WO2024186166A1 - Double-stranded rna having innate immune response-inducing effect, and use thereof - Google Patents
Double-stranded rna having innate immune response-inducing effect, and use thereof Download PDFInfo
- Publication number
- WO2024186166A1 WO2024186166A1 PCT/KR2024/003026 KR2024003026W WO2024186166A1 WO 2024186166 A1 WO2024186166 A1 WO 2024186166A1 KR 2024003026 W KR2024003026 W KR 2024003026W WO 2024186166 A1 WO2024186166 A1 WO 2024186166A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- double
- stranded rna
- innate immune
- immune response
- strand
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/39—Medicinal preparations containing antigens or antibodies characterised by the immunostimulating additives, e.g. chemical adjuvants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
Definitions
- the present invention relates to a double-stranded RNA having an innate immune response inducing effect and a use thereof, and more particularly, to a double-stranded RNA comprising a nucleotide into which an innate immune activation motif and a chemical modification have been introduced.
- the innate immune response is a non-specific immune response of the body, and it acts as the first line of defense of the body to recognize external factors and respond first. Since it has been known that pathological conditions such as cancer, pathogenic infection, and hereditary diseases that are difficult to treat can be improved by enhancing or activating the innate immune response, research on therapeutics using the innate immune response has been actively conducted. To this end, research has been conducted on the mechanism of action that can activate or regulate the innate immune response.
- Pattern recognition receptors are proteins present in the cell membrane or cytoplasm of cells, which recognize pattern recognition molecules and mediate innate immune responses. Pattern recognition receptors are distributed in dendritic cells, macrophages, monocytes, neutrophils, epithelial cells, etc., and recognize pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs).
- PAMPs pathogen-associated molecular patterns
- DAMPs damage-associated molecular patterns
- TLR Toll-like receptors
- CLR C-type lectin receptors
- RLR retinoic acid-inducible gene-I-like receptors
- NLR nucleotide oligomerization domain-like receptors
- ARR absence-in-melanoma-like receptors
- RNA interference is a method of suppressing target gene expression by inducing cleavage of target mRNA.
- short interfering RNA small interfering RNA
- siRNA an external substance
- the introduction of modified RNA nucleotides for example, the introduction of nucleotides with chemical modifications, has been suggested as a technology for reducing the innate immune response by administration of siRNA.
- RNA-based therapeutics can be applied not only to the aforementioned RNAi agents, but also to passive and active immunotherapy, vaccine agents, or genetic engineering in general.
- RNA molecules can also be used as therapeutics for replacement therapy, such as protein replacement therapy to replace missing or mutated proteins.
- the inventors of the present invention have conducted research efforts to develop a double-stranded RNA capable of specifically activating an innate immune response to enhance therapeutic efficacy, and as a result, have confirmed the effect of enhancing an innate immune response resulting from chemical modification of specific motifs and nucleotides, and have completed the present invention based on this.
- the purpose of the present invention is to provide double-stranded RNA capable of specifically activating an innate immune response.
- Another object of the present invention is to provide a composition for enhancing an innate immune response comprising the double-stranded RNA, a pharmaceutical composition comprising the double-stranded RNA, or a method for treating a disease or improving a pathological condition comprising a step of administering the pharmaceutical composition to a subject.
- One aspect is a double-stranded RNA having 25 to 60 base pairs (bp) comprising a first strand; and a second strand complementarily binding to the first strand, wherein one strand of the double-stranded RNA contains at least 10 nucleotides to which a chemical modification has been introduced, and wherein the double-stranded RNA enhances an MDA5 (Melanoma differentiation-associated protein 5)-mediated innate immune response.
- MDA5 Melnoma differentiation-associated protein 5
- Another aspect provides a double-stranded RNA of 25 to 60 base pairs, comprising a first strand; and a second strand complementarily binding to said first strand, said double-stranded RNA comprising an innate immune activation motif of 10 to 30 base pairs, wherein one strand of said double-stranded RNA comprises at least 10 nucleotides to which a chemical modification has been introduced.
- composition for enhancing innate immunity comprising the double-stranded RNA.
- Another aspect provides a pharmaceutical composition comprising the double-stranded RNA as an active ingredient.
- Another aspect provides a method of treating a disease comprising administering to a subject the double-stranded RNA.
- Another aspect provides a medicinal use of said double-stranded RNA for the treatment or amelioration of a disease or pathological condition.
- Double-stranded RNA can induce a high level of innate immune response by including an innate immune response Activating Motif and a chemically modified nucleotide.
- double-stranded RNA may enhance the efficacy of vaccine formulations or immunotherapeutic agents by enhancing innate immune responses.
- Figure 1 is a schematic diagram showing the structure of a double-stranded RNA containing an innate immune activation motif according to one aspect.
- Figure 2 shows the results of confirming the mRNA expression levels of IFIT1, CXCL10, and ISG15 after transfecting a T98G cell line with a double-stranded RNA containing an innate immune activation sequence of sequence number 6 according to one aspect.
- Figure 3 shows the results of confirming the mRNA expression levels of TNF- ⁇ and IL-1 ⁇ after transfecting a T98G cell line with a double-stranded RNA containing an innate immune activation sequence of sequence number 6 according to one aspect.
- Figure 4 shows the results of confirming the mRNA expression levels of IFIT1, CXCL10, and ISG15 after transfecting a T98G cell line with a double-stranded RNA containing an innate immune activation sequence of sequence number 8 according to one aspect.
- Figure 5 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting a double-stranded RNA containing an innate immune activation sequence of sequence number 6 according to one aspect into a wild-type cell line (WT) or a cell line lacking RIG-I (RIG-I KO).
- IRF interferon regulatory factor
- Figure 6 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting a double-stranded RNA containing an innate immune activation sequence of sequence number 8 according to one aspect into a wild-type cell line (WT) or a cell line lacking RIG-I (RIG-I KO).
- IRF interferon regulatory factor
- Figure 7 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting a double-stranded RNA containing an innate immune activation sequence of sequence number 2 or sequence number 4 according to one aspect into a wild-type cell line (WT) or a cell line lacking RIG-I (RIG-I KO).
- IRF interferon regulatory factor
- Figure 8 is according to the daily aspect. This is the result of confirming the mRNA expression level of the Lamin A/C gene after transfecting the Huh7 cell line with a 30-bp double-stranded RNA containing the innate immune activation sequence of sequence number 2.
- Figure 9 is according to the daily aspect. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting each of the double-stranded RNAs containing the innate immune activation sequence of sequence number 2, wherein the innate immune activation sequence within the double-stranded RNA is located at the 5' end of the second strand or the 3' end of the first strand, into a wild-type cell line (WT) and a cell line lacking RIG-I (RIG-KO).
- IRF interferon regulatory factor
- Figure 10 is a diagram showing an embodiment in which a chemical modification is introduced into either the first strand or the second strand. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
- IRF interferon regulatory factor
- Figure 11 is a diagram showing an embodiment of a method in which chemical modifications are introduced into both the first and second strands. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
- IRF interferon regulatory factor
- Figure 12 shows an aspect in which 10 nucleotides into which chemical modifications are introduced are positioned at the 5' end or the 3' end of the first strand (#1 5'-1-10, #1 3'-1-10), or the position of the first nucleotide into which chemical modifications are introduced in the first strand is changed (#1 5'-1-10, #1 5'-2-10). This is the result of confirming the activity level of interferon regulatory factors after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
- WT wild-type cell line
- RIG-I deficient cell line RIG-I deficient cell line
- Figure 13 shows chemical modifications introduced into 8, 10, 12 or 15 nucleotides, respectively. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
- IRF interferon regulatory factor
- Figure 14 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting chemically modified double-stranded RNA into a wild-type cell line (WT), a cell line lacking RIG-I (RIG-I KO), or a cell line lacking MDA5 (MDA5 KO), respectively.
- IRF interferon regulatory factor
- Figure 15 shows the results of examining the activity level of interferon regulatory factor (IRF) over time after transfecting a human monocyte cell line (THP-1 Dual) with double-stranded RNA with chemical modifications.
- IRF interferon regulatory factor
- Figure 16 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell lines with double-stranded RNAs having different nucleotide lengths of 20 to 40 bp, in which the innate immune activation motif is located at the 5' end of the second strand and at least 10 chemical modifications are introduced into the first strand.
- IRF interferon regulatory factor
- Figure 17 shows the results of examining the activity level of interferon regulatory factor (IRF) over time after transfecting a human monocyte cell line (THP-1 Dual) with double-stranded RNA containing or not containing an innate immune activation motif in a 30bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
- IRF interferon regulatory factor
- FIG. 18 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell line with a double-stranded RNA including an innate immune activation sequence of SEQ ID NO: 2, an innate immune activation sequence of SEQ ID NO: 8 (20 nt), or an innate immune activation sequence of SEQ ID NO: 62 (30 nt) in a 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
- IRF interferon regulatory factor
- FIG. 19 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell lines with double-stranded RNA containing chemical modification of 2' -OCH3(-O-methyl) or chemical modification of 2' -F in 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
- IRF interferon regulatory factor
- FIG. 20 shows the results of examining the activity level of interferon regulatory factor (IRF) over time after transfecting a human monocyte cell line (THP-1 Dual) with a double-stranded RNA containing a chemical modification of 2' -OCH3(-O-methyl) or a chemical modification of 2' -F in a 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
- IRF interferon regulatory factor
- FIG. 21 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell line with double-stranded RNA in which two adjacent nucleotide bonds at both ends of the first strand, the second strand, or both the first strand and the second strand are modified with phosphorothioate bonds in a 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
- IRF interferon regulatory factor
- One aspect provides a double-stranded RNA of 25 to 60 base pairs, comprising a first strand; and a second strand complementarily binding to the first strand, wherein the double-stranded RNA comprises an innate immune activation motif of 10 to 30 base pairs, wherein one strand of the double-stranded RNA comprises at least 10 nucleotides to which a chemical modification has been introduced.
- double-stranded RNA means RNA having two strands (a first strand and a second strand) that complementarily bind to each other.
- the double-stranded RNA may be used interchangeably with "RNA-based immune enhancer/adjuvant”, “RNA-based immunogenic composition”, and “RNAi-inducing nucleic acid molecule”, depending on the intended use.
- first strand or “second strand” may be referred to as the antisense strand or the sense strand, respectively, when the double-stranded RNA comprises a region for RNA interference.
- the double-stranded RNA may have a length of 25 to 60 base pairs, for example, 25 to 60, 25 to 55, 25 to 50, 25 to 45, 25 to 40, 25 to 35, 25 to 30, 26 to 60, 26 to 55, 26 to 50, 26 to 45, 26 to 40, 26 to 35, 26 to 30, 30 to 60, 30 to 55, 30 to 50, 30 to 45, 30 to 40, 30 to 35, 35 to 60, 35 to It may have a length of 55, 35 to 50, 35 to 45, or 35 to 40 base pairs.
- complementarity refers to their generally accepted meanings in the art.
- the terms may generally refer to the formation or presence of hydrogen bond(s) between one nucleic acid sequence and another nucleic acid sequence, either by traditional Watson-Crick or other non-traditional types of binding as described herein. Perfect complementarity may mean that every adjacent residue in one nucleic acid sequence hydrogen bonds with the same number of adjacent residues in the other nucleic acid sequence.
- Partial complementarity can include various mismatches or non-based paired nucleotides within a nucleic acid molecule (e.g., more than 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 mismatches, e.g., 1 to 3 mismatches, non-nucleotide linkers, or non-base paired nucleotides).
- the partial complementarity can result in bulges, loops, overhangs, or blunt ends between the first and second strands of a double-stranded RNA, or between the first strand of a double-stranded RNA and a corresponding target nucleic acid molecule.
- Innate Immune Response Activating Motif refers to a region in a double-stranded RNA that induces or enhances an innate immune response in a subject to which the double-stranded RNA has been administered, and refers to a double-stranded structure composed of an innate immune activating sequence and a sequence that forms a complementary bond with the sequence.
- innate immune response may refer to an in vivo innate defense immune system induced from double-stranded RNA, which may contribute to improving the efficacy of treatment by enhancing the immunity of an individual from a therapeutic perspective.
- the innate immune response may be, for example, induced by interferon, or mediated by RIG-I (Retinoic acid-inducible gene I), or MDA5 (Melanoma differentiation-associated gene 5).
- the innate immune response by double-stranded RNA depends on the terminal region structure of the double-strand. Therefore, as a technical means for enhancing the innate immune response, an approach of introducing 5'-triphosphate or introducing a modified terminal structure has been reported. In addition, it has been reported that the MDA5-mediated immune response is induced by long-length double-stranded RNA. Meanwhile, the present inventors have elucidated that the innate immune response can be induced or enhanced through the innate immune activation motif and any chemical modification even though the double-stranded RNA according to one embodiment has a relatively short length.
- the term "innate immune activation sequence” refers to a nucleotide sequence located on either strand of a double-stranded RNA that activates an innate immune response.
- the innate immune activation sequence may comprise a structure capable of activating an innate immune response mediated by RIG-I.
- the innate immune activation sequence may be a nucleotide sequence of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62.
- the innate immune activation sequence may comprise a variant of the nucleotide sequence having at least 80% sequence identity with any one of the nucleotide sequences of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62.
- the innate immune activation motif can be comprised of a nucleotide sequence of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62, and a nucleotide sequence complementarily binding to said nucleotide sequence.
- the innate immune activation motif can be located at one terminal region of the double-stranded RNA, for example, can be located at the 3' end of the first strand or the 5' end of the second strand.
- the innate immune activation motif comprises 10 to 30 nucleotides comprising the nucleotide sequence of SEQ ID NO: 2, for example, 10 to 30, 10 to 28, 10 to 26, 10 to 24, 10 to 22, 10 to 20, 10 to 18, 10 to 16, 10 to 14, 10 to 12, 12 to 30, 12 to 28, 12 to 26, 12 to 24, 12 to 22, 12 to 20, 12 to 18, 12 to 16, 12 to 14, 14 to 30, 14 to It may consist of 28, 14 to 26, 14 to 24, 14 to 22, 14 to 20, 14 to 18, or 14 to 16 nucleotides and another nucleotide sequence complementarily binding to said sequence.
- the innate immune activation motif may have a length of 10 to 30 base pairs, for example, 10 to 30, 10 to 28, 10 to 26, 10 to 24, 10 to 22, 10 to 20, 10 to 18, 10 to 16, 10 to 14, 10 to 12, 12 to 30, 12 to 28, 12 to 26, 12 to 24, 12 to 22, 12 to 20, 12 to 18, 12 to 16, 12 to 14, 14 to 30, 14 to 28, It may have a length of 14 to 26, 14 to 24, 14 to 22, 14 to 20, 14 to 18, 14 to 16, 16 to 30, 16 to 28, 16 to 26, 16 to 24, 16 to 22, 16 to 20, 16 to 18, 18 to 30, or 18 to 20 base pairs.
- variants refer to entities that exhibit substantial structural identity with a reference entity (e.g., a wild type sequence) but are structurally different from the reference entity in one or more respects.
- a polynucleotide may differ from a reference polynucleotide by one or more differences in the nucleotide sequence and/or by one or more differences in the chemical moieties (e.g., carbohydrates, lipids, etc.) covalently linked to the polynucleotide backbone.
- the variants exhibit at least 60%, 65%, 70%, 75%, 80%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% overall sequence identity with the reference polynucleotide.
- identity refers to the overall relatedness between polymer molecules, for example, between nucleic acids (e.g., DNA molecules and/or RNA molecules).
- polynucleotide sequences are considered to be “substantially identical” to one another if their sequences are at least 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 99% identical.
- Calculating the percent identity of two polynucleotide sequences can be performed, for example, by aligning the two sequences for optimal comparison purposes (e.g., gaps can be introduced into one or both of the first and second sequences for optimal alignment, and non-identical sequences can be ignored for comparison purposes).
- Determining the percent identity between two sequences and comparing the sequences can be accomplished using mathematical algorithms. As is well known to those skilled in the art, nucleotide sequences may be compared using any of a variety of algorithms, including those available in commercial computer programs such as BLASTN.
- the innate immune activation motif is introduced into one terminal region of the double-stranded RNA, so that it can operate independently of other regions other than the innate immune activation motif.
- Double-stranded RNA with chemical modifications Double-stranded RNA with chemical modifications
- either the first strand or the second strand may include a nucleotide into which a chemical modification has been introduced.
- the nucleotide into which the chemical modification is introduced may comprise one or more chemical modifications selected from the group consisting of: replacement of the -OH group at the 2' carbon position of the sugar structure in the nucleotide with -H, -CH 3 (methyl), -OCH 3 (-O-methyl), -NH 2 , -F, -O-2-methoxyethyl-O-propyl, -O-2-methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl.
- the nucleotides to which the chemical modification has been introduced may be included in either the first strand or the second strand, and the number of nucleotides to which the chemical modification has been introduced may be at least 10 or more.
- the number of nucleotides into which the chemical modification is introduced is, for example, 10 to 60, 10 to 55, 10 to 50, 10 to 45, 10 to 40, 10 to 35, 10 to 30, 10 to 25, 10 to 20, 10 to 15, 15 to 60, 15 to 55, 15 to 50, 15 to 45, 15 to 40, 15 to 35, 15 to 30, 15 to 25, 15 to 20, 20 to 60, 20 to 55, 20 to 50, 20 to It can be 45, 20 to 40, 20 to 35, 20 to 30, 20 to 25, or 15 to 20.
- the nucleotides into which the chemical modification has been introduced may be arranged either continuously or discontinuously within either the first strand or the second strand.
- the nucleotide into which the chemical modification is introduced may be included in either the first strand or the second strand and may be located in a region within the innate immune activation motif and/or a region outside the innate immune activation motif.
- the nucleotides to which the chemical modification has been introduced are included in at least 10 nucleotides in the first strand, and may be arranged alternatively with nucleotides to which the chemical modification has not been introduced.
- nucleotides to which the chemical modification has been introduced in which the -OH group at the 2' carbon position is substituted with -OCH 3 (-O-methyl) and nucleotides to which the chemical modification has not been introduced may be arranged alternatively.
- 50% or less, for example, 50%, 45%, 40%, 35%, or 30% of the total nucleotides in the first strand may be nucleotides to which the chemical modification has been introduced.
- the nucleotides to which the chemical modification has been introduced are included in the second strand at least 10 times, and may be arranged alternately with nucleotides to which the chemical modification has not been introduced, for example, nucleotides to which the chemical modification has been introduced in which the -OH group at the 2' carbon position is substituted with -OCH 3 (-O-methyl) and nucleotides to which the chemical modification has not been introduced may be arranged alternately.
- 50% or less, for example, 50%, 45%, 40%, 35%, or 30% of the total nucleotides of the second strand may be nucleotides to which the chemical modification has been introduced.
- the nucleotide to which the chemical modification has been introduced can induce or enhance an innate immune response.
- the nucleotide to which the chemical modification has been introduced can induce or enhance an MDA5-mediated innate immune response.
- the RNA duplex according to the present invention comprising the nucleotide to which the chemical modification has been introduced can be for inducing or enhancing an RIG-I-mediated innate immune response and/or an MDA5-mediated innate immune response.
- MDA5 Melnoma differentiation-associated gene 5
- RIG-I RIG-I-like receptor
- CARDs N-tandem amino-terminal caspase activation and recruitment domains
- CCD C-terminal domain
- IRFs interferon regulatory factors
- a double-stranded RNA having the structure of the aforementioned double-stranded RNA as its basic backbone and additionally having chemical modifications can induce a strong innate immune response despite the processing of double-stranded RNA having a relatively short base length, through an additional signaling mechanism in which MDA5 acts as a key factor.
- the double-stranded RNA may be one in which at least one inter-nucleotide bond is modified with phosphorothioate, boranophosphate, or methyl phosphonate.
- the double-stranded RNA may be one in which at least one, for example, 2 to 4, inter-nucleotide bonds adjacent to the 5'-end and/or the 3'-end of the first strand and/or the second strand are modified with phosphorothioate, boranophosphate, or methyl phosphonate.
- a double-stranded RNA having the structure of a double-stranded RNA with the chemical modifications described above as its basic backbone and further modified in the bonds between nucleotides can enable the induction of a strong innate immune response despite such modifications.
- the double-stranded RNA may be one that induces or enhances an innate immune response.
- the double-stranded RNA may satisfy the following conditions: 1) having a length of 25 to 60 base pairs, 2) the innate immune activation motif is located at one terminal region of one strand of the double-stranded RNA, 3) either the first strand or the second strand includes at least 10 nucleotides to which a chemical modification has been introduced.
- the double-stranded RNA may additionally have one or both ends of the double-stranded RNA be blunt ends. In one specific embodiment, one end of the double-stranded RNA may be an overhang. In one specific embodiment, the double-stranded RNA may have an innate immune activation motif positioned at the 3' end of the first strand and the 5' end of the second strand complementary thereto, forming a blunt end. For example, in the double-stranded RNA, one end at which the innate immune activation motif is positioned may be a blunt end, and the other end where the innate immune activation motif is not positioned may have an overhang. For example, in the double-stranded RNA, both one end at which the innate immune activation motif is positioned and the other end where the innate immune activation motif is not positioned may be blunt ends.
- the double-stranded RNA may be a double-stranded RNA having a length of 25 to 60 base pairs, and may have a property of inducing an innate immune response mediated by RIG-I and MDA-5, and for this purpose, the double-stranded RNA may include an innate immune activation motif, and either the first strand or the second strand may have a structure including at least 10 nucleotides to which a chemical modification has been introduced.
- the double-stranded RNA may be a double-stranded RNA having a length of 25 to 60 base pairs, and may have a property of inducing RNA interference that inhibits the expression of a specific gene, and for this purpose, the double-stranded RNA may additionally include a region that complementarily binds to a specific gene in a terminal region.
- the double-stranded RNA may have an innate immune activation sequence located at the 3' end of the first strand and the 5' end region of the second strand complementary thereto.
- the innate immune activation sequence may be 10 to 30 nucleotides including the nucleotide sequence of SEQ ID NO: 2, for example, the innate immune activation sequence may refer to a nucleotide consisting of any one of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62, or a variant of the nucleotide.
- the double-stranded RNA may contribute to enhancing an innate immune response of an individual, specifically, a RIG-I-mediated innate immune response and/or an MDA5-mediated innate immune response, by including the above-mentioned innate immune activation motif and the structure of the double-stranded RNA (e.g., chemical modification, terminal form).
- the double-stranded RNA comprises a RIG-I-mediated innate immune activation motif located at one end and at least 10 nucleotides into which a chemical modification has been introduced, and the double-stranded RNA having the structure described above can induce a high level of innate immune response, regardless of the sequence of a region outside the innate immune activation motif, the type of strand on which the innate immune activation motif is located, and the region located within the double strand.
- the term "effective ingredient” means an appropriate effective amount of an ingredient that affects a beneficial or desirable clinical or biochemical result. Specifically, it may mean an effective amount of a preparation, an active agent, or a double-stranded RNA.
- the pharmaceutical composition may optionally include a pharmaceutically acceptable carrier, diluent, excipient, buffer, salt, surfactant, cryoprotectant, etc.
- the effective amount may be administered once or more and may be an amount appropriate for preventing a disease, or for alleviating symptoms, reducing the extent of a disease, stabilizing (i.e. not worsening) a disease, delaying or reducing the rate of disease progression, or improving or temporarily alleviating and alleviating (partially or completely) a disease.
- prevention refers to any action that prevents the occurrence of a disease in advance, suppresses a disease, or delays its progression. For example, it refers to preventing the occurrence of the disease or its characteristic characteristics, interfering with the occurrence, or protecting or protecting against the occurrence of the disease or its characteristic characteristics.
- treatment refers to both therapeutic treatment and preventive or prophylactic measures. It also refers to any action that improves or beneficially alters the symptoms of a disease. For example, preventing, reducing or ameliorating the disease or its characteristic features, or delaying (attenuating) the progression of the disease or its characteristic features in a subject.
- the disease or pathological condition may be an infection caused by a virus or bacteria, a disease associated with immunosuppression, cancer (e.g., a solid tumor such as breast cancer, liver cancer, esophageal cancer, pancreatic cancer, lung cancer, stomach cancer, squamous cell carcinoma of the head and neck, prostate cancer, colon cancer, lymphoma, gallbladder cancer, kidney cancer, multiple myeloma, ovarian cancer, cervical cancer or glioma, or a non-solid tumor such as leukemia).
- cancer e.g., a solid tumor such as breast cancer, liver cancer, esophageal cancer, pancreatic cancer, lung cancer, stomach cancer, squamous cell carcinoma of the head and neck, prostate cancer, colon cancer, lymphoma, gallbladder cancer, kidney cancer, multiple myeloma, ovarian cancer, cervical cancer or glioma, or a non-solid tumor such as leukemia).
- the term "effective amount” refers to its generally accepted meaning in the art.
- the term can generally mean an amount of a molecule, compound, or composition that will elicit a desired biological response (e.g., a beneficial response) in a cell, tissue, system, animal, or human, as sought by a researcher, veterinarian, physician, or other clinician.
- a "therapeutically effective amount” can mean an amount of a molecule, compound, or composition that will elicit a desired medical response, such as a therapeutically relevant change in a measurable parameter associated with a disease or disorder, such that a particular clinical treatment can be considered efficacious.
- a therapeutically effective amount of a drug for treating the disease or disorder can be the amount necessary to effect a therapeutically relevant change in the parameter.
- pharmaceutically acceptable excipient may be one which, when combined/mixed with double-stranded RNA, maintains the activity of the double-stranded RNA.
- examples thereof include, but are not limited to, any standard pharmaceutical excipient, such as a buffer system such as phosphate buffered saline, a surfactant, water, emulsions such as oil/water emulsions and various forms of wetting agents, starches, milk, sugars, certain forms of clay, gelatin, stearic acid or its salts, magnesium or calcium stearate, talc, vegetable oils, gums, glycols, or other known excipients.
- a buffer system such as phosphate buffered saline
- surfactant water
- emulsions such as oil/water emulsions and various forms of wetting agents
- starches starches, milk, sugars, certain forms of clay, gelatin, stearic acid or its salts, magnesium or calcium stearate, talc
- the method of administration of the above pharmaceutical composition can be determined by a person skilled in the art based on the symptoms of the patient and the severity of the disease.
- it can be formulated in various forms such as powder, tablet, capsule, liquid, injection, ointment, syrup, etc., and can be provided in unit-dose or multi-dose containers, such as sealed ampoules and bottles.
- the pharmaceutical composition of the present invention can be administered orally or parenterally.
- the route of administration of the composition according to the present invention is not limited thereto, but for example, oral, intravenous, intramuscular, intraarterial, intramedullary, intrathecal, intracardiac, transdermal, subcutaneous, intraperitoneal, enteral, sublingual, or topical administration is possible.
- the dosage of the composition according to the present invention varies depending on the patient's weight, age, sex, health condition, diet, administration time, method, excretion rate, or disease severity, and can be easily determined by a person skilled in the art.
- the composition of the present invention can be formulated into a suitable dosage form using a known technique for clinical administration.
- composition for enhancing innate immunity refers to a biological agent containing the above-mentioned double-stranded RNA and for inducing or enhancing the innate immune response of an administered subject.
- the composition for enhancing innate immunity can be used interchangeably with an immune enhancer/adjuvant, an RNA-based immunogenic composition, or an immunostimulation agent.
- the composition can optionally contain a pharmaceutically acceptable carrier, diluent, excipient, buffer, salt, surfactant, cryoprotectant, etc., and the implementation conditions and implementation embodiments are as described for the pharmaceutical composition.
- composition for enhancing innate immunity can be utilized as an effective ingredient in a vaccine adjuvant, an immune anticancer agent, or an antiviral agent (e.g., hepatitis B virus), depending on the intended use.
- an immune anticancer agent e.g., hepatitis B virus
- the composition for enhancing innate immunity may, for example, form a complex with one or more lipid components to form liposomes, lipid nanoparticles and/or lipoplexes.
- the lipid nanoparticles include an ionizable cationic lipid as one component, and may also include other components such as a helper lipid or a stabilizer that encapsulates mRNA or helps with delivery efficiency and stabilization.
- the lipid nanoparticles may apply components known in the art, and may include cationic lipids, helper lipids, and PEG-conjugated lipids.
- techniques known in the art may be utilized without limitation with respect to effective amounts, formulations, administration methods, combination preparations, etc.
- Another aspect provides a method of treating a disease or ameliorating a pathological condition comprising administering to a subject the double-stranded RNA.
- Another aspect provides a method of enhancing an innate immune response comprising administering to a subject the pharmaceutical composition.
- Another aspect provides the use of said double-stranded RNA for the manufacture of a medicament for treating or ameliorating a disease or pathological condition.
- Another aspect provides the use of said double-stranded RNA for the manufacture of a medicament for enhancing innate immune response.
- the above method or use includes or utilizes the above-described double-stranded RNA or pharmaceutical composition as it is, the description of common contents between them is omitted to avoid excessive complexity of this specification.
- the disease or pathological condition is as described above.
- subject means a subject in need of treatment for a disease, specifically a condition, and more specifically, may include any mammal, such as a human or non-human primate, mouse, dog, cat, horse, cow, sheep, pig, goat, camel, or antelope.
- double-stranded RNAs of various lengths capable of activating innate immunity were produced.
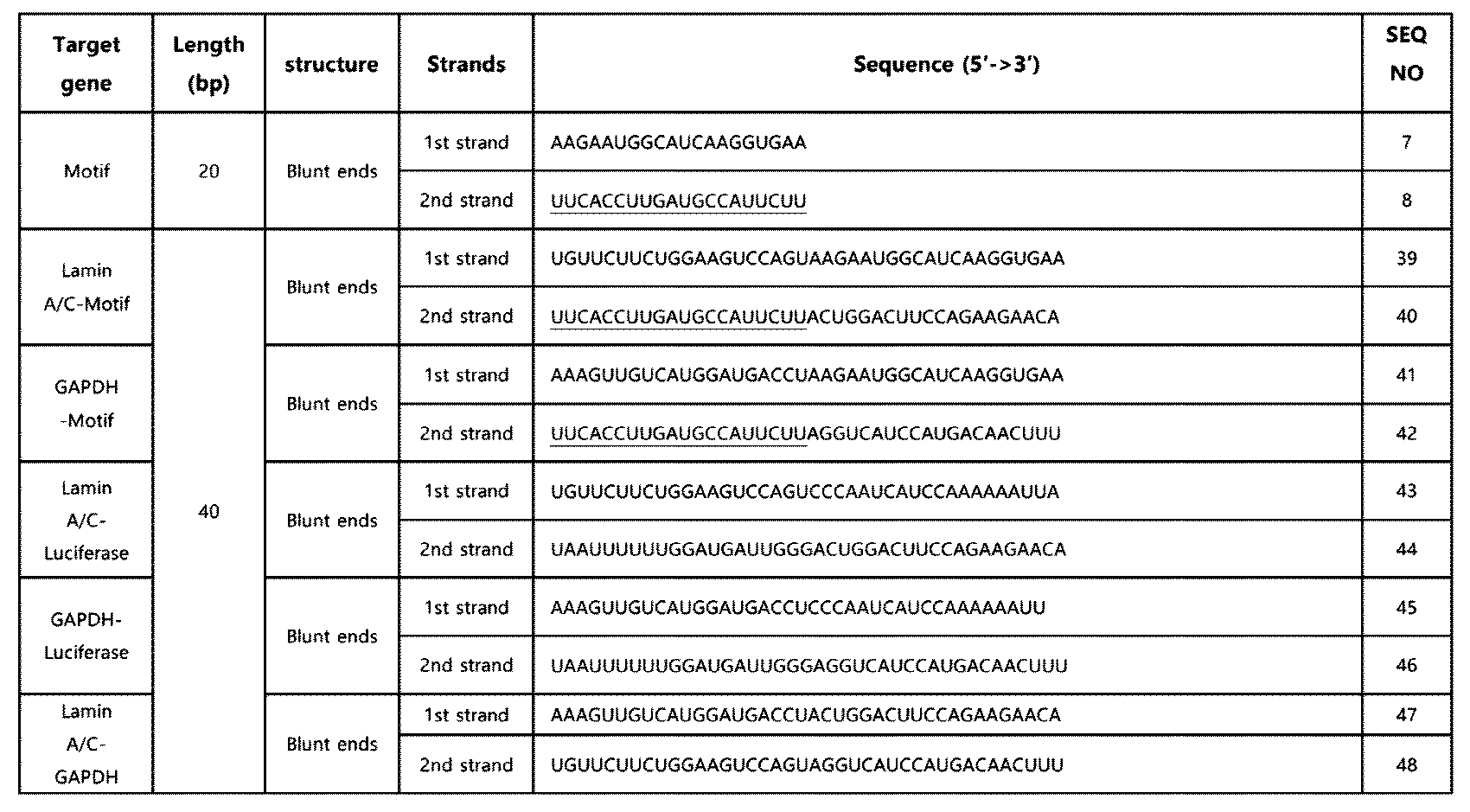
- the double-stranded RNA is composed of 38 to 60 base pairs, which are composed of a first strand and a second strand complementarily binding to the first strand, and includes an innate immune activation motif located at the 3' terminal region of the first strand and the 5' terminal region of the second strand, wherein the innate immune activation sequence of SEQ ID NO: 6 (19 nt) or SEQ ID NO: 8 (20 nt) is located at the 5' terminal region of the second strand.
- the terminal region of the double-stranded RNA was modified based on the above-described basic structure to produce double-stranded RNAs having a smooth end, one protrusion, or two protrusions, respectively.
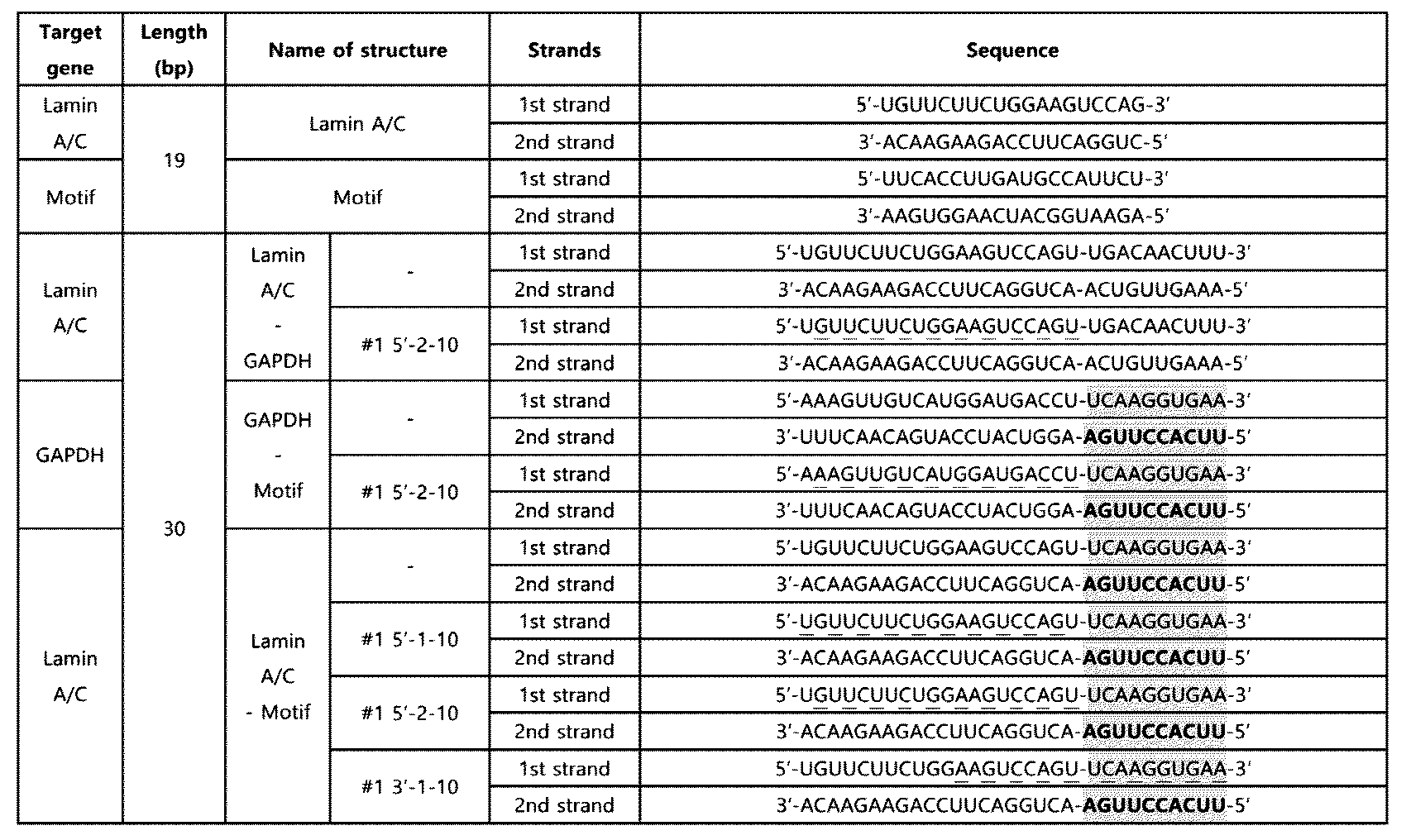
- the double-stranded RNA was purchased from Bioneer Co. Ltd (Republic of Korea), and Dharmacon, Inc (UK), and the double-stranded RNA comprising the innate immune activation sequence of SEQ ID NO: 6 is as shown in Table 1, and the double-stranded RNA comprising the innate immune activation sequence of SEQ ID NO: 8 is as shown in Table 2.
- Tables 1 and 2 the innate immune activation sequence is underlined, and the protrusion formed in the terminal region is shaded.
- the knockdown effect of the double-stranded RNA according to Example 1 on the target gene was confirmed.
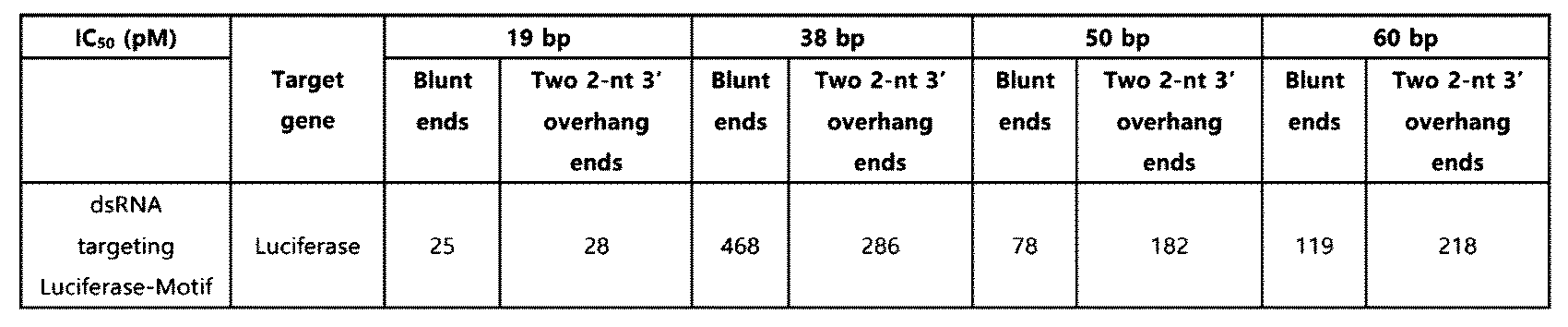
- the knockdown effect of the 19, 38, 50, or 60 bp double-stranded RNA (Table 1) containing the innate immune activation motif according to the above Example 1 on the Luciferase gene was confirmed in HeLa (CCL-2, ATCC) cell line treated with Luciferase plasmid (E1741, Promega).
- the HeLa (CCL-2, ATCC) cell line was plated in a 24-well plate at 30% confluency in complete medium without antibiotics. Then, the cell line was transfected with double-stranded RNA using Lipofectamine RNAiMAX (13778150; Invitrogen).
- Primers for the target gene were mixed with SYBR Green PCR Master Mix and quantified by relative standard curve. The target gene mRNA levels were measured using the relative standard curve quantitation method. Then, IC 50 values were calculated based on the dose-response curve using GraphPad Prism 8.
- cytokines related to the innate immune response was analyzed for the 38, 50, or 60 bp double-stranded RNAs comprising the innate immune activation sequence of SEQ ID NO: 6 according to Example 1 (Table 1) and the 40 bp double-stranded RNAs comprising the innate immune activation sequence of SEQ ID NO: 8 (Table 2).
- T98G (CRL-1690, ATCC) cell line was plated in a 24-well plate at 30% confluency in complete medium without antibiotics.
- the double-stranded RNA was transfected into the cell line using Lipofectamine RNAiMAX (13778150; Invitrogen), and the transfected cells were harvested after 12 or 24 hours, respectively. Thereafter, the levels of IFIT1, ISG15, and CXCL10 were analyzed using qRT-PCR according to the method of Example 2-1. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as a group to which only the transfection reagent was added (mock) and a group not treated with double-stranded RNA (NT).
- the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 6 induced high expression of IFIT1, CXCL10, ISG15, TNF- ⁇ , and IL-1 ⁇ related to the innate immune response.
- this effect was more excellent when the terminal structure of the double-stranded RNA was a blunt end, and was also very remarkable compared to the effect of the double-stranded RNA having the same structure as above but without the motif introduced at the terminal of the double-stranded RNA.
- the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 8 also induced high expression of IFIT1, CXCL10, and ISG15 related to the innate immune response.
- the group treated with the 40 bp double-stranded RNA (Lamin A/G-GAPDH, Lamin A/C-Luciferase, GAPDH-Luciferase) having the same structure as above but without the motif introduced at the end of the double-stranded RNA, and the group treated by mixing each of the short-length target binding sequence and motif sequence (mixture of 19 bp) did not induce an effective level of innate immune response. Therefore, it is shown that the above-mentioned innate immune response induction effect is derived from the innate immune activation motif according to one embodiment introduced into the double-stranded RNA.
- the RAW-Lucia ISG cell line and the RAW-Lucia ISG-KO-RIG-I cell line were plated at 1 ⁇ 10 5 cells per well in a 96-well plate. Thereafter, the double-stranded RNA (10 nM) was transfected into the cell line using Lipofectamine RNAiMAX (13778150; Invitrogen), and the transfected cells were each cultured for 24 hours to obtain the supernatant. Thereafter, the interferon regulatory factor (IRF) activity level was detected in the supernatant using a plate reader (VICTORX2; PerkinElmer).
- IRF interferon regulatory factor
- the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
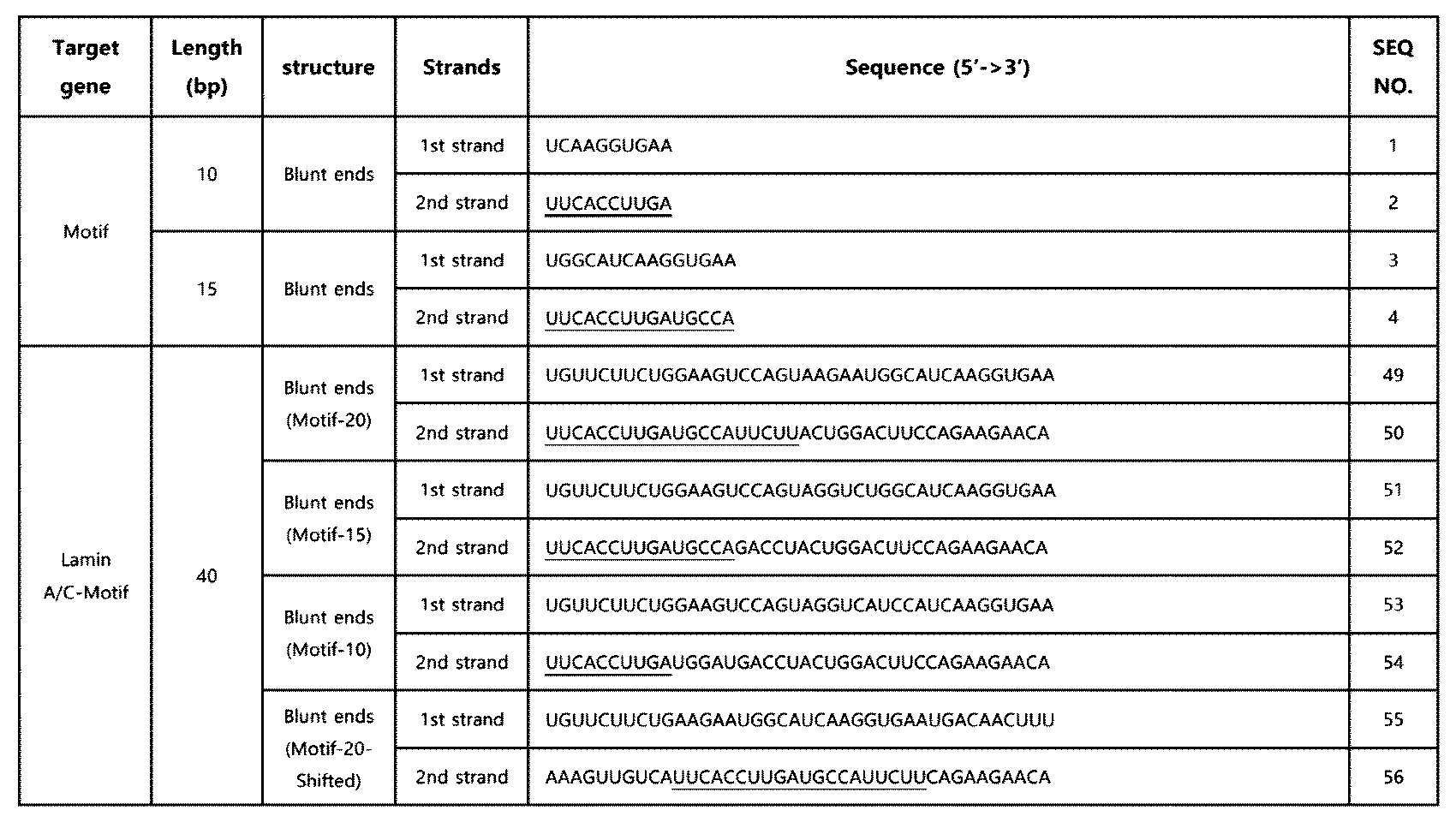
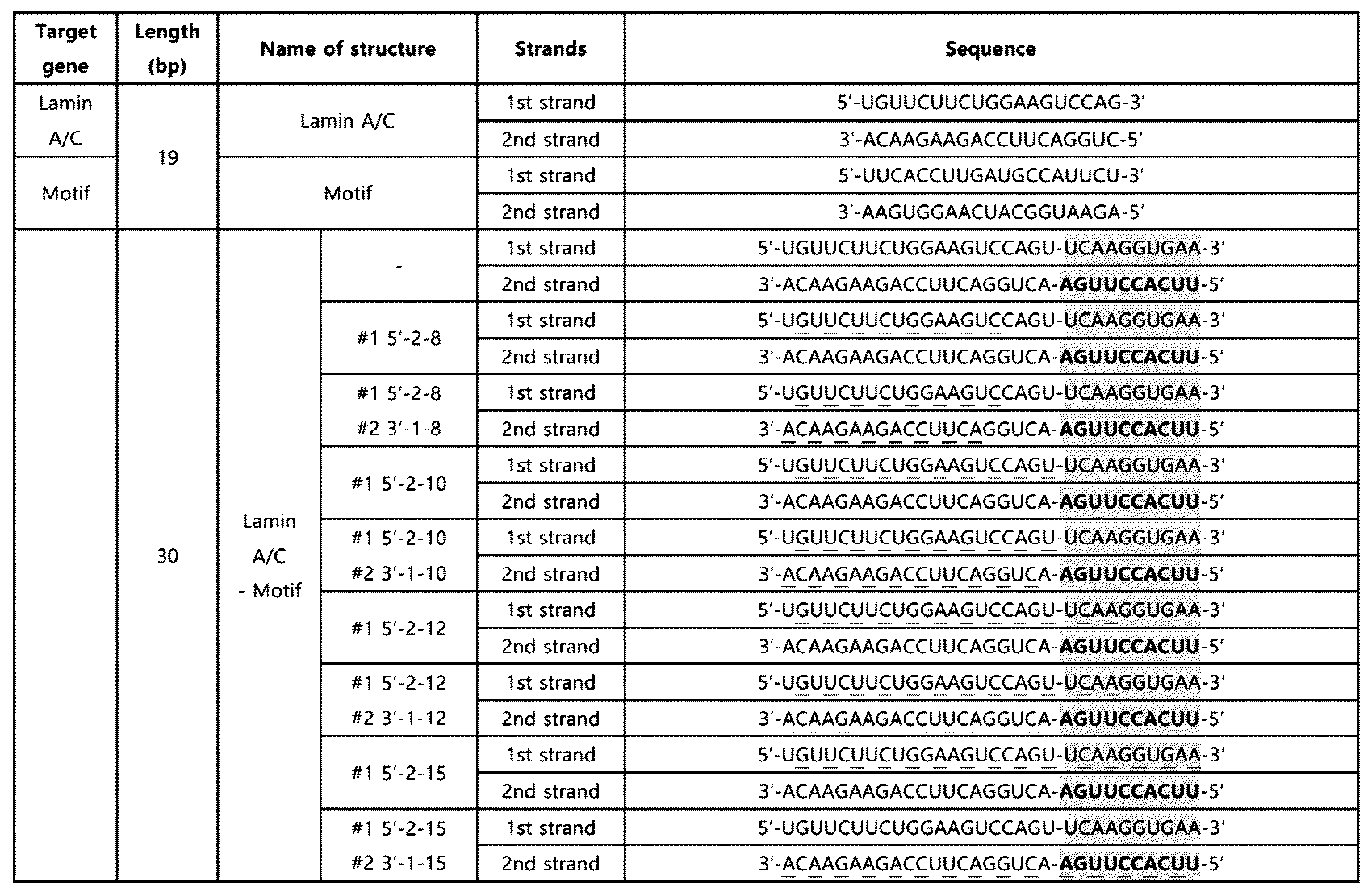
- RNA including a modified innate immune activation motif was produced in which the nucleotide sequence of SEQ ID NO: 2 (10 nt) or the nucleotide sequence of SEQ ID NO: 4 (15 nt) is located at the 5' end of the second strand, and its specific structure is as shown in Fig. 1.
- a double-stranded RNA in which the nucleotide sequence of SEQ ID NO: 8 was introduced into the extra-terminal region was additionally produced.
- the double-stranded RNA was obtained from Bioneer Co. Ltd (Republic of Korea), and Dharmacon, Inc (UK), and the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 2 or SEQ ID NO: 4 is as shown in Table 4.
- the innate immune activation sequence is underlined.
- the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured.
- the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
- the double-stranded RNA including the nucleotide sequence of SEQ ID NO: 2 or the nucleotide sequence of SEQ ID NO: 4 at the terminal was also able to enhance the RIG-I-mediated innate immune response, similarly to Example 2-3.
- the double-stranded RNA (Motif-20-Shifted) in which the nucleotide sequence of SEQ ID NO: 8 was shifted to the center of the entire sequence showed a significant decrease in this effect.
- the motif of SEQ ID NO: 2 consisting of 10 nt can be applied as a unit or core sequence for inducing the RIG-I-mediated innate immune response, and that the sequence can exhibit effective efficacy when introduced or positioned at the terminal in the double-stranded RNA.
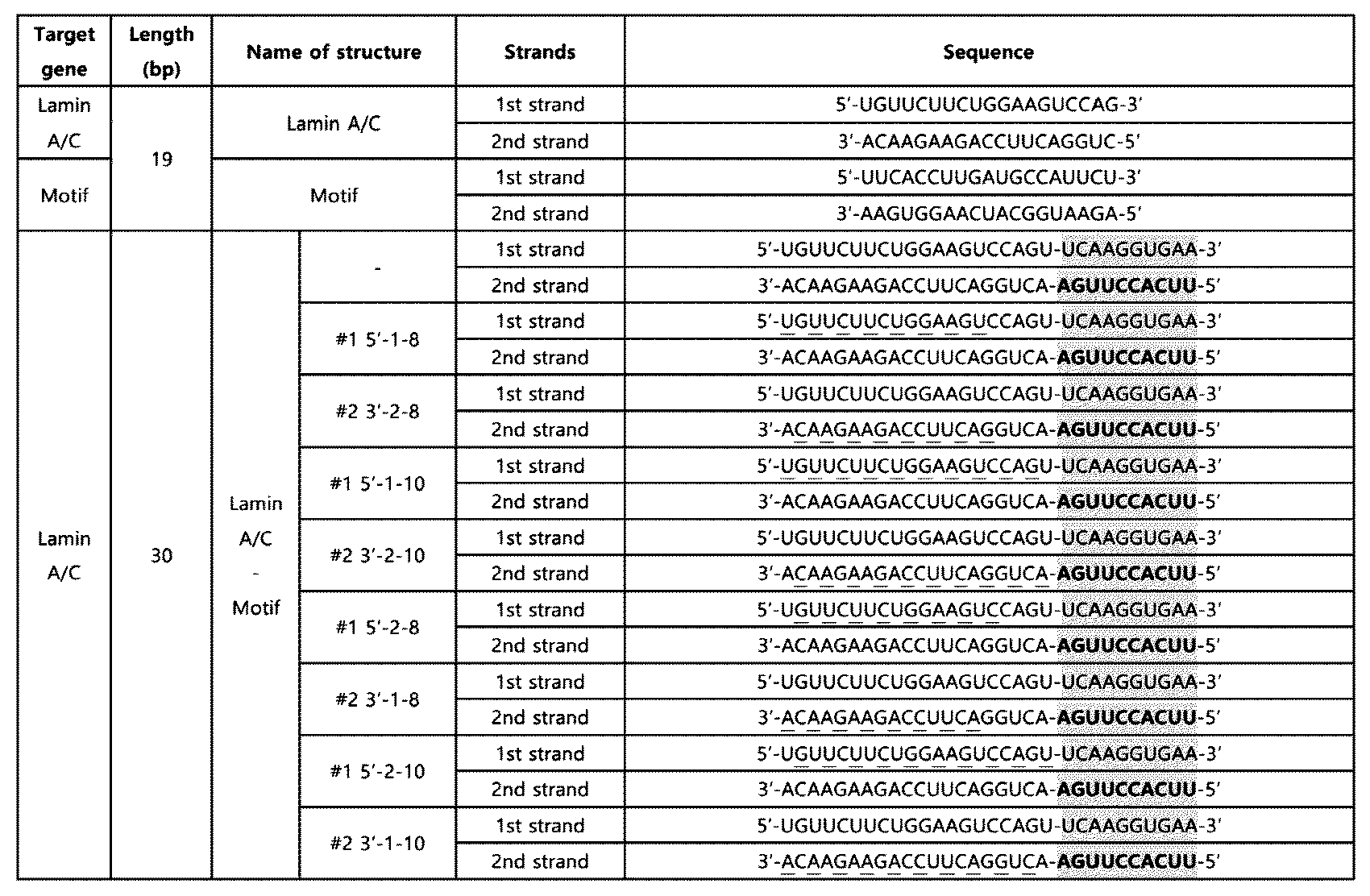
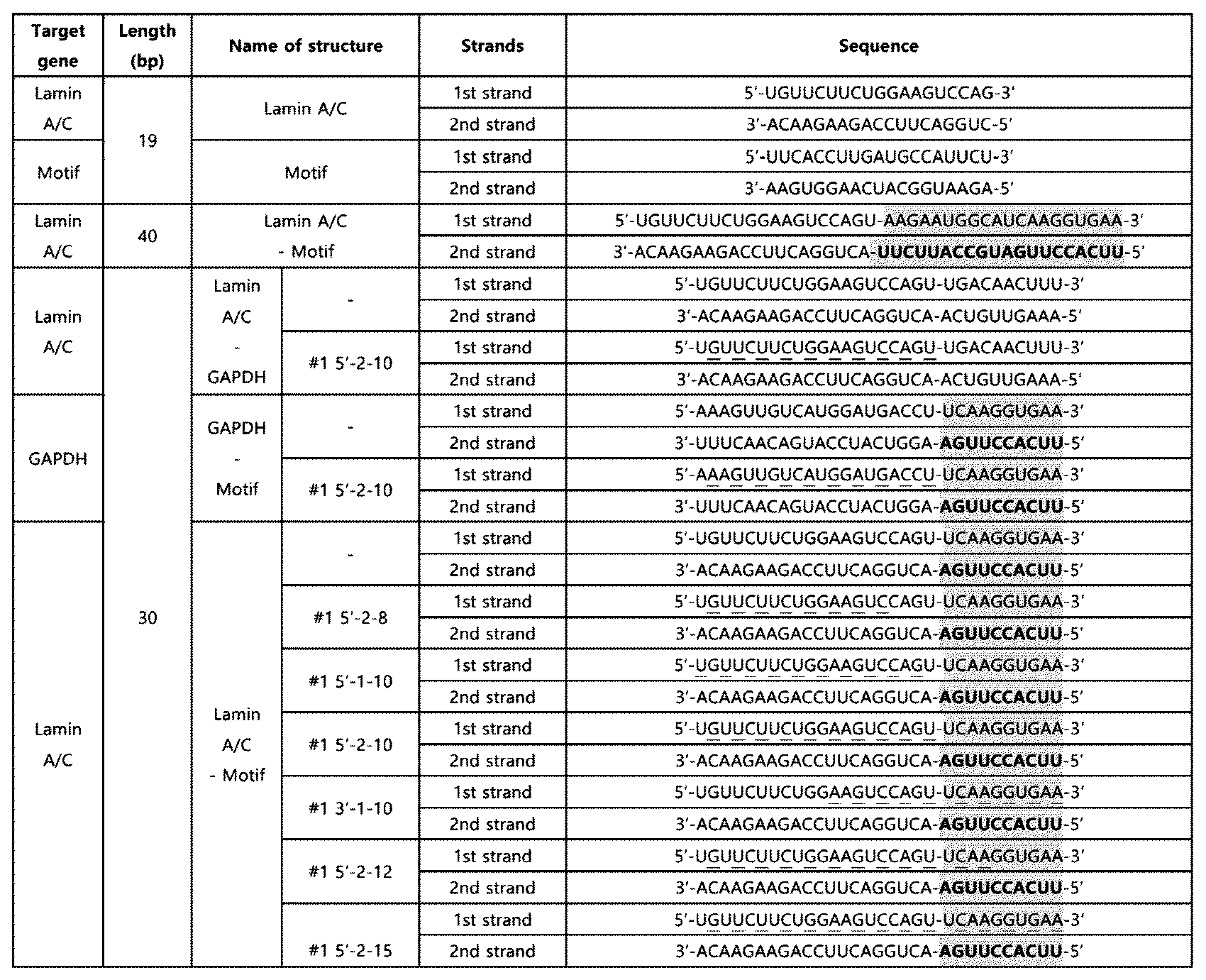
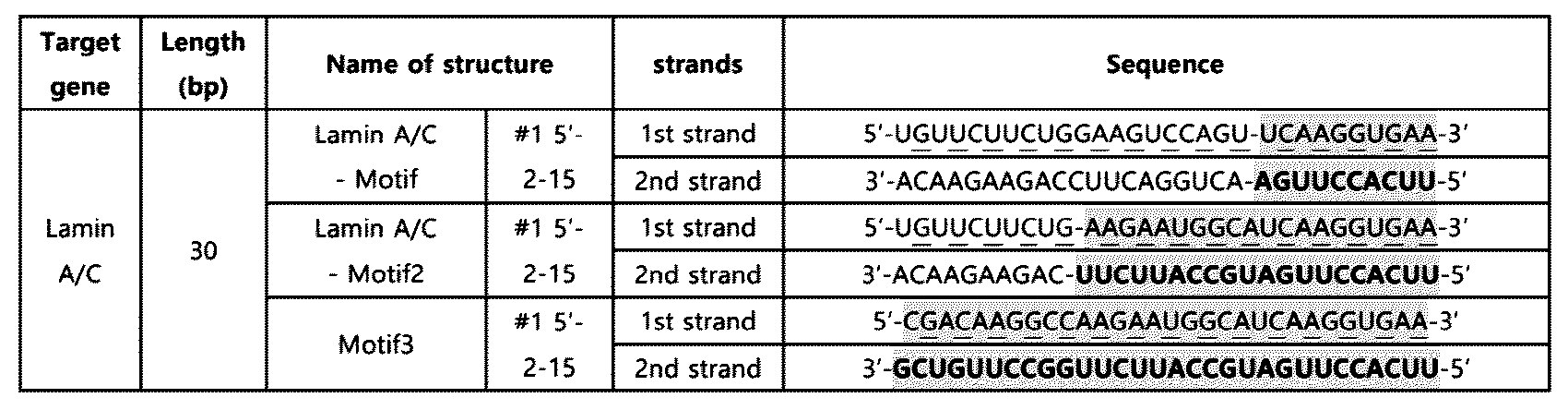
- RNA based on the experimental results of Example 2 and Example 3-1, a 30 bp double-stranded RNA was prepared in which the nucleotide sequence (10 nt) of SEQ ID NO: 2 is located at the 5' end of the second strand or the 3' end of the first strand in the double-stranded RNA and both ends are blunt ends.
- a double-stranded RNA (LaminA/C-Motif-10) in which the innate immune activation sequence is located at the 5' end of the second strand and a double-stranded RNA (LaminA/C-inv.
- the double-stranded RNA was obtained from Bioneer Co. Ltd (Republic of Korea), and Dharmacon, Inc (UK), and the above-mentioned double-stranded RNAs are as shown in Table 5.
- Table 5 the innate immune activation sequences are underlined.
- the knockdown effect of the target gene, Lamin A/C, upon treatment with 10 nM of double-stranded RNA was confirmed using the Huh7 cell line in the same manner as in Example 2-1.
- the double-stranded RNA having a length of 30 bp also showed a knockdown effect on the target gene, similar to Example 2-1.
- a double-stranded RNA containing an innate immune activation motif was prepared by introducing chemical modifications into a double-stranded RNA, and the innate immune response induction effect of the double-stranded RNA with the chemical modifications was evaluated.
- RNA having the innate immune activation sequence of sequence number 2 inserted at the 5' end of the second strand was prepared as a basic backbone, and a double-stranded RNA was prepared in which a chemical modification was introduced in which the -OH group at the 2' carbon position of the nucleotide was substituted with -OCH 3 (-O-methyl).
- Double-stranded RNA having a chemical modification introduced into either one of the first strand or the second strand, and double-stranded RNA having chemical modifications introduced into both the first strand and the second strand were respectively prepared.
- double-stranded RNA having a chemical modification introduced into either one of the first strand or the second strand are as shown in Table 6, and double-stranded RNA having chemical modifications introduced into both the first strand and the second strand are as shown in Table 7.
- Tables 6 and 7 the nomenclature of double-stranded RNA is expressed as [strand having chemical modification introduced]-[terminus having chemical modification introduced and first nucleotide position having chemical modification introduced]-[number of nucleotides having chemical modification introduced].
- the innate immune activation motif and the innate immune activation sequence are respectively shaded and/or bolded, and the nucleotides having 2'-OCH 3 (-O-methyl) chemical modification introduced are underlined.
- the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured.
- the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
- the double-stranded RNA containing eight nucleotides to which chemical modifications were introduced (#1 5'-1-8, #2 3'-2-8, #1 5'-2-8, and #2 3'-1-8) completely lost the effect derived from the innate immune response motif, i.e., the innate immune response induction effect.
- the double-stranded RNA with the chemical modifications introduced as described above is as shown in Table 8.
- Table 8 above innate immune activation motifs and innate immune activation sequences are shaded and/or bold, respectively, and nucleotides with 2'-OCH 3 (-O-methyl) chemical modifications are underlined.
- the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured.
- the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
- the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured.
- the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
- the double-stranded RNA including the innate immune activation motif when at least 10 chemical modifications (10, 12, 15) were introduced, a high level of innate immunity induction effect was exhibited. In addition, this effect was significantly reduced in the ISG-KO-RIG-I cell line lacking RIG-I.
- the double-stranded RNA including 8 nucleotides into which chemical modifications were introduced, and the double-stranded RNA into which chemical modifications were introduced into both the first strand and the second strand all innate immune response induction effects derived from the innate immune response motif were lost.
- the effect of enhancing the RIG-I-mediated innate immune response is achieved regardless of the sequence of the region outside the innate immune activation motif in the double-stranded RNA and the location of the innate immune activation motif in the single strand.
- the double-stranded RNA with the chemical modification of Example 4 above showed an enhanced innate immune response induction effect compared to the double-stranded RNA according to Examples 1 to 3 without the chemical modification.
- the double-stranded RNA with the chemical modification introduced unlike the double-stranded RNA without the chemical modification introduced, showed a certain level of IRF activity without completely disappearing or significantly reducing the innate immune response in the ISG-KO-RIG-I cell line lacking RIG-I (see FIGS. 10, 12, and 13).
- the activity of IRF was measured for RAW-Lucia ISG cell line (WT), RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO), and RAW-Lucia ISG-KO-MDA5-I cell line (MDA5 KO) in the same manner as in Example 2-3.
- the double-stranded RNAs into which chemical modifications were introduced used in this example are as shown in Table 10.
- the innate immune activation motifs and innate immune activation sequences are indicated in shaded and/or bold, respectively, and the nucleotides into which 2'-OCH 3 (-O-methyl) chemical modifications were introduced are indicated in underline.
- Example 6 Verification of the effect of inducing innate immune response using human monocyte cell line
- the effect of inducing an innate immune response according to the treatment of the double-stranded RNA with the chemical modification of Example 4 was verified again, using the human monocyte cell line THP1-Dual (thpd-nfis) cell line.
- a 30-bp double-stranded RNA in which an innate immune activation sequence consisting of nucleotides of SEQ ID NO: 2 is inserted at the 5' end of the second strand as a basic framework was prepared to introduce a chemical modification in which the -OH group at the 2' carbon position of the nucleotide is substituted with -OCH 3 (-O-methyl).
- Tables 6, 7, and 9 information on the double-stranded RNA with the chemical modification introduced used in this example is as shown in Tables 6, 7, and 9 above.
- double-stranded RNA (10 nM) was transfected into the THP1-Dual (thpd-nfis) cell line using Lipofectamine RNAiMAX (13778150; Invitrogen), and the transfected cells were cultured for 24 or 48 hours, respectively, and then the supernatant was obtained. Thereafter, the level of interferon regulatory factor activity was detected in the supernatant using a plate reader (VICTORX2; PerkinElmer).
- the positive control group was a group administered with Poly I:C, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
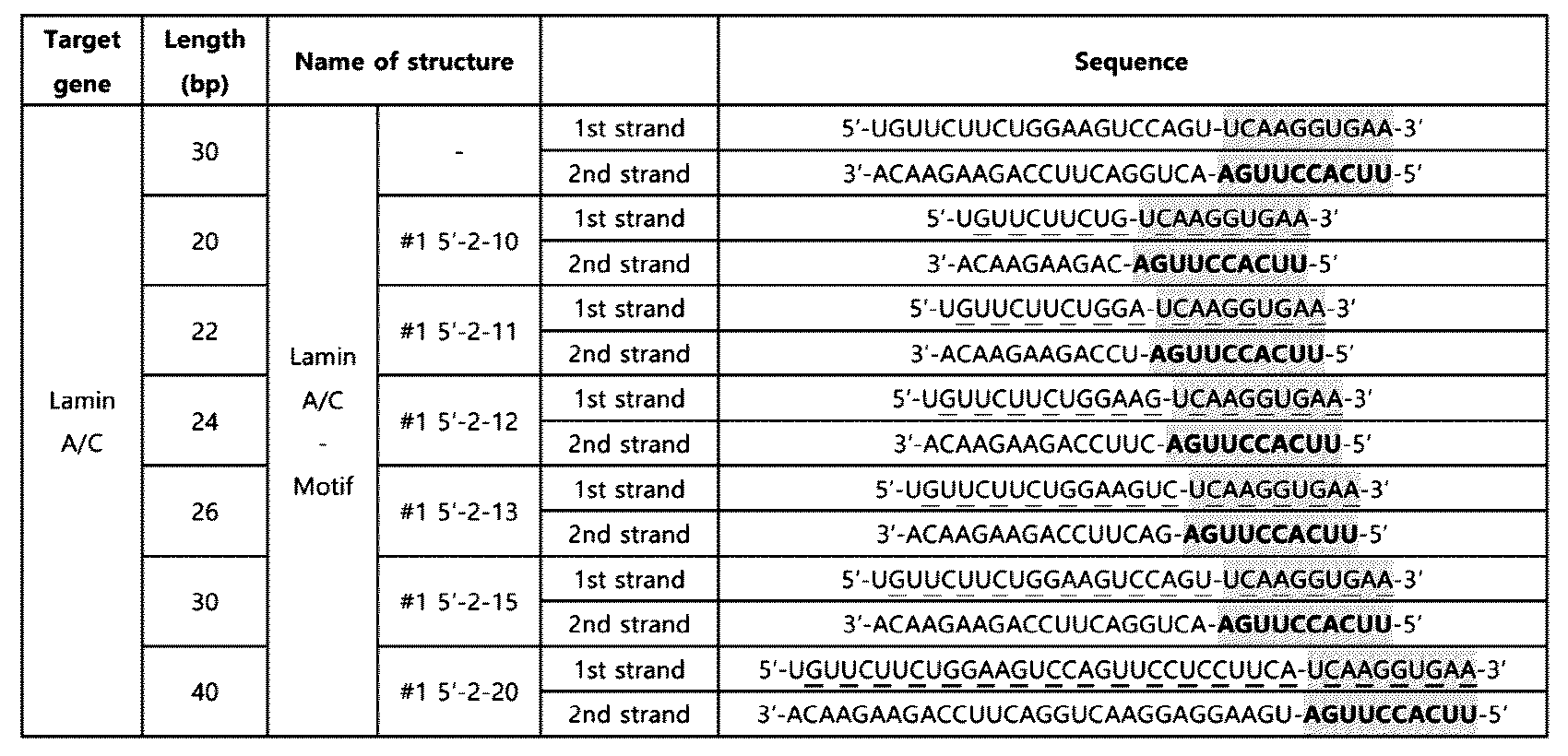
- the 30 bp double-stranded RNA (#1 5'-2-15), which showed a high level of innate immunity induction effect in all of the above Examples 4-3, 5, and 6, was used to evaluate the innate immune response induction effect according to the modification of the entire nucleotide length.
- the innate immune activation sequence of SEQ ID NO: 2 was inserted at the 5' end of the second strand, and a double-stranded RNA in which 50% of the chemical modifications for the entire nucleotide length were introduced into the first strand was used as the basic backbone, and the length of the nucleotides was changed to 20 to 40 bp so as to include at least 10 or more chemical modifications (#1 5'-2-10, #1 5'-2-11, #1 5'-2-12, #1 5'-2-13, #1 5'-2-15, #1 5'-2-20).
- RNAs into which the above-mentioned chemical modifications were introduced are as shown in Table 11.
- innate immune activation motifs and innate immune activation sequences are shaded and/or bold, respectively, and nucleotides with 2'-OCH 3 (-O-methyl) chemical modifications are underlined.
- the 20-24 bp double-stranded RNA containing the innate immune activation motif and having at least 10 2'-OCH3(-O-methyl) chemical modifications introduced into either the first strand or the second strand completely lost the effect derived from the innate immune response motif, i.e., the innate immune response induction effect.
- the 26-40 bp double-stranded RNA containing the innate immune activation motif and having at least 10 2'-OCH3(-O-methyl) chemical modifications introduced into either the first strand or the second strand exhibited a high level of innate immune induction effect.
- the double-stranded RNA containing an innate immune activation motif according to one embodiment and having at least 10 chemical modifications introduced into either the first strand or the second strand exhibited efficacy due to the innate immune activation motif and chemical modifications in the range of a total nucleotide length of 26 bp to 40 bp.
- the 30 bp double-stranded RNA that showed a high level of innate immunity induction effect in Example 7 was used as the target for the human monocyte cell line, THP1-Dual (thpd-nfis), to evaluate the innate immune response induction effect according to the presence or absence of the innate immune activation motif.
- THP1-Dual thpd-nfis
- a double-stranded RNA with or without an innate immune activation motif was produced.
- the 30 bp double-stranded RNAs with or without the above-mentioned innate immune activation motif are as shown in Table 12.
- the innate immune activation motif and the innate immune activation sequence are indicated in shaded and/or bold, respectively, and the nucleotides introduced with a 2'-OCH 3 (-O-methyl) chemical modification are indicated in underline.
- Example 6 Thereafter, in the same manner as in Example 6, the activity of IRF for the THP1-Dual (thpd-nfis) cell line was measured. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT). The results are shown in Fig. 17. The result using 30 bp Lamin A/C-GAPDH without 2'-OCH3(-O-methyl) chemical modification in Table 12 is denoted as Lamin A/C-GAPDH in Fig.
- the 30 bp double-stranded RNA containing the innate immune activation motif and having at least 10 2'-OCH3(-O-methyl) chemical modifications introduced into either the first or second strand exhibited a high level of innate immune induction effect in human monocyte cell lines, similar to the experimental result of Example 7.
- the group treated with the 30 bp double-stranded RNA (Lamin A/G-GAPDH) having the same structure as above but without the motif introduced into the end of the double-stranded RNA did not induce a significant level of innate immune response. Therefore, it was confirmed once again that the introduction of the innate immune activation motif is an essential element for the innate immune induction effect.
- the innate immune activation sequences of SEQ ID NO: 8 (20 nt) and SEQ ID NO: 62 (30 nt) are shown in Table 13, and the 30 bp double-stranded RNA (Lamin A/C- Motif2, Motif3) containing the innate immune activation sequence of SEQ ID NO: 8 (20 nt) or SEQ ID NO: 62 (30 nt) in the 5'-terminal region of the second strand are as shown in Table 14.
- the innate immune activation motif and the innate immune activation sequence are shaded and/or bold, respectively, and the nucleotides introduced with the 2'-OCH 3 (-O-methyl) chemical modification are underlined.
- the double-stranded RNA including the nucleotide sequence of sequence number 8 at the end also exhibited a high level of innate immunity induction effect, similar to Example 7, and it was confirmed that the nucleotide sequence of sequence number 62 also exhibited an excellent innate immunity induction effect.
- the double-stranded RNA with chemical modification introduced according to one embodiment can exhibit effective innate immune induction efficacy even in the modified innate immune activation motif.
- the 30 bp double-stranded RNA into which the above-mentioned 2'-F chemical modification was introduced is as shown in Table 15.
- the innate immune activation motif and the innate immune activation sequence are indicated in shaded and/or bold, respectively, and the nucleotides into which the 2'- OCH3 (-O-methyl) or 2'-F chemical modification was introduced are indicated in underline.
- the activity of IRF for the RAW-Lucia ISG cell line was measured in the same manner as in the above Example 2-3.
- the activity of IRF for the THP1-Dual (thpd-nfis) cell line was measured in the same manner as in the above Example 6.
- the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT).
- the results for the RAW-Lucia ISG cell line are shown in Fig. 19, and the results for the THP1-Dual (thpd-nfis) cell line are shown in Fig. 20.
- the double-stranded RNA into which 2'-F or 2'-OCH 3 (-O-methyl) chemical modifications were introduced exhibited an effective innate immune response induction effect, and in particular, it was confirmed that the double-stranded RNA into which 2'-OCH 3 (-O-methyl) chemical modifications were introduced exhibited a much higher level of innate immune induction effect. In addition, it was confirmed that this tendency was similarly shown in Fig. 20 using a human monocyte cell line.
- RNA stability we attempted to verify whether the innate immunity induction effect could be maintained even when the nucleotide bond was modified to improve RNA stability.
- a double-stranded RNA was additionally produced in which two nucleotide bonds adjacent to each other from at least one strand, i.e., the bond between the 1st nucleotide and the 2nd nucleotide and the bond between the 2nd nucleotide and the 3rd nucleotide, were modified from phosphodiester bond to phosphorothioate bond.
- the 30 bp double-stranded RNA in which the nucleotide bond described above was modified is as shown in Table 16.
- innate immune activation motifs and innate immune activation sequences are shaded and/or bold, respectively, nucleotides with 2'-OCH3(-O-methyl) chemical modifications are underlined, and forms in which nucleotide bonds are substituted with phosphorothioate (PS) bonds are marked with "*".
- the 30 bp double-stranded RNA which showed a high level of innate immunity induction effect in the above Example 7, was treated to various cell lines to re-verify the innate immune response induction effect.
- RAW-Lucia ISG, EMT-6, or CT-26 cell lines were used to measure the protein expression levels of RIG-I and MDA5 by Western blot, and the expression levels of RIG-I, MDA5, and IFIT1 mRNA by RT-qPCR.
- THP1-Dual (thpd-nfis), THP, THP-PMA, PBMS, HCC-1143, SK-BR-3, HCC-70, HCC-38, BT-549, SK BR3, Hep3 B, SN12C, or A498 cell lines were used to measure the mRNA expression levels of RIG-I and MDA5 by RT-qPCR. The results are shown in Table 17 below.
- Example 7 As a result, as shown in Table 17 above, when the 30 bp double-stranded RNA of Example 7 was applied, it was confirmed that MDA and RIG-I-mediated innate immune responses could be effectively induced in human immune cells such as THP, THP differentiated by PMA treatment (THP-PMA), and PBMC, as well as breast, liver, and renal cancer cell lines.
- human immune cells such as THP, THP differentiated by PMA treatment (THP-PMA), and PBMC, as well as breast, liver, and renal cancer cell lines.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Epidemiology (AREA)
- Biotechnology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Immunology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Microbiology (AREA)
- General Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Mycology (AREA)
- Plant Pathology (AREA)
- Biophysics (AREA)
- Physics & Mathematics (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
본 발명은 선천성 면역 반응 유도 효과를 갖는 이중가닥 RNA 및 이의 용도에 관한 것으로서, 더욱 상세하게는 선천성 면역 활성화 모티프 및 화학적 변형이 도입된 뉴클레오티드를 포함하는 이중가닥 RNA를 제공한다. The present invention relates to a double-stranded RNA having an innate immune response inducing effect and a use thereof, and more particularly, to a double-stranded RNA comprising a nucleotide into which an innate immune activation motif and a chemical modification have been introduced.
선천성/후천성 면역 반응의 유도는 다양한 질병의 치료에서 중요한 역할을 한다. 이 중에서도 선천성 면역 반응 (innate immune response)은 신체의 비특이적 면역 반응으로 외부 인자를 인식한 후, 가장 먼저 반응하는 신체의 1차적 방어선 역할을 수행한다. 선천성 면역 반응을 증진 또는 활성화시켜 치료가 어려운 암, 병원균 감염, 유전병과 같은 병리적 상태를 개선시킬 수 있다는 점이 알려진 후, 상기 선천성 면역 반응을 이용한 치료제에 대한 연구가 활발하게 진행되었다. 이를 위하여, 선천성 면역 반응을 활성화 또는 조절할 수 있는 작용 기작 등에 대한 연구가 이루어져 왔다. 예컨대, 항암 치료로서 선천성 면역 반응을 활성화시키는 경우, 케모카인 생성 및 염증 촉진을 통한 암세포 부위로 대식세포 또는 수지상세포 모집이 활성화되고, 정상 상태를 유도하기 위해 인터페론과 같은 사이토카인의 생성을 증가시킬 수 있음이 보고된 바 있다. Induction of innate/adoptive immune responses plays an important role in the treatment of various diseases. Among these, the innate immune response is a non-specific immune response of the body, and it acts as the first line of defense of the body to recognize external factors and respond first. Since it has been known that pathological conditions such as cancer, pathogenic infection, and hereditary diseases that are difficult to treat can be improved by enhancing or activating the innate immune response, research on therapeutics using the innate immune response has been actively conducted. To this end, research has been conducted on the mechanism of action that can activate or regulate the innate immune response. For example, it has been reported that when activating the innate immune response as an anticancer treatment, the recruitment of macrophages or dendritic cells to the cancer cell site is activated through chemokine production and promotion of inflammation, and the production of cytokines such as interferon can be increased to induce a normal state.
또한, 외부로부터 유래하거나 손상된 핵산을 감지하는데 특화된 일부 선천성 면역 수용체가 확인되었다. 패턴 인식 수용체 (pattern recognition receptor: PRR)는 세포의 세포막이나 세포질에 존재하는 단백질로서, 유형 인식 분자를 인식하여 선천성 면역 반응을 매개하는 역할을 한다. 패턴 인식 수용체는 수지상 세포, 대식세포, 단핵구, 호중구, 상피세포 등에 분포하여, 병원체연관분자유형 (pathogen-associated molecular pattern: PAMP) 및 손상연관분자유형 (damage-associated molecular pattern: DAMP)을 인식한다. 대표적인 패턴 인식 수용체로는 톨 유사 수용체 (Toll-like receptor: TLR), C형 렉틴 수용체 (C-type lectin receptor, CLR), RIG-I-유사 수용체 (Retinoic acid-inducible gene-I-like receptor: RLR), NOD-유사 수용체 (Nucleotide oligomerization domain-like receptor: NLR), AIM-유사 수용체 (Absent-in-melanoma-like receptor: ALR) 등이 있다. 각각의 수용체들은 서로 다른 종류의 리간드를 인식하여 다양한 방식으로 면역계 증진에 기여한다. In addition, some innate immune receptors specialized in detecting foreign or damaged nucleic acids have been identified. Pattern recognition receptors (PRRs) are proteins present in the cell membrane or cytoplasm of cells, which recognize pattern recognition molecules and mediate innate immune responses. Pattern recognition receptors are distributed in dendritic cells, macrophages, monocytes, neutrophils, epithelial cells, etc., and recognize pathogen-associated molecular patterns (PAMPs) and damage-associated molecular patterns (DAMPs). Representative pattern recognition receptors include Toll-like receptors (TLR), C-type lectin receptors (CLR), retinoic acid-inducible gene-I-like receptors (RLR), nucleotide oligomerization domain-like receptors (NLR), and absence-in-melanoma-like receptors (ALR). Each receptor recognizes different types of ligands and contributes to the enhancement of the immune system in various ways.
한편, RNA 기반 치료로서 RNA 간섭 (RNA interference, RNAi) 현상을 이용한 기술이 개발되어 왔고, 이러한 RNA 간섭은 표적 mRNA의 절단을 유도하여 표적 유전자 발현을 억제하는 방법이다. 구체적으로, RNAi 제제로서, 짧은 간섭 RNA(small interfering RNA)를 사용할 수 있는데, 이 경우, 외부 물질인 siRNA의 투여에 의한 선천성 면역 반응의 유도는 비특이적 반응으로, 오히려 RNAi 제제의 효능을 저해시키는 요인으로 인식되었다. 이에, 변형된 RNA 뉴클레오티드의 도입, 예를 들어, 화학적 변형이 도입된 뉴클레오티드의 도입이 siRNA의 투여에 의한 선천성 면역 반응을 감소시키기 위한 기술로 제시된 바 있다. 다만, RNA 기반 치료제는 전술한 RNAi 제제로의 사용 뿐만 아니라, 수동 및 능동 면역 요법, 백신 제제, 또는 유전 공학 전반에서 적용될 수 있다. 또한, RNA 분자는 예를 들어, 결손되거나 돌연변이된 단백질을 대체하기 위한 단백질 대체 요법과 같은 대체 요법을 위한 치료제로도 사용될 수 있다.Meanwhile, technologies utilizing the RNA interference (RNAi) phenomenon have been developed as RNA-based treatments, and this RNA interference is a method of suppressing target gene expression by inducing cleavage of target mRNA. Specifically, short interfering RNA (small interfering RNA) can be used as an RNAi agent, and in this case, the induction of an innate immune response by administration of an external substance, siRNA, is recognized as a non-specific response and rather as a factor that inhibits the efficacy of the RNAi agent. Accordingly, the introduction of modified RNA nucleotides, for example, the introduction of nucleotides with chemical modifications, has been suggested as a technology for reducing the innate immune response by administration of siRNA. However, RNA-based therapeutics can be applied not only to the aforementioned RNAi agents, but also to passive and active immunotherapy, vaccine agents, or genetic engineering in general. In addition, RNA molecules can also be used as therapeutics for replacement therapy, such as protein replacement therapy to replace missing or mutated proteins.
이러한 기술적 배경 하에서, 본 발명자들은 선천성 면역 반응을 특이적으로 활성화하여 치료 효능을 향상시킬 수 있는 이중가닥 RNA를 개발하기 위해 연구 노력한 결과, 특정 모티프 및 뉴클레오티드의 화학적 변형으로부터 비롯된 선천성 면역 반응 증진 효과를 확인하고, 이에 기초하여 본 발명을 완성하였다. Under this technical background, the inventors of the present invention have conducted research efforts to develop a double-stranded RNA capable of specifically activating an innate immune response to enhance therapeutic efficacy, and as a result, have confirmed the effect of enhancing an innate immune response resulting from chemical modification of specific motifs and nucleotides, and have completed the present invention based on this.
본 발명의 목적은 선천성 면역 반응을 특이적으로 활성화시킬 수 있는 이중가닥 RNA를 제공하는데 있다.The purpose of the present invention is to provide double-stranded RNA capable of specifically activating an innate immune response.
본 발명의 다른 목적은 상기 이중가닥 RNA를 포함하는 선천성 면역 반응 증진용 조성물, 상기 이중가닥 RNA를 포함하는 약학적 조성물, 또는 상기 약학적 조성물을 개체에 투여하는 단계를 포함하는 질병을 치료하는 방법 또는 병리적 상태를 개선하는 방법을 제공하는데 있다. Another object of the present invention is to provide a composition for enhancing an innate immune response comprising the double-stranded RNA, a pharmaceutical composition comprising the double-stranded RNA, or a method for treating a disease or improving a pathological condition comprising a step of administering the pharmaceutical composition to a subject.
일 양상은 제1 가닥; 및 상기 제1 가닥과 상보적으로 결합하는 제 2가닥으로 이루어지는, 25개 내지 60개 염기쌍 (bp)의 이중가닥 RNA로서, 상기 이중가닥 RNA 중 어느 하나의 가닥은 화학적 변형 (Chemical modification)이 도입된 뉴클레오티드를 적어도 10개 이상 포함하며, 상기 이중가닥 RNA는 MDA5 (Melanoma differentiation-associated protein 5) 매개 선천성 면역 반응을 증진시키는 것인, 이중가닥 RNA를 제공한다.One aspect is a double-stranded RNA having 25 to 60 base pairs (bp) comprising a first strand; and a second strand complementarily binding to the first strand, wherein one strand of the double-stranded RNA contains at least 10 nucleotides to which a chemical modification has been introduced, and wherein the double-stranded RNA enhances an MDA5 (Melanoma differentiation-associated protein 5)-mediated innate immune response.
다른 양상은 제1 가닥; 및 상기 제1 가닥과 상보적으로 결합하는 제2 가닥으로 이루어지는, 25개 내지 60개 염기쌍의 이중가닥 RNA로서, 상기 이중가닥 RNA는 10개 내지 30개 염기쌍의 선천성 면역 활성화 모티프를 포함하고, 상기 이중가닥 RNA 중 어느 하나의 가닥은 화학적 변형이 도입된 뉴클레오티드를 적어도 10개 이상 포함하는 이중가닥 RNA를 제공한다. Another aspect provides a double-stranded RNA of 25 to 60 base pairs, comprising a first strand; and a second strand complementarily binding to said first strand, said double-stranded RNA comprising an innate immune activation motif of 10 to 30 base pairs, wherein one strand of said double-stranded RNA comprises at least 10 nucleotides to which a chemical modification has been introduced.
또 다른 양상은 상기 이중가닥 RNA를 포함하는 선천성 면역 증진용 조성물을 제공한다.Another aspect provides a composition for enhancing innate immunity comprising the double-stranded RNA.
또 다른 양상은 상기 이중가닥 RNA를 유효성분으로 포함하는 약학적 조성물을 제공한다.Another aspect provides a pharmaceutical composition comprising the double-stranded RNA as an active ingredient.
또 다른 양상은 상기 이중가닥 RNA를 개체에 투여하는 단계를 포함하는 질병의 치료방법을 제공한다.Another aspect provides a method of treating a disease comprising administering to a subject the double-stranded RNA.
또 다른 양상은 질병 또는 병리적 상태에 대한 치료 또는 개선을 위한 상기 이중가닥 RNA의 의약적 용도를 제공한다.Another aspect provides a medicinal use of said double-stranded RNA for the treatment or amelioration of a disease or pathological condition.
일 양상에 따른 이중가닥 RNA는 선천성 면역 활성화 모티프(Innate Immune response Activating Motif) 및 화학적 변형이 도입된 뉴클레오티드를 포함함으로써, 높은 수준의 선천성 면역 반응을 유도할 수 있다. Double-stranded RNA according to one aspect can induce a high level of innate immune response by including an innate immune response Activating Motif and a chemically modified nucleotide.
또한, 일 양상에 따른 이중가닥 RNA는 선천성 면역 반응을 증진시킴으로써, 백신 제제 또는 면역 치료제의 효능을 향상시킬 수 있다. Additionally, double-stranded RNA according to one aspect may enhance the efficacy of vaccine formulations or immunotherapeutic agents by enhancing innate immune responses.
도 1은 일 양상에 따른 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA의 구조를 개략적으로 나타낸 도이다. Figure 1 is a schematic diagram showing the structure of a double-stranded RNA containing an innate immune activation motif according to one aspect.
도 2는 일 양상에 따른 서열번호 6의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 T98G 세포주에 형질감염시킨 후, IFIT1, CXCL10, 및 ISG15 의 mRNA 발현 수준을 확인한 결과이다. Figure 2 shows the results of confirming the mRNA expression levels of IFIT1, CXCL10, and ISG15 after transfecting a T98G cell line with a double-stranded RNA containing an innate immune activation sequence of sequence number 6 according to one aspect.
도 3은 일 양상에 따른 서열번호 6의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 T98G 세포주에 형질감염시킨 후, TNF-α 및 IL-1β의 mRNA 발현 수준을 확인한 결과이다.Figure 3 shows the results of confirming the mRNA expression levels of TNF-α and IL-1β after transfecting a T98G cell line with a double-stranded RNA containing an innate immune activation sequence of sequence number 6 according to one aspect.
도 4는 일 양상에 따른 서열번호 8의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 T98G 세포주에 형질감염시킨 후, IFIT1, CXCL10, 및 ISG15 의 mRNA 발현 수준을 확인한 결과이다. Figure 4 shows the results of confirming the mRNA expression levels of IFIT1, CXCL10, and ISG15 after transfecting a T98G cell line with a double-stranded RNA containing an innate immune activation sequence of
도 5는 일 양상에 따른 서열번호 6의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 야생형 세포주(WT) 또는 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다. Figure 5 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting a double-stranded RNA containing an innate immune activation sequence of sequence number 6 according to one aspect into a wild-type cell line (WT) or a cell line lacking RIG-I (RIG-I KO).
도 6은 일 양상에 따른 서열번호 8의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 야생형 세포주(WT) 또는 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 6 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting a double-stranded RNA containing an innate immune activation sequence of
도 7은 일 양상에 따른 서열번호 2 또는 서열번호 4의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 야생형 세포주(WT) 또는 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 7 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting a double-stranded RNA containing an innate immune activation sequence of
도 8은 일 양상에 따른 서열번호 2의 선천성 면역 활성화 서열을 포함하는 30bp의 이중가닥 RNA를 Huh7 세포주에 형질감염시킨 후, Lamin A/C 유전자의 mRNA 발현 수준을 확인한 결과이다. Figure 8 is according to the daily aspect. This is the result of confirming the mRNA expression level of the Lamin A/C gene after transfecting the Huh7 cell line with a 30-bp double-stranded RNA containing the innate immune activation sequence of
도 9는 일 양상에 따른 서열번호 2의 선천성 면역 활성화 서열을 포함하는 30bp의 이중가닥 RNA에서, 이중가닥 내 선천성 면역 활성화 서열이 제2 가닥의 5'말단 또는 제1 가닥의 3'말단에 위치하는 이중가닥 RNA 각각을 야생형 세포주(WT) 및 RIG-Ⅰ가 결여된 세포주(RIG-KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 9 is according to the daily aspect. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting each of the double-stranded RNAs containing the innate immune activation sequence of
도 10은 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥에 화학적 변형이 도입된, 일 양상에 따른 이중가닥 RNA를 각각 야생형 세포주(WT) 및 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 10 is a diagram showing an embodiment in which a chemical modification is introduced into either the first strand or the second strand. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
도 11은 제1 가닥 및 제2 가닥 모두에 화학적 변형이 도입된, 일 양상에 따른 이중가닥 RNA를 각각 야생형 세포주(WT) 및 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 11 is a diagram showing an embodiment of a method in which chemical modifications are introduced into both the first and second strands. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
도 12는 화학적 변형이 도입된 10개의 뉴클레오티드를 제1 가닥의 5'말단 또는 3' 말단으로 위치시키거나(#1 5'-1-10, #1 3'-1-10), 제1 가닥 내 화학적 변형이 도입된 첫번째 뉴클레오티드 위치를 변경시킨(#1 5'-1-10, #1 5'-2-10), 일 양상에 따른 이중가닥 RNA를 각각 야생형 세포주(WT) 및 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자의 활성 수준을 확인한 결과이다.Figure 12 shows an aspect in which 10 nucleotides into which chemical modifications are introduced are positioned at the 5' end or the 3' end of the first strand (#1 5'-1-10, #1 3'-1-10), or the position of the first nucleotide into which chemical modifications are introduced in the first strand is changed (#1 5'-1-10, #1 5'-2-10). This is the result of confirming the activity level of interferon regulatory factors after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
도 13은 8개, 10개, 12개 또는 15개의 뉴클레오티드에 화학적 변형이 도입된, 각각의 이중가닥 RNA를 각각 야생형 세포주(WT) 및 RIG-Ⅰ가 결여된 세포주(RIG-I KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 13 shows chemical modifications introduced into 8, 10, 12 or 15 nucleotides, respectively. This is the result of confirming the activity level of interferon regulatory factor (IRF) after transfecting double-stranded RNA into wild-type cell line (WT) and RIG-I deficient cell line (RIG-I KO), respectively.
도 14는 화학적 변형이 도입된 이중가닥 RNA를 각각 야생형 세포주(WT), RIG-Ⅰ가 결여된 세포주(RIG-I KO), 또는 MDA5가 결여된 세포주(MDA5 KO)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다.Figure 14 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting chemically modified double-stranded RNA into a wild-type cell line (WT), a cell line lacking RIG-I (RIG-I KO), or a cell line lacking MDA5 (MDA5 KO), respectively.
도 15는 화학적 변형이 도입된 이중가닥 RNA를 각각 인간 단핵구 세포주(THP-1 Dual)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 시간의 경과에 따라 확인한 결과이다.Figure 15 shows the results of examining the activity level of interferon regulatory factor (IRF) over time after transfecting a human monocyte cell line (THP-1 Dual) with double-stranded RNA with chemical modifications.
도 16은 선천성 면역 활성화 모티프가 제 2가닥의 5' 말단에 위치하고, 적어도 10개의 화학적 변형이 제1 가닥에 도입된, 20bp~40bp의 서로 다른 뉴클레오티드 길이를 갖는 이중가닥 RNA를 각각 RAW-Lucia ISG 세포주에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다. Figure 16 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell lines with double-stranded RNAs having different nucleotide lengths of 20 to 40 bp, in which the innate immune activation motif is located at the 5' end of the second strand and at least 10 chemical modifications are introduced into the first strand.
도 17은 일 실시예에 따른 30bp의 이중가닥 RNA(#1 5'-2-15)에서, 선천성 면역 활성화 모티프를 포함하거나 포함하지 않는 이중가닥 RNA를 각각 인간 단핵구 세포주(THP-1 Dual)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 시간의 경과에 따라 확인한 결과이다.Figure 17 shows the results of examining the activity level of interferon regulatory factor (IRF) over time after transfecting a human monocyte cell line (THP-1 Dual) with double-stranded RNA containing or not containing an innate immune activation motif in a 30bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
도 18은 일 실시예에 따른 30bp의 이중가닥 RNA(#1 5'-2-15)에서, 서열번호 2의 선천성 면역 활성화 서열, 서열번호 8(20nt)의 선천성 면역 활성화 서열, 또는 서열번호 62(30nt)의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA를 각각 RAW-Lucia ISG 세포주에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다. FIG. 18 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell line with a double-stranded RNA including an innate immune activation sequence of SEQ ID NO: 2, an innate immune activation sequence of SEQ ID NO: 8 (20 nt), or an innate immune activation sequence of SEQ ID NO: 62 (30 nt) in a 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
도 19는 일 실시예에 따른 30bp의 이중가닥 RNA(#1 5'-2-15)에서, 2' -OCH3(-O-methyl)의 화학적 변형 또는 2' -F의 화학적 변형을 포함하는 이중가닥 RNA를 각각 RAW-Lucia ISG 세포주에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다. FIG. 19 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell lines with double-stranded RNA containing chemical modification of 2' -OCH3(-O-methyl) or chemical modification of 2' -F in 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
도 20은 일 실시예에 따른 30bp의 이중가닥 RNA(#1 5'-2-15)에서, 2' -OCH3(-O-methyl)의 화학적 변형 또는 2' -F의 화학적 변형을 포함하는 이중가닥 RNA를 각각 인간 단핵구 세포주(THP-1 Dual)에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 시간의 경과에 따라 확인한 결과이다.FIG. 20 shows the results of examining the activity level of interferon regulatory factor (IRF) over time after transfecting a human monocyte cell line (THP-1 Dual) with a double-stranded RNA containing a chemical modification of 2' -OCH3(-O-methyl) or a chemical modification of 2' -F in a 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
도 21은 일 실시예에 따른 30bp의 이중가닥 RNA(#1 5'-2-15)에서, 제1 가닥, 제 2가닥, 또는 제1 가닥 및 제2 가닥 모두의 양 말단에서 인접한 2개의 뉴클레오티드 결합이 포스포로티오에이트(phosphorothioate) 결합으로 변형된 이중가닥 RNA를 각각 RAW-Lucia ISG 세포주에 형질감염시킨 후, 인터페론 조절 인자(IRF)의 활성 수준을 확인한 결과이다. FIG. 21 shows the results of confirming the activity level of interferon regulatory factor (IRF) after transfecting RAW-Lucia ISG cell line with double-stranded RNA in which two adjacent nucleotide bonds at both ends of the first strand, the second strand, or both the first strand and the second strand are modified with phosphorothioate bonds in a 30 bp double-stranded RNA (#1 5'-2-15) according to one embodiment.
본 출원에서 개시된 각각의 설명 및 실시형태는 각각의 다른 설명 및 실시 형태에도 적용될 수 있다. 즉, 본 출원에서 개시된 다양한 요소들의 모든 조합이 본 출원의 범주에 속한다. 또한, 하기 기술된 구체적인 서술에 의하여 본 출원의 범주가 제한된다고 볼 수 없다.Each description and embodiment disclosed in this application can also be applied to each other description and embodiment. That is, all combinations of various elements disclosed in this application fall within the scope of this application. In addition, the scope of this application cannot be considered limited by the specific description described below.
일 양상은 제1 가닥; 및 상기 제1 가닥과 상보적으로 결합하는 제2 가닥으로 이루어지는, 25개 내지 60개 염기쌍의 이중가닥 RNA로서, 상기 이중가닥 RNA는 10개 내지 30개 염기쌍의 선천성 면역 활성화 모티프를 포함하고, 상기 이중가닥 RNA 중 어느 하나의 가닥은 화학적 변형이 도입된 뉴클레오티드를 적어도 10개 이상 포함하는 이중가닥 RNA를 제공한다.One aspect provides a double-stranded RNA of 25 to 60 base pairs, comprising a first strand; and a second strand complementarily binding to the first strand, wherein the double-stranded RNA comprises an innate immune activation motif of 10 to 30 base pairs, wherein one strand of the double-stranded RNA comprises at least 10 nucleotides to which a chemical modification has been introduced.
이중가닥 RNAdouble-stranded RNA
본 명세서에서 용어, "이중가닥 RNA (double-stranded RNA)"는 서로 상보적으로 결합을 하는 두 가닥 (제1 가닥 및 제2 가닥)의 RNA를 의미한다. 상기 이중가닥 RNA는 사용 목적에 따라, RNA 기반의 면역 증강제/애주번트", "RNA 기반의 면역원성 조성물", "RNAi 유도용 핵산 분자"와 상호 교환적으로 사용될 수 있다. As used herein, the term "double-stranded RNA" means RNA having two strands (a first strand and a second strand) that complementarily bind to each other. The double-stranded RNA may be used interchangeably with "RNA-based immune enhancer/adjuvant", "RNA-based immunogenic composition", and "RNAi-inducing nucleic acid molecule", depending on the intended use.
본 명세서에 용어, "제1 가닥" 또는 "제2 가닥"은 상기 이중가닥 RNA가 RNA 간섭을 위한 영역을 포함하는 경우, 각각 안티센스 가닥 또는 센스 가닥으로 지칭될 수 있다. As used herein, the terms “first strand” or “second strand” may be referred to as the antisense strand or the sense strand, respectively, when the double-stranded RNA comprises a region for RNA interference.
상기 이중가닥 RNA는 25개 내지 60개의 염기쌍 길이를 갖는 것일 수 있으며, 예를 들어, 25개 내지 60개, 25개 내지 55개, 25개 내지 50개, 25개 내지 45개, 25개 내지 40개, 25개 내지 35개, 25개 내지 30개, 26 내지 60개, 26개 내지 55개, 26개 내지 50개, 26개 내지 45개, 26개 내지 40개, 26개 내지 35개, 26개 내지 30개, 30개 내지 60개, 30개 내지 55개, 30개 내지 50개, 30개 내지 45개, 30개 내지 40개, 30개 내지 35개, 35개 내지 60개, 35개 내지 55개, 35개 내지 50개, 35개 내지 45개, 또는 35개 내지 40개 염기쌍의 길이를 갖는 것일 수 있다. The double-stranded RNA may have a length of 25 to 60 base pairs, for example, 25 to 60, 25 to 55, 25 to 50, 25 to 45, 25 to 40, 25 to 35, 25 to 30, 26 to 60, 26 to 55, 26 to 50, 26 to 45, 26 to 40, 26 to 35, 26 to 30, 30 to 60, 30 to 55, 30 to 50, 30 to 45, 30 to 40, 30 to 35, 35 to 60, 35 to It may have a length of 55, 35 to 50, 35 to 45, or 35 to 40 base pairs.
본 명세서에서 용어, "상보성 (complementarity)" 또는 "상보적인 (complementary)"은 당업계에서 일반적으로 허용되는 의미를 지칭한다. 상기 용어는 일반적으로 전통적인 왓슨-크릭 (Watson-Crick) 또는 본 명세서에 기술된 다른 비-전통적 유형의 결합에 의해, 하나의 핵산 서열과 다른 핵산 서열 사이에서의 수소 결합 (들)의 형성 또는 존재를 지칭할 수 있다. 완벽한 상보성은 하나의 핵산 서열의 모든 인접 잔기가 다른 핵산 서열에서 동일한 수의 인접 잔기와 수소 결합한다는 것을 의미할 수 있다. 부분 상보성은 핵산 분자 내에서, 다양한 미스매치 (mismatch) 또는 비-염기쌍 뉴클레오티드 (non-based paired nucleotides) (예를 들어, 1, 2, 3, 4, 5, 6, 7, 8, 9, 또는 10 개 이상의 미스매치, 예를 들어, 1 내지 3개의 미스매치, 비-뉴클레오티드 링커 (non-nucleotide linker), 또는 비-염기쌍 뉴클레오티드)를 포함할 수 있다. 상기 부분 상보성은, 이중가닥 RNA의 제1 가닥과 제2 가닥 사이에서, 또는 이중가닥 RNA의 제1 가닥과 이와 상응하는 표적 핵산 분자 사이에서, 벌지 (bulges), 루프 (loops), 돌출 (overhang), 또는 평활 말단(blunt end)을 초래할 수 있다.As used herein, the terms "complementarity" or "complementary" refer to their generally accepted meanings in the art. The terms may generally refer to the formation or presence of hydrogen bond(s) between one nucleic acid sequence and another nucleic acid sequence, either by traditional Watson-Crick or other non-traditional types of binding as described herein. Perfect complementarity may mean that every adjacent residue in one nucleic acid sequence hydrogen bonds with the same number of adjacent residues in the other nucleic acid sequence. Partial complementarity can include various mismatches or non-based paired nucleotides within a nucleic acid molecule (e.g., more than 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 mismatches, e.g., 1 to 3 mismatches, non-nucleotide linkers, or non-base paired nucleotides). The partial complementarity can result in bulges, loops, overhangs, or blunt ends between the first and second strands of a double-stranded RNA, or between the first strand of a double-stranded RNA and a corresponding target nucleic acid molecule.
선천성 면역 활성화 모티프 Innate immune activation motif
본 명세서에서 용어, "선천성 면역 활성화 모티프(Innate Immune Response Activating Motif)"는 이중가닥 RNA가 투여된 개체에서 선천성 면역 반응을 유도 또는 증진시키기 위한 이중가닥 RNA 내 영역을 지칭하며, 선천성 면역 활성화 서열 및 상기 서열과 상보적인 결합을 형성하는 서열로 이루어진 이중가닥의 구조물을 지칭한다. The term "Innate Immune Response Activating Motif" as used herein refers to a region in a double-stranded RNA that induces or enhances an innate immune response in a subject to which the double-stranded RNA has been administered, and refers to a double-stranded structure composed of an innate immune activating sequence and a sequence that forms a complementary bond with the sequence.
본 명세서에서 용어, "선천성 면역 반응"은 이중가닥 RNA로부터 유도되는 생체 내 선천성 방어 면역 시스템으로서, 치료학적 관점에서 개체의 면역력을 향상시켜 치료의 효능을 향상시키는데 기여할 수 있는 생체 내 반응을 지칭할 수 있다. 상기 선천성 면역 반응은 예를 들어, 인터페론에 유도되거나, RIG-Ⅰ(Retinoic acid-inducible gene Ⅰ), 또는 MDA5 (Melanoma differentiation-associated gene 5)에 의해 매개되는 것일 수 있다.The term "innate immune response" as used herein may refer to an in vivo innate defense immune system induced from double-stranded RNA, which may contribute to improving the efficacy of treatment by enhancing the immunity of an individual from a therapeutic perspective. The innate immune response may be, for example, induced by interferon, or mediated by RIG-I (Retinoic acid-inducible gene I), or MDA5 (Melanoma differentiation-associated gene 5).
이중가닥 RNA에 의한 선천성 면역 반응은 이중가닥의 말단 영역 구조에 의존하는 것으로 알려져 있다. 따라서, 선천성 면역 반응을 증진시키기 위한 기술적 수단으로서, 5'-트리포스페이트를 도입하거나, 변형된 말단의 구조를 도입하는 접근법이 보고된 바 있다. 또한, MDA5 매개 면역 반응은 긴 길이의 이중가닥 RNA에 의해 야기되는 것으로 보고된 바 있다. 한편, 본 발명자들은 일 실시예에 따른 이중가닥 RNA가 상대적으로 짧은 길이를 가지고 있음에도, 선천성 면역 활성화 모티프 및 임의의 화학적 변형을 통해 선천성 면역 반응이 유도 또는 증진될 수 있음을 규명하였다. It is known that the innate immune response by double-stranded RNA depends on the terminal region structure of the double-strand. Therefore, as a technical means for enhancing the innate immune response, an approach of introducing 5'-triphosphate or introducing a modified terminal structure has been reported. In addition, it has been reported that the MDA5-mediated immune response is induced by long-length double-stranded RNA. Meanwhile, the present inventors have elucidated that the innate immune response can be induced or enhanced through the innate immune activation motif and any chemical modification even though the double-stranded RNA according to one embodiment has a relatively short length.
본 명세서에서 용어, "선천성 면역 활성화 서열"은 선천성 면역 반응을 활성화시키는 이중가닥 RNA 내 어느 하나의 가닥에 위치하는 뉴클레오티드 서열을 지칭한다. 예를 들어, 상기 선천성 면역 활성화 서열은 RIG-Ⅰ에 의해 매개되는 선천성 면역 반응을 활성화할 수 있는 구조를 포함할 수 있다. 구체적으로, 상기 선천성 면역 활성화 서열은 서열번호 2, 서열번호 4, 서열번호 6, 서열번호 8, 또는 서열번호 62의 뉴클레오티드 서열일 수 있다. 일 구체예에서, 상기 선천성 면역 활성화 서열은 서열번호 2, 서열번호 4, 서열번호 6, 서열번호 8, 또는 서열번호 62 중 어느 하나의 뉴클레오티드 서열과 적어도 80% 이상의 서열 동일성을 갖는 상기 뉴클레오티드 서열의 변이체를 포함하는 것일 수 있다. As used herein, the term "innate immune activation sequence" refers to a nucleotide sequence located on either strand of a double-stranded RNA that activates an innate immune response. For example, the innate immune activation sequence may comprise a structure capable of activating an innate immune response mediated by RIG-I. Specifically, the innate immune activation sequence may be a nucleotide sequence of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62. In one specific embodiment, the innate immune activation sequence may comprise a variant of the nucleotide sequence having at least 80% sequence identity with any one of the nucleotide sequences of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62.
일 구체예에서, 선천성 면역 활성화 모티프는 서열번호 2, 서열번호 4, 서열번호 6, 서열번호 8, 또는 서열번호 62의 뉴클레오티드 서열과, 상기 뉴클레오티드 서열과 상보적으로 결합하는 뉴클레오티드 서열로 이루어질 수 있다. 상기 선천성 면역 활성화 모티프는 이중가닥 RNA의 일측 말단 영역에 위치할 수 있으며, 예를 들어, 제1 가닥의 3' 말단 또는 제2 가닥의 5'말단에 위치하는 것일 수 있다. In one specific embodiment, the innate immune activation motif can be comprised of a nucleotide sequence of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62, and a nucleotide sequence complementarily binding to said nucleotide sequence. The innate immune activation motif can be located at one terminal region of the double-stranded RNA, for example, can be located at the 3' end of the first strand or the 5' end of the second strand.
상기 선천성 면역 활성화 모티프는 서열번호 2의 뉴클레오티드 서열을 포함하는 10개 내지 30개의 뉴클레오티드, 예를 들어, 10개 내지 30개, 10개 내지 28개, 10개 내지 26개, 10개 내지 24개, 10개 내지 22개, 10개 내지 20개, 10개 내지 18개, 10개 내지 16개, 10개 내지 14개, 10개 내지 12개, 12개 내지 30개, 12개 내지 28개, 12개 내지 26개, 12개 내지 24개, 12개 내지 22개, 12개 내지 20개, 12개 내지 18개, 12개 내지 16개, 12개 내지 14개, 14개 내지 30개, 14개 내지 28개, 14개 내지 26개, 14개 내지 24개, 14개 내지 22개, 14개 내지 20개, 14개 내지 18개, 또는 14개 내지 16개의 뉴클레오티드와 상기 서열에 상보적으로 결합하는 또 다른 뉴클레오티드 서열로 이루어질 수 있다. The innate immune activation motif comprises 10 to 30 nucleotides comprising the nucleotide sequence of SEQ ID NO: 2, for example, 10 to 30, 10 to 28, 10 to 26, 10 to 24, 10 to 22, 10 to 20, 10 to 18, 10 to 16, 10 to 14, 10 to 12, 12 to 30, 12 to 28, 12 to 26, 12 to 24, 12 to 22, 12 to 20, 12 to 18, 12 to 16, 12 to 14, 14 to 30, 14 to It may consist of 28, 14 to 26, 14 to 24, 14 to 22, 14 to 20, 14 to 18, or 14 to 16 nucleotides and another nucleotide sequence complementarily binding to said sequence.
상기 선천성 면역 활성화 모티프는 10개 내지 30개의 염기쌍 길이를 갖는 것일 수 있으며, 예를 들어, 10개 내지 30개, 10개 내지 28개, 10개 내지 26개, 10개 내지 24개, 10개 내지 22개, 10개 내지 20개, 10개 내지 18개, 10개 내지 16개, 10개 내지 14개, 10개 내지 12개, 12개 내지 30개, 12개 내지 28개, 12개 내지 26개, 12개 내지 24개, 12개 내지 22개, 12개 내지 20개, 12개 내지 18개, 12개 내지 16개, 12개 내지 14개, 14개 내지 30개, 14개 내지 28개, 14개 내지 26개, 14개 내지 24개, 14개 내지 22개, 14개 내지 20개, 14개 내지 18개, 14개 내지 16개, 16개 내지 30개, 16개 내지 28개, 16개 내지 26개, 16개 내지 24개, 16개 내지 22개, 16개 내지 20개, 16개 내지 18개, 18개 내지 30개, 또는 18개 내지 20개 염기쌍 길이를 갖는 것일 수 있다. The innate immune activation motif may have a length of 10 to 30 base pairs, for example, 10 to 30, 10 to 28, 10 to 26, 10 to 24, 10 to 22, 10 to 20, 10 to 18, 10 to 16, 10 to 14, 10 to 12, 12 to 30, 12 to 28, 12 to 26, 12 to 24, 12 to 22, 12 to 20, 12 to 18, 12 to 16, 12 to 14, 14 to 30, 14 to 28, It may have a length of 14 to 26, 14 to 24, 14 to 22, 14 to 20, 14 to 18, 14 to 16, 16 to 30, 16 to 28, 16 to 26, 16 to 24, 16 to 22, 16 to 20, 16 to 18, 18 to 30, or 18 to 20 base pairs.
본 명세서에서, "변이체 (variants)"는 참조 엔터티 (entity)(예컨대, 야생형 서열)와 상당한 구조적 동일성을 나타내지만 하나 이상에서 참조 엔터티와 구조적으로 상이한 엔터티를 지칭한다. 예를 들어, 폴리뉴클레오티드는 뉴클레오티드 서열의 하나 이상의 차이 및/또는 폴리뉴클레오티드 백본에 공유 결합된 화학적 모이어티 (예를 들어, 탄수화물, 지질 등)의 하나 이상의 차이로 인해 참조 폴리뉴클레오티드와 상이할 수 있다. 상기 변이체는 적어도 60%, 65%, 70%, 75%, 80%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98% 또는 99%의, 참조 폴리뉴클레오티드와의 전체 서열 동일성을 나타낸다. As used herein, "variants" refer to entities that exhibit substantial structural identity with a reference entity (e.g., a wild type sequence) but are structurally different from the reference entity in one or more respects. For example, a polynucleotide may differ from a reference polynucleotide by one or more differences in the nucleotide sequence and/or by one or more differences in the chemical moieties (e.g., carbohydrates, lipids, etc.) covalently linked to the polynucleotide backbone. The variants exhibit at least 60%, 65%, 70%, 75%, 80%, 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% overall sequence identity with the reference polynucleotide.
본 명세서에서 용어, "동일성"은 폴리머 분자들 간의, 예를 들어, 핵산(예를 들어, DNA 분자들 및/또는 RNA 분자들) 간의 전체 관련성을 지칭한다. 예를 들어, 폴리뉴클레오티드 서열은 이들의 서열이 적어도, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 또는 99% 동일하다면 서로 "실질적으로 동일한" 것으로 간주된다. 두 폴리뉴클레오티드 서열의 동일성 퍼센트의 계산은, 예를 들어, 최적의 비교 목적을 위해 두 서열을 정렬함으로써 수행될 수 있다(예를 들어, 최적의 정렬을 위해 제1 및 제2 서열 중 하나 또는 둘 모두에 갭이 도입될 수 있고, 비교 목적을 위해 비-동일 서열이 무시될 수 있다). 두 서열 간의 동일성 퍼센트의 결정 및 서열의 비교는 수학적 알고리즘을 이용하여 달성될 수 있다. 당업자에게 널리 공지된 바와 같이, 뉴클레오티드 서열의 경우 BLASTN와 같이 상업적 컴퓨터 프로그램에서 입수 가능한 것들을 포함하는 임의의 다양한 알고리즘을 이용하여 비교될 수 있다.As used herein, the term "identity" refers to the overall relatedness between polymer molecules, for example, between nucleic acids (e.g., DNA molecules and/or RNA molecules). For example, polynucleotide sequences are considered to be "substantially identical" to one another if their sequences are at least 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, or 99% identical. Calculating the percent identity of two polynucleotide sequences can be performed, for example, by aligning the two sequences for optimal comparison purposes (e.g., gaps can be introduced into one or both of the first and second sequences for optimal alignment, and non-identical sequences can be ignored for comparison purposes). Determining the percent identity between two sequences and comparing the sequences can be accomplished using mathematical algorithms. As is well known to those skilled in the art, nucleotide sequences may be compared using any of a variety of algorithms, including those available in commercial computer programs such as BLASTN.
일 실시예에 따르면, 상기 선천성 면역 활성화 모티프는 이중가닥 RNA의 일측 말단 영역에 도입되어, 선천성 면역 활성화 모티프 외 다른 영역과 독립적으로 작동될 수 있다.In one embodiment, the innate immune activation motif is introduced into one terminal region of the double-stranded RNA, so that it can operate independently of other regions other than the innate immune activation motif.
화학적인 변형이 도입된 이중가닥 RNADouble-stranded RNA with chemical modifications
본 명세서의 이중가닥 RNA에서, 상기 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥은 화학적 변형이 도입된 뉴클레오티드를 포함하는 것일 수 있다. In the double-stranded RNA of the present specification, either the first strand or the second strand may include a nucleotide into which a chemical modification has been introduced.
일 구체예에서, 상기 화학적 변형이 도입된 뉴클레오티드는 다음으로 구성된 군에서 선택된 하나 이상의 화학적 변형을 포함할 수 있다: 뉴클레오티드 내 당 구조의 2' 탄소 위치에서 -OH기가 -H, -CH3(methyl), -OCH3(-O-methyl), -NH2, -F, -O-2-메톡시에틸-O-프로필(-O-2-methoxyethyl-O-propyl), -O-2-메틸티오에틸(methylthioethyl), -O-3-아미노프로필(O-3-aminopropyl), 또는 -O-3-디메틸아미노프로필(O-3-dimethylaminopropyl)로 치환.In one specific embodiment, the nucleotide into which the chemical modification is introduced may comprise one or more chemical modifications selected from the group consisting of: replacement of the -OH group at the 2' carbon position of the sugar structure in the nucleotide with -H, -CH 3 (methyl), -OCH 3 (-O-methyl), -NH 2 , -F, -O-2-methoxyethyl-O-propyl, -O-2-methylthioethyl, -O-3-aminopropyl, or -O-3-dimethylaminopropyl.
일 구체예에서, 상기 화학적 변형이 도입된 뉴클레오티드는 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥에 포함되는 것일 수 있고, 상기 화학적 변형이 도입된 뉴클레오티드는 적어도 10개 이상일 수 있다. 상기 화학적 변형이 도입된 뉴클레오티드의 개수는 예를 들어, 단일 가닥 내 10개 내지 60개, 10개 내지 55개, 10개 내지 50개, 10개 내지 45개, 10개 내지 40개, 10개 내지 35개, 10개 내지 30개, 10개 내지 25개, 10개 내지 20개, 10개 내지 15개, 15개 내지 60개, 15개 내지 55개, 15개 내지 50개, 15개 내지 45개, 15개 내지 40개, 15개 내지 35개, 15개 내지 30개, 15개 내지 25개, 15개 내지 20개, 20개 내지 60개, 20개 내지 55개, 20개 내지 50개, 20개 내지 45개, 20개 내지 40개, 20개 내지 35개, 20개 내지 30개, 20개 내지 25개, 또는 15개 내지 20개일 수 있다. In one specific embodiment, the nucleotides to which the chemical modification has been introduced may be included in either the first strand or the second strand, and the number of nucleotides to which the chemical modification has been introduced may be at least 10 or more. The number of nucleotides into which the chemical modification is introduced is, for example, 10 to 60, 10 to 55, 10 to 50, 10 to 45, 10 to 40, 10 to 35, 10 to 30, 10 to 25, 10 to 20, 10 to 15, 15 to 60, 15 to 55, 15 to 50, 15 to 45, 15 to 40, 15 to 35, 15 to 30, 15 to 25, 15 to 20, 20 to 60, 20 to 55, 20 to 50, 20 to It can be 45, 20 to 40, 20 to 35, 20 to 30, 20 to 25, or 15 to 20.
일 구체예에서, 상기 화학적 변형이 도입된 뉴클레오티드는 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥 내에서 연속적으로 또는 불연속적으로 배열되는 것일 수 있다. In one specific embodiment, the nucleotides into which the chemical modification has been introduced may be arranged either continuously or discontinuously within either the first strand or the second strand.
일 구체예에서, 상기 화학적 변형이 도입된 뉴클레오티드는 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥에 포함되는 것일 수 있고, 선천성 면역 활성화 모티프 내 영역 및/또는 선천 면역 활성화 모티프 외 영역에 위치하는 것일 수 있다. In one specific embodiment, the nucleotide into which the chemical modification is introduced may be included in either the first strand or the second strand and may be located in a region within the innate immune activation motif and/or a region outside the innate immune activation motif.
일 구체예에서, 상기 화학적 변형이 도입된 뉴클레오티드는 제1 가닥에 적어도 10개 이상 포함되는 것으로서, 화학적 변형이 도입되지 않은 뉴클레오티드와 교대로(alternatively) 배열되는 것일 수 있으며, 예를 들어, 2' 탄소 위치에서 -OH기가 -OCH3 (-O-methyl)로 치환된 화학적 변형이 도입된 뉴클레오티드와 화학적 변형이 도입되지 않은 뉴클레오티드가 교대로 배열되는 것일 수 있다. 이 경우, 제1 가닥의 전체 뉴클레오티드에 대하여 50% 이하, 예를 들어, 50%, 45%, 40%, 35%, 또는 30%의 뉴클레오티드가 상기 화학적 변형이 도입된 뉴클레오티드일 수 있다.In one specific embodiment, the nucleotides to which the chemical modification has been introduced are included in at least 10 nucleotides in the first strand, and may be arranged alternatively with nucleotides to which the chemical modification has not been introduced. For example, nucleotides to which the chemical modification has been introduced in which the -OH group at the 2' carbon position is substituted with -OCH 3 (-O-methyl) and nucleotides to which the chemical modification has not been introduced may be arranged alternatively. In this case, 50% or less, for example, 50%, 45%, 40%, 35%, or 30% of the total nucleotides in the first strand may be nucleotides to which the chemical modification has been introduced.
일 구체예에서, 상기 화학적 변형이 도입된 뉴클레오티드는 제2 가닥에 적어도 10개 이상 포함되는 것으로서, 화학적 변형이 도입되지 않은 뉴클레오티드와 교대로 배열되는 것일 수 있으며, 예를 들어, 2' 탄소 위치에서 -OH기가 -OCH3 (-O-methyl)로 치환된 화학적 변형이 도입된 뉴클레오티드와 화학적 변형이 도입되지 않은 뉴클레오티드가 교대로 배열되는 것일 수 있다. 이 경우, 제2 가닥의 전체 뉴클레오티드에 대하여 50% 이하, 예를 들어, 50%, 45%, 40%, 35%, 또는 30%의 뉴클레오티드가 상기 화학적 변형이 도입된 뉴클레오티드일 수 있다.In one specific embodiment, the nucleotides to which the chemical modification has been introduced are included in the second strand at least 10 times, and may be arranged alternately with nucleotides to which the chemical modification has not been introduced, for example, nucleotides to which the chemical modification has been introduced in which the -OH group at the 2' carbon position is substituted with -OCH 3 (-O-methyl) and nucleotides to which the chemical modification has not been introduced may be arranged alternately. In this case, 50% or less, for example, 50%, 45%, 40%, 35%, or 30% of the total nucleotides of the second strand may be nucleotides to which the chemical modification has been introduced.
상기 화학적 변형이 도입된 뉴클레오티드는 선천성 면역 반응을 유도 또는 증진할 수 있다. 구체적으로, 상기 화학적 변형이 도입된 뉴클레오티드는 MDA5 매개 선천성 면역 반응을 유도 또는 증진시킬 수 있다. 또한, 상기 화학적 변형이 도입된 뉴클레오티드를 포함하는 본 발명에 따른 RNA 이중가닥은 RIG-Ⅰ 매개 선천성 면역 반응 및/또는 MDA5 매개 선천성 면역 반응을 유도 또는 증진시키기 위한 것일 수 있다. The nucleotide to which the chemical modification has been introduced can induce or enhance an innate immune response. Specifically, the nucleotide to which the chemical modification has been introduced can induce or enhance an MDA5-mediated innate immune response. In addition, the RNA duplex according to the present invention comprising the nucleotide to which the chemical modification has been introduced can be for inducing or enhancing an RIG-I-mediated innate immune response and/or an MDA5-mediated innate immune response.
본 명세서에서 용어,"MDA5 (Melanoma differentiation-associated gene 5)"는 RIG-Ⅰ 유사 수용체로, 외부 침입체를 인식하기 위한 패턴 인식 수용체 단백질로 기능하는 세포질 RNA 헬리카제이다. RIG-Ⅰ와 MDA5의 구조는 유사하여 N-텐덤 아미노-말단 카스파제 활성 및 모집 도메인 (tandem amino-terminal caspase activation and recruitment domains: CARDs) 구조 및 C- 말단 도메인(CTD)를 공유하며, CTD가 이중가닥 RNA를 인식하면 CARD가 방출되고, 이후의 연속된 신호는 인터페론 조절 인자(IRF)를 활성화하는 것으로 알려져 있다. 상기 MDA5 및 RIG-Ⅰ는 외부 RNA, 즉, 이중가닥 RNA에 부착되어 신호전달의 개시가 이루어지나, 짧은 dsRNA와의 부착을 선호하는 RIG-Ⅰ와 달리, MDA5는 상대적으로 긴 길이의 dsRNA에 의해 신호전달의 개시가 이루어지는 것을 알려져 있다. As used herein, the term "MDA5 (Melanoma differentiation-associated gene 5)" is a RIG-I-like receptor, a cytoplasmic RNA helicase that functions as a pattern recognition receptor protein to recognize foreign invaders. The structures of RIG-I and MDA5 are similar and share the N-tandem amino-terminal caspase activation and recruitment domains (CARDs) structure and the C-terminal domain (CTD). It is known that when the CTD recognizes double-stranded RNA, the CARDs are released, and the subsequent continuous signal activates interferon regulatory factors (IRFs). The above MDA5 and RIG-I bind to foreign RNA, i.e., double-stranded RNA, and initiate signal transduction. However, unlike RIG-I, which prefers attachment to short dsRNA, it is known that MDA5 initiates signal transduction by relatively long dsRNA.
일 실시예에 따르면, 전술한 이중가닥 RNA의 구조를 기본 골격으로 하여 화학적 변형이 추가로 도입된 이중가닥 RNA는 MDA5가 주요 인자로서 작용하는 추가적인 신호전달 기작을 통해, 상대적으로 짧은 염기상 길이를 갖는 이중가닥 RNA의 처리도 불구하고, 강력한 선천성 면역 반응의 유도를 가능하게 할 수 있다. In one embodiment, a double-stranded RNA having the structure of the aforementioned double-stranded RNA as its basic backbone and additionally having chemical modifications can induce a strong innate immune response despite the processing of double-stranded RNA having a relatively short base length, through an additional signaling mechanism in which MDA5 acts as a key factor.
일 구체예에서, 상기 이중가닥 RNA는 적어도 하나의 뉴클레오티드 간의 결합이 포스포로티오에이트(phosphorothioate), 보라노포페이트(boranophosphate), 또는 메틸포스포네이트(methyl phosphonate)로 변형된 것일 수 있다. 구체적으로, 상기 이중가닥 RNA는, 제1 가닥 및/또는 제2가닥의 5'말단 및/또는 3'말단으로부터 인접한 적어도 하나, 예를 들어, 2개 내지 4개의 뉴클레오티드 간 결합이 포스포로티오에이트(phosphorothioate), 보라노포페이트(boranophosphate), 또는 메틸포스포네이트(methyl phosphonate)로 변형된 것일 수 있다.In one specific embodiment, the double-stranded RNA may be one in which at least one inter-nucleotide bond is modified with phosphorothioate, boranophosphate, or methyl phosphonate. Specifically, the double-stranded RNA may be one in which at least one, for example, 2 to 4, inter-nucleotide bonds adjacent to the 5'-end and/or the 3'-end of the first strand and/or the second strand are modified with phosphorothioate, boranophosphate, or methyl phosphonate.
일 실시예에 따르면, 전술한 화학적 변형이 도입된 이중가닥 RNA의 구조를 기본 골격으로 하여 뉴클레오티드 간의 결합이 추가로 변형된 이중가닥 RNA는 그러한 변형에도 불구하고, 강력한 선천성 면역 반응의 유도를 가능하게 할 수 있다.In one embodiment, a double-stranded RNA having the structure of a double-stranded RNA with the chemical modifications described above as its basic backbone and further modified in the bonds between nucleotides can enable the induction of a strong innate immune response despite such modifications.
선천성 면역 반응 유도 효과를 갖는 이중가닥 RNADouble-stranded RNA with innate immune response induction effect
본 명세서에서, 이중가닥 RNA는 선천성 면역 반응을 유도 또는 증진시키는 것일 수 있다. 이를 위하여, 상기 이중가닥 RNA는 다음과 같은 조건을 만족하는 것일 수 있다: 1) 25 내지 60개 염기쌍의 길이를 갖을 것, 2) 상기 선천성 면역 활성화 모티프가 이중가닥 RNA중 한 가닥의 일측 말단 영역에 위치할 것, 3) 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥은 화학적 변형이 도입된 뉴클레오티드를 적어도 10개 이상을 포함할 것.In the present specification, the double-stranded RNA may be one that induces or enhances an innate immune response. To this end, the double-stranded RNA may satisfy the following conditions: 1) having a length of 25 to 60 base pairs, 2) the innate immune activation motif is located at one terminal region of one strand of the double-stranded RNA, 3) either the first strand or the second strand includes at least 10 nucleotides to which a chemical modification has been introduced.
일 구체예에서, 상기 이중가닥 RNA는 추가적으로, 상기 이중가닥 RNA의 일측 말단 또는 양 말단은 평활 말단(blunt end)일 수 있다. 일 구체예에서, 상기 이중가닥 RNA의 일측 말단은 돌출부 (Overhang)일 수 있다. 일 구체예에서, 상기 이중가닥 RNA는 상기 제1 가닥의 3'말단과 이에 상보적인 상기 제2 가닥의 5'말단에 선천성 면역 활성화 모티프가 위치하고, 평활 말단을 형성하는 것일 수 있다. 예를 들어, 상기 이중가닥 RNA에서, 선천성 면역 활성화 모티프가 위치하는 일측 말단은 평활 말단, 선천성 면역 활성화 모티프가 위치하지 않은 다른 말단은 돌출부를 갖는 것일 수 있다. 예를 들어, 상기 이중가닥 RNA에서, 선천성 면역 활성화 모티프가 위치하는 일측 말단 및 선천성 면역 활성화 모티프가 위치하지 않은 다른 말단 모두 평활 말단일 수 있다. In one specific embodiment, the double-stranded RNA may additionally have one or both ends of the double-stranded RNA be blunt ends. In one specific embodiment, one end of the double-stranded RNA may be an overhang. In one specific embodiment, the double-stranded RNA may have an innate immune activation motif positioned at the 3' end of the first strand and the 5' end of the second strand complementary thereto, forming a blunt end. For example, in the double-stranded RNA, one end at which the innate immune activation motif is positioned may be a blunt end, and the other end where the innate immune activation motif is not positioned may have an overhang. For example, in the double-stranded RNA, both one end at which the innate immune activation motif is positioned and the other end where the innate immune activation motif is not positioned may be blunt ends.
일 구체예에서, 상기 이중가닥 RNA는 25개 내지 60개 염기쌍의 길이를 갖는 이중가닥 RNA로서, RIG-Ⅰ및 MDA-5 매개의 선천성 면역 반응을 유도하는 특성을 갖는 것일 수 있고, 이를 위하여, 상기 이중가닥 RNA는 선천성 면역 활성화 모티프를 포함하고, 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥은 화학적 변형이 도입된 뉴클레오티드를 적어도 10개 이상을 포함하는 구조를 갖는 것일 수 있다. 상기 이중가닥 RNA는 25개 내지 60개 염기쌍의 길이를 갖는 이중가닥 RNA로서, 특정 유전자의 발현을 저해하는 RNA 간섭을 유도하는 특성을 갖는 것일 수 있으며, 이를 위하여 상기 이중가닥 RNA는 말단 영역에 추가적으로 특정 유전자와 상보적으로 결합하는 영역을 추가로 포함할 수 있다. In one specific example, the double-stranded RNA may be a double-stranded RNA having a length of 25 to 60 base pairs, and may have a property of inducing an innate immune response mediated by RIG-I and MDA-5, and for this purpose, the double-stranded RNA may include an innate immune activation motif, and either the first strand or the second strand may have a structure including at least 10 nucleotides to which a chemical modification has been introduced. The double-stranded RNA may be a double-stranded RNA having a length of 25 to 60 base pairs, and may have a property of inducing RNA interference that inhibits the expression of a specific gene, and for this purpose, the double-stranded RNA may additionally include a region that complementarily binds to a specific gene in a terminal region.
일 구체예에서, 상기 이중가닥 RNA는 상기 제1 가닥의 3' 말단과 이에 상보적인 상기 제2 가닥의 5' 말단 영역에 선천성 면역 활성화 서열이 위치하는 것일 수 있다. 상기 선천성 면역 활성화 서열은 서열번호 2의 뉴클레오티드 서열을 포함하는 10개 내지 30개의 뉴클레오티드일 수 있고, 예를 들어, 상기 선천성 면역 활성화 서열은 서열번호 2, 서열번호 4, 서열번호 6, 서열번호 8, 또는 서열번호 62 중 어느 하나의 서열로 이루어지는 뉴클레오티드 또는 상기 뉴클레오티드의 변이체를 지칭할 수 있다. 상기 이중가닥 RNA는 전술한 선천성 면역 활성화 모티프 및 이중가닥 RNA의 구조 (예를 들어, 화학적 변형, 말단 형태)를 포함함으로써, 개체의 선천성 면역 반응, 구체적으로, RIG-Ⅰ 매개 선천성 면역 반응 및/또는 MDA5 매개 선천성 면역 반응을 증진시키는데 기여할 수 있다.In one specific example, the double-stranded RNA may have an innate immune activation sequence located at the 3' end of the first strand and the 5' end region of the second strand complementary thereto. The innate immune activation sequence may be 10 to 30 nucleotides including the nucleotide sequence of SEQ ID NO: 2, for example, the innate immune activation sequence may refer to a nucleotide consisting of any one of SEQ ID NO: 2, SEQ ID NO: 4, SEQ ID NO: 6, SEQ ID NO: 8, or SEQ ID NO: 62, or a variant of the nucleotide. The double-stranded RNA may contribute to enhancing an innate immune response of an individual, specifically, a RIG-I-mediated innate immune response and/or an MDA5-mediated innate immune response, by including the above-mentioned innate immune activation motif and the structure of the double-stranded RNA (e.g., chemical modification, terminal form).
일 실시예에 따르면, 일 실시예에 따른 이중가닥 RNA는 일측 말단에 위치하는 RIG-Ⅰ매개 선천성 면역 활성화 모티프 및 화학적 변형이 도입된 뉴클레오티드를 적어도 10개 이상을 포함하며, 전술한 구조를 갖는 이중가닥 RNA는 선천성 면역 활성 모티프외 영역의 서열 및 선천성 면역 활성 모티프가 위치하는 가닥의 종류 및 이중 가닥 내 위치하는 영역에 구애받지 않고, 높은 수준의 선천성 면역 반응을 유도할 수 있었다. According to one embodiment, the double-stranded RNA according to one embodiment comprises a RIG-I-mediated innate immune activation motif located at one end and at least 10 nucleotides into which a chemical modification has been introduced, and the double-stranded RNA having the structure described above can induce a high level of innate immune response, regardless of the sequence of a region outside the innate immune activation motif, the type of strand on which the innate immune activation motif is located, and the region located within the double strand.
약학적 조성물 Pharmaceutical composition
본 명세서에서 용어, "유효성분 (effective ingredient)"은 이롭거나 바람직한 임상적 또는 생화학적 결과에 영향을 주는 적절한 유효량의 성분을 의미한다. 구체적으로는, 유효량의 제제, 활성제, 또는 이중가닥 RNA를 의미할 수 있다. 상기 약학적 조성물은 약제학적으로 허용 가능한 담체, 희석제, 부형제, 완충제, 염, 계면활성제, 저온보호물질 등을 선택적으로 포함할 수 있다. As used herein, the term "effective ingredient" means an appropriate effective amount of an ingredient that affects a beneficial or desirable clinical or biochemical result. Specifically, it may mean an effective amount of a preparation, an active agent, or a double-stranded RNA. The pharmaceutical composition may optionally include a pharmaceutically acceptable carrier, diluent, excipient, buffer, salt, surfactant, cryoprotectant, etc.
상기 유효량은 한번 또는 그 이상 투여될 수 있고, 질병을 예방하거나, 질병 상태를 비제한적으로, 증상의 완화, 질병 범위의 감소, 질병 상태의 안정화 (즉, 악화되지 않음), 질병 진행의 지연 또는 속도의 감소, 또는 질병 상태의 개선 또는 일시적 완화 및 경감 (부분적이거나 전체적으로)을 위한 적절한 양일 수 있다.The effective amount may be administered once or more and may be an amount appropriate for preventing a disease, or for alleviating symptoms, reducing the extent of a disease, stabilizing (i.e. not worsening) a disease, delaying or reducing the rate of disease progression, or improving or temporarily alleviating and alleviating (partially or completely) a disease.
본 명세서에서 용어, "예방 (prevention)"은 질환의 발생을 미리 차단하거나, 질환을 억제하거나 진행을 지연시키는 모든 행위를 의미한다. 예를 들어, 상기 질환 또는 이의 특징적인 특성의 발생을 막거나, 발생을 방해하거나, 상기 질환 또는 이의 특징적인 특성의 발생으로부터 방어 또는 보호하는 것을 지칭한다. The term "prevention" as used herein refers to any action that prevents the occurrence of a disease in advance, suppresses a disease, or delays its progression. For example, it refers to preventing the occurrence of the disease or its characteristic characteristics, interfering with the occurrence, or protecting or protecting against the occurrence of the disease or its characteristic characteristics.
본 명세서에서 용어, "치료 (treatment)"는 치료학적 치료 및 예방적 또는 예방조치 방법 모두를 의미한다. 또한, 질환의 증상이 호전 또는 이롭게 변경되는 모든 행위를 의미한다. 예를 들어, 상기 질환 또는 이의 특징적인 특성을 예방, 감소 또는 개선하거나, 또는 개체에서 상기 질환 또는 이의 특징적인 특성의 진행을 지연 (약화)시키는 것이다. The term "treatment" as used herein refers to both therapeutic treatment and preventive or prophylactic measures. It also refers to any action that improves or beneficially alters the symptoms of a disease. For example, preventing, reducing or ameliorating the disease or its characteristic features, or delaying (attenuating) the progression of the disease or its characteristic features in a subject.
상기 질환 또는 병리적 상태는 바이러스 또는 세균에 의한 감염증, 면역 저하와 연관된 질환, 암 (예를 들어, 유방암, 간암, 식도암, 췌장암, 폐암, 위암, 두경부 편평 세포 암종, 전립선암, 결장암, 림프종, 담낭암, 신장암, 다발성 골수종, 난소암, 자궁경부암 또는 신경교종과 같은 고형 종양, 또는 백혈병 등과 같은 비고형 종양)일 수 있다. The disease or pathological condition may be an infection caused by a virus or bacteria, a disease associated with immunosuppression, cancer (e.g., a solid tumor such as breast cancer, liver cancer, esophageal cancer, pancreatic cancer, lung cancer, stomach cancer, squamous cell carcinoma of the head and neck, prostate cancer, colon cancer, lymphoma, gallbladder cancer, kidney cancer, multiple myeloma, ovarian cancer, cervical cancer or glioma, or a non-solid tumor such as leukemia).
본 명세서에서 용어, "유효량 (effective amount)"은 당업계에서 일반적으로 허용되는 의미를 지칭한다. 상기 용어는 일반적으로 연구자, 수의사, 의사, 또는 기타 임상의사 등이 추구하는 세포, 조직, 시스템, 동물, 또는 인간의 의도된 생물학적 반응 (예: 유익한 반응)을 이끌어낼 분자, 화합물, 또는 구성물의 양을 의미할 수 있다. 구체적으로, "치료학적 유효량 (therapeutically effective amount)"은 예컨대, 질병이나 장애와 관련된 측정 가능한 파라미터에 있어서 치료적으로 관련된 변화가 있어 특정 임상적 치료가 효과적이라고 간주될 수 있는 정도의 바람직한 의학적 반응을 이끌어낼 수 있는 분자, 화합물, 또는 구성물의 양을 의미할 수 있다. 상기 질병 또는 장애의 치료를 위한 약물의 치료학적 유효량은 상기 파라미터에서 치료적으로 관련된 변화를 초래하는데 필요한 양일 수 있다. As used herein, the term "effective amount" refers to its generally accepted meaning in the art. The term can generally mean an amount of a molecule, compound, or composition that will elicit a desired biological response (e.g., a beneficial response) in a cell, tissue, system, animal, or human, as sought by a researcher, veterinarian, physician, or other clinician. Specifically, a "therapeutically effective amount" can mean an amount of a molecule, compound, or composition that will elicit a desired medical response, such as a therapeutically relevant change in a measurable parameter associated with a disease or disorder, such that a particular clinical treatment can be considered efficacious. A therapeutically effective amount of a drug for treating the disease or disorder can be the amount necessary to effect a therapeutically relevant change in the parameter.
본 명세서에서 용어, "약학적으로 허용가능한 부형제"는 이중가닥 RNA와 결합/혼합되는 경우 이중가닥 RNA의 활성을 유지하는 것일 수 있다. 일례로서, 인산염 완충 식염수와 같은 버퍼 시스템, 계면활성제, 물, 오일/물 에멀젼과 같은 에멀젼 및 다양한 형태의 습윤제, 전분, 우유, 당, 특정 형태의 클레이, 젤라틴, 스테아르산 또는 그 염, 마그네슘 또는 칼슘 스테아레이트, 탈크, 식물성 유지, 검, 글리콜, 또는 다른 공지된 부형제와 같은 임의의 표준 약학 부형제를 포함하나, 이에 제한되는 것은 아니다.The term "pharmaceutically acceptable excipient" as used herein may be one which, when combined/mixed with double-stranded RNA, maintains the activity of the double-stranded RNA. Examples thereof include, but are not limited to, any standard pharmaceutical excipient, such as a buffer system such as phosphate buffered saline, a surfactant, water, emulsions such as oil/water emulsions and various forms of wetting agents, starches, milk, sugars, certain forms of clay, gelatin, stearic acid or its salts, magnesium or calcium stearate, talc, vegetable oils, gums, glycols, or other known excipients.
상기 약학적 조성물의 투여방법은 통상의 환자의 증후와 질병의 심각도에 기초하여 본 기술분야의 통상의 전문가가 결정할 수 있다. 또한, 산제, 정제, 캡슐제, 액제, 주사제, 연고제, 시럽제 등의 다양한 형태로 제제화 할 수 있으며 단위-투여량 또는 다-투여량 용기, 예를 들면 밀봉된 앰플 및 병 등으로 제공될 수도 있다.The method of administration of the above pharmaceutical composition can be determined by a person skilled in the art based on the symptoms of the patient and the severity of the disease. In addition, it can be formulated in various forms such as powder, tablet, capsule, liquid, injection, ointment, syrup, etc., and can be provided in unit-dose or multi-dose containers, such as sealed ampoules and bottles.
본 발명의 약학적 조성물은 경구 또는 비경구 투여가 가능하다. 본 발명에 따른 조성물의 투여경로는 이들로 한정되는 것은 아니지만, 예를 들면, 구강, 정맥 내, 근육 내, 동맥 내, 골수 내, 경막 내, 심장 내, 경피, 피하, 복강 내, 장관, 설하 또는 국소 투여가 가능하다. 본 발명에 따른 조성물의 투여량은 환자의 체중, 연령, 성별, 건강상태, 식이, 투여시간, 방법, 배설율 또는 질병의 중증도 등에 따라 그 범위가 다양하며, 본 기술분야의 통상의 전문가가 용이하게 결정할 수 있다. 또한, 임상 투여를 위해 공지의 기술을 이용하여 본 발명의 조성물을 적합한 제형으로 제제화할 수 있다.The pharmaceutical composition of the present invention can be administered orally or parenterally. The route of administration of the composition according to the present invention is not limited thereto, but for example, oral, intravenous, intramuscular, intraarterial, intramedullary, intrathecal, intracardiac, transdermal, subcutaneous, intraperitoneal, enteral, sublingual, or topical administration is possible. The dosage of the composition according to the present invention varies depending on the patient's weight, age, sex, health condition, diet, administration time, method, excretion rate, or disease severity, and can be easily determined by a person skilled in the art. In addition, the composition of the present invention can be formulated into a suitable dosage form using a known technique for clinical administration.
선천성 면역 증진용 조성물Composition for enhancing innate immunity
본 명세서에서 용어, "선천성 면역 증진용 조성물"은 전술한 이중가닥 RNA를 포함하고, 투여된 개체의 선천성 면역 반응을 유도 또는 증진시키기 위한 생물학적 제제를 지칭한다. 상기 선천성 면역 증진용 조성물은 면역 증강제/애주번트, RNA 기반의 면역원성 조성물, 또는 면역 자극 제제 (immunostimulation agent)와 상호 교환적으로 사용될 수 있다. 상기 조성물은 약제학적으로 허용 가능한 담체, 희석제, 부형제, 완충제, 염, 계면활성제, 저온보호물질 등을 선택적으로 포함할 수 있으며, 실시 조건 및 실시 태양은 상기 약학적 조성물에 기재한 바와 같다. As used herein, the term "composition for enhancing innate immunity" refers to a biological agent containing the above-mentioned double-stranded RNA and for inducing or enhancing the innate immune response of an administered subject. The composition for enhancing innate immunity can be used interchangeably with an immune enhancer/adjuvant, an RNA-based immunogenic composition, or an immunostimulation agent. The composition can optionally contain a pharmaceutically acceptable carrier, diluent, excipient, buffer, salt, surfactant, cryoprotectant, etc., and the implementation conditions and implementation embodiments are as described for the pharmaceutical composition.
상기 선천성 면역 증진용 조성물은 사용 목적에 따라, 백신 애주번트, 면역 항암제, 또는 항바이러스제 (예를 들어, hepatitis B virus) 내 유효 성분으로 활용될 수 있다. The above composition for enhancing innate immunity can be utilized as an effective ingredient in a vaccine adjuvant, an immune anticancer agent, or an antiviral agent (e.g., hepatitis B virus), depending on the intended use.
상기 선천성 면역 증진용 조성물은 예를 들어, 하나 이상의 지질 성분과 복합체를 형성하여, 리포솜 (liposomes), 지질 나노입자 (lipid nanoparticles) 및/또는 리포플렉스 (lipoplexes)를 형성하는 것일 수 있다. 상기 지질 나노입자 (lipid nanoparticles)는 이온화 가능한 양이온성 지질 (ionizable cationic lipid)을 일 구성 성분으로 포함하며, mRNA를 캡슐화하거나 전달 효율, 안정화에 도움을 주는 헬퍼 지질 (helper lipid), 안정화제 (stabilizer) 와 같은 기타 성분 또한 포함할 수 있다. 상기 지질 나노입자는 당업계의 공지의 성분을 적용할 수 있으며, 양이온성 지질, 헬퍼 지질, 및 PEG-접합된 지질을 포함할 수 있다. 상기 언급된 구성 성분이외에도, 유효량, 제형, 투여 방식, 병용 제제 등에 대하여, 당업계에서 공지의 기술이 비제한적으로 활용될 수 있다. The composition for enhancing innate immunity may, for example, form a complex with one or more lipid components to form liposomes, lipid nanoparticles and/or lipoplexes. The lipid nanoparticles include an ionizable cationic lipid as one component, and may also include other components such as a helper lipid or a stabilizer that encapsulates mRNA or helps with delivery efficiency and stabilization. The lipid nanoparticles may apply components known in the art, and may include cationic lipids, helper lipids, and PEG-conjugated lipids. In addition to the above-mentioned components, techniques known in the art may be utilized without limitation with respect to effective amounts, formulations, administration methods, combination preparations, etc.
또 다른 양상은 상기 이중가닥 RNA를 개체에 투여하는 단계를 포함하는 질병을 치료하는 방법 또는 병리적 상태를 개선하는 방법을 제공한다. Another aspect provides a method of treating a disease or ameliorating a pathological condition comprising administering to a subject the double-stranded RNA.
또 다른 양상은 상기 약학적 조성물을 개체에 투여하는 단계를 포함하는 선천성 면역 반응을 증진시키는 방법을 제공한다.Another aspect provides a method of enhancing an innate immune response comprising administering to a subject the pharmaceutical composition.
또 다른 양상은 질병 또는 병리적 상태에 대한 치료 또는 개선을 위한 의약의 제조를 위한 상기 이중가닥 RNA의 용도를 제공한다.Another aspect provides the use of said double-stranded RNA for the manufacture of a medicament for treating or ameliorating a disease or pathological condition.
또 다른 양상은 선천성 면역 반응을 증진시키기 위한 의약의 제조를 위한 상기 이중가닥 RNA의 용도를 제공한다.Another aspect provides the use of said double-stranded RNA for the manufacture of a medicament for enhancing innate immune response.
상기 방법 또는 용도는 전술한 이중가닥 RNA 또는 약학적 조성물을 그대로 포함하거나 이를 이용하기 때문에, 이들 사이에 공통된 내용은 본 명세서의 과도한 복잡성을 피하기 위하여, 그 기재를 생략한다. 또한, 상기 질병 또는 병리적 상태에 대해서는 전술한 바와 같다.Since the above method or use includes or utilizes the above-described double-stranded RNA or pharmaceutical composition as it is, the description of common contents between them is omitted to avoid excessive complexity of this specification. In addition, the disease or pathological condition is as described above.
본 명세서에서 용어, "개체"는 질병, 구체적으로 질환의 치료를 필요로 하는 대상을 의미하고, 보다 구체적으로는, 인간 또는 비-인간인 영장류, 생쥐 (mouse), 개, 고양이, 말, 소, 양, 돼지, 염소, 낙타, 영양 등의 포유류를 모두 포함할 수 있다.The term "subject" as used herein means a subject in need of treatment for a disease, specifically a condition, and more specifically, may include any mammal, such as a human or non-human primate, mouse, dog, cat, horse, cow, sheep, pig, goat, camel, or antelope.
이하 본 발명을 실시예를 통하여 보다 상세하게 설명한다. 그러나, 이들 실시예는 본 발명을 예시적으로 설명하기 위한 것으로 본 발명의 범위가 이들 실시예에 한정되는 것은 아니다.Hereinafter, the present invention will be described in more detail through examples. However, these examples are intended to exemplify the present invention and the scope of the present invention is not limited to these examples.
실시예 1. 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA의 제작 Example 1. Production of double-stranded RNA containing an innate immune activation motif
본 실시예에서는 선천성 면역을 활성화시킬 수 있는 다양한 길이의 이중가닥 RNA를 제작하였다. 도 1에 도시한 바와 같이, 상기 이중가닥 RNA는 제1 가닥 및 상기 제1 가닥과 상보적으로 결합하는 제2 가닥으로 구성되는 38 내지 60개 염기쌍으로 이루어지고, 제1 가닥의 3' 말단 영역 및 제2 가닥의 5' 말단 영역에 위치하는 선천성 면역 활성화 모티프를 포함하며, 여기서, 제2 가닥의 5' 말단 영역에 서열번호 6 (19nt) 또는 서열번호 8 (20nt)의 선천성 면역 활성화 서열이 위치하도록 제작되었다. 또한, 상기 이중가닥 RNA의 구조에 따른 효능을 비교하기 위하여, 전술한 기본 구조를 바탕으로 이중가닥 RNA의 말단 영역을 변형하여, 평활 말단, 1개의 돌출부, 또는 2개의 돌출부를 갖는 이중가닥 RNA를 각각 제작하였다. 구체적으로, 상기 이중가닥 RNA는 Bioneer Co. Ltd (Republic of Korea), 및 Dharmacon, Inc (UK)로부터 수득하였고, 서열번호 6의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA는 표 1에 나타낸 바와 같으며, 서열번호 8의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA는 표 2에 나타낸 바와 같다. 표 1 및 표 2에서, 선천성 면역 활성화 서열은 밑줄로 표기하였고, 말단 영역에 형성된 돌출부는 음영으로 표기하였다. In this example, double-stranded RNAs of various lengths capable of activating innate immunity were produced. As illustrated in Fig. 1, the double-stranded RNA is composed of 38 to 60 base pairs, which are composed of a first strand and a second strand complementarily binding to the first strand, and includes an innate immune activation motif located at the 3' terminal region of the first strand and the 5' terminal region of the second strand, wherein the innate immune activation sequence of SEQ ID NO: 6 (19 nt) or SEQ ID NO: 8 (20 nt) is located at the 5' terminal region of the second strand. In addition, in order to compare the efficacy according to the structure of the double-stranded RNA, the terminal region of the double-stranded RNA was modified based on the above-described basic structure to produce double-stranded RNAs having a smooth end, one protrusion, or two protrusions, respectively. Specifically, the double-stranded RNA was purchased from Bioneer Co. Ltd (Republic of Korea), and Dharmacon, Inc (UK), and the double-stranded RNA comprising the innate immune activation sequence of SEQ ID NO: 6 is as shown in Table 1, and the double-stranded RNA comprising the innate immune activation sequence of SEQ ID NO: 8 is as shown in Table 2. In Tables 1 and 2, the innate immune activation sequence is underlined, and the protrusion formed in the terminal region is shaded.
[표 1][Table 1]
[표 2][Table 2]
실시예 2. 선천성 면역 활성화 모티프 도입에 따른 효과 확인 Example 2. Confirmation of the effect of introducing an innate immune activation motif
2-1. 표적 유전자에 대한 넉다운 효과2-1. Knockdown effect on target gene
본 실시예에서는 상기 실시예 1에 따른 이중가닥 RNA의 표적 유전자에 대한 넉다운 효과를 확인하고자 하였다. 상기 실시예 1에 따른 선천성 면역 활성화 모티프를 포함하는 19, 38, 50, 또는 60bp의 이중가닥 RNA (표 1)의 Luciferase 유전자에 대한 넉다운 효과를 Luciferase plasmid (E1741, Promega가 처리된 HeLa (CCL-2, ATCC) 세포주를 대상으로 확인하였다. 구체적으로, 상기 HeLa (CCL-2, ATCC) 세포주를 항생제가 없는 완전 배지에서 30% 컨플루언시 (confluency)로 24웰 플레이트에 플레이팅하였다. 이후, 상기 세포주에 이중가닥 RNA를 Lipofectamine RNAiMAX (13778150; Invitrogen)를 사용하여 형질감염시켰다. 형질감염 시점으로부터 24시간 후, Tri-RNA Reagent (Favorgen)를 사용하여 총 RNA를 추출하고, 0.5㎍의 총 RNA를 cDNA 합성에 사용하였다. 표적 유전자에 대한 프라이머를 SYBR Green PCR Master Mix와 혼합하고 상대 표준 곡선 정량 방법 (relative standard curve quantitation method)을 사용하여 표적 유전자 mRNA 수준을 측정하였다. 이후, GraphPad Prism 8을 사용하여, 용량-반응 곡선에 기초한 IC50 값을 산출하였다. In this example, the knockdown effect of the double-stranded RNA according to Example 1 on the target gene was confirmed. The knockdown effect of the 19, 38, 50, or 60 bp double-stranded RNA (Table 1) containing the innate immune activation motif according to the above Example 1 on the Luciferase gene was confirmed in HeLa (CCL-2, ATCC) cell line treated with Luciferase plasmid (E1741, Promega). Specifically, the HeLa (CCL-2, ATCC) cell line was plated in a 24-well plate at 30% confluency in complete medium without antibiotics. Then, the cell line was transfected with double-stranded RNA using Lipofectamine RNAiMAX (13778150; Invitrogen). After 24 hours from the time of transfection, total RNA was extracted using Tri-RNA Reagent (Favorgen), and 0.5 μg of total RNA was used for cDNA synthesis. Primers for the target gene were mixed with SYBR Green PCR Master Mix and quantified by relative standard curve. The target gene mRNA levels were measured using the relative standard curve quantitation method. Then, IC 50 values were calculated based on the dose-response curve using
그 결과, 표 3에 나타낸 바와 같이, 모든 이중가닥 RNA는 길이 또는 말단 구조에 관계없이 표적 유전자에 대해 0.5nM 미만의 IC50 값을 나타내었다. 상기 결과는 상기 이중가닥 RNA 내 선천성 면역 활성화 모티프의 도입은 표적 유전자 넉다운 효과에 영향을 미치지 않으며, 즉, 이중가닥 RNA 본연의 기능에 영향을 미치지 않은 상태로 독립적으로 작용할 수 있음을 나타내는 것이다. As a result, as shown in Table 3, all double-stranded RNAs showed IC 50 values of less than 0.5 nM for the target gene, regardless of length or terminal structure. The results indicate that the introduction of the innate immune activation motif into the double-stranded RNA does not affect the target gene knockdown effect, i.e., it can act independently without affecting the original function of the double-stranded RNA.
[표 3][Table 3]
2-2. 선천성 면역 반응 유도 효과2-2. Effect of inducing innate immune response
본 실시예에서는 이중가닥 RNA의 말단에 도입된 선천성 면역 활성화 모티프가 선천성 면역 반응에 미치는 영향을 확인하고자 하였다. 상기 실시예 1에 따른 서열번호 6의 선천성 면역 활성화 서열을 포함하는 38, 50, 또는 60bp의 이중가닥 RNA (표 1) 및 서열번호 8의 선천성 면역 활성화 서열을 포함하는 40bp의 이중가닥 RNA (표 2)에 대하여 선천성 면역 반응과 관련된 사이토카인 발현을 분석하였다. 구체적으로, T98G (CRL-1690, ATCC) 세포주를 항생제가 없는 완전 배지에서 30% 컨플루언시로 24웰 플레이트에 플레이팅하였다. 이후 상기 세포주에 이중가닥 RNA를 Lipofectamine RNAiMAX (13778150; Invitrogen)를 사용하여 형질감염시키고, 상기 형질전환된 세포를 각각 12시간 또는 24시간 후에 수득하였다. 이후, IFIT1, ISG15, CXCL10 수준을 상기 실시예 2-1의 방법에 따라 qRT-PCR을 사용하여 분석하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다.In this example, we attempted to determine the effect of the innate immune activation motif introduced at the end of double-stranded RNA on the innate immune response. The expression of cytokines related to the innate immune response was analyzed for the 38, 50, or 60 bp double-stranded RNAs comprising the innate immune activation sequence of SEQ ID NO: 6 according to Example 1 (Table 1) and the 40 bp double-stranded RNAs comprising the innate immune activation sequence of SEQ ID NO: 8 (Table 2). Specifically, T98G (CRL-1690, ATCC) cell line was plated in a 24-well plate at 30% confluency in complete medium without antibiotics. Thereafter, the double-stranded RNA was transfected into the cell line using Lipofectamine RNAiMAX (13778150; Invitrogen), and the transfected cells were harvested after 12 or 24 hours, respectively. Thereafter, the levels of IFIT1, ISG15, and CXCL10 were analyzed using qRT-PCR according to the method of Example 2-1. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as a group to which only the transfection reagent was added (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 2 및 도 3에 나타낸 바와 같이, 서열번호 6의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA는 선천성 면역 반응과 관련된 IFIT1, CXCL10, ISG15, TNF-α 및 IL-1β의 높은 발현을 유도하였다. 특히, 이러한 효과는 이중가닥 RNA의 말단 구조가 평활 말단인 경우에 보다 우수하였으며, 상기와 동일한 구조를 지니되, 이중가닥 RNA의 말단에 상기 모티프가 도입되지 않은 이중가닥 RNA의 효과에 비해서도 매우 현저한 것이었다. 이러한 결과는 상기 선천성 면역 반응의 유도는 이중가닥 RNA의 말단 영역에 위치하는 선천성 면역 활성화 모티프에 영향을 받음을 나타내는 것이다. As a result, as shown in FIGS. 2 and 3, the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 6 induced high expression of IFIT1, CXCL10, ISG15, TNF-α, and IL-1β related to the innate immune response. In particular, this effect was more excellent when the terminal structure of the double-stranded RNA was a blunt end, and was also very remarkable compared to the effect of the double-stranded RNA having the same structure as above but without the motif introduced at the terminal of the double-stranded RNA. These results indicate that the induction of the innate immune response is affected by the innate immune activation motif located at the terminal region of the double-stranded RNA.
또한, 도 4에 나타낸 바와 같이, 서열번호 8의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA 역시 선천성 면역 반응과 관련된 IFIT1, CXCL10, 및 ISG15의 높은 발현을 유도하였다. 한편, 상기와 동일한 구조를 지니되, 이중가닥 RNA의 말단에 상기 모티프가 도입되지 않은 40bp의 이중가닥 RNA (Lamin A/G-GAPDH, Lamin A/C-Luciferase, GAPDH-Luciferase)를 처리한 군과, 짧은 길이의 표적 결합 서열 및 모티프 서열 각각을 혼합하여 처리한 군 (mixture of 19bp)에서는 모두 유효한 수준의 선천성 면역 반응을 유도하지 못하였다. 따라서, 전술한 선천성 면역 반응 유도 효과는 이중가닥 RNA에 도입된, 일 실시예에 따른 선천성 면역 활성화 모티프로부터 비롯된 것임을 나타낸다. Also, as shown in FIG. 4, the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 8 also induced high expression of IFIT1, CXCL10, and ISG15 related to the innate immune response. On the other hand, the group treated with the 40 bp double-stranded RNA (Lamin A/G-GAPDH, Lamin A/C-Luciferase, GAPDH-Luciferase) having the same structure as above but without the motif introduced at the end of the double-stranded RNA, and the group treated by mixing each of the short-length target binding sequence and motif sequence (mixture of 19 bp) did not induce an effective level of innate immune response. Therefore, it is shown that the above-mentioned innate immune response induction effect is derived from the innate immune activation motif according to one embodiment introduced into the double-stranded RNA.
2-3. RIG-I 매개 선천성 면역 반응 유도 효과2-3. Effect of RIG-I mediated innate immune response induction
본 실시예에서는 선천성 면역 반응을 매개하는 인자로서, RIG-Ⅰ와의 관련성을 확인하고자 하였다. 상기 실시예 1에 따른 서열번호 6의 선천성 면역 활성화 서열을 포함하는 38, 50, 또는 60bp의 이중가닥 RNA (표 1) 및 서열번호 8의 선천성 면역 활성화 서열을 포함하는 또는 40bp의 이중가닥 RNA (표 2)에 대하여 RAW-Lucia ISG 세포주 (WT) 및 RAW-Lucia ISG-KO-RIG-I 세포주 (RIG-I KO)에 대한 IRF의 활성도를 측정하였다. 구체적으로, RAW-Lucia ISG 세포주 및 RAW-Lucia ISG-KO-RIG-I 세포주를 96웰 플레이트에 웰당 1 × 105 개씩 플레이팅하였다. 이후, 상기 세포주에 상기 이중가닥 RNA (10nM)를 Lipofectamine RNAiMAX (13778150; Invitrogen)를 사용하여 형질감염시키고, 상기 형질전환된 세포를 각각 24시간 배양한 후에 상등액 수득하였다. 이후, 상등액을 대상으로, 플레이트 판독기 (VICTORX2; PerkinElmer)를 사용하여, 인터페론 조절 인자(Interferon Regulatory Factor, IRF) 활성 수준을 검출하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C 및/또는 LPS를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다.In this example, we attempted to confirm the relationship with RIG-I as a factor mediating the innate immune response. The activity of IRF for the RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured for the 38, 50, or 60 bp double-stranded RNA (Table 1) including the innate immune activation sequence of SEQ ID NO: 6 according to Example 1 and the 40 bp double-stranded RNA (Table 2) including the innate immune activation sequence of SEQ ID NO: 8. Specifically, the RAW-Lucia ISG cell line and the RAW-Lucia ISG-KO-RIG-I cell line were plated at 1 × 10 5 cells per well in a 96-well plate. Thereafter, the double-stranded RNA (10 nM) was transfected into the cell line using Lipofectamine RNAiMAX (13778150; Invitrogen), and the transfected cells were each cultured for 24 hours to obtain the supernatant. Thereafter, the interferon regulatory factor (IRF) activity level was detected in the supernatant using a plate reader (VICTORX2; PerkinElmer). Meanwhile, in this example, the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 5 및 도 6에 나타낸 바와 같이, 서열번호 6 또는 서열번호 8의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA이 처리된 RAW-Lucia ISG 세포주에서는 높은 수준의 선천성 면역 반응이 확인되었다. 특히, 상기 이중가닥 RNA 중에서도 양 말단이 모두 평활 말단이거나, 선천성 면역 활성화 모티프와 인접된 말단이 평활 말단인 경우에 전술한 선천성 면역 반응이 유도됨을 알 수 있었다. 반면, RIG-Ⅰ가 결여된 ISG-KO-RIG-I 세포주에서는 이러한 선천성 면역 반응이 소실되거나 현격하게 감소되었다. 특히, 도 6에 나타낸 바와 같이, 상기 이중가닥 RNA에서, 양 말단이 모두 평활 말단을 형성하는 경우뿐만 아니라, 선천성 면역 활성화 모티프와 인접하는 어느 하나의 말단 영역에 평활 말단을 갖는 경우에도 유사한 효과를 보여주었다. 상기 결과는 이중가닥 RNA의 말단 영역에 위치하는 모티프로부터 비롯된 선천성 면역 반응은 RIG-Ⅰ가 매개하는 것임을 나타내는 것이다. As a result, as shown in FIGS. 5 and 6, a high level of innate immune response was confirmed in the RAW-Lucia ISG cell line treated with the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 6 or SEQ ID NO: 8. In particular, it was found that the innate immune response described above was induced when, among the double-stranded RNAs, both ends were blunt-ended or the end adjacent to the innate immune activation motif was blunt-ended. On the other hand, in the ISG-KO-RIG-I cell line lacking RIG-I, the innate immune response was lost or significantly reduced. In particular, as shown in FIG. 6, in the double-stranded RNA, a similar effect was shown not only when both ends formed blunt-ended, but also when one of the terminal regions adjacent to the innate immune activation motif had a blunt-ended end. The above results indicate that the innate immune response derived from motifs located in the terminal region of double-stranded RNA is mediated by RIG-I.
실시예 3. 변형된 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA의 선천성 면역 반응 유도 효과 확인Example 3. Confirmation of the innate immune response-inducing effect of double-stranded RNA containing a modified innate immune activation motif
3-1. 40bp의 길이를 갖는 이중가닥 RNA의 효능 확인 3-1. Confirmation of the efficacy of double-stranded RNA with a length of 40 bp
본 실시예에서는 변형된 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에 의한 선천성 면역 활성화 수준을 평가하고자 하였다. 이를 위하여, 서열번호 2의 뉴클레오티드 서열 (10nt), 또는 서열번호 4의 뉴클레오티드 서열 (15nt)이 제 2가닥의 5' 말단에 위치하는 이중가닥 RNA를 제작하였으며, 구체적인 구조는 도 1에 도시한 바와 같다. 이와 더불어, 상기 선천성 면역 활성화 모티프가 위치하는 영역에 따른 효과의 차이를 비교하고자, 서열번호 8의 뉴클레오티드 서열이 말단외 영역에 도입된 이중가닥 RNA를 추가로 제작하였다. 구체적으로, 상기 이중가닥 RNA는 Bioneer Co. Ltd (Republic of Korea), 및 Dharmacon, Inc (UK)로부터 수득하였고, 서열번호 2 또는 서열번호 4의 선천성 면역 활성화 서열을 포함하는 이중가닥 RNA는 표 4에 나타낸 바와 같다. 표 4에서, 선천성 면역 활성화 서열은 밑줄로 표기하였다. In this example, we aimed to evaluate the level of innate immune activation by double-stranded RNA including a modified innate immune activation motif. To this end, a double-stranded RNA was produced in which the nucleotide sequence of SEQ ID NO: 2 (10 nt) or the nucleotide sequence of SEQ ID NO: 4 (15 nt) is located at the 5' end of the second strand, and its specific structure is as shown in Fig. 1. In addition, in order to compare the difference in effect depending on the region in which the innate immune activation motif is located, a double-stranded RNA in which the nucleotide sequence of SEQ ID NO: 8 was introduced into the extra-terminal region was additionally produced. Specifically, the double-stranded RNA was obtained from Bioneer Co. Ltd (Republic of Korea), and Dharmacon, Inc (UK), and the double-stranded RNA including the innate immune activation sequence of SEQ ID NO: 2 or SEQ ID NO: 4 is as shown in Table 4. In Table 4, the innate immune activation sequence is underlined.
[표 4][Table 4]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주 (WT) 및 RAW-Lucia ISG-KO-RIG-I 세포주 (RIG-I KO)에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C 및/또는 LPS를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다.Thereafter, in the same manner as in Example 2-3, the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured. Meanwhile, in this example, the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 7에 나타낸 바와 같이, 서열번호 2의 뉴클레오티드 서열, 또는 서열번호 4의 뉴클레오티드 서열이 말단에 포함된 이중가닥 RNA 역시 상기 실시예 2-3과 동일하게, RIG-Ⅰ매개 선천성 면역 반응을 증진시킬 수 있었다. 다만, 서열번호 8의 뉴클레오티드 서열이 전체 서열의 중앙으로 이동된 이중가닥 RNA (Motif-20-Shifted)는 이러한 효과가 현격하게 감소되었다. 상기 결과로부터 10nt로 이루어진 서열번호 2의 모티프는 RIG-Ⅰ매개 선천성 면역 반응 유도를 위한 단위체 또는 핵심 서열로 적용될 수 있고, 상기 서열은 이중가닥 RNA 내 말단에 도입 또는 위치하는 경우에 유효한 효능이 발휘될 수 있음을 알 수 있었다. As a result, as shown in FIG. 7, the double-stranded RNA including the nucleotide sequence of SEQ ID NO: 2 or the nucleotide sequence of SEQ ID NO: 4 at the terminal was also able to enhance the RIG-I-mediated innate immune response, similarly to Example 2-3. However, the double-stranded RNA (Motif-20-Shifted) in which the nucleotide sequence of SEQ ID NO: 8 was shifted to the center of the entire sequence showed a significant decrease in this effect. From the above results, it was found that the motif of SEQ ID NO: 2 consisting of 10 nt can be applied as a unit or core sequence for inducing the RIG-I-mediated innate immune response, and that the sequence can exhibit effective efficacy when introduced or positioned at the terminal in the double-stranded RNA.
3-2. 30bp의 길이를 갖는 이중가닥 RNA의 효능 확인 3-2. Confirmation of the efficacy of double-stranded RNA with a length of 30 bp
본 실시예에서는 실시예 2 및 실시예 3-1의 실험 결과를 토대로, 서열번호 2의 뉴클레오티드 서열 (10nt)이 이중가닥 RNA 내 제2 가닥의 5'말단 또는 제1 가닥의 3'말단에 위치하며, 양 말단이 평활 말단인, 30bp의 이중가닥 RNA를 제조하였다. 상기 선천성 면역 활성화 모티프의 이중가닥 내 위치가 선천성 면역 반응에 미치는 영향을 확인하고자, 제2 가닥의 5'말단에 선천성 면역 활성화 서열이 위치하는 이중가닥 RNA (LaminA/C-Motif-10), 및 제1 가닥의 3'말단에 선천성 면역 활성화 서열이 위치하는 이중가닥 RNA (LaminA/C-inv. Motif-10)를 각각 제조하였다. 구체적으로, 상기 이중가닥 RNA는 Bioneer Co. Ltd (Republic of Korea), 및 Dharmacon, Inc (UK)로부터 수득하였고, 전술한 이중가닥 RNA는 표 5에 나타낸 바와 같다. 표 5에서, 선천성 면역 활성화 서열은 밑줄로 표기하였다. In this example, based on the experimental results of Example 2 and Example 3-1, a 30 bp double-stranded RNA was prepared in which the nucleotide sequence (10 nt) of SEQ ID NO: 2 is located at the 5' end of the second strand or the 3' end of the first strand in the double-stranded RNA and both ends are blunt ends. In order to confirm the effect of the position of the innate immune activation motif in the double-stranded RNA on the innate immune response, a double-stranded RNA (LaminA/C-Motif-10) in which the innate immune activation sequence is located at the 5' end of the second strand and a double-stranded RNA (LaminA/C-inv. Motif-10) in which the innate immune activation sequence is located at the 3' end of the first strand were prepared, respectively. Specifically, the double-stranded RNA was obtained from Bioneer Co. Ltd (Republic of Korea), and Dharmacon, Inc (UK), and the above-mentioned double-stranded RNAs are as shown in Table 5. In Table 5, the innate immune activation sequences are underlined.
[표 5][Table 5]
이후, Huh7 세포주를 대상으로 상기 실시예 2-1과 동일한 방식으로 10nM의 이중가닥 RNA의 처리에 따른 표적 유전자인 Lamin A/C의 넉다운 효과를 확인하였다. 그 결과, 도 8에 나타낸 바와 같이, 30bp의 길이를 갖는 이중가닥 RNA 역시 실시예 2-1과 유사하게, 표적 유전자에 대한 넉다운 효과를 보여주었다. Thereafter, the knockdown effect of the target gene, Lamin A/C, upon treatment with 10 nM of double-stranded RNA was confirmed using the Huh7 cell line in the same manner as in Example 2-1. As a result, as shown in Fig. 8, the double-stranded RNA having a length of 30 bp also showed a knockdown effect on the target gene, similar to Example 2-1.
또한, 실시예 2-3과 동일한 방식으로, 10nM의 이중가닥 RNA의 처리에 따른 RAW-Lucia ISG 세포주 (WT) 및 RAW-Lucia ISG-KO-RIG-I 세포주 (RIG-I KO)에 대한 IRF의 활성도를 측정하였다. 그 결과, 도 9에 나타낸 바와 같이, 선천성 면역 활성화 서열이 제2 가닥의 5' 말단에 위치하는 경우뿐만 아니라, 제1 가닥의 3' 말단에 위치하는 경우에도, 선천성 면역 반응을 증진시킬 수 있으며, 이러한 선천성 면역 반응 역시 RIG-Ⅰ이 매개하는 것임을 알 수 있었다. In addition, in the same manner as in Example 2-3, the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) according to treatment with 10 nM of double-stranded RNA was measured. As a result, as shown in Fig. 9, it was found that the innate immune response can be enhanced not only when the innate immune activation sequence is located at the 5' end of the second strand but also when it is located at the 3' end of the first strand, and that this innate immune response is also mediated by RIG-I.
실시예 4. 화학적 변형이 도입된 이중가닥 RNA의 선천성 면역 반응 유도 효과 확인Example 4. Confirmation of the innate immune response induction effect of double-stranded RNA with chemical modifications
본 실시예에서는 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에서, 화학적 변형이 도입된 이중가닥 RNA를 제조하고, 상기 화학적 변형이 도입된 이중가닥 RNA의 선천성 면역 반응 유도 효과를 평가하였다. In this example, a double-stranded RNA containing an innate immune activation motif was prepared by introducing chemical modifications into a double-stranded RNA, and the innate immune response induction effect of the double-stranded RNA with the chemical modifications was evaluated.
4-1. 화학적 변형이 도입된 가닥 (strand)에 따른 효과 확인 4-1. Confirmation of the effect according to the strand into which chemical modification was introduced
본 실시예에서는 서열번호 2의 선천성 면역 활성화 서열이 제2 가닥의 5' 말단에 삽입된 30bp의 이중가닥 RNA를 기본 골격으로 하여, 뉴클레오티드의 2' 탄소 위치의 -OH기가 -OCH3(-O-methyl)로 치환된 화학적 변형이 도입된 이중가닥 RNA를 제조하였다. 선천성 면역 반응 유도 효과에 화학적 변형이 도입된 가닥의 종류가 미치는 영향을 확인하고자, 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥에 화학적 변형이 도입된 이중가닥 RNA와, 제1 가닥 및 제2 가닥 모두에 화학적 변형이 도입된 이중가닥 RNA를 각각 제조하였다. 구체적으로, 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥에 화학적 변형이 도입된 이중가닥 RNA는 표 6에 나타낸 바와 같고, 제1 가닥 및 제2 가닥 모두에 화학적 변형이 도입된 이중가닥 RNA는 표 7에 나타낸 바와 같다. 표 6 및 표 7에서, 이중가닥 RNA의 명명은 [화학적 변형이 도입된 가닥]-[화학적 변형이 도입된 말단 및 화학적 변형이 도입된 첫번째 뉴클레오티드 위치]-[화학적 변형이 도입된 뉴클레오티드 개수]로 표기하였다. 또한, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다. In this example, a 30-bp double-stranded RNA having the innate immune activation sequence of
[표 6][Table 6]
[표 7][Table 7]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주 (WT) 및 RAW-Lucia ISG-KO-RIG-I 세포주 (RIG-I KO)에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C 및/또는 LPS를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다.Thereafter, in the same manner as in Example 2-3, the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured. Meanwhile, in this example, the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 10에 나타낸 바와 같이, 제1 가닥 또는 제2 가닥 중 어느 하나의 가닥에 화학적 변형이 도입된 이중가닥 RNA에서, 화학적 변형이 도입된 8개의 뉴클레오티드를 포함하는 이중가닥 RNA (#1 5'-1-8, #2 3'-2-8, #1 5'-2-8, 및 #2 3'-1-8)는 선천성 면역 반응 모티프로부터 비롯된 효과, 즉, 선천성 면역 반응 유도 효과가 모두 소실되었다. 반면, 화학적 변형이 도입된 10개의 뉴클레오티드가 제1 가닥 또는 제2 가닥 중 어느 하나에 도입된 이중가닥 RNA (#1 5'-1-10, #2 3'-2-10, 및 #1 5'-2-10, #2 3'-1-10)는 높은 수준의 선천성 면역 유도 효과를 나타내었으며, 이러한 효과는 RIG-Ⅰ가 결여된 ISG-KO-RIG-I 세포주에서 유의적으로 감소되었다. 또한, 도 11에 나타낸 바와 같이, 제1 가닥 및 제2 가닥 모두에 화학적 변형이 도입된 이중가닥 RNA는 선천성 면역 반응 유도 효과가 모두 소실되었다.As a result, as shown in Fig. 10, in the double-stranded RNA having chemical modifications introduced into either the first strand or the second strand, the double-stranded RNA containing eight nucleotides to which chemical modifications were introduced (#1 5'-1-8, #2 3'-2-8, #1 5'-2-8, and #2 3'-1-8) completely lost the effect derived from the innate immune response motif, i.e., the innate immune response induction effect. In contrast, the double-stranded RNA having ten nucleotides to which chemical modifications were introduced into either the first strand or the second strand (#1 5'-1-10, #2 3'-2-10, and #1 5'-2-10, #2 3'-1-10) exhibited a high level of innate immune induction effect, and this effect was significantly reduced in the ISG-KO-RIG-I cell line lacking RIG-I. In addition, as shown in Fig. 11, double-stranded RNA with chemical modifications introduced to both the first and second strands completely lost the innate immune response induction effect.
4-2. 화학적 변형이 도입된 뉴클레오티드의 위치에 따른 효과 확인 4-2. Confirmation of the effect according to the position of the nucleotide where chemical modification is introduced
선천성 면역 반응 유도 효과에 화학적 변형이 도입된 뉴클레오티드의 위치가 미치는 영향을 확인하고자 하였다. 이를 위하여, 상기 실시예 4-1의 이중가닥 RNA를 기본 골격으로 하여, 화학적 변형이 도입된 10개의 뉴클레오티드를 제1 가닥의 5'말단 또는 3' 말단으로 위치시키거나 (#1 5'-1-10, #1 3'-1-10), 화학적 변형이 도입된 첫번째 뉴클레오티드 위치를 변경하였다 (#1 5'-1-10, #1 5'-2-10). 추가적으로, 이중가닥 RNA 내 선천성 면역 활성 모티프외 영역의 서열을 변경하면서, 화학적 변형이 도입된 이중가닥 RNA를 제조하였다(GAPDH-Motif). 구체적으로, 전술한 화학적 변형이 도입된 이중가닥 RNA는 표 8에 나타낸 바와 같다. 상기 표 8에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다. This study was conducted to determine the effect of the position of the chemically modified nucleotides on the innate immune response induction effect. To this end, using the double-stranded RNA of Example 4-1 as the basic framework, 10 nucleotides with chemical modifications were positioned at the 5' end or 3' end of the first strand (#1 5'-1-10, #1 3'-1-10), or the position of the first nucleotide with chemical modifications was changed (#1 5'-1-10, #1 5'-2-10). Additionally, a double-stranded RNA with chemical modifications was prepared by changing the sequence of a region other than the innate immune activation motif in the double-stranded RNA (GAPDH-Motif). Specifically, the double-stranded RNA with the chemical modifications introduced as described above is as shown in Table 8. In Table 8 above, innate immune activation motifs and innate immune activation sequences are shaded and/or bold, respectively, and nucleotides with 2'-OCH 3 (-O-methyl) chemical modifications are underlined.
[표 8][Table 8]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주(WT) 및 RAW-Lucia ISG-KO-RIG-I 세포주(RIG-I KO)에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C 및/또는 LPS를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군(mock) 및 이중가닥 RNA를 처리하지 않은 군(NT)을 사용하였다.Thereafter, in the same manner as in Example 2-3, the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured. Meanwhile, in this example, the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 12에 나타낸 바와 같이, 선천성 면역 활성화 모티프를 포함하지 않은 이중가닥 RNA는 화학적 변형의 도입과 관계 없이, 유효한 수준의 선천성 면역 반응을 유도하지 못했던 반면(Lamin A/C-GAPDH, Lamin A/C-GAPDH #1 5'-2-10), 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에서는, 10개의 화학적 변형이 도입된 경우, 이중가닥 RNA 내 선천성 면역 활성 모티프외 영역의 서열 및 단일 가닥 내 선천성 면역 활성 모티프의 위치에 구애받지 않고, 높은 수준의 선천성 면역 유도 효과를 나타내었다. As a result, as shown in Fig. 12, the double-stranded RNA without the innate immune activation motif did not induce an effective level of innate immune response regardless of the introduction of chemical modifications (Lamin A/C-GAPDH, Lamin A/C-
4-3. 화학적 변형이 도입된 뉴클레오티드의 개수에 따른 효과 확인 4-3. Confirmation of the effect according to the number of nucleotides to which chemical modifications have been introduced
선천성 면역 반응 유도 효과에 화학적 변형이 도입된 뉴클레오티드의 개수가 미치는 영향을 확인하고자 하였다. 이를 위하여, 상기 실시예 4-1의 이중가닥 RNA 를 기본 골격으로 하여, 화학적 변형이 도입된 8개, 10개, 12개, 또는 15개의 뉴클레오티드를 제1 가닥의 5'말단 위치시켰다 (AS 5'-2-8, AS 5'-2-10, AS 5'-2-12, 및 AS 5'-2-15). 추가적으로, 상기의 화학적 변형과 함께, 제1 가닥 및 제2 가닥 모두에 화학적 변형이 도입된 이중가닥 RNA를 제조하여, 이들간 선천성 면역 반응 유도 효과를 비교하였다. 구체적으로, 전술한 화학적 변형이 도입된 이중가닥 RNA는 표 9에 나타낸 바와 같다. 상기 표 9에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였으며, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다.This study was conducted to determine the effect of the number of chemically modified nucleotides on the innate immune response induction effect. To this end, using the double-stranded RNA of Example 4-1 as the basic framework, 8, 10, 12, or 15 chemically modified nucleotides were positioned at the 5' end of the first strand (AS 5'-2-8, AS 5'-2-10, AS 5'-2-12, and AS 5'-2-15). Additionally, double-stranded RNAs in which chemical modifications were introduced to both the first strand and the second strand were prepared along with the chemical modifications described above, and their innate immune response induction effects were compared. Specifically, the double-stranded RNAs in which the chemical modifications described above were introduced are as shown in Table 9. In Table 9 above, innate immune activation motifs and innate immune activation sequences are shaded and/or bolded, respectively, and nucleotides with 2'-OCH 3 (-O-methyl) chemical modifications are underlined.
[표 9][Table 9]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주 (WT) 및 RAW-Lucia ISG-KO-RIG-I 세포주 (RIG-I KO)에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C 및/또는 LPS를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다.Thereafter, in the same manner as in Example 2-3, the activity of IRF for RAW-Lucia ISG cell line (WT) and RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO) was measured. Meanwhile, in this example, the positive control group was a group administered with Poly I:C and/or LPS, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 13에 나타낸 바와 같이, 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에서, 적어도 10개 이상의 화학적 변형이 도입된 경우 (10개, 12개, 15개), 높은 수준의 선천성 면역 유도 효과를 나타내었다. 또한, 이러한 효과는 RIG-Ⅰ가 결여된 ISG-KO-RIG-I 세포주에서 유의적으로 감소되었다. 아울러, 전술한 실시예의 결과와 마찬가지로, 화학적 변형이 도입된 8개의 뉴클레오티드를 포함하는 이중가닥 RNA, 및 제1 가닥 및 제2 가닥 모두에 화학적 변형이 도입된 이중가닥 RNA는 선천성 면역 반응 모티프로부터 비롯된 선천성 면역 반응 유도 효과가 모두 소실되었다. As a result, as shown in Fig. 13, in the double-stranded RNA including the innate immune activation motif, when at least 10 chemical modifications (10, 12, 15) were introduced, a high level of innate immunity induction effect was exhibited. In addition, this effect was significantly reduced in the ISG-KO-RIG-I cell line lacking RIG-I. In addition, similar to the results of the examples described above, the double-stranded RNA including 8 nucleotides into which chemical modifications were introduced, and the double-stranded RNA into which chemical modifications were introduced into both the first strand and the second strand, all innate immune response induction effects derived from the innate immune response motif were lost.
상기 결과를 종합하면, 일 실시예에 따른 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에서, 적어도 10개 이상의 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 하나에 도입된 경우, 이중가닥 RNA 내 선천성 면역 활성 모티프외 영역의 서열 및 단일 가닥 내 선천성 면역 활성 모티프의 위치에 구애받지 않고, RIG-Ⅰ매개 선천성 면역 반응의 증진 효과가 달성됨을 알 수 있었다.In summary of the above results, it was found that when at least 10 chemical modifications are introduced into either the first strand or the second strand in a double-stranded RNA including an innate immune activation motif according to one embodiment, the effect of enhancing the RIG-I-mediated innate immune response is achieved regardless of the sequence of the region outside the innate immune activation motif in the double-stranded RNA and the location of the innate immune activation motif in the single strand.
실시예 5. 화학적 변형이 도입된 이중가닥 RNA의 선천성 면역 반응 유도 기작 확인 Example 5. Confirmation of the mechanism of induction of innate immune response by double-stranded RNA with chemical modifications
상기 실시예 4의 화학적 변형이 도입된 이중가닥 RNA는, 화학적 변형이 도입되지 않은 실시예 1 내지 3에 따른 이중가닥 RNA에 비해 향상된 선천성 면역 반응 유도 효과를 보여 주었다. 또한, 상기 화학적 변형이 도입된 이중가닥 RNA는 화학적 변형이 도입되지 않은 이중가닥 RNA와 달리, RIG-Ⅰ가 결여된 ISG-KO-RIG-I 세포주에서 선천성 면역 반응이 완전히 소실되거나 현격하게 감소되지 않고, 일정 수준의 IRF의 활성을 보여 주었다 (도 10, 도 12, 도 13 참고). The double-stranded RNA with the chemical modification of Example 4 above showed an enhanced innate immune response induction effect compared to the double-stranded RNA according to Examples 1 to 3 without the chemical modification. In addition, the double-stranded RNA with the chemical modification introduced, unlike the double-stranded RNA without the chemical modification introduced, showed a certain level of IRF activity without completely disappearing or significantly reducing the innate immune response in the ISG-KO-RIG-I cell line lacking RIG-I (see FIGS. 10, 12, and 13).
따라서, 본 실시예에서는 화학적 변형의 도입에 따른 선천성 면역 반응 유도 효과는 RIG-Ⅰ와는 다른 기작에 의해 이루어질 수 있다는 전제 하에서, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주 (WT), RAW-Lucia ISG-KO-RIG-I 세포주 (RIG-I KO) 및 RAW-Lucia ISG-KO-MDA5-I 세포주 (MDA5 KO)에 대한 IRF의 활성도를 측정하였다. 구체적으로, 본 실시예에서 사용된 화학적 변형이 도입된 이중가닥 RNA는 표 10에 나타낸 바와 같다. 상기 표 10에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다.Therefore, in this example, under the premise that the innate immune response induction effect by introduction of chemical modifications can be achieved by a different mechanism from RIG-I, the activity of IRF was measured for RAW-Lucia ISG cell line (WT), RAW-Lucia ISG-KO-RIG-I cell line (RIG-I KO), and RAW-Lucia ISG-KO-MDA5-I cell line (MDA5 KO) in the same manner as in Example 2-3. Specifically, the double-stranded RNAs into which chemical modifications were introduced used in this example are as shown in Table 10. In Table 10, the innate immune activation motifs and innate immune activation sequences are indicated in shaded and/or bold, respectively, and the nucleotides into which 2'-OCH 3 (-O-methyl) chemical modifications were introduced are indicated in underline.
[표 10][Table 10]
그 결과, 도 14에 나타낸 바와 같이, 상기 실시예 4의 실험 결과와 마찬 가지로, 선천성 면역 활성화 모티프를 포함하지 않은 이중가닥 RNA는 유효한 수준의 선천성 면역 반응을 유도하지 못했던 반면, 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에서, 적어도 10개 이상의 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 하나에 도입된 경우, 높은 수준의 선천성 면역 유도 효과를 나타내었다. 특히, 이러한 효과는 종전 실시예의 실험 결과와 마찬가지로, RIG-Ⅰ가 결여된 ISG-KO-RIG-I 세포주에서 일부 감소되는 경향을 보인 반면, MDA5가 결여된 ISG-KO-MDA5 세포주에서는 소실되거나 현격하게 감소됨을 확인하였다. As a result, as shown in Fig. 14, similar to the experimental results of Example 4, double-stranded RNA not containing an innate immune activation motif did not induce an effective level of innate immune response, whereas in double-stranded RNA containing an innate immune activation motif, when at least 10 or more chemical modifications were introduced to either the first strand or the second strand, a high level of innate immune induction effect was exhibited. In particular, it was confirmed that this effect tended to be somewhat reduced in the ISG-KO-RIG-I cell line lacking RIG-I, similar to the experimental results of the previous examples, whereas it was lost or significantly reduced in the ISG-KO-MDA5 cell line lacking MDA5.
상기 결과로부터, 일 실시예에 따른 화학적 변형이 도입된 이중가닥 RNA의 선천성 면역 반응은, RIG-I외에도 MDA5가 주요 인자로서 매개하는 것임을 알 수 있었다. From the above results, it was found that the innate immune response to double-stranded RNA with chemical modification introduced according to one embodiment is mediated by MDA5 as a major factor in addition to RIG-I.
실시예 6. 인간 단핵구 세포주를 이용한 선천성 면역 반응 유도 효과 검증 Example 6. Verification of the effect of inducing innate immune response using human monocyte cell line
본 실시예에서는 인간 단핵구 세포주인 THP1-Dual(thpd-nfis) 세포주를 대상으로, 상기 실시예 4의 화학적 변형이 도입된 이중가닥 RNA의 처리에 따른 선천성 면역 반응 유도 효과를 재차 검증하였다. 본 실시예에서는 서열번호 2의 뉴클레오티드로 이루어지는 선천성 면역 활성화 서열이 제2 가닥의 5' 말단에 삽입된 30bp의 이중가닥 RNA를 기본 골격으로 하여, 뉴클레오티드의 2' 탄소 위치의 -OH기가 -OCH3(-O-methyl)로 치환된 화학적 변형이 도입된 이중가닥 RNA를 제조하였다. 구체적으로, 본 실시예에서 사용된 화학적 변형이 도입된 이중가닥 RNA의 정보는 상기 표 6, 표 7, 표 9에 나타낸 바와 같다. In this example, the effect of inducing an innate immune response according to the treatment of the double-stranded RNA with the chemical modification of Example 4 was verified again, using the human monocyte cell line THP1-Dual (thpd-nfis) cell line. In this example, a 30-bp double-stranded RNA in which an innate immune activation sequence consisting of nucleotides of SEQ ID NO: 2 is inserted at the 5' end of the second strand as a basic framework was prepared to introduce a chemical modification in which the -OH group at the 2' carbon position of the nucleotide is substituted with -OCH 3 (-O-methyl). Specifically, information on the double-stranded RNA with the chemical modification introduced used in this example is as shown in Tables 6, 7, and 9 above.
상기 실시예 2-3과 동일한 방식으로, 상기 THP1-Dual (thpd-nfis) 세포주에 이중가닥 RNA (10nM)를 Lipofectamine RNAiMAX (13778150; Invitrogen)를 사용하여 형질감염시키고, 상기 형질전환된 세포를 각각 24시간 또는 48시간 배양한 후에 상등액을 수득하였다. 이후, 상등액을 대상으로, 플레이트 판독기 (VICTORX2; PerkinElmer)를 사용하여, 인터페론 조절 인자 활성 수준을 검출하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다.In the same manner as in the above Example 2-3, double-stranded RNA (10 nM) was transfected into the THP1-Dual (thpd-nfis) cell line using Lipofectamine RNAiMAX (13778150; Invitrogen), and the transfected cells were cultured for 24 or 48 hours, respectively, and then the supernatant was obtained. Thereafter, the level of interferon regulatory factor activity was detected in the supernatant using a plate reader (VICTORX2; PerkinElmer). Meanwhile, in this example, the positive control group was a group administered with Poly I:C, and the control group was a group added with only the transfection reagent (mock) and a group not treated with double-stranded RNA (NT).
그 결과, 도 15에 나타낸 바와 같이, 일 실시예에 따른 선천성 면역 활성화 모티프를 포함하는 이중가닥 RNA에서, 적어도 10개 이상의 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 하나에 도입된 경우, 선천성 면역 반응의 증진 효과가 달성됨을 알 수 있었다.As a result, as shown in FIG. 15, it was found that in a double-stranded RNA including an innate immune activation motif according to one embodiment, when at least 10 chemical modifications were introduced to either the first strand or the second strand, the effect of enhancing the innate immune response was achieved.
실시예 7. 뉴클레오티드 길이에 따른 선천성 면역반응 유도 효과 확인Example 7. Confirmation of the effect of inducing innate immune response according to nucleotide length
본 실시예에서는, 상기 실시예 4-3, 5, 및 6에서 모두 높은 수준의 선천성 면역 유도 효과를 나타낸 30 bp 이중가닥 RNA(#1 5'-2-15)를 이용하여, 뉴클레오티드 전체 길이의 변형에 따른 선천성 면역 반응 유도 효과를 평가하였다. 이를 위하여, 상기 실시예 4-3, 5, 및 6의 30 bp 이중가닥 RNA(#1 5'-2-15)에서와 같이, 서열번호 2의 선천성 면역 활성화 서열이 제2 가닥의 5' 말단에 삽입되고, 뉴클레오티드 길이 전체에 대하여 50%의 화학적 변형이 제1 가닥에 도입된 이중가닥 RNA를 기본 골격으로 하여, 적어도 10개 이상의 화학적 변형을 포함하도록 뉴클레오티드의 길이를 20bp~40bp로 변경하였다 (#1 5'-2-10, #1 5'-2-11, #1 5'-2-12, #1 5'-2-13, #1 5'-2-15, #1 5'-2-20). 구체적으로, 전술한 화학적 변형이 도입된 20bp~40bp의 이중가닥 RNA는 표 11에 나타낸 바와 같다. 표 11에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다. In this example, the 30 bp double-stranded RNA (#1 5'-2-15), which showed a high level of innate immunity induction effect in all of the above Examples 4-3, 5, and 6, was used to evaluate the innate immune response induction effect according to the modification of the entire nucleotide length. To this end, as in the 30 bp double-stranded RNA of Examples 4-3, 5, and 6 (#1 5'-2-15), the innate immune activation sequence of SEQ ID NO: 2 was inserted at the 5' end of the second strand, and a double-stranded RNA in which 50% of the chemical modifications for the entire nucleotide length were introduced into the first strand was used as the basic backbone, and the length of the nucleotides was changed to 20 to 40 bp so as to include at least 10 or more chemical modifications (#1 5'-2-10, #1 5'-2-11, #1 5'-2-12, #1 5'-2-13, #1 5'-2-15, #1 5'-2-20). Specifically, the 20 to 40 bp double-stranded RNAs into which the above-mentioned chemical modifications were introduced are as shown in Table 11. In Table 11, innate immune activation motifs and innate immune activation sequences are shaded and/or bold, respectively, and nucleotides with 2'-OCH 3 (-O-methyl) chemical modifications are underlined.
[표 11][Table 11]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다. 그 결과를 도 16에 나타내었다. 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형이 도입되지 않은 30bp Lamin A/C-motif를 사용한 결과는 도 16에 Lamin A/C-motif로 표기하였고; 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-10이 도입된 20bp Lamin A/C-motif를 사용한 결과는 도 16에 20bp 2'-OMe Lamin A/C-motif로 표기하였고; 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-11이 도입된 22bp Lamin A/C-motif를 사용한 결과는 도 16에 22bp 2'-OMe Lamin A/C-motif로 표기하였고; 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-12이 도입된 24bp Lamin A/C-motif를 사용한 결과는 도 16에 24bp 2'-OMe Lamin A/C-motif로 표기하였고; 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-13이 도입된 26bp Lamin A/C-motif를 사용한 결과는 도 16에 26bp 2'-OMe Lamin A/C-motif로 표기하였고; 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Lamin A/C-motif를 사용한 결과는 도 16에 30bp 2'-OMe Lamin A/C-motif로 표기하였고; 상기 표 11에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-20이 도입된 40bp Lamin A/C-motif를 사용한 결과는 도 16에 40bp 2'-OMe Lamin A/C-motif로 표기하였다.Thereafter, the activity of IRF for RAW-Lucia ISG cell line was measured in the same manner as in Example 2-3. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT). The results are shown in Fig. 16. The result using 30 bp Lamin A/C-motif without 2'-OCH3(-O-methyl) chemical modification in Table 11 is indicated as Lamin A/C-motif in Fig. 16; The result using 20 bp Lamin A/C-motif with 2'-OCH3(-O-methyl) chemical modification #1 5'-2-10 introduced in Table 11 is indicated as 20 bp 2'-OMe Lamin A/C-motif in Fig. 16; The result using the 22 bp Lamin A/C-motif into which the 2'-OCH3(-O-methyl) chemical modification #1 5'-2-11 was introduced in Table 11 above is indicated as 22 bp 2'-OMe Lamin A/C-motif in FIG. 16; The result using the 24 bp Lamin A/C-motif into which the 2'-OCH3(-O-methyl) chemical modification #1 5'-2-12 was introduced in Table 11 above is indicated as 24 bp 2'-OMe Lamin A/C-motif in FIG. 16; The result using the 26 bp Lamin A/C-motif into which the 2'-OCH3(-O-methyl) chemical modification #1 5'-2-13 was introduced in Table 11 above is indicated as 26 bp 2'-OMe Lamin A/C-motif in FIG. 16; The results using the 30 bp Lamin A/C-motif introduced with the 2'-OCH3(-O-methyl) chemical modification #1 5'-2-15 in Table 11 above are indicated as 30 bp 2'-OMe Lamin A/C-motif in Figure 16; The results using the 40 bp Lamin A/C-motif introduced with the 2'-OCH3(-O-methyl) chemical modification #1 5'-2-20 in Table 11 above are indicated as 40 bp 2'-OMe Lamin A/C-motif in Figure 16.
그 결과, 도 16에 나타낸 바와 같이, 선천성 면역 활성화 모티프를 포함하고 적어도 10개의 2'-OCH3(-O-methyl) 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 한 가닥에 도입된 20bp-24bp의 이중 가닥 RNA는 선천성 면역 반응 모티프로부터 비롯된 효과, 즉, 선천성 면역 반응 유도 효과가 모두 소실되었다. 반면, 선천성 면역 활성화 모티프를 포함하고 적어도 10개의 2'-OCH3(-O-methyl) 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 한 가닥에 도입된 26bp-40bp의 이중가닥 RNA는 높은 수준의 선천성 면역 유도 효과를 나타내었다. As a result, as shown in FIG. 16, the 20-24 bp double-stranded RNA containing the innate immune activation motif and having at least 10 2'-OCH3(-O-methyl) chemical modifications introduced into either the first strand or the second strand completely lost the effect derived from the innate immune response motif, i.e., the innate immune response induction effect. In contrast, the 26-40 bp double-stranded RNA containing the innate immune activation motif and having at least 10 2'-OCH3(-O-methyl) chemical modifications introduced into either the first strand or the second strand exhibited a high level of innate immune induction effect.
상기 결과로부터, 일 실시예에 따른 선천성 면역 활성화 모티프를 포함하고 적어도 10개의 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 한 가닥에 도입된 이중가닥 RNA는, 전체 뉴클레오티드 길이 26bp-40bp의 범위에서, 선천성 면역 활성화 모티프 및 화학적 변형에 의한 효능이 발휘됨을 알 수 있었다. From the above results, it was found that the double-stranded RNA containing an innate immune activation motif according to one embodiment and having at least 10 chemical modifications introduced into either the first strand or the second strand exhibited efficacy due to the innate immune activation motif and chemical modifications in the range of a total nucleotide length of 26 bp to 40 bp.
실시예 8. 선천성 면역 활성화 모티프에 의한 선천성 면역반응 유도 효과 검증Example 8. Verification of the effect of inducing innate immune response by innate immune activation motif
8-1. 선천성 면역 활성화 모티프 유무에 따른 효능 평가8-1. Evaluation of efficacy according to presence or absence of innate immune activation motif
본 실시예에서는 인간 단핵구 세포주인 THP1-Dual(thpd-nfis) 세포주를 대상으로, 상기 실시예 7에서 높은 수준의 선천성 면역 유도 효과를 나타낸 30bp의 이중가닥 RNA를 이용하여 선천성 면역 활성화 모티프 유무에 따른 선천성 면역 반응 유도 효과를 평가하였다. 본 실시예에서는 상기 실시예 7의 30bp의 이중가닥 RNA(#1 5'-2-15)를 기본 골격으로 하여, 선천성 면역 활성화 모티프를 포함하거나 포함하지 않는 이중가닥 RNA를 제작하였다. 구체적으로, 전술한 선천성 면역 활성화 모티프 포함 유무에 따른 30bp의 이중가닥 RNA는 표 12에 나타낸 바와 같다. 표 12에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다.In this example, the 30 bp double-stranded RNA that showed a high level of innate immunity induction effect in Example 7 was used as the target for the human monocyte cell line, THP1-Dual (thpd-nfis), to evaluate the innate immune response induction effect according to the presence or absence of the innate immune activation motif. In this example, using the 30 bp double-stranded RNA (#1 5'-2-15) of the above Example 7 as a basic framework, a double-stranded RNA with or without an innate immune activation motif was produced. Specifically, the 30 bp double-stranded RNAs with or without the above-mentioned innate immune activation motif are as shown in Table 12. In Table 12, the innate immune activation motif and the innate immune activation sequence are indicated in shaded and/or bold, respectively, and the nucleotides introduced with a 2'-OCH 3 (-O-methyl) chemical modification are indicated in underline.
[표 12][Table 12]
이후, 상기 실시예 6과 동일한 방식으로, THP1-Dual (thpd-nfis) 세포주에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다. 그 결과를 도 17에 나타내었다. 상기 표 12에서 2'-OCH3(-O-methyl) 화학적 변형이 도입되지 않은 30bp Lamin A/C-GAPDH를 사용한 결과는 도 17에 Lamin A/C-GAPDH로 표기하였고; 상기 표 12에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Lamin A/C-GAPDH를 사용한 결과는 도 17에 2'-OMe Lamin A/C-GAPDH로 표기하였고; 상기 표 12에서 2'-OCH3(-O-methyl) 화학적 변형이 도입되지 않은 30bp Lamin A/C-Motif를 사용한 결과는 도 17에 Lamin A/C-Motif로 표기하였고; 상기 표 12에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Lamin A/C-Motif를 사용한 결과는 도 17에 2'-OMe Lamin A/C-Motif로 표기하였다.Thereafter, in the same manner as in Example 6, the activity of IRF for the THP1-Dual (thpd-nfis) cell line was measured. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT). The results are shown in Fig. 17. The result using 30 bp Lamin A/C-GAPDH without 2'-OCH3(-O-methyl) chemical modification in Table 12 is denoted as Lamin A/C-GAPDH in Fig. 17; The result using 30 bp Lamin A/C-GAPDH with 2'-OCH3(-O-methyl)
그 결과, 도 17에 나타낸 바와 같이, 선천성 면역 활성화 모티프를 포함하고 적어도 10개의 2'-OCH3(-O-methyl) 화학적 변형이 제1 가닥 또는 제2 가닥 중 어느 한 가닥에 도입된 30bp의 이중가닥 RNA는 인간 단핵구 세포주에서도 상기 실시예 7의 실험 결과와 마찬가지로, 높은 수준의 선천성 면역 유도 효과를 나타내었다. 반면, 상기와 동일한 구조를 지니되, 이중가닥 RNA의 말단에 상기 모티프가 도입되지 않은 30bp의 이중가닥 RNA (Lamin A/G-GAPDH)를 처리한 군에서는 유효한 수준의 선천성 면역 반응을 유도하지 못하였다. 따라서, 선천성 면역 유도 효과에 있어 선천성 면역 활성화 모티프의 도입은 필수적인 요소임을 재차 확인하였다.As a result, as shown in Fig. 17, the 30 bp double-stranded RNA containing the innate immune activation motif and having at least 10 2'-OCH3(-O-methyl) chemical modifications introduced into either the first or second strand exhibited a high level of innate immune induction effect in human monocyte cell lines, similar to the experimental result of Example 7. On the other hand, the group treated with the 30 bp double-stranded RNA (Lamin A/G-GAPDH) having the same structure as above but without the motif introduced into the end of the double-stranded RNA did not induce a significant level of innate immune response. Therefore, it was confirmed once again that the introduction of the innate immune activation motif is an essential element for the innate immune induction effect.
8-2. 변형된 선천성 면역 활성화 모티프의 적용에 따른 효능 평가8-2. Efficacy evaluation according to application of modified innate immune activation motif
본 실시예에서는 상기 실시예 7에서 높은 수준의 선천성 면역 유도 효과를 나타낸 30bp의 이중가닥 RNA를 기초로 선천성 면역 활성화 모티프의 길이를 변형함에 따른 선천성 면역 반응 유도 효과에의 영향을 확인하고자 하였다. 이를 위하여, 상기 실시예 7의 30bp의 이중가닥 RNA(#1 5'-2-15)를 기본 골격으로 하여, 서열번호 2의 선천성 면역 활성화 서열 대신 서열번호 8(20nt) 또는 서열번호 62 (30nt)의 선천성 면역 활성화 서열을 도입한 이중가닥 RNA를 추가적으로 제작하였다. 구체적으로, 서열번호 8(20nt) 및 서열번호 62(30nt)의 선천성 면역 활성화 서열은 표 13에 나타내었으며, 서열번호 8(20nt) 또는 서열번호 62(30nt)의 선천성 면역 활성화 서열을 제2 가닥의 5' 말단 영역에 포함하는 30bp의 이중가닥 RNA(Lamin A/C- Motif2, Motif3)는 표 14에 나타낸 바와 같다. 표 13 및 표 14에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다.In this example, we attempted to confirm the effect of modifying the length of the innate immune activation motif on the innate immune response induction effect based on the 30 bp double-stranded RNA that showed a high level of innate immune induction effect in the above Example 7. To this end, using the 30 bp double-stranded RNA (#1 5'-2-15) of the above Example 7 as the basic framework, we additionally produced a double-stranded RNA in which the innate immune activation sequence of SEQ ID NO: 8 (20 nt) or SEQ ID NO: 62 (30 nt) was introduced instead of the innate immune activation sequence of SEQ ID NO: 2. Specifically, the innate immune activation sequences of SEQ ID NO: 8 (20 nt) and SEQ ID NO: 62 (30 nt) are shown in Table 13, and the 30 bp double-stranded RNA (Lamin A/C- Motif2, Motif3) containing the innate immune activation sequence of SEQ ID NO: 8 (20 nt) or SEQ ID NO: 62 (30 nt) in the 5'-terminal region of the second strand are as shown in Table 14. In Tables 13 and 14, the innate immune activation motif and the innate immune activation sequence are shaded and/or bold, respectively, and the nucleotides introduced with the 2'-OCH 3 (-O-methyl) chemical modification are underlined.
[표 13][Table 13]
[표 14][Table 14]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다. 그 결과를 도 18에 나타내었다. 상기 표 14에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Lamin A/C-Motif를 사용한 결과는 도 18에 2'-OMe Lamin A/C-Motif 로 표기하였고; 상기 표 14에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Lamin A/C-Motif2를 사용한 결과는 도 18에 2'-OMe Lamin A/C-Motif2로 표기하였고; 상기 표 14에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Motif3을 사용한 결과는 도 18에 2'-OMe Motif3으로 표기하였다.Thereafter, the activity of IRF for RAW-Lucia ISG cell line was measured in the same manner as in Example 2-3. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT). The results are shown in Fig. 18. The result using 30 bp Lamin A/C-Motif into which 2'-OCH3(-O-methyl)
그 결과, 도 18에 나타낸 바와 같이, 서열번호 8의 뉴클레오티드 서열이 말단에 포함된 이중가닥 RNA 역시 상기 실시예 7과 동일하게 높은 수준의 선천성 면역 유도 효과를 나타내었으며, 서열번호 62의 뉴클레오티드 서열의 경우에도 우수한 선천성 면역 유도 효과를 나타내는 것을 확인하였다.As a result, as shown in Fig. 18, the double-stranded RNA including the nucleotide sequence of
따라서, 일 실시예에 따른 화학적 변형이 도입된 이중가닥 RNA는 변형된 선천성 면역 활성화 모티프에서도 유효한 선천성 면역 유도 효능을 발휘할 수 있는 것을 확인하였다.Therefore, it was confirmed that the double-stranded RNA with chemical modification introduced according to one embodiment can exhibit effective innate immune induction efficacy even in the modified innate immune activation motif.
실시예 9. 도입된 화학적 변형의 형태에 의한 선천성 면역반응 유도 효과 확인 Example 9. Confirmation of the effect of inducing innate immune response by the form of introduced chemical modification
본 실시예에서는 상기 실시예 7에서 높은 수준의 선천성 면역 유도 효과를 나타낸 30bp의 이중가닥 RNA를 기초로 도입된 화학적 변형의 형태에 따른 선천성 면역 반응 유도 효과에의 영향을 확인하고자 하였다. 이를 위하여, 상기 실시예 7의 30bp의 이중가닥 RNA(#1 5'-2-15)를 기본 골격으로 하여, 뉴클레오티드의 2' 탄소 위치의 -OH기가 -OCH3(-O-methyl) 대신 -F 로 치환된 화학적 변형이 도입된 이중가닥 RNA를 추가적으로 제작하였다. 구체적으로, 전술한 2'-F 화학적 변형이 도입된 30bp의 이중가닥 RNA는 표 15에 나타낸 바와 같다. 표 15에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 또는 2'-F 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였다.In this example, we attempted to confirm the influence of the type of chemical modification introduced based on the 30 bp double-stranded RNA showing a high level of innate immune induction effect in the above Example 7 on the innate immune response induction effect. To this end, using the 30 bp double-stranded RNA (#1 5'-2-15) of the above Example 7 as a basic framework, a double-stranded RNA was additionally produced in which a chemical modification was introduced in which the -OH group at the 2' carbon position of the nucleotide was substituted with -F instead of -OCH3(-O-methyl). Specifically, the 30 bp double-stranded RNA into which the above-mentioned 2'-F chemical modification was introduced is as shown in Table 15. In Table 15, the innate immune activation motif and the innate immune activation sequence are indicated in shaded and/or bold, respectively, and the nucleotides into which the 2'- OCH3 (-O-methyl) or 2'-F chemical modification was introduced are indicated in underline.
[표 15][Table 15]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주에 대한 IRF의 활성도를 측정하였다. 또한, 상기 실시예 6과 동일한 방식으로, THP1-Dual (thpd-nfis) 세포주에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다. RAW-Lucia ISG 세포주에 대한 결과를 도 19에 나타내었으며, THP1-Dual (thpd-nfis) 세포주에 대한 결과를 도 20에 나타내었다. 상기 표 15에서 2'-OCH3(-O-methyl)나 2'-F의 화학적 변형이 도입되지 않은 30bp Lamin A/C-motif를 사용한 결과는 도 19 및 도 20에 Lamin A/C-motif로 표기하였고; 상기 표 15에서 2'-OCH3(-O-methyl) 화학적 변형이 #1 5'-2-15 위치에 도입된 30bp Lamin A/C-Motif를 사용한 결과는 도 19 및 도 20에 2'-OMe Lamin A/C-Motif로 표기하였고; 상기 표 15에서 2'-F 화학적 변형 #1 5'-2-15이 도입된 30bp Motif를 사용한 결과는 도 19 및 도 20에 2'-F Lamin A/C-Motif로 표기하였다.Thereafter, the activity of IRF for the RAW-Lucia ISG cell line was measured in the same manner as in the above Example 2-3. In addition, the activity of IRF for the THP1-Dual (thpd-nfis) cell line was measured in the same manner as in the above Example 6. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT). The results for the RAW-Lucia ISG cell line are shown in Fig. 19, and the results for the THP1-Dual (thpd-nfis) cell line are shown in Fig. 20. The results using the 30 bp Lamin A/C-motif without chemical modification of 2'-OCH3(-O-methyl) or 2'-F in Table 15 are indicated as Lamin A/C-motif in Figs. 19 and 20; The results using the 30 bp Lamin A/C-Motif in which the 2'-OCH3(-O-methyl) chemical modification was introduced at
그 결과, 도 19에 나타낸 바와 같이, 2'-F 또는 2'-OCH3(-O-methyl) 화학적 변형이 도입된 이중가닥 RNA에서도 유효한 선천성 면역 반응 유도 효과가 나타났으며, 특히, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 이중가닥 RNA에서는 훨씬 더 높은 수준의 선천성 면역 유도 효과를 나타내는 것을 확인하였다. 또한, 이러한 경향성은 인간 단핵구 세포주를 사용한 도 20에서도 유사하게 나타나는 것을 확인하였다.As a result, as shown in Fig. 19, the double-stranded RNA into which 2'-F or 2'-OCH 3 (-O-methyl) chemical modifications were introduced exhibited an effective innate immune response induction effect, and in particular, it was confirmed that the double-stranded RNA into which 2'-OCH 3 (-O-methyl) chemical modifications were introduced exhibited a much higher level of innate immune induction effect. In addition, it was confirmed that this tendency was similarly shown in Fig. 20 using a human monocyte cell line.
실시예 10. 뉴클레오티드 결합의 변형에 따른 효능 평가Example 10. Evaluation of efficacy according to modification of nucleotide bond
본 실시예에서는 RNA 안정성 향상을 위해 뉴클레오티드 결합을 변형할 경우에도 선천성 면역 유도 효과가 유지될 수 있는지를 확인하고자 하였다. 이를 위하여, 상기 실시예 7에서 높은 수준의 선천성 면역 유도 효과를 나타낸 30bp의 이중가닥 RNA(#1 5'-2-15)를 기본 골격으로 하여, 적어도 하나의 가닥에서 양 말단으로부터 인접한 2개의 뉴클레오티드 결합, 즉, 1번째 뉴클레오티와 2번째 뉴클레오티드 사이 결합, 및 2번째 뉴클레오티드와 3번째 뉴클레오티드 사이의 결합이 포스포다이에스터 결합에서 포스포로티오에이트 결합으로 변형된 이중가닥 RNA를 추가적으로 제작하였다. 구체적으로, 전술한 뉴클레오티드 결합이 변형된 30bp의 이중가닥 RNA는 표 16에 나타낸 바와 같다. 표 16에서, 선천성 면역 활성화 모티프 및 선천성 면역 활성화 서열은 각각 음영 또는/및 진하게 표기하였고, 2'-OCH3(-O-methyl) 화학적 변형이 도입된 뉴클레오티드는 밑줄로 표기하였으며, 뉴클레오티드 결합이 포스포로티오에이트(phosphorothioate; PS) 결합으로 치환된 형태는 "*"로 표기하였다.In this example, we attempted to verify whether the innate immunity induction effect could be maintained even when the nucleotide bond was modified to improve RNA stability. To this end, using the 30 bp double-stranded RNA (#1 5'-2-15) that showed a high level of innate immunity induction effect in Example 7 as a basic framework, a double-stranded RNA was additionally produced in which two nucleotide bonds adjacent to each other from at least one strand, i.e., the bond between the 1st nucleotide and the 2nd nucleotide and the bond between the 2nd nucleotide and the 3rd nucleotide, were modified from phosphodiester bond to phosphorothioate bond. Specifically, the 30 bp double-stranded RNA in which the nucleotide bond described above was modified is as shown in Table 16. In Table 16, innate immune activation motifs and innate immune activation sequences are shaded and/or bold, respectively, nucleotides with 2'-OCH3(-O-methyl) chemical modifications are underlined, and forms in which nucleotide bonds are substituted with phosphorothioate (PS) bonds are marked with "*".
[표 16][Table 16]
이후, 상기 실시예 2-3과 동일한 방식으로, RAW-Lucia ISG 세포주에 대한 IRF의 활성도를 측정하였다. 한편, 본 실시예에서 양성 대조군은 Poly I:C를 투여한 군, 대조군은 형질 감염 시약만을 첨가한 군 (mock) 및 이중가닥 RNA를 처리하지 않은 군 (NT)을 사용하였다. 그 결과를 도 21에 나타내었다. 상기 표 16에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입된 30bp Lamin A/C-Motif를 사용한 결과는 도 21에 2'-OMe Lamin A/C-Motif 로 표기하였고; 상기 표 16에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입되고, 제1 가닥에 양 말단에 2개의 PS 결합이 도입된 30bp Lamin A/C-Motif(AS 2PS)를 사용한 결과는 도 21에 AS 2PS로 표기하였고; 상기 표 16에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입되고, 제2 가닥에 양 말단에 2개의 PS 결합이 도입된 30bp Lamin A/C-Motif(SS 2PS)를 사용한 결과는 도 21에 SS 2PS로 표기하였고; 상기 표 16에서 2'-OCH3(-O-methyl) 화학적 변형 #1 5'-2-15이 도입되고, 제1 가닥 및 제2 가닥 모두의 양 말단에 2개의 PS 결합이 도입된 30bp Lamin A/C-Motif(AS 2PS SS 2PS)를 사용한 결과는 도 21에 AS 2PS SS 2PS로 표기하였다.Thereafter, the activity of IRF for RAW-Lucia ISG cell line was measured in the same manner as in Example 2-3. Meanwhile, in this example, the positive control group was administered Poly I:C, and the control group was used as the group added only with the transfection reagent (mock) and the group not treated with double-stranded RNA (NT). The results are shown in Fig. 21. The result using the 30 bp Lamin A/C-Motif introduced with the 2'-OCH3(-O-methyl) chemical modification #1 5'-2-15 in Table 16 is indicated as 2'-OMe Lamin A/C-Motif in Fig. 21; The results using 30 bp Lamin A/C-Motif (AS 2PS) in which 2'-OCH3(-O-methyl) chemical modification #1 5'-2-15 was introduced in Table 16 above and two PS bonds were introduced at both ends in the first strand were denoted as AS 2PS in FIG. 21; The results using 30 bp Lamin A/C-Motif (SS 2PS) in which 2'-OCH3(-O-methyl) chemical modification #1 5'-2-15 was introduced in Table 16 above and two PS bonds were introduced at both ends in the second strand were denoted as SS 2PS in FIG. 21; The results using 30 bp Lamin A/C-Motif (AS 2PS SS 2PS) with 2'-OCH3(-O-methyl) chemical modification #1 5'-2-15 introduced in Table 16 above and two PS bonds introduced at both ends of both the first and second strands are denoted as AS 2PS SS 2PS in Figure 21.
그 결과, 도 21에 나타낸 바와 같이, 포스포로티오에이트(phosphorothioate; PS) 변형에도 불구하고, 선천성 면역 반응 유도 효과가 현저하게 유지되는 것을 확인하였으며, PS 결합이 제1 가닥에만 도입된 경우, 제2 가닥에만 도입된 경우, 제1 가닥 및 제2 가닥에 모두 도입된 경우를 불문하고, 모두 높은 수준의 선천성 면역 유도 효과를 나타내는 것을 확인하였다.As a result, as shown in Fig. 21, it was confirmed that the innate immune response induction effect was significantly maintained despite phosphorothioate (PS) modification, and it was confirmed that a high level of innate immune response induction effect was exhibited in all cases, regardless of whether the PS bond was introduced only to the first strand, only to the second strand, or both to the first and second strands.
실시예 11. 다양한 세포주에서의 선천성 면역반응 유도 효과 확인Example 11. Confirmation of the effect of inducing innate immune response in various cell lines
본 실시예에서는 상기 실시예 7에서 높은 수준의 선천성 면역 유도 효과를 나타낸 30bp의 이중가닥 RNA를 다양한 세포주에 처리하여 선천성 면역 반응 유도 효과를 재차 검증하였다. 마우스 세포주의 경우, RAW-Lucia ISG, EMT-6 또는 CT-26 세포주를 사용하여, Western blot으로 RIG-I, MDA5 단백질 발현 수준을 측정하고, RT-qPCR 방식으로 RIG-I, MDA5, IFIT1 mRNA 발현 수준을 측정하였다. 또한, 인간 세포주의 경우, THP1-Dual (thpd-nfis), THP, THP-PMA, PBMS, HCC-1143, SK-BR-3, HCC-70, HCC-38, BT-549, SK BR3, Hep3 B, SN12C, 또는 A498 세포주를 사용하여, RT-qPCR 방식으로 RIG-I, MDA5 mRNA 발현 수준을 측정하였다. 그 결과를 하기 표 17에 나타내었다.In this example, the 30 bp double-stranded RNA, which showed a high level of innate immunity induction effect in the above Example 7, was treated to various cell lines to re-verify the innate immune response induction effect. In the case of mouse cell lines, RAW-Lucia ISG, EMT-6, or CT-26 cell lines were used to measure the protein expression levels of RIG-I and MDA5 by Western blot, and the expression levels of RIG-I, MDA5, and IFIT1 mRNA by RT-qPCR. In addition, in the case of human cell lines, THP1-Dual (thpd-nfis), THP, THP-PMA, PBMS, HCC-1143, SK-BR-3, HCC-70, HCC-38, BT-549, SK BR3, Hep3 B, SN12C, or A498 cell lines were used to measure the mRNA expression levels of RIG-I and MDA5 by RT-qPCR. The results are shown in Table 17 below.
[표 17][Table 17]
그 결과, 상기 표 17에 나타난 바와 같이, 상기 실시예 7의 30bp의 이중가닥 RNA를 적용한 경우, 인간 면역세포인 THP, PMA 처리하여 분화시킨 THP (THP-PMA), 및 PBMC를 비롯하여, 유방, 간, 및 신장 암세포주에서도 MDA 및 RIG-Ⅰ 매개 선천성 면역 반응이 유효하게 유도될 수 있음을 확인하였다.As a result, as shown in Table 17 above, when the 30 bp double-stranded RNA of Example 7 was applied, it was confirmed that MDA and RIG-I-mediated innate immune responses could be effectively induced in human immune cells such as THP, THP differentiated by PMA treatment (THP-PMA), and PBMC, as well as breast, liver, and renal cancer cell lines.
이상으로 본 발명 내용의 특정한 부분을 상세히 기술하였는 바, 당업계의 통상의 지식을 가진 자에게 있어서 이러한 구체적 기술은 단지 바람직한 실시 양태일 뿐이며, 이에 의해 본 발명의 범위가 제한되는 것이 아닌 점은 명백할 것이다. 따라서, 본 발명의 실질적인 범위는 첨부된 청구항들과 그것들의 등가물에 의하여 정의된다고 할 것이다.While the specific parts of the present invention have been described in detail above, it will be apparent to those skilled in the art that such specific descriptions are merely preferred embodiments and that the scope of the present invention is not limited thereby. Accordingly, the substantial scope of the present invention will be defined by the appended claims and their equivalents.
Claims (23)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2023-0030816 | 2023-03-08 | ||
| KR20230030816 | 2023-03-08 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024186166A1 true WO2024186166A1 (en) | 2024-09-12 |
Family
ID=92675354
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2024/003026 Ceased WO2024186166A1 (en) | 2023-03-08 | 2024-03-08 | Double-stranded rna having innate immune response-inducing effect, and use thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024186166A1 (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20170369882A1 (en) * | 2015-04-03 | 2017-12-28 | University Of Massachusetts | Fully stabilized asymmetric sirna |
| KR20180002528A (en) * | 2016-06-29 | 2018-01-08 | 올릭스 주식회사 | Compounds improving RNA interference of small interfering RNA and use thereof |
| US20180085432A1 (en) * | 2008-08-04 | 2018-03-29 | University Of Miami | STING (Stimulator of Interferon Genes), A Regulator of Innate Immune Responses |
| KR20190108167A (en) * | 2017-02-10 | 2019-09-23 | 성균관대학교산학협력단 | Long double stranded RNA for RNA interference |
-
2024
- 2024-03-08 WO PCT/KR2024/003026 patent/WO2024186166A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20180085432A1 (en) * | 2008-08-04 | 2018-03-29 | University Of Miami | STING (Stimulator of Interferon Genes), A Regulator of Innate Immune Responses |
| US20170369882A1 (en) * | 2015-04-03 | 2017-12-28 | University Of Massachusetts | Fully stabilized asymmetric sirna |
| KR20180002528A (en) * | 2016-06-29 | 2018-01-08 | 올릭스 주식회사 | Compounds improving RNA interference of small interfering RNA and use thereof |
| KR20190108167A (en) * | 2017-02-10 | 2019-09-23 | 성균관대학교산학협력단 | Long double stranded RNA for RNA interference |
Non-Patent Citations (1)
| Title |
|---|
| ZHONGJI MENG, MENGJI LU: "RNA Interference-Induced Innate Immunity, Off-Target Effect, or Immune Adjuvant?", FRONTIERS IN IMMUNOLOGY, vol. 8, XP055750431, DOI: 10.3389/fimmu.2017.00331 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2013103249A1 (en) | High-efficiency nanoparticle-type double-helical oligo-rna structure and method for preparing same | |
| WO2011056005A2 (en) | Novel sirna structure for minimizing off-target effects caused by antisense strands, and use thereof | |
| WO2011071232A1 (en) | Method for diagnosis/prognosis of cancers using an epigenetic marker consisting of a specific single cpg site in ttp promoter and treatment of cancers by regulating its epigenetic status | |
| WO2024210530A1 (en) | RNAi AGENTS TARGETING LPA GENE AND USE THEREOF | |
| WO2016018088A1 (en) | Novel biomarker for predicting sensitivity to met inhibitor, and use thereof | |
| WO2022191431A1 (en) | Nanoliposome carrier composition having kras and p53 gene editing function | |
| WO2019225968A1 (en) | Amphiregulin gene-specific double-stranded oligonucleotide and composition, for preventing and treating fibrosis-related diseases and respiratory diseases, comprising same | |
| WO2019194586A1 (en) | Novel target for anti-cancer and immune-enhancing | |
| WO2022216096A1 (en) | Novel target for anti-cancer effect and immunity enhancement | |
| WO2024186166A1 (en) | Double-stranded rna having innate immune response-inducing effect, and use thereof | |
| WO2018155890A1 (en) | Asymmetric sirna for inhibiting expression of male pattern hair loss target gene | |
| WO2023096126A1 (en) | Method for screening mdsc inhibitor | |
| WO2020235936A1 (en) | Asymmetric sirna inhibiting expression of pd-1 | |
| WO2017171373A2 (en) | Composition for suppressing resistance to egfr-targeting agent | |
| WO2024177416A1 (en) | Pharmaceutical composition for treating egfr targeted therapy-resistant cancer | |
| WO2023282704A1 (en) | Rnai agent targeting marc1 gene, and use thereof | |
| WO2019066519A1 (en) | Pharmaceutical composition for prevention or treatment of age-related macular degeneration containing rna complex targeting connective tissue growth factor | |
| RU2636003C2 (en) | Composition for treatment of cancer associated with hpv infection | |
| WO2017188707A1 (en) | Dicer substrate rna nanostructures with enhanced gene silencing effect and preparation method thereof | |
| WO2022145949A2 (en) | Composition comprising tead4 inhibitor for preventing or treating colorectal cancer | |
| WO2023163497A1 (en) | Pharmaceutical composition for preventing or treating colorectal cancer comprising ednra inhibitor as active ingredient | |
| WO2019245269A1 (en) | Composition comprising flt3 inhibitor as effective ingredient for inhibiting drug resistance in chronic myelogenous leukemia | |
| WO2023043239A1 (en) | Double-stranded rna having innate immune response-inducing effect, and use thereof | |
| WO2022131883A1 (en) | Rnai agent for inhibiting hbv expression and use thereof | |
| WO2020116688A1 (en) | Anticancer supplement comprising nc886 and/or pkr inhibitor, and method for providing information of drug for cancer treatment |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24767453 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |