WO2024227154A1 - Complement component 3 (c3)-specific antibodies and uses thereof - Google Patents
Complement component 3 (c3)-specific antibodies and uses thereof Download PDFInfo
- Publication number
- WO2024227154A1 WO2024227154A1 PCT/US2024/026808 US2024026808W WO2024227154A1 WO 2024227154 A1 WO2024227154 A1 WO 2024227154A1 US 2024026808 W US2024026808 W US 2024026808W WO 2024227154 A1 WO2024227154 A1 WO 2024227154A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- amino acid
- acid sequence
- antibody
- lcdr1
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Definitions
- Embodiments of the present disclosure relate to the treatment and/or prevention of age- related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA).
- AMD age-related macular degeneration
- GA Geographic Atrophy
- the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3) proteins.
- C3 Complement Component 3
- Age-Related Macular Degeneration is the leading cause of severe vision loss in adults over age 50.
- the Centers for Disease Control and Prevention estimate that 1.8 million people have AMD and another 7.3 million are at substantial risk for vision loss from AMD.
- This eye disease occurs when there are changes to the macula, a small portion of the retina that is located on the inside back layer of the eye.
- AMD is a loss of central vision that can occur in two forms: “dry” (atrophic) and “wet” (exudative). Most people with macular degeneration have the dry form.
- GA Geographic atrophy
- AMD is considered the late stage of the dry form of AMD, characterized by focal and progressive loss of photoreceptors.
- GA is less common than neovascular AMD, and it is responsible for 10-20% of cases of legal blindness in this condition, affecting more than 5 million people worldwide.
- Embodiments of the present disclosure include compositions and methods pertaining to an antibody, or antigen binding fragment thereof, which specifically binds Complement Component 3 (C3) polypeptides, optionally wherein said C3 is a Complement Component 3a (C3a) polypeptide or a Complement Component 3b (C3b) polypeptide, which is optionally a polypeptide comprising or consisting of the amino acid sequence of SEQ ID NO: 1064.
- the present disclosure provides an antibody or a fragment thereof, which exhibits any one or more the following functional characteristics: (a) reduces complement activation and/or attenuates complement component 3 (C3) convertase activity, which is optionally measured in an ELISA; and/or (b) cross-reacts with at least one of human C3b protein, Cynomolgus monkey C3 protein, and/or human C3a protein; and/or (c) binds to human C3 with a KD -1 of about lOOnM or lower; and/or (d) binds to the same epitope on human C3 as an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) competes for binding to human C3 with an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6.
- C3 complement component 3
- the antibody or fragment thereof which is a monoclonal antibody. In some embodiments, the antibody or fragment thereof, which is a human, humanized or chimeric antibody. In further embodiments, the antibody or fragment thereof, is a whole antibody, a single chain variable fragment (scFv), a variable fragment (Fv), a fragment antigen-binding region (Fab), a Fab-C, a Fab’-SH, a (Fab’)2, a single-domain antibody (sdAb), a nanobody, a diabody, a triabody, or an aptamer, optionally wherein the antibody is a whole antibody comprising an Fc region such as a human IgGl , IgG2, IgG3 or IgG4 Fc region.
- an Fc region such as a human IgGl , IgG2, IgG3 or IgG4 Fc region.
- the antibody or fragment thereof is conjugated to at least one additional moiety, optionally selected from: (a) a therapeutic or cytotoxic moiety; (b) a detection moiety; (c) a purification moiety; (d) a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody.
- additional moiety optionally selected from: (a) a therapeutic or cytotoxic moiety; (b) a detection moiety; (c) a purification moiety; (d) a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody.
- the present disclosure provides an antibody or a fragment thereof, which is a polypeptide comprising: (a) one, two or all three HCDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also one, two or all three of the corresponding LCDRs of said exemplary antibody; and/or (b) a VH sequence having at least 90% identity to the VH sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also a VL sequence having at least 90% identity to the corresponding VL sequence of said exemplary antibody, preferably wherein variation is not permitted in the HCDRs or LCDRs; and/or (c) all six CDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (d) the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) the full length heavy chain (VH + constant) sequence of any one of the
- an expression vector comprises a polynucleotide, which is optionally an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, or a retrovirus vector.
- AAV adeno-associated virus
- LV lentiviral
- HSV herpes simplex virus
- Embodiments of the present disclosure include compositions and methods pertaining to antibodies directed against Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, including antigen binding fragments thereof.
- the present disclosure provides a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3.
- VH heavy chain variable region
- CDRs complementarity determining regions
- VL light chain variable region
- the HCDR1 comprises one of the following amino acid sequences: the HCDR1 comprises one of the following amino acid sequences: (a) X1YX2X3X4 (SEQ ID NO: 1), wherein Xi is G, S, N, D, T, or A; X2 is Y, A, G, W, or T; X 3 is L, M, I, or V; and X 4 is H, N, S, or G; (b) X1X2 X3MX4 (SEQ ID NO: 57), wherein Xi is D, N, S, or A; X2 is Y, or A; X3 is A, W, Y, S, C, D, or G; and X4 is H, S, or N; (c) X1X2X3WWX4 (SEQ ID NO: 95), wherein Xi is I, S, R, N, or K; X 2 is N, D, S, or G; X 3 is N
- the HCDR2 comprises one of the following amino acid sequences: (a) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 20), wherein Xi is W, I, or V; X 2 is N, S, or Y; X 3 is P, T, A, G, or Y; X 4 is N, Y, G, D, H, or A; X 5 is S, T, N, G, or D; X 6 is G, or S; X 7 is D, G, T, N, V, A, or H; X 8 is T, P, or K; X9 is N, Y, T, H, R, I, D, or K; X 10 is Y, or A; Xu is V, Q, A, or S; X12 is Q, K, P, or D; X13 is N, F, K, G, S, or Q; X14 is
- the HCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 39-56; (b) SEQ ID NOs: 83-94; (c) SEQ ID NOs: 153- 180; (d) SEQ ID NOs: 197-203; (e) SEQ ID NOs: 220-226; or (f) SEQ ID NOs: 234-235.
- the LCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 237-267, SEQ ID NOs: 332-333, SEQ ID NOs: 341-348, or SEQ ID NOs: 368-401;
- the LCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 269-299, SEQ ID NOs: 335- 336, SEQ ID NOs: 350-357, or SEQ ID NOs 403-436;
- the LCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 300-330, SEQ ID NOs: 339-339, SEQ ID NOs: 359-366, or SEQ ID NOs: 438-471.
- Embodiments of the present disclosure also include antibodies directed against Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, including antigen binding fragments thereof, comprising a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3.
- VH heavy chain variable region
- CDRs complementarity determining regions
- VL light chain variable region
- the LCDR1 comprises one of the following amino acid sequences: (a) X1ASQX2X3X4X5X6X7X8 (SEQ ID NO: 236), wherein Xi is R or Q; X 2 is D, N, T, G, or S; X 3 is V or I; X 4 is S, I, R, G, T, or K; X 5 is T, N, R, or S; X 6 is A, W, F, D, Y, N, or C; X 7 is V or L; X 8 is A, N, G, or H; (b) RSSQX1LLHX2NGYNYLD (SEQ ID NO: 331), wherein Xi is S or I; X 2 is S or T; or (c) RASQSX1X2X3X4X5X6 (SEQ ID NO: 340), wherein Xi is V or I; X 2 is S or R; X 3 is S, D, or R;
- the LCDR2 comprises one of the following amino acid sequences: (a) X1ASX2X3X4X5 (SEQ ID NO: 268), wherein Xi is S, K, A, Y, T, D, or V; X 2 is F, S, N, Q, or T; X 3 is L, S, or F; X 4 is Y, E, Q, or F; X 5 is S or T; (b) LGSXiRAS (SEQ ID NO: 334), wherein Xi is N or Y; (c) X1ASX2RAT (SEQ ID NO: 349), wherein Xi is D or G; X 2 is S or N; or (d) WASXiRES (SEQ ID NO: 402), wherein Xi is N or T.
- SEQ ID NO: 268 X1ASX2X3X4X5
- Xi is S, K, A, Y, T, D, or V
- X 2 is F, S,
- the LCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 300-330; (b) MQX1LQTPX2T (SEQ ID NO: 337), wherein Xi is T or A; X 2 is P or L; (c) QQX1X2X3X4PX5T (SEQ ID NO: 358), wherein Xi is R or Y; X 2 is S, G, or D; X 3 is N or S; X 4 is W or S; X 5 is L, R, I, Y, F, or W; or (d) QQX1X2X3TX4X5 (SEQ ID NO: 437), wherein Xi is Y or F; X 2 is Y or F; X 3 is S, G, or D; X4 is P or R; X5 is Y, W, R, T, or P.
- the HCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 2-19, SEQ ID NOs: 58-69, SEQ ID NOs: 96-123, SEQ ID NOs: 182-188, SEQ ID NOs: 205-211, or SEQ ID NOs: 228-229;
- the HCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 21-38, SEQ ID NOs: 71- 82, SEQ ID NOs: 125-152, SEQ ID NOs 190-196, SEQ ID NOs 213-219, or SEQ ID NOs 231- 232;
- the HCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 39-56, SEQ ID NOs: 83-94, SEQ ID NOs: 153-180, SEQ ID NOs: 197-203, SEQ ID NOs: 220-226, or SEQ ID NOs: 234-235.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 2; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 21; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 39.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 3; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 22; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 40.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 4; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 23; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 41.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 5; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 24; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 42.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 6; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 25; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 43.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 7; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 26; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 44.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 8; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 27; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 45.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 9; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 28; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 46.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 10; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 29; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 47.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 11; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 30; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 48.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 12; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 31; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 49.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 13; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 32; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 50.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 14; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 33; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 51.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 15; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 34; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 52.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 16; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 35; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 53.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 17; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 36; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 54.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 18; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 37; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 55.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 19; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 38; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 56.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 58; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 71; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 83.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 60; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 73; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 85.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 61; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 74; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 86.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 62; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 75; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 87.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 63; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 76; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 88.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 64; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 77; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 89.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 65; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 78; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 90.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 66; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 79; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 91.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 67; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 80; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 92.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 68; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 81; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 93.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 69; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 82; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 94.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 96; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 125; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 153.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 97; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 126; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 154.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 98; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 127; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 155.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 99; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 128; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 156.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 100; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 129; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 157.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 101; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 130; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 158.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 102; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 131; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 159.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 103; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 132; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 160.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 104; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 133; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 161.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 105; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 134; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 162.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 106; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 135; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 163.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 107; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 136; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 164.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 108; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 137; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 165.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 109; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 138; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 166.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 110; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 139; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 167.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 111; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 140; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 168.
- HCDR1 comprises the amino acid sequence of SEQ ID NO: 112; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 141; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 169.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 113; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 142; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 170.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 114; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 143; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 171.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 115; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 144; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 172.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 116; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 145; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 173.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 117; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 146; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 174.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 118; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 147; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 175.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 119; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 148; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 176.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 120; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 149; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 177.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 121; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 150; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 178.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 122; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 151; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 179.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 123; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 152; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 180.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 182; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 190; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 197.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 183; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 191; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 198.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 184; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 192; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 199.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 185; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 193; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 200.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 186; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 194; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 201.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 187; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 195; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 202.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 188; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 196; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 203.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 205; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 213; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 220.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 206; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 214; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 221.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 207; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 215; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 222.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 208; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 216; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 223.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 209; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 217; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 224.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 210; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 218; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 225.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 211; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 219; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 226.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 228; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 231; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 234.
- the HCDR1 comprises the amino acid sequence of SEQ ID NO: 229; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 232; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 35.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 237; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 269; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 300.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 238; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 270; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 301.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 239; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 271; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 302.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 240; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 272; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 303.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 241; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 273; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 304.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 242; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 274; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 305.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 243; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 275; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 306.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 244; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 276; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 307.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 245; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 277; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 308.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 246; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 278; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 309.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 247; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 279; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 310.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 248; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 280; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 311.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 249; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 281; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 312.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 250; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 282; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 313.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 251; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 283; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 314.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 252; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 284; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 315.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 253; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 285; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 316.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 254; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 286; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 317.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 255; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 287; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 318.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 256; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 288; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 319.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 257; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 289; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 320.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 258; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 290; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 321.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 259; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 291; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 322.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 260; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 292; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 323.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 261; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 293; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 324.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 262; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 294; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 325.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 263; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 295; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 326.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 264; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 296; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 327.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 265; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 297; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 328.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 266; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 298; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 329.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 267; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 299; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 330.
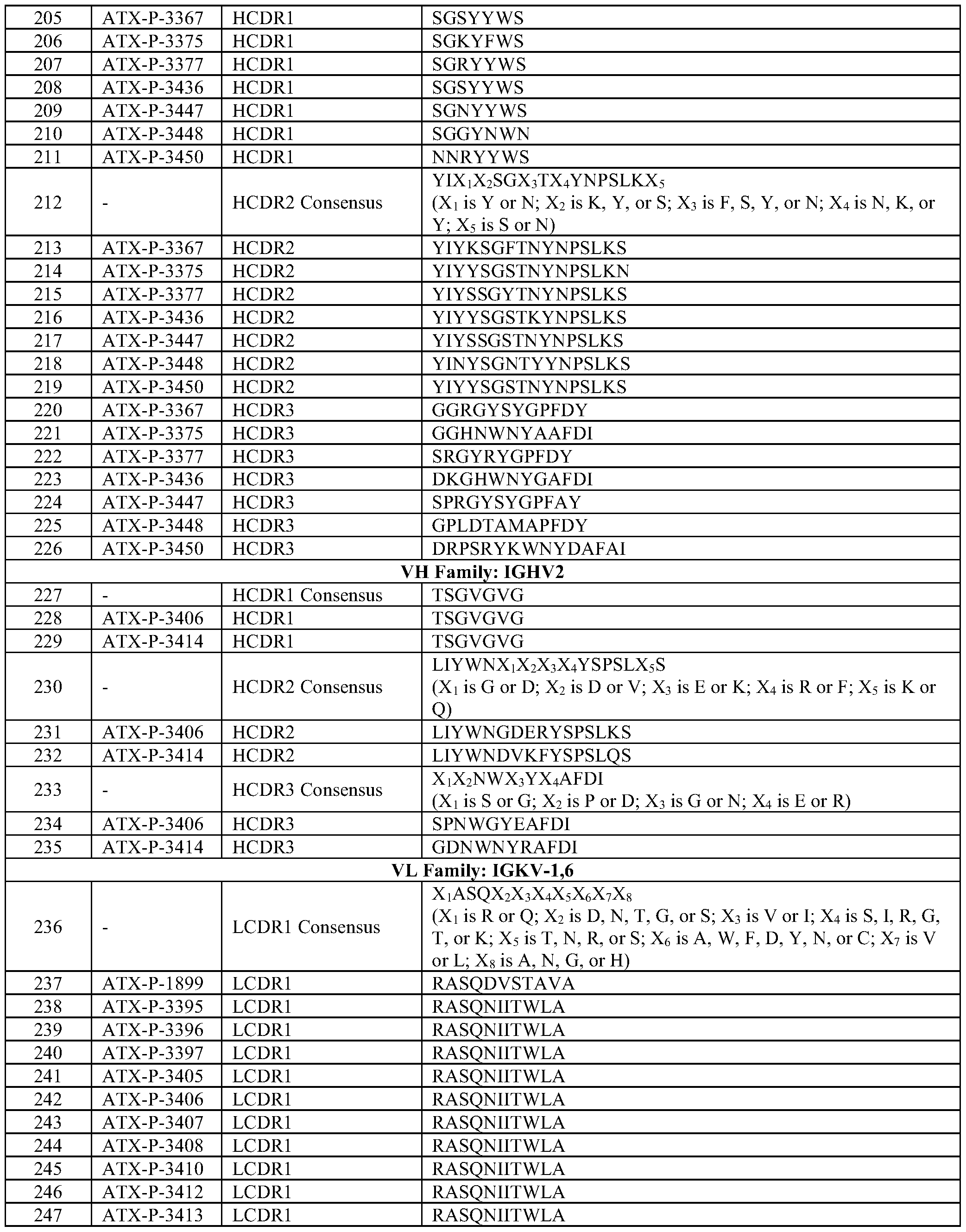
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 332; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 335; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 338.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 333; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 336; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 339.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 341; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 350; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 359.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 342; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 351; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 360.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 343; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 352; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 361.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 344; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 353; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 362.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 345; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 354; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 363.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 346; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 355; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 364.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 347; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 356; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 365.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 348; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 357; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 366.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 368; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 403; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 438.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 369; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 404; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 439.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 370; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 405; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 440.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 371; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 406; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 441.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 372; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 407; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 442.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 373; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 408; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 443.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 374; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 409; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 444.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 375; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 410; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 445.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 376; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 411; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 446.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 377; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 412; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 447.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 378; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 413; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 448.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 379; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 414; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 449.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 380; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 415; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 450.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 381; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 416; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 451.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 382; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 417; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 452.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 383; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 418; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 453.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 384; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 419; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 454.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 385; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 420; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 455.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 386; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 421; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 456.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 387; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 422; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 457.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 388; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 423; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 458.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 389; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 424; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 459.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 390; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 425; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 460.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 391; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 426; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 461.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 392; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 427; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 462.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 393; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 428; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 463.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 394; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 429; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 464.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 395; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 430; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 465.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 396; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 431; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 466.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 397; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 432; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 467.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 398; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 433; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 468.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 399; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 434; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 469.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 400; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 435; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 470.
- the LCDR1 comprises the amino acid sequence of SEQ ID NO: 401; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 436; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 471.
- the VH comprises an amino acid sequence that is at least 90% identical to any of SEQ ID NOs: 546-619.
- the VL comprises an amino acid sequence that is at least 90% identical to any of SEQ ID NOs: 694-767.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 546 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 694. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 547 and the and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 695. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 548 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 696.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 549 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 697. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 550 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 698. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 551 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 699.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 552 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 700. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 553 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 701. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 554 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 702.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 555 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 703.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 556 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 704.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 557 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 705.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 558 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 706. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 559 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 707. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 560 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 708.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 561 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 709. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 562 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 710. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 563 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 711.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 564 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 712. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 565 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 713. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 566 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 714.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 567 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 715. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 568 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 716. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 569 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 717.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 570 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 718. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 571 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 719. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 572 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 720.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 573 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 721. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 574 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 722. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 575 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 723.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 576 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 724. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 577 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 725. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 578 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 726.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 579 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 727. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 580 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 728. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 581 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 729.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 582 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 730. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 583 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 731. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 584 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 732.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 585 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 733. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 586 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 734. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 587 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 735.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 588 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 736. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 589 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 737. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 590 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 738.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 591 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 739. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 592 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 740. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 593 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 741.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 594 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 742. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 595 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 743. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 596 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 744.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 597 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 745. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 598 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 746. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 599 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 747.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 600 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 748. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 601 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 749. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 602 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 750.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 603 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 751. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 604 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 752. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 605 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 753.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 606 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 754. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 607 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 755. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 608 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 756.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 609 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 757. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 610 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 758. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 611 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 759.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 612 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 760. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 613 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 761. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 614 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 762.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 615 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 763. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 616 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 764. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 617 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 765.
- the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 618 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 766. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 619 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 767.
- the anti-C3 antibodies of the present disclosure bind an epitope from a C3 polypeptide, or fragment thereof, having an amino acid sequence of SEQ ID NO: 1064. In some embodiments, the anti-C3 antibodies of the present disclosure bind C3, C3a, and/or C3b, and reduce complement activation. In some embodiments, the anti-C3 antibodies of the present disclosure bind C3, C3a, and/or C3b, with a KD of about 100 nM or lower. In some embodiments, the anti-C3 antibodies of the present disclosure bind C3, C3a, and/or C3b, and attenuate C3 convertase activity.
- the antibody is a monoclonal antibody, a human antibody, a humanized antibody, and/or a chimeric antibody.
- the antibody is a fragment selected from the group consisting of Fab, Fab-C, Fab'-SH, Fv, scFv, and (Fab')? fragments.
- the antibody is a monospecific antibody.
- the antibody is a bispecific antibody.
- the antibody comprises a detection moiety. In some embodiments, the antibody comprises a purification moiety.
- the antibody comprises a half-life extension moiety.
- the half-life extension moiety comprises a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residues.
- the half-life extension polypeptide is attached to the C-terminus or N-terminus of the antibody.
- Embodiments of the present disclosure also includes a pharmaceutical composition comprising any of the anti-C3 antibodies described herein.
- the composition is suitable for ocular administration.
- ocular administration comprises injection into vitreous fluid.
- ocular administration comprises delivering the antibody using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant.
- Embodiments of the present disclosure also include a method of treating age-related macular degeneration (AMD) comprising administering a pharmaceutical composition comprising an effective amount of any of the anti-C3 antibodies described herein to a subject in need thereof.
- the pharmaceutical composition is administered ocularly and treats at least one AMD symptom.
- AMD comprises wet AMD.
- AMD comprises dry AMD.
- at least one AMD symptom comprises visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light.
- administering the pharmaceutical composition reduces complement activation in the subject’s eye.
- the pharmaceutical composition is administered at a dose ranging from about 0.0001 mg/dose to about 100 mg/dose. In some embodiments, the pharmaceutical composition is administered at a dose ranging from about 0.0001 mg/ml to about 100 mg/ml.
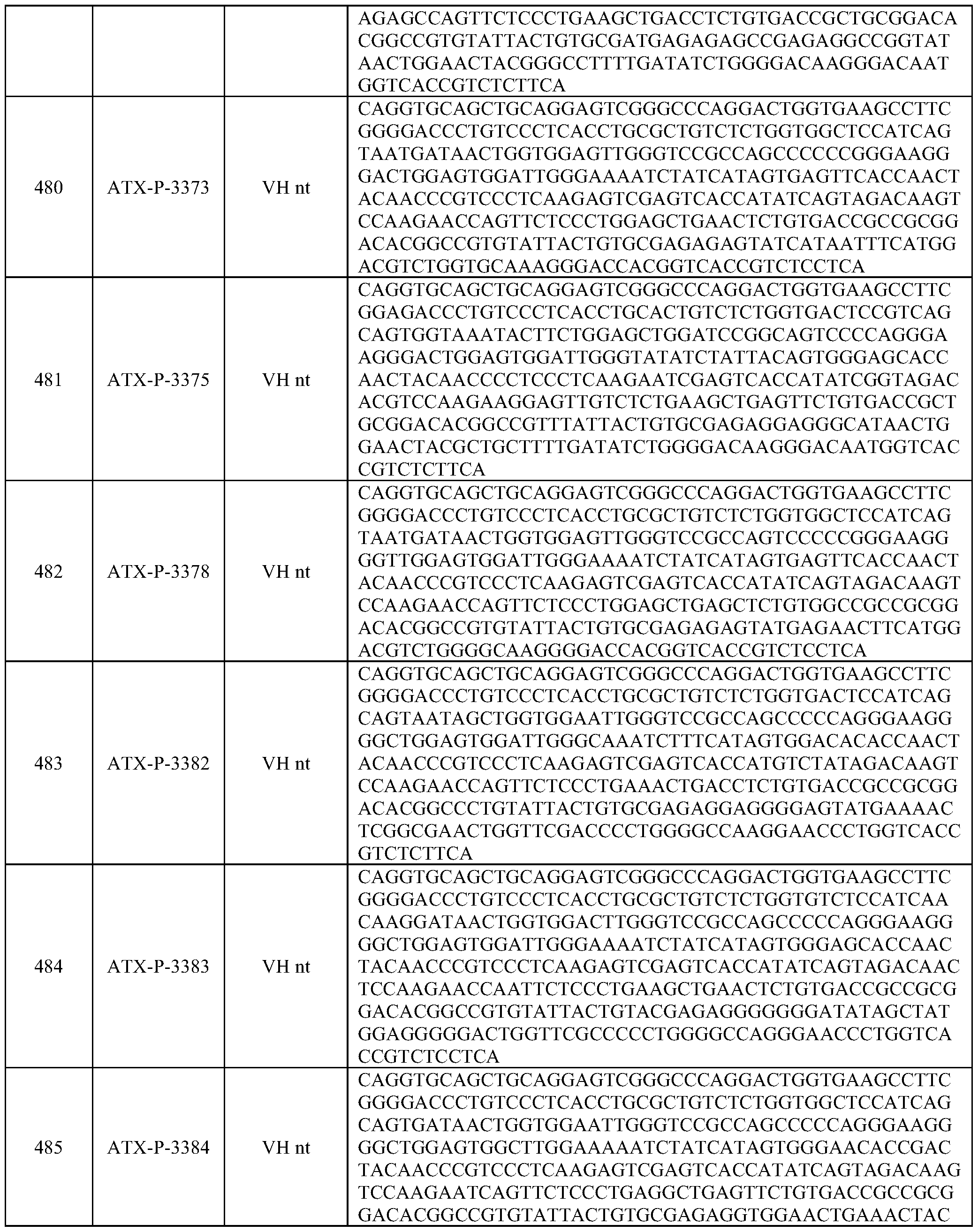
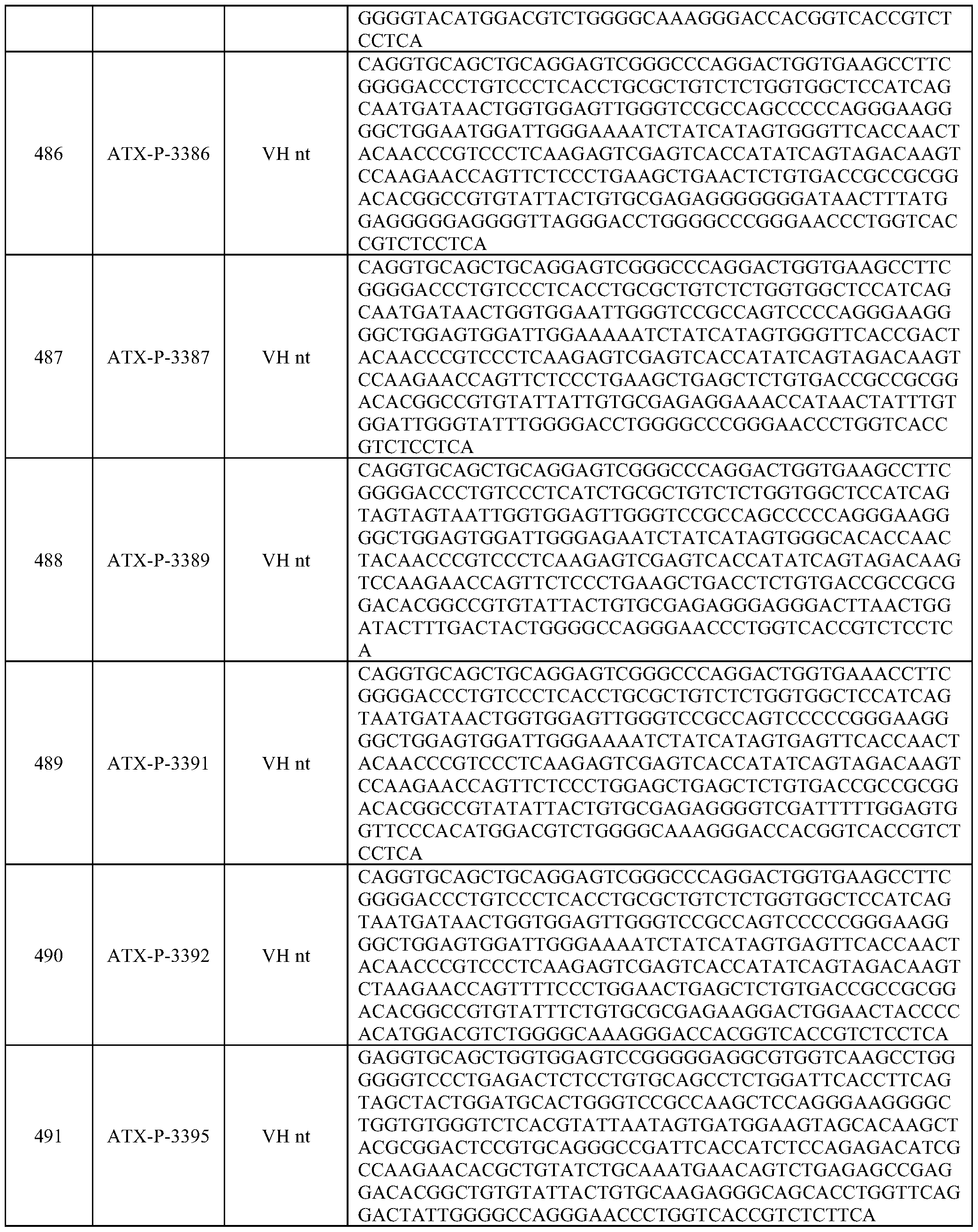
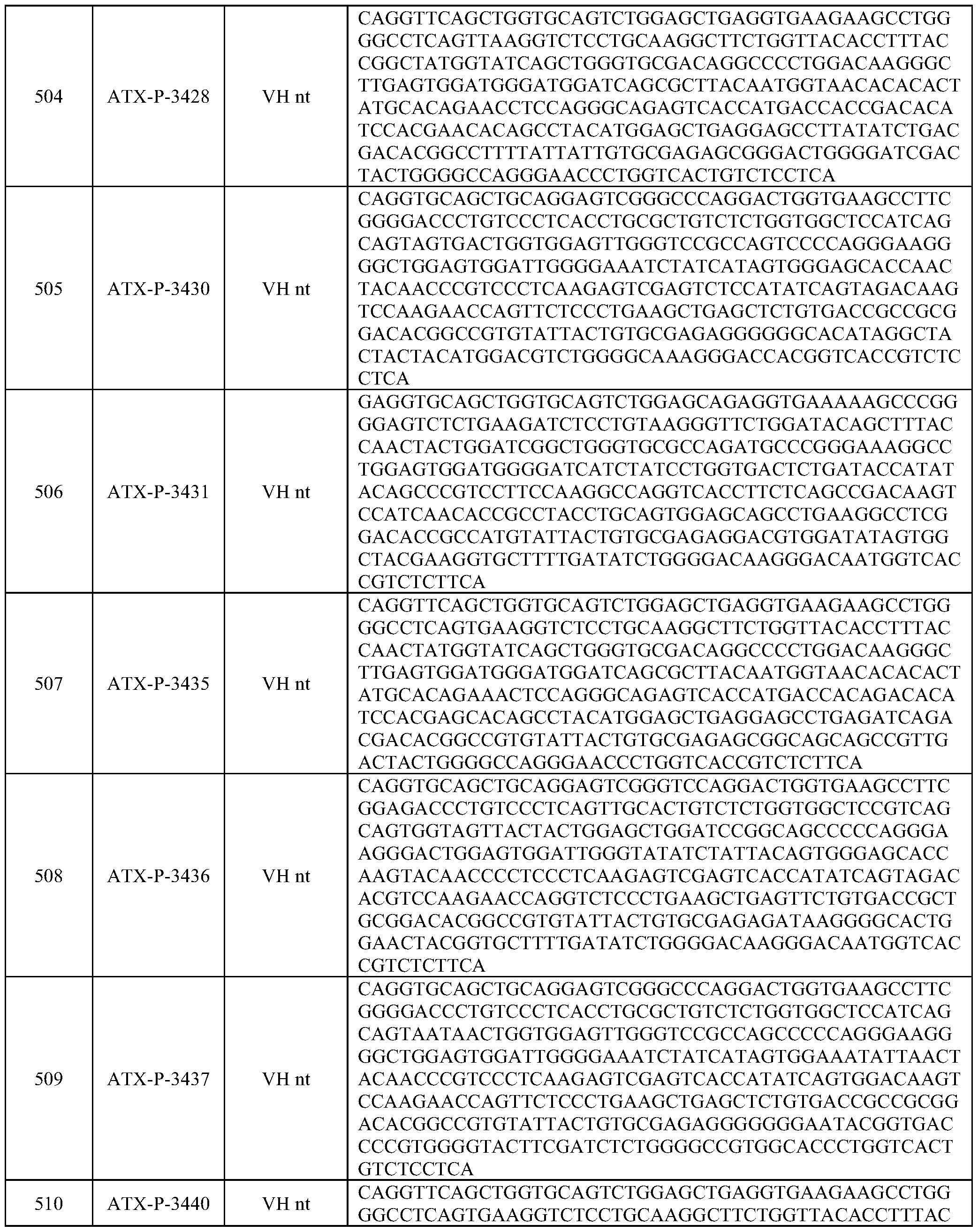
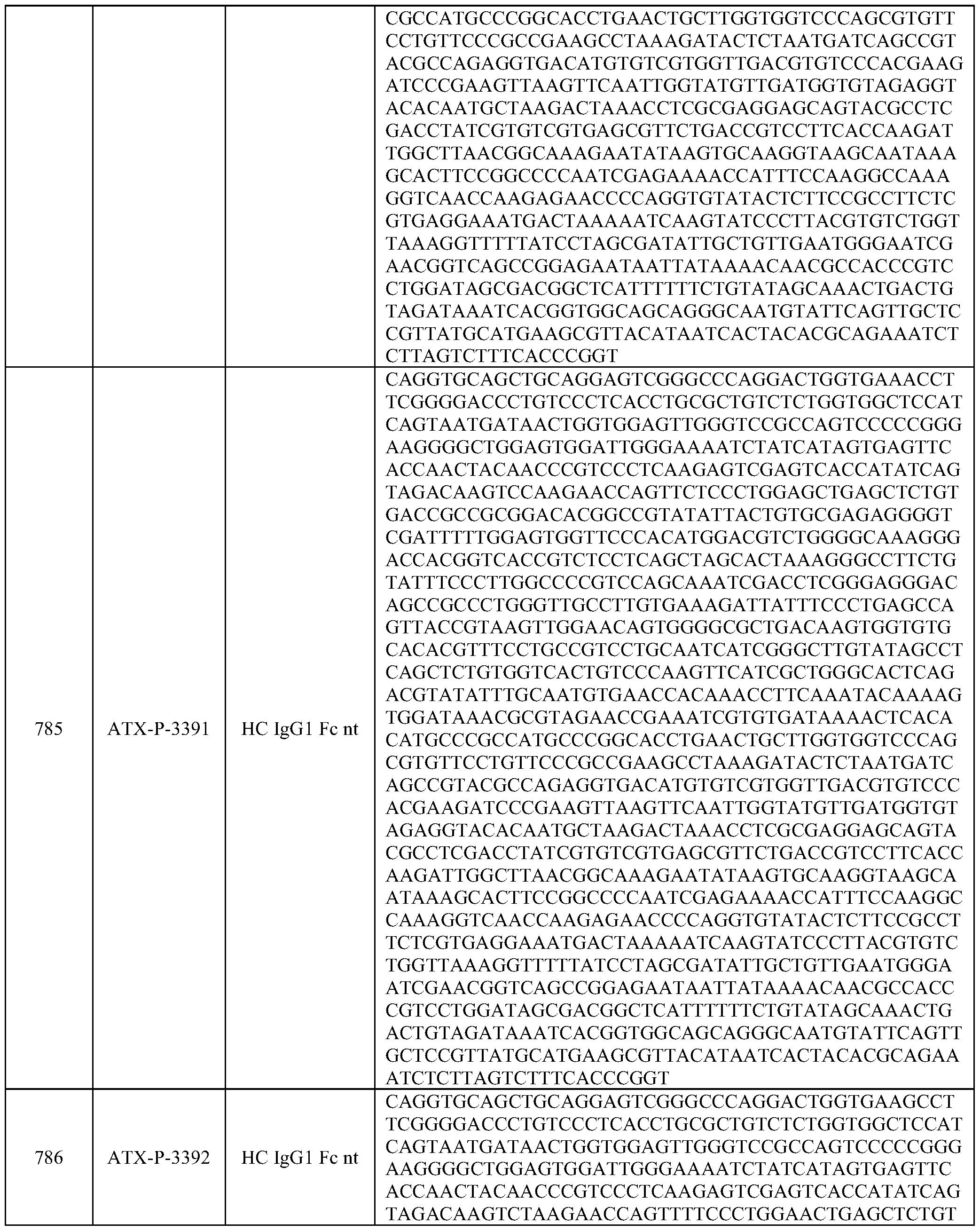
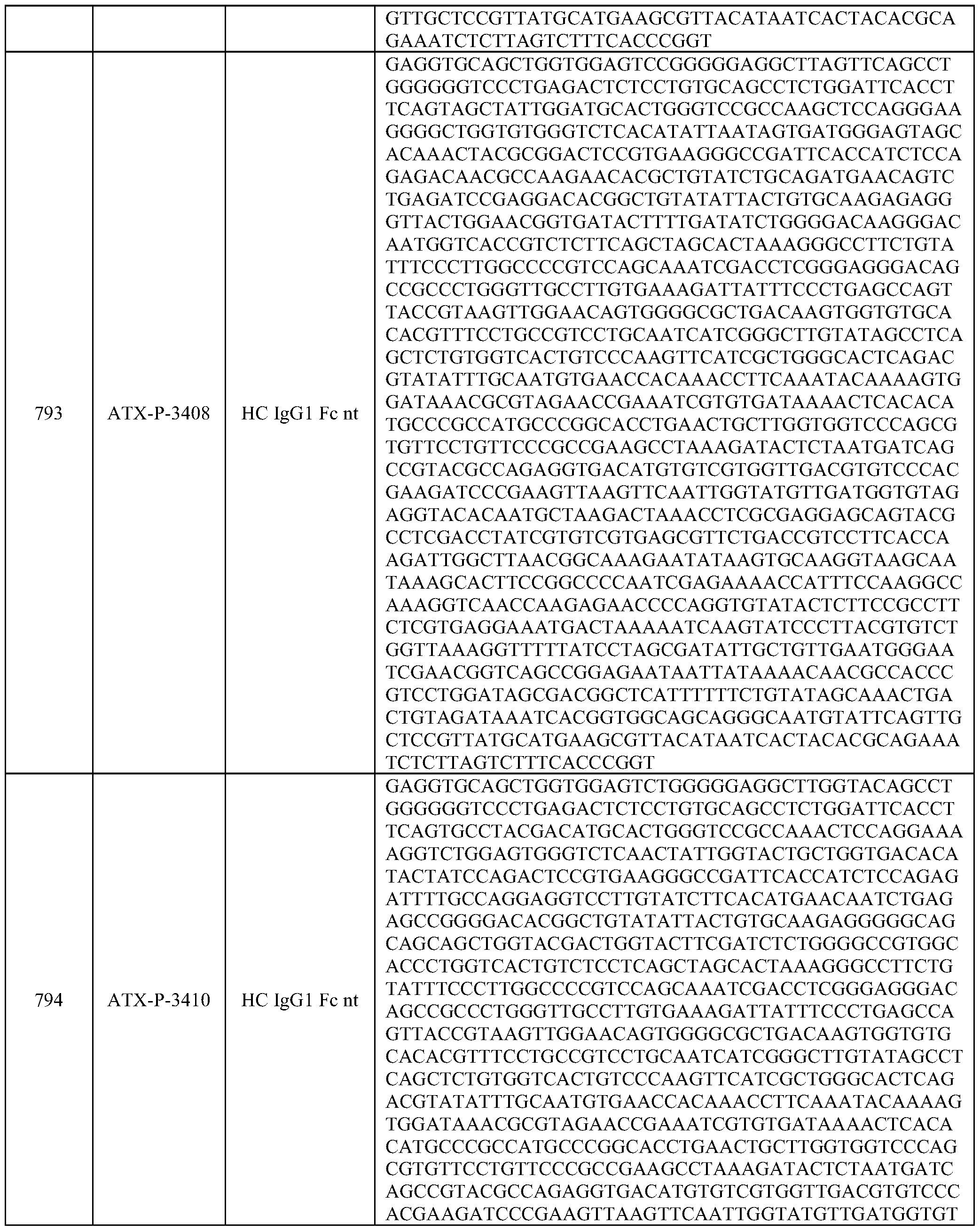
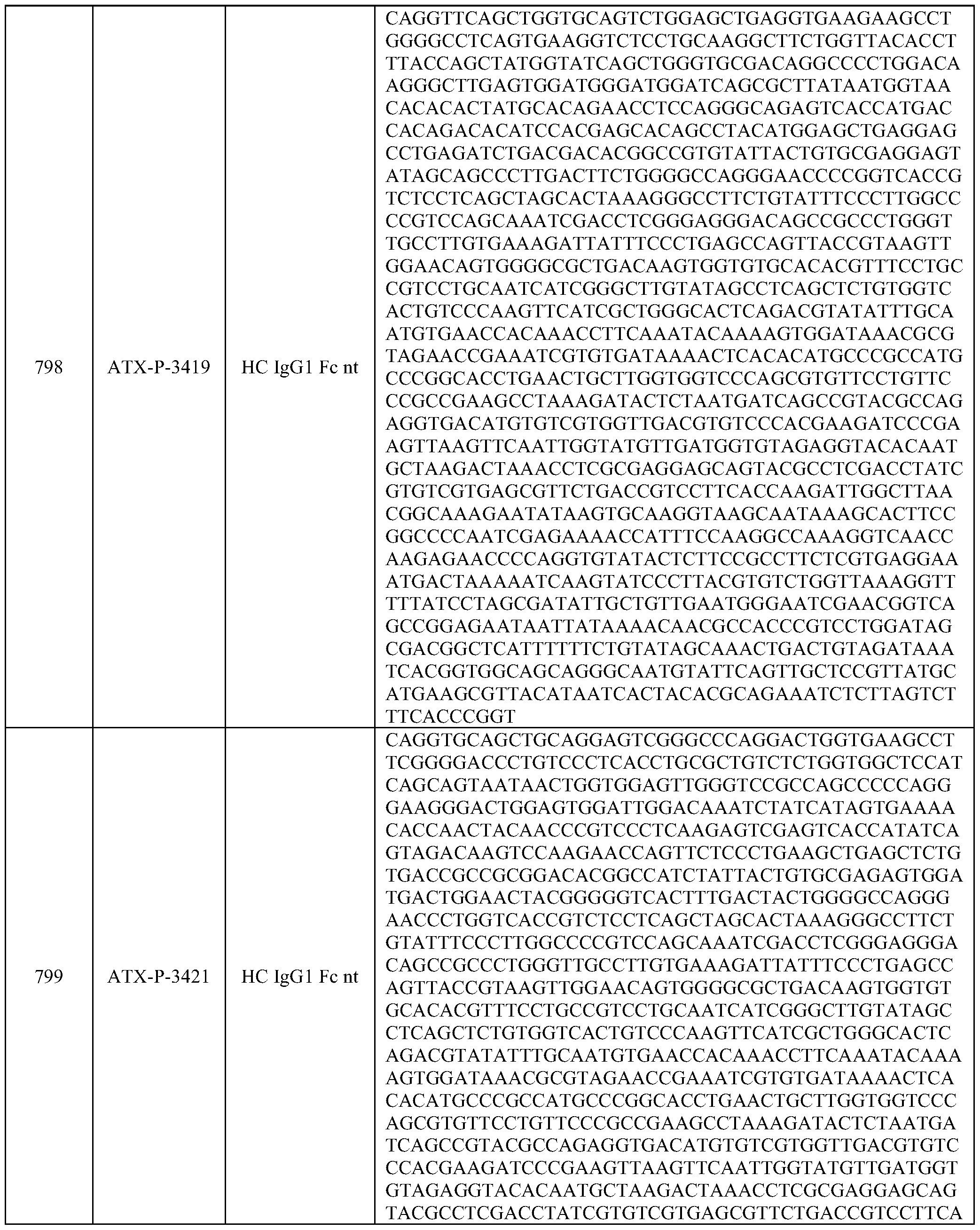
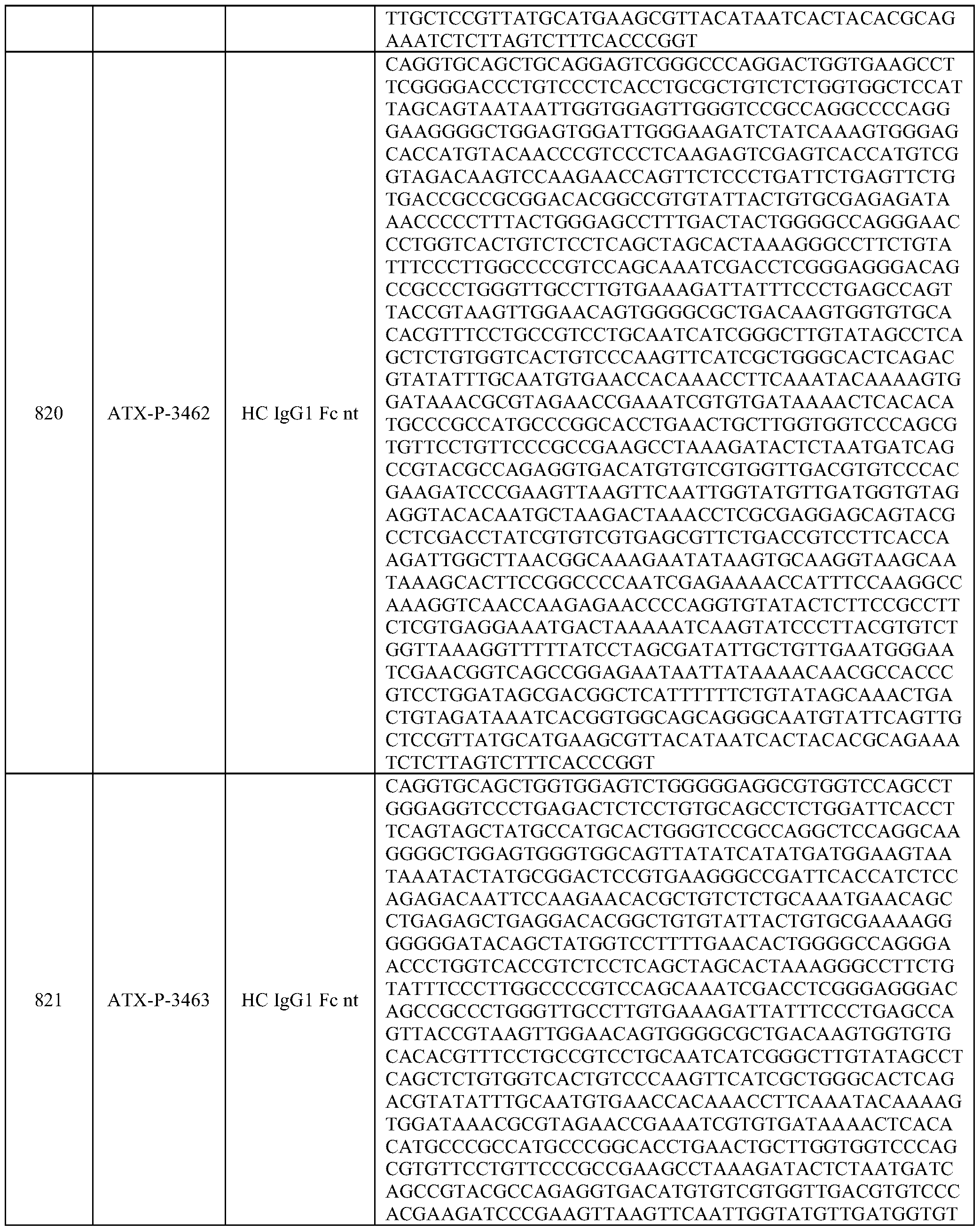
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 70% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 70% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
- Embodiments of the present disclosure also include an expression vector comprising any of the polynucleotides described herein.
- the expression vector is at least one of: an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, and a retrovirus vector.
- AAV adeno-associated virus
- LV lentiviral
- HSV herpes simplex virus
- retrovirus vector retrovirus vector
- Embodiments of the present disclosure also include a method of administering ocular gene therapy to a subject in need thereof comprising injecting a pharmaceutical composition comprising an effective amount of an expression vector described herein.
- Embodiments of the present disclosure also include a method of treating age-related macular degeneration (AMD) comprising administering a pharmaceutical composition comprising an effective amount of an expression vector described herein, wherein administering the pharmaceutical composition treats at least one AMD symptom.
- AMD age-related macular degeneration
- FIGS. 1 A-1D Representative ELISA results used to determine antigen positive serum titers of mice immunized with human C3 and cyno C3, according to different immunization protocols/cohorts (Cohort 1: FIG. 1A (human C3) and FIG. IB (cyno C3); Cohort 2: FIG. 1C (human C3); FIG. ID (cyno C3)).
- FIG. 2 Representing ELISA results of polyclonal phage pools to confirm enrichment and examine cross reactivity.
- FIG. 3 Representative table containing binding kinetics of the C3 antibodies of the present disclosure.
- FIG. 4 Representative table containing results of C3 antibody cross-reactivity with human C3b protein, Cynomolgus monkey C3 protein, and human C3a protein.
- FIGS. 5A-5B Representative tables containing binding kinetics of C3 antibodies with respect to human C3 protein (FIG. 5A) and human C3b protein (FIG. 5B).
- FIGS. 6A-6D Representative results of C3 antibody epitope experiments, including a table containing binning allocations (FIG. 6A), data from a representative heatmap analyzing the ability of the antibodies to block one another for binding to the antigen (FIG. 6B), and representative network plots (FIG. 6C and FIG. 6D), which progressively groups antibodies with similar competition profiles.
- FIGS. 7A-7E Representative binning network plots based on recombinant human affinities and cyno binding performed with C3b (FIG. 7A); C3b affinity and ligand blocking (FIG. 7B); bins vs. communities for CFHR4b blocking (FIG. 7C); bins vs. communities for CFH blocking (FIG. 7D); and material availability and common vs. diverse light chains (FIG. 7E).
- FIG. 8 Representative results of C3 antibody polyreactivity with baculo virus particles (BVPs).
- FIG. 9 Representative results of C3 antibody polyreactivity with the various proteins indicated.
- FIGS. 10A-10B Representative results of C3 antibody polyreactivity with CFHR4 (FIG. 10A) and CFH (FIG. 10B).
- FIG. 11 Representative results of the effects of C3 antibodies on alternative pathway complement activation based on the quantification of red blood cell hemolysis.
- Embodiments of the present disclosure relate to the treatment and/or prevention of age- related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA).
- AMD age-related macular degeneration
- GA Geographic Atrophy
- the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, and antigen binding fragments thereof.
- the human complement system comprises a family of proteins that are essential to the human immune response against infections. Complement recognizes microbes or damaged host cells and subsequently triggers an enzymatic cascade that mainly serves to label target cells for phagocytosis by immune cells, produce chemoattractants, and directly kill target cells via pore formation. Unwanted complement activation on the body’s own cells is a key pathological driver in a wide spectrum of immune diseases including autoimmune, inflammatory, and degenerative diseases. For current and future development of therapeutic complement inhibitors, knowledge of complement activation and how it can be regulated is of great importance.
- Convertase enzymes fulfill a central role in the complement cascade as they cleave C3 and C5, which mediate nearly all complement effector functions.
- C3 convertases cleave C3 into C3a, a chemoattractant molecule, and C3b, which covalently binds to target surfaces and triggers phagocytosis.
- C5 convertases cleave C5 into C5a, a potent mediator of leukocyte recruitment and inflammation, and C5b, the initiator of the membrane attack complex and cell lysis.
- the complement cascade begins via specific recognition of target cells in the classical (CP) and lectin (LP) pathways.
- CP antibodies bind epitopes on the target cell and subsequently recruit the Cl complex (Clqr2S2).
- Clq-associated protease Cis converts C4 and C2 to generate a C3 convertase enzyme (C4b2a) on the cell surface.
- the lectin pathway also forms C4b2a via activation of mannose-binding lectin-associated serine proteases.
- the resulting CP/LP C4b2a convertases cleave C3 into C3a and C3b. Following cleavage, a reactive thioester in C3b is exposed, which enables its covalent attachment to target cell surfaces, leading to recognition of the cells by phagocytes.
- the labeling of target cells with C3b is amplified by the alternative pathway (AP) in which surface-bound C3b binds factor B (FB).
- AP surface-bound C3b binds factor B
- FB surface-bound C3b binds factor B
- FD factor D
- C3bBb active C3 convertase complex that consists of C3b and the protease fragment Bb
- C3bBb active AP C3 convertase
- C3bBb active AP C3 convertase
- substrate cleavage results in generation of additional convertases, further propagating C3b deposition.
- the existing C3 convertases C4b2a and C3bBb
- Age-related macular degeneration is a progressive retinal disease in which the early stage is characterized by relatively few small drusen within the macula. When AMD progresses, drusen size and number increase, eventually leading towards more advanced stages of AMD.
- Two forms of advanced AMD are distinguished.
- the first form, neovascular AMD is characterized by infiltration of abnormal blood vessels into the retina. These newly formed vessels are fragile and when they break, the leakage of blood constituents in the retina leads to sudden vision loss.
- the second form of advanced AMD is the result of gradual degeneration of the RPE and photoreceptors cells. Although neovascularization occurs in only 15- 20% of AMD cases, it is responsible for the vast majority of vision loss caused by AMD.
- VEGF vascular endothelial growth factor
- one or more refers to a number higher than one.
- the term “one or more” encompasses any of the following: two or more, three or more, four or more, five or more, six or more, seven or more, eight or more, nine or more, ten or more, eleven or more, twelve or more, thirteen or more, fourteen or more, fifteen or more, twenty or more, fifty or more, 100 or more, or an even greater number.
- the higher number can be 10,000, 1,000, 100, 50, etc.
- the higher number can be approximately 50 (e.g., 50, 49, 48, 47, 46, 45, 44, 43, 42, 41, 40, 39, 38, 37, 36, 35, 34, 33, 32, 31, 32, 30, 29, 28, 27, 26, 25, 24, 23, 22, 21, 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3 or 2).
- immunoglobulin refers to a protein that is found in blood or other bodily fluids of vertebrates, which is used by the immune system to identify and neutralize foreign objects, such as bacteria and viruses.
- an immunoglobulin or antibody is a protein that comprises at least one complementarity determining region (CDR).
- CDRs form the “hypervariable region” of an antibody, which is responsible for antigen binding (discussed further below).
- a whole antibody typically consists of four polypeptides: two identical copies of a heavy (H) chain polypeptide and two identical copies of a light (L) chain polypeptide.
- Each of the heavy chains contains one N-terminal variable (VH) region and three C-terminal constant (CHI, CH2, and Cm) regions, and each light chain contains one N-terminal variable (VL) region and one C-terminal constant (CL) region.
- the light chains of antibodies can be assigned to one of two distinct types, either kappa (K) or lambda (X), based upon the amino acid sequences of their constant domains.
- K kappa
- X lambda
- each light chain is linked to a heavy chain by disulfide bonds, and the two heavy chains are linked to each other by disulfide bonds.
- the light chain variable region is aligned with the variable region of the heavy chain
- the light chain constant region is aligned with the first constant region of the heavy chain.
- the remaining constant regions of the heavy chains are aligned with each other.
- variable regions of each pair of light and heavy chains form the antigen binding site of an antibody.
- the VH and VL regions have the same general structure, with each region comprising four framework (FW or FR) regions.
- framework region refers to the relatively conserved amino acid sequences within the variable region which are located between the CDRs.
- the framework regions form the [:) sheets that provide the structural framework of the variable region (see, e.g., C. A. Janeway et al. (eds.), Immunobiology, 5th Ed., Garland Publishing, New York, N.Y. (2001)).
- the framework regions are connected by three CDRs.
- the three CDRs known as CDR1, CDR2, and CDR3, form the “hypervariable region” of an antibody, which is responsible for antigen binding.
- the CDRs form loops connecting, and in some cases comprising part of, the beta-sheet structure formed by the framework regions.
- the constant regions of the light and heavy chains are not directly involved in binding of the antibody to an antigen, the constant regions can influence the orientation of the variable regions.
- the constant regions also exhibit various effector functions, such as participation in antibody-dependent complement- mediated lysis or antibody-dependent cellular toxicity via interactions with effector molecules and cells.
- an antibody or other entity e.g., antigen binding domain
- an antibody or other entity e.g., antigen binding domain
- affinity which is substantially higher means affinity that is high enough to enable detection of an antigen or epitope which is distinguished from entities using a desired assay or measurement apparatus.
- binding affinity having a binding constant (K a ) of at least 10 7 M' 1 (e.g., >10 7 M’ 1 , >10 8 M’ 1 , >10 9 M’ 1 , >1O 10 M’ 1 , >10 n M’ 1 , >10 12 M’ 1 , >10 13 M’ 1 , etc.).
- K a binding constant
- an antibody is capable of binding different antigens so long as the different antigens comprise that particular epitope.
- homologous proteins from different species may comprise the same epitope.
- fragment of an antibody refers to one or more fragments of an antibody that retain the ability to specifically bind to an antigen (see, generally, Holliger et al., Nat. Biotech., 25(9): 1126-1129 (2005)). Any antigen-binding fragment of the antibody described herein is within the scope of the present disclosure.
- the antibody fragment desirably comprises, for example, one or more CDRs, the variable region (or portions thereof), the constant region (or portions thereof), or combinations thereof.
- antibody fragments include, but are not limited to, (i) a Fab fragment, which is a monovalent fragment consisting of the VL, VH, CL, and CHI domains, (ii) a F(ab’)2 fragment, which is a bivalent fragment comprising two Fab fragments linked by a disulfide bridge at the hinge region, (iii) a Fv fragment consisting of the VL and VH domains of a single arm of an antibody, (iv) a Fab’ fragment, which results from breaking the disulfide bridge of an F(ab’)2 fragment using mild reducing conditions, (v) a disulfide-stabilized Fv fragment (dsFv), and (vi) a domain antibody (dAb), which is an antibody single variable region domain (VH or VL) polypeptide that specifically binds antigen.
- a Fab fragment which is a monovalent fragment consisting of the VL, VH, CL, and CHI domains
- the term “monoclonal antibody,” as used herein, refers to an antibody produced by a single clone of B lymphocytes that is directed against a single epitope on an antigen.
- Monoclonal antibodies typically are produced using hybridoma technology, as first described in Kohler and Milstein, Eur. J. Immunol., 5: 511-519 (1976).
- Monoclonal antibodies may also be produced using recombinant DNA methods (see, e.g., U.S. Patent 4,816,567), isolated from phage display antibody libraries (see, e.g., Clackson et al. Nature, 352: 624-628 (1991)); and Marks et al., J. Mol.
- polyclonal antibodies are antibodies that are secreted by different B cell lineages within an animal. Polyclonal antibodies are a collection of immunoglobulin molecules that recognize multiple epitopes on the same antigen.
- nucleic acid refers to a polymer or oligomer of pyrimidine and/or purine bases, preferably cytosine, thymine, and uracil, and adenine and guanine, respectively (See Albert L. Lehninger, Principles of Biochemistry, at 793-800 (Worth Pub. 1982)).
- the terms encompass any deoxyribonucleotide, ribonucleotide, or peptide nucleic acid component, and any chemical variants thereof, such as methylated, hydroxymethylated, or glycosylated forms of these bases.
- the polymers or oligomers may be heterogenous or homogenous in composition, may be isolated from naturally occurring sources, or may be artificially or synthetically produced.
- the nucleic acids may be DNA or RNA, or a mixture thereof, and may exist permanently or transitionally in single-stranded or double-stranded form, including homoduplex, heteroduplex, and hybrid states.
- a nucleic acid or nucleic acid sequence comprises other kinds of nucleic acid structures such as, for instance, a DNA/RNA helix, peptide nucleic acid (PNA), morpholino nucleic acid (see, e.g., Braasch and Corey, Biochemistry, 41()A): 4503-4510 (2002) and U.S. Patent 5,034,506), locked nucleic acid (LNA; see Wahlestedt et al., Proc. Natl. Acad. Sci. U.S.A., 97: 5633-5638 (2000)), cyclohexenyl nucleic acids (see Wang, J. Am. Chem.
- nucleic acid and “nucleic acid sequence” may also encompass a chain comprising nonnatural nucleotides, modified nucleotides, and/or non-nucleotide building blocks that can exhibit the same function as natural nucleotides (e.g., “nucleotide analogs”).
- peptide refers to a polymeric form of amino acids of any length, which can include coded and non-coded amino acids, chemically or biochemically modified or derivatized amino acids, and polypeptides having modified peptide backbones.
- nucleic acid or “nucleic acid molecule” generally refers to any ribonucleic acid or deoxyribonucleic acid, which may be unmodified or modified DNA or RNA.
- Nucleic acids include, without limitation, single- and double-stranded nucleic acids.
- nucleic acid also includes DNA as described above that contains one or more modified bases.
- nucleic acid DNA with a backbone modified for stability or for other reasons is a “nucleic acid.”
- nucleic acid as it is used herein embraces such chemically, enzymatically, or metabolically modified forms of nucleic acids, as well as the chemical forms of DNA characteristic of viruses and cells, including for example, simple and complex cells.
- oligonucleotide or “polynucleotide” or “nucleotide” or “nucleic acid” refer to a molecule having two or more deoxyribonucleotides or ribonucleotides, preferably more than three, and usually more than ten. The exact size will depend on many factors, which in turn depends on the ultimate function or use of the oligonucleotide.
- the oligonucleotide may be generated in any manner, including chemical synthesis, DNA replication, reverse transcription, or a combination thereof.
- Typical deoxyribonucleotides for DNA are thymine, adenine, cytosine, and guanine.
- Typical ribonucleotides for RNA are uracil, adenine, cytosine, and guanine.
- complementarity refers to nucleotides (e.g., 1 nucleotide) or polynucleotides (e.g., a sequence of nucleotides) related by the base-pairing rules.
- sequence 5’-A-G-T-3’ is complementary to the sequence 3'-T-C-A-5'.
- Complementarity may be “partial,” in which only some of the nucleic acids’ bases are matched according to the base pairing rules. Or, there may be “complete” or “total” complementarity between the nucleic acids.
- the degree of complementarity between nucleic acid strands affects the efficiency and strength of hybridization between nucleic acid strands. This is of particular importance in amplification reactions and in detection methods that depend upon binding between nucleic acids.
- the term “gene” refers to a nucleic acid (e.g., DNA or RNA) sequence that comprises coding sequences necessary for the production of an RNA, or of a polypeptide or its precursor.
- a functional polypeptide can be encoded by a full-length coding sequence or by any portion of the coding sequence as long as the desired activity or functional properties (e.g., enzymatic activity, ligand binding, signal transduction, etc.) of the polypeptide are retained.
- portion when used in reference to a gene refers to fragments of that gene. The fragments may range in size from a few nucleotides to the entire gene sequence minus one nucleotide. Thus, “a nucleotide comprising at least a portion of a “gene” may comprise fragments of the gene or the entire gene.
- the term “gene” also encompasses the coding regions of a structural gene and includes sequences located adjacent to the coding region on both the 5' and 3' ends, e.g., for a distance of about 1 kb on either end, such that the gene corresponds to the length of the full-length mRNA (e.g., comprising coding, regulatory, structural and other sequences).
- the sequences that are located 5' of the coding region and that are present on the mRNA are referred to as 5' non- translated or untranslated sequences.
- the sequences that are located 3' or downstream of the coding region and that are present on the mRNA are referred to as 3' non-translated or 3' untranslated sequences.
- genomic form or clone of a gene contains the coding region interrupted with noncoding sequences termed “introns” or “intervening regions” or “intervening sequences.”
- Introns are segments of a gene that are transcribed into nuclear RNA (hnRNA); introns may contain regulatory elements such as enhancers. Introns are removed or “spliced out” from the nuclear or primary transcript; introns therefore are absent in the messenger RNA (mRNA) transcript.
- mRNA messenger RNA
- genomic forms of a gene may also include sequences located on both the 5' and 3' ends of the sequences that are present on the RNA transcript. These sequences are referred to as “flanking” sequences or regions (these flanking sequences are located 5' or 3' to the non-translated sequences present on the mRNA transcript).
- the 5' flanking region may contain regulatory sequences such as promoters and enhancers that control or influence the transcription of the gene.
- the 3' flanking region may contain sequences that direct the termination of transcription, posttranscriptional cleavage, and polyadenylation.
- wild-type when made in reference to a gene refers to a gene that has the characteristics of a gene isolated from a naturally occurring source.
- wild-type when made in reference to a gene product refers to a gene product that has the characteristics of a gene product isolated from a naturally occurring source.
- wild-type when made in reference to a protein refers to a protein that has the characteristics of a naturally occurring protein.
- naturally-occurring as applied to an object refers to the fact that an object can be found in nature.
- a polypeptide or polynucleotide sequence that is present in an organism (including viruses) that can be isolated from a source in nature, and which has not been intentionally modified by the hand of a person in the laboratory is naturally-occurring.
- a wild-type gene is often that gene or allele that is most frequently observed in a population and is thus arbitrarily designated the “normal” or “wild-type” form of the gene.
- the term “modified” or “mutant” when made in reference to a gene or to a gene product refers, respectively, to a gene or to a gene product that displays modifications in sequence and/or functional properties (e.g., altered characteristics) when compared to the wild-type gene or gene product.
- naturally-occurring mutants can be isolated; these are identified by the fact that they have altered characteristics when compared to the wild-type gene or gene product.
- allele refers to a variation of a gene; the variations include but are not limited to variants and mutants, polymorphic loci, and single nucleotide polymorphic loci, frameshift, and splice mutations. An allele may occur naturally in a population, or it might arise during the lifetime of any particular individual of the population.
- variant and mutant when used in reference to a nucleotide sequence refer to a nucleic acid sequence that differs by one or more nucleotides from another, usually related, nucleotide acid sequence.
- a “variation” is a difference between two different nucleotide sequences; typically, one sequence is a reference sequence.
- immunogen and “antigen” are used interchangeably herein and refer to any molecule, compound, or substance that induces an immune response in an animal (e.g., a mammal).
- An “immune response” can entail, for example, antibody production and/or the activation of immune effector cells.
- An antigen in the context of the disclosure can comprise any subunit, fragment, or epitope of any proteinaceous or non-proteinaceous (e.g., carbohydrate or lipid) molecule that provokes an immune response in a mammal.
- epitope refers to a sequence of an antigen that is recognized by an antibody or an antigen receptor.
- an epitope is a region of an antigen that is specifically bound by an antibody.
- an epitope may include chemically active surface groupings of molecules such as amino acids, sugar side chains, phosphoryl, or sulfonyl groups.
- an epitope may have specific three- dimensional structural characteristics (e.g., a “conformational” epitope) and/or specific charge characteristics.
- the antigen can be a protein or peptide of viral, bacterial, parasitic, fungal, protozoan, prion, cellular, or extracellular origin, which provokes an immune response in a mammal, preferably leading to protective immunity.
- a “pharmaceutically acceptable carrier” as used herein generally refers to an ingredient in a pharmaceutical formulation, other than an active ingredient, which is nontoxic to a subject.
- a pharmaceutically acceptable carrier includes, but is not limited to, a buffer, excipient, stabilizer, or preservative.
- pharmaceutical formulation generally refers to a preparation which is in such form as to permit the biological activity of an active ingredient (e.g., an anti-C3 antibody, an antibody conjugate, a fusion protein, or a polymeric formulation) contained therein to be effective, and which contains no additional components which are unacceptably toxic to a subject to which the formulation would be administered.
- an active ingredient e.g., an anti-C3 antibody, an antibody conjugate, a fusion protein, or a polymeric formulation
- treatment generally refers to clinical intervention in an attempt to alter the natural course of the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis.
- anti-C3 antibodies of the present disclosure or other compositions that include an anti-C3 antibody of the present disclosure are used to delay development of a disease or to slow the progression of a disease.
- half-life generally refers to the time required for the concentration of a substance (e.g., an anti-3 antibody, an antibody conjugate, a fusion protein (e.g., a Fab fusion protein), or a polymeric formulation) to decrease by one-half, in vivo (e.g., in the eye (e.g., the vitreous)) or in vitro.
- a substance e.g., an anti-3 antibody, an antibody conjugate, a fusion protein (e.g., a Fab fusion protein), or a polymeric formulation
- an “effective amount” of an agent e.g., a pharmaceutical formulation, as used herein generally refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
- An “individual” or “subject” is a mammal. Mammals include, but are not limited to, domesticated animals (e.g., cows, sheep, cats, dogs, and horses), primates (e.g., humans and non- human primates such as monkeys), rabbits, and rodents (e.g., mice and rats). In certain embodiments, the individual or subject is a human. A “subject” may be a “patient.”
- embodiments of the present disclosure relate to the treatment and/or prevention of age-related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA).
- AMD age-related macular degeneration
- GA Geographic Atrophy
- the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b).
- anti-C3 antibodies were generated, and their structural and functional properties were elucidated. Based on these data, embodiments of the present disclosure include anti-C3 antibodies, or antigen-binding fragments thereof, that are comprised of a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3.
- VH heavy chain variable region
- CDRs complementarity determining regions
- VL light chain variable region
- the HCDR1 comprises one of the following amino acid sequences: (a) X1YX2X3X4 (SEQ ID NO: 1), wherein Xi is G, S, N, D, T, or A; X 2 is Y, A, G, W, or T; X 3 is L, M, I, or V; and X 4 is H, N, S, or G; (b) X1X2 X3MX4 (SEQ ID NO: 57), wherein Xi is D, N, S, or A; X 2 is Y, or A; X 3 is A, W, Y, S, C, D, or G; and X 4 is H, S, or N; (c) X1X2X3WWX4 (SEQ ID NO: 95), wherein Xi is I, S, R, N, or K; X 2 is N, D, S, or G; X 3 is N, S, T, D, Y, or H; and
- the HCDR2 comprises one of the following amino acid sequences: (a) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 20), wherein Xi is W, I, or V; X 2 is N, S, or Y; X 3 is P, T, A, G, or Y; X 4 is N, Y, G, D, H, or A; X 5 is S, T, N, G, or D; X 6 is G, or S; X 7 is D, G, T, N, V, A, or H; X 8 is T, P, or K; X9 is N, Y, T, H, R, I, D, or K; X10 is Y, or A; Xu is V, Q, A, or S; X12 is Q, K, P, or D; X13 is N, F, K, G, S, or Q; X14 is
- the HCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 39-56; (b) SEQ ID NOs: 83-94; (c) SEQ ID NOs: 153-180; (d) SEQ ID NOs: 197- 203; (e) SEQ ID NOs: 220-226; or (f) SEQ ID NOs: 234-235.
- Embodiments of the present disclosure also include antibodies directed against Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, including antigen binding fragments thereof, comprising a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3.
- VH heavy chain variable region
- CDRs complementarity determining regions
- VL light chain variable region
- the LCDR1 comprises one of the following amino acid sequences: (a) XIASQX2X3X4X 5 X6X 7 X 8 (SEQ ID NO: 236), wherein Xi is R or Q; X 2 is D, N, T, G, or S; X 3 is V or I; X 4 is S, I, R, G, T, or K; X 5 is T, N, R, or S; X 6 is A, W, F, D, Y, N, or C; X 7 is V or L; X 8 is A, N, G, or H; (b) RSSQX1LLHX2NGYNYLD (SEQ ID NO: 331), wherein Xi is S or I; X 2 is S or T; or (c) RASQSX1X2X3X4X5X6 (SEQ ID NO: 340), wherein Xi is V or I; X 2 is S or R; X 3 is S, D, or R
- the LCDR2 comprises one of the following amino acid sequences: (a) X1ASX2X3X4X5 (SEQ ID NO: 268), wherein Xi is S, K, A, Y, T, D, or V; X 2 is F, S, N, Q, or T; X 3 is L, S, or F; X 4 is Y, E, Q, or F; X 5 is S or T; (b) LGSXiRAS (SEQ ID NO: 334), wherein Xi is N or Y; (c) X1ASX2RAT (SEQ ID NO: 349), wherein Xi is D or G; X 2 is S or N; or (d) WASXiRES (SEQ ID NO: 402), wherein Xi is N or T.
- SEQ ID NO: 268 X1ASX2X3X4X5
- Xi is S, K, A, Y, T, D, or V
- X 2 is F, S,
- the LCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 300-330; (b) MQX1LQTPX2T (SEQ ID NO: 337), wherein Xi is T or A; X 2 is P or L; (c) QQX1X2X3X4PX5T (SEQ ID NO: 358), wherein Xi is R or Y; X 2 is S, G, or D; X 3 is N or S; X 4 is W or S; X 5 is L, R, I, Y, F, or W; or (d) QQX1X2X3TX4X5 (SEQ ID NO: 437), wherein Xi is Y or F; X 2 is Y or F; X 3 is S, G, or D; X4 is P or R; X5 is Y, W, R, T, or P.
- the HCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 2-19, SEQ ID NOs: 58-69, SEQ ID NOs: 96-123, SEQ ID NOs: 182-188, SEQ ID NOs: 205-211, or SEQ ID NOs: 228-229;
- the HCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 21-38, SEQ ID NOs: 71- 82, SEQ ID NOs: 125-152, SEQ ID NOs 190-196, SEQ ID NOs 213-219, or SEQ ID NOs 231- 232;
- the HCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 39-56, SEQ ID NOs: 83-94, SEQ ID NOs: 153-180, SEQ ID NOs: 197-203, SEQ ID NOs: 220-226, or SEQ ID NOs: 234-235.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 2; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 21; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 39.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 3; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 22; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 40.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 4; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 23; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 41.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 5; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 24; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 42.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 6; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 25; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 43.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 7; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 26; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 44.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 8; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 27; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 45.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 9; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 28; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 46.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 10; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 29; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 47.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 11; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 30; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 48.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 12; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 31 ; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 49.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 13; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 32; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 50.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 14; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 33; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 51.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 15; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 34; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 52.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 16; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 35; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 53.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 17; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 36; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 54.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 18; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 37; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 55.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 19; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 38; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 56.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 58; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 71; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 83.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 59; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 72; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 84.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 60; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 73; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 85.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 61; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 74; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 86. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 62; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 75; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 87.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 63; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 76; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 88.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 64; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 77; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 89.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 65; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 78; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 90.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 66; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 79; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 91.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 67; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 80; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 92.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 68; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 81; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 93. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 69; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 82; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 94.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 96; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 125; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 153. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 97; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 126; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 154.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 98; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 127; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 155. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 99; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 128; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 156.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 100; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 129; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 157.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 101; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 130; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 158.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 102; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 131; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 159.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 103; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 132; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 160.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 104; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 133; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 161.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 105; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 134; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 162.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 106; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 135; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 163.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 107; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 136; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 164.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 108; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 137; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 165.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 109; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 138; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 166.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 110; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 139; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 167. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 111; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 140; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 168.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 112; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 141; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 169. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 113; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 142; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 170.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 114; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 143; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 171.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 115; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 144; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 172.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 116; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 145; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 173.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 117; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 146; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 174.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 118; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 147; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 175.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 119; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 148; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 176.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 120; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 149; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 177.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 121; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 150; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 178.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 122; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 151; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 179.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 123; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 152; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 180.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 182; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 190; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 197. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 183; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 191; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 198.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 184; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 192; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 199.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 185; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 193; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 200.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 186; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 194; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 201.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 187; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 195; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 202.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 188; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 196; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 203. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 205; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 213; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 220.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 206; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 214; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 221.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 207; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 215; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 222.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 208; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 216; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 223.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 209; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 217; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 224.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 210; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 218; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 225. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 211; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 219; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 226.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 228; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 231; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 234.
- an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 229; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 232; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 35.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 237; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 269; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 300.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 238; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 270; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 301.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 239; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 271; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 302.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 240; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 272; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 303.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 241; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 273; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 304.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 242; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 274; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 305.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 243; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 275; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 306.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 244; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 276; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 307.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 245; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 277; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 308.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 246; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 278; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 309.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 247; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 279; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 310.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 248; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 280; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 311.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 249; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 281; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 312.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 250; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 282; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 313.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 251; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 283; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 314.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 252; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 284; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 315.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 253; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 285; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 316.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 254; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 286; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 317.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 255; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 287; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 318.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 256; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 288; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 319.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 257; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 289; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 320.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 258; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 290; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 321.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 259; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 291; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 322.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 260; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 292; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 323.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 261; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 293; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 324.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 262; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 294; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 325.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 263; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 295; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 326.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 264; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 296; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 327.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 265; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 297; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 328.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 266; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 298; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 329.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 267; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 299; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 330.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 332; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 335; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 338.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 333; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 336; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 339.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 341; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 350; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 359.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 342; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 351; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 360.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 343; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 352; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 361.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 344; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 353; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 362.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 345; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 354; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 363.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 346; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 355; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 364.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 347; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 356; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 365.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 348; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 357; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 366.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 368; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 403; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 438.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 369; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 404; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 439.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 370; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 405; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 440.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 371; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 406; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 441.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 372; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 407; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 442.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 373; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 408; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 443.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 374; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 409; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 444.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 375; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 410; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 445.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 376; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 411; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 446.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 377; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 412; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 447.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 378; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 413; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 448.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 379; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 414; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 449.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 380; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 415; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 450.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 381; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 416; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 451.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 382; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 417; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 452.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 383; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 418; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 453.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 384; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 419; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 454.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 385; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 420; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 455.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 386; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 421; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 456.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 387; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 422; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 457.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 388; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 423; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 458.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 389; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 424; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 459.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 390; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 425; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 460.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 391; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 426; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 461.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 392; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 427; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 462.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 393; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 428; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 463.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 394; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 429; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 464.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 395; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 430; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 465.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 396; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 431 ; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 466.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 397; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 432; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 467.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 398; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 433; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 468.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 399; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 434; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 469.
- an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 400; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 435; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 470.
- an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 401; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 436; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 471.
- the VH of the anti-C3 antibodies of the present disclosure includes an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to any of SEQ ID NOs: 546-619.
- the VL of the anti-C3 antibodies of the present disclosure includes an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to any of SEQ ID NOs: 694-767.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 546 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 694.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 547 and the and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 695.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 548 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 696.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 549 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 697.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 550 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 698.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 551 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 699.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 552 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 700.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 553 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 701.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 554 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 702.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 555 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 703.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 556 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 704.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 557 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 705.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 558 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 706.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 559 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 707.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 560 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 708.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 561 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 709.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 562 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 710.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 563 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 711.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 564 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 712.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 565 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 713.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 566 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 714.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 567 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 715.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 568 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 716.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 569 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 717.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 570 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 718.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 571 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 719.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 572 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 720.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 573 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 721.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 574 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 722.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 575 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 723.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 576 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 724.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 577 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 725.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 578 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 726.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 579 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 727.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 580 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 728.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 581 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 729.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 582 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 730.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 583 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 731.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 584 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 732.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 585 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 733.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 586 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 734.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 587 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 735.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 588 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 736.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 589 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 737.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 590 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 738.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 591 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 739.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 592 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 740.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 593 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 741.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 594 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 742.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 595 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 743.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 596 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 744.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 597 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 745.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 598 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 746.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 599 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 747.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 600 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 748.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 601 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 749.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 602 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 750.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 603 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 751.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 604 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 752.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 605 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 753.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 606 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 754.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 607 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 755.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 608 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 756.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 609 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 757.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 610 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 758.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 611 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 759.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 612 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 760.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 613 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 761.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 614 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 762.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 615 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 763.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 616 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 764.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 617 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 765.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 618 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 766.
- the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 619 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 767.
- Nucleic acid or amino acid sequence “identity,” as described herein, can be determined by comparing a nucleic acid or amino acid sequence of interest to a reference nucleic acid or amino acid sequence.
- a number of mathematical algorithms for obtaining the optimal alignment and calculating identity between two or more sequences are known and incorporated into a number of available software programs. Examples of such programs include CLUSTAL-W, T-Coffee, and ALIGN (for alignment of nucleic acid and amino acid sequences), BLAST programs (e.g., BLAST 2.1, BL2SEQ, and later versions thereof) and FASTA programs (e.g., FASTA3x, FASTM, and SSEARCH) (for sequence alignment and sequence similarity searches).
- Sequence alignment algorithms also are disclosed in, for example, Altschul et al., J. Molecular Biol., 215(3): 403-410 (1990), Beigert et al., Proc. Natl. Acad. Sci. USA, 106( Q>): 3770-3775 (2009), Durbin et al., eds., Biological Sequence Analysis: Probabilistic Models of Proteins and Nucleic Acids, Cambridge University Press, Cambridge, UK (2009), Soding, Bioinformatics, 21(7): 951-960 (2005), Altschul et al., Nucleic Acids Res., 25(17): 3389-3402 (1997), and Gusfield, Algorithms on Strings, Trees and Sequences, Cambridge University Press, Cambridge UK (1997)).
- one or more amino acids of the aforementioned anti-C3 antibodies can be replaced or substituted with a different amino acid.
- An amino acid “replacement” or “substitution” refers to the replacement of one amino acid at a given position or residue by another amino acid at the same position or residue within a polypeptide sequence.
- Amino acids are broadly grouped as “aromatic” or “aliphatic.” An aromatic amino acid includes an aromatic ring.
- aromatic amino acids include histidine (H or His), phenylalanine (F or Phe), tyrosine (Y or Tyr), and tryptophan (W or Trp).
- Non-aromatic amino acids are broadly grouped as “aliphatic.” Examples of “aliphatic” amino acids include glycine (G or Gly), alanine (A or Ala), valine (V or Vai), leucine (L or Leu), isoleucine (I or He), methionine (M or Met), serine (S or Ser), threonine (T or Thr), cysteine (C or Cys), proline (P or Pro), glutamic acid (E or Glu), aspartic acid (A or Asp), asparagine (N or Asn), glutamine (Q or Gin), lysine (K or Lys), and arginine (R or Arg).
- Aliphatic amino acids may be sub-divided into four sub-groups.
- the “large aliphatic non-polar sub-group” consists of valine, leucine, and isoleucine.
- the “aliphatic slightly-polar sub-group” consists of methionine, serine, threonine, and cysteine.
- the “aliphatic polar/charged sub-group” consists of glutamic acid, aspartic acid, asparagine, glutamine, lysine, and arginine.
- the “small-residue sub-group” consists of glycine and alanine.
- the group of charged/polar amino acids may be sub-divided into three sub-groups: the “positively-charged sub-group” consisting of lysine and arginine, the “negatively-charged subgroup” consisting of glutamic acid and aspartic acid, and the “polar sub-group” consisting of asparagine and glutamine.
- Aromatic amino acids may be sub-divided into two sub-groups: the “nitrogen ring sub-group” consisting of histidine and tryptophan and the “phenyl sub-group” consisting of phenylalanine and tyrosine.
- the amino acid replacement or substitution can be conservative, semi-conservative, or non-conservative.
- the phrase “conservative amino acid substitution” or “conservative mutation” refers to the replacement of one amino acid by another amino acid with a common property.
- a functional way to define common properties between individual amino acids is to analyze the normalized frequencies of amino acid changes between corresponding proteins of homologous organisms (Schulz and Schirmer, Principles of Protein Structure, Springer-Verlag, New York (1979)). According to such analyses, groups of amino acids may be defined where amino acids within a group exchange preferentially with each other, and therefore resemble each other most in their impact on the overall protein structure.
- conservative amino acid substitutions include substitutions of amino acids within the sub-groups described above, for example, lysine for arginine and vice versa such that a positive charge may be maintained, glutamic acid for aspartic acid and vice versa such that a negative charge may be maintained, serine for threonine such that a free -OH can be maintained, and glutamine for asparagine such that a free -NH2 can be maintained.
- “Semi-conservative mutations” include amino acid substitutions of amino acids within the same groups listed above, but not within the same sub-group. For example, the substitution of aspartic acid for asparagine, or asparagine for lysine, involves amino acids within the same group, but different sub-groups.
- “Non-conservative mutations” involve amino acid substitutions between different groups, for example, lysine for tryptophan, or phenylalanine for serine, etc.
- one or more amino acids can be inserted into the anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies), or antigen-binding fragments thereof (e.g., insertion into the heavy and/or light chain variable region amino acid sequence).
- Any number of suitable amino acids can be inserted into the amino acid sequence of the antibody or antigen-binding fragment thereof.
- at least one amino acid e.g., 2 or more, 5 or more, or 10 or more amino acids
- 20 amino acids e.g., 18 or less, 15 or less, or 12 or less amino acids
- 1- 10 amino acids may be inserted into the amino acid sequence of the monoclonal antibody or antigen-binding fragment thereof.
- the amino acid(s) can be inserted into an antibody or antigen-binding fragment thereof in any suitable location.
- the amino acid(s) are inserted into a CDR (e.g., CDR1, CDR2, or CDR3) of the antibody or antigen-binding fragment thereof.
- amino acid sequences of the anti-C3 antibodies are not limited to the specific amino acid sequences described herein.
- an anti-C3 antibody e.g., C3, C3a, and C3b antibody
- antigenbinding fragment thereof can comprise any heavy chain polypeptide or light chain polypeptide that competes with the anti-C3 antibodies or antigen-binding fragments thereof for conformational binding to C3.
- Antibody competition can be assayed using routine peptide competition assays such as, for example, ELISA, Western blot, or immunohistochemistry methods (see, e.g., U.S. Patents 4,828,981 and 8,568,992; and Braitbard et al., Proteome Sci., 4 12 (2006)).
- An anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure may be a whole antibody, or an antigen-binding fragment of a whole antibody.
- antigen-binding antibody fragments encompassed by the present disclosure include, but are not limited to, F(ab’)2, Fab’, Fab, Fv, scFv, dsFv, dAb, and single chain binding polypeptides.
- Antibody fragments and their therapeutic utility are further described in, e.g., Nelson, A.L., MAbs.
- the anti-C3 antigen-binding fragment is a single-chain variable fragment (scFv), which is an engineered antibody generated by the fusion of the heavy (VH) and light chains (VL) of immunoglobulins through a short polypeptide linker.
- scFv single-chain variable fragment
- Single chain variable domain (Fv) fragments are used in the art in a variety of clinical and therapeutic applications, primarily due to their improved pharmacokinetic properties as compared to the parent monoclonal antibodies and the relative ease of producing them in large quantities at low cost (Monnier et al., Antibodies 2013, 2(2), 193-208; doi.org/10.3390/antib2020193; Safdari et al., Mol Med. 2016; 22: 258-270; and Lu, R., Hwang, Y., Liu, I. et al. Development of therapeutic antibodies for the treatment of diseases. J Biomed Sci 27, 1 (2020). https://doi.org/10.1186/sl2929-019-0592-z).
- An anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure may be a diabody.
- Diabodies are antibody fragments with two antigen-binding sites that may be bivalent or bispecific. See, for example, EP 404,097; WO 1993/01161; Hudson et al., Nat. Med. 9:129-134 (2003); and Hollinger et al., Proc. Natl. Acad. Sci. USA 90: 6444-6448 (1993). Triabodies and tetrabodies are also described in Hudson et al., Nat. Med. 9:129-134 (2003).
- An anti-C3 antibody of the present disclosure may be a single-domain antibody (also referred to as a nanobody).
- Single-domain antibodies are antibody fragments comprising all or a portion of the heavy chain variable domain or all or a portion of the light chain variable domain of an antibody.
- a single-domain antibody is a human single-domain antibody (Domantis, Inc., Waltham, Mass.; see, e.g., U.S. Pat. No. 6,248,516 Bl).
- Antibody fragments can be made by various techniques, including but not limited to proteolytic digestion of an intact antibody as well as production by recombinant host cells (e.g., E. coli or phage), as described herein.
- the anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure is a whole antibody.
- a whole antibody comprises two identical copies of a heavy (H) chain polypeptide and two identical copies of a light (L) chain polypeptide.
- Each of the heavy chains contains one N-terminal variable (VH) region and three C- terminal constant (CHI, CH2, and Cm) regions, and each light chain contains one N-terminal variable (VL) region and one C-terminal constant (CL).
- the heavy chain C-terminal constant region contains the fragment crystallizable (Fc) domain, which determines antibody class and is responsible for humoral and cellular effector functions.
- Antibodies are divided into five major classes (or “isotypes”), IgG, IgM, IgA, IgD and IgE, which differ in their function in the immune system.
- IgGs are the most abundant immunoglobulins in the blood, representing 60% of total serum antibodies in humans.
- IgG antibodies may be subclassified as IgGl, IgG2, IgG3, and IgG4, named in order of their abundance in serum (IgGl being the most abundant) (Vidarsson et al., Frontiers in Immunology. 5: 520 (2014)).
- a whole anti-C3 monoclonal antibody described herein may be of any suitable class and/or subclass.
- the monoclonal antibody is of class IgG (e.g., IgGl, IgG2, IgG3, or IgG4).
- the monoclonal antibody may be an IgGl antibody.
- the Fc domain mediates several effector functions of antibodies, such as binding to receptors on target cells and complement fixation (triggering effector functions that eliminate the antigen).
- the Fc domain may be modified or engineered to alter its effector functions.
- Fc domains may be modified to improve antibodydependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP), and to control serum half-life.
- the Fc domain of the anti-C3 antibody may be engineered to modulate affinity for an Fc receptor, such as Fey receptors (FcyRs) and the neonatal Fc receptor (FcRn).
- the Fc domain also may be modified to improve serum half-life, e.g., by engineering IgG Fc for higher FcRn binding (Zalevsky et al., Nat. Biotechnol. 28, 157-159 (2010); and Dall’Acqua et al., J. Immunol. 169, 5171-5180 (2002)).
- the Fc domain may be modified to create monovalency or antibody bispecificity for improving therapeutic potency.
- an Fc domain may be generated that does not form a homodimer but remains as a soluble monomer, mFc, that exhibits high affinity for FcyRI but no detectable binding to FcyRIIIa.
- a heterodimeric Fc domain may be generated to obtain bispecific properties for antigen binding to circumvent homodimer formation.
- Engineered Fc domains may be generated by inducing point mutations or by modifying glycosylation of the Fc domain (Saunders, K.O., Front Immunol. 2019; 10: 1296; Kelley, R.F., Meng, Y.G., Liu et al., J Biol Chem. 2014;289:3571-90; Monnet et al., MAbs. 2014;6:422-36; Li et al., Proc Natl Acad Sci U S A. 2017;114:3485-90; and Lin et al., Proc Natl Acad Sci U S A. 2015;112:10611-6; Kang and Jung, supra).
- Embodiments of the present disclosure include compositions and methods pertaining to an antibody, or antigen binding fragment thereof, which specifically binds Complement Component 3 (C3) polypeptides, optionally wherein said C3 is a Complement Component 3a (C3a) polypeptide or a Complement Component 3b (C3b) polypeptide, which is optionally a polypeptide comprising or consisting of the amino acid sequence of SEQ ID NO: 1064.
- the present disclosure provides an antibody or a fragment thereof, which exhibits any one or more the following functional characteristics: (a) reduces complement activation and/or attenuates complement component 3 (C3) convertase activity, which is optionally measured in an ELISA; and/or (b) cross-reacts with at least one of human C3b protein, Cynomolgus monkey C3 protein, and/or human C3a protein; and/or (c) binds to human C3 with a KD -1 of about lOOnM or lower; and/or (d) binds to the same epitope on human C3 as an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) competes for binding to human C3 with an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6.
- C3 complement component 3
- the antibody or fragment thereof which is a monoclonal antibody. In some embodiments, the antibody or fragment thereof, which is a human, humanized or chimeric antibody. In further embodiments, the antibody or fragment thereof, is a whole antibody, a single chain variable fragment (scFv), a variable fragment (Fv), a fragment antigen-binding region (Fab), a Fab-C, a Fab’-SH, a (Fab’)2, a single-domain antibody (sdAb), a nanobody, a diabody, a triabody, or an aptamer, optionally wherein the antibody is a whole antibody comprising an Fc region such as a human IgGl , IgG2, IgG3 or IgG4 Fc region.
- an Fc region such as a human IgGl , IgG2, IgG3 or IgG4 Fc region.
- the antibody or fragment thereof is conjugated to at least one additional moiety, optionally selected from: (a) a therapeutic or cytotoxic moiety; (b) a detection moiety; (c) a purification moiety; (d) a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody.
- additional moiety optionally selected from: (a) a therapeutic or cytotoxic moiety; (b) a detection moiety; (c) a purification moiety; (d) a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody.
- this is referred to as “XTENylation,” as described further in US8933197, US7846445, US7855279, US8492530, US9938331, US8673860, US9371369, US9926351, US10961287, US10172953, and US 10953073.
- the present disclosure provides an antibody or a fragment thereof, which is a polypeptide comprising: (a) one, two or all three HCDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also one, two or all three of the corresponding LCDRs of said exemplary antibody; and/or (b) a VH sequence having at least 90% identity to the VH sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also a VL sequence having at least 90% identity to the corresponding VL sequence of said exemplary antibody, preferably wherein variation is not permitted in the HCDRs or LCDRs; and/or (c) all six CDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (d) the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) the full length heavy chain (VH + constant) sequence of any one of the
- an expression vector comprises a polynucleotide, which is optionally an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, or a retrovirus vector.
- AAV adeno-associated virus
- LV lentiviral
- HSV herpes simplex virus
- Embodiments of the present disclosure include a pharmaceutical composition comprising an antibody or fragment, a polynucleotide, or a vector and optionally (a) at least one pharmaceutically acceptable carrier, a buffer, excipient, stabilizer, diluent, or preservative; and/or (b) at least one additional active ingredient.
- the pharmaceutical composition which is suitable for ocular administration to a subject, optionally by delivery using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant, preferably by injection into the vitreous fluid.
- the antibody or fragment, the polynucleotide, the vector, the composition for use as a medicament, optionally for use in a method of treating a disease of the eye in a subject.
- the antibody or fragment, the polynucleotide, the vector, the composition for use, wherein said disease is characterized by increased activation of the complement system, in particular the alternative pathway, and in particular within the eye of the subject, for example in drusen or retinal pigment epithelium (RPE) cells of the subject.
- RPE retinal pigment epithelium
- the antibody or fragment, the polynucleotide, the vector, the composition for use wherein the method comprises ocular administration of the antibody, preferably by injection into the vitreous fluid, and wherein said administration preferably relieves at least one symptom in the subject selected from visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light.
- AMD age-related macular degeneration
- said AMD is dry AMD, which may be at an early, intermediate, or advanced stage (the latter otherwise known as geographic atrophy, GA).
- the anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies) of the present disclosure can be a monoclonal antibody, a human antibody, a humanized antibody, and/or a chimeric antibody.
- the antibody is a fragment selected from the group consisting of Fab, Fab-C, Fab'-SH, Fv, scFv, and (Fab')2 fragments.
- the anti-C3 antibody is a monospecific antibody.
- the anti-C3 antibody is a bispecific antibody.
- the anti-C3 antibody comprises two or more singledomain antibodies that form a bivalent antibody, a trivalent antibody, or a tetravalent antibody that recognizes different epitopes on the same or different antigens.
- an anti-C3 antibody provided herein is a chimeric antibody.
- Certain chimeric antibodies are described, for example, in U.S. Pat. No. 4,816,567; and Morrison et al., Proc. Natl. Acad. Sci. USA. 81:6851-6855 (1984).
- a chimeric antibody comprises a non-human variable region (e.g., a variable domain derived from a mouse, rat, hamster, rabbit, or non-human primate, such as a monkey) and a human constant domain.
- a chimeric antibody is a “class switched” antibody in which the class or subclass has been changed from that of the parent antibody.
- Chimeric antibodies include antigen-binding fragments thereof.
- a chimeric antibody is a humanized antibody.
- a non-human antibody is humanized to reduce immunogenicity to humans, while retaining the specificity and affinity of the parental non-human antibody.
- a humanized antibody comprises one or more variable domains in which HVRs, for example, CDRs, (or portions thereof) are derived from a non-human antibody, and FRs (or portions thereof) are derived from human antibody sequences.
- a humanized antibody optionally will also comprise at least a portion of a human constant region.
- some FR residues in a humanized antibody are substituted with corresponding residues from a non-human antibody (e.g., the antibody from which the HVR residues are derived), e.g., to restore or improve antibody specificity or affinity.
- a non-human antibody e.g., the antibody from which the HVR residues are derived
- the anti-C3 antibodies e.g., C3, C3a, and C3b antibodies
- the anti-C3 antibodies can be made into bivalent, trivalent, or tetravalent formats.
- an anti-C3 antibody of the present disclosure can be a bivalent, bispecific antibody with heteromeric heavy chains (e.g., Triomab, knobs-into-holes (KIH), Duobody, etc).
- An anti-C3 antibody of the present disclosure can be a tetravalent multispecific antibody comprised of IgGs with other binding domains fused to either the N- or C-termini of either the heavy or light chains (e.g., dual variable domain [DVD], IgG-scFv fusion, Mabtyrin (IgG with non-antibody binding scaffold “centyrin” fused to C-terminal end of heavy chains).
- An anti-C3 antibody of the present disclosure can be comprised of IgGs to which additional antigen combining sites have been added within the structure (e.g., two-in-one antibodies, MAT “Modular Antibody Technology” platform from F-Star).
- An anti-C3 antibody of the present disclosure can be an engineered antibody fragment linked by short peptide linkers which can be made into bivalent, trivalent, or tetravalent formats addressing two to three targets (e.g., bispecific T-cell engager (BiTE), Nanobody platform, dual- affinity re -targeting (DART) antibodies, “tandem antibody” structures (TandAbs)).
- targets e.g., bispecific T-cell engager (BiTE), Nanobody platform, dual- affinity re -targeting (DART) antibodies, “tandem antibody” structures (TandAbs)
- an anti-C3 antibody of the present disclosure can be comprised of chemically coupled IgGs.
- an anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure is a multispecific antibody, such as a bispecific antibody, which have binding specificities for at least two different antigens.
- the anti-C3 antibodies of the present disclosure, or antigen-binding fragments thereof can be used to form one arm (e.g., antigen-binding portion) of a bispecific antibody, whereas the other arm of the bispecific antibody can be specific for a different antigen.
- the other antigen includes, but is not limited to, interleukin- 1 beta (IL-1J3), interleukin-6 (IL-6); interleukin-6 receptor (IL-6R); interleukin- 13 (IL-13); IL-13 receptor (IL-13R); PDGF (e.g., PDGF-BB); angiopoietin; angiopoietin 2 (Ang2); Tie2; SIP; integrins av03, av05, and a501; betacellulin; apelin/APJ; erythropoietin; complement factor D; TNFa; HtrAl; a VEGF receptor (e.g., VEGFR1, VEGFR2, VEGFR3, membrane-bound VEGF -receptor (mbVEGFR), or soluble VEGF receptor (sVEGFR)); ST-2 receptor; and proteins genetically linked to age-related macular degeneration (AMD) risk, such as complement pathway components C2, factor B
- AMD
- the bispecific antibody may have binding specificity for C3/C3a/C3b and IL-1
- a bispecific antibody of the present disclosure includes an anti- C3 antibody (e.g., C3, C3a, and C3b antibody), or an antigen-binding fragment thereof, and an anti-VEGF antibody, or an antigen-binding fragment thereof.
- an anti- C3 antibody e.g., C3, C3a, and C3b antibody
- an anti-VEGF antibody or an antigen-binding fragment thereof.
- Such bispecific antibodies can be used to target the complement activation pathway from two different mechanisms, and thus provide additional therapeutic benefits.
- the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure
- the anti-VEGF arm can be any VEGF antagonist, including but not limited to, anti-VEGF antibodies (e.g., bevacizumab, sevacizumab, and ranibizumab), anti-VEGFR2 antibodies and related molecules (e.g., ramucirumab, tanibirumab, aflibercept), anti-VEGFRl antibodies and related molecules (e.g., icrucumab, aflibercept (VEGF Trap-Eye; EYLEA®), and ziv-aflibercept (VEGF Trap; ZALTRAP®)), anti-VEGF arms of VEGF bispecific antibodies (e.g., MP-0250, vanucizumab (VEGF-ANG2)), including anti-VEGF, anti- VEGFRl, and anti-VEGFR2 arms.
- VEGF antibodies e.g., bevacizumab, sevac
- a bispecific antibody of the present disclosure includes an anti- C3 antibody (e.g., C3, C3a, and C3b antibody), or an antigen-binding fragment thereof, and an anti-CFHR4 antibody, or an antigen-binding fragment thereof.
- an anti-C3 antibody e.g., C3, C3a, and C3b antibody
- an anti-CFHR4 antibody or an antigen-binding fragment thereof.
- Such bispecific antibodies can be used to target the complement activation pathway from two different mechanisms, and thus provide additional therapeutic benefits.
- the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure (e.g., C3, C3a, and C3b antibodies), and the anti-CFHR4 arm can be any CFHR4 antagonist.
- a bispecific antibody of the present disclosure includes an anti- C3 antibody (e.g., C3, C3a, and C3b antibody), or an antigen-binding fragment thereof, and an anti-C5 antibody, or an antigen-binding fragment thereof.
- the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure
- the anti-C5 arm can be any C5 antagonist, including but not limited to, eculizumab and tesidolumab (LFG316).
- a bispecific antibody of the present disclosure includes an anti-C3 antibody, or an antigen-binding fragment thereof, and an anti-FD antibody, or an antigen-binding fragment thereof.
- the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure
- the anti-C5 arm can be any C5 antagonist, including but not limited to, lampalizumab.
- the present disclosure provides anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies) comprising various functional characteristics.
- the anti-C3 antibodies described herein bind an antigen on C3 (SEQ ID NO: 1064), or a variant or isoform thereof, via interaction with its antigenic determinants (epitopes).
- binding of an anti-C3 antibody to C3, C3a, and/or C3b reduces complement activation.
- the anti-C3 antibody binds human C3, C3a, and/or C3b with a KD of about 100 nM or lower (FIG. 3).
- FIG. 4 includes representative data of C3 antibody cross-reactivity with human C3b protein, Cynomolgus monkey C3 protein, and human C3a protein.
- FIGS. 5A-5B include representative data of binding kinetics of C3 antibodies with respect to human C3 protein (FIG. 5A) and human C3b protein (FIG. 5B).
- FIG. 8 includes representative results of C3 antibody polyreactivity with baculovirus particles (BVPs);
- FIG. 9 includes representative results of C3 antibody polyreactivity with the various proteins indicated; and
- FIGS. 10A-10B include representative results of C3 antibody polyreactivity with CFHR4 (FIG. 10A) and CFH (FIG. 10B).
- Embodiments of the present disclosure also include a polynucleotide encoding any of the anti-C3 antibodies of the present disclosure.
- the polynucleotide comprises a sequence that is at least 70% identical (e.g., at least 70%, 75%, 80%, 85%, 90%, 95% or 100%) to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 70% identical (e.g., at least 70%, 75%, 80%, 85%, 90%, 95% or 100%) to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
- Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
- the present disclosure includes an expression vector comprising any of the polynucleotides encoding an anti-C3 antibody of the present disclosure.
- the expression vector is suitable for manufacturing an anti-C3 antibody of the present disclosure for delivery of the antibody to a subject.
- the nucleic acid sequence is in the form of a vector.
- the vector can be, for example, a plasmid, episome, cosmid, viral vector (e.g., retroviral or adenoviral), or phage.
- Suitable vectors and methods of vector preparation are well known in the art (see, e.g., Sambrook et al., Molecular Cloning, a Laboratory Manual, 4th edition, Cold Spring Harbor Press, Cold Spring Harbor, N.Y. (2012), and Ausubel et al., Current Protocols in Molecular Biology, Greene Publishing Associates and John Wiley & Sons, New York, N.Y. (1994)).
- the vector desirably comprises expression control sequences, such as promoters, enhancers, polyadenylation signals, transcription terminators, internal ribosome entry sites (IRES), and the like, that provide for the expression of the antibodyencoding nucleic sequence in a host cell.
- expression control sequences such as promoters, enhancers, polyadenylation signals, transcription terminators, internal ribosome entry sites (IRES), and the like, that provide for the expression of the antibodyencoding nucleic sequence in a host cell.
- Exemplary expression control sequences are known in the art and described in, for example, Goeddel, Gene Expression Technology: Methods in Enzymology, Vol. 185, Academic Press, San Diego, Calif. (1990).
- a vector comprising a nucleic acid sequence encoding an anti-C3 antibody (e.g., C3, C3a, and C3b antibody), or antigen-binding fragment thereof, may be introduced into a host cell that is capable of expressing the polypeptides encoded thereby, including any suitable prokaryotic or eukaryotic cell.
- suitable prokaryotic cells include, but are not limited to, cells from the genera Bacillus (such as Bacillus subtilis and Bacillus brevis), Escherichia (such as E. coll). Pseudomonas, Streptomyces, Salmonella, and Erwinia.
- Particularly useful prokaryotic cells include the various strains of Escherichia coli (e.g., K12, HB101 (ATCC No. 33694), DH5a, DH10, MC1061 (ATCC No. 53338), and CC102).
- Suitable eukaryotic cells are known in the art and include, for example, yeast cells, insect cells, and mammalian cells. Examples of suitable yeast cells include those from the genera Hansenula, Kluyveromyces, Pichia, Rhinosporidium, Saccharomyces, and Schizosaccharomyces .
- Suitable insect cells include Sf-9 and HIS cells (Invitrogen, Carlsbad, Calif.) and are described in, for example, Kitts et al., Biotechniques, 14: 810-817 (1993); Lucklow, Curr. Opin. Biotechnol., 4: 564-572 (1993); and Lucklow et al., J. Virol., 67'. 4566-4579 (1993).
- suitable mammalian cells include, but are not limited to, Chinese hamster ovary cells (CHO) (ATCC No. CCL61), CHO DHFR-cells (Urlaub et al., Proc. Natl. Acad. Sci.
- HEK human embryonic kidney
- HEK human embryonic kidney
- CRL1573 human embryonic kidney
- 3T3 cells ATCC No. CCL92
- Other suitable mammalian cell lines are the monkey COS-1 (ATCC No. CRL1650) and COS-7 cell lines (ATCC No. CRL1651), as well as the CV-1 cell line (ATCC No. CCL70).
- Further exemplary mammalian host cells include primate cell lines and rodent cell lines, including transformed cell lines. Normal diploid cells, cell strains derived from in vitro culture of primary tissue, as well as primary explants also are suitable.
- suitable mammalian cell lines include, but are not limited to, mouse neuroblastoma N2A cells, HeLa, mouse L-929 cells, and BHK or HaK hamster cell lines, all of which are available from the ATCC. Methods for selecting suitable mammalian host cells and methods for transformation, culture, amplification, screening, and purification of such cells are well known in the art (see, e.g., Ausubel et al., eds., Short Protocols in Molecular Biology, 5th ed., John Wiley & Sons, Inc., Hoboken, N.J. (2002)). Preferably, the mammalian cell is a human cell.
- the vector can include means for attaching a detection moiety to an anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure.
- the vector can include means for attaching a purification moiety to an anti-C3 antibody of the present disclosure.
- Exemplary detection and/or purification moieties/tags that can be coupled to an anti-C3 antibody of the present disclosure includes, but is not limited to, hemagglutinin (HA), c-Myc, V5, DYKDDDDK, His tag (e.g., 6x-HIS), Glutathione S-Transferase (GST), Maltose Binding Protein (MBP), a fluorophore (e.g., Green Fluorescent Protein (GFP), Red Fluorescent Protein (RFP), mCherry, a chromophore, and/or a luminescent peptide (e.g., luciferase).
- HA hemagglutinin
- c-Myc V5
- His tag e.g., 6x-HIS
- GST Glutathione S-Transferase
- MBP Maltose Binding Protein
- a fluorophore e.g., Green Fluorescent Protein
- the expression vector is suitable for use in gene therapy (e.g., an expression vector for delivering a polynucleotide encoding an anti-C3 antibody of the present disclosure to a subject).
- the expression vector is a herpes simplex virus (HSV) vector, or a retrovirus vector.
- the expression vector is an adeno- associated virus (AAV) vector, or comprises an AAV backbone.
- AAV vectors have been designed, produced and used to mediate gene delivery in human subjects, including for therapeutic purposes.
- AAV vectors for use in gene transfer comprise a replication defective AAV genome lacking functional Rep and Cap coding viral sequences.
- such replication defective AAV vectors lack most or all of the Rep and Cap coding sequences, and essentially retain one or two AAV ITR sequences and a packaging sequence.
- the defective genome is packaged in a viral particle, to form a defective, recombined AAV virus, also termed “AAV vector”.
- Methods of producing such AAV vectors have been disclosed in the literature, including using packaging cells, auxiliary viruses or plasmids, and/or baculovirus systems (Samulski et al., (1989) J. Virology 63, 3822; Xiao et al., (1998) J. Virology 72, 2224; Inoue et al., (1998) J. Virol.
- AAV vectors may be prepared or derived from various serotypes of AAVs, which may be even mixed together or with other types of viruses to produce chimeric (e.g., pseudotyped) AAV viruses.
- tAAVs are human AAV4 vectors, human AAV7 vectors, human AAV9 vectors, human AAV10 vectors, or bovine AAV vectors.
- the AAV vector may be derived from a single AAV serotype or comprise sequences or components originating from at least two distinct AAV serotypes (pseudotyped AAV vector), e.g., an AAV vector comprising an AAV genome derived from one AAV serotype (for example AAV9), and a capsid derived at least in part from a distinct AAV serotype.
- An AAV vector as used herein, is a vector which comprises at least one component part derivable from an adeno-associated virus. Preferably, that component part is involved in the biological mechanisms by which the vector infects or transduces target cells and expresses an anti-C3 antibody of the present disclosure (e.g., ocular delivery/expression).
- the expression vector is a lentiviral vector (LV), or comprises an LV backbone.
- Lentiviruses are part of a larger group of retroviruses. A detailed list of lentiviruses may be found in Coffin et al (1997) “Retroviruses” Cold Spring Harbour Laboratory Press Eds: JM Coffin, SM Hughes, HE Varmus pp 758-763).
- lentiviruses can be divided into primate and non-primate groups. Examples of primate lentiviruses include but are not limited to: the human immunodeficiency virus (HIV), the causative agent of human auto immunodeficiency syndrome (AIDS), and the simian immunodeficiency virus (SIV).
- HIV human immunodeficiency virus
- AIDS causative agent of human auto immunodeficiency syndrome
- SIV simian immunodeficiency virus
- the non- primate lentiviral group includes the prototype “slow virus” visna/maedi virus (VMV), as well as the related caprine arthritis-encephalitis virus (CAEV), equine infectious anaemia virus (EIAV), feline immunodeficiency virus (FIV), Maedi visna virus (MW) and bovine immunodeficiency virus (BIV).
- VMV low virus
- CAEV caprine arthritis-encephalitis virus
- EIAV equine infectious anaemia virus
- FIV feline immunodeficiency virus
- MW Maedi visna virus
- bovine immunodeficiency virus BIV
- the lentiviral vector is derived from HIV- 1, HIV-2, SIV, FIV, BIV, EIAV, CAEV or Visna lentivirus.
- the lentivirus family differs from retroviruses in that lentiviruses have the capability to infect both dividing and non-dividing cells (Lewis et al (1992) EM BO J 11 (8): 3053-3058 and Lewis and Emerman (1994) J Virol 68 (1):510-516).
- retroviruses such as MLV
- a lentiviral vector is a vector which comprises at least one component part derivable from a lentivirus.
- that component part is involved in the biological mechanisms by which the vector infects or transduces target cells and expresses an anti-C3 antibody of the present disclosure (e.g., ocular delivery/expression).
- compositions and method for ocular gene therapy can be found in, e.g., Bordet, T., and Behar-Cohen, F., “Ocular gene therapies in clinical practice: viral vectors and nonviral alternatives,” Drug Discovery Today, Volume 24, Issue 8, August 2019, Pages 1685- 1693).
- gene therapy platforms, methods, and compositions that can be used to deliver an anti-C3 antibody of the present disclosure to a subject includes the platforms, methods, and compositions disclosed in US20220025396, US20220011308, US20210371877, US20210363192, US20190078099, US20190038724, and US10494646B2, which are incorporated herein by reference.
- gene therapy platforms, methods, and compositions that can be used to deliver an anti-C3 antibody of the present disclosure to a subject includes the platforms, methods, and compositions based on HMR59 (Hemera Biosciences), which through its protein product soluble CD59, blocks the membrane attack complex that forms during the terminal step in the complement cascade.
- HMR59 is designed to be administered as a single intraocular injection.
- the present disclosure also provides a method of administering ocular gene therapy to a subject in need thereof comprising injecting a pharmaceutical composition comprising an effective amount of an expression vector described herein (e.g., an expression vector comprising a polynucleotides encoding an anti-C3 antibody of the present disclosure).
- a pharmaceutical composition comprising an effective amount of an expression vector described herein (e.g., an expression vector comprising a polynucleotides encoding an anti-C3 antibody of the present disclosure).
- administering the pharmaceutical composition treats at least one AMD symptom and/or GA.
- the anti-C3 antibodies can be administered as part of a pharmaceutical composition in a therapeutically effective amount to treat an eye disease (e.g., AMD or GA).
- the composition is suitable for ocular administration.
- ocular administration comprises injection into vitreous fluid.
- ocular administration comprises delivering the antibody using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant (e.g., SusvimoTM).
- Recent methods and formulations for ocular administration can be found in, e.g., Souto, E.B., et al. “Advanced Formulation Approaches for Ocular Drug Delivery: State-Of-The-Art and Recent Patents,” Pharmaceutics, 2019 Sep; 11(9): 460).
- the methods include administering a pharmaceutical composition comprising a therapeutically effective amount of an anti-C3 antibody of the present disclosure.
- the pharmaceutical composition is administered ocularly and treats at least one AMD symptom.
- AMD comprises wet AMD.
- AMD comprises dry AMD (e.g., GA).
- the at least one AMD symptom comprises visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light.
- administering the pharmaceutical composition reduces complement activation in the subject’s eye.
- the pharmaceutical composition comprising a therapeutically effective amount of an anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure is administered at a dose ranging from about 0.0001 mg/dose to about 100 mg/dose.
- the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/dose to about 100 mg/dose.
- the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/dose to about 100 mg/dose.
- the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/dose to about 100 mg/dose.
- the anti-C3 antibody is administered at a dose ranging from about 1.0 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 10 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 10 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 1.0 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 0.1 mg/dose.
- the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 0.001 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/dose to about 10 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/dose to about 1.0 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/dose to about 10 mg/dose.
- the pharmaceutical composition comprising a therapeutically effective amount of an anti-C3 antibody of the present disclosure is administered at a dose ranging from about 0.0001 mg/ml to about 100 mg/ml.
- the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/ml to about 100 mg/ml.
- the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/ml to about 100 mg/ml.
- the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/ml to about 100 mg/ml.
- the anti-C3 antibody is administered at a dose ranging from about 1.0 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 10 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 10 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 1.0 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 0.1 mg/ml.
- the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 0.01 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 0.001 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/ml to about 10 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/ml to about 1.0 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/ml to about 10 mg/ml.
- the terms “treatment,” “treating,” and the like refer to obtaining a desired pharmacologic and/or physiologic effect.
- the effect is therapeutic (e.g., the effect partially or completely cures a disease and/or adverse symptom attributable to the disease).
- the methods of the present disclosure comprise administering a “therapeutically effective amount” of an anti-C3 antibody (e.g., C3, C3a, and C3b antibody), or composition comprising an anti-C3 antibody.
- a “therapeutically effective amount” refers to an amount effective, at dosages and for periods of time necessary, to achieve a desired therapeutic result.
- the therapeutically effective amount may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the monoclonal antibody to elicit a desired response in the individual.
- a therapeutically effective amount of an anti-C3 antibody of the present disclosure is an amount which treats at least one AMD and/or GA symptom in a subject.
- the pharmacologic and/or physiologic effect may be prophylactic (e.g., the effect completely or partially prevents a disease or symptom thereof).
- the methods of the present disclosure comprise administering a “prophylactically effective amount” of an anti-C3 antibody or composition comprising an anti-C3 antibody.
- a “prophylactically effective amount” refers to an amount effective, at dosages and for periods of time necessary, to achieve a desired prophylactic result (e.g., prevention of AMD and/or GA onset).
- a typical dose of a therapeutically effective amount of an anti-C3 antibody of the present disclosure can range from, for example, about 0.0001 mg/dose to about 100 mg/dose for each eye to be treated.
- a therapeutically effective amount of an anti-C3 antibody of the present disclosure can range from about 0.001 mg/dose to about 100 mg/dose, from about 0.01 mg/dose to about 100 mg/dose, from about 0.05 mg/dose to about 50 mg/dose, from about 0.1 mg/dose to about 10 mg/dose, from about 0.5 mg/dose to about 5 mg/dose, and from about 1 mg/dose to about 10 mg/dose.
- a therapeutically effective concentration of an anti-C3 antibody of the present disclosure can range from, for example, about 0.0001 mg to about 100 mg of the antibody per milliliter of solution. In some embodiments, a therapeutically effective concentration of an anti-C3 antibody of the present disclosure can range from about 0.001 mg/ml to about 100 mg/ml, from about 0.01 mg/ml to about 100 mg/ml, from about 0.1 mg/ml to about 100 mg/ml, from about 1.0 mg/ml and about 100 mg/ml, from about 0.001 mg/ml and about 50 mg/ml, from about 0.01 mg/ml and about 50 mg/ml, from about 0.1 mg/ml and about 50 mg/ml, from about 0.1 mg/ml and about 25 mg/ml, from about 0.1 mg/ml and about 10 mg/ml, and from about 1.0 mg/ml and about 10 mg/ml.
- a therapeutically effective dose of an anti-C3 antibody of the present disclosure can be, exactly or approximately, 0.1 mg, 0.2 mg, 0.25 mg, 0.3 mg, 0.35 mg, 0.4 mg, 0.45 mg, 0.5 mg, 0.55 mg, 0.6 mg, 0.65 mg, 0.7 mg, 0.75 mg, 0.8 mg, 0.85 mg, 0.9 mg, 0.95 mg, 1.0 mg, 2.0 mg, 3.0 mg, 4.0 mg, 5.0 mg, 10.0 mg, 15.0 mg, 20.0 mg, or 25.0 mg, or can fall within a range delimited by any two of the foregoing values.
- a sustained release formulation (e.g., an ocular implant) can be, exactly or approximately, 0.1 mg, 0.2 mg, 0.25 mg, 0.3 mg, 0.35 mg, 0.4 mg, 0.45 mg, 0.5 mg, 0.55 mg, 0.6 mg, 0.65 mg, 0.7 mg, 0.75 mg, 0.8 mg, 0.85 mg, 0.9 mg, 0.95 mg, 1.0 mg, 2.0 mg, 3.0 mg, 4.0 mg, 5.0 mg, 10.0 mg, 15.0 mg, 20.0 mg, or 25.0 mg of an anti-C3 antibody, or an amount that falls within a range delimited by any two of the foregoing values.
- Therapeutic or prophylactic efficacy can be monitored by periodic assessment of treated patients. For repeated administrations over several days or longer, depending on the condition, the treatment is repeated until a desired suppression of disease symptoms occurs.
- the desired dosage can be delivered by a single bolus administration of the composition, by multiple bolus administrations of the composition, or by continuous infusion administration of the composition.
- the composition comprising an anti-C3 antibody, or antigen-binding fragment thereof can be administered to a mammal using standard administration techniques, including ocular, oral, intravenous, intraperitoneal, subcutaneous, pulmonary, transdermal, intramuscular, intranasal, buccal, sublingual, or suppository administration.
- the composition preferably is suitable for ocular administration.
- embodiments of the present disclosure include anti-C3 antibodies that have enhanced half-life (e.g., after ocular administration), such that the anti-C3 antibody can be administered less often to a subject.
- the antibody comprises a half-life extension moiety.
- the half-life extension moiety comprises a polypeptide that can be coupled to an anti-C3 antibody of the present disclosure by any means known in the art (e.g., generation of a fusion protein).
- the polypeptide that can be coupled to an anti-C3 antibody of the present disclosure is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residues.
- the half-life extension polypeptide is attached to the C-terminus or N-terminus of the antibody. In some embodiments, this is referred to as “XTENylation,” as described further in US8933197, US7846445, US7855279, US8492530, US9938331, US8673860, US9371369, US9926351, US10961287, US10172953, and US10953073.
- the present disclosure also provides a composition comprising any of the anti-C3 antibodies or antigen-binding fragments thereof described herein.
- the composition desirably is a pharmaceutically acceptable (e.g., physiologically acceptable) composition, which comprises a carrier, preferably a pharmaceutically acceptable (e.g., physiologically acceptable) carrier, and the anti-C3 antibody or antigen-binding fragment thereof.
- a pharmaceutically acceptable composition which comprises a carrier, preferably a pharmaceutically acceptable (e.g., physiologically acceptable) carrier, and the anti-C3 antibody or antigen-binding fragment thereof.
- a carrier preferably a pharmaceutically acceptable (e.g., physiologically acceptable) carrier
- the composition may contain preservatives, such as, for example, methylparaben, propylparaben, sodium benzoate, and benzalkonium chloride. A mixture of two or more preservatives optionally may be used.
- buffering agents may be included in the composition. Suitable buffering agents include, for example, citric acid, sodium citrate, phosphoric acid, potassium phosphate, and various other acids and salts. A mixture of two or more buffering agents optionally may be used. Methods for preparing compositions for pharmaceutical use are known to those skilled in the art and are described in, for example, Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins; 21st ed. (May 1, 2005). [00118] Once administered to a mammal (e.g., a human), the biological activity of the anti-C3 antibody, or antigen-binding fragment thereof, can be measured by any suitable method known in the art.
- a mammal e.g., a human
- the biological activity can be assessed by determining the stability of the anti- 03 antibody.
- the biological activity of the anti-C3 antibody also can be assessed by determining its binding affinity to C3, C3a, and/or C3b peptides and/or by assessing its binding affinity to peptides with which it may cross-react.
- affinity refers to the equilibrium constant for the reversible binding of two agents and is expressed as the dissociation constant (KD).
- Affinity of a binding agent to a ligand can be, for example, from about 1 femtomolar (fM) to about 1 millimolar (mM) (e.g., from about 1 picomolar (pM) to about 1 nanomolar (nM), or from about 1 nM to about 1 micromolar (pM)).
- the affinity of an anti-C3 antibody may be from about 1 nm to about 20 nm, and desirably from about 5 nm to about 10 nm.
- Antibody affinity for an antigen or epitope of interest can be measured using any art-recognized assay.
- Such methods include, for example, fluorescence activated cell sorting (FACS), separable beads (e.g., magnetic beads), antigen panning, and/or ELISA (see, e.g., Janeway et al. (eds.), Immunobiology, 5th ed., Garland Publishing, New York, N.Y., 2001).
- FACS fluorescence activated cell sorting
- separable beads e.g., magnetic beads
- antigen panning e.g., antigen panning
- ELISA see, e.g., Janeway et al. (eds.), Immunobiology, 5th ed., Garland Publishing, New York, N.Y., 2001).
- an anti-C3 antibody, or composition comprising an anti-C3 antibody may be administered alone or in combination with other drugs.
- the anti- C3 antibody can be administered in combination with other agents for the treatment or prevention of AMD and/or GA, as disclosed herein.
- anti-C3 antibodies of the present disclosure, or antibody conjugates, fusion proteins, or polymeric formulations thereof can be used either alone or in combination with other agents in a therapy.
- an anti-C3 antibody may be coadministered with at least one additional therapeutic agent.
- an additional therapeutic agent is another antibody, a chemotherapeutic agent, a cytotoxic agent, an anti- angiogenic agent, an immunosuppressive agent, a prodrug, a cytokine, a cytokine antagonist, cytotoxic radiotherapy, a corticosteroid, an anti-emetic, a cancer vaccine, an analgesic, a growth- inhibitory agent, or combinations thereof.
- any of the preceding methods further comprises administering one or more additional compounds.
- the anti-C3 antibody, antibody conjugate, fusion protein, or polymeric formulation is administered simultaneously with the additional compound(s).
- the anti-C3 antibody, antibody conjugate, fusion protein, or polymeric formulation is administered before or after the additional compound(s).
- the additional compound binds to a second biological molecule selected from the group consisting of VEGF, IL-10; IL-6; IL-6R; IL-13; IL-13R; PDGF; angiopoietin; Ang2; Tie2; SIP; integrins av03, av05, and a501; betacellulin; apelin/APJ; erythropoietin; complement factor D; TNFa; HtrAl; a VEGF receptor; ST-2 receptor; and proteins genetically linked to AMD risk, such as complement pathway components C2, factor B, factor H, CFHR3, CFHR4, C5, C5a, HtrAl, ARMS2, TIMP3, HLA, interleukin-8 (IL-8), CX3CR1, TLR3, TLR4, CETP, LIPC, COL10A1, and TNFRSF10A.
- a second biological molecule selected from the group consisting of VEGF, IL-10; IL-6;
- the additional compound is an antibody or antigen-binding fragment thereof.
- the ocular disorder is an intraocular neovascular disease selected from the group consisting of proliferative retinopathies, choroidal neovascularization (CNV), age-related macular degeneration (AMD), geographic atrophy (GA), diabetic and other ischemia-related retinopathies, diabetic macular edema, pathological myopia, von Hippel-Lindau disease, histoplasmosis of the eye, retinal vein occlusion (RVO), including CRVO and BRVO, corneal neovascularization, retinal neovascularization, and retinopathy of prematurity (ROP).
- CNV proliferative retinopathies
- AMD age-related macular degeneration
- GA geographic atrophy
- diabetic and other ischemia-related retinopathies diabetic macular edema
- pathological myopia von Hippel-Lindau disease
- an anti-C3 antibody of the present disclosure may be administered in combination with at least one additional therapeutic agent for treatment of an ocular disorder, for example, an ocular disorder described herein (e.g., AMD (e.g., wet AMD or dry AMD), GA, DME, DR, or RVO).
- AMD e.g., wet AMD or dry AMD
- GA e.g., GA, DME, DR, or RVO
- VEGF antagonists include, without limitation, anti-angiogenic agents, such as VEGF antagonists, including, for example, anti-VEGF antibodies (e.g., the anti-VEGF Fab LUCENTIS® (ranibizumab)), soluble receptor fusion proteins (e.g., the recombinant soluble receptor fusion protein EYLEA® (aflibercept, also known as VEGF Trap Eye; Regeneron/ Aventis)), aptamers (e.g., the anti-VEGF pegylated aptamer MACUGEN® (pegaptanib sodium; NeXstar Pharmaceuticals/OSI Pharmaceuticals)), and VEGFR tyrosine kinase inhibitors (e.g., 4-(4-bromo- 2-fluoroanilino)-6-methoxy-7-(l -methyip iperidin-4-ylmethoxy)quinazoline (ZD6474), 4-(4-(4-bromo- 2-fluor
- additional therapeutic agents that can be used in combination with an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO), include, but are not limited to, VISUDYNE® (verteporfin; a light-activated drug that is typically used in conjunction with photodynamic therapy with a non-thermal laser), PKC412, Endovion (NS 3728; NeuroSearch A/S), neurotrophic factors (e.g., glial derived neurotrophic factor (GDNF) and ciliary neurotrophic factor (CNTF)), diltiazem, dorzolamide, PHOTOTROP®, 9-cis-retinal, eye medication (e.g., phospholine iodide, echothiophate, or carbonic anhydrase inhibitors), veovastat (AE-941; AEterna Laboratories,
- An anti-C3 antibody of the present disclosure may be administered in combination with a therapy or surgical procedure for treatment of an ocular disorder (e.g., AMD, GA DME, DR, or RVO), including, for example, laser photocoagulation (e.g., panretinal photocoagulation (PRP)), drusen lasering, macular hole surgery, macular translocation surgery, implantable miniature telescopes, PHI-motion angiography (also known as micro-laser therapy and feeder vessel treatment), proton beam therapy, microstimulation therapy, retinal detachment and vitreous surgery, scleral buckle, submacular surgery, transpup illary thermotherapy, photosystem I therapy, use of RNA interference (RNAi), extracorporeal rheopheresis (also known as membrane differential filtration and rheotherapy), microchip implantation, stem cell therapy, gene replacement therapy, ribozyme gene therapy (including gene therapy
- RNAi RNA interference
- extracorporeal rheopheresis
- an anti-C3 antibody of the present disclosure can be administered in combination with an anti-angiogenic agent for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO).
- an anti-angiogenic agent for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO).
- Any suitable anti-angiogenic agent can be used in combination with an antibody of the present disclosure, including, but not limited to, those listed by Carmeliet et al. Nature 407:249-257, 2000.
- the anti-angiogenic agent is a VEGF antagonist, including, but not limited to, an anti-VEGF antibody (e.g., the anti-VEGF Fab LUCENTIS® (ranibizumab), RTH-258 (formerly ESBA-1008, an anti-VEGF single-chain antibody fragment; Novartis), or a bispecific anti-VEGF antibody (e.g., an anti-VEGF/anti- angiopoietin 2 bispecific antibody such as RG-7716; Roche)), a soluble recombinant receptor fusion protein (e.g., EYLEA® (aflibercept)), a VEGF variant, a soluble VEGFR fragment, an aptamer capable of blocking VEGF (e.g., pegaptanib) or VEGFR, a neutralizing anti-VEGFR antibody, a small molecule inhibitor of VEGFR tyrosine kinases, an anti-VEGF DARPin® (e.g., ab
- Suitable anti-angiogenic agents that may be administered in combination with an antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO) include corticosteroids, angiostatic steroids, anecortave acetate, angiostatin, endostatin, tyrosine kinase inhibitors, matrix metalloproteinase (MMP) inhibitors, insulin-like growth factor-binding protein 3 (IGFBP3), stromal derived factor (SDF-1) antagonists (e.g., anti-SDF-1 antibodies), pigment epithelium-derived factor (PEDF), gamma-secretase, Delta-like ligand 4, integrin antagonists, hypoxia-inducible factor (HIF)-la antagonists, protein kinase CK2 antagonists, agents that inhibit stem cell (e.g., endothelial progen), cortic
- an anti-C3 antibody of the present disclosure can be administered in combination with an agent that has activity against neovascularization for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO), such as an anti-inflammatory drug, a mammalian target of rapamycin (mTOR) inhibitor (e.g., rapamycin, AFINITOR® (everolimus), and TORISEL® (temsirolimus)), cyclosporine, a tumor necrosis factor (TNF) antagonist (e.g., an anti- TNFa antibody or antigen-binding fragment thereof (e.g., infliximab, adalimumab, certolizumab pegol, and golimumab) or a soluble receptor fusion protein (e.g., etanercept)), an anti-complement agent, a nonsteroidal rapamycin (mTOR) inhibitor (e.g., rapamycin, AFINITOR® (ever
- Any suitable AMD therapeutic agent can be administered as an additional therapeutic agent in combination with an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO), including, but not limited to, a VEGF antagonist, for example, an anti- VEGF antibody (e.g., LUCENTIS® (ranibizumab), RTH-258 (formerly ESBA-1008, an anti-VEGF single-chain antibody fragment; Novartis), or a bispecific anti- VEGF antibody (e.g., an anti-VEGF/anti-angiopoietin 2 bispecific antibody such as RG-7716; Roche)), a soluble VEGF receptor fusion protein (e.g., EYLEA® (aflibercept)), an anti- VEGF DARPin® (e.g., abicipar pegol; Molecular Partners AG/ Allergan),
- AMD therapeutic agents can be co-formulated.
- the anti-PDGFR antibody REGN2 176-3 can be co-formulated with aflibercept (EYLEA®).
- EYLEA® aflibercept
- such a coformulation can be administered in combination with an antibody of the present disclosure.
- the ocular disorder is AMD (e.g., wet AMD).
- an anti-C3 antibody or antigen-binding fragment, described herein can be used in diagnostic or research applications.
- Research applications include, for example, methods that utilize the anti-C3 antibody and a label to detect C3, C3a, and/or C3b in a sample, e.g., in a human body fluid or in a cell or tissue extract.
- the anti-C3 antibody or antigen-binding fragment thereof may be employed in any suitable assay for measuring C3, C3a, and/or C3b in a sample for diagnostic and/or research purposes.
- Such assays include, but are not limited to, sandwich immunoassays, enzyme immunoassays (EIA), enzyme-linked immunosorbent assays (ELISA), lateral flow assays, competitive inhibition immunoassays (e.g., forward and reverse), competitive binding assays, Forster resonance energy transfer (FRET), one- step antibody detection assays, single molecule detection assays, radioimmunoassays (RIA), and FACS.
- sandwich immunoassays enzyme immunoassays (EIA), enzyme-linked immunosorbent assays (ELISA), lateral flow assays, competitive inhibition immunoassays (e.g., forward and reverse), competitive binding assays, Forster resonance energy transfer (FRET), one- step antibody detection assays, single molecule detection assays, radioimmunoassays (RIA), and FACS.
- EIA enzyme immunoassays
- ELISA enzyme-linked immunosorbent assays
- lateral flow assays e
- the anti-C3 antibody or antigen-binding fragment thereof can be provided in a kit, e.g., a packaged combination of reagents in predetermined amounts with instructions for performing an assay using the antibody (e.g., an assay that detects C3, C3a, and/or C3b).
- a kit comprising the antibody or antigen-binding fragment described herein and instructions for use thereof.
- the instructions can be in paper form or computer-readable form, such as a disk, CD, DVD, etc.
- the kit can comprise a calibrator or control, and/or at least one container (e.g., tube, microtiter plates, or strips) for conducting an assay, and/or a buffer, such as an assay buffer or a wash buffer.
- the kit comprises all components, i.e., reagents, standards, buffers, diluents, etc., which are necessary to perform the assay.
- Other additives may be included in the kit, such as stabilizers, buffers (e.g., a blocking buffer or lysis buffer), and the like.
- the relative amounts of the various reagents can be varied to provide for concentrations in solution of the reagents which substantially optimize the sensitivity of the assay.
- the reagents may be provided as dry powders (typically lyophilized), including excipients which on dissolution will provide a reagent solution having the appropriate concentration.
- C3 antibody sequences were recovered from immunized mice followed by phage display with either fixed or diverse light chains. Selected leads were produced as full aglyco IgGls and extensively characterized.
- C3 Recombinant Proteins were procured from Complement Technologies (Tyler, TX); the proteins were purified directly from the relevant species (human or cynomolgus) sera. Recombinant proteins were qualified with analytic SEC and binding studies to established antibodies.
- Sample bleeds were conducted at the fourth week and tested for antigen positive titer and cross reactive titer (human for cynomolgus cohort or cynomolgus for human cohort).
- ELISA plates were coated with 1 ug/ml of relevant recombinant protein overnight. Coated plates were incubated with sera across a descending 5 x 10-fold range starting at 1:100 and terminating at 1 : 10E7 dilutions.
- Antibodies bound to the coated antigen were detected with anti-mouse IgG HRP secondary antibody and quantified with the aid of one step TMB solution and OD determination at 450 nM using a microplate reader. Note, the positive control antibody used for the ELISA experiment is not cross reactive to the cyno C3 protein (FIGS. 1A-1D).
- RNA was extracted from said tissues.
- Two sequence recovery strategies were used in this study, a fixed light strategy and a diverse light chain strategy.
- variable heavy chains were amplified from the RNA prepared from the spleen tissues via reverse transcription.
- viable gene specific primers and PCR were used to create DNA fragments which could be cloned into a phage display vector designed to express Fabs on phage g3p protein.
- the diverse light chain strategy the same approach was used to amplify the natural diversity of the light chain from the spleen RNA and clone into the Fab vector.
- the relevant part of the Fab vector was fixed to two specific sequences with constructs synthesized commercially that match the LC of ATX-P-569 and ATX-P-592.
- Purified phages expressing Fabs were selected for antigen binding as described earlier in a subsequent 2nd round. Phage from the second round output were diluted and infected in TGI cells. Polyclonal pools of phage output from both rounds were tested by ELISA to confirm enrichment and examine cross reactivity (FIG. 2).
- Antibody Production Unique variable heavy and light chain pairs from the phage display campaigns were cloned into vectors designed to express full length antibodies as algyco IgGls in HEK293 cells under the control of a CMV promoter. Antibody expression vectors were complexed with polyethylenimine and transfected into HEK293 cultures. After 5 days of shaking at 37 C in 293 cell culture media, antibodies were captured on agarose-based protein A resin. After several stringent washes, antibodies were eluted in citrate solution, pH 3, neutralized with Hepes, pH 9, and buffer exchanged into PBS.
- the kinetic data was referenced with the interstitial reference spots and double-referenced to a buffer cycle, and then fit globally to a 1 : 1 binding model to determine their apparent association and dissociation kinetic rate constants (K a and KD values).
- K a and KD values apparent association and dissociation kinetic rate constants.
- Epitope Binning High-throughput epitope binning experiment was done on real-time label-free biosensors (Carterra LSA) to sort large panels of mAbs into bins based on their ability to block one another for binding to the antigen.
- antigen and antibody 2 analyte antibody
- H200M sensor chip covalently pre-loaded with antibody 1 (ligand antibody).
- An increase in response upon exposure to the analyte antibody indicates non-competition between the two antibodies, whereas a lack of change in the signal indicates competition.
- Antibodies having the same blocking profiles towards others in the test set are grouped into one bin.
- C3 Antibody Polyreactivity Experiments were conducted to determine the reactivity of the C3 antibodies of the present disclosure with various other proteins using ELISA and Carterra LSA platforms (FIGS. 8 and 9). The methods used were similar as reported by Hotzel et al. (2012). Briefly, baculovirus particles (BVP, Lake Pharma) was diluted 1:100 in 50 rnM sodium bicarbonate (pH 9.3). After overnight incubation of 50 pL of BVP on ELISA plates (3369; Corning) at 4 °C overnight, unbound BVPs were aspirated from the wells. All remaining steps were performed at RT.
- BVP baculovirus particles
- the plate was blocked with 100 pL of blocking buffer (PBS with 1% BSA) for 1 h before three washes with 100 pL of PBS.
- 50 pL of 16 nM testing antibodies was added to the wells and incubated for 1 h followed by washes with 100 pL of PBS.
- HRP-conjugated goat anti-human IgG antibody at 1:1000 Jackson ImmunoResearch
- 50 pL of TMB substrate (34021; Fisher Scientific) was added to each well and incubated for 10-15 min. The reactions were stopped by adding 50 pL of 2 M sulfuric acid to each well.
- the absorbance was read at 450 nm and BVP score determined by normalizing absorbance by control wells with no test antibody.
- lOOpl of the erythrocyte suspension with GVB+ + diluted with an equal volume of serum was diluted to 30%, 15%, 7.5%, and 3.75%.
- the freshly prepared mouse serum was diluted at 15%, 7.5%, 3.75%, 1.875%.
- SRBC sheep red blood cell
- FIG. 11 an assay was conducted to test whether the C3b antibodies of the present disclosure were capable of activating the alternative complement pathway by quantifying sheep red blood cell (SRBC) hemolysis.
- the assay was run twice in parallel, once with sensitized SRBC (FIG. 11) and once with non-sensitized SRBC.
- eight C3b antibodies were tested in parallel with compstatin, a cyclic peptide that binds to C3 and inhibits its cleavage.
- the compstatin assays were run with and without normal human serum (NHS). NHS is the source of complement components and in its absence there is no SRBC lysis.
- Table 6 Summary of SEQ ID NOs for individual exemplary antibodies of the present disclosure.
- Human Complement Component 3 which includes Complement Component 3a (C3a) and Complement Component 3b (C3b), was used to generate the anti-C3 antibodies of the present disclosure (see, e.g., UniProt Accession No.
- M0QYC8 ITHRIHWESASLLRSEETKENEGFTVTAEGKGQGTLSVVTMYHAKAKDQLT CNKFDLKVTIKPAPETGIPSPIFLSSVFLEKRPQDAKNTMILEICTRYRGDQDATMSILDIS MMTGFAPDTDDLKQLANGVDRYISKYELDKAFSDRNTLIIYLDKVSHSEDDCLAFKVH QYFNVELIQPGAVKVYAYYNL (SEQ ID NO: 1064).
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Immunology (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Animal Behavior & Ethology (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Ophthalmology & Optometry (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Peptides Or Proteins (AREA)
Abstract
The present disclosure relates to the treatment and/or prevention of age-related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA). In particular, the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3) proteins.
Description
COMPLEMENT COMPONENT 3 (C3)-SPECIFIC ANTIBODIES AND USES THEREOF
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to and the benefit of U.S. Provisional Patent Application No. 63/462,638 filed April 28, 2023, which is incorporated herein by reference in its entirety for all purposes.
SEQUENCE LISTING STATEMENT
[0002] The text of the computer readable sequence listing filed herewith, titled “BWING_41801_601_SequenceListing,” created on April 23, 2024, having a fde size of 1,199,632 bytes, is hereby incorporated by reference in its entirety
FIELD
[0003] Embodiments of the present disclosure relate to the treatment and/or prevention of age- related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA). In particular, the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3) proteins.
BACKGROUND
[0004] Age-Related Macular Degeneration (AMD) is the leading cause of severe vision loss in adults over age 50. The Centers for Disease Control and Prevention estimate that 1.8 million people have AMD and another 7.3 million are at substantial risk for vision loss from AMD. This eye disease occurs when there are changes to the macula, a small portion of the retina that is located on the inside back layer of the eye. AMD is a loss of central vision that can occur in two forms: “dry” (atrophic) and “wet” (exudative). Most people with macular degeneration have the dry form. Geographic atrophy (GA) is considered the late stage of the dry form of AMD, characterized by focal and progressive loss of photoreceptors. GA is less common than neovascular AMD, and it is responsible for 10-20% of cases of legal blindness in this condition, affecting more than 5 million people worldwide. Currently there is no approved or effective treatment to prevent either the onset
or progression of GA; however, in recent years, significant progress has been made in understanding the pathogenesis of GA, which has led to a number of new potential therapies.
[0005] Genetic and molecular studies have identified the complement system as an important factor in the etiology and progression of AMD. Many of the genetic risk variants cluster in genes of the alternative pathway of the complement system, and complement activation products are elevated in AMD patients. Nevertheless, attempts in treating AMD via complement regulators have not yet been successful, suggesting a level of complexity that could not be predicted only from a genetic point of view. Thus, there remains a need for new therapeutic approaches for the treatment of AMD and GA.
SUMMARY
[0006] Embodiments of the present disclosure include compositions and methods pertaining to an antibody, or antigen binding fragment thereof, which specifically binds Complement Component 3 (C3) polypeptides, optionally wherein said C3 is a Complement Component 3a (C3a) polypeptide or a Complement Component 3b (C3b) polypeptide, which is optionally a polypeptide comprising or consisting of the amino acid sequence of SEQ ID NO: 1064. In accordance with these embodiments, the present disclosure provides an antibody or a fragment thereof, which exhibits any one or more the following functional characteristics: (a) reduces complement activation and/or attenuates complement component 3 (C3) convertase activity, which is optionally measured in an ELISA; and/or (b) cross-reacts with at least one of human C3b protein, Cynomolgus monkey C3 protein, and/or human C3a protein; and/or (c) binds to human C3 with a KD-1 of about lOOnM or lower; and/or (d) binds to the same epitope on human C3 as an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) competes for binding to human C3 with an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6. In some embodiments, the antibody or fragment thereof, which is a monoclonal antibody. In some embodiments, the antibody or fragment thereof, which is a human, humanized or chimeric antibody. In further embodiments, the antibody or fragment thereof, is a whole antibody, a single chain variable fragment (scFv), a variable fragment (Fv), a fragment antigen-binding region (Fab), a Fab-C, a Fab’-SH, a (Fab’)2, a single-domain
antibody (sdAb), a nanobody, a diabody, a triabody, or an aptamer, optionally wherein the antibody is a whole antibody comprising an Fc region such as a human IgGl , IgG2, IgG3 or IgG4 Fc region. [0007] In some embodiments, the antibody or fragment thereof, is conjugated to at least one additional moiety, optionally selected from: (a) a therapeutic or cytotoxic moiety; (b) a detection moiety; (c) a purification moiety; (d) a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody. In accordance with these embodiments, the present disclosure provides an antibody or a fragment thereof, which is a polypeptide comprising: (a) one, two or all three HCDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also one, two or all three of the corresponding LCDRs of said exemplary antibody; and/or (b) a VH sequence having at least 90% identity to the VH sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also a VL sequence having at least 90% identity to the corresponding VL sequence of said exemplary antibody, preferably wherein variation is not permitted in the HCDRs or LCDRs; and/or (c) all six CDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (d) the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) the full length heavy chain (VH + constant) sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally the corresponding full length light chain (VL + constant) sequence of said exemplary antibody.
[0008] Embodiments of the present disclosure include compositions and methods pertaining to a polynucleotide encoding an antibody or fragment according to any one of the preceding claims, optionally wherein said polynucleotide comprises or consists of a nucleic acid sequence having at least 70, 80, 90 or 100% identity to a nucleic acid sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6. In some embodiments, an expression vector comprises a polynucleotide, which is optionally an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, or a retrovirus vector.
[0009] Embodiments of the present disclosure include compositions and methods pertaining to antibodies directed against Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, including antigen binding
fragments thereof. In accordance with these embodiments, the present disclosure provides a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3. In some embodiments, the HCDR1 comprises one of the following amino acid sequences: the HCDR1 comprises one of the following amino acid sequences: (a) X1YX2X3X4 (SEQ ID NO: 1), wherein Xi is G, S, N, D, T, or A; X2 is Y, A, G, W, or T; X3 is L, M, I, or V; and X4 is H, N, S, or G; (b) X1X2 X3MX4 (SEQ ID NO: 57), wherein Xi is D, N, S, or A; X2 is Y, or A; X3 is A, W, Y, S, C, D, or G; and X4 is H, S, or N; (c) X1X2X3WWX4 (SEQ ID NO: 95), wherein Xi is I, S, R, N, or K; X2 is N, D, S, or G; X3 is N, S, T, D, Y, or H; and X4 is S, N, or T; (d) X1YYX2X3 (SEQ ID NO: 181), wherein Xi is S, or D; X2 is W, or L; and X3 is S, or T; (e) X 1X2X3 YX4WX5 (SEQ ID NO: 204), wherein Xi is S or N; X2 is G or N; X3 is S, K, R, N, or G; X4 is Y, F, or N; and X5 is S or N; or (f) TSGVGVG (SEQ ID NO: 227). In some embodiments, the HCDR2 comprises one of the following amino acid sequences: (a) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 20), wherein Xi is W, I, or V; X2 is N, S, or Y; X3 is P, T, A, G, or Y; X4 is N, Y, G, D, H, or A; X5 is S, T, N, G, or D; X6 is G, or S; X7 is D, G, T, N, V, A, or H; X8 is T, P, or K; X9 is N, Y, T, H, R, I, D, or K; X10 is Y, or A; Xu is V, Q, A, or S; X12 is Q, K, P, or D; X13 is N, F, K, G, S, or Q; X14 is F, Q, L, or V; and X15 is Q, G, T, or K; (b) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 70), wherein Xi is G, R, N, S, H, T, V, or A; X2 is S, K, N, or G; X3 is W, S, Q, T, G, or Y; X4 is N, K, D, S, A, or Y; X5 is S, T, or G; X6 is G, D, or S; X7 is S, G, E, Y, T, or N; X8 is I, G, T, K, or Y; X9 is A, T, S, Y, or N; X10 is Y, T, P, or F; Xu is A, D, or V; X12 is D, Y, S, or A; X13 is S, A, or V; X14 is V, A, or K; and X15 is K, P, Q, or G; (c) X1IX2X3SX4X5X6X7X8NPSLX9S (SEQ ID NO: 124), wherein Xi is K, Q, R, E, or D; X2 is Y or F; X3 is H or Q; X4 is G, E, or R; X5 is N, I, S, H, F, or T; Xe is T or I; X7 is D, N, or M; X8 is Y or H; and X9 is K or R; (d) X1X2X3X4X5GX6TX7YNPSLKS (SEQ ID NO: 189) wherein Xi is Y or E; X2 is V or I; X3 is Y or N; X4 is Y or H; X5 is S or I; Xe is Y, F, or S; and X7 is N or K; (e) YIX1X2SGX3TX4YNPSLKX5 (SEQ ID NO: 212) wherein Xi is Y or N; X2 is K, Y, or S; X3 is F, S, Y, or N; X4 is N, K, or Y; and X5 is S or N; or (f) LIYWNX1X2X3X4YSPSLX5S (SEQ ID NO: 230) wherein Xi is G or D; X2 is D or V; X3 is E or K; X4 is R or F; and X5 is K or Q. In some embodiment, the HCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 39-56; (b) SEQ ID NOs: 83-94; (c) SEQ ID NOs: 153-
180; (d) SEQ ID NOs: 197-203; (e) SEQ ID NOs: 220-226; or (f) SEQ ID NOs: 234-235. In accordance with these embodiments, the LCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 237-267, SEQ ID NOs: 332-333, SEQ ID NOs: 341-348, or SEQ ID NOs: 368-401; the LCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 269-299, SEQ ID NOs: 335- 336, SEQ ID NOs: 350-357, or SEQ ID NOs 403-436; and the LCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 300-330, SEQ ID NOs: 339-339, SEQ ID NOs: 359-366, or SEQ ID NOs: 438-471.
[0010] Embodiments of the present disclosure also include antibodies directed against Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, including antigen binding fragments thereof, comprising a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3. In some embodiments, the LCDR1 comprises one of the following amino acid sequences: (a) X1ASQX2X3X4X5X6X7X8 (SEQ ID NO: 236), wherein Xi is R or Q; X2 is D, N, T, G, or S; X3 is V or I; X4 is S, I, R, G, T, or K; X5 is T, N, R, or S; X6 is A, W, F, D, Y, N, or C; X7 is V or L; X8 is A, N, G, or H; (b) RSSQX1LLHX2NGYNYLD (SEQ ID NO: 331), wherein Xi is S or I; X2 is S or T; or (c) RASQSX1X2X3X4X5X6 (SEQ ID NO: 340), wherein Xi is V or I; X2 is S or R; X3 is S, D, or R; X4 is Y or S; X5 is L or Y; X6 is A, F, or L; or (d) KSSQX1X2LYX3SNX4KNYLA (SEQ ID NO: 367), wherein Xi is S or T; X2 is V or I; X3 is R or S; X4 is N or I. In some embodiments, the LCDR2 comprises one of the following amino acid sequences: (a) X1ASX2X3X4X5 (SEQ ID NO: 268), wherein Xi is S, K, A, Y, T, D, or V; X2 is F, S, N, Q, or T; X3 is L, S, or F; X4 is Y, E, Q, or F; X5 is S or T; (b) LGSXiRAS (SEQ ID NO: 334), wherein Xi is N or Y; (c) X1ASX2RAT (SEQ ID NO: 349), wherein Xi is D or G; X2 is S or N; or (d) WASXiRES (SEQ ID NO: 402), wherein Xi is N or T. In some embodiments, the LCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 300-330; (b) MQX1LQTPX2T (SEQ ID NO: 337), wherein Xi is T or A; X2 is P or L; (c) QQX1X2X3X4PX5T (SEQ ID NO: 358), wherein Xi is R or Y; X2 is S, G, or D; X3 is N or S; X4 is W or S; X5 is L, R, I, Y, F, or W; or (d) QQX1X2X3TX4X5 (SEQ ID NO: 437), wherein Xi is Y or F; X2 is Y or F; X3 is S, G, or D; X4 is P or R; X5 is Y, W, R, T, or P. In accordance with these embodiments, the
HCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 2-19, SEQ ID NOs: 58-69, SEQ ID NOs: 96-123, SEQ ID NOs: 182-188, SEQ ID NOs: 205-211, or SEQ ID NOs: 228-229; the HCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 21-38, SEQ ID NOs: 71- 82, SEQ ID NOs: 125-152, SEQ ID NOs 190-196, SEQ ID NOs 213-219, or SEQ ID NOs 231- 232; and the HCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 39-56, SEQ ID NOs: 83-94, SEQ ID NOs: 153-180, SEQ ID NOs: 197-203, SEQ ID NOs: 220-226, or SEQ ID NOs: 234-235.
[0011] In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 2; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 21; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 39. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 3; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 22; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 40. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 4; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 23; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 41. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 5; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 24; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 42. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 6; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 25; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 43. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 7; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 26; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 44. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 8; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 27; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 45. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 9; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 28; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 46. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 10; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 29; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 47. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 11; the HCDR2 comprises the
amino acid sequence of SEQ ID NO: 30; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 48. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 12; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 31; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 49. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 13; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 32; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 50. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 14; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 33; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 51. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 15; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 34; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 52. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 16; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 35; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 53. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 17; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 36; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 54. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 18; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 37; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 55. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 19; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 38; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 56. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 58; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 71; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 83. In some embodiments, the amino acid sequence of SEQ ID NO: 59; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 72; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 84. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 60; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 73; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 85. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 61; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 74; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 86. In some embodiments,
the HCDR1 comprises the amino acid sequence of SEQ ID NO: 62; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 75; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 87. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 63; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 76; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 88. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 64; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 77; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 89. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 65; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 78; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 90. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 66; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 79; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 91. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 67; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 80; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 92. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 68; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 81; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 93. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 69; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 82; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 94. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 96; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 125; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 153. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 97; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 126; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 154. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 98; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 127; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 155. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 99; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 128; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 156. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 100; the HCDR2 comprises the
amino acid sequence of SEQ ID NO: 129; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 157. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 101; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 130; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 158. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 102; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 131; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 159. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 103; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 132; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 160. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 104; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 133; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 161. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 105; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 134; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 162. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 106; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 135; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 163. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 107; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 136; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 164. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 108; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 137; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 165. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 109; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 138; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 166. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 110; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 139; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 167. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 111; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 140; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 168. In some embodiments, HCDR1 comprises the amino acid sequence of SEQ ID NO: 112; the HCDR2 comprises the amino acid sequence of
SEQ ID NO: 141; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 169. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 113; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 142; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 170. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 114; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 143; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 171. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 115; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 144; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 172. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 116; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 145; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 173. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 117; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 146; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 174. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 118; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 147; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 175. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 119; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 148; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 176. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 120; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 149; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 177. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 121; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 150; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 178. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 122; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 151; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 179. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 123; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 152; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 180. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 182; the HCDR2 comprises the amino acid sequence of SEQ ID NO:
190; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 197. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 183; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 191; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 198. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 184; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 192; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 199. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 185; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 193; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 200. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 186; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 194; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 201. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 187; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 195; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 202. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 188; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 196; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 203. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 205; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 213; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 220. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 206; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 214; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 221. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 207; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 215; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 222. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 208; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 216; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 223. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 209; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 217; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 224. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 210; the HCDR2 comprises the amino acid sequence of SEQ ID NO:
218; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 225. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 211; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 219; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 226. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 228; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 231; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 234. In some embodiments, the HCDR1 comprises the amino acid sequence of SEQ ID NO: 229; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 232; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 35.
[0012] In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 237; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 269; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 300. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 238; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 270; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 301. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 239; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 271; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 302. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 240; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 272; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 303. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 241; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 273; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 304. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 242; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 274; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 305. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 243; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 275; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 306. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 244; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 276; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 307. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 245; the LCDR2
comprises the amino acid sequence of SEQ ID NO: 277; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 308. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 246; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 278; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 309. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 247; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 279; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 310. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 248; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 280; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 311. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 249; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 281; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 312. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 250; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 282; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 313. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 251; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 283; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 314. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 252; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 284; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 315. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 253; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 285; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 316. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 254; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 286; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 317. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 255; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 287; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 318. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 256; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 288; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 319. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 257; the LCDR2
comprises the amino acid sequence of SEQ ID NO: 289; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 320. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 258; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 290; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 321. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 259; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 291; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 322. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 260; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 292; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 323. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 261; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 293; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 324. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 262; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 294; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 325. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 263; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 295; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 326. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 264; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 296; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 327. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 265; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 297; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 328. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 266; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 298; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 329. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 267; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 299; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 330. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 332; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 335; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 338. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 333; the LCDR2
comprises the amino acid sequence of SEQ ID NO: 336; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 339. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 341; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 350; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 359. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 342; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 351; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 360. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 343; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 352; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 361. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 344; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 353; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 362. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 345; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 354; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 363. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 346; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 355; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 364. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 347; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 356; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 365. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 348; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 357; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 366. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 368; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 403; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 438. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 369; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 404; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 439. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 370; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 405; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 440. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 371; the LCDR2
comprises the amino acid sequence of SEQ ID NO: 406; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 441. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 372; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 407; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 442. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 373; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 408; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 443. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 374; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 409; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 444. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 375; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 410; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 445. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 376; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 411; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 446. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 377; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 412; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 447. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 378; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 413; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 448. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 379; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 414; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 449. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 380; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 415; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 450. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 381; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 416; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 451. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 382; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 417; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 452. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 383; the LCDR2
comprises the amino acid sequence of SEQ ID NO: 418; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 453. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 384; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 419; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 454. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 385; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 420; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 455. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 386; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 421; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 456. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 387; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 422; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 457. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 388; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 423; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 458. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 389; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 424; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 459. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 390; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 425; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 460. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 391; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 426; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 461. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 392; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 427; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 462. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 393; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 428; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 463. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 394; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 429; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 464. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 395; the LCDR2
comprises the amino acid sequence of SEQ ID NO: 430; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 465. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 396; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 431; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 466. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 397; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 432; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 467. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 398; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 433; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 468. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 399; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 434; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 469. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 400; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 435; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 470. In some embodiments, the LCDR1 comprises the amino acid sequence of SEQ ID NO: 401; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 436; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 471.
[0013] In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to any of SEQ ID NOs: 546-619. In some embodiments, the VL comprises an amino acid sequence that is at least 90% identical to any of SEQ ID NOs: 694-767.
[0014] In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 546 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 694. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 547 and the and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 695. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 548 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 696. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 549 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 697. In some embodiments, the VH comprises an amino acid sequence that is at least 90%
identical to SEQ ID NO: 550 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 698. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 551 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 699. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 552 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 700. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 553 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 701. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 554 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 702. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 555 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 703. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 556 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 704. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 557 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 705. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 558 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 706. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 559 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 707. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 560 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 708. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 561 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 709. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 562 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 710. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 563 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 711. In some
embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 564 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 712. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 565 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 713. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 566 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 714. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 567 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 715. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 568 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 716. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 569 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 717. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 570 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 718. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 571 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 719. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 572 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 720. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 573 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 721. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 574 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 722. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 575 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 723. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 576 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 724. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 577 and the VL comprises an
amino acid sequence that is at least 90% identical to SEQ ID NO: 725. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 578 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 726. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 579 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 727. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 580 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 728. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 581 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 729. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 582 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 730. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 583 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 731. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 584 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 732. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 585 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 733. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 586 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 734. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 587 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 735. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 588 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 736. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 589 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 737. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 590 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 738. In some embodiments, the VH comprises an amino acid sequence
that is at least 90% identical to SEQ ID NO: 591 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 739. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 592 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 740. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 593 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 741. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 594 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 742. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 595 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 743. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 596 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 744. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 597 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 745. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 598 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 746. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 599 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 747. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 600 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 748. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 601 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 749. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 602 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 750. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 603 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 751. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 604 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID
NO: 752. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 605 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 753. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 606 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 754. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 607 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 755. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 608 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 756. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 609 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 757. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 610 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 758. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 611 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 759. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 612 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 760. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 613 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 761. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 614 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 762. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 615 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 763. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 616 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 764. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 617 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 765. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 618 and the
VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 766. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 619 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 767.
[0015] In some embodiments, the anti-C3 antibodies of the present disclosure bind an epitope from a C3 polypeptide, or fragment thereof, having an amino acid sequence of SEQ ID NO: 1064. In some embodiments, the anti-C3 antibodies of the present disclosure bind C3, C3a, and/or C3b, and reduce complement activation. In some embodiments, the anti-C3 antibodies of the present disclosure bind C3, C3a, and/or C3b, with a KD of about 100 nM or lower. In some embodiments, the anti-C3 antibodies of the present disclosure bind C3, C3a, and/or C3b, and attenuate C3 convertase activity.
[0016] In some embodiments, the antibody is a monoclonal antibody, a human antibody, a humanized antibody, and/or a chimeric antibody. In some embodiments, the antibody is a fragment selected from the group consisting of Fab, Fab-C, Fab'-SH, Fv, scFv, and (Fab')? fragments. In some embodiments, the antibody is a monospecific antibody. In some embodiments, the antibody is a bispecific antibody.
[0017] In some embodiments, the antibody comprises a detection moiety. In some embodiments, the antibody comprises a purification moiety.
[0018] In some embodiments, the antibody comprises a half-life extension moiety. In some embodiments, the half-life extension moiety comprises a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residues. In some embodiments, the half-life extension polypeptide is attached to the C-terminus or N-terminus of the antibody.
[0019] Embodiments of the present disclosure also includes a pharmaceutical composition comprising any of the anti-C3 antibodies described herein. In some embodiments, the composition is suitable for ocular administration. In some embodiments, ocular administration comprises injection into vitreous fluid. In some embodiments, ocular administration comprises delivering the antibody using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant.
[0020] Embodiments of the present disclosure also include a method of treating age-related macular degeneration (AMD) comprising administering a pharmaceutical composition comprising
an effective amount of any of the anti-C3 antibodies described herein to a subject in need thereof. In some embodiments, the pharmaceutical composition is administered ocularly and treats at least one AMD symptom. In some embodiments, AMD comprises wet AMD. In some embodiments, AMD comprises dry AMD. In some embodiments, at least one AMD symptom comprises visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light. In some embodiments, administering the pharmaceutical composition reduces complement activation in the subject’s eye. In some embodiments, the pharmaceutical composition is administered at a dose ranging from about 0.0001 mg/dose to about 100 mg/dose. In some embodiments, the pharmaceutical composition is administered at a dose ranging from about 0.0001 mg/ml to about 100 mg/ml.
[0021] Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 70% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 70% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
[0022] Embodiments of the present disclosure also include an expression vector comprising any of the polynucleotides described herein. In some embodiments, the expression vector is at least one of: an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, and a retrovirus vector.
[0023] Embodiments of the present disclosure also include a method of administering ocular gene therapy to a subject in need thereof comprising injecting a pharmaceutical composition comprising an effective amount of an expression vector described herein.
[0024] Embodiments of the present disclosure also include a method of treating age-related macular degeneration (AMD) comprising administering a pharmaceutical composition comprising an effective amount of an expression vector described herein, wherein administering the pharmaceutical composition treats at least one AMD symptom.
BRIEF DESCRIPTION OF THE DRAWING(S)
[0025] FIGS. 1 A-1D: Representative ELISA results used to determine antigen positive serum titers of mice immunized with human C3 and cyno C3, according to different immunization protocols/cohorts (Cohort 1: FIG. 1A (human C3) and FIG. IB (cyno C3); Cohort 2: FIG. 1C (human C3); FIG. ID (cyno C3)).
[0026] FIG. 2: Representing ELISA results of polyclonal phage pools to confirm enrichment and examine cross reactivity.
[0027] FIG. 3: Representative table containing binding kinetics of the C3 antibodies of the present disclosure.
[0028] FIG. 4: Representative table containing results of C3 antibody cross-reactivity with human C3b protein, Cynomolgus monkey C3 protein, and human C3a protein.
[0029] FIGS. 5A-5B: Representative tables containing binding kinetics of C3 antibodies with respect to human C3 protein (FIG. 5A) and human C3b protein (FIG. 5B).
[0030] FIGS. 6A-6D: Representative results of C3 antibody epitope experiments, including a table containing binning allocations (FIG. 6A), data from a representative heatmap analyzing the ability of the antibodies to block one another for binding to the antigen (FIG. 6B), and representative network plots (FIG. 6C and FIG. 6D), which progressively groups antibodies with similar competition profiles.
[0031] FIGS. 7A-7E: Representative binning network plots based on recombinant human affinities and cyno binding performed with C3b (FIG. 7A); C3b affinity and ligand blocking (FIG. 7B); bins vs. communities for CFHR4b blocking (FIG. 7C); bins vs. communities for CFH blocking (FIG. 7D); and material availability and common vs. diverse light chains (FIG. 7E).
[0032] FIG. 8: Representative results of C3 antibody polyreactivity with baculo virus particles (BVPs).
[0033] FIG. 9: Representative results of C3 antibody polyreactivity with the various proteins indicated.
[0034] FIGS. 10A-10B: Representative results of C3 antibody polyreactivity with CFHR4 (FIG. 10A) and CFH (FIG. 10B).
[0035] FIG. 11 : Representative results of the effects of C3 antibodies on alternative pathway complement activation based on the quantification of red blood cell hemolysis.
DETAILED DESCRIPTION
[0036] Embodiments of the present disclosure relate to the treatment and/or prevention of age- related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA). In particular, the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, and antigen binding fragments thereof.
[0037] The human complement system comprises a family of proteins that are essential to the human immune response against infections. Complement recognizes microbes or damaged host cells and subsequently triggers an enzymatic cascade that mainly serves to label target cells for phagocytosis by immune cells, produce chemoattractants, and directly kill target cells via pore formation. Unwanted complement activation on the body’s own cells is a key pathological driver in a wide spectrum of immune diseases including autoimmune, inflammatory, and degenerative diseases. For current and future development of therapeutic complement inhibitors, knowledge of complement activation and how it can be regulated is of great importance.
[0038] Convertase enzymes fulfill a central role in the complement cascade as they cleave C3 and C5, which mediate nearly all complement effector functions. C3 convertases cleave C3 into C3a, a chemoattractant molecule, and C3b, which covalently binds to target surfaces and triggers phagocytosis. C5 convertases cleave C5 into C5a, a potent mediator of leukocyte recruitment and inflammation, and C5b, the initiator of the membrane attack complex and cell lysis. The complement cascade begins via specific recognition of target cells in the classical (CP) and lectin (LP) pathways. In the CP, antibodies bind epitopes on the target cell and subsequently recruit the Cl complex (Clqr2S2). Upon binding to the antibody platforms, Clq-associated protease Cis
converts C4 and C2 to generate a C3 convertase enzyme (C4b2a) on the cell surface. Similarly, the lectin pathway also forms C4b2a via activation of mannose-binding lectin-associated serine proteases. The resulting CP/LP C4b2a convertases cleave C3 into C3a and C3b. Following cleavage, a reactive thioester in C3b is exposed, which enables its covalent attachment to target cell surfaces, leading to recognition of the cells by phagocytes. The labeling of target cells with C3b is amplified by the alternative pathway (AP) in which surface-bound C3b binds factor B (FB). The proconvertase C3bB is then cleaved by factor D (FD) to form an active C3 convertase complex that consists of C3b and the protease fragment Bb (C3bBb). Since the resulting active AP C3 convertase (C3bBb) is comprised of C3b itself, substrate cleavage results in generation of additional convertases, further propagating C3b deposition. When the density of C3b molecules on the cell surface becomes sufficiently high, the existing C3 convertases (C4b2a and C3bBb) gain the ability to cleave C5, leading to formation of C5a and C5b.
[0039] Age-related macular degeneration (AMD) is a progressive retinal disease in which the early stage is characterized by relatively few small drusen within the macula. When AMD progresses, drusen size and number increase, eventually leading towards more advanced stages of AMD. Two forms of advanced AMD are distinguished. The first form, neovascular AMD, is characterized by infiltration of abnormal blood vessels into the retina. These newly formed vessels are fragile and when they break, the leakage of blood constituents in the retina leads to sudden vision loss. The second form of advanced AMD, geographic atrophy (GA), is the result of gradual degeneration of the RPE and photoreceptors cells. Although neovascularization occurs in only 15- 20% of AMD cases, it is responsible for the vast majority of vision loss caused by AMD. Drugs targeting vascular endothelial growth factor (VEGF), one of the central molecules in neovascularization, have proven to be very successful in neovascular AMD. However, no treatment is available for the remaining majority of early, intermediate or geographic atrophy AMD cases, and furthermore there are no effective means of preventing progression of early to advanced stages. Although it is known that AMD is the result of a complex interaction of environmental and genetic risk factors, studies into the molecular constituents of drusen suggest that AMD may have an immunological component. This suggestion arose after proteins involved in inflammation and/or other immune-associated responses, including components of the complement system, were found within drusen.
[0040] In light of this, experiments were performed to determine the role that C3, C3a, and C3b play in the etiology of AMD and GA, and concomitantly, to develop a therapeutic platform based on modulating C3 activity using anti-C3 antibodies.
Definitions
[0041] To facilitate an understanding of the present technology, a number of terms and phrases are defined below. Additional definitions are set forth throughout the detailed description.
[0042] The use of the terms “a” and “an” and “the” and “at least one” and similar referents in the context of describing the embodiments of the present disclosure (especially in the context of the following claims) are to be construed to cover both the singular and the plural, unless otherwise indicated herein or clearly contradicted by context. The use of the term “at least one” followed by a list of one or more items (for example, “at least one of A and B”) is to be construed to mean one item selected from the listed items (A or B) or any combination of two or more of the listed items (A and B), unless otherwise indicated herein or clearly contradicted by context. The terms “comprising,” “having,” “including,” and “containing” are to be construed as open-ended terms (i.e., meaning “including, but not limited to,”) unless otherwise noted. Recitation of ranges of values herein are merely intended to serve as a shorthand method of referring individually to each separate value falling within the range, unless otherwise indicated herein, and each separate value is incorporated into the specification as if it were individually recited herein. All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly contradicted by context. The use of any and all examples, or exemplary language (e.g., “such as”) provided herein, is intended merely to better illuminate the various embodiments of the present disclosure, and does not pose a limitation on the scope of these embodiment unless otherwise claimed. No language in the specification should be construed as indicating any nonclaimed element as essential to the practice of the various embodiments of the present disclosure. [0043] As used herein, the term “or” is an inclusive “or” operator and is equivalent to the term “and/or” unless the context clearly dictates otherwise. The term “based on” is not exclusive and allows for being based on additional factors not described, unless the context clearly dictates otherwise. In addition, throughout the specification, the meaning of “a,” “an,” and “the” include plural references. The meaning of “in” includes “in” and “on.”
[0044] The transitional phrase “consisting essentially of’ as used in claims in the present application limits the scope of a claim to the specified materials or steps “and those that do not materially affect the basic and novel characteristic(s)” of the claimed invention, as discussed in In re Herz, 537 F.2d 549, 551-52, 190 USPQ 461, 463 (CCPA 1976). For example, a composition “consisting essentially of’ recited elements may contain an unrecited contaminant at a level such that, though present, the contaminant does not alter the function of the recited composition as compared to a pure composition, i.e., a composition “consisting of’ the recited components.
[0045] The term “one or more,” as used herein, refers to a number higher than one. For example, the term “one or more” encompasses any of the following: two or more, three or more, four or more, five or more, six or more, seven or more, eight or more, nine or more, ten or more, eleven or more, twelve or more, thirteen or more, fourteen or more, fifteen or more, twenty or more, fifty or more, 100 or more, or an even greater number.
[0046] The term “one or more but less than a higher number,” “two or more but less than a higher number,” “three or more but less than a higher number,” “four or more but less than a higher number,” “five or more but less than a higher number,” “six or more but less than a higher number,” “seven or more but less than a higher number,” “eight or more but less than a higher number,” “nine or more but less than a higher number,” “ten or more but less than a higher number,” “eleven or more but less than a higher number,” “twelve or more but less than a higher number,” “thirteen or more but less than a higher number,” “fourteen or more but less than a higher number,” or “fifteen or more but less than a higher number” is not limited to a higher number. For example, the higher number can be 10,000, 1,000, 100, 50, etc. For example, the higher number can be approximately 50 (e.g., 50, 49, 48, 47, 46, 45, 44, 43, 42, 41, 40, 39, 38, 37, 36, 35, 34, 33, 32, 31, 32, 30, 29, 28, 27, 26, 25, 24, 23, 22, 21, 20, 19, 18, 17, 16, 15, 14, 13, 12, 11, 10, 9, 8, 7, 6, 5, 4, 3 or 2).
[0047] The term “immunoglobulin” or “antibody,” as used herein, refers to a protein that is found in blood or other bodily fluids of vertebrates, which is used by the immune system to identify and neutralize foreign objects, such as bacteria and viruses. Typically, an immunoglobulin or antibody is a protein that comprises at least one complementarity determining region (CDR). The CDRs form the “hypervariable region” of an antibody, which is responsible for antigen binding (discussed further below). A whole antibody typically consists of four polypeptides: two identical
copies of a heavy (H) chain polypeptide and two identical copies of a light (L) chain polypeptide. Each of the heavy chains contains one N-terminal variable (VH) region and three C-terminal constant (CHI, CH2, and Cm) regions, and each light chain contains one N-terminal variable (VL) region and one C-terminal constant (CL) region. The light chains of antibodies can be assigned to one of two distinct types, either kappa (K) or lambda (X), based upon the amino acid sequences of their constant domains. In a typical antibody, each light chain is linked to a heavy chain by disulfide bonds, and the two heavy chains are linked to each other by disulfide bonds. The light chain variable region is aligned with the variable region of the heavy chain, and the light chain constant region is aligned with the first constant region of the heavy chain. The remaining constant regions of the heavy chains are aligned with each other.
[0048] The variable regions of each pair of light and heavy chains form the antigen binding site of an antibody. The VH and VL regions have the same general structure, with each region comprising four framework (FW or FR) regions. The term “framework region,” as used herein, refers to the relatively conserved amino acid sequences within the variable region which are located between the CDRs. There are four framework regions in each variable domain, which are designated FR1, FR2, FR3, and FR4. The framework regions form the [:) sheets that provide the structural framework of the variable region (see, e.g., C. A. Janeway et al. (eds.), Immunobiology, 5th Ed., Garland Publishing, New York, N.Y. (2001)).
[0049] The framework regions are connected by three CDRs. As discussed above, the three CDRs, known as CDR1, CDR2, and CDR3, form the “hypervariable region” of an antibody, which is responsible for antigen binding. The CDRs form loops connecting, and in some cases comprising part of, the beta-sheet structure formed by the framework regions. While the constant regions of the light and heavy chains are not directly involved in binding of the antibody to an antigen, the constant regions can influence the orientation of the variable regions. The constant regions also exhibit various effector functions, such as participation in antibody-dependent complement- mediated lysis or antibody-dependent cellular toxicity via interactions with effector molecules and cells.
[0050] As used herein, when an antibody or other entity (e.g., antigen binding domain) “specifically recognizes” or “specifically binds” an antigen or epitope, it preferentially recognizes the antigen in a complex mixture of proteins and/or macromolecules, and binds the antigen or
epitope with affinity which is substantially higher than to other entities not displaying the antigen or epitope. In this regard, “affinity which is substantially higher” means affinity that is high enough to enable detection of an antigen or epitope which is distinguished from entities using a desired assay or measurement apparatus. Typically, it means binding affinity having a binding constant (Ka) of at least 107 M'1 (e.g., >107 M’1, >108 M’1, >109 M’1, >1O10 M’1, >10n M’1, >1012 M’1, >1013 M’1, etc.). In certain such embodiments, an antibody is capable of binding different antigens so long as the different antigens comprise that particular epitope. In certain instances, for example, homologous proteins from different species may comprise the same epitope.
[0051] The terms “fragment of an antibody,” “antibody fragment,” and “antigen-binding fragment” of an antibody are used interchangeably herein to refer to one or more fragments of an antibody that retain the ability to specifically bind to an antigen (see, generally, Holliger et al., Nat. Biotech., 25(9): 1126-1129 (2005)). Any antigen-binding fragment of the antibody described herein is within the scope of the present disclosure. The antibody fragment desirably comprises, for example, one or more CDRs, the variable region (or portions thereof), the constant region (or portions thereof), or combinations thereof. Examples of antibody fragments include, but are not limited to, (i) a Fab fragment, which is a monovalent fragment consisting of the VL, VH, CL, and CHI domains, (ii) a F(ab’)2 fragment, which is a bivalent fragment comprising two Fab fragments linked by a disulfide bridge at the hinge region, (iii) a Fv fragment consisting of the VL and VH domains of a single arm of an antibody, (iv) a Fab’ fragment, which results from breaking the disulfide bridge of an F(ab’)2 fragment using mild reducing conditions, (v) a disulfide-stabilized Fv fragment (dsFv), and (vi) a domain antibody (dAb), which is an antibody single variable region domain (VH or VL) polypeptide that specifically binds antigen.
[0052] The term “monoclonal antibody,” as used herein, refers to an antibody produced by a single clone of B lymphocytes that is directed against a single epitope on an antigen. Monoclonal antibodies typically are produced using hybridoma technology, as first described in Kohler and Milstein, Eur. J. Immunol., 5: 511-519 (1976). Monoclonal antibodies may also be produced using recombinant DNA methods (see, e.g., U.S. Patent 4,816,567), isolated from phage display antibody libraries (see, e.g., Clackson et al. Nature, 352: 624-628 (1991)); and Marks et al., J. Mol. Biol., 222: 581-597 (1991)), or produced from transgenic mice carrying a fully human immunoglobulin system (see, e.g., Lonberg, Nat. Biotechnol., 25(9): 1117-25 (2005), and Lonberg,
Handb. Exp. Pharmacol., 181: 69-97 (2008)). In contrast, “polyclonal” antibodies are antibodies that are secreted by different B cell lineages within an animal. Polyclonal antibodies are a collection of immunoglobulin molecules that recognize multiple epitopes on the same antigen.
[0053] The terms “nucleic acid,” “polynucleotide,” “nucleotide sequence,” and “oligonucleotide” are used interchangeably herein and refer to a polymer or oligomer of pyrimidine and/or purine bases, preferably cytosine, thymine, and uracil, and adenine and guanine, respectively (See Albert L. Lehninger, Principles of Biochemistry, at 793-800 (Worth Pub. 1982)). The terms encompass any deoxyribonucleotide, ribonucleotide, or peptide nucleic acid component, and any chemical variants thereof, such as methylated, hydroxymethylated, or glycosylated forms of these bases. The polymers or oligomers may be heterogenous or homogenous in composition, may be isolated from naturally occurring sources, or may be artificially or synthetically produced. In addition, the nucleic acids may be DNA or RNA, or a mixture thereof, and may exist permanently or transitionally in single-stranded or double-stranded form, including homoduplex, heteroduplex, and hybrid states. In some embodiments, a nucleic acid or nucleic acid sequence comprises other kinds of nucleic acid structures such as, for instance, a DNA/RNA helix, peptide nucleic acid (PNA), morpholino nucleic acid (see, e.g., Braasch and Corey, Biochemistry, 41()A): 4503-4510 (2002) and U.S. Patent 5,034,506), locked nucleic acid (LNA; see Wahlestedt et al., Proc. Natl. Acad. Sci. U.S.A., 97: 5633-5638 (2000)), cyclohexenyl nucleic acids (see Wang, J. Am. Chem. Soc., 122: 8595-8602 (2000)), and/or a ribozyme. The terms “nucleic acid” and “nucleic acid sequence” may also encompass a chain comprising nonnatural nucleotides, modified nucleotides, and/or non-nucleotide building blocks that can exhibit the same function as natural nucleotides (e.g., “nucleotide analogs”).
[0054] The terms “peptide,” “polypeptide,” and “protein” are used interchangeably herein and refer to a polymeric form of amino acids of any length, which can include coded and non-coded amino acids, chemically or biochemically modified or derivatized amino acids, and polypeptides having modified peptide backbones.
[0055] As used herein, a “nucleic acid” or “nucleic acid molecule” generally refers to any ribonucleic acid or deoxyribonucleic acid, which may be unmodified or modified DNA or RNA. “Nucleic acids” include, without limitation, single- and double-stranded nucleic acids. As used herein, the term “nucleic acid” also includes DNA as described above that contains one or more
modified bases. Thus, DNA with a backbone modified for stability or for other reasons is a “nucleic acid.” The term “nucleic acid” as it is used herein embraces such chemically, enzymatically, or metabolically modified forms of nucleic acids, as well as the chemical forms of DNA characteristic of viruses and cells, including for example, simple and complex cells.
[0056] The terms “oligonucleotide” or “polynucleotide” or “nucleotide” or “nucleic acid” refer to a molecule having two or more deoxyribonucleotides or ribonucleotides, preferably more than three, and usually more than ten. The exact size will depend on many factors, which in turn depends on the ultimate function or use of the oligonucleotide. The oligonucleotide may be generated in any manner, including chemical synthesis, DNA replication, reverse transcription, or a combination thereof. Typical deoxyribonucleotides for DNA are thymine, adenine, cytosine, and guanine. Typical ribonucleotides for RNA are uracil, adenine, cytosine, and guanine.
[0057] The terms “complementary” and “complementarity” refer to nucleotides (e.g., 1 nucleotide) or polynucleotides (e.g., a sequence of nucleotides) related by the base-pairing rules. For example, the sequence 5’-A-G-T-3’ is complementary to the sequence 3'-T-C-A-5'. Complementarity may be “partial,” in which only some of the nucleic acids’ bases are matched according to the base pairing rules. Or, there may be “complete” or “total” complementarity between the nucleic acids. The degree of complementarity between nucleic acid strands affects the efficiency and strength of hybridization between nucleic acid strands. This is of particular importance in amplification reactions and in detection methods that depend upon binding between nucleic acids.
[0058] The term “gene” refers to a nucleic acid (e.g., DNA or RNA) sequence that comprises coding sequences necessary for the production of an RNA, or of a polypeptide or its precursor. A functional polypeptide can be encoded by a full-length coding sequence or by any portion of the coding sequence as long as the desired activity or functional properties (e.g., enzymatic activity, ligand binding, signal transduction, etc.) of the polypeptide are retained. The term “portion” when used in reference to a gene refers to fragments of that gene. The fragments may range in size from a few nucleotides to the entire gene sequence minus one nucleotide. Thus, “a nucleotide comprising at least a portion of a “gene” may comprise fragments of the gene or the entire gene.
[0059] The term “gene” also encompasses the coding regions of a structural gene and includes sequences located adjacent to the coding region on both the 5' and 3' ends, e.g., for a distance of
about 1 kb on either end, such that the gene corresponds to the length of the full-length mRNA (e.g., comprising coding, regulatory, structural and other sequences). The sequences that are located 5' of the coding region and that are present on the mRNA are referred to as 5' non- translated or untranslated sequences. The sequences that are located 3' or downstream of the coding region and that are present on the mRNA are referred to as 3' non-translated or 3' untranslated sequences. The term “gene” encompasses both cDNA and genomic forms of a gene. In some organisms (e.g., eukaryotes), a genomic form or clone of a gene contains the coding region interrupted with noncoding sequences termed “introns” or “intervening regions” or “intervening sequences.” Introns are segments of a gene that are transcribed into nuclear RNA (hnRNA); introns may contain regulatory elements such as enhancers. Introns are removed or “spliced out” from the nuclear or primary transcript; introns therefore are absent in the messenger RNA (mRNA) transcript. The mRNA functions during translation to specify the sequence or order of amino acids in a nascent polypeptide.
[0060] In addition to containing introns, genomic forms of a gene may also include sequences located on both the 5' and 3' ends of the sequences that are present on the RNA transcript. These sequences are referred to as “flanking” sequences or regions (these flanking sequences are located 5' or 3' to the non-translated sequences present on the mRNA transcript). The 5' flanking region may contain regulatory sequences such as promoters and enhancers that control or influence the transcription of the gene. The 3' flanking region may contain sequences that direct the termination of transcription, posttranscriptional cleavage, and polyadenylation.
[0061] The term “wild-type” when made in reference to a gene refers to a gene that has the characteristics of a gene isolated from a naturally occurring source. The term “wild-type” when made in reference to a gene product refers to a gene product that has the characteristics of a gene product isolated from a naturally occurring source. The term “wild-type” when made in reference to a protein refers to a protein that has the characteristics of a naturally occurring protein. The term “naturally-occurring” as applied to an object refers to the fact that an object can be found in nature. For example, a polypeptide or polynucleotide sequence that is present in an organism (including viruses) that can be isolated from a source in nature, and which has not been intentionally modified by the hand of a person in the laboratory is naturally-occurring. A wild-type gene is often that gene or allele that is most frequently observed in a population and is thus arbitrarily designated the
“normal” or “wild-type” form of the gene. In contrast, the term “modified” or “mutant” when made in reference to a gene or to a gene product refers, respectively, to a gene or to a gene product that displays modifications in sequence and/or functional properties (e.g., altered characteristics) when compared to the wild-type gene or gene product. It is noted that naturally-occurring mutants can be isolated; these are identified by the fact that they have altered characteristics when compared to the wild-type gene or gene product.
[0062] The term “allele” refers to a variation of a gene; the variations include but are not limited to variants and mutants, polymorphic loci, and single nucleotide polymorphic loci, frameshift, and splice mutations. An allele may occur naturally in a population, or it might arise during the lifetime of any particular individual of the population.
[0063] Thus, the terms “variant” and “mutant” when used in reference to a nucleotide sequence refer to a nucleic acid sequence that differs by one or more nucleotides from another, usually related, nucleotide acid sequence. A “variation” is a difference between two different nucleotide sequences; typically, one sequence is a reference sequence.
[0064] The terms “immunogen” and “antigen” are used interchangeably herein and refer to any molecule, compound, or substance that induces an immune response in an animal (e.g., a mammal). An “immune response” can entail, for example, antibody production and/or the activation of immune effector cells. An antigen in the context of the disclosure can comprise any subunit, fragment, or epitope of any proteinaceous or non-proteinaceous (e.g., carbohydrate or lipid) molecule that provokes an immune response in a mammal. The term “epitope” refers to a sequence of an antigen that is recognized by an antibody or an antigen receptor. Epitopes also are referred to in the art as “antigenic determinants.” In certain embodiments, an epitope is a region of an antigen that is specifically bound by an antibody. In certain embodiments, an epitope may include chemically active surface groupings of molecules such as amino acids, sugar side chains, phosphoryl, or sulfonyl groups. In certain embodiments, an epitope may have specific three- dimensional structural characteristics (e.g., a “conformational” epitope) and/or specific charge characteristics. The antigen can be a protein or peptide of viral, bacterial, parasitic, fungal, protozoan, prion, cellular, or extracellular origin, which provokes an immune response in a mammal, preferably leading to protective immunity.
[0065] A “pharmaceutically acceptable carrier” as used herein generally refers to an ingredient in a pharmaceutical formulation, other than an active ingredient, which is nontoxic to a subject. A pharmaceutically acceptable carrier includes, but is not limited to, a buffer, excipient, stabilizer, or preservative.
[0066] The term “pharmaceutical formulation” as used herein generally refers to a preparation which is in such form as to permit the biological activity of an active ingredient (e.g., an anti-C3 antibody, an antibody conjugate, a fusion protein, or a polymeric formulation) contained therein to be effective, and which contains no additional components which are unacceptably toxic to a subject to which the formulation would be administered.
[0067] As used herein, “treatment” (and grammatical variations thereof such as “treat” or “treating”) generally refers to clinical intervention in an attempt to alter the natural course of the individual being treated, and can be performed either for prophylaxis or during the course of clinical pathology. Desirable effects of treatment include, but are not limited to, preventing occurrence or recurrence of disease, alleviation of symptoms, diminishment of any direct or indirect pathological consequences of the disease, preventing metastasis, decreasing the rate of disease progression, amelioration or palliation of the disease state, and remission or improved prognosis. In some embodiments, anti-C3 antibodies of the present disclosure or other compositions that include an anti-C3 antibody of the present disclosure (e.g., an antibody conjugate, a fusion protein, or a polymeric formulation) are used to delay development of a disease or to slow the progression of a disease.
[0068] The term “half-life” as used herein generally refers to the time required for the concentration of a substance (e.g., an anti-3 antibody, an antibody conjugate, a fusion protein (e.g., a Fab fusion protein), or a polymeric formulation) to decrease by one-half, in vivo (e.g., in the eye (e.g., the vitreous)) or in vitro.
[0069] An “effective amount” of an agent, e.g., a pharmaceutical formulation, as used herein generally refers to an amount effective, at dosages and for periods of time necessary, to achieve the desired therapeutic or prophylactic result.
[0070] An “individual” or “subject” is a mammal. Mammals include, but are not limited to, domesticated animals (e.g., cows, sheep, cats, dogs, and horses), primates (e.g., humans and non-
human primates such as monkeys), rabbits, and rodents (e.g., mice and rats). In certain embodiments, the individual or subject is a human. A “subject” may be a “patient.”
Anti-C3 Antibodies
[0071] As described further herein, embodiments of the present disclosure relate to the treatment and/or prevention of age-related macular degeneration (AMD), including the advanced form of dry AMD referred to as Geographic Atrophy (GA). In particular, the present disclosure provides novel therapeutic antibodies that target components of the alternative pathway of the complement activation system, including Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b).
[0072] As described further herein, anti-C3 antibodies were generated, and their structural and functional properties were elucidated. Based on these data, embodiments of the present disclosure include anti-C3 antibodies, or antigen-binding fragments thereof, that are comprised of a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3. In some embodiments, the HCDR1 comprises one of the following amino acid sequences: (a) X1YX2X3X4 (SEQ ID NO: 1), wherein Xi is G, S, N, D, T, or A; X2 is Y, A, G, W, or T; X3 is L, M, I, or V; and X4 is H, N, S, or G; (b) X1X2 X3MX4 (SEQ ID NO: 57), wherein Xi is D, N, S, or A; X2 is Y, or A; X3 is A, W, Y, S, C, D, or G; and X4 is H, S, or N; (c) X1X2X3WWX4 (SEQ ID NO: 95), wherein Xi is I, S, R, N, or K; X2 is N, D, S, or G; X3 is N, S, T, D, Y, or H; and X4 is S, N, or T; (d) X1YYX2X3 (SEQ ID NO: 181), wherein Xi is S, or D; X2 is W, or L; and X3 is S, or T; (e) X1X2X3YX4WX5 (SEQ ID NO: 204), wherein Xi is S or N; X2 is G or N; X3 is S, K, R, N, or G; X4 is Y, F, or N; and X5 is S or N; or (f) TSGVGVG (SEQ ID NO: 227). In some embodiments, the HCDR2 comprises one of the following amino acid sequences: (a) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 20), wherein Xi is W, I, or V; X2 is N, S, or Y; X3 is P, T, A, G, or Y; X4 is N, Y, G, D, H, or A; X5 is S, T, N, G, or D; X6 is G, or S; X7 is D, G, T, N, V, A, or H; X8 is T, P, or K; X9 is N, Y, T, H, R, I, D, or K; X10 is Y, or A; Xu is V, Q, A, or S; X12 is Q, K, P, or D; X13 is N, F, K, G, S, or Q; X14 is F, Q, L, or V; and X15 is Q, G, T, or K; (b) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 70), wherein Xi is G, R, N, S, H, T, V, or A; X2 is S, K, N, or G; X3 is W, S, Q, T, G, or Y; X4 is N, K, D, S, A, or Y; X5 is S, T, or G;
X6 is G, D, or S; X7 is S, G, E, Y, T, or N; X8 is I, G, T, K, or Y; X9 is A, T, S, Y, or N; Xio is Y, T, P, or F; Xu is A, D, or V; X12 is D, Y, S, or A; X13 is S, A, or V; X14 is V, A, or K; and X15 is K, P, Q, or G; (c) X1IX2X3SX4X5X6X7X8NPSLX9S (SEQ ID NO: 124), wherein Xi is K, Q, R, E, or D; X2 is Y or F; X3 is H or Q; X4 is G, E, or R; X5 is N, I, S, H, F, or T; Xe is T or I; X7 is D, N, or M; X8 is Y or H; and X9 is K or R; (d) X1X2X3X4X5GX6TX7YNPSLKS (SEQ ID NO: 189) wherein Xi is Y or E; X2 is V or I; X3 is Y or N; X4 is Y or H; X5 is S or I; Xe is Y, F, or S; and X7 is N or K; (e) YIX1X2SGX3TX4YNPSLKX5 (SEQ ID NO: 212) wherein Xi is Y or N; X2 is K, Y, or S; X3 is F, S, Y, orN; X4 is N, K, or Y; andX5 is S orN; or (f) LIYWNX1X2X3X4YSPSLX5S (SEQ ID NO: 230) wherein Xi is G or D; X2 is D or V; X3 is E or K; X4 is R or F; and X5 is K or Q. In some embodiments, the HCDR3 comprises one of the following amino acid sequences: (a) SEQ ID NOs: 39-56; (b) SEQ ID NOs: 83-94; (c) SEQ ID NOs: 153-180; (d) SEQ ID NOs: 197- 203; (e) SEQ ID NOs: 220-226; or (f) SEQ ID NOs: 234-235.
[0073] Embodiments of the present disclosure also include antibodies directed against Complement Component 3 (C3), Complement Component 3a (C3a), and Complement Component 3b (C3b) polypeptides and proteins, including antigen binding fragments thereof, comprising a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3. In some embodiments, the LCDR1 comprises one of the following amino acid sequences: (a) XIASQX2X3X4X5X6X7X8 (SEQ ID NO: 236), wherein Xi is R or Q; X2 is D, N, T, G, or S; X3 is V or I; X4 is S, I, R, G, T, or K; X5 is T, N, R, or S; X6 is A, W, F, D, Y, N, or C; X7 is V or L; X8 is A, N, G, or H; (b) RSSQX1LLHX2NGYNYLD (SEQ ID NO: 331), wherein Xi is S or I; X2 is S or T; or (c) RASQSX1X2X3X4X5X6 (SEQ ID NO: 340), wherein Xi is V or I; X2 is S or R; X3 is S, D, or R; X4 is Y or S; X5 is L or Y; X6 is A, F, or L; or (d) KSSQX1X2LYX3SNX4KNYLA (SEQ ID NO: 367), wherein Xi is S or T; X2 is V or I; X3 is R or S; X4 is N or I. In some embodiments, the LCDR2 comprises one of the following amino acid sequences: (a) X1ASX2X3X4X5 (SEQ ID NO: 268), wherein Xi is S, K, A, Y, T, D, or V; X2 is F, S, N, Q, or T; X3 is L, S, or F; X4 is Y, E, Q, or F; X5 is S or T; (b) LGSXiRAS (SEQ ID NO: 334), wherein Xi is N or Y; (c) X1ASX2RAT (SEQ ID NO: 349), wherein Xi is D or G; X2 is S or N; or (d) WASXiRES (SEQ ID NO: 402), wherein Xi is N or T. In some embodiments, the LCDR3
comprises one of the following amino acid sequences: (a) SEQ ID NOs: 300-330; (b) MQX1LQTPX2T (SEQ ID NO: 337), wherein Xi is T or A; X2 is P or L; (c) QQX1X2X3X4PX5T (SEQ ID NO: 358), wherein Xi is R or Y; X2 is S, G, or D; X3 is N or S; X4 is W or S; X5 is L, R, I, Y, F, or W; or (d) QQX1X2X3TX4X5 (SEQ ID NO: 437), wherein Xi is Y or F; X2 is Y or F; X3 is S, G, or D; X4 is P or R; X5 is Y, W, R, T, or P. In accordance with these embodiments, the HCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 2-19, SEQ ID NOs: 58-69, SEQ ID NOs: 96-123, SEQ ID NOs: 182-188, SEQ ID NOs: 205-211, or SEQ ID NOs: 228-229; the HCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 21-38, SEQ ID NOs: 71- 82, SEQ ID NOs: 125-152, SEQ ID NOs 190-196, SEQ ID NOs 213-219, or SEQ ID NOs 231- 232; and the HCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 39-56, SEQ ID NOs: 83-94, SEQ ID NOs: 153-180, SEQ ID NOs: 197-203, SEQ ID NOs: 220-226, or SEQ ID NOs: 234-235.
[0074] In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 2; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 21; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 39. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 3; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 22; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 40. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 4; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 23; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 41. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 5; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 24; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 42. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 6; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 25; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 43. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 7; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 26; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 44. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 8; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 27; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 45. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 9; the HCDR2 comprises the amino acid sequence of SEQ ID NO:
28; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 46. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 10; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 29; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 47. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 11; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 30; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 48. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 12; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 31 ; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 49. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 13; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 32; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 50. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 14; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 33; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 51. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 15; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 34; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 52. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 16; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 35; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 53. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 17; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 36; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 54. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 18; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 37; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 55. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 19; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 38; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 56. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 58; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 71; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 83. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 59; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 72; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 84. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 60; the HCDR2 comprises the amino
acid sequence of SEQ ID NO: 73; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 85. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 61; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 74; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 86. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 62; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 75; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 87. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 63; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 76; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 88. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 64; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 77; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 89. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 65; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 78; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 90. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 66; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 79; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 91. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 67; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 80; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 92. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 68; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 81; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 93. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 69; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 82; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 94. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 96; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 125; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 153. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 97; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 126; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 154. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 98; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 127; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 155. In some embodiments, an anti-C3 antibody comprises the
HCDR1 of SEQ ID NO: 99; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 128; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 156. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 100; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 129; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 157. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 101; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 130; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 158. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 102; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 131; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 159. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 103; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 132; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 160. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 104; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 133; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 161. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 105; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 134; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 162. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 106; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 135; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 163. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 107; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 136; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 164. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 108; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 137; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 165. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 109; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 138; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 166. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 110; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 139; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 167. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 111; the HCDR2 comprises the amino acid sequence of
SEQ ID NO: 140; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 168. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 112; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 141; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 169. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 113; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 142; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 170. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 114; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 143; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 171. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 115; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 144; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 172. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 116; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 145; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 173. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 117; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 146; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 174. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 118; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 147; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 175. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 119; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 148; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 176. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 120; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 149; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 177. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 121; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 150; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 178. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 122; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 151; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 179. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 123; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 152; and the HCDR3 comprises the amino acid sequence of SEQ ID
NO: 180. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 182; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 190; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 197. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 183; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 191; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 198. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 184; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 192; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 199. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 185; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 193; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 200. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 186; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 194; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 201. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 187; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 195; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 202. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 188; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 196; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 203. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 205; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 213; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 220. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 206; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 214; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 221. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 207; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 215; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 222. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 208; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 216; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 223. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 209; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 217; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 224. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO:
210; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 218; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 225. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 211; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 219; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 226. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 228; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 231; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 234. In some embodiments, an anti-C3 antibody comprises the HCDR1 of SEQ ID NO: 229; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 232; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 35.
[0075] In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 237; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 269; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 300. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 238; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 270; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 301. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 239; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 271; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 302. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 240; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 272; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 303. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 241; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 273; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 304. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 242; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 274; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 305. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 243; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 275; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 306. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 244; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 276; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 307. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 245; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 277; and the
LCDR3 comprises the amino acid sequence of SEQ ID NO: 308. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 246; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 278; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 309. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 247; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 279; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 310. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 248; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 280; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 311. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 249; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 281; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 312. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 250; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 282; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 313. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 251; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 283; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 314. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 252; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 284; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 315. 1 In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 253; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 285; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 316. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 254; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 286; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 317. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 255; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 287; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 318. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 256; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 288; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 319. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 257; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 289; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 320. In some embodiments, an anti-C3 antibody comprises
the LCDR1 of SEQ ID NO: 258; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 290; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 321. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 259; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 291; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 322. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 260; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 292; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 323. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 261; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 293; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 324. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 262; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 294; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 325. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 263; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 295; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 326. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 264; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 296; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 327. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 265; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 297; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 328. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 266; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 298; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 329. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 267; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 299; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 330. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 332; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 335; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 338. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 333; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 336; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 339. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 341; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 350; and the
LCDR3 comprises the amino acid sequence of SEQ ID NO: 359. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 342; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 351; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 360. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 343; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 352; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 361. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 344; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 353; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 362. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 345; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 354; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 363. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 346; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 355; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 364. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 347; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 356; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 365. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 348; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 357; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 366. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 368; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 403; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 438. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 369; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 404; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 439. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 370; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 405; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 440. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 371; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 406; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 441. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 372; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 407; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 442. In some embodiments, an anti-C3 antibody comprises
the LCDR1 of SEQ ID NO: 373; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 408; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 443. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 374; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 409; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 444. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 375; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 410; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 445. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 376; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 411; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 446. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 377; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 412; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 447. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 378; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 413; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 448. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 379; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 414; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 449. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 380; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 415; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 450. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 381; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 416; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 451. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 382; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 417; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 452. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 383; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 418; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 453. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 384; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 419; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 454. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 385; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 420; and the
LCDR3 comprises the amino acid sequence of SEQ ID NO: 455. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 386; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 421; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 456. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 387; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 422; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 457. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 388; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 423; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 458. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 389; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 424; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 459. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 390; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 425; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 460. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 391; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 426; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 461. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 392; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 427; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 462. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 393; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 428; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 463. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 394; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 429; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 464. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 395; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 430; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 465. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 396; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 431 ; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 466. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 397; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 432; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 467. In some embodiments, an anti-C3 antibody comprises
the LCDR1 of SEQ ID NO: 398; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 433; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 468. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 399; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 434; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 469. In some embodiments, an anti-C3 antibody comprises the LCDR1 of SEQ ID NO: 400; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 435; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 470. In some embodiments, an anti- 03 antibody comprises the LCDR1 of SEQ ID NO: 401; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 436; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 471.
[0076] In some embodiments, the VH of the anti-C3 antibodies of the present disclosure includes an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to any of SEQ ID NOs: 546-619. In some embodiments, the VL of the anti-C3 antibodies of the present disclosure includes an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to any of SEQ ID NOs: 694-767.
[0077] In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 546 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 694. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 547 and the and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 695. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to
SEQ ID NO: 548 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 696. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 549 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 697. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 550 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 698. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 551 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 699. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 552 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 700. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 553 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 701. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at
least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 554 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 702. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 555 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 703. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 556 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 704. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 557 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 705. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 558 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 706. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 559 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 707. In some embodiments, the VH comprises an
amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 560 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 708. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 561 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 709. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 562 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 710. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 563 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 711. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 564 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 712. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 565 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%,
at least 99%, or 100% identical) to SEQ ID NO: 713. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 566 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 714. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 567 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 715. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 568 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 716. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 569 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 717. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 570 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 718. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 571 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least
91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 719. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 572 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 720. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 573 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 721. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 574 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 722. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 575 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 723. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 576 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 724. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID
NO: 577 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 725. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 578 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 726. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 579 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 727. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 580 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 728. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 581 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 729. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 582 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 730. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%,
at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 583 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 731. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 584 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 732. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 585 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 733. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 586 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 734. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 587 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 735. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 588 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 736. In some embodiments, the VH comprises an amino acid
sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 589 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 737. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 590 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 738. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 591 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 739. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 592 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 740. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 593 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 741. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 594 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or
100% identical) to SEQ ID NO: 742. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 595 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 743. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 596 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 744. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 597 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 745. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 598 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 746. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 599 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 747. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 600 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%,
at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 748. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 601 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 749. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 602 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 750. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 603 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 751. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 604 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 752. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 605 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 753. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 606 and the VL
comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 754. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 607 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 755. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 608 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 756. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 609 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 757. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 610 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 758. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 611 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 759. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least
96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 612 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 760. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 613 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 761. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 614 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 762. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 615 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 763. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 616 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 764. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 617 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 765. In some embodiments, the VH comprises an amino acid sequence that is at least
90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 618 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 766. In some embodiments, the VH comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 619 and the VL comprises an amino acid sequence that is at least 90% identical (e.g., at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 767.
[0078] Nucleic acid or amino acid sequence “identity,” as described herein, can be determined by comparing a nucleic acid or amino acid sequence of interest to a reference nucleic acid or amino acid sequence. A number of mathematical algorithms for obtaining the optimal alignment and calculating identity between two or more sequences are known and incorporated into a number of available software programs. Examples of such programs include CLUSTAL-W, T-Coffee, and ALIGN (for alignment of nucleic acid and amino acid sequences), BLAST programs (e.g., BLAST 2.1, BL2SEQ, and later versions thereof) and FASTA programs (e.g., FASTA3x, FAS™, and SSEARCH) (for sequence alignment and sequence similarity searches). Sequence alignment algorithms also are disclosed in, for example, Altschul et al., J. Molecular Biol., 215(3): 403-410 (1990), Beigert et al., Proc. Natl. Acad. Sci. USA, 106( Q>): 3770-3775 (2009), Durbin et al., eds., Biological Sequence Analysis: Probabilistic Models of Proteins and Nucleic Acids, Cambridge University Press, Cambridge, UK (2009), Soding, Bioinformatics, 21(7): 951-960 (2005), Altschul et al., Nucleic Acids Res., 25(17): 3389-3402 (1997), and Gusfield, Algorithms on Strings, Trees and Sequences, Cambridge University Press, Cambridge UK (1997)).
[0079] As would be recognized by one of ordinary skill in the art based on the present disclosure, one or more amino acids of the aforementioned anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies), or antigen fragments thereof, can be replaced or substituted with a different amino acid. An amino acid “replacement” or “substitution” refers to the replacement of one amino acid at a given position or residue by another amino acid at the same position or residue within a polypeptide sequence. Amino acids are broadly grouped as “aromatic” or “aliphatic.” An aromatic
amino acid includes an aromatic ring. Examples of “aromatic” amino acids include histidine (H or His), phenylalanine (F or Phe), tyrosine (Y or Tyr), and tryptophan (W or Trp). Non-aromatic amino acids are broadly grouped as “aliphatic.” Examples of “aliphatic” amino acids include glycine (G or Gly), alanine (A or Ala), valine (V or Vai), leucine (L or Leu), isoleucine (I or He), methionine (M or Met), serine (S or Ser), threonine (T or Thr), cysteine (C or Cys), proline (P or Pro), glutamic acid (E or Glu), aspartic acid (A or Asp), asparagine (N or Asn), glutamine (Q or Gin), lysine (K or Lys), and arginine (R or Arg). Aliphatic amino acids may be sub-divided into four sub-groups. The “large aliphatic non-polar sub-group” consists of valine, leucine, and isoleucine. The “aliphatic slightly-polar sub-group” consists of methionine, serine, threonine, and cysteine. The “aliphatic polar/charged sub-group” consists of glutamic acid, aspartic acid, asparagine, glutamine, lysine, and arginine. The “small-residue sub-group” consists of glycine and alanine. The group of charged/polar amino acids may be sub-divided into three sub-groups: the “positively-charged sub-group” consisting of lysine and arginine, the “negatively-charged subgroup” consisting of glutamic acid and aspartic acid, and the “polar sub-group” consisting of asparagine and glutamine. Aromatic amino acids may be sub-divided into two sub-groups: the “nitrogen ring sub-group” consisting of histidine and tryptophan and the “phenyl sub-group” consisting of phenylalanine and tyrosine.
[0080] The amino acid replacement or substitution can be conservative, semi-conservative, or non-conservative. The phrase “conservative amino acid substitution” or “conservative mutation” refers to the replacement of one amino acid by another amino acid with a common property. A functional way to define common properties between individual amino acids is to analyze the normalized frequencies of amino acid changes between corresponding proteins of homologous organisms (Schulz and Schirmer, Principles of Protein Structure, Springer-Verlag, New York (1979)). According to such analyses, groups of amino acids may be defined where amino acids within a group exchange preferentially with each other, and therefore resemble each other most in their impact on the overall protein structure. Examples of conservative amino acid substitutions include substitutions of amino acids within the sub-groups described above, for example, lysine for arginine and vice versa such that a positive charge may be maintained, glutamic acid for aspartic acid and vice versa such that a negative charge may be maintained, serine for threonine such that a free -OH can be maintained, and glutamine for asparagine such that a free -NH2 can be
maintained. “Semi-conservative mutations” include amino acid substitutions of amino acids within the same groups listed above, but not within the same sub-group. For example, the substitution of aspartic acid for asparagine, or asparagine for lysine, involves amino acids within the same group, but different sub-groups. “Non-conservative mutations” involve amino acid substitutions between different groups, for example, lysine for tryptophan, or phenylalanine for serine, etc.
[0081] In addition, one or more amino acids can be inserted into the anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies), or antigen-binding fragments thereof (e.g., insertion into the heavy and/or light chain variable region amino acid sequence). Any number of suitable amino acids can be inserted into the amino acid sequence of the antibody or antigen-binding fragment thereof. In this respect, at least one amino acid (e.g., 2 or more, 5 or more, or 10 or more amino acids), but not more than 20 amino acids (e.g., 18 or less, 15 or less, or 12 or less amino acids), can be inserted into the amino acid sequence of the antibody or antigen-binding fragment thereof. For example, 1- 10 amino acids (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids) may be inserted into the amino acid sequence of the monoclonal antibody or antigen-binding fragment thereof. In this respect, the amino acid(s) can be inserted into an antibody or antigen-binding fragment thereof in any suitable location. Preferably, the amino acid(s) are inserted into a CDR (e.g., CDR1, CDR2, or CDR3) of the antibody or antigen-binding fragment thereof.
[0082] The amino acid sequences of the anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies), or antigen-binding fragments thereof, are not limited to the specific amino acid sequences described herein. Indeed, an anti-C3 antibody (e.g., C3, C3a, and C3b antibody), or antigenbinding fragment thereof, can comprise any heavy chain polypeptide or light chain polypeptide that competes with the anti-C3 antibodies or antigen-binding fragments thereof for conformational binding to C3. Antibody competition can be assayed using routine peptide competition assays such as, for example, ELISA, Western blot, or immunohistochemistry methods (see, e.g., U.S. Patents 4,828,981 and 8,568,992; and Braitbard et al., Proteome Sci., 4 12 (2006)).
[0083] An anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure may be a whole antibody, or an antigen-binding fragment of a whole antibody. As defined herein, antigen-binding antibody fragments encompassed by the present disclosure include, but are not limited to, F(ab’)2, Fab’, Fab, Fv, scFv, dsFv, dAb, and single chain binding polypeptides. Antibody fragments and their therapeutic utility are further described in, e.g., Nelson, A.L., MAbs.
2010 Jan-Feb; 2(1): 77-83; Joosten et al., Microbial Cell Factories volume 2, Article number: 1 (2003); and Bates A, Power CA., Antibodies (Basel). 2019;8(2):28; doi:10.3390/antib8020028). In some embodiments, the anti-C3 antigen-binding fragment is a single-chain variable fragment (scFv), which is an engineered antibody generated by the fusion of the heavy (VH) and light chains (VL) of immunoglobulins through a short polypeptide linker. Single chain variable domain (Fv) fragments (scFv) are used in the art in a variety of clinical and therapeutic applications, primarily due to their improved pharmacokinetic properties as compared to the parent monoclonal antibodies and the relative ease of producing them in large quantities at low cost (Monnier et al., Antibodies 2013, 2(2), 193-208; doi.org/10.3390/antib2020193; Safdari et al., Mol Med. 2016; 22: 258-270; and Lu, R., Hwang, Y., Liu, I. et al. Development of therapeutic antibodies for the treatment of diseases. J Biomed Sci 27, 1 (2020). https://doi.org/10.1186/sl2929-019-0592-z).
[0084] An anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure may be a diabody. Diabodies are antibody fragments with two antigen-binding sites that may be bivalent or bispecific. See, for example, EP 404,097; WO 1993/01161; Hudson et al., Nat. Med. 9:129-134 (2003); and Hollinger et al., Proc. Natl. Acad. Sci. USA 90: 6444-6448 (1993). Triabodies and tetrabodies are also described in Hudson et al., Nat. Med. 9:129-134 (2003). An anti-C3 antibody of the present disclosure may be a single-domain antibody (also referred to as a nanobody). Single-domain antibodies are antibody fragments comprising all or a portion of the heavy chain variable domain or all or a portion of the light chain variable domain of an antibody. In certain embodiments, a single-domain antibody is a human single-domain antibody (Domantis, Inc., Waltham, Mass.; see, e.g., U.S. Pat. No. 6,248,516 Bl). Antibody fragments can be made by various techniques, including but not limited to proteolytic digestion of an intact antibody as well as production by recombinant host cells (e.g., E. coli or phage), as described herein.
[0085] In other embodiments, the anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure is a whole antibody. As defined herein, a whole antibody comprises two identical copies of a heavy (H) chain polypeptide and two identical copies of a light (L) chain polypeptide. Each of the heavy chains contains one N-terminal variable (VH) region and three C- terminal constant (CHI, CH2, and Cm) regions, and each light chain contains one N-terminal variable (VL) region and one C-terminal constant (CL). The heavy chain C-terminal constant region contains the fragment crystallizable (Fc) domain, which determines antibody class and is
responsible for humoral and cellular effector functions. Antibodies are divided into five major classes (or “isotypes”), IgG, IgM, IgA, IgD and IgE, which differ in their function in the immune system. IgGs are the most abundant immunoglobulins in the blood, representing 60% of total serum antibodies in humans. IgG antibodies may be subclassified as IgGl, IgG2, IgG3, and IgG4, named in order of their abundance in serum (IgGl being the most abundant) (Vidarsson et al., Frontiers in Immunology. 5: 520 (2014)). A whole anti-C3 monoclonal antibody described herein may be of any suitable class and/or subclass. In some embodiments, the monoclonal antibody is of class IgG (e.g., IgGl, IgG2, IgG3, or IgG4). For example, the monoclonal antibody may be an IgGl antibody.
[0086] As discussed above, the Fc domain mediates several effector functions of antibodies, such as binding to receptors on target cells and complement fixation (triggering effector functions that eliminate the antigen). In some embodiments, the Fc domain may be modified or engineered to alter its effector functions. For example, Fc domains may be modified to improve antibodydependent cellular cytotoxicity (ADCC) and antibody-dependent cellular phagocytosis (ADCP), and to control serum half-life. In some embodiments, the Fc domain of the anti-C3 antibody may be engineered to modulate affinity for an Fc receptor, such as Fey receptors (FcyRs) and the neonatal Fc receptor (FcRn). Indeed, optimization of the interactions between antibodies and Fey Rs has emerged as a promising approach for enhancing the activity of therapeutic antibodies for the treatment of various diseases (Mimoto et al., Curr. Pharm. Biotechnol. 17, 1298-1314 (2016); Lazar et al., Proc. Natl Acad. Sci. USA 103, 4005-4010 (2006); Richards et al., Mol. Cancer Ther. 7, 2517-2527 (2008); Nordstrom et al., Breast Cancer Res. 13, R123 (2011); and Kang, T.H., Jung, S.T., Exp Mol Med 51, 1-9 (2019)). The Fc domain also may be modified to improve serum half-life, e.g., by engineering IgG Fc for higher FcRn binding (Zalevsky et al., Nat. Biotechnol. 28, 157-159 (2010); and Dall’Acqua et al., J. Immunol. 169, 5171-5180 (2002)). In other embodiments, the Fc domain may be modified to create monovalency or antibody bispecificity for improving therapeutic potency. For example, an Fc domain may be generated that does not form a homodimer but remains as a soluble monomer, mFc, that exhibits high affinity for FcyRI but no detectable binding to FcyRIIIa. In other embodiments, a heterodimeric Fc domain may be generated to obtain bispecific properties for antigen binding to circumvent homodimer formation. Engineered Fc domains may be generated by inducing point mutations or by modifying
glycosylation of the Fc domain (Saunders, K.O., Front Immunol. 2019; 10: 1296; Kelley, R.F., Meng, Y.G., Liu et al., J Biol Chem. 2014;289:3571-90; Monnet et al., MAbs. 2014;6:422-36; Li et al., Proc Natl Acad Sci U S A. 2017;114:3485-90; and Lin et al., Proc Natl Acad Sci U S A. 2015;112:10611-6; Kang and Jung, supra).
[0087] Embodiments of the present disclosure include compositions and methods pertaining to an antibody, or antigen binding fragment thereof, which specifically binds Complement Component 3 (C3) polypeptides, optionally wherein said C3 is a Complement Component 3a (C3a) polypeptide or a Complement Component 3b (C3b) polypeptide, which is optionally a polypeptide comprising or consisting of the amino acid sequence of SEQ ID NO: 1064. In accordance with these embodiments, the present disclosure provides an antibody or a fragment thereof, which exhibits any one or more the following functional characteristics: (a) reduces complement activation and/or attenuates complement component 3 (C3) convertase activity, which is optionally measured in an ELISA; and/or (b) cross-reacts with at least one of human C3b protein, Cynomolgus monkey C3 protein, and/or human C3a protein; and/or (c) binds to human C3 with a KD-1 of about lOOnM or lower; and/or (d) binds to the same epitope on human C3 as an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) competes for binding to human C3 with an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6. In some embodiments, the antibody or fragment thereof, which is a monoclonal antibody. In some embodiments, the antibody or fragment thereof, which is a human, humanized or chimeric antibody. In further embodiments, the antibody or fragment thereof, is a whole antibody, a single chain variable fragment (scFv), a variable fragment (Fv), a fragment antigen-binding region (Fab), a Fab-C, a Fab’-SH, a (Fab’)2, a single-domain antibody (sdAb), a nanobody, a diabody, a triabody, or an aptamer, optionally wherein the antibody is a whole antibody comprising an Fc region such as a human IgGl , IgG2, IgG3 or IgG4 Fc region. In some embodiments, the antibody or fragment thereof, is conjugated to at least one additional moiety, optionally selected from: (a) a therapeutic or cytotoxic moiety; (b) a detection moiety; (c) a purification moiety; (d) a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody. In some embodiments, this is
referred to as “XTENylation,” as described further in US8933197, US7846445, US7855279, US8492530, US9938331, US8673860, US9371369, US9926351, US10961287, US10172953, and US 10953073. In accordance with these embodiments, the present disclosure provides an antibody or a fragment thereof, which is a polypeptide comprising: (a) one, two or all three HCDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also one, two or all three of the corresponding LCDRs of said exemplary antibody; and/or (b) a VH sequence having at least 90% identity to the VH sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also a VL sequence having at least 90% identity to the corresponding VL sequence of said exemplary antibody, preferably wherein variation is not permitted in the HCDRs or LCDRs; and/or (c) all six CDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (d) the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or (e) the full length heavy chain (VH + constant) sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally the corresponding full length light chain (VL + constant) sequence of said exemplary antibody.
[0088] Embodiments of the present disclosure include compositions and methods pertaining to a polynucleotide encoding an antibody or fragment according to any one of the preceding claims, optionally wherein said polynucleotide comprises or consists of a nucleic acid sequence having at least 70, 80, 90 or 100% identity to a nucleic acid sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6. In some embodiments, an expression vector comprises a polynucleotide, which is optionally an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, or a retrovirus vector.
[0089] Embodiments of the present disclosure include a pharmaceutical composition comprising an antibody or fragment, a polynucleotide, or a vector and optionally (a) at least one pharmaceutically acceptable carrier, a buffer, excipient, stabilizer, diluent, or preservative; and/or (b) at least one additional active ingredient. In some embodiments, the pharmaceutical composition, which is suitable for ocular administration to a subject, optionally by delivery using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant, preferably by injection into the vitreous fluid. In some embodiments, the antibody or fragment, the polynucleotide, the vector, the
composition, for use as a medicament, optionally for use in a method of treating a disease of the eye in a subject. In further embodiments, the antibody or fragment, the polynucleotide, the vector, the composition for use, wherein said disease is characterized by increased activation of the complement system, in particular the alternative pathway, and in particular within the eye of the subject, for example in drusen or retinal pigment epithelium (RPE) cells of the subject. In some embodiments, the antibody or fragment, the polynucleotide, the vector, the composition for use, wherein the method comprises ocular administration of the antibody, preferably by injection into the vitreous fluid, and wherein said administration preferably relieves at least one symptom in the subject selected from visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light. In accordance with these embodiments, where the disease is age-related macular degeneration (AMD), optionally wherein said AMD is dry AMD, which may be at an early, intermediate, or advanced stage (the latter otherwise known as geographic atrophy, GA).
Multi-specific Anti-C3 Antibodies
[0090] As described above, the anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies) of the present disclosure can be a monoclonal antibody, a human antibody, a humanized antibody, and/or a chimeric antibody. In some embodiments, the antibody is a fragment selected from the group consisting of Fab, Fab-C, Fab'-SH, Fv, scFv, and (Fab')2 fragments. In some embodiments, the anti-C3 antibody is a monospecific antibody. In some embodiments, the anti-C3 antibody is a bispecific antibody. In some embodiments, the anti-C3 antibody comprises two or more singledomain antibodies that form a bivalent antibody, a trivalent antibody, or a tetravalent antibody that recognizes different epitopes on the same or different antigens.
[0091] In some embodiments, an anti-C3 antibody provided herein is a chimeric antibody. Certain chimeric antibodies are described, for example, in U.S. Pat. No. 4,816,567; and Morrison et al., Proc. Natl. Acad. Sci. USA. 81:6851-6855 (1984). In one example, a chimeric antibody comprises a non-human variable region (e.g., a variable domain derived from a mouse, rat, hamster, rabbit, or non-human primate, such as a monkey) and a human constant domain. In a further example, a chimeric antibody is a “class switched” antibody in which the class or subclass has been changed from that of the parent antibody. Chimeric antibodies include antigen-binding fragments thereof.
[0092] In certain embodiments, a chimeric antibody is a humanized antibody. Typically, a non-human antibody is humanized to reduce immunogenicity to humans, while retaining the specificity and affinity of the parental non-human antibody. Generally, a humanized antibody comprises one or more variable domains in which HVRs, for example, CDRs, (or portions thereof) are derived from a non-human antibody, and FRs (or portions thereof) are derived from human antibody sequences. A humanized antibody optionally will also comprise at least a portion of a human constant region. In some embodiments, some FR residues in a humanized antibody are substituted with corresponding residues from a non-human antibody (e.g., the antibody from which the HVR residues are derived), e.g., to restore or improve antibody specificity or affinity.
[0093] Humanized antibodies and methods of making them are reviewed, for example, in Almagro and Fransson, Front. Biosci. 13:1619-1633 (2008), and are further described, for example, in Riechmann et al., Nature 332:323-329 (1988); Queen et al., Proc. Nat'l Acad. Set. USA 86:10029-10033 (1989); U.S. Pat. Nos. 5,821,337, 7,527,791, 6,982,321, and 7,087,409; Kashmiri et al., Methods 36:25-34 (2005) (describing specificity determining region (SDR) grafting); Padlan, Mol. Immunol. 28:489-498 (1991) (describing “resurfacing”); DalfAcqua et al., Methods 36:43-60 (2005) (describing “FR shuffling”); and Osbourn et al., Methods 36:61-68 (2005) and Klimka et al., Br. J. Cancer, 83:252-260 (2000) (describing the “guided selection” approach to FR shuffling).
[0094] In accordance with the above embodiments, the anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies) of the present disclosure can be made into bivalent, trivalent, or tetravalent formats. For example, an anti-C3 antibody of the present disclosure can be a bivalent, bispecific antibody with heteromeric heavy chains (e.g., Triomab, knobs-into-holes (KIH), Duobody, etc). An anti-C3 antibody of the present disclosure can be a tetravalent multispecific antibody comprised of IgGs with other binding domains fused to either the N- or C-termini of either the heavy or light chains (e.g., dual variable domain [DVD], IgG-scFv fusion, Mabtyrin (IgG with non-antibody binding scaffold “centyrin” fused to C-terminal end of heavy chains). An anti-C3 antibody of the present disclosure can be comprised of IgGs to which additional antigen combining sites have been added within the structure (e.g., two-in-one antibodies, MAT “Modular Antibody Technology” platform from F-Star). An anti-C3 antibody of the present disclosure can be an engineered antibody fragment linked by short peptide linkers which can be made into bivalent,
trivalent, or tetravalent formats addressing two to three targets (e.g., bispecific T-cell engager (BiTE), Nanobody platform, dual- affinity re -targeting (DART) antibodies, “tandem antibody” structures (TandAbs)). And an anti-C3 antibody of the present disclosure can be comprised of chemically coupled IgGs.
[0095] In some embodiments, an anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure is a multispecific antibody, such as a bispecific antibody, which have binding specificities for at least two different antigens. In some embodiments, the anti-C3 antibodies of the present disclosure, or antigen-binding fragments thereof, can be used to form one arm (e.g., antigen-binding portion) of a bispecific antibody, whereas the other arm of the bispecific antibody can be specific for a different antigen. In some embodiments, the other antigen includes, but is not limited to, interleukin- 1 beta (IL-1J3), interleukin-6 (IL-6); interleukin-6 receptor (IL-6R); interleukin- 13 (IL-13); IL-13 receptor (IL-13R); PDGF (e.g., PDGF-BB); angiopoietin; angiopoietin 2 (Ang2); Tie2; SIP; integrins av03, av05, and a501; betacellulin; apelin/APJ; erythropoietin; complement factor D; TNFa; HtrAl; a VEGF receptor (e.g., VEGFR1, VEGFR2, VEGFR3, membrane-bound VEGF -receptor (mbVEGFR), or soluble VEGF receptor (sVEGFR)); ST-2 receptor; and proteins genetically linked to age-related macular degeneration (AMD) risk, such as complement pathway components C2, factor B, factor H, CFHR3, CFHR4, C5, C5a, HtrAl, ARMS2, TIMP3, HLA, interleukin-8 (IL-8), CX3CR1, TLR3, TLR4, CETP, LIPC, COL10A1, and TNFRSF10A. In other embodiments, the bispecific antibody may have binding specificity for C3/C3a/C3b and IL-1|3; C3/C3a/C3b and IL-6; C3/C3a/C3b and IL-6R; C3/C3a/C3b and IL-13; C3/C3a/C3b and IL-13R; C3/C3a/C3b and PDGF (e.g., PDGF-BB); C3/C3a/C3b and angiopoietin; C3/C3a/C3b and Ang2; C3/C3a/C3b and Tie2; C3/C3a/C3b and SIP; C3/C3a/C3b and integrin av[:13; C3/C3a/C3b and integrin av[:15; C3/C3a/C3b and integrin a501; C3/C3a/C3b and betacellulin; C3/C3a/C3b and apelin/APJ; C3/C3a/C3b and erythropoietin; C3/C3a/C3b and complement factor D; C3/C3a/C3b and TNFa; C3/C3a/C3b and HtrAl; C3/C3a/C3b and a VEGF receptor (e.g., VEGFR1 , VEGFR2, VEGFR3, mbVEGFR, or sVEGFR); C3/C3a/C3b and ST-2 receptor; C3/C3a/C3b and C2; C3/C3a/C3b and factor B; C3/C3a/C3b and factor H; C3/C3a/C3b and CFHR3; C3/C3a/C3b and CFHR4; C3/C3a/C3b and C5; C3/C3a/C3b and C5a; C3/C3a/C3b and ARMS2; C3/C3a/C3b and TIMP3; C3/C3a/C3b and HLA; C3/C3a/C3b and IL-8; C3/C3a/C3b and CX3CR1; C3/C3a/C3b and TLR3; C3/C3a/C3b and TLR4;
C3/C3a/C3b and CETP; C3/C3a/C3b and LIPC; C3/C3a/C3b and COL10A1; or C3/C3a/C3b and TNFRSF10A.
[0096] In some embodiments, a bispecific antibody of the present disclosure includes an anti- C3 antibody (e.g., C3, C3a, and C3b antibody), or an antigen-binding fragment thereof, and an anti-VEGF antibody, or an antigen-binding fragment thereof. Such bispecific antibodies can be used to target the complement activation pathway from two different mechanisms, and thus provide additional therapeutic benefits. For example, the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure, and the anti-VEGF arm can be any VEGF antagonist, including but not limited to, anti-VEGF antibodies (e.g., bevacizumab, sevacizumab, and ranibizumab), anti-VEGFR2 antibodies and related molecules (e.g., ramucirumab, tanibirumab, aflibercept), anti-VEGFRl antibodies and related molecules (e.g., icrucumab, aflibercept (VEGF Trap-Eye; EYLEA®), and ziv-aflibercept (VEGF Trap; ZALTRAP®)), anti-VEGF arms of VEGF bispecific antibodies (e.g., MP-0250, vanucizumab (VEGF-ANG2)), including anti-VEGF, anti- VEGFRl, and anti-VEGFR2 arms.
[0097] In other embodiments, a bispecific antibody of the present disclosure includes an anti- C3 antibody (e.g., C3, C3a, and C3b antibody), or an antigen-binding fragment thereof, and an anti-CFHR4 antibody, or an antigen-binding fragment thereof. Such bispecific antibodies can be used to target the complement activation pathway from two different mechanisms, and thus provide additional therapeutic benefits. For example, the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure (e.g., C3, C3a, and C3b antibodies), and the anti-CFHR4 arm can be any CFHR4 antagonist.
[0098] In other embodiments, a bispecific antibody of the present disclosure includes an anti- C3 antibody (e.g., C3, C3a, and C3b antibody), or an antigen-binding fragment thereof, and an anti-C5 antibody, or an antigen-binding fragment thereof. For example, the anti-C3 arm can be any of the anti-C3 antibodies of the present disclosure, and the anti-C5 arm can be any C5 antagonist, including but not limited to, eculizumab and tesidolumab (LFG316). In other embodiments, a bispecific antibody of the present disclosure includes an anti-C3 antibody, or an antigen-binding fragment thereof, and an anti-FD antibody, or an antigen-binding fragment thereof. For example, the anti-C3 arm can be any of the anti-C3 antibodies of the present
disclosure, and the anti-C5 arm can be any C5 antagonist, including but not limited to, lampalizumab.
Functional Characteristics of Anti-C3 Antibodies
[0099] In accordance with the above embodiments, the present disclosure provides anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies) comprising various functional characteristics. In some embodiments, the anti-C3 antibodies described herein bind an antigen on C3 (SEQ ID NO: 1064), or a variant or isoform thereof, via interaction with its antigenic determinants (epitopes). In some embodiments, binding of an anti-C3 antibody to C3, C3a, and/or C3b reduces complement activation. In some embodiments, the anti-C3 antibody binds human C3, C3a, and/or C3b with a KD of about 100 nM or lower (FIG. 3). As further described herein, the anti-C3 antibodies of the present disclosure were evaluated for cross-reactivity with various other components of the complement activation pathway. For example, FIG. 4 includes representative data of C3 antibody cross-reactivity with human C3b protein, Cynomolgus monkey C3 protein, and human C3a protein. FIGS. 5A-5B include representative data of binding kinetics of C3 antibodies with respect to human C3 protein (FIG. 5A) and human C3b protein (FIG. 5B). Additionally, FIG. 8 includes representative results of C3 antibody polyreactivity with baculovirus particles (BVPs); FIG. 9 includes representative results of C3 antibody polyreactivity with the various proteins indicated; and FIGS. 10A-10B include representative results of C3 antibody polyreactivity with CFHR4 (FIG. 10A) and CFH (FIG. 10B).
Polypeptides and Expression Vectors
[00100] Embodiments of the present disclosure also include a polynucleotide encoding any of the anti-C3 antibodies of the present disclosure. In some embodiments, the polynucleotide comprises a sequence that is at least 70% identical (e.g., at least 70%, 75%, 80%, 85%, 90%, 95% or 100%) to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 70% identical (e.g., at least 70%, 75%, 80%, 85%, 90%, 95% or 100%) to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ
ID NOs: 768-841, and/or SEQ ID NOs: 916-989. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693. Embodiments of the present disclosure also include a polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
[00101] In accordance with these embodiments, the present disclosure includes an expression vector comprising any of the polynucleotides encoding an anti-C3 antibody of the present disclosure. In some embodiments, the expression vector is suitable for manufacturing an anti-C3 antibody of the present disclosure for delivery of the antibody to a subject. In certain embodiments, the nucleic acid sequence is in the form of a vector. The vector can be, for example, a plasmid, episome, cosmid, viral vector (e.g., retroviral or adenoviral), or phage. Suitable vectors and methods of vector preparation are well known in the art (see, e.g., Sambrook et al., Molecular Cloning, a Laboratory Manual, 4th edition, Cold Spring Harbor Press, Cold Spring Harbor, N.Y. (2012), and Ausubel et al., Current Protocols in Molecular Biology, Greene Publishing Associates and John Wiley & Sons, New York, N.Y. (1994)).
[00102] In addition to the nucleic acid encoding an anti-C3 antibody (e.g., C3, C3a, and C3b antibody), or antigen-binding fragment thereof, the vector desirably comprises expression control sequences, such as promoters, enhancers, polyadenylation signals, transcription terminators, internal ribosome entry sites (IRES), and the like, that provide for the expression of the antibodyencoding nucleic sequence in a host cell. Exemplary expression control sequences are known in the art and described in, for example, Goeddel, Gene Expression Technology: Methods in Enzymology, Vol. 185, Academic Press, San Diego, Calif. (1990).
[00103] A vector comprising a nucleic acid sequence encoding an anti-C3 antibody (e.g., C3, C3a, and C3b antibody), or antigen-binding fragment thereof, may be introduced into a host cell that is capable of expressing the polypeptides encoded thereby, including any suitable prokaryotic or eukaryotic cell. Examples of suitable prokaryotic cells include, but are not limited to, cells from the genera Bacillus (such as Bacillus subtilis and Bacillus brevis), Escherichia (such as E. coll). Pseudomonas, Streptomyces, Salmonella, and Erwinia. Particularly useful prokaryotic cells include the various strains of Escherichia coli (e.g., K12, HB101 (ATCC No. 33694), DH5a, DH10, MC1061 (ATCC No. 53338), and CC102). Suitable eukaryotic cells are known in the art
and include, for example, yeast cells, insect cells, and mammalian cells. Examples of suitable yeast cells include those from the genera Hansenula, Kluyveromyces, Pichia, Rhinosporidium, Saccharomyces, and Schizosaccharomyces . Suitable insect cells include Sf-9 and HIS cells (Invitrogen, Carlsbad, Calif.) and are described in, for example, Kitts et al., Biotechniques, 14: 810-817 (1993); Lucklow, Curr. Opin. Biotechnol., 4: 564-572 (1993); and Lucklow et al., J. Virol., 67'. 4566-4579 (1993). Examples of suitable mammalian cells include, but are not limited to, Chinese hamster ovary cells (CHO) (ATCC No. CCL61), CHO DHFR-cells (Urlaub et al., Proc. Natl. Acad. Sci. USA, 97: 4216-4220 (1980)), human embryonic kidney (HEK) 293 or 293T cells (ATCC No. CRL1573), and 3T3 cells (ATCC No. CCL92). Other suitable mammalian cell lines are the monkey COS-1 (ATCC No. CRL1650) and COS-7 cell lines (ATCC No. CRL1651), as well as the CV-1 cell line (ATCC No. CCL70). Further exemplary mammalian host cells include primate cell lines and rodent cell lines, including transformed cell lines. Normal diploid cells, cell strains derived from in vitro culture of primary tissue, as well as primary explants also are suitable. Other suitable mammalian cell lines include, but are not limited to, mouse neuroblastoma N2A cells, HeLa, mouse L-929 cells, and BHK or HaK hamster cell lines, all of which are available from the ATCC. Methods for selecting suitable mammalian host cells and methods for transformation, culture, amplification, screening, and purification of such cells are well known in the art (see, e.g., Ausubel et al., eds., Short Protocols in Molecular Biology, 5th ed., John Wiley & Sons, Inc., Hoboken, N.J. (2002)). Preferably, the mammalian cell is a human cell.
[00104] In some embodiments, the vector can include means for attaching a detection moiety to an anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure. In some embodiments, the vector can include means for attaching a purification moiety to an anti-C3 antibody of the present disclosure. Exemplary detection and/or purification moieties/tags that can be coupled to an anti-C3 antibody of the present disclosure includes, but is not limited to, hemagglutinin (HA), c-Myc, V5, DYKDDDDK, His tag (e.g., 6x-HIS), Glutathione S-Transferase (GST), Maltose Binding Protein (MBP), a fluorophore (e.g., Green Fluorescent Protein (GFP), Red Fluorescent Protein (RFP), mCherry, a chromophore, and/or a luminescent peptide (e.g., luciferase).
[00105] In some embodiments, the expression vector is suitable for use in gene therapy (e.g., an expression vector for delivering a polynucleotide encoding an anti-C3 antibody of the present
disclosure to a subject). In some embodiments, the expression vector is a herpes simplex virus (HSV) vector, or a retrovirus vector. In some embodiments, the expression vector is an adeno- associated virus (AAV) vector, or comprises an AAV backbone. For example, AAV vectors have been designed, produced and used to mediate gene delivery in human subjects, including for therapeutic purposes. Typically, AAV vectors for use in gene transfer comprise a replication defective AAV genome lacking functional Rep and Cap coding viral sequences. In some embodiments, such replication defective AAV vectors lack most or all of the Rep and Cap coding sequences, and essentially retain one or two AAV ITR sequences and a packaging sequence. The defective genome is packaged in a viral particle, to form a defective, recombined AAV virus, also termed “AAV vector”. Methods of producing such AAV vectors have been disclosed in the literature, including using packaging cells, auxiliary viruses or plasmids, and/or baculovirus systems (Samulski et al., (1989) J. Virology 63, 3822; Xiao et al., (1998) J. Virology 72, 2224; Inoue et al., (1998) J. Virol. 72, 7024; WO98/22607; W02005/072364). Methods of producing pseudotyped AAV vectors have also been reported (e.g., WO00/28004), as well as various modifications or formulations of AAV vectors, to reduce their immunogenicity upon in vivo administration (see e.g., W001/23001; WOOO/73316; WO04/112727; W005/005610; WO99/06562). AAV vectors may be prepared or derived from various serotypes of AAVs, which may be even mixed together or with other types of viruses to produce chimeric (e.g., pseudotyped) AAV viruses. Examples of tAAVs are human AAV4 vectors, human AAV7 vectors, human AAV9 vectors, human AAV10 vectors, or bovine AAV vectors. The AAV vector may be derived from a single AAV serotype or comprise sequences or components originating from at least two distinct AAV serotypes (pseudotyped AAV vector), e.g., an AAV vector comprising an AAV genome derived from one AAV serotype (for example AAV9), and a capsid derived at least in part from a distinct AAV serotype. An AAV vector, as used herein, is a vector which comprises at least one component part derivable from an adeno-associated virus. Preferably, that component part is involved in the biological mechanisms by which the vector infects or transduces target cells and expresses an anti-C3 antibody of the present disclosure (e.g., ocular delivery/expression).
[00106] In other embodiments, the expression vector is a lentiviral vector (LV), or comprises an LV backbone. Lentiviruses are part of a larger group of retroviruses. A detailed list of lentiviruses may be found in Coffin et al (1997) “Retroviruses” Cold Spring Harbour Laboratory
Press Eds: JM Coffin, SM Hughes, HE Varmus pp 758-763). For example, lentiviruses can be divided into primate and non-primate groups. Examples of primate lentiviruses include but are not limited to: the human immunodeficiency virus (HIV), the causative agent of human auto immunodeficiency syndrome (AIDS), and the simian immunodeficiency virus (SIV). The non- primate lentiviral group includes the prototype “slow virus” visna/maedi virus (VMV), as well as the related caprine arthritis-encephalitis virus (CAEV), equine infectious anaemia virus (EIAV), feline immunodeficiency virus (FIV), Maedi visna virus (MW) and bovine immunodeficiency virus (BIV). In one embodiment, the lentiviral vector is derived from HIV- 1, HIV-2, SIV, FIV, BIV, EIAV, CAEV or Visna lentivirus. The lentivirus family differs from retroviruses in that lentiviruses have the capability to infect both dividing and non-dividing cells (Lewis et al (1992) EM BO J 11 (8): 3053-3058 and Lewis and Emerman (1994) J Virol 68 (1):510-516). In contrast, other retroviruses, such as MLV, are unable to infect non-dividing or slowly dividing cells such as those that make up, for example, muscle, brain, lung and liver tissue. A lentiviral vector, as used herein, is a vector which comprises at least one component part derivable from a lentivirus. Preferably, that component part is involved in the biological mechanisms by which the vector infects or transduces target cells and expresses an anti-C3 antibody of the present disclosure (e.g., ocular delivery/expression).
[00107] Additional compositions and method for ocular gene therapy can be found in, e.g., Bordet, T., and Behar-Cohen, F., “Ocular gene therapies in clinical practice: viral vectors and nonviral alternatives,” Drug Discovery Today, Volume 24, Issue 8, August 2019, Pages 1685- 1693). In some embodiments, gene therapy platforms, methods, and compositions that can be used to deliver an anti-C3 antibody of the present disclosure to a subject (e.g., ocular delivery) includes the platforms, methods, and compositions disclosed in US20220025396, US20220011308, US20210371877, US20210363192, US20190078099, US20190038724, and US10494646B2, which are incorporated herein by reference. In other embodiments, gene therapy platforms, methods, and compositions that can be used to deliver an anti-C3 antibody of the present disclosure to a subject (e.g., ocular delivery) includes the platforms, methods, and compositions based on HMR59 (Hemera Biosciences), which through its protein product soluble CD59, blocks the membrane attack complex that forms during the terminal step in the complement cascade. HMR59 is designed to be administered as a single intraocular injection.
[00108] In accordance with these embodiments, the present disclosure also provides a method of administering ocular gene therapy to a subject in need thereof comprising injecting a pharmaceutical composition comprising an effective amount of an expression vector described herein (e.g., an expression vector comprising a polynucleotides encoding an anti-C3 antibody of the present disclosure). As described further below, the present disclosure also provides a method of treating AMD and/or GA comprising administering a pharmaceutical composition comprising an effective amount of an expression vector described herein (e.g., an expression vector comprising a polynucleotides encoding an anti-C3 antibody of the present disclosure). In some embodiments, administering the pharmaceutical composition treats at least one AMD symptom and/or GA.
Compositions and Methods of Treatment
[00109] The anti-C3 antibodies (e.g., C3, C3a, and C3b antibodies) of the present disclosure can be administered as part of a pharmaceutical composition in a therapeutically effective amount to treat an eye disease (e.g., AMD or GA). In some embodiments, the composition is suitable for ocular administration. In some embodiments, ocular administration comprises injection into vitreous fluid. In some embodiments, ocular administration comprises delivering the antibody using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant (e.g., Susvimo™). Recent methods and formulations for ocular administration can be found in, e.g., Souto, E.B., et al. “Advanced Formulation Approaches for Ocular Drug Delivery: State-Of-The-Art and Recent Patents,” Pharmaceutics, 2019 Sep; 11(9): 460).
[00110] In accordance with these embodiments, the methods include administering a pharmaceutical composition comprising a therapeutically effective amount of an anti-C3 antibody of the present disclosure. In some embodiments, the pharmaceutical composition is administered ocularly and treats at least one AMD symptom. In some embodiments, AMD comprises wet AMD. In some embodiments, AMD comprises dry AMD (e.g., GA). In some embodiments, the at least one AMD symptom comprises visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light. In some embodiments, administering the pharmaceutical composition reduces complement activation in the subject’s eye.
[00111] In some embodiments, the pharmaceutical composition comprising a therapeutically effective amount of an anti-C3 antibody (e.g., C3, C3a, and C3b antibody) of the present disclosure is administered at a dose ranging from about 0.0001 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 1.0 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 10 mg/dose to about 100 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 10 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 1.0 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 0.1 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/dose to about 0.001 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/dose to about 10 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/dose to about 1.0 mg/dose. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/dose to about 10 mg/dose.
[00112] In some embodiments, the pharmaceutical composition comprising a therapeutically effective amount of an anti-C3 antibody of the present disclosure is administered at a dose ranging from about 0.0001 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 1.0 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 10 mg/ml to about 100 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 10 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose
ranging from about 0.0001 mg/ml to about 1.0 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 0.1 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 0.01 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.0001 mg/ml to about 0.001 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.01 mg/ml to about 10 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.001 mg/ml to about 1.0 mg/ml. In some embodiments, the anti-C3 antibody is administered at a dose ranging from about 0.1 mg/ml to about 10 mg/ml.
[00113] As used herein, the terms “treatment,” “treating,” and the like refer to obtaining a desired pharmacologic and/or physiologic effect. In some embodiments, the effect is therapeutic (e.g., the effect partially or completely cures a disease and/or adverse symptom attributable to the disease). To this end, the methods of the present disclosure comprise administering a “therapeutically effective amount” of an anti-C3 antibody (e.g., C3, C3a, and C3b antibody), or composition comprising an anti-C3 antibody. A “therapeutically effective amount” refers to an amount effective, at dosages and for periods of time necessary, to achieve a desired therapeutic result. The therapeutically effective amount may vary according to factors such as the disease state, age, sex, and weight of the individual, and the ability of the monoclonal antibody to elicit a desired response in the individual. For example, a therapeutically effective amount of an anti-C3 antibody of the present disclosure is an amount which treats at least one AMD and/or GA symptom in a subject. In some embodiments, the pharmacologic and/or physiologic effect may be prophylactic (e.g., the effect completely or partially prevents a disease or symptom thereof). In this respect, the methods of the present disclosure comprise administering a “prophylactically effective amount” of an anti-C3 antibody or composition comprising an anti-C3 antibody. A “prophylactically effective amount” refers to an amount effective, at dosages and for periods of time necessary, to achieve a desired prophylactic result (e.g., prevention of AMD and/or GA onset).
[00114] A typical dose of a therapeutically effective amount of an anti-C3 antibody of the present disclosure can range from, for example, about 0.0001 mg/dose to about 100 mg/dose for each eye to be treated. In some embodiments, a therapeutically effective amount of an anti-C3 antibody of the present disclosure can range from about 0.001 mg/dose to about 100 mg/dose, from
about 0.01 mg/dose to about 100 mg/dose, from about 0.05 mg/dose to about 50 mg/dose, from about 0.1 mg/dose to about 10 mg/dose, from about 0.5 mg/dose to about 5 mg/dose, and from about 1 mg/dose to about 10 mg/dose. In some embodiments, a therapeutically effective concentration of an anti-C3 antibody of the present disclosure can range from, for example, about 0.0001 mg to about 100 mg of the antibody per milliliter of solution. In some embodiments, a therapeutically effective concentration of an anti-C3 antibody of the present disclosure can range from about 0.001 mg/ml to about 100 mg/ml, from about 0.01 mg/ml to about 100 mg/ml, from about 0.1 mg/ml to about 100 mg/ml, from about 1.0 mg/ml and about 100 mg/ml, from about 0.001 mg/ml and about 50 mg/ml, from about 0.01 mg/ml and about 50 mg/ml, from about 0.1 mg/ml and about 50 mg/ml, from about 0.1 mg/ml and about 25 mg/ml, from about 0.1 mg/ml and about 10 mg/ml, and from about 1.0 mg/ml and about 10 mg/ml. In some embodiments, a therapeutically effective dose of an anti-C3 antibody of the present disclosure can be, exactly or approximately, 0.1 mg, 0.2 mg, 0.25 mg, 0.3 mg, 0.35 mg, 0.4 mg, 0.45 mg, 0.5 mg, 0.55 mg, 0.6 mg, 0.65 mg, 0.7 mg, 0.75 mg, 0.8 mg, 0.85 mg, 0.9 mg, 0.95 mg, 1.0 mg, 2.0 mg, 3.0 mg, 4.0 mg, 5.0 mg, 10.0 mg, 15.0 mg, 20.0 mg, or 25.0 mg, or can fall within a range delimited by any two of the foregoing values. For example, in certain embodiments, a sustained release formulation, (e.g., an ocular implant) can be, exactly or approximately, 0.1 mg, 0.2 mg, 0.25 mg, 0.3 mg, 0.35 mg, 0.4 mg, 0.45 mg, 0.5 mg, 0.55 mg, 0.6 mg, 0.65 mg, 0.7 mg, 0.75 mg, 0.8 mg, 0.85 mg, 0.9 mg, 0.95 mg, 1.0 mg, 2.0 mg, 3.0 mg, 4.0 mg, 5.0 mg, 10.0 mg, 15.0 mg, 20.0 mg, or 25.0 mg of an anti-C3 antibody, or an amount that falls within a range delimited by any two of the foregoing values.
[00115] Therapeutic or prophylactic efficacy can be monitored by periodic assessment of treated patients. For repeated administrations over several days or longer, depending on the condition, the treatment is repeated until a desired suppression of disease symptoms occurs. However, other dosage regimens may be useful and are within the scope of the present disclosure. The desired dosage can be delivered by a single bolus administration of the composition, by multiple bolus administrations of the composition, or by continuous infusion administration of the composition. The composition comprising an anti-C3 antibody, or antigen-binding fragment thereof, can be administered to a mammal using standard administration techniques, including ocular, oral, intravenous, intraperitoneal, subcutaneous, pulmonary, transdermal, intramuscular,
intranasal, buccal, sublingual, or suppository administration. The composition preferably is suitable for ocular administration.
[00116] In accordance with the compositions and methods of treatment described herein, embodiments of the present disclosure include anti-C3 antibodies that have enhanced half-life (e.g., after ocular administration), such that the anti-C3 antibody can be administered less often to a subject. In some embodiments, the antibody comprises a half-life extension moiety. In some embodiments, the half-life extension moiety comprises a polypeptide that can be coupled to an anti-C3 antibody of the present disclosure by any means known in the art (e.g., generation of a fusion protein). In some embodiments, the polypeptide that can be coupled to an anti-C3 antibody of the present disclosure is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residues. In some embodiments, the half-life extension polypeptide is attached to the C-terminus or N-terminus of the antibody. In some embodiments, this is referred to as “XTENylation,” as described further in US8933197, US7846445, US7855279, US8492530, US9938331, US8673860, US9371369, US9926351, US10961287, US10172953, and US10953073.
[00117] The present disclosure also provides a composition comprising any of the anti-C3 antibodies or antigen-binding fragments thereof described herein. The composition desirably is a pharmaceutically acceptable (e.g., physiologically acceptable) composition, which comprises a carrier, preferably a pharmaceutically acceptable (e.g., physiologically acceptable) carrier, and the anti-C3 antibody or antigen-binding fragment thereof. Any suitable carrier can be used within the context of the present disclosure, and such carriers are well known in the art. For example, the composition may contain preservatives, such as, for example, methylparaben, propylparaben, sodium benzoate, and benzalkonium chloride. A mixture of two or more preservatives optionally may be used. In addition, buffering agents may be included in the composition. Suitable buffering agents include, for example, citric acid, sodium citrate, phosphoric acid, potassium phosphate, and various other acids and salts. A mixture of two or more buffering agents optionally may be used. Methods for preparing compositions for pharmaceutical use are known to those skilled in the art and are described in, for example, Remington: The Science and Practice of Pharmacy, Lippincott Williams & Wilkins; 21st ed. (May 1, 2005).
[00118] Once administered to a mammal (e.g., a human), the biological activity of the anti-C3 antibody, or antigen-binding fragment thereof, can be measured by any suitable method known in the art. For example, the biological activity can be assessed by determining the stability of the anti- 03 antibody. The biological activity of the anti-C3 antibody also can be assessed by determining its binding affinity to C3, C3a, and/or C3b peptides and/or by assessing its binding affinity to peptides with which it may cross-react. The term “affinity” refers to the equilibrium constant for the reversible binding of two agents and is expressed as the dissociation constant (KD). Affinity of a binding agent to a ligand, such as affinity of an antibody for an epitope, can be, for example, from about 1 femtomolar (fM) to about 1 millimolar (mM) (e.g., from about 1 picomolar (pM) to about 1 nanomolar (nM), or from about 1 nM to about 1 micromolar (pM)). In some embodiments, the affinity of an anti-C3 antibody may be from about 1 nm to about 20 nm, and desirably from about 5 nm to about 10 nm. Antibody affinity for an antigen or epitope of interest can be measured using any art-recognized assay. Such methods include, for example, fluorescence activated cell sorting (FACS), separable beads (e.g., magnetic beads), antigen panning, and/or ELISA (see, e.g., Janeway et al. (eds.), Immunobiology, 5th ed., Garland Publishing, New York, N.Y., 2001).
[00119] In some embodiments, an anti-C3 antibody, or composition comprising an anti-C3 antibody, may be administered alone or in combination with other drugs. For example, the anti- C3 antibody can be administered in combination with other agents for the treatment or prevention of AMD and/or GA, as disclosed herein. For example, anti-C3 antibodies of the present disclosure, or antibody conjugates, fusion proteins, or polymeric formulations thereof, can be used either alone or in combination with other agents in a therapy. For instance, an anti-C3 antibody may be coadministered with at least one additional therapeutic agent. In certain embodiments, an additional therapeutic agent is another antibody, a chemotherapeutic agent, a cytotoxic agent, an anti- angiogenic agent, an immunosuppressive agent, a prodrug, a cytokine, a cytokine antagonist, cytotoxic radiotherapy, a corticosteroid, an anti-emetic, a cancer vaccine, an analgesic, a growth- inhibitory agent, or combinations thereof.
[00120] For example, in certain embodiments, any of the preceding methods further comprises administering one or more additional compounds. In certain embodiments, the anti-C3 antibody, antibody conjugate, fusion protein, or polymeric formulation is administered simultaneously with the additional compound(s). In certain embodiments, the anti-C3 antibody, antibody conjugate,
fusion protein, or polymeric formulation is administered before or after the additional compound(s). In certain embodiments, the additional compound binds to a second biological molecule selected from the group consisting of VEGF, IL-10; IL-6; IL-6R; IL-13; IL-13R; PDGF; angiopoietin; Ang2; Tie2; SIP; integrins av03, av05, and a501; betacellulin; apelin/APJ; erythropoietin; complement factor D; TNFa; HtrAl; a VEGF receptor; ST-2 receptor; and proteins genetically linked to AMD risk, such as complement pathway components C2, factor B, factor H, CFHR3, CFHR4, C5, C5a, HtrAl, ARMS2, TIMP3, HLA, interleukin-8 (IL-8), CX3CR1, TLR3, TLR4, CETP, LIPC, COL10A1, and TNFRSF10A. In some embodiments, the additional compound is an antibody or antigen-binding fragment thereof. In some embodiments according to (or as applied to) any of the embodiments above, the ocular disorder is an intraocular neovascular disease selected from the group consisting of proliferative retinopathies, choroidal neovascularization (CNV), age-related macular degeneration (AMD), geographic atrophy (GA), diabetic and other ischemia-related retinopathies, diabetic macular edema, pathological myopia, von Hippel-Lindau disease, histoplasmosis of the eye, retinal vein occlusion (RVO), including CRVO and BRVO, corneal neovascularization, retinal neovascularization, and retinopathy of prematurity (ROP).
[00121] In some instances, an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, may be administered in combination with at least one additional therapeutic agent for treatment of an ocular disorder, for example, an ocular disorder described herein (e.g., AMD (e.g., wet AMD or dry AMD), GA, DME, DR, or RVO). Exemplary additional therapeutic agents for combination therapy for treatment of ocular disorders include, without limitation, anti-angiogenic agents, such as VEGF antagonists, including, for example, anti-VEGF antibodies (e.g., the anti-VEGF Fab LUCENTIS® (ranibizumab)), soluble receptor fusion proteins (e.g., the recombinant soluble receptor fusion protein EYLEA® (aflibercept, also known as VEGF Trap Eye; Regeneron/ Aventis)), aptamers (e.g., the anti-VEGF pegylated aptamer MACUGEN® (pegaptanib sodium; NeXstar Pharmaceuticals/OSI Pharmaceuticals)), and VEGFR tyrosine kinase inhibitors (e.g., 4-(4-bromo- 2-fluoroanilino)-6-methoxy-7-(l -methyip iperidin-4-ylmethoxy)quinazoline (ZD6474), 4-(4- fLuoro-2-methylindol-5-yloxy)-6-methoxy-7-(3-pyrrolidin-l -ylpropoxyjquinazoline (AZD2171), vatalanib (PTK787), semaxaminib (SU5416; SUGEN), and SUTENT® (sunitinib));
Tryptophanyl-tRNA synthetase (TrpRS); squalamine; RETAANE® (anecortave acetate for depot suspension; Alcon, Inc.); Combretastatin A4 Prodrug (CA4P); MIFEPREX® (mifepristone- ru486); subtenon triamcinolone acetonide; intravitreal crystalline triamcinolone acetonide; matrix metalloproteinase inhibitors (e.g., Prinomastat (AG3340; Pfizer)); fluocinolone acetonide (including fluocinolone intraocular implant; Bausch & Lomb/Control Delivery Systems); linomide; inhibitors of integrin [S3 function; angiostatin, and combinations thereof.
[00122] Further examples of additional therapeutic agents that can be used in combination with an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO), include, but are not limited to, VISUDYNE® (verteporfin; a light-activated drug that is typically used in conjunction with photodynamic therapy with a non-thermal laser), PKC412, Endovion (NS 3728; NeuroSearch A/S), neurotrophic factors (e.g., glial derived neurotrophic factor (GDNF) and ciliary neurotrophic factor (CNTF)), diltiazem, dorzolamide, PHOTOTROP®, 9-cis-retinal, eye medication (e.g., phospholine iodide, echothiophate, or carbonic anhydrase inhibitors), veovastat (AE-941; AEterna Laboratories, Inc.), Sirna-027 (AGF-745; Sirna Therapeutics, Inc.), neurotrophins (including, by way of example only, NT-4/5, Genentech), Cand5 (Acuity Pharmaceuticals), INS-37217 (Inspire Pharmaceuticals), integrin antagonists (including those from Jerini AG and Abbott Laboratories), EG-3306 (Ark Therapeutics Ltd.), BDM-E (BioDiem Ltd.), thalidomide (as used, for example, by EntreMed, Inc.), cardiotrophin-1 (Genentech), 2-methoxyestradiol (Allergan/Oculex), DL-8234 (Toray Industries), NTC-200 (Neurotech), tetrathiomolybdate (University of Michigan), LYN-002 (Lynkeus Biotech), microalgal compound (Aquasearch/ Albany, Mera Pharmaceuticals), D-9120 (Celitech Group pic), ATX-S10 (Hamamatsu Photonics), TGF-beta 2 (Genzyme/Celtrix), tyrosine kinase inhibitors (e.g., those from Allergan, SUGEN, or Pfizer), NX-278-L (NeXstar Pharmaceuticals/Gilead Sciences), Opt-24 (OPTIS France SA), retinal cell ganglion neuroprotectants (Cogent Neurosciences), N-nitropyrazole derivatives (Texas A&M University System), KP-102 (Krenitsky Pharmaceuticals), cyclosporin A, therapeutic agents used in photodynamic therapy (e.g., VISUDYNE®; receptor-targeted PDT, Bristol-Myers Squibb, Co.; porfimer sodium for injection with PDT; verteporfin, QLT Inc.; rostaporfin with PDT, Miravent Medical Technologies; talaporfin sodium with PDT, Nippon Petroleum; and motexafin lutetium, Pharmacy dies, Inc.),
antisense oligonucleotides (including, by way of example, products tested by Novagali Pharma SA and ISIS- 13650, Isis Pharmaceuticals), and combinations thereof.
[00123] An anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, may be administered in combination with a therapy or surgical procedure for treatment of an ocular disorder (e.g., AMD, GA DME, DR, or RVO), including, for example, laser photocoagulation (e.g., panretinal photocoagulation (PRP)), drusen lasering, macular hole surgery, macular translocation surgery, implantable miniature telescopes, PHI-motion angiography (also known as micro-laser therapy and feeder vessel treatment), proton beam therapy, microstimulation therapy, retinal detachment and vitreous surgery, scleral buckle, submacular surgery, transpup illary thermotherapy, photosystem I therapy, use of RNA interference (RNAi), extracorporeal rheopheresis (also known as membrane differential filtration and rheotherapy), microchip implantation, stem cell therapy, gene replacement therapy, ribozyme gene therapy (including gene therapy for hypoxia response element, Oxford Biomedica; Lentipak, Genetix; and PDEF gene therapy, GenVec), photoreceptor/retinal cells transplantation (including transplantable retinal epithelial cells, Diacrin, Inc.; retinal cell transplant, Cell Genesys, Inc.), acupuncture, and combinations thereof.
[00124] In some embodiments, an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, can be administered in combination with an anti-angiogenic agent for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO). Any suitable anti-angiogenic agent can be used in combination with an antibody of the present disclosure, including, but not limited to, those listed by Carmeliet et al. Nature 407:249-257, 2000. In some embodiments, the anti-angiogenic agent is a VEGF antagonist, including, but not limited to, an anti-VEGF antibody (e.g., the anti-VEGF Fab LUCENTIS® (ranibizumab), RTH-258 (formerly ESBA-1008, an anti-VEGF single-chain antibody fragment; Novartis), or a bispecific anti-VEGF antibody (e.g., an anti-VEGF/anti- angiopoietin 2 bispecific antibody such as RG-7716; Roche)), a soluble recombinant receptor fusion protein (e.g., EYLEA® (aflibercept)), a VEGF variant, a soluble VEGFR fragment, an aptamer capable of blocking VEGF (e.g., pegaptanib) or VEGFR, a neutralizing anti-VEGFR antibody, a small molecule inhibitor of VEGFR tyrosine kinases, an anti-VEGF DARPin® (e.g., abicipar pegol), a small interfering RNAs which inhibits expression of VEGF or VEGFR, a
VEGFR tyrosine kinase inhibitor (e.g., 4-(4-bromo-2-fluoroanilino)-6-methoxy-7-(l- methylpiperidin-4-ylmethoxy)quinazoline (ZD6474), 4-(4-fhioro-2-methylindol-5-yloxy)-6- methoxy-7-(3-pyrrolidin-l-ylpropoxy)quinazoline (AZD2171), vatalanib (PTK787), semaxaminib (SU5416; SUGEN), and SUTENT® (sunitinib)), and combinations thereof.
[00125] Other suitable anti-angiogenic agents that may be administered in combination with an antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO) include corticosteroids, angiostatic steroids, anecortave acetate, angiostatin, endostatin, tyrosine kinase inhibitors, matrix metalloproteinase (MMP) inhibitors, insulin-like growth factor-binding protein 3 (IGFBP3), stromal derived factor (SDF-1) antagonists (e.g., anti-SDF-1 antibodies), pigment epithelium-derived factor (PEDF), gamma-secretase, Delta-like ligand 4, integrin antagonists, hypoxia-inducible factor (HIF)-la antagonists, protein kinase CK2 antagonists, agents that inhibit stem cell (e.g., endothelial progenitor cell) homing to the site of neovascularization (e.g., an anti-vascular endothelial cadherin (CD-144) antibody and/or an anti- SDF-1 antibody), and combinations thereof.
[00126] In some embodiments, an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, can be administered in combination with an agent that has activity against neovascularization for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO), such as an anti-inflammatory drug, a mammalian target of rapamycin (mTOR) inhibitor (e.g., rapamycin, AFINITOR® (everolimus), and TORISEL® (temsirolimus)), cyclosporine, a tumor necrosis factor (TNF) antagonist (e.g., an anti- TNFa antibody or antigen-binding fragment thereof (e.g., infliximab, adalimumab, certolizumab pegol, and golimumab) or a soluble receptor fusion protein (e.g., etanercept)), an anti-complement agent, a nonsteroidal anti-inflammatory agent (NS AID), or combinations thereof.
[00127] Any suitable AMD therapeutic agent can be administered as an additional therapeutic agent in combination with an anti-C3 antibody of the present disclosure, or an antibody conjugate, fusion protein, and/or polymeric formulation thereof, for treatment of an ocular disorder (e.g., AMD, GA, DME, DR, or RVO), including, but not limited to, a VEGF antagonist, for example, an anti- VEGF antibody (e.g., LUCENTIS® (ranibizumab), RTH-258 (formerly ESBA-1008, an anti-VEGF single-chain antibody fragment; Novartis), or a bispecific anti- VEGF antibody (e.g.,
an anti-VEGF/anti-angiopoietin 2 bispecific antibody such as RG-7716; Roche)), a soluble VEGF receptor fusion protein (e.g., EYLEA® (aflibercept)), an anti- VEGF DARPin® (e.g., abicipar pegol; Molecular Partners AG/ Allergan), or an anti- VEGF aptamer (e.g., MACUGEN® (pegaptanib sodium)); a platelet-derived growth factor (PDGF) antagonist, for example, an anti- PDGF antibody, an anti-PDGFR antibody (e.g., REGN2176-3), an anti-PDGF-BB pegylated aptamer (e.g., FOVISTA®; Ophthotech/Novartis), a soluble PDGFR receptor fusion protein, or a dual PDGF/VEGF antagonist (e.g., a small molecule inhibitor (e.g., DE- 120 (Santen) or X-82 (TyrogeneX)) or a bispecific anti-PDGF/anti-VEGF antibody)); VISUDYNE® (verteporfin) in combination with photodynamic therapy; an antioxidant; a complement system antagonist, for example, a complement factor C5 antagonist (e.g., a small molecule inhibitor (e.g., ARC-1905; Opthotech) or an anti-C5 antibody (e.g., LFG-316; Novartis), a properdin antagonist (e.g., an antiproperdin antibody, e.g., CLG-561; Alcon), or a complement factor D antagonist (e.g., an anticomplement factor D antibody, e.g., lampalizumab; Roche)); a visual cycle modifier (e.g., emixustat hydrochloride); squalamine (e.g., OHR-102; Ohr Pharmaceutical); vitamin and mineral supplements (e.g., those described in the Age-Related Eye Disease Study 1 (AREDS1; zinc and/or antioxidants) and Study 2 (AREDS2; zinc, antioxidants, lutein, zeaxanthin, and/or omega-3 fatty acids)); a cell-based therapy, for example, NT-501 (Renexus); PH-05206388 (Pfizer), huCNS-SC cell transplantation (StemCells), CNTO-2476 (Janssen), OpRegen (Cell Cure Neurosciences), or MA09-hRPE cell transplantation (Ocata Therapeutics); a tissue factor antagonist (e.g., hl-conl; Iconic Therapeutics); an alpha-adrenergic receptor agonist (e.g., brimonidine tartrate); a peptide vaccine (e.g., S-646240; Shionogi); an amyloid beta antagonist (e.g., an anti-beta amyloid monoclonal antibody, e.g., GSK-933776); an SIP antagonist (e.g., an anti-SIP antibody, e.g., iSONEP™; Lpath Inc); a ROBO4 antagonist (e.g., an anti-ROBO4 antibody, e.g., DS-7080a; Daiichi Sankyo); a lentiviral vector expressing endostatin and angiostatin (e.g., RetinoStat); and any combination thereof. In some instances, AMD therapeutic agents (including any of the preceding AMD therapeutic agents) can be co-formulated. For example, the anti-PDGFR antibody REGN2 176-3 can be co-formulated with aflibercept (EYLEA®). In some instances, such a coformulation can be administered in combination with an antibody of the present disclosure. In some instances, the ocular disorder is AMD (e.g., wet AMD).
[00128] In addition to therapeutic uses, an anti-C3 antibody or antigen-binding fragment, described herein can be used in diagnostic or research applications. Research applications include, for example, methods that utilize the anti-C3 antibody and a label to detect C3, C3a, and/or C3b in a sample, e.g., in a human body fluid or in a cell or tissue extract. The anti-C3 antibody or antigen-binding fragment thereof may be employed in any suitable assay for measuring C3, C3a, and/or C3b in a sample for diagnostic and/or research purposes. Such assays include, but are not limited to, sandwich immunoassays, enzyme immunoassays (EIA), enzyme-linked immunosorbent assays (ELISA), lateral flow assays, competitive inhibition immunoassays (e.g., forward and reverse), competitive binding assays, Forster resonance energy transfer (FRET), one- step antibody detection assays, single molecule detection assays, radioimmunoassays (RIA), and FACS. Such methods are disclosed in, for example, U.S. Patents 6,143,576; 6,113,855; 6,019,944; 5,985,579; 5,947,124; 5,939,272; 5,922,615; 5,885,527; 5,851,776; 5,824,799; 5,679,526; 5,525,524; and 5,480,792; and Adamczyk et al., Anal. Chim. Acta, 579(1): 61-67 (2006).
[00129] The anti-C3 antibody or antigen-binding fragment thereof can be provided in a kit, e.g., a packaged combination of reagents in predetermined amounts with instructions for performing an assay using the antibody (e.g., an assay that detects C3, C3a, and/or C3b). As such, the disclosure provides a kit comprising the antibody or antigen-binding fragment described herein and instructions for use thereof. The instructions can be in paper form or computer-readable form, such as a disk, CD, DVD, etc. Alternatively or additionally, the kit can comprise a calibrator or control, and/or at least one container (e.g., tube, microtiter plates, or strips) for conducting an assay, and/or a buffer, such as an assay buffer or a wash buffer. Ideally, the kit comprises all components, i.e., reagents, standards, buffers, diluents, etc., which are necessary to perform the assay. Other additives may be included in the kit, such as stabilizers, buffers (e.g., a blocking buffer or lysis buffer), and the like. The relative amounts of the various reagents can be varied to provide for concentrations in solution of the reagents which substantially optimize the sensitivity of the assay. The reagents may be provided as dry powders (typically lyophilized), including excipients which on dissolution will provide a reagent solution having the appropriate concentration.
[00130] The following examples further illustrate the various embodiments of the present disclosure but should not be construed as in any way limiting its scope.
EXAMPLES
[00131] It will be readily apparent to those skilled in the art that other suitable modifications and adaptations of the methods of the present disclosure described herein are readily applicable and appreciable, and may be made using suitable equivalents without departing from the scope of the present disclosure or the aspects and embodiments disclosed herein. Having now described the present disclosure in detail, the same will be more clearly understood by reference to the following examples, which are merely intended only to illustrate some aspects and embodiments of the disclosure, and should not be viewed as limiting to the scope of the disclosure. The disclosures of all journal references, U.S. patents, and publications referred to herein are hereby incorporated by reference in their entireties.
[00132] The present disclosure has multiple aspects, illustrated by the following non-limiting examples.
EXAMPLE 1
[00133] Recovery of C3 antibody sequences from immunized mice. C3 antibody sequences were recovered from immunized mice followed by phage display with either fixed or diverse light chains. Selected leads were produced as full aglyco IgGls and extensively characterized.
[00134] C3 Recombinant Proteins. C3 proteins were procured from Complement Technologies (Tyler, TX); the proteins were purified directly from the relevant species (human or cynomolgus) sera. Recombinant proteins were qualified with analytic SEC and binding studies to established antibodies.
[00135] C3 Immunization. Two cohorts of Alloy Therapeutics transgenic humanized mice, ATX-GK Mix, were immunized with either human C3 or cynomolgus C3 using the following 5 week protocols: Cohort 1: 5 ATX-GX mix mice immunized with human C3 (REA23) using standard 5-week RIMMS protocol with 10 ug subcutaneous dosing of antigen. The first dose was emulsified in complete Freund’s adjuvant with 4 follow on weekly 10 ug doses in incomplete Freund’s adjuvant. Cohort 2 As Cohort 1 but with cynomolgus C3 (REA20).
[00136] Sample bleeds were conducted at the fourth week and tested for antigen positive titer and cross reactive titer (human for cynomolgus cohort or cynomolgus for human cohort). ELISA plates were coated with 1 ug/ml of relevant recombinant protein overnight. Coated plates were
incubated with sera across a descending 5 x 10-fold range starting at 1:100 and terminating at 1 : 10E7 dilutions. Antibodies bound to the coated antigen were detected with anti-mouse IgG HRP secondary antibody and quantified with the aid of one step TMB solution and OD determination at 450 nM using a microplate reader. Note, the positive control antibody used for the ELISA experiment is not cross reactive to the cyno C3 protein (FIGS. 1A-1D).
[00137] Antibody Sequence Recovery - Phage Display. Spleen tissues from high titer mice were preserved at harvest for sequence recovery via phage display. RNA was extracted from said tissues. Two sequence recovery strategies were used in this study, a fixed light strategy and a diverse light chain strategy. For both strategies, variable heavy chains were amplified from the RNA prepared from the spleen tissues via reverse transcription. Following reverse transcription, viable gene specific primers and PCR were used to create DNA fragments which could be cloned into a phage display vector designed to express Fabs on phage g3p protein. For the diverse light chain strategy, the same approach was used to amplify the natural diversity of the light chain from the spleen RNA and clone into the Fab vector. For the fixed light chain strategy, the relevant part of the Fab vector was fixed to two specific sequences with constructs synthesized commercially that match the LC of ATX-P-569 and ATX-P-592.
[00138] Once complete, the libraries of phage expressing unique Fabs or Fabs with fixed light chains were amplified, purified, and depleted on non-specific binders. Phage were allowed to bind to biotinylated C3 antigens (human and/or cyno) captured on streptavidin magnetic beads. Phage remaining bound to antigens beads after several stringent washes was eluted using a basic triethylamine solution and neutralized with Tris buffer pH 8.0. Eluted phages were reinfected into TGI bacterial cells, amplified by co-infection with M12 helper phage, and purified by PEG precipitation. A small sample was saved for polyclonal analysis. Purified phages expressing Fabs were selected for antigen binding as described earlier in a subsequent 2nd round. Phage from the second round output were diluted and infected in TGI cells. Polyclonal pools of phage output from both rounds were tested by ELISA to confirm enrichment and examine cross reactivity (FIG. 2).
[00139] Follow on monoclonal ELISA on individual colonies was conducted and from this second ELISA, clones were identified for rolling circle amplification and Sanger Sequencing of the variable heavy and light chain regions of the Fab where appropriate (for diverse libraries only, for fixed LC libraries only sequencing the HC was required). From the sequence analysis, a group
of clones were advanced for reformatting into full length antibodies. The selected group broadly represented the clonotype diversity discovered in both the fixed/cLC and diverse libraries. In total, 113 paired variable heavy and light chain sequences were put into production across the fixed and diverse libraries.
[00140] Antibody Production. Unique variable heavy and light chain pairs from the phage display campaigns were cloned into vectors designed to express full length antibodies as algyco IgGls in HEK293 cells under the control of a CMV promoter. Antibody expression vectors were complexed with polyethylenimine and transfected into HEK293 cultures. After 5 days of shaking at 37 C in 293 cell culture media, antibodies were captured on agarose-based protein A resin. After several stringent washes, antibodies were eluted in citrate solution, pH 3, neutralized with Hepes, pH 9, and buffer exchanged into PBS.
EXAMPLE 2
[00141] C3 Antibody Binding Kinetics. Experiments were conducted to determine the binding kinetics of anti-C3 antibodies, species cross-reactivity, and epitope cross-blocking.
[00142] Kinetic experiments were performed on Carterra LSA with a running buffer of PBS pH 7.40, 1% BSA, 0.05% Tween20. Antibodies were captured on an anti-human Fc capture chip prepared with an HC30M chip. For kinetics analysis, C3a, C3b from Complement Technology, or C3-His from Aero Biosystems, were injected sequentially at a concentration from 0.076 nM to 1500 nM (a serial 3-fold dilution). For each concentration, there was a 5 Min association followed by 15 Min dissociation. Results were processed and analyzed in Carterra LSA Kinetics Software. The kinetic data was referenced with the interstitial reference spots and double-referenced to a buffer cycle, and then fit globally to a 1 : 1 binding model to determine their apparent association and dissociation kinetic rate constants (Ka and KD values). The ratio KD / Ka was used to derive the KD value of each antigen/mAb interaction (KD = KD / Ka) (FIG. 3).
[00143] Cross-reactivity assay by SPRi. The binding experiments were performed on Carterra LSA with a running buffer of PBS pH 7.40, 1% BSA, 0.05% Tween20. Antibodies were covalently printed on an HC30M chip. Chip was activated with 33 mM s-NHS and 133 mM EDC in 100 mM MES pH 5.5 for 7 minutes. Antibodies at 10 mg/ml in acetic acid buffer pH 4.5 were used for printing for 10 min. The printed chip was then quenched with 1 M ethanolamine pH 8.5 for 7 min.
For the reactivity assay, human C3, C3a, C3b, and cyno C3 from Complement Technology was tested with 5 Min association followed by 15 Min dissociation. Results were processed and analyzed in Carterra LSA Kinetics Software. The data was referenced with the interstitial reference spots and double-referenced to a buffer cycle, and then the responses (nm) after association were reported. Isotype control was used to determine the cutoff response for positive binding (FIGS. 4 and 5).
[00144] Epitope Binning. High-throughput epitope binning experiment was done on real-time label-free biosensors (Carterra LSA) to sort large panels of mAbs into bins based on their ability to block one another for binding to the antigen. In a pairwise epitope binning analysis, antigen and antibody 2 (analyte antibody) are sequentially applied to the sensor chip (HC200M) covalently pre-loaded with antibody 1 (ligand antibody). An increase in response upon exposure to the analyte antibody indicates non-competition between the two antibodies, whereas a lack of change in the signal indicates competition. Antibodies having the same blocking profiles towards others in the test set are grouped into one bin. Community network plots are used to explore clustering of mAbs that share similar but not necessarily identical competition profiles. Rather than relying strictly on the sandwiching/blocking assignments in the heat map, as the Bin network plots do, hierarchical clustering is applied to the sorted heat map to generate dendrograms, which progressively group mAbs (FIGS. 6 and 7).
EXAMPLE 3
[00145] C3 Antibody Polyreactivity. Experiments were conducted to determine the reactivity of the C3 antibodies of the present disclosure with various other proteins using ELISA and Carterra LSA platforms (FIGS. 8 and 9). The methods used were similar as reported by Hotzel et al. (2012). Briefly, baculovirus particles (BVP, Lake Pharma) was diluted 1:100 in 50 rnM sodium bicarbonate (pH 9.3). After overnight incubation of 50 pL of BVP on ELISA plates (3369; Corning) at 4 °C overnight, unbound BVPs were aspirated from the wells. All remaining steps were performed at RT. The plate was blocked with 100 pL of blocking buffer (PBS with 1% BSA) for 1 h before three washes with 100 pL of PBS. Next, 50 pL of 16 nM testing antibodies was added to the wells and incubated for 1 h followed by washes with 100 pL of PBS. HRP-conjugated goat anti-human IgG antibody at 1:1000 (Jackson ImmunoResearch) was used as the secondary
antibody, and incubated for 1 h followed by washes as before. Finally, 50 pL of TMB substrate (34021; Fisher Scientific) was added to each well and incubated for 10-15 min. The reactions were stopped by adding 50 pL of 2 M sulfuric acid to each well. The absorbance was read at 450 nm and BVP score determined by normalizing absorbance by control wells with no test antibody.
[00146] Additional experiments were conducted to determine the blocking potential of the C3 antibodies of the present disclosure for CFHR4 (FIG. 10B) and CFH (FIG. 10B).
EXAMPLE 4
[00147] Experiments were conducted to assess the effects of the C3 antibodies of the present disclosure on alternative pathway complement activation based on the quantification of red blood cell hemolysis (FIG. 11). Briefly, to measure complement activity, sheep or human red blood cells (SRBCs and HRBCs) were used to perform a hemolytic assay. Erythrocytes were washed and suspended in Gelatin Veronal buffer containing Mg2+ and Ca2+ (GVB++). A 1% erythrocyte suspension was incubated for 30 min at 37 °C with 1/200 diluted rabbit anti-SRBC or rabbit anti- HRBC serum. The Ab-sensitized erythrocytes were washed and re-suspended in GVB+ + to 2%. Then, lOOpl of the erythrocyte suspension with GVB+ + diluted with an equal volume of serum. To lyse the SRBCs, the serum was diluted to 30%, 15%, 7.5%, and 3.75%. To lyse the HRBCs, the freshly prepared mouse serum was diluted at 15%, 7.5%, 3.75%, 1.875%.
[00148] Experiments were conducted in 96-well plates in triplicate. The plates were incubated for 30 min at 37 °C. Unlysed cells were removed by centrifugation, and hemoglobin in the supernatant was measured on the basis of the ABS450. The percentage lysis was calculated as follows: % LYSIS = (test ABS45O - blank ABS45o/total lysis ABS450) x 100.
[00149] To reconstitute the complement-mediated hemolysis caused by the C9-/- serum, Ab - sensitized HRBCs were incubated primarily with GVB+ + - diluted 15% mC9-/- derived serum. The unlysed erythrocytes were washed and collected by centrifugation. A 2% erythrocyte suspension was prepared by adding normal saline (NS) to the HRBC pellet. mC9+/+, mC9+/- or mC9-/- serum was pretreated with 10 mM EDTA for 30 min and then diluted with NS before addition of an equal volume to the erythrocyte suspension. Hemoglobin in the supernatant and the percentage lysis were analyzed as described above.
[00150] As shown in FIG. 11, an assay was conducted to test whether the C3b antibodies of the present disclosure were capable of activating the alternative complement pathway by quantifying sheep red blood cell (SRBC) hemolysis. The assay was run twice in parallel, once with sensitized SRBC (FIG. 11) and once with non-sensitized SRBC. As shown, eight C3b antibodies were tested in parallel with compstatin, a cyclic peptide that binds to C3 and inhibits its cleavage. The compstatin assays were run with and without normal human serum (NHS). NHS is the source of complement components and in its absence there is no SRBC lysis. (This is the expected result as any lysis in the absence of NHS would be non-complement mediated, e.g., the result of residual detergent.) The results indicated that the C3b antibodies tested reduced percent hemolysis at increasing concentrations, thus demonstrating their efficacy at inhibiting activation of the alternative complement pathway.
[00151] Sequences
[00152] The various amino acid sequences and nucleic acid sequences referenced herein are provided below.
[00158] Table 6: Summary of SEQ ID NOs for individual exemplary antibodies of the present disclosure.
KEY: aa = amino acid; HCDR 1, 2, 3 = heavy chain CDRs 1, 2, and 3 (in numerical order); LCDRs 1, 2, 3 = light chain CDRs 1, 2, and 3 (in numerical order); VH = heavy chain variable region; VL = light chain variable region; VH + constant = heavy chain variable region sequence with an exemplary human heavy chain constant region sequence (typically IgGl); VL + constant = light chain variable region sequence with an exemplary human light chain constant region sequence (typically kappa). All numbers except in “Antibody Name” correspond to SEQ ID NOs in the sequence listing and Tables 1, 2, 3, 4, and 5.
[00159] Human Complement Component 3 (C3), which includes Complement Component 3a (C3a) and Complement Component 3b (C3b), was used to generate the anti-C3 antibodies of the present disclosure (see, e.g., UniProt Accession No. M0QYC8): ITHRIHWESASLLRSEETKENEGFTVTAEGKGQGTLSVVTMYHAKAKDQLT CNKFDLKVTIKPAPETGIPSPIFLSSVFLEKRPQDAKNTMILEICTRYRGDQDATMSILDIS MMTGFAPDTDDLKQLANGVDRYISKYELDKAFSDRNTLIIYLDKVSHSEDDCLAFKVH QYFNVELIQPGAVKVYAYYNL (SEQ ID NO: 1064).
[00160] Various embodiments of the present disclosure are described herein. Variations of those embodiments may become apparent to those of ordinary skill in the art upon reading the foregoing description. The inventors expect skilled artisans to employ such variations as appropriate, and the inventors intend for the various embodiments of the present disclosure to be practiced otherwise than as specifically described herein. Accordingly, embodiments of the present disclosure include all modifications and equivalents of the subject matter recited in the claims appended hereto as permitted by applicable law. Moreover, any combination of the abovedescribed elements in all possible variations thereof is encompassed by the various embodiments of the present disclosure unless otherwise indicated herein or otherwise clearly contradicted by context.
Claims
1. An antibody, or antigen binding fragment thereof, which specifically binds Complement Component 3 (C3) polypeptides, optionally wherein said C3 is a Complement Component 3a (C3a) polypeptide or a Complement Component 3b (C3b) polypeptide, which is optionally a polypeptide comprising or consisting of the amino acid sequence of SEQ ID NO: 1064.
2. The antibody or fragment of claim 1, which exhibits any one or more the following functional characteristics: a. reduces complement activation and/or attenuates complement component 3 (C3) convertase activity, which is optionally measured in an ELISA; and/or b. cross-reacts with at least one of human C3b protein, Cynomolgus monkey C3 protein, and/or human C3a protein; and/or c. binds to human C3 with a KD-1 of about lOOnM or lower; and/or d. binds to the same epitope on human C3 as an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or e. competes for binding to human C3 with an antibody comprising the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6.
3. The antibody or fragment according to any one of the preceding claims, which is a monoclonal antibody.
4. The antibody or fragment according to any one of the preceding claims, which is a human, humanized, or chimeric antibody.
5. The antibody or fragment according to any one of the preceding claims, which is a whole antibody, a single chain variable fragment (scFv), a variable fragment (Fv), a fragment antigen-
binding region (Fab), a Fab-C, a Fab’-SH, a (Fab’)2, a single-domain antibody (sdAb), a nanobody, a diabody, a triabody, or an aptamer, optionally wherein the antibody is a whole antibody comprising an Fc region such as a human IgGl, IgG2, IgG3 or IgG4 Fc region.
6. The antibody or fragment according to any one of the preceding claims, which is conjugated to at least one additional moiety, optionally selected from: a. a therapeutic or cytotoxic moiety; b. a detection moiety; c. a purification moiety; d. a half-life extension moiety, optionally a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residue, which polypeptide is conjugated to the C- or N- terminus of the antibody.
7. The antibody or fragment according to any one of the preceding claims, which is a polypeptide comprising: a. one, two or all three HCDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also one, two or all three of the corresponding LCDRs of said exemplary antibody; and/or b. a VH sequence having at least 90% identity to the VH sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally also a VL sequence having at least 90% identity to the corresponding VL sequence of said exemplary antibody, preferably wherein variation is not permitted in the HCDRs or LCDRs; and/or c. all six CDRs of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or d. the VH and VL sequences of any one of the exemplary antibodies the sequences of which are provided in Table 6; and/or e. the full length heavy chain (VH + constant) sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6, and optionally the corresponding full length light chain (VL + constant) sequence of said exemplary antibody.
8. A polynucleotide encoding an antibody or fragment according to any one of the preceding claims, optionally wherein said polynucleotide comprises or consists of a nucleic acid sequence having at least 70, 80, 90 or 100% identity to a nucleic acid sequence of any one of the exemplary antibodies the sequences of which are provided in Table 6.
9. An expression vector comprising the polynucleotide of claim 8, which is optionally an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, or a retrovirus vector.
10. A pharmaceutical composition comprising an antibody or fragment, a polynucleotide, or a vector according to any one of the preceding claims, and optionally a. at least one pharmaceutically acceptable carrier, a buffer, excipient, stabilizer, diluent, or preservative; and/or b. at least one additional active ingredient.
11. The pharmaceutical composition of claim 10, which is suitable for ocular administration to a subject, optionally by delivery using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant, preferably by injection into the vitreous fluid.
12. The antibody or fragment according to any one of claims 1 to 7, the polynucleotide of claim 8, the vector of claim 9, or the composition of claim 10 or 11, for use as a medicament, optionally for use in a method of treating a disease of the eye in a subject.
13. The antibody, fragment, polynucleotide, vector, or composition for use according to claim 12, wherein said disease is characterized by increased activation of the complement system, in particular the alternative pathway, and in particular within the eye of the subject, for example in drusen or retinal pigment epithelium (RPE) cells of the subject.
14. The antibody, fragment, polynucleotide, vector, or composition for use according to claim 12 or 13, wherein the method comprises ocular administration of the antibody, preferably by injection into the vitreous fluid, and wherein said administration preferably relieves at least one symptom in the subject selected from visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light.
15. The antibody, fragment, polynucleotide, vector, or composition for use according to any one of claims 12 to 14, where the disease is age-related macular degeneration (AMD), optionally wherein said AMD is dry AMD, which may be at an early, intermediate, or advanced stage (the latter otherwise known as geographic atrophy, GA).
16. An antibody directed against Complement Component 3 (C3) peptides, or an antigenbinding fragment thereof, comprising a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3, wherein: the HCDR1 comprises one of the following amino acid sequences:
(a) X1YX2X3X4 (SEQ ID NO: 1), wherein Xi is G, S, N, D, T, or A; X2 is Y, A, G, W, or T; X3 is L, M, I, or V; and X4 is H, N, S, or G;
(b) X1X2 X3MX4 (SEQ ID NO: 57), wherein Xi is D, N, S, or A; X2 is Y, or A; X3 is A, W, Y, S, C, D, or G; and X4 is H, S, or N;
(c) X1X2X3WWX4 (SEQ ID NO: 95), wherein Xi is I, S, R, N, or K; X2 is N, D, S, or G; X3 is N, S, T, D, Y, or H; and X4 is S, N, or T;
(d) X1YYX2X3 (SEQ ID NO: 181), wherein Xi is S, or D; X2 is W, or L; and X3 is S, or T;
(e) X1X2X3YX4WX5 (SEQ ID NO: 204), wherein Xi is S or N; X2 is G or N; X3 is S, K, R, N, or G; X4 is Y, F, or N; and X5 is S or N; or
(f) TSGVGVG (SEQ ID NO: 227);
wherein the HCDR2 comprises one of the following amino acid sequences:
(a) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 20), wherein Xi is W, I, or V; X2 is N, S, or Y; X3 is P, T, A, G, or Y; X4 is N, Y, G, D, H, or A; X5 is S, T, N, G, or D; X6 is G, or S; X7 is D, G, T, N, V, A, or H; X8 is T, P, or K; X9 is N, Y, T, H, R, I, D, or K; X10 is Y, or A; Xu is V, Q, A, or S; X12 is Q, K, P, or D; X13 is N, F, K, G, S, or Q; X14 is F, Q, L, or V; and X15 is Q, G, T, or K;
(b) X1IX2X3X4X5X6X7X8X9X10X11X12X13X14X15 (SEQ ID NO: 70), wherein Xi is G, R, N, S, H, T, V, or A; X2 is S, K, N, or G; X3 is W, S, Q, T, G, or Y; X4 is N, K, D, S, A, or Y; X5 is S, T, or G; X6 is G, D, or S; X7 is S, G, E, Y, T, or N; X8 is I, G, T, K, or Y; X9 is A, T, S, Y, or N; X10 is Y, T, P, or F; Xu is A, D, or V; X12 is D, Y, S, or A; X13 is S, A, or V; X14 is V, A, or K; and X15 is K, P, Q, or G;
(c) X1IX2X3SX4X5X6X7X8NPSLX9S (SEQ ID NO: 124), wherein Xi is K, Q, R, E, or D; X2 is Y or F; X3 is H or Q; X4 is G, E, or R; X5 is N, I, S, H, F, or T; Xe is T or I; X7 is D, N, or M; Xs is Y or H; and X9 is K or R;
(d) X1X2X3X4X5GX6TX7YNPSLKS (SEQ ID NO: 189) wherein Xi is Y or E; X2 is V or I; X3 is Y or N; X4 is Y or H; X5 is S or I; Xe is Y, F, or S; and X7 is N or K;
(e) YIX1X2SGX3TX4YNPSLKX5 (SEQ ID NO: 212) wherein Xi is Y or N; X2 is K, Y, or S; X3 is F, S, Y, or N; X4 is N, K, or Y; and X5 is S or N; or
(f) LIYWNX1X2X3X4YSPSLX5S (SEQ ID NO: 230) wherein Xi is G or D; X2 is D or V; X3 is E or K; X4 is R or F; and X5 is K or Q; wherein the HCDR3 comprises one of the following amino acid sequences:
(a) SEQ ID NOs: 39-56;
(b) SEQ ID NOs: 83-94;
(c) SEQ ID NOs: 153-180;
(d) SEQ ID NOs: 197-203;
(e) SEQ ID NOs: 220-226; or
(f) SEQ ID NOs: 234-235; and
wherein the LCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 237-267, SEQ ID NOs: 332-333, SEQ ID NOs: 341-348, or SEQ ID NOs: 368-401; the LCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 269-299, SEQ ID NOs: 335-336, SEQ ID NOs: 350-357, or SEQ ID NOs 403-436; and the LCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 300-330, SEQ ID NOs: 339-339, SEQ ID NOs: 359-366, or SEQ ID NOs: 438- 471.
17. An antibody directed against Complement Component 3 (C3) peptides, or an antigenbinding fragment thereof, comprising a heavy chain variable region (VH) comprising complementarity determining regions (CDRs) HCDR1, HCDR2, and HCDR3, and a light chain variable region (VL) comprising complementarity determining regions (CDRs) LCDR1, LCDR2, and LCDR3, wherein: the LCDR1 comprises one of the following amino acid sequences:
(a) X1ASQX2X3X4X5X6X7X8 (SEQ ID NO: 236), wherein Xi is R or Q; X2 is D, N, T, G, or S; X3 is V or I; X4 is S, I, R, G, T, or K; X5 is T, N, R, or S; X6 is A, W, F, D, Y, N, or C; X7 is V or L; Xs is A, N, G, or H;
(b) RSSQX1LLHX2NGYNYLD (SEQ ID NO: 331), wherein Xi is S or I; X2 is S or T; or
(c) RASQSX1X2X3X4X5X6 (SEQ ID NO: 340), wherein Xi is V or I; X2 is S or R; X3 is S, D, or R; X4 is Y or S; X5 is L or Y; Xe is A, F, or L; or
(d) KSSQX1X2LYX3SNX4KNYLA (SEQ ID NO: 367), wherein Xi is S or T; X2 is V or I; X3 is R or S; X4 is N or I; wherein the LCDR2 comprises one of the following amino acid sequences:
(a) X1ASX2X3X4X5 (SEQ ID NO: 268), wherein Xi is S, K, A, Y, T, D, or V; X2 is F, S, N, Q, or T; X3 is L, S, or F; X4 is Y, E, Q, or F; X5 is S or T;
(b) LGSXiRAS (SEQ ID NO: 334), wherein Xi is N or Y;
(c) Xi ASX2RAT (SEQ ID NO: 349), wherein Xi is D or G; X2 is S or N; or
(d) WASXiRES (SEQ ID NO: 402), wherein Xi is N or T;
wherein the LCDR3 comprises one of the following amino acid sequences:
(a) SEQ ID NOs: 300-330;
(b) MQX1LQTPX2T (SEQ ID NO: 337), wherein Xi is T or A; X2 is P or L;
(c) QQX1X2X3X4PX5T (SEQ ID NO: 358), wherein Xi is R or Y; X2 is S, G, or D; X3 is N or S; X4 is W or S; X5 is L, R, I, Y, F, or W; or
(d) QQX1X2X3TX4X5 (SEQ ID NO: 437), wherein Xi is Y or F; X2 is Y or F; X3 is S, G, or D; X4 is P or R; X5 is Y, W, R, T, or P; and wherein the HCDR1 comprises an amino acid sequence of any of SEQ ID NOs: 2-19, SEQ ID NOs: 58-69, SEQ ID NOs: 96-123, SEQ ID NOs: 182-188, SEQ ID NOs: 205-211, or SEQ ID NOs: 228-229; the HCDR2 comprises an amino acid sequence of any of SEQ ID NOs: 21-38, SEQ ID NOs: 71-82, SEQ ID NOs: 125-152, SEQ ID NOs 190-196, SEQ ID NOs 213-219, or SEQ ID NOs 231-232; and the HCDR3 comprises an amino acid sequence of any of SEQ ID NOs: 39-56, SEQ ID NOs: 83-94, SEQ ID NOs: 153-180, SEQ ID NOs: 197-203, SEQ ID NOs: 220-226, or SEQ ID NOs: 234-235.
18. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 2; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 21; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 39.
19. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 3; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 22; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 40.
20. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 4; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 23; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 41.
21. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 5; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 24; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 42.
22. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 6; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 25; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 43.
23. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 7; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 26; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 44.
24. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 8; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 27; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 45.
25. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 9; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 28; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 46.
26. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 10; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 29; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 47.
27. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 11; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 30; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 48.
28. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 12; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 31; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 49.
29. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 13; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 32; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 50.
30. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 14; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 33; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 51.
31. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 15; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 34; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 52.
32. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 16; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 35; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 53.
33. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 17; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 36; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 54.
34. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 18; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 37; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 55.
35. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 19; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 38; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 56.
36. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 58; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 71; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 83.
37. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 59; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 72; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 84.
38. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 60; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 73; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 85.
39. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 61; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 74; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 86.
40. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 62; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 75; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 87.
41. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 63; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 76; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 88.
42. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 64; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 77; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 89.
43. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 65; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 78; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 90.
44. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 66; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 79; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 91.
45. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 67; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 80; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 92.
46. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 68; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 81; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 93.
47. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 69; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 82; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 94.
48. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 96; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 125; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 153.
49. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 97; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 126; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 154.
50. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 98; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 127; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 155.
51. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 99; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 128; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 156.
52. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 100; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 129; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 157.
53. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 101; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 130; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 158.
54. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 102; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 131; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 159.
55. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 103; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 132; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 160.
56. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 104; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 133; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 161.
57. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 105; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 134; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 162.
58. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 106; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 135; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 163.
59. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 107; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 136; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 164.
60. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 108; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 137; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 165.
61. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 109; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 138; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 166.
62. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 110; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 139; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 167.
63. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 111; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 140; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 168.
64. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 112; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 141; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 169.
65. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 113; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 142; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 170.
66. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 114; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 143; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 171.
67. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 115; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 144; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 172.
68. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 116; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 145; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 173.
69. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 117; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 146; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 174.
70. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 118; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 147; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 175.
71. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 119; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 148; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 176.
72. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 120; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 149; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 177.
73. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 121; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 150; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 178.
74. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 122; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 151; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 179.
75. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 123; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 152; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 180.
76. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 182; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 190; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 197.
77. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 183; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 191; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 198.
78. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 184; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 192; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 199.
79. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 185; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 193; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 200.
80. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 186; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 194; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 201.
81. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 187; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 195; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 202.
82. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 188; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 196; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 203.
83. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 205; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 213; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 220.
84. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 206; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 214; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 221.
85. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 207; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 215; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 222.
86. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 208; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 216; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 223.
87. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 209; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 217; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 224.
88. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 210; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 218; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 225.
89. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 211; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 219; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 226.
90. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 228; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 231; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 234.
91. The antibody of claim 16 or claim 17, wherein the HCDR1 comprises the amino acid sequence of SEQ ID NO: 229; the HCDR2 comprises the amino acid sequence of SEQ ID NO: 232; and the HCDR3 comprises the amino acid sequence of SEQ ID NO: 35.
92. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 237; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 269; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 300.
93. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 238; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 270; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 301.
94. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 239; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 271; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 302.
95. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 240; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 272; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 303.
96. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 241; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 273; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 304.
97. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 242; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 274; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 305.
98. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 243; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 275; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 306.
99. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 244; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 276; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 307.
100. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 245; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 277; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 308.
101. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 246; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 278; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 309.
102. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 247; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 279; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 310.
103. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 248; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 280; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 311.
104. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 249; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 281; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 312.
105. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 250; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 282; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 313.
106. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 251; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 283; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 314.
107. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 252; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 284; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 315.
108. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 253; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 285; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 316.
109. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 254; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 286; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 317.
110. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 255; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 287; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 318.
111. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 256; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 288; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 319.
112. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 257; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 289; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 320.
113. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 258; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 290; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 321.
114. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 259; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 291; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 322.
115. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 260; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 292; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 323.
116. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 261; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 293; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 324.
117. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 262; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 294; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 325.
118. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 263; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 295; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 326.
119. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 264; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 296; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 327.
120. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 265; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 297; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 328.
121. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 266; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 298; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 329.
122. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 267; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 299; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 330.
123. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 332; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 335; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 338.
124. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 333; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 336; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 339.
125. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 341; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 350; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 359.
126. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 342; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 351; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 360.
127. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 343; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 352; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 361.
128. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 344; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 353; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 362.
129. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 345; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 354; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 363.
130. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 346; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 355; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 364.
131. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 347; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 356; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 365.
132. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 348; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 357; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 366.
133. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 368; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 403; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 438.
134. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 369; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 404; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 439.
135. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 370; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 405; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 440.
136. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 371; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 406; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 441.
137. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 372; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 407; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 442.
138. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 373; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 408; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 443.
139. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 374; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 409; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 444.
140. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 375; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 410; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 445.
141. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 376; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 411; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 446.
142. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 377; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 412; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 447.
143. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 378; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 413; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 448.
144. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 379; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 414; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 449.
145. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 380; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 415; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 450.
146. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 381; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 416; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 451.
147. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 382; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 417; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 452.
148. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 383; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 418; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 453.
149. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 384; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 419; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 454.
150. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 385; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 420; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 455.
151. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 386; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 421; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 456.
152. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 387; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 422; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 457.
153. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 388; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 423; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 458.
154. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 389; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 424; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 459.
155. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 390; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 425; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 460.
156. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 391; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 426; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 461.
157. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 392; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 427; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 462.
158. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 393; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 428; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 463.
159. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 394; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 429; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 464.
160. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 395; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 430; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 465.
161. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 396; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 431; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 466.
162. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 397; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 432; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 467.
163. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 398; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 433; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 468.
164. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 399; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 434; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 469.
165. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 400; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 435; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 470.
166. The antibody of claim 16 or claim 17, wherein the LCDR1 comprises the amino acid sequence of SEQ ID NO: 401; the LCDR2 comprises the amino acid sequence of SEQ ID NO: 436; and the LCDR3 comprises the amino acid sequence of SEQ ID NO: 471.
167. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to any of SEQ ID NOs: 546-619.
168. The antibody of claim 16 or claim 17, wherein the VL comprises an amino acid sequence that is at least 90% identical to any of SEQ ID NOs: 694-767.
169. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 546 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 694.
170. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 547 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 695.
171. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 548 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 696.
172. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 549 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 697.
173. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 550 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 698.
174. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 551 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 699.
175. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 552 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 700.
176. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 553 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 701.
177. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 554 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 702.
178. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 555 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 703.
179. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 556 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 704.
180. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 557 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 705.
181. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 558 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 706.
182. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 559 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 707.
183. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 560 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 708.
184. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 561 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 709.
185. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 562 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 710.
186. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 563 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 711.
187. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 564 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 712.
188. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 565 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 713.
189. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 566 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 714.
190. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 567 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 715.
191. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 568 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 716.
192. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 569 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 717.
193. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 570 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 718.
194. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 571 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 719.
195. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 572 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 720.
196. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 573 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 721.
197. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 574 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 722.
198. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 575 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 723.
199. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 576 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 724.
200. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 577 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 725.
201. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 578 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 726.
202. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 579 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 727.
203. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 580 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 728.
204. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 581 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 729.
205. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 582 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 730.
206. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 583 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 731.
207. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 584 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 732.
208. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 585 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 733.
209. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 586 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 734.
210. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 587 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 735.
211. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 588 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 736.
212. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 589 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 737.
213. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 590 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 738.
214. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 591 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 739.
215. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 592 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 740.
216. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 593 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 741.
217. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 594 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 742.
218. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 595 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 743.
219. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 596 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 744.
220. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 597 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 745.
221. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 598 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 746.
222. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 599 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 747.
223. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 600 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 748.
224. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 601 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 749.
225. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 602 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 750.
226. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 603 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 751.
227. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 604 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 752.
228. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 605 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 753.
229. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 606 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 754.
230. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 607 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 755.
231. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 608 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 756.
232. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 609 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 757.
233. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 610 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 758.
234. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 611 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 759.
235. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 612 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 760.
236. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 613 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 761.
237. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 614 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 762.
238. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 615 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 763.
239. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 616 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 764.
240. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 617 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 765.
241. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 618 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 766.
242. The antibody of claim 16 or claim 17, wherein the VH comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 619 and the VL comprises an amino acid sequence that is at least 90% identical to SEQ ID NO: 767.
243. The antibody of claim 16 or claim 17, wherein the antibody binds C3 and reduces complement activation.
244. The antibody of claim 16 or claim 17, wherein the antibody binds human C3 with a KD of about 100 nM or lower.
245. The antibody of claim 16 or claim 17, wherein the antibody attenuates complement component 3 (C3) convertase activity.
246. The antibody of claim 16 or claim 17, wherein the antibody is a monoclonal antibody, a human antibody, a humanized antibody, and/or a chimeric antibody.
247. The antibody of claim 16 or claim 17, wherein the antibody is a fragment selected from the group consisting of Fab, Fab-C, Fab'-SH, Fv, scFv, and (Fab')2 fragments.
248. The antibody of claim 16 or claim 17, wherein the antibody is a monospecific antibody.
249. The antibody of claim 16 or claim 17, wherein the antibody is a bispecific antibody.
250. The antibody of claim 16 or claim 17, wherein the antibody comprises a detection moiety.
251. The antibody of claim 16 or claim 17, wherein the antibody comprises a purification moiety.
252. The antibody of claim 16 or claim 17, wherein the antibody comprises a half-life extension moiety.
253. The antibody of claim 237, wherein the half-life extension moiety comprises a polypeptide that is at least 20 amino acids in length and comprises any combination of G, A, S T, E, and P residues.
254. The antibody of claim 237, wherein the half-life extension polypeptide is attached to the C-terminus or N-terminus of the antibody.
255. A pharmaceutical composition comprising any of the antibodies of claims 1 to 239.
256. The composition of claim 240, wherein the composition is suitable for ocular administration.
257. The composition of claim 241, wherein the ocular administration comprises injection into vitreous fluid.
258. The composition of claim 241, wherein the ocular administration comprises delivering the antibody using a conjunctival insert, a contact lens, a gel, a nanoparticle, a mucoadhesive polymer, an ointment, a solution, a suspension, eye drops, and/or an implant.
259. A method of treating age-related macular degeneration (AMD) comprising administering a pharmaceutical composition comprising an effective amount of the antibody of claim 1 or claim 2 to a subject in need thereof.
260. The method of claim 244, wherein the pharmaceutical composition is administered ocularly and treats at least one AMD symptom.
261. The method of claim 244, wherein AMD comprises wet AMD.
262. The method of claim 244, wherein AMD comprises dry AMD.
263. The method of any of claims 244-247, wherein the at least one AMD symptom comprises visual distortion, reduced central vision, blurred vision, and/or difficulty adapting to low light.
264. The method of any of claims 244-248, wherein administering the pharmaceutical composition reduces complement activation in the subject’s eye.
265. The method of any of claims 244-249, wherein the pharmaceutical composition is administered at a dose ranging from about 0.0001 mg/dose to about 100 mg/dose.
266. The method of any of claims 244-163, wherein the pharmaceutical composition is administered at a dose ranging from about 0.0001 mg/ml to about 100 mg/ml.
267. A polynucleotide comprising a nucleic acid sequence that is at least 70% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
268. A polynucleotide comprising a nucleic acid sequence that is at least 70% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
269. A polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
270. A polynucleotide comprising a nucleic acid sequence that is at least 80% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
271. A polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 472-545, and/or SEQ ID NOs: 620-693.
272. A polynucleotide comprising a nucleic acid sequence that is at least 90% identical to any of SEQ ID NOs: 768-841, and/or SEQ ID NOs: 916-989.
273. An expression vector comprising any of the polynucleotides of claims 252-257.
274. The expression vector of claim 258, wherein the expression vector is at least one of: an adeno-associated virus (AAV) vector, a lentiviral (LV) vector, a herpes simplex virus (HSV) vector, and a retrovirus vector.
275. A method of administering ocular gene therapy to a subject in need thereof comprising injecting a pharmaceutical composition comprising an effective amount of the expression vector of claim 259.
276. A method of treating age-related macular degeneration (AMD) comprising administering a pharmaceutical composition comprising an effective amount of the expression vector of claim 259, wherein administering the pharmaceutical composition treats at least one AMD symptom.
277. The antibody of claim 16 or claim 17, wherein the antibody binds an epitope from a C3 polypeptide having an amino acid sequence of SEQ ID NO: 1064.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363462638P | 2023-04-28 | 2023-04-28 | |
| US63/462,638 | 2023-04-28 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024227154A1 true WO2024227154A1 (en) | 2024-10-31 |
Family
ID=91185108
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/026808 Pending WO2024227154A1 (en) | 2023-04-28 | 2024-04-29 | Complement component 3 (c3)-specific antibodies and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024227154A1 (en) |
Citations (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US4828981A (en) | 1983-08-24 | 1989-05-09 | Synbiotics Corporation | Immunoassays for determining Dirofilaria immitis infection using antiidiotype monoclonal antibody reagents |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| US5034506A (en) | 1985-03-15 | 1991-07-23 | Anti-Gene Development Group | Uncharged morpholino-based polymers having achiral intersubunit linkages |
| WO1993001161A1 (en) | 1991-07-11 | 1993-01-21 | Pfizer Limited | Process for preparing sertraline intermediates |
| US5480792A (en) | 1990-09-14 | 1996-01-02 | Biosite Diagnostics, Inc. | Antibodies to complexes of ligand receptors and ligands and their utility in ligand-receptor assays |
| US5525524A (en) | 1991-04-10 | 1996-06-11 | Biosite Diagnostics, Inc. | Crosstalk inhibitors and their uses |
| US5679526A (en) | 1989-01-10 | 1997-10-21 | Biosite Diagnostics Incorporated | Threshold ligand-receptor assay |
| WO1998022607A1 (en) | 1996-11-22 | 1998-05-28 | Rhone-Poulenc Rorer S.A. | Method for producing recombinant virus |
| US5821337A (en) | 1991-06-14 | 1998-10-13 | Genentech, Inc. | Immunoglobulin variants |
| US5824799A (en) | 1993-09-24 | 1998-10-20 | Biosite Diagnostics Incorporated | Hybrid phthalocyanine derivatives and their uses |
| US5851776A (en) | 1991-04-12 | 1998-12-22 | Biosite Diagnostics, Inc. | Conjugates and assays for simultaneous detection of multiple ligands |
| WO1999006562A1 (en) | 1997-07-31 | 1999-02-11 | Chiron Corporation | Method enabling readministration of aav vector via immunosuppression of host |
| US5885527A (en) | 1992-05-21 | 1999-03-23 | Biosite Diagnostics, Inc. | Diagnostic devices and apparatus for the controlled movement of reagents without membrances |
| US5922615A (en) | 1990-03-12 | 1999-07-13 | Biosite Diagnostics Incorporated | Assay devices comprising a porous capture membrane in fluid-withdrawing contact with a nonabsorbent capillary network |
| US5939272A (en) | 1989-01-10 | 1999-08-17 | Biosite Diagnostics Incorporated | Non-competitive threshold ligand-receptor assays |
| US5947124A (en) | 1997-03-11 | 1999-09-07 | Biosite Diagnostics Incorporated | Diagnostic for determining the time of a heart attack |
| WO2000028004A1 (en) | 1998-11-10 | 2000-05-18 | The University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| US6113855A (en) | 1996-11-15 | 2000-09-05 | Biosite Diagnostics, Inc. | Devices comprising multiple capillarity inducing surfaces |
| US6143576A (en) | 1992-05-21 | 2000-11-07 | Biosite Diagnostics, Inc. | Non-porous diagnostic devices for the controlled movement of reagents |
| WO2000073316A2 (en) | 1999-05-28 | 2000-12-07 | Cell Genesys, Inc. | Virus immunologic determinants |
| WO2001023001A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Rapid peg-modification |
| US6248516B1 (en) | 1988-11-11 | 2001-06-19 | Medical Research Council | Single domain ligands, receptors comprising said ligands methods for their production, and use of said ligands and receptors |
| WO2004112727A2 (en) | 2003-06-19 | 2004-12-29 | Avigen, Inc. | Aav virions with decreased immunoreactivity and uses therefor |
| WO2005005610A2 (en) | 2003-06-30 | 2005-01-20 | The Regents Of The University Of California | Mutant adeno-associated virus virions and methods of use thereof |
| WO2005072364A2 (en) | 2004-01-27 | 2005-08-11 | University Of Florida | A modified baculovirus expression system for production of pseudotyped raav vector |
| US6982321B2 (en) | 1986-03-27 | 2006-01-03 | Medical Research Council | Altered antibodies |
| US7087409B2 (en) | 1997-12-05 | 2006-08-08 | The Scripps Research Institute | Humanization of murine antibody |
| WO2008154251A2 (en) * | 2007-06-07 | 2008-12-18 | Genentech, Inc. | C3b antibodies and methods for the prevention and treatment of complement- associated disorders |
| US7527791B2 (en) | 2004-03-31 | 2009-05-05 | Genentech, Inc. | Humanized anti-TGF-beta antibodies |
| US7846445B2 (en) | 2005-09-27 | 2010-12-07 | Amunix Operating, Inc. | Methods for production of unstructured recombinant polymers and uses thereof |
| US7855279B2 (en) | 2005-09-27 | 2010-12-21 | Amunix Operating, Inc. | Unstructured recombinant polymers and uses thereof |
| WO2013152024A1 (en) * | 2012-04-03 | 2013-10-10 | Novelmed Therapeutics, Inc | Humanized and chimeric anti-factor c3 antibodies and uses thereof |
| US8568992B2 (en) | 2001-12-03 | 2013-10-29 | Amgen Fremont Inc. | Antibody categorization based on binding characteristics |
| US8673860B2 (en) | 2009-02-03 | 2014-03-18 | Amunix Operating Inc. | Extended recombinant polypeptides and compositions comprising same |
| US8933197B2 (en) | 2007-08-15 | 2015-01-13 | Amunix Operating Inc. | Compositions comprising modified biologically active polypeptides |
| US10172953B2 (en) | 2012-02-27 | 2019-01-08 | Amunix Operating Inc. | XTEN conjugate compositions and methods of making same |
| US20190038724A1 (en) | 2015-05-01 | 2019-02-07 | Spark Therapeutics, Inc. | Adeno-Associated Virus-Mediated CRISPR-Cas9 Treatment of Ocular Disease |
| US20190078099A1 (en) | 2016-03-30 | 2019-03-14 | Spark Therapeutics, Inc. | Cell line for recombinant protein and/or viral vector production |
| US10494646B2 (en) | 2015-02-02 | 2019-12-03 | Meiragtx Uk Ii Limited | Regulation of gene expression by aptamer-mediated modulation of alternative splicing |
| US20210363192A1 (en) | 2018-04-27 | 2021-11-25 | Spark Therapeutics, Inc. | Engineered aav capsids with increased tropism and aav vectors comprising the engineered capsids and methods of making and using same |
| US20210371877A1 (en) | 2017-12-18 | 2021-12-02 | Spark Therapeutics, Inc. | Adeno-associated virus (aav) vector lipid nanoparticle compositions and methods of use |
| US20220011308A1 (en) | 2018-11-16 | 2022-01-13 | Spark Therapeutics, Inc. | In vitro assay for detecting enhancers and inhibitors of adeno associated virus (aav) vector transduction and/or detecting or quantitating anti-aav binding antibodies |
| US20220025396A1 (en) | 2018-05-07 | 2022-01-27 | Spark Therapeutics, Inc. | Plasmid free aav vector producing cell lines |
| WO2023212298A1 (en) * | 2022-04-29 | 2023-11-02 | Broadwing Bio Llc | Bispecific antibodies and methods of treating ocular disease |
-
2024
- 2024-04-29 WO PCT/US2024/026808 patent/WO2024227154A1/en active Pending
Patent Citations (53)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US4828981A (en) | 1983-08-24 | 1989-05-09 | Synbiotics Corporation | Immunoassays for determining Dirofilaria immitis infection using antiidiotype monoclonal antibody reagents |
| US5034506A (en) | 1985-03-15 | 1991-07-23 | Anti-Gene Development Group | Uncharged morpholino-based polymers having achiral intersubunit linkages |
| US6982321B2 (en) | 1986-03-27 | 2006-01-03 | Medical Research Council | Altered antibodies |
| US6248516B1 (en) | 1988-11-11 | 2001-06-19 | Medical Research Council | Single domain ligands, receptors comprising said ligands methods for their production, and use of said ligands and receptors |
| US5939272A (en) | 1989-01-10 | 1999-08-17 | Biosite Diagnostics Incorporated | Non-competitive threshold ligand-receptor assays |
| US5679526A (en) | 1989-01-10 | 1997-10-21 | Biosite Diagnostics Incorporated | Threshold ligand-receptor assay |
| EP0404097A2 (en) | 1989-06-22 | 1990-12-27 | BEHRINGWERKE Aktiengesellschaft | Bispecific and oligospecific, mono- and oligovalent receptors, production and applications thereof |
| US5922615A (en) | 1990-03-12 | 1999-07-13 | Biosite Diagnostics Incorporated | Assay devices comprising a porous capture membrane in fluid-withdrawing contact with a nonabsorbent capillary network |
| US5480792A (en) | 1990-09-14 | 1996-01-02 | Biosite Diagnostics, Inc. | Antibodies to complexes of ligand receptors and ligands and their utility in ligand-receptor assays |
| US5985579A (en) | 1990-09-14 | 1999-11-16 | Biosite Diagnostics, Inc. | Antibodies to complexes of ligand receptors and ligands and their utility in ligand-receptor assays |
| US5525524A (en) | 1991-04-10 | 1996-06-11 | Biosite Diagnostics, Inc. | Crosstalk inhibitors and their uses |
| US5851776A (en) | 1991-04-12 | 1998-12-22 | Biosite Diagnostics, Inc. | Conjugates and assays for simultaneous detection of multiple ligands |
| US5821337A (en) | 1991-06-14 | 1998-10-13 | Genentech, Inc. | Immunoglobulin variants |
| WO1993001161A1 (en) | 1991-07-11 | 1993-01-21 | Pfizer Limited | Process for preparing sertraline intermediates |
| US5885527A (en) | 1992-05-21 | 1999-03-23 | Biosite Diagnostics, Inc. | Diagnostic devices and apparatus for the controlled movement of reagents without membrances |
| US6143576A (en) | 1992-05-21 | 2000-11-07 | Biosite Diagnostics, Inc. | Non-porous diagnostic devices for the controlled movement of reagents |
| US6019944A (en) | 1992-05-21 | 2000-02-01 | Biosite Diagnostics, Inc. | Diagnostic devices and apparatus for the controlled movement of reagents without membranes |
| US5824799A (en) | 1993-09-24 | 1998-10-20 | Biosite Diagnostics Incorporated | Hybrid phthalocyanine derivatives and their uses |
| US6113855A (en) | 1996-11-15 | 2000-09-05 | Biosite Diagnostics, Inc. | Devices comprising multiple capillarity inducing surfaces |
| WO1998022607A1 (en) | 1996-11-22 | 1998-05-28 | Rhone-Poulenc Rorer S.A. | Method for producing recombinant virus |
| US5947124A (en) | 1997-03-11 | 1999-09-07 | Biosite Diagnostics Incorporated | Diagnostic for determining the time of a heart attack |
| WO1999006562A1 (en) | 1997-07-31 | 1999-02-11 | Chiron Corporation | Method enabling readministration of aav vector via immunosuppression of host |
| US7087409B2 (en) | 1997-12-05 | 2006-08-08 | The Scripps Research Institute | Humanization of murine antibody |
| WO2000028004A1 (en) | 1998-11-10 | 2000-05-18 | The University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| WO2000073316A2 (en) | 1999-05-28 | 2000-12-07 | Cell Genesys, Inc. | Virus immunologic determinants |
| WO2001023001A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Rapid peg-modification |
| US8568992B2 (en) | 2001-12-03 | 2013-10-29 | Amgen Fremont Inc. | Antibody categorization based on binding characteristics |
| WO2004112727A2 (en) | 2003-06-19 | 2004-12-29 | Avigen, Inc. | Aav virions with decreased immunoreactivity and uses therefor |
| WO2005005610A2 (en) | 2003-06-30 | 2005-01-20 | The Regents Of The University Of California | Mutant adeno-associated virus virions and methods of use thereof |
| WO2005072364A2 (en) | 2004-01-27 | 2005-08-11 | University Of Florida | A modified baculovirus expression system for production of pseudotyped raav vector |
| US7527791B2 (en) | 2004-03-31 | 2009-05-05 | Genentech, Inc. | Humanized anti-TGF-beta antibodies |
| US7855279B2 (en) | 2005-09-27 | 2010-12-21 | Amunix Operating, Inc. | Unstructured recombinant polymers and uses thereof |
| US8492530B2 (en) | 2005-09-27 | 2013-07-23 | Amunix Operating Inc. | Unstructured recombinant polymers and compositions comprising same |
| US7846445B2 (en) | 2005-09-27 | 2010-12-07 | Amunix Operating, Inc. | Methods for production of unstructured recombinant polymers and uses thereof |
| US9938331B2 (en) | 2005-09-27 | 2018-04-10 | Amunix Operating Inc. | Biologically active proteins having increased in vivo and/or in vitro stability |
| WO2008154251A2 (en) * | 2007-06-07 | 2008-12-18 | Genentech, Inc. | C3b antibodies and methods for the prevention and treatment of complement- associated disorders |
| US8933197B2 (en) | 2007-08-15 | 2015-01-13 | Amunix Operating Inc. | Compositions comprising modified biologically active polypeptides |
| US8673860B2 (en) | 2009-02-03 | 2014-03-18 | Amunix Operating Inc. | Extended recombinant polypeptides and compositions comprising same |
| US9371369B2 (en) | 2009-02-03 | 2016-06-21 | Amunix Operating Inc. | Extended recombinant polypeptides and compositions comprising same |
| US9926351B2 (en) | 2009-02-03 | 2018-03-27 | Amunix Operating Inc. | Extended recombinant polypeptides and compositions comprising same |
| US10961287B2 (en) | 2009-02-03 | 2021-03-30 | Amunix Pharmaceuticals, Inc | Extended recombinant polypeptides and compositions comprising same |
| US10953073B2 (en) | 2012-02-27 | 2021-03-23 | Amunix Pharmaceuticals, Inc. | XTEN conjugate compositions and methods of making same |
| US10172953B2 (en) | 2012-02-27 | 2019-01-08 | Amunix Operating Inc. | XTEN conjugate compositions and methods of making same |
| WO2013152024A1 (en) * | 2012-04-03 | 2013-10-10 | Novelmed Therapeutics, Inc | Humanized and chimeric anti-factor c3 antibodies and uses thereof |
| US10494646B2 (en) | 2015-02-02 | 2019-12-03 | Meiragtx Uk Ii Limited | Regulation of gene expression by aptamer-mediated modulation of alternative splicing |
| US20190038724A1 (en) | 2015-05-01 | 2019-02-07 | Spark Therapeutics, Inc. | Adeno-Associated Virus-Mediated CRISPR-Cas9 Treatment of Ocular Disease |
| US20190078099A1 (en) | 2016-03-30 | 2019-03-14 | Spark Therapeutics, Inc. | Cell line for recombinant protein and/or viral vector production |
| US20210371877A1 (en) | 2017-12-18 | 2021-12-02 | Spark Therapeutics, Inc. | Adeno-associated virus (aav) vector lipid nanoparticle compositions and methods of use |
| US20210363192A1 (en) | 2018-04-27 | 2021-11-25 | Spark Therapeutics, Inc. | Engineered aav capsids with increased tropism and aav vectors comprising the engineered capsids and methods of making and using same |
| US20220025396A1 (en) | 2018-05-07 | 2022-01-27 | Spark Therapeutics, Inc. | Plasmid free aav vector producing cell lines |
| US20220011308A1 (en) | 2018-11-16 | 2022-01-13 | Spark Therapeutics, Inc. | In vitro assay for detecting enhancers and inhibitors of adeno associated virus (aav) vector transduction and/or detecting or quantitating anti-aav binding antibodies |
| WO2023212298A1 (en) * | 2022-04-29 | 2023-11-02 | Broadwing Bio Llc | Bispecific antibodies and methods of treating ocular disease |
Non-Patent Citations (67)
| Title |
|---|
| "Biological Sequence Analysis: Probabilistic Models of Proteins and Nucleic Acids", 2009, CAMBRIDGE UNIVERSITY PRESS |
| "Immunobiology", 2001, GARLAND PUBLISHING |
| "Remington: The Science and Practice of Pharmacy", 1 May 2005, LIPPINCOTT WILLIAMS & WILKINS |
| "Short Protocols in Molecular Biology", 2002, JOHN WILEY & SONS, INC. |
| ADAMCZYK ET AL., ANAL. CHIM. ACTA, vol. 579, no. 1, 2006, pages 61 - 67 |
| ALBERT L. LEHNINGER: "Principles of Biochemistry", 1982, WORTH PUB., pages: 793 - 800 |
| ALMAGROFRANSSON, FRONT. BIOSCI., vol. 13, 2008, pages 1619 - 1633 |
| ALTSCHUL ET AL., J. MOLECULAR BIOL., vol. 215, no. 3, 1990, pages 403 - 410 |
| ALTSCHUL ET AL., NUCLEIC ACIDS RES., vol. 25, no. 17, 1997, pages 3389 - 3402 |
| BATES APOWER CA., ANTIBODIES (BASEL)., vol. 8, no. 2, 2019, pages 28 |
| BEIGERT ET AL., PROC. NATL. ACAD. SCI. USA, vol. 106, no. 10, 2009, pages 3770 - 3775 |
| BORDET, T.BEHAR-COHEN, F.: "Ocular gene therapies in clinical practice: viral vectors and nonviral alternatives", DRUG DISCOVERY TODAY, vol. 24, August 2019 (2019-08-01), pages 1685 - 1693, XP085782111, DOI: 10.1016/j.drudis.2019.05.038 |
| BRAASCHCOREY, BIOCHEMISTRY, vol. 41, no. 14, 2002, pages 4503 - 4510 |
| BRAITBARD ET AL., PROTEOME SCI., vol. 4, 2006, pages 12 |
| CARMELIET ET AL., NATURE, vol. 407, 2000, pages 249 - 257 |
| CLACKSON ET AL., NATURE, vol. 352, 1991, pages 624 - 628 |
| COFFIN ET AL.: "Algorithms on Strings, Trees and Sequences", 1997, COLD SPRING HARBOUR LABORATORY PRESS, pages: 758 - 763 |
| DALL'ACQUA ET AL., J. IMMUNOL., vol. 169, 2002, pages 5171 - 5180 |
| DALL'ACQUA ET AL., METHODS, vol. 36, 2005, pages 61 - 68 |
| GOEDDEL: "Gene Expression Technology: Methods in Enzymology", vol. 185, 1990, ACADEMIC PRESS |
| HOLLIGER ET AL., NAT. BIOTECH., vol. 23, no. 9, 2005, pages 1126 - 1129 |
| HOLLINGER ET AL., PROC. NATL. ACAD. SCI. USA, vol. 90, 1993, pages 6444 - 6448 |
| HUDSON ET AL., NAT. MED., vol. 9, 2003, pages 129 - 134 |
| INOUE ET AL., J. VIROL., vol. 72, 1998, pages 7024 |
| JOOSTEN ET AL., MICROBIAL CELL FACTORIES, vol. 2, 2003 |
| KANG, T.H.JUNG, S.T., EXP MOL MED, vol. 51, 2019, pages 1 - 9 |
| KELLEY, R.F.MENG, Y.G.LIU ET AL., J BIOL CHEM., vol. 289, 2014, pages 3571 - 90 |
| KENNETH J. KATSCHKE ET AL: "Structural and Functional Analysis of a C3b-specific Antibody That Selectively Inhibits the Alternative Pathway of Complement", JOURNAL OF BIOLOGICAL CHEMISTRY, vol. 284, no. 16, 17 April 2009 (2009-04-17), US, pages 10473 - 10479, XP055568372, ISSN: 0021-9258, DOI: 10.1074/jbc.M809106200 * |
| KITTS ET AL., BIOTECHNIQUES, vol. 14, 1993, pages 810 - 817 |
| KLIMKA ET AL., BR. J. CANCER, vol. 83, 2000, pages 252 - 260 |
| KOHLERMILSTEIN, EUR. J. IMMUNOL., vol. 5, 1976, pages 511 - 519 |
| LAZAR ET AL., PROC. NATL ACAD. SCI. USA, vol. 103, 2006, pages 4005 - 4010 |
| LEWIS ET AL., EM BO J, vol. 11, no. 8, 1992, pages 3053 - 3058 |
| LEWISEMERMAN, J VIROL, vol. 68, no. 1, 1994, pages 510 - 516 |
| LI ET AL., PROC NATL ACAD SCI U S A., vol. 114, 2017, pages 3485 - 90 |
| LIN ET AL., PROC NATL ACAD SCI U S A., vol. 112, 2015, pages 10611 - 6 |
| LONBERG, HANDB. EXP. PHARMACOL., vol. 181, 2008, pages 69 - 97 |
| LONBERG, NAT. BIOTECHNOL., vol. 23, no. 9, 2005, pages 1117 - 25 |
| LU, R.HWANG, Y.LIU, I. ET AL.: "Development of therapeutic antibodies for the treatment of diseases", J BIOMED SCI, vol. 27, 2020, pages 1, Retrieved from the Internet <URL:https://doi.org/10.1186/s12929-019-0592-z> |
| LUCKLOW ET AL., J. VIROL., vol. 67, 1993, pages 4566 - 4579 |
| LUCKLOW, CURR. OPIN. BIOTECHNOL., vol. 4, 1993, pages 564 - 572 |
| MARKS ET AL., J. MOL. BIOL., vol. 222, 1991, pages 581 - 597 |
| MIMOTO ET AL., CURR. PHARM. BIOTECHNOL., vol. 17, 2016, pages 1298 - 1314 |
| MONNET ET AL., MABS, vol. 6, 2014, pages 422 - 36 |
| MONNIER ET AL., ANTIBODIES, vol. 2, no. 2, 2013, pages 193 - 208 |
| MORRISON ET AL., PROC. NATL. ACAD. SCI. USA., vol. 81, 1984, pages 6851 - 6855 |
| NELSON, A.L., MABS, vol. 2, no. 1, January 2010 (2010-01-01), pages 77 - 83 |
| NORDSTROM ET AL., BREAST CANCER RES., vol. 13, 2011, pages R123 |
| PADLAN, MOL. IMMUNOL., vol. 28, 1991, pages 489 - 498 |
| QUEEN ET AL., PROC. NAT'L ACAD. SCI. USA, vol. 86, 1989, pages 10029 - 10033 |
| RICHARDS ET AL., MOL. CANCER THER., vol. 7, 2008, pages 2517 - 2527 |
| RIECHMANN ET AL., NATURE, vol. 332, 1988, pages 323 - 329 |
| RUDIKOFF S ET AL: "Single amino acid substitution altering antigen-binding specificity", PROCEEDINGS OF THE NATIONAL ACADEMY OF SCIENCES, NATIONAL ACADEMY OF SCIENCES, vol. 79, no. 6, 1 March 1982 (1982-03-01), pages 1979 - 1983, XP002683593, ISSN: 0027-8424, DOI: 10.1073/PNAS.79.6.1979 * |
| SAFDARI ET AL., MOL MED., vol. 22, 2016, pages 258 - 270 |
| SAMBROOK ET AL.: "Molecular Cloning, a Laboratory Manual", 2012, COLD SPRING HARBOR PRESS |
| SAMULSKI ET AL., J. VIROLOGY, vol. 63, 1989, pages 3822 |
| SAUNDERS, K.O., FRONT IMMUNOL., vol. 10, 2019, pages 1296 |
| SCHULZSCHIRMER: "Principles of Protein Structure", 1979, SPRINGER-VERLAG |
| SODING, BIOINFORMATICS, vol. 21, no. 7, 2005, pages 951 - 960 |
| SOUTO, E.B. ET AL.: "Advanced Formulation Approaches for Ocular Drug Delivery: State-Of-The-Art and Recent Patents", PHARMACEUTICS, vol. 11, no. 9, September 2019 (2019-09-01), pages 460 |
| URLAUB ET AL., PROC. NATL. ACAD. SCI. USA, vol. 97, 1980, pages 4216 - 4220 |
| VIDARSSON ET AL., FRONTIERS IN IMMUNOLOGY, vol. 5, 2014, pages 520 |
| WAHLESTEDT ET AL., PROC. NATL. ACAD. SCI. U.S.A., vol. 97, 2000, pages 5633 - 5638 |
| WANG, J. AM. CHEM. SOC., vol. 122, 2000, pages 8595 - 8602 |
| WINKLER K ET AL: "Changing the antigen binding specificity by single point mutations of an anti-p24 (HIV-1) antibody", THE JOURNAL OF IMMUNOLOGY, WILLIAMS & WILKINS CO, US, vol. 165, no. 8, 15 October 2000 (2000-10-15), pages 4505 - 4514, XP002579393, ISSN: 0022-1767 * |
| XIAO ET AL., J. VIROLOGY, vol. 72, 1998, pages 2224 |
| ZALEVSKY ET AL., NAT. BIOTECHNOL., vol. 28, 2010, pages 157 - 159 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2022101682A (en) | Antibodies and methods of use targeting HIV gp120 | |
| US11008393B2 (en) | Pharmaceutical compositions comprising a polypeptide that binds to IL-6 | |
| JP2024501811A (en) | Multispecific antibody with specificity for IL-4R and IL-31 | |
| CN106565840B (en) | Antibody against hepatitis B surface antigen and its use | |
| US20250346653A1 (en) | Bispecific antibodies and method of treating ocular disease | |
| JP2014526886A (en) | Antibodies cross-reactive with macrophage migration inhibitory factor (MIF) and D-dopachrome tomerase (D-DT) | |
| JP2025013590A (en) | Antibodies against feline McDonough sarcoma (FMS)-like tyrosine kinase 3 receptor ligand (FLT3L) and their use for treating autoimmune and inflammatory diseases - Patent Application 20070123333 | |
| US20250289873A1 (en) | Complement factor h related 4-specific antibodies and uses thereof | |
| WO2024227154A1 (en) | Complement component 3 (c3)-specific antibodies and uses thereof | |
| JP2022540859A (en) | Novel BSSL antibody | |
| US20250296996A1 (en) | Angiopoietin-related protein 7-specific antibodies and uses thereof | |
| CA3250574A1 (en) | Bispecific antibodies and methods of treating ocular disease | |
| OA21947A (en) | Angiopoietin-related protein 7-specific antibodies and uses thereof. | |
| WO2024259273A2 (en) | Mhc class i chain-related protein a (mica)-specific antibodies and uses thereof | |
| CN121532429A (en) | MHC class I chain-associated protein A (MICA) specific antibodies and their uses | |
| HK40041682A (en) | Antibodies to feline mcdonough sarcoma (fms)-like tyrosine kinase 3 receptor ligand (flt3l) and uses thereof for treating autoimmune and inflammatory diseases |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24727125 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |