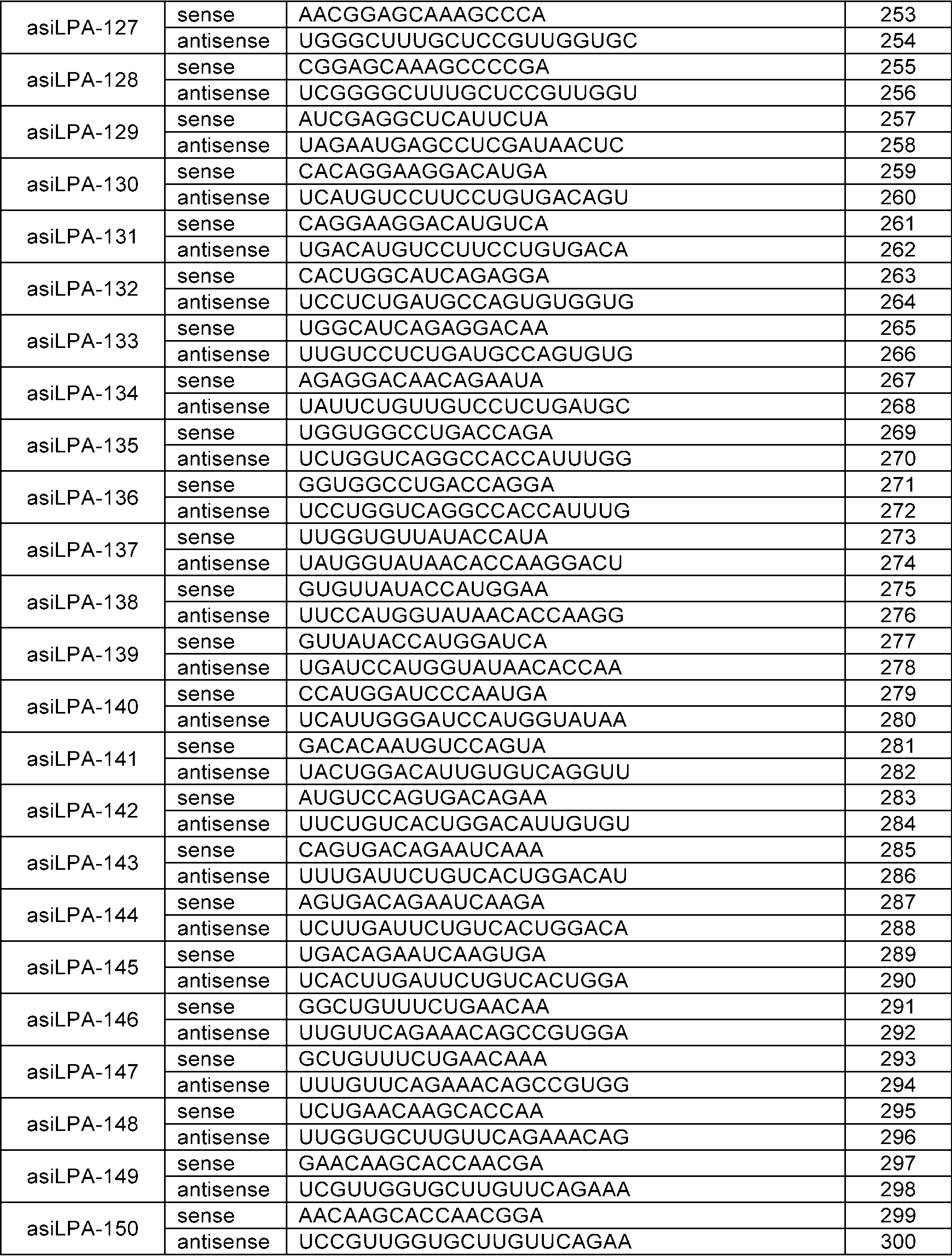
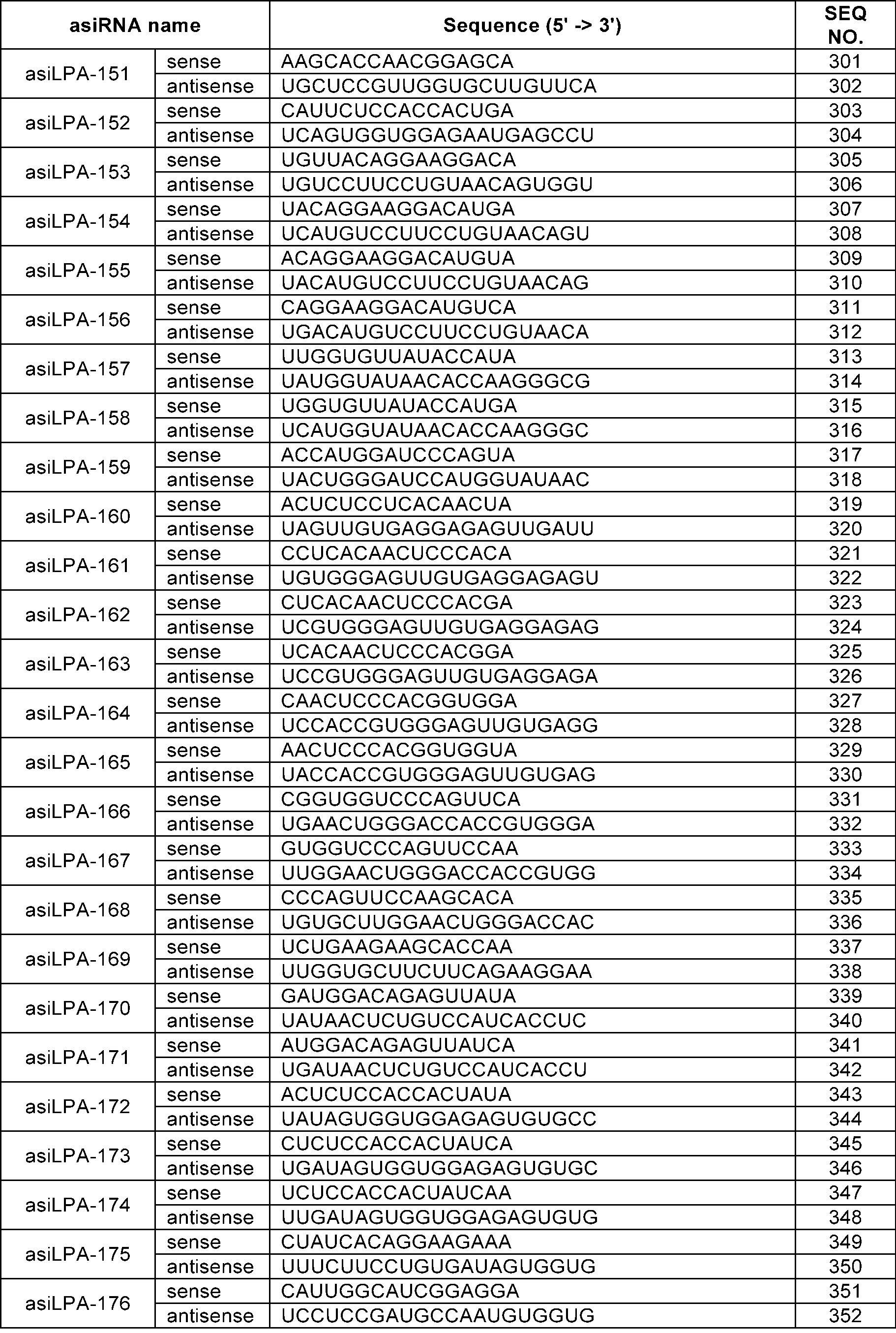
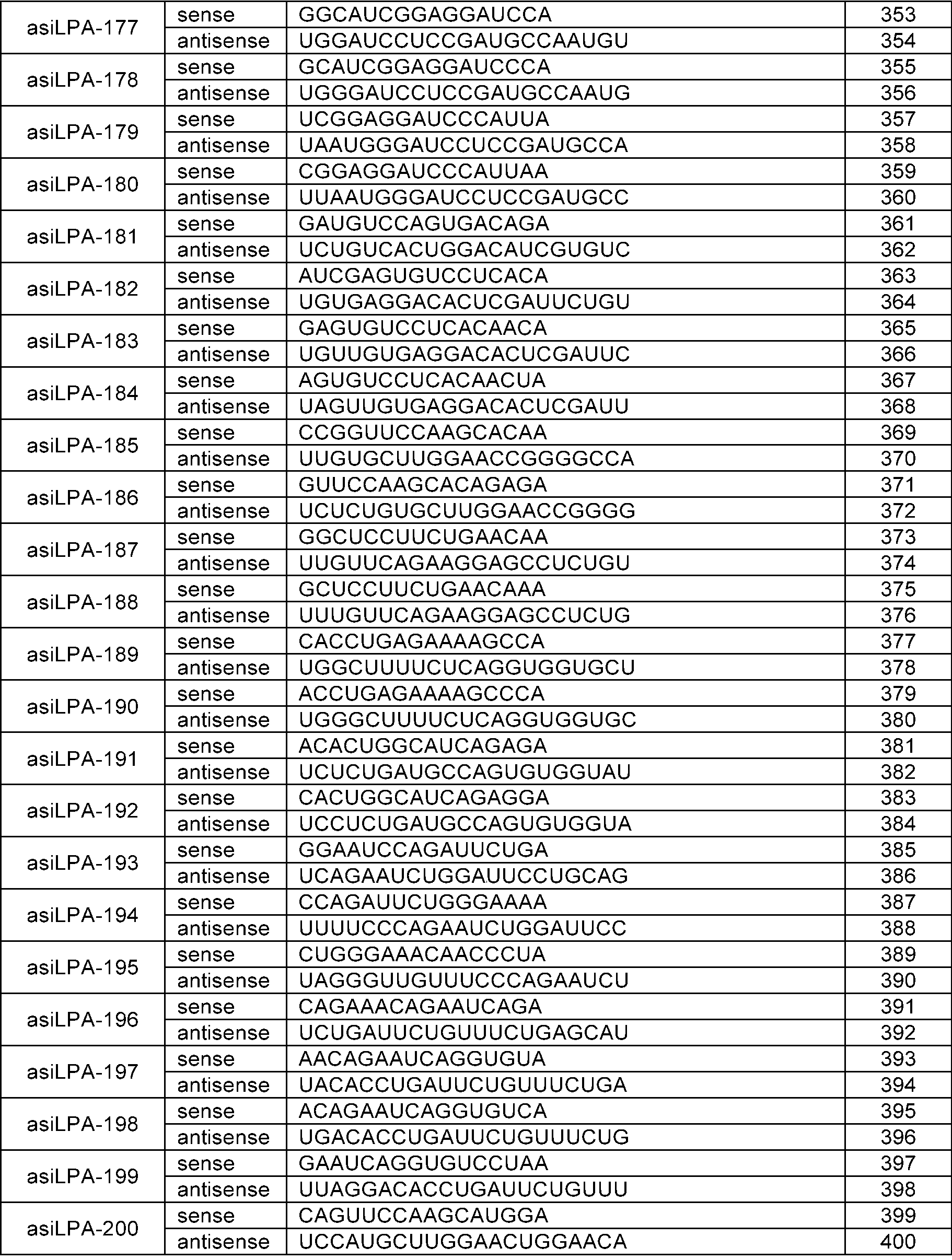
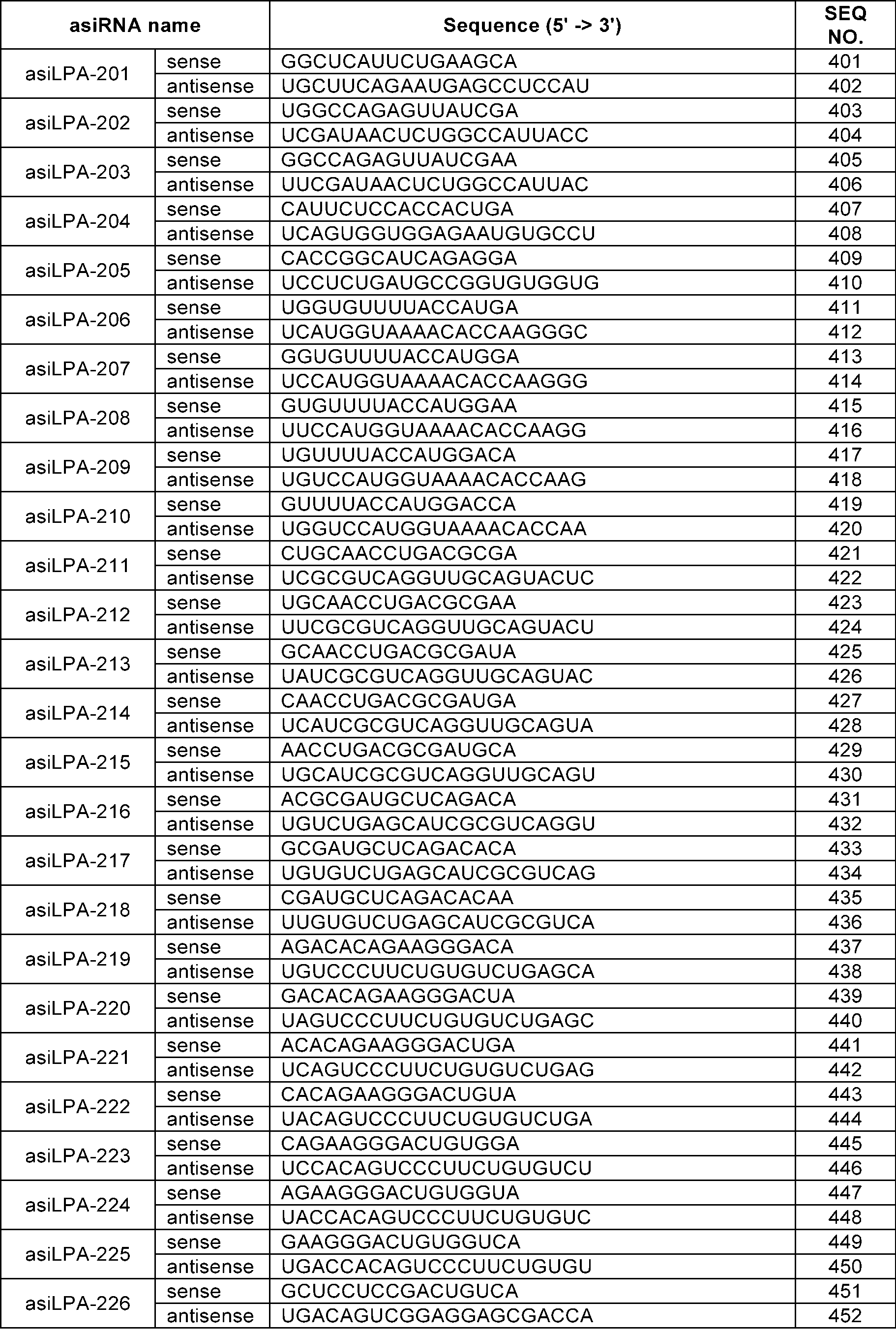
WO2024210530A1 - RNAi AGENTS TARGETING LPA GENE AND USE THEREOF - Google Patents
RNAi AGENTS TARGETING LPA GENE AND USE THEREOF Download PDFInfo
- Publication number
- WO2024210530A1 WO2024210530A1 PCT/KR2024/004361 KR2024004361W WO2024210530A1 WO 2024210530 A1 WO2024210530 A1 WO 2024210530A1 KR 2024004361 W KR2024004361 W KR 2024004361W WO 2024210530 A1 WO2024210530 A1 WO 2024210530A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- rnai agent
- lpa
- antisense strand
- sense strand
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/549—Sugars, nucleosides, nucleotides or nucleic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/06—Antihyperlipidemics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/14—Type of nucleic acid interfering nucleic acids [NA]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/312—Phosphonates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/312—Phosphonates
- C12N2310/3125—Methylphosphonates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/31—Chemical structure of the backbone
- C12N2310/315—Phosphorothioates
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/321—2'-O-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/32—Chemical structure of the sugar
- C12N2310/322—2'-R Modification
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/351—Conjugate
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/352—Nature of the modification linked to the nucleic acid via a carbon atom
- C12N2310/3521—Methyl
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/352—Nature of the modification linked to the nucleic acid via a carbon atom
- C12N2310/3525—MOE, methoxyethoxy
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/30—Chemical structure
- C12N2310/35—Nature of the modification
- C12N2310/353—Nature of the modification linked to the nucleic acid via an atom other than carbon
- C12N2310/3533—Halogen
Definitions
- the present disclosure relates to an RNAi agent targeting an LPA gene and use thereof, more specifically, an RNAi agent including an antisense strand having sequence complementarity with an LPA mRNA sequence, and a sense strand having sequence complementarity with the antisense strand, and a pharmaceutical composition for preventing or treating cardiovascular disease, including the RNAi agent.
- cardiovascular diseases are the main cause of morbidity and mortality worldwide, and it has been reported that approximately 8.9 million deaths occurred due to cardiovascular diseases worldwide in 2019.
- the cardiovascular diseases refer to diseases of the circulatory system such as the heart and blood vessels, and arteriosclerosis is known as a direct and main cause.
- the arteriosclerosis is a disease in which cholesterol or triglyceride is accumulated in the blood vessels to narrow, harden, and block the blood vessels, and there is no sure way to reduce or eliminate the above symptoms.
- LPA or Lp(a) lipoprotein(a)
- ESC European Society of Cardiology
- EAS Atherosclerosis Society
- a genetically high level of LPA of 180 mg/dL or higher has been determined to have a similar risk of cardiovascular disease to heterozygous familial hypercholesterolemia, and a treatment target of less than 50 mg/dL has been suggested.
- a therapy for lowering LPA may be applied to treat diseases such as stroke, atherosclerosis, thrombosis and cardiovascular diseases such as coronary heart disease, and aortic stenosis.
- diseases such as stroke, atherosclerosis, thrombosis and cardiovascular diseases such as coronary heart disease, and aortic stenosis.
- a level of LPA has very limited variability due to diet, exercise, or other life cycle changes, a new technique capable of controlling an expression level of the LPA gene is needed.
- RNAi agent capable of specifically inhibiting expression of LPA mRNA by binding thereto, and thereby completed the present disclosure.
- An object of the present disclosure is to provide an RNAi agent that specifically inhibits expression of LPA.
- Another object of the present disclosure is to provide a pharmaceutical composition for preventing or treating cardiovascular diseases, including the RNAi agent, or a method of preventing or treating cardiovascular diseases including administering the pharmaceutical composition to a subject.
- an aspect provides an RNAi agent including: an antisense strand having sequence complementarity to an LPA mRNA sequence, and is 19 nt to 21 nt in length; and a sense strand having sequence complementarity to the antisense strand, and is 15 nt to 17 nt in length, wherein the 5' end of the antisense strand and the 3' end of the sense strand form a blunt end.
- Another aspect provides a pharmaceutical composition including the RNAi agent as an active ingredient for preventing or treating cardiovascular disease.
- Still another aspect provides a method of preventing or treating cardiovascular disease, including administering the RNAi agent to a subject.
- Still another aspect provides use of the RNAi agent for manufacturing a pharmaceutical product for preventing or treating cardiovascular disease.
- An aspect provides an RNAi agent including: an antisense strand having sequence complementarity to an LPA mRNA sequence, and is 19 nt to 21 nt in length; and a sense strand having sequence complementarity to the antisense strand, and is 15 nt to 17 nt in length, wherein the 5' end of the antisense strand and the 3' end of the sense strand form a blunt end.
- RNA interference refers to a mechanism for suppressing expression of a target gene by inducing degradation of mRNA of the target gene by introduction of double-stranded RNA (dsRNA) composed of a strand having a sequence homology to mRNA of a target gene and a strand having a sequence complementary to the aforementioned strand.
- dsRNA double-stranded RNA
- RNAi agents or “nucleic acid molecules inducing RNAi”, used herein, refers to any agent or nucleic acid molecule that is capable of inhibiting or downregulating gene expression or viral replication by mediating the RNA interference in a sequence-specific manner.
- the term may refer to both an individual nucleic acid molecule, a plurality of the nucleic acid molecules, or a pool of the nucleic acid molecules.
- the RNAi agent may be siRNA.
- small interfering RNA refers to a short double-stranded RNA (dsRNA) that sequence-specifically mediates efficient gene silencing.
- the term "gene”, used herein, should be considered in its broadest sense, and may encode a structural protein or a regulatory protein.
- the regulatory protein includes a transcription factor, a heat shock protein, or a protein involved in DNA/RNA replication, transcription, and/or translation.
- the target gene to be suppressed is inherent in the viral genome, and may be integrated into an animal genome or exist as an extrachromosomal component.
- antisense strand refers to a polynucleotide that is substantially or 100 % complementary to a target nucleic acid of interest, and may be complementary in whole or in part with, for example, messenger RNA (mRNA), non-mRNA RNA sequences (e.g., microRNA, piwiRNA, tRNA, rRNA and hnRNA), or coding or non-coding DNA sequences.
- mRNA messenger RNA
- non-mRNA RNA sequences e.g., microRNA, piwiRNA, tRNA, rRNA and hnRNA
- coding or non-coding DNA sequences coding or non-coding DNA sequences.
- ense strand refers to a polynucleotide having the same nucleic acid sequence as a target nucleic acid, or a polynucleotide in whole or in part the same with, for example, messenger RNA (mRNA), non-mRNA RNA sequences (e.g., microRNA, piwiRNA, tRNA, rRNA and hnRNA), or coding or non-coding DNA sequences.
- mRNA messenger RNA
- non-mRNA RNA sequences e.g., microRNA, piwiRNA, tRNA, rRNA and hnRNA
- coding or non-coding DNA sequences coding or non-coding DNA sequences.
- complementarity refers to a generally accepted meaning in the art.
- the term generally may refer to formation or presence of hydrogen bond(s) between one nucleic acid sequence and another nucleic acid sequence, either by traditional Watson-Crick bonding or other non-traditional types of bonding as described herein. Perfect complementarity may mean that all contiguous residues of a nucleic acid sequence hydrogen bond with the same number of contiguous residues in a second nucleic acid sequence.
- Partial complementarity within a nucleic acid molecule may include various mismatches or non-base-paired nucleotides (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more mismatches, e.g., 1 to 3 mismatches, non-nucleotide linkers, or non-base pair nucleotides).
- the partial complementarity may result in bulges, loops, overhangs, or blunt ends between a sense strand and an antisense strand of a nucleic acid molecule, or between an antisense strand of a nucleic acid molecule and a corresponding target nucleic acid molecule.
- blunt end refers to a generally accepted meaning in the art.
- the term may refer to an end of a double-stranded siRNA molecule that lacks overhanging nucleotides.
- the siRNA molecules described herein may be such that the 5'-end of an antisense strand and the 3'-end of a sense strand form a blunt end.
- lipoprotein(a) is a heterogeneous low-density lipoprotein particle mainly expressed in the liver, and is composed of apolipoprotein(a) (Apo(a)) linked to low density lipoprotein (LDL) by an ApoB polypeptide. It has been reported that a high level of LPA in the serum is not affected by diet or exercise, and is closely related to an increased risk of cardiovascular diseases by increasing possibility of developing atherosclerosis. In addition, with respect to diagnostic and prophylactic medicine, the high level of LPA in a patient's serum is known to be a significantly dominant, independent, and genetic risk factor for coronary heart disease and aortic stenosis.
- the LPA gene may be a potential therapeutic target for cardiovascular diseases.
- the LPA protein may be interpreted to include the naturally occurring wild-type LPA and functional variants thereof, and the sequence of the LPA protein or a gene encoding the same may be obtained from a known database such as GenBank of the US National Institutes of Health.
- expression refers to the generally accepted meaning in the art.
- the term may generally refer to a process by which a gene ultimately produces a protein.
- the expression includes, but is not limited to, transcription, splicing, post-transcriptional modification, or translation.
- an expression level may be determined or monitored by detection of an mRNA level or a protein level.
- the reduction may be, for example, at least 30 %, 35 %, 40 %, 45 %, 50 %, 55 %, 60 %, 65 %, 70 %, 75 %, 80 %, 85 %, 90 %, or 95 % or more, but it may be below the detection limit, depending on the detection or measurement method.
- siRNA is a small interfering RNA and is involved in RNA interference (RNAi).
- RNAi is an intracellular gene regulation mechanism first discovered in 1998 in Caenorthabditis elegans, and its mechanism of action is known to induce target gene degradation by an antisense strand, which is one of RNA double strands which are introduced into the cell, that complementarily binds to mRNA of a target gene, and RNAi has recently been one of the most popular candidate technologies for developing new drugs.
- siRNA is an effective method for directly regulating expression of a target gene, it is difficult to develop a therapeutic agent due to these issues.
- asymmetric shorter duplex siRNA (asiRNA) is an asymmetric RNAi-induced structure having a shorter double helix length compared to the 19+2 structure of siRNA in the art.
- asiRNA technology overcame issues such as off-target effect, saturation of RNAi mechanism, and immune response by TLR3, which are identified in the existing siRNA structure technology, and accordingly, it is possible to develop new RNAi drugs with low side effects.
- siRNA including a sense strand and an antisense strand complementary to the sense strand is presented in this example, and siRNA according to an example does not cause issues such as off-target effect, saturation of RNAi machinery, etc., and thereby, may inhibit expression of an LPA gene to a desired degree while stably maintaining high delivery efficiency.
- asymmetric siRNAs (asiRNAs) targeting LPA were designed and prepared, and after transfection of the asiRNAs into cells expressing LPA, nucleic acid molecules for inducing RNAi with excellent knockdown efficiency, i.e., LPA asiRNAs, were selected.
- the RNAi agent may be characterized in that the sense strand has a length of 15 nt to 17 nt, and the antisense strand has a length of 19 nt to 21 nt. More preferably, a length of the sense strand may be 16 nt, and a length of the antisense strand complementary thereto may be 19 nt, 20 nt, or 21 nt, but is not limited thereto.
- the 5'-end of the antisense strand and the 3'-end of the sense strand may form a blunt end.
- the 3' end of the antisense strand may include, for example, an overhang of 2 nt to 6 nt.
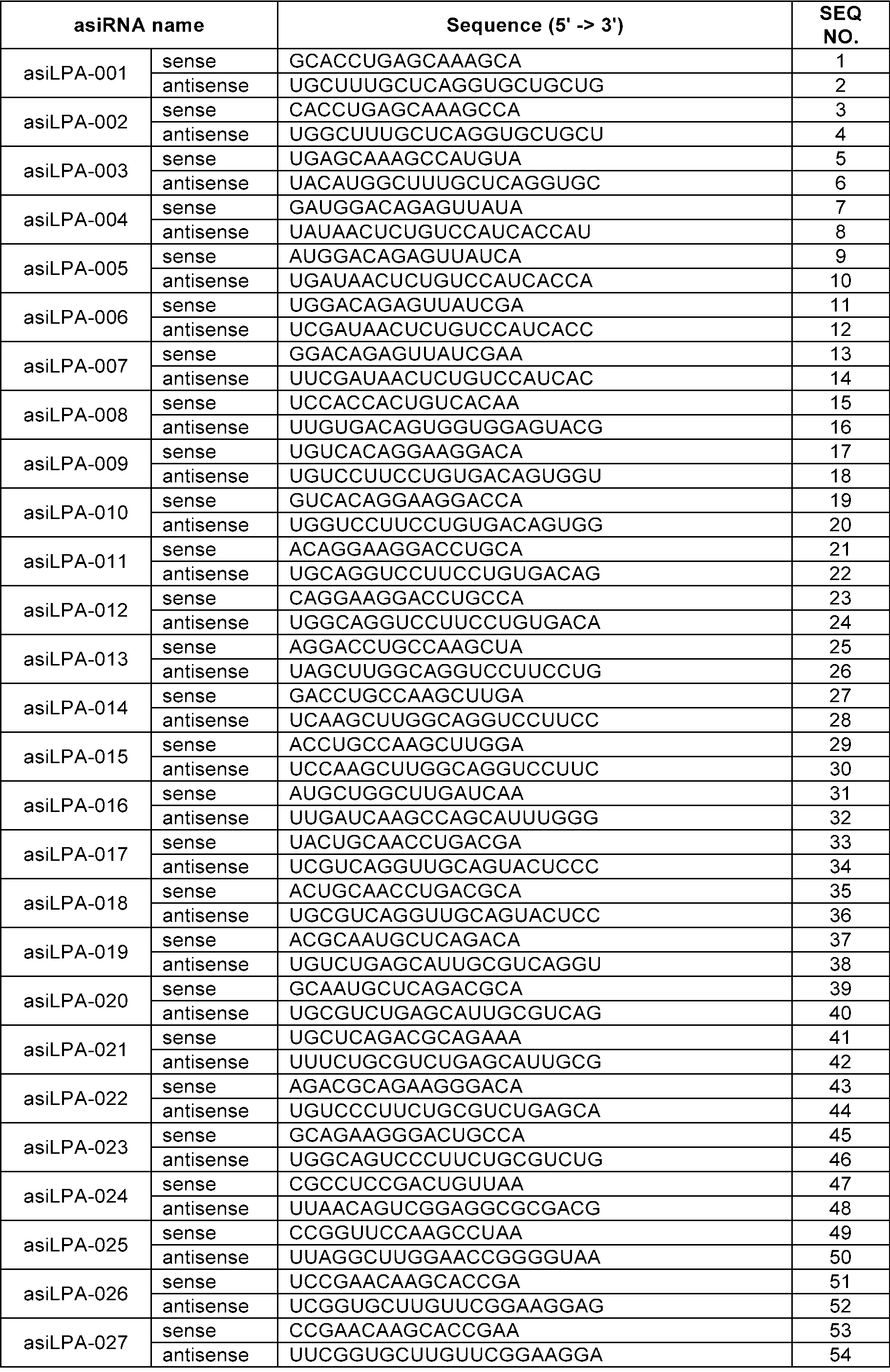
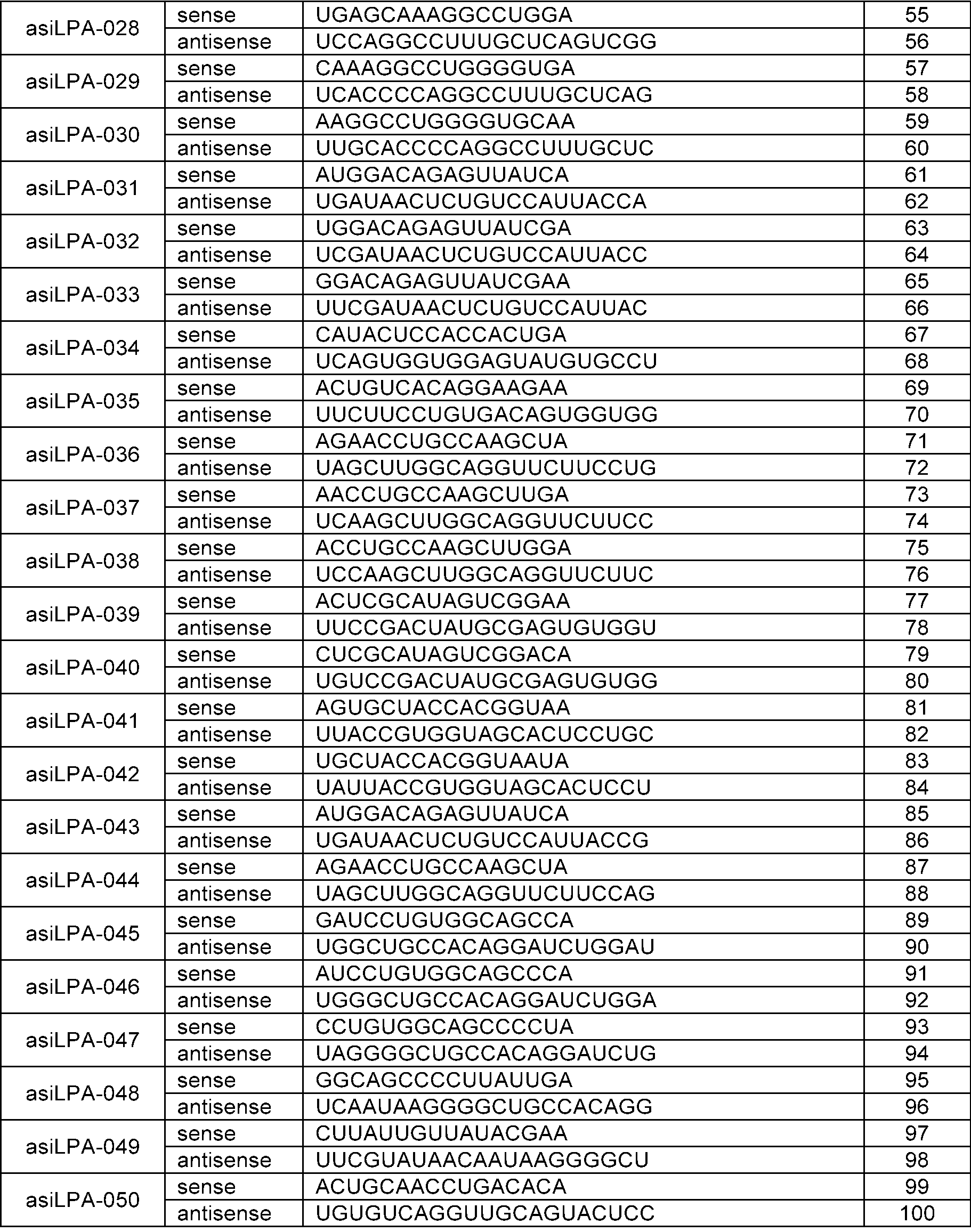
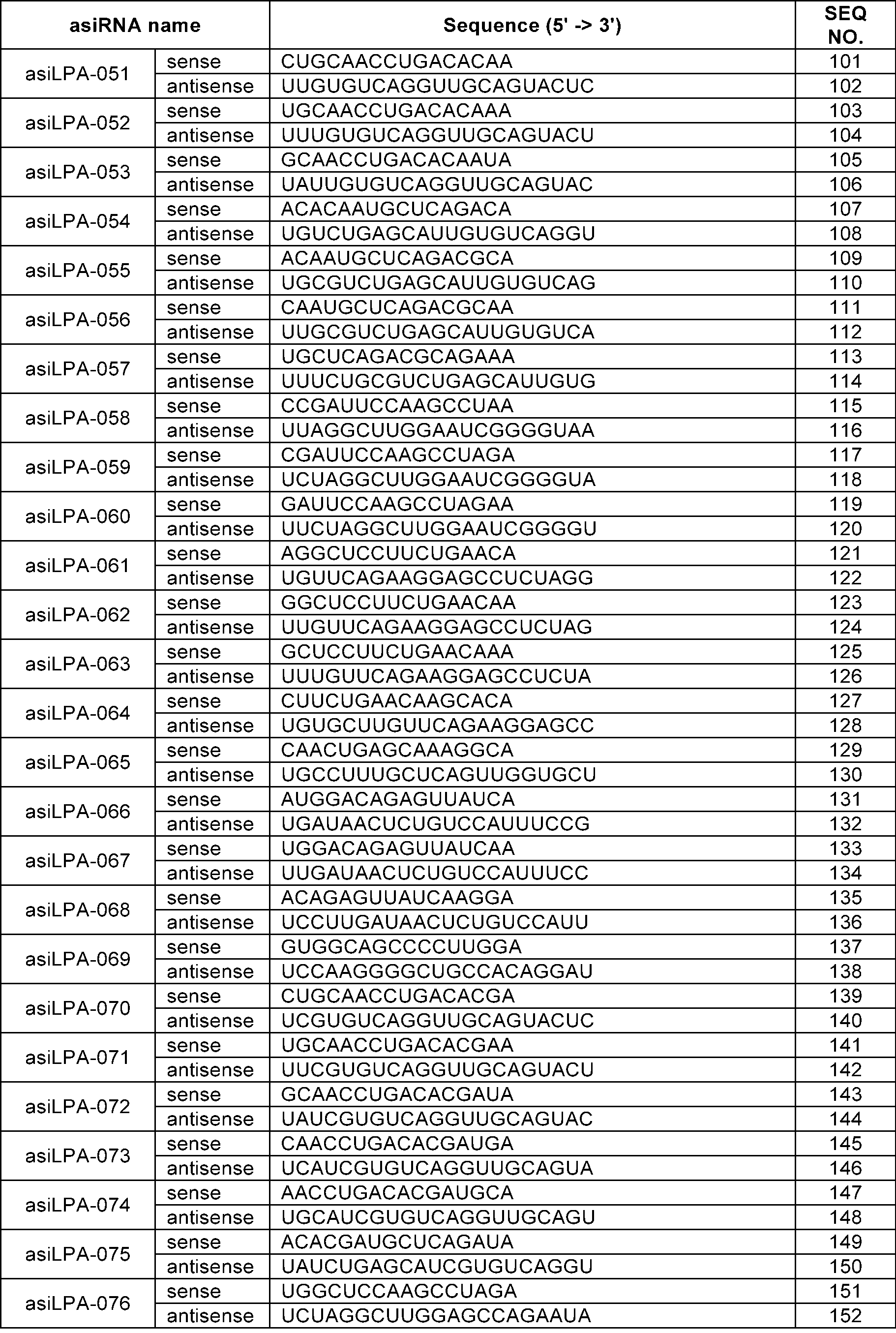
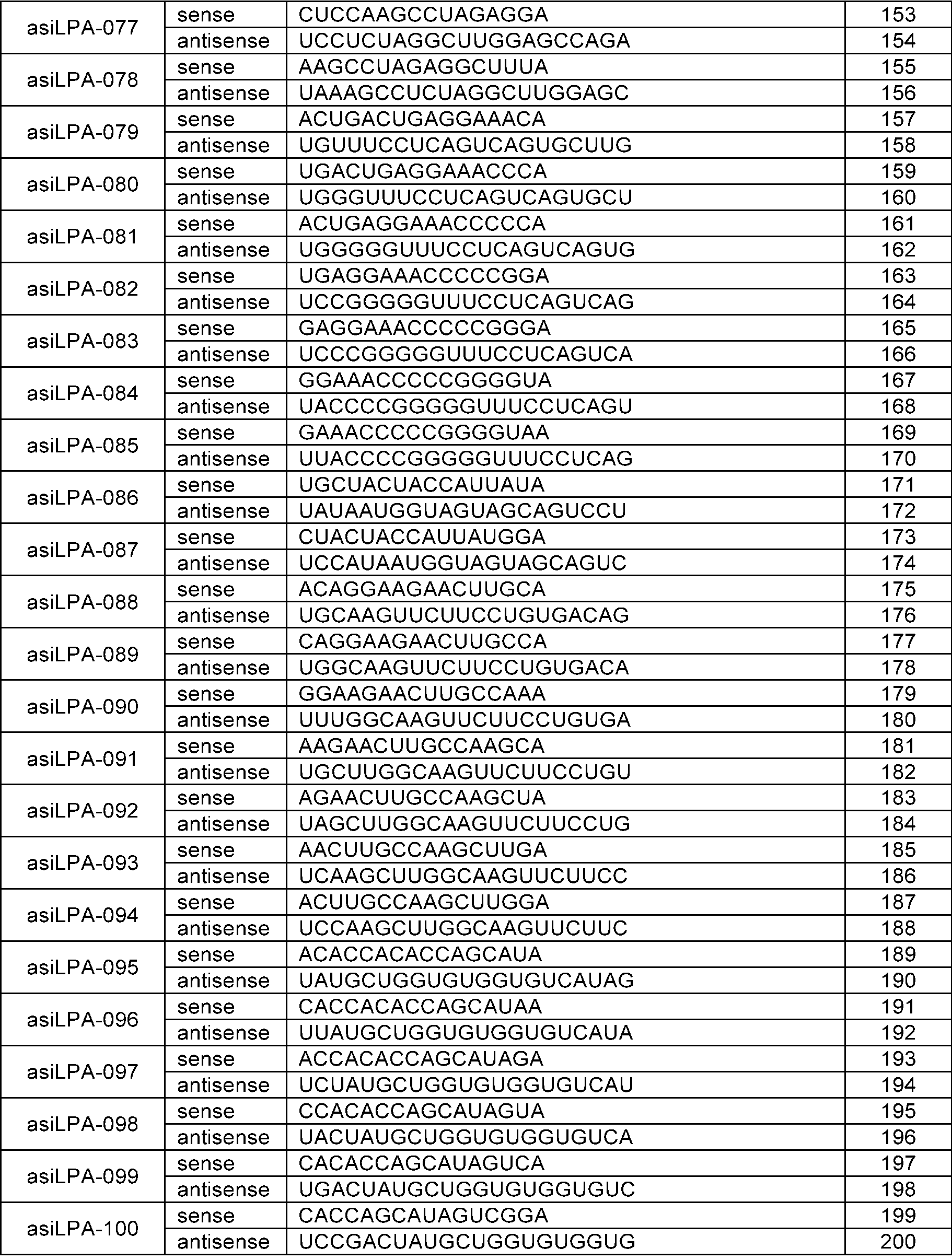
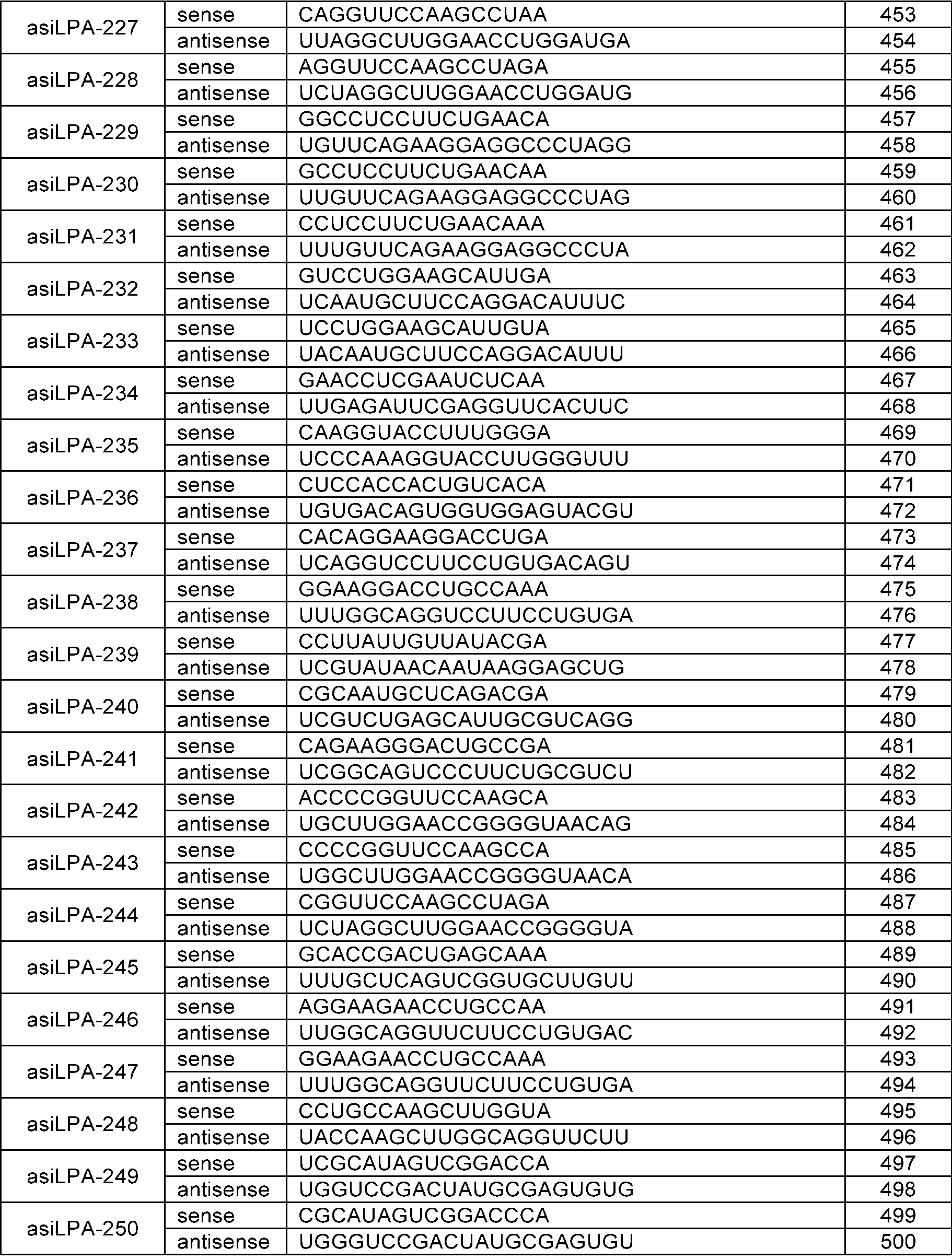
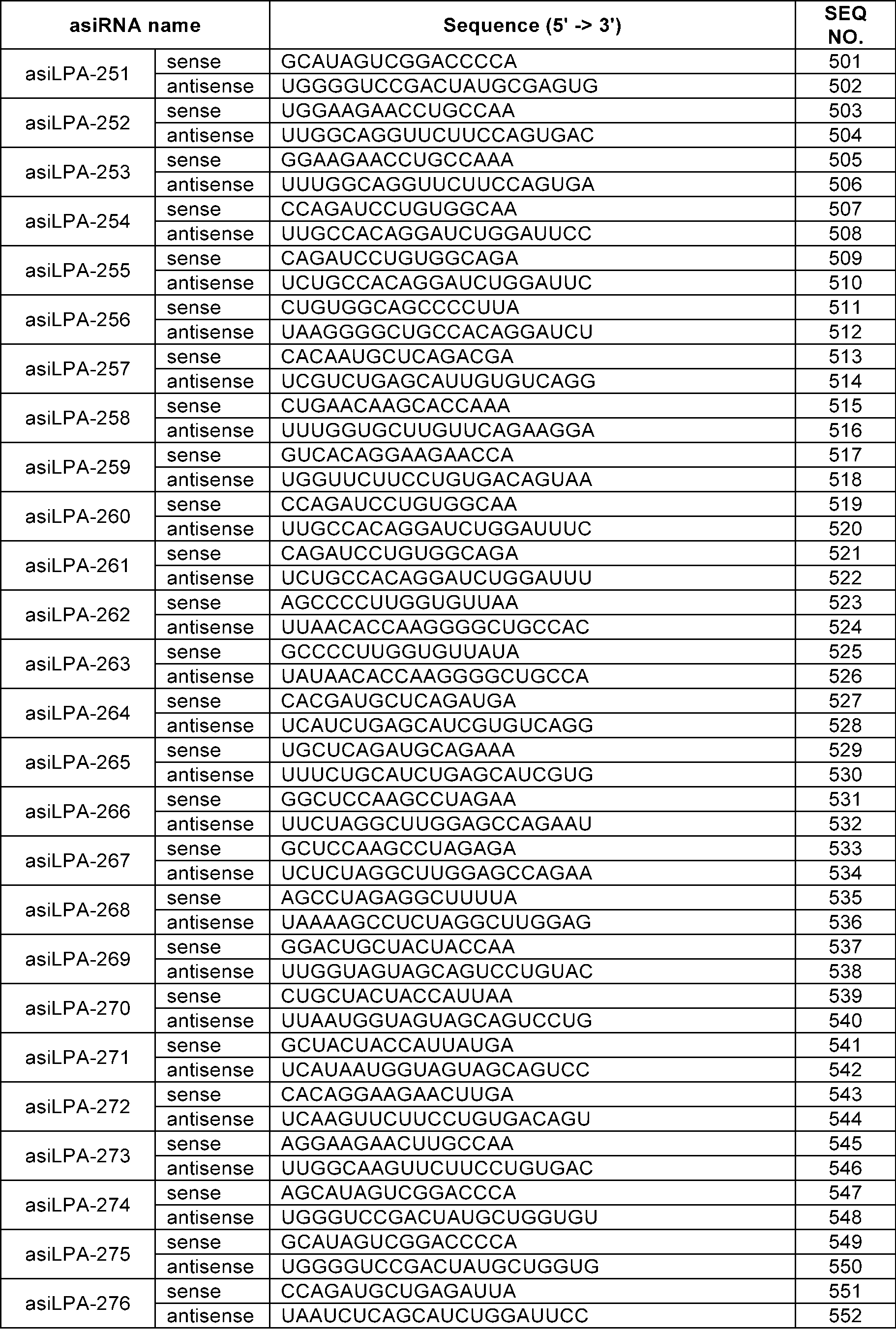
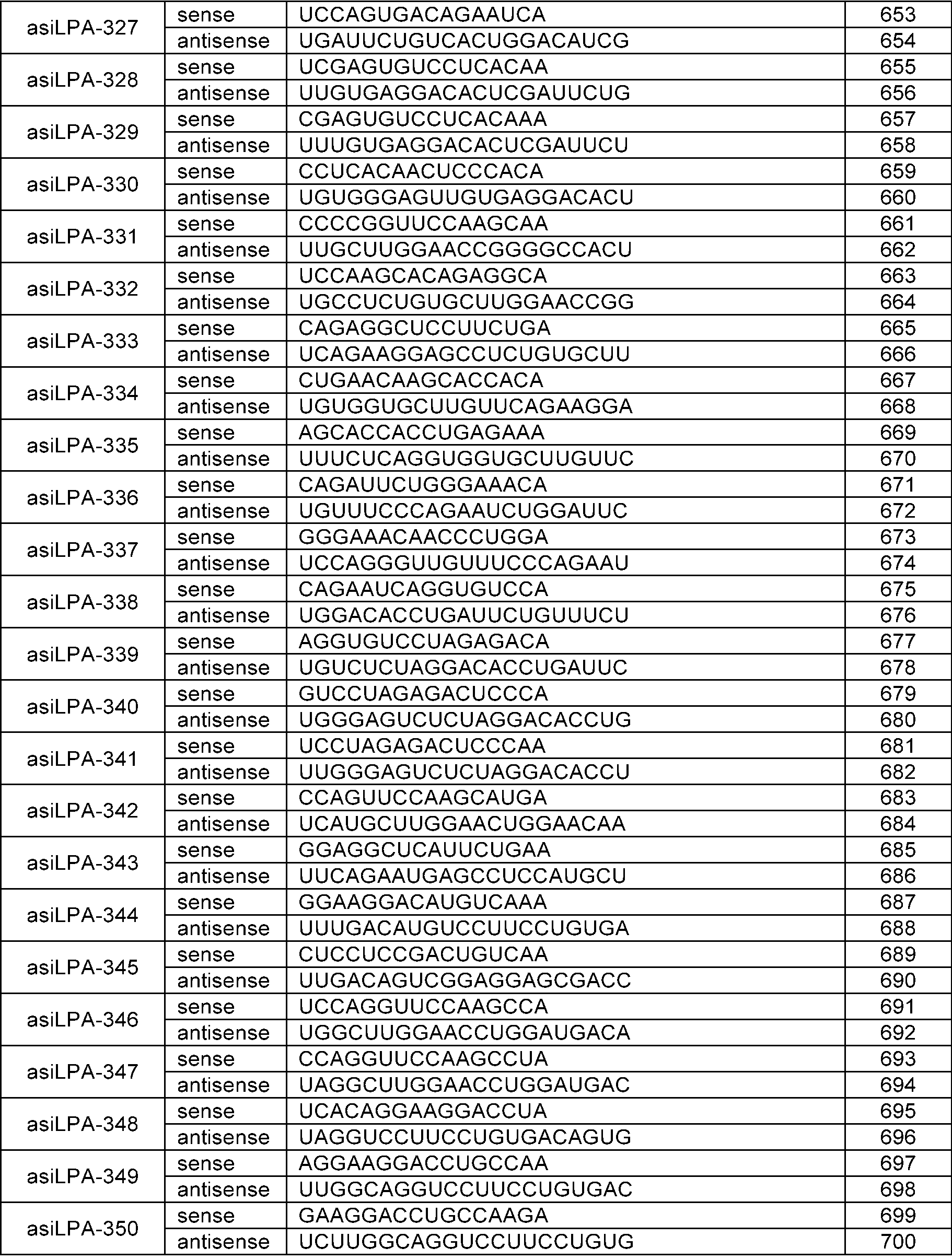
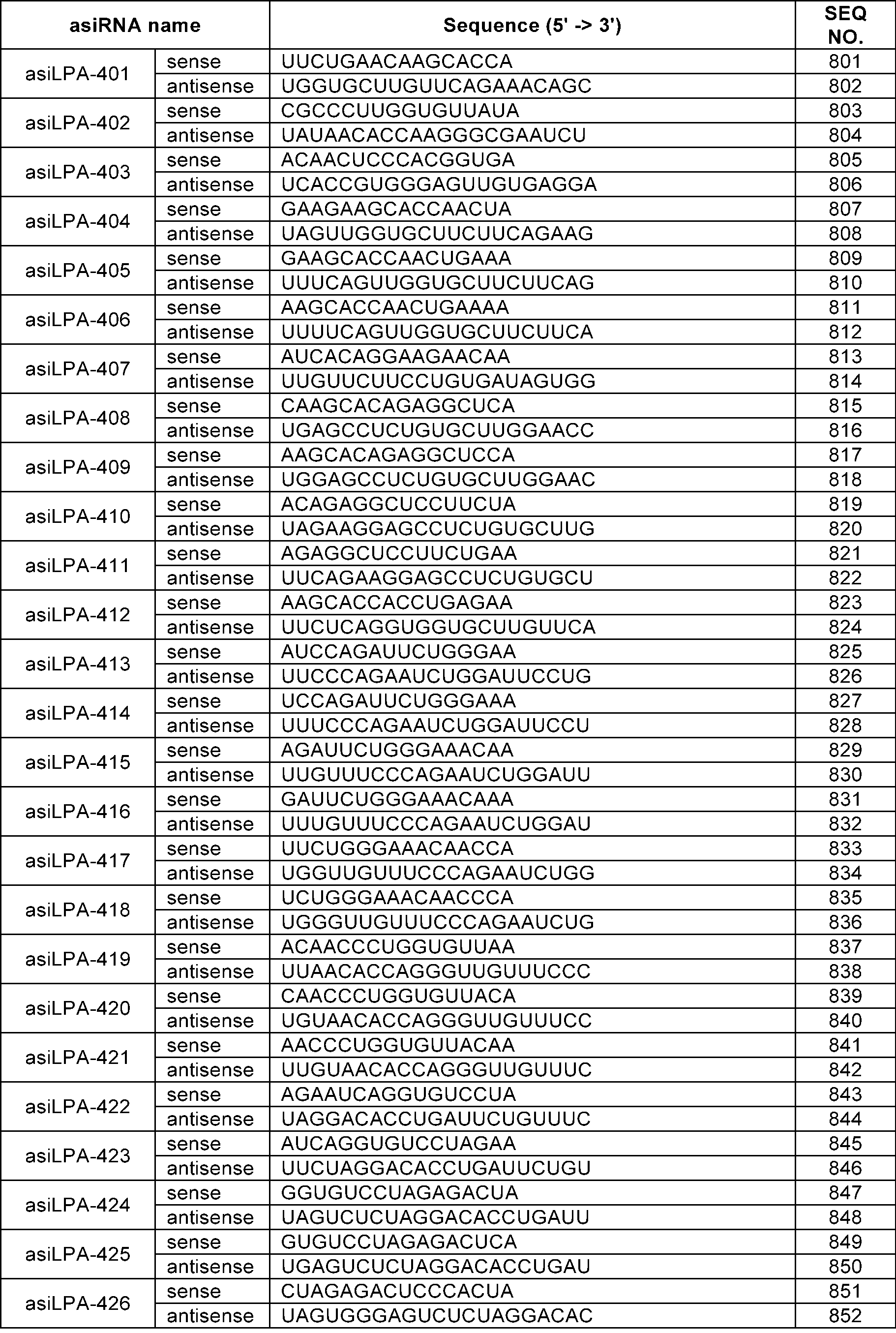
- the sense strand may be a sequence selected from the sense strand sequences listed in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, and Table 9, and the antisense strand may include a sequence selected from the antisense strand sequences listed in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, and Table 9.
- the sense strand may be, for example, at least one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 31, SEQ ID NO: 35, SEQ ID NO: 39, SEQ ID NO: 47, SEQ ID NO: 51, SEQ ID NO: 53, SEQ ID NO: 63, SEQ ID NO: 65, SEQ ID NO: 67, SEQ ID NO: 71, SEQ ID NO: 77, SEQ ID NO: 79, SEQ ID NO: 83, SEQ ID NO: 85, SEQ ID NO: 87, SEQ ID NO: 103, SEQ ID NO: 123, SEQ ID NO: 125, SEQ ID NO: 127, SEQ ID NO: 183, SEQ ID NO: 215, SEQ ID NO: 339, SEQ ID NO: 341, SEQ ID NO: 373, SEQ ID NO: 375, SEQ ID NO: 471, SEQ ID NO: 477, SEQ ID NO: 495, SEQ ID NO: 701, SEQ ID NO:
- the sense strand may be, for example, at least one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 63, SEQ ID NO: 65, SEQ ID NO: 67, SEQ ID NO: 71, SEQ ID NO: 85, SEQ ID NO: 123, SEQ ID NO: 127, SEQ ID NO: 183, SEQ ID NO: 215, SEQ ID NO: 339, SEQ ID NO: 341, SEQ ID NO: 373, and SEQ ID NO: 737, for example, it may be any one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 65, SEQ ID NO: 123, and SEQ ID NO: 183.
- the sense strand may be any one selected from SEQ ID NO: 65, SEQ ID NO: 123, and SEQ ID NO: 183.
- the antisense strand may be, for example, at least one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 32, SEQ ID NO: 36, SEQ ID NO: 40, SEQ ID NO: 48, SEQ ID NO: 52, SEQ ID NO: 54, SEQ ID NO: 64, SEQ ID NO: 66, SEQ ID NO: 68, SEQ ID NO: 72, SEQ ID NO: 78, SEQ ID NO: 80, SEQ ID NO: 84, SEQ ID NO: 86, SEQ ID NO: 88, SEQ ID NO: 104, SEQ ID NO: 124, SEQ ID NO: 126, SEQ ID NO: 128, SEQ ID NO: 184, SEQ ID NO: 216, SEQ ID NO: 340, SEQ ID NO: 342, SEQ ID NO: 374, SEQ ID NO: 376, SEQ ID NO: 472, SEQ ID NO: 478, SEQ ID NO: 496, SEQ ID NO: 702, SEQ ID NO: 12
- the antisense strand may be, for example, at least one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 64, SEQ ID NO: 66, SEQ ID NO: 68, SEQ ID NO: 72, SEQ ID NO: 86, SEQ ID NO: 124, SEQ ID NO: 128, SEQ ID NO: 184, SEQ ID NO: 216, SEQ ID NO: 340, SEQ ID NO: 342, SEQ ID NO: 374, and SEQ ID NO: 738, for example, the antisense strand may be any one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 66, SEQ ID NO: 124, and SEQ ID NO: 184.
- the antisense strand may be any one selected from SEQ ID NO: 66, SEQ ID NO: 124, and SEQ ID NO: 184.
- the RNAi agent has a sense strand as shown in SEQ ID NO: 65 and an antisense strand as shown in SEQ ID NO: 66.
- the RNAi agent has a sense strand as shown in SEQ ID NO: 123 and an antisense strand as shown in SEQ ID NO: 124.
- the RNAi agent has a sense strand as shown in SEQ ID NO: 183 and an antisense strand as shown in SEQ ID NO: 184.
- the sense strand or the antisense strand may include one or more chemical modifications.
- the present disclosure provides novel chemical modification patterns for improving the properties and effects of RNAi agents.
- siRNA cannot pass through the cell membrane due to high negative charge and a high molecular weight of the phosphate backbone structure, and is rapidly degraded and eliminated from the blood, making it difficult to deliver a sufficient amount to an actual target site for RNAi induction.
- many high-efficiency delivery methods using cationic lipids and cationic polymers have been developed, however, for in vivo , it is difficult to deliver siRNA with as high efficiency as in vitro , and there is an issue of reduced siRNA delivery efficiency due to interaction with various proteins existing in the living body.
- RNAi agent having improved hepatocyte targeted delivery ability by introduction of chemical modifications to the asymmetric siRNA structure
- an asymmetric siRNA construct (GalNAc asymmetric siRNA, GalNAc-asiRNA) capable of effective intracellular delivery without a separate carrier.
- the chemical modification in the sense strand or the antisense strand may include one or more selected from: binding with an N-acetylgalactosamine (GalNAc) derivative, or a cell-penetrating peptide; modification of a nucleotide bond with phosphorothioate, boranophosphate, or methyl phosphonate; substitution of an -OH group at the 2' carbon position of the sugar structure of a nucleotide with -H, -CH 3 (methyl), -OCH 3 (-O-methyl), -NH 2 , -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and substitution of an -OH group at the 5' end with E-vinylphosphonate.
- GalNAc N-acetylgalactosamine
- the N-acetylgalactosamine (GalNAc) derivative may have a structure of Formula 1 below.
- the N-acetylgalactosamine (GalNAc) derivative recognizes asialoglycoprotein (ASGPR) receptors on a surface of a hepatocyte and helps RNAi agents to flow into the hepatocyte, that is, acts as an ASGPR targeting moiety, and thus, the RNAi agent, in which the N-acetylgalactosamine (GalNAc) derivative is bound to an end have improved delivery to hepatocytes and may provide an effective targeted treatment for liver diseases.
- ASGPR asialoglycoprotein
- the sense strand may include one or more chemical modifications selected from: modification of 2 to 4 nucleotide bonds adjacent to the 5' end with phosphorothioate, boranophosphate, or methyl phosphonate; substitution of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide with -H, -CH 3 (methyl), -OCH 3 (-O-methyl), -NH 2 , -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and binding with an N-acetylgalactosamine (GalNAc) derivative, or a cell penetrating peptide at the 3' end.
- GalNAc N-acetylgalactosamine
- the antisense strand may include one or more chemical modifications selected from: modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end with phosphorothioate, boranophosphate, or methyl phosphonate; substitution of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide with -H, -CH 3 (methyl), -OCH 3 (-O-methyl), -NH 2 , -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and substitution of an -OH group at the 5' end to E-vinylphosphonate.
- the RNAi agent may include at least one modification selected from: modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end in a sense or an antisense strand to phosphorothioate; modification of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide in a sense strand or an antisense strand to -H, -OCH 3 (-O-methyl), or -F; binding to an N-acetylgalactosamine (GalNAc) derivative at the 3' end of the sense strand; and substitution of an -OH group at the 5' end of an antisense strand to E-vinylphosphonate.
- modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end in a sense or an antisense strand to phosphorothioate modification of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide in a sense strand or
- the RNAi agent as described above having an antisense strand and sense strand, wherein in the antisense strand, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand, from 5' to 3', positions 2, 4, 5, 6 are modified by 2'-fluoro, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative.
- GalNAc N-acetylgalactosamine
- the RNAi agent as described above having a sense strand from 5' to 3': positions 2, 4, 5, 6, 14 and 16 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe.
- the RNAi agent as described above having a sense strand from 5' to 3': positions 2, 4, 5, 6 and 12 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe.
- the chemical modification pattern is: in the antisense strand of a RNAi agent, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are modified as deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand of a RNAi agent, from 5' to 3', positions 2, 4, 5, 6, 14 and 16 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative.
- GalNAc N-acetylgalactosamine
- the chemical modification pattern is: in the antisense strand of a RNAi agent, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are modified as deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand of a RNAi agent, from 5' to 3', positions 2, 4, 5, 6 and 12 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative.
- GalNAc N-acetylgalactosamine
- RNAi agent in the sense strand of the RNAi agent as described above, from 5' to 3' may be modified as:
- the trivalent GalNAc derivative is Formula 1.
- RNAi agent in the sense strand of the RNAi agent as described above, from 5' to 3' may be modified as:
- the trivalent GalNAc derivative is Formula 1.
- the sense strand may be a sequence selected from the sense strand sequences listed in Table 15, and the antisense strand may include a sequence selected from the antisense strand sequences listed in Table 15.
- the RNAi agent is selected from the GalNAc-asiRNA conjugates listed in Table 15.
- the sense strand may be any one selected from: (A) to (D) in the table below
- the antisense strand may be any one selected from: (a) to (d) in the table below.
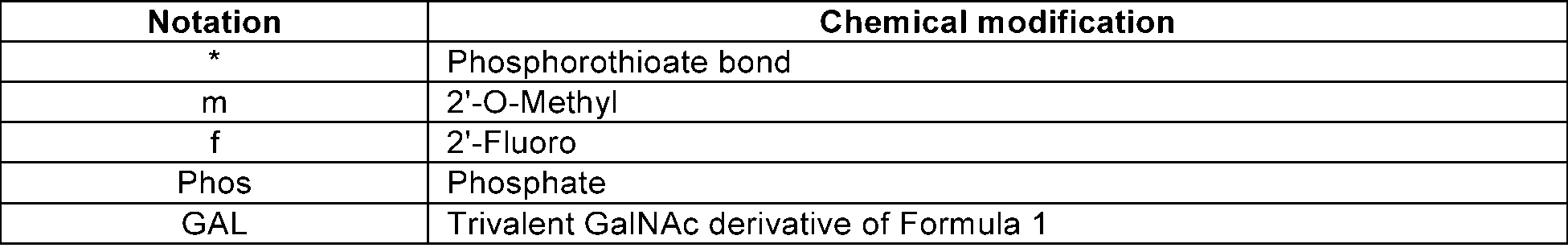
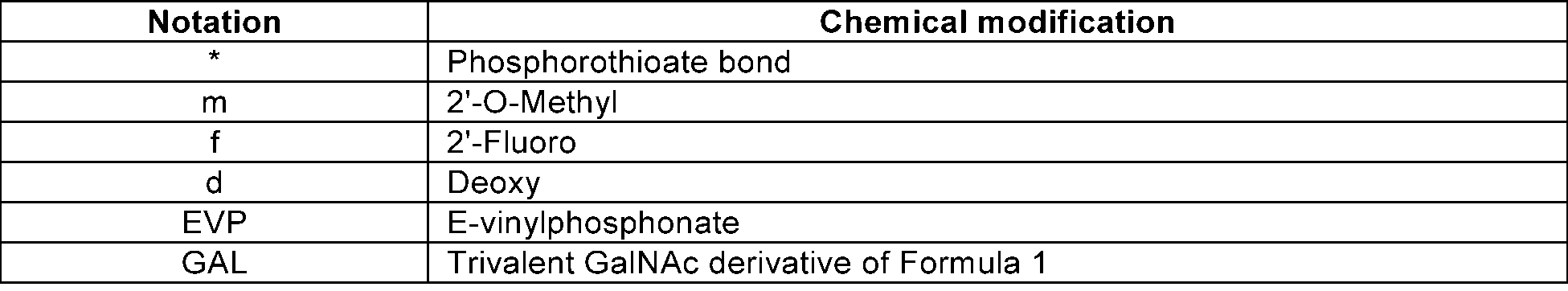
- * denotes a phosphorothioated bond
- m denotes 2'-O-methyl
- f denotes 2'-fluoro
- d denotes a form in which existing 2'-OH is substituted with -H
- EVP denotes E-vinylphosphonate
- GAL denotes a trivalent GalNAc derivative of Formula 1.
- the sense strand and the antisense strand of the RNAi agent may be sequences of the sense strand and antisense strand below selected from the table above, for example, the RNAi agent may have a combination of a sense strand (A) and an antisense strand (a); a combination of a sense strand (B) and an antisense strand (b); a combination of a sense strand (C) and an antisense strand (c); or a combination of a sense strand (D) and an antisense strand (d).
- Another aspect provides a pharmaceutical composition including the RNAi agent as an active ingredient, for preventing or treating cardiovascular disease.
- Still another aspect provides use of the RNAi agent for manufacturing a pharmaceutical product, for preventing or treating cardiovascular disease.
- the pharmaceutical composition contains or uses the above-described RNAi agent as it is, descriptions thereof are omitted in order to avoid excessive complexity of the present specification.
- the pharmaceutical composition may be utilized as an active ingredient of a pharmaceutical composition for preventing or treating cardiovascular disease by inhibiting expression of an LPA gene.
- the cardiovascular disease may collectively refer to primary diseases directly caused by various causes such as abnormality, dysfunction, and damage of the circulatory system in vivo , as well as secondary diseases derived from the primary diseases.
- the cardiovascular disease is a disease closely related to an expression level of the LPA gene, and may be for example, any one selected from: Berger's disease, peripheral artery disease, coronary artery disease, metabolic syndrome, acute coronary syndrome, aortic valve stenosis, myocardial infarction, aortic valve regurgitation, aortic dissection, retinal artery occlusion, cerebrovascular disease, mesenteric ischemia, superior mesenteric artery occlusion, renal artery stenosis, stable/unstable angina, acute coronary syndrome, heterozygous or homozygous familial hypercholesterolemia, hyperaphobeta lipoproteinemia, cerebrovascular atherosclerosis, and venous thrombosis, but is not limited thereto.
- RNAi agent means an appropriate effective amount of an ingredient that brings about a beneficial or desirable clinical or biochemical outcome. Specifically, the term may refer to an agent, an active agent, or an RNAi agent of an effective amount.
- the effective amount may be administered one or more times, and unlimitedly may be an appropriate amount for preventing a disease, alleviating symptoms, reducing an extent of the disease, stabilizing (i.e., not exacerbating) the disease state, delaying or reducing a rate of disease progression, or improving or temporarily alleviating and ameliorating (partially or fully) a disease state.
- prevention refers to any action that blocks an occurrence of a disease in advance, suppresses a disease, or delays progression thereof.
- the term refers to preventing the cardiovascular disease or occurrence of characteristics thereof, interrupting the occurrence, or defending or protecting from the cardiovascular disease or occurrence of the characteristics thereof.
- treatment refers to both therapeutic treatment and preventive or prophylactic measures. In addition, it refers to any action in which the symptoms of a disease are improved or beneficially changed. For example, the term refers to preventing, reducing or improving the cardiovascular disease or characteristics thereof, or delaying (attenuating) progression of the cardiovascular disease or characteristics thereof in a subject.
- ⁇ ективное amount refers to a generally accepted meaning in the art.
- the term generally refers to an amount of a molecule, compound, or construct that elicits an intended biological response (e.g., a beneficial response) in a cell, tissue, a system, an animal, or a human, sought by researchers, veterinarians, physicians, or other clinicians, etc.
- a "therapeutically effective amount” refers to an amount of a molecule, compound, or construct that is capable of eliciting a desirable medical response to the extent that a particular clinical treatment may be considered effective, due to, for example, a therapeutically relevant change in a measurable parameter associated with the disease or disorder.
- the therapeutically effective amount of a drug for treatment of a disease or a disorder may be an amount necessary to cause a therapeutically relevant change in the parameter.
- a method of administering the pharmaceutical composition may be determined by a person skilled in the art based on the symptoms of the patient and the severity of the disease.
- the pharmaceutical composition may be formulated in various forms such as powders, tablets, capsules, solutions, injections, ointments, syrups, etc., and may be provided in unit-dose or multi-dose containers, for example, sealed ampoules and bottles.
- composition of the present disclosure may be administered orally or parenterally.
- Routes of administration of the composition according to the present disclosure may be, for example, oral, intravenous, intramuscular, intraarterial, intramedullary, intrathecal, intracardiac, transdermal, subcutaneous, intraperitoneal, intestinal, sublingual, or topical, but are not limited thereto.
- Dosage of the composition according to the present disclosure varies depending on the patient's weight, age, sex, health status, diet, administration time, method, excretion rate, or severity of disease, etc. and may be readily determined by those of average skill in the art.
- the composition of the present disclosure may be formulated into a suitable formulation for clinical administration using known techniques.
- Still another aspect provides a method of preventing or treating cardiovascular disease, including administering the RNAi agent to a subject.
- the method of treating cardiovascular disease includes or uses the above-described pharmaceutical composition as it is, descriptions thereof are omitted in order to avoid excessive complexity of the present specification.
- subject refers to a subject in need of treatment for a disease, specifically, cardiovascular disease, and more specifically, the term may include all mammals such as a human or a non-human primate, a mouse, a dog, a cat, a horse, a cow, sheep, a pig, a goat, a camel, and an antelope.
- An RNAi agent according to an aspect may inhibit expression of the LPA protein by binding to and degrading LPA mRNA, while alleviating side effects such as non-specific immune responses and off-target effects.
- RNAi agent according to an aspect is effectively delivered to hepatocytes and may be usefully as a target therapeutic agent for cardiovascular diseases.
- FIG. 1 is a diagram schematically showing a structure of LPA asiRNA consisting of a 16-mer sense strand and a 21-mer antisense strand;
- FIG. 2a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 55 types of LPA asiRNAs (asiLPA-001 to asiLPA-055) according to an aspect, at a concentration of 10 nM;
- FIG. 2b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-056 to asiLPA-109) according to an aspect, at a concentration of 10 nM;
- FIG. 3a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 55 types of LPA asiRNAs (asiLPA-110 to asiLPA-164) according to an aspect, at a concentration of 10 nM;
- FIG. 3b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-165 to asiLPA-218) according to an aspect, at a concentration of 10 nM;
- FIG. 4a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 55 types of LPA asiRNAs (asiLPA-219 to asiLPA-273) according to an aspect, at a concentration of 10 nM;
- FIG. 4b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-274 to asiLPA-327) according to an aspect, at a concentration of 10 nM;
- FIG. 5a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-328 to asiLPA-381) according to an aspect, at a concentration of 10 nM;
- FIG. 5b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 53 types of LPA asiRNAs (asiLPA-382 to asiLPA-434) according to an aspect, at a concentration of 10 nM;
- FIG. 6 shows results of confirming expression levels of LPA mRNA, after respectively treating Hep3B cells with 65 types of LPA asiRNAs, at a concentration of 1 nM;
- FIG. 7 shows results of confirming expression levels of LPA mRNA, after respectively treating Hep3B cells with 65 types of LPA asiRNAs, at a concentration of 1 nM;
- FIG. 8 shows results of confirming expression levels of LPA mRNA, after respectively treating Hep3B cells with 40 types of LPA asiRNAs according to an aspect, at a concentration of 0.1 nM;
- FIG. 9 shows results of confirming expression levels of LPA mRNA, after respectively treating primary human hepatocytes with 40 types of LPA asiRNAs according to an aspect, at a concentration of 10 nM;
- FIG. 10 shows results of confirming expression levels of LPA mRNA, after respectively treating primary cynomolgus monkey hepatocytes with 40 types of LPA asiRNAs according to an aspect, at a concentration of 10 nM;
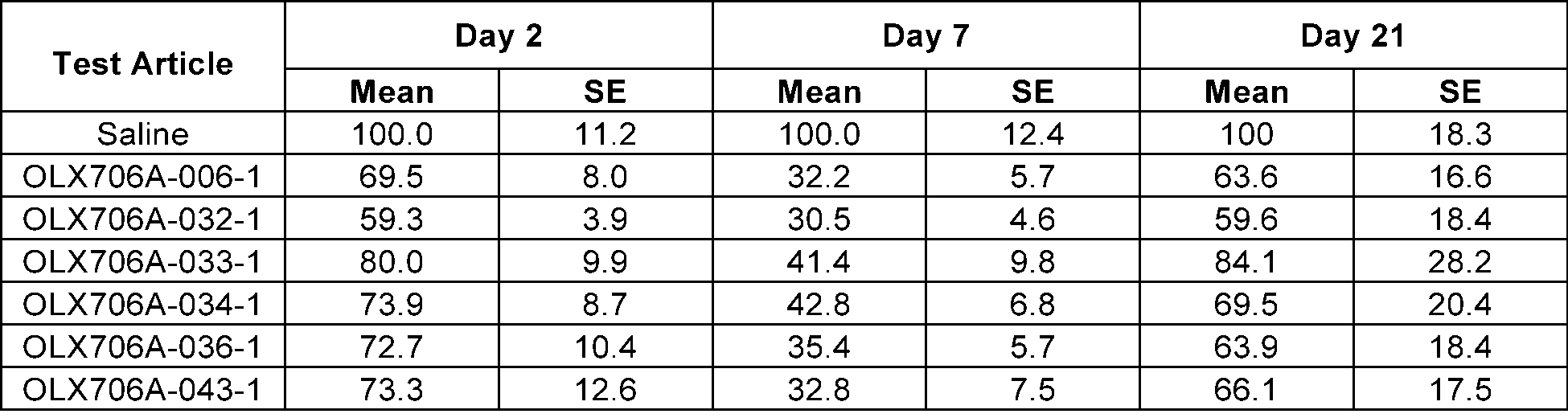
- FIG. 11 shows results of confirming changes in LPA expression levels over time, after subcutaneously administering 6 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-032-1, OLX706A-033-1, OLX706A-034-1, OLX706A-036-1, OLX706A -043-1) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P1) HDI mice at a single dose of 8 mg/kg (mpk);
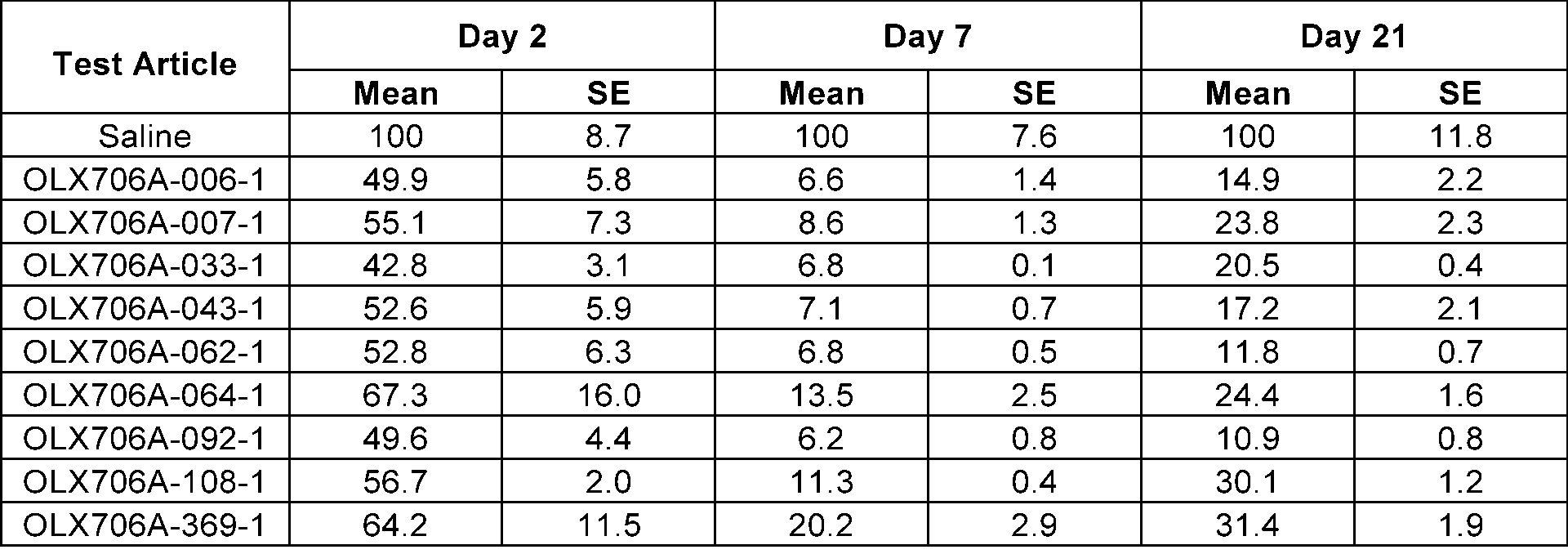
- FIG. 12 shows results of confirming changes in LPA expression levels over time, after subcutaneously administering 9 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-007-1, OLX706A-033-1, OLX706A-043-1, OLX706A-062-1, OLX706A-064-1, OLX706A-092-1, OLX706A-108-1, OLX706A-369-1) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P2) HDI mice at a single dose of 8 mg/kg (mpk);
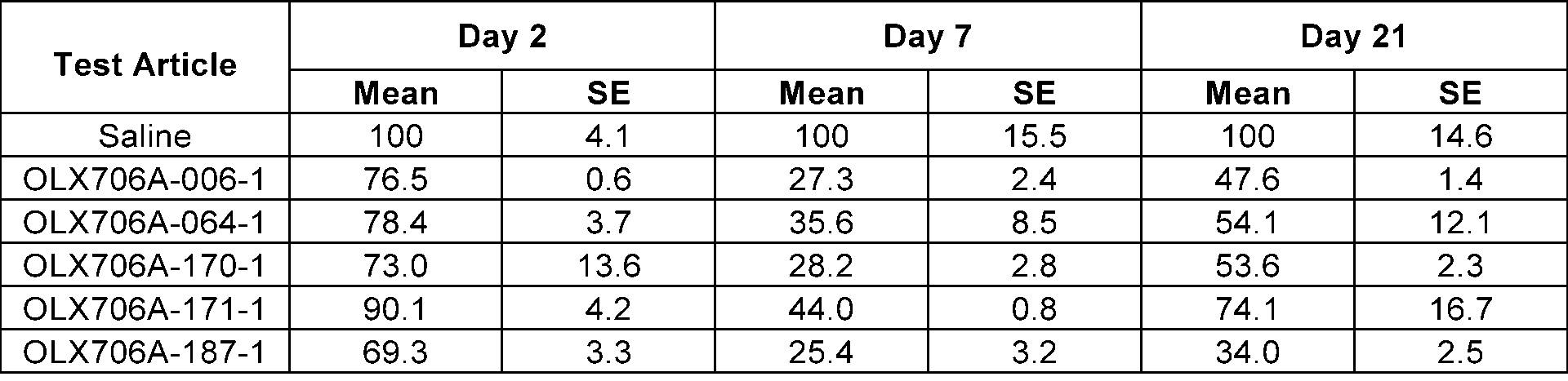
- FIG. 13 shows results of confirming changes in LPA expression levels over time, after subcutaneously administering 5 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-064-1, OLX706A-170-1, OLX706A-171-1, OLX706A-187-1) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P3) HDI mice at a single dose of 8 mg/kg (mpk);
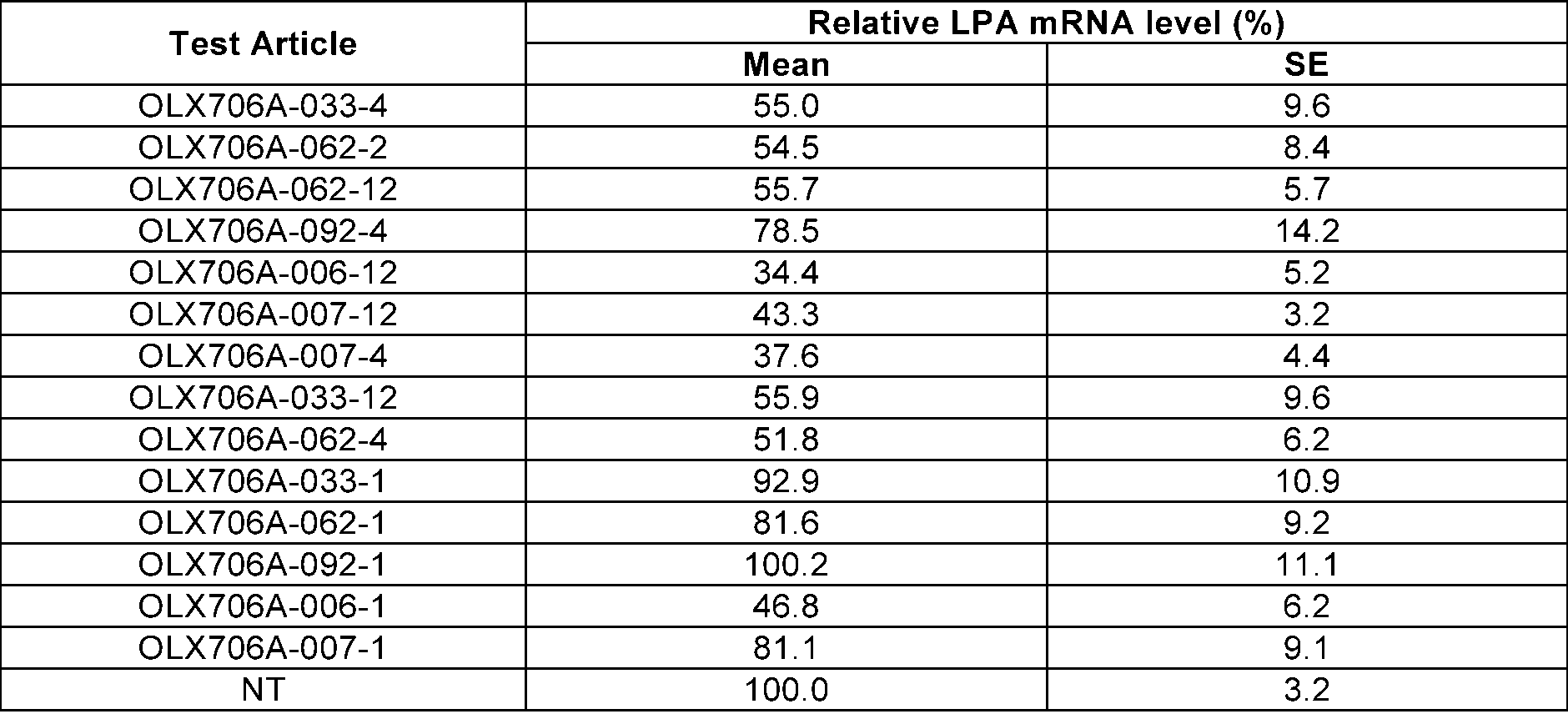
- FIG. 14 shows results of confirming expression levels of LPA mRNA, after respectively treating primary human hepatocytes with 14 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-2, OLX706A-062-12, OLX706A-092-4, OLX706A-033-1, OLX706A-062-1, OLX706A-092-1, OLX706A-006-12, OLX706A-007-12, OLX706A-007-4, OLX706A-033-12, OLX706A-062-4, OLX706A-006-1, OLX706A-007-1) according to an aspect, into which chemical modifications are introduced, at a concentration of 200 nM; and
- FIG. 15 shows results of confirming changes in LPA expression levels, after subcutaneously administering 6 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, OLX706A-092-4) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P2) HDI mice at a single dose of 5 mg/kg (mpk).
- 6 types of LPA GalNAc-asiRNAs OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, OLX706A-092-4
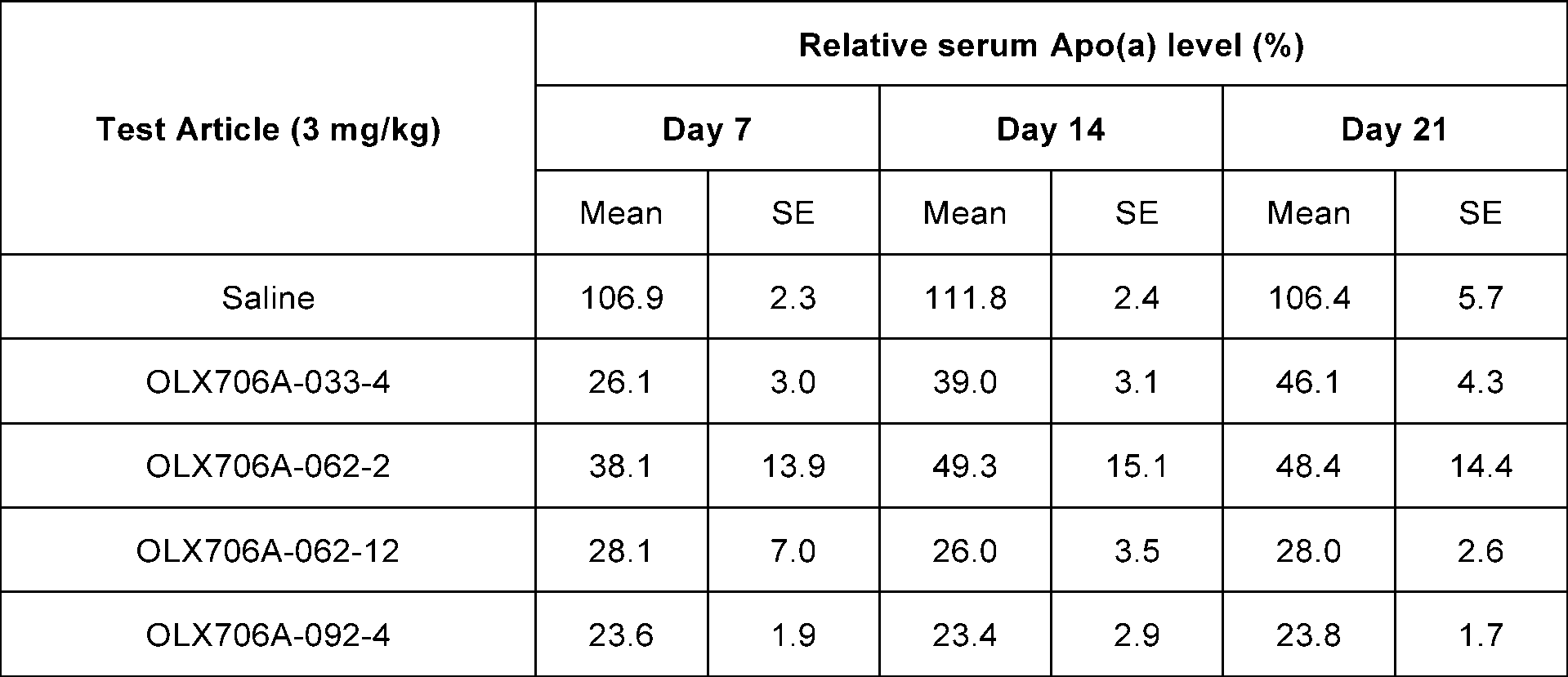
- FIG. 16a shows results of confirming changes in Apo(a) expression levels over time, after subcutaneously administering 4 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-2, OLX706A-062-12, OLX706A-092-4) according to an aspect, into which chemical modifications are introduced, to Apo(a) TG mice at a single dose of 3 mg/kg (mpk);
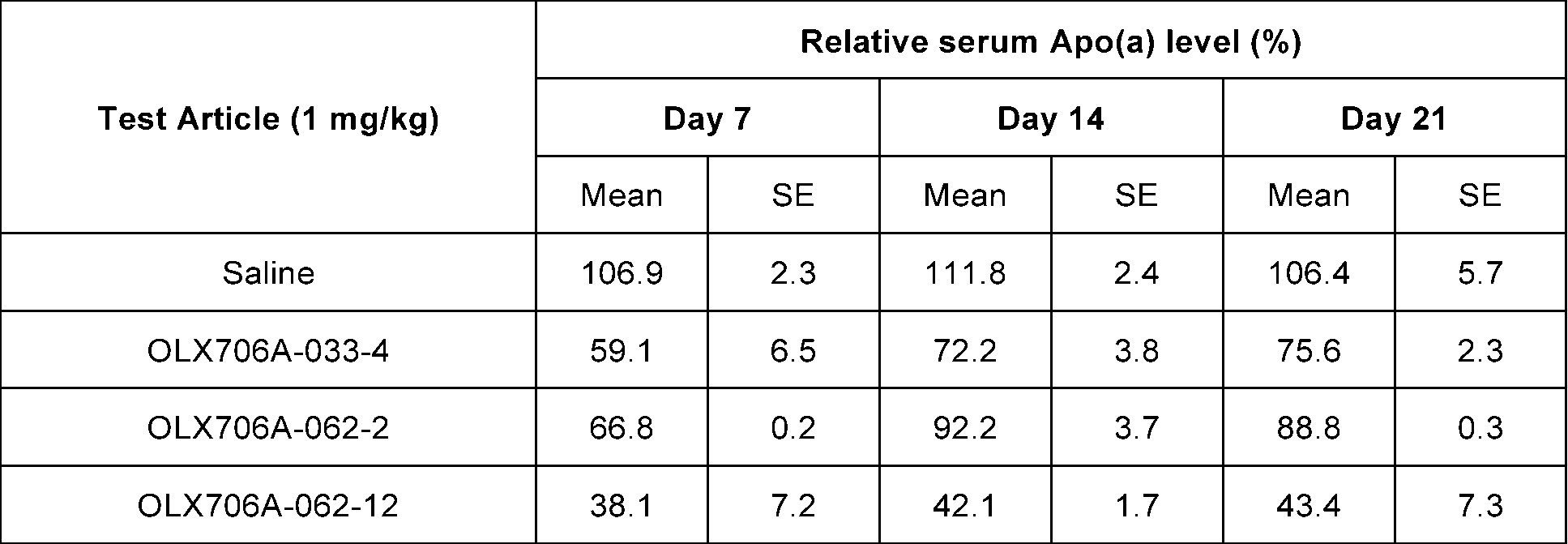
- FIG. 16b shows results of confirming changes in Apo(a) expression levels over time, after subcutaneously administering 3 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-2, OLX706A-062-12) according to an aspect, into which chemical modifications are introduced, to Apo(a) TG mice at a single dose of 1 mg/kg (mpk);
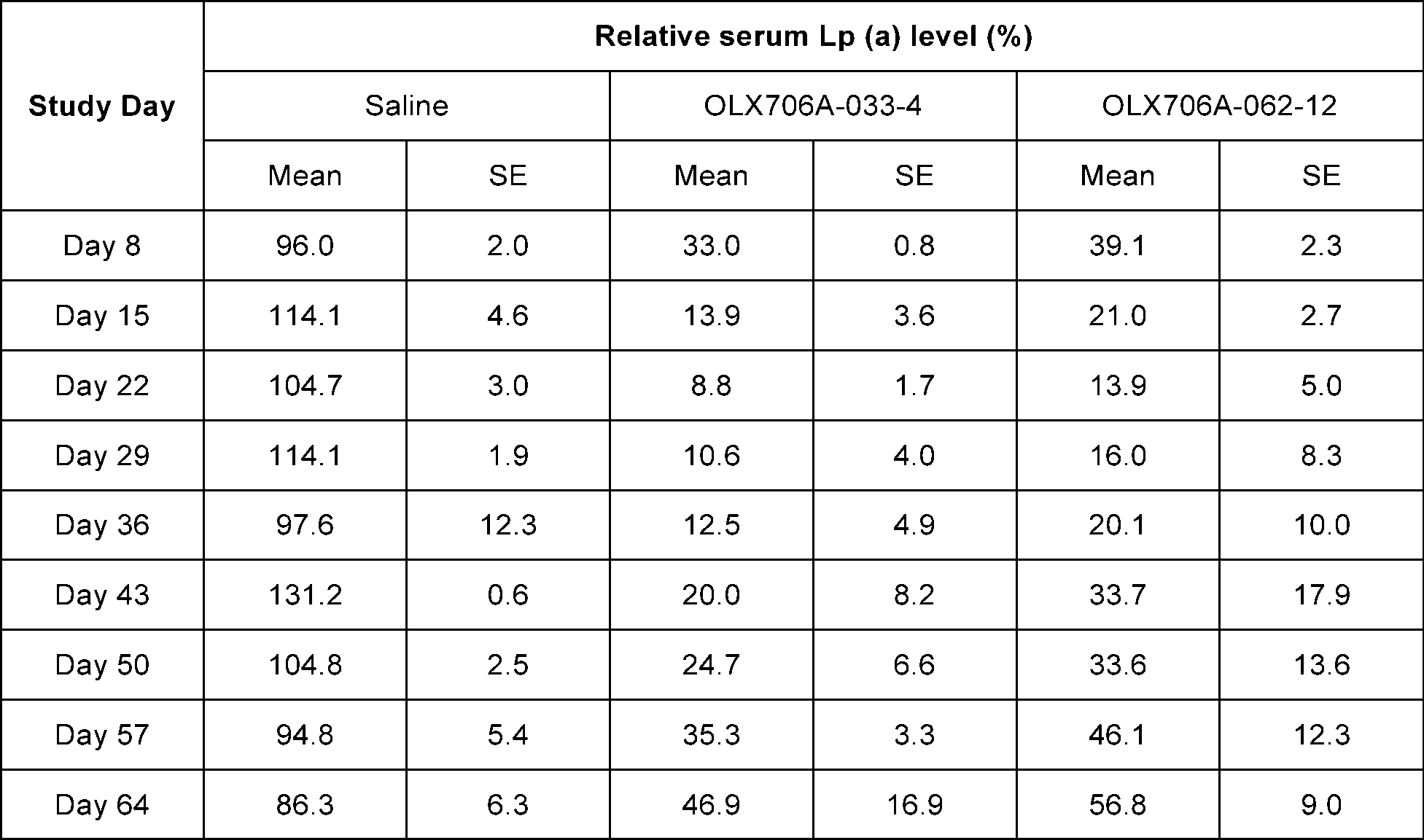
- FIG. 17 shows results of confirming changes in Lp(a) expression levels over time, after subcutaneously administering 2 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-12) according to an aspect, into which chemical modifications are introduced, to cynomolgus monkey at a single dose of 3 mg/kg (mpk).
- Example 1 Screening of nucleic acid molecules for inducing RNAi targeting LPA
- LPA asiRNAs LPA asymmetric siRNAs
- Hep3B cells which are human liver cancer cells, were transfected with the 434 types of LPA asiRNAs at a concentration of 10 nM, and a dual-luciferase reporter assay was performed to measure expression levels of LPA mRNA.
- LPA asiRNA (10 nM) and psiCHECK-2-LPA plasmid DNA (10 ng/well) were delivered into Hep3B cells by using lipofectamine 2000 (Invitrogen, 11668019, 0.2 ⁇ l/well).
- LPA mRNA expressed by LPA mRNA
- LPA mRNA expressed by LPA mRNA
- a dual luciferase reporter assay system Promega, Cat.E1980
- Hep3B cells which are human liver cancer cells, were transfected with the 130 types of selected LPA asiRNAs at a concentration of 1 nM, and a dual-luciferase reporter assay was performed to measure expression levels of LPA mRNA, in the same manner as in Example 1-2.
- LPA asiRNAs showing relatively good expression inhibition efficiency were identified (asiLPA-002, 006, 007, 008, 016, 018, 020, 024, 026, 027, 032, 033, 034, 036, 039, 040, 042, 044, 052, 063, 064, 092, 102, 108, 170, 171, 187, 188, 213, 236, 351, 355, 356, 357, 365, 368, 369, 370, 372, 388).
- Hep3B cells, primary human hepatocytes, or primary cynomolgus monkey hepatocytes were transfected with the 40 types of LPA asiRNAs, respectively, and dual-luciferase reporter assay, or qRT-PCR was performed to measure expression levels of LPA mRNA.
- the Hep3B cells were transfected with the total of 40 types of LPA asiRNAs at a concentration of 0.1 nM, and expression levels of LPA mRNA were measured in the same manner as in Example 1-2.
- the primary human hepatocytes were transfected with the total of 40 types of LPA asiRNAs at a concentration of 10 nM, and expression levels of LPA mRNA were measured by performing qRT-PCR.
- the primary human hepatocytes were seeded in a 96 well plate at 1.0 ⁇ 10 4 cells/well, and transfected with LPA asiRNA (10 nM), by using lipofectamine 2000 (Invitrogen, 13778150). 24 hours after the transfection, cell lysate was prepared by using SuperPrepTM Cell lysis & RT kit for qPCR Kit II (TOYOBO, SCQ-401), and cDNA was synthesized through reverse transcription using the mRNA included in the lysate as a template.
- the primary cynomolgus monkey hepatocytes were also transfected with the 40 types of LPA asiRNAs at a concentration of 10 nM, and expression levels of LPA mRNA were measured by performing qRT-PCR. Specifically, the primary cynomolgus monkey hepatocytes were seeded in a 96 well plate at 4.5 ⁇ 10 4 cells/well, and transfected with LPA asiRNA (10 nM), by using lipofectamine 2000 (Invitrogen, 13778150).
- cell lysate was prepared using SuperPrepTM Cell lysis & RT kit for qPCR Kit II (TOYOBO, SCQ-401), and cDNA was synthesized through reverse transcription using the mRNA contained in the lysate as a template. Then, by using the synthesized cDNA as a template, quantitative PCR was performed by using TaqMan® Gene Expression Assays, and a probe (Mf02789271_m1: cyno LPA, Mf04354341_g1: ACTB, Applied Biosystems). Then, expression levels of LPA mRNA were identified by using a CFX Connect Real-Time PCR System (Bio-Rad).
- Example 1 40 types of LPA asiRNAs showing relatively good expression inhibition efficiency were identified (asiLPA-006, 007, 016, 018, 020, 024, 026, 027, 032, 033, 034, 036, 039, 040, 042, 043, 044, 052, 062, 063, 064, 092, 108, 170, 171, 187, 188, 236, 239, 248, 351, 355, 356, 357, 358, 368, 369, 370, 372, 388) for further screening.
- Example 2 Screening by using RNAi-inducing nucleic acid molecules with chemical modifications introduced
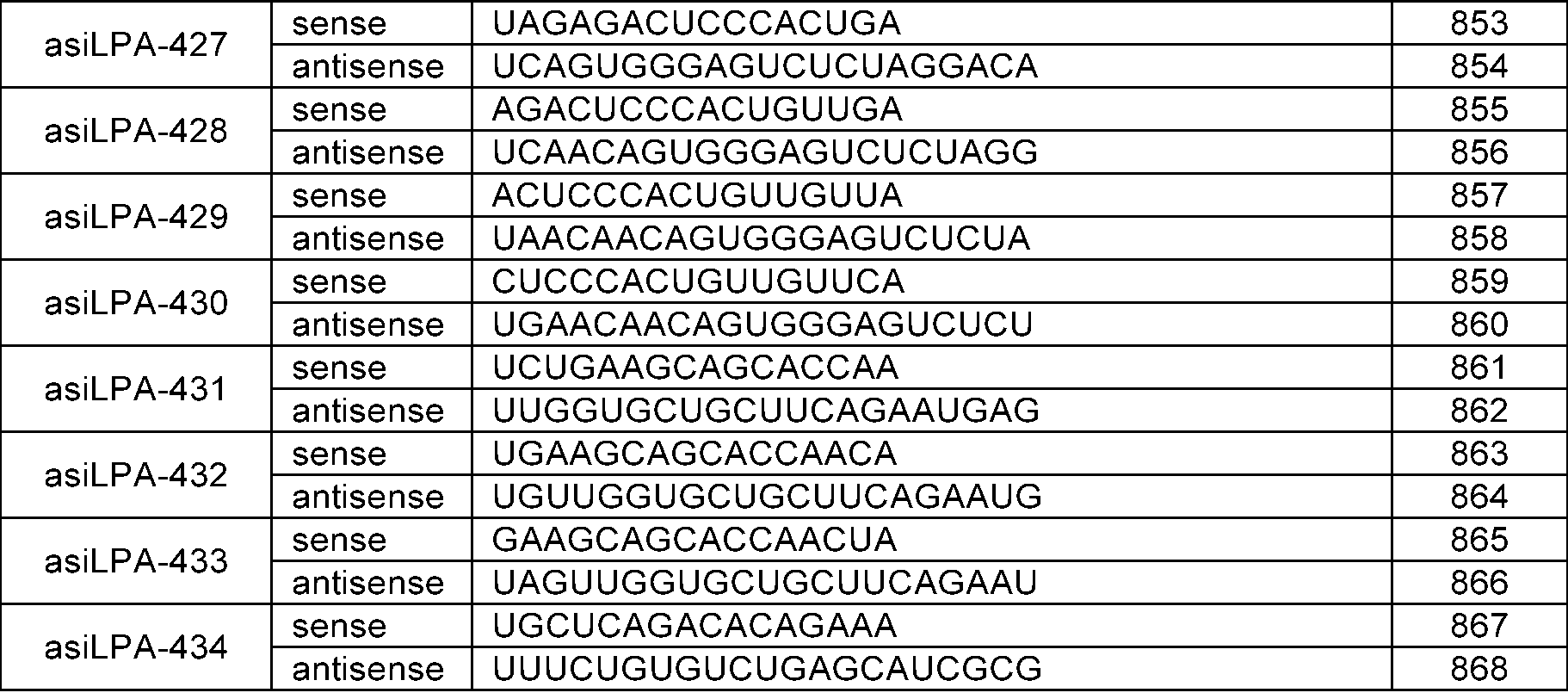
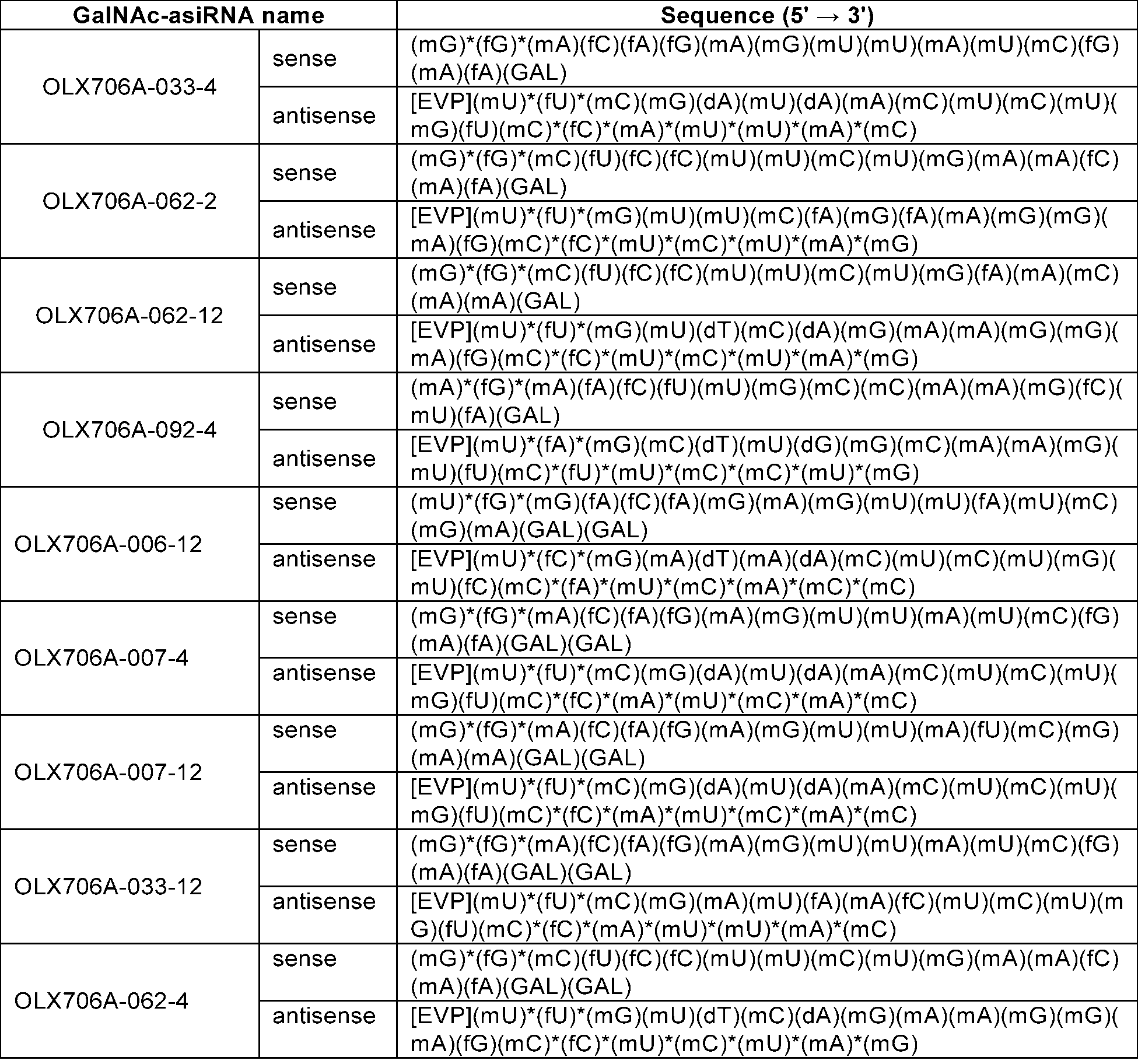
- LPA GalNAc-asiRNAs As a double-stranded nucleic acid molecule that induces high-efficiency RNA interference targeting LPA, LPA GalNAc-asiRNAs with chemical modifications introduced were designed. Specifically, in consideration of the experimental results of Example 1, 15 types of LPA asiRNAs (asiLPA-006, 007, 032, 033, 034, 036, 043, 062, 064, 092, 108, 170, 171, 187, 369) was selected as basic skeletons.
- LPA GalNAc-asiRNAs 15 types were designed with each of the basic skeletons, into which are introduced 1) substitution of an -OH group at the 2' carbon position of the sugar structure in the nucleotides of the sense and antisense strands with -OMe or -F, 2) introduction of phosphorothioate modifications in the backbone of the 5' end or 3' end of the antisense strand, and the 5' end of the sense strand, 3) introduction of phosphate at the 5' end of the antisense strand, and 4) introduction of GalNAc at the 3' end of the sense strand.
- Sequence information of the chemically modified LPA GalNAc-asiRNA prepared in this example is shown in Table 10 below.
- "*” means a form in which an existing phosphodiester bond is substituted with a phosphorothioate bond
- "m” means a form in which an existing 2'-OH is substituted with 2'-O-methyl
- "f” means, for example, in a case of fG, a form in which 2'-OH of an existing guanine (G) is substituted with fluorine.
- Phos refers to a form in which 5'-OH is substituted with phosphate
- GAL refers to a form in which a trivalent GalNAc derivative of Formula 1 is bonded to the 3' end of the sense strand (see Table 11).
- the 15 types of LPA GalNAc-asiRNAs introduced with the above chemical modifications were respectively administered to SEAP-LPA HDI mice in which 3 types of plasmids (P1, P2, and P3) were injected, and accordingly, changes in LPA levels were measured.
- the 3 types of plasmids were prepared by dividing the LPA from a Kringle IV site to a protease domain site into P1 (from Kringle IV-1 to Kringle IV-6), P2 (from Kringle IV-7 to Kringle IV-34), and P3 (from Kringle IV-35 to a protease domain).
- mice (Balb/c, female, 6 weeks old) through hydrodynamic injection (HDI).
- the mice were subcutaneously injected with LPA GalNAc-asiRNAs at a single dose of 8 mg/kg (mpk).
- SEAP levels in the serum were measured by using the Phospha-LightTM SEAP Reporter Gene Assay System (InvitrogenTM, T1015), and the levels of LPA in the serum derived from mice were confirmed by normalizing the measured value with a level of SEAP in the serum of a control group to which saline was administered on the day the LPA levels were measured, and with a SEAP level in the serum before the administration of LPA GalNAc-asiRNAs.
- FIG.11 and Table 12 in mice with P1 plasmids introduced, with FIG. 12 and Table 13 in mice with P2 plasmids introduced, and with FIG. 13 and Table 14 in mice introduced with P3 plasmids.
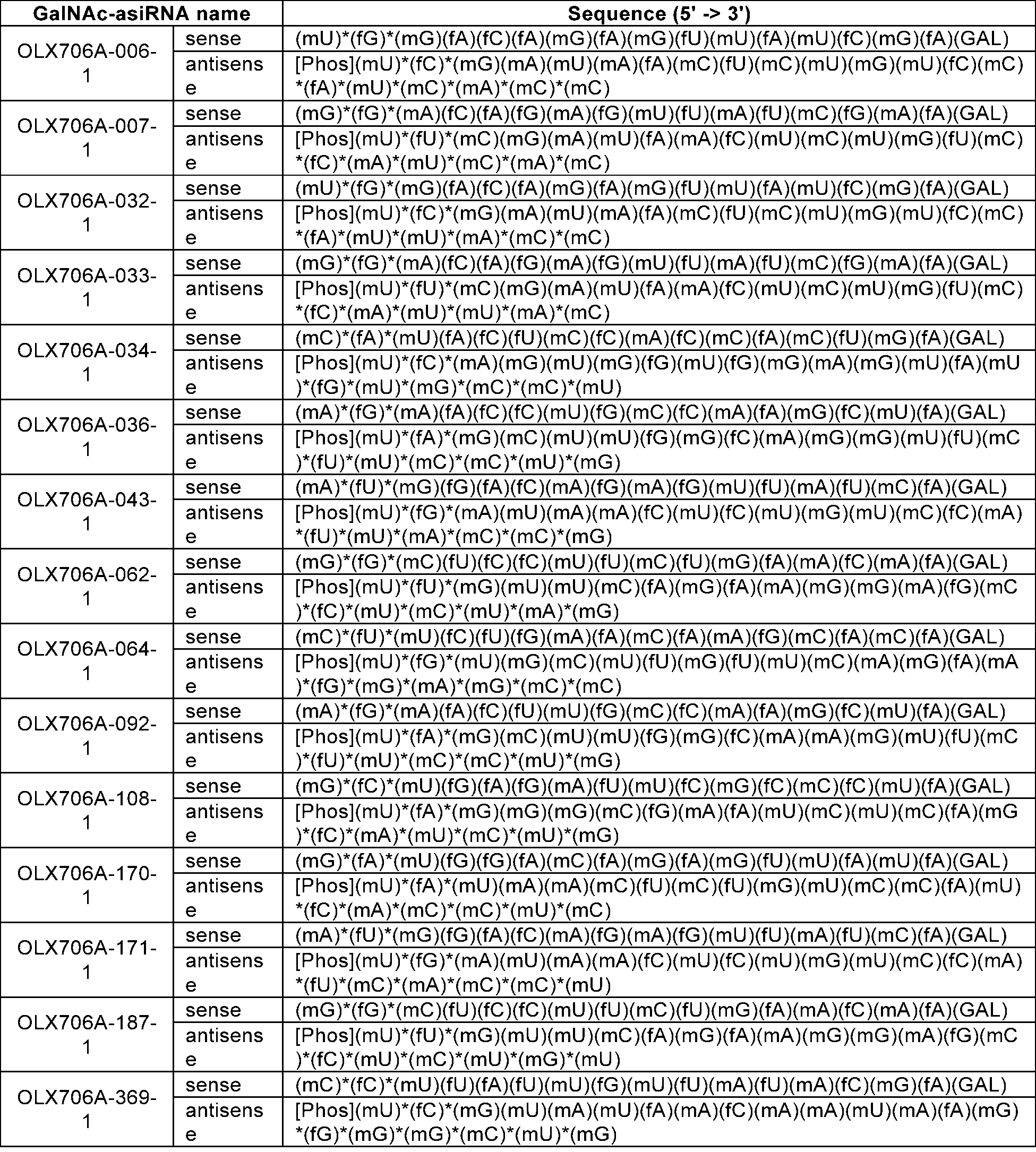
- LPA GalNAc-asiRNAs As a double-stranded nucleic acid molecule that induces high-efficiency RNA interference targeting LPA, LPA GalNAc-asiRNAs with chemical modifications introduced were designed.
- LPA GalNAc-asiRNAs different types, wherein in the LPA GalNAc-asiRNAs, 1) an -OH group at the 2' carbon position of the sugar structure in the nucleotides of the sense and antisense strands is substituted with -OMe or -F, 2) a phosphorothioate modification is introduced to a backbone of the 5' or 3' end of the antisense strand and the 5' end of the sense strand, 3) E-vinylphosphonate is introduced at the 5' end of the antisense strand, and 4) GalNAc is introduced at the 3' end of the sense strand.
- LPA asiRNAs (asiLPA-006,asiLPA-007,asiLPA-033, asiLPA-062, and asiLPA-092) of Example 2 having an effective inhibitory effect on LPA mRNA expression were used as a basic framework, and various modification patterns designed from the chemical modifications of Table 16 were introduced to the basic frameworks to obtained many different GalNAc-asiRNAs conjugates, e.g., OLX706A-006-12, OLX706A-007-12, OLX706A-007-4, OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, and OLX706A-092-4 were prepared.
- OLX706A-006-1, OLX706A-007-1, OLX706A-033-1, OLX706A-062-1, and OLX706A-092-1 in this Example were prepared in the same manner as in Example 2.
- Sequence information of the chemically modified LPA GalNAc-asiRNA prepared in this example is shown in Table 15 below.
- "*” means a form in which an existing phosphodiester bond is substituted by a phosphorothioate bond
- "m” means a form in which an existing 2'-OH is substituted with 2'-O-methyl
- "f” means, for example, in a case of fG, a form in which 2'-OH of an existing guanine (G) is substituted with fluorine.
- d refers to a form in which existing 2'-OH is substituted with -H
- EDP refers to a form in which an existing -OH group at the 5' end is substituted with E-vinylphosphonate
- GAL refers to a form in which the trivalent GalNAc derivative of Formula 1 below is bonded to the 3' end of a sense strand (see Table 16).
- Example 4 Confirmation of effect of suppressing expression of LPA of nucleic acid molecule with chemical modifications introduced for inducing RNAi
- qRT-PCR was performed to measure expression levels of LPA mRNA.
- the primary human hepatocytes were seeded in a 96 well plate at 3.0 ⁇ 10 4 cells/well, and treated with LPA GalNAc-asiRNA (200 nM).
- cell lysate was prepared using SuperPrepTM Cell lysis & RT kit for qPCR Kit II (TOYOBO, SCQ-401), and cDNA was synthesized through reverse transcription using the mRNA contained in the lysate as a template.
- LPA GalNAc-asiRNAs with the above chemical modifications introduced were administered to HDI mice introduced with LPA, and resulting changes in LPA levels were measured. Specifically, plasmids including LPA and a secreted embryonic alkaline phosphatase (SEAP) were injected into mice (Balb/c, female, 6 weeks old) through hydrodynamic injection (HDI). The mice were subcutaneously injected with LPA GalNAc-asiRNAs at a single dose of 5 mg/kg (mpk).
- SEAP secreted embryonic alkaline phosphatase
- SEAP levels in the serum were measured by using the Phospha-LightTM SEAP Reporter Gene Assay System (InvitrogenTM, T1015), and the measured values were normalized with a level of SEAP in the serum before the administration of LPA GalNAc-asiRNAs, and with a level of SEAP in the serum of a control group to which saline was administered, in order to confirm LPA levels in the serum derived from the mice.
- Apo(a)8K TG mice were generated from the frozen stock of the mouse described in the literature (Merki et al., 2011, JACC, doi:10.1016/j.jacc.2010.10.052). Blood samples were collected on Study Day -2 (pre-treatment bleeding) and serum apo(a) protein levels were determined by human lipoprotein A ELISA kit (Abcam, ab108878). Mice were grouped so that their initial apo(a) levels were even among experimental groups. Each cohort contained three animals (at least two female subjects). The GalNAc-conjugated LPA asiRNAs were administered subcutaneously at a single dose of 1 or 3 mpk on Study Day 0. Blood samples were collected weekly after dosing. Inhibition of LPA expression was determined by measuring serum apo(a) protein from treated mice. For normalization, values from pre-dose samples were averaged and set to 100%, and data were analyzed as relative values compared to the pre-dose.
- OLX706A-062-12 showed 72% inhibition of LPA expression on day 7, 74% inhibition on day 14 and 72% inhibition on day 21.
- OLX706A-033-4 showed 74% inhibition of LPA expression on day 7, 61% inhibition on day 14 and 54% inhibition on day 21.
- OLX706A-092-4 showed 76% inhibition of LPA expression on day 7, 77% inhibition on day 14 and 76% inhibition on day 21.
- OLX706A-062-12 showed 62% inhibition of LPA expression on day 7, 58% inhibition on day 14 and 57% inhibition on day 21.
- OLX706A-033-4 showed 41% inhibition of LPA expression on day 7, 28% inhibition on day 14 and 24% inhibition on day 21.
- the cynomolgus monkeys (Macaca fascicularis) were grouped so that their mean body weights (about 2 ⁇ 4 kg) were comparable between the control and experimental groups. Each cohort contained two animals.
- the GalNAc-conjugated LPA asiRNAs were administered subcutaneously at a single dose of 3 mpk on Study Day 1. Blood samples were collected on Study Days -6 and 1, and weekly after dosing. At the indicated time points, inhibition of LPA expression was determined by measuring Lp (a) serum concentration from treated NHPs using the Lp (a) ELISA (Mercodia, 10-1106-01). Values from pre-dose samples were averaged and set to 100%, and data were analyzed as relative values compared to the pre-dose.
- OLX706A-033-4 showed 89% inhibition of LPA expression on day 29, 80% inhibition on day 43 and 53% inhibition on day 64.
- OLX706A-062-12 showed 84% inhibition of LPA expression on day 29, 66% inhibition on day 43 and 43% inhibition on day 64. Data from the experiment are shown in the following Table 21 and FIG. 17.
- RNAi agent may inhibit expression of the LPA protein by binding to and degrading LPA mRNA, while alleviating side effects such as non-specific immune responses and off-target effects.
- a study was also performed to evaluate the toxicity of RNAi agents of the present disclosure in ICR mice after a single-dose subcutaneous administration followed by 8-day observation periods, no significant sign of toxicity was found.
- RNAi agent according to an aspect is effectively delivered to hepatocytes and may be usefully as a target therapeutic agent for cardiovascular diseases.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- Biotechnology (AREA)
- Heart & Thoracic Surgery (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- General Engineering & Computer Science (AREA)
- Cardiology (AREA)
- Epidemiology (AREA)
- Physics & Mathematics (AREA)
- Biophysics (AREA)
- Plant Pathology (AREA)
- Microbiology (AREA)
- Urology & Nephrology (AREA)
- Vascular Medicine (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Provided are an RNAi agent targeting a lipoprotein(a) (LPA) gene, and use thereof, more specifically, an RNAi agent including an antisense strand having sequence complementarity with an LPA mRNA sequence, and a sense strand having sequence complementarity with the antisense strand, and a pharmaceutical composition for preventing or treating cardiovascular disease, including the RNAi agent.
Description
The present disclosure relates to an RNAi agent targeting an LPA gene and use thereof, more specifically, an RNAi agent including an antisense strand having sequence complementarity with an LPA mRNA sequence, and a sense strand having sequence complementarity with the antisense strand, and a pharmaceutical composition for preventing or treating cardiovascular disease, including the RNAi agent.
Recently, development of cardiovascular diseases is increasing day by day due to changes in living environment, and a continuous increase in intake of animal-derived foods due to westernized eating habit of frequent consumption of meat, fast food, and instant food. According to the World Health Organization (WHO), cardiovascular diseases are the main cause of morbidity and mortality worldwide, and it has been reported that approximately 8.9 million deaths occurred due to cardiovascular diseases worldwide in 2019. The cardiovascular diseases refer to diseases of the circulatory system such as the heart and blood vessels, and arteriosclerosis is known as a direct and main cause. The arteriosclerosis is a disease in which cholesterol or triglyceride is accumulated in the blood vessels to narrow, harden, and block the blood vessels, and there is no sure way to reduce or eliminate the above symptoms. Currently, when there is already atherosclerosis, the focus is on preventing further progression or preventing death or complications due to the atherosclerosis, and in cases where atherosclerosis has not yet occurred, methods of prevention by controlling or removing risk factors for atherosclerosis are being applied.
Meanwhile, LPA or Lp(a) (lipoprotein(a)) is mainly expressed in a liver tissue and is known as one of genetic risk factors associated with occurrence of cardiovascular diseases. The European Society of Cardiology (ESC) and the Atherosclerosis Society (EAS) recommend measuring LPA at least once in a lifetime to determine the risk of cardiovascular disease. In addition, a genetically high level of LPA of 180 mg/dL or higher has been determined to have a similar risk of cardiovascular disease to heterozygous familial hypercholesterolemia, and a treatment target of less than 50 mg/dL has been suggested. That is, a therapy for lowering LPA may be applied to treat diseases such as stroke, atherosclerosis, thrombosis and cardiovascular diseases such as coronary heart disease, and aortic stenosis. However, since a level of LPA has very limited variability due to diet, exercise, or other life cycle changes, a new technique capable of controlling an expression level of the LPA gene is needed.
Accordingly, the present inventors, as a result of intensive research efforts to develop a new drug for treating cardiovascular diseases, developed an RNAi agent capable of specifically inhibiting expression of LPA mRNA by binding thereto, and thereby completed the present disclosure.
An object of the present disclosure is to provide an RNAi agent that specifically inhibits expression of LPA.
Another object of the present disclosure is to provide a pharmaceutical composition for preventing or treating cardiovascular diseases, including the RNAi agent, or a method of preventing or treating cardiovascular diseases including administering the pharmaceutical composition to a subject.
In order to achieve the above objects, an aspect provides an RNAi agent including: an antisense strand having sequence complementarity to an LPA mRNA sequence, and is 19 nt to 21 nt in length; and a sense strand having sequence complementarity to the antisense strand, and is 15 nt to 17 nt in length, wherein the 5' end of the antisense strand and the 3' end of the sense strand form a blunt end.
Another aspect provides a pharmaceutical composition including the RNAi agent as an active ingredient for preventing or treating cardiovascular disease.
Still another aspect provides a method of preventing or treating cardiovascular disease, including administering the RNAi agent to a subject.
Still another aspect provides use of the RNAi agent for manufacturing a pharmaceutical product for preventing or treating cardiovascular disease.
Reference will now be made in detail to embodiments, embodiments of which are illustrated in the accompanying drawings, wherein like reference numerals refer to like elements throughout. In this regard, the present embodiments may have different forms and should not be construed as being limited to the descriptions set forth herein. Accordingly, the embodiments are merely described below, by referring to the figures, to explain aspects of the present description. As used herein, the term "and/or" includes any and all combinations of at least one of the associated listed items. Expressions such as "at least one of," when preceding a list of elements, modify the entire list of elements and do not modify the individual elements of the list.
Each description and embodiment disclosed in the present application may also be applied to other descriptions and embodiments. That is, all combinations of the various elements disclosed in the application fall within the scope of the application. In addition, it should not be construed that the scope of the present application is limited by the detailed description described below.
An aspect provides an RNAi agent including: an antisense strand having sequence complementarity to an LPA mRNA sequence, and is 19 nt to 21 nt in length; and a sense strand having sequence complementarity to the antisense strand, and is 15 nt to 17 nt in length, wherein the 5' end of the antisense strand and the 3' end of the sense strand form a blunt end.
RNAi agent
The term "RNA interference (RNAi)" refers to a mechanism for suppressing expression of a target gene by inducing degradation of mRNA of the target gene by introduction of double-stranded RNA (dsRNA) composed of a strand having a sequence homology to mRNA of a target gene and a strand having a sequence complementary to the aforementioned strand.
The term "RNAi agents" or "nucleic acid molecules inducing RNAi", used herein, refers to any agent or nucleic acid molecule that is capable of inhibiting or downregulating gene expression or viral replication by mediating the RNA interference in a sequence-specific manner. The term may refer to both an individual nucleic acid molecule, a plurality of the nucleic acid molecules, or a pool of the nucleic acid molecules. In an embodiment, the RNAi agent may be siRNA.
The term "small interfering RNA (siRNA; short interfering RNA)", used herein, refers to a short double-stranded RNA (dsRNA) that sequence-specifically mediates efficient gene silencing.
The term "gene", used herein, should be considered in its broadest sense, and may encode a structural protein or a regulatory protein. In this regard, the regulatory protein includes a transcription factor, a heat shock protein, or a protein involved in DNA/RNA replication, transcription, and/or translation. In the present disclosure, the target gene to be suppressed is inherent in the viral genome, and may be integrated into an animal genome or exist as an extrachromosomal component.
The term "antisense strand", used herein, refers to a polynucleotide that is substantially or 100 % complementary to a target nucleic acid of interest, and may be complementary in whole or in part with, for example, messenger RNA (mRNA), non-mRNA RNA sequences (e.g., microRNA, piwiRNA, tRNA, rRNA and hnRNA), or coding or non-coding DNA sequences.
The term "sense strand", used herein, refers to a polynucleotide having the same nucleic acid sequence as a target nucleic acid, or a polynucleotide in whole or in part the same with, for example, messenger RNA (mRNA), non-mRNA RNA sequences (e.g., microRNA, piwiRNA, tRNA, rRNA and hnRNA), or coding or non-coding DNA sequences.
The term "complementarity" or "complementary", used herein, refers to a generally accepted meaning in the art. The term generally may refer to formation or presence of hydrogen bond(s) between one nucleic acid sequence and another nucleic acid sequence, either by traditional Watson-Crick bonding or other non-traditional types of bonding as described herein. Perfect complementarity may mean that all contiguous residues of a nucleic acid sequence hydrogen bond with the same number of contiguous residues in a second nucleic acid sequence. Partial complementarity within a nucleic acid molecule may include various mismatches or non-base-paired nucleotides (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 or more mismatches, e.g., 1 to 3 mismatches, non-nucleotide linkers, or non-base pair nucleotides). The partial complementarity may result in bulges, loops, overhangs, or blunt ends between a sense strand and an antisense strand of a nucleic acid molecule, or between an antisense strand of a nucleic acid molecule and a corresponding target nucleic acid molecule.
The term "blunt end", used herein, refers to a generally accepted meaning in the art. In relation to the RNAi agents or nucleic acid molecules herein, the term may refer to an end of a double-stranded siRNA molecule that lacks overhanging nucleotides. The siRNA molecules described herein may be such that the 5'-end of an antisense strand and the 3'-end of a sense strand form a blunt end.
RNAi agent for silencing expression of LPA
"lipoprotein(a) (LPA)" is a heterogeneous low-density lipoprotein particle mainly expressed in the liver, and is composed of apolipoprotein(a) (Apo(a)) linked to low density lipoprotein (LDL) by an ApoB polypeptide. It has been reported that a high level of LPA in the serum is not affected by diet or exercise, and is closely related to an increased risk of cardiovascular diseases by increasing possibility of developing atherosclerosis. In addition, with respect to diagnostic and prophylactic medicine, the high level of LPA in a patient's serum is known to be a significantly dominant, independent, and genetic risk factor for coronary heart disease and aortic stenosis. Therefore, the LPA gene may be a potential therapeutic target for cardiovascular diseases. The LPA protein may be interpreted to include the naturally occurring wild-type LPA and functional variants thereof, and the sequence of the LPA protein or a gene encoding the same may be obtained from a known database such as GenBank of the US National Institutes of Health.
The term "expression", used herein, refers to the generally accepted meaning in the art. The term may generally refer to a process by which a gene ultimately produces a protein. The expression includes, but is not limited to, transcription, splicing, post-transcriptional modification, or translation. As used herein, an expression level may be determined or monitored by detection of an mRNA level or a protein level.
The term "inhibition" or "reduction", as used in reference to LPA gene expression in a subject, refers to a statistically significant decrease compared to an untreated or normal control group. The reduction may be, for example, at least 30 %, 35 %, 40 %, 45 %, 50 %, 55 %, 60 %, 65 %, 70 %, 75 %, 80 %, 85 %, 90 %, or 95 % or more, but it may be below the detection limit, depending on the detection or measurement method.
siRNA is a small interfering RNA and is involved in RNA interference (RNAi). RNAi is an intracellular gene regulation mechanism first discovered in 1998 in Caenorthabditis elegans, and its mechanism of action is known to induce target gene degradation by an antisense strand, which is one of RNA double strands which are introduced into the cell, that complementarily binds to mRNA of a target gene, and RNAi has recently been one of the most popular candidate technologies for developing new drugs.
However, contrary to this possibility, side effects and disadvantages of siRNA have been continuously reported. For the development of RNAi-based therapeutic agents, it is necessary to overcome barriers such as 1) absence of an effective delivery system, 2) off-target effect, 3) induction of immune response, and 4) saturation of RNAi machinery in cells. Although siRNA is an effective method for directly regulating expression of a target gene, it is difficult to develop a therapeutic agent due to these issues. In this regard, asymmetric shorter duplex siRNA (asiRNA) is an asymmetric RNAi-induced structure having a shorter double helix length compared to the 19+2 structure of siRNA in the art. asiRNA technology overcame issues such as off-target effect, saturation of RNAi mechanism, and immune response by TLR3, which are identified in the existing siRNA structure technology, and accordingly, it is possible to develop new RNAi drugs with low side effects.
Based on this, asymmetric siRNA including a sense strand and an antisense strand complementary to the sense strand is presented in this example, and siRNA according to an example does not cause issues such as off-target effect, saturation of RNAi machinery, etc., and thereby, may inhibit expression of an LPA gene to a desired degree while stably maintaining high delivery efficiency.
In an example, asymmetric siRNAs (asiRNAs) targeting LPA were designed and prepared, and after transfection of the asiRNAs into cells expressing LPA, nucleic acid molecules for inducing RNAi with excellent knockdown efficiency, i.e., LPA asiRNAs, were selected.
In an embodiment, the RNAi agent may be characterized in that the sense strand has a length of 15 nt to 17 nt, and the antisense strand has a length of 19 nt to 21 nt. More preferably, a length of the sense strand may be 16 nt, and a length of the antisense strand complementary thereto may be 19 nt, 20 nt, or 21 nt, but is not limited thereto.
The 5'-end of the antisense strand and the 3'-end of the sense strand may form a blunt end. The 3' end of the antisense strand may include, for example, an overhang of 2 nt to 6 nt.
In an embodiment, the sense strand may be a sequence selected from the sense strand sequences listed in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, and Table 9, and the antisense strand may include a sequence selected from the antisense strand sequences listed in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, and Table 9.
In an embodiment, the sense strand may be, for example, at least one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 31, SEQ ID NO: 35, SEQ ID NO: 39, SEQ ID NO: 47, SEQ ID NO: 51, SEQ ID NO: 53, SEQ ID NO: 63, SEQ ID NO: 65, SEQ ID NO: 67, SEQ ID NO: 71, SEQ ID NO: 77, SEQ ID NO: 79, SEQ ID NO: 83, SEQ ID NO: 85, SEQ ID NO: 87, SEQ ID NO: 103, SEQ ID NO: 123, SEQ ID NO: 125, SEQ ID NO: 127, SEQ ID NO: 183, SEQ ID NO: 215, SEQ ID NO: 339, SEQ ID NO: 341, SEQ ID NO: 373, SEQ ID NO: 375, SEQ ID NO: 471, SEQ ID NO: 477, SEQ ID NO: 495, SEQ ID NO: 701, SEQ ID NO: 709, SEQ ID NO: 711, SEQ ID NO: 713, SEQ ID NO: 715, SEQ ID NO: 735, SEQ ID NO: 737, SEQ ID NO: 739, SEQ ID NO: 743 and SEQ ID NO: 775.
In an embodiment, the sense strand may be, for example, at least one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 63, SEQ ID NO: 65, SEQ ID NO: 67, SEQ ID NO: 71, SEQ ID NO: 85, SEQ ID NO: 123, SEQ ID NO: 127, SEQ ID NO: 183, SEQ ID NO: 215, SEQ ID NO: 339, SEQ ID NO: 341, SEQ ID NO: 373, and SEQ ID NO: 737, for example, it may be any one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 65, SEQ ID NO: 123, and SEQ ID NO: 183. Preferably, the sense strand may be any one selected from SEQ ID NO: 65, SEQ ID NO: 123, and SEQ ID NO: 183.
In an embodiment, the antisense strand may be, for example, at least one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 32, SEQ ID NO: 36, SEQ ID NO: 40, SEQ ID NO: 48, SEQ ID NO: 52, SEQ ID NO: 54, SEQ ID NO: 64, SEQ ID NO: 66, SEQ ID NO: 68, SEQ ID NO: 72, SEQ ID NO: 78, SEQ ID NO: 80, SEQ ID NO: 84, SEQ ID NO: 86, SEQ ID NO: 88, SEQ ID NO: 104, SEQ ID NO: 124, SEQ ID NO: 126, SEQ ID NO: 128, SEQ ID NO: 184, SEQ ID NO: 216, SEQ ID NO: 340, SEQ ID NO: 342, SEQ ID NO: 374, SEQ ID NO: 376, SEQ ID NO: 472, SEQ ID NO: 478, SEQ ID NO: 496, SEQ ID NO: 702, SEQ ID NO: 710, SEQ ID NO: 712, SEQ ID NO: 714, SEQ ID NO: 716, SEQ ID NO: 736, SEQ ID NO: 738, SEQ ID NO: 740, SEQ ID NO: 744 and SEQ ID NO: 776.
In an embodiment, the antisense strand may be, for example, at least one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 64, SEQ ID NO: 66, SEQ ID NO: 68, SEQ ID NO: 72, SEQ ID NO: 86, SEQ ID NO: 124, SEQ ID NO: 128, SEQ ID NO: 184, SEQ ID NO: 216, SEQ ID NO: 340, SEQ ID NO: 342, SEQ ID NO: 374, and SEQ ID NO: 738, for example, the antisense strand may be any one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 66, SEQ ID NO: 124, and SEQ ID NO: 184. Preferably, the antisense strand may be any one selected from SEQ ID NO: 66, SEQ ID NO: 124, and SEQ ID NO: 184. In a preferred embodiment, the RNAi agent has a sense strand as shown in SEQ ID NO: 65 and an antisense strand as shown in SEQ ID NO: 66. In an embodiment, the RNAi agent has a sense strand as shown in SEQ ID NO: 123 and an antisense strand as shown in SEQ ID NO: 124. In an embodiment, the RNAi agent has a sense strand as shown in SEQ ID NO: 183 and an antisense strand as shown in SEQ ID NO: 184.
RNAi agent with chemical modifications introduced
In the RNAi agent, the sense strand or the antisense strand may include one or more chemical modifications. In one aspect, the present disclosure provides novel chemical modification patterns for improving the properties and effects of RNAi agents.
General siRNA cannot pass through the cell membrane due to high negative charge and a high molecular weight of the phosphate backbone structure, and is rapidly degraded and eliminated from the blood, making it difficult to deliver a sufficient amount to an actual target site for RNAi induction. Currently, for in vitro delivery, many high-efficiency delivery methods using cationic lipids and cationic polymers have been developed, however, for in vivo, it is difficult to deliver siRNA with as high efficiency as in vitro, and there is an issue of reduced siRNA delivery efficiency due to interaction with various proteins existing in the living body.
Accordingly, provided in the example is an RNAi agent having improved hepatocyte targeted delivery ability by introduction of chemical modifications to the asymmetric siRNA structure, more specifically, provided is an asymmetric siRNA construct (GalNAc asymmetric siRNA, GalNAc-asiRNA) capable of effective intracellular delivery without a separate carrier.
In the present disclosure, the chemical modification in the sense strand or the antisense strand may include one or more selected from: binding with an N-acetylgalactosamine (GalNAc) derivative, or a cell-penetrating peptide; modification of a nucleotide bond with phosphorothioate, boranophosphate, or methyl phosphonate; substitution of an -OH group at the 2' carbon position of the sugar structure of a nucleotide with -H, -CH3 (methyl), -OCH3 (-O-methyl), -NH2, -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and substitution of an -OH group at the 5' end with E-vinylphosphonate.
In an embodiment, the N-acetylgalactosamine (GalNAc) derivative may have a structure of Formula 1 below. The N-acetylgalactosamine (GalNAc) derivative recognizes asialoglycoprotein (ASGPR) receptors on a surface of a hepatocyte and helps RNAi agents to flow into the hepatocyte, that is, acts as an ASGPR targeting moiety, and thus, the RNAi agent, in which the N-acetylgalactosamine (GalNAc) derivative is bound to an end have improved delivery to hepatocytes and may provide an effective targeted treatment for liver diseases.
Formula 1
In an embodiment, the sense strand may include one or more chemical modifications selected from: modification of 2 to 4 nucleotide bonds adjacent to the 5' end with phosphorothioate, boranophosphate, or methyl phosphonate; substitution of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide with -H, -CH3 (methyl), -OCH3 (-O-methyl), -NH2, -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and binding with an N-acetylgalactosamine (GalNAc) derivative, or a cell penetrating peptide at the 3' end.
In an embodiment, the antisense strand may include one or more chemical modifications selected from: modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end with phosphorothioate, boranophosphate, or methyl phosphonate; substitution of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide with -H, -CH3 (methyl), -OCH3 (-O-methyl), -NH2, -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and substitution of an -OH group at the 5' end to E-vinylphosphonate.
In another embodiment, the RNAi agent may include at least one modification selected from: modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end in a sense or an antisense strand to phosphorothioate; modification of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide in a sense strand or an antisense strand to -H, -OCH3 (-O-methyl), or -F; binding to an N-acetylgalactosamine (GalNAc) derivative at the 3' end of the sense strand; and substitution of an -OH group at the 5' end of an antisense strand to E-vinylphosphonate.
In another embodiment, the RNAi agent as described above having an antisense strand and sense strand, wherein in the antisense strand, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand, from 5' to 3', positions 2, 4, 5, 6 are modified by 2'-fluoro, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative.
In an embodiment, the RNAi agent as described above having a sense strand, from 5' to 3': positions 2, 4, 5, 6, 14 and 16 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe. In an embodiment, the RNAi agent as described above having a sense strand, from 5' to 3': positions 2, 4, 5, 6 and 12 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe. In some embodiment, the chemical modification pattern is: in the antisense strand of a RNAi agent, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are modified as deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand of a RNAi agent, from 5' to 3', positions 2, 4, 5, 6, 14 and 16 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative. In some embodiment, the chemical modification pattern is: in the antisense strand of a RNAi agent, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are modified as deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand of a RNAi agent, from 5' to 3', positions 2, 4, 5, 6 and 12 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative.
In an embodiment, in the sense strand of the RNAi agent as described above, from 5' to 3' may be modified as:
(m)*(f)*(m)(f)(f)(f)(m)(m)(m)(m)(m)(m)(m)(f)(m)(f)(GAL);
and in the antisense strand of the RNAi agent as described above, from 5' to 3' may be modified as:
[EVP](m)*(f)*(m)(m)(d)(m)(d)(m)(m)(m)(m)(m)(m)(f)(m)*(f)*(m)*(m)*(m)*(m)*(m);
wherein, * denotes a phosphorothioated bond, (m) denotes 2'-O-methyl, (f) denotes 2'-fluoro, (d) denotes a form in which existing 2'-OH is substituted with -H, [EVP] denotes E-vinylphosphonate, and (GAL) denotes a trivalent GalNAc derivative. Preferably, the trivalent GalNAc derivative is Formula 1.
In an embodiment, in the sense strand of the RNAi agent as described above, from 5' to 3' may be modified as:
(m)*(f)*(m)(f)(f)(f)(m)(m)(m)(m)(m)(f)(m)(m)(m)(m)(GAL);
and in the antisense strand of the RNAi agent as described above, from 5' to 3' may be modified as:
[EVP](m)*(f)*(m)(m)(d)(m)(d)(m)(m)(m)(m)(m)(m)(f)(m)*(f)*(m)*(m)*(m)*(m)*(m);
wherein, * denotes a phosphorothioated bond, (m) denotes 2'-O-methyl, (f) denotes 2'-fluoro, (d) denotes a form in which existing 2'-OH is substituted with -H, [EVP] denotes E-vinylphosphonate, and (GAL) denotes a trivalent GalNAc derivative. Preferably, the trivalent GalNAc derivative is Formula 1.
In an embodiment, the sense strand may be a sequence selected from the sense strand sequences listed in Table 15, and the antisense strand may include a sequence selected from the antisense strand sequences listed in Table 15. In a preferred embodiment, the RNAi agent is selected from the GalNAc-asiRNA conjugates listed in Table 15.
In an embodiment, the sense strand may be any one selected from: (A) to (D) in the table below, and the antisense strand may be any one selected from: (a) to (d) in the table below.
In the above sequences, * denotes a phosphorothioated bond, m denotes 2'-O-methyl, f denotes 2'-fluoro, d denotes a form in which existing 2'-OH is substituted with -H, EVP denotes E-vinylphosphonate, and GAL denotes a trivalent GalNAc derivative of Formula 1.
Specifically, the sense strand and the antisense strand of the RNAi agent may be sequences of the sense strand and antisense strand below selected from the table above, for example, the RNAi agent may have a combination of a sense strand (A) and an antisense strand (a); a combination of a sense strand (B) and an antisense strand (b); a combination of a sense strand (C) and an antisense strand (c); or a combination of a sense strand (D) and an antisense strand (d).
Another aspect provides a pharmaceutical composition including the RNAi agent as an active ingredient, for preventing or treating cardiovascular disease.
Still another aspect provides use of the RNAi agent for manufacturing a pharmaceutical product, for preventing or treating cardiovascular disease.
Since the pharmaceutical composition contains or uses the above-described RNAi agent as it is, descriptions thereof are omitted in order to avoid excessive complexity of the present specification.
Cardiovascular disease
The pharmaceutical composition may be utilized as an active ingredient of a pharmaceutical composition for preventing or treating cardiovascular disease by inhibiting expression of an LPA gene.
The cardiovascular disease may collectively refer to primary diseases directly caused by various causes such as abnormality, dysfunction, and damage of the circulatory system in vivo, as well as secondary diseases derived from the primary diseases. The cardiovascular disease is a disease closely related to an expression level of the LPA gene, and may be for example, any one selected from: Berger's disease, peripheral artery disease, coronary artery disease, metabolic syndrome, acute coronary syndrome, aortic valve stenosis, myocardial infarction, aortic valve regurgitation, aortic dissection, retinal artery occlusion, cerebrovascular disease, mesenteric ischemia, superior mesenteric artery occlusion, renal artery stenosis, stable/unstable angina, acute coronary syndrome, heterozygous or homozygous familial hypercholesterolemia, hyperaphobeta lipoproteinemia, cerebrovascular atherosclerosis, and venous thrombosis, but is not limited thereto.
Pharmaceutical composition
The term "effective ingredient", used herein, means an appropriate effective amount of an ingredient that brings about a beneficial or desirable clinical or biochemical outcome. Specifically, the term may refer to an agent, an active agent, or an RNAi agent of an effective amount.
The effective amount may be administered one or more times, and unlimitedly may be an appropriate amount for preventing a disease, alleviating symptoms, reducing an extent of the disease, stabilizing (i.e., not exacerbating) the disease state, delaying or reducing a rate of disease progression, or improving or temporarily alleviating and ameliorating (partially or fully) a disease state.
The term "prevention", used herein, refers to any action that blocks an occurrence of a disease in advance, suppresses a disease, or delays progression thereof. For example, the term refers to preventing the cardiovascular disease or occurrence of characteristics thereof, interrupting the occurrence, or defending or protecting from the cardiovascular disease or occurrence of the characteristics thereof.
The term "treatment", used herein, refers to both therapeutic treatment and preventive or prophylactic measures. In addition, it refers to any action in which the symptoms of a disease are improved or beneficially changed. For example, the term refers to preventing, reducing or improving the cardiovascular disease or characteristics thereof, or delaying (attenuating) progression of the cardiovascular disease or characteristics thereof in a subject.
The term "effective amount", used herein, refers to a generally accepted meaning in the art. The term generally refers to an amount of a molecule, compound, or construct that elicits an intended biological response (e.g., a beneficial response) in a cell, tissue, a system, an animal, or a human, sought by researchers, veterinarians, physicians, or other clinicians, etc. Specifically, a "therapeutically effective amount" refers to an amount of a molecule, compound, or construct that is capable of eliciting a desirable medical response to the extent that a particular clinical treatment may be considered effective, due to, for example, a therapeutically relevant change in a measurable parameter associated with the disease or disorder. The therapeutically effective amount of a drug for treatment of a disease or a disorder may be an amount necessary to cause a therapeutically relevant change in the parameter.
A method of administering the pharmaceutical composition may be determined by a person skilled in the art based on the symptoms of the patient and the severity of the disease. In addition, the pharmaceutical composition may be formulated in various forms such as powders, tablets, capsules, solutions, injections, ointments, syrups, etc., and may be provided in unit-dose or multi-dose containers, for example, sealed ampoules and bottles.
The pharmaceutical composition of the present disclosure may be administered orally or parenterally. Routes of administration of the composition according to the present disclosure may be, for example, oral, intravenous, intramuscular, intraarterial, intramedullary, intrathecal, intracardiac, transdermal, subcutaneous, intraperitoneal, intestinal, sublingual, or topical, but are not limited thereto. Dosage of the composition according to the present disclosure varies depending on the patient's weight, age, sex, health status, diet, administration time, method, excretion rate, or severity of disease, etc. and may be readily determined by those of average skill in the art. In addition, the composition of the present disclosure may be formulated into a suitable formulation for clinical administration using known techniques.
Still another aspect provides a method of preventing or treating cardiovascular disease, including administering the RNAi agent to a subject.
Since the method of treating cardiovascular disease includes or uses the above-described pharmaceutical composition as it is, descriptions thereof are omitted in order to avoid excessive complexity of the present specification.
The term "subject", used herein, refers to a subject in need of treatment for a disease, specifically, cardiovascular disease, and more specifically, the term may include all mammals such as a human or a non-human primate, a mouse, a dog, a cat, a horse, a cow, sheep, a pig, a goat, a camel, and an antelope.
An RNAi agent according to an aspect may inhibit expression of the LPA protein by binding to and degrading LPA mRNA, while alleviating side effects such as non-specific immune responses and off-target effects.
In addition, the RNAi agent according to an aspect is effectively delivered to hepatocytes and may be usefully as a target therapeutic agent for cardiovascular diseases.
The above and other aspects, features, and advantages of certain embodiments of the disclosure will be more apparent from the following description taken in conjunction with the accompanying drawings, in which:
FIG. 1 is a diagram schematically showing a structure of LPA asiRNA consisting of a 16-mer sense strand and a 21-mer antisense strand;
FIG. 2a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 55 types of LPA asiRNAs (asiLPA-001 to asiLPA-055) according to an aspect, at a concentration of 10 nM;
FIG. 2b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-056 to asiLPA-109) according to an aspect, at a concentration of 10 nM;
FIG. 3a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 55 types of LPA asiRNAs (asiLPA-110 to asiLPA-164) according to an aspect, at a concentration of 10 nM;
FIG. 3b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-165 to asiLPA-218) according to an aspect, at a concentration of 10 nM;
FIG. 4a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 55 types of LPA asiRNAs (asiLPA-219 to asiLPA-273) according to an aspect, at a concentration of 10 nM;
FIG. 4b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-274 to asiLPA-327) according to an aspect, at a concentration of 10 nM;
FIG. 5a shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 54 types of LPA asiRNAs (asiLPA-328 to asiLPA-381) according to an aspect, at a concentration of 10 nM;
FIG. 5b shows results of confirming expression levels of LPA mRNA after respectively treating Hep3B cells with 53 types of LPA asiRNAs (asiLPA-382 to asiLPA-434) according to an aspect, at a concentration of 10 nM;
FIG. 6 shows results of confirming expression levels of LPA mRNA, after respectively treating Hep3B cells with 65 types of LPA asiRNAs, at a concentration of 1 nM;
FIG. 7 shows results of confirming expression levels of LPA mRNA, after respectively treating Hep3B cells with 65 types of LPA asiRNAs, at a concentration of 1 nM;
FIG. 8 shows results of confirming expression levels of LPA mRNA, after respectively treating Hep3B cells with 40 types of LPA asiRNAs according to an aspect, at a concentration of 0.1 nM;
FIG. 9 shows results of confirming expression levels of LPA mRNA, after respectively treating primary human hepatocytes with 40 types of LPA asiRNAs according to an aspect, at a concentration of 10 nM;
FIG. 10 shows results of confirming expression levels of LPA mRNA, after respectively treating primary cynomolgus monkey hepatocytes with 40 types of LPA asiRNAs according to an aspect, at a concentration of 10 nM;
FIG. 11 shows results of confirming changes in LPA expression levels over time, after subcutaneously administering 6 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-032-1, OLX706A-033-1, OLX706A-034-1, OLX706A-036-1, OLX706A -043-1) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P1) HDI mice at a single dose of 8 mg/kg (mpk);
FIG. 12 shows results of confirming changes in LPA expression levels over time, after subcutaneously administering 9 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-007-1, OLX706A-033-1, OLX706A-043-1, OLX706A-062-1, OLX706A-064-1, OLX706A-092-1, OLX706A-108-1, OLX706A-369-1) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P2) HDI mice at a single dose of 8 mg/kg (mpk);
FIG. 13 shows results of confirming changes in LPA expression levels over time, after subcutaneously administering 5 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-064-1, OLX706A-170-1, OLX706A-171-1, OLX706A-187-1) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P3) HDI mice at a single dose of 8 mg/kg (mpk);
FIG. 14 shows results of confirming expression levels of LPA mRNA, after respectively treating primary human hepatocytes with 14 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-2, OLX706A-062-12, OLX706A-092-4, OLX706A-033-1, OLX706A-062-1, OLX706A-092-1, OLX706A-006-12, OLX706A-007-12, OLX706A-007-4, OLX706A-033-12, OLX706A-062-4, OLX706A-006-1, OLX706A-007-1) according to an aspect, into which chemical modifications are introduced, at a concentration of 200 nM; and
FIG. 15 shows results of confirming changes in LPA expression levels, after subcutaneously administering 6 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, OLX706A-092-4) according to an aspect, into which chemical modifications are introduced, to SEAP-LPA (P2) HDI mice at a single dose of 5 mg/kg (mpk).
FIG. 16a shows results of confirming changes in Apo(a) expression levels over time, after subcutaneously administering 4 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-2, OLX706A-062-12, OLX706A-092-4) according to an aspect, into which chemical modifications are introduced, to Apo(a) TG mice at a single dose of 3 mg/kg (mpk);
FIG. 16b shows results of confirming changes in Apo(a) expression levels over time, after subcutaneously administering 3 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-2, OLX706A-062-12) according to an aspect, into which chemical modifications are introduced, to Apo(a) TG mice at a single dose of 1 mg/kg (mpk);
FIG. 17 shows results of confirming changes in Lp(a) expression levels over time, after subcutaneously administering 2 types of LPA GalNAc-asiRNAs (OLX706A-033-4, OLX706A-062-12) according to an aspect, into which chemical modifications are introduced, to cynomolgus monkey at a single dose of 3 mg/kg (mpk).
Hereinafter, the present disclosure will be described in more detail through examples. However, these examples are intended to illustrate the present disclosure, and the scope of the present disclosure is not limited to these examples.
Example 1: Screening of nucleic acid molecules for inducing RNAi targeting LPA
1-1. Design and preparation of LPA asiRNA
In this example, in order to secure a double-stranded nucleic acid molecule that induces high-efficiency RNA interference targeting LPA, after selecting target sequences for the LPA gene, LPA asymmetric siRNAs (LPA asiRNAs) for the same were designed (sense strand (16-mer), antisense strand (21-mer), see FIG. 1).
Specifically, after acquiring LPA gene information through searching the NCBI database, a total of 434 asiRNAs were designed in consideration of animal experiments, and synthesized by OliX US, at a 4-nmole scale. Thereafter, the synthesized asiRNAs were gel-electrophoresed by using 15 % polyacrylamide gel, and then quality control (QC) was performed by using a ChemiDoc UV transilluminator (Biorad). Sequence information of the LPA asiRNA prepared according to the above method is as shown in Tables 1 to 9 below.
[Table 1]
[Table 2]
[Table 3]
[Table 4]
[Table 5]
[Table 6]
[Table 7]
[Table 8]
[Table 9]
1-2. Selection of 130 types of LPA asiRNAs
Hep3B cells, which are human liver cancer cells, were transfected with the 434 types of LPA asiRNAs at a concentration of 10 nM, and a dual-luciferase reporter assay was performed to measure expression levels of LPA mRNA. Specifically, LPA asiRNA (10 nM) and psiCHECK-2-LPA plasmid DNA (10 ng/well) were delivered into Hep3B cells by using lipofectamine 2000 (Invitrogen, 11668019, 0.2 μl/well). Then, the expression levels of LPA mRNA were evaluated by measuring intensities of renilla luciferases (expressed by LPA mRNA) and firefly luciferases by using a dual luciferase reporter assay system (Promega, Cat.E1980), and normalizing the expression levels of LPA mRNA with a level of intensity of firefly luciferases constitutively expressed in a state in which psiCHECK-2-LPA plasmid DNA is introduced.
As a result, as shown in FIGS. 2 to 5, an effect of inhibiting expression of renilla luciferases according to LPA asiRNA treatment was confirmed. In addition, 130 types of LPA asiRNAs showing relatively good expression inhibition efficiency were identified (asiLPA-001, 002, 003, 004, 005, 006, 007, 008, 009, 010, 013, 016, 017, 018, 019, 020, 021, 022, 023, 024, 025, 026, 027, 031, 033, 032, 034, 035, 036, 038, 039, 040, 041, 042, 043, 044, 050, 051, 052, 053, 054, 055, 056, 059, 061, 062, 063, 064, 066, 070, 079, 088, 092, 098, 102, 105, 108, 110, 119, 120, 121, 124, 129, 130, 131, 146, 147, 149, 152, 161, 167, 168, 170, 171, 178, 187, 188, 204, 213, 214, 216, 217, 221, 230, 236, 238, 239, 240, 244, 245, 248, 249, 250, 251, 258, 265, 275, 334, 343, 349, 351, 353, 354, 355, 356, 357, 358, 359, 360, 361, 362, 364, 365, 367, 368, 369, 370, 371, 372, 373,374, 379, 388, 400, 406, 411, 418, 422, 431, 434).
1-3. Selection of 40 types of LPA asiRNAs
Hep3B cells, which are human liver cancer cells, were transfected with the 130 types of selected LPA asiRNAs at a concentration of 1 nM, and a dual-luciferase reporter assay was performed to measure expression levels of LPA mRNA, in the same manner as in Example 1-2.
As a result, as shown in FIGS. 6 and 7, an effect of inhibiting expression of LPA mRNA according to LPA asiRNA treatment was confirmed. In addition, 40 types of LPA asiRNAs showing relatively good expression inhibition efficiency were identified (asiLPA-002, 006, 007, 008, 016, 018, 020, 024, 026, 027, 032, 033, 034, 036, 039, 040, 042, 044, 052, 063, 064, 092, 102, 108, 170, 171, 187, 188, 213, 236, 351, 355, 356, 357, 365, 368, 369, 370, 372, 388).
1-4. Confirmation of effect of suppressing expression of 40 types of LPA asiRNAs
Hep3B cells, primary human hepatocytes, or primary cynomolgus monkey hepatocytes were transfected with the 40 types of LPA asiRNAs, respectively, and dual-luciferase reporter assay, or qRT-PCR was performed to measure expression levels of LPA mRNA.
Specifically, for experiments on Hep3B cells, the Hep3B cells were transfected with the total of 40 types of LPA asiRNAs at a concentration of 0.1 nM, and expression levels of LPA mRNA were measured in the same manner as in Example 1-2. In addition, for experiments on primary human hepatocytes, the primary human hepatocytes were transfected with the total of 40 types of LPA asiRNAs at a concentration of 10 nM, and expression levels of LPA mRNA were measured by performing qRT-PCR. Specifically, the primary human hepatocytes were seeded in a 96 well plate at 1.0Х104 cells/well, and transfected with LPA asiRNA (10 nM), by using lipofectamine 2000 (Invitrogen, 13778150). 24 hours after the transfection, cell lysate was prepared by using SuperPrepTM Cell lysis & RT kit for qPCR Kit II (TOYOBO, SCQ-401), and cDNA was synthesized through reverse transcription using the mRNA included in the lysate as a template. Then, by using the synthesized cDNA as a template, quantitative PCR was performed by using TaqMan® Gene Expression Assays, and a probe (Hs00916691_m1: human LPA, Hs03928985_g1: RNA18S5, Applied Biosystems). Then, expression levels of LPA mRNA were identified by using a CFXOpus 384 Real-Time PCR System (Bio-Rad). In addition, for experiments on primary cynomolgus monkey hepatocytes, the primary cynomolgus monkey hepatocytes were also transfected with the 40 types of LPA asiRNAs at a concentration of 10 nM, and expression levels of LPA mRNA were measured by performing qRT-PCR. Specifically, the primary cynomolgus monkey hepatocytes were seeded in a 96 well plate at 4.5Х104 cells/well, and transfected with LPA asiRNA (10 nM), by using lipofectamine 2000 (Invitrogen, 13778150). 24 hours after the transfection, cell lysate was prepared using SuperPrepTM Cell lysis & RT kit for qPCR Kit II (TOYOBO, SCQ-401), and cDNA was synthesized through reverse transcription using the mRNA contained in the lysate as a template. Then, by using the synthesized cDNA as a template, quantitative PCR was performed by using TaqMan® Gene Expression Assays, and a probe (Mf02789271_m1: cyno LPA, Mf04354341_g1: ACTB, Applied Biosystems). Then, expression levels of LPA mRNA were identified by using a CFX Connect Real-Time PCR System (Bio-Rad).
As a result, as shown in FIGS. 8 to 10, an effect of inhibiting expression of LPA mRNA according to LPA asiRNA treatment was confirmed in each of Hep3B cells, primary human hepatocytes, or primary cynomolgus monkey hepatocytes.
In view of all the experimental results of Example 1, 40 types of LPA asiRNAs showing relatively good expression inhibition efficiency were identified (asiLPA-006, 007, 016, 018, 020, 024, 026, 027, 032, 033, 034, 036, 039, 040, 042, 043, 044, 052, 062, 063, 064, 092, 108, 170, 171, 187, 188, 236, 239, 248, 351, 355, 356, 357, 358, 368, 369, 370, 372, 388) for further screening.
Example 2: Screening by using RNAi-inducing nucleic acid molecules with chemical modifications introduced
2-1. Nucleic acid molecules for inducing RNAi with chemical modifications introduced
In the present example, as a double-stranded nucleic acid molecule that induces high-efficiency RNA interference targeting LPA, LPA GalNAc-asiRNAs with chemical modifications introduced were designed. Specifically, in consideration of the experimental results of Example 1, 15 types of LPA asiRNAs (asiLPA-006, 007, 032, 033, 034, 036, 043, 062, 064, 092, 108, 170, 171, 187, 369) was selected as basic skeletons. Then, 15 types of LPA GalNAc-asiRNAs were designed with each of the basic skeletons, into which are introduced 1) substitution of an -OH group at the 2' carbon position of the sugar structure in the nucleotides of the sense and antisense strands with -OMe or -F, 2) introduction of phosphorothioate modifications in the backbone of the 5' end or 3' end of the antisense strand, and the 5' end of the sense strand, 3) introduction of phosphate at the 5' end of the antisense strand, and 4) introduction of GalNAc at the 3' end of the sense strand.
Sequence information of the chemically modified LPA GalNAc-asiRNA prepared in this example is shown in Table 10 below. Specifically, in Table 10, "*" means a form in which an existing phosphodiester bond is substituted with a phosphorothioate bond, and "m" means a form in which an existing 2'-OH is substituted with 2'-O-methyl. In addition, "f" means, for example, in a case of fG, a form in which 2'-OH of an existing guanine (G) is substituted with fluorine. In addition, "Phos" refers to a form in which 5'-OH is substituted with phosphate, and "GAL" refers to a form in which a trivalent GalNAc derivative of Formula 1 is bonded to the 3' end of the sense strand (see Table 11).
[Table 10]
[Table 11]
Formula 1
2-2. Confirmation of LPA expression suppression effect by using animal models
The 15 types of LPA GalNAc-asiRNAs introduced with the above chemical modifications were respectively administered to SEAP-LPA HDI mice in which 3 types of plasmids (P1, P2, and P3) were injected, and accordingly, changes in LPA levels were measured. In this regard, the 3 types of plasmids were prepared by dividing the LPA from a Kringle IV site to a protease domain site into P1 (from Kringle IV-1 to Kringle IV-6), P2 (from Kringle IV-7 to Kringle IV-34), and P3 (from Kringle IV-35 to a protease domain). Specifically, 3 types of plasmids including LPA and a secreted embryonic alkaline phosphatase (SEAP) were respectively injected into mice (Balb/c, female, 6 weeks old) through hydrodynamic injection (HDI). The mice were subcutaneously injected with LPA GalNAc-asiRNAs at a single dose of 8 mg/kg (mpk). On days 2, 7, and 21 from the day of subcutaneous injection into the mice, SEAP levels in the serum were measured by using the Phospha-Light™ SEAP Reporter Gene Assay System (Invitrogen™, T1015), and the levels of LPA in the serum derived from mice were confirmed by normalizing the measured value with a level of SEAP in the serum of a control group to which saline was administered on the day the LPA levels were measured, and with a SEAP level in the serum before the administration of LPA GalNAc-asiRNAs.
As a result, as shown in FIGS. 11 to 13 and Tables 12 to 14, an effect of inhibiting expression of SEAP reporters was confirmed according to treatment of 15 types of LPA GalNAc-asiRNAs (OLX706A-006-1, OLX706A-007-1, OLX706A-032-1, OLX706A-033-1, OLX706A-034-1, OLX706A-036-1, OLX706A-043-1, OLX706A-062-1, OLX706A-064-1, OLX706A-092-1, OLX706A-108-1, OLX706A-170-1, OLX706A-171-1, OLX706A-187-1, OLX706A-369-1). Specifically, an effect of inhibiting expression of SEAP reporters according to treatment of LPA GalNAc-asiRNAs was confirmed with FIG.11 and Table 12 in mice with P1 plasmids introduced, with FIG. 12 and Table 13 in mice with P2 plasmids introduced, and with FIG. 13 and Table 14 in mice introduced with P3 plasmids.
[Table 12]
[Table 13]
[Table 14]
Example 3: Preparation of nucleic acid molecules for inducing RNAi with chemical modifications introduced
In the present example, as a double-stranded nucleic acid molecule that induces high-efficiency RNA interference targeting LPA, LPA GalNAc-asiRNAs with chemical modifications introduced were designed. For this, different types of LPA GalNAc-asiRNAs were designed, wherein in the LPA GalNAc-asiRNAs, 1) an -OH group at the 2' carbon position of the sugar structure in the nucleotides of the sense and antisense strands is substituted with -OMe or -F, 2) a phosphorothioate modification is introduced to a backbone of the 5' or 3' end of the antisense strand and the 5' end of the sense strand, 3) E-vinylphosphonate is introduced at the 5' end of the antisense strand, and 4) GalNAc is introduced at the 3' end of the sense strand. Specifically, the 5 types of LPA asiRNAs (asiLPA-006,asiLPA-007,asiLPA-033, asiLPA-062, and asiLPA-092) of Example 2 having an effective inhibitory effect on LPA mRNA expression were used as a basic framework, and various modification patterns designed from the chemical modifications of Table 16 were introduced to the basic frameworks to obtained many different GalNAc-asiRNAs conjugates, e.g., OLX706A-006-12, OLX706A-007-12, OLX706A-007-4, OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, and OLX706A-092-4 were prepared. Meanwhile, OLX706A-006-1, OLX706A-007-1, OLX706A-033-1, OLX706A-062-1, and OLX706A-092-1 in this Example were prepared in the same manner as in Example 2.
Sequence information of the chemically modified LPA GalNAc-asiRNA prepared in this example is shown in Table 15 below. Specifically, in Table 15, "*" means a form in which an existing phosphodiester bond is substituted by a phosphorothioate bond, and "m" means a form in which an existing 2'-OH is substituted with 2'-O-methyl. In addition, "f" means, for example, in a case of fG, a form in which 2'-OH of an existing guanine (G) is substituted with fluorine. In addition, "d" refers to a form in which existing 2'-OH is substituted with -H, "EVP" refers to a form in which an existing -OH group at the 5' end is substituted with E-vinylphosphonate, and "GAL" refers to a form in which the trivalent GalNAc derivative of Formula 1 below is bonded to the 3' end of a sense strand (see Table 16).
[Table 15]
[Table 16]
Example 4: Confirmation of effect of suppressing expression of LPA of nucleic acid molecule with chemical modifications introduced for inducing RNAi
4-1. Confirmation of LPA expression suppression effect
in vitro
After treating primary human hepatocytes with the above GalNAc-asiRNAs, qRT-PCR was performed to measure expression levels of LPA mRNA. Specifically, the primary human hepatocytes were seeded in a 96 well plate at 3.0Х104 cells/well, and treated with LPA GalNAc-asiRNA (200 nM). 24 hours after the transfection, cell lysate was prepared using SuperPrep™ Cell lysis & RT kit for qPCR Kit II (TOYOBO, SCQ-401), and cDNA was synthesized through reverse transcription using the mRNA contained in the lysate as a template. Then, by using the synthesized cDNA as a template, quantitative PCR was performed by using TaqMan® Gene Expression Assays, and a probe (Hs00916691_m1: human LPA, Hs03928985_g1: RNA18S5, Applied Biosystems). Then, expression levels of LPA mRNA were identified by using a CFXOpus 384 Real-Time PCR System (Bio-Rad).
As a result, as shown in FIG. 14, and Table 17, an effect of suppressing LPA mRNA expression was confirmed after treatment of OLX706A-006-12, OLX706A-007-12, OLX706A-007-4, OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, and OLX706A-092-4 in primary human hepatocytes.
[Table 17]
4-2. Confirmation of LPA expression suppression effect by using animal models
LPA GalNAc-asiRNAs with the above chemical modifications introduced were administered to HDI mice introduced with LPA, and resulting changes in LPA levels were measured. Specifically, plasmids including LPA and a secreted embryonic alkaline phosphatase (SEAP) were injected into mice (Balb/c, female, 6 weeks old) through hydrodynamic injection (HDI). The mice were subcutaneously injected with LPA GalNAc-asiRNAs at a single dose of 5 mg/kg (mpk). On days 7, 21, and 35 from the day of subcutaneous injection into the mice, SEAP levels in the serum were measured by using the Phospha-Light™ SEAP Reporter Gene Assay System (Invitrogen™, T1015), and the measured values were normalized with a level of SEAP in the serum before the administration of LPA GalNAc-asiRNAs, and with a level of SEAP in the serum of a control group to which saline was administered, in order to confirm LPA levels in the serum derived from the mice.
As a result, as shown in FIG. 15, and Table 18, an effect of suppressing expression of SEAP reporters was confirmed according to treatment of OLX706A-033-4, OLX706A-033-12, OLX706A-062-2, OLX706A-062-4, OLX706A-062-12, and OLX706A-092-4 in the mice.
[Table 18]
Example 5. LPA Knockdown Efficacy Studies in Apo(a) Transgenic Model
Apo(a)8K TG mice were generated from the frozen stock of the mouse described in the literature (Merki et al., 2011, JACC, doi:10.1016/j.jacc.2010.10.052). Blood samples were collected on Study Day -2 (pre-treatment bleeding) and serum apo(a) protein levels were determined by human lipoprotein A ELISA kit (Abcam, ab108878). Mice were grouped so that their initial apo(a) levels were even among experimental groups. Each cohort contained three animals (at least two female subjects). The GalNAc-conjugated LPA asiRNAs were administered subcutaneously at a single dose of 1 or 3 mpk on Study Day 0. Blood samples were collected weekly after dosing. Inhibition of LPA expression was determined by measuring serum apo(a) protein from treated mice. For normalization, values from pre-dose samples were averaged and set to 100%, and data were analyzed as relative values compared to the pre-dose.
As a result, as shown in FIG. 16a, and Table 19, OLX706A-062-12 showed 72% inhibition of LPA expression on day 7, 74% inhibition on day 14 and 72% inhibition on day 21. OLX706A-033-4 showed 74% inhibition of LPA expression on day 7, 61% inhibition on day 14 and 54% inhibition on day 21. OLX706A-092-4 showed 76% inhibition of LPA expression on day 7, 77% inhibition on day 14 and 76% inhibition on day 21.
As shown in FIG. 16b, and Table 20, OLX706A-062-12 showed 62% inhibition of LPA expression on day 7, 58% inhibition on day 14 and 57% inhibition on day 21. OLX706A-033-4 showed 41% inhibition of LPA expression on day 7, 28% inhibition on day 14 and 24% inhibition on day 21.
[Table 19]
[Table 20]
Example 6. Non-Human Primate Studies of GalNAc-asiLPAs to assess Lp (a) reduction in the serum
The cynomolgus monkeys (Macaca fascicularis) were grouped so that their mean body weights (about 2~4 kg) were comparable between the control and experimental groups. Each cohort contained two animals. The GalNAc-conjugated LPA asiRNAs were administered subcutaneously at a single dose of 3 mpk on Study Day 1. Blood samples were collected on Study Days -6 and 1, and weekly after dosing. At the indicated time points, inhibition of LPA expression was determined by measuring Lp (a) serum concentration from treated NHPs using the Lp (a) ELISA (Mercodia, 10-1106-01). Values from pre-dose samples were averaged and set to 100%, and data were analyzed as relative values compared to the pre-dose.
OLX706A-033-4 showed 89% inhibition of LPA expression on day 29, 80% inhibition on day 43 and 53% inhibition on day 64. OLX706A-062-12 showed 84% inhibition of LPA expression on day 29, 66% inhibition on day 43 and 43% inhibition on day 64. Data from the experiment are shown in the following Table 21 and FIG. 17.
[Table 21]
Thus far, specific portions of the content of the present disclosure have been described in detail, and it will be apparent to those of ordinary skill in the art that these specific descriptions are only preferred embodiments, and that the scope of the present disclosure is not limited thereby. Accordingly, it is intended that the substantial scope of the present disclosure is defined by the appended claims and their equivalents.
An RNAi agent according to an aspect may inhibit expression of the LPA protein by binding to and degrading LPA mRNA, while alleviating side effects such as non-specific immune responses and off-target effects. A study was also performed to evaluate the toxicity of RNAi agents of the present disclosure in ICR mice after a single-dose subcutaneous administration followed by 8-day observation periods, no significant sign of toxicity was found.
In addition, the RNAi agent according to an aspect is effectively delivered to hepatocytes and may be usefully as a target therapeutic agent for cardiovascular diseases.
It should be understood that embodiments described herein should be considered in a descriptive sense only and not for purposes of limitation. Descriptions of features or aspects within each embodiment should typically be considered as available for other similar features or aspects in other embodiments. While one or more embodiments have been described with reference to the figures, it will be understood by those of ordinary skill in the art that various changes in form and details may be made therein without departing from the spirit and scope of the disclosure as defined by the following claim.
Claims (19)
- An RNAi agent comprising: an antisense strand having sequence complementarity to an LPA (lipoprotein(a)) mRNA sequence, and is 19 nt to 21 nt in length; and a sense strand having sequence complementarity to the antisense strand, and is 15 nt to 17 nt in length, wherein the 5' end of the antisense strand and the 3' end of the sense strand form a blunt end.
- The RNAi agent of claim 1, wherein the sense strand has a sequence selected from the sense strand sequences listed in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, and Table 9.
- The RNAi agent of claim 1, wherein the sense strand is any one selected from: SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 31, SEQ ID NO: 35, SEQ ID NO: 39, SEQ ID NO: 47, SEQ ID NO: 51, SEQ ID NO: 53, SEQ ID NO: 63, SEQ ID NO: 65, SEQ ID NO: 67, SEQ ID NO: 71, SEQ ID NO: 77, SEQ ID NO: 79, SEQ ID NO: 83, SEQ ID NO: 85, SEQ ID NO: 87, SEQ ID NO: 103, SEQ ID NO: 123, SEQ ID NO: 125, SEQ ID NO: 127, SEQ ID NO: 183, SEQ ID NO: 215, SEQ ID NO: 339, SEQ ID NO: 341, SEQ ID NO: 373, SEQ ID NO: 375, SEQ ID NO: 471, SEQ ID NO: 477, SEQ ID NO: 495, SEQ ID NO: 701, SEQ ID NO: 709, SEQ ID NO: 711, SEQ ID NO: 713, SEQ ID NO: 715, SEQ ID NO: 735, SEQ ID NO: 737, SEQ ID NO: 739, SEQ ID NO: 743 and SEQ ID NO: 775.
- The RNAi agent of claim 1, wherein the sense strand is any one selected from: SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 63, SEQ ID NO: 65, SEQ ID NO: 67, SEQ ID NO: 71, SEQ ID NO: 85, SEQ ID NO: 123, SEQ ID NO: 127, SEQ ID NO: 183, SEQ ID NO: 215, SEQ ID NO: 339, SEQ ID NO: 341, SEQ ID NO: 373, and SEQ ID NO: 737; and preferably the sense strand is any one selected from SEQ ID NO: 11, SEQ ID NO: 13, SEQ ID NO: 65, SEQ ID NO: 123, and SEQ ID NO: 183.
- The RNAi agent of claim 1, wherein the antisense strand has a sequence selected from the antisense strand sequences listed in Table 1, Table 2, Table 3, Table 4, Table 5, Table 6, Table 7, Table 8, and Table 9.
- The RNAi agent of claim 1, wherein the antisense strand is any one selected from: SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 32, SEQ ID NO: 36, SEQ ID NO: 40, SEQ ID NO: 48, SEQ ID NO: 52, SEQ ID NO: 54, SEQ ID NO: 64, SEQ ID NO: 66, SEQ ID NO: 68, SEQ ID NO: 72, SEQ ID NO: 78, SEQ ID NO: 80, SEQ ID NO: 84, SEQ ID NO: 86, SEQ ID NO: 88, SEQ ID NO: 104, SEQ ID NO: 124, SEQ ID NO: 126, SEQ ID NO: 128, SEQ ID NO: 184, SEQ ID NO: 216, SEQ ID NO: 340, SEQ ID NO: 342, SEQ ID NO: 374, SEQ ID NO: 376, SEQ ID NO: 472, SEQ ID NO: 478, SEQ ID NO: 496, SEQ ID NO: 702, SEQ ID NO: 710, SEQ ID NO: 712, SEQ ID NO: 714, SEQ ID NO: 716, SEQ ID NO: 736, SEQ ID NO: 738, SEQ ID NO: 740, SEQ ID NO: 744 and SEQ ID NO: 776.
- The RNAi agent of claim 1, wherein the antisense strand is any one selected from: SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 64, SEQ ID NO: 66, SEQ ID NO: 68, SEQ ID NO: 72, SEQ ID NO: 86, SEQ ID NO: 124, SEQ ID NO: 128, SEQ ID NO: 184, SEQ ID NO: 216, SEQ ID NO: 340, SEQ ID NO: 342, SEQ ID NO: 374, and SEQ ID NO: 738; and preferably the antisense strand is any one selected from SEQ ID NO: 12, SEQ ID NO: 14, SEQ ID NO: 66, SEQ ID NO: 124, and SEQ ID NO: 184; more preferably the RNAi agent has a sense strand as shown in SEQ ID NO: 65, and has an antisense strand as shown in SEQ ID NO: 66; the RNAi agent has a sense strand as shown in SEQ ID NO: 123, and has an antisense strand as shown in SEQ ID NO: 124; or the RNAi agent has a sense strand as shown in SEQ ID NO: 183, and has an antisense strand as shown in SEQ ID NO:184.
- The RNAi agent of claim 1, wherein the sense strand is bound to an N-acetylgalactosamine (GalNAc) derivative at the 3' end.
- The RNAi agent of claim 1, wherein the sense strand or the antisense strand comprises at least one chemical modification.
- The RNAi agent of claim 9, wherein the sense strand comprises at least one chemical modification selected from:modification of 2 to 4 nucleotide bonds adjacent to the 5' end with phosphorothioate, boranophosphate, or methyl phosphonate;substitution of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide with -CH3 (methyl), -OCH3 (-O-methyl), -NH2, -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; andbinding with an N-acetylgalactosamine (GalNAc) derivative, or a cell penetrating peptide at the 3' end.
- The RNAi agent of claim 9, wherein the antisense strand comprises at least one chemical modification selected from:modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end with phosphorothioate, boranophosphate, or methyl phosphonate; andsubstitution of an -OH group at the 2' carbon position of the sugar structure of at least one nucleotide with -H, -CH3 (methyl), -OCH3 (-O-methyl), -NH2, -F, -O-2-methoxyethyl-O-propyl, -O-2-methyl methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl; and substitution of an -OH group at the 5' end with E-vinylphosphonate.
- The RNAi agent of claim 9, comprising at least one modification selected from:modification of 2 to 7 nucleotide bonds adjacent to the 3' end or the 5' end in a sense or an antisense strand to phosphorothioate;modification of an-OH group at the 2' carbon position of the sugar structure of at least one nucleotide in a sense strand or an antisense strand to -H, -OCH3 (-O-methyl), or -F;binding to an N-acetylgalactosamine (GalNAc) derivative at the 3' end of the sense strand; andsubstitution of an -OH group at the 5' end of the antisense strand with E-vinylphosphonate.
- The RNAi agent of any one of claims 1-12, wherein in the antisense strand, from 5' to 3', positions 2, 14, 16 are modified by 2'-fluoro, positions 5 and 7 are deoxynucleosides, and the rest positions are modified by 2'-OMe, at the 3' end, there are six phosphorothioate linkages, at the 5' end, there is a 5' (E)-vinylphosphonate nucleotide; in the sense strand, from 5' to 3', positions 2, 4, 5, 6 are modified by 2'-fluoro, and optionally at the 3' end, there is a nucleotide linked with an N-acetylgalactosamine (GalNAc) derivative.
- The RNAi agent of claim 13, wherein in the sense strand, from 5' to 3': positions 2, 4, 5, 6, 14 and 16 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe; or positions 2, 4, 5, 6 and 12 are modified by 2'-fluoro, and the rest positions are modified by 2'-OMe.
- The RNAi agent of claim 9, wherein the sense strand has a sequence selected from sense strand sequences listed in Table 15.
- The RNAi agent of claim 9, wherein the antisense strand has a sequence selected from antisense strand sequences listed in Table 15, preferably the RNAi agent is selected from the GalNAc-asiRNA conjugates listed in Table 15.
- A pharmaceutical composition comprising the RNAi agent according to any one of claims 1 to 16 as an active ingredient and pharmaceutically acceptable carriers.
- A use of the RNAi agent according to any one of claims 1 to 16 or the pharmaceutical composition of claim 17 for manufacturing a pharmaceutical product for preventing or treating cardiovascular disease.
- The use of claim 18, wherein the cardiovascular disease is selected from: Berger's disease, peripheral artery disease, coronary artery disease, metabolic syndrome, acute coronary syndrome, aortic valve stenosis, myocardial infarction, aortic valve regurgitation, aortic dissection, retinal artery occlusion, cerebrovascular disease, mesenteric ischemia, superior mesenteric artery occlusion, renal artery stenosis, stable/unstable angina, acute coronary syndrome, heterozygous or homozygous familial hypercholesterolemia, hyperaphobeta lipoproteinemia, cerebrovascular atherosclerosis, and venous thrombosis.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202480002533.5A CN119384503A (en) | 2023-04-03 | 2024-04-03 | RNAi agent targeting LPA gene and use thereof |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR20230043712 | 2023-04-03 | ||
| KR10-2023-0043712 | 2023-04-03 | ||
| CN2023140973 | 2023-12-22 | ||
| CNPCT/CN2023/140973 | 2023-12-22 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024210530A1 true WO2024210530A1 (en) | 2024-10-10 |
Family
ID=92972226
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2024/004361 Ceased WO2024210530A1 (en) | 2023-04-03 | 2024-04-03 | RNAi AGENTS TARGETING LPA GENE AND USE THEREOF |
Country Status (2)
| Country | Link |
|---|---|
| CN (2) | CN118773191A (en) |
| WO (1) | WO2024210530A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN120344664A (en) * | 2023-10-27 | 2025-07-18 | 北京安龙生物医药有限公司 | Oligonucleotides targeting lipoprotein a and uses thereof |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025252159A1 (en) * | 2024-06-06 | 2025-12-11 | 大睿生物医药科技(上海)有限公司 | Dsrna molecule for regulating lpa expression |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108368506A (en) * | 2015-10-01 | 2018-08-03 | 箭头药业股份有限公司 | Compositions and methods for inhibiting gene expression of LPA |
| WO2022079221A1 (en) * | 2020-10-16 | 2022-04-21 | Sanofi | Rna compositions and methods for inhibiting lipoprotein(a) |
| US20230078200A1 (en) * | 2019-12-09 | 2023-03-16 | Amgen Inc. | RNAi CONSTRUCTS AND METHODS FOR INHIBITING LPA EXPRESSION |
| WO2023041079A1 (en) * | 2021-09-18 | 2023-03-23 | 上海金中锘美生物医药科技有限公司 | Lpa inhibitor and use thereof |
| CN115851723A (en) * | 2022-10-24 | 2023-03-28 | 厦门甘宝利生物医药有限公司 | A kind of RNA inhibitor that suppresses LPA gene expression and application thereof |
-
2024
- 2024-04-03 CN CN202410405283.1A patent/CN118773191A/en active Pending
- 2024-04-03 CN CN202480002533.5A patent/CN119384503A/en active Pending
- 2024-04-03 WO PCT/KR2024/004361 patent/WO2024210530A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN108368506A (en) * | 2015-10-01 | 2018-08-03 | 箭头药业股份有限公司 | Compositions and methods for inhibiting gene expression of LPA |
| US20230078200A1 (en) * | 2019-12-09 | 2023-03-16 | Amgen Inc. | RNAi CONSTRUCTS AND METHODS FOR INHIBITING LPA EXPRESSION |
| WO2022079221A1 (en) * | 2020-10-16 | 2022-04-21 | Sanofi | Rna compositions and methods for inhibiting lipoprotein(a) |
| WO2023041079A1 (en) * | 2021-09-18 | 2023-03-23 | 上海金中锘美生物医药科技有限公司 | Lpa inhibitor and use thereof |
| CN115851723A (en) * | 2022-10-24 | 2023-03-28 | 厦门甘宝利生物医药有限公司 | A kind of RNA inhibitor that suppresses LPA gene expression and application thereof |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN120344664A (en) * | 2023-10-27 | 2025-07-18 | 北京安龙生物医药有限公司 | Oligonucleotides targeting lipoprotein a and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN119384503A (en) | 2025-01-28 |
| CN118773191A (en) | 2024-10-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2024210530A1 (en) | RNAi AGENTS TARGETING LPA GENE AND USE THEREOF | |
| US11534452B2 (en) | Nucleic acid molecules for reduction of PAPD5 or PAPD7 mRNA for treating hepatitis B infection | |
| WO2013176477A1 (en) | Rna-interference-inducing nucleic acid molecule able to penetrate into cells, and use therefor | |
| JP2018068243A (en) | Method for canceling oocyte maturation inhibition of useful shrimps | |
| WO2018155890A1 (en) | Asymmetric sirna for inhibiting expression of male pattern hair loss target gene | |
| US20230287427A1 (en) | Inhibition of lncExACT1 to Treat Heart Disease | |
| WO2009151304A2 (en) | Composition comprised of akap12 and uses of akap12 mutant zebrafish as an animal model | |
| WO2020235936A1 (en) | Asymmetric sirna inhibiting expression of pd-1 | |
| WO2023282704A1 (en) | Rnai agent targeting marc1 gene, and use thereof | |
| WO2019066519A1 (en) | Pharmaceutical composition for prevention or treatment of age-related macular degeneration containing rna complex targeting connective tissue growth factor | |
| WO2020213982A1 (en) | Composition for prevention or treatment of neurodegenerative diseases using cd9 and method for screening therapeutic agent for neurodegenerative diseases | |
| WO2022131883A1 (en) | Rnai agent for inhibiting hbv expression and use thereof | |
| WO2023163497A1 (en) | Pharmaceutical composition for preventing or treating colorectal cancer comprising ednra inhibitor as active ingredient | |
| US20210310000A1 (en) | Compounds of chemically modified oligonucleotides and methods of use thereof | |
| WO2010095879A2 (en) | Sirna to be bound to transcripts of apoptosis-related genes, and compositions for treating cancer using same | |
| WO2015093886A1 (en) | Sirna targeting prk2, which is hepatitis c virus therapeutic agent | |
| WO2011087343A2 (en) | Composition for treating cancer related to an hpv infection | |
| WO2017061828A1 (en) | Pharmaceutical composition for preventing or treating cancer comprising inhibitor of plrg1 (pleiotropic regulator 1) as active ingredient | |
| WO2022145949A2 (en) | Composition comprising tead4 inhibitor for preventing or treating colorectal cancer | |
| WO2015065020A1 (en) | Pharmaceutical composition for preventing or treating hepatitis c virus infectious disease | |
| WO2020122534A1 (en) | Asymmetric sirna for inhibiting expression of snai1 | |
| WO2024186166A1 (en) | Double-stranded rna having innate immune response-inducing effect, and use thereof | |
| WO2015147506A1 (en) | Novel biomarker piro for osteoclast fusion | |
| Ge et al. | Bovine CAPN3 core promoter initiates expression of foreign genes in skeletal muscle cells by MyoD transcriptional regulation | |
| WO2025053611A1 (en) | Pharmaceutical composition comprising pdk3 inhibitor for preventing or treating peripheral nerve tumor, and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| WWE | Wipo information: entry into national phase |
Ref document number: 202480002533.5 Country of ref document: CN |
|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24785231 Country of ref document: EP Kind code of ref document: A1 |
|
| WWP | Wipo information: published in national office |
Ref document number: 202480002533.5 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |