). FcRs are defined by their specificity for immunoglobulin classes; Fc receptors for IgG antibodies are referred to as FcyR, for IgE as FcsR, for IgA as FcaR and so on and neonatal Fc receptors are referred to as FcRn. Fc receptor binding is described for example in Ravetch, J. V., and Kinet, J. P., Annu. Rev. Immunol. 9 (1991) 457-492; Capel, P. J., et al., Immunomethods 4 (1994) 25-34; de Haas, M., et al., J Lab. Clin. Med. 126 (1995) 330-341; and Gessner, J. E., et al., Ann. Hematol. 76 (1998) 231-248.
Cross-linking of receptors by the Fc domain of native IgG antibodies (FcyR) triggers a wide variety of effector functions including phagocytosis, antibody-dependent cellular cytotoxicity, and release of inflammatory mediators, as well as immune complex clearance and regulation of antibody production. Fc moi eties providing cross-linking of receptors (e.g., FcyR) are contemplated herein. In humans, three classes of FcyR have been characterized to-date, which are: (i) FcyRI (CD64), which binds monomeric IgG with high affinity and is expressed on macrophages, monocytes, neutrophils and eosinophils; (ii) FcyRII (CD32), which binds complexed IgG with medium to low affinity, is widely expressed, in particular on leukocytes, is
believed to be a central player in antibody-mediated immunity, and which can be divided into FcyRIIA, FcyRIIB and FcyRIIC, which perform different functions in the immune system, but bind with similar low affinity to the IgG-Fc, and the ectodomains of these receptors are highly homologuous; and (iii) FcyRIII (CD 16), which binds IgG with medium to low affinity and has been found in two forms: FcyRIIIA, which has been found on NK cells, macrophages, eosinophils, and some monocytes and T cells, and is believed to mediate ADCC; and FcyRIIIB, which is highly expressed on neutrophils.
FcyRIIA is found on many cells involved in killing (e.g. macrophages, monocytes, neutrophils) and seems able to activate the killing process. FcyRIIB seems to play a role in inhibitory processes and is found on B-cells, macrophages and on mast cells and eosinophils. Importantly, it has been shown that 75% of all FcyRIIB is found in the liver (Ganesan, L. P. et al., 2012: “FcyRIIb on liver sinusoidal endothelium clears small immune complexes,” loumal of Immunology 189: 4981-4988). FcyRIIB is abundantly expressed on Liver Sinusoidal Endothelium, called LSEC, and in Kupffer cells in the liver and LSEC are the major site of small immune complexes clearance (Ganesan, L. P. et al., 2012: FcyRIIb on liver sinusoidal endothelium clears small immune complexes. lournal of Immunology 189: 4981-4988).
In some embodiments, the antibodies disclosed herein and the antigen-binding fragments thereof comprise an Fc polypeptide or fragment thereof for binding to FcyRIIb, in particular an Fc region, such as, for example IgG-type antibodies. Moreover, it is possible to engineer the Fc moiety to enhance FcyRIIB binding by introducing the mutations S267E and L328F as described by Chu, S. Y. et al., 2008: Inhibition of B cell receptor-mediated activation of primary human B cells by co-engagement of CD19 and FcgammaRIIb with Fc-engineered antibodies. Molecular Immunology 45, 3926-3933. Thereby, the clearance of immune complexes can be enhanced (Chu, S., et al., 2014: Accelerated Clearance of IgE In Chimpanzees Is Mediated By Xmab7195, An Fc-Engineered Antibody With Enhanced Affinity For Inhibitory Receptor FcyRIIb. Am I Respir Crit, American Thoracic Society International Conference Abstracts). In some embodiments, the antibodies of the present disclosure, or the antigen binding fragments thereof, comprise an engineered Fc moiety with the mutations S267E and L328F, in particular as described by Chu, S. Y. et al., 2008: Inhibition of b cell receptor-mediated activation of primary human B cells by co-engagement of CD19 and FcgammaRIIb with Fc-engineered antibodies. Molecular Immunology 45, 3926-3933.
On B cells, FcyRIIB may function to suppress further immunoglobulin production and isotype switching to, for example, the IgE class. On macrophages, FcyRIIB is thought to inhibit
phagocytosis as mediated through FcyRIIA. On eosinophils and mast cells, the B form may help to suppress activation of these cells through IgE binding to its separate receptor.
Regarding FcyRI binding, modification in native IgG of at least one of E233-G236, P238, D265, N297, A327 and P329 reduces binding to FcyRI. IgG2 residues at positions 233-236, substituted into corresponding positions IgGl and IgG4, reduces binding of IgGl and IgG4 to FcyRI by 103-fold and eliminated the human monocyte response to antibody-sensitized red blood cells (Armour, K. L., et al. Eur. J. Immunol. 29 (1999) 2613-2624).
Regarding FcyRII binding, reduced binding for FcyRIIA is found, e.g., for IgG mutation of at least one of E233-G236, P238, D265, N297, A327, P329, D270, Q295, A327, R292 and K414.
Two allelic forms of human FcyRIIA are the “H131” variant, which binds to IgGl Fc with higher affinity, and the “R131” variant, which binds to IgGl Fc with low affinity. See, e.g., Bruhns et al, Blood 11331X6-3125 (2009).
Regarding FcyRIII binding, reduced binding to FcyRIIIA is found, e.g., for mutation of at least one of E233-G236, P238, D265, N297, A327, P329, D270, Q295, A327, S239, E269, E293, Y296, V303, A327, K338 and D376. Mapping of the binding sites on human IgGl for Fc receptors, the above-mentioned mutation sites, and methods for measuring binding to FcyRI and FcyRIIA, are described in Shields, R. L., et al., J. Biol. Chem. 276 (2001) 6591-6604.
Two allelic forms of human FcyRIIIA are the “Fl 58” variant, which binds to IgGl Fc with lower affinity, and the “V158” variant, which binds to IgGl Fc with higher affinity. See, e.g., Bruhns et al., Blood 11331X6-3125 (2009).
Regarding binding to FcyRII, two regions of native IgG Fc appear to be involved in interactions between FcyRIIs and IgGs, namely (i) the lower hinge site of IgG Fc, in particular amino acid residues L, L, G, G (234 - 237, EU numbering), and (ii) the adjacent region of the CH2 domain of IgG Fc, in particular a loop and strands in the upper CH2 domain adjacent to the lower hinge region, e.g. in a region of P331 (Wines, B.D., et al., I. Immunol. 2000; 164: 5313 - 5318). Moreover, FcyRI appears to bind to the same site on IgG Fc, whereas FcRn and Protein A bind to a different site on IgG Fc, which appears to be at the CH2-CH3 interface (Wines, B.D., et al., I. Immunol. 2000; 164: 5313 - 5318).
Also contemplated are mutations that increase binding affinity of an Fc polypeptide or fragment thereof of the present disclosure to a (i.e., one or more) Fey receptor (e.g., as compared to a reference Fc polypeptide or fragment thereof or containing the same that does not comprise the mutation(s)). See, e.g., Delillo and Ravetch, Cell 161(5): 1035-1045 (2015) and Ahmed et al.,
J. Struc. Biol. 194(1):78 (2016), the Fc mutations and techniques of which are incorporated herein by reference.
In any of the herein disclosed embodiments, an antibody or antigen-binding fragment can comprise a Fc polypeptide or fragment thereof comprising a mutation selected from G236A; S239D; A330L; and I332E; or a combination comprising any two or more of the same; e.g., S239D/I332E; S239D/A330L/I332E; G236A/S239D/I332E; G236A/A330L/I332E (also referred to herein as “GAALIE”); or G236A/S239D/A330L/I332E. In some embodiments, the Fc polypeptide or fragment thereof does not comprise S239D. In some embodiments, the Fc polypeptide or fragment thereof comprises S at position 239 (EU numbering).
In certain embodiments, the Fc polypeptide or fragment thereof may comprise or consist of at least a portion of an Fc polypeptide or fragment thereof that is involved in FcRn binding. In certain embodiments, the Fc polypeptide or fragment thereof comprises one or more amino acid modifications that improve binding affinity for (e.g., enhance binding to) FcRn (e.g., at a pH of about 6.0) and, in some embodiments, thereby extend in vivo half-life of a molecule comprising the Fc polypeptide or fragment thereof (e.g., as compared to a reference Fc polypeptide or fragment thereof or antibody that is otherwise the same but does not comprise the modification(s)). In certain embodiments, the Fc polypeptide or fragment thereof comprises or is derived from a IgG Fc and a half-life-extending mutation comprises any one or more of: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q311I; D376V; T307A; E380A (EU numbering). In certain embodiments, a half-life-extending mutation comprises M428L/N434S (also referred to herein as “MLNS”, “LS”, “_LS”, and “-LS”). In certain embodiments, a half-life-extending mutation comprises M252Y/S254T/T256E. In certain embodiments, a half-life-extending mutation comprises T250Q/M428L. In certain embodiments, a half-life-extending mutation comprises P257EQ311I. In certain embodiments, a half-life-extending mutation comprises P257I/N434H. In certain embodiments, a half-life- extending mutation comprises D376V/N434H. In certain embodiments, a half-life-extending mutation comprises T307A/E380A/N434A. In certain embodiments, a half-life-extending mutation comprises M428L/N434A.
In some embodiments, an antibody or antigen-binding fragment includes a Fc moiety that comprises the substitution mutations M428L/N434S. In some embodiments, an antibody or antigen-binding fragment includes a Fc moiety that comprises the substitution mutations M428L/N434A. In some embodiments, an antibody or antigen-binding fragment includes a Fc polypeptide or fragment thereof that comprises the substitution mutations G236A/A330L/I332E.
In certain embodiments, an antibody or antigen-binding fragment includes a (e.g., IgG) Fc moiety that comprises a G236A mutation, an A330L mutation, and a I332E mutation (GAALIE), and does not comprise a S239D mutation (e.g., comprises a native S at position 239). In particular embodiments, an antibody or antigen-binding fragment includes an Fc polypeptide or fragment thereof that comprises the substitution mutations: M428L/N434S and G236A/A330L/I332E, and optionally does not comprise S239D (e.g., comprises S at 239). In certain embodiments, an antibody or antigen-binding fragment includes a Fc polypeptide or fragment thereof that comprises the substitution mutations: M428L/N434S and G236 A/S239D/A330L/I332E.
Also contemplated are mutations that reduce binding of an (e.g., IgGl Fc or engineered variant thereof) to Staphylococcus Protein A. For example, in certain embodiments, an antibody or antigen-binding fragment comprises an IgGl Fc polypeptide (or an engineered variant thereof) comprising an H435R mutation, a Y436F mutation, or an H435R mutation and a Y436F mutation. See, e.g., Jendeberg et al. J Immunol Methods 207(l):25-34 (1997); doi: 10.1016/s0022-1759(96)00215-3. In some embodiments, an antibody or antigen-binding fragment includes an (e.g., IgGl) Fc polypeptide that comprises the following mutations: (i) M428L, N434S, H435R; (ii) M428L, N434S, Y436F; or (iii) (ii) M428L, N434S, H435R, and Y436F. Alternatively, in some embodiments, an antibody or antigen-binding fragment may be expressed as (e.g., recombinant) human IgG3 (IgG3 contains an arginine “R” residue at EU position 435). In some embodiments, an antibody has a native IgGl isotype and is expressed recombinantly as an IgG3 isotype.
In certain embodiments, the antibody or antigen-binding fragment comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or the antibody or antigen-binding fragment is partially or fully aglycosylated and/or is partially or fully afucosylated. Host cell lines and methods of making partially or fully aglycosylated or partially or fully afucosylated antibodies and antigen-binding fragments are known (see, e.g., PCT Publication No. WO 2016/181357; Suzuki et al. Clin. Cancer Res. 73(6): 1875-82 (2007); Huang et al. mAbs 6: 1-12 (2018)). In certain embodiments, an antibody or antigen-binding fragment comprises a heavy chain that comprises one or more mutations in the hinge, CH2, and/or CH3 (or in the Fc), wherein the antibody or antigen-binding fragment has one or more improved characteristics over, for example, the antibody or antigenbinding fragment comprising reference wild-type Fc polypeptide and/or comprising a known variant Fc polypeptide). Presently disclosed Fc variants possess, for example: increased binding
to one or more human FcyRA (e.g., a FcyRIIA and/or a FcyRIIIA; decreased/reduced binding to a human FcyRIIB; increased binding to one or more human FcyRA as compared to binding to a human FcyRIIB; increased thermostability as compared to known Fc polypeptides; increased binding to human Clq; increased human FcyRIIIA signaling in a host cell expressing the FcyRIIIA, increased human FcyRIIIA signaling in a host cell expressing the FcyRIIA, decreased human FcyRIIB signaling in a host cell expressing the FcyRIIB, a relative increase in binding to FcyRA as compared to FcyRIIB, improved qualities for production as compared to known Fc polypeptides; and combinations of such features.
In certain embodiments, antibodies comprising a variant Fc polypeptide of the present disclosure provide surprising advantages, such as any one or more of the following: increased binding affinity (e.g. as determined by surface plasmon resonance, e.g. using a Biacore instrument and/or as determined by a electrochemiluminescence assay, such as a meso scale discovery (MSD) assay) for and/or inducing increased signaling (e.g. as determined using (1) an Fc variant antibody (2) antigen-expressing target cells and (3) reporter cells expressing one or more human FcyRA, optionally driving expression of a reporter gene such as, for example, GFP or luciferase) by one or more human FcyRA, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; decreased binding affinity for and/or inducing decreased signaling of human FcyRIIB, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; a unique and optionally improved binding profile across human FcyRIIA-H, human FcyRIIA-R, human FcyRIIB, human FcyRIIIA-F, and human FcyRIIIA-V, wherein improved binding comprises an overall increase in binding to and/or activation of FcyRA signaling relative to binding to and/or activation of inhibitory FcyR signaling, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; increased binding affinity for human Clq , as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; no detrimental effect or no substantial detrimental effect on thermal stability, a reduced negative effect on thermal stability as compared to a variant Fc polypeptide or fragment thereof not comprising the mutation(s) and/or fucosylation state (e.g., a human IgGl Fc comprising the mutations G236A, A330L, and I332E (e.g. having a smaller decreasing effect, or no decreasing effect, on melting temperature as compared to the antibody comprising a human IgGl Fc comprising the mutations G236A, A330L, and I332E), or having a higher melting temperature than the antibody comprising a human IgGl Fc comprising the mutations G236A, A330L, and I332E)); increasing
specific lysis (e.g. via ADCC) by natural killer cells and/or PBMCs (e.g. expressing Fl 58/VI 58 or VI 58/VI 58 FcyRIIIA) against antigen-expressing target cells, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state (e.g. the antibody comprising a human IgGl Fc comprising the mutations G236A, A330L, and I332E); increasing ADCP by monocytes (e.g. CD14+ monocytes, optionally expressing F 158/VI 58 FcyRIIA and R131/H131 FcyRIIA or F 158/F 158 FcyRIIA and R131/H131 FcyRIIA) against antigen-expressing target cells, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state; increasing the percentage of CD83+ cells (e.g. moDCs) and/or increasing expression of CD83 by moDCs in a sample when provided in combination with the antigen, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state, when provided in combination with the antigen; increasing production of one or more cytokine (optionally selected from the group consisting of IL-ip, IFN-y, IL-6, and TNF-a) by moDCs in a sample when provided in combination with the antigen, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state, when provided in combination with the antigen; and/or increasing the ability of moDCs to stimulate antigenspecific CD4+ T cells when provided to the moDCs in combination with the antigen, as compared to the antibody comprising a reference Fc polypeptide not comprising the mutation(s) and/or fucosylation state, when provided to the moDCs in combination with the antigen, wherein, optionally, (1) the moDCs and the CD4+ T cells are from the same (optionally antigen- vaccinated) subject and/or (2) stimulation of antigen-specific CD4+ T cells is determined by an increase in CD25 expression and/or an increase in proliferation (e.g. as determined by a reduction in CFSE staining over time) and/or an increase in expression of CD69 and/or an increase in expression of NF AT and/or an increase in expression of CD44, by the antigenspecific CD4+ T cells.
In some embodiments, an engineered Fc or Fc fragment of the present disclosure (or a polypeptide comprising the same) comprises two or more substitution mutations as compared to a reference wild-type Fc or Fc fragment, and the combined effect of the two or more substitutions is different than, and is optionally greater than, would be expected based on the effects of the individual component substitution mutations and/or based on the effects of a subset of the two or more substitution mutations. In other words, in some embodiments, combination mutations comprise a non-additive or synergistic effect with reference to the individual component mutations and/or to a subset thereof.
In some embodiments, presently disclosed antibodies or antigen-binding fragments comprising an Fc variant possess characteristics such as effector functions, ability to bind human Clq, ability to induce FcyRA-mediated cell signaling, ability to bind to human FcRn, ability to promote ADCP, ability to promote ADCC, ability to promote activation of CD4+ T cells, and the like.
Presently disclosed polypeptides include those that comprise a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises one or more modifications as compared to the IgG Fc polypeptide or fragment thereof. It will be understood that, unless stated otherwise, a “reference” polypeptide or antibody (e.g., reference IgG Fc polypeptide or fragment thereof, reference antibody, reference CH2 polypeptide, reference IgG hinge-CH2, reference IgG hinge-Fc polypeptide, reference CH3 polypeptide) is preferably identical to the recited molecule (e.g., variant of an Fc polypeptide or fragment thereof; polypeptide comprising such a variant; antibody comprising a variant of an Fc polypeptide) except for the recited difference or differences.
For example, it will be understood that for a variant IgGl Fc polypeptide that comprises an alanine (A) amino acid at EU position 236, a reference Fc polypeptide includes an IgGl Fc polypeptide that is otherwise identical to the variant except that a native glycine (G) amino acid is found at EU position 236. As another example, for a variant of an Fc polypeptide fragment (e.g., containing a CH2 and a portion of a CH3), a reference Fc polypeptide fragment is preferably of an identical length to the variant and preferably differs from the variant only by the recited features (e.g., amino acid mutation or mutations present in the variant). In some embodiments, a reference Fc polypeptide, Fc polypeptide fragment, or antibody comprises a wild-type amino acid sequence (e.g., wild-type human IgGl). Excepting the recited differences present in the variant, a reference Fc polypeptide, Fc polypeptide fragment, or antibody will be of the same isotype, and, preferably, of the same allotype, as the variant. In the case of a reference antibody, the Fabs or other antigen-binding domains will preferably be identical to those present in the specified antibody comprising a variant Fc polypeptide or fragment thereof. In some embodiments, variants of IgG Fc polypeptides or fragments thereof include one or more amino acid substitution as compared to a reference (e.g. wild-type) IgG Fc polypeptide or fragment thereof. Herein, the position of an amino acid in a variant IgG Fc polypeptide or fragment may be described by referencing the “EU position”; it will be understood that “the EU position” follows the EU numbering system as set forth in Kabat. By way of illustration, it will be understood that in the example of a human IgGl CH1-CH3 amino acid sequence provided by
SEQ ID NO:251, the first amino acid (A) corresponds to EU position 118, and the last amino acid (K) corresponds to EU position 447:
ASTKGPSVFP LAPSSKSTSG GTAALGCLVK DYFPEPVTVS WNSGALTSGV HTFPAVLQSS GLYSLSSVVT VPSSSLGTQT YICNVNHKPS NTKVDKKVEP KSCDKTHTCP PCPAPELLGG PSVFLFPPKP KDTLMISRTP EVTCVVVDVS HEDPEVKFNW YVDGVEVHNA KTKPREEQYN STYRVVS VLT VLHQDWLNGK EYKCKVSNKA LPAPIEKTIS KAKGQPREPQ VYTLPPSRDE LTKNQVSLTC LVKGFYPSDI AVEWESNGQP ENNYKTTPPV LDSDGSFFLY SKLTVDKSRW QQGNVFSCSV MHEALHNHYT QKSLSLSPGK.
Accordingly, it will be understood that unless otherwise indicated, the position of a recited amino acid(s) follows EU numbering for human IgGl even if a complete antibody heavy chain, complete CH1-CH3, complete CH2-CH3, or the like is not present or is not explicitly recited. In other words, for example, if only a hinge-CH2 is described and a CH3 and/or CHI may not be present, the position of the amino acids in the hinge-CH2 is described with reference to EU numbering, unless stated otherwise. Correspondence between EU numbering, Kabat numbering, IMGT exon numbering, and IMGT unique numbering for immunoglobulin G heavy chain constant domain is known in the art and is shown, for example, in the IMGT Scientific chart (www.imgt.org/IMGTScientificChart/Numbering/Hu_IGHGnber.html; created May 17, 2001, accessed May 23, 2021, last updated January 20, 2020).
Certain embodiments of Fc variants (fucosylated, unless otherwise indicated) and nonlimiting properties of the same are summarized in Table A; see also PCT/US2022/030556.
Additional features of disclosed Fc variant-containing antibodies are described herein.
It will be understood that two or more amino acid substitutions present in a variant can be expressed in a variety of ways, for example, as G236A_Y300L, or as G236A/Y300L. Moreover, a mutation or combination mutation may be referenced using a short form including the original amino acid(s) and the amino acid(s) resulting from the substitution(s). For example, G236A may be described as “GA” or “236A”; G236A_Y300L may be described as “GAYL”; G236A_L328V_Q295E may be described as “GALVQE”; G236A_R292P_Y300L may be described as “GARPYL”, G236A_R292P_I377N may be described as “GARPIN”, or the like.
In any of the presently disclosed embodiments, a variant of an Fc polypeptide or fragment thereof can be derived from or comprise a human Fc polypeptide or fragment thereof, and/or can be derived from or comprise a human IgGl, a human IgG2, a human IgG3, or a human IgG4 isotype. In this context, the expression “derived from” means that the variant is the same as the referenced polypeptide or isotype, with the exception of the specified modification(s) (e.g., amino acid substitution(s)). By way of example, a variant Fc polypeptide which comprises a wild-type human IgGl Fc amino acid sequence with the exception of the amino acid substitution mutations G236A L328V Q295E (and, optionally, other amino acid substitutions) can be said to be “derived from” wild-type human IgGl Fc. In any of the presently disclosed embodiments, a polypeptide, CH2, Fc, Fc fragment, or antibody may comprise human Ig sequence, such as human IgGl sequence. In some embodiments, the polypeptide, CH2, Fc, Fc fragment, or antibody can comprise a native or wild-type human Ig sequence with the exception of the described mutation(s), or can comprise a human Ig (e.g. IgG) sequence that contains one or more additional mutations.
An antibody or antigen-binding fragment, may be of any allotype or combination of allotypes. “Allotype” refers to the allelic variation found among the IgG subclasses. For example, an allotype may comprise Glml (or Glm(a)), Glm2 (or Glm(x)), Glm3 (or Glm(f)),
Glml7 (or Gm(z))m), Glm27, and/or Glm28 (Glm27 and Glm28 have been described as “alloallotypes”).
The Glm3 and Glml7 allotypes are located at the same position in the CHI domain (position 214 according to EU numbering). Glm3 comprises R214 (EU), while Glml7 comprises K214 (EU). The Glml allotype is located in the CH3 domain (at positions 356 and 358 (EU)) and refers to the replacements E356D and M358L. The Glm2 allotype refers to a replacement of the alanine in position 431 (EU) by a glycine. Glm allotypes, alloallotypes, and features thereof are known in the art and described at, for example, www.imgt.org/IMGTrepertoire/Proteins/allotypes/human/IGH/IGHC/Glm_allotypes.html and Lefranc, M.-P. and Lefranc, G. Human Gm, Km and Am allotypes and their molecular characterization: a remarkable demonstration of polymorphism In: B. Tait, F. Christiansen (Eds.), Immunogenetics, chap. 34, Humana Press, Springer, New York, USA. Methods Mol. Biol. 2012; 882, 635-680. PMID: 22665258, LIGM: 406, the contents and allotypes and allotype information of which are incorporated herein by reference.
The Glml allotype may be combined, for example, with the Glm3, Glm 17, Glm27, Glm2, and/or Glm28 allotype. In some embodiments, an allotype is Glm3 with no Glml (Glm3,-1). In some embodiments, an allotype is Glml7,l allotype. In some embodiments, an allotype is Glm3,l. In some embodiments, an allotype is Glml7 with no Glml (Glml7,-1). Optionally, these allotypes may be combined (or not combined) with the Glm2, Glm27 or Glm28 allotype. For example, an allotype may be Glml7,l,2.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a Glm3 allotype or a Glm3,l allotype. In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a Glm3 allotype and comprises M428L and N434S or M428L and N434A mutations or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a Glm3,l allotype and comprises M428L and N434S or M428L and N434A mutations or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a Glml7, 1 allotype. In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a Glml7, 1 allotype and comprises M428L and N434S or M428L and N434A mutations or any other mutation(s) that enhance binding to a human FcRn, as described further herein.
In some embodiments, the antibody or antigen-binding fragment comprises an IgG3 isotype. In some embodiments, the antibody or antigen-binding fragment comprises an IgG3m(b*) allotype, an IgG3m(b**) allotype, an IgG3m(c3*) allotype, an IgG3m(c3c5*) allotype, an IgG3m(g*) allotype, an IgG3m(s*) allotype, an IgG3m(st*) allotype, an IgG3 GM5 allotype, an IgG3 G4m5 allotype, or any combination thereof.
In certain embodiments, an antibody or antigen-binding fragment comprises a CH3 sequence engineered to reduce potential immunogenicity of the antibody or antigen-binding fragment by replacing specific amino acids of one allotype with those of another allotype (these may be referred to as isoallotype mutations), as described in more detail in Stickler et al. (Genes Immun. 2011 Apr; 12(3): 213-221), which is herein incorporated by reference in its entirety, including, for example, the isoallotype mutations disclosed therein. In particular embodiments, specific amino acids of the Glml allotype are replaced. In a preferred embodiment, isoallotype mutations D356E and L358M are made in the CH3 sequence.
In certain embodiments, a variant of an Fc polypeptide comprises only the specified or recited amino acid mutations (e.g. substitutions), and does not comprise any further amino acid substitutions or mutations; e.g., relative to the reference polypeptide (e.g., a wild-type Fc polypeptide or fragment thereof). For example, in some embodiments, a variant Fc polypeptide comprising the amino acid substitutions G236A Y300L does not comprise any other amino acid substitutions; i.e., comprises an amino acid sequence that is wild-type except for G236A and Y300L.
In some embodiments, a variant of an Fc polypeptide may comprise one or more additional amino acid mutations (e.g. substitutions), which can be specified (e.g., M428L_N434S; M428L_N434A). In some embodiments, a further amino acid mutation or mutations is physically remote to the recited amino acid positions in tertiary structure, and/or is of such nature (e.g. is a conservative substitution), so that one or more function of the recited Fc variant or fragment thereof is not reduced or is reduced by no more than 50%, no more than 40%, no more than 30%, no more than 25%, no more than 20%, no more than 15%, no more than 10% or no more than 5%, or by no more than 10-fold, no more than 9-fold, no more than 8- fold, no more than 7-fold, no more than 6-fold, no more than 5-fold, no more than 4-fold, no more than 3-fold, no more than 2-fold, or no more than 1.5-fold. In some embodiments, variant of an Fc polypeptide comprises the mutations M428L and N434S or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, including those described herein.
In some embodiments, an antibody or antigen-binding fragment (described further herein) is provided that comprises, in a(n e.g. human) IgGl heavy chain, the amino acid mutation(s) set forth in any one of (i)-(xviii): (i) G236A, L328V, and Q295E; (ii) G236A, P230A, and Q295E; (iii) G236A, R292P, and I377N; (iv) G236A, K334A, and Q295E; (v) G236S, R292P, and Y300L; (vi) G236A and Y300L; (vii) G236A, R292P, and Y300L; (viii) G236S, G420V, G446E, and L309T; (ix) G236A and R292P; (x) R292P and Y300L; (xi) G236A and R292P; (xii) Y300L; (xiii) E345K, G236S, L235Y, and S267E; (xiv) E272R, L309T, S219Y, and S267E; (xv) G236Y; (xvi) G236W; (xvii) F243L, G446E, P396L, and S267E; (xviii) G236A, S239D, and H268E, wherein the numbering of amino acid residues is according to the EU index as set forth in Kabat. In certain embodiments, the antibody or antigen-binding fragment is afucosylated. In some embodiments, the antibody or antigen-binding fragment further comprises one or more mutation that enhances binding to a human FcRn, such as M428L and N434S mutations or M428L and N434A mutations (EU numbering) or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated. In some embodiments, the IgGl heavy chain comprises a CH1-CH3 or a CH2-CH3 or a hinge-CH2-CH3, wherein the CH1-CH3 or CH2-CH3 or hinge-CH2-CH3 has at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity (or similarity) to a wild-type human IgGl CH1-CH3 or CH2-CH3 or hinge-CH2-CH3, respectively. In certain embodiments, an antibody or antigen-binding fragment of the present disclosure comprises an Fc variant comprising an amino acid sequence having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity (or similarity) to the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765.
In some embodiments, the antibody or antigen-binding fragment comprises the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765, or a variant thereof, e.g. that further comprises one or more mutation that enhances binding to a human FcRn, such as M428L and N434S mutations or M428L and N434A mutations (EU numbering) or any other mutation(s) that enhance binding to a human FcRn, including those described herein. In some embodiments, the antibody or antigen-binding fragment comprises an amino acid sequence that differs from the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765 only by one or more IgGl allotype-specific mutations and/or by the presence of M428L and N434S
mutations or M428L and N434A mutations or other mutation(s) that enhance binding to a human FcRn.
Also provided are embodiments wherein an antibody or antigen-binding fragment comprises an Fc polypeptide comprising one or more of the following mutations to, e.g., in a human IgGl background, can promote binding to Cl : K236WZE333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L. Additionally or alternatively, an Fc polypeptide can comprise any mutation or mutations described in Table A to increase binding to Cl (or Clq).
Also provided are embodiments wherein an antibody or antigen-binding fragment comprises an Fc polypeptide comprising E345R/E430G/S440Y mutations to, e.g., in a human IgGl background, can promote oligomerization.
An antibody or antigen-binding fragment of the present disclosure can be fucosylated (e.g., comprising one or more fucosyl moiety, and typically comprising a native (wild-type) fucosylation pattern or a fucosylation pattern that includes one or more additional, or fewer, fucosyl moieties as compared to native), or can be afucosylated. In particular, native IgGl antibodies carry a glycan site at N297, and this is typically the only site where a core fucose moiety may be found in the antibody, though some glycan sites may arise through mutation (e.g. in the variable domains) during antibody development. Fucosylation of an antibody or antigenbinding fragment, can be affected by introducing amino acid mutations to introduce or disrupt a fucosylation site (e.g. a mutation at N297, such as N297Q or N297A, to disrupt formation of a glycan that can include a core fucose moiety), though typically it is preferred to maintain N297 and the glycan thereof, such as by expressing the antibody or antigen-binding fragment in a host cell which has been genetically engineered to lack the ability (or have an inhibited or compromised ability) to fucosylate the antibody or antigen-binding fragment; by expressing the antibody or antigen-binding fragment under conditions in which a host cell is impaired in its ability to fucosylate the polypeptide (e.g, in the presence of 2-fluoro-L-fucose (2FF)), or the like. An afucosylated antibody or antigen-binding fragment can comprise no fucose moieties, or substantially no fucose moieties, and/or can be expressed by a host cell that is genetically engineered to lack the ability (or have an inhibited or compromised ability) to fucosylate the antibody or antigen-binding fragment and/or can be expressed under conditions in which a host cell is impaired in its ability to fucosylate the antibody or antigen-binding fragment (e.g, in the presence of 2-fluoro-L-fucose (2FF)). In some embodiments, an antibody or antigen-binding fragment does not comprise a core fucose moiety at Asn297. In some embodiments,
afucosylated antibodies or antigen-binding fragments have increased binding to FcyRIIIA. In some contexts, addition of 2FF to a culture media comprising host cells expressing an antibody results in about 85% or more of the antibodies or antigen-binding fragments not carrying a fucose moiety. Accordingly, a plurality of antibodies or antigen-binding fragments may be described as “afucosylated” when the plurality was produced in the presence of 2FF or like reagent. In some contexts, a plurality of antibodies or antigen-binding fragments may be described as, for example, afucosylated, meaning that about 85% or more of the single antibody or antigen-binding fragment molecules of the plurality do not comprise a fucose moiety. In certain preferred embodiments, an afucosylated antibody or antigen-binding fragment or a population or a plurality thereof comprises an asparagine (N) at EU position 297. Fucosylation or lack thereof can be assessed using, for example, mass spectrometry (e.g. Electrospray mass spectrometry (ESI-MS)). In some embodiments, compositions are provided that comprise a plurality of any one or more of the presently disclosed antibodies or antigen-binding fragments, wherein the composition comprises afucosylated antibodies or antigen-binding fragments.
In certain embodiments, presently disclosed variants of IgG Fc polypeptides or fragments thereof possess one or more function that is distinct from (e.g. improved as compared to) the corresponding function of a reference Fc polypeptide that comprises the following mutation or mutations: G236A; G236S; G236A_A330L_I332E; G236A_A330L_I332E_M428L_N434S; A330L I332E; or G236A_S239D_A330L_I332E. For example, in certain embodiments, a presently disclosed variant of an IgG Fc polypeptide or fragment thereof possesses one or more of the following properties, as compared to a reference Fc polypeptide that comprises the following mutation or mutations: G236A; G236S; G236A_A330L_I332E; G236A_A330L_I332E_M428L_N434S; A330LJ332E; or G236A_S239D_A330L_I332E: increased binding (e.g. affinity) to and/or signaling via a human FcyRIIa H131; increased binding (e.g. affinity) to and/or signaling via a human FcyRIIa R131; decreased binding to (e.g. affinity) and/or signaling via human FcyRIIb; an increased ratio of binding to (e.g. affinity) and/or signaling via a human FcyRIIa (H131, R131, or both) versus the ratio of binding to or signaling via (respectively) a human FcyRIIb; increased binding (e.g. affinity) to and/or signaling via a human FcyRIIIa (V158, F158, or both); increased binding (e.g. affinity) to a human Clq; a higher Tm; an improved production titer; an improved signaling in a host cell via a FcyRIIa (H131, R131, or both); increased facilitation of ADCP and/or ADCC by human NK cells and/or human PBMCs when in the presence of antigen-presenting cells; and an improved ability to stimulate moDCs when in an immune complex with antigen.
In the present disclosure, binding of a variant Fc polypeptide or fragment may be described as increased (or “greater than”, or the like) or decreased (or “reduced” or “less than”, or the like) as compared to the binding of a comparator (e.g., to a reference wild-type IgGl Fc, or to a reference IgGl Fc that is wild-type except for M428L and N434S mutations or except for M428L and N434A mutations or to a variant IgGl Fc comprising G236A_A330L_I332E mutations) to a same binding partner. Binding interactions between a variant Fc polypeptide or fragment (or an antibody or polypeptide comprising the same) and a binding partner (e.g. a human FcyR, FcRn, or Clq) can preferably be determined using an electrochemiluminescence assay, more preferably using the Meso Scale Discovery (“MSD”; mesoscale.com) platform. MSD binding assay is similar to ELISA though MSD uses electrochemiluminescence, as opposed to colorimetry, as a detection technique. Other techniques for measuring binding interactions are known and include, for example, ELISA, surface plasmon resonance (SPR), biolayer interferometry (BLI), and the like.
In some embodiments, binding includes affinity, avidity, or both. Affinity refers to the strength of a bond between a binding molecule and its binding partner. In some contexts, binding can include affinity and/or avidity. Unless otherwise indicated, avidity refers to the total binding strength of a molecule to a binding partner, and reflects binding affinity, valency of binding sites (or domains) (e.g., whether an Fc polypeptide comprises one, two, or more binding sites (or domains)), and, for example, whether another agent is present that can affect the binding (e.g., a non-competitive inhibitor of the Fc polypeptide).
A binding interaction between a variant molecule of the present disclosure and a binding partner can be expressed in terms of fold-change relative to the binding interaction between a reference molecule and the binding partner. For example, binding of a presently disclosed antibody comprising a variant Fc to a human FcyRIIa may be stronger than the binding of the antibody comprising a wild-type Fc to the human FcyRIIa, and the relative increased strength of the variant can be expressed in terms of fold-change e.g., linear scale of area-under-the-curve) relative to the reference molecule binding using the same assay. For example, a variant Fc polypeptide or fragment may bind to a FcyRIIa with a 2-fold, 3-fold, 4-fold, or 5-fold greater binding strength than a reference Fc polypeptide or fragment binds to the FcyRIIa. As another example, a variant Fc polypeptide or fragment thereof may bind less strongly to a FcyRIIb as compared to a reference Fc or fragment thereof; e.g., may have a 0.9-fold binding, 0.8-fold binding, 0.7-fold binding, 0.6-fold binding, or the like, as compared to the reference Fc polypeptide or fragment thereof. It will be understood that, for example, the expression “2-fold
greater binding as compared to the binding of a reference” means a 2-fold increase in binding as compared to the reference.
Moreover, binding of a variant Fc molecule of the present disclosure to two different partner molecules can be described in terms of a ratio, and this ratio can be compared to a like ratio obtained using a reference molecule with the same assay. For example, a variant Fc polypeptide may bind to a human FcyRIIa H131 five times more strongly than it binds to a human FcyRIIb, while a reference wild-type Fc polypeptide binds to FcyRIIa H131 as strongly as it binds to a human FcyRIIb. In this example, the variant Fc polypeptide can be said to have a 5: 1 (binding FcyRIIIa H131 :binding FcyRIIb) binding ratio, which can be compared to the 1 : 1 (binding FcyRIIIa H131 :binding FcyRIIb) binding ratio of the reference wild-type Fc polypeptide.
Variant Fc molecules of the present disclosure may also be described in terms of ability to induce signaling in a host cell, wherein the host cell expresses or over-expresses one or more FcyR (e.g., FcyRIIa H131, FcyRIIa R131, FcyRIIb, FcyRIIIa F158, or FcyRIIIa V158) and the signaling is induced by binding of the variant molecule to the FcyR. Reporter cells useful for determining signaling include, for example, cells in which NF AT drives expression of a luciferase reporter (e.g., available from Promega®).
Unless stated otherwise, FcyRs, FcRn, and Cl (e.g., Clq) as described herein are human.
In some embodiments, an antibody or antigen-binding fragment comprising a variant Fc polypeptide or fragment is preferably capable of inducing one or more of: antibody-dependent cell cytotoxicity (ADCC), antibody-dependent cellular phagocytosis (ADCP); and complementdependent cytotoxicity. Assays for measuring these functions are known.
In some embodiments, a variant Fc polypeptide or fragment (or antibody or antigenbinding fragment comprising the same) preferably has comparable binding to a human FcRn (e.g., at pH 6.0) and/or a comparable in vivo half-life in a mammal as compared to a reference Fc polypeptide, fragment, antibody, or antigen-binding fragment, respectively.
In some embodiments, a variant Fc polypeptide or fragment (or antibody or antigenbinding fragment comprising the same) preferably has increased binding to a human FcRn (e.g., at pH 6.0) and/or increased in vivo half-life in a mammal as compared to a reference Fc polypeptide, fragment, antibody, or antigen-binding fragment, respectively.
In some embodiments, a variant Fc polypeptide or fragment (or antibody or antigenbinding fragment comprising the same) preferably has a melting temperature (Tm) that is less than 12°C, less than 11°C, less than 10°C, less than 9°C, less than 8°C, less than 7°C, less than
6°C, less than 5°C, less than 4°C, less than 3°C, less than 2°C, or less than 1°C below the Tm of a reference Fc polypeptide (or antibody or antigen-binding fragment comprising the same), or has a Tm that is higher than the Tm of the reference Fc polypeptide or fragment (or polypeptide or antibody comprising the same). In some embodiments, the reference polypeptide or fragment is or comprises a wild-type human Fc polypeptide (or antibody comprising the same).
In some embodiments, a variant Fc polypeptide or fragment (or antibody or antigenbinding fragment comprising the same) has a melting temperature that is higher than the melting temperature of a reference Fc polypeptide or fragment (or antibody or antigen-binding fragment comprising the same) that comprises the mutations G236A, A330L, I332E, and, optionally, M428L and N434S,
In some embodiments, a variant Fc polypeptide or fragment (or antibody or antigenbinding fragment comprising the same) is preferably capable of being produced in a host cell line (e.g., a CHO cell line) at least about as efficiently (e.g., produces at least about the same titer and/or within less than O. l-fold, less than 0.09-fold, less than 0.08-fold, less than 0.07-fold, less than 0.06-fold, less than 0.05-fold, less than 0.04-fold, less than 0.03-fold, less than 0.02-fold, or less than 0.02 -fold less) as compared to a reference Fc polypeptide or fragment (or antibody or antigen-binding fragment comprising the same).
In certain embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a valine (V) at EU position 328, and a glutamic acid (E) at EU position 295. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GALVQE”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG hinge-CH2 polypeptide; or (ii) an IgG hinge-Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an alanine (A) at EU position 230, and a glutamic acid (E) at EU position 295. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wildtype) IgGl Fc polypeptide or fragment thereof (“GAPAQE”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the
mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a proline (P) at EU position 292, and an asparagine (N) at EU position 377. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARPIN”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an alanine (A) at EU position 334, and a glutamic acid (E) at EU position 295. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GAKAQE”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a serine (S) at EU position 236, a proline (P) at EU position 292, and a leucine (L) at EU position 300. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GSRPYL”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a proline (P)
at EU position 292, and a leucine (L) at EU position 300. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARPYL”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236 and a leucine (L) at EU position 300. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GAYL”). In some embodiments, the antibody or antigen-binding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In certain other embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of: (i) an IgG CH2 polypeptide; or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an aspartic acid (D) at EU position 239, and a glutamic acid (E) at EU position 268. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GASDHE”). In some embodiments, the antibody or antigenbinding fragment further comprises the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein. In certain embodiments, the antibody or antigen-binding fragment is afucosylated.
In some embodiments, the antibody or antigen-binding fragment has increased binding to a human FcyRIIa and/or has decreased binding to a human FcyRIIb, as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa or the human FcyRIIb, respectively, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
In certain embodiments, the increased binding to a human FcyRIIa comprises more than 1-fold, at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold greater binding to the human FcyRIIa as
compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa.
In some embodiments, the human FcyRIIa comprises H131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold greater binding to the human FcyRIIa H131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa H131.
In some embodiments, the human FcyRIIa comprises R131 and, optionally, the increased binding to the human FcyRIIa R131 comprises more than 1-fold, at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold greater binding to the human FcyRIIa R131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa R131.
In some embodiments, the decreased binding to a human FcyRIIb comprises less than 0.9-fold, less than 0.8-fold, less than 0.7-fold, less than 0.6-fold, or between 0.5-fold and 0.9- fold, of the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIb.
In any of the presently disclosed embodiments, (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a reference polypeptide to the human FcyRIIa to (iv) the binding of the reference antibody or antigen-binding fragment to the human FcyRIIb, wherein the reference antibody or antigen-binding fragment optionally comprises a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery. In some embodiments, the human FcyRIIa comprises H131, R131, or both. In some embodiments, the ratio in (1) is more than 1-fold, at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11 -fold, at least 12-fold, at least 13-fold, or at least 14-fold greater than the ratio in (2).
Also provided is an antibody or antigen-binding fragment of the present disclosure comprising a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment
thereof, wherein the variant comprises an alanine (A) at EU position 236 and a leucine (L) at EU position 300. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GAYL”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
In some embodiments, the antibody or antigen-binding fragment has increased binding to a human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
In some embodiments, the increased binding to a human FcyRIIa comprises at least 4- fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13-fold, at least 14-fold, at least 15-fold, at least 16-fold, at least 17-fold, or at least 18-fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa.
In some embodiments, the human FcyRIIa comprises H131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 4-fold, at least 5-fold, at least 6- fold, at least 7-fold, at least 8-fold, at least 9-fold, or at least 10-fold, at least 11-fold, at least 12- fold, at least 13-fold, at least 14-fold, at least 15-fold, at least 16-fold, at least 17-fold, or at least 18-fold greater binding to the human FcyRIIa H131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g. IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa H131.
In some embodiments, the human FcyRIIa comprises R131 and, optionally, the increased binding to the human FcyRIIa R131 comprises at least 4-fold greater binding to the human FcyRIIa R131 as compared to the binding of a reference antibody or antigen-binding fragment (optionally comprising a wild-type human IgG (e.g IgGl) Fc polypeptide or a fragment thereof) to the human FcyRIIa R131.
In certain embodiments, (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa to (iv) the binding of the reference polypeptide to the human FcyRIIb, wherein the reference antibody or antigen-binding fragment
comprises a wild-type human IgG Fc polypeptide or a fragment thereof. In certain embodiments, the human FcyRIIa comprises H131, R131, or both. In further embodiments, the ratio in (1) is at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13-fold, at least 14-fold, at least 15-fold, at least 16-fold, or at least 17-fold greater than the ratio in (2).
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, a proline (P) at EU position 292, and a leucine (L) at EU position 300. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARPYL”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
In certain embodiments, the antibody or antigen-binding fragment has increased binding to a human FcyRIIIa, as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
In some embodiments, the increased binding to a human FcyRIIa comprises at least 2- fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11 -fold, at least 12-fold, at least 13 -fold, or at least 14-fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa.
In some embodiments, the human FcyRIIa comprises H131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 2-fold, at least 3-fold, at least 4- fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13-fold, or at least 14-fold greater binding to the human FcyRIIa H131 as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa H131.
In some embodiments, the human FcyRIIa comprises R131 and, optionally, the increased binding to the human FcyRIIa H131 comprises at least 2-fold greater binding to the human
FcyRIIa R131 as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa R131.
In certain embodiments, (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment, respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa to (iv) the binding of the reference antibody or antigen-binding fragment to the human FcyRIIb, wherein the reference antibody or antigen-binding fragment optionally comprises a wild-type human IgG Fc polypeptide or a fragment thereof, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery. In some embodiments, the human FcyRIIa comprises H131, R131, or both. In some embodiments, the ratio in (1) is at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 9-fold, at least 10-fold, at least 11-fold, at least 12-fold, at least 13- fold, at least 14-fold, or at least 15-fold greater than the ratio in (2).
In certain embodiments, the antibody or antigen-binding fragment has increased binding to a human FcyRIIIa, as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery. In some embodiments, the human FcyRIII comprises V158, F158, or both. In certain further embodiments, the increased binding to a human FcyRIIIa comprises greater than 2-fold, at least 2.1-fold, at least 2.2-fold, at least 2.3-fold, at least 2.4-fold, at least 2.5-fold, at least 2.6-fold, at least 2.7-fold, at least 2.8-fold, at least 2.9-fold, at least 3.0 fold, at least 3.1-fold, at least 3.2- fold, at least 3.3-fold, at least 3.4-fold, at least 3.5-fold, at least 3.6-fold, or at least 3.7-fold greater binding to the human FcyRIIIa as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIIa.
In certain embodiments, the antibody or antigen-binding fragment is capable of binding to a human complement component Iq (Clq), wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of an IgG Fc polypeptide, wherein the variant comprises a serine (S) at EU position 236, a valine (V) at EU position 420, a glutamic acid (E) at EU position 446, and a
threonine (T) at EU position 309. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GSGVGELT”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigenbinding fragment is afucoyslated.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide, wherein the variant comprises an alanine (A) at EU position 236 and a proline (P) at EU position 292. In some embodiments, the antibody or antigen-binding fragment comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GARP”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
In certan embodiments, the antibody or antigen-binding fragment has decreased binding to a human FcyRIIb as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIb, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery. In some embodiments, the decreased binding to a human FcyRIIb comprises less than 0.9-fold, less than 0.8-fold, less than 0.7-fold, less than 0.6-fold, less than 0.5-fold, or less than 0.4-fold as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIb.
In further embodiments, the antibody or antigen-binding fragment has increased binding to a human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIa, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery.
In some embodiments, the increased binding to the human FcyRIIa comprises greater than 1-fold, at least 2-fold, at least 3 -fold, at least 4-fold, or at least 5 -fold greater binding to the human FcyRIIa as compared to the binding of a reference antibody or antigen-binding fragment comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa.
In certain embodiments, the human FcyRIIa comprises H131, R131, or both.
In some embodiments, (1) a ratio of (i) the binding of the antibody or antigen-binding fragment to a human FcyRIIa to (ii) the binding of the antibody or antigen-binding fragment,
respectively, to a human FcyRIIb is greater than (2) a ratio of (iii) the binding of a antibody or antigen-binding fragment to the human FcyRIIa to (iv) the binding of the reference antibody or antigen-binding fragment to the human FcyRIIb, wherein the reference antibody or antigenbinding fragment optionally comprises a wild-type human IgG Fc polypeptide or a fragment thereof, wherein, optionally, binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery. In some embedments, the human FcyRIIa comprises H131, R131, or both. In certain embodiments, the ratio in (1) is at least 2-fold, at least 3-fold, at least 4-fold, at least 5-fold, at least 6-fold, at least 7-fold, at least 8-fold, at least 10-fold, at least 11-fold, or at least 12-fold greater than the ratio in (2).
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide, wherein the variant comprises a proline (P) at EU position 292 and a leucine (L) at EU position 300, and wherein, optionally, variant and, further optionally, the antibody or antigen-binding fragment has increased binding to a human FcyRIIIa with as compared to the binding of a reference antibody or antigen-binding fragment to the human FcyRIIIa, wherein, optionally, the binding is as determined using an electrochemiluminescence assay, further optionally Meso Scale Discovery. In some embodiments, the antibody or antigen-binding fragment comprises an (e.g., otherwise wild-type) IgGl CH2 polypeptide or IgG Fc polypeptide (“RPYL”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
In certain embodiments, the human FcyRIIIa comprises V158, F158, or both, and wherein the increased binding to the human FcyRIIIa comprises at least 4-fold, at least 4.5-fold, at least 5-fold, at least 5.1-fold, or at least 5.2-fold greater binding as compared to the binding of a reference antibody or antigen-binding fragment optionally comprising a wild-type human IgG Fc polypeptide or a fragment thereof to the human FcyRIIa.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a leucine (L) at EU position 300. In some embodiments, the IgG CH2 polypeptide or IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“YL”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any
other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a lysine (K) at EU position 345, a serine (S) at EU position 236, tyrosine (Y) at EU position 235, and a glutamic acid (E) at EU position 267. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GSEKLYSE”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
Also provided is antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG hinge-CH2 polypeptide or (ii) an IgG hinge-Fc polypeptide or a fragment thereof, wherein the variant comprises an arginine (R) at EU position 272, a threonine (T) at EU position 309, a tyrosine (Y) at EU position 219, and a glutamic acid (E) at EU position 267. In some embodiments, the IgG hinge-CH2 polypeptide or an IgG hinge-Fc polypeptide or a fragment thereof comprises an (e.g. otherwise wild-type) IgGl hinge-CH2 polypeptide or IgG hinge-Fc polypeptide or a fragment thereof (“SYSEERLT”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a tyrosine (Y) at EU position 236. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GY”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a tryptophan (W) at EU position 236. In some embodiments, the IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof
comprises an (e.g., otherwise wild-type) IgGl CH2 polypeptide or Fc polypeptide or fragment thereof (“GW”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigenbinding fragment is afucoyslated.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, wherein the IgG Fc polypeptide or fragment thereof, and optionally the polypeptide, is afucosylated, and wherein, further optionally, the variant comprises a leucine (L) at EU position 330 and a glutamic acid (E) at EU postion 332, wherein, still further optionally, the variant does not comprise an aspartic acid (D) at EU position 239, and, even further optionally, comprises a serine (S) at EU position 239. In some embodiments, the IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof comprises an (e.g., otherwise wild-type) IgGl CH2 polypeptide or Fc polypeptide or fragment thereof (“GA-afuc” or “GAALIE-afuc”, respectively). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present.
Also provided is an antibody or antigen-binding fragment of the present disclosure that comprises a variant of: an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises a leucine (L) at EU position 243, a glutamic acid (E) at EU position 446, a leucine (L) at EU position 396, and a glutamic acid (E) at EU position 267. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“FLSEPLGE”). In certain further embodiments, the mutations M428L and N434S, or the mutations M428L and N434A, or any other mutation(s) that enhance binding to a human FcRn, such as those described herein, are present. In certain embodiments, the antibody or antigen-binding fragment is afucoyslated.
Also provided is an antibody or antigen-binding fragment that comprises a variant of: (i) an IgG CH2 polypeptide or (ii) an IgG Fc polypeptide or a fragment thereof, wherein the variant comprises an alanine (A) at EU position 236, an aspartic acid (D) at EU position 239, a glutamic acid (E) and EU position 332, a leucine (L) at EU position 428, and a serine (S) or an alanine (A) at EU position 434. In some embodiments, the IgG Fc polypeptide or fragment thereof comprises an (e.g., otherwise wild-type) IgGl Fc polypeptide or fragment thereof (“GASDIEMLNS” or “GASDIEMLNA”).
In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of an (e.g. IgGl) IgG Fc polypeptide, wherein the variant comprises the following mutations, according to EU numbering: (i) M428L, N434S, G236A, L328V, and Q295E; (ii) M428L, N434S, G236A, R292P, and I377N; (iii) M428L, N434S, G236A, and Y300L; (iv) M428L, N434S, G236A, R292P, and Y300L; (v) M428L, N434S, G236A, L328V, and Q295E, wherein the antibody or antigen-binding fragment is afucosylated; (vi) M428L, N434S, G236A, R292P, and I377N, wherein the antibody or antigen-binding fragment is afucosylated; (vii) M428L, N434S, G236A, and Y300L, wherein the antibody or antigen-binding fragment is afucosylated; or (viii) M428L, N434S, G236A, R292P, and Y300L, wherein the antibody or antigen-binding fragment is afucosylated. In some embodiments, the variant of an (e.g. IgGl) IgG Fc polypeptide comprises amino acid substitutions that consist essentially of the substitution mutations in (i), (ii), (iii), (iv), (v), (vi), (vii), or (viii) above. In some embodiments, the antibody or antigen-binding fragment comprises a kappa light chain.
In some embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a variant of an (e.g. IgGl) IgG Fc polypeptide, wherein the variant comprises the following mutations, according to EU numbering: (i) M428L, N434A, G236A, L328V, and Q295E; (ii) M428L, N434A, G236A, R292P, and I377N; (iii) M428L, N434A, G236A, and Y300L; (iv) M428L, N434A, G236A, R292P, and Y300L; (v) M428L, N434A, G236A, L328V, and Q295E, wherein the antibody or antigen-binding fragment is afucosylated; (vi) M428L, N434A, G236A, R292P, and I377N, wherein the antibody or antigen-binding fragment is afucosylated; (vii) M428L, N434A, G236A, and Y300L, wherein the antibody or antigenbinding fragment is afucosylated; or (viii) M428L, N434A, G236A, R292P, and Y300L, wherein the antibody or antigen-binding fragment is afucosylated. In some embodiments, the variant of an IgG Fc polypeptide comprises amino acid substitutions that consist essentially of the substitution mutations in (i), (ii), (iii), (iv), (v), (vi), (vii), or (viii) above. In some embodiments, the antibody comprises a kappa light chain. In certain embodiments, the antibody or antigenbinding fragment is capable of eliciting continued protection in vivo in a subject even once no detectable levels of the antibody or antigen-binding fragment can be found in the subject (z.e., when the antibody or antigen-binding fragment has been cleared from the subject following administration). Such protection is referred to herein as a vaccinal effect. Without wishing to be bound by theory, it is believed that dendritic cells can internalize complexes of antibody and antigen and thereafter induce or contribute to an endogenous immune response against antigen. In certain embodiments, an antibody or antigen-binding fragment comprises one or more
modifications, such as, for example, mutations in the Fc comprising G236A, A330L, and I332E, that are capable of activating dendritic cells that may induce, e.g., T cell immunity to the antigen. In any of the presently disclosed embodiments, the antibody or antigen-binding fragment comprises a Fc polypeptide or a fragment thereof, including a CH2 (or a fragment thereof, a CH3 (or a fragment thereof), or a CH2 and a CH3, wherein the CH2, the CH3, or both can be of any isotype and may contain amino acid substitutions or other modifications as compared to a corresponding wild-type CH2 or CH3, respectively. In certain embodiments, a Fc of the present disclosure comprises two CH2-CH3 polypeptides that associate to form a dimer.
Non-limiting examples of heavy chain or Fc amino sequences - including certain sequences comprising amino acid substitution mutations as disclosed herein - are provided in SEQ ID NOs.:251-277 and 765. In certain embodiments, an antibody or antigen-binding fragment of the present disclosure comprises a heavy chain comprising an amino acid sequence having at least 85%, at least 86%, at least 87%, at least 88%, at least 89%, at least 90%, at least 91%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity (or similarity) to, or comprising or consisting of, the amino acid sequence set forth in any one of SEQ ID NOs.:251-277 and 765.
In any of the presently disclosed embodiments, the antibody or antigen-binding fragment can be monoclonal. The term “monoclonal antibody” (mAb) as used herein refers to an antibody obtained from a population of substantially homogeneous antibodies, z.e., individual antibodies comprising the population are identical except for possible naturally occurring mutations that may be present, in some cases in minor amounts. Monoclonal antibodies are highly specific, being directed against a single antigenic site. Furthermore, in contrast to polyclonal antibody preparations that include different antibodies directed against different epitopes, each monoclonal antibody is directed against a single epitope of the antigen. In addition to their specificity, the monoclonal antibodies are advantageous in that they may be synthesized uncontaminated by other antibodies. The term “monoclonal” is not to be construed as requiring production of the antibody by any particular method. For example, monoclonal antibodies useful in the present invention may be prepared by the hybridoma methodology first described by Kohler et al., Nature 256 :495 (1975), or may be made using recombinant DNA methods in bacterial, eukaryotic animal, or plant cells (see, e.g., U.S. Pat. No. 4,816,567). Monoclonal antibodies may also be isolated from phage antibody libraries using the techniques described in Clackson et al., Nature, 352:624-628 (1991) and Marks et al., J. Mol. Biol., 222:581-597 (1991),
for example. Monoclonal antibodies may also be obtained using methods disclosed in PCT Publication No. WO 2004/076677A2.
Antibodies and antigen-binding fragments of the present disclosure include “chimeric antibodies” in which a portion of the heavy and/or light chain is identical with or homologous to corresponding sequences in antibodies derived from a particular species or belonging to a particular antibody class or subclass, while the remainder of the chain(s) is identical with or homologous to corresponding sequences in antibodies derived from another species or belonging to another antibody class or subclass, as well as fragments of such antibodies, so long as they exhibit the desired biological activity (see, U.S. Pat. Nos. 4,816,567; 5,530,101 and 7,498,415; and Morrison et al., Proc. Natl. Acad. Sci. USA, 57:6851-6855 (1984)). For example, chimeric antibodies may comprise human and non-human residues. Furthermore, chimeric antibodies may comprise residues that are not found in the recipient antibody or in the donor antibody. These modifications are made to further refine antibody performance. For further details, see Jones etal., Nature 321 :522-525 (1986); Riechmann etal., Nature 332:323-329 (1988); and Presta, Curr. Op. Struct. Biol. 2:593-596 (1992). Chimeric antibodies also include primatized and humanized antibodies.
A “humanized antibody” is generally considered to be a human antibody that has one or more amino acid residues introduced into it from a source that is non-human. These non-human amino acid residues are typically taken from a variable domain. Humanization may be performed following the method of Winter and co-workers (Jones et al., Nature, 321 : 522-525 (1986); Reichmann et al., Nature, 332:323-327 (1988); Verhoeyen et al., Science, 239: 1534- 1536 (1988)), by substituting non-human variable sequences for the corresponding sequences of a human antibody. Accordingly, such “humanized” antibodies are chimeric antibodies (U.S. Pat. Nos. 4,816,567; 5,530,101 and 7,498,415) wherein substantially less than an intact human variable domain has been substituted by the corresponding sequence from a non-human species. In some instances, a “humanized” antibody is one which is produced by a non-human cell or animal and comprises human sequences, e.g., He domains.
A “human antibody” is an antibody containing only sequences that are present in an antibody that is produced by a human (i.e., sequences that are encoded by human antibodyencoding genes). However, as used herein, human antibodies may comprise residues or modifications not found in a naturally occurring human antibody (e.g., an antibody that is isolated from a human), including those modifications and variant sequences described herein. These are typically made to further refine or enhance antibody performance. In some instances,
human antibodies are produced by transgenic animals. For example, see U.S. Pat. Nos. 5,770,429; 6,596,541 and 7,049,426.
In certain embodiments, an antibody or antigen-binding fragment of the present disclosure is chimeric, humanized, or human.
In some embodiments, various pharmacokinetic (“PK”) parameters are used to describe or characterize the antibodies or antigen-binding fragments provided herein. Details regarding collection of antibody serum concentrations for purpose of evaluating PK parameters are described in association with the Examples herein. The term “ti/2” or “half-life” refers to the elimination half-life of the antibody or antigen-binding fragment included in the pharmaceutical composition administered to a subject. The term “Clast” generally refers to the last measurable plasma concentration (z.e., subsequent thereto, the substance is not present at a measurable concentration in plasma).
In some embodiments, the antibody, or the antigen-binding fragment, comprises a human antibody, a monoclonal antibody, a purified antibody, a single chain antibody, a Fab, a Fab’, a F(ab’)2, or Fv.
In some embodiments, the antibody or antigen-binding fragment is a multi-specific antibody or antigen-binding fragment, wherein, optionally, the antibody or antigen-binding fragment is a bi-specific antibody or antigen-binding fragment.
In some embodiments, the antibody or antigen-binding fragment comprises an (e.g., IgGl) Fc polypeptide or a fragment thereof. In some embodiments, the antibody or antigenbinding fragment comprises an IgG, IgA, IgM, IgE, or IgD isotype. In further embodiments, the antibody or antigen-binding fragment comprises an IgG isotype selected from IgGl, IgG2, IgG3, and IgG4. In certain embodiments, the antibody or antigen-binding fragment comprises an IgGl isotype. In some embodiments, the antibody or antigen-binding fragment comprises an IgGlm3 allotype, an IgGlml7 allotype, an IgGl ml allotype, or any combination thereof. In some embodiments, the antibody or antigen-binding fragment comprises an IgG3 isotype. In some embodiments, the antibody or antigen-binding fragment comprises an IgG3m(b*) allotype, an IgG3m(b**) allotype, an IgG3m(c3*) allotype, an IgG3m(c3c5*) allotype, an IgG3m(g*) allotype, an IgG3m(s*) allotype, an IgG3m(st*) allotype, an IgG3 GM5 allotype, an IgG3 G4m5 allotype, or any combination thereof.
In some embodiments, the Fc polypeptide or fragment thereof comprises: (i) a mutation that increases binding affinity to a human FcRn (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols)), as
compared to a reference Fc polypeptide that does not comprise the mutation; and/or (ii) a mutation that increases binding affinity to a human FcyR (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols, and/or as measured using mesoscale discovery (MSD))) as compared to a reference Fc polypeptide that does not comprise the mutation.
In some embodiments, the mutation that increases binding affinity to a human FcRn comprises: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q31 II; D376V; T307A; E380A; or any combination thereof. In some embodiments, the mutation that increases binding affinity to a human FcRn comprises: (i) M428L/N434S; (ii) M252Y/S254T/T256E; (iii) T250Q/M428L; (iv) P257VQ311I; (v) P257I/N434H; (vi) D376V/N434H; (vii) T307A/E380A/N434A; (viii) M428L/N434A; or (ix) any combination of (i)-(viii). In some embodiments, the mutation that increases binding affinity to a human FcRn comprises M428L/N434S or M428L/N434A.
In some embodiments, the mutation that enhances binding to a FcyR comprises S239D; I332E; A330L; G236A; or any combination thereof. In some embodiments, the mutation that enhances binding to a FcyR comprises: (i) S239D/I332E; (ii) S239D/A330L/I332E; (iii) G236A/S239D/I332E; or (iv) G236A/A330L/I332E, wherein the Fc polypeptide or fragment thereof optionally comprises Ser at position 239.
In some embodiments, the antibody or antigen-binding fragment comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or which is aglycosylated, and/or which is afucosylated.
Also provided are antibody conjugates that comprise an antibody or antigen-binding fragment of the present disclosure conjugated to a payload molecule or moiety, such as, for example, a drug compound.
Conjugation of a compound to an antibody or antigen-binding fragment can be accomplished using known means. Antibody or antigen-binding fragment structures amenable to conjugation include, for example, lysine amino acid residues, cysteine amino acid residues, nonnatural amino acids (e.g., amino acid analogs), carbohydrate moi eties, and N-termini of heavy and light chains (or of VH and VL). Such structures may be native to the antibody or antigenbinding fragment, or may be introduced (e.g., a native non-lysine or non-cysteine amino acid of the antibody may be replaced by a lysine or cysteine, and/or a lysine or cysteine may be inserted into the amino acid sequence of the antibody or antigen-binding fragment, as an additional amino acid). Also contemplated are conjugation techniques in which a protein with a strong interaction
with an antibody or antigen-binding fragment (e.g., FcRn, Protein A, Protein G, or fragments of these) is conjugated to a compound and non-covalent interaction between the protein and the antibody or antigen-binding fragment effectively couples the compound to the antibody or antigen-binding fragment, as well as modification in nucleotide-binding sites (e.g., for modification by photoaffinity labeling), or in catalytic sites (if present). Non-limiting examples of antibody or antigen-binding fragment conjugation technologies and mutations to facilitate conjugation are disclosed in, for example, PCT Publication Nos. WO 2006/034488, WO 2011/005481, WO 2011/156328, WO 2016/040856, WO 2015/157595, WO 2013/093809, WO 2005/018572, WO 1996/014339, WO 2008/070593, WO 2009/092011, WO 2020/191306, WO 2006/074397, WO 2008/038024, WO 2010/141902, and WO 2004/042017, as well as in Sang et al., Methods Mol. Biol. 2078:235-250 (2020), Haque et al. Chem. Commun. 57:10689-10702 (2021), Brun and Gauzy -Lazo, Methods Mol. Biol. 1045 Al 3 -187 (2013), and Yoder et al., Mol. Pharmaceuticals 76(9):3926-3927 (2019); the techniques, linkers, conjugation sites and conjugation chemistries, and mutations described in the foregoing documents are incorporated herein by reference. Non-limiting examples of mutations include the following (all residue positions according to EU numbering of human IgGl): Al 18C, S239C, T289C, A330C, and S442C in the heavy chain, and K149C in the light chain.
An antibody or antigen-binding fragment of a conjugate can comprise one or more native lysine, one or more native cysteine, one or more introduced lysine, one or more introduced cysteine, or any combination thereof. In embodiments where an antibody or antigen-binding fragment comprises its native lysines, its native cysteines, or both. In some embodiments, an antibody or antigen-binding fragment does not comprise modifications in its amino acid sequence for conjugation. Conjugation may comprise forming conjugates at all, or at fewer than all, of the available conjugation sites.
In certain embodiments, a payload molecule is covalently linked by a linker to the antibody or antigen-binding fragment thereof. In certain embodiments the linker is selected from a cleavable linker and a non-cleavable linker. In certain embodiments the cleavable linker is a protease-sensitive linker, a pH-sensitive linker, or a glutathione-sensitive linker. In certain embodiments, the cleavable linker is a protease-sensitive linker comprising a valine-citrulline dipeptide. In some embodiments, the linker comprises a maleimide group. In certain embodiments, the herein disclosed antibody or antigen-binding fragment thereof comprises a reduced disulfide bridge in a hinge region and the reduced disulfide bridge is coupled to the
maleimide group. Also provided herein are embodiments in which the linker further comprises a self-demolishing group, such as, for example, para-amino benzyl alcohol (PABC).
Various techniques may be used to couple a payload molecule to an antibody or antigenbinding fragment thereof to form an antibody or antigen-binding fragment conjugate of the present disclosure. In some embodiments, an antibody antigen-binding fragment conjugate comprises payload molecule that is covalently linked by a linker to the antibody or antigenbinding fragment thereof. Linkers used in antibody antigen-binding fragment conjugates may be organic compounds that fall into one of two groups, organized according to the mechanism by which the payload molecule is released from the carrier molecule. Cleavable linkers are designed to be selectively degraded or cleaved according to an inherent property of the target cell: three types of cleavable linkers are protease-sensitive linkers (whereby cleavage of the linker, e.g., a linker comprising a valine-citrulline or phenylalanine-lysine dipeptide or a tetrapeptide (e.g., GFLG or ALAL), by proteases present in a tumor cell lysosome releases the payload molecule); pH-sensitive linkers, containing an acid labile group that is selectively hydrolyzed by the lower pH of endosomal and lysosomal compartments, relative to cytosolic pH; and glutathione-sensitive linkers, which comprise a disulfide bridge that is reduced by intracellular glutathione. Non-cleavable linkers rely on non-specific degradation of the antibody conjugate to release the payload molecule.
Specific linkers, payloads, linker chemistries, and related mechanisms and methods are disclosed in Nareshkumar et al., Pharm. Res. 32:3526-3540 (2015), which compositions, methods, and techniques are herein incorporated by reference in their entirety. In certain embodiments, an antibody or antigen-binding fragment conjugate comprises a linker is selected from a cleavable linker and a non-cleavable linker. In further embodiments, the linker is a cleavable linker selected from a protease-sensitive linker, a pH-sensitive linker, or a glutathionesensitive linker. In specific embodiments, a cleavable linker is a protease-sensitive linker comprising a valine-citrulline dipeptide.
A linker may be connected or coupled to the antibody or antigen-binding fragment thereof using any appropriate technique or mechanism. In some embodiments, a linker comprises a maleimide group (optionally PEGylated) capable of reacting with a reduced disulfide bridge in a hinge region of the antibody or antigen-binding fragment thereof. Other sites on the carrier molecule (i.e., the antibody or antigen-binding fragment thereof) suitable for conjugation to a linker may be introduced or engineered using recombinant techniques, such as introducing cysteine residues or non-natural amino acids for site-specific conjugation. Methods for
introducing such modifications include, for example, the method described in Examples 6.3-7 of PCT Publication No. WO 2012/032181.
In some embodiments, a linker further comprises a self-demoli shing group, also referred to as a self-immolative group or a self-immolative spacer, to assist in a selective cleavage reaction. In certain embodiments, the self-demoli shing group is para-amino benzyl alcohol (PABC).
Click chemistries useful for generating antibody conjugates include those described in Meyer et al.. Bioconjug. Chem. 27(12):2791-2807 (2016), and are herein incorporated by reference in their entirety.
In any of the antibody or antigen-binding fragment conjugates described herein, the payload molecule may be selected from a therapeutic agent and a detectable indicator.
Polynucleotides, Vectors, DNA Therapeutics, RNA Therapeutics, and Host cells
In another aspect, the present disclosure provides isolated polynucleotides that encode any of the presently disclosed antibodies or an antigen-binding fragment thereof, or a portion thereof (e.g., a CDR, a VH, a VL, a Fab, a Fd, a heavy chain, or a light chain, or a heavy chain and a light chain), or that encode a presently disclosed polypeptide.
In certain embodiments, the polynucleotide comprises deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), wherein the RNA optionally comprises messenger RNA (mRNA).
In some embodiments, the polynucleotide comprises a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof. In certain embodiments, the polynucleotide comprises a pseudouridine, a N6-methyladenonsine, a 5-methylcytidine, a 2- thiouridine, or any combination thereof. In some embodiments, the pseudouridine comprises N1 -methylpseudouridine.
In certain embodiments, the polynucleotide is codon-optimized for expression in a host cell (e.g., a human cell, or a CHO cell). Once a coding sequence is known or identified, codon optimization can be performed using known techniques and tools, e.g., using the GenScript® OptimumGene™ tool, or the like). Codon-optimized sequences include sequences that are partially codon-optimized (z.e., one or more codon is optimized for expression in the host cell) and those that are fully codon-optimized.
It will also be appreciated that polynucleotides encoding antibodies and antigen-binding fragments of the present disclosure may possess different nucleotide sequences while still
encoding a same antibody or antigen-binding fragment due to, for example, the degeneracy of the genetic code, splicing, and the like.
In any of the presently disclosed embodiments, the polynucleotide can comprise deoxyribonucleic acid (DNA) or ribonucleic acid (RNA). In some embodiments, the RNA comprises messenger RNA (mRNA), more specifically circRNA, taRNA, or saRNA. In some embodiments the polynucleotide may be optimized for expression in a host cell. In embodiments in which the polynucleotide comprises taRNA or saRNA, one or more sequences encoding a replication protein or peptide, one or more sequences encoding an antibody or antigen-binding fragment, or combinations thereof may be optimized for expression in human cells.
In certain embodiments, a polynucleotide comprises (i) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the VH polynucleotide sequence of an antibody as set forth in Table 3, and (ii) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the corresponding VL polynucleotide sequence of the antibody as set forth in Table 3. For example, in certain embodiments, a polynucleotide comprises (i) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to SEQ ID NO.:29, and (ii) a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to SEQ ID NO.:30.
In any of the presently disclosed embodiments, the polynucleotide can comprise deoxyribonucleic acid (DNA) or ribonucleic acid (RNA). In some embodiments, the RNA comprises messenger RNA (mRNA). In further embodiments, in which the polynucleotide is an saRNA, the first polynucleotide may further comprise a nucleic acid sequence encoding a replication protein or peptide. In other further embodiments in which the polynucleotide is a taRNA, a second polynucleotide is provided that comprises a nucleic acid sequence encoding a replication protein or peptide suitable to cause replication of the first polynucleotide.
In other more specific embodiments, at least two polynucleotides are provided, in which the first polynucleotide comprises or consists of a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the VH polynucleotide sequence of an antibody as set forth in Table 3 and the second polynucleotide comprises or consists of a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 98%, or at least 99% identity (or similarity) to the corresponding VL polynucleotide sequence of the antibody as set forth in Table 3. In further embodiments, in which the polynucleotides are mRNAs at least one and optionally both of the first and second polynucleotide is a saRNA, and further comprise a nucleic acid sequence encoding a replication protein or peptide. In other further embodiments in which the polynucleotides are taRNAs, a third polynucleotide is provided that comprises a nucleic acid sequence encoding a replication protein or peptide suitable to cause replication of the first polynucleotide and second polynucleotide. Vectors are also provided, wherein the vectors comprise or contain a polynucleotide as disclosed herein (e.g., a polynucleotide that encodes an antibody or antigen-binding fragment). A vector can comprise any one or more of the vectors disclosed herein. In particular embodiments, a vector is provided that comprises a DNA plasmid construct encoding the antibody or antigen-binding fragment, or a portion thereof (e.g., so-called “DMAb”; see, e.g., Muthumani et al., J Infect Dis. 2/-/(3):369-378 (2016); Muthumani etal., Hum Vaccin Immunother 9:2253-2262 (2013)); Flingai et al., Sci Rep. 5: 12616 (2015); and Elliott et al., NPJ Vaccines 18 (2017), which antibody-coding DNA constructs and related methods of use, including administration of the same, are incorporated herein by reference). In certain embodiments, a DNA plasmid construct comprises a single open reading frame encoding a heavy chain and a light chain (or a VH and a VL) of the antibody or antigen-binding fragment, wherein the sequence encoding the heavy chain and the sequence encoding the light chain are optionally separated by polynucleotide encoding a protease cleavage site and/or by a polynucleotide encoding a self-cleaving peptide. In some embodiments, the substituent components of the antibody or antigen-binding fragment are encoded by a polynucleotide comprised in a single plasmid. In other embodiments, the substituent components of the antibody or antigen-binding fragment are encoded by a polynucleotide comprised in two or more plasmids (e.g, a first plasmid comprises a polynucleotide encoding a heavy chain, VH, or
VH+CH1, and a second plasmid comprises a polynucleotide encoding the cognate light chain, or VL). In certain embodiments, a single plasmid comprises a polynucleotide encoding a heavy chain and/or a light chain from two or more antibodies or antigen-binding fragments of the present disclosure. An exemplary expression vector is pVaxl, available from Invitrogen®. A DNA plasmid of the present disclosure can be delivered to a subject by, for example, electroporation (e.g., intramuscular electroporation), or with an appropriate formulation (e.g., hyaluronidase).
In some embodiments, the vector may comprise or consist of circRNA, taRNA, or saRNA. In embodiments where the RNA therapeutic consists of a naked circRNA, taRNA, or saRNA vector, the cirRNA, taRNA, or saRNA may, in particular, contain modified nucleosides or other modifications disclosed herein to facilitate delivery to and expression in a human host cell in vivo.
Similarly, naked DNA vectors or other naked DNA therapeutics may contain modifications to facilitate delivery to and expression in a human host cell in vivo.
DNA therapeutics and RNA therapeutics, including circRNA therapeutics, taRNA therapeutics, or saRNA therapeutics, are also provided, wherein the DNA therapeutics or RNA therapeutics comprise or contain a polynucleotide as disclosed herein (e.g., a polynucleotide that encodes an antibody or antigen-binding fragment that binds to two or more bacterial antigens as described herein).
In a further aspect, the present disclosure also provides a host cell expressing an antibody or antigen-binding fragment according to the present disclosure; or comprising or containing a DNA therapeutic, RNA therapeutic, vector, or polynucleotide according the present disclosure.
In some embodiments, a method is provided that comprises administering to a subject a first polynucleotide (e.g., mRNA) encoding an antibody heavy chain, a VH, or a Fd (VH + CHI), and administering to the subject a second polynucleotide (e.g., mRNA) encoding the cognate antibody light chain, or VL.
In certain embodiments, a method is provided that comprises administering to a subject a first plasmid or vector that comprises a polynucleotide (e.g. mRNA) encoding a VH, a VH + CHI, or a heavy chain, and a second plasmid or vector is provided that comprises a polynucleotide (e.g. mRNA) encoding the cognate VL or light chain. The first plasmid or vector and/or the second plasmid or vector can be comprised in a composition that further comprises a pharmaceutically acceptable carrier, excipient, or diluent. In some embodiments, a polynucleotide (e.g., mRNA) is provided that encodes a heavy chain and a light chain of an
antibody or antigen-binding fragment thereof. In some embodiments, a polynucleotide (e.g., mRNA) is provided that encodes two heavy chains and two light chains of an antibody or antigen-binding fragment thereof. See, e.g. Li, JQ., Zhang, ZR., Zhang, HQ. et al. Intranasal delivery of replicating mRNA encoding neutralizing antibody against SARS-CoV-2 infection in mice. Sig Transduct Target Ther 6, 369 (2021). https://doi.org/10.1038/s41392-021-00783-l, the antibody-encoding mRNA constructs, vectors, and related techniques of which are incorporated herein by reference. In some embodiments, a polynucleotide is delivered to a subject via an alphavirus replicon particle (VRP) delivery system. In some embodiments, a replicon comprises a modified VEEV replicon comprising two subgenomic promoters. In some embodiments, a polynucleotide or replicon can translate simultaneously the heavy chain (or VH, or VH+1) and the light chain (or VL) of an antibody or antigen-binding fragment thereof. In some embodiments, a method is provided that comprises delivering to a subject such a polynucleotide or replicon.
In a further aspect, the present disclosure also provides a host cell expressing an antibody or antigen-binding fragment according to the present disclosure; or comprising or containing a vector or polynucleotide according the present disclosure.
Examples of such cells include but are not limited to, eukaryotic cells, e.g., yeast cells, animal cells, insect cells, plant cells; and prokaryotic cells, including A. coli. In some embodiments, the cells are mammalian cells, such as human B cells. In certain such embodiments, the cells are a mammalian cell line such as CHO cells (e.g., DHFR- CHO cells (Urlaub et al., PNAS 77:4216 (1980)), human embryonic kidney cells (e.g., HEK293T cells), PER.C6 cells, Y0 cells, Sp2/0 cells. NS0 cells, human liver cells, e.g. Hepa RG cells, myeloma cells or hybridoma cells. Other examples of mammalian host cell lines include mouse sertoli cells (e.g., TM4 cells); monkey kidney CV1 line transformed by SV40 (COS-7); baby hamster kidney cells (BHK); African green monkey kidney cells (VERO-76); monkey kidney cells (CV1); human cervical carcinoma cells (HELA); human lung cells (W138); human liver cells (Hep G2); canine kidney cells (MDCK; buffalo rat liver cells (BRL 3 A); mouse mammary tumor (MMT 060562); TRI cells; MRC 5 cells; and FS4 cells. Mammalian host cell lines suitable for antibody production also include those described in, for example, Yazaki and Wu, Methods in Molecular Biology, Vol. 248 (B. K. C. Lo, ed., Humana Press, Totowa, N.J.), pp. 255-268 (2003).
In certain embodiments, a host cell is a prokaryotic cell, such as an E. coli. The expression of peptides in prokaryotic cells such as A. coli is well established (see, e.g.,
Pluckthun, A. Bio/Technology 9:545-551 (1991). For example, antibodies may be produced in bacteria, in particular when glycosylation and Fc effector function are not needed. For expression of antibody fragments and polypeptides in bacteria, see, e.g., U.S. Pat. Nos. 5,648,237; 5,789,199; and 5,840,523.
In particular embodiments, the cell may be transfected with a vector, DNA therapeutic, or RNA therapeutic according to the present description with an expression vector. The term “transfection” refers to the introduction of nucleic acid molecules, such as DNA or RNA (e.g. mRNA) molecules, into cells, such as into eukaryotic cells. In the context of the present description, the term “transfection” encompasses any method known to the skilled person for introducing nucleic acid molecules into cells, such as into eukaryotic cells, including into mammalian cells. Such methods encompass, for example, electroporation, lipofection, e.g., based on cationic lipids and/or liposomes, calcium phosphate precipitation, nanoparticle based transfection, virus based transfection, or transfection based on cationic polymers, such as DEAE- dextran or polyethylenimine, etc. In certain embodiments, the introduction is non-viral.
Moreover, host cells of the present disclosure may be transfected stably or transiently with a vector, DNA therapeutic, or RNA therapeutic according to the present disclosure, e.g. for expressing an antibody, or an antigen-binding fragment thereof, according to the present disclosure. In such embodiments, the cells may be stably transfected with the vector or DNA therapeutic as described herein. Alternatively, cells may be transiently transfected with a vector, DNA therapeutic, or RNA therapeutic according to the present disclosure encoding an antibody or antigen-binding fragment as disclosed herein. In any of the presently disclosed embodiments, a polynucleotide may be heterologous to the host cell.
Accordingly, the present disclosure also provides recombinant host cells that heterologously express an antibody or antigen-binding fragment of the present disclosure. For example, the cell may be of a species that is different to the species from which the antibody was fully or partially obtained (e.g., CHO cells expressing a human antibody or an engineered human antibody). In some embodiments, the cell type of the host cell does not express the antibody or antigen-binding fragment in nature. Moreover, the host cell may impart a post-translational modification (PTM; e.g., glycosylation or fucosylation), or a lack thereof, on the antibody or antigen-binding fragment that is not present in a native state of the antibody or antigen-binding fragment (or in a native state of a parent antibody from which the antibody or antigen binding fragment was engineered or derived). Such a PTM, or a lack thereof, may result in a functional difference (e.g., reduced immunogenicity). Accordingly, an antibody or antigen-binding
fragment of the present disclosure that is produced by a host cell as disclosed herein may include one or more post-translational modification that is distinct from the antibody (or parent antibody) in its native state (e.g., a human antibody produced by a host cell can comprise one or more post- translational modification, or can include fewer post-translational modification(s), such that it is distinct from the antibody when isolated from the human and/or produced by the native human B cell or plasma cell).
Insect cells useful expressing a binding protein of the present disclosure are known in the art and include, for example, Spodoptera frugipera Sf9 cells, Trichoplusia ni BTI-TN5B1-4 cells, and Spodoptera frugipera SfSWTOl “Mimic™” cells. See, e.g., Palmberger et al., J. BiotechnoL 753(3-4): 160-166 (2011). Numerous baculoviral strains have been identified which may be used in conjunction with insect cells, particularly for transfection of Spodoptera frugiperda cells.
Eukaryotic microbes such as filamentous fungi or yeast are also suitable hosts for cloning or expressing protein-encoding vectors, DNA therapeutics, or RNA therapeutics and include fungi and yeast strains with “humanized” glycosylation pathways, resulting in the production of an antibody with a partially or fully human glycosylation pattern. See Gerngross, Nat.
Biotech. 22:1409-1414 (2004); Li et al., Nat. Biotech. 24:210-215 (2006).
Plant cells can also be utilized as hosts for expressing an antibody or antigen-binding fragment of the present disclosure. For example, PLANTIBODIES™ technology (described in, for example, U.S. Pat. Nos. 5,959,177; 6,040,498; 6,420,548; 7,125,978; and 6,417,429) employs transgenic plants to produce antibodies.
In certain embodiments, the host cell comprises a mammalian cell. In particular embodiments, the host cell is a CHO cell, a HEK293 cell, a PER.C6 cell, a Y0 cell, a Sp2/0 cell, a NS0 cell, a human liver cell, a myeloma cell, or a hybridoma cell.
In some embodiments relating to an RNA therapeutic, particularly a circRNA therapeutic, taRNA therapeutic, or saRNA therapeutic, or a DNA therapeutic, the production host cell may be any host cell described above or otherwise disclosed herein. The in vivo human host cell may be any human cell that is a host cell as described above or otherwise disclosed herein.
In a related aspect, the present disclosure provides methods for producing an antibody, or antigen-binding fragment, wherein the methods comprise culturing a host cell of the present disclosure under conditions and for a time sufficient to produce the antibody, or the antigenbinding fragment. Methods useful for isolating and purifying recombinantly produced
antibodies, by way of example, may include obtaining supernatants from suitable host cell/vector systems that secrete the recombinant antibody into culture media and then concentrating the media using a commercially available filter. Following concentration, the concentrate may be applied to a single suitable purification matrix or to a series of suitable matrices, such as an affinity matrix or an ion exchange resin. One or more reverse phase HPLC steps may be employed to further purify a recombinant polypeptide. These purification methods may also be employed when isolating an immunogen from its natural environment. Methods for large scale production of one or more of the isolated/recombinant antibody described herein include batch cell culture, which is monitored and controlled to maintain appropriate culture conditions. Purification of soluble antibodies may be performed according to methods described herein and known in the art and that comport with laws and guidelines of domestic and foreign regulatory agencies.
Compositions
Also provided herein are compositions that comprise any one or more of the presently disclosed antibodies, antigen-binding fragments, polypeptides, polynucleotides, vectors, DNA therapeutics, RNA therapeutics) (including mRNA therapeutics, circ RNA therapeutics, taRNA therapeutics, and saRNA therapeutics), or host cells, singly or in any combination, and can further comprise a pharmaceutically acceptable carrier, excipient, or diluent. Such compositions, as well as carriers, excipients, and diluents, are discussed in further detail herein.
In certain embodiments, a composition comprises a first vector comprising a first plasmid, and a second vector comprising a second plasmid, wherein the first plasmid comprises a polynucleotide encoding a heavy chain, VH, or VH+CH1, and a second plasmid comprises a polynucleotide encoding the cognate light chain or VL of the antibody or antigen-binding fragment thereof. In certain embodiments, a composition comprises a polynucleotide (e.g., mRNA) coupled to a suitable delivery vehicle or carrier. In certain embodiments, a composition comprises a first polynucleotide (e.g., mRNA) encoding an antibody heavy chain, a VH, or a Fd (VH + CHI), and a second polynucleotide (e.g., mRNA) encoding the cognate antibody light chain or VL.
Exemplary vehicles or carriers for administration to a human subject include a lipid or lipid-derived delivery vehicle, such as a liposome, solid lipid nanoparticle, oily suspension, submicron lipid emulsion, lipid microbubble, inverse lipid micelle, cochlear liposome, lipid microtubule, lipid microcylinder, or lipid nanoparticle (LNP) or a nanoscale platform see, e.g.,
Li et al. Wilery Interdiscip Rev. Nanomed Nanobiotechnol. 77(2):el530 (2019)). Principles, reagents, and techniques for designing appropriate mRNA and formulating mRNA-LNP and delivering the same are described in, for example, Pardi et al. (J Control Release 277345-351 (2015)); Thess et al. Mol Ther 23: 1456-1464 (2015)); Thran et al. (EMBO Mol Med 9(10): 1434-1448 (2017); Kose et al. (Set. Immunol. 4 eaaw6647 (2019); and Sabnis et al. Mol. Ther. 26: 1509-1519 (2018)), which techniques, include capping, codon optimization, nucleoside modification, purification of mRNA, incorporation of the mRNA into stable lipid nanoparticles (e.g., ionizable cationic lipid/phosphatidylcholine/cholesterol/PEG-lipid; ionizable lipid:distearoyl PC:cholesterol:polyethylene glycol lipid), and subcutaneous, intramuscular, intradermal, intravenous, intraperitoneal, and intratracheal administration of the same, are incorporated herein by reference.
In certain embodiments, a composition comprises a first antibody or antigen-binding fragment of the present disclosure and a second antibody or antigen-binding fragment of the present disclosure, wherein of the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are different. In some embodiments, the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment bind to the following, respectively: an LTA and a Gmd; an LTA and a PFT (e.g., an alpha toxin, a gamma toxin, or a leukotoxin); an LTA and a SpA; an LTA and a ClfA; a Gmd and a PFT; a Gmd and a SpA; a Gmd and a ClfA; a PFT and a SpA; a PFT and a ClfA; a SpA and a ClfA; a Sbi and an LTA; a Sbi and a Gmd; a Sbi and a PFT; a Sbi and a SpA; or a Sbi and a ClfA. In other embodiments, the first antibody or antigen-binding fragment and the second antibody or antigenbinding fragment are different and each bind to: an LTA; a PFT; a Gmd; a SpA; a Sbi; or a ClfA. In certain embodiments, the first antibody or antigen-binding domain comprises the six CDR sequences, and optionally the VH and VL, of a first anti-LTA antibody and the second antibody or antigen-binding domain comprises the six CDR sequences, and optionally the VH and VL, of a second anti-LTA antibody. In some embodiments, the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are selected from, or are derived from, a first and a second antibody disclosed in Table 1.
In some embodiments, a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to an opsonizing target and a second antibody or antigen-binding fragment thereof binds to a neutralizing target.
In some embodiments, a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to a target selected from LTA, ClfA, Protein A, and Gmd and a second antibody or antigen-binding fragment thereof binds to an alpha toxin.
In some embodiments, a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to a target selected from LTA, ClfA, Protein A, and Gmd and a second antibody or antigen-binding fragment thereof binds to a neutralizing target.
In some embodiments, a first antibody or antigen-binding fragment thereof comprises a first antibody which binds to an opsonizing target and a second antibody or antigen-binding fragment thereof binds to an alpha toxin.
In some embodiments, a combination or composition comprises a first antibody or antigen-binding fragment thereof and a second antigen-binding fragment thereof, wherein the first antibody or antigen-binding fragment thereof or the second antibody or antigen-binding fragment thereof comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC10; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF10; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE150; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
In some embodiments, a combination or composition comprises a first antibody or antigen-binding fragment thereof and a second antigen-binding fragment thereof, wherein the first antibody or antigen-binding fragment thereof and the second antibody or antigen-binding fragment thereof comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively. In certain embodiments, the first antibody or antigen-binding
fragment and/or the second antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S/H435R/Y436F mutations.
In certain embodiments, a composition comprises polynucleotide, a vector, a RNA therapeutic, or an DNA therapeutic.
In some embodiments, the composition comprises a single polynucleotide encoding i) a heavy chain, VH, or VH+CH; and ii) a cognate light chain, VL, or VL+CL, of the antibody or antigen-binding fragment. In some such embodiments, the polynucleotide is an saRNA and further encodes a replication protein or peptide. In some embodiments, the polynucleotide is a circRNA.
In some embodiments, the composition comprises a first taRNA encoding i) a heavy chain, VH, or VH+CH; and ii) a cognate light chain, VL, or VL+CL, of the antibody or antigenbinding fragment, and a second taRNA that encodes a replication protein or peptide. In some embodiments, at least one, or both the first and second taRNA is a circRNA.
In some embodiments, the composition comprises a first polynucleotide encoding a heavy chain, VH, or VH+CH, and a second polynucleotide encoding a cognate light chain, VL, or VL+CL, of the antibody or antigen-binding fragment. In some such embodiments in which the polynucleotides are mRNAs, the first polynucleotide, the second polynucleotide, or both are saRNAs or taRNAs an further encode a replication protein or peptide. In some embodiments, at least one, at least two, or all of the polynucleotides is a circRNA.
In some embodiments, the composition comprises a first mRNA encoding a heavy chain, VH, or VH+CH, and a second mRNA encoding a cognate light chain, VL, or VL+CL, of the antibody or antigen-binding fragment. In some such embodiments in which the polynucleotides are mRNAs, the composition is a taRNA composition that comprises a third taRNA that encodes a replication protein or peptide. In some embodiments, at least one, at least two, or all of the polynucleotides is a circRNA.
Methods and Uses
Also provided herein are methods for use of an antibody or antigen-binding fragment, nucleic acid, vector, cell, or composition of the present disclosure in the diagnosis of an infection antigens (e.g., in a human subject, or in a sample obtained from a human subject) by a bacteria expressing one or more herein disclosed antigens (lipoteichoic acid (LTA), e.g., a Type I LTA, a Type II LTA, a Type IV LTA, or any combination thereof; a glucosaminidase (Gmd); Pore Forming Toxin (PFT) such as a leukocidin (e.g., LukED, a LukSF, or both), an alpha toxin (e.g.,
Hla), or a gamma toxin or gamma hemolysin (e.g., HIgAB); a Staphylococcal Protein A (SpA); a clumping factor A (ClfA); Sbi; or any combination thereof). In some embodiments, an infection comprises infection by bacteria of any or more of: genus Staphylococcus,' genus Enterococcus,' genus Streptococcus,' and genus Lactococcus. In some embodiments, an infection comprises infection by: S. aureus (e.g., MRS A); Coagulase-negative staphylococci (CoNS); S. epidermidis,' S. pneumoniae,' S. lugdunensis,' or S. hominis,' S. aureus FPR3757; S. az//vz/.s NE284 TE2 mutant; S. epidermidis RP62A; S. epidermidis 1200; S. epidermidis NIH04008; S. pyogenes,' E.faecalis,' S. agalactiae, S. hominis,' L. garvieae,' S. pneumoniae,' E. coli, or any combination of thereof. ). In some embodiments, an infection comprises infection by a gram-positive bacteria.
Methods of diagnosis (e.g., in vitro, ex vivo) may include contacting an antibody, antibody fragment (e.g., antigen binding fragment) with a sample. Such samples may be isolated from a subject, for example an isolated tissue sample taken from, for example, nasal passages, sinus cavities, salivary glands, lung, liver, pancreas, kidney, ear, eye, placenta, alimentary tract, heart, ovaries, pituitary, adrenals, thyroid, brain, skin, or blood. The methods of diagnosis may also include the detection of an antigen/antibody complex, in particular following the contacting of an antibody or antibody fragment with a sample. Such a detection step can be performed at the bench, i.e. without any contact to the human or animal body. Examples of detection methods are well-known to the person skilled in the art and include, e.g., ELISA (enzyme-linked immunosorbent assay), including direct, indirect, and sandwich ELISA.
Also provided herein are methods of treating a subject using an antibody or antigenbinding fragment of the present disclosure, or a composition comprising the same, wherein the subject has, is believed to have, or is at risk for having an infection by a bacteria expressing one or more herein-disclosed antigens. “Treat,” “treatment,” or “ameliorate” refers to medical management of a disease, disorder, or condition of a subject e.g., a human or non-human mammal, such as a primate, horse, cat, dog, goat, mouse, or rat). In general, an appropriate dose or treatment regimen comprising an antibody or composition of the present disclosure is administered in an amount sufficient to elicit a therapeutic or prophylactic benefit. Therapeutic or prophylactic/preventive benefit includes improved clinical outcome; lessening or alleviation of symptoms associated with a disease; decreased occurrence of symptoms; improved quality of life; longer disease-free status; diminishment of extent of disease, stabilization of disease state; delay or prevention of disease progression; remission; survival; prolonged survival; or any combination thereof. In certain embodiments, therapeutic or prophylactic/preventive benefit includes reduction or prevention of hospitalization for treatment of a bacterial infection i.e., in a
statistically significant manner). In certain embodiments, therapeutic or prophylactic/preventive benefit includes a reduced duration of hospitalization for treatment of a bacterial infection (z.e., in a statistically significant manner). In certain embodiments, therapeutic or prophylactic/preventive benefit includes a reduced or abrogated need for respiratory intervention, such as intubation and/or the use of a respirator device. In certain embodiments, therapeutic or prophylactic/preventive benefit includes reduction inflammation, reduction in cell death, reduction or reversal of sepsis, reduced or abrogated need for antibiotics, or any combination thereof. In certain embodiments, therapeutic or prophylactic/preventive benefit includes reversing a late-stage disease pathology and/or reducing mortality.
In some embodiments, an infection comprises one or more of the following indications or settings: bacteremia; blood stream infection (BSI); skin and soft tissue infection (SSTI); wound infection; prosthetic joint infection (PJI); surgical implant; surgical site infection (SSI); hemodialysis associated/line associated bacteremia; neutropenia; ventilator associated pneumonia (VAP); toxic shock syndrome; menstrual toxic shock syndrome; peritonitis; pneumonia; infective endocarditis; sepsis; osteomyelitis; lower respiratory infections (LRI) and any related infection of the thorax; diarrhea; urinary tract infection (UTI); pyelonephritis; bacterial infections of the skin and/or subcutaneous system; typhoid; paratyphoid; invasive non- typhoidal salmonella disease; meningitis and other bacterial central nervous system (CNS) infections; cardiac infections; infections of bones, joints, related organs; gonorrhea; and chlamydia. In some embodiments, an infection is in a subject who receives dialysis. In some embodiments, an infection is in a subject who has recurrent bacteremia and/or recurrent BSI. In some embodiments, an infection is in a subject who has undergone, is undergoing, or will undergo surgery. In some embodiments, an infection is in a subject who used a ventilator, is using a ventilator, or will use a ventilator. In some embodiments, an infection is in a subject who has received, is receiving, or will receive, a prosthetic joint or other joint replacement (e.g., hip, knee, and/or shoulder). In some embodiments, an infection is in a subject who is carrying a wound (e.g., abrasion, cut, projectile) or who has received treatment for a wound. In some embodiments, an infection is in a subject who has undergone, is undergoing, or will undergo hemodialysis. In some embodiments, an infection is in a subject who has undergone, is undergoing, or will undergo placement of a line.
In certain embodiments, for a single dose, e.g. a daily, weekly or monthly dose, the amount of the antibody or antigen-binding fragment in a composition or method according to the present disclosure, may not exceed 1 g or 500 mg. In some embodiments, for a single dose, the
amount of the antibody or antigen-binding fragment in a composition or method according to the present disclosure, may not exceed 200 mg, or 100 mg. For example, in some embodiments, for a single dose, the amount of the antibody or antigen-binding fragment in a composition or method according to the present disclosure, may not exceed 50 mg.
A “therapeutically effective amount” or “effective amount” of an antibody, antigenbinding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition of this disclosure refers to an amount of the composition or molecule sufficient to result in a therapeutic effect, including improved clinical outcome; lessening or alleviation of symptoms associated with a disease; decreased occurrence of symptoms; improved quality of life; longer disease-free status; diminishment of extent of disease, stabilization of disease state; delay of disease progression; remission; survival; or prolonged survival in a statistically significant manner. When referring to an individual active ingredient, administered alone, a therapeutically effective amount refers to the effects of that ingredient or cell expressing that ingredient alone. When referring to a combination, a therapeutically effective amount refers to the combined amounts of active ingredients or combined adjunctive active ingredient with a cell expressing an active ingredient that results in a therapeutic effect, whether administered serially, sequentially, or simultaneously.
Accordingly, in certain embodiments, methods are provided for treating a bacterial infection in a subject, wherein the methods comprise administering to the subject an effective amount of an antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition as disclosed herein.
Subjects that can be treated by the present disclosure are, in general, human and other primate subjects, such as monkeys and apes for veterinary medicine purposes. Other model organisms, such as mice and rats, may also be treated according to the present disclosure. In any of the aforementioned embodiments, the subject may be a human subject. The subjects can be male or female and can be any suitable age, including infant, juvenile, adolescent, adult, and geriatric subjects.
A number of criteria are believed to contribute to high risk for severe symptoms or death associated with a bacterial infection. These include, but are not limited to, age, occupation, general health, pre-existing health conditions, locale, and lifestyle habits. Risk factors can also include: dialysis; use of a ventilator; bacteremia; current, previous, or recurrent blood stream infection; current, previous, or recurrent skin and soft tissue infection; receipt of surgery; receipt of a prosthetic joint or other implant or joint replacement; or any combination of these. In some
embodiments, a subject treated according to the present disclosure comprises one or more risk factors.
In certain embodiments, a human subject treated according to the present disclosure is an infant, a child, a young adult, an adult of middle age, or an elderly person. In certain embodiments, a human subject treated according to the present disclosure is less than 1 year old, or is 1 to 5 years old, or is between 5 and 125 years old (e.g., 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 105, 110, 115, or 125 years old, including any and all ages therein or therebetween). In certain embodiments, a human subject treated according to the present disclosure is 0-19 years old, 20-44 years old, 45-54 years old, 55-64 years old, 65-74 years old, 75-84 years old, or 85 years old, or older. Persons of middle, and especially of elderly age are can be at particular risk. In particular embodiments, the human subject is 45-54 years old, 55-64 years old, 65-74 years old, 75-84 years old, or 85 years old, or older. In some embodiments, the human subject is male. In some embodiments, the human subject is female.
In certain embodiments, a subject treated according to the present disclosure has received a vaccine for a bacterial infection and the vaccine is determined to be ineffective, e.g., by postvaccine infection or symptoms in the subject, by clinical diagnosis or scientific or regulatory consensus.
Prophylaxis of a bacterial infection refers in particular to prophylactic settings, wherein the subject was not diagnosed with infection by the bacteria (either no diagnosis was performed or diagnosis results were negative) and/or the subject does not show or experience symptoms of bacterial infection. Prophylaxis of bacterial infection is particularly useful in subjects at greater risk of severe disease or complications when infected, such as pregnant women, children (such as children under 59 months), the elderly, subjects with chronic medical conditions (such as chronic cardiac, pulmonary, renal, metabolic, neurodevelopmental, liver or hematologic diseases) and individuals with immunosuppressive conditions (such as HIV/AIDS, receiving chemotherapy or steroids, or malignancy), subjects using a ventilator, subjects undergoing or having recently undergone surgery, subjects with a blood stream infection or history of blood stream infections, subjects receiving or having received a prosthetic joint or other implant, subjects undergoing or having undergone dialysis, subjects, subjects with bacteremia or a history of bacteremia, subjects having wound, or the like. Moreover, prophylaxis of bacterial infection is also particularly useful in subjects at greater risk acquiring bacterial infection, e.g., due to increased exposure, for example subjects working or staying in unsanitary areas.
In certain embodiments, treatment is administered as peri-exposure or pre-exposure prophylaxis. In certain embodiments, treatment is administered as post-exposure prophylaxis.
In therapeutic settings, in contrast, the subject has a bacterial infection, is diagnosed with a bacterial infection, and/or shows symptoms of a bacterial infection. Of note, the terms “treatment” and “therapy”/” therapeutic” of bacterial infection can refer to (complete) cure as well as attenuation/reduction of bacterial infection and/or related symptoms (e.g., attenuation/reduction of severity of infection and/or symptoms, number of symptoms, duration of infection and/or symptoms, or any combination thereof).
It will be understood that reference herein to a reduced number and/or severity of symptoms, which reduction results from administration of a presently disclosed pharmaceutical composition, describes a comparison with a reference subject who did not receive a disclosed pharmaceutical composition. A reference subject can be, for example, (i) the same subject during an earlier period of time, (ii) a subject of a same or a similar: age or age group; gender; pregnancy status; chronic medical condition (such as chronic cardiac, pulmonary, renal, metabolic, neurodevelopmental, liver or hematologic diseases) or lack thereof; and/or immunosuppressive condition or lack thereof; or (iii) a typical subject within a population (e.g., local, regional, or national, including of a same or similar age or age range and/or general state of health) during a period of time. Prophylaxis can be determined by, for example, the failure to develop a diagnosed bacterial infection and/or the lack of symptoms associated with a bacterial infection over a period of time.
In some embodiments, treatment and/or prevention comprises post-exposure prophylaxis.
In some embodiments, the subject has received, is receiving, or will receive an antibiotic agent. In some embodiments, the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti- infectives, or any combination thereof. In some embodiments, the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin- clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof. In some embodiments, a subject has received, is receiving, or will receive, wound drainage, removal of a medical device, or both.
Typical routes of administering the presently disclosed compositions include, without limitation, oral, topical, transdermal, inhalation, parenteral, sublingual, buccal, rectal, vaginal,
and intranasal. The term “parenteral”, as used herein, includes subcutaneous injections, intravenous, intramuscular, intrasternal injection or infusion techniques. In certain embodiments, administering comprises administering by a route that is selected from oral, intravenous, parenteral, intragastric, intrapleural, intrapulmonary, intrarectal, intradermal, intraperitoneal, intratumoral, subcutaneous, topical, transdermal, intracisternal, intrathecal, intranasal, and intramuscular. In particular embodiments, a method comprises orally administering the antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
Pharmaceutical compositions according to certain embodiments of the present invention are formulated so as to allow the active ingredients contained therein to be bioavailable upon administration of the composition to a patient. Compositions that will be administered to a subject or patient may take the form of one or more dosage units, where for example, a tablet may be a single dosage unit, and a container of a herein described an antibody or antigen-binding in aerosol form may hold a plurality of dosage units. Actual methods of preparing such dosage forms are known, or will be apparent, to those skilled in this art; for example, see Remington: The Science and Practice of Pharmacy, 20th Edition (Philadelphia College of Pharmacy and Science, 2000). The composition to be administered will, in any event, contain an effective amount of an antibody or antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition of the present disclosure, for treatment of a disease or condition of interest in accordance with teachings herein.
A composition may be in the form of a solid or liquid. In some embodiments, the carrier(s) are particulate, so that the compositions are, for example, in tablet or powder form. The carrier(s) may be liquid, with the compositions being, for example, an oral oil, injectable liquid or an aerosol, which is useful in, for example, inhalatory administration. When intended for oral administration, the pharmaceutical composition is preferably in either solid or liquid form, where semi solid, semi liquid, suspension and gel forms are included within the forms considered herein as either solid or liquid.
As a solid composition for oral administration, the pharmaceutical composition may be formulated into a powder, granule, compressed tablet, pill, capsule, chewing gum, wafer or the like. Such a solid composition will typically contain one or more inert diluents or edible carriers. In addition, one or more of the following may be present: binders such as carboxymethylcellulose, ethyl cellulose, microcrystalline cellulose, gum tragacanth or gelatin; excipients such as starch, lactose or dextrins, disintegrating agents such as alginic acid, sodium
alginate, Primogel, com starch and the like; lubricants such as magnesium stearate or Sterotex; glidants such as colloidal silicon dioxide; sweetening agents such as sucrose or saccharin; a flavoring agent such as peppermint, methyl salicylate or orange flavoring; and a coloring agent. When the composition is in the form of a capsule, for example, a gelatin capsule, it may contain, in addition to materials of the above type, a liquid carrier such as polyethylene glycol or oil.
The composition may be in the form of a liquid, for example, an elixir, syrup, solution, emulsion or suspension. The liquid may be for oral administration or for delivery by injection, as two examples. When intended for oral administration, preferred compositions contain, in addition to the present compounds, one or more of a sweetening agent, preservatives, dye/colorant and flavor enhancer. In a composition intended to be administered by injection, one or more of a surfactant, preservative, wetting agent, dispersing agent, suspending agent, buffer, stabilizer and isotonic agent may be included.
Liquid pharmaceutical compositions, whether they be solutions, suspensions or other like form, may include one or more of the following adjuvants: sterile diluents such as water for injection, saline solution, preferably physiological saline, Ringer’s solution, isotonic sodium chloride, fixed oils such as synthetic mono or diglycerides which may serve as the solvent or suspending medium, polyethylene glycols, glycerin, propylene glycol or other solvents; antibacterial agents such as benzyl alcohol or methyl paraben; antioxidants such as ascorbic acid or sodium bisulfite; chelating agents such as ethylenediaminetetraacetic acid; buffers such as acetates, citrates or phosphates and agents for the adjustment of tonicity such as sodium chloride or dextrose. The parenteral preparation can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic. Physiological saline is a preferred adjuvant. An injectable pharmaceutical composition is preferably sterile.
A liquid composition intended for either parenteral or oral administration should contain an amount of an antibody or antigen-binding fragment as herein disclosed such that a suitable dosage will be obtained. Typically, this amount is at least 0.01% of the antibody or antigenbinding fragment in the composition. When intended for oral administration, this amount may be varied to be between 0.1 and about 70% of the weight of the composition. Certain oral pharmaceutical compositions contain between about 4% and about 75% of the antibody or antigen-binding fragment. In certain embodiments, pharmaceutical compositions and preparations according to the present invention are prepared so that a parenteral dosage unit contains between 0.01 to 10% by weight of antibody or antigen-binding fragment prior to dilution.
The composition may be intended for topical administration, in which case the carrier may suitably comprise a solution, emulsion, ointment or gel base. The base, for example, may comprise one or more of the following: petrolatum, lanolin, polyethylene glycols, bee wax, mineral oil, diluents such as water and alcohol, and emulsifiers and stabilizers. Thickening agents may be present in a composition for topical administration. If intended for transdermal administration, the composition may include a transdermal patch or iontophoresis device. The pharmaceutical composition may be intended for rectal administration, in the form, for example, of a suppository, which will melt in the rectum and release the drug. The composition for rectal administration may contain an oleaginous base as a suitable nonirritating excipient. Such bases include, without limitation, lanolin, cocoa butter and polyethylene glycol.
A composition may include various materials which modify the physical form of a solid or liquid dosage unit. For example, the composition may include materials that form a coating shell around the active ingredients. The materials that form the coating shell are typically inert, and may be selected from, for example, sugar, shellac, and other enteric coating agents. Alternatively, the active ingredients may be encased in a gelatin capsule. The composition in solid or liquid form may include an agent that binds to the antibody or antigen-binding fragment of the disclosure and thereby assists in the delivery of the compound. Suitable agents that may act in this capacity include monoclonal or polyclonal antibodies, one or more proteins or a liposome. The composition may consist essentially of dosage units that can be administered as an aerosol. The term aerosol is used to denote a variety of systems ranging from those of colloidal nature to systems consisting of pressurized packages. Delivery may be by a liquefied or compressed gas or by a suitable pump system that dispenses the active ingredients. Aerosols may be delivered in single phase, bi phasic, or tri phasic systems in order to deliver the active ingredient(s). Delivery of the aerosol includes the necessary container, activators, valves, subcontainers, and the like, which together may form a kit. One of ordinary skill in the art, without undue experimentation, may determine preferred aerosols.
It will be understood that compositions of the present disclosure also encompass carrier molecules for polynucleotides, as described herein (e.g., lipid nanoparticles, nanoscale delivery platforms, and the like).
The pharmaceutical compositions may be prepared by methodology well known in the pharmaceutical art. For example, a composition intended to be administered by injection can be prepared by combining a composition that comprises an antibody, antigen-binding fragment thereof, or antibody conjugate as described herein and optionally, one or more of salts, buffers
and/or stabilizers, with sterile, distilled water so as to form a solution. A surfactant may be added to facilitate the formation of a homogeneous solution or suspension. Surfactants are compounds that non-covalently interact with the peptide composition so as to facilitate dissolution or homogeneous suspension of the antibody or antigen-binding fragment thereof in the aqueous delivery system.
In general, an appropriate dose and treatment regimen provide the composition(s) in an amount sufficient to provide therapeutic and/or prophylactic benefit (such as described herein, including an improved clinical outcome (e.g., a decrease in frequency, duration, or severity of diarrhea or associated dehydration, or inflammation, or longer disease-free and/or overall survival, or a lessening of symptom severity). For prophylactic use, a dose should be sufficient to prevent, delay the onset of, or diminish the severity of a disease associated with disease or disorder. Prophylactic benefit of the compositions administered according to the methods described herein can be determined by performing pre-clinical (including in vitro and in vivo animal studies) and clinical studies and analyzing data obtained therefrom by appropriate statistical, biological, and clinical methods and techniques, all of which can readily be practiced by a person skilled in the art.
Compositions are administered in an effective amount (e.g., to treat a bacterial (e.g., Staphylococcus infection), which will vary depending upon a variety of factors including the activity of the specific compound employed; the metabolic stability and length of action of the compound; the age, body weight, general health, sex, and diet of the subject; the mode and time of administration; the rate of excretion; the drug combination; the severity of the particular disorder or condition; and the subject undergoing therapy. In certain embodiments, following administration of therapies according to the formulations and methods of this disclosure, test subjects will exhibit about a 10% up to about a 99% reduction in one or more symptoms associated with the disease or disorder being treated as compared to placebo-treated or other suitable control subjects.
Generally, a therapeutically effective dose of an antibody or antigen binding fragment is (for a 70 kg mammal) from about 0.001 mg/kg (z.e., 0.07 mg) to about 100 mg/kg (i.e., 7.0 g); preferably a therapeutically effective dose is (for a 70 kg mammal) from about 0.01 mg/kg (i.e., 0.7 mg) to about 50 mg/kg (i.e., 3.5 g); more preferably a therapeutically effective dose is (for a 70 kg mammal) from about 1 mg/kg (i.e., 70 mg) to about 25 mg/kg (i.e., 1.75 g). For polynucleotides, vectors, DNA therapeutics, RNA therapeutics, host cells, and related compositions of the present disclosure, a therapeutically effective dose may be different than for
an antibody or antigen-binding fragment. In such cases, the dose may be sufficient to cause endogenous production of the antibody or antigen-binding fragment in a human patient that results, within a specified period of time, in serum concentrations equivalent to that of a therapeutically effective daily dose of the antibody or antigen-binding fragment. In some embodiments, the specified period of time may be 1, 2, 5, 7, 14, or 30 days from the time of administration.
In some embodiments, the serum concentration equivalent to that of a therapeutically effective daily dose of the antibody or antigen-binding fragment may be maintained for at least 2 weeks, 1 month, 2 months, 3 months, or 6 months, or for at least a time between 2 weeks and 6 months, 2 weeks and 3 months, 2 weeks and 2 months, 2 weeks and 1 month, 1 and 6 months, 1 and 3 months, 1 and 2 months, 2 and 6 months, 2 and 3 months, or 3 and 6 months from the time of administration.
In some embodiments, the polynucleotide may not integrate into the human host cell genome or persist in the human host, such that the antibody or antigen-binding fragment may be undetectable in serum by at least 1, 2, 3, 6, or 12 months from the time of administration.
In some embodiments in which administration is repeated, the time of administration may be the time of first administration.
In some embodiments, particularly those in which RNA therapeutics are used, serum concentrations may typically exhibit an initial increase, followed by a time of steady concentration, then a decrease.
The period of time to reach a therapeutically effective daily dose of the antibody or antigen-binding fragment in the serum, the period of time for which such dose is maintained, and the period of time required for the antibody or antigen-binding fragment to become undetectable in serum, and the doses required to achieve these results are influenced by the nature of the polynucleotide delivered. For example, in some embodiments, taRNA therapeutics or saRNA therapeutics may achieve results similar to those of non-self-amplifying mRNAs at much smaller doses because the RNA encoding the antibody or antigen-binding fragment is amplified in the in vivo human host cell.
In certain embodiments, a method comprises administering the antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject 2, 3, 4, 5, 6, 7, 8, 9, 10 times, or more. In certain embodiments, a method comprises administering the antibody, antigen-binding fragment, polynucleotide, vector, DNA
therapeutic, RNA therapeutic, host cell, or composition to the subject at least 2, 3, 4, 5, 6, 7, 8, 9, 10 times, or more.
In certain embodiments, a method comprises administering the antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, or composition to the subject a plurality of times, wherein a second or successive administration is performed at about 6, about 7, about 8, about 9, about 10, about 11, about 12, about 24, about 48, about 74, about 96 hours, or more, following a first or prior administration, respectively.
In certain embodiments, a method comprises administering the antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition at least one time prior to the subject being infected by the bacteria.
Compositions comprising an antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition of the present disclosure may also be administered simultaneously with, prior to, or after administration of one or more other therapeutic agents, such as, for example, one or more antibiotic agent, such as described herein. Such combination therapy may include administration of a single pharmaceutical dosage formulation which contains a compound of the invention and one or more additional active agents, as well as administration of compositions comprising an antibody or antigen-binding fragment of the disclosure and each active agent in its own separate dosage formulation. For example, an antibody or antigen-binding fragment thereof as described herein and the other active agent can be administered to the patient together in a single oral dosage composition such as a tablet or capsule, or each agent administered in separate oral dosage formulations.
Similarly, an antibody or antigen-binding fragment as described herein and the other active agent can be administered to the subject together in a single parenteral dosage composition such as in a saline solution or other physiologically acceptable solution, or each agent administered in separate parenteral dosage formulations. Where separate dosage formulations are used, the compositions comprising an antibody or antigen-binding fragment and one or more additional active agents can be administered at essentially the same time, z.e., concurrently, or at separately staggered times, z.e., sequentially and in any order; combination therapy is understood to include all these regimens.
In some embodiments, an antibody (or one or more nucleic acid, host cell, DNA therapeutic, RNA therapeutic, vector, or composition) is administered to a subject who has previously received one or more anti-inflammatory agent and/or one or more antibiotic agent. In certain embodiments, a combination therapy is provided that comprises one or more antibody or
antigen-binding fragment (or one or more nucleic acid, host cell, vector, DNA therapeutic, RNA therapeutic, or composition) of the present disclosure and one or more anti-inflammatory agent and/or one or more antibiotic agent. In some embodiments, the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof. In some embodiments, the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin-clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof. In particular embodiments, the one or more anti-inflammatory agent comprises a corticosteroid such as, for example, dexamethasone, prednisone, or the like. In some embodiments, the one or more anti-inflammatory agents comprise a cytokine antagonist such as, for example, an antibody that binds to IL6 (such as siltuximab), or to IL-6R (such as tocilizumab), or to IL-ip, IL-7, IL-8, IL-9, IL-10, FGF, G-CSF, GM-CSF, IFN-y, IP-10, MCP-1, MIP-1A, MIP1-B, PDGR, TNF-a, or VEGF. In some embodiments, anti-inflammatory agents such as leronlimab, ruxolitinib and/or anakinra are used. Other anti-inflammatory agents for use in a combination therapy of the present disclosure include non-steroidal anti-inflammatory drugs (NSAIDS). It will be appreciated that in such a combination therapy, the one or more antibody (or one or more nucleic acid, host cell, vector, DNA therapeutic, RNA therapeutic, or composition) and the one or more anti-inflammatory agent and/or one or the more antibiotic agent can be administered in any order and any sequence, or together.
In some embodiments, an antibody (or one or more nucleic acid, host cell, vector, DNA therapeutic, RNA therapeutic, or composition) is administered to a subject who has previously received one or more anti-inflammatory agent and/or one or more antibiotic agent. In some embodiments, one or more anti-inflammatory agent and/or one or more antibiotic agent is administered to a subject who has previously received an antibody (or one or more nucleic acid, host cell, vector, DNA therapeutic, RNA therapeutic, or composition).
In a related aspect, uses of the presently disclosed antibodies, antigen-binding fragments, vectors, DNA therapeutics, RNA therapeutics, host cells, and compositions (e.g., in the diagnosis, prophylaxis, and/or treatment of a bacterial (e.g., Staphylococcus) infection, in the manufacture of a medicament for preventing or treating a bacterial (e.g., Staphylococcus) infection) are provided.
The present disclosure further provides a kit comprising one or more of any antibodies, antigen-binding fragments, polynucleotides, nucleic acids, vectors, DNA therapeutics, RNA therapeutics, or other compositions disclosed herein.
In certain embodiments, an antibody, antigen-binding fragment, polynucleotide, vector, DNA therapeutic, RNA therapeutic, host cell, or composition is provided for use in a method of treating or preventing a bacterial infection in a subject.
In certain embodiments, an antibody, antigen-binding fragment, or composition is provided for use in a method of manufacturing or preparing a medicament for treating or preventing a bacterial infection in a subject.
Also provided is a library comprising a plurality of antibodies or antigen-binding fragments, or comprising polynucleotides encoding the same, wherein the library comprises or encodes certain human variable domain alleles. Briefly, the present disclosure teaches that certain alleles and combinations of alleles are used by clonally related antibodies that can bind to a herein disclosed antigen. A library can, for example, comprise polynucleotides having a genomic allele sequence, and/or can contain alternative polynucleotide sequences that encode a same amino acid sequence encoded by the genomic allele. The library can be enriched for these sequences. Alternatively or additionally, a library can comprise antibodies or antigen-binding fragments. The antibodies or antigen-binding fragments can be expressed (e.g., the polynucleotides can be expressed by a plurality of host cell, such as, for example, yeast, human B cells, or human plasma cells, and antibody variable domain genes may undergo recombination, somatic hypermutation, or both) and the library can be screened for antibodies or antigen-binding fragments that bind to a Staphylococcus antigen as provided herein.
In certain embodiments, a library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences encoded by any one or more of the following human immunoglobulin genes: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4- 1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6- 57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1-16;
IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof. In some embodiments, the human immunoglobulin genes in the library consist essentially of, or consist of, any one or more of the foregoing human immunoglobulin genes.
In certain embodiments, a library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences from: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3- 33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3- 21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1 ; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1 ; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6- 57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3- 49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
An antibody, antigen-binding fragment, or polynucleotide encoding the same in a library can further comprise (or encode) additional variable domain amino acid sequence; the additional
variable domain amino acid sequence may be randomized in this regard. A library can comprise host cells that express the plurality of antibodies or antigen-binding fragments. Any suitable host cell can be used, for example, in certain embodiments, the library comprises a yeast display library, B cells, plasma cells, or any combination thereof. Preferably, a library encompasses genetic variation (e.g., randomized and/or rationally designed) among the plurality of antibodies, antigen-binding fragments, or polynucleotides) while maintaining amino acid sequences from any one the human immunoglobulin genes or combinations of human immunoglobulin genes described above. Variation can, for example, be found in one or more of the CDRs, such as CDRH3, CDRL3, or both. In some embodiments, variation is found in two or more of the CDRs. In some embodiments, variation is found in one or more framework region sequences. A library can be screened to identify antibodies and/or antigen-binding fragments that bind to or more antigen as described herein (e.g., an LTA, ClfA, Gmd, Hla, HIgA, HIgB, a Luk (e.g., LukE, LukD, LukF, LukS, LukED, or LukSF), a SpA, an alpha toxin, a gamma toxin, a Sbi or any combination thereof). The antigen can be isolated, fixed (e.g., immobilized on a plate) or expressed by a host cell (e.g., bacterium).
Table 4 summarizes CDR amino acid sequences, variable domain amino acid sequences, constant domain sequences, and examples of corresponding polynucleotide sequences (SEQ ID NOS.) of certain antibodies of the present disclosure. IMGT CDR sequences are shown in bold font. “Vkappa” and “Vlambda” indicate light chain variable domain sequences. Underlining in the CDR and V region sequences show mutation as compared to the germline sequence.
The present disclosure also provides the following non-limiting enumerated Embodiments: Embodiment 1. An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and/or CDRL3 amino acid sequence(s), or functional variants thereof (and optionally comprising the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, or functional variants thereof), of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314,
respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively, wherein the antibody or antigen-binding fragment is capable of binding to: a lipoteichoic acid (LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; or a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 2. An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, and optionally the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences, of one of the following antibodies: SSG20; SSC35; SSE73; SSA12; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSE1; SSE158; SSF2; SSF11; SSF10; SSB18; SSB24; SSB54; SSB65; SSB67; SSH3; SSF33; SSL5; SSL17; SSL35; SSG50; SSG54; SSM47; SSG44; SSG48; SSM37; SSH4; SSH7; SSG27; SSG39; SSM45; SSF52; SSG51; SSF34; SSG59; SSAA8; SSAA26; SSAB5; SSAI3; SSAI8; SSAI2; SSAF1; SSAL2; SSAM6; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; SSF69; SSN2; SSN6; SSH8; SSM35; SSM36; and SSM40, wherein the antibody or antigen-binding fragment is capable of binding to: a lipoteichoic acid (LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; or a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin) .
Embodiment 3. The antibody or antigen-binding fragment of Embodiment 1 or Embodiment 2, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences are according to: (i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system;(vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
Embodiment 4. An antibody, or an antigen-binding fragment thereof, comprising:
(i) a complementarity determining region (CDR)H1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.: 42, 32, 122, 22, 132, 142,
152, 162, 242, 172, 2, 12, 182, 52, 192, 212, 102, 222, 112, 232, 82, 92, 62, 72, 202, 281, 291, 301, 311,
321, 331, 341, 351, 361, 371, 381, 391, 401, 411, 421, 433, 443, 453, 463, 473, 483, 493, 503, 513, 523,
533, 543, 553, 563, 573, 583, 593, 603, 613, 623, 633, 643, 653, 663, 673, 683, 693, 703, 713, 723, 733,
743, and 753, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline- encoded amino acid;
(ii) a CDRH2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:43, 33, 123, 23, 133, 143, 153, 163, 243, 173, 3, 13, 183, 53, 193,
213, 103, 223, 113, 233, 83, 93, 63, 73, 203, 282, 292, 302, 312, 322, 332, 342, 352, 362, 372, 382, 392,
402, 412, 422, 434, 444, 454, 464, 474, 484, 494, 504, 514, 524, 534, 544, 554, 564, 574, 584, 594, 604,
614, 624, 634, 644, 654, 664, 674, 684, 694, 704, 714, 724, 734, 744, and 754 or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid;
(iii) a CDRH3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.: 44, 34, 124, 24, 134, 144, 154, 164, 244, 174, 4, 14, 184, 54, 194,
214, 104, 224, 114, 234, 84, 94, 64, 74, 204, 283, 293, 303, 313, 323, 333, 343, 353, 363, 373, 383, 393,
403, 413, 423, 435, 445, 455, 465, 475, 485, 495, 505, 515, 525, 535, 545, 555, 565, 575, 585, 595, 605,
615, 625, 635, 645, 655, 665, 675, 685, 695, 705, 715, 725, 735, 745, and 755, or a sequence variant
thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid;
(iv) a CDRL1 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:46, 36, 126, 26, 136, 146, 156, 166, 246, 176, 6, 16, 186, 56, 196,
216, 106, 226, 116, 236, 86, 96, 66, 76, 206, 285, 295, 305, 315, 325, 335, 345, 355, 365, 375, 385, 395,
405, 415, 425, 437, 447, 457, 467, 477, 487, 497, 507, 517, 527, 537, 547, 557, 567, 577, 587, 597, 607,
617, 627, 637, 647, 657, 667, 677, 687, 697, 707, 717, 727, 737, 747, and 757, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline -encoded amino acid;
(v) a CDRL2 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:47, 37, 127, 27, 137, 147, 157, 167, 247, 177, 7, 17, 187, 57, 197,
217, 107, 227, 117, 237, 87, 97, 67, 77, 207, 286, 296, 306, 316, 326, 336, 346, 356, 366, 376, 386, 396,
406, 416, 426, 438, 448, 458, 468, 478, 488, 498, 508, 518, 528, 538, 548, 558, 568, 578, 588, 598, 608,
618, 628, 638, 648, 658, 668, 678, 688, 698, 708, 718, 728, 738, 748, and 758, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid; and/or
(vi) a CDRL3 comprising, consisting essentially of, or consisting of the amino acid sequence set forth in any one of SEQ ID NOs.:48, 38, 128, 28, 138, 148, 158, 168, 248, 178, 8, 18, 188, 58, 198,
218, 108, 228, 118, 238, 88, 98, 68, 78, 208, 287, 297, 307, 317, 327, 337, 347, 357, 367, 377, 387, 397,
407, 417, 427, 439, 449, 459, 469, 479, 489, 499, 509, 519, 529, 539, 549, 559, 569, 579, 589, 599, 609,
619, 629, 639, 649, 659, 669, 679, 689, 699, 709, 719, 729, 739, 749, and 759, or a sequence variant thereof comprising one, two, or three acid substitutions, one or more of which substitutions is optionally a conservative substitution and/or is a substitution to a germline-encoded amino acid, wherein the antibody or antigen-binding fragment is capable of binding to: a lipoteichoic acid (LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; or a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 5. The antibody or antigen-binding fragment of Embodiment 4, comprising a CDRH3 and a CDRL3, wherein the CDRH3 and the CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (i) 44 and 48, respectively; (ii) 34 and 38, respectively; (iii) 124 and 128, respectively; (iv) 24 and 28, respectively; (v) 134 and 138, respectively; (vi) 144 and 148, respectively; (vii) 154 and 158, respectively; (viii) 164 and 168, respectively; (ix) 244 and 248, respectively; (x) 174 and 178, respectively; (xi) 4 and 8, respectively; (xii) 14 and 18,
respectively; (xiii) 184 and 188, respectively; (xiv) 54 and 58, respectively; (xv) 194 and 198, respectively; (xvi) 214 and 218, respectively; (xvii) 104 and 108, respectively; (xviii) 224 and 228, respectively; (xix) 114 and 118, respectively; (xx) 234 and 238, respectively; (xxi) 84 and 88, respectively; (xxii) 94 and 98, respectively; (xxiii) 64 and 68, respectively; (xxiv) 74 and 78, respectively; (xxv) 204 and 208, respectively; (xxvi) 283 and 287, respectively; (xxvii) 293 and 297, respectively; (xxviii) 303 and 307, respectively; (xxix) 313 and 317, respectively; (xxx) 323 and 327, respectively; (xxxi) 333 and 337, respectively; (xxxii) 343 and 347, respectively; (xxxiii) 353 and 357, respectively; (xxxiv) 363 and 367, respectively; (xxxv) 373 and 377, respectively, (xxxvi) 383 and 387, respectively; (xxxvii) 393 and 397, respectively; (xxxviii) 403 and 407, respectively; (xxxix) 413 and 417, respectively; (xl) 423 and 427, respectively; (xli) 435 and 439, respectively; (xlii) 445 and 449, respectively; (xliii) 455 and 459, respectively; (xliv) 465 and 469, respectively; (xlv) 475 and 479, respectively; (xlvi) 485 and 489, respectively; (xlvii) 495 and 499, respectively; (xlviii) 505 and 509, respectively; (xlix) 515 and 519, respectively; (1) 525 and 529, respectively; (li) 535 and 539, respectively; (lii) 545 and 549, respectively; (liii) 555 and 559, respectively; (liv) 565 and 569, respectively; (Iv) 575 and 579, respectively; (Ivi) 585 and 589, respectively; (Ivii) 595 and 599, respectively; (Iviii) 605 and 609, respectively; (lix) 615 and 619, respectively; (lx) 625 and 629, respectively; (Ixi) 635 and 639, respectively; (Ixii) 645 and 649, respectively; (Ixiii) 655 and 659, respectively; (Ixiv) 665 and 669, respectively; (Ixv) 675 and 679, respectively; (Ixvi) 685 and 689, respectively; (Ixvii) 695 and 699, respectively; (Ixviii) 705 and 709, respectively; (Ixix) 715 and 719, respectively; (Ixx) 725 and 729, respectively; (Ixxi) 735 and 739, respectively; (Ixxii) 745 and 749, respectively; or (Ixxiii) 755 and 759, respectively.
Embodiment 6. An antibody, or an antigen-binding fragment thereof, comprising a complementarity determining region (CDR)H1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (i) 42-44 and 46-48, respectively; (ii) 32-34 and 36-38, respectively; (iii) 122-124 and 126-128, respectively; (iv) 22-24 and 26-28, respectively; (v) 132-134 and 136-138, respectively; (vi) 142-144 and 146-148, respectively; (vii) 152-154 and 156-158, respectively; (viii) 162-164 and 166-168, respectively; (ix) 242-244 and 246-248, respectively; (x) 172-174 and 176-178, respectively; (xi) 2-4 and 6-8, respectively; (xii) 12-14 and 16-18, respectively; (xiii) 182-184 and 186-188, respectively; (xiv) 52-54 and 56-58, respectively; (xv) 192-194 and 196-198, respectively; (xvi) 212-214 and 216-218, respectively; (xvii) 102-104 and 106-108, respectively; (xviii) 222-224 and 226-228, respectively; (xix) 112-114 and 116-118, respectively; (xx) 232-234 and 236-238, respectively; (xxi) 82-84 and 86-88, respectively; (xxii) 92-94 and 96-98,
respectively; (xxiii) 62-64 and 66-68, respectively; (xxiv) 72-74 and 76-78, respectively; (xxv) 202-204 and 206-208, respectively; (xxvi) 281-283 and 285-287, respectively; (xxvii) 291-293 and 295-297, respectively; (xxviii) 301-303 and 305-307, respectively; (xxix) 311-313 and 315-317, respectively; (xxx) 321-323 and 325-327, respectively; (xxxi) 331-333 and 335-337, respectively; (xxxii) 341-343 and 345- 347, respectively; (xxxiii) 351-353 and 355-357, respectively; (xxxiv) 361-363 and 365-367, respectively; (xxxv) 371-373 and 375-377, respectively; (xxxvi) 381-383 and 385-387, respectively; (xxxvii) 391-393 and 395-397, respectively; (xxxviii) 401-403 and 405-407, respectively; (xxxix) 411-413 and 415-417, respectively; (xl) 421-423 and 425-427, respectively, ; (xli) 433-435 and 437-439, respectively; (xlii) 443-445 and 447-449, respectively; (xliii) 453-455 and 457-459, respectively; (xliv) 463-465 and 467- 469, respectively; (xlv) 473-475 and 477-479, respectively; (xlvi) 483-485 and 487-489, respectively; (xlvii) 493-495 and 497-499, respectively; (xlviii) 503-505 and 507-509, respectively; (xlix) 513-515 and 517-519, respectively; (1) 523-525 and 527-529, respectively; (li) 533-535 and 537-539, respectively; (lii) 543-545 and 547-549, respectively; (liii) 553-555 and 557-559, respectively; (liv) 563-565 and 567-569, respectively; (Iv) 573-575 and 577-579, respectively; (Ivi) 583-585 and 587-589, respectively; (Ivii) 593- 595 and 597-599, respectively; (Iviii) 603-605 and 607-609, respectively; (lix) 613-615 and 617-619, respectively; (lx) 623-625 and 627-629, respectively; (Ixi) 633-635 and 637-639, respectively; (Ixii) 643- 645 and 647-649, respectively; (Ixiii) 653-655 and 657-659, respectively; (Ixiv) 663-665 and 667-669, respectively; (Ixv) 673-675 and 677-679, respectively; (Ixvi) 683-685 and 687-689, respectively; (Ixvii) 693-695 and 697-699, respectively; (Ixviii) 703-705 and 707-709, respectively; (Ixix) 713-715 and 717- 719, respectively; (Ixx) 723-725 and 727-729, respectively; (Ixxi) 733-735 and 737-739, respectively; (Ixxii) 743-745 and 747-749, respectively; or (Ixxiii) 753-755 and 757-759, respectively, wherein the antibody or antigen-binding fragment is capable of binding to: a lipoteichoic acid (LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; or a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 7. The antibody or antigen-binding fragment of any one of Embodiments 1-6, wherein the antibody or antigen-binding fragment is capable of binding to a LTA, wherein, optionally, the antibody or antigen-binding binding fragment is capable of binding to: (i) a Type I LTA; (ii) a Type II LTA; (iii) a Type IV LTA; or (iv) any combination of (i)-(iii).
Embodiment 8. The antibody or antigen-binding fragment of any one of Embodiments 1-7, wherein the antibody or antigen-binding fragment is capable of binding to any one, any two, any three, any four, any five, any six, any seven, any eight, any nine, any ten, any eleven, or all twelve of: .S' aureus LPR3757; .S' aureus NE284 TE2 mut; .S' epidermidis RP62A; .S' epidermidis 1200; .S' epidermidis
NIH04008; .S'. lugdunensis', S. pyogenes,' E.faecalis', S. agalactiac, S. hominis', L. garvieac, and .S'. pneumoniae.
Embodiment 9. The antibody or antigen-binding fragment of any one of Embodiments 1-8, wherein the antibody or antigen-binding binding fragment is capable of binding to a Gmd, wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a .S' aureus, to .S' epide rmidis. or to .S' aureus and to .S' epidermidis .
Embodiment 10. The antibody or antigen-binding fragment of any one of Embodiments 1-9, wherein the antibody or antigen-binding fragment is capable of binding to a ClfA.
Embodiment 11. The antibody or antigen-binding fragment of Embodiment 10, wherein the antibody or antigen-binding binding is capable of binding to a ClfA_001, a ClfA_002, and/or a ClfA_004.
Embodiment 12. The antibody or antigen -binding fragment of any one of Embodiments 1-11, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA), wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a SPAKKAA.
Embodiment 13. The antibody or antigen-binding fragment of any one of Embodiments 1-12, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin, wherein, optionally, the alpha toxin comprises a-hemolysin (also called Hla).
Embodiment 14. The antibody or antigen-binding fragment of any one of Embodiments 1-13, wherein the antibody or antigen-binding fragment is capable of binding to a gamma toxin.
Embodiment 15. The antibody or antigen-binding fragment of any one of Embodiments 1-14, wherein the antibody or antigen-binding fragment is capable of binding to a leukocidin, wherein, optionally, the leukocidin comprises a LukD, a LukE, a LukF, a LukS, or any combination thereof.
Embodiment 16. The antibody or antigen-binding fragment of Embodiment 15, wherein the antibody or antigen-binding fragment is capable of binding to a LukED, a LukSF, or to a LukED and a LukSF.
Embodiment 17. The antibody or antigen-binding fragment of any one of Embodiments 1-16, wherein the antibody or antigen-binding fragment is capable of binding to a HIgA, a HIgB, or both.
Embodiment 18. The antibody or antigen-binding fragment of any one of Embodiments 1-17, wherein the antibody is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin.)
Embodiment 19. The antibody or antigen-binding fragment of any one of Embodiments 1-18, wherein the antibody or antigen-binding fragment comprises an amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising,
consisting essentially of, or consisting of, the amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3- 49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3- 11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2- 30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1-16; IGKV3-20; IGLV3-21; IGHV1- 69; IGHV3-43, or any combination thereof.
Embodiment 20. The antibody or antigen-binding fragment of any one of Embodiments 1-19, wherein the antibody or antigen-binding fragment comprises amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1- 5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and
IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
Embodiment 21. The antibody or antigen-binding fragment of any one of Embodiments 1-20, wherein the antibody or antigen-binding fragment comprises a framework amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, a framework amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1- 8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1- 16; IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof
Embodiment 22. The antibody or antigen-binding fragment of any one of Embodiments 1-21, wherein the antibody or antigen-binding fragment comprises framework amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the framework amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3- 7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D- 12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4- 61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3,
IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
Embodiment 23. The antibody or antigen-binding fragment of any one of Embodiments 1-22, comprising a heavy chain variable domain framework region (VHFR)1, a VHFR2, a VHFR3, a light chain variable domain framework region (VLFR)1, a VLFR2, a VLFR3, and/or a VLFR4 (or a variant of the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4 comprising one, two, three, four, or five substitutions, insertions, and/or deletions, or a variant having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4, respectively) of the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and
404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively.
Embodiment 24. The antibody or antigen-binding fragment of any one of Embodiments 21-23, wherein the framework region or regions are according to: (i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system;(vi) the North numbering system; (vii) the Contact numbering system; (viii)the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
Embodiment 25. The antibody or antigen-binding fragment of any one of Embodiments 1-24, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively;
(xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively.
Embodiment 26. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
Embodiment 27. The anti -LTA antibody or antigen-binding fragment of Embodiment 26, wherein the CDRs are according to: (i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system;(vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
Embodiment 28. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences set forth in SEQ ID NOs.:22-24 and 26-28, respectively.
Embodiment 29. The anti-LTA antibody or antigen-binding fragment of any one of Embodiments 26-28, comprising an amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequence encoded by: IGHV3-7; IGHJ3; IGKV1-6; IGKJ1; or any combination thereof.
Embodiment 30. The anti-LTA antibody or antigen-binding fragment of any one of Embodiments 26-29, comprising amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequences encoded by: IGHV3-7; IGHJ3; IGKV1-6; and IGKJ1.
Embodiment 31. The anti-LTA antibody or antigen-binding fragment of any one of Embodiments 1-30, comprising a framework amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, a framework amino acid sequence encoded by: IGHV3-7; IGHJ3; IGKV1-6; or IGKJ1.
Embodiment 32. The anti-LTA antibody or antigen-binding fragment of any one of Embodiments 1-31, comprising framework amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the framework amino acid sequences encoded by: IGHV3-7; IGHJ3; IGKV1-6; and IGKJ1.
Embodiment 33. The anti-LTA antibody or antigen-binding fragment of any one of Embodiments 1-32, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
Embodiment 34. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising a heavy chain variable domain (VH) and a light chain variable domain (VL), wherein the VH and VL comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOs.:21 and 25, respectively.
Embodiment 35. The antibody or antigen-binding fragment of any one of Embodiments 1-34, wherein the antibody, or the antigen-binding fragment, comprises a human antibody, a monoclonal antibody, a purified antibody, a single chain antibody (e.g., scFv), a Fab, a Fab’, a F(ab’)2, or Fv.
Embodiment 36. The antibody or antigen -binding fragment of any one of Embodiments 1-35, wherein the antibody or antigen-binding fragment is a multi specific antibody or antigen-binding fragment, wherein, optionally, the antibody or antigen-binding fragment is a bi-specific antibody or antigen-binding fragment.
Embodiment 37. The antibody or antigen-binding fragment of embodiment 36, which comprises (1) the six CDRS, and optionally the VH and VL, of a first antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1) and (2) the six CDRS, and optionally the VH and VL, of a second, different antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1).
Embodiment 38. The antibody or antigen-binding fragment of Embodiment 36 or 37, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and a ClfA; a SpA and a ClfA; a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; or a Sbi and a ClfA.
Embodiment 39. The antibody or antigen-binding fragment of any one of Embodiments 36-38, comprising a first antigen-binding domain and second antigen-binding domain, wherein the first antigenbinding domain or the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; or (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC10; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF10; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30;
(Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE150; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
Embodiment 40. The antibody or antigen-binding fragment of any one of Embodiments 36-39, comprising a first antigen-binding domain and second antigen-binding domain, wherein the first antigenbinding domain and the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S or M428L/N434S/H435R/Y436F mutations.
Embodiment 41. An antibody, or an antigen -binding fragment thereof, which is a multi-specific antibody or antigen-binding fragment thereof, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and a ClfA; a SpA and a ClfA; a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; or a Sbi and a ClfA.
Embodiment 42. The antibody or antigen -binding fragment of any one of Embodiments 36-41, which comprises or is of a format selected from: a Bispecific T cell Engager (BiTE); a DART; a Knobs- Into-Holes (KIH) assembly; a scFv-CH3-KIH assembly; a KIH Common Light-Chain antibody; a TandAb; a Triple Body; a TriBi Minibody; a Fab-scFv; a scFv-CH-CL-scFv; a F(ab’)2-scFv2; a tetravalent Hcab; an intrabody; a CrossMab; a Dual Action Fab (DAF) (two-in-one or four-in-one); a DutaMab; a DT-IgG, a charge paired antibody; a Fab-arm Exchange antibody, a SEEDbody; a Triomab; a LUZ-Y assembly, an Fcab; a K/.-body: an orthogonal Fabs antibody; a DVD-Ig; am IgG(H)-scFv; an scFv-(H)IgG; an IgG(L)-scFv; an scFv-(L)IgG; an IgG(L,H)-Fv; an IgG(H)-V; a V(H)-IgG; an IgG(L)-V; a V(L)-IgG; a KIH IgG-scFab; a 2scFv-IgG; a IgG-2scFv; a scFv4-Ig; a Zybody; a DVI-IgG (four-in- one), a FIT-Ig; a WuxiBody; and an In-Elbow-Insert Ig.
Embodiment 43. The antibody or antigen -binding fragment of any one of Embodiments 1-42, wherein the antibody or antigen-binding fragment comprises an (e.g., IgGl or IgG3) Fc polypeptide or a fragment thereof.
Embodiment 44. The antibody or antigen -binding fragment of any one of Embodiments 1-43, which comprises an IgG, IgA, IgM, IgE, or IgD isotype.
Embodiment 45. The antibody or antigen-binding fragment of any one of Embodiments 1-44, which comprises an IgG isotype selected from IgGl, IgG2, IgG3, and IgG4, optionally an IgGl isotype comprising a heavy chain with a C-terminal lysine removed or a C-terminal glycine -lysine removed.
Embodiment 46. The antibody or antigen -binding fragment of any one of Embodiments 1-45, which comprises an IgGl isotype.
Embodiment 47. The antibody or antigen-binding fragment of any one of Embodiments 1-46, which comprises an IgGlm3 allotype, an IgGlml7 allotype, an IgGlml allotype, or any combination thereof.
Embodiment 48. The antibody or antigen-binding fragment of any one of Embodiments 1-45, which comprises an IgG3 isotype.
Embodiment 49. The antibody or antigen-binding fragment of any one of Embodiments 43-48, wherein the Fc polypeptide or fragment thereof comprises: (i) a mutation that increases binding affinity to a human FcRn (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols)), as compared to a reference Fc polypeptide that does not comprise the mutation; and/or (ii) a mutation that increases binding affinity to a human FcyR (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols, and/or as measured using mesoscale discovery (MSD))) as compared to a reference Fc polypeptide that does not comprise the mutation.
Embodiment 50. The antibody or antigen -binding fragment of Embodiment 49, wherein the mutation that increases binding affinity to a human FcRn comprises: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q311I; D376V; T307A; E380A; or any combination thereof.
Embodiment 51. The antibody or antigen-binding fragment of Embodiment 49 or 50, wherein the mutation that increases binding affinity to a human FcRn comprises: (i) M428L/N434S; (ii) M252Y/S254T/T256E; (iii) T250Q/M428L; (iv) P257I/Q31 II; (v) P257I/N434H; (vi) D376V/N434H; (vii) T307A/E380A/N434A; (viii) M428L/N434A; or (ix) any combination of (i)-(viii).
Embodiment 52. The antibody or antigen-binding fragment of any one of Embodiments 49-51, wherein the mutation that increases binding affinity to a human FcRn comprises M428L/N434S or M428L/N434A.
Embodiment 53. The antibody or antigen -binding fragment of any one of Embodiments 49-52, wherein the mutation that enhances binding to a FcyR comprises S239D; I332E; A330L; G236A; or any combination thereof.
Embodiment 54. The antibody or antigen-binding fragment of any one of Embodiments 49-53, wherein the mutation that enhances binding to a FcyR comprises: (i) S239D/I332E; (ii) S239D/A330L/I332E; (iii) G236A/S239D/I332E; or (iv) G236A/A330L/I332E, wherein the Fc polypeptide or fragment thereof optionally comprises Ser at position 239.
Embodiment 55. The antibody or antigen-binding fragment of any one of Embodiments 1-54, which comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or which is aglycosylated, and/or which is afucosylated.
Embodiment 56. The antibody or antigen -binding fragment of any one of Embodiments 1-55, comprising in a heavy chain thereof, the amino acid mutation(s) set forth in any one of (i)-(xix): (i) G236A, L328V, and Q295E; (ii) G236A, P230A, and Q295E; (iii) G236A, R292P, and I377N; (iv) G236A, K334A, and Q295E; (v) G236S, R292P, and Y300L; (vi) G236A and Y300L; (vii) G236A, R292P, and Y300L; (viii) G236S, G420V, G446E, and L309T; (ix) G236A and R292P; (x) R292P and Y300L; (xi) G236A and R292P; (xii) Y300L; (xiii) E345K, G236S, L235Y, and S267E; (xiv) E272R, L309T, S219Y, and S267E; (xv) G236Y; (xvi) G236W; (xvii) F243L, G446E, P396L, and S267E; (xviii) G236A, S239D, and H268E; (xix) any one or more of the mutations shown in Figure 14A, Figure 14B, Figure 14C, Figure 14E, Figure 14F, Figure 14G, Figure 14H, Figure 141, Figure 14J, or Figure 14K.
Embodiment 57. The antibody or antigen-binding fragment of any one of Embodiments 1-56, comprising in a heavy chain thereof, one or more of the following the amino acid mutations: G236A; Y300L; R292P.
Embodiment 58. The antibody or antigen-binding fragment of any one of Embodiments 1-57, comprising, in a heavy chain thereof, an amino acid mutation that promotes complement activation, wherein, optionally, the amino acid mutation that promotes complement activation comprises or consists of: K236W/E333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L; G236A/Y300L; or G236A/R292P/Y300L.
Embodiment 59. The antibody or antigen-binding fragment of any one of Embodiments 1-58, comprising, in a heavy chain or a CH3 thereof, an amino acid mutation that reduces binding by Staphylococcus Protein A, wherein, the optionally, amino acid mutation that reduces binding by Staphylococcus Protein A comprises or consists of: H435R; Y436F; or H435R/Y436F.
Embodiment 60. The antibody or antigen -binding fragment of any one of Embodiments 1-59, comprising a human IgGl isotype and comprising, in a heavy chain or a CH3 thereof, the amino acid mutations M428L/N434S/H435R/Y436F.
Embodiment 61. The antibody or antigen-binding fragment of any one of Embodiments 1-60, which is capable of neutralizing an infection (in vitro, ex vivo, and/or in vivo) by a bacteria expressing the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi.
Embodiment 62. The antibody or antigen-binding fragment of any one of Embodiments 1-61, which is capable of neutralizing an S. aureus infection in vitro, ex vivo, or in vivo.
Embodiment 63. The antibody or antigen-binding fragment of any one of Embodiments 1-62, which is capable of opsonizing and/or is capable of promoting phagocytosis of a bacteria expressing the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, Sbi, or gamma toxin, wherein, optionally, the bacteria comprises .S', aureus.
Embodiment 64. The antibody or antigen-binding fragment of any one of Embodiments 1-63, which: is afucosylated; has been produced in a host cell that is incapable of fucosylation or that is inhibited in its ability to fucosylate a polypeptide; has been produced under conditions that inhibit fucosylation thereof by a host cell; or any combination thereof.
Embodiment 65. The antibody or antigen-binding fragment of any one of Embodiments 1-64, which is human, humanized, or chimeric.
Embodiment 66. An antibody, or an antigen-binding fragment, that is capable of binding to an S. aureus antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi, wherein, optionally: (1) the antibody or antigenbinding fragment is human, humanized, or chimeric; (2) the antibody or antigen-binding fragment binds to a LTA and does not complete with pagibaximab for binding to the LTA; (3) the antibody or antigenbinding fragment comprises an amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequence encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3;
IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1-16; IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof; (4) the antibody or antigen-binding fragment comprises amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1- 6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4- 61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2; (5) the antibody or antigen-binding fragment comprises a framework amino acid sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, a framework amino acid sequence
encoded by: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1- 24; IGKV1-16; IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof; (6) the antibody or antigen-binding fragment comprises framework amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising or consisting essentially of or consisting of, the framework amino acid sequences encoded by: (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1- 5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2;
(xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2; (7) the antibody or antigen-binding fragment comprises a VH and a VL that comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively; and/or (8) the antibody or antigen-binding fragment comprises an Fc polypeptide or fragment thereof, or a heavy chain, according to any one of embodiments 43-60.
Embodiment 67. An isolated polynucleotide encoding the antibody or antigen-binding fragment of any one of Embodiments 1-66, or encoding a VH, a Fd, a heavy chain, a VL, and/or a light chain of the antibody or the antigen-binding fragment.
Embodiment 68. The polynucleotide of Embodiment 67, wherein the polynucleotide comprises deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), wherein the RNA optionally comprises messenger RNA (mRNA)
Embodiment 69. The polynucleotide of Embodiment 68, wherein the polynucleotide comprises self-amplifying RNA (saRNA), trans-amplifying RNA (taRNA), or circular RNA (circRNA).
Embodiment 70. The polynucleotide of any one of Embodiments 67-69, comprising a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof, wherein, optionally, the polynucleotide comprises a pseudouridine, aN6-methyladenonsine, a 5 -methylcytidine, a 2-thiouridine, or any combination thereof, wherein, further optionally, the pseudouridine comprises Nl- methylpseudouridine .
Embodiment 71. The polynucleotide of any one of Embodiments 67-70, which is codon- optimized for expression in a host cell.
Embodiment 72. The polynucleotide of Embodiment 71, wherein the host cell comprises a human cell or a CHO cell.
Embodiment 73. The polynucleotide of Embodiment 71 , wherein the host cell is a production host cell.
Embodiment 74. The polynucleotide of Embodiment 71, wherein the host cell is an in vivo human host cell.
Embodiment 75. A recombinant vector, DNA therapeutic, or RNA therapeutic comprising the polynucleotide of any one of Embodiments 67-74.
Embodiment 76. A DNA therapeutic construct or RNA therapeutic construct comprising the polynucleotide of any one of Embodiments 67-74 encapsulated in a carrier, wherein the carrier optionally comprises a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
Embodiment 77. A recombinant vector comprising the polynucleotide of any one of Embodiments 67-74.
Embodiment 78. A host cell comprising the polynucleotide of any one of Embodiments 67-74 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77, wherein the
polynucleotide is optionally heterologous to the host cell and/or wherein the host cell is capable of expressing the encoded antibody or antigen-binding fragment or polypeptide.
Embodiment 79. An isolated human B cell comprising the polynucleotide of any one of Embodiments 67-74 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77, wherein polynucleotide is optionally heterologous to the human B cell and/or wherein the human B cell is immortalized.
Embodiment 80. A composition or combination comprising: (i) the antibody or antigen-binding fragment of any one of Embodiments 1-66; (ii) the polynucleotide of any one of Embodiments 67-74; (iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77; (iv) the host cell of Embodiment 78; and/or (v) the human B cell of Embodiment 79, and, optionally, a pharmaceutically acceptable excipient, carrier, or diluent.
Embodiment 81. The composition or combination of Embodiment 80, which comprises (1) a first antibody or antigen -binding fragment of any one of Embodiments 1-66 and (2) a second, different antibody or antigen -binding fragment of any one of Embodiments 1-66.
Embodiment 82. The composition or combination of Embodiment 81, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are capable of binding to: an LTA and a Gmd, respectively; an LTA and an alpha toxin, respectively; an LTA and a gamma toxin, respectively; an LTA and a leucocidin, respectively; an LTA and a SpA, respectively; an LTA and a ClfA, respectively; an alpha toxin and a Gmd, respectively; an alpha toxin and a gamma toxin, respectively; an alpha toxin and a leucocidin, respectively; an alpha toxin and a SpA, respectively; an alpha toxin and a ClfA, respectively; a gamma toxin and a Gmd, respectively; a gamma toxin and a leucocidin, respectively; a gamma toxin and a SpA, respectively; a gamma toxin and a ClfA, respectively; a leucocidin and a SpA, respectively; a leucodin and a Gmd, respectively; a leucocidin and a ClfA, respectively; a Gmd and a ClfA, respectively; a SpA and a ClfA, respectively; a Sbi and a LTA, respectively; a Sbi and a Gmd, respectively; a Sbi and an alpha toxin, respectively; a Sbi and a gamma toxin, respectively; a Sbi and a leucocidin, respectively; a Sbi and a SpA, respectively; or a Sbi and a SlfA, respectively.
Embodiment 83. The composition or combination of any one of Embodiments 80-82, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment or the second antibody or antigen-binding fragment comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii)
SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC1O; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF1O; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE15O; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
Embodiment 84. The composition or combination of any one of Embodiments 80-83, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment comprise the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S/H435R/Y436F mutations.
Embodiment 85. The composition or combination of Embodiment 80, comprising a first polynucleotide and a second polynucleotide, each according to any one of Embodiments 67-74, or a recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77, encoding two or more antibodies or antigen-binding fragments, wherein the first antibody or antigen-binding fragment encoded by the first polypeptide and second antibody or antigen-binding fragment encoded by the second polypeptide, respectively, or the multispecific antibody or antigen-binding fragment, comprise(s) CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3, and optionally VH and VL, according to any two antibodies of the present disclosure, or any antigen-binding fragments thereof.
Embodiment 86. A composition comprising the polynucleotide of any one of Embodiments 67- 74 or the vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77 encapsulated in a carrier molecule, wherein the carrier molecule optionally comprises a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
Embodiment 87. A(n, e.g., in vitro or ex vivo) method of making an antibody or antigen-binding fragment of any one of Embodiments 1-66, comprising culturing the host cell of Embodiment 78 or the human B cell of Embodiment 79 for a time and under conditions sufficient for the host cell or human B cell, respectively, to express the antibody or antigen-binding fragment.
Embodiment 88. The method of making an antibody or antigen-binding fragment of Embodiment 87, wherein the host cell of Embodiment 78 or the human B cell of Embodiment 79 comprises the polynucleotide of any one of Embodiments 67-74 and/or recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77.
Embodiment 89. The method of Embodiment 87 or 88, further comprising isolating the antibody or antigen-binding fragment.
Embodiment 90. A method of treating or preventing a bacterial infection in a(n, e.g., human) subject, wherein the bacterial infection is optionally bacteremia, wherein further optionally the bacteremia is hemodialysis associated/line associated bacteremia, the method comprising administering to the subject an effective amount of: (i) the antibody or antigen -binding fragment of any one of Embodiments 1-66; (ii) the polynucleotide of any one of Embodiments 67-74; (iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77; (iv) the host cell of Embodiment 78; (v) the human B cell of Embodiment 79; and/or (vi) the composition or combination of any one of Embodiments 80-86, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S', aureus.
Embodiment 91. The method of Embodiment 90, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'. Aureus (MRSA); (2) a Coagulase-negative Staphylococcus (CoNS); (3) .S', epidermidis,' (4) E. faecalis,' (5) .S', pyogenes,' (6) .S'. agalactiae,' (7) L. garviae,' (8) .S', pneumoniae,' (9) .S', lugdunensis,' (10) .S', hominis,' (11) .S', epidermidis,' or (12) any combination of (l)-(l 1).
Embodiment 92. The method of Embodiment 90 or 91, comprising administering a single dose of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
Embodiment 93. The method of any one of Embodiments 90-92, comprising administering two or more doses of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
Embodiment 94. The method of any one of Embodiments 90-93, comprising administering the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition intramuscularly, subcutaneously, or intravenously.
Embodiment 95. The method of any one of Embodiments 90-94, wherein the treatment and/or prevention comprises pre-exposure or post-exposure prophylaxis.
Embodiment 96. The method of any one of Embodiments 90-95, wherein the subject: is using a ventilator; is undergoing, will undergo, or has recently undergone surgery; has a blood stream infection or a history of blood stream infections; is receiving, will receive, or has received a prosthetic joint or other implant; is undergoing, will undergo, or has undergone dialysis; has bacteremia or a history of bacteremia; has neutropenia or a history of neutropenia; has hemodialysis associated/line associated bacteremia or a history of hemodialysis associated/line associated bacteremia; has a wound; or any combination of the foregoing.
Embodiment 97. The method of any one of Embodiments 90-96, further comprising administering to the subject or more antibiotic agent, or wherein the subject has received one or more antibiotic agent.
Embodiment 98. The method of Embodiment 97, wherein the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof.
Embodiment 99. The method of Embodiment 97 or 98, wherein the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin-clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof.
Embodiment 100. The antibody or antigen-binding fragment of any one of Embodiments 1- 61a, the polynucleotide of any one of Embodiments 62-66b, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 67-67b, the host cell of Embodiment 68, the human B cell of Embodiment 69, and/or the composition or combination of any one of Embodiments 70-71, for use in a method of treating or preventing a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
Embodiment 101. The antibody or antigen-binding fragment of any one of Embodiments 1- 66, the polynucleotide of any one of Embodiments 67-74, the recombinant vector, DNA therapeutic, or
RNA therapeutic of any one of Embodiments 75-77, the host cell of Embodiment 78, the human B cell of Embodiment 79, and/or the composition or combination of any one of Embodiments 80-86, for use in the preparation of a medicament for the treatment or prevention of a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
Embodiment 102. A method for in vitro diagnosis of a bacterial infection, the method comprising:
(i) contacting a sample from a subject with an antibody or antigen-binding fragment of any one of Embodiments 1-66; and (ii) detecting a complex comprising an antigen and the antibody, or comprising an antigen and the antigen-binding fragment, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises S. aureus.
Embodiment 103. The antibody, antigen-binding fragment, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, human B cell, or composition for use of Embodiment 100 or 101, or the method of Embodiment 102, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'. Aureus (MRSA); (2) a Coagulase -negative Staphylococcus (CoNS); (3) .S', epidermidis,' (4) E.faecalis,' (5) .S', pyogenes,' (6) .S', agalactiae,' (7) L. garviae,' (8) .S', pneumoniae,' (9) .S', lugdunensis,' (10) .S', hominis,' (11) .S', epidermidis,' or (12) any combination of (l)-(l 1).
Embodiment 104. A library comprising a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprising a plurality of polynucleotides encoding amino acid sequences encoded by any one or more of the following human immunoglobulin genes: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10- 1; IGHV1-24; IGKV1-16; IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof, preferably (1) IGHV3-23/IGHJ3, (2) IGHV3-7/IGVK1-6, or (3) IGHV3-23.
Embodiment 105. The library of Embodiment 104, wherein the human immunoglobulin genes in the library consist essentially of, or consist of, any one or more of the human immunoglobulin genes of Embodiment 104.
Embodiment 106. The library of Embodiment 104 or 105, wherein the library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences from any one or more of (i)-(xxiii): (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3- 23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3- 21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
Embodiment 107. The library of Embodiment 106, wherein the plurality of antibodies or antigenbinding fragments, or the plurality of polynucleotides encoding the antibodies or antigen-binding fragments, further comprises or encodes additional amino acid sequences, wherein the additional amino
acid sequences are optionally randomized and/or are limited to one or more CDR regions of an antibody or antigen-binding fragment.
Embodiment 108. A method comprising screening one or more antibodies of the library of any one of Embodiments 104-107 for binding to a bacterial (e.g., .S', aureus) antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi.
Embodiment 109. A kit comprising a liquid composition comprising and antibody or antigenbinding fragment of any one of Embodiments 1-66, 100, 101, or 103, the polynucleotide of any one of Embodiments 67-74, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of Embodiments 75-77, the host cell of Embodiment 78, the human B cell of Embodiment 79, and/or the composition of any one of Embodiments 80-86, and instructions for use thereof in treating a bacterial (e.g., Staphylococcus) infection in a subject.
Embodiment 201. An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and/or CDRL3 amino acid sequence(s), or functional variants thereof (and optionally comprising the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, or functional variants thereof), of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: (a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532 and 536, respectively; (xii) 542 and 546, respectively; (xiii) 552 and 556, respectively; (xiv) 562 and 566, respectively; (xv) 572 and 576, respectively; (xvi) 582 and 586, respectively; (xvii) 592 and 596, respectively; (xviii) 602 and 606, respectively; (xix) 612 and 616, respectively; (xx) 622 and 626, respectively; (xxi) 632 and 636, respectively; (xxii) 642 and 646, respectively; (xxiii) 652 and 656, respectively; (xxiv) 662 and 666, respectively; (xxv) 672 and 676, respectively; (xxvi) 682 and 686, respectively; or (xxvii) 692 and 696, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA); (b) (i) 41 and 45, respectively; (ii) 181 and 185, respectively; (iii) 51 and 55, respectively; (iv) 191 and 195, respectively; (v) 280 and 284, respectively; (vi) 290 and 294, respectively; (vii) 300 and 304, respectively; (viii) 380 and 384, respectively; (ix) 390 and 394, respectively; (x) 400 and 404, respectively; or (xi) 420 and 424, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd); (c) (i) 61 and 65, respectively; (ii) 71 and 75, respectively; (iii) 201 and 205, respectively; (iv)
360 and 364, respectively; or (v) 370 and 374, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA); (d) (i) 81 and 85, respectively; (ii) 91 and 95, respectively; (iii) 310 and 314, respectively; (iv) 320 and 324, respectively; (v) 330 and 334, respectively; (vi) 410 and 414, respectively; (vii) 702 and 706, respectively; (viii) 712 and 716, respectively; (ix) 722 and 726, respectively; (x) 732 and 736, respectively; (xi) 742 and 746, respectively; or (xii) 752 and 756, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA); (e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively; (iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or (g) (i) 340 and 344, respectively; or (ii) 350 and 354, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 202. An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, and optionally the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences, of one of the following antibodies: (a) SSC35; SSE73; SSA12; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; or SSF69, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA); (b) SSG20; SSF2; SSF11; SSF10; SSG50; SSG54; SSM47; SSM45; SSF52; SSG51; or SSG59, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd); (c) SSL5; SSL17; SSL35; SSG27; or SSG39, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA); (d) SSH3; SSF33; SSG44; SSG48; SSM37; SSF34; SSN2; SSN6; SSH8; SSM35; SSM36; or SSM40, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA); (e) SSE1; SSE158; SSAA8; SSAA26; SSAB5; SSAI3; SSAI8; SSAI2; SSAF1; SSAL2; or SSAM6; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) SSB18; SSB24; SSB54; SSB65; or SSB67; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or (g) SSH4; or
SSH7; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 203. The antibody or antigen-binding fragment of embodiment 201 or 202, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences are according to: (i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system; (vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
Embodiment 204. An antibody or antigen-binding fragment thereof comprising a CDRH3 and a CDRL3, wherein the CDRH3 and the CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (a) (i) 34 and 38, respectively; (ii) 124 and 128, respectively; (iii) 24 and 28, respectively; (iv) 134 and 138, respectively; (v) 144 and 148, respectively; (vi) 154 and 158, respectively; (vii) 164 and 168, respectively; (viii) 244 and 248, respectively; (ix) 174 and 178, respectively; (x) 525 and 529, respectively; (xi) 535 and 539, respectively; (xii) 545 and 549, respectively; (xiii) 555 and 559, respectively; (xiv) 565 and 569, respectively; (xv) 575 and 579, respectively; (xvi) 585 and 589, respectively; (xvii) 595 and 599, respectively; (xviii) 605 and 609, respectively; (xix) 615 and 619, respectively; (xx) 625 and 629, respectively; (xxi) 635 and 639, respectively; (xxii) 645 and 649, respectively; (xxiii) 655 and 659, respectively; (xxiv) 665 and 669, respectively; (xxv) 675 and 679, respectively; (xxvi) 685 and 689, respectively; or (xxvii) 695 and 699, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA); (b) (i) 44 and 48, respectively; (ii) 184 and 188, respectively; (iii) 54 and 58, respectively; (iv) 194 and 198, respectively; (v) 283 and 287, respectively; (vi) 293 and 297, respectively; (vii) 303 and 307, respectively; (viii) 383 and 387, respectively; (ix) 393 and 397, respectively; (x) 403 and 407, respectively; or (xi) 423 and 427, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd); (c) (i) 64 and 68, respectively; (ii) 74 and 78, respectively; (iii) 204 and 208, respectively; (iv) 363 and 367, respectively; or (v) 373 and 377, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA); (d) (i) 84 and 88, respectively; (ii) 94 and 98, respectively; (iii) 313 and 317, respectively; (iv) 323 and 327, respectively; (v) 333 and 337, respectively; (vi) 413 and 417, respectively; (vii) 705 and
709, respectively; (viii) 715 and 719, respectively; (ix) 725 and 729, respectively; (x) 735 and 739, respectively; (xi) 745 and 749, respectively; or (xii) 755 and 759, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a. Staphylococcus Protein A (SpA); (e) (i) 4 and 8, respectively; (ii) 14 and 18, respectively; (iii) 435 and 439, respectively; (iv) 445 and 449, respectively; (v) 455 and 459, respectively; (vi) 465 and 469, respectively; (vii) 475 and 479, respectively; (viii) 485 and 489, respectively; (ix) 495 and 499, respectively; (x) 505 and 509, respectively; or (xi) 515 and 519, respectively; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) (i) 214 and 218, respectively; (ii) 104 and 108, respectively; (iii) 224 and 228, respectively; (iv) 114 and 118, respectively; or (v) 234 and 238, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or (g) (i) 343 and 347, respectively; or (ii) 353 and 357, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 205. An antibody, or an antigen-binding fragment thereof, comprising a complementarity determining region (CDR)H1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.: (a) (i) 32-34 and 36-38, respectively; (ii) 122-124 and 126-128, respectively; (iii) 22-24 and 26-28, respectively; (iv) 132-134 and 136-138, respectively; (v) 142-144 and 146-148, respectively; (vi) 152-154 and 156-158, respectively; (vii) 162-164 and 166-168, respectively; (viii) 242-244 and 246-248, respectively; (ix) 172-174 and 176- 178, respectively; (x) 523-525 and 527-529, respectively; (xi) 533-535 and 537-539, respectively; (xii) 543-545 and 547-549, respectively; (xiii) 553-555 and 557-559, respectively; (xiv) 563-565 and 567-569, respectively; (xv) 573-575 and 577-579, respectively; (xvi) 583-585 and 587-589, respectively; (xvii) 593-595 and 597-599, respectively; (xviii) 603-605 and 607-609, respectively; (xix) 613-615 and 617- 619, respectively; (xx) 623-625 and 627-629, respectively; (xxi) 633-635 and 637-639, respectively; (xxii) 643-645 and 647-649, respectively; (xxiii) 653-655 and 657-659, respectively; (xxiv) 663-665 and 667-669, respectively; (xxv) 673-675 and 677-679, respectively; (xxvi) 683-685 and 687-689, respectively; or (xxvii) 693-695 and 697-699, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA); (b) (i) 42-44 and 46-48, respectively; (ii) 182-184 and 186-188, respectively; (iii) 52-54 and 56-58, respectively; (iv) 192-194 and 196-198, respectively; (v) 281-283 and 285-287, respectively; (vi) 291-293 and 295-297, respectively; (vii) 301 - 303 and 305-307, respectively; (viii) 381-383 and 385-387, respectively; (ix) 391-393 and 395-397, respectively; (x) 401-403 and 405-407, respectively; or (xi) 421-423 and 425-427, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd); (c) (i) 62-64
and 66-68, respectively; (ii) 72-74 and 76-78, respectively; (iii) 202-204 and 206-208, respectively; (iv) 361-363 and 365-367, respectively; or (v) 371-373 and 375-377, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA); (d) (i) 82-84 and 86-88, respectively; (ii) 92-94 and 96-98, respectively; (iii) 311-313 and 315-317, respectively; (iv) 321-323 and 325-327, respectively; (v) 331-333 and 335-337, respectively; (vi) 411-413 and 415-417, respectively; (vii) 703-705 and 707-709, respectively; (viii) 713-715 and 717-719, respectively; (ix) 723-725 and 727- 729, respectively; (x) 733-735 and 737-739, respectively; (xi) 743-745 and 747-749, respectively; or (xii) 753-755 and 757-759, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA); (e) (i) 2-4 and 6-8, respectively; (ii) 12-14 and 16-18, respectively; (iii) 433-435 and 437-439, respectively; (iv) 443-445 and 447-449, respectively; (v) 453- 455 and 457-459, respectively; (vi) 463-465 and 467-469, respectively; (vii) 473-475 and 477-479, respectively; (viii) 483-485 and 487-489, respectively; (ix) 493-495 and 497-499, respectively; (x) 503- 505 and 507-509, respectively; or (xi) 513-515 and 517-519, respectively; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) (i) 212-214 and 216-218, respectively; (ii) 102-104 and 106-108, respectively; (iii) 222-224 and 226-228, respectively; (iv) 112-114 and 116-118, respectively; or (v) 232-234 and 236-238, respectively; wherein the antibody or antigenbinding fragment is capable of binding to a leucocidin and/or gamma toxin; or (g) (i) 341-343 and 345- 347, respectively; or (ii) 351-353 and 355-357, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 206. The antibody or antigen-binding fragment of any one of embodiments 201- 205, comprising a heavy chain variable domain framework region (VHFR)1, a VHFR2, a VHFR3, a light chain variable domain framework region (VLFR)1, a VLFR2, a VLFR3, and/or a VLFR4 (or a variant of the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4 comprising one, two, three, four, or five substitutions, insertions, and/or deletions, or a variant having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4, respectively) of the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101
and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65, respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively.
Embodiment 207. The antibody or antigen-binding fragment of embodiment 206, wherein the framework region or regions are according to: (i)the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system; (vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
Embodiment 208. The antibody or antigen-binding fragment of any one of embodiments 201- 207, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL
amino acid sequences set forth in SEQ ID NOs.: (a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532 and 536, respectively; (xii) 542 and 546, respectively; (xiii) 552 and 556, respectively; (xiv) 562 and 566, respectively; (xv) 572 and 576, respectively; (xvi) 582 and 586, respectively; (xvii) 592 and 596, respectively; (xviii) 602 and 606, respectively; (xix) 612 and 616, respectively; (xx) 622 and 626, respectively; (xxi) 632 and 636, respectively; (xxii) 642 and 646, respectively; (xxiii) 652 and 656, respectively; (xxiv) 662 and 666, respectively; (xxv) 672 and 676, respectively; (xxvi) 682 and 686, respectively; or (xxvii) 692 and 696, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA); (b) (i) 41 and 45, respectively; (ii) 181 and 185, respectively; (iii) 51 and 55, respectively; (iv) 191 and 195, respectively; (v) 280 and 284, respectively; (vi) 290 and 294, respectively; (vii) 300 and 304, respectively; (viii) 380 and 384, respectively; (ix) 390 and 394, respectively; (x) 400 and 404, respectively; or (xi) 420 and 424, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd); (c) (i) 61 and 65, respectively; (ii) 71 and 75, respectively; (iii) 201 and 205, respectively; (iv) 360 and 364, respectively; or (v) 370 and 374, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA); (d) (i) 81 and 85, respectively; (ii) 91 and 95, respectively; (iii) 310 and 314, respectively; (iv) 320 and 324, respectively; (v) 330 and 334, respectively; (vi) 410 and 414, respectively; (vii) 702 and 706, respectively; (viii) 712 and 716, respectively; (ix) 722 and 726, respectively; (x) 732 and 736, respectively; (xi) 742 and 746, respectively; or (xii) 752 and 756, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a. Staphylococcus Protein A (SpA); (e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively; (iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin; (f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or (g) (i) 340 and 344, respectively; or (ii) 350 and 354, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
Embodiment 209. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a LTA, wherein,
optionally, the antibody or antigen-binding binding fragment is capable of binding to: (i) a Type I LTA; (ii) a Type II LTA; (iii) a Type IV LTA; or (iv) any combination of (i)-(iii).
Embodiment 210. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding binding fragment is capable of binding to a Gmd, wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a .S'. aureus, to .S'. epide rmidis. or to .S' aureus and to .S' epidermidis .
Embodiment 211. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a ClfA.
Embodiment 212. The antibody or antigen-binding fragment of embodiment 211, wherein the antibody or antigen-binding binding is capable of binding to a ClfA OOl, a ClfA_002, and/or a ClfA_004.
Embodiment 213. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA), wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a SPAKKAA-
Embodiment 214. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin, wherein, optionally, the alpha toxin comprises a-hemolysin (also called Hla).
Embodiment 215. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody or antigen-binding fragment is capable of binding to a gamma toxin.
Embodiment 216. The antibody or antigen-binding fragment of any one of embodiments 201-208 and 215, wherein the antibody or antigen-binding fragment is capable of binding to a leukocidin, wherein, optionally, the leukocidin comprises a LukD, a LukE, a LukF, a LukS, or any combination thereof.
Embodiment 217. The antibody or antigen-binding fragment of embodiment 216, wherein the antibody or antigen-binding fragment is capable of binding to a LukED, a LukSF, or to a LukED and a LukSF.
Embodiment 218. The antibody or antigen-binding fragment of any one of embodiments 201-208 and 215-217, wherein the antibody or antigen-binding fragment is capable of binding to a HIgA, a HIgB, or both.
Embodiment 219. The antibody or antigen-binding fragment of any one of embodiments 201- 208, wherein the antibody is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin.).
Embodiment 220. The antibody or antigen-binding fragment of any one of embodiments 201- 219, wherein the antibody or antigen-binding fragment is capable of binding to any one, any two, any
three, any four, any five, any six, any seven, any eight, any nine, any ten, any eleven, or all twelve of: .S'. aureus FPR3757; .S'. aureus NE284 TE2 mut; .S' epidermidis RP62A; .S' epidermidis 1200; .S' epidermidis NIH04008; .S' lugdunensis, S. pyogenes^ E.faecalis', S. agalactiae S. hominis,' L. garvieae, and .S' pneumonia.
Embodiment 221. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
Embodiment 222. The anti -LTA antibody or antigen-binding fragment of embodiment 221, wherein the CDRS are according to: (i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences); (ii) the Kabat numbering system; (iii) the Chothia numbering system; (iv) the Enhanced Chothia (also referred to as “Martin”) numbering system; (v) the AHo numbering system; (vi) the North numbering system; (vii) the Contact numbering system; (viii) the EU numbering system; (ix) the AbM numbering system; or (x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
Embodiment 223. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences set forth in SEQ ID NOs.:22-24 and 26-28, respectively.
Embodiment 24. The anti -LTA antibody or antigen-binding fragment of any one of embodiments 221-223, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
Embodiment 225. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising a heavy chain variable domain (VH) and a light chain variable domain (VL), wherein the VH and VL comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOs.:21 and 25, respectively.
Embodiment 226. The antibody or antigen-binding fragment of any one of embodiments 201- 225, wherein the antibody, or the antigen-binding fragment, comprises a human antibody, a monoclonal antibody, a purified antibody, a single chain antibody (e.g., scFv), a Fab, a Fab’, a F(ab’)2, or Fv.
Embodiment 227. The antibody or antigen-binding fragment of any one of embodiments 201- 226, wherein the antibody or antigen-binding fragment is a multi-specific antibody or antigen-binding fragment, wherein, optionally, the antibody or antigen-binding fragment is a bi-specific antibody or antigen-binding fragment.
Embodiment 228. The antibody or antigen-binding fragment of embodiment 227, which comprises (1) the six CDRS, and optionally the VH and VL, of a first antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1) and (2) the six CDRS, and optionally the VH and VL, of a second, different antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1).
Embodiment 229. The antibody or antigen-binding fragment of embodiment 227 or 228, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and a ClfA; a SpA and a ClfA; a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; or a Sbi and a ClfA.
Embodiment 230. The antibody or antigen-binding fragment of any one of embodiments 227-
229, comprising a first antigen-binding domain and second antigen-binding domain, wherein the first antigen-binding domain or the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; or (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSE52; (xxi) SSG51; (xxii) SSE34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC10; (xxxi) SSC15; (xxxii) SSE2; (xxxiii) SSE11; (xxxiv) SSE10; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSE33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAE1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE150; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSE69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
Embodiment 231. The antibody or antigen-binding fragment of any one of embodiments 227-
230, comprising a first antigen-binding domain and second antigen-binding domain, wherein the first
antigen-binding domain and the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S or M428L/N434S/FI435R/Y436F mutations.
Embodiment 232. An antibody, or an antigen-binding fragment thereof, which is a multi-specific antibody or antigen-binding fragment thereof, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and a ClfA; a SpA and a ClfA; a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; or a Sbi and a ClfA.
Embodiment 233. The antibody or antigen-binding fragment of any one of embodiments 227-
232, which comprises or is of a format selected from: a Bispecific T cell Engager (BiTE); a DART; a Knobs-Into-Holes (KIH) assembly; a scFv-CH3-KIH assembly; a KIH Common Light-Chain antibody; a TandAb; a Triple Body; a TriBi Minibody; a Fab-scFv; a scFv-CH-CL-scFv; a F(ab’)2-scFv2; a tetravalent Hcab; an intrabody; a CrossMab; a Dual Action Fab (DAF) (two-in-one or four-in-one); a DutaMab; a DT-IgG, a charge paired antibody; a Fab-arm Exchange antibody, a SEEDbody; a Triomab; a LUZ-Y assembly, an Fcab; a K/.-body: an orthogonal Fabs antibody; a DVD-Ig; am IgG(H)-scFv; an scFv-(H)IgG; an IgG(L)-scFv; an scFv-(L)IgG; an IgG(L,H)-Fv; an IgG(H)-V; a V(H)-IgG; an IgG(L)-V; a V(L)-IgG; a KIH IgG-scFab; a 2scFv-IgG; a IgG-2scFv; a scFv4-Ig; a Zybody; a DVI-IgG (four-in- one), a FIT-Ig; a WuxiBody ; and an In-Elbow-Insert Ig.
Embodiment 234. The antibody or antigen-binding fragment of any one of embodiments 201-
233, wherein the antibody or antigen-binding fragment comprises an (e.g., IgGl or IgG3) Fc polypeptide or a fragment thereof.
Embodiment 235. The antibody or antigen-binding fragment of any one of embodiments 201-
234, which comprises an IgG, IgA, IgM, IgE, or IgD isotype.
Embodiment 236. The antibody or antigen-binding fragment of any one of embodiments 201-
235, which comprises an IgG isotype selected from IgGl, IgG2, IgG3, and IgG4, optionally an IgGl
isotype comprising a heavy chain with a C-terminal lysine removed or a C-terminal glycine-lysine removed.
Embodiment 237. The antibody or antigen-binding fragment of any one of embodiments 201-
236, which comprises an IgGl isotype.
Embodiment 238. The antibody or antigen-binding fragment of any one of embodiments 201-
237, which comprises an IgGlm3 allotype, an IgGlml7 allotype, an IgGlml allotype, or any combination thereof.
Embodiment 239. The antibody or antigen-binding fragment of any one of embodiments 201- 236, which comprises an IgG3 isotype.
Embodiment 240. The antibody or antigen-binding fragment of any one of embodiments 234- 239, wherein the Fc polypeptide or fragment thereof comprises: (i) a mutation that increases binding affinity to a human FcRn (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols)), as compared to a reference Fc polypeptide that does not comprise the mutation; and/or (ii) a mutation that increases binding affinity to a human FcyR (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols, and/or as measured using mesoscale discovery (MSD))) as compared to a reference Fc polypeptide that does not comprise the mutation.
Embodiment 241. The antibody or antigen-binding fragment of embodiment 240, wherein the mutation that increases binding affinity to a human FcRn comprises: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q311I; D376V; T307A; E380A; or any combination thereof.
Embodiment 242. The antibody or antigen-binding fragment of embodiment 240 or 241, wherein the mutation that increases binding affinity to a human FcRn comprises: (i) M428L/N434S; (ii) M252Y/S254T/T256E; (iii) T250Q/M428L; (iv) P257I/Q31 II; (v) P257I/N434H; (vi) D376V/N434H; (vii) T307A/E380A/N434A; (viii) M428L/N434A; or (ix) any combination of (i)-(viii).
Embodiment 243. The antibody or antigen-binding fragment of any one of embodiments 240-
242, wherein the mutation that increases binding affinity to a human FcRn comprises M428L/N434S or M428L/N434A.
Embodiment 244. The antibody or antigen-binding fragment of any one of embodiments 240-
243, wherein the mutation that enhances binding to a FcyR comprises S239D; I332E; A330L; G236A; or any combination thereof.
Embodiment 245. The antibody or antigen-binding fragment of any one of embodiments 240-
244, wherein the mutation that enhances binding to a FcyR comprises: (i) S239D/I332E; (ii)
S239D/A330L/I332E; (iii) G236A/S239D/I332E; or (iv) G236A/A330L/I332E, wherein the Fc polypeptide or fragment thereof optionally comprises Ser at position 239.
Embodiment 246. The antibody or antigen-binding fragment of any one of embodiments 201-
245, which comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or which is aglycosylated, and/or which is afucosylated.
Embodiment 247. The antibody or antigen-binding fragment of any one of embodiments 201-
246, comprising in a heavy chain thereof, the amino acid mutation(s) set forth in any one of (i)-(xix): (i) G236A, L328V, and Q295E; (ii) G236A, P230A, and Q295E; (iii) G236A, R292P, and I377N; (iv) G236A, K334A, and Q295E; (v) G236S, R292P, and Y300L; (vi) G236A and Y300L; (vii) G236A, R292P, and Y300L; (viii) G236S, G420V, G446E, and L309T; (ix) G236A and R292P; (x) R292P and Y300L; (xi) G236A and R292P; (xii) Y300L; (xiii) E345K, G236S, L235Y, and S267E; (xiv) E272R, L309T, S219Y, and S267E; (xv) G236Y; (xvi) G236W; (xvii) F243L, G446E, P396L, and S267E; (xviii) G236A, S239D, and H268E; (xix) any one or more of the mutations shown in Figure 14A, Figure 14B, Figure 14C, Figure 14E, Figure 14F, Figure 14G, Figure 14H, Figure 141, Figure 14J, or Figure 14K.
Embodiment 248. The antibody or antigen-binding fragment of any one of embodiments 201-
247, comprising in a heavy chain thereof, one or more of the following the amino acid mutations: G236A; Y300L; R292P.
Embodiment 249. The antibody or antigen-binding fragment of any one of embodiments 201-
248, comprising, in a heavy chain thereof, an amino acid mutation that promotes complement activation, wherein, optionally, the amino acid mutation that promotes complement activation comprises or consists of: K236W/E333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L; G236A/Y300L; or G236A/R292P/Y300L.
Embodiment 250. The antibody or antigen-binding fragment of any one of embodiments 201-
249, comprising, in a heavy chain or a CH3 thereof, an amino acid mutation that reduces binding by Staphylococcus Protein A, wherein, the optionally, amino acid mutation that reduces binding by Staphylococcus Protein A comprises or consists of: H435R; Y436F; or H435R/Y436F.
Embodiment 251. The antibody or antigen-binding fragment of any one of embodiments 201-
250, comprising a human IgGl isotype and comprising, in a heavy chain or a CH3 thereof, the amino acid mutations M428L/N434S/H435R/Y436F.
Embodiment 252. The antibody or antigen-binding fragment of any one of embodiments 201-
251, which is capable of neutralizing an infection (in vitro, ex vivo, and/or in vivo) by a bacteria expressing the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi.
Embodiment 253. The antibody or antigen-binding fragment of any one of embodiments 201-
252, which is capable of neutralizing an .S'. aureus infection in vitro, ex vivo, or in vivo.
Embodiment 254. The antibody or antigen-binding fragment of any one of embodiments 201-
253, which is capable of opsonizing and/or is capable of promoting phagocytosis of a bacteria expressing the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, Sbi, or gamma toxin, wherein, optionally, the bacteria comprises .S', aureus.
Embodiment 255. The antibody or antigen-binding fragment of any one of embodiments 201-
254, which: is afucosylated; has been produced in a host cell that is incapable of fucosylation or that is inhibited in its ability to fucosylate a polypeptide; has been produced under conditions that inhibit fucosylation thereof by a host cell; or any combination thereof.
Embodiment 256. The antibody or antigen-binding fragment of any one of embodiments 201-
255, which is human, humanized, or chimeric.
Embodiment 257. An antibody, or an antigen-binding fragment, that is capable of binding to an .S', aureus antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi, wherein, optionally:
(i) the antibody or antigen-binding fragment is human, humanized, or chimeric;
(ii) the antibody or antigen-binding fragment binds to a LTA and does not complete with pagibaximab for binding to the LTA;
(iii) the antibody or antigen-binding fragment comprises a VH and a VL that comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.:
(a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively;
(iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532 and 536, respectively; (xii) 542 and 546, respectively; (xiii) 552 and 556, respectively; (xiv) 562 and 566, respectively; (xv) 572 and 576, respectively; (xvi) 582 and 586, respectively; (xvii) 592 and 596, respectively; (xviii) 602 and 606, respectively; (xix) 612 and 616, respectively; (xx) 622 and 626, respectively; (xxi) 632 and 636, respectively; (xxii) 642 and 646,
respectively; (xxiii) 652 and 656, respectively; (xxiv) 662 and 666, respectively; (xxv) 672 and 676, respectively; (xxvi) 682 and 686, respectively; or (xxvii) 692 and 696, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA);
(b) (i) 41 and 45, respectively; (ii) 181 and 185, respectively; (iii) 51 and 55, respectively;
(iv) 191 and 195, respectively; (v) 280 and 284, respectively; (vi) 290 and 294, respectively; (vii) 300 and 304, respectively; (viii) 380 and 384, respectively; (ix) 390 and 394, respectively; (x) 400 and 404, respectively; or (xi) 420 and 424, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) (i) 61 and 65, respectively; (ii) 71 and 75, respectively; (iii) 201 and 205, respectively; (iv) 360 and 364, respectively; or (v) 370 and 374, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA);
(d) (i) 81 and 85, respectively; (ii) 91 and 95, respectively; (iii) 310 and 314, respectively;
(iv) 320 and 324, respectively; (v) 330 and 334, respectively; (vi) 410 and 414, respectively; (vii) 702 and 706, respectively; (viii) 712 and 716, respectively; (ix) 722 and 726, respectively; (x) 732 and 736, respectively; (xi) 742 and 746, respectively; or (xii) 752 and 756, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively;
(iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) (i) 340 and 344, respectively; or (ii) 350 and 354, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin); and/or
(iv) the antibody or antigen-binding fragment comprises an Fc polypeptide or fragment thereof, or a heavy chain, according to any one of embodiments 234-251.
Embodiment 258. An isolated polynucleotide encoding the antibody or antigen-binding fragment of any one of embodiments 201-257, or encoding a VH, a Fd, a heavy chain, a VL, and/or a light chain of the antibody or the antigen-binding fragment.
Embodiment 259. The polynucleotide of embodiment 258, wherein the polynucleotide comprises deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), wherein the RNA optionally comprises messenger RNA (mRNA).
Embodiment 260. The polynucleotide of embodiment 259, wherein the polynucleotide comprises self-amplifying RNA (saRNA), trans-amplifying RNA (taRNA), or circular RNA (circRNA).
Embodiment 261. The polynucleotide of any one of embodiments 258-260, comprising a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof, wherein, optionally, the polynucleotide comprises a pseudouridine, a N6-methyladenonsine, a 5 -methylcytidine, a 2- thiouridine, or any combination thereof, wherein, further optionally, the pseudouridine comprises Nl- methylpseudouridine .
Embodiment 262. The polynucleotide of any one of embodiments 258-261, which is codon- optimized for expression in a host cell.
Embodiment 263. The polynucleotide of embodiment 262, wherein the host cell comprises a human cell or a CHO cell.
Embodiment 264. The polynucleotide of embodiment 262, wherein the host cell is a production host cell.
Embodiment 265. The polynucleotide of embodiment 262, wherein the host cell is an in vivo human host cell.
Embodiment 266. A recombinant vector, DNA therapeutic, or RNA therapeutic comprising the polynucleotide of any one of embodiments 258-265.
Embodiment 267. A DNA therapeutic construct or RNA therapeutic construct comprising the polynucleotide of any one of embodiments 258-265 encapsulated in a carrier, wherein the carrier optionally comprises a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
Embodiment 268. A recombinant vector comprising the polynucleotide of any one of embodiments 258-265.
Embodiment 269. A host cell comprising the polynucleotide of any one of embodiments 258-265 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, wherein the
polynucleotide is optionally heterologous to the host cell and/or wherein the host cell is capable of expressing the encoded antibody or antigen-binding fragment or polypeptide.
Embodiment 270. An isolated human B cell comprising the polynucleotide of any one of embodiments 258-265 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, wherein polynucleotide is optionally heterologous to the human B cell and/or wherein the human B cell is immortalized.
Embodiment 271. A composition or combination comprising: (i) the antibody or antigen-binding fragment of any one of embodiments 201-257; (ii) the polynucleotide of any one of embodiments 258- 265; (iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266- 268; (iv) the host cell of embodiment 269; and/or (v) the human B cell of embodiment 270, and, optionally, a pharmaceutically acceptable excipient, carrier, or diluent.
Embodiment 272. The composition or combination of embodiment 271, which comprises (1) a first antibody or antigen-binding fragment of any one of embodiments 201-257 and (2) a second, different antibody or antigen-binding fragment of any one of embodiments 201-257.
Embodiment 273. The composition or combination of embodiment 272, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are capable of binding to: an LTA and a Gmd, respectively; an LTA and an alpha toxin, respectively; an LTA and a gamma toxin, respectively; an LTA and a leucocidin, respectively; an LTA and a SpA, respectively; an LTA and a ClfA, respectively; an alpha toxin and a Gmd, respectively; an alpha toxin and a gamma toxin, respectively; an alpha toxin and a leucocidin, respectively; an alpha toxin and a SpA, respectively; an alpha toxin and a ClfA, respectively; a gamma toxin and a Gmd, respectively; a gamma toxin and a leucocidin, respectively; a gamma toxin and a SpA, respectively; a gamma toxin and a ClfA, respectively; a leucocidin and a SpA, respectively; a leucodin and a Gmd, respectively; a leucocidin and a ClfA, respectively; a Gmd and a ClfA, respectively; a SpA and a ClfA, respectively; a Sbi and a LTA, respectively; a Sbi and a Gmd, respectively; a Sbi and an alpha toxin, respectively; a Sbi and a gamma toxin, respectively; a Sbi and a leucocidin, respectively; a Sbi and a SpA, respectively; or a Sbi and a SlfA, respectively.
Embodiment 274. The composition or combination of any one of embodiments 271-273, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment or the second antibody or antigenbinding fragment comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7;
(xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC1O; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF1O; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE15O; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
Embodiment 275. The composition or combination of any one of embodiments 271-274, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment and the second antibody or antigenbinding fragment comprise the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S/H435R/Y436F mutations.
Embodiment 276. The composition or combination of embodiment 271, comprising a first polynucleotide and a second polynucleotide, each according to any one of embodiments 258-265, or a recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, encoding two or more antibodies or antigen-binding fragments, wherein the first antibody or antigen-binding fragment encoded by the first polypeptide and second antibody or antigen-binding fragment encoded by the second polypeptide, respectively, or the multispecific antibody or antigen-binding fragment, comprise(s) CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3, and optionally VH and VL, according to any two antibodies of the present disclosure, or any antigen-binding fragments thereof.
Embodiment 277. A composition comprising the polynucleotide of any one of embodiments 258- 265 or the vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268 encapsulated in a carrier molecule, wherein the carrier molecule optionally comprises a lipid, a lipidderived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
Embodiment 278. A(n, e.g. , in vitro or ex vivo) method of making an antibody or antigen-binding fragment of any one of embodiments 201-257, comprising culturing the host cell of embodiment 269 or the human B cell of embodiment 270 for a time and under conditions sufficient for the host cell or human B cell, respectively, to express the antibody or antigen-binding fragment.
Embodiment 279. The method of making an antibody or antigen-binding fragment of embodiment 278, wherein the host cell of embodiment 269 or the human B cell of embodiment 270 comprises the polynucleotide of any one of embodiment 258-265 and/or recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268.
Embodiment 280. The method of embodiment 278 or 279, further comprising isolating the antibody or antigen-binding fragment.
Embodiment 281. A method of treating or preventing a bacterial infection in a(n, e.g. , human) subject, wherein the bacterial infection is optionally bacteremia, wherein further optionally the bacteremia is hemodialysis associated/line associated bacteremia, the method comprising administering to the subject an effective amount of: (i) the antibody or antigen-binding fragment of any one of embodiments 201-257;
(11) the polynucleotide of any one of embodiments 258-265; (iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268; (iv) the host cell of embodiment 269; (v) the human B cell of embodiment 270; and/or (vi) the composition or combination of any one of embodiments 271-277, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S', aureus.
Embodiment 282. The method of embodiment 281, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'. Aureus (MRSA); (2) a Coagulase-negative Staphylococcus (CoNS); (3) .S', epidermidis,' (4) E. faecalis,' (5) .S', pyogenes,' (6) .S'. agalactiae,' (7) L. garviae,' (8) .S', pneumoniae,' (9) .S', lugdunensis,' (10) .S', hominis,' (11) .S', epidermidis,' or
(12) any combination of (l)-(l 1).
Embodiment 283. The method of embodiment 281 or 282, comprising administering a single dose of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
Embodiment 284. The method of any one of embodiments 281-283, comprising administering two or more doses of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
Embodiment 285. The method of any one of embodiments 281-284, comprising administering the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition intramuscularly, subcutaneously, or intravenously.
Embodiment 286. The method of any one of embodiments 281-285, wherein the treatment and/or prevention comprises pre-exposure or post-exposure prophylaxis.
Embodiment 287. The method of any one of embodiments 281-286, wherein the subject: is using a ventilator; is undergoing, will undergo, or has recently undergone surgery; has a blood stream infection or a history of blood stream infections; is receiving, will receive, or has received a prosthetic joint or other implant; is undergoing, will undergo, or has undergone dialysis; has bacteremia or a history of bacteremia; has neutropenia or a history of neutropenia; has hemodialysis associated/line associated bacteremia or a history of hemodialysis associated/line associated bacteremia; has a wound; or any combination of the foregoing.
Embodiment 288. The method of any one of embodiments 281-287, further comprising administering to the subject or more antibiotic agent, or wherein the subject has received one or more antibiotic agent.
Embodiment 289. The method of embodiment 288, wherein the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof.
Embodiment 290. The method of embodiment 288 or 289, wherein the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin-clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof.
Embodiment 291. The antibody or antigen-binding fragment of any one of embodiments 201- 257, the polynucleotide of any one of embodiments 258-265, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, the host cell of embodiment 269, the human B cell of embodiment 270, and/or the composition or combination of any one of embodiments 271-277, for use in a method of treating or preventing a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S', aureus.
Embodiment 292. The antibody or antigen-binding fragment of any one of embodiments 201- 257, the polynucleotide of any one of embodiments 258-265, the recombinant vector, DNA therapeutic, RNA or therapeutic of any one of embodiments 266-268, the host cell of embodiment 269, the human B cell of embodiment 270, and/or the composition or combination of any one of embodiments 271-277, for use in the preparation of a medicament for the treatment or prevention of a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
Embodiment 293. A method for in vitro diagnosis of a bacterial infection, the method comprising: (i) contacting a sample from a subject with an antibody or antigen-binding fragment of any one of embodiments 201-257; and (ii) detecting a complex comprising an antigen and the antibody, or comprising an antigen and the antigen-binding fragment, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises .S'. aureus.
Embodiment 294. The antibody, antigen-binding fragment, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, human B cell, or composition for use of embodiment 291 or 292, or the method of embodiment 293, wherein wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) .S', aureus, wherein, further optionally, the .S', aureus comprises a methicillin-resistant .S'. Aureus (MRSA); (2) a Coagulase -negative Staphylococcus (CoNS); (3) .S', epidermidis,' (4) E. faecalis,' (5) .S', pyogenes,' (6) .S', agalactiae,' (7) L. garviae,' (8) .S', pneumoniae,' (9) .S', lugdunensis,' (10) .S', hominis,' (11) .S', epidermidis,' or (12) any combination of (l)-(l 1).
Embodiment 295. A library comprising a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprising a plurality of polynucleotides encoding amino acid sequences encoded by any one or more of the following human immunoglobulin genes: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3-15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2-14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-
1; IGHV1-24; IGKV1-16; IGKV3-20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof, preferably (1) IGHV3-23/IGHJ3, (2) IGHV3-7/IGVK1-6, or (3) IGHV3-23.
Embodiment 296. The library of embodiment 295, wherein the human immunoglobulin genes in the library consist essentially of, or consist of, any one or more of the human immunoglobulin genes of embodiment 295.
Embodiment 297. The library of embodiment 295 or 296, wherein the library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences from any one or more of (i)-(xxiii): (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3- 23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3- 21, and IGLJ2; (xiv) IGHV5-51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1-39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1-69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3; (xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
Embodiment 298. The library of embodiment 297, wherein the plurality of antibodies or antigenbinding fragments, or the plurality of polynucleotides encoding the antibodies or antigen-binding fragments, further comprises or encodes additional amino acid sequences, wherein the additional amino acid sequences are optionally randomized and/or are limited to one or more CDR regions of an antibody or antigen-binding fragment.
Embodiment 299. A method comprising screening one or more antibodies of the library of any one of embodiments 104-107 for binding to a bacterial (e.g. , .S', aureus) antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi.
Embodiment 300. A kit comprising a liquid composition comprising and antibody or antigenbinding fragment of any one of embodiments 201-257, 291, 292, or 294, the polynucleotide of any one of embodiments 258-265, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of embodiments 266-268, the host cell of embodiment 269, the human B cell of embodiment 270, and/or the composition of any one of embodiment 271-277, and instructions for use thereof in treating a bacterial (e.g., Staphylococcus) infection in a subject.
EXAMPLES
EXAMPLE 1
IDENTIFICATION AND TESTING OF ANTI-STAPHYLOCOCCUS ANTIBODIES
Monoclonal antibodies (mAbs) reactive against one or more Staphylococcus antigen were identified from a human tonsil bank and from PBMCs from human donors recovered from S. aureus, as shown and described for Figures 1A-1D. MAbs were cloned, recombinantly expressed (in some contexts, carrying one or more Fc mutation), and characterized for binding specificity, neutralization, and other functions, as shown and described for Figures 2A-6C.
EXAMPLE 2
ADDITIONAL STUDIES or Staphylococcus, opsonizing antibodies include anti-LTA antibodies, anti-Gmd antibodies, anti-ClfA antibodies, and anti-SpA antibodies. Neutralizing antibodies include anti- Hla antibodies and anti-bicomponent (Luk, HIgAB) antibodies.
For anti-LTA antibodies, further experiments assess breadth of binding to live bacteria (FACS) and epitope-binning. For anti-Gmd antibodies, further experiments assess breadth of binding to different strains of Staphylococcus, epitope-binning, and effect on Gmd enzymatic activity. For anti-ClfA antibodies, opsonophagocytic killing and fibrinogen inhibition of binding are assessed. For anti-Hla antibodies, synergism of Hla neutralization and neutralization on THP1 cells are assessed. For anti-bicomponent toxins, neutralization studies are performed.
Human whole blood assays and in vivo mouse models are used for further studies.
Human whole blood (received within 1-2 hours of draw from donor) is used in an assay where exponential-phase S. aureus cells are mixed with mAbs and added to whole blood. After incubation, human cells are lysed, and the sample is plated for S. aureus survival. Briefly, prior work has shown that results from whole blood assays using S. aureus wild-type strain MW2 is predictive of efficacy in vivo (Chen et al. PNAS 779(4):e2114478119 (2022); doi:
10.1073/pnas.2114478119). Mice are used to study bacteremia, pneumonia, dermonecrosis, a prosthetic joint infection model, a foreign body model, and a surgical site infection model. In a foreign body model, beads coated with 500 CFU of S. aureus are injected into the skin, representative of a real-life infection dose. S. aureus introduced on a foreign object is not cleared as easily as suspension cells and presents as biofilm-like. In a model of prosthetic joint infection, a surgically placed stainless steel K-wire implant can be inserted into the knee joints of mice and inoculated with S. aureus to model a prosthetic joint infection.
EXAMPLE 3 TESTING OF ANTI-HLA ANTIBODIES
The following experiments were performed using anti-Hla antibodies SSE1 and SSE158. Figure 7A shows results from SSE1 and SSE158 binding and neutralization studies, with MEDI4893 included as a comparator. SSE1 and SSE158 bind to H1A and neutralize H1A activity on rabbit red blood cells (“RBCs”) and THP-1 cells.
Figure 7B shows results (neutralization of infection) from a synergy/antagonism study with SSE1 and SSE158. SSE1 and SSE158 did not show synergistic neutralization in this assay.
Figure 7C shows that SSE1 and SSE158 protect BALB/c mice in an in vivo model of S. aureus pneumonia. The experimental set-up was as shown in the schematic in the bottom of Figure 7C. Percent survival was measured post-infection.
EXAMPLE 4
TESTING OF ANTI-GMD ANTIBODIES
The following experiments were performed using certain anti-Gmd antibodies of the present disclosure.
Figures 8A-8B show binding (ELISA) to Gmd S. aureus and Gmd S. epidermis, with 1C11 included as a comparator. Figure 8C shows quantified binding EC50 values to Gmd S. aureus and Gmd S. epidermis,' to strains of S. aureus, S. epidermidis, and S. lugdunensis,' and V- domain gene usage of the indicated antibodies.
EXAMPLE 5
NEUTRALIZATION OF LTA ACTIVITY
Figures 9A-9B relate to neutralization of LTA activity on TLR-2-expressing cells by certain antibodies of the present disclosure. Briefly, HEK-Blue hTLR2 cells were co-transfected with hTLR2 and SEAP (secreted embryonic alkaline phosphatase) reporter genes under the control of the IFN-b minimal promoter fused to NF-kB and AP-l-binding sites. Stimulation of TLR2 activates NF-kB and AP-1 which induces the production of SEAP. The hydrolysis of the substrate in the medium by SEAP produces an easily detected purple/blue color.
EXAMPLE 6
TESTING OF ANTI-CLFA ANTIBODIES
The following experiments were performed using certain anti-ClfA antibodies of the present disclosure. Figure 10A shows binding (ELISA) by certain antibodies of the present disclosure to strains of S. aureus with different ClfA genotypes (ClfA_001, ClfA_002, and ClfA_004), with E. coli included as a negative control. Figure 10B shows quantified binding EC50 values to ClfA_001, ClfA_002, ClfA_004, and strains of S. aureus,' binding affinity values (Kato ClfA OOl); inhibition of ClfA binding to fibrinogen (IC50); and V-domain gene usage of certain antibodies of the present disclosure. Tefibazumab was included as a comparator. Figure 10C shows inhibition of binding between ClfA and fibrinogen (Fg) by certain antibodies of the present disclosure, with tefibazumab included as a comparator. Figure 10D shows sequence alignments between ClfA_001, ClfA_002, and ClfA_004 genotypes and a schematic of a representative recombinant ClfA expression construct. Figure 10E shows inhibition of binding between ClfA and Fg by certain antibodies of the present disclosure. Briefly, antibody inhibition
of ClfA and Fg binding was assessed using the following method: Fg was coated in PBS; a blocking agent was applied (1% Casein in PBS); a mix of recombinantly expressed ClfA OOl, ClfA_002, and ClfA_004 polypeptides comprising a series of tag peptides and linkers for detection/purification (see Figures IOC and 10D) and anti-ClfA antibodies were added; and binding between Clfa and Fg was monitored by development of “Strep-Tactin AP”. S2X303 was included as an isotype control and tefibazumab was included as a comparator. Figure 10F shows antibodies grouped according to like binding profiles (Figure 10B).
EXAMPLE 7
TESTING OF ANTI-SPA ANTIBODIES
The following experiments were performed using certain anti-SpA antibodies of the present disclosure. Figure 11A shows binding (EC50 as determined by ELISA) to SpA-KKAA, percent inhibition of SpA binding to IgG, binding affinity values of antibodies of the present disclosure to Spa-KKAA (Ka), V-domain gene usage, and epitope binning of certain antibodies of the present disclosure. Figure 11B shows neutralization of binding between SpA and IgGl by the indicated antibodies of the present disclosure (Octet). Figure 11B shows antibody neutralization of Protein A binding of IgGs assessed using BLI (Octet).
EXAMPLE 8
WHOLE BLOOD ASSAYS
Exponential-phase bacteria were mixed with mAbs and added to whole blood from a human donor. After incubation, human cells were lysed, and the sample was plated to evaluate bacterial survival. See Figure 12A. Prior work has shown that results from whole blood assays using S. aureus wild-type strain MW2 is predictive of efficacy in vivo (Chen et al. PNAS 779(4):e2114478119 (2022); doi: 10.1073/pnas.2114478119). Human whole blood assays were performed to test the effect of certain antibodies of the present disclosure on S. aureus (Figures 12B and 14J) and S. epidermidis (Figures 14H) survival.
EXAMPLE 9
Fc MODIFICATIONS
The effects of additional Fc mutations and/or production under conditions that inhibit antibody glycosylation were evaluated in SSA12-LS-RF. (See Figures 14A-14K). Numbering of
the Fc mutations indicated in Figures 14A-14K is per the EU numbering system with reference to Human IgGl. The effects of the Fc modifications on in vitro FcyR and Cl binding were measured by SPR, MSD, BLI, and ELISA. The effects of the additional Fc modifications on SSA12-LS-RF were also measured by human whole blood assays. Fc-variant-antibody binding to human Cl with S. aureus and mouse Clq with S. aureus is compared in Figures 15A-15B.
EXAMPLE 10
IN VIVO STUDIES
In vivo studies are performed to assess efficacy of antibodies against different opsonizing targets in an i.v. infection model in mice. Briefly, C57BL/6 mice (n = 5 per group) receive different doses of antibody i.v. two days before S. aureus i.v. At day 4 post- aureus, mice are sacrificed (30% weight-loss cutoff) and kidneys are harvested to assess colony-forming units. The mice are appropriately matched for prevention in dialysis in humans.
EXAMPLE 11
TESTING OF ANTI-SBI ANTIBODIES
The following experiments were using certain anti-Sbi antibodies of the present disclosure. A variant of Sbi (SbiKK) containing substitution mutations (Q9K, Q10K, D36A, D37A, in each of five its Ig-binding domains) and lacking IgG-Fc binding was used for selection and characterization of anti-Sbi antibodies. Figure 18A shows a schematic of Sbi structure.
Figure 18B shows (top) binding quantified by ELISA and (bottom) quantified binding values (EC50), percent IgG-binding inhibition, binding affinity (KD), and V-domain gene usage of SSH4 and SSH7. Figure 18C shows sensorgram curves showing that antibodies SSH4 and SSH7 of the present disclosure do not compete with one another for Sbi binding.
Results from additional assays are shown in Figures 23 and 24.
EXAMPLE 12
COMPARING MABS AGAINST DIFFERENT STAPHYLOCOCCUS TARGETS IN FPR3757 BACTEREMIA MODEL IN BL6 MICE
To determine the level of protection provided by anti-Staphylococcal antibodies in a disseminated infection model, anti-LTA, anti-ClfA, anti-SpA, and anti-Gmd mAbs were administered to 8-week-old female C57BL/6 mice at 10 mg/kg intravenously 2 days prior to challenge with 2xlOA7 cfu of S. aureus strain FPR3757 via retroorbital injection (Figure 25). Mice were then monitored for weight loss and kidney burden was determined 4 days post infection by organ homogenization and serial dilution. As a control, one group was treated with daptomycin (50 mg/kg subcutaneously) at days 2 and 3 post infection to compare the relative weight reduction and kidney burden of antibiotics in a treatment model. Additional control groups included mice dosed with non-binding isotype control mAbs and a histidine buffer group. Mouse serum Ig level was measured at day 1 post-dosing using meso scale discovery (goat antihuman IgG as capture antibody, sulfo tag anti-human CH2 as detection antibody; Figure 26). 514G3 RF antibody had 2.3-fold lower serum Ig level compared to isotype control, similar to results for 514G3 IgG3 antibody format. SSF11 antibody had 3-folder lower serum Ig level compared to isotype control. SSC35 antibody had 1.7-fold higher serum Ig level compared to isotype control. Figures 27A-27B show kidney burden at 4 days post infection for individual mice (Figure 27 A), weight loss over time and the weight loss calculated from the area under the curve (Figure 27B). Mice treated with isotype control antibodies had uniformly high kidney burden with pronounced weight loss, while daptomycin treated mice showed a 1-3 log reduction in S. aureus kidney burden associated with a minor reduction in weight loss. Individual mice treated with anti-Staphylococcal antibodies varied from no reduction to 6 log reduction in kidney burden (Figure 28). Mice dosed with select anti-Staphylococcal mAbs had kidney burden below the limit of detection in 2/5 (anti-LTA SSA8 and anti-ClfA SSL5) and 3/5 mice (anti-ClfA SSG27). Weight loss was reduced in many of the mice treated with n -Staphylococcal antibodies. Largely, mice with reduced weight loss correlated with mice showing a reduction in
kidney burden. Anti-ClfA mAb SSG27 and tefibazumab dosed mice demonstrated the most consistent reduction in weight loss across the groups tested.
Together, these data show that select anh-Staphylococcal antibodies administered prior to infection can reduce the kidney burden and weight loss associated with S. aureus infection. Among anti-LTA antibodies, more mice treated with SSA8 antibody cleared infection compared to Pagibaximab and other anti-LTA mAbs. Among anti-ClfA antibodies, more mice treated with SSG27 antibody cleared infection and lost less weight compared to tefibazumab and other anti- LTA antibodies. Mice receiving tefibazumab lost the least weight. No difference was observed between SSH3 antibody and 514G3 IgG3 antibody; 514G3 IgG3 antibody showed a trend of less weight loss. No difference was observed between SSF11 antibody and 1C11 antibody, consistent with a previous study. Combination of SSG27 antibody + SSE1 antibody (10 mpk of each) did not further reduce kidney bacterial titer compared to SSG27 antibody (10 mpk) alone. No difference was observed between SSA12-LS-RF antibody and SSA12-LS antibody.
The various embodiments described above can be combined to provide further embodiments. All of the U.S. patents, U.S. patent application publications, U.S. patent applications, foreign patents, foreign patent applications and non-patent publications referred to in this specification and/or listed in the Request, including U.S. Provisional Application No. 63/512,279 filed on July 6, 2023, US Provisional Application No. 63/609,814 filed on December 13, 2023, and US Provisional Application 63/639,554 filed on April 26, 2024, are incorporated herein by reference, in their entirety. Aspects of the embodiments can be modified, if necessary to employ concepts of the various patents, applications and publications to provide yet further embodiments.
These and other changes can be made to the embodiments in light of the above-detailed description. In general, in the following claims, the terms used should not be construed to limit the claims to the specific embodiments disclosed in the specification and the claims, but should be construed to include all possible embodiments along with the full scope of equivalents to which such claims are entitled. Accordingly, the claims are not limited by the disclosure.
Claims
1. An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and/or CDRL3 amino acid sequence(s), or functional variants thereof (and optionally comprising the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, or functional variants thereof), of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs. :
(a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532 and 536, respectively; (xii) 542 and 546, respectively; (xiii) 552 and 556, respectively; (xiv) 562 and 566, respectively; (xv) 572 and 576, respectively; (xvi) 582 and 586, respectively; (xvii) 592 and 596, respectively; (xviii) 602 and 606, respectively; (xix) 612 and 616, respectively; (xx) 622 and 626, respectively; (xxi) 632 and 636, respectively; (xxii) 642 and 646, respectively; (xxiii) 652 and 656, respectively; (xxiv) 662 and 666, respectively; (xxv) 672 and 676, respectively; (xxvi) 682 and 686, respectively; or (xxvii) 692 and 696, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LT A);
(b) (i) 41 and 45, respectively; (ii) 181 and 185, respectively; (iii) 51 and 55, respectively; (iv) 191 and 195, respectively; (v) 280 and 284, respectively; (vi) 290 and 294, respectively; (vii) 300 and 304, respectively; (viii) 380 and 384, respectively; (ix) 390 and 394, respectively; (x) 400 and 404, respectively; or (xi) 420 and 424, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) (i) 61 and 65, respectively; (ii) 71 and 75, respectively; (iii) 201 and 205, respectively; (iv) 360 and 364, respectively; or (v) 370 and 374, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA);
(d) (i) 81 and 85, respectively; (ii) 91 and 95, respectively; (iii) 310 and 314, respectively; (iv) 320 and 324, respectively; (v) 330 and 334, respectively; (vi) 410 and 414, respectively; (vii) 702 and 706, respectively; (viii) 712 and 716, respectively; (ix) 722 and 726, respectively; (x) 732 and 736, respectively; (xi) 742 and 746, respectively; or (xii) 752 and 756,
respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively; (iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) (i) 340 and 344, respectively; or (ii) 350 and 354, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
2. An antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)H1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences, and optionally the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences, of one of the following antibodies:
(a) SSC35; SSE73; SSA12; SSA8; SSA9; SSA10; SSC1; SSC10; SSC15; SSAA5; SSAA7; SSAA15; SSAA21; SSAA60; SSAA65; SSAA68; SSAA69; SSAA71; SSAA72; SSAB24; SSAB30; SSAC1; SSE137; SSE150; SSE151; SSE153; or SSF69, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LT A);
(b) SSG20; SSF2; SSF11; SSF10; SSG50; SSG54; SSM47; SSM45; SSF52; SSG51; or SSG59, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) SSL5; SSL17; SSL35; SSG27; or SSG39, wherein the antibody or antigenbinding fragment is capable of binding to a clumping factor A (ClfA);
(d) SSH3; SSF33; SSG44; SSG48; SSM37; SSF34; SSN2; SSN6; SSH8; SSM35; SSM36; or SSM40, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) SSE1; SSE158; SSAA8; SSAA26; SSAB5; SSAI3; SSAI8; SSAI2; SSAF1; SSAL2; or SSAM6; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) SSB18; SSB24; SSB54; SSB65; or SSB67; wherein the antibody or antigenbinding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) SSH4; or SSH7; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
3. The antibody or antigen-binding fragment of claim 1 or 2, wherein the CDRH1,
CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences are according to:
(i) the IMGT numbering system (optionally using the junction definitions for the
CDRH3 and CDRL3 sequences);
(ii) the Kabat numbering system;
(iii) the Chothia numbering system;
(iv) the Enhanced Chothia (also referred to as “Martin”) numbering system;
(v) the AHo numbering system;
(vi) the North numbering system;
(vii) the Contact numbering system;
(viii) the EU numbering system;
(ix) the AbM numbering system; or
(x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
4. An antibody or antigen-binding fragment thereof comprising a CDRH3 and a CDRL3, wherein the CDRH3 and the CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.:
(a) (i) 34 and 38, respectively; (ii) 124 and 128, respectively; (iii) 24 and 28, respectively; (iv) 134 and 138, respectively; (v) 144 and 148, respectively; (vi) 154 and 158, respectively; (vii) 164 and 168, respectively; (viii) 244 and 248, respectively; (ix) 174 and 178, respectively; (x) 525 and 529, respectively; (xi) 535 and 539, respectively; (xii) 545 and 549,
respectively; (xiii) 555 and 559, respectively; (xiv) 565 and 569, respectively; (xv) 575 and 579, respectively; (xvi) 585 and 589, respectively; (xvii) 595 and 599, respectively; (xviii) 605 and 609, respectively; (xix) 615 and 619, respectively; (xx) 625 and 629, respectively; (xxi) 635 and 639, respectively; (xxii) 645 and 649, respectively; (xxiii) 655 and 659, respectively; (xxiv) 665 and 669, respectively; (xxv) 675 and 679, respectively; (xxvi) 685 and 689, respectively; or (xxvii) 695 and 699, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LT A);
(b) (i) 44 and 48, respectively; (ii) 184 and 188, respectively; (iii) 54 and 58, respectively; (iv) 194 and 198, respectively; (v) 283 and 287, respectively; (vi) 293 and 297, respectively; (vii) 303 and 307, respectively; (viii) 383 and 387, respectively; (ix) 393 and 397, respectively; (x) 403 and 407, respectively; or (xi) 423 and 427, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) (i) 64 and 68, respectively; (ii) 74 and 78, respectively; (iii) 204 and 208, respectively; (iv) 363 and 367, respectively; or (v) 373 and 377, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA);
(d) (i) 84 and 88, respectively; (ii) 94 and 98, respectively; (iii) 313 and 317, respectively; (iv) 323 and 327, respectively; (v) 333 and 337, respectively; (vi) 413 and 417, respectively; (vii) 705 and 709, respectively; (viii) 715 and 719, respectively; (ix) 725 and 729, respectively; (x) 735 and 739, respectively; (xi) 745 and 749, respectively; or (xii) 755 and 759, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) (i) 4 and 8, respectively; (ii) 14 and 18, respectively; (iii) 435 and 439, respectively; (iv) 445 and 449, respectively; (v) 455 and 459, respectively; (vi) 465 and 469, respectively; (vii) 475 and 479, respectively; (viii) 485 and 489, respectively; (ix) 495 and 499, respectively; (x) 505 and 509, respectively; or (xi) 515 and 519, respectively; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) (i) 214 and 218, respectively; (ii) 104 and 108, respectively; (iii) 224 and 228, respectively; (iv) 114 and 118, respectively; or (v) 234 and 238, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) (i) 343 and 347, respectively; or (ii) 353 and 357, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
5. An antibody, or an antigen-binding fragment thereof, comprising a complementarity determining region (CDR)H1, a CDRH2, a CDRH3, a CDRL1, a CDRL2, and a CDRL3, wherein the CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOS.:
(a) (i) 32-34 and 36-38, respectively; (ii) 122-124 and 126-128, respectively; (iii) 22-24 and 26-28, respectively; (iv) 132-134 and 136-138, respectively; (v) 142-144 and 146-148, respectively; (vi) 152-154 and 156-158, respectively; (vii) 162-164 and 166-168, respectively; (viii) 242-244 and 246-248, respectively; (ix) 172-174 and 176-178, respectively; (x) 523-525 and 527-529, respectively; (xi) 533-535 and 537-539, respectively; (xii) 543-545 and 547-549, respectively; (xiii) 553-555 and 557-559, respectively; (xiv) 563-565 and 567-569, respectively; (xv) 573-575 and 577-579, respectively; (xvi) 583-585 and 587-589, respectively; (xvii) 593-595 and 597-599, respectively; (xviii) 603-605 and 607-609, respectively; (xix) 613-615 and 617- 619, respectively; (xx) 623-625 and 627-629, respectively; (xxi) 633-635 and 637-639, respectively; (xxii) 643-645 and 647-649, respectively; (xxiii) 653-655 and 657-659, respectively; (xxiv) 663-665 and 667-669, respectively; (xxv) 673-675 and 677-679, respectively; (xxvi) 683-685 and 687-689, respectively; or (xxvii) 693-695 and 697-699, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA);
(b) (i) 42-44 and 46-48, respectively; (ii) 182-184 and 186-188, respectively; (iii) 52-54 and 56-58, respectively; (iv) 192-194 and 196-198, respectively; (v) 281-283 and 285-287, respectively; (vi) 291-293 and 295-297, respectively; (vii) 301-303 and 305-307, respectively; (viii) 381-383 and 385-387, respectively; (ix) 391-393 and 395-397, respectively; (x) 401-403 and 405-407, respectively; or (xi) 421-423 and 425-427, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) (i) 62-64 and 66-68, respectively; (ii) 72-74 and 76-78, respectively; (iii) 202- 204 and 206-208, respectively; (iv) 361-363 and 365-367, respectively; or (v) 371-373 and 375- 377, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA);
(d) (i) 82-84 and 86-88, respectively; (ii) 92-94 and 96-98, respectively; (iii) 311- 313 and 315-317, respectively; (iv) 321-323 and 325-327, respectively; (v) 331-333 and 335- 337, respectively; (vi) 411-413 and 415-417, respectively; (vii) 703-705 and 707-709, respectively; (viii) 713-715 and 717-719, respectively; (ix) 723-725 and 727-729, respectively;
(x) 733-735 and 737-739, respectively; (xi) 743-745 and 747-749, respectively; or (xii) 753-755 and 757-759, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) (i) 2-4 and 6-8, respectively; (ii) 12-14 and 16-18, respectively; (iii) 433-435 and 437-439, respectively; (iv) 443-445 and 447-449, respectively; (v) 453-455 and 457-459, respectively; (vi) 463-465 and 467-469, respectively; (vii) 473-475 and 477-479, respectively; (viii) 483-485 and 487-489, respectively; (ix) 493-495 and 497-499, respectively; (x) 503-505 and 507-509, respectively; or (xi) 513-515 and 517-519, respectively; wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) (i) 212-214 and 216-218, respectively; (ii) 102-104 and 106-108, respectively; (iii) 222-224 and 226-228, respectively; (iv) 112-114 and 116-118, respectively; or (v) 232-234 and 236-238, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) (i) 341-343 and 345-347, respectively; or (ii) 351-353 and 355-357, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
6. The antibody or antigen-binding fragment of any one of claims 1-5, comprising a heavy chain variable domain framework region (VHFR)1, a VHFR2, a VHFR3, a light chain variable domain framework region (VLFR)1, a VLFR2, a VLFR3, and/or a VLFR4 (or a variant of the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4 comprising one, two, three, four, or five substitutions, insertions, and/or deletions, or a variant having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to the VHFR1, VHFR2, VHFR3, VHFR4, VLFR1, VLFR2, VLFR3, or VLFR4, respectively) of the VH and VL amino acid sequences set forth in SEQ ID NOs.: (i) 41 and 45, respectively; (ii) 31 and 35, respectively; (iii) 121 and 125, respectively; (iv) 21 and 25, respectively; (v) 131 and 135, respectively; (vi) 141 and 145, respectively; (vii) 151 and 155, respectively; (viii) 161 and 165, respectively; (ix) 241 and 245, respectively; (x) 171 and 175, respectively; (xi) 1 and 5, respectively; (xii) 11 and 15, respectively; (xiii) 181 and 185, respectively; (xiv) 51 and 55, respectively; (xv) 191 and 195, respectively; (xvi) 211 and 215, respectively; (xvii) 101 and 105, respectively; (xviii) 221 and 225, respectively; (xix) 111 and 115, respectively; (xx) 231 and 235, respectively; (xxi) 81 and 85, respectively; (xxii) 91 and 95, respectively; (xxiii) 61 and 65,
respectively; (xxiv) 71 and 75, respectively; (xxv) 201 and 205, respectively; (xxvi) 280 and 284, respectively; (xxvii) 290 and 294, respectively; (xxviii) 300 and 304, respectively; (xxix) 310 and 314, respectively; (xxx) 320 and 324, respectively; (xxxi) 330 and 334, respectively; (xxxii) 340 and 344, respectively; (xxxiii) 350 and 354, respectively; (xxxiv) 360 and 364, respectively; (xxxv) 370 and 374, respectively; (xxxvi) 380 and 384, respectively; (xxxvii) 390 and 394, respectively; (xxxviii) 400 and 404, respectively; (xxxix) 410 and 414, respectively; (xl) 420 and 424, respectively; (xli) 432 and 436, respectively; (xlii) 442 and 446, respectively; (xliii) 452 and 456, respectively; (xliv) 462 and 466, respectively; (xlv) 472 and 476, respectively; (xlvi) 482 and 486, respectively; (xlvii) 492 and 496, respectively; (xlviii) 502 and 506, respectively; (xlix) 512 and 516, respectively; (1) 522 and 526, respectively; (li) 532 and 536, respectively; (lii) 542 and 546, respectively; (liii) 552 and 556, respectively; (liv) 562 and 566, respectively; (Iv) 572 and 576, respectively; (Ivi) 582 and 586, respectively; (Ivii) 592 and 596, respectively; (Iviii) 602 and 606, respectively; (lix) 612 and 616, respectively; (lx) 622 and 626, respectively; (Ixi) 632 and 636, respectively; (Ixii) 642 and 646, respectively; (Ixiii) 652 and 656, respectively; (Ixiv) 662 and 666, respectively; (Ixv) 672 and 676, respectively; (Ixvi) 682 and 686, respectively; (Ixvii) 692 and 696, respectively; (Ixviii) 702 and 706, respectively; (Ixix) 712 and 716, respectively; (Ixx) 722 and 726, respectively; (Ixxi) 732 and 736, respectively; (Ixxii) 742 and 746, respectively; or (Ixxiii) 752 and 756, respectively.
7. The antibody or antigen-binding fragment of claim 6, wherein the framework region or regions are according to:
(i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences);
(ii) the Kabat numbering system;
(iii) the Chothia numbering system;
(iv) the Enhanced Chothia (also referred to as “Martin”) numbering system;
(v) the AHo numbering system;
(vi) the North numbering system;
(vii) the Contact numbering system;
(viii) the EU numbering system;
(ix) the AbM numbering system; or
(x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems
combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
8. The antibody or antigen-binding fragment of any one of claims 1-7, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.:
(a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532 and 536, respectively; (xii) 542 and 546, respectively; (xiii) 552 and 556, respectively; (xiv) 562 and 566, respectively; (xv) 572 and 576, respectively; (xvi) 582 and 586, respectively; (xvii) 592 and 596, respectively; (xviii) 602 and 606, respectively; (xix) 612 and 616, respectively; (xx) 622 and 626, respectively; (xxi) 632 and 636, respectively; (xxii) 642 and 646, respectively; (xxiii) 652 and 656, respectively; (xxiv) 662 and 666, respectively; (xxv) 672 and 676, respectively; (xxvi) 682 and 686, respectively; or (xxvii) 692 and 696, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LT A);
(b) (i) 41 and 45, respectively; (ii) 181 and 185, respectively; (iii) 51 and 55, respectively; (iv) 191 and 195, respectively; (v) 280 and 284, respectively; (vi) 290 and 294, respectively; (vii) 300 and 304, respectively; (viii) 380 and 384, respectively; (ix) 390 and 394, respectively; (x) 400 and 404, respectively; or (xi) 420 and 424, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) (i) 61 and 65, respectively; (ii) 71 and 75, respectively; (iii) 201 and 205, respectively; (iv) 360 and 364, respectively; or (v) 370 and 374, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA);
(d) (i) 81 and 85, respectively; (ii) 91 and 95, respectively; (iii) 310 and 314, respectively; (iv) 320 and 324, respectively; (v) 330 and 334, respectively; (vi) 410 and 414, respectively; (vii) 702 and 706, respectively; (viii) 712 and 716, respectively; (ix) 722 and 726, respectively; (x) 732 and 736, respectively; (xi) 742 and 746, respectively; or (xii) 752 and 756,
respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively; (iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) (i) 340 and 344, respectively; or (ii) 350 and 354, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin).
9. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody or antigen-binding fragment is capable of binding to a LTA, wherein, optionally, the antibody or antigen-binding binding fragment is capable of binding to: (i) a Type I LTA; (ii) a Type II LTA; (iii) a Type IV LTA; or (iv) any combination of (i)-(iii).
10. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody or antigen-binding binding fragment is capable of binding to a Gmd, wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a S. aureus, to S. epider midis, or to S. aureus and to S. epidermidis.
11. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody or antigen-binding fragment is capable of binding to a ClfA.
12. The antibody or antigen-binding fragment of claim 11, wherein the antibody or antigen-binding binding is capable of binding to a ClfA_001, a ClfA_002, and/or a ClfA_004.
13. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA),
wherein, optionally, the antibody or antigen-binding fragment is capable of binding to a SpAKKAA.
14. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin, wherein, optionally, the alpha toxin comprises a-hemolysin (also called Hla).
15. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody or antigen-binding fragment is capable of binding to a gamma toxin.
16. The antibody or antigen-binding fragment of any one of claims 1-8 and 15, wherein the antibody or antigen-binding fragment is capable of binding to a leukocidin, wherein, optionally, the leukocidin comprises a LukD, a LukE, a LukF, a LukS, or any combination thereof.
17. The antibody or antigen-binding fragment of claim 16, wherein the antibody or antigen-binding fragment is capable of binding to a LukED, a LukSF, or to a LukED and a LukSF.
18. The antibody or antigen-binding fragment of any one of claims 1-8 and 15-17, wherein the antibody or antigen-binding fragment is capable of binding to a HIgA, a HIgB, or both.
19. The antibody or antigen-binding fragment of any one of claims 1-8, wherein the antibody is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin.).
20. The antibody or antigen-binding fragment of any one of claims 1-19, wherein the antibody or antigen-binding fragment is capable of binding to any one, any two, any three, any four, any five, any six, any seven, any eight, any nine, any ten, any eleven, or all twelve of: S. aureus FPR3757; S. aureus NE284 TE2 mut; S. epidermidis RP62A; S. epidermidis 1200; S. epidermidis NH404008; S. lugdunensis: S. pyogenes,' E.faecalis,' S. agalacliae S. hominis,' L. garvieae: and S. pneumonia.
21. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)Hl, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences of the heavy chain variable domain (VH) and light chain variable domain (VL) amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
22. The anti -LTA antibody or antigen-binding fragment of claim 21, wherein the CDRS are according to:
(i) the IMGT numbering system (optionally using the junction definitions for the CDRH3 and CDRL3 sequences);
(ii) the Kabat numbering system;
(iii) the Chothia numbering system;
(iv) the Enhanced Chothia (also referred to as “Martin”) numbering system;
(v) the AHo numbering system;
(vi) the North numbering system;
(vii) the Contact numbering system;
(viii) the EU numbering system;
(ix) the AbM numbering system; or
(x) a combination of any two or more of: IMGT, Kabat, Chothia, Enhanced Chothia, AHo, EU, North, AbM, and Contact, wherein, optionally, the two or more numbering systems combined produce CDR definitions having the greatest length of the CDRs according to any numbering system or combination of numbering systems.
23. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising the complementarity determining region (CDR)Hl, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3 amino acid sequences set forth in SEQ ID NOs.:22-24 and 26-28, respectively.
24. The anti-LTA antibody or antigen-binding fragment of any one of claims 21-23, comprising a VH and a VL, wherein the VH and the VL comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or
consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.: 21 and 25, respectively.
25. An anti-lipoteichoic acid (LTA) antibody, or an antigen-binding fragment thereof, comprising a heavy chain variable domain (VH) and a light chain variable domain (VL), wherein the VH and VL comprise, consist essentially of, or consist of the amino acid sequences set forth in SEQ ID NOs.:21 and 25, respectively.
26. The antibody or antigen-binding fragment of any one of claims 1-25, wherein the antibody, or the antigen-binding fragment, comprises a human antibody, a monoclonal antibody, a purified antibody, a single chain antibody (e.g., scFv), a Fab, a Fab’, a F(ab’)2, or Fv.
27. The antibody or antigen-binding fragment of any one of claims 1-26, wherein the antibody or antigen-binding fragment is a multi-specific antibody or antigen-binding fragment, wherein, optionally, the antibody or antigen-binding fragment is a bi-specific antibody or antigen-binding fragment.
28. The antibody or antigen-binding fragment of claim 27, which comprises (1) the six CDRS, and optionally the VH and VL, of a first antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1) and (2) the six CDRS, and optionally the VH and VL, of a second, different antibody or antigen-binding fragment of the present disclosure (e.g., as shown in Table 1).
29. The antibody or antigen-binding fragment of claim 27 or 28, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and a ClfA; a SpA and a ClfA; a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; or a Sbi and a ClfA.
30. The antibody or antigen-binding fragment of any one of claims 27-29, comprising a first antigen-binding domain and second antigen-binding domain, wherein the first antigenbinding domain or the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; or (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC10; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF10; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE150; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
31. The antibody or antigen-binding fragment of any one of claims 27-30, comprising a first antigen-binding domain and second antigen-binding domain, wherein the first antigenbinding domain and the second antigen-binding domain comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S or M428L/N434S/H435R/Y436F mutations.
32. An antibody, or an antigen-binding fragment thereof, which is a multi-specific antibody or antigen-binding fragment thereof, which is capable of binding to: an LTA and a Gmd; an LTA and an alpha toxin; an LTA and a gamma toxin; an LTA and a leucocidin; an LTA and a SpA; an LTA and a ClfA; an alpha toxin and a Gmd; an alpha toxin and a gamma toxin; an alpha toxin and a leucocidin; an alpha toxin and a SpA; an alpha toxin and a ClfA; a gamma toxin and a Gmd; a gamma toxin and a leucocidin; a gamma toxin and a SpA; a gamma toxin and a ClfA; a leucocidin and a SpA; a leucodin and a Gmd; a leucocidin and a ClfA; a Gmd and
a ClfA; a SpA and a ClfA; a Sbi and a LTA; a Sbi and a Gmd; a Sbi and an alpha toxin; a Sbi and a gamma toxin; a Sbi and a leucocidin; a Sbi and a SpA; or a Sbi and a ClfA.
33. The antibody or antigen-binding fragment of any one of claims 27-32, which comprises or is of a format selected from: a Bispecific T cell Engager (BiTE); a DART; a Knobs-Into-Holes (KIH) assembly; a scFv-CH3-KIH assembly; a KIH Common Light-Chain antibody; a TandAb; a Triple Body; a TriBi Minibody; a Fab-scFv; a scFv-CH-CL-scFv; a F(ab’)2-scFv2; a tetravalent Hcab; an intrabody; a CrossMab; a Dual Action Fab (DAF) (two-in- one or four-in-one); a DutaMab; a DT-IgG, a charge paired antibody; a Fab-arm Exchange antibody, a SEEDbody; a Triomab; a LUZ-Y assembly, an Fcab; a KX-body; an orthogonal Fabs antibody; a DVD-Ig; am IgG(H)-scFv; an scFv-(H)IgG; an IgG(L)-scFv; an scFv-(L)IgG; an IgG(L,H)-Fv; an IgG(H)-V; a V(H)-IgG; an IgG(L)-V; a V(L)-IgG; a KIH IgG-scFab; a 2scFv- IgG; a IgG-2scFv; a scFv4-Ig; a Zybody; a DVI-IgG (four-in-one), a FIT-Ig; a WuxiBody ; and an In-Elbow-Insert Ig.
34. The antibody or antigen-binding fragment of any one of claims 1-33, wherein the antibody or antigen-binding fragment comprises an (e.g., IgGl or IgG3) Fc polypeptide or a fragment thereof.
35. The antibody or antigen-binding fragment of any one of claims 1-34, which comprises an IgG, IgA, IgM, IgE, or IgD isotype.
36. The antibody or antigen-binding fragment of any one of claims 1-35, which comprises an IgG isotype selected from IgGl, IgG2, IgG3, and IgG4, optionally an IgGl isotype comprising a heavy chain with a C-terminal lysine removed or a C-terminal glycine-lysine removed.
37. The antibody or antigen-binding fragment of any one of claims 1-36, which comprises an IgGl isotype.
38. The antibody or antigen-binding fragment of any one of claims 1-37, which comprises an IgGlm3 allotype, an IgGlml7 allotype, an IgGlml allotype, or any combination thereof.
39. The antibody or antigen-binding fragment of any one of claims 1-36, which comprises an IgG3 isotype.
40. The antibody or antigen-binding fragment of any one of claims 34-39, wherein the Fc polypeptide or fragment thereof comprises:
(i) a mutation that increases binding affinity to a human FcRn (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols)), as compared to a reference Fc polypeptide that does not comprise the mutation; and/or
(ii) a mutation that increases binding affinity to a human FcyR (e.g., as measured using surface plasmon resonance (SPR) (e.g., Biacore, e.g., T200 instrument, using manufacturer’s protocols, and/or as measured using mesoscale discovery (MSD))) as compared to a reference Fc polypeptide that does not comprise the mutation.
41. The antibody or antigen-binding fragment of claim 40, wherein the mutation that increases binding affinity to a human FcRn comprises: M428L; N434S; N434H; N434A; N434S; M252Y; S254T; T256E; T250Q; P257I; Q311I; D376V; T307A; E380A; or any combination thereof.
42. The antibody or antigen-binding fragment of claim 40 or 41, wherein the mutation that increases binding affinity to a human FcRn comprises: (i) M428L/N434S; (ii) M252Y/S254T/T256E; (iii) T250Q/M428L; (iv) P257VQ311I; (v) P257I/N434H; (vi) D376V/N434H; (vii) T307A/E380A/N434A; (viii) M428L/N434A; or (ix) any combination of (i)-(viii).
43. The antibody or antigen-binding fragment of any one of claims 40-42, wherein the mutation that increases binding affinity to a human FcRn comprises M428L/N434S or M428L/N434A.
44. The antibody or antigen-binding fragment of any one of claims 40-43, wherein the mutation that enhances binding to a FcyR comprises S239D; I332E; A330L; G236A; or any combination thereof.
45. The antibody or antigen-binding fragment of any one of claims 40-44, wherein the mutation that enhances binding to a FcyR comprises: (i) S239D/I332E; (ii) S239D/A330L/I332E; (iii) G236A/S239D/I332E; or (iv) G236A/A330L/I332E, wherein the Fc polypeptide or fragment thereof optionally comprises Ser at position 239.
46. The antibody or antigen-binding fragment of any one of claims 1-45, which comprises a mutation that alters glycosylation, wherein the mutation that alters glycosylation comprises N297A, N297Q, or N297G, and/or which is aglycosylated, and/or which is afucosylated.
47. The antibody or antigen-binding fragment of any one of claims 1-46, comprising in a heavy chain thereof, the amino acid mutation(s) set forth in any one of (i)-(xix): (i) G236A, L328V, and Q295E; (ii) G236A, P230A, and Q295E; (iii) G236A, R292P, and I377N; (iv) G236A, K334A, and Q295E; (v) G236S, R292P, and Y300L; (vi) G236A and Y300L; (vii) G236A, R292P, and Y300L; (viii) G236S, G420V, G446E, and L309T; (ix) G236A and R292P; (x) R292P and Y300L; (xi) G236A and R292P; (xii) Y300L; (xiii) E345K, G236S, L235Y, and S267E; (xiv) E272R, L309T, S219Y, and S267E; (xv) G236Y; (xvi) G236W; (xvii) F243L, G446E, P396L, and S267E; (xviii) G236A, S239D, and H268E; (xix) any one or more of the mutations shown in Figure 14A, Figure 14B, Figure 14C, Figure 14E, Figure 14F, Figure 14G, Figure 14H, Figure 141, Figure 14J, or Figure 14K.
48. The antibody or antigen-binding fragment of any one of claims 1-47, comprising in a heavy chain thereof, one or more of the following the amino acid mutations: G236A; Y300L; R292P.
49. The antibody or antigen-binding fragment of any one of claims 1-48, comprising, in a heavy chain thereof, an amino acid mutation that promotes complement activation, wherein, optionally, the amino acid mutation that promotes complement activation comprises or consists of: K236W/E333S; K326A/E333A; S267E/H268F/S324T; S267E/H268F/S324T/G236A/I332E; Y300L; G236A/Y300L; or G236A/R292P/Y300L.
50. The antibody or antigen-binding fragment of any one of claims 1-49, comprising, in a heavy chain or a CH3 thereof, an amino acid mutation that reduces binding by
Staphylococcus Protein A, wherein, the optionally, amino acid mutation that reduces binding by
Staphylococcus Protein A comprises or consists of: H435R; Y436F; or H435R/Y436F.
51. The antibody or antigen-binding fragment of any one of claims 1-50, comprising a human IgGl isotype and comprising, in a heavy chain or a CH3 thereof, the amino acid mutations M428L/N434S/H435R/Y436F.
52. The antibody or antigen-binding fragment of any one of claims 1-51, which is capable of neutralizing an infection (in vitro, ex vivo, and/or in vivo) by a bacteria expressing the LT A, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi.
53. The antibody or antigen-binding fragment of any one of claims 1-52, which is capable of neutralizing an S. aureus infection in vitro, ex vivo, or in vivo.
54. The antibody or antigen-binding fragment of any one of claims 1-53, which is capable of opsonizing and/or is capable of promoting phagocytosis of a bacteria expressing the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, Sbi, or gamma toxin, wherein, optionally, the bacteria comprises S. aureus.
55. The antibody or antigen-binding fragment of any one of claims 1-54, which: is afucosylated; has been produced in a host cell that is incapable of fucosylation or that is inhibited in its ability to fucosylate a polypeptide; has been produced under conditions that inhibit fucosylation thereof by a host cell; or any combination thereof.
56. The antibody or antigen-binding fragment of any one of claims 1-55, which is human, humanized, or chimeric.
57. An antibody, or an antigen-binding fragment, that is capable of binding to an S. aureus antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi, wherein, optionally:
(i) the antibody or antigen-binding fragment is human, humanized, or chimeric;
(ii) the antibody or antigen-binding fragment binds to a LTA and does not complete with pagibaximab for binding to the LTA;
(iii) the antibody or antigen-binding fragment comprises a VH and a VL that comprise, consist essentially of, or consist of, amino acid sequences having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity or similarity to, or comprising, consisting essentially of, or consisting of, the VH and VL amino acid sequences set forth in SEQ ID NOs.:
(a) (i) 31 and 35, respectively; (ii) 121 and 125, respectively; (iii) 21 and 25, respectively; (iv) 131 and 135, respectively; (v) 141 and 145, respectively; (vi) 151 and 155, respectively; (vii) 161 and 165, respectively; (viii) 241 and 245, respectively; (ix) 171 and 175, respectively; (x) 522 and 526, respectively; (xi) 532 and 536, respectively; (xii) 542 and 546, respectively; (xiii) 552 and 556, respectively; (xiv) 562 and 566, respectively; (xv) 572 and 576, respectively; (xvi) 582 and 586, respectively; (xvii) 592 and 596, respectively; (xviii) 602 and 606, respectively; (xix) 612 and 616, respectively; (xx) 622 and 626, respectively; (xxi) 632 and 636, respectively; (xxii) 642 and 646, respectively; (xxiii) 652 and 656, respectively; (xxiv) 662 and 666, respectively; (xxv) 672 and 676, respectively; (xxvi) 682 and 686, respectively; or (xxvii) 692 and 696, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a lipoteichoic acid (LTA);
(b) (i) 41 and 45, respectively; (ii) 181 and 185, respectively; (iii) 51 and 55, respectively; (iv) 191 and 195, respectively; (v) 280 and 284, respectively; (vi) 290 and 294, respectively; (vii) 300 and 304, respectively; (viii) 380 and 384, respectively; (ix) 390 and 394, respectively; (x) 400 and 404, respectively; or (xi) 420 and 424, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a glucosaminidase (Gmd);
(c) (i) 61 and 65, respectively; (ii) 71 and 75, respectively; (iii) 201 and 205, respectively; (iv) 360 and 364, respectively; or (v) 370 and 374, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a clumping factor A (ClfA);
(d) (i) 81 and 85, respectively; (ii) 91 and 95, respectively; (iii) 310 and 314, respectively; (iv) 320 and 324, respectively; (v) 330 and 334, respectively; (vi)
410 and 414, respectively; (vii) 702 and 706, respectively; (viii) 712 and 716, respectively; (ix) 722 and 726, respectively; (x) 732 and 736, respectively; (xi) 742 and 746, respectively; or (xii) 752 and 756, respectively, wherein the antibody or antigen-binding fragment is capable of binding to a Staphylococcus Protein A (SpA);
(e) (i) 1 and 5, respectively; (ii) 11 and 15, respectively; (iii) 432 and 436, respectively; (iv) 442 and 446, respectively; (v) 452 and 456, respectively; (vi) 462 and 466, respectively; (vii) 472 and 476, respectively; (viii) 482 and 486, respectively; (ix) 492 and 496, respectively; (x) 502 and 506, respectively; or (xi) 512 and 516, respectively, wherein the antibody or antigen-binding fragment is capable of binding to an alpha toxin;
(f) (i) 211 and 215, respectively; (ii) 101 and 105, respectively; (iii) 221 and 225, respectively; (iv) 111 and 115, respectively; or (v) 231 and 235, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a leucocidin and/or gamma toxin; or
(g) (i) 340 and 344, respectively; or (ii) 350 and 354, respectively; wherein the antibody or antigen-binding fragment is capable of binding to a second immunoglobulin-binding protein (Sbi, also called a Staphylococcal binder of immunoglobulin); and/or
(iv) the antibody or antigen-binding fragment comprises an Fc polypeptide or fragment thereof, or a heavy chain, according to any one of claims 34-51.
58. An isolated polynucleotide encoding the antibody or antigen-binding fragment of any one of claims 1-57, or encoding a VH, a Fd, a heavy chain, a VL, and/or a light chain of the antibody or the antigen-binding fragment.
59. The polynucleotide of claim 58, wherein the polynucleotide comprises deoxyribonucleic acid (DNA) or ribonucleic acid (RNA), wherein the RNA optionally comprises messenger RNA (mRNA).
60. The polynucleotide of claim 59, wherein the polynucleotide comprises selfamplifying RNA (saRNA), trans-amplifying RNA (taRNA), or circular RNA (circRNA).
61. The polynucleotide of any one of claims 58-60, comprising a modified nucleoside, a cap-1 structure, a cap-2 structure, or any combination thereof, wherein, optionally, the polynucleotide comprises a pseudouridine, a N6-methyladenonsine, a 5-methylcytidine, a 2- thiouridine, or any combination thereof, wherein, further optionally, the pseudouridine comprises N1 -methylpseudouridine.
62. The polynucleotide of any one of claims 58-61, which is codon-optimized for expression in a host cell.
63. The polynucleotide of claim 62, wherein the host cell comprises a human cell or a CHO cell.
64. The polynucleotide of claim 62, wherein the host cell is a production host cell.
65. The polynucleotide of claim 62, wherein the host cell is an in vivo human host cell.
66. A recombinant vector, DNA therapeutic, or RNA therapeutic comprising the polynucleotide of any one of claims 58-65.
67. A DNA therapeutic construct or RNA therapeutic construct comprising the polynucleotide of any one of claims 58-65 encapsulated in a carrier, wherein the carrier optionally comprises a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
68. A recombinant vector comprising the polynucleotide of any one of claims 58-65.
69. A host cell comprising the polynucleotide of any one of claims 58-65 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68, wherein the polynucleotide is optionally heterologous to the host cell and/or wherein the host cell is capable of expressing the encoded antibody or antigen-binding fragment or polypeptide.
70. An isolated human B cell comprising the polynucleotide of any one of claims 58- 65 and/or the vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68, wherein polynucleotide is optionally heterologous to the human B cell and/or wherein the human B cell is immortalized.
71. A composition or combination comprising:
(i) the antibody or antigen-binding fragment of any one of claims 1-57;
(ii) the polynucleotide of any one of claims 58-65;
(iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68;
(iv) the host cell of claim 69; and/or
(v) the human B cell of claim 70, and, optionally, a pharmaceutically acceptable excipient, carrier, or diluent.
72. The composition or combination of claim 71, which comprises (1) a first antibody or antigen-binding fragment of any one of claims 1-57 and (2) a second, different antibody or antigen-binding fragment of any one of claims 1-57.
73. The composition or combination of claim 72, wherein the first antibody or antigen-binding fragment and the second antibody or antigen-binding fragment are capable of binding to: an LTA and a Gmd, respectively; an LTA and an alpha toxin, respectively; an LTA and a gamma toxin, respectively; an LTA and a leucocidin, respectively; an LTA and a SpA, respectively; an LTA and a ClfA, respectively; an alpha toxin and a Gmd, respectively; an alpha toxin and a gamma toxin, respectively; an alpha toxin and a leucocidin, respectively; an alpha toxin and a SpA, respectively; an alpha toxin and a ClfA, respectively; a gamma toxin and a Gmd, respectively; a gamma toxin and a leucocidin, respectively; a gamma toxin and a SpA, respectively; a gamma toxin and a ClfA, respectively; a leucocidin and a SpA, respectively; a leucodin and a Gmd, respectively; a leucocidin and a ClfA, respectively; a Gmd and a ClfA, respectively; a SpA and a ClfA, respectively; a Sbi and a LTA, respectively; a Sbi and a Gmd, respectively; a Sbi and an alpha toxin, respectively; a Sbi and a gamma toxin, respectively; a Sbi and a leucocidin, respectively; a Sbi and a SpA, respectively; or a Sbi and a SlfA, respectively.
74. The composition or combination of any one of claims 71-73, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment or the second antibody or antigen-binding fragment comprises the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12; (ii) SSG20; (iii) SSH3; (iv) SSL17; (v) SSE1; (vi) SSE158; (vii) SSB24; (viii) SSB65; (ix) SSG50; (x) SSG54; (xi) SSM47; (xii) SSG44; (xiii) SSG48; (xiv) SSM37; (xv) SSH4; (xvi) SSH7; (xvii) SSG27; (xviii) SSG39; (xix) SSM45; (xx) SSF52; (xxi) SSG51; (xxii) SSF34; (xxiii) SSG59; (xxiv) SSC35; (xxv) SSE73; (xxvi) SSA8; (xxvii) SSA9; (xxviii) SSA10; (xxix) SSC1; (xxx) SSC10; (xxxi) SSC15; (xxxii) SSF2; (xxxiii) SSF11; (xxxiv) SSF10; (xxxv) SSB18; (xxxvi) SSB54; (xxxvii) SSB67; (xxxviii) SSF33; (xxxix) SSL5; (xl) SSL35; (xli) SSAA8; (xlii) SSAA26; (xliii) SSAB5; (xliv) SSAI3; (xlv) SSAI8; (xlvi) SSAI2; (xlvii) SSAF1; (xlviii) SSAL2; (xlix) SSAM6; (1) SSAA5; (li) SSAA7; (lii) SSAA15; (liii) SSAA21; (liv) SSAA60; (Iv) SSAA65; (Ivi) SSAA68; (Ivii) SSAA69; (Iviii) SSAA71; (lix) SSAA72; (lx) SSAB24; (Ixi) SSAB30; (Ixii) SSAC1; (Ixiii) SSE137; (Ixiv) SSE150; (Ixv) SSE151; (Ixvi) SSE153; (Ixvii) SSF69; (Ixviii) SSN2; (Ixix) SSN6; (Ixx) SSH8; (Ixxi) SSM35; (Ixxii) SSM36; or (Ixxiii) SSM40.
75. The composition or combination of any one of claims 71-74, comprising a first antibody or antigen-binding fragment and a second antibody or antigen-binding fragment, wherein the first antibody or antigen-binding fragment and the second antibody or antigenbinding fragment comprise the six CDR sequences (e.g., according to IMGT), and optionally the VH and VL, of: (i) SSA12 and SSG20, respectively; (ii) SSA12 and SSL17, respectively; (iii) SSA12 and SSH3, respectively; (iv) SSG20 and SSL17, respectively; (v) SSG20 and SSH3, respectively; (vi) SSL17 and SSH3, respectively; (vii) SSE1 and SSE158, respectively; or (viii) SSB24 and SSB65, respectively, wherein, optionally, the antibody or antigen-binding fragment comprises a human IgGl Fc polypeptide comprising M428L/N434S/H435R/Y436F mutations.
76. The composition or combination of claim 71, comprising a first polynucleotide and a second polynucleotide, each according to any one of claims 58-65, or a recombinant vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68, encoding two or more antibodies or antigen-binding fragments, wherein the first antibody or antigen-binding fragment encoded by the first polypeptide and second antibody or antigen-binding fragment encoded by
the second polypeptide, respectively, or the multispecific antibody or antigen-binding fragment, comprise(s) CDRH1, CDRH2, CDRH3, CDRL1, CDRL2, and CDRL3, and optionally VH and VL, according to any two antibodies of the present disclosure, or any antigen-binding fragments thereof.
77. A composition comprising the polynucleotide of any one of claims 58-65 or the vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68 encapsulated in a carrier molecule, wherein the carrier molecule optionally comprises a lipid, a lipid-derived delivery vehicle, such as a liposome, a solid lipid nanoparticle, an oily suspension, a submicron lipid emulsion, a lipid microbubble, an inverse lipid micelle, a cochlear liposome, a lipid microtubule, a lipid microcylinder, lipid nanoparticle (LNP), or a nanoscale platform.
78. A(n, e.g., in vitro or ex vivo) method of making an antibody or antigen-binding fragment of any one of claims 1-57, comprising culturing the host cell of claim 69 or the human B cell of claim 70 for a time and under conditions sufficient for the host cell or human B cell, respectively, to express the antibody or antigen-binding fragment.
79. The method of making an antibody or antigen-binding fragment of claim 78, wherein the host cell of claim 69 or the human B cell of claim 70 comprises the polynucleotide of any one of claim 58-65 and/or recombinant vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68.
80. The method of claim 78 or 79, further comprising isolating the antibody or antigen-binding fragment.
81. A method of treating or preventing a bacterial infection in a(n, e.g. , human) subject, wherein the bacterial infection is optionally bacteremia, wherein further optionally the bacteremia is hemodialysis associated/line associated bacteremia, the method comprising administering to the subject an effective amount of
(i) the antibody or antigen-binding fragment of any one of claims 1-57;
(ii) the polynucleotide of any one of claims 58-65;
(iii) the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68;
(iv) the host cell of claim 69;
(v) the human B cell of claim 70; and/or
(vi) the composition or combination of any one of claims 71-77, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises S. aureus.
82. The method of claim 81, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) S. aureus, wherein, further optionally, the S. aureus comprises a methicillin-resistant S. Aureus (MRSA); (2) a Coagulase-negative Staphylococcus (CoNS); (3) S. epidermidis,' (4) E. faecalis,' (5) S. pyogenes,' (6) S. agalactiae,' (7) L. garviae,' (8) S. pneumoniae,' (9) S. lugdunensis,' (10) A hominis,' (11) S. epidermidis,' or (12) any combination of (l)-(l 1).
83. The method of claim 81 or 82, comprising administering a single dose of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
84. The method of any one of claims 81-83, comprising administering two or more doses of the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition to the subject.
85. The method of any one of claims 81-84, comprising administering the antibody or antigen-binding fragment, polypeptide, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, or composition intramuscularly, subcutaneously, or intravenously.
86. The method of any one of claims 81-85, wherein the treatment and/or prevention comprises pre-exposure or post-exposure prophylaxis.
87. The method of any one of claims 81-86, wherein the subject: is using a ventilator; is undergoing, will undergo, or has recently undergone surgery; has a blood stream infection or a history of blood stream infections; is receiving, will receive, or has received a prosthetic joint or other implant; is undergoing, will undergo, or has undergone dialysis; has bacteremia or a history
of bacteremia; has neutropenia or a history of neutropenia; has hemodialysis associated/line associated bacteremia or a history of hemodialysis associated/line associated bacteremia; has a wound; or any combination of the foregoing.
88. The method of any one of claims 81-87, further comprising administering to the subject or more antibiotic agent, or wherein the subject has received one or more antibiotic agent.
89. The method of claim 88, wherein the antibiotic agent comprises a penicillin, a fluoroquinolone, a cephalosporin, a macrolide, a beta-lactam with increased activity (e.g. amoxicillin-clavulanate), a tetracycline, a trimethoprim-sulfamethoxazole, a lincosamide (e.g. clindamycin), a urinary anti-infectives, or any combination thereof.
90. The method of claim 88 or 89, wherein the antibiotic agent comprises vancomycin, cefazolin, nafcillin, oxacillin, daptomycin, linezolid, penicillin, amoxicillin, azithromycin, erythromycin, cephalexin, cefdinir, ciproflaxin, levofloxacin, amoxicillin- clavulanate, ceftazidime/avibactam, tetracycline, doxycycline, trimethoprim-sulfamethoxazole, nitrofurantoin, clindamycin, or any combination thereof.
91. The antibody or antigen-binding fragment of any one of claims 1-57, the polynucleotide of any one of claims 58-65, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68, the host cell of claim 69, the human B cell of claim 70, and/or the composition or combination of any one of claims 71-77, for use in a method of treating or preventing a bacterial infection in a subject, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises S. aureus.
92. The antibody or antigen-binding fragment of any one of claims 1-57, the polynucleotide of any one of claims 58-65, the recombinant vector, DNA therapeutic, RNA or therapeutic of any one of claims 66-68, the host cell of claim 69, the human B cell of claim 70, and/or the composition or combination of any one of claims 71-77, for use in the preparation of a medicament for the treatment or prevention of a bacterial infection in a subject, wherein the
bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises S. aureus.
93. A method for in vitro diagnosis of a bacterial infection, the method comprising:
(i) contacting a sample from a subject with an antibody or antigen-binding fragment of any one of claims 1-57; and
(ii) detecting a complex comprising an antigen and the antibody, or comprising an antigen and the antigen-binding fragment, wherein the bacteria expresses the LTA, Gmd, ClfA, SpA, alpha toxin, leucocidin, gamma toxin, or Sbi, wherein, optionally, the bacteria comprises S. aureus.
94. The antibody, antigen-binding fragment, polynucleotide, recombinant vector, DNA therapeutic, RNA therapeutic, host cell, human B cell, or composition for use of claim 91 or 92, or the method of claim 93, wherein the bacteria comprises a bacteria of genus Staphylococcus and/or a bacteria of genus Enterococcus and/or a bacteria of genus Streptococcus and/or a bacteria of genus Lactococcus, wherein, optionally, the bacteria comprises: (1) S. aureus, wherein, further optionally, the S. aureus comprises a methicillin-resistant S. Aureus (MRSA); (2) a Coagulase-negative Staphylococcus (CoNS); (3) S. epidermidis,' (4) E. faecalis,' (5) S. pyogenes,' (6) S. agalactiae,' (7) L. garviae,' (8) S. pneumoniae,' (9) S. lugdunensis,' (10) A hominis,' (11) S. epidermidis,' or (12) any combination of ( 1 )-( 11).
95. A library comprising a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprising a plurality of polynucleotides encoding amino acid sequences encoded by any one or more of the following human immunoglobulin genes: IGHV3-23; IGHV3-7; IGHJ1; IGHJ3; IGLV3-21; IGKV1-5; IGKV1-6; IGLJ2; IGKJ2; IGKJ1; IGHV1-8; IGHV3-30; IGHV3-49; IGHV4-39; IGHJ6; IGHJ3; IGHJ4; IGKV1-27; IGLV3-21; IGKV1-5; IGKV1D-12; IGKV1-9; IGLV1-40; IGVK1-6; IGLV1-47; IGKJ1; IGLJ2; IGKJ2; IGKJ1; IGLJ1; IGHV1-3; IGHV1-18; IGHV3-11; IGHV3- 15; IGHV3-64; IGHV3-74; IGHV4-61; IGHV5-51; IGHJ5; IGHJ2; IGLV1-51; IGKV4-1; IGKV3-15; IGKV1-39; IGLV1-44; IGKV1-33; IGLV3-21; IGKV1-12; IGLV2-23; IGLV6-57; IGKV2-30; IGLJ3; IGKJ4; IGHV4-31; IGHV3-33; IGHV1-2; IGHV3-53; IGKV1-16; IGLV2- 14; IGKV3-11; IGKJ5; IGHV3-30-3; IGHV4-31; IGHV5-10-1; IGHV1-24; IGKV1-16; IGKV3-
20; IGLV3-21; IGHV1-69; IGHV3-43, or any combination thereof, preferably (1) IGHV3- 23/IGHJ3, (2) IGHV3-7/IGVK1-6, or (3) IGHV3-23.
96. The library of claim 95, wherein the human immunoglobulin genes in the library consist essentially of, or consist of, any one or more of the human immunoglobulin genes of claim 95.
97. The library of claim 95 or 96, wherein the library comprises a plurality of antibodies or antigen-binding fragments comprising amino acid sequences from any one or more of, and/or comprises a plurality of polynucleotides encoding amino acid sequences from any one or more of (i)-(xxiii): (i) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (ii) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ1; (iii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (iv) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (v) IGHV3-23, IGHJ1, IGLV3-21, and IGLJ2; (vi) IGHV3-23, IGHJ3, IGKV1-5, and IGKJ2; (vii) IGHV3-23, IGHJ4, IGKV1D-12, and IGKJ2; (viii) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ1; (ix) IGHV3-7, IGHJ3, IGKV1-6, and IGKJ2; (x) IGHV1-3, IGHJ4, IGLV1-51, and IGLJ1; (xi) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ1; (xii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ3; (xiii) IGHV3-74, IGHJ4, IGLV3-21, and IGLJ2; (xiv) IGHV5- 51, IGHJ4, IGLV6-57, and IGLFJ3; (xv) IGHV5-51, IGHJ6, IGKV2-30, and IGKJ4; (xvi) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ1; (xvii) IGHV3-33, IGHJ4, IGLV2-14, and IGLJ1; (xviii) IGHV4-31, IGHJ4, IGKV1-16, and IGKJ2; (xix) IGHV4-39, IGHJ4, IGKV3-11, and IGKJ5; (xx) IGHV1-2, IGHJ3, IGKV1-5, and IGKJ2; (xxi) IGHV3-53, IGHJ4, IGLV3-21, and IGLJ3; (xxii) IGHV4-61, IGHJ2, IGLV2-23, and IGLJ2; (xxiii) IGHV3-15, IGHJ6, IGLV1-44, and IGLJ3; (xxiv) IGHV3-30-3, IGHJ4, IGLV3-21, and IGLJ3; (xxv) IGHV4-31, IGHJ4, IGKV1-16, and, IGHJ4; (xxvi) IGHV5-10-1, IGHJ4, IGLV3-21, and IGLJ2; (xxvii) IGHV1-24, IGHJ4, IGKV3-20, and IGKJ4; (xxviii) IGHV1-8, IGHJ6, IGKV1-27, and IGKJ1; (xxix) IGHV3-30, IGHJ4, IGKV1-9, and IGKJ1; (xxx) IGHV3-49, IGHJ4, IGLV1-40, and IGLJ1; (xxxi) IGHV4-39, IGHJ4, IGLV1-47, and IGLJ2; (xxxii) IGHV1-3, IGHJ4, IGKV4-1, and IGKJ2; (xxxiii) IGHV1-18, IGHJ4, IGKV3-15, and IGKJ1; (xxxiv) IGHV3-11, IGHJ4, IGKV1- 39, and IGKJ2; (xxxv) IGHV3-64, IGHJ6, IGKV1-33, and IGKJ4; (xxxvi) IGHV4-39, IGHJ5, IGKV1-12, and IGKJ1; (xxxvii) IGHV5-51, IGHJ4, IGLV6-57, and IGLJ3; (xxxviii) IGHV1- 69, IGHJ4, IGLV6-57, and IGHJ4; (xxxix) IGHV4-31, IGHJ5, IGKV1-16, and IGHJ5; (xl) IGHV4-31, IGHJ4, IGKV1-16, and IGHJ4; (xli) IGHV3-33, IGHJ2, IGKV4-1, and IGKJ4; (xlii) IGHV3-43, IGHJ6, IGLV1-44, and IGLJ2; (xliii) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ3;
(xliv) IGHV5-10-1, IGHJ5, IGLV3-21, and IGLJ2; (xlv) IGHV3-49, IGHJ5, IGKV1-12, and IGKJ1; (xlvi) IGHV1-3, IGHJ3, IGKV1-5, and IGKJ2; (xlvii) IGHV3-23, IGHJ4, IGLV1-44, and IGLJ1; or (xlviii) IGHV3-23, IGHJ4, IGLV1-40, and IGLJ2.
98. The library of claim 97, wherein the plurality of antibodies or antigen-binding fragments, or the plurality of polynucleotides encoding the antibodies or antigen-binding fragments, further comprises or encodes additional amino acid sequences, wherein the additional amino acid sequences are optionally randomized and/or are limited to one or more CDR regions of an antibody or antigen-binding fragment.
99. A method comprising screening one or more antibodies of the library of any one of claims 104-107 for binding to a bacterial (e.g., S. aureus) antigen selected from: a lipoteichoic acid (LTA) (optionally a Type I, a Type II, and/or a Type IV LTA); a glucosaminidase (Gmd); a clumping factor A (ClfA); a Staphylococcus Protein A (SpA); an alpha toxin; a leukocidin; a gamma toxin; and a Sbi.
100. A kit comprising a liquid composition comprising and antibody or antigenbinding fragment of any one of claims 1-57, 91, 92, or 94, the polynucleotide of any one of claims 58-65, the recombinant vector, DNA therapeutic, or RNA therapeutic of any one of claims 66-68, the host cell of claim 69, the human B cell of claim 70, and/or the composition of any one of claim 71-77, and instructions for use thereof in treating a bacterial (e.g., Staphylococcus) infection in a subject.
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363512279P | 2023-07-06 | 2023-07-06 | |
| US63/512,279 | 2023-07-06 | ||
| US202363609814P | 2023-12-13 | 2023-12-13 | |
| US63/609,814 | 2023-12-13 | ||
| US202463639554P | 2024-04-26 | 2024-04-26 | |
| US63/639,554 | 2024-04-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025010424A1 true WO2025010424A1 (en) | 2025-01-09 |
Family
ID=92106502
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/036949 Pending WO2025010424A1 (en) | 2023-07-06 | 2024-07-05 | Antibodies against staphylococcus antigens and methods of using the same |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025010424A1 (en) |
Citations (43)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4751180A (en) | 1985-03-28 | 1988-06-14 | Chiron Corporation | Expression using fused genes providing for protein product |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US4935233A (en) | 1985-12-02 | 1990-06-19 | G. D. Searle And Company | Covalently linked polypeptide cell modulators |
| US5283173A (en) | 1990-01-24 | 1994-02-01 | The Research Foundation Of State University Of New York | System to detect protein-protein interactions |
| WO1996014339A1 (en) | 1994-11-05 | 1996-05-17 | The Wellcome Foundation Limited | Antibodies |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US5648237A (en) | 1991-09-19 | 1997-07-15 | Genentech, Inc. | Expression of functional antibody fragments |
| US5770429A (en) | 1990-08-29 | 1998-06-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5789199A (en) | 1994-11-03 | 1998-08-04 | Genentech, Inc. | Process for bacterial production of polypeptides |
| US5840523A (en) | 1995-03-01 | 1998-11-24 | Genetech, Inc. | Methods and compositions for secretion of heterologous polypeptides |
| US5959177A (en) | 1989-10-27 | 1999-09-28 | The Scripps Research Institute | Transgenic plants expressing assembled secretory antibodies |
| US6040498A (en) | 1998-08-11 | 2000-03-21 | North Caroline State University | Genetically engineered duckweed |
| US6420548B1 (en) | 1999-10-04 | 2002-07-16 | Medicago Inc. | Method for regulating transcription of foreign genes |
| US6596541B2 (en) | 2000-10-31 | 2003-07-22 | Regeneron Pharmaceuticals, Inc. | Methods of modifying eukaryotic cells |
| WO2004042017A2 (en) | 2002-10-31 | 2004-05-21 | Genentech, Inc. | Methods and compositions for increasing antibody production |
| WO2004076677A2 (en) | 2003-02-26 | 2004-09-10 | Institute For Research In Biomedicine | Monoclonal antibody production by ebv transformation of b cells |
| WO2005018572A2 (en) | 2003-08-22 | 2005-03-03 | Biogen Idec Ma Inc. | Improved antibodies having altered effector function and methods for making the same |
| WO2006034488A2 (en) | 2004-09-23 | 2006-03-30 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| US7049426B2 (en) | 1999-06-10 | 2006-05-23 | Abgenix, Inc. | Transgenic animals for producing specific isotypes of human antibodies via non-cognate switch regions |
| WO2006074397A2 (en) | 2005-01-05 | 2006-07-13 | Biogen Idec Ma Inc. | Cripto binding molecules |
| US7125978B1 (en) | 1999-10-04 | 2006-10-24 | Medicago Inc. | Promoter for regulating expression of foreign genes |
| WO2008038024A1 (en) | 2006-09-29 | 2008-04-03 | Ucb Pharma S.A. | Altered antibodies |
| US7364738B2 (en) | 2001-01-26 | 2008-04-29 | Inhibitex, Inc. | Monoclonal antibodies to the CLFA protein and method of use in treating infections |
| WO2008070593A2 (en) | 2006-12-01 | 2008-06-12 | Seattle Genetics, Inc. | Variant target binding agents and uses thereof |
| WO2008116118A2 (en) * | 2007-03-20 | 2008-09-25 | Avanir Pharmaceuticals | Fully human antibodies to gram positive bacteria |
| US7498415B2 (en) | 2003-09-24 | 2009-03-03 | Kyowa Hakko Kogyo Co., Ltd. | Recombinant antibody against human insulin-like growth factor |
| WO2009092011A1 (en) | 2008-01-18 | 2009-07-23 | Medimmune, Llc | Cysteine engineered antibodies for site-specific conjugation |
| WO2010141902A2 (en) | 2009-06-04 | 2010-12-09 | Novartis Ag | METHODS FOR IDENTIFICATION OF SITES FOR IgG CONJUGATION |
| WO2011005481A1 (en) | 2009-06-22 | 2011-01-13 | Medimmune, Llc | ENGINEERED Fc REGIONS FOR SITE-SPECIFIC CONJUGATION |
| WO2011156328A1 (en) | 2010-06-08 | 2011-12-15 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| US8119772B2 (en) | 2006-09-29 | 2012-02-21 | California Institute Of Technology | MART-1 T cell receptors |
| WO2012032181A2 (en) | 2010-09-10 | 2012-03-15 | Allozyne, Inc | Novel antibody derivatives |
| US8258268B2 (en) | 2005-08-19 | 2012-09-04 | Abbott Laboratories | Dual variable domain immunoglobulin and uses thereof |
| WO2013093809A1 (en) | 2011-12-23 | 2013-06-27 | Pfizer Inc. | Engineered antibody constant regions for site-specific conjugation and methods and uses therefor |
| WO2015103072A1 (en) | 2013-12-30 | 2015-07-09 | Epimab Biotherapeutics | Fabs-in-tandem immunoglobulin and uses thereof |
| WO2015157595A1 (en) | 2014-04-11 | 2015-10-15 | Medimmune, Llc | Conjugated compounds comprising cysteine-engineered antibodies |
| WO2016040856A2 (en) | 2014-09-12 | 2016-03-17 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| WO2016181357A1 (en) | 2015-05-13 | 2016-11-17 | Zumutor Biologics, Inc. | Afucosylated protein, cell expressing said protein and associated methods |
| US20180179267A1 (en) | 2015-04-17 | 2018-06-28 | Arsanis Biosciences Gmbh | Anti-staphylococcus aureus antibody combination preparation |
| WO2019024979A1 (en) | 2017-07-31 | 2019-02-07 | Institute For Research In Biomedicine | Antibodies with functional domains in the elbow region |
| WO2019057122A1 (en) | 2017-09-22 | 2019-03-28 | Wuxi Biologics (Shanghai) Co., Ltd. | Novel bispecific polypeptide complexes |
| WO2020191306A1 (en) | 2019-03-21 | 2020-09-24 | Immunogen, Inc. | Methods of preparing cell-binding agent-drug conjugates |
| WO2022251119A2 (en) | 2021-05-24 | 2022-12-01 | Vir Biotechnology, Inc. | Engineered polypeptides |
-
2024
- 2024-07-05 WO PCT/US2024/036949 patent/WO2025010424A1/en active Pending
Patent Citations (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US4751180A (en) | 1985-03-28 | 1988-06-14 | Chiron Corporation | Expression using fused genes providing for protein product |
| US4935233A (en) | 1985-12-02 | 1990-06-19 | G. D. Searle And Company | Covalently linked polypeptide cell modulators |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US6417429B1 (en) | 1989-10-27 | 2002-07-09 | The Scripps Research Institute | Transgenic plants expressing assembled secretory antibodies |
| US5959177A (en) | 1989-10-27 | 1999-09-28 | The Scripps Research Institute | Transgenic plants expressing assembled secretory antibodies |
| US5283173A (en) | 1990-01-24 | 1994-02-01 | The Research Foundation Of State University Of New York | System to detect protein-protein interactions |
| US5468614A (en) | 1990-01-24 | 1995-11-21 | The Research Foundation Of State University Of New York | System to detect protein-protein interactions |
| US5770429A (en) | 1990-08-29 | 1998-06-23 | Genpharm International, Inc. | Transgenic non-human animals capable of producing heterologous antibodies |
| US5648237A (en) | 1991-09-19 | 1997-07-15 | Genentech, Inc. | Expression of functional antibody fragments |
| US5789199A (en) | 1994-11-03 | 1998-08-04 | Genentech, Inc. | Process for bacterial production of polypeptides |
| WO1996014339A1 (en) | 1994-11-05 | 1996-05-17 | The Wellcome Foundation Limited | Antibodies |
| US5840523A (en) | 1995-03-01 | 1998-11-24 | Genetech, Inc. | Methods and compositions for secretion of heterologous polypeptides |
| US6040498A (en) | 1998-08-11 | 2000-03-21 | North Caroline State University | Genetically engineered duckweed |
| US7049426B2 (en) | 1999-06-10 | 2006-05-23 | Abgenix, Inc. | Transgenic animals for producing specific isotypes of human antibodies via non-cognate switch regions |
| US7125978B1 (en) | 1999-10-04 | 2006-10-24 | Medicago Inc. | Promoter for regulating expression of foreign genes |
| US6420548B1 (en) | 1999-10-04 | 2002-07-16 | Medicago Inc. | Method for regulating transcription of foreign genes |
| US6596541B2 (en) | 2000-10-31 | 2003-07-22 | Regeneron Pharmaceuticals, Inc. | Methods of modifying eukaryotic cells |
| US7364738B2 (en) | 2001-01-26 | 2008-04-29 | Inhibitex, Inc. | Monoclonal antibodies to the CLFA protein and method of use in treating infections |
| WO2004042017A2 (en) | 2002-10-31 | 2004-05-21 | Genentech, Inc. | Methods and compositions for increasing antibody production |
| WO2004076677A2 (en) | 2003-02-26 | 2004-09-10 | Institute For Research In Biomedicine | Monoclonal antibody production by ebv transformation of b cells |
| WO2005018572A2 (en) | 2003-08-22 | 2005-03-03 | Biogen Idec Ma Inc. | Improved antibodies having altered effector function and methods for making the same |
| US7498415B2 (en) | 2003-09-24 | 2009-03-03 | Kyowa Hakko Kogyo Co., Ltd. | Recombinant antibody against human insulin-like growth factor |
| WO2006034488A2 (en) | 2004-09-23 | 2006-03-30 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| WO2006074397A2 (en) | 2005-01-05 | 2006-07-13 | Biogen Idec Ma Inc. | Cripto binding molecules |
| US8258268B2 (en) | 2005-08-19 | 2012-09-04 | Abbott Laboratories | Dual variable domain immunoglobulin and uses thereof |
| WO2008038024A1 (en) | 2006-09-29 | 2008-04-03 | Ucb Pharma S.A. | Altered antibodies |
| US8119772B2 (en) | 2006-09-29 | 2012-02-21 | California Institute Of Technology | MART-1 T cell receptors |
| WO2008070593A2 (en) | 2006-12-01 | 2008-06-12 | Seattle Genetics, Inc. | Variant target binding agents and uses thereof |
| WO2008116118A2 (en) * | 2007-03-20 | 2008-09-25 | Avanir Pharmaceuticals | Fully human antibodies to gram positive bacteria |
| WO2009092011A1 (en) | 2008-01-18 | 2009-07-23 | Medimmune, Llc | Cysteine engineered antibodies for site-specific conjugation |
| WO2010141902A2 (en) | 2009-06-04 | 2010-12-09 | Novartis Ag | METHODS FOR IDENTIFICATION OF SITES FOR IgG CONJUGATION |
| WO2011005481A1 (en) | 2009-06-22 | 2011-01-13 | Medimmune, Llc | ENGINEERED Fc REGIONS FOR SITE-SPECIFIC CONJUGATION |
| WO2011156328A1 (en) | 2010-06-08 | 2011-12-15 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| WO2012032181A2 (en) | 2010-09-10 | 2012-03-15 | Allozyne, Inc | Novel antibody derivatives |
| WO2013093809A1 (en) | 2011-12-23 | 2013-06-27 | Pfizer Inc. | Engineered antibody constant regions for site-specific conjugation and methods and uses therefor |
| WO2015103072A1 (en) | 2013-12-30 | 2015-07-09 | Epimab Biotherapeutics | Fabs-in-tandem immunoglobulin and uses thereof |
| WO2015157595A1 (en) | 2014-04-11 | 2015-10-15 | Medimmune, Llc | Conjugated compounds comprising cysteine-engineered antibodies |
| WO2016040856A2 (en) | 2014-09-12 | 2016-03-17 | Genentech, Inc. | Cysteine engineered antibodies and conjugates |
| US20180179267A1 (en) | 2015-04-17 | 2018-06-28 | Arsanis Biosciences Gmbh | Anti-staphylococcus aureus antibody combination preparation |
| WO2016181357A1 (en) | 2015-05-13 | 2016-11-17 | Zumutor Biologics, Inc. | Afucosylated protein, cell expressing said protein and associated methods |
| WO2019024979A1 (en) | 2017-07-31 | 2019-02-07 | Institute For Research In Biomedicine | Antibodies with functional domains in the elbow region |
| WO2019025391A1 (en) | 2017-07-31 | 2019-02-07 | Institute For Research In Biomedicine | Antibodies with functional domains in the elbow region between variable adn constant domain |
| WO2019057122A1 (en) | 2017-09-22 | 2019-03-28 | Wuxi Biologics (Shanghai) Co., Ltd. | Novel bispecific polypeptide complexes |
| WO2020191306A1 (en) | 2019-03-21 | 2020-09-24 | Immunogen, Inc. | Methods of preparing cell-binding agent-drug conjugates |
| WO2022251119A2 (en) | 2021-05-24 | 2022-12-01 | Vir Biotechnology, Inc. | Engineered polypeptides |
Non-Patent Citations (102)
| Title |
|---|
| "Remington: The Science and Practice of Pharmacy", 2000, PHILADELPHIA COLLEGE OF PHARMACY AND SCIENCE |
| ABHINANDANMARTIN, MOL IMMUNOL, vol. 45, 2008, pages 3832 - 9 |
| AHMED ET AL., J. STRUC. BIOL., vol. 194, no. 1, 2016, pages 78 |
| ALTSCHUL ET AL., NUCLEIC ACIDS RES, vol. 25, 1997, pages 3389 - 3402 |
| ARMOUR, K. L. ET AL., EUR. J. IMMUNOL., vol. 29, 1999, pages 2613 - 2624 |
| BIOINFORMATICS, vol. 15, 2016, pages 298 - 300 |
| BRINKMANNKONTERMANN, MABS, vol. 9, no. 2, 2017, pages 182 - 212 |
| BRUHNS ET AL., BLOOD, vol. 113, 2009, pages 3716 - 3725 |
| BRUNGAUZY-LAZO, METHODS MOL. BIOL., vol. 1045, 2013, pages 173 - 187 |
| BURTON, D. R., MOL. IMMUNOL., vol. 22, 1985, pages 161 - 206 |
| CAPEL, P. J. ET AL., IMMUNOMETHODS, vol. 113, 1994, pages 269 - 315 |
| CHAUDHARY ET AL., PROC. NATL. ACAD. SCI. USA, vol. 87, 1990, pages 1066 - 1070 |
| CHEN ET AL., PNAS, vol. 119, no. 4, 2022, pages e2114478119 |
| CHOTHIALESK, J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| CHU, S. ET AL.: "Accelerated Clearance of IgE In Chimpanzees Is Mediated By Xmab7195, An Fc-Engineered Antibody With Enhanced Affinity For Inhibitory Receptor FcγRIIb", AM J RESPIR CRIT, 2014 |
| CHU, S. Y. ET AL.: "Inhibition of b cell receptor-mediated activation of primary human B cells by co-engagement of CD19 and FcgammaRIIb with Fc-engineered antibodies", MOLECULAR IMMUNOLOGY, vol. 45, 2008, pages 3926 - 3933, XP002498116, DOI: 10.1016/j.molimm.2008.06.027 |
| CLACKSON ET AL., NATURE, vol. 352, 1991, pages 624 - 628 |
| COFFIN, J. M. ET AL.: "Fundamental Virology", 1996, LIPPINCOTT-RAVEN PUBLISHERS, article "Retroviridae: The viruses and their replication" |
| COREY ET AL., MOLECULAR THERAPY METHODS & CLINICAL DEVELOPMENT, vol. 28, 2023, pages 1 - 10 |
| DE HAAS, M. ET AL., J LAB. CLIN. MED., vol. 126, 1995, pages 330 - 341 |
| DELILLORAVETCH, CELL, vol. 161, no. 5, 2015, pages 1035 - 1045 |
| DUNCAN, A. R.WINTER, G., NATURE, vol. 332, 1988, pages 323 - 327 |
| ELLIOTT ET AL., NPJ VACCINES, 2017, pages 18 |
| ENGELS ET AL., HUM. GENE THER., vol. 14, 2003, pages 1155 |
| EYLER ET AL., PNAS, vol. 116, no. 46, 2019, pages 23068 - 23071 |
| FLINGAI ET AL., SCI REP, vol. 5, 2015, pages 12616 |
| FRECHA ET AL., MOL. THER., vol. 18, 2010, pages 1748 |
| GANESAN, L. P. ET AL.: "FcyRIIb on liver sinusoidal endothelium clears small immune complexes", JOURNAL OF IMMUNOLOGY, vol. 189, 2012, pages 4981 - 4988, XP002724347, DOI: 10.4049/jimmunol.1202017 |
| GANESAN, L. P. ET AL.: "FcγRIIb on liver sinusoidal endothelium clears small immune complexes", JOURNAL OF IMMUNOLOGY, vol. 189, 2012, pages 4981 - 4988, XP002724347, DOI: 10.4049/jimmunol.1202017 |
| GERNGROSS, NAT. BIOTECH., vol. 22, 2004, pages 1409 - 1414 |
| GESSNER, J. E. ET AL., ANN. HEMATOL., vol. 76, 1998, pages 231 - 248 |
| GEURTS ET AL., MOL. THER., vol. 8, 2003, pages 108 |
| HAQUE ET AL., CHEM. COMMUN., vol. 57, 2021, pages 10689 - 10702 |
| HONEGGERPLUCKTHUN, J. MOL. BIO., vol. 309, 2001, pages 657 - 670 |
| HUANG ET AL., MABS, vol. 6, 2018, pages 1 - 12 |
| JENDEBERG ET AL., J IMMUNOL METHODS, vol. 201, no. 1, 1997, pages 25 - 34 |
| JOLLY, D J.: "The Development of Human Gene Therapy", 1999, COLD SPRING HARBOR LAB, article "Emerging Viral Vectors", pages: 209 - 40 |
| JONES ET AL., NATURE, vol. 321, 1986, pages 522 - 525 |
| KIM ET AL., J EXP MED, vol. 207, no. 9, 2010, pages 1863 - 1870 |
| KOHLER ET AL., NATURE, vol. 256, 1975, pages 495 |
| KOSE ET AL., SCI. IMMUNOL., vol. 4, 2019, pages eaaw6647 |
| KRISKY ET AL., GENE THER, vol. 5, 1998, pages 1517 |
| LEFRANC ET AL., DEV. COMP. IMMUNOL., vol. 27, 2003, pages 55 |
| LEFRANC, M.-P.LEFRANC, G.: "Immunogenetics", vol. 882, 2012, HUMANA PRESS, article "Human Gm, Km and Am allotypes and their molecular characterization: a remarkable demonstration of polymorphism", pages: 635 - 680 |
| LI ET AL., NAT. BIOTECH., vol. 24, 2006, pages 210 - 215 |
| LI ET AL., WILERY INTERDISCIP REV. NANOMED NANOBIOTECHNOL, vol. 11, no. 2, 2019, pages e1530 |
| LI ET AL., WILERY INTERDISCIP REV. NANOMEDNANOBIOTECHNOL, vol. 11, no. 2, 2019, pages e1530 |
| LI, JQ., ZHANG, ZR., ZHANG, HQ: "Intranasal delivery of replicating mRNA encoding neutralizing antibody against SARS-CoV-2 infection in mice", SIG TRANSDUCT TARGET THER, vol. 6, 2021, pages 369, Retrieved from the Internet <URL:https://doi.org/10.1038/s41392-021-00783-1> |
| LIU ET AL., MABS, vol. 6, no. 5, 2014, pages 1145 - 1154 |
| MARATEA ET AL., GENE, vol. 40, 1985, pages 39 - 46 |
| MARKS ET AL., J. MOL. BIOL., vol. 222, 1991, pages 581 - 597 |
| MATES ET AL., NAT. GENET., vol. 41, 2009, pages 753 |
| MEYER ET AL., BIOCONJUG. CHEM., vol. 27, no. 12, 2016, pages 2791 - 2807 |
| MOORE ET AL., MABS, vol. 2, no. 2, 2010, pages 181 - 189 |
| MORRISON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 81, 1984, pages 6851 - 6855 |
| MURPHY ET AL., PROC. NATL. ACAD. SCI. USA, vol. 83, 1986, pages 8258 - 8262 |
| MUTHUMANI ET AL., HUM VACCIN IMMUNOTHER, vol. 9, 2013, pages 2253 - 2262 |
| MUTHUMANI ET AL., J INFECT DIS, vol. 214, no. 3, 2016, pages 369 - 378 |
| NANCEMEIER, ACS CENT. SCI., vol. 7, no. 5, 2021, pages 748 - 756 |
| NARESHKUMAR ET AL., PHARM. RES., vol. 32, 2015, pages 3526 - 3540 |
| NEGA ET AL., COMMUN BIOL, vol. 3, no. 1, 2020, pages 695 |
| NIE SIWEI ET AL: "Biology drives the discovery of bispecific antibodies as innovative therapeutics", ANTIBODY THERAPEUTICS, vol. 3, no. 1, 1 January 2020 (2020-01-01), pages 18 - 62, XP055776258, Retrieved from the Internet <URL:http://academic.oup.com/abt/article-pdf/3/1/18/34842496/tbaa003.pdf> DOI: 10.1093/abt/tbaa003 * |
| NORTH ET AL., J MOL BIOL., vol. 406, 2011, pages 228 - 56 |
| PALMBERGER ET AL., J. BIOTECHNOL., vol. 153, no. 3-4, 2011, pages 160 - 166 |
| PARDI ET AL., J CONTROL RELEASE, 2015, pages 351 |
| PARDI ET AL., J CONTROLRELEASE, 2015, pages 217345 - 351 |
| PATELKAUFMAN, EXPERT OPIN BIOL THER, vol. 15, no. 4, 2015, pages 595 - 600 |
| PLUCKTHUN, A, BIO/TECHNOLOGY, vol. 9, 1991, pages 545 - 551 |
| PRESTA, CURR. OP. STRUCT. BIOL., vol. 2, 1992, pages 593 - 596 |
| RAAFAT DINA ET AL: "FightingStaphylococcus aureusBiofilms with Monoclonal Antibodies", TRENDS IN MICROBIOLOGY, vol. 27, no. 4, 1 April 2019 (2019-04-01), pages 303 - 322, XP085628085, ISSN: 0966-842X, DOI: 10.1016/J.TIM.2018.12.009 * |
| RAVETCH, J. V.KINET, J. P., ANNU. REV. IMMUNOL., vol. 9, 1991, pages 457 - 492 |
| SABNIS ET AL., MOL. THER., vol. 26, 2018, pages 1509 - 1519 |
| SAMBROOK ET AL.: "A Laboratory Manual", 1989, COLD SPRING HARBOR LABORATORY, article "Molecular Cloning" |
| SANG ET AL., METHODS MOL. BIOL., vol. 2078, 2020, pages 235 - 250 |
| SCATCHARD ET AL., ANN. N.Y. ACAD. SCI., vol. 51, 1949, pages 660 |
| SHIELDS, R. L. ET AL., J. BIOL. CHEM., vol. 276, 2001, pages 6591 - 6604 |
| SPIESS ET AL., MOL. IMMUNOL, vol. 67, no. 2, 2015, pages 95 |
| STICKLER ET AL., GENES IMMUN, vol. 12, no. 3, April 2011 (2011-04-01), pages 213 - 221 |
| SUZUKI ET AL., CLIN. CANCER RES, vol. 13, no. 6, 2007, pages 1875 - 82 |
| TABOR ET AL., ANTIMICROB AGENTS CHEMOTHER., vol. 60, no. 9, 2016, pages 5312 - 21 |
| THESS ET AL., MOL THER, vol. 23, 2015, pages 1456 - 1464 |
| THRAN ET AL., EMBO MOL MED, vol. 9, no. 10, 2017, pages 1434 - 1448 |
| THRAN ET AL., EMBO MOLMED, vol. 9, no. 10, 2017, pages 1434 - 1448 |
| URLAUB ET AL., PNAS, vol. 77, 1980, pages 4216 |
| VAN DE WINKEL, J. G.ANDERSON, C. L., J. LEUKOC. BIOL., vol. 49, 1991, pages 511 - 524 |
| VAN HOECKEROOSE, J. TRANSLATIONAL MED, vol. 17, 2019, pages 54, Retrieved from the Internet <URL:https://doi.org/10.1186/s12967-019-1804-8> |
| VARRONE ET AL., J ORTHOP RES, vol. 32, no. 10, 2014, pages 1389 - 96 |
| VARSHNEY AKKUZMICHEVA GALIN J ET AL., PLOS ONE, vol. 13, no. 1, 24 January 2018 (2018-01-24), pages e0190537 |
| VERHOEYEN ET AL., METHODS MOL. BIOL., vol. 506, 2009, pages 97 |
| VERHOEYEN ET AL., SCIENCE, vol. 239, 1988, pages 1534 - 1536 |
| WALCHLI ET AL., PLOS ONE, vol. 6, 2011, pages 327930 |
| WARD, E. S.GHETIE, V., THER. IMMUNOL., vol. 2, 1995, pages 77 - 94 |
| WEEMS JJ JRSTEINBERG JPFILLER S ET AL., ANTIMICROB AGENTS CHEMOTHER, vol. 50, no. 8, 2006, pages 2751 - 2755 |
| WILSON, SCIENCE, vol. 295, 2002, pages 2103 |
| WINES, B.D. ET AL., J. IMMUNOL., vol. 164, 2000, pages 5313 - 5318 |
| WOLFF ET AL., CANCER RES, vol. 53, 1993, pages 2560 |
| YAZAKIWU: "Methods in Molecular Biology", vol. 248, 2003, HUMANA PRESS, pages: 255 - 268 |
| YODER ET AL., MOL. PHARMACEUTICALS, vol. 16, no. 9, 2019, pages 3926 - 3927 |
| YUJIA Z. ET AL.: "Principles of RNA methylation and their implications for biology and medicine", BIOMEDICINE & PHARMACOTHERAPY, vol. 131, 2020, pages 110731, XP086322129, DOI: 10.1016/j.biopha.2020.110731 |
| ZHANG ET AL., FRONT. IMMUNOL., 2019 |
| ZHANG ET AL., FRONT. IMMUNOL., DOI=10.3389/FIMMU.2019.00594, 2019 |
| ZHAO ET AL., J. IMMUNOL., vol. 174, 2005, pages 4415 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11168128B2 (en) | Antibodies against SARS-CoV-2 and methods of using the same | |
| US20250263466A1 (en) | Antibodies against sars-cov-2 and methods of using the same | |
| CA3177169A1 (en) | Antibodies against sars-cov-2 | |
| CA3218489A1 (en) | Engineered polypeptides | |
| WO2022204202A1 (en) | Antibodies that bind to multiple sarbecoviruses | |
| CA3199023A1 (en) | Broadly neutralizing antibodies against influenza neuraminidase | |
| CA3199429A1 (en) | Anti-influenza antibodies and combinations thereof | |
| CA3194162A1 (en) | Antibodies against sars-cov-2 | |
| CA3197537A1 (en) | Antibodies against influenza a viruses | |
| WO2024006472A1 (en) | Antibodies that bind to multiple sarbecoviruses | |
| EP4529464A2 (en) | Broadly neutralizing antibodies against influenza neuraminidase | |
| WO2025010424A1 (en) | Antibodies against staphylococcus antigens and methods of using the same | |
| WO2024118998A2 (en) | Engineered anti-sars-cov-2 antibodies and methods of using the same | |
| WO2024112818A1 (en) | Engineered anti-sars-cov-2 antibodies and uses thereof | |
| WO2023245078A1 (en) | Anti-parvovirus antibodies and uses thereof | |
| HK40059225B (en) | Antibodies against sars-cov-2 | |
| HK40098455A (en) | Antibodies against sars-cov-2 | |
| HK40059225A (en) | Antibodies against sars-cov-2 | |
| TW202210505A (en) | Antibodies against sars-cov-2 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24748503 Country of ref document: EP Kind code of ref document: A1 |