WO2023083201A1 - Aminopyrazole derivative, and preparation method therefor and use thereof - Google Patents
Aminopyrazole derivative, and preparation method therefor and use thereof Download PDFInfo
- Publication number
- WO2023083201A1 WO2023083201A1 PCT/CN2022/130771 CN2022130771W WO2023083201A1 WO 2023083201 A1 WO2023083201 A1 WO 2023083201A1 CN 2022130771 W CN2022130771 W CN 2022130771W WO 2023083201 A1 WO2023083201 A1 WO 2023083201A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- pharmaceutically acceptable
- compound
- independently selected
- haloalkyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/14—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D231/38—Nitrogen atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the disclosure relates to an aminopyrazole derivative, a preparation method and application thereof, and belongs to the field of medicine.
- Cyclin-dependent kinases are members of the serine/threonine kinase subfamily, and each CDK/cyclin complex is responsible for a specific phase transition or progression within the cell cycle, which plays an important role in regulating eukaryotic cell division and important role in proliferation. Cyclin-dependent kinase catalytic units are activated by regulatory subunits called cyclins. At least 16 mammalian cell cyclins have been identified (Annu. Rev. Pharmacol. Toxicol. (1999) 39:295-312).
- Cyclin B/CDK1, cyclin A/CDK2, cyclin E/CDK2, cyclin D/CDK4, cyclin D/CDK6 and possibly other heterodynes are important regulators of cell cycle progression.
- Other functions of cyclin/CDK heterodynes include transcriptional regulation, DNA repair, differentiation and apoptosis (Annu. Rev. Cell. Dev. Biol. (1997) 13:261-291).
- CDK2 Overexpression of CDK2 is associated with abnormal regulation of the cell cycle.
- the cyclin E/CDK2 complex plays an important role in regulating the G1/S transition, histone biosynthesis and centrosome duplication. Progressive phosphorylation of Rb by cyclin D/Cdk4/6 and cyclin E/Cdk2 releases the G1 transcription factor E2F and promotes S phase entry. Activation of cyclin A/CDK2 during early S phase promotes phosphorylation of endogenous substrates that allow DNA replication and inactivation of E2F to complete S phase (Nat.Rev.Drug.Discov.2015; 14( 2): 130-146).
- Cyclin E is overexpressed in a variety of cancers, especially breast cancer, lung cancer, leukemia, and lymphoma (Guo Cuiping et al., Cyclin E regulation and malignant tumors. International Journal of Oncology, 2012,39(005):337 -340), the amplification or overexpression of cyclin E is also associated with poor prognosis in ovarian, gastric, endometrial and other cancers.
- CDK2 cyclin A/E
- CDK2 also binds cyclin A for progression through S phase and participates in DNA repair.
- major companies have identified and discovered a series of selective CDK 2 inhibitors for the treatment of cancer and other diseases, such as Seliciclib, Dinaciclib, etc., but in order to achieve better cancer treatment effects, better meet Market demand still requires the development of a new generation of selective CDK2 inhibitors with high efficiency and low toxicity.
- the present disclosure provides a compound represented by formula I or a pharmaceutically acceptable salt thereof,
- said R1 and R2 are each independently selected from hydrogen, alkyl, haloalkyl, hydroxyalkyl, cycloalkyl or heterocycloalkyl, said alkyl, haloalkyl, hydroxyalkyl, ring Alkyl or heterocycloalkyl is optionally substituted by one or more substituents independently selected from R 1A ;
- the R 1A is selected from halogen, hydroxyl, alkyl, alkoxy, haloalkyl, haloalkoxy, heterocycloalkyl or NR'(R"), and the alkyl, alkoxy, haloalkyl, Haloalkoxy or heterocycloalkyl is optionally substituted by one or more substituents independently selected from R 5 ;
- R 1 and R 2 form a 3- to 7-membered heterocycloalkyl group with the nitrogen atom to which they are jointly connected, and the heterocycloalkyl group is optionally substituted by one or more substituents independently selected from R 1B ;
- the R 1B is selected from independently halogen, cyano, hydroxyl, amino, alkyl or alkoxy;
- the R 3 is selected from H, COR' or
- the ring A is selected from 5 to 6 membered aryl, 5 to 6 membered heteroaryl, cycloalkyl or heterocycloalkyl;
- R 4A two adjacent R 4 together form ring A optionally substituted by one or more independently selected from R 4A
- the alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, aryl, heteroaryl are any is selected to be replaced by one or more R 8 ;
- Said X is independently selected from O or S;
- the R 5 is selected from halogen, hydroxyl, alkyl, cyano, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl or NR'(R");
- the R and R are each independently selected from hydrogen, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl or heterocycloalkyl;
- the R is selected from halogen, hydroxyl, cyano, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl or NR'(R");
- R' and R" are each independently selected from H or alkyl
- n is selected from 1, 2, 3, 4, 5.
- the compound represented by formula I or a pharmaceutically acceptable salt thereof in the present disclosure is a compound represented by formula II or a pharmaceutically acceptable salt thereof,
- R 1 , R 2 , R 3 , R 4 , and n are respectively as defined in the compound represented by formula I.
- the compound represented by formula I or a pharmaceutically acceptable salt thereof in the present disclosure is a compound represented by formula III or a pharmaceutically acceptable salt thereof,
- R 1 , R 2 , R 4 , and n are respectively as defined in the compound represented by formula I.
- the compound represented by formula I or a pharmaceutically acceptable salt thereof in the present disclosure is a compound represented by formula IV or a pharmaceutically acceptable salt thereof,
- R 1 , R 2 , R 4 , and n are respectively as defined in the compound represented by formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from phenyl, five-membered nitrogen-containing heteroaryl, six-membered nitrogen-containing heteroaryl.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from pyridyl, pyrimidinyl, piperidinyl, imidazolyl, Pyrrolyl, pyrazolyl.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from pyridyl.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from phenyl.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein Ring A is I-1,
- the G 1 , G 2 , G 3 , G 4 and G 5 are each independently selected from CR 4 or N, wherein at least one of G 1 , G 2 , G 3 , G 4 and G 5 is CR 4 ,
- the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein Ring A is I-2,
- the G 1 , G 2 , G 3 and G 4 are each independently selected from CR 4 or N or NR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 3 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 1 , G 3 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound represented by formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 1 , G 2 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the G 3 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the G 2 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 3 and G 4 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the G 1 , G 3 and G 4 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 3 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the G 1 , G 3 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 3 and G 4 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 4 is CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 3 is CR 4 , and the R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein G 1 , G 2 , G 3 , G 4 and G 5 are each independently selected from CR4 .
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 2 , G 3 , G 4 and G 5 are selected from CR 4 , the selected from
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, the for
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 3 , G 4 and G 5 are each independently selected from CR 4 .
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is said selected from
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 3 , G 4 and G 5 are each independently selected from CR 4 .
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 4 and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound represented by formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 3 , G 4 and G 5 are each independently selected from CR 4 .
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 4 and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound represented by formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 3 , and G 4 are each independently selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 3 , and G 4 are each independently selected from CR 4 .
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein ring A is selected from
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 3 , and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 3 and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound represented by formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 3 and G 4 are each independently selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 4 is selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 3 is selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
- two adjacent R 4 together with ring A form an optionally substituted by one or more R 4A Wherein ring A is defined as described in any scheme herein.
- the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein said R is selected from hydrogen, alkyl, haloalkyl, hydroxyalkane group, cycloalkyl or heterocycloalkyl.
- the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein, said R 1 is selected from hydrogen, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 hydroxyalkyl, 3-7 membered cycloalkyl or 3-7 membered heterocycloalkyl.
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein said R 1 is selected from hydrogen, C 1-3 alkyl or C 1-3 haloalkyl.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 1 is hydrogen.
- the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein, said R 2 is selected from the group consisting of alkyl, haloalkyl, hydroxyalkyl, Cycloalkyl or heterocycloalkyl, said alkyl, haloalkyl, hydroxyalkyl, cycloalkyl or heterocycloalkyl are optionally substituted by one or more R 1A , said R 1A is selected from haloalkane group, alkoxy, haloalkyl, haloalkoxy, heterocycloalkyl or NR'(R"), the said alkyl, alkoxy, haloalkyl, haloalkoxy, heterocycloalkyl are optionally replaced by one Or multiple R 5 substitutions.
- said R 2 is selected from the group consisting of alkyl, haloalkyl, hydroxyalkyl, Cycloalkyl or heterocycloalkyl, said alkyl, hal
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 2 is selected from C 1-6 alkyl, C 1- 6 haloalkyl, C 1-6 hydroxyalkyl, 3 to 7 membered cycloalkyl or 3 to 7 membered heterocycloalkyl, said C 1-6 alkyl, C 1-6 haloalkyl, C 1 -6 hydroxyalkyl, 3 to 7-membered cycloalkyl or 3 to 7-membered heterocycloalkyl are optionally substituted by one or more R 1A ; said R 1A is selected from halogen, C 1-6 alkyl .
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein said R 2 is selected from methyl, ethyl, isopropyl, Tert-butyl, cyclopropyl, cyclobutyl, cyclopentyl, described cyclopropyl, cyclobutyl, cyclopentyl are substituted by one or more R 1A , described R 1A is selected from methyl, ethyl base, isopropyl.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 2 is selected from isopropyl or methylcyclopropyl.
- the present disclosure provides compounds represented by formulas I and II or pharmaceutically acceptable salts thereof, wherein said R is selected from Wherein X is O, the R 6 and R 7 are each independently selected from hydrogen, C 1-6 alkyl.
- the present disclosure provides compounds represented by formulas I and II or pharmaceutically acceptable salts thereof, wherein said R is selected from Wherein X is O, and the R 6 and R 7 are each independently selected from hydrogen, methyl, ethyl.
- the present disclosure provides compounds represented by formulas I and II or pharmaceutically acceptable salts thereof, wherein said R is selected from
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein R 4 is oxo.
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of the R 4 is independently selected from hydrogen, cyano, halogen, hydroxyl , alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, heteroaryl, SO 2 R', -NHSO 2 R', COR', -NHCOR', COOR', CONR'(R"), or
- the alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, heteroaryl are optionally substituted by one or more R 4A ;
- R 4A is selected from halogen, cyano, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy base, SO 2 R', -NHSO 2 R', COR', -NHCOR', COOR', CONR'(R") or
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein each R 4 is independently selected from hydrogen, cyano or halogen.
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of the R 4 is independently selected from hydrogen, COR', -NHSO 2 R', SO 2 R' or
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of said R 4 is independently selected from hydrogen, alkyl, hydroxyalkyl , the alkyl is optionally substituted by one or more R 4A , the R 4A is selected from halogen, cyano, alkoxy, SO 2 R' or
- the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of the R 4 is independently selected from hydrogen, alkoxy, heterocyclic Alkyloxy, or each R 4 is independently selected from C 1-6 alkoxy or 3 to 7 membered heterocycloalkyloxy.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 1 is hydrogen.
- the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 2 is selected from isopropyl or methylcyclopropyl.
- the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein the ring A is pyridyl, and at least one of the R 4 is cyano.
- the compound shown in the formula I, II, III, IV provided by the present disclosure or a pharmaceutically acceptable salt thereof wherein, in an optional embodiment, the compound of the formula I, II, III provided in the present disclosure , the compound shown in IV or a pharmaceutically acceptable salt thereof, wherein R' and R" are each independently selected from hydrogen or C 1-6 alkyl.
- the compound shown in the formula I, II, III, IV provided by the present disclosure or a pharmaceutically acceptable salt thereof wherein, in an optional embodiment, the compound of the formula I, II, III provided in the present disclosure , the compound shown in IV or a pharmaceutically acceptable salt thereof, wherein R' and R" are each independently selected from hydrogen or methyl and ethyl.
- the compound provided by the present disclosure or a pharmaceutically acceptable salt thereof is selected from:
- the present disclosure also provides isotopic substitutions of the compounds described above.
- the isotope substitution is deuterium atom substitution.
- the present disclosure also provides a pharmaceutical composition, which comprises the compound represented by the aforementioned formula I, II, III, IV or its pharmaceutically acceptable salt or its isotope substitution and at least one pharmaceutically acceptable carrier and diluent or excipients.
- the unit dose of the pharmaceutical composition is 0.001 mg-1000 mg.
- the pharmaceutical composition contains 0.01-99.99% of the compound represented by the aforementioned formula I, II, III, IV or a pharmaceutically acceptable salt thereof or an isotope substitution thing. In certain embodiments, the pharmaceutical composition contains 0.1-99.9% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof. In some embodiments, the pharmaceutical composition contains 0.5%-99.5% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof.
- the pharmaceutical composition contains 1%-99% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof. In certain embodiments, the pharmaceutical composition contains 2%-98% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof.
- the pharmaceutical composition contains 0.01%-99.99% of pharmaceutically acceptable excipients based on the total weight of the composition. In certain embodiments, the pharmaceutical composition contains 0.1%-99.9% of pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical composition contains 0.5%-99.5% of pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical composition contains 1%-99% of pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical composition contains 2%-98% of pharmaceutically acceptable excipients.
- the present disclosure also provides a method for preventing and/or treating a patient suffering from a protein-dependent kinase-related disease, by administering to the patient a therapeutically effective amount of the formulas I, II, III, and IV described in the present disclosure.
- the present disclosure also provides a method for preventing and/or treating a patient suffering from a cyclin-related disease, by administering to the patient a therapeutically effective amount of the compounds represented by formulas I, II, III, and IV described in the present disclosure.
- the protein-dependent kinase-related disease or cyclin-related disease is selected from cell proliferative disease, cancer or immune disease.
- the protein-dependent kinase-associated disease or cyclin-associated disease is selected from breast cancer, ovarian cancer, prostate cancer, melanoma, brain tumor, esophageal cancer, gastric cancer, liver cancer, pancreatic cancer, Colorectal cancer, lung cancer, kidney cancer, skin cancer, glioblastoma, neuroblastoma, sarcoma.
- the cancer is selected from cancers in which cyclin E1 and/or cyclin E2 are amplified.
- the present disclosure also provides a method for preventing and/or treating a patient suffering from cancer, by administering to the patient a therapeutically effective amount of the compound represented by the formulas I, II, III, IV described in the present disclosure or its alternative Pharmaceutically acceptable salt or its isotope substitution or the aforementioned pharmaceutical composition, the cancer is selected from breast cancer, ovarian cancer, prostate cancer, melanoma, brain tumor, esophageal cancer, gastric cancer, liver cancer, pancreatic cancer, colorectal cancer, Lung cancer, kidney cancer, skin cancer, glioblastoma, neuroblastoma, sarcoma.
- the present disclosure provides a therapeutically effective dose of the compounds shown in formulas I, II, III, and IV described in the present disclosure or their pharmaceutically acceptable salts or isotope substitutions or the aforementioned pharmaceutical compositions in the preparation for preventing and/or Use in medicines for the treatment of diseases associated with protein-dependent kinases,
- the protein-dependent kinase is selected from CDK2, and the disease related to the protein-dependent kinase is selected from cell proliferative diseases, cancer or immune diseases.
- the present disclosure provides a therapeutically effective amount of the compound shown in formula I, II, III, IV described in the present disclosure or its pharmaceutically acceptable salt or its isotope substitution or the aforementioned pharmaceutical composition in preparation for use in Use in medicines for preventing and/or treating diseases related to cell cyclins.
- the cyclin is selected from cyclin E, such as cyclin E1, cyclin E2.
- the cyclin-associated disease is selected from cell proliferative disease, cancer or immune disease.
- the present disclosure provides a therapeutically effective amount of a compound represented by formula I, II, III, IV of the present disclosure or a pharmaceutically acceptable salt thereof or an isotope substitution thereof or the aforementioned pharmaceutical composition in the preparation of a drug for the treatment of cancer Uses in medicine.
- the cancer is selected from breast cancer, ovarian cancer, prostate cancer, melanoma, brain tumor, esophageal cancer, gastric cancer, liver cancer, pancreatic cancer, colorectal cancer, lung cancer, kidney cancer, skin cancer Carcinoma, glioblastoma, neuroblastoma, sarcoma.
- the cancer is selected from cancers in which cyclin E1 and/or cyclin E2 are amplified.
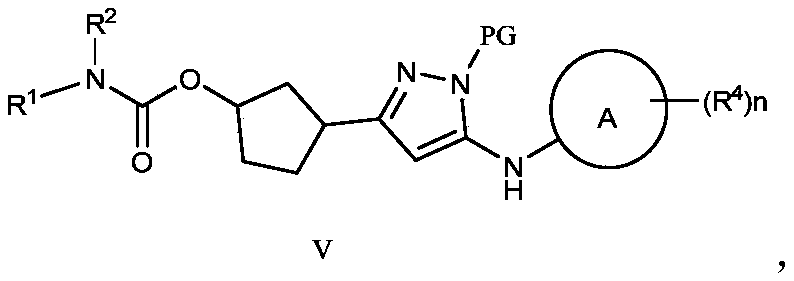
- Another aspect of the present disclosure provides a preparation method of the compound represented by formula III or its pharmaceutically acceptable salts and isotope substitutions, which includes the step of removing the amino protecting group PG from the compound represented by formula V under acidic conditions,
- R 1 , R 2 , R 4 , and n are respectively as defined in the compound shown in formula III, and PG is an amino protecting group, which can be tert-butyl, acetyl, trifluoroacetyl, trityl, benzyl base, formyl.
- Another aspect of the present disclosure provides a compound represented by formula V,
- R 1 , R 2 , R 4 , and n are respectively as defined in the compound shown in formula III, and PG is an amino protecting group, which can be tert-butyl, acetyl, trifluoroacetyl, trityl, benzyl base, formyl.
- the pharmaceutically acceptable salts of the compounds described in the present disclosure are selected from inorganic salts or organic salts, and the compounds described in the present disclosure can react with acidic or basic substances to form corresponding salts.
- the disclosed compound has a good inhibitory effect on cell cycle protein-dependent kinase, and the IC 50 value of the inhibitory activity to CDK2/Cyclin E is 0.01 to 1000nM, and the IC 50 value of the inhibitory activity of some compounds on CDK2/Cyclin E In the range of 0.01 to 500nM, the IC 50 values of the inhibitory activity of some compounds on CDK2/Cyclin E are in the range of 0.01 to 300nM, and the IC 50 values of the inhibitory activity of some compounds on CDK2/Cyclin E are in the range of 0.01 to 200nM.
- the IC50 value of the inhibitory activity of CDK2/Cyclin E is between 0.01 and 100nM, and the IC50 value of the inhibitory activity of some compounds against CDK2/Cyclin E is ⁇ 100nM.
- Some embodiments provide compounds that achieve ⁇ 50 nM inhibitory activity against CDK2/Cyclin E. Other embodiments provide compounds that achieve ⁇ 20 nM inhibitory activity against CDK2/Cyclin E. Other embodiments provide compounds that have ⁇ 10 nM inhibitory activity against CDK2/Cyclin E. Other embodiments provide compounds that achieve ⁇ 5nM inhibitory activity on CDK2/Cyclin E. Other embodiments provide compounds that achieve ⁇ 1 nM inhibitory activity against CDK2/Cyclin E.
- the disclosed compound also has a good inhibitory effect on CDK2/Cyclin A.
- Some embodiments provide compounds that achieve ⁇ 500 nM inhibitory activity against CDK2/Cyclin A.
- Other embodiments provide compounds that achieve ⁇ 200 nM inhibitory activity against CDK2/Cyclin A.
- Other embodiments provide compounds that achieve ⁇ 150 nM inhibitory activity against CDK2/Cyclin A.
- Other embodiments provide compounds that achieve ⁇ 100 nM inhibitory activity against CDK2/Cyclin A.
- Other embodiments provide compounds that have CDK2/Cyclin A inhibitory activity ⁇ 50 nM.
- Other embodiments provide compounds that achieve ⁇ 20 nM inhibitory activity against CDK2/Cyclin A.
- Other embodiments provide compounds that achieve ⁇ 10 nM inhibitory activity against CDK2/Cyclin A.
- the compounds of the present disclosure have weak inhibitory activity on other kinases such as CDK1, CDK4, CDK5, CDK7, CDK9 or GSK3 ⁇ , and have CDK2-specific selection.
- the disclosed compounds may exist in particular geometric or stereoisomeric forms.
- This disclosure contemplates all such compounds, including cis and trans isomers, (-)- and (+)-enantiomers, (R)- and (S)-enantiomers, diastereomers isomers, (D)-isomers, (L)-isomers, and their racemic and other mixtures, such as enantiomerically or diastereomerically enriched mixtures, all of which are within the scope of this disclosure.
- Additional asymmetric carbon atoms may be present in substituents such as alkyl groups. All such isomers, as well as mixtures thereof, are included within the scope of this disclosure.
- tautomer or tautomeric form refers to structural isomers of different energies that can interconvert via a low energy barrier.
- proton tautomers also known as prototropic tautomers
- interconversions via migration of a proton such as keto-enol and imine-enamine, lactam-lactam, pyrazole base isomerization.
- Compounds of the present disclosure may be asymmetric, eg, possess one or more stereoisomers. Unless otherwise stated, all stereoisomers are included, such as enantiomers and diastereomers.
- Compounds of the present disclosure containing asymmetric carbon atoms can be isolated in optically pure or racemic forms. Optically pure forms can be resolved from racemic mixtures or synthesized by using chiral starting materials or reagents.
- Optically active (R)- and (S)-isomers as well as D and L-isomers can be prepared by chiral synthesis or chiral reagents or other conventional techniques. If one enantiomer of a compound of the present disclosure is desired, it can be prepared by asymmetric synthesis or derivatization with chiral auxiliary agents, wherein the resulting diastereomeric mixture is separated and the auxiliary group is cleaved to provide pure desired enantiomer.
- a diastereoisomeric salt is formed with an appropriate optically active acid or base, and then a diastereomeric salt is formed by a conventional method known in the art. Diastereomeric resolution is performed and the pure enantiomers are recovered. Furthermore, the separation of enantiomers and diastereomers is usually accomplished by the use of chromatography using chiral stationary phases, optionally in combination with chemical derivatization methods (e.g. amines to amino groups formate).
- the present disclosure also includes certain isotopically labeled compounds of the disclosure that are identical to those described herein, but wherein one or more atoms are replaced by an atom of an atomic mass or mass number different from that normally found in nature.
- isotopes that can be incorporated into compounds of the present disclosure include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, iodine, and chlorine, such as 2 H, 3 H, 11 C, 13 C, 14 C, 13 N, 15 N, 15 O, 17 O, 18 O, 31 P, 32 P, 35 S, 18 F, 123 I, 125 I and 36 Cl, etc.
- deuterium when a position is specifically designated as deuterium (D), the position is understood to have an abundance of deuterium (i.e., at least 10 % deuterium incorporation).
- exemplary compounds having a natural abundance greater than deuterium can be at least 1000 times more abundant deuterium, at least 2000 times more abundant deuterium, at least 3000 times more abundant deuterium, at least 4000 times more abundant deuterium, at least 5000 times more abundant deuterium, at least 6000 times more abundant deuterium, or more abundant deuterium.
- the present disclosure also includes various deuterated forms of compounds of formula I. Each available hydrogen atom attached to a carbon atom can be independently replaced by a deuterium atom.
- deuterated starting materials can be used in the preparation of deuterated forms of compounds of formula I, or they can be synthesized using conventional techniques using deuterated reagents, including but not limited to deuterated borane, trideuterated borane Tetrahydrofuran solution, deuterated lithium aluminum hydride, deuterated ethyl iodide and deuterated methyl iodide, etc.
- C 1-6 alkyl optionally substituted by halogen or cyano means that halogen or cyano may but not necessarily exist, and this description includes the case where the alkyl is substituted by halogen or cyano and the alkyl is not substituted by halogen And the case of cyano substitution.
- the bond Indicates unassigned configuration, i.e. if chiral isomers exist in the chemical structure, the bond can be or or both and Two configurations.
- the present invention may include all isomers such as tautomers, rotamers, geometric isomers, diastereoisomers body, racemate and enantiomer.
- the bond If the configuration is not specified, it can be Z configuration or E configuration, or both configurations.
- “Pharmaceutical composition” means a mixture containing one or more compounds described herein, or a physiologically acceptable salt or prodrug thereof, and other chemical components, as well as other components such as physiologically acceptable carriers and excipients. Forming agent.
- the purpose of the pharmaceutical composition is to promote the administration to the organism, facilitate the absorption of the active ingredient and then exert the biological activity.
- “Pharmaceutically acceptable excipients” include, but are not limited to, any adjuvants, carriers, glidants, sweeteners, diluents that have been approved by the U.S. Food and Drug Administration (FDA) to be acceptable for human or livestock use , preservative, dye/colorant, flavor enhancer, surfactant, wetting agent, dispersant, suspending agent, stabilizer, isotonic agent, solvent or emulsifier.
- FDA Food and Drug Administration
- an “effective amount” or “therapeutically effective amount” as used in the present disclosure includes an amount sufficient to ameliorate or prevent a symptom or condition of a medical condition.
- An effective amount also means an amount sufficient to permit or facilitate diagnosis.
- Effective amounts for a particular patient or veterinary subject may vary depending on factors such as the condition being treated, the general health of the patient, the method, route and dosage of administration, and the severity of side effects.
- An effective amount may be the maximum dose or dosing regimen that avoids significant side effects or toxic effects.
- Alkyl refers to a saturated aliphatic hydrocarbon group, including straight and branched chain groups of 1 to 20 carbon atoms. An alkyl group containing 1 to 6 carbon atoms. Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 2,2-dimethylpropyl and various branched isomers, etc.
- Alkyl groups may be substituted or unsubstituted, and when substituted, substituents may be substituted at any available point of attachment, preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, cyano, amino, C 1-6 alkyl, C 1-6 alkoxy, 3 to 6 membered cycloalkyl or 3 to 6 membered heterocycloalkyl, the alkyl, alkoxy, cycloalkyl or Heterocycloalkyl is optionally substituted with halo, hydroxy, nitro, cyano or amino.
- alkenyl refers to an alkyl compound containing at least one carbon-carbon double bond in the molecule, wherein alkyl is as defined above.
- Alkenyl groups may be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from the group consisting of alkoxy, halo, haloalkyl, haloalkoxy, hydroxy, hydroxyalkane
- alkynyl refers to an alkyl compound containing at least one carbon-carbon triple bond in the molecule, wherein alkyl is as defined above.
- Alkynyl groups may be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from alkoxy, halo, haloalkyl, haloalkoxy, hydroxy, hydroxyalkane
- cycloalkyl refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent, the cycloalkyl ring containing 3 to 20 carbon atoms, preferably containing 3 to 6 carbon atoms.
- monocyclic cycloalkyls include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, etc.
- multicyclic cycloalkyls include spiro Cycloalkyls of rings, parallel rings and bridged rings.
- Cycloalkyl groups may be substituted or unsubstituted, and when substituted, the substituents may be substituted at any available point of attachment, preferably one or more of the following groups, independently selected from halogen, hydroxy, oxo , cyano, amino, C 1-6 alkyl, C 1-6 alkoxy, 3 to 6 membered cycloalkyl or 3 to 6 membered heterocycloalkyl, the alkyl, alkoxy, cycloalkyl Or heterocycloalkyl is optionally substituted with halo, hydroxy, nitro, cyano or amino.
- heterocycloalkyl refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent comprising 3 to 20 ring atoms, one or more of which are selected from nitrogen, oxygen or A heteroatom of S(O) m (where m is an integer from 0 to 2), excluding ring portions of -OO-, -OS- or -SS-, the remaining ring atoms being carbon.
- m is an integer from 0 to 2
- it contains 3 to 12 ring atoms, of which 1 to 4 are heteroatoms; more preferably it contains 3 to 7 ring atoms.
- Non-limiting examples of “heterocycloalkyl” include:
- heterocycloalkyl ring may be fused to an aryl or heteroaryl ring, wherein the ring bonded to the parent structure is a heterocycloalkyl, non-limiting examples of which include:
- Heterocycloalkyl groups may be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, cyano, amino, C 1-6 alkyl, C 1-6 alkoxy, 3 to 6 membered cycloalkyl or 3 to 6 membered heterocycloalkyl, the alkyl, alkoxy, cycloalkyl or heterocycloalkyl optionally Substituted by halogen, hydroxy, nitro, cyano or amino.
- alkoxy refers to -O-(alkyl), wherein alkyl is as defined above.
- alkoxy include: methoxy, ethoxy, propoxy, butoxy.
- Alkoxy may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, cyano, amino, C -6 alkyl, C 1-6 alkoxy, 3 to 7 membered cycloalkyl or 3 to 7 membered heterocycloalkyl, said alkyl, alkoxy, cycloalkyl or heterocycloalkyl are optionally Halogen, hydroxy, nitro, cyano or amino substituted.
- cycloalkyloxy and “heterocycloalkyloxy” are as defined above for “alkoxy”, specifically -O-(cycloalkyl), -O-(heterocycloalkyl).
- alkylthio refers to -S-(alkyl), wherein alkyl is as defined above.
- alkoxy include: methylthio, ethylthio, propylthio, butylthio.
- Alkylthio may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups independently selected from C 1-6 alkoxy, 3 to 6 membered cycloalkane group, 3 to 6 membered heterocycloalkyl, 3 to 6 membered cycloalkoxy, 3 to 6 membered heterocycloalkyloxy, C 1-6 alkylthio, 3 to 6 membered cycloalkylthio, 3 to 6 6-membered heterocycloalkylthio, the alkoxy, cycloalkyl, heterocycloalkyl, cycloalkoxy, heterocyclyloxy, alkylthio, cycloalkylthio, heterocycloalkylthio are optionally Halogen, hydroxy, cyano or amino substituted.
- cycloalkylthio and “heterocycloalkylthio” have the same definition as the above-mentioned “alkylthio”.
- “Monovalent group” means that a compound is “formally” eliminated from a monovalent atom or group. “Subunit” refers to a compound formed by “formally” eliminating two monovalent or one divalent atoms or atomic groups.
- alkylene means what remains of an alkane molecule after removal of 2 hydrogen atoms, including straight and branched chain subgroups of 1 to 20 carbon atoms.
- An alkylene group having 1 to 6 carbon atoms include methylene (-CH 2 -), ethylene (eg -CH 2 CH 2 - or -CH(CH 3 )-).
- the alkylene group may be substituted or unsubstituted, and when substituted, the substituent may be substituted at any available point of attachment, preferably one or more of the following groups, independently selected from halogen , hydroxyl, cyano, amino, C 1-6 alkyl or C 1-6 alkoxy.
- alkyleneoxy Similarly, the definitions of "alkyleneoxy”, “alkenylene”, “alkenyleneoxy”, “cycloalkylene”, and “heterocycloalkylene” are the same as “alkylene”.
- aryl refers to a 6 to 14 membered all-carbon monocyclic or fused polycyclic (that is, rings sharing adjacent pairs of carbon atoms) group, preferably 6 to 12 membered, having a conjugated ⁇ -electron system, such as benzene base and naphthyl.
- the aryl ring may be fused to a heteroaryl, heterocycloalkyl or cycloalkyl ring, where the ring bonded to the parent structure is an aryl ring, non-limiting examples of which include:
- Aryl groups may be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, nitro, cyano, C1-6 Alkyl, C 1-6 alkoxy, C 2-6 alkenyloxy, C 2-6 alkynyloxy, 3 to 6-membered cycloalkoxy, 3 to 6-membered heterocycloalkyloxy, C 3- 8 cycloalkenyloxy, 5 to 6 membered aryl or heteroaryl, said C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyloxy, C 2-6 alkynyloxy, 3 to 6-membered cycloalkoxy, 3 to 6-membered heterocycloalkyloxy, 3 to 8-membered cycloalkenyloxy, 5 to 6-membered aryl or heteroaryl are optionally replaced by one or more members selected from halogen, Hydroxy, cyano, amino
- heteroaryl refers to a heteroaromatic system comprising 1 to 4 heteroatoms, 5 to 14 ring atoms, wherein the heteroatoms are selected from oxygen, sulfur and nitrogen.
- the heteroaryl group is preferably 6 to 12 membered, more preferably 5 or 6 membered.
- Non-limiting examples thereof include: imidazolyl, furyl, thienyl, thiazolyl, pyrazolyl, oxazolyl, isoxazolyl, pyrrolyl, tetrazolyl, pyridyl, pyrimidinyl , Thiadiazole, pyrazinyl, triazolyl, indazolyl, benzimidazolyl, wait.
- the heteroaryl ring may be fused to an aryl, heterocycloalkyl or cycloalkyl ring, wherein the ring bonded to the parent structure is a heteroaryl ring, non-limiting examples of which include:
- Heteroaryl may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups independently selected from halogen, hydroxy, cyano, amino, C alkane group or C 1-6 alkoxy group (here needs to be adjusted according to the claims!).
- spiro refers to a compound in which two rings share one atom.
- spirocycloalkyl groups include:
- merged ring refers to a compound formed by combining two or more rings by sharing two adjacent atoms.
- cycloalkyl groups include:
- bridged ring refers to a structure formed by two or more ring structures sharing two non-adjacent ring atoms with each other. According to the number of constituent rings, it can be divided into bicyclic, tricyclic, tetracyclic or polycyclic bridged cycloalkyl groups, preferably bicyclic, tricyclic or tetracyclic, more preferably bicyclic or tricyclic.
- bridged cycloalkyl groups include:
- heterocyclic ring refers to atoms other than carbon atoms that make up the ring, and includes heterocycloalkyl and heteroaryl rings.
- hydroxyl refers to a -OH group.
- halogen refers to fluorine, chlorine, bromine or iodine.
- cyano refers to -CN.
- amino refers to -NH2 .
- nitro refers to -NO2 .
- Substituted means that one or more hydrogen atoms in a group, preferably up to 5, more preferably 1 to 3 hydrogen atoms are independently substituted by the corresponding number of substituents.
- two (2) hydrogens on the atom are replaced.
- Substituted by one or more means that it can be substituted by single or multiple substituents. When substituted by a plurality of substituents, it may be a plurality of the same substituents, or one or a combination of a plurality of different substituents.
- NMR nuclear magnetic resonance
- MS mass spectroscopy
- MS was determined by Shimadzu 2010 Mass Spectrometer or Agilent 6110A MSD mass spectrometer.
- HPLC uses Shimadzu LC-20A systems, Shimadzu LC-2010HT series or Agilent Agilent 1200LC high pressure liquid chromatography (Ultimate XB-C18 3.0*150mm column or Xtimate C18 2.1*30mm column).
- Chiralpak IC-3 100 ⁇ 4.6mm I.D., 3um, Chiralpak AD-3 150 ⁇ 4.6mm I.D., 3um, Chiralpak AD-3 50 ⁇ 4.6mm I.D., 3um, Chiralpak AS-3 150 ⁇ 4.6mm were used for chiral HPLC analysis and determination I.D., 3um, Chiralpak AS-3 100 ⁇ 4.6mm I.D., 3 ⁇ m, ChiralCel OD-3 150 ⁇ 4.6mm I.D., 3um, Chiralcel OD-3 100 ⁇ 4.6mm I.D., 3 ⁇ m, ChiralCel OJ-H 150 ⁇ 4.6mm I.D., 5um, Chiralcel OJ-3 150 ⁇ 4.6mm I.D., 3um column;
- the thin-layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate.
- the specification of the silica gel plate used in thin-layer chromatography (TLC) is 0.15mm-0.2mm, and the specification of thin-layer chromatography separation and purification products is 0.4mm. ⁇ 0.5mm.
- the chiral preparative column uses DAICEL CHIRALPAK IC (250mm*30mm, 10um) or Phenomenex-Amylose-1 (250mm*30mm, 5um).
- the CombiFlash rapid preparation instrument uses Combiflash Rf150 (TELEDYNE ISCO).
- the known starting materials of the present disclosure can be adopted or synthesized according to methods known in the art, or can be purchased from ABCR GmbH & Co. KG, Acros Organics, Aldrich Chemical Company, Shaoyuan Chemical Technology (Accela ChemBio Inc), Darui chemical companies.
- the reactions can all be carried out under an argon atmosphere or a nitrogen atmosphere.
- Argon atmosphere or nitrogen atmosphere means that the reaction bottle is connected to an argon or nitrogen balloon with a volume of about 1 L.
- the hydrogen atmosphere means that the reaction bottle is connected to a hydrogen balloon with a capacity of about 1L.
- the pressurized hydrogenation reaction uses Parr 3916EKX hydrogenation instrument and Qinglan QL-500 hydrogen generator or HC2-SS hydrogenation instrument.
- the hydrogenation reaction is usually vacuumized and filled with hydrogen, and the operation is repeated 3 times.
- the solution refers to an aqueous solution.
- reaction temperature is room temperature, which is 20°C to 30°C.
- the monitoring of the reaction process in the embodiment adopts thin-layer chromatography (TLC), the developing agent used in reaction, the eluent system of the eluent system of the column chromatography that purification compound adopts and the developing agent system of thin-layer chromatography, the volume of solvent
- TLC thin-layer chromatography
- the ratio is adjusted according to the polarity of the compound, and it can also be adjusted by adding a small amount of basic or acidic reagents such as triethylamine and acetic acid.
- Compound 4 was prepared according to the synthesis steps of Example 1 by replacing the corresponding raw materials.
- 2-Bromo-5-chloropyrazine 5a (500mg, 2.6mmol) was first dissolved in N,N-dimethylformamide (20ml) followed by the addition of tert-butyl 2-cyanoacetate (733mg, 5.2mmol) , cesium carbonate (2.1g, 6.5mmol), react at room temperature for 4 hours. The crude product is obtained after extraction, drying and concentration, which can be directly used in the next reaction.
- Compound 6 was prepared according to the similar steps of Example 1 by substituting corresponding raw materials and reagents.
- Compound 7 was prepared according to the synthesis steps of Example 1 by replacing the corresponding raw materials.
- Compound 12 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
- reaction solution was acidified with 1M hydrochloric acid, extracted with ethyl acetate, the organic phases were combined, washed with saturated brine, dried, and concentrated under reduced pressure to obtain the title compound 13e (424.6 mg, yield 90 %).
- reaction solution was quenched with water, extracted with dichloromethane, the organic phases were combined, and the organic phase was successively washed with 1M hydrochloric acid, 1M aqueous sodium hydroxide solution and saturated brine, dried, concentrated under reduced pressure, and the residue was separated and purified by silica gel column chromatography to obtain The title compound 13f (247.4 mg, 60% yield).
- Compound 16 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
- Compound 18 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
- Compound 19 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
- Compound 20 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
- Compound 21 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
- Compound 22 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
- Deuteromethylamine hydrochloride (154mg, 2.2mmol) was dissolved in 15mL of dichloromethane, triethylamine (557mg, 5.52mmol), 4-dimethylaminopyridine (45mg, 0.37mmol) were added to the system, 0 Add 4-bromo-3-fluorobenzenesulfonyl chloride (500mg, 1.84mmol) at °C and stir overnight at room temperature. After the reaction, the reaction solution was concentrated under reduced pressure, and the residue was purified by silica gel chromatography to obtain the title compound 23b (145 mg, yield 29.2%).
- Compound 23c was prepared according to the synthetic procedure of Example 11 by replacing the corresponding raw materials. (56 mg, yield 75.9%).
- Compound 23 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials. (19 mg, yield 38.2%).
- deuterated methylamine hydrochloride (210mg, 3mmol) and DIEA (650mg, 5mmol) were dissolved in anhydrous dichloromethane (10mL), and after clarification, 5-bromopyridine-2-sulfonyl chloride ( 250mg, 1mmol), continue to stir, TLC monitors until the end of the reaction, add water (10mL) to wash once, and saturated brine (10mL) to wash once, collect the organic phase and dry it over anhydrous sodium sulfate, spin to a small volume and directly normalize Column chromatography gave the title compound 25b (200 mg, yield 82%).
- the title compound 27 was prepared according to the synthesis procedure of Example 25 by replacing the corresponding raw materials.
- Compound 28 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
- Compound 29 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
- Compound 30 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
- Compound 32 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
- Compound 33 was prepared according to the synthetic steps of Example 8 by replacing the corresponding raw materials.
- Compound 34 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
- Compound 35 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
- Compound 36 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials and reagents.
- Compound 37 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials and reagents.
- Compound 38 was prepared according to the synthetic steps of Example 11 by replacing corresponding raw materials and reagents.
- Compound 39 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials and reagents.
- Compound 40 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
- Compound 41 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
- Compound 42 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
- Test Example 1 Test of the Inhibitory Activity of the Compounds of the Disclosure on Ovarian Cancer Cells (OVCAR3)
- Ovarian cancer cells OVCAR3 were cultured in RPMI 1640 with 10% FBS in a 37%, 5% CO2 incubator. On the first day, cells were plated in 96-well plates at a concentration of 2500 cells/well and incubated overnight in an incubator. Compound treatment was performed on the second day, the highest concentration of compound treatment was 10 uM, 3-fold dilution, 9 concentrations, and the final concentration of DMSO was 0.1%. After the cells continued to be cultured in the incubator for 7 days, the cell viability was tested using the Celltiter Glo assay kit (Promega), and the test method was consistent with the operation method provided by the kit. Data were processed and IC50 calculated using GraphPad Prism 8.
- X log value of compound concentration
- Y % inhibition.
- Test Example 2 Detection of the activity of the disclosed compounds on cyclin-dependent kinases (CDK1, 2, 9).
- Compound dilutions were transferred to each well of the assay plate using an Echo 550 (784075, Greiner). Seal the assay plate and centrifuge the assay plate at 1000g for 1 min; prepare 2x enzyme in 1x kinase buffer ((40mM Tris-HCl, pH 7.4, 20mM MgCl2, 0.1mg/ml BSA, 50uM DTT) by dissolving 2.5 ⁇ l of 2x enzyme Add to 384-well assay plate, centrifuge the plate at 1000g for 30s, and let it stand at room temperature for 10 minutes. Prepare 2x substrate and ATP mix in 1x kinase buffer, add 2.5 ⁇ l 2x substrate and ATP mix to start the reaction.
- 1x kinase buffer ((40mM Tris-HCl, pH 7.4, 20mM MgCl2, 0.1mg/ml BSA, 50uM DTT)
- the luminescent signal of each well was read on an Envision 2104 plate reader.
- Percent inhibition 100 - (cmpd signal - Ave_PC signal) / (Ave_VC signal - Ave_PC signal) x 100. cmpd signal: compound signal; Ave_PC signal: positive control signal average; Ave_VC signal: vector group signal average.
- Test example 3 the pharmacokinetic test of embodiment 8 of the present invention and embodiment 9
- Example 8 Taking Balb/c nude mouse as the test animal, the LC/MS/MS method was used to determine the drug concentration in the blood plasma at different times after the oral administration of Example 8 and Example 9. Study the pharmacokinetic behavior of the compound of the present invention in Balb/c nude mouse body, and evaluate its pharmacokinetic characteristics.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
本公开涉及一种氨基吡唑衍生物及其制备方法和用途,属于医药领域。The disclosure relates to an aminopyrazole derivative, a preparation method and application thereof, and belongs to the field of medicine.
细胞周期蛋白-依赖性激酶(CDK)是丝氨酸/苏氨酸激酶亚家族的成员,每个CDK/细胞周期蛋白复合物负责细胞周期内特定期的转换或进展,其在调节真核细胞分裂和增殖中发挥重要作用。细胞周期蛋白-依赖性激酶催化单元被称为细胞周期蛋白的调节亚基激活。已经鉴定出至少16种哺乳动物细胞周期蛋白(Annu.Rev.Pharmacol.Toxicol.(1999)39:295-312)。细胞周期蛋白B/CDK1、细胞周期蛋白A/CDK2、细胞周期蛋白E/CDK2、细胞周期蛋白D/CDK4、细胞周期蛋白D/CDK6和可能的其他heterodynes是细胞周期进展的重要调节因子。细胞周期蛋白/CDK heterodynes的其他功能包括转录调节、DNA修复、分化和凋亡(Annu.Rev.Cell.Dev.Biol.(1997)13:261-291)。Cyclin-dependent kinases (CDKs) are members of the serine/threonine kinase subfamily, and each CDK/cyclin complex is responsible for a specific phase transition or progression within the cell cycle, which plays an important role in regulating eukaryotic cell division and important role in proliferation. Cyclin-dependent kinase catalytic units are activated by regulatory subunits called cyclins. At least 16 mammalian cell cyclins have been identified (Annu. Rev. Pharmacol. Toxicol. (1999) 39:295-312). Cyclin B/CDK1, cyclin A/CDK2, cyclin E/CDK2, cyclin D/CDK4, cyclin D/CDK6 and possibly other heterodynes are important regulators of cell cycle progression. Other functions of cyclin/CDK heterodynes include transcriptional regulation, DNA repair, differentiation and apoptosis (Annu. Rev. Cell. Dev. Biol. (1997) 13:261-291).
近年来,乳腺癌治疗领域最大的进展无疑是CDK4/6单用或联合内分泌治疗在激素受体阳性晚期乳腺癌,如帕博西尼(palbociclib)、瑞博西尼(ribociclib)和玻玛西尼(abemaciclib)已被批准与芳香酶抑制剂组合用于治疗绝经后妇女的激素受体(HR)-阳性、人类表皮生长因子受体2(HER2)-阴性晚期或转移性乳腺癌,并且帕博西尼和玻玛西尼(abemaciclib)已被批准与氟维司群组合用于在内分泌疗法后疾病进展后治疗绝经后妇女的激素受体(HR)-阳性、人类表皮生长因子受体2(HER2)-阴性晚期或转移性乳腺癌(Nature Reviews(2016)13:417-430、J Clin Oncol 2017,35,2875-2884)。尽管CDK4/6抑制剂在雌激素受体ER阳性转移性乳腺癌中显示出显著的临床功效,但与其他激酶一样,它们的作用可能随着时间的推移被原发性或获得性抗性的发展限制。In recent years, the greatest progress in the field of breast cancer treatment is undoubtedly the CDK4/6 monotherapy or combined with endocrine therapy in hormone receptor positive advanced breast cancer, such as palbociclib, ribociclib and bomaciclib Abemaciclib has been approved in combination with an aromatase inhibitor for the treatment of postmenopausal women with hormone receptor (HR)-positive, human epidermal growth factor receptor 2 (HER2)-negative advanced or metastatic breast cancer, and Bociclib and abemaciclib have been approved in combination with fulvestrant for the treatment of hormone receptor (HR)-positive, human epidermal growth factor receptor 2 in postmenopausal women after disease progression on endocrine therapy (HER2)-negative advanced or metastatic breast cancer (Nature Reviews (2016) 13:417-430, J Clin Oncol 2017, 35, 2875-2884). Although CDK4/6 inhibitors have shown remarkable clinical efficacy in estrogen receptor ER-positive metastatic breast cancer, like other kinases, their effects may be over time controlled by primary or acquired resistance. development restrictions.
CDK2的过表达与细胞周期的异常调节有关。细胞周期蛋白E/CDK2复合物在调节G1/S转换、组蛋白生物合成和中心体复制中起重要作用。细胞周期蛋白D/Cdk4/6和细胞周期蛋白E/Cdk2对Rb的进行性磷酸化释放G1转录因子E2F,并促进S期进入。在早期S期期间细胞周期蛋白A/CDK2的激活促进内源性底物的磷酸化,其允许DNA复制和E2F的失活,以完成S期(Nat.Rev.Drug.Discov.2015;14(2):130-146)。细胞周期蛋白E在多种癌症中过度表达,特别是乳腺癌、肺癌、白血病、淋巴瘤(郭翠萍等,细胞周期蛋白E的调控与恶性肿瘤.国际肿瘤学杂志,2012,39(005):337-340),细胞周期蛋白E的扩增或过表达也与卵巢癌、胃癌、子宫内膜癌和其他癌症的不良预后有关。Overexpression of CDK2 is associated with abnormal regulation of the cell cycle. The cyclin E/CDK2 complex plays an important role in regulating the G1/S transition, histone biosynthesis and centrosome duplication. Progressive phosphorylation of Rb by cyclin D/Cdk4/6 and cyclin E/Cdk2 releases the G1 transcription factor E2F and promotes S phase entry. Activation of cyclin A/CDK2 during early S phase promotes phosphorylation of endogenous substrates that allow DNA replication and inactivation of E2F to complete S phase (Nat.Rev.Drug.Discov.2015; 14( 2): 130-146). Cyclin E is overexpressed in a variety of cancers, especially breast cancer, lung cancer, leukemia, and lymphoma (Guo Cuiping et al., Cyclin E regulation and malignant tumors. International Journal of Oncology, 2012,39(005):337 -340), the amplification or overexpression of cyclin E is also associated with poor prognosis in ovarian, gastric, endometrial and other cancers.
研究表明,抑制CDK2激酶会诱导肿瘤细胞调亡,但对于正常细胞只会造成较小的损伤。CDK激酶的单体形式是无活性的,而细胞周期蛋白A/E与 CDK2结合并促发磷酸化的结合激活CDK2。CDK2还可结合细胞周期蛋白A用于S期的整个进展并参与DNA修复。近几年各大公司分别鉴定发现了一系列选择性抑制CDK 2的抑制剂,用于治疗癌症等疾病,如Seliciclib、Dinaciclib等,但是为了达到更好的癌症治疗效果的目的,更好的满足市场需求,仍需要开发出新一代的高效低毒的选择性CDK2抑制剂。Studies have shown that inhibition of CDK2 kinase induces tumor cell apoptosis, but causes only minor damage to normal cells. The monomeric form of the CDK kinase is inactive, whereas the binding of cyclin A/E to CDK2 and triggering phosphorylation activates CDK2. CDK2 also binds cyclin A for progression through S phase and participates in DNA repair. In recent years, major companies have identified and discovered a series of selective CDK 2 inhibitors for the treatment of cancer and other diseases, such as Seliciclib, Dinaciclib, etc., but in order to achieve better cancer treatment effects, better meet Market demand still requires the development of a new generation of selective CDK2 inhibitors with high efficiency and low toxicity.
发明内容Contents of the invention
本公开提供一种式I所示的化合物或其可药用的盐,The present disclosure provides a compound represented by formula I or a pharmaceutically acceptable salt thereof,
其中,所述的R 1和R 2各自独立地选自氢、烷基、卤代烷基、羟烷基、环烷基或杂环烷基,所述的烷基、卤代烷基、羟烷基、环烷基或杂环烷基任选被一个或多个独立地选自R 1A的取代基取代; Wherein, said R1 and R2 are each independently selected from hydrogen, alkyl, haloalkyl, hydroxyalkyl, cycloalkyl or heterocycloalkyl, said alkyl, haloalkyl, hydroxyalkyl, ring Alkyl or heterocycloalkyl is optionally substituted by one or more substituents independently selected from R 1A ;
所述的R 1A选自卤素、羟基、烷基、烷氧基、卤代烷基、卤代烷氧基、杂环烷基或NR'(R”),所述的烷基、烷氧基、卤代烷基、卤代烷氧基或杂环烷基任选被一个或者多个独立地选自R 5的取代基取代; The R 1A is selected from halogen, hydroxyl, alkyl, alkoxy, haloalkyl, haloalkoxy, heterocycloalkyl or NR'(R"), and the alkyl, alkoxy, haloalkyl, Haloalkoxy or heterocycloalkyl is optionally substituted by one or more substituents independently selected from R 5 ;
或者R 1和R 2与其共同连接的氮原子形成3至7元的杂环烷基,所述的杂环烷基任选被一个或者多个独立地选自R 1B的取代基取代; Or R 1 and R 2 form a 3- to 7-membered heterocycloalkyl group with the nitrogen atom to which they are jointly connected, and the heterocycloalkyl group is optionally substituted by one or more substituents independently selected from R 1B ;
所述的R 1B选各自独立地自卤素、氰基、羟基、氨基、烷基或烷氧基; The R 1B is selected from independently halogen, cyano, hydroxyl, amino, alkyl or alkoxy;
所述的R 3选自H、COR'或 The R 3 is selected from H, COR' or
所述的环A选自5至6元的芳基、5至6元的杂芳基、环烷基或杂环烷基;The ring A is selected from 5 to 6 membered aryl, 5 to 6 membered heteroaryl, cycloalkyl or heterocycloalkyl;
所述的R 4各自独立地选自卤素、氰基、硝基、羟基、烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基、SR'、SOR'、SO 2R'、-NHSO 2R'、SO 2NR'(R”)、NR'(R”)、COR'、-NHCOR'、COOR'、CONR'(R”)、-(P=O)R'(R”)、 或氧代基,所述的烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基任选被一个或多个各自独立地选自R 4A的取代基取代; The R4 are each independently selected from halogen, cyano, nitro, hydroxyl, alkenyl, alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocyclic Alkyl, cycloalkyloxy, heterocycloalkyloxy, aryl, heteroaryl, SR', SOR', SO 2 R', -NHSO 2 R', SO 2 NR'(R"), NR '(R"), COR', -NHCOR', COOR', CONR'(R"), -(P=O)R'(R"), Or oxo, the alkenyl, alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkane Oxygen, aryl, heteroaryl are optionally substituted by one or more substituents independently selected from R 4A ;
或者,两个相邻的R 4与环A一起形成任选被一个或多个独立地选自R 4A取代 的 Alternatively, two adjacent R 4 together form ring A optionally substituted by one or more independently selected from R 4A
所述的R 4A选自卤素、氰基、硝基、羟基、烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基、SR'、SOR'、SO 2R'、-NHSO 2R'、SO 2NR'(R”)、NR'(R”)、COR'、-NHCOR'、COOR'、CONR'(R”)、-(P=O)R'(R”)或 所述的烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基任选被一个或者多个R 8取代; Described R 4A is selected from halogen, cyano, nitro, hydroxyl, alkenyl, alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, Cycloalkyloxy, heterocycloalkyloxy, aryl, heteroaryl, SR', SOR', SO 2 R', -NHSO 2 R', SO 2 NR'(R"), NR'(R "), COR', -NHCOR', COOR', CONR'(R"), -(P=O)R'(R"), or The alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, aryl, heteroaryl are any is selected to be replaced by one or more R 8 ;
所述X独立地选自O或S;Said X is independently selected from O or S;
所述的R 5选自卤素、羟基、烷基、氰基、烷氧基、卤代烷基、卤代烷氧基、羟烷基或NR'(R”); The R 5 is selected from halogen, hydroxyl, alkyl, cyano, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl or NR'(R");
所述R 6和R 7各自独立地选自氢、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基或杂环烷基; The R and R are each independently selected from hydrogen, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl or heterocycloalkyl;
所述的R 8选自卤素、羟基、氰基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基或NR'(R”); The R is selected from halogen, hydroxyl, cyano, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl or NR'(R");
所述的R'和R”各自独立地选自H或烷基;The R' and R" are each independently selected from H or alkyl;
所述的n选自1、2、3、4、5。Said n is selected from 1, 2, 3, 4, 5.
可选的实施方案中,本公开中的式I所示的化合物或其可药用的盐,其为式II所示化合物或其可药用的盐,In an optional embodiment, the compound represented by formula I or a pharmaceutically acceptable salt thereof in the present disclosure is a compound represented by formula II or a pharmaceutically acceptable salt thereof,
其中,所述的R 1、R 2、R 3、R 4、n分别如式I所示的化合物中定义。 Wherein, the R 1 , R 2 , R 3 , R 4 , and n are respectively as defined in the compound represented by formula I.
可选的实施方案中,本公开中的式I所示的化合物或其可药用的盐,其为式III所示化合物或其可药用的盐,In an optional embodiment, the compound represented by formula I or a pharmaceutically acceptable salt thereof in the present disclosure is a compound represented by formula III or a pharmaceutically acceptable salt thereof,
其中,所述的R 1、R 2、R 4、n分别如式I所示的化合物中定义。 Wherein, the R 1 , R 2 , R 4 , and n are respectively as defined in the compound represented by formula I.
可选的实施方案中,本公开中的式I所示的化合物或其可药用的盐,其为式IV所示化合物或其可药用的盐,In an optional embodiment, the compound represented by formula I or a pharmaceutically acceptable salt thereof in the present disclosure is a compound represented by formula IV or a pharmaceutically acceptable salt thereof,
其中,所述的R 1、R 2、R 4、n分别如式I所示的化合物中定义。 Wherein, the R 1 , R 2 , R 4 , and n are respectively as defined in the compound represented by formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A选自苯基、五元含氮杂芳基、六元含氮杂芳基。In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from phenyl, five-membered nitrogen-containing heteroaryl, six-membered nitrogen-containing heteroaryl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A选自吡啶基、嘧啶基、哌啶基、咪唑基、吡咯基、吡唑基。In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from pyridyl, pyrimidinyl, piperidinyl, imidazolyl, Pyrrolyl, pyrazolyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A选自吡啶基。In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from pyridyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A选自苯基。In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein ring A is selected from phenyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为I-1, 所述G 1、G 2、G 3、G 4和G 5各自独立地选自CR 4或N,其中,G 1、G 2、G 3、G 4和G 5中至少有一个为CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein Ring A is I-1, The G 1 , G 2 , G 3 , G 4 and G 5 are each independently selected from CR 4 or N, wherein at least one of G 1 , G 2 , G 3 , G 4 and G 5 is CR 4 , The R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为I-2, 所述G 1、G 2、G 3和G 4各自独立地选自CR 4或N或NR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein Ring A is I-2, The G 1 , G 2 , G 3 and G 4 are each independently selected from CR 4 or N or NR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 2、G 3、G 4和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 3 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 1、G 3、G 4和G 5各自独立地选自CR 4,所 述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 1 , G 3 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound represented by formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 1、G 2、G 4和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 1 , G 2 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 3、G 4和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 3 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 2、G 4和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 4 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 2、G 3和G 4各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 3 and G 4 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 1、G 3和G 4各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 1 , G 3 and G 4 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 2、G 3和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 2 , G 3 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用 的盐,其中,环A为 所述G 1、G 3和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 1 , G 3 and G 5 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 3和G 4各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 3 and G 4 are each independently selected from CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 4为CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 4 is CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述G 3为CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is The G 3 is CR 4 , and the R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,G 1、G 2、G 3、G 4和G 5各自独立地选自CR 4。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein G 1 , G 2 , G 3 , G 4 and G 5 are each independently selected from CR4 .
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 1、G 2、G 3、G 4和G 5选自CR 4,所述 选自 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 2 , G 3 , G 4 and G 5 are selected from CR 4 , the selected from
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,所述 为 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, the for
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用 的盐,其中,环A为 G 2、G 3、G 4和G 5各自独立地选自CR 4。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 3 , G 4 and G 5 are each independently selected from CR 4 .
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 所述 选自 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is said selected from
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 1、G 3、G 4和G 5各自独立地选自CR 4。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 3 , G 4 and G 5 are each independently selected from CR 4 .
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 2、G 4和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 4 and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound represented by formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 3、G 4和G 5各自独立地选自CR 4。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 3 , G 4 and G 5 are each independently selected from CR 4 .
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用 的盐,其中,环A为 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 2、G 4和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 4 and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound represented by formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 2、G 3、G 4各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 3 , and G 4 are each independently selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 1、G 3、G 4各自独立地选自CR 4。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 3 , and G 4 are each independently selected from CR 4 .
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A选自 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein ring A is selected from
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 2、G 3、G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 2 , G 3 , and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 1、G 3和G 5各自独立地选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 1 , G 3 and G 5 are each independently selected from CR 4 , and R 4 is as defined in the compound represented by formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 3、G 4各自独立地选自CR 4,所述R 4如式I所 示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 3 and G 4 are each independently selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 4选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 4 is selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,环A为 G 3选自CR 4,所述R 4如式I所示化合物中定义。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein Ring A is G 3 is selected from CR 4 , and R 4 is as defined in the compound shown in formula I.
可选的实施方案中,两个相邻的R 4与环A一起形成任选被一个或多个R 4A取代的 其中环A的定义如本文任一方案所述。 In an alternative embodiment, two adjacent R 4 together with ring A form an optionally substituted by one or more R 4A Wherein ring A is defined as described in any scheme herein.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 1选自氢、烷基、卤代烷基、羟烷基、环烷基或杂环烷基。 In an optional embodiment, the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein said R is selected from hydrogen, alkyl, haloalkyl, hydroxyalkane group, cycloalkyl or heterocycloalkyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 1选自氢、C 1-6烷基、C 1-6卤代烷基、C 1-6羟烷基、3至7元的环烷基或3-7元的杂环烷基。 In an optional embodiment, the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein, said R 1 is selected from hydrogen, C 1-6 alkyl, C 1-6 haloalkyl, C 1-6 hydroxyalkyl, 3-7 membered cycloalkyl or 3-7 membered heterocycloalkyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 1选自氢、C 1-3烷基或C 1-3卤代烷基。 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein said R 1 is selected from hydrogen, C 1-3 alkyl or C 1-3 haloalkyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 1为氢。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 1 is hydrogen.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 2选自烷基、卤代烷基、羟烷基、环烷基或杂环烷基,所述的烷基、卤代烷基、羟烷基、环烷基或杂环烷基任选被一个或多个R 1A取代,所述的R 1A选自卤烷基、烷氧基、卤代烷基、卤代烷氧基、杂环烷基或NR'(R”),所述的烷基、烷氧基、卤代烷基、卤代烷氧基、杂环烷基任选被一个或者多个R 5取代。 In an optional embodiment, the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein, said R 2 is selected from the group consisting of alkyl, haloalkyl, hydroxyalkyl, Cycloalkyl or heterocycloalkyl, said alkyl, haloalkyl, hydroxyalkyl, cycloalkyl or heterocycloalkyl are optionally substituted by one or more R 1A , said R 1A is selected from haloalkane group, alkoxy, haloalkyl, haloalkoxy, heterocycloalkyl or NR'(R"), the said alkyl, alkoxy, haloalkyl, haloalkoxy, heterocycloalkyl are optionally replaced by one Or multiple R 5 substitutions.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 2选自C 1-6烷基、C 1-6卤代烷基、C 1-6羟烷基、3至7元的环烷基或3至7元的杂环烷基,所述的C 1-6烷基、C 1-6卤代烷基、C 1-6羟烷基、3至7元的环烷基或3至7元的杂环烷基任选被一个或多个R 1A取代;所述的R 1A选自卤素、C 1-6烷基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 2 is selected from C 1-6 alkyl, C 1- 6 haloalkyl, C 1-6 hydroxyalkyl, 3 to 7 membered cycloalkyl or 3 to 7 membered heterocycloalkyl, said C 1-6 alkyl, C 1-6 haloalkyl, C 1 -6 hydroxyalkyl, 3 to 7-membered cycloalkyl or 3 to 7-membered heterocycloalkyl are optionally substituted by one or more R 1A ; said R 1A is selected from halogen, C 1-6 alkyl .
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 2选自甲基、乙基、异丙基、叔丁基、环丙基、环丁基、环戊基,所述的环丙基、环丁基、环戊基被一个或多个R 1A取代,所述的R 1A选自甲 基、乙基、异丙基。 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein said R 2 is selected from methyl, ethyl, isopropyl, Tert-butyl, cyclopropyl, cyclobutyl, cyclopentyl, described cyclopropyl, cyclobutyl, cyclopentyl are substituted by one or more R 1A , described R 1A is selected from methyl, ethyl base, isopropyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 2选自异丙基或甲基环丙基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 2 is selected from isopropyl or methylcyclopropyl.
可选的实施方案中,本公开提供的式I、II所示的化合物或其可药用的盐,其中,所述的R 3选自 其中X为O,所述R 6和R 7各自独立地选自氢、C 1-6烷基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I and II or pharmaceutically acceptable salts thereof, wherein said R is selected from Wherein X is O, the R 6 and R 7 are each independently selected from hydrogen, C 1-6 alkyl.
可选的实施方案中,本公开提供的式I、II所示的化合物或其可药用的盐,其中,所述的R 3选自 其中X为O,所述R 6和R 7各自独立地选自氢、甲基、乙基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I and II or pharmaceutically acceptable salts thereof, wherein said R is selected from Wherein X is O, and the R 6 and R 7 are each independently selected from hydrogen, methyl, ethyl.
可选的实施方案中,本公开提供的式I、II所示的化合物或其可药用的盐,其中,所述的R 3选自 In an optional embodiment, the present disclosure provides compounds represented by formulas I and II or pharmaceutically acceptable salts thereof, wherein said R is selected from
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,R 4选自卤素、氰基、硝基、羟基、烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基、SR'、SOR'、SO 2R'、-NHSO 2R'、SO 2NR'(R”)、NR'(R”)、COR'、-NHCOR'、COOR'、CONR'(R”)、-(P=O)R'(R”)或 所述的烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基任选被一个或多个R 4A取代,其中,R 4A如式I所示的化合物中定义。 In an optional embodiment, the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein R 4 is selected from halogen, cyano, nitro, hydroxyl, alkenyl, Alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, aryl, heteroaryl, SR', SOR', SO 2 R', -NHSO 2 R', SO 2 NR'(R"), NR'(R"), COR', -NHCOR', COOR', CONR'(R"), -(P=O)R'(R") or The alkenyl, alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, aromatic The radical, heteroaryl is optionally substituted by one or more R 4A , wherein R 4A is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,R 4选自卤素、氰基、硝基、羟基、烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基、SR'、SOR'、SO 2R'、-NHSO 2R'、SO 2NR'(R”)、NR'(R”)、COR'、-NHCOR'、COOR'、CONR'(R”)、-(P=O)R'(R”)、 或氧代基,所述的烯基、炔基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、芳基、杂芳基任选被一个或多个R 4A取代,其中,R 4A如式I所示的化合物中定义。 In an optional embodiment, the present disclosure provides a compound of formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein R 4 is selected from halogen, cyano, nitro, hydroxyl, alkenyl, Alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, aryl, heteroaryl, SR', SOR', SO 2 R', -NHSO 2 R', SO 2 NR'(R"), NR'(R"), COR', -NHCOR', COOR', CONR'(R"), -(P=O)R'(R"), Or oxo, the alkenyl, alkynyl, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkane Baseoxy, aryl, heteroaryl are optionally substituted by one or more R 4A , wherein R 4A is as defined in the compound shown in formula I.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,R 4为氧代基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein R 4 is oxo.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述R 4各自独立地选自氢、氰基、卤素、羟基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、杂芳基、SO 2R'、-NHSO 2R'、COR'、-NHCOR'、COOR'、CONR'(R”)、或 所述的烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、杂芳基任选被一个或多个R 4A取代; In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of the R 4 is independently selected from hydrogen, cyano, halogen, hydroxyl , alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy, heteroaryl, SO 2 R', -NHSO 2 R', COR', -NHCOR', COOR', CONR'(R"), or The alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, heteroaryl are optionally substituted by one or more R 4A ;
所述的R 4A选自卤素、氰基、烷基、烷氧基、卤代烷基、卤代烷氧基、羟烷基、环烷基、杂环烷基、环烷基氧基、杂环烷基氧基、SO 2R'、-NHSO 2R'、COR'、-NHCOR'、COOR'、CONR'(R”)或 Described R 4A is selected from halogen, cyano, alkyl, alkoxy, haloalkyl, haloalkoxy, hydroxyalkyl, cycloalkyl, heterocycloalkyl, cycloalkyloxy, heterocycloalkyloxy base, SO 2 R', -NHSO 2 R', COR', -NHCOR', COOR', CONR'(R") or
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,各个R 4各自独立地选自氢、氰基或卤素。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein each R 4 is independently selected from hydrogen, cyano or halogen.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述R 4各自独立地选自氢、COR'、-NHSO 2R'、SO 2R'或 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of the R 4 is independently selected from hydrogen, COR', -NHSO 2 R', SO 2 R' or
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述R 4各自独立地选自氢、烷基、羟烷基、所述烷基任选被一个或者多个R 4A取代,所述的R 4A选自卤素、氰基、烷氧基、SO 2R'或 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of said R 4 is independently selected from hydrogen, alkyl, hydroxyalkyl , the alkyl is optionally substituted by one or more R 4A , the R 4A is selected from halogen, cyano, alkoxy, SO 2 R' or
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述R 4各自独立地选自氢、烷氧基、杂环烷基氧基,或各个R 4各自独立地选自C 1-6烷氧基或3至7元的杂环烷基氧基。 In an optional embodiment, the present disclosure provides a compound represented by formula I, II, III, IV or a pharmaceutically acceptable salt thereof, wherein each of the R 4 is independently selected from hydrogen, alkoxy, heterocyclic Alkyloxy, or each R 4 is independently selected from C 1-6 alkoxy or 3 to 7 membered heterocycloalkyloxy.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 1为氢。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 1 is hydrogen.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的R 2选自异丙基或甲基环丙基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, IV or pharmaceutically acceptable salts thereof, wherein said R 2 is selected from isopropyl or methylcyclopropyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,所述的环A为吡啶基,所述R 4至少有一个为氰基。 In an optional embodiment, the present disclosure provides compounds represented by formulas I, II, III, and IV or pharmaceutically acceptable salts thereof, wherein the ring A is pyridyl, and at least one of the R 4 is cyano.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,R'和R”各自独立地选自氢或者C 1-6烷基。 In an optional embodiment, the compound shown in the formula I, II, III, IV provided by the present disclosure or a pharmaceutically acceptable salt thereof, wherein, in an optional embodiment, the compound of the formula I, II, III provided in the present disclosure , the compound shown in IV or a pharmaceutically acceptable salt thereof, wherein R' and R" are each independently selected from hydrogen or C 1-6 alkyl.
可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,可选的实施方案中,本公开提供的式I、II、III、IV所示的化合物或其可药用的盐,其中,R'和R”各自独立地选自氢或甲基、乙基。In an optional embodiment, the compound shown in the formula I, II, III, IV provided by the present disclosure or a pharmaceutically acceptable salt thereof, wherein, in an optional embodiment, the compound of the formula I, II, III provided in the present disclosure , the compound shown in IV or a pharmaceutically acceptable salt thereof, wherein R' and R" are each independently selected from hydrogen or methyl and ethyl.
本公开提供的化合物或其可药用的盐,其选自:The compound provided by the present disclosure or a pharmaceutically acceptable salt thereof is selected from:
本公开还提供上述化合物的同位素取代物。The present disclosure also provides isotopic substitutions of the compounds described above.
一些实施方案中,所述的同位素取代物为氘原子取代。In some embodiments, the isotope substitution is deuterium atom substitution.
本公开还提供一种药物组合物,其包含前述式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物和至少一种药学上可接受的载体、稀释剂或者赋形剂。在一些实施方案中,所述的药物组合物的单位剂量为0.001mg-1000mg。The present disclosure also provides a pharmaceutical composition, which comprises the compound represented by the aforementioned formula I, II, III, IV or its pharmaceutically acceptable salt or its isotope substitution and at least one pharmaceutically acceptable carrier and diluent or excipients. In some embodiments, the unit dose of the pharmaceutical composition is 0.001 mg-1000 mg.
在某些实施方案中,基于组合物的总重量,所述的药物组合物含有0.01-99.99%的前述式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物。在某些实施方案中,所述的药物组合物含有0.1-99.9%的前述式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物。在某些实施方案中,所述的药物组合物含有0.5%-99.5%的前述式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物。在某些实施方案中,所述的药物组合物含有1%-99%的前述式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物。在某些实施方案中,所述的药物组合物含有2%-98%的前述式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物。In certain embodiments, based on the total weight of the composition, the pharmaceutical composition contains 0.01-99.99% of the compound represented by the aforementioned formula I, II, III, IV or a pharmaceutically acceptable salt thereof or an isotope substitution thing. In certain embodiments, the pharmaceutical composition contains 0.1-99.9% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof. In some embodiments, the pharmaceutical composition contains 0.5%-99.5% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof. In certain embodiments, the pharmaceutical composition contains 1%-99% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof. In certain embodiments, the pharmaceutical composition contains 2%-98% of the compound represented by the aforementioned formulas I, II, III, IV or pharmaceutically acceptable salts or isotope substitutions thereof.
在某些实施方案中,基于组合物的总重量,所述的药物组合物含有0.01%-99.99%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有0.1%-99.9%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有0.5%-99.5%的药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有1%-99%的 药学上可接受的赋形剂。在某些实施方案中,所述的药物组合物含有2%-98%的药学上可接受的赋形剂。In certain embodiments, the pharmaceutical composition contains 0.01%-99.99% of pharmaceutically acceptable excipients based on the total weight of the composition. In certain embodiments, the pharmaceutical composition contains 0.1%-99.9% of pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical composition contains 0.5%-99.5% of pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical composition contains 1%-99% of pharmaceutically acceptable excipients. In certain embodiments, the pharmaceutical composition contains 2%-98% of pharmaceutically acceptable excipients.
本公开还提供一种预防和/或治疗患有与蛋白依赖性激酶相关疾病的患者的方法,其通过向所述患者施用治疗有效量的本公开所述的式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物或前述药物组合物。The present disclosure also provides a method for preventing and/or treating a patient suffering from a protein-dependent kinase-related disease, by administering to the patient a therapeutically effective amount of the formulas I, II, III, and IV described in the present disclosure. The compound shown or its pharmaceutically acceptable salt or its isotope substitution or the aforementioned pharmaceutical composition.
本公开还提供一种预防和/或治疗患有与细胞周期蛋白相关疾病的患者的方法,其通过向所述患者施用治疗有效量的本公开所述的式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物或前述药物组合物。The present disclosure also provides a method for preventing and/or treating a patient suffering from a cyclin-related disease, by administering to the patient a therapeutically effective amount of the compounds represented by formulas I, II, III, and IV described in the present disclosure. The compound or its pharmaceutically acceptable salt or its isotopic substitution or the aforementioned pharmaceutical composition.
在一些实施方案中,所述与蛋白依赖性激酶相关疾病或与细胞周期蛋白相关疾病选细胞增殖性疾病,癌症或免疫性疾病。In some embodiments, the protein-dependent kinase-related disease or cyclin-related disease is selected from cell proliferative disease, cancer or immune disease.
在一些实施方案中,所述与蛋白依赖性激酶相关疾病或与细胞周期蛋白相关疾病选自乳腺癌、卵巢癌、前列腺癌、黑色素瘤、脑瘤、食道癌、胃癌、肝癌、胰腺癌、结直肠癌、肺癌、肾癌、皮肤癌、成胶质细胞瘤、神经母细胞瘤、肉瘤。In some embodiments, the protein-dependent kinase-associated disease or cyclin-associated disease is selected from breast cancer, ovarian cancer, prostate cancer, melanoma, brain tumor, esophageal cancer, gastric cancer, liver cancer, pancreatic cancer, Colorectal cancer, lung cancer, kidney cancer, skin cancer, glioblastoma, neuroblastoma, sarcoma.
一些具体的实施方案中,所述的癌症选自细胞周期蛋白E1和/或细胞周期蛋白E2扩增的癌症。In some specific embodiments, the cancer is selected from cancers in which cyclin E1 and/or cyclin E2 are amplified.
本公开还提供一种预防和/或治疗患有癌症的患者的方法,其通过向所述患者施用治疗有效量的本公开所述的式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物或前述药物组合物,所述的癌症选自乳腺癌、卵巢癌、前列腺癌、黑色素瘤、脑瘤、食道癌、胃癌、肝癌、胰腺癌、结直肠癌、肺癌、肾癌、皮肤癌、成胶质细胞瘤、神经母细胞瘤、肉瘤。The present disclosure also provides a method for preventing and/or treating a patient suffering from cancer, by administering to the patient a therapeutically effective amount of the compound represented by the formulas I, II, III, IV described in the present disclosure or its alternative Pharmaceutically acceptable salt or its isotope substitution or the aforementioned pharmaceutical composition, the cancer is selected from breast cancer, ovarian cancer, prostate cancer, melanoma, brain tumor, esophageal cancer, gastric cancer, liver cancer, pancreatic cancer, colorectal cancer, Lung cancer, kidney cancer, skin cancer, glioblastoma, neuroblastoma, sarcoma.
本公开提供了治疗有效量的本公开所述的式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物或前述药物组合物在制备用于预防和/或治疗与蛋白依赖性激酶相关疾病的药物中的用途,The present disclosure provides a therapeutically effective dose of the compounds shown in formulas I, II, III, and IV described in the present disclosure or their pharmaceutically acceptable salts or isotope substitutions or the aforementioned pharmaceutical compositions in the preparation for preventing and/or Use in medicines for the treatment of diseases associated with protein-dependent kinases,
一些具体的实施方案中,蛋白依赖性激酶选自CDK2,与蛋白依赖性激酶相关疾病选自细胞增殖性疾病,癌症或免疫性疾病。In some specific embodiments, the protein-dependent kinase is selected from CDK2, and the disease related to the protein-dependent kinase is selected from cell proliferative diseases, cancer or immune diseases.
另一方面,本公开提供了治疗有效量的本公开所述的式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物或前述药物组合物在制备用于预防和/或治疗与细胞周期蛋白相关疾病的药物中的用途。On the other hand, the present disclosure provides a therapeutically effective amount of the compound shown in formula I, II, III, IV described in the present disclosure or its pharmaceutically acceptable salt or its isotope substitution or the aforementioned pharmaceutical composition in preparation for use in Use in medicines for preventing and/or treating diseases related to cell cyclins.
一些具体的实施方案中,细胞周期蛋白选自细胞周期蛋白E,例如细胞周期蛋白E1、细胞周期蛋白E2。与细胞周期蛋白相关疾病选自细胞增殖性疾病,癌症或免疫性疾病。In some specific embodiments, the cyclin is selected from cyclin E, such as cyclin E1, cyclin E2. The cyclin-associated disease is selected from cell proliferative disease, cancer or immune disease.
在一些实施方案中,本公开提供了治疗有效量的本公开式I、II、III、IV所示的化合物或其可药用的盐或其同位素取代物或前述药物组合物在制备治疗癌症的药物中的用途。In some embodiments, the present disclosure provides a therapeutically effective amount of a compound represented by formula I, II, III, IV of the present disclosure or a pharmaceutically acceptable salt thereof or an isotope substitution thereof or the aforementioned pharmaceutical composition in the preparation of a drug for the treatment of cancer Uses in medicine.
一些具体的实施方案中,所述的癌症选自选自乳腺癌、卵巢癌、前列腺癌、黑色素瘤、脑瘤、食道癌、胃癌、肝癌、胰腺癌、结直肠癌、肺癌、肾癌、皮肤 癌、成胶质细胞瘤、神经母细胞瘤、肉瘤。In some specific embodiments, the cancer is selected from breast cancer, ovarian cancer, prostate cancer, melanoma, brain tumor, esophageal cancer, gastric cancer, liver cancer, pancreatic cancer, colorectal cancer, lung cancer, kidney cancer, skin cancer Carcinoma, glioblastoma, neuroblastoma, sarcoma.
一些具体的实施方案中,所述的癌症选自细胞周期蛋白E1和/或细胞周期蛋白E2扩增的癌症。In some specific embodiments, the cancer is selected from cancers in which cyclin E1 and/or cyclin E2 are amplified.
本公开另一方面提供一种式III所示化合物或其可药用的盐、同位素取代物的制备方法,其包括式V所示化合物在酸性条件下脱除氨基保护基PG的步骤,Another aspect of the present disclosure provides a preparation method of the compound represented by formula III or its pharmaceutically acceptable salts and isotope substitutions, which includes the step of removing the amino protecting group PG from the compound represented by formula V under acidic conditions,
其中,R 1、R 2、R 4、n分别如式III所示化合物中所定义,PG为氨基保护基,可选为叔丁基、乙酰基、三氟乙酰基、三苯甲基、苄基、甲酰基。 Among them, R 1 , R 2 , R 4 , and n are respectively as defined in the compound shown in formula III, and PG is an amino protecting group, which can be tert-butyl, acetyl, trifluoroacetyl, trityl, benzyl base, formyl.
本公开另一方面提供一种式V所示的化合物,Another aspect of the present disclosure provides a compound represented by formula V,
其中,R 1、R 2、R 4、n分别如式III所示化合物中所定义,PG为氨基保护基,可选为叔丁基、乙酰基、三氟乙酰基、三苯甲基、苄基、甲酰基。 Among them, R 1 , R 2 , R 4 , and n are respectively as defined in the compound shown in formula III, and PG is an amino protecting group, which can be tert-butyl, acetyl, trifluoroacetyl, trityl, benzyl base, formyl.
本公开中所述化合物可药用盐选自无机盐或有机盐,本公开所述化合物可与酸性或碱性物质反应成相应盐。The pharmaceutically acceptable salts of the compounds described in the present disclosure are selected from inorganic salts or organic salts, and the compounds described in the present disclosure can react with acidic or basic substances to form corresponding salts.
本公开化合物对细胞周期蛋白-依赖性激酶具有很好的抑制作用,对CDK2/Cyclin E的抑制活性的IC 50值在0.01至1000nM,某些化合物对CDK2/Cyclin E的抑制活性的IC 50值在0.01至500nM,某些化合物对CDK2/Cyclin E的抑制活性的IC 50值在0.01至300nM,某些化合物对CDK2/Cyclin E的抑制活性的IC 50值在0.01至200nM,某些化合物对CDK2/Cyclin E的抑制活性的IC 50值在0.01至100nM,某些化合物对CDK2/Cyclin E的抑制活性的IC50值<100nM。 The disclosed compound has a good inhibitory effect on cell cycle protein-dependent kinase, and the IC 50 value of the inhibitory activity to CDK2/Cyclin E is 0.01 to 1000nM, and the IC 50 value of the inhibitory activity of some compounds on CDK2/Cyclin E In the range of 0.01 to 500nM, the IC 50 values of the inhibitory activity of some compounds on CDK2/Cyclin E are in the range of 0.01 to 300nM, and the IC 50 values of the inhibitory activity of some compounds on CDK2/Cyclin E are in the range of 0.01 to 200nM. The IC50 value of the inhibitory activity of CDK2/Cyclin E is between 0.01 and 100nM, and the IC50 value of the inhibitory activity of some compounds against CDK2/Cyclin E is <100nM.
一些实施方案提供的化合物对CDK2/Cyclin E抑制活性达到<50nM。另一些实施方案提供的化合物对CDK2/Cyclin E抑制活性达到<20nM。另一些实施方案提供的化合物对CDK2/Cyclin E抑制活性达到<10nM。另一些实施方案提供的化合物对CDK2/Cyclin E抑制活性达到<5nM。另一些实施方案提供的化合物对CDK2/Cyclin E抑制活性达到<1nM。Some embodiments provide compounds that achieve <50 nM inhibitory activity against CDK2/Cyclin E. Other embodiments provide compounds that achieve <20 nM inhibitory activity against CDK2/Cyclin E. Other embodiments provide compounds that have <10 nM inhibitory activity against CDK2/Cyclin E. Other embodiments provide compounds that achieve <5nM inhibitory activity on CDK2/Cyclin E. Other embodiments provide compounds that achieve <1 nM inhibitory activity against CDK2/Cyclin E.
另一方面,本公开化合物对CDK2/Cyclin A也有较好的抑制效果。一些实施方案提供的化合物对CDK2/Cyclin A抑制活性达到<500nM。另一些实施方案提供的化合物对CDK2/Cyclin A抑制活性达到<200nM。另一些实施方案提供的化合物对CDK2/Cyclin A抑制活性达到<150nM。另一些实施方案提供的化合物对CDK2/Cyclin A抑制活性达到<100nM。另一些实施方案提供的化合物对 CDK2/Cyclin A抑制活性达到<50nM。另一些实施方案提供的化合物对CDK2/Cyclin A抑制活性达到<20nM。另一些实施方案提供的化合物对CDK2/Cyclin A抑制活性达到<10nM。On the other hand, the disclosed compound also has a good inhibitory effect on CDK2/Cyclin A. Some embodiments provide compounds that achieve <500 nM inhibitory activity against CDK2/Cyclin A. Other embodiments provide compounds that achieve <200 nM inhibitory activity against CDK2/Cyclin A. Other embodiments provide compounds that achieve <150 nM inhibitory activity against CDK2/Cyclin A. Other embodiments provide compounds that achieve <100 nM inhibitory activity against CDK2/Cyclin A. Other embodiments provide compounds that have CDK2/Cyclin A inhibitory activity <50 nM. Other embodiments provide compounds that achieve <20 nM inhibitory activity against CDK2/Cyclin A. Other embodiments provide compounds that achieve <10 nM inhibitory activity against CDK2/Cyclin A.
在另一些实施方案中,本公开化合物对其他激酶如CDK1、CDK4、CDK5、CDK7、CDK9或GSK3β抑制活性偏弱,具有CDK2特异性选择。In other embodiments, the compounds of the present disclosure have weak inhibitory activity on other kinases such as CDK1, CDK4, CDK5, CDK7, CDK9 or GSK3β, and have CDK2-specific selection.
另一方面,本公开化合物可以存在特定的几何或立体异构体形式。本公开设想所有的这类化合物,包括顺式和反式异构体、(-)-和(+)-对对映体、(R)-和(S)-对映体、非对映异构体、(D)-异构体、(L)-异构体,及其外消旋混合物和其他混合物,例如对映异构体或非对映体富集的混合物,所有这些混合物都属于本公开的范围之内。烷基等取代基中可存在另外的不对称碳原子。所有这些异构体以及它们的混合物,均包括在本公开的范围之内。On the other hand, the disclosed compounds may exist in particular geometric or stereoisomeric forms. This disclosure contemplates all such compounds, including cis and trans isomers, (-)- and (+)-enantiomers, (R)- and (S)-enantiomers, diastereomers isomers, (D)-isomers, (L)-isomers, and their racemic and other mixtures, such as enantiomerically or diastereomerically enriched mixtures, all of which are within the scope of this disclosure. Additional asymmetric carbon atoms may be present in substituents such as alkyl groups. All such isomers, as well as mixtures thereof, are included within the scope of this disclosure.
另外,本公开的化合物和中间体还可以以不同的互变异构体形式存在,并且所有这样的形式包含于本公开的范围内。术语“互变异构体”或“互变异构体形式”是指可经由低能垒互变的不同能量的结构异构体。例如,质子互变异构体(也称为质子转移互变异构体)包括经由质子迁移的互变,如酮-烯醇及亚胺-烯胺、内酰胺-内酰亚胺、吡唑基异构化。In addition, the compounds and intermediates of the present disclosure may also exist in different tautomeric forms, and all such forms are included within the scope of the present disclosure. The term "tautomer" or "tautomeric form" refers to structural isomers of different energies that can interconvert via a low energy barrier. For example, proton tautomers (also known as prototropic tautomers) include interconversions via migration of a proton, such as keto-enol and imine-enamine, lactam-lactam, pyrazole base isomerization.
吡唑基平衡实例是在如下所示的E和F之间:An example of a pyrazolyl equilibrium is between E and F as shown below:
具体到本申请:Specific to this application:
所有的互变异构形式在本发明的范围内。化合物的命名不排除任何互变异构体。All tautomeric forms are within the scope of the invention. The naming of compounds does not exclude any tautomers.
本公开化合物可以是不对称的,例如,具有一个或多个立体异构体。除非另有说明,所有立体异构体都包括,如对映异构体和非对映异构体。本公开的含有不对称碳原子的化合物可以以光学活性纯的形式或外消旋形式被分离出来。光学活性纯的形式可以从外消旋混合物拆分,或通过使用手性原料或手性试剂合成。Compounds of the present disclosure may be asymmetric, eg, possess one or more stereoisomers. Unless otherwise stated, all stereoisomers are included, such as enantiomers and diastereomers. Compounds of the present disclosure containing asymmetric carbon atoms can be isolated in optically pure or racemic forms. Optically pure forms can be resolved from racemic mixtures or synthesized by using chiral starting materials or reagents.
可以通过的手性合成或手性试剂或者其他常规技术制备光学活性的(R)-和(S)-异构体以及D和L异构体。如果想得到本公开某化合物的一种对映体,可以通过不对称合成或者具有手性助剂的衍生作用来制备,其中将所得非对映体混合物分离,并且辅助基团裂开以提供纯的所需对映异构体。或者,当分子中含有碱性官能团(如氨基)或酸性官能团(如羧基)时,与适当的光学活性的酸或碱形成非对映异 构体的盐,然后通过本领域所公知的常规方法进行非对映异构体拆分,然后回收得到纯的对映体。此外,对映异构体和非对映异构体的分离通常是通过使用色谱法完成的,所述色谱法采用手性固定相,并任选地与化学衍生法相结合(例如由胺生成氨基甲酸盐)。Optically active (R)- and (S)-isomers as well as D and L-isomers can be prepared by chiral synthesis or chiral reagents or other conventional techniques. If one enantiomer of a compound of the present disclosure is desired, it can be prepared by asymmetric synthesis or derivatization with chiral auxiliary agents, wherein the resulting diastereomeric mixture is separated and the auxiliary group is cleaved to provide pure desired enantiomer. Alternatively, when the molecule contains a basic functional group (such as an amino group) or an acidic functional group (such as a carboxyl group), a diastereoisomeric salt is formed with an appropriate optically active acid or base, and then a diastereomeric salt is formed by a conventional method known in the art. Diastereomeric resolution is performed and the pure enantiomers are recovered. Furthermore, the separation of enantiomers and diastereomers is usually accomplished by the use of chromatography using chiral stationary phases, optionally in combination with chemical derivatization methods (e.g. amines to amino groups formate).
本公开还包括一些与本文中记载的那些相同的,但一个或多个原子被原子量或质量数不同于自然中通常发现的原子量或质量数的原子置换的同位素标记的本公开化合物。可结合到本公开化合物的同位素的实例包括氢、碳、氮、氧、磷、硫、氟、碘和氯的同位素,诸如分别为 2H、 3H、 11C、 13C、 14C、 13N、 15N、 15O、 17O、 18O、 31P、 32P、 35S、 18F、 123I、 125I和 36Cl等。 The present disclosure also includes certain isotopically labeled compounds of the disclosure that are identical to those described herein, but wherein one or more atoms are replaced by an atom of an atomic mass or mass number different from that normally found in nature. Examples of isotopes that can be incorporated into compounds of the present disclosure include isotopes of hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, iodine, and chlorine, such as 2 H, 3 H, 11 C, 13 C, 14 C, 13 N, 15 N, 15 O, 17 O, 18 O, 31 P, 32 P, 35 S, 18 F, 123 I, 125 I and 36 Cl, etc.
除另有说明,当一个位置被特别地指定为氘(D)时,该位置应理解为具有大于氘的天然丰度(其为0.015%)至少1000倍的丰度的氘(即,至少10%的氘掺入)。示例中化合物的具有大于氘的天然丰度可以是至少1000倍的丰度的氘、至少2000倍的丰度的氘、至少3000倍的丰度的氘、至少4000倍的丰度的氘、至少5000倍的丰度的氘、至少6000倍的丰度的氘或更高丰度的氘。本公开还包括各种氘化形式的式I化合物。与碳原子连接的各个可用的氢原子可独立地被氘原子替换。本领域技术人员能够参考相关文献合成氘化形式的式I化合物。在制备氘代形式的式I化合物时可使用市售的氘代起始物质,或它们可使用常规技术采用氘代试剂合成,氘代试剂包括但不限于氘代硼烷、三氘代硼烷四氢呋喃溶液、氘代氢化锂铝、氘代碘乙烷和氘代碘甲烷等。Unless otherwise stated, when a position is specifically designated as deuterium (D), the position is understood to have an abundance of deuterium (i.e., at least 10 % deuterium incorporation). Exemplary compounds having a natural abundance greater than deuterium can be at least 1000 times more abundant deuterium, at least 2000 times more abundant deuterium, at least 3000 times more abundant deuterium, at least 4000 times more abundant deuterium, at least 5000 times more abundant deuterium, at least 6000 times more abundant deuterium, or more abundant deuterium. The present disclosure also includes various deuterated forms of compounds of formula I. Each available hydrogen atom attached to a carbon atom can be independently replaced by a deuterium atom. Those skilled in the art can refer to the relevant literature to synthesize the deuterated form of the compound of formula I. Commercially available deuterated starting materials can be used in the preparation of deuterated forms of compounds of formula I, or they can be synthesized using conventional techniques using deuterated reagents, including but not limited to deuterated borane, trideuterated borane Tetrahydrofuran solution, deuterated lithium aluminum hydride, deuterated ethyl iodide and deuterated methyl iodide, etc.
“任选地”或“任选”是指意味着随后所描述的事件或环境可以但不必发生,该说明包括该事件或环境发生或不发生的场合。例如“任选的被卤素或者氰基取代的C 1-6烷基”是指卤素或者氰基可以但不必须存在,该说明包括烷基被卤素或者氰基取代的情形和烷基不被卤素和氰基取代的情形。 "Optionally" or "optionally" means that the subsequently described event or circumstance can but need not occur, and that the description includes instances where the event or circumstance occurs or does not occur. For example, "C 1-6 alkyl optionally substituted by halogen or cyano" means that halogen or cyano may but not necessarily exist, and this description includes the case where the alkyl is substituted by halogen or cyano and the alkyl is not substituted by halogen And the case of cyano substitution.
本发明所述化合物的化学结构中,键 表示未指定构型,即如果化学结构中存在手性异构体,键 可以为 或 或者同时包含 和 两种构型。虽然为简便起见将全部上述结构式画成某些异构体形式,但是本发明可以包括所有的异构体,如互变异构体、旋转异构体、几何异构体、非对映异构体、外消旋体和对映异构体。 In the chemical structure of the compound of the present invention, the bond Indicates unassigned configuration, i.e. if chiral isomers exist in the chemical structure, the bond can be or or both and Two configurations. Although all of the above formulas are drawn as certain isomeric forms for simplicity, the present invention may include all isomers such as tautomers, rotamers, geometric isomers, diastereoisomers body, racemate and enantiomer.
本公开所述化合物的化学结构中,键 并未指定构型,即可以为Z构型或E构型,或者同时包含两种构型。 In the chemical structures of the compounds described in this disclosure, the bond If the configuration is not specified, it can be Z configuration or E configuration, or both configurations.
术语解释Terminology Explanation
“药物组合物”表示含有一种或多种本文所述化合物或其生理学上可药用的盐或前体药物与其他化学组分的混合物,以及其他组分例如生理学可药用的载体和赋形剂。药物组合物的目的是促进对生物体的给药,利于活性成分的吸收进而 发挥生物活性。"Pharmaceutical composition" means a mixture containing one or more compounds described herein, or a physiologically acceptable salt or prodrug thereof, and other chemical components, as well as other components such as physiologically acceptable carriers and excipients. Forming agent. The purpose of the pharmaceutical composition is to promote the administration to the organism, facilitate the absorption of the active ingredient and then exert the biological activity.
“可药用赋形剂”包括但不限于任何已经被美国食品和药物管理局(FDA)批准对于人类或家畜动物使用可接受的任何助剂、载体、助流剂、甜味剂、稀释剂、防腐剂、染料/着色剂、增香剂、表面活性剂、润湿剂、分散剂、助悬剂、稳定剂、等渗剂、溶剂或乳化剂。"Pharmaceutically acceptable excipients" include, but are not limited to, any adjuvants, carriers, glidants, sweeteners, diluents that have been approved by the U.S. Food and Drug Administration (FDA) to be acceptable for human or livestock use , preservative, dye/colorant, flavor enhancer, surfactant, wetting agent, dispersant, suspending agent, stabilizer, isotonic agent, solvent or emulsifier.
本公开中所述“有效量”或“有效治疗量”包含足以改善或预防医学病症的症状或病症的量。有效量还意指足以允许或促进诊断的量。用于特定患者或兽医学受试者的有效量可依据以下因素而变化:如待治疗的病症、患者的总体健康情况、给药的方法途径和剂量以及副作用严重性。有效量可以是避免显著副作用或毒性作用的最大剂量或给药方案。An "effective amount" or "therapeutically effective amount" as used in the present disclosure includes an amount sufficient to ameliorate or prevent a symptom or condition of a medical condition. An effective amount also means an amount sufficient to permit or facilitate diagnosis. Effective amounts for a particular patient or veterinary subject may vary depending on factors such as the condition being treated, the general health of the patient, the method, route and dosage of administration, and the severity of side effects. An effective amount may be the maximum dose or dosing regimen that avoids significant side effects or toxic effects.
“烷基”指饱和的脂族烃基团,包括1至20个碳原子的直链和支链基团。含有1至6个碳原子的烷基。非限制性实施例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基及其各种支链异构体等。烷基可以是取代的或未取代的,当被取代时,取代基可以在任何可使用的连接点上被取代,优选一个或多个以下基团,独立地选自卤素、羟基、氧代、氰基、氨基、C 1-6烷基、C 1-6烷氧基、3至6元环烷基或3至6元杂环烷基,所述烷基、烷氧基、环烷基或杂环烷基任选被卤素、羟基、硝基、氰基或氨基所取代。 "Alkyl" refers to a saturated aliphatic hydrocarbon group, including straight and branched chain groups of 1 to 20 carbon atoms. An alkyl group containing 1 to 6 carbon atoms. Non-limiting examples include methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 2,2-dimethylpropyl and various branched isomers, etc. Alkyl groups may be substituted or unsubstituted, and when substituted, substituents may be substituted at any available point of attachment, preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, cyano, amino, C 1-6 alkyl, C 1-6 alkoxy, 3 to 6 membered cycloalkyl or 3 to 6 membered heterocycloalkyl, the alkyl, alkoxy, cycloalkyl or Heterocycloalkyl is optionally substituted with halo, hydroxy, nitro, cyano or amino.
术语“烯基”指分子中含有至少一个碳碳双键的烷基化合物,其中烷基的定义如上所述。烯基可以是取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷氧基、卤素、卤代烷基、卤代烷氧基、羟基、羟烷基、氰基、氨基、硝基、环烷基、杂环烷基、芳基和杂芳基中的一个或多个取代基。The term "alkenyl" refers to an alkyl compound containing at least one carbon-carbon double bond in the molecule, wherein alkyl is as defined above. Alkenyl groups may be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from the group consisting of alkoxy, halo, haloalkyl, haloalkoxy, hydroxy, hydroxyalkane One or more substituents in radical, cyano, amino, nitro, cycloalkyl, heterocycloalkyl, aryl and heteroaryl.
术语“炔基”指分子中含有至少一个碳碳三键的烷基化合物,其中烷基的定义如上所述。炔基可以是取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自烷氧基、卤素、卤代烷基、卤代烷氧基、羟基、羟烷基、氰基、氨基、硝基、环烷基、杂环烷基、芳基和杂芳基中的一个或多个取代基。The term "alkynyl" refers to an alkyl compound containing at least one carbon-carbon triple bond in the molecule, wherein alkyl is as defined above. Alkynyl groups may be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from alkoxy, halo, haloalkyl, haloalkoxy, hydroxy, hydroxyalkane One or more substituents in radical, cyano, amino, nitro, cycloalkyl, heterocycloalkyl, aryl and heteroaryl.
术语“环烷基”指饱和或部分不饱和单环或多环环状烃取代基,环烷基环包含3至20个碳原子,优选包含3至6个碳原子。单环环烷基的非限制性实例包括环丙基、环丁基、环戊基、环戊烯基、环己基、环己烯基、环己二烯基等;多环环烷基包括螺环、并环和桥环的环烷基。环烷基可以是取代的或未取代的,当被取代时,取代基可以在任何可使用的连接点上被取代,优选一个或多个以下基团,独立地选自卤素、羟基、氧代、氰基、氨基、C 1-6烷基、C 1-6烷氧基、3至6元环烷基或3至6元杂环烷基,所述烷基、烷氧基、环烷基或杂环烷基任选被卤素、羟基、硝基、氰基或氨基所取代。 The term "cycloalkyl" refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent, the cycloalkyl ring containing 3 to 20 carbon atoms, preferably containing 3 to 6 carbon atoms. Non-limiting examples of monocyclic cycloalkyls include cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, etc.; multicyclic cycloalkyls include spiro Cycloalkyls of rings, parallel rings and bridged rings. Cycloalkyl groups may be substituted or unsubstituted, and when substituted, the substituents may be substituted at any available point of attachment, preferably one or more of the following groups, independently selected from halogen, hydroxy, oxo , cyano, amino, C 1-6 alkyl, C 1-6 alkoxy, 3 to 6 membered cycloalkyl or 3 to 6 membered heterocycloalkyl, the alkyl, alkoxy, cycloalkyl Or heterocycloalkyl is optionally substituted with halo, hydroxy, nitro, cyano or amino.
术语“杂环烷基(Heterocycloalkyl)”指饱和或部分不饱和单环或多环环状烃取代基,其包含3至20个环原子,其中一个或多个环原子为选自氮、氧或S(O) m(其中m是整数0至2)的杂原子,但不包括-O-O-、-O-S-或-S-S-的环部分,其余环原子为碳。优选包含3至12个环原子,其中1~4个是杂原子;更优选包含3至7个环原子。“杂环烷基”非限制性实例包括: The term "heterocycloalkyl" refers to a saturated or partially unsaturated monocyclic or polycyclic cyclic hydrocarbon substituent comprising 3 to 20 ring atoms, one or more of which are selected from nitrogen, oxygen or A heteroatom of S(O) m (where m is an integer from 0 to 2), excluding ring portions of -OO-, -OS- or -SS-, the remaining ring atoms being carbon. Preferably it contains 3 to 12 ring atoms, of which 1 to 4 are heteroatoms; more preferably it contains 3 to 7 ring atoms. Non-limiting examples of "heterocycloalkyl" include:
等等。 etc.
所述杂环烷基环可以稠合于芳基或杂芳基环上,其中与母体结构连接在一起的环为杂环烷基,其非限制性实例包括:The heterocycloalkyl ring may be fused to an aryl or heteroaryl ring, wherein the ring bonded to the parent structure is a heterocycloalkyl, non-limiting examples of which include:
等。 wait.
杂环烷基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自卤素、羟基、氧代、氰基、氨基、C 1-6烷基、C 1-6烷氧基、3至6元环烷基或3至6元杂环烷基,所述烷基、烷氧基、环烷基或杂环烷基任选被卤素、羟基、硝基、氰基或氨基所取代。 Heterocycloalkyl groups may be optionally substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, cyano, amino, C 1-6 alkyl, C 1-6 alkoxy, 3 to 6 membered cycloalkyl or 3 to 6 membered heterocycloalkyl, the alkyl, alkoxy, cycloalkyl or heterocycloalkyl optionally Substituted by halogen, hydroxy, nitro, cyano or amino.
术语“烷氧基”指-O-(烷基),其中烷基的定义如上所述。烷氧基的非限制性实例包括:甲氧基、乙氧基、丙氧基、丁氧基。烷氧基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自卤素、羟基、氧代、氰基、氨基、C 1-6烷基、C 1-6烷氧基、3至7元环烷基或3至7元杂环烷基,所述烷基、烷氧基、环烷基或杂环烷基任选被卤素、羟基、硝基、氰基或氨基所取代。 The term "alkoxy" refers to -O-(alkyl), wherein alkyl is as defined above. Non-limiting examples of alkoxy include: methoxy, ethoxy, propoxy, butoxy. Alkoxy may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, cyano, amino, C -6 alkyl, C 1-6 alkoxy, 3 to 7 membered cycloalkyl or 3 to 7 membered heterocycloalkyl, said alkyl, alkoxy, cycloalkyl or heterocycloalkyl are optionally Halogen, hydroxy, nitro, cyano or amino substituted.
同理,“环烷基氧基”、“杂环烷基氧基”同上述“烷氧基”定义,具体可以是-O-(环烷基)、-O-(杂环烷基)。Similarly, "cycloalkyloxy" and "heterocycloalkyloxy" are as defined above for "alkoxy", specifically -O-(cycloalkyl), -O-(heterocycloalkyl).
术语“烷硫基”指-S-(烷基),其中烷基的定义如上所述。烷氧基的非限制性实例包括:甲硫基、乙硫基、丙硫基、丁硫基。烷硫基可以是任选取代的或非取代 的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自C 1-6烷氧基、3至6元环烷基、3至6元杂环烷基、3至6元环烷氧基、3至6元杂环烷基氧基、C 1-6烷硫基、3至6元环烷硫基、3至6元杂环烷硫基,所述烷氧基、环烷基、杂环烷基、环烷氧基、杂环氧基、烷硫基、环烷硫基、杂环烷硫基任选被卤素、羟基、氰基或氨基所取代。 The term "alkylthio" refers to -S-(alkyl), wherein alkyl is as defined above. Non-limiting examples of alkoxy include: methylthio, ethylthio, propylthio, butylthio. Alkylthio may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups independently selected from C 1-6 alkoxy, 3 to 6 membered cycloalkane group, 3 to 6 membered heterocycloalkyl, 3 to 6 membered cycloalkoxy, 3 to 6 membered heterocycloalkyloxy, C 1-6 alkylthio, 3 to 6 membered cycloalkylthio, 3 to 6 6-membered heterocycloalkylthio, the alkoxy, cycloalkyl, heterocycloalkyl, cycloalkoxy, heterocyclyloxy, alkylthio, cycloalkylthio, heterocycloalkylthio are optionally Halogen, hydroxy, cyano or amino substituted.
同理,“环烷硫基”、“杂环烷硫基”同上述“烷硫基”定义。Similarly, "cycloalkylthio" and "heterocycloalkylthio" have the same definition as the above-mentioned "alkylthio".
“一价基团”是指一个化合物从“形式上”消除一个单价的原子或基团。“亚基”则是指化合物从“形式上”消除两个单价或一个双价形成的原子或原子团。"Monovalent group" means that a compound is "formally" eliminated from a monovalent atom or group. "Subunit" refers to a compound formed by "formally" eliminating two monovalent or one divalent atoms or atomic groups.
术语“亚烷基”表示烷烃分子中去除2个氢原子后余下的部分,包括1至20个碳原子的直链和支链亚基团。含有1至6个碳原子的亚烷基,非限制性实施例包括亚甲基(-CH 2-)、亚乙基(如-CH 2CH 2-或-CH(CH 3)-)。如无特殊说明,亚烷基可以是取代的或未取代的,当被取代时,取代基可以在任何可使用的连接点上被取代,优选一个或多个以下基团,独立地选自卤素、羟基、氰基、氨基、C 1-6烷基或C 1-6烷氧基。 The term "alkylene" means what remains of an alkane molecule after removal of 2 hydrogen atoms, including straight and branched chain subgroups of 1 to 20 carbon atoms. An alkylene group having 1 to 6 carbon atoms, non-limiting examples include methylene (-CH 2 -), ethylene (eg -CH 2 CH 2 - or -CH(CH 3 )-). Unless otherwise specified, the alkylene group may be substituted or unsubstituted, and when substituted, the substituent may be substituted at any available point of attachment, preferably one or more of the following groups, independently selected from halogen , hydroxyl, cyano, amino, C 1-6 alkyl or C 1-6 alkoxy.
同理,“亚烷氧基”、“亚烯基”、“亚烯氧基”、“亚环烷基”、“亚杂环烷基”的定义如“亚烷基”。Similarly, the definitions of "alkyleneoxy", "alkenylene", "alkenyleneoxy", "cycloalkylene", and "heterocycloalkylene" are the same as "alkylene".
术语“芳基”指具有共轭的π电子体系的6至14元全碳单环或稠合多环(也就是共享毗邻碳原子对的环)基团,优选为6至12元,例如苯基和萘基。所述芳基环可以稠合于杂芳基、杂环烷基或环烷基环上,其中与母体结构连接在一起的环为芳基环,其非限制性实例包括:The term "aryl" refers to a 6 to 14 membered all-carbon monocyclic or fused polycyclic (that is, rings sharing adjacent pairs of carbon atoms) group, preferably 6 to 12 membered, having a conjugated π-electron system, such as benzene base and naphthyl. The aryl ring may be fused to a heteroaryl, heterocycloalkyl or cycloalkyl ring, where the ring bonded to the parent structure is an aryl ring, non-limiting examples of which include:
芳基可以是取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自卤素、羟基、氧代、硝基、氰基、C 1-6烷基、C 1-6烷氧基、C 2-6烯氧基、C 2-6炔氧基、3至6元环烷氧基、3至6元杂环烷基氧基、C 3-8环烯氧基、5至6元芳基或杂芳基,所述C 1-6烷基、C 1-6烷氧基、C 2-6烯氧基、C 2-6炔氧基、3至6元环烷氧基、3至6元杂环烷基氧基、3至8元环烯氧基、5至6元芳基或杂芳基任选被一个或多个选自卤素、羟基、氰基、氨基、C 1-6烷基或C 1-6烷氧基。 Aryl groups may be substituted or unsubstituted, and when substituted, the substituents are preferably one or more of the following groups independently selected from halogen, hydroxy, oxo, nitro, cyano, C1-6 Alkyl, C 1-6 alkoxy, C 2-6 alkenyloxy, C 2-6 alkynyloxy, 3 to 6-membered cycloalkoxy, 3 to 6-membered heterocycloalkyloxy, C 3- 8 cycloalkenyloxy, 5 to 6 membered aryl or heteroaryl, said C 1-6 alkyl, C 1-6 alkoxy, C 2-6 alkenyloxy, C 2-6 alkynyloxy, 3 to 6-membered cycloalkoxy, 3 to 6-membered heterocycloalkyloxy, 3 to 8-membered cycloalkenyloxy, 5 to 6-membered aryl or heteroaryl are optionally replaced by one or more members selected from halogen, Hydroxy, cyano, amino, C 1-6 alkyl or C 1-6 alkoxy.
术语“杂芳基”指包含1至4个杂原子、5至14个环原子的杂芳族体系,其中杂原子选自氧、硫和氮。杂芳基优选为6至12元,更优选为5元或6元。例如。其非限制性实例包括:咪唑基、呋喃基、噻吩基、噻唑基、吡唑基、噁唑基(oxazolyl)、异噁唑基(isoxazolyl)、吡咯基、四唑基、吡啶基、嘧啶基、噻二唑、吡嗪基、三唑 基、吲唑基、苯并咪唑基、 等。 The term "heteroaryl" refers to a heteroaromatic system comprising 1 to 4 heteroatoms, 5 to 14 ring atoms, wherein the heteroatoms are selected from oxygen, sulfur and nitrogen. The heteroaryl group is preferably 6 to 12 membered, more preferably 5 or 6 membered. For example. Non-limiting examples thereof include: imidazolyl, furyl, thienyl, thiazolyl, pyrazolyl, oxazolyl, isoxazolyl, pyrrolyl, tetrazolyl, pyridyl, pyrimidinyl , Thiadiazole, pyrazinyl, triazolyl, indazolyl, benzimidazolyl, wait.
所述杂芳基环可以稠合于芳基、杂环烷基或环烷基环上,其中与母体结构连接在一起的环为杂芳基环,其非限制性实例包括:The heteroaryl ring may be fused to an aryl, heterocycloalkyl or cycloalkyl ring, wherein the ring bonded to the parent structure is a heteroaryl ring, non-limiting examples of which include:
杂芳基可以是任选取代的或非取代的,当被取代时,取代基优选为一个或多个以下基团,其独立地选自卤素、羟基、氰基、氨基、C 1-6烷基或C 1-6烷氧基(此处需要根据权利要求调整!)。 Heteroaryl may be optionally substituted or unsubstituted, and when substituted, the substituent is preferably one or more of the following groups independently selected from halogen, hydroxy, cyano, amino, C alkane group or C 1-6 alkoxy group (here needs to be adjusted according to the claims!).
术语“螺环”指两环共用一个原子的化合物。螺环烷基的非限制性实例包括:The term "spiro" refers to a compound in which two rings share one atom. Non-limiting examples of spirocycloalkyl groups include:
术语“并环”指两个或两个以上环通过共用两个相邻的原子并合而成的化合物。并环烷基的非限制性实例包括:The term "merged ring" refers to a compound formed by combining two or more rings by sharing two adjacent atoms. Non-limiting examples of cycloalkyl groups include:
术语“桥环”指两个或两个以上环状结构彼此共用两个非相邻的环原子所形成的结构。根据组成环的数目可以分为双环、三环、四环或多环桥环烷基,优选为双环、三环或四环,更有选为双环或三环。桥环烷基的非限制性实例包括:The term "bridged ring" refers to a structure formed by two or more ring structures sharing two non-adjacent ring atoms with each other. According to the number of constituent rings, it can be divided into bicyclic, tricyclic, tetracyclic or polycyclic bridged cycloalkyl groups, preferably bicyclic, tricyclic or tetracyclic, more preferably bicyclic or tricyclic. Non-limiting examples of bridged cycloalkyl groups include:
术语“杂环”指构成环的原子除碳原子外还有其他原子,其包括杂环烷基和杂芳环。The term "heterocyclic ring" refers to atoms other than carbon atoms that make up the ring, and includes heterocycloalkyl and heteroaryl rings.
术语“羟基”指-OH基团。The term "hydroxyl" refers to a -OH group.
术语“卤素”指氟、氯、溴或碘。The term "halogen" refers to fluorine, chlorine, bromine or iodine.
术语“氰基”指-CN。The term "cyano" refers to -CN.
术语“氨基”指-NH 2。 The term "amino" refers to -NH2 .
术语“硝基”指-NO 2。 The term "nitro" refers to -NO2 .
术语“氧代”指=O取代基。The term "oxo" refers to a =O substituent.
“取代的”指基团中的一个或多个氢原子,优选为最多5个,更优选为1~3个氢原子彼此独立地被相应数目的取代基取代。当取代基是酮或氧代(即,=O)时,则原子上有两个(2个)氢被替代。"Substituted" means that one or more hydrogen atoms in a group, preferably up to 5, more preferably 1 to 3 hydrogen atoms are independently substituted by the corresponding number of substituents. When a substituent is keto or oxo (ie, =0), then two (2) hydrogens on the atom are replaced.
“被一个或多个……取代”是指可以被单个或多个取代基取代。当被多个取代基取代时,可以是复数个相同取代基,也可以是一个或复数个不同取代基的组合。"Substituted by one or more" means that it can be substituted by single or multiple substituents. When substituted by a plurality of substituents, it may be a plurality of the same substituents, or one or a combination of a plurality of different substituents.
以下结合实施例进一步描述本公开,但这些实施例并非限制着本公开的范围。The present disclosure is further described below in conjunction with examples, but these examples do not limit the scope of the present disclosure.
本公开实施例中未注明具体条件的实验方法,通常按照常规条件,或按照原料或商品制造厂商所建议的条件。未注明具体来源的试剂,为市场购买的常规试剂。The experimental methods in the embodiments of the present disclosure that do not indicate specific conditions are generally in accordance with conventional conditions, or in accordance with the conditions suggested by the raw material or commodity manufacturers. Reagents without specific sources indicated are conventional reagents purchased in the market.
化合物的结构是通过核磁共振(NMR)或/和质谱(MS)来确定的。NMR位移(δ)以10 -6(ppm)的单位给出。NMR的测定是用Bruker AVANCE-400核磁仪,测定溶剂为氘代二甲基亚砜(DMSO-d 6),氘代氯仿(CDCl 3),氘代甲醇(CD 3OD),内标为四甲基硅烷(TMS)。 Compound structures were determined by nuclear magnetic resonance (NMR) or/and mass spectroscopy (MS). NMR shifts (δ) are given in units of 10 -6 (ppm). The determination of NMR is to use Bruker AVANCE-400 nuclear magnetic instrument, and the determination solvent is deuterated dimethyl sulfoxide (DMSO-d 6 ), deuterated chloroform (CDCl 3 ), deuterated methanol (CD 3 OD), and the internal standard is four Methylsilane (TMS).
MS的测定用Shimadzu 2010 Mass Spectrometer或Agilent 6110A MSD质谱仪。MS was determined by Shimadzu 2010 Mass Spectrometer or Agilent 6110A MSD mass spectrometer.
HPLC的测定使用Shimadzu LC-20A systems、Shimadzu LC-2010HT series或安捷伦Agilent 1200LC高压液相色谱仪(Ultimate XB-C18 3.0*150mm色谱柱或Xtimate C18 2.1*30mm色谱柱)。The determination of HPLC uses Shimadzu LC-20A systems, Shimadzu LC-2010HT series or Agilent Agilent 1200LC high pressure liquid chromatography (Ultimate XB-C18 3.0*150mm column or Xtimate C18 2.1*30mm column).
手性HPLC分析测定使用Chiralpak IC-3 100×4.6mm I.D.,3um、Chiralpak AD-3 150×4.6mm I.D.,3um、Chiralpak AD-3 50×4.6mm I.D.,3um、Chiralpak AS-3 150×4.6mm I.D.,3um、Chiralpak AS-3 100×4.6mm I.D.,3μm、ChiralCel OD-3 150×4.6mm I.D.,3um、Chiralcel OD-3 100×4.6mm I.D.,3μm、ChiralCel OJ-H 150×4.6mm I.D.,5um、Chiralcel OJ-3 150×4.6mm I.D.,3um色谱柱;Chiralpak IC-3 100×4.6mm I.D., 3um, Chiralpak AD-3 150×4.6mm I.D., 3um, Chiralpak AD-3 50×4.6mm I.D., 3um, Chiralpak AS-3 150×4.6mm were used for chiral HPLC analysis and determination I.D., 3um, Chiralpak AS-3 100×4.6mm I.D., 3μm, ChiralCel OD-3 150×4.6mm I.D., 3um, Chiralcel OD-3 100×4.6mm I.D., 3μm, ChiralCel OJ-H 150×4.6mm I.D., 5um, Chiralcel OJ-3 150×4.6mm I.D., 3um column;
薄层层析硅胶板使用烟台黄海HSGF254或青岛GF254硅胶板,薄层色谱法(TLC)使用的硅胶板采用的规格是0.15mm~0.2mm,薄层层析分离纯化产品采用的规格是0.4mm~0.5mm。The thin-layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate. The specification of the silica gel plate used in thin-layer chromatography (TLC) is 0.15mm-0.2mm, and the specification of thin-layer chromatography separation and purification products is 0.4mm. ~0.5mm.
柱层析一般使用烟台黄海硅胶100~200目、200~300目或300~400目硅胶为载体。Column chromatography generally uses Yantai Huanghai silica gel 100-200 mesh, 200-300 mesh or 300-400 mesh silica gel as the carrier.
手性制备柱使用DAICEL CHIRALPAK IC(250mm*30mm,10um)或 Phenomenex-Amylose-1(250mm*30mm,5um)。The chiral preparative column uses DAICEL CHIRALPAK IC (250mm*30mm, 10um) or Phenomenex-Amylose-1 (250mm*30mm, 5um).
CombiFlash快速制备仪使用Combiflash Rf150(TELEDYNE ISCO)。The CombiFlash rapid preparation instrument uses Combiflash Rf150 (TELEDYNE ISCO).
本公开的已知的起始原料可以采用或按照本领域已知的方法来合成,或可购买自ABCR GmbH&Co.KG,Acros Organics,Aldrich Chemical Company,韶远化学科技(Accela ChemBio Inc)、达瑞化学品等公司。The known starting materials of the present disclosure can be adopted or synthesized according to methods known in the art, or can be purchased from ABCR GmbH & Co. KG, Acros Organics, Aldrich Chemical Company, Shaoyuan Chemical Technology (Accela ChemBio Inc), Darui chemical companies.
实施例中无特殊说明,反应能够均在氩气氛或氮气氛下进行。Unless otherwise specified in the examples, the reactions can all be carried out under an argon atmosphere or a nitrogen atmosphere.
氩气氛或氮气氛是指反应瓶连接一个约1L容积的氩气或氮气气球。Argon atmosphere or nitrogen atmosphere means that the reaction bottle is connected to an argon or nitrogen balloon with a volume of about 1 L.
氢气氛是指反应瓶连接一个约1L容积的氢气气球。The hydrogen atmosphere means that the reaction bottle is connected to a hydrogen balloon with a capacity of about 1L.
加压氢化反应使用Parr 3916EKX型氢化仪和清蓝QL-500型氢气发生器或HC2-SS型氢化仪。The pressurized hydrogenation reaction uses Parr 3916EKX hydrogenation instrument and Qinglan QL-500 hydrogen generator or HC2-SS hydrogenation instrument.
氢化反应通常抽真空,充入氢气,反复操作3次。The hydrogenation reaction is usually vacuumized and filled with hydrogen, and the operation is repeated 3 times.
微波反应使用CEM Discover-S 908860型微波反应器。For the microwave reaction, a CEM Discover-S 908860 microwave reactor was used.
实施例中无特殊说明,溶液是指水溶液。Unless otherwise specified in the examples, the solution refers to an aqueous solution.
实施例中无特殊说明,反应的温度为室温,为20℃~30℃。Unless otherwise specified in the examples, the reaction temperature is room temperature, which is 20°C to 30°C.
实施例中的反应进程的监测采用薄层色谱法(TLC),反应所使用的展开剂,纯化化合物采用的柱层析的洗脱剂的体系和薄层色谱法的展开剂体系,溶剂的体积比根据化合物的极性不同而进行调节,也可以加入少量的三乙胺和醋酸等碱性或酸性试剂进行调节。The monitoring of the reaction process in the embodiment adopts thin-layer chromatography (TLC), the developing agent used in reaction, the eluent system of the eluent system of the column chromatography that purification compound adopts and the developing agent system of thin-layer chromatography, the volume of solvent The ratio is adjusted according to the polarity of the compound, and it can also be adjusted by adding a small amount of basic or acidic reagents such as triethylamine and acetic acid.
实施例1Example 1
(1R,3S)-3-(3-((4-氰基吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((4-cyanopyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate
第一步first step
(1R,3S)-3-(1-(叔-丁基)-5-((4-氰基吡啶-2-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯1b(1R,3S)-3-(1-(tert-butyl)-5-((4-cyanopyridin-2-yl)amino)-1H-pyrazol-3-yl)cyclopentylisopropyl carbamate 1b
将(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(40mg,0.13mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得),2-氯代异烟腈(20mg,0.14mmol),醋酸钯(5.8mg,0.03mmol),1,1'-联萘-2.2'-二苯膦(32.3mg,0.05mmol),碳酸铯(84.5mg,0.26mmol)和1,4-二氧六环(1.5mL)置于微波管中,用氮气吹1分钟。反应液在100℃微波反应2小时。反应结束后,冷却至室温, 减压浓缩得到粗品1b呈黑色固体,直接用于下一步反应(80mg,产率30%)。(1R,3S)-3-(5-Amino-1-(tert-butyl)-1H-pyrazol-3-yl)cyclopentyl isopropyl carbamate (40mg, 0.13mmol, using patent Prepared by the method disclosed in the application "WO 2020/157652 A2"), 2-chloroisonicotinonitrile (20mg, 0.14mmol), palladium acetate (5.8mg, 0.03mmol), 1,1'-binaphthalene-2.2' - Diphenylphosphine (32.3 mg, 0.05 mmol), cesium carbonate (84.5 mg, 0.26 mmol) and 1,4-dioxane (1.5 mL) were placed in a microwave tube and blown with nitrogen for 1 minute. The reaction solution was microwaved at 100° C. for 2 hours. After the reaction, it was cooled to room temperature and concentrated under reduced pressure to obtain the crude product 1b as a black solid, which was directly used in the next reaction (80 mg, yield 30%).
MS m/z(ESI):411.4[M+H] +。 MS m/z (ESI): 411.4 [M+H] + .
第二步second step
(1R,3S)-3-(3-((4-氰基吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯1(1R,3S)-3-(3-((4-cyanopyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate 1
在室温下,向(1R,3S)-3-(1-(叔-丁基)-5-((4-氰基吡啶-2-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(80mg,0.04mmol)中加入甲酸(1.5mL)。反应液在100℃下搅拌反应1个小时。反应结束后,反应液直接用C-18反相色谱法纯化得到标题化合物1(5.4mg,产率38%)。At room temperature, to (1R,3S)-3-(1-(tert-butyl)-5-((4-cyanopyridin-2-yl)amino)-1H-pyrazol-3-yl) ring Formic acid (1.5 mL) was added to pentyl isopropyl carbamate (80 mg, 0.04 mmol). The reaction solution was stirred and reacted at 100° C. for 1 hour. After the reaction, the reaction solution was directly purified by C-18 reverse phase chromatography to obtain the title compound 1 (5.4 mg, yield 38%).
MS m/z(ESI):355.4[M+H] +。 MS m/z (ESI): 355.4 [M+H] + .
1H NMR(400MHz,DMSO):δ11.99(s,1H),9.69(s,1H),8.33(s,1H),7.67(s,1H),7.00(d,J=35.1Hz,2H),6.08(s,1H),5.00(s,1H),3.58(s,1H),3.06(s,1H),1.96(m,3H),1.67(m,3H),1.13-0.92(m,6H)。 1 H NMR(400MHz,DMSO):δ11.99(s,1H),9.69(s,1H),8.33(s,1H),7.67(s,1H),7.00(d,J=35.1Hz,2H) ,6.08(s,1H),5.00(s,1H),3.58(s,1H),3.06(s,1H),1.96(m,3H),1.67(m,3H),1.13-0.92(m,6H ).
实施例2Example 2
(1R,3S)-3-(3-((5-(氰基甲基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((5-(cyanomethyl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate
第一步first step
(1R,3S)-3-(1-(叔-丁基)-5-((5-(氰基甲基)吡啶-2-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯2b(1R,3S)-3-(1-(tert-butyl)-5-((5-(cyanomethyl)pyridin-2-yl)amino)-1H-pyrazol-3-yl)cyclopentene isopropyl carbamate 2b
氮气氛下,依次将2-(6-氯吡啶-3-基)乙酰腈(20mg,0.13mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(40.4mg,0.13mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、醋酸钯(5.9mg,0.03mmol)、1,1'-联萘-2,2'-双二苯膦(16.3mg,0.03mmol)溶于1,4-二氧六环(3mL)中。加入碳酸铯(106.8mg,0.33mmol),80℃条件下反应2小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后得到标题化合物2b(40mg,产率71.8%)。Under nitrogen atmosphere, 2-(6-chloropyridin-3-yl)acetonitrile (20mg, 0.13mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)-1H -pyrazol-3-yl) cyclopentyl isopropyl carbamate (40.4mg, 0.13mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), palladium acetate (5.9mg, 0.03 mmol), 1,1'-binaphthyl-2,2'-bisdiphenylphosphine (16.3 mg, 0.03 mmol) were dissolved in 1,4-dioxane (3 mL). Add cesium carbonate (106.8mg, 0.33mmol) and react at 80°C for 2 hours. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected and concentrated under reduced pressure to obtain the title compound 2b (40 mg, yield 71.8%).
MS m/z(ESI):425.6[M+H] +。 MS m/z (ESI): 425.6 [M+H] + .
第二步second step
(1R,3S)-3-(3-((5-(氰基甲基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯2(1R,3S)-3-(3-((5-(cyanomethyl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate 2
将(1R,3S)-3-(1-(叔-丁基)-5-((5-(氰基甲基)吡啶-2-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(40mg,0.09mmol)溶于6mL甲酸中,在80℃条件下反应3小时。冷却至室温,反应液直接用C-18反相色谱法纯化得到标题化合物2(12mg,产率34.5%)。(1R,3S)-3-(1-(tert-butyl)-5-((5-(cyanomethyl)pyridin-2-yl)amino)-1H-pyrazol-3-yl)ring Amyl isopropyl carbamate (40mg, 0.09mmol) was dissolved in 6mL of formic acid and reacted at 80°C for 3 hours. After cooling to room temperature, the reaction solution was directly purified by C-18 reverse phase chromatography to obtain the title compound 2 (12 mg, yield 34.5%).
MS m/z(ESI):369.4[M+H] +。 MS m/z (ESI): 369.4 [M+H] + .
1H NMR(400MHz,DMSO):δ11.84(s,1H),9.20(s,1H),8.06(d,J=2.4Hz,1H),7.52(dd,J=8.7,2.5Hz,1H),7.29(s,1H),6.96(d,J=7.9Hz,1H),6.07(s,1H),5.00(s,1H),3.88(s,2H),3.58(m,2H),3.11–2.97(m,1H),2.00(t,J=8.3Hz,1H),1.90(m,1H),1.73(d,J=9.8Hz,2H),1.61(s,1H),1.03(d,J=6.5,6H)。 1 H NMR(400MHz,DMSO):δ11.84(s,1H),9.20(s,1H),8.06(d,J=2.4Hz,1H),7.52(dd,J=8.7,2.5Hz,1H) ,7.29(s,1H),6.96(d,J=7.9Hz,1H),6.07(s,1H),5.00(s,1H),3.88(s,2H),3.58(m,2H),3.11– 2.97(m,1H),2.00(t,J=8.3Hz,1H),1.90(m,1H),1.73(d,J=9.8Hz,2H),1.61(s,1H),1.03(d,J = 6.5, 6H).
实施例3Example 3
(1R,3S)-3-(3-((4-(1H-1,2,4-三唑-1-基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((4-(1H-1,2,4-triazol-1-yl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)ring Amyl isopropyl carbamate
第一步first step
2-氯-4-(1H-1,2,4-三唑-1-基)吡啶3b2-Chloro-4-(1H-1,2,4-triazol-1-yl)pyridine 3b
氮气氛下,将1H-1,2,4-三唑(420mg,6.08mmol)溶于N,N-二甲基甲酰胺(8mL)中。在室温下将氢化钠(243.2mg,6.08mmol)分批加入到反应液中,在室温下搅拌半小时,将2,4-二氯吡啶(300mg,2.02mmol)分批加入到反应液中,在100℃下搅拌2小时,降温至室温,反应液加入50ml水,用乙酸乙酯萃取,合并有机相,将有机相洗涤,干燥,减压浓缩后残余物用C-18反相色谱法纯化得到标题化合物3b(120mg,产率32.8%)。1H-1,2,4-triazole (420 mg, 6.08 mmol) was dissolved in N,N-dimethylformamide (8 mL) under nitrogen atmosphere. Sodium hydride (243.2 mg, 6.08 mmol) was added to the reaction solution in batches at room temperature, stirred at room temperature for half an hour, 2,4-dichloropyridine (300 mg, 2.02 mmol) was added to the reaction solution in batches, Stir at 100°C for 2 hours, cool down to room temperature, add 50ml of water to the reaction solution, extract with ethyl acetate, combine the organic phases, wash the organic phases, dry, and concentrate under reduced pressure, the residue is purified by C-18 reverse phase chromatography The title compound 3b (120 mg, 32.8% yield) was obtained.
MS m/z(ESI):181.2[M+H] +。 MS m/z (ESI): 181.2 [M+H] + .
1H NMR(400MHz,DMSO):δ9.58(s,1H),8.58(d,J=5.5Hz,1H),8.38(s,1H),8.11(d,J=2.0Hz,1H),7.95(dd,J=5.6,1.9Hz,1H)。 1 H NMR (400MHz, DMSO): δ9.58(s, 1H), 8.58(d, J=5.5Hz, 1H), 8.38(s, 1H), 8.11(d, J=2.0Hz, 1H), 7.95 (dd, J = 5.6, 1.9 Hz, 1H).
第二步second step
(1R,3S)-3-(5-((4-(1H-1,2,4-三唑-1-基)吡啶-2-基)氨基)-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯3c(1R,3S)-3-(5-((4-(1H-1,2,4-triazol-1-yl)pyridin-2-yl)amino)-1-(tert-butyl)-1H -pyrazol-3-yl)cyclopentylisopropylcarbamate 3c
氮气氛下,依次将2-氯-4-(1H-1,2,4-三唑-1-基)吡啶(15mg,0.08mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(25.6mg,0.08mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、醋酸钯(3.7mg,0.02mmol)、1,1'-联萘-2,2'-双二苯膦(10.3mg,0.02mmol)溶于1,4-二氧六环(2mL)中。加入碳酸铯(67.7mg,0.21mmol),80℃条件下反应2小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后得到标题化合物3c(30mg,产率79.8%)Under nitrogen atmosphere, 2-chloro-4-(1H-1,2,4-triazol-1-yl)pyridine (15 mg, 0.08 mmol), (1R,3S)-3-(5-amino-1 -(tert-butyl)-1H-pyrazol-3-yl)cyclopentylisopropylcarbamate (25.6mg, 0.08mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2" ), palladium acetate (3.7mg, 0.02mmol), 1,1'-binaphthyl-2,2'-bisdiphenylphosphine (10.3mg, 0.02mmol) were dissolved in 1,4-dioxane (2mL) . Add cesium carbonate (67.7mg, 0.21mmol) and react at 80°C for 2 hours. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected and concentrated under reduced pressure to obtain the title compound 3c (30 mg, yield 79.8%)
MS m/z(ESI):453.7[M+H] +。 MS m/z (ESI): 453.7 [M+H] + .
第三步third step
(1R,3S)-3-(3-((4-(1H-1,2,4-三唑-1-基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯3(1R,3S)-3-(3-((4-(1H-1,2,4-triazol-1-yl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)ring Amyl isopropyl carbamate 3
将(1R,3S)-3-(5-((4-(1H-1,2,4-三唑-1-基)吡啶-2-基)氨基)-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(30mg,0.07mmol)溶于3mL甲酸中,在80℃条件下反应12小时。冷却至室温,反应液直接用C-18反相色谱法纯化得到标题化合物3(10.7mg,产率40.7%)。(1R,3S)-3-(5-((4-(1H-1,2,4-triazol-1-yl)pyridin-2-yl)amino)-1-(tert-butyl)- 1H-pyrazol-3-yl) cyclopentyl isopropyl carbamate (30mg, 0.07mmol) was dissolved in 3mL formic acid, and reacted at 80°C for 12 hours. After cooling to room temperature, the reaction solution was directly purified by C-18 reverse phase chromatography to obtain the title compound 3 (10.7 mg, yield 40.7%).
MS m/z(ESI):397.4[M+H] +。 MS m/z (ESI): 397.4 [M+H] + .
1H NMR(400MHz,DMSO-d6):δ12.21-11.66(m,1H),9.52-9.42(m,1H),9.42-9.33(m,1H),8.34-8.18(m,2H),7.81(s,1H),7.20(d,J=2.1Hz,1H),7.04-6.83(m,1H),6.05(s,1H),5.05-4.95(m,1H),3.69-3.47(m,3H),2.10-1.96(m,1H),1.95-1.83(m,1H),1.79-1.67(m,2H),1.66-1.54(m,1H),1.03(br d,J=6.4Hz,6H)。 1 H NMR(400MHz,DMSO-d6):δ12.21-11.66(m,1H),9.52-9.42(m,1H),9.42-9.33(m,1H),8.34-8.18(m,2H),7.81 (s,1H),7.20(d,J=2.1Hz,1H),7.04-6.83(m,1H),6.05(s,1H),5.05-4.95(m,1H),3.69-3.47(m,3H ),2.10-1.96(m,1H),1.95-1.83(m,1H),1.79-1.67(m,2H),1.66-1.54(m,1H),1.03(br d,J=6.4Hz,6H) .
实施例4Example 4
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({4-[(甲基氨基)二氧亚基-6-硫基]苯基}氨基)-2H-吡唑-3-基]环戊基酯(Propan-2-ylamino)methane-(1R,3S)-3-[5-({4-[(methylamino)dioxylidene-6-thio]phenyl}amino)-2H- Pyrazol-3-yl]cyclopentyl ester
根据实施例1合成步骤替换相应原料制得化合物4。Compound 4 was prepared according to the synthesis steps of Example 1 by replacing the corresponding raw materials.
MS m/z(ESI):422.2[M+H] +。 MS m/z (ESI): 422.2 [M+H] + .
实施例5Example 5
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(氰基甲基)吡嗪-2-基]氨基}-2H-吡唑-3-基)环戊基酯(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[5-(cyanomethyl)pyrazin-2-yl]amino}-2H-pyrazol-3-yl ) cyclopentyl ester
第一步first step
2-(5-溴吡嗪-2-基)-2-氰基乙酸-2-甲基丙-2-基酯5b2-(5-Bromopyrazin-2-yl)-2-cyanoacetic acid-2-methylpropan-2-yl ester 5b
首先将2-溴-5-氯吡嗪5a(500mg,2.6mmol)溶于N,N-二甲基甲酰胺(20ml)中,随后加入2-氰基乙酸叔丁酯(733mg,5.2mmol)、碳酸铯(2.1g,6.5mmol),室温反应4小时。经萃取、干燥、浓缩后得到粗产品,该粗产物可直接用于下一步反应。2-Bromo-5-chloropyrazine 5a (500mg, 2.6mmol) was first dissolved in N,N-dimethylformamide (20ml) followed by the addition of tert-butyl 2-cyanoacetate (733mg, 5.2mmol) , cesium carbonate (2.1g, 6.5mmol), react at room temperature for 4 hours. The crude product is obtained after extraction, drying and concentration, which can be directly used in the next reaction.
MS m/z(ESI):298.4[M+H] +。 MS m/z (ESI): 298.4 [M+H] + .
第二步second step
(5-溴吡嗪-2-基)乙腈5c(5-Bromopyrazin-2-yl)acetonitrile 5c
将2-(5-溴吡嗪-2-基)-2-氰基乙酸-2-甲基丙-2-基酯5b溶于二氯甲烷(10ml)中,随后加入过量的三氟乙酸(2ml),室温反应1小时。经萃取、干燥、浓缩后得到粗产品,正相柱层析分离同时得到(5-溴吡嗪-2-基)乙腈5c(150mg,29%)。2-(5-Bromopyrazin-2-yl)-2-cyanoacetic acid-2-methylpropan-2-yl ester 5b was dissolved in dichloromethane (10 ml), followed by the addition of excess trifluoroacetic acid ( 2ml), react at room temperature for 1 hour. The crude product was obtained after extraction, drying and concentration, and (5-bromopyrazin-2-yl)acetonitrile 5c (150 mg, 29%) was obtained by normal phase column chromatography.
MS m/z(ESI):198.2[M+H] +。 MS m/z (ESI): 198.2 [M+H] + .
第三步third step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(氰基甲基)吡嗪-2-基]氨基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯5d(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[5-(cyanomethyl)pyrazin-2-yl]amino}-1-(2-methylpropane -2-yl)pyrazol-3-yl)cyclopentyl ester 5d
氮气氛下,依次将(5-溴吡嗪-2-基)乙腈(50mg,0.25mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(77mg,0.25mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、XantPhos(8.7mg 0.015mmol)、Pd2dba3(11.4mg,0.0125mmol)、碳酸铯(81.2mg,0.25mmol)溶于二氧六环(2mL)中,微波120℃条件下反应1小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后正相分离后得到标题化合物5d(50mg,产率47%)。Under nitrogen atmosphere, (5-bromopyrazin-2-yl)acetonitrile (50mg, 0.25mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)-1H-pyridine Azol-3-yl) cyclopentyl isopropyl carbamate (77mg, 0.25mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), XantPhos (8.7mg 0.015mmol), Pd2dba3 ( 11.4mg, 0.0125mmol), cesium carbonate (81.2mg, 0.25mmol) were dissolved in dioxane (2mL), and microwaved at 120°C for 1 hour. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected, concentrated under reduced pressure and separated from the normal phase to obtain the title compound 5d (50 mg, yield 47%).
MS m/z(ESI):426.2[M+H] +。 MS m/z (ESI): 426.2 [M+H] + .
第四步the fourth step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(氰基甲基)吡嗪-2-基]氨基}-2H-吡唑-3-基)环戊基酯5(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[5-(cyanomethyl)pyrazin-2-yl]amino}-2H-pyrazol-3-yl ) cyclopentyl ester 5
将(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(氰基甲基)吡嗪-2-基]氨基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯(40mg,0.09mmol)溶于3mL甲酸中,在100℃条件下反应1小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物5(8mg,产率23.0%)。(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[5-(cyanomethyl)pyrazin-2-yl]amino}-1-(2-methyl Propan-2-yl)pyrazol-3-yl)cyclopentyl ester (40mg, 0.09mmol) was dissolved in 3mL formic acid and reacted at 100°C for 1 hour. After cooling to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title compound 5 (8 mg, yield 23.0%).
MS m/z(ESI):370.2[M+H] +。 MS m/z (ESI): 370.2 [M+H] + .
1H NMR(400MHz,DMSO):δ12.00(s,1H),9.70(s,1H),8.55(s,1H),8.11(d,J=1.4Hz,1H),6.94(d,J=7.7Hz,1H),6.19(s,1H),5.00(s,1H),4.05(s,2H),3.63–3.54(m,1H),3.11-3.02(m,1H),2.10-1.84(m,3H),1.79-1.68(m,2H),1.64-1.60(m,1H),1.04(dJ=6.7Hz,6H)。 1 H NMR (400MHz, DMSO): δ12.00(s, 1H), 9.70(s, 1H), 8.55(s, 1H), 8.11(d, J=1.4Hz, 1H), 6.94(d, J= 7.7Hz, 1H), 6.19(s, 1H), 5.00(s, 1H), 4.05(s, 2H), 3.63–3.54(m, 1H), 3.11-3.02(m, 1H), 2.10-1.84(m , 3H), 1.79-1.68 (m, 2H), 1.64-1.60 (m, 1H), 1.04 (dJ=6.7Hz, 6H).
实施例6Example 6
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(5-氰基吡嗪-2-基)氨基]-2H-吡唑-3-基}环戊基酯(Propan-2-ylamino)methanoic acid-(1R,3S)-3-{5-[(5-cyanopyrazin-2-yl)amino]-2H-pyrazol-3-yl}cyclopentyl ester
根据实施例1类似步骤替换相应原料和试剂制得化合物6。Compound 6 was prepared according to the similar steps of Example 1 by substituting corresponding raw materials and reagents.
MS m/z(ESI):356.4[M+H] +。 MS m/z (ESI): 356.4 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ12.21(s,1H),10.47(s,1H),7.93(d,J=9.6Hz,1H),7.69(d,J=Hz,1H),6.93(d,J=7.2Hz,1H),6.19(s,1H),5.01(d,J=4.4Hz,1H),3.62-3.54(m,1H),3.11-3.05(m,1H),2.08-1.58(m,6H),1.03(d,J=6.4Hz,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ12.21(s, 1H), 10.47(s, 1H), 7.93(d, J=9.6Hz, 1H), 7.69(d, J=Hz, 1H) ,6.93(d,J=7.2Hz,1H),6.19(s,1H),5.01(d,J=4.4Hz,1H),3.62-3.54(m,1H),3.11-3.05(m,1H), 2.08-1.58 (m, 6H), 1.03 (d, J=6.4Hz, 6H).
实施例7Example 7
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(6-氰基-1,2-二氮杂环己熳-3-基)氨基]-2H-吡唑-3-基}环戊基酯(Propan-2-ylamino)methane-(1R,3S)-3-{5-[(6-cyano-1,2-diazacyclohexyl-3-yl)amino]-2H-pyridine Azol-3-yl}cyclopentyl ester
根据实施例1合成步骤替换相应原料制得化合物7。Compound 7 was prepared according to the synthesis steps of Example 1 by replacing the corresponding raw materials.
MS m/z(ESI):356.4[M+H] +。 MS m/z (ESI): 356.4 [M+H] + .
实施例8Example 8
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({4-[(氨基)二氧亚基-6-硫基]苯基}氨基)-2H-吡唑-3-基]环戊基酯(Propan-2-ylamino)methane-(1R,3S)-3-[5-({4-[(amino)dioxylidene-6-thio]phenyl}amino)-2H-pyrazole -3-yl]cyclopentyl ester
第一步first step
(1R,3S)-3-(1-(叔丁基)-5-((4-磺酰苯基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯8b(1R,3S)-3-(1-(tert-butyl)-5-((4-sulfonylphenyl)amino)-1H-pyrazol-3-yl)cyclopentylisopropylcarbamate 8b
氮气氛下,依次将4-溴苯磺酰胺(23.5mg,0.10mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(30.8mg,0.10mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、XantPhos Pd G3(9.5mg,0.01mmol)、磷酸钾(63.6mg,0.30mmol)溶于NMP(2mL)中,微波80℃条件下反应2小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后正相分离后得到标题化合物8b(40mg,产率85%)。Under nitrogen atmosphere, 4-bromobenzenesulfonamide (23.5mg, 0.10mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)-1H-pyrazol-3-yl ) cyclopentyl isopropyl carbamate (30.8mg, 0.10mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), XantPhos Pd G3 (9.5mg, 0.01mmol), potassium phosphate ( 63.6mg, 0.30mmol) was dissolved in NMP (2mL), and reacted under microwave at 80°C for 2 hours. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected, concentrated under reduced pressure and separated from the normal phase to obtain the title compound 8b (40 mg, yield 85%).
MS m/z(ESI):464.3[M+H] +。 MS m/z (ESI): 464.3 [M+H] +.
第三步third step
(1R,3S)-3-(3-((4-磺酰苯基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯8(1R,3S)-3-(3-((4-sulfonylphenyl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate 8
将8b(40mg,0.08mmol)溶于3mL甲酸中,在80℃条件下反应12小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物8(7.7mg,产率18.9%)。8b (40mg, 0.08mmol) was dissolved in 3mL formic acid and reacted at 80°C for 12 hours. After cooling to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title compound 8 (7.7 mg, yield 18.9%).
MS m/z(ESI):408.2[M+H] +。 MS m/z (ESI): 408.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.87(s,1H),8.86(s,1H),7.64-7.57(m,2H),7.40(d,J=8.4Hz,2H),7.03-6.91(m,3H),5.69(s,1H),5.00(s,1H),3.63-3.53(m,1H),3.11-3.01(m,1H),2.49-2.41(m,1H),2.05-1.99(m,1H),1.97-1.84(m,1H),1.73(m,2H),1.27-1.22(m,1H),1.08-1.00(d,J=6.6,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.87(s, 1H), 8.86(s, 1H), 7.64-7.57(m, 2H), 7.40(d, J=8.4Hz, 2H), 7.03 -6.91(m,3H),5.69(s,1H),5.00(s,1H),3.63-3.53(m,1H),3.11-3.01(m,1H),2.49-2.41(m,1H),2.05 -1.99 (m, 1H), 1.97-1.84 (m, 1H), 1.73 (m, 2H), 1.27-1.22 (m, 1H), 1.08-1.00 (d, J=6.6, 6H).
实施例9Example 9
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({4-[(氨基)二氧亚基-6-硫基]-2氟苯基}氨基)-2H-吡唑-3-基]环戊基酯(Propan-2-ylamino)methane-(1R,3S)-3-[5-({4-[(amino)dioxylidene-6-sulfanyl]-2fluorophenyl}amino)-2H -pyrazol-3-yl]cyclopentyl ester
第一步first step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[4-(氨基二氧亚基-6-硫基)-2-氟苯基]氨基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯9b(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[4-(Aminodioxyylidene-6-thio)-2-fluorophenyl]amino}-1- (2-Methylprop-2-yl)pyrazol-3-yl)cyclopentyl ester 9b
将化合物9a(2.7g,10.6mmol),1a(3.0g,9.7mmol),Xantphos-Pd-G3(300mg,0.3mmol)和磷酸钾(2.5g,11.6mmol),溶于N-甲基吡咯烷酮(15mL)中,在微波下120℃反应5小时。待反应完全后,向反应液中加入水(30mL),用EtOAc(20mL×2)萃取。合并有机相并用无水Na 2SO 4干燥,过滤,真空减压浓缩得到化合物粗品。粗品通过C18柱纯化得到化合物9b(4.0g,产率85.7%)。 Compound 9a (2.7g, 10.6mmol), 1a (3.0g, 9.7mmol), Xantphos-Pd-G3 (300mg, 0.3mmol) and potassium phosphate (2.5g, 11.6mmol), were dissolved in N-methylpyrrolidone ( 15 mL), react under microwave at 120°C for 5 hours. After the reaction was complete, water (30 mL) was added to the reaction solution, and extracted with EtOAc (20 mL×2). The organic phases were combined and dried over anhydrous Na 2 SO 4 , filtered, and concentrated in vacuo to obtain the crude compound. The crude product was purified by C18 column to obtain compound 9b (4.0 g, yield 85.7%).
MS m/z(ESI):482.2[M+H] +。 MS m/z (ESI): 482.2 [M+H] + .
第二步second step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({4-[(氨基)二氧亚基-6-硫基]-2氟苯基}氨基)-2H-吡唑-3-基]环戊基酯9(Propan-2-ylamino)methane-(1R,3S)-3-[5-({4-[(amino)dioxylidene-6-sulfanyl]-2fluorophenyl}amino)-2H -pyrazol-3-yl]cyclopentyl ester 9
将化合物9b(4.0g,8.3mmol)溶于甲酸(20mL)中,在120℃反应5小时。待反应完全后,将反应液浓缩得到化合物粗品,粗品通过反相HPLC纯化得到化合物9(1.7g,产率48.2%)。Compound 9b (4.0 g, 8.3 mmol) was dissolved in formic acid (20 mL), and reacted at 120° C. for 5 hours. After the reaction was complete, the reaction solution was concentrated to obtain a crude compound, which was purified by reverse-phase HPLC to obtain compound 9 (1.7 g, yield 48.2%).
MS m/z(ESI):426.5[M+H] +。 MS m/z (ESI): 426.5 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.96(s,1H),8.62(s,1H),8.19(t,J=8.5Hz,1H),7.51(d,2H),7.17(s,2H),6.94(d,J=7.8Hz,1H),5.83(s,1H),5.08-4.96(m,1H),3.69-3.48(m,1H),3.16-2.92(m,1H),2.48-2.40(m,1H),2.09-1.98(m,1H),1.96-1.86(m,1H),1.76-1.69(m,2H),1.66-1.55(m,1H),1.04(d,J=6.6,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.96(s, 1H), 8.62(s, 1H), 8.19(t, J=8.5Hz, 1H), 7.51(d, 2H), 7.17(s ,2H),6.94(d,J=7.8Hz,1H),5.83(s,1H),5.08-4.96(m,1H),3.69-3.48(m,1H),3.16-2.92(m,1H), 2.48-2.40(m,1H),2.09-1.98(m,1H),1.96-1.86(m,1H),1.76-1.69(m,2H),1.66-1.55(m,1H),1.04(d,J = 6.6, 6H).
实施例10Example 10
{[(2S)-丁-2-基]氨基}甲烷酸-(1R,3S)-3-(5-{[4-(氨基二氧亚基-6-硫基)-2-氟苯基]氨基}-2H-吡唑-3-基)环戊基酯{[(2S)-Butan-2-yl]amino}methane-(1R,3S)-3-(5-{[4-(aminodioxyylidene-6-thio)-2-fluorophenyl ]amino}-2H-pyrazol-3-yl)cyclopentyl ester
第一步first step
{[(2S)-丁-2-基]氨基}甲烷酸-(1R,3S)-3-(5-{[4-(氨基二氧亚基-6-硫基)-2-氟苯基]氨 基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯10c {[(2S)-Butan-2-yl]amino}methane-(1R,3S)-3-(5-{[4-(aminodioxyylidene-6-thio)-2-fluorophenyl ] amino }-1-(2-methylprop-2-yl)pyrazol-3-yl)cyclopentyl ester 10c
将化合物10a(0.5g,1.96mmol),10b(0.65g,2.0mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得),Xantphos-Pd-G3(95mg,0.1mmol),磷酸钾(0.53g,2.5mmol),溶于N-甲基吡咯烷酮(10mL)中,在微波下120℃反应5小时。待反应完全,向反应液中加入水(30mL),用EtOAc(20mL×2)萃取。合并有机相并用无水Na 2SO 4干燥,过滤,真空减压浓缩得到化合物粗品。粗品通过C18硅胶柱纯化得到化合物10c(454mg,0.91mmol,产率:46.4%)。 Compound 10a (0.5g, 1.96mmol), 10b (0.65g, 2.0mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), Xantphos-Pd-G3 (95mg, 0.1mmol), phosphoric acid Potassium (0.53g, 2.5mmol) was dissolved in N-methylpyrrolidone (10mL), and reacted under microwave at 120°C for 5 hours. After the reaction was complete, water (30 mL) was added to the reaction liquid, and extracted with EtOAc (20 mL×2). The organic phases were combined and dried over anhydrous Na 2 SO 4 , filtered, and concentrated in vacuo to obtain the crude compound. The crude product was purified by C18 silica gel column to obtain compound 10c (454 mg, 0.91 mmol, yield: 46.4%).
MS(ESI)m/z 496.4[M+H] + MS(ESI)m/z 496.4[M+H] +
第二步second step
{[(2S)-丁-2-基]氨基}甲烷酸-(1R,3S)-3-(5-{[4-(氨基二氧亚基-6-硫基)-2-氟苯基]氨基}-2H-吡唑-3-基)环戊基酯10{[(2S)-Butan-2-yl]amino}methane-(1R,3S)-3-(5-{[4-(aminodioxyylidene-6-thio)-2-fluorophenyl ]amino}-2H-pyrazol-3-yl)cyclopentyl ester 10
将化合物10c(454mg,0.91mmol)溶于甲酸(10mL)中,在120℃反应5小时。待反应完全后,将反应液浓缩得到化合物粗品,粗品通过HPLC纯化得到化合物10(250mg,0.57mmol,产率:62.5%)。Compound 10c (454mg, 0.91mmol) was dissolved in formic acid (10mL) and reacted at 120°C for 5 hours. After the reaction was complete, the reaction solution was concentrated to obtain a crude compound, which was purified by HPLC to obtain compound 10 (250 mg, 0.57 mmol, yield: 62.5%).
MS m/z(ESI):440.2[M+H] +。 MS m/z (ESI): 440.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.95(s,1H),8.61(s,1H),8.19(t,J=8.5Hz,1H),7.58-7.46(m,2H),7.17(s,2H),6.88(d,J=8.3Hz,1H),5.83(s,1H),5.07-4.95(m,1H),3.47-3.35(m,1H),3.15-2.99(m,1H),2.49-2.40(m,1H),2.11-1.99(m,1H),1.98-1.84(m,1H),1.80-1.66(m,2H),1.66-1.53(m,1H),1.43-1.31(m,2H),1.02(d,J=6.6Hz,3H),0.81(t,J=7.4Hz,3H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.95(s, 1H), 8.61(s, 1H), 8.19(t, J=8.5Hz, 1H), 7.58-7.46(m, 2H), 7.17 (s,2H),6.88(d,J=8.3Hz,1H),5.83(s,1H),5.07-4.95(m,1H),3.47-3.35(m,1H),3.15-2.99(m,1H ),2.49-2.40(m,1H),2.11-1.99(m,1H),1.98-1.84(m,1H),1.80-1.66(m,2H),1.66-1.53(m,1H),1.43-1.31 (m, 2H), 1.02 (d, J=6.6Hz, 3H), 0.81 (t, J=7.4Hz, 3H).
实施例11Example 11
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({6-[(甲基氨基)二氧亚基-6-硫基]吡啶-3-基}氨基)-2H-吡唑-3-基]环戊基酯(Propan-2-ylamino)methanoic acid-(1R,3S)-3-[5-({6-[(methylamino)dioxylidene-6-thio]pyridin-3-yl}amino) -2H-pyrazol-3-yl]cyclopentyl ester
第一步first step
5-溴-N-甲基吡啶-2-磺酰胺11b5-Bromo-N-methylpyridine-2-sulfonamide 11b
室温下,将甲胺盐酸盐(180mg,3mmol)、DIEA(650mg,5mmol)溶于无水二氯甲烷(10mL)中,澄清后慢慢加入5-溴吡啶-2-磺酰氯(250mg,1mmol),继续搅拌,TLC监测至反应结束后,加入水(10mL)洗涤1次,饱和食盐水(10mL)洗涤1次,收集有机相层无水硫酸钠干燥,旋至小体积直接正相柱层析,得到11b(180mg,产率72%)。At room temperature, dissolve methylamine hydrochloride (180mg, 3mmol) and DIEA (650mg, 5mmol) in anhydrous dichloromethane (10mL), and slowly add 5-bromopyridine-2-sulfonyl chloride (250mg, 1mmol), continue to stir, TLC monitoring until the end of the reaction, add water (10mL) to wash once, saturated brine (10mL) wash once, collect the organic phase layer anhydrous sodium sulfate and dry, spin to a small volume direct normal phase column Chromatography afforded 11b (180 mg, 72% yield).
第二步second step
(1R,3S)-3-(1-(叔丁基)-5-((6-(N-甲基氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯11c(1R,3S)-3-(1-(tert-butyl)-5-((6-(N-methylaminosulfonyl)pyridin-3-yl)amino)-1H-pyrazol-3-yl) Cyclopentyl isopropyl carbamate 11c
氮气氛下,依次将5-溴-N-甲基吡啶-2-磺酰胺(25.1mg,0.10mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(30.8mg,0.10mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、Pd 2(dba) 3(9.1mg,0.01mmol)、Xantphos(11.4mg,0.02mmol)、CS 2CO 3(96.2mg,0.30mmol)溶于1,4-二氧六环(2mL)中,微波80℃条件下反应2小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后正相分离后得到标题化合物11c(15mg,产率32%)。 Under nitrogen atmosphere, 5-bromo-N-methylpyridine-2-sulfonamide (25.1 mg, 0.10 mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)- 1H-pyrazol-3-yl) cyclopentyl isopropyl carbamate (30.8 mg, 0.10 mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), Pd 2 (dba) 3 (9.1mg, 0.01mmol), Xantphos (11.4mg, 0.02mmol), CS 2 CO 3 (96.2mg, 0.30mmol) were dissolved in 1,4-dioxane (2mL), and reacted under microwave at 80°C 2 Hour. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected, concentrated under reduced pressure and separated from the normal phase to obtain the title compound 11c (15 mg, yield 32%).
MS m/z(ESI):479.3[M+H] +。 MS m/z (ESI): 479.3 [M+H] + .
第三步third step
(1R,3S)-3-(3-((6-(N-甲基氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯11(1R,3S)-3-(3-((6-(N-Methylsulfonyl)pyridin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylaminomethyl Ester 11
将((1R,3S)-3-(1-(叔丁基)-5-((6-(N-甲基氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(15mg,0.03mmol)溶于2mL甲酸中,在80℃条件下反应12小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物(2.2mg,产率17.3%)。((1R,3S)-3-(1-(tert-butyl)-5-((6-(N-methylaminosulfonyl)pyridin-3-yl)amino)-1H-pyrazole-3- Base) cyclopentyl isopropyl carbamate (15 mg, 0.03 mmol) was dissolved in 2 mL of formic acid and reacted at 80 ° C for 12 hours. Cooled to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title Compound (2.2 mg, yield 17.3%).
MS m/z(ESI):423.2[M+H] +。 MS m/z (ESI): 423.2 [M+H] + .
实施例12Example 12
{[(2S)-丁-2-基]氨基}甲烷酸-(1R,3S)-3-[5-({6-[(乙基氨基)二氧亚基-6-硫基]吡啶-3-基}氨基)-2H-吡唑-3-基]环戊基酯{[(2S)-Butan-2-yl]amino}methane-(1R,3S)-3-[5-({6-[(Ethylamino)dioxyylidene-6-thio]pyridine- 3-yl}amino)-2H-pyrazol-3-yl]cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物12。Compound 12 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):451.2[M+H] +。 MS m/z (ESI): 451.2 [M+H] + .
1H NMR(400MHz,DMSO-d6):δ12.00(s,1H),9.02(s,1H),8.57(d,J=1.6Hz,1H),7.97(dd,J 1=1.6Hz,J 2=8.4Hz,1H),7.73(d,J=8.8Hz,1H),7.43(t,J=5.6Hz,1H),6.87(d,J=8.4Hz,1H),5.72(s,1H),5.00(d,J=2.4Hz,1H),3.42-3.35(m,1H),3.09-3.05(m,1H),2.90-2.83(m,2H),2.08-1.56(m,6H),1.40-1.30(m,2H),1.01(d,J=6.4Hz,3H),0.96(t,J=7.2Hz,3H),0.80(t,J=7.6Hz,3H)。 1 H NMR (400MHz, DMSO-d6): δ12.00(s, 1H), 9.02(s, 1H), 8.57(d, J=1.6Hz, 1H), 7.97(dd, J 1 =1.6Hz, J 2 =8.4Hz,1H),7.73(d,J=8.8Hz,1H),7.43(t,J=5.6Hz,1H),6.87(d,J=8.4Hz,1H),5.72(s,1H) ,5.00(d,J=2.4Hz,1H),3.42-3.35(m,1H),3.09-3.05(m,1H),2.90-2.83(m,2H),2.08-1.56(m,6H),1.40 -1.30(m, 2H), 1.01(d, J=6.4Hz, 3H), 0.96(t, J=7.2Hz, 3H), 0.80(t, J=7.6Hz, 3H).
实施例13Example 13
(1R,3S)-3-(3-((5-(2-(氘代甲氨基)-2-氧代乙基)吡嗪-2-基)氨基)-1H-吡唑-5-基)环戊基(S)-仲丁基氨基甲酸酯(1R,3S)-3-(3-((5-(2-(Deuteromethylamino)-2-oxoethyl)pyrazin-2-yl)amino)-1H-pyrazol-5-yl ) cyclopentyl (S)-sec-butyl carbamate
第一步first step
2-(5-氯吡嗪-2-基)丙二酸二乙酯13bDiethyl 2-(5-chloropyrazin-2-yl)malonate 13b
氮气氛下,依次将丙二酸二乙酯(2.49g,15.51mmol),2-吡啶甲酸(127.3mg,1.04mmol),碳酸铯(5.06g,15.51mmol)和碘化亚铜(393.9mg,2.07mmol)加到2-溴-5-氯吡嗪13a(1g,5.17mmol)的二氧六环(30mL)溶液中。在105℃下反应过夜,降至室温,将反应液减压浓缩,加入30mL水,用乙酸乙酯萃取,合并有机相,将有机相用饱和食盐水洗涤,干燥,减压浓缩后残余物用硅胶柱色谱分离纯化得到标题化合物13b(1.1g,产率79%)。Under nitrogen atmosphere, diethyl malonate (2.49g, 15.51mmol), 2-pyridinecarboxylic acid (127.3mg, 1.04mmol), cesium carbonate (5.06g, 15.51mmol) and cuprous iodide (393.9mg, 2.07 mmol) was added to a solution of 2-bromo-5-chloropyrazine 13a (1 g, 5.17 mmol) in dioxane (30 mL). Reacted overnight at 105°C, lowered to room temperature, concentrated the reaction solution under reduced pressure, added 30 mL of water, extracted with ethyl acetate, combined the organic phases, washed the organic phase with saturated brine, dried, and concentrated under reduced pressure. Separation and purification by silica gel column chromatography gave the title compound 13b (1.1 g, yield 79%).
MS m/z(ESI):273.1[M+H] +。 MS m/z (ESI): 273.1 [M+H] + .
第二步second step
2-(5-氯吡嗪-2-基)乙酸乙酯13c2-(5-Chloropyrazin-2-yl) ethyl acetate 13c
将2-(5-氯吡嗪-2-基)丙二酸二乙酯13b(1g,3.68mmol)用N,N-二甲基亚砜(20mL)溶解,向溶液中加氯化钠(234.6mg,4mmol)和水(132.4mg,7.35mmol)。在145℃下反应,待反应结束后将反应液降至室温,加入30mL水,用乙酸乙酯萃取,合并有机相,将有机相用饱和食盐水洗涤,干燥,减压浓缩后残余物用硅胶柱色谱分离纯化得到标题化合物13c(514.7mg,产率70%)。Diethyl 2-(5-chloropyrazin-2-yl)malonate 13b (1 g, 3.68 mmol) was dissolved in N,N-dimethylsulfoxide (20 mL), and sodium chloride ( 234.6mg, 4mmol) and water (132.4mg, 7.35mmol). React at 145°C. After the reaction is completed, the reaction solution is lowered to room temperature, 30 mL of water is added, extracted with ethyl acetate, the organic phases are combined, the organic phase is washed with saturated brine, dried, and concentrated under reduced pressure, and the residue is washed with silica gel Purified by column chromatography to obtain the title compound 13c (514.7 mg, yield 70%).
MS m/z(ESI):201.1[M+H] +。 MS m/z (ESI): 201.1 [M+H] + .
第三步third step
2-(5-((1-(叔丁基)-3-((1S,3R)-3-((((S)-仲丁基)氨基甲酰基)氧基)环戊基)-1H-吡唑-5-基)氨基)吡嗪-2-基)乙酸乙酯13d2-(5-((1-(tert-butyl)-3-((1S,3R)-3-((((S)-sec-butyl)carbamoyl)oxy)cyclopentyl)-1H -pyrazol-5-yl)amino)pyrazin-2-yl)ethyl acetate 13d
将2-(5-氯吡嗪-2-基)乙酸乙酯(500mg,2.5mmol),(1R,3S)-3-(5-氨基-1-(叔丁基)-1H-吡唑-3-基)环戊基((S)-仲丁基)氨基甲酸酯(805mg,2.5mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得),碳酸铯(2.45g,7.5mmol)和XantPhos Pd G3(119mg,0.125mmol)用1,4-二氧六环(10mL)溶解,氮气保护下回流,待反应结束后将反应液降至室温,加入20ml水,用乙酸乙酯萃取,合并有机相,将有机相用饱和食盐水洗涤,干燥,减压浓缩后残余物用硅胶柱色谱分离纯化得到标题化合物13d(850.5mg,产率70%)。Ethyl 2-(5-chloropyrazin-2-yl)acetate (500mg, 2.5mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)-1H-pyrazole- 3-yl) cyclopentyl ((S)-sec-butyl) carbamate (805mg, 2.5mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), cesium carbonate (2.45g, 7.5mmol) and XantPhos Pd G3 (119mg, 0.125mmol) were dissolved in 1,4-dioxane (10mL), and refluxed under nitrogen protection. After the reaction was completed, the reaction solution was lowered to room temperature, and 20ml of water was added. The ester was extracted, and the organic phases were combined. The organic phase was washed with saturated brine, dried, and concentrated under reduced pressure. The residue was separated and purified by silica gel column chromatography to obtain the title compound 13d (850.5 mg, yield 70%).
MS m/z(ESI):487.5[M+H] +。 MS m/z (ESI): 487.5 [M+H] + .
第四步the fourth step
2-(5-((1-(叔丁基)-3-((1S,3R)-3-((((S)-仲丁基)氨基甲酰基)氧基)环戊基)-1H-吡唑-5-基)氨基)吡嗪-2-基)乙酸13e2-(5-((1-(tert-butyl)-3-((1S,3R)-3-((((S)-sec-butyl)carbamoyl)oxy)cyclopentyl)-1H -pyrazol-5-yl)amino)pyrazin-2-yl)acetic acid 13e
将2-(5-((1-(叔丁基)-3-((1S,3R)-3-((((S)-仲丁基)氨基甲酰基)氧基)环戊基)-1H-吡唑-5-基)氨基)吡嗪-2-基)乙酸乙酯13d(500mg,1.03mmol)溶于四氢呋喃(4mL)中,向溶液中加氢氧化锂水溶液(2M,2.06mmol)。室温反应,待反应结束后将反应液用1M盐酸酸化,用乙酸乙酯萃取,合并有机相,将有机相用饱和食盐水洗涤,干燥,减压浓缩得到标题化合物13e(424.6mg,产率90%)。2-(5-((1-(tert-butyl)-3-((1S,3R)-3-((((S)-sec-butyl)carbamoyl)oxy)cyclopentyl)- 1H-pyrazol-5-yl)amino)pyrazin-2-yl)ethyl acetate 13d (500mg, 1.03mmol) was dissolved in THF (4mL), and to the solution was added aqueous lithium hydroxide (2M, 2.06mmol) . Reaction at room temperature, after the reaction was completed, the reaction solution was acidified with 1M hydrochloric acid, extracted with ethyl acetate, the organic phases were combined, washed with saturated brine, dried, and concentrated under reduced pressure to obtain the title compound 13e (424.6 mg, yield 90 %).
MS m/z(ESI):457.4[M-H] -。 MS m/z (ESI): 457.4 [MH] - .
第五步the fifth step
(1R,3S)-3-(1-(叔丁基)-5-((5-(2-(氘代甲氨基)-2-氧代乙基)吡嗪-2-基)氨基)-1H-吡唑-3-基)环戊基((S)-仲丁基)氨基甲酸酯13f(1R,3S)-3-(1-(tert-butyl)-5-((5-(2-(deuteromethylamino)-2-oxoethyl)pyrazin-2-yl)amino)- 1H-pyrazol-3-yl)cyclopentyl((S)-sec-butyl)carbamate 13f
将13e(400mg,0.87mmol)用二氯甲烷(5mL)溶解,向溶液中加1,1’羰基二咪唑(141.6mg,0.88mmol),反应液在室温下搅拌1小时。将氘代甲胺盐酸盐(62.2mg,0.87mmol)用二氯甲烷(1mL)溶解,向其中加入三乙胺(138μL,1mmol),室温下搅拌30分钟。将该悬浊液缓慢加入到上述混合液中,继续反应过夜。反应液用水淬灭,用二氯甲烷萃取,合并有机相,将有机相依次用1M盐酸,1M氢氧化钠水溶液和饱和食盐水洗涤,干燥,减压浓缩后残余物用硅胶柱色谱分离纯化得到标题化合物13f(247.4mg,产率60%)。13e (400mg, 0.87mmol) was dissolved in dichloromethane (5mL), 1,1'carbonyldiimidazole (141.6mg, 0.88mmol) was added to the solution, and the reaction solution was stirred at room temperature for 1 hour. Deuteromethylamine hydrochloride (62.2 mg, 0.87 mmol) was dissolved in dichloromethane (1 mL), triethylamine (138 μL, 1 mmol) was added thereto, and stirred at room temperature for 30 minutes. The suspension was slowly added to the above mixture, and the reaction was continued overnight. The reaction solution was quenched with water, extracted with dichloromethane, the organic phases were combined, and the organic phase was successively washed with 1M hydrochloric acid, 1M aqueous sodium hydroxide solution and saturated brine, dried, concentrated under reduced pressure, and the residue was separated and purified by silica gel column chromatography to obtain The title compound 13f (247.4 mg, 60% yield).
MS m/z(ESI):475.5[M+H] +。 MS m/z (ESI): 475.5 [M+H] + .
第六步step six
(1R,3S)-3-(3-((5-(2-(氘代甲氨基)-2-氧代乙基)吡嗪-2-基)氨基)-1H-吡唑-5-基)环戊基-(S)-仲丁基氨基甲酸酯13(1R,3S)-3-(3-((5-(2-(Deuteromethylamino)-2-oxoethyl)pyrazin-2-yl)amino)-1H-pyrazol-5-yl ) cyclopentyl-(S)-sec-butylcarbamate 13
将13f(173mg,0.36mmol)溶于甲酸(3mL),反应液在100℃下反应约2小时。待反应液冷却至室温减压浓缩后残余物用C-18反相色谱法纯化得到标题化合物13(30mg,产率20%)。13f (173mg, 0.36mmol) was dissolved in formic acid (3mL), and the reaction solution was reacted at 100°C for about 2 hours. After the reaction solution was cooled to room temperature and concentrated under reduced pressure, the residue was purified by C-18 reverse phase chromatography to obtain the title compound 13 (30 mg, yield 20%).
MS m/z(ESI):419.4[M+H] +。 MS m/z (ESI): 419.4 [M+H] + .
实施例14Example 14
(1R,3S)-3-(3-((5-(氰甲基)-3-甲基吡啶-2-基)氨基)-1H-吡唑-5-基)异丙基氨基甲酸环戊酯(1R,3S)-3-(3-((5-(cyanomethyl)-3-methylpyridin-2-yl)amino)-1H-pyrazol-5-yl)isopropylcarbamate cyclopentyl ester
第一步first step
4-(6-氯-5-甲基吡啶-3-基)异噁唑14b4-(6-Chloro-5-methylpyridin-3-yl)isoxazole 14b
氮气氛下,将5-溴-2-氯-3-甲基吡啶(1g,4.84mmol)溶于N,N-二甲基甲酰胺(5mL)和水(5ml)中,依次加入4-异噁唑硼酸频那醇酯(1.04g,5.32mmol)、氟化钾(0.84g,14.52mmol)、Pd(dppf)Cl 2(0.35g,0.48mmol),升至80℃反应16h,降至室温, 将反应液倒入10ml水中,乙酸乙酯(10ml*2)萃取,合并有机相,将有机相洗涤,干燥,减压浓缩得标题化合物14b(1g,产率105%)。 Under nitrogen atmosphere, 5-bromo-2-chloro-3-methylpyridine (1g, 4.84mmol) was dissolved in N,N-dimethylformamide (5mL) and water (5ml), and 4-iso Oxazole boronic acid pinacol ester (1.04g, 5.32mmol), potassium fluoride (0.84g, 14.52mmol), Pd(dppf)Cl 2 (0.35g, 0.48mmol), rise to 80°C for 16h, then cool to room temperature , The reaction solution was poured into 10ml of water, extracted with ethyl acetate (10ml*2), the organic phases were combined, washed, dried, and concentrated under reduced pressure to obtain the title compound 14b (1g, yield 105%).
MS m/z(ESI):195.2[M+H] +。 MS m/z (ESI): 195.2 [M+H] + .
第二步second step
2-(6-氯-5-甲基吡啶-3-基)乙腈14c2-(6-Chloro-5-methylpyridin-3-yl)acetonitrile 14c
将4-(6-氯-5-甲基吡啶-3-基)异噁唑(1g,5mmol)溶于甲醇(10ml)和水(1ml)中,再加入氟化钾(60mg,1mmol),升温至90℃,反应1h,降至室温,旋去甲醇,加入3ml水,用乙酸乙酯(5ml*2)萃取,合并有机相,拌样过柱得到标题化合物14c(350mg,产率40.9%)。Dissolve 4-(6-chloro-5-methylpyridin-3-yl)isoxazole (1g, 5mmol) in methanol (10ml) and water (1ml), then add potassium fluoride (60mg, 1mmol), Raise the temperature to 90°C, react for 1h, cool down to room temperature, spin off methanol, add 3ml of water, extract with ethyl acetate (5ml*2), combine the organic phases, mix the sample and pass through the column to obtain the title compound 14c (350mg, yield 40.9% ).
MS m/z(ESI):167.2[M+H] +。 MS m/z (ESI): 167.2 [M+H] + .
第三步third step
(1R,3S)-3-(1-(叔丁基)-5-((5-(氰甲基)-3-甲基吡啶-2-基)氨基)-1H-吡唑-3-基)异丙基氨基甲酸环戊酯14d(1R,3S)-3-(1-(tert-butyl)-5-((5-(cyanomethyl)-3-methylpyridin-2-yl)amino)-1H-pyrazol-3-yl ) Cyclopentyl isopropylcarbamate 14d
依次将14c(35.8mg,0.21mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(60mg,0.19mmol)采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、Pd2(dba)3(17.8mg,0.02mmol)、1,1'-联萘-2,2'-双二苯膦(24.3mg,0.04mmol)溶于1,4-二氧六环(2mL)中。加入磷酸钾(103.4mg,0.49mmol),微波反应120℃,4h。反应液冷却至室温,将反应液倒入5ml水中,乙酸乙酯(5ml*2)萃取,有机相合并,拌样过柱得标题化合物14d(40mg,产率46.9%)。14c (35.8 mg, 0.21 mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)-1H-pyrazol-3-yl)cyclopentylisopropylaminomethyl Ester (60mg, 0.19mmol) prepared by the method disclosed in the patent application "WO 2020/157652 A2"), Pd2(dba)3 (17.8mg, 0.02mmol), 1,1'-binaphthyl-2,2 '-Bisdiphenylphosphine (24.3 mg, 0.04 mmol) was dissolved in 1,4-dioxane (2 mL). Potassium phosphate (103.4mg, 0.49mmol) was added, and microwaved at 120°C for 4h. The reaction solution was cooled to room temperature, poured into 5ml of water, extracted with ethyl acetate (5ml*2), the organic phases were combined, mixed and passed through the column to obtain the title compound 14d (40mg, yield 46.9%).
MS m/z(ESI):439.3[M+H] +。 MS m/z (ESI): 439.3 [M+H] + .
第四步the fourth step
(1R,3S)-3-(3-((5-(氰甲基)-3-甲基吡啶-2-基)氨基)-1H-吡唑-5-基)异丙基氨基甲酸环戊酯14(1R,3S)-3-(3-((5-(cyanomethyl)-3-methylpyridin-2-yl)amino)-1H-pyrazol-5-yl)isopropylcarbamate cyclopentyl Ester 14
将14d(40mg,0.07mmol)溶于4mL甲酸中,在100℃条件下反应3小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物14(20mg,产率57.1%)。14d (40mg, 0.07mmol) was dissolved in 4mL formic acid and reacted at 100°C for 3 hours. After cooling to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title compound 14 (20 mg, yield 57.1%).
MS m/z(ESI):383.3[M+H] +。 MS m/z (ESI): 383.3 [M+H] + .
1H NMR(400MHz,DMSO-d6):δ11.94(s,1H),8.17(s,1H),7.98-7.92(m,1H),7.38(s,1H),6.93(d,J=7.6Hz,1H),6.28(s,1H),5.00(d,J=6.9Hz,1H),3.87(s,2H),3.58(h,J=6.7Hz,1H),3.09-2.99(m,1H),2.5-2.40(m,1H),2.22(s,3H),2.00(t,J=7.8Hz,1H),1.90(ddd,J=13.3,8.0,5.0Hz,1H),1.74(s,2H),1.65(s,1H),1.03(dd,J=6.6,2.1Hz,6H)。 1 H NMR(400MHz,DMSO-d6):δ11.94(s,1H),8.17(s,1H),7.98-7.92(m,1H),7.38(s,1H),6.93(d,J=7.6 Hz,1H),6.28(s,1H),5.00(d,J=6.9Hz,1H),3.87(s,2H),3.58(h,J=6.7Hz,1H),3.09-2.99(m,1H ),2.5-2.40(m,1H),2.22(s,3H),2.00(t,J=7.8Hz,1H),1.90(ddd,J=13.3,8.0,5.0Hz,1H),1.74(s, 2H), 1.65 (s, 1H), 1.03 (dd, J=6.6, 2.1 Hz, 6H).
实施例15Example 15
(1R,3S)-3-(3-((5-(氰甲基)-4-甲基吡啶-2-基)氨基)-1H-吡唑-5-基)异丙基氨基甲酸环戊酯(1R,3S)-3-(3-((5-(cyanomethyl)-4-methylpyridin-2-yl)amino)-1H-pyrazol-5-yl)isopropylcarbamate cyclopentyl ester
合成参考实施例14,变换相关原料可合成化合物15。Synthesis Reference Example 14, compound 15 can be synthesized by changing the relevant raw materials.
MS m/z(ESI):383.3[M+H] +。 MS m/z (ESI): 383.3 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.82(s,1H),9.09(s,1H),7.99(s,1H),7.16(s,1H),6.94(d,J=7.8Hz,1H),6.05(s,1H),5.00(d,J=7.3Hz,1H),3.87(s,2H),3.58(h,J=6.6Hz,1H),3.11-2.97(m,1H),2.45(dd,J=14.2,7.4Hz,1H),2.25(s,3H),2.01(q,J=8.5,6.9Hz,1H),1.96-1.83(m,1H),1.73(q,J=7.3,6.2Hz,2H),1.59(d,J=14.3Hz,1H),1.04(d,J=6.6Hz,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.82(s, 1H), 9.09(s, 1H), 7.99(s, 1H), 7.16(s, 1H), 6.94(d, J=7.8Hz ,1H),6.05(s,1H),5.00(d,J=7.3Hz,1H),3.87(s,2H),3.58(h,J=6.6Hz,1H),3.11-2.97(m,1H) ,2.45(dd,J=14.2,7.4Hz,1H),2.25(s,3H),2.01(q,J=8.5,6.9Hz,1H),1.96-1.83(m,1H),1.73(q,J = 7.3, 6.2Hz, 2H), 1.59 (d, J = 14.3Hz, 1H), 1.04 (d, J = 6.6Hz, 6H).
实施例16Example 16
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(甲酰胺基甲基)吡嗪-2-基]氨基}-2H-吡唑-3-基)环戊基酯(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[5-(formamidomethyl)pyrazin-2-yl]amino}-2H-pyrazole-3- base) cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物16。Compound 16 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):388.4[M+H] +。 MS m/z (ESI): 388.4 [M+H] + .
1H NMR(400MHz,DMSO):δ11.93(s,1H),9.44(s,1H),8.51(s,1H),8.02(d,J=1.4Hz,1H),7.43(s,1H),6.93(s,2H),6.15(s,1H),5.00(s,1H),3.63-3.54(m,1H),3.46(s,1H),3.10-3.01(m,1H),2.06-1.83(m,3H),1.81-1.55(m,4H),1.04(d,J=6.4Hz,6H)。 1 H NMR(400MHz,DMSO):δ11.93(s,1H),9.44(s,1H),8.51(s,1H),8.02(d,J=1.4Hz,1H),7.43(s,1H) ,6.93(s,2H),6.15(s,1H),5.00(s,1H),3.63-3.54(m,1H),3.46(s,1H),3.10-3.01(m,1H),2.06-1.83 (m, 3H), 1.81-1.55 (m, 4H), 1.04 (d, J=6.4Hz, 6H).
实施例17Example 17
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[6-(氰基甲基)吡嗪-2-基]氨基}-2H-吡唑-3-基)环戊基酯(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[6-(cyanomethyl)pyrazin-2-yl]amino}-2H-pyrazol-3-yl ) cyclopentyl ester
第一步first step
2-(6-溴吡嗪-2-基)-2-氰基乙酸-2-甲基丙-2-基酯17b2-(6-Bromopyrazin-2-yl)-2-cyanoacetic acid-2-methylpropan-2-yl ester 17b
首先将2,6-二溴吡嗪(1g,4.2mmol)溶于N,N-二甲基甲酰胺(40ml)中,随后加入2-氰基乙酸叔丁酯(1.2g,8.4mmol)、碳酸铯(3.4g,10.5mmol),室温反应4小时。经萃取、干燥、浓缩后得到粗产品,该粗产物可直接用于下一步反应。First, 2,6-dibromopyrazine (1 g, 4.2 mmol) was dissolved in N, N-dimethylformamide (40 ml), followed by the addition of tert-butyl 2-cyanoacetate (1.2 g, 8.4 mmol), Cesium carbonate (3.4g, 10.5mmol) was reacted at room temperature for 4 hours. The crude product is obtained after extraction, drying and concentration, which can be directly used in the next reaction.
MS m/z(ESI):298.2[M+H] +。 MS m/z (ESI): 298.2 [M+H] + .
第二步second step
(6-溴吡嗪-2-基)乙腈17c(6-Bromopyrazin-2-yl)acetonitrile 17c
首先将所得粗产品2-(6-溴吡嗪-2-基)-2-氰基乙酸-2-甲基丙-2-基酯溶于二氯甲烷(20ml)中,随后加入过量的三氟乙酸(4ml),室温反应1小时。经萃取、干燥、浓缩后得到粗产品,正相柱层析分离得到(6-溴吡嗪-2-基)乙腈17c(250mg,30%)。Firstly, the resulting crude product 2-(6-bromopyrazin-2-yl)-2-cyanoacetic acid-2-methylpropan-2-yl ester was dissolved in dichloromethane (20ml), then an excess of tris Fluoroacetic acid (4ml), react at room temperature for 1 hour. The crude product was obtained after extraction, drying and concentration, and (6-bromopyrazin-2-yl)acetonitrile 17c (250 mg, 30%) was separated by normal phase column chromatography.
MS m/z(ESI):198.2[M+H] +。 MS m/z (ESI): 198.2 [M+H] + .
第三步third step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[6-(氰基甲基)吡嗪-2-基]氨基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯17e(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[6-(cyanomethyl)pyrazin-2-yl]amino}-1-(2-methylpropane -2-yl)pyrazol-3-yl)cyclopentyl ester 17e
氮气氛下,依次将(6-溴吡嗪-2-基)乙腈(50mg,0.25mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(77mg,0.25mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、XantPhos(8.7mg 0.015mmol)、Pd2dba3(11.4mg,0.0125mmol)、碳酸铯(81.2mg,0.25mmol)溶于二氧六环(2mL)中,微波120℃条件下反应1小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后正相分离后得到标题化合物17e(38mg,产率36%)。Under nitrogen atmosphere, (6-bromopyrazin-2-yl) acetonitrile (50mg, 0.25mmol), (1R,3S)-3-(5-amino-1-(tert-butyl)-1H-pyridine Azol-3-yl) cyclopentyl isopropyl carbamate (77mg, 0.25mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), XantPhos (8.7mg 0.015mmol), Pd2dba3 ( 11.4mg, 0.0125mmol), cesium carbonate (81.2mg, 0.25mmol) were dissolved in dioxane (2mL), and microwaved at 120°C for 1 hour. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected, concentrated under reduced pressure and separated from the normal phase to obtain the title compound 17e (38 mg, yield 36%).
MS m/z(ESI):426.4[M+H] +。 MS m/z (ESI): 426.4 [M+H] + .
第四步the fourth step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[6-(氰基甲基)吡嗪-2-基]氨基}-2H-吡唑-3- 基)环戊基酯17(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[6-(cyanomethyl)pyrazin-2-yl]amino}-2H-pyrazol-3-yl ) cyclopentyl ester 17
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[6-(氰基甲基)吡嗪-2-基]氨基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯(38mg,0.09mmol)溶于3mL甲酸中,在100℃条件下反应1小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物636(9mg,产率28%)。(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[6-(cyanomethyl)pyrazin-2-yl]amino}-1-(2-methylpropane -2-yl)pyrazol-3-yl)cyclopentyl ester (38mg, 0.09mmol) was dissolved in 3mL formic acid and reacted at 100°C for 1 hour. After cooling to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title compound 636 (9 mg, yield 28%).
MS m/z(ESI):370.2[M+H] +。 MS m/z (ESI): 370.2 [M+H] + .
实施例18Example 18
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[6-(甲酰胺基甲基)吡嗪-2-基]氨基}-2H-吡唑-3-基)环戊基酯(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[6-(formamidomethyl)pyrazin-2-yl]amino}-2H-pyrazole-3- base) cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物18。Compound 18 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
1H NMR(400MHz,DMSO):δ11.93(s,1H),9.45(s,1H),8.51(s,1H),8.02(d,J=1.5Hz,1H),7.43(s,1H),6.93(s,1H),6.88(d,J=8.3Hz,1H),6.15(s,1H),5.00(d,J=8.4Hz,1H),3.46(s,2H),3.41-3.36(m,1H),3.13-3.01(m,1H),2.09-1.84(m,3H),1.82-1.54(m,3H),1.01(d,J=6.6Hz,6H)。 1 H NMR(400MHz,DMSO):δ11.93(s,1H),9.45(s,1H),8.51(s,1H),8.02(d,J=1.5Hz,1H),7.43(s,1H) ,6.93(s,1H),6.88(d,J=8.3Hz,1H),6.15(s,1H),5.00(d,J=8.4Hz,1H),3.46(s,2H),3.41-3.36( m, 1H), 3.13-3.01 (m, 1H), 2.09-1.84 (m, 3H), 1.82-1.54 (m, 3H), 1.01 (d, J=6.6Hz, 6H).
实施例19Example 19
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[6-(氰基甲基)-1,2-二氮杂环己熳-3-基]氨基}-2H-吡唑-3-基)环戊基酯(Propan-2-ylamino)methane acid-(1R,3S)-3-(5-{[6-(cyanomethyl)-1,2-diazacyclohexyl-3-yl]amino} -2H-pyrazol-3-yl)cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物19。Compound 19 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):370.2[M+H] +。 MS m/z (ESI): 370.2 [M+H] + .
实施例20Example 20
(丁-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(甲酰胺基甲基)吡嗪-2-基]氨基}-2H-吡唑-3-基)环戊基酯(But-2-ylamino)methane-(1R,3S)-3-(5-{[5-(formamidomethyl)pyrazin-2-yl]amino}-2H-pyrazole-3- base) cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物20。Compound 20 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):402.2[M+H] +。 MS m/z (ESI): 402.2 [M+H] + .
1H NMR(400MHz,DMSO):δ11.94(s,1H),9.54(s,1H),8.45(s,1H),7.83(s,1H),7.54(s,1H),7.00(s,1H),6.94(d,J=7.9Hz,1H),6.18(s,1H),5.01(s,1H),3.63-3.53(m,1H),3.48(s,2H),3.09-3.00(m,1H),2.49-2.41(m,1H),2.07-1.96(m,3H),1.94-1.85(m,1H),1.78-1.57(m,3H),1.06-1.04(m,3H),1.03-1.02(m,3H)。 1 H NMR (400MHz, DMSO): δ11.94(s,1H),9.54(s,1H),8.45(s,1H),7.83(s,1H),7.54(s,1H),7.00(s, 1H), 6.94(d, J=7.9Hz, 1H), 6.18(s, 1H), 5.01(s, 1H), 3.63-3.53(m, 1H), 3.48(s, 2H), 3.09-3.00(m ,1H),2.49-2.41(m,1H),2.07-1.96(m,3H),1.94-1.85(m,1H),1.78-1.57(m,3H),1.06-1.04(m,3H),1.03 -1.02(m,3H).
实施例21Example 21
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({4-[二甲基(氧亚基)-5-甲磷基]苯基}氨基)-2H-吡唑-3-基]环戊基酯(Propan-2-ylamino)methane acid-(1R,3S)-3-[5-({4-[Dimethyl(oxyethylene)-5-methylphosphoryl]phenyl}amino)-2H- Pyrazol-3-yl]cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物21。Compound 21 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):405.2[M+H] +。 MS m/z (ESI): 405.2 [M+H] + .
实施例22Example 22
(1R,3S)-3-(3-((6-氰基哒嗪-3-基)氨基)-1H-吡唑-5-基)环戊基((S)-仲丁基)氨基甲酸酯(1R,3S)-3-(3-((6-cyanopyridazin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentyl((S)-sec-butyl)aminomethyl Ester
第一步first step
(1R,3S)-3-(1-(叔丁基)-5-((6-氰基哒嗪-3-基)氨基)-1H-吡唑-3-基)环戊基((S)-仲丁基)氨基甲酸酯(1R,3S)-3-(1-(tert-butyl)-5-((6-cyanopyridazin-3-yl)amino)-1H-pyrazol-3-yl)cyclopentyl ((S )-sec-butyl)carbamate
将(1R,3S)-3-(5-氨基-1-(叔丁基)-1H-吡唑-3-基)环戊基((S)-仲丁基)氨基甲酸酯(84mg,0.26mmol)溶于N,N-二甲基甲酰胺(10mL)中,0℃下加入氢化钠(13mg,0.31mmol),0℃下搅拌0.5小时,随后加入6-氯哒嗪-3-甲腈22a(36mg,0.26mmol),待反应结束后,将反应液用1mL水洗涤,20mL乙酸乙酯萃取,20mL饱和氯化钠水溶液洗涤,收集有机相,无水硫酸钠干燥,过滤,收集滤液。滤液减压浓缩,残留物用硅胶色谱法纯化得到标题化合物22c(40mg,产率40.9%)。(1R,3S)-3-(5-Amino-1-(tert-butyl)-1H-pyrazol-3-yl)cyclopentyl((S)-sec-butyl)carbamate (84mg, 0.26mmol) was dissolved in N,N-dimethylformamide (10mL), sodium hydride (13mg, 0.31mmol) was added at 0°C, stirred at 0°C for 0.5 hours, then 6-chloropyridazine-3-formazine was added Nitrile 22a (36mg, 0.26mmol), after the reaction was completed, the reaction solution was washed with 1mL of water, extracted with 20mL of ethyl acetate, washed with 20mL of saturated aqueous sodium chloride solution, the organic phase was collected, dried over anhydrous sodium sulfate, filtered, and the filtrate was collected . The filtrate was concentrated under reduced pressure, and the residue was purified by silica gel chromatography to obtain the title compound 22c (40 mg, yield 40.9%).
MS m/z(ESI):426.2[M+H] +。 MS m/z (ESI): 426.2 [M+H] + .
第二步second step
(1R,3S)-3-(3-((6-氰基哒嗪-3-基)氨基)-1H-吡唑-5-基)环戊基((S)-仲丁基)氨基甲酸酯(1R,3S)-3-(3-((6-cyanopyridazin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentyl((S)-sec-butyl)aminomethyl Ester
根据实施例11合成步骤替换相应原料制得化合物22。Compound 22 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):370.2[M+H] +。 MS m/z (ESI): 370.2 [M+H] + .
实施例23Example 23
(1R,3S)-3-(3-((2-氟-4-(N-(氘代甲基)磺酰)苯基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((2-fluoro-4-(N-(deuteromethyl)sulfonyl)phenyl)amino)-1H-pyrazol-5-yl)cyclopentyliso Propyl carbamate
第一步first step
4-溴-3-氟-N-(氘代甲基)苯磺酰胺23b4-Bromo-3-fluoro-N-(deuteromethyl)benzenesulfonamide 23b
将氘代甲胺盐酸盐(154mg,2.2mmol)溶于15mL二氯甲烷中,向体系中加入三乙胺(557mg,5.52mmol),4-二甲氨基吡啶(45mg,0.37mmol),0℃下加入4-溴-3-氟苯磺酰氯(500mg,1.84mmol),室温下搅拌过夜。反应结束后,将反应液减压浓缩,残留物用硅胶色谱法纯化得到标题化合物23b(145mg,产率29.2%)。Deuteromethylamine hydrochloride (154mg, 2.2mmol) was dissolved in 15mL of dichloromethane, triethylamine (557mg, 5.52mmol), 4-dimethylaminopyridine (45mg, 0.37mmol) were added to the system, 0 Add 4-bromo-3-fluorobenzenesulfonyl chloride (500mg, 1.84mmol) at ℃ and stir overnight at room temperature. After the reaction, the reaction solution was concentrated under reduced pressure, and the residue was purified by silica gel chromatography to obtain the title compound 23b (145 mg, yield 29.2%).
MS m/z(ESI):270.9[M+H] +。 MS m/z (ESI): 270.9 [M+H] + .
第二步second step
(1R,3S)-3-(1-(叔丁基)-5-((2-氟-4-(N-(氘代甲基)磺酰)苯基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯23c(1R,3S)-3-(1-(tert-butyl)-5-((2-fluoro-4-(N-(deuteromethyl)sulfonyl)phenyl)amino)-1H-pyrazole- 3-yl) cyclopentyl isopropyl carbamate 23c
根据实施例11合成步骤替换相应原料制得化合物23c。(56mg,产率75.9%)。Compound 23c was prepared according to the synthetic procedure of Example 11 by replacing the corresponding raw materials. (56 mg, yield 75.9%).
MS m/z(ESI):499.2[M+H] +。 MS m/z (ESI): 499.2 [M+H] + .
第三步third step
(1R,3S)-3-(3-((2-氟-4-(N-(氘代甲基)磺酰)苯基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯23(1R,3S)-3-(3-((2-fluoro-4-(N-(deuteromethyl)sulfonyl)phenyl)amino)-1H-pyrazol-5-yl)cyclopentyliso Propyl carbamate 23
根据实施例11合成步骤替换相应原料制得化合物23。(19mg,产率38.2%)。Compound 23 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials. (19 mg, yield 38.2%).
MS m/z(ESI):443.2[M+H] +。 MS m/z (ESI): 443.2 [M+H] + .
1H NMR(400MHz,DMSO-d6):δ11.99(s,1H),8.68(d,J=1.6Hz,1H),8.23-8.19(m,1H),7.47-7.43(m,2H),7.18(s,1H),6.96(d,J=7.2Hz,1H),5.84(s,1H),5.01(d,J=4.0Hz,1H),3.61-3.56(m,1H),3.09-3.05(m,1H),2.06-1.60(m,6H),1.03(d,J=6.4Hz,6H)。 1 H NMR (400MHz, DMSO-d6): δ11.99(s, 1H), 8.68(d, J=1.6Hz, 1H), 8.23-8.19(m, 1H), 7.47-7.43(m, 2H), 7.18(s,1H),6.96(d,J=7.2Hz,1H),5.84(s,1H),5.01(d,J=4.0Hz,1H),3.61-3.56(m,1H),3.09-3.05 (m, 1H), 2.06-1.60 (m, 6H), 1.03 (d, J=6.4Hz, 6H).
实施例24Example 24
(1R,3S)-3-(3-((4-(N-(氘代甲基)磺酰)苯基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((4-(N-(Deuteromethyl)sulfonyl)phenyl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylaminomethyl Ester
根据实施例23合成步骤替换相应原料制得化合物24(19mg,产率38.2%)。Compound 24 (19 mg, yield 38.2%) was obtained by replacing the corresponding raw materials according to the synthetic procedure of Example 23.
MS m/z(ESI):425.2[M+H] +。 MS m/z (ESI): 425.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.91(s,1H),8.93(s,1H),7.55(d,J=8.8Hz,2H),7.41(d,J=8.0Hz,2H),7.02(s,1H),6.94(d,J=7.2Hz,1H),5.71(s,1H),5.00(d,J=2.8Hz,1H),3.61-3.56(m,1H),3.08-3.04(m,1H),2.06-1.61(m,6H),1.03(d,J=6.4Hz,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.91(s, 1H), 8.93(s, 1H), 7.55(d, J=8.8Hz, 2H), 7.41(d, J=8.0Hz, 2H ),7.02(s,1H),6.94(d,J=7.2Hz,1H),5.71(s,1H),5.00(d,J=2.8Hz,1H),3.61-3.56(m,1H),3.08 -3.04 (m, 1H), 2.06-1.61 (m, 6H), 1.03 (d, J=6.4Hz, 6H).
实施例25Example 25
(1R,3S)-3-(3-((6-(N-(甲基-d3)氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-5-基)环戊基异丙氨基甲酸酯(1R,3S)-3-(3-((6-(N-(methyl-d3)aminosulfonyl)pyridin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentyliso Propyl carbamate
第一步first step
5-溴-N-(甲基-d3)吡啶-2-磺酰胺25b5-Bromo-N-(methyl-d3)pyridine-2-sulfonamide 25b
室温下,将氘代甲胺盐酸盐(210mg,3mmol)、DIEA(650mg,5mmol)溶于无水二氯甲烷(10mL)中,澄清后慢慢加入5-溴吡啶-2-磺酰氯(250mg,1mmol),继续搅拌,TLC监测至反应结束后,加入水(10mL)洗涤1次,饱和食盐水(10mL)洗涤1次,收集有机相层无水硫酸钠干燥,旋至小体积直接正相柱层析,得到标题化合物25b(200mg,产率82%)。At room temperature, deuterated methylamine hydrochloride (210mg, 3mmol) and DIEA (650mg, 5mmol) were dissolved in anhydrous dichloromethane (10mL), and after clarification, 5-bromopyridine-2-sulfonyl chloride ( 250mg, 1mmol), continue to stir, TLC monitors until the end of the reaction, add water (10mL) to wash once, and saturated brine (10mL) to wash once, collect the organic phase and dry it over anhydrous sodium sulfate, spin to a small volume and directly normalize Column chromatography gave the title compound 25b (200 mg, yield 82%).
MS m/z(ESI):253.9[M+H] +。 MS m/z (ESI): 253.9 [M+H] + .
第二步second step
(1R,3S)-3-(1-(叔丁基)-5-((6-(N-(甲基-d3)氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-3-基)环戊基异丙氨基甲酸酯25c(1R,3S)-3-(1-(tert-butyl)-5-((6-(N-(methyl-d3)aminosulfonyl)pyridin-3-yl)amino)-1H-pyrazole- 3-yl) cyclopentyl isopropyl carbamate 25c
氮气氛下,依次将5-溴-N-(甲基-d3)吡啶-2-磺酰胺(25.3mg,0.10mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(30.8mg,0.10mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、Pd2(dba)3(9.1mg,0.01mmol)、Xantphos(11.4mg,0.02mmol)、CS2CO3(96.2mg,0.30mmol)溶于1,4-二氧六环(2mL)中,微波80℃条件下反应2小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后正相分离后得到标题化合物25c(16mg,产率32%)。Under nitrogen atmosphere, 5-bromo-N-(methyl-d3)pyridine-2-sulfonamide (25.3 mg, 0.10 mmol), (1R,3S)-3-(5-amino-1-(tert- Butyl)-1H-pyrazol-3-yl)cyclopentylisopropylcarbamate (30.8mg, 0.10mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), Pd2( dba)3 (9.1mg, 0.01mmol), Xantphos (11.4mg, 0.02mmol), CS2CO3 (96.2mg, 0.30mmol) were dissolved in 1,4-dioxane (2mL), and reacted under microwave conditions at 80°C 2 Hour. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected, concentrated under reduced pressure and separated from the normal phase to obtain the title compound 25c (16 mg, yield 32%).
MS m/z(ESI):482.3[M+H] +。 MS m/z (ESI): 482.3 [M+H] + .
第三步third step
(1R,3S)-3-(3-((6-(N-(甲基-d3)氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-5-基)环戊基异丙氨基甲酸酯25(1R,3S)-3-(3-((6-(N-(methyl-d3)aminosulfonyl)pyridin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentyliso Propyl carbamate 25
将25c(16mg,0.03mmol)溶于2mL甲酸中,在80℃条件下反应12小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物25(2.5mg,产率19.6%)。25c (16mg, 0.03mmol) was dissolved in 2mL formic acid and reacted at 80°C for 12 hours. After cooling to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title compound 25 (2.5 mg, yield 19.6%).
MS m/z(ESI):426.2[M+H] +。 MS m/z (ESI): 426.2 [M+H] + .
实施例26Example 26
(1R,3S)-3-(3-((6-(N-甲基氨基磺酰基)吡啶-3-基)氨基)-1H-吡唑-5-基)环戊基((S)-仲丁基)氨基甲酸酯(1R,3S)-3-(3-((6-(N-Methylaminosulfonyl)pyridin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentyl ((S)- sec-butyl) carbamate
根据实施例25合成步骤替换相应原料制得标题化合物26。The title compound 26 was prepared according to the synthesis procedure of Example 25 by replacing the corresponding raw materials.
MS m/z(ESI):437.2[M+H] +。 MS m/z (ESI): 437.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ12.01(s,1H),9.23(s,1H),8.58(d,J=2.6Hz,1H),8.45(s,1H),7.99(d,J=8.6Hz,1H),7.74(d,J=8.7Hz,1H),7.33(q,J=5.0Hz, 1H),6.89(d,J=8.3Hz,1H),5.73(s,1H),5.00(s,1H),3.12-3.03(m,1H),2.47(d,J=4.8Hz,3H),2.01(dp,J=12.9,7.0,6.3Hz,2H),1.91(ddd,J=12.9,8.4,4.3Hz,1H),1.78-1.70(m,2H),1.61(t,J=14.2Hz,1H),1.47(d,J=7.3Hz,1H),1.41-1.30(m,1H),1.02(d,J=6.6Hz,3H),0.83(dt,J=23.5,6.9Hz,3H)。 1 H NMR (400MHz, DMSO-d 6 ): δ12.01(s, 1H), 9.23(s, 1H), 8.58(d, J=2.6Hz, 1H), 8.45(s, 1H), 7.99(d ,J=8.6Hz,1H),7.74(d,J=8.7Hz,1H),7.33(q,J=5.0Hz,1H),6.89(d,J=8.3Hz,1H),5.73(s,1H ),5.00(s,1H),3.12-3.03(m,1H),2.47(d,J=4.8Hz,3H),2.01(dp,J=12.9,7.0,6.3Hz,2H),1.91(ddd, J=12.9,8.4,4.3Hz,1H),1.78-1.70(m,2H),1.61(t,J=14.2Hz,1H),1.47(d,J=7.3Hz,1H),1.41-1.30(m , 1H), 1.02 (d, J=6.6Hz, 3H), 0.83 (dt, J=23.5, 6.9Hz, 3H).
实施例27Example 27
(1R,3S)-3-(3-((5-(N-甲基氨基磺酰基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((5-(N-methylaminosulfonyl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylaminomethyl Ester
根据实施例25合成步骤替换相应原料制得标题化合物27。The title compound 27 was prepared according to the synthesis procedure of Example 25 by replacing the corresponding raw materials.
MS m/z(ESI):423.2[M+H] +。 MS m/z (ESI): 423.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ12.03(s,1H),9.84(s,1H),8.44(d,J=2.5Hz,1H),7.82(dd,J=9.0,2.6Hz,1H),7.34(s,1H),7.26(d,J=5.5Hz,1H),6.94(d,J=7.7Hz,1H),6.18(s,1H),5.01(s,1H),3.63-3.53(m,1H),3.07(t,J=8.9Hz,1H),2.41(d,J=4.8Hz,3H),2.00(p,J=6.9,6.5Hz,1H),1.74(d,J=9.0Hz,1H),1.62(s,1H),1.46(t,J=7.1Hz,1H),1.25(m,4H),1.04(dd,J=6.6,2.2Hz,3H)。 1 H NMR (400MHz, DMSO-d 6 ): δ12.03(s, 1H), 9.84(s, 1H), 8.44(d, J=2.5Hz, 1H), 7.82(dd, J=9.0, 2.6Hz ,1H),7.34(s,1H),7.26(d,J=5.5Hz,1H),6.94(d,J=7.7Hz,1H),6.18(s,1H),5.01(s,1H),3.63 -3.53(m,1H),3.07(t,J=8.9Hz,1H),2.41(d,J=4.8Hz,3H),2.00(p,J=6.9,6.5Hz,1H),1.74(d, J=9.0Hz, 1H), 1.62(s, 1H), 1.46(t, J=7.1Hz, 1H), 1.25(m, 4H), 1.04(dd, J=6.6, 2.2Hz, 3H).
实施例28Example 28
(1R,3S)-3-(3-((6-氰基吡啶-3-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((6-cyanopyridin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate
根据实施例11合成步骤替换相应原料制得化合物28。Compound 28 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):355.2[M+H] +。 MS m/z (ESI): 355.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ12.08(s,1H),9.43(s,1H),8.58(d,J=2.6Hz,1H),7.96(dd,J=8.8,2.7Hz,1H),7.77(d,J=8.7Hz,1H),6.94(d,J=7.8Hz,1H),5.75(s,1H),5.00(dd,J=6.6,3.2Hz,1H),3.58(dt,J=9.8,5.0Hz,1H),3.12-3.01(m,1H),2.09-1.97(m,1H),2.00-1.84(m,1H),1.74(q,J=7.6,4.7Hz,2H),1.61(s,1H),1.24(d,J=3.7Hz,1H),1.04(dd,J=6.6,2.3Hz,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ12.08(s, 1H), 9.43(s, 1H), 8.58(d, J=2.6Hz, 1H), 7.96(dd, J=8.8, 2.7Hz ,1H),7.77(d,J=8.7Hz,1H),6.94(d,J=7.8Hz,1H),5.75(s,1H),5.00(dd,J=6.6,3.2Hz,1H),3.58 (dt,J=9.8,5.0Hz,1H),3.12-3.01(m,1H),2.09-1.97(m,1H),2.00-1.84(m,1H),1.74(q,J=7.6,4.7Hz , 2H), 1.61 (s, 1H), 1.24 (d, J=3.7Hz, 1H), 1.04 (dd, J=6.6, 2.3Hz, 6H).
实施例29Example 29
(1R,3S)-3-(3-((5-氰基吡啶-3-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((5-cyanopyridin-3-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate
根据实施例11合成步骤替换相应原料制得化合物29。Compound 29 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):355.2[M+H] + MS m/z(ESI): 355.2[M+H] +
1H NMR(400MHz,DMSO-d 6):δ11.97(s,1H),9.15(s,1H),8.70(d,J=2.5Hz,1H),8.34-8.27(m,2H),6.94(d,J=7.7Hz,1H),5.70(d,J=1.9Hz,1H),5.00(s,1H),3.62-3.53(m,1H),3.12-3.02(m,1H),2.09-1.84(m,1H),1.73(s,1H),1.61(s,1H),1.46(s,1H),1.24(s,2H),1.04(d,J=6.5Hz,6H). 1 H NMR (400MHz, DMSO-d 6 ): δ11.97(s, 1H), 9.15(s, 1H), 8.70(d, J=2.5Hz, 1H), 8.34-8.27(m, 2H), 6.94 (d,J=7.7Hz,1H),5.70(d,J=1.9Hz,1H),5.00(s,1H),3.62-3.53(m,1H),3.12-3.02(m,1H),2.09- 1.84(m,1H),1.73(s,1H),1.61(s,1H),1.46(s,1H),1.24(s,2H),1.04(d,J=6.5Hz,6H).
实施例30Example 30
(1R,3S)-3-(3-((4-(甲磺酰基)苯基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((4-(methylsulfonyl)phenyl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylcarbamate
根据实施例11合成步骤替换相应原料制得化合物30。Compound 30 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):407.2[M+H] +。 MS m/z (ESI): 407.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.95(s,1H),9.06(s,1H),7.67(d,J=8.8Hz,2H),7.50-7.43(m,2H),6.94(d,J=6.8Hz,1H),5.72(s,1H),5.01(s,1H),3.57(s,1H),3.08(s,3H),2.03(dt,J=11.0,7.5Hz,1H),1.99-1.84(m,1H),1.73(d,J=8.5Hz,2H),1.61(s,1H),1.24(s,1H),1.04(dd,J=6.7,2.2Hz,6H)。 1 H NMR(400MHz,DMSO-d 6 ):δ11.95(s,1H),9.06(s,1H),7.67(d,J=8.8Hz,2H),7.50-7.43(m,2H),6.94 (d,J=6.8Hz,1H),5.72(s,1H),5.01(s,1H),3.57(s,1H),3.08(s,3H),2.03(dt,J=11.0,7.5Hz, 1H), 1.99-1.84(m, 1H), 1.73(d, J=8.5Hz, 2H), 1.61(s, 1H), 1.24(s, 1H), 1.04(dd, J=6.7, 2.2Hz, 6H ).
实施例31Example 31
(1R,3S)-3-(3-((5-((甲磺酰基)甲基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯(1R,3S)-3-(3-((5-((methylsulfonyl)methyl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylaminomethyl Ester
第一步first step
2-氯-5-((甲硫基)甲基)吡啶31b2-Chloro-5-((methylthio)methyl)pyridine 31b
室温下,将2-氯-5-(氯甲基)吡啶(1000mg,6.2mmol)溶于95%乙醇(10mL)中,澄清后慢慢加入甲硫醇钠(700mg,10mmol),继续搅拌,TLC监测至反应结束后,加入水(10mL)洗涤1次,饱和食盐水(10mL)洗涤1次,收集有机相层无水硫酸钠干燥,减压旋去溶剂,柱层析后得到化合物31b(800mg,产率74%)。Dissolve 2-chloro-5-(chloromethyl)pyridine (1000mg, 6.2mmol) in 95% ethanol (10mL) at room temperature, add sodium methylthiolate (700mg, 10mmol) slowly after clarification, and continue stirring, After TLC monitoring to the end of the reaction, water (10 mL) was added to wash once, saturated brine (10 mL) was washed once, the organic phase was collected and dried over anhydrous sodium sulfate, the solvent was spin-off under reduced pressure, and compound 31b was obtained after column chromatography ( 800 mg, yield 74%).
MS m/z(ESI):174.0[M+H] +。 MS m/z (ESI): 174.0 [M+H] + .
第二步second step
2-氯-5-((甲磺酰基)甲基)吡啶31c2-Chloro-5-((methylsulfonyl)methyl)pyridine 31c
室温下,将2-氯-5-((甲硫基)甲基)吡啶(800mg,4.5mmol)溶于二氯甲烷(10mL)中,澄清后慢慢加入m-CPBA(1100mg,6.75mmol),继续搅拌,TLC监测至反应结束后,滤除沉淀,加入水(10mL)洗涤1次,饱和食盐水(10mL)洗涤1次,收集有机相层,无水硫酸钠干燥,减压旋去溶剂,柱层析后得到化合物31c(800mg,产率85%)。Dissolve 2-chloro-5-((methylthio)methyl)pyridine (800mg, 4.5mmol) in dichloromethane (10mL) at room temperature, add m-CPBA (1100mg, 6.75mmol) slowly after clarification , continue to stir, TLC monitors until the end of the reaction, filter the precipitate, add water (10mL) to wash once, and saturated brine (10mL) to wash once, collect the organic layer, dry over anhydrous sodium sulfate, and spin off the solvent under reduced pressure , Compound 31c (800 mg, yield 85%) was obtained after column chromatography.
MS m/z(ESI):206.0[M+H] +。 MS m/z (ESI): 206.0 [M+H] + .
第三步third step
(1R,3S)-3-(1-(叔丁基)-5-((5-((甲磺酰)甲基)吡啶-2-基)氨基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯31d(1R,3S)-3-(1-(tert-butyl)-5-((5-((methylsulfonyl)methyl)pyridin-2-yl)amino)-1H-pyrazol-3-yl) Cyclopentyl isopropyl carbamate 31d
氮气氛下,依次将2-氯-5-((甲磺酰基)甲基)吡啶(20.6mg,0.10mmol)、(1R,3S)-3-(5-氨基-1-(叔-丁基)-1H-吡唑-3-基)环戊基异丙基氨基甲酸酯(30.8mg,0.10mmol,采用专利申请“WO 2020/157652 A2”公开的方法制备而得)、Pd 2(dba) 3(9.1mg,0.01mmol)、Xantphos(11.4mg,0.02mmol)、CS 2CO 3(96.2mg,0.30mmol)溶于1,4-二氧六环(2mL)中,微波80℃条件下反应2小时。反应液冷却至室温,过滤,收集滤液,减压浓缩后正相分离后得到标题化合物31d(5mg,产率10%)。 Under nitrogen atmosphere, 2-chloro-5-((methylsulfonyl)methyl)pyridine (20.6mg, 0.10mmol), (1R,3S)-3-(5-amino-1-(tert-butyl )-1H-pyrazol-3-yl)cyclopentylisopropylcarbamate (30.8mg, 0.10mmol, prepared by the method disclosed in the patent application "WO 2020/157652 A2"), Pd 2 (dba ) 3 (9.1mg, 0.01mmol), Xantphos (11.4mg, 0.02mmol), CS 2 CO 3 (96.2mg, 0.30mmol) were dissolved in 1,4-dioxane (2mL), and microwaved at 80°C React for 2 hours. The reaction solution was cooled to room temperature, filtered, and the filtrate was collected, concentrated under reduced pressure and separated from the normal phase to obtain the title compound 31d (5 mg, yield 10%).
MS m/z(ESI):478.3[M+H] +。 MS m/z (ESI): 478.3 [M+H] + .
第四步the fourth step
(1R,3S)-3-(3-((5-((甲磺酰基)甲基)吡啶-2-基)氨基)-1H-吡唑-5-基)环戊基异丙基氨基甲酸酯31(1R,3S)-3-(3-((5-((methylsulfonyl)methyl)pyridin-2-yl)amino)-1H-pyrazol-5-yl)cyclopentylisopropylaminomethyl Ester 31
将(1R,3S)-3-(1-(叔丁基)-5-((5-((甲磺酰)甲基)吡啶-2-基)氨基)-1H-吡唑-3-基)环 戊基异丙基氨基甲酸酯(5mg,0.01mmol)溶于1mL甲酸中,在80℃条件下反应12小时。冷却至室温,反应液用C-18反相色谱法纯化得到标题化合物31(1.2mg,产率28.3%)。(1R,3S)-3-(1-(tert-butyl)-5-((5-((methylsulfonyl)methyl)pyridin-2-yl)amino)-1H-pyrazol-3-yl ) cyclopentyl isopropyl carbamate (5 mg, 0.01 mmol) was dissolved in 1 mL of formic acid, and reacted at 80° C. for 12 hours. After cooling to room temperature, the reaction solution was purified by C-18 reverse phase chromatography to obtain the title compound 31 (1.2 mg, yield 28.3%).
MS m/z(ESI):422.2[M+H] +。 MS m/z (ESI): 422.2 [M+H] + .
实施例32Example 32
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(4-甲酰胺基苯基)氨基]-1H-吡唑-3-基}环戊基(Propan-2-ylamino)methanoic acid-(1R,3S)-3-{5-[(4-formamidophenyl)amino]-1H-pyrazol-3-yl}cyclopentyl 酯ester
根据实施例8合成步骤替换相应原料制得化合物32。Compound 32 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
MS m/z(ESI):372.2[M+H] +。 MS m/z (ESI): 372.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.81(s,1H),8.66(s,1H),7.71(d,J=8.8Hz,2H),7.61(s,1H),7.30(d,J=8.3Hz,2H),6.94(d,J=6.9Hz,2H),5.67(s,1H),5.08–4.95(m,1H),3.76-3.51(m,1H),3.16-2.95(m,1H),2.47-2.40(m,1H),2.09-1.97(m,1H),1.93-1.85(m,1H),1.80-1.64(m,2H),1.64-1.58(m,1H),1.08-1.00(m,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.81(s, 1H), 8.66(s, 1H), 7.71(d, J=8.8Hz, 2H), 7.61(s, 1H), 7.30(d ,J=8.3Hz,2H),6.94(d,J=6.9Hz,2H),5.67(s,1H),5.08–4.95(m,1H),3.76-3.51(m,1H),3.16-2.95( m,1H),2.47-2.40(m,1H),2.09-1.97(m,1H),1.93-1.85(m,1H),1.80-1.64(m,2H),1.64-1.58(m,1H), 1.08-1.00(m,6H).
实施例33Example 33
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({2-氟-4-[(甲基氨基)二氧亚基-6-硫基]苯基}氨(Propan-2-ylamino)methanoic acid-(1R,3S)-3-[5-({2-fluoro-4-[(methylamino)dioxylidene-6-sulfanyl]phenyl}ammonia 基)-1H-吡唑-3-基]环戊基酯Base)-1H-pyrazol-3-yl]cyclopentyl ester
根据实施例8合成步骤替换相应原料制得化合物33。Compound 33 was prepared according to the synthetic steps of Example 8 by replacing the corresponding raw materials.
MS m/z(ESI):440.2[M+H] +。 MS m/z (ESI): 440.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.99(s,1H),8.69(s,1H),8.21(t,J=8.7Hz,1H),7.54-7.41(m,2H),7.26–7.15(m,1H),6.94(d,J=7.8Hz,1H),5.84(s,1H),5.03-4.98(m,1H),3.60-3.55(m,1H),3.14-3.00(m,1H),2.49-2.43(m,1H),2.39(d,J=5.1Hz,3H),2.10-1.84(m,2H),1.76-1.65(m,2H),1.64-1.54(m,1H),1.04(d,J=6.8,2.1Hz,6H)。 1 H NMR(400MHz,DMSO-d 6 ):δ11.99(s,1H),8.69(s,1H),8.21(t,J=8.7Hz,1H),7.54-7.41(m,2H),7.26 –7.15(m,1H),6.94(d,J=7.8Hz,1H),5.84(s,1H),5.03-4.98(m,1H),3.60-3.55(m,1H),3.14-3.00(m ,1H),2.49-2.43(m,1H),2.39(d,J=5.1Hz,3H),2.10-1.84(m,2H),1.76-1.65(m,2H),1.64-1.54(m,1H ), 1.04 (d, J=6.8, 2.1 Hz, 6H).
实施例34Example 34
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(4-{[(2-羟基乙基)氨基]二氧亚基-6-硫基}苯基)(Propan-2-ylamino)methane-(1R,3S)-3-{5-[(4-{[(2-hydroxyethyl)amino]dioxylidene-6-thio}phenyl) 氨基]-1H-吡唑-3-基}环戊基酯Amino]-1H-pyrazol-3-yl}cyclopentyl ester
根据实施例8合成步骤替换相应原料制得化合物34。Compound 34 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
MS m/z(ESI):452.2[M+H] +。 MS m/z (ESI): 452.2 [M+H] + .
实施例35Example 35
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(5-氰基嘧啶-2-基)氨基]-1H-吡唑-3-基}环戊基(Propan-2-ylamino)methanoic acid-(1R,3S)-3-{5-[(5-cyanopyrimidin-2-yl)amino]-1H-pyrazol-3-yl}cyclopentyl 酯ester
根据实施例8合成步骤替换相应原料制得化合物35。Compound 35 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
MS m/z(ESI):356.2[M+H] +。 MS m/z (ESI): 356.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ12.21(s,1H),9.74(s,1H),8.94(s,2H),6.94(s,1H),5.79(s,1H),5.00(s,1H),3.62-3.55(m,1H),3.13-3.04(m,1H),2.48-2.38(m,1H),2.09-1.94(m,1H),1.97-1.86(m,1H),1.77-1.69(m,2H),1.64-1.59(m,1H),1.04(d,J=6.5Hz,6H)。 1 H NMR(400MHz,DMSO-d 6 ):δ12.21(s,1H),9.74(s,1H),8.94(s,2H),6.94(s,1H),5.79(s,1H),5.00 (s,1H),3.62-3.55(m,1H),3.13-3.04(m,1H),2.48-2.38(m,1H),2.09-1.94(m,1H),1.97-1.86(m,1H) , 1.77-1.69 (m, 2H), 1.64-1.59 (m, 1H), 1.04 (d, J=6.5Hz, 6H).
实施例36Example 36
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(5-甲酰胺基吡嗪-2-基)氨基]-2H-吡唑-3-基}环(Propan-2-ylamino)methanoic acid-(1R,3S)-3-{5-[(5-formamidopyrazin-2-yl)amino]-2H-pyrazol-3-yl}ring 戊基酯Amyl esters
根据实施例11合成步骤替换相应原料和试剂制得化合物36。Compound 36 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials and reagents.
MS m/z(ESI):374.2[M+H] +。 MS m/z (ESI): 374.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ12.12(s,1H),10.14(s,1H),8.67(s,1H),8.47(s,1H),7.77(s,1H),7.40(s,1H),6.95(d,J=7.7Hz,1H),6.29(s,1H),5.04-4.99(m, 1H),3.65-3.52(m,1H),3.13-3.04(m,1H),2.48–2.42(m,1H),2.09-1.99(m,1H),1.98-1.83(m,1H),1.78-1.71(m,2H),1.65-1.60(m,1H),1.04(d,J=6.6,2.4Hz,6H)。 1 H NMR(400MHz,DMSO-d 6 ):δ12.12(s,1H),10.14(s,1H),8.67(s,1H),8.47(s,1H),7.77(s,1H),7.40 (s,1H),6.95(d,J=7.7Hz,1H),6.29(s,1H),5.04-4.99(m,1H),3.65-3.52(m,1H),3.13-3.04(m,1H ),2.48–2.42(m,1H),2.09-1.99(m,1H),1.98-1.83(m,1H),1.78-1.71(m,2H),1.65-1.60(m,1H),1.04(d , J=6.6, 2.4Hz, 6H).
实施例37Example 37
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[5-(甲酰胺基甲基)吡啶-2-基]氨基}-2H-吡唑-3-(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[5-(formamidomethyl)pyridin-2-yl]amino}-2H-pyrazole-3- 基)环戊基酯base) cyclopentyl ester
根据实施例11合成步骤替换相应原料和试剂制得化合物37。Compound 37 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials and reagents.
MS m/z(ESI):387.3[M+H] +。 MS m/z (ESI): 387.3 [M+H] + .
实施例38Example 38
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-({4-[二氧亚基(丙酰基氨基)-6-硫基]苯基}氨基)-2H-吡唑-3-基]环戊基酯(Propan-2-ylamino)methane acid-(1R,3S)-3-[5-({4-[Dioxyylidene(propionylamino)-6-thio]phenyl}amino)-2H- Pyrazol-3-yl]cyclopentyl ester
根据实施例11合成步骤替换相应原料和试剂制得化合物38。Compound 38 was prepared according to the synthetic steps of Example 11 by replacing corresponding raw materials and reagents.
MS m/z(ESI):464.2[M+H] +。 MS m/z (ESI): 464.2 [M+H] + .
实施例39Example 39
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(4-氰基嘧啶-2-基)氨基]-2H-吡唑-3-基}环戊基(Propan-2-ylamino)methanoic acid-(1R,3S)-3-{5-[(4-cyanopyrimidin-2-yl)amino]-2H-pyrazol-3-yl}cyclopentyl 酯ester
根据实施例11合成步骤替换相应原料和试剂制得化合物39。Compound 39 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials and reagents.
MS m/z(ESI):356.2[M+H] +。 MS m/z (ESI): 356.2 [M+H] + .
实施例40Example 40
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[1-(甲基二氧亚基-6-硫基)六氢吡啶-4-基]氨(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[1-(methyldioxyylidene-6-thio)hexahydropyridin-4-yl]ammonia 基}-2H-吡唑-3-基)环戊基酯Base}-2H-pyrazol-3-yl)cyclopentyl ester
第一步first step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[1-(甲基二氧亚基-6-硫基)六氢吡啶-4-基]氨(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[1-(methyldioxyylidene-6-thio)hexahydropyridin-4-yl]ammonia 基}-1-(2-甲基丙-2-基)吡唑-3-基)环戊基酯40bBase}-1-(2-methylprop-2-yl)pyrazol-3-yl)cyclopentyl ester 40b
将化合物1-N-甲磺酰基-4-哌啶酮(178mg,1.0mmol),1a(310mg,1.0mmol),溶于二氯甲烷(10mL)中,加入干燥的4A分子筛室温搅拌1小时。加入冰乙酸(1滴),氰基硼氢化钠(124mg,2.0mmol)室温反应过夜。向反应液中加入水(30mL),用二氯甲烷(20mL×2)萃取。合并有机相并用无水Na 2SO 4干燥,过滤,真空减压浓缩得到化合物粗品。粗品通过C18纯化得到化合物40b(234mg,0.5mmol,产率:50%)。MS(ESI)m/z 470.3[M+H] Compound 1-N-methylsulfonyl-4-piperidone (178 mg, 1.0 mmol), 1a (310 mg, 1.0 mmol) was dissolved in dichloromethane (10 mL), added to dry 4A molecular sieves and stirred at room temperature for 1 hour. Add glacial acetic acid (1 drop), sodium cyanoborohydride (124 mg, 2.0 mmol) and react overnight at room temperature. Water (30 mL) was added to the reaction liquid, and extracted with dichloromethane (20 mL×2). The organic phases were combined and dried over anhydrous Na 2 SO 4 , filtered, and concentrated in vacuo to obtain the crude compound. The crude product was purified by C18 to obtain compound 40b (234 mg, 0.5 mmol, yield: 50%). MS(ESI)m/z 470.3[M+H]
第二步second step
(丙-2-基氨基)甲烷酸-(1R,3S)-3-(5-{[1-(甲基二氧亚基-6-硫基)六氢吡啶-4-基]氨(Propan-2-ylamino)methanoic acid-(1R,3S)-3-(5-{[1-(methyldioxyylidene-6-thio)hexahydropyridin-4-yl]ammonia 基}-2H-吡唑-3-基)环戊基酯40Base}-2H-pyrazol-3-yl)cyclopentyl ester 40
根据实施例8合成步骤替换相应原料制得化合物40。Compound 40 was prepared according to the synthesis steps of Example 8 by replacing the corresponding raw materials.
MS m/z(ESI):414.3[M+H] +。 MS m/z (ESI): 414.3 [M+H] + .
实施例41Example 41
(丙-2-基氨基)甲烷酸-(1R,3S)-3-[5-(1,2-二氮杂环己熳-3-基氨基)-2H-吡唑-3-基]环(Propan-2-ylamino)methane-(1R,3S)-3-[5-(1,2-diazacyclohexyl-3-ylamino)-2H-pyrazol-3-yl]ring 戊基酯Amyl esters
根据实施例11合成步骤替换相应原料制得化合物41。Compound 41 was prepared according to the synthetic steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):331.2[M+H] +。 MS m/z (ESI): 331.2 [M+H] + .
1H NMR(400MHz,DMSO-d 6):δ11.94(s,1H),9.51(s,1H),8.59(d,1H),7.54(d,J=9.1Hz,1H),7.44-7.35(m,1H),6.95(d,J=7.7Hz,1H),6.22(s,1H),5.05-4.98 (m,1H),3.65-3.53(m,1H),3.11-3.02(m,1H),2.49-2.43(m,1H),2.07-1.97(m,1H),1.98-1.83(m,1H),1.80-1.67(m,2H),1.65-1.60(m,1H),1.04(d,J=6.7,2.0Hz,6H)。 1 H NMR (400MHz, DMSO-d 6 ): δ11.94(s, 1H), 9.51(s, 1H), 8.59(d, 1H), 7.54(d, J=9.1Hz, 1H), 7.44-7.35 (m,1H),6.95(d,J=7.7Hz,1H),6.22(s,1H),5.05-4.98(m,1H),3.65-3.53(m,1H),3.11-3.02(m,1H ),2.49-2.43(m,1H),2.07-1.97(m,1H),1.98-1.83(m,1H),1.80-1.67(m,2H),1.65-1.60(m,1H),1.04(d , J=6.7, 2.0Hz, 6H).
实施例42Example 42
(丙-2-基氨基)甲烷酸-(1R,3S)-3-{5-[(5-氰基-4-甲基吡啶-2-基)氨基]-2H-吡唑-3-基}(Propan-2-ylamino)methanoic acid-(1R,3S)-3-{5-[(5-cyano-4-methylpyridin-2-yl)amino]-2H-pyrazol-3-yl } 环戊基酯Cyclopentyl ester
根据实施例11合成步骤替换相应原料制得化合物42。Compound 42 was prepared according to the synthesis steps of Example 11 by replacing the corresponding raw materials.
MS m/z(ESI):369.2[M+H] +。 MS m/z (ESI): 369.2 [M+H] + .
生物学评价biological evaluation
以下结合测试例进一步描述解释本公开,但这些实施例并非意味着限制本公开的范围。The following further describes and explains the present disclosure in combination with test examples, but these examples are not meant to limit the scope of the present disclosure.
测试例1.本公开化合物对卵巢癌细胞(OVCAR3)抑制活性测试Test Example 1. Test of the Inhibitory Activity of the Compounds of the Disclosure on Ovarian Cancer Cells (OVCAR3)
表1.实验材料和仪器Table 1. Experimental materials and instruments
1.实验步骤1. Experimental steps
卵巢癌细胞OVCAR3使用10%FBS的RPMI 1640培养在37%,5%CO 2的细胞培养箱中培养。第一天,细胞铺板在96-孔板中,铺板细胞浓度为2500个细胞/孔,在培养箱中培养过夜。第二天进行化合物处理,化合物处理最高浓度为10uM,3倍稀释,9个浓度,DMSO的终浓度为0.1%。细胞继续在培养箱中培养7天后,使用Celltiter Glo assay kit(Promega)测试细胞活力,测试方法与试剂盒提供的操作方法保持一致。使用GraphPad Prism 8处理数据并计算IC 50。 Ovarian cancer cells OVCAR3 were cultured in RPMI 1640 with 10% FBS in a 37%, 5% CO2 incubator. On the first day, cells were plated in 96-well plates at a concentration of 2500 cells/well and incubated overnight in an incubator. Compound treatment was performed on the second day, the highest concentration of compound treatment was 10 uM, 3-fold dilution, 9 concentrations, and the final concentration of DMSO was 0.1%. After the cells continued to be cultured in the incubator for 7 days, the cell viability was tested using the Celltiter Glo assay kit (Promega), and the test method was consistent with the operation method provided by the kit. Data were processed and IC50 calculated using GraphPad Prism 8.
计算公式Y=Bottom+(Top-Bottom)/(1+10^((LogIC50-X)*HillSlope))。Calculation formula Y=Bottom+(Top-Bottom)/(1+10^((LogIC50-X)*HillSlope)).
X:化合物浓度的对数值;Y:%抑制。X: log value of compound concentration; Y: % inhibition.
本公开化合物对OVCAR3的IC 50(nM),A≤400nM,400nM<B≤600nM,600nM<C≤1000nM。 The IC 50 (nM) of the disclosed compound on OVCAR3, A≤400nM, 400nM<B≤600nM, 600nM<C≤1000nM.
表2.本公开化合物的对OVCAR3的IC 50(nM) Table 2. IC 50 (nM) for OVCAR3 of Compounds of the Disclosure
注:根据WO2022174031A的实施例32、实施例402和实施例563分别合成 得到化合物EX32、EX402和EX563。Note: Compounds EX32, EX402 and EX563 were synthesized respectively according to Example 32, Example 402 and Example 563 of WO2022174031A.
测试例2.本公开化合物对细胞周期蛋白-依赖性激酶(CDK1,2,9)活性检测。Test Example 2. Detection of the activity of the disclosed compounds on cyclin-dependent kinases (CDK1, 2, 9).
2.1实验材料及仪器(见表3)2.1 Experimental materials and instruments (see Table 3)
表3.实验材料和仪器Table 3. Experimental materials and instruments
2.2实验步骤2.2 Experimental steps
使用Echo 550将化合物稀释液转移到测定板的每个孔中(784075,Greiner)。密封测定板,以1000g离心测定板1分钟;在1x激酶缓冲液((40mM Tris-HCl,pH7.4,20mM MgCl2,0.1mg/ml BSA,50uM DTT)中准备2x酶,将2.5μl 2x酶加入384孔测定板,将板在1000g下离心30s,在室温放置10分钟。在1x激酶缓冲液中制备2x底物和ATP混合物,加入2.5μl 2x底物和ATP混合物开始反应。将板以1000g离心30秒,密封测定板,室温反应1小时。加入4μl ADP-Glo试剂,在室温下孵育40分钟,再加入8μl激酶检测试剂,在室温下孵育40分钟。Compound dilutions were transferred to each well of the assay plate using an Echo 550 (784075, Greiner). Seal the assay plate and centrifuge the assay plate at 1000g for 1 min; prepare 2x enzyme in 1x kinase buffer ((40mM Tris-HCl, pH 7.4, 20mM MgCl2, 0.1mg/ml BSA, 50uM DTT) by dissolving 2.5 μl of 2x enzyme Add to 384-well assay plate, centrifuge the plate at 1000g for 30s, and let it stand at room temperature for 10 minutes. Prepare 2x substrate and ATP mix in 1x kinase buffer, add 2.5μl 2x substrate and ATP mix to start the reaction. Centrifuge the plate at 1000g Centrifuge for 30 seconds, seal the assay plate, and react at room temperature for 1 hour. Add 4 μl ADP-Glo reagent, incubate at room temperature for 40 minutes, then add 8 μl kinase detection reagent, and incubate at room temperature for 40 minutes.
在Envision 2104读板器上读取每个孔发光信号。The luminescent signal of each well was read on an Envision 2104 plate reader.
抑制百分率计算如下:抑制百分率=100-(cmpd信号-Ave_PC信号)/(Ave_VC信号-Ave_PC信号)×100。cmpd信号:化合物信号;Ave_PC信号:阳性对照信 号平均值;Ave_VC信号:载体组信号平均值。Percent inhibition was calculated as follows: Percent inhibition = 100 - (cmpd signal - Ave_PC signal) / (Ave_VC signal - Ave_PC signal) x 100. cmpd signal: compound signal; Ave_PC signal: positive control signal average; Ave_VC signal: vector group signal average.
使用GraphPad 8.0通过将抑制百分率值和化合物浓度的对数拟合为非线性回归(剂量响应–可变斜率)来计算IC 50。Y=底部+(顶部-底部)/(1+10^((LogIC 50-X)*斜率)),其中X:抑制剂浓度的对数;Y:%抑制。 IC50s were calculated using GraphPad 8.0 by fitting the percent inhibition values and the logarithm of the compound concentration to a non-linear regression (dose response - variable slope). Y=Bottom+(Top-Bottom)/(1+10^(( LogIC50 -X)*Slope)), where X: logarithm of inhibitor concentration; Y: % inhibition.
表4.本公开化合物对CDK1/B、CDK2/Cyclin E、CDK9/Cyclin T1的IC 50值 Table 4. IC 50 values of compounds of the present disclosure to CDK1/B, CDK2/Cyclin E, CDK9/Cyclin T1
注:根据WO2022174031A的实施例32、实施例402和实施例563分别合成得到化合物EX32、EX402和EX563。Note: Compounds EX32, EX402 and EX563 were synthesized according to Example 32, Example 402 and Example 563 of WO2022174031A, respectively.
测试例3.本发明实施例8和实施例9的药代动力学测试Test example 3. the pharmacokinetic test of embodiment 8 of the present invention and embodiment 9
以Balb/c nude mouse为受试动物,应用LC/MS/MS法测定其灌胃给予实施例8和实施例9后不同时刻血浆中的药物浓度。研究本发明的化合物在Balb/c nude mouse体内的药代动力学行为,评价其药动学特征。Taking Balb/c nude mouse as the test animal, the LC/MS/MS method was used to determine the drug concentration in the blood plasma at different times after the oral administration of Example 8 and Example 9. Study the pharmacokinetic behavior of the compound of the present invention in Balb/c nude mouse body, and evaluate its pharmacokinetic characteristics.
3.1实验动物3.1 Experimental animals
健康的雌性Balb/c nude mouse2只,购自集萃药康公司。Two healthy female Balb/c nude mice were purchased from Jizui Yaokang Company.
3.2药物配制3.2 Drug preparation
称取适量的样品,以5%DMSO/40%PEG400/55%saline为溶媒,制得7.5mg/mL溶液。Weigh an appropriate amount of sample, and use 5% DMSO/40% PEG400/55% saline as a solvent to prepare a 7.5 mg/mL solution.
3.3给药3.3 Administration
健康的雌性Balb/c nude mouse 2只,禁食一夜后分别灌胃给药,剂量为75mg/kg,给药体积10mL/kg。Two healthy female Balb/c nude mice were intragastrically administered after overnight fasting, the dose was 75mg/kg, and the administration volume was 10mL/kg.
于给药前及给药后0.25、0.5、1、2、4、8、24小时采血,置于肝素化抗凝试管中,离心分离血浆,低温保存。用LC/MS/MS法测定血浆中的待测化合物含量。血浆样品经过沉淀蛋白预处理后进行分析。Blood was collected before administration and at 0.25, 0.5, 1, 2, 4, 8, and 24 hours after administration, placed in a heparinized anticoagulant test tube, centrifuged to separate plasma, and stored at low temperature. The content of the test compound in plasma was determined by LC/MS/MS method. Plasma samples were pretreated to precipitate proteins for analysis.
3.4药代动力学参数结果3.4 Results of pharmacokinetic parameters
表5.本公开化合物的药代动力学参数如下:Table 5. The pharmacokinetic parameters of the disclosed compounds are as follows:
Claims (23)
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111319542 | 2021-11-09 | ||
| CN202111319542.1 | 2021-11-09 | ||
| CN202210871359.0 | 2022-07-22 | ||
| CN202210871359 | 2022-07-22 | ||
| CN202210998936 | 2022-08-19 | ||
| CN202210998936.2 | 2022-08-19 | ||
| CN202211174775.1 | 2022-09-26 | ||
| CN202211174775 | 2022-09-26 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023083201A1 true WO2023083201A1 (en) | 2023-05-19 |
Family
ID=86335097
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2022/130771 Ceased WO2023083201A1 (en) | 2021-11-09 | 2022-11-09 | Aminopyrazole derivative, and preparation method therefor and use thereof |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202325280A (en) |
| WO (1) | WO2023083201A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023208143A1 (en) * | 2022-04-28 | 2023-11-02 | 正大天晴药业集团股份有限公司 | Pyrazole-substituted cyclopentanol ester derivative and use thereof |
| WO2024056019A1 (en) * | 2022-09-15 | 2024-03-21 | Beigene, Ltd. | Bicyclic compounds as cdk inhibitors |
| WO2024104455A1 (en) * | 2022-11-17 | 2024-05-23 | 山东绿叶制药有限公司 | Cdk2 inhibitor, preparation method therefor, and use thereof |
| US12097261B2 (en) | 2021-05-07 | 2024-09-24 | Kymera Therapeutics, Inc. | CDK2 degraders and uses thereof |
| WO2024217431A1 (en) * | 2023-04-17 | 2024-10-24 | 正大天晴药业集团股份有限公司 | Cyclopentylpyrazolamine derivative and use thereof |
| WO2025041076A1 (en) * | 2023-08-24 | 2025-02-27 | Beigene Switzerland Gmbh | Bicyclic compounds as cdk inhibitors |
| WO2025237418A1 (en) * | 2024-05-17 | 2025-11-20 | 山东绿叶制药有限公司 | Salt and crystal form of cdk2 inhibitor, and preparation method therefor and use thereof |
| WO2026024674A1 (en) | 2024-07-22 | 2026-01-29 | Genesis Therapeutics, Inc. | Methods of treating skp2-associated cancers |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1518543A (en) * | 2000-08-31 | 2004-08-04 | �Ʒ� | Pyrazole derivatives and their use as protein kinase inhibitors |
| WO2005051919A1 (en) * | 2003-11-26 | 2005-06-09 | Pfizer Products Inc. | Aminopyrazole derivatives as gsk-3 inhibitors |
| CN113330000A (en) * | 2019-01-31 | 2021-08-31 | 辉瑞公司 | 3-carbonylamino-5-cyclopentyl-1 FI-pyrrole compound having inhibitory activity against CDK2 |
| WO2022135365A1 (en) * | 2020-12-22 | 2022-06-30 | Anrui Biomedical Technology (Guangzhou) Co., Ltd. | Disubstituted cyclopentane kinase inhibitors |
| WO2022174031A1 (en) * | 2021-02-12 | 2022-08-18 | Relay Therapeutics, Inc. | Cdk inhibitors and methods of use thereof |
-
2022
- 2022-11-09 WO PCT/CN2022/130771 patent/WO2023083201A1/en not_active Ceased
- 2022-11-09 TW TW111142871A patent/TW202325280A/en unknown
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1518543A (en) * | 2000-08-31 | 2004-08-04 | �Ʒ� | Pyrazole derivatives and their use as protein kinase inhibitors |
| WO2005051919A1 (en) * | 2003-11-26 | 2005-06-09 | Pfizer Products Inc. | Aminopyrazole derivatives as gsk-3 inhibitors |
| CN113330000A (en) * | 2019-01-31 | 2021-08-31 | 辉瑞公司 | 3-carbonylamino-5-cyclopentyl-1 FI-pyrrole compound having inhibitory activity against CDK2 |
| WO2022135365A1 (en) * | 2020-12-22 | 2022-06-30 | Anrui Biomedical Technology (Guangzhou) Co., Ltd. | Disubstituted cyclopentane kinase inhibitors |
| WO2022174031A1 (en) * | 2021-02-12 | 2022-08-18 | Relay Therapeutics, Inc. | Cdk inhibitors and methods of use thereof |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12097261B2 (en) | 2021-05-07 | 2024-09-24 | Kymera Therapeutics, Inc. | CDK2 degraders and uses thereof |
| WO2023208143A1 (en) * | 2022-04-28 | 2023-11-02 | 正大天晴药业集团股份有限公司 | Pyrazole-substituted cyclopentanol ester derivative and use thereof |
| WO2024056019A1 (en) * | 2022-09-15 | 2024-03-21 | Beigene, Ltd. | Bicyclic compounds as cdk inhibitors |
| WO2024104455A1 (en) * | 2022-11-17 | 2024-05-23 | 山东绿叶制药有限公司 | Cdk2 inhibitor, preparation method therefor, and use thereof |
| WO2024217431A1 (en) * | 2023-04-17 | 2024-10-24 | 正大天晴药业集团股份有限公司 | Cyclopentylpyrazolamine derivative and use thereof |
| WO2025041076A1 (en) * | 2023-08-24 | 2025-02-27 | Beigene Switzerland Gmbh | Bicyclic compounds as cdk inhibitors |
| WO2025237418A1 (en) * | 2024-05-17 | 2025-11-20 | 山东绿叶制药有限公司 | Salt and crystal form of cdk2 inhibitor, and preparation method therefor and use thereof |
| WO2026024674A1 (en) | 2024-07-22 | 2026-01-29 | Genesis Therapeutics, Inc. | Methods of treating skp2-associated cancers |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202325280A (en) | 2023-07-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2023083201A1 (en) | Aminopyrazole derivative, and preparation method therefor and use thereof | |
| RU2602233C2 (en) | Substituted quinazine derivatives as fgfr-kinase inhibitors for treatment of cancer | |
| RU2701517C2 (en) | Quinazolinone derivatives applicable as modulators of fgfr kinase | |
| JP6564406B2 (en) | Imidazo-pyridazine derivatives as casein kinase 1 delta / epsilon inhibitors | |
| WO2020125513A1 (en) | Macrocyclic compound as cdk inhibitor, preparation method therefor, and use thereof in medicine | |
| TW202317564A (en) | Cdk2 inhibitor, a preparation method and a use thereof | |
| BR112014010179B1 (en) | compound, pharmaceutical composition, and, use of a compound | |
| JP2003513091A (en) | Pyrazolones substituted with heterocycles | |
| WO2012019427A1 (en) | Phthalazinone ketone derivative, preparation method thereof, and pharmaceutical use thereof | |
| CN101282960A (en) | RAF inhibitor compounds and methods | |
| JP6283688B2 (en) | Novel pyrazole-substituted imidazopyrazine as casein kinase 1D / E inhibitor | |
| WO2021197452A1 (en) | Crystal form of free alkali of nitrogen-containing aromatic derivatives | |
| WO2024074497A1 (en) | Parg inhibitory compound | |
| WO2020200069A1 (en) | Pyrroloheterocyclic derivative, preparation method therefor, and application thereof in medicine | |
| CN107207504A (en) | Phthalazinone derivatives, preparation method and the usage | |
| CN114195771B (en) | Multi-kinase inhibitors and uses thereof | |
| JP2021504334A (en) | Pyrazolopyridinone compound | |
| WO2023051302A1 (en) | Heterocyclic compound having cyclin-dependent kinase inhibitory activity, preparation method therefor and medical use thereof | |
| WO2020238776A1 (en) | Substituted fused bicyclic derivative, preparation method therefor, and application thereof in medicines | |
| TW202246243A (en) | Aminopyrimidine compounds and methods of their use | |
| WO2025087267A1 (en) | Thiazolyl-containing piperidinopyrimidine derivative, preparation method therefor and medical use thereof | |
| CN115843296B (en) | CDK9 inhibitors and uses thereof | |
| WO2025160079A1 (en) | Indazolo-amino-pyrimidinone compounds | |
| TWI548637B (en) | Phthalazinone derivatives, preparation process and pharmaceutical use thereof | |
| WO2025073870A1 (en) | Parg inhibitory compound |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22891995 Country of ref document: EP Kind code of ref document: A1 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22891995 Country of ref document: EP Kind code of ref document: A1 |