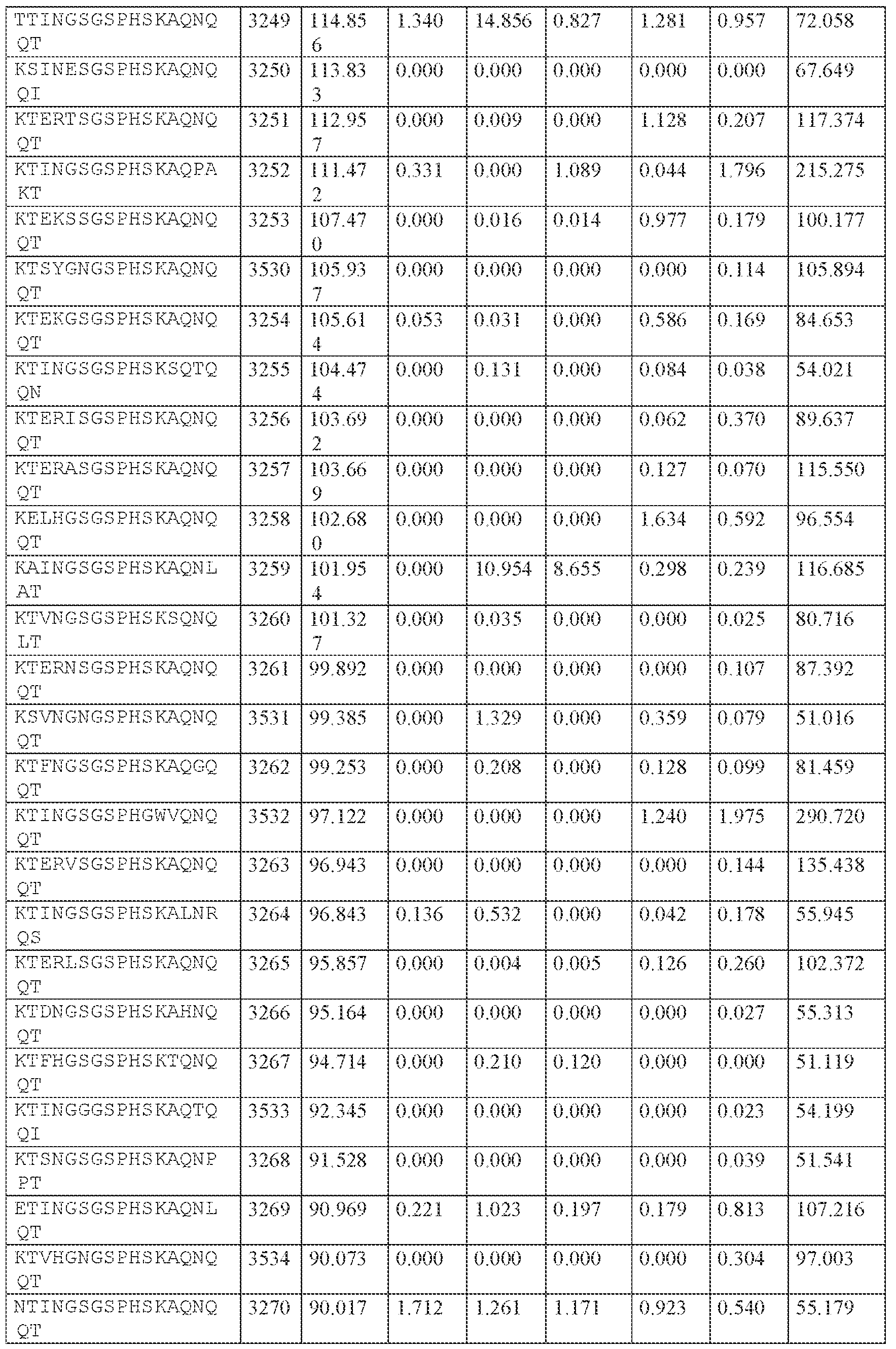
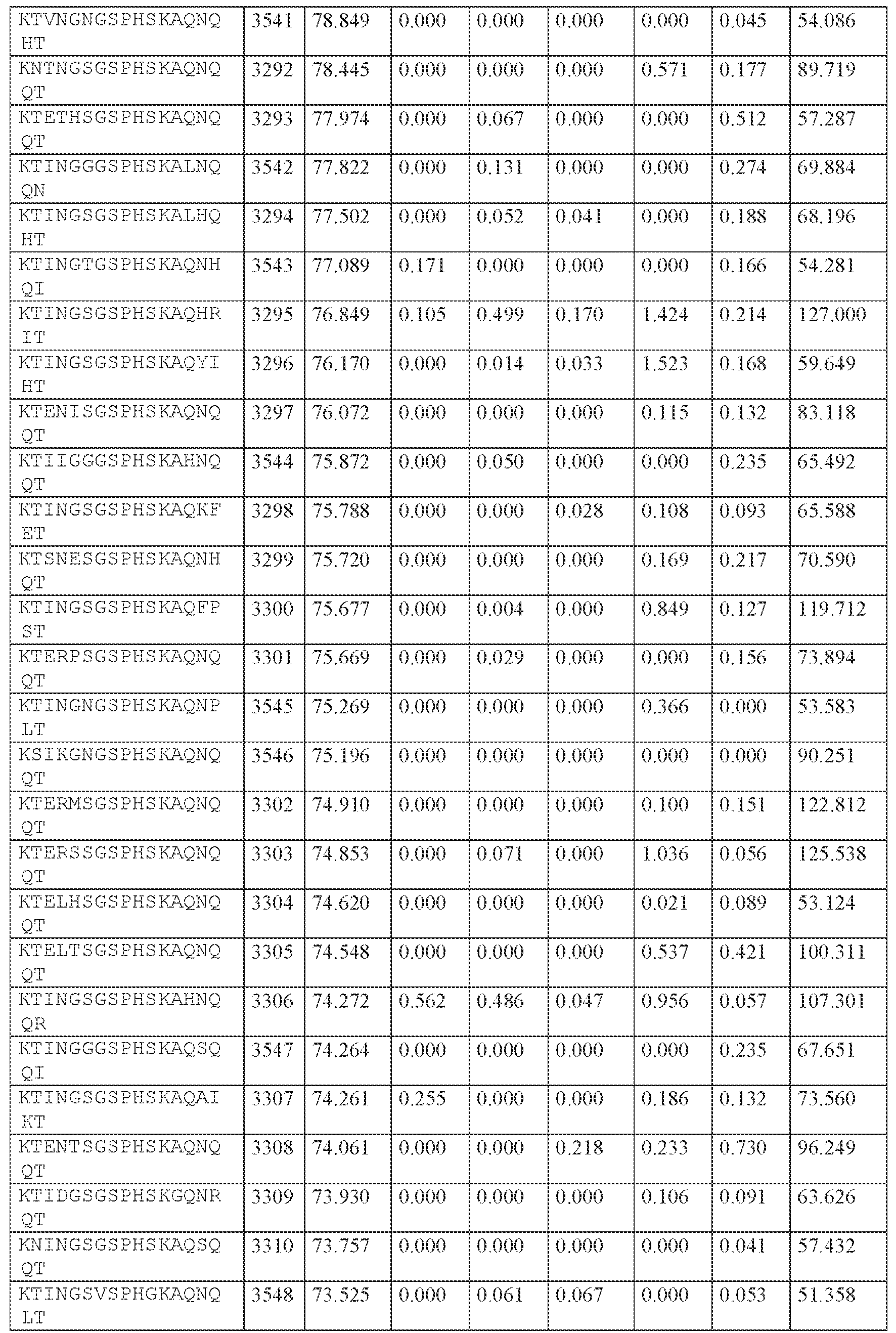
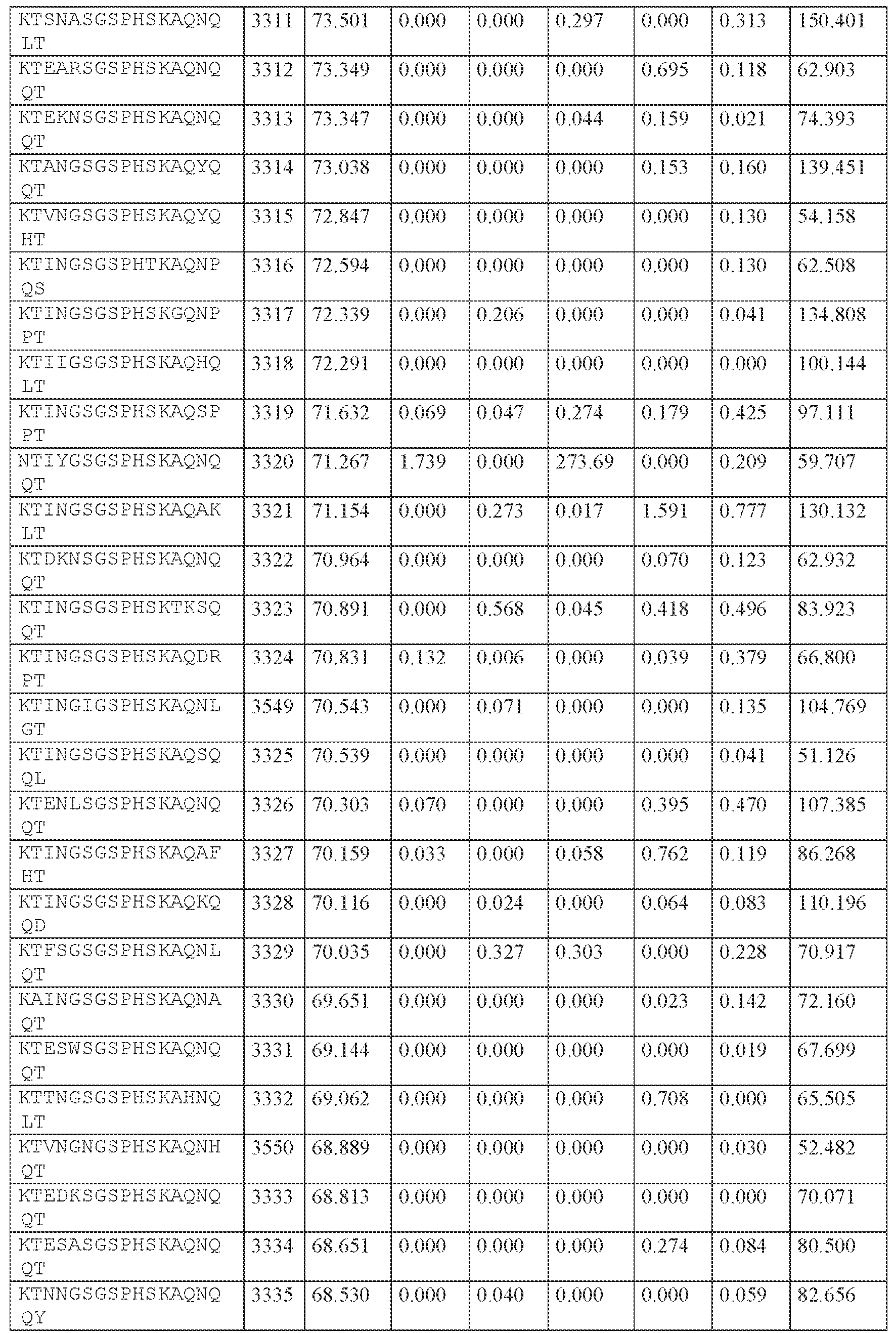
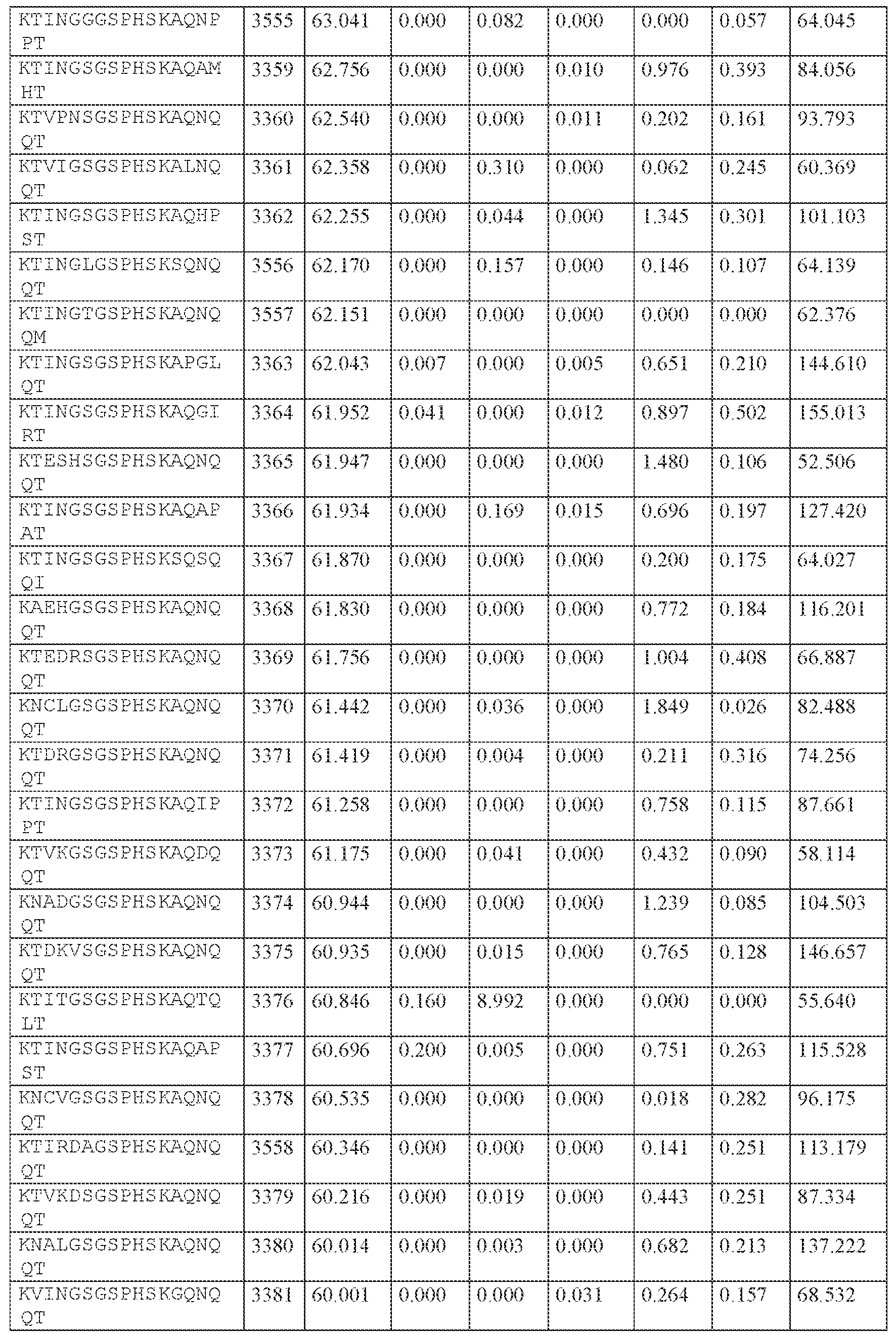
WO2024229173A2 - Compositions and methods for the treatment of disorders related to ataxin-2 - Google Patents
Compositions and methods for the treatment of disorders related to ataxin-2 Download PDFInfo
- Publication number
- WO2024229173A2 WO2024229173A2 PCT/US2024/027327 US2024027327W WO2024229173A2 WO 2024229173 A2 WO2024229173 A2 WO 2024229173A2 US 2024027327 W US2024027327 W US 2024027327W WO 2024229173 A2 WO2024229173 A2 WO 2024229173A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- amino acid
- acid sequence
- aav particle
- aav
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14122—New viral proteins or individual genes, new structural or functional aspects of known viral proteins or genes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14143—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14145—Special targeting system for viral vectors
Definitions
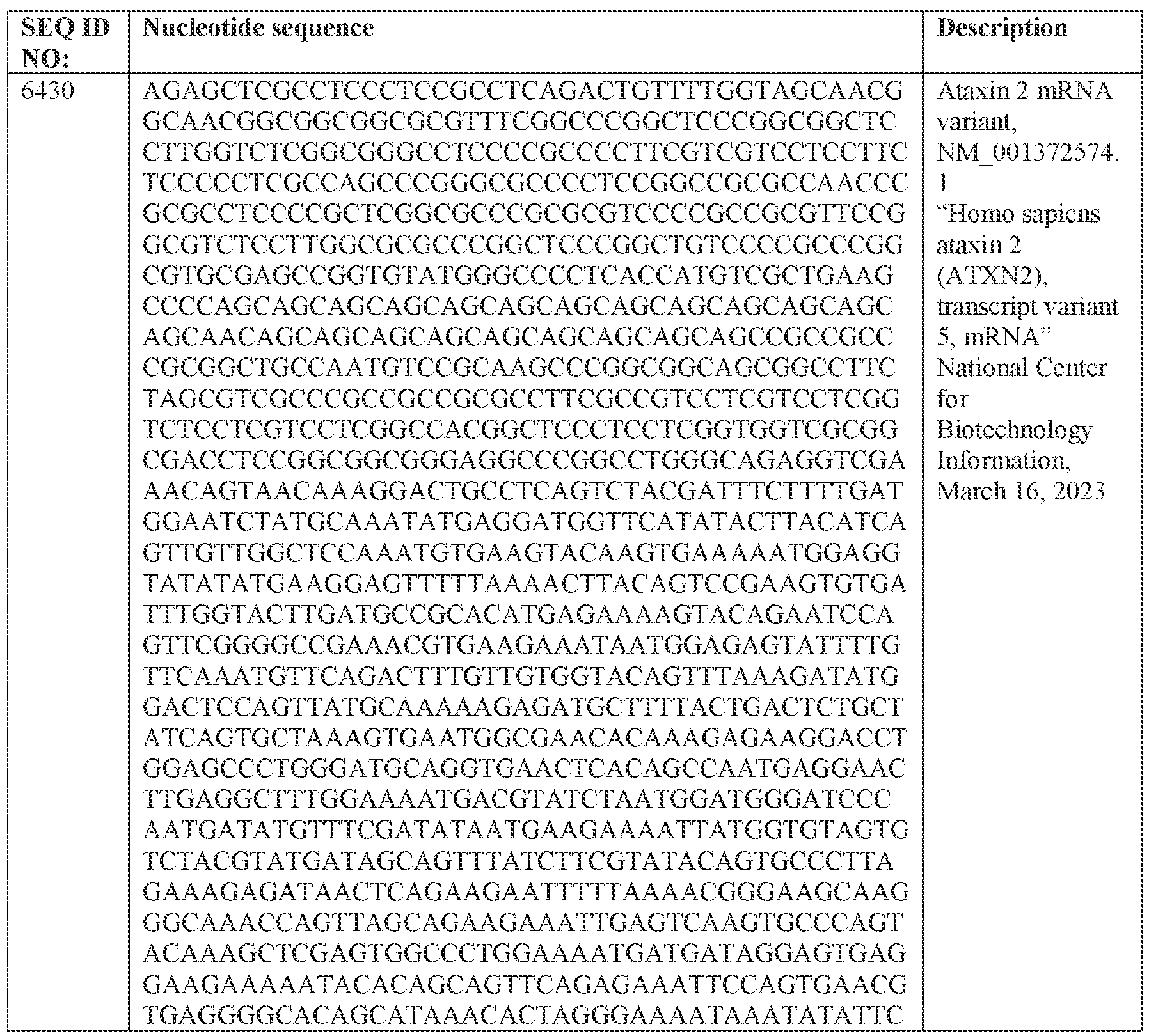
- compositions and methods relating to adeno-associated virus (AAV) viral particles for the delivery- of polynucleotides e.g., modulatory polynucleotides for reducing or eliminating expression of ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2. protein
- AAV adeno-associated virus
- SCA2 spinocerebellar ataxia type 2
- compositions described herein may be used to treat a subject in need thereof, such as a human subject diagnosed with SCA2 or another condition resulting from mutant ATXN2 protein or aberrant ATXN2 protein expression or activity.
- Ataxin-2 is a protein encoded by the ATXN2 gene (Ensembl Gene ID No. EN SG00000204842), which is also known as ATX2 and TNR.C 13. It is located on chromosome 12 of the human genome.
- Ataxin-2 The function of Ataxin-2 is not known, but it is found in the cytoplasm, and is thought to interact with the endoplasmic reticulum.
- ATXN2 mutations cause disease in human subjects.
- ATXN2 mutations are known to cause spinocerebellar ataxia type 2 (SCA2).
- SCA2 is an autosomal dominant cerebellar ataxia.
- ATXN2 mutations also play a role in other ATXN2-related disorders, including amyotrophic lateral sclerosis (ALS), Parkinson’s disease (PD), and frontotemporal lobar degeneration (FTLD).
- ALS amyotrophic lateral sclerosis
- PD Parkinson’s disease
- FTLD frontotemporal lobar degeneration
- SCA2 is typically caused by CAG trinucleotide repeat expansions in the ATXN2 gene on I2q24.1. Patients with SCA2 have ATXN2 alleles with 32 or more trinucleotide repeats, resulting in ATXN2 protein with polyQ expansions. PolyQ expansions in ATXN2 (e.g., intermediate length polyQ expansions) have been associated with other diseases, like ALS, as well.
- the age of onset of SCA2 is thought to correlate with the number of CAG trinucleotide repeats. Onset is typically later than age 60 in subjects with 32-34 CAG repeats (SEQ ID NO: 6432). Onset is Apically between ages 20 and. 60 in subjects with 35-40 CAG repeats (SEQ ID NO: 6433). Onset is typically before age 20 for patients with 45 or more CAG repeats (SEQ ID NO: 6434).
- Mutated ATXN2 is thought to produce an Ataxin-2. protein with an abnormally long stretch of glutamine residues. The cellular impact of mutant Ataxin-2 protein is not confirmed. Without wishing to be bound by any particular theory, mutant Ataxin-2 may accumulate in cells and cause cell death.
- SCA2 is characterized by progressive cerebellar ataxia, including nystagmus, and slow saccadic eye movements. It may also present with ophthalmoparesis and/or parkinsonism. Later stages of SCA2 mainly involve the brainstem and cerebellum.
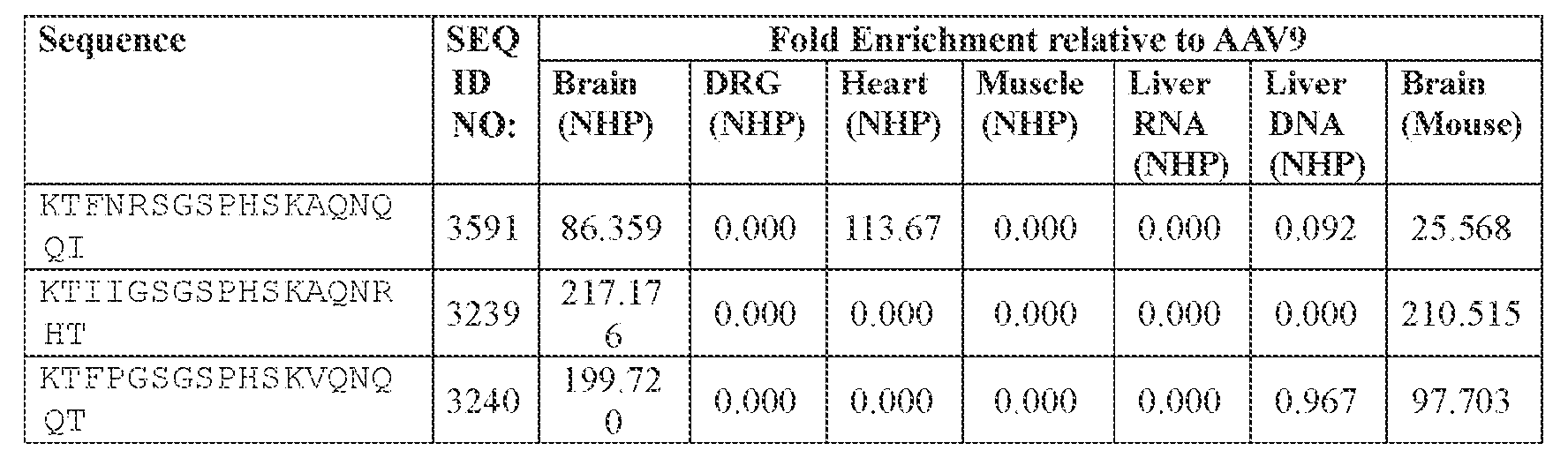
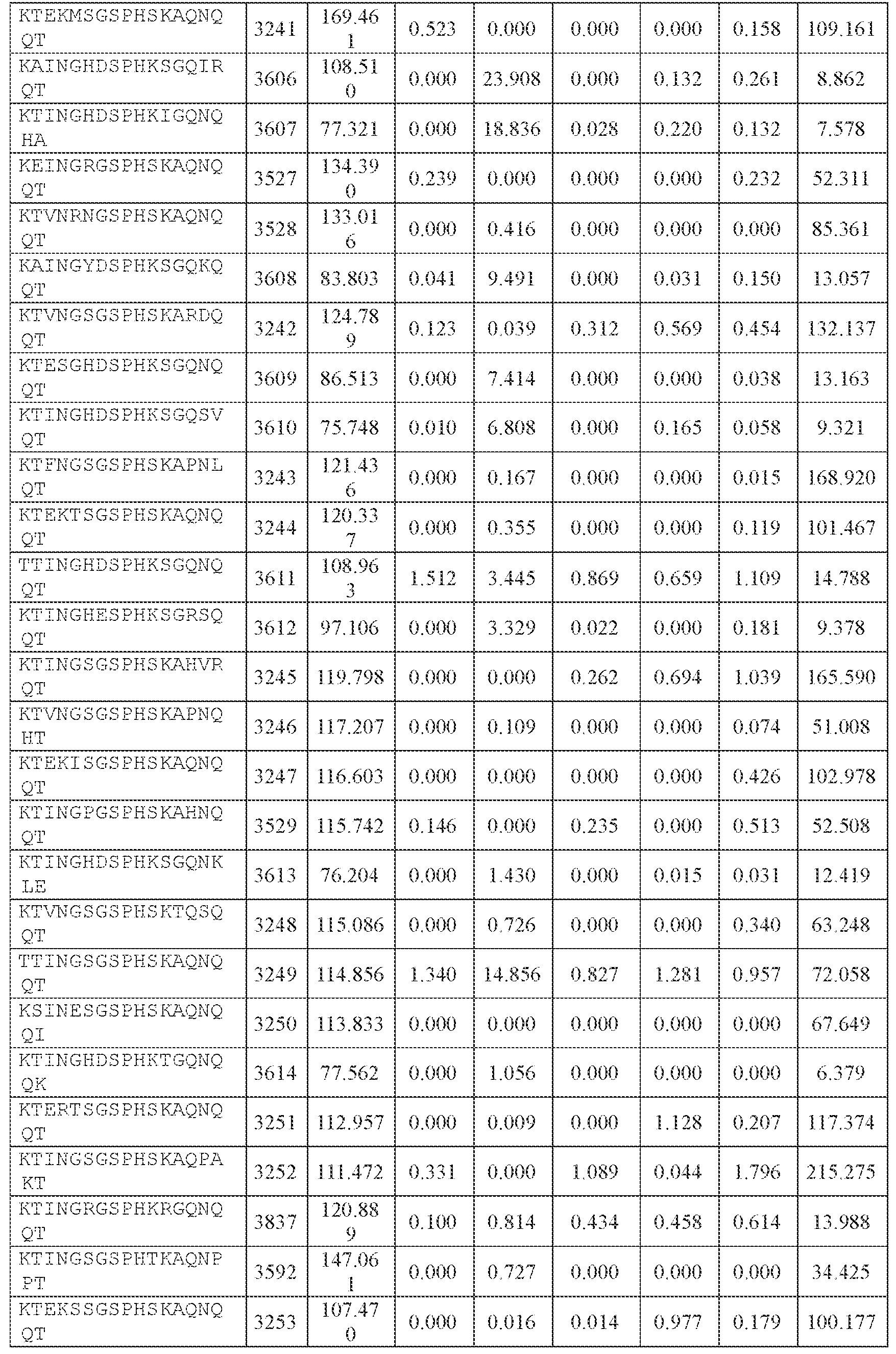
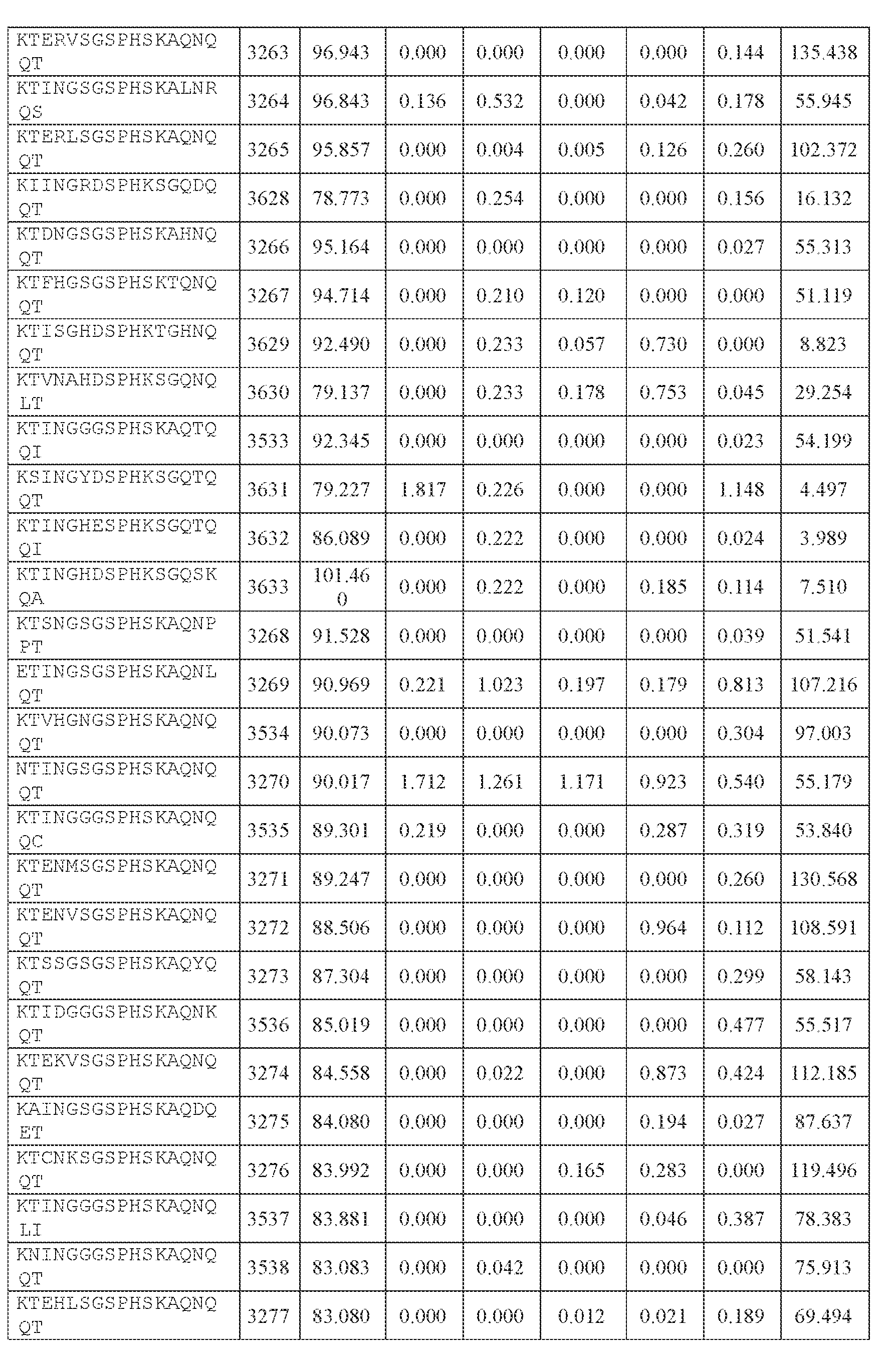
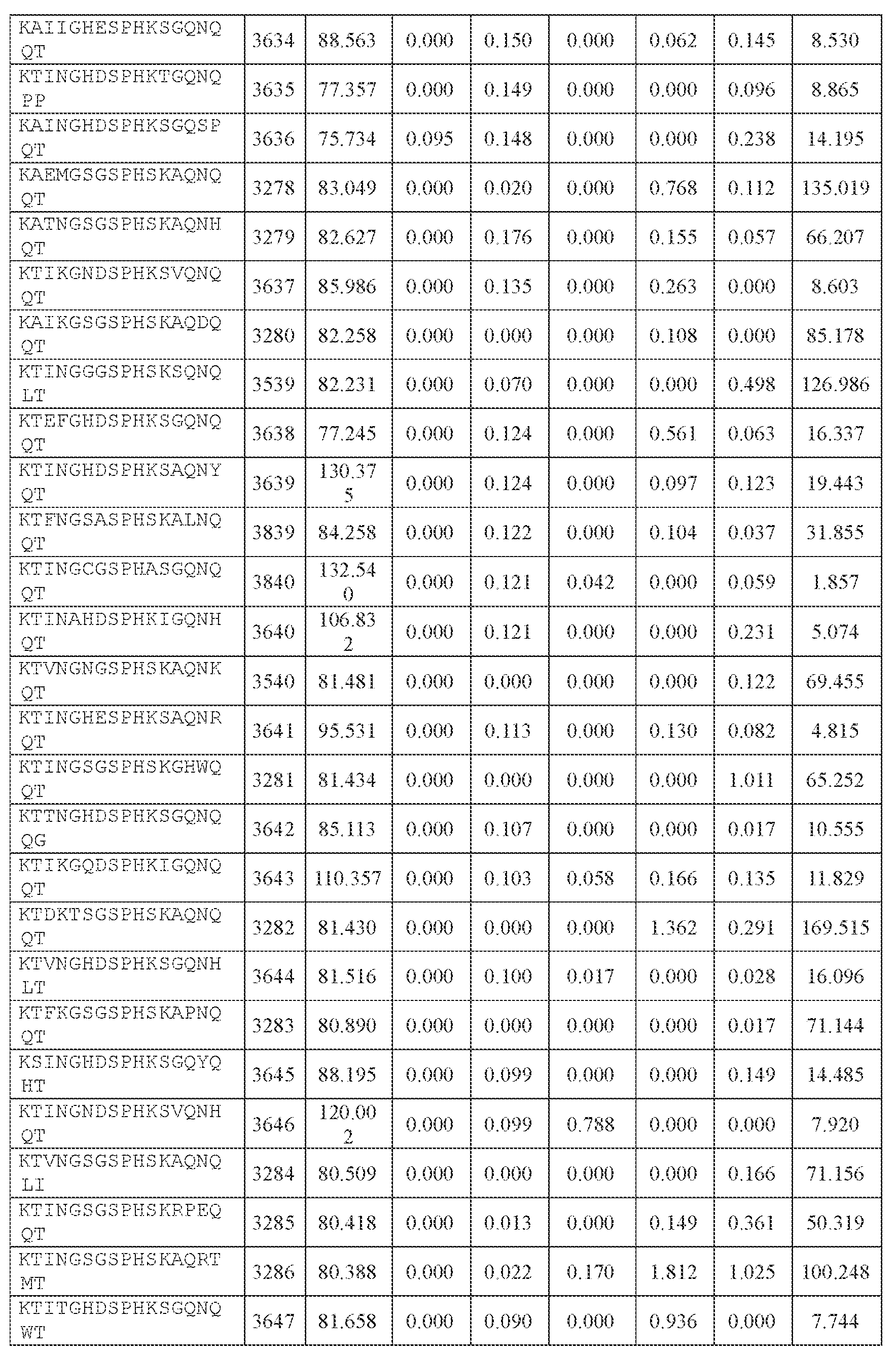
- AAV capsid variants that are capable of delivering a modulatory polynucleotide for reducing or eliminating expression of human ATXN2 mRNA (thereby reducing or eliminating ATXN2 protein), to a target cell or tissue, e.g., a CNS cell or tissue.
- compositions and methods directed to AAV-based gene delivery of modulatory polynucleotides for reducing or eliminating expression of ATXN2 to treat SCA2.
- the compositions and methods reduce or eliminate expression of ATXN2 mRNA, and thereby reduce or eliminate ATXN2 protein, to treat SCA2.
- the compositions and methods are useful to reduce the effects of mutant ATXN2 or aberrant ATXN2 protein expression or activity, and to slow, halt or reverse muscular and other symptoms of SCA2.
- aberrant ATXN2 expression refers to expression of ATXN2 comprising 32 or more CAG trinucleotide repeats (SEQ ID NO: 6435) (and thus polyQ expansions).
- ATXN2, ATX2, and TNRC13 are synonymous terms and are used interchangeably to refer to ATXN2.
- ATXN2 refers to the transcript encoding the ATXN2 (Ataxin 2) protein
- ATXN2 protein refers to the protein encoded by the ATXN2 gene and mRNA.
- SCA2 is used to refer to spinocerebellar ataxia type 2.
- the present disclosure provides an A AV particle comprising an AAV capsid and a nucleotide sequence encoding a modulatory polynucleotide tor reducing or eliminating expression of ATXN2.
- the modulatory polynucleotide reduces or eliminates mRNA encoding ATXN2.
- the modulatory polynucleotide reduces or eliminates ATXN2 protein (e.g., a human ATXN2 protein).
- the AAV capsid is an AAV capsid variant.
- the AAV capsid variant is an AAV9 capsid variant.
- the AAV capsid variant is an AAV9 capsid variant comprising a peptide insert in the loop IV region.
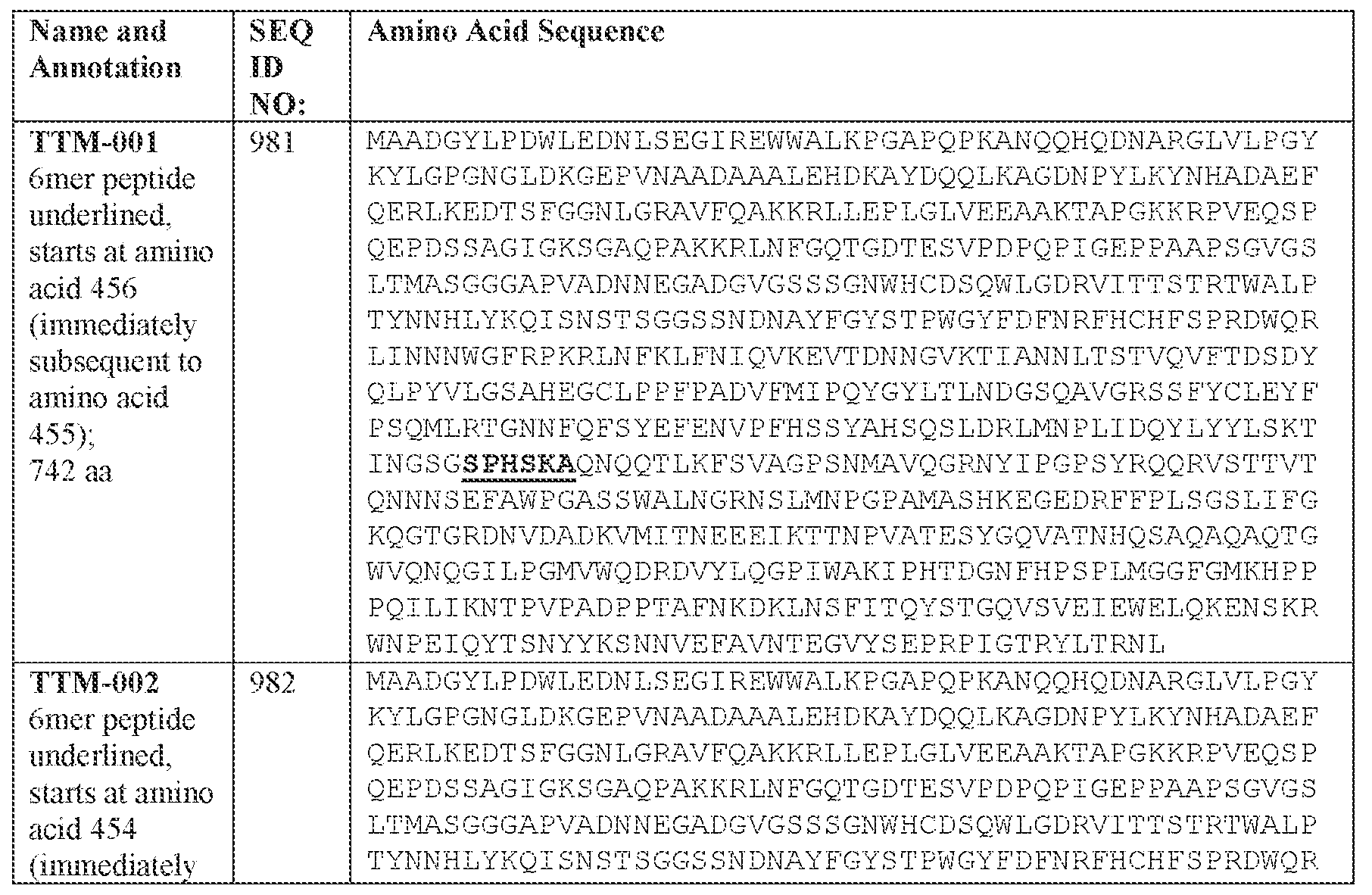
- the AAV capsid variant comprises the amino acid sequence of SPH in loop IV.
- the AAV capsid variant comprises the amino acid sequence of SPH in loop IV wherein the amino acid sequence (SPH) is present immediately subsequent to position 455 as numbered according to SEQ ID NO: 138.
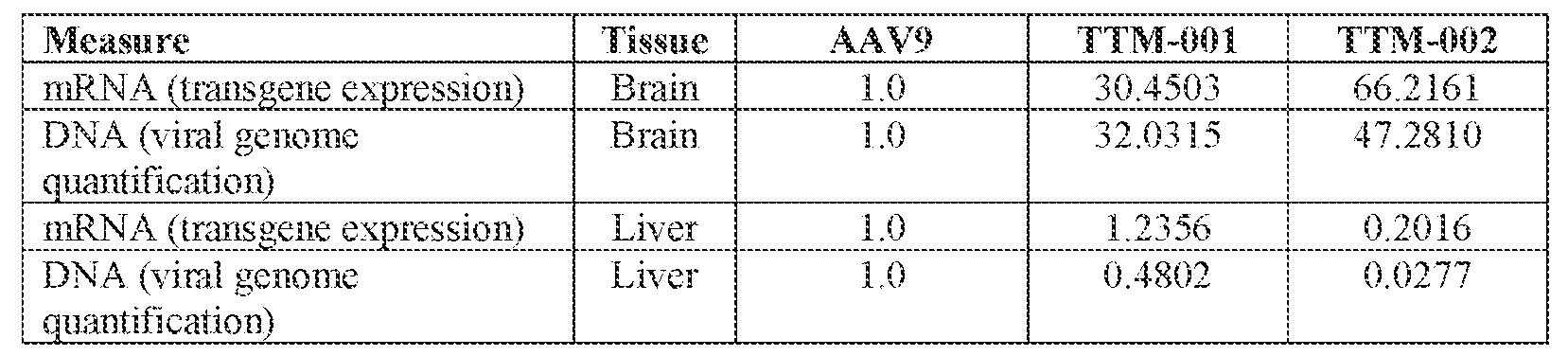
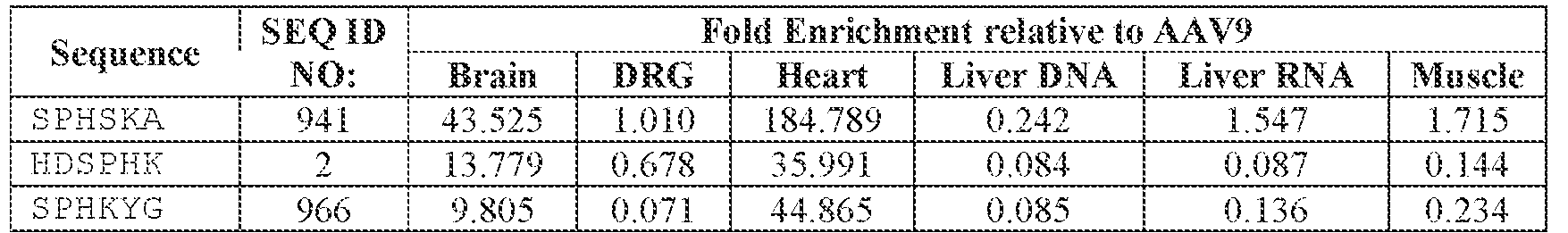
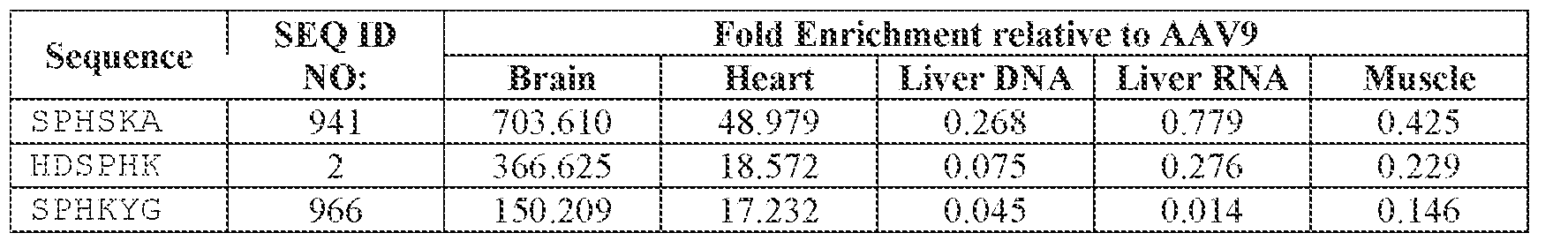
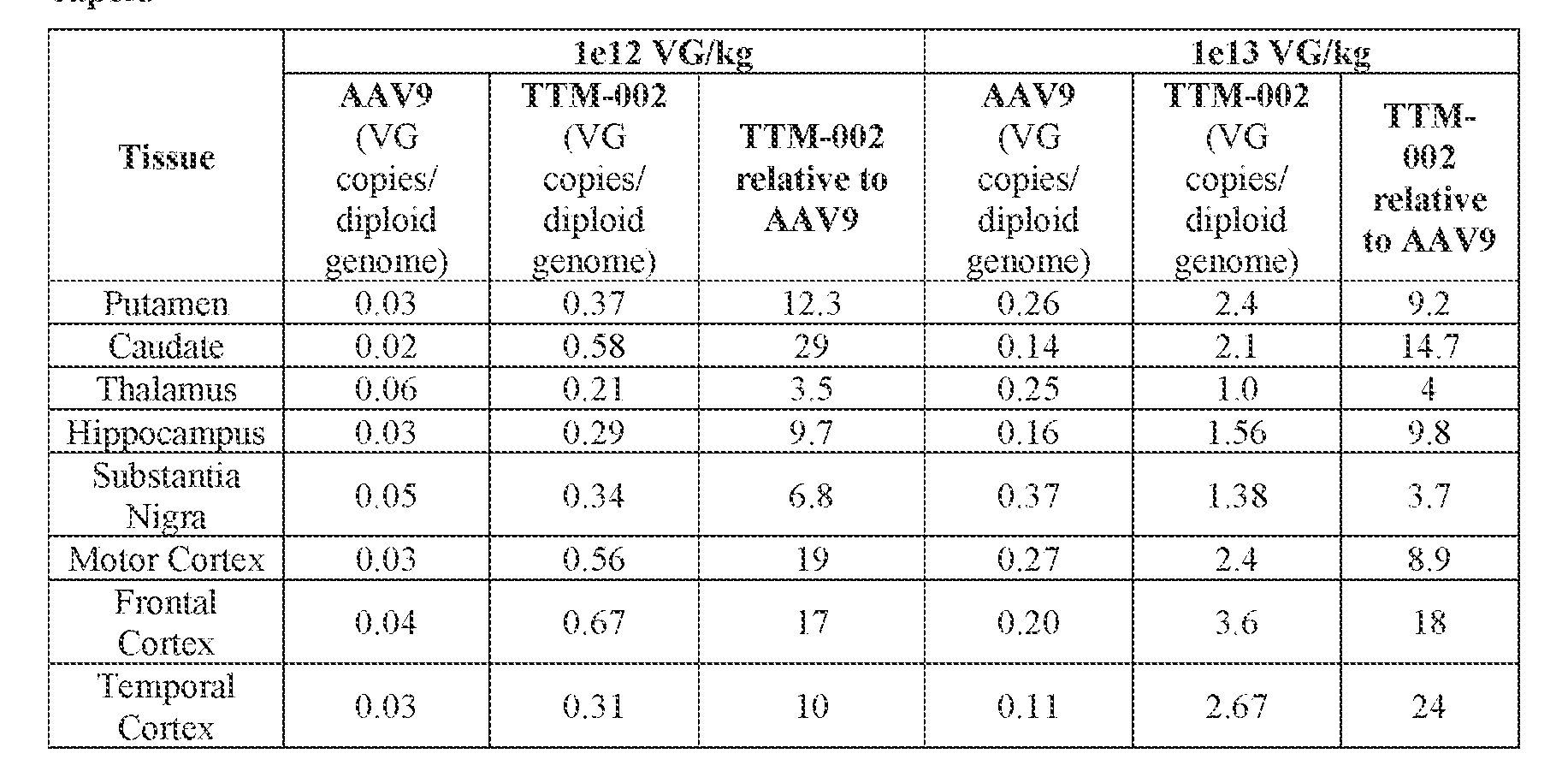
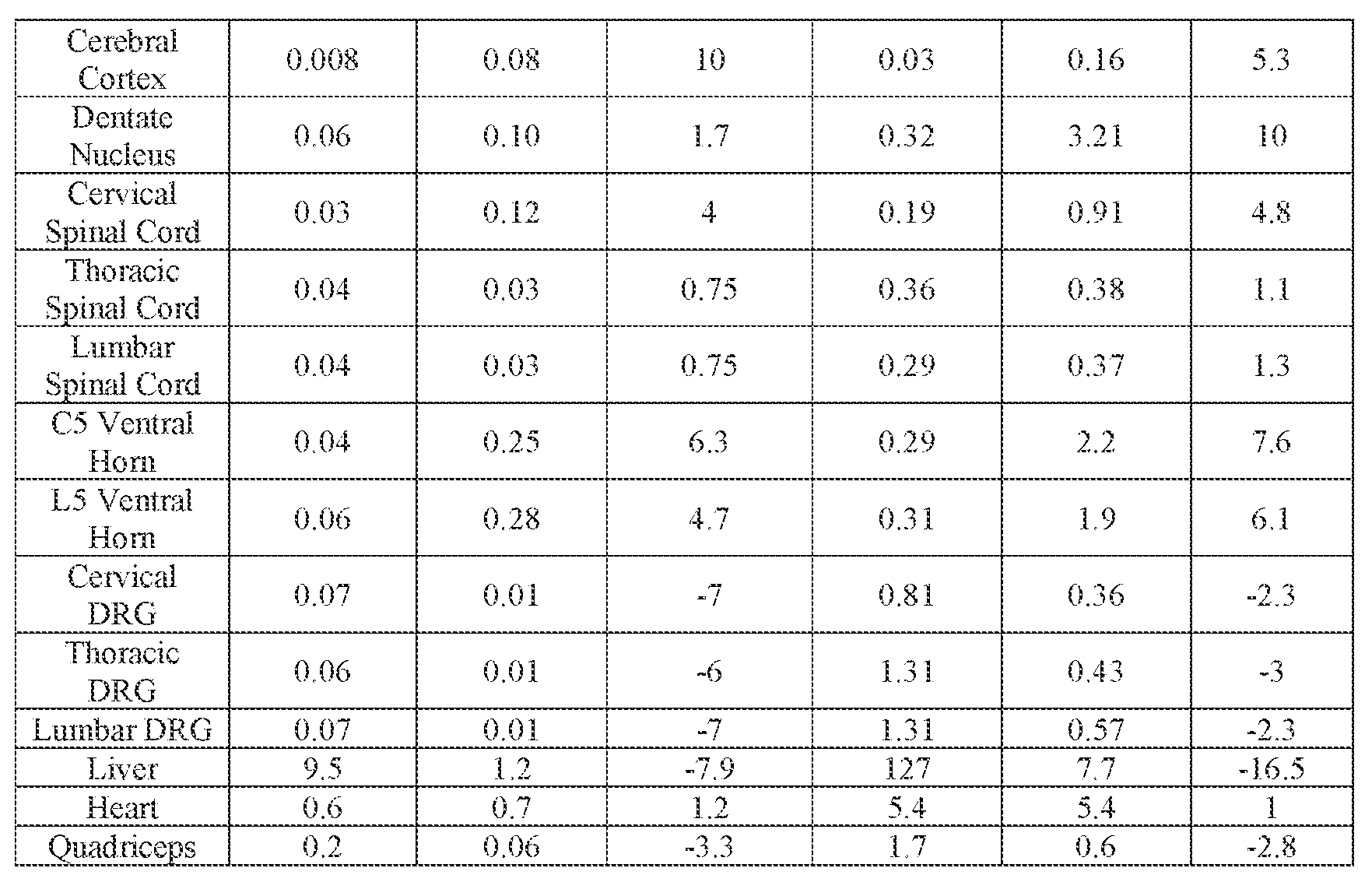
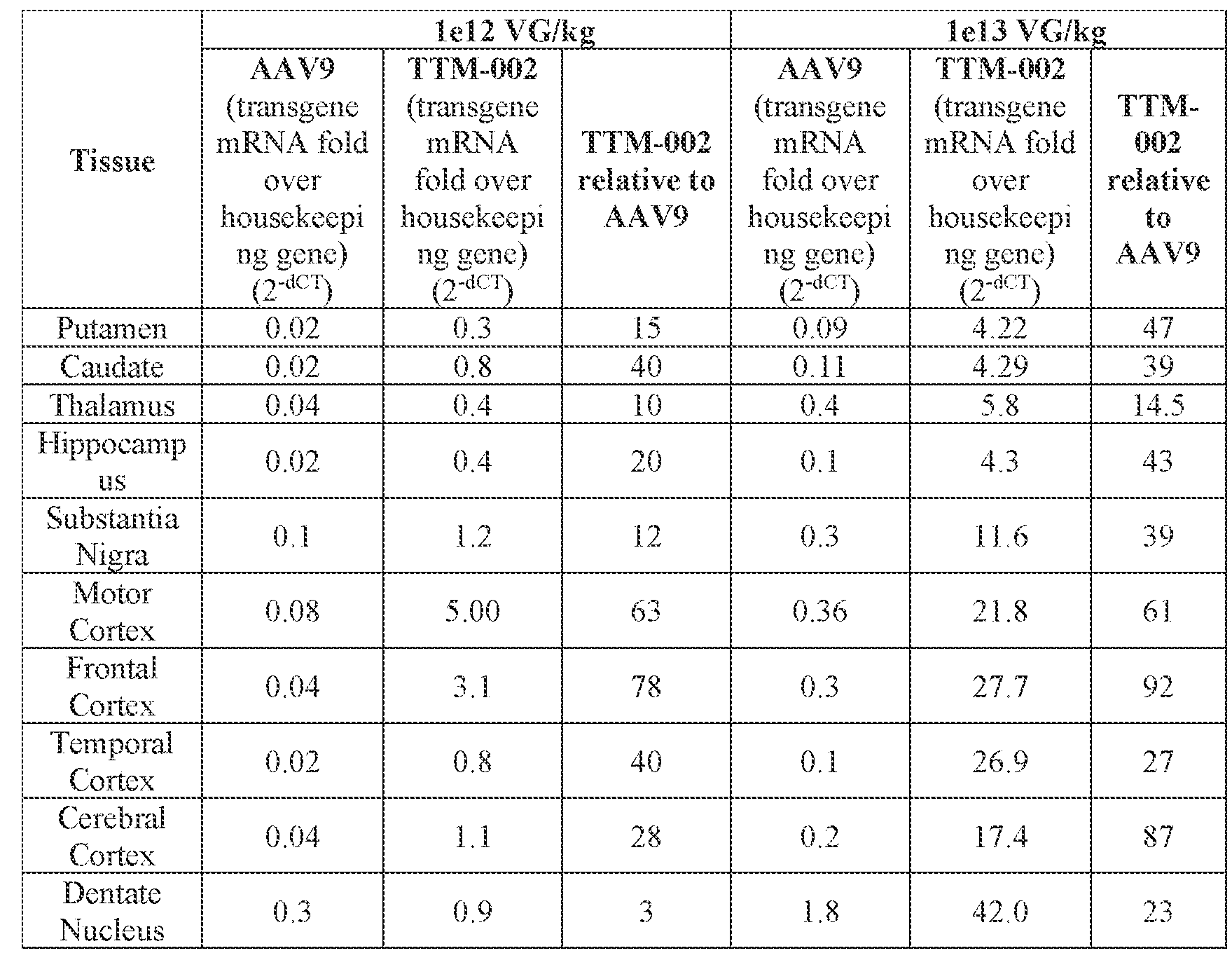
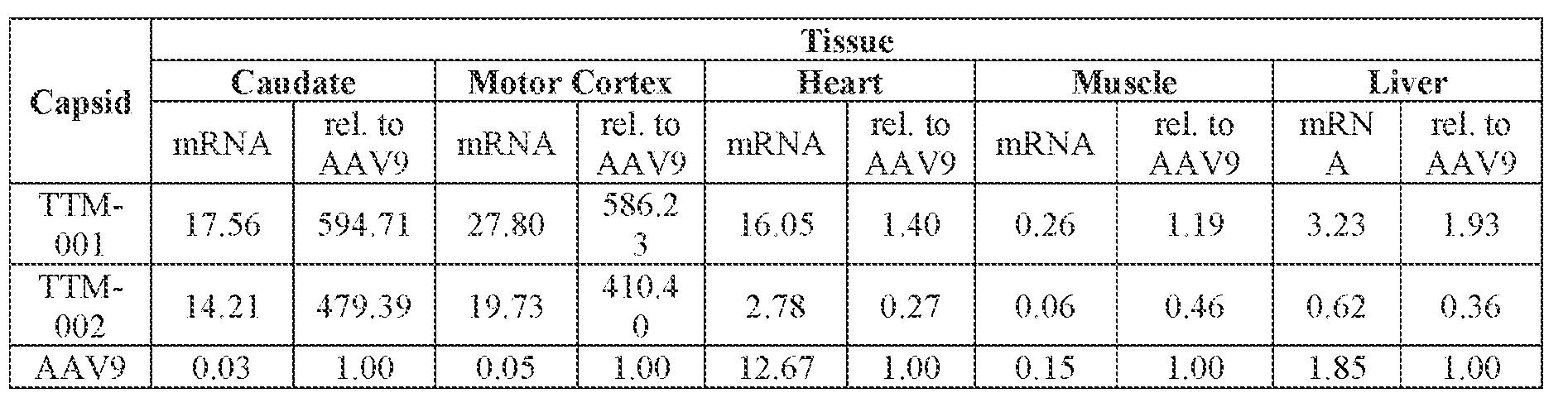
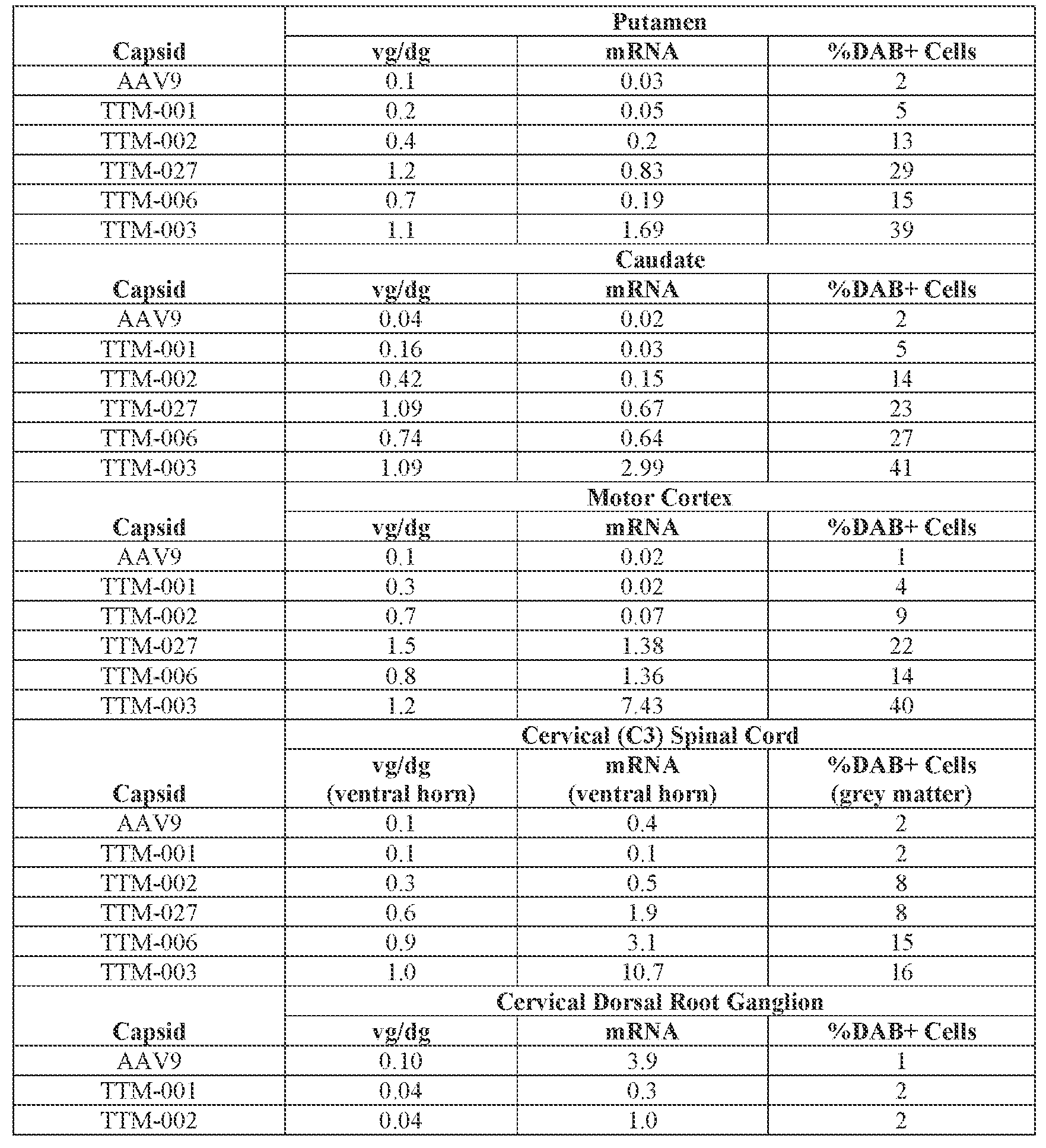
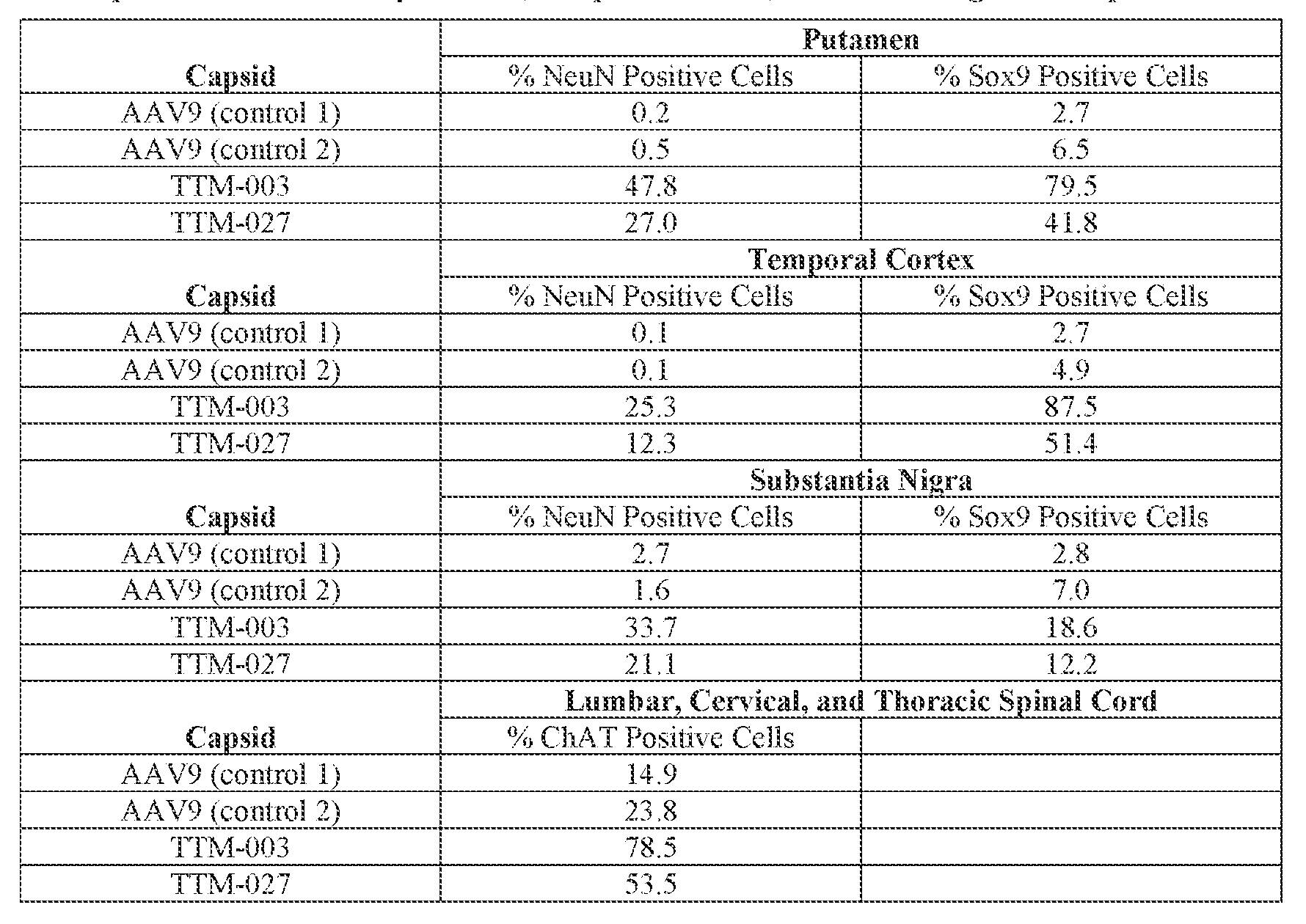
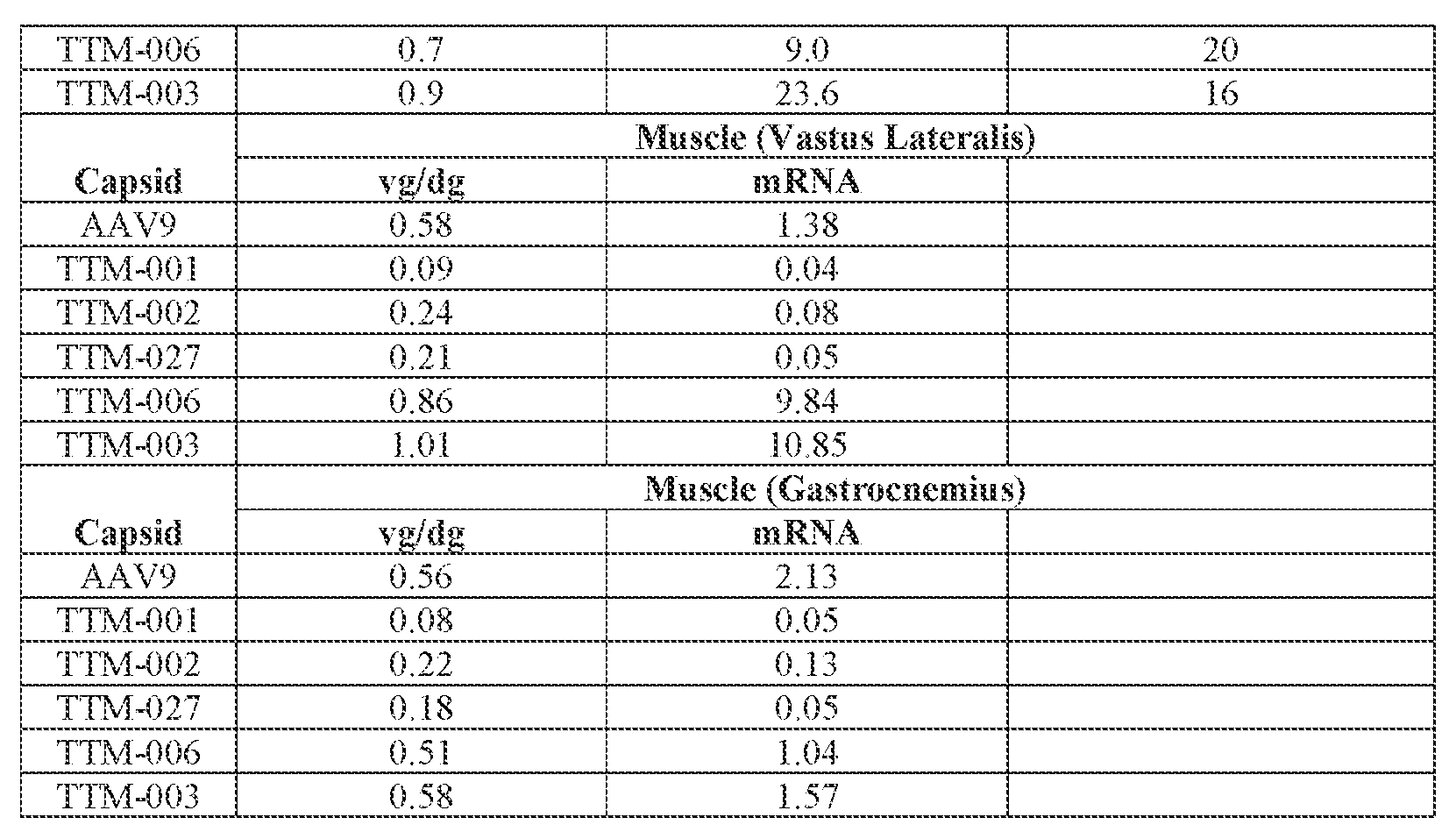
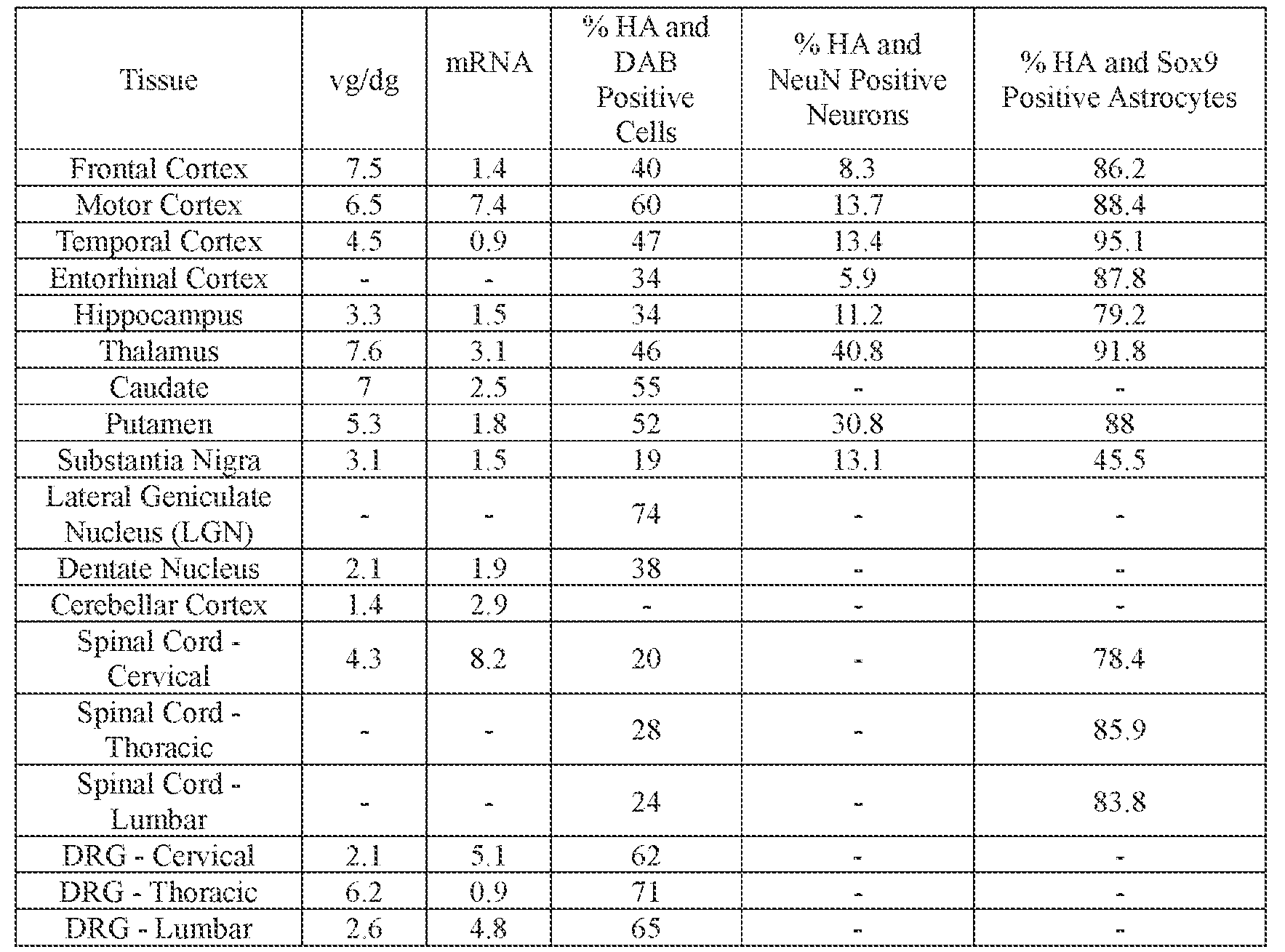
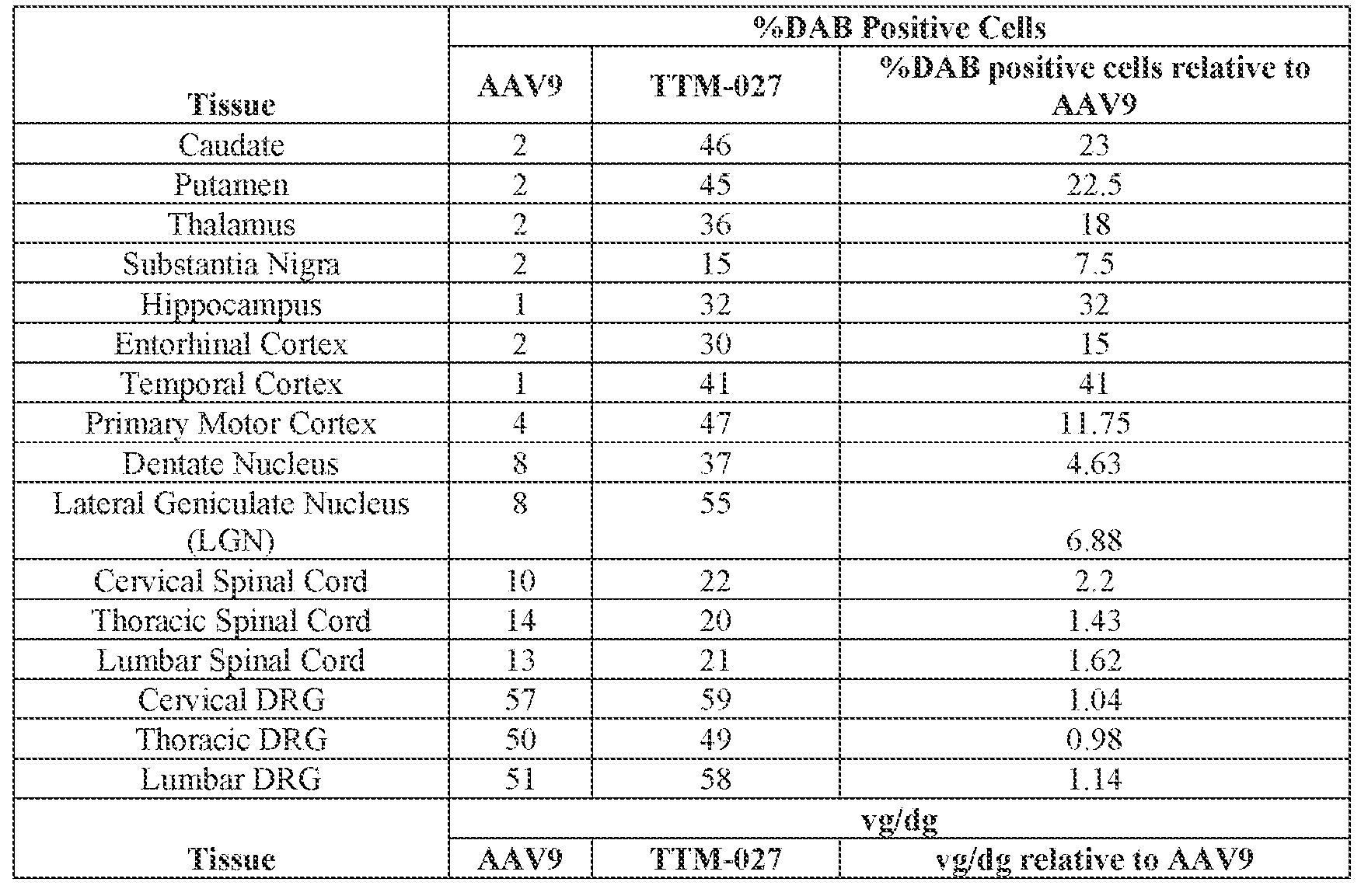
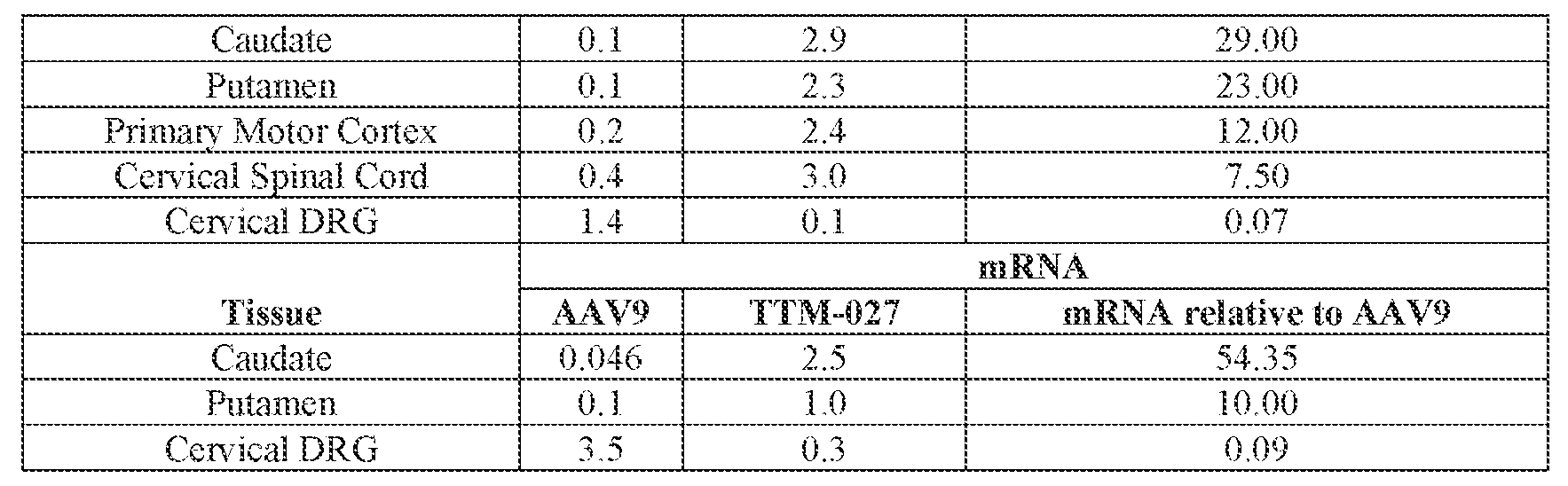
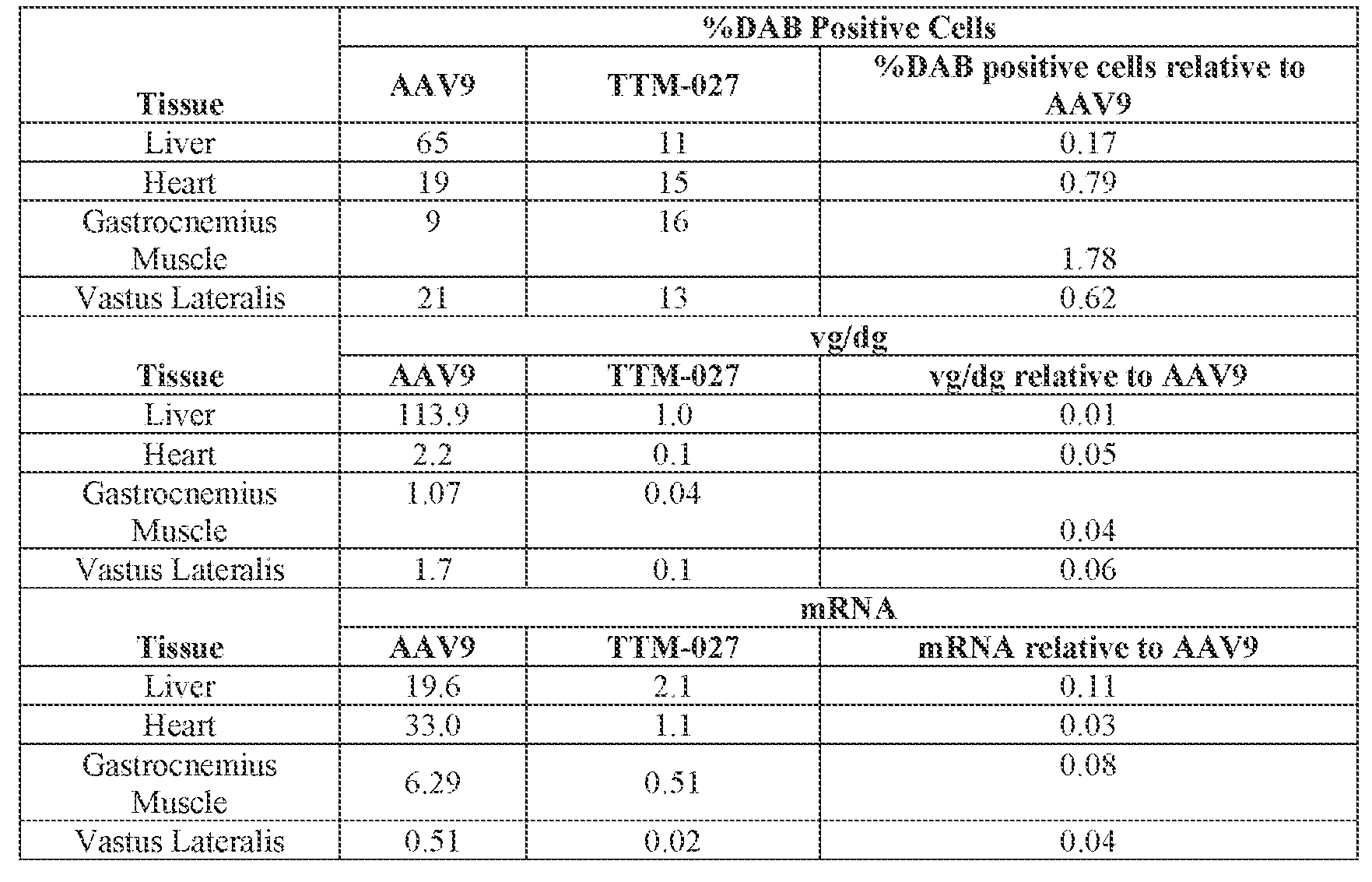
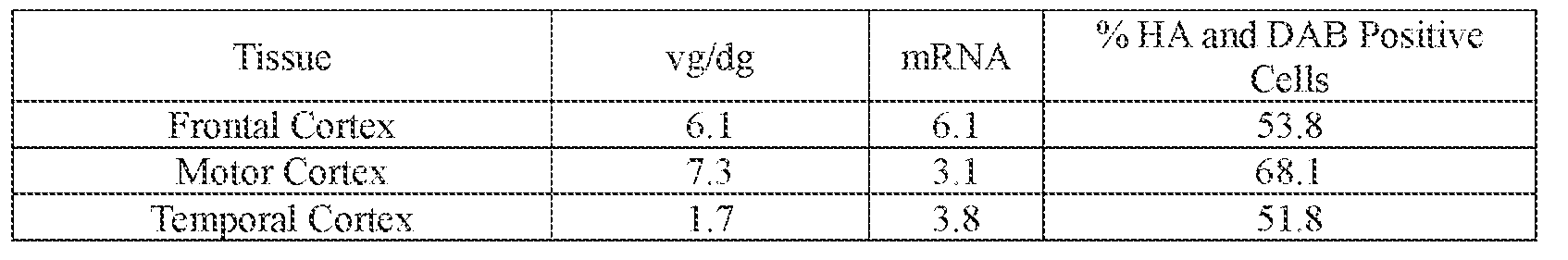
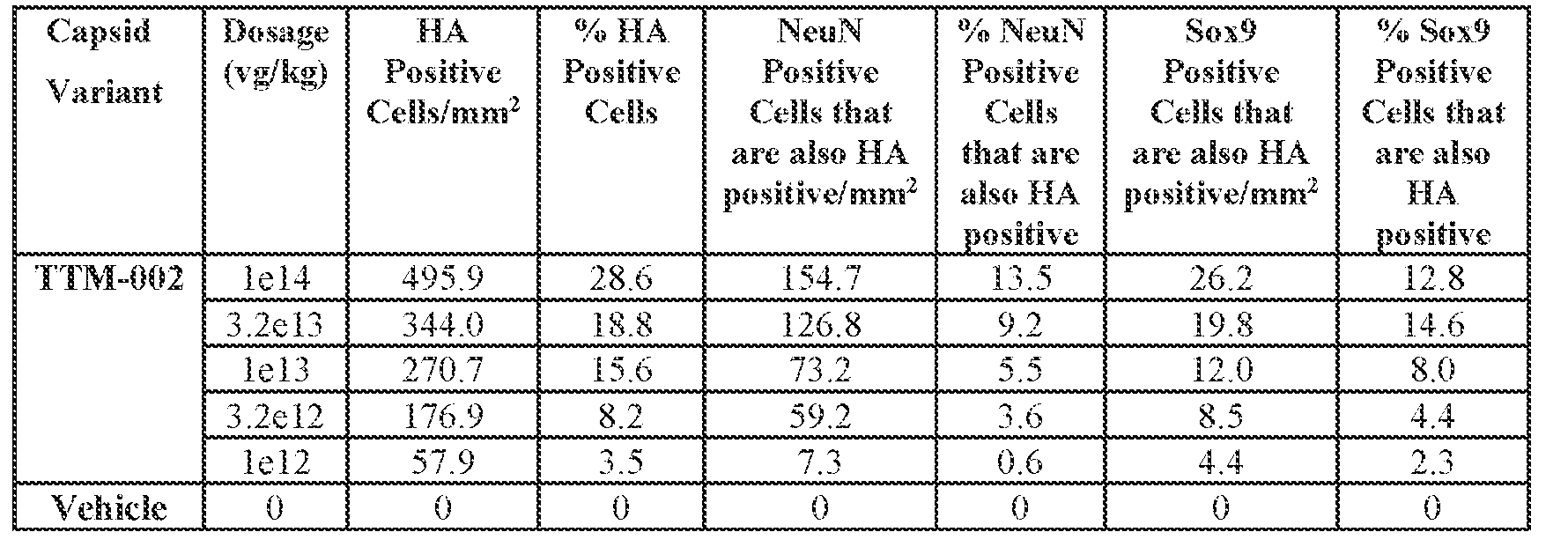
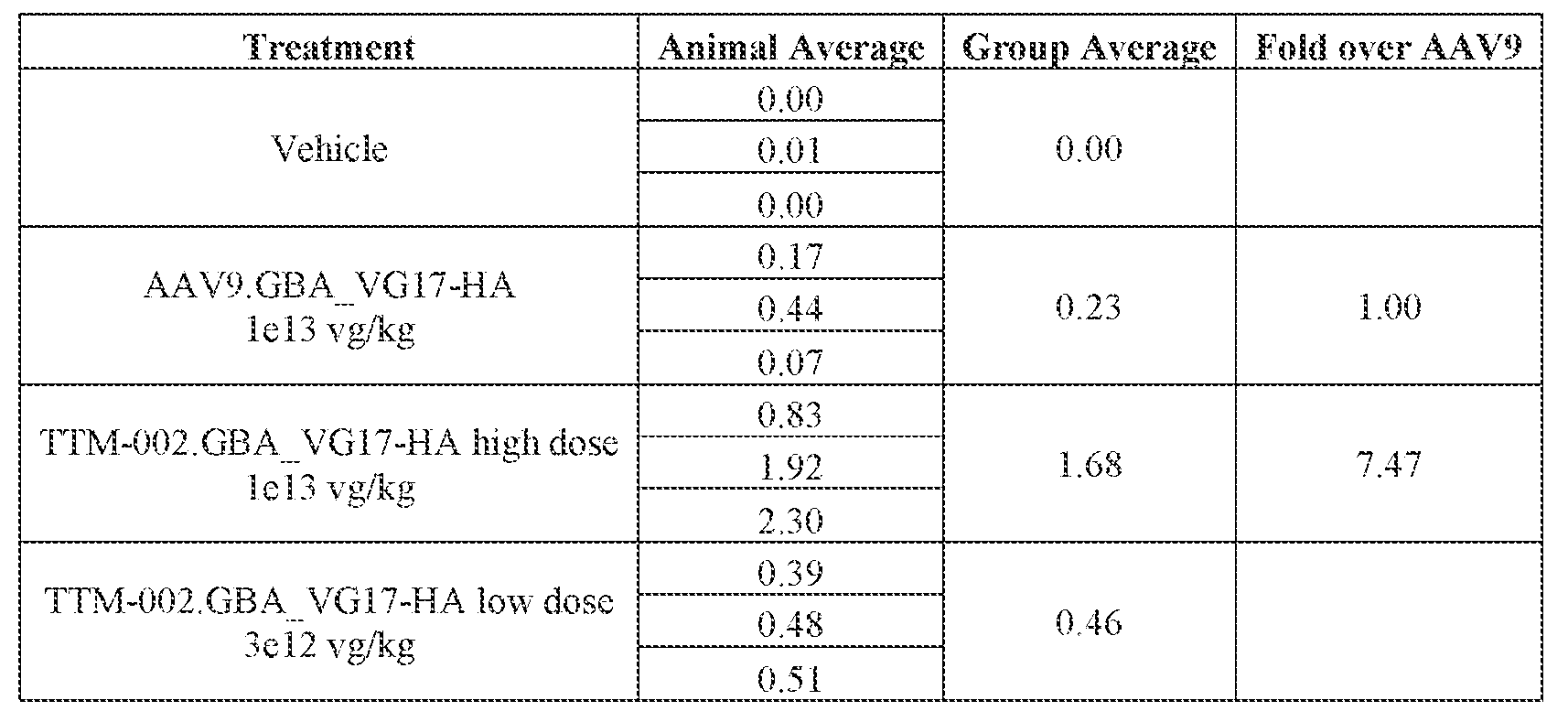
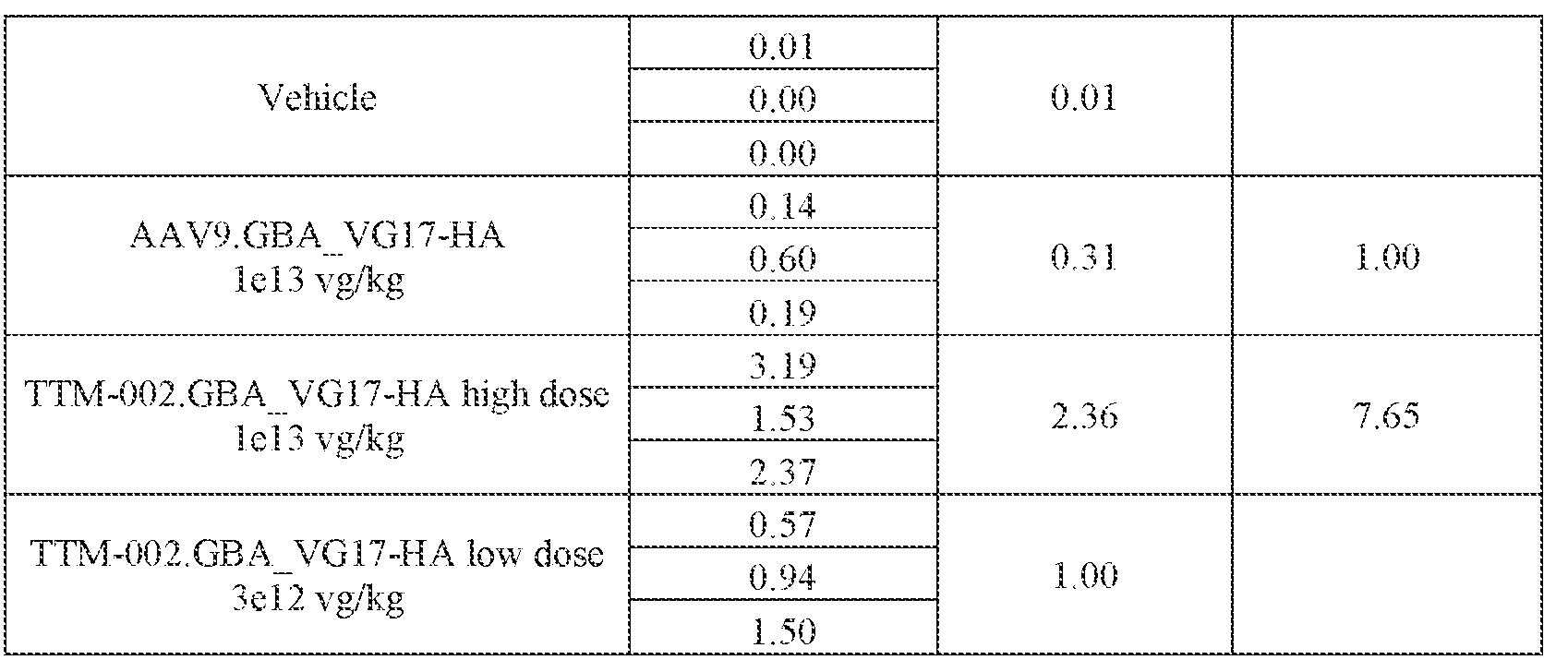
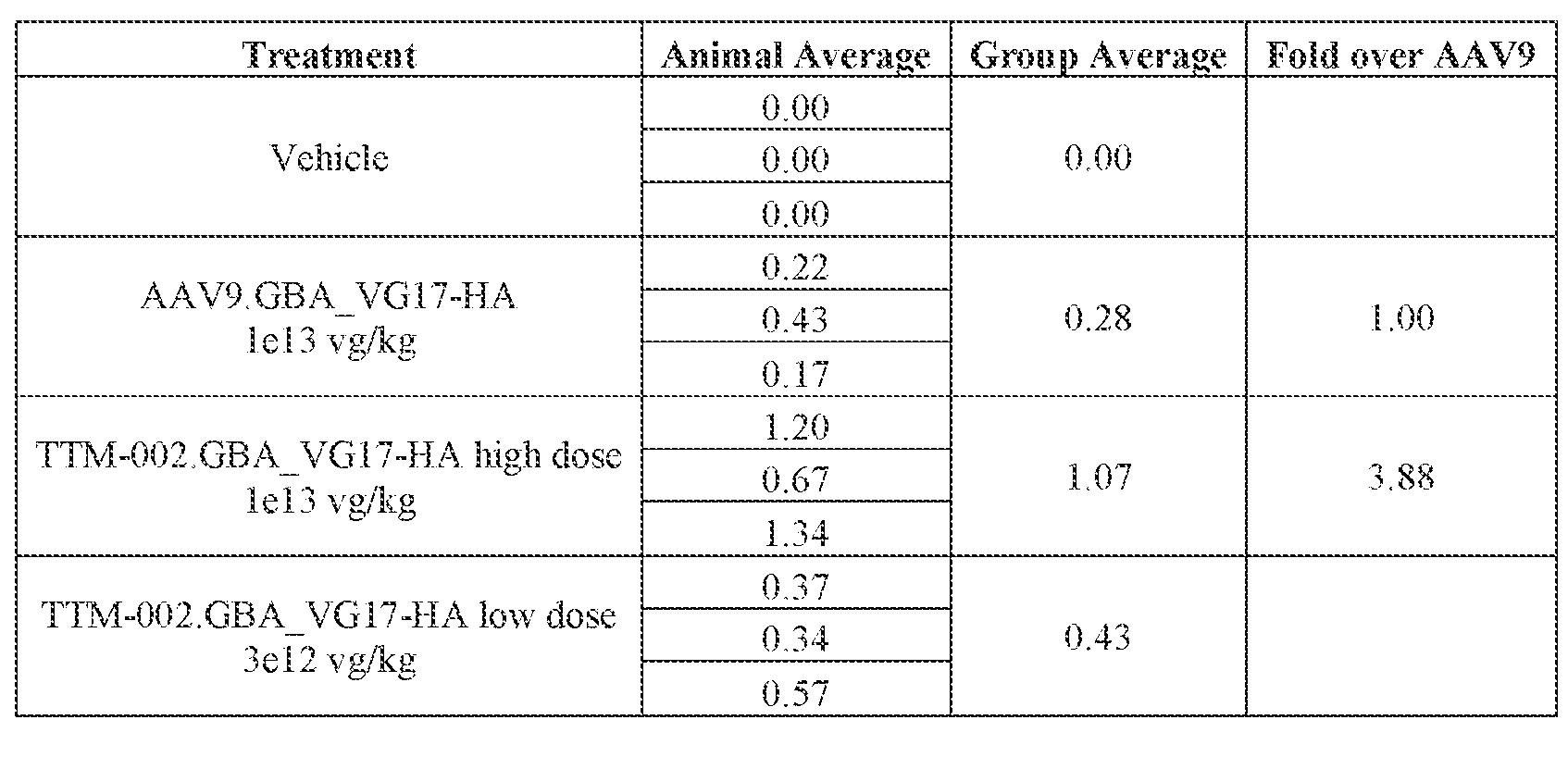
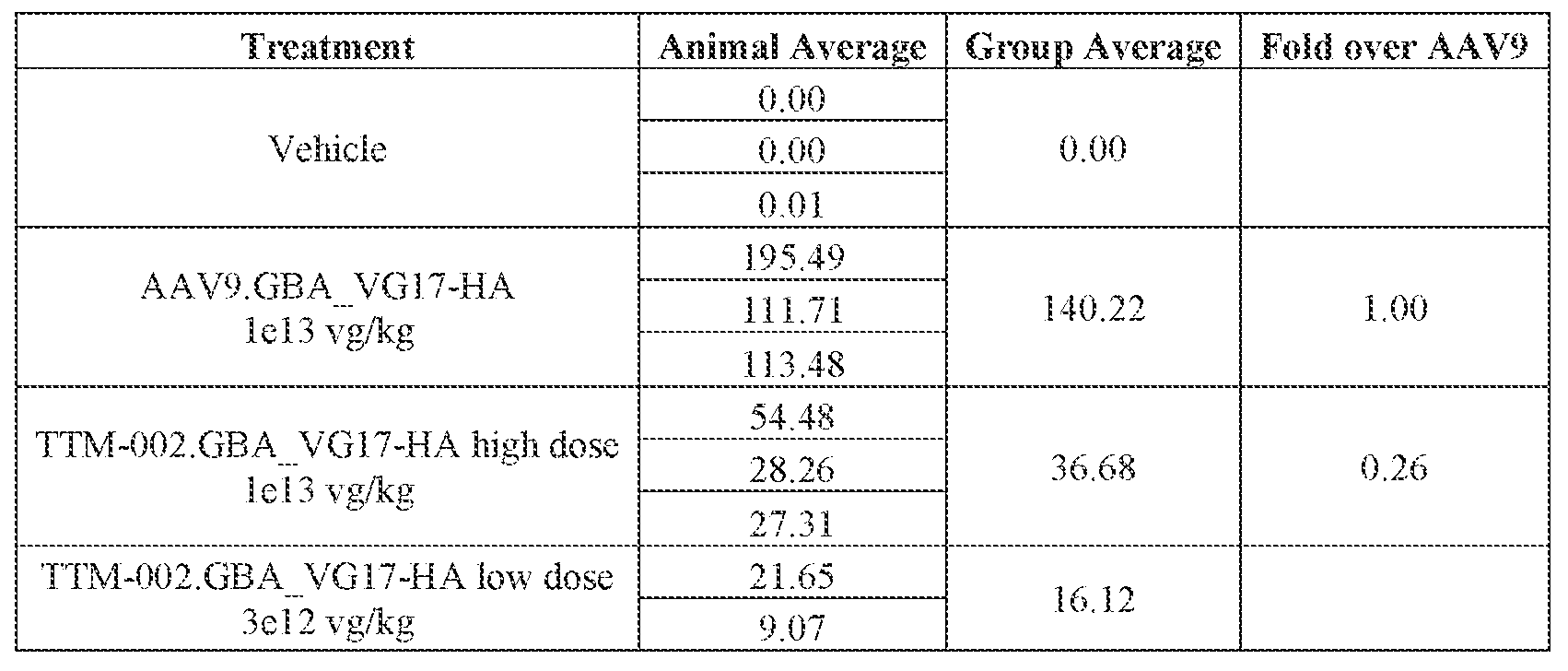
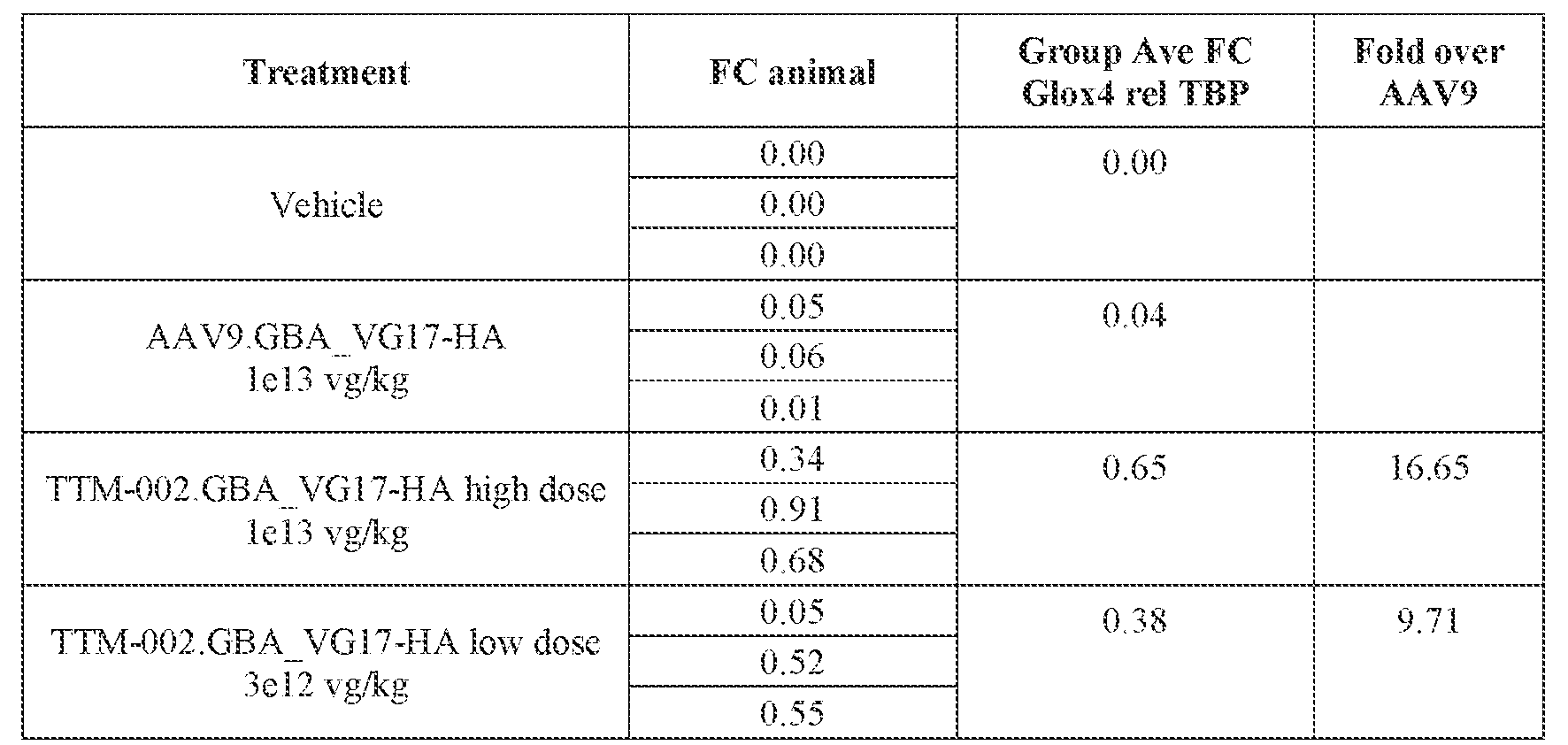
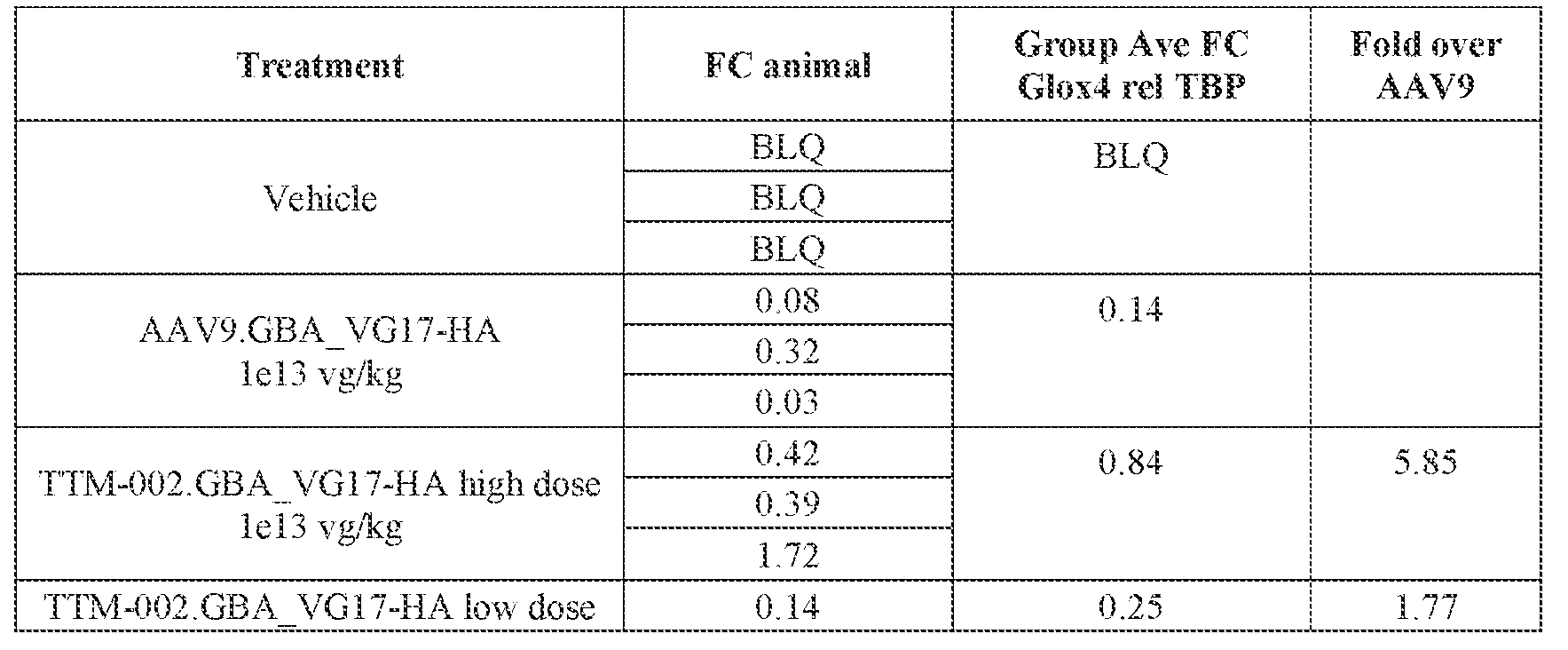
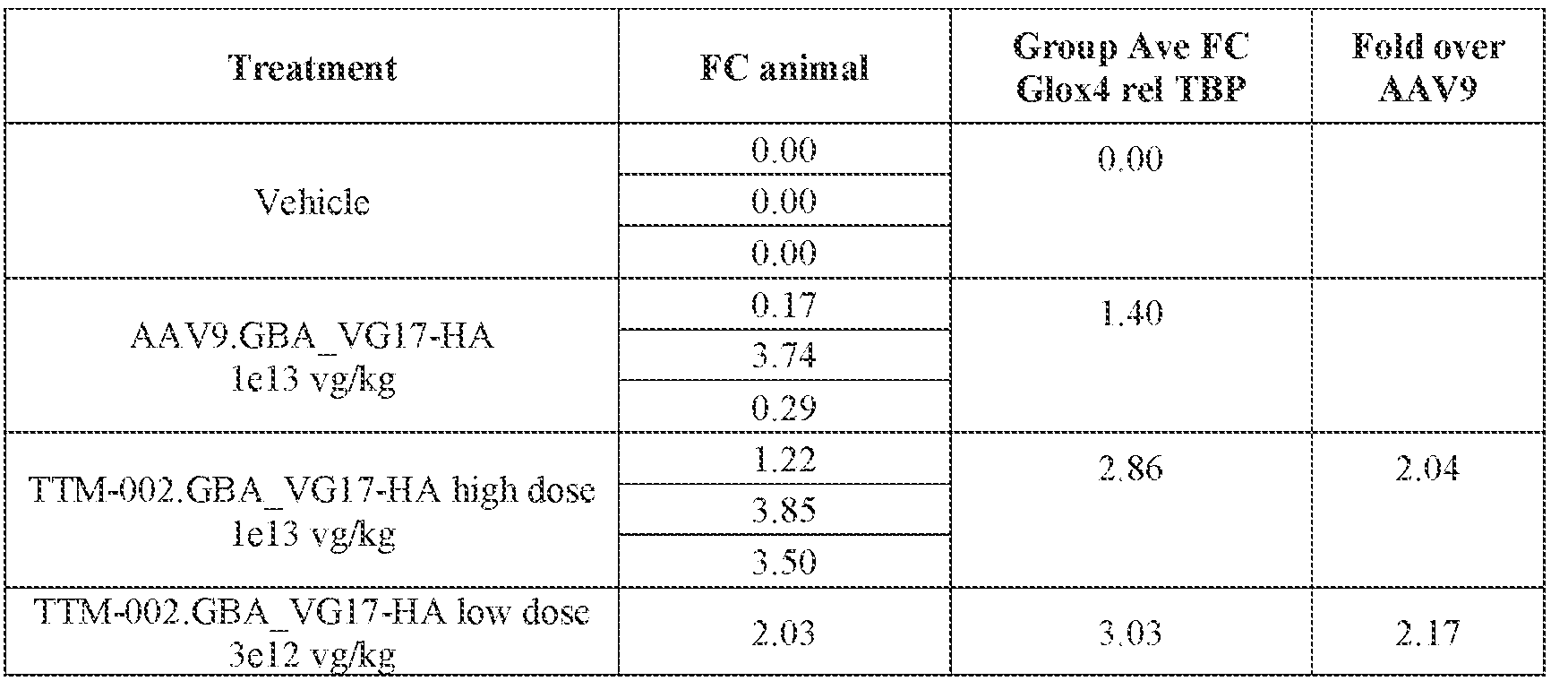
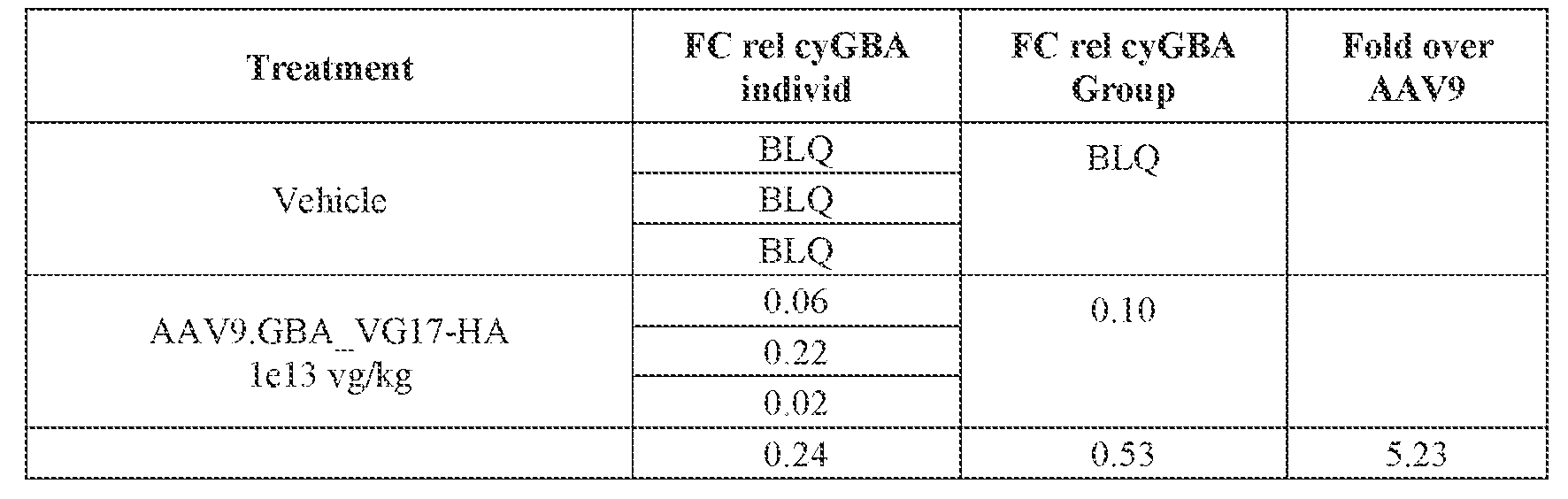
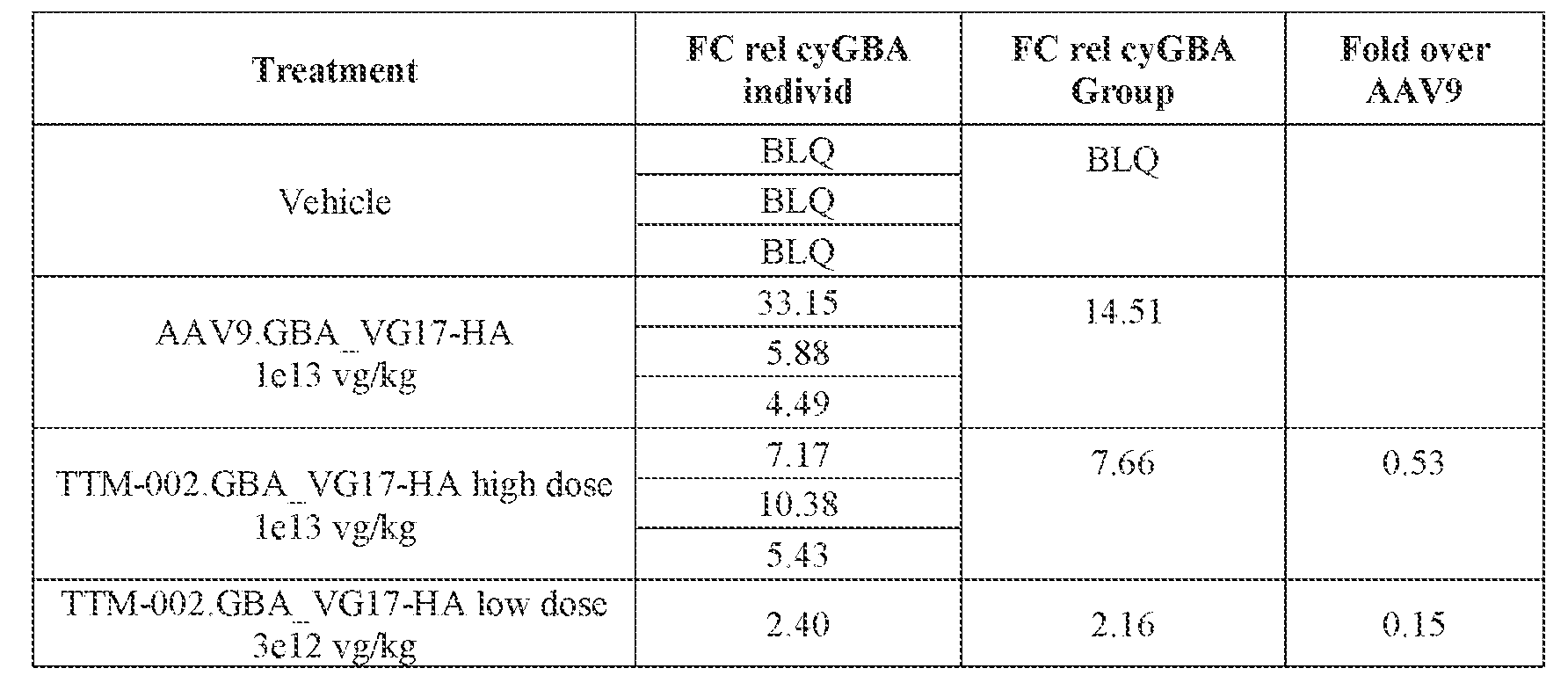
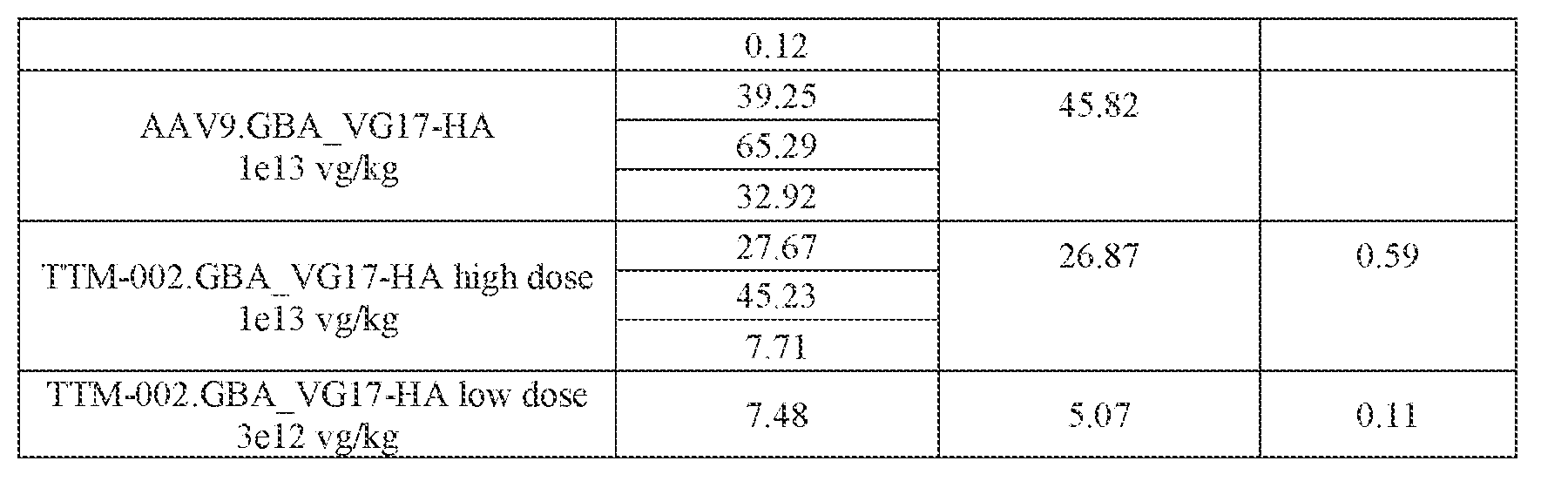
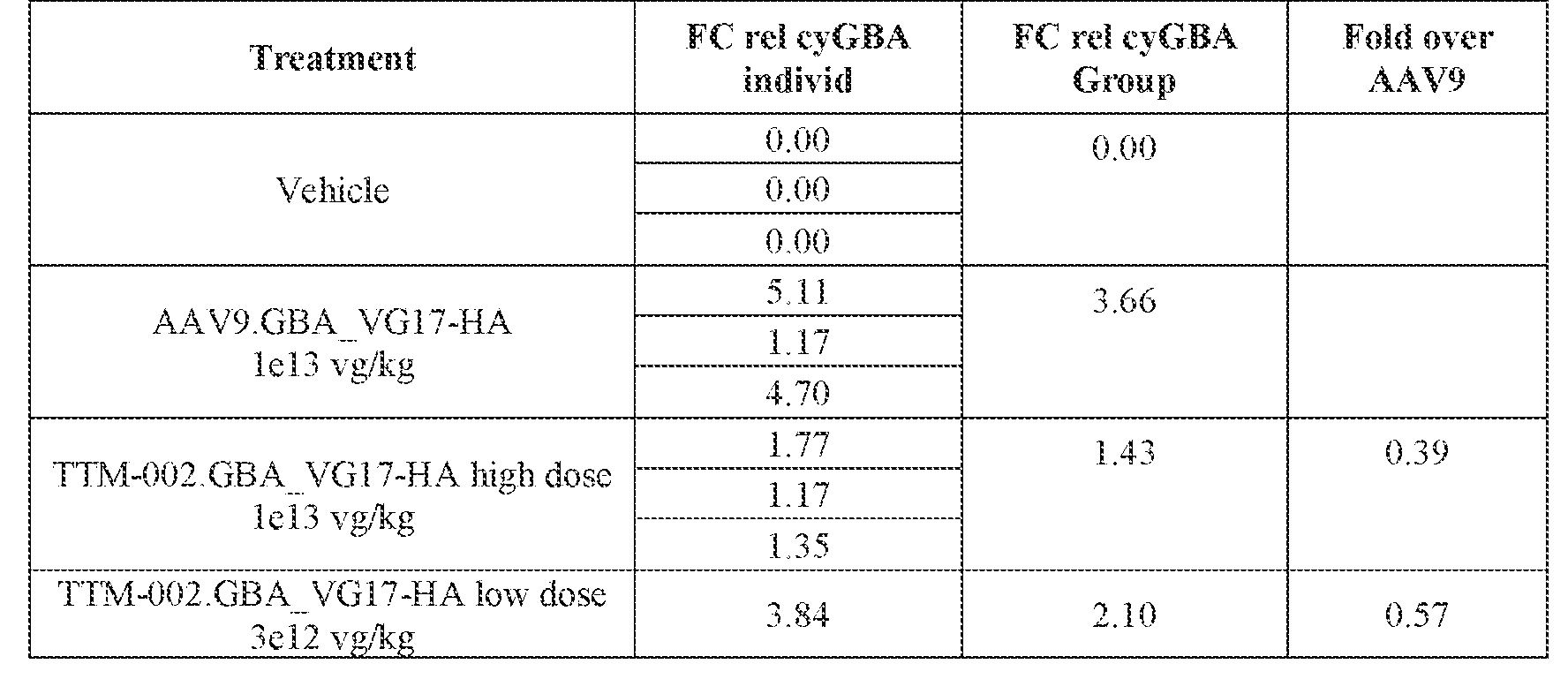
- the AAV particles described herein surprisingly provide high expression of the modulatory polynucleotide in the brain (e.g., the cortex, striatum, and brainstem), high modulatory polynucleotide activity in the brain, reduced immunogenicity, and/or reduced toxicity to other tissues of the body.
- the AAV particles described herein surprisingly provide reduced modulatory polynucleotide expression in the liver while retaining high modulatory polynucleotide activity in other areas of the brain (e.g., the brain stem), e.g., relative to AAV9.
- the AAV particles described herein can be administered to a. subject having a ATXN2-related disorder such as SCA2, ALS, PD, or FTLD.
- administration of an A AV particle to a. subject results m greater reduction in the expression and/or activity of ATXN2 in CNS cells or tissues of the subject as compared to administration of an AAV9 particle comprising a wildtype AAV9 capsid and the modulatory polynucleotide sequence.
- the present disclosure provides an adeno-associated virus (AAV) particle comprising: a) an AAV capsid variant comprising an amino acid sequence having the following formula [N1 ]-[N2]-[N3], wherein (i) optionally [N1] comprises X1, X2, and X3, wherein at least one of XL X2, or X3 is G; (ii) [N2] comprises the amino acid sequence of SPH; and (iii) [N3] comprises X4, X5, and X6, wherein at least one of X4, X5, or X6 is a basic amino acid: and b) a viral genome comprising a nucleic acid sequence encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2).
- AAV adeno-associated virus
- the modulatory polynucleotide comprises an RNAi agent targeting ATXN2 mRNA.
- the amino acid sequence [N1]-[N2]-[N3] is in hypervariable loop IV of the AAV capsid variant.
- the .AAV capsid variant is an AAV9 capsid variant.
- [N1] comprises XL X2, and X3, wherein at least one of X1, X2 or X3 is G.
- [N1]-[N2]-[N3] is presen t immediately subsequent to a position corresponding to the amino acid position 452 of SEQ ID MO: 982 and the AAV capsid variant comprises an amino acid sequence at least 90% identical, e.g., at least 91%, at least 92%, at least 93%, at least 94%), at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of positions 203-742 of SEQ ID NO: 982.
- [N1]-[N2]-[N3] is present immediately subsequent to a position corresponding to the amino acid position 452 of SEQ ID NO: 982 and the AAV capsid variant comprises an amino acid sequence at least 90% identical, e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 982.
- [N1] comprises GHD. In some embodiments, [N1] comprises the amino acid G at a position corresponding to position 453, the amino acid H at position 454, and the amino acid D at position 455 of SEQ ID NO: 138 or SEQ ID NO: 982. In some embodiments [N3] comprises KSG.
- the AAV capsid variant comprises: (i) a VP 1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to SEQ ID NO; 982; (ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to positions 138-742 SEQ ID NO: 982; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 982.
- the AAV capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to SEQ ID NO; 982; (ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 982; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 982.
- the AAV capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to SEQ ID NO; 982; (ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to positions 138-742 SEQ ID NO: 982; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 982.
- the AAV capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982; or (iii) a. VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO; 982.
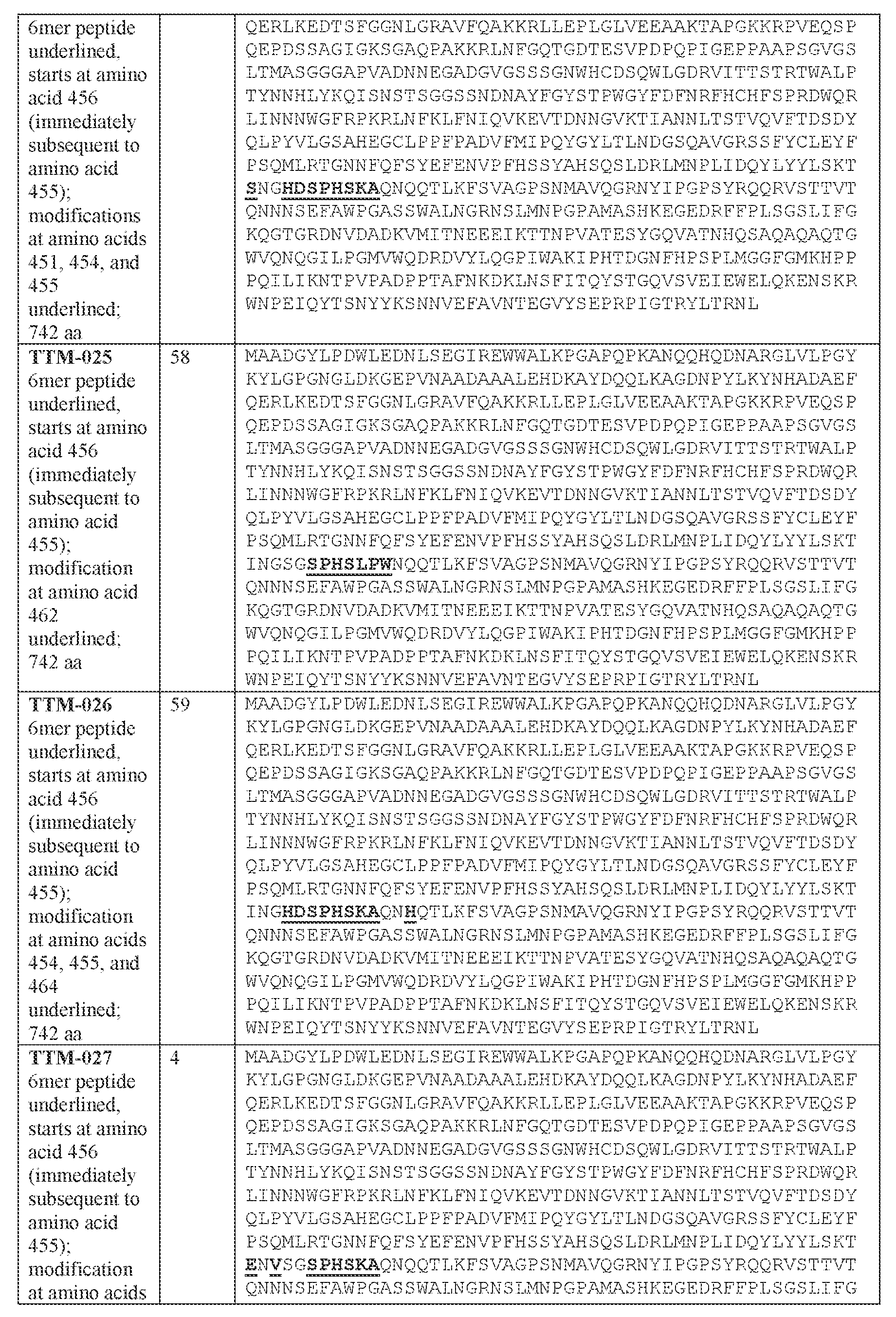
- [N2]-[N3] comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941).
- the present disclosure provides an adeno-associated virus (AAV) particle comprising (a) a viral genome comprising a nucleic acid sequence encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2), and (b) an AAV9 capsid variant comprising the amino acid sequence of SPHSKA (SEQ ID NO: 941).
- AAV9 capsid variant comprising the amino acid sequence of SPHSKA (SEQ ID NO: 941).
- the amino acid sequence of SPHSKA (SEQ ID NO: 941) is in hypervariable loop IV of the AAV capsid variant.
- the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO: 36 or SEQ ID NO: 4.
- the AAV 9 capsid variant comprises one, two, or all of: an E at an amino acid position corresponding to position 451, an R at an amino acid position corresponding to position 452, and/or a V at an amino acid position corresponding to position 453 of SEQ ID NO: 36.
- the AAV9 capsid variant comprises the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589).
- the AAV9 capsid variant comprises: (i) a VP1 protein composing an amino acid sequence having at least 90% identity to SEQ ID NO: 36; (ii) a VP2 protein comprising an amino acid sequence having at least 90% identity to positions 138-742 SEQ ID NO: 36; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 36.
- the AA.V9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 95% identity to SEQ ID NO: 36; (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 36; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 36.
- the AAV9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 36; (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to positions 138-742 of SEQ ID NO: 36; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 36.
- the AAV9 capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 36; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 36; and/or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 36.
- the AAV9 capsid variant comprises: (i) the amino acid sequence SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO 36; (ii) an E at an amino acid position corresponding to position 451 , an R at an amino acid position corresponding to position 452, and a. V at an amino acid position corresponding to position 453 of SEQ ID NO: 36; and (iii) no other modifications relative to wild type AAV 9.
- SPHSKA SEQ ID NO: 941
- the AAV9 capsid variant comprises one, two, or all of: an N at an amino acid position corresponding to position 452, an E at an amino acid position corresponding to position 451, and/or a V at an amino acid position corresponding to position 453 of SEQ ID NO: 4.
- the AAV9 capsid variant comprises the amino acid sequence of KTENVSGSPHSKAQNQQT (SEQ ID NO: 3272).
- the AA.V9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 90% identity to SEQ ID NO: 4; (ii) a VP2 protein comprising an amino acid sequence having at least 90% identity to positions 138-742 of SEQ ID NO: 4; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 4.
- the AAV9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 95% identity to SEQ ID NO: 4; (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 4; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 4.
- the AAV9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 4; (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to positions 138-742 of SEQ ID NO: 4; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 4.
- the AAV9 capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 4; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 4; and/or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 4,
- the AAV9 capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO 4; (ii) an E at an amino acid position corresponding to position 451 and a. V at an amino acid position corresponding to position 453 of SEQ ID NO: 4; and (iii) no other modifications relative to wild type AAV9.
- the modulatory polynucleotide comprises: (a) a 5’ flanking region comprising any one of SEQ ID NOs: 6413-6416 or a nucleotide sequence at least 95% identical thereto; (b) a loop region comprising any one of SEQ ID NOs: 6417-6421 or a nucleotide sequence at least 95% identical thereto; and (c) a 3’ flanking region comprising any one of SEQ ID NOs: 6422.-6427 or a nucleotide sequence at least 95% identical thereto.
- the 5' flanking region comprises SEQ ID NO: 6414 or SEQ ID NO: 6415, or a nucleotide sequence at least 95% identical thereto;
- the loop region comprises SEQ ID NO: 6417, SEQ ID NO: 6418, or SEQ ID NO: 6421 , or a nucleotide sequence at least 95% identical thereto;
- the 3' flanking region comprises SEQ ID NO: 6423, SEQ ID NO: 6424, or SEQ ID NO: 6425, or a nucleotide sequence at least 95% identical thereto.
- the 5’ flanking region comprises SEQ ID NO; 6414 or a nucleotide sequence at least 95% identical thereto
- the loop region comprises SEQ ID NO: 6417 or a nucleotide sequence at least 95% identical thereto
- the 3’ flanking region comprises SEQ ID NO; 6423 or a. nucleotide sequence at least 95% identical thereto.
- the 5’ flanking region comprises SEQ ID NO: 6415 or a nucleotide sequence at least 95% identical thereto
- the loop region comprises SEQ ID NO: 6421 or a nucleotide sequence at least 95% identical thereto
- the 3’ flanking region comprises SEQ ID NO: 6425 or a nucleotide sequence at least 95% identical thereto.
- the 5’ flanlang region comprises SEQ ID NO: 6414 or a nucleotide sequence at least 95% identical thereto
- the loop region comprises SEQ ID NO: 6417 or a nucleotide sequence at least 95% identical thereto
- the 3" flanking region comprises SEQ ID NO: 6424 or a nucleotide sequence at least 95% identical thereto.
- the 5’ flanking region comprises SEQ ID NO: 6414 or a nucleotide sequence at least 95% identical thereto
- the loop region comprises SEQ ID NO: 6418 or a nucleotide sequence at least 95% identical thereto
- the 3’ flanking region comprises SEQ ID NO: 6423 or a nucleotide sequence at least 95% identical thereto.
- the modulatory polynucleotide comprises siRNA .
- the modulatory polynucleotide comprises shRNA.
- the modulatory polynucleotide further comprises a passenger strand and a guide strand, wherein the guide strand binds to and reduces or eliminates expression of one or more ATXN2 mRNA transcripts.
- the modulatory polynucleotide comprises from 5 ' to 3‘: the 5' flanking region, the passenger strand, the loop region, tire guide strand, and the 3' flanking region.
- the modulatory polynucleotide comprises from 5' to 3': the 5' flanking region, the guide strand, the loop region, the passenger strand; and the 3' flanking region.
- the passenger strand is 15-30 nucleotides in length. In some embodiments, the guide strand is 15-30 nucleotides in length. In some embodiments, the guide strand is 21-25 nucleotides in length. In some embodiments, the passenger strand is 21- 25 nucleotides in length. In some embodiments, the passenger strand is at least 70%, at least 80%, at least 90%, at least 95% or is 100% complementary to the guide strand.
- the one or more ATXN2 mRNA transcripts comprises SEQ ID NO: 6428, SEQ ID NO: 6429, SEQ ID NO: 6430, and/or SEQ ID NO: 6431, or a trinucleotide repeat expansion thereof.
- the viral genome comprises a promoter operably linked to the nucleic acid sequence encoding the modulatory polynucleotide.
- the viral genome further comprises an inverted terminal repeat (ITR) sequence.
- ITR inverted terminal repeat
- the viral genome comprises an ITR sequence positioned 5’ relative to the nucleic acid sequence encoding the modulatory polynucleotide.
- the viral genome comprises an ITR sequence positioned 3' relative to the nucleic acid sequence encoding the modulatory polynucleotide .
- viral genome comprises an ITR sequence positioned 5’ relative to the nucleic acid sequence encoding the modulatory polynucleotide, and an ITR sequence positioned 3’ relative to the nucleic, acid sequence encoding the modulatory polynucleotide.
- the present disclosure provides a cell comprising an AAV particle described herein.
- the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial ceil.
- the present disclosure provides a method of making an AAV particle described herein, the method comprising (i) providing a cell comprising the viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle.
- the AAV capsid variant comprises (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least. 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 982; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least.
- a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least. 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 982; (ii) a VP
- the AAV capsid variant comprises (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 36; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 138-742 SEQ ID NO: 36; and/or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO:
- the AAV capsid variant comprises (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%), at least 95%, at least 96%, at least 97%>, at least 98%, or at least 99% identity) to SEQ ID NO: 4; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 4 or an amino acid, sequence having at. least 90% identity (e.g., at least. 90%, at.
- a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%>, at least 97%, at least 98%, or at least 99% identity) to positions 203-742 of SEQ ID NO: 4.
- the method further comprises, prior to step (i), introducing a first nucleic acid molecule comprising the viral genome into the cell.
- the cell comprises a second nucleic acid molecule encoding the AAV capsid variant.
- the method further comprises, prior to step (i), introducing the second nucleic acid molecule into the cell.
- the cell comprises a mammalian cell, e.g., an HEK293 cell, an insect cell, e.g., an Sf9 cell, or a bacterial cell.
- the present disclosure provides a pharmaceutical composition comprising an AAV particle described herein, and a pharmaceutically acceptable excipient.
- the present disclosure provides a method of delivering a modulatory polynucleotide for reducing or eliminating expression of ATXN2 to a subject, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein, thereby delivering the modulatory polynucleotide.
- the method reduces or eliminates ATXN2 protein in the subject.
- the subject has, has been diagnosed with having, or is at risk of having an ATXN2-related disorder.
- the ATXN2-related disorder is spinocerebellar ataxia type 2 (SCA2).
- the present disclosure provides a method of treating an ATXN2-related disorder in a subject, comprising administering to tire subject an effective amount of a pharmaceutical composition or AAV particle described herein, thereby treating the ATXN2-related disorder.
- the subject has, has been diagnosed, or is at risk of having the ATXN2 -related disorder.
- the subject has one or more mutations in the ATXN2 gene.
- the one or more mutations in the ATXN2 gene comprises a trinucleotide repeat expansion.
- the trinucleotide repeat expansion in the ATXX2 gene comprises 32 or more CAG repeats.
- treating results in prevention of progression of the ATXN2- related disorder in the subject.
- the treating results in amelioration of at least one symptom of the ATXN 2 -related disorder and/or a change in at least one biomarker of the AT.XN2 -related disorder in the subject.
- the at least one symptom comprises progressive cerebellar ataxia, nystagmus, slow saccadic eye movements, ophthahnoparesis, parkinsonism, or a combination thereof.
- the ATXN2-related disorder is spinocerebellar ataxia type 2 (SCA2).
- the present disclosure provides a method of treating spinocerebellar ataxia type 2 (SCA2) in a. subject, comprising administering to the subject an effective amount of the pharmaceutical composition described herein or the AAV particle described herein, thereby treating SCA2.
- the subject has, has been diagnosed with having, or is at risk of having SCA2.
- the subject is a human.
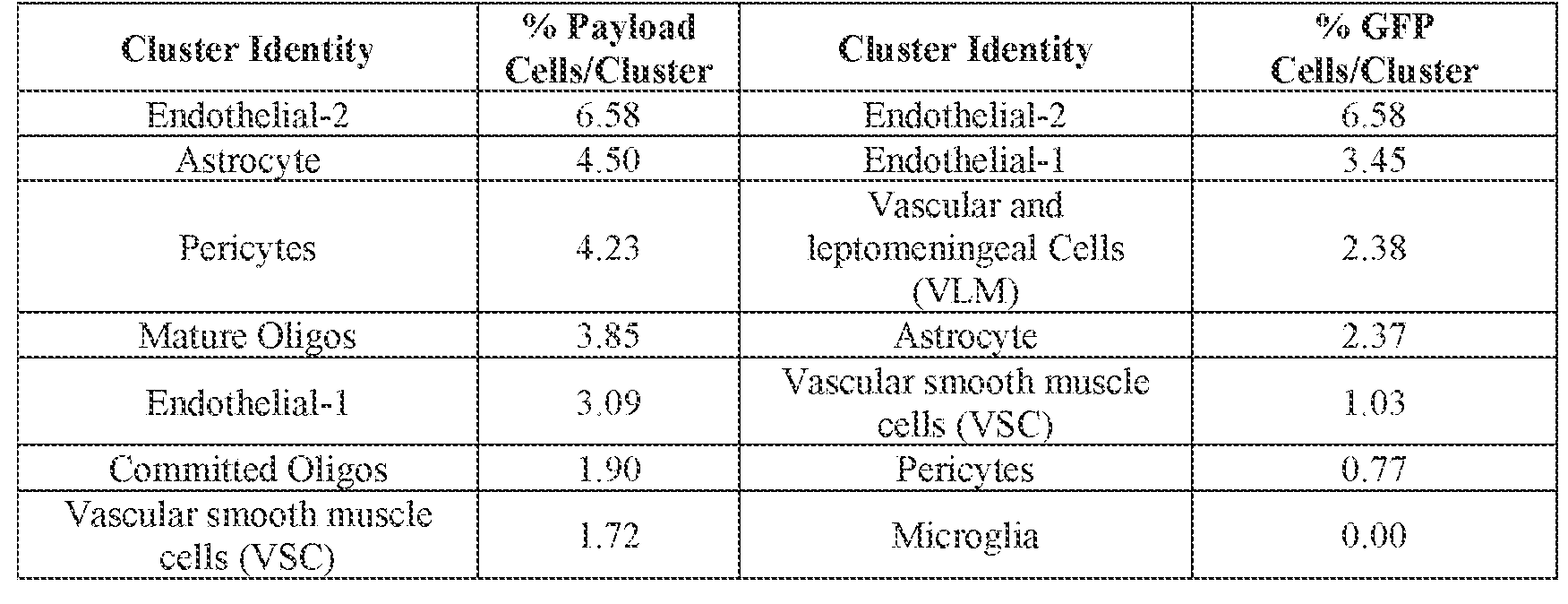
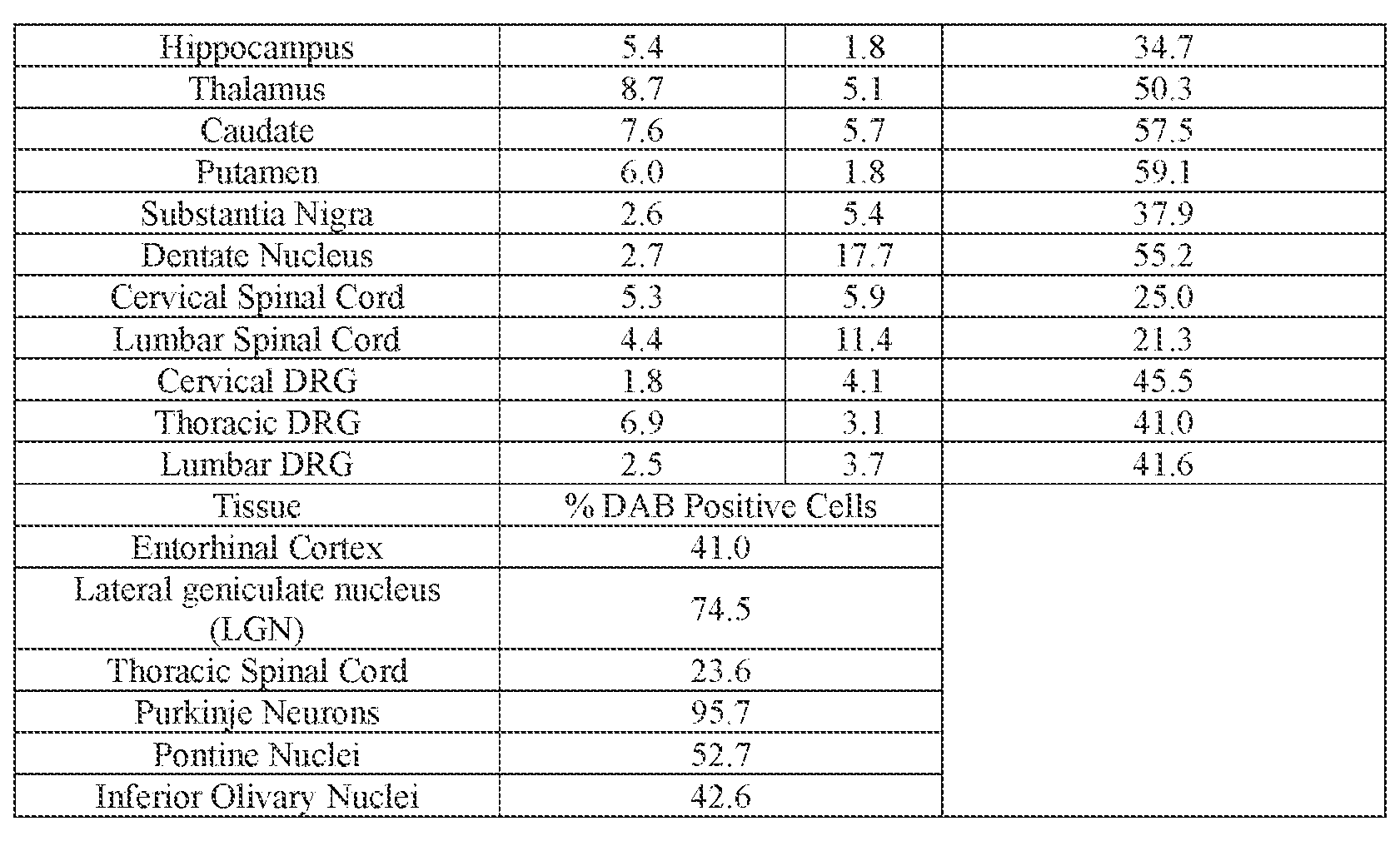
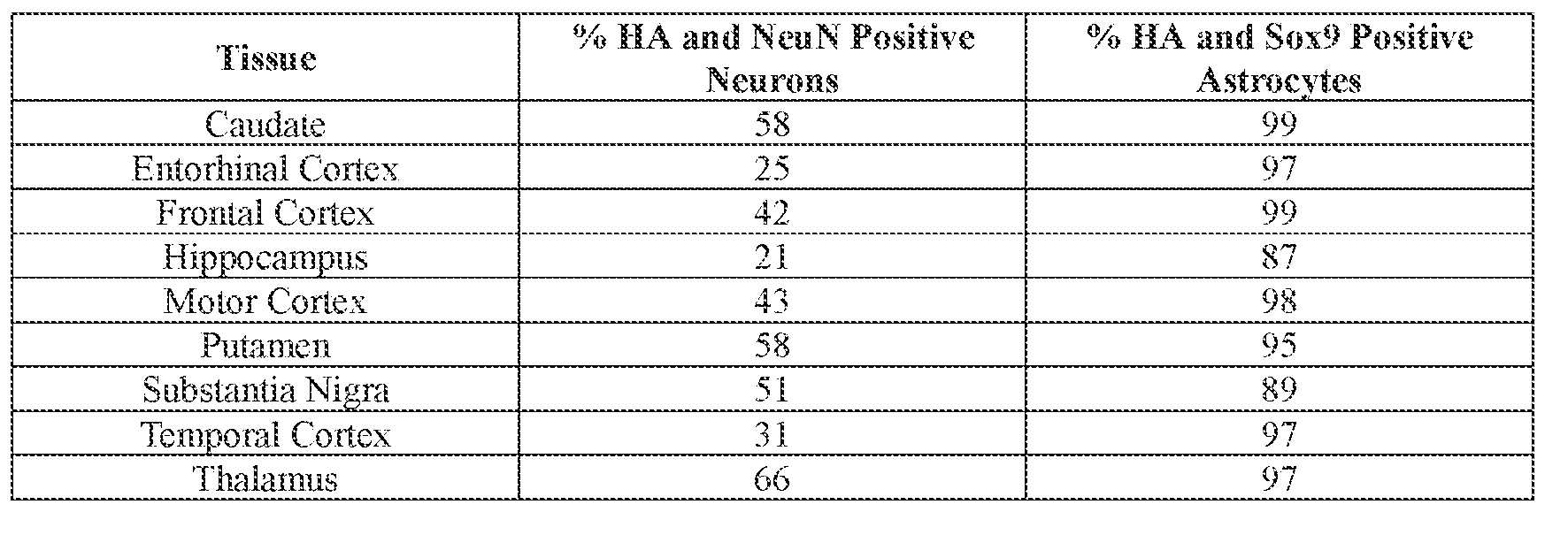
- the AAV particle or the pharmaceutical composition is delivered to a cell or tissue of the CNS.
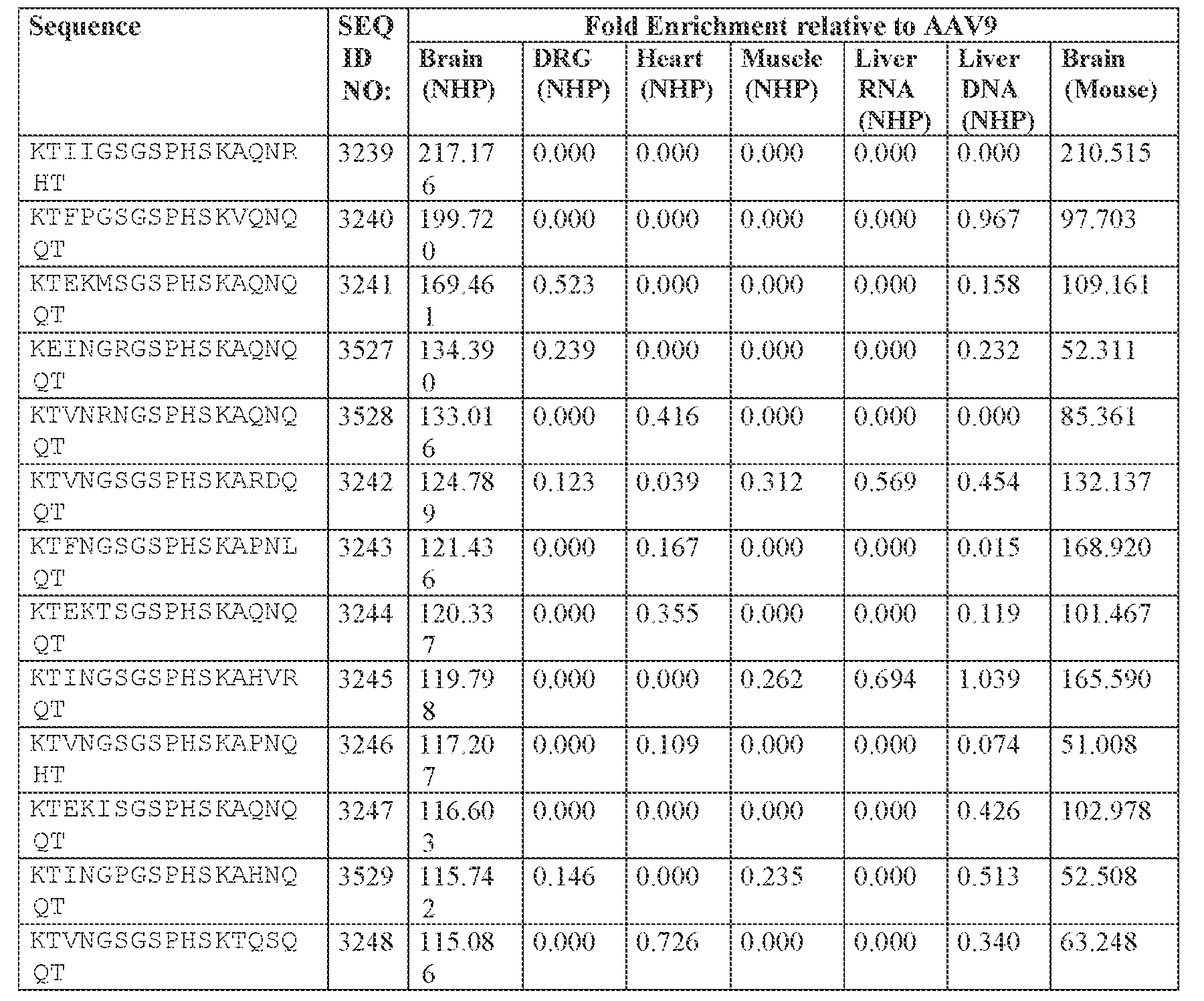
- the cell or tissue of the CNS is a cell or tissue of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region).
- the method of delivering or treating further comprises evaluating, e.g., measuring, the level of modulatory polynucleotide expression, and/or the level of ATXN2 expression, e.g., ATXN2 gene, ATXN2 mRNA, and/or ATXN2 protein expression, in the subject, e.g., in a cell, tissue, or fluid, of the subject.
- the level of ATXN2 protein is measured by an ELISA, a Western blot, or an immunohistochemistry assay.
- the evaluating of the subject’s level of modulatory polynucleotide expression and/or level of ATXN2 expression is performed prior to and/or subsequent to administration of the pharmaceutical composition or AAV particle, optionally wherein the subject’s level of modulatory polynucleotide expression and/or the subject’s level of ATXN2 expression prior to administration is compared to the subject’s level of modulatory polynucleotide expression and/or the subject’s level of ATXN2 expression subsequent to administration.
- the subject’s level of ATXN2 protein expression subsequent to administration is decreased relative to the subject’s level of ATXN2 protein expression prior to administration.
- the method of delivering or treating further comprises evaluating, e.g., measuring, the level of modulatory polynucleotide activity and/or ATXN2 activity in the subject, e.g., in a cell or tissue of the subject.
- the administration results in: (i) a decrease in ATXN2 protein expression in a cell, tissue (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region)), and/or fluid (e.g., CSF and/or serum) of the subject relative to baseline and/or
- tissue
- VG viral genomes
- a CNS tissue e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei)
- cortex e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex
- external cuneate nucleus geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord
- the method of delivering or treating further comprises administering to the subject at least one additional therapeutic agent and/or therapy.
- the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the ATXN2-related disorder.
- the at least one additional therapeutic agent and/or therapy comprises one or more of: growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2-related disorders.
- the ATXN2-related disorder is SCA2, amyotrophic lateral sclerosis (ALS), Parkinson’s disease (PD), or frontotemporal lobar degeneration (FTLD). In some embodiments, the ATXN2-related disorder is SCA2.
- the method of delivering or treating further comprises administering an immunosuppressant to the subject.
- the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
- the present, disclosure provides a method of treating a subject having or diagnosed with having ALS, PD, or FTLD, comprising administering to the subject an effective amount of a pharmaceutical composition or A AV particle described herein.
- the method further comprises administering to the subject at least one additional therapeutic agent and/or therapy.
- the at least one additional therapeutic agent and/or therapy comprises one or more of: growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2-related disorders.
- the method further comprises administering an immunosuppressant to the subject.
- the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, rnycophenolatemofetil, tacrolimus, rituximab, and/or ecuiizumab hydroxychloroquine.
- a corticosteroid e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone
- rapamycin e.g., rapamycin, rnycophenolatemofetil, tacrolimus, rituximab, and/or ecuiizumab hydroxychloroquine.
- the present, disclosure provides a pharmaceutical composition or AAV particle described herein for use in a method of treating a disorder described herein.
- the ATXN2-related disorder is SCA2.
- the subject has, has been diagnosed 'with having, or is at risk of having the ATXN2-relat.ed disorder, optionally wherein the ATXN2-relat.ed disorder is SCA2.
- the present disclosure provides a pharmaceutical composition or AAV particle described herein for use in treating an ATXN2 -related disorder in a subject.
- the ATXN2-related disorder is SCA2.
- the subject has, has been diagnosed with having, or is at risk of having the ATXN2-related disorder, optionally wherein the ATXN2 -related disorder is SCA2.
- the present disclosure pro vides a use of a pharmaceutical composition or A AV particle described herein in the manufacture of a medicament for the treatment of an ATXN2-related disorder in a subject.
- the ATXN2- related disorder is SCA2.
- the subject has, has been diagnosed with having, or is at risk of having the ATXN2-related disorder, optionally wherein the ATXN2- related disorder is SCA2.
- An adeno-associated virus (AAV) particle comprising an AAV capsid, variant (e.g., an AAV9 capsid variant) and a viral genome comprising a. nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein (e.g., human ATXN2 protein), wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]- [N3], wherein:
- [N2] comprises the amino acid sequence of SPH
- (iii) [N3] comprises X4, X5, and X6, wherein at least one of X4, X5, or X6 is a basic amino acid, e.g., a K or R.
- X 4 of [N3] is: K, S, A, V, T, G, F, W, V, N, or R;
- X5 of [N3] is: S, K, T, F, I, L, Y, H, M, or R; and/or
- X6 of [N3] is: G, A, R, M, I, N, T, Y, D, P, V, L, E, W, N, Q, K, or S; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c).
- AAV particle of any one of embodiments 1-4, wherein [N3] comprises SK, KA, KS, AR, RM, VK, AS, SR, VK, KR, KK, KN, VR, RS, RK, KT, TS, KF, FG, KI, IG, KL, LG, TT, TY, KY, YG, KD), KP, TR, RG, VR, GA, SL, SS, FL,, WK, SA, RA, LR, KW, RR, GK, TK, NK, AK, KV, KG, KH, KM, TG, SE, SV, SW, SN, HG, SQ, LW, MG, MA, or SG.
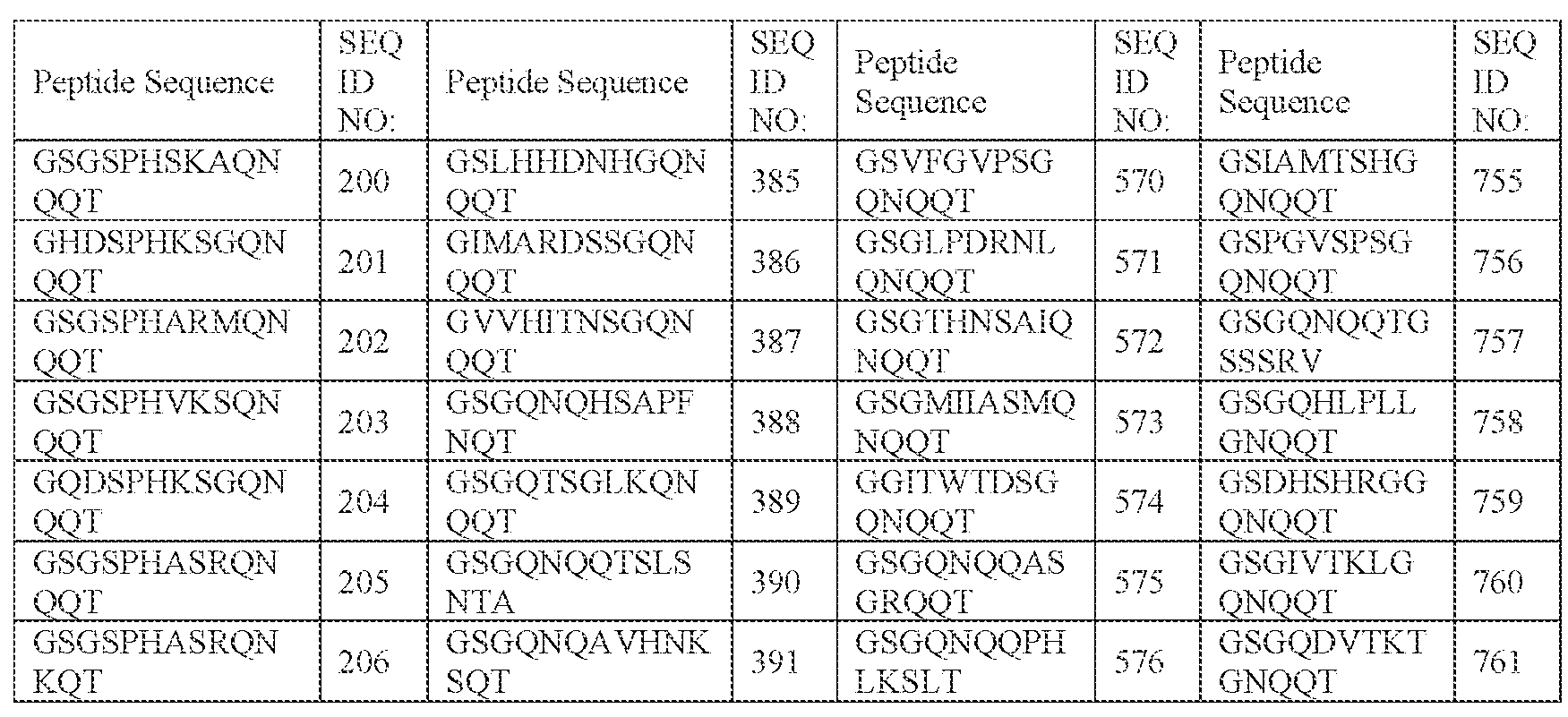
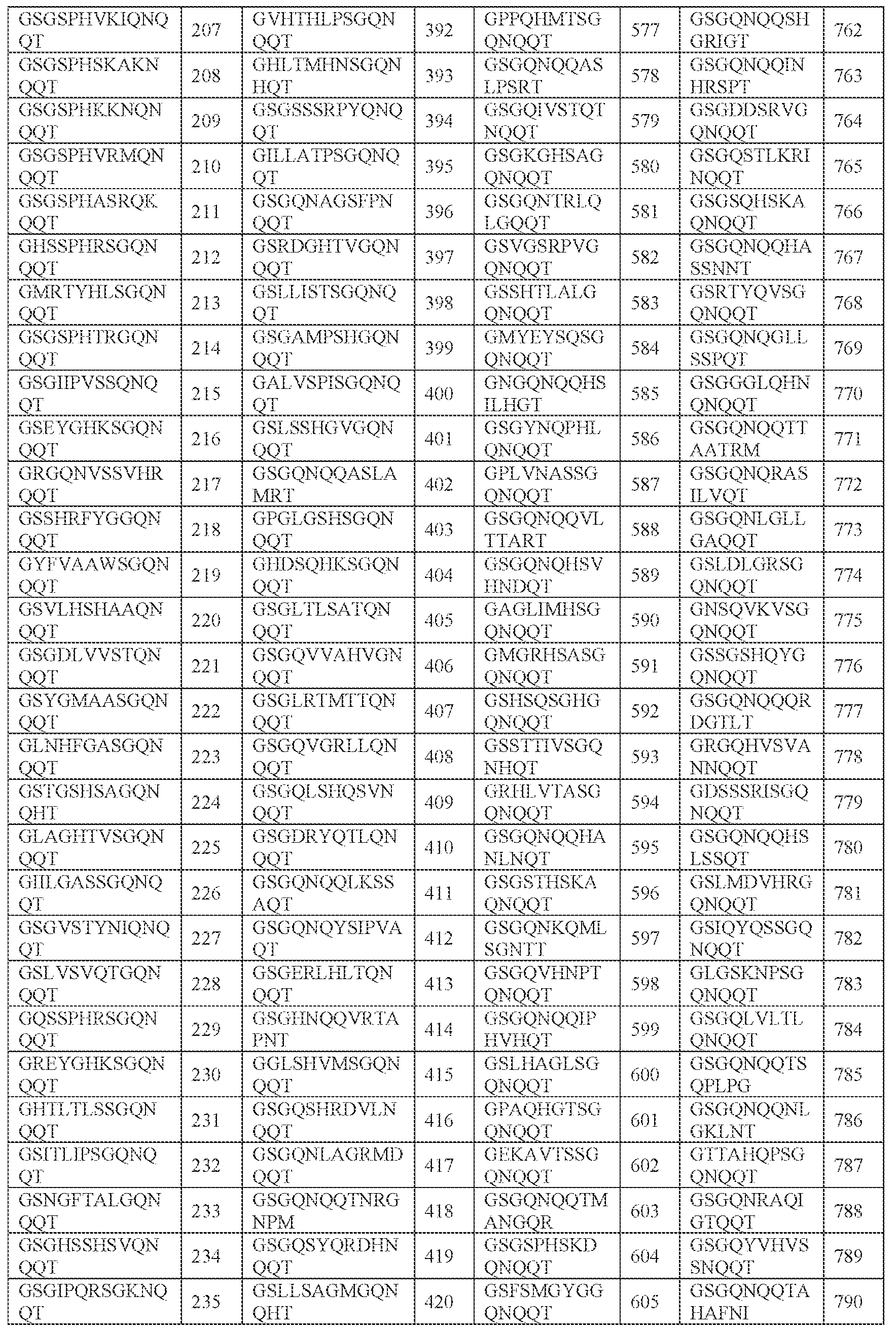
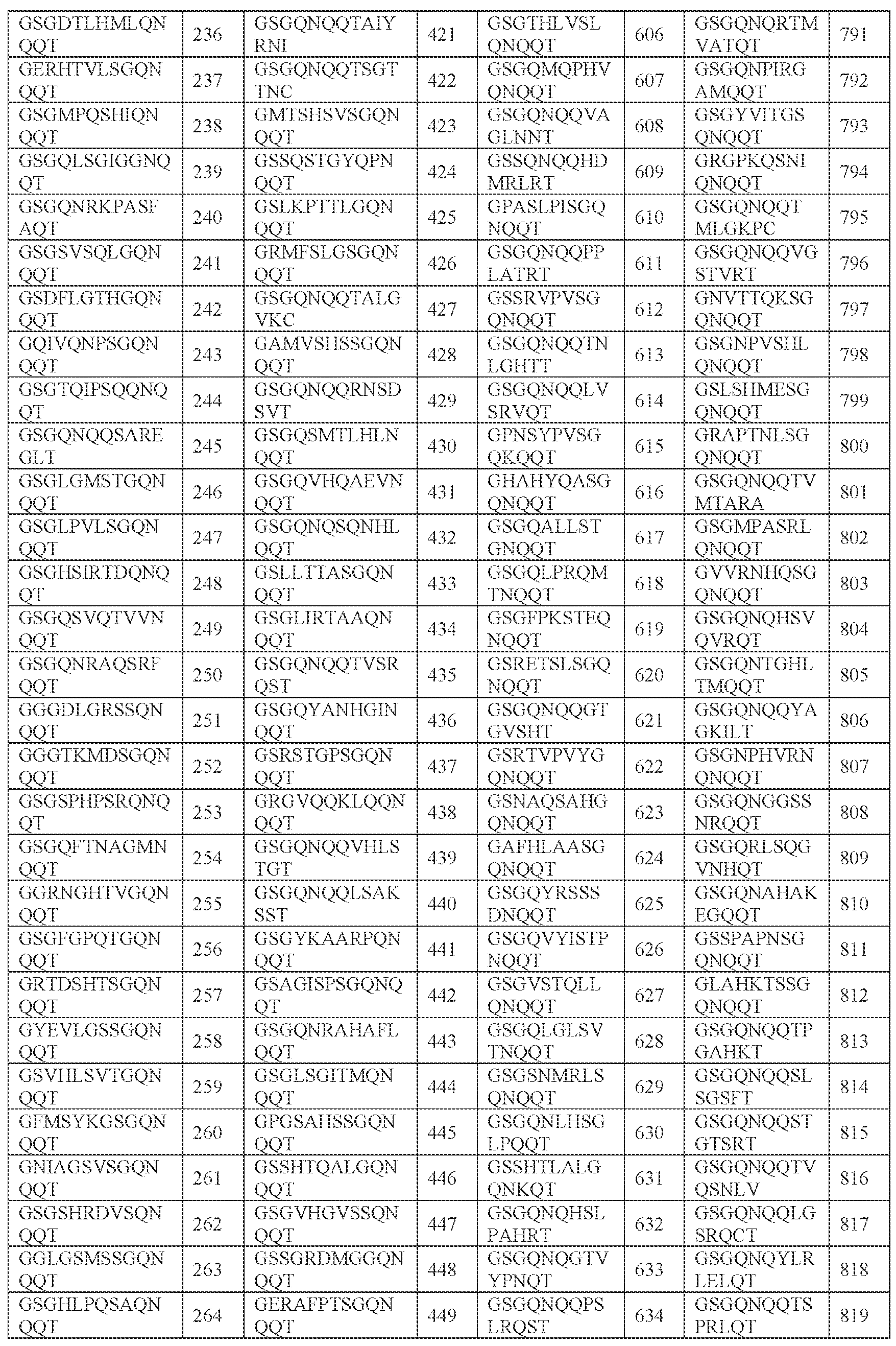
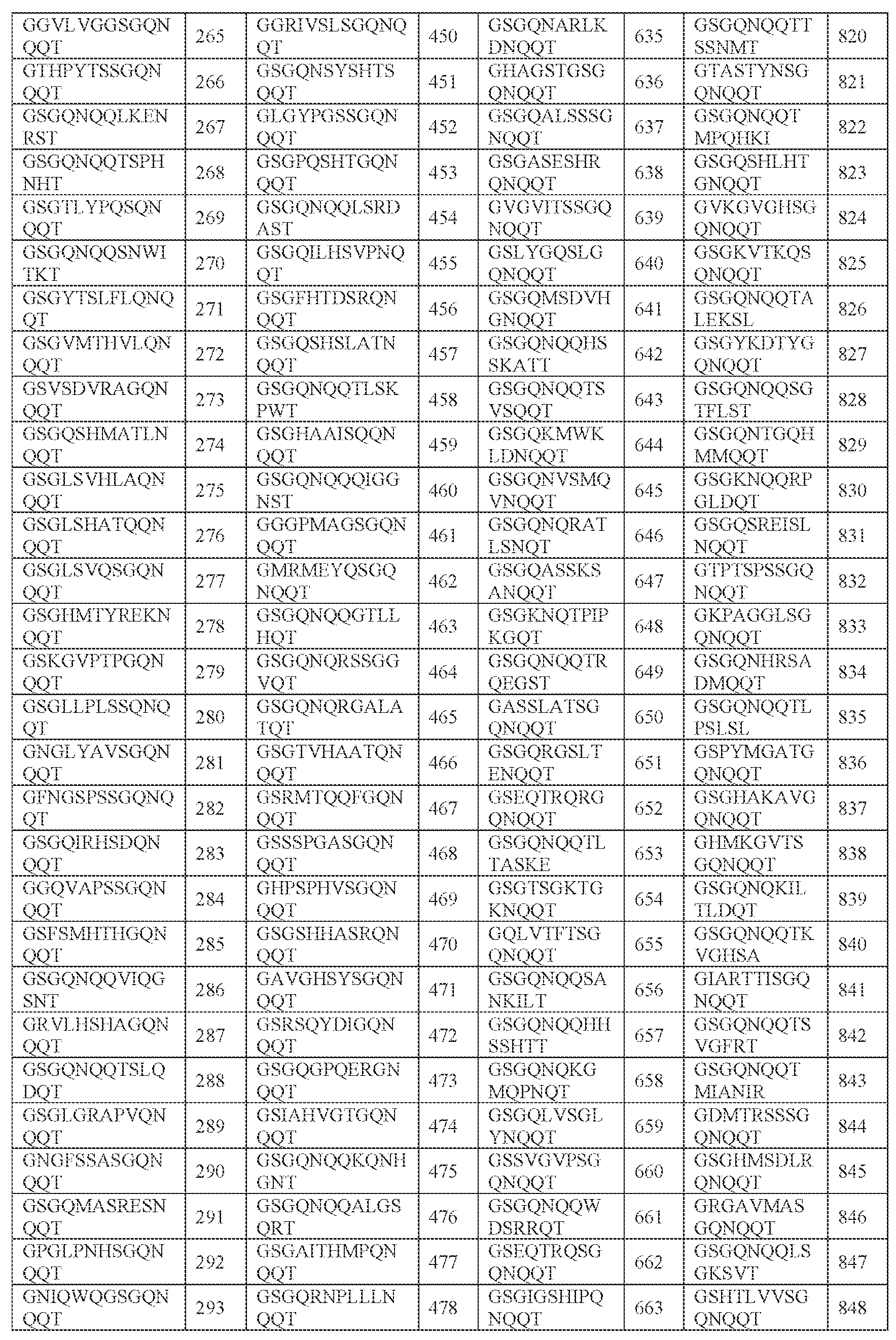
- [N2]-[N3] comprises SPHSK (SEQ ID NO: 4701), SPHKS (SEQ ID NO: 4704), SPHAR (SEQ ID NO: 4705), SPHVK (SEQ ID NO: 4706), SPHAS (SEQ ID NO: 4707), SPHKK (SEQ ID NO: 4708), SPHVR (SEQ ID NO: 4709), SPHRK (SEQ ID NO: 4710), SPHKT (SEQ ID NO: 4711), SPHKF (SEQ ID NO: 4712), SPHKI (SEQ ID NO: 4713), SPHKL (SEQ ID NO: 4714), SPHKY (SEQ ID NO: 4715), SPHTR (SEQ ID NO: 4716), SPHKR (SEQ ID NO: 4717), SPHGA (SEQ ID NO: 4718), SPHSR (SEQ ID NO: 4719), SPHSL (SEQ ID NO: 4720), SPHSS (SEQ ID NO: 4711), SPHSK (SEQ ID NO: 4701),
- SPHSKA SEQ ID NO: 941
- SPHKSG SEQ ID NO: 946
- SPHARM SEQ ID NO: 947
- SPHVKS SEQ ID NO: 948
- SPHASR SEQ ID NO: 949
- SPHVKI SEQ ID NO: 950
- SPHKKN SEQ ID NO: 954
- SPHVRM SEQ ID NO: 955)
- SPHRKA SEQ ID NO: 956
- SPHKFG SEQ ID NO: 957)
- SPHKIG SEQ ID NO: 958
- SPHKLG SEQ ID NO: 959
- SPHKTS SEQ ID NO: 963
- SPHKTT SEQ ID NO: 964
- SPHKTY SEQ ID NO: 965
- SPHKYG SEQ ID NO: 966
- SPHSKD SEQ ID NO: 967
- SPHSKP SEQ ID NO: 968
- SPHTRG SEQ ID NO: 972
- SPHVRG SEQ ID NO: 973
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- amino acid other than G at position 453 e.g., V, R, D, E, M, T, I, S, A, N, L, K, H, P, W, or C
- an amino acid other than S at position 454 e.g.
- (a) X1 of [N1] is: G, V, R, D, E, M, T, I, S, A, N, I.,, K, H, P, W, or C;
- X2 of [N1] is: S, V, L, N, D, H, R, P, G, T, I, A, E, Y, M, or Q; and/or
- X3 of [N1] is: G, C, L, D, E, Y, H, V, A, N, P, or S; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g,, a. conservative substitution, of any of the aforesaid amino acids in (a)-(c). 14.
- AAV particle of any one of embodiments 1-14, wherein [N1] is or comprises GSG, GHD, GQD, VSG, CSG, GRG, CSH, GQS, GSH, RVG, GSC, GLL, GDD, GHE, GNY, MSG, RNG, TSG, ISG, GPG, ESG, SSG, GNG, ASG, NSG, LSG, GGG, KSG, HSG, GTG, PSG, GSV, RSG, GIG, WSG, DSG, IDG, GLG, DAG, DGG, MEG, ENG, GSA, KNG, KEG, AIG, GYD, GHG, GRD, GND, GPD, GMG, GQV, GHN, GHP, or GHS.
- SGSPH SEQ ID NO: 4752
- HDSPH SEQ ID NO: 4703
- QDSPH SEQ ID NO: 4753
- RGSPH SEQ ID NO: 4754
- SHSPH SEQ ID NO: 4755
- QSSPH SEQ ID NO: 4756
- DDSPH SEQ ID NO: 4757
- HESPH SEQ ID NO: 4758
- NYSPH SEQ ID NO: 4759
- VGSPH SEQ ID NO: 4760
- SCSPH SEQ ID NO: 4761
- LLSPH SEQ ID NO: 4762
- NGSPH SEQ ID NO: 4763
- PGSPH SEQ ID NO: 4764
- GGSPH SEQ ID NO: 4765
- TGSPH SEQ ID NO: 4766
- SVSPH SEQ ID NO: 4767
- IGSPH SEQ ID NO: 4768
- DGSPH (SEQ ID NO: 4769), LGSPH (SEQ ID NO: 4770), AGSPH (SEQ ID NO: 4771), EGSPH (SEQ ID NO: 4772), SASPH (SEQ ID NO: 4773), YDSPH (SEQ ID NO: 4774), HGSPH (SEQ ID NO: 4775), RDSPH (SEQ ID NO: 4776), NDSPH (SEQ ID NO: 4777), PDSPH (SEQ ID NO: 4778), MGSPH (SEQ ID NO: 4779), QVSPH (SEQ ID NO: 4780), HNSPH (SEQ ID NO: 4781), HPSPH (SEQ ID NO: 4782). or HSSPH (SEQ ID NO: 4783);
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof:
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV particle of any one of embodiments 1-16, wherein [N1]-[N2] is or comprises: (i) GSGSPH (SEQ ID NO: 4695), GHDSPII (SEQ ID NO: 4784), GQDSPH (SEQ ID NO: 4785), VSGSPH (SEQ ID NO: 4786), CSGSPH (SEQ ID NO: 4787), GRGSPH (SEQ ID NO: 4788), CSHSPH (SEQ ID NO: 4789), GQSSPH (SEQ ID NO: 4790), GSHSPH (SEQ ID NO: 4791), GDDSPII (SEQ ID NO: 4792), GIIESPII (SEQ ID NO: 4793), GNYSPH (SEQ ID NO: 4794), RVGSPH (SEQ ID NO: 4795), GSCSPH (SEQ ID NO: 4796).
- GLLSPH (SEQ ID NO: 4797), MSGSPH (SEQ ID NO: 4798), RNGSPH (SEQ ID NO: 4799), TSGSPH (SEQ ID NO: 4800), ISGSPH (SEQ ID NO: 4801), GPGSPH (SEQ ID NO: 4802), ESGSPH (SEQ ID NO: 4803), SSGSPH (SEQ ID NO: 4804), GNGSPII (SEQ ID NO: 4805), ASGSPH (SEQ ID NO: 4806), NSGSPH (SEQ ID NO: 4807), LSGSPH (SEQ ID NO: 4808), GGGSPH (SEQ ID NO: 4809), KSGSPH (SEQ ID NO: 4810), HSGSPH (SEQ ID NO: 4811), GTGSPH (SEQ ID NO: 4812), PSGSPH (SEQ ID NO: 4813), GSVSPH (SEQ ID NO: 4814), RSGSPH (SEQ ID NO: 4815), GIGSPH (SEQ
- WSGSPH (SEQ ID NO: 4817), DSGSPH (SEQ ID NO: 4818), IDGSPH (SEQ ID NO: 4819), GLGSPH (SEQ ID NO: 4820), DAGSPH (SEQ ID NO: 4821), DGGSPH (SEQ ID NO: 4822), MEGSPH (SEQ ID NO: 4823), ENGSPH (SEQ ID NO: 4824), GSASPH (SEQ ID NO: 4825), KNGSPH (SEQ ID NO: 4826), KEGSPH (SEQ ID NO: 4827), AIGSPH (SEQ ID NO: 4828), GYDSPH (SEQ ID NO: 4829), GHGSPH (SEQ ID NO: 4830), GRDSPH (SEQ ID NO: 4831), GNDSPH (SEQ ID NO: 4832), GPDSPH (SEQ ID NO: 4833), GMGSPH (SEQ ID NO: 4834), GQVSPH (SEQ ID NO: 4835), GHNSPH (SEQ ID NO:
- amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different: amino acids, relative to any one of the amino acid sequences in (i).
- SGSPHSK SEQ ID NO: 4839
- HDSPHKS SEQ ID NO: 4840
- SGSPHAR SEQ ID NO: 4841
- SGSPHVK SEQ ID NO: 4842
- QDSPHKS SEQ ID NO: 4843
- SGSPHKK SEQ ID NO: 4844
- SGSPHVR SEQ ID NO: 4845
- SGSPHAS SEQ ID NO: 4846
- SGSPHRK SEQ ID NO: 4847
- SGSPHKT SEQ ID NO: 4848
- SHSPHKS SEQ ID NO: 4849
- QSSPHRS SEQ ID NO: 4850
- RGSPHAS SEQ ID NO: 4851
- RGSPHSK SEQ ID NO: 4852
- SGSPHKF SEQ ID NO: 4853
- SGSPHKI SEQ ID NO: 4854
- SGSPHKL SEQ ID NO: 4855
- SGSPHKY SEQ ID NO: 4856
- SGSPHTR SEQ ID NO: 4857
- SGSPHFL (SEQ ID NO; 4869), LLSPHWK (SEQ ID NO: 4870), NGSPHSK (SEQ ID NO: 4871), PGSPHSK (SEQ ID NO: 4872), GGSPHSK (SEQ ID NO: 4873), TGSPHSK (SEQ ID NO: 4874), SVSPHGK (SEQ ID NO: 4875), SGSPHTK (SEQ ID NO: 4876), IGSPHSK (SEQ ID NO: 4877), DGSPHSK (SEQ ID NO: 4878), SGSPHNK (SEQ ID NO: 4879), LGSPHSK (SEQ ID NO: 4880), AGSPHSK (SEQ ID NO: 4881), EGSPHSK (SEQ ID NO: 4882), SASPHSK (SEQ ID NO: 4883), SGSPHAK (SEQ ID NO: 4884), HDSPHKI (SEQ ID NO: 4885), YDSPHKS (SEQ ID NO: 4886), HDSPHKT (SEQ ID NO: 4887), RGSPHKR
- NDSPHKI SEQ ID NO: 4901
- HDSPHKI. SEQ ID NO: 4902
- HPSPHWK SEQ ID NO: 4903
- HDSPHKM SEQ ID NO: 4904
- HSSPHRS HSSPHRS
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g,, consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
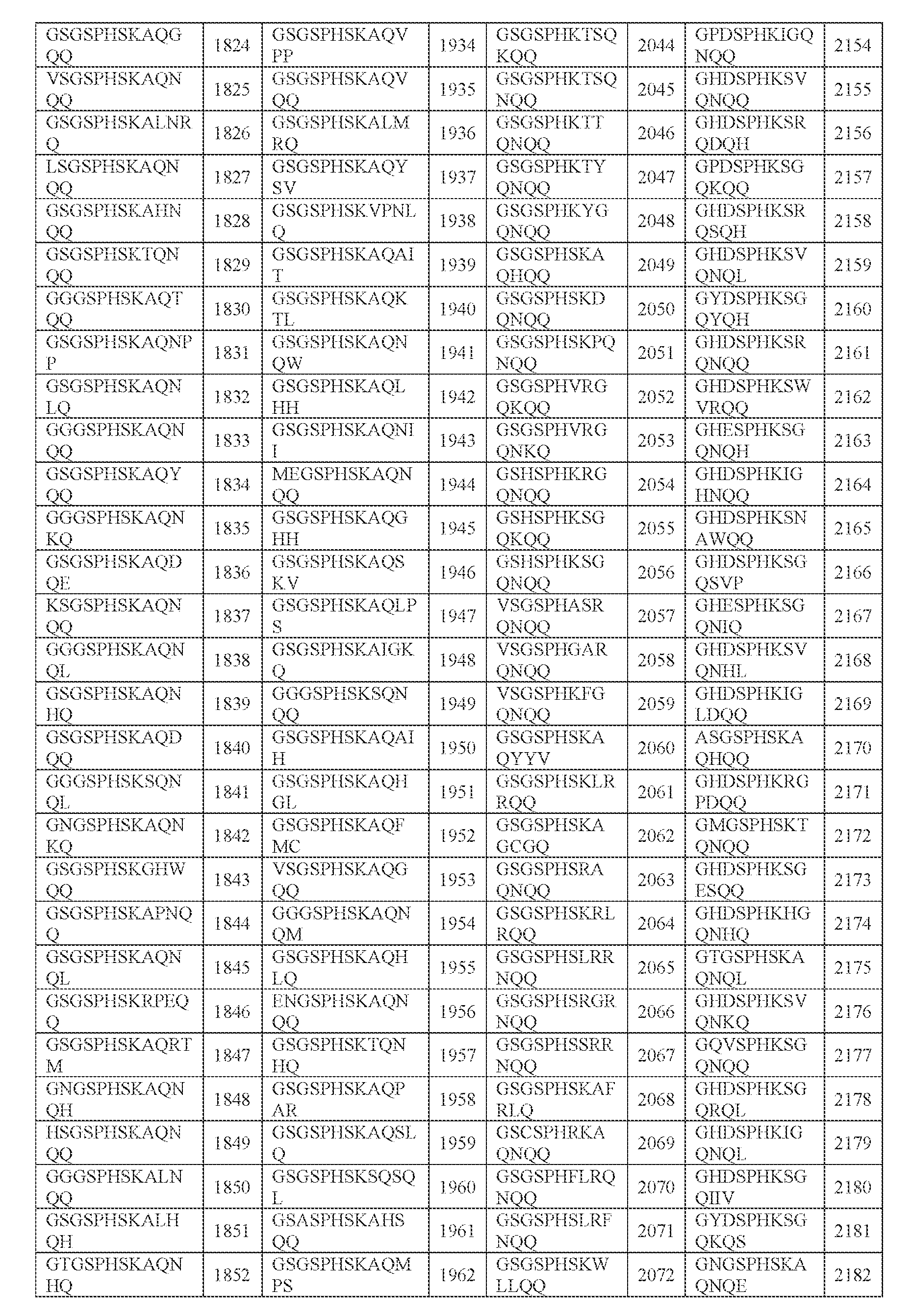
- GSGSPHSKA SEQ ID NO; 4697
- GHDSPHKSG SEQ ID NO; 4698
- GSGSPHARM SEQ ID NO: 4906
- GSGSPHVKS SEQ ID NO: 4907
- GQDSPHKSG SEQ ID NO: 4908
- GSGSPHASR SEQ ID NO; 4909
- GSGSPHVKI SEQ ID NO: 4910
- GSGSPHKKN SEQ ID NO: 491 1
- GSGSPHVRM SEQ ID NO: 4912
- VSGSPHSKA SEQ ID NO: 4913
- CSGSPHSKA SEQ ID NO: 4914
- GSGSPHRKA SEQ ID NO: 4915
- CSGSPHKTS SEQ ID NO: 4916
- CSHSPHKSG SEQ ID NO: 4917
- GQSSPHRSG SEQ ID NO: 4918
- GRGSPHASR SEQ ID NO: 4919
- GRGSPHSKA SEQ ID NO: 4920
- GSGSPHKFG SEQ ID NO
- amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, or 8 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- KSG KSG, or KYG.
- SPHSK SEQ ID NO: 4701
- SPHKS SEQ ID NO: 4704
- SPHKY SEQ ID NO: 4715
- AAV particle of any one of embodiments 1-23 or 26-27, wherein [N1]-[N2]-[N3] comprises SGSPHSK (SEQ ID NO: 4839).
- AAV particle of any one of embodiments 1-22, 24, 26, or 28, wherein [N1]-[N2]- [N3] comprises HDSPHKS (SEQ ID NO: 4840).
- AAV particle of any one of embodiments 1-37, wherein the AAV capsid variant comprises:
- X7 is: Q, W, K, R, G, L, V, S, P, II, K, I, M, A, E, or F;
- X8 is: N, Y, C, K, T, H, R, D, V, S, P, G, W, E, F, A, I, M, Q, or L;
- (c) .X9 is: Q, G, K, H, R, T, L, D, A, P, I, F, V, M, W, Y, S, E, N, or Y; and
- X10 is: Q, II, L, R, W, K, A, P, E, M, I, S, G, N, Y, C, V, T, D, or V; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(d).
- X10 of [N4] is Q, L, or R.
- QNQQ (SEQ ID NO: 5028), WNQQ (SEQ ID NO: 5029).
- QERP (SEQ ID NO: 5109), QDLQ (SEQ ID NO: 5110), QAMH (SEQ ID NO: 5111), QHPS (SEQ ID NO: 5112), PGLQ (SEQ ID NO: 5113), QGIR (SEQ ID NO: 5114), QAPA (SEQ ID NO: 5115), QIPP (SEQ ID NO: 5116), QTQL (SEQ ID NO: 5117), QAPS (SEQ ID NO: 5118), QNTY (SEQ ID NO: 5119), QDKQ (SEQ ID NO: 5120), QNIIL (SEQ ID NO: 5121), QIGM (SEQ ID NO: 5122), LNKQ (SEQ ID NO: 5123), PNQL (SEQ ID NO: 5124), QLQQ (SEQ ID NO: 5125), QRMS (SEQ ID NO: 5126), QGIL (SEQ ID NO: 5127), QDRQ (SEQ ID NO: 5128), RDWQ (SEQ ID NO: 5129), Q
- QNLH (SEQ ID NO: 5206), QNQE (SEQ ID NO: 5207), LNQP (SEQ ID NO: 5208), QNQD (SEQ ID NO: 5209), QNLL (SEQ ID NO: 5210), QLVI (SEQ ID NO: 5211), RTQE (SEQ ID NO: 5212), QTHQ (SEQ ID NO: 5213), QDQH (SEQ ID NO: 5214), QSQH (SEQ ID NO: 5215), VRQQ (SEQ ID NO: 5216).
- AWQQ (SEQ ID NO: 5217), QSVP (SEQ ID NO: 5218), QNIQ (SEQ ID NO: 5219), LDQQ (SEQ ID NO: 5220), PDQQ (SEQ ID NO: 5221), ESQQ (SEQ ID NO: 5222), QRQL (SEQ ID NO: 5223), QIIV (SEQ ID NO: 5224), QKQS (SEQ ID NO: 5225), QSHQ (SEQ ID NO: 5226), QFVV (SEQ ID NO: 5227), QSQP (SEQ ID NO: 5228), QNEQ (SEQ ID NO: 5229), INQQ (SEQ ID NO: 5230), RNRQ (SEQ ID NO: 5231 ), RDQK (SEQ ID NO: 5232), QWKR (SEQ ID NO: 5233), ENRQ (SEQ ID NO: 5234), QTQP (SEQ ID NO: 5235), QKQL (SEQ ID NO: 5236), RN
- amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, or 3 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g,, any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
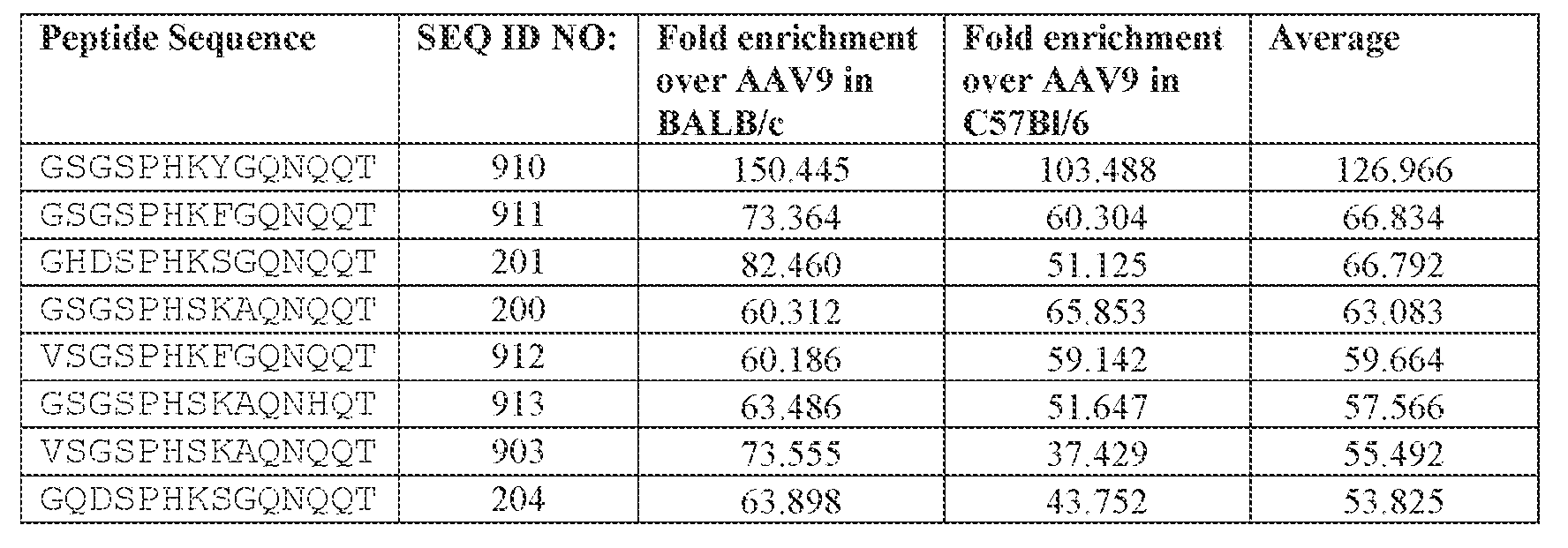
- is or comprises GSGSPHKYGQNQQT (SEQ ID NO: 910).
- amino acid other than T at position 450 e.g., S, Y, M, A, C, I, R, L, D, F, V, Q,
- X A is: T, S, Y, M, A, C, I. R. L. D. F. V. Q. N, H. E, or G;
- X B is: I, M, P, E, N, D, S, A, T, G, Q, F, V, I.,, C, H, R, W, or L;
- X C is: N, M, E, G, Y, W, T, I, Q, F, V, A, L, I, P, K, R, H, S, D, or S; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c).
- AAV particle of embodiment 48 wherein [N0] is or comprises TIN, SMN, TIM, YLS, GLS, MPE, MEG, MEY, AEW, CEW, ANN, IPE.
- TEE TRE, QGE, SEK, NVN, GGE, EFV, SDK, TEQ, EVQ, TEY, NOW, TDV, SDI, NSI, NSL, EVV, TEP, SEL, TWQ, TEV, AVN, GVL, TEN, TEG, TRD, NAT, AEN, AET, ETA, NNL, or any dipeptide thereof.
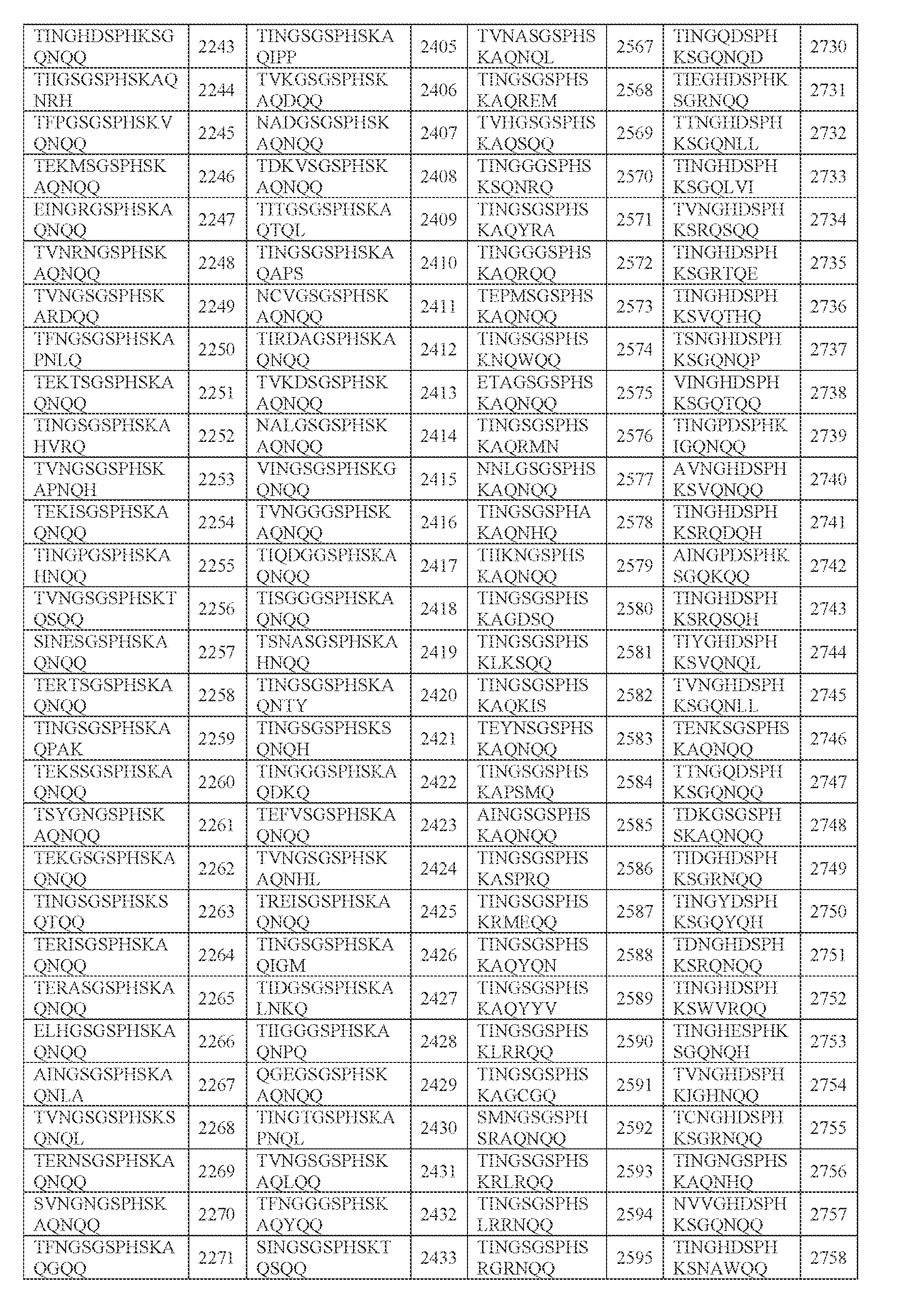
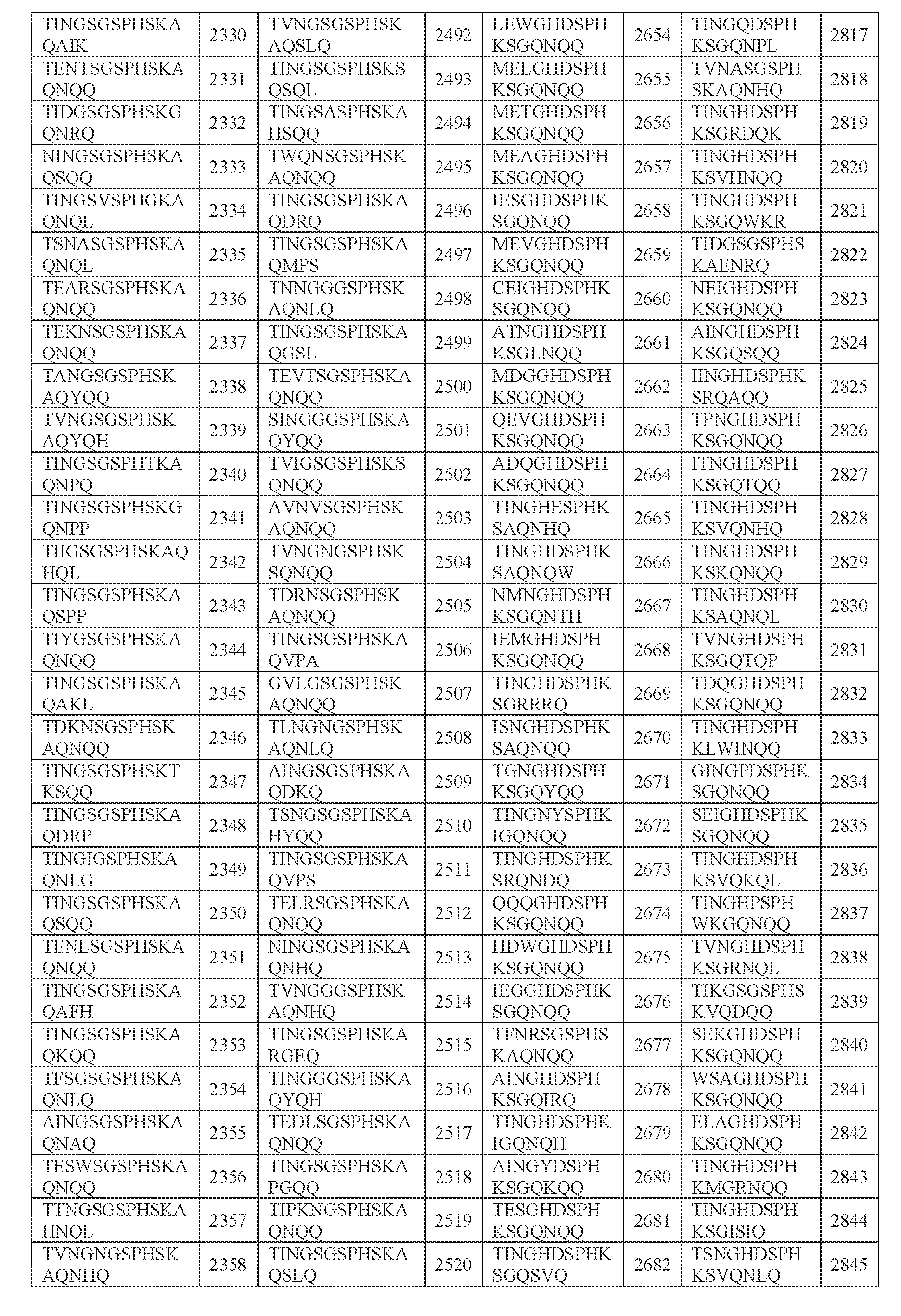
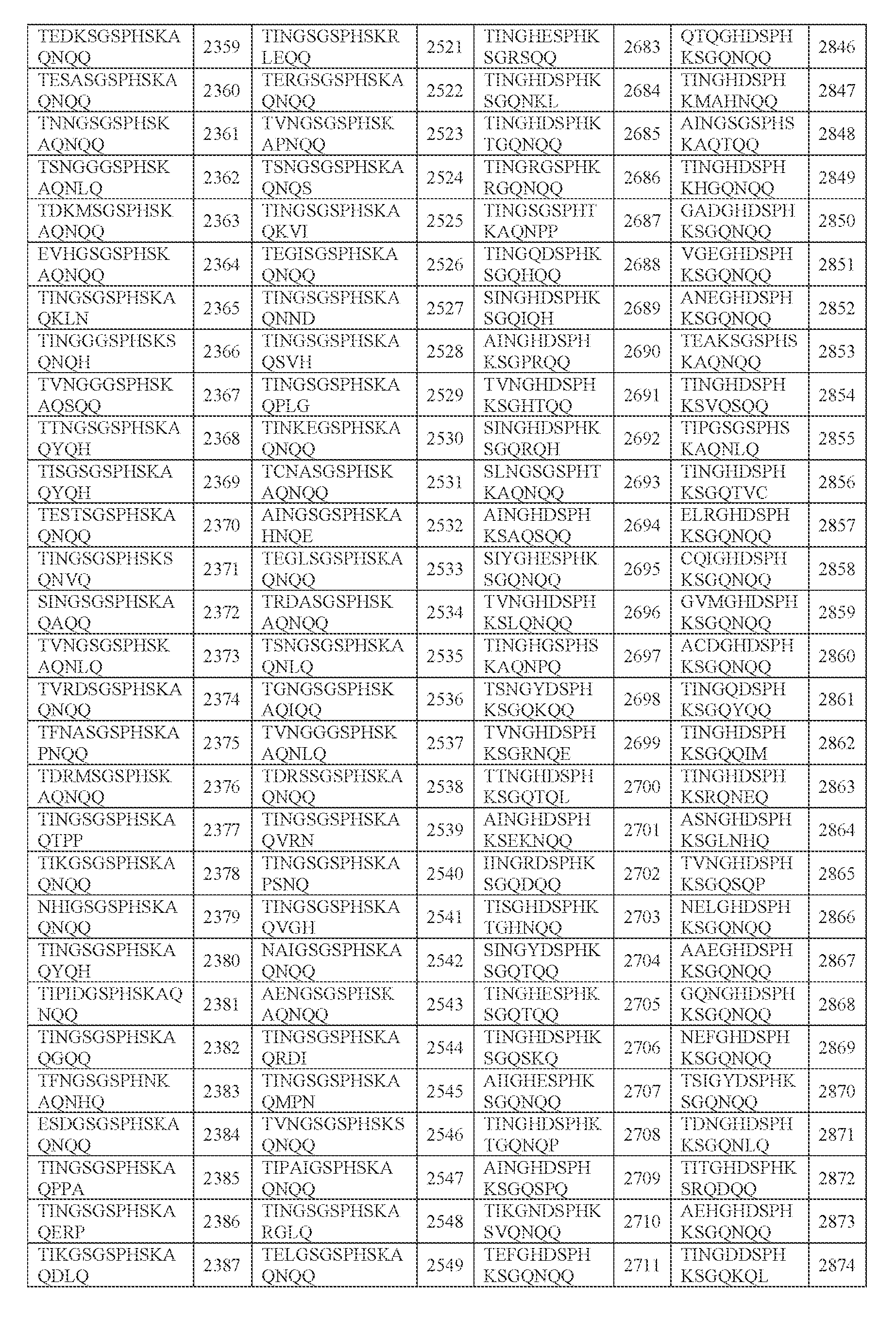
- amino acid sequence of any one of SEQ ID NOs: 2242-2886 (i) the amino acid sequence of any one of SEQ ID NOs: 2242-2886; (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- N0]-[N1 ]-[N2]-[N3]-[N4] is or comprises TINGHDSPHKSGQNQQ (SEQ ID NO: 2243).
- AAV particle of any one of embodiments 48-52, wherein [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TINGSGSPHKYGQNQQT (SEQ ID NO: 5246).
- 450, 451, and 452. (e.g., T450, I451, and N452), numbered according to SEQ ID NO: 138. 59.
- [N0] replaces positions 450-452 (e.g., T450, 1451, and N452), numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
- substitution at position 454 e.g., S454H
- substitution at position 455 e.g., G455D
- AAV particle of any one of embodiments 1-72, 79, or 80, wherein the .AAV capsid variant comprises the amino acid S at position 454 and the amino acid G at position 455, numbered according to SEQ ID NO: 981.
- [N2] corresponds to positions 456-458 (e.g., S456, P457, H458) of any one of SEQ ID NOs: 4 or 36-59.
- [N2]-[N3]-[N4] corresponds to positions 456-465 (e.g., S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465) of SEQ ID NO: 981.
- [N2]-[N3]-[N4] corresponds to positions 456-465 (e.g., S456, P457, H458, K459, S460, G461, Q462, N463, Q464, Q465) of SEQ ID NO: 982.
- [N1] [N2]-[N3]-[N4] corresponds to positions 453-465 (e.g., G453, H454, D455, S456, P457, H458, K459, S460, G461, Q462, N463, Q464, Q465) of SEQ ID NO: 982.
- -[N2]-[N3] corresponds to positions 453-461 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461) of SEQ ID NO: 981.
- positions 453-461 e.g., G453, H454, D455, S456, P457, H458, K459, S460, G461
- N1 ]-[N2]-[N3]- [N4] replaces positions 450-459 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
- positions 450-465 e.g., T450, 1451 , N452, G453, S454, G455, S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465
- [N4] replaces positions 462-465 (e.g., Q462, N463, Q464, and. Q465), numbered according to SEQ ID NO: 4, 36, 981 , or 982.
- replaces positions 462-465 (e.g., Q462, N463, Q464, and Q465), numbered according to SEQ ID NO: 4, 36, 981, or 982.
- [N2]-[N3]-[N4] replaces positions 462-465 (e.g., Q462, N463, Q464, and Q465 ), numbered according to SEQ ID NO: 4, 36, 981, or 982.
- the AAV particle of any one of embodiments .1-119, wirerein the AAV capsid, variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3].
- AAV particle of any one of embodiments 48-122, wherein the A AV capsid vanant comprises, from N-terminus to C -terminus, [N0]-[N1]-[N2]-[N3]-[N4].
- amino acid other T at position 460 e.g., N, I, C, H, R, L, D, Y, A, M, Q, 1, E, K, P, G or S
- AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid other T at position 466 e.g., N, I, C, H, R, L, D, Y, A, M, Q, I, E, K, P, G or S
- amino acid other T at position 466 e.g., N, I, C, H, R, L, D, Y, A, M, Q, I, E, K, P, G or S
- AAV adeno-associated virus
- [A] comprises the amino acid sequence of GSGSPH (SEQ ID NO: 4695).
- X1 is: S, C, F, or V;
- X2 is: K, L, R, I, E, Y, V, or S;
- X3 is: A, R, L, G, I, Y, S, F, or W;
- X4 is: W, Q, R, G, L, V, S, or F;
- X5 is: N, Y, R. C, K, or L;
- X6 is: Q, G, K, R, T, L, or Y;
- (g) X7 is: Q, L, R, or V; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(g).
- X3 is: A, R. or L;
- (g) X7 is: Q, L, or R.
- SLLWNQQ SEQ ID NO: 5247
- SKAQYYV SEQ ID NO: 5248
- SKLRRQQ SEQ ID NO: 5249
- SIWQNQQ SIWQNQQ
- SEQ ID NO: 5250 SKAGCGQ
- SEQ ID NO: 5251 SRAQNQQ
- SEQ ID NO: 5252 SKRLRQQ
- SLRRNQQ SEQ ID NO: 5254
- SRGRNQQ SEQ ID NO: 5255
- SEIVNQQ SEQ ID NO: 5256
- SSRRNQQ SEQ ID NO: 5257
- CLLQNQQ SEQ ID NO: 5258
- SKAFRLQ SEQ ID NO: 5259
- CLAQNQQ SEQ ID NO: 5260
- FLRQNQQ SEQ ID NO: 5261
- SLRFNQQ SEQ ID NO: 5262
- SYLRNQQ SEQ ID NO: 5263
- SKARLRQ SEQ ID NO: 5279
- SKASKRQ SEQ ID NO: 5280
- VRRQNQQ SEQ ID NO: 5281
- SKAQLYR SEQ ID NO: 5282
- SLFRNQQ SEQ ID NO: 5283
- SKAQLTV SEQ ID NO: 5284
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g,, any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- GSGSPHSLLWNQQ (SEQ ID NO: 5285), GSGSPHSKAQYYV (SEQ ID NO: 2060), GSGSPHSKLRRQQ (SEQ ID NO: 2061), GSGSPHSIWQNQQ (SEQ ID NO: 5286), GSGSPHSKAGCGQ (SEQ ID NO: 2062), GSGSPHSRAQNQQ (SEQ ID NO: 2063), GSGSPHSKRLRQQ (SEQ ID NO: 2064), GSGSPHSLRRNQQ (SEQ ID NO: 2065).
- GSGSPHSRGRNQQ (SEQ ID NO: 2066), GSGSPHSEIVNQQ (SEQ ID NO: 5287), GSGSPHSSRRNQQ (SEQ ID NO: 2067), GSGSPHCLLQNQQ (SEQ ID NO: 5288), GSGSPHSKAFRLQ (SEQ ID NO: 2068), GSGSPHCLAQNQQ (SEQ ID NO: 5289), GSGSPHFLRQNQQ (SEQ ID NO: 2070), GSGSPHSLRFNQQ (SEQ ID NO: 2071), GSGSPHSYLRNQQ (SEQ ID NO: 5290), GSGSPHCSLQNQQ (SEQ ID NO: 52.91), GSGSPHVLWQNQQ (SEQ ID NO: 5292), GSGSPHSKWLLQQ (SEQ ID NO: 2072), GSGSPHSLWSNQQ (SEQ ID NO: 5293), GSGSPHSKRRLQQ (SEQ ID NO: 2073), GSGSPH
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof;
- an amino acid other than T at position 450 e.g., S, Y, or G
- an amino acid other than I at position 451 e.g., M or L
- N at position 452 numbered according to SEQ ID NO: 138.
- AAV particle of any one of embodiments 130-134, wherein the .AAV capsid variant further comprises a Y at position 450, an L at position 451, and an S at position 452, numbered according to SEQ ID NO: 138.
- [A] replaces positions 453-455 (e.g., G453, S454, G455), numbered according to SEQ ID NO: 138.
- [A] [B] replaces positions 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, Q459), numbered according to SEQ ID NO: 138.
- positions 453-459 e.g., G453, S454, G455, Q456, N457, Q458, Q459
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV 9 capsid vanant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises [A] [B] (SEQ ID NO; 4699), wherein:
- [A] comprises X 1 X 2 X3 X4 X5 X6, wherein
- X1 is T, M, A, C, I, R, L, D, F, V, Q, N, or II;
- X2 is 1, P, E, N, D, S, A, T, M, or Q;
- X3 is N, E, G, Y, W. M, T, I, K, Q. F. S, V, A, or L;
- X4 is G. D, R, or E;
- X5 is H, Q, N, or D;
- X6 is D or R; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(f); and
- (ti) [B] comprises SPHKSG (SEQ ID NO: 946).
- X1 is: T, M, A, or I;
- X3 is: N, Q, Y, I, M, or V;
- TINGHD SEQ ID NO: 5297
- MPEGHD SEQ ID NO: 5298
- MEGGHD SEQ ID NO: 5299
- MEYGHD SEQ ID NO: 5300
- AEWGIID SEQ ID NO: 5301
- CEWGHD SEQ ID NO: 5302
- ANNGQD SEQ ID NO: 5303
- IPEGHD SEQ ID NO: 5304
- ADMGHD SEQ ID NO: 5305
- IEYGHD SEQ ID NO: 5306
- ADYGHD SEQ ID NO: 5307
- IETGHD SEQ ID NO: 5308
- MEWGHD SEQ ID NO: 5309
- CEYGHD SEQ ID NO: 5310
- RINGHD SEQ ID NO: 5311
- MEIGHD SEQ ID NO: 5312
- LEYGHD SEQ ID NO: 5313
- ADWGHD SEQ ID NO: 5314
- IEIGHD SEQ ID NO: 5315
- TIKDND SEQ ID NO:
- lEVGHD (SEQ ID NO: 5327), MEMGHD (SEQ ID NO: 5328), AEVGHD (SEQ ID NO: 5329), MDAGHD (SEQ ID NO: 5330), VEWGHD (SEQ ID NO: 5331), AEQGHD (SEQ ID NO: 5332), LEWGHD (SEQ ID NO: 5333), MELGHD (SEQ ID NO: 5334).
- METGHD (SEQ ID NO: 5335), MEAGHD (SEQ ID NO: 5336), TINRQR (SEQ ID NO: 5337), lESGHD (SEQ ID NO: 5338), TAKDIID (SEQ ID NO: 5339), MEVGHD (SEQ ID NO: 5340), CEIGHD (SEQ ID NO: 5341), ATNGHD (SEQ ID NO: 5342), MDGGHD (SEQ ID NO: 5343), QEVGHD (SEQ ID NO: 5344), ADQGHD (SEQ ID NO: 5345), NMNGHD (SEQ ID NO: 5346), TPWEHD (SEQ ID NO: 5347), lEMGHD (SEQ ID NO: 5348), TANEHD (SEQ ID NO: 5349), QQQGHD (SEQ ID NO: 5350), TPQDHD (SEQ ID NO: 5351), HDWGIID (SEQ ID NO: 5352), IEGGHD (SEQ ID NO: 5353) (ii) an amino
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV particle of any one of embodiments 147-149, wherein [A][B] comprises: (i) T1NGHDSPHKR (SEQ ID NO: 5354), MPEGHDSPHKS (SEQ ID NO: 5355), MEGGHDSPHKS (SEQ ID NO: 5356), MEYGHDSPHKS (SEQ ID NO: 5357), AEWGHDSPHKS (SEQ ID NO: 5358), CEWGHDSPHKS (SEQ ID NO: 5359), ANNGQDSPHKS (SEQ ID NO: 5360), IPEGHDSPHKS (SEQ ID NO: 5361), ADMGHDSPHKS (SEQ ID NO: 5362), IEYGHDSPHKS (SEQ ID NO: 5363).
- ADYGHDSPHKS (SEQ ID NO: 5364), IETGHDSPHKS (SEQ ID NO: 5365), MEWGHDSPHKS (SEQ ID NO: 5366), CEYGHDSPHKS (SEQ ID NO: 5367), RINGHDSPHKS (SEQ ID NO: 5368), MEIGHDSPHKS (SEQ ID NO: 5369), LEYGHDSPHKS (SEQ ID NO: 5370), ADWGHDSPHKS (SEQ ID NO: 5371), IEIGHDSPHKS (SEQ ID NO: 5372), TIKDNDSPHKS (SEQ ID NO: 5373), DIMGHDSPHKS (SEQ ID NO: 5374), FEQGHDSPHKS (SEQ ID NO: 5375), MEFGHDSPHKS (SEQ ID NO: 5376), CDQGHDSPHKS (SEQ ID NO: 5377).
- LPEGHDSPHKS (SEQ ID NO: 5378), IENGHDSPHKS (SEQ ID NO: 5379), MESGHDSPHKS (SEQ ID NO: 5380), AEIGIIDSPHKS (SEQ ID NO: 5381 ), VEYGHDSPHKS (SEQ ID NO: 5382), TSNGDDSPHKS (SEQ ID NO: 5383), IEYGHDSPHKS (SEQ ID NO: 5384), MEMGHDSPHKS (SEQ ID NO: 5385), AEVGHDSPHKS (SEQ ID NO: 5386), MDAGHDSPHKS (SEQ ID NO: 5387), VEWGHDSPHKS (SEQ ID NO: 5388), AEQGHDSPHKS (SEQ ID NO: 5389), LEWGHDSPHKS (SEQ ID NO: 5390), MELGHDSPHKS (SEQ ID NO: 5391), MEIGHDSPHKS (SEQ ID NO: 5392), MEAGHDSPHKS (SEQ ID NO: 5393), TINRQRSPHKS (
- amino acid sequence comprising any portion of an amino acid sequence in (i). e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof:
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- Q at position 456 e.g., R or L
- N at position 457 e.g., H, K, or R
- Q at position 458 e.g., R or T
- Q at position 459 H
- N or S T at position 460
- positions 450-453 e.g., T450, 1451, N452, G453
- [A][B] replaces positions 450-455 (e.g., T450, 1451, N452, G453, S454, G455), numbered according to SEQ ID NO: 138.
- positions 450-455 e.g., T450, 1451, N452, G453, S454, G455
- AAV particle of any one of embodiments 147-166, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [A] [B] .
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a. viral genome comprising a nucleic acid encoding a modulator ⁇ ' polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]-[N3] (SEQ ID NO: 6407), wherein:
- [N1] comprises X1, X2, and X3, wherein X2 is S and X3 is G;
- [N2] comprises the amino acid sequence SPH;
- (iii) [N3] comprises X4, X5, and X6, wherein X5 is K.
- X5 of [N3] is A, V, T, S, G, R, L, or N: optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid, amino acids in (i) or (ii).
- AA V particle of any one of embodiments 168-170, wherein [N3] comprises SK, TK, NK, AK, KA, KV, KT, KS, KG, KR, KL, or KN.
- SPHSKA SEQ ID NO: 941
- SPHSKV SEQ ID NO: 4737
- SPHSKT SEQ ID NO: 4731
- SPHSKS SEQ ID NO: 962
- SPHSKG SEQ ID NO: 4732
- SPHSKR SEQ ID NO: 978
- SPHTKA SEQ ID NO: 4739
- SPHNKA SEQ ID NO: 4734
- SPHSKL SEQ ID NO: 960
- SPHSKN SEQ ID NO: 4735
- SPHAKA SEQ ID NO: 4736
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV particle of any one of embodiments 168-176, wherein the AAV capsid variant comprises an amino acid other than G at position 453 e.g., M, T, I, E, S, A, N, V, L, K, H P, R, W, or D
- amino acid other than G at position 453 e.g., M, T, I, E, S, A, N, V, L, K, H P, R, W, or D
- AAV particle of any one of embodiments 168-179, wherein [N1] comprises SG, GS, MS, TS, IS, ES, SS, AS, NS, VS, LS, KS, HS, PS, RS, WS, or DS. 181.
- N1 ] is or comprises:
- GSG MSG, TSG, ISG, ESG, SSG, ASG, NSG, VSG, LSG, KSG, HSG, PSG, RSG, WSG, or DSG.
- GSGSPH SEQ ID NO: 4695
- MSGSPH SEQ ID NO: 4798
- TSGSPH SEQ ID NO: 4800
- ISGSPH SEQ ID NO: 4801
- ESGSPH SEQ ID NO: 4803
- SSGSPH SEQ ID NO: 4804
- ASGSPH SEQ ID NO: 4806
- NSGSPH SEQ ID NO: 4807
- VSGSPH SEQ ID NO: 4786
- ESGSPH SEQ ID NO: 4808
- KSGSPH SEQ ID NO: 4810
- HSGSPH SEQ ID NO: 4811
- PSGSPH SEQ ID NO: 4813
- RSGSPH SEQ ID NO: 4815
- WSGSPH SEQ ID NO: 4817
- DSGSPH SEQ ID NO: 4818
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- GSGSPHSKA (SEQ ID NO; 4697), GSGSPHSKV (SEQ ID NO; 4956), MSGSPHSKA (SEQ ID NO; 4957), TSGSPHSKA (SEQ ID NO: 4959), ISGSPHSKA (SEQ ID NO: 4960), GSGSPHSKT (SEQ ID NO: 4962), ESGSPHSKA (SEQ ID NO: 4963), SSGSPHSKA (SEQ ID NO: 4964), GSGSPHSKS (SEQ ID NO: 4953), ASGSPHSKA (SEQ ID NO: 4966), NSGSPHSKA (SEQ ID NO: 4967), VSGSPHSKA (SEQ ID NO: 4913), LSGSPHSKA (SEQ ID NO; 4968), KSGSPHSKA (SEQ ID NO: 4970), GSGSPHSKG (SEQ ID NO: 4972), GSGSPHSKR (SEQ ID NO: 4945), HSGSPHSKA (SEQ ID NO: 4973), PSGSPHSKA (SEQ ID NO: 4931),
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV capsid variant of any one of embodiments 168-186 which comprises an amino acid other than Q at position 456 (e.g., R, P, H, L, K, I, G, S, M, or E), an amino acid other than N at position 457 (e.g., D, V, S, P, T, G, ⁇ , W, E, R, H, K, F, A, I, L, or M), an amino acid other than Q at position 458 (e.g., R, L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, or D), an amino acid other than Q at position 459 (e.g., H, K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G), and/or an amino acid other than T at position 460 (e.g., I, N, S, H, R, L, D, Y,
- Q amino acid other than Q at position 462
- N e.g., N at position 463
- N e.g., D, V, S, P, T, G, Y, W, E, R, H, K, F, A, I, L, or M
- position 465 e.g,, H, K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G
- an amino acid other than T at position 466 e.g., I, N, S, FI, R, L, D, Y, A, or Q
- SEQ ID NO: 981 numbered according to SEQ ID NO: 981 .
- AAV particle of any one of embodiments 168-188, wherein the AAV capsid variant comprises the amino acid Q at position 456, the amino acid N at. position 457, the amino acid Q at position 458, the amino acid Q at position 459, and/or the amino acid T at position 460, numbered according to SEQ ID NO: 138.
- AAV particle of any one of embodiments 168-189, wherein the AAV capsid variant comprises the amino acid Q at position 462, the amino acid N at position 463, the amino acid Q at position 464, the amino acid Q at position 465, and/or the amino acid T at position 466, numbered according to SEQ ID NO: 981.
- X7 is Q, R, P, II, L, K, I, G, S, M, or E;
- X8 is N, D, V, S, P, T, G, Y, W, E, R, H, K, F, A, I, L, or M;
- X9 is Q, R. L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, D;
- X10 is Q, H, K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G; and.
- X11 is T, I, N, S, II, R L, D, Y, A, Q; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(e).
- QNQQT (SEQ ID NO: 5412), QNRHT (SEQ ID NO: 5413), RDQQT (SEQ ID NO: 5414), PNLQT (SEQ ID NO: 5415), HVRQT (SEQ ID NO: 5416), PNQHT (SEQ ID NO: 5417), QSQQT (SEQ ID NO: 5418), QNQQI (SEQ ID NO: 5419), QPAKT (SEQ ID NO: 5420), QTQQN (SEQ ID NO: 5421), QNLAT (SEQ ID NO: 5422), QNQLT (SEQ ID NO: 5423), QGQQT (SEQ ID NO: 5424), LNRQS (SEQ ID NO: 5425), HNQQT (SEQ ID NO: 5426), QNPPT (SEQ ID NO: 5427), QNLQT (SEQ ID NO: 5428), QYQQT (SEQ ID NO: 5429), QDQET (SEQ ID NO: 5430), QNI
- PNQQT (SEQ ID NO: 5434), QNQLI (SEQ ID NO: 5435), PEQQT (SEQ ID NO: 5436), QRTMT (SEQ ID NO: 5437), QNQQH (SEQ ID NO: 5438), LFIQIIT (SEQ ID NO: 5439), QHRIT (SEQ ID NO: 5440), QYIHT (SEQ ID NO: 5441), QKFET (SEQ ID NO: 5442), QFPST (SEQ ID NO: 5443), HNQQR (SEQ ID NO: 5444), QAIKT (SEQ ID NO: 5445).
- QNRQT (SEQ ID NO: 5446), QYQHT (SEQ ID NO: 5447), QN.PQS (SEQ ID NO: 5448), QHQLT (SEQ ID NO: 5449), QSPPT (SEQ ID NO: 5450), QAKLT (SEQ ID NO: 5451), KSQQT (SEQ ID NO: 5452), QDRPT (SEQ ID NO: 5453), QSQQL (SEQ ID NO: 5454), QAFHT (SEQ ID NO: 5455), QKQQD (SEQ ID NO: 5456), QNAQT (SEQ ID NO: 5457), HNQLT (SEQ ID NO: 5458), QNQQY (SEQ ID NO: 5459), QKLNT (SEQ ID NO: 5460), QNVQT (SEQ ID NO: 5461), QAQQT (SEQ ID NO: 5462), QNLQA (SEQ ID NO: 5463), QTPPT (SEQ ID NO: 5464), QYQHA (SEQ ID
- QNHQN (SEQ ID NO: 5514), QPART (SEQ ID NO: 5515), QSLQT (SEQ ID NO: 5516), QSQLT (SEQ ID NO: 5517), QDRQS (SEQ ID NO: 5518), QMPST (SEQ ID NO: 5519), QGSLT (SEQ ID NO: 5520), QVPAT (SEQ ID NO: 5521), QDKQT (SEQ ID NO: 5522), I1YQQT (SEQ ID NO: 5523), QVPST (SEQ ID NO: 5524), RGEQT (SEQ ID NO: 5525), PGQQT (SEQ ID NO: 5526).
- QSLQI (SEQ ID NO: 5527), LEQQT (SEQ ID NO: 5528), QNQST (SEQ ID NO: 5529), QKVIT (SEQ ID NO: 5530), QNNDQ (SEQ ID NO: 5531), QSVHT (SEQ ID NO: 5532), QPLGT (SEQ ID NO: 5533), IINQET (SEQ ID NO: 5534).
- QNLQI (SEQ ID NO: 5535), QIQQT (SEQ ID NO: 5536), QVRNT (SEQ ID NO: 5537), PSNQT (SEQ ID NO: 5538), QVGHT (SEQ ID NO: 5539), QRDIT (SEQ ID NO: 5540), QMPNT (SEQ ID NO: 5541), RGEQT (SEQ ID NO: 5542), QKQQT (SEQ ID NO: 5543), PSLQT (SEQ ID NO: 5544), QRDQT (SEQ ID NO: 5545), QAKGT (SEQ ID NO: 5546), QSAHT (SEQ ID NO: 5547).
- QSTMT (SEQ ID NO: 5548), QREMT (SEQ ID NO: 5549), QYRAT (SEQ ID NO: 5550), QWQQT (SEQ ID NO: 5551), QRMNT (SEQ ID NO: 5552), GDSQT (SEQ ID NO: 5553), QKIST (SEQ ID NO: 5554), PSMQT (SEQ ID NO: 5555), SPRQT (SEQ ID NO: 5556), MEQQT (SEQ ID NO: 5557), QYQNT (SEQ ID NO: 5558), QIIQQT (SEQ ID NO; 5559), INQQT (SEQ ID NO: 5560), PNQQH (SEQ ID NO: 5561), ENRQT (SEQ ID NO; 5562), QTQQA (SEQ ID NO: 5563), or QNQAT (SEQ ID NO: 5564);
- amino acid sequence comprising any portion of an amino acid sequence in (i). e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, or 13 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- K at position 449 e.g., T, E, or N
- T at position 450 e.g., S, E, A, N, V, Q, or G
- an amino acid other than I at position 451 e.g.,
- AAV particle of any one of embodiments 168-196, wherein the AAV capsid variant comprises the amino acid K at position 449, the amino acid T at position 450, the amino acid I at position 451, and/or the amino acid N at position 452, numbered according to SEQ ID NO: 138 or 981.
- X A is K, T, E, or N;
- X C is I, F, E, V, L. D. S. C, T, A. N. H. R, G, or W;
- X D is N, I, P, K, R, H, S, M, Q, D, T, L, A, ⁇ , V, F, E, W, or G; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids m (a)-(d).
- KTII SEQ ID NO: 5565
- KTFP SEQ ID NO: 5566
- KTEK SEQ ID NO: 5567
- KTVN SEQ ID NO: 5568
- KTFN SEQ ID NO: 5569
- KT1N SEQ ID NO: 5570
- TTIN SEQ ID NO: 5571
- KSIN SEQ ID NO: 5572
- KTER SEQ ID NO: 5573
- KELH SEQ ID NO: 5574
- KAIN SEQ ID NO: 5575
- KTDN SEQ ID NO: 5576
- KTFH SEQ ID NO: 5577
- KTSN SEQ ID NO: 5578
- ETIN SEQ ID NO: 5579
- NTIN SEQ ID NO: 5580
- KTEN SEQ ID NO: 5581
- KTSS SEQ ID NO: 5582
- KTCN SEQ ID NO: 5583
- KTEH SEQ ID NO: 5584
- KAEM SEQ ID NO: 5585
- KATN SEQ ID NO: 5586
- KAIK SEQ ID NO: 5587
- KTDK SEQ ID NO: 5588
- KTFK SEQ ID NO: 5589
- KSDQ SEQ ID NO: 5590
- KTEI SEQ ID NO: 5591
- KTID SEQ ID NO: 5592
- KNTN SEQ ID NO: 5593
- KTET SEQ ID NO: 5594
- KIEL SEQ ID NO: 5595
- KNTN SEQ ID NO: 5596
- KTEA SEQ ID NO: 5597
- KT AN SEQ ID NO: 5598
- NTIY SEQ ID NO: 5599
- KFS SEQ ID NO: 5600
- KTES SEQ ID NO: 5601
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, or 3 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, or 17 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]-[N3] (SEQ ID NO: 6408), wherein:
- [N1] comprises X1, X2, and X3, wherein X2 is an amino acid other than S and X3 is an amino acid other than G;
- [N2] comprises the amino acid sequence SPH;
- (iii) [N3] comprises X4, X5, and X6, wherein X4 is K.
- X5 of [N3] is S, I, T. R. H, Y, L, or M;
- X6 of [N3] is G, A, L, E, V, R, W, N, Q, or K; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (i) or (ii).
- KT, KR, KH, KY, KL, KM SG, IG, TG, RG, SA, SL, SE, SV, SR, SW, SN, IIG, YG, SQ, IV, SK, LW, MG, or MA.
- KSG KIG, KTG, KRG, KSA, KSL, KSE, KSV, KSR, KSW, KSN, KIIG, KYG, KSQ, KIV, KSK, KLW, KMG, or KMA.
- AA V particle of any one of embodiments 203-208, wherein [N2]-[N3] comprises SPHKS (SEQ ID NO: 4704), SPIIKI (SEQ ID NO: 4713), SPIIKT (SEQ ID NO: 471 1), SPHKR (SEQ ID NO: 4717), NPHKS (SEQ ID NO: 5661), SPHKH (SEQ ID NO: 4728),
- SPHKY SEQ ID NO: 4715
- SPHKL SEQ ID NO: 4714
- SPHKM SEQ ID NO: 4729
- SPHKSG SEQ ID NO: 946
- SPHKIG SEQ ID NO: 958
- SPHKTG SEQ ID NO: 4738
- SPHKRG SEQ ID NO: 974
- NPHKSG SEQ ID NO: 5662
- SPHKSA SEQ ID NO: 977
- SPHKSL SEQ ID NO: 4740
- SPHKSE SEQ ID NO: 4741
- SPHKSV SEQ ID NO: 4742
- SPHKSR SEQ ID NO: 951
- SPHKSW SEQ ID NO: 4743
- SPHKSN SEQ ID NO: 4744
- SPHKHG SEQ ID NO: 4745
- SPHKYG SEQ ID NO: 966
- SPHKSQ SEQ ID NO: 4746
- SPHKIV SEQ ID NO: 5663
- SPHKSK SEQ ID NO: 4747
- SPHKLW SEQ ID NO: 4748
- SPHKMG SEQ ID NO: 4750
- SPHKAIA SEQ ID NO: 4751
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV particle of any one of embodiments 203-211, wherein the AAV capsid variant comprises an amino acid other than G at position 453 e.g.. A, K, W, R, L, I, M, N, T, E, Q, Y, H, F, or V
- amino acid other than G at position 453 e.g.. A, K, W, R, L, I, M, N, T, E, Q, Y, H, F, or V
- AAV particle of any one of embodiments 203-212, wherein the AAV capsid variant composes the amino acid G at position 453, numbered according to SEQ ID NO: 138 or 981.
- X1 of [N1] is G, A, K, W, R, L, I, M, N, T, E, Q, Y, H, F, or V;
- (li) X2 of [N1] is H, Y, R . Q. N, P, or D;
- X3 of [N1] is D, E, G, V, or N; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (i), (ii), or (iii).
- Q V. DD, HN, or NG comprises GH, HD, GY, GR, GQ, AH, GN, KH, GP, WH, RH, LH, TH, MH, GD, NH, TH, EH, QH, YH, HH , F H , VH, YD, HE, RG, QI).
- RD ND, PI).
- AAV capsid variant of any one of embodiments 203-217, wherein [N1] is or comprises GHD, GYD, GHE, GRG, GQD, GRD, AHD, GND, KHD, GPD, WHD, RHD, LHD, GQV, IHD, MHD, GDD, GHN, NHD, THD, GNG, EHD, QHD, YHD, HHD, FHD, or VHD.
- AAV particle of any one of embodiments 203-219, wherein [N1]-[N2] comprises HDSPH (SEQ ID NO: 4703).
- GHDSPH (SEQ ID NO: 4784).
- GYDSPH (SEQ ID NO: 4829), GHESPH (SEQ ID NO: 4793), GRGSPH (SEQ ID NO: 4788), GHDNPH (SEQ ID NO: 5664), GQDSPH (SEQ ID NO: 4785), GRDSPH (SEQ ID NO: 4831), AHDSPH (SEQ ID NO: 5665), GNDSPH (SEQ ID NO: 4832), KHDSPH (SEQ ID NO: 5666), GPDSPH (SEQ ID NO: 4833), WHDSPH (SEQ ID NO: 5667), RHDSPH (SEQ ID NO: 5668), LHDSPH (SEQ ID NO: 5669), GQVSPH (SEQ ID NO: 4835), IHDSPH (SEQ ID NO: 5670), MHDSPH (SEQ ID NO: 5671), GDDSPH (SEQ ID NO: 4792), GHNSPH (SEQ ID NO: 4836), NHDSPH (
- amino acid sequence comprising any portion of an amino acid sequence in (i). e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
- an ami no acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- GHDSPHKSG (SEQ ID NO: 4698), GHDSPHKIG (SEQ ID NO: 4996), GYDSPHKSG (SEQ ID NO: 4997), GHESPHKSG (SEQ ID NO: 4998), GHDSPHKTG (SEQ ID NO: 4999), GRGSPHKRG (SEQ ID NO: 5000), GHDNPHKSG (SEQ ID NO: 5680), GQDSPHKSG (SEQ ID NO: 4908), GHDSPHKSA (SEQ ID NO: 4940), GHDSPHKSL (SEQ ID NO: 5001), GHDSPHKSE (SEQ ID NO: 5003), GRDSPHKSG (SEQ ID NO: 5004), AHDSPHKSG (SEQ ID NO: 5681), GNDSPHKSV (SEQ ID NO: 5005), AHDSPHKIG (SEQ ID NO: 5682), GHESPHKSA (SEQ ID NO: 4939), GQDSPHKIG (SEQ ID NO: 5006), GHDSPHKSV (SEQ ID NO:
- GHDSPHKHG (SEQ ID NO: 5013), EHDSPHKSG (SEQ ID NO: 5686), GQVSPHKSG (SEQ ID NO: 5014), IHDSPHKSG (SEQ ID NO: 5687), MHDSPHKSG (SEQ ID NO: 5688), GDDSPHKSV (SEQ ID NO: 5015).
- GHNSPHKSG (SEQ ID NO: 5016), NHDSPHKSG (SEQ ID NO: 5689), THDSPHKSG (SEQ ID NO: 5690), GNGSPHKRG (SEQ ID NO: 5017), EHDSPHKSG (SEQ ID NO: 5691), GIIDSPHKYG (SEQ ID NO: 5018), GHDSPHKSQ (SEQ ID NO: 5019), QHDSPHKSG (SEQ ID NO: 5692), RHDSPHKIV (SEQ ID NO: 5693), YHDSPHKSG (SEQ ID NO: 5694).
- GNDSPHKIG (SEQ ID NO: 5020), HHDSPHKSG (SEQ ID NO: 5695), GHDSPHKSK (SEQ ID NO: 5021), EHDSPHKSG (SEQ ID NO: 5696), GHDSPHKLW (SEQ ID NO: 5022), YHDSPHKSG (SEQ ID NO: 5697), GIIDSPHKMG (SEQ ID NO: 5024), GHDSPHKMA (SEQ ID NO: 5025), or GDDSPHKSG (SEQ ID NO: 4938);
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- amino acid other than Q at position 456 e.g., R,
- amino acid other than Q at position 462 e.g., R, P
- X7 is Q, R, P, H , L, K, I, G, S, M, or E;
- X8 is N, D, V, S, P, T, G, Y, W, E, R, H, K, F, A, 1, L, or M:
- X9 is Q, R, L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, D;
- X10 is Q, H , K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G;
- X11 is T, I, N, S, H, R, L, D, Y, A, Q; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid, amino acids in (a)-(e).
- QNQQT (SEQ ID NO: 5412), Q1RQT (SEQ ID NO: 5698), QNQHA (SEQ ID NO: 5699), QKQQT (SEQ ID NO: 5543), QSVQT (SEQ ID NO: 5700), RSQQT (SEQ ID NO: 5701), QNKLE (SEQ ID NO: 5702), QNQQK (SEQ ID NO: 5703), QHQQA (SEQ ID NO: 5704), QIQHT (SEQ ID NO: 5705), PRQQT (SEQ ID NO: 5706), HTQQT (SEQ ID NO: 5707), QRQHT (SEQ ID NO: 5708), QSQQT (SEQ ID NO: 5418), QNQQS (SEQ ID NO: 5709), RNQET (SEQ ID NO: 5710), QTQLT (SEQ ID NO: 5478), KNQQT (SEQ ID NO: 5711), QD
- QYQHT (SEQ ID NO: 5447), QNQWT (SEQ ID NO: 5503), QNQHT (SEQ ID NO: 5718), QTRQT (SEQ ID NO: 5719), QNLHT (SEQ ID NO: 5720), LNQQT (SEQ ID NO: 5471), QNQET (SEQ ID NO: 5721), QHLQT (SEQ ID NO: 5513), LNQPT (SEQ ID NO: 5722), QNQDT (SEQ ID NO: 5723), RNQQT (SEQ ID NO: 5724), QNLLT (SEQ ID NO: 5725).
- QLVIT (SEQ ID NO: 5726), RTQET (SEQ ID NO: 5727), QTHQT (SEQ ID NO: 5728), QNQPA (SEQ ID NO: 5729), QDQHT (SEQ ID NO: 5730), QSQHT (SEQ ID NO: 5731), RNQQI (SEQ ID NO: 5732), VRQQT (SEQ ID NO: 5733), QNQHS (SEQ ID NO: 5734), AWQQT (SEQ ID NO: 5735), QSVPT (SEQ ID NO: 5736), QNIQP (SEQ ID NO: 5737), QNHLN (SEQ ID NO: 5738), LDQQT (SEQ ID NO: 5739), PDQQS (SEQ ID NO: 5740), ESQQT (SEQ ID NO: 5741), QNKQT (SEQ ID NO: 5742), QRQLT (SEQ ID NO: 5743), Q
- QSLQT (SEQ ID NO: 5516).
- RNRQT (SEQ ID NO: 5750), QSKQT (SEQ ID NO: 5751), QNPLT (SEQ ID NO: 5752), RDQKT (SEQ ID NO: 5753), HNQQN (SEQ ID NO: 5754), QWKRT (SEQ ID NO: 5755), QSQQI (SEQ ID NO: 5476), QAQQT (SEQ ID NO: 5462), QNHQI (SEQ ID NO: 5756), QNQQA (SEQ ID NO: 5757), QNQLN (SEQ ID NO: 5758), QTQPT (SEQ ID NO: 5759), INQQT (SEQ ID NO: 5560), QKQLT (SEQ ID NO: 5760), RNQLA (SEQ ID NO: 5761), RNQQS (SEQ ID NO: 5762), ISIQT (SEQ ID NO: 5763), QNQQ
- QYQQI (SEQ ID NO: 5767), QQIMT (SEQ ID NO: 5768), QNEQS (SEQ ID NO: 5769), LNHQT (SEQ ID NO: 5770), QMIHT (SEQ ID NO: 5771), RNHQS (SEQ ID NO: 5772), QKMNT (SEQ ID NO: 5773), QSQQN (SEQ ID NO: 5774), QYQIIA (SEQ ID NO: 5465);
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g,, any 2, 3, or 4 amino acids, e.g,, consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, .10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof;
- an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i). 231.
- T T
- T at position 450 e.g.. A, S, I, V, N, E, Y, C, G, W, or Q
- an amino acid other than I at position 451 e.g., E, V, S, T, N, D
- AAV particle of any one of embodiments 203-232, wherein the AAV capsid variant comprises the amino acid K at position 449, the amino acid T at position 450, the amino acid 1 at position 45 I , and/or the amino acid N at position 452, numbered according to SEQ ID NO: 138 or 982.
- X B is T, A, S, I, V, N, E, Y, C, G, W, or Q;
- X C is I, E, V, S, T, N, D, C, G, Q, L, P, A;
- AAV capsid, variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(d).
- KAIN SEQ ID NO: 5575
- KTIN SEQ ID NO: 5570
- KTES SEQ ID NO: 5601
- TTIN SEQ ID NO: 5571
- KSIN SEQ ID NO: 5572
- KTVN SEQ ID NO: 5568
- KSIY SEQ ID NO: 5775
- KTSN SEQ ID NO: 5578
- KTTN (SEQ ID NO: 5602), KIIN (SEQ ID NO: 5776), KITS (SEQ ID NO: 5606), KA.II (SEQ ID NO: 5777), KTIK (SEQ ID NO: 5612), KTEF (SEQ ID NO: 5624), KITT (SEQ ID NO: 5620), KTNN (SEQ ID NO: 5604), KTID (SEQ ID NO: 5592), KAIS (SEQ ID NO: 5778), KTVD (SEQ ID NO: 5779), KTIE (SEQ ID NO: 5780), KTEG (SEQ ID NO: 5647), KVIN (SEQ ID NO: 5623), KAVN (SEQ ID NO: 5645), KTIY (SEQ ID NO: 5781), KTDN (SEQ ID NO: 5576), KTCN (SEQ ID NO: 5583), KNVV (SEQ ID NO: 5782), KTEL (SEQ ID NO: 5595), KTDA (SEQ ID NO
- KTET SEQ ID NO: 5594
- KTIG SEQ ID NO: 5797
- KTDP SEQ ID NO: 5798
- KELV SEQ ID NO: 5799
- KELM SEQ ID NO: 5800
- KNEI SEQ ID NO: 5801
- KTPN SEQ ID NO: 5802
- KTDQ SEQ ID NO: 5805
- KGIN SEQ ID NO: 5806
- KSEI SEQ ID NO: 5807
- KSEK SEQ ID NO: 5627
- KWSA SEQ ID NO: 5808
- KELA SEQ ID NO: 5809
- KQTQ SEQ ID NO: 5810
- KGAD SEQ ID NO: 5811
- KVGE SEQ ID NO: 5812
- KANE SEQ ID NO: 5813
- KTDT SEQ ID NO: 5810
- KELR (SEQ ID NO: 5816), KCQI (SEQ ID NO: 5817), KGVM (SEQ ID NO: 5818), KA CD (SEQ ID NO: 5819), KNEE (SEQ ID NO: 5820), KAAE (SEQ ID NO: 5821), KGQN (SEQ ID NO: 5822), KNEE (SEQ ID NO: 5823), KTSI (SEQ ID NO: 5824), KAEH (SEQ ID NO: 5616), KCDQ (SEQ ID NO: 5825), KEIL (SEQ ID NO: 5826), KIER (SEQ ID NO: 5573), KNAI (SEQ ID NO: 5650), KTDK (SEQ ID NO: 5588), KTPD (SEQ ID NO: 5827), KTIH (SEQ ID NO: 5659), or KTEI (SEQ ID NO: 5591);
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, or 3 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV particle of embodiment 234or 235 wherein [N0]-[N1]-[N2]-[N3]-[N4] is or comprises:
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, or 17 amino acids, e.g., consecutive amino acids, thereof;
- AAV particle of any one of embodiments 234-236, wherein [N0]-[N1]-[N2]-[N3]- [N4] is or comprises KAEIGHDSPHKSGQNQQT (SEQ ID NO: 1754).
- [N0] replaces positions 449-452 (e.g., K449, T450, 1451, and N452), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
- [N0] replaces positions 449-452 (e.g., K449, T450, 1451, and N452), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
- [N1] replaces positions 453-455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
- X3 of [N1] corresponds to position 455 (e.g., G455), wherein (i), (ii), and (iii) are numbered according to SEQ ID NO: 138 or SEQ ID NO:
- X3 of [N1] corresponds to position 455 (e.g., G455); wherein (i), (ii), and (iii) are numbered according to SEQ ID NO: 138 or SEQ ID NO:
- positions 459-460 e.g., S459, K460, A461 of SEQ ID NO: 36, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51 , 52, 53, 54, 55, 57, or 59.
- 456-461 e.g., S456, P457, H458, S459, K460, A461
- [N3] is present immediately subsequent to [N1]-[N2] and replaces positions 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
- [N1 ]-[N2]- [N3]-[N4] replaces positions 453-460 (e.g., G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138. 280.
- positions 453-460 e.g., G453, S454, G455, Q456, N457, Q458, Q459, and T460
- [N1]-[N2]-[N3]-[N4] corresponds to positions 453-466 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465, and T466) of SEQ ID NO: 981.
- [N1]-[N2]-[N3] corresponds to positions 453-461 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461) of SEQ ID NO: 981 .
- positions 453-466 e.g... G453, H454, D455, S456, P457, H458, K459, S460, G461, Q462, N463, Q464, Q465, T466) of SEQ ID NO: 982.
- positions 453-461 e.g., G453, H454, D455, S456, P457, H458, K459, S460, G461
- [N0]-[N1]- [N2]-[N3]-[N4] replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
- positions 449-460 e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460
- K449, T450, 1451, N452, G453, S454, G455, S456, P457, H458, S459, K460, A461 , Q462, N463, Q464, Q465, T466 of SEQ ID NO: 981.
- positions 449-466 e.g., K449, T450, 1451, N452, G453, H454, D455, S456, P457, II458, K459, S460, G461 , Q462, N463, Q464, Q465, T466) of SEQ ID NO: 982.
- [N4] replaces positions 462-466 (e.g., Q462, N463, Q464, Q465, and T466), numbered according to SEQ ID NO: 4, 36, 981, or 982.
- [N2]-[N3]- [N4] replaces positions 462-466 (e.g., Q462, N463, Q464, Q465, and T466), numbered according to SEQ ID NO: 4, 36, 981 , or 982,
- the AAV particle of any one of embodiments 168-294, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N2]-[N3],
- AAV particle of any one of embodiments 168-295, wherein the AAV capsid variant comprises, from N-terminus to C-tenninus, [N1]-[N2]-[N3
- AAV particle of any one of embodiments 168-296, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3].
- AAV particle of any one of embodiments 168-297, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4],
- AAV particle of any one of embodiments 168-298, wherein the AAV capsid variant composes, from N-terminus to C-tenninus, [N0]-[N1]-[N2.]-[N3]-[N4].
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV 9 capsid vanant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the formula [A
- [A] comprises GSGSPH (SEQ ID NO: 4695);
- X1 is S, I, F, V, C, Y, W, R, P, L, Q, M, K, or G;
- X2 is K, M, R, F, V, C. P, Y, L, W, G, N, S. T, I, or A:
- X3 is A, Y, L, R, W, C, T, F, H, I, P, M, K, S, V, G, Q, or N;
- X4 is Q, M, F, K, H, R, C, W, P, V, L, G, S, Y, I, A, T, D, N, or E;
- X5 is A, N, Y, R, K, L. I, M, Q, S, C, W, F, T, G, V, or P; optionally wherein the AAV capsid, variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(e).
- X1 is S, L, R, V, or P;
- X2 is K, C, F, L, P, R, S, or V;
- X3 is A, C, F, I, K, L, M, P, R, T, W, or Y:
- X4 is Q, R, S, T, C, F, K, L, P or Y ;
- X5 is N, R, S, T, K, M, Q or Y; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a. conservative substitution, of any of the aforesaid amino acids in (a)-(e).
- AAV particle of embodiment 300 or 301, wherein [B] comprises SKA, SMY, SKL, SKR, SKW, SRC, SFT, SKF, IVW, SKY, SCH, FPW, SKI, VYY, SLY, SKP, SRF, SRM, SVK, SWA, SLW, SFR, SKK, SYA, SCS, SGA, SFP, SFF, SMC, SKT, SGK, FYR, CRV, YGI, VNC, SLA, WSY, RWL, PSC, SSW, SKG, VPW, SGC, STT, PKR, SKC, WVP, SFW, RIK, SKM, LRW, LPT, SYM, LLC, RCC, LCV, SYL, QGC, MAF, SFQ, SLC, RPW, RPR, SCP, SVR, SLP, VYII, SYT, LVY, YRY,
- YMN AFY, LKR, RHR, AQK, WRL, CRN, TCN, FFI, AQY, WQN.
- YFM ARQ, HQN, IRR, YQN, YWN, APS, FWN, AQC, MRN, K.KN, APN, WKN, ARW.
- RPN RPN, KVF, AFN, ACS, RLW, SRN, CPN, ACN, FRQ, PEN, FGN, CQN, LFW, TRK, KRN, RQN, VQN, IQN, AQR, PFR, AWN, RSY, LQN, WLN, RRA, AQT, GCT, RYT, TPN, ARM, CFL, PQN, WSN, FKN, KQN. APR. RYN, MIC, TQN, WKS, AAR, LTR, IRG. LVN, FQN, ACQ, WGL, IL R.
- KAQ MYM, KAF, KLK, KRH, KWR, RCR, FTC, KFF, VWQ, KYF, KAR, CHQ, PWQ, KIR, YYQ, LYW, KPK, RFW, RMR, VKK, WAP, LWK, FRP, KKV, YAF, KAC, KRL, CSR, RCP, GAC, KFR, FFG, MCQ, KEF, KTR, GKR, YRQ, RVQ, GIQ, NCQ, KPF, LAW, KRS, SYQ, WLQ, KRR, KGC, KRY, GCQ, FTP, ITC.
- VAQ VAQ
- LYQ KVR
- ALQ SCT
- KNS KRK
- CTQ TCL
- YAR KQR
- KRV SGQ
- YYS LTC
- CQS CQS
- KAK KPQ
- PFQ PFQ
- KCT or VFE.
- AAV particle of any one of embodiments 300-302, wherein [B] comprises SKAQ (SEQ ID NO: 5829), SMYM (SEQ ID NO: 5830), SKAF (SEQ ID NO: 5831), SKLK (SEQ ID NO: 5832), SKRTI (SEQ ID NO: 5833), SKWR (SEQ ID NO: 5834), SRCR (SEQ ID NO: 5835), SFTC (SEQ ID NO: 5836), SKFF (SEQ ID NO: 5837), IVWQ (SEQ ID NO: 5838), SKYF (SEQ ID NO: 5839), SKAR (SEQ ID NO: 5840), SCIIQ (SEQ ID NO: 5841), FPWQ (SEQ ID NO: 5842), SKIR (SEQ ID NO: 5843), VYYQ (SEQ ID NO: 5844), SLYW (SEQ ID NO: 5845), SKPK (SEQ ID NO: 58
- SK U' (SEQ ID NO: 5855), SKRL (SEQ ID NO: 5856), SCSR (SEQ ID NO: 5857), SRCP (SEQ ID NO: 5858), SGAC (SEQ ID NO: 5859), SKFR (SEQ ID NO: 5860), SFPF (SEQ ID NO: 5861), SFFG (SEQ ID NO: 5862), SMCQ (SEQ ID NO: 5863), SKLF (SEQ ID NO: 5864), SKTR (SEQ ID NO: 5865), SGKR (SEQ ID NO: 5866), FYRQ (SEQ ID NO: 5867), CRVQ (SEQ ID NO: 5868), YGIQ (SEQ ID NO: 5869), VNCQ (SEQ ID NO: 5870), SKPF (SEQ ID NO: 5871), SLAW (SEQ ID NO: 5872), SKRS (SEQ ID NO: 5873), WSYQ (
- LVYQ (SEQ ID NO: 5923), YRYQ (SEQ ID NO: 5924), SWLK (SEQ ID NO: 5925), CPAQ (SEQ ID NO: 5926), SMCT (SEQ ID NO: 5927), SPPD (SEQ ID NO: 5928), SKRN (SEQ ID NO: 5929), RWTQ (SEQ ID NO: 5930), PRKQ (SEQ ID NO: 5931), SKCS (SEQ ID NO: 5932), PFVQ (SEQ ID NO: 5933), SKLP (SEQ ID NO: 5934), SKSE (SEQ ID NO: 5935), WVAQ (SEQ ID NO: 5936), SLYQ (SEQ ID NO: 5937), SKVR (SEQ ID NO: 5938).
- CALQ (SEQ ID NO: 5939), SSCT (SEQ ID NO: 5940), SKNS (SEQ ID NO: 5941), SKRK (SEQ ID NO: 5942), LCTQ (SEQ ID NO: 5943), STCL (SEQ ID NO: 5944), SY AR (SEQ ID NO: 5945), SKQR (SEQ ID NO: 5946), SKRY (SEQ ID NO: 5947), KSGQ (SEQ ID NO: 5948), SYYS (SEQ ID NO: 5949), SLTC (SEQ ID NO: 5950), SCQS (SEQ ID NO: 5951), SKAK (SEQ ID NO: 5952), SKPQ (SEQ ID NO: 5953), FPFQ (SEQ ID NO: 5954), SKCT (SEQ ID NO: 5955), SAFE (SEQ ID NO: 5956), GRYQ (SEQ ID NO: 5957), KAQA (SEQ
- KWRL (SEQ ID NO: 5965), RCRN (SEQ ID NO: 5966), FTCN (SEQ ID NO: 5967), KFFI (SEQ ID NO: 5968), KAQY (SEQ ID NO: 5969), VWQN (SEQ ID NO: 5970), KYFM (SEQ ID NO: 5971), KARQ (SEQ ID NO: 5972), CHQN (SEQ ID NO: 5973), PWQN (SEQ ID NO: 5974), KIRR (SEQ ID NO: 5975), YYQN (SEQ ID NO: 5976), LYWN (SEQ ID NO: 5977), KPKR (SEQ ID NO: 5978), KAFS (SEQ ID NO: 5979), RFWN (SEQ ID NO: 5980), KAQC (SEQ ID NO: 5981), RMRN (SEQ ID NO: 5982), VKKN (SEQ ID NO: 5983), WAPN (SEQ ID
- FRPN (SEQ ID NO: 5987), KKVF (SEQ ID NO: 5988), YAFN (SEQ ID NO: 5989), KACS (SEQ ID NO: 5990), KRLW (SEQ ID NO: 5991), CSRN (SEQ ID NO: 5992), RCPN (SEQ ID NO: 5993), GACN (SEQ ID NO: 5994), KFRQ (SEQ ID NO: 5995), FPFN (SEQ ID NO: 5996), FFGN (SEQ ID NO: 5997), MCQN (SEQ ID NO: 5998), KLFW (SEQ ID NO: 5999), KTRK (SEQ ID NO: 6000), GKRN (SEQ ID NO: 6001), YRQN (SEQ ID NO: 6002), RVQN (SEQ ID NO: 6003), GIQN (SEQ ID NO: 6004), KAQR (SEQ ID NO: 6005), NCQN (SEQ ID NO: 6006), KP
- KCFL (SEQ ID NO: 6023), VPQN (SEQ ID NO: 6024), FWSN (SEQ ID NO: 6025), KFKN (SEQ ID NO: 6026), IKQN (SEQ ID NO: 6027), KAPR (SEQ ID NO: 6028), FRYN (SEQ ID NO: 6029), KMIC (SEQ ID NO: 6030), RWQN (SEQ ID NO: 6031), PTQN (SEQ ID NO: 6032), KWKS (SEQ ID NO: 6033).
- YMRN (SEQ ID NO: 6034), KAAR (SEQ ID NO: 6035), LCQN (SEQ ID NO: 6036), CCQN (SEQ ID NO: 6037), CVQN (SEQ ID NO: 6038), KLTR (SEQ ID NO: 6039), KLCT (SEQ ID NO: 6040), KIRG (SEQ ID NO: 6041), YLVN (SEQ ID NO: 6042), AFQN (SEQ ID NO: 6043), KACQ (SEQ ID NO: 6044), KWGL (SEQ ID NO: 6045), KIER (SEQ ID NO: 6046), FQ1N (SEQ ID NO: 6047), KACI (SEQ ID NO: 6048), KALR (SEQ ID NO: 6049), KARA (SEQ ID NO: 6050), LCLN (SEQ ID NO: 6051), KAFV (SEQ ID NO: 6052), PRQN (SEQ ID NO: 6053), CPQN (SEQ ID NO:
- SKAQA SEQ ID NO: 6102
- SKAQN SEQ ID NO: 6103
- SMYMN SEQ ID NO: 6104
- SKAFY SEQ ID NO: 6105
- SKLKR SEQ ID NO: 6106
- SKRHR SEQ ID NO: 6107
- SKAQK SEQ ID NO: 6108
- SKWRL SEQ ID NO: 6109
- SRCRN SEQ ID NO: 61 10
- SFTCN SEQ ID NO: 6111.
- SKFFI SEQ ID NO: 6112
- SKAQY SEQ ID NO: 6113
- IVWQN SEQ ID NO: 6114
- SKYFM SEQ ID NO: 6115
- SKARQ SEQ ID NO: 6116
- SCHQN SEQ ID NO: 6117
- FPWQN SEQ ID NO: 6118
- SKIRR SEQ ID NO: 6119
- VYYQN SEQ ID NO: 6120
- SLYWN SEQ ID NO: 6121
- SKPKR SEQ ID NO: 6122
- SKAFS SEQ ID NO: 6123
- SRFWN SEQ ID NO: 6124
- SKAQC SEQ ID NO: 6125
- SRMRN SEQ ID NO: 6126
- SVKKN SEQ ID NO: 6127
- SWAPN SEQ ID NO: 6128
- SLWKN (SEQ ID NO: 6129), SKARW (SEQ ID NO: 6130), SFRPN (SEQ ID NO: 6131), SKKVF (SEQ ID NO: 6132), SYAFN (SEQ ID NO: 6133), SKACS (SEQ ID NO: 6134), SKRLW (SEQ ID NO: 6135), SCSRN (SEQ ID NO: 6136), SRCPN (SEQ ID NO: 6137), SGACN (SEQ ID NO: 6138), SKFRQ (SEQ ID NO: 6139), SFPFN (SEQ ID NO: 6140), SFFGN (SEQ ID NO: 6141), SMCQN (SEQ ID NO: 6142), SKLFW (SEQ ID NO: 6143), SKTRK (SEQ ID NO: 6144), SGKRN (SEQ ID NO: 6145), FYRQN (SEQ ID NO: 6146), CRVQN (SEQ ID NO: 6147), YGIQN (SEQ ID NO: 6148
- SKCFL SEQ ID NO: 6168
- WVPQN SEQ ID NO: 6169
- SFWSN SEQ ID NO: 6170
- SKFKN SEQ ID NO: 6171
- RIKQN SEQ ID NO: 6172
- SKAPR SEQ ID NO: 6173
- SFRYN SEQ ID NO: 6174
- SKMIC SEQ ID NO: 6175
- LRWQN SEQ ID NO: 6176
- LPTQN SEQ ID NO: 6177
- SKWKS SEQ ID NO: 6178
- SYMRN SEQ ID NO: 6179
- SKAAR SEQ ID NO: 6180
- LLCQN SEQ ID NO: 6181
- RCCQN SEQ ID NO: 6182
- LCVQN SEQ ID NO: 6183
- SKLTR SEQ ID NO: 6184
- SKLCT SEQ ID NO: 6185
- SKIRG SEQ ID NO: 6186
- SYLVN SEQ
- MAFQN (SEQ ID NO: 6189), SKACQ (SEQ ID NO: 6190).
- SKWGL (SEQ ID NO: 6191), SKIER (SEQ ID NO: 6192), SFQIN (SEQ ID NO: 6193), SKACI (SEQ ID NO: 6194), SKALR (SEQ ID NO: 6195), SKAHA (SEQ ID NO: 6196), SLCLN (SEQ ID NO: 6197), SKAFV (SEQ ID NO: 6198), RPWQN (SEQ ID NO: 6199), RPRQN (SEQ ID NO: 6200), SCPQN (SEQ ID NO: 6201), SKAQF (SEQ ID NO: 6202), SVRYN (SEQ ID NO: 6203), SVRCN (SEQ ID NO: 6204), SKMPC (SEQ ID NO: 6205), SKKTS (SEQ ID NO: 6206), SLPYN (SEQ ID NO: 6207), VYHQN (SEQ ID NO
- SYTRN (SEQ ID NO: 6210), LVYQN (SEQ ID NO: 6211), YRYQN (SEQ ID NO: 6212), SWLKN (SEQ ID NO: 6213), SKAQM (SEQ ID NO: 6214), CPAQN (SEQ ID NO: 6215), SMCTN (SEQ ID NO: 6216), SPPDN (SEQ ID NO: 6217), SKRNY (SEQ ID NO: 6218), RWTQN (SEQ ID NO: 6219), SKACR (SEQ ID NO: 6220), PRKQN (SEQ ID NO: 6221), SKCSV (SEQ ID NO: 6222), SKARI (SEQ ID NO: 6223).
- PFVQN (SEQ ID NO: 6224), SKLPK (SEQ ID NO: 6225), SKSEQ (SEQ ID NO: 6226), WVAQN (SEQ ID NO: 6227), SLYQN (SEQ ID NO: 6228), SKVRM (SEQ ID NO: 6229), CALQN (SEQ ID NO: 6230).
- SSCTN SEQ ID NO: 6231
- SKNSR SEQ ID NO: 6232
- SKRKR SEQ ID NO: 6233
- LCTQN SEQ ID NO: 6234
- STCLN SEQ ID NO: 6235
- SYARN SEQ ID NO: 6236
- SKQRP SEQ ID NO: 6237
- SKRVV SEQ ID NO: 6238
- KSGQN SEQ ID NO: 6239
- SYYSN SEQ ID NO: 6240
- SLTCN SEQ ID NO: 6241
- SCQSN SEQ ID NO: 6242
- SKAKG SEQ ID NO: 6243
- SKPQN SEQ ID NO: 6244
- FPFQN SEQ ID NO: 6245
- SKCTS SEQ ID NO: 6246
- SVFEN SEQ ID NO: 6247
- SKAKK SEQ ID NO: 6248
- GRYQN SEQ ID NO: 6249
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- GSGSPHSKAQA (SEQ ID NO: 6250), GSGSPHSKAQN (SEQ ID NO: 6251), GSGSPHSMYMN (SEQ ID NO: 6252), GSGSPHSKAFY (SEQ ID NO: 6253), GSGSPHSKLKR (SEQ ID NO: 6254), GSGSPHSKRHR (SEQ ID NO: 6255), GSGSPHSKAQK (SEQ ID NO: 6256), GSGSPHSKWRL (SEQ ID NO: 6257), GSGSPHSRCRN (SEQ ID NO: 6258).
- GSGSPHSFTCN (SEQ ID NO: 6259), GSGSPHSKFFI (SEQ ID NO: 6260), GSGSPHSKAQY (SEQ ID NO: 6261), GSGSPHTVWQN (SEQ ID NO: 6262), GSGSPHSKYFM (SEQ ID NO: 6263), GSGSPHSKARQ (SEQ ID NO: 6264), GSGSPHSCHQN (SEQ ID NO: 6265), GSGSPHFPWQN (SEQ ID NO: 6266), GSGSPHSKIRR (SEQ ID NO: 6267), GSGSPHVYYQN (SEQ ID NO: 6268), GSGSPHSLYWN (SEQ ID NO: 62.69), GSGSPHSKPKR (SEQ ID NO: 6270), GSGSPHSKAFS (SEQ ID NO: 6271), GSGSPHSRFWN (SEQ ID NO: 6272), GSGSPHSK AQC (SEQ ID NO: 6273), GSG
- GSGSPHSKAAR (SEQ ID NO: 6328), GSGSPHELCQN (SEQ ID NO: 6329), GSGSPHRCCQN (SEQ ID NO: 6330), GSGSPHLCVQN (SEQ ID NO: 6331), GSGSPHSKLTR (SEQ ID NO: 6332), GSGSPHSKLCT (SEQ ID NO: 6333), GSGSPHSKIRG (SEQ ID NO: 6334), GSGSPHSYLVN (SEQ ID NO: 6335), GSGSPHQGCQN (SEQ ID NO: 6336), GSGSPHMAFQN (SEQ ID NO: 6337), GSGSPHSKACQ (SEQ ID NO: 6338), GSGSPHSKWGL (SEQ ID NO: 6339), GSGSPHSKILR (SEQ ID NO: 6340), GSGSPHSFQLN (SEQ ID NO: 6341), GSGSPHSKACI (SEQ ID NO: 6342), GSGSPHSKALR (
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof;
- the AAV particle of any one of embodiments 300-306, w herein the AAV capsid variant comprises one, two, or all of an amino acid other than Q at position 458 (e.g., R, C, S, W, L, F, Y, H, I, V, A, or P), an amino acid other than Q at position 459 (e.g., K, I, R, L or S), and/or an amino acid other than T at position 460 (e.g., R), numbered according to SEQ ID NO: 138.
- an amino acid other than Q at position 458 e.g., R, C, S, W, L, F, Y, H, I, V, A, or P
- an amino acid other than Q at position 459 e.g., K, I, R, L or S
- T at position 460 numbered according to SEQ ID NO: 138.
- AAV particle of any one of embodiments 300-307, wherein the AAV capsid variant comprises:
- an amino acid other than T at position 450 e.g., Y, P, W, R, K, S, or F
- an amino acid other than I at position 451 e.g., R, S, Y, I.,, V, H, P, A, or F
- N e.g., V, W, A, T, F, Y, L, R, H
- AAV particle of any one of embodiments 300-31 1, wherein the AAV capsid variant comprises:
- AAV particle of any one of embodiments 300-315, wherein the .AAV capsid variant comprises:
- amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, or 17 amino acids, e.g., consecutive amino acids, thereof;
- amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
- AAV particle of any one of embodiments 300-320, w-herein [A] is present immediately subsequent to position 452, and wherein [A] replaces positions 453-455 (e.g., G453, S454, G455), numbered according to SEQ ID NO: 138 or 981.
- [A]-[B] replaces positions 453-457 (e.g,, G453, S454, G455, Q456, N457), numbered according to SEQ ID NO: 138.
- amino acid residues e.g., acidic and/or basic amino acid residues
- the AA V particle of embodiment 327 or 328, wherein the AAV capsid variant comprises less than tour, less than three, less than two (e.g., two or one) charged amino acid residues (e.g., acidic and/or basic amino acid residues) relative to SEQ ID NO: 138.
- the AA V capsid variant comprises one charged amino acid residues (e.g., an acidic or basic amino acid residue) relative to SEQ ID NO: 138, optionally at any one of positions 450-455 numbered relative to SEQ ID NO: 138.
- an acidic amino acid e.g., D or E
- a negatively charged amino acid e.g., D or E
- E or D an acidic amino acid
- a basic amino acid e.g., R, K, or H
- E charged amino acid residue
- R or K charged amino acid residue
- AAV particle of any one of embodiments 327-347, wherein the .AAV capsid variant comprises E at position 451 and R at position 452, numbered according to SEQ ID NO: 138 or SEQ ID NO: 982.
- amino acid sequence of SPH is present at positions 456-458 numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
- the AAV particle of embodiment 350 wherein the amino acid sequence of SPH is present at positions 456-458 numbered according to any one of SEQ ID NOs: 36-59, 981, or 982. 352.
- one charged amino acid residues e.g., a basic amino acid residue
- a basic amino acid e.g., R or K
- residue e.g., K or R
- SPH sequence e.g., at position 459 numbered according to SEQ ID NO: 981.
- a. charged amino acid residue e.g., R or K
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
- amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, or at least 17 consecutive amino acids from any one of the sequences provided in Tables 1, 2A, 2B, or 15-21; or
- an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to any one of the sequences provided in Tables 1, 2A, 2B. or 15-21; or
- amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of the sequences provided in Tables 1, 2A, 2B, or 15-21.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid, variant comprises:
- amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 945-980 or 985-986;
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
- an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, or at least 13 consecutive amino acids from any one of SEQ ID NOs: 2, 200, 2.01, 941 , 943, 204, 208, 404, or 903-909;
- an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941. 943, 204, 208, 404, or 903-909: or
- an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (c.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
- amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16 or at least 17 consecutive amino acids from any one of SEQ ID NOs: 3849- 4051 or 4681-4693;
- amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 3849-4051 or 4681-4693; or
- an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 3849-4051 or 4681 -4693. 373.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (AT.XN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
- amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16 or at least 17 consecutive amino acids from any one of SEQ ID NOs: 4052- 4092;
- amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 4052-4092; or
- amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid, sequence of any one of SEQ ID NOs: 4052-4092.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRMA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
- an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16 or at least 17 consecutive amino acids from any one of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097;
- an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097; or
- an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097. 375.
- the AAV particle of embodiment 369 or 371, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, or at least 13 consecutive amino acids from any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909.
- AAV particle of any one of embodiments 369-371 or 376, wherein the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700).
- SPHSK SEQ ID NO: 4701.
- AAV particle of any one of embodiments 369-371 or 380, wherein the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO; 4702).
- HDSPH SEQ ID NO: 4703
- HDSPHK SEQ ID NO: 2.
- the at least 4 consecutive amino acids comprise SPHK (SEQ ID NO: 6398);
- the at least 5 consecutive amino acids comprise SPHKY (SEQ ID NO; 4715); and/or (iv) the at least 6 consecutive amino acids comprise SPHKYG (SEQ ID NO: 966).
- AA V particle of embodiment 369 or 371, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909.
- AAV particle of embodiment 370, wherein the AAV capsid variant comprises:
- amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications, relative to the amino acid, sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589);
- AAV particle of embodiment 369 or 371, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941 , 943, 204, 208, 404, or 903-909.
- AAV particle of embodiment 369, wherein the AAV capsid variant comprises:
- amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589);
- amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of KTINGHDSPHSKAQNLQT (SEQ ID NO: 4100); or
- amino acid sequence comprising at least one, at least two, or at least three but. no more than four different amino acids relative to the amino acid sequence of KTVNGHDSPHSKAQNQQT (SEQ ID NO: 4062).
- .AAV capsid variant comprises the amino acid sequence of
- ERVSGSPHSKA (SEQ ID NO: 6399), optionally wherein the amino acid sequence is present immediately subsequent to position 450 and replaces positions 451-455 (e.g., 1451, N542, G453, S454, G455), numbered according to SEQ ID NO: 138.
- K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460 numbered according to SEQ ID NO: 138.
- KAEIGHDSPHKSGQNQQT SEQ ID NO: 1754
- amino acid sequence is present immediately subsequent to position 448 and replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460), numbered according to SEQ ID
- EKMSGSPHSKA SEQ ID NO: 6401
- positions 451-455 e.g., 1451 , N452, G453, S454, G455
- K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460 numbered according to SEQ ID NO: 138.
- SEQ ID NO: 4100 amino acid sequence of KTTNGHDSPHSKAQNLQT
- K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460 numbered according to SEQ ID NO: 138.
- nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NO: 942; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 942; or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 942.
- nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NO: 3; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 3; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 3; a nucleotide sequence comprising at least one, at least two, at least three,
- AAV particle of any one of embodiments 415-419, wherein the AAV capsid variant comprises:
- the AA V particle of embodiment 415 or 416, wherein the AAV capsid variant further comprises an amino acid other than I at position 451, an amino acid other than N at position 452, and/or G at position 453, numbered according to SEQ ID NO: 39 or 138.
- AAV particle of embodiment 415 or 416, wherein the .AAV capsid variant further comprises an amino acid other than S at position 454, an amino acid other than G at position 455, and/or Q at position 458, numbered according to SEQ ID NO: 138.
- the AA V particle of embodiment 415 or 416, wherein the AAV capsid variant further comprises an amino acid other than I at position 451, an amino acid other than S at position 454, and/or an amino acid other than G at position 455, numbered according to SEQ ID NO: 52 or 138.
- AAV particle of any one of embodiments 415, 416, or 429-431, wherein the .AAV capsid variant comprises:
- the AAV particle of any one of embodiments 369-435, wherein the AAV capsid variant composes:
- AAV particle of any one of embodiments 369-436, wherein the AAV capsid variant comprises the amino acids HD at position 454 and 455, and further comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), which is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
- AAV particle of any one of embodiments 433-435 or 438, wherein the AAV capsid variant further comprises A at position 450, E at position 451, and I at position 452, numbered according to SEQ ID NO: 138 or 982.
- AAV particle of any one of embodiments 433, 434, or 438-440, wherein the AAV capsid variant comprises:
- An adeno-associated virus (AAV) particle comprising an .AAV capsid variant comprising the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to the amino acid sequence of SEQ ID NO: 982, wherein the AAV particle further comprises viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein.
- AAV adeno-associated virus
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a. viral genome comprising a nucleic acid encoding a modulatory polynucleotide tor reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to the amino acid sequence of SEQ ID NO: 981 .
- AAV capsid variant e.g., an AAV9 capsid variant
- viral genome comprising a nucleic acid encoding a modulatory polynucleotide tor reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein
- the AAV capsid variant comprises the amino acid sequence of SP
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant comprising the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to the amino acid sequence of SEQ ID NO: 37, and optionally further comprising:
- AAV particle further comprises a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein. 446.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant comprising the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to the amino acid sequence of any one of SEQ ID NO: 36, 38-55, 57, or 59, wherein the AAV particle further comprises a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein.
- AAV adeno-associated virus
- substitutions e.g., conservative substitutions
- substitutions e.g., conservative substitutions
- the AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid, sequence with at least 98% identity to SEQ ID NO: 138. 452.
- the AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid sequence encoded by a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at. least. 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 137.
- nucleotide sequence encoding the capsid vanant comprises at least 80% (e.g., at least 80%, at. least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 137.
- a VP2 of SEQ ID NO: 981, 982, 36, or 4
- at least 80% e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity thereto.
- the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701); or (iv) the at least 6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941); wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 981; (b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 981; (c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 981 ; or (d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences in (a)-(c).
- the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein:
- the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700);
- the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701); or
- the at. least 6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941); wherein the AAV capsid variant comprises an amino acid sequence has at least 90%
- a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 981;
- the at least 3 consecutive amino acids comprise HDS;
- the at least 4 consecutive amino acids comprise IIDSP (SEQ ID NO: 4702);
- the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703); or
- the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO; 982; (b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982; (c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982; or (d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences in (a)-(c).
- the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO: 4702);
- the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703); or
- the at. least 6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2); wherein the AAV capsid variant comprises an amino acid sequence at: least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 982.
- a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982
- a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982;
- a VP3 protein comprising the amino acid, sequence of positions 203-742. of SEQ ID NO: 982; or
- amino acid sequence with at least 90% e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to any of the amino acid sequences m (a)-(c).
- AAV particle of any one of embodiments 1-468, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981 or 982, or an amino acid sequence with at least 80% (e.g., at least 80%, at least. 85%, at. least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity- thereto.
- SEQ ID NO: 981 amino acid sequence with at least 80% (
- substitutions e.
- the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983 or 984, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%), at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- the AAV particle of any one of embodiments 1-478, wherein the nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NOs: 983 or 984, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- 480 e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity thereto.
- nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity there
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a. viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981. 485.
- AAV capsid variant e.g., an AAV9 capsid variant
- viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981. 485.
- the AAV particle of embodiment 483 or 484, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence at least 90%, at least 95%, or at least 99% identical thereto.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982.
- AAV capsid variant e.g., an AAV9 capsid variant
- AAV capsid variant e.g., an AAV9 capsid variant
- viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982.
- the AA V particle of embodiment 486 or 487, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence at least 90%, at least 95%, or at least 99% identical thereto.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NOs: 983 or 984, or a nucleotide sequence at least 95% identical thereto.
- AAV capsid variant e.g., an AAV9 capsid variant
- a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein
- AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid, variant comprises the amino acid sequence of any one of SEQ ID NOs: 4 or 36-59, optionally wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 4 or 36. 491.
- AAV capsid variant e.g., an AAV9 capsid variant
- a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein
- AAV capsid, variant comprises the amino acid sequence of any one of
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of any one of SEQ ID NOs: 12-35, or a nucleotide sequence at least 95% identical thereto.
- AAV capsid variant e.g., an AAV9 capsid variant
- a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein
- AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence
- the AAV particle of 490 or 491, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of any one of SEQ ID NOs: 12-35, or a nucleotide sequence at least 95% identical thereto.
- the AAV particle of any one of embodiments 1-299, 369-371, or 375-492 which has an increased tropism for a CNS cell or ti ssue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising a capsid comprising the amino acid sequence of SEQ ID NO: 138.
- an assay e.g., an immunohistochemistry assay or a qPCR assay, e.g., as described in Example 2.
- an assay e.g., an immunolnstochemistiy assay or a qPCR assay, e.g., as described in Example 2.
- a non-human primate and rodent e.g., mouse
- a non-human primate and rodent e.g., mouse
- AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1 or 5.
- AA V particle of embodiment. 498 or 499 wherein the at least two to at least three species are Macaca fasciculans, Chlorocebus sabaeus, Callithrix jacchus, and/or mouse (e.g., BALB/c mice, C57B1/6 mice, and/or CD-I outbred mice).
- an assay e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2 or 8).
- an assay e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2 or 8).
- a midbrain region e.g., the hippocampus or thalamus
- DRG dorsal root ganglia
- DDG dorsal root ganglia.
- the AAV particle of any one of the preceding embodiments which is capable of transducing non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes).
- non-neuronal cells e.g., glial cells (e.g., oligodendrocytes or astrocytes).
- non-neuronal ceils comprise glial cells, oligodendrocytes (e.g., Olig2 positive oligodendrocytes), or astrocytes (e.g., Olig2 positive astrocytes).
- oligodendrocytes e.g., Olig2 positive oligodendrocytes
- astrocytes e.g., Olig2 positive astrocytes
- the .AAV particle of any one of the preceding embodiments which is capable of transducing Olig2 positive cells, e.g., Olig2 positive astrocytes or Olig2 positive oligodendrocyte s .
- the AAV particle of embodiment 516 or 517, wherein the muscle cell or tissue is a heart muscle (e.g., a heart ventricle or a heart atrium, or both), a quadriceps muscle, or both.
- a heart muscle e.g., a heart ventricle or a heart atrium, or both
- a quadriceps muscle e.g., a quadriceps muscle, or both.
- RNAi agent optionally a dsRNA, siRNA, shRNA, pre-miRN A, pri-miRNA, miRNA, stRNA, IncRNA, piRNA, or snoRNA, or a combination thereof.
- the AAV particle of embodiment 577, wdierein the modulatory polynucleotide comprises an siRNA.
- Embodiments 585 is intentionally absent.
- 586 The AAV particle of any one of the preceding embodiments, wherein the viral genome comprises a promoter operably linked to the nucleic acid sequence encoding the modulators' polynucleotide.
- EFla human elongation factor la-subunit
- CMV cytomegalovirus
- CBA chicken p-actin
- CAG promoter a CAG derivative promoter
- CMV immediate-early enhancer and/or promoter a CMV promoter, a ⁇ glucuronidase (GUSB) promoter, a ubiquitin C (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF- ⁇ ) promoter, an intercellular adhesion molecule 2 (ICAM-2) promoter, a.
- GUSB ⁇ glucuronidase
- UBC ubiquitin C
- NSE neuron-specific enolase
- PDGF platelet-derived growth factor
- PDGF- ⁇ platelet-derived growth factor B-chain
- IAM-2 intercellular adhesion molecule 2
- synapsin (Syn) promoter a methyl- CpG binding protein 2 (MeCP2) promoter, a Ca2+/calmodulin-dependent protein kinase II (CaMKII) promoter, a metabotropic glutamate receptor 2 (mGluR2) promoter, a neurofilament light (NFL) or heavy (NFH) promoter, a P-globin minigene n ⁇ 2 promoter, a.
- MeCP2 methyl- CpG binding protein 2
- CaMKII Ca2+/calmodulin-dependent protein kinase II
- mGluR2 metabotropic glutamate receptor 2
- NFL neurofilament light
- NFH neurofilament light
- P-globin minigene n ⁇ 2 promoter a.
- preproenkephalin (PPE) promoter preproenkephalin (PPE) promoter, an enkephalin (Enk) and excitatory.' amino acid transporter 2 (EAAT2) promoter , a glial fibrillary acidic protein (GFAP) promoter, a myelin basic protein (MBP) promoter, a cardiovascular promoter (e.g., aMHC, cTnT, and CMV-MLC2k), a liver promoter (e.g., hAAT, TBG), a skeletal muscle promoter (e.g., desmin, MCK, C512) or a fragment, e.g., a truncation, or a functional variant thereof.
- PPE preproenkephalin
- Enk enkephalin
- EAAT2 amino acid transporter 2
- GFAP glial fibrillary acidic protein
- MBP myelin basic protein
- a cardiovascular promoter e.g.,
- ITR inverted terminal repeat
- a Rep protein e.g., a non-structural protein
- the Rep protein comprises a Rep78 protein, a Rep68 protein, a Rep52 protein, and/or a. Rep40 protein (e.g,, a. R.ep78 and a. Rep52 protein).
- a Rep protein e.g., a non-stmctural protein
- the Rep protein comprises a Rep78 protein, a. Rep68 protein, a Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 and a Rep52 protein).
- the AAV particle of any one of embodiments 586-618, wherein the viral genome further comprises a. nucleic acid sequence encoding the AAV capsid variant of the AAV particle of any one of embodiments 1-519, 566, or 574.
- a cell e.g., a host cell, comprising the AAV particle of any one of the preceding embodiments.
- cell of embodiment 624 or 625 wherein the cell is a neuron, a sensory neuron, a motor neuron, an astrocyte, a glial cell, or an oligodendrocyte.
- a method of making an AAV particle comprising
- a pharmaceutical composition comprising the AAV particle of any one of embodiments 1-620, and a pharmaceutically acceptable excipient. 632.
- a method of delivering a payload to a. cell or tissue comprising administering an effective amount of the pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Genetics & Genomics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Biophysics (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Molecular Biology (AREA)
- Wood Science & Technology (AREA)
- General Health & Medical Sciences (AREA)
- General Engineering & Computer Science (AREA)
- Virology (AREA)
- Biomedical Technology (AREA)
- Biotechnology (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Gastroenterology & Hepatology (AREA)
- Microbiology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Medicinal Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The disclosure relates to compositions and methods for modulating, e.g., reducing or eliminating, the expression of ATXN2 via delivery of a modulatory polynucleotide using an adeno-associated viral (AAV) capsid variant. The compositions and methods of the present disclosure are useful in the treatment of subjects diagnosed with, or suspected of having, spinocerebellar ataxia type 2 or another ATXN2-related disorder.
Description
COMPOSITIONS AND METHODS FOR THE TREATMENT OF DISORDERS
RELATED TO ATAXIN-2
RELATED APPLICATIONS
[81] This application claims the benefit of and priority to US Provisional Application Serial No. 63/463,841, filed May 3, 2.023, US Provisional Application Serial No. 63/593,857, filed October 27, 2023, and US Provisional Application Serial No. 63/564,435, filed March 12, 2024, the contents of each of which are incorporated herein by reference in their entirety .
SEQUENCE LISTING
[02] The present application is being filed along with a Sequence Listing in electronic format. The Sequence Listing file, entitled “14640 0083-00304 SL.xnil”, was created on March 21, 2024, and. is 5,499,942 bytes in size. The information in electronic format of the Sequence Listing is incorporated herein by reference in its entirety.
FIELD
[03] Described herein are compositions and methods relating to adeno-associated virus (AAV) viral particles for the delivery- of polynucleotides, e.g., modulatory polynucleotides for reducing or eliminating expression of ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2. protein, and their use in the treatment of spinocerebellar ataxia type 2 (SCA2) and other disorders associated with aberrant ATXN2 protein expression or activity or mutated ATXN2 protein. In some embodiments, compositions described herein may be used to treat a subject in need thereof, such as a human subject diagnosed with SCA2 or another condition resulting from mutant ATXN2 protein or aberrant ATXN2 protein expression or activity.
BACKGROUND
[04] Ataxin-2 is a protein encoded by the ATXN2 gene (Ensembl Gene ID No. EN SG00000204842), which is also known as ATX2 and TNR.C 13. It is located on chromosome 12 of the human genome.
[05] The function of Ataxin-2 is not known, but it is found in the cytoplasm, and is thought to interact with the endoplasmic reticulum.
[06] Mutations to ATXN2. cause disease in human subjects. ATXN2 mutations are known to cause spinocerebellar ataxia type 2 (SCA2). SCA2 is an autosomal dominant
cerebellar ataxia. ATXN2 mutations also play a role in other ATXN2-related disorders, including amyotrophic lateral sclerosis (ALS), Parkinson’s disease (PD), and frontotemporal lobar degeneration (FTLD).
[07] SCA2 is typically caused by CAG trinucleotide repeat expansions in the ATXN2 gene on I2q24.1. Patients with SCA2 have ATXN2 alleles with 32 or more trinucleotide repeats, resulting in ATXN2 protein with polyQ expansions. PolyQ expansions in ATXN2 (e.g., intermediate length polyQ expansions) have been associated with other diseases, like ALS, as well.
[08] The age of onset of SCA2 is thought to correlate with the number of CAG trinucleotide repeats. Onset is typically later than age 60 in subjects with 32-34 CAG repeats (SEQ ID NO: 6432). Onset is Apically between ages 20 and. 60 in subjects with 35-40 CAG repeats (SEQ ID NO: 6433). Onset is typically before age 20 for patients with 45 or more CAG repeats (SEQ ID NO: 6434).
[09] Mutated ATXN2 is thought to produce an Ataxin-2. protein with an abnormally long stretch of glutamine residues. The cellular impact of mutant Ataxin-2 protein is not confirmed. Without wishing to be bound by any particular theory, mutant Ataxin-2 may accumulate in cells and cause cell death.
[010] SCA2 is characterized by progressive cerebellar ataxia, including nystagmus, and slow saccadic eye movements. It may also present with ophthalmoparesis and/or parkinsonism. Later stages of SCA2 mainly involve the brainstem and cerebellum.
[Oil] There are no specific treatments for SCA2. Trials have considered riluzole, which inhibits glutamic acid release, for treating SCA2. However, there are no treatments targeting the cause of SCA2. Thus, there remains a long -felt need to develop pharmaceutical compositions and methods for the treatment of SCA2. In particular, a need exists for treatments of SCA2 targeting the brainstem and cerebellum.
[012] There remains a need for effective methods of treatment using AAV capsid variants that are capable of delivering a modulatory polynucleotide for reducing or eliminating expression of human ATXN2 mRNA (thereby reducing or eliminating ATXN2 protein), to a target cell or tissue, e.g., a CNS cell or tissue.
SUMMARY
[013] The present disclosure addresses these challenges by providing AAV -based compositions comprising AAV capsid variants and methods for treating SCA2 -related
disorder in subjects. Disclosed herein are compositions and methods directed to AAV-based gene delivery of modulatory polynucleotides for reducing or eliminating expression of ATXN2 to treat SCA2. In some embodiments, the compositions and methods reduce or eliminate expression of ATXN2 mRNA, and thereby reduce or eliminate ATXN2 protein, to treat SCA2. The compositions and methods are useful to reduce the effects of mutant ATXN2 or aberrant ATXN2 protein expression or activity, and to slow, halt or reverse muscular and other symptoms of SCA2. In some embodiments, aberrant ATXN2 expression refers to expression of ATXN2 comprising 32 or more CAG trinucleotide repeats (SEQ ID NO: 6435) (and thus polyQ expansions). Unless otherwise specified, ATXN2, ATX2, and TNRC13 are synonymous terms and are used interchangeably to refer to ATXN2. ATXN2 refers to the transcript encoding the ATXN2 (Ataxin 2) protein, and ATXN2 protein refers to the protein encoded by the ATXN2 gene and mRNA. SCA2 is used to refer to spinocerebellar ataxia type 2.
[014] In some embodiments, the present disclosure provides an A AV particle comprising an AAV capsid and a nucleotide sequence encoding a modulatory polynucleotide tor reducing or eliminating expression of ATXN2. In some embodiments, the modulatory polynucleotide reduces or eliminates mRNA encoding ATXN2. In some embodiments, the modulatory polynucleotide reduces or eliminates ATXN2 protein (e.g., a human ATXN2 protein). In some embodiments, the AAV capsid is an AAV capsid variant. In some embodiments, the AAV capsid variant is an AAV9 capsid variant.
[015] In some embodiments, the AAV capsid variant is an AAV9 capsid variant comprising a peptide insert in the loop IV region. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SPH in loop IV. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SPH in loop IV wherein the amino acid sequence (SPH) is present immediately subsequent to position 455 as numbered according to SEQ ID NO: 138.
[016] In some embodiments, the AAV particles described herein surprisingly provide high expression of the modulatory polynucleotide in the brain (e.g., the cortex, striatum, and brainstem), high modulatory polynucleotide activity in the brain, reduced immunogenicity, and/or reduced toxicity to other tissues of the body. In some embodiments, the AAV particles described herein surprisingly provide reduced modulatory polynucleotide expression in the liver while retaining high modulatory polynucleotide activity in other areas of the brain (e.g., the brain stem), e.g., relative to AAV9. In some embodiments, the AAV particles described
herein can be administered to a. subject having a ATXN2-related disorder such as SCA2, ALS, PD, or FTLD.
[017] In some embodiments, administration of an A AV particle to a. subject results m greater reduction in the expression and/or activity of ATXN2 in CNS cells or tissues of the subject as compared to administration of an AAV9 particle comprising a wildtype AAV9 capsid and the modulatory polynucleotide sequence.
[018] In some embodiments, the present disclosure provides an adeno-associated virus (AAV) particle comprising: a) an AAV capsid variant comprising an amino acid sequence having the following formula [N1 ]-[N2]-[N3], wherein (i) optionally [N1] comprises X1, X2, and X3, wherein at least one of XL X2, or X3 is G; (ii) [N2] comprises the amino acid sequence of SPH; and (iii) [N3] comprises X4, X5, and X6, wherein at least one of X4, X5, or X6 is a basic amino acid: and b) a viral genome comprising a nucleic acid sequence encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2). In some embodiments, the modulatory polynucleotide comprises an RNAi agent targeting ATXN2 mRNA. In some embodiments, the amino acid sequence [N1]-[N2]-[N3] is in hypervariable loop IV of the AAV capsid variant. In some embodiments, the .AAV capsid variant is an AAV9 capsid variant. In some embodiments, [N1] comprises XL X2, and X3, wherein at least one of X1, X2 or X3 is G.
[019] In some embodiments, [N1]-[N2]-[N3] is presen t immediately subsequent to a position corresponding to the amino acid position 452 of SEQ ID MO: 982 and the AAV capsid variant comprises an amino acid sequence at least 90% identical, e.g., at least 91%, at least 92%, at least 93%, at least 94%), at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of positions 203-742 of SEQ ID NO: 982. In some embodiments, [N1]-[N2]-[N3] is present immediately subsequent to a position corresponding to the amino acid position 452 of SEQ ID NO: 982 and the AAV capsid variant comprises an amino acid sequence at least 90% identical, e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 982.
[020] In some embodiments, [N1] comprises GHD. In some embodiments, [N1] comprises the amino acid G at a position corresponding to position 453, the amino acid H at position 454, and the amino acid D at position 455 of SEQ ID NO: 138 or SEQ ID NO: 982. In some embodiments [N3] comprises KSG.
[021] In some embodiments, the AAV capsid variant comprises: (i) a VP 1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at
least 90% identity to SEQ ID NO; 982; (ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to positions 138-742 SEQ ID NO: 982; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 982.
[822] In some embodiments, the AAV capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to SEQ ID NO; 982; (ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 982; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 982.
[023] In some embodiments, the AAV capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to SEQ ID NO; 982; (ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to positions 138-742 SEQ ID NO: 982; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 982.
[024] In some embodiments, the AAV capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982; or (iii) a. VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO; 982.
[025] In some embodiments, [N2]-[N3] comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941).
[026] In some embodiments, the present disclosure provides an adeno-associated virus (AAV) particle comprising (a) a viral genome comprising a nucleic acid sequence encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2), and (b) an AAV9 capsid variant comprising the amino acid sequence of SPHSKA (SEQ ID NO: 941). In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is in hypervariable loop IV of the AAV capsid variant. In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO: 36 or SEQ ID NO: 4.
[027] In some embodiments, the AAV 9 capsid variant comprises one, two, or all of: an E at an amino acid position corresponding to position 451, an R at an amino acid position corresponding to position 452, and/or a V at an amino acid position corresponding to position 453 of SEQ ID NO: 36. In some embodiments, the AAV9 capsid variant comprises the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589).
[028] In some embodiments, the AAV9 capsid variant comprises: (i) a VP1 protein composing an amino acid sequence having at least 90% identity to SEQ ID NO: 36; (ii) a VP2 protein comprising an amino acid sequence having at least 90% identity to positions 138-742 SEQ ID NO: 36; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 36.
[029] In some embodiments, the AA.V9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 95% identity to SEQ ID NO: 36; (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 36; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 36.
[030] In some embodiments, the AAV9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 36; (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to positions 138-742 of SEQ ID NO: 36; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 36.
[031] In some embodiments, the AAV9 capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 36; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 36; and/or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 36.
[032] In some embodiments, the AAV9 capsid variant comprises: (i) the amino acid sequence SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO 36; (ii) an E at an amino acid position corresponding to position 451 , an R at an amino acid position corresponding to position 452, and a. V at an amino acid position corresponding to position 453 of SEQ ID NO: 36; and (iii) no other modifications relative to wild type AAV 9.
[033] In some embodiments, the AAV9 capsid variant comprises one, two, or all of: an N at an amino acid position corresponding to position 452, an E at an amino acid position corresponding to position 451, and/or a V at an amino acid position corresponding to position
453 of SEQ ID NO: 4. In some embodiments, the AAV9 capsid variant comprises the amino acid sequence of KTENVSGSPHSKAQNQQT (SEQ ID NO: 3272).
[034] In some embodiments, the AA.V9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 90% identity to SEQ ID NO: 4; (ii) a VP2 protein comprising an amino acid sequence having at least 90% identity to positions 138-742 of SEQ ID NO: 4; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 4.
[035] In some embodiments, the AAV9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 95% identity to SEQ ID NO: 4; (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 4; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 4.
[036] In some embodiments, the AAV9 capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 4; (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to positions 138-742 of SEQ ID NO: 4; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 4.
[037] In some embodiments, the AAV9 capsid variant comprises: (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 4; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 4; and/or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 4,
[038] In some embodiments, the AAV9 capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO 4; (ii) an E at an amino acid position corresponding to position 451 and a. V at an amino acid position corresponding to position 453 of SEQ ID NO: 4; and (iii) no other modifications relative to wild type AAV9.
[039] In some embodiments, the modulatory polynucleotide comprises: (a) a 5’ flanking region comprising any one of SEQ ID NOs: 6413-6416 or a nucleotide sequence at least 95% identical thereto; (b) a loop region comprising any one of SEQ ID NOs: 6417-6421 or a nucleotide sequence at least 95% identical thereto; and (c) a 3’ flanking region comprising any one of SEQ ID NOs: 6422.-6427 or a nucleotide sequence at least 95% identical thereto. [040] In some embodiments, (a.) the 5' flanking region comprises SEQ ID NO: 6414 or SEQ ID NO: 6415, or a nucleotide sequence at least 95% identical thereto; (b) the loop
region comprises SEQ ID NO: 6417, SEQ ID NO: 6418, or SEQ ID NO: 6421 , or a nucleotide sequence at least 95% identical thereto; and (c) the 3' flanking region comprises SEQ ID NO: 6423, SEQ ID NO: 6424, or SEQ ID NO: 6425, or a nucleotide sequence at least 95% identical thereto.
[041] In some embodiments, the 5’ flanking region comprises SEQ ID NO; 6414 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6417 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO; 6423 or a. nucleotide sequence at least 95% identical thereto. In some embodiments, the 5’ flanking region comprises SEQ ID NO: 6415 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6421 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO: 6425 or a nucleotide sequence at least 95% identical thereto. In some embodiments, the 5’ flanlang region comprises SEQ ID NO: 6414 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6417 or a nucleotide sequence at least 95% identical thereto, and the 3" flanking region comprises SEQ ID NO: 6424 or a nucleotide sequence at least 95% identical thereto. In some embodiments, the 5’ flanking region comprises SEQ ID NO: 6414 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6418 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO: 6423 or a nucleotide sequence at least 95% identical thereto.
[042] In some embodiments, the modulatory polynucleotide comprises siRNA .
[043] In some embodiments, the modulatory polynucleotide comprises shRNA.
[044] In some embodiments, the modulatory polynucleotide further comprises a passenger strand and a guide strand, wherein the guide strand binds to and reduces or eliminates expression of one or more ATXN2 mRNA transcripts. In some embodiments, the modulatory polynucleotide comprises from 5' to 3‘: the 5' flanking region, the passenger strand, the loop region, tire guide strand, and the 3' flanking region. In some embodiments, the modulatory polynucleotide comprises from 5' to 3': the 5' flanking region, the guide strand, the loop region, the passenger strand; and the 3' flanking region.
[045] In some embodiments, the passenger strand is 15-30 nucleotides in length. In some embodiments, the guide strand is 15-30 nucleotides in length. In some embodiments, the guide strand is 21-25 nucleotides in length. In some embodiments, the passenger strand is 21- 25 nucleotides in length. In some embodiments, the passenger strand is at least 70%, at least 80%, at least 90%, at least 95% or is 100% complementary to the guide strand.
[046] In some embodiments, the one or more ATXN2 mRNA transcripts comprises SEQ ID NO: 6428, SEQ ID NO: 6429, SEQ ID NO: 6430, and/or SEQ ID NO: 6431, or a trinucleotide repeat expansion thereof.
[047] In some embodiments, the viral genome comprises a promoter operably linked to the nucleic acid sequence encoding the modulatory polynucleotide.
[048] In some embodiments, the viral genome further comprises an inverted terminal repeat (ITR) sequence. In some embodiments, the viral genome comprises an ITR sequence positioned 5’ relative to the nucleic acid sequence encoding the modulatory polynucleotide. In some embodiments, the viral genome comprises an ITR sequence positioned 3' relative to the nucleic acid sequence encoding the modulatory polynucleotide . In some embodiments, viral genome comprises an ITR sequence positioned 5’ relative to the nucleic acid sequence encoding the modulatory polynucleotide, and an ITR sequence positioned 3’ relative to the nucleic, acid sequence encoding the modulatory polynucleotide.
[049] In some embodiments, the present disclosure provides a cell comprising an AAV particle described herein. In some embodiments, the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial ceil.
[050] In some embodiments, the present disclosure provides a method of making an AAV particle described herein, the method comprising (i) providing a cell comprising the viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle.
[051] In some embodiments, the AAV capsid variant comprises (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least. 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 982; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least. 90% identity (e.g,, at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 138-742 SEQ ID NO: 982; and/or (in) a VP3 protein comprising tire amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91 %, at least 92%), at least 93%, at least 94%, at least 95%, at least 96%, at least 97%), at least 98%), or at least 99%) identity) to positions 203-742 of SEQ ID NO: 982.
[052] In some embodiments, the AAV capsid variant comprises (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 36; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 138-742 SEQ ID NO: 36; and/or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91 %, at least 92%), at least 93%, at least 94%, at least 95%, at least 96%, at least 97%), at least 98%), or at least 99%) identity) to positions 203-742 of SEQ ID NO: 36.
[853] In some embodiments, the AAV capsid variant comprises (i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%), at least 95%, at least 96%, at least 97%>, at least 98%, or at least 99% identity) to SEQ ID NO: 4; (ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 4 or an amino acid, sequence having at. least 90% identity (e.g., at least. 90%, at. least 91%), at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%o, or at least 99%o identity) to positions 138-742 SEQ ID NO: 4; or (iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%>, at least 97%, at least 98%, or at least 99% identity) to positions 203-742 of SEQ ID NO: 4.
[054] In some embodiments, the method, further comprises, prior to step (i), introducing a first nucleic acid molecule comprising the viral genome into the cell. In some embodiments, the cell comprises a second nucleic acid molecule encoding the AAV capsid variant. In some embodiments, the method further comprises, prior to step (i), introducing the second nucleic acid molecule into the cell. In some embodiments, the cell comprises a mammalian cell, e.g., an HEK293 cell, an insect cell, e.g., an Sf9 cell, or a bacterial cell.
[055] In some embodiments, the present disclosure provides a pharmaceutical composition comprising an AAV particle described herein, and a pharmaceutically acceptable excipient.
[056] In some embodiments, the present disclosure provides a method of delivering a modulatory polynucleotide for reducing or eliminating expression of ATXN2 to a subject, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein, thereby delivering the modulatory polynucleotide. In some embodiments, the method reduces or eliminates ATXN2 protein in the subject. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having an ATXN2-related disorder. In some embodiments, the ATXN2-related disorder is spinocerebellar ataxia type 2 (SCA2).
[057] In some embodiments, the present disclosure provides a method of treating an ATXN2-related disorder in a subject, comprising administering to tire subject an effective amount of a pharmaceutical composition or AAV particle described herein, thereby treating the ATXN2-related disorder. In some embodiments, the subject has, has been diagnosed, or is at risk of having the ATXN2 -related disorder.
[058] In some embodiments, the subject has one or more mutations in the ATXN2 gene. In some embodiments, the one or more mutations in the ATXN2 gene comprises a trinucleotide repeat expansion. In some embodiments, the trinucleotide repeat expansion in the ATXX2 gene comprises 32 or more CAG repeats.
[059] In some embodiments, treating results in prevention of progression of the ATXN2- related disorder in the subject. In some embodiments, the treating results in amelioration of at least one symptom of the ATXN 2 -related disorder and/or a change in at least one biomarker of the AT.XN2 -related disorder in the subject. In some embodiments, the at least one symptom comprises progressive cerebellar ataxia, nystagmus, slow saccadic eye movements, ophthahnoparesis, parkinsonism, or a combination thereof. In some embodiments, the ATXN2-related disorder is spinocerebellar ataxia type 2 (SCA2).
[060] In some embodiments, the present disclosure provides a method of treating spinocerebellar ataxia type 2 (SCA2) in a. subject, comprising administering to the subject an effective amount of the pharmaceutical composition described herein or the AAV particle described herein, thereby treating SCA2. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having SCA2.
[061] In some embodiments, the subject is a human.
[062] In some embodiments, the AAV particle or the pharmaceutical composition is delivered to a cell or tissue of the CNS. In some embodiments, the cell or tissue of the CNS is a cell or tissue of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal
cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region). In some embodiments, the AAV particle or the pharmaceutical composition is delivered via intravenous administration.
[063] In some embodiments, the method of delivering or treating further comprises evaluating, e.g., measuring, the level of modulatory polynucleotide expression, and/or the level of ATXN2 expression, e.g., ATXN2 gene, ATXN2 mRNA, and/or ATXN2 protein expression, in the subject, e.g., in a cell, tissue, or fluid, of the subject. In some embodiments, the level of ATXN2 protein is measured by an ELISA, a Western blot, or an immunohistochemistry assay. In some embodiments, the evaluating of the subject’s level of modulatory polynucleotide expression and/or level of ATXN2 expression is performed prior to and/or subsequent to administration of the pharmaceutical composition or AAV particle, optionally wherein the subject’s level of modulatory polynucleotide expression and/or the subject’s level of ATXN2 expression prior to administration is compared to the subject’s level of modulatory polynucleotide expression and/or the subject’s level of ATXN2 expression subsequent to administration. In some embodiments, the subject’s level of ATXN2 protein expression subsequent to administration is decreased relative to the subject’s level of ATXN2 protein expression prior to administration.
[064] In some embodiments, the method of delivering or treating further comprises evaluating, e.g., measuring, the level of modulatory polynucleotide activity and/or ATXN2 activity in the subject, e.g., in a cell or tissue of the subject. In some embodiments, the administration results in: (i) a decrease in ATXN2 protein expression in a cell, tissue (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region)), and/or fluid (e.g., CSF and/or serum) of the subject relative to baseline and/or relative to ATXN2 protein expression in a. cell, tissue, or fluid, of an individual with an ATXN2-relat.ed disorder who has not been administered the pharmaceutical composition or AAV particle; (ii) an increase in the number and/or level of viral genomes (VG) per cell in a
CNS tissue (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or (iii) a decrease in ATXN2 mRNA expression in a cell or tissue (e.g., a cell or tissue of the CNS, e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region) of the subject relative to baseline and/or relative to ATXN2 mRNA expression in a cell or tissue of an individual with an ATXN2-related disorder who has not been administered the pharmaceutical composition or AAV particle.
[065] In some embodiments, the method of delivering or treating further comprises administering to the subject at least one additional therapeutic agent and/or therapy. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the ATXN2-related disorder. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises one or more of: growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2-related disorders.
[866] In some embodiments, the ATXN2-related disorder is SCA2, amyotrophic lateral sclerosis (ALS), Parkinson’s disease (PD), or frontotemporal lobar degeneration (FTLD). In some embodiments, the ATXN2-related disorder is SCA2.
[067] In some embodiments, the method of delivering or treating further comprises administering an immunosuppressant to the subject. In some embodiments, the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
[068] In some embodiments, the present, disclosure provides a method of treating a subject having or diagnosed with having ALS, PD, or FTLD, comprising administering to the subject an effective amount of a pharmaceutical composition or A AV particle described herein. In some embodiments, the method further comprises administering to the subject at least one additional therapeutic agent and/or therapy. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises one or more of: growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2-related disorders. In some embodiments, the method further comprises administering an immunosuppressant to the subject. In some embodiments, the immunosuppressant, comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, rnycophenolatemofetil, tacrolimus, rituximab, and/or ecuiizumab hydroxychloroquine.
[069] In some embodiments, the present, disclosure provides a pharmaceutical composition or AAV particle described herein for use in a method of treating a disorder described herein. In some embodiments, the ATXN2-related disorder is SCA2. In some embodiments, the subject has, has been diagnosed 'with having, or is at risk of having the ATXN2-relat.ed disorder, optionally wherein the ATXN2-relat.ed disorder is SCA2.
[070] In some embodiments, the present disclosure provides a pharmaceutical composition or AAV particle described herein for use in treating an ATXN2 -related disorder in a subject. In some embodiments, the ATXN2-related disorder is SCA2. In some embodiments, the subject, has, has been diagnosed with having, or is at risk of having the ATXN2-related disorder, optionally wherein the ATXN2 -related disorder is SCA2.
[071] In some embodiments, the present disclosure pro vides a use of a pharmaceutical composition or A AV particle described herein in the manufacture of a medicament for the treatment of an ATXN2-related disorder in a subject. In some embodiments, the ATXN2- related disorder is SCA2. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having the ATXN2-related disorder, optionally wherein the ATXN2- related disorder is SCA2.
[072] Those skilled in the art will recognize or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments of the invention described herein. Such equivalents are intended, to be encompassed by the following enumerated embodiments.
Enumerated Embodiments
1, An adeno-associated virus (AAV) particle comprising an AAV capsid, variant (e.g., an AAV9 capsid variant) and a viral genome comprising a. nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein (e.g., human ATXN2 protein), wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]- [N3], wherein:
(i) optionally [N1] comprises X1, X2, and X3, wherein at least one of X1, X2, or X3 is G;
(ii) [N2] comprises the amino acid sequence of SPH; and
(iii) [N3] comprises X4, X5, and X6, wherein at least one of X4, X5, or X6 is a basic amino acid, e.g., a K or R.
2. The AAV particle of embodiment 1, wherein X4, X5, or both of [N3] is a K.
3. The AAV particle of embodiment 1 or 2, wherein X4, X5, or X6 of [N3] is an R.
4. The AAV particle of any one of embodiments 1-3, wherein:
(a) X 4 of [N3] is: K, S, A, V, T, G, F, W, V, N, or R;
(b) X5 of [N3] is: S, K, T, F, I, L, Y, H, M, or R; and/or
(c) X6 of [N3] is: G, A, R, M, I, N, T, Y, D, P, V, L, E, W, N, Q, K, or S; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c).
5. The AAV particle of any one of embodiments 1-4, wherein [N3] comprises SK, KA, KS, AR, RM, VK, AS, SR, VK, KR, KK, KN, VR, RS, RK, KT, TS, KF, FG, KI, IG, KL, LG, TT, TY, KY, YG, KD), KP, TR, RG, VR, GA, SL, SS, FL,, WK, SA, RA, LR, KW, RR, GK, TK, NK, AK, KV, KG, KH, KM, TG, SE, SV, SW, SN, HG, SQ, LW, MG, MA, or SG.
6. The AAV particle of any one of embodiments 1-5, wherein [N3] is or comprises SKA, KSG, ARM, VKS, ASR, VKI, KKN, VRM, RKA, KTS, KFG, KIG, KLG, KTT, KTY,
KYG, SKD, SKP, TRG, VRG, KRG, GAR, KSA, KSR, SKL, SRA, SKR, SLR, SRG, SSR,
FLR, SKW, SKS, WK A, VRR, SKV, SKT, SKG, GKA, TKA. NKA, SKL, SKN, AKA,
KTG, KSL, KSE, KSV, KSW, KSN, KHG, KSQ, KSK, KLW, WKG, KMG, KMA, or RSG.
7. The AAV particle of any one of embodiments 1-6, wherein [N2]-[N3] comprises SPHSK (SEQ ID NO: 4701), SPHKS (SEQ ID NO: 4704), SPHAR (SEQ ID NO: 4705), SPHVK (SEQ ID NO: 4706), SPHAS (SEQ ID NO: 4707), SPHKK (SEQ ID NO: 4708), SPHVR (SEQ ID NO: 4709), SPHRK (SEQ ID NO: 4710), SPHKT (SEQ ID NO: 4711), SPHKF (SEQ ID NO: 4712), SPHKI (SEQ ID NO: 4713), SPHKL (SEQ ID NO: 4714), SPHKY (SEQ ID NO: 4715), SPHTR (SEQ ID NO: 4716), SPHKR (SEQ ID NO: 4717), SPHGA (SEQ ID NO: 4718), SPHSR (SEQ ID NO: 4719), SPHSL (SEQ ID NO: 4720), SPHSS (SEQ ID NO: 4721), SPHFL (SEQ ID NO: 4722), SPIIWK (SEQ ID NO: 4723), SPHGK (SEQ ID NO: 4724), SPHTK (SEQ ID NO: 4725), SPUNK (SEQ ID NO: 4726), SPHAK (SEQ ID NO: 4727), SPHKH (SEQ ID NO: 4728), SPHKM (SEQ ID NO: 4729), or SPHRS (SEQ ID NO: 4730),
8. The AAV particle of any one of embodiments 1-7, wherein [N2] [N3] is or comprises:
(i) SPHSKA (SEQ ID NO: 941), SPHKSG (SEQ ID NO: 946), SPHARM (SEQ ID NO: 947), SPHVKS (SEQ ID NO: 948), SPHASR (SEQ ID NO: 949), SPHVKI (SEQ ID NO: 950), SPHKKN (SEQ ID NO: 954), SPHVRM (SEQ ID NO: 955), SPHRKA (SEQ ID NO: 956), SPHKFG (SEQ ID NO: 957), SPHKIG (SEQ ID NO: 958), SPHKLG (SEQ ID NO: 959), SPHKTS (SEQ ID NO: 963), SPHKTT (SEQ ID NO: 964), SPHKTY (SEQ ID NO: 965), SPHKYG (SEQ ID NO: 966), SPHSKD (SEQ ID NO: 967), SPHSKP (SEQ ID NO: 968), SPHTRG (SEQ ID NO: 972), SPHVRG (SEQ ID NO: 973), SPHKRG (SEQ ID NO: 974), SPHGAR (SEQ ID NO: 975), SPHKSA (SEQ ID NO: 977), SPHKSR (SEQ ID NO: 951), SPHSKL (SEQ ID NO: 960), SPHSR A (SEQ ID NO: 969), SPHSKR (SEQ ID NO: 978), SPHSLR (SEQ ID NO: 952), SPHSRG (SEQ ID NO: 961), SPHSSR (SEQ ID NO: 970), SPHFLR (SEQ ID NO: 979), SPHSKW (SEQ ID NO: 953), SPHSKS (SEQ ID NO: 962), SPHWKA (SEQ ID NO: 971), SPHVRR (SEQ ID NO: 980), SPHSKT (SEQ ID NO: 4731), SPHSKG (SEQ ID NO: 4732), SPHGKA (SEQ ID NO: 4733), SPHNKA (SEQ ID NO: 4734), SPHSKN (SEQ ID NO: 4735), SPHAKA (SEQ ID NO: 4736), SPHSKV (SEQ ID NO: 4737), SPHKTG (SEQ ID NO: 4738), SPHTKA (SEQ ID NO: 4739), SPHKSL (SEQ ID NO: 4740), SPHKSE (SEQ ID NO: 4741), SPHKSV (SEQ ID NO: 4742), SPHKSW (SEQ ID NO: 4743), SPHKSN (SEQ ID NO: 4744), SPHKHG (SEQ ID NO: 4745), SPHKSQ (SEQ ID NO: 4746), SPHKSK (SEQ ID NO: 4747), SPHKLW (SEQ ID
NO: 4748), SPHWKG (SEQ ID NO: 4749), SPHKMG (SEQ ID NO: 4750), SPHKMA (SEQ ID NO: 4751), or SPHRSG (SEQ ID NO: 976);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
9, The AAV particle of any one of embodiments 1-8, wherein the AAV capsid variant comprises an amino acid other than G at position 453 (e.g., V, R, D, E, M, T, I, S, A, N, L, K, H, P, W, or C), an amino acid other than S at position 454 (e.g., V, L, N, D, H, R, P, G, T, I, A, E, Y, M, or Q), and/or an amino acid other than G at position 455 (e.g., C, L, D, E, Y, H, V, A, N, P, or S), numbered according to any one of SEQ ID NOs: 36-59, 138, 981, or 982.
10. The AAV particle of any one of embodiments 1-8, wherein the AAV capsid variant comprises the amino acid G at position 453, the amino acid S at position 454, and the amino acid G at position 455, numbered according to SEQ ID NO: 138 or 981 .
11. The AAV particle of any one of embodiments 1-9, wherein the AAV capsid variant comprises the amino acid G at position 453, the amino acid H at position 454, and the amino acid D at position 455, numbered according to SEQ ID NO: 138 or 982.
12. The AAV particle of any one of embodiments 1-11, wherein [N1] comprises X1, X2, and X3, wherein at least one of X L X2, or X3 is G.
13. The AAV particle of any one of embodiments 1-12, wherein:
(a) X1 of [N1] is: G, V, R, D, E, M, T, I, S, A, N, I.,, K, H, P, W, or C;
(b) X2 of [N1] is: S, V, L, N, D, H, R, P, G, T, I, A, E, Y, M, or Q; and/or
(c) X3 of [N1] is: G, C, L, D, E, Y, H, V, A, N, P, or S; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g,, a. conservative substitution, of any of the aforesaid amino acids in (a)-(c).
14. The AAV particle of any one of embodiments 1-13, wherein [N1] comprises GS, SG,
GH, HD, GQ, QD, VS, CS, GR, RG, QS, SH, MS, RN, TS, IS, GP, ES, SS, GN, AS, NS, LS, GG, KS, GT, PS, RS, GI, WS, DS, ID, GL, DA, DG, ME, EN, KN, KE, Al, NG, PG, TG, SV, IG, LG, AG, EG, SA, YD, HE, HG, RD, ND, PD, MG, QV, DD, HN, HP, GY, GM, GD, or HS.
15. The AAV particle of any one of embodiments 1-14, wherein [N1] is or comprises GSG, GHD, GQD, VSG, CSG, GRG, CSH, GQS, GSH, RVG, GSC, GLL, GDD, GHE, GNY, MSG, RNG, TSG, ISG, GPG, ESG, SSG, GNG, ASG, NSG, LSG, GGG, KSG, HSG, GTG, PSG, GSV, RSG, GIG, WSG, DSG, IDG, GLG, DAG, DGG, MEG, ENG, GSA, KNG, KEG, AIG, GYD, GHG, GRD, GND, GPD, GMG, GQV, GHN, GHP, or GHS.
16. The AAV particle of any one of embodiments 1-15, wherein [N1]-[N2] comprises:
(i) SGSPH (SEQ ID NO: 4752), HDSPH (SEQ ID NO: 4703), QDSPH (SEQ ID NO: 4753), RGSPH (SEQ ID NO: 4754), SHSPH (SEQ ID NO: 4755), QSSPH (SEQ ID NO: 4756), DDSPH (SEQ ID NO: 4757), HESPH (SEQ ID NO: 4758), NYSPH (SEQ ID NO: 4759), VGSPH (SEQ ID NO: 4760), SCSPH (SEQ ID NO: 4761), LLSPH (SEQ ID NO: 4762), NGSPH (SEQ ID NO: 4763), PGSPH (SEQ ID NO: 4764), GGSPH (SEQ ID NO: 4765), TGSPH (SEQ ID NO: 4766), SVSPH (SEQ ID NO: 4767), IGSPH (SEQ ID NO: 4768). DGSPH (SEQ ID NO: 4769), LGSPH (SEQ ID NO: 4770), AGSPH (SEQ ID NO: 4771), EGSPH (SEQ ID NO: 4772), SASPH (SEQ ID NO: 4773), YDSPH (SEQ ID NO: 4774), HGSPH (SEQ ID NO: 4775), RDSPH (SEQ ID NO: 4776), NDSPH (SEQ ID NO: 4777), PDSPH (SEQ ID NO: 4778), MGSPH (SEQ ID NO: 4779), QVSPH (SEQ ID NO: 4780), HNSPH (SEQ ID NO: 4781), HPSPH (SEQ ID NO: 4782). or HSSPH (SEQ ID NO: 4783);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof:
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
17. The AAV particle of any one of embodiments 1-16, wherein [N1]-[N2] is or comprises:
(i) GSGSPH (SEQ ID NO: 4695), GHDSPII (SEQ ID NO: 4784), GQDSPH (SEQ ID NO: 4785), VSGSPH (SEQ ID NO: 4786), CSGSPH (SEQ ID NO: 4787), GRGSPH (SEQ ID NO: 4788), CSHSPH (SEQ ID NO: 4789), GQSSPH (SEQ ID NO: 4790), GSHSPH (SEQ ID NO: 4791), GDDSPII (SEQ ID NO: 4792), GIIESPII (SEQ ID NO: 4793), GNYSPH (SEQ ID NO: 4794), RVGSPH (SEQ ID NO: 4795), GSCSPH (SEQ ID NO: 4796). GLLSPH (SEQ ID NO: 4797), MSGSPH (SEQ ID NO: 4798), RNGSPH (SEQ ID NO: 4799), TSGSPH (SEQ ID NO: 4800), ISGSPH (SEQ ID NO: 4801), GPGSPH (SEQ ID NO: 4802), ESGSPH (SEQ ID NO: 4803), SSGSPH (SEQ ID NO: 4804), GNGSPII (SEQ ID NO: 4805), ASGSPH (SEQ ID NO: 4806), NSGSPH (SEQ ID NO: 4807), LSGSPH (SEQ ID NO: 4808), GGGSPH (SEQ ID NO: 4809), KSGSPH (SEQ ID NO: 4810), HSGSPH (SEQ ID NO: 4811), GTGSPH (SEQ ID NO: 4812), PSGSPH (SEQ ID NO: 4813), GSVSPH (SEQ ID NO: 4814), RSGSPH (SEQ ID NO: 4815), GIGSPH (SEQ ID NO: 4816). WSGSPH (SEQ ID NO: 4817), DSGSPH (SEQ ID NO: 4818), IDGSPH (SEQ ID NO: 4819), GLGSPH (SEQ ID NO: 4820), DAGSPH (SEQ ID NO: 4821), DGGSPH (SEQ ID NO: 4822), MEGSPH (SEQ ID NO: 4823), ENGSPH (SEQ ID NO: 4824), GSASPH (SEQ ID NO: 4825), KNGSPH (SEQ ID NO: 4826), KEGSPH (SEQ ID NO: 4827), AIGSPH (SEQ ID NO: 4828), GYDSPH (SEQ ID NO: 4829), GHGSPH (SEQ ID NO: 4830), GRDSPH (SEQ ID NO: 4831), GNDSPH (SEQ ID NO: 4832), GPDSPH (SEQ ID NO: 4833), GMGSPH (SEQ ID NO: 4834), GQVSPH (SEQ ID NO: 4835), GHNSPH (SEQ ID NO: 4836), GHPSPH (SEQ ID NO: 4837), or GHSSPH (SEQ ID NO: 4838):
(ii) an amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different: amino acids, relative to any one of the amino acid sequences in (i).
18. The AAV particle of any one of embodiments 1-17, wherein [N1]-[N2]-[N3] comprises:
(i) SGSPHSK (SEQ ID NO: 4839), HDSPHKS (SEQ ID NO: 4840), SGSPHAR (SEQ ID NO: 4841), SGSPHVK (SEQ ID NO: 4842), QDSPHKS (SEQ ID NO: 4843), SGSPHKK (SEQ ID NO: 4844), SGSPHVR (SEQ ID NO: 4845), SGSPHAS (SEQ ID NO: 4846), SGSPHRK (SEQ ID NO: 4847), SGSPHKT (SEQ ID NO: 4848), SHSPHKS (SEQ ID NO: 4849), QSSPHRS (SEQ ID NO: 4850), RGSPHAS (SEQ ID NO: 4851 ), RGSPHSK (SEQ ID NO: 4852), SGSPHKF (SEQ ID NO: 4853), SGSPHKI (SEQ ID NO: 4854),
SGSPHKL (SEQ ID NO: 4855), SGSPHKY (SEQ ID NO: 4856), SGSPHTR (SEQ ID NO: 4857), SHSPHKR (SEQ ID NO: 4858), SGSPHGA (SEQ ID NO: 4859), HDSPHKR (SEQ ID NO: 4860), DDSPHKS (SEQ ID NO: 4861), HESPHKS (SEQ ID NO: 4862), NYSPHKI (SEQ ID NO: 4863), SGSPHSR (SEQ ID NO; 4864), SGSPHSL (SEQ ID NO; 4865), SGSPHSS (SEQ ID NO: 4866), VGSPHSK (SEQ ID NO; 4867), SCSPHRK (SEQ ID NO: 4868). SGSPHFL (SEQ ID NO; 4869), LLSPHWK (SEQ ID NO: 4870), NGSPHSK (SEQ ID NO: 4871), PGSPHSK (SEQ ID NO: 4872), GGSPHSK (SEQ ID NO: 4873), TGSPHSK (SEQ ID NO: 4874), SVSPHGK (SEQ ID NO: 4875), SGSPHTK (SEQ ID NO: 4876), IGSPHSK (SEQ ID NO: 4877), DGSPHSK (SEQ ID NO: 4878), SGSPHNK (SEQ ID NO: 4879), LGSPHSK (SEQ ID NO: 4880), AGSPHSK (SEQ ID NO: 4881), EGSPHSK (SEQ ID NO: 4882), SASPHSK (SEQ ID NO: 4883), SGSPHAK (SEQ ID NO: 4884), HDSPHKI (SEQ ID NO: 4885), YDSPHKS (SEQ ID NO: 4886), HDSPHKT (SEQ ID NO: 4887), RGSPHKR (SEQ ID NO: 4888), HGSPHSK (SEQ ID NO: 4889), RDSPHKS (SEQ ID NO: 4890), NDSPHKS (SEQ ID NO: 4891), QDSPHKI (SEQ ID NO: 4892), PDSPHKI (SEQ ID NO: 4893), PDSPHKS (SEQ ID NO: 4894), MGSPHSK (SEQ ID NO: 4895), HDSPHKH (SEQ ID NO: 4896), QVSPHKS (SEQ ID NO: 4897), HNSPHKS (SEQ ID NO: 4898), NGSPHKR (SEQ ID NO: 4899), HDSPHKY (SEQ ID NO: 4900). NDSPHKI (SEQ ID NO: 4901), HDSPHKI. (SEQ ID NO: 4902), HPSPHWK (SEQ ID NO: 4903), HDSPHKM (SEQ ID NO: 4904), or HSSPHRS (SEQ ID NO: 4905);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g,, consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
19. The AAV particle of any one of embodiments 1-18, wherein [N1]-[N2]-[N3] is or comprises:
(i) GSGSPHSKA (SEQ ID NO; 4697), GHDSPHKSG (SEQ ID NO; 4698), GSGSPHARM (SEQ ID NO: 4906), GSGSPHVKS (SEQ ID NO: 4907), GQDSPHKSG (SEQ ID NO: 4908), GSGSPHASR (SEQ ID NO; 4909), GSGSPHVKI (SEQ ID NO: 4910), GSGSPHKKN (SEQ ID NO: 491 1), GSGSPHVRM (SEQ ID NO: 4912), VSGSPHSKA (SEQ ID NO: 4913), CSGSPHSKA (SEQ ID NO: 4914), GSGSPHRKA (SEQ ID NO: 4915), CSGSPHKTS (SEQ ID NO: 4916), CSHSPHKSG (SEQ ID NO: 4917),
GQSSPHRSG (SEQ ID NO: 4918), GRGSPHASR (SEQ ID NO: 4919), GRGSPHSKA (SEQ ID NO: 4920), GSGSPHKFG (SEQ ID NO: 4921), GSGSPHKIG (SEQ ID NO: 4922), GSGSPHKLG (SEQ ID NO: 4923), GSGSPHKTS (SEQ ID NO: 4924), GSGSPHKTT (SEQ ID NO: 4925), GSGSPHKTY (SEQ ID NO: 4926), GSGSPHKYG (SEQ ID NO: 4927), GSGSPHSKD (SEQ ID NO: 4928), GSGSPHSKP (SEQ ID NO: 4929), GSGSPHTRG (SEQ ID NO: 4930), GSGSPHVRG (SEQ ID NO: 4931), GSHSPHKRG (SEQ ID NO: 4932), GSHSPHKSG (SEQ ID NO: 4933), VSGSPHASR (SEQ ID NO: 4934), VSGSPHGAR (SEQ ID NO: 4935), VSGSPHKFG (SEQ ID NO: 4936), GHDSPHKRG (SEQ ID NO: 4937), GDDSPHKSG (SEQ ID NO: 4938), GHESPHKSA (SEQ ID NO: 4939), GHDSPHKSA (SEQ ID NO: 4940), GNYSPHKIG (SEQ ID NO: 4941), GHDSPHKSR (SEQ ID NO: 4942), GSGSPHSKL (SEQ ID NO: 4943), GSGSPHSRA (SEQ ID NO: 4944), GSGSPHSKR (SEQ ID NO: 4945), GSGSPHSLR (SEQ ID NO: 4946), GSGSPHSRG (SEQ ID NO: 4947), GSGSPHSSR (SEQ ID NO: 4948), RVGSPHSKA (SEQ ID NO: 4949), GSCSPHRKA (SEQ ID NO: 4950), GSGSPHFLR (SEQ ID NO: 4951), GSGSPHSKW (SEQ ID NO: 4952), GSGSPHSKS (SEQ ID NO: 4953), GLLSPIIWKA (SEQ ID NO: 4954), GSGSPHVRR (SEQ ID NO: 4955), GSGSPHSKV (SEQ ID NO: 4956), MSGSPHSKA (SEQ ID NO: 4957), RNGSPHSKA (SEQ ID NO: 4958), TSGSPHSK A (SEQ ID NO: 4959), ISGSPHSKA (SEQ ID NO: 4960), GPGSPHSKA (SEQ ID NO: 4961), GSGSPHSKT (SEQ ID NO: 4962), ESGSPHSKA (SEQ ID NO: 4963), SSGSPHSKA (SEQ ID NO: 4964), GNGSPHSKA (SEQ ID NO: 4965), ASGSPHSKA (SEQ ID NO: 4966), NSGSPHSKA (SEQ ID NO: 4967), ESGSPHSKA (SEQ ID NO: 4968), GGGSPHSKA (SEQ ID NO: 4969), KSGSPHSKA (SEQ ID NO: 4970), GGGSPHSKS (SEQ ID NO: 4971), GSGSPHSKG (SEQ ID NO: 4972), HSGSPHSKA (SEQ ID NO: 4973), GTGSPHSKA (SEQ ID NO: 4974), PSGSPHSKA (SEQ ID NO: 4975), GSVSPHGKA (SEQ ID NO: 4976), RSGSPHSKA (SEQ ID NO: 4977), GSGSPHTKA (SEQ ID NO: 4978), GIGSPHSKA (SEQ ID NO: 4979), WSGSPHSKA (SEQ ID NO: 4980), DSGSPHSKA (SEQ ID NO: 4981), IDGSPHSKA (SEQ ID NO: 4982), GSGSPHNKA (SEQ ID NO: 4983), GLGSPHSKS (SEQ ID NO: 4984), DAGSPHSKA (SEQ ID NO: 4985), DGGSPHSKA (SEQ ID NO: 4986), MEGSPHSKA (SEQ ID NO: 4987), ENGSPHSKA (SEQ ID NO: 4988), GSASPHSKA (SEQ ID NO: 4989), GNGSPHSKS (SEQ ID NO: 4990), KNGSPHSKA (SEQ ID NO: 4991), KEGSPHSKA (SEQ ID NO: 4992), AIGSPHSKA (SEQ ID NO: 4993), GSGSPHSKN (SEQ ID NO: 4994), GSGSPHAKA (SEQ ID NO: 4995), GHDSPHKIG (SEQ ID NO: 4996), GYDSPHKSG (SEQ ID NO: 4997), GHESPHKSG (SEQ ID NO: 4998), GHDSPHKTG (SEQ ID NO: 4999), GRGSPHKRG
(SEQ ID NO: 5000), GQDSPHKSG (SEQ ID NO: 4908), GHDSPHKSL (SEQ ID NO: 5001), GHGSPHSKA (SEQ ID NO: 5002), GHDSPHKSE (SEQ ID NO: 5003), VSGSPHSKA (SEQ ID NO: 4913), GRDSPHKSG (SEQ ID NO: 5004), GNDSPHKSV (SEQ ID NO: 5005), GQDSPHKIG (SEQ ID NO: 5006), GITDSPHKSV (SEQ ID NO: 5007), GPDSPHKIG (SEQ ID NO: 5008), GPDSPHKSG (SEQ ID NO: 5009), GHDSPHKSW (SEQ ID NO: 5010), GHDSPHKSN (SEQ ID NO: 5011), GMGSPHSKT (SEQ ID NO: 50.12), GHDSPHKHG (SEQ ID NO: 5013), GQVSPHKSG (SEQ ID NO: 5014), GDDSPHKSV (SEQ ID NO: 5015), GHNSPHKSG (SEQ ID NO: 5016), GNGSPHKRG (SEQ ID NO: 5017), GHDSPHKYG (SEQ ID NO: 5018), GHDSPHKSQ (SEQ ID NO: 5019), GNDSPHKIG (SEQ ID NO: 5020), GHDSPHKSK (SEQ ID NO: 5021), GHDSPHKLW (SEQ ID NO: 5022), GHPSPHWKG (SEQ ID NO: 5023), GHDSPHKMG (SEQ ID NO: 5024), GHDSPHKMA (SEQ ID NO: 5025), or GHSSPHRSG (SEQ ID NO: 5026):
(ii) an amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, or 8 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
20. The AAV particle of any one of embodiments 1-19, wherein [N3] comprises SK, KA, KS, or SG.
21. The AAV particle of any one of embodiments 1-20, wherein [N3| is or comprises SK A.
KSG, or KYG.
22. The AAV particle of any one of embodiments 1-21, wherein [N2]-[N3] comprises
SPHSK (SEQ ID NO: 4701), SPHKS (SEQ ID NO: 4704), or SPHKY (SEQ ID NO: 4715).
23. The AAV particle of any one of embodiments 1-22, wherein [N2]-[N3] is or comprises SPHSK A (SEQ ID NO: 941).
24. The AAV particle of any one of embodiments 1-22, wherein [N2]-[N3] is or comprises SPHKSG (SEQ ID NO: 946).
25. The AAV particle of any one of embodiments 1-22, wherein [N2]-[N3] is or comprises SPHKYG (SEQ ID NO: 966).
26. The AAV particle of any one of embodiments 1-25, wherein [N1] comprises GS, SG, GH, or HD.
27. The AAV particle of any one of embodiments 1-26, wherein [N1] is or comprises GSG.
28. The AAV particle of any one of embodiments 1-26, wherein [N1] is or comprises GHD.
29. The AAV particle of any one of embodiments 1-23 or 26-27, wherein [N1]-[N2]-[N3] comprises SGSPHSK (SEQ ID NO: 4839).
30. The AAV particle of any one of embodiments 1-22, 24, 26, or 28, wherein [N1]-[N2]- [N3] comprises HDSPHKS (SEQ ID NO: 4840).
31. The AAV particle of any one of embodiments 1-22 or 2.5-27, wherein [N1]-[N2]-[N3] comprises SGSPHKYG (SEQ ID NO: 5027).
32. The AAV particle of any one of embodiments 1-8, 10, 12-2.3, 26-27, or 2.9, wherein [N1]- [N2]-[N3] is or comprises GSGSPHSKA (SEQ ID NO: 4697).
33. The AAV particle of any one of embodiments 1-9, 11-22, 24, 26, 28, or 30, wherein [N1]- [N2]-[N3] is or comprises GHDSPHKSG (SEQ ID NO: 4698).
34. The AAV particle of any one of embodiments 1-8, 10, 12-22, 25-27, or 31, wherein [N1] [N2]-[N3] is or comprises GSGSPHKYG (SEQ ID NO: 4927).
35. The AAV particle of any one of embodiments 1-34, wherein [N1 ]-[N2]-[N3] replaces positions 453-455, numbered according to SEQ ID NO: 138.
36. The AAV particle of any one of embodiments 1-35, wherein the AAV capsid variant comprises an amino acid other than Q at position 456 (e.g., W, K, R. G, L, V, S, P, H, K, I,
M, A, E, or F), an amino acid other than N at position 457 (e.g., Y, C, K, T, II, R, D, V, S, P, G, W, E, F, A, I, M, Q, or L), an amino acid other than Q at position 458 (e.g., G, K, H, R, T, L, D, A, P, I, F, V, M, W, Y, S, E, N, or Y), and/or an amino acid, other than Q at position 459 (e.g., H, L, R, W, K, A, P, E, M, I, S, G, N, Y, C, V, T, D, or V), numbered according to SEQ ID NO: 138.
37. The AAV particle of any one of embodiments 1-36, wherein the AAV capsid variant comprises an amino acid other than Q at position 462 (e.g., W, K, R, G, L, V, S, P, H, K, I, M, A, E, or F), an amino acid other than N at position 463 (e.g., Y, C, K, T, H, R, D, V, S, P, G, W, E, F, A, I, M, Q, or L), an amino acid other than Q at position 464 (e.g., G, K, H, R, T, L, D, A, P, I, F, V, M, W, Y, S, E, N, or Y), and/or an amino acid other than Q at position 465 (e.g., H, L, R, W, K, A, P, E, M, I, S, G, N, Y, C, V, T, D, or V), numbered according to SEQ ID NO: 981, 982, 36, 37, 39, 40. 42-46, 48, 49, 50, 52, 53. 56, or 57.
38. The AAV particle of any one of embodiments 1-37, wherein the AAV capsid variant comprises:
(a) the amino acid Q at position 456, the amino acid N at position 457, the amino acid Q at position 458, and/or the amino acid Q at position 459, numbered according to SEQ ID NO: 138; or
(b) the amino acid Q at position 462, the amino acid N at position 463, the amino acid Q at position 464, and/or the amino acid Q at position 465, numbered according to SEQ ID NO: 981 , 982, 36, 37, 39, 40, 42-46, 48, 49, 50, 52, 53, 56, or 57.
39. 'The AAV particle of any one of embodiments 1-38, wherein the AAV capsid variant further comprises [N4] , wherein [N4] comprises X7 X8 X9 X10, and wherein:
(a) X7 is: Q, W, K, R, G, L, V, S, P, II, K, I, M, A, E, or F;
(b) X8 is: N, Y, C, K, T, H, R, D, V, S, P, G, W, E, F, A, I, M, Q, or L;
(c) .X9 is: Q, G, K, H, R, T, L, D, A, P, I, F, V, M, W, Y, S, E, N, or Y; and
(d) X10 is: Q, II, L, R, W, K, A, P, E, M, I, S, G, N, Y, C, V, T, D, or V; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(d).
40. The AAV particle of embodiment 39, wherein:
(a) X7 of [N4] is Q or R;
(b) X8 of [N4] is N or R;
(c) X9 of [N4] is Q or R; and
(d) X10 of [N4] is Q, L, or R.
41. The AAV particle of embodiment 39 or 40, wherein [N4] is or comprises:
(i) QNQQ (SEQ ID NO: 5028), WNQQ (SEQ ID NO: 5029). QYYV (SEQ ID NO: 5030), RRQQ (SEQ ID NO: 5031), GCGQ (SEQ ID NO: 5032), LRQQ (SEQ ID NO: 5033), RNQQ (SEQ ID NO: 5034), VNQQ (SEQ ID NO: 5035), FREQ (SEQ ID NO: 5036), FNQQ (SEQ ID NO: 5037), LLQQ (SEQ ID NO: 5038), SNQQ (SEQ ID NO: 5039), RLQQ (SEQ ID NO: 5040), LNQQ (SEQ ID NO: 5041), QRKL (SEQ ID NO: 5042), LRRQ (SEQ ID NO: 5043), QRLR (SEQ ID NO: 5044), QRRL (SEQ ID NO: 5045), RRLQ (SEQ ID NO: 5046), RL.RQ (SEQ ID NO: 5047), SKRQ (SEQ ID NO: 5048), QLYR (SEQ ID NO: 5049), QLTV (SEQ ID NO: 5050), QNKQ (SEQ ID NO: 5051), KNQQ (SEQ ID NO: 5052), QKQQ (SEQ ID NO: 5053), QTQQ (SEQ ID NO: 5054), QNHQ (SEQ ID NO: 5055), QIIQQ (SEQ ID NO: 5056), QNQII (SEQ ID NO: 5057), QIIRQ (SEQ ID NO: 5058), LTQQ (SEQ ID NO: 5059), QNQW (SEQ ID NO: 5060), QNTH (SEQ ID NO: 5061), RRRQ (SEQ ID NO: 5062), QYQQ (SEQ ID NO: 5063), QNDQ (SEQ ID NO: 5064), QNRH (SEQ ID NO: 5065), RDQQ (SEQ ID NO: 5066), PNLQ (SEQ ID NO: 5067), HVRQ (SEQ ID NO: 5068), PNQH (SEQ ID NO: 5069), HNQQ (SEQ ID NO: 5070), QSQQ (SEQ ID NO: 5071), QPAK (SEQ ID NO: 5072), QNLA (SEQ ID NO: 5073), QNQL (SEQ ID NO: 5074), QGQQ (SEQ ID NO: 5075), LNRQ (SEQ ID NO: 5076), QNPP (SEQ ID NO: 5077), QNLQ (SEQ ID NO: 5078), QDQE (SEQ ID NO: 5079), QDQQ (SEQ ID NO: 5080), HWQQ (SEQ ID NO: 5081), PNQQ (SEQ ID NO: 5082), PEQQ (SEQ ID NO: 5083), QRTM (SEQ ID NO: 5084), LHQH (SEQ ID NO: 5085), QHRI (SEQ ID NO: 5086), QYIH (SEQ ID NO: 5087), QKFE (SEQ ID NO: 5088), QFPS (SEQ ID NO: 5089), QNPL (SEQ ID NO: 5090), QAIK (SEQ ID NO: 5091), QNRQ (SEQ ID NO: 5092), QYQII (SEQ ID NO: 5093), QNPQ (SEQ ID NO: 5094), QHQL (SEQ ID NO: 5095), QSPP (SEQ ID NO: 5096), QAKL (SEQ ID NO: 5097), KSQQ (SEQ ID NO: 5098), QDRP (SEQ ID NO: 5099), QNLG (SEQ ID NO: 5100), QAFH (SEQ ID NO: 5101), QNAQ (SEQ ID NO: 5102), FINQL (SEQ ID NO: 5103), QKLN (SEQ ID NO: 5104), QNVQ (SEQ ID NO: 5105), QAQQ (SEQ ID NO: 5106), QIPP (SEQ ID NO: 5107), QPPA (SEQ ID NO: 5108). QERP (SEQ ID NO: 5109), QDLQ (SEQ ID NO: 5110), QAMH (SEQ ID NO: 5111), QHPS (SEQ ID NO: 5112), PGLQ (SEQ ID NO: 5113), QGIR (SEQ ID NO: 5114), QAPA (SEQ ID NO: 5115), QIPP (SEQ ID NO: 5116), QTQL (SEQ ID NO: 5117), QAPS (SEQ ID NO: 5118),
QNTY (SEQ ID NO: 5119), QDKQ (SEQ ID NO: 5120), QNIIL (SEQ ID NO: 5121), QIGM (SEQ ID NO: 5122), LNKQ (SEQ ID NO: 5123), PNQL (SEQ ID NO: 5124), QLQQ (SEQ ID NO: 5125), QRMS (SEQ ID NO: 5126), QGIL (SEQ ID NO: 5127), QDRQ (SEQ ID NO: 5128), RDWQ (SEQ ID NO: 5129), QERS (SEQ ID NO: 5130), QNYQ (SEQ ID NO: 5131), QRTC (SEQ ID NO: 5132), QIGH (SEQ ID NO: 5133), QGAI (SEQ ID NO: 5134), QVPP (SEQ ID NO: 5135), QVQQ (SEQ ID NO: 5136), LMRQ (SEQ ID NO: 5137), QYSV (SEQ ID NO: 5138), QAIT (SEQ ID NO: 5139), QKTL (SEQ ID NO: 5140), QLHH (SEQ ID NO: 5141), QNII (SEQ ID NO: 5142), QGHH (SEQ ID NO: 5143), QSKV (SEQ ID NO: 5144), QLPS (SEQ ID NO: 5145), 1GKQ (SEQ ID NO: 5146), QAIH (SEQ ID NO: 5147), QHGL (SEQ ID NO: 5148), QFMC (SEQ ID NO: 5149), QNQM (SEQ ID NO: 5150), QHLQ (SEQ ID NO: 5151), QPAR (SEQ ID NO: 5152), QSLQ (SEQ ID NO: 5153), QSQL (SEQ ID NO: 5154), HSQQ (SEQ ID NO: 5155), QMPS (SEQ ID NO: 5156), QGSL (SEQ ID NO: 5157), QVPA (SEQ ID NO: 5158), HYQQ (SEQ ID NO: 5159), QVPS (SEQ ID NO: 5160), RGEQ (SEQ ID NO: 5161), PGQQ (SEQ ID NO: 5162), LEQQ (SEQ ID NO: 5163), QNQS (SEQ ID NO: 5164), QKVI (SEQ ID NO: 5165), QNND (SEQ ID NO: 5166), QSVH (SEQ ID NO: 5167), QPLG (SEQ ID NO: 5168), HNQE (SEQ ID NO: 5169), QIQQ (SEQ ID NO: 5170), QVRN (SEQ ID NO: 5171), PSNQ (SEQ ID NO: 5172), QVGH (SEQ ID NO: 5173), QRDI (SEQ ID NO: 5174), QMPN (SEQ ID NO: 5175), RGEQ (SEQ ID NO: 5176), PSLQ (SEQ ID NO: 5177), QRDQ (SEQ ID NO: 5178), QAKG (SEQ ID NO: 5179), QSAH (SEQ ID NO: 5180), QSTM (SEQ ID NO: 5181), QREM (SEQ ID NO: 5182), QYRA (SEQ ID NO: 5183), QRQQ (SEQ ID NO: 5184), QWQQ (SEQ ID NO: 5185), QRMN (SEQ ID NO: 5186), GDSQ (SEQ ID NO: 5187), QKIS (SEQ ID NO: 5188), PSMQ (SEQ ID NO: 5189), SPRQ (SEQ ID NO: 5190), MEQQ (SEQ ID NO: 5191), QYQN (SEQ ID NO: 5192), QIRQ (SEQ ID NO: 5193), QSVQ (SEQ ID NO: 5194), RSQQ (SEQ ID NO: 5195), QNKL (SEQ ID NO: 5196), QIQH (SEQ ID NO: 5197), PRQQ (SEQ ID NO: 5198), HTQQ (SEQ ID NO: 5199), QRQH (SEQ ID NO: 5200), RNQE (SEQ ID NO: 5201), QSKQ (SEQ ID NO: 5202), QNQP (SEQ ID NO: 5203), QSPQ (SEQ ID NO: 5204), QTRQ (SEQ ID NO: 5205). QNLH (SEQ ID NO: 5206), QNQE (SEQ ID NO: 5207), LNQP (SEQ ID NO: 5208), QNQD (SEQ ID NO: 5209), QNLL (SEQ ID NO: 5210), QLVI (SEQ ID NO: 5211), RTQE (SEQ ID NO: 5212), QTHQ (SEQ ID NO: 5213), QDQH (SEQ ID NO: 5214), QSQH (SEQ ID NO: 5215), VRQQ (SEQ ID NO: 5216). AWQQ (SEQ ID NO: 5217), QSVP (SEQ ID NO: 5218), QNIQ (SEQ ID NO: 5219), LDQQ (SEQ ID NO: 5220), PDQQ (SEQ ID NO: 5221), ESQQ (SEQ ID NO: 5222), QRQL (SEQ ID NO: 5223), QIIV (SEQ ID NO: 5224), QKQS (SEQ ID NO: 5225), QSHQ (SEQ ID NO: 5226), QFVV (SEQ ID NO: 5227), QSQP (SEQ ID NO: 5228),
QNEQ (SEQ ID NO: 5229), INQQ (SEQ ID NO: 5230), RNRQ (SEQ ID NO: 5231 ), RDQK (SEQ ID NO: 5232), QWKR (SEQ ID NO: 5233), ENRQ (SEQ ID NO: 5234), QTQP (SEQ ID NO: 5235), QKQL (SEQ ID NO: 5236), RNQL (SEQ ID NO: 5237), ISIQ (SEQ ID NO: 5238), QTVC (SEQ ID NO: 5239), QQIM (SEQ ID NO: 5240), LNIIQ (SEQ ID NO: 5241), QNQA (SEQ ID NO: 5242), QMIH (SEQ ID NO: 5243), RNHQ (SEQ ID NO: 5244), or QKMN (SEQ ID NO: 5245):
(ii) an amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, or 3 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
42. The AAV particle of any one of embodiments 39-41, wherein [N1]-[N2]-[N3]-[N4] is or comprises:
(i) the amino acid sequence of any of SEQ ID NOs: 1800-2241;
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g,, any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
43. The AAV particle of any one of embodiments 39-42, wherein [N1]-[N2]-[N3]-[N4] is or comprises GSGSPHSKAQNQQ (SEQ ID NO: 1801).
44. The AAV particle of any one of embodiments 39-42, wherein [N1]-[N2]-[N3]-[N4] is or composes GHDSPHKSGQNQQ (SEQ ID NO: 1800).
45. The AAV particle of any one of embodiments 39-42, wherein [N1]-[N2]-[N3]-[N4| is or comprises GSGSPHKYGQNQQT (SEQ ID NO: 910).
46. The AAV particle of any one of embodiments 1-45. wherein the AAV capsid variant comprises an amino acid other than T at position 450 (e.g., S, Y, M, A, C, I, R, L, D, F, V, Q,
N, H, E, or G), an amino acid other than I at position 451 (e.g., M, P, E, N, D, S, A, T, G, Q, F, V, L, C, H, R, W, or L), and/or an amino acid other than N at position 452 (e.g., M, E, G, Y, W, T, I, Q, F, V, A, L, I, P, K, R, H, S, D, or S), numbered according to any one of SEQ ID NOs: 36-59, 138, 981, or 982.
47. The AAV particle of any one of embodiments 1-46, wherein the AAV capsid variant compnses the amino acid T at position 450, the amino acid I at position 451 , and/or the amino acid N at position 452, numbered according to any one of SEQ ID NOs: 138, 981, or 982.
48. The AAV particle of any one of embodiments 1-47, wherein the AAV capsid variant further comprises [N0], wherein [N0] comprises XA XB and XC, and wherein:
(a) XA is: T, S, Y, M, A, C, I. R. L. D. F. V. Q. N, H. E, or G;
(b) XB is: I, M, P, E, N, D, S, A, T, G, Q, F, V, I.,, C, H, R, W, or L; and
(c) XC is: N, M, E, G, Y, W, T, I, Q, F, V, A, L, I, P, K, R, H, S, D, or S; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c).
49. The AAV particle of embodiment 48, wherein [N0] is or comprises TIN, SMN, TIM, YLS, GLS, MPE, MEG, MEY, AEW, CEW, ANN, IPE. ADM, IEY, ADY, IET, MEW, CEY, RIN, MEI, LEY, ADW, lEI, DIM, FEQ, MEF, CDQ, L PE, IEN, MES, AEI, VEY, UN, TSN, IEV, MEM, AEV, MDA, VEW, AEQ, LEW, MEL, MET, MEA, IES, MEV, CEI, ATN, MDG, QEV, ADQ, NMN, IEM, ISN, TGN, QQQ, HDW, LEG, TII, TFP, TEK, EIN, TVN, TEN, SIN, TER, TSY, ELH, AIN. SVN, TDN, TFH, TVH, TEN. TSS, TID, TCN, NIN, TEH, AEM, AIK, TDK, TFK, SDQ, TEI, NTN, TET, SIK, TEL., TEA, TAN, TTY, TFS, TES, TEN, TED, TNN, EVH, TIS, TVR, TDR, TIK, NHI, TIP, ESD, TDL, TVP, TVI, AEH, NCL, TVK, NAD, TIT, NCV, TIR, NAL. VLN. HQ. TEE, TRE, QGE, SEK, NVN, GGE, EFV, SDK, TEQ, EVQ, TEY, NOW, TDV, SDI, NSI, NSL, EVV, TEP, SEL, TWQ, TEV, AVN, GVL, TEN, TEG, TRD, NAT, AEN, AET, ETA, NNL, or any dipeptide thereof.
50. The MV particle of embodiment 48 or 49, wherein [N0]-[N1]-[N2]-[N3]-[N4] is or compnses:
(i) the amino acid sequence of any one of SEQ ID NOs: 2242-2886;
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i): or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
51. The AAV particle of any one of embodiments 48-50, wherein [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TINGSGSPHSKAQNQQ (SEQ ID NO: 2242).
52. The AAV particle of any one of embodiments 48-50, wherein |N0]-[N1 ]-[N2]-[N3]-[N4] is or comprises TINGHDSPHKSGQNQQ (SEQ ID NO: 2243).
53. The AAV particle of any one of embodiments 48-52, wherein [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TINGSGSPHKYGQNQQT (SEQ ID NO: 5246).
54. The AAV particle of any one of embodiments 1-53, wherein [N1]-[N2]-[N3] is present in loop IV.
55. The AAV particle of any one of embodiments 48-54, wherein [N0] and [N4] are present in loop IV.
56. The AAV particle of any one of embodiments 48-55, wherein [N0] is present immediately subsequent to position 449, numbered according to SEQ ID NO: 138.
57. The AAV particle of any one of embodiments 48-56, wherein [N0] is present immediately subsequent to position 449, numbered according to any one of SEQ ID NOs: 36- 59, 981, 01' 982.
58. lire AAV particle of any one of embodiments 48-57, wherein [N0] replaces positions
450, 451, and 452. (e.g., T450, I451, and N452), numbered according to SEQ ID NO: 138.
59. The AAV particle of any one of embodiments 48-58, wherein [N0] replaces positions 450-452 (e.g., T450, 1451, and N452), numbered according to any one of SEQ ID NOs: 36- 59, 981 , or 982.
60. The AAV particle of any one of embodiments 48-59, wherein [N0] corresponds to positions 450-452 of any one of SEQ ID NOs: 36-59, 138, 981 or 982.
61 . The AAV particle of any one of embodiments 48-60, wherein [N0] is present immediately subsequent to position 449 and wherein [N0] replaces positions 450-452 (e.g., T450, 1451, and N452), numbered according to SEQ ID NO: 138.
62. The AAV particle of any one of embodiments 48-61, wherein [N0] is present immediately subsequent to position 449 and wherein [N0] replaces positions 450-452 (e.g., T450, 1451, and N452), numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
63. The AAV particle of any one of embodiments 1-62, wherein [N1] is present immediately subsequent to position 452, numbered according to the amino acid sequence of SEQ ID NO: 138.
64. The AAV particle of any one of embodiments 1-63, wherein [N1] is present immediately subsequent to position 452, numbered according to SEQ ID NO: 981 or 982.
65. The AAV particle of any one of embodiments 1-61, wherein [N1] replaces positions 453
455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO; 138.
66. The AAV particle of any one of embodiments 1-64, wherein [N1] replaces positions 453 (e.g., G453), numbered according to SEQ ID NO: 138.
67. The AAV particle of any one of embodiments 1-65, wherein [N1] replaces positions 453- 455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 981.
68. The AAV particle of any one of embodiments 1-65 or 67, wherein [N1] replaces positions 453-455, numbered according to SEQ ID NO: 982.
69. The AAV particle of any one of embodiments 1-65, 67, or 68, wherein [N1] is present immediately subsequent to position 452 and wherein [N1] replaces positions 453-455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 138.
70. The AAV particle of any one of embodiments 1-64 or 66, wherein [N1] is present immediately subsequent to position 452 and wherein [N1] replaces positions 453 (e.g., G453), numbered according to SEQ ID NO: 138.
71. The AAV particle of any one of embodiments 1-64, 66 or 70, wherein [N1] is present immediately subsequent to position 452 and wherein [N1] replaces positions 453-455, numbered according to SEQ ID NO; 4, 36, 981, or 982.
72. The AAV particle of any one of embodiments 1-71, wherein [N1] corresponds to positions 453-455, numbered according to any one of SEQ ID NOs: 4, 36-59, 981, or 982.
73. The AAV particle of any one of embodiments 1-72, wherein the AAV capsid variant comprises an amino acid other than S at position 454 and/or an amino acid other than G at position 455, numbered according to SEQ ID NO: 138, 981 , or 982.
74. The AAV particle of any one of embodiments 1-73, wherein the AAV capsid variant comprises the amino acid H at position 454 and the amino acid D at position 455, numbered according to SEQ ID NO: 138 or 982.
75. The AAV particle of any one of embodiments 1-74, wherein the AAV capsid variant comprises a substitution at position 454 (e.g., S454H) and/or a substitution at position 455 (e.g., G455D), numbered according to SEQ ID NO: 138.
76. The AAV particle of any one of embodiments 1-75, wherein the AAV capsid variant comprises the amino acid II at position 454 and the amino acid D at position 455, and further comprises the amino acid sequence SPHSKA (SEQ ID NO: 941) immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
77. The AAV particle of any one of embodiments 1-76, wherein the AAV capsid variant comprises the amino acid H at position 454 and the amino acid D at position 455, numbered according to SEQ ID NO: 982.
78. The AAV particle of any one of embodiments 1-77, wherein the AAV capsid variant comprises the amino acid H at position 454 and the amino acid I) at position 455, and fiirther composes the amino acid sequence SPHSKA (SEQ ID NO: 941) immediately subsequent to position 455, numbered according to SEQ ID NO: 982.
79. The A AV particle of any one of embodiments 1-72, wherein the AAV capsid vanant comprises the amino acid S at position 454 and the amino acid G at position 455, numbered according to SEQ ID NO: 138.
80. The AAV particle of any one of embodiments 1-72 or 79, wherein the AAV capsid variant comprises the amino acid S at position 454 and the amino acid G at position 455, and further comprises the amino acid sequence SPHSKA (SEQ ID NO: 941) immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
81. The AAV particle of any one of embodiments 1-72, 79, or 80, wherein the .AAV capsid variant comprises the amino acid S at position 454 and the amino acid G at position 455, numbered according to SEQ ID NO: 981.
82. The AAV particle of any one of embodiments 1-72 or 79-81, wherein the AAV capsid variant comprises the amino acid S at position 454 and the amino acid G at position 455, and fiirther comprises the amino acid sequence SPHSKA (SEQ ID NO: 941) immediately subsequent to position 455, numbered according to SEQ ID NO: 981.
83. The AAV particle of any one of embodiments 1-82, wherein [N2] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
84. The AAV particle of any one of embodiments 1-83, wherein [N2] corresponds to positions 456-458 (e.g., S456, P457, H458) of SEQ ID NO: 981 or 982.
85. The AAV particle of any one of embodiments 1-83, wherein [N2] corresponds to positions 456-458 (e.g., S456, P457, H458) of any one of SEQ ID NOs: 4 or 36-59.
86. The AAV particle of any one of embodiments 1-85, wherein [N2]-[N3] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
87. The AAV particle of any one of embodiments 1-86, wherein [N2] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 4, 36, 981 , or 982.
88. The AAV particle of any one of embodiments 1-87, wherein [N2]-[N3] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 4, 36, 981, or 982.
89. The AAV particle of any one of embodiments 1-88, wherein [N2]-[N3] corresponds to positions 456-461 (e.g., S456, P457, H458, S459, K460, A461) of SEQ ID NO: 981.
90. The AAV particle of any one of embodiments 1-88, wherein [N2]-[N3] corresponds to positions 456-461 (e.g., S456, P457, H458, K459, S460, G461) of SEQ ID NO: 982.
91. The AAV particle of any one of embodiments 1-90, wherein [N2] is present immediately subsequent to [N1] .
92. The AAV particle of any one of embodiments 1-64, 66, 70, or 71, wherein [N3] is present immediately subsequent to [N2] and replaces positions 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
93. The AAV particle of any one of embodiments 1-1-64, 66, 70, 71, or 92, wherein [N3] is present immediately subsequent to [N1]-[N2] and replaces positions 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
94. lire AAV particle of any one of embodiments 39-93, wherein [N4] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
95. The AAV particle of any one of embodiments 39-94, wherein [N4] replaces positions 456-459 (e.g., Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
96. The AAV particle of any one of embodiments 39-95, wherein [N4] corresponds to positions 462-465 (e.g., Q462, N463, Q464, Q465) of SEQ ID NO: 4, 36, 981, or 982.
97. The AAV particle of any one of embodiments 39-96, wherein [N2]-[N3]-[N4] replaces positions 456-459 (e.g., Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
98. The AAV particle of any one of embodiments 39-97, wherein [N2]-[N3]-[N4] is present immediately subsequent to position 455, and wherein [N2]-[N3]-[N4] replaces positions 456- 459 (e.g., Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
99. The AAV particle of any one of embodiments 39-98, wherein [N2]-[N3]-[N4] corresponds to positions 456-465 (e.g., S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465) of SEQ ID NO: 981.
100. The AAV particle of any one of embodiments 39-98, wherein [N2]-[N3]-[N4] corresponds to positions 456-465 (e.g., S456, P457, H458, K459, S460, G461, Q462, N463, Q464, Q465) of SEQ ID NO: 982.
101. The AAV particle of any one of embodiments 39-98, wherein [N2]-[N3]-[N4] corresponds to positions 456-465 of any one of SEQ ID NOs: 4 or 36-59.
102. The AAV particle of any one of embodiments 39-101 , wherein [N1]-[N2]-[N3]-[N4] replaces positions 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
103. The AAV particle of any one of embodiments 39-102, wherein [N1]- [N2]-[N3]-[N4] is present immediately subsequent to position 452, and wherein [N1]-[N2]-[N3]-[N4] replaces positions 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
104. The AAV particle of any one of embodiments 39-99, 102, or 103, wherein [N1]-[N2]- [N3]-[N4] corresponds to positions 453-465 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465) of SEQ ID NO: 981.
105. The AAV particle of any one of embodiments 39-98, 100, 102, or 103, wherein [N1] [N2]-[N3]-[N4] corresponds to positions 453-465 (e.g., G453, H454, D455, S456, P457, H458, K459, S460, G461, Q462, N463, Q464, Q465) of SEQ ID NO: 982.
106. The AAV particle of any one of embodiments 39-98, 102, or 103, wherein [N1]-[N2] [N3]-[N4] corresponds to positions 453-465 of any one of SEQ ID NOs: 4 or 36-59.
107. The AAV particle of any one of embodiments 1-99 or 102-104, wherein [N1 |-[N2]-[N3] corresponds to positions 453-461 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461) of SEQ ID NO: 981.
108. The AAV particle of any one of embodiments 1-98, 100, 102, 103, or 105, wherein [N1]-[N2]-[N3] corresponds to positions 453-461 (e.g., G453, H454, D455, S456, P457, H458, K459, S460, G461) of SEQ ID NO: 982.
109. The AAV particle of any one of embodiments 39-98, 102, 103, or 106, wherein [N1]- [N2]-[N3] corresponds to positions 453-461 of any one of SEQ ID NOs: 36-59.
110. The AAV particle of any one of embodiments 48-109, wherein [N0]-|N1 ]-[N2]-[N3]- [N4] replaces positions 450-459 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
111. The AAV particle of any one of embodiments 48-110, wherein [N0]-[N1]-[N2]-[N3]- [N4] is present immediately subsequent to position 449, and wherein [N0]-[N1]-[N2]-[N3]- [N4] replaces positions 450-459 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
112. The AAV particle of any one of embodiments 48-99, 102-104, or 106, wherein [N0]- [N1]-[N2]-[N3]-[N4] corresponds to positions 450-465 (e.g., T450, 1451 , N452, G453, S454,
G455, S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465) of SEQ ID NO: 981.
113. The AAV particle of any one of embodiments 48-98, 100, 102, 103, 105, or 108, wherein [N0]-[N1J-[N2]-[N3]-[N4] corresponds to positions 450-465 (e.g., T450, 1451, N452, G453. H454, D455. S456, P457, H458, K459, S460. G461, Q462, N463, Q464, Q465) of SEQ ID NO: 982.
114. The AAV particle of any one of embodiments 48-98, 102, 103, 106, or 109, wherein [N0]-[N1]-[N2]-[N3]-[N4] corresponds to positions 450-465 of any one of SEQ ID NOs: 36- 59.
115. The AAV particle of any one of embodiments 39-114, wherein [N4] replaces positions 462-465 (e.g., Q462, N463, Q464, and. Q465), numbered according to SEQ ID NO: 4, 36, 981 , or 982.
116. The AAV particle of any one of embodiments 39-115, wherein [N2]-|N3]-[N4| replaces positions 462-465 (e.g., Q462, N463, Q464, and Q465), numbered according to SEQ ID NO: 4, 36, 981, or 982.
1 17. The AAV particle of any one of embodiments 39-1 .16, wherein [N2]-[N3]-[N4] is present immediately subsequent to position 455, and wherein [N2]-[N3]-[N4] replaces positions 462-465 (e.g., Q462, N463, Q464, and Q465 ), numbered according to SEQ ID NO: 4, 36, 981, or 982.
118. The AAV particle of any one of embodiments 1-117, wherein [N3] is present immediately subsequent to [N2].
1 19. The AAV particle of any one of embodiments 1 -1 18, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N2]-[N3],
120. The AAV particle of any one of embodiments .1-119, wirerein the AAV capsid, variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3].
121. The AAV particle of any one of embodiments 48-120, wherein the AAV capsid variant comprises, from N-terminus to C -terminus, |N0]-[N1]-[N2]-[N3].
122. The AAV particle of any one of embodiments 39-121 , wherein the AAV capsid variant comprises, from N-terminus to C -terminus, [N1]-[N2]-[N3]-[N4J.
123. The AAV particle of any one of embodiments 48-122, wherein the A AV capsid vanant comprises, from N-terminus to C -terminus, [N0]-[N1]-[N2]-[N3]-[N4].
124. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid other T at position 460 (e.g., N, I, C, H, R, L, D, Y, A, M, Q, 1, E, K, P, G or S), numbered according to SEQ ID NO: 138.
125. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises the amino acid N, I, C, H, R, L, D, Y, A, M, Q, I, E, K, P, G or S at position 460, numbered according to SEQ ID NO: 138.
126. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid other T at position 466 (e.g., N, I, C, H, R, L, D, Y, A, M, Q, I, E, K, P, G or S), numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
127. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises the amino acid N, I, C, H, R, L, D, Y, A, M, Q, I, E, K, P, G or S at position 466, numbered according to any one of SEQ ID NOs: 36-59, 981 or 982.
128. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid other K at position 449 (e.g., an E, an N, or a T), numbered according to any one of SEQ ID NOs: 36-59, 138, 981 , or 982,
129. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises the amino E, N, or T at position 449, numbered according to any one of SEQ ID NOs: 36-59, 138, 981 or 982.
130. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises [A] [B] (SEQ ID NO: 4694), wherein:
(i) [A] comprises the amino acid sequence of GSGSPH (SEQ ID NO: 4695); and
(ii) [B] comprises X1 X2 X3 X4 X5 X6 X7 , wherein:
(a) X1 is: S, C, F, or V;
(b) X2 is: K, L, R, I, E, Y, V, or S;
(c) X3 is: A, R, L, G, I, Y, S, F, or W;
(d) X4 is: W, Q, R, G, L, V, S, or F;
(e) X5 is: N, Y, R. C, K, or L;
(f) X6 is: Q, G, K, R, T, L, or Y; and
(g) X7 is: Q, L, R, or V; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(g).
131. The AA V particle of embodiment 130, wherein
(a) X1 is S;
(b) X2 is K or L;
(c) X3 is: A, R. or L;
(d) X4 is: Q or R;
(e) X5 is: N or R;
(f) X6 is: Q or R; and
(g) X7 is: Q, L, or R.
132. The AAV particle of embodiment 130 or 131, wherein [B] comprises:
(i) SLLWNQQ (SEQ ID NO: 5247), SKAQYYV (SEQ ID NO: 5248), SKLRRQQ (SEQ ID NO: 5249), SIWQNQQ (SEQ ID NO: 5250), SKAGCGQ (SEQ ID NO: 5251), SRAQNQQ (SEQ ID NO: 5252), SKRLRQQ (SEQ ID NO: 5253), SLRRNQQ (SEQ ID NO: 5254), SRGRNQQ (SEQ ID NO: 5255), SEIVNQQ (SEQ ID NO: 5256), SSRRNQQ (SEQ ID NO: 5257), CLLQNQQ (SEQ ID NO: 5258), SKAFRLQ (SEQ ID NO: 5259), CLAQNQQ (SEQ ID NO: 5260), FLRQNQQ (SEQ ID NO: 5261), SLRFNQQ (SEQ ID NO: 5262), SYLRNQQ (SEQ ID NO: 5263), CSLQNQQ (SEQ ID NO: 5264), VLWQNQQ (SEQ
ID NO: 5265), SKWLLQQ (SEQ ID NO: 5266), SLWSNQQ (SEQ ID NO: 5267), SKRRLQQ (SEQ ID NO: 5268), SVYLNQQ (SEQ ID NO: 5269), SLWLNQQ (SEQ ID NO: 5270), SKAQRKL (SEQ ID NO: 5271), SKALRRQ (SEQ ID NO: 5272), SKAQRLR (SEQ ID NO: 5273), SKAQNQQ (SEQ ID NO: 5274), SKAQRRL (SEQ ID NO: 5275), SKARRQQ (SEQ ID NO: 5276), SKARRLQ (SEQ ID NO: 5277), SKSRRQQ (SEQ ID NO: 5278). SKARLRQ (SEQ ID NO: 5279), SKASKRQ (SEQ ID NO: 5280), VRRQNQQ (SEQ ID NO: 5281), SKAQLYR (SEQ ID NO: 5282), SLFRNQQ (SEQ ID NO: 5283), SKAQLTV (SEQ ID NO: 5284);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g,, any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i): or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
133. The .AAV particle of any one of embodiments 130-132, wherein [A][B] comprises:
(i) GSGSPHSLLWNQQ (SEQ ID NO: 5285), GSGSPHSKAQYYV (SEQ ID NO: 2060), GSGSPHSKLRRQQ (SEQ ID NO: 2061), GSGSPHSIWQNQQ (SEQ ID NO: 5286), GSGSPHSKAGCGQ (SEQ ID NO: 2062), GSGSPHSRAQNQQ (SEQ ID NO: 2063), GSGSPHSKRLRQQ (SEQ ID NO: 2064), GSGSPHSLRRNQQ (SEQ ID NO: 2065). GSGSPHSRGRNQQ (SEQ ID NO: 2066), GSGSPHSEIVNQQ (SEQ ID NO: 5287), GSGSPHSSRRNQQ (SEQ ID NO: 2067), GSGSPHCLLQNQQ (SEQ ID NO: 5288), GSGSPHSKAFRLQ (SEQ ID NO: 2068), GSGSPHCLAQNQQ (SEQ ID NO: 5289), GSGSPHFLRQNQQ (SEQ ID NO: 2070), GSGSPHSLRFNQQ (SEQ ID NO: 2071), GSGSPHSYLRNQQ (SEQ ID NO: 5290), GSGSPHCSLQNQQ (SEQ ID NO: 52.91), GSGSPHVLWQNQQ (SEQ ID NO: 5292), GSGSPHSKWLLQQ (SEQ ID NO: 2072), GSGSPHSLWSNQQ (SEQ ID NO: 5293), GSGSPHSKRRLQQ (SEQ ID NO: 2073), GSGSPHSVYLNQQ (SEQ ID NO: 5294), GSGSPHSLWLNQQ (SEQ ID NO: 5295), GSGSPHSKAQRKL (SEQ ID NO: 2074), GSGSPHSKALRRQ (SEQ ID NO: 2075), GSGSPHSKAQRLR (SEQ ID NO: 2076), GSGSPHSKAQNQQ (SEQ ID NO: 1801), GSGSPHSKAQRRL (SEQ ID NO: 2077), GSGSPHSKARRQQ (SEQ ID NO: 2078), GSGSPHSKARRLQ (SEQ ID NO: 2079), GSGSPHSKSRRQQ (SEQ ID NO: 2080), GSGSPHSKARLRQ (SEQ ID NO: 2082), GSGSPHSKASKRQ (SEQ ID NO: 2083),
GSGSPHVRRQNQQ (SEQ ID NO: 2084), GSGSPHSKAQLYR (SEQ ID NO: 2085), GSGSPHSLFRNQQ (SEQ ID NO: 5296), GSGSPHSKAQLTV (SEQ ID NO: 2086):.
(li) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in ( i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i)
134. The AAV particle of any one of embodiments 130-133, wherein the AAV capsid variant further comprises one, two, or all of an amino acid other than T at position 450 (e.g., S, Y, or G), an amino acid other than I at position 451 (e.g., M or L), and/or an amino acid other than N at position 452 (e.g., S), numbered according to SEQ ID NO: 138.
135. The AAV particle of any one of embodiments 130-134, wherein the AAV capsid variant further comprises an S at position 450 and an M at position 451, numbered according to SEQ ID NO: 138.
136. The AAV particle of any one of embodiments 130-134, wherein the .AAV capsid variant further comprises a Y at position 450, an L at position 451, and an S at position 452, numbered according to SEQ ID NO: 138.
137. The AAV particle of any one of embodiments 130-134, wherein the AAV capsid variant further comprises a G at position 450, an L at position 451, and an S at position 452, numbered according to SEQ ID NO: 138.
138. The AAV particle of any one of embodiments 130-137, wherein [A] [B] is present in loop IV.
139. The AAV particle of any one of embodiments 130-138, wherein [A] is present immediately subsequent to position 452, numbered according to SEQ ID NO: 138.
140. The AAV particle of any one of embodiments 130-139, wherein [A] replaces positions 453-455 (e.g., G453, S454, G455), numbered according to SEQ ID NO: 138.
141. The AAV particle of any one of embodiments 130-140, wherein [A] is present immediately subsequent to position 452, and wherein [A] replaces positions 453-455 (e.g., G453, S454, G455), numbered according to SEQ ID NO: 138.
142. The AAV particle of any one of embodiments 130-141, wherein [B] is present immediately subsequent to [A],
143. The AAV particle of any one of embodiments 130-142, wherein [B] replaces positions 456-459 (e.g., Q456, N457, Q458, Q459), numbered according to SEQ ID NO: 138.
144. The AAV particle of any one of embodiments 130-143, wherein [A] [B] replaces positions 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, Q459), numbered according to SEQ ID NO: 138.
145. The AAV particle of any one of embodiments 130-144, wherein [A] [B] is present immediately subsequent to position 452, and wherein [A][B] replaces positions 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, Q459), numbered according to SEQ ID NO: 138.
146. The AAV particle of any one of embodiments 130-145, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [A][B],
147. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV 9 capsid vanant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises [A] [B] (SEQ ID NO; 4699), wherein:
(i) [A] comprises X 1 X 2 X3 X4 X5 X6, wherein
(a) X1 is T, M, A, C, I, R, L, D, F, V, Q, N, or II;
(b) X2 is 1, P, E, N, D, S, A, T, M, or Q;
(c) X3 is N, E, G, Y, W. M, T, I, K, Q. F. S, V, A, or L;
(d) X4 is G. D, R, or E;
(e) X5 is H, Q, N, or D;
(f) X6 is D or R;
optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(f); and
(ti) [B] comprises SPHKSG (SEQ ID NO: 946).
148. The AAV particle of embodiment 147, wherein
(a) X1 is: T, M, A, or I;
(b) X2 is: E, I or I);
(c) X3 is: N, Q, Y, I, M, or V;
(d) X4 is G;
(e) X5 is H; and
(f) X6 is D.
149. The AAV particle of embodiment 147 or 148, wherein [A] comprises:
(i) TINGHD (SEQ ID NO: 5297), MPEGHD (SEQ ID NO: 5298), MEGGHD (SEQ ID NO: 5299), MEYGHD (SEQ ID NO: 5300), AEWGIID (SEQ ID NO: 5301), CEWGHD (SEQ ID NO: 5302), ANNGQD (SEQ ID NO: 5303), IPEGHD (SEQ ID NO: 5304), ADMGHD (SEQ ID NO: 5305), IEYGHD (SEQ ID NO: 5306), ADYGHD (SEQ ID NO: 5307), IETGHD (SEQ ID NO: 5308), MEWGHD (SEQ ID NO: 5309), CEYGHD (SEQ ID NO: 5310), RINGHD (SEQ ID NO: 5311), MEIGHD (SEQ ID NO: 5312), LEYGHD (SEQ ID NO: 5313), ADWGHD (SEQ ID NO: 5314), IEIGHD (SEQ ID NO: 5315), TIKDND (SEQ ID NO: 5316), DIMGHD (SEQ ID NO: 5317), FEQGHD (SEQ ID NO: 5318), MEFGHD (SEQ ID NO: 5319), CDQGHD (SEQ ID NO: 5320), LPEGHD (SEQ ID NO: 5321), IENGHD (SEQ ID NO: 5322), MESGHD (SEQ ID NO: 5323), AEIGHD (SEQ ID NO: 5324), VEYGHD (SEQ ID NO: 5325), TSNGDD (SEQ ID NO: 5326). lEVGHD (SEQ ID NO: 5327), MEMGHD (SEQ ID NO: 5328), AEVGHD (SEQ ID NO: 5329), MDAGHD (SEQ ID NO: 5330), VEWGHD (SEQ ID NO: 5331), AEQGHD (SEQ ID NO: 5332), LEWGHD (SEQ ID NO: 5333), MELGHD (SEQ ID NO: 5334). METGHD (SEQ ID NO: 5335), MEAGHD (SEQ ID NO: 5336), TINRQR (SEQ ID NO: 5337), lESGHD (SEQ ID NO: 5338), TAKDIID (SEQ ID NO: 5339), MEVGHD (SEQ ID NO: 5340), CEIGHD (SEQ ID NO: 5341), ATNGHD (SEQ ID NO: 5342), MDGGHD (SEQ ID NO: 5343), QEVGHD (SEQ ID NO: 5344), ADQGHD (SEQ ID NO: 5345), NMNGHD (SEQ ID NO: 5346), TPWEHD (SEQ ID NO: 5347), lEMGHD (SEQ ID NO: 5348), TANEHD (SEQ ID NO: 5349), QQQGHD (SEQ ID NO: 5350), TPQDHD (SEQ ID NO: 5351), HDWGIID (SEQ ID NO: 5352), IEGGHD (SEQ ID NO: 5353)
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof:
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
150. The AAV particle of any one of embodiments 147-149, wherein [A][B] comprises: (i) T1NGHDSPHKR (SEQ ID NO: 5354), MPEGHDSPHKS (SEQ ID NO: 5355), MEGGHDSPHKS (SEQ ID NO: 5356), MEYGHDSPHKS (SEQ ID NO: 5357), AEWGHDSPHKS (SEQ ID NO: 5358), CEWGHDSPHKS (SEQ ID NO: 5359), ANNGQDSPHKS (SEQ ID NO: 5360), IPEGHDSPHKS (SEQ ID NO: 5361), ADMGHDSPHKS (SEQ ID NO: 5362), IEYGHDSPHKS (SEQ ID NO: 5363). ADYGHDSPHKS (SEQ ID NO: 5364), IETGHDSPHKS (SEQ ID NO: 5365), MEWGHDSPHKS (SEQ ID NO: 5366), CEYGHDSPHKS (SEQ ID NO: 5367), RINGHDSPHKS (SEQ ID NO: 5368), MEIGHDSPHKS (SEQ ID NO: 5369), LEYGHDSPHKS (SEQ ID NO: 5370), ADWGHDSPHKS (SEQ ID NO: 5371), IEIGHDSPHKS (SEQ ID NO: 5372), TIKDNDSPHKS (SEQ ID NO: 5373), DIMGHDSPHKS (SEQ ID NO: 5374), FEQGHDSPHKS (SEQ ID NO: 5375), MEFGHDSPHKS (SEQ ID NO: 5376), CDQGHDSPHKS (SEQ ID NO: 5377). LPEGHDSPHKS (SEQ ID NO: 5378), IENGHDSPHKS (SEQ ID NO: 5379), MESGHDSPHKS (SEQ ID NO: 5380), AEIGIIDSPHKS (SEQ ID NO: 5381 ), VEYGHDSPHKS (SEQ ID NO: 5382), TSNGDDSPHKS (SEQ ID NO: 5383), IEYGHDSPHKS (SEQ ID NO: 5384), MEMGHDSPHKS (SEQ ID NO: 5385), AEVGHDSPHKS (SEQ ID NO: 5386), MDAGHDSPHKS (SEQ ID NO: 5387), VEWGHDSPHKS (SEQ ID NO: 5388), AEQGHDSPHKS (SEQ ID NO: 5389), LEWGHDSPHKS (SEQ ID NO: 5390), MELGHDSPHKS (SEQ ID NO: 5391), MEIGHDSPHKS (SEQ ID NO: 5392), MEAGHDSPHKS (SEQ ID NO: 5393), TINRQRSPHKS (SEQ ID NO: 5394), IESGHDSPHKS (SEQ ID NO: 5395), TAKDHDSPHKS (SEQ ID NO: 5396), MEYGHDSPHKS (SEQ ID NO: 5397), CEIGHDSPHKS (SEQ ID NO: 5398), ATNGHDSPHKS (SEQ ID NO: 5399), MDGGHDSPHKS (SEQ ID NO: 5400), QEVGHDSPHKS (SEQ ID NO: 5401), ADQGHDSPHKS (SEQ ID NO: 5402), NMNGHDSPHKS (SEQ ID NO: 5403), TPWEHDSPHKS (SEQ ID NO: 5404), IEMGHDSPHKS (SEQ ID NO: 5405),
TANEIIDSPHKS (SEQ ID NO: 5406), TINGHDSPHKS (SEQ ID NO: 5407), QQQGHDSPHKS (SEQ ID NO: 5408), TPQDHDSPHKS (SEQ ID NO: 5409), HDWGHDSPHKS (SEQ ID NO: 5410), lEGGHDSPHKS (SEQ ID NO: 5411)
(li) an amino acid sequence comprising any portion of an amino acid sequence in (i). e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof:
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
151. The AAV particle of any one of embodiments 147-150, wherein the AAV capsid variant further comprises one, two, three, four, or all of an amino acid other than Q at position 456 (e.g., R or L), N at position 457 (e.g., H, K, or R), Q at position 458 (e.g., R or T), Q at position 459 (H), and/or T at position 460 (N or S), numbered according to SEQ ID NO: 138.
152. The AAV particle of any one of embodiments 147-151, wherein the AAV capsid variant further comprises an R at position 456, numbered according to SEQ ID NO: 138.
153. The AAV particle of any one of embodiments 147-151, wherein the .AAV capsid variant further comprises an L at position 456, numbered according to SEQ ID NO: 138.
154. The AAV particle of any one of embodiments 147-153, wherein the AAV capsid variant further comprises an H at position 457 and an R at position 458, numbered according to SEQ ID NO: 138.
155. The AAV particle of any one of embodiments 147-153, wherein the AAV capsid variant further comprises a K at position 457 and an N at position 460, numbered according to SEQ ID NO: 138.
156. The AAV particle of any one of embodiments 147-153, wherein the AAV capsid variant further comprises a T at position 458, an H at position 459, and an S at position 460, numbered according to SEQ ID NO: 138.
157. The AAV particle of any one of embodiments 147-151, wherein the AAV capsid variant further comprises an R at position 456, an R at position 457, and an R at position 458, numbered according to SEQ ID NO: 138.
158. The AAV particle of any one of embodiments 147-157, wherein [A] [B | is present in loop IV.
159. The AAV particle of any one of embodiments 147-158, wherein [A] is present immediately subsequent to position 449, numbered according to SEQ ID NO: 138.
160. The AAV particle of any one of embodiments 147-159, wherein [A] replaces positions 450-453 (e.g., T450, 1451, N452, G453), numbered according to SEQ ID NO: 138.
161. The AAV particle of any one of embodiments 147-160, wherein [A] is present immediately subsequent to position 449, and wherein [A] replaces positions 450-453 (e.g., T450, 1451, N452, G453), numbered according to SEQ ID NO: 138.
162. The AAV particle of any one of embodiments 147-161, wherein [A][B] replaces positions 450-455 (e.g., T450, 1451, N452, G453, S454, G455), numbered according to SEQ ID NO: 138.
163. The AAV particle of any one of embodiments 147-162, wherein [A][B] is present immediately subsequent to position 449, and wherein [A][B] replaces positions 450-455 (e.g., T450, 1451, N452, G453, S454, G455), numbered according to SEQ ID NO: 138.
164. The AAV particle of any one of embodiments 147-163, wherein [B] is present immediately subsequent [A], and replaces positions 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
165. The .AAV particle of any one of embodiments 147-164, wherein [B] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 4, 36, 981, or 982.
166. The AAV particle of any one of embodiments 147-165, wherein [B] is present immediately subsequent to [A],
167. The AAV particle of any one of embodiments 147-166, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [A] [B] .
168. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a. viral genome comprising a nucleic acid encoding a modulator}' polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]-[N3] (SEQ ID NO: 6407), wherein:
(i) [N1] comprises X1, X2, and X3, wherein X2 is S and X3 is G;
(ii) [N2] comprises the amino acid sequence SPH; and
(iii) [N3] comprises X4, X5, and X6, wherein X5 is K.
169. The AAV particle of embodiment 168, wherein:
(i) X4 of [N3] is S, T. N, or A: and.
(ii) X5 of [N3] is A, V, T, S, G, R, L, or N: optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid, amino acids in (i) or (ii).
170. The AAV particle of embodiment 168 or 169, wherein X4 is S and/or X5 is A.
171. The AA V particle of any one of embodiments 168-170, wherein [N3] comprises SK, TK, NK, AK, KA, KV, KT, KS, KG, KR, KL, or KN.
172. The AAV particle of any one of embodiments 168-171, wherein [N3] is or comprises SKA, SKV, SKT, SKS, SKG, SKR, TKA, NKA, SKL, SKN, or AKA.
173. The AAV particle of any one of embodiments 168-172, wherein [N3] is or comprises SKA.
174. The AAV particle of any one of embodiments 168-173, wherein [N2]-[N3] comprises SPHSK (SEQ ID NO: 4701), SPHTK (SEQ ID NO: 4725), SPHNK (SEQ ID NO: 4726), or SPHAK (SEQ ID NO: 4727).
175. The AAV particle of any one of embodiments 168-174, wherein [N2]-[N3] is or comprises:
(i) SPHSKA (SEQ ID NO: 941), SPHSKV (SEQ ID NO: 4737), SPHSKT (SEQ ID NO: 4731 ), SPHSKS (SEQ ID NO: 962), SPHSKG (SEQ ID NO: 4732), SPHSKR (SEQ ID NO: 978), SPHTKA (SEQ ID NO: 4739), SPHNKA (SEQ ID NO: 4734), SPHSKL (SEQ ID NO: 960), SPHSKN (SEQ ID NO: 4735), or SPHAKA (SEQ ID NO: 4736):
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
176. The AA V particle of any one of embodiments 168-175, wherein [N2]-[N3] is or comprises SPHSKA (SEQ ID NO: 941).
177. The AAV particle of any one of embodiments 168-176, wherein the AAV capsid variant comprises an amino acid other than G at position 453 (e.g., M, T, I, E, S, A, N, V, L, K, H P, R, W, or D), numbered according to SEQ ID NO: 138 or 981.
178. The AAV particle of any one of embodiments 168-177, wherein the AAV capsid variant comprises the amino acid G at position 453, numbered according to SEQ ID NO: 138 or 981.
179. The AAV particle of any one of embodiments 168-178, wherein X1 of [N1] is G, M, T, I, E, S, A, N, V, L, K, H, P, R, W, or D; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids.
180. The AAV particle of any one of embodiments 168-179, wherein [N1] comprises SG, GS, MS, TS, IS, ES, SS, AS, NS, VS, LS, KS, HS, PS, RS, WS, or DS.
181. The .AAV particle of any one of embodiments 168-180, wherein |N1 ] is or comprises:
GSG, MSG, TSG, ISG, ESG, SSG, ASG, NSG, VSG, LSG, KSG, HSG, PSG, RSG, WSG, or DSG.
182. lire AAV particle of any one of embodiments 168-181, wherein [Ml] is or comprises GSG.
183. The .AAV particle of any one of embodiments 168-182, wherein |N1]-[N2| comprises SGSPH (SEQ ID NO: 4752).
184. The AAV particle of any one of embodiments 168-183, wherein [N1]-[N2] is or comprises:
(i) GSGSPH (SEQ ID NO: 4695), MSGSPH (SEQ ID NO: 4798), TSGSPH (SEQ ID NO: 4800), ISGSPH (SEQ ID NO: 4801 ), ESGSPH (SEQ ID NO: 4803), SSGSPH (SEQ ID NO: 4804), ASGSPH (SEQ ID NO: 4806), NSGSPH (SEQ ID NO: 4807), VSGSPH (SEQ ID NO: 4786), ESGSPH (SEQ ID NO: 4808). KSGSPH (SEQ ID NO: 4810), HSGSPH (SEQ ID NO: 4811), PSGSPH (SEQ ID NO: 4813), RSGSPH (SEQ ID NO: 4815), WSGSPH (SEQ ID NO: 4817), DSGSPH (SEQ ID NO: 4818):
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
185. The AAV particle of any one of embodiments 168-184, wherein [N1]-[N2]-[N3] is or comprises:
(i) GSGSPHSKA (SEQ ID NO; 4697), GSGSPHSKV (SEQ ID NO; 4956), MSGSPHSKA (SEQ ID NO; 4957), TSGSPHSKA (SEQ ID NO: 4959), ISGSPHSKA (SEQ ID NO: 4960), GSGSPHSKT (SEQ ID NO: 4962), ESGSPHSKA (SEQ ID NO: 4963), SSGSPHSKA (SEQ ID NO: 4964), GSGSPHSKS (SEQ ID NO: 4953), ASGSPHSKA (SEQ ID NO: 4966), NSGSPHSKA (SEQ ID NO: 4967), VSGSPHSKA (SEQ ID NO: 4913), LSGSPHSKA (SEQ ID NO; 4968), KSGSPHSKA (SEQ ID NO: 4970), GSGSPHSKG (SEQ
ID NO: 4972), GSGSPHSKR (SEQ ID NO: 4945), HSGSPHSKA (SEQ ID NO: 4973), PSGSPHSKA (SEQ ID NO: 4975), RSGSPHSKA (SEQ ID NO: 4977), GSGSPHTKA (SEQ ID NO: 4978), WSGSPHSKA (SEQ ID NO: 4980), DSGSPHSKA (SEQ ID NO: 4981), GSGSPHNKA (SEQ ID NO: 4983), GSGSPHSKL (SEQ ID NO: 4943), GSGSPHSKN (SEQ ID NO: 4994), or GSGSPHAKA (SEQ ID NO: 4995):
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
186. The AAV particle of any one of embodiments 168-185, wherein [N1]-[N2]-[N3] is or composes GSGSPHSKA (SEQ ID NO: 4697).
187. The AAV capsid variant of any one of embodiments 168-186, which comprises an amino acid other than Q at position 456 (e.g., R, P, H, L, K, I, G, S, M, or E), an amino acid other than N at position 457 (e.g., D, V, S, P, T, G, ¥, W, E, R, H, K, F, A, I, L, or M), an amino acid other than Q at position 458 (e.g., R, L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, or D), an amino acid other than Q at position 459 (e.g., H, K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G), and/or an amino acid other than T at position 460 (e.g., I, N, S, H, R, L, D, Y, A, or Q), numbered according to SEQ ID NO: 138.
188. The AAV particle of any one of embodiments 168-187, wherein the AAV capsid variant comprises an amino acid other than Q at position 462 (e.g., R, P, H, L, K, I, G, S, M, or E), an amino acid other than N at position 463 (e.g., D, V, S, P, T, G, Y, W, E, R, H, K, F, A, I, L, or M), an amino acid other than Q at position 464 (e.g., R, L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, or D), an amino acid other than Q at. position 465 (e.g,, H, K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G), and/or an amino acid other than T at position 466 (e.g., I, N, S, FI, R, L, D, Y, A, or Q), numbered according to SEQ ID NO: 981 .
189. The AAV particle of any one of embodiments 168-188, wherein the AAV capsid variant comprises the amino acid Q at position 456, the amino acid N at. position 457, the amino acid
Q at position 458, the amino acid Q at position 459, and/or the amino acid T at position 460, numbered according to SEQ ID NO: 138.
190. The AAV particle of any one of embodiments 168-189, wherein the AAV capsid variant comprises the amino acid Q at position 462, the amino acid N at position 463, the amino acid Q at position 464, the amino acid Q at position 465, and/or the amino acid T at position 466, numbered according to SEQ ID NO: 981.
191. The .AAV particle of any one of embodiments 168-190, wherein the AAV capsid variant further comprises [N4] wherein [N4] comprises X7, X8, X9, X10, and. X11, wherein:
(a) X7 is Q, R, P, II, L, K, I, G, S, M, or E;
(b) X8 is N, D, V, S, P, T, G, Y, W, E, R, H, K, F, A, I, L, or M;
(c) X9 is Q, R. L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, D;
(d) X10 is Q, H, K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G; and.
(e) X11 is T, I, N, S, II, R L, D, Y, A, Q; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(e).
192. The AAV particle of embodiment 191, wherein [N4] is or comprises:
(i) QNQQT (SEQ ID NO: 5412), QNRHT (SEQ ID NO: 5413), RDQQT (SEQ ID NO: 5414), PNLQT (SEQ ID NO: 5415), HVRQT (SEQ ID NO: 5416), PNQHT (SEQ ID NO: 5417), QSQQT (SEQ ID NO: 5418), QNQQI (SEQ ID NO: 5419), QPAKT (SEQ ID NO: 5420), QTQQN (SEQ ID NO: 5421), QNLAT (SEQ ID NO: 5422), QNQLT (SEQ ID NO: 5423), QGQQT (SEQ ID NO: 5424), LNRQS (SEQ ID NO: 5425), HNQQT (SEQ ID NO: 5426), QNPPT (SEQ ID NO: 5427), QNLQT (SEQ ID NO: 5428), QYQQT (SEQ ID NO: 5429), QDQET (SEQ ID NO: 5430), QNIIQT (SEQ ID NO: 5431), QDQQT (SEQ ID NO: 5432), HWQQT (SEQ ID NO: 5433). PNQQT (SEQ ID NO: 5434), QNQLI (SEQ ID NO: 5435), PEQQT (SEQ ID NO: 5436), QRTMT (SEQ ID NO: 5437), QNQQH (SEQ ID NO: 5438), LFIQIIT (SEQ ID NO: 5439), QHRIT (SEQ ID NO: 5440), QYIHT (SEQ ID NO: 5441), QKFET (SEQ ID NO: 5442), QFPST (SEQ ID NO: 5443), HNQQR (SEQ ID NO: 5444), QAIKT (SEQ ID NO: 5445). QNRQT (SEQ ID NO: 5446), QYQHT (SEQ ID NO: 5447), QN.PQS (SEQ ID NO: 5448), QHQLT (SEQ ID NO: 5449), QSPPT (SEQ ID NO: 5450), QAKLT (SEQ ID NO: 5451), KSQQT (SEQ ID NO: 5452), QDRPT (SEQ ID NO: 5453), QSQQL (SEQ ID NO: 5454), QAFHT (SEQ ID NO: 5455), QKQQD (SEQ ID
NO: 5456), QNAQT (SEQ ID NO: 5457), HNQLT (SEQ ID NO: 5458), QNQQY (SEQ ID NO: 5459), QKLNT (SEQ ID NO: 5460), QNVQT (SEQ ID NO: 5461), QAQQT (SEQ ID NO: 5462), QNLQA (SEQ ID NO: 5463), QTPPT (SEQ ID NO: 5464), QYQHA (SEQ ID NO: 5465), QGQQA (SEQ ID NO: 5466), QPPAT (SEQ ID NO: 5467), QERPT (SEQ ID NO: 5468), QDLQT (SEQ ID NO: 5469), QAMHT (SEQ ID NO: 5470), LNQQT (SEQ ID NO: 5471), QHPST (SEQ ID NO: 5472), PGLQT (SEQ ID NO: 5473), QGIRT (SEQ ID NO: 5474), QAPAT (SEQ ID NO: 5475), QSQQI (SEQ ID NO: 5476), QIPPT (SEQ ID NO: 5477), QTQLT (SEQ ID NO: 5478), QAPST (SEQ ID NO: 5479), QNTYA (SEQ ID NO: 5480), QNQHI (SEQ ID NO: 5481), QNHLT (SEQ ID NO: 5482), QIGMT (SEQ ID NO: 5483), LNKQT (SEQ ID NO: 5484), QLQQT (SEQ ID NO: 5485), QRMST (SEQ ID NO: 5486), QGILT (SEQ ID NO: 5487), QDRQT (SEQ ID NO: 5488), RDWQT (SEQ ID NO: 5489), QNTHD (SEQ ID NO: 5490), PNLQI (SEQ ID NO: 5491), QERST (SEQ ID NO: 5492). QNYQT (SEQ ID NO: 5493), QRTCT (SEQ ID NO: 5494), QIGHT (SEQ ID NO: 5495), QGAIT (SEQ ID NO: 5496), QVPPT (SEQ ID NO: 5497), QVQQI (SEQ ID NO: 5498), LMRQT (SEQ ID NO: 5499), QYSVT (SEQ ID NO: 5500), QAITT (SEQ ID NO: 5501), QKTLT (SEQ ID NO: 5502), QNQWT (SEQ ID NO: 5503), QLHHT (SEQ ID NO: 5504), QNIII (SEQ ID NO: 5505), QGHHT (SEQ ID NO: 5506), QSKVT (SEQ ID NO: 5507), QLPST (SEQ ID NO: 5508), IGKQT (SEQ ID NO: 5509), QAIHT (SEQ ID NO: 5510), QHGLT (SEQ ID NO: 5511), QFMCT (SEQ ID NO: 5512), QHLQT (SEQ ID NO: 5513). QNHQN (SEQ ID NO: 5514), QPART (SEQ ID NO: 5515), QSLQT (SEQ ID NO: 5516), QSQLT (SEQ ID NO: 5517), QDRQS (SEQ ID NO: 5518), QMPST (SEQ ID NO: 5519), QGSLT (SEQ ID NO: 5520), QVPAT (SEQ ID NO: 5521), QDKQT (SEQ ID NO: 5522), I1YQQT (SEQ ID NO: 5523), QVPST (SEQ ID NO: 5524), RGEQT (SEQ ID NO: 5525), PGQQT (SEQ ID NO: 5526). QSLQI (SEQ ID NO: 5527), LEQQT (SEQ ID NO: 5528), QNQST (SEQ ID NO: 5529), QKVIT (SEQ ID NO: 5530), QNNDQ (SEQ ID NO: 5531), QSVHT (SEQ ID NO: 5532), QPLGT (SEQ ID NO: 5533), IINQET (SEQ ID NO: 5534). QNLQI (SEQ ID NO: 5535), QIQQT (SEQ ID NO: 5536), QVRNT (SEQ ID NO: 5537), PSNQT (SEQ ID NO: 5538), QVGHT (SEQ ID NO: 5539), QRDIT (SEQ ID NO: 5540), QMPNT (SEQ ID NO: 5541), RGEQT (SEQ ID NO: 5542), QKQQT (SEQ ID NO: 5543), PSLQT (SEQ ID NO: 5544), QRDQT (SEQ ID NO: 5545), QAKGT (SEQ ID NO: 5546), QSAHT (SEQ ID NO: 5547). QSTMT (SEQ ID NO: 5548), QREMT (SEQ ID NO: 5549), QYRAT (SEQ ID NO: 5550), QWQQT (SEQ ID NO: 5551), QRMNT (SEQ ID NO: 5552), GDSQT (SEQ ID NO: 5553), QKIST (SEQ ID NO: 5554), PSMQT (SEQ ID NO: 5555), SPRQT (SEQ ID NO: 5556), MEQQT (SEQ ID NO: 5557), QYQNT (SEQ ID NO:
5558), QIIQQT (SEQ ID NO; 5559), INQQT (SEQ ID NO: 5560), PNQQH (SEQ ID NO: 5561), ENRQT (SEQ ID NO; 5562), QTQQA (SEQ ID NO: 5563), or QNQAT (SEQ ID NO: 5564);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i). e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
193. The AAV particle of embodiment 191 or 192, wherein [N1]-[N2]-[N3]-[N4] is or comprises:
(i) the amino acid sequence of any of SEQ ID NOs: 2.00 or 2887-3076;
(ii) an amino acid sequence comprising any portion of an amino acid, sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , 12, or 13 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
194. The AAV particle of any one of embodiments 191 -193, wherein [N1]-[N2]-[N3]-[N4] is or composes GSGSPHSKAQNQQT (SEQ ID NO: 200).
195. The AAV particle of any one of embodiments 191-193, wherein [N1]-[N2]-[N3]-[N4] is or comprises VSGSPHSKAQNQQT (SEQ ID NO: 903).
196. The AAV particle of any one of embodiments 168-195, wherein the AAV capsid variant comprises an amino acid other than K at position 449 (e.g., T, E, or N), T at position 450 (e.g., S, E, A, N, V, Q, or G), an amino acid other than I at position 451 (e.g., F, E, V, L, D, S, C, T, A, N, H, R, G, or W), and/or an amino acid other than N at position 452 (e.g., I, P, K, R, H, S, M, Q, I), T, L, A, Y, V, F, E, W, or G), numbered, according to SEQ ID NO: 138 or 981.
197. The AAV particle of any one of embodiments 168-196, wherein the AAV capsid variant comprises the amino acid K at position 449, the amino acid T at position 450, the amino acid I at position 451, and/or the amino acid N at position 452, numbered according to SEQ ID NO: 138 or 981.
198. The AAV particle of any one of embodiments 168-197, wherein the AAV capsid variant further composes [N0], wherein [N0] comprises XA, XB, XC, and XD, wherein:
(a) XA is K, T, E, or N;
(b) XB IS T, S, E, A, N, V, Q, or G.
(c) XC is I, F, E, V, L. D. S. C, T, A. N. H. R, G, or W; and
(d) XD is N, I, P, K, R, H, S, M, Q, D, T, L, A, ¥, V, F, E, W, or G; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids m (a)-(d).
199. The AAV particle of embodiment 198, wherein [N0] is or comprises:
(i) KTII (SEQ ID NO: 5565), KTFP (SEQ ID NO: 5566), KTEK (SEQ ID NO: 5567), KTVN (SEQ ID NO: 5568), KTFN (SEQ ID NO: 5569), KT1N (SEQ ID NO: 5570), TTIN (SEQ ID NO: 5571), KSIN (SEQ ID NO: 5572), KTER (SEQ ID NO: 5573), KELH (SEQ ID NO: 5574), KAIN (SEQ ID NO: 5575), KTDN (SEQ ID NO: 5576), KTFH (SEQ ID NO: 5577). KTSN (SEQ ID NO: 5578), ETIN (SEQ ID NO: 5579), NTIN (SEQ ID NO: 5580). KTEN (SEQ ID NO: 5581), KTSS (SEQ ID NO: 5582), KTCN (SEQ ID NO: 5583), KTEH (SEQ ID NO: 5584), KAEM (SEQ ID NO: 5585), KATN (SEQ ID NO: 5586), KAIK (SEQ ID NO: 5587), KTDK (SEQ ID NO: 5588), KTFK (SEQ ID NO: 5589), KSDQ (SEQ ID NO: 5590), KTEI (SEQ ID NO: 5591), KTID (SEQ ID NO: 5592), KNTN (SEQ ID NO: 5593), KTET (SEQ ID NO: 5594), KIEL (SEQ ID NO: 5595), KNTN (SEQ ID NO: 5596), KTEA (SEQ ID NO: 5597), KT AN (SEQ ID NO: 5598), NTIY (SEQ ID NO: 5599), KTFS (SEQ ID NO: 5600), KTES (SEQ ID NO: 5601), KTT'N (SEQ ID NO: 5602), KTED (SEQ ID NO: 5603), KTNN (SEQ ID NO: 5604), KEVH (SEQ ID NO: 5605), KTIS (SEQ ID NO: 5606), KTVR (SEQ ID NO: 5607), KTDR (SEQ ID NO: 5608), ETIK (SEQ ID NO: 5609), KNHI (SEQ ID NO: 5610), KESD (SEQ ID NO: 5611), KTIK (SEQ ID NO: 5612), KTDL (SEQ ID NO: 5613), KTVP (SEQ ID NO: 5614), KTVI (SEQ ID NO: 5615), KAEH (SEQ ID NO: 5616), KNCL (SEQ ID NO: 5617), KTVK (SEQ ID NO: 5618), KNAD (SEQ ID NO: 5619), KTIT (SEQ ID NO: 5620), KNCV (SEQ ID NO: 5621), KNAL (SEQ ID NO: 5622), KVIN (SEQ ID NO: 5623), KTEF (SEQ ID NO: 5624), KTRE (SEQ ID NO: 5625),
KQGE (SEQ ID NO: 5626), KSEK (SEQ ID NO: 5627), KNVN (SEQ ID NO: 5628), KGGE (SEQ ID NO: 5629), KEFV (SEQ ID NO: 5630), KSDK (SEQ ID NO: 5631), KTEQ (SEQ ID NO: 5632), KEVQ (SEQ ID NO: 5633), KTEY (SEQ ID NO: 5634), KNCW (SEQ ID NO: 5635), KTDV (SEQ ID NO: 5636), KSDI (SEQ ID NO: 5637), KNSI (SEQ ID NO: 5638), KNSL (SEQ ID NO: 5639), KEVV (SEQ ID NO: 5640), KTEP (SEQ ID NO: 5641), KSEL (SEQ ID NO: 5642), KTWQ (SEQ ID NO: 5643), KIEV (SEQ ID NO: 5644), KAVN (SEQ ID NO: 5645), KGVL (SEQ ID NO: 5646), KTEG (SEQ ID NO: 5647), KTRD (SEQ ID NO: 5648), KTGN (SEQ ID NO: 5649), KNAI (SEQ ID NO: 5650), KAEN (SEQ ID NO: 5651), KAET (SEQ ID NO: 5652), KTVH (SEQ ID NO: 5653), KETA (SEQ ID NO: 5654), KNNL (SEQ ID NO: 5655), EAIN (SEQ ID NO: 5656), KSLN (SEQ ID NO: 5657), KTIP (SEQ ID NO: 5658), or KTIH (SEQ ID NO: 5659);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, or 3 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
200. The AAV particle of embodiment 198 or 199, wherein [N0]-[N1]-[N2]-[N3]-[N4] is or comprises:
(i) the amino acid sequence of any one of SEQ ID NOs: 3239-3526 or 3591-3605;
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, or 17 amino acids, e.g., consecutive amino acids, thereof;
(lii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
201. The AAV particle of any one of embodiments 198-200, wherein [N0]-[N1]-[N2]-[N3] [N4] is or comprises KTINGSGSPHSKAQNQQT (SEQ ID NO: 5660).
202. The AAV particle of any one of embodiments 198-200, wherein [N0]-[N1]-[N2]-[N3]- [N4] is or comprises KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589).
203. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]-[N3] (SEQ ID NO: 6408), wherein:
(i) [N1] comprises X1, X2, and X3, wherein X2 is an amino acid other than S and X3 is an amino acid other than G;
(ii) [N2] comprises the amino acid sequence SPH; and
(iii) [N3] comprises X4, X5, and X6, wherein X4 is K.
204. The AAV particle of embodiment 203, wherein:
(i) X5 of [N3] is S, I, T. R. H, Y, L, or M; and
(ii) X6 of [N3] is G, A, L, E, V, R, W, N, Q, or K; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (i) or (ii).
205. The AAV particle of embodiment 203 or 204, wherein X5 is S and/or X6 is G.
206. The AAV particle of any one of embodiments 203-205, wherein [N3] comprises KS, KI,
KT, KR, KH, KY, KL, KM, SG, IG, TG, RG, SA, SL, SE, SV, SR, SW, SN, IIG, YG, SQ, IV, SK, LW, MG, or MA.
207. The AAV particle of any one of embodiments 203-206, wherein [N3] is or comprises
KSG, KIG, KTG, KRG, KSA, KSL, KSE, KSV, KSR, KSW, KSN, KIIG, KYG, KSQ, KIV, KSK, KLW, KMG, or KMA.
208. The AAV particle of any one of embodiments 203-207, wherein [N3] is or comprises KSG.
209. The AA V particle of any one of embodiments 203-208, wherein [N2]-[N3] comprises SPHKS (SEQ ID NO: 4704), SPIIKI (SEQ ID NO: 4713), SPIIKT (SEQ ID NO: 471 1),
SPHKR (SEQ ID NO: 4717), NPHKS (SEQ ID NO: 5661), SPHKH (SEQ ID NO: 4728),
SPHKY (SEQ ID NO: 4715), SPHKL (SEQ ID NO: 4714), or SPHKM (SEQ ID NO: 4729).
210. The AAV particle of any one of embodiments 203-209, wherein [N2]-[N3] is or comprises:
(i) SPHKSG (SEQ ID NO: 946), SPHKIG (SEQ ID NO: 958), SPHKTG (SEQ ID NO: 4738), SPHKRG (SEQ ID NO: 974), NPHKSG (SEQ ID NO: 5662), SPHKSA (SEQ ID NO: 977), SPHKSL (SEQ ID NO: 4740), SPHKSE (SEQ ID NO: 4741), SPHKSV (SEQ ID NO: 4742), SPHKSR (SEQ ID NO: 951), SPHKSW (SEQ ID NO: 4743), SPHKSN (SEQ ID NO: 4744), SPHKHG (SEQ ID NO: 4745), SPHKYG (SEQ ID NO: 966), SPHKSQ (SEQ ID NO: 4746), SPHKIV (SEQ ID NO: 5663), SPHKSK (SEQ ID NO: 4747), SPHKLW (SEQ ID NO: 4748), SPHKMG (SEQ ID NO: 4750), or SPHKAIA (SEQ ID NO: 4751);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
211. The AAV particle of any one of embodiments 203-210, wherein [N2'|-[N3] is or composes SPHKSG (SEQ ID NO: 946).
212. The AAV particle of any one of embodiments 203-211, wherein the AAV capsid variant comprises an amino acid other than G at position 453 (e.g.. A, K, W, R, L, I, M, N, T, E, Q, Y, H, F, or V), numbered, according to SEQ ID NO: 138 or 981 .
213. The AAV particle of any one of embodiments 203-212, wherein the AAV capsid variant composes the amino acid G at position 453, numbered according to SEQ ID NO: 138 or 981.
214. The AAV particle of any one of embodiments 203-214, wherein:
(i) X1 of [N1] is G, A, K, W, R, L, I, M, N, T, E, Q, Y, H, F, or V;
(li) X2 of [N1] is H, Y, R . Q. N, P, or D;
(iii) X3 of [N1] is D, E, G, V, or N;
optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (i), (ii), or (iii).
215. The AAV particle of any one of embodiments 203-214, wherein X2 of [N1] is II and X3 of [N1] is D.
2.16. The AAV particle of any one of embodiments 203-215, wherein X1 of [N1] is G, X2. of [N1] is H and X3 of [N1] is D.
217. The AA V particle of any one of embodiments 203-216, wherein [N1] comprises GH, HD, GY, GR, GQ, AH, GN, KH, GP, WH, RH, LH, TH, MH, GD, NH, TH, EH, QH, YH, HH , F H , VH, YD, HE, RG, QI). RD, ND, PI). Q V. DD, HN, or NG.
2.18. The AAV capsid variant, of any one of embodiments 203-217, wherein [N1] is or comprises GHD, GYD, GHE, GRG, GQD, GRD, AHD, GND, KHD, GPD, WHD, RHD, LHD, GQV, IHD, MHD, GDD, GHN, NHD, THD, GNG, EHD, QHD, YHD, HHD, FHD, or VHD.
219. The AAV particle of any one of embodiments 203-218, wherein [N1] is or comprises GHD.
220. The AAV particle of any one of embodiments 203-219, wherein [N1]-[N2] comprises HDSPH (SEQ ID NO: 4703).
221. The AA V particle of any one of embodiments 203-22.0, wherein [N1]-[N2] is or composes:
(i) GHDSPH (SEQ ID NO: 4784). GYDSPH (SEQ ID NO: 4829), GHESPH (SEQ ID NO: 4793), GRGSPH (SEQ ID NO: 4788), GHDNPH (SEQ ID NO: 5664), GQDSPH (SEQ ID NO: 4785), GRDSPH (SEQ ID NO: 4831), AHDSPH (SEQ ID NO: 5665), GNDSPH (SEQ ID NO: 4832), KHDSPH (SEQ ID NO: 5666), GPDSPH (SEQ ID NO: 4833), WHDSPH (SEQ ID NO: 5667), RHDSPH (SEQ ID NO: 5668), LHDSPH (SEQ ID NO: 5669), GQVSPH (SEQ ID NO: 4835), IHDSPH (SEQ ID NO: 5670), MHDSPH (SEQ ID NO: 5671), GDDSPH (SEQ ID NO: 4792), GHNSPH (SEQ ID NO: 4836), NHDSPH (SEQ ID NO: 5672), THDSPH (SEQ ID NO: 5673), GNGSPH (SEQ ID NO: 4805), EHDSPH
(SEQ ID NO: 5674), QHDSPH (SEQ ID NO: 5675), YHDSPH (SEQ ID NO: 5676), HHDSPH (SEQ ID NO: 5677), FHDSPH (SEQ ID NO: 5678), or VHDSPH (SEQ ID NO: 5679);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i). e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an ami no acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
222. The AAV particle of any one of embodiments 203-221 , wherein [N1]-[N2]-[N3] is or comprises:
(i) GHDSPHKSG (SEQ ID NO: 4698), GHDSPHKIG (SEQ ID NO: 4996), GYDSPHKSG (SEQ ID NO: 4997), GHESPHKSG (SEQ ID NO: 4998), GHDSPHKTG (SEQ ID NO: 4999), GRGSPHKRG (SEQ ID NO: 5000), GHDNPHKSG (SEQ ID NO: 5680), GQDSPHKSG (SEQ ID NO: 4908), GHDSPHKSA (SEQ ID NO: 4940), GHDSPHKSL (SEQ ID NO: 5001), GHDSPHKSE (SEQ ID NO: 5003), GRDSPHKSG (SEQ ID NO: 5004), AHDSPHKSG (SEQ ID NO: 5681), GNDSPHKSV (SEQ ID NO: 5005), AHDSPHKIG (SEQ ID NO: 5682), GHESPHKSA (SEQ ID NO: 4939), GQDSPHKIG (SEQ ID NO: 5006), GHDSPHKSV (SEQ ID NO: 5007), GHDSPHKSR (SEQ ID NO: 4942), KHDSPHKSG (SEQ ID NO: 5683), GPDSPHKIG (SEQ ID NO: 5008), GPDSPHKSG (SEQ ID NO: 5009), GHDSPHKSW (SEQ ID NO: 5010), WHDSPHKSG (SEQ ID NO: 5684), RHDSPHKSG (SEQ ID NO: 5685), GHDSPHKSN (SEQ ID NO: 5011), GHDSPHKRG (SEQ ID NO: 4937). GHDSPHKHG (SEQ ID NO: 5013), EHDSPHKSG (SEQ ID NO: 5686), GQVSPHKSG (SEQ ID NO: 5014), IHDSPHKSG (SEQ ID NO: 5687), MHDSPHKSG (SEQ ID NO: 5688), GDDSPHKSV (SEQ ID NO: 5015). GHNSPHKSG (SEQ ID NO: 5016), NHDSPHKSG (SEQ ID NO: 5689), THDSPHKSG (SEQ ID NO: 5690), GNGSPHKRG (SEQ ID NO: 5017), EHDSPHKSG (SEQ ID NO: 5691), GIIDSPHKYG (SEQ ID NO: 5018), GHDSPHKSQ (SEQ ID NO: 5019), QHDSPHKSG (SEQ ID NO: 5692), RHDSPHKIV (SEQ ID NO: 5693), YHDSPHKSG (SEQ ID NO: 5694). GNDSPHKIG (SEQ ID NO: 5020), HHDSPHKSG (SEQ ID NO: 5695), GHDSPHKSK (SEQ ID NO: 5021), EHDSPHKSG (SEQ ID NO: 5696), GHDSPHKLW (SEQ ID NO: 5022), YHDSPHKSG (SEQ ID NO: 5697),
GIIDSPHKMG (SEQ ID NO: 5024), GHDSPHKMA (SEQ ID NO: 5025), or GDDSPHKSG (SEQ ID NO: 4938);
(li) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
223. The AAV particle of any one of embodiments 203-22.2, wherein [N1]-[N2]-[N3] is or comprises GIIDSPHKSG (SEQ ID NO: 4698).
224. The AAV particle of any one of embodiments 203-223, wherein the AAV capsid variant comprises an amino acid other than Q at position 456 (e.g., R, P, H, K, L, V, A, E, or I), an amino acid other than N at position 457 (e.g., I, K, S, H, R, T, D, Y, L, W, F, A, Q, or M), an amino acid other than Q at position 458 (e.g., R, V, K, P, Y, H, L, I, E, or M), an amino acid other than Q at position 459 (e.g., H, L, E, P, W, D, I, V, S, K, R, C, M, or N), and/or an amino acid other than T at position 460 (e.g.. A, E, K, S, I, P, G, or N), numbered according to SEQ ID NO: 138.
225. The AAV particle of any one of embodiments 203-224, wherein the AAV capsid variant comprises an amino acid other than Q at position 462 (e.g., R, P, II, K, L, V, A, E, or I), an amino acid other than N at position 463 (e.g., I, K, S, H, R, T, D, Y, L, W, F, A, Q, or M), an amino acid other than Q at position 464 (e.g., R, V, K, P, Y, H, L, I, E, or M), an amino acid other than Q at position 465 (e.g., H, L, E, P, W, D, I, V, S, K, R, C, M, or N), and/or an amino acid other than T at position 466 (e.g., A, E, K, S, I, P, G, or N), numbered according to SEQ ID NO: 982.
226. The AAV particle of any one of embodiments 203-225, wherein the AAV capsid variant comprises the amino acid Q at position 456, the amino acid N at position 457, the amino acid Q at position 458, the amino acid Q at position 459, and/or the amino acid T at position 460, numbered according to SEQ ID NO: 138.
227. The AAV particle of any one of embodiments 203-226, wherein the AAV capsid variant comprises the amino acid Q at position 462, the amino acid N at position 463, the amino acid Q at position 464, the amino acid Q at position 465, and/or the amino acid T at position 466, numbered according to SEQ ID NO; 138.
228. The AAV particle of any one of embodiments 203-227, wherein the AAV capsid variant further composes [N4], wherein [N4] comprises X7, X8, X9, X10, and X11, wherein:
(a) X7 is Q, R, P, H , L, K, I, G, S, M, or E;
(b) X8 is N, D, V, S, P, T, G, Y, W, E, R, H, K, F, A, 1, L, or M:
(c) X9 is Q, R, L, A, P, H, T, I, F, K, V, M, G, W, Y, S, E, N, D;
(d) X10 is Q, H , K, A, L, P, E, M, I, S, N, R, Y, C, V, T, W, D, G; and
(e) X11 is T, I, N, S, H, R, L, D, Y, A, Q; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid, amino acids in (a)-(e).
229. The AAV particle of embodiment 228, wherein [N4] is or comprises;
(i) QNQQT (SEQ ID NO: 5412), Q1RQT (SEQ ID NO: 5698), QNQHA (SEQ ID NO: 5699), QKQQT (SEQ ID NO: 5543), QSVQT (SEQ ID NO: 5700), RSQQT (SEQ ID NO: 5701), QNKLE (SEQ ID NO: 5702), QNQQK (SEQ ID NO: 5703), QHQQA (SEQ ID NO: 5704), QIQHT (SEQ ID NO: 5705), PRQQT (SEQ ID NO: 5706), HTQQT (SEQ ID NO: 5707), QRQHT (SEQ ID NO: 5708), QSQQT (SEQ ID NO: 5418), QNQQS (SEQ ID NO: 5709), RNQET (SEQ ID NO: 5710), QTQLT (SEQ ID NO: 5478), KNQQT (SEQ ID NO: 5711), QDQQT (SEQ ID NO: 5432), HNQQT (SEQ ID NO: 5426), QNQLT (SEQ ID NO: 5423), QTQQT (SEQ ID NO: 5712), QTQQI (SEQ ID NO: 5713), QSKQA (SEQ ID NO: 5714), QNQPP (SEQ ID NO: 5715), QSPQT (SEQ ID NO: 5716), QNYQT (SEQ ID NO: 5493), QNHQT (SEQ ID NO: 5431), QNRQT (SEQ ID NO: 5446), QNQQG (SEQ ID NO: 5717), QNHLT (SEQ ID NO: 5482). QYQHT (SEQ ID NO: 5447), QNQWT (SEQ ID NO: 5503), QNQHT (SEQ ID NO: 5718), QTRQT (SEQ ID NO: 5719), QNLHT (SEQ ID NO: 5720), LNQQT (SEQ ID NO: 5471), QNQET (SEQ ID NO: 5721), QHLQT (SEQ ID NO: 5513), LNQPT (SEQ ID NO: 5722), QNQDT (SEQ ID NO: 5723), RNQQT (SEQ ID NO: 5724), QNLLT (SEQ ID NO: 5725). QLVIT (SEQ ID NO: 5726), RTQET (SEQ ID NO: 5727), QTHQT (SEQ ID NO: 5728), QNQPA (SEQ ID NO: 5729), QDQHT (SEQ ID NO: 5730), QSQHT (SEQ ID NO: 5731), RNQQI (SEQ ID NO: 5732), VRQQT (SEQ ID NO: 5733), QNQHS (SEQ ID NO: 5734), AWQQT (SEQ ID NO: 5735), QSVPT (SEQ ID
NO: 5736), QNIQP (SEQ ID NO: 5737), QNHLN (SEQ ID NO: 5738), LDQQT (SEQ ID NO: 5739), PDQQS (SEQ ID NO: 5740), ESQQT (SEQ ID NO: 5741), QNKQT (SEQ ID NO: 5742), QRQLT (SEQ ID NO: 5743), QIIVT (SEQ ID NO: 5744), QKQST (SEQ ID NO: 5745), QSHQT (SEQ ID NO: 5746), QFVVT (SEQ ID NO: 5747), QNLQT (SEQ ID NO: 5428), QNQQI (SEQ ID NO: 5419), QSQPT (SEQ ID NO: 5748), QNEQT (SEQ ID NO: 5749). QSLQT (SEQ ID NO: 5516). RNRQT (SEQ ID NO: 5750), QSKQT (SEQ ID NO: 5751), QNPLT (SEQ ID NO: 5752), RDQKT (SEQ ID NO: 5753), HNQQN (SEQ ID NO: 5754), QWKRT (SEQ ID NO: 5755), QSQQI (SEQ ID NO: 5476), QAQQT (SEQ ID NO: 5462), QNHQI (SEQ ID NO: 5756), QNQQA (SEQ ID NO: 5757), QNQLN (SEQ ID NO: 5758), QTQPT (SEQ ID NO: 5759), INQQT (SEQ ID NO: 5560), QKQLT (SEQ ID NO: 5760), RNQLA (SEQ ID NO: 5761), RNQQS (SEQ ID NO: 5762), ISIQT (SEQ ID NO: 5763), QNQQN (SEQ ID NO: 5764), QSQQS (SEQ ID NO: 5765), QTVCT (SEQ ID NO: 5766). QYQQI (SEQ ID NO: 5767), QQIMT (SEQ ID NO: 5768), QNEQS (SEQ ID NO: 5769), LNHQT (SEQ ID NO: 5770), QMIHT (SEQ ID NO: 5771), RNHQS (SEQ ID NO: 5772), QKMNT (SEQ ID NO: 5773), QSQQN (SEQ ID NO: 5774), QYQIIA (SEQ ID NO: 5465);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g,, any 2, 3, or 4 amino acids, e.g,, consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in ( i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
230. The AAV particle of embodiment 228 or 229, wherein [N1]-[N2]-[N3]-[N4] is or comprises:
(i) the amino acid sequence of any of SEQ ID NOs: 201 or 3160-3237;
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, .10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
231. The AAV particle of any one of embodiments 228-230, wherein [N1]-[N2]-[N3]-[N4] is or comprises GHDSPHKSGQNQQT (SEQ ID NO; 201).
232. The AAV particle of any one of embodiments 203-231, wherein the AAV capsid variant comprises an amino acid other than K at position 449 (e.g., T), T at position 450 (e.g.. A, S, I, V, N, E, Y, C, G, W, or Q), an amino acid other than I at position 451 (e.g., E, V, S, T, N, D, C, G, Q, L, P, A), and/or an amino acid other than N at position 452 (e.g., S, Y, I, K, F, T, D, E, G, V, L, A, M, Q, II, P, or R), numbered according to SEQ ID NO: 138 or 982.
233. The AAV particle of any one of embodiments 203-232, wherein the AAV capsid variant comprises the amino acid K at position 449, the amino acid T at position 450, the amino acid 1 at position 45 I , and/or the amino acid N at position 452, numbered according to SEQ ID NO: 138 or 982.
234. The AAV particle of any one of embodiments 203-233, wherein the AAV capsid variant further comprises [N0], wherein [N0] comprises XA, XB, XC, and XD, wherein:
(a) XA is K or T;
(b) XB is T, A, S, I, V, N, E, Y, C, G, W, or Q;
(c) XC is I, E, V, S, T, N, D, C, G, Q, L, P, A; and
(d) XD IS N, S, Y, I, K, F, T, D, E, G, V. L, A, M, Q, H, P, or R; optionally wherein the AAV capsid, variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(d).
235. The AAV particle of embodiment 234, wherein [N0] is or comprises:
(i) KAIN (SEQ ID NO: 5575), KTIN (SEQ ID NO: 5570), KTES (SEQ ID NO: 5601), TTIN (SEQ ID NO: 5571), KSIN (SEQ ID NO: 5572), KTVN (SEQ ID NO: 5568), KSIY (SEQ ID NO: 5775), KTSN (SEQ ID NO: 5578). KTTN (SEQ ID NO: 5602), KIIN (SEQ ID NO: 5776), KITS (SEQ ID NO: 5606), KA.II (SEQ ID NO: 5777), KTIK (SEQ ID NO: 5612), KTEF (SEQ ID NO: 5624), KITT (SEQ ID NO: 5620), KTNN (SEQ ID NO: 5604), KTID (SEQ ID NO: 5592), KAIS (SEQ ID NO: 5778), KTVD (SEQ ID NO: 5779), KTIE (SEQ ID NO: 5780), KTEG (SEQ ID NO: 5647), KVIN (SEQ ID NO: 5623), KAVN (SEQ ID NO: 5645), KTIY (SEQ ID NO: 5781), KTDN (SEQ ID NO: 5576), KTCN (SEQ ID NO: 5583), KNVV (SEQ ID NO: 5782), KTEL (SEQ ID NO: 5595), KTDA (SEQ ID NO: 5783), KIEV (SEQ ID NO: 5644), KSEL (SEQ ID NO: 5642), KTEM (SEQ ID NO:
5784), KTEQ (SEQ ID NO: 5632), KTII (SEQ ID NO: 5565), KIVN (SEQ ID NO: 5785), KTEK (SEQ ID NO: 5567), KTEN (SEQ ID NO: 5581), KIGN (SEQ ID NO: 5786), KEVM (SEQ ID NO: 5787), KYQV (SEQ ID NO: 5788), KTEA (SEQ ID NO: 5597), KATN (SEQ ID NO: 5586), KTEH (SEQ ID NO: 5584), KTVE (SEQ ID NO: 5789), KAID (SEQ ID NO: 5790), KTIM (SEQ ID NO: 5791), KEVG (SEQ ID NO: 5792), KSEM (SEQ ID NO: 5793), KAQQ (SEQ ID NO: 5794), KCGE (SEQ ID NO: 5795), KASN (SEQ ID NO: 5796). KTET (SEQ ID NO: 5594), KTIG (SEQ ID NO: 5797), KTDP (SEQ ID NO: 5798), KELV (SEQ ID NO: 5799), KELM (SEQ ID NO: 5800), KNEI (SEQ ID NO: 5801), KTPN (SEQ ID NO: 5802), KITN (SEQ ID NO: 5803), KTDI (SEQ ID NO: 5804), KTDQ (SEQ ID NO: 5805), KGIN (SEQ ID NO: 5806), KSEI (SEQ ID NO: 5807), KSEK (SEQ ID NO: 5627), KWSA (SEQ ID NO: 5808), KELA (SEQ ID NO: 5809), KQTQ (SEQ ID NO: 5810), KGAD (SEQ ID NO: 5811), KVGE (SEQ ID NO: 5812), KANE (SEQ ID NO: 5813), KTDT (SEQ ID NO: 5814), KTCI (SEQ ID NO: 5815). KELR (SEQ ID NO: 5816), KCQI (SEQ ID NO: 5817), KGVM (SEQ ID NO: 5818), KA CD (SEQ ID NO: 5819), KNEE (SEQ ID NO: 5820), KAAE (SEQ ID NO: 5821), KGQN (SEQ ID NO: 5822), KNEE (SEQ ID NO: 5823), KTSI (SEQ ID NO: 5824), KAEH (SEQ ID NO: 5616), KCDQ (SEQ ID NO: 5825), KEIL (SEQ ID NO: 5826), KIER (SEQ ID NO: 5573), KNAI (SEQ ID NO: 5650), KTDK (SEQ ID NO: 5588), KTPD (SEQ ID NO: 5827), KTIH (SEQ ID NO: 5659), or KTEI (SEQ ID NO: 5591);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, or 3 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
236. The AAV particle of embodiment 234or 235, wherein [N0]-[N1]-[N2]-[N3]-[N4] is or comprises:
(i) the amino acid sequence of any one of SEQ ID NOs: 3606-3836;
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, or 17 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
237. The AAV particle of any one of embodiments 234-236, wherein [N0]-[N1]-[N2]-[N3]- [N4] is or comprises KTINGHDSPFIKSGQNQQT (SEQ ID NO: 5828).
2.38. The AAV particle of any one of embodiments 234-236, wherein [N0]-[N1]-[N2]-[N3]- [N4] is or comprises KAEIGHDSPHKSGQNQQT (SEQ ID NO: 1754).
239. The AA V particle of any one of embodiments 234-236, wherein [N0]-[N1]-[N2]-[N3]- [N4] is or comprises KTEKMSGSPHSKAQNQQT (SEQ ID NO: 3241).
240. The AAV particle of any one of embodiments 168-239, wherein [N1]-[N2]-[N3] is present in loop IV, e.g., numbered according to SEQ ID NO: 4, 36, 138, 981 , or 982,
241. The AAV particle of any one of embodiments 198-202 or 234-240, wherein [N0] and [N4] are present in loop IV, e.g., numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
242. The AAV particle of any one of embodiments 198-202 or 234-241, wherein [N0] is present immediately subsequent to position 448, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
243. The AAV particle of any one of embodiments 198-202 or 234-242, wherein [N0] replaces positions 449-452 (e.g., K449, T450, 1451, and N452), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
244. The AAV particle of any one of embodiments 198-202 or 234-243, wherein [N0] is present immediately subsequent to position 448 and wherein [N0] replaces positions 449-452 (e.g., K449, T450, 1451, and N452), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
245. The AAV particle of any one of embodiments 198-202 or 234-244, wherein [N0] corresponds to positions 449-452 (e.g., K449, T450, 1451 , and N452) of any one of SEQ ID NOs: 4, 36, 138, 981, or 982.
246. The AAV particle of any one of embodiments 168-245, wherein [N1] is present immediately subsequent to position 452, numbered according to SEQ ID NO: 4, 36, 138, 981 , or 982.
247. The AAV particle of any one of embodiments 168-246, wherein [N1] replaces positions 453-455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
248. The AAV particle of any one of embodiments 168-246, wherein [N1] replaces position
453 (e.g., G453), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
249. The AAV particle of any one of embodiments 168-177, 179-181, 183-185, 187-193, 195-200, 202, or 240-246, wherein:
(i) X1 of [N1] replaces position 453 (e.g., G453);
(ii) X2 of [N1] corresponds to position 454 (e.g., S454); and
(iii) X3 of [N1] corresponds to position 455 (e.g., G455), wherein (i), (ii), and (iii) are numbered according to SEQ ID NO: 138 or SEQ ID NO:
981.
250. The AAV particle of any one of embodiments 168-176, 178-201, or 240-246, wherein :
(i) X1 of [N1] corresponds to position 453 (e.g., G453);
(ii) X2 of [N1] corresponds to position 454 (e.g., S454); and
(iii) X3 of [N1] corresponds to position 455 (e.g., G455); wherein (i), (ii), and (iii) are numbered according to SEQ ID NO: 138 or SEQ ID NO:
981.
251. The AAV particle of any one of embodiments 203-248, wherein:
(i) X1 of [N1] corresponds to position 453 (e.g., G453);
(ii) X2 of [N1] replaces position 454 (e.g., S454); and
(iii) X3 of [N1] replaces position 455 (e.g., G455), wherein (i), (ii), and (iii) are numbered according to SEQ ID NO: 138 or SEQ ID NO:
982.
252. The AAV particle of any one of embodiments 203-248 or 251, wherein [N1] corresponds to positions 453-455 (e.g., G453, H454, D455) of SEQ ID NO 982.
253. The AAV particle of any one of embodiments 168-176, 178-201, 240-247, or 250, wherein [N1] corresponds to positions 453-455 (e.g., G453, S454, G455) of SEQ ID NO: 138 or 981.
254. The AAV particle of any one of embodiments 168-253, wherein [N2] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982.
255. The AAV particle of any one of embodiments 168-254, wherein [M2] corresponds to positions 456-458 (e.g., S456, P457, and H458) of SEQ ID NO: 981 or 982.
256. The AAV particle of any one of embodiments 168-254, wherein [N2] corresponds to positions 456-458 (e.g., S456, P457, and H458) of any one of SEQ ID NOs: 4 or 36-59.
257. The AA V particle of any one of embodiments 168-256, wherein [N2] is present immediately subsequent to [N1].
258. The AAV particle of any one of embodiments 168-202, 240-247, or 249-257, wherein [N3] corresponds to positions 459-460 (e.g., S459, K460, A461) of SEQ ID NO: 981 .
259. The AAV particle of any one of embodiments 168-202, 240-247, or 249-257, wherein [N3] corresponds to positions 459-460 (e.g., S459, K460, A461) of SEQ ID NO: 36, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51 , 52, 53, 54, 55, 57, or 59.
260. The AAV particle of any one of embodiments 168-259, wherein [N2]-[N3] is present immediately subsequent to position 455, numbered according to any one of SEQ ID NOs: 4, 36, 138, 981, or 982.
261. The AA V particle of any one of embodiments 168-259, wherein [N2]-[N3] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 981.
262. The AAV particle of any one of embodiments 168-261, wherein [N2]-[N3] corresponds to positions 456-461 (e.g., S456, P457, H458, S459, K460, A461) of SEQ ID NO: 981.
263. The AAV particle of any one of embodiments 168-262, wherein [N2]-[N3] corresponds to positions 456-461 (e.g., S456, P457, H458, S459, K460, A461) of any one of SEQ ID NOs: 36, 38, 39. 40, 41, 42, 43, 44, 45, 46. 47, 48, 49, 50, 51, 52, 53. 54, 55, 57, or 59.
264. The AAV particle of any one of embodiments 203-257 or 259-261, wherein [N3] corresponds to positions 459-460 (e.g., K459, S460, G461) of SEQ ID NO: 982.
265. The AAV particle of any one of embodiments 203-257 or 259-261, wherein [N3] corresponds to positions 459-460 (e.g., K459, S460, G461) of SEQ ID NO: 37.
2.66. The AAV particle of any one of embodiments 203-265, wherein [N2.]-[N3] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 982.
267. The AAV particle of any one of embodiments 203-246, 248, 252, 255, 2.57, 260, 263
266, wherein [N3] replaces positions 454 and. 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
2.68. The AAV particle of any one of embodiments 203-246, 2.48, 252, 255, 257, 260, 263-
267, wherein [N3] is present immediately subsequent to [N2] and replaces positions 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
269. The AA V particle of any one of embodiments 203-246, 248, 252, 255, 2.57, 260, 263-
268, wherein [N3] is present immediately subsequent to [N1]-[N2] and replaces positions 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138.
270. The AAV particle of any one of embodiments 203-257, 260, 264-269, wherein [N2]- [N3] corresponds to positions 456-461 (e.g., S456, P457, H458, K459, S460, G461) of SEQ ID NO: 982.
271 . The AAV particle of any one of embodiments 203-257, 260, 264-270, wherein [N2]- [N3] corresponds to positions 456-461 (e.g., S456, P457, H458, K459, S460, G461) of SEQ ID NO: 37.
272. The AAV particle of any one of embodiments 191-202 or 228-271, wherein [N4] is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
273. The AAV particle of any one of embodiments 191-202 or 228-272, wherein [N4] replaces positions 456-460 (e.g., Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
274. The AAV particle of any one of embodiments 191-202 or 228-273, wherein [N4] corresponds to positions 462-466 (e.g., Q462, N463, Q464, Q465, and T466) of SEQ ID NO: 981 or 982.
275. The AAV particle of any one of embodiments 191-202 or 228-273, wherein [N4] corresponds to positions 462-466 of any one of SEQ ID NOs; 4 or 36-59.
276. The AAV particle of any one of embodiments 191-202 or 228-274, wherein [N4] corresponds to positions 456-460 (e.g., Q456, N457, Q458. Q459, and T460) of SEQ ID NO: 138.
277. The .AAV particle of any one of embodiments 191-202 or 228-276, wherein [N2]-[N3] [N4] replaces positions 456-460 (e.g., Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
278. The AAV particle of any one of embodiments 191-202 or 228-277, wherein [N2]-[N3j- [N4] is present immediately subsequent to position 455, and wherein [N2]-[N3]-[N4] replaces positions 456-460 (e.g., Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
279. The AA V particle of any one of embodiments 191-202 or 228-278, wherein [N1 ]-[N2]- [N3]-[N4] replaces positions 453-460 (e.g., G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
280. The AAV particle of any one of embodiments 191-202 or 228-279, wherein [N1]-[N2] [N3]-[N4] is present immediately subsequent to position 452, and wherein [N1]-[N2]-[N3]- [N4] replaces positions 453-460 (e.g., G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
281. The AAV particle of any one of embodiments 191-202, 2.40-247, 249, 250, 253-263,
266, or 272-280, wherein [N1]-[N2]-[N3]-[N4] corresponds to positions 453-466 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461, Q462, N463, Q464, Q465, and T466) of SEQ ID NO: 981.
282. The AAV particle of any one of embodiments 168-202, 240-247, 249, 250, 253-263,
266, or 272-280, wherein [N1]-[N2]-[N3] corresponds to positions 453-461 (e.g., G453, S454, G455, S456, P457, H458, S459, K460, A461) of SEQ ID NO: 981 .
283. The AAV particle of any one of embodiments 228-257, 260, 261, 264-282, wherein [N1]-[N2]-[N3]-[N4] corresponds to positions 453-466 (e.g.. G453, H454, D455, S456, P457, H458, K459, S460, G461, Q462, N463, Q464, Q465, T466) of SEQ ID NO: 982.
284. The AAV particle of any one of embodiments 203-257, 260, 261, 264-283, wherein [N1]-[N2]-[N3] corresponds to positions 453-461 (e.g., G453, H454, D455, S456, P457, H458, K459, S460, G461) of SEQ ID NO: 982.
285. The AAV particle of any one of embodiments 228-257, 260, 261, 264-282, wherein [N1]-[N2]-[N3]-[N4] corresponds to positions 453-466 of any one of SEQ ID NOs: 4 or 36- 59.
2.86. The AAV particle of any one of embodiments 198-202 or 234-286, wherein [N0]-[N1]- [N2]-[N3]-[N4] replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
287. The AA V particle of any one of embodiments 198-202 or 234-286, wherein [N0]-[N1]- [N2]-[N3]-[N4] is present immediately subsequent to position 448, and wherein [N0]-[N1]-
[N2]-[N3]-[N4] replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered according to SEQ ID NO: 138.
288. The AAV particle of any one of embodiments 198-202, 240-247, 249, 250, 253-263, 266, 272-281, 286, or 287 , wherein [N0]- [N1]-[N2]-[N3]-[N4] corresponds to positions 449- 466 (e.g., K449, T450, 1451, N452, G453, S454, G455, S456, P457, H458, S459, K460, A461 , Q462, N463, Q464, Q465, T466) of SEQ ID NO: 981.
289. The AAV particle of any one of embodiments 234-257, 260, 261, 264-284, 286, or 287, wherein [N0]-[N1]-[N2]-[N3]-[N4] corresponds to positions 449-466 (e.g., K449, T450, 1451, N452, G453, H454, D455, S456, P457, II458, K459, S460, G461 , Q462, N463, Q464, Q465, T466) of SEQ ID NO: 982.
2.90. The AAV particle of any one of embodiments 234-257, 2.60, 261, 264-284, 2.86, or 287, wherein [N0]-[N1]-[N2]-[N3]-[N4] corresponds to positions 449-466 of any one of SEQ ID NOs: 4 or 36-59.
291. The AA V particle of any one of embodiments 191-202 or 228-290, wherein [N4] is present immediately subsequent to position 461, numbered according to SEQ ID NO; 4, 36, 981, or 982.
292. The AAV particle of any one of embodiments 191 -202 or 228-291 , wherein [N4] replaces positions 462-466 (e.g., Q462, N463, Q464, Q465, and T466), numbered according to SEQ ID NO: 4, 36, 981, or 982.
293. The AAV particle of any one of embodiments 191-202 or 228-292, wherein [N2]-[N3]- [N4] replaces positions 462-466 (e.g., Q462, N463, Q464, Q465, and T466), numbered according to SEQ ID NO: 4, 36, 981 , or 982,
294. The AAV particle of any one of embodiments 191-202 or 228-293, wherein [N2]-[N3]- [N4] is present immediately subsequent to position 455, and wherein [N2]-[N3]-[N4] replaces positions 462-466 (e.g., Q462, N463, Q464, Q465, and T466), numbered according to SEQ ID NO: 4, 36, 981, or 982.
295. The AAV particle of any one of embodiments 168-294, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N2]-[N3],
296. The AAV particle of any one of embodiments 168-295, wherein the AAV capsid variant comprises, from N-terminus to C-tenninus, [N1]-[N2]-[N3|.
297. The AAV particle of any one of embodiments 168-296, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3].
298. The AAV particle of any one of embodiments 168-297, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4],
299. The AAV particle of any one of embodiments 168-298, wherein the AAV capsid variant composes, from N-terminus to C-tenninus, [N0]-[N1]-[N2.]-[N3]-[N4].
300. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV 9 capsid vanant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the formula [A|-[B] (SEQ ID NO: 4696), wherein:
(i) [A] comprises GSGSPH (SEQ ID NO: 4695); and
(ii) [B] comprises X1, X2, X3, X4, and X5, wherein:
(a) X1 is S, I, F, V, C, Y, W, R, P, L, Q, M, K, or G;
(b) X2 is K, M, R, F, V, C. P, Y, L, W, G, N, S. T, I, or A:
(c) X3 is A, Y, L, R, W, C, T, F, H, I, P, M, K, S, V, G, Q, or N;
(d) X4 is Q, M, F, K, H, R, C, W, P, V, L, G, S, Y, I, A, T, D, N, or E; and
(e) X5 is A, N, Y, R, K, L. I, M, Q, S, C, W, F, T, G, V, or P; optionally wherein the AAV capsid, variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(e).
301. The AAV particle of embodiment 300, wherein:
(a) X1 is S, L, R, V, or P;
(b) X2 is K, C, F, L, P, R, S, or V;
(c) X3 is A, C, F, I, K, L, M, P, R, T, W, or Y:
(d) X4 is Q, R, S, T, C, F, K, L, P or Y ; and
(e) X5 is N, R, S, T, K, M, Q or Y; optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a. conservative substitution, of any of the aforesaid amino acids in (a)-(e).
302. The AAV particle of embodiment 300 or 301, wherein [B] comprises SKA, SMY, SKL, SKR, SKW, SRC, SFT, SKF, IVW, SKY, SCH, FPW, SKI, VYY, SLY, SKP, SRF, SRM, SVK, SWA, SLW, SFR, SKK, SYA, SCS, SGA, SFP, SFF, SMC, SKT, SGK, FYR, CRV, YGI, VNC, SLA, WSY, RWL, PSC, SSW, SKG, VPW, SGC, STT, PKR, SKC, WVP, SFW, RIK, SKM, LRW, LPT, SYM, LLC, RCC, LCV, SYL, QGC, MAF, SFQ, SLC, RPW, RPR, SCP, SVR, SLP, VYII, SYT, LVY, YRY, SWL, CPA, SPP, RWT, PRK, PFV, SKS, WVA, SKV, CAL, SSC, SKN, LCT, STC, SKQ, KSG, SYY, SLT, SCQ, FPF, SVF, GRY, AQA, AQN. YMN, AFY, LKR, RHR, AQK, WRL, CRN, TCN, FFI, AQY, WQN. YFM, ARQ, HQN, IRR, YQN, YWN, APS, FWN, AQC, MRN, K.KN, APN, WKN, ARW. RPN, KVF, AFN, ACS, RLW, SRN, CPN, ACN, FRQ, PEN, FGN, CQN, LFW, TRK, KRN, RQN, VQN, IQN, AQR, PFR, AWN, RSY, LQN, WLN, RRA, AQT, GCT, RYT, TPN, ARM, CFL, PQN, WSN, FKN, KQN. APR. RYN, MIC, TQN, WKS, AAR, LTR, IRG. LVN, FQN, ACQ, WGL, IL R. QIN, ACI, ALR, AHA, CLN, AFV, AQF, RC.N, MPC, KTS, PYN, AQS, TRN, LKN, AQM, CTN, PDN, RNY, ACR, CSV, ARI, LPK, SEQ, VRM, NSR, RKR, ARN, QRP, RVV, GQN. YSN, QSN, AKG, CIS, FEN, AKK. KAQ, MYM, KAF, KLK, KRH, KWR, RCR, FTC, KFF, VWQ, KYF, KAR, CHQ, PWQ, KIR, YYQ, LYW, KPK, RFW, RMR, VKK, WAP, LWK, FRP, KKV, YAF, KAC, KRL, CSR, RCP, GAC, KFR, FFG, MCQ, KEF, KTR, GKR, YRQ, RVQ, GIQ, NCQ, KPF, LAW, KRS, SYQ, WLQ, KRR, KGC, KRY, GCQ, FTP, ITC. KRQ, KCF, VPQ, FWS, KFK, IKQ, K AP. FRY, KM1, RWQ, PTQ, KWK, YMR, KAA, LCQ, CCQ, CVQ, KLT, KLC, YLV, AFQ, KWG, KIL, FQI, KAL, KAH, LCL, PRQ, CPQ, VRY, VRC, KMP, KKT, LPY, YHQ, YTR, VYQ, RYQ, WLK, PAQ, MCT, PPD, WTQ, RKQ, KCS, FVQ, KLP, KSE. VAQ, LYQ, KVR, ALQ, SCT, KNS, KRK, CTQ, TCL, YAR, KQR, KRV, SGQ, YYS, LTC, CQS, KAK, KPQ, PFQ, KCT, or VFE.
303. The AAV particle of any one of embodiments 300-302, wherein [B] comprises SKAQ (SEQ ID NO: 5829), SMYM (SEQ ID NO: 5830), SKAF (SEQ ID NO: 5831), SKLK (SEQ ID NO: 5832), SKRTI (SEQ ID NO: 5833), SKWR (SEQ ID NO: 5834), SRCR (SEQ ID NO: 5835), SFTC (SEQ ID NO: 5836), SKFF (SEQ ID NO: 5837), IVWQ (SEQ ID NO:
5838), SKYF (SEQ ID NO: 5839), SKAR (SEQ ID NO: 5840), SCIIQ (SEQ ID NO: 5841), FPWQ (SEQ ID NO: 5842), SKIR (SEQ ID NO: 5843), VYYQ (SEQ ID NO: 5844), SLYW (SEQ ID NO: 5845), SKPK (SEQ ID NO: 5846), SKEW (SEQ ID NO: 5847), SRMR (SEQ ID NO: 5848), SVKK (SEQ ID NO: 5849), SWAP (SEQ ID NO: 5850), SLWK (SEQ ID NO: 5851), SFRP (SEQ ID NO: 5852), SKKV (SEQ ID NO: 5853), SYAF (SEQ ID NO: 5854). SK U' (SEQ ID NO: 5855), SKRL (SEQ ID NO: 5856), SCSR (SEQ ID NO: 5857), SRCP (SEQ ID NO: 5858), SGAC (SEQ ID NO: 5859), SKFR (SEQ ID NO: 5860), SFPF (SEQ ID NO: 5861), SFFG (SEQ ID NO: 5862), SMCQ (SEQ ID NO: 5863), SKLF (SEQ ID NO: 5864), SKTR (SEQ ID NO: 5865), SGKR (SEQ ID NO: 5866), FYRQ (SEQ ID NO: 5867), CRVQ (SEQ ID NO: 5868), YGIQ (SEQ ID NO: 5869), VNCQ (SEQ ID NO: 5870), SKPF (SEQ ID NO: 5871), SLAW (SEQ ID NO: 5872), SKRS (SEQ ID NO: 5873), WSYQ (SEQ ID NO: 5874), RWLQ (SEQ ID NO: 5875), PSCQ (SEQ ID NO: 5876), SSWL (SEQ ID NO: 5877), SKRR (SEQ ID NO: 5878), SKGC (SEQ ID NO: 5879), VPWQ (SEQ ID NO: 5880), SKRY (SEQ ID NO: 5881), SGCQ (SEQ ID NO: 5882), Sl i p (SEQ ID NO: 5883), STIC (SEQ ID NO: 5884), PKRQ (SEQ ID NO: 5885), SKCF (SEQ ID NO: 5886), WVPQ (SEQ ID NO: 5887), SEWS (SEQ ID NO: 5888), SKFK (SEQ ID NO: 5889), RIKQ (SEQ ID NO: 5890), SKAP (SEQ ID NO: 5891), SFRY (SEQ ID NO: 5892), SKMI (SEQ ID NO: 5893), LRWQ (SEQ ID NO: 5894), LPTQ (SEQ ID NO: 5895), SKWK (SEQ ID NO: 5896), SYMR (SEQ ID NO: 5897), SKAA (SEQ ID NO: 5898), LLCQ (SEQ ID NO: 5899), RCCQ (SEQ ID NO: 5900), LCVQ (SEQ ID NO: 5901), SKLT (SEQ ID NO: 5902), SKLC (SEQ ID NO: 5903), SYLV (SEQ ID NO: 5904), QGCQ (SEQ ID NO: 5905), MAFQ (SEQ ID NO: 5906), SKWG (SEQ ID NO: 5907), SKIL (SEQ ID NO: 5908), SFQI (SEQ ID NO: 5909), SKAL (SEQ ID NO: 5910), SKAH (SEQ ID NO: 5911), SLCL (SEQ ID NO: 5912), RPWQ (SEQ ID NO: 5913), RPRQ (SEQ ID NO: 5914), SCPQ (SEQ ID NO: 5915), SVRY (SEQ ID NO: 5916), SVRC (SEQ ID NO: 5917), SKMP (SEQ ID NO: 5918), SKKT (SEQ ID NO: 5919), SLPY (SEQ ID NO: 5920), VYIIQ (SEQ ID NO: 5921), SYTR (SEQ ID NO: 5922). LVYQ (SEQ ID NO: 5923), YRYQ (SEQ ID NO: 5924), SWLK (SEQ ID NO: 5925), CPAQ (SEQ ID NO: 5926), SMCT (SEQ ID NO: 5927), SPPD (SEQ ID NO: 5928), SKRN (SEQ ID NO: 5929), RWTQ (SEQ ID NO: 5930), PRKQ (SEQ ID NO: 5931), SKCS (SEQ ID NO: 5932), PFVQ (SEQ ID NO: 5933), SKLP (SEQ ID NO: 5934), SKSE (SEQ ID NO: 5935), WVAQ (SEQ ID NO: 5936), SLYQ (SEQ ID NO: 5937), SKVR (SEQ ID NO: 5938). CALQ (SEQ ID NO: 5939), SSCT (SEQ ID NO: 5940), SKNS (SEQ ID NO: 5941), SKRK (SEQ ID NO: 5942), LCTQ (SEQ ID NO: 5943), STCL (SEQ ID NO: 5944), SY AR (SEQ ID NO: 5945), SKQR (SEQ ID NO: 5946), SKRY (SEQ ID NO: 5947), KSGQ (SEQ ID NO:
5948), SYYS (SEQ ID NO: 5949), SLTC (SEQ ID NO: 5950), SCQS (SEQ ID NO: 5951), SKAK (SEQ ID NO: 5952), SKPQ (SEQ ID NO: 5953), FPFQ (SEQ ID NO: 5954), SKCT (SEQ ID NO: 5955), SAFE (SEQ ID NO: 5956), GRYQ (SEQ ID NO: 5957), KAQA (SEQ ID NO: 5958), KAQN (SEQ ID NO: 5959), MYMN (SEQ ID NO: 5960), KAFY (SEQ ID NO: 5961), KLKR (SEQ ID NO: 5962), KRHR (SEQ ID NO: 5963), KAQK (SEQ ID NO: 5964). KWRL (SEQ ID NO: 5965), RCRN (SEQ ID NO: 5966), FTCN (SEQ ID NO: 5967), KFFI (SEQ ID NO: 5968), KAQY (SEQ ID NO: 5969), VWQN (SEQ ID NO: 5970), KYFM (SEQ ID NO: 5971), KARQ (SEQ ID NO: 5972), CHQN (SEQ ID NO: 5973), PWQN (SEQ ID NO: 5974), KIRR (SEQ ID NO: 5975), YYQN (SEQ ID NO: 5976), LYWN (SEQ ID NO: 5977), KPKR (SEQ ID NO: 5978), KAFS (SEQ ID NO: 5979), RFWN (SEQ ID NO: 5980), KAQC (SEQ ID NO: 5981), RMRN (SEQ ID NO: 5982), VKKN (SEQ ID NO: 5983), WAPN (SEQ ID NO: 5984), LWKN (SEQ ID NO: 5985), KARW (SEQ ID NO: 5986). FRPN (SEQ ID NO: 5987), KKVF (SEQ ID NO: 5988), YAFN (SEQ ID NO: 5989), KACS (SEQ ID NO: 5990), KRLW (SEQ ID NO: 5991), CSRN (SEQ ID NO: 5992), RCPN (SEQ ID NO: 5993), GACN (SEQ ID NO: 5994), KFRQ (SEQ ID NO: 5995), FPFN (SEQ ID NO: 5996), FFGN (SEQ ID NO: 5997), MCQN (SEQ ID NO: 5998), KLFW (SEQ ID NO: 5999), KTRK (SEQ ID NO: 6000), GKRN (SEQ ID NO: 6001), YRQN (SEQ ID NO: 6002), RVQN (SEQ ID NO: 6003), GIQN (SEQ ID NO: 6004), KAQR (SEQ ID NO: 6005), NCQN (SEQ ID NO: 6006), KPFR (SEQ ID NO: 6007), LAWN (SEQ ID NO: 6008), KRSY (SEQ ID NO: 6009), SYQN (SEQ ID NO: 6010), WLQN (SEQ ID NO: 6011), SCQN (SEQ ID NO: 6012), SWEN (SEQ ID NO: 6013), KRRA (SEQ ID NO: 6014), KAQT (SEQ ID NO: 6015), KGCT (SEQ ID NO: 6016), KRYT (SEQ ID NO: 6017), GCQN (SEQ ID NO: 6018), FTPN (SEQ ID NO: 6019), TTCN (SEQ ID NO: 6020), KARM (SEQ ID NO: 6021), KRQN (SEQ ID NO: 6022). KCFL (SEQ ID NO: 6023), VPQN (SEQ ID NO: 6024), FWSN (SEQ ID NO: 6025), KFKN (SEQ ID NO: 6026), IKQN (SEQ ID NO: 6027), KAPR (SEQ ID NO: 6028), FRYN (SEQ ID NO: 6029), KMIC (SEQ ID NO: 6030), RWQN (SEQ ID NO: 6031), PTQN (SEQ ID NO: 6032), KWKS (SEQ ID NO: 6033). YMRN (SEQ ID NO: 6034), KAAR (SEQ ID NO: 6035), LCQN (SEQ ID NO: 6036), CCQN (SEQ ID NO: 6037), CVQN (SEQ ID NO: 6038), KLTR (SEQ ID NO: 6039), KLCT (SEQ ID NO: 6040), KIRG (SEQ ID NO: 6041), YLVN (SEQ ID NO: 6042), AFQN (SEQ ID NO: 6043), KACQ (SEQ ID NO: 6044), KWGL (SEQ ID NO: 6045), KIER (SEQ ID NO: 6046), FQ1N (SEQ ID NO: 6047), KACI (SEQ ID NO: 6048), KALR (SEQ ID NO: 6049), KARA (SEQ ID NO: 6050), LCLN (SEQ ID NO: 6051), KAFV (SEQ ID NO: 6052), PRQN (SEQ ID NO: 6053), CPQN (SEQ ID NO: 6054), KAQF (SEQ ID NO: 6055), VRYN (SEQ ID NO: 6056), VRCN (SEQ
ID NO: 6057), KMPC (SEQ ID NO: 6058), KKTS (SEQ ID NO: 6059), LPYN (SEQ ID NO: 6060), YHQN (SEQ ID NO: 6061), KAQS (SEQ ID NO: 6062), YTRN (SEQ ID NO: 6063), VYQN (SEQ ID NO: 6064), RYQN (SEQ ID NO: 6065), WLKN (SEQ ID NO: 6066), KAQM (SEQ ID NO: 6067), PAQN (SEQ ID NO: 6068), MCTN (SEQ ID NO: 6069), PPDN (SEQ ID NO: 6070), KRNY (SEQ ID NO: 6071), WTQN (SEQ ID NO: 6072), KACR (SEQ ID NO: 6073), RKQN (SEQ ID NO: 6074), ROSY (SEQ ID NO: 6075), KARI (SEQ ID NO: 6076), FVQN (SEQ ID NO: 6077), KLPK (SEQ ID NO: 6078), KSEQ (SEQ ID NO: 6079), VAQN (SEQ ID NO: 6080), LYQN (SEQ ID NO: 6081), KVRM (SEQ ID NO: 6082), ALQN (SEQ ID NO: 6083), SCTN (SEQ ID NO: 6084), KNSR (SEQ ID NO: 6085), KRKR (SEQ ID NO: 6086), CTQN (SEQ ID NO: 6087), TCLN (SEQ ID NO: 6088), YARN (SEQ ID NO: 6089), KQRP (SEQ ID NO: 6090), KRVV (SEQ ID NO: 6091), SGQN (SEQ ID NO: 6092), YYSN (SEQ ID NO: 6093), ETON (SEQ ID NO: 6094), CQSN (SEQ ID NO: 6095), KAKG (SEQ ID NO: 6096), KPQN (SEQ ID NO: 6097), PFQN (SEQ ID NO: 6098), KCTS (SEQ ID NO: 6099), VEEN (SEQ ID NO: 6100), or KAKK (SEQ ID NO: 6101).
304. The AAV particle of any one of embodiments 300-303, wherein [B] is or comprises:
(i) SKAQA (SEQ ID NO: 6102), SKAQN (SEQ ID NO: 6103), SMYMN (SEQ ID NO: 6104), SKAFY (SEQ ID NO: 6105), SKLKR (SEQ ID NO: 6106), SKRHR (SEQ ID NO: 6107), SKAQK (SEQ ID NO: 6108), SKWRL (SEQ ID NO: 6109). SRCRN (SEQ ID NO: 61 10), SFTCN (SEQ ID NO: 6111.), SKFFI (SEQ ID NO: 6112), SKAQY (SEQ ID NO: 6113), IVWQN (SEQ ID NO: 6114), SKYFM (SEQ ID NO: 6115), SKARQ (SEQ ID NO: 6116), SCHQN (SEQ ID NO: 6117), FPWQN (SEQ ID NO: 6118), SKIRR (SEQ ID NO: 6119), VYYQN (SEQ ID NO: 6120), SLYWN (SEQ ID NO: 6121), SKPKR (SEQ ID NO: 6122), SKAFS (SEQ ID NO: 6123), SRFWN (SEQ ID NO: 6124), SKAQC (SEQ ID NO: 6125), SRMRN (SEQ ID NO: 6126), SVKKN (SEQ ID NO: 6127), SWAPN (SEQ ID NO: 6128). SLWKN (SEQ ID NO: 6129), SKARW (SEQ ID NO: 6130), SFRPN (SEQ ID NO: 6131), SKKVF (SEQ ID NO: 6132), SYAFN (SEQ ID NO: 6133), SKACS (SEQ ID NO: 6134), SKRLW (SEQ ID NO: 6135), SCSRN (SEQ ID NO: 6136), SRCPN (SEQ ID NO: 6137), SGACN (SEQ ID NO: 6138), SKFRQ (SEQ ID NO: 6139), SFPFN (SEQ ID NO: 6140), SFFGN (SEQ ID NO: 6141), SMCQN (SEQ ID NO: 6142), SKLFW (SEQ ID NO: 6143), SKTRK (SEQ ID NO: 6144), SGKRN (SEQ ID NO: 6145), FYRQN (SEQ ID NO: 6146), CRVQN (SEQ ID NO: 6147), YGIQN (SEQ ID NO: 6148), SKAQR (SEQ ID NO: 6149), VNCQN (SEQ ID NO: 6150), SKPFR (SEQ ID NO: 6151), SLAWN (SEQ ID NO:
6152), SKRSY (SEQ ID NO: 6153), WSYQN (SEQ ID NO: 6154), RWLQN (SEQ ID NO: 6155), PSCQN (SEQ ID NO: 6156), SSWLN (SEQ ID NO: 6157), SKRRA (SEQ ID NO: 6158), SKAQT (SEQ ID NO: 6159), SKGCT (SEQ ID NO: 6160), VPWQN (SEQ ID NO: 6161), SKRYT (SEQ ID NO: 6162), SGCQN (SEQ ID NO: 6163), SFTPN (SEQ ID NO: 6164), STTCN (SEQ ID NO: 6165), SKARM (SEQ ID NO: 6166), PKRQN (SEQ ID NO: 6167). SKCFL (SEQ ID NO: 6168), WVPQN (SEQ ID NO: 6169), SFWSN (SEQ ID NO: 6170), SKFKN (SEQ ID NO: 6171), RIKQN (SEQ ID NO: 6172), SKAPR (SEQ ID NO: 6173), SFRYN (SEQ ID NO: 6174), SKMIC (SEQ ID NO: 6175), LRWQN (SEQ ID NO: 6176), LPTQN (SEQ ID NO: 6177), SKWKS (SEQ ID NO: 6178), SYMRN (SEQ ID NO: 6179), SKAAR (SEQ ID NO: 6180), LLCQN (SEQ ID NO: 6181), RCCQN (SEQ ID NO: 6182), LCVQN (SEQ ID NO: 6183), SKLTR (SEQ ID NO: 6184), SKLCT (SEQ ID NO: 6185), SKIRG (SEQ ID NO: 6186), SYLVN (SEQ ID NO: 6187), QGCQN (SEQ ID NO: 6188). MAFQN (SEQ ID NO: 6189), SKACQ (SEQ ID NO: 6190). SKWGL (SEQ ID NO: 6191), SKIER (SEQ ID NO: 6192), SFQIN (SEQ ID NO: 6193), SKACI (SEQ ID NO: 6194), SKALR (SEQ ID NO: 6195), SKAHA (SEQ ID NO: 6196), SLCLN (SEQ ID NO: 6197), SKAFV (SEQ ID NO: 6198), RPWQN (SEQ ID NO: 6199), RPRQN (SEQ ID NO: 6200), SCPQN (SEQ ID NO: 6201), SKAQF (SEQ ID NO: 6202), SVRYN (SEQ ID NO: 6203), SVRCN (SEQ ID NO: 6204), SKMPC (SEQ ID NO: 6205), SKKTS (SEQ ID NO: 6206), SLPYN (SEQ ID NO: 6207), VYHQN (SEQ ID NO: 6208), SKAQS (SEQ ID NO: 6209). SYTRN (SEQ ID NO: 6210), LVYQN (SEQ ID NO: 6211), YRYQN (SEQ ID NO: 6212), SWLKN (SEQ ID NO: 6213), SKAQM (SEQ ID NO: 6214), CPAQN (SEQ ID NO: 6215), SMCTN (SEQ ID NO: 6216), SPPDN (SEQ ID NO: 6217), SKRNY (SEQ ID NO: 6218), RWTQN (SEQ ID NO: 6219), SKACR (SEQ ID NO: 6220), PRKQN (SEQ ID NO: 6221), SKCSV (SEQ ID NO: 6222), SKARI (SEQ ID NO: 6223). PFVQN (SEQ ID NO: 6224), SKLPK (SEQ ID NO: 6225), SKSEQ (SEQ ID NO: 6226), WVAQN (SEQ ID NO: 6227), SLYQN (SEQ ID NO: 6228), SKVRM (SEQ ID NO: 6229), CALQN (SEQ ID NO: 6230). SSCTN (SEQ ID NO: 6231), SKNSR (SEQ ID NO: 6232), SKRKR (SEQ ID NO: 6233), LCTQN (SEQ ID NO: 6234), STCLN (SEQ ID NO: 6235), SYARN (SEQ ID NO: 6236), SKQRP (SEQ ID NO: 6237), SKRVV (SEQ ID NO: 6238), KSGQN (SEQ ID NO: 6239), SYYSN (SEQ ID NO: 6240), SLTCN (SEQ ID NO: 6241), SCQSN (SEQ ID NO: 6242), SKAKG (SEQ ID NO: 6243), SKPQN (SEQ ID NO: 6244), FPFQN (SEQ ID NO: 6245), SKCTS (SEQ ID NO: 6246), SVFEN (SEQ ID NO: 6247), SKAKK (SEQ ID NO: 6248), or GRYQN (SEQ ID NO: 6249);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof;
(lii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
305. The AAV particle of any one of embodiments 300-304, wherein [A]-[B] is or comprises:
(i) GSGSPHSKAQA (SEQ ID NO: 6250), GSGSPHSKAQN (SEQ ID NO: 6251), GSGSPHSMYMN (SEQ ID NO: 6252), GSGSPHSKAFY (SEQ ID NO: 6253), GSGSPHSKLKR (SEQ ID NO: 6254), GSGSPHSKRHR (SEQ ID NO: 6255), GSGSPHSKAQK (SEQ ID NO: 6256), GSGSPHSKWRL (SEQ ID NO: 6257), GSGSPHSRCRN (SEQ ID NO: 6258). GSGSPHSFTCN (SEQ ID NO: 6259), GSGSPHSKFFI (SEQ ID NO: 6260), GSGSPHSKAQY (SEQ ID NO: 6261), GSGSPHTVWQN (SEQ ID NO: 6262), GSGSPHSKYFM (SEQ ID NO: 6263), GSGSPHSKARQ (SEQ ID NO: 6264), GSGSPHSCHQN (SEQ ID NO: 6265), GSGSPHFPWQN (SEQ ID NO: 6266), GSGSPHSKIRR (SEQ ID NO: 6267), GSGSPHVYYQN (SEQ ID NO: 6268), GSGSPHSLYWN (SEQ ID NO: 62.69), GSGSPHSKPKR (SEQ ID NO: 6270), GSGSPHSKAFS (SEQ ID NO: 6271), GSGSPHSRFWN (SEQ ID NO: 6272), GSGSPHSK AQC (SEQ ID NO: 6273), GSGSPHSRMRN (SEQ ID NO: 6274), GSGSPHSVKKN (SEQ ID NO: 6275), GSGSPHSWAPN (SEQ ID NO: 6276), GSGSPHSLWKN (SEQ ID NO: 6277), GSGSPHSKARW (SEQ ID NO: 6278), GSGSPHSFRPN (SEQ ID NO: 6279), GSGSPHSKKVF (SEQ ID NO: 6280), GSGSPHSYAFN (SEQ ID NO: 6281), GSGSPHSKACS (SEQ ID NO: 62.82), GSGSPHSKRLW (SEQ ID NO: 6283), GSGSPHSCSRN (SEQ ID NO: 6284), GSGSPHSRCPN (SEQ ID NO: 6285), GSGSPHSGACN (SEQ ID NO: 6286), GSGSPHSKFRQ (SEQ ID NO: 6287), GSGSPHSFPFN (SEQ ID NO: 62.88), GSGSPHSFFGN (SEQ ID NO: 6289), GSGSPHSMCQN (SEQ ID NO: 6290), GSGSPHSKLFW (SEQ ID NO: 6291 ), GSGSPHSKTRK (SEQ ID NO: 6292), GSGSPHSGKRN (SEQ ID NO: 6293), GSGSPHFYRQN (SEQ ID NO: 6294), GSGSPHCRVQN (SEQ ID NO: 6295), GSGSPHYGIQN (SEQ ID NO: 62.96), GSGSPHSKAQR (SEQ ID NO: 6297), GSGSPHVNCQN (SEQ ID NO: 6298), GSGSPHSKPFR (SEQ ID NO: 6299), GSGSPHSLAWN (SEQ ID NO: 6300), GSGSPHSKRSY (SEQ ID NO: 6301),
GSGSPHWSYQN (SEQ ID NO: 6302), GSGSPHRWLQN (SEQ ID NO: 6303), GSGSPHPSCQN (SEQ ID NO: 6304), GSGSPHSSWLN (SEQ ID NO: 6305), GSGSPHSKRRA (SEQ ID NO: 6306), GSGSPHSKAQT (SEQ ID NO: 6307), GSGSPHSKGCT (SEQ ID NO: 6308), GSGSPHVPWQN (SEQ ID NO: 6309), GSGSPHSKRYT (SEQ ID NO: 6310), GSGSPHSGCQN (SEQ ID NO: 6311 ), GSGSPHSFTPN (SEQ ID NO: 6312), GSGSPHSITCN (SEQ ID NO: 6313), GSGSPHSKARM (SEQ ID NO: 6314), GSGSPHPKRQN (SEQ ID NO: 6315), GSGSPHSKCFL (SEQ ID NO: 6316), GSGSPHWVPQN (SEQ ID NO: 6317), GSGSPHSFWSN (SEQ ID NO: 6318), GSGSPHSKFKN (SEQ ID NO: 6319), GSGSPHRIKQN (SEQ ID NO: 6320), GSGSPHSKAPR (SEQ ID NO: 6321), GSGSPHSFRYN (SEQ ID NO: 6322), GSGSPHSKMIC (SEQ ID NO: 6323), GSGSPHLRWQN (SEQ ID NO: 6324), GSGSPHLPTQN (SEQ ID NO: 6325), GSGSPHSKWKS (SEQ ID NO: 6326), GSGSPHSYMRN (SEQ ID NO: 6327). GSGSPHSKAAR (SEQ ID NO: 6328), GSGSPHELCQN (SEQ ID NO: 6329), GSGSPHRCCQN (SEQ ID NO: 6330), GSGSPHLCVQN (SEQ ID NO: 6331), GSGSPHSKLTR (SEQ ID NO: 6332), GSGSPHSKLCT (SEQ ID NO: 6333), GSGSPHSKIRG (SEQ ID NO: 6334), GSGSPHSYLVN (SEQ ID NO: 6335), GSGSPHQGCQN (SEQ ID NO: 6336), GSGSPHMAFQN (SEQ ID NO: 6337), GSGSPHSKACQ (SEQ ID NO: 6338), GSGSPHSKWGL (SEQ ID NO: 6339), GSGSPHSKILR (SEQ ID NO: 6340), GSGSPHSFQLN (SEQ ID NO: 6341), GSGSPHSKACI (SEQ ID NO: 6342), GSGSPHSKALR (SEQ ID NO: 6343), GSGSPHSKAHA (SEQ ID NO: 6344), GSGSPHSLCLN (SEQ ID NO: 6345), GSGSPHSKAFV (SEQ ID NO: 6346), GSGSPHRPWQN (SEQ ID NO: 6347), GSGSPHRPRQN (SEQ ID NO: 6348), GSGSPHSCPQN (SEQ ID NO: 6349), GSGSPHSKAQF (SEQ ID NO: 6350), GSGSPHSVRYN (SEQ ID NO: 6351), GSGSPHSVRCN (SEQ ID NO: 6352), GSGSPHSKMPC (SEQ ID NO: 6353), GSGSPHSKKTS (SEQ ID NO: 6354), GSGSPHSLPYN (SEQ ID NO: 6355), GSGSPHVYHQN (SEQ ID NO: 6356), GSGSPHSKAQS (SEQ ID NO: 6357), GSGSPHSYTRN (SEQ ID NO: 6358), GSGSPHLVYQN (SEQ ID NO: 6359), GSGSPHYRYQN (SEQ ID NO: 6360), GSGSPHSWLKN (SEQ ID NO: 6361), GSGSPHSKAQM (SEQ ID NO: 6362), GSGSPHCPAQN (SEQ ID NO: 6363), GSGSPHSMCTN (SEQ ID NO: 6364), GSGSPHSPPDN (SEQ ID NO: 6365), GSGSPHSKRNY (SEQ ID NO: 6366), GSGSPHRWTQN (SEQ ID NO: 6367), GSGSPHSKACR (SEQ ID NO: 6368), GSGSPHPRKQN (SEQ ID NO: 6369),
GSGSPHSKCSV (SEQ ID NO: 6370), GSGSPHSKARI (SEQ ID NO: 6371 ), GSGSPHPFVQN (SEQ ID NO: 6372), GSGSPHSKLPK (SEQ ID NO: 6373), GSGSPHSKSEQ (SEQ ID NO: 6374), GSGSPHWVAQN (SEQ ID NO: 6375), GSGSPHSLYQN (SEQ ID NO: 6376), GSGSPHSKVRM (SEQ ID NO: 6377), GSGSPHCALQN (SEQ ID NO: 6378), GSGSPHSSCTN (SEQ ID NO: 6379), GSGSPHSKNSR (SEQ ID NO: 6380), GSGSPHSKRKR (SEQ ID NO: 6381), GSGSPHLCTQN (SEQ ID NO: 6382), GSGSPHSTCLN (SEQ ID NO: 6383), GSGSPHSYARN (SEQ ID NO: 6384), GSGSPHSKQRP (SEQ ID NO: 6385), GSGSPHSKRVV (SEQ ID NO: 6386), GSGSPHKSGQN (SEQ ID NO: 6387), GSGSPHSYYSN (SEQ ID NO: 6388), GSGSPHSLTCN (SEQ ID NO: 6389), GSGSPHSCQSN (SEQ ID NO: 6390), GSGSPHSKAKG (SEQ ID NO: 6391 ), GSGSPHSKPQN (SEQ ID NO: 6392), GSGSPHFPFQN (SEQ ID NO: 6393), GSGSPHSKCTS (SEQ ID NO: 6394). GSGSPHSVFEN (SEQ ID NO: 6395), GSGSPHSKAKK (SEQ ID NO: 6396), or GSGSPHGRYQN (SEQ ID NO: 6397);
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i)
306. The AAV particle of any one of embodiments 300-305, wherein [A]-[B] does not comprise the amino acid sequence of GSGSPHSKAQN (SEQ ID NO: 6251).
307. The AAV particle of any one of embodiments 300-306, w herein the AAV capsid variant comprises one, two, or all of an amino acid other than Q at position 458 (e.g., R, C, S, W, L, F, Y, H, I, V, A, or P), an amino acid other than Q at position 459 (e.g., K, I, R, L or S), and/or an amino acid other than T at position 460 (e.g., R), numbered according to SEQ ID NO: 138.
308. The AAV particle of any one of embodiments 300-307, wherein the AAV capsid variant comprises:
(i) the amino acid R at position 458;
(ii) the amino acid W at position 458;
(iii) the amino acid Y at position 458;
(iv) the amino acid F at position 458;
(v) the amino acid. S at position 458;
(vi) the amino acid C at position 458;
(vii) the amino acid I at position 458;
(viii) the amino acid L at position 458;
(ix) the amino acid P at position 458;
(x) the amino acid I at position 459;
(xi) the amino acid FI at position 458; or
(xii) the amino acid V at position 458; wherein (i)-(xii) are numbered according to SEQ ID NO: 138.
309. The AAV particle of any one of embodiments 300-307, wherein the AAV capsid variant composes:
(i) the amino acid R at position 458 and the amino acid K at position 459;
(ii) the amino acid C at position 458 and the amino acid I at position 459;
(lii) the amino acid S at position 458 and the amino acid R at position 459;
(iv) tire amino acid L at position 458 and tire amino acid K at position 459;
(v) the amino acid F at position 458 and the amino acid K at position 459;
(vi) the amino acid C at position 458 and the amino acid R at position 459;
(vii) the amino acid H at position 458 and the amino acid R at position 459;
(viii) the amino acid I at position 458 and the amino acid L at position 459;
(ix) the amino acid V at position 458 and the amino acid R at position 459;
(x) the amino acid A at position 458 and the amino acid K at position 459;
(xi) tire amino acid I at position 458 and the amino acid. K at position 459;
(xii) the amino acid C at position 458 and the amino acid S at. position 459; or
(xiii) the amino acid C at position 458 and the amino acid L at position 459; wherein (i)-(xiii) are numbered, according to SEQ ID NO: 138.
310. The AAV particle of any one of embodiments 300-307, wherein the AAV capsid variant comprises the amino acid F at position 458, the amino acid K at position 459, and the amino acid R at position 460, numbered according to SEQ ID NO: 138.
31 1. The AAV particle of any one of embodiments 300-310, wherein the AAV capsid variant comprises one, two, or all of an amino acid other than T at position 450 (e.g., Y, P, W, R, K, S, or F), an amino acid other than I at position 451 (e.g., R, S, Y, I.,, V, H, P, A, or F), and/or an amino acid other than N at position 452 (e.g., V, W, A, T, F, Y, L, R, H, S, or M), numbered according to SEQ ID NO: 138.
312. The AAV particle of any one of embodiments 300-311, wherein the AAV capsid variant comprises the amino acid V at position 452, numbered according to SEQ ID NO: 138.
313. The AAV particle of any one of embodiments 300-312, wherein the AAV capsid variant comprises the amino acid Y at position 450 and the amino acid V at position 452, numbered according to SEQ ID NO: 138.
314. The AAV particle of any one of embodiments 300-312, wherein the AAV capsid variant comprises the amino acid R at position 450 and the amino acid Y at position 451 , numbered according to SEQ ID NO: 138.
315. The AAV particle of any one of embodiments 300-31 1, wherein the AAV capsid variant comprises:
(i) the amino acid P at position 450, the amino acid R at position 451, and the amino acid W at position 452;
(ii) the amino acid Y at. position 450, the amino acid S at position 451 , and the amino acid A at position 452;
(iii) the amino acid Y at position 450, the amino acid Y at position 451, and the amino acid T at position 452;
(iv) the amino acid P at: position 450, the amino acid R at. position 451, and the amino acid F at position 452;
(v) the amino acid W at. position 450, the amino acid L at position 451 , and the amino acid T at position 452;
( vi) the amino acid R at position 450, the amino acid S at position 451 , and the amino acid Y at position 452;
(vii) the amino acid Y at position 450, the amino acid V at position 451, and the amino acid F at position 452;
(viii) the amino acid K at position 450, the amino acid H at position 451, and the amino acid L at position 452;
(ix) the amino acid P at position 450, the amino acid. P at position 451, and the amino acid L at position 452;
(x) the amino acid P at position 450, the amino acid A at position 451, and the amino acid R at position 452;
(xi) the amino acid S at position 450, the amino acid R at position 451 , and the amino acid R at position 452;
(xii) the amino acid F at position 450, the amino acid F at position 451 , and the amino acid H at position 452;
(xiii) the amino acid R at position 450, the amino acid F at position 451, and the amino acid S at position 452;
(xiv) the amino acid Y at position 450, the amino acid S at position 451, and the amino acid M at position 452; or
(xv) the amino acid P at position 450, the amino acid F at position 451, and the amino acid L at position 452; wherein (i)-(xv) is numbered according to SEQ ID NO: 138.
316. The AAV particle of any one of embodiments 300-315, wherein the .AAV capsid variant comprises:
(i) the amino acid sequence of any one of SEQ ID NOs: 3849-3982, 2984-4010, 4681- 4693;
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, or 17 amino acids, e.g., consecutive amino acids, thereof;
(iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
(iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the amino acid sequences in (i).
317. The AAV particle of any one of embodiments 300-316, wherein the AAV capsid variant does not comprise the amino acid sequence of GSGSPHSKAQNQQ (SEQ ID NO: 1801) or GSGSPHSKAQNQQT (SEQ ID NO: 200).
318. The AAV particle of any one of embodiments 300-317, wherein [A]-[B] is present in loop IV.
319. The AAV particle of any one of embodiments 300-318, wherein [A] is present immediately subsequent to position 452, numbered according to SEQ ID NO: 138 or 981.
320. The AAV particle of any one of embodiments 300-319, wherein [A] replaces positions 453-455 (e.g., G453, S454, G455), numbered according to SEQ ID NO: 138 or 981.
321. The AAV particle of any one of embodiments 300-320, w-herein [A] is present immediately subsequent to position 452, and wherein [A] replaces positions 453-455 (e.g., G453, S454, G455), numbered according to SEQ ID NO: 138 or 981.
322. The AAV particle of any one of embodiments 300-321, wherein [B] is present immediately subsequent to [A],
323. The AAV particle of any one of embodiments 300-322, wherein [B] replaces positions 456 and 457 (e.g., Q456, N457), numbered according to SEQ ID NO: 138.
324. The AAV particle of any one of embodiments 300-323, wherein [A]-[B] replaces positions 453-457 (e.g,, G453, S454, G455, Q456, N457), numbered according to SEQ ID NO: 138.
325. The AAV particle of any one of embodiments 300-324, wherein [A]-[B] is present immediately subsequent to position 452, and wherein [A]-[B] replaces positions 453-457 (e.g., G453, S454, G455, Q456, N457), numbered according to SEQ ID NO: 138.
32.6. The AAV particle of any one of embodiments 300-325, wherein the AAV capsid variant comprises, from N-terminus to C -terminus, [A][B],
327. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises at least one, at least two, at least three, or at least four (e.g., from 1 -4 to 1 - 5) charged amino acid residues (e.g., acidic and/or basic amino acid residues) relative to SEQ ID NO: 138, which is present N-terminal to the amino acid sequence of SPH (e.g., within 1,
2, 3, 4, 5, or 6 amino acids from the start of the SPH amino acid sequence (e.g., within positions 450-455 numbered according to SEQ ID NO: 138)), optionally wherein the amino acid sequence of SPH is present at positions 456-458 numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
328. The AAV particle of embodiment 327, wherein the amino acid sequence of SPH is present at positions 456-458 numbered according to any one of SEQ ID NOs: 36-59, 981 , or 982.
329. The AA V particle of embodiment 327 or 328, wherein the AAV capsid variant comprises less than tour, less than three, less than two (e.g., two or one) charged amino acid residues (e.g., acidic and/or basic amino acid residues) relative to SEQ ID NO: 138.
330. The AAV particle of any one of embodiments 327-329, wherein the AA V capsid variant comprises one charged amino acid residues (e.g., an acidic or basic amino acid residue) relative to SEQ ID NO: 138, optionally at any one of positions 450-455 numbered relative to SEQ ID NO: 138.
331. The AAV particle of any one of embodiments 327-330, wherein the charged amino acid residue is an acidic amino acid (e.g., D or E).
332. The AAV particle of any one of embodiments 327-331, wherein the charged amino acid residue is a negatively charged amino acid (e.g., D or E).
333. The AAV particle of any one of embodiments 327-332, wherein the charged amino acid residue is D.
334. The AAV particle of any one of embodiments 32.7-333, wherein the charged amino acid residue is E.
335. The AAV particle of any one of embodiments 327-334, wherein the charged amino acid residue is a basic amino acid (e.g., K, R, or H),
336. Hre AAV particle of any one of embodiments 327-335, wherein the charged amino acid residue is a positively charged amino acid (e.g., K, R, or H).
337. The AAV particle of any one of embodiments 327-336, wherein the charged amino acid residue is H.
338. lire AAV particle of any one of embodiments 327-337, wherein the charged amino acid residue is R.
339. The AAV particle of any one of embodiments 327-338, wherein tire charged amino acid residue is K.
340. lire AAV particle of any one of embodiments 327-339, wherein the AAV capsid variant comprises an acidic amino acid (e.g., E or D) and a basic amino acid (e.g., R, K, or H).
341 . The .AAV particle of any one of embodiments 327-340, wherein at least one, two, three or four charged amino acid residues is present within 1, 2, 3, 4, 5, or 6 (e.g., 1-6) amino acids from the start, of the SPH amino acid sequence.
342. lire AAV particle of any one of embodiments 327-341, wherein the AAV capsid variant composes two charged amino acid residues immediately preceding the amino acid sequence of SPH (e.g., at. positions 454 and 455, numbered according to SEQ ID NO: 138 or SEQ ID NO: 982).
343. The AAV particle of any one of embodiments 327-342, wherein the AAV capsid variant comprises a charged amino acid residue (e.g., E) within 1 , 2, 3, 4, 5 (e.g., 5) amino acids from the start of the SPH amino acid sequence.
344. Hre AAV particle of any one of embodiments 327-343, wherein the AAV capsid variant comprises a charged amino acid residue (e.g., E) at position 451, numbered according to any one of SEQ ID NO: 138, 981. or 982.
345. The AAV particle of any one of embodiments 327-344, wherein the AAV capsid variant comprises E at position 451, numbered according to any one of SEQ ID NOs: 138, 981, or 982.
346. The AAV particle of any one of embodiments 327-345, wherein the .AAV capsid variant comprises a charged amino acid residue (e.g., R or K) at position 452, numbered according to any one of SEQ ID NOs: 138, 981 , or 982,
347. The AAV particle of any one of embodiments 327-346, wherein the AAV capsid variant comprises R at position 452, numbered according to SEQ ID NO: 138 or SEQ ID NO: 982.
348. The AAV particle of any one of embodiments 327-347, wherein the .AAV capsid variant comprises E at position 451 and R at position 452, numbered according to SEQ ID NO: 138 or SEQ ID NO: 982.
349. The AAV particle of any one of embodiments 327-348, wherein the AAV capsid variant has decreased tropism for a liver cell or tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981.
350. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises at least one, at least two, at least three, or at least four (e.g., from 1-4 to 1- 5) charged amino acid residues (e.g., basic amino acid residues) relative to SEQ ID NO: 138, which is present C -terminal to the amino acid sequence of SPH (e.g., within 1, 2, 3, 4, 5, 6, or 7 amino acids from the end of the SPH amino acid sequence (e.g., within positions 459-465 numbered according to any one of SEQ ID NOs: 36-59, or 981)), optionally wherein the amino acid sequence of SPH is present at positions 456-458 numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
351 . The AAV particle of embodiment 350, wherein the amino acid sequence of SPH is present at positions 456-458 numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
352. The AAV particle of embodiment 350 or 351, wherein the AAV capsid variant comprises less than four, less than three, less than two (e.g., two or one) charged amino acid residues (e.g,, basic amino acid residues) relative to SEQ ID NO: 138.
353. The AAV particle of any one of embodiments 350-352, wherein the .AAV capsid variant comprises one charged amino acid residues (e.g., a basic amino acid residue) relative to SEQ ID NO: 138, optionally at any one of positions 456-460 numbered according to SEQ ID NO: 138, or at positions 462-466 numbered according to any one of SEQ ID NOs: 36-59, 981, or 982.
354. The AAV particle of any one of embodiments 350-353, wherein the charged amino acid residue is a basic amino acid (e.g., R or K).
355. The AAV particle of any one of embodiments 350-354, wherein the charged amino acid residue is a positively charged amino acid (e.g., R or K).
356. The AAV particle of any one of embodiments 350-355, wherein the charged amino acid residue is R.
357. The AAV particle of any one of embodiments 350-355, wherein the charged amino acid residue is K,
358. The AAV particle of any one of embodiments 350-357, wherein at least one, two, three or four charged amino acid residues is present within 1, 2, 3, 4, 5, 6, 7 (e.g., 1-7) amino acids from the end of the SPH amino acid sequence.
359. The AAV particle of any one of embodiments 350-358, wherein the AAV capsid variant comprises a. charged amino acid, residue (e.g., K or R) immediately after the SPH sequence (e.g., at position 459 numbered according to SEQ ID NO: 981).
360. The AAV particle of any one of embodiments 350-359, wherein the AAV capsid variant comprises a charged amino acid residue (e.g., K or R) at position 459, numbered according to SEQ ID NO: 138 or SEQ ID NO: 982.
361 . The AAV particle of any one of embodiments 350-360, wherein the AAV capsid variant comprises K at position 459, numbered according to SEQ ID NO: 981.
362. The AAV particle of any one of embodiments 350-360, wherein the AAV capsid variant comprises R at position 459, numbered according to SEQ ID NO: 981.
363. The AAV particle of any one of embodiments 350-362, wherein the AAV capsid variant comprises a. charged amino acid residue (e.g., R or K) at one, two three, four, five, or all of positions 460, 461, 462, 463, 464, and/or 465, numbered according to SEQ ID NO: 138 or 981.
364. The AAV particle of any one of embodiments 300-326 or 350-363, wherein the AAV capsid variant has increased tropism for a liver cell or tissue, relative to the tropism of an AAV capsid, comprising the amino acid sequence of SEQ ID NO: 138.
365. The .AAV particle of any one of embodiments 300-326 or 350-364, wherein the AAV capsid variant is enriched at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100, at least 105, at least 110, at least 115, at least 120, at least 125, at least 130, at least 135, at least 140, at least 150, at least 160, at least 170, at least 180, at least 190, or at least. 200-fold, in the liver compared to an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 4.
366. The AAV particle of any one of embodiments 300-326, 364, or 365, wherein the AAV capsid variant has reduced tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138.
367. The .AAV particle of any one of embodiments 300-326 or 364-366, wherein the AAV capsid variant shows preferential transduction in a liver region relative to the transduction in the brain and/or dorsal root ganglia (DRG).
368. The AAV particle of any one of embodiments 300-326 or 364-367, wherein the AAV capsid variant shows preferential transduction in a liver region relative to the transduction in the heart and/or muscle (e.g., quadriceps).
369. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
(a) the amino acid sequence of any of the sequences provided in Tables 1, 2A, 2B, or 15-21 ;
(b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, or at least 17 consecutive amino acids from any one of the sequences provided in Tables 1, 2A, 2B, or 15-21; or
(c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to any one of the sequences provided in Tables 1, 2A, 2B. or 15-21; or
(d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of the sequences provided in Tables 1, 2A, 2B, or 15-21.
370. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid, variant comprises:
(a) the amino acid sequence of any of SEQ ID NOs: 945-980 or 985-986;
(b) an amino acid sequence comprising at least 3, at least 4, or at least 5 consecutive amino acids from any one of SEQ ID NOs: 945-980 or 985-986; or
(c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 945-980 or 985-986;
(d) an amino sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 945-980 or 985-986.
371. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
(a) the amino acid sequence of any of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909;
(b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, or at least 13 consecutive amino acids from any one of SEQ ID NOs: 2, 200, 2.01, 941 , 943, 204, 208, 404, or 903-909;
(c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941. 943, 204, 208, 404, or 903-909: or
(d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909.
372. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (c.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
(a) the amino acid sequence of any of SEQ ID NOs: 3849-4051 or 4681-4693;
(b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16 or at least 17 consecutive amino acids from any one of SEQ ID NOs: 3849- 4051 or 4681-4693;
(c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 3849-4051 or 4681-4693; or
(d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 3849-4051 or 4681 -4693.
373. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (AT.XN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
(a) the amino acid sequence of any of SEQ ID NOs: 4052-4092;
(b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16 or at least 17 consecutive amino acids from any one of SEQ ID NOs: 4052- 4092;
(c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 4052-4092; or
(d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid, sequence of any one of SEQ ID NOs: 4052-4092.
374. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRMA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises:
(a) the amino acid sequence of any of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097;
(b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16 or at least 17 consecutive amino acids from any one of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097;
(c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097; or
(d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 4056, 4058, 4059, 4062-4064, 4066, 4067, 4080, 4084, 4090, or 4095-4097.
375. The AAV particle of embodiment 369 or 371, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, or at least 13 consecutive amino acids from any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909.
376. The AAV particle of any one of embodiments 369-375, wherein the at least 3 consecutive amino acids comprise SPH.
377. The AAV particle of any one of embodiments 369-371 or 376, wherein the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700).
378. The AAV particle of any one of embodiments 369-371, 376, or 377, wherein the at least
5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701).
379. The AAV particle of any one of embodiments 369-371 or 376-378, wherein the at least
6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941).
380. The AA V particle of embodiment 369-371, wherein the at least 3 consecutive amino acids comprise HDS.
381. The AAV particle of any one of embodiments 369-371 or 380, wherein the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO; 4702).
382. The AAV particle of any one of embodiments 369-371, 380, or 381, wherein the at least
5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703).
383. The AAV particle of any one of embodiments 369-371 or 380-382, wherein the at least
6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2).
384. The AAV particle of any one of embodiments 369-371, wherein:
(i) the at least 3 consecutive amino acids comprise SPH;
(ii) the at least 4 consecutive amino acids comprise SPHK (SEQ ID NO: 6398);
(iii) the at least 5 consecutive amino acids comprise SPHKY (SEQ ID NO; 4715); and/or
(iv) the at least 6 consecutive amino acids comprise SPHKYG (SEQ ID NO: 966).
385. The AA V particle of embodiment 369 or 371, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909.
386. The AAV particle of any one of embodiments 369, 371, or 385, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941).
387. The AAV particle of any one of embodiments 369, 371, or 385, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2).
388. The AA V particle of any one of embodiments 369-371, 384, or 385, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of SPHKYG (SEQ ID NO: 966).
389. The AAV particle of embodiment 370, wherein the AAV capsid variant comprises:
(i) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications, relative to the amino acid, sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589);
(ii) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications, relative to the amino acid sequence of KAEIGHDSPHKSGQNQQT (SEQ ID NO: 1754)
(iii) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications, relative to the amino acid sequence of KTEKMSGSPHSKAQNQQT (SEQ ID NO: 3241);
(iv) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications, relative to the amino acid sequence of KTINGHDSPHSKAQNLQT (SEQ ID NO: 4100); or
(v) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications, relative to the amino acid sequence of KTVNGHDSPHSKAQNQQT (SEQ ID NO: 4062).
390. The AAV particle of embodiment 369 or 371, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941 , 943, 204, 208, 404, or 903-909.
391. The AAV particle of any one of embodiments 369, 371, or 390, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941).
392. The AA V particle of any one of embodiments 369, 371, or 390, wherein the AA V capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2).
393. The AAV particle of any one of embodiments 369, 371, 384, or 390, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of SPHKYG (SEQ ID NO: 966).
394. The AAV particle of embodiment 369, wherein the AAV capsid variant comprises:
(i) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589);
(ti) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of KAEIGHDSPHKSGQNQQT (SEQ ID NO: 1754);
(iii) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of KTEKMSGSPHSKAQNQQT (SEQ ID NO: 3241);
(iv) an amino acid sequence comprising at least one, at least two, or at least three but no more than four different amino acids relative to the amino acid sequence of KTINGHDSPHSKAQNLQT (SEQ ID NO: 4100); or
(v) an amino acid sequence comprising at least one, at least two, or at least three but. no more than four different amino acids relative to the amino acid sequence of KTVNGHDSPHSKAQNQQT (SEQ ID NO: 4062).
395. The AAV particle of any one of embodiments 1-129, 269, 271 , 375-388, or 390-394, wherein the .AAV capsid variant comprises the amino acid sequence of any one of SEQ ID NOs: 2. 200, 201, 941, 943, 204, 208, 404, or 903-909.
396. The AAV particle of any one of embodiments 295-297, 301-305, 313, 314, 318, 319, or
323, wherein the .AAV capsid variant comprises the amino acid sequence of
ERVSGSPHSKA (SEQ ID NO: 6399), optionally wherein the amino acid sequence is present immediately subsequent to position 450 and replaces positions 451-455 (e.g., 1451, N542, G453, S454, G455), numbered according to SEQ ID NO: 138.
397. The AAV particle of any one of embodiments 369-371, 375-379, 385, 386, 389-391 , or 394-396, wherein the AAV capsid variant comprises the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589), optionally wherein the amino acid sequence is present immediately subsequent to position 448 and replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460), numbered according to SEQ ID NO: 138.
398. The AAV particle of any one of embodiments 269-371, 375, 380-383, 385, 387, 389, 390, 393, or 394, wherein the AAV capsid variant comprises the amino acid sequence of AEIGHDSPHKSG (SEQ ID NO: 6400), optionally wherein the amino acid sequence is present immediately subsequent to position 449 and replaces positions 450-455 (e.g., T450, 1451, N452, G453, S454, G455), numbered according to SEQ ID NO: 138.
399. The AAV particle of any one of embodiments 369-371, 375, 380-383, 385, 387, 389, 390, 393, 394, or 398, wherein the AAV capsid variant comprises the amino acid sequence of KAEIGHDSPHKSGQNQQT (SEQ ID NO: 1754), optionally wherein the amino acid sequence is present immediately subsequent to position 448 and replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460), numbered according to SEQ ID NO: 138.
400. The AAV particle of any one of embodiments 369-371, 375-379, 390, 391 , or 395, wherein the AAV capsid variant comprises the amino acid sequence of EKMSGSPHSKA (SEQ ID NO: 6401), optionally wherein the amino acid sequence is present immediately subsequent to position 450 and replaces positions 451-455 (e.g., 1451 , N452, G453, S454, G455), numbered according to SEQ ID NO: 138.
401. The AAV particle of any one of embodiments 369-371, 375-379, 390, 391 , or 395, wherein the AAV capsid variant comprises the amino acid sequence of KTEKMSGSPHSKAQNQQT (SEQ ID NO: 3241), optionally wherein the amino acid sequence is present immediately subsequent to position 448 and replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460), numbered according to SEQ ID NO: 138.
402. The AAV particle of any one of embodiments 369-371, 375-379, 390, 391 , or 395, wherein the AAV capsid variant comprises the amino acid sequence of HDSPHSKAQNL (SEQ ID NO: 6402), optionally wherein the amino acid sequence is present immediately subsequent to position 453 and replaces positions 456-458 (e.g., Q456, N457, Q458), numbered according to SEQ ID NO: 138.
403. lire AAV particle of any one of embodiments 369-371, 375-379, 390, 391, or 395, wherein the AAV capsid variant comprises the amino acid sequence of KTTNGHDSPHSKAQNLQT (SEQ ID NO: 4100), optionally wherein the amino acid sequence is present immediately subsequent to position 448 and replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456. N457, Q458, Q459, T460), numbered according to SEQ ID NO: 138.
404. The AAV particle of any one of embodiments 369-371, 375-379, 390, 391 , or 395, wherein the AAV capsid variant comprises the amino acid sequence of VNGHDSPHSKA (SEQ ID NO: 6403), optionally wherein the amino acid sequence is present immediately subsequent to position 450 and replaces positions 451-455 (e.g., 1451 , N452, G453, S454, G455), numbered according to SEQ ID NO: 138.
405. The AAV particle of any one of embodiments 369-371, 375-379, 390, 391 , or 395, wherein the AAV capsid variant comprises the amino acid sequence of KTVNGHDSPHSKAQNQQT (SEQ ID NO: 4062), optionally wherein the amino acid sequence is present immediately subsequent to position 448 and replaces positions 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, T460), numbered according to SEQ ID NO: 138.
406. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51 , 54-72, 69- 89, 91, 94-99, 102-104, 107, 1 10-112, 115-129, 168-202, 02, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, or 395, wherein the AAV capsid variant comprises an amino acid sequence encoded by: the nucleotide sequence of SEQ ID NO: 942; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 942; or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 942.
407. The AAV particle of any one of embodiments 1-9, 11 , 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105. 110, 111. 113-129, 203-248, 251, 252, 2.54-257, 2.60, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, or 395, wherein the AAV capsid variant comprises an amino acid sequence encoded by: the nucleotide sequence of SEQ ID NO: 3; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 3; or a nucleotide sequence comprising at least one, at least two, at
least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 3.
408. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91, 94-99, 102-104, 107, 110-112, 115-129, 168-202, 02, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288. 291-299, 327-363, 369-371, 375-379, 385, 386, 390. 391, 395, or 406, wherein the nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NO: 942; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 942; or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 942.
409. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88. 90-97, 100-103, 105, 110. 111, 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, or 407, wherein the nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NO: 3; a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, e.g., substitutions (e.g., conservative substitutions), but no more than ten modifications, e.g., substitutions (e.g., conservative substitutions), relative to the nucleotide sequence of SEQ ID NO: 3; or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 3.
410. The AAV particle of any one of embodiments 369-409, wherein the amino acid. sequence is present in loop IV, e.g., relative to the amino acid sequence of SEQ ID NO: 138.
411. The AAV particle of any one of embodiments 369-410, wherein the amino acid sequence is present immediately subsequent to position 448, 449, 450, 451, 452, 453, 454, or 455, numbered according to SEQ ID NO: 138.
412. The AAV particle of any one of embodiments 369-411, wherein the amino acid sequence replaces amino acids 449, 450, 451, 452, 453, 454, 455, 456, 457, 458, 459, and/or 460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and/or T460), numbered according to SEQ ID NO: 138.
413. The AAV particle of any one of embodiments 369-412, wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
414. The AAV particle of any one of embodiments 369-413, wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to SEQ ID NO: 138.
415. The AAV particle of any one of embodiments 1-2.3, 26-2.9, 32, 35-43, 46-51 , 54-72, 69- 89, 91, 94-99, 102-104, 107, 1 10-112, 115-129, 168-202, 02, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, 395, 406, 408, or 410-413, wherein the AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
416. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51 , 54-72, 69- 89, 91, 94-99, 102-104, 107, 1 10-112, 115-129, 168-202, 02, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, or 415, wherein the AAV capsid variant the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 981.
417. The AAV particle of embodiment 415 or 416, wherein the AA V capsid variant further comprises an amino acid other than I at position 451 , an amino acid other than N at position 452, and an amino acid oilier than G at position 453, numbered according to any one of SEQ ID NOs: 36, 138, or 981.
418. The AAV particle of any one of embodiments 415-417, wherein the AAV capsid variant further comprises E at position 451, R at position 452, and V at position 453, numbered according to any one of SEQ ID NOs: 36, 138, or 981 .
419. The AAV particle of any one of embodiments 415-418, wherein the .AAV capsid variant further comprises the substitutions I451E, N452R, and G453V, numbered according to any one of SEQ ID NOs: 36, 138, or 981 .
420. The AAV particle of any one of embodiments 415-419, wherein the AAV capsid variant comprises:
(i) E at position 451, R at position 452, and V at position 453, numbered according to any one of SEQ ID NOs: 36, 138, or 981; and
(ii) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to any one of SEQ ID NOs: 36, 138, or 981 (at amino acids 456-461, numbered according to SEQ ID NO: 36 or 981).
421. The AA V particle of embodiment 415 or 416, wherein the AAV capsid variant further comprises an amino acid other than I at position 451, an amino acid other than N at position 452, and/or G at position 453, numbered according to SEQ ID NO: 39 or 138.
422. The AAV particle of any one of embodiments 415, 416, or 421 , wherein the AAV capsid variant further comprises E at position 451, K at position 452, and/or M at position 453, numbered according to SEQ ID NO: 138 or 39.
423. The AAV particle of any one of embodiments 415, 416, 421, or 422, wherein the AAV capsid variant further comprises the substitutions I451E, N452K, and G453M, numbered according to SEQ ID NO: 39 or 138.
424. The .AAV particle of any one of embodiments 415, 416, or 421-423, wherein the AAV capsid variant comprises:
(i) E at position 451, K at position 452, and M at position 453, numbered according to SEQ ID NO: 39 or 138; and
(ii) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 39 or 138.
425. The AAV particle of embodiment 415 or 416, wherein the .AAV capsid variant further comprises an amino acid other than S at position 454, an amino acid other than G at position 455, and/or Q at position 458, numbered according to SEQ ID NO: 138.
426. The .AAV particle of any one of embodiments 415, 416, or 425, wherein the AAV capsid variant further comprises H at position 454, D at position 455, and/or L at position 458, numbered according to SEQ ID NO: 138.
427. The AAV particle of any one of embodiments 415, 416, 425, or 426, wherein the AAV capsid variant further comprises the substitutions S454H, G455I), and Q458L, numbered according to SEQ ID NO: 138.
428. The AAV particle of any one of embodiments 415, 416, or 425-427, wherein the AAV capsid variant comprises:
(i) H at position 454, D at position 455, and/or L at position 458, numbered according to SEQ ID NO: 138; and
(ii) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
429. The AA V particle of embodiment 415 or 416, wherein the AAV capsid variant further comprises an amino acid other than I at position 451, an amino acid other than S at position 454, and/or an amino acid other than G at position 455, numbered according to SEQ ID NO: 52 or 138.
430. The .AAV particle of any one of embodiments 415, 416, or 429, wherein the AAV capsid variant further comprises V at position 451, H at position 454, and/or D at position 455, numbered according to SEQ ID NO: 52 or 138.
431 . The AAV particle of any one of embodiments 415, 416, 429, or 430, wherein the AAV capsid variant further comprises the substitutions I451V, S454H, and/or G455D, numbered according to SEQ ID NO: 52 or 138.
432. The AAV particle of any one of embodiments 415, 416, or 429-431, wherein the .AAV capsid variant comprises:
(i) V at position 451, H at position 454, and/or D at position 455, numbered according to SEQ ID NO: 52 or 138; and
(ii) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 52 or 138.
433. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111, 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, or 414, wherein the AAV capsid variant the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to the amino acid sequence of SEQ ID NO: 138.
434. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 1 11, 113-129, 203-248, 251 , 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, or 433, comprising the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid, sequence is present immediately subsequent to position 453, numbered according to the amino acid sequence of SEQ ID NO: 982.
435. The AAV particle of any one of embodiments 1 -9, 11, 12-2.2, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 1 11, 113-129, 203-248, 251 , 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433, or 434, wherein the AAV capsid variant the amino acid sequence of SPHKSG (SEQ ID NO: 946), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 982.
436. The AAV particle of any one of embodiments 369-435, wherein the AAV capsid variant composes:
(i) the amino acid sequence of HDSPHSKA (SEQ ID NO: 4486), which is present immediately subsequent to position 453; and
(ii) a deletion of amino acids SG at position 454 and 455; wherein (i) and (ii) are numbered according to SEQ ID NO: 138.
437. The AAV particle of any one of embodiments 369-436, wherein the AAV capsid variant comprises the amino acids HD at position 454 and 455, and further comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), which is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138.
438. The AAV particle of any one of embodiments 433-435, wherein the AAV capsid variant further composes an amino acid other than T at position 450, an amino acid other than I at position 451, and an amino acid other than N at position 452, numbered according to SEQ ID NO: 138 or 982.
439. The AAV particle of any one of embodiments 433-435 or 438, wherein the AAV capsid variant further comprises A at position 450, E at position 451, and I at position 452, numbered according to SEQ ID NO: 138 or 982.
440. The AAV particle of any one of embodiments 433-435, 438, or 439, wherein the AAV capsid variant further comprises the substitutions T450A, I451E, and N452I, numbered according to SEQ ID NO: 138 or 982.
441. The AAV particle of any one of embodiments 433, 434, or 438-440, wherein the AAV capsid variant comprises:
(i) A at position 450, E at position 451, and. I at position 452, numbered according to SEQ ID NO: 138 or 982; and
(ii) the amino acid sequence of HDSPHK (SEQ ID NO: 2), which is present immediately subsequent to positions 453, numbered according to SEQ ID NO: 138 or 982.
442. The AAV particle of any one of embodiments 1-22, 25-27, 31 , 34-42, 45-50, 53-63, 69, 79, 83-86, 91-98, 102, 103, 110, 111, 118-129, 369-371, 384, 385, 390, 393, 395, 410-413,
wherein the AAV capsid variant the amino acid sequence of SPHKYG (SEQ ID NO: 966), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to the amino acid sequence of SEQ ID NO: 138.
443. An adeno-associated virus (AAV) particle comprising an .AAV capsid variant comprising the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to the amino acid sequence of SEQ ID NO: 982, wherein the AAV particle further comprises viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein.
444. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a. viral genome comprising a nucleic acid encoding a modulatory polynucleotide tor reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to the amino acid sequence of SEQ ID NO: 981 .
445. An adeno-associated virus (AAV) particle comprising an AAV capsid variant comprising the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to the amino acid sequence of SEQ ID NO: 37, and optionally further comprising:
(i) one, two, or all of an amino acid other than T at position 450, an amino acid other than I at position 541, and/or an amino acid other than N at position 452, numbered according to SEQ ID NO: 138 or 37;
(ii) one, two, or all of A at position 450, E at position 451, and/or I at position 452, numbered according to SEQ ID NO: 138 or 37; wherein the AAV particle further comprises a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein.
446. An adeno-associated virus (AAV) particle comprising an AAV capsid variant comprising the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to the amino acid sequence of any one of SEQ ID NO: 36, 38-55, 57, or 59, wherein the AAV particle further comprises a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein.
447. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant further comprises:
(i) a modification in loop I, II, VI and/or VIII; and/or
(ii) a substitution at position K449, e.g., a K449R substitution, numbered according to SEQ ID NO: 138.
448. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, e.g., substitutions (e.g., conservative substitutions), but not more than 30, not more than 20, or not more than 10 modifications, e.g., substitutions (e.g., conservative substitutions), relative to the amino acid sequence of SEQ ID NO: 138.
449. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than 30, not more than 20, or not more than 10 different amino acids relative to the amino acid sequence of SEQ ID NO: 138.
450. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138.
451. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid, sequence with at least 98% identity to SEQ ID NO: 138.
452. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises an amino acid sequence encoded by a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at. least. 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 137.
453. The AAV particle of any one of the preceding embodiments, wherein the nucleotide sequence encoding the capsid vanant comprises at least 80% (e.g., at least 80%, at. least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 137.
454. The AAV particle of any one of the preceding embodiments, wherein the AAV capsid variant comprises a VP 1 protein, a VP2 protein, a VP3 protein, or a combination thereof.
455. The AAV particle of any one of embodiments 1-454, wherein the AAV capsid variant comprises the amino acid sequence corresponding to positions 138-742, e.g., a VP2, of SEQ ID NO: 981, 982, 36, or 4, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
456. The AAV particle of any one of embodiments 1-455, wherein the AAV capsid variant comprises the amino acid sequence corresponding to positions 203-742, e.g., a VP3, of SEQ ID NO: 981, 982, 36, or 4, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
457. The AAV particle of any one of embodiments 1-456, wherein the AAV capsid variant comprises an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138.
458. The AAV particle of any one of embodiments 1-457, wherein the AAV capsid variant comprises an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138.
459. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91 , 94-99, 102-104, 107, 110-112, 115-129, 168-202, 240-247, 249, 250, 253-263, 266, 272-281 , 286, 288, 291 -299, 327-363, 369-371, 375-379, 385, 386, 390, 391 , 395, 406, 408, 410-413, 415-432, 444, or 446-458, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein:
(i) the at least 3 consecutive amino acids comprise SPH;
(ii) the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700):
(lii) the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701); or (iv) the at least 6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941); wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 981; (b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 981; (c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 981 ; or (d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences in (a)-(c).
460. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91, 94-99, 102-104, 107, 110-112, 115-129, 168-202, 240-247, 249, 250. 253-2.63. 266, 272-281, 286, 288, 291-299, 327-363, 369-371 , 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, or 446-459, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein:
(i) the at least 3 consecutive amino acids comprise SPH;
(ii) the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700);
(iii) the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701); or
(iv) the at. least 6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941); wherein the AAV capsid variant comprises an amino acid sequence has at least 90%
(e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to the amino acid sequence of SEQ ID NO: 981.
461. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91, 94-99, 102-104, 107, 110-112, 115-129, 168-202, 240-247, 249, 250, 253-263, 266,
272-281, 286, 288, 291-299, 327-363, 369-371 , 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, or 446-460, wherein the AAV capsid variant comprises one or two, but no more than three substitutions relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941 ), wherein the AAV capsid variant comprises:
(a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 981;
(b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ
ID NO: 981:
(c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 981; or
(d) an amino acid sequence with at least 90% (e.g,, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences in (a)-(c).
462. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51 , 54-72, 69- 89, 91, 94-99, 102-104, 107, 1 10-112, 115-129, 168-202, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, or 446-461, wherein the AAV capsid variant comprises one or two, but no more than three substitutions relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the AAV capsid variant comprises an amino acid sequence at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981 ,
463. The AAV particle of any one of embodiments 459-462, wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138 or 981.
464. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111, 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, or 447-458, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein:
(i) the at least 3 consecutive amino acids comprise HDS;
(ii) the at least 4 consecutive amino acids comprise IIDSP (SEQ ID NO: 4702);
(iii) the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703); or
(iv) tire at least 6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2); wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO; 982; (b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982; (c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982; or (d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences in (a)-(c).
465. The AAV particle of any one of embodiments 1-9, 1 1 , 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111, 113-129, 203-248, 251, 252, 254-257, 260, 261, 2.64-280, 2.83-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, or 464, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein:
(i) the at least 3 consecutive amino acids comprise HDS;
(ii) the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO: 4702);
(iii) the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703); or
(iv) the at. least 6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2); wherein the AAV capsid variant comprises an amino acid sequence at: least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 982.
466. The AAV particle of any one of embodiments 1-9, 1 1, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105. 110, 111. 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, 464, or 465, wherein the AAV capsid variant comprises one or two, but no more than three substitutions relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the AAV capsid variant comprises:
(a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982;
(b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982;
(c) a VP3 protein comprising the amino acid, sequence of positions 203-742. of SEQ ID NO: 982; or
(d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences m (a)-(c).
467. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111 , 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, or 464-466, wherein the AAV capsid variant comprises one or two, but no more than three substitutions relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the AAV capsid variant comprises an amino acid sequence at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 982.
468. The AAV particle of any one of embodiments 464-468, wherein the amino acid sequence is present immediately subsequent to position 453, numbered according to SEQ ID NO: 138 or 982.
469. The AAV particle of any one of embodiments 1-468, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981 or 982, or an amino acid sequence with at least 80% (e.g., at least 80%, at least. 85%, at. least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity- thereto.
470. The AAV particle of any one of embodiments 1-469, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three modifications, e.g., substitutions (e.g., conservative substitutions), but not more than 30, not more than 20 or not more than 10 modifications, e.g., substitutions (e.g., conservative substitutions), relative to the amino acid sequence of SEQ ID NO: 981 or 982.
471 . The AAV particle of any one of embodiments, 1-470, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three, but not more than 30, not more than 20 or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 981 or 982.
472. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91, 94-99, 102-104, 107, 110-112, 115-129, 168-202, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371 , 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, 446-463, or 469-471, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981, or an amino acid, sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
473. The AAV particle of any one of embodiments 1-2.3, 26-29, 32, 35-43, 46-51 , 54-72, 69- 89, 91, 94-99, 102-104, 107, 1 10-112, 115-129, 168-202, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, 446-463, or 469-472, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three modifications, e.g., substitutions (e.g., conservative substitutions), but not more than 30, not more than 20 or not more than 10 modifications, e.g., substitutions (e.g., conservative substitutions), relative to the amino acid sequence of SEQ ID NO: 981.
474. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91, 94-99, 102-104, 107. 110-112, 115-129, 168-202, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291 -299, 327-363, 369-371, 375-379, 385, 386, 390, 391 , 395, 406, 408, 410-413, 415-432, 444, 446-463, or 469-473, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three, but not more than 30, not more than 20 or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 981.
475. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111 , 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, or 464-471, wherein
the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
476. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111, 113-129, 203-248, 251, 252, 254-257, 260, 261, 2.64-280, 2.83-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, 464-471 , or 475, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three modifications, e.g., substitutions (e.g., conservative substitutions), but not more than 30, not more than 20 or not more than 10 modifications, e.g.. substitutions (e.g., conservative substitutions), relative to the amino acid sequence of SEQ ID NO: 982.
477. The AAV particle of any one of embodiments 1 -9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111, 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-2.80, 283-2.87, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, 464-471, 475, or 476, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three, but not more than 30, not more than 20 or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 982.
478. The .AAV particle of any one of embodiments 1-477, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983 or 984, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%), at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
479. The AAV particle of any one of embodiments 1-478, wherein the nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NOs: 983 or 984, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
480. The AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51, 54-72, 69- 89, 91, 94-99, 102-104, 107, 110-112, 115-129, 168-202, 02, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, 446-463, 469-474, 478, or 479, wherein the nucleotide sequence encoding the capsid variant comprises the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
481. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42, 44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105, 110, 111 , 113-129, 203-248, 251, 252, 254-257, 260, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386, 390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, 464-471, or 475- 479, wherein the nucleotide sequence encoding the capsid variant comprises tire nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
482. The AAV particle of any one of the preceding embodiments, wherein the nucleotide sequence encoding the capsid variant is codon optimized.
483. An AAV particle of any one of embodiments 1-23, 26-29, 32, 35-43, 46-51 , 54-72, 69- 89, 91, 94-99, 102-104, 107, 1 10-112, 115-129, 168-202, 02, 240-247, 249, 250, 253-263, 266, 272-281, 286, 288, 291-299, 327-363, 369-371, 375-379, 385, 386, 390, 391, 395, 406, 408, 410-413, 415-432, 444, 446-463, 469-474, 478-480, or 482, and further comprising an amino acid sequence at least 95% identical to SEQ ID NO: 981,
484. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a. viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981.
485. The AAV particle of embodiment 483 or 484, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence at least 90%, at least 95%, or at least 99% identical thereto.
486. The AAV particle of any one of embodiments 1-9, 11, 12-22, 24, 26, 28, 30, 33, 35-42,
44, 46-50, 52, 54-77, 83-88, 90-97, 100-103, 105. 110, 111. 113-129, 203-248, 251, 252,
2.54-257, 2.60, 261, 264-280, 283-287, 289-299, 327-363, 369, 369, 371, 380-383, 385, 386,
390, 392, 395, 407, 409-412, 414, 433-435, 438-441, 443, 445, 447-458, 464-471 , 475-479,
481, or 482, and further comprising an amino acid sequence at least 95% identical to SEQ ID NO: 982.
487. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982.
488. The AA V particle of embodiment 486 or 487, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence at least 90%, at least 95%, or at least 99% identical thereto.
489. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NOs: 983 or 984, or a nucleotide sequence at least 95% identical thereto.
490. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid, variant comprises the amino acid sequence of any one of SEQ ID NOs: 4 or 36-59, optionally wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 4 or 36.
491. An adeno-associated virus (AAV) particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) and a viral genome comprising a nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2) mRNA, thereby reducing or eliminating ATXN2 protein, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of any one of SEQ ID NOs: 12-35, or a nucleotide sequence at least 95% identical thereto.
492. The AAV particle of 490 or 491, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of any one of SEQ ID NOs: 12-35, or a nucleotide sequence at least 95% identical thereto.
493. The AAV particle of any one of embodiments 1-299, 369-371, or 375-492, which has an increased tropism for a CNS cell or ti ssue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising a capsid comprising the amino acid sequence of SEQ ID NO: 138.
494. The AA V particle of any one of embodiments 1-129, 168-299, 369-371 , or 375-493, which transduces a brain region, e.g., a midbrain region (e.g., the hippocampus, or thalamus) or the brain stem, optionally wherein the level of transduction is at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65 -fold greater as compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay, e.g., an immunohistochemistry assay or a qPCR assay, e.g., as described in Example 2.
495. The AAV particle of any one of embodiments 1-129, 168-299, 369-371 , or 375-494, which transduces a brain region, e.g., a midbrain region (e.g., the hippocampus, or thalamus) or the brain stem, optionally wherein the level of transduction is at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65-fold greater as compared to an .AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay, e.g., an immunolnstochemistiy assay or a qPCR assay, e.g., as described in Example 2.
496. The AAV particle of any one of embodiments 1-129, 168-299, 369-371 , or 375-495, which is enriched at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at
least 10-fold, in the brain compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1.
497. The AAV particle of any one of embodiments 1-129, 168-299, 369-371 , or 375-496, which is enriched at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80 or at least 85-fold, in the brain compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1.
498. The AA V particle of any one of embodiments 1-129, 168-299, 369-371 , or 375-497, which is enriched in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., mouse), e.g., as compared to an AAV particle comprising a capsid of SEQ ID NO: 138.
499. The AAV particle of any one of embodiments 1-129, 168-299, 369-371, or 375-498, which is enriched at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least
40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least
80, at least 85, at least 90, at least 95, at least 100, at least 105, at least 115, at least 120, at least 125, at least 130, at least 135, at least 140, at least 145, at least 150, at least 155, at least
160, at least 165, at least 170, at least 175, at least 180, at least 190, at least 200, at least 205, or at least 210-fold, in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., mouse), compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1 or 5.
500. The AA V particle of embodiment. 498 or 499, wherein the at least two to at least three species are Macaca fasciculans, Chlorocebus sabaeus, Callithrix jacchus, and/or mouse (e.g., BALB/c mice, C57B1/6 mice, and/or CD-I outbred mice).
501. The AAV particle of any one of embodiments 130-146, 369, 410-414, 447-454, 457, 458, 482, or 493, which is enriched at least 2, at least 2.5, at least 3, at least 3.5, at least 4, at least 4.5, at least 5, at least 5.5, at least 6, at least 6.5, at least 7, at least 7.5, or at least 8-fold, in the brain compared, to an AAV particle comprising a capsid of SEQ ID NO: 981, e.g., when measured by an assay as described in Example 3.
502. The AAV particle of any one of embodiments 147-167, 369, 410-414, 447-454, 457, 458, 482, or 493, which is enriched at least 2, at least 2.5, at least 3, at least 3.5, at least 4, at least 4.5, at least 5, or at least 5.5 -fold, in the brain compared, to an AAV particle comprising a capsid of SEQ ID NO; 982, e.g., when measured by an assay as described in Example 3.
503. The AAV particle of any one of embodiments 1-129, 168-299, 369-371, or 375-500, which delivers an increased level of a payload to a brain region, optionally wherein the level of the payload is increased by at least 10, at least 12, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, or at least 70-fold, as compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2 or 8).
504. The AAV particle of any one of embodiments 1-12.9, 168-2.99, 369-371, 375-500, or 503, which delivers an increased level of viral genomes to a brain region, optionally wherein the level of viral genomes is increased by at least 5, at least 10, at least 15, at least 1.7, at least 18, at least 19, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, or at least 50-fold, as compared to an AAV particle comprising a. capsid of SEQ ID NO: 138, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2 or 8).
505. The AAV particle of embodiment 503 or 504, wherein the brain region is a midbrain region (e.g., the hippocampus or thalamus), frontal cortex, temporal cortex, motor cortex, cerebral cortex, caudate, putamen, dentate nucleus, substantia nigra, or the brainstem.
506. The AAV particle of any one of embodiments 1-129, 168-299, 369-371 , 375-500, or 503-505, which is enriched at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least. 35-fold, in the spinal cord compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1 or 8, optionall y wherein the region of the spinal cord is a thoracic spinal cord region, cervical spmal cord region, C5 ventral horn region, lumbar spinal cord region, or L5 ventral horn region.
507. The AAV particle of any one of embodiments 1-129, 168-299, 369-371, 375-500, or 503-506, which shows preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG).
508. The AAV particle of any one of embodiments 1-129, 168-299, 369-371, 375-500, or 503-507, which shows preferential transduction in a brain region relative to the liver.
509. The AAV particle of any one of embodiments 1-129, 168-299, 369-371, 375-500, or 503-508, which shows preferential transduction in a brain region relative to the transduction in the heart.
510. The AAV particle of any one of embodiments 1-129, 168-299, 369-371, 375-500, or 503-509, which shows preferential transduction in a brain region relative to the transduction in the dorsal root ganglia. (DRG) and the heart.
51 1. The AAV particle of any one of the preceding embodiments, which is capable of transducing non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes).
512. The AAV particle of embodiment 511, wherein the non-neuronal ceils comprise glial cells, oligodendrocytes (e.g., Olig2 positive oligodendrocytes), or astrocytes (e.g., Olig2 positive astrocytes).
513. The .AAV particle of any one of the preceding embodiments, which is capable of transducing Olig2 positive cells, e.g., Olig2 positive astrocytes or Olig2 positive oligodendrocyte s .
514. The AAV particle of any one of embodiments 369, 373, 447-454, 457, 458, or 482, which has increased tropism for a heart cell or tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of an AAV particle comprising a capsid of SEQ ID NO: 138.
515. The AAV particle of any one of embodiments 369, 373, 447-454, 457, 458. 482, or 514, which is enriched at least 4, at least 5, at least 8, at least 10, at least 11, at least 12, at least 13, at least 14, at least 18, at least 19, at least 20, at least 21 , at least 22, at least 24, at least 25, at least 27, at least 31, at least 33, or at least 34-fokl, in the heart compared to an AAV particle
comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 4.
516. The AAV particle of any one of embodiments 369, 374, 447-454, 457, 458, 482, which has an increased tropism for a muscle cell or tissue (e.g., a quadriceps cell or a quadriceps tissue), relative to the tropism of an AAV particle comprising a capsid comprising the amino acid sequence of SEQ ID NO: 138.
517. The AAV particle of any one of embodiments 369, 374, 447-454, 457, 458, 482, which is enriched, at least 4, at least 5, at least 8, at least 12, at least 17, at least 18, at least 20, at least 26, at least 27, at least 28, at least 30, or at least 36-fold, in the muscle compared to an AAV particle comprising a capsid of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 4.
518. The AAV particle of embodiment 516 or 517, wherein the muscle cell or tissue is a heart muscle (e.g., a heart ventricle or a heart atrium, or both), a quadriceps muscle, or both.
519. The AAV particle of any one of the preceding embodiments, which is isolated and/or recombinant.
[Embodiments 520-576 are intentionally absent.]
577. The AAV particle of any one of the preceding embodiments, wherein the encoded modulatory polynucleotide comprises an RNAi agent, optionally a dsRNA, siRNA, shRNA, pre-miRN A, pri-miRNA, miRNA, stRNA, IncRNA, piRNA, or snoRNA, or a combination thereof.
[Embodiments 578-583 are intentionally absent.]
584. The AAV particle of embodiment 577, wdierein the modulatory polynucleotide comprises an siRNA.
[Embodiments 585 is intentionally absent.]
586. The AAV particle of any one of the preceding embodiments, wherein the viral genome comprises a promoter operably linked to the nucleic acid sequence encoding the modulators' polynucleotide.
587. The AAV particle of embodiment 586, wherein the promoter is a human elongation factor la-subunit (EFla) promoter, cytomegalovirus (CMV) promoter, a chicken p-actin (CBA) promoter, a CAG promoter, a CAG derivative promoter, a. CMV immediate-early enhancer and/or promoter, a CMV promoter, a β glucuronidase (GUSB) promoter, a ubiquitin C (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF-β) promoter, an intercellular adhesion molecule 2 (ICAM-2) promoter, a. synapsin (Syn) promoter, a methyl- CpG binding protein 2 (MeCP2) promoter, a Ca2+/calmodulin-dependent protein kinase II (CaMKII) promoter, a metabotropic glutamate receptor 2 (mGluR2) promoter, a neurofilament light (NFL) or heavy (NFH) promoter, a P-globin minigene nβ2 promoter, a. preproenkephalin (PPE) promoter, an enkephalin (Enk) and excitatory.' amino acid transporter 2 (EAAT2) promoter , a glial fibrillary acidic protein (GFAP) promoter, a myelin basic protein (MBP) promoter, a cardiovascular promoter (e.g., aMHC, cTnT, and CMV-MLC2k), a liver promoter (e.g., hAAT, TBG), a skeletal muscle promoter (e.g., desmin, MCK, C512) or a fragment, e.g., a truncation, or a functional variant thereof.
[Embodiments 588 and 589 are intentionally absent.]
590. The AAV particle of any one of embodiments 586-589, wherein the viral genome further comprises a polyadenylation (poly A) sequence.
591. The AAV particle of any one of embodiments 586-590, wherein the viral genome further comprises an inverted terminal repeat (ITR) sequence.
592. The AAV particle of any one of embodiments 586-591, wherein the viral genome comprises an ITR sequence positioned 5’ relative to the nucleic acid sequence encoding the modulatory polynucleotide .
593. The AAV particle of any one of embodiments 586-592, wherein the viral genome comprises an ITR sequence positioned 3’ relative to the nucleic acid sequence encoding the modulatory polynucleotide.
594. The AAV particle of any one of embodiments 586-593, wherein the viral genome comprises an ITR sequence positioned 5" relative to the nucleic acid sequence encoding the modulatory polynucleotide and. an ITR sequence positioned 3’ relative to the nucleic acid sequence encoding the modulatory polynucleotide.
595. The AA V particle of any one of embodiments 586-594, wherein the viral genome further comprises an enhancer, a Kozak sequence, an intron region, and/or an exon region.
[Embodiments 596-615 are intentionally absent.]
616. The AAV particle of any one of embodiments 586-595, wherein the viral genome further comprises a nucleotide sequence encoding a Rep protein, e.g., a non-structural protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, a Rep52 protein, and/or a. Rep40 protein (e.g,, a. R.ep78 and a. Rep52 protein).
617. The AAV particle of any one of embodiments 586-615, wherein the AAV particle further composes a. nucleotide sequence encoding a. Rep protein, e.g., a non-stmctural protein, wherein the Rep protein comprises a Rep78 protein, a. Rep68 protein, a Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 and a Rep52 protein).
618. The AA V particle of embodiment 616 or 617, wherein the Rep78 protein, the Rep68 protein, the Rep52 protein, and/or the Rep40 protein are encoded by at least one Rep gene.
619. The AAV particle of any one of embodiments 586-618, wherein the viral genome further comprises a. nucleic acid sequence encoding the AAV capsid variant of the AAV particle of any one of embodiments 1-519, 566, or 574.
620. The AAV particle of any one of embodiments 575-519, wherein the AAV particle is an isolated and/or recombinant AAV particle.
[Embodiment 621 is intentionally absent.]
622. A cell, e.g., a host cell, comprising the AAV particle of any one of the preceding embodiments.
623. lire cell of embodiment 622, wherein the cell is a mammalian cell or an insect cell.
[Embodiment 624 is intentionally absent.]
625. The cell of embodiment 622 or 623, wherein the cell is a cell of a brain region or a spinal cord region, optionally a cell of the brain stem, hippocampus, or thalamus.
626. lire cell of embodiment 624 or 625, wherein the cell is a neuron, a sensory neuron, a motor neuron, an astrocyte, a glial cell, or an oligodendrocyte.
627. A method of making an AAV particle, comprising
(i) providing a host cell comprising a viral genome encoding a modulatory polynucleotide for reducing or eliminating expression of A taxin-2 (ATXN2) mRNA; and
(ii) incubating the host cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant as described in any one of embodiments 1-620; thereby making the AAV particle.
628. The method of embodiment 627, further comprising, prior to step (i), introducing a first nucleic acid molecule comprising the viral genome into the host cell.
629. The method of embodiment 628, wherein the host cell comprises a second nucleic acid encoding the capsid variant.
630. The method of embodiment 629, wherein the second nucleic acid molecule is introduced into the host cell prior to, concurrently with, or after the first nucleic acid molecule.
631. A pharmaceutical composition comprising the AAV particle of any one of embodiments 1-620, and a pharmaceutically acceptable excipient.
632. A method of delivering a payload to a. cell or tissue (e.g., a CNS cell or CNS tissue), comprising administering an effective amount of the pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620.
[Embodiments 633-635 are intentionally absent.]
636. The method of embodiment 632, wherein the cell or tissue is within a subject.
637. The method of embodiment 636, wherein the subject has, has been diagnosed with having, or is at risk of having a genetic disorder, e.g,, a monogenic disorder or a polygenic disorder.
638. The method of embodiment 636 or 637, wherein the subject has, has been diagnosed with having, or is at risk of a disorder associated with aberrant expression or activity of ATXN2.
639. The method of any one of embodiments 636-638, wherein the subject has, has been diagnosed with having, or is at risk of having a muscular disorder or a neuromuscular disorder.
640. The method of any one of embodiments 636-639, wherein the subject has, has been diagnosed with having, or is at risk of having spinocerebellar ataxia type 2 (SCA2).
641. A method of treating a subject having or diagnosed with having a genetic disorder, e.g., a monogenic disorder or a. polygenic disorder, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620.
642. A method of treating a. subject having or diagnosed with having a. muscular disorder or a neuromuscular disorder associated with aberrant expression or activity of ATXN2, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620.
643. A method of treating a. subject having or diagnosed with having a. spinocerebellar ataxia type 2 (SCA2), comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620.
[Embodiments 644 and 645 are intentionally absent.]
646. The method of any one of embodiments 641-643, where treating comprises prevention of progression of the disease or disorder in the subject.
647. The method of embodiment 636-646, wherein the subject is a human.
648. The method of any one of embodiments 636-647, wherein the AAV particle is administered to the subject intravenously, intrathecally, intraarterially, or intramuscularly.
649. The method of any one of embodiments 636-648, wherein the AAV particle is administered to the subject via focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
650. The method of any one of embodiments 636-649, wherein the AAV particle is administered to the subject intravenously.
[Embodiment 651 is intentionally absent.]
652. The method of any one of embodiments 636-651 , wherein the AAV particle is administered to the subject intraarterially.
653. The method of any one of embodiments 648-652, wherein administration of the AAV particle results in a decreased presence, level, and/or activity of an ATXN2 gene, mRMA, protein, or combination thereof.
[Embodiment 654 is intentionally absent.]
655. The pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620 for use in a method of delivering a modulatory polynucleotide for reducing or elimination expression of ATXN2 to a. cell or tissue.
656. The pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620 for use in a method of treating a genetic disorder, a muscular disorder, or a neuromuscular disorder.
657. The pharmaceutical composition of embodiment 631 or the AAV particle of any one of embodiments 1-620 for use in the manufacture of a medicament.
658. Use of the pharmaceutical composition of embodiment 631 or the AAV particle of anyone of embodiments 1-620 in the manufacture of a medicament.
659. Use of the pharmaceutical composition of embodiment. 631 or the AAV particle of anyone of embodiments 1-620 in the manufacture of a medicament for treating a genetic disorder, a muscular disorder, or a neuromuscular disorder.
660. The AAV particle, pharmaceutical composition, cell, method, or use or any one of the preceding embodiments for reducing or eliminating expression of ATXN2 (e.g., of ATXN2 mRNA. and/or protein).
[Embodiments 661 -664 are intentionally absent.]
665. The AA V particle of embodiment 660, wherein the AAV capsid vanant comprises (a) a VP 1 protein comprising the amino acid sequence of SEQ ID NO: 982 or a. sequence at least 90% identical thereto; (b) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or a sequence at least 90% identical thereto; or (c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or a sequence at least 90% identical thereto; or wherein the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a sequence at least 90% identical thereto.
666. The AAV particle of any one of embodiments 660-664, wherein the AAV capsid variant comprises no more than three amino acid substitutions relative to the amino acid sequence of
HDSPHK (SEQ ID NO: 2), wherein the AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 982.
[Embodiments 666-674 are intentionally absent.]
675. The AAV particle of any one of the preceding embodiments, wherein the encoded modulatory polynucleotide comprises a molecular scaffold, comprising a 5’ flanking region, a loop region, and a. 3' flanking region, optionally wherein
(a) the 5’ flanking region comprises any one of SEQ ID NOs: 6413-6416; or a nucleotide sequence at least 75%, at least 80%, at least 85%, at least 90%, at least 95% at least 96%, at least 97%, at least 98%, or at least 99% identical to any one of SEQ ID NOs: 6413-6416;
(b) the loop region comprises any one of SEQ ID NOs: 6417-6421; or a nucleotide sequence at least 75%, at least 80%, at least 85%, at least 90%, at least 95% at least 96%, at least 97%, at least 98%, or at least 99% identical to any one of SEQ ID NOs: 6417-6421; and
(c) the 3’ flanking region comprises any one of SEQ ID NOs: 6422-6427; or a nucleotide sequence at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical to any one of SEQ ID NOs: 6422-6427.
676. The AAV particle of embodiment 675, wherein:
(a) the 5’ flanking region of the molecular scaffold comprises SEQ ID NO: 6414 or SEQ ID NO: 6415;
(b) the loop region of the molecular scaffold comprises SEQ ID NO: 6417, SEQ ID NO: 6418, or SEQ ID NO: 6421 ; and
(c) the 3’ flanking region of the molecular scaffold comprises SEQ ID NO: 6423, SEQ ID NO: 6424. or SEQ ID NO: 6425.
677. The AAV particle of embodiment 675, wherein:
(a) the 5’ thinking region of the molecular scaffold comprises SEQ ID NO: 6414, the loop region of the molecular scaffold comprises SEQ ID NO: 6417, and the 3" flanking region of the molecular scaffold comprises SEQ ID NO: 6423;
(b) the 5’ flanking region of the molecular scaffold comprises SEQ ID NO; 6415, the loop region of the molecular scaffold comprises SEQ ID NO: 6421, and the 3’ flanking region of the molecular scaffold comprises SEQ ID NO: 6425; or
(c) the 5’ flanking region of the molecular scaffold comprises SEQ ID NO: 6414, the loop region of the molecular scaffold comprises SEQ ID NO: 6417, and the 3’ flanking region of the molecular scaffold comprises SEQ ID NO: 6424; or
(d) the 5’ flanking region of the molecular scaffold comprises SEQ ID NO: 6414, the loop region of the molecular scaffold comprises SEQ ID NO: 6418, and the 3’ flanking region of the molecular scaffold comprises SEQ ID NO: 6423.
677a. The AAV particle of embodiment 677, wherein the modulatory polynucleotide comprises an siRNA comprising a passenger strand and a guide strand, wherein the passenger and guide strands are substantially complementary, and wherein the guide strand targets (e.g., reduces expression or silences expression of) ATXN2 mRNA, thereby reducing or eliminating ATXN2 protein.
677b. The AAV particle of embodiment 677a, wherein the passenger strand and the guide strand, are located, respectively, on a 5’ arm and a 3’ arm of a stem loop structure, wherein the passenger strand is located between the 5 ’ flanking region of the molecular scaffold and the loop region of the molecular scaffold and the guide strand is located between the loop region of the molecular scaffold and the 3’ flanking region of the molecular scaffold.
677c. The AAV particle of embodiment 677a, wherein the guide strand and the passenger strand are located, respectively; on a 5’ arm and a 3’ arm of a stem loop structure, wherein the guide strand is located between the 5’ flanking region of the molecular scaffold, and the loop region of the molecular scaffold and the passenger strand is located between the loop region of the molecular scaffold and the 3’ flanking region of the molecular scaffold.
677d. The AAV particle of any one of embodiments 677a-677c, wherein the passenger strand is 15-30 nucleotides in length, optionally 21-25 nucleotides in length.
677e. The AAV particle of any one of embodiments 677a-677d, wherein the guide strand is 15-30 nucleotides in length, optionally 21-25 nucleotides in length.
677f. Tlte AAV particle of embodiment 677e, wherein the passenger strand and guide strand are at least 70% complementary.
677g. The AAV particle of any one of embodiments 677a-677f, wherein the modulatory polynucleotide (e.g., the guide strand of the modulatory polynucleotide) binds to a coding region of ATXN2 mRNA.
677h. Tlte AAV particle of any one of embodiments 677a-677g, wherein the modulatory polynucleotide (e.g., the guide strand of the modulatory polynucleotide) binds to a noncoding region of ATXN2 mRNA,
678. The AAV particle of any one of embodiments 667-677h, wherein the viral genome further comprises a promoter operably linked to the nucleic acid sequence encoding the modulatory polynucleotide .
679. The AAV particle of any one of embodiments 667-678, wherein the viral genome further comprises an enhancer.
[Embodiments 680 and 681 are intentionally absent.]
682. The AAV particle of embodiment 678 or 679, wherein the promoter comprises a tissue- specific promoter.
683. The AAV particle of embodiment 678 or 679, wherein the promoter comprises a ubiquitous promoter.
684. lire AAV particle of any one of embodiments 678-683, wherein the promoter composes:
(i) an EF-la promoter, a CB promoter, a chicken β -actin (CBA) promoter, a CAG promoter, a CMV immediate-early enhancer and/or promoter, a CMV promoter, an Hl promoter, a β glucuronidase (GUSB) promoter, a ubiquitin C (UBC) promoter, a neuron- specific enolase (NSE), a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF-β) promoter, an intercellular adhesion molecule 2 (ICAM-2) promoter, a synapsin (Syn) promoter, a methyl-CpG binding protein 2 (MeCP2) promoter, a
Ca2+/calmodul in-depen dent protein kinase II (CaMKII) promoter, a metabotropic glutamate receptor 2 (mGluR2) promoter, a neurofilament light (NFL) or heavy (NFH) promoter, a p- globin minigene np2 promoter, a preproenkephalin (PPE) promoter, an enkephalin (Enk) and. excitatory amino acid transporter 2 (EAAT2) , a glial fibrillary acidic protein (GFAP) promoter, a myelin basic protein (MBP) promoter, a cardiovascular promoter (e.g., aMHC, cTnT, and CMV-MLC2k), a liver promoter (e.g., hAAT, TEG), a skeletal muscle promoter (e.g., desmin, MCK, C512) or a fragment, e.g., a truncation, or a functional variant thereof.
713. The AAV particle of any one of embodiments 667-713, wherein the viral genome further comprises an inverted terminal repeat (ITR) sequence.
714. The AAV particle of embodiment 713, wherein the ITR sequence is positioned 5’ relative to the nucleic acid encoding the modulatory polynucleotide.
715. The AAV particle of embodiment 713 or embodiment 714, wherein the ITR sequence is positioned 3’ relative to the nucleic acid encoding the modulatory polynucleotide.
716. The AA V particle of any one of embodiments 713-715, wherein the viral genome comprises an ITR positioned 5' relative to the nucleic acid encoding the modulatory polynucleotide and an ITR positioned 3 ’ relative to the nucleic acid encoding the modulatory polynucleotide.
717. The AAV particle of any one of embodiments 667-716, wherein the viral genome further comprises a polyadenylation (polyA) sregion.
718. The AAV particle of any one of embodiments 667-717, wherein the viral genome further comprises an intron region.
[Embodiments 719-726 are intentionally absent.]
727. The AAV particle of any one of embodiments 667-718, wherein the viral genome further comprises an exon region, e.g., at least one, at least two, or at least three exon regions.
728. The AAV particle of any one of embodiments 667-727, wherein the viral genome further comprises a Kozak sequence.
[Embodiments 729-748 are intentionally absent.]
749. An AAV particle comprising an AAV capsid variant and a viral genome, wherein the capsid variant comprises:
(a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or a sequence at least 90% identical thereto:
(b) a VP2 protein composing the amino acid sequence of positions 138-742 of SEQ
ID NO: 982 or a sequence at least 90% identical thereto; and/or
(c) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ
ID NO: 982 or a sequence at least 90% identical thereto and wherein the viral genome encodes a modulatory polynucleotide (e.g... siRNA) comprising a stem-loop structure comprising;
(i) a 5’ flanking region, a passenger strand, a loop region, a guide strand that targets ATXN2 mRNA, and a 3 ’ flanking region; or
(ii) a 5’ flanking region, a guide strand that targets ATXN2 mRNA, a loop region, a passenger strand, and a 3’ flanking region.
750. The AAV particle of embodiment 749, w herein the AAV capsid variant comprises no more than three amino acid substitutions relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 982.
751. The AAV particle of embodiment 749, wherein the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
[Embodiments 752-761 are intentionally absent.]
762. The AAV particle of any one of embodiments 660-751, further comprising a nucleic acid encoding a Rep protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, a. Rep52 protein, and/or a Rep40 protein.
763. The AAV particle of embodiment 762, wherein the Rep78 protein, the Rep68 protein, the Rep52 protein, and/or the Rep40 protein are encoded by at least one Rep gene.
764. A vector encoding the AAV particle of any one of embodiments 660-763.
765. A cell comprising the AAV particle of any one of embodiments 660-763 or the vector of embodiment 764.
766. The cell of embodiment 765, which is a mammalian cell, e.g., an HEK293 cell, an insect cell, e.g., an Sf9 cell, or a bacterial cell.
767. A method of making a recombinant AAV particle, the method comprising
(i) providing a host cell comprising a viral genome comprising the nucleotide sequence encoding a modulatory polynucleotide of the disclosure; and
(ii) incubating the host cell under conditions suitable to enclose the viral genome in a capsid variant comprising the amino acid sequence of SEQ ID NO: 982; thereby making the recombinant AAV particle.
768. The method of embodiment 767, further comprising, prior to step (i), introducing a first nucleic acid molecule comprising the viral genome into the host cell.
769. The method of embodiment 767 or embodiment 768, wherein the host cell comprises a second nucleic acid encoding the capsid variant.
770. The method of embodiment 769, further comprising introducing the second nucleic acid into the cell.
771. The method, of embodiment 769 or embodiment 770, wherein the second nucleic acid molecule is introduced into the host cell prior to, concurrently with, or after the first nucleic acid molecule.
772. The method of any one of embodiments 767-772, wherein the host cell comprises a mammalian cell, e.g., an HEK293 cell, an insect cell, e.g,, an Sf9 cell, or a bacterial cell .
773. A pharmaceutical composition comprising the AAV particle of any one of embodiments 660-763 and a pharmaceutically acceptable excipient.
774. A method of delivering a modulatory polynucleotide to a subject comprising administering an effective amount of the pharmaceutical composition of embodiment 773 or the AAV particle of any one of embodiments 660-763, thereby delivering the nucleic acid encoding a modulatory polynucleotide to the subject.
775. The method of embodiment 774, wherein the subject has, has been diagnosed with having, or is at risk of having a. disease associated with aberrant expression or activity of ATXN2, e.g., of a ATXN2 gene, ATXN2 mRNA, and/or ATXN2 protein.
776. The method of embodiment 774 or embodiment 775, wherein the subject has, has been diagnosed with having, or is at risk of having a spinocerebellar ataxia, type 2 (SCA2).
777. A method of treating a subject having or diagnosed with having a disease associated with aberrant ATXN2. protein expression or activity or mutant ATXN2 protein, composing administering an effective amount of the pharmaceutical composition of embodiment 773 or the AAV particle of any one of embodiments 1-751, thereby treating the disease associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein in the subject.
[Embodiments 778-779 are intentionally absent.]
780. A method of treating a. subject having or diagnosed with having SCA2, comprising administering an effective amount, of the pharmaceutical composition of embodiment 773 or the AAV particle of any one of embodiments 1-751, thereby treating SCA2 in the subject.
781. The method, of embodiment 779 or embodiment 780, wherein the subject has one or more mutations in ATXN2.
[Embodiments 782-785 are intentionally absent]
786. The method, of embodiment 781, wherein the subject’s ATXN2 gene has 32 or more CAG repeats (SEQ ID NO: 6435).
787. The method of embodiment 781, wherein the subject’s ATXN2 gene has 35 or more CAG repeats (SEQ ID NO: 6436).
788. The method of embodiment 781, w'herein the subject’s ATXN2 gene has 45 or more CA.G repeats (SEQ ID NO: 6434).
789. The method of any one of embodiments 774-789, wherein the subject has an increased level of ATXN2 activity as compared to a reference level.
790. The method of embodiment 789, wherein the reference level comprises the level of ATXN2 activity in a subject that does not have a disease associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein.
791. The method of any one of embodiments 777-790, wherein treating results in amelioration of at least one biomarker or symptom of the disease associated with aberrant AT.XN2 protein expression or activity or mutant AT.XN2 protein.
792. The method of embodiment 791, w'herein the at least one biomarker or symptom comprises progressive cerebellar ataxia, nystagmus, slow saccadic eye movements, ophthalmoparesis, parkinsonism, or a. combination thereof.
793. The method of any one of embodiments 774-792, wherein tire subject is a human.
794. The method of any one of embodiments 774-793, wherein the subject is a juvenile, e.g., between 6 years of age to 20 years of age.
795. The method, of any one of embodiments 774-793, wherein the subject is an adult, e.g., above 20 years of age.
796. The method of any one of embodiments 774-795. wherein the subject has one or more mutations in a ATXN2 gene, ATXM2 mRNA, and/or ATXN2 protein.
797. The method of any one of embodiments 774-796, wherein the AAV particle is administered to the subject intravenously, intracerebrally, via intrathalamic (ITH) administration, intramuscularly, intrathecally, intracerebroventricularly, via intraparenchymal administration, via focused ultrasound (FUS), e.g., coupled, with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration, or via intra-cisterna magna injection (ICM).
798. The method of any one of embodiments 774-796, wherein the AAV particle is administered via dual ITH and ICM administration.
799. The method of any one of embodiments 774-796, wherein the AAV particle is administered via intravenous injection, optionally wherein the intravenous injection is via focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
800. The method of any one of embodiments 774-799, wherein the AAV particle is administered to a ceil, tissue, or region of tire CNS, e.g., a region of the brain or spinal cord, e.g., the parenchyma, the cortex, substantia nigra, caudate cerebellum, striatum, corpus callosum, cerebellum, brain stem caudate-putamen, thalamus, superior colliculus, the spinal cord, or a combination thereof.
801. The method, of any one of embodiments 774-800, wherein the AAV particle is administered to at least two tissues, or regions of the CNS, e.g., bilateral administration.
802. The method of any one of embodiments 774-799, wherein the AAV particle is administered to the cerebral spinal fluid, the serum, or a combination thereof.
803. The method of any one of embodiments 774-802, further comprising performing a blood test, performing an imaging test, collecting a tissue biopsy, or collecting a blood or serum sample.
804. The method of any one of embodiments 774-803, which further comprises evaluating, e.g., measuring, the level of ATXN2 expression, e.g., ATXN2 gene, ATXN2 mRNA, and/or ATXN2 protein expression, in the subject, e.g., in a cell, tissue, or fluid, of the subject, optionally wherein the level of ATXN2 protein is measured by an assay described herein, e.g., an ELISA, a Western blot, or an immunohistochemistry assay.
805. The method of embodiment 804, wherein measuring the level of ATXN2 expression is performed prior to, during, or subsequent to treatment with the AAV particle.
806. The method, of embodiment 804 or embodiment 805, wherein the cell or tissue is a. cell or tissue of the central nervous system (e.g., parenchyma) or peripheral cell or tissue (e.g., the liver, heart, and/or spleen).
807. The method of any one of embodiments 774-806, wherein the administration results in decreased level of ATXN2 protein expression in a cell or tissue of the subject, relative to a. reference level, e.g., a subject that has not received treatment, e.g., has not been administered the AAV particle.
808. The method of any one of embodiments 774-807, which further comprises evaluating, e.g., measuring, the level of ATXN2 activity m the subject, e.g., in a cell or tissue of the subject, optionally wherein the level of ATXN2 activity is measured by an assay described herein.
808a. The method of any one of embodiments 774-808, wherein the administration results in an increase in the level of viral genomes (VG) per cell in a CNS cell or tissue (e.g. parenchyma).
809. The method of any one of embodiments 774-808a, wherein the administration results in a decrease in at least one of:
(i) the level of ATXN2 activity in a cell, tissue, and/or fluid, e.g., a CNS cell or tissue, of the subject: and/or
(ii) the level of ATXN2 mRNA expression in a cell, tissue, and/or fluid, e.g., a CNS cell or tissue, of the subject.
810. The method of any one of embodiments 774-809, further comprising administration of an additional therapeutic agent and/or therapy suitable for treatment or prevention of the disease associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein, e.g., spinocerebellar ataxia type 2.
811. The method of embodiment 810, wherein the additional therapeutic agent comprises riluzole.
812. The pharmaceutical composition of embodiment 773 or the AAV particle of any one of embodiments 1-751 for use in the treatinent of a disease associated, with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein, e.g., spinocerebellar ataxia type 2.
813. Use of an effective amount of the pharmaceutical composition of embodiment 773 or the AAV particle of any one of embodiments 660-763 in the manufacture of a medicament for the treatment of a. disease associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein, e.g., spinocerebellar ataxia type 2.
[073] The details of various aspects or embodiments of the present disclosure are set forth below. Other features, objects, and advantages of the disclosure will be apparent from the description and the claims. In the description, the singular forms also include the plural unless the context clearly dictates otherwise. Unless defined otherwise, all technical and scientific, terms used herein have the same meaning as commonly understood by one of ordinary skill in the art in the field of this disclosure. In the case of conflict, the present description will control.
BRIEF DESCRIPTION OF THE DRAWINGS
[074] FIG. 1 depicts biodistribution (VG/cell) in the motor cortex, frontal cortex, putamen, substantia nigra, dentate nucleus, cervical spinal cord ventral hom, DRG, liver, and heart in cynomolgus monkeys at 28 days post-IV injection of TTM-002.GBA_VG17-HA, AAV9.GBA_VG17-HA, or vehicle control.
[075] FIG. 2 depicts niRNA expression of GBA1 transgene in the motor cortex, frontal cortex, putamen, substantia nigra, dentate nucleus, cervical spinal cord ventral horn, DRG, liver, and heart in cynomolgus monkeys at 28 days post-IV injection of TTM- 002.GBA VG17-HA, AAV9.GBA VG17-HA, or vehicle control.
[076] FIG. 3A is a. graph showing the percentage of HA positive cells (percent of cells transduced by the indicated capsid variant) in the cortex in mice on the Y axis at the indicated doses on the X-axis (from highest dose to lowest dose: le 14 vg/kg, 3.2el 3 vg/kg, l el3 vg/kg, 3.2el2 vg/kg, or lei 2 vg/kg) at 28 days post-intravenous administration of AAV particles comprising the TTM-002 or TTM-027 AAV capsid variant. FIG. 3B is a graph showing the mRNA transgene expression relative to the housekeeping gene in the brains of the mice on the Y axis at the indicated doses on the X-axis (from highest to lowest dose: le 14 vg/kg, 3.2el3 vg/kg, lel.3 vg/kg, 3.2el 2 vg/kg, or 1 e 12 vg/kg) at 28 days post-intravenous administration of AAV particles comprising the TTM-002 or TTM-027 AAV capsid variant. [077] FIG. 4A is a graph showing the percentage of transduced cells having HA+ nuclei as measured by co-local izati on of nuclear H2B-HA staining and hematoxylin (%HA+ cells) in the indicated brain regions (temporal cortex, caudate, thalamus, or hippocampus) of African green monkey s. Measurements are at day 28 post-intravenous injection of AAV particles comprising the TTM-002 capsid variant or the AAV9 capsid control and a self- complementary genome encoding a histone 2B protein with an HA -tag at a dose of lel 3 VG/kg. FIG. 4B is a graph showing the percentage of HA+ cells among cells positive for the indicated marker (NeuN+ neurons, SM311+ Neurons, GFAP+ astrocytes, or Sox9 + astrocytes) in the indicated brain regions (temporal cortex, caudate, thalamus, or hippocampus) of African green monkey s. Measurements are at day 28 post-intravenous injection of AAV particles comprising the TTM-002 capsid variant and a self-complementary genome encoding a histone 2.B protein with an HA-tag at a dose of le!3 VG/kg. Plotted data, in FIGs. 4A-4B represent one slice per monkey (n=2). Quantitative image analysis was performed on le3 to le5 cells according to region size. All P values are derived from an unpaired two-tailed t-test.
[078] FIGs. 5A-5D are a series of graphs showing tropism of TTM-001 and TTM~002 relative to the AAV9 control in the brain and liver at 28 days post-intravenous administration in mice at a dose of le 13 VG/kg. FIG. 5A shows the viral genomes (VG)/diploid genomes (DG) in the brain for the AAV9 control, TTM-001 , or TTM-002; FIG. 5B shows brain RNA (fold vs AAV9) for the AAV9 control, TTM-001, or TTM-002; FIG. 5C shows the VG/DG in the liver for the .AAV 9 control, TTM-001, or TTM-002; and FIG. 5D shows the liver RNA (fold vs AAV9) for the AAV9 control, TTM-001, or TTM-002. Each data point represents an individual mouse and all plotted values represent mean ± SD (n=3). P values are derived from an unpaired two-tailed t-test.
DETAILED DESCRIPTION
Overview
[079] Described herein, inter alia, are compositions comprising an AAV capsid, variant tor delivery of modulatory polynucleotides for reducing or eliminating expression of ATXN2 to treat SCA2 or other disorders associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein. In some embodiments, the modulatory polynucleotides reduce or eliminate expression of ATXN2 mRNA, thereby reducing or eliminating ATXN2 protein. The AAV capsid variants described herein have enhanced tropism for a cell or tissue, e.g., for the delivery of a payload to said cell or tissue, for example a CNS tissue or a CNS cell.
[080] AAVs have proven to be usefill as a biological tool due to their relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing ceils) without integration into the host genome and without replicating, and their relatively benign immunogenic profile. Engineered adeno-associated virus (AAV) capsids with improved brain tropism represent an attractive solution to the limitations of CNS delivery. AAV-derived vectors are promising tools for clinical gene transfer because of their non -pathogenic nature, their low immunogenic profile, low rate of integration into the host genome, and long-term transgene expression in non-dividing cells. However, the transduction efficiency of naturally occurring AAVs in certain organs is too low for clinical applications, and capsid neutralization by pre-existing neutralizing antibodies may prevent treatment of a large proportion of patients. For these reasons, considerable efforts have been devoted to obtaining capsid variants with enhanced properties. Of many approaches tested so far, significant, advances have resulted from directed evolution of -AAV capsids using in vitro or in vivo selection of capsid variants created by capsid sequence randomization using either error- prone PCR, shuffling of various parent serotypes, or insertion of fully randomized short peptides at. defined positions.
[681] The genome of tire virus may be modified to contain a minim um of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver a desired payload . The genome of the virus may be modified to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to express or deliver a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA (thereby reducing or eliminating ATXN2 protein), which may be delivered to a target, cell, tissue, or organism. In some embodiments, the genome encodes a modulatory
polynucleotide for reducing or eliminating expression of human ATXN2 mRNA, thereby reducing or eliminating human ATXN2 protein. In some embodiments, the target cell is a CNS cell. In some embodiments, the target tissue is a CNS tissue. The target CNS tissue maybe brain tissue. In some embodiments, the brain target comprises caudate, putamen, thalamus, superior colliculus, cortex, and corpus collosum.
[682] Gene therapy presents an alternative approach for SCA2 and related diseases sharing single-gene etiology. AAVs are commonly- used, in gene therapy approaches as a result of a number of advantageous features. Without being bound by theory, it is believed, in some embodiments, that expression vectors, e.g., an adeno-associated viral vector (AAVs) or AAV particle, e.g., an AAV particle described herein, can be used to administer and/or deliver a modulatory poly-nucleotide, in order to achieve sustained, high concentrations, allowing for longer lasting efficacy, fewer dose treatments, broad biodistribution, and/or more consistent levels of the modulatory polynucleotide, relative to a non-AAV therapy.
[083] Provided herein are compositions and methods with improved features compared to prior A AV-mediated approaches, including (i) increased biodistribution throughout the CNS (e.g., the cortex, striatum, thalamus, cerebellum, brainstem, and/or spinal cord), and the periphery (e.g., the liver), (ii) elevated payload expression, e.g., modulatory polynucleotide expression, in multiple brain regions (e.g., cortex, thalamus, and brain stem) and the periphery (e.g., the liver): and/or (iii) increased modulatory polynucleotide in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., the cortex, striatum, thalamus, cerebellum, and/or brainstem), and/or fluid (e.g., CSF and/or serum), of the subject. In some embodiments, an AAV particle comprising an AAV capsid variant (e.g., comprising the amino acid sequence of SEQ ID NO: 4, 36, or 982), and a viral genome comprising a nucleic acid encoding a modulatory- poly nucleotide, yields increased biodistribution and/or elevated expression of the modulatory- polynucleotide in the cerebellum, e.g., Purkinje cells.
[084] Also provided herein, are compositions comprising an AAV capsid variant, e.g., an AAV capsid variant described herein for delivery, e.g., vectorized delivery, of a nucleic acid encoding modulatory- poly-nucleotides described herein, and methods of making and using the same. As demonstrated in the Examples below, certain AAV capsid variants described herein show multiple advantages over wild-type AAV9, including (i) increased penetrance through the blood brain barrier following intravenous administration, (ii) wider distribution throughout the multiple brain regions, e.g., frontal cortex, sensory- cortex, motor cortex, putamen, thalamus, cerebellar cortex, dentate nucleus, caudate, and/or hippocampus, and/or (iii) elevated payload expression in multiple brain regions. Without being bound by theory, it
is believed that these advantages may be due, in part, to the dissemination of the AAV capsid variants through the brain vasculature. In some embodiments, the AAV capsids described herein enhance tire delivery of a payload to multiple regions of the brain including for example, the frontal cortex, sensory cortex, motor cortex, putamen, thalamus, cerebellar cortex, dentate nucleus, caudate, and/or hippocampus.
[885] Thus, the compositions and methods described herein can be used in the treatment of disorders associated with mutant ATXN2 protein or aberrant ATXN2 protein expression or activity, such as SCA2. In some embodiments, the disclosure provides an AAV particle comprising one of the AAV capsid variants disclosed herein and an .AAV viral genome comprising a modulatory polynucleotide for use in treating disorders associated with mutant ATXN2 protein or aberrant ATXN2 protein expression or activity, such as SCA2.
I. Compositions
Adeno-associated viral (AAV) Particles
[086] AAVs have a genome of about 5,000 nucleotides in length and contain two open reading frames encoding the proteins responsible for replication (Rep) and the structural protein of the capsid (Cap). The open reading frames are flanked by two Inverted Terminal Repeat (ITR) sequences, which serve as the origin of replication of the viral genome. The wild-type AA V viral genome comprises nucleotide sequences for two open reading frames, one for the four non -structural Rep proteins (Rep78, Rep68, Rep52, Rep40, encoded by Rep genes) and one for the three capsid, or structural, proteins (VP1, VP2, VP3, encoded by capsid genes or Cap genes). The Rep proteins are important for replication and packaging, while the capsid proteins are assembled to create the protein shell of the AAV, or AAV capsid. Alternative splicing and alternate initiation codons and promoters result in the generation of four different Rep proteins from a single open reading frame and the generation of three capsid proteins from a single open reading frame. Though it varies by AAV serotype, as a. non-limiting example, for AAV9/11U.14 (SEQ ID NO: 123 of US 7,906, 11 1 , the contents of which are herein incorporated by reference in their entirety) VP1 refers to amino acids 1- 736, VP2 refers to amino acids 138-736, and VP3 refers to amino acids 203-736. In some embodiments, with reference to the amino acid sequence of SEQ ID NO: 982, 36, or 4, VP1 comprises amino acids 1-742, VP2 comprises amino acids 138-742, and VP3 comprises amino acids 203-742. In other words, VP I is the full-length capsid protein sequence, while VP2 and VP3 are shorter components of the whole. As a result, changes in the sequence in the VP3 region are also changes to VP1 and VP2; however, the percent difference as compared to the parent sequence will be greatest for VP3 since it is the shortest sequence of
the three. Though described here in relation to the amino acid sequence, the nucleic acid sequence encoding these proteins can be similarly described. Together, the three capsid proteins assemble to create the AAV capsid. While not wishing to be bound by theory, the AAV capsid typically comprises a molar ratio of 1:1: 10 of VP1 :VP2:VP3.
[087] The AAV particle typically requires a co-helper (e.g., adenovirus) to undergo productive infection in cells. In the absence of such helper functions, the AAV virions essentially enter host cells but do not integrate into the cells’ genome.
[088] AAV particles have been investigated for delivery of gene therapeutics because of several unique features. Non-limiting examples of the features include (i) the ability to infect both dividing and non-dividing cells; (ii) a broad host range for infectivity, including human cells; (iii) wild-type AAV has not been associated with any disease and has not been shown to replicate in infected ceils; (iv) the lack of cell-mediated immune response against the particle, and ( v) the non-integrative nature in a host chromosome thereby reducing potential for long-term genetic alterations. Moreover, infection with AAV particles has minimal influence on changing the pattern of cellular gene expression (Stilwell and Samulski el al, Biotechniques, 2003, 34, 148, the contents of which are herein incorporated by reference in their entirety).
[089] Typically, A AV particles for modulatory-' polynucleotide delivery may be recombinant viral particles which are replication defective as they lack sequences encoding functional Rep and Cap proteins within the viral genome. In some cases, the replication detective A AV particles may lack most or all coding sequences and essentially only contain one or two AAV ITR sequences and a. nucleic acid sequence encoding a modulatory polynucleotide.
[090] In some embodiments, the AAV particles of the present disclosure may be introduced into mammalian cells.
[091] AAV particles may be modified to enhance the efficiency of delivery. Such modified AAV particles of the present disclosure can be packaged efficiently and can be used to successfully infect the target cells at high frequency and with minimal toxicity.
[092] In other embodiments, AAV particles of the present disclosure may be used to deliver a modulatory polynucleotide to the central nervous system (see, e.g., U.S. Pat. No.
6, 180,613; the contents of which are herein incorporated by reference in their entirety) or to specific tissues of the CNS.
[093] It is understood that the compositions described herein may have additional conservative or non-essential amino acid substitutions, which do not have a substantial effect on their functions.
[094] In some embodiments, an AAV capsid variant disclosed herein comprises a modification in loop IV of AAV9, e.g., at positions between 449-460, e.g., at position 454 and/or 456, numbered relative to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, loop (e.g,, loop IV) is used interchangeably herein with the term variable region (e.g., variable region IV), or VR (e.g., VR-IV). In some embodiments loop IV comprises positions 449-475 (e.g., amino acids KTINGSGQNQQTLKFSVAGPSNMAVQG (SEQ ID NO: 6404)), numbered according to SEQ ID NO: 138. In some embodiments loop IV comprises positions 449-460 (e.g., amino acids KTINGSGQNQQT (SEQ ID NO: 6405)), numbered according to SEQ ID NO: 138. In some embodiments, loop IV or variable region IV (VR-IV) is as described m DiMattia et al. ‘"Structural Insights into the Unique Properties of the Adeno- Associated Virus Serotype 9,’" Journal of Virology, I2(86):6947-6958 (the contents of which are hereby incorporated by reference in their entirety), e.g., comprising positions 452-460 (e.g., NGSGQNQQT (SEQ ID NO: 4487)), numbered according to SEQ ID NO: 138.
[095] lire AAV particles and payloads of the disclosure may be delivered to one or more target cells, tissues, organs, or organisms. In some embodiments, the AAV particles demonstrate enhanced tropism for a target cell type, tissue or organ. As a non-limiting example, the AAV particle may have enhanced tropism for cells and tissues of the central or peripheral nervous systems (CNS and. PNS, respectively). In some embodiments, an AAV particle may, in addition, or alternatively, have decreased tropism tor a. cell-ty-pe, tissue or organ.
[096] In some embodiments, AAV particles are used as a biological tool due to a relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile. The genome of tire virus may be manipulated to contain a minimum of components for the assembly of a. functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver a desired payload.
[097] In some embodiments, the AAV particle is a recombinant AAV particle. In some embodiments, the wild-type AAV viral genome is a linear, single-stranded DNA (ssDNA) molecule approximately 5,000 nucleotides (nt) in length. In some embodiments, inverted terminal repeats (ITRs) cap the viral genome at both the 5’ and the 3’ end, providing origins
of replication for the viral genome. In some embodiments, an AAV viral genome comprises two ITR sequences. In some embodiments, the ITRs have a characteristic T-shaped hairpin structure defined by a self-complementary region (145nt in wild-type AAV) at the 5’ and 3’ ends of the ssDNA which form an energetically stable double stranded region. In some embodiments, the double stranded hairpin structures comprise multiple functions including, but not limited to, acting as an origin for DNA replication by functioning as primers for the endogenous DNA polymerase complex of the host viral replication cell.
[098) In some embodiments, the wild-type AAV viral genome further comprises nucleotide sequences for two open reading frames, one for the four non-structural Rep proteins (R.ep'78, Rep68, Rep52, Rep40, encoded by Rep genes) and one for the three capsid, or structural, proteins (VP1 , VP2, VP3, encoded by capsid genes or Cap genes). The Rep proteins are used for replication and packaging, while the capsid proteins are assembled to create tire protein shell of the AAV, or AAV capsid polypeptide, e.g., an AAV capsid variant. Alternative splicing and alternate initiation codons and. promoters result m the generation of four different Rep proteins from a single open reading frame and the generation of three capsid proteins from a single open reading frame. Though it varies by AAV serotype, as a non-limiting example, for AAV9/hu.l4 (SEQ ID NO: 123 of US 7,906,111, the contents of which are herein incorporated by reference in their entirety) VP1 refers to amino acids 1-736, VP2 refers to amino acids 138-736, and VP3 refers to amino acids 203-736. In some embodiments, for any one of the amino acid sequences of SEQ ID NO: 4, 36, 981, or 982, VP1 comprises amino acids 1-742, VP2 comprises amino acids 138-742, and VP3 comprises amino acids 203-742. In other words, VP I is the full-length capsid sequence, while VP2 and VP3 are shorter components of the whole. As a result, changes in the sequence in the VP3 region, are also changes to VP1 and VP2, however, the percent difference as compared to the parent sequence wall be greatest for VP3 since it is the shortest sequence of the three.
Though described here in relation to the amino acid sequence, the nucleic acid sequence encoding these proteins can be similarly described. Together, the three capsid proteins assemble to create the AAV capsid protein. While not being bound by theory, the AAV capsid protein typically comprises a molar ratio of 1: 1 : 10 of VP1 :VP2;VP3.
[099] AAV particles of the present disclosure may be produced recombinantly and maybe based on AAV reference sequences. In addition to single-stranded AAV viral genomes (e.g., ssAAVs), the present disclosure also provides for self-complementary AAV (scAAVs) viral genomes. scAAV viral genomes contain DNA strands that anneal together to form double-stranded DNA. By skipping second strand synthesis, scAAVs allow' for rapid
expression in the transduced celi. In some embodiments, the AAV particle of the present disclosure is an scAAV. In some embodiments, the AAV particle of the present disclosure is an ssAAV.
[0100] Methods for producing and/or modifying AAV particles are disclosed in the art such as pseudotyped AAV particles (PCT Patent Publication Nos. W0200028004;
W0200123001; W02004112727; W02005005610; and W02005072364, the content of each of which is incorporated herein by reference in its entirety).
[0101] As described herein, the AAV particles of the disclosure comprising an AAV capsid variant, and a viral genome, have enhanced tropism for a cell-type or a tissue, e.g., a CNS cell-type, region, or tissue.
AAV Capsid Variants
[0102] Disclosed herein are AAV particles comprising an AAV capsid variant comprising one or more modifications (e.g., comprising one or more insertions and/or substitutions relative to a wildtype AAV capsid) for enhanced or improved transduction of a target tissue (e.g., cells, regions, and/or tissues of the CNS and/or PNS). In some embodiments, the one or more modifications comprises a peptide insertion and/or amino acid substitution relative to a wildtype AA V capsid. In some embodiments, the one or more modifications is present in a capsid protein of the AAV particle. In some embodiments, the one or more modification is present in a VP1, VP2, and/or VP3 protein of the AAV particle.
[0103] In some embodiments, the one or more modifications (e.g., the peptide insertion relative to a wildtype AAV capsid) is in loop IV of the AAV capsid variant. In some embodiments, the AAV capsid variant is an AAV9 capsid variant.
[0104] In some embodiments, the variant is an insertional variant. As used herein, the term “insertional variant” refers to a polypeptide comprising one or more amino acids inserted, e.g,, “immediately adjacent” or “immediately subsequent.” to a position in a reference amino acid sequence. “Immediately adjacent” or “immediately subsequent” to an amino acid refers to the insertion sequence being connected to either the alpha-carboxy or alpha-amino functional group of the amino acid. In some embodiments, the variant is a deletion variant. As used herein, the tenn “deletion variant” refers to a polypeptide comprising one or more amino acids removed from a reference amino acid sequence. In some embodiments, the variant is a substitution variant. As used herein, the term “substitution variant.” refers to a polypeptide comprising one or more amino acid, changes from a reference
amino acid sequence. In some embodiments, the variant is an msertional variant and a substitution variant.
[0105] In some embodiments, the one or more modifications in the AAV capsid may increase distribution of an AAV particle to a ceil, region, or tissue of the CNS. The cell of the CNS may be, but is not limited to, neurons (e.g., excitatory, inhibitory, motor, sensory. autonomic, sympathetic, parasympathetic, Purkinje, Betz, etc.), glial ceils (e.g., microglia, astrocytes, oligodendrocytes) and/or supporting cells of the brain such as immune cells (e.g..
T cells). The tissue of the CNS may be, but is not limited to, the cortex (e.g., frontal, parietal occipital, and/or temporal), thalamus, hypothalamus, striatum, putamen, caudate nucleus. hippocampus, entorhinal cortex, basal ganglia, or deep cerebellar nuclei.
[0106] In some embodiments, the one or more modifications may increase distribution of an AAV particle to the CNS (e.g., the cortex) after intravenous administration. In some embodiments, the one or more modifications may increase distribution of an AAV particle to the CNS (e.g., the cortex) following focused, ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration .
[0107] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence as set forth in Table 1 , In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence as set forth in Table 2A or Table 2B. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence set forth in Table 17. In some embodiments, the AAV capsid variant comprises
(e.g., in loop IV) an amino acid sequence as set forth in Table 18.
[0108] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence having the formula [N1]-[N2]-[N3], wherein [N2] comprises the amino acid sequence of SPH and [N3] comprises amino acids X4, X5, and X6, wherein at least one of X4, X5, or X6 is a basic amino acid, e.g,, a. K or R. In some embodiments, X4 of [N2] is
K. In some embodiments, X5 of [N2] is K.
[8109] In some embodiments, the AAV capsid variant comprises an amino acid sequence having the formula [N1]-[N2]-[N3], wherein: [N1] comprises .XL X2, and X3, wherein at least one of X1, X2, or X3 is G; [N2] comprises the amino acid sequence of SPH; and [N3] comprises X4, X5, and X6, wherein at least one of X4, X5, or X6 is a basic amino acid, e.g.. a K or R; wherein [N I]-[N2]-[N3] is present in hypervariable loop IV: and wherein the AAV capsid variant comprises an amino acid sequence at least 95% identical to the amino acid sequence corresponding to positions 203-736 of SEQ ID NO: 138.
[0110] In some embodiments, [N1] comprises X1, X2, and X3, wherein at least one of X1,
X2, or X3 is G. In some embodiments, X1 of [N1] is G, V, R, D, E, M, T, I, S, A, N, L, K, H,
P, W, or C. In some embodiments, X2 of [N1] is: S, V, I.,, N, D, H, R. P, G, T, I, A, E, Y, M, or Q. In some embodiments, X3 of [N1] is: G, C, L, D, E, Y, H, V, A, N, P, or S. In some embodiments. [N1] comprises GS, SG. GH, HD, GQ, QD, VS, CS, GR, RG, QS, SH, MS,
RN, TS, IS, GP, ES, SS, GN, AS, NS, LS, GG, KS, GT, PS, RS, GI, WS, DS, ID, GE, DA, DG, ME, EN, KN, KE, Al, NG, PG, TG, SV, IG, LG, AG, EG, SA, YD, HE, HG, RD, ND, PD, MG, QV, DD, HN, HP, GY, GM, GD, or HS. In some embodiments, [N1] comprises GS, SG, GH, or HD. In some embodiments, [N1] is or comprises GSG, GHD, GQD, VSG, CSG, GRG, CSH, GQS, GSH, RVG, GSC, GLL, GDD, GHE, GNY, MSG, RNG, TSG, ISG, GPG. ESG, SSG, GNG, ASG, NSG, LSG, GGG, KSG, HSG, GIG. PSG, GSV, RSG, GIG, WSG, DSG, IDG, GLG, DAG, DGG, MEG, ENG, GSA, KNG, KEG, AIG, GYD, GHG, GRD, GND, GPD, GMG, GQV, GHN, GHP, or GHS. In some embodiments, [N1] is or comprises GSG. In some embodiments, [N1] is or comprises GHD. In some embodiments, [N1]-[N2] comprises SGSPH (SEQ ID NO: 4752), HDSPH (SEQ ID NO: 4703), QDSPH (SEQ ID NO: 4753), RGSPH (SEQ ID NO: 4754), SHSPH (SEQ ID NO: 4755), QSSPH (SEQ ID NO: 4756), DDSPH (SEQ ID NO: 4757), HESPH (SEQ ID NO: 4758), NYSPH (SEQ ID NO: 4759), VGSPH (SEQ ID NO: 4760). SCSPH (SEQ ID NO: 4761). LLSPH (SEQ ID NO: 4762), NGSPH (SEQ ID NO: 4763), PGSPH (SEQ ID NO: 4764), GGSPH (SEQ ID NO: 4765), TGSPH (SEQ ID NO: 4766), SVSPH (SEQ ID NO: 4767), IGSPH (SEQ ID NO: 4768), DGSPH (SEQ ID NO: 4769), LGSPH (SEQ ID NO: 4770), AGSPH (SEQ ID NO: 4771), EGSPH (SEQ ID NO: 4772), SASPH (SEQ ID NO: 4773), YDSPH (SEQ ID NO: 4774), HGSPH (SEQ ID NO: 4775), RDSPH (SEQ ID NO: 4776), NDSPH (SEQ ID NO: 4777), PDSPH (SEQ ID NO: 4778), MGSPH (SEQ ID NO: 4779), QVSPH (SEQ ID NO: 4780), HNSPH (SEQ ID NO: 4781). HPSPH (SEQ ID NO: 4782), or HSSPH (SEQ ID NO: 4783); an amino acid sequence comprising any portion of any of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications; or an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [N1]-[N2] is or comprises GSGSPH (SEQ ID NO: 4695), GHDSPH (SEQ ID NO: 4784), GQDSPH (SEQ ID NO: 4785), VSGSPH (SEQ ID NO: 4786), CSGSPH (SEQ ID NO: 4787), GRGSPH (SEQ ID NO: 4788), CSHSPH (SEQ ID NO: 4789), GQSSPH (SEQ ID NO: 4790), GSHSPH (SEQ ID NO: 4791), GDDSPH (SEQ ID NO: 4792), GHESPH (SEQ ID NO: 4793), GNYSPH (SEQ ID NO: 4794), RVGSPH (SEQ ID NO: 4795), GSCSPH (SEQ ID NO: 4796), GLLSPH (SEQ ID NO: 4797), MSGSPH (SEQ ID NO: 4798), RNGSPH (SEQ ID NO: 4799), TSGSPH (SEQ ID NO: 4800), ISGSPH (SEQ ID NO: 4801), GPGSPH (SEQ ID NO: 4802), ESGSPH (SEQ ID NO: 4803), SSGSPH (SEQ ID NO: 4804), GNGSPH (SEQ ID NO: 4805), ASGSPH
(SEQ ID NO: 4806), NSGSPH (SEQ ID NO: 4807), LSGSPH (SEQ ID NO: 4808), GGGSPH (SEQ ID NO: 4809), KSGSPH (SEQ ID NO: 4810), HSGSPH (SEQ ID NO: 4811), GTGSPH (SEQ ID NO: 4812), PSGSPH (SEQ ID NO: 4813), GSVSPH (SEQ ID NO: 4814), RSGSPII (SEQ ID NO: 4815), GIGSPH (SEQ ID NO: 4816), WSGSPII (SEQ ID NO: 4817), DSGSPH (SEQ ID NO: 4818), IDGSPH (SEQ ID NO: 4819), GLGSPH (SEQ ID NO: 4820), DAGSPH (SEQ ID NO: 4821). DGGSPH (SEQ ID NO: 4822), MEGSPH (SEQ ID NO: 4823), ENGSPH (SEQ ID NO: 4824), GSASPH (SEQ ID NO: 4825), KNGSPH (SEQ ID NO: 4826), KEGSPH (SEQ ID NO: 4827), AIGSPH (SEQ ID NO: 4828), GYDSPH (SEQ ID NO: 4829), GHGSPH (SEQ ID NO: 4830), GRDSPH (SEQ ID NO: 4831), GNDSPH (SEQ ID NO: 4832), GPDSPH (SEQ ID NO: 4833), GMGSPH (SEQ ID NO: 4834), GQVSPII (SEQ ID NO: 4835), GHNSPH (SEQ ID NO: 4836), GIIPSPH (SEQ ID NO: 4837), or GHSSPH (SEQ ID NO: 4838); an amino acid sequence comprising any portion of any of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [N1]-[N2] is or comprises GSGSPH (SEQ ID NO: 4695). In some embodiments, [N1]-[N2] is or comprises GHDSPH (SEQ ID NO: 4784). [0111] In some embodiments, X4, X5, or both of [N3] are K. In some embodiments, X4, X5, or X6 of [N3] is R. In some embodiments, X4 of [N3] is: A, K, V, S, T, G, F, W, V, N, or R. In some embodiments, X5 of [N3] is: S, K, T, F, I, L, Y, H, M, or R. In some embodiments, X6 of [N3] is: G, R, A, M, I, N, T, Y, D, P, V, L, E, W, N, Q, K, or S. In some embodiments, [N3] comprises SK, KA, KS, AR, RM, VK, AS, SR, VK, KR, KK, KN, VR, RS, RK, KT, TS, KF, FG, KI, IG. KL, LG. TT, TY, KY, YG, KD, KP, TR. RG, VR, GA, SL, SS, FL, WK, SA, RA, LR, KW, RR, GK, TK, NK, AK, KV, KG, KH, KM, TG, SE, SV, SW, SN, IIG, SQ, LW, MG, MA, or SG. In some embodiments, [N3] comprises SK, KA, KS, or SG. In some embodiments, [N3] is or comprises SKA, KSG, ARM, VKS, ASR, VKI, KKN, VRM, RKA, KTS, KFG, KIG, KLG, KTT, KTY, KYG, SKD, SKP, TRG, VRG, KRG, GAR, KSA, KSR, SKL, SRA, SKR, SLR, SRG, SSR, FLR, SKW, SKS, WKA, VRR, SKV, SKT, SKG, GIGA, TKA, NKA, SKL, SKN, AKA, KTG, KSL, KSE, KSV, KSW, KSN, KHG, KSQ, KSK, KLW. WKG, KMG, KMA, or RSG. In some embodiments, [N3] is or comprises SKA . In some embodiments, [N3] is or comprises KSG. In some embodiments, [N2]-[N3] comprises SPHSK (SEQ ID NO: 4701), SPIIKS (SEQ ID NO: 4704), SPHAR (SEQ ID NO: 4705), SPHVK (SEQ ID NO: 4706), SPHAS (SEQ ID NO: 4707), SPHKK
(SEQ ID NO: 4708), SPHVR (SEQ ID NO: 4709), SPHRK. (SEQ ID NO: 4710), SPHKT (SEQ ID NO: 4711), SPHKF (SEQ ID NO: 4712), SPHKI (SEQ ID NO: 4713), SPHKL (SEQ ID NO: 4714), SPHKY (SEQ ID NO: 4715), SPHTR (SEQ ID NO: 4716), SPHKR (SEQ ID NO: 4717), SPHGA (SEQ ID NO: 4718), SPHSR (SEQ ID NO: 4719), SPHSL (SEQ ID NO: 4720), SPHSS (SEQ ID NO: 4721), SPHFL (SEQ ID NO: 4722), SPHWK (SEQ ID NO: 4723), SPHGK (SEQ ID NO: 4724). SPHTK (SEQ ID NO: 4725), SPHNK (SEQ ID NO: 4726), SPHAK (SEQ ID NO: 4727), SPHKH (SEQ ID NO: 4728), SPHKM (SEQ ID NO: 4729), or SPHRS (SEQ ID NO: 4730). In some embodiments [N2]-[N3] comprises SPHSK (SEQ ID NO: 4701) or SPHKS (SEQ ID NO: 4704). In some embodiments, [N2]-[N3] is or comprises SPHSKA (SEQ ID NO: 941), SPHKSG (SEQ ID NO: 946), SPHARM (SEQ ID NO: 947), SPHVKS (SEQ ID NO: 948), SPHASR (SEQ ID NO: 949), SPHVKI (SEQ ID NO: 950), SPHKKN (SEQ ID NO: 954), SPHVRM (SEQ ID NO: 955), SPHRKA (SEQ ID NO: 956), SPHKFG (SEQ ID NO: 957), SPHKIG (SEQ ID NO: 958), SPHKLG (SEQ ID NO: 959), SPHKTS (SEQ ID NO: 963), SPHKTT (SEQ ID NO: 964), SPHKTY (SEQ ID NO: 965), SPHKYG (SEQ ID NO: 966), SPHSKD (SEQ ID NO: 967), SPHSKP (SEQ ID NO: 968), SPHTRG (SEQ ID NO: 972), SPHVRG (SEQ ID NO: 973), SPHKRG (SEQ ID NO: 974). SPHGAR (SEQ ID NO: 975), SPHKSA (SEQ ID NO: 977), SPHKSR (SEQ ID NO: 951), SPHSKL (SEQ ID NO: 960), SPHSRA (SEQ ID NO: 969), SPHSKR (SEQ ID NO: 978), SPHSLR (SEQ ID NO: 952), SPHSRG (SEQ ID NO: 961), SPHSSR (SEQ ID NO: 970), SPHFLR (SEQ ID NO: 979), SPHSKW (SEQ ID NO: 953), SPHSKS (SEQ ID NO: 962), SPHWKA (SEQ ID NO: 971), SPHVRR (SEQ ID NO: 980), SPHSKT (SEQ ID NO: 4731), SPHSKG (SEQ ID NO: 4732), SPHGKA (SEQ ID NO: 4733), SPHNKA (SEQ ID NO: 4734), SPHSKN (SEQ ID NO: 4735), SPHAKA (SEQ ID NO: 4736), SPHSKV (SEQ ID NO: 4737), SPHKTG (SEQ ID NO: 4738), SPHTKA (SEQ ID NO: 4739), SPHKSL (SEQ ID NO: 4740), SPHKSE (SEQ ID NO: 4741), SPHKSV (SEQ ID NO: 4742), SPHKSW (SEQ ID NO: 4743), SPHKSN (SEQ ID NO: 4744), SPHKHG (SEQ ID NO: 4745), SPHKSQ (SEQ ID NO: 4746), SPHKSK (SEQ ID NO: 4747), SPHKLW (SEQ ID NO: 4748), SPHWKG (SEQ ID NO: 4749), SPHKMG (SEQ ID NO: 4750), SPHKMA (SEQ ID NO: 4751), or SPHRSG (SEQ ID NO: 976). In some embodiments, [N2]-[N3] is or comprises SPHSKA (SEQ ID NO: 941). In some embodiments, [N2]-[N3] is or comprises SPHKSG (SEQ ID NO: 946).
[0112] In some embodiments, [N1]-[N2]-[N3] comprises SGSPHSK (SEQ ID NO: 4839), HDSPHKS (SEQ ID NO: 4840), SGSPHAR (SEQ ID NO: 4841), SGSPHVK (SEQ ID NO: 4842), QDSPHKS (SEQ ID NO: 4843), SGSPHKK (SEQ ID NO: 4844), SGSPHVR (SEQ
ID NO: 4845), SGSPHAS (SEQ ID NO: 4846), SGSPIIRK (SEQ ID NO: 4847), SGSPHKT (SEQ ID NO: 4848), SHSPHKS (SEQ ID NO: 4849), QSSPHRS (SEQ ID NO: 4850), RGSPHAS (SEQ ID NO: 4851), RGSPHSK (SEQ ID NO: 4852), SGSPHKF (SEQ ID NO: 4853), SGSPHKI (SEQ ID NO: 4854), SGSPHKL (SEQ ID NO: 4855), SGSPHKY (SEQ ID NO: 4856), SGSPHTR (SEQ ID NO: 4857), SHSPHKR (SEQ ID NO: 4858), SGSPHGA (SEQ ID NO: 4859), HDSPHKR (SEQ ID NO: 4860), DDSPHKS (SEQ ID NO: 4861), HESPHKS (SEQ ID NO: 4862), NYSPHKI (SEQ ID NO: 4863), SGSPHSR (SEQ ID NO: 4864), SGSPHSL (SEQ ID NO: 4865), SGSPHSS (SEQ ID NO: 4866), VGSPHSK (SEQ ID NO: 4867), SCSPHRK (SEQ ID NO: 4868), SGSPHFL (SEQ ID NO: 4869), LLSPHWK (SEQ ID NO: 4870), NGSPHSK (SEQ ID NO: 4871), PGSPHSK (SEQ ID NO: 4872), GGSPHSK (SEQ ID NO: 4873), TGSPHSK (SEQ ID NO: 4874), SVSPHGK (SEQ ID NO: 4875), SGSPHTK (SEQ ID NO: 4876), 1GSPHSK (SEQ ID NO: 4877), DGSPHSK (SEQ ID NO: 4878), SGSPHNK (SEQ ID NO: 4879), LGSPHSK (SEQ ID NO: 4880), AGSPHSK (SEQ ID NO: 4881), EGSPHSK (SEQ ID NO: 4882), SASPHSK (SEQ ID NO: 4883), SGSPHAK (SEQ ID NO: 4884), IIDSPIIKI (SEQ ID NO: 4885), YDSPHKS (SEQ ID NO: 4886), HDSPHKT (SEQ ID NO: 4887), RGSPHKR (SEQ ID NO: 4888), HGSPHSK (SEQ ID NO: 4889), RDSPHKS (SEQ ID NO: 4890), NDSPHKS (SEQ ID NO: 4891), QDSPHKI (SEQ ID NO: 4892), PDSPHKI (SEQ ID NO: 4893), PDSPHKS (SEQ ID NO: 4894), MGSPHSK (SEQ ID NO: 4895), HDSPHKH (SEQ ID NO: 4896), QVSPHKS (SEQ ID NO: 4897), HNSPHKS (SEQ ID NO: 4898), NGSPHKR (SEQ ID NO: 4899), HDSPHKY (SEQ ID NO: 4900), NDSPHKI (SEQ ID NO: 4901), HDSPHKL (SEQ ID NO: 4902), HPSPHWK (SEQ ID NO: 4903), IIDSPHKM (SEQ ID NO: 4904), or HSSPHRS (SEQ ID NO: 4905).
In some embodiments, [N1]-[N2]-[N3] is GSGSPHSKA (SEQ ID NO: 4697), GHDSPHKSG (SEQ ID NO: 4698), GSGSPHARM (SEQ ID NO: 4906), GSGSPHVKS (SEQ ID NO: 4907), GQDSPHKSG (SEQ ID NO: 4908), GSGSPHASR (SEQ ID NO: 4909), GSGSPHVKI (SEQ ID NO: 4910), GSGSPHKKN (SEQ ID NO: 4911), GSGSPHVRM (SEQ ID NO: 4912), VSGSPHSKA (SEQ ID NO: 4913), CSGSPHSKA (SEQ ID NO: 4914), GSGSPHRKA (SEQ ID NO: 4915), CSGSPHKTS (SEQ ID NO: 4916), CSHSPHKSG (SEQ ID NO: 4917), GQSSPHRSG (SEQ ID NO: 4918), GRGSPHASR (SEQ ID NO: 4919), GRGSPHSKA (SEQ ID NO: 4920), GSGSPHKFG (SEQ ID NO: 4921), GSGSPHKIG (SEQ ID NO: 4922), GSGSPHKLG (SEQ ID NO: 4923), GSGSPHKTS (SEQ ID NO: 4924), GSGSPHKTT (SEQ ID NO: 4925), GSGSPHKTY (SEQ ID NO: 4926), GSGSPHKYG (SEQ ID NO: 4927), GSGSPHSKD (SEQ ID NO: 4928), GSGSPHSKP (SEQ ID NO: 4929), GSGSPHTRG (SEQ ID NO: 4930), GSGSPHVRG (SEQ ID NO: 4931), GSHSPHKRG
(SEQ ID NO: 4932), GSHSPHKSG (SEQ ID NO: 4933), VSGSPHASR (SEQ ID NO: 4934), VSGSPHGAR (SEQ ID NO: 4935), VSGSPHKFG (SEQ ID NO: 4936), GHDSPHKRG (SEQ ID NO: 4937), GDDSPHKSG (SEQ ID NO: 4938), GHESPHKSA (SEQ ID NO: 4939), GHDSPHKSA (SEQ ID NO: 4940), GNYSPHKIG (SEQ ID NO: 4941), GHDSPHKSR (SEQ ID NO: 4942), GSGSPHSKL (SEQ ID NO: 4943), GSGSPHSRA (SEQ ID NO: 4944), GSGSPHSKR (SEQ ID NO: 4945), GSGSPHSLR (SEQ ID NO: 4946), GSGSPHSRG (SEQ ID NO: 4947), GSGSPHSSR (SEQ ID NO: 4948), RVGSPHSKA (SEQ ID NO: 4949), GSCSPHRKA (SEQ ID NO: 4950), GSGSPHFLR (SEQ ID NO: 4951), GSGSPHSKW (SEQ ID NO: 4952), GSGSPHSKS (SEQ ID NO: 4953), GLLSPHWKA (SEQ ID NO: 4954), GSGSPHVRR (SEQ ID NO: 4955), GSGSPHSKV (SEQ ID NO: 4956), MSGSPHSKA (SEQ ID NO: 4957), RNGSPHSKA (SEQ ID NO: 4958), TSGSPHSKA (SEQ ID NO: 4959), ISGSPHSKA (SEQ ID NO: 4960), GPGSPHSKA (SEQ ID NO: 4961), GSGSPHSKT (SEQ ID NO: 4962), ESGSPHSKA (SEQ ID NO: 4963), SSGSPHSKA (SEQ ID NO: 4964), GNGSPHSKA (SEQ ID NO: 4965), ASGSPHSKA (SEQ ID NO: 4966), NSGSPHSKA (SEQ ID NO: 4967), ESGSPHSKA (SEQ ID NO: 4968), GGGSPHSKA (SEQ ID NO: 4969), KSGSPHSKA (SEQ ID NO: 4970), GGGSPHSKS (SEQ ID NO: 4971), GSGSPHSKG (SEQ ID NO: 4972), ESGSPHSKA (SEQ ID NO: 4973), GTGSPHSKA (SEQ ID NO: 4974), PSGSPHSKA (SEQ ID NO: 4975), GSVSPHGKA (SEQ ID NO: 4976), RSGSPHSKA (SEQ ID NO: 4977), GSGSPHTKA (SEQ ID NO: 4978), GIGSPHSKA (SEQ ID NO: 4979), WSGSPHSKA (SEQ ID NO: 4980), DSGSPHSKA (SEQ ID NO: 4981), IDGSPHSKA (SEQ ID NO: 4982), GSGSPHNKA (SEQ ID NO: 4983), GLGSPHSKS (SEQ ID NO: 4984), DAGSPHSKA (SEQ ID NO: 4985), DGGSPHSKA (SEQ ID NO: 4986), MEGSPHSKA (SEQ ID NO: 4987), ENGSPHSKA (SEQ ID NO: 4988), GSASPHSKA (SEQ ID NO: 4989), GNGSPHSKS (SEQ ID NO: 4990), KNGSPHSKA (SEQ ID NO: 4991), KEGSPHSKA (SEQ ID NO: 4992), AIGSPHSKA (SEQ ID NO: 4993), GSGSPHSKN (SEQ ID NO: 4994), GSGSPHAKA (SEQ ID NO: 4995), GHDSPHKIG (SEQ ID NO: 4996), GYDSPHKSG (SEQ ID NO: 4997), GHESPHKSG (SEQ ID NO: 4998), GHDSPHKTG (SEQ ID NO: 4999), GRGSPHKRG (SEQ ID NO: 5000), GQDSPHKSG (SEQ ID NO: 4908), GHDSPHKSL (SEQ ID NO: 5001), GHGSPHSKA (SEQ ID NO: 5002), GHDSPHKSE (SEQ ID NO: 5003), VSGSPHSKA (SEQ ID NO: 4913), GRDSPHKSG (SEQ ID NO: 5004), GNDSPHKSV (SEQ ID NO: 5005), GQDSPHKIG (SEQ ID NO: 5006), GHDSPHKSV (SEQ ID NO: 5007), GPDSPHKIG (SEQ ID NO: 5008), GPDSPHKSG (SEQ ID NO: 5009), GHDSPHKSW (SEQ ID NO: 5010), GHDSPHKSN (SEQ ID NO: 5011),
GMGSPHSKT (SEQ ID NO: 5012), GHDSPHKHG (SEQ ID NO: 5013), GQVSPHKSG (SEQ ID NO: 5014), GDDSPHKSV (SEQ ID NO: 5015), GHINSPHKSG (SEQ ID NO: 5016), GNGSPHKRG (SEQ ID NO: 5017), GHDSPHKYG (SEQ ID NO: 5018), GHDSPHKSQ (SEQ ID NO: 5019), GNDSPHKIG (SEQ ID NO: 5020), GHDSPHKSK (SEQ ID NO: 5021), GHDSPHKLW (SEQ ID NO: 5022), GHPSPHWKG (SEQ ID NO: 5023). GHDSPHKMG (SEQ ID NO: 5024), GHDSPHKMA (SEQ ID NO: 5025), or GHSSPHRSG (SEQ ID NO: 5026); an amino acid sequence comprising any portion of any of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, or 8 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [N1]-[N2]-[N3] is or comprises GSGSPHSKA (SEQ ID NO: 4697). In some embodiments. [N1]-[N2]-[N3] is or comprises GHDSPHKSG (SEQ ID NO: 4698).
[0113] In some embodiments, the AAV capsid variant comprising an amino acid sequence having the formula [N1]-[N2]-[N3] (e.g., in loop IV) further comprises [N4], which comprises X7 X8 X9 X10. In some embodiments, X7 of [N4] is W, Q, K, R. G, L, V, S, P.
H, K, I, M, A, E, or F. In some embodiments, X8 of [N4] is N, Y, C, K, T, H, R, D, V, S, P, G, W, E, F, A, I, M, Q, or L. In some embodiments, X9 of [N4] is Q, G, K, FI, R, T, L, D, A, P, I, F, V, M, W, Y, S, E, N, or Y. In some embodiments, X10 of [N4] is Q. H, L, R. W, K, A, P, E, M, I, S, G, N, Y, C, V, T, D, or V. In some embodiments [N4] is or comprises QNQQ (SEQ ID NO: 5028), WNQQ (SEQ ID NO: 5029), QYYV (SEQ ID NO: 5030), RRQQ (SEQ ID NO: 5031), QNQQ (SEQ ID NO: 5028), GCGQ (SEQ ID NO: 5032), LRQQ (SEQ ID NO: 5033), RNQQ (SEQ ID NO: 5034). VNQQ (SEQ ID NO: 5035). FREQ (SEQ ID NO: 5036), FNQQ (SEQ ID NO: 5037), LLQQ (SEQ ID NO: 5038), SNQQ (SEQ ID NO: 5039), RLQQ (SEQ ID NO: 5040), LNQQ (SEQ ID NO: 5041), QRKL (SEQ ID NO: 5042), LRRQ (SEQ ID NO: 5043). QRLR (SEQ ID NO: 5044), QRRL (SEQ ID NO: 5045), RRLQ (SEQ ID NO: 5046), RL.RQ (SEQ ID NO: 5047), SKRQ (SEQ ID NO: 5048), QLYR (SEQ ID NO: 5049), QLTV (SEQ ID NO: 5050), QNKQ (SEQ ID NO: 5051), KNQQ (SEQ ID NO: 5052), QKQQ (SEQ ID NO: 5053), QTQQ (SEQ ID NO: 5054), QNHQ (SEQ ID NO: 5055), QHQQ (SEQ ID NO: 5056), QNQH (SEQ ID NO: 5057), QHRQ (SEQ ID NO: 5058), L.TQQ (SEQ ID NO: 5059), QNQW (SEQ ID NO: 5060), QNTH (SEQ ID NO: 5061), RRRQ (SEQ ID NO: 5062), QYQQ (SEQ ID NO: 5063), QNDQ (SEQ ID NO: 5064), QNRH (SEQ ID NO: 5065), RDQQ (SEQ ID NO: 5066),
PNLQ (SEQ ID NO: 5067), HVRQ (SEQ ID NO: 5068), PNQII (SEQ ID NO: 5069), FINQQ (SEQ ID NO: 5070), QSQQ (SEQ ID NO: 5071), QPAK (SEQ ID NO: 5072), QNLA (SEQ ID NO: 5073), QNQL (SEQ ID NO: 5074), QGQQ (SEQ ID NO: 5075), ENRQ (SEQ ID NO: 5076), QNPP (SEQ ID NO: 5077), QNLQ (SEQ ID NO: 5078), QDQE (SEQ ID NO: 5079), QDQQ (SEQ ID NO: 5080), HWQQ (SEQ ID NO: 5081), PNQQ (SEQ ID NO: 5082). PEQQ (SEQ ID NO: 5083), QRTM (SEQ ID NO: 5084), LHQH (SEQ ID NO: 5085), QHRI (SEQ ID NO: 5086), QYIH (SEQ ID NO: 5087), QKFE (SEQ ID NO: 5088), QFPS (SEQ ID NO: 5089), QNPL (SEQ ID NO: 5090), QAIK (SEQ ID NO: 5091), QNRQ (SEQ ID NO: 5092), QYQH (SEQ ID NO: 5093), QNPQ (SEQ ID NO: 5094), QHQL (SEQ ID NO: 5095), QSPP (SEQ ID NO: 5096), QAKL (SEQ ID NO: 5097), KSQQ (SEQ ID NO: 5098), QDRP (SEQ ID NO: 5099), QNLG (SEQ ID NO: 5100), QAFFI (SEQ ID NO: 5101), QNAQ (SEQ ID NO: 5102), HNQL (SEQ ID NO: 5103), QKLN (SEQ ID NO: 5104), QNVQ (SEQ ID NO: 5105), QAQQ (SEQ ID NO: 5106), QTPP (SEQ ID NO: 5107), QPPA (SEQ ID NO: 5108), QERP (SEQ ID NO: 5109), QDLQ (SEQ ID NO: 5110), QAMH (SEQ ID NO: 5111), QIIPS (SEQ ID NO: 5112), PGLQ (SEQ ID NO: 5113), QGIR (SEQ ID NO: 5114), QAPA (SEQ ID NO: 5115), QIPP (SEQ ID NO: 5116), QTQL (SEQ ID NO: 5117), QAPS (SEQ ID NO: 5118), QNTY (SEQ ID NO: 5119), QDKQ (SEQ ID NO: 5120), QNHL (SEQ ID NO: 5121), QIGM (SEQ ID NO: 5122), LNKQ (SEQ ID NO: 5123), PNQL (SEQ ID NO: 5124), QLQQ (SEQ ID NO: 5125), QRMS (SEQ ID NO: 5126), QGIL (SEQ ID NO: 5127). QDRQ (SEQ ID NO: 5128), RDWQ (SEQ ID NO: 5129), QERS (SEQ ID NO: 5130), QNYQ (SEQ ID NO: 5131), QRTC (SEQ ID NO: 5132), QIGH (SEQ ID NO: 5133), QGAI (SEQ ID NO: 5134), QVPP (SEQ ID NO: 5135), QVQQ (SEQ ID NO: 5136), LMRQ (SEQ ID NO: 5137), QYSV (SEQ ID NO: 5138), QAIT (SEQ ID NO: 5139), QKTL (SEQ ID NO: 5140), QLHH (SEQ ID NO: 5141), QNII (SEQ ID NO: 5142), QGHH (SEQ ID NO: 5143), QSKV (SEQ ID NO: 5144), QLPS (SEQ ID NO: 5145), IGKQ (SEQ ID NO: 5146), QAIH (SEQ ID NO: 5147), QHGL (SEQ ID NO: 5148), QFMC (SEQ ID NO: 5149), QNQM (SEQ ID NO: 5150), QHLQ (SEQ ID NO: 5151), QPAR (SEQ ID NO: 5152), QSLQ (SEQ ID NO: 5153), QSQL (SEQ ID NO: 5154), HSQQ (SEQ ID NO: 5155), QMPS (SEQ ID NO: 5156), QGSL (SEQ ID NO: 5157), QVPA (SEQ ID NO: 5158), HYQQ (SEQ ID NO: 5159), QVPS (SEQ ID NO: 5160), RGEQ (SEQ ID NO: 5161), PGQQ (SEQ ID NO: 5162), LEQQ (SEQ ID NO: 5163), QNQS (SEQ ID NO: 5164), QKVI (SEQ ID NO: 5165). QNND (SEQ ID NO: 5166), QSVH (SEQ ID NO: 5167), QPLG (SEQ ID NO: 5168), HNQE (SEQ ID NO: 5169), QIQQ (SEQ ID NO: 5170), QVRN (SEQ ID NO: 5171), PSNQ (SEQ ID NO: 5172), QVGH (SEQ ID NO: 5173), QRDI (SEQ ID NO: 5174), QMPN (SEQ ID NO: 5175), RGLQ
(SEQ ID NO: 5176), PSLQ (SEQ ID NO: 5177), QRDQ (SEQ ID NO: 5178), QAKG (SEQ ID NO: 5179), QSAH (SEQ ID NO: 5180), QSTM (SEQ ID NO: 5181), QREM (SEQ ID NO: 5182), QYRA (SEQ ID NO: 5183), QRQQ (SEQ ID NO: 5184), QWQQ (SEQ ID NO: 5185), QRMN (SEQ ID NO: 5186), GDSQ (SEQ ID NO: 5187), QKIS (SEQ ID NO: 5188), PSMQ (SEQ ID NO: 5189), SPRQ (SEQ ID NO: 5190), MEQQ (SEQ ID NO: 5191), QYQN (SEQ ID NO: 5192), QIRQ (SEQ ID NO: 5193), QSVQ (SEQ ID NO: 5194), RSQQ (SEQ ID NO: 5195), QNKL (SEQ ID NO: 5196), QIQH (SEQ ID NO: 5197), PRQQ (SEQ ID NO: 5198), IITQQ (SEQ ID NO: 5199), QRQII (SEQ ID NO: 5200), RNQE (SEQ ID NO: 5201), QSKQ (SEQ ID NO: 5202), QNQP (SEQ ID NO: 5203), QSPQ (SEQ ID NO: 5204), QTRQ (SEQ ID NO: 5205), QNLH (SEQ ID NO: 5206), QNQE (SEQ ID NO: 5207), LNQP (SEQ ID NO: 5208), QNQD (SEQ ID NO: 5209), QNLL (SEQ ID NO: 5210), QLVI (SEQ ID NO: 5211), RTQE (SEQ ID NO: 5212), QTHQ (SEQ ID NO: 5213), QDQH (SEQ ID NO: 5214), QSQH (SEQ ID NO: 5215), VRQQ (SEQ ID NO: 5216), AWQQ (SEQ ID NO: 5217), QSVP (SEQ ID NO: 5218), QNIQ (SEQ ID NO: 5219), LDQQ (SEQ ID NO: 5220), PDQQ (SEQ ID NO: 5221), ESQQ (SEQ ID NO: 5222), QRQL (SEQ ID NO: 5223), QIIV (SEQ ID NO: 5224), QKQS (SEQ ID NO: 5225), QSHQ (SEQ ID NO: 5226), QFVV (SEQ ID NO: 5227), QSQP (SEQ ID NO: 5228), QNEQ (SEQ ID NO: 5229). INQQ (SEQ ID NO: 5230), RNRQ (SEQ ID NO: 5231), RDQK (SEQ ID NO: 5232), QWKR (SEQ ID NO: 5233), ENRQ (SEQ ID NO: 5234), QTQP (SEQ ID NO: 5235), QKQL (SEQ ID NO: 5236), RNQL (SEQ ID NO: 5237), ISIQ (SEQ ID NO: 5238), QTVC (SEQ ID NO: 5239), QQIM (SEQ ID NO: 5240), LNHQ (SEQ ID NO: 5241), QNQA (SEQ ID NO: 5242), QMIH (SEQ ID NO: 5243), RNIIQ (SEQ ID NO: 5244), or QKMN (SEQ ID NO: 5245), or any dipeptide or tripeptide thereof. In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises: the amino acid sequence of any of SEQ ID NOs: 1800-2241; an amino acid sequence comprising any portion of any of the aforesaid amino acid, sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the aforesaid amino acid sequences; or an amino acid, sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises GSGSPHSKAQNQQ (SEQ ID NO: 1801). In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises GHDSPHKSGQNQQ (SEQ ID NO: 1800).
[0114] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence having the formula [N1]-[N2]-[N3]; and further comprises [N0], which
comprises XA XB and XC. In some embodiments, XA of [N0] is T, S, Y, M, A, C, I, R, L, D, F, V, Q, N, H, E, or G. In some embodiments, XB of [N0] is I, M, P, E, N, D, S, A, T, G, Q, F, V, L, C, H, R, W, or L. In some embodiments, Xc of [N0] is N, M, E, G, Y, W, T, I, Q, F, V, A, L, I, P, K, R, H, S, D, or S. In some embodiments, [N0] is or comprises TIN, SMN, TIM, YES, GLS, MPE, MEG, MEY, AEW, CEW, ANN, IPE, ADM, IEY, ADY, IET, MEW, CEY, RIN, MEL LEY, ADW, IEI, DIM, FEQ, MEF, CDQ, LPE, IEN, MES, AEL VEY, TIN, TSN, IEV, MEM, AEV, MDA, VEW, AEQ, LEW, MEL,, MET, MEA, IES, MEV, CEI, ATN, MDG, QEV, ADQ, NMN, IEM, ISN, TGN, QQQ, HDW, IEG, TII, TFP, TEK, EIN, TVN, TFN, SIN, TER, TSY, ELH, AIN, SVN, TDN, TEH, TVH, TEN, TSS, TTD, TCN, NIN, TEH, AEM, AIK, TDK, TFK, SDQ, TEI, NTN, TET, SIK, TEL,, TEA, TAN, TIY, TFS, TES, TTN, TED, TNN, EVH, TIS, TVR, TDR, TIK, NHI, TIP, ESD, TDL, TVP, TVI, AEH, NCL, TVK, NAD, TIT, NCV, HR, NAL, VIN, TIQ, TEF, TRE, QGE, SEK, NVN, GGE, EFV, SDK, TEQ, EVQ, TEY, NCW, TDV, SDI, NSI, NSL, EVV, TEP, SEE, TWQ, TEV, AVN, GVL, TEN, TEG, TRD, NAI, AEN, AET, ETA, NNE, or any dipeptide thereof. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is or comprises the amino acid sequence of any one of SEQ ID NOs: 2242-2886; an amino acid sequence comprising any portion of any of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g,, consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TINGSGSPHSKAQNQQ (SEQ ID NO: 2242). In some embodiments, [N0]-[N1]-[N2]- [N3]-[N4] is or comprises TINGHDSPHKSGQNQQ (SEQ ID NO: 2243).
[0115] In some embodiments, [N3] is present immediately subsequent to [N2], In some embodiments, the amino acid sequence comprises, from N-terminus to C-terminus, [N2]-[N3]. In some embodiments, the amino acid sequence comprises, from N-terminus to C- terminus, [N1]-[N2]-[N3], In some embodiments, the amino acid, sequence comprises, from N-terminus to C-tenninus, [N1]-[N2]-[N3]-[N4], In some embodiments, the amino acid sequence comprises, from N-terminus to C-tenninus, [N0]-[N1]-[N2]-[N3], In some embodiments, the amino acid sequence comprises, from N-terminus to C-terminus, [N0]- [N1]-[N2]-[N3]-[N4],
[0116] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence having the formula [A][B] (SEQ ID NO: 4694), wherein [A] comprises
the amino acid sequence of GSGSPH (SEQ ID NO: 4695) and [B] comprises X1 X2 X3 X4 X5 X6 X7. In some embodiments, X1 of [BJ is S, C, F, or V. In some embodiments, X2 of [B] is K, L, R, I, E, Y, V, or S. In some embodiments, X3 of [B] is A, R, L, G, I, Y, S, F, or W. In some embodiments X4 of [B] is W, Q, R, G, L, V, S, or F. In some embodiments, X5 of [B] is N, Y, R, C, K, or L. In some embodiments, X6 of [B] is Q, G, K, R, T, L, or Y. In some embodiment, X7 of [B] is Q, L, R, or V. In some embodiments, [B] comprises SLLWNQQ (SEQ ID NO: 5247), SKAQYYV (SEQ ID NO: 5248), SKLRRQQ (SEQ ID NO: 5249), SIWQNQQ (SEQ ID NO: 5250), SKAGCGQ (SEQ ID NO: 5251), SRAQNQQ (SEQ ID NO: 5252), SKRLRQQ (SEQ ID NO: 5253), SLRRNQQ (SEQ ID NO: 5254), SRGRNQQ (SEQ ID NO: 5255), SEIVNQQ (SEQ ID NO: 5256), SSRRNQQ (SEQ ID NO: 5257), CLLQNQQ (SEQ ID NO: 5258), SKAFRLQ (SEQ ID NO: 5259), CLAQNQQ (SEQ ID NO: 5260), FLRQNQQ (SEQ ID NO: 5261), SLRFNQQ (SEQ ID NO: 5262), SYLRNQQ (SEQ ID NO: 5263), CSLQNQQ (SEQ ID NO: 5264), VLWQNQQ (SEQ ID NO: 5265), SKWLLQQ (SEQ ID NO: 5266), SLWSNQQ (SEQ ID NO: 5267), SKRRLQQ (SEQ ID NO: 5268), SVYLNQQ (SEQ ID NO: 5269), SLWLNQQ (SEQ ID NO: 5270), SKAQRKL (SEQ ID NO: 5271), SKALRRQ (SEQ ID NO: 5272), SKAQRLR (SEQ ID NO: 5273), SKAQNQQ (SEQ ID NO: 5274), SKAQRRL (SEQ ID NO: 5275), SKARRQQ (SEQ ID NO: 5276), SKARRLQ (SEQ ID NO: 5277), SKSRRQQ (SEQ ID NO: 5278), SKARLRQ (SEQ ID NO: 5279), SKASKRQ (SEQ ID NO: 5280), VRRQNQQ (SEQ ID NO: 5281), SKAQLYR (SEQ ID NO: 5282), SLFRNQQ (SEQ ID NO: 5283), SKAQLTV (SEQ ID NO: 5284), or any dipeptide, tripeptide, tetrapeptide, pentapeptide, or hexapeptide thereof. In some embodiments, [A] [B] comprises GSGSPHSLLWNQQ (SEQ ID NO: 5285), GSGSPHSKAQYYV (SEQ ID NO: 2060), GSGSPHSKLRRQQ (SEQ ID NO: 2061), GSGSPHSIWQNQQ (SEQ ID NO: 5286), GSGSPHSKAGCGQ (SEQ ID NO: 2062), GSGSPHSRAQNQQ (SEQ ID NO: 2063), GSGSPHSKRLRQQ (SEQ ID NO: 2064), GSGSPHSLRRNQQ (SEQ ID NO: 2065), GSGSPHSRGRNQQ (SEQ ID NO: 2066), GSGSPHSEIVNQQ (SEQ ID NO: 5287), GSGSPHSSRRNQQ (SEQ ID NO: 2067), GSGSPHCLLQNQQ (SEQ ID NO: 5288), GSGSPHSKAFRLQ (SEQ ID NO: 2068), GSGSPHCLAQNQQ (SEQ ID NO: 5289), GSGSPHFLRQNQQ (SEQ ID NO: 2070), GSGSPHSLRFNQQ (SEQ ID NO: 2071), GSGSPHSYLRNQQ (SEQ ID NO: 5290), GSGSPHCSLQNQQ (SEQ ID NO: 5291), GSGSPHVLWQNQQ (SEQ ID NO: 5292), GSGSPHSKWLLQQ (SEQ ID NO: 2072), GSGSPHSLWSNQQ (SEQ ID NO: 5293), GSGSPHSKRRLQQ (SEQ ID NO: 2073), GSGSPHSVYLNQQ (SEQ ID NO: 5294), GSGSPHSLWLNQQ (SEQ ID NO: 5295), GSGSPHSKAQRKL (SEQ ID NO: 2074),
GSGSPHSKALRRQ (SEQ ID NO: 2075), GSGSPHSKAQRLR (SEQ ID NO: 2076), GSGSPHSKAQNQQ (SEQ ID NO: 1801), GSGSPHSKAQRRL (SEQ ID NO: 2077), GSGSPHSKARRQQ (SEQ ID NO: 2078), GSGSPHSKARRLQ (SEQ ID NO: 2079), GSGSPHSKSRRQQ (SEQ ID NO: 2080), GSGSPHSKARLRQ (SEQ ID NO: 2082), GSGSPHSKASKRQ (SEQ ID NO: 2083), GSGSPHVRRQNQQ (SEQ ID NO: 2084), GSGSPHSKAQLYR (SEQ ID NO: 2085), GSGSPHSLFRNQQ (SEQ ID NO: 5296), GSGSPHSKAQLTV (SEQ ID NO: 2086), or any portion thereof, e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , or 12 amino acids, e.g., consecutive amino acids, thereof. In some embodiments, [B] is present immediately subsequent to [A], In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising, from N-terrninus to C- terminus, [A][B],
[0117] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence having the formula [A] [13] (SEQ ID NO: 4699), wherein [A] comprises X1 X2 X3 X4 X5 X6 and [B] comprises SPHKSG (SEQ ID NO: 946). In some embodiments, X1 of [A] is T, M, A, C, I, R L, D, F, V, Q, N, or H. In some embodiments, X2 of [A] is I, P, E, N, D, S, A, T, M, or Q. In some embodiments, X3 of [A] is N, E, G, Y, W, M, T, I, K, Q, F, S, V, A, or L. In some embodiments. X4 of [A] is G, D, R, or E. In some embodiments, X5 of [A] is H, Q, N, or D In some embodiments, X6 of [A] is D or R. In some embodiments, [A] comprises TINGHD (SEQ ID NO: 5297), MPEGHD (SEQ ID NO: 5298), MEGGHD (SEQ ID NO: 5299), MEYGHD (SEQ ID NO: 5300), AEWGHD (SEQ ID NO: 5301), CEWGHD (SEQ ID NO: 5302), ANNGQD (SEQ ID NO: 5303), IPEGHD (SEQ ID NO: 5304), ADMGHD (SEQ ID NO: 5305), IEYGIID (SEQ ID NO: 5306), ADYGHD (SEQ ID NO: 5307), IETGHD (SEQ ID NO: 5308), MEWGHD (SEQ ID NO: 5309), CEYGHD (SEQ ID NO: 5310). R1NGHD (SEQ ID NO: 5311), MEIGHD (SEQ ID NO: 5312), LEYGHD (SEQ ID NO: 5313), ADWGHD (SEQ ID NO: 5314), IEIGHD (SEQ ID NO: 5315), TIKDND (SEQ ID NO: 5316), DIMGIID (SEQ ID NO: 5317), FEQGHD (SEQ ID NO: 5318), MEFGHD (SEQ ID NO: 5319), CDQGHD (SEQ ID NO: 5320), LPEGHD (SEQ ID NO: 5321), lENGHD (SEQ ID NO: 5322), MESGHD (SEQ ID NO: 5323), AEIGIID (SEQ ID NO: 5324), VEYGIID (SEQ ID NO: 5325), TSNGDD (SEQ ID NO: 5326), IEVGHD (SEQ ID NO: 5327), MEMGHD (SEQ ID NO: 5328), AEVGHD (SEQ ID NO: 5329), MDAGHD (SEQ ID NO: 5330), VEWGHD (SEQ ID NO: 5331), AEQGHD (SEQ ID NO: 5332), LEWGHD (SEQ ID NO: 5333), MELGHD (SEQ ID NO: 5334), METGHD (SEQ ID NO: 5335), MEAGHD (SEQ ID NO: 5336), TTNRQR (SEQ ID NO: 5337), IESGHD (SEQ ID NO: 5338), TAKDHD (SEQ ID NO: 5339), MEVGHD (SEQ
ID NO: 5340), CEIGHD (SEQ ID NO: 5341), ATNGHD (SEQ ID NO: 5342), MDGGIID (SEQ ID NO: 5343), QEVGHD (SEQ ID NO: 5344), ADQGHD (SEQ ID NO: 5345), NMNGHD (SEQ ID NO: 5346), TPWEHD (SEQ ID NO: 5347), IEMGHD (SEQ ID NO: 5348), TANEHD (SEQ ID NO: 5349), QQQGIID (SEQ ID NO: 5350), TPQDHD (SEQ ID NO: 5351), HDWGHD (SEQ ID NO: 5352), IEGGHD (SEQ ID NO: 5353), or any dipeptide, tripeptide, tetrapeptide, or pentapeptide thereof. In some embodiments, [A][B] comprises TINGHDSPHKR (SEQ ID NO: 5354), MPEGHDSPHKS (SEQ ID NO: 5355), MEGGHDSPHKS (SEQ ID NO: 5356), MEYGHDSPHKS (SEQ ID NO: 5357), AEWGHDSPHKS (SEQ ID NO: 5358), CEWGHDSPHKS (SEQ ID NO: 5359), ANNGQDSPHKS (SEQ ID NO: 5360), IPEGHDSPHKS (SEQ ID NO: 5361), ADMGHDSPIIKS (SEQ ID NO: 5362), IEYGIIDSPHKS (SEQ ID NO: 5363), ADYGHDSPHKS (SEQ ID NO: 5364), IETGHDSPHKS (SEQ ID NO: 5365), MEWGHDSPHKS (SEQ ID NO: 5366), CEYGHDSPHKS (SEQ ID NO: 5367), RINGHDSPHKS (SEQ ID NO: 5368), MEIGHDSPHKS (SEQ ID NO: 5369), LEYGHDSPHKS (SEQ ID NO: 5370), ADWGHDSPHKS (SEQ ID NO: 5371), IEIGHDSPHKS (SEQ ID NO: 5372), TIKDNDSPHKS (SEQ ID NO: 5373), DIMGHDSPHKS (SEQ ID NO: 5374). FEQGHDSPHKS (SEQ ID NO: 5375), MEFGHDSPHKS (SEQ ID NO: 5376), CDQGHDSPHKS (SEQ ID NO: 5377), LPEGHDSPHKS (SEQ ID NO: 5378), IENGHDSPHKS (SEQ ID NO: 5379), MESGHDSPHKS (SEQ ID NO: 5380), AEIGHDSPHKS (SEQ ID NO: 5381), VEYGHDSPHKS (SEQ ID NO: 5382), TSNGDDSPHKS (SEQ ID NO: 5383), IEVGHDSPHKS (SEQ ID NO: 5384), MEMGHDSPIIKS (SEQ ID NO: 5385), AEVGHDSPHKS (SEQ ID NO: 5386), MDAGHDSPHKS (SEQ ID NO: 5387), VEWGHDSPHKS (SEQ ID NO: 5388). AEQGHDSPHKS (SEQ ID NO: 5389), LEWGHDSPHKS (SEQ ID NO: 5390), MELGHDSPHKS (SEQ ID NO: 5391), METGHDSPHKS (SEQ ID NO: 5392), MEAGHDSPIIKS (SEQ ID NO: 5393), TINRQRSPHKS (SEQ ID NO: 5394), IESGHDSPHKS (SEQ ID NO: 5395), TAKDHDSPHKS (SEQ ID NO: 5396), MEVGHDSPHKS (SEQ ID NO: 5397), CEIGHDSPHKS (SEQ ID NO: 5398), ATNGHD SPHKS (SEQ ID NO: 5399), MDGGHDSPHKS (SEQ ID NO: 5400), QEVGHDSPHKS (SEQ ID NO: 5401), ADQGHDSPHKS (SEQ ID NO: 5402), NMNGHDSPHKS (SEQ ID NO: 5403), TPWEHDSPHKS (SEQ ID NO: 5404), IEMGHDSPHKS (SEQ ID NO: 5405), TANEHDSPHKS (SEQ ID NO: 5406), TINGHDSPHKS (SEQ ID NO: 5407), QQQGHDSPHKS (SEQ ID NO: 5408), TPQDHDSPHKS (SEQ ID NO: 5409),
HDWGHDSPIIKS (SEQ ID NO: 5410), IEGGHDSPHKS (SEQ ID NO: 5411 ), or any portion thereof, e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof. In some embodiments, [B] is present immediately subsequent to [A], In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising, from N-terminus to C -terminus, [A][B],
[8118] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, at least 16, or at least 17 consecutive amino acids from any one of the sequences provided in Table 1, 2A, 2B, or 11-21. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least 3, at least 4, or at least 5 consecutive amino acids from any one of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, or at least 13 consecutive amino acids from any one of SEQ ID NOs: 2, 200, 201, 941 , 943, 204, 208, 404, or 903-909.
[0119] In some embodiments, the at least 3 consecutive amino acids comprise SPH. In some embodiments, the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700). In some embodiments, the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701). In some embodiments, the at least 6 consecutive amino acids comprise SPHS KA (SEQ ID NO: 941).
[0128] In some embodiments, at least 3 consecutive amino acids comprise HDS. In some embodiments, the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO: 4702). In some embodiments, the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703). In some embodiments, the at least 6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2).
[0121] In some embodiments, the at least 3 consecutive amino acids comprise SPH. In some embodiments, the at least 4 consecutive amino acids comprise SPHK (SEQ ID NO: 6398). In some embodiments, the at least 5 consecutive amino acids comprise SPHKY (SEQ ID NO: 4715). In some embodiments, the at least 6 consecutive amino acids comprise SPHKYG (SEQ ID NO: 966).
[0122] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of any one of the sequences provided
in Table 1, 2A, 2B, or 11-21. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids, relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, or 11-21. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of any one of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941. 943, 2.04, 208, 404, or 903-909. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid, sequence comprising at least one, at least two, or at least three, but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of SEQ ID NO: 3589. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid, sequence comprising at least one, at least two, or at least three, but no more than tour different amino acids relative to the amino acid sequence of SEQ ID NO: 3589. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of SEQ ID NO: 1754. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids relative to the amino acid sequence of SEQ ID NO: 1754.
[0123] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941). In some embodiments, the peptide comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941).
[0124] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2). In some embodiments, the AAV capsid variant comprises (e.g., in loop IA7) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2). [0125] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of SPHKYG (SEQ ID NO: 966). In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids relative to the amino acid sequence of SPHKYG (SEQ ID NO: 966). [8126] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of any of the sequences provided in Table 1, 2A, 2B, or 11-21 . In some embodiments, the AAV capsid variant comprises (e.g., in loop IA7) the amino acid sequence of any of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of any of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 2.08, 404, or 903-909. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 941. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 943. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 2. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 3589. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV ) the amino acid sequence of SEQ ID NO: 1754. In some embodiments, the AAV capsid variant comprises (e.g., in loop IA7) the amino acid sequence of SEQ ID NO: 3241 . In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 4100. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 4062. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) the amino acid sequence of SEQ ID NO: 4486.
[0127] In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by a nucleotide sequence described herein, e.g., a nucleotide sequence of Table 2A. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by a nucleotide sequence comprising at least one.
at least two, at least three, at least four, at least five, at least six, or at least severs modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides, relative to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at. least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
In some embodiments, the .AAV capsid variant comprises (e.g., in loop IV) an amino acid sequence encoded by a nucleotide sequence described herein, e.g., a nucleotide sequence of Table 2.A. In some embodiments, the nucleotide sequence is codon optimized. In some embodiments, the nucleotide sequence is an isolated nucleotide sequence. In some embodiments, the nucleotide sequence is a recombinant nucleotide sequence.
[0128] In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relati ve to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides.
relative to the nucleotide sequence of SEQ ID NO; 942. In some embodiments, the nucleic acid sequence encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
[8129] In some embodiments, the nucleic acid encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 3, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 3. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides, relative to the nucleotide sequence of SEQ ID NO: 3. In some embodiments, the nucleic acid encoding an AAV capsid variant comprises a nucleotide sequence comprising the nucleotide sequence of SEQ ID NO: 3, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
[0130] In some embodiments, the nucleic acid encoding an A AV capsid vanant comprises the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments the nucleic acid encoding an AAV capsid variant comprises a nucleotide sequence comprising the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at: least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
[0131] Also provided herein are polynucleotide sequences encoding any of the AAV capsid variants described above, AAV particles, vectors, and cells comprising the same. [0132] In some embodiments, [N1]-[N2]-[N3] is present in loop IV of the AAV capsid variant. In some embodiments, [N0] and [N4] are present in loop IV of the AAV capsid
variant. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is present in loop IV of the AAV capsid variant.
[0133] In some embodiments, [N0] is present immediately subsequent to amino acid 449, relative to a reference sequence of SEQ ID NO: 138 (i.e., at a sequence position corresponding to that in SEQ ID NO: 138). In some embodiments, [N0] is present immediately subsequent to amino acid 449, numbered according to SEQ ID NO: 4, 36, 981, or 982 (i.e., at a sequence position corresponding to that in SEQ ID NO: 4, 36, 981, or 982). In some embodiments, [N0] replaces amino acids 450, 451 , and 452 (e.g., amino acids T450, 1451, and N452), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, [N0] is present immediately subsequent to amino acid 449 and [N0] replaces amino acids 450-452 (e.g., T450, 1451, and N452), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, [N1] is present immediately subsequent to sequence of SEQ ID NO: 453- 455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 138, 981, or 982. In some embodiments, [N1] is present immediately subsequent to amino acid 452, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, [N1] replaces amino acids 453-455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 4, 36, amino acid 452 and wherein [N1] replaces amino acids 4, 36, 138, 981, or 982. In some embodiments, [N2] is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, [N2]-[N3] is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments [N1]-[N2]-[N3] is present immediately subsequent to amino acid 452, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, [N1 ]-[N2]-[N3] replaces amino acids 453-455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 4, 36. 138, 981, or 982. In some embodiments, [N1] is present immediately subsequent to amino acid 452 and wherein [N1]-[N2]-[N3] replaces amino acids 453-455 (e.g., G453, S454, and G455), numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, [N4] is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 138, In some embodiments, [N4] replaces amino acids 456-459 (e.g., Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N4] is present immediately subsequent to amino acid 455, and [N4] replaces amino acids 456-459 (e.g., Q456, N457, Q458, and. Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N2]-[N3]-[N4] replaces amino acids 456-459 (e.g., Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N2]-[N3]-[N4] is
present immediately subsequent to amino acid 455, wherein [N2]-[N3]-[N4] replaces amino acids 456-459 (e.g., Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N1]-[N2]-[N3]-[N4] replaces amino acids 453-459 (e.g,, G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N1 ]-[N2]-[N3]-[N4] is present immediately subsequent to amino acid 452, and [N1]-[N2]-[N3]-[N4] replaces amino acids 453-459 (e.g., G453, S454, G455, Q456. N457, Q458, and. Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] replaces amino acids 450-459 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is present immediately subsequent, to amino acid 449, and wherein [N0]-[N1]-[N2]-[N3]-[N4] replaces amino acids 450-459 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, and Q459), numbered according to SEQ ID NO: 138.
[0134] In some embodiments, [N3] is present, immediately subsequent to [N2],
[0135] In some embodiments, the AAV capsid variant comprises, from N-terminus to C- terminus, [N2j-[N3], In some embodiments, the AAV capsid variant comprises, from N- terminus to C-terminus, [N1]-[N2]-[N3]. In some embodiments, the AAV capsid variant comprises, from N-termmus to C-terminus, [N1]-[N2]-[N3]-[N4], In some embodiments, the AAV capsid variant comprises, from N-terminus to C-terminus, [N0]-|NI]-[N2]-[N3]. In some embodiments, the AAV capsid variant comprises, from N-termmus to C-terminus, [N0]-[N1]-[N2]-[N3]-[N4] .
[0136] In some embodiments, an AAV capsid variant comprises an amino acid sequence having the formula [A][B] (SEQ ID NO: 4694), wherein [A] comprises the amino acid sequence of GSGSPH (SEQ ID NO: 4695) and [B] comprises X1 X2 X3 X4 X5 X6 X7. In some embodiments, X1 of [B] is S, C, F, or V. In some embodiments, .X2 of [B] is K, I.,, R I, E, Y, V, or S. In some embodiments, X3 of [B] is A, R, L, G, I, Y, S, F, or W. In some embodiments, X4 of [B] is W, Q, R, G, L, V, S, or F. In some embodiments, X5 of [B] is N, Y, R, C, K, or L. In some embodiments, X6 of [B] is Q, G, K, R, T, L, or Y. In some embodiments, X7 of [B] is Q, L, R, or V. In some embodiments, [B] comprises SLLWNQQ (SEQ ID NO: 5247), SKAQYYV (SEQ ID NO: 5248), SKLRRQQ (SEQ ID NO: 5249), SIWQNQQ (SEQ ID NO: 5250), SKAGCGQ (SEQ ID NO: 5251), SRAQNQQ (SEQ ID NO: 5252), SKRLRQQ (SEQ ID NO: 5253), SLRRNQQ (SEQ ID NO: 5254), SRGRNQQ (SEQ ID NO: 5255), SEIVNQQ (SEQ ID NO: 5256), SSRRNQQ (SEQ ID NO: 5257), CLLQNQQ (SEQ ID NO: 5258), SKAFRLQ (SEQ ID NO: 5259), CLAQNQQ (SEQ ID
NO: 5260), FLRQNQQ (SEQ ID NO: 5261), SLRFNQQ (SEQ ID NO: 5262), SYLRNQQ (SEQ ID NO: 5263), CSLQNQQ (SEQ ID NO: 5264), VLWQNQQ (SEQ ID NO: 5265), SKWLLQQ (SEQ ID NO: 5266), SLWSNQQ (SEQ ID NO: 5267), SKRRLQQ (SEQ ID NO: 5268), SVYLNQQ (SEQ ID NO: 5269), SLWLNQQ (SEQ ID NO: 5270), SKAQRKL (SEQ ID NO: 5271), SKALRRQ (SEQ ID NO: 5272), SKAQRLR (SEQ ID NO: 5273), SKA QNQQ (SEQ ID NO: 5274), SKAQRRL (SEQ ID NO: 5275), SKARRQQ (SEQ ID NO: 5276), SKARRLQ (SEQ ID NO: 5277), SKSRRQQ (SEQ ID NO: 5278), SKARLRQ (SEQ ID NO: 5279), SKASKRQ (SEQ ID NO: 5280), VRRQNQQ (SEQ ID NO: 5281), SKAQLYR (SEQ ID NO: 5282), SLFRNQQ (SEQ ID NO: 5283), SKAQLTV (SEQ ID NO: 5284), or any dipeptide, tripeptide, tetrapeptide, pentapeptide, or bexapeptide thereof. In some embodiments, [A][B] comprises GSGSPHSLLWNQQ (SEQ ID NO: 5285), GSGSPHSKAQYYV (SEQ ID NO: 2060), GSGSPHSKLRRQQ (SEQ ID NO: 2061), GSGSPHSIWQNQQ (SEQ ID NO: 5286), GSGSPHSKAGCGQ (SEQ ID NO: 2062), GSGSPHSRAQNQQ (SEQ ID NO: 2063), GSGSPHSKRLRQQ (SEQ ID NO: 2064), GSGSPHSLRRNQQ (SEQ ID NO: 2065), GSGSPHSRGRNQQ (SEQ ID NO: 2066), GSGSPHSEIVNQQ (SEQ ID NO: 5287), GSGSPHSSRRNQQ (SEQ ID NO: 2067), GSGSPHCLLQNQQ (SEQ ID NO: 5288). GSGSPHSKAFRLQ (SEQ ID NO: 2068), GSGSPHCLAQNQQ (SEQ ID NO: 5289), GSGSPHFLRQNQQ (SEQ ID NO: 2070), GSGSPHSLRFNQQ (SEQ ID NO: 2071), GSGSPHSYLRNQQ (SEQ ID NO: 5290), GSGSPHCSLQNQQ (SEQ ID NO: 5291), GSGSPHVLWQNQQ (SEQ ID NO: 5292), GSGSPHSKWLLQQ (SEQ ID NO: 2072), GSGSPHSLWSNQQ (SEQ ID NO: 5293), GSGSPHSKRRLQQ (SEQ ID NO: 2073), GSGSPHSVYLNQQ (SEQ ID NO: 5294), GSGSPHSLWLNQQ (SEQ ID NO: 5295), GSGSPHSKAQRKL (SEQ ID NO: 2074), GSGSPHSKALRRQ (SEQ ID NO: 2075), GSGSPHSKAQRLR (SEQ ID NO: 2076), GSGSPHSKAQNQQ (SEQ ID NO: 1801), GSGSPHSKAQRRL (SEQ ID NO: 2077), GSGSPHSKARRQQ (SEQ ID NO: 2078), GSGSPIISKARRLQ (SEQ ID NO: 2079), GSGSPHSKSRRQQ (SEQ ID NO: 2080), GSGSPHSKARLRQ (SEQ ID NO: 2082), GSGSPHSKASKRQ (SEQ ID NO: 2083), GSGSPHVRRQNQQ (SEQ ID NO: 2084), GSGSPHSKAQLYR (SEQ ID NO: 2085), GSGSPHSLFRNQQ (SEQ ID NO: 5296), GSGSPHSKAQLTV (SEQ ID NO: 2086), or any portion thereof; e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof.
[0137] In some embodiments, [A] [IB] is present in loop IV of the AAV capsid variant. In some embodiments, [A] is present immediately subsequent to amino acid 452, relative to a reference sequence of SEQ ID NO: 138 (i.e., at a sequence position corresponding to that in
SEQ ID NO: 138). In some embodiments, [A] replaces amino acids 453-455 (e.g., G453, S454, G455), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [A] is present immediately subsequent to amino acid 452, and [A] replaces amino acids 453- 455 (e.g., G453, S454, G455), relative to a. reference sequence of SEQ ID NO: 138. In some embodiments, [B] is present immediately subsequent to [A], In some embodiments, [B] replaces amino acids 456-459 (e.g., Q456, N457, Q458, Q459), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [A][B] replaces amino acids 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, Q459), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [A][B] is present immediately subsequent to amino acid 452, and wherein [A][B] replaces amino acids 453-459 (e.g., G453, S454, G455, Q456, N457, Q458, Q459), relative to a. reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises, from N-terminus to C-terminus, [A] [B] . [0138] In some embodiments, an AAV capsid variant comprises an amino acid sequence having the formula [A][B] (SEQ ID NO: 4699), wherein [A] comprises X1 X2 X3 X4 X5 X6 and [B] comprises SPHKSG (SEQ ID NO: 946). In some embodiments, X1 of [A] is T, M, A, C, I, R, L, D, F, V, Q, N, or H. In some embodiments, X2 of [A] is I, P, E, N, D, S, A, T,
M, or Q. In some embodiments, X 3 of [A] is N, E, G, Y, W, M, T. I, K, Q, F, S. V. A. or L. In some embodiments, X4 of [A] is G, D, R, or E, In some embodiments, X5 of [A] is H, Q,
N, or D. In some embodiments, X6 of [A] is D or R. In some embodiments, [A] comprises TINGHD (SEQ ID NO: 5297), MPEGHD (SEQ ID NO: 5298), MEGGHD (SEQ ID NO: 5299), MEYGHD (SEQ ID NO: 5300), AEWGHD (SEQ ID NO: 5301), CEWGHD (SEQ ID NO: 5302), ANNGQD (SEQ ID NO: 5303), IPEGIID (SEQ ID NO: 5304), ADMGHD (SEQ ID NO: 5305), IEYGHD (SEQ ID NO: 5306), ADYGHD (SEQ ID NO: 5307), IETGHD (SEQ ID NO: 5308), MEWGHD (SEQ ID NO: 5309), CEYGHD (SEQ ID NO: 5310), RINGHD (SEQ ID NO: 5311), MEIGHD (SEQ ID NO: 5312), LEYGHD (SEQ ID NO: 5313), ADWGHD (SEQ ID NO: 5314), IEIGIID (SEQ ID NO: 5315), TIKDND (SEQ ID NO: 5316), DIMGHD (SEQ ID NO: 5317), FEQGHD (SEQ ID NO: 5318), MEFGHD (SEQ ID NO: 5319), CDQGHD (SEQ ID NO: 5320), LPEGHD (SEQ ID NO: 5321), IE.NGHD (SEQ ID NO: 5322), MESGHD (SEQ ID NO: 5323), AEIGIID (SEQ ID NO: 5324), VEYGHD (SEQ ID NO: 5325), TSNGDD (SEQ ID NO: 5326), IEVGHD (SEQ ID NO: 5327), MEMGHD (SEQ ID NO: 5328). AEVGHD (SEQ ID NO: 5329), MDAGHD (SEQ ID NO: 5330), VEWGHD (SEQ ID NO: 5331), AEQGHD (SEQ ID NO: 5332), LEWGHD (SEQ ID NO: 5333), MELGHD (SEQ ID NO: 5334), METGIID (SEQ ID NO: 5335), MEAGHD (SEQ ID NO: 5336), TINRQR (SEQ ID NO: 5337), IESGHD (SEQ ID NO:
5338), TAKDIID (SEQ ID NO; 5339), MEVGHD (SEQ ID NO: 5340), CEIGHD (SEQ ID NO: 5341), ATNGHD (SEQ ID NO: 5342), MDGGHD (SEQ ID NO: 5343), QEVGHD (SEQ ID NO: 5344), ADQGHD (SEQ ID NO: 5345), NMNGHD (SEQ ID NO: 5346), TPWEIID (SEQ ID NO; 5347), IEMGIID (SEQ ID NO: 5348), TANEHD (SEQ ID NO: 5349), QQQGHD (SEQ ID NO: 5350), TPQDHD (SEQ ID NO: 5351), HDWGHD (SEQ ID NO: 5352), IEGGHD (SEQ ID NO: 5353), or any dipeptide, tripeptide, tetrapeptide, or pentapeptide thereof. In some embodiments, [A][B] comprises TINGHDSPHKR (SEQ ID NO: 5354), MPEGHDSPHKS (SEQ ID NO: 5355), MEGGHDSPHKS (SEQ ID NO: 5356), MEYGHDSPHKS (SEQ ID NO: 5357), AEWGHDSPHKS (SEQ ID NO: 5358), CEWGHDSPHKS (SEQ ID NO: 5359), ANNGQDSPHKS (SEQ ID NO: 5360), IPEGHDSPHKS (SEQ ID NO: 5361), ADMGHDSPHKS (SEQ ID NO: 5362), 1EYGHDSPHKS (SEQ ID NO: 5363), ADYGHDSPHKS (SEQ ID NO: 5364), IETGHDSPHKS (SEQ ID NO: 5365), MEWGHDSPHKS (SEQ ID NO: 5366). CEYGHDSPHKS (SEQ ID NO: 5367), RINGHDSPHKS (SEQ ID NO: 5368), MEIGHDSPHKS (SEQ ID NO: 5369), LEYGHDSPHKS (SEQ ID NO: 5370), ADWGHDSPHKS (SEQ ID NO: 5371), IEIGHDSPHKS (SEQ ID NO: 5372), TIKDNDSPHKS (SEQ ID NO: 5373), DIMGHDSPHKS (SEQ ID NO: 5374), FEQGHDSPHKS (SEQ ID NO: 5375), MEFGHDSPHKS (SEQ ID NO: 5376), CDQGHDSPHKS (SEQ ID NO: 5377), LPEGHDSPHKS (SEQ ID NO: 5378), IENGHDSPHKS (SEQ ID NO: 5379). MESGHDSPHKS (SEQ ID NO: 5380), AEIGHDSPHKS (SEQ ID NO: 5381), VEYGHDSPHKS (SEQ ID NO: 5382), TSNGDDSPHKS (SEQ ID NO: 5383), IEVGIIDSPHKS (SEQ ID NO: 5384), MEMGHDSPHKS (SEQ ID NO: 5385), AEVGHDSPHKS (SEQ ID NO: 5386), MDAGHDSPHKS (SEQ ID NO: 5387), VEWGHDSPHKS (SEQ ID NO: 5388). AEQGHDSPHKS (SEQ ID NO: 5389), LEWGHDSPHKS (SEQ ID NO: 5390), MELGHDSPHKS (SEQ ID NO: 5391), METGHDSPHKS (SEQ ID NO: 5392), MEAGHDSPHKS (SEQ ID NO: 5393), TINRQRSPHKS (SEQ ID NO: 5394), IESGHDSPHKS (SEQ ID NO: 5395), TAKDHDSPHKS (SEQ ID NO: 5396), MEYGHDSPHKS (SEQ ID NO: 5397), CEIGHDSPHKS (SEQ ID NO: 5398), ATNGHDSPHKS (SEQ ID NO: 5399), MDGGHDSPHKS (SEQ ID NO: 5400), QEVGHDSPHKS (SEQ ID NO: 5401), ADQGHDSPHKS (SEQ ID NO: 5402), NMNGHDSPHKS (SEQ ID NO: 5403), TPWEHDSPHKS (SEQ ID NO: 5404), IEMGHDSPHKS (SEQ ID NO: 5405), TANEHDSPHKS (SEQ ID NO: 5406), TINGHDSPHKS (SEQ ID NO: 5407), QQQGHDSPHKS (SEQ ID NO: 5408),
TPQDHDSPHKS (SEQ ID NO: 5409k HDWGHDSPHKS (SEQ ID NO: 5410), IEGGHDSPHKS (SEQ ID NO: 5411), or any portion thereof; e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , or 12 amino acids, e.g., consecutive amino acids, thereof.
[0139] In some embodiments, [A] [B] is present in loop IV of the AAV capsid variant. In some embodiments, [A] is present immediately subsequent to amino acid 449, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [A] replaces amino acids 450-455 (e.g., T450, 1451, N452, G453, S454, G455), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [A] is present immediately subsequent to amino acid 449, and wherein [A] replaces amino acids 450-455 (e.g., T450, 1451, N452, G453, S454, G455), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [B] is present immediately subsequent to [A], In some embodiments, [A][B] replaces amino acids 450-455 (e.g., T450, 1451, N452, G453, S454, G455), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, [A][B] is present immediately subsequent to amino acid 449, and wherein [A][B] replaces amino acids 450-455 (e.g., T450, 1451, N452, G453, S454, G455), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the peptide comprises, from N-tenninus to C -terminus, [A][B|.
[0140] In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 16, or at least 17 consecutive amino acids from anyone of the sequences provided in Table 1, 2A, 2B, or 11-21. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, or at least 5 consecutive amino acids from any one of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, or at least 13 consecutive amino acids from any one of SEQ ID NOs: 2, 200, 201, 941 , 943, 204, 208, 404, or 903-909. In some embodiments, the amino acid sequence is present in loop IV. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 448, 452, 453, or 455, numbered according to SEQ ID NO: 4, 36, 138, 981 , or 982 (i.e., at a sequence position corresponding to that in SEQ ID NO: 4, 36, 138, 981 , or 982). In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 982. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 453, numbered according to SEQ ID NO: 981. In some
embodiments, the amino acid sequence is present immediately subsequent to amino acid 453, numbered according to SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or all of amino acids 499 (e.g,, K499), 450 (e.g., T450), 451 (e.g., 1451 ), 452 (e.g., N452), 453 (e.g., G453), 454 (e.g., S454), 455 (e.g., G455), 456 (e.g., Q456), 457 (e.g., N457), 458 (e.g., Q458), 459 (e.g., Q459), and 460 (e.g., T460), numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises one or more amino acid substitutions at amino acids 499 (e.g., K499), 450 (e.g,, T450), 451 (e.g., 1451), 452 (e.g., N452), 453 (e.g., G453), 454 (e.g., S454), 455 (e.g., G455), 456 (e.g., Q456), 457 (e.g., N457), 458 (e.g., Q458), 459 (e.g., Q459), and/or 460 (e.g., T460), numbered according to SEQ ID NO: 138.
[0141] In some embodiments, the at. least 3 consecutive amino acids comprise SPH. In some embodiments, the at least 3 consecutive amino acids comprise SPH in an AAV9 variant. In some embodiments, the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700). In some embodiments, the at least 4 consecutive amino acids comprise SPHS (SEQ ID NO: 4700) in an AAV9 variant. In some embodiments, the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701). In some embodiments, the at least 5 consecutive amino acids comprise SPHSK (SEQ ID NO: 4701) in an AAV9 variant. In some embodiments, the at least 6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941). in some embodiments, the at least 6 consecutive amino acids comprise SPHSKA (SEQ ID NO: 941) in an AAV9 variant.
[0142] In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present at amino acids 456-461, numbered according to SEQ ID NO: 981. In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present at amino acids 456-461 of an AAV9 variant, numbered according to SEQ ID NO: 981. In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present at amino acids 456-461 , numbered according to SEQ ID NO: 4. In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present at amino acids 456-461 of an AAV9 variant, numbered according to SEQ ID NO: 4. In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present at amino acids 456-461, numbered according to SEQ ID NO: 36. In some embodiments, the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present at amino acids 456-461 of an AAV9 variant, numbered according to SEQ ID NO: 36.
[0143] In some embodiments, the at least 3 consecutive amino acids comprise HDS In some embodiments, the at least 3 consecutive amino acids comprise HDS in an AAV9
variant. In some embodiments, the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO: 4702). In some embodiments, the at least 4 consecutive amino acids comprise HDSP (SEQ ID NO: 4702) in an AAV9 variant. In some embodiments, the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703). In some embodiments, the at least 5 consecutive amino acids comprise HDSPH (SEQ ID NO: 4703) in an AAV9 variant. In some embodiments, the at least 6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2). In some embodiments, the at least 6 consecutive amino acids comprise HDSPHK (SEQ ID NO: 2) in an AAV9 variant.
[0144] In some embodiments, the amino acid sequence of HDSPHK (SEQ ID NO: 2) is present in at amino acids 454-459, numbered according to SEQ ID NO: 982. In some embodiments, the amino acid sequence of HDSPHK (SEQ ID NO: 2) is present in an AAV9 variant at amino acids 454-459, numbered according to SEQ ID NO: 982.
[0145] In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of any one of the sequences provided in Table 1 , 2A, 2B, or 11-21 . In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids, relative to the amino acid sequence of any one of the sequences provided m Table 1 , 2A, 2B, or 11-21. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of any one of SEQ II) NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than tour modifications, relative to the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids, from the amino acid sequence of any one of SEQ ID NOs: 2, 200, 201, 941, 943, 204, 208, 404, or 903-909. In some embodiments, the amino acid sequence is present in loop IV. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 448, 452, 453, or 455, numbered according to SEQ ID NO: 4, 36, 138, 981, or 982. In some embodiments, the amino acid sequence is present immediately
subsequent to amino acid 455, numbered according to SEQ ID NO: 982. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 138. In some embodiments, the amino acid, sequence is present immediately subsequent to amino acid 453, numbered according to SEQ ID NO: 981, 4, or 36. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 453, numbered according to SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 , or all of amino acids 499 (e.g., K499), 450 (e.g., T450), 451 (e.g., 1451), 452 (e.g., N452), 453 (e.g., G453), 454 (e.g., S454), 455 (e.g., G455), 456 (e.g., Q456), 457 (e.g., N457), 458 (e.g., Q458), 459 (e.g., Q459), and 460 (e.g., T460), numbered according to SEQ ID NO: 138.
[0146] In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of SPHSKA (SEQ ID NO: 941). In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids from the amino acid sequence of SPHSKA (SEQ ID NO: 941).
[0147] In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four modifications, relative to the amino acid sequence of HDSPHK (SEQ ID NO: 2). In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than four different amino acids that relative to the amino acid, sequence of HDSPHK (SEQ ID NO: 2).
[0148] In some embodiments, the AAV capsid variant comprises an amino acid sequence provided in Table 1, 2A, 2B, or 11-21. In some embodiments, the amino acid sequence comprises any one of SEQ ID NOs: 945-980 or 985-986. In some embodiments, the AAV capsid variant comprises the amino acid sequence of any of SEQ ID NOs: 2, 200, 201 , 941, 943, 204, 208, 404, or 903-909. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 941. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 2. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 943. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 3589. In some embodiments, the AAV capsid variant comprises the amino acid, sequence of SEQ ID NO: 1754. In some embodiments, the amino acid sequence is present in loop IV. In some embodiments, the amino acid sequence is present immediately subsequent to amino
acid 448, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 448 and replaces amino acids 449-460 (e.g., K449, T450, 1451, N452, G453, S454, G455, Q456. N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 449, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 450-460 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 449, and replaces amino acids 450-460 (e.g., T450, 1451, N452, G453, S454, G455, Q456, N457. Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 450, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 451-460 (e.g., 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 450 and replaces amino acids 451-460 (e.g., 1451, N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 451, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 452-460 (e.g., N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 451 and replaces amino acids 452-460 (e.g., N452, G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 452, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 453-460 (e.g., G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 452, and replaces amino acids 453-460 (e.g., G453, S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 453, relative to a reference
sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 453, and replaces amino acids 454 and 455 (e.g., S454 and G455), numbered according to SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces amino acids 454-460 (e.g., S454, G455, Q456. N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138, In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 453, and replaces amino acids 454-460 (e.g., S454, G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present, immediately subsequent to amino acid 454, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 454, relative to a reference sequence of SEQ ID NO: 981. In some embodiments, the amino acid sequence replaces amino acids 455-460 (e.g., amino acids G455, Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acids 454, and replaces amino acids 455-460 (e.g., amino acids G455, Q456, N457, Q458, Q459. and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent, to amino acid 455, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 455, relative to a reference sequence of SEQ ID NO: 982. In some embodiments, the amino acid sequence replaces amino acids 456-460 (e.g., Q456, N457, Q458, Q459, and T460), numbered relative to SEQ ID NO: 138. In some embodiments, the amino acid sequence is present immediately subsequent to amino acid 455, and replaces amino acids 456-460 (e.g., Q456, N457, Q458, Q459, and T460), numbered, relative to SEQ ID NO: 138.
[0149] In some embodiments, the AAV capsid variant (e.g., an AAV capsid variant described herein), comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 942 or 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the AAV capsid variant described herein comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 3 or 942, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ
ID NO: 3 or 942. In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 3 or 942.
[0150] In some embodiments, the nucleotide sequence encoding the .AAV capsid variant (e.g., an AAV capsid variant described herein), comprises tire nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleic acid sequence encoding the AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequences of SEQ ID NO: 942. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides, relative to the nucleotide sequence of SEQ ID NO: 942.
[0151] In some embodiments, the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 3, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleic acid sequence encoding the AA V capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least tour, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequences of SEQ ID NO: 3. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 3.
[0152] In some embodiments, the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 5, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleic acid sequence encoding the AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least
five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequences of SEQ ID NO: 5. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 5.
[8153] In some embodiments, the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 12, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleic acid sequence encoding the AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequences of SEQ ID NO: 12. In some embodiments, the nucleotide sequence encoding an AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 12.
[0154] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to amino acid 455, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to amino acid 455, relative to a reference sequence of SEQ ID NO: 981.
[0155] In some embodiments, an AAV capsid variant comprises the amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to amino acid 453, relative to a. reference sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant comprises tire amino acid sequence of HDSPHK (SEQ ID NO: 2), wherein the amino acid sequence is present immediately subsequent to amino acid 453, relative to a reference sequence of SEQ ID NO: 982.
[0156] In some embodiments, an AAV capsid variant comprises (i) the amino acid sequence ofHDSPHSKA (SEQ ID NO: 4486), which is present immediately subsequent to amino acid 453; and (ii) a deletion of amino acids SG at amino acid 454 and 455; wherein (i) and (ii) are numbered according to SEQ ID NO: 138.
[0157] In some embodiments, an AAV capsid variant comprises an amino acid other than S at amino acid 454 and/or an amino acid other than G at amino acid 455, numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises the amino acid II at amino acid 454 and the amino acid D at amino acid 455, numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941). In some embodiments, the AAV capsid variant comprises: (i) the amino acid H at amino acid 454 and the amino acid D at amino acid 455, and (ii) the amino acid sequence SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence of SPHKSG (SEQ ID NO: 946) is present immediately subsequent to amino acid 455, wherein (i) and. (ii) are numbered according to SEQ ID NO: 138, [0158] In some embodiments, an AAV capsid variant comprises a modification, e.g., substitution, relative to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a modification, e.g., substitution, at amino acid S454 and/or G455, numbered relative to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a S454H substitution and/or G455D substitution, numbered relative to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a S454H substitution and a G455D substitution, numbered relative to SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941). In some embodiments, the AAV capsid variant comprises: (i) a S454H substitution and a G455D substitution, and (ii) the amino acid sequence SPHKSG (SEQ ID NO: 946), wherein the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present immediately subsequent to amino acid 455, wherein (i) and (ii) are numbered according to SEQ ID NO: 138.
[0159] In some embodiments, the .AAV capsid variant further comprises one, two, or all of an amino acid other than T at amino acid 450 (e.g., S, Y, or G), an amino acid other than I at amino acid 451 (e.g., M or L), and/or an amino acid other than N at amino acid 452 (e.g., S), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an S at amino acid 450 and an M at amino acid 451, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a Y at amino acid 450, an L at amino acid 451, and an S at amino acid 452, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a G at amino acid 450, an L at amino acid 451, and an S at amino acid 452, relative to a reference sequence of SEQ ID NO: 138.
[0160] In some embodiments, the AAV capsid variant further comprises one, two, three, four, or ail of an amino acid other than Q at amino acid 456 (e.g., R or L), N at amino acid
457 (e.g., H , K, or R), Q at amino acid 458 (e.g., R or T), Q at amino acid 459 (H), and/or T at amino acid 460 (N or S), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an R at amino acid 456, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an L at amino acid 456, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an H at amino acid 457 and an R at amino acid 458, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a K at amino acid 457 and an N at amino acid 460, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T at amino acid 458, an H at amino acid 459, and an S at amino acid 460, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an R at amino acid 456, an R at amino acid 457, and an R at amino acid 458, relative to a reference sequence of SEQ ID NO: 138.
[0161] In some embodiments, an AAV capsid variant comprises an amino acid other than I at amino acid 451, an amino acid other than N at amino acid 452, and an amino acid other than G at amino acid 453, numbered according to SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant comprises E at amino acid 45 I, R at amino acid 452, and V at amino acid 453, numbered according to SEQ ID NO: 138 or 981.
[0162] In some embodiments, the AAV capsid variant comprises the substitutions I451E, N452R, and G453V, numbered according to SEQ ID NO: 138 or 981.
[0163] In some embodiments, the AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to amino acid 455 and wherein the AAV capsid variant comprises the E at amino acid 451, R at amino acid 452, and V at amino acid 453, numbered according to SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant comprises the substitutions 145 IE, N452R and G453V, and further comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to amino acid 455, numbered according to SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant comprises the amino acid sequence of ERVSGSPHSKA (SEQ ID NO: 6399), wherein the amino acid sequence is present immediately subsequent to amino acid 449 and replaces amino acids 450-455, numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589), wherein the amino acid sequence is
present immediately subsequent to amino acid 448 and replaces amino acids 449-460, numbered according to SEQ ID NO: 138.
[0164] In some embodiments, an AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138, 981, or 4; and (ii) one or both of E at position 451 and/or V at position 453, numbered according to the amino acid sequence of SEQ ID NO : 4, 138, or 981. In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138, 981, or 4; and (ii) one or both of E at position 451 and/or V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 4, 138, or 981 , wherein the AAV capsid variant is an AAV9 variant.
[8165] In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138, 981, or 4; and (ii) E at position 451 and V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 4, 138, or 981. In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to position 455, numbered according to SEQ ID NO: 138, 981, or 4; and (ii) E at position 451 and V at position 453, numbered according to the amino acid, sequence of SEQ ID NO: 4, 138, or 981, wherein the AAV capsid variant is an AAV9 variant.
[0166] In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 4, 138, or 981 (re., at a sequence position corresponding to that in SEQ ID NO: 4, 138, or 981) and (ii) one or both of E at position 451 and/or V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 4, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 4, 138, or 981). In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 4, 138, or 981 and (ii) one or both of E at position 451 and/or V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 4, 138, or 981, wherein the AAV capsid variant is an AAV9 variant.
[0167] In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 4, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 4, 138, or 981); and (ii) E at position 451 and V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 4, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 4, 138, or 981). In some embodiments, the AAV capsid variant comprises: (i) the amino acid, sequence of SPHSKA (SEQ ID NO: 941 ), wherein the amino acid sequence is present at amino acids 456- 461, numbered according to SEQ ID NO: 4, 138, or 981, and (ii) E at position 451 and V at position 453, numbered according to the amino acid, sequence of SEQ ID NO: 4, 138, or 981, wherein the AAV capsid variant is an AAV9 variant.
[0168] In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 36, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 36, 138, or 981), and (ii) one, two, or all of E at position 451, R at position 452, and/or V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 36, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 36, 138, or 981). In some embodiments, the AAV capsid, variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 36, 138, or 981 and (ii) one, two, or all of E at position 451 , R at position 452, and/or V at. position 453, numbered according to the amino acid sequence of SEQ ID NO: 36, 138, or 981, wherein the .AAV capsid variant is an AAV9 variant.
[0169] In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 36, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 36, 138, or 981); and (ii) E at position 451, R at position 452, and V at position 453, numbered according to the amino acid sequence of SEQ ID NO: 36, 138, or 981 (i.e., at a sequence position corresponding to that in SEQ ID NO: 36, 138, or 981). In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present at amino acids 456-461, numbered according to SEQ ID NO: 36, 138, or 981 and (ii) E at position 451, R at position 452, and V at position 453, numbered according to the amino
acid sequence of SEQ ID NO: 36, 138, or 981, wherein the AAV capsid variant is an AAV9 variant.
[0170] In some embodiments, an AAV capsid variant described herein comprises an amino acid other than T at amino acid 450, an amino acid other than I at amino acid 451, and an amino acid other than N at amino acid 452, numbered according to SEQ ID NO: 138 or 982. In some embodiments, the AAV capsid variant comprises A at amino acid 450, E at amino acid 451, and I at amino acid 452, numbered according to SEQ ID NO: 138 or 982. In some embodiments, the AAV capsid variant comprises the substitutions T450A, 145 IE, and N452I, numbered according to SEQ ID NO: 138 or 982.
[0171] In some embodiments, the AAV capsid variant comprises the amino acid sequence of HDSPHK (SEQ ID NO: 2), which is present immediately subsequent to amino acids 453, and further comprises A at amino acid 450, E at amino acid 451 , and I at amino acid 452, all numbered according to SEQ ID NO: 138 or 982. In some embodiments, the AAV capsid variant comprises the substitutions T450A, 145 IE, and N452.I, and further comprises the amino acid sequence HDSPHK (SEQ ID NO: 2) present immediately subsequent to amino acid 453, all numbered according to SEQ ID NO: 138 or 982. In some embodiments, the AAV capsid variant comprises the amino acid sequence of AEIGHDSPHKSG (SEQ ID NO: 6400), wherein the amino acid sequence is present immediately subsequent to amino acid 449 and replaces amino acids 450-455, numbered according to SEQ ID NO: 138.
[6172] In some embodiments, the AAV capsid variant comprises the amino acid sequence of KAEIGHDSPHKSGQNQQT (SEQ ID NO: 1754), wherein the amino acid, sequence is present immediately subsequent to amino acid 448 and replaces amino acids 449- 460, numbered according to SEQ ID NO: 138.
[0173] In some embodiments, the AAV capsid variant further comprises a substitution at amino acid K449, e.g., a K449R substitution, numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant, further comprises an amino acid other than K at amino acid 449 (e.g., R), relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises an R at amino acid 449, relative to a reference sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a modification, e.g., an insertion, substitution, and/or deletion in loop I, II, VI, and/or VIII.
[0174] In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, of the amino acid sequence of SEQ ID NO:
138. In some embodiments, the AAV capsid variant further comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 amino acids that differ from the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises the amino acid sequence of SEQ ID NO: 138, or an amino acid sequence with at least 70% (e.g., at least 80%, at least 85%, at least 90, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
[0175] In some embodiments, the AAV capsid variant comprises (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982, 36, or 4; (b) a VP2 protein comprising amino acids 138-742 of SEQ ID NO: 982, 36, or 4; (c) a VP3 protein comprising amino acids 203-742 of SEQ ID NO: 982, 36, or 4; or (d) an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any of the amino acid sequences in (a)-(c), an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids relative to any of the amino acid sequences in (a)-(c), or an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 2.0, or not more than 10 modifications, relative to any of the amino acid sequences in (a)-(c).
[0176] In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence that is at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 137. In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 2.0, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 137. In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 137.
[0177] In some embodiments, the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence that is at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%%) identical to SEQ ID NO: 137. In some embodiments, the nucleotide sequence
encoding the AAV capsid variant comprises a. nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more tlian 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 137. In some embodiments, the nucleotide sequence encoding the AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, or at least three, but not more tlian 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 137.
[0178] In some embodiments, an AAV capsid variant of the present disclosure comprises an amino acid sequence as described herein, e.g., an amino acid sequence of an AAV capsid variant of TIM-001 or TIM-002, e.g., as described in Tables 3 and 4. In some embodiments, an AAV capsid variant of the present disclosure comprises an amino acid sequence as described herein, e.g., an amino acid sequence of an AAV capsid variant of TTM-003, TTM-004, TTM-005. TIM-006, TTM-007, TTM-008, TIM-009, TTM-010, TTM-01 1, TTM-012, TTM-013, TIM-014, TTM-015, TTM-016, TTM-017, TTM-018, TIM-019, TIM-020, TIM-021, TTM-022, TIM-023, TTM-024, TTM-025, TIM-026, or TIM-027, e.g., as described in Table 4. In some embodiments, the AAV capsid variant comprises an amino acid sequence of SEQ ID NO: 36 (TTM-003; comprising a peptide of SEQ ID NO: 3589), SEQ ID NO: 39 (TTM-006; comprising a peptide of SEQ ID NO: 3241), or SEQ ID NO: 4 (TIM-027; comprising a peptide of SEQ ID NO: 3272).
[8179] In some embodiments, an AAV capsid variant comprises a VP1, VP2, and/or VP3 protein comprising an amino acid sequence described herein, e.g., an amino acid, sequence of an AAV capsid variant of TTM-001 or TIM-002, e.g., as described in Tables 3 and 4. In some embodiments, an AAV capsid variant comprises a VP I, VP2, and/or VP3 protein comprising an amino acid sequence described herein, e.g., an amino acid sequence of an AAV capsid variant of TTM-003, TTM-004, TTM-005, TTM-006, TTM-007, TTM-008, TTM-009, TTM-010, TTM-011 , TTM-012, TTM-013, TTM-014, TTM-015, TTM-016, TTM-017, TTM-018, TTM-019. TIM-020, TIM-021, TTM-022, TIM-023, TTM-024, TTM-025, TIM-026 or TTM-027 e.g., as described in Table 4.
[0188] In some embodiments, an AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence as described herein, e.g., a nucleotide sequence of an AAV capsid variant of TTM-001 or TIM-002, e.g., as described in Tables 3 and 5. In some embodiments, an AAV capsid, variant comprises an amino acid sequence encoded by a nucleotide sequence as described herein, e.g., a nucleotide sequence of an AAV capsid variant of TTM-003, TTM-004, TTM-005, TTM-006, TTM-007, TTM-008, TTM-009,
TIM-010, TTM-011, TTM-012, TTM-013, TTM-014, TIM-015, TTM-016, TTM-017,
TTM-018, TTM-019, TTM-020, TTM-021, TTM-022, TTM-023, TTM-024, TIM-025,
TTM-026, or TTM-027 e.g., as described in Table 5.
[0181] In some embodiments, a polynucleotide or nucleic acid encoding an AAV capsid variant of the present disclosure comprises a nucleotide sequence described herein, e.g., a nucleotide sequence of an AAV capsid variant of TTM-001 or TTM-002, e.g., as described in
Tables 3 and 5. In some embodiments, a polynucleotide or nucleic acid encoding an AAV capsid variant of the present disclosure comprises a nucleotide sequence described herein. e.g., a nucleotide sequence of an .AAV capsid variant of TTM-003, TTM-004, TTM-005,
TIM-006, TTM-007, TIM-008, TIM-009, TTM-010, TTM-011, TTM-012, TTM-013,
TTM-014, TIM-015, TTM-016, TTM-017, TTM-018, TTM-019, TTM-020, TTM-021 ,
TTM-022, TTM-023, TTM-024, TIM-025, TTM-026, or TTM-027, e.g., as described in
Table 5.
[0182] In some embodiments, the polynucleotide encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 983 or 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least
95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
[0183] In some embodiments, the polynucleotide encoding an AAV capsid vanant comprises the nucleotide sequence of any one of SEQ ID NOs: 5, 12-35, or a. nucleotide
sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto,
[0184] In some embodiments, the polynucleotide encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 12 or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
[0185] In some embodiments, the polynucleotide encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 5 or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
[0186] In some embodiments, the polynucleotide encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three modifications but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 983. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions relative to the amino acid sequence of SEQ ID NO: 983. In some embodiments, the nucleic acid sequence encoding an AAV capsid variant is codon optimized. In some embodiments, the polynucleotide encoding an AAV capsid variant comprises SEQ ID NO: 983. In some embodiments, the polynucleotide encoding an AAV capsid variant comprises SEQ ID NO: 983. In some embodiments, the polynucleotide encoding an AAV capsid variant consists of SEQ ID NO: 983.
[0187] In some embodiments, the polynucleotide encoding an AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three
modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID MO: 984. In some embodiments, the nucleotide sequence encoding an A AV capsid variant described, herein, comprises a. nucleotide sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 984. In some embodiments, the nucleic acid sequence encoding an AAV capsid, variant is codon optimized. In some embodiments, the polynucleotide encoding an AAV capsid variant comprises SEQ ID NO: 984. In some embodiments, the polynucleotide encoding an AAV capsid variant consists of SEQ ID NO: 984.
[0188] In some embodiments, an AAV capsid variant comprises the amino acid sequence of any one of SEQ ID NOs: 4, 36-59, 981 , or 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant, comprises an amino acid sequence comprising at least one, at least, two, or at least, three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 4, 36-59, 981, or 982. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not. more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 4, 36-59, 981, or 982.
[0189] In some embodiments, the AAV capsid variant comprises one or more substitutions in loop IV and comprises the amino acid sequence of SEQ ID NO: 981 , or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 981. In some embodiments, an AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions, relative to the amino acid sequence of SEQ ID NO: 981.
[0190] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 70% (e.g., at least. 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least
98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 982. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 982. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 97% identical to SEQ ID NO: 982. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 98% identical to SEQ ID NO: 982. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 982. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982. In some embodiments, an AAV capsid variant consists of the amino acid sequence of SEQ ID NO: 982. In some embodiments, an AAV capsid variant comprises amino acid residues 2-742 of SEQ ID NO: 982. In some embodiments, an AAV capsid variant consists of amino acid residues of 2-742 of SEQ ID NO: 982.
[0191] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 36, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 36. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 36. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 97% identical to SEQ ID NO: 36. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 98% identical to SEQ ID NO: 36. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 36. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 36. In some embodiments, an AAV capsid variant consists of the amino acid sequence of SEQ ID NO: 36. In some embodiments, an AAV capsid variant comprises
amino acid residues 2-742 of SEQ ID NO: 36. In some embodiments, an AAV capsid variant consists of amino acid residues of 2-742 of SEQ ID NO: 36.
[0192] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 4, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 4. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 4. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 97% identical to SEQ ID NO: 4. In some embodiments, an AAV capsid variant comprises an amino acid, sequence that is at least 98% identical to SEQ ID NO: 4. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 4. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 4. In some embodiments, an AAV capsid variant consists of the amino acid sequence of SEQ ID NO: 4. In some embodiments, an .AAV capsid variant comprises amino acid residues 2-742 of SEQ ID NO: 4. In some embodiments, an AAV capsid variant consists of amino acid residues of 2-742 of SEQ ID NO: 4.
[0193] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 39, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid, sequence of SEQ ID NO: 39. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 39. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 97% identical to SEQ ID NO: 39. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 98% identical to SEQ ID NO: 39. In some embodiments, an AAV
capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 39. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 39, In some embodiments, an AAV capsid variant consists of the amino acid sequence of SEQ ID NO: 39. In some embodiments, an AAV capsid variant comprises amino acid residues 2-742 of SEQ ID NO: 39. In some embodiments, an AAV capsid variant consists of amino acid residues of 2-742 of SEQ ID NO: 39.
[0194] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 51 , or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 51. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 51. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 97% identical to SEQ ID NO: 51. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 98% identical to SEQ ID NO: 51. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 51. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 51. In some embodiments, an AAV capsid variant consists of the amino acid sequence of SEQ ID NO: 51. In some embodiments, an AAV capsid variant comprises amino acid residues 2-742 of SEQ ID NO: 51. In some embodiments, an AAV capsid variant consists of amino acid, residues of 2-742 of SEQ ID NO: 51 .
[0195] In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 52, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 52. In some embodiments, the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different
amino adds, relative to the amino acid sequence of SEQ ID NO: 52. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 97% identical to SEQ ID NO: 52. In some embodiments, an AAV capsid variant, comprises an amino acid sequence that is at least 98% identical to SEQ ID NO: 52. In some embodiments, an AAV capsid variant comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 52. In some embodiments, an AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 52. In some embodiments, an AAV capsid variant consists of the amino acid sequence of SEQ ID NO: 52. In some embodiments, an AAV capsid variant comprises amino acid residues 2-742 of SEQ ID NO: 52. In some embodiments, an AAV capsid variant consists of amino acid, residues of 2-742 of SEQ ID NO: 52.
[0196] In some embodiments, an AAV capsid variant, comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983 or 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least. 95%, at. least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions, relative to the amino acid sequence of SEQ ID NO: 983. In some embodiments, an AAV capsid, variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 983.
[0197] In some embodiments, an AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of any one of SEQ ID NOs: 5, 12-35, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at. least. 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 2.0, or not more than 10 different nucleotides, relative to the amino acid sequence of any one of SEQ ID NOs: 5, 12-35. In some embodiments, an AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of any one of SEQ ID NOs: 5, 12-35.
[0198] In some embodiments, an AAV capsid variant comprises a VP1, VP2, VP3 protein, the VP1, VP2, and VP3 comprise one or more insertions in loop IV. In some embodiments, an AAV capsid variant comprises the amino acid, sequence corresponding to amino acids 138-742, e.g., a VP2, of SEQ ID NO; 981 or 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid protein comprises the amino acid sequence corresponding to amino acids 203-742, e.g., a VP3, of SEQ ID NO: 981 or 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence corresponding to amino acids 1-742, e.g., a VP1, of SEQ ID NO: 981 or 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto, [0199] In some embodiments, an AAV capsid variant comprises the amino acid sequence corresponding to amino acids 138-742, e.g., a VP2, of SEQ ID NO: 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%, e.g., 100%) sequence identity thereto. In some embodiments, the AAV capsid protein comprises the amino acid sequence corresponding to amino acids 203-742, e.g., a VP3, of SEQ ID NO: 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%, e.g., 100%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence corresponding to amino acids 1-742, e.g., a VP1, of SEQ ID NO: 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%, e.g., 100%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises or consists of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the AAV capsid variant comprises or consists of amino acid residues 2-742 of SEQ ID NO; 982.
[0200] In some embodiments, an AAV capsid variant comprises the amino acid sequence corresponding to amino acids 138-742, e.g., a VP2, of SEQ ID NO: 36, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid protein comprises the amino acid sequence
corresponding to amino acids 203-742, e.g., a VP3, of SEQ ID NO: 36, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence corresponding to amino acids 1-742, e.g., a VP1, of SEQ ID NO: 36, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises or consists of the amino acid sequence of SEQ ID NO: 36. In some embodiments, the AAV capsid variant comprises or consists of amino acid residues 2-742 of SEQ ID NO: 36.
[0201] In some embodiments, an AAV capsid variant comprises the amino acid sequence corresponding to amino acids 138-742, e.g., a VP2, of SEQ ID NO: 4, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid protein comprises the amino acid sequence corresponding to amino acids 203-742, e.g., a VP3, of SEQ ID NO: 4, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence corresponding to amino acids 1-742, e.g., a VP1, of SEQ ID NO: 4, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises or consists of the amino acid sequence of SEQ ID NO: 4. In some embodiments, the AAV capsid variant comprises or consists of amino acid residues 2-742 of SEQ ID NO: 4.
[0202] In some embodiments, an AAV capsid variant comprises a VP1 , VP2, VP3 protein, or a combination thereof. In some embodiments, an AAV capsid variant comprises the amino acid sequence corresponding to amino acids 138-742, e.g., a VP2, of any one of SEQ ID NOs: 4, 36-59, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid protein comprises the amino acid sequence corresponding to amino acids 203-742, e.g., a VP3, of any one of SEQ ID NOs: 4, 36-59, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least
98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence corresponding to amino acids 1-742, e.g., a VP1, of any one of SEQ ID NOs: 4, 36-59, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
[8203] In some embodiments, an AAV capsid variant has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord, cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising SEQ ID NO: 138.
[0204] In some embodiments, an AAV capsid variant transduces a brain region, e.g., a midbrain region (e.g., the hippocampus, or thalamus) or the brain stem. In some embodiments, the level of transduction is at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65-fold greater as compared to a reference sequence of SEQ ID NO: 138. In some embodiments, the level of transduction is at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65 -fold greater as compared to an AAV capsid variant comprising SEQ ID NO: 138.
[0205] In some embodiments, an AAV capsid variant is enriched at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, or at least 10-fold in the brain compared to a reference sequence of SEQ ID NO: 138. In some embodiments, an .AAV capsid variant is enriched at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80 or at least 85-fold in the brain compared to an AAV capsid variant comprising SEQ ID NO: 138.
[0206] In some embodiments, an AAV capsid variant is enriched in the brain of at least two to three species, e.g., a non-human primate and rodent (e.g., mouse) species, compared to an AAV capsid variant comprising SEQ ID NO: 138. In some embodiments, an AAV capsid variant is enriched at least 2, at least 3, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100, at least 105, at least 1 15, at least 120, at least 125, at least 130, at least 135, at least 140, at least 145, at least 150, at least 155, at least 160, at least 165, at least 170, at least 175, at least 180, at least 190, at least 200, at least 205, or at least 210-fold in the brain of at least two to three species, e.g., a non-huraan primate and rodent (e.g., mouse) species, compared to an AAV capsid variant comprising SEQ ID NO: 138. In some embodiments, the at least two to three
species are Macacafascicularis, CMorocebus sabaeus, Callithnx jacchus, and/or mouse (e.g., outbred mice).
[0207] In some embodiments, an AAV capsid variant is enriched at least 2, at least 2.5, at least 3, at least 3.5, at least 4, at least 4.5, at least 5, at least 5.5, at least 6, at least 6.5, at least 7, at least 7.5, or at least 8-fold, in the brain compared to an AAV capsid variant comprising SEQ ID NO: 981. In some embodiments, an AAV capsid variant is enriched about 2, 2.5, 3, 3.5, 4, 4.5, 5, or 5.5-fold, in the brain compared to an AAV capsid variant comprising SEQ ID NO: 982.
[0208] In some embodiments, an AAV capsid variant delivers an increased level of viral genomes to a brain region. In some embodiments, the level of viral genomes is increased by at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, or at least 50-fold, as compared to an AAV capsid comprising SEQ ID NO: 138. In some embodiments, the brain region comprises a midbrain region (e.g., the hippocampus or thalamus) and/or the brainstem. [0209] In some embodiments, an AA V capsid variant delivers an increased level of a payload to a brain region. In some embodiments, the level of the payload is increased by at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, or at least 70-fold, as compared to an AAV capsid comprising SEQ ID NO: 138, In some embodiments, the brain region comprises a midbrain region (e.g., the hippocampus or thalamus) and/or the brainstem.
[8210] In some embodiments, an AAV capsid variant is enriched at least 5, at least 10, at least 15, at least 2.0, at least 2.5, at least 30, or at least 35-fold, in the spinal cord compared, to an AAV capsid comprising SEQ ID NO: 138.
[0211] In some embodiments, an AAV capsid variant shows preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG). In some embodiments, the AAV capsid variant shows preferential transduction in a brain region relative to the transduction in the liver. In some embodiments, the AAV capsid variant shows preferential transduction in a brain region relative to the transduction in the liver and the DRG In some embodiments, the AAV capsid variant shows preferential transduction in a brain region relative to the transduction in the heart. In some embodiments, the AAV capsid variant shows preferential transduction in a brain region relative to the transduction in the heart and DRG In some embodiments, the AAV capsid variant shows preferential transduction in a brain region relative to the transduction in the heart, DRG, and liver. In some embodiments, the AAV capsid variant show's preferential transduction in a brain region and/or a heart region relative to the transduction in the liver and DRG.
[0212] In some embodiments, an AAV capsid variant is capable of transducing non- neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes). In some embodiments, the AAV capsid variant is capable of transducing neuronal cells and non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes). In some embodiments, the non-neuronal cells are glial cells, oligodendrocytes (e.g., Olig2 positive oligodendrocytes), or astrocytes (e.g., Olig2 positive astrocytes). In some embodiments, the AAV capsid variant is capable of transducing Olig2 positive cells, e.g,, Olig2 positive astrocytes or Olig2 positive oligodendrocytes.
[0213] In some embodiments, an AAV capsid variant of the present disclosure has decreased tropism for the liver. In some embodiments, an AAV capsid variant comprises a modification that results in reduced, tropism (e.g., de-targeting) and/or activity in the liver. In some embodiments, the reduced tropism in the liver is compared to an otherwise similar capsid that does not comprise the modification, e.g., a wild-type capsid polypeptide. In some embodiments, an AAV capsid variant comprises a modification that results in one or more of the following properties: (1) reduced tropism in the liver; (2) reduced, e.g., de-targeted expression in the liver: (3) reduced activity in the liver: and/or (4) reduced binding to galactose. In some embodiments, the reduction in any one or all of properties (l)-(3) is compared to an otherwise similar AAV capsid variant that does not compose the modification.
[8214] Exemplary modifications are provided in WO 2018/119330; Pulicherla et al. (2011)A/o/. Ther. 19(6): 1070-1078; Adachi et al. (2014) Nature Communications 5(3075), DOI: 10.1038/ncomms4075; and Bell et al. (2012) J. Virol. 86(13): 7326-33; the contents of which are hereby incorporated by reference in their entirety. In some embodiments, the AAV capsid variant comprises a modification at amino acid N470 (e.g., N470A), D271 (e.g., D271A), N272 (e.g., N272A), Y446 (e.g., Y446A), N498 (e.g., N498Y or N498I), W503 (e.g., W503R or W503A), L620 (e.g., L620F), or a combination thereof, as numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises one, two, three, four, five, or all of an amino acid other than N at amino acid 470 (e.g.. A), an amino acid other than D at amino acid 271 (e.g., A), an amino acid other than N at amino acid 272 (e.g., A), an amino acid other than Y at amino acid 446 (e.g.. A), and amino acid other than N at amino acid 498/ (e.g., Y or I), and amino acid other th an W at amino acid 503 (e.g., R or A), and amino acid other than L at amino acid 620 (e.g., F), as numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a. modification at amino acid N470 (e.g., N470A), D271 (e.g., D271A), N272 (e.g., N272A),
¥446 (e.g., Y446A), and W503 (e.g., W503R or W503A), numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a modification at N498 (e.g., N498Y) and L620 (e.g., L620F).
[0215] In some embodiments, the AAV capsid variant comprises a modification as described in Adachi et al. (2014) Nature Communications 5(3075), DOI; 10.1038/ncomms4075, the contents of which are hereby incorporated by reference in its entirety. Exemplary modifications that alter or do not alter tissue transduction in at least the brain, liver, heart, lung, and/or kidney can be found in Supplementary Data 2 showing the AAV Barcode-Seq data obtained with AAV9-AA-VBCLib of Adachi et al. (supra), the contents of which are hereby incorporated by reference in their entirety .
[0216] In some embodiments, the AAV capsid variant is an isolated capsid variant. In some embodiments, the AAV capsid variant is a recombinant capsid variant. In some embodiments, a polynucleotide encoding an AAV capsid polypeptide, e.g., an AAV capsid variant is an isolated and/or a. recombinant AAV capsid polypeptide .
[0217] Also provided herein are polynucleotide sequences encoding any of the AAV capsid variants described above and .AAV particles, vectors, and cells comprising the same. Certain Properties of AAV Capsids
[0218] In some embodiments, an AAV particle of the present disclosure may comprise a capsid protein or variant thereof any natural or recombinant AAV serotype. AAV serotypes may differ in characteristics such as, but not limited to, packaging, tropism, transduction, and immunogenic profiles,
[0219] In some embodiments, an AAV capsid variant described herein allows for blood brain barrier penetration following intravenous administration. In some embodiments, the AAV capsid variant allows for blood brain barrier penetration following intravenous administration, focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. In some embodiments the AAV capsid variant allows for increased distribution to a brain region. In some embodiments, the brain region comprises a frontal cortex, sensory cortex, motor cortex, caudate, dentate nucleus, cerebellar cortex, cerebral cortex, brain stem, hippocampus, thalamus, putamen, or a combination thereof. In some embodiments, the .AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the dorsal root ganglia. (DRG). In some embodiments, the AAV capsid variant allows for preferential transduction in a. brain region relative to the transduction in the liver. In some embodiments, the AAV capsid variant allows for transduction in a non-
neuronal cell, e.g., a. glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof).
[0220] In some embodiments, an AAV capsid variant allows for increased distribution to a spinal cord region. In some embodiments, the spinal region comprises a. cervical spinal cord region, thoracic spinal cord region, and/or lumbar spinal cord region.
[8221] In some embodiments, the initiation codon for translation of the AAV VP1 capsid protein, e.g., a capsid variant, described herein may be CTG, TTG, or GTG as described in US Patent No. USS 163543, the con ten ts of which are herein incorporated by reference in its entirety.
[0222] The present disclosure refers to structural capsid proteins (including VP1, VP2. and VP3) which are encoded by capsid (Cap) genes. These capsid, proteins form an outer protein structural shell (e.g., capsid) of a viral vector such as AAV. VP capsid proteins synthesized from Cap polynucleotides generally include a methionine as the first amino acid in the peptide sequence (Metl), which is associated with the start codon (AUG or ATG) m the corresponding Cap nucleotide sequence. However, it is common for a first-methionine (Met1) residue or generally any first amino acid (AA 1) to be cleaved off after or during polypeptide synthesis by protein processing enzymes such as Met-aminopeptidases. This “Met/ AA -clipping” process often correlates with a corresponding acetylation of the second amino acid in the polypeptide sequence (e.g., alanine, valine, serine, threonine, etc.). Met- clipping commonly occurs with VP1 and VP3 capsid proteins but can also occur with VP2 capsid proteins.
[0223] Where the Met/ AA -clipping is incomplete, a. mixture of one or more (one, two or three) VP capsid proteins comprising the viral capsid may be produced, some of which may include a Metl/AA1 amino acid (Met+/AA+) and some of which may lack a Metl/AA1 amino acid as a result of Met/A A-clipping (Met-ZAA-). For further discussion regarding Met/AA-clipping in capsid proteins, see Jin, et al. Direct Liquid Chromatography/Mass Spectrometry Analysis for Complete Characterization of Recombinant Adeno-Associated Vims Capsid Proteins. Hum Gene Ther Methods . 2017 Oct. 28(5):255-267; Hwang, et al. N- Terminal Acetylation of Cellular Proteins Creates Specific Degradation Signals. Science. 2010 February-' 19. 327(5968): 973 -977; the contents of which are each incorporated herein by reference in its entirety.
[0224] According to the present disclosure, references to capsid proteins, e.g., AAV capsid variants, is not limited to either clipped (Met-ZAA-) or unclipped (Met+/AA+) and may, in context, refer to independent capsid proteins, viral capsids comprised of a mixture of
capsid proteins, and/or polynucleotide sequences (or fragments thereof) which encode, describe, produce, or result in capsid proteins of the present disclosure. A direct reference to a capsid protein or capsid polypeptide (such as VP1, VP2 or VP2) may also comprise VP capsid proteins which include a Metl/AA1 amino acid (Met+/AA+) as well as corresponding VP capsid proteins which lack the Metl/AA1 amino acid as a result of Met/AA-clipping (Met-/AA-).
[0225] Further according to the present disclosure, a reference to a specific SEQ ID NO: (whether a. protein or nucleic acid) which comprises or encodes one or more capsid proteins which include a Metl/AA1 amino acid (Met+/AA+) should be understood to teach the VP capsid proteins which lack the Metl/AA1 amino acid as upon review of the sequence, it is readily apparent any sequence which merely lacks the first listed amino acid (whether or not Metl/AA1 ).
[0226] As a non-limiting example, reference to a VP1 polypeptide sequence which is 736 amino acids in length, and which includes a “ Metl ” amino acid (Met+) encoded, by the AUG/ATG start codon may also be understood to teach a VP1 polypeptide sequence which is
735 amino acids in length, and which does not include the “Metl ” amino acid (Met-) of the
736 amino acid Met+ sequence. As a second non-limiting example, reference to a VP1 polypeptide sequence which is 736 amino acids in length, and which includes an “AA1.” amino acid (AA1+) encoded by any NNN initiator codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length, and which does not include the “AA1.” amino acid (AA1-) of the 736 amino acid A.A1+ sequence.
[0227] References to viral capsids formed from VP capsid proteins (such as reference to specific AAV capsid serotypes), can incorporate VP capsid proteins which include a Metl/AA1 amino acid (Met+/AA1+), corresponding VP capsid proteins which lack the Metl/AA1 amino acid as a. result, of Met/AA1 -clipping (Met-ZAA1-), and. combinations thereof (Met.+/AA1+ and Met-/AA1 -).
[0228] As a non-limiting example, an AAV capsid serotype can include VP1 (Met+ AA1.+), VP1 (Met-./AA 1-), or a combination of VP1 (Met+/AA1+) and VP1 (Met- /AA1-). An AAV capsid serotype can also include VP3 (Met+/AA1+), VP3 (Met-ZAA1 -), or a combination of VP3 (Met+/AA1+) and VP3 (Met-/AA1- ); and can also include similar optional combinations of VP2 (Metv/AA1 ) and VP2 (Met-ZAA1 -).
Additional AAV Sequences
[0229] In some embodiments, an AAV capsid polypeptide or AAV capsid variant described herein may comprise a VOY101 capsid polypeptide, an AAVPHP.B (PHP.B)
capsid polypeptide, a AAVPHP.N (PHP.N) capsid polypeptide, an AAV1 capsid polypeptide, an AAV2 capsid polypeptide, an AAV5 capsid polypeptide, an AAV9 capsid polypeptide, an AAV9 K449R capsid polypeptide, an AAVrhl 0 capsid polypeptide, or a functional variant thereof. In some embodiments, the AAV capsid polypeptide, e.g., AAV capsid variant, comprises an amino acid sequence of any of the AAV capsid polypeptides in Table 6, or an amino acid sequence substantially identical (e.g., having at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleotide sequence encoding the AAV capsid polypeptide comprises any one of the nucleotide sequences in Table 6, or a nucleotide sequence substantially identical (e.g., having at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
[0230] In some embodiments, an .AAV capsid polypeptide or an AAV capsid variant described herein comprises an amino acid sequence having at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity) to SEQ ID NO: 138. In some embodiments the AAV capsid polypeptide or the AAV capsid variant, comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, e.g., substitutions (e.g., conservative substitutions), but no more than 30, no more than 20, or no more than 10 modifications, e.g,, substitutions (e.g., conservative substitutions), relative to the amino acid sequence of SEQ ID NO: 138. In some embodiments, the .AAV capsid polypeptide or the AAV capsid variant, comprises an amino acid sequence encoded by a nucleotide sequence having at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) to SEQ ID NO: 137. In some embodiments, the nucleotide sequence encoding the AAV capsid polypeptide or the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 137 or a nucleotide sequence substantially identical (e.g,, having at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the AAV capsid polypeptide or the AAV capsid variant, comprises substitution at position K449, e.g., a K449R substitution, numbered, relative to SEQ ID NO: 138.
[0231] In some embodiments, the AAV capsid polypeptide or the AAV capsid variant, comprises a peptide comprising the amino acid sequence of TLAVPFK (SEQ ID NO: 4680). In some embodiments, the peptide is present immediately subsequent to position 588, numbered according to SEQ ID NO: 138. In some embodiments, the capsid polypeptide comprises the amino acid substitutions of A587D and Q588G, numbered according to SEQ ID NO: 138.
[0232] In some embodiments, the AAV capsid polypeptide or the AAV capsid variant comprises the amino acid substitution of K449R, numbered according to SEQ ID NO: 138; and a peptide comprising the amino acid sequence of TLAVPFK (SEQ ID NO: 4680), wherein the peptide is present immediately subsequent to position 588, numbered according to SEQ ID NO: 138.
[0233] In some embodiments, the AAV capsid polypeptide or the AAV capsid variant comprises the amino acid substitution of K449R, numbered according to SEQ ID NO: 138; a peptide comprising the amino acid sequence of TLAVPFK (SEQ ID NO: 4680), wherein the peptide is present immediately subsequent to position 588, numbered according to SEQ ID NO: 138; and the amino acid substitutions of A587D and Q588G, numbered according to SEQ ID NO: 138.
[0234] In some embodiments, the AAV capsid polypeptide or the AAV capsid variant comprises a peptide comprising the amino acid sequence of TLAVPFK (SEQ ID NO: 4680), wherein the insert is present immediately subsequent to position 588, numbered, according to SEQ ID NO: 138; and the amino acid substitutions of A587D and Q588G, numbered according to SEQ ID NO: 138.
[0235] In some embodiments, the AAV capsid polypeptide or the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 11 or an amino acid sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments the AAV capsid polypeptide or the AAV capsid variant, comprises an amino acid sequence comprising at least one, two, or three modifications, e.g., substitutions (e.g., conservative substitutions), but no more than 30, no more than 20, or no more than 10 modifications, e.g., substitutions (conservative substitutions), relative to the amino acid sequence of SEQ ID NO: 1 I, optionally wherein position 449 is not R.
[8236] In some embodiments, the AAV capsid polypeptide or AAV capsid variant, comprises the amino acid sequence of SEQ ID NO: I or an amino acid sequence substantially identical (e.g., having at. least. 70%, at least 75%, at least 80%, at least 85%, at least 90%, at. least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments the AAV capsid polypeptide or the AAV capsid variant, comprises an amino acid sequence comprising at least one, two, or three modifications, e.g., substitutions (e.g., conservative substitutions), but no more than 30, no more than 20, or no more than 10
modifications, e.g., substitutions (e.g., conservative substitutions), relative to the amino acid sequence of SEQ ID NO; 1.
AAV Viral Genome
[0237] In some embodiments, the AA V particle of the present disclosure serves as an expression vector comprising a. viral genome that encodes a modulatory polynucleotide. In various embodiments, a gene product can be expressed from an expression vector. In some embodiments, an mRNA encoding a protein is transcribed from the expression vector and translated into a protein. In some embodiments, a modulatory polynucleotide is transcribed from an expression vector and serves to reduce or eliminate expression of a separate gene product, such as a mutant ATXN2 mRNA or protein. In some embodiments, various regulatory elements may be used in an expression vector.
[0238] In some embodiments, an AAV particle, e.g., an AAV particle for the vectorized
delivery of modulatory polynucleotide described herein, comprises a. viral genome, e.g., an AAV viral genome (e.g., an AAV genome, vector genome, or .AAV vector genome). In some embodiments, the viral genome, e.g,, the AAV viral genome, further comprises an inverted terminal repeat (ITR) region, an enhancer, a. promoter, an intron region, an exon region, a. nucleic acid encoding a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA described herein), a polyA region, or a combination thereof. In some embodiments, the viral genome, e.g., the AAV viral genome, further comprises at least one miRNA binding site.
Viral Genome Component: Inverted Terminal Repeats (ITRs)
[0239] In some embodiments, the viral genome may comprise at least one inverted terminal repeat (ITR) region. The AAV particles of the present disclosure comprise a viral genome with at least one ITR region and a payload region. In some embodiments, the viral genome has two ITRs. These two ITRs flank the payload region at the 5’ and 3" ends. In some embodiments, the ITR functions as an origin of replication comprising a recognition site for replication. In some embodiments, the ITR comprises a. sequence region which can be complementary and symmetrically arranged. In some embodiments, the ITR incorporated into a viral genome described herein may be comprised of a naturally occurring polynucleotide sequence or a. recombinantly derived polynucleotide sequence.
[0240] In some embodiments, the ITR is of the same serotype as the capsid, selected from any of the serotypes known in the art, or a derivative thereof. In some embodiments. The ITR is of a different serotype than the capsid. In some embodiments, the AA V particle has more than one ITR. In some embodiments, the AAV particle comprises a viral genome comprising two ITRs. In some embodiments, the ITRs are of th e same serotype as one another. In some embodiments, the ITRs are of different serotypes. Non-limiting examples include zero, one, or both of the ITRs having the same serotype as the capsid , Viral Genome Component: Promoters and Enhancers
[0241] In some embodiments, the viral genome comprises at least one element to enhance the transgene target specificity and expression. See, e.g. , Powell et al. Viral Expression Cassette Elements to Enhance Transgene Target. Specificity and Expression in Gene Therapy, 2015; the contents of which are herein incorporated by reference in their entirety. Non- limiting examples of elements to enhance tire transgene target specificity and expression include promoters, endogenous miRNAs, post-transcriptional regulatory elements (PREs), polyadenylation (polyA) region, upstream enhancers (USEs), CMV enhancers, and introns.
[0242] In some embodiments, expression of the modulatory polynucleotide in a target cell may be driven by a specific promoter, including but not limited to, a promoter that is species specific, inducible, tissue-specific, or cell cycle-specific (Parr et al... Nat. M.ed3; V 145-9 (1997); the contents of which are herein incorporated by reference in their entirety).
[0243] In some embodiments, the viral genome comprises a promoter that is sufficient for expression, e.g., in a target cell, of a modulatory polynucleotide for reducing or eliminating expression of ATXN2, e.g., mutant ATXN2. protein. In some embodiments, the promoter is deemed to be efficient when it drives expression of the modulatory polynucleotide encoded in the viral genome of the AAV particle.
[0244] In some embodiments, the promoter is deemed to be efficient when it drives expression of the modulatory-' polynucleotide in the cell or tissue being targeted ,
[0245] Promoters may be naturally occurring or non-naturally occurring. Non-limiting examples of promoters include viral promoters, plant promoters and mammalian promoters. In some embodiments, the promoters may be human promoters. In some embodiments, the promoter may be truncated.
[0246] In some embodiments, the viral genome comprises a promoter that results in expression of the modulatory polynucleotide in one or more cells and/or tissues. In some embodiments, the promoter is a ubiquitous promoter. In some embodiments, a promoter which drives or promotes expression in most mammalian tissues includes, but is not limited to, human elongation factor la-subunit (EFlα) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken β-actin (CBA) promoter, a CAG promoter, a β glucuronidase (GUSB) promoter, and a. ubiquitin C (UBC) promoter.
[0247] In some embodiments, the viral genome comprises a nervous system specific promoter, e.g., a promoter that results in expression of a modulatory polynucleotide in a neuron, an astrocyte, and/or an oligodendrocyte. Non-limiting examples of tissue-specific expression elements for neurons include synapsm (Syn) or synapsin 1 (Syn1), e.g., human synapsm or synapsin 1.
[0248] In some embodiments, the promoter may be less than 1 kb.
[0249] In some embodiments, the promoter may be a combination of two or more components of the same or different starting or parental promoters.
[0250] In some embodiments, the viral genome comprises an enhancer.
[0251] In some embodiments, the viral genome comprises an engineered promoter.
Viral Genome Component: Introns
[0252] In some embodiments, the viral genome comprises at least one intron or a fragment or derivative thereof. In some embodiments, the viral genome comprises at least one exon or a fragment or derivative thereof.
[0253] In some embodiments, the intron may be 100-600 nucleotides in length.
[0254] In some embodiments, the modulatory polynucleotide-encoding sequence may be located within 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, or more than 30 nucleotides downstream from a promoter comprising an intron (e.g., 3’ relative to the promoter comprising an intron) and/or upstream of the polyadenylation sequence (e.g., 5" relative to the polyadenylation sequence). In some embodiments, the modulator}' polynucleotide-encoding sequence may be located within 1-5, 1-10, 1-15, 1-20, 1 -25, 1-30, 5-10, 5-15, 5-20, 5-25, 5-30, 10-15, 10-20, 10-25, 10-30, 15-20, 15-25, 15-30, 20-25, 20-30, or 25-30 nucleotides downstream from the intron (e.g., 3’ relative to the intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence). In some embodiments, the modulatory polynucleotide-encoding sequence may be located within the first 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 15%, 20%, 25%, or more than 25% of the nucleotides downstream from the intron (e.g., 3" relative to the intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence). In some embodiments, the modulator}' polynucleotide-encoding sequence may be located within the first 1-5%, 1-10%, 1-15%, 1-20%, 1-25%, 5-10%, 5- 15%, 5-20%, 5-25%, 10-15%, 10-20%, 10-25%, 15-20%, 15-25%, or 20-2.5% of the sequence downstream from the intron (e.g., 3’ relative to the intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence).
[8255] In certain embodiments, the intron sequence is not an enhancer sequence. In some embodiments, the intron sequence is not a. sub-component of a promoter sequence. In some embodiments, the intron sequence is a. sub-component of a. promoter sequence.
Viral Genome Component: Untranslated Regions (UTRs)
[0256] In some embodiments, a wild type untranslated region (UTR) of a gene is transcribed but not. translated. Generally, the 5’ UTR. starts at. the transcription start site and. ends at the start codon and the 3’ UTR starts immediately following the stop codon and continues until the termination signal for transcription.
[0257] Features typically found in abundantly expressed genes of specific target organs may be engineered into UTRs to enhance the stability and protein production, As a non- limiting example, a 5’ UTR from mRNA normally expressed in the liver (e.g., albumin,
serum amyloid A, Apolipoprotein A/B/E, transferrin, alpha fetoprotein, erythropoietin, or Factor VIII) may be used in the viral genomes of the .AAV particles of the disclosure to enhance expression in hepatic cell lines or liver.
[0258] In some embodiments, the viral genome encoding a modulatory polynucleotide described herein (e.g., a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA) comprises a Kozak sequence. While not wishing to be bound by theory, wild-type 5' untranslated regions (UTRs) include features that play roles in translation initiation. Kozak sequences, which are commonly known to be involved in the process by which the ribosome initiates translation of many genes, are usually included in 5’ UTRs. Kozak sequences have the consensus CCR(A/G)CCAUGG, where R is a purine (adenine or guanine) three bases upstream of the start codon (ATG), which is followed by another ’G’.
[0259] In some embodiments, the 5 ’UTR in the viral genome includes a Kozak sequence.
[8260] In some embodiments, the 5 "UTR in the viral genome does not include a Kozak sequence.
[0261] While not wishing to be bound by theory-, wild-type 3' UTRs are known to have stretches of adenosines and uridines embedded therein. These AU rich signatures are particularly prevalent in genes with high rates of turnover. Based on their sequence features and functional properties, the AU rich elements (AREs) can be separated into three classes (Chen et al, 1995, the contents of which are herein incorporated by reference in their entirety): Class I AREs, such as, but not limited to, c-Myc and MyoD, contain several dispersed copies of an AUUUA motif within U-rich regions. Class II AREs, such as, but not limited to, GM-CSF and TNF-a, possess two or more overlapping UUAUUUA(U/A)(U/A) nonamers. Class III ARES, such as, but not limited to, c-Jun and Myogenin, are less well defined. These U rich regions do not contain an AUUUA motif. Most proteins binding to the AREs are known to destabilize the messenger, whereas members of the ELAV family, most notably HuR, have been documented to increase the stability of mRNA. HuR binds to AREs of all the three classes. Engineering the HuR specific binding sites into the 3' UTR of nucleic acid molecules will lead, to HuR binding and thus, stabilization of the message in vivo.
[0262] Introduction, removal or modification of 3' UTR AU rich elements (AREs) can be used to modulate the stability of polynucleotides. When engineering specific polynucleotides, e.g., payload regions of viral genomes, one or more copies of an ARE can be introduced to make polynucleotides less stable and thereby curtail translation and decrease production of the resultant protein. Likewise, AREs can be identified and removed or mutated to increase the intracellular stability and thus increase translation and production of the resultant protein.
[0263] In some embodiments, the 3' UTR of the viral genome may include an oligo(dT) sequence for templated addition of a poly-A tail.
[0264] Any UTR. from any gene known in the art may be incorporated into the viral genome of the AAV particle. These UTRs, or portions thereof, may be placed in the same orientation as in the gene from which they were selected or they may be altered in orientation or location. In some embodiments, the UTR used in the viral genome of the AAV particle may be inverted, shortened, lengthened, or made with one or more other 5'' UTRs or 3'' UTRs known in the art. As used herein, the term “altered,” as it relates to a UTR, means that the UTR has been changed in some way in relation to a reference sequence. For example, a 3' or 5'' UTR may be altered relative to a wild type or native UTR. by the change in orientation or location as taugh t above or may be altered by the inclusion of additional nucleotides, deletion of nucleotides, swapping or transposition of nucleotides.
[8265] In some embodiments, the viral genome of the AAV particle comprises at least one artificial UTR, which is not a variant of a wild type UTR .
[0266] In some embodiments, the viral genome of the AAV particle comprises UTRs which have been selected from a family of transcripts -whose proteins share a common function, structure, feature, or property.
Viral Genome Component: Filler (Stuffer) Sequence
[0267] As used herein, the terms “staffer sequence” and “filler sequence” are used interchangeably. In some embodiments, the AAV particle viral genome comprises at least one filler sequence.
[0268] In some embodiments, the viral genome comprises one or more filler sequences. The filler sequence may be a wild-type sequence or an engineered sequence. A filler sequence may be a variant of a wild-type sequence.
[0269] In some embodiments, the viral genome comprises one or more filler sequences in order to have the length of the viral genome be the optimal size for packaging. In some embodiments, the viral genome comprises at least one filler sequence m order to have the length of the viral genome be about 2.3 kb. In some embodiments, the viral genome comprises at least one filler sequence in order to have the length of the viral genome be about 4.6 kb.
Viral Genome Component: Modulatory polynudeotide-eneoding sequences
[0278] In some embodiments, the disclosure provides an AAV particle comprising a viral genome encoding a modulatory polynucleotide, e.g., a modulatory polynucleotide for
reducing or eliminating expression of ATXN2, thereby reducing or eliminating ATXN2 protein. In some embodiments, the modulatory polynucleotide comprises or encodes an
RNAi agent, e.g., an siRNA or an ASO. In some embodiments, the modulatory polynucleotide comprises or encodes an RNAi agent. In some embodiments, the RNAi agent comprises an siRNA.
[8271] In some embodiments, the modulatory polynucleotides of the disclosure reduce or eliminate AT.XN2 mRNA, thereby reducing or eliminating ATXN2 protein. Exemplary
ATXN2 mRNA sequences are provided in Table 34. The mRNA sequences and accompanying information in Table 34 are hereby incorporated by reference in their entirety.
In some embodiments, the modulatory polynucleotide of the disclosure reduces or eliminates expression of one or more of (e.g., one, two, three or all four of) SEQ ID NOs: 6428, 6429,
6430, and 6431, or a trinucleotide repeat expansion of any of the foregoing.
[0272] In some embodiments, the modulatory polynucleotide comprises a guide strand and a passenger strand, wherein the guide strand targets and thus reduces or eliminates expression of one or more of (e.g., one, two, three or all four of) SEQ ID NOs: 642.8-6431 or a trinucleotide repeat expansion thereof. In some embodiments, the guide strand targeting any one of SEQ ID NOs: 6428-6431 or a trinucleotide repeat expansion thereof is fully complementary to a region of that ATXN2 mRNA sequence. In some embodiments, the guide strand targeting any one of SEQ ID NOs: 6428-6431 or a trinucleotide repeat expansion thereof is partially (e.g., substantially) complementary (e.g., at least 70%, at least
75%, at least 80%, at least 85%, at least 90% identical at least 91%, at least 92%, at least
93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%
complementary) to a. region of that ATXN2 mRNA sequence. In some embodiments, the guide strand has up to 1, up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 mismatches relative to a region of ATXN2 mRNA, e.g., to a region of any one of SEQ ID NOs: 6428-6431 or a trinucleotide repeat expansion thereof.
[0273] In some embodiments, the passenger strand and guide strand are fully complementary. In some embodiments, the passenger strand and guide strand are partially (e.g., substantially) complementary (permitting one or more mismatches). In some embodiments, the passenger strand is at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90% at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100%) complementary to the guide strand. In some embodiments, the passenger strand has up to up to 1 , up to 2, up to 3, up to 4, up to 5, up to 6, up to 7, up to 8, up to 9, or up to 10 mismatches relative to the guide strand.
[0274] In some embodiments, an shRNA comprises the passenger strand and. guide strand. In some embodiments, an siRNA comprises the passenger strand and guide strand.
[0275] In some embodiments, the modulatory polynucleotide binds to a coding region of ATXN2 mRN A. In some embodiments, the modulatory polynucleotide binds to a noncoding region of ATXN2 mRNA,
[0276] In some embodiments, the disclosure herein provides constructs that allow tor improved expression of a modulatory polynucleotide delivered by gene therapy vectors.
[0277] In some embodiments, the disclosure provides constructs that allow for improved biodistribution of a modulatory polynucleotide delivered by gene therapy vectors.
[0278] In some embodiments, the disclosure provides constructs that allow for improved sub-cellular distribution or trafficking of a modulatory polynucleotide delivered by gene therapy vectors.
[0279] In some embodiments, the disclosure provides constructs that allow for improved trafficking of a modulatory polynucleotide to lysosomal membranes delivered by gene therapy vectors.
[0280] In some embodiments, the present disclosure relates to a. composition containing or comprising a nucleic acid sequence encoding modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA or a functional fragment or variant thereof and methods of administering the composition in vitro or in vivo in a subject, e.g,, a. human subject and/or an animal model of disease, e.g., an ATXN2-related disorder.
[0281] In some embodiments, the disclosure provides a nucleotide sequence encoding a
modulatory polynucleotide for use in an AAV genome. In some embodiments, the nucleotide sequence further comprises one or more, e.g., all of, a 5’ ITR sequence, an enhancer sequence, a promoter sequence, an intron sequence, a poly A. sequence, and a 3’ ITR sequence.
[0282] In some embodiments, the AAV genome encodes a payload construct that comprises a combination of coding and non-coding nucleic acid sequences.
[0283] In some embodiments, the viral genome encodes more than one payload. As a non- limiting example, a viral genome encoding more than one payload may be replicated and packaged into a viral particle. A target cell transduced with a viral particle comprising more than one payload may express each of the payloads in a single cell.
[0284] In some embodiments, the viral genome encodes an inhibitory polynucleotide, e.g., an antisense oligonucleotide (ASO) or RNAi agent (e.g., a dsRNA, siRNA, shRNA, pre- miRNA, pri-miRNA, miRNA, stRNA, IncRN A, piRNA, or snoRNA). In some embodiments, the modulatory agent is an RNAi agent. In some embodiments, the RNAi agent is a siRNA. In some embodiments, the RNAi agent is a shRNA. In some embodiments, the modulatory agent is an ASO. In some embodiments, the ASO or siRNA comprises at least one (e.g., one or more or all) modified nucleotides.
[0285] A viral genome encoding a modulatory polynucleotide may further comprise or encode a selectable marker. A selectable marker may comprise a gene sequence or a protein or polypeptide encoded by a gene sequence expressed in a host cell that allows for the identification, selection, and/or purification of the host cell from a population of cells that may or may not express the selectable marker. In some embodiments, the selectable marker provides resistance to survive a selection process that would otherwise kill the host cell, such as treatment with an antibiotic. In some embodiments, an antibiotic selectable marker may comprise one or more antibiotic resistance factors, including but not limited to neomycin resistance (e.g., neo), hygromycin resistance, kanamyem resistance, and/or puromycin resistance.
[0286] In some embodiments, a viral genome encoding a modulatory polynucleotide may comprise a selectable marker including, but not limited to, p-lactamase, luciferase, β- galactosidase, or any other reporter gene as that term is understood in the art, including cell- surface markers, such as CD4 or the truncated nerve growth factor (NGFR) (for GFP, see WO 96/23810; Heim et al.. Current Biology 2: 178-182 (1996); Heim et al., Proc. Natl. Acad. Sci. USA (1995); or Heim et al. , Science 373:663-664 (1995); for P-lactamase, see WO 96/30540); the contents of each of which are herein incorporated by reference in their
entirety.
[0287] In some embodiments, a viral genome encoding a selectable marker may comprise a fluorescent protein. A. fluorescent protein as herein described may comprise any fluorescent marker including but not limited to green, yellow, and/or red fluorescent protein (GFP, YFP, and/or RFP). In some embodiments, the selectable marker may comprise a human influenza hemagglutinin (HA) tag.
[0288] In certain embodiments, a nucleic acid for expression of a modulatory polynucleotide in a target cell will be incorporated into the viral genome and located between two ITR sequences.
Viral Genome Component: Molecular Scaffolds
[0289] In some embodiments, the molecular scaffold of the modulatory polynucleotide is a known or wild type pri- or pre-microRNA. In other embodiments, the molecular scaffold of the modulatory polynucleotide is designed ab initio. In some embodiments, the molecular scaffold along with the payload (e.g., passenger strand and guide strand) form a stem loop structure.
[8290] In some embodiments, the molecular scaffold comprises a 5’ flanking region. In some embodiments, the molecular scaffold comprises a 3’ flanking region. In some embodiments, the molecular scaffold comprises a loop region, -wherein the loop region is present between the passenger and guide strand of the stem loop structure. In some embodiments, the molecular scaffold comprises one or more spacers between one or more modules of the modulatory polynucleotide. A module of the modulatory polynucleotide may include a 5’ flanking region, a 5' arm, a loop region, a 3’ arm, or a 3' flanking region. In some embodiments, a spacer is of sufficient length to form approximately one helical turn of the sequence.
[0291] In some embodiments, the molecular scaffold comprises a 5’ flanking region, a. loop region, and a 3’ flanking region. In some embodiments, the modulatory polynucleotide comprises, in the 5' to 3' direction, a 5' flanking sequence, a 5' arm comprising a passenger strand, or guide strand, a loop region, a 3' arm comprising a guide strand or passenger strand, respectively, and a. 3’ flanking sequence.
[0292] Exemplary sequences for the 5' flanking region, the loop region, and the 3' flanking region that may be used in the molecular scaffolds described herein are shown in Tables 7-9.
Table 7. Exemplary 5’ Flanking Regions for Molecular Scaffold
[0293] In some embodiments, the molecular scaffold comprises a 5' flanking region comprising any one of SEQ ID NOs: 6413-6416, or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least
95%, at least 96%, at least 97%, at least 98%, or at least 99% identical to any one of SEQ ID
NOs: 6413-6416; a loop region comprising any one of SEQ ID NOs: 6417-6421, or a. nucleotide sequence at least 75%, at least 80%, at least 85%, at least 90%, at least 95% at least 96%, at least 97%, at least 98%, or at least 99% identical to any one of SEQ ID NOs:
6417-6421 ; and a 3 ’ flanking region comprising any one of SEQ ID NOs: 6422-6427, or a nucleotide sequence at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical to any one of SEQ ID NOs: 6422-6427.
[0294] In some embodiments, the molecular scaffold comprises a 5’ flanking region comprising any one of SEQ ID NOs; 6413-6416; a loop region comprising any one of SEQ ID NOs: 6417-6421 ; and a 3’ flanking region comprising any one of SEQ ID NOs: 6422- 6427.
[0295] In some embodiments, the molecular scaffold comprises a 5’ flanking region comprising SEQ ID NO: 6414 or SEQ ID NO: 6415; a loop region comprising SEQ ID NO: 6417, SEQ ID NO: 6418, or SEQ ID NO: 6421 ; and a 3’ flanking region composing SEQ ID NO: 6422, SEQ ID NO: 6423, SEQ ID NO: 6424, or SEQ ID NO: 6425. In some embodiments, the molecular scaffold comprises a 5" flanking region comprising SEQ ID NO: 6414 or SEQ ID NO: 6415; a loop region composing SEQ ID NO: 6417, SEQ ID NO: 6418, or SEQ ID NO: 6421 ; and a 3’ flanking region comprising SEQ ID NO: 6423, SEQ ID NO: 6424, or SEQ ID NO: 6425.
[0296] In some embodiments, the molecular scaffold comprises a 5’ flanking region comprising SEQ ID NO: 6414, a loop region comprising SEQ ID NO: 6417, and a. 3’ flanking region comprising SEQ ID NO: 6423. In some embodiments, the molecular scaffold comprises a 5’ flanking region comprising SEQ ID NO: 6415, a loop region comprising SEQ ID NO: 6421, and a 3’ flanking region comprising SEQ ID NO: 6425. In some embodiments, the molecular scaffold comprises a 5' flanking region comprising SEQ ID NO: 6414, a loop region comprising SEQ ID NO: 6417, and a 3’ flanking region comprising SEQ ID NO: 6424. In some embodiments, the molecular scaffold comprises a 5’ flanking region comprises SEQ ID NO: 6414, a loop region comprising SEQ ID NO: 6418, and a 3’ flanking region comprising SEQ ID NO: 6423.
Exemplary AAV Capsid Sequences
[0297] In some embodiments, a viral genome, e.g., an AAV viral genome, AAV genome, or vector genome, described herein, comprises a promoter operably linked to a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRN A, thereby reducing or eliminating ATXN2 protein. In some embodiments, the viral genome further comprises an inverted terminal repeat region, an enhancer, an intron, a poly A region, or a combination thereof. In some embodiments, the AAV particle comprises a viral genome encoding a
capsid protein, e.g., a structural protein. In some embodiments, the capsid protein comprises a VP1 polypeptide, a VP2 polypeptide, and/or a VP3 polypeptide. In some embodiments, the VP1 polypeptide, the VP2 polypeptide, and/or the VP3 polypeptide are encoded by at least one Cap gene. In some embodiments, the AAV viral genome further comprises a nucleic acid encoding a Rep protein, e.g., a non-structural protein. In some embodiments, the Rep protein comprises a Rep78 protein, a Rep68 protein, a Rep52 protein, and/or a Rep40 protein. In some embodiments, the Rep78 protein, the Rep68 protein, the Rep52 protein, and/or the Rep40 protein are encoded by at least one Rep gene.
[0298] In some embodiments, the AAV particle comprises a viral genome encoding a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA, wherein the viral genome is packaged in an AAV capsid variant comprising an amino acid sequence selected from Table 3 or Table 4.
[8299] In some embodiments, the AAV particle comprising the viral genome encoding a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA, comprises an AAV capsid variant comprising: an amino acid sequence having the formula [N1]-[N2]-[N3], wherein: (i) [N I] comprises X1, X2, and X3, wherein at least one of X1, X2, or X3 is G; (ii) [N2] comprises the amino acid sequence of SPH; and (iii) [N3] comprises X4, X5, and X6, wherein at least one of X4, X5, or X6 is a. basic amino acid, e.g., a K or R; wherein [N1]-[N2]-[N3] is present in hypervariable loop IV ; and wherein the AAV capsid variant comprises an amino acid sequence at least 95% identical to the amino acid sequence oppositions 203-736 of SEQ ID NO: 138.
[0308] In some embodiments, the AAV capsid variant comprises the amino acid sequence of IIDSPHK (SEQ ID NO: 2), which is present in loop IV, e.g., between amino acids 449- 460 numbered according to SEQ ID NO: 982 (i.e., at a sequence position corresponding to that in SEQ ID NO: 982). In some embodiments, the AAV capsid vanant comprises: (i) a. VP 1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) a VP2 protein comprising or consisting of the amino acid sequence according to positions 138-742 of SEQ ID NO: 982 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto: and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence according to positions 203-742 of SEQ ID NO: 982 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
[0301] In some embodiments, the AAV capsid variant comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941), which is present in loop IV, e.g., between amino acids 449- 460 numbered according to SEQ ID NO: 36 (i.e., at a sequence position corresponding to that in SEQ ID NO: 36). In some embodiments, the AAV capsid variant comprises the amino acid E at position 451, the amino acid R at position 452, and the amino acid V at position 453, numbered according to SEQ ID NO: 36. In some embodiments, the AAV capsid variant composes: (i) the amino acid sequence of SPHSKA (SEQ II) NO: 941) present immediately subsequent to position 455 (e.g., at positions 456-461), numbered according to SEQ ID NO: 36: and (ii) the amino acid E at position 451, the amino acid R at position 452, and the amino acid V at position 453, numbered according to SEQ ID NO: 36. In some embodiments, the AAV capsid variant comprises the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589) in loop IV, e.g., between amino acids 449-460 numbered according to SEQ ID NO: 36. In some embodiments, the AAV capsid variant comprises: (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 36 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto: (ii) a VP2 protein comprising or consisting of the amino acid sequence according to positions 138-742 of SEQ ID NO: 36 or an amino acid sequence that is at least 95% identical (e.g,, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence according to positions 203-742 of SEQ ID NO: 36 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
[0302] In some embodiments, the AAV capsid variant comprises the amino acid sequence of SPSHKA (SEQ ID NO: 941), which is present in loop IV, e.g., between amino acids 449- 460 numbered according to SEQ ID NO: 4 (i.e,, at a sequence position corresponding to that in SEQ ID NO: 4). In some embodiments, the AAV capsid variant comprises the amino acid E at position 451, and the amino acid V at position 453, numbered according to SEQ ID NO: 4. In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SPHSKA (SEQ ID NO: 941 ) present immediately subsequent to position 455 (e.g., at positions 456-461), numbered according to SEQ ID NO: 4: and (ii) the amino acid E at position 451 and the amino acid V at position 453, numbered according to SEQ ID NO: 4. In some embodiments, the AAV capsid, variant comprises the amino acid, sequence of KTENVSGSPHSKAQNQQT (SEQ ID NO: 3272) in loop IV, e.g., between amino acids 449- 460 numbered according to SEQ ID NO: 4. In some embodiments, the AAV capsid variant
comprises: (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 4 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) a VP2 protein comprising or consisting of the amino acid sequence according to positions 138-742 of SEQ ID NO: 4 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto: and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence according to positions 203-742 of SEQ ID NO: 4 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
[0303] In some embodiments, the AAV particle comprises a viral genome comprising the nucleotide sequence comprising a modulatory polynucleotide of the disclosure. In some embodiments, the AAV capsid of the AAV particle comprises SEQ ID NO: 982.
[0304] In some embodiments, the AAV particle comprises a viral genome comprising the nucleotide sequence comprising a modulatory polynucleotide of the disclosure. In some embodiments, the AAV capsid of the AAV particle comprises SEQ ID NO: 36.
[0305] In some embodiments, the AAV particle comprises a viral genome comprising the nucleotide sequence comprising a modulatory polynucleotide of the disclosure. In some embodiments, the AAV capsid of the AAV particle comprises SEQ ID NO: 4.
[0306] In some embodiments, the AAV particle comprising the viral genome comprising the nucleotide sequence comprising a modulatory polynucleotide of the disclosure comprises an AA V capsid variant comprising an amino acid sequence selected, from Table 3 or Table 4. [0307] The present disclosure provides in some embodiments, vectors, cells, and/or AAV particles comprising the above-identified viral genomes and/or capsid variants.
Self-Complementary and Single Strand Vectors
[0308] In some embodiments, the AAV viral genome used in the present disclosure is single-stranded.
[0309] In some embodiments, the AAV viral genome is capable of forming double- stranded DNA. In some embodiments, the AAV viral genome is self-complementary. See, e.g., US Patent No. 7,465,583. scAAV particles contain both DNA strands that anneal together to form double stranded DNA. By skipping second strand synthesis, scAAVs allow for rapid expression in the cell.
[0310] Methods for producing and/or modifying AAV viral genome and. particles are disclosed in the art such as pseudotyped AAV particles (International Patent Publication Nos.
W0200028004; W0200123001; W02004112727; WO 2005005610 and WO 2005072364, the content of each of which are incorporated herein by reference in their entirety.
II. AAV Production
[0311] Viral production disclosed herein describes processes and methods for producing AAV particles (with enhanced, improved and/or increased tropism for a target tissue), e.g., an AAV particle comprising an AAV capsid variant that may be used to contact a target cell to deliver a payload.
[0312] In some embodiments, disclosed herein is a method of making an AAV particle of the present disclosure, e.g., an AAV particle comprising an .AAV capsid variant disclosed herein, wherein the method comprises: (i) providing a cell comprising a viral genome and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; e.g., an AAV capsid variant thereby making the AAV particle. In some embodiments, the method comprises, prior to step (i), introducing a nucleic acid comprising the viral genome into the cell. In some embodiments, the method comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. In some embodiments, the ceil comprises a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an SI9 cell), or a bacterial cell. In some embodiments, the AAV particle described herein is an isolated AAV particle. In some embodiments, the AAV particle described herein is a recombinant .AAV particle.
[8313] Any method known in the art may be used for the preparation of AAV particles. In some embodiments, AAV particles are produced, in mammalian cells (e.g., HEK293 cells). In some embodiments, AAV particles are produced in insect cells (e.g., Sf9 cells).
[0314] Methods of making AAV particles are well known in the art and are described in e.g., U.S. Patent Nos. US6204059, US5756283, US6258595, US6261551, US6270996, US6281010, US6365394, US6475769, US6482634, US6485966, US6943019, US6953690, US7022519, US7238526, US7291498 and US7491508, US5064764, US6194191, US6566118, US8137948; or International Publication Nos. WO1996039530, W01998010088, WO1999014354, WO1999015685, WO1999047691, W02000055342, W02000075353 and W02001023597; Methods In Molecular Biology, ed. Richard, Humana Press, NJ (1995); O'Reilly et al., Baculovirus Expression Vectors, A Laboratory Manual, Oxford Univ. Press (1994); Samulski et al., J. Fir.63:3822-8 (1989); Kajigaya et al., Proc. Nat'L Acad. Sci. USA 88: 4646-50 (1991): Ruffing et al., J. Vir. 66:6922-30 (1992);
Kimbauer et al., Vir., 219:37-44 (1996); Zhao et al., Vir.272: 382-93 (2000); the contents of each of which are herein incorporated by reference in their entirety. In some embodiments.
the AAV particles are made using the methods described in International Patent Publication WO2015191508, the contents of which are herein incorporated by reference in their entirety.
III. Pharmaceutical Compositions
[0315] The present disclosure additionally provides a method for treating an ATXN2- related disorder, e.g., in a. human subject, comprising administering to the subject any of the AAV polynucleotides or .AAV genomes described herein or administering to the subject a particle comprising said AAV polynucleotide or AAV genome, or administering to the subject any of the described compositions, including pharmaceutical compositions.
[0316] In some embodiments, a composition described herein comprises an AAV polynucleotide or AAV genome or AAV particle and at least one excipient.
[0317] Although pharmaceutical compositions provided herein, e.g., comprising A AV particles comprising a modulatory polynucleotide-encoding sequence, are principally directed to pharmaceutical compositions that are suitable for administration to humans, it will be understood by the skilled artisan that such compositions may be suitable for administration to anyr other animal, e.g., non-human mammals. Modification of pharmaceutical compositions suitable for administration to humans in order to render the compositions suitable for administration to various non-human animals is well understood, and the ordinarily skilled veterinary pharmacologist can design and/or perform such modification with merely ordinary-, if any, experimentation. Subjects to which administration of the pharmaceutical compositions is contemplated include, but are not limited to, humans and/or other primates; mammals, including commercially relevant mammals such as cattle, pigs, horses, sheep, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as poultry, chickens, ducks, geese, and/or turkeys.
[0318] In some embodiments, compositions are administered to humans, e.g., human patients or human subjects.
[0319] In some embodiments, the AAV particle formulations described herein may contain a nucleic acid encoding at least one modulatory polynucleotide for reducing or eliminating expression of ATXN2 (e.g., aberrantly expressed, aberrantly active, or mutant ATXN2). In some embodiments, the formulations may contain a nucleic acid encoding 1, 2, 3, 4, or 5 modulatory- polynucleotide(s) for reducing or eliminating expression of ATXN2 (e.g., aberrantly expressed, aberrantly active, or mutant ATXN2). In some embodiments, the modulatory polynucleotide is a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA, thereby reducing or eliminating ATXN2 protein.
[0320] A pharmaceutical composition in accordance with the present disclosure may be prepared, packaged, and/or sold in bulk, as a single unit dose, and/or as a plurality of single unit doses. As used herein, a “unit dose” refers to a discrete amount of the pharmaceutical composition comprising a predetermined amount of the active ingredient. The amount of the active ingredient is generally equal to the dosage of the active ingredient which would be administered to a subject and/or a convenient fraction of such a dosage such as, for example, one-half or one-third of such a dosage.
IV. Formulations
[0321] Formulations of the AAV pharmaceutical compositions described herein may be prepared, by any method known or hereafter developed in the art of pharmacology. In general, such preparatory- methods include the step of bringing the active ingredient into association with an excipient and/or one or more other accessory ingredients, and then, if necessary and/or desirable, dividing, shaping and/or packaging the product into a desired single- or multi-dose unit.
[0322] Relative amounts of the active ingredient, the pharmaceutically acceptable excipient, and/or any additional ingredients in a pharmaceutical composition in accordance with the disclosure will vary, depending upon the identity, size, and/or condition of the subject treated and. further depending upon the route by which the composition is to be administered.
[8323] For example, the composition may comprise about 0.1% to about 99% (w/w) of the active ingredient. By way of example, the composition may comprise about 0,1% to about.
100%, e.g., about 0.5% to about 50%, about 1% to about 30%, about 5% to about 80%, or at least 80% (w/w) active ingredient.
[0324] The AAV particles of the disclosure can be formulated using one or more excipients to: (1) increase stability; (2) increase cell transfection or transduction; (3) permit the sustained or delayed release; (4) alter the biodistribution (e.g., target the viral particle to specific tissues or cell types); (5) increase the translation of encoded protein in vivo; (6) alter the release profile of encoded protein in vivo and/or (7) allow' for regulatable expression of the modulatory- polynucleotide.
[0325] Formulations of the present disclosure can include, without limitation, saline, lipidoids, liposomes, lipid nanoparticles, polymers, lipoplexes, core-shell nanoparticles, peptides, proteins, cells transfected with viral vectors (e.g,, for transplantation into a subject), nanoparticle mimics and combinations thereof. Further, the viral vectors of the present disclosure may be formulated using self-assembled nucleic acid nanoparticles.
[0326] In some embodiments, the viral vectors encoding a modulatory polynucleotide may be formulated to optimize baricity and/or osmolality. In some embodiments, the baricity and/or osmolality of the formulation may be optimized to ensure optimal drug distribution in the central nervous system or a region or component of the central nervous system.
Excipients
[0327] The formulations of the disclosure can include one or more excipients, each in an amount that together increases the stability of the AAV particle, increases cell transfection or transduction by the viral particle, increases the expression of viral particle encoded protein, and/or alters the release profile of AAV particle encoded proteins. In some embodiments, a pharmaceutically acceptable excipient may be at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% pure. In some embodiments, an excipient is approved for use for humans and for veterinary use. In some embodiments, an excipient may be approved by United States Food and Drug Administration. In some embodiments, an excipient may be of pharmaceutical grade. In some embodiments, an excipient may meet the standards of the United States Pharmacopoeia (USP), the European Pharmacopoeia (EP), the British Pharmacopoeia, and/or the International Pharmacopoeia.
[0328] Excipients, which, as used herein, include, but are not limited to, any and all solvents, dispersion media, diluents, or other liquid, vehicles, dispersion or suspension aids, surface active agents, isotonic agents, thickening or emulsifying agents, preservatives, and the like, as suited to the particular dosage form desired. Various excipients for formulating pharmaceutical compositions and techniques for preparing the composition are known in the art (see Remington; The Science and Practice of pharma.cy, 21st Edition, A. R. Gennaro, Lippincott, Williams & Wilkins, Baltimore, MD, 2006; the contents of which are herein incorporated by reference in their entirety). The use of a conventional excipient medium may be contemplated within the scope of the present disclosure, except insofar as any conventional excipient medium may be incompatible with a substance or its derivatives, such as by producing any undesirable biological effect or otherwise interacting in a deleterious manner with any other component(s) of the phannaceutical composition.
Inactive Ingredients
[0329] In some embodiments, AAV formulations may comprise at least one excipient which is an inactive ingredient. As used herein, the term “inactive ingredient” refers to one or more agents that do not contribute to the activity of the pharmaceutical composition included, in formulations. In some embodiments, ail, none, or some of the inactive ingredients which may be used in the formulations of the present disclosure may be approved by the US Food
and Drag Administration (FDA).
[0330] Formulations of AAV particles may include cations or anions. In some embodiments, the formulations include metal cations such as, but not limited, to, Zn2+, Ca2+, Cu2+, Mg+, or combinations thereof. In one embodiment, formulations may include polymers or polynucleotides complexed with a metal cation (see, e.g., U.S. Pat. Nos. 6,265,389 and 6,555,525, the contents of each of which are herein incorporated by reference m their entirety).
V. Uses and Applications
[0331] The compositions of the disclosure may be administered to a subject or used in the manufacture of a medicament for administration to a subject having aberrant ATXN2 protein expression or activity or mutant ATXN2 protein. As used herein, “associated with aberrant ATXN2 levels” or “associated with increased expression” means that one or more symptoms of a disease are caused by aberrant ATXN2 protein expression or activity or mutant ATXN2 protein in a. target tissue or in a biofluid such as blood. A disease or condition associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein may be a disorder of the central nervous system, and/or a neurological and/or neuromuscular disorder. Also specifically contemplated herein is spinocerebellar ataxia type 2 (SCA2) and related disorders, e.g., related disorders arising from expression of ATXN2 gene product with trinucleotide repeat expansions, e.g., ATXN2 with 32 or more CAG repeats (SEQ ID NO: 6435) (polyQ expansions).
[0332] The present disclosure addresses the need for new technologies by providing ATXN2-related treatment deliverable by AAV-based compositions and complexes for the treatment of ATXN2-related disorders.
[0333] In some embodiments, the disclosure provides a method for treating SCA2 with at least one mutation in a AT.XN2 gene. In certain embodiments, the AAV particles including modulatory polynucleotides for reducing or eliminating expression of ATXN2 may be administered to a subject to treat SCA2. In some embodiments, administration of the AAV particles comprising viral genomes that encode a modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA may protect central nervous system cells and/or tissues from cell death.
[0334] In some embodiments, the delivery of the AAV particles may halt or slow progression of ATXN2-related disorders. In certain embodiments, the delivery of the AAV particles improves symptoms of ATXN2-related disorders, including, for example, cognitive, muscular, physical, and sensory symptoms of ATXN2-related disorders.
[0335] In some embodiments, the present disclosure encompasses the delivery- of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may- improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body.
[0336] In certain embodiments, the pharmaceutical compositions described herein are used as research tools, particularly in in vitro investigations using human cell lines such as HEK293T and. in vivo testing in nonhuman primates which will occur prior to human clinical trials.
CNS diseases
[0337] The present disclosure provides a method for treating a disease, disorder and/or condition in a mammalian subject, including a human subject, comprising administering to the subject any of the viral particles e.g., AAV, AAV particle, or AAV viral genome that expresses a modulatory polynucleotide described herein or administering to the subject a particle comprising said AA V particle or AAV genome, or administering to the subject any of the described compositions, including pharmaceutical compositions.
[0338] In some embodiments, AAV particles of the present disclosure, through delivery of a functional payload that is a therapeutic product comprising a modulatory polynucleotide that can modulate the level or function of a gene product in the CNS.
[0339] A functional payload may alleviate or reduce symptoms that result from abnormal level and/or function of a gene product (e.g., an absence or defect in a protein) in a subject in need thereof or that otherwise confers a benefit to a CNS disorder in a subject in need thereof. [0340] As non-limiting examples, companion or combination therapeutic products delivered by AAV particles of the present disclosure may include, but are not limited to, growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2-related disorders.
[0341] In some embodiments, AAV particles of the present disclosure may be used to treat diseases that are associated with impairments of the growth and development of the CNS, e.g., neurodevelopmental disorders. In some embodiments, such neurodevelopmental disorders may be caused by genetic mutations.
[0342] In some embodiments, the neurological disorders may be functional neurological disorders with motor and/or sensory symptoms which have neurological origin in the CNS. As non-limiting examples, functional neurological and/or neuromuscular disorders may be chronic pain, seizures, speech problems, involuntary movements, sleep disturbances.
myotonia, muscle weakness, cardiac conduction abnormalities, learning difficulties, or hypotonia.
[0343] In some embodiments, the neurological or neuromuscular disease, disorder, and/or condition is ATXN2-related disorders. In some embodiments, the delivery of the AAV particles may halt or slow the disease progression of ATXN2-related disorders by 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or more than 95% using a known analysis method and comparator group for AT.XN2-related disorders. As a non-limiting example, the delivery of the AAV particles may halt or slow progression of ATXN2-related disorders as measured by cholesterol accumulation in CNS cells (as determined, for example, by filipin staining and quantification).
[0344] In some embodiments, delivery of an AAV particle described herein increases the amount of ATXN2 protein in a tissue by 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11 %, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99%, or more than 100%, In some embodiments, the AAV particle encoding a payload may decrease the amount of ATXN2 protein in a tissue effective to reduce one or more symptoms of a disease associated with aberrant ATXN2 protein expression or activity or mutated ATXN2 protein, optionally caused by trinucleotide repeat (and. thus poiyQ) expansion.
[0345] In some embodiments, the AAV particles and AAV viral genomes described herein, upon administration to subject or introduction to a target cell, decrease ATXN2 activity 2-3 fold over baseline ATXN2 activity . In the case of subjects or target cells with aberrant ATXN2 activity, as in the case of subjects having a ATXN2-related disorder or cells or tissues harboring at least one mutation in a ATXN2 gene, the AAV particles and .AAV viral genomes described herein delivery modulatory polynucleotide to inhibit activity of aberrant ATXN2 and restore ATXN2 activity to normal levels, as defined by ATXN2 activity levels in subjects, tissues, and cells not afflicted with a ATXN2-related disorder or not harboring a ATXN2 gene mutation.
Therapeutic applications
[0346] The present disclosure additionally provides methods for treating non-infectious diseases and/or disorders in a mammalian subject, including a human subject, comprising administering to the subject any of the AAV particles or pharmaceutical compositions described. herein. In some embodiments, non-infectious diseases and/or disorders treated according to the methods described herein include but are not limited to spinocerebellar ataxia type 2 (SCA2).
[0347] The present disclosure provides a method for administering to a subject in need thereof, including a human subject, a therapeutically effective amount of the AAV particles of the disclosure to slow, stop or reverse disease progression. As a. non-limiting example, disease progression may be measured by tests or diagnostic tool(s) known to those skilled in the art. As another non-limiting example, disease progression may be measured by change in the pathological features of the brain, CSF, or other tissues or fluids of the subject.
VI. Delivery of AAV Particles
Delivery' to Cells
[0348] In some aspects, the present disclosure provides a me thod of delivering to a ceil or tissue any of the above-described AAV particles, comprising contacting the cell or tissue with said AAV particle or contacting the cell or tissue with a formulation comprising said AAV particle, or contacting the cell or tissue with any of the described compositions, including pharmaceutical compositions. The method of delivering the AAV particle to a cell or tissue can be accomplished in vitro, ex vivo, or in vivo.
Delivery- to Subjects
[0349] In some aspects, the present disclosure additionally provides a method of delivering to a subject, including a mammalian subject, any of the above-described AAV particles comprising administering to the subject said. AAV particle, or administering to the subject a formulation comprising said AAV particle, or administering to the subject any of the described compositions, including pharmaceutical compositions.
[0350] In some embodiments, the AAV particles may be delivered to bypass anatomical blockages (e.g., the blood brain barrier).
[0351] In some embodiments, the .AAV particles may be formulated and delivered to a subject by a route which increases the speed of drug effect as compared to oral delivery.
[0352] In some embodiments, the AAV particles may be delivered using intrathecal infusion.
[0353] In some embodiments, a subject may be administered the AAV particles described herein using a bolus infusion.
[0354] In some embodiments, the AAV particles may be delivered in a continuous and/or bolus infusion. Each site of delivery may use a different dosing regimen, or the same dosing regimen may be used for each site of delivery. As a non-limiting example, the sites of delivery may be in the cervical and the lumbar region. As another non-limiting example, the sites of delivery- may be in the cervical region. As another non-limiting example, the sites of delivery may be in the lumbar region.
[0355] In some embodiments, the AAV particles may be delivered to a subject via a single route of administration.
[0356] In some embodiments, the AAV particles may be delivered to a. subject via. a. multi- site route of administration. For example, a. subject may be administered the AAV particles at 2, 3, 4, 5, or more than 5 sites.
[0357] In some embodiments, a subject may be administered the AAV particles described herein using sustained delivery over a period of minutes, hours, or days. The infusion rate may be changed depending on the subject, distribution, formulation, or another delivery parameter known to those in the art.
[0358] In some embodiments, if continuous delivery (continuous infusion) of the AAV particles is used, the continuous infusion may be for 1 hour, 2, hours, 3 hours, 4 hours, 5 hours, 6 hours, 7 hours, 8 hours, 9 hours, 10 hours, 11 hours, 12 hours, 13 hours, 14 hours, 15 hours, 16 hours, 17 hours, 18 hours, 19 hours, 20 hours, 21 hours, 22 hours, 23 hours, 24 hours, or more than 24 hours.
[0359] In some embodiments, the intracranial pressure may be evaluated prior to administration. The route, volume, AAV particle concentration, infusion duration and/or vector titer may be optimized based on the intracranial pressure of a subject.
[0360] In some embodiments, the AAV particles may be delivered by systemic delivery.
In some embodiments, the systemic delivery may be by intravascular administration. In some embodiments, the systemic delivery may be by intravenous administration.
[0361] In some embodiments, the AA V particles may be delivered by injection into the CSF pathway. Non-limiting examples of delivery to the CSF pathway include intrathecal and intracerebroventricular administration.
[0362] In some embodiments, an AAV particle described herein is administered intravenously.
[0363] In some embodiments, the AAV particles may be delivered by direct (intraparenchymal) injection into the substance of an organ, e.g,, one or more regions of the brain.
[0364] In some embodiments, the AAV particles may be delivered by subpial injection into the spinal cord. For example, subjects may be placed into a spinal immobilization apparatus, A dorsal laminectomy may be performed to expose the spinal cord. Guiding tubes and XYZ manipulators may be used to assist catheter placement. Subpial catheters may be placed into the subpial space by advancing the catheter from the guiding tube and AAV particles may be injected through the catheter (Miyanohara et al., Mol Ther Methods Clin
Dev. 2016; 3: 16046). In some cases, the AAV particles may be injected into the cervical subpial space. In some cases, the AAV particles may be injected into the thoracic subpial space.
[0365] In some embodiments, the AAV particies may be delivered by direct injection to the CNS of a subject. In some embodiments, direct injection is intracerebral injection, intraparenchyrnal injection, intrathecal injection, mtra-cisterna magna injection, or any combination thereof. In some embodiments, direct injection to the CNS of a subject comprises convection enhanced delivery (CED). In some embodiments, administration comprises peripheral injection. In some embodiments, peripheral injection is intravenous injection.
[0366] In some embodiments, the AAV particles may be delivered to a. subject in order to decrease mutated ATXN2 mRNA levels in the caudate, putamen, thalamus, superior colliculus, cortex, and/or corpus callosum by transducing cells in these CNS regions.
Transduction may also be referred, to as the amount of cells that are positive for the modulatory polynucleotide. The transduction may be greater than or equal to 1%, greater than or equal to 5%, greater than or equal to 10%, greater than or equal to 15%, greater than or equal to 20%, greater than or equal to 25%, greater than or equal to 30%, greater than or equal to 35%, greater than or equal to 40%, greater than or equal to 45%, greater than or equal to 50%, greater than or equal to 55%, greater than or equal to 60%, greater than or equal to 65%, greater than or equal to 70%, greater than or equal to 75%, greater than or equal to 80%, greater than or equal to 85%, greater than or equal to 90%, greater than or equal to 95%, or greater than or equal to 99% of cells in these CNS regions.
[0367] In some embodiments, delivery of -AAV particles comprising a viral genome encoding a modulatory polynucleotide described herein to neurons in the caudate-putamen, thalamus, superior colliculus, cortex, and/or corpus callosum will lead to an increased expression of a. modulatory polynucleotide and/or decreased expression of mutated ATXN2 mRNA. lire increased expression of a modulatory polynucleotide and/or decreased expression of mutated ATXN2 mRNA may lead to improved survival and/or function of various cell types in these CNS regions and/or improvement of ATXN2-related disorder.
[0368] In some embodiments, the .AAV particles may be delivered to a subject in order to establish widespread distribution of the modulatory polynucleotide throughout the CNS, e.g., by administering the AAV particles to the thalamus of the subject.
[0369] In some embodiments, the increased expression of modulatory polynucleotide protein may lead to improved gait, sensory capability, coordination of movement and
strength, functional capacity, cognition, and/or quality of life.
Administration
[0370] In some embodiments, the present disclosure provides methods comprising administering a viral vector comprising a payload construct in accordance with the disclosure to a subject in need thereof. Viral vector pharmaceutical, diagnostic, or prophylactic compositions thereof, may be administered to a subject using any amount and any route of administration effective for treating, or diagnosing a disease, disorder, and/or condition associated with aberrant ATXN2 protein expression or activity or mutant ATXN2 protein). In some embodiments, the disease, disorder, and/or condition is ATXN2-related disorder (e.g., SCA2).
[0371] Compositions in accordance with the disclosure may be formulated in unit dosage form for ease of administration and uniformity of dosage. It will be understood, however, that the total daily usage of the compositions of the present disclosure may be decided by the attending physician within the scope of sound medical judgment. The specific therapeutically effective, prophylactically effective, or appropriate imaging dose level for any particular patient will depend upon a variety of factors including the disorder being treated and the seventy of the disorder; the activity of the specific compound employed: the specific composition employed; the age, body weight, general health , sex, and diet of the patient; the time of administration, route of administration, and rate of excretion of the specific protein employed; the duration of the treatment; drugs used in combination or coincidental with the specific compound employed; and like factors well known in the medical arts,
[0372] In certain embodiments, the desired dosage may be delivered using multiple administrations (e.g., two, three, four, five, six, seven, eight, nine, ten, eleven, twelve, thirteen, fourteen, or more administrations). When multiple administrations are employed, split dosing regimens such as those described herein may be used. As used herein, a “split dose” is the division of single unit dose or total daily dose into two or more doses, e.g., two or more administrations of the single unit dose. As used herein, a “single unit dose” is a dose of any therapeutic composition administered in one dose/at one time/single route/smgle point of contact, e.g., single administration event. In some embodiments, a. single unit dose is provided as a discrete dosage form (e.g., a tablet, capsule, patch, loaded syringe, vial, etc.). As used herein, a “total daily dose” is an amount given or prescribed in 2.4-hour period. It may be administered as a single unit dose. The viral particles may be formulated in buffer only or in a formulation described herein.
[0373] In some embodiments, a pharmaceutical composition described herein can be formulated into a topical, intranasal, pulmonary, intratracheal, or injectable dosage form. In some embodiments, a. pharmaceutical composition described herein can be formulated in a dosage form suitable for intravenous, intraocular, intravitreal, intramuscular, intracardiac, intraperitoneal, and/or subcutaneous administration.
[0374] In some embodiments, delivery of the AAV particles described herein results in minimal serious adverse events (SAEs) as a result of the delivery of the AAV particles. Combinations
[0375] The AAV particles may be used in combination with one or more other therapeutic, prophylactic, diagnostic, or imaging agents, lire phrase “in combination with,” is not intended to require that the agents must be administered at the same time and/or formulated for delivery together, although these methods of delivery are within the scope of the present disclosure. Compositions can be administered concurrently with, prior to, or subsequent to, one or more other desired therapeutics or medical procedures. In general, each agent will be administered at a dose and/or on a time schedule determined forthat agent. In some embodiments, the present disclosure encompasses the delivery-- of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body.
[0376] The therapeutic agents may be approved by the US Food and Drug Administration or may be in clinical trial or at the preclinical research stage. The therapeutic agents may utilize any therapeutic modality known in the art, with non-limiting examples including gene silencing or interference (i.e.., miRNA, siRNA, RNAi, shRNA), gene editing (i.e.., TALEN, CRISPR/Cas9 systems, zinc finger nucleases), and gene, protein or enzyme replacement.
[0377] In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the SCA2-related disorder. In some embodiments, the at least one additional therapeutic agent and/or therapy for treating the SCA2-related disorder comprises enzyme replacement therapy- (E.RT) (e.g., imiglucerase, velaglucerase alfa, or taliglucera.se alfa); substrate reduction therapy (SRT) (e.g., eliglustat or miglustat), levodopa, carbidopa, Safinamide, a dopamine agonist (e.g., pramipexole, rotigotme, or ropinirole), a dopamine antagonist (e.g., quetiapine, clozapine), an anticholinergic (e.g., benztropine or trihexyphenidyl), a cholinesterase inhibitor (e.g., rivastigmine, donepezil, or galantamine), an N -methyl -d-aspartate (NMD A) receptor antagonist (e.g., memantine), or a combination thereof.
[0378] In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an immunosuppressant. In some embodiments, the immunosuppressant may be administered to the subject prior to administration of an A AV particle or pharmaceutical composition described herein. In some embodiments, the immunosuppressant may be administered to the subject simultaneously with administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the immunosuppressant may be administered to the subject after administration of an A AV particle or pharmaceutical composition described herein. In some embodiments, the AAV particle or pharmaceutical composition is administered to a subject who is receiving or has received an immunosuppressant. In some embodiments, the immunosuppressant comprises a corticosteroid (for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. In some embodiments, the corticosteroid comprises prednisone, prednisolone, methylprednisolone, and/or dexamethasone.
Measurement of Expression
[0379] Expression of modulatory polynucleotide from viral genomes may be determined using various methods known in the art such as, but not limited to immunochemistry (e. g. , IHC), enzyme-linked immunosorbent assay (ELISA), affinity ELISA, ELISPOT, flow cytometry, immunocytology, surface plasmon resonance analysis, kinetic exclusion assay, liquid chromatography-mass spectrometry (LCMS), high-performance liquid chromatography (HPLC), BCA assay, immunoelectrophoresis. Western blot, SDS-PAGE, protein immunoprecipitation, PCR, and/or in situ hybridization (ISH). In some embodiments, modulatory polynucleotide delivered in different AAV capsids may have different expression levels in different CNS tissues.
[0380] Methods of detecting modulatory polynucleotide expression are known.
VII. Kits and Devices
Kits
[0381] In some aspects, the present disclosure provides a variety of kits for conveni ently and/or effectively carrying out methods of the present disclosure. Typically, kits will comprise sufficient amounts and/or numbers of components to allow' a user to perform multiple treatments of a subjects) and/or to perform multiple experiments.
[0382] Any of the vectors, constructs, or modulatory polynucleotides of the present disclosure may be comprised in a kit. In some embodiments, kits may further include reagents and/or instructions for creating and/or synthesizing compounds and/or compositions
of the present disclosure. In some embodiments, kits may also include one or more buffers. In some embodiments, kits of the disclosure may include components for making protein or nucleic acid arrays or libraries and thus, may include, for example, solid supports, [0383] In some embodiments, kit components may be packaged either in aqueous media or in lyophilized form. The container means of the kits will generally include at least one vial, test tube, flask, bottle, syringe or other container means, into which a component may be placed, and suitably aliquoted, Where there is more than one kit component, (labeling reagent and label may be packaged together), kits may also generally contain second, third or other additional containers into which additional components may be separately placed. In some embodiments, kits may also comprise second container means for containing sterile, pharmaceutically acceptable buffers and/or other diluents. In some embodiments, various combinations of components may be comprised in one or more vial. Kits of the present disclosure may also typically include means for containing compounds and/or compositions of the present disclosure, e.g. , proteins, nucleic acids, and any other reagent containers in close confinement for commercial sale. Such containers may include injection or blow- molded plastic containers into which desired vials are retained.
[0384] In some embodiments, kit components are provided in one and/or more liquid solutions. In some embodiments, liquid solutions are aqueous solutions, with sterile aqueous solutions being particularly used. In some embodiments, kit components may be provided as dried powder(s). When reagents and/or components are provided as dry powders, such powders may be reconstituted by the addition of suitable volumes of solvent. In some embodiments, it is envisioned that solvents may also be provided in another container means. In some embodiments, labeling dyes are provided as dried powders. In some embodiments, it is contemplated that 10, 20, 30, 40. 50, 60, 70, 80, 90. 100, 120, 120, 130, 140, 150, 160. 170, 180, 190, 200, 300, 400, 500, 600, 700, 800, 900, 1000 micrograms or at least or at most those amounts of dried dye are provided in kits of the disclosure. In such embodiments, dye may then be resuspended in any suitable solvent, such as DMSO.
[0385] In some embodiments, kits may include instructions for employing kit components as well the use of any other reagen t not included in the kit. Instructions may include variations that may be implemented.
Devices
[0386] In some embodiments, compounds and/or compositions of the present disclosure may be combined with, coated onto or embedded in a device. Devices may include, but are not limited to, dental implants, stents, bone replacements, artificial joints, valves, pacemakers
and/or other implantable therapeutic device.
[0387] The present disclosure provides for devices which may incorporate viral vectors that encode one or more modulatory polynucleotide molecules. These devices contain in a stable formulation the viral vectors which may be immediately delivered to a subject in need thereof, such as a human patient.
[0388] Devices for administration may be employed to deliver the viral vectors encoding modulatory- polynucleotide of the present disclosure according to single, multi- or split- dosing regimens taught herein.
[0389] Method and devices known in the art for multi-administration to cells, organs and tissues are contemplated for use in conjunction with the methods and compositions disclosed herein as embodiments of the present disclosure.
VIII. Definitions
[0390] At various places in the present specification, substituents of compounds of the present disclosure are disclosed in groups or in ranges. It is specifically intended that the present disclosure include each and every individual sub-combination of the members of such groups and ranges. The following is a non-limiting list of term definitions.
[0391] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the present invention pertains.
[0392] The articles "a,” “an,” and “the” may mean one or more than one unless indicated to the contrary-' or otherwise evident from the context. Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. The disclosure includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process. The disclosure includes embodiments in which more than one, or the entire group members are present in, employed in, or otherwise relevant to a given product or process.
[0393] The term “comprising” is intended to be open and pennits but does not require the inclusion of additional elements or steps.
[0394] Where ranges are given, endpoints are included. Furthermore, it is to be understood that unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of
the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise.
[0395] Adeno-associated virus: As used herein, the term “adeno-associated virus” or “AAV” refers to members of the dependovirus genus or a functional variant thereof. Unless stated otherwise, “AAV” may refer to wildtype (i.e., naturally occurring) .AAV or recombinant AAV.
[0396] AAV Particle: As used herein, an “AAV particle” refers to a particle comprising an AAV capsid, e.g., an AAV capsid variant (such as a parent capsid sequence with at least one peptide insert), and a polynucleotide, e.g., a viral genome or a vector genome. The AAV particle may be capable of delivering a polynucleotide to cells. The cells may be mammalian cells, e.g., human cells. In some embodiments, an AAV particle of the present disclosure may be produced recombinandy. In some embodiments, an AAV particle may be derived from any serotype, described herein or known in the art, including combinations of serotypes (e.g., “pseudotyped” AAV) or from various genomes (e.g., single stranded or self- complementary). In some embodiments, the AAV particle may be replication defective and/or targeted. In some embodiments, the AAV particle may comprise a peptide present in, e.g., inserted into, the capsid to enhance tropism for a desired target tissue. It is to be understood that reference to the A AV particle of the disclosure also includes pharmaceutical compositions thereof, even if not explicitly recited.
[0397] Administering: As used herein, the term “administering” refers to providing a pharmaceutical agent or composition to a subject.
[0398] Amelioration: As used herein, the term “amelioration” or “ameliorating” refers to a lessening of severity of at least one indicator of a condition or disease. For example, in the context of a neurodegenerative disorder, amelioration includes the reduction or stabilization of neuron loss.
[0399] Antisense strand: As used herein, the term “the antisense strand” or “the first strand” or “the guide strand” of an siRN A molecule refers to a polynucleotide strand that is substantially complementary to a section of about 10-50 nucleotides, e.g., about 15-30, 16-25, 18-23, or 19-22 nucleotides, of the mRNA of a. gene targeted for silencing, e.g., by the RNAi machinery or process.
[0400] Approximately: As used herein, the term “approximately” or “about,” as applied to one or more values of interest, refers to a value that is similar to, i.e., within 10% of, a stated reference value.
[0401] Baseline: The term “baseline,” when used to describe a. measurement in a subject receiving or about to receive a treatment, refers to a measurement made before starting the treatment.
[0402] ATXN2: As used herein, and unless indicated otherwise by context, the terms “ATXN2” and “ATXN2 protein” are used interchangeably to refer to a protein product or portion thereof of the ATXN2 gene (Ensembl Gene ID No. ENSG00000204842).
[0403] Capsid: As used herein, the term “capsid” refers to the exterior, e.g,, a. protein shell, of a virus particle, e.g., an AAV particle, that is substantially (e.g., >50%, >60%, >70%, >80%, >90%, >95%, >99%, or 100%) protein. In some embodiments, the capsid is an AAV capsid comprising an AAV capsid protein described herein, e.g., a VP1, VP2, and/or VP3 polypeptide. The AAV capsid protein can be a wild-type AAV capsid protein or a variant, e.g., a structural and/or functional variant from a wild-type or a reference capsid protein, referred to herein as an “AAV capsid variant.” For example, and without limitation, an AAV capsid variant may refer to at least a VP1 protein, a VP2 protein, or a VP3 protein (e.g., all of the VP1 , VP2, and VP3 proteins forming the AAV capsid) as will be clear from context. In some embodiments, the AAV capsid variant described herein may comprise a peptide insertion. In some embodiments, the AAV capsid variant described herein has the ability to encapsulate a viral genome and/or is capable of entry into a cell, e.g., a mammalian cell. In some embodiments, the AAV capsid variant described herein may have modified tropism compared to that of a wild-type AAV capsid, e.g., the corresponding wild-type capsid.
[0404] Cis-Elements: As used herein, cis-elements or the synonymous term “cis- regulatory elements” refer to regions of non-coding DNA which regulate the transcription of nearby genes, lire Latin prefix “cis” translates to “on this side.” Cis-elements are found in tire vicinity of the gene, or genes, they regulate. Examples of cis-elements include a Kozak sequence, SV40 introns, or a portion of the backbone.
[0405] CNS structures: As used herein, “CNS structures” refers to structures of the central nervous system and sub-structures thereof. Non-limiting examples of structures in the spinal cord may include, ventral horn, dorsal horn, white matter, and nervous system pathways or nuclei within. Non-limiting examples of structures in the brain include, forebrain, midbrain, hindbrain, diencephalon, telencephalon, myelencephalon, metencephalon, mesencephalon, prosencephalon, rhombencephalon, cortices, frontal lobe, parietal lobe, temporal lobe, occipital lobe, cerebrum, thalamus, hypothalamus, tectum, tegmentum, cerebellum, pons, medulla, amygdala, hippocampus, basal ganglia, corpus callosum, pituitary- gland, putamen,
striatum, ventricles and sub-structures thereof.
[0406] CNS Cells: As used herein, “CNS cells” refers to cells of the central nervous system and sub-structures thereof. Non-limiting examples of CNS cells include, neurons and. sub-types thereof, glia, microglia, oligodendrocytes, ependymal cells and astrocytes. Non- limiting examples of neurons include sensory neurons, motor neurons, interneurons, unipolar cells, bipolar cells, multipolar cells, pseudounipolar cells, pyramidal cells, basket cells, stellate cells, Purkinje cells, Betz cells, amacrine cells, granule cell, ovoid cell, medium aspmy neurons, large aspiny neurons, GABAergic neurons and/or glutamatergic neurons.
[0407] Codon optimization; codon optimization” refers to a process of changing codons of a given gene in such a manner that the polypeptide sequence encoded by the gene remains the same.
[0408] Complementary and substantially complementary: As used herein, the term “complementary” refers to the ability of polynucleotides to form base pairs with one another. Perfect complementarity or 100% complementarity refers to the situation in which each nucleotide unit of one polynucleotide strand can form a hydrogen bond with a. nucleotide unit of a second polynucleotide strand. Less than perfect complementarity refers to the situation in which some, but not all, nucleotide units of two strands can form hydrogen bond with each other. For example, for two 20-mers, if only two base pairs on each strand can form a hydrogen bond with each other, the polynucleotide strands exhibit 10% complementarity. In the same example, if 18 base pairs on each strand can form hydrogen bonds with. each other, the polynucleotide strands exhibit 90% (e.g., 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or 100% complementarity. The term “complementary” as used herein can encompass fully complementary or partially (e.g., substantially) complementary. “Fully complementary”, “perfect complementarity”, or “100% complementarity” refers to the situation m which each nucleotide unit of one polynucleotide or oligonucleotide strand can base-pair with a nucleotide unit of a second polynucleotide or oligonucleotide strand. As used herein, the term “substantially complementary” means that >50% of the nucleotide units of a first polynucleotide strand can base pair with nucleotide units on a second polynucleotide strand. When used, in the context of RNA silencing, “substantially complementary” refers to an siRNA that has a sequence (e.g., in the antisense strand) that is sufficient to bind the desired target mRNA and to trigger the RNA silencing of the target mRNA. “Fully complementary”, “perfect complementarity”, or “100% complementarity” refers to the situation in which each nucleotide unit of one polynucleotide or oligonucleotide strand can base-pair with a nucleotide unit of a second polynucleotide or oligonucleotide strand.
[0409] Conservative substitution: As used herein, a conservative substitution, as applied to an amino acid sequence, also referred to as a “conservative amino acid substitution," is one in which the amino acid residue is replaced with an amino acid residue having similar biochemical properties. When used in reference to a nucleic acid sequence, the term “conservative substitution" refers to a nucleotide replacement that results in an amino acid residue having similar biochemical properties compared to a reference sequence. Families of amino acid residues having similar biochemical properties have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine).
[0410] Corresponding to: As used herein, the phrase “corresponding to” in the context of an amino acid sequence refers to the location of an amino acid in a reference sequence or the equivalent position in a modified sequence when aligned. For example, an arnino acid corresponding to position 5 of SEQ ID NO: 36 refers to the amino acid at the fifth position from the N-terminus in SEQ ID NO: 36 or the equivalent position in an aligned sequence. As used herein, an amino acid at a position corresponding to that in a designated sequence may also be referred to as an amino acid at a particular position, numbered according to the designated, sequence. For instance, an amino acid corresponding to position 5 of SEQ ID NO: 36 may also be referred to as an amino acid at position 5, numbered according to SEQ ID NO: 36, relative to a reference sequence of SEQ ID: 36, or as numbered according to a sequence corresponding to SEQ ID NO: 36.
[0411] Derivative: As used herein, the term “derivative” refers to a composition (e.g., sequence, compound, formulation, etc.) that is derived from, or finds its basis in, a parent composition. Non-limiting examples of a parent composition include a wild-type or original amino acid or nucleic acid sequence, or an undiluted formulation. In some embodiments, a derivative is a variant of a parent composition. A derivative may differ from the parent composition by less than about 1%, less than about 5%, less than about 10%, less than about 15%, less than about 20%, less than about 25%, less than about 30%, less than about 35%, less than about 40%, less than about 45%, or less than about 50%. In certain embodiments, a derivative may differ from a. parent composition by more than about 50%. In certain embodiments, a derivative may differ from a parent composition by more than about 75%. In
some embodiments, a derivative may be a fragment or truncation of a parent amino acid or nucleotide sequence. As a non-limiting example, a derivative may be a sequence with a nucleotide, amino acid, or peptide substitution and/or insertion as compared to a parent nucleic acid or amino acid sequence (e.g., as compared to AAV9).
[0412] Effective amount: As used herein, the term "‘effective amount” or “therapeutically effective amount” of an agent is that amount sufficient to effect beneficial or desired results. An effective amount is provided in a single dose or multiple doses to treat, improve symptoms of, delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition.
[0413] Excipient: As used herein, the term “excipient” refers to an inactive substance that serves as the vehicle or medium for an active pharmaceutical agent or other active substance.
[0414] Formulation: As used herein, a “formulation” includes at least one active ingredient (e.g., an AAV particle) and at least one inactive ingredient (e.g., a pharmaceuti cally acceptabl e excipient) .
[0415] Fragment: A “fragment,” as used herein, refers to a. contiguous portion of a reference sequence. A fragment may comprise a functional fragment that retains at least one activity of the reference sequence. For example, fragments of proteins may comprise polypeptides obtained by digesting full-length protein isolated from cultured, cells, A fragment may also refer to a truncation (e.g., an N-terminal and/or C -terminal truncation) of a protein or a truncation (e.g., at tire 5’ and/or 3" end) of a nucleic acid. A protein fragment may be obtained by expression of a truncated nucleic acid, such that the nucleic acid encodes a portion of the full-length protein.
[0416] Humanized: As used herein, the term “humanized” refers to a non-human sequence of a polynucleotide or a polypeptide which has been altered to increase its similarity to a corresponding human sequence.
[0417] Identity: As used herein, the term “identity” refers to the overall relatedness between polymeric molecules, e.g., between oligonucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of the percent identity of two polynucleotide sequences, for example, may be performed by aligning the two sequences for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second nucleic acid sequences for optimal alignment and non-identical sequences can be disregarded, for comparison purposes). The nucleotides at corresponding nucleotide positions are then compared. When a position in the first sequence is occupied by the same nucleotide as the corresponding position in the second sequence, then the molecules are
identical at that position. The percent identity between the two sequences is a function of the number of identical positions shared by the sequences, taking into account the number of gaps, and the length of each gap, which needs to be introduced for optimal alignment of the two sequences. The comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm. For example, the percent identity between two nucleotide sequences can be determined using methods such as those described in Computational Molecular Biology, Lesk, A, M,, ed., Oxford University Press, New York, 1988; Biocomputing: Informatics and Genome Projects, Smith, D W., ed.. Academic Press, New York, 1993; Sequence Analysis in Molecular Biology, von Heinje, G., Academic Press, 1987; Computer Analysis of Sequence Data, Part I, Griffin, A. M., and Griffin, II G., eds., Humana Press, New Jersey, 1994; and Sequence Analysis Primer, Gribskov, M. and Devereux, J., eds., M Stockton Press, New York, 1991; each of which is incorporated herein by reference in its entirety. For example, the percent identity between two nucleotide sequences can be determined using the algorithm of Myers and Mi ller (CABIOS, 1989, 4: 11-17), which has been incorporated into the ALIGN program (version 2.0) using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4. The percent identity between two nucleotide sequences can, alternatively, be determined using the GAP program in the GCG software package using an NWSgapdna.CMP matrix. Methods commonly employed to determine percent identity between sequences include, but are not limited to those disclosed in Carillo, H., and Lipman, D., SIAM J Applied Math., 48: 1073 (1988); incorporated herein by reference in its entirety. Techniques for determining identity are codified in publicly available computer programs. Computer software to determine homology between two sequences include, but are not limited to, GCG program package, Devereux, J., et al., Nucleic Acids Research, 12(1), 387 (1984)), the Basic Local Alignment Search Tool (BLAST, which includes, e.g., BLASTP for protein sequences and BLASTN for nucleic acid sequences), and PASTA Altschul, S. F. et al., J. Molecular Biol., 215, 403 (1990)), EMBOSS Needle, Clustal Omega, Benchling, and Geneious. In preferred embodiments, sequence identity may be determined using BLA ST, Clustal Omega, or EMBOSS Needle.
[0418] Inhibit expression of a gene: As used herein, the phrase “inhibit expression of a gene” means to cause a reduction in the amount of an expression product of the gene. The expression product can be an RNA transcribed from the gene (e.g,, an mRNA) or a. polypeptide translated from an mRNA transcribed from the gene. Typically, a. reduction in the level of an mRNA results in a reduction in the level of a polypeptide translated therefrom.
The level of expression may be determined using standard techniques for measuring mRNA or protein.
[0419] Inverted terminal repeat: As used herein, the term "inverted terminal repeat” or “ITR” refers to a cis-regulatory element for the packaging of polynucleotide sequences into viral capsids.
[0420] Isolated: As used herein, the term "isolated” refers to a substance or entity that is altered or removed from the natural state, e.g., altered or removed, from at least some of component with which it is associated in the natural state. For example, a nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is “isolated.” An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell. Such polynucleotides could be part of a vector and/or such polynucleotides or polypeptides could be part of a composition, and. still be isolated, in that, such vector or composition is not part of the environment in which it is found in nature. In some embodiments, an isolated nucleic acid is recombinant, e.g., incorporated into a vector.
[0421] Modification: As used herein, the term “modification” or “modified,” refers to any substance compound or molecule that has been changed in any way. For example, a. modification in an amino acid sequence may comprise a substitution (e.g., a conservative substitution), an insertion, and/or a deletion of one or more amino acids in the sequence.
[0422] Modulatory polynucleotide: As used herein, a modulatory polynucleotide reduces or eliminates expression of a target, gene, e.g., thereby reducing or eliminated the protein encoded by the target gene. RNAi agents, such as siRNAs, are exemplary modulatory polynucleotides.
[0423] Molecular scaffold: As used herein, a “molecular scaffold” is a nucleic acid framework within which an inhibitory nucleic acid molecule is provided. For example, in the context of an siRNA molecule, a molecular scaffold is a nucleic acid framework within with a passenger strand and a guide strand is provided to form a stem loop structure.
[0424] Neurological disease: As used herein, a. “neurological disease” is any disease associated with the central or peripheral nervous system and components thereof (e.g., neurons).
[0425] Operably linked: As used herein, the phrase “operably linked” refers to a. functional connection between two or more molecules, constructs, transcripts, entities, moieties or the like.
[0426] Payload: As used herein, “payload,” “payload sequence,” or “payload region” refers to one or more polynucleotides or polynucleotide regions encoded by or within a viral genome or an expression product of such polynucleotide or polynucleotide region, e.g., a modulatory (e.g., inhibitor}') polynucleotide, e.g., an siRNA.
[0427] Payload construct: As used herein, “payload construct” is one or more polynucleotide regions encoding or comprising a payload that is flanked on one or both sides by an inverted terminal repeat (ITR) sequence. The payload construct is a template that is replicated in a viral production cell to produce a viral genome.
[0428] Payload construct vector: As used herein, “payload construct vector” is a vector encoding or comprising a payload construct, and regulator}' regions for replication and expression in bacterial cells. The payload construct vector may also comprise a component for viral expression in a viral replication cell .
[0429] Pharmaceutically acceptable: lire phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are suitable for use in contact with the tissues of human beings and animals.
[0430] Pharmaceutically acceptable excipients: As used herein, the term “pharmaceutically acceptable excipient,” as used herein, refers to any ingredient other than active agents (e.g,, as described herein) present in pharmaceutical compositions that can function as vehicles for suspending and/or dissolving active agents.
[0431] Pharmaceutically acceptable salts: Pharmaceutically acceptable salts of tire compounds described herein are forms of the disclosed compounds wherein the acid or base moiety is in its salt form (e.g., as generated by reacting a free base group with a suitable organic acid). Examples of pharmaceutically acceptable salts include, but are not limited to, mineral or organic acid salts of basic residues such as amines; alkali or organic salts of acidic residues such as carboxylic acids; and the like.
[0432] Pharmaceutical Composition: As used herein, the term “pharmaceutical composition” or pharmaceutically acceptable composition” comprises AAV polynucleotides, AAV genomes, or AAV particle and one or more pharmaceutically acceptable excipients, solvents, adjuvants, and/or the like.
[0433] Position: The term “position,” as used herein amino acid sequence, refers to the location of a particular amino acid or set of amino acids relative to a larger sequence. A position or positions of amino acids may interchangeably be referred to by an amino acid number or numbers of a reference sequence. For example, and unless otherwise specified, “positions 1-742, as numbered according to SEQ ID NO: 982” is interchangeable with
“amino acids 1-742” Preventing: As used herein, the term “preventing” refers to partially or completely delaying onset of an infection, disease, disorder and/or condition; partially or completely delaying onset of one or more symptoms, features, or clinical manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying onset of one or more symptoms, features, or manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying progression from an infection, a particular disease, disorder and/or condition ; and/or decreasing the risk of developing pathology associated with the infection, the disease, disorder, and/or condition. The term “prevention” or “preventing” of an infection, disease, disorder and/or condition may be considered a subset within the meaning with the term “treatment” or treating” of the infection, disease, disorder and/or condition.
[0434] Reduce or eliminate expression: As used herein, the term “reduce or eliminate expression” refers to the action of a modulatory polynucleotide to reduce the amount of the target gene expression product (e.g., ATXN2 mRNA or protein). Reduction may be partial reduction, or complete reduction (i.e., elimination). Reducing or eliminating expression may comprise targeting the gene, the mRNA and/or the protein. In some embodiments, reduced expression is reduced expression compared to the subject prior to administration. In some embodiments, reduced expression is reduced expression compared to a subject with SCA2 to whom a modulatory polynucleotide has not been provided. In some embodiments, reduced expression is reduced expression compared to a known reference level. In some embodiments, eliminating expression results in ATXN2 mRNA or protein being undetectable.
[0435] Region: As used herein, the term “region” refers to a zone or general area. In some embodiments, when referring to a protein or protein module, a region may comprise a linear sequence of amino acids along the protein or protein module or may comprise a three- dimensional area. In some embodiments, regions comprise terminal regions. As used herein, the term “terminal region” refers to regions located at the ends or termini of a given agent. When referring to proteins, terminal regions may comprise N- and/or C -termini. N -termini refer to the end of a protein comprising an amino acid with a free amino group. C-termmi refer to the end of a protein comprising an amino acid with a free carboxyl group. N- and/or C -terminal regions may comprise the N- and/or C -termini as well as surrounding amino acids. When referring to a polynucleotide, a region may comprise a linear sequence of nucleic acids along the polynucleotide or may comprise a three-dimensional area, secondary structure, or tertiary structure. In some embodiments, regions comprise terminal regions. As
used herein, the term “terminal region” refers to regions located at the ends or termini of a. given agent. When referring to polynucleotides, terminal regions may comprise 5’ and/or 3’ termini.
[0436] RNA interfering or RNAi: As used herein, the term “RNA interfering” or “RNAi” refers to a sequence specific regulatory mechanism mediated by RNA molecules which results in the inhibition, interfering, or “silencing” of the expression of a corresponding protein-coding gene. RNAi may result, in the knocking down (i.e., decreasing) or knocking out. (re., eliminating) of gene expression, which may be detected at the RNA level and/or protein level. RNAi has been observed in many types of organisms, including plants, animals and fungi. RNAi occurs in cells naturally to remove foreign RNAs (e.g., viral RNAs). Natural RNAi proceeds via fragments cleaved from free dsRNA which direct the degradative mechanism to other similar RNA sequences. The dsRNA molecules can be introduced into cells exogenously. Exogenous dsRNA initiates RNAi by activating the ribonuclease protein Dicer, which binds and cleaves dsRNAs to produce double-stranded fragments of 21-25 base pairs "with a few unpaired overhang bases on each end. These short double stranded fragments are called small interfering RNAs (siRNAs).
[0437] RNAi agent: As used herein, the term “RNAi agent” refers to an RNA molecule, or its derivative, that can induce inhibition, interfering, or “silencing” of the expression of a target gene and/or its protein product. An RNAi agent may knock-out (eliminate) expression, or knock-down (lessen or decrease) expression. The RNAi agent may be, but is not limited to, dsRNA, siRNA, shRNA, pre-miRNA, pri-miRNA, miRNA, stRNA, IncRNA, piRNA, or snoRNA.
[0438] Sample: As used herein, the term “sample” or “biological sample” refers to a subset of tissues, cells, nucleic acids, or a component or part of tire body (e.g. a body fluid, including but not limited to blood, mucus, lymphatic fluid, synovial fluid, cerebrospinal fluid, saliva, amniotic fluid, amniotic cord blood, urine, vaginal fluid and semen).
[0439] Sense Strand: As used herein, the tenn “the sense strand” or “the second strand” or “the passenger strand” of an inhibitory polynucleotide, e.g., an siRNA , refers to a strand that is capable of hybridizing to an antisense strand (also referred to as the first, strand or the guide strand). -As used herein, a “siRNA duplex” includes a siRNA strand having sufficient complementarity to a section of abo ut 10-50 nucleotides of the mRNA of the gene targeted for silencing and a siRNA strand having sufficient complementarity to form a duplex with the other siRNA strand.
[0440] Serotype: As used herein, the term “serotype"' refers to distinct variations in a
capsid of an AAV based on surface antigens which allow epidemiologic classifications of the AAVs at the sub-species level.
[0441] Silent Mutation: As used herein, a “silent mutation” or “silent substitution” refers to a nucleotide replacement that results in the same amino acid residue as a. reference sequence.
[0442] Similarity: As used herein, the term “similarity” refers to the overall relatedness between polymeric molecules, e.g., between polynucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of percent similarity of polymeric molecules to one another can be performed in the same manner as a calculation of percent identity, except that calculation of percent similarity takes into account conservative substitutions as is understood in the art.
[0443] Short interfering RNA or siRNA; As used herein, the terms “short interfering RNA,” “small interfering RNA” or “siRNA” refer to an RNA molecule (or RNA analog) composing about 5-60 nucleotides (or nucleotide analogs) and which is capable of directing or mediating RNAi. The term “short” siRNA refers to a siRNA comprising 5-23 nucleotides. The term “long” siRNA refers to a siRNA comprising 24-60 nucleotides. siRNAs can be single stranded RNA molecules (ss-siRNAs) or double stranded RNA molecules (ds-siRNAs) comprising a. sense strand and an antisense strand which hybridized to form a duplex structure called an siRNA duplex.
[0444] Spacer: As used herein, a “spacer” is generally any selected nucleic acid sequence of, e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides in length, which is located between two or more consecutive miR binding site sequences. In some embodiments, spacers may also be more than 10 nucleotides in length, e.g., 20, 30, 40, or 50 or more than 50 nucleotides.
[0445] Subject: As used herein, the term “subject” or “patient” refers to any organism to which a composition in accordance with the disclosure may be administered, e.g., for experimental, diagnostic, prophylactic, and/or therapeutic purposes. Similarly, “subject” or “patient” refers to an organism who may seek, who may require, who is receiving, or who will receive treatment or who is under care by a. trained, professional for a particular disease or condition. Typical subjects include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and humans). As used herein, a subject or patient may be susceptible to or suspected of having a ATXN2-related disorder, such as SCA2.
[0446] Substantially: As used herein, the team “substantially” refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. One of ordinary skill in the biological arts will understand that biological and
chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena.
[0447] Suffering from: An individual who is "‘suffering from” a disease, disorder, and/or condition has been diagnosed with or displays one or more symptoms of a disease, disorder, and/or condition.
[0448] Susceptible to: An individual who is “susceptible to” a. disease, disorder, and/or condition has not been diagnosed with and/or may not exhibit symptoms of the disease, disorder, and/or condition but harbors a. propensity to develop a disease or its symptoms. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition may be characterized by one or more of the following: (1) a genetic mutation associated with development of the disease, disorder, and/or condition; (2) a genetic polymorphism associated, with development of the disease, disorder, and/or condition; (3) increased, and/or decreased expression and/or activity of a. protein and/or nucleic acid associated with the disease, disorder, and/or condition; (4) habits and/or lifestyles associated with development of the disease, disorder, and/or condition; (5) a family history of the disease, disorder, and/or condition; and (6) exposure to and/or infection with a microbe associated with development of the disease, disorder, and/or condition. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition will develop the disease, disorder, and/or condition. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition wall not develop the disease, disorder, and/or condition.
[0449] Target Cells: As used herein, “target cells” refers to any one or more cells of interest. The cells may be found in vitro, in vi vo, in situ or in the tissue or organ of an organism. The organism may be an animal, preferably a mammal, more preferably a human and most preferably a. human patient.
[0450] Target Tissue: As used herein, “target tissue” refers to a tissue of interest that maybe found m vitro, in situ, or as part of an animal, preferably- a mammal, more preferably- a human and most preferably a human patient.
[0451] Therapeutic -Agent: The term “therapeutic agent” refers to any agent that, when administered to a subject, elicits a desired biological and/or pharmacological effect.
[0452] Therapeutically- Effective Outcome: As used herein, the term “therapeutically effective outcome” means an outcome that, is sufficient in a. subject suffering from or susceptible to an infection, disease, disorder, and/or condition, to treat, improve symptoms of.
delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition.
[0453] Treating: As used herein, the term “treating” refers to partially or completely alleviating, ameliorating, improving, relieving, delaying onset of, inhibiting progression of, reducing severity of, reducing incidence of, and/or preventing one or more symptoms or features of a particular infection, disease, disorder, and/or condition. Treatment may be administered to a subject who does not exhibit signs of a disease, disorder, and/or condition and/or to a subject who exhibits only early signs of a disease, disorder, and/or condition for the purpose of decreasing the risk of developing pathology associated with the disease, disorder, and/or condition.
[0454] Unmodified: As used herein, “unmodified” refers to any substance, compound or molecule prior to being changed in any way. Unmodified may, but does not always, refer to the wild-type or native form of a biomolecule or entity. Molecules or entities may undergo a series of modifications whereby each modified product may serve as the “unm odified” starting molecule or entity- for a. subsequent modification.
[0455] Variant: The term “variant” refers to a polypeptide or polynucleotide that has an amino acid or a nucleotide sequence that has at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to a reference sequence. The variant may be a functional variant. As used herein, the term “functional variant” refers to a polypeptide variant or a polynucleotide variant that has at least one activity of the reference sequence.
[0456] Vector; As used herein, a “vector” is any molecule or moiety which transports, transduces, or otherwise acts as a carrier of a heterologous molecule. Vectors of the present disclosure may be produced recombinantly and may be based on and/or may comprise adeno- associated virus (AAV) parent or reference sequence(s).
[0457] Viral genome: As used herein, a. “viral genome”, “vector genome”, or “VG” is a polynucleotide comprising at least one inverted tenninal repeat (ITR) and at least one nucleic acid sequence encoding a payload. A viral genome encodes at least one copy of the payload. EXAMPLES
[0458] The present disclosure is further illustrated by the following non-limiting examples.
Example 1. High-throughput screen of TRACER AAV library in NHP and Mice [0459] A TRACER based method as described in W02020072683 , WO 2021/202651 , and WO2021230987, the contents of which are herein incorporated by reference in their
entirety, was used to generate the AAV capsid variants described herein. An orthogonal evolution approach was combined with a high throughput screening by NGS. Briefly, the library of AAV capsid variants was generated using a. sliding window approach, where 6 amino acid sequences were inserted into 8 different positions across loop IV of AAV9, including immediately subsequent to positions 453, 454, 455, 456, 457, 458, 459, and 460, relative to a reference sequence numbered according to SEQ ID NO: 138. lire initial library was passed twice through non-human primates (NHP, 2-4 years of age). After the second passage (e.g., 28 days post injection into two NHPs), RNA was extracted from six brain regions. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate fold enrichment relative to an AAV9 wild-type control. Following these two passages, approximately 21 195 variants were identified with an average fold change greater than wild-type . Of the 21195 variants, 1558 demonstrated a fold-change of greater than 6 compared to wild-type and were detected across all brain regions investigated. Of these 1558, approximately 1470 variants were selected for constructing a synthetic library and a third passage through two NHPs. Within the 1470 variants selected for further characterization and investigation, there was a relatively even distribution for each insertion position of the sliding window used to generate the initial library.
[0460] After creation of the synthetic library with the sub-selected, variants, the synthetic library was screened (passage 3) in two NHPs (2-4 years of age) and two strains of mice, BALB/c (n=3, 6-8 weeks of age) and C57B1/6 mice (n=3, 6-8 weeks of age), in a first cross- species evolution screen. The animals were injected intravenously with the synthetic library-'. After a period in vivo, (e.g., 28-days) RNA was extracted from nervous tissue, e.g., brain, spinal cord, and DRG of the NHPs and the brains of mice. Following RNA recovery and RT- PCR amplification, a systematic NGS enrichment analysis was performed, and the peptides comprised within the variants were identified and the capsid enrichment ratio for each variant compared to the w'ild-type AAV9 control was calculated (fold enrichment relative to wild- type AAV9) (Table 11). Values above 1 indicate an increase in expression relative to AAV9. All animals were dosed intravenously at 2-3 VG/kg across the screen.
[0461] As shown in Table 11, approximately 700 variants demonstrated an increase in expression relative to AAV9, and several variants demonstrated a greater than 10-fold enrichment relative to AAV9 m the brain of NHPs. Further, the variants demonstrating the greatest fold enrichment in the brain also demonstrated, the greatest fold enrichment in the spinal cord relative to AAV9 in NHPs. These variants also demonstrated de-targeting in the DRG (data not shown). For instance, the variant comprising GSGSPHSKAQNQQT (SEQ ID
[0462] NO: 200) demonstrated a 76.6 fold enrichment in the brain, a 29.4 fold enrichment in the spinal cord, and 0.4 fold enrichment in the DRG of NHPs relative to AAV9; and
GHDSPHKSGQNQQT (SEQ ID NO: 201) demonstrated a 62.6 fold enrichment in the brain. a 15.6 fold enrichment in the spinal cord, and 0.0 fold enrichment in the DRG of NHPs relative to AAV9. Also, across the peptides comprised -within the AAV capsid variants with the greatest fold-enrichment in the NHP brain relative wild-type AAV9, it was observed that each of these peptides comprised an SPH motif in the same position (e.g., immediately subsequent to position 455, relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138), regardless of the insertion position within the variant capsid, as well as a. positive amino acid, (e.g., K or R) in one of the next three residues subsequent to the SPH motif.
[0463] Those variants with the greatest fold enrichment in the brains of NHPs also had the greatest fold enrichment in the brains of both mouse species. Also, when comparing the fold enrichment relative to wild-type for each variant between the two species of mice investigated (C57B1/6 and BALB/c mice), they were R2= 0.8591).
[0464] A second cross-species evolution screen was performed using an AAV capsid variant library with a modification in loop IV introduced as described above and passaging it once through
NHPs (passage 1) and then subsequently injected it into two different strains of mice (passage 2),
C57B1/6 and BALB/c. The fold-enrichment for each variant in the brain of each mouse species was calculated by systematic NGS enrichment analysis following RNA recovery and R’T-PCR amplification. The fold enrichment values in the second passage in mice were compared to those fold enrichment values from the second pass that was performed in NHPs as described above. As shown in Table 12. when comparing the second pass fold enrichment values in the mice versus NHPs, 12 variants were identified tliat had a fold-enrichment value greater than 10 in all three animal groups.
Further, 10 of these 12 variants comprised the SPH motif and a positive residue in one of the next
three subsequent residues (Table 12).
Table 12. NGS fold-enrichment of AAV capsid variants from a second passage (P2) in NHPs or mice (C57BI/6 or BALB/c) following a first passage in NHPs
[0465] Following the second passage in mice, a synthetic library was generated using those variants that demonstrated a fold-change in enrichment relative to wikl-type AAV9 that was above 10 in the brain of either strain of mice, as measured by systematic NGS enrichment analysis following RNA recovery and RT-PCR amplification. There were approximately 500 variants in this synthetic library. Tins synthetic library was then injected back into both strains of mice (C57B1/6 and BALB/c; passage 3). RNA was recovered from the mouse brains, RT-PCR amplification was performed, and fold-enrichment relative to wild-type AAV9 was calculated by NGS analysis, which is provided in Table 13. As shown in Table 13, the variants with the greatest fold-enrichment in the brain in each strain, were highly correlated across strains (R2 "0.8458).
Table 13. NGS fold-enrichment of AAV capsid variants in the brain from a third passage (P3) in mice (C57BI/6 or BALB/c) following a first and second passage in mice
[0466] Taken together, these results demonstrate that after 3 rounds of screening of this
AAV9 variant library with loop IV modifications in NHP and mice, many AAV capsid variants outperformed the wild-type AAV9, for example, in penetration of the blood brain barrier (BBB) and spinal cord expression. These capsid variants were able to cross-species,
evidenced by expression and tropism in the NHP brain/spinal cord as well as in the brain of two different mouse species.
Example 2. Individual Capsid Characterization in Mice
[0467] The goal of these experiments was to determine the transduction level, tropism, ability to cross the blood brain barrier, and overall spatial distribution in the central nervous system (CNS) of 2 capsid variants selected from the study described m Example 1 relative to AAV9 following intravenous injection in mice. The 2. capsid vanants were TTM-001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941) and TTM-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 2), as outlined in Table 3 above. The amino acid and DNA. sequences of TTM-001 and TTM-002 are provided, e.g., in Tables 4 and 5, respectively.
[0468] AAV particles were generated with each of these capsid variants encapsulating a luciferase-EGFP transgene driven by a CMV, -'chicken beta actin promoter in a single stranded viral genome. Each capsid variant and AA.V9 control were tested, by intravenously administering by tail vein injection, the AAV particle formulation at. 5e11 VG/dose (2.5E13 vg/kg) to three female BALB/c mice. The in-life period was 28 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA, transgene protein, and viral DNA (biodistribution).
[0469] At 28 days post-injection of the AAV particles encapsulated in the TTM-001 capsid variant (AAV TTM-001), mice were injected with. luciferin and their brains were harvested for IVIS imaging. Robust luciferase signal was observed in mice injected with AAV particles encapsulated in the TTM-001 capsid variant, and this was greatly increased relative to AAV particles encapsulated in the wild-type AAV9 control capsid.
[0470] The brains isolated from mice injected with the AAV particles encapsulated in the TTM-001 capsid variant (AAVJTTM-001) or the TTM-002 capsid variant. (AAVJTTM-002) were assayed by qPCR for the presence of transgene RNA as a. measure of transgene expression, and the presence of viral DNA as a measure of viral genome levels. Data were provided as fold over AAV9 (Table 14). As shown in Table 14, when compared to the wiId- type AAV9 capsid control, TTM-001 and TTM-002 demonstrated a 30-fold and 66-fold increase, respectively, in transgene mRNA levels and expression in the brain, indicative of enhanced payload delivery. This correlated with a 32-fold (TTM-001) and 47-fold (TTM- 002) increase, respectively, in viral genome (DNA) concentrations in the brain relative to the AAV9 capsid control, which is indicative of enhanced CNS tropism and transduction (Table 14).
Table 14. Transgene mRNA and viral genome levels (DNA) in mice relative to the
[0471] The brain tissues and spinal cords of the mice were also subjected to anti-GFP immunohistochemistry staining to evaluate overall CNS tropism and biodistribution. Immunohistochemical staining correlated with the qPCR analysis, as TTM-001 and TTM- 002 showed significantly stronger staining and payload expression in the brain and spinal cord, as compared to the AAV9 control. More specifically, TTM-001 and TTM-002 demonstrated localization and strong payload expression and transduction in the mid-brain region, with increased staining observed in the hippocampus and thalamus, as well as in the brain stem, compared to AAV9. Less staining was observed in the cortical regions of the brain compared to tire midbrain. However, staining in these cortical regions was stronger for TTM-001 and TTM-002 compared to the AAV9 control. It also appeared that the TTM-001 and TTM-002 capsid variants were able to transduce non-neuronal cells, including glial cells and oligodendrocytes. With respect to the spinal cord, staining and payload expression for TTM-01 and TTM-002 were localized, to the ventral horns of the grey matter.
[0472] Peripheral tissues were also isolated from the mice intravenously injected with the AAV particles encapsulated in the TTM-001 capsid variant or the TTM-002 capsid variant for analysis by qPCR and/or GFP immunohistochemical staining. Transgene mRNA levels and viral genome DNA. levels were quantified, in the liver by qPCR and the fold over AAV9 was calculated for each capsid variant (Table 14). TTM-001 resulted in similar levels of payload expression (mRNA levels) as compared to wild-type AAV9, but only half as much viral genome DNA was quantified in the liver compared, to AAV9. TTM-002 demonstrated greatly reduced mRNA and viral genome DNA levels in the liver compared to AAV9. GFP immunohistochemical staining of the spleen, heart, skeletal muscle, kidneys, and lungs of mice injected with AAV particles encapsulated in the TTM-001 capsid variant or the TTM- 002 capsid variant showed similar levels of payload expression as compared to those mice injected -with AAV particles encapsulated in the wild-type AAV9 control capsid.
[0473] Taken together, these data demonstrate that TTM-001 and TTM-002 are enhanced
CNS tropic capsids in mice that can infect non-neuronal cells. Additionally, these capsid variants were able to successfully penetrate the blood brain barrier following intravenous injection.
Example 3. Maturation of TTM-001 and TTM-002 Capsid in Mice
[0474] Ulis Example describes maturation of the TTM-001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941) and TTM-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 2) capsid variants to further enhance their transduction and biodistribution in the central nervous system and evolve the AAV capsid variants to provide further cross-species compatibility. Two approaches were used to mature the TTM-001 and TTM-002 capsid sequences in order to randomize and mutate within and around the peptide insert: comprised within loop IV of the capsid variant. As many of the AAA' capsid variants that demonstrated the greatest fold-enrichment in the NHP brain relative wild-type AAV9 comprised an SPH motif in the same position (e.g., immediately subsequent to position 455, relative to a. reference sequence numbered according to the amino acid, sequence of SEQ ID NO: 138) (see Example 1 ), the SPH motif was not mutated in either approach to mature the TTM-001 and TTM-002 capsid variants. In the first maturation approach, sets of three contiguous amino acids were randomized across the mutagenesis region in the TTM-001 and TTM-002 sequences, which spanned from position 450 to position 466, numbered according to SEQ ID NO: 981 and 982. In the second maturation approach, mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region m the TTM-001 and TTM-002 sequences ranging from position 449 to position 466, numbered according to SEQ ID NO: 981 and 982. AAV capsid variants arising from each maturation approach for TTM-001 were pooled together and AAV capsid variants arising from each maturation approach for TTM-002 were also pooled together, for subsequent testing and characterization in mice.
[0475] The library of pooled matured AAV capsid variants generated from TTM-001 or library of pooled matured AAV capsid variants generated from the TTM-002 matured AAV capsid variant each were intravenously injected, into the tail vein of three female CD-1 Outbred mice (Charles River) at a dose of 1.0 x 1012 VG/dose. After 14-da.ys in life, the brains of the mice were isolated and RNA was extracted. Following RNA recovery and RT- PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the corresponding TTM-001 or TTM-002 control, and the peptides comprised within the variants were identified. The data for the TTM-001 matured
capsid variants is provided in Table 15 and the data for the TTM-002 matured capsid variants is provided in Table 16.
[0476] As shown in Table 15, approximately 714 TTM-001 matured capsid variants demonstrated at least a 2-fold increase in expression relative to the non-matured TTM-001 control, and several variants demonstrated greater than a four-fold enrichment relative to the non-matured TTM-001 control. Also, across the peptides comprised within the TTM-001 matured capsid variants with the greatest fold-enrichment relative to the non-matured TTM-
001 capsid in the brain, it was observed that the modifications in the variant sequences appeared in the region C-terminal to the SPH motif present within the capsid variant. This indicates that modifications that appeared to improve TTM-001 capsid tropism in the CNS of mice were skewed to the C-terminal portion of the peptide insertion in loop IV of the sequence. -Additionally, a number of these C-terminal modifications were the incorporation of an arginine (R) or leucine (L) residue.
Table 15. NGS fold-enrichment of TTM-001 matured AAV capsid variants in the brain of CD-I Outbred mice
[0477] As shown in Table 16, approximately 72 TTM-002 matured capsid variants demonstrated at least a 2-fold increase in expression relative to the non-matured TTM-002 control, with a few variants demonstrating greater than a three- to five -fold enrichment relative to the non-matured TTM-002 control. Also, across the peptides comprised within the
TTM-002 matured capsid variants with the greatest fold-enrichment relative to the non matured TTM-002 capsid in the brain, it was observed that the modifications in the variant sequences appeared in the region N-terminal to the SPH motif present within the capsid variant. This indicates that modifications that appeared to improve TTM-002 capsid tropism in the CNS of mice were skew ed to the N -terminal portion of the peptide insertion in loop IV of the sequence. Additionally, a number of these N-terminal modifications that were incorporated into the matured TTM-002 capsid variants were negatively charged amino acids
(particularly glutamic acid (E)).
Table 16, NGS fold-enrichment of TTM-002 matured AAV capsid variants in the brain of CD-I Outbred mice
[0478] These data demonstrate that following two maturation approaches, matured TTM-
001 and TTM-002 capsid variants with loop IV modifications were generated with significantly enhanced CNS tropism in mice compared, to the corresponding non-matured
TTM-001 and TTM-002 capsid variants, which already exhibited a significant fold enrichment over AAV9 in the mouse brain.
Example 4. Maturation of TTM-001 and TTM-002 Capsid in NHPs
[0479] This Example describes maturation of the AAV9 capsid variants, TTM-001 (SEQ
ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941 (encoded by SEQ ID
NO: 942)) and TTM-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ
ID NO: 2 (encoded by SEQ ID NO: 3)) in NHPs to further enhance their transduction and. biodistribution in the central nervous system as well as other tissues, and evolve the AAV capsid variants to provide further cross-species compatibility. Two approaches were used to mature the TTM-001 and. TTM-002 capsid, sequences in order to randomize and mutate within and around the peptide insert comprised within loop IV of the capsid variant. As many of the AAV capsid variants that demonstrated the greatest fold-enrichment in the NHP brain
relative wild-type AAV9 comprised an SPH motif in the same position (e.g., immediately subsequent to position 455, relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138) (see Example 1), the SPH motif was not mutated in either approach to mature the TTM-001 and TTM-002 capsid variants. In the first maturation approach, sets of three contiguous amino acids were randomized across the mutagenesis region in the TTM-001 and TTM-002 sequences, which spanned from position 450 to position 466, numbered according to SEQ ID NO: 981 and 982. In the second maturation approach, mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region in the TTM-001 and TTM-002 sequences ranging from position 449 to position 466, numbered according to SEQ ID NO: 981 and 982, AAV capsid variants arising from each maturation approach for TTM-001 and TTM-002 were pooled together, for subsequent testing and characterization in NHPs.
[0480] The library of pooled matured AAV capsid variants generated using the first maturation approach and the second maturation approach for the TTM-001 and TTM-002 AAV capsid variants were injected into two NHPs. After a period in life, the brains, heart, liver, muscle, and DRG of the NHPs were isolated and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic. NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to an AAV9 control, and. the peptides comprised within the variants were identified.
[0481] Following the RNA recovery and NGS analysis from the second maturation approach, approximately 680,000 capsid variants were identified. The 680,000 matured capsid variants were then filtered based on samples with a raw virus count greater than 10 and a coefficient of variance (CV) of less than 1, which was calculated for each peptide across the brain samples taken from the two NHPs. Those that had a CV value <1 were identified, as these were the peptides that were reliably detected in the majority of samples isolated from the brains of the two NHPs. Using this filtering criteria, this led to approximately 64,000 matured capsid variants.
[0482] Table 17 provides the peptide sequences of the matured capsid variants having a raw virus count greater than 10, a CV of less than 1 for the brain samples isolated, and that also demonstrated a 50-fold or greater fold-increase in expression in the brain relative to the AAV 9 control in both mice and NHPs. The matured variants in Table 17, were also those variants that had a fold-change in expression that was less than 2 relative to the AA V9 control in the liver and the DRG. Applying these criteria, approximately 350 matured capsid variants were identified that demonstrated high transduction in the brain in NHPs and mice.
cross-species compatibility in mice and NHPs, and were de-targeted in the liver and DRG, relative to the AAV 9 control. Several variants as shown in Table 1.7, led to greater than 100- fold increase in expression relative to AAV9 in the NHP and/or mouse brain, with one variant resulting in a greater than 200-fold increase in expression relative to AAV9 in both species.
J0483] Fold-change in expression for the TTM-001 and TTM-002 matured variants in
Table 17 that showed increased expression in the brain of the NHPs and mice, w ere also calculated for the DRG, muscle, liver (RNA and DNA), and. heart of the NHPs following each maturation approach. As shown in Table 17, many variants were de-targeted in the peripheral tissues with a lower fold-change in expression relative to the AAV9 control. demonstrating CNS-specific tropism and a preferential transduction of the brain and CNS,
Some variants demonstrated increased expression to AAV9 in multiple tissues, including the brain and peripheral tissues, demonstrating pan-tropism.
Table 17. NGS fold-enrichment of TTM-001 and TTM-002 matured AAV capsid variants in the brain of NHPs and mice
[0484] Table 18 provides the peptide sequence of 341 matured capsid variants, and the fold enrichment of these matured capsid variants relative to the AAV9 control that demonstrated a 75-fokl or greater increase in expression in the brain of NHPs relative to the
AAV9 control and had a fold-change in expression that was less than 2 relative to the AAV9 control in the liver and the DRG
Table 18. NGS fold-enrichment of TTM-001 and TTM-002 matured AAV capsid variants in the brain of NHPs
5
QT 3
[0485] Table 19 provides the sequences of 216 matured capsid variants having a CV of less than 1 for the liver RNA samples isolated and a 10-fold or greater increase in expression relative to AAV 9 in the liver of NHPs. These matured variants showed preferential transduction of the liver over other tissues as shown by a low value for fold-enrichment relative to AAV9 in the other tissues investigated including the brain, DRG, heart and muscle. As such. Table 19 provides TTM-001 and TTM-002 matured AAV capsid variants with liver-specific tropism. Across the peptides within the matured capsid vanants in Table
19, approximately 175 of them comprised the sequence GSGSPH (SEQ ID NO: 4695) and further comprised additional modifications in the C -terminal region of the sequence.
Table 19. NGS fold-enrichment of TTM-001 and TTM-002 matured AAV capsid variants in the liver of NIIPs
[0486] Table 20 provides the peptide sequences of 43 matured capsid variants having a raw virus count greater than 10, a CV of less than 1 for the heart samples isolated, and that also demonstrated a 4-fold or greater fold-increase in expression in the heart relative to the
AAV9 control. A number of the matured variants shown in Table 20 also demonstrated increased expression in other tissues isolated from the NHPs, including the brain, muscle, and/or liver, and are therefore pan-tropic.
Table 20. NGS fold-enrichment of TTM-001 and TTM-002 matured AAV capsid variants in the heart of NHPs
[0487] Table 21 provides the peptide sequences of 14 matured capsid variants having a raw virus count greater than 10, a CV of less than 1 for the muscle samples isolated (e.g.. quadriceps), and that also demonstrated a. 4-fold or greater fold-increase in expression in the
muscle relative to the AAV9 control. A number of the matured variants shown in Table 21 also demonstrated increased expression in other tissues isolated from the NHPs, including the brain, heart, and/or liver, and are therefore pan-tropic.
Table 21. NGS fold-enrichment of TTM-001 and TTM-002 matured AAV capsid variants in the muscle (e.g., quadriceps) of NHPs
[0488] Additional variants were identified following generation and screening in NHPs that had the following properties. TTM-001 and TTM-002 capsid variants comprising the amino acid sequence of SEQ ID NOs: 4253, 4281, 4290-4295, 4304, 4305, 4320, 4328-4335,
4337-4340, 4353, 4355, 4369, 4387, 4421, 4424-4428. 4430, 4432, 4433, 4435, 4436-4449,
4452, 4455, 4476, 4483, or 4484 had a raw virus count 10 or greater, a CV of less than 1 for the brain samples isolated from the NHPs, demonstrated a 50-fold or greater increase in expression in the brain of mice and NHPs relative to AAV9, and demonstrated 2-fold or less expression in the liver and DRG of NHPs relative to AAV9. TTM-001 and TTM-002 capsid
variants comprising the amino acid sequence of SEQ ID NOs: 4098-4105, 4254-4280, 4282- 4289, 4296-4303, 4306-4327, 4336, 4341-4352, 4354, 4356-4420, 4422, 4423, 4425, 4429, 4431, 4434, 4444, 4450, 4451, 4453, 4454, 4456-4475, 4477-4482, or 4485 had a. CV of less than 1 in across the brain samples isolated from the NHPs and demonstrated a 100-fold or greater increase in expression in the brain of NHPs relative to AAV9. TTM-001 and TTM- 002 capsid variants comprising the amino acid sequence of SEQ ID NOs: 4102 and 4106 4252 had normalized virus counts of greater than or equal to 0,01, a CV of less than 1 across the liver RNA samples isolated from the NHPs, and demonstrated a 20-fold or greater increase in expression in the liver of NHPs relative to AAV9. TM-001 and TTM-002 capsid variants comprising the amino acid sequence of SEQ ID NO: 4105 had a raw virus count 9.9 or greater, a CV of less than 1 across the muscle samples isolated from the NHPs, and 5-fold or greater increase in expression in the muscle of the NHPs relative to AAV9. TM-001 and TTM-002 capsid variants comprising the amino acid sequence of SEQ ID NO: 4105 also had a raw virus count 9.9 or greater, a CV of less than 1 across the samples isolated from the heart of the NHPs, and 5-fold or greater increase in expression in the heart of the NHPs relative to AAV9.
[0489] These data demonstrate that following two maturation approaches, matured TTM- 001 and TTM-002 capsid variants (AAV9 capsid variants) with loop IV modifications were generated with significantly enhanced CNS tropism over wild-type AAV9 controls in both NHPs and mice, while also exhibiting de-targeting in peripheral tissues (e.g., the liver and DRG). These resulting matured, variants therefore demonstrated cross-species CNS tropism in both NHPs and mice. Matured TTM-001 and TTM-002 capsid variants with liver-specific tropism were also generated with at least 10 times the expression compared to wild-type AAV 9 in the liver of NHPs. Several matured variants were also generated with increased expression in the heart and skeletal muscle (e.g., quadriceps) relative to wild-type AAV9 in NHPs.
Example 5. Evaluation of TTM-001 and TTM-002 AAV capsid variants in Diverse Primate Species
[0490] This Example evaluates the tropism and cross-species compatibility of the TTM- 001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941) and TTM-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 2) capsid variants in two diverse primate species, mannosets (Callithrixjacchus) and African green monkeys {Chlorocebus sabaeus), as compared to their tropism in cynomolgus macaques
(M acaca fascicularis) provided in Example 1. The cross-species compatibility and tropism of an AAV9 capsid variant comprising the amino acid sequence of SPHKYG (SEQ ID NO: 966) was also investigated in this example. The amino acid and. DNA sequences of TTM-001 and TTM-002 are provided, e.g., in Tables 4 and 5, respectively.
|0491] To investigate tropism in African green monkeys, .AAV particles comprising the TTM-001 capsid variant, the TTM-002 capsid variant, an AAV9 capsid variant comprising SEQ ID NO: 966, or an AAV9 control under the control of a synapsin promoter, were intravenously injected into NHPs (n=2, 3-12 years of age) at a. dose of 2E13 vg/kg. After 14- days in life, the brains and tissues (liver, DRG, quadriceps, and heart) of the NHPs were collected and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the AAV9 wild-type control.
[0492] To investigate tropism in marmoset monkeys, AAV particles comprising the TTM- 001 capsid variant, the TTM-002. capsid variant, an AAV9 capsid variant comprising SEQ ID NO: 966, or an AAV9 control, were intravenously injected into NHPs (n=2, >10 months of age) at a dose of 2E13 vg/kg (8.75E12 vg/rnL). After 28-days in life, the brains and tissues (liver quadriceps, and heart) of the NHPs were collected and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the AAV9 wild-type control. [0493] As provided in Table 22 (African green monkeys) and Table 23 (mannosets), both the TTM-001 and TTM-002. capsid variants demonstrated, increased. CNS tropism in diverse primate species. The TTM-001 capsid variant demonstrated a 73.6-fold increase in expression relative to AAV9 in the brain of cynomolgus macaques (Table 11, Example 1), a 43.5-fold increase in expression relative to AAV9 in the brain of African green monkeys, and a 703.3-fold increase in expression relative to AAV9 in the brain of marmosets. The TTM- 002 capsid variant demonstrated a 62.6-fold increase in expression relative to AAV9 in the brain of cynomolgus macaques (Table 11), a 13.8-fold increase in expression relative to AAV9 in the brain of African green monkeys, and a 366.6-fold, increase in expression relative to AAV9 in the brain of mannosets. Both TTM-001 and TTM-002 led to a. significan t increase in expression relative to AAV9 in the heart of both African green monkeys and marmosets (Table 22 and Table 23). The AAV9 capsid variant comprising SEQ ID NO: 966 also demonstrated in increase in expression relative to AAV9 in the brain and heart of both African green monkeys and marmosets. Furthermore, TTM-001 , TTM-002, and the AAV9 capsid variant comprising SEQ ID NO: 966, also all led to increased expression in the brain
of both BALB/c and C57B1/6 mice (Table 13, Example 1), demonstrating an average fold change in expression relative to .AAV 9 across both species of mice of 63.1, 66.8, and 126.97, respectively.
Table 22. NGS-foId enrichment of TTM-001 (comprises SEQ ID NO: 941), TTM-002 (comprises SEQ ID NO: 2), and an AAV9 capsid variant comprising SEQ ID NO: 966 in African green monkeys
Table 23. NGS-foId enrichment of TTM-001 (comprises SEQ ID NO: 941), TTM-002 (comprises SEQ ID NO: 2), and an AAV9 capsid variant comprising SEQ ID NO: 966 in marmosets
[0494] Taken together, these data demonstrate that the AAV9 capsid variants of TTM~001 and TTM-002 demonstrated increased CNS tropism relative to the AAV9 control in the CNS across three diverse primate species and two species of mice, providing evidence of strong cross-species capacity. The AA.V9 capsid variant comprising the amino acid sequence of SEQ ID NO: 966 also demonstrated strong CNS expression relative to the AAV9 control in two species of NHPs and two species of mice, also showing strong cross-species capacity. Example 6. Advanced maturation of TTM-002 capsid variant in mice
[0495] This Example describes additional maturation of the TTM-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 2) capsid variant in mice. In order to mature the TTM-002 capsid variant, sets of three contiguous amino acids were randomized across the mutagenesis region in TTM-002 sequence, which spanned from position 450 to position 466, numbered according to SEQ ID NO: 982. Unlike the maturation performed in in Example 3, where the SPH motif that was observed in the AAV capsid variants that demonstrated the greatest fold-enrichment in the NHP brain relative wild-type AAV9 was not disrupted, in the maturation approach used in this Example, the SPH motif was not held constant to further explore the role of this motif in the capsid variant. The matured TTM-002
capsid variants that resulted from the maturation approach were pooled together for subsequent testing and characterization in mice.
[0496] The library of matured AA V capsid variants generated, from the TTM-002 matured AAV capsid variant were intravenously injected into the tail vein of three CD-I Outbred mice (Charles River; 6-8 weeks of age) at a dose of 1 .0 x 1012 VG/dose. After about 28 days in life, the brains of the mice were isolated, and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the corresponding TTM-002 non-matured control, and the peptides comprised within the variants were identified. Variants were filtered by those with a raw virus count in the sample above 10 and a coefficient of variance (CV) that was greater than 1 (identifies the peptides/variants reliably detected in the majority of the samples isolated from the three mice).
[0497] Following the advanced maturation screen and filtering of the variants, 1302 variants demonstrated an increase in expression relative to the non-matured TTM-002 capsid, variant in the brain of the outbred mice. Of the 1302 variants with improved tropism relative to the non-matured TTM-002, 1283 comprised the SPH motif in the same position as the non- matured TTM-002 capsid variant (e.g., immediately subsequent to position 455, relative to a reference sequence numbered, according to the amino acid sequence of SEQ ID NO: 138 or 982). Mutations in the region of the SPH motif present in the non-matured TTM-002 capsid variant only consistently appear in those variants with a fold change of 0.2 or 0.1 or lower relative to the non-matured TTM-002 control in the brain of the mice. This indicates that the
SPH motif may be important to the increased brain tropism that observed for the TTM-002 capsid variant. In instances when the SPH motif was disrupted, the fold change of the matured variants of TTM-002 decreased considerably in relation to the non-matured TTM- 002 variant, which comprised the SPH motif
Example 7. Tropism of TTM-002 AAV capsid variant
[0498] This Example further investigates the tropism and CNS cells transduced by the TTM-002 capsid variant (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 2), as outlined in Table 3 above. The amino acid and DNA sequences of TTM-002 are provided, e.g., in Tables 4 and 5, respectively.
[0499] AAV particles were generated with the TTM-002 capsid variant encapsulating a GFP transgene (AAV JTTM-002.GFP) or a payload driven by a. heterologous CBA constitutive promoter (AAV_TTM-002.Payload).
[0500] Two tandem single cell RNA sequencing runs (scRNA-Seq) of mouse cells derived from the midbrain area were performed. In the first run, cells were pooled from two mice at day 28 post treatment with AAV_TTM-002. Payload particles. In the second run we treated with AAV_TTM-002.GFP particles, in the same manner but without xenografts. Orthotopic xenografts of MDA-MB-361-Luc#l high passage cells grown as tumorspheres (in tumorsphere media; Sigma # C -28070) were injected (250,000 cells/2pL/mouse) intracranially into 2 -month old female SCID CB17 (Mutation: Icr-Prkdcscid/IcrlcoCrl) congenic immunodeficient mice (Charles River Laboratories). The injections were 2.5mm (lateral), -1mm (posterior) with respect to bregma, lowered -3mm ventral and raised +.5 mm dorsal to a final -2.5mm ventral position. Two days later, dilutions of the AAV_TTM- 002.Payload particles (run 1 ), or in the case without xenografts, dilutions of AAV_TTM- 002.GFP particles (run 2) were prepared. IV injections of lOOpL (2.5el 1 VG/anirnal) of the AAV TTM-002. payload particles or AAV TTM-002..GFP particles were administered through the tail veins of mice (n=5 mice per groups). At 7 days post-injection, mice from run 1 were imaged in an AmiHTX (Spectral Imager) for bioluminescence of the human tumor cells due to expression of luciferase in response to intraperitoneal luciferin injections.
[0501] At 28 days post-injection 'with the AAV TFM-002. pay load particles or AAV_TTM-002.GFP particles, two mice from each ran were necropsied, brain samples were isolated, and the midbrain was dissected and isolated. The midbrain samples were then exposed to a cold protease inhibitor (Creative Biomart
) and were dissociated at 6 degrees centigrade. For the samples collected from the mice of run I (AAV_TTM- 002. Payload particles), myelin depletion was performed (Miltenyi, #130-096-731), cells were filtered through a 40μM mesh to filter out neurons) and loaded on a 10X chromium G chip. scRNA-Seq was performed (10X Genomics) and samples were sequenced on aNextGen500 Sequencing machine (Illumina). For the samples collected from run 2. (AAV_TTM-002.GFP particles and no xenografts), the cells were not myelin depleted or filtered through 40μM mesh to include neurons. The cells isolated after run 2 were FACS sorted for GFP+/7AAD-
(live GFP+ ceils). The resultant cells were loaded on a 10X chromium G chip and the scRNA-Seq was run and processed (10X Genomics).
[0502) For rim 1, the scRNA-Seq data was filtered to include cells with only greater than 1000 genes per cell and less than 5000, and less than 20 percent mitochondrial gene expression. For run 2, the scRNA-Seq data was filtered to include cells with only greater than 200 genes per cell and less than 5000, and less than 20 percent mitochondrial gene expression. The data were normalized, scaled, and integrated into one combined dataset.
Clusters were generated with a resolution of 0.3 and each cluster identity was determined using a panel of cell type specific genes (e.g., as described in Brown et al., 2021. “Deep Parallel Characterization of AA V Tropism and AAV-Mediated Transcriptional Changes via Single-Cell RNA Sequencing”. Front. Immunol. 12:730825; the contents of which are hereby incorporated by reference in its entirety). Th e percentage of GFP sorted cells per cluster -was calculated as was the percentage of pay load expressing genes per cluster as parallel measures of TTM-002 transduction .
[0503] For payload expressing cells, endothelial cells had the highest proportion of payload positive cells, followed by astrocytes (Table 24). For GFP+ sorted cells, endothelial cells had the highest proportion of GFP positive cells, and astrocytes were the third highest cell type when sorting by proportion of cells expressing GFP (Table 24). These data indicate TTM-002 transduction exhibits an endothelial and astrocytic tropism. Furthermore, the astrocytic cluster had the second highest level of expression of Olig2 (oligodendrocytes demonstrated the greatest Olig2 expression). IHC staining was performed, on brain samples isolated from AAV TTM-002. GFP infected mice and demonstrated that GFP co-localized with some but not all Olig2+ cells. No co-staining was observed with mylein basic protein (MBP), a marker of oligodendrocytes. Co-staining with GFP was also not observed in NeuN positive cells (neurons), GFAP positive cells (astrocytes), and Ibal positive cells (microglia). GFP staining was observed throughout the sagittal section of the mouse brain, which was demonstrative of increased staining in the midbrain, lire GFP expressing cells observed did not have a bipolar morphology like oligodendrocyte progenitor (OPC) cells and therefore, together with the scRNA-Seq data, these results indicated that at day 28 post AAV treatment, Olig2+ astrocytes in the midbrain are being transduced by AAV particles comprising a TTM- 002 capsid, in a cell type specific tropism.
Example 8. Individual Capsid Characterization of TTM-001, TTM-002, TTM-003, TTM-006, and TTM-027 in NHPs
[0504] This example describes the transduction level, tropism, ability to cross the blood brain barrier, and overall spatial distribution in the central nervous system (CNS) and peripheral tissues of the AAV capsid variants TTM-002 (SEQ ID NO: 982 (amino acid) and SEQ ID NO: 984 (DNA), comprising SEQ ID NO: 2), TTM-001 (SEQ ID NO: 981 (amino acid) and SEQ ID NO: 983 (DNA), comprising SEQ ID NO: 941); TTM-003 (SEQ ID NO: 36 (amino acid) and SEQ ID NO: 12 (DNA), comprising SEQ ID NO: 3589), TTM-006 (SEQ ID NO: 39 (amino acid) and SEQ ID NO: 15 (DNA), comprising SEQ ID NO: 3241), and/or TTM-027 (SEQ ID NO: 4 (amino acid) and SEQ ID NO: 5 (DNA), comprising SEQ ID NO: 3272), relative to AAV9 following intravenous administration in African green monkeys (Chlorocebus sabaeus mannosets (Callithrix jacchus and/or cynomolgus macaques (Macaca fascicularis) .
A. Evaluation of TTM-002 in African Green Monkeys (Chlorocebus sabaeus)
[0505] AAV particles were generated with the TTM-002 capsid vanant or the AAV9 capsid control which comprised a self-complementary viral genome encoding an histone H2b protein with an HA tag driven by a ubiquitous CBA promoter. The .AAV particles comprising the TIM-002 capsid variant or the AAV9 capsid control were administered to the NHPs (n=2) intravenously at a dose of le 12 VG/kg or le 13 VG/kg. The in-life period was 28 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR and viral DNA (biodistribution) by ddPCR.
[0506] As shown m Table 25, the TTM-002 capsid variant resulted, in increased brain biodistribution in all brain regions investigated as compared to AAV9 at both doses tested. The TTM-002 capsid variant also led to increased transgene expression in the brain relative to AAV9 at both doses tested (Table 26). In the spinal cord, the TTM-002 capsid variant distributed, to the cervical spinal cord and the spinal cord ventral hom at a higher level relative to AAV9 (Table 25) and it mediated higher transgene expression than AAV9 in both the full spinal cord and the ventral horn (Table 26). When administered at a dose of lel.3
VG/kg, TTM-002 delivered 1-2 viral genomes (VGs) per cell on average across multiple brain areas, outperforming AAV9 by 4- to 24-fold, and was capable of expressing 16- to 186- fold more transgene RNA (Table 25 and Table 26), The TTM-002 capsid variant, exhibited lower biodistribution (Table 25) and transgene expression (Table 26) in the DRG relative to AAV9, indicating that TTM-002 capsid variant was detargeted in the DRG relative to AAV9. Similar expression and distribution were observed by immunohistochemistry performed on these CNS tissues. High-throughput analysis of immunohistochemistry stainings indicated that TTM-002 was capable of targeting upwards of 50% of cells in the brain (FIG. 4A), including both astrocytes and neurons (FIG. 4B). In contrast with the tropism in mice as provided in Example 9, TTM-002 demonstrated a bias towards Sox9(+) astrocytes over neurons, labeled with either NeuN or SMI31 1 .
[0507] Distribution and transgene expression was also measured in the peripheral tissues of the liver, heart, and quadriceps. In the liver, TTM-002 capsid variant exhibited lower biodistribution (Table 25) and transgene expression (Table 26) relative to AAV9, indicating that TTM-002 capsid variant was detargeted in the liver relative to AAV9. In the heart, the TTM-002 capsid variant exhibited comparable levels of biodistribution relative to AAV9 (Table 25), but increased transgene expression relative to AAV9 (Table 2.6). In the quadriceps, TTM-002. capsid variant exhibited lower biodistribution (Table 2.5) and lower transgene expression (Table 26), relative to AAV9. Similar expression and distribution were observed by immunohistochemistry performed on these peripheral tissues.
Table 25: Quantification of viral genome copies per diploid genome (biodistribution) by ddPCR following intravenous administration of AAV particles comprising a TTM-002 capsid
Table 26: Quantification of transgene mRNA by RT-qPCR following intravenous administration of AAV particles comprising a TTM-002 capsid
[0508] Taken together, these data demonstrate that TTM-002 is an enhanced CNS tropic capsid in NHPs (African green monkeys) that can infect non-neuronal cells. TTM-002 was also detargeted. in the DRG and liv er relative to AAV9, but showed increased transgene expression in the heart relative to AAV9. Additionally, the TTM-002 capsid variant was able to successfully penetrate the blood brain barrier following intravenous injection.
B, Evaluation of TTM-001 and TTM-002 in Marmosets (Callithrix jacchus)
[0509] AAV particles were generated with the TTM-002 capsid vanant, the TTM-001 capsid variant, or the AAV9 capsid control which comprised a self-complementary viral genome encoding a histone H2b protein with an MYC tag (TTM-002 capsid variant), His tag (TTM-001 capsid variant), or HA tag (AAV9 control capsid) driven by a ubiquitous CAG promoter. The AAV particles comprising the TTM-002 capsid variant, the TTM-001 capsid variant, or the AAV 9 capsid control were administered to the marmosets (Callithrix jacchus) (n™3) intravenously in a single solution, at the doses indicated in Table 51. The in-life period was 2.8 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR, protein expression by IHC, and viral DNA
(biodistribution) by ddPCR. Data, were then normalized to the dose of each viral vector in the dosing solution.
[0510] As shown m Table 52, both the TTM-001 and TTM-002 capsid vanants demonstrated increased biodistribution in the caudate and motor cortex in the brain of the marmosets relative to the AAV9 control. The TTM-001 and TTM-002 capsid variants also led to increased transgene expression (Table 53) in the caudate and motor cortex in the brain of the marmosets. In fact, biodistribution and transgene expression were increased over 100- 400 fold for both TTM-001 and TTM-002 in the brain relative to AAV9. TTM-002 delivered upwards of 280-fold more viral genomes and expressed 500-fold higher transgene RNA levels than AAV9 (Tables 52. and 53). Similar expression and distribution was observed by immunohistochemistry. More specifically, staining for TTM-001 and TTM-002 was detected in the mid-brain, caudate, putamen, thalamus, and cerebellum, and this staining was increased for both capsid variants in each of these brain tissues reiative to AAV9. Staining for TTM- 001 and TTM-002 was also observed m the molecular and granule layer of the cerebellum. [0511] Biodistribution and transgene expression were also measured in the peripheral tissues of the liver, heart, and quadriceps. In the liver, the TTM-002 capsid variant exhibited lower biodistribution (Table 52) and transgene expression (Table 53) reiative to AAV9, indicating that the TTM-002 capsid variant was detargeted in the liver relative to AAV9 in marmosets. The TTM-001 capsid variant demonstrated comparable biodistribution and transgene expression in the liver (Table 52 and Table 53) as well as comparable transgene expression in the heart and muscle (Table 53) relative to AAV9. Both TTM-001 and TTM- 002. led to decreased biodistribution (Table 52) relative to AAV9 in the heart and muscle, and TTM-002 also resulted in lower transgene expression in the heart and muscle relative to AAV9 (Table 53).
Table 52. Quantification of viral genome copies per diploid genome (biodistribution) by ddPCR following intravenous administration of AAV partides comprising a TTM-001
capsid or a TTM-002 capsid normalized to the actual titer of the viral vector in the dosing solution (vg/dg = viral genome copies/ diploid genome)
Table 53. Quantification of transgene mRNA by RT-qPCR following intravenous administration of .AAV particles comprising a TTM-001 capsid or a TTM-002 capsid normalized to the actual titer of the viral vector in the dosing solution (mRNA = transgene mRNA fold over housekeeping gene; rel. to AAV9= transgene mRNA fold
over housekeeping gene relative to AAV9)
These data, in marmosets for TTM-002 were similar to those observed in African green monkeys, further demonstrating cross-species compatibility of the TTM-002 capsid variant. Taken together, these data demonstrate that TIM-001 and TTM-002 are enhanced CMS tropic capsids in mannosets. TTM-002 was also detargeted in the liver, heart, and muscle relative to AAV9 in mannosets, where TTM-001 demonstrated comparable biodistribution and/or transgene expression in the liver, heart, and muscle compared to AAV9. Additionally, the TTM-001 and TTM-002 capsid variants were able to successfully penetrate the blood brain barrier following intravenous injection.
C. Evaluation of TTM-001, TTM-002, TTM-003. TTM-006, and TTM-027 in
Cynomolgus Macaques (Macaco fascicularis)
[0512] AAV particles were generated with the TTM-002 capsid, variant, the TTM-001 capsid variant, the TTM-003 capsid variant, the TTM-006 capsid variant, the TTM-027 capsid variant, or the AAV9 capsid control which comprised a self-complementary viral genome encoding a histone H2b protein driven by a ubiquitous CAG promoter. The AAV
particles comprising the TTM-002 capsid variant, the TTM-001 capsid variant, the TTM-027 capsid variant or the AAV9 capsid control were administered to a first group of male cynomolgus macaques M acaca fascicularis; 4-6 kg body weight; over 2 years old) intravenously in a single solution, at a total dose per group of 2e 13 V G/kg or a. dose per capsid of 4el2 VG/kg. The AAV particles comprising the TTM-003 capsid variant or the TTM-006 capsid variant were administered to a second group of male cynomolgus macaques (Macaca fascicularis', 4-6 kg body weight; over 2 years old) intravenously in a. single solution, at a total dose per group of 2el3 VG/kg or a. dose per capsid of 4el2 VG/kg. The in- life period was 28 days for both groups, and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR; protein expression by IHC/chromogenic staining (e.g., DAB staining for percent of DAB+ cells indicating the percent of cells transduced); percent positive cells (e.g., neurons, motor neurons, and astrocytes) in brain and spinal cord regions by immunofluorescence microscopy; and viral DNA (biodistribution) by ddPCR.
[0513] As shown in Table 54, TTM-001 , TTM-002, TTM-003, TTM-006, and TTM-027 demonstrated increased CNS transduction and/or biodistribution in several regions of the brain (greater than 30% of cells transduced observed in several regions for multiple capsid variants) and spinal cord of the cynomolgus macaques after intravenous administration at a relatively low' dose of 4el2 vg/kg. More specifically, TTM-003 was capable of transducing up to 40% of cells in the caudate, putamen, and cortex; TTM-027 was capable of transducing up to 30% of cells in the caudate, putamen, and. cortex; and. both showed improved delivery to the spinal cord relative to AAV9 and TTM-002, at a. dose of 4el2 vg/kg.
[0514] Cell-typing was also performed in the putamen, substantia nigra, and temporal cortex of the brain to measure the percent of neurons (NeuN+ cells) and astrocytes (Sox9-i- cells) that were transduced by the AAV particles comprising the TTM-003 and TTM-027 capsid variants or the AAV9 controls (Table 55). TTM-003 was capable of transducing up to 47.8% of neurons and 79.5% of astrocytes in the putamen; 25.3% of neurons and 87.5% of astrocytes in the temporal cortex; and 33.7% of neurons and. 18.6% of astrocytes in the substantia nigra (Table 55). TTM-027 was capable of transducing up to 27% of neurons and 41.8% of astrocytes in the putamen; 12.3% of neurons and 51.4% of astrocytes in the temporal cortex; and 21.1% of neurons and 12.2% of astrocytes in the substantia nigra (Table 55). Co-localization of TTM-027 and TTM-003 with motor neurons (ChAT+ cells) was also observed in the spinal cord by immunofluorescence microscopy (Table 55). Across the
lumbar, cervical, and thoracic spinal cord, TTM-003 was capable of transducing 78.5% of motor neurons and TTM-027 was capable of transducing 53.5% of motor neurons (Table 55).
[0515] In the peripheral tissues, all of the TTM-001, TTM-002, TTM-003, TTM-006, and.
TTM-027 capsid variants tested exhibited robust liver de-targeting relative to AAV9 (Table
56).
Table 54. Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR, transgene mRNA by RT-qPCR (mRNA = transgene mRNA fold over housekeeping gene), and percent of DAB+ cells in tissues of the CNS of cynomolgus macaques
Table 55. Quantification of neurons (%NeuN positive cells), motor neurons (%chAT positive cell) and/or astrocytes (% Sox9 cells) transduced with the TTM-003 and TTM- 027 capsid variants in the putamen, temporal cortex, substantia nigra, and spinal cord
Table 56. Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR, transgene mRNA by RT-qPCR (mRNA = transgene mRNA fold over housekeeping gene), and percent of DAB+ cells in liver and heart of cynomolgus macaques
[0516] Takers together, these data demonstrate that TTM-001, TTM-002, TTM-003, TTM- 006, and TTM-027 are enhanced CNS tropic capsids in cynomolgus macaques that were capable of crossing the blood brain barrier following intravenous injection, even at a low' dose of 4el 2 vg/kg. TTM-003 and TTM-027 were also capable of transducing both neurons and astrocytes in several brain tissues as well as motor neurons in the spinal cord. All capsids variants also demonstrated robust liver de-targeting relative to AAV9.
D. Individual Evaluation of TTM-003 in Cynomolgus Macaques (Macaca fascicularis} [0517] AAV particles were generated with the TTM-003 capsid variant which comprised a self-complementary viral genome encoding a histone H2b protein with an HA tag driven by a ubiquitous CAG promoter. The AAV particles comprising the TTM-003 capsid variant were administered to cynomolgus macaques (Macaca fascicularis) (n=3 male monkeys; 4-12 years of age) intravenously at a dose of 3el3 VG/kg. The in-life period, was 28 days and. then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-ddPCR, viral DNA (biodistribution) by ddPCR, and immunohistochemistry (IHC)/chromogenic quantification of percent positive cells in various tissues (cellular tropism).
[0518] As shown in Table 57, substantial and widespread transduction of TTM-003 was observed in the brain and spinal cord in NHPs following intra veno us administration of the AAV particles comprising the TTM-003 capsid variant. When administered at a dose of 3el3 VG/kg, TTM-003 was capable of transducing multiple cell types in the brain and spinal cord, including neurons and astrocytes, as quantified in Table 57. Distribution and transgene
expression was also measured in the peripheral tissues of liven heart, and muscle as provided in Table 58.
[0519] Taken together, these data demonstrate that TTM-003 is a. CNS-tropic capsid in
NHPs (cynomolgus macaques) that can infect both neuronal and non-neuronal cells.
Additionally, the TTM-003 capsid variant was able to successfully penetrate the blood brain barrier following intravenous injection.
Table 57: Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR; transgene mRNA expression by RT-ddPCR (mRNA = transgene mRNA expression relative to a housekeeping gene (mTBP)); and the percentage of cells transduced with the TTM-003 capsid variant measured by co- localized staining of HA (payload tag) and DAB, NeuN (neurons), or Sox9 (astrocytes) in the brain and spinal cord of NHPs (each value represents the average from 3 NHPs)
Table 58: Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR; transgene mRNA expression by RT-ddPCR (mRNA = transgene mRNA relative to a housekeeping gene (mTBP)); and percentage of cells transduced with the TTM-003 capsid variant measured by co-localized HA (payload tag) and DAB staining in the peripheral tissues of NHPs (each value represents the average of three individual NHPs)
E. Evaluation of TTM-027 in Cynomolgus Macaques (M acaca fascicularis)
[0520] AAV particles were generated with the TTM-027 capsid variant or the AAV 9 capsid control which comprised, a self-complementary viral genome encoding a histone H2b protein driven by a ubiquitous C-AG promoter. The AAV particles comprising the TTM-027 capsid variant or the AAV9 capsid control were administered to a group of male cynomolgus macaques (Macaca fascicularis; 8-9 kg body weight; 4-10 years old; n=3) intravenously in a single solution, at. a total dose per group of 2el3 VG/kg or a dose per capsid of 4e12 VG/kg. The in-life period was 28 days, and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-ddPCR; protein expression by IHC/chromogenic staining and quantification of percent positive cells; and viral DNA (biodistribution) by ddPCR.
[0521] As shown in Table 59, TTM-027 demonstrated increased CNS transduction in several brain regions and the spinal cord of the cynomolgus macaques after intravenous administration at a relatively low dose of 4e12 vg/kg.
Table 59. Quantification of percent DAB+ cells; viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR; and transgene mRNA expression by RT-ddPCR (mRNA = transgene mRNA. expression relative to a housekeeping gene (mTBP)) in tissues of the CNS of cynomolgus macaques
[0522] The percentage of DAB positive cells, biodistribution, and mRNA expression were also quantified in the peripheral tissues of the liver, heart, and muscle, which is provided in Table 60. The
TTM-027 capsid variant exhibited robust liver de-targeting relative to AAV9 (Table 60).
Table 60. Quantification of percent DAB+ cells; viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR; and transgene mRNA expression by RT-ddPCR (mRNA = transgene mRNA expression relative to a housekeeping gene (mTBP)) in the peripheral tissues of cynomolgus macaques
[0523] Taken together, these data demonstrate that TTM-027 is an enhanced CNS tropic capsid in cynomolgus macaques that was capable of crossing the blood brain barrier following intravenous injection, even at a low dose of 4e12 vg/kg, which is consistent with what was observed in Example 8C above.
F. Individual Evaluation of TTM-027 in Cynomolgus Macaques (Macaca fascicularis) [0524] AAV particles were generated with the TTM-027 capsid variant which each comprised a self-complementary viral genome encoding a histone H2b protein with an HA tag driven by a ubiquitous CAG promoter. The AAV particles comprising the TTM-027 capsid variant were administered to cynomolgus macaques (Macaca fascicularis) (n=3 male monkeys; 7.4-11 years of age) intravenously at a dose of 3el3 VG/kg. The in-life period was 28 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR. viral DNA (biodistribution) by ddPCR, and immunohistochemistry (IHC)Zchromogenic and irnmunofluorescent quantification of percent positive cells in various tissues (cellular tropism).
[0525] As shown in Table 61 substantial and widespread transduction of TTM-027 was observed in the brain and spinal cord of NHPs following intravenous administration of the AAV particles comprising the TTM-027 capsid variant. More specifically, TTM-027 demonstrated superior viral genome biodistribution in a variety of CNS tissues and regions, broader expression in the CNS as shown by both transgene mRNA expression and IHC (Table 61 and Table 62), as well as a highly neurotropic and astrocytic tropism in the brain and the spinal cord (Table 62 and Table 63). TIM-027 when administered intravenously at a dose of 3el 3 vg/kg was capable of transducing up to 21 -65% of neurons (HA. and NeuN positive cells) and 87-97% of astrocytes (FLA and Sox9 positive cells) in multiple brain regions; up to 96% of Purkinje Neurons in the cerebellum; up to 84-94% of motor neurons (HA and ChAT positive cells) in the spinal cord; and up to 93-97% of astrocytes (HA and. Sox9 positive cells) in the spinal cord (Table 63). TTM-027 was also able to transduce 97.9% of the dopaminergic neurons in the substantia nigra, as indicated by cells that were positive for both tyrosine hydroxylase (TH) and HA (pay load tag). The TTM-027 capsid variant was well tolerated, in the NHPs.
Table 61 : Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR; transgene mRNA expression by RT-qPCR (mRNA = transgene mRNA expression relative to a housekeeping gene (mTBP)); and the percentage of cells transduced with the TTM-027 capsid variant measured by co- localized staining of HA (payload tag) and DAB in the brain and spinal cord of NHPs (each value represents the average from 3 NHPs; for regions of the DRG, sensory neuron data are shown)
Table 62: Quantification of the percentage of cells transduced with the TTM-027 capsid variant measured by co-localized staining of HA (payload tag) and either NeuN (neurons) or Sox9 (astrocytes) in the brain of NHPs (each value represents the average from 3 NHPs)
Table 63: Quantification of the percentage of cells transduced with the TTM-027 capsid variant measured by co-localized staining of HA (payload tag) and either ChAT (motor neurons) or Sox9 (astrocytes) in the gray matter of the spinal cord of NHPs (each value represents the average from 3 NHPs)
[0526] The biodistribution and mRNA expression following intravenous administration of
AAV particle comprising the TTM-027 capsid variant at a dose of 3e 13 vg/kg was also measured in the peripheral tissues of the liver, heart, and muscle (vastus lateralis and
gastrocnemius) as provided in Table 64). TTM-027 showed very low biodistribution and mRNA expression in the liver in NHPs (Table 64), and demonstrated substantially reduced liver tropism.
Table 64: Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR, and transgene mRNA expression relative to a housekeeping gene (mTBP) by RT-ddPCR in the peripheral tissues of NHPs (each value represents the average from 3 NHPs)
]0527] Taken together, the individual characterization of the TTM-027 capsid variant further demonstrates and confirms that the TTM-027 is an enhanced CNS tropic capsid m cynomolgus macaques, capable of crossing the blood brain barrier following intravenous injection, consistent with what was observed in Examples 8C and 8E above.
Example 9, Dose Response Evaluation of the TTM-002 and TTM-027 AAV capsid variants
[0528] This Example investigates transduction of the TTM-002 (SEQ ID NO: 982 (amino acid) and SEQ ID NO: 984 (DNA), comprising SEQ ID NO: 2) and TTM-027 (SEQ ID NO: 4 (amino acid) and SEQ ID NO: 5 (DNA), comprising SEQ ID NO: 32.72) capsid variants following intravenous administration at increasing doses in mice.
[0529] AAV particles were generated with the TTM-002 capsid variant or the TTM-027 capsid variant which comprised a single stranded viral genome encoding a histone protein with an HA tag (H3F3-HA) and a woodchuck hepatitis virus posttranscriptional regulatory element (WERE) driven by a ubiquitous CBA promoter. The AAV particles comprising the TTM-002 capsid variant or the TTM-027 AAV capsid variant were administered to mice (11=3 mice per dosing group; Balb/c; 6-8 weeks of age) via tail vein injection at increasing doses of 1e12 vg/kg, 3.2e12 vg/kg, 1e13 vg/kg, 3.2e13 vg/kg, or 1e 14 vg/kg, The dose of 3.2e 12 vg/kg was approximately equivalent to the dose used per capsid in the cynomolgus macaques (Macaca fas cicu lari s) in Example 8C above. The in-life period was 28 days and CNS tissues were collected for measuring transgene mRNA (expression) by qPCR and the percent of HA positive cells in the brain.
[0530] As shown in Table 65 and FIG. 3A, a dose dependent increase in transduction of both the TTM-002 and TTM-027 capsid variants was observed in the mouse cortex foilowing intravenous administration of the AAV particles at the increasing doses. The percent of cells transduced in the mouse cortex with TTM-027 was higher at all doses compared to TTM- 002. At the highest dose tested, 1e14 vg/kg, TTM-027 transduced 65% of cells in the cortex, whereas TTM-002 transduced 38% of cells, with TIM-002 demonstrating an even distribution between neurons and astrocytes, identified by NeuN and Sox9 markers, respectively (Table 66). It was also observed that an increase in dose from 3 ,2e 12 to 3 ,2e 13 vg/kg resulted in a greater than 3 fold increase in percent positive ceils. Consistent with the percentage of TTM-002 or TTM-027 positive cells, a dose dependent increase in transgene mRNA expression was also observed in the mouse brain following intravenous administration of the TTM-002 and TTM-027 capsid variants at the increasing doses (FIG. 3B and Table 65).
[0531] Measurements in Table 65 are by co-localization of chromogenic HA staining and hematoxylin, as well as quantification by qPCR of transgene mRNA expression relative to a housekeeping gene (mTBP) in the mouse brain, following intravenous administration (each val ue is the average of three individual measurements within each cortex taken from three mice). Measurements in Table 66 are in the mouse cortex by fluorescence microscopy and co-staining with HA and cell-type specific markers (each value is the average of three individual measurements within each cortex taken from three mice).
[0532] Taken together, these data demonstrate a dose-dependent response across a 2 -log dose range in mouse ( 1e 12 to 1e 14 vg/kg), without reaching saturation at the maximal dose (Table 65 and Table 66).
Table 65. Quantification of cells positive for the TTM-002 or TTM-027 capsid variant per mm2 (HA positive cells/mm2) or percentage of cells transduced with the TTM-002 or TTM-027 capsid variants (% HA positive cells) in the mouse cortex
Table 66. Quantification of total cells (HA positive cells/mm2), neurons (NeuN positive cells that are also HA positive/mm2), and astrocytes (Sox9 positive cells that are also HA positive/mm2) positive for the TTM-002 capsid variant per mm2 or percentage of total cells (%HA positive cells), neurons (% NeuN positive cells that are also HA positive), and astrocytes (% Sox9 positive cells that are also HA positive) transduced with the TTM-002 capsid variant
Example 10: Individual Characterization of TTM-002 and TTM-001 Capsid Variants in
Mice
[0533] The goal of these experiments was to determine the transduction level, tropism. ability to cross the blood brain barrier, and overall spatial distribution in the brain, heart, and liver of the TTM-001 and TTM-002 capsids and variants thereof relative to AAV9 following intravenous injection in mice. TTM-001 (SEQ ID NO: 981), TTM-002 (SEQ ID NO: 982), and variants of the TTM-002 and TTM-001 capsids comprising local modifications in loop
IV were investigated including TTM-003 (SEQ ID NO: 36), TTM-006 (SEQ ID NO: 39),
TTM-018 (SEQ ID NO: 51), and TTM-019 (SEQ ID NO: 52). The amino acid sequences for these capsid variants are provided, e.g., in Table 4.
[0534] AAV particles were generated with each of these capsid variants encapsulating a. fluorescent reporter construct, ZsGreen-HA, driven by a. CAG promoter. Each capsid variant and AAV9 control were tested by intravenously administering by tail vein injection, the .AAV particle formulation at le 13 VG/kg to three BALB/c mice. The in-life period was 28 days
and then various CNS and peripheral tissues were collected for measuring transgene mRNA expression.
[0535] The brains isolated from mice injected with the AAV particles encapsulated in the TTM-002 capsid, the TTM-001 capsid, or variants of the TTM-001 or TTM-002 capsid comprising local modifications were assayed to calculate ZsGreen expression and/or transgene DNA. Data were provided as fold over the AAV9 control (Table 67). All of the variants of the TTM-001 and TTM-002 capsids as well as TTM-001 and TTM-002 demonstrated increased CNS tropism and expression in the brain relative to AAV9 (Table 67; FIGs. 5A-5B). More specifically, TTM-001 and TTM-002 showed a broad distribution throughout the entire brain and spinal cord, outperforming the AAV9 control by approximately 30- and 40-fold, respectively, in terms of viral DNA biodistribution and transgene RNA expression (FIGs. 5A-5B; Table 67). The variants of the TTM-001 and TTM-002 capsids as well as TTM-001 and TTM-002 also demonstrated reduced mRNA and DNA expression in the liver by qPCR relative to the AA V9 control, with TTM-002. showing 14-fold lower gene expression than the AAV 9 control (Table 67; FIGs. 5C-5D). Similar results were observed by immunohistochemistry (IHC) staining of the brain (including the cortex, thalamus, and cerebellum), spinal cord (grey matter), and liver for transduction by AAV particles comprising the AAV capsid variants investigated. Transduction of the heart, skeletal muscle, and kidney did not show major differences between AAV9 and TTM-002.
Table 67. ZsGreen Expression and Transgene DNA and/or RNA expression for variants of the TTM-001 and TTM-002 capsids relative to AAV9 in mice
Brain Liver
Capsid ZsGreen Transgene DNA Transgene RNA Transgene DNA fold Variant Expression fold fold change change relative to ibid change change relative to relative to
AAV9 AAV9 relative to AAV9 AAV9
AAV9 1.0 1.0 LOGO 1.000
TTM- 001 32.3 31.9 0.337 0.348
TTM-
002 40.9 37.0 0.072 0.034
TTM-
006 16.1 36.9 0.036 0.013
TTM- 018 22.8 27.0 0.231 0.223
TTM-
019 14.6 14.4 0.117 0.112
TTM- 003 17.3 14.6 0.015 0.026
Example 11 : In vivo Evaluation of TTM-002 AAV Capsid Variant Comprising an HA- tagged Nucleotide Sequence Encoding GBA in Cynomolgus Monkeys
[0536] This Example investigates the distribution and efficacy of an AAV comprising a TTM-002 AAV capsid variant (SEQ ID NO: 982) and a codon-optimized nucleotide sequence (SEQ ID NO: 1773) encoding a GBA and an HA-tag (TTM-002.GBA VG17-HA). Mauritius male cynomolgus monkeys (Macaca Jascicularis; 3-6 years of age; 3-8 kg in weight; n= 3 per group) were administered TTM-002.GBA_VG17-HA at 3el2 vg/kg or 1e 13 vg/kg by intravenous administration. Monkeys were also administered a vehicle control (modified PBS) or 1e13 vg/kg of an AAV9 capsid comprising the same payload construct was used as control (AAV9.GBA__VG17-HA) for comparison. Biodistribution of GBA 1-HA protein was assessed following a 28-day treatment period. Primary readouts included biochemical analysis of viral genomes and mRNA, immunohistochemical analysis of HA levels, and histopathology readouts in the brain, spinal cord, liver, kidney, and heart tissues.
Secondary-' readouts included biochemical analysis of GCase activity, LC/MS-MS analysis of GBA 1 levels, and cage-side observations including weekly body weight.
[0537] Histopathology analysis revealed no signs of toxicity in the kidneys, hearts, brains, and spinal cords of the treated monkeys at both AAV dosages.
[0538] TTM-002. GBA VG17-HA led to increased viral genome biodistribution in all
CNS tissues (FIG, 1; Table 46A-H) and reduced liver transduction as compared to AAV9.GBA VG17-HA.
Table 46D: Viral genome biodistribution in the substantia nigra
Table 46E: Viral genome biodistribution in the C3 Ventral Horn
[0539] TTM-002.GBA VG17-IIA led to increased human GBA 1 mRNA expression over the expression of the TBP housekeeping gene in the dentate nucleus, putamen, substantia nigra, motor and frontal cortex, and spinal cord (Tables 47A-E). Compared to animals treated with AAV9.GBA_VG17-HA, comparable levels of human GBA1 mRNA expression were observed in the DRG, liver, or heart of animals treated with TTM-002.GBA VG17-HA
(Tables 47F-H).
Table 47A: GBA1 mRNA expression relative to TBP in the frontal cortex
Table 47B: GBA1 mRNA expression relative to TBP in the motor cortex
[0540] As compared to AAV9.GBA_VG17-HA, TTM-002.GBA_VGl 7-HA led to increased human GBA1 mRNA in the dentate nucleus, cervical spinal cord, frontal cortex, motor cortex, putamen, and substantia nigra (FIG. 2; Tables 48A-E). Compared to animals treated with AAV9.GBA_VG17-HA, comparable levels of human GBA 1 mRNA expression were observed in the DRG, liver, or heart of animals treated with TTM-002.GBA VG17-HA relative to endogenous cynomolgus GBA1 (Tables 48F-H).
Table 48A: Human GBA1 mRNA expression relative to endogenous cynomolgus GBA1 mRNA in the frontal cortex
Table 48B: Human GBA1 mRNA expression relative to endogenous cynomoigus GBA1 mRNA in the motor cortex
Table 48D: Unman GBA1 mRNA expression relative to endogenous cynomoigus GBA1 mRNA in the substantia nigra
Table 48E: Human GBA1 mRNA expression relative to endogenous eynomoigus GBA1 mRNA in the spinal cord
[0541] Immunohistochemical analysis showed that TTM-002.GBA VG17-HA led to greater GBA 1-HA levels in the brain as compared to AAV9 (Table 49).
Table 49: HA chromogenic IHC quantitative image analysis following
[0542] Co-staining analysis showed that HA protein expression was detected in both neurons and astrocytes of the putamen and substantia nigra of subjects treated with
TTM002.GBA_VG17-HA but not in subjects treated with AAV9.GBA_VG17-HA or vehicle (Table 50).
Table 50: Astrocyte co-staining analysis following TTM002.GBA_VG17-HA or
IX. Equivalents and Scope
[0543] Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments in accordance with the Detailed Description provided herein. The scope of the present disclosure is not intended to be limited to the above Detailed Description, but rather is as set forth in the appended claims.
[0544] Where ranges are given, endpoints are included. Furthermore, it is to be understood that unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise. [0545] In addition, it is to be understood that any particular embodiment of the present disclosure that falls within the prior art may be explicitly excluded from any one or more of the claims. Since such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the compositions of the disclosure (e.g, , any, composition, therapeutic or active ingredient; any method of production; any method of use; etc?) can be excluded from any one or more claims, for any reason, whether or not related to the existence of prior art.
[0546] It is to be understood that the words which have been used are w ords of description rather than limitation, and that changes may be made within the purview of the appended claims without departing from the true scope and spirit of the disclosure m its broader aspects.
[0547] While the present disclosure has been described at some length and with some particularity with respect to the several described embodiments, it is not intended that it should be limited to any such particulars or embodiments or any particular embodiment, but it is to be construed with references to the appended claims so as to provide the broadest possible interpretation of such claims in view of the prior art and, therefore, to effectively encompass the intended scope of the disclosure.
[0548] All publications, patent applications, patents, and other references mentioned herein are incorporated by reference in their entirety. In case of conflict, the present specification, including definitions, will control. In addition, section headings, the materials, methods, and. examples are illustrative only and not intended to be limiting.
Claims
1. An adeno-associated virus (AAV) particle comprising: a) an AAV capsid variant comprising an amino acid sequence having the following formula:
[N1]-[N2]-[N3], wherein:
(i) optionally [N1] comprises XL _X2, and X3, wherein at least one of X1, X2, or X3 is G;
(ii) [N2] comprises the amino acid sequence of SPH; and
(iii) [N3] comprises X4, X5, and. X6, wherein at least one of X4, X5, or X6 is a basic amino acid; and b) a viral genome comprising a nucleic acid sequence encoding a modulatory polynucleotide for reducing or eliminating expression of At.axin-2 (ATXN2), optionally wherein the modulatory polynucleotide comprises an RNAi agent targeting ATXN2 mRNA.
2. The AAV particle of claim 1, wherein the amino acid sequence [N1]-[N2]-[N3] is in hypervariable loop IV of the AAV capsid, variant.
3. The AAV particle of claim 1 or claim 2, wherein the AAV capsid variant is an AAV9 capsid variant.
4. The AAV particle of any one of claims 1-3, wherein [N1] comprises X1, X2, and X3, wherein at least one of X1, X2, or X3 is G.
5. The AAV particle of any one of claims 1 -4, wherein [N1]-[N2]-[N3] is present immediately subsequent to a position corresponding to the amino acid position 452 of SEQ ID NO: 982; and wherein the AAV capsid variant comprises an amino acid sequence at least 90% identical, e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 982, e.g., to positions 203-742 of SEQ ID NO: 982.
6. The AAV particle of any one of claims 1-5, wherein [N1] comprises GHD.
7. The AAV particle of any one of claims 1-6, wherein [N1] comprises the amino acid G at a position corresponding to position 453, the amino acid H at position 454, and the amino acid. D at. position 455 of SEQ ID NO: 138 or SEQ ID NO: 982.
8. The AAV particle of any one of claims 1-7, wherein [M3] comprises KSG.
9. The AAV particle of any one of claims 1-8, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to SEQ ID NO: 982;
(ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to positions 138-742 SEQ ID NO: 982; or
(iii) a VP3 protein compri sing the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 982.
10. The AAV particle of any one of claims 1 -9, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to SEQ ID NO: 982;
(ii) a. VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 982; or
(iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid, sequence having at least. 95% identity to positions 203-742 of SEQ ID NO: 982.
1 1 . The AAV particle of any one of claims 1-10, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to SEQ ID NO: 982;
(ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid, sequence having at least 99% identity to positions 138-742 SEQ ID NO: 982; or
(iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 982.
12. The AAV particle of any one of claims 1-11, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982;
(ii) a. VP2 protein comprising the amino acid, sequence of positions 138-742 of SEQ ID NO: 982; or
(iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982.
13. The AAV particle of any one of claims 1-4, wherein [N2]-[N3] comprises the amino acid sequence of SPHSKA (SEQ ID NO: 941).
14. An adeno-associated virus (AAV) particle comprising (a) a viral genome comprising a nucleic acid sequence encoding a modulatory polynucleotide for reducing or eliminating expression of Ataxin-2 (ATXN2), and (b) an AAV 9 capsid variant comprising the amino acid sequence of SPHSKA (SEQ ID NO: 941).
15. The AAV particle of claim 14, wherein tire amino acid sequence of SPHSKA (SEQ ID NO: 941) is in hypervariabie loop IV of the AAV capsid variant.
16. The AAV particle of claim 14 or claim 15, wherein the amino acid sequence of SPHSKA (SEQ ID NO: 941) is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO: 36 or SEQ ID NO: 4.
17. The AAV particle of claim 16, wherein tire AAV9 capsid variant further comprises one, two, or all of: an E at an amino acid position corresponding to position 451 , an R at an amino acid position corresponding to position 452, and/or a V at an amino acid position corresponding to position 453 of SEQ ID NO: 36.
18. The AAV particle of any one of claims 14-17, wherein the A AV 9 capsid variant comprises the amino acid sequence of KTERVSGSPHSKAQNQQT (SEQ ID NO: 3589).
19. The AAV particle of any one of claims 14-1.8, wherein the AAV9 capsid variant comprises:
(i) a VP1 protein comprising an amino acid sequence having at least 90% identity to SEQ ID NO: 36;
(ii) a VP2 protein comprising an amino acid sequence having at least 90% identity to positions 138-742 SEQ ID NO: 36; and/or
(iii) a VP3 protein comprising an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 36.
20. The AAV particle of any one of claims 14-19, wherein the A AV9 capsid variant composes:
(i) a VP1 protein comprising an amino acid sequence having at least 95% identity to SEQ ID NO: 36;
(ii) a. VP2 protein comprising an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 36; and/or
(iii) a VP3 protein comprising an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 36.
21. The AAV particle of any one of claims 14-20, wherein the AAV9 capsid variant comprises:
(i) a VP1 protein comprising an amino acid, sequence having at least 99% identity to SEQ ID NO: 36;
(ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to positions 138-742 of SEQ ID NO: 36; and/or
(iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 36.
22. The AAV particle of any one of claims 14-21 , wherein the AAV9 capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 36;
(ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ
ID NO: 36; and/or
(iii) a. VP3 protein comprising the amino acid sequence of posi tions 203-742 of SEQ
ID NO: 36.
23. The AAV particle of any one of claims 14-21, wherein the AAV9 capsid variant compnses:
(i) the amino acid sequence SPHSKA (SEQ ID NO: 941 ), wherein the amino acid sequence is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO: 36;
(ii) an E at an amino acid position corresponding to position 451 , an R at an amino acid position corresponding to position 452, and a. V at an amino acid position corresponding to position 453 of SEQ ID NO: 36; and
(iii) no other modifications relative to wild, type AA.V9.
24. The AAV particle of any one of claims 14-16, wherein the AAV9 capsid variant further comprises one, two, or all of: an N at an amino acid position corresponding to position 452, an E at an amino acid, position corresponding to position 451, and/or a V at an amino acid position corresponding to position 453 of SEQ ID NO: 4.
25. The AAV particle of claim 23 or claim 24, wherein the AAV9 capsid variant comprises the amino acid sequence of KTENVSGSPHSKAQNQQT (SEQ ID NO: 3272).
26. The AAV particle of any one of claims 23-25, wherein the AAV9 capsid variant compnses:
(i) a VP1 protein comprising an amino acid sequence having at least 90% identity to SEQ ID NO: 4;
(ii) a VP2 protein comprising an amino acid sequence having at least 90% identity to positions 138-742 of SEQ ID NO: 4; and/or
(iii) a. VP3 protein comprising an amino acid sequence having at least 90% identity to positions 203-742 of SEQ ID NO: 4.
27. The AAV particle of any one of claims 23-26, wherein the AAV9 capsid variant compnses:
(1) a VP1 protein comprising an amino acid sequence having at least 95% identity to SEQ ID NO: 4:
(ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to positions 138-742 SEQ ID NO: 4; and/or
(iii) a VP3 protein comprising an amino acid sequence having at least 95% identity to positions 203-742 of SEQ ID NO: 4.
28. The AAV particle of any one of claims 23-27, wherein the AAV9 capsid variant comprises:
(i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 4;
(ii) a. VP2 protein comprising an amino acid sequence having at least 99% identity to positions 138-742 of SEQ ID NO: 4; and/or
(iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to positions 203-742 of SEQ ID NO: 4.
29. The AAV particle of any one of claims 23-28, wherein the AAV9 capsid variant composes:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 4;
(ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ
ID NO: 4: and/or
(iii) a VP3 protein comprising the amino acid sequence of positions 203-742. of SEQ ID NO: 4.
30. The AAV particle of any one of claims 23-28, wherein the A AV 9 capsid variant comprises:
(i) the amino acid sequence of SPHSKA (SEQ ID NO: 941), wherein the amino acid sequence is present immediately subsequent to an amino acid position corresponding to position 455 of SEQ ID NO: 4;
(ii) an E at an amino acid position corresponding to position 451 and a V at an amino acid position corresponding to position 453 of SEQ ID NO: 4; and
(iii) no other modifications relative to wild type AAV9.
31. The AAV particle of any one of claims 1-30, wherein the modulatory polynucleotide compnses:
(a) a 5’ flanking region comprising any one of SEQ ID NOs: 6413-6416 or a nucleotide sequence at least 95% identical thereto;
(b) a loop region comprising any one of SEQ ID NOs: 6417-6421 or a nucleotide sequence at least 95% identical thereto: and
(c) a 3’ flanking region comprising any one of SEQ ID NOs: 6422-6427 or a nucleotide sequence at least 95% identical thereto.
32. The AAV particle of claim 31, wherein:
(a) the 5’ flanking region composes SEQ ID NO: 6414 or SEQ ID NO: 6415, or a nucleotide sequence at least 95% identical thereto;
(b) the loop region comprises SEQ ID NO: 6417, SEQ ID NO: 6418, or SEQ ID NO: 6421, or a nucleotide sequence at least 95% identical thereto; and
(c) the 3’ flanking region comprises SEQ ID NO: 6423, SEQ ID NO: 6424, or SEQ ID NO: 6425, or a nucleotide sequence at least 95% identical thereto.
33. The AAV particle of claim 31, wherein the 5’ flanking region comprises SEQ ID NO: 6414 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6417 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO: 6423 or a nucleotide sequence at least 95% identical thereto.
34. The AAV particle of claim 31, wherein the 5’ flanking region comprises SEQ ID NO: 6415 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6421 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO: 6425 or a. nucleotide sequence at least 95% identical thereto.
35. The AAV particle of claim 31, wherein the 5’ flanking region comprises SEQ ID NO: 6414 or a. nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6417 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO: 6424 or a nucleotide sequence at least 95% identical thereto.
36. The AAV particle of claim 31, wherein the 5' flanking region comprises SEQ ID NO: 6414 or a nucleotide sequence at least 95% identical thereto, the loop region comprises SEQ ID NO: 6418 or a nucleotide sequence at least 95% identical thereto, and the 3’ flanking region comprises SEQ ID NO: 6423 or a nucleotide sequence at least 95% identical thereto.
37. The AAV particle of any one of claims 1-36, wherein the modulatory polynucleotide comprises siRNA or shRNA.
38. The AAV particle of any one of claims 1 -37, wherein the modulatory polynucleotide further comprises a passenger strand and a guide strand, wherein the guide strand binds to and reduces or eliminates expression of one or more ATXN2 mRNA transcripts, where in the modulatory polynucleotide comprises from 5' to 3': the 5' flanking region, the passenger strand, the loop region, the guide strand; and the 3' flanking region.
39. The A AV particle of any one of claims 1-37, wherein the modulator}' polynucleotide further comprises a passenger strand and a guide strand, wherein the guide strand binds to and reduces or eliminates expression of one or more ATXN2 mRNA transcripts, wherein the modulatory polynucleotide comprises from 5' to 3': the 5* flanking region, the guide strand, the loop region, the passenger strand; and the 3' flanking region.
40. The AAV particle of claim 38 or claim 39, wherein the passenger strand is 15-30 nucleotides in length.
41. The AAV particle of any one of claims 38-40, wherein the guide strand is 15-30 nucleotides in length.
42. The AAV particle of claim 41, wherein the guide strand is 21-25 nucleotides in length and/or the passenger strand is 21-25 nucleotides in length.
43. The AAV particle of any one of claims 38-42, wherein the passenger strand is at least 70%, at least 80%, at least 90%, at least 95% or is 100% complementary to the guide strand.
44. The AAV particle of any one of claims 38-43, wherein the one or more ATXN2 mRNA transcripts comprises SEQ ID NO: 6428, SEQ ID NO: 6429, SEQ ID NO: 6430, and/or SEQ ID NO: 6431, or a trinucleotide repeat expansion thereof.
45. The A AV particle of any one of claims 1-44, wherein the viral genome comprises a. promoter operably linked to the nucleic acid sequence encoding the modulator}- polynucleotide.
46. The AAV particle of any one of claims 1-45, wherein the viral genome further comprises an inverted terminal repeat (ITR) sequence.
47. The AAV particle of claim 46, wherein the viral genome comprises an ITR sequence positioned 5’ relative to the nucleic acid sequence encoding the modulatory polynucleotide.
48. The AAV particle of claim 46 or claim 47, wherein the viral genome comprises an ITR sequence positioned 3’ relative to the nucleic acid sequence encoding the modulatory polynucleotide.
49. The AAV particle of any of claims 46-48, wherein the viral genome comprises an ITR sequence positioned 5 ’ relative to the nucleic acid sequence encoding the modulatory polynucleotide, and an ITR sequence positioned 3 ’ relati ve to the nucleic acid sequence encoding the modulatory polynucleotide.
50. A cell comprising the AAV particle of any one of claims 1-49, optionally wherein the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g,, an Sf9 cell), or a bacterial cell.
51 . A method of making the AA V particle of any one of claims 1-49, the method comprising
(i) providing a. cell comprising the viral genome comprising a nucleic acid encoding a. modulatory polynucleotide for reducing or eliminating expression of ATXN2 mRNA; and
(li) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid vanant; thereby making the AAV particle.
52. The method of claim 51, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 982;
(ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at
least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 138-742 SEQ ID NO: 982; and/or
(lii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 203-742 of SEQ ID NO: 982.
53. The method of claim 51, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 36;
(ii) a VP2 protein comprising the amino acid sequence oppositions 138-742 of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 138-742 SEQ ID NO: 36; and/or
(iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 36 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 203-742 of SEQ ID NO: 36.
54. The method of claim 51, wherein the AAV capsid variant comprises:
(i) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to SEQ ID NO: 4;
(ii) a VP2 protein comprising the amino acid sequence of positions 138-742 of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91 %, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 138-742 SEQ ID NO: 4; or
(iii) a VP3 protein comprising the amino acid sequence of positions 203-742 of SEQ ID NO: 4 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) to positions 203-742 of SEQ ID NO: 4.
55. The method of any one of claims 51-54, further comprising, prior to step (i), introducing a first nucleic acid molecule comprising the viral genome into the cell .
56. The method of any one of claims 51-55, wherein the ceil comprises a second nucleic acid molecule encoding the AAV capsid variant.
57. The method of claim 56, further comprising, prior to step (i), introducing the second nucleic acid molecule into the cell.
58. The method of any one of claims 51 -57, wherein the cell comprises a mammalian cell, e.g., an HEK293 cell, an insect cell, e.g., an Sf9 cell, or a bacterial cell.
59. A pharmaceutical composition comprising the AAV particle of any one of claims 1-49, and a. pharmaceutically acceptable excipient.
60. A pharmaceutical composition comprising the AAV particle of any one of claims 13-30 and a pharmaceutically acceptable excipient.
61. A pharmaceutical composition comprising the AAV particle of any one of claims 17-23 and a pharmaceutically acceptable excipient.
62. A pharmaceutical composition comprising the AAV particle of any one of claims 24-30 and a pharmaceutically acceptable excipient.
63. A method of delivering a modulatory polynucleotide for reducing or eliminating expression of ATXN2 to a subject comprising administering to the subject an effective amount of the pharmaceutical composition of any one of claims 58-61 or the AAV particle of any one of claims 1 -49, thereby delivering the modulatory polynucleotide.
64. lire method of claim 63, wherein the method reduces or eliminates ATXN2 protein in tire subject.
65. The method of claim 63 or claim 64, wherein the subject has, has been diagnosed with having, or is at risk of having an ATXN2-related disorder.
66. The method of claim 65, wherein the ATXN2-related disorder is spinocerebellar ataxia type 2 (SCA2).
67. A method of treating an ATXN2-related disorder in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of any one of claims 59-62 or the AAV particle of any one of claims I -49, thereby treating the ATXN2-related disorder.
68. A method of treating an ATXN2-related disorder in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 60 or the AAV particle of any one of claims 13-30, thereby treating the ATXN2-related disorder.
69. A method of treating an ATXN2-related disorder in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 61 or the AAV particle of any one of claim s 17-23, thereby treating the ATXN2 -related, disorder.
70. A method of treating an ATXN2-related disorder in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 62 or the AAV particle of any one of claims 24-30, thereby treating the ATXN2-related disorder.
71. The method of any one of claims 67-70, wherein the subject has, has been diagnosed, or is at risk of having the ATXN2-related disorder.
72. The method of any one of claims 63-71, wherein the subject has one or more mutations in the ATXN2 gene.
73. The method of claim 72, wherein the one or more mutations in the ATXN2 gene comprises a trinucleotide repeat expansion.
74. The method of claim 73, wherein the trinucleotide repeat expansion in the ATXN2 gene comprises 32 or more CAG repeats.
75. The method of any one of claims 67-74, wherein the treating results in prevention of progression of the A TXN2 -related. disorder in the subject.
76. The method of claim 67-75, wherein the treating results in amelioration of at least one symptom of the ATXN2-related disorder and/or a change in at least one biomarker of the ATXN2-related disorder in the subject.
77. The method of claim 76, wherein the at least one symptom comprises progressive cerebellar ataxia, nystagmus, slow saccadic eye movements, ophthalmoparesis, parkinsonism, or a. combination thereof.
78. The method of any one of claims 67-77, wherein the ATXN2-related disorder is spinocerebellar ataxia type 2 (SCA2).
79. A method of treating spinocerebellar ataxia type 2 (SCA2) in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of anyone of claims 59-62 or the AAV particle of any one of claims 1-49, thereby treating SCA2,
80. A method of treating spinocerebellar ataxia type 2 (SCA2) in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 60 or the AAV particle of any one of claims 13-30, thereby treating SCA2.
81. A method of treating spinocerebellar ataxia type 2 (SCA2) in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 61 or the AAV particle of any one of claims 17-23, thereby treating SCA2.
82. A method of treating spinocerebellar ataxia type 2 (SCA2.) in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 62 or the AAV particle of any one of chums 24-30, thereby treating SCA2.
83. The method of any one of claims 79-82, wherein the subject has, has been diagnosed with having, or is at risk of having SCA2.
84. The method of any one of claims 63-83, wherein the subject is a human.
85. The method of any one of claims 63-84, wherein the A AV particle or the pharmaceutical composition is delivered to a cell or tissue of the CNS, optionally wherein the AAV particle or the pharmaceutical composition is delivered via intravenous administration.
86. The method of claim 85, wherein the cell or tissue of the CNS is a cell or tissue of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region).
87. The method of any one of claims 63-86, further comprising evaluating, e.g., measuring, the level of modulatory polynucleotide expression, and/or the level of ATXN2 expression, e.g,, ATXN2 gene, ATXN2 mRNA, and/or ATXN2 protein expression, in the subject, e.g,, in a cell, tissue, or fluid, of the subject.
88. The method of claim 87, wherein the level of ATXN2 protein is measured by an ELISA, a Western blot, or an immunohistochemistry' assay.
89. lire method of claim 87 or claim 88, wherein the evaluating of the subject’s level of modulatory polynucleotide expression and/or level of ATXN2 expression is performed prior to and/or subsequent to administration of the pharmaceutical composition or AAV particle, optionally wherein the subject’s level of modulatory polynucleotide expression and/or the subject’s level of ATXN2 expression prior to administration is compared to the subject’s level of modulatory polynucleotide expression and/or the subject’s level of ATXN2 expression subsequent to administration.
90. The method of any one of claims 63-89, wherein the subject’s level of ATXN2 protein expression subsequent to administration is decreased relative to the subject’s level of ATXN2 protein expression prior to administration.
91 . The method of any one of claims 63-90, further comprising evaluating, e.g., measuring, the level of modulatory polynucleotide activity and/or ATXN2 activity in the subject, e.g,, in a cell or tissue of the subject.
92. The method of any one of claims 63-91, wherein the administration results in:
(i) a decrease in ATXN2 protein expression in a cell, tissue (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region)), and/or fluid (e.g., CSF and/or serum) of the subject relative to baseline and/or relative to ATXN2 protein expression in a cell, tissue, or fluid of an individual with an ATXN2-related disorder who has not been administered the pharmaceutical composition or AAV particle;
(ii) an increase in the number and/or level of viral genomes (VG) per cell i n a CNS tissue (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary- complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or
(iii) a decrease in ATXN2 mRNA expression in a cell or tissue (e.g., a cell or tissue of the CNS, e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguous, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region) of the subject relative to baseline and/or relative to
ATXN2 mRNA expression in a cell or tissue of an individual with an ATXN2-related disorder who has not been administered the pharmaceutical composition or AAV particle.
93. The method of any one of claims 63-92, further comprising administering to the subject at least one additional therapeutic agent and/or therapy.
94. The method of claim 93, wherein the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy tor treating the ATXN2-related disorder.
95. The method of claim 93 or claim 94, wherein the at least one additional therapeutic agent and/or therapy comprises one or more of; growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2 -related disorders .
96. The method of claim 94 or claim 93, wherein the ATXN2 -related disorder is SCA2, amyotrophic lateral sclerosis (ALS), Parkinson’s disease (PD), or frontotemporal lobar d egene rati on (FTLD) .
97. The method of any one of claims 94-96, wherein the ATXN2 -related disorder is SCA2.
98. The method of any one of claims 63-97, further comprising administering an immunosuppressant to the subject.
99. The method of claim 98, wherein the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
100. A method of treating a. subject having or diagnosed with having amyotrophic lateral sclerosis (ALS), Parkinson’s disease (PD), or frontotemporal lobar degeneration (FTLD), comprising administering to the subject an effective amount of the pharmaceutical composition of any one of claims 59-62 or the AAV particle of any one of claims 1-49.
101. The method of claim 100, further comprising administering to the subject at least one additional therapeutic agent and/or therapy.
102. The method of claim 101 , wherein the at least one additional therapeutic agent and/or therapy comprises one or more of; growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, modulatory polynucleotides, and any protein known to be mutated in pathological disorders such as ATXN2-related disorders.
103. The method, of any one of claims 100-102, further comprising administering an immunosuppressant to the subject.
104. The method of claim 103, wherein the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolatemofet.il, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
105. The pharmaceutical composition of any one of claims 59-62 or the AAV particle of any one of claims 1-49 for use in a. method of treating a disorder according to any one of claim s 67-104.
106. The pharmaceutical composition of any one of claims 59-62 or the AAV particle of any one of claims 1-49 for use in treating an ATXN2-related disorder in a. subject, optionallywherein the ATXN2-related disorder is SCA2.
107. The pharmaceutical composition or the AA V particle for use of claim 106, wherein the subject has, has been diagnosed with having, or is at risk of having the ATXN2-related disorder, optionally wherein the ATXN2-related disorder is SCA2.
108. Use of the pharmaceutical composition of any one of claims 59-62 or the AAV particle of any one of claims 1-49 in the manufacture of a medicament for the treatment of an ATXN2-related disorder in a subject, optionally wherein the ATXN2-related disorder is SCA2.
109. The use of claim 108, wherein the subject has, has been diagnosed with having, or is at risk of having the ATXN2 -related disorder, optionally wherein the ATXN2-related disorder is SCA2.
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363463841P | 2023-05-03 | 2023-05-03 | |
| US63/463,841 | 2023-05-03 | ||
| US202363593857P | 2023-10-27 | 2023-10-27 | |
| US63/593,857 | 2023-10-27 | ||
| US202463564435P | 2024-03-12 | 2024-03-12 | |
| US63/564,435 | 2024-03-12 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2024229173A2 true WO2024229173A2 (en) | 2024-11-07 |
| WO2024229173A3 WO2024229173A3 (en) | 2024-12-12 |
Family
ID=91302105
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/027327 Pending WO2024229173A2 (en) | 2023-05-03 | 2024-05-01 | Compositions and methods for the treatment of disorders related to ataxin-2 |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202509226A (en) |
| WO (1) | WO2024229173A2 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12296025B2 (en) | 2021-11-02 | 2025-05-13 | Voyager Therapeutics, Inc. | AAV capsid variants and uses thereof |
| US12467046B2 (en) | 2018-10-02 | 2025-11-11 | Voyager Therapeutics, Inc. | Redirection of tropism of AAV capsids |
Citations (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5064764A (en) | 1988-12-20 | 1991-11-12 | Commissariat A L'energie Atomique | Mineral hollow fiber bioreactor for the cultivation of animal cells |
| WO1996023810A1 (en) | 1994-11-10 | 1996-08-08 | The Regents Of The University Of California | Modified green fluorescenct proteins |
| WO1996030540A2 (en) | 1995-03-20 | 1996-10-03 | The Regents Of The University Of California | Substrates for beta-lactamase and uses thereof |
| WO1996039530A2 (en) | 1995-06-05 | 1996-12-12 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines, and methods of production and use thereof |
| WO1998010088A1 (en) | 1996-09-06 | 1998-03-12 | Trustees Of The University Of Pennsylvania | An inducible method for production of recombinant adeno-associated viruses utilizing t7 polymerase |
| US5756283A (en) | 1995-06-05 | 1998-05-26 | The Trustees Of The University Of Pennsylvania | Method for improved production of recombinant adeno-associated viruses for gene therapy |
| WO1999014354A1 (en) | 1997-09-19 | 1999-03-25 | The Trustees Of The University Of The Pennsylvania | Methods and vector constructs useful for production of recombinant aav |
| WO1999015685A1 (en) | 1997-09-19 | 1999-04-01 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| WO1999047691A1 (en) | 1998-03-20 | 1999-09-23 | Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000028004A1 (en) | 1998-11-10 | 2000-05-18 | The University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| WO2000055342A1 (en) | 1999-03-18 | 2000-09-21 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000075353A1 (en) | 1999-06-02 | 2000-12-14 | Trustees Of The University Of Pennsylvania | Compositions and methods useful for production of recombinant viruses which require helper viruses |
| US6194191B1 (en) | 1996-11-20 | 2001-02-27 | Introgen Therapeutics, Inc. | Method for the production and purification of adenoviral vectors |
| US6204059B1 (en) | 1994-06-30 | 2001-03-20 | University Of Pittsburgh | AAV capsid vehicles for molecular transfer |
| WO2001023597A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Cell lines and constructs useful in production of e1-deleted adenoviruses in absence of replication competent adenovirus |
| WO2001023001A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Rapid peg-modification |
| US6265389B1 (en) | 1995-08-31 | 2001-07-24 | Alkermes Controlled Therapeutics, Inc. | Microencapsulation and sustained release of oligonucleotides |
| US6485966B2 (en) | 1999-03-18 | 2002-11-26 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US6566118B1 (en) | 1997-09-05 | 2003-05-20 | Targeted Genetics Corporation | Methods for generating high titer helper-free preparations of released recombinant AAV vectors |
| WO2004112727A2 (en) | 2003-06-19 | 2004-12-29 | Avigen, Inc. | Aav virions with decreased immunoreactivity and uses therefor |
| WO2005000560A2 (en) | 2003-06-25 | 2005-01-06 | Newtec International Group (Sa) | System for the thermal conditioning of preforms of plastic containers |
| WO2005005610A2 (en) | 2003-06-30 | 2005-01-20 | The Regents Of The University Of California | Mutant adeno-associated virus virions and methods of use thereof |
| WO2005072364A2 (en) | 2004-01-27 | 2005-08-11 | University Of Florida | A modified baculovirus expression system for production of pseudotyped raav vector |
| US6953690B1 (en) | 1998-03-20 | 2005-10-11 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US7291498B2 (en) | 2003-06-20 | 2007-11-06 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| US7465583B2 (en) | 2000-06-01 | 2008-12-16 | The University Of North Carolina At Chapel Hill | Duplexed parvovirus vectors |
| US7491508B2 (en) | 2003-06-20 | 2009-02-17 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| US8137948B2 (en) | 2003-05-21 | 2012-03-20 | Genzyme Corporation | Methods for producing preparations of recombinant AAV virions substantially free of empty capsids |
| US8163543B2 (en) | 2005-10-20 | 2012-04-24 | Amsterdam Molecular Therapeutics B.V. | AAV vectors produced in insect cells |
| WO2015191508A1 (en) | 2014-06-09 | 2015-12-17 | Voyager Therapeutics, Inc. | Chimeric capsids |
| WO2018119330A2 (en) | 2016-12-22 | 2018-06-28 | Oregon Health & Science University | Adeno associated viral vectors |
| WO2020072683A1 (en) | 2018-10-02 | 2020-04-09 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021202651A1 (en) | 2020-04-01 | 2021-10-07 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021230987A1 (en) | 2020-05-13 | 2021-11-18 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3830107A2 (en) * | 2018-08-03 | 2021-06-09 | Voyager Therapeutics, Inc. | Aav variants with enhanced tropism |
| US20220281922A1 (en) * | 2019-08-02 | 2022-09-08 | Voyager Therapeutics, Inc. | Aav variants with enhanced tropism |
-
2024
- 2024-05-01 WO PCT/US2024/027327 patent/WO2024229173A2/en active Pending
- 2024-05-02 TW TW113116412A patent/TW202509226A/en unknown
Patent Citations (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5064764A (en) | 1988-12-20 | 1991-11-12 | Commissariat A L'energie Atomique | Mineral hollow fiber bioreactor for the cultivation of animal cells |
| US6204059B1 (en) | 1994-06-30 | 2001-03-20 | University Of Pittsburgh | AAV capsid vehicles for molecular transfer |
| WO1996023810A1 (en) | 1994-11-10 | 1996-08-08 | The Regents Of The University Of California | Modified green fluorescenct proteins |
| WO1996030540A2 (en) | 1995-03-20 | 1996-10-03 | The Regents Of The University Of California | Substrates for beta-lactamase and uses thereof |
| WO1996039530A2 (en) | 1995-06-05 | 1996-12-12 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines, and methods of production and use thereof |
| US5756283A (en) | 1995-06-05 | 1998-05-26 | The Trustees Of The University Of Pennsylvania | Method for improved production of recombinant adeno-associated viruses for gene therapy |
| US6281010B1 (en) | 1995-06-05 | 2001-08-28 | The Trustees Of The University Of Pennsylvania | Adenovirus gene therapy vehicle and cell line |
| US6270996B1 (en) | 1995-06-05 | 2001-08-07 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines and methods of production and use thereof |
| US6261551B1 (en) | 1995-06-05 | 2001-07-17 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines, and methods of production and use thereof |
| US6555525B2 (en) | 1995-08-31 | 2003-04-29 | Alkermes Controlled Therapeutics, Inc. | Microencapsulation and sustained release of oligonucleotides |
| US6265389B1 (en) | 1995-08-31 | 2001-07-24 | Alkermes Controlled Therapeutics, Inc. | Microencapsulation and sustained release of oligonucleotides |
| WO1998010088A1 (en) | 1996-09-06 | 1998-03-12 | Trustees Of The University Of Pennsylvania | An inducible method for production of recombinant adeno-associated viruses utilizing t7 polymerase |
| US6194191B1 (en) | 1996-11-20 | 2001-02-27 | Introgen Therapeutics, Inc. | Method for the production and purification of adenoviral vectors |
| US6566118B1 (en) | 1997-09-05 | 2003-05-20 | Targeted Genetics Corporation | Methods for generating high titer helper-free preparations of released recombinant AAV vectors |
| US7238526B2 (en) | 1997-09-19 | 2007-07-03 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| WO1999014354A1 (en) | 1997-09-19 | 1999-03-25 | The Trustees Of The University Of The Pennsylvania | Methods and vector constructs useful for production of recombinant aav |
| US6943019B2 (en) | 1997-09-19 | 2005-09-13 | The Trustees Of The University Of Pennsylvania | Methods and vector constructs useful for production of recombinant AAV |
| WO1999015685A1 (en) | 1997-09-19 | 1999-04-01 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| US6475769B1 (en) | 1997-09-19 | 2002-11-05 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| US6482634B1 (en) | 1997-09-19 | 2002-11-19 | The Trustees Of The University Of Pennsylvania | Methods and vector constructs useful for production of recombinant AAV |
| US6953690B1 (en) | 1998-03-20 | 2005-10-11 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO1999047691A1 (en) | 1998-03-20 | 1999-09-23 | Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000028004A1 (en) | 1998-11-10 | 2000-05-18 | The University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| US6485966B2 (en) | 1999-03-18 | 2002-11-26 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000055342A1 (en) | 1999-03-18 | 2000-09-21 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US7022519B2 (en) | 1999-03-18 | 2006-04-04 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000075353A1 (en) | 1999-06-02 | 2000-12-14 | Trustees Of The University Of Pennsylvania | Compositions and methods useful for production of recombinant viruses which require helper viruses |
| US6365394B1 (en) | 1999-09-29 | 2002-04-02 | The Trustees Of The University Of Pennsylvania | Cell lines and constructs useful in production of E1-deleted adenoviruses in absence of replication competent adenovirus |
| WO2001023001A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Rapid peg-modification |
| WO2001023597A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Cell lines and constructs useful in production of e1-deleted adenoviruses in absence of replication competent adenovirus |
| US7465583B2 (en) | 2000-06-01 | 2008-12-16 | The University Of North Carolina At Chapel Hill | Duplexed parvovirus vectors |
| US8137948B2 (en) | 2003-05-21 | 2012-03-20 | Genzyme Corporation | Methods for producing preparations of recombinant AAV virions substantially free of empty capsids |
| WO2004112727A2 (en) | 2003-06-19 | 2004-12-29 | Avigen, Inc. | Aav virions with decreased immunoreactivity and uses therefor |
| US7491508B2 (en) | 2003-06-20 | 2009-02-17 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| US7291498B2 (en) | 2003-06-20 | 2007-11-06 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| WO2005000560A2 (en) | 2003-06-25 | 2005-01-06 | Newtec International Group (Sa) | System for the thermal conditioning of preforms of plastic containers |
| WO2005005610A2 (en) | 2003-06-30 | 2005-01-20 | The Regents Of The University Of California | Mutant adeno-associated virus virions and methods of use thereof |
| WO2005072364A2 (en) | 2004-01-27 | 2005-08-11 | University Of Florida | A modified baculovirus expression system for production of pseudotyped raav vector |
| US8163543B2 (en) | 2005-10-20 | 2012-04-24 | Amsterdam Molecular Therapeutics B.V. | AAV vectors produced in insect cells |
| WO2015191508A1 (en) | 2014-06-09 | 2015-12-17 | Voyager Therapeutics, Inc. | Chimeric capsids |
| WO2018119330A2 (en) | 2016-12-22 | 2018-06-28 | Oregon Health & Science University | Adeno associated viral vectors |
| WO2020072683A1 (en) | 2018-10-02 | 2020-04-09 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021202651A1 (en) | 2020-04-01 | 2021-10-07 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021230987A1 (en) | 2020-05-13 | 2021-11-18 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
Non-Patent Citations (26)
| Title |
|---|
| "Biocomputing: Informatics and Genome Projects", 1993, ACADEMIC PRESS |
| "Methods In Molecular Biology", 1995, HUMANA PRESS |
| "Remington: The Science and Practice of Pharmacy", 2006, WILLIAMS & WILKINS |
| "Sequence Analysis Primer", 1991, M STOCKTON PRESS |
| ADACHI ET AL., NATURE COMMUNICATIONS, vol. 5, no. 3075, 2014 |
| ALTSCHUL, S. F. ET AL., J. MOLECULAR BIOL., vol. 215, 1990, pages 403 |
| BELL ET AL., J. VTROL., vol. 86, no. 13, 2012, pages 7326 - 33 |
| CABIOS., vol. 4, 1989, pages 11 - 17 |
| CARILLO. H.LIPMAN, D., SIAM J APPLIED MATH., vol. 48, 1988, pages 1073 |
| DEVEREUX, J. ET AL., NUCLEIC .4CIDV RESEARCH., vol. 12, no. 1, 1984, pages 387 |
| HEIM ET AL., CURRENT BIOLOGY, vol. 2, 1996, pages 178 - 182 |
| HEIM ET AL., PROC. NATL. ACAD. SCI. USA, 1995 |
| HEIM ET AL., SCIENCE, vol. 373, 1995, pages 663 - 664 |
| HWANG ET AL.: "N-Terminal Acetylation of Cellular Proteins Creates Specific Degradation Signals", SCIENCE, vol. 327, no. 5968, 19 February 2010 (2010-02-19), pages 973 - 977, XP055369420, DOI: 10.1126/science.1183147 |
| JIN. ET AL.: "Direct Liquid Chcumatography/Λ tass Spectrometry Analysis for Complete Characterization of Recombinant Adeno-Associated Virus Capsid Proteins", FFUM GENE THER METHODS, vol. 28, no. 5, October 2017 (2017-10-01), pages 255 - 267 |
| KAJIGAYA ET AL., PROC. NAL'L. ACAD. SCI. USA, vol. 88, 1991, pages 4646 - 50 |
| KIMBAUER ET AL., I'IR.., vol. 219, 1996, pages 37 - 44 |
| MIYANOHARA ET AL., MOL THER METHODS CLIN DEV., vol. 3, 2016, pages 16046 |
| O'REILLY ET AL.: "Baculovirus Expression Vectors, A Laboratory Manual", 1994, OXFORD UNIV. PRESS |
| PARR ET AL., N AT. MED., vol. 3, 1997, pages 1145 - 9 |
| POWELL ET AL., VIRAL EXPRESSION CASSETTE ELEMENTS TO ENHANCE TRANSGENE TARGET SPECIFICITY AND EXPRESSION IN GENE THERAPY, 2015 |
| PULICHERLA ET AL., MOL. THER., vol. 19, no. 6, 2011, pages 1070 - 1078 |
| RUFFING ET AL., J. VIR., vol. 66, 1992, pages 6922 - 30 |
| SAMULSKI ET AL., J. VIR., vol. 63, 1989, pages 3822 - 8 |
| VON HEINJE, G.: "Sequence Analysis in Molecular Biology", 1987, ACADEMIC PRESS |
| ZHAO ET AL., I'IR., vol. 272, 2000, pages 382 - 93 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12467046B2 (en) | 2018-10-02 | 2025-11-11 | Voyager Therapeutics, Inc. | Redirection of tropism of AAV capsids |
| US12296025B2 (en) | 2021-11-02 | 2025-05-13 | Voyager Therapeutics, Inc. | AAV capsid variants and uses thereof |
| US12419969B2 (en) | 2021-11-02 | 2025-09-23 | Voyager Therapeutics, Inc. | AAV capsid variants and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024229173A3 (en) | 2024-12-12 |
| TW202509226A (en) | 2025-03-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12419969B2 (en) | AAV capsid variants and uses thereof | |
| WO2024229173A2 (en) | Compositions and methods for the treatment of disorders related to ataxin-2 | |
| WO2024229164A2 (en) | Compositions and methods for the treatment of disorders related to dystrophia myotonica protein kinase | |
| CA3190309A1 (en) | Compositions and methods for the treatment of neurological disorders related to glucosylceramidase beta deficiency | |
| WO2024229167A1 (en) | Compositions and methods for the treatment of disorders related to syntaxin-binding protein 1 deficiency | |
| WO2024229163A1 (en) | Compositions and methods for the treatment of disorders related to cdkl5 deficiency | |
| WO2024229125A2 (en) | Compositions and methods for the treatment of disorders related to frataxin deficiency | |
| WO2024229161A1 (en) | Compositions and methods for the treatment of disorders related to glucosylceramidase beta 1 deficiency | |
| WO2024226761A2 (en) | Compositions and methods for treating amyotrophic lateral sclerosis | |
| TW202444917A (en) | Aav capsid variants and uses thereof | |
| WO2025038802A1 (en) | Compositions and methods for the treatment of disorders related to ataxin-2 | |
| WO2025038795A1 (en) | Compositions and methods for the treatment of disorders related to dystrophia myotonica protein kinase | |
| WO2025038796A1 (en) | Compositions and methods for the treatment of disorders related to cdkl5 deficiency | |
| WO2025038805A1 (en) | Compositions and methods for the treatment of disorders related to glucosylceramidase beta 1 deficiency | |
| AU2024266140A1 (en) | Compositions and methods for regulating mapt | |
| US20230285596A1 (en) | Compositions and methods for the treatment of niemann-pick type c1 disease | |
| KR20260021096A (en) | Compositions and methods for treating disorders associated with CDKL5 deficiency | |
| KR20260020501A (en) | Compositions and methods for treating disorders associated with syndrome-binding protein 1 deficiency | |
| KR20260020502A (en) | Compositions and methods for treating disorders associated with glucosylceramidase beta 1 deficiency | |
| CN121488045A (en) | AAV capsid variants and uses thereof | |
| CN118632861A (en) | AAV capsid variants and their uses | |
| HK40111250A (en) | Aav capsid variants and uses thereof | |
| WO2025137219A1 (en) | Compositions and methods for the treatment of disorders related to dystrophia myotonica protein kinase | |
| WO2025235734A9 (en) | Aav capsid variants and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24729477 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |