WO2023201267A1 - Combination therapy for treating trop-2 expressing cancers - Google Patents
Combination therapy for treating trop-2 expressing cancers Download PDFInfo
- Publication number
- WO2023201267A1 WO2023201267A1 PCT/US2023/065682 US2023065682W WO2023201267A1 WO 2023201267 A1 WO2023201267 A1 WO 2023201267A1 US 2023065682 W US2023065682 W US 2023065682W WO 2023201267 A1 WO2023201267 A1 WO 2023201267A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cancer
- antibody
- inhibitor
- therapy
- trop
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/68037—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being a camptothecin [CPT] or derivatives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7052—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides
- A61K31/706—Compounds having saccharide radicals and heterocyclic rings having nitrogen as a ring hetero atom, e.g. nucleosides, nucleotides containing six-membered rings with nitrogen as a ring hetero atom
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/3955—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against proteinaceous materials, e.g. enzymes, hormones, lymphokines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39533—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals
- A61K39/39558—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum against materials from animals against tumor tissues, cells, antigens
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6855—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from breast cancer cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6861—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from kidney or bladder cancer cell
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6851—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell
- A61K47/6869—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a determinant of a tumour cell the tumour determinant being from a cell of the reproductive system: ovaria, uterus, testes, prostate
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2818—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against CD28 or CD152
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
- A61K2039/507—Comprising a combination of two or more separate antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/54—Medicinal preparations containing antigens or antibodies characterised by the route of administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2300/00—Mixtures or combinations of active ingredients, wherein at least one active ingredient is fully defined in groups A61K31/00 - A61K41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/24—Immunoglobulins specific features characterized by taxonomic origin containing regions, domains or residues from different species, e.g. chimeric, humanized or veneered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
Definitions
- the present disclosure relates to methods of treating, mitigating, or preventing or delaying the recurrence or metastasis of a Trop-2 expressing cancer (c.g., metastatic castrate resistant prostate cancer, non-small cell lung cancer) in a subject by administering an effective amount of: (a) an anti-Trop-2 antibody drug conjugate (ADC) (e.g., sacituzumab govitecan); and (b) an adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat) to the subject.
- ADC anti-Trop-2 antibody drug conjugate
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- the present disclosure further relates to methods of treating, mitigating, or preventing or delaying the recurrence or metastasis of a tumor antigen (TA) expressing cancer in a subject by administering an effective amount of: a) a tumor antigen targeted antibody-drug conjugate (ADC) comprising a topoisomerase I inhibitor (Topi ADC); and b) an adenosine pathway inhibitor.
- ADC tumor antigen targeted antibody-drug conjugate
- Topici ADC topoisomerase I inhibitor
- Anti-Trop-2 antibody drug conjugates such as sacituzumab govitecan and datopotamab deruxtecan, are under clinical investigation for the treatment of a variety of Trop-2 expressing cancers. While evidence of clinical efficacy has been obtained in a monotherapy setting, further therapeutic benefits for patients are desired.
- Trop-2 expression has been reported for a variety of epithelial cancers, including breast, bladder, lung, colorectal and prostate cancers.
- Prostate cancer is the most commonly diagnosed cancer and the second most frequent cause of cancer-related mortality among men in the United States (US).
- US United States
- Adenocarcinoma comprises 95% of prostatic carcinomas (Lie AK. Histology of prostate cancer. Oncolex Oncology Encyclopedia. Accessed 25 February 2020 at oncolex.org/Prostate-cancer/Background/Histology.).
- Most cases (90%) are diagnosed at a local or regional stage for which treatment options include active surveillance, surgery, or radiation.
- the 5-ycar relative survival is 98% across all stages but is 30% for disease diagnosed at an advanced stage.
- Metastatic or advanced prostate cancer has a more complex treatment algorithm. Patients with locally advanced disease initially receive androgen ablation with radiation or prostatectomy, whereas first-line therapy in the metastatic setting is androgen ablation plus systemic treatment (e.g., second-generation hormonal therapy or docetaxel-based chemotherapy).
- Disease progression in the context of androgen deprivation is known as castrate resistant prostate cancer (CRPC).
- PCWG2 Prostate Cancer Working Group 2
- PCWG2 Prostate Cancer Working Group 2
- PSA serum prostate-specific antigen
- the progression of pre-existing local or metastatic disease and/or the appearance of new metastases in the setting of castrate levels of serum testosterone ( ⁇ 50 ng/dL; Scher HI, et al. J Clin Oncol. (2008) 26(7): 1148- 1159.).
- Prognosis for CRPC is associated with several factors, including performance status, presence of bone pain, extent of disease on bone scan, and serum alkaline phosphatase (ALP) levels. Bone metastases will occur in approximately 80% of men with CRPC and can produce significant morbidity, including pain, pathologic fractures, spinal cord compression, and bone marrow failure. Paraneoplastic effects are also common, including anemia, weight loss, fatigue, hypercoagulability, and increased susceptibility to infection (Saad F and Hotte S. Can Urol Assoc J. (2010) 4(6):380-384).
- Lung cancer is the leading cause of cancer-related mortality worldwide. It was estimated that in 2020, there were over 2 million new cases of lung cancer and approximately 1.8 million deaths worldwide. In the United States (US) in 2021, it is estimated that there will be over 235,000 new cases of lung cancer and over 131,000 deaths. Approximately 80% to 85% of all lung cancers are non-small cell lung cancer (NSCLC) (Ettinger et al. J Natl Compr Cane Netw (2019); 17(12): 1464-72) and more than half of these arc identified at an advanced stage (Siegel ct al. CA Cancer J Clin (2019);69 (l):7-34).
- NSCLC non-small cell lung cancer
- EGFR epidermal growth factor receptor
- ALK anaplastic lymphoma kinase
- ROS1 ROS proto-oncogene 1
- BRAF proto-oncogene B-raf
- RET alterations neurotrophic tyrosine receptor kinase
- NSRK neurotrophic tyrosine receptor kinase
- Chemotherapy /immunotherapy regimens such as pembrolizumab/(carboplatin or cisplatin)/pemetrexed are recommended for patients with non-squamous NSCLC regardless of PD-L1 expression.
- chemotherapy/immunotherapy regimens such as pembrolizumab/carboplatin/ (paclitaxel or albumin-bound paclitaxel) are recommended (Ettinger et al. J Natl Compr Cane Netw (2019);17 (12): 1464-72).
- Immune checkpoint inhibitors have reinvigorated clinical development interest in anticancer immunotherapy. The latter relies on therapeutic modulation of the tumor microenvironment or other aspects of the immune system to overcome mechanisms of immune suppression that a tumor elicits on the host immune system. Despite their proven benefit in numerous tumor types as evidenced by the approvals of nivolumab, pembrolizumab, atezolizumab, and ipilimumab, checkpoint inhibition has not proven effective in the treatment of metastatic prostate cancer where response rates to monotherapy are less than 5% (Antonarakis ES, et al. J Clin Oncol. (2020) 38(5):395-405.).
- ADCs Antibody-drug conjugates
- ADCs are a fast-growing drug class in oncology with several different ADCs currently approved as cancer treatments and many more in preclinical and clinical development (e.g., Drago, J.Z. et al., Nat Rev Clin Oncol (2021) 18, 327-344).
- ADCs are generally composed of monoclonal antibodies linked to cytotoxic drugs (payloads).
- ADCs are designed to limit the delivery of cytotoxic drugs specifically to cells expressing the target antigen of the respective antibody (e.g., Trop-2, Her-2, Nectin-4) and immediately surrounding tumor tissue (bystander effect).
- ADCs As a result of their tumor targeted delivery of cytotoxic agents ADCs often have much improved therapeutic windows compared to systemically administered cytotoxic agents.
- ADCs that have obtained marketing approval by the U.S. Food and Drug Administration (US FDA) include gemtuzumab ozogamicin, brentuximab vedotin, ado- trastuzumab emtansine (T-DM1), inotuzumab ozogamicin, trastuzumab deruxtecan (T-DXd), polatuzumab vcdotin, sacituzumab deruxtecan, enfortumab vedotin, and bclantamab mafodotin.
- Adenosine pathway mediates immunosuppressive effects in the tumor microenvironment (e.g., Allard, B., et al. Nat Rev Clin Oncol (2020) 17, 611-629).
- Adenosine pathway inhibitors including various CD39 inhibitors, CD73 inhibitors, and adenosine receptor antagonists, are under clinical investigation as promising agents stimulating anti-cancer immune responses.
- a Trop-2 positive cancer comprising co-administering to a subject an effective amount of: a) an anti-Trop-2 antibody-drug conjugate (ADC); and b) an adenosine pathway inhibitor.
- ADC anti-Trop-2 antibody-drug conjugate
- the methods provided herein are for treating a Trop-2 positive cancer comprising co-administering to a subject an effective amount of: a) an anti-Trop-2 antibody-drug conjugate (ADC); and b) an adenosine pathway inhibitor.
- ADC anti-Trop-2 antibody-drug conjugate
- the anti-Trop-2 ADC comprises a topoisomerase I inhibitor.
- the topoisomerase I inhibitor is a camptothecin (CPT).
- the topoisomerase I inhibitor is a topotecan, irinotecan, belotecan or exatecan.
- the topoisomerase I inhibitor is SN38 or Dxd.
- the topoisomerase I inhibitor is selected from the group consisting of irinotecan, topetecan, and SN-38.
- the topoisomerase I inhibitor is SN38.
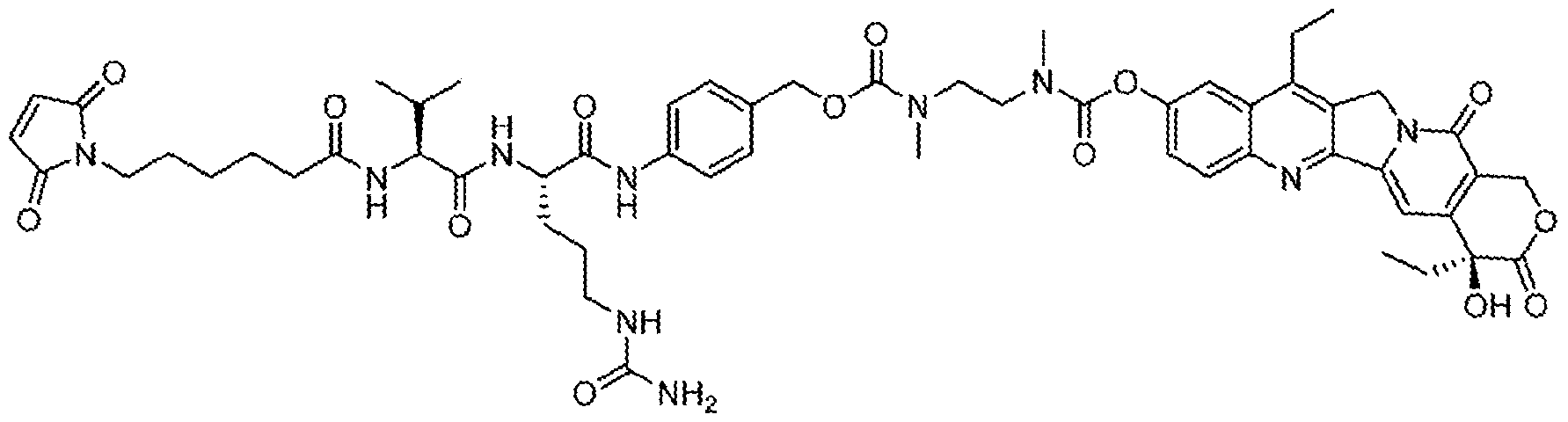
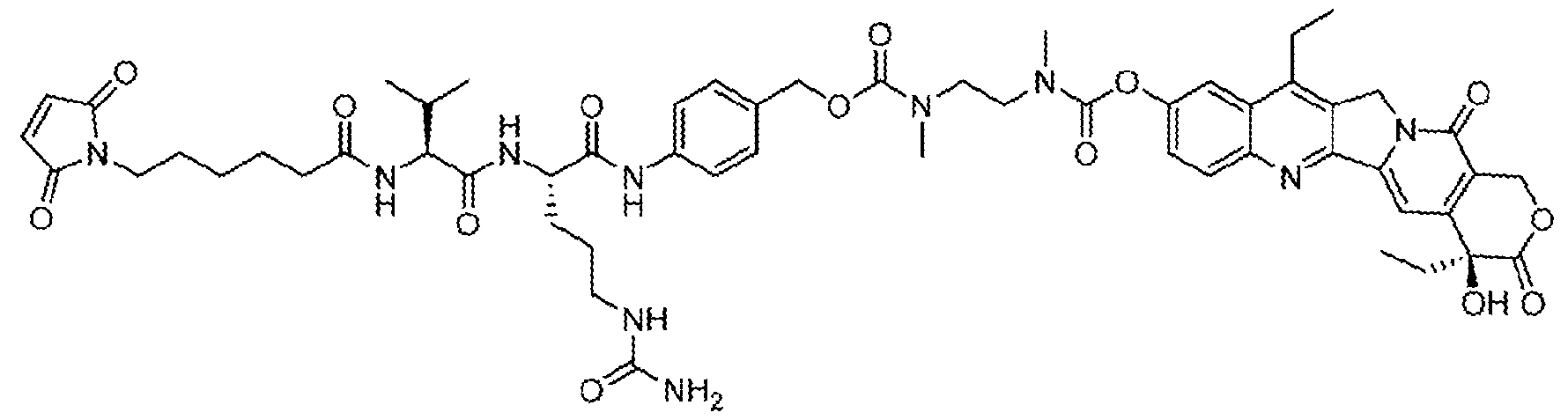
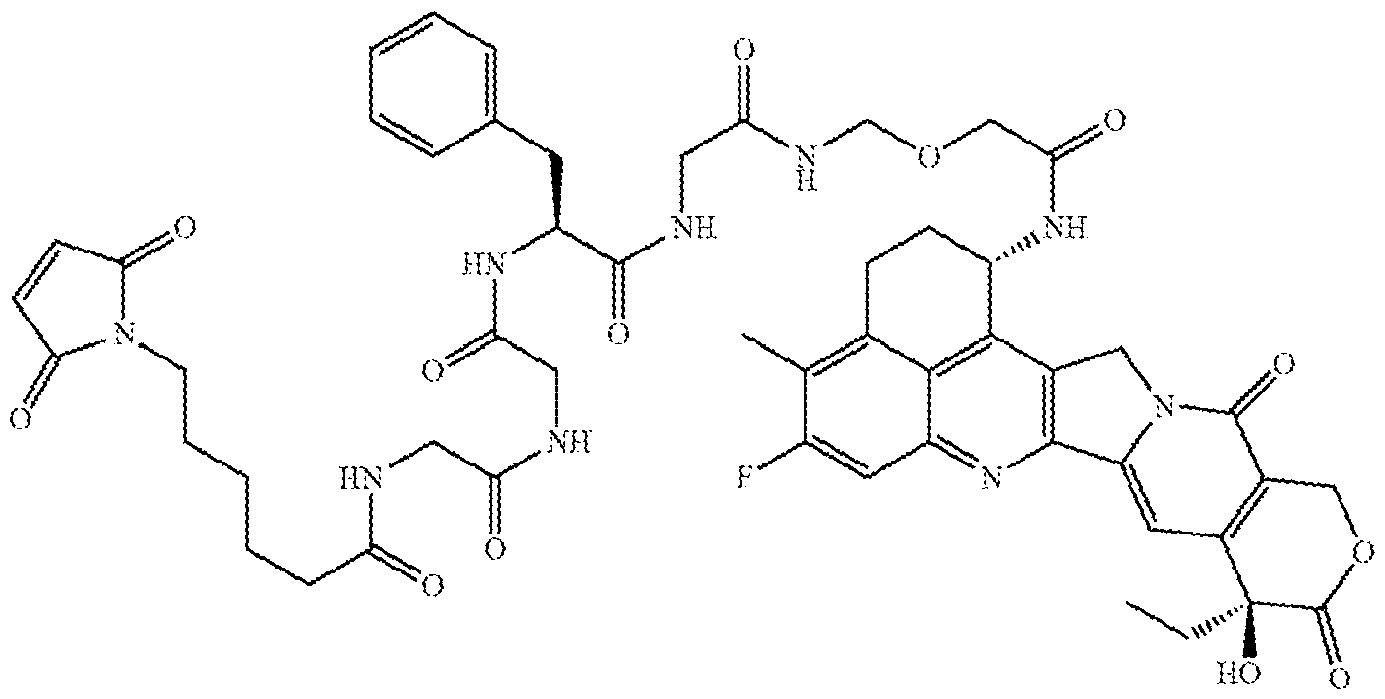
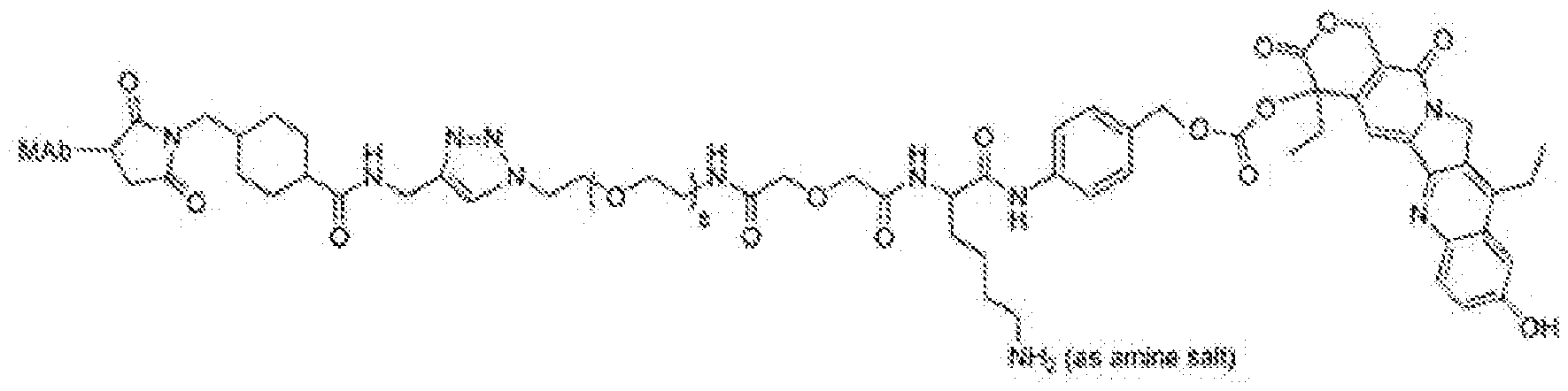
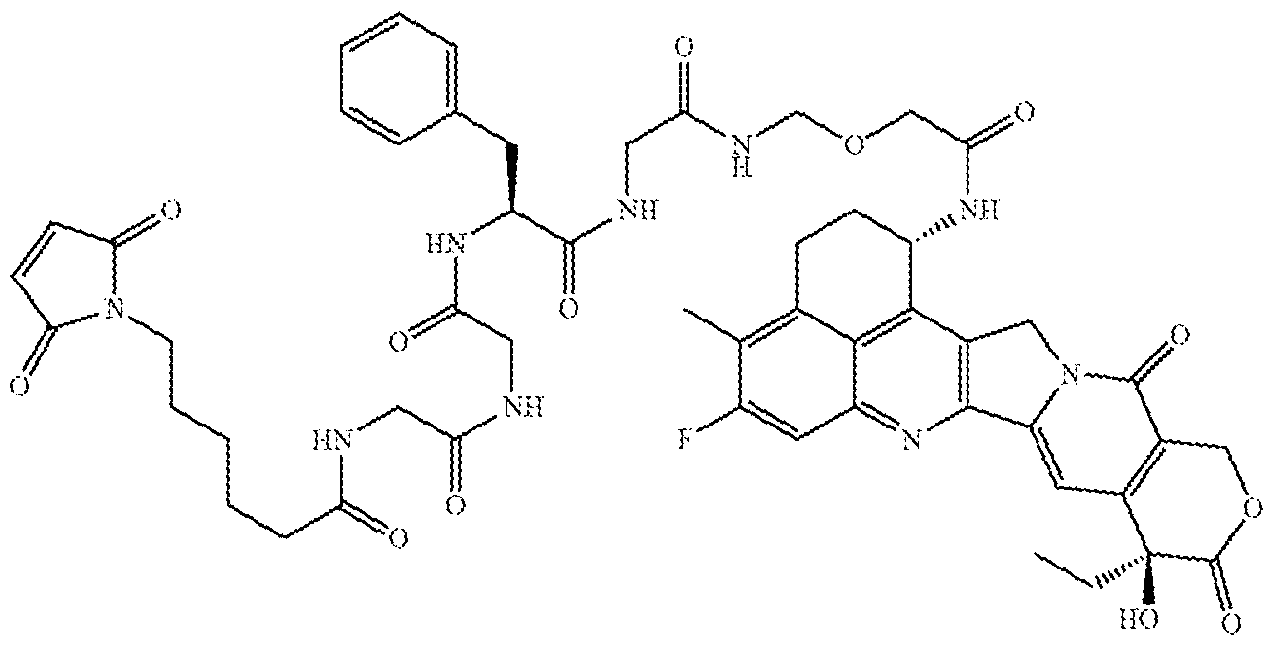
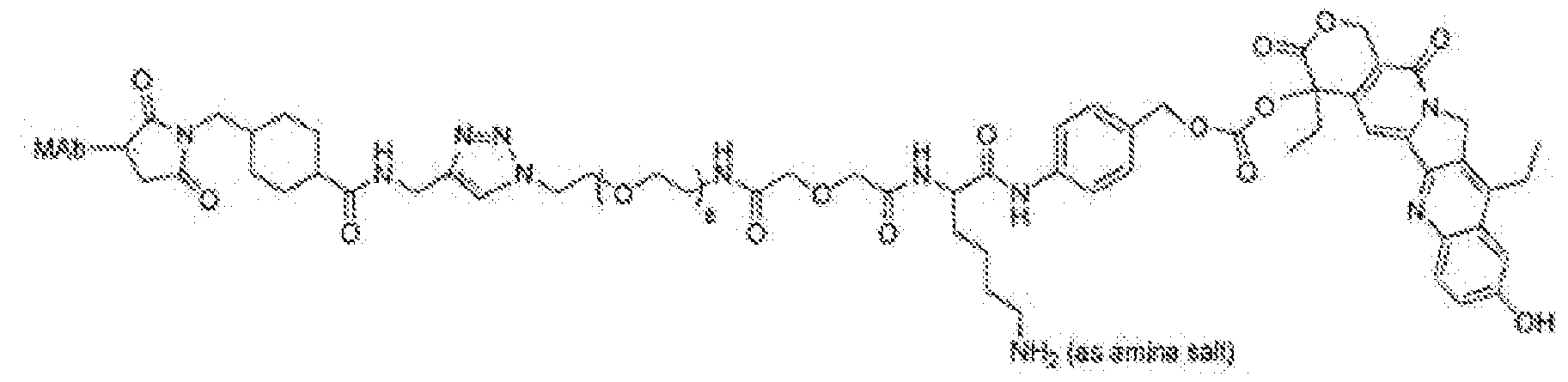
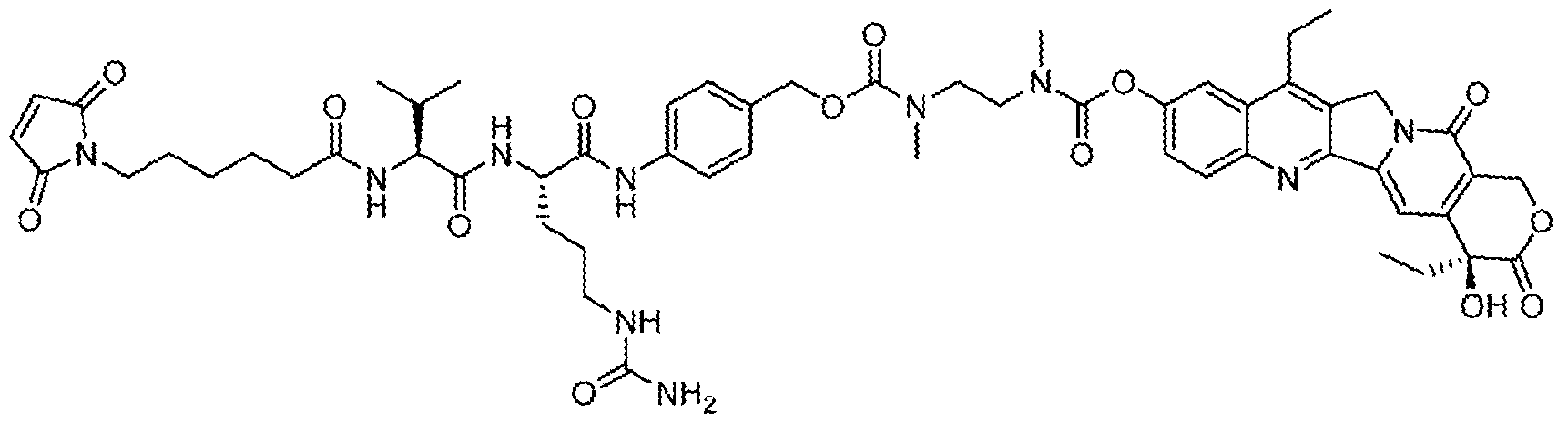
- the anti-Trop-2 ADC has a structural formula of mAb-CL2A-SN- 38, with a structure represented by:
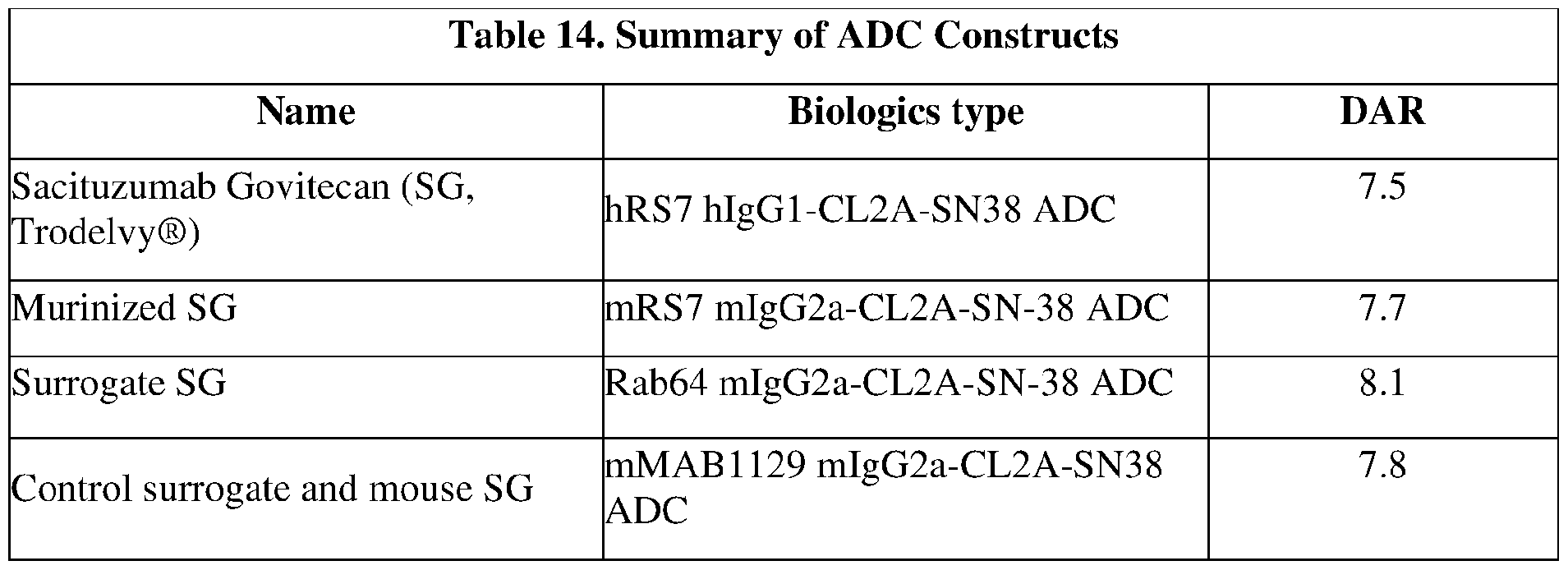
- the anti-Trop-2 ADC comprises sacituzumab (hRS7; described, e.g., in W02003074566, Figures 3 and 4).
- the anti-Trop-2 ADC is selected from the group consisting of sacituzumab govitecan, datopotamab deruxtecan (DS-1062), ESG-401, SKB-264, DAC-02 and BAT-8003.
- the anti-Trop-2 ADC is sacituzumab govitecan.
- the adenosine pathway inhibitor is a CD39 inhibitor, a CD73 inhibitor, or an adenosine receptor antagonist.
- the adenosine pathway inhibitor is a plurality of adenosine pathway inhibitors.
- the plurality of adenosine pathway inhibitors comprises a CD73 inhibitor and an adenosine receptor antagonist.
- the CD73 inhibitor is quemliclustat and the adenosine receptor antagonist is etrumadenant.
- the adenosine pathway inhibitor is an adenosine receptor antagonist.
- the adenosine pathway inhibitor is imaradenant, NIR178, ID11902, IN-A003, NTI-55, TT-10, TT-228, PBF-1129 (Palobiofarma), TT-702, etrumadenant, INCB 106385, M1069, HM87277, RVU-330, or TT-53.
- the adenosine pathway inhibitor is a dual antagonist of adenosine A2A receptor (A2AR; ADORA2A) and A2B receptor (A2BR; ADORA2B).
- A2AR adenosine A2A receptor
- A2BR A2B receptor
- the adenosine pathway inhibitor is etrumadenant (AB928; GS- 0928), taminadcnant, TT-10, TT-4, or M1069.
- the adenosine pathway inhibitor is etrumadenant.
- the adenosine pathway inhibitor is a CD73 inhibitor.
- the CD73 inhibitor is oleclumab, B MS-986179, uliledlimab, AK119, quemliclustat, mupadolimab, HLX23, INCA00186, IB 1325, NZV930, ORIC-533, Sym024, IPH5301, IOA-237, JAB-BX100, PT199, TRB010, CD73 ASO, ABSK-051, AK131, BR101, BP1200, CB708, GB7002, or ATG-037.
- the CD73 inhibitor is quemliclustat (AB680, GS-0680), uliledlimab, mupadolimab, ORIC-533, ATG-037, PT-199, AK131, NZV93O, BMS-986179, or oleclumab.
- the CD73 inhibitor is quemliclustat.
- the method further comprises co-administering an additional therapeutic agent or therapeutic modality.
- the additional therapeutic agent or therapeutic modality comprises one, two, three, or four additional therapeutic agents and/or therapeutic modalities.
- the additional therapeutic agent comprises an anti-PD-(L)l antibody.
- the anti-PD-(L)l antibody is pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, or zimberelimab.
- the anti-PD-(L)l antibody is zimberelimab.
- the additional therapeutic agent comprises an anti-TIGIT antibody.
- the anti-TIGIT antibody is tiragolumab, vibostolimab, domvanalimab, AB3O8, AK127, BMS-986207, ralzapastotug, or etigilimab.
- the anti-TTGTT antibody is domvanalimab.
- the anti-TIGIT antibody is ralzapastotug.
- the additional therapeutic agent comprises a) zimberelimab and domvanalimab, b) zimberelimab and AB308, c) atezolizumab and tiragolumab, d) pembrolizumab and vibostolimab, e) pembrolizumab and domvanalimab, f) pembrolizumab and AB308, g) MK- 7684A (pembrolizumab/vibostolimab coformulation), h) durvalumab and domvanalimab, i) zimberelimab and ralzapastotug, or j) pembrolizumab and ralzapastotug.
- the additional therapeutic agent comprises zimberelimab and domvanalimab.
- the Trop-2 positive cancer is a solid epithelial cancer.
- the solid epithelial cancer is selected from breast cancer (e.g., triple negative breast cancer (TNBC), HR + /Her2- breast cancer, HR + /Her2 low breast cancer), colorectal cancer, lung cancer, stomach cancer, urinary tract cancer, urothelial cancer, bladder cancer, renal cancer, pancreatic cancer, ovarian cancer, uterine cancer, esophageal cancer and prostatic cancer.
- breast cancer e.g., triple negative breast cancer (TNBC), HR + /Her2- breast cancer, HR + /Her2 low breast cancer
- colorectal cancer e.g., lung cancer, stomach cancer, urinary tract cancer, urothelial cancer, bladder cancer, renal cancer, pancreatic cancer, ovarian cancer, uterine cancer, esophageal cancer and prostatic cancer.
- the breast cancer is triple negative breast cancer (TNBC), HR + /Her2’ breast cancer, or HR + /Her2 low breast cancer.
- the prostatic cancer is castrate-resistant prostate cancer (CRPC).
- CRPC castrate-resistant prostate cancer
- the lung cancer is non-small lung cancer (NSCLC).
- NSCLC non-small lung cancer
- the lung cancer is (i) squamous NSCLC or (ii) non-squamous NSCLC.
- the lung cancer is without EGFR, ALK, or other actionable genomic alterations.
- the Trop-2 positive cancer is (i) unresectable, locally advanced or (ii) metastatic.
- the cancer has progressed following at least one prior anti-cancer therapy.
- Tn some embodiments, the cancer has progressed after prior new hormonal agent treatment (NHA; first- or second-generation non-steroidal antiandrogens, abiratcronc).
- NHA first- or second-generation non-steroidal antiandrogens, abiratcronc
- the cancer has progressed or recurred after platinum-based chemotherapy.
- the cancer has progressed or recurred after checkpoint inhibitor therapy (CPI) therapy.
- CPI checkpoint inhibitor therapy
- the cancer has progressed or recurred after platinum-based chemotherapy and anti-PD-(L)l antibody therapy, received either in combination or sequentially in any order.
- the cancer has progressed or recurred after a tyrosine kinase inhibitor therapy.
- the subject is treatment naive.
- the subject has not received a prior therapy selected from the group consisting of a taxane therapy (taxane naive), checkpoint inhibitor therapy (CPI naive), and topoisomerase I inhibitor therapy.
- a prior therapy selected from the group consisting of a taxane therapy (taxane naive), checkpoint inhibitor therapy (CPI naive), and topoisomerase I inhibitor therapy.
- the subject has not received a prior taxane therapy (taxane naive), checkpoint inhibitor therapy (CPI naive), or topoisomerase I inhibitor therapy.
- taxane naive prior taxane therapy
- CPI naive checkpoint inhibitor therapy
- topoisomerase I inhibitor therapy topoisomerase I inhibitor therapy
- the taxane therapy comprises paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, or cabazitaxel.
- the checkpoint inhibitor therapy comprises an anti-CTLA4 antibody or an anti-PD(L)l antibody.
- the topoisomerase I inhibitor therapy comprises a topotecan, irinotecan, belotecan or exatecan.
- the anti-Trop-2 ADC and the adenosine pathway inhibitor are coadministered concurrently.
- the anti-Trop-2 ADC and the adenosine pathway inhibitor are coadministered sequentially.
- the subject is human.
- the anti-Trop-2 ADC is administered at one or more doses in the range of 8 mg/kg to 10 mg/kg.
- the anti-Trop2 ADC is administered at one or more doses of 8 mg/kg or 10 mg/kg.
- the anti-Trop2 ADC is administered at one or more doses of 10 mg/kg.
- the anti-Trop-2 ADC is administered intravenously.
- the anti-Trop-2 ADC is administered on days 1 and 8 of a 21-day cycle.
- the adenosine pathway inhibitor is administered at one or more doses of 75 mg or 150 mg.
- the adenosine pathway inhibitor is administered at one or more doses of 150 mg.
- the adenosine pathway inhibitor is administered orally (PO).
- the adenosine pathway inhibitor is administered once daily (QD).
- the anti-PD(L)l antibody is administered at one or more doses of 360 mg.
- the anti-PD(L)l antibody is administered intravenously (IV).
- the anti-PD(L)l antibody is administered once every three weeks (Q3W).
- sacituzumab govitecan is administered intravenously (IV) at a dose of 10 mg/kg on day 1 and day 8 of a 21 -day treatment cycle
- etrumadenant is administered orally (PO) once a day (QD) at a dose of 150 mg on each day of the 21-day treatment cycle
- zimberelimab is administered intravenously (IV) on day 1 of the 21-day treatment cycle (Q3W).
- an anti-cancer effect is observed as determined by objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), PSA response rate, radiographic response rate, change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers, or a combination thereof.
- ORR objective response rate
- DCR disease control rate
- PFS progression free survival
- DOR duration of response
- OS overall survival
- CR complete response
- PR partial response
- PSA response rate radiographic response rate
- change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers or a combination thereof.
- tumor response or progression is determined according to RECIST version 1.1.
- a method of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of castrate-resistant prostate cancer comprising co-administering to a human patient an effective amount of a) sacituzumab govitecan; and b) etrumadenant.
- CRPC castrate-resistant prostate cancer
- Tn some embodiments, provided herein is a method of treating metastatic castrate-resistant prostate cancer (mCRPC) comprising co-administering to a human patient an effective amount of a) sacituzumab govitecan; and b) etrumadenant.
- the method further comprises co-administering an anti-PD-(L)l antibody to the human patient.
- the anti-PD-(L)l antibody is selected from the group consisting of pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the method further comprises co-administering zimberelimab to the human patient.
- the human mCRPC patient has previously progressed on ADT.
- the human mCRPC patient has previously progressed on NHA.
- the human mCRPC patient is CPI and taxane naive.
- the human mCRPC patient has RECIST 1.1 measurable or non-measurable disease.
- the CRPC is metastatic CRPC (mCRPC).
- the CRPC is resistant or refractory to one or more anti-cancer therapies.
- the CRPC has progressed following prior NHA therapy (first- or second-generation non-steroidal antiandrogens, abiraterone).
- prior NHA therapy first- or second-generation non-steroidal antiandrogens, abiraterone.
- the human patient has not received a prior therapy selected from taxanc therapy (taxanc naive), checkpoint inhibitor therapy (CPI naive), and topoisomerase I inhibitor therapy.
- the human patient has not received a prior taxane therapy (taxane naive), checkpoint inhibitor therapy (CPI naive), or topoisomerase I inhibitor therapy.
- taxane naive prior taxane therapy
- CPI naive checkpoint inhibitor therapy
- topoisomerase I inhibitor therapy topoisomerase I inhibitor therapy
- the taxane therapy comprises paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, or cabazitaxel.
- the checkpoint inhibitor therapy comprises an anti-CTLA4 antibody or an anti-PD(L)! antibody.
- the topoisomerase I inhibitor therapy comprises a topotecan, irinotecan, belotecan or exatecan.
- NSCLC non-small cell lung cancer
- the anti-PD-(L)l antibody is selected from the group consisting of pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD-(L)l antibody is zimberelimab.
- the NSCLC has progressed or recurred after platinum-based chemotherapy. In some embodiments, the NSCLC has progressed or recurred after checkpoint inhibitor therapy (CPI) therapy. In some embodiments, the NSCLC has progressed or recurred after platinum based chemotherapy and anti-PD-(L)l antibody therapy, received either in combination or sequentially in any order. In some embodiments, the NSCLC has progressed or recurred after a tyrosine kinase inhibitor therapy. In some embodiments, the NSCLC is (i) unresectable, locally advanced or (ii) metastatic.
- sacituzumab govitecan is administered intravenously (IV) at a dose of 8 mg/kg or 10 mg/kg on day 1 and day 8 of a 21 -day treatment cycle and etrumadenant is administered orally (PO) once a day (QD) at a dose of 75 mg or 150 mg on each day of the 21 -day treatment cycle.
- sacituzumab govitecan is administered intravenously (IV) at a dose of 10 mg/kg on day 1 and day 8 of a 21-day treatment cycle and etrumadenant is administered orally (PO) once a day (QD) at a dose of 150 mg on each day of the 21-day treatment cycle.
- zimberelimab is administered intravenously (IV) on day 1 of the 21-day treatment cycle (Q3W).
- an anti-cancer effect is observed as determined by objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), PSA response rate, radiographic response rate, change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers, or combinations thereof.
- an anti-cancer effect is observed as determined by objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), overall survival (OS), complete response (CR), partial response (PR), PSA response rate, radiographic response rate, or change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- an anti-CD47 antibody e.g., magrolimab
- an anti-CD47 antibody is not co-administered to the subject or human patient.
- an MCL1 inhibitor e.g., GS-9716
- GS-9716 is not co-administered to the subject or human patient.
- a FLT3 agonist e.g., GS-3583, CDX-301
- GS-3583, CDX-301 is not administered to the subject or human patient.
- TA + tumor antigen positive
- TA + tumor antigen positive
- TA + tumor antigen positive
- TA + tumor antigen positive
- a tumor antigen targeted ADC comprising a topoisomerase I inhibitor (Topi ADC)
- Topici ADC topoisomerase I inhibitor
- adenosine pathway inhibitor an adenosine pathway inhibitor
- optionally an anti-PD(L)l antibody optionally an anti-PD(L)l antibody.
- an anti-Trop-2 ADC for use in combination with an adenosine pathway inhibitor and optionally an anti-PD(L)l antibody in a method of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of Trop-2 positive cancers, wherein the method comprises administering the anti-Trop-2 ADC, adenosine pathway inhibitor, and optionally the additional anti-PD(L) 1 antibody to a subject.
- a Topi ADC for use in combination with an adenosine pathway inhibitor and optionally an anti-PD(L)l antibody in a method of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of tumor antigen positive (TA + ) cancers, wherein the method comprises administering the Topi ADC, adenosine pathway inhibitor, and optionally the anti-PD(L)l antibody to a subject.
- arc combination therapies for treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of Trop-2 positive (Trop-2 + ) cancers by co-administering effective amounts of a) an anti-Trop-2 antibody-drug conjugate (anti-Trop-2 ADC) and b) an adenosine pathway inhibitor to a subject.
- an anti-Trop-2 antibody-drug conjugate anti-Trop-2 ADC
- combination therapies for treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of a tumor antigen positive (TA + ) cancer by co-administering an effective amount of a) a tumor antigen (TA) targeted ADC comprising a topoisomerase I inhibitor (Topi ADC) and b) an effective amount of an adenosine pathway inhibitor to a subject.
- the combination therapies provided herein further comprise co-administering an additional therapeutic agent, such as a checkpoint inhibitor, or an additional therapeutic modality (e.g., surgery, radiation).
- the anti-Trop-2 ADC is sacituzumab govitecan.
- the Topi ADC is trastuzumab-deruxtecan (T-DXd).
- the adenosine pathway inhibitor is a CD39 inhibitor (e.g., TTX-030, IPH5201, SRF617), a CD73 inhibitor (e.g., quemliclustat, oleclumab), or an adenosine receptor antagonist (e.g., etrumadenant, imaradenant, NIR178).
- the checkpoint inhibitor is an anti-PD(L)l antibody (e.g., zimberelimab, pembrolizumab, nivolumab, atezolizumab, durvalumab).
- the Trop-2 positive cancer is castrate-resistant prostate cancer (CRPC) or non-small cell lung cancer (NSCLC).
- an anti-Trop-2 ADC e.g., sacituzumab govitecan
- an adenosine pathway inhibitor e.g., etrumadenant
- an additional therapeutic agent such as a checkpoint inhibitor (e.g., anti-PD(L)l antibody), or an additional therapeutic modality (e.g., surgery, radiation) in a method of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of Trop-2 positive cancers
- the method comprises administering the anti-Trop-2 ADC, adenosine pathway inhibitor, and optionally the additional therapeutic agent or additional therapeutic modality to a subject (e.g., a human cancer patient).
- the anti-Trop-2 ADC is sacituzumab govitecan.
- the adenosine pathway inhibitor is a CD39 inhibitor (e.g., TTX-030, IPH5201, SRF617), a CD73 inhibitor (e.g., quemliclustat, oleclumab), or an adenosine receptor antagonist (e.g., etrumadenant, imaradenant, NIR178).
- the checkpoint inhibitor is an anti-PD(L)l antibody (e.g., zimberelimab, pembrolizumab, nivolumab, atezolizumab, durvalumab).
- the Trop-2 positive cancer is castrate-resistant prostate cancer (CRPC) or non-small cell lung cancer (NSCLC).
- a Topi ADC for use in combination with an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, adenosine receptor antagonist) and optionally an additional therapeutic agent, such as a checkpoint inhibitor (e.g., anti-PD(L)l antibody), or an additional therapeutic modality (e.g., surgery, radiation) in a method of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of TA positive cancers, wherein the method comprises administering the Topi ADC, adenosine pathway inhibitor, and optionally the additional therapeutic agent or additional therapeutic modality to a subject (e.g., a human cancer patient).
- an adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, adenosine receptor antagonist
- an additional therapeutic agent such as a checkpoint inhibitor (e.g., anti-PD(L)l antibody), or an additional therapeutic modality (e.g., surgery, radiation) in
- the Topi ADC is trastuzumab-deruxtecan (T-DXd).
- the adenosine pathway inhibitor is a CD39 inhibitor (e.g., TTX-030, IPH5201, SRF617), a CD73 inhibitor (e.g., quemliclustat, oleclumab), or an adenosine receptor antagonist (e.g., etrumadenant, imaradenant, NIR178).
- the checkpoint inhibitor is an anti-PD(L)l antibody (e.g., zimberelimab, pembrolizumab, nivolumab, atezolizumab, durvalumab).
- the Trop-2 positive cancer is castrate-resistant prostate cancer (CRPC) or non-small cell lung cancer (NSCLC).
- This disclosure is based, at least in part, on the realization that a combination therapy comprising co-administration of a) an anti-Trop2 ADC or a Topi ADC and b) an adenosine pathway inhibitor can have improved anticancer effects compared to single-agent therapies with one of the combined agents a) and b).
- Improved anti-cancer effects can include, for example, improved overall response rates (ORR), improved partial response rates (PR), improved complete response rates (CR), improved durations of response (DOR), improved overall survival (OS), improved progression free survival (PFS), improved quality of life (QoL) indicators, or the like.
- the combination therapies described herein generally have tolerable safety profiles, c.g., as determined by the incidence and/or severity of observed adverse events (AEs) or serious adverse events (SAEs).
- the combination therapies described herein have improved safety profiles, e.g., as compared to monotherapies involving the co-administered agents, or relative to a standard of care for a given indication (e.g., a chemotherapy regimen of physician’s choice).
- the term “antibody” refers to a polypeptide that includes canonical immunoglobulin sequence elements sufficient to confer specific binding to a particular target antigen (e.g., a heavy chain variable domain, a light chain variable domain, and/or one or more CDRs sufficient to confer specific binding to a particular target antigen).
- a particular target antigen e.g., a heavy chain variable domain, a light chain variable domain, and/or one or more CDRs sufficient to confer specific binding to a particular target antigen.
- the term antibody includes, for example, and without limitation, human antibodies, non-human antibodies, antibody fragments, and antigen-binding agents that include antibody fragments, inclusive of synthetic, engineered, and modified forms thereof.
- the term antibody includes, by way of example, both naturally occurring and non-naturally occurring antibodies.
- an antibody may comprise at least two heavy (H) chains and two light (L) chains interconnected by disulfide bonds, or an antigen-binding molecule thereof.
- Each H chain comprises a heavy chain variable region (abbreviated herein as VH) and a heavy chain constant region.
- the heavy chain constant region comprises three constant domains, CHI, CH2 and CH3.
- Each light chain comprises a light chain variable region (abbreviated herein as VL) and a light chain constant region.
- the light chain constant region comprises one constant domain, CL.
- the VH and VL regions can be further subdivided into regions of hypervariability, termed complementarity determining regions (CDRs), interspersed with regions that are more conserved, termed framework regions (FR).
- CDRs complementarity determining regions
- Each VH and VL comprises three CDRs and four FRs, arranged from amino-terminus to carboxy-terminus in the following order: FR1, CDR1, FR2, CDR2, FR3, CDR3, and FR4.
- the variable regions of the heavy and light chains contain a binding domain that interacts with an antigen.
- the constant regions of the antibodies (Abs) may mediate the binding of the immunoglobulin to host tissues or factors, including various cells of the immune system (e.g., effector cells) and the first component (Clq) of the classical complement system.
- Naturally -produced antibodies are glycosylated, typically on the CH2 domain.
- antibodies include monoclonal antibodies, monospecific antibodies, polyclonal antibodies, multispecific antibodies (including bispecific antibodies), engineered antibodies, rccombinantly produced antibodies, wholly synthetic antibodies, humanized antibodies, chimeric antibodies, immunoglobulins, tetrameric antibodies comprising two heavy chain and two light chain molecules, antibody light chain monomers, antibody heavy chain monomers, antibody light chain dimers, antibody heavy chain dimers, antibody light chain- antibody heavy chain pairs, intrabodies, antibody fusions (sometimes referred to herein as “antibody conjugates”), heteroconjugate antibodies, single domain antibodies, monovalent antibodies, single chain antibodies or single-chain Fvs (scFv), camelized antibodies, affybodies, Fab fragments, Fab' fragments, F(ab’)2 fragments, Fd' fragments, Fd fragments, isolated CDRs, single chain Fvs, polypeptide-Fc fusions, single domain antibodies (e.g., shark single domain antibodies such as IgNAR or fragments thereof); came
- the term “antibody-drug conjugate” generally refers to a compound comprising an antibody targeting a tumor antigen and an anticancer agent payload, optionally connected by a linker.
- the tumor antigen is tumor- associated calcium signal transducer 2 (Trop-2; NCBI Gene ID: 4070).
- the tumor antigen targeted antibody is an anti-Trop-2 antibody (e.g., sacituzumab or datopotamab).
- the payload is a topoisomerase I inhibitor (e.g., SN38 or Dxd).
- ADC linker chemistries are known to a skilled artisan and referenced herein (e.g., CL2A).
- topoisomerase I inhibitor refers to small molecule compounds capable of inhibiting the activity of a DNA topoisomerase type I enzyme.
- Type I topoisomerases can catalyze changes in DNA topology via transient single-stranded breaks in DNA.
- Type I topoisomerases can be further classified as Type 1 A and a Type IB subtypes.
- a description of type I topoisomerases can be found, for example, in Baker et al. (2009) Nucleic Acids Res 37(3), 693- 701.
- Topoisomerase inhibitors that can be used as payloads in the ADCs described herein include camptothecin (CPT) and non-camptothecin based inhibitors.
- Useful camptothecins include, for example, topotecan, irinotecan, belotecan, exatecan, and derivatives thereof.
- Useful non- camptothccins include, for example, indcnosinoquinolincs (c.g., indcno[l,2-c]isoquinolinc, NSC314622, indotecan (LMP-400), indimitecan (LMP-776)), phenanthridines (e.g., topovale (ARC- 111), and indolocarbazoles (e.g., BE-13793C).
- the topoisomerase I inhibitor is a camptothecin (e.g., an irinotecan, topotecan, belotecan, or exatecan derivative, such as SN38 or Dxd). In some embodiments, the topoisomerase I inhibitor is SN38. In some embodiments, the topoisomerase I inhibitor is Dxd.
- camptothecin e.g., an irinotecan, topotecan, belotecan, or exatecan derivative, such as SN38 or Dxd.
- the topoisomerase I inhibitor is SN38. In some embodiments, the topoisomerase I inhibitor is Dxd.
- anti-PD(L)l antibody refers to antibodies that a) bind to programmed cell death protein 1 (PD-1, CD279; NCBI Gene ID: 5133) or programmed deathligand 1 (PD-L1 , CD274; NCBI Gene ID: 29126); and b) inhibit the PD-1/PD-L1 interaction and PD-1/PD-L1 pathway.
- PD-1, CD279; NCBI Gene ID: 5133 programmed cell death protein 1
- PD-L1 , CD274 NCBI Gene ID: 29126
- the PD-1/PD-L1 pathway and its role in cancer immunotherapy is described, for example, in Salmaninejad etal, J. Cell Physiol (2019) 234 (10): 16824-16837.
- Anti- PD(L)1 antibodies that can be used in the methods provided herein include, for example, pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD(L)l antibody is zimberelimab.
- the terms “effective amount” or “therapeutically effective amount” refer to that amount of a therapeutic agent administered in the methods provided herein (e.g., ADC, adenosine pathway inhibitor, checkpoint inhibitor) that, when administered alone or in combination with another therapeutic agent to a cell, tissue, or subject is sufficient to effect treatment or a beneficial result in the subject.
- the therapeutically effective amount may vary depending on the subject, and disease or condition being treated, the weight and age of the subject, the severity of the disease or condition, and the manner of administering, which can readily be determined by one of ordinary skill in the art.
- an effective amount further refers to that amount of the therapeutic agent, which when used in the context of the combination therapies provided herein, is sufficient to treat, prevent, alleviate, ameliorate or mitigate a disease condition, or delay or slow the progression of a disease, and that amount sufficient to effect an increase in rate of treatment, healing, prevention or amelioration of such conditions.
- an effective amount refers to that active ingredient alone.
- a therapeutically effective amount refers to combined amounts of the active ingredients that result in the therapeutic effect, whether administered in combination, serially or simultaneously.
- an effective amount or therapeutically effective amount of a therapeutic agent e.g., ADC, adenosine pathway inhibitor, checkpoint inhibitor
- a therapeutic agent e.g., ADC, adenosine pathway inhibitor, checkpoint inhibitor
- the amount is sufficient to ameliorate, palliate, lessen, and/or delay one or more of symptoms of cancer.
- treatment refers to reversing, alleviating, delaying the onset of, or inhibiting the progress of a disease or disorder, or one or more symptoms thereof, as described herein.
- treatment may be administered after one or more symptoms have developed.
- treatment may be administered in the absence of symptoms.
- treatment may be administered to a susceptible individual prior to the onset of symptoms e.g., in light of a history of symptoms and/or in light of genetic or other susceptibility factors). Treatment may also be continued after symptoms have resolved, for example, to prevent or delay their recurrence.
- the methods provided herein refer to the treatment of a subject having cancer (e.g., a human cancer patient).
- treating a subject having cancer comprises inhibiting cancer or cancer cell proliferation in the treated subject.
- treating a human cancer patient using the methods provided herein results in the observation of anti-tumor effects or anti-cancer effects in the treated patient.
- the terms “inhibition of cancer” and “inhibition of cancer cell proliferation” refer to the inhibition of the growth, division, maturation or viability of cancer cells, and/or causing the death of cancer cells, individually or in aggregate with other cancer cells, by cytotoxicity, nutrient depletion, or the induction of apoptosis.
- anti-tumor effect refers to a biological effect that can present as a decrease in tumor volume, a decrease in the number of tumor cells, a decrease in tumor cell proliferation, a decrease in the number of metastases, an increase in overall or progression-free survival, an increase in life expectancy, or amelioration of various physiological symptoms associated with the tumor.
- anti-cancer effects are measured using one or more of the endpoint criteria applied in the clinical studies described herein (e.g., primary, secondary, or exploratory endpoints).
- Exemplary clinical endpoint criteria that can be used to measure anti-cancer effects in connection with the methods provided herein include objective response rate (ORR), complete response (CR) rate, partial response (PR) rate, disease control rate (DCR), progression-free survival (PFS), duration of response (DOR), overall survival (OS), biomarker-based signals, e.g., of intratumoral immune activation or induction of cancer cell death (e.g., tumor tissue or blood based biomarkers), patient quality of life (QoL) indicators (e.g., based on patient surveys), and others.
- anti-tumor effects e.g., tumor responses or progression
- RECIST version 1.1 Eisenhauer et al. Eur J Cancer (2009);45 (2):228-47.
- Anti-cancer effects can be observed using any diagnostic methods known to a skilled artisan, such as computed tomography (CT), magnetic resonance imaging (MRI), radiography, or the like.
- an “increased” or “enhanced” amount is typically a “statistically significant” amount (e.g., with respect to tumor size, cancer cell proliferation or growth), and may include an increase that is 1.1, 1.2, 1.3, 1.4, 1.5, 1.6, 1.7, 1.8, 1.9, 2, 2.5, 3, 3.5, 4, 4.5, 5, 6, 7, 8, 9, 10, 15, 20, 30, 40, or 50 or more times (e.g., 100, 500, 1000 times) (including all integers and decimal points in between and above 1, e.g., 2.1, 2.2, 2.3, 2.4, etc.) an amount or level described herein.
- It may also include an increase of at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, at least 90%, at least 100%, at least 150%, at least 200%, at least 500%, or at least 1000% of an amount or level described herein.
- a “decreased” or “reduced” or “lesser” amount refers to a decrease that is about 1.1, 1.2, 1.3, 1.4, 1.5, 1.6
- an amount or level described herein may also include a decrease of at least 10%, at least 20%, at least 30%, at least 40%, at least 50%, at least 60%, at least 70%, at least 80%, or at least 90%, at least 100%, at least 150%, at least 200%, at least 500%, or at least 1000% of an amount or level described herein.
- AE adverse event
- Adverse event refers to any untoward medical occurrence in a clinical study participant administered a study drug that does not necessarily have a causal relationship with the treatment.
- An AE can therefore be any unfavorable and/or unintended sign (including clinically significant abnormal laboratory findings), symptom, or disease temporally associated with the use of a study drug, whether or not the AE is considered related to the study drug.
- Adverse events may also include pretreatment or posttreatment complications that occur as a result of protocol-specified procedures or special situations.
- preexisting events that increase in severity or change in nature after study drug initiation or during or as a consequence of participation in the clinical study are also considered AEs.
- the term “serious adverse event” refers to a) death; b) a lifethreatening situation; c) in-participant hospitalization or prologation of existing hospitalization; d) persistent or significant disability or incapacity; e) a congenital anomaly or birth defect; or f) a medically important event or reaction as determined by an attending physician.
- medically important events include intensive treatment in an emergency room or at home for allergic bronchospasm; blood dyscrasias or convulsions that do not result in hospitalization; and development of drug dependency or drug abuse.
- tumor antigen expressing cancer or “tumor antigen positive cancer” are used interchangeably to refer to cancers having detectable levels of tumor antigen (TA) expression.
- the tumor antigen is Trop-2.
- Tumor antigen expression in a cancer tissue or cancer cell can be detected in a sample from a subject having cancer (e.g., a human cancer patient) by any method known to a skilled artisan, e.g., as a protein, mRNA, or cell-surface expression level.
- tumor antigen expression can be determined by methods such as immunohistochemistry (IHC), western blot, fluorescence in-situ hybridization (FISH), polymerase chain reaction (PCR), next-generation exome sequencing, or fluorescence associated cell sorting (FACS).
- IHC immunohistochemistry
- FISH fluorescence in-situ hybridization
- PCR polymerase chain reaction
- FACS fluorescence associated cell sorting
- TA expressing cancer or “TA positive cancer”
- less than 99%, less than 95%, less than 90%, less than 80%, less than 70%, less than 60%, less than 50%, less than 40%, less than 30%, less than 20%, or less than 10% of cells in a tumor or tumor sample have detectable levels of tumor antigen expression.
- 99% or more, 95% or more, 90% or more, 80% or more, 70% or more, 60% or more, 50% or more, 40% or more, 30% or more, 20% or more, or 10% or more of cells in a tumor or tumor sample have detectable levels of tumor antigen expression.
- TA expressing cancer or “TA positive cancer” refers to a cancer for which treatment with a certain anti-Trop-2 ADC or Topi ADC is indicated either as a single-agent therapy or in a combination.
- TA positive cancer indications of anti-Trop-2 ADCs or Topi ADCs that have received a marketing authorization from a regulatory health agency e.g., FDA, EMA are listed, for example, on agency approved drug product labels.
- a TA positive (e.g., Trop-2 + ) cancer is a cancer in which an anti-Trop-2 ADC or Topi ADC has demonstrated an anti-cancer effect that is attributable to the anti-Trop-2 ADC or Topi ADC.
- Such anti-cancer effects can be demonstrated in a preclinical model (e.g., a mouse xenograft or syngeneic cancer model) or in a clinical trial conducted with human cancer patients.
- the TA expressing cancer or TA positive cancer does not express detectable levels of Trop-2, e.g., as determined by IHC and/or FISH analysis.
- the term “tumor antigen targeted antibody-drug conjugate (ADC)” generally refers to an ADC comprising a tumor antigen binding antibody.
- ADC tumor antigen targeted antibody-drug conjugate
- Such TA binding antibodies can generally direct an ADC to TA expressing cancer cells in a tumor tissue.
- the TA binding antibody is a neutralizing or blocking antibody (competing with the binding of another binding partner to the TA).
- the TA binding antibody can modulate TA associated molecular or cellular signaling events.
- the TA binding antibody has antagonistic or agonistic activity relative to the TA.
- TA targeted ADCs that can be used in the methods provided herein include, without limitation, gemtuzumab ozogamicin, brentuximab vedotin, ado-trastuzumab emtansine (T-DM1), inotuzumab ozogamicin, trastuzumab deruxtecan (T-DXD), datopotamab deruxtecan (DATO-DXD), polatuzumab vedotin, sacituzumab govitecan, labetuzumab govitecan, enfortumab vedotin, and belantamab mafodotin.
- the TA targeted ADC does not bind to Trop-2.
- ADCs Antibody-Drug-Conjugates
- the treatment methods provided herein comprise co-administering an antibody-drug- conjugate (ADC) to a subject, such as a human cancer patient.
- ADC antibody-drug- conjugate
- the ADC comprises an anti-Trop-2 antibody, an anti-cancer agent payload, and an optional linker connecting the anti-Trop-2 antibody and payload (anti-Trop-2 ADC).
- the anti-cancer agent payload in the anti-Trop-2 ADC is a topoisomerase I inhibitor (e.g., SN38, Dxd).
- the anti-cancer agent payload in the anti-Trop-2 ADC does not include a topoisomerase I inhibitor.
- the ADC comprises a tumor antigen (TA) targeted antibody, a topoisomerase I inhibitor payload, and an optional linker connecting the TA targeted antibody and payload (Topi ADC).
- TA targeted antibody in the Topi ADC is an anti-Trop-2 antibody.
- the TA targeted antibody in the TropI ADC does not include an anti-Trop-2 antibody.
- ADCs that can be used in the methods provided herein can comprise antibodies or antigen- binding fragments thereof of any format.
- the ADC can include, without limitation, a monospecific or multispecific (e.g., bispecific, trispecific) antibody, or an antigenbinding fragment thereof, in any format, such as DART®, Duobody®, BiTE®, BiKE, TriKE, XmAb®, TandAb®, scFv, Fab, or Fab derivative.
- the ADC comprises a non-immunoglobulin antibody mimetic (e.g., including adnectin, affibody, affilin, affimer, affitin, alphabody, anticalin, peptide aptamer, armadillo repeat protein (ARM), atrimer, avimer, designed ankyrin repeat protein (DARPin®), fynomer, knottin, Kunitz domain peptide, monobody, and nanoCLAMPs).
- a non-immunoglobulin antibody mimetic e.g., including adnectin, affibody, affilin, affimer, affitin, alphabody, anticalin, peptide aptamer, armadillo repeat protein (ARM), atrimer, avimer, designed ankyrin repeat protein (DARPin®), fynomer, knottin, Kunitz domain peptide, monobody, and nanoCLAMPs).
- the ADC antibody is a blocking antibody. In some embodiments, the ADC antibody is a neutralizing antibody. In some embodiments, the ADC antibody is an agonistic or activating antibody. Tn some embodiments, the ADC antibody is an antagonistic or inhibitory antibody.
- the ADC comprises an IgG antibody or antigen-binding fragment thereof.
- the IgG antibody or antigen-binding fragment thereof can be of various isotypes, such as IgGl, IgG2, IgG3 or IgG4.
- the ADC antibody comprises human IgGl hinge and constant region sequences.
- the ADC antibody can be a chimeric human-mouse, a chimeric human-primate, a humanized (human framework and murine hypervariable (CDR) regions), or a fully human antibody, as well as a variation thereof.
- the ADC antibody is a half-IgG4 antibody (referred to as “unibody”), as described, e.g., by van der Neut Kolfschoten et al. (Science 2007; 317:1554-1557).
- the ADC antibody or antigen-binding fragment thereof is designed or selected to comprise human constant region sequences that belong to specific allotypes, which may result in reduced immunogenicity when the antibody or ADC is administered to a human subject.
- the ADC antibody or antigen-binding fragment thereof is of a non-Glml allotype (nGlml), such as Glm3, Glm3,l, Glm3,2 or Glm3,l,2.
- the allotype is selected from the group consisting of the nGlml, Glm3, nGlml, 2 and Km3 allotypes.
- the ADCs that can be used in the methods provided herein comprise an anti-Trop-2 antibody, an anti-cancer agent payload, and an optional linker connecting the anti-Trop-2 antibody and payload (Anti-Trop-2 ADC).
- the pay load is a topoisomerase I inhibitor. In some embodiments, the payload does not include a toposisomerase I inhibitor.
- anti-Trop-2 antibodies that can be used in anti-Trop-2 ADCs to perform the methods provided herein include, but are not limited to, those described in W02020016662 (Abmart), W02020249063 (Bio-Thera Solutions), US20190048095 (Bio-Thera Solutions), WO2013077458 (LivTech/Chiome), EP20110783675 (Chiome), W02015098099 (Daiichi Sankyo), WG2017002776 (Daiichi Sankyo), W02020130125 (Daiichi Sankyo), W02020240467 (Daiichi Sankyo), US2021093730 (Daiichi Sankyo), US9850312 (Daiichi Sankyo), CN112321715 (Biosion), US2006193865 (Immunomedics/Gilead), WO2011068845 (Immunomedic s/Gilead), US2016296633 (
- WO2020257648 (Gilead), US2013039861 (Gilead), WO2014163684 (Gilead), US9427464 (LivTech/Chiome), US10501555 (Abruzzo Theranostic/Oncoxx), WO2018036428 (Sichuan Kelun Pharma), WO2013068946 (Pfizer), W02007095749 (Roche), and W02020094670 (SynAffix).
- the anti-Trop-2 ADC comprises an antibody selected from sacituzumab (hRS7), datopotamab (hTINA HILI), and a Trop-2 binding fragment thereof.
- the anti-Trop-2 ADC is sacituzumab (hRS7).
- the anti-Trop-2 antibody is datopotamab (hTINA HILI).
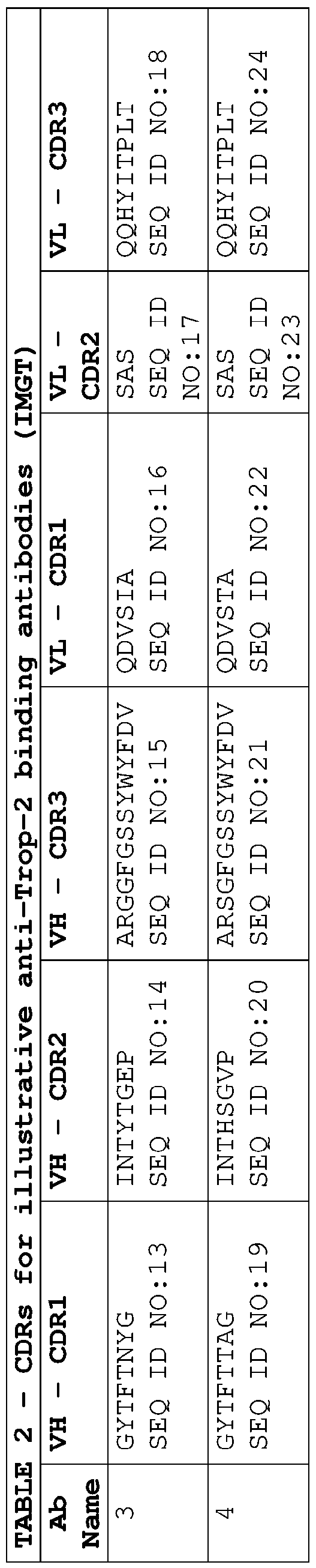
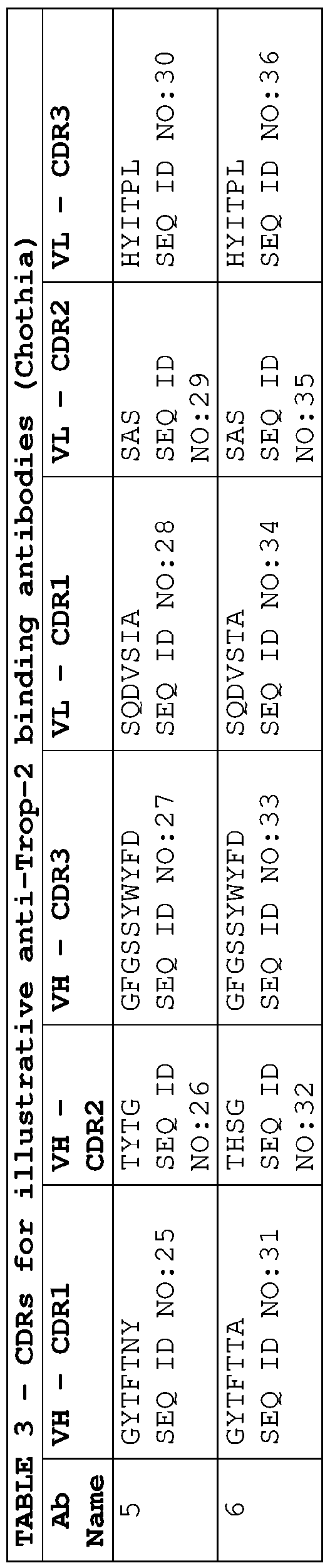
- the anti-Trop-2 ADC comprises a VH-CDR1, a VH-CDR2, a VH-CDR3, a VL-CDR1, a VL-CDR2 and a VL-CDR3 selected from one of Tables 1 to 4.
- the anti-Trop-2 ADC comprises the following VH- CDR1 , a VH-CDR2, a VH-CDR3, a VL-CDR1 , a VL-CDR2 and a VL-CDR3 amino acid sequences (according to Kabat), respectively:
- the anti-Trop-2 ADC comprise variable domains (VH and VL) selected from Table 5.
- the anti-Trop-2 ADC comprises the following VH and VL amino acid sequences, respectively:
- the anti-Trop-2 ADC comprises an anti-Trop-2 antibody, an anti-cancer agent payload, and an optional linker connecting antibody and pay load.
- the linker is non-cleavable (e.g., a maleimidocaproyl or maleimidomethyl cyclohexane- 1 -carboxylate linker).
- the linker is cleavable.
- the linker is acid cleavable (e.g., a hydrazone linker).
- the cleavable linker is reducible (e.g., a disulphide linker).
- the linker is protease cleavable (e.g., a dipeptide or tetrapeptide linker). In some embodiments, the linker is selected from linkers disclosed in USPN 7,999,083 (e.g., CL2A, CL6, CL7, CLX, or CLY). In some embodiments, the linker is CL2A.
- Exemplary anti-cancer agent payloads that can be used in anti-Trop-2 ADCs in the methods provided herein include, for example, microtubule inhibitors, DNA cleavage agents, and topoisomerase I inhibitors.
- the microtubule inhibitor is an auristatin (e.g., monomethyl auristatin E (MMAE) or monomethyl auristatin F (MMAF), a taxane, a vinca alkaloid, an epothilone) or maytansinoid (e.g., mertansine (DM1) or ravtansine (DM4)).
- auristatin e.g., monomethyl auristatin E (MMAE) or monomethyl auristatin F (MMAF)
- MMAE monomethyl auristatin E
- MMAF monomethyl auristatin F
- DM1 mertansine
- DM4 mertansine
- the DNA cleavage agent is a calicheamicin (e.g., ozogamicin).
- the topoisomerase I inhibitor is a camptothecin (e.g., an irinotecan, topotecan, belotecan, or exatecan derivative, such as SN38 or Dxd).
- the topoisomerase I inhibitor is SN38.
- the topoisomerase I inhibitor is Dxd.
- the topoisomerase I inhibitor is not a camptothecin (i.e. non-camptothecin topoisomerase 1 inhibitor).
- the non-camptothecin topoisomerase 1 inhibitor is selected from an indenosinoquinoline (e.g., indeno[l,2-c]isoquinoline, NSC314622, indotecan (LMP-400), indimitecan (LMP-776)), a phenanthridine (e.g., topovale (ARC-111), and a indolocarbazole (e.g., BE-13793C).
- indenosinoquinoline e.g., indeno[l,2-c]isoquinoline, NSC314622, indotecan (LMP-400), indimitecan (LMP-776)
- a phenanthridine e.g., topovale (ARC-111)
- a indolocarbazole e.g., BE-13793C
- Additional illustrative anti-cancer agent payloads that can be conjugated to anti-Trop-2 ADCs include without limitation anthracyline (e.g., doxorubicin, daunorubicin, epirubicin, idarubicin), pyrrolobenzodiazepine (PBD), or dimer thereof, DNA cross-linking agent SC-DR002 (D6.5), duocarmycin, a duocarmycin (A, B l, B2, Cl, C2, D, SA, CC-1065), tubulysin B and analogs thereof (e.g., Tubl96), and other anti-cancer agents described herein.
- anthracyline e.g., doxorubicin, daunorubicin, epirubicin, idarubicin
- PBD pyrrolobenzodiazepine
- SC-DR002 DNA cross-linking agent SC-DR002 (D6.5)
- duocarmycin a duocarmycin (A, B
- Exemplary anti-Trop-2 ADCs that can be used in the methods provided herein are described in WO21225892 (Shanghai Escugen Biotechnology; ESG-401, STI-3258), W022010797 (BiOneCure Therapeutics; BIO-106), CN112237634 (Shanghai Fudan-Zhangjiang Biopharmaceutical; FDA018-ADC), WO19114666 (Sichuan Kelun Pharmaceutical; KLA264), WO22078524 (Hangzhou DAC Biotech; DAC-002), W015098099 (Daiichi Sankyo; datopotamab deruxtecan), WO21147993 (Jiangsu Hengrui Medicine; SHR-A1921), and WO21052402 (Sichuan Baili Pharmaceutical; BL-M02D1 ).
- the anti-Trop-2 ADC is selected from sacituzumab govitecan (Immunomedic s/Gilead), datopotamab deruxtecan (DS- 1062, Dato- Dxd; Daiichi Snakyo/AstraZeneca), SKB-264 (KL-A264; Klus Pharma, Sichuan Kelun Pharma), ESG-401 (Shanghai Escugen Bio technology/Le vena Biopharma), JS-108 (DAC-002; Junshi Bio/Hangzhou DAC), FDA018-ADC (Shanghai Fudan Zhangjiang Bio Pharma), STI-3258 (Sorrento), OXG-64 (Oncoxx), BD1-4702 (OBI Pharma), BL-M02D1 (Systimmune), Anti-Trop- 2 Ab (Mediterrania Theranostic/Legochem), KD-065 (Nanjing KAEDI Bio
- the anti-Trop-2 ADC is sacituzumab govitecan (Immunomedics/Gilead). In some embodiments, the anti-Trop-2 ADC is selected from sacituzumab govitecan, datopotamab deruxtecan (DS-1062), ESG-401, SKB-264, DAC-02 and BAT-8003. In some embodiments, the anti-Trop-2 ADC is sacituzumab govitecan. In some embodiments, the anti-Trop-2 ADC is datopotamab deruxtecan (DS-1062, Dato-Dxd; Daiichi Snakyo/AstraZeneca). Further examples of useful anti-Trop-2 therapeutics include, but are not limited to, those described in W02016201300 (Gilead), and CN108440674 (Hangzhou Lonzymc Biological Technology).
- anti-Trop-2 ADCs that can be used in the methods provided herein are described, for example, in USPN 7,999,083 and USPN 9,028,833, which are hereby incorporated herein by reference in their entireties.
- the anti-Trop-2 ADC comprises a topoisomerase I inhibitor.
- the topoisomerase I inhibitor is selected from irinotecan, topotecan, and SN-38.
- the anti-Trop-2 ADC has a structural formula of mAb-CL2A-SN-38, with a structure represented by:
- the anti-Trop-2 ADC comprises sacituzumab (hRS7; described, e.g., in W02003074566, Figures 3 and 4).
- the anti-Trop-2 ADC is sacituzumab govitecan (IMMU-132). Sacituzumab govitecan (SG) is an antibody-drug conjugate (ADC) composed of the following 3 components:
- the humanized monoclonal antibody hRS7 IgGlK which binds to trophoblast cellsurface antigen 2 (Trop-2; TACSTD2; EGP-1; NCBI Gene ID: 4070), a transmembrane calcium signal transducer that is overexpressed in many epithelial cancers, including triple-negative breast cancer (TNBC).
- camptothecin-derived agent SN-38 a topoisomerase I inhibitor.
- a hydrolyzable linker that links the humanized monoclonal antibody to SN-38.
- CL2A hydrolyzable linker
- Additional exemplary anti-Trop-2 ADCs that can be used in the methods provided herein arc described in WO21225892 (Shanghai Escugcn Biotechnology).
- the anti-Trop-2 ADC comprises a linker-payload conjugate having a structure represented by: attached to an anti-Trop-2 antibody (e.g., hRS7).
- the anti-Trop-2 ADC has a DAR of between 1 and 8.
- the anti-Trop-2 ADC has a DAR of between 7.0 and 8.0.
- the anti-Trop-2 ADC is ESG-401 (STI-3258).
- the anti-Trop-2 ADC comprises a linker-payload conjugate (TL035) having a structure represented by:
- the anti-Trop-2 ADC has a DAR of between 1 and 8. In some embodiments, the anti-Trop-2 ADC has a DAR of between 7.0 and 8.0. In some embodiments, the anti-Trop-2 ADC has a DAR of about 7.0. In some embodiments, the anti-Trop-2 ADC is KL-A264.
- anti-Trop-2 ADC comprises a linker-payload conjugate having a structure represented by:
- the anti-Trop-2 ADC has a DAR of between 1 and 8. In some embodiments, the anti-Trop-2 ADC has a DAR of ⁇ 7.0. In some embodiments, the anti-Trop-2 ADC has a DAR of about 4. In some embodiments, the anti-Trop-2 ADC is datopotamab deruxtecan.
- the ADCs that can be co-administered in the methods provided herein comprise a tumor antigen (TA) targeted antibody, a topoisomerase I inhibitor payload, and an optional linker connecting the TA targeted antibody and payload (Topi ADC).
- TA tumor antigen
- Topici ADC topoisomerase I inhibitor
- the TA targeted antibody in the Topi ADC is an anti-Trop-2 antibody.
- the TA targeted antibody in the Topi ADC does not comprise an anti-Trop-2 antibody.
- the Topi ADCs that can be co-administered in the methods provided herein comprise an antibody that binds a tumor antigen selected from carbonic anhydrase IX, B7, CCCL19, CCCL21, CSAp, HER-2/neu, BrE3, CD1, CDla, CD2, CD3, CD4, CD5, CD8, CD11A, CD14, CD15, CD16, CD18, CD19, CD20, CD21, CD22, CD23, CD25, CD29, CD30, CD32b, CD33, CD37, CD38, CD40, CD40L, CD44, CD45, CD46, CD52, CD54, CD55, CD59, CD64, CD67, CD70, CD74, CD79a, CD80, CD83, CD95, CD126, CD133, CD138, CD147, CD154, CEACAM5, CEACAM-6, alpha-fetoprotein (AFP), VEGF, ED-B fibronectin, EGP-1, EGP
- the Topi ADCs that can be co-administered in the methods provided herein comprise an antibody that binds a tumor antigen selected from CEACAM5 (NCBI Gene ID: 1048), CEACAM6 (NCBI Gene ID: 4680), CD74 (NCBI Gene ID: 972), CD19 (NCBI Gene ID: 930), CD20 (NCBI Gene ID: 931), CD22 (NCBI Gene ID: 933), CSAp (NCBI Gene ID: 126731), HLA-DR, HLA-G, MUC5ac (NCBI Gene ID: 4586), and AFP (NCBI Gene ID: 174).
- a tumor antigen selected from CEACAM5 (NCBI Gene ID: 1048), CEACAM6 (NCBI Gene ID: 4680), CD74 (NCBI Gene ID: 972), CD19 (NCBI Gene ID: 930), CD20 (NCBI Gene ID: 931), CD22 (NCBI Gene ID: 933), CSAp (NCBI Gene ID: 126731), HLA-
- the Topi ADCs that can be used to perform the methods provided herein comprise an antibody selected from gemtuzumab, brentuximab, belantamab, camidanlumab. trastuzumab, inotuzumab, glembatumumab, anetumab, mirvetuximab, depatuxizumab, vadastuximab, labetuzumab, ladiratuzumab, loncastuximab, patritumab, lifastuzumab, indusatumab, polatuzumab, pinatuzumab, coltuximab, upifitamab, indatuximab, milatuzumab, rovalpituzumab, enfortumab, tisotumab, tusamitamab. disitamab, telisotuzumab, and antigen-
- the Topi ADC that can be used in the methods provided herein comprise an antibody selected from hLLl (anti-CD74; USPN 7,312,318), 11LL2 (anti-CD22; USPN 7,074,403), hRFB4 (anti-CD22), hPAM4 (anti-MUC5ac; USPN 7,282,567), hMN-3 (anti- NOTCH3; USPN 7,541,440), hMN-14 (labetuzumab; anti-CEAC AM5 ; USPN 6,676,924); hMN15 (anti-CEACAM6; USPN 7,541,440); hA19 (anti-CD19; USPN 7,109,304), hA20 (anti- CD22; USPN 7,251,164), hMu-9 (anti-CSAp; USPN 7,387,773), hL243 (anti-HLA-DR; USPN 7,612,180), and hIMMU-31 (anti- AFP; USPN 7,
- the Topi ADC comprises a linker connecting a topoisomerase I inhibitor payload with a tumor antigen targeted antibody.
- the linker is non-cleavable (e.g., a maleimidocaproyl or maleimidomethyl cyclohexane- 1 -carboxylate linker).
- the linker is cleavable.
- the linker is acid clcav blc (c.g., a hydrazone linker).
- the cleavable linker is reducible (e.g., a disulphide linker).
- the linker is protease cleavable (e.g., a dipeptide or tetrapeptide linker). In some embodiments, the linker is selected from linkers disclosed in USPN 7,999,083 (e.g., CL2A, CL6, CL7, CLX, or CLY). In some embodiments, the linker is CL2A.
- the Topi ADC comprises a topoisomerase I inhibitor that is a camptothecin (e.g., an irinotecan, topotecan, belotecan, or exatecan derivative, such as Dxd or SN38).
- a camptothecin e.g., an irinotecan, topotecan, belotecan, or exatecan derivative, such as Dxd or SN38.
- the topoisomerase I inhibitor in the Topi ADC is Dxd.
- the topoisomerase I inhibitor in the Topi ADC is SN38.
- the topoisomerase I inhibitor in the Topi ADC is not a camptothecin.
- the non-camptothecin topoisomerase I inhibitor is selected from an indenosinoquinoline (e.g., indeno[l,2-c]isoquinoline, NSC314622, indotecan (LMP-400), indimitecan (LMP-776)), a phenanthridine (e.g., topovale (ARC-111), and a indolocarbazole (e.g., BE-13793C).
- indenosinoquinoline e.g., indeno[l,2-c]isoquinoline, NSC314622, indotecan (LMP-400), indimitecan (LMP-776)
- a phenanthridine e.g., topovale (ARC-111)
- a indolocarbazole e.g., BE-13793C
- the Topi ADC has a structural formula of mAb-CL2A-SN-38, with a structure represented by:
- the Topi ADC that can be used in a method provided herein includes an antibody targeting carcinocmbryonic antigen-related cell adhesion molecule 5 (CEACAM5; CD66e; NCBI Gene ID: 1048).
- CEACAM5 an antibody targeting carcinocmbryonic antigen-related cell adhesion molecule 5
- the CEACAM5 antibody is hMN-14 (e.g., as described in WO1996011013).
- the anti-CEACAM5 ADC is as described in W02010093395 (anti-CEACAM5-CL2A-SN38).
- the Topi ADC is labetuzumab govitecan (IMMU-130).
- the Topi ADC that can be used in a method provided herein comprises an antibody targeting MHC class II cell surface receptor encoded by the human leukocyte antigen complex (HLA-DR).
- HLA-DR antibody is hL243 (e.g., as described in W02006094192).
- the HLA-DR-ADC is as described in W02010093395 (anti-HLA-DR-CL2A-SN38).
- the antibody and/or fusion protein provided herein is administered with the HLA-DR-ADC IMMU-140.
- Topi ADCs that can be co-administered in the methods provided herein are described in WO21225892 (Shanghai Escugen Biotechnology).
- the Topi ADC comprises a linker-payload conjugate having a structure represented by: attached to a tumor antigen targeted antibody.
- Topi ADCs that can be co-administered in the methods provided herein are described in US20210101906 (Sichuan Kelun Pharmaceutical).
- the Topi ADC comprises a linker-payload conjugate (TLO35) having a structure represented by: attached to a tumor antigen targeting antibody.
- TLO35 linker-payload conjugate
- Topi ADCs that can be co-administered in the methods provided herein are described in US2016297890 (Daiichi Sankyo).
- the Topi ADC comprises a linker-payload conjugate having a structure represented by: attached to a tumor antigen targeting antibody (e.g., trastuzumab).
- a tumor antigen targeting antibody e.g., trastuzumab
- the Topi ADC has a DAR of about 4.
- the Topi ADC is trastuzumab-deruxtecan (T- DXd).
- the adenosine pathway is one of several pathways that can promote tumor immune evasion in the tumor microenvironment. It is believed that the conversion of pro-inflammatory extracellular ATP into immunosuppressive adenosine (eADO) can favor tumor progression and escape from antitumor immunity.
- eADO immunosuppressive adenosine
- the role of the adenosine pathway in immuno-oncology is described, for example, in Allard et al., (2020) Nat Rev Clin Oncol 17, 611-629.
- Adenosine pathway inhibitors that can be co-administered in the methods provided herein can include, for example, inhibitors of CD39 (cctonuclcosidc triphosphate diphospohydrolasc-1; ENTPD1; NCBI Gene ID: 953); inhibitors of CD73 (exto-5’ -nucleotidase; NT5E; NCBI Gene ID: 4907), or antagonists of adenosine receptors, such as adenosine A 2A receptor (AD0RA2A; NCBI Gene ID: 135) or adenosine AIB receptor (AD0RA2B; NCBI Gene ID: 136).
- the adenosine pathway inhibitors that can be co-administered in the methods provided herein include Inhibitors of CD38 (cyclic ADP ribose hydrolase; NCBI Gene ID: 952).
- CD39 inhibitors that can be co-administered in the methods provided herein include small molecule inhibitors and large molecule inhibitors (e.g., anti-CD39 antibodies) of CD39.
- Exemplary CD39 inhibitors are described, for example in WO09095478, WO12085132, WO16073845, WO17157948, WO18049145, WO18065552, WO18065622, WO19027935, WO19178269, W021056610, WG21037037, WO21055329, WO21088838, and WO22111576.
- the CD39 inhibitor is selected from TTX-030 (AbbVie/Trishula), IPH5201 (AstraZeneca/Innate Pharma), SRF617 (Surface Oncology), CD39 ASO (Secama Pharmaceuticals), JS-019 (Shanghai Junshi Biosciences); anti-CD39 (Arcus Biosciences), ES002 (Elpiscience Biopharmaceuticals), and CD39xPDl (Biotheus).
- CD73 inhibitors that can be co-administered in the methods provided herein include small molecule inhibitors and large molecule inhibitors (e.g., anti-CD73 antibodies) of CD73.
- CD73 inhibitors that can be used in the methods provided herein are described, for example, in US Patent No. 11,001,603, the compounds of which are hereby incorporated by reference herein. Additional illustrative CD73 inhibitors that can be co-administered in the methods provided herein are described, for example in WO15164573, W016055609, W016075099, WO16081746, WO16081748, W017064043, WO17098421, W017100670, WO17118613, WO17153952, WO18013611, WO18067424, WO18065627, WO18094148, WO18110555, WO18119284, WO18137598, WO18183635, W018208980, WO18208727, WO18215535, WO18237173, WO18237157, WO19053617, W019090111, WO19129059, WO19168744, WO
- the CD73 inhibitor is quemliclustat (AB680, GS-0680), uliledlimab, mupadolimab, ORIC-533, ATG-037, PT-199, AK131, NZV93O, BMS-986179, or oleclumab.
- the CD73 inhibitor is oleclumab or quemliclustat. In some embodiments, the CD73 inhibitor is quemliclustat.
- the adenosine receptor antagonists that can be co-administered in the methods provided herein can be selective antagonists of adenosine A2A receptor (A2AR; ADORA2A) or A2B receptor (A2BR; ADORA2B), or dual A2A/2BR antagonists.
- the adenosine receptor antagonist is a selective A2aR antagonist.
- the adenosine receptor antagonist is an adenosine A2A receptor (A2AR; ADORA2A) selective antagonist selected from imaradenant (AstraZeneca), NIR178 (Novartis/Palobiofarma) ID11902 (Ildong), IN-A003 (Inno.n), NTI-55 (A2aR/TLR7, Nammi), TT-10 (Tarns Therapeutics), and TT-228 (Teon Therapeutics).
- the adenosine receptor antagonist is a selective A2BR antagonist.
- the adenosine receptor antagonist is an adenosine A2B receptor (A2BR; ADORA2B) selective antagonist selected from PBF-1129 (Palobiofarma) and TT-702 (Toon Therapeutics).
- A2BR adenosine A2B receptor
- ADORA2B adenosine A2B receptor
- the adenosine receptor antagonist is a dual A2A/2BR antagonist.
- the adenosine receptor antagonist is a dual adenosine A2A/A2B receptor antagonist selected from etrumadenant (AB928, Arcus Biosciences), INCB 106385 (Incyte), M1069 (Merck KGaA), A2aR/A2bR (Domain/Merck KgaA), HM87277 (Al/A2aR/A2bR, Hanmi Pharmaceutical), RVU-330 (Ryvu), and TT-53 (Tarns Therapeutics).
- the adenosine receptor antagonist is etrumadenant.
- the adenosine receptor antagonists can be small molecule antagonists or large molecule antagonists.
- adenosine receptor antagonists that can be co-administered in the methods provided herein are described, for example, in WO07103776, WO07134958, W008086201, W009009178, WG09033161, WO09037463, WO09037468, WO09037467, WO09055548, W009055308, W010008775, WO11027805, WO11050160, WO11055391, WO12135084, W014101120, WO14101113, WO14106861, WO15027431, W016081290, WO16126570, W016200717, WO16209787, W017008205, WO18013951, W019086074, WO2019118313, WO20053263, W020106558, W020103930, W020103939, W020106560, W020112706, W020112700, WO20216152
- adenosine receptor antagonists that can be used in the methods provided herein are described, for example, in US Patent No. 10,399,962, the compounds of which are hereby incorporated by reference.
- the adenosine pathway inhibitor is a dual antagonist of adenosine A2A receptor (A2AR; ADORA2A) and A2B receptor (A2BR; ADORA2B).
- the adenosine pathway inhibitor is etrumadenant (AB928; GS-0928), taminadenant, TT-10, TT-4, or M 1069.
- the adenosine pathway inhibitor is etrumadenant.
- Etrumadenant is a small molecule dual antagonist of both A2AR and A2BR that can inhibit the adcnosinc-drivcn impairment of tumor-infiltrating lymphocytes (mainly through A2AR on CD8+ T cells and NK cells) and myeloid cells (through A2BR on dendritic cells and macrophages) in the absence of any agonist activity.
- Etrumadenant can achieve high penetration of tumor tissue, robust potency in the presence of high adenosine concentrations, and shows low nonspecific protein binding.
- the combination treatment methods provided herein comprise coadministering a PD(L)1 inhibitor.
- PD(L)1 inhibitors that can be co-administered can include small molecule PD(L)1 inhibitors and large molecule PD(L)1 inhibitors (e.g., anti-PD(L)-l antibodies).
- Exemplary small molecule PD(L)1 inhibitors that can be co-administered in the methods provided herein include, for example, CA-170, GS-4224, GS-4416 and lazertinib (GNS-1480; PD- Ll/EGFR). Additional illustrative small molecule PD(L)1 inhibitors are described, for example, in Guzik et al. (2019) Molecules 24(11), 2071.
- anti-PD-(L)l antibodies that can be co-administered in the method provided herein include, for example, pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD-(L)! antibody is zimberelimab.
- Additional illustrative anti-PD-(L)l antibodies that can be co-administered in the methods provided herein include pembrolizumab, nivolumab, cemiplimab, pidilizumab, AMP- 224, MEDI0680 (AMP-514), spartalizumab, atezolizumab, avelumab, durvalumab, BMS-936559, cosibelimab (CK-301), sasanlimab (PF-06801591), tislelizumab (BGB-A317), GLS-010 (WBP- 3055), AK-103 (HX-008), AK-105, CS-1003, HLX-10, retifanlimab (MGA-012), BI-754091, balstilimab (AGEN-2034), AMG-404, toripalimab (JS-001), cetrelimab (JNJ-637232
- anti-TIGIT antibodies that can be co-administered in the methods provided herein include, for example, tiragolumab, vibostolimab, domvanalimab, AB3O8, AK127, BMS- 986207, ralzapastotug, and etigilimab.
- the anti-TIGIT antibody is domvanalimab.
- the anti-TIGIT antibody is AB308.
- the anti-TIGIT antibody is ralzapastotug.
- the anti-TIGIT antibody is an Fc-silent antibody. In some embodiments, the anti-TIGIT antibody comprises one or more mutations in the Fc region to reduce, prevent, or eliminate binding to an Fc receptor. In some embodiments, the anti-TIGIT antibody comprises one or more mutations in the Fc region to reduce, prevent, or eliminate binding to FcyR. In some embodiments, any of the antibodies disclosed herein comprise one or more mutations in the Fc region to reduce, prevent, or eliminate binding to FcyRIIIA. In some embodiments, any of the antibodies disclosed herein comprise one or more mutations in the Fc region to reduce, prevent, or eliminate binding to FcyRIV.
- the anti-TIGIT antibody comprises one or more mutations in the Fc region to reduce, prevent, or eliminate ADCC, ADCP, and/or CDC. In some embodiments, the anti-TIGIT antibody comprises one or more substitutions in the Fc region to reduce, prevent, or eliminate binding to Fc receptors, wherein the one or more substitutions occur at EU index positions 228, 233, 234, 235, 235, 235, 236, 237, 265, 297, 322, 327, 328, 330, 331, and any combination thereof.
- the anti-TIGIT antibody comprises one or more substitutions in the Fc region to reduce, prevent, or eliminate binding to Fc receptors, wherein the one or more substitutions comprise S228P, E233P, E234A, L235A, E235E, E235F, G236R, G237A, D265A, N297A, K322A, A327G, E328R, A330S, P331S, and any combination thereof. Additional mutations in the Fc region that reduce, prevent, or eliminate binding to Fc receptors and alternative strategies for reducing, preventing, or eliminating binding to Fc receptors are described in, e.g., Saunders, Front Immunol.
- the anti-TIGIT antibody is an Fc-enabled antibody. In some embodiments, the anti-TIGIT antibody comprises one or more mutations in the Fc region to enable or enhance binding to an Fc receptor. In some embodiments, the anti-TIGIT antibody comprises one or more mutations in the Fc region to enable or enhance binding to FcyR. In some embodiments, any of the antibodies disclosed herein comprise one or more mutations in the Fc region to enable or enhance binding to FcyRIIIA. In some embodiments, any of the antibodies disclosed herein comprise one or more mutations in the Fc region to enable or enhance binding to FcyRIV.
- the anti-TIGIT antibody comprises one or more mutations in the Fc region to enable or enhance ADCC, ADCP, and/or CDC.
- the anti-PD- (E)l antibody comprises one or more substitutions in Fc region to enhance or enable binding to Fc receptors, wherein the one or more substitutions occur at EU index positions 234, 235, 236, 239, 243, 247, 267, 268, 292, 298, 300, 305, 324, 326, 330, 332, 333, 334, 339, 345, 396, 430, and any combination thereof.
- the anti-TIGIT antibody comprises one or more substitutions in the Fc region to enhance or enable binding to Fc receptors, wherein the one or more substitutions comprise F234L, L235V, G236A, S239D, F243L, P247I, S267E, H268E, R292P, S298A, Y300L, V305I, S324T, K326W, A33OE, I332E, E333A, E333S, K334A, A339Q, E345G, P396L, E430G, and any combination thereof.
- substitutions comprise F234L, L235V, G236A, S239D, F243L, P247I, S267E, H268E, R292P, S298A, Y300L, V305I, S324T, K326W, A33OE, I332E, E333A, E333S, K334A, A339Q, E345G
- the Fc-enabled antibody comprises a modified IgGl domain characterized by substitutions at S239D, A33OL, and I332E (Eu numbering).
- the anti-TIGIT antibody contains or has a glycoform perturbation.
- the anti-TIGIT antibody contains or has an N-linked Fc glycosylation.
- the anti-TTGTT antibody contains or has sialylation, galactosylation, bisecting sugars, fucosylation, or any combination thereof. Additional mutations in the Fc region that enhance or enable binding to Fc receptors and alternative strategies for enhancing or enabling binding to Fc receptors are described in Saunders, 2019.
- adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor
- the methods provided herein include treating a Trop-2 positive cancer comprising co-administering to a subject an effective amount of: a) an anti-Trop-2 antibody-drug conjugate (ADC); and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- the methods provided herein include treating the recurrence or metastasis of a Trop-2 positive cancer comprising co-administering to a subject an effective amount of: a) an anti-Trop-2 antibody-drug conjugate (ADC); and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- the methods provided herein include preventing or delaying the recurrence or metastasis of a Trop-2 positive cancer comprising co-administering to a subject an effective amount of: a) an anti-Trop-2 antibody-drug conjugate (ADC); and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- ADC anti-Trop-2 antibody-drug conjugate
- the adenosine pathway inhibitor is a plurality of adenosine pathway inhibitors.
- the plurality of adenosine pathway inhibitors comprises a CD73 inhibitor and an adenosine receptor antagonist.
- the CD73 inhibitor is quemliclustat and the adenosine receptor antagonist is etrumadenant.
- the methods provided herein are for treating a Trop-2 positive cancer comprising co-administering to a subject an effective amount of: a) an anti-Trop-2 antibody-drug conjugate (ADC); and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- ADC anti-Trop-2 antibody-drug conjugate
- adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor
- TA + tumor antigen positive
- methods of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of a tumor antigen positive (TA + ) cancer comprising co-administering to a subject an effective amount of: a) a tumor antigen (TA) targeted antibody-drug conjugate (ADC) comprising a topoisomerase I inhibitor (Topi ADC); and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- TA tumor antigen
- ADC targeted antibody-drug conjugate
- Topici ADC topoisomerase I inhibitor
- an adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor
- the methods provided herein include treating a tumor antigen positive (TA+) cancer comprising co-administering to a subject an effective amount of: a) a tumor antigen targeted antibody-drug conjugate (ADC) comprising a topoisomerase I inhibitor; and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- ADC tumor antigen targeted antibody-drug conjugate
- adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor
- the methods provided herein include treating the recurrence or metastasis of a tumor antigen positive cancer comprising co-administering to a subject an effective amount of: a) a tumor antigen targeted antibody-drug conjugate (ADC) comprising a topoisomerase I inhibitor; and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- ADC tumor antigen targeted antibody-drug conjugate
- an adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor.
- the methods provided herein include preventing or delaying the recurrence or metastasis of a tumor antigen positive cancer comprising co-administering to a subject an effective amount of: a) a tumor antigen targeted antibody-drug conjugate (ADC) comprising a topoisomerase I inhibitor; and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- ADC tumor antigen targeted antibody-drug conjugate
- an adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor.
- the methods provided herein are for treating a tumor antigen positive cancer comprising co-administering to a subject an effective amount of: a) a tumor antigen targeted antibody-drug conjugate (ADC) comprising a topoisomerase I inhibitor; and b) an adenosine pathway inhibitor (e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor).
- ADC tumor antigen targeted antibody-drug conjugate
- adenosine pathway inhibitor e.g., CD39 inhibitor, CD73 inhibitor, or A2R inhibitor
- provided herein are methods of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of a tumor antigen (e.g., Trop-2) positive cancer comprising co-administering to a subject the combinations provided herein.
- the treatment methods provided herein mitigate the occurrence or recurrence of a tumor antigen (e.g., Trop-2) positive cancer by administering the combinations provided herein.
- the treatment methods provided herein reduce the occurrence or recurrence of a tumor antigen (e.g., Trop-2) positive cancer by administering the combinations provided herein.
- the treatment methods provided herein prevent the occurrence or recurrence of a tumor antigen (e.g., Trop-2) positive cancer by administering the combinations provided herein. In some embodiments, the treatment methods provided herein delay the occurrence or recurrence of a tumor antigen (e.g., Trop-2) positive cancer by administering the combinations provided herein.
- a tumor antigen e.g., Trop-2
- the treatment methods provided herein delay the occurrence or recurrence of a tumor antigen (e.g., Trop-2) positive cancer by administering the combinations provided herein.
- the methods provided herein further comprise co-administering an additional therapeutic agent or therapeutic modality, or a combination thereof.
- the methods provided herein comprise co-administering one, two, or three additional therapeutic agents or therapeutic modalities, or combinations thereof.
- the additional therapeutic agent comprises chemotherapy (e.g., a chemotherapy treatment of physician’s choice, or an indicated standard of care for a specific therapeutic setting).
- the additional therapeutic agent comprises an immune checkpoint inhibitor (CPI; e.g., anti-CTLA4 antibody, anti-PD(L)! antibody, anti-TTGTT antibody).
- CPI immune checkpoint inhibitor
- the additional therapeutic agent comprises an anti-PD(L)l antibody (e.g., an anti- PD-1 antibody or an anti-PD-Ll antibody), and optionally an anti-TIGIT antibody.
- the additional therapeutic agent comprises an anti-PD-(L)l antibody.
- the additional therapeutic agent comprises an anti-TIGIT antibody.
- the additional therapeutic agent comprises an anti-PD-(Ll) antibody and an anti- TIGIT antibody.
- the additional therapeutic agent comprises a) zimberelimab and domvanalimab, b) zimberelimab and AB308, c) atezolizumab and tiragolumab, d) pembrolizumab and vibostolimab, e) pembrolizumab and domvanalimab, f) pembrolizumab and AB3O8, g) MK-7684A (pembrolizumab/vibostolimab coformulation), h) durvalumab and domvanalimab, i) zimberelimab and ralzapastotug, or j) pembrolizumab and ralzapastotug.
- the additional therapeutic agent comprises zimberelimab and domvanalimab. In some embodiments, the additional therapeutic agent comprises zimberelimab and ralzapastotug. In some embodiments, the additional therapeutic modality comprises surgery or radiation therapy.
- the subject is a cancer patient.
- the subject is a human cancer patient.
- the subject is treatment naive.
- the subject e.g., human cancer patient
- has not received a taxane e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel
- a checkpoint inhibitor therapy e.g., anti-CTLA4 antibody, anti-PD(L)l antibody
- the subject e.g. human cancer patient
- a topoisomerase I inhibitor therapy e.g., irinotecan
- the subject has received one or more lines of previous anti-cancer therapy before administration of a combination provided herein.
- the subject e.g., human (m)CRPC patient
- a new hormonal agent NHA; first- or second-generation nonsteroidal antiandrogens, e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- NHA new hormonal agent
- first- or second-generation nonsteroidal antiandrogens e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- the subject e.g., human (m)CRPC patient
- has shown disease progression after prior NHA treatment first- or second-generation non-steroidal antiandrogens, e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- first- or second-generation non-steroidal antiandrogens e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- the subject has received a prior anticancer therapy selected from surgery, radiation therapy, chemotherapy (including NHA therapy), checkpoint inhibitor therapy (e.g. anti-PD(L)l antibody).
- the subject has a cancer that is resistant or refractory to one or more anti-cancer therapies.
- the cancer is resistant or refractory to one or more anti-cancer therapies selected from radiation therapy, chemotherapy (including NHA therapy), and checkpoint inhibitor therapy (e.g., anti- PD(L)1 antibody).
- the subject e.g., human (m)CRPC patient
- first- or second-generation non-steroidal antiandrogens e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- a taxane e.g., docetaxel, cabazitaxel
- checkpoint inhibitor e.g., anti-CTLA-4 antibody; anti-PD(L)l antibody
- a topoisomerase I inhibitor e.g., irinotecan
- the subject e.g., human (m)NSCLC patient
- the subject e.g., human (m)NSCLC patient
- has shown disease progression after checkpoint inhibitor therapy e.g., anti-PD-(L)l antibody or anti-CTLA4 antibody therapy.
- the subject has progressed after platinum-based chemotherapy and checkpoint inhibitor therapy (e.g., anti-PD-(L)l antibody or anti-CTLA4 antibody therapy), received either in combination or sequentially in any order.
- the subject e.g., human (m)NSCLC patient
- a tyrosine kinase inhibitor therapy targeting specific genomic alterations in a cancer.
- Exemplary tyrosine kinase inhibitors that are useful to address specific genomic alterations in lung cancer include gefitinib, erlotinib, afatinib, dacomitinib, neratinib, osimertinib, rociletinib, olmutinib, ASP8273 (Astellas), moninib, PF-06747775 (Pfizer), avitinib, and HS- 10296 (Jiangsu Hansoh).
- the subject e.g., human (m)CRPC or (m)NSCLC patient
- tumor antigen e.g., Trop-2
- tumor antigen expression levels e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or next-generation DNA sequencing.
- the subject is a human prostate cancer patient.
- the human patient has castrate resistant prostate cancer (CRPC).
- the human patient has metastatic CRPC (mCRPC).
- the human patient with CRPC or mCRPC ((m)CRPC) is treatment naive.
- the human patient with (m)CRPC has not received a taxane (e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel) therapy before administration of a combination therapy provided herein.
- a taxane e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel
- the human patient with (m)CRPC has not received a checkpoint inhibitor therapy (e.g., anti-CTLA4 antibody, anti-PD(L)l antibody) before administration of a combination therapy provided herein.
- a checkpoint inhibitor therapy e.g., anti-CTLA4 antibody, anti-PD(L)l antibody
- the human patient with (m)CRPC has not received a topoisomerase I inhibitor therapy (e.g., irinotecan) before administration of a combination therapy provided herein.
- the human patient with (m)CRPC has received one or more lines of anti-cancer therapy before administration of a combination provided herein.
- the human patient with (m)CRPC has shown disease progression on one or more lines of anti-cancer therapy before administration of a combination provided herein.
- the human patient with (m)CRPC has received a new hormonal agent (NHA; first- or second-generation non-steroidal antiandrogens, abiraterone) (NHA experienced) prior to administration of a combination therapy provided herein.
- NHA new hormonal agent
- the human patient with (m)CRPC has shown disease progression after prior NHA treatment (first- or second-generation non-steroidal antiandrogens, e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof) before administration of a combination therapy provided herein
- the human patient with (m)CRPC has received a prior anti-cancer therapy selected from surgery, radiation therapy, chemotherapy (including NHA therapy), checkpoint inhibitor therapy (e.g., anti-CTLA4 antibody, anti-PD(L)l antibody).
- the human patient with (m)CRPC has a cancer that is resistant or refractory to one or more anti-cancer therapies.
- the cancer is resistant or refractory to one or more anti-cancer therapies selected from radiation therapy, chemotherapy (including NHA therapy), and checkpoint inhibitor therapy (e.g., anti-CTLA4 antibody, anti- PD(L)1 antibody).
- the human patient with (m)CRPC has received a prior NHA treatment (first- or second-generation non-steroidal antiandrogens, e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof) (NHA experienced) and has not received a taxane (e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel) or checkpoint inhibitor (e.g., anti-CTLA-4 antibody; anti-PD(L)l antibody) (taxane and CPI naive) before administration of a combination therapy provided herein.
- first- or second-generation non-steroidal antiandrogens e.
- the human patient with (m)CRPC has received a prior NHA treatment (first- or second-generation nonsteroidal antiandrogens, e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof) (NHA experienced) and has not received a taxane (e.g., paclitaxel, nab- paclitaxel (ABRAXANE®), docetaxel, cabazitaxel), checkpoint inhibitor (e.g., anti-CTLA-4 antibody; anti-PD(L)l antibody), or topoisomerase I inhibitor (e.g., irinotecan) before administration of a combination therapy provided herein.
- first- or second-generation nonsteroidal antiandrogens e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- NHA experienced and has not received a taxane (e.g., pac
- the human patient with (m)CRPC has shown disease progression after prior NHA treatment (first- or second- generation non-steroidal antiandrogens, e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof) and has not received a taxane (e.g., paclitaxel, nab- paclitaxel (ABRAXANE®), docetaxel, cabazitaxel), checkpoint inhibitor (e.g., anti-CTLA-4 antibody; anti-PD(L)l antibody), or a topoisomerase I inhibitor (e.g., irinotecan) before administration of a combination therapy provided herein.
- first- or second- generation non-steroidal antiandrogens e.g., abiraterone, enzalutamide, darolutamide, apalutamide, or combinations thereof
- a taxane e.g., paclitaxel, na
- the human patient with (m)CRPC has histologically confirmed adenocarcinoma of the prostate and metastatic castrate resistant with tumor progression while on androgen deprivation therapy (ADT; including orchiectomy) with castrate levels of serum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by PSA and/or radiographic criteria according to PCWG3.
- ADT on androgen deprivation therapy
- the human patient (m)CRPC has metastatic castrate resistant adenocarcinoma of the prostate with tumor progression while on androgen deprivation therapy (e.g., including orchiectomy) with castrate levels of serum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by prostate specific antigen (PSA) and/or radiographic criteria according to The Prostate Cancer Working Group 3 (PCWG3) and measurable or non-measurable disease per the Response Evaluation Criteria in Solid Tumors (RECIST) v 1.1.
- the human patient with (m)CRPC has Eastern Cooperative Oncology Group performance status 0 or 1 with a life expectancy >3 months.
- the human patient with (m)CRPC has been tested for tumor antigen (e.g., Trop-2) expression levels (e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or next-generation DNA sequencing).
- tumor antigen e.g., Trop-2
- expression levels e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or next-generation DNA sequencing.
- the subject is a human lung cancer patient.
- the human patient has non-small cell lung cancer (NSCLC).
- NSCLC non-small cell lung cancer
- the NSCLC is squamous.
- the NSCLC is non-squamous.
- the lung cancer does not have an actionable genomic alteration (e.g., an EGFR or ALK mutation, insertion, deletion, or the like) for which treatment with a targeted therapy (e.g., a targeted tyrosine kinase inhibitor therapy) is indicated (actionable genomic alteration).
- a targeted therapy e.g., a targeted tyrosine kinase inhibitor therapy
- Exemplary actionable genomic alterations and targeted tyrosine kinase inhibitor therapies are described, e.g., in Sullivan and Planchard Front.
- the human NSCLC patient has shown disease progression after platinum-based chemotherapy. In some embodiments, the human NSCLC patient has shown disease progression after checkpoint inhibitor therapy (e.g., anti-PD- (L)l antibody or anti-CTLA4 antibody therapy). In some embodiments, the human NSCLC patient has progressed after platinum-based chemotherapy and checkpoint inhibitor therapy (e.g., anti-PD-(L)! antibody or anti-CTLA4 antibody therapy), received either in combination or sequentially in any order. In some embodiments, the human NSCLC patient has progressed after a tyrosine kinase inhibitor therapy targeting specific genomic alterations in a cancer.
- checkpoint inhibitor therapy e.g., anti-PD- (L)l antibody or anti-CTLA4 antibody therapy
- checkpoint inhibitor therapy e.g., anti-PD-(L)! antibody or anti-CTLA4 antibody therapy
- Exemplary tyrosine kinase inhibitors that are useful to address specific genomic alterations in lung cancer (e.g., EGFR mutations or deletions) include gefitinib, erlotinib, afatinib, dacomitinib, neratinib, osimertinib, rociletinib, olmutinib, ASP8273 (Astellas), Feliartinib, PF-06747775 (Pfizer), avitinib, and HS- 10296 (Jiangsu Hansoh).
- the combinations provided herein are administered in a neoadjuvant setting (e.g., in preparation for surgery or radiation therapy).
- the combinations provided herein are administered in an adjuvant setting (e.g., following a primary treatment such as surgery or radiation therapy).
- the combinations provided herein are administered in a therapeutic setting (e.g., as the primary therapy).
- the combinations provided herein are administered in a maintenance setting.
- the cancer is a hematological cancer.
- the cancer includes a solid tumor.
- the cancer includes a malignant tumor.
- the cancer includes a metastatic cancer.
- the cancer is resistant or refractory to one or more anticanccr therapies.
- greater than about 50% of the cancer cells delectably express one or more cell surface immune checkpoint receptors (e.g., so-called “hot” cancer or tumor).
- greater than about 1% and less than about 50% of the cancer cells detectably express one or more cell surface immune checkpoint receptors (e.g., so called “warm” cancer or tumor).
- less than about 1% of the cancer cells detectably express one or more cell surface immune checkpoint receptors (e.g., so called “cold” cancer or tumor).
- the cancer is a hematological cancer, e.g., a leukemia (e.g., Acute Myelogenous Leukemia (AML), Acute Lymphoblastic Leukemia (ALL), B-cell ALL, Myelodysplastic Syndrome (MDS), myeloproliferative disease (MPD), Chronic Myelogenous Leukemia (CML), Chronic Lymphocytic Leukemia (CLL), undifferentiated leukemia), a lymphoma (e.g., small lymphocytic lymphoma (SLL), mantle cell lymphoma (MCL), follicular lymphoma (FL), T-cell lymphoma, B-cell lymphoma, diffuse large B-cell lymphoma (DLBCL), marginal zone lymphoma (MZL), Waldestrom’s macroglobulinemia (WM)) and/or a myeloma (e.g., multiple myeloma (e.g., multiple mye
- the cancer is an epithelial tumor (e.g., a carcinoma, a squamous cell carcinoma, a basal cell carcinoma, a squamous intraepithelial neoplasia), a glandular tumor (e.g., an adenocarcinoma, an adenoma, an adenomyoma), a mesenchymal or soft tissue tumor (e.g., a sarcoma, a rhabdomyosarcoma, a leiomyosarcoma, a liposarcoma, a fibrosarcoma, a dermatofibrosarcoma, a neurofibrosarcoma, a fibrous histiocytoma, an angiosarcoma, an angiomyxoma, a leiomyoma, a chondroma, a chondrosarcoma, an alve
- a glandular tumor e.g., an
- the cancer includes a solid tumor in or arising from a tissue or organ, such as:
- bone e.g., adamantinoma, aneurysmal bone cysts, angiosarcoma, chondroblastoma, chondroma, chondromyxoid fibroma, chondrosarcoma, chordoma, dedifferentiated chondrosarcoma, enchondroma, epithelioid hemangioendothelioma, fibrous dysplasia of the bone, giant cell tumour of bone, haemangiomas and related lesions, osteoblastoma, osteochondroma, osteosarcoma, osteoid osteoma, osteoma, periosteal chondroma, Desmoid tumor, Ewing sarcoma);
- bone e.g., adamantinoma, aneurysmal bone cysts, angiosarcoma, chondroblastoma, chondroma, chondromyxoid fibroma, chondrosarcoma, chord
- lips and oral cavity e.g., odontogenic ameloblastoma, oral leukoplakia, oral squamous cell carcinoma, primary oral mucosal melanoma
- salivary glands e.g., pleomorphic salivary gland adenoma, salivary gland adenoid cystic carcinoma, salivary gland mucoepidermoid carcinoma, salivary gland Warthin's tumors
- esophagus e.g., Barrett's esophagus, dysplasia and adenocarcinoma
- stomach e.g., gastric adenocarcinoma, primary gastric lymphoma, gastrointestinal stromal tumors (GISTs), metastatic deposits, gastric carcinoids, gastric sarcomas, neuroendocrine carcinoma, gastric primary squamous cell carcinoma, gastric adenoacanthomas), intestines and smooth muscle (e.g., intravenous leiomyomatosis), colon (e.g., colorectal adenocarcinoma), rectum, anus;
- stomach e.g., gastric adenocarcinoma, primary gastric lymphoma, gastrointestinal stromal tumors (GISTs), metastatic deposits, gastric carcinoids, gastric sarcomas, neuroendocrine carcinoma, gastric primary squamous cell carcinoma, gastric adenoacanthomas), intestines and smooth muscle (e.g., intravenous leiomyomatosis), colon (e.g
- pancreas e.g., serous neoplasms, including microcystic or macrocystic serous cystadenoma, solid serous cystadenoma, Von Hippel-Landau (VHL)-associated serous cystic neoplasm, serous cystadenocarcinoma; mucinous cystic neoplasms (MCN), intraductal papillary mucinous neoplasms (IPMN), intraductal oncocytic papillary neoplasms (IOPN), intraductal tubular neoplasms, cystic acinar neoplasms, including acinar cell cystadenoma, acinar cell cystadcnocarcinoma, pancreatic adenocarcinoma, invasive pancreatic ductal adenocarcinomas, including tubular adenocarcinoma, adenosquamous carcinoma, colloid carcinoma, medullary carcinoma, he
- neuro-endocrine e.g., adrenal cortical carcinoma, carcinoid tumors, phaeochromocytoma, pituitary adenomas
- thyroid e.g., anaplastic (undifferentiated) carcinoma, medullary carcinoma, oncocytic tumors, papillary carcinoma, adenocarcinoma);
- liver e.g., adenoma, combined hepatocellular and cholangiocarcinoma, fibrolamellar carcinoma, hepatoblastoma, hepatocellular carcinoma, mesenchymal, nested stromal epithelial tumor, undifferentiated carcinoma; hepatocellular carcinoma, intrahepatic cholangiocarcinoma, bile duct cystadenocarcinoma, epithelioid hemangioendothelioma, angiosarcoma, embryonal sarcoma, rhabdomyosarcoma, solitary fibrous tumor, teratoma, York sac tumor, carcinosarcoma, rhabdoid tumor);
- kidney e.g., ALK-rearranged renal cell carcinoma, chromophobe renal cell carcinoma, clear cell renal cell carcinoma, clear cell sarcoma, metanephric adenoma, metanephric adenofibroma, mucinous tubular and spindle cell carcinoma, nephroma, nephroblastoma (Wilms tumor), papillary adenoma, papillary renal cell carcinoma, renal oncocytoma, renal cell carcinoma, succinate dehydrogenase-deficient renal cell carcinoma, collecting duct carcinoma);
- ALK-rearranged renal cell carcinoma e.g., ALK-rearranged renal cell carcinoma, chromophobe renal cell carcinoma, clear cell renal cell carcinoma, clear cell sarcoma, metanephric adenoma, metanephric adenofibroma, mucinous tubular and spindle cell carcinoma, nephroma, nephroblastoma (Wil
- breast e.g. , invasive ductal carcinoma, including without limitation, acinic cell carcinoma, adenoid cystic carcinoma, apocrine carcinoma, cribriform carcinoma, glycogen-rich/clear cell, inflammatory carcinoma, lipid-rich carcinoma, medullary carcinoma, metaplastic carcinoma, micropapillary carcinoma, mucinous carcinoma, neuroendocrine carcinoma, oncocytic carcinoma, papillary carcinoma, sebaceous carcinoma, secretory breast carcinoma, tubular carcinoma; lobular carcinoma, including without limitation, pleomorphic carcinoma, signet ring cell carcinoma);
- peritoneum e.g., mesothelioma; primary peritoneal cancer
- female sex organ tissues including ovary (e.g., choriocarcinoma, epithelial tumors, germ cell tumors, sex cord-stromal tumors), Fallopian tubes (e.g., serous adenocarcinoma, mucinous adenocarcinoma, endometrioid adenocarcinoma, clear cell adenocarcinoma, transitional cell carcinoma, squamous cell carcinoma, undifferentiated carcinoma, Mullerian tumors, adenosarcoma, leiomyosarcoma, teratoma, germ cell tumors, choriocarcinoma, trophoblastic tumors), uterus (e.g., carcinoma of the cervix, endometrial polyps, endometrial hyperplasia, intraepithelial carcinoma (EIC), endometrial carcinoma (e.g., endometrioid carcinoma, serous carcinoma, clear cell carcinoma, mucinous carcinoma
- male sex organ tissues including prostate, testis (e.g., germ cell tumors, spermatocytic seminoma), penis;
- bladder e.g., squamous cell carcinoma, urothelial carcinoma, bladder urothelial carcinoma
- brain e.g., gliomas (e.g., astrocytomas, including non-infiltrating, low-grade, anaplastic, glioblastomas; oligodendrogliomas, ependymomas)), meningiomas, gangliogliomas, schwannomas (neurilemmomas), craniopharyngiomas, chordomas, Non-Hodgkin lymphomas (NHLs), indolent non-Hodgkin’s lymphoma (iNHL), refractory iNHL, pituitary tumors;
- gliomas e.g., astrocytomas, including non-infiltrating, low-grade, anaplastic, glioblastomas; oligodendrogliomas, ependymomas
- Meningiomas e.g., astrocytomas, including non-infiltrating, low-grade, anaplastic, glioblastomas;
- eye e.g., retinoma, retinoblastoma, ocular melanoma, posterior uveal melanoma, iris hamartoma
- eye e.g., retinoma, retinoblastoma, ocular melanoma, posterior uveal melanoma, iris hamartoma
- head and neck e.g., nasopharyngeal carcinoma, Endolymphatic Sac Tumor (ELST), epidermoid carcinoma, laryngeal cancers including squamous cell carcinoma (SCC) (e.g., glottic carcinoma, supraglottic carcinoma, subglottic carcinoma, transglottic carcinoma), carcinoma in situ, verrucous, spindle cell and basaloid SCC, undifferentiated carcinoma, laryngeal adenocarcinoma, adenoid cystic carcinoma, neuroendocrine carcinomas, laryngeal sarcoma), head and neck paragangliomas (e.g., carotid body, jugulotympanic, vagal); thymus (e.g., thymoma);
- heart e.g., cardiac myxoma
- lung e.g., small cell carcinoma (SCLC), non-small cell lung carcinoma (NSCLC), including squamous cell carcinoma (SCC), adenocarcinoma and large cell carcinoma, carcinoids (typical or atypical), carcinosarcomas, pulmonary blastomas, giant cell carcinomas, spindle cell carcinomas, pleuropulmonary blastoma);
- SCLC small cell carcinoma
- NSCLC non-small cell lung carcinoma
- SCC squamous cell carcinoma
- carcinoids typically or atypical
- carcinosarcomas pulmonary blastomas
- giant cell carcinomas e.g., giant cell carcinomas, spindle cell carcinomas, pleuropulmonary blastoma
- pleuropulmonary blastoma e.g., small cell carcinoma (SCLC), non-small cell lung carcinoma (NSCLC), including squamous cell carcinoma (SCC), adenocarcinoma and large cell carcinoma, carcinoids (typical or atypical), carcino
- lymphomas including Hodgkin’s lymphoma, non-Hodgkin’s lymphoma (NHL), indolent non-Hodgkin’s lymphoma (iNHL), refractory iNHL, Epstein-Barr virus (EBV)-associatcd lymphoproliferative diseases, including B cell lymphomas and T cell lymphomas e.g., Burkitt lymphoma; large B cell lymphoma, diffuse large B-cell lymphoma (DLBCL), mantle cell lymphoma, indolent B-cell lymphoma, low grade B cell lymphoma, fibrin-associated diffuse large cell lymphoma; primary effusion lymphoma; plasmablastic lymphoma; extranodal NK/T cell lymphoma, nasal type; peripheral T cell lymphoma, cutaneous T cell lymphoma, angioimmunoblastic T cell lymphoma; follicular
- central nervous system e.g., gliomas including astrocytic tumors (e.g., pilocytic astrocytoma, pilomyxoid astrocytoma, subependymal giant cell astrocytoma, pleomorphic xanthoastrocytoma, diffuse astrocytoma, fibrillary astrocytoma, gemistocytic astrocytoma, protoplasmic astrocytoma, anaplastic astrocytoma, glioblastoma (e.g., giant cell glioblastoma, gliosarcoma, glioblastoma multiforme) and gliomatosis cerebri), oligodendroglial tumors (e.g., oligodendroglioma, anaplastic oligodendroglioma), oligoastrocytic tumors (e.g., oligoastrocytoma, an astrocy
- skin e.g., clear cell hidradenoma, cutaneous benign fibrous histiocytomas, cylindroma, hidradenoma, melanoma (including cutaneous melanoma, mucosal melanoma), pilomatricoma, Spitz tumors); and
- soft tissues e.g. , aggressive angiomyxoma, alveolar rhabdomyosarcoma, alveolar soft part sarcoma, angiofibroma, angiomatoid fibrous histiocytoma, synovial sarcoma, biphasic synovial sarcoma, clear cell sarcoma, dermatofibrosarcoma protuberans, desmoid-type fibromatosis, small round cell tumor, desmoplastic small round cell tumor, elastofibroma, embryonal rhabdomyosarcoma, Ewing's tumors/primitive neurectodermal tumors (PNET), extraskeletal myxoid chondrosarcoma, extraskeletal osteosarcoma, paraspinal sarcoma, inflammatory myofibroblastic tumor, lipoblastoma, lipoma, chondroid lipoma, liposarcoma I malignant lipomatous tumors, lipo
- the cancer is positive for a tumor antigen selected from carbonic anhydrase IX, B7, CCCL19, CCCL21, CSAp, HER-2/neu, BrE3, CD1, CDla, CD2, CD3, CD4, CD5, CD8, CD11A, CD14, CD15, CD16, CD18, CD19, CD20 (e.g., C2B8, hA20, 1F5 MAbs), CD21, CD22, CD23, CD25, CD29, CD30, CD32b, CD33, CD37, CD38, CD40, CD40L, CD44, CD45, CD46, CD52, CD54, CD55, CD59, CD64, CD67, CD70, CD74, CD79a, CD80, CD83, CD95, CD126, CD133, CD138, CD147, CD154, CEACAM5, CEACAM-6, alpha-fetoprotein (AFP), VEGF, fibronectin splice variant, ED-B fibronectin
- a tumor antigen selected
- the cancer is positive for a tumor antigen selected from CD74, CD22, Trop-2, CEA, CSAp Mu-9, AFP, CC49, and PSMA.
- the cancer is Trop-2 positive.
- the Trop-2 positive cancer is a solid epithelial cancer.
- the solid epithelial cancer is selected from breast cancer (e.g., triple negative breast cancer (TNBC); HR positive/Her-2 negative (HR + /Her-2 ) breast cancer; HR positive/Her-2 low breast cancer), colorectal cancer, lung cancer, stomach cancer, urinary tract cancer, urothelial cancer, bladder cancer, renal cancer, pancreatic cancer, ovarian cancer, uterine cancer, esophageal cancer, and prostatic cancer.
- the prostatic cancer is castrate-resistant prostate cancer (CRPC).
- the lung cancer is non-small lung cancer (NSCLC).
- the Trop- 2 positive cancer is (i) unresectable, locally advanced or (ii) metastatic cancer (e.g., mCRPC or mNSCLC). In some embodiments, the Trop-2 positive cancer is resistant or refractive to one or more anti-cancer therapy.
- the Trop-2 positive cancer is prostate cancer. In some embodiments, the Trop-2 positive cancer is metatstatic prostate cancer. In some embodiments, the Trop-2 positive cancer is castrate resistant prostate cancer (CRPC). In some embodiments, the Trop-2 positive cancer is metatstatic castrate resistant prostate cancer (mCRPC).
- the Trop-2 positive cancer is metastatic castrate resistant adenocarcinoma of the prostate showing tumor progression on androgen deprivation therapy (e.g., including orchiectomy) with castrate levels of serum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by prostate specific antigen (PSA) and/or radiographic criteria according to The Prostate Cancer Working Group 3 (PCWG3); measurable or non-measurable disease per the Response Evaluation Criteria in Solid Tumors (RECIST) vl.l.
- tumor progression on androgen deprivation therapy e.g., including orchiectomy
- PSA prostate specific antigen
- PCWG3 Prostate Cancer Working Group 3
- RECIST Solid Tumors
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop2 antibody drug conjugate; and b) an adenosine pathway inhibitor.
- the anti-Trop-2 ADC comprises a topoisomerase I inhibitor.
- the topoisomerase inhibitor is a camptothecin analog.
- the camptothecin analog is an irinotecan derivative, a topotecan derivative, or an exatecan derivative.
- the camptothecin analog is SN38 or Dxd.
- the camptothecin analog is SN38. In some embodiments, the camptothecin analog is connected to an anti-Trop-2 antibody via a hydrolyzable linker. In some embodiments, the hydrolyzable linker is CL2A (e.g., as described in US 7,999,083). In some embodiments, the camptothecin analog is connected to an anti-Trop-2 antibody via a protease cleavable linker. In some embodiments, the anti-Trop-2 ADC has a structure mAb-CL2A-SN-38, represented by:
- the drug-antibody ratio (DAR) of CL2A-SN38 to anti-Trop-2 antibody in the anti-Trop2 ADC is between 7.0 and 8.0. In some embodiments, the DAR of CL2A-SN38 to anti-Trop-2 antibody in the anti-Trop-2 ADC is about 7.6. In some embodiments, the anti-Trop-2 ADC comprises the anti-Trop-2 antibody sacituzumab (hRS7, described, e.g., in W02003074566, Figures 3 and 4).
- the anti-Trop-2 ADC is selected from sacituzumab govitecan, datopotamab deruxtecan (DS- 1062), ESG-401, SKB-264, DAC-02 and BAT-8003. In some embodiments, the anti-Trop-2 ADC is sacituzumab govitecan. In some embodiments, the anti-Trop-2 ADC is datopotamab deruxtecan (DS- 1062). In some embodiments, the adenosine pathway inhibitor is a CD39 inhibitor, a CD73 inhibitor, or an adenosine receptor antagonist.
- the CD39 inhibitor is selected from TTX-030 (AbbVie/Trishula), IPH5201 (AstraZeneca/Innate Pharma), SRF617 (Surface Oncology), CD39 ASO (Secarna Pharmaceuticals), JS-019 (Shanghai Junshi Biosciences); AB598 (anti-CD39) (Arcus Biosciences), ES002 (Elpiscience Biopharmaceuticals), and CD39xPDl (Biotheus).
- the CD73 inhibitor is selected from oleclumab (AstraZeneca), BMS-986179 (BMS), uliledlimab (I-MAB Biopharma), AK119 (Akeso Biopharma), quemliclustat (AB680, Arcus Biosciences), mupadolimab (Corvus Pharmaceuticals), HLX23 (Shanghai Hcnlius Biotech), INCA00186 (Incytc), IBI325 (Innovcnt Bio), NZV930 (Novartis/Surface Oncology), ORIC-533 (ORIC Pharma), Sym024 (Servier), IPH5301 (Innate), IOA-237 (iOnctura), JAB-BX100 (Jacobio), PT199 (Phanes Therapeutics), TRB010 (Trican Biotechnology), CD73 ASO (Secarna Pharmaceuticals), 622 (3SBio), ABSK-051 (Abbisko Therapeutics), AK131 (CD73xPD
- the CD73 inhibitor is oleclumab or quemliclustat. In some embodiments, the CD73 inhibitor is quemliclustat.
- the adenosine receptor antagonist is an adenosine A2A receptor (A2AR; ADORA2A) selective antagonist, such as imaradenant (AstraZeneca), NIR178 (Novartis/Palobiofarma) ID11902 (Ildong), IN-A003 (Inno.n), NTI-55 (A2aR/TLR7, Nammi), TT-10 (Tarus Therapeutics), or TT- 228 (Teon Therapeutics).
- A2AR adenosine A2A receptor
- ADORA2A adenosine A2A receptor selective antagonist
- the adenosine receptor antagonist is an adenosine A2B receptor (A2BR; ADORA2B) antagonist, such as PBF-1129 (Palobiofarma) or TT-702 (Teon Therapeutics).
- A2BR adenosine A2B receptor
- ADORA2B adenosine A2B receptor
- PBF-1129 Palobiofarma
- TT-702 Teon Therapeutics
- the adenosine receptor antagonist is a dual adenosine A2A/A2B receptor antagonist, such as etrumadenant (AB928, Arcus Biosciences), INCB 106385 (Incyte), M1069 (Merck KgaA), A2aR/A2bR (Domain/Merck KgaA), HM87277 (Al/A2aR/A2bR, Hanmi Pharmaceutical), RVU-330 (Ryvu), TT-53 (Tarus Therapeutics).
- the adenosine receptor antagonist is etrumadenant.
- the adenosine pathway inhibitor is quemliclustat or etrumadenant.
- the adenosine pathway inhibitor is etrumadenant.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dxd); and b) an adenosine pathway inhibitor selected from a CD39 inhibitor, a CD73 inhibitor, and an adenosine receptor antagonist.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dxd); and b) an adenosine pathway inhibitor selected from a CD73 inhibitor and an adenosine receptor antagonist.
- a subject e.g., a human cancer patient
- an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dxd)
- an adenosine pathway inhibitor selected from a CD73 inhibitor and an adenosine receptor antagonist.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dxd); and b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, ulilcdlimab, imaradenant, NIR178, and ctrumadcnant.
- a subject e.g., a human cancer patient
- an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dxd)
- an adenosine pathway inhibitor selected from oleclumab, BMS-986179, ulilcdlimab, imaradenant, NIR178, and ctrumadcnant.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC having a structure mAb-CL2A-SN-38, represented by: and b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, uliledlimab, imaradenant, NIR178, and etrumadenant.
- a subject e.g., a human cancer patient
- an anti-Trop-2 ADC having a structure mAb-CL2A-SN-38, represented by: and b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, uliledlimab, imaradenant, NIR178, and etrumadenant.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan or datopotamab deruxtecan (DS-1062); and b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, uliledlimab, imaradenant, NIR178, and etrumadenant.
- a subject e.g., a human cancer patient
- a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan or datopotamab deruxtecan (DS-1062); and b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, uliledlimab, imaradenant, NIR178, and etrumadenant.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan; and b) etrumadenant.
- the methods provided herein further comprise co-administering an additional therapeutic agent or therapeutic modality, or a combination thereof.
- the additional therapeutic agent comprises an immune checkpoint inhibitor (CPI).
- the CPI comprises an anti-PD(L)l antibody (e.g., an anti-PD-1 antibody or an anti- PD-L1 antibody), and optionally an anti-TIGIT antibody.
- the anti-PD(L)l antibody is selected from pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD(L)l antibody is zimberelimab.
- the anti-TIGIT antibody is selected from tiragolumab, vibostolimab, domvanalimab, AB308, AK127, BMS- 986207, ralzapastotug, and etigilimab.
- the anti-TIGIT antibody is domvanalimab.
- the additional therapeutic agent comprises an anti-PD(L)! antibody and an anti-TIGIT antibody.
- the additional therapeutic agent comprises a) zimberelimab and domvanalimab, b) zimberelimab and AB3O8, c) atezolizumab and tiragolumab, d) pembrolizumab and vibostolimab, e) MK-7684A (pembrolizumab/vibostolimab coformulation), f) durvalumab and domvanalimab, g) zimberelimab and ralzapastotug, or h) pembrolizumab and ralzapastotug.
- the anti-PD(L)l antibody is zimberelimab and the anti-TIGIT antibody is domvanalimab.
- the additional therapeutic modality comprises surgery or radiation therapy.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dxd); b) an adenosine pathway inhibitor selected from oleclumab, BMS- 986179, uliledlimab, imaradenant, NIR178, and etrumadenant, and c) an anti-PD(L)l antibody.
- a subject e.g., a human cancer patient
- a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC comprising a topoisomerase I inhibitor (e.g., SN38 or Dx
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) an anti-Trop-2 ADC having a structure mAb-CL2A-SN-38, represented by: b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, uliledlimab, imaradenant, NIR178, and etrumadenant, and c) an anti-PD(L)l antibody.
- the methods provided herein comprise co-administering to a subject (e.g.
- a human cancer patient having a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan or datopotamab deruxtecan (DS- 1062); b) an adenosine pathway inhibitor selected from oleclumab, BMS-986179, uliledlimab, imaradenant, NIR178, and etrumadenant; and c) and anti-PD(L)l antibody.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan; b) etrumadenant; and c) an anti-PD(L)l antibody.
- a subject e.g., a human cancer patient
- a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan; b) etrumadenant; and c) an anti-PD(L)l antibody.
- the anti-PD(L)l antibody is selected from pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cctrclimab, gcnolimzumab, prolgolimab, lodapolimab, camrclizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD(L)l antibody is zimberelimab.
- the methods provided herein comprise co-administering to a subject (e.g., a human cancer patient) having a Trop-2 positive cancer an effective amount of a) sacituzumab govitecan; b) etrumadenant; and c) zimberelimab.
- the Trop-2 positive cancer is castrate resistant prostate cancer (CRPC) or non-small cell lung cancer (NSCLC).
- the Trop-2 positive cancer is CRPC.
- the Trop-2 positive cancer is metastatic (e.g., mCRPC, mNSCLC).
- the Trop-2 positive cancer is resistant or refractive of one or more anti-cancer therapy (e.g., NHA resistant or refractive mCRPC).
- the methods provided herein comprise co-administering to a subject e.g., a human cancer patient) having a tumor antigen positive (TA + ) cancer an effective amount of a) a Topi ADC; and b) an adenosine pathway inhibitor.
- the Topi ADC comprises a camptothecin analog.
- the camptothecin analog is an irinotecan derivative, a topotecan derivative, or an exatecan derivative.
- the camptothecin analog is SN38 or Dxd.
- the camptothecin analog is SN38.
- the camptothecin analog is connected to a tumor antigen targeted antibody via a hydrolyzable linker.
- the hydrolyzable linker is CL2A (e.g., as described in US 7,999,083).
- the camptothecin analog is connected to a tumor antigen targeted antibody via a protease cleavable linker.
- the Topi ADC has a structure mAb-CL2A-SN-38, represented by:
- the tumor antigen targeted antibody in the Topi ADC is selected from gemtuzumab, brentuximab, belantamab, camidanlumah, trastuzumab, inotuzumab, glembatumumab, anetumab, mirvetuximab, dcpatuxizumab, vadastuximab, labctuzumab, ladiratuzumab, loncastuximab, patritumab, lifastuzumab, indusatumab, polatuzumab, pinatuzumab, coltuximab, upifitamab, indatuximab, milatuzumab, rovalpituzumab, enfortumab, tisotumab, tusamitamab, disitamab
- the tumor antigen targeted antibody in the Topi ADC is selected from hLLl, hLL2, RFB4, hA19, hA20, hRS7, hPAM4, hMN-3, hMN-14, hMu-9, hRl, CC49, hL243, D2/B, hlmmu-31, and antigen binding fragments thereof.
- the anti-Trop-2 ADC comprises the anti-Trop-2 antibody sacituzumab (hRS7, described, e.g., in W02003074566, Figures 3 and 4).
- the Topi ADC is trastuzumab deruxtecan.
- the adenosine pathway inhibitor is a CD39 inhibitor, a CD73 inhibitor, or an adenosine receptor antagonist.
- the CD39 inhibitor is selected from TTX-030 (AbbVie/Trishula), IPH5201 (AstraZeneca/Innate Pharma), SRF617 (Surface Oncology), CD39 ASO (Secama Pharmaceuticals), JS-O19 (Shanghai Junshi Biosciences); anti-CD39 (Arcus Biosciences), ES002 (Elpiscience Biopharmaceuticals), and CD39xPDl (Biotheus).
- the CD73 inhibitor is selected from oleclumab (AstraZeneca), BMS-986179 (BMS), uliledlimab (I-MAB Biopharma), AK119 (Akeso Biopharma), quemliclustat (AB68O, Arcus Biosciences), mupadolimab (Corvus Pharmaceuticals), HLX23 (Shanghai Henlius Biotech), TNCAOO186 (Tncyte), IBT325 (Tnnovent Bio), NZV930 (Novartis/Surface Oncology), ORIC-533 (ORIC Pharma), Sym024 (Servier), IPH5301 (Innate), IOA-237 (iOnctura), JAB- BX100 (Jacobio), PT199 (Phanes Therapeutics), TRB010 (Trican Biotechnology), CD73 ASO (Secarna Pharmaceuticals), 622 (3SBio), ABSK-051 (Abbisko Therapeutics), AK131 (CD73xPDl
- the CD73 inhibitor is oleclumab or quemliclustat. In some embodiments, the CD73 inhibitor is quemliclustat.
- the adenosine receptor antagonist is an adenosine A2A receptor (A2AR; AD0RA2A) selective antagonist, such as imaradenant (AstraZeneca), NIR178 (Novartis/Palobiofarma) ID11902 (Ildong), IN-A003 (Inno.n), NTI-55 (A2aR/TLR7, Nammi), TT-10 (Tarns Therapeutics), or TT-228 (Teon Therapeutics).
- A2AR adenosine A2A receptor
- AD0RA2A adenosine A2A receptor selective antagonist
- the adenosine receptor antagonist is an adenosine A2B receptor (A2BR; AD0RA2B) antagonist, such as PBF-1129 (Palobiofarma) or TT-702 (Teon Therapeutics).
- A2BR adenosine A2B receptor
- AD0RA2B adenosine A2B receptor
- PBF-1129 Palobiofarma
- TT-702 Teon Therapeutics
- the adenosine receptor antagonist is a dual adenosine A2A/A2B receptor antagonist, such as etrumadenant (AB928, Arcus Biosciences), INCB 106385 (Incyte), M1069 (Merck KGaA), A2aR/A2bR (Domain/Merck KGaA), HM87277 (Al/A2aR/A2bR, Hanmi Pharmaceutical), RVU-330 (Ryvu), TT-53 (Tarns Therapeutics).
- the methods provided herein further comprise co-administering an additional therapeutic agent or therapeutic modality, or a combination thereof.
- the additional therapeutic agent comprises an immune checkpoint inhibitor.
- the CPI comprises an anti-PD(L)l antibody (e.g., an anti-PD-1 antibody or an anti-PD-Ll antibody), and optionally an anti-TIGIT antibody.
- the anti-PD(L)l antibody is selected from pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD(L)l antibody is zimberelimab.
- the anti-TIGIT antibody is selected from tiragolumab, vibostolimab, domvanalimab, AB3O8, AK127, BMS-986207, ralzapastotug, and etigilimab.
- the anti-TIGIT antibody is domvanalimab.
- the additional therapeutic agent comprises an anti-PD(L)l antibody and an anti- TIGIT antibody.
- the additional therapeutic agent comprises a) zimberelimab and domvanalimab, b) zimberelimab and AB308, c) atezolizumab and tiragolumab, d) pembrolizumab and vibostolimab, e) MK-7684A (pembrolizumab/vibostolimab coformulation), f) durvalumab and domvanalimab, g) zimberelimab and ralzapastotug, or h) pembrolizumab and ralzapastotug.
- the anti-PD(L)l antibody is zimberelimab and the anti-TIGIT antibody is domvanalimab.
- the additional therapeutic modality comprises surgery or radiation therapy.
- the tumor antigen positive cancer is positive for a tumor antigen selected from carbonic anhydrase IX, B7, CCCL19, CCCL21, CSAp, HER-2/neu, BrE3, CD1, CDla, CD2, CD3, CD4, CD5, CD8, CD11A, CD14, CD15, CD16, CD18, CD19, CD20, CD21, CD22, CD23, CD25, CD29, CD30, CD32b, CD33, CD37, CD38, CD40, CD40L, CD44, CD45, CD46, CD52, CD54, CD55, CD59, CD64, CD67, CD70, CD74, CD79a, CD80, CD83, CD95, CD126, CD133, CD138, CD147, CD154, CEACAM5, CEACAM5, CEACAM5, CEACAM5, CE
- General dosing and administration regimens and pharmaceutical compositions for antibody-drug conjugates, adenosine pathway inhibitors, and additional therapeutic agents that can be used in the methods provide herein are known to a skilled artisan.
- general dosing and administration regimens and pharmaceutical compositions are described in W02014/092804A1 for exemplary ADCs, including sacituzumab govitecan, in WO2017120508 for exemplary CD73 inhibitors, including quemliclustat, in W02018136700A1 for exemplary adenosine receptor antagonists, including etrumadenant, and in W02017025051A1 for exemplary anti-PD(L)l antibodies, including zimberelimab.
- the ADC e.g., sacituzumab govitecan
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- the anti-PD(L)l antibody e.g., zimberelimab
- the ADC e.g., sacituzumab govitecan
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- the anti-PD(Ll) antibody e.g., zimberelimab
- the same day e.g., on day 1 of a 21-day treatment cycle.
- the ADC e.g., sacituzumab govitecan
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- the anti-PD(Ll) antibody e.g., zimberelimab
- the ADC e.g., sacituzumab govitecan
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- the anti-PD(Ll) antibody e.g., zimberelimab
- the ADC e.g., sacituzumab govitecan
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- the anti-PD(Ll) antibody e.g., zimberelimab
- sacituzumab govitecan and etrumadenant are co-administered sequentially on day 1 of a 21 -day treatment cycle, wherein etrumadenant is co-administcrcd orally at least 30 minutes prior to the start of an intravenous infusion of sacituzumab govitecan.
- sacituzumab govitecan, etrumadenant, and zimberelimab are co-administered sequentially on day 1 of a 21 -day treatment cycle, wherein etrumadenant is co-administered orally at least 30 minutes prior to the start of an intravenous infusion of zimberelimab, and sacituzumab govitecan is coadministered intravenously after completion of the zimberelimab infusion.
- sacituzumab govitecan is coadministered on day 1 and day 8 of a 21 -day treatment cycle.
- sacituzumab govitecan is co-administered intravenously (IV).
- IV intravenously
- sacituzumab govitecan is co-administered at a dose of 8 mg/kg to 10 mg/kg.
- sacituzumab govitecan is co-administered at a dose of 8 mg/kg or 10 mg/kg.
- sacituzumab govitecan is co-administered at a dose of 10 mg/kg.
- sacituzumab govitecan is coadministered intravenously (IV) at a dose of 8 mg/kg or 10 mg/kg on day 1 and day 8 of a 21-day treatment cycle.
- etrumadenant is co-administered once daily (QD). In some embodiments, etrumadenant is co-administered orally (PO). In some embodiments, etrumadenant is co-administered at a dose of 75 mg or 150 mg. In some embodiments, etrumadenant is co-administered at a dose of 150 mg. Tn some embodiments, etrumadenant is co-administered orally (PO) once daily (QD) at a dose of 75 mg or 150 mg. In some embodiments, etrumadenant is co-administered orally (PO) once daily (QD) at a dose of 150 mg.
- an anti-PD(L)l antibody e.g., zimberelimab
- Q3W once every three weeks
- an anti-PD(L)1 antibody is co-administered intravenously (IV).
- an anti-PD(L)l antibody is co-administered at a dose of 360 mg.
- sacituzumab govitecan is coadministered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 8 mg/kg or 10 mg/kg; etrumadenant is co-administered orally (PO) once daily (QD) at a dose of 75 mg or 150 mg on each day of the 21 -day treatment cycle, and, optionally, zimberelimab is coadministered on day 1 of the 21-day treatment cycle (Q3W) at a dose of 360 mg.
- IV intravenously
- etrumadenant is co-administered orally (PO) once daily (QD) at a dose of 75 mg or 150 mg on each day of the 21 -day treatment cycle
- Zmberelimab is coadministered on day 1 of the 21-day treatment cycle (Q3W) at a dose of 360 mg.
- sacituzumab govitecan is coadministered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 10 mg/kg; etrumadenant is co-administered orally (PO) once daily (QD) at a dose of 150 mg on each day of the 21-day treatment cycle, and, optionally, zimberelimab is co-administered on day 1 of the 21-day treatment cycle (Q3W) at a dose of 360 mg.
- IV intravenously
- etrumadenant is co-administered orally (PO) once daily (QD) at a dose of 150 mg on each day of the 21-day treatment cycle
- Zmberelimab is co-administered on day 1 of the 21-day treatment cycle (Q3W) at a dose of 360 mg.
- the methods provided herein have anti-cancer effects as determined by one or more efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), PSA response rate, radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- ORR objective response rate
- DCR disease control rate
- PFS progression free survival
- DOR duration of response
- OS overall survival
- CR complete response
- PR partial response
- PSA response rate radiographic response rate
- tumor response or progression is determined according to RECIST version 1.1.
- the ORR is defined as the composite proportion of participants with a PSA and/or radiographic complete and partial response determined by the investigator according to the Prostate Cancer Working Group 3 (PCWG3) criteria.
- PCWG3 Prostate Cancer Working Group 3
- the PSA response is defined as the proportion of participants with a confirmed PSA decrease from baseline of 50% or more based on 2 consecutive assessments measured at least 3 to 4 weeks apart.
- the radiographic response defined as the proportion of participants with a best overall response of CR or PR according to RECIST v 1.1.
- DCR is defined as the proportion of participants with a best overall RECIST response of CR, PR, or stable disease (SD).
- SD stable disease
- the DCR is at least 6 months.
- PFS is defined as the time from treatment assignment until first documentation of progressive disease (PSA progression, radiographic progression, bone scan progression, or other) or death, whichever occurs first. Depending on data availability, time to progression due to specific reasons (e.g., PSA progression or radiographic progression) may also be presented individually.
- OS is defined as the time from treatment assignment until death due to any cause.
- the methods provided herein further comprise determining tumor antigen (e.g., Trop-2) expression levels in a sample from the subject.
- the determination of tumor antigen (e.g., Trop-2) expression levels can occur with any clinical analytics method known to a skilled artisan.
- Samples can include liquid biopsy samples (e.g., blood samples) and solid tumor biopsy samples.
- Tumor antigen (e.g., Trop-2) expression levels can be determined at the DNA, RNA, or protein level.
- Illustrative methods for the determination of tumor antigen (e.g., Trop-2) expression levels include western blot, immunohistochemistry, QPCR, exome sequencing, FACS, and the like.
- an anti-CD47 antibody is not coadministered to the subject or human patient (CD47; integrin associated protein; IAP; NCBI Gene ID: 961).
- the subject or human patient is not co-administered an anti-CD47 antibody selected from magrolimab, lemzoparlimab, letaplimab, ligufalimab, AO- 176, IBI-322, ZL-1201, IMC-002, SRF-231, CC-90002 (a.k.a., INBRX-103), NI-1701 (a.k.a., TG-1801) and STI-6643.
- the subject or human patient is not co-administered magrolimab.
- an MCL1 inhibitor is not coadministered to the subject or human patient (MCL1; myeloid leukemia cell differentiation protein; NCBI Gene ID: 4170).
- MCL1 myeloid leukemia cell differentiation protein
- NCBI Gene ID: 4170 the subject or human patient is not coadministered an MCL1 inhibitor selected from GS-9716, AMG-397, AMG-176, PRT-1419, and S6431.
- the subject or human patient is not co-administered GS-9716.
- a FLT3 agonist is not coadministered to the subject or human patient (FLT3; fms like tyrosine kinase; CD135; NCBI Gene ID: 2322).
- the FLT3 agonist is selected from GS-3583, CDX-301, TAK- 605, ONCR-177, Alb-Ftl3L, and SYM-027.
- the FLT3 agonist is GS-3583.
- kits for use as a medicament comprising a) TROP-2- targeted antibody-drug conjugate (ADC) comprising an anti-TROP-2 antibody (anti-TROP-2 ADC); b) an adenosine pathway inhibitor; and c) optionally an anti-PD-(L)l antibody.
- ADC TROP-2- targeted antibody-drug conjugate
- kits for use as a medicament comprising a) a tumor antigen (TA) targeted ADC comprising a topoisomerase I inhibitor (Topi ADC); b) an adenosine pathway inhibitor; and c) optionally an anti-PD-(L)! antibody.
- TA tumor antigen
- Topici ADC topoisomerase I inhibitor
- adenosine pathway inhibitor optionally an anti-PD-(L)! antibody.
- kits comprising one or more unitary doses of the active agents, e.g., a) an anti-Trop-2 ADC (c.g., sacituzumab govitccan) or Topi ADC; b) an adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat); and optionally c) an anti-PD(L)l antibody (e.g., zimberelimab), and formulations thereof, as described herein, and instructions for use.
- an anti-Trop-2 ADC c.g., sacituzumab govitccan
- Topi ADC adenosine pathway inhibitor
- an anti-PD(L)l antibody e.g., zimberelimab
- the a) anti-Trop-2 ADC e.g., sacituzumab govitecan
- Topi ADC e.g., adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat); and optionally c) anti-PD(L)l antibody (e.g., zimberelimab)
- the kit can further contain a least one additional reagent, e.g. an anti-TIGIT antibody.
- Kits typically include a label indicating the intended use of the contents of the kit. The term label includes any writing, or recorded material supplied on or with the kit, or which otherwise accompanies the kit.
- one or more of the a) anti-Trop-2 ADC e.g., sacituzumab govitecan) or Topi ADC
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- c) anti-PD(L)l antibody e.g., zimberelimab
- a dosage form e.g., a therapeutically effective dosage form
- one or more of the a) anti-Trop-2 ADC e.g., sacituzumab govitecan) or Topi ADC
- b) adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- optionally c) anti-PD(L)l antibody e.g., zimberelimab
- two or more different dosage forms e.g., two or more different therapeutically effective dosage forms.
- one or more of the a) anti-Trop-2 ADC e.g., sacituzumab govitecan) or Topi ADC
- b) adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- c) anti-PD(L)l antibody e.g., zimberelimab
- any convenient packaging e.g., stick pack, dose pack, etc.
- anti-Trop-2 ADC e.g., sacituzumab govitecan
- Topi ADC e.g., adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat); and optionally c) anti-PD(L)l antibody (e.g., zimberelimab)
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- anti-PD(L)l antibody e.g., zimberelimab
- the a) anti-Trop-2 ADC e.g., sacituzumab govitecan
- Topi ADC e.g., adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat); and optionally c) anti-PD(L)l antibody (e.g., zimberelimab) are provided in separate containers.
- adenosine pathway inhibitor e.g., etrumadenant, quemliclustat
- anti-PD(L)l antibody e.g., zimberelimab
- compositions comprising one or more of the a) anti-Trop-2 ADC (e.g., sacituzumab govitecan) or Topi ADC; b) adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat); and optionally c) anti-PD(L)l antibody (e.g., zimberelimab) are provided in one or more containers, the containers having a label.
- Suitable containers include, for example, bottles, vials, ampoules, syringes (including pre-loaded syringes), and test tubes.
- the containers may be formed from a variety of materials such as glass or plastic.
- the container holds a composition which is effective for treating the condition and may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle).
- the active agent in one composition is a) an anti-Trop-2 ADC (e.g., sacituzumab govitecan) or Topi ADC.
- the active agent in a second composition is an adenosine pathway inhibitor (e.g., etrumadenant, quemliclustat).
- the active agent in an optional third composition is optionally c) anti-PD(L)l antibody (e.g., zimberelimab).
- the label on, or associated with, the container indicates that the composition is used for treating the condition of choice.
- the article of manufacture may further comprise one or more containers comprising a pharmaceutically- acceptable buffer, e.g., for use as diluent.
- a pharmaceutically- acceptable buffer include without limitation phosphate -buffered saline, Ringer’s solution and/or dextrose solution.
- the kit may further include other materials desirable from a commercial and user standpoint, including other buffers, diluents, filters, needles, syringes, and package inserts with instructions for use.
- the subject kits may further include (in certain embodiments) instructions for practicing the subject methods.
- These instructions may be present in the subject kits in a variety of forms, one or more of which may be present in the kit.
- One form in which these instructions may be present is as printed information on a suitable medium or substrate, e.g., a piece or pieces of paper on which the information is printed, in the packaging of the kit, in a package insert, and the like.
- Yet another form of these instructions is a computer readable medium, e.g., diskette, compact disk (CD), flash drive, and the like, on which the information has been recorded.
- Yet another form of these instructions that may be present is a website address which may be used via the internet to access the information at a removed site.
- mCRPC metastatic castrateresistant prostate cancer
- the method further comprises co-administering zimberelimab to the human patient.
- the human mCRPC patient has previously progressed on androgen deprivation therapy (ADT).
- ADT androgen deprivation therapy
- the human mCRPC patient has previously progressed on one or more next generation hormonal agents (NHAs, e.g., abiraterone, enzalutamide, darolutamide, apalutamide).
- NHAs next generation hormonal agents
- the human mCRPC patient is checkpoint inhibitor (CPI) and taxanc naive.
- the human mCRPC patient has RECIST 1.1 measurable or non-measurable disease.
- a method of treating castrate resistant prostate cancer comprising co-administering to a human patient an effective amount of a) an anti- Trop-2 ADC; b) a CD73 inhibitor or adenosine receptor antagonist; and, optionally, c) an anti- PD(L)1 antibody.
- the CRPC is metastatic CRPC (mCRPC).
- the anti-Trop-2 ADC has a structural formula of mAb-CL2A-SN-38, with a structure represented by:
- the anti-Trop-2 ADC comprises a linker-payload conjugate having a structure represented by: attached to an anti-Trop-2 antibody (e.g., hRS7).
- the anti-Trop-2 ADC comprises a linker-payload conjugate (TL035) having a structure represented by:
- the anti-Trop-2 ADC comprises a linker-payload conjugate having a structure represented by: attached to an anti-Trop-2 antibody (e.g., hTINAl-HILl).
- the anti-Trop-2 ADC has a DAR of about 4.
- the anti-Trop-2 ADC is selected from sacituzumab govitecan, datopotamab deruxtecan (DS-1062), ESG-401, SKB-264, DAC-02 and BAT-8003.
- the anti-Trop-2 antibody is sacituzumab govitecan or datopotamab deruxtecan.
- the anti-Trop-2 antibody is sacituzumab govitecan.
- the CD73 inhibitor is selected from oleclumab (AstraZeneca), BMS-986179 (BMS), uliledlimab (I-MAB Biopharma), AK119 (Akeso Biopharma), quemliclustat (AB680, Arcus Biosciences), mupadolimab (Corvus Pharmaceuticals), HLX23 (Shanghai Henlius Biotech), INCA00186 (Incyte), IBI325 (Innovent Bio), NZV930 (Novartis/Surface Oncology), ORIC-533 (ORIC Pharma), Sym024 (Servier), IPH5301 (Innate), IOA-237 (iOnctura), JAB-BX100 (Jacobio), PT199 (Phanes Therapeutics), TRB010 (Trican Biotechnology), CD73 ASO (Secarna Pharmaceuticals), 622 (3Sbio),
- the CD73 inhibitor is selected from oleclumab and quemliclustat. In some embodiments, the CD73 inhibitor is quemliclustat.
- the adenosine receptor antagonist is an adenosine A2A receptor (A2AR; AD0RA2A) selective antagonist, such as imaradenant (AstraZeneca), NIR178 (Novartis/Palobiofarma) ID11902 (Ildong), IN-A003 (Inno.n), NTI-55 (A2aR/TLR7, Nammi), TT-10 (Tarus Therapeutics), or TT-228 (Teon Therapeutics).
- A2AR adenosine A2A receptor
- AD0RA2A adenosine A2A receptor selective antagonist
- the adenosine receptor antagonist is an adenosine A2B receptor (A2BR; AD0RA2B) antagonist, such as PBF- 1129 (Palobiofarma) or TT-702 (Teon Therapeutics).
- A2BR adenosine A2B receptor
- AD0RA2B adenosine A2B receptor
- PBF- 1129 Palobiofarma
- TT-702 Teon Therapeutics
- the adenosine receptor antagonist is a dual adenosine A2A/A2B receptor antagonist, such as etrumadenant (AB928, Arcus Biosciences), INCB 106385 (Incyte), M1069 (Merck KgaA), A2aR/A2bR (Domain/Merck KgaA), HM87277 (Al/A2aR/A2bR, Hanmi Pharmaceutical), RVU-330 (Ryvu), and TT-53 (Tarus Therapeutics).
- the adenosine receptor antagonist is etrumadenant.
- the anti-PD(L)1 antibody is selected from pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD(L)l antibody is zimberelimab.
- the human patient is not co-administered an additional therapeutic agent selected from an MCL-1 inhibitor, an anti-CD47 antibody, and a FLT3 agonist.
- a method of treating castrate resistant prostate cancer comprising co-administering to a human patient an effective amount of a) sacituzumab govitecan; and b) etrumadenant.
- the CRPC is metastatic CRPC (mCRPC).
- the CRPC or mCRPC ((m)CRPC) is resistant or refractive to at least one anti-cancer therapy.
- the human patient has shown disease progression after prior treatment with a new hormonal agent (NHA; first- or second- generation non-steroidal antiandrogens, abiraterone, enzalutamide, darolutamide, apalutamide).
- the human patient has not received a prior taxane therapy (e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel), checkpoint inhibitor therapy (e.g., anti- CTLA4 antibody, anti-PD(L)l antibody), topoisomerase I inhibitor therapy (e.g., irinotecan).
- a prior taxane therapy e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel
- checkpoint inhibitor therapy e.g., anti- CTLA4 antibody, anti-PD(L)l antibody
- topoisomerase I inhibitor therapy e.g., irinotecan
- the human patient with (m)CRPC has histologically confirmed adenocarcinoma of the prostate and metastatic castrate resistant with tumor progression while on androgen deprivation therapy (ADT ; including orchiectomy) with castrate levels of scrum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by PSA and/or radiographic criteria according to PCWG3.
- ADT on androgen deprivation therapy
- the human patient (m)CRPC has metastatic castrate resistant adenocarcinoma of the prostate with tumor progression while on androgen deprivation therapy (e.g., including orchiectomy) with castrate levels of serum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by prostate specific antigen (PSA) and/or radiographic criteria according to The Prostate Cancer Working Group 3 (PCWG3) and measurable or non-measurable disease per the Response Evaluation Criteria in Solid Tumors (RECIST) vl.l.
- the human patient with (m)CRPC has Eastern Cooperative Oncology Group performance status 0 or 1 with a life expectancy >3 months.
- the human patient with (m)CRPC has been tested for tumor antigen (e.g., Trop-2) expression levels (e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or next-generation DNA sequencing).
- tumor antigen e.g., Trop-2
- sacituzumab govitecan is administered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 8 mg/kg or 10 mg/kg and etrumadenant is administered orally (PO) once daily (QD) at a dose of 75 mg or 150 mg on each day of the 21-day treatment cycle.
- sacituzumab govitecan is administered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 10 mg/kg and etrumadenant is administered orally (PO) once daily (QD) at a dose of 150 mg on each day of the 21-day treatment cycle.
- the methods provided herein have anti-cancer effects as determined by one or more efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), overall survival (OS), complete response (CR), partial response (PR), PSA response rate, radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- the ORR is defined as the composite proportion of participants with a PSA and/or radiographic complete and partial response determined by the investigator according to the Prostate Cancer Working Group 3 (PCWG3) criteria.
- the PSA response is defined as the proportion of participants with a confirmed PSA decrease from baseline of 50% or more based on 2 consecutive assessments measured at least 3 to 4 weeks apart.
- the radiographic response defined as the proportion of participants with a best overall response of CR or PR according to RECIST vl.l.
- DCR is defined as the proportion of participants with a best overall RECTST response of CR, PR, or stable disease (SD). Tn some embodiments, the DCR is at least 6 months.
- PFS is defined as the time from treatment assignment until first documentation of progressive disease (PSA progression, radiographic progression, bone scan progression, or other) or death, whichever occurs first. Depending on data availability, time to progression due to specific reasons (e.g., PSA progression or radiographic progression) may also be presented individually.
- OS is defined as the time from treatment assignment until death due to any cause.
- the human patient is not coadministered an additional therapeutic agent selected from an MCL-1 inhibitor, an anti-CD47 antibody, and a FLT3 agonist.
- a method of treating castrate resistant prostate cancer comprising co-administering to a human patient an effective amount of a) sacituzumab govitecan; b) etrumadenant and c) zimberelimab.
- the CRPC is metastatic CRPC (mCRPC).
- the CRPC or mCRPC ((m)CRPC) is resistant or refractive to at least one anti-cancer therapy.
- the human patient has shown disease progression after prior treatment with a new hormonal agent (NHA; first- or second-generation non-steroidal antiandrogens, e.g, abiraterone, enzalutamide, darolutamide, apalutamide).
- NHA new hormonal agent
- the human patient has not received a prior taxane therapy (e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel), checkpoint inhibitor therapy (e.g., anti-CTLA4 antibody, anti-PD(L)l antibody), topoisomerase I inhibitor therapy (e.g., irinotecan).
- a prior taxane therapy e.g., paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, cabazitaxel
- checkpoint inhibitor therapy e.g., anti-CTLA4 antibody, anti-PD(L)l antibody
- the human patient with (m)CRPC has histologically confirmed adenocarcinoma of the prostate and metastatic castrate resistant with tumor progression while on androgen deprivation therapy (ADT ; including orchiectomy) with castrate levels of serum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by PSA and/or radiographic criteria according to PCWG3.
- ADT on androgen deprivation therapy
- the human patient (m)CRPC has metastatic castrate resistant adenocarcinoma of the prostate with tumor progression while on androgen deprivation therapy (e.g., including orchiectomy) with castrate levels of serum (total) testosterone ( ⁇ 1.7 nmol/L or 50 ng/dL) defined by prostate specific antigen (PSA) and/or radiographic criteria according to The Prostate Cancer Working Group 3 (PCWG3) and measurable or non-measurable disease per the Response Evaluation Criteria in Solid Tumors (RECTST) vl .l . Tn some embodiments, the human patient with (m)CRPC has Eastern Cooperative Oncology Group performance status 0 or 1 with a life expectancy >3 months.
- the human patient with (m)CRPC has been tested for tumor antigen (e.g., Trop-2) expression levels (e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or nextgeneration DNA sequencing).
- tumor antigen e.g., Trop-2
- sacituzumab govitecan is administered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 8 mg/kg or 10 mg/kg
- etrumadenant is administered orally (PO) once daily (QD) at a dose of 75 mg or 150 mg on each day of the 21-day treatment cycle
- zimberelimab is administered on day 1 of the 21- day treatment cycle (Q3W) at a dose of 360 mg.
- sacituzumab govitecan is administered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 10 mg/kg
- etrumadenant is administered orally (PO) once daily (QD) at a dose of 150 mg on each day of the 21-day treatment cycle
- zimberelimab is administered on day 1 of the 21-day treatment cycle (Q3W) at a dose of 360 mg.
- the methods provided herein have anticancer effects as determined by one or more efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), overall survival (OS), complete response (CR), partial response (PR), PSA response rate, radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers, hr some embodiments, the ORR is defined as the composite proportion of participants with a PSA and/or radiographic complete and partial response determined by the investigator according to the Prostate Cancer Working Group 3 (PCWG3) criteria.
- PCWG3 Prostate Cancer Working Group 3
- the PSA response is defined as the proportion of participants with a confirmed PSA decrease from baseline of 50% or more based on 2 consecutive assessments measured at least 3 to 4 weeks apart.
- the radiographic response defined as the proportion of participants with a best overall response of CR or PR according to RECIST vl.l.
- DCR is defined as the proportion of participants with a best overall RECIST response of CR, PR, or stable disease (SD).
- the DCR is at least 6 months.
- PFS is defined as the time from treatment assignment until first documentation of progressive disease (PSA progression, radiographic progression, bone scan progression, or other) or death, whichever occurs first.
- time to progression due to specific reasons may also be presented individually.
- OS is defined as the time from treatment assignment until death due to any cause.
- the human patient is not co-administered an additional therapeutic agent selected from an MCL-1 inhibitor, an anti-CD47 antibody, and a FLT3 agonist.
- Tn some embodiments, provided herein is a method of treating metastatic non-small cell lung cancer (mNSCLC) comprising co-administcring to a human mNSCLC patient an effective amount of a) sacituzumab govitecan; b) etrumadenant; and c) an anti-PD-(L)l antibody.
- mNSCLC metastatic non-small cell lung cancer
- the anti-PD-(L)l antibody is selected from the group consisting of pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD-(L)l antibody is zimberelimab.
- the human mNSCLC patient has progressed after platinumbased chemotherapy.
- the human mNSCLC patient has progressed after checkpoint inhibitor therapy (e.g., anti-PD-(L)l antibody or anti-CTLA4 antibody therapy).
- the human mNSCLC patient has progressed after platinum-based chemotherapy and anti-PD-(L)l antibody therapy, received either in combination or sequentially (in any order).
- the human mNSCLC patient is treatment naive.
- NSCLC non-small cell lung cancer
- a method of treating non-small cell lung cancer comprising co-administering to a human patient an effective amount of a) an anti-Trop- 2 ADC; b) a CD73 inhibitor or adenosine receptor antagonist; and, optionally, c) an anti-PD(L)l antibody.
- the NSCLC is metastatic NSCLC (mNSCLC).
- the anti-Trop-2 ADC has a structural formula of mAb-CL2A-SN-38, with a structure represented by:
- the anti-Trop-2 ADC comprises a linker-payload conjugate having a structure represented by:
- the anti-Trop-2 ADC comprises a linker-payload conjugate (TL035) having a structure represented by:
- the anti-Trop-2 ADC comprises a linker-payload conjugate having a structure represented by: attached to an anti-Trop-2 antibody (e.g., hTINAl-HILl).
- the anti-Trop-2 ADC has a DAR of about 4.
- the anti-Trop-2 ADC is selected from sacituzumab govitecan, datopotamab deruxtecan (DS-1062), ESG-401, SKB-264, DAC-02 and BAT-8003.
- the anti-Trop-2 antibody is sacituzumab govitecan or datopotamab deruxtecan. In some embodiments, the anti-Trop-2 antibody is sacituzumab govitecan.
- the CD73 inhibitor is selected from oleclumab (AstraZeneca), B MS-986179 (BMS), ulilcdlimab (I-MAB Biopharma), AK119 (Akcso Biopharma), quemliclustat (AB680, Arcus Biosciences), mupadolimab (Corvus Pharmaceuticals), HLX23 (Shanghai Henlius Biotech), INCA00186 (Incyte), IBI325 (Innovent Bio), NZV930 (Novartis/Surface Oncology), ORIC-533 (ORIC Pharma), Sym024 (Servier), IPH5301 (Innate), IOA-237 (iOnctura), JAB-BX100 (Jacobio),
- the CD73 inhibitor is selected from oleclumab and quemliclustat. In some embodiments, the CD73 inhibitor is quemliclustat.
- the adenosine receptor antagonist is an adenosine A2A receptor (A2AR; ADORA2A) selective antagonist, such as imaradenant (AstraZeneca), NIR178 (Novartis/Palobiofarma) ID11902 (Ildong), IN-A003 (Inno.n), NTI-55 (A2aR/TLR7, Nammi), TT-10 (Tarus Therapeutics), or TT-228 (Teon Therapeutics).
- A2AR adenosine A2A receptor
- ADORA2A adenosine A2A receptor selective antagonist
- the adenosine receptor antagonist is an adenosine A2B receptor (A2BR; ADORA2B) antagonist, such as PBF- 1129 (Palobiofarma) or TT-702 (Teon Therapeutics).
- A2BR adenosine A2B receptor
- ADORA2B adenosine A2B receptor
- PBF- 1129 Palobiofarma
- TT-702 Teon Therapeutics
- the adenosine receptor antagonist is a dual adenosine A2A/A2B receptor antagonist, such as etrumadenant (AB928, Arcus Biosciences), INCB 106385 (Incyte), M1069 (Merck KGaA), A2aR/A2bR (Domain/Merck KGaA), HM87277 (Al/A2aR/A2bR, Hanmi Pharmaceutical), RVU-330 (Ryvu), and TT-53 (Tarus Therapeutics).
- the adenosine receptor antagonist is etrumadenant.
- the anti-PD-(L)l antibody is selected from pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD-(L)l antibody is zimberelimab.
- the human patient is not co-administered an additional therapeutic agent selected from an MCL-1 inhibitor, an anti-CD47 antibody, and a FLT3 agonist.
- NSCLC non-small cell lung cancer
- the anti-PD- (L)1 antibody is selected from the group consisting of pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atczolizumab, avclumab, durvalumab, cosibclimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the anti-PD-(L)l antibody is zimberelimab.
- the NSCLC is metastatic NSCLC (mNSCLC).
- the NSCLC or mNSCLC ((m)NSCLC) is resistant or refractive to at least one anti-cancer therapy.
- the human mNSCLC patient has progressed after platinum-based chemotherapy.
- the human mNSCLC patient has progressed after checkpoint inhibitor therapy (e.g., anti-PD-(L)l antibody or anti-CTLA4 antibody therapy).
- the human mNSCLC patient has progressed after platinum-based chemotherapy and anti-PD-(L)l antibody therapy, received either in combination or sequentially (in any order).
- the human mNSCLC patient is treatment naive.
- the human patient with (m)NSCLC has been tested for tumor antigen (e.g., Trop-2) expression levels (e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or next-generation DNA sequencing).
- tumor antigen e.g., Trop-2
- tumor antigen expression levels e.g., liquid or solid tumor biopsy, followed by tumor antigen expression analysis, e.g., by IHC or next-generation DNA sequencing.
- sacituzumab govitecan is administered intravenously (IV) on day 1 and day 8 of a 21 -day treatment cycle at a dose of 8 mg/kg or 10 mg/kg and etrumadenant is administered orally (PO) once daily (QD) at a dose of 75 mg or 150 mg on each day of the 21-day treatment cycle.
- sacituzumab govitecan is administered intravenously (IV) on day 1 and day 8 of a 21-day treatment cycle at a dose of 10 mg/kg and etrumadenant is administered orally (PO) once daily (QD) at a dose of 150 mg on each day of the 21-day treatment cycle.
- the methods provided herein have anti-cancer effects as determined by one or more efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- tumor response or progression is determined according to RECIST version 1.1.
- the human patient is not co-administered an additional therapeutic agent selected from an MCL-1 inhibitor, an anti-CD47 antibody, and a FLT3 agonist.
- [0215] Disclosed herein are methods of treating, mitigating, reducing, preventing or delaying the recurrence or metastasis of breast cancer comprising co-administering to a human patient an effective amount of: (a) sacituzumab govitecan; and (b) a CD73 inhibitor.
- the CD73 inhibitor is olcclumab, BMS-986179, ulilcdlimab, AK119, qucmliclustat, mupadolimab, HLX23, INCA00186, IB 1325, NZV930, ORIC-533, Sym024, IPH5301, IOA-237, JAB-BX100, PT199, TRB010, CD73 ASO, ABSK-051, AK131, BR101, BP1200, CB708, GB7002, or ATG-037.
- the CD73 inhibitor is quemliclustat (AB680, GS- 0680), uliledlimab, mupadolimab, ORIC-533, ATG-037, PT-199, AK131, NZV930, BMS- 986179, or oleclumab. In some embodiments, the CD73 inhibitor is quemliclustat (AB680, GS- 0680). In some embodiments, the method further comprises co-administering an anti-PD-(L)l antibody to the human patient.
- the anti-PD-(L)l antibody is selected from the group consisting of pembrolizumab, nivolumab, cemiplimab, pidilizumab, spartalizumab, atezolizumab, avelumab, durvalumab, cosibelimab, sasanlimab, tislelizumab, retifanlimab, balstilimab, toripalimab, cetrelimab, genolimzumab, prolgolimab, lodapolimab, camrelizumab, budigalimab, avelumab, dostarlimab, envafolimab, sintilimab, and zimberelimab.
- the method further comprises co-administering zimberelimab to the human patient.
- the breast cancer is metastatic breast cancer.
- the breast cancer is resistant or refractory to one or more anti-cancer therapies.
- the breast has progressed following prior anti-cancer therapy (first- or second-generation anti-cancer therapy, e.g., hormone therapy).
- the human patient has not received a prior therapy selected from taxane therapy (taxane naive), checkpoint inhibitor therapy (CPI naive), and topoisomerase I inhibitor therapy.
- the human patient has not received a prior taxane therapy (taxane naive), checkpoint inhibitor therapy (CPI naive), or topoisomerase I inhibitor therapy.
- taxane therapy comprises paclitaxel, nab-paclitaxel (ABRAXANE®), docetaxel, or cabazitaxel.
- the checkpoint inhibitor therapy comprises an anti-CTLA4 antibody or an anti- PD(L)1 antibody.
- the topoisomerase I inhibitor therapy comprises a topotecan, irinotecan, belotecan, or exatecan.
- the methods provided herein have anti-cancer effects as determined by one or more efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- efficacy endpoints selected from objective response rate (ORR), disease control rate (DCR), progression free survival (PFS), duration of response (DOR), overall survival (OS), complete response (CR), partial response (PR), radiographic response rate, and change from baseline in blood and tumor tissue microenvironment pharmacodynamic (PD) biomarkers.
- tumor response or progression is determined according to RECTST version 1.1.
- the human patient is not co-administcrcd an additional therapeutic agent selected from an MCL- 1 inhibitor, an anti-CD47 antibody, and a FLT3 agonist.
- a clinical study is conducted administering human patients with metastatic castrate resistant prostate cancer (mCRPC) with a combination of an anti-Trop-2 antibody and an adenosine receptor antagonist.
- mCRPC metastatic castrate resistant prostate cancer
- a subgroup of patients is treated with a combination of an anti-Trop-2 antibody, an adenosine receptor antagonist, and an anti-PD-(L)l antibody.
- human mCRPC patients are treated with a combination of sacituzumab govitecan and etrumadenant.
- human mCRPC patients are treated with a combination of sacituzumab govitecan (SG), etrumadenant, and zimberelimab.
- the patient population in this study can include previously treated mCRPC patients who have progressed on androgen deprivation therapy (ADT) and/or next generation hormonal agents (NHAs). mCRPC patients who are CPI and taxane naive can also be included. Additional patients can have RECIST 1.1 measurable or non-measurable disease.
- ADT androgen deprivation therapy
- NHAs next generation hormonal agents
- Primary endpoints in the study can include composite overall response rate (ORR; PSA/RECIST Response) and safety.
- Secondary endpoints can include ORR per RECIST 1.1, PSA response rate per PCWG3, disease control rate (DCR), or pharmacokinetics (PK).
- ORR per RECIST 1.1 PSA response rate per PCWG3, disease control rate (DCR), or pharmacokinetics (PK).
- DCR disease control rate
- PK pharmacokinetics
- Exploratory endpoints can include progression free survival, overall survival, or certain biomarkers. Investigational Products
- Sacituzumab govitecan is an antibody-drug conjugate (ADC) composed of the following 3 components: o The humanized monoclonal antibody hRS7 IgGlK, which binds to trophoblast cellsurface antigen 2 (Trop-2), a transmembrane calcium signal transducer that is overexpressed in many epithelial cancers, including triple-negative breast cancer (TNBC) and non-small cell lung cancer (NSCLC). o The camptothecin-derived agent SN-38, a topoisomerase I inhibitor. o A hydrolyzable linker CL2A that links the humanized monoclonal antibody to SN-38.
- ADC antibody-drug conjugate
- SG delivers significantly greater amounts of SN 38 to a Trop 2-cxprcssing tumor than conventional irinotecan chemotherapy (Sharkey RM, et al. Clinical Cancer Research (2015) 21(22):5131-8).
- the extracellular release of SN 38 from SG also allows for bystander killing of Trop 2 negative tumor cells (Lopez S, et al. Oncotarget (2020) l l(5):560;Perrone E, et al. Frontiers in Oncology (2020): 118; Zeybek B, et al. Scientific Reports (2020) 10(l):973)Thus
- SG can deliver cytotoxic chemotherapy to tumors, including adjacent cancer cells, in concentrations that are higher than those with standard chemotherapy and may reduce toxic effects in normal tissues that do not express the target.
- sacituzumab govitecan is generally administered at 10 mg/kg as an IV infusion on Days 1 and 8 of a 21-day cycle.
- Etrumadenant is generally administered at 10 mg/kg as an IV infusion on Days 1 and 8 of a 21-day cycle.
- Etrumadenant also known as AB928, GS-0928
- Etrumadenant is a low-molecular-weight, orally bioavailable, selective dual antagonist of adenosine receptors adenosine 2a receptor (A2aR) and adenosine 2b receptor (A2b ).
- A2aR adenosine 2a receptor
- A2b adenosine 2b receptor
- etrumadenant has been shown to selectively reverse the immunosuppressive effects caused by high concentrations of adenosine, without causing any immune activation effects on its own.
- Tn prostate cancer, A2bR is upregulated and the activity of prostatic acid phosphatase (PAP) produces additional adenosine, suggesting this tumor type may be more susceptible than others to adenosine-mediated immunosuppression.
- PAP prostatic acid phosphatase
- Etrumadenant can achieve high penetration of tumor tissue, robust potency in the presence of high adenosine concentrations, and only small shift in potency from nonspecific protein binding.
- Etrumadenant exhibits PK/pharmacodynamics consistent with once-daily dosing and has been well tolerated in dose escalation studies as a single agent and in Phase lb/2 studies administered in combination with chemo/immunotherapy across multiple advanced solid tumor indications.
- Tn the clinical studies described herein, etrumadenant is generally administered orally at a dose of 75 mg or 150 mg QD.
- Zimberelimab is a fully human IgG4 monoclonal antibody targeting human PD- 1.
- PD- 1 is a type I transmembrane protein that is part of the immunoglobulin gene superfamily and the CD28 family of cell surface receptors.
- the structure of PD-1 consists of 1 immunoglobulin variable-like extracellular domain and 1 cytoplasmic domain containing an immunoreceptor tyrosine-based inhibitory motif and an immunoreceptor tyrosine-based switch motif.
- PD-1 has 2 known ligands, PD-L1 (B7 Hl and CD274) and programmed cell death ligand 2 (PD-L2; B7 DC and CD73), which are members of the B7 family and are expressed on the plasma membrane of cancer cells and tumor-infiltrating leukocytes. Both PD-L1 and PD-L2 are B7 homologs that bind to PD-1, but they do not bind to other CD28 family members. [0229] PD-1 is an inhibitory immune checkpoint protein that is expressed on activated B cells, T cells, and myeloid cells, and it plays a key role in limiting the activity of effector T cells. It also provides a major resistance mechanism by which tumor cells can escape immune surveillance.
- PD-1 When activated by its ligands, PD-1 induces a state of anergy or unresponsiveness in T cells, and the cells are unable to produce optimal levels of effector cytokines or carry out other effector T- cell functions. PD-1 may also induce apoptosis in T cells via its ability to inhibit survival signals. Under normal circumstances, PD-1 is important for limiting the extent of T cell-mediated immune responses. PD- 1 -deficient animals develop various autoimmune phenotypes, including autoimmune cardiomyopathy and a lupus-like syndrome with arthritis and nephritis.
- PD-1/PD-L1 The interaction of PD-1 expressed on activated T cells and PD-L1 expressed on tumor cells negatively regulates immune response and dampens anticancer immunity.
- PD-L1 is abundantly expressed on a variety of human tumors, and its expression correlates with reduced patient survival in esophageal, pancreatic, and other types of cancers. Therefore, the PD-1/PD-L1 pathway is an important target for tumor immunotherapy.
- Activation of the PD-1/PD-L1 signaling pathway results in a decrease in tumor-infiltrating lymphocytes, a decrease in T cell proliferation, and an increase in immune evasion by cancerous cells.
- Immune suppression can be reversed by inhibiting the local interaction of PD-1 with PD-L1, and the effect is additive when the interaction of PD-1 with PD-L2 is also blocked.
- the selected dose of zimberelimab is generally 360 mg administered IV Q3W.
- the treatment arms will be conducted in 2 stages: Stage 1 and Stage 2. Depending on the treatment arm and stage, enrollment may or may not involve randomization. The decision to begin randomized enrollment in a specific treatment arm will be made by the sponsor for each combination therapy. Stage 1
- Stage 1 approximately 15 participants will be enrolled and will receive investigational products at the single agent recommended dose for expansion; standard of care therapies will be administered according to label instructions. The decision to begin enrollment in a specific treatment arm in Stage 1 will be made by the sponsor. The study includes the following anus:
- Etrumadenant Sacituzumab govitecan 10 mg/kg on Days 1 and 8 of a 21-day cycle + Etrumadenant 150 mg QD.
- SG+ Etrumadenant + Zimberelimab Sacituzumab govitecan 10 mg/kg on Days 1 and 8 of a 21-day cycle + Etrumadenant 150 mg QD + Zimberelimab 360 mg Q3W.
- CPI checkpoint inhibitor
- NHA new hormonal agent
- SG sacituzumab govitecan
- Safety assessments will consist of monitoring and recording AEs, including SAEs and AEs of special interest (AESI), performing protocol- specified safety laboratory assessments, measuring protocol- specified vital signs, and conducting other protocol- specified tests that are deemed critical to the safety evaluation of the study. All AEs and laboratory abnormalities will be graded according to the Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Objectives and Endpoints
- ORR Objective response rate
- PCWG3 Prostate Cancer Working Group 3
- ORR defined as the composite proportion of participants with a PSA and/or radiographic complete and partial response determined by PCWG3 criteria.
- Participants for biopsy should have at least 2 measurable lesions at baseline: 1 for tissue sampling and 1 for radiographic response assessment.
- HIV Negative human immunodeficiency virus
- HBsAg hepatitis B surface antigen
- HBcAb total hepatitis B core antibody
- HCV hepatitis C virus
- Prior anticancer treatment for the disease under study including approved agents, systemic radiotherapy, or investigational therapy, within 4 weeks (or 5 half-lives) prior to initiation of study treatment.
- Prior focal radiotherapy must be completed at least 2 weeks prior to the initiation of study treatment
- systemic immunostimulatory agents including, but not limited to, interferon and interleukin-2
- systemic immunostimulatory agents including, but not limited to, interferon and interleukin-2
- systemic immunosuppressive medication including, but not limited to, corticosteroids, cyclophosphamide, azathioprine, methotrexate, thalidomide, and antitumor necrosis factor-a agents
- systemic immunosuppressive medication including, but not limited to, corticosteroids, cyclophosphamide, azathioprine, methotrexate, thalidomide, and antitumor necrosis factor-a agents
- a Patients who received low dose ( ⁇ 10 mg/day prednisone or equivalent), systemic immunosuppressant medications or a 1-time pulse dose of systemic immunosuppressant medication (eg, 48 hours of corticosteroids for a contrast allergy) are eligible for the study after medical monitor approval has been obtained.
- CNS metastases a. Patients with a history of treated CNS metastases arc eligible, if all of the following criteria are met: i. The patient has no history of intracranial hemorrhage or spinal cord hemorrhage. ii. Metastases are limited to the cerebellum or the supratentorial region (ie, no metastases to the midbrain, pons, medulla, or spinal cord). iii. There is no evidence of interim progression between completion of CNS directed therapy and the screening brain scan. iv. The patient has not received stereotactic radiotherapy within 7 days prior to initiation of study treatment or whole brain radiotherapy within 14 days prior to initiation of study treatment.
- IV intravenous
- Severe infection within 4 weeks prior to initiation of study treatment including, but not limited to, hospitalization for complications of infection, bacteremia, or severe pneumonia
- Known allergy or hypersensitivity to any of the study drugs or their excipients may affect the interpretation of the results, or may render the patient at high risk from treatment complications 22.
- autoimmune disease or immune deficiency including, but not limited to, myasthenia gravis, myositis, autoimmune hepatitis, systemic lupus erythematosus, rheumatoid arthritis, inflammatory bowel disease, antiphospholipid antibody syndrome, Wegener granulomatosis, Sjogren syndrome, Guillain Barre syndrome, or multiple sclerosis with the following exceptions: a. Patients with a history of autoimmune related hypothyroidism who are on thyroid replacement hormone are eligible for the study. b. Patients with controlled Type 1 diabetes mellitus who, in the judgment of the investigator, arc on a stable insulin regimen arc eligible for the study. c.
- Treatment with known strong CYP3A4 inducers eg, rifampin, phenytoin, carbamazepine, phenobarbital, and St. John’s Wort
- strong CYP3A4 inhibitors eg, clarithromycin, grapefruit juice, itraconazole, ketoconazole, posaconazole, telithromycin, and voriconazole
- Sacituzumab govitecan 10 mg/kg, IV, on Days 1 and 8 of a 21-day cycle
- Etrumadenant 150 mg, oral, once daily continuous dosing (21-day cycle)
- Table 7 provides study intervention details: Table 7: Etrumadenant + sacituzumab govitecan with or without zimberelimab
- a participant may receive etrumadenant as a capsule or tablet but cannot receive both formulations.
- the dose of SG will be calculated based on actual weight at randomization (using weight obtained either at screening or on Cycle 1 Day 1) and remains constant throughout the study, unless there is a > 10% change in body weight from baseline. Modifications to the study drug doses administered should be made for a > 10% change in body weight from baseline and according to local and regional prescribing standards. Dose modifications for changes in body weight ⁇ 10% may be made according to local institutional guidelines. Sacituzumab govitecan is administered via IV infusion.
- Sacituzumab govitecan will be administered in 21-day cycles on Days 1 and 8; the next cycle should start a minimum 14 days after the Day 8 dose (i.e., the Day 8 infusion will be counted as the first day of that 14-day period).
- Etrumadenant should be taken at least 30 minutes prior to the start of IV infusion of SG. Sacituzumab govitecan is administered via IV infusion. The first infusion is administered over 3 hours. Subsequent infusions may be administered over 1 to 2 hours if previous infusions were well tolerated.
- Etrumadenant should be taken at least 30 minutes prior to the start of IV infusion of zimberelimab. Administer zimberelimab over a 60-minute ( ⁇ 5 min) IV infusion followed by a 30- minute observation (+15 min) interval. Following the zimberelimab observation period, sacituzumab govitecan should be administered as described above.
- Table 8 provides the study treatment administration schedule.
- 1 experimental treatment cycle is 21 days in duration (Cl, C2, C3, C4).
- Concomitant medications or treatments may be prescribed if considered necessary for adequate prophylactic or supportive care except for those medications identified as prohibited further below.
- Anticoagulant therapy including low-molecular-weight heparins, are allowed as clinically indicated. Applicable participants should undergo routine monitoring of coagulation parameters, including INR, according to institutional guidelines.
- Premedication is permitted for treatment with SG.
- Guidance for premedication for prevention of toxicities associated with SG is presented in Table 9.
- ANC absolute neutrophil count
- ASCO American Society of Clinical Oncology
- ESMO European Society for Medical Oncology
- IV intravenous
- PO orally
- SG Sacituzumab govitecan
- Systemic immuno stimulatory agents including but not limited to interferons and interleukin-2.
- Systemic immunosuppressive medications including but not limited to corticosteroids, cyclophosphamide, azathioprinc, methotrexate, thalidomide, and antitumor necrosis factor-a agents
- Systemic immunosuppressive medications including but not limited to corticosteroids, cyclophosphamide, azathioprinc, methotrexate, thalidomide, and antitumor necrosis factor-a agents
- acute, low dose ⁇ 10 mg/day prednisone or equivalent
- systemic immunosuppressant medications for management of AEs are allowed after medical monitor approval has been obtained.
- UGT1A1 inducers that should be avoided while receiving SG include: Carbamazepine, efavirenz, ethinylestradiol, lamotrigine, phenobarbital, phenytoin, primidone, rifampicin, ritonavir, and tipranavir.
- Stage 1 Participants will be enrolled into various treatment arms based on their prior cancer history. Enrollment may be paused after the accrual of approximately 6 participants in a treatment arm to allow for safety evaluation. A rate of toxicity of ⁇ 33% will be targeted, with toxicity being defined as the occurrence of either a treatment-related Grade 4 AE or a treatment- related Grade 3 AE that is not responsive to supportive care during Cycle 1. If 2 or more such events are observed, enrollment in that arm may not continue.
- Radiographic response is defined as either a CR or PR by RECIST vl.l
- PSA response is defined as percent change from baseline PSA of > 50%.
- At least 1 radiographic CR or PR or 2 PSA responses out of 15 evaluable participants must be observed in order to consider opening Stage 2. In this case, across a range of prior beta distributions, observing 1 or fewer PSA responses would provide greater than 80% posterior probability confidence that the PSA response rate is less than 20%.
- Participants must have at least 1 postbaseline radiographic disease evaluation or consecutive postbaseline PSA assessments measured at least 3 to 4 weeks apart to be considered evaluable for Stage 1 decision gating.
- Stage 2 up to 25 additional participants may be enrolled either onto an experimental arm or a standard of care control arm. The goal of this stage is to better characterize potential treatment differences between experimental arms and standard of care among concurrently enrolled participants. Representative estimates of 90% confidence intervals for potential differences in response between treatment arms are provided. Safety Endpoints
- Safety analyses will be conducted in the safety-evaluable population, defined as all participants who are enrolled and receive any amount of study treatment. Safety will be assessed through summaries of AEs, as well as relevant changes in laboratory test results, vital signs, and electrocardiograms. Verbatim AE terms will be mapped and coded using the Medical Dictionary for Regulatory Activities. All AEs will be assessed for severity according to National Cancer Institute Common Terminology Criteria for Adverse Events (NCI CTCAE) v5.0.
- NCI CTCAE National Cancer Institute Common Terminology Criteria for Adverse Events
- Treatment-emergent adverse events defined as AEs with onset date on or after initiation of study treatment or as AEs present at baseline but which worsened after initiation of study treatment, will be summarized. Adverse events will be summarized on the participant-level using the maximum severity or grade reported. All TEAEs, SAEs, AEs leading to study treatment discontinuation, Grade 3 or higher AEs, and deaths will be listed and summarized by treatment arm with the mapped preferred terms, thesaurus level and NCI CTCAE grade, as appropriate.
- Efficacy analyses will be primarily conducted in the efficacy-evaluable population, defined as all participants who are enrolled and receive at least 1 dose of each drug of their assigned treatment regimen. The following prioritized efficacy endpoints will be analyzed according to PCWG3 and based on the investigator’s assessment:
- ORR defined as the composite proportion of participants with a PSA response or a radiographic CR or PR (defined below).
- PSA response defined as the proportion of participants with a confirmed PSA decrease from baseline of 50% or more based on 2 consecutive assessments measured at least 3 to 4 weeks apart.
- Radiographic response defined as the proportion of participants with a best overall response of CR or PR according to RECIST vl.l.
- DCR is defined as the proportion of participants with a best overall RECIST response of CR, PR, or stable disease (SD).
- PFS is defined as the time from treatment assignment until first documentation of progressive disease (PSA progression, radiographic progression, bone scan progression, or other) or death, whichever occurs first. Depending on data availability, time to progression due to specific reasons (e.g., PSA progression or radiographic progression) may also be presented individually.
- OS is defined as the time from treatment assignment until death due to any cause.
- the primary efficacy endpoint is the ORR, defined as the composite proportion of participants with a PSA response or a radiographic complete or partial response determined by the investigator according to the PCWG3 criteria. PSA response and radiographic complete or partial response may be reported individually. For clarity, PSA response is defined as the proportion of participants with a confirmed PSA decrease from baseline of 50% or more based on 2 consecutive assessments measured 3 to 4 weeks apart. Radiographic response is defined as the percentage of participants with measurable disease at baseline who achieve a best overall response of CR or PR according to RECIST vl.l.
- tumor assessments every 12 weeks ( ⁇ 7 days) from start of study treatment until disease progression (regardless of whether the participant is still receiving treatment), the start of new anticancer therapy, withdrawal of consent, death, or the end of the study.
- tumor assessments may be repeated at any time if disease progression is suspected. All participants who discontinue study treatment for reasons other than disease progression (e.g., AEs) will continue tumor assessments until death, disease progression, initiation of another systemic anticancer therapy, lost to follow-up, withdrawal of consent, or study termination, whichever occurs first.
- Measurable and evaluable lesions should be assessed and documented at screening. Tumor assessments performed as standard of care prior to obtaining informed consent and within 28 days prior to enrollment do not have to be repeated at screening.
- Baseline disease assessments for all participants will include: CT scan with contrast of the chest and abdomen/pelvis, Full-body bone scan (technetium-99m [TC-99m]), and MRI scan of the brain.
- Postbaseline disease assessments for all participants will include:
- All scans should be performed in accordance with RECIST vl.l and with contrast. If contrast is medically contraindicated, a chest CT without contrast and an abdomen/pelvis MRI without contrast may be performed. If a brain MRI is contraindicated, a brain CT with contrast should be performed. If a CT scan for tumor assessment is performed in a positron emission tomography (PET)/CT scanner, the CT acquisition must be consistent with standard for a full contrast diagnostic CT scan.
- PET positron emission tomography
- All measurable and evaluable lesions identified at baseline should be reassessed at each subsequent postbaseline tumor evaluation.
- the same radiographic procedures used to assess disease sites at screening should be used for subsequent tumor assessments (e.g., same contrast protocol for CT scans). Response will be assessed by the investigator using PCWG3. Assessments should be performed by the same evaluator, if possible, to ensure internal consistency across disease evaluations. Results must be reviewed by the investigator before dosing at the next planned study treatment cycle.
- An adverse event is any untoward medical occurrence in a clinical investigation participant administered a pharmaceutical product regardless of causal attribution.
- An AE can therefore be any of the following: Any unfavorable and unintended sign (including an abnormal laboratory finding), symptom, or disease temporally associated with the use of a medicinal product, whether or not considered related to the medicinal product
- a serious adverse event is any AE that meets any of the following criteria:
- Severity refers to the intensity of an AE (e.g., rated as mild, moderate, or severe, or according to the National Cancer Institute [NCI] CTCAE); the event itself may be of relatively minor medical significance (such as severe headache without any further findings). Assessment of Severity
- CTCAE Instrumental activities of daily living refer to preparing meals, shopping for groceries or clothes, using the telephone, managing money, etc.
- Examples of self-care activities of daily living include bathing, dressing and undressing, feeding oneself, using the toilet, and taking medications, as performed by patients who are not bedridden.
- c If an event is assessed as a “significant medical event”, it must be reported as an SAE d Grade 4 and 5 events must be reported as SAEs should the event meet the SAE definition.
- This study will evaluate the efficacy and safety of combination treatment regimens for patients with advanced or metastatic NSCLC with progression on or recurrence after platinumbased chemotherapy and PD-1/PD-L1 immunotherapy, received either in combination or sequentially.
- Participants must have a diagnosis of advanced or metastatic squamous or non-squamous NSCLC.
- Participants with EGFR, ALK, or any other known actionable genomic alterations must have also received treatment with at least 1 approved tyrosine kinase inhibitor appropriate to the genomic alteration.
- participants will be randomly assigned to an experimental arm. Randomization will be stratified by histology (squamous versus non-squamous) and prior therapy for an actionable genomic alteration (yes versus no).
- participant will be randomly assigned to the comparator arm or an experimental arm. Participants in the comparator arm will receive either sacituzumab govitecan or docetaxel, with the comparator arm choice based on the treatment landscape at the time of initiation of the expansion stage.
- ORR Objective response rate
- PFS Progression-free survival
- DOR Duration of response
- OS overall survival
- Tumor biopsy must have been performed on or after progression on prior line of therapy and before enrollment with no anticancer treatment between collection of tissue and enrollment.
- Topoisomerase 1 inhibitors Any agent including an ADC containing a chemotherapeutic agent targeting topoisomerase 1.
- Trop-2-targeted therapy c) Docetaxel as monotherapy or in combination with other agents.
- Docetaxel as monotherapy or in combination with other agents.
- Sacituzumab govitecan, etrumadenant, and zimberelimab are as described in Example 1.
- Etrumadenant is administered orally, followed by zimberelimab IV, followed by sacituzumab govitecan IV.
- the comparator arm during the expansion phase of the study will be either docetaxel or sacituzumab govitecan monotherapy (Table 12).
- EGFR testing will be performed using the Cobas® EGFR Mutation Test (Roche).
- ALK the Vysis ALK Break Apart FISH probe test (Abbott) will be used.
- Additional biomarkers in blood and tissue may include, but are not limited to, protein expression, analyses of specific immune and tumor signatures (RNA), as well as tumor mutational burden and tumor mutations (DNA). Tumor and blood samples will be collected to measure biomarkers of response and resistance and to better understand molecular attributes predictive of treatment in lung cancer. Examples may include, but will not be limited to, PD-L1 and Trop-2 - expression, other proteins as well as mutations/gene expression (WES/RNAseq) related to any of the study treatments or related to lung cancer, tumor mutational burden, oncogenic mutations, composition of immune subsets in tumor microenvironment, and pathological features of the tumor.
- WES/RNAseq mutations/gene expression
- hRS7 mAb, SG, Rab64 Ab and surrogate SG to Trop-2 was characterized by surface plasmon resonance (SPR, Table 13) and demonstrated that surrogate SG bound to both mouse and human Trop-2 in the low nanomolar range (1 .37 nM to mTrop-2 and 1.1 nM to hTrop-2).
- a mouse isotype antibody control (mMAB1129/mG2a/mKap) was used as control ADC and coupled to CL2A linker/SN-38 payload in the same condition that Rab64 mAb.
- Table 14 summarizes the ADC constructs.
- the purpose of this study is to determine the in vivo efficacy of murinized SG combined to anti-mouse PD1 and/or quemliclustat in an immunocompetent mouse model of orthotopic breast cancer expressing human Trop-2.
- mice were acquired from Biocytogen. Female (5-7 weeks old) are injected into the left fourth mammary fat pad with huTrop-2 transduced EO771 tumor cells. When the tumor volume reaches a mean volume of 100 mm 3 , mice are randomized into 12 groups (10 mice per group) and treated for four weeks with
- Murinized SG ADC was produced as described in Example 3.
- the anti-mouse PD1 antibody RMP1.14 mlgGl D265A is a commercially available antibody that can be purchased, for example, from Invivogen (cat. code mpdl-mabl5-l).
- Palpable tumors are measured with calipers. Mice are sacrificed when tumor sizes reached -2000 mm 3 . It is contemplated that the group treated with triple combinations (group 8) will survive the longest due to the combination of the anti-tumor effect of murinized SG to the activation of the tumor infiltrate via IO drugs.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Immunology (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- Cell Biology (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- Mycology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Biophysics (AREA)
- Oncology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biomedical Technology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Endocrinology (AREA)
- Reproductive Health (AREA)
- Urology & Nephrology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Peptides Or Proteins (AREA)
- Medicinal Preparation (AREA)
Abstract
Description
Claims
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202380033738.5A CN119173276A (en) | 2022-04-13 | 2023-04-12 | Combination therapy for the treatment of Trop-2 expressing cancers |
| CA3256033A CA3256033A1 (en) | 2022-04-13 | 2023-04-12 | Combination therapy for treating trop-2 expressing cancers |
| KR1020247037701A KR20250004779A (en) | 2022-04-13 | 2023-04-12 | Combination therapy for treating TROP-2 expressing cancers |
| JP2024560326A JP2025512384A (en) | 2022-04-13 | 2023-04-12 | Combination Therapies for Treating TROP-2-Expressing Cancers |
| AU2023252914A AU2023252914A1 (en) | 2022-04-13 | 2023-04-12 | Combination therapy for treating trop-2 expressing cancers |
| EP23721581.9A EP4507736A1 (en) | 2022-04-13 | 2023-04-12 | Combination therapy for treating trop-2 expressing cancers |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202263330700P | 2022-04-13 | 2022-04-13 | |
| US63/330,700 | 2022-04-13 | ||
| US202263370228P | 2022-08-02 | 2022-08-02 | |
| US63/370,228 | 2022-08-02 | ||
| US202263377918P | 2022-09-30 | 2022-09-30 | |
| US63/377,918 | 2022-09-30 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023201267A1 true WO2023201267A1 (en) | 2023-10-19 |
Family
ID=86328489
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2023/065682 Ceased WO2023201267A1 (en) | 2022-04-13 | 2023-04-12 | Combination therapy for treating trop-2 expressing cancers |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP4507736A1 (en) |
| JP (1) | JP2025512384A (en) |
| KR (1) | KR20250004779A (en) |
| CN (1) | CN119173276A (en) |
| AU (1) | AU2023252914A1 (en) |
| CA (1) | CA3256033A1 (en) |
| TW (1) | TW202345845A (en) |
| WO (1) | WO2023201267A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025186213A1 (en) | 2024-03-04 | 2025-09-12 | Debiopharm International S.A. | Combination of a wee1 inhibitor and a topoisomerase 1 inhibitor |
| WO2025188583A1 (en) * | 2024-03-06 | 2025-09-12 | Merck Sharp & Dohme Llc | Combination therapies for the treatment of hr+/her2- metastatic or locally advanced breast cancer |
| WO2025190251A1 (en) * | 2024-03-12 | 2025-09-18 | 百奥泰生物制药股份有限公司 | Use of anti-trop2 antibody-drug conjugate and pd-1 antagonist in combined treatment of solid tumor |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| AU2023254836A1 (en) * | 2022-04-13 | 2024-11-07 | Arcus Biosciences, Inc. | Combination therapy for treating tumor antigen expressing cancers |
| CN119592567B (en) * | 2024-12-10 | 2025-12-02 | 浙江大学 | Antisense oligonucleotides based on tumor splicing transformation therapy and their applications |
Citations (220)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996011013A1 (en) | 1994-10-05 | 1996-04-18 | Immunomedics, Inc. | Cdr-grafted type iii anti-cea humanized mouse monoclonal antibodies |
| WO2003074566A2 (en) | 2002-03-01 | 2003-09-12 | Immunomedics, Inc. | Rs7 antibodies |
| US6624821B1 (en) | 1999-02-05 | 2003-09-23 | Samsung Electronics Co., Ltd. | Image texture retrieving method and apparatus thereof |
| US20060193865A1 (en) | 2002-12-13 | 2006-08-31 | Immunomedics, Inc. | Camptothecin-binding moiety conjugates |
| WO2006094192A2 (en) | 2005-03-03 | 2006-09-08 | Immunomedics, Inc. | Humanized l243 antibodies |
| WO2007095749A1 (en) | 2006-02-24 | 2007-08-30 | Arius Research Inc. | Cytotoxicity mediation of cells evidencing surface expression of trop-2 |
| WO2007103776A2 (en) | 2006-03-02 | 2007-09-13 | Cv Therapeutics, Inc. | A2a adenosine receptor antagonists |
| WO2007134958A1 (en) | 2006-05-18 | 2007-11-29 | F. Hoffmann-La Roche Ag | Thiazolo-pyramidine / pyridine urea derivatives as adenosine a2b receptor antagonists |
| WO2008086201A1 (en) | 2007-01-05 | 2008-07-17 | Biogen Idec Ma Inc. | Polymorphs and solvates of a pharmaceutical and methods of making |
| WO2009009178A2 (en) | 2007-04-06 | 2009-01-15 | The General Hospital Corporation | Systems and methods for optical imaging using early arriving photons |
| WO2009033161A1 (en) | 2007-09-07 | 2009-03-12 | The John Hopkins University | Adenosine receptor agonists and antagonists to modulate t cell responses |
| WO2009037463A1 (en) | 2007-09-21 | 2009-03-26 | Vernalis (R & D) Ltd. | Thienopyrimidine compounds |
| WO2009037468A1 (en) | 2007-09-21 | 2009-03-26 | Vernalis (R & D) Ltd. | Thienopyrimidine compounds and compositions |
| WO2009037467A1 (en) | 2007-09-21 | 2009-03-26 | Vernalis (R & D) Ltd. | Pyrrolopyrimidine compounds |
| WO2009055308A1 (en) | 2007-10-24 | 2009-04-30 | Janssen Pharmaceutica, N.V. | Arylindenopyrimidines and their use as adenosine a2a receptor antagonists |
| WO2009055548A1 (en) | 2007-10-25 | 2009-04-30 | Janssen Pharmaceutica, N.V. | Arylindenopyrimidines and their use as adenosine a2a receptor antagonists |
| WO2009095478A1 (en) | 2008-01-31 | 2009-08-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Antibodies against human cd39 and use thereof for inhibiting t regulatory cells activity |
| WO2010008775A1 (en) | 2008-06-23 | 2010-01-21 | Ligand Pharmaceuticals Inc. | Aminopyridopyrazinone derivatives for treating neurodegenerative diseases |
| WO2010093395A1 (en) | 2009-02-13 | 2010-08-19 | Immunomedics, Inc. | Immunoconjugates with an intracellularly-cleavable linkage |
| WO2011027805A1 (en) | 2009-09-02 | 2011-03-10 | 協和発酵キリン株式会社 | Therapeutic agent for mood disorders |
| WO2011050160A1 (en) | 2009-10-22 | 2011-04-28 | Biogen Idec Ma Inc. | Pharmaceuticals, compositions and methods of making and using the same |
| WO2011055391A1 (en) | 2009-11-09 | 2011-05-12 | Advinus Therapeutics Private Limited | Substituted fused pyrimidine compounds, its preparation and uses thereof |
| WO2011068845A1 (en) | 2009-12-02 | 2011-06-09 | Immunomedics, Inc. | Combining radioimmunotherapy and antibody-drug conjugates for improved cancer therapy |
| WO2012085132A1 (en) | 2010-12-22 | 2012-06-28 | Orega Biotech | Antibodies against human cd39 and use thereof |
| WO2012135084A1 (en) | 2011-03-31 | 2012-10-04 | Merck Sharp & Dohme Corp. | METABOLITES OF 2-(FURAN-2-YL)-7-(2-(4-(4-(2-METHOXYETHOXY)PHENYL)PIPERAZIN-1-YL)ETHYL)-7H-PYRAZOLO[4,3-e][1,2,4]TRIAZOLO[1,5-c]PYRIMIDIN-5-AMINE AND THEIR UTILITY AS ADENOSINE A2a RECEPTOR ANTAGONISTS |
| US20130039861A1 (en) | 2005-04-06 | 2013-02-14 | Immunomedics, Inc. | Dye Conjugated Peptides for Fluorescent Imaging |
| US8435529B2 (en) | 2002-06-14 | 2013-05-07 | Immunomedics, Inc. | Combining radioimmunotherapy and antibody-drug conjugates for improved cancer therapy |
| US8435539B2 (en) | 2004-02-13 | 2013-05-07 | Immunomedics, Inc. | Delivery system for cytotoxic drugs by bispecific antibody pretargeting |
| WO2013068946A2 (en) | 2011-11-11 | 2013-05-16 | Rinat Neuroscience Corp. | Antibodies specific for trop-2 and their uses |
| WO2013077458A1 (en) | 2011-11-22 | 2013-05-30 | 株式会社リブテック | Anti-human trop-2 antibody exhibiting antitumor activity in vivo |
| WO2014092804A1 (en) | 2012-12-13 | 2014-06-19 | Immunomedics, Inc. | Dosages of immunoconjugates of antibodies and sn-38 for improved efficacy and decreased toxicity |
| WO2014101113A1 (en) | 2012-12-28 | 2014-07-03 | Merck Sharp & Dohme Corp. | Piperazine-substituted 7-methoxy-[1,2,4]triazolo[1,5-c]quinazolin-5-amine compounds with a2a antagonist properties |
| WO2014101120A1 (en) | 2012-12-28 | 2014-07-03 | Merck Sharp & Dohme Corp. | Heterobicyclo-substituted-7-methoxy-[1,2,4]triazolo[1,5-c]quinazolin-5-amine compounds with a2a antagonist properties |
| WO2014106861A1 (en) | 2013-01-03 | 2014-07-10 | Council Of Scientific & Industrial Research | (4e)-4-(4-substituted benzylideneamino)-2,3-dihydro-3- substituted-2-thioxothiazole-5-carbonitriles as a2ar antagonist and process for preparation thereof |
| WO2014163684A1 (en) | 2013-04-03 | 2014-10-09 | Ibc Pharmaceuticals, Inc. | Combination therapy for inducing immune response to disease |
| WO2015027431A1 (en) | 2013-08-29 | 2015-03-05 | Merck Sharp & Dohme Corp. | 2,2-difluorodioxolo a2a receptor antagonists |
| WO2015098099A1 (en) | 2013-12-25 | 2015-07-02 | 第一三共株式会社 | Anti-trop2 antibody-drug conjugate |
| WO2015164573A1 (en) | 2014-04-25 | 2015-10-29 | Vitae Pharmaceuticals, Inc. | Purine derivatives as cd73 inhibitors for the treatment of cancer |
| WO2016055609A1 (en) | 2014-10-10 | 2016-04-14 | Innate Pharma | Cd73 blockade |
| WO2016073845A1 (en) | 2014-11-07 | 2016-05-12 | Igenica Biotherapeutics, Inc. | Anti-cd39 antibodies and uses thereof |
| WO2016075099A1 (en) | 2014-11-10 | 2016-05-19 | Medimmune Limited | Binding molecules specific for cd73 and uses thereof |
| WO2016081290A1 (en) | 2014-11-18 | 2016-05-26 | Merck Sharp & Dohme Corp. | Aminopyrazine compounds with a2a antagonist properties |
| WO2016081748A2 (en) | 2014-11-21 | 2016-05-26 | Bristol-Myers Squibb Company | Antibodies against cd73 and uses thereof |
| WO2016081746A2 (en) | 2014-11-21 | 2016-05-26 | Bristol-Myers Squibb Company | Antibodies comprising modified heavy constant regions |
| WO2016126570A1 (en) | 2015-02-06 | 2016-08-11 | Merck Sharp & Dohme Corp. | Aminoquinazoline compounds as a2a antagonist |
| US20160296633A1 (en) | 2013-07-23 | 2016-10-13 | Immunomedics, Inc. | Combining anti-hla-dr or anti-trop-2 antibodies with microtubule inhibitors, parp inhibitors, bruton kinase inhibitors or phosphoinositide 3-kinase inhibitors significantly improves therapeutic outcome in cancer |
| US9492566B2 (en) | 2012-12-13 | 2016-11-15 | Immunomedics, Inc. | Antibody-drug conjugates and uses thereof |
| WO2016200717A1 (en) | 2015-06-11 | 2016-12-15 | Merck Sharp & Dohme Corp. | Aminopyrazine compounds with a2a antagonist properties |
| WO2016201300A1 (en) | 2015-06-12 | 2016-12-15 | Immunomedics, Inc. | Disease therapy with chimeric antigen receptor (car) constructs and t cells (car-t) or nk cells (car-nk) expressing car constructs |
| WO2016209787A1 (en) | 2015-06-26 | 2016-12-29 | Merck Sharp & Dohme Corp. | Sustained release formulation and tablets prepared therefrom |
| WO2017008205A1 (en) | 2015-07-10 | 2017-01-19 | Merck Sharp & Dohme Corp. | Substituted aminoquinazoline compounds as a2a antagonist |
| US20170021017A1 (en) | 2012-08-14 | 2017-01-26 | Ibc Pharmaceuticals, Inc. | Combination therapy for inducing immune response to disease |
| WO2017025051A1 (en) | 2015-08-11 | 2017-02-16 | Wuxi Biologics (Shanghai) Co. Ltd. | Novel anti-pd-1 antibodies |
| WO2017064043A1 (en) | 2015-10-12 | 2017-04-20 | Innate Pharma | Cd73 blocking agents |
| WO2017100670A1 (en) | 2015-12-09 | 2017-06-15 | Corvus Pharmaceuticals, Inc. | Humanized anti-cd73 antibodies |
| WO2017098421A1 (en) | 2015-12-08 | 2017-06-15 | Glaxosmithkline Intellectual Property Development Limited | Benzothiadiazine compounds |
| WO2017118613A1 (en) | 2016-01-08 | 2017-07-13 | Syddansk Universitet | Bispecific antibodies targeting human cd73 |
| WO2017120508A1 (en) | 2016-01-08 | 2017-07-13 | Arcus Biosciences, Inc. | Modulators of 5'-nucleotidase, ecto and the use thereof |
| US20170209594A1 (en) | 2015-06-25 | 2017-07-27 | Immunomedics, Inc. | Synergistic effect of anti-trop-2 antibody-drug conjugate in combination therapy for triple-negative breast cancer when used with microtubule inhibitors or parp inhibitors |
| WO2017153952A1 (en) | 2016-03-10 | 2017-09-14 | Glaxosmithkline Intellectual Property Development Limited | 5-sulfamoyl-2-hydroxybenzamide derivatives |
| WO2017157948A1 (en) | 2016-03-14 | 2017-09-21 | Innate Pharma | Anti-cd39 antibodies |
| WO2018013611A1 (en) | 2016-07-11 | 2018-01-18 | Corvus Pharmaceuticals, Inc. | Anti-cd73 antibodies |
| WO2018013951A1 (en) | 2016-07-15 | 2018-01-18 | Northwestern University | Targeting adenosine a2a receptors for the treatment of levodopa-induced dyskinesias |
| WO2018036428A1 (en) | 2016-08-22 | 2018-03-01 | 广东纳路纳米科技有限公司 | Metal nanowire-oxidation-resistant material combined transparent conductive film and preparation therefor |
| WO2018049145A1 (en) | 2016-09-09 | 2018-03-15 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2018065627A1 (en) | 2016-10-07 | 2018-04-12 | Secarna Pharmaceuticals Gmbh & Co Kg | Immunosuppression-reverting oligonucleotides inhibiting the expression of cd73 |
| WO2018065622A1 (en) | 2016-10-07 | 2018-04-12 | Secarna Pharmaceuticals Gmbh & Co Kg | Immunosuppression-reverting oligonucleotides inhibiting the expression of cd39 |
| WO2018067424A1 (en) | 2016-10-03 | 2018-04-12 | Arcus Biosciences, Inc. | Inhibitors of adenosine 5'-nucleotidase |
| WO2018065552A1 (en) | 2016-10-06 | 2018-04-12 | Innate Pharma | Anti-cd39 antibodies |
| US20180110772A1 (en) | 2012-12-13 | 2018-04-26 | Immunomedics, Inc. | Therapy for metastatic urothelial cancer with the antibody-drug conjugate, sacituzumab govitecan (immu-132) |
| WO2018094148A1 (en) | 2016-11-18 | 2018-05-24 | Arcus Biosciences, Inc. | Inhibitors of cd73-mediated immunosuppression |
| WO2018110555A1 (en) | 2016-12-13 | 2018-06-21 | アステラス製薬株式会社 | Anti-human cd73 antibody |
| WO2018119284A1 (en) | 2016-12-22 | 2018-06-28 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| US20180185351A1 (en) | 2012-12-13 | 2018-07-05 | Immunomedics, Inc. | Therapy of small-cell lung cancer (sclc) with a topoisomerase-i inhibiting antibody-drug conjugate (adc) targeting trop-2 |
| WO2018136700A1 (en) | 2017-01-20 | 2018-07-26 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| WO2018137598A1 (en) | 2017-01-24 | 2018-08-02 | I-Mab | Anti-cd73 antibodies and uses thereof |
| CN108440674A (en) | 2018-04-28 | 2018-08-24 | 杭州荣泽生物科技有限公司 | A kind of Trop-2 specific chimerics antigen receptor cell is prepared and application thereof |
| US20180271992A1 (en) | 2017-03-27 | 2018-09-27 | Immunomedics, Inc. | Treatment of high trop-2 expressing triple negative breast cancer (tnbc) with sacituzumab govitecan (immu-132) overcomes homologous recombination repair (hrr) rescue mediated by rad51 |
| WO2018183635A1 (en) | 2017-03-31 | 2018-10-04 | Peloton Therapeutics, Inc. | Cd73 inhibitors and uses thereof |
| WO2018195321A1 (en) | 2017-04-20 | 2018-10-25 | Gilead Sciences, Inc. | Pd-1/pd-l1 inhibitors |
| WO2018208727A1 (en) | 2017-05-08 | 2018-11-15 | Eternity Bioscience Inc. | Nucleoside and nucleotide analogues as cd73 inhibitors and therapeutic uses thereof |
| WO2018208980A1 (en) | 2017-05-10 | 2018-11-15 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2018215535A1 (en) | 2017-05-23 | 2018-11-29 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Novel cd73 antibody, preparation and uses thereof |
| WO2018217227A1 (en) | 2017-05-24 | 2018-11-29 | Immunomedics, Inc. | Novel anti-pd-1 checkpoint inhibitor antibodies that block binding of pd-l1 to pd-1 |
| WO2018237173A1 (en) | 2017-06-22 | 2018-12-27 | Novartis Ag | ANTIBODY MOLECULES DIRECTED AGAINST CD73 AND CORRESPONDING USES |
| WO2018237157A1 (en) | 2017-06-22 | 2018-12-27 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| WO2019027935A1 (en) | 2017-07-31 | 2019-02-07 | Tizona Therapeutics | Anti-cd39 antibodies, compositions comprising anti-cd39 antibodies and methods of using anti-cd39 antibodies |
| US20190048095A1 (en) | 2017-08-11 | 2019-02-14 | Bio-Thera Solutions, Ltd. | Compounds and methods for the treatment of trop2 positive diseases |
| WO2019053617A1 (en) | 2017-09-12 | 2019-03-21 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| WO2019090111A1 (en) | 2017-11-03 | 2019-05-09 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2019086074A1 (en) | 2017-10-31 | 2019-05-09 | Helmholtz-Zentrum Dresden-Rossendorf E.V. | 4-(furan-2-yl)-1h-pyrazolo[3,4-d]pyrimidin-6-amine derivative and use thereof |
| WO2019118313A1 (en) | 2017-12-13 | 2019-06-20 | Merck Sharp & Dohme Corp. | Imidazo [1,2-c] quinazolin-5-amine compounds with a2a antagonist properties |
| WO2019114666A1 (en) | 2017-12-15 | 2019-06-20 | 四川科伦博泰生物医药股份有限公司 | Bioactive conjugate, preparation method therefor and use thereof |
| WO2019129059A1 (en) | 2017-12-29 | 2019-07-04 | 上海和誉生物医药科技有限公司 | Phosphonic acid derivative having cd73 inhibitory activity, and preparation method and use thereof |
| US20190248917A1 (en) | 2016-02-10 | 2019-08-15 | Immunomedics, Inc. | Combination of abcg2 inhibitors with sacituzumab govitecan (immu-132) overcomes resistance to sn-38 in trop-2 expressing cancers |
| WO2019160882A1 (en) | 2018-02-13 | 2019-08-22 | Gilead Sciences, Inc. | Pd-1/pd-l1 inhibitors |
| WO2019168744A1 (en) | 2018-03-01 | 2019-09-06 | Eli Lilly And Company | Cd73 inhibitors |
| WO2019166701A1 (en) | 2018-03-02 | 2019-09-06 | Turun Yliopisto | Anti-adenosine signaling pathway antibodies conjugated or fused with adenosine deaminase or capable of binding adenosine deaminase |
| WO2019170131A1 (en) | 2018-03-07 | 2019-09-12 | 复旦大学 | Targeted cd73 antibody and antibody-drug conjugate, and preparation method therefor and uses thereof |
| WO2019173692A2 (en) | 2018-03-09 | 2019-09-12 | Agenus Inc. | Anti-cd73 antibodies and methods of use thereof |
| WO2019173291A1 (en) | 2018-03-09 | 2019-09-12 | Phanes Therapeutics, Inc. | Anti-cd73 antibodies and uses thereof |
| WO2019178269A2 (en) | 2018-03-14 | 2019-09-19 | Surface Oncology, Inc. | Antibodies that bind cd39 and uses thereof |
| WO2019224025A2 (en) | 2018-05-19 | 2019-11-28 | Boehringer Ingelheim International Gmbh | Antagonizing cd73 antibody |
| WO2019232244A2 (en) | 2018-05-31 | 2019-12-05 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| US10501555B2 (en) | 2014-12-04 | 2019-12-10 | Abruzzo Theranostic S.R.L. | Humanized anti-trop-2 monoclonal antibodies and uses thereof |
| WO2019246403A1 (en) | 2018-06-21 | 2019-12-26 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2020014643A1 (en) | 2018-07-13 | 2020-01-16 | Gilead Sciences, Inc. | Pd-1/pd-l1 inhibitors |
| WO2020016662A2 (en) | 2018-07-09 | 2020-01-23 | Abmart Inc. | Antibodies specific to trophoblast antigen 2 (trop2) |
| WO2020047082A1 (en) | 2018-08-28 | 2020-03-05 | Eternity Bioscience Inc. | Cd73 inhibitors and therapeutic uses thereof |
| WO2020046813A1 (en) | 2018-08-27 | 2020-03-05 | Arcus Biosciences, Inc. | Cd73 inhibitors |
| WO2020053263A1 (en) | 2018-09-11 | 2020-03-19 | Iteos Therapeutics S.A. | Thiocarbamate derivatives as a2a inhibitors, pharmaceutical composition thereof and combinations with anticancer agents |
| WO2020051686A1 (en) | 2018-09-11 | 2020-03-19 | Risen (Suzhou) Pharma Tech Co. Ltd. | Cd73 inhibitors and pharmaceutical uses thereof |
| WO2020097127A2 (en) | 2018-11-05 | 2020-05-14 | Corvus Pharmaceuticals, Inc. | B-cell activating cd73 antibodies |
| WO2020094670A1 (en) | 2018-11-05 | 2020-05-14 | Synaffix B.V. | Antibody-conjugates for targeting of tumours expressing trop-2 |
| WO2020098599A1 (en) | 2018-11-12 | 2020-05-22 | 江苏恒瑞医药股份有限公司 | Anti-cd73 antibody, antigen-binding fragment thereof and application thereof |
| WO2020106560A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020106558A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020103939A1 (en) | 2018-11-22 | 2020-05-28 | 上海科技大学 | Triazolo cycle compound, preparation method therefor, intermediate thereof and application thereof |
| WO2020103930A1 (en) | 2018-11-22 | 2020-05-28 | 上海科技大学 | Thiazolo ring compound, preparation method therefor, intermediate thereof, and application thereof |
| WO2020112706A1 (en) | 2018-11-30 | 2020-06-04 | Merck Sharp & Dohme Corp. | 7-, 8-, and 10-SUBSTITUTED AMINO TRIAZOLO QUINAZOLINE DERIVATIVES AS ADENOSINE RECEPTOR ANTAGONISTS, PHARMACEUTICAL COMPOSITIONS AND THEIR USE |
| WO2020112700A1 (en) | 2018-11-30 | 2020-06-04 | Merck Sharp & Dohme Corp. | 9-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020130125A1 (en) | 2018-12-21 | 2020-06-25 | 第一三共株式会社 | Combination of antibody-drug conjugate and kinase inhibitor |
| WO2020139803A1 (en) | 2018-12-24 | 2020-07-02 | Dcb-Usa Llc | Benzothiadiazine derivatives and compositions comprising the same for treating disorders mediated by adenosine |
| WO2020135195A1 (en) | 2018-12-28 | 2020-07-02 | 四川科伦博泰生物医药股份有限公司 | Aminopyridine compound, preparation method therefor and use thereof |
| WO2020135210A1 (en) | 2018-12-28 | 2020-07-02 | 四川科伦博泰生物医药股份有限公司 | Substituted aryl compound and preparation method therefor and use thereof |
| WO2020143710A1 (en) | 2019-01-11 | 2020-07-16 | 康诺亚生物医药科技(成都)有限公司 | Anti-cd73 monoclonal antibody and application thereof |
| WO2020143836A1 (en) | 2019-01-11 | 2020-07-16 | 上海开拓者生物医药有限公司 | Cd73 antibody, preparation method therefor and application thereof |
| WO2020150677A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | Heterocyclic compounds as adenosine antagonists |
| WO2020150675A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | Compounds and uses thereof |
| WO2020150676A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | 1,8-naphthyridinone compounds and uses thereof |
| WO2020150674A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | Heterocyclic compounds as adenosine antagonists |
| WO2020151707A1 (en) | 2019-01-22 | 2020-07-30 | 江苏恒瑞医药股份有限公司 | Novel small molecule cd73 inhibitor, preparation method therefor and pharmaceutical use thereof |
| WO2020152132A1 (en) | 2019-01-22 | 2020-07-30 | Merck Patent Gmbh | Thiazolopyridine derivatives as adenosine receptor antagonists |
| WO2020156505A1 (en) | 2019-02-02 | 2020-08-06 | 江苏恒瑞医药股份有限公司 | 2-amionpyrimidine derivative, preparation method therefor and application thereof in medicines |
| WO2020159905A1 (en) | 2019-01-29 | 2020-08-06 | Incyte Corporation | Pyrazolopyridines and triazolopyridines as a2a / a2b inhibitors |
| CN111534585A (en) | 2020-03-23 | 2020-08-14 | 至本医疗科技(上海)有限公司 | Method for immunotherapy prognosis of non-small cell lung cancer (NSCLC) patient |
| WO2020202038A1 (en) | 2019-04-02 | 2020-10-08 | Medimmune, Llc | Anti-cd73, anti-pd-l1 antibodies and chemotherapy for treating tumors |
| WO2020205538A1 (en) | 2019-03-29 | 2020-10-08 | Eternity Bioscience Inc. | Cd73 inhibitors and therapeutic uses thereof |
| WO2020210970A1 (en) | 2019-04-16 | 2020-10-22 | Bioardis Llc | Imidazotriazine derivatives as cd73 inhibitors |
| WO2020210938A1 (en) | 2019-04-15 | 2020-10-22 | Bioardis Llc | Quinazoline derivatives as cd73 inhibitors |
| WO2020216697A1 (en) | 2019-04-23 | 2020-10-29 | Innate Pharma | Cd73 blocking antibodies |
| WO2020216152A1 (en) | 2019-04-24 | 2020-10-29 | Dongguan Hec New Drug R&D Co., Ltd. | 8-substituted aryl vinyl xanthine derivatives and uses thereof |
| WO2020221209A1 (en) | 2019-04-28 | 2020-11-05 | 上海和誉生物医药科技有限公司 | Cd73 inhibitor, preparation method therefor and application thereof |
| WO2020227156A1 (en) | 2019-05-03 | 2020-11-12 | Nektar Therapeutics | Adenosine 2 receptor antagonists |
| WO2020240467A1 (en) | 2019-05-29 | 2020-12-03 | Daiichi Sankyo Company, Limited | Dosage of an antibody-drug conjugate |
| WO2020244606A1 (en) | 2019-06-06 | 2020-12-10 | 北京加科思新药研发有限公司 | Binding molecule specific for cd73 and use of binding molecule |
| WO2020249063A1 (en) | 2019-06-13 | 2020-12-17 | Bio-Thera Solutions, Ltd. | Methods for the treatment of trop2 positive diseases |
| WO2020257648A1 (en) | 2019-06-20 | 2020-12-24 | Fred Hutchinson Cancer Research Center | Microlumenal targeting of cancer cells |
| WO2020253568A1 (en) | 2019-06-19 | 2020-12-24 | 海正生物制药有限公司 | Anti-cd73 antibody and application thereof |
| WO2020257429A1 (en) | 2019-06-20 | 2020-12-24 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2020253867A1 (en) | 2019-06-21 | 2020-12-24 | 中国人民解放军军事科学院军事医学研究院 | Small-molecule compound having a2a adenosine receptor antagonism |
| WO2020260196A1 (en) | 2019-06-24 | 2020-12-30 | Helmholtz-Zentrum Dresden-Rossendorf E.V. | Deuterated 7-(3-(4-(2-([18f]fluor)ethoxy)phenyl)propyl)-2-(furan-2-yl)-7h-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-amine derivatives |
| WO2020260857A1 (en) | 2019-06-13 | 2020-12-30 | AdoRx Therapeutics Limited | Hydroxamate compounds as antagonists of the adenosine a2a receptor |
| WO2020263058A1 (en) | 2019-06-28 | 2020-12-30 | 한미약품 주식회사 | Aminocyanopyridine derivative and use thereof |
| WO2021001873A1 (en) | 2019-07-01 | 2021-01-07 | 日本電信電話株式会社 | Path control device, path control method, program, and network system |
| CN112237634A (en) | 2019-07-19 | 2021-01-19 | 上海复旦张江生物医药股份有限公司 | Antibody drug conjugate, intermediate thereof, preparation method and application |
| WO2021011670A1 (en) | 2019-07-17 | 2021-01-21 | Teon Therapeutics, Inc. | Adenosine a2a receptor antagonists and uses thereof |
| WO2021011689A1 (en) | 2019-07-16 | 2021-01-21 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2021018172A1 (en) | 2019-07-30 | 2021-02-04 | 杭州阿诺生物医药科技有限公司 | Adenosine receptor antagonist |
| CN112321715A (en) | 2020-11-03 | 2021-02-05 | 博奥信生物技术(南京)有限公司 | anti-TROP 2 nano antibody and preparation method and application thereof |
| WO2021029450A1 (en) | 2019-08-09 | 2021-02-18 | 한국화학연구원 | Novel pyrimidine sulfonamide derivative, and pharmaceutical composition for preventing or treating cancer comprising same as active ingredient |
| WO2021032173A1 (en) | 2019-08-21 | 2021-02-25 | Harbour Biomed (Shanghai) Co., Ltd. | Anti-cd73 antibody and application thereof |
| WO2021037037A1 (en) | 2019-08-27 | 2021-03-04 | Elpiscience (Suzhou) Biopharma, Ltd. | Novel anti-cd39 antibodies |
| WO2021041360A1 (en) | 2019-08-26 | 2021-03-04 | Incyte Corporation | Triazolopyrimidines as a2a / a2b inhibitors |
| WO2021041319A1 (en) | 2019-08-29 | 2021-03-04 | Eli Lilly And Company | Crystalline forms of a cd73 inhibitor |
| WO2021040356A1 (en) | 2019-08-23 | 2021-03-04 | Kainos Medicine, Inc. | C-nucleosides, c-nucleotides and their analogs, equivalents and prodrugs thereof for ectonucleotidase inhibition |
| WO2021043229A1 (en) | 2019-09-05 | 2021-03-11 | 复旦大学 | Tumor-targeting recombinant bifunctional fusion protein and application thereof |
| WO2021044005A1 (en) | 2019-09-06 | 2021-03-11 | Symphogen A/S | Anti-cd73 antibodies |
| WO2021052402A1 (en) | 2019-09-18 | 2021-03-25 | 四川百利药业有限责任公司 | Camptothecin derivative and conjugate thereof |
| WO2021055329A1 (en) | 2019-09-16 | 2021-03-25 | Surface Oncology, Inc. | Anti-cd39 antibody compositions and methods |
| US20210093730A1 (en) | 2019-10-01 | 2021-04-01 | Immunomedics, Inc. | Biomarkers for antibody-drug conjugate monotherapy or combination therapy |
| WO2021056610A1 (en) | 2019-09-25 | 2021-04-01 | Shanghai Epimab Biotherapeutics Co., Ltd. | High affinity antibodies to cd39 and uses thereof |
| WO2021087136A1 (en) | 2019-10-30 | 2021-05-06 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2021087463A1 (en) | 2019-11-01 | 2021-05-06 | Corvus Pharmaceuticals, Inc. | Immunomodulatory anti-cd73 antibodies and uses thereof |
| WO2021088901A1 (en) | 2019-11-05 | 2021-05-14 | Bioardis Llc | Compounds as cd73 inhibitors |
| WO2021088838A1 (en) | 2019-11-05 | 2021-05-14 | 北京加科思新药研发有限公司 | Binding molecule specifically for cd39 and use thereof |
| WO2021093701A1 (en) | 2019-11-11 | 2021-05-20 | Sunshine Lake Pharma Co., Ltd. | 8-substituted styryl xanthine derivatives and uses thereof |
| WO2021097223A2 (en) | 2019-11-15 | 2021-05-20 | Genzyme Corporation | Biparatopic cd73 antibodies |
| WO2021099832A2 (en) | 2019-11-19 | 2021-05-27 | Ildong Pharmaceutical Co., Ltd. | Adenosine receptor antagonist compounds |
| WO2021105916A1 (en) | 2019-11-26 | 2021-06-03 | Aurigene Discovery Technologies Limited | Sulfonamide compounds targeting cd73 and adenosine receptors |
| WO2021113625A1 (en) | 2019-12-06 | 2021-06-10 | Plexxikon Inc. | Compounds and methods for cd73 modulation and indications therefor |
| WO2021138467A1 (en) | 2020-01-03 | 2021-07-08 | Incyte Corporation | Anti-cd73 antibodies and uses thereof |
| WO2021146631A1 (en) | 2020-01-17 | 2021-07-22 | Nuvation Bio Inc. | Heterocyclic compounds as adenosine antagonists |
| WO2021147993A1 (en) | 2020-01-22 | 2021-07-29 | 江苏恒瑞医药股份有限公司 | Anti-trop-2 antidody-exatecan analog conjugate and medical use thereof |
| WO2021156439A1 (en) | 2020-02-06 | 2021-08-12 | Astrazeneca Ab | Triazole compounds as adenosine receptor antagonists |
| WO2021179074A1 (en) | 2020-03-10 | 2021-09-16 | Marvel Biotechnology | Purine compounds for treating disorders |
| WO2021185256A1 (en) | 2020-03-16 | 2021-09-23 | 上海海雁医药科技有限公司 | Substituted pyrimidine or pyridine amine derivative, composition thereof, and medical use thereof |
| WO2021191379A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | 5-amino-8-(4-pyridyl)-[1,2,4]triazolo[4,3-c]pyrimidin-3-one compounds for use against cancer |
| WO2021191380A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | Triazolone compounds |
| WO2021194623A1 (en) | 2020-03-26 | 2021-09-30 | Inspyr Therapeutics, Inc. | 8-substituted diaryl xanthines as dual a 2a-a 2b antagonists |
| WO2021191376A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | Triazolone compounds |
| WO2021191378A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | 5-amino-8-(4-pyridyl)-[1,2,4]triazolo[4,3-c]pyrimidin-3-one compounds for use against cancer |
| WO2021205383A1 (en) | 2020-04-09 | 2021-10-14 | Aprilbio Co., Ltd. | Monoclonal antibodies and antigen binding fragments thereof for suppressing cd73 immune checkpoint and uses thereof |
| WO2021213466A1 (en) | 2020-04-22 | 2021-10-28 | 中山康方生物医药有限公司 | Anti-cd73 antibody and use thereof |
| WO2021216898A1 (en) | 2020-04-23 | 2021-10-28 | Plexxikon Inc. | Compounds and methods for cd73 modulation and indications therefor |
| WO2021213475A1 (en) | 2020-04-22 | 2021-10-28 | 中山康方生物医药有限公司 | Anti-cd73-anti-pd-1 bispecific antibody and use thereof |
| WO2021222522A1 (en) | 2020-05-01 | 2021-11-04 | Gilead Sciences, Inc. | Cd73 inhibiting 2,4-dioxopyrimidine compounds |
| WO2021224636A1 (en) | 2020-05-07 | 2021-11-11 | AdoRx Therapeutics Limited | Antagonists of the adenosine a2a receptor |
| WO2021225892A1 (en) | 2020-05-03 | 2021-11-11 | Levena (Suzhou) Biopharma Co., Ltd. | Antibody-drug conjugates (adcs) comprising an anti-trop-2 antibody, compositions comprising such adcs, as well as methods of making and using the same |
| WO2021227306A1 (en) | 2020-05-12 | 2021-11-18 | 普米斯生物技术(珠海)有限公司 | Anti-cd73 antibody and use thereof |
| WO2021241729A1 (en) | 2020-05-29 | 2021-12-02 | ブライトパス・バイオ株式会社 | Anti-cd73 antibody and use thereof |
| WO2021257643A1 (en) | 2020-06-17 | 2021-12-23 | Arcus Biosciences, Inc. | Crystalline forms of a cd73 inhibitor and uses thereof |
| WO2021259199A1 (en) | 2020-06-22 | 2021-12-30 | 信达生物制药(苏州)有限公司 | Anti-cd73 antibody and use thereof |
| WO2022007677A1 (en) | 2020-07-07 | 2022-01-13 | 贝达药业股份有限公司 | Cd73 inhibitor and application in pharmaceutical use |
| WO2022010797A2 (en) | 2020-07-07 | 2022-01-13 | Bionecure Therapeutics, Inc. | Novel maytansinoids as adc payloads and their use for the treatment of cancer |
| WO2022020552A1 (en) | 2020-07-24 | 2022-01-27 | Merck Sharp & Dohme Corp. | Adenosine a2a and a2b receptor dual antagonists for immuno-oncology |
| WO2022020550A1 (en) | 2020-07-24 | 2022-01-27 | Merck Sharp & Dohme Corp. | Adenosine a2a and a2b receptor dual antagonists for immuno-oncology |
| WO2022023772A1 (en) | 2020-07-31 | 2022-02-03 | AdoRx Therapeutics Limited | Antagonist compounds |
| WO2022052886A1 (en) | 2020-09-08 | 2022-03-17 | 贝达药业股份有限公司 | Cd73 inhibitor and application thereof in medicine |
| WO2022068929A1 (en) | 2020-09-30 | 2022-04-07 | 武汉人福创新药物研发中心有限公司 | Pyrimidinedione compound and use thereof |
| WO2022072601A1 (en) | 2020-09-30 | 2022-04-07 | Na Biotech Corp | ANTI-ADENOSINE RECEPTOR (A2aR) ANTIBODIES |
| WO2022078524A2 (en) | 2021-11-03 | 2022-04-21 | Hangzhou Dac Biotech Co., Ltd. | Specific conjugation of an antibody |
| WO2022083049A1 (en) | 2020-10-19 | 2022-04-28 | 中山康方生物医药有限公司 | Anti-cd73 antibody and use thereof |
| WO2022090711A1 (en) | 2020-10-26 | 2022-05-05 | AdoRx Therapeutics Limited | Compounds as cd73 inhibitors |
| WO2022095975A1 (en) | 2020-11-05 | 2022-05-12 | 武汉人福创新药物研发中心有限公司 | Cd73 inhibitor and use thereof |
| WO2022096020A1 (en) | 2020-11-09 | 2022-05-12 | 江苏中新医药有限公司 | Anti-ecto-5'-nucleotidase antibody sequence |
| WO2022095953A1 (en) | 2020-11-05 | 2022-05-12 | 武汉人福创新药物研发中心有限公司 | Pyridazine alkyne compounds and use thereof |
| WO2022111576A1 (en) | 2020-11-27 | 2022-06-02 | Elpiscience (Suzhou) Biopharma, Ltd. | Novel conjugate molecules targeting cd39 and tgfβeta |
| WO2022123272A1 (en) | 2020-12-11 | 2022-06-16 | AdoRx Therapeutics Limited | Antagonists of the adenosine a2a receptor |
| WO2022121914A1 (en) | 2020-12-10 | 2022-06-16 | 上海翰森生物医药科技有限公司 | Oxo-nitrogen ring derivative regulator, preparation method therefor, and application thereof |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111936169A (en) * | 2018-04-06 | 2020-11-13 | 西雅图遗传学公司 | Camptothecin Peptide Conjugate |
-
2023
- 2023-04-12 KR KR1020247037701A patent/KR20250004779A/en active Pending
- 2023-04-12 WO PCT/US2023/065682 patent/WO2023201267A1/en not_active Ceased
- 2023-04-12 AU AU2023252914A patent/AU2023252914A1/en active Pending
- 2023-04-12 JP JP2024560326A patent/JP2025512384A/en active Pending
- 2023-04-12 EP EP23721581.9A patent/EP4507736A1/en active Pending
- 2023-04-12 CN CN202380033738.5A patent/CN119173276A/en active Pending
- 2023-04-12 CA CA3256033A patent/CA3256033A1/en active Pending
- 2023-04-13 TW TW112113867A patent/TW202345845A/en unknown
Patent Citations (229)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1996011013A1 (en) | 1994-10-05 | 1996-04-18 | Immunomedics, Inc. | Cdr-grafted type iii anti-cea humanized mouse monoclonal antibodies |
| US6624821B1 (en) | 1999-02-05 | 2003-09-23 | Samsung Electronics Co., Ltd. | Image texture retrieving method and apparatus thereof |
| WO2003074566A2 (en) | 2002-03-01 | 2003-09-12 | Immunomedics, Inc. | Rs7 antibodies |
| US8435529B2 (en) | 2002-06-14 | 2013-05-07 | Immunomedics, Inc. | Combining radioimmunotherapy and antibody-drug conjugates for improved cancer therapy |
| US20060193865A1 (en) | 2002-12-13 | 2006-08-31 | Immunomedics, Inc. | Camptothecin-binding moiety conjugates |
| US7999083B2 (en) | 2002-12-13 | 2011-08-16 | Immunomedics, Inc. | Immunoconjugates with an intracellularly-cleavable linkage |
| US8435539B2 (en) | 2004-02-13 | 2013-05-07 | Immunomedics, Inc. | Delivery system for cytotoxic drugs by bispecific antibody pretargeting |
| WO2006094192A2 (en) | 2005-03-03 | 2006-09-08 | Immunomedics, Inc. | Humanized l243 antibodies |
| US20130039861A1 (en) | 2005-04-06 | 2013-02-14 | Immunomedics, Inc. | Dye Conjugated Peptides for Fluorescent Imaging |
| WO2007095749A1 (en) | 2006-02-24 | 2007-08-30 | Arius Research Inc. | Cytotoxicity mediation of cells evidencing surface expression of trop-2 |
| WO2007103776A2 (en) | 2006-03-02 | 2007-09-13 | Cv Therapeutics, Inc. | A2a adenosine receptor antagonists |
| WO2007134958A1 (en) | 2006-05-18 | 2007-11-29 | F. Hoffmann-La Roche Ag | Thiazolo-pyramidine / pyridine urea derivatives as adenosine a2b receptor antagonists |
| WO2008086201A1 (en) | 2007-01-05 | 2008-07-17 | Biogen Idec Ma Inc. | Polymorphs and solvates of a pharmaceutical and methods of making |
| WO2009009178A2 (en) | 2007-04-06 | 2009-01-15 | The General Hospital Corporation | Systems and methods for optical imaging using early arriving photons |
| WO2009033161A1 (en) | 2007-09-07 | 2009-03-12 | The John Hopkins University | Adenosine receptor agonists and antagonists to modulate t cell responses |
| WO2009037468A1 (en) | 2007-09-21 | 2009-03-26 | Vernalis (R & D) Ltd. | Thienopyrimidine compounds and compositions |
| WO2009037463A1 (en) | 2007-09-21 | 2009-03-26 | Vernalis (R & D) Ltd. | Thienopyrimidine compounds |
| WO2009037467A1 (en) | 2007-09-21 | 2009-03-26 | Vernalis (R & D) Ltd. | Pyrrolopyrimidine compounds |
| WO2009055308A1 (en) | 2007-10-24 | 2009-04-30 | Janssen Pharmaceutica, N.V. | Arylindenopyrimidines and their use as adenosine a2a receptor antagonists |
| WO2009055548A1 (en) | 2007-10-25 | 2009-04-30 | Janssen Pharmaceutica, N.V. | Arylindenopyrimidines and their use as adenosine a2a receptor antagonists |
| WO2009095478A1 (en) | 2008-01-31 | 2009-08-06 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Antibodies against human cd39 and use thereof for inhibiting t regulatory cells activity |
| WO2010008775A1 (en) | 2008-06-23 | 2010-01-21 | Ligand Pharmaceuticals Inc. | Aminopyridopyrazinone derivatives for treating neurodegenerative diseases |
| WO2010093395A1 (en) | 2009-02-13 | 2010-08-19 | Immunomedics, Inc. | Immunoconjugates with an intracellularly-cleavable linkage |
| WO2011027805A1 (en) | 2009-09-02 | 2011-03-10 | 協和発酵キリン株式会社 | Therapeutic agent for mood disorders |
| WO2011050160A1 (en) | 2009-10-22 | 2011-04-28 | Biogen Idec Ma Inc. | Pharmaceuticals, compositions and methods of making and using the same |
| WO2011055391A1 (en) | 2009-11-09 | 2011-05-12 | Advinus Therapeutics Private Limited | Substituted fused pyrimidine compounds, its preparation and uses thereof |
| WO2011068845A1 (en) | 2009-12-02 | 2011-06-09 | Immunomedics, Inc. | Combining radioimmunotherapy and antibody-drug conjugates for improved cancer therapy |
| WO2012085132A1 (en) | 2010-12-22 | 2012-06-28 | Orega Biotech | Antibodies against human cd39 and use thereof |
| WO2012135084A1 (en) | 2011-03-31 | 2012-10-04 | Merck Sharp & Dohme Corp. | METABOLITES OF 2-(FURAN-2-YL)-7-(2-(4-(4-(2-METHOXYETHOXY)PHENYL)PIPERAZIN-1-YL)ETHYL)-7H-PYRAZOLO[4,3-e][1,2,4]TRIAZOLO[1,5-c]PYRIMIDIN-5-AMINE AND THEIR UTILITY AS ADENOSINE A2a RECEPTOR ANTAGONISTS |
| WO2013068946A2 (en) | 2011-11-11 | 2013-05-16 | Rinat Neuroscience Corp. | Antibodies specific for trop-2 and their uses |
| US9427464B2 (en) | 2011-11-22 | 2016-08-30 | Chiome Bioscience Inc. | Anti-human TROP-2 antibody having an antitumor activity in vivo |
| WO2013077458A1 (en) | 2011-11-22 | 2013-05-30 | 株式会社リブテック | Anti-human trop-2 antibody exhibiting antitumor activity in vivo |
| US20170021017A1 (en) | 2012-08-14 | 2017-01-26 | Ibc Pharmaceuticals, Inc. | Combination therapy for inducing immune response to disease |
| WO2014092804A1 (en) | 2012-12-13 | 2014-06-19 | Immunomedics, Inc. | Dosages of immunoconjugates of antibodies and sn-38 for improved efficacy and decreased toxicity |
| US20180110772A1 (en) | 2012-12-13 | 2018-04-26 | Immunomedics, Inc. | Therapy for metastatic urothelial cancer with the antibody-drug conjugate, sacituzumab govitecan (immu-132) |
| US20180185351A1 (en) | 2012-12-13 | 2018-07-05 | Immunomedics, Inc. | Therapy of small-cell lung cancer (sclc) with a topoisomerase-i inhibiting antibody-drug conjugate (adc) targeting trop-2 |
| US20210069343A1 (en) | 2012-12-13 | 2021-03-11 | Immunomedics, Inc. | Antibody-drug conjugates and uses thereof |
| US9492566B2 (en) | 2012-12-13 | 2016-11-15 | Immunomedics, Inc. | Antibody-drug conjugates and uses thereof |
| WO2014101113A1 (en) | 2012-12-28 | 2014-07-03 | Merck Sharp & Dohme Corp. | Piperazine-substituted 7-methoxy-[1,2,4]triazolo[1,5-c]quinazolin-5-amine compounds with a2a antagonist properties |
| WO2014101120A1 (en) | 2012-12-28 | 2014-07-03 | Merck Sharp & Dohme Corp. | Heterobicyclo-substituted-7-methoxy-[1,2,4]triazolo[1,5-c]quinazolin-5-amine compounds with a2a antagonist properties |
| WO2014106861A1 (en) | 2013-01-03 | 2014-07-10 | Council Of Scientific & Industrial Research | (4e)-4-(4-substituted benzylideneamino)-2,3-dihydro-3- substituted-2-thioxothiazole-5-carbonitriles as a2ar antagonist and process for preparation thereof |
| WO2014163684A1 (en) | 2013-04-03 | 2014-10-09 | Ibc Pharmaceuticals, Inc. | Combination therapy for inducing immune response to disease |
| US20160296633A1 (en) | 2013-07-23 | 2016-10-13 | Immunomedics, Inc. | Combining anti-hla-dr or anti-trop-2 antibodies with microtubule inhibitors, parp inhibitors, bruton kinase inhibitors or phosphoinositide 3-kinase inhibitors significantly improves therapeutic outcome in cancer |
| US20170274093A1 (en) | 2013-07-23 | 2017-09-28 | Immunomedics, Inc. | Combining anti-hla-dr or anti-trop-2 antibodies with microtubule inhibitors, parp inhibitors, bruton kinase inhibitors or phosphoinositide 3-kinase inhibitors significantly improves therapeutic outcome in cancer |
| WO2015027431A1 (en) | 2013-08-29 | 2015-03-05 | Merck Sharp & Dohme Corp. | 2,2-difluorodioxolo a2a receptor antagonists |
| US9850312B2 (en) | 2013-12-25 | 2017-12-26 | Daiichi Sankyo Company, Limited | Anti-TROP2 antibody-drug conjugate |
| US20160297890A1 (en) | 2013-12-25 | 2016-10-13 | Daiichi Sankyo Company, Limited | Anti-trop2 antibody-drug conjugate |
| WO2015098099A1 (en) | 2013-12-25 | 2015-07-02 | 第一三共株式会社 | Anti-trop2 antibody-drug conjugate |
| WO2015164573A1 (en) | 2014-04-25 | 2015-10-29 | Vitae Pharmaceuticals, Inc. | Purine derivatives as cd73 inhibitors for the treatment of cancer |
| WO2016055609A1 (en) | 2014-10-10 | 2016-04-14 | Innate Pharma | Cd73 blockade |
| WO2016073845A1 (en) | 2014-11-07 | 2016-05-12 | Igenica Biotherapeutics, Inc. | Anti-cd39 antibodies and uses thereof |
| WO2016075099A1 (en) | 2014-11-10 | 2016-05-19 | Medimmune Limited | Binding molecules specific for cd73 and uses thereof |
| WO2016081290A1 (en) | 2014-11-18 | 2016-05-26 | Merck Sharp & Dohme Corp. | Aminopyrazine compounds with a2a antagonist properties |
| WO2016081746A2 (en) | 2014-11-21 | 2016-05-26 | Bristol-Myers Squibb Company | Antibodies comprising modified heavy constant regions |
| WO2016081748A2 (en) | 2014-11-21 | 2016-05-26 | Bristol-Myers Squibb Company | Antibodies against cd73 and uses thereof |
| US10501555B2 (en) | 2014-12-04 | 2019-12-10 | Abruzzo Theranostic S.R.L. | Humanized anti-trop-2 monoclonal antibodies and uses thereof |
| WO2016126570A1 (en) | 2015-02-06 | 2016-08-11 | Merck Sharp & Dohme Corp. | Aminoquinazoline compounds as a2a antagonist |
| WO2016200717A1 (en) | 2015-06-11 | 2016-12-15 | Merck Sharp & Dohme Corp. | Aminopyrazine compounds with a2a antagonist properties |
| WO2016201300A1 (en) | 2015-06-12 | 2016-12-15 | Immunomedics, Inc. | Disease therapy with chimeric antigen receptor (car) constructs and t cells (car-t) or nk cells (car-nk) expressing car constructs |
| US20170209594A1 (en) | 2015-06-25 | 2017-07-27 | Immunomedics, Inc. | Synergistic effect of anti-trop-2 antibody-drug conjugate in combination therapy for triple-negative breast cancer when used with microtubule inhibitors or parp inhibitors |
| WO2016209787A1 (en) | 2015-06-26 | 2016-12-29 | Merck Sharp & Dohme Corp. | Sustained release formulation and tablets prepared therefrom |
| WO2017008205A1 (en) | 2015-07-10 | 2017-01-19 | Merck Sharp & Dohme Corp. | Substituted aminoquinazoline compounds as a2a antagonist |
| WO2017025051A1 (en) | 2015-08-11 | 2017-02-16 | Wuxi Biologics (Shanghai) Co. Ltd. | Novel anti-pd-1 antibodies |
| WO2017064043A1 (en) | 2015-10-12 | 2017-04-20 | Innate Pharma | Cd73 blocking agents |
| WO2017098421A1 (en) | 2015-12-08 | 2017-06-15 | Glaxosmithkline Intellectual Property Development Limited | Benzothiadiazine compounds |
| WO2017100670A1 (en) | 2015-12-09 | 2017-06-15 | Corvus Pharmaceuticals, Inc. | Humanized anti-cd73 antibodies |
| WO2017120508A1 (en) | 2016-01-08 | 2017-07-13 | Arcus Biosciences, Inc. | Modulators of 5'-nucleotidase, ecto and the use thereof |
| US11001603B2 (en) | 2016-01-08 | 2021-05-11 | Arcus Biosciences, Inc. | Modulators of 5′-nucleotidase, ecto and the use thereof |
| WO2017118613A1 (en) | 2016-01-08 | 2017-07-13 | Syddansk Universitet | Bispecific antibodies targeting human cd73 |
| US20190248917A1 (en) | 2016-02-10 | 2019-08-15 | Immunomedics, Inc. | Combination of abcg2 inhibitors with sacituzumab govitecan (immu-132) overcomes resistance to sn-38 in trop-2 expressing cancers |
| WO2017153952A1 (en) | 2016-03-10 | 2017-09-14 | Glaxosmithkline Intellectual Property Development Limited | 5-sulfamoyl-2-hydroxybenzamide derivatives |
| WO2017157948A1 (en) | 2016-03-14 | 2017-09-21 | Innate Pharma | Anti-cd39 antibodies |
| WO2018013611A1 (en) | 2016-07-11 | 2018-01-18 | Corvus Pharmaceuticals, Inc. | Anti-cd73 antibodies |
| WO2018013951A1 (en) | 2016-07-15 | 2018-01-18 | Northwestern University | Targeting adenosine a2a receptors for the treatment of levodopa-induced dyskinesias |
| WO2018036428A1 (en) | 2016-08-22 | 2018-03-01 | 广东纳路纳米科技有限公司 | Metal nanowire-oxidation-resistant material combined transparent conductive film and preparation therefor |
| WO2018049145A1 (en) | 2016-09-09 | 2018-03-15 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2018067424A1 (en) | 2016-10-03 | 2018-04-12 | Arcus Biosciences, Inc. | Inhibitors of adenosine 5'-nucleotidase |
| WO2018065552A1 (en) | 2016-10-06 | 2018-04-12 | Innate Pharma | Anti-cd39 antibodies |
| WO2018065627A1 (en) | 2016-10-07 | 2018-04-12 | Secarna Pharmaceuticals Gmbh & Co Kg | Immunosuppression-reverting oligonucleotides inhibiting the expression of cd73 |
| WO2018065622A1 (en) | 2016-10-07 | 2018-04-12 | Secarna Pharmaceuticals Gmbh & Co Kg | Immunosuppression-reverting oligonucleotides inhibiting the expression of cd39 |
| WO2018094148A1 (en) | 2016-11-18 | 2018-05-24 | Arcus Biosciences, Inc. | Inhibitors of cd73-mediated immunosuppression |
| WO2018110555A1 (en) | 2016-12-13 | 2018-06-21 | アステラス製薬株式会社 | Anti-human cd73 antibody |
| WO2018119284A1 (en) | 2016-12-22 | 2018-06-28 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2018136700A1 (en) | 2017-01-20 | 2018-07-26 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| US10399962B2 (en) | 2017-01-20 | 2019-09-03 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| WO2018137598A1 (en) | 2017-01-24 | 2018-08-02 | I-Mab | Anti-cd73 antibodies and uses thereof |
| US20180271992A1 (en) | 2017-03-27 | 2018-09-27 | Immunomedics, Inc. | Treatment of high trop-2 expressing triple negative breast cancer (tnbc) with sacituzumab govitecan (immu-132) overcomes homologous recombination repair (hrr) rescue mediated by rad51 |
| WO2018183635A1 (en) | 2017-03-31 | 2018-10-04 | Peloton Therapeutics, Inc. | Cd73 inhibitors and uses thereof |
| WO2018195321A1 (en) | 2017-04-20 | 2018-10-25 | Gilead Sciences, Inc. | Pd-1/pd-l1 inhibitors |
| WO2018208727A1 (en) | 2017-05-08 | 2018-11-15 | Eternity Bioscience Inc. | Nucleoside and nucleotide analogues as cd73 inhibitors and therapeutic uses thereof |
| WO2018208980A1 (en) | 2017-05-10 | 2018-11-15 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2018215535A1 (en) | 2017-05-23 | 2018-11-29 | Helmholtz Zentrum München - Deutsches Forschungszentrum für Gesundheit und Umwelt (GmbH) | Novel cd73 antibody, preparation and uses thereof |
| WO2018217227A1 (en) | 2017-05-24 | 2018-11-29 | Immunomedics, Inc. | Novel anti-pd-1 checkpoint inhibitor antibodies that block binding of pd-l1 to pd-1 |
| WO2018237157A1 (en) | 2017-06-22 | 2018-12-27 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| WO2018237173A1 (en) | 2017-06-22 | 2018-12-27 | Novartis Ag | ANTIBODY MOLECULES DIRECTED AGAINST CD73 AND CORRESPONDING USES |
| WO2019027935A1 (en) | 2017-07-31 | 2019-02-07 | Tizona Therapeutics | Anti-cd39 antibodies, compositions comprising anti-cd39 antibodies and methods of using anti-cd39 antibodies |
| US20190048095A1 (en) | 2017-08-11 | 2019-02-14 | Bio-Thera Solutions, Ltd. | Compounds and methods for the treatment of trop2 positive diseases |
| WO2019053617A1 (en) | 2017-09-12 | 2019-03-21 | Glaxosmithkline Intellectual Property Development Limited | Chemical compounds |
| WO2019086074A1 (en) | 2017-10-31 | 2019-05-09 | Helmholtz-Zentrum Dresden-Rossendorf E.V. | 4-(furan-2-yl)-1h-pyrazolo[3,4-d]pyrimidin-6-amine derivative and use thereof |
| WO2019090111A1 (en) | 2017-11-03 | 2019-05-09 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2019118313A1 (en) | 2017-12-13 | 2019-06-20 | Merck Sharp & Dohme Corp. | Imidazo [1,2-c] quinazolin-5-amine compounds with a2a antagonist properties |
| WO2019114666A1 (en) | 2017-12-15 | 2019-06-20 | 四川科伦博泰生物医药股份有限公司 | Bioactive conjugate, preparation method therefor and use thereof |
| US20210101906A2 (en) | 2017-12-15 | 2021-04-08 | Sichuan Kelun-Biotech Biopharmaceutical Co., Ltd. | Bioactive conjugate, preparation method therefor and use thereof |
| WO2019129059A1 (en) | 2017-12-29 | 2019-07-04 | 上海和誉生物医药科技有限公司 | Phosphonic acid derivative having cd73 inhibitory activity, and preparation method and use thereof |
| WO2019160882A1 (en) | 2018-02-13 | 2019-08-22 | Gilead Sciences, Inc. | Pd-1/pd-l1 inhibitors |
| WO2019168744A1 (en) | 2018-03-01 | 2019-09-06 | Eli Lilly And Company | Cd73 inhibitors |
| WO2019166701A1 (en) | 2018-03-02 | 2019-09-06 | Turun Yliopisto | Anti-adenosine signaling pathway antibodies conjugated or fused with adenosine deaminase or capable of binding adenosine deaminase |
| WO2019170131A1 (en) | 2018-03-07 | 2019-09-12 | 复旦大学 | Targeted cd73 antibody and antibody-drug conjugate, and preparation method therefor and uses thereof |
| WO2019173291A1 (en) | 2018-03-09 | 2019-09-12 | Phanes Therapeutics, Inc. | Anti-cd73 antibodies and uses thereof |
| WO2019173692A2 (en) | 2018-03-09 | 2019-09-12 | Agenus Inc. | Anti-cd73 antibodies and methods of use thereof |
| WO2019178269A2 (en) | 2018-03-14 | 2019-09-19 | Surface Oncology, Inc. | Antibodies that bind cd39 and uses thereof |
| CN108440674A (en) | 2018-04-28 | 2018-08-24 | 杭州荣泽生物科技有限公司 | A kind of Trop-2 specific chimerics antigen receptor cell is prepared and application thereof |
| WO2019224025A2 (en) | 2018-05-19 | 2019-11-28 | Boehringer Ingelheim International Gmbh | Antagonizing cd73 antibody |
| WO2019232244A2 (en) | 2018-05-31 | 2019-12-05 | Novartis Ag | Antibody molecules to cd73 and uses thereof |
| WO2019246403A1 (en) | 2018-06-21 | 2019-12-26 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2020016662A2 (en) | 2018-07-09 | 2020-01-23 | Abmart Inc. | Antibodies specific to trophoblast antigen 2 (trop2) |
| WO2020014643A1 (en) | 2018-07-13 | 2020-01-16 | Gilead Sciences, Inc. | Pd-1/pd-l1 inhibitors |
| WO2020046813A1 (en) | 2018-08-27 | 2020-03-05 | Arcus Biosciences, Inc. | Cd73 inhibitors |
| WO2020047082A1 (en) | 2018-08-28 | 2020-03-05 | Eternity Bioscience Inc. | Cd73 inhibitors and therapeutic uses thereof |
| WO2020051686A1 (en) | 2018-09-11 | 2020-03-19 | Risen (Suzhou) Pharma Tech Co. Ltd. | Cd73 inhibitors and pharmaceutical uses thereof |
| WO2020053263A1 (en) | 2018-09-11 | 2020-03-19 | Iteos Therapeutics S.A. | Thiocarbamate derivatives as a2a inhibitors, pharmaceutical composition thereof and combinations with anticancer agents |
| WO2020097127A2 (en) | 2018-11-05 | 2020-05-14 | Corvus Pharmaceuticals, Inc. | B-cell activating cd73 antibodies |
| WO2020094670A1 (en) | 2018-11-05 | 2020-05-14 | Synaffix B.V. | Antibody-conjugates for targeting of tumours expressing trop-2 |
| WO2020098599A1 (en) | 2018-11-12 | 2020-05-22 | 江苏恒瑞医药股份有限公司 | Anti-cd73 antibody, antigen-binding fragment thereof and application thereof |
| WO2020106560A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020106558A1 (en) | 2018-11-20 | 2020-05-28 | Merck Sharp & Dohme Corp. | Substituted amino triazolopyrimidine and amino triazolopyrazine adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020103930A1 (en) | 2018-11-22 | 2020-05-28 | 上海科技大学 | Thiazolo ring compound, preparation method therefor, intermediate thereof, and application thereof |
| WO2020103939A1 (en) | 2018-11-22 | 2020-05-28 | 上海科技大学 | Triazolo cycle compound, preparation method therefor, intermediate thereof and application thereof |
| WO2020112700A1 (en) | 2018-11-30 | 2020-06-04 | Merck Sharp & Dohme Corp. | 9-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use |
| WO2020112706A1 (en) | 2018-11-30 | 2020-06-04 | Merck Sharp & Dohme Corp. | 7-, 8-, and 10-SUBSTITUTED AMINO TRIAZOLO QUINAZOLINE DERIVATIVES AS ADENOSINE RECEPTOR ANTAGONISTS, PHARMACEUTICAL COMPOSITIONS AND THEIR USE |
| WO2020130125A1 (en) | 2018-12-21 | 2020-06-25 | 第一三共株式会社 | Combination of antibody-drug conjugate and kinase inhibitor |
| WO2020139803A1 (en) | 2018-12-24 | 2020-07-02 | Dcb-Usa Llc | Benzothiadiazine derivatives and compositions comprising the same for treating disorders mediated by adenosine |
| WO2020135210A1 (en) | 2018-12-28 | 2020-07-02 | 四川科伦博泰生物医药股份有限公司 | Substituted aryl compound and preparation method therefor and use thereof |
| WO2020135195A1 (en) | 2018-12-28 | 2020-07-02 | 四川科伦博泰生物医药股份有限公司 | Aminopyridine compound, preparation method therefor and use thereof |
| WO2020143836A1 (en) | 2019-01-11 | 2020-07-16 | 上海开拓者生物医药有限公司 | Cd73 antibody, preparation method therefor and application thereof |
| WO2020143710A1 (en) | 2019-01-11 | 2020-07-16 | 康诺亚生物医药科技(成都)有限公司 | Anti-cd73 monoclonal antibody and application thereof |
| WO2020150677A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | Heterocyclic compounds as adenosine antagonists |
| WO2020150675A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | Compounds and uses thereof |
| WO2020150676A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | 1,8-naphthyridinone compounds and uses thereof |
| WO2020150674A1 (en) | 2019-01-18 | 2020-07-23 | Nuvation Bio Inc. | Heterocyclic compounds as adenosine antagonists |
| WO2020151707A1 (en) | 2019-01-22 | 2020-07-30 | 江苏恒瑞医药股份有限公司 | Novel small molecule cd73 inhibitor, preparation method therefor and pharmaceutical use thereof |
| WO2020152132A1 (en) | 2019-01-22 | 2020-07-30 | Merck Patent Gmbh | Thiazolopyridine derivatives as adenosine receptor antagonists |
| WO2020159905A1 (en) | 2019-01-29 | 2020-08-06 | Incyte Corporation | Pyrazolopyridines and triazolopyridines as a2a / a2b inhibitors |
| WO2020156505A1 (en) | 2019-02-02 | 2020-08-06 | 江苏恒瑞医药股份有限公司 | 2-amionpyrimidine derivative, preparation method therefor and application thereof in medicines |
| WO2020205538A1 (en) | 2019-03-29 | 2020-10-08 | Eternity Bioscience Inc. | Cd73 inhibitors and therapeutic uses thereof |
| WO2020202038A1 (en) | 2019-04-02 | 2020-10-08 | Medimmune, Llc | Anti-cd73, anti-pd-l1 antibodies and chemotherapy for treating tumors |
| WO2020210938A1 (en) | 2019-04-15 | 2020-10-22 | Bioardis Llc | Quinazoline derivatives as cd73 inhibitors |
| WO2020210970A1 (en) | 2019-04-16 | 2020-10-22 | Bioardis Llc | Imidazotriazine derivatives as cd73 inhibitors |
| WO2020216697A1 (en) | 2019-04-23 | 2020-10-29 | Innate Pharma | Cd73 blocking antibodies |
| WO2020216152A1 (en) | 2019-04-24 | 2020-10-29 | Dongguan Hec New Drug R&D Co., Ltd. | 8-substituted aryl vinyl xanthine derivatives and uses thereof |
| WO2020221209A1 (en) | 2019-04-28 | 2020-11-05 | 上海和誉生物医药科技有限公司 | Cd73 inhibitor, preparation method therefor and application thereof |
| WO2020227156A1 (en) | 2019-05-03 | 2020-11-12 | Nektar Therapeutics | Adenosine 2 receptor antagonists |
| WO2020240467A1 (en) | 2019-05-29 | 2020-12-03 | Daiichi Sankyo Company, Limited | Dosage of an antibody-drug conjugate |
| WO2020244606A1 (en) | 2019-06-06 | 2020-12-10 | 北京加科思新药研发有限公司 | Binding molecule specific for cd73 and use of binding molecule |
| WO2020260857A1 (en) | 2019-06-13 | 2020-12-30 | AdoRx Therapeutics Limited | Hydroxamate compounds as antagonists of the adenosine a2a receptor |
| WO2020249063A1 (en) | 2019-06-13 | 2020-12-17 | Bio-Thera Solutions, Ltd. | Methods for the treatment of trop2 positive diseases |
| WO2020253568A1 (en) | 2019-06-19 | 2020-12-24 | 海正生物制药有限公司 | Anti-cd73 antibody and application thereof |
| WO2020257429A1 (en) | 2019-06-20 | 2020-12-24 | Calithera Biosciences, Inc. | Ectonucleotidase inhibitors and methods of use thereof |
| WO2020257648A1 (en) | 2019-06-20 | 2020-12-24 | Fred Hutchinson Cancer Research Center | Microlumenal targeting of cancer cells |
| WO2020253867A1 (en) | 2019-06-21 | 2020-12-24 | 中国人民解放军军事科学院军事医学研究院 | Small-molecule compound having a2a adenosine receptor antagonism |
| WO2020260196A1 (en) | 2019-06-24 | 2020-12-30 | Helmholtz-Zentrum Dresden-Rossendorf E.V. | Deuterated 7-(3-(4-(2-([18f]fluor)ethoxy)phenyl)propyl)-2-(furan-2-yl)-7h-pyrazolo[4,3-e][1,2,4]triazolo[1,5-c]pyrimidin-5-amine derivatives |
| WO2020263058A1 (en) | 2019-06-28 | 2020-12-30 | 한미약품 주식회사 | Aminocyanopyridine derivative and use thereof |
| WO2021001873A1 (en) | 2019-07-01 | 2021-01-07 | 日本電信電話株式会社 | Path control device, path control method, program, and network system |
| WO2021011689A1 (en) | 2019-07-16 | 2021-01-21 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2021011670A1 (en) | 2019-07-17 | 2021-01-21 | Teon Therapeutics, Inc. | Adenosine a2a receptor antagonists and uses thereof |
| CN112237634A (en) | 2019-07-19 | 2021-01-19 | 上海复旦张江生物医药股份有限公司 | Antibody drug conjugate, intermediate thereof, preparation method and application |
| WO2021018172A1 (en) | 2019-07-30 | 2021-02-04 | 杭州阿诺生物医药科技有限公司 | Adenosine receptor antagonist |
| WO2021029450A1 (en) | 2019-08-09 | 2021-02-18 | 한국화학연구원 | Novel pyrimidine sulfonamide derivative, and pharmaceutical composition for preventing or treating cancer comprising same as active ingredient |
| WO2021032173A1 (en) | 2019-08-21 | 2021-02-25 | Harbour Biomed (Shanghai) Co., Ltd. | Anti-cd73 antibody and application thereof |
| WO2021040356A1 (en) | 2019-08-23 | 2021-03-04 | Kainos Medicine, Inc. | C-nucleosides, c-nucleotides and their analogs, equivalents and prodrugs thereof for ectonucleotidase inhibition |
| WO2021041360A1 (en) | 2019-08-26 | 2021-03-04 | Incyte Corporation | Triazolopyrimidines as a2a / a2b inhibitors |
| WO2021037037A1 (en) | 2019-08-27 | 2021-03-04 | Elpiscience (Suzhou) Biopharma, Ltd. | Novel anti-cd39 antibodies |
| WO2021041319A1 (en) | 2019-08-29 | 2021-03-04 | Eli Lilly And Company | Crystalline forms of a cd73 inhibitor |
| WO2021043229A1 (en) | 2019-09-05 | 2021-03-11 | 复旦大学 | Tumor-targeting recombinant bifunctional fusion protein and application thereof |
| WO2021044005A1 (en) | 2019-09-06 | 2021-03-11 | Symphogen A/S | Anti-cd73 antibodies |
| WO2021055329A1 (en) | 2019-09-16 | 2021-03-25 | Surface Oncology, Inc. | Anti-cd39 antibody compositions and methods |
| WO2021052402A1 (en) | 2019-09-18 | 2021-03-25 | 四川百利药业有限责任公司 | Camptothecin derivative and conjugate thereof |
| WO2021056610A1 (en) | 2019-09-25 | 2021-04-01 | Shanghai Epimab Biotherapeutics Co., Ltd. | High affinity antibodies to cd39 and uses thereof |
| US20210093730A1 (en) | 2019-10-01 | 2021-04-01 | Immunomedics, Inc. | Biomarkers for antibody-drug conjugate monotherapy or combination therapy |
| WO2021087136A1 (en) | 2019-10-30 | 2021-05-06 | Oric Pharmaceuticals, Inc. | Cd73 inhibitors |
| WO2021087463A1 (en) | 2019-11-01 | 2021-05-06 | Corvus Pharmaceuticals, Inc. | Immunomodulatory anti-cd73 antibodies and uses thereof |
| WO2021088901A1 (en) | 2019-11-05 | 2021-05-14 | Bioardis Llc | Compounds as cd73 inhibitors |
| WO2021088838A1 (en) | 2019-11-05 | 2021-05-14 | 北京加科思新药研发有限公司 | Binding molecule specifically for cd39 and use thereof |
| WO2021093701A1 (en) | 2019-11-11 | 2021-05-20 | Sunshine Lake Pharma Co., Ltd. | 8-substituted styryl xanthine derivatives and uses thereof |
| WO2021097223A2 (en) | 2019-11-15 | 2021-05-20 | Genzyme Corporation | Biparatopic cd73 antibodies |
| WO2021099832A2 (en) | 2019-11-19 | 2021-05-27 | Ildong Pharmaceutical Co., Ltd. | Adenosine receptor antagonist compounds |
| WO2021105916A1 (en) | 2019-11-26 | 2021-06-03 | Aurigene Discovery Technologies Limited | Sulfonamide compounds targeting cd73 and adenosine receptors |
| WO2021113625A1 (en) | 2019-12-06 | 2021-06-10 | Plexxikon Inc. | Compounds and methods for cd73 modulation and indications therefor |
| WO2021138467A1 (en) | 2020-01-03 | 2021-07-08 | Incyte Corporation | Anti-cd73 antibodies and uses thereof |
| WO2021146631A1 (en) | 2020-01-17 | 2021-07-22 | Nuvation Bio Inc. | Heterocyclic compounds as adenosine antagonists |
| WO2021147993A1 (en) | 2020-01-22 | 2021-07-29 | 江苏恒瑞医药股份有限公司 | Anti-trop-2 antidody-exatecan analog conjugate and medical use thereof |
| WO2021156439A1 (en) | 2020-02-06 | 2021-08-12 | Astrazeneca Ab | Triazole compounds as adenosine receptor antagonists |
| WO2021179074A1 (en) | 2020-03-10 | 2021-09-16 | Marvel Biotechnology | Purine compounds for treating disorders |
| WO2021185256A1 (en) | 2020-03-16 | 2021-09-23 | 上海海雁医药科技有限公司 | Substituted pyrimidine or pyridine amine derivative, composition thereof, and medical use thereof |
| CN111534585A (en) | 2020-03-23 | 2020-08-14 | 至本医疗科技(上海)有限公司 | Method for immunotherapy prognosis of non-small cell lung cancer (NSCLC) patient |
| WO2021194623A1 (en) | 2020-03-26 | 2021-09-30 | Inspyr Therapeutics, Inc. | 8-substituted diaryl xanthines as dual a 2a-a 2b antagonists |
| WO2021191380A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | Triazolone compounds |
| WO2021191379A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | 5-amino-8-(4-pyridyl)-[1,2,4]triazolo[4,3-c]pyrimidin-3-one compounds for use against cancer |
| WO2021191376A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | Triazolone compounds |
| WO2021191378A1 (en) | 2020-03-26 | 2021-09-30 | Astrazeneca Ab | 5-amino-8-(4-pyridyl)-[1,2,4]triazolo[4,3-c]pyrimidin-3-one compounds for use against cancer |
| WO2021205383A1 (en) | 2020-04-09 | 2021-10-14 | Aprilbio Co., Ltd. | Monoclonal antibodies and antigen binding fragments thereof for suppressing cd73 immune checkpoint and uses thereof |
| WO2021213466A1 (en) | 2020-04-22 | 2021-10-28 | 中山康方生物医药有限公司 | Anti-cd73 antibody and use thereof |
| WO2021213475A1 (en) | 2020-04-22 | 2021-10-28 | 中山康方生物医药有限公司 | Anti-cd73-anti-pd-1 bispecific antibody and use thereof |
| WO2021216898A1 (en) | 2020-04-23 | 2021-10-28 | Plexxikon Inc. | Compounds and methods for cd73 modulation and indications therefor |
| WO2021222522A1 (en) | 2020-05-01 | 2021-11-04 | Gilead Sciences, Inc. | Cd73 inhibiting 2,4-dioxopyrimidine compounds |
| WO2021225892A1 (en) | 2020-05-03 | 2021-11-11 | Levena (Suzhou) Biopharma Co., Ltd. | Antibody-drug conjugates (adcs) comprising an anti-trop-2 antibody, compositions comprising such adcs, as well as methods of making and using the same |
| WO2021224636A1 (en) | 2020-05-07 | 2021-11-11 | AdoRx Therapeutics Limited | Antagonists of the adenosine a2a receptor |
| WO2021227306A1 (en) | 2020-05-12 | 2021-11-18 | 普米斯生物技术(珠海)有限公司 | Anti-cd73 antibody and use thereof |
| WO2021241729A1 (en) | 2020-05-29 | 2021-12-02 | ブライトパス・バイオ株式会社 | Anti-cd73 antibody and use thereof |
| WO2021257643A1 (en) | 2020-06-17 | 2021-12-23 | Arcus Biosciences, Inc. | Crystalline forms of a cd73 inhibitor and uses thereof |
| WO2021259199A1 (en) | 2020-06-22 | 2021-12-30 | 信达生物制药(苏州)有限公司 | Anti-cd73 antibody and use thereof |
| WO2022007677A1 (en) | 2020-07-07 | 2022-01-13 | 贝达药业股份有限公司 | Cd73 inhibitor and application in pharmaceutical use |
| WO2022010797A2 (en) | 2020-07-07 | 2022-01-13 | Bionecure Therapeutics, Inc. | Novel maytansinoids as adc payloads and their use for the treatment of cancer |
| WO2022020552A1 (en) | 2020-07-24 | 2022-01-27 | Merck Sharp & Dohme Corp. | Adenosine a2a and a2b receptor dual antagonists for immuno-oncology |
| WO2022020550A1 (en) | 2020-07-24 | 2022-01-27 | Merck Sharp & Dohme Corp. | Adenosine a2a and a2b receptor dual antagonists for immuno-oncology |
| WO2022023772A1 (en) | 2020-07-31 | 2022-02-03 | AdoRx Therapeutics Limited | Antagonist compounds |
| WO2022052886A1 (en) | 2020-09-08 | 2022-03-17 | 贝达药业股份有限公司 | Cd73 inhibitor and application thereof in medicine |
| WO2022068929A1 (en) | 2020-09-30 | 2022-04-07 | 武汉人福创新药物研发中心有限公司 | Pyrimidinedione compound and use thereof |
| WO2022072601A1 (en) | 2020-09-30 | 2022-04-07 | Na Biotech Corp | ANTI-ADENOSINE RECEPTOR (A2aR) ANTIBODIES |
| WO2022083049A1 (en) | 2020-10-19 | 2022-04-28 | 中山康方生物医药有限公司 | Anti-cd73 antibody and use thereof |
| WO2022090711A1 (en) | 2020-10-26 | 2022-05-05 | AdoRx Therapeutics Limited | Compounds as cd73 inhibitors |
| CN112321715A (en) | 2020-11-03 | 2021-02-05 | 博奥信生物技术(南京)有限公司 | anti-TROP 2 nano antibody and preparation method and application thereof |
| WO2022095953A1 (en) | 2020-11-05 | 2022-05-12 | 武汉人福创新药物研发中心有限公司 | Pyridazine alkyne compounds and use thereof |
| WO2022095975A1 (en) | 2020-11-05 | 2022-05-12 | 武汉人福创新药物研发中心有限公司 | Cd73 inhibitor and use thereof |
| WO2022096020A1 (en) | 2020-11-09 | 2022-05-12 | 江苏中新医药有限公司 | Anti-ecto-5'-nucleotidase antibody sequence |
| WO2022111576A1 (en) | 2020-11-27 | 2022-06-02 | Elpiscience (Suzhou) Biopharma, Ltd. | Novel conjugate molecules targeting cd39 and tgfβeta |
| WO2022121914A1 (en) | 2020-12-10 | 2022-06-16 | 上海翰森生物医药科技有限公司 | Oxo-nitrogen ring derivative regulator, preparation method therefor, and application thereof |
| WO2022123272A1 (en) | 2020-12-11 | 2022-06-16 | AdoRx Therapeutics Limited | Antagonists of the adenosine a2a receptor |
| WO2022078524A2 (en) | 2021-11-03 | 2022-04-21 | Hangzhou Dac Biotech Co., Ltd. | Specific conjugation of an antibody |
Non-Patent Citations (39)
| Title |
|---|
| ALLARD, B. ET AL., NAT REV CLIN ONCOL, vol. 17, 2020, pages 611 - 629 |
| ANONYMOUS: "Adenosine Receptor Antagonist Combination Therapy for Metastatic Castrate Resistant Prostate Cancer", CLINICALTRIALS.GOV, 11 May 2020 (2020-05-11), XP093066189, Retrieved from the Internet <URL:https%3A%2F%2Fclassic.clinicaltrials.gov%2Fct2%2Fshow%2FNCT04381832> [retrieved on 20230721] * |
| ANONYMOUS: "Sacituzumab Govitecan +/- Pembrolizumab in Metastatic TNBC", CLINICALTRIALS.GOV, 13 July 2020 (2020-07-13), XP093066142, Retrieved from the Internet <URL:https%3A%2F%2Fclassic.clinicaltrials.gov%2Fct2%2Fshow%2FNCT04468061> [retrieved on 20230721] * |
| ANTONARAKIS ES ET AL., J CLIN ONCOL., vol. 38, no. 5, 2020, pages 395 - 405 |
| ARMOUR, EUR. J. IMMUNOL., vol. 29, no. 8, 1999, pages 2613 - 2624 |
| AUGUSTIN RYAN C ET AL: "Next steps for clinical translation of adenosine pathway inhibition in cancer immunotherapy", JOURNAL FOR IMMUNOTHERAPY OF CANCER, BIOMED CENTRAL, LONDON, GB, vol. 10, no. 2, 31 January 2022 (2022-01-31), pages 1 - 13, XP009537691, ISSN: 2051-1426, DOI: 10.1136/JITC-2021-004089 * |
| BAKER ET AL., NUCLEIC ACIDS RES, vol. 37, no. 3, 2009, pages 693 - 701 |
| BARDIA A. ET AL: "Sacituzumab govitecan, a Trop-2-directed antibody-drug conjugate, for patients with epithelial cancer: final safety and efficacy results from the phase I/II IMMU-132-01 basket trial", ANNALS OF ONCOLOGY, vol. 32, no. 6, 1 June 2021 (2021-06-01), pages 746 - 756, XP093066113, ISSN: 0923-7534, DOI: 10.1016/j.annonc.2021.03.005 * |
| CANFIELDMORRISON, J EXP MED., vol. 173, no. 6, 1991, pages 1483 - 1491 |
| CHAUDHRY A. ET AL: "ARC-7: A phase II study to evaluate the safety and efficacy of zimberelimab alone, AB154 in combination with zimberelimab, and AB154 in combination with zimberelimab and AB928 in front-line, PD-L1 expressing, non-small cell lung cancer (NSCLC)", ANNALS OF ONCOLOGY, vol. 31, 1 September 2020 (2020-09-01), pages S897, XP093066165, ISSN: 0923-7534, DOI: 10.1016/j.annonc.2020.08.1733 * |
| COCKCROFT DWGAULT MH, NEPHRON, vol. 16, no. 1, 1976, pages 31 - 41 |
| DRAGO, J.Z. ET AL., NAI REV CLIN ONCOL, vol. 18, 2021, pages 327 344 |
| EISENHAUER EA ET AL., EUR J CANCER., vol. 45, no. 2, 2009, pages 228 - 247 |
| EISENHAUER ET AL., EUR J CANCER, vol. 45, no. 2, 2009, pages 228 - 47 |
| ETTINGER ET AL., J NATL COMPR CANC NETW, vol. 17, no. 12, 2019, pages 1464 - 72 |
| ETTINGER ET AL., J NATL COMPR CANE NETW, vol. 17, no. 12, 2019, pages 1464 - 72 |
| GOLDENBERG DM ET AL., ONCOTARGET, vol. 6, no. 26, 2015, pages 22496 |
| GOVINDAN SV ET AL., MOLECULAR CANCER THERAPEUTICS, vol. 12, no. 6, 2013, pages 968 - 78 |
| GUZIK ET AL., MOLECULES, vol. 24, no. 11, 2019, pages 2071 |
| HORN ET AL., J CLIN ONCO, vol. 35, no. 35, 2017, pages 3924 - 33 |
| LIE AK: "Histology of prostate cancer", ONCOLEX ONCOLOGY ENCYCLOPEDIA, 25 February 2020 (2020-02-25) |
| LOPEZ S ET AL., ONCOTARGET, vol. 11, no. 5, 2020, pages 560 |
| MARKHAM ANTHONY: "Zimberelimab: First Approval", 8 November 2021 (2021-11-08), XP093060623, Retrieved from the Internet <URL:http://www.ncbi.nlm.nih.gov/pubmed/34709602> [retrieved on 20230704] * |
| MAZIERES ET AL., JOURNAL OF THORACIC ONCOLOGY, vol. 16, no. 1, 2021, pages 140 - 50 |
| PERRONE E ET AL., FRONTIERS IN ONCOLOGY, 2020, pages 118 |
| ROHATGI A ET AL: "Abstract CT049: VELOCITY-Lung: A phase 2 study evaluating safety and efficacy of sacituzumab govitecan (SG) + zimberelimab (zim) + etrumadenant (etruma) in patients (pts) with advanced or metastatic non-small cell lung cancer (mNSCLC) progressing on or after platinum (PT)-based chemotherapy and chec", CANCER RES, 14 April 2023 (2023-04-14), American Association for Cancer Research Annual Meeting, ACCR 2023 - 20230414 to 20230419 - Orlando, FL, XP093060050, Retrieved from the Internet <URL:https://aacrjournals.org/cancerres/article/83/8_Supplement/CT049/725196/Abstract-CT049-VELOCITY-Lung-A-phase-2-study> [retrieved on 20230703] * |
| SAAD FHOTTE S., CAN UROL ASSOC J., vol. 4, no. 6, 2010, pages 380 - 384 |
| SALMANINEJAD ET AL., J. CELL PHYSIOL, vol. 234, no. 10, 2019, pages 16824 - 16837 |
| SAUNDERS, FRONT IMMUNOL., vol. 10, 2019, pages 1296 |
| SCHER HI ET AL., J CLIN ONCOL., vol. 26, no. 7, 2008, pages 1148 - 1159 |
| SHARKEY RM ET AL., CLINICAL CANCER RESEARCH, vol. 21, no. 22, 2015, pages 5131 - 8 |
| SHEPHERD ET AL., J CLIN ONCOL, vol. 18, no. 10, 2000, pages 2095 - 103 |
| SHIELDS, J. BIOL. CHEM., vol. 276, no. 9, 2001, pages 6591 - 6604 |
| SHIH LB ET AL., JOURNAL OF NUCLEAR MEDICINE, vol. 35, no. 5, 1994, pages 899 - 908 |
| SIEGEL ET AL., CA CANCER J CLIN, vol. 69, no. 1, 2019, pages 7 - 34 |
| STEIN R ET AL., CANCER RESEARCH, vol. 55, no. 14, 1995, pages 3132 - 9 |
| TAO, J EXP MED., vol. 178, 1993, pages 661 - 667 |
| VAN DER NEUT KOLFSCHOTEN ET AL., SCIENCE, vol. 317, 2007, pages 1554 - 1557 |
| ZEYBEK B ET AL., SCIENTIFIC REPORTS, vol. 10, no. 1, 2020, pages 973 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025186213A1 (en) | 2024-03-04 | 2025-09-12 | Debiopharm International S.A. | Combination of a wee1 inhibitor and a topoisomerase 1 inhibitor |
| WO2025188583A1 (en) * | 2024-03-06 | 2025-09-12 | Merck Sharp & Dohme Llc | Combination therapies for the treatment of hr+/her2- metastatic or locally advanced breast cancer |
| WO2025190251A1 (en) * | 2024-03-12 | 2025-09-18 | 百奥泰生物制药股份有限公司 | Use of anti-trop2 antibody-drug conjugate and pd-1 antagonist in combined treatment of solid tumor |
Also Published As
| Publication number | Publication date |
|---|---|
| CN119173276A (en) | 2024-12-20 |
| EP4507736A1 (en) | 2025-02-19 |
| JP2025512384A (en) | 2025-04-17 |
| AU2023252914A1 (en) | 2024-10-17 |
| KR20250004779A (en) | 2025-01-08 |
| TW202345845A (en) | 2023-12-01 |
| CA3256033A1 (en) | 2023-10-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2023201267A1 (en) | Combination therapy for treating trop-2 expressing cancers | |
| CN110536905B (en) | Anti-PD-1 antibodies for lung cancer treatment | |
| EP3307778A1 (en) | Treatment of cancer by combined blockade of the pd-1 and cxcr4 signaling pathways | |
| CN107922502A (en) | Use the method for immunity inspection point inhibitor for treating cancer | |
| US20220135682A1 (en) | Anti-ICOS Antibodies for the Treatment of Cancer | |
| US20230279096A1 (en) | Combination therapy with anti-il-8 antibodies and anti-pd-1 antibodies for treating cancer | |
| CN114599680A (en) | Anti-OX40 antibody and its use | |
| US20250249115A1 (en) | Combination therapy for treating tumor antigen expressing cancers | |
| TW202311287A (en) | Transforming growth factor-beta ligand traps for the treatment of disease | |
| CN114630839B (en) | Anti-OX40 antibodies and uses thereof | |
| AU2018353432A1 (en) | Combination product for the treatment of cancer | |
| KR20240099362A (en) | LAG-3 antagonist therapy for hematologic malignancies | |
| EP4096713A1 (en) | Anti-cd30 antibody-drug conjugates and their use for the treatment of non-hodgkin lymphoma | |
| CN113164599B (en) | Use of Anti-PD-L1 Monoclonal Antibody to Treat Cancer | |
| US20240270838A1 (en) | Method for prophylactic therapy of cytokine release syndrome and/or immune effector cell-associated neurotoxicity syndrome (icans) | |
| WO2024050515A1 (en) | Combination therapy for treating trop-2 expressing prostate cancers | |
| WO2023192478A1 (en) | Combination therapy with anti-il-8 antibodies and anti-pd-1 antibodies for treating cancer | |
| HK40053430A (en) | Anti-ox40 antibody and use thereof | |
| CN117580589A (en) | Combination of anti-galectin-9 antibodies and chemotherapeutic agents for use in cancer therapy | |
| HK40053431A (en) | Anti-ox40 antibody and use thereof | |
| HK40074868A (en) | Anti-ox40 antibody and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23721581 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: AU2023252914 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024560326 Country of ref document: JP |
|
| ENP | Entry into the national phase |
Ref document number: 2023252914 Country of ref document: AU Date of ref document: 20230412 Kind code of ref document: A |
|
| ENP | Entry into the national phase |
Ref document number: 20247037701 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020247037701 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023721581 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2023721581 Country of ref document: EP Effective date: 20241113 |