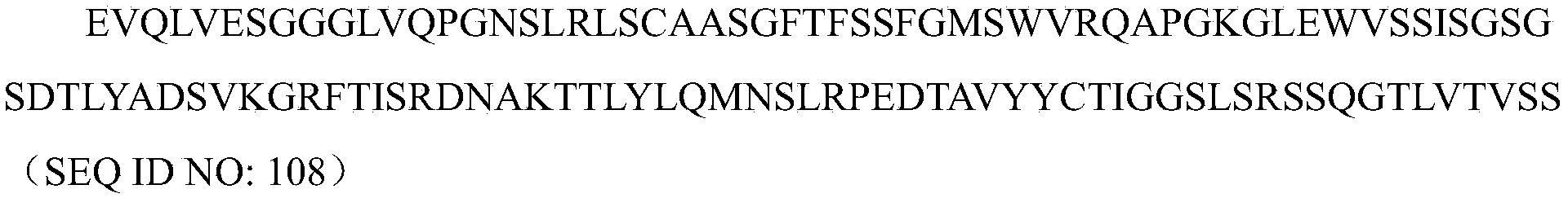WO2023035272A1 - Il17 antibody, preparation method therefor and application thereof - Google Patents
Il17 antibody, preparation method therefor and application thereof Download PDFInfo
- Publication number
- WO2023035272A1 WO2023035272A1 PCT/CN2021/118023 CN2021118023W WO2023035272A1 WO 2023035272 A1 WO2023035272 A1 WO 2023035272A1 CN 2021118023 W CN2021118023 W CN 2021118023W WO 2023035272 A1 WO2023035272 A1 WO 2023035272A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- sequence shown
- antibody according
- cdr2
- cdr1
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
Definitions
- the invention belongs to the field of biomedicine, and in particular relates to an IL17 antibody and its preparation method and application.
- the cytokine IL17 can come from a variety of different immune cells, among which the secretion of Th17 helper T cells is the main source, and CD8 + T cells, NK T cells, LLC3 (Type 3 innate lymphoid cells) and ⁇ T cells can also secrete IL17 (Nat Rev Immunol. 2010, 10, 479).
- the cytokine IL17 protein family mainly includes IL17A, IL17B, IL17C, IL17D, IL17E (IL25) and IL17F, among which IL17A and IL17F have a large sequence similarity and can be expressed simultaneously in Th17 cells.
- IL17A and IL17F can exist as a homodimer, or as an IL17A/F heterodimer composed of IL17A and IL17F monomers (Cold Spring Harb Perspect Biol. 2018, 10, a028522).
- IL17 receptor protein There are multiple subunits of IL17 receptor protein, including five receptor subunits including IL17RA, IL17RB, IL17RC, IL17RD and IL17RE, among which IL17RA is often used as the common core subunit of IL17 receptor and participates in the binding of different IL17 molecules, such as IL17A , IL17F and IL17A/F both bind to the receptor complex composed of IL17RA/IL17RC, IL17C binds to the receptor complex composed of IL17RA/IL17RE, IL17E binds to the receptor complex composed of IL17RA/IL17RB (Cytokine 2013,64,477; Trends Immunol. 2017, 38, 310).
- IL17RA is expressed in both hematopoietic stem cells and non-hematopoietic cells, but IL17RC is commonly expressed in non-hematopoietic stem cells, so dimers such as IL17A, IL17F and IL17A/F activate non-hematopoietic stem cells, such as epithelial cells, by binding to the IL17RA/IL17RC receptor complex cells and stromal cells, etc. After the cells are activated, they produce a series of pro-inflammatory factors, such as cytokines such as IL-1, IL-6, G-CSF and TNF, and also induce the expression of CXCL1, CXCL2, CXCL5, CCL2, CCL7, CCL20 and IL-8.
- cytokines such as IL-1, IL-6, G-CSF and TNF
- cytokine IL17 plays an important role in resisting external bacteria and fungal infections, maintaining the integrity of the mucosa and epidermis, and promoting wound healing (Immunol Rev. 2008, 226, 57). Conversely, once the activity of IL17 is not normally regulated, it will cause chronic inflammation and autoimmune diseases, and may even induce the formation of tumors.
- TIP-DC TNF- ⁇ /iNOS
- TIP-DC cells are activated by binding TNF- ⁇ , and the activated TIP-DC cells Further secrete TNF- ⁇ , IL23, IL12 and other cytokines, IL23 activates Th17 cells, prompts Th17 cells to secrete excessive cytokines such as IL17A, IL17F and IL22, and these cytokines further drive abnormal differentiation and proliferation of keratinocytes, thus forming psoriasis disease (J Am Acad Dermatol.2014,71,141; J Dermatol.2018,45,264; Int J Mol Sci.2020,21,1690). Inhibition of the IL23-IL17 immune axis (Nat. Rev. Immunol.
- Th17 cell subsets are involved in regulating the inflammation of neutrophils and macrophages in the lungs, and are associated with severe neutrophil asthma and chronic obstructive pulmonary disease (Nat.Rev.Immunol.2008,8,183; Ann.Rev. Physiol. 2010, 72, 495).
- Studies have shown that the expression levels of IL17A and IL17F are related to the severity of asthma. The correlation is because IL17 can effectively promote the expression of neutrophil growth factors and chemokines, such as IL-6, G-CSF, IL8, etc., thus Promote the enrichment of neutrophils (Expert Rev. Respir. Med. 2014,8,25).
- IL17 plays a certain connection role in the process of inflammatory response, wound healing and tumor formation (J. Exp. Med. 2019, 217, e20190297; Nat. Immunol. 2019, 20, 1594).
- IL17 promotes the pathogenesis of various cancers, including colon cancer (Immunity, 2014, 41, 1052; J Immunol. 2017, 199, 3849), skin cancer (Cancer Res 2010, 70, 10112; J Exp. Med.2019,216,195), pancreatic cancer (Gastroenterology 2018,155,210), liver cancer (Dig.Dis.Sci.2016,61,474), lung cancer (Cell 2019,176,998) and myeloma (Nat.Commun.2018,9,4832) wait.
- IL17 induces cytokines and chemokines to promote myeloid suppressor cells, thereby further promoting angiogenesis (Nat.Med.2013, 19, 1114; PNAS 2014, 111, 5664), or enhancing the tumor microenvironment to form an anti-tumor inhibitory effect , to promote the formation and development of tumors (J Immunol.2010, 184, 2281; Nature, 2015, 522, 345; Adv Exp Med Biol. 2020, 1240, 47).
- J Immunol.2010, 184, 2281; Nature, 2015, 522, 345; Adv Exp Med Biol. 2020, 1240, 47 There is also research evidence that the activation of IL17 signaling helps to form tumor resistance to radiotherapy and chemotherapy, thereby reducing the therapeutic effect (J Exp Med.2020, 217, e20190297).
- anti-IL17 antibodies also have great application prospects for anti-tumor therapy.
- Psoriasis is an autoimmune disease with a high incidence rate.
- the prevalence rate in Europe and the United States is 1%-3%.
- the prevalence rate in China is relatively low, and it is estimated that there are more than 6 million patients.
- patients with moderate to severe psoriasis and/or psoriatic arthritis who are ineffective or poorly tolerated by traditional therapies, they can be treated by injecting biological agents; currently targeting IL17A is used to treat psoriasis.
- Sonelokimab a multivalent single-domain antibody targeting IL17A, IL17F and IL17A/F
- 120 mg or less dose of sonelokimab showed significant clinical benefits compared with placebo, with rapid onset of action , durability and acceptable safety (Lancet, 2021, 397, 1564).
- multivalent antibodies targeting IL17A and other cytokines, such as TNF- ⁇ , BAFF, and IL13 are also in different development stages for the treatment of various indications such as chronic inflammation of the lung.
- single domain antibody As a strict complete monomer, single domain antibody has the advantages of small molecular weight, high solubility, strong tissue permeability, high affinity and high degree of humanization. Its special structural characteristics and properties endow it with some other conventional antibodies or antibodies A property that fragments do not have (Annu Rev Biochem. 2013, 82, 775).
- V H H single-domain antibodies have good druggability to soluble targets, membrane protein targets including GPCRs and ion channels (Biomolecules 2021, 11, 63), and are widely used in anti-tumor drugs and anti-viruses , autoimmune diseases, and CAR-T immune cell therapy have been widely used (BioDrugs 2020, 34, 11; Antib Ther 2020, 3, 257; Biomolecules 2021, 11, 238).
- Single-domain antibodies are easy to modify and are ideal building blocks for multivalent antibodies. They can be aggregated through short linker sequences (linker) to construct multivalent forms with higher antigen affinity than monovalent antibodies, or they can be used to combine multiple different antibodies at the same time. antigenic target. Due to the small molecular weight of the multivalent VHH antibody, not only the traditional CHO expression production method can be used, but also the production system such as E.coli, P.pastoris, etc., and in the development of new dosage forms of biological drugs, including subcutaneous injection, inhalation And oral drugs, etc., which have advantages over traditional monoclonal antibodies (MAbs 2018, 10, 778; Mucosal Immunol. 2010, 3, 49).
- single/multivalent single-domain antibodies targeting IL17A and IL17F has multiple potential applications, such as multivalent antibodies targeting multiple targets such as IL17A, IL17A/F and IL17F at the same time, for psoriasis and ankylosing spondylitis, also including indications such as plaque psoriasis, psoriatic arthritis, and nonradiographic axial spondyloarthritis (Expert Opin Biol Ther 2019,19,45); or, with anti-TNF- One or more of cytokine antibodies such as ⁇ , BAFF and IL13 form a multivalent antibody, which is used for the treatment of various autoimmune diseases including chronic lung diseases, rheumatoid diseases, and Sjögren's syndrome.
- the technical problem to be overcome by the present invention is to provide an IL17 antibody and its preparation method and application in order to overcome the lack of effective single/multivalent single-domain antibodies targeting IL17A and IL17F in the prior art.
- the present invention solves the above-mentioned technical problems through the following technical solutions.
- the present invention provides an IL17 antibody comprising a heavy chain variable region, wherein:
- the heavy chain variable region comprises a sequence such as CDR1 shown in any one of SEQ ID NOs: 1 to 24, a sequence such as CDR2 shown in any one of SEQ ID NOs: 25 to 47, and a sequence such as SEQ ID NO : CDR3 shown in any one of 48-71.
- the heavy chain variable region of the present invention is a heavy chain variable region of the present invention.
- CDR1 comprises the sequence shown in SEQ ID NO:1
- CDR2 comprises the sequence shown in SEQ ID NO:25
- CDR3 comprises the sequence shown in SEQ ID NO:48;
- CDR1 comprises the sequence shown in SEQ ID NO:2
- CDR2 comprises the sequence shown in SEQ ID NO:26
- CDR3 comprises the sequence shown in SEQ ID NO:49;
- CDR1 comprises the sequence shown in SEQ ID NO:3
- CDR2 comprises the sequence shown in SEQ ID NO:27
- CDR3 comprises the sequence shown in SEQ ID NO:50;
- CDR1 comprises the sequence shown in SEQ ID NO:4
- CDR2 comprises the sequence shown in SEQ ID NO:28
- CDR3 comprises the sequence shown in SEQ ID NO:51;
- CDR1 comprises the sequence shown in SEQ ID NO:5
- CDR2 comprises the sequence shown in SEQ ID NO:29
- CDR3 comprises the sequence shown in SEQ ID NO:52;
- CDR1 comprises the sequence shown in SEQ ID NO:6
- CDR2 comprises the sequence shown in SEQ ID NO:28
- CDR3 comprises the sequence shown in SEQ ID NO:53;
- CDR1 comprises the sequence shown in SEQ ID NO:7
- CDR2 comprises the sequence shown in SEQ ID NO:30
- CDR3 comprises the sequence shown in SEQ ID NO:54;
- CDR1 comprises the sequence shown in SEQ ID NO:8
- CDR2 comprises the sequence shown in SEQ ID NO:31
- CDR3 comprises the sequence shown in SEQ ID NO:55;
- CDR1 comprises the sequence shown in SEQ ID NO:9
- CDR2 comprises the sequence shown in SEQ ID NO:32
- CDR3 comprises the sequence shown in SEQ ID NO:56; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:10
- CDR2 comprises the sequence shown in SEQ ID NO:33
- CDR3 comprises the sequence shown in SEQ ID NO:55;
- CDR1 comprises the sequence shown in SEQ ID NO:11
- CDR2 comprises the sequence shown in SEQ ID NO:34
- CDR3 comprises the sequence shown in SEQ ID NO:57;
- CDR1 comprises the sequence shown in SEQ ID NO:12
- CDR2 comprises the sequence shown in SEQ ID NO:35
- CDR3 comprises the sequence shown in SEQ ID NO:58; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:13
- CDR2 comprises the sequence shown in SEQ ID NO:36
- CDR3 comprises the sequence shown in SEQ ID NO:59; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:14
- CDR2 comprises the sequence shown in SEQ ID NO:34
- CDR3 comprises the sequence shown in SEQ ID NO:60; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:15
- CDR2 comprises the sequence shown in SEQ ID NO:37
- CDR3 comprises the sequence shown in SEQ ID NO:61;
- CDR1 comprises the sequence shown in SEQ ID NO:9
- CDR2 comprises the sequence shown in SEQ ID NO:38
- CDR3 comprises the sequence shown in SEQ ID NO:62;
- CDR1 comprises the sequence shown in SEQ ID NO:14
- CDR2 comprises the sequence shown in SEQ ID NO:34
- CDR3 comprises the sequence shown in SEQ ID NO:60;
- CDR1 comprises the sequence shown in SEQ ID NO:15
- CDR2 comprises the sequence shown in SEQ ID NO:37
- CDR3 comprises the sequence shown in SEQ ID NO:63;
- CDR1 comprises the sequence shown in SEQ ID NO:16
- CDR2 comprises the sequence shown in SEQ ID NO:39
- CDR3 comprises the sequence shown in SEQ ID NO:64;
- CDR1 comprises the sequence shown in SEQ ID NO:17
- CDR2 comprises the sequence shown in SEQ ID NO:40
- CDR3 comprises the sequence shown in SEQ ID NO:65; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:18
- CDR2 comprises the sequence shown in SEQ ID NO:41
- CDR3 comprises the sequence shown in SEQ ID NO:66; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:19
- CDR2 comprises the sequence shown in SEQ ID NO:42
- CDR3 comprises the sequence shown in SEQ ID NO:67; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:20
- CDR2 comprises the sequence shown in SEQ ID NO:43
- CDR3 comprises the sequence shown in SEQ ID NO:67; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:21
- CDR2 comprises the sequence shown in SEQ ID NO:44
- CDR3 comprises the sequence shown in SEQ ID NO:68; Or,
- CDR1 comprises the sequence shown in SEQ ID NO:22
- CDR2 comprises the sequence shown in SEQ ID NO:45
- CDR3 comprises the sequence shown in SEQ ID NO:69;
- CDR1 comprises the sequence shown in SEQ ID NO:23
- CDR2 comprises the sequence shown in SEQ ID NO:46
- CDR3 comprises the sequence shown in SEQ ID NO:70
- CDR1 comprises the sequence shown in SEQ ID NO:24
- CDR2 comprises the sequence shown in SEQ ID NO:47
- CDR3 comprises the sequence shown in SEQ ID NO:71.
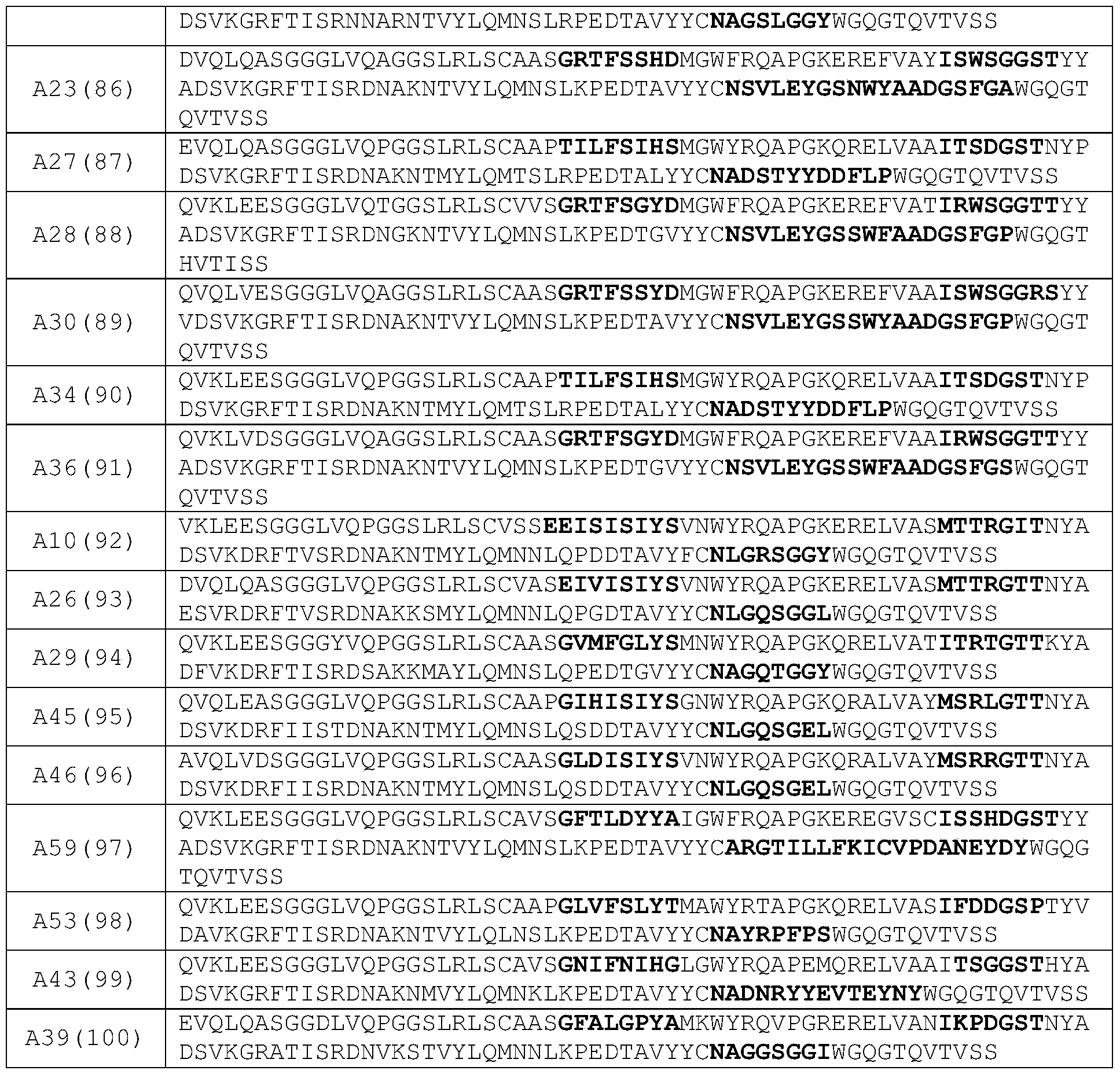
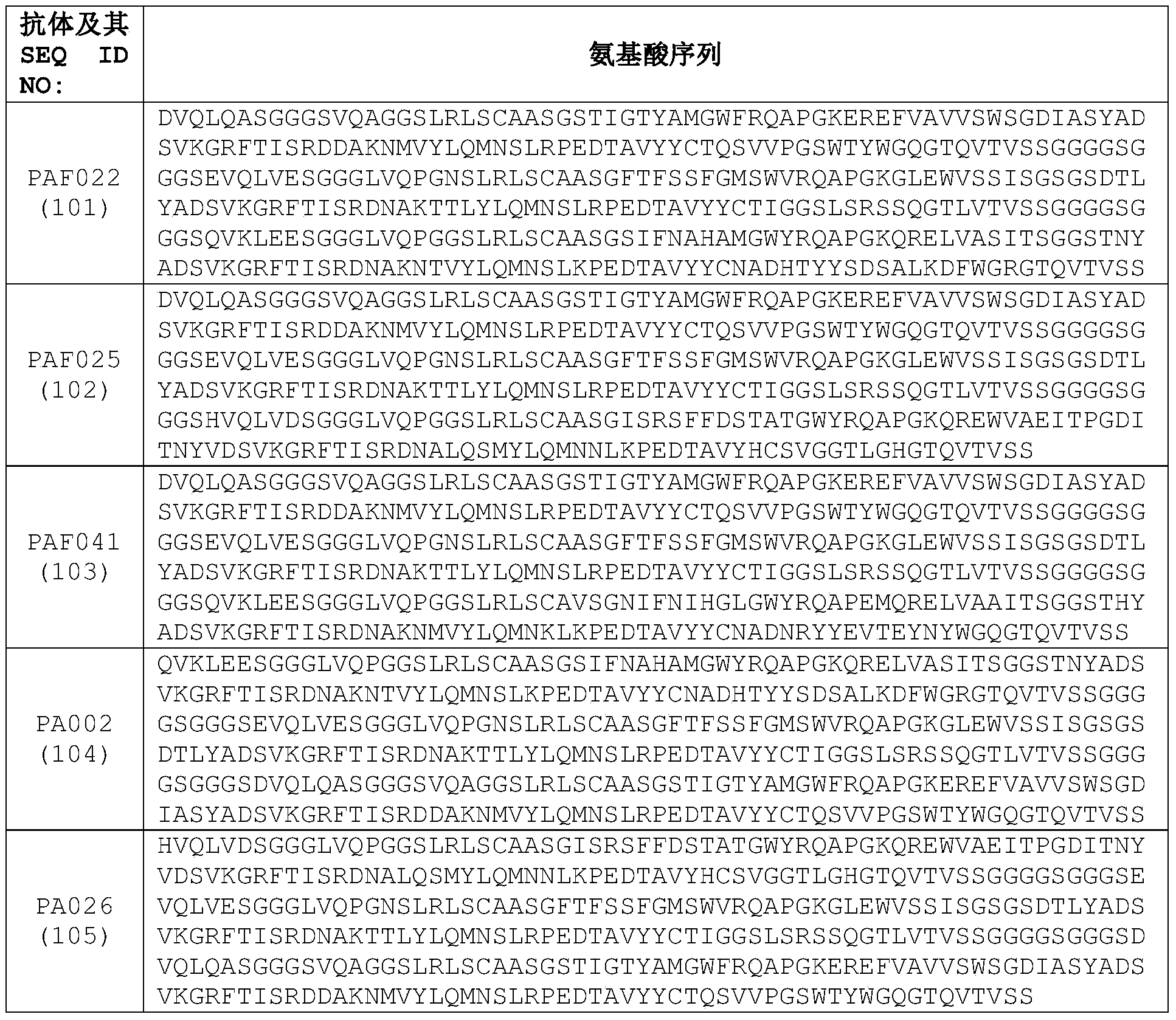
- the heavy chain variable region comprises a sequence as shown in any one of SEQ ID NO: 72-100.
- antibodies in the present invention include, but are not limited to, full-length antibodies, heavy chain antibodies (HCAb), antigen-binding fragments (Fab, Fab', F(ab) 2 , Fv fragments, F(ab') 2 , scFv, di-scFv and/or dAb), immunoconjugates, multispecific antibodies (e.g. bispecific antibodies), antibody fragments, antibody derivatives, antibody analogs or fusion proteins, etc., as long as they exhibit the desired antigen binding activity; preferably a single domain antibody or a VHH fragment thereof.
- the present invention also provides a trivalent single-domain antibody, which comprises the above-mentioned IL17 antibody.
- the trivalent single domain antibody preferably contains three VHHs connected sequentially from the N-terminus to the C - terminus: VHH1 - VHH2 - VHH3 .
- VHH2 is preferably an anti-HSA VHH ; more preferably contains the sequence shown in SEQ ID NO:108.
- VHH 1 and the above-mentioned VHH3 are preferably IL17 antibodies in the form of single-domain antibodies; preferably:
- the VHH1 contains the CDR combination as defined in any one of (1) to (6) above, and the VHH3 contains the CDR combination as defined in any one of the above (7) to (22); More preferably, the VHH1 contains the sequence shown in any one of SEQ ID NO:72-79 , and the VHH3 contains the sequence shown in any one of SEQ ID NO:80-100 ;
- the VHH3 contains the CDR combination defined in any one of (1) to (6) above, and the VHH1 contains the CDR combination defined in any one of the above (7) to (22) more preferably, the VHH3 contains the sequence shown in any one of SEQ ID NO:72 ⁇ 79, and the VHH1 contains the sequence shown in any one of SEQ ID NO:80 ⁇ 100 sequence.
- the VHH1 or VHH3 contains the CDR combination as defined in ( 2 ) above, and the VHH3 or VHH1 contains the above (7), ( 10) or the CDR combination defined in (26).
- the VHH1 contains the sequence shown in SEQ ID NO:73
- the VHH3 contains the sequence shown in SEQ ID NO:80, 83 or 99;
- the VHH1 contains the sequence shown in SEQ ID NO:80, 83 or 99
- the VHH3 contains the sequence shown in SEQ ID NO:73.
- the above different VHHs are preferably operably connected through a linker; the linker preferably contains the sequence shown in SEQ ID NO:107.
- the trivalent single domain antibody contains the amino acid sequence shown in any one of SEQ ID NO: 101-106.
- the present invention also provides an isolated nucleic acid encoding the IL17 antibody as described above or the trivalent single domain antibody as described above.
- the present invention also provides an expression vector comprising the isolated nucleic acid as described above.
- the present invention also provides a host cell comprising the expression vector as described above; preferably, the host cell is a prokaryotic cell or a eukaryotic cell.
- the present invention also provides a method for preparing an IL17 antibody or a trivalent single-domain antibody, which comprises culturing the above-mentioned host cells, and obtaining the antibody from the culture.
- the present invention also provides a pharmaceutical composition, which comprises the above-mentioned IL17 antibody or the above-mentioned trivalent single-domain antibody.
- the present invention also provides the application of the above-mentioned IL17 antibody, the above-mentioned trivalent single-domain antibody or the above-mentioned pharmaceutical composition in the preparation of drugs for preventing and treating IL17-related diseases or diseases.
- the disease or condition preferably includes chronic inflammation of the lung, rheumatoid arthritis, intestinal inflammation, dry eye disease, psoriasis, ankylosing spondylitis and various cancers including skin cancer, more preferably psoriasis, ankylosing spondylitis, chronic inflammation of the lungs and rheumatoid arthritis; psoriasis, ankylosing spondylitis and rheumatoid arthritis are further preferred.
- the present invention also provides a chimeric antigen receptor comprising the above-mentioned IL17 antibody or the above-mentioned trivalent single-domain antibody.
- the present invention also provides an antibody-drug conjugate, which comprises a cytotoxic agent, and the aforementioned IL17 antibody or the aforementioned trivalent single-domain antibody; preferably, the cytotoxic agent is MMAF or MMAE.
- the present invention also provides a kit comprising the above-mentioned IL17 antibody, the above-mentioned trivalent single domain antibody, the above-mentioned chimeric antigen receptor, the above-mentioned antibody drug conjugate and/or A pharmaceutical composition as described above;
- the kit further comprises (i) a device for administering the antibody or antigen-binding fragment thereof or antibody drug conjugate or pharmaceutical composition; and/or (ii) instructions for use.
- the present invention also provides a kit of medicines, which includes a medicine box A and a medicine box B, wherein:
- the kit A contains the above-mentioned IL17 antibody, the above-mentioned trivalent single domain antibody, the above-mentioned chimeric antigen receptor, the above-mentioned antibody drug conjugate and/or the above-mentioned pharmaceutical composition;
- the kit B contains other anti-tumor antibodies or pharmaceutical compositions containing the other anti-tumor antibodies, and/or consists of hormone preparations, targeted small molecule preparations, proteasome inhibitors, imaging agents, diagnostic agents, chemotherapeutic agents, One or more of the group consisting of oncolytic drugs, cytotoxic agents, cytokines, activators of co-stimulatory molecules, inhibitors of inhibitory molecules, and vaccines.
- the present invention also provides a method for diagnosing, treating and/or preventing IL17-mediated diseases or disorders, the method comprising administering to a patient in need a therapeutically effective amount of the above-mentioned IL17 antibody, the above-mentioned trivalent
- the present invention also provides a method for immune detection or determination of IL17, which includes using the above-mentioned IL17 antibody, the above-mentioned trivalent single-domain antibody, the above-mentioned chimeric antigen receptor, and the above-mentioned antibody drug
- the conjugate or the pharmaceutical composition as described above is detected after incubation with the analyte; preferably, the detection or the determination is for non-diagnostic and/or therapeutic purposes.
- the present invention also provides a combination therapy, which comprises separately administering the above-mentioned IL17 antibody, the above-mentioned trivalent single domain antibody, the above-mentioned chimeric antigen receptor, the above-mentioned antibody to patients in need
- a drug conjugate or a pharmaceutical composition as described above, and a second therapeutic agent preferably comprises other anti-tumor antibodies or a pharmaceutical composition comprising the other anti-tumor antibodies, and/or consists of Hormonal agents, targeted small molecule agents, proteasome inhibitors, imaging agents, diagnostic agents, chemotherapeutic agents, oncolytic agents, cytotoxic agents, cytokines, activators of co-stimulatory molecules, inhibitors of inhibitory molecules, and vaccine components One or more of the groups.
- the present invention uses IL17A homodimer, IL17F homodimer and IL17A/F heterodimer to immunize alpaca multiple times, isolates mRNA of peripheral blood lymphocytes, synthesizes cDNA and constructs V H H Antibody library, using phage display technology, ELISA technology and other methods to screen and obtain multiple V H H antibodies with high affinity and specific binding to IL17A, IL17F and IL17A/F, followed by V H H antibody expression purification, affinity, specificity and stability By combining with Anti-HSA antibody, construct multiple multivalent V H H antibodies, and complete the evaluation of expression purification, affinity, specificity, stability and epitope binning. The results show that these monovalent and multivalent VHH antibodies have unique advantages, mainly in the following aspects:
- V H H antibodies can selectively and high-affinity bind to one or more of human cytokines IL17A, IL17F and IL17A/F, and can bind cynomolgus monkey IL17A and IL17F with high affinity and specificity;
- V H H antibody sequence has good thermal stability (T m ⁇ 65°C);
- the multivalent antibody constructed by combining different V H H antibody sequences can simultaneously bind IL17A, IL17A/F and IL17F with high affinity and specificity;
- Monovalent and multivalent antibodies can be efficiently expressed and folded correctly in the intercellular space of E.coli, and can be purified and prepared by protein A-labeled affinity chromatography and ion exchange chromatography;
- V H H antibodies are expected to be used in diseases caused by abnormal regulation of cytokines such as IL17A, IL17F and IL17A/F;
- V H H antibodies can be easily combined with V H H single domain antibodies of other targets to form multivalent antibodies, which can be used for the treatment of anti-tumor, lung disease, autoimmune disease, anti-virus, anti-infection, etc.
- Fig. 1A is the SDS-PAGE electrophoresis diagram of V H H-A19 affinity chromatography
- 1B is the Superdex 75 10/300 chromatogram
- Fig. 1C is the protein SDS-PAGE electrophoresis diagram after Superdex 75 10/300 chromatography.
- Fig. 2 is a diagram of the affinity test between V H H-A19 and human IL17A.
- Figure 3 is the test chart of T m value of V H H-AF6.
- Fig. 4 is a Paratope analysis diagram of the interaction between V H H-A8 and IL17A.
- 5A is the purification diagram of rProtein A sepharose FF of PAF022
- 5B is the purification diagram of Hitrap Q column
- 5C is the SDS-PAGE electrophoresis diagram of sampling after Hitrap Q column purification.
- Figure 6 is the T m test chart of PAF022.
- Fig. 7A shows the neutralizing activity of PAF022, PAF025 and PAF041 on IL17A;
- Fig. 7B shows the neutralizing activity of PAF022, PAF025 and PAF041 on IL17A/F.
- FIG 8 shows the way V H H is connected to anti-HSA through amino acid Linker (GGGGSGGGS).
- Example 1 Alpaca immunization, titer detection and lymphocyte separation
- IL17A (Sino Biological, 12047-HNAS), IL17A/F (Sino Biological, CT047-HNAE) and IL7F (Sino Biological, 11855-HNAE) three proteins, refer to (Methods Mol Biol 2012, 911, 211; Curr Protoc Immunol 2013, 103, 2.17.1; Nat Protoc 2014, 9, 674) and other documents described in the immunization method, immunized three alpacas.
- the amount of 100 ⁇ g protein/alpaca was injected on both sides of the neck muscle of the alpaca, and a total of two injections were injected.
- Complete Freund's adjuvant was used for the first time and recorded as day 0.
- incomplete Freund's adjuvant was used for the second to sixth booster immunization.
- the immune effect of alpaca was tested by ELISA method (J Vis Exp 2019, 143). The results showed that the titer of the serum diluted 10,000 times after five immunizations was significantly higher than that of the negative serum diluted 1,000 times, reaching the level set before immunization. Determined potency requirements.
- the lymphocyte RNA obtained in Example 1 was synthesized into cDNA according to the instructions of the PrimeScript TM II 1st Strand cDNA Synthesis Kit (TAKARA, 6210A), and amplified by nested PCR using KOD-FX (TOYOBO, KFX-101)
- the single domain antibody (V H H) gene was obtained and double digested with BamHI-HF (NEB, #R3136L) and XhoI (NEB, #R0146L) overnight at 37°C.
- BamHI-HF (NEB, #R3136L) and XhoI (NEB, #R0146L) were used to digest the pComb3XSS-GFP vector (transformed from pComb3XSS (BioVector NTCC Inc.), and the sequence between the enzyme cutting site SfiI was replaced with the GFP gene, At the same time, BamHI and XhoI were added at both ends, so that the connection sequence was 5'-OmpA signal peptide-BamHI-GFP-XhoI-gene III-stop codon-3').
- the double-digested V H H fragment and the pComb3xss-GFP vector were incubated with T4DNA Ligase (NEB, #M0202L) at 16°C and ligated overnight. After further ion purification of the ligated product, TG1 competent cells (Lucigen, 60502- 2), collect the Escherichia coli cultured overnight to complete the library construction. In the experiment, 6.8-9.2 ⁇ 10 8 cDNA libraries with a fragment insertion rate of more than 95% and good sequence diversity were obtained for three alpacas.
- the cDNA library is screened by phage display technology and liquid phase method. Taking the screening of antigen IL17A as an example, the specific steps are as follows:
- Bacterial culture solution was centrifuged at 3000g at 4°C for 10 minutes, the supernatant was mixed with PEG/NaCl solution at a volume ratio of 4:1, then cooled on ice for 1 hour, centrifuged at 3000g at 4°C for 20 minutes, and the precipitate was collected. Resuspend in 10mL PBS, centrifuge at 3000g at 4°C for 20 minutes, collect the supernatant and add 2.5mL PEG/NaCl solution, mix well and keep on ice for 30 minutes; centrifuge at 3000g at 4°C for 30 minutes, resuspend the pellet with 1mL PBS to obtain a phage solution .
- the V H H gene obtained in the third round of screening was cloned into the expression vector pSJF2, and the positive clone was obtained by ELISA detection method, and the V H H sequence of the positive clone was obtained by sequencing analysis.
- the experimental method is as follows:
- BbsI-HF (NEB, #R3539L) and BamHI-HF (NEB, #R3136L) restriction sites at both ends were designed to obtain V After digestion of H H gene, BbsI-HF and BamHI-HF, use T4 DNA ligase to connect the same digested pSJF2 vector, and transform TG1 competent cells, spread LB plate containing 100 ⁇ g/mL ampicillin, and culture at 37°C overnight.
- the antigen IL17A (Sino Biological, 12047-HNAS) was diluted to 1 ⁇ g/mL with carbonate buffer, 100 ⁇ L/well, added to an ELISA plate (ThermoFisher scientific, 439454), and incubated overnight at 4°C.
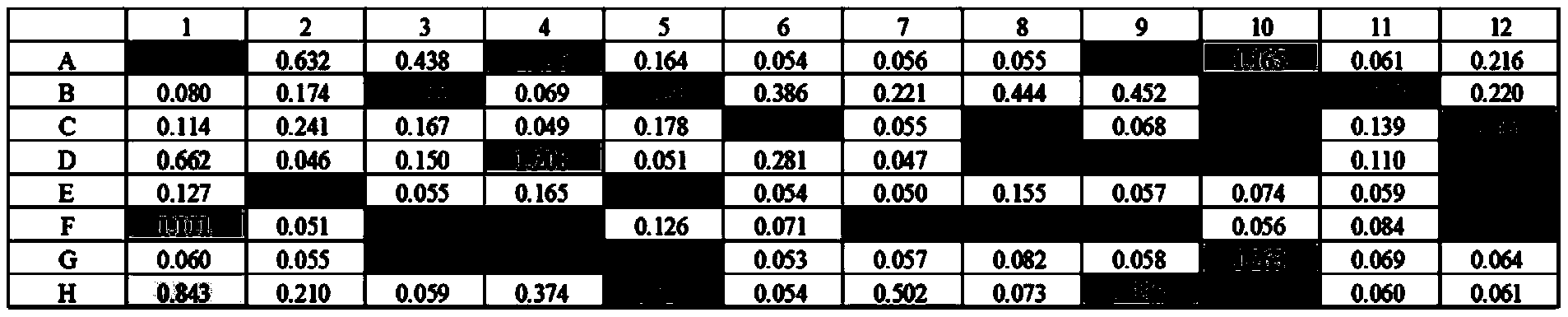
- the results of the ELISA test are shown in Table 1: consistent with the 96-well deep-well plate, the first column A to H is the column number, the first row 1-12 is the horizontal row number, and the clones are numbered and identified by using the horizontal and vertical numbers as coordinates.
- the clones of antigens IL17A/F and IL17F were detected using the same method.
- the gene was resynthesized and cloned into the pET28a(+) vector to construct an expression plasmid. From the N-terminal to the C-terminal of the expression sequence, there are OmpA signal peptide, V H H, linker (amino acid sequence is GAA) and 6 ⁇ His tag.
- the synthesized expression plasmid was transformed into BL21(DE3) competent cells, and the plate was cultured overnight at 37°C.
- Inoculate 400 mL of 2 ⁇ YT medium containing 50 ⁇ g/mL kanamycin at a ratio of 1:100 to 400 mL of 2 ⁇ YT medium containing 50 ⁇ g/mL kanamycin after monoclonal cultivation overnight, and cultivate to OD 600 0.8 at 250 rpm at 37°C, add IPTG with a final concentration of 0.5mM, and cultivate at 16°C 16 hours.
- Collect the bacteria by low-speed centrifugation, add 40mL of Buffer A (10mM/Tris-HCl, pH 8.0, 1mM/EDTA, 25% sucrose) to thoroughly resuspend the bacteria, shake and incubate at room temperature for 15 minutes; remove the supernatant after high-speed centrifugation, and pre-cool 20mL Buffer B (30mM Tris-HCl, pH 8.0, 5mM MgSO 4 ) was resuspended; the supernatant was taken after high-speed centrifugation again, and an appropriate amount of NaCl solution, imidazole solution and DTT solution were added to make the final concentrations respectively 150mM, 20mM and 1mM.
- Buffer A 10mM/Tris-HCl, pH 8.0, 1mM/EDTA, 25% sucrose
- a in Fig. 1 is the SDS-PAGE electrophoresis image of the sample during A19 affinity chromatography.
- S represents the supernatant after lysing bacteria by "superosmotic method"
- FT represents the combined flow-through
- 40 and 80 represent the buffer after washing Ni-Resin with 1 ⁇ PBS buffer containing 40mM and 80mM imidazole
- E represents Samples eluted with 1 ⁇ PBS buffer containing 300mM imidazole
- B represents the electrophoresis results of Ni-Resin boiled samples after elution.
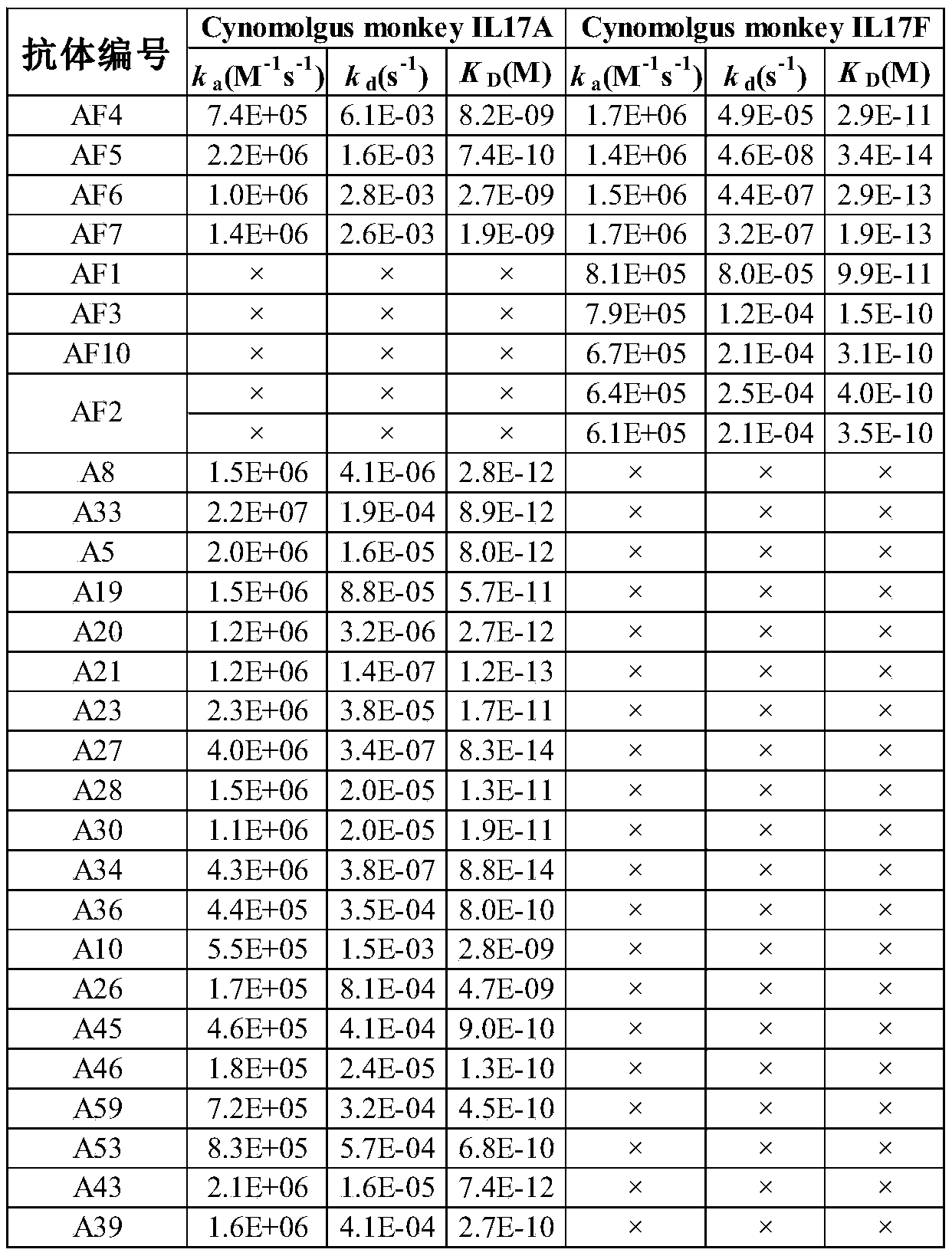
- the affinity test of the monovalent single-domain antibody was completed using Biacore T200, and the His-tag monoclonal antibody was bound to the CM5 chip (GE Healthcare, BR- 1005-30), used to capture monovalent V H H antibody with His tag, human IL17A, IL17A/F and IL17F were used as mobile phase for affinity detection.
- Figure 2 is the binding and dissociation curves and fitting diagrams of antibody A19 and IL17A, and the affinity value between V H H antibody and antigen was obtained through calculation and analysis.
- the affinity data are shown in Table 3, where " ⁇ " means no binding.
- the thermal stability of candidate VHH antibody molecules was initially screened by differential scanning fluorimetry (DSF). During the screening process, using SYPRO Orange Protein Gel Stain (Sigma, S5692) and gradually heating with a qPCR instrument, a preliminary test of the Tm value of the purified VHH antibody was performed. In the initial screening, the T m value of VHH antibody is ⁇ 55°C as the screening condition, and the VHH antibodies listed in Table 2 all meet this requirement.
- Fig. 3 is a test chart of antibody AF6 using DSF to test the T m value, and its T m value reaches 74°C.
- the ability of V H H antibody to bind one or more antigens in IL17A, IL17A/F or/and IL17F with high affinity in order to further evaluate the ability of specific binding of IL17 family proteins, follow the steps 2)
- the affinity between the antibody and human IL17B Prospec, CYT-753
- IL17C Acro Biosystems, ILC-H52H7
- IL17D Atagenix, ATAP01505
- IL17E Acro Biosystems, IL5-H4221
- the 29 candidate VHH antibody molecules can all bind IL17A/F with strong affinity, and IL17A/F is used as a positive control molecule to test the candidate VHH antibody and IL17B, IL17C, IL17D and IL17E specific binding capacity.
- the test results showed that the binding Ru values of 29 candidate V H H antibody molecules to IL17A/F ranged from 30 to 120, while most of the binding Ru values to IL17B, IL17C, IL17D and IL17E were ⁇ 10, far from The absolute value of Ru is lower than that of IL17A/F. Therefore, these antibodies have no obvious binding to IL17B, IL17C, IL17D and IL17E, and the specificity meets the requirements.
- V H H was coupled to a CM5 chip through a His tag as immobilization, and IL17A and IL17F molecules of cynomolgus monkey were used as mobile phase for detection.
- the detection results are shown in Table 3, where ⁇ represents no binding.
- the present invention uses SPR technology to perform cluster analysis on the above V H H according to the difference of Epitope, and the method is as follows. Optimize and synthesize the expressed gene of E.Coli codon preference, so that the sequence of the expression frame is OmpA signal peptide, V H H, SASA (from US 2013/0129727 A1, which can be combined with BSA), and His tag; the synthesized gene follows 1) Expression and purification of SASA-V H H was performed using the monovalent single domain antibody purification method described in .
- BSA is first coupled to the CM5 chip, then SASA-V H H (V H H 1 ) is combined with BSA through the flow path to be immobilized on the chip, and then IL17A/F is combined with V H H 1 through the flow path to form BSA-SASA-V H H (V H H 1 )-IL17A/F complex, and finally the V H H (V H H 2 ) antibody passed through the flow path as the mobile phase, and the response value Ru was recorded at this time.
- V H H 1 and V H H 2 have different epitopes, then V H H 2 can be compared with BSA-SASA-V H H ( V H H 1 )-IL17A/F complex binding, the Ru value will be higher; otherwise, it will be low.
- Table 4 shows the results obtained by using this method. It can be seen that some antibodies have different epitopes, such as combinations of AF5 and A8, AF10 and A5, etc., where Ru is positive and has a large value; some antibodies have the same epitope, Such as the combination of A8 and A19, AF2 and A21 with Ru around 0.
- Hydrogen-deuterium exchange mass spectrometry (HDX-MS) epitope mapping is an effective method that can quickly provide complete information on epitope structure.
- the principle is that the hydrogen on the protein amide bond can exchange controllably with the deuterium in heavy water.
- the hydrogen on the surface of the protein is in close contact with the heavy water, and the exchange rate is fast; while the hydrogen inside the protein and the hydrogen that forms the hydrogen bond are difficult to contact. Heavy water, slow exchange rate.
- mass spectrometry uses mass spectrometry to measure the number of deuterated peptide sequences under different reaction times, and then calculate the hydrogen-deuterium exchange rate of each peptide sequence, the spatial structure information of the protein can be judged.
- the technique uses mass spectrometry to measure the solvent accessibility of amino acid residues in proteins to determine the interaction sites of antigen-antibody complexes in their native solution without introducing any modification to the antigen or antibody.
- the experimental steps are as follows: Dissolve 5-10 ⁇ M antigen, antibody, and antigen-antibody complex in 50mM HEPES, pH 7.4, 150mM NaCl, 4mM TCEP solution at a molar ratio of 1:1, and place them at 4°C for 1 hour to ensure Form a stable antigen-antibody complex. After standing for 1 hour, at 4°C, dilute 5 ⁇ L of different samples into 20 ⁇ L D2O (deuterium) respectively, and place them at different HDX time points (for example, 0, 10, 60, 300, 900 seconds), by cooling with 25 ⁇ L 100mM NaH 2 PO 4 mixed with 1M TCEP to stop the reaction.
- the sample tube Place the sample tube on dry ice immediately after stopping the reaction until the sample is injected into the HDX LEAP PAL3.0 platform.
- the sample passed through the immobilized pepsin column at a flow rate of 120 ⁇ L/min, and the enzymatically digested peptides were captured and desalted by a 2.1 mm ⁇ 5 cm C18 column (1.9 ⁇ m Hypersil Gold, Thermo Fisher).
- the desalted peptides were separated with a linear gradient of 4-40% acetonitrile and 0.3% formic acid over 8 minutes.
- protein digestion and peptide separation were performed at 4°C.
- the eluted and separated peptides were analyzed using an Orbitrap mass spectrometer (Orbitrap Fusion TM Tribrid TM Mass Spectrometer, Thermo Fisher), with a measurement resolution of 65,000 (m/z 400), and hydrogen-deuterium exchanged mass spectrometry data were obtained.
- Orbitrap mass spectrometer Orbitrap Fusion TM Tribrid TM Mass Spectrometer, Thermo Fisher
- HDX assays triplicates
- the average m/z centroid value of the mass spectrum peak intensity of each enzymatic peptide was calculated by HDX Workbench software (generally, the accuracy is 10ppm), and then converted into the percentage of deuterium incorporation.
- the key amino acid sequences involved in the spatial epitope are calculated, and the difference in Delta%D (comparing the change in the percentage of deuterium incorporation on the same peptide) is determined by calculating the difference between the two samples. Differences in Delta %D outside -5 to 5% were considered significant. In addition, HDX Workbench detects statistically significant (p ⁇ 0.05) differences between samples at each time point by student's t test.
- the present invention uses the above method to identify the binding site of Epitope/Paratope.
- a total of five interactions AF5-IL17A, AF5-IL17A/F, AF5-IL17F, A8-IL17A and A8-IL17A/F, were carried out.
- Figure 4 is the Paratope analysis of the interaction between A8 and IL17A. The results show that SGSIFNAHAMGW and CNADHTYYSDSAL are potential Paratopes, mainly focusing on CDR1 and CDR3.
- AF5 has strong binding affinity with IL17A, IL17A/F and IL17F, but the binding mode of AF5 is not exactly the same according to the Paratope involved in the binding.
- the collated detection results are shown in Table 5.
- A8 mainly interacts with IL17A and IL17A/F through CDR1 and CDR3.
- AF5 mainly binds to IL17A through partial sequences of CDR2 and CDR3; interacts with IL17A/F through partial amino acids of CDR3 and Framework-4; binds to IL17F through CDR2.
- the present invention After characterization and analysis of monovalent single-domain antibodies, the present invention has obtained 29 high-affinity VHH sequences with good specificity and good thermal stability, and these antibodies can be used to combine to construct multivalent antibodies to achieve simultaneous high-affinity binding to IL17A , IL17A/F and IL17F purposes.
- Table 1 in view of the higher affinity between A series V H H and IL17A, and the higher affinity between AF series V H H and IL17A/F and IL17F, when combining AF series, A series One for each VHH , combined by Anti-HSA ( VHH , from US 2007/0269422 A1, ALB11).
- V H H is connected to anti-HSA through amino acid Linker (GGGGSGGGS) (as shown in Figure 8) to construct different trivalent antibodies.
- the expression method of the trivalent single-domain antibody is the same as that of the monovalent single-domain antibody. After two steps of purification, a high-purity trivalent single-domain antibody can be obtained.
- the purification method is as follows: After expressing the trivalent single-domain antibody, add an appropriate amount of pre-cooled Lysis buffer (1 ⁇ PBS, 1mM DTT, 1mM EDTA), ultrasonicate (400W, 3s on/3s off, 15min) and then centrifuge at a high speed , load the supernatant to the pretreated HiTrap TM MabSelect SuRe Column (Cytiva, 11003493), equilibrate with 1 ⁇ PBS until the A280 baseline goes flat to zero, wash with 100mM glycine-hydrochloric acid (PH3.0) at a flow rate of 1mL/min Remove and collect the target peak; after adjusting the pH of the eluted protein to 10.0, pass through the Hitrap Q column (Cytiva, 17115401)
- a in Figure 5 is the protein A affinity chromatography purification diagram of the antibody PAF022
- B in Figure 5 is the purification diagram of the Hitrap Q column
- C in Figure 5 is the SDS-PAGE electrophoresis diagram of the Hitrap Q column after purification.
- the Anti-His in the His tag capture kit was first coupled to the CM5 chip for Capture human IL17A (Acro Biosystems, ILA-H82Q1), IL17A/F (Acro Biosystems, ILF-H52W6) and IL17F (Sino Biological, 11855-H07H) with His-tagged, trivalent single domain antibody as mobile phase for testing .
- the test results are shown in Table 9.
- the present invention uses a differential scanning calorimeter (TA Instruments, Discovery DSC2500/250/25) to test the prepared control antibody MS3091 (US 2014/0314743 A1), A total of seven trivalent antibodies, PAF022, PAF025, PAF041, PA002, PA026 and PA154, were tested for thermal stability.
- the specific method is to take 20 ⁇ L of the sample to be tested (2mg/mL) and add it to the sample plate, seal it, put it into the instrument, heat it from 40°C to 95°C at a speed of 1°C/min, and record the heat flow between the sample plate and the reference plate Variety.
- the T m of PA022 is 76.7°C
- Figure 6 shows the heat flow curve of PA022.
- the T m value of the positive control is 82.0°C
- PAF025 and PAF041 are 79.4°C and 78.6°C, respectively
- PA002 is 76.0°C, all of which are greater than 65°C, showing good thermal stability.
- IL17 can stimulate epidermal cells (Hs27 cell line, human epidermal fibroblasts, ATCC#CRL-1634) to secrete GRO ⁇ . If IL17 antibody is added, it will block the interaction between IL17 and cell membrane surface receptors by binding to IL17 protein, thereby achieving Inhibiting the effect of epidermal cells secreting GRO ⁇ , by detecting the amount of GRO ⁇ secretion, the IC50 value of the candidate antibody can be obtained.
- epidermal cells Hs27 cell line, human epidermal fibroblasts, ATCC#CRL-1634
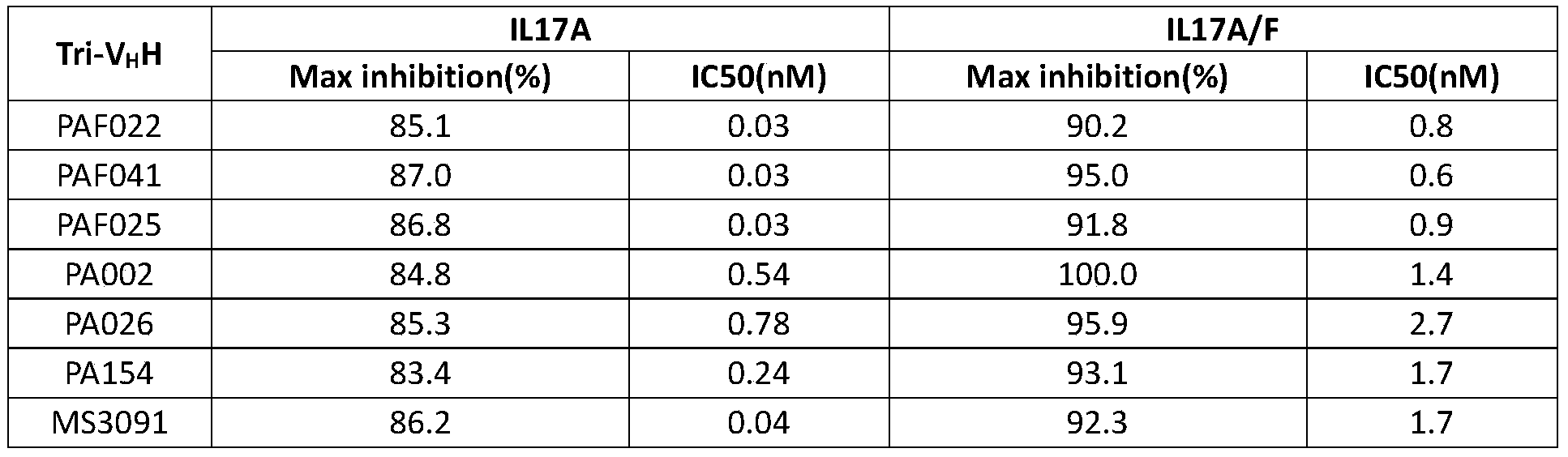
- the present invention has tested the activity of trivalent antibodies, and some results are shown in Figure 7 and Table 10.
- the maximum inhibitory rates of the six tested trivalent antibodies to IL17A were all greater than 83%; the IC50 of antibodies PAF022, PAF025 and PAF041 were all In the vicinity of 0.03nM, compared with the IC50 value of MS3091 of 0.04nM, the activity was comparable, and the IC50 of antibodies PA002, PA026 and PA154 were 10-20 times lower than MS3091.
- the six trivalent antibodies tested had a maximum inhibitory rate of more than 90% on IL17A/F, and their IC50 were all between 0.6-2.7nM, which was only 3-5 times different from MS3091.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Immunology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biochemistry (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Microbiology (AREA)
- Oncology (AREA)
- Communicable Diseases (AREA)
- Rheumatology (AREA)
- Pain & Pain Management (AREA)
- Epidemiology (AREA)
- Mycology (AREA)
- Peptides Or Proteins (AREA)
Abstract
Description
本发明属于生物医药领域,具体涉及一种IL17抗体及其制备方法和应用。The invention belongs to the field of biomedicine, and in particular relates to an IL17 antibody and its preparation method and application.
细胞因子IL17可以来自多种不同的免疫细胞,其中以Th17辅助T细胞分泌为主要来源,CD8 +T细胞、NK T细胞、LLC3(Type 3 innate lymphoid cells)和γδT细胞等也可以分泌IL17(Nat Rev Immunol.2010,10,479)。细胞因子IL17蛋白家族主要包括IL17A、IL17B、IL17C、IL17D、IL17E(IL25)和IL17F,其中IL17A和IL17F序列相似度大,可以在Th17细胞中同时表达。IL17A和IL17F可以是同二聚体的存在形式,也可以由IL17A和IL17F单体组成的IL17A/F异二聚体(Cold Spring Harb Perspect Biol.2018,10,a028522)。IL17受体蛋白存在多个亚基,包括IL17RA、IL17RB、IL17RC、IL17RD和IL17RE等五个受体亚单位,其中IL17RA经常作为IL17受体的通用核心亚单位,参与不同IL17分子的结合,比如IL17A、IL17F和IL17A/F都与IL17RA/IL17RC组成的受体复合物结合、IL17C与IL17RA/IL17RE组成的受体复合物结合、IL17E与IL17RA/IL17RB组成的受体复合物结合(Cytokine 2013,64,477;Trends Immunol.2017,38,310)。 The cytokine IL17 can come from a variety of different immune cells, among which the secretion of Th17 helper T cells is the main source, and CD8 + T cells, NK T cells, LLC3 (Type 3 innate lymphoid cells) and γδ T cells can also secrete IL17 (Nat Rev Immunol. 2010, 10, 479). The cytokine IL17 protein family mainly includes IL17A, IL17B, IL17C, IL17D, IL17E (IL25) and IL17F, among which IL17A and IL17F have a large sequence similarity and can be expressed simultaneously in Th17 cells. IL17A and IL17F can exist as a homodimer, or as an IL17A/F heterodimer composed of IL17A and IL17F monomers (Cold Spring Harb Perspect Biol. 2018, 10, a028522). There are multiple subunits of IL17 receptor protein, including five receptor subunits including IL17RA, IL17RB, IL17RC, IL17RD and IL17RE, among which IL17RA is often used as the common core subunit of IL17 receptor and participates in the binding of different IL17 molecules, such as IL17A , IL17F and IL17A/F both bind to the receptor complex composed of IL17RA/IL17RC, IL17C binds to the receptor complex composed of IL17RA/IL17RE, IL17E binds to the receptor complex composed of IL17RA/IL17RB (Cytokine 2013,64,477; Trends Immunol. 2017, 38, 310).
IL17RA在造血干细胞和非造血细胞中都表达,但IL17RC常见非造血干细胞中表达,因此IL17A、IL17F和IL17A/F等二聚体通过结合IL17RA/IL17RC受体复合物,激活非造血干细胞,比如上皮细胞和基质细胞等。细胞被激活之后,产生一系列促炎症因子,比如IL-1、IL-6、G-CSF和TNF等细胞因子,也诱导表达CXCL1、CXCL2、CXCL5、CCL2、CCL7、CCL20和IL-8等趋化因子,也会促进基质金素蛋白酶MMP1、MMP3、MMP9和MMP13的表达,以及产生一些抗菌肽,比如β-defensins、S-100蛋白等(Immunity 2011,34,149)。细胞因子IL17在抵御外部细菌、真菌感染、保持粘膜和维持表皮的完整性、促进伤口愈合方面,扮演重要的角色(Immunol Rev.2008,226,57)。反过来说,一旦IL17的活性不受正常的调控,会引发慢性炎症和自身免疫疾病,甚至可能诱发形成肿瘤。IL17RA is expressed in both hematopoietic stem cells and non-hematopoietic cells, but IL17RC is commonly expressed in non-hematopoietic stem cells, so dimers such as IL17A, IL17F and IL17A/F activate non-hematopoietic stem cells, such as epithelial cells, by binding to the IL17RA/IL17RC receptor complex cells and stromal cells, etc. After the cells are activated, they produce a series of pro-inflammatory factors, such as cytokines such as IL-1, IL-6, G-CSF and TNF, and also induce the expression of CXCL1, CXCL2, CXCL5, CCL2, CCL7, CCL20 and IL-8. It also promotes the expression of matrix metalloproteinases MMP1, MMP3, MMP9 and MMP13, and produces some antimicrobial peptides, such as β-defensins, S-100 protein, etc. (Immunity 2011, 34, 149). The cytokine IL17 plays an important role in resisting external bacteria and fungal infections, maintaining the integrity of the mucosa and epidermis, and promoting wound healing (Immunol Rev. 2008, 226, 57). Conversely, once the activity of IL17 is not normally regulated, it will cause chronic inflammation and autoimmune diseases, and may even induce the formation of tumors.
IL17A及相关细胞因子的表达水平上调,可引发一系列的自身免疫疾病,包括银屑病(psoriasis)、类风湿性关节炎(rheumatoid arthritis)、硬皮症(scleroderma)和狼疮(lupus)等。在银屑病形成的过程中,能分泌TNF-α/iNOS的DC细胞(TIP-DC)扮演了重要的角色,TIP-DC细胞通过结合TNF-α而被活化,活化后的TIP-DC细胞进一步分泌TNF-α和IL23、IL12等细胞因子,IL23进而活化Th17细胞,促使Th17细胞分泌过量的IL17A、IL17F 和IL22等细胞因子,这些细胞因子进一步驱使角质细胞异常分化和增殖,从而形成银屑病(J Am Acad Dermatol.2014,71,141;J Dermatol.2018,45,264;Int J Mol Sci.2020,21,1690)。抑制IL23-IL17免疫轴(Nat.Rev.Immunol.2014,14,585)已在银屑病等相关疾病的治疗中取得了良好疗效。除此之外,IL17A、IL17F血清浓度上升与婴儿的特应性皮炎(atopic dermatitis)的严重性存在正相关。近来也有相关研究显示,IL17A也是败血症(Sepsis)的潜在治疗靶点(Front Immunol.2020,11,1558)。在神经性疾病领域,除了多发性硬化(multiple sclerosis)外,IL17调控异常也被认为是慢性神经炎、实验性自身免疫性脑脊髓炎、阿尔茨海默氏病和缺血性脑损伤的致病因素(Front Immunol.2020,11,947)。The increased expression of IL17A and related cytokines can cause a series of autoimmune diseases, including psoriasis, rheumatoid arthritis, scleroderma and lupus. In the process of psoriasis formation, DC cells that can secrete TNF-α/iNOS (TIP-DC) play an important role. TIP-DC cells are activated by binding TNF-α, and the activated TIP-DC cells Further secrete TNF-α, IL23, IL12 and other cytokines, IL23 activates Th17 cells, prompts Th17 cells to secrete excessive cytokines such as IL17A, IL17F and IL22, and these cytokines further drive abnormal differentiation and proliferation of keratinocytes, thus forming psoriasis disease (J Am Acad Dermatol.2014,71,141; J Dermatol.2018,45,264; Int J Mol Sci.2020,21,1690). Inhibition of the IL23-IL17 immune axis (Nat. Rev. Immunol. 2014, 14, 585) has achieved good results in the treatment of psoriasis and other related diseases. In addition, elevated serum concentrations of IL17A and IL17F were positively correlated with the severity of atopic dermatitis in infants. Recent studies have also shown that IL17A is also a potential therapeutic target for sepsis (Front Immunol. 2020, 11, 1558). In the field of neurological diseases, in addition to multiple sclerosis (multiple sclerosis), dysregulation of IL17 is also considered to be the cause of chronic neuritis, experimental autoimmune encephalomyelitis, Alzheimer's disease and ischemic brain injury. Disease factors (Front Immunol.2020, 11, 947).
Th17细胞亚群参与调控肺部中性粒细胞、巨噬细胞的炎症,与重度中性粒细胞性哮喘和慢性阻塞性肺疾病有关联(Nat.Rev.Immunol.2008,8,183;Ann.Rev.Physiol.2010,72,495)。有研究显示IL17A、IL17F的表达水平与哮喘的严重程度相关,其相关性由于IL17能有效促进中性粒生长因子、化学趋化因子的表达,比如IL-6、G-CSF、IL8等,从而促进中性粒细胞的富集(Expert Rev.Respir.Med.2014,8,25)。在患有慢性阻塞性肺疾病、支气管哮喘和同时患有慢性肺阻塞肺疾病和哮喘的患者中,血清中IL17A浓度上升,并且浓度提高程度与疾病严重程度正相关(Mediators Inflamm.2020,2020,4652898)。通过研究慢性阻塞性肺疾病亚型人群的生物标志物,发现一类对激素治疗反应不敏感的人群,与IL17引发的炎症相关,意味着结合IL17的抗体,对这类慢性阻塞性肺疾病亚型人群的治疗具有临床价值(J Clin Invest.2019,129,169)。Th17 cell subsets are involved in regulating the inflammation of neutrophils and macrophages in the lungs, and are associated with severe neutrophil asthma and chronic obstructive pulmonary disease (Nat.Rev.Immunol.2008,8,183; Ann.Rev. Physiol. 2010, 72, 495). Studies have shown that the expression levels of IL17A and IL17F are related to the severity of asthma. The correlation is because IL17 can effectively promote the expression of neutrophil growth factors and chemokines, such as IL-6, G-CSF, IL8, etc., thus Promote the enrichment of neutrophils (Expert Rev. Respir. Med. 2014,8,25). In patients with chronic obstructive pulmonary disease, bronchial asthma, and patients with both chronic obstructive pulmonary disease and asthma, the concentration of IL17A in serum is increased, and the degree of increased concentration is positively correlated with the severity of the disease (Mediators Inflamm.2020,2020, 4652898). By studying the biomarkers of subtypes of chronic obstructive pulmonary disease, it was found that a group of people who are insensitive to hormone therapy is associated with inflammation triggered by IL17, which means that antibodies that bind to IL17 are effective for this subtype of chronic obstructive pulmonary disease. The treatment of
除了自身免疫性疾病之外,越来越多的证据显示IL17失调,对癌症的早期发生、中晚期的发展,具有促进作用。通过测试一系列不同实体瘤患者血清中IL17的浓度,发现拥有更高IL17浓度的肿瘤患者,其预后效果更差(Oncoimmunology 2015,4,e984547)。IL17作为一类促炎症因子,在炎症反应、伤口愈合和肿瘤形成过程中起到某种连接作用(J.Exp.Med.2019,217,e20190297;Nat.Immunol.2019,20,1594)。有研究显示IL17促进了多种癌症的发病,这些癌症包括结肠癌(Immunity,2014,41,1052;J Immunol.2017,199,3849)、皮肤癌(Cancer Res 2010,70,10112;J Exp.Med.2019,216,195)、胰腺癌(Gastroenterology 2018,155,210)、肝癌(Dig.Dis.Sci.2016,61,474)、肺癌(Cell 2019,176,998)和骨髓瘤(Nat.Commun.2018,9,4832)等。总之,IL17诱导细胞因子、趋化因子促进髓样抑制细胞,从而进一步促进血管形成(Nat.Med.2013,19,1114;PNAS 2014,111,5664),或者提升肿瘤微环境形成抗肿瘤抑制效果,促进肿瘤的形成和发展(J Immunol.2010,184,2281;Nature,2015,522,345;Adv Exp Med Biol.2020,1240,47)。也有研究证据显示,IL17信号的激活,有助于形成肿瘤对放、化疗的抵抗,从而降低治疗效果(J Exp Med.2020,217, e20190297)。基于以上研究,抗IL17的抗体对于抗肿瘤治疗,也具有巨大的应用前景。In addition to autoimmune diseases, more and more evidences show that the dysregulation of IL17 can promote the early occurrence and the development of middle and late stages of cancer. By testing the concentration of IL17 in the serum of a series of different solid tumor patients, it was found that tumor patients with higher IL17 concentration had worse prognosis (Oncoimmunology 2015, 4, e984547). As a kind of pro-inflammatory factor, IL17 plays a certain connection role in the process of inflammatory response, wound healing and tumor formation (J. Exp. Med. 2019, 217, e20190297; Nat. Immunol. 2019, 20, 1594). Studies have shown that IL17 promotes the pathogenesis of various cancers, including colon cancer (Immunity, 2014, 41, 1052; J Immunol. 2017, 199, 3849), skin cancer (Cancer Res 2010, 70, 10112; J Exp. Med.2019,216,195), pancreatic cancer (Gastroenterology 2018,155,210), liver cancer (Dig.Dis.Sci.2016,61,474), lung cancer (Cell 2019,176,998) and myeloma (Nat.Commun.2018,9,4832) wait. In short, IL17 induces cytokines and chemokines to promote myeloid suppressor cells, thereby further promoting angiogenesis (Nat.Med.2013, 19, 1114; PNAS 2014, 111, 5664), or enhancing the tumor microenvironment to form an anti-tumor inhibitory effect , to promote the formation and development of tumors (J Immunol.2010, 184, 2281; Nature, 2015, 522, 345; Adv Exp Med Biol. 2020, 1240, 47). There is also research evidence that the activation of IL17 signaling helps to form tumor resistance to radiotherapy and chemotherapy, thereby reducing the therapeutic effect (J Exp Med.2020, 217, e20190297). Based on the above studies, anti-IL17 antibodies also have great application prospects for anti-tumor therapy.
发明内容Contents of the invention
银屑病是一种发病率较高的自身免疫疾病,欧美患病率为1%-3%,中国患病率相对较低,据估算也有600万以上患者。对于传统疗法治疗无效或耐受性较差的中至重度银屑病和(或)患有银屑病关节炎的患者,可通过注射生物制剂进行治疗;目前靶向IL17A靶点用于治疗银屑病的单克隆抗体药物,有三个(Cosentyx/Secukinumab、Taltz/Ixekizumab和Netakimab)先后被批准用于治疗银屑病、强直性脊柱炎等适应症,效果得到很好的认可。新一代靶向IL17A、IL17F的多价单克隆抗体药,比如比利时优时比公司的Bimekizumab,在头对头的临床试验中,显示出对银屑病的良好治疗效果。靶向IL17A、IL17F和IL17A/F的多价单域抗体Sonelokimab,在临床IIb期多中心的试验中,120mg或更少剂量的sonelokimab进行治疗比安慰剂显示出显著的临床益处,具有起效快,持久耐用,安全性可接受的特点(Lancet,2021,397,1564)。同时,靶向IL17A以及其它细胞因子的多价抗体,比如TNF-α、BAFF和IL13等,用于治疗肺部慢性炎症等多种适应症,也处于不同的开发阶段。Psoriasis is an autoimmune disease with a high incidence rate. The prevalence rate in Europe and the United States is 1%-3%. The prevalence rate in China is relatively low, and it is estimated that there are more than 6 million patients. For patients with moderate to severe psoriasis and/or psoriatic arthritis who are ineffective or poorly tolerated by traditional therapies, they can be treated by injecting biological agents; currently targeting IL17A is used to treat psoriasis. Three monoclonal antibody drugs for psoriasis (Cosentyx/Secukinumab, Taltz/Ixekizumab and Netakimab) have been approved for the treatment of psoriasis, ankylosing spondylitis and other indications, and the effect has been well recognized. A new generation of multivalent monoclonal antibody drugs targeting IL17A and IL17F, such as Bimekizumab from Belgium-based UCB, has shown good therapeutic effects on psoriasis in head-to-head clinical trials. Sonelokimab, a multivalent single-domain antibody targeting IL17A, IL17F and IL17A/F, in a clinical phase IIb multicenter trial, 120 mg or less dose of sonelokimab showed significant clinical benefits compared with placebo, with rapid onset of action , durability and acceptable safety (Lancet, 2021, 397, 1564). At the same time, multivalent antibodies targeting IL17A and other cytokines, such as TNF-α, BAFF, and IL13, are also in different development stages for the treatment of various indications such as chronic inflammation of the lung.
单域抗体作为一种严格的完整单体,具有分子量小、可溶性高、组织渗透性强、亲和力高和人源化程度高等优势,其特殊的结构特征和性质赋予了其一些其它常规抗体或抗体片段所不具有的特性(Annu Rev Biochem.2013,82,775)。V
HH单域抗体对可溶性靶点、包括GPCR、离子通道在内的膜蛋白靶点(Biomolecules 2021,11,63),都有很好的成药性,被广泛应用于抗肿瘤药物、抗病毒、自身免疫疾病、CAR-T免疫细胞治疗等方面得到广泛应用(BioDrugs 2020,34,11;Antib Ther 2020,3,257;Biomolecules 2021,11,238)。单域抗体改造容易,是理想的多价抗体的构建单元,可通过短小的链接序列(linker)聚合在一起构建比单价抗体具有更高抗原亲和力的多价形式,或者用于同时结合多个不同的抗原靶点。由于多价V
HH抗体的分子量较小,不仅可以采用传统CHO表达生产方式,也可以采用E.coli、P.pastoris等生产体系,且在生物药新剂型开发方面,包括皮下注射、吸入剂和口服药等,具有优于传统单克隆抗体的优势(MAbs 2018,10,778;Mucosal Immunol.2010,3,49)。
As a strict complete monomer, single domain antibody has the advantages of small molecular weight, high solubility, strong tissue permeability, high affinity and high degree of humanization. Its special structural characteristics and properties endow it with some other conventional antibodies or antibodies A property that fragments do not have (Annu Rev Biochem. 2013, 82, 775). V H H single-domain antibodies have good druggability to soluble targets, membrane protein targets including GPCRs and ion channels (
因此,开发靶向IL17A和IL17F的单/多价单域抗体,具有多方面的潜在应用,比如同时靶向IL17A、IL17A/F和IL17F等多个靶点的多价抗体,用于银屑病和强直性脊柱炎,也包括斑块银屑病、银屑病关节炎和非放射学性轴性脊柱关节炎等适应症(Expert Opin Biol Ther 2019,19,45);或者,与抗TNF-α、BAFF和IL13等细胞因子抗体中的一/多个 组成多价抗体,用于包括肺部慢性疾病、类风湿疾病、干燥综合症等多种自身免疫疾病的治疗。Therefore, the development of single/multivalent single-domain antibodies targeting IL17A and IL17F has multiple potential applications, such as multivalent antibodies targeting multiple targets such as IL17A, IL17A/F and IL17F at the same time, for psoriasis and ankylosing spondylitis, also including indications such as plaque psoriasis, psoriatic arthritis, and nonradiographic axial spondyloarthritis (Expert Opin Biol Ther 2019,19,45); or, with anti-TNF- One or more of cytokine antibodies such as α, BAFF and IL13 form a multivalent antibody, which is used for the treatment of various autoimmune diseases including chronic lung diseases, rheumatoid diseases, and Sjögren's syndrome.
本发明所要克服的技术问题是为克服现有技术中缺乏有效的靶向IL17A和IL17F的单/多价单域抗体的缺陷,提供一种IL17抗体及其制备方法和应用。The technical problem to be overcome by the present invention is to provide an IL17 antibody and its preparation method and application in order to overcome the lack of effective single/multivalent single-domain antibodies targeting IL17A and IL17F in the prior art.
本发明通过以下技术方案解决上述技术问题。The present invention solves the above-mentioned technical problems through the following technical solutions.
本发明提供一种IL17抗体,其包含重链可变区,其中:The present invention provides an IL17 antibody comprising a heavy chain variable region, wherein:
所述重链可变区包含序列如SEQ ID NO:1~24中任一项所示的CDR1、序列如SEQ ID NO:25~47中任一项所示的CDR2,以及序列如SEQ ID NO:48~71中任一项所示的CDR3。The heavy chain variable region comprises a sequence such as CDR1 shown in any one of SEQ ID NOs: 1 to 24, a sequence such as CDR2 shown in any one of SEQ ID NOs: 25 to 47, and a sequence such as SEQ ID NO : CDR3 shown in any one of 48-71.
较佳地,本发明所述重链可变区:Preferably, the heavy chain variable region of the present invention:
(1)其CDR1包含如SEQ ID NO:1所示的序列,CDR2包含如SEQ ID NO:25所示的序列,且CDR3包含如SEQ ID NO:48所示的序列;或者,(1) its CDR1 comprises the sequence shown in SEQ ID NO:1, CDR2 comprises the sequence shown in SEQ ID NO:25, and CDR3 comprises the sequence shown in SEQ ID NO:48; Or,
(2)其CDR1包含如SEQ ID NO:2所示的序列,CDR2包含如SEQ ID NO:26所示的序列,且CDR3包含如SEQ ID NO:49所示的序列;或者,(2) its CDR1 comprises the sequence shown in SEQ ID NO:2, CDR2 comprises the sequence shown in SEQ ID NO:26, and CDR3 comprises the sequence shown in SEQ ID NO:49; Or,
(3)其CDR1包含如SEQ ID NO:3所示的序列,CDR2包含如SEQ ID NO:27所示的序列,且CDR3包含如SEQ ID NO:50所示的序列;或者,(3) its CDR1 comprises the sequence shown in SEQ ID NO:3, CDR2 comprises the sequence shown in SEQ ID NO:27, and CDR3 comprises the sequence shown in SEQ ID NO:50; Or,
(4)其CDR1包含如SEQ ID NO:4所示的序列,CDR2包含如SEQ ID NO:28所示的序列,且CDR3包含如SEQ ID NO:51所示的序列;或者,(4) Its CDR1 comprises the sequence shown in SEQ ID NO:4, CDR2 comprises the sequence shown in SEQ ID NO:28, and CDR3 comprises the sequence shown in SEQ ID NO:51; Or,
(5)其CDR1包含如SEQ ID NO:5所示的序列,CDR2包含如SEQ ID NO:29所示的序列,且CDR3包含如SEQ ID NO:52所示的序列;或者,(5) its CDR1 comprises the sequence shown in SEQ ID NO:5, CDR2 comprises the sequence shown in SEQ ID NO:29, and CDR3 comprises the sequence shown in SEQ ID NO:52; or,
(6)其CDR1包含如SEQ ID NO:6所示的序列,CDR2包含如SEQ ID NO:28所示的序列,且CDR3包含如SEQ ID NO:53所示的序列;或者,(6) Its CDR1 comprises the sequence shown in SEQ ID NO:6, CDR2 comprises the sequence shown in SEQ ID NO:28, and CDR3 comprises the sequence shown in SEQ ID NO:53; Or,
(7)其CDR1包含如SEQ ID NO:7所示的序列,CDR2包含如SEQ ID NO:30所示的序列,且CDR3包含如SEQ ID NO:54所示的序列;或者,(7) Its CDR1 comprises the sequence shown in SEQ ID NO:7, CDR2 comprises the sequence shown in SEQ ID NO:30, and CDR3 comprises the sequence shown in SEQ ID NO:54; Or,
(8)其CDR1包含如SEQ ID NO:8所示的序列,CDR2包含如SEQ ID NO:31所示的序列,且CDR3包含如SEQ ID NO:55所示的序列;或者,(8) Its CDR1 comprises the sequence shown in SEQ ID NO:8, CDR2 comprises the sequence shown in SEQ ID NO:31, and CDR3 comprises the sequence shown in SEQ ID NO:55; Or,
(9)其CDR1包含如SEQ ID NO:9所示的序列,CDR2包含如SEQ ID NO:32所示的序列,且CDR3包含如SEQ ID NO:56所示的序列;或者,(9) Its CDR1 comprises the sequence shown in SEQ ID NO:9, CDR2 comprises the sequence shown in SEQ ID NO:32, and CDR3 comprises the sequence shown in SEQ ID NO:56; Or,
(10)其CDR1包含如SEQ ID NO:10所示的序列,CDR2包含如SEQ ID NO:33所示的序列,且CDR3包含如SEQ ID NO:55所示的序列;或者,(10) its CDR1 comprises the sequence shown in SEQ ID NO:10, CDR2 comprises the sequence shown in SEQ ID NO:33, and CDR3 comprises the sequence shown in SEQ ID NO:55; Or,
(11)其CDR1包含如SEQ ID NO:11所示的序列,CDR2包含如SEQ ID NO:34所 示的序列,且CDR3包含如SEQ ID NO:57所示的序列;或者,(11) its CDR1 comprises the sequence shown in SEQ ID NO:11, CDR2 comprises the sequence shown in SEQ ID NO:34, and CDR3 comprises the sequence shown in SEQ ID NO:57; Or,
(12)其CDR1包含如SEQ ID NO:12所示的序列,CDR2包含如SEQ ID NO:35所示的序列,且CDR3包含如SEQ ID NO:58所示的序列;或者,(12) Its CDR1 comprises the sequence shown in SEQ ID NO:12, CDR2 comprises the sequence shown in SEQ ID NO:35, and CDR3 comprises the sequence shown in SEQ ID NO:58; Or,
(13)其CDR1包含如SEQ ID NO:13所示的序列,CDR2包含如SEQ ID NO:36所示的序列,且CDR3包含如SEQ ID NO:59所示的序列;或者,(13) Its CDR1 comprises the sequence shown in SEQ ID NO:13, CDR2 comprises the sequence shown in SEQ ID NO:36, and CDR3 comprises the sequence shown in SEQ ID NO:59; Or,
(14)其CDR1包含如SEQ ID NO:14所示的序列,CDR2包含如SEQ ID NO:34所示的序列,且CDR3包含如SEQ ID NO:60所示的序列;或者,(14) Its CDR1 comprises the sequence shown in SEQ ID NO:14, CDR2 comprises the sequence shown in SEQ ID NO:34, and CDR3 comprises the sequence shown in SEQ ID NO:60; Or,
(15)其CDR1包含如SEQ ID NO:15所示的序列,CDR2包含如SEQ ID NO:37所示的序列,且CDR3包含如SEQ ID NO:61所示的序列;或者,(15) Its CDR1 comprises the sequence shown in SEQ ID NO:15, CDR2 comprises the sequence shown in SEQ ID NO:37, and CDR3 comprises the sequence shown in SEQ ID NO:61; Or,
(16)其CDR1包含如SEQ ID NO:9所示的序列,CDR2包含如SEQ ID NO:38所示的序列,且CDR3包含如SEQ ID NO:62所示的序列;或者,(16) Its CDR1 comprises the sequence shown in SEQ ID NO:9, CDR2 comprises the sequence shown in SEQ ID NO:38, and CDR3 comprises the sequence shown in SEQ ID NO:62; Or,
(17)其CDR1包含如SEQ ID NO:14所示的序列,CDR2包含如SEQ ID NO:34所示的序列,且CDR3包含如SEQ ID NO:60所示的序列;或者,(17) Its CDR1 comprises the sequence shown in SEQ ID NO:14, CDR2 comprises the sequence shown in SEQ ID NO:34, and CDR3 comprises the sequence shown in SEQ ID NO:60; Or,
(18)其CDR1包含如SEQ ID NO:15所示的序列,CDR2包含如SEQ ID NO:37所示的序列,且CDR3包含如SEQ ID NO:63所示的序列;或者,(18) Its CDR1 comprises the sequence shown in SEQ ID NO:15, CDR2 comprises the sequence shown in SEQ ID NO:37, and CDR3 comprises the sequence shown in SEQ ID NO:63; Or,
(19)其CDR1包含如SEQ ID NO:16所示的序列,CDR2包含如SEQ ID NO:39所示的序列,且CDR3包含如SEQ ID NO:64所示的序列;或者,(19) Its CDR1 comprises the sequence shown in SEQ ID NO:16, CDR2 comprises the sequence shown in SEQ ID NO:39, and CDR3 comprises the sequence shown in SEQ ID NO:64; Or,
(20)其CDR1包含如SEQ ID NO:17所示的序列,CDR2包含如SEQ ID NO:40所示的序列,且CDR3包含如SEQ ID NO:65所示的序列;或者,(20) Its CDR1 comprises the sequence shown in SEQ ID NO:17, CDR2 comprises the sequence shown in SEQ ID NO:40, and CDR3 comprises the sequence shown in SEQ ID NO:65; Or,
(21)其CDR1包含如SEQ ID NO:18所示的序列,CDR2包含如SEQ ID NO:41所示的序列,且CDR3包含如SEQ ID NO:66所示的序列;或者,(21) Its CDR1 comprises the sequence shown in SEQ ID NO:18, CDR2 comprises the sequence shown in SEQ ID NO:41, and CDR3 comprises the sequence shown in SEQ ID NO:66; Or,
(22)其CDR1包含如SEQ ID NO:19所示的序列,CDR2包含如SEQ ID NO:42所示的序列,且CDR3包含如SEQ ID NO:67所示的序列;或者,(22) Its CDR1 comprises the sequence shown in SEQ ID NO:19, CDR2 comprises the sequence shown in SEQ ID NO:42, and CDR3 comprises the sequence shown in SEQ ID NO:67; Or,
(23)其CDR1包含如SEQ ID NO:20所示的序列,CDR2包含如SEQ ID NO:43所示的序列,且CDR3包含如SEQ ID NO:67所示的序列;或者,(23) Its CDR1 comprises the sequence shown in SEQ ID NO:20, CDR2 comprises the sequence shown in SEQ ID NO:43, and CDR3 comprises the sequence shown in SEQ ID NO:67; Or,
(24)其CDR1包含如SEQ ID NO:21所示的序列,CDR2包含如SEQ ID NO:44所示的序列,且CDR3包含如SEQ ID NO:68所示的序列;或者,(24) Its CDR1 comprises the sequence shown in SEQ ID NO:21, CDR2 comprises the sequence shown in SEQ ID NO:44, and CDR3 comprises the sequence shown in SEQ ID NO:68; Or,
(25)其CDR1包含如SEQ ID NO:22所示的序列,CDR2包含如SEQ ID NO:45所示的序列,且CDR3包含如SEQ ID NO:69所示的序列;或者,(25) Its CDR1 comprises the sequence shown in SEQ ID NO:22, CDR2 comprises the sequence shown in SEQ ID NO:45, and CDR3 comprises the sequence shown in SEQ ID NO:69; Or,
(26)其CDR1包含如SEQ ID NO:23所示的序列,CDR2包含如SEQ ID NO:46所示的序列,且CDR3包含如SEQ ID NO:70所示的序列;或者,(26) Its CDR1 comprises the sequence shown in SEQ ID NO:23, CDR2 comprises the sequence shown in SEQ ID NO:46, and CDR3 comprises the sequence shown in SEQ ID NO:70; Or,
(27)其CDR1包含如SEQ ID NO:24所示的序列,CDR2包含如SEQ ID NO:47所示的序列,且CDR3包含如SEQ ID NO:71所示的序列。(27) Its CDR1 comprises the sequence shown in SEQ ID NO:24, CDR2 comprises the sequence shown in SEQ ID NO:47, and CDR3 comprises the sequence shown in SEQ ID NO:71.
在本发明一较佳实施方案中,所述重链可变区包含如SEQ ID NO:72~100任一项所示的序列。In a preferred embodiment of the present invention, the heavy chain variable region comprises a sequence as shown in any one of SEQ ID NO: 72-100.
本发明中所述抗体的实例包括但不限于全长抗体、重链抗体(HCAb)、抗原结合片段(Fab,Fab’、F(ab) 2、Fv片段、F(ab’) 2、scFv、di-scFv和/或dAb)、免疫缀合物、多特异性抗体(例如双特异性抗体)、抗体片段、抗体衍生物、抗体类似物或融合蛋白等,只要它们显示出所需的抗原结合活性即可;优选单域抗体或其V HH片段。 Examples of antibodies in the present invention include, but are not limited to, full-length antibodies, heavy chain antibodies (HCAb), antigen-binding fragments (Fab, Fab', F(ab) 2 , Fv fragments, F(ab') 2 , scFv, di-scFv and/or dAb), immunoconjugates, multispecific antibodies (e.g. bispecific antibodies), antibody fragments, antibody derivatives, antibody analogs or fusion proteins, etc., as long as they exhibit the desired antigen binding activity; preferably a single domain antibody or a VHH fragment thereof.
本发明还提供一种三价单域抗体,其包含如上所述的IL17抗体。The present invention also provides a trivalent single-domain antibody, which comprises the above-mentioned IL17 antibody.
本发明中,所述三价单域抗体优选含有三个自N端至C端依次连接的V HH:V HH 1-V HH 2-V HH 3。 In the present invention, the trivalent single domain antibody preferably contains three VHHs connected sequentially from the N-terminus to the C - terminus: VHH1 - VHH2 - VHH3 .
以上所述V HH 2优选抗HSA的V HH;更优选含有如SEQ ID NO:108所示的序列。 The above-mentioned VHH2 is preferably an anti-HSA VHH ; more preferably contains the sequence shown in SEQ ID NO:108.
以上所述V HH 1和所述V HH 3优选为单域抗体形式的如上所述IL17抗体;较佳地: The above-mentioned VHH 1 and the above-mentioned VHH3 are preferably IL17 antibodies in the form of single-domain antibodies; preferably:
所述V HH 1含有如上(1)~(6)中任一项所定义的CDR组合,所述V HH 3含有如上(7)~(22)中任一项所定义的CDR组合;更佳地,所述V HH 1含有如SEQ ID NO:72~79任一项所示的序列,且所述V HH 3含有如SEQ ID NO:80~100任一项所示的序列; The VHH1 contains the CDR combination as defined in any one of (1) to (6) above, and the VHH3 contains the CDR combination as defined in any one of the above (7) to (22); More preferably, the VHH1 contains the sequence shown in any one of SEQ ID NO:72-79 , and the VHH3 contains the sequence shown in any one of SEQ ID NO:80-100 ;
或者所述V HH 3含有如上(1)~(6)中任一项所定义的CDR组合,所述V HH 1含有如上(7)~(22)中任一项所定义的CDR组合;更佳地,所述V HH 3含有如SEQ ID NO:72~79任一项所示的序列,且所述V HH 1含有如SEQ ID NO:80~100任一项所示的序列。 Or the VHH3 contains the CDR combination defined in any one of (1) to (6) above, and the VHH1 contains the CDR combination defined in any one of the above (7) to (22) more preferably, the VHH3 contains the sequence shown in any one of SEQ ID NO:72~79, and the VHH1 contains the sequence shown in any one of SEQ ID NO:80~100 sequence.
在本发明一较佳实施方案中,所述V HH 1或者V HH 3含有如上(2)所定义的CDR组合,所述V HH 3或者V HH 1含有如上(7)、(10)或者(26)所定义的CDR组合。 In a preferred embodiment of the present invention, the VHH1 or VHH3 contains the CDR combination as defined in ( 2 ) above, and the VHH3 or VHH1 contains the above (7), ( 10) or the CDR combination defined in (26).
在本发明一具体实施方案中,所述V HH 1含有如SEQ ID NO:73所示的序列,且所述V HH 3含有如SEQ ID NO:80、83或者99所示的序列; In a specific embodiment of the present invention, the VHH1 contains the sequence shown in SEQ ID NO:73 , and the VHH3 contains the sequence shown in SEQ ID NO:80, 83 or 99;
在本发明另一实施方案中,所述V HH 1含有如SEQ ID NO:80、83或者99所示的序列,所述V HH 3含有如SEQ ID NO:73所示的序列。 In another embodiment of the present invention, the VHH1 contains the sequence shown in SEQ ID NO:80, 83 or 99, and the VHH3 contains the sequence shown in SEQ ID NO:73.
以上不同V HH之间优选通过接头可操作性地连接;所述接头优选含有如SEQ ID NO:107所示的序列。 The above different VHHs are preferably operably connected through a linker; the linker preferably contains the sequence shown in SEQ ID NO:107.
在本发明一具体实施方案中,所述三价单域抗体含有如SEQ ID NO:101~106任一项所示的氨基酸序列。In a specific embodiment of the present invention, the trivalent single domain antibody contains the amino acid sequence shown in any one of SEQ ID NO: 101-106.
本发明还提供一种分离的核酸,其编码如上所述的IL17抗体或者如上所述的三价单域抗体。The present invention also provides an isolated nucleic acid encoding the IL17 antibody as described above or the trivalent single domain antibody as described above.
本发明还提供一种包含如上所述的分离的核酸的表达载体。The present invention also provides an expression vector comprising the isolated nucleic acid as described above.
本发明还提供一种宿主细胞,其包含如上所述的表达载体;优选地,所述宿主细胞为原核细胞或真核细胞。The present invention also provides a host cell comprising the expression vector as described above; preferably, the host cell is a prokaryotic cell or a eukaryotic cell.
本发明还提供一种IL17抗体或者三价单域抗体的制备方法,其包含培养如上所述的宿主细胞,从培养物中获得抗体。The present invention also provides a method for preparing an IL17 antibody or a trivalent single-domain antibody, which comprises culturing the above-mentioned host cells, and obtaining the antibody from the culture.
本发明还提供一种药物组合物,其包含如上所述的IL17抗体或者如上所述的三价单域抗体。The present invention also provides a pharmaceutical composition, which comprises the above-mentioned IL17 antibody or the above-mentioned trivalent single-domain antibody.
本发明还提供如上所述的IL17抗体、如上所述的三价单域抗体或者如上所述的药物组合物在制备防治IL17相关的疾病或病症的药物中的应用。The present invention also provides the application of the above-mentioned IL17 antibody, the above-mentioned trivalent single-domain antibody or the above-mentioned pharmaceutical composition in the preparation of drugs for preventing and treating IL17-related diseases or diseases.
所述疾病或病症优选包括肺部慢性炎症、类风湿关节炎、肠道炎症、干眼病、银屑病、强直性脊柱炎和包括皮肤癌在内的多种癌症,更优选银屑病、强直性脊柱炎、肺部慢性炎症和类风湿关节炎;进一步优选银屑病、强直性脊柱炎和类风湿关节炎。The disease or condition preferably includes chronic inflammation of the lung, rheumatoid arthritis, intestinal inflammation, dry eye disease, psoriasis, ankylosing spondylitis and various cancers including skin cancer, more preferably psoriasis, ankylosing spondylitis, chronic inflammation of the lungs and rheumatoid arthritis; psoriasis, ankylosing spondylitis and rheumatoid arthritis are further preferred.
本发明还提供一种嵌合抗原受体,其包含如上所述的IL17抗体或者如上所述的三价单域抗体。The present invention also provides a chimeric antigen receptor comprising the above-mentioned IL17 antibody or the above-mentioned trivalent single-domain antibody.
本发明还提供一种抗体药物偶联物,其包含细胞毒性剂,以及如上所述的IL17抗体或者如上所述的三价单域抗体;优选地,所述细胞毒性剂为MMAF或MMAE。The present invention also provides an antibody-drug conjugate, which comprises a cytotoxic agent, and the aforementioned IL17 antibody or the aforementioned trivalent single-domain antibody; preferably, the cytotoxic agent is MMAF or MMAE.
本发明还提供一种试剂盒,其包括如上所述的IL17抗体、如上所述的三价单域抗体、如上所述的嵌合抗原受体、如上所述的抗体药物偶联物和/或如上所述的药物组合物;The present invention also provides a kit comprising the above-mentioned IL17 antibody, the above-mentioned trivalent single domain antibody, the above-mentioned chimeric antigen receptor, the above-mentioned antibody drug conjugate and/or A pharmaceutical composition as described above;
优选地,所述试剂盒还包括(i)施用抗体或其抗原结合片段或抗体药物偶联物或药物组合物的装置;和/或(ii)使用说明。Preferably, the kit further comprises (i) a device for administering the antibody or antigen-binding fragment thereof or antibody drug conjugate or pharmaceutical composition; and/or (ii) instructions for use.
本发明还提供一种套装药盒,其包含药盒A和药盒B,其中:The present invention also provides a kit of medicines, which includes a medicine box A and a medicine box B, wherein:
所述药盒A含有如上所述的IL17抗体、如上所述的三价单域抗体、如上所述的嵌合抗原受体、如上9所述的抗体药物偶联物和/或如上所述的药物组合物;The kit A contains the above-mentioned IL17 antibody, the above-mentioned trivalent single domain antibody, the above-mentioned chimeric antigen receptor, the above-mentioned antibody drug conjugate and/or the above-mentioned pharmaceutical composition;
所述药盒B含有其他抗肿瘤抗体或者包含所述其他抗肿瘤抗体的药物组合物,和/或由激素制剂、靶向小分子制剂、蛋白酶体抑制剂、成像剂、诊断剂、化疗剂、溶瘤药物、细胞毒性剂、细胞因子、共刺激分子的激活剂、抑制性分子的抑制剂以及疫苗组成的群组中的一种或多种。The kit B contains other anti-tumor antibodies or pharmaceutical compositions containing the other anti-tumor antibodies, and/or consists of hormone preparations, targeted small molecule preparations, proteasome inhibitors, imaging agents, diagnostic agents, chemotherapeutic agents, One or more of the group consisting of oncolytic drugs, cytotoxic agents, cytokines, activators of co-stimulatory molecules, inhibitors of inhibitory molecules, and vaccines.
本发明还提供一种诊断、治疗和/或预防IL17介导的疾病或病症的方法,所述方法包括向有需要的患者施用治疗有效量的如上所述的IL17抗体、如上所述的三价单域抗体、如上所述的嵌合抗原受体、如上所述的抗体药物偶联物或如上所述的药物组合物,或者使用如上所述的套装药盒治疗有需要的患者。其中,关于所述的疾病或病症如以上所定 义。The present invention also provides a method for diagnosing, treating and/or preventing IL17-mediated diseases or disorders, the method comprising administering to a patient in need a therapeutically effective amount of the above-mentioned IL17 antibody, the above-mentioned trivalent The single domain antibody, chimeric antigen receptor as above, antibody drug conjugate as above or pharmaceutical composition as above, or use the kit as above to treat patients in need. Wherein, with regard to said disease or disease as defined above.
本发明还提供一种免疫检测或者测定IL17的方法,其包括使用如上所述的IL17抗体、如上所述的三价单域抗体、如上所述的嵌合抗原受体、如上所述的抗体药物偶联物或如上所述的药物组合物与待测物孵育后检测;优选地,所述检测或者所述测定为非诊断和/或治疗目的的。The present invention also provides a method for immune detection or determination of IL17, which includes using the above-mentioned IL17 antibody, the above-mentioned trivalent single-domain antibody, the above-mentioned chimeric antigen receptor, and the above-mentioned antibody drug The conjugate or the pharmaceutical composition as described above is detected after incubation with the analyte; preferably, the detection or the determination is for non-diagnostic and/or therapeutic purposes.
本发明还提供一种联合疗法,其包括分别向有需要的患者施用如上所述的IL17抗体、如上所述的三价单域抗体、如上所述的嵌合抗原受体、如上所述的抗体药物偶联物或如上所述的药物组合物,和第二治疗剂;所述第二治疗剂较佳地包含其他抗肿瘤抗体或者包含所述其他抗肿瘤抗体的药物组合物,和/或由激素制剂、靶向小分子制剂、蛋白酶体抑制剂、成像剂、诊断剂、化疗剂、溶瘤药物、细胞毒性剂、细胞因子、共刺激分子的激活剂、抑制性分子的抑制剂以及疫苗组成的群组中的一种或多种。The present invention also provides a combination therapy, which comprises separately administering the above-mentioned IL17 antibody, the above-mentioned trivalent single domain antibody, the above-mentioned chimeric antigen receptor, the above-mentioned antibody to patients in need A drug conjugate or a pharmaceutical composition as described above, and a second therapeutic agent; the second therapeutic agent preferably comprises other anti-tumor antibodies or a pharmaceutical composition comprising the other anti-tumor antibodies, and/or consists of Hormonal agents, targeted small molecule agents, proteasome inhibitors, imaging agents, diagnostic agents, chemotherapeutic agents, oncolytic agents, cytotoxic agents, cytokines, activators of co-stimulatory molecules, inhibitors of inhibitory molecules, and vaccine components One or more of the groups.
本发明使用IL17A同源二聚体、IL17F同源二聚体和IL17A/F异二聚体,分别对羊驼进行了多次免疫,分离外周血淋巴细胞的mRNA,合成cDNA并构建V HH抗体文库,利用噬菌体展示技术、ELISA技术等方法筛选获得多个高亲和力且特异性结合IL17A、IL17F和IL17A/F的V HH抗体,随后进行V HH抗体表达纯化、亲和力、特异性和稳定性等方面的评价;通过与Anti-HSA抗体组合,构建多个多价V HH抗体,并完成表达纯化、亲和力、特异性、稳定性和epitope binning等方面的评价。结果显示,这些单价、多价V HH抗体具有独特的优点,主要表现在以下方面: The present invention uses IL17A homodimer, IL17F homodimer and IL17A/F heterodimer to immunize alpaca multiple times, isolates mRNA of peripheral blood lymphocytes, synthesizes cDNA and constructs V H H Antibody library, using phage display technology, ELISA technology and other methods to screen and obtain multiple V H H antibodies with high affinity and specific binding to IL17A, IL17F and IL17A/F, followed by V H H antibody expression purification, affinity, specificity and stability By combining with Anti-HSA antibody, construct multiple multivalent V H H antibodies, and complete the evaluation of expression purification, affinity, specificity, stability and epitope binning. The results show that these monovalent and multivalent VHH antibodies have unique advantages, mainly in the following aspects:
1.这些V HH抗体能选择性、高亲和力的结合人源细胞因子IL17A、IL17F和IL17A/F中的一个或者多个,并能够高亲和力、特异性结合食蟹猴IL17A和IL17F; 1. These V H H antibodies can selectively and high-affinity bind to one or more of human cytokines IL17A, IL17F and IL17A/F, and can bind cynomolgus monkey IL17A and IL17F with high affinity and specificity;
2.V HH抗体序列具备很好的热稳定性(T m≥65℃); 2. The V H H antibody sequence has good thermal stability (T m ≥ 65°C);
3.在结合IL17A、IL17F和IL17A/F时,具有全新的抗原决定簇;3. When combined with IL17A, IL17F and IL17A/F, it has a new antigenic determinant;
4.通过把不同V HH抗体序列组合在一起,构建的多价抗体,可同时高亲和力、特异性结合IL17A、IL17A/F和IL17F; 4. The multivalent antibody constructed by combining different V H H antibody sequences can simultaneously bind IL17A, IL17A/F and IL17F with high affinity and specificity;
5.单价、多价抗体能在E.coli的细胞间隙中高效表达、正确折叠,并可通过Protein A标签的亲和层析、离子交换层析纯化制备;5. Monovalent and multivalent antibodies can be efficiently expressed and folded correctly in the intercellular space of E.coli, and can be purified and prepared by protein A-labeled affinity chromatography and ion exchange chromatography;
6.细胞活性测试显示能有效阻断IL17A、IL17A/F与相应受体的结合;6. Cell activity test shows that it can effectively block the binding of IL17A, IL17A/F and corresponding receptors;
7.单价、多价V HH抗体有望用于IL17A、IL17F和IL17A/F等细胞因子异常调控导致的疾病; 7. Monovalent and multivalent V H H antibodies are expected to be used in diseases caused by abnormal regulation of cytokines such as IL17A, IL17F and IL17A/F;
8.V HH抗体容易与其它靶点的V HH单域抗体组合成多价抗体,用于抗肿瘤、肺部疾病、自身免疫疾病、抗病毒、抗感染等方面的治疗。 8. V H H antibodies can be easily combined with V H H single domain antibodies of other targets to form multivalent antibodies, which can be used for the treatment of anti-tumor, lung disease, autoimmune disease, anti-virus, anti-infection, etc.
图1中图1A为V HH-A19亲和层析SDS-PAGE电泳图,1B为Superdex 75 10/300层析图,图1C为Superdex 75 10/300层析后蛋白SDS-PAGE电泳图。 In Fig. 1, Fig. 1A is the SDS-PAGE electrophoresis diagram of V H H-A19 affinity chromatography, 1B is the Superdex 75 10/300 chromatogram, and Fig. 1C is the protein SDS-PAGE electrophoresis diagram after Superdex 75 10/300 chromatography.
图2为V HH-A19与人源IL17A亲和力测试图。 Fig. 2 is a diagram of the affinity test between V H H-A19 and human IL17A.
图3为V HH-AF6的T m值测试图。 Figure 3 is the test chart of T m value of V H H-AF6.
图4为V HH-A8与IL17A相互作用的Paratope分析图。 Fig. 4 is a Paratope analysis diagram of the interaction between V H H-A8 and IL17A.
图5中5A为PAF022的rProtein A sepharose FF纯化图,5B为其Hitrap Q column纯化图,5C为其Hitrap Q column纯化后取样SDS-PAGE电泳图。In Figure 5, 5A is the purification diagram of rProtein A sepharose FF of PAF022, 5B is the purification diagram of Hitrap Q column, and 5C is the SDS-PAGE electrophoresis diagram of sampling after Hitrap Q column purification.
图6为PAF022的T m测试图。 Figure 6 is the T m test chart of PAF022.
图7中:图7A为PAF022、PAF025和PAF041对IL17A的中和活性;图7B为PAF022、PAF025和PAF041对IL17A/F的中和活性。In Fig. 7: Fig. 7A shows the neutralizing activity of PAF022, PAF025 and PAF041 on IL17A; Fig. 7B shows the neutralizing activity of PAF022, PAF025 and PAF041 on IL17A/F.
图8显示V HH通过氨基酸Linker(GGGGSGGGS)与anti-HSA连接方式。 Figure 8 shows the way V H H is connected to anti-HSA through amino acid Linker (GGGGSGGGS).
实施例1 羊驼免疫、效价检测与淋巴细胞分离Example 1 Alpaca immunization, titer detection and lymphocyte separation
采购IL17A(Sino Biological,12047-HNAS)、IL17A/F(Sino Biological,CT047-HNAE)和IL7F(Sino Biological,11855-HNAE)三个蛋白,参考(Methods Mol Biol 2012,911,211;Curr Protoc Immunol 2013,103,2.17.1;Nat Protoc 2014,9,674)等文献所描述的免疫方法,对三只羊驼进行免疫。Purchase IL17A (Sino Biological, 12047-HNAS), IL17A/F (Sino Biological, CT047-HNAE) and IL7F (Sino Biological, 11855-HNAE) three proteins, refer to (Methods Mol Biol 2012, 911, 211; Curr Protoc Immunol 2013, 103, 2.17.1; Nat Protoc 2014, 9, 674) and other documents described in the immunization method, immunized three alpacas.
每次免疫按100μg蛋白/羊驼的量,在羊驼颈部肌肉两侧分别注射,总共注射两针。第一次使用完全弗氏佐剂,并记为第0天。在后续的第14天、28天、42天、56天和70天,采用不完全弗氏佐剂,进行第二到六次强化免疫。羊驼免疫效果使用ELISA法(J Vis Exp 2019,143)进行效价检测,结果显示五次免疫后的血清稀释10000倍后的效价,显著高于稀释1000倍的阴性血清,达到免疫之前设定的效价要求。For each immunization, the amount of 100 μg protein/alpaca was injected on both sides of the neck muscle of the alpaca, and a total of two injections were injected. Complete Freund's adjuvant was used for the first time and recorded as
在第五次免疫后一周、第六次免疫后一周,分别进行大规模采血,采血体积为80mL。大量采血后,使用QIAamp RNA Blood Mini Kit(QIAGEN,52304),按照说明书的实验步骤分离淋巴细胞,并完成RNA提取。One week after the fifth immunization and one week after the sixth immunization, large-scale blood collection was carried out, and the blood collection volume was 80 mL. After a large amount of blood was collected, using the QIAamp RNA Blood Mini Kit (QIAGEN, 52304), the lymphocytes were separated according to the experimental procedures in the manual, and the RNA extraction was completed.
实施例2 文库构建与筛选Example 2 Library Construction and Screening
参考单域抗体发现的常用方法,构建抗体文库和筛选(Nat Protoc 2014,9,674;Nat Protoc 2007,2,3001),获取与抗原特异性结合的高亲和力序列。简单来讲,首先将单域抗 体基因插入噬菌体展示载体,然后包装展示在噬菌体上,根据实验目的设计相应的筛选条件,最终获得符合要求的单域抗体序列。具体实验方法如下:Refer to the common method of single domain antibody discovery, construct antibody library and screen (Nat Protoc 2014, 9, 674;
将实施例1中获取的淋巴细胞RNA,按照PrimeScript TM II 1st Strand cDNA Synthesis Kit试剂盒(TAKARA,6210A)的使用说明书合成cDNA,使用KOD-FX(TOYOBO,KFX-101)通过巢式PCR扩增获得单域抗体(V HH)基因,并用BamHI-HF(NEB,#R3136L)和XhoI(NEB,#R0146L)于37℃双酶切过夜。同时采用BamHI-HF(NEB,#R3136L)和XhoI(NEB,#R0146L)酶切pComb3XSS-GFP载体(改造自pComb3XSS(BioVector NTCC Inc.),酶切位点SfiI之间的序列替换为GFP基因、同时两端添加BamHI与XhoI,使连接顺序为5’-OmpA信号肽-BamHI-GFP-XhoI-gene III-终止密码子-3’)。进一步把双酶切的V HH片段和pComb3xss-GFP载体通过T4DNA Ligase(NEB,#M0202L)在16℃孵育、连接过夜,连接产物进一步离子纯化后,电击转化TG1感受态细胞(Lucigen,60502-2),收集过夜培养的大肠杆菌完成文库构建。实验中,针对3只羊驼分别获得了6.8~9.2×10 8、片段插入率大于95%、序列多样性好的cDNA文库。 The lymphocyte RNA obtained in Example 1 was synthesized into cDNA according to the instructions of the PrimeScript TM II 1st Strand cDNA Synthesis Kit (TAKARA, 6210A), and amplified by nested PCR using KOD-FX (TOYOBO, KFX-101) The single domain antibody (V H H) gene was obtained and double digested with BamHI-HF (NEB, #R3136L) and XhoI (NEB, #R0146L) overnight at 37°C. At the same time, BamHI-HF (NEB, #R3136L) and XhoI (NEB, #R0146L) were used to digest the pComb3XSS-GFP vector (transformed from pComb3XSS (BioVector NTCC Inc.), and the sequence between the enzyme cutting site SfiI was replaced with the GFP gene, At the same time, BamHI and XhoI were added at both ends, so that the connection sequence was 5'-OmpA signal peptide-BamHI-GFP-XhoI-gene III-stop codon-3'). Further, the double-digested V H H fragment and the pComb3xss-GFP vector were incubated with T4DNA Ligase (NEB, #M0202L) at 16°C and ligated overnight. After further ion purification of the ligated product, TG1 competent cells (Lucigen, 60502- 2), collect the Escherichia coli cultured overnight to complete the library construction. In the experiment, 6.8-9.2×10 8 cDNA libraries with a fragment insertion rate of more than 95% and good sequence diversity were obtained for three alpacas.
cDNA文库采用噬菌体展示技术,以液相法进行筛选,以抗原IL17A的筛选为例,具体步骤如下:The cDNA library is screened by phage display technology and liquid phase method. Taking the screening of antigen IL17A as an example, the specific steps are as follows:
文库菌接种至50mL包含100μg/mL ampicillin和2%葡萄糖的2×YT培养基,接种后OD 600=0.1,37℃,220rpm培养直到OD 600=0.5。按照20:1(噬菌体:细菌)的比例加入辅助噬菌体M13KO7(NEB,#N0315S),混匀后于37℃静置30分钟;室温3000g离心10分钟后弃上清,并用50mL加了100μg/mL ampicillin和50μg/mL kanamycin的2×YT培养基重悬,26℃,220rpm培养过夜。 The library bacteria were inoculated into 50 mL of 2×YT medium containing 100 μg/mL ampicillin and 2% glucose, after inoculation, OD 600 =0.1, and cultured at 37° C., 220 rpm until OD 600 =0.5. Add helper phage M13KO7 (NEB, #N0315S) at a ratio of 20:1 (phage:bacteria), mix well and let stand at 37°C for 30 minutes; centrifuge at 3000g at room temperature for 10 minutes, discard the supernatant, and add 100μg/mL to 50mL Resuspend ampicillin and 50 μg/mL kanamycin in 2×YT medium, and culture overnight at 26°C and 220 rpm.
细菌培养液于4℃3000g离心10分钟,上清与PEG/NaCl溶液按体积比4:1混匀后冰浴1小时,4℃3000g离心20分钟,收集沉淀。用10mL PBS重悬,4℃3000g离心20分钟,收集上清并加入2.5mL PEG/NaCl溶液,混匀后冰浴30分钟;4℃3000g离心30分钟,沉淀用1mL PBS重悬,获得噬菌体溶液。Bacterial culture solution was centrifuged at 3000g at 4°C for 10 minutes, the supernatant was mixed with PEG/NaCl solution at a volume ratio of 4:1, then cooled on ice for 1 hour, centrifuged at 3000g at 4°C for 20 minutes, and the precipitate was collected. Resuspend in 10mL PBS, centrifuge at 3000g at 4°C for 20 minutes, collect the supernatant and add 2.5mL PEG/NaCl solution, mix well and keep on ice for 30 minutes; centrifuge at 3000g at 4°C for 30 minutes, resuspend the pellet with 1mL PBS to obtain a phage solution .
取500μL上述噬菌体溶液,先加500μL 1%(w/v)BSA溶液封闭1小时,再加入100nM生物素化标记的IL17A(ARCO Biosystems,ILA-H82Q1)溶液,室温、翻转孵育1小时,接着加入50μL经1%BSA溶液封闭的链霉亲和素磁珠(ThermoFisher scientific,11206D),室温、翻转孵育30分钟,用磁铁吸附磁珠,0.1%PBST、PBS分别洗涤10次,加三乙基胺(Sigma,T0886)溶液,混匀后室温静置10分钟,加入适量预冷的1M Tris-HCl(pH7.4)混匀,磁铁吸附后取上清感染10mL TG1菌株(预先活化,OD 600=0.5),涂布含100μg/mL ampicillin和2%葡萄糖的平板、32℃倒置培养过夜。收集平板上所有菌体 并保存菌种,梯度稀释计数的小板上单克隆挑取适量进行测序分析。 Take 500 μL of the above phage solution, first add 500 μL of 1% (w/v) BSA solution to block for 1 hour, then add 100 nM biotinylated IL17A (ARCO Biosystems, ILA-H82Q1) solution, incubate at room temperature for 1 hour, then add 50 μL of streptavidin magnetic beads (ThermoFisher scientific, 11206D) blocked by 1% BSA solution, incubate at room temperature for 30 minutes with inversion, adsorb the magnetic beads with a magnet, wash 10 times with 0.1% PBST and PBS respectively, add triethylamine (Sigma, T0886) solution, after mixing, let it stand at room temperature for 10 minutes, add an appropriate amount of pre-cooled 1M Tris-HCl (pH7.4) and mix evenly, after magnet adsorption, take the supernatant to infect 10mL TG1 strain (pre-activated, OD 600 = 0.5), spread a plate containing 100 μg/mL ampicillin and 2% glucose, and culture overnight at 32° C. upside down. Collect all the bacteria on the plate and save the strains, and pick an appropriate amount of single clones on the small plate for serial dilution and counting for sequencing analysis.
降低抗原IL17A浓度,分别使用10nM和1nM重复以上流程完成第二和第三轮筛选,争取获得高亲和力的V HH序列。 Reduce the concentration of the antigen IL17A, repeat the above process with 10nM and 1nM respectively to complete the second and third rounds of screening, and strive to obtain high-affinity VHH sequences.
抗原IL17A/F和IL17F的文库使用相同方法完成三轮筛选。Libraries of antigens IL17A/F and IL17F were screened in three rounds using the same method.
实施例3 阳性单域抗体序列获取Example 3 Acquisition of Positive Single Domain Antibody Sequence
为进一步确定高亲和力序列,将第三轮筛选获得的V HH基因克隆到表达载体pSJF2上,并用ELISA检测的方法获取阳性克隆,阳性克隆经测序分析获得V HH序列。实验方法如下: In order to further determine the high-affinity sequence, the V H H gene obtained in the third round of screening was cloned into the expression vector pSJF2, and the positive clone was obtained by ELISA detection method, and the V H H sequence of the positive clone was obtained by sequencing analysis. The experimental method is as follows:
设计两端分别携带BbsI-HF(NEB,#R3539L)和BamHI-HF(NEB,#R3136L)酶切位点的引物,从第二、第三轮筛选后子库的质粒中PCR扩增获取V HH基因,BbsI-HF和BamHI-HF酶切完成后,使用T4DNA连接酶连接相同酶切过的pSJF2载体,并转化TG1感受态细胞,涂布含100μg/mL ampicillin的LB平板,37℃培养过夜。 Primers carrying BbsI-HF (NEB, #R3539L) and BamHI-HF (NEB, #R3136L) restriction sites at both ends were designed to obtain V After digestion of H H gene, BbsI-HF and BamHI-HF, use T4 DNA ligase to connect the same digested pSJF2 vector, and transform TG1 competent cells, spread LB plate containing 100 μg/mL ampicillin, and culture at 37°C overnight.
挑取培养过夜的单克隆至加有200μL 2×YT(100μg/mL ampicillin)培养基的96孔深孔板,37℃培养4小时后,离心弃上清,并更换诱导培养基(2×YT+100μg/mL ampicillin+1%Glycine+1%TritonX-100+0.5mM IPTG+5%sucrose),27℃培养过夜。Pick the single clone cultured overnight to a 96-well deep-well plate with 200 μL of 2×YT (100 μg/mL ampicillin) medium, culture at 37°C for 4 hours, centrifuge to discard the supernatant, and replace the induction medium (2×YT +100μg/mL ampicillin+1%Glycine+1%TritonX-100+0.5mM IPTG+5%sucrose), cultivate overnight at 27°C.
将抗原IL17A(Sino Biological,12047-HNAS)用碳酸盐缓冲液稀释至1μg/mL,100μL/孔,加入ELISA板(ThermoFisher scientific,439454),4℃孵育过夜。ELISA板用PBS洗三次后,加380μL/孔的1%BSA,37℃封闭2小时;PBST洗3次,加菌液上清100μL/孔,37℃孵育1小时;PBST洗3次,加1%BSA稀释过的HRP标记的His标签抗体(Sino Biological,105327-MM02T-H),37℃孵育1小时;PBST洗5次,加入TMB显色液(Sigma,T8665),100μL/孔,室温避光反应10分钟;2M H 2SO 4终止反应,50μL/孔,酶标仪检测OD 450数值。ELISA检测结果如表1所示:与96孔深孔板一致,第一列A~H为纵列编号、第一排1-12为横排编号,通过横纵编号为坐标对克隆进行编号和定位(A1~H12),将深孔板内克隆与OD 450检测读数对应起来;A1~H12这96个检测孔的读数(第一排和第一列除外的数值)使用不同灰度突出显示,读数越大则颜色越深、理论上亲和力更高(因非定量实验、EILSA误差较大等因素,实际亲和力与ELISA检测读数并不能一一对应,仅作参考),阴性克隆读数约为0.06(G12和H12),本次检测将读数大于0.6的克隆视为阳性克隆。将161个阳性克隆进行测序分析,对测序结果进行分析,最终得到阳性候选V HH基因序列。 The antigen IL17A (Sino Biological, 12047-HNAS) was diluted to 1 μg/mL with carbonate buffer, 100 μL/well, added to an ELISA plate (ThermoFisher scientific, 439454), and incubated overnight at 4°C. After washing the ELISA plate three times with PBS, add 380 μL/well of 1% BSA, and block for 2 hours at 37°C; wash 3 times with PBST, add 100 μL/well of the bacterial supernatant, and incubate at 37°C for 1 hour; wash 3 times with PBST, add 1 HRP-labeled His-tag antibody (Sino Biological, 105327-MM02T-H) diluted with %BSA, incubated at 37°C for 1 hour; washed 5 times with PBST, added to TMB chromogenic solution (Sigma, T8665), 100 μL/well, kept at room temperature Light reaction for 10 minutes; stop the reaction with 2M H 2 SO 4 , 50 μL/well, and detect the OD 450 value with a microplate reader. The results of the ELISA test are shown in Table 1: consistent with the 96-well deep-well plate, the first column A to H is the column number, the first row 1-12 is the horizontal row number, and the clones are numbered and identified by using the horizontal and vertical numbers as coordinates. Positioning (A1~H12), corresponding to the clones in the deep well plate and the OD 450 detection readings; the readings of the 96 detection wells (A1~H12 except the values in the first row and the first column) are highlighted with different gray levels, The larger the reading, the darker the color, and the theoretically higher affinity (due to factors such as non-quantitative experiments, large errors in EILSA, etc., the actual affinity and ELISA detection readings cannot be one-to-one correspondence, only for reference), and the reading of negative clones is about 0.06 ( G12 and H12), clones with reads greater than 0.6 were considered positive clones in this test. The 161 positive clones were sequenced and analyzed, and the sequence results were analyzed to finally obtain the positive candidate V H H gene sequence.
表1 ELISA检测读数Table 1 ELISA detection readings
抗原IL17A/F和IL17F的克隆使用相同方法检测。The clones of antigens IL17A/F and IL17F were detected using the same method.
实施例4 单价单域抗体表征Example 4 Characterization of Monovalent Single Domain Antibody
1)单价单域抗体纯化1) Purification of monovalent single domain antibody
根据ELISA阳性序列多样性分析,挑选出70个代表性序列,经E.coli密码子优化后,重新合成基因并克隆到pET28a(+)载体上,构建表达质粒。表达序列N端到C端依次为OmpA信号肽、V HH、linker(氨基酸序列为GAA)和6×His标签。合成后的表达质粒转化BL21(DE3)感受态细胞,涂布平板37℃培养过夜。 According to the ELISA positive sequence diversity analysis, 70 representative sequences were selected, and after E.coli codon optimization, the gene was resynthesized and cloned into the pET28a(+) vector to construct an expression plasmid. From the N-terminal to the C-terminal of the expression sequence, there are OmpA signal peptide, V H H, linker (amino acid sequence is GAA) and 6×His tag. The synthesized expression plasmid was transformed into BL21(DE3) competent cells, and the plate was cultured overnight at 37°C.
单克隆培养过夜后的种子液按1:100接种至400mL含有50μg/mL kanamycin的2×YT培养基,37℃,250rpm培养至OD
600=0.8,加入终浓度为0.5mM的IPTG,16℃培养16小时。低速离心收菌,加入40mL的Buffer A(10mM/Tris-HCl,pH 8.0,1mM/EDTA,25%蔗糖)彻底重悬菌体,室温振荡孵育15分钟;高速离心后去上清,20mL预冷的Buffer B(30mM Tris-HCl,pH 8.0,5mM MgSO
4)重悬;再次高速离心后取上清,加入适量NaCl溶液、咪唑溶液和DTT溶液,使其终浓度分别为150mM、20mM、1mM。
加入200μL Ni NTA Beads 6FF(Smart lifesciences,SA005025),摇床缓慢混匀30分钟,转移混合液至层析柱中(Bio-Rad,7311550),收集流穿液(flow through,FT);经过5mL包含了40mM咪唑溶液的1×PBS洗涤后,用0.5mL包含了300mM咪唑的1×PBS溶液洗脱抗体。图1中A为A19亲和层析过程中样品SDS-PAGE电泳图。其中,S代表“超渗法”裂解细菌后的上清;FT代表结合流穿液;40、80代表采用包含40mM、80mM咪唑的1×PBS缓冲液清洗Ni-Resin后的缓冲液;E代表采用包含300mM咪唑的1×PBS缓冲液洗脱的样品;B代表洗脱后的Ni-Resin煮样电泳结果。Add 200 μL Ni NTA Beads 6FF (Smart lifesciences, SA005025), shake the shaker slowly for 30 minutes, transfer the mixture to the chromatography column (Bio-Rad, 7311550), collect the flow through (flow through, FT); After washing with 1×PBS containing 40 mM imidazole, the antibody was eluted with 0.5 mL of 1×PBS containing 300 mM imidazole. A in Fig. 1 is the SDS-PAGE electrophoresis image of the sample during A19 affinity chromatography. Among them, S represents the supernatant after lysing bacteria by "superosmotic method"; FT represents the combined flow-through; 40 and 80 represent the buffer after washing Ni-Resin with 1×PBS buffer containing 40mM and 80mM imidazole; E represents Samples eluted with 1×PBS buffer containing 300mM imidazole; B represents the electrophoresis results of Ni-Resin boiled samples after elution.
洗脱样品浓缩后使用Superdex 75 10/300进行分子筛层析,随后BCA(上海碧云天生物技术有限公司,P0010)法定量分析,并进行SDS-PAGE分析。图1中B为A19的Superdex 75 10/300分子筛层析图,图1中C为A19分子筛层析后蛋白成品的SDS-PAGE电泳图。可见通过2步纯化后获得了高纯度的V HH样品。 After concentration, the eluted samples were subjected to molecular sieve chromatography using Superdex 75 10/300, followed by BCA (Shanghai Biyuntian Biotechnology Co., Ltd., P0010) method for quantitative analysis and SDS-PAGE analysis. B in Figure 1 is the Superdex 75 10/300 molecular sieve chromatogram of A19, and C in Figure 1 is the SDS-PAGE electrophoresis of the finished protein product after A19 molecular sieve chromatography. It can be seen that a high-purity VHH sample was obtained after two steps of purification.
2)V HH亲和力测试 2) V H H affinity test
单价单域抗体的亲和力测试,使用Biacore T200完成,按照His标签蛋白捕获试剂盒(GE Healthcare,28-9950-56)描述的方法,将His标签单克隆抗体结合到CM5芯片(GE Healthcare,BR-1005-30)上,用于捕获带有His标签的单价V HH抗体,人源IL17A、IL17A/F和IL17F作为流动相,进行亲和力检测。 The affinity test of the monovalent single-domain antibody was completed using Biacore T200, and the His-tag monoclonal antibody was bound to the CM5 chip (GE Healthcare, BR- 1005-30), used to capture monovalent V H H antibody with His tag, human IL17A, IL17A/F and IL17F were used as mobile phase for affinity detection.
图2为抗体A19与IL17A结合与解离曲线及拟合图,经计算分析得到V HH抗体与抗原的亲和力数值。在纳入表征的70个V HH抗体中,得到29个表达纯化较好、亲和力较高的V HH抗体,其亲和力数据如表3所示,其中“×”代表不结合。 Figure 2 is the binding and dissociation curves and fitting diagrams of antibody A19 and IL17A, and the affinity value between V H H antibody and antigen was obtained through calculation and analysis. Among the 70 VHH antibodies included in the characterization, 29 VHH antibodies with better expression and purification and higher affinity were obtained. The affinity data are shown in Table 3, where "×" means no binding.
表2单价V HH抗体与人源IL17A、IL17A/F和IL17F的亲和力测试 Table 2 Affinity test of monovalent V H H antibody and human IL17A, IL17A/F and IL17F
3)V HH热稳定性分析 3) V H H thermal stability analysis
采用荧光分析法(differential scanning fluorimetry,DSF)对候选V HH抗体分子,进行热稳定性的初步筛查。在筛查过程中,使用SYPRO Orange Protein Gel Stain(Sigma,S5692),用qPCR仪器逐步加热,对所纯化的V HH抗体进行了T m值的初步测试。初步筛查时,以V HH抗体T m值≥55℃为筛选的条件,表2所列V HH抗体,都满足该要求。图3为抗体AF6采用DSF测试T m值的测试图,其T m值达到74℃。 The thermal stability of candidate VHH antibody molecules was initially screened by differential scanning fluorimetry (DSF). During the screening process, using SYPRO Orange Protein Gel Stain (Sigma, S5692) and gradually heating with a qPCR instrument, a preliminary test of the Tm value of the purified VHH antibody was performed. In the initial screening, the T m value of VHH antibody is ≥ 55°C as the screening condition, and the VHH antibodies listed in Table 2 all meet this requirement. Fig. 3 is a test chart of antibody AF6 using DSF to test the T m value, and its T m value reaches 74°C.
4)V HH特异性分析 4) V H H specificity analysis
如表2中亲和力测试结果所示,V HH抗体高亲和力结合IL17A、IL17A/F或/和IL17F中的一个或者多个抗原的能力,为进一步评价IL17家族蛋白特异性结合的能力,按照步骤2)中测试方法,检测抗体与人源IL17B(Prospec,CYT-753)、IL17C(Acro Biosystems,ILC-H52H7)、IL17D(Atagenix,ATAP01505)和IL17E(Acro Biosystems,IL5-H4221)之间的亲和力。如表2所示的29个候选V HH抗体分子,都能以较强的亲和力结合IL17A/F,以IL17A/F作为阳性对照分子,测试候选V HH抗体与IL17B、IL17C、IL17D和IL17E的特异性结合能力。测试结果显示,29个候选V HH抗体分子,与IL17A/F的结合Ru值介于30~120,而与IL17B、IL17C、IL17D和IL17E的结合Ru绝对值大多数都处于≤10,远远低于其与IL17A/F的Ru绝对值。所以,这些抗体与IL17B、IL17C、IL17D和IL17E没有明显的结合,特异性符合要求。 As shown in the affinity test results in Table 2, the ability of V H H antibody to bind one or more antigens in IL17A, IL17A/F or/and IL17F with high affinity, in order to further evaluate the ability of specific binding of IL17 family proteins, follow the steps 2) In the test method, the affinity between the antibody and human IL17B (Prospec, CYT-753), IL17C (Acro Biosystems, ILC-H52H7), IL17D (Atagenix, ATAP01505) and IL17E (Acro Biosystems, IL5-H4221) was detected . As shown in Table 2, the 29 candidate VHH antibody molecules can all bind IL17A/F with strong affinity, and IL17A/F is used as a positive control molecule to test the candidate VHH antibody and IL17B, IL17C, IL17D and IL17E specific binding capacity. The test results showed that the binding Ru values of 29 candidate V H H antibody molecules to IL17A/F ranged from 30 to 120, while most of the binding Ru values to IL17B, IL17C, IL17D and IL17E were ≤10, far from The absolute value of Ru is lower than that of IL17A/F. Therefore, these antibodies have no obvious binding to IL17B, IL17C, IL17D and IL17E, and the specificity meets the requirements.
5)V HH与食蟹猴(Cynomolgus monkey)IL17A、IL17F亲和力测试 5) Affinity test between V H H and Cynomolgus monkey (Cynomolgus monkey) IL17A, IL17F
为了进一步检测上述V HH的结合活性,并为后面抗体药在食蟹猴进行动物实验做准备,需检测V HH与食蟹猴IL17A和IL17F之间的亲和力,检测方法同步骤2),V HH通过His标签耦联到CM5芯片上作为固定,相食蟹猴的IL17A和IL17F分子作为流动相进行检测。检测结果如表3所示,其中×代表不结合。Biacore测试结果显示,除A29未检测外,其余抗体与食蟹猴IL17A和IL17F都有一定的结合能力。其次,对比表1、表3的测试结果,大多数候选V HH抗体与人源IL17A或IL17F的结合亲和力,与食蟹猴IL17A或IL17F的结合亲和力,不存在数量级的差别。 In order to further detect the binding activity of the above-mentioned VHH , and to prepare for the subsequent animal experiments of antibody drugs in cynomolgus monkeys, it is necessary to detect the affinity between VHH and cynomolgus monkey IL17A and IL17F, and the detection method is the same as step 2), V H H was coupled to a CM5 chip through a His tag as immobilization, and IL17A and IL17F molecules of cynomolgus monkey were used as mobile phase for detection. The detection results are shown in Table 3, where × represents no binding. The results of Biacore test showed that except for A29 which was not detected, all the other antibodies had certain binding ability to IL17A and IL17F in cynomolgus monkeys. Secondly, comparing the test results in Table 1 and Table 3, there is no order of magnitude difference between the binding affinity of most candidate V H H antibodies to human IL17A or IL17F, and the binding affinity to cynomolgus monkey IL17A or IL17F.
表3 V HH与Cynomolgus monkey IL17A和IL17F的亲和力 Table 3 Affinity between V H H and Cynomolgus monkey IL17A and IL17F
6)V HH的Epitope binning分析 6) Epitope binning analysis of V H H
本发明使用SPR技术对以上V HH根据Epitope的不同进行了聚类分析,方法如下所述。优化合成E.Coli密码子偏好的表达基因,使表达框序列依次为OmpA信号肽、V HH、SASA(来自US 2013/0129727 A1,可结合BSA)、His标签;合成后的基因按照1)中所述单价单域抗体纯化方法进行SASA-V HH的表达和纯化。检测时首先将BSA耦合到CM5芯片上,随后SASA-V HH(V HH 1)经过流路与BSA结合从而固定在芯片上,然后使IL17A/F经过流路与V HH 1结合形成BSA-SASA-V HH(V HH 1)-IL17A/F复合体,最后V HH(V HH 2)抗体作为流动相经过流路,此时记录响应值Ru。根据Ru值得变化来判断两个抗体是否具有相同得Epitope,如果V HH 1和V HH 2具有不同的表位,则V HH 2经过流路时能与BSA-SASA-V HH(V HH 1)-IL17A/F复合体结合,Ru值会较高;反之则低。 The present invention uses SPR technology to perform cluster analysis on the above V H H according to the difference of Epitope, and the method is as follows. Optimize and synthesize the expressed gene of E.Coli codon preference, so that the sequence of the expression frame is OmpA signal peptide, V H H, SASA (from US 2013/0129727 A1, which can be combined with BSA), and His tag; the synthesized gene follows 1) Expression and purification of SASA-V H H was performed using the monovalent single domain antibody purification method described in . During the detection, BSA is first coupled to the CM5 chip, then SASA-V H H (V H H 1 ) is combined with BSA through the flow path to be immobilized on the chip, and then IL17A/F is combined with V H H 1 through the flow path to form BSA-SASA-V H H (V H H 1 )-IL17A/F complex, and finally the V H H (V H H 2 ) antibody passed through the flow path as the mobile phase, and the response value Ru was recorded at this time. Judging whether two antibodies have the same epitope according to the change of Ru value, if V H H 1 and V H H 2 have different epitopes, then V H H 2 can be compared with BSA-SASA-V H H ( V H H 1 )-IL17A/F complex binding, the Ru value will be higher; otherwise, it will be low.
表4是使用该方法检测所获取的结果,可知部分抗体具有不同的表位,如AF5与A8、AF10与A5等Ru为正值且数值较大的组合;部分抗体则具有相同的表位,如A8与A19、AF2与A21等Ru在0附近的组合。Table 4 shows the results obtained by using this method. It can be seen that some antibodies have different epitopes, such as combinations of AF5 and A8, AF10 and A5, etc., where Ru is positive and has a large value; some antibodies have the same epitope, Such as the combination of A8 and A19, AF2 and A21 with Ru around 0.
表4 Epitope binning检测结果Table 4 Epitope binning detection results
7)采用H/D交换方法鉴定Epitope/Paratope结合位点7) Identification of Epitope/Paratope binding sites by H/D exchange method
氢氘交换质谱法(HDX-MS)表位定位能快速提供表位结构完整信息的有效方法。其原理是蛋白质酰胺键上的氢可以与重水中的氘发生可控交换,将蛋白浸入重水中,蛋白表面的氢与重水密切接触,交换速率快;而蛋白内部及形成氢键的氢难以接触重水,交换速率慢。利用质谱测定不同反应时间下肽段序列的氘代数量,进而算出每条肽段序列的氢氘交换速率,即可判断蛋白质的空间结构信息。该技术利用质谱法测量蛋白质中氨基酸残基的溶剂可及性,来确定抗原-抗体复合物在其天然溶液中的相互作用位点,并且不会对抗原或抗体引入任何修饰。Hydrogen-deuterium exchange mass spectrometry (HDX-MS) epitope mapping is an effective method that can quickly provide complete information on epitope structure. The principle is that the hydrogen on the protein amide bond can exchange controllably with the deuterium in heavy water. When the protein is immersed in heavy water, the hydrogen on the surface of the protein is in close contact with the heavy water, and the exchange rate is fast; while the hydrogen inside the protein and the hydrogen that forms the hydrogen bond are difficult to contact. Heavy water, slow exchange rate. Using mass spectrometry to measure the number of deuterated peptide sequences under different reaction times, and then calculate the hydrogen-deuterium exchange rate of each peptide sequence, the spatial structure information of the protein can be judged. The technique uses mass spectrometry to measure the solvent accessibility of amino acid residues in proteins to determine the interaction sites of antigen-antibody complexes in their native solution without introducing any modification to the antigen or antibody.
实验步骤如下:将5-10μM抗原、抗体、抗原抗体复合物按1:1的摩尔比溶解于50mM HEPES,pH 7.4,150mM NaCl,4mM TCEP溶液中,并分别在4℃下放置1小时,确保形成稳定的抗原抗体复合物。放置1小时后,在4℃条件下,将5μL不同样品分别稀释到20μL D 2O(氘)中,并放置到不同的HDX时间点(例如,0,10,60,300,900秒),通过与25μL冰冷的100mM NaH 2PO 4 1M TCEP混合来终止反应。在停止反应后立即将样品管置于干冰上,直至样品注入HDX LEAP PAL3.0平台。注射到全自动氢氘交换平台后,样品以120μL/min流速通过固定的胃蛋白酶柱,并将酶解的肽段通过2.1mm×5cm C18柱(1.9μm Hypersil Gold,Thermo Fisher)捕获并脱盐。在8分钟内,以4-40%乙腈和0.3%甲酸的线性梯度分离脱盐的肽段。在样品处理过程中,蛋白质酶解和肽段分离均在4℃下进行。 The experimental steps are as follows: Dissolve 5-10μM antigen, antibody, and antigen-antibody complex in 50mM HEPES, pH 7.4, 150mM NaCl, 4mM TCEP solution at a molar ratio of 1:1, and place them at 4°C for 1 hour to ensure Form a stable antigen-antibody complex. After standing for 1 hour, at 4°C, dilute 5 μL of different samples into 20 μL D2O (deuterium) respectively, and place them at different HDX time points (for example, 0, 10, 60, 300, 900 seconds), by cooling with 25 μL 100mM NaH 2 PO 4 mixed with 1M TCEP to stop the reaction. Place the sample tube on dry ice immediately after stopping the reaction until the sample is injected into the HDX LEAP PAL3.0 platform. After injection into the fully automatic hydrogen-deuterium exchange platform, the sample passed through the immobilized pepsin column at a flow rate of 120 μL/min, and the enzymatically digested peptides were captured and desalted by a 2.1 mm×5 cm C18 column (1.9 μm Hypersil Gold, Thermo Fisher). The desalted peptides were separated with a linear gradient of 4-40% acetonitrile and 0.3% formic acid over 8 minutes. During sample processing, protein digestion and peptide separation were performed at 4°C.
使用Orbitrap质谱仪(Orbitrap Fusion TM Tribrid TM Mass Spectrometer,Thermo Fisher) 对洗脱、分离的肽段进行分析,其测量的分辨率为65,000(m/z 400),获得氢氘交换的质谱数据。每一个样品在每一个时间点都有三次HDX测定(triplicates)。通过HDX Workbench软件来计算每个酶解肽段的质谱峰强度平均m/z质心值(一般情况下为10ppm精确度),随后转换成氘掺入百分比。计算参与空间表位的关键氨基酸序列,通过计算两个样本之间的差异来确定Delta%D的差值(比较同一肽段上氘掺入百分比的变化)。Delta%D在-5至5%之外的差异被认为是有显著性差异。此外,HDX Workbench通过student’s t test检测每个时间点的样品之间的统计学显着性(p<0.05)差异。 The eluted and separated peptides were analyzed using an Orbitrap mass spectrometer (Orbitrap Fusion TM Tribrid TM Mass Spectrometer, Thermo Fisher), with a measurement resolution of 65,000 (m/z 400), and hydrogen-deuterium exchanged mass spectrometry data were obtained. Each sample has three HDX assays (triplicates) at each time point. The average m/z centroid value of the mass spectrum peak intensity of each enzymatic peptide was calculated by HDX Workbench software (generally, the accuracy is 10ppm), and then converted into the percentage of deuterium incorporation. The key amino acid sequences involved in the spatial epitope are calculated, and the difference in Delta%D (comparing the change in the percentage of deuterium incorporation on the same peptide) is determined by calculating the difference between the two samples. Differences in Delta %D outside -5 to 5% were considered significant. In addition, HDX Workbench detects statistically significant (p<0.05) differences between samples at each time point by student's t test.
本发明采用上述方法进行了Epitope/Paratope结合位点的鉴定。以A8、AF5两个V HH为例,根据具体相互作用模式,AF5-IL17A、AF5-IL17A/F、AF5-IL17F、A8-IL17A和A8-IL17A/F总共五个相互作用,进行了H/D交换的实验。图4是A8与IL17A相互作用的Paratope分析,结果显示SGSIFNAHAMGW、CNADHTYYSDSAL是潜在Paratope,主要集中在CDR1和CDR3上。AF5与IL17A、IL17A/F和IL17F都有较强的结合亲和力,但是从参与结合的Paratope来看,其结合模式并不完全一样。整理后的检测结果见表5,A8主要通过CDR1和CDR3与IL17A和IL17A/F相互作用。AF5主要通过CDR2、CDR3的部分序列,结合IL17A;通过CDR3以及Framework-4的部分氨基酸与IL17A/F相互作用;通过CDR2结合IL17F。 The present invention uses the above method to identify the binding site of Epitope/Paratope. Taking the two V H Hs of A8 and AF5 as an example, according to the specific interaction mode, a total of five interactions, AF5-IL17A, AF5-IL17A/F, AF5-IL17F, A8-IL17A and A8-IL17A/F, were carried out. Experiment with the /D swap. Figure 4 is the Paratope analysis of the interaction between A8 and IL17A. The results show that SGSIFNAHAMGW and CNADHTYYSDSAL are potential Paratopes, mainly focusing on CDR1 and CDR3. AF5 has strong binding affinity with IL17A, IL17A/F and IL17F, but the binding mode of AF5 is not exactly the same according to the Paratope involved in the binding. The collated detection results are shown in Table 5. A8 mainly interacts with IL17A and IL17A/F through CDR1 and CDR3. AF5 mainly binds to IL17A through partial sequences of CDR2 and CDR3; interacts with IL17A/F through partial amino acids of CDR3 and Framework-4; binds to IL17F through CDR2.
表5 AF5、A8与IL17A、IL17A/F和IL17F的Paratope序列Table 5 Paratope sequences of AF5, A8 and IL17A, IL17A/F and IL17F
在获得了抗体-抗原相互作用的Paratope信息之后,进一步分析IL17A、IL17A/F和IL17F上的Epitope信息,结果见表6。After obtaining the Paratope information of the antibody-antigen interaction, the Epitope information on IL17A, IL17A/F and IL17F was further analyzed, and the results are shown in Table 6.
表6 AF5、A8与IL17A、IL17A/F和IL17F的Epitope序列Table 6 Epitope sequences of AF5, A8 and IL17A, IL17A/F and IL17F
8)候选V HH序列 8) Candidate V H H sequences
经过以上多个步骤的表征,获取了29个高亲和力、特异性好、热稳定性好的单域抗体,其氨基酸序列见表7(每条序列中粗体部分从前往后依次为CDR1、CDR2和CDR3)。After the characterization of the above multiple steps, 29 single-domain antibodies with high affinity, good specificity, and good thermal stability were obtained, and their amino acid sequences are shown in Table 7 (the bold parts in each sequence are CDR1, CDR2 from front to back) and CDR3).
表7候选V HH氨基酸序列 Table 7 Candidate V H H Amino Acid Sequences
实施例5 三价单域抗体构建、表征与功能验证Example 5 Construction, Characterization and Functional Verification of Trivalent Single Domain Antibody
1)三价单域抗体构建1) Construction of trivalent single domain antibody
经过单价单域抗体表征分析后,本发明获取了29个特异性较好、热稳定性较好的高亲和力V HH序列,这些抗体都可用于组合构建多价抗体,实现同时高亲和力结合IL17A、IL17A/F和IL17F的目的。如表1所示,鉴于A系列的V HH与IL17A的亲和力更高,而AF系列的V HH与IL17A/F和IL17F的亲和力更高,在组合的时候,将AF系列、A系列的V HH各一个,通过Anti-HSA(V HH,来自US 2007/0269422 A1,ALB11)进行组合。考虑空间位阻对V HH与IL17A、IL17A/F和IL17F相互作用的影响,将Anti-HSA置于中间,AF系列、A系列的V HH分别置于两端,且构建V HH位置颠倒的两种组合:第一种是AF系列的V HH在N端且A系列的V HH在C端,第二种是A系列的V HH在N端且AF系 列的V HH在C端。V HH通过氨基酸Linker(GGGGSGGGS)与anti-HSA连接(如图8所示)构建不同的三价抗体。 After characterization and analysis of monovalent single-domain antibodies, the present invention has obtained 29 high-affinity VHH sequences with good specificity and good thermal stability, and these antibodies can be used to combine to construct multivalent antibodies to achieve simultaneous high-affinity binding to IL17A , IL17A/F and IL17F purposes. As shown in Table 1, in view of the higher affinity between A series V H H and IL17A, and the higher affinity between AF series V H H and IL17A/F and IL17F, when combining AF series, A series One for each VHH , combined by Anti-HSA ( VHH , from US 2007/0269422 A1, ALB11). Considering the influence of steric hindrance on the interaction between V H H and IL17A, IL17A/F and IL17F, Anti-HSA was placed in the middle, and V H H of AF series and A series were placed at both ends respectively, and the V H H positions were constructed Two reversed combinations: the first one is the V H H of the AF series at the N terminal and the V H H of the A series is at the C terminal, the second is the V H H of the A series at the N terminal and the V H H of the AF series at the C end. V H H is connected to anti-HSA through amino acid Linker (GGGGSGGGS) (as shown in Figure 8) to construct different trivalent antibodies.
Anti-HSA(US 2007/0269422 A1,ALB11):Anti-HSA (US 2007/0269422 A1, ALB11):
以AF5、A8、A19和A43这4个V HH为例,构建6个不同的三价抗体,其中PAF022由AF5、Anti-HSA和A8依次连接组成,PAF025由AF5、Anti-HSA和A19依次连接组成,PAF041由AF5、Anti-HSA和A43依次连接组成,PA002由A8、Anti-HSA和AF5依次连接组成,PA026由A19、Anti-HSA和AF5依次连接组成,PA154由A43、Anti-HSA和AF5依次连接组成。6个三价单域抗体氨基酸序列(见表8),添加OmpA信号肽和终止密码子,优化密码子后合成基因并克隆到pET28a(+)上。 Taking the four V H Hs of AF5, A8, A19 and A43 as an example, six different trivalent antibodies were constructed, among which PAF022 was composed of AF5, Anti-HSA and A8 in sequence, and PAF025 was composed of AF5, Anti-HSA and A19 in sequence PAF041 is composed of AF5, Anti-HSA and A43, PA002 is composed of A8, Anti-HSA and AF5, PA026 is composed of A19, Anti-HSA and AF5, and PA154 is composed of A43, Anti-HSA and AF5 is sequentially connected to form. Amino acid sequences of 6 trivalent single domain antibodies (see Table 8), OmpA signal peptide and stop codon were added, codons were optimized and the gene was synthesized and cloned into pET28a(+).
表8 6个三价单域抗体氨基酸序列Table 8 Amino acid sequences of 6 trivalent single domain antibodies
2)三价单域抗体制备2) Preparation of trivalent single domain antibody
三价单域抗体的表达方法与单价单域抗体相同,经2步纯化后可得高纯度得三价单域抗体。纯化方法如下:三价单域抗体表达后的菌体,加入适量预冷的Lysis buffer(1×PBS,1mM DTT,1mM EDTA),超声破碎(400W,3s on/3s off,15min)后高速离心,上清液上样至预处理的HiTrap TM MabSelect SuRe Column(Cytiva,11003493),1×PBS平衡至A280基线走平至零,用100mM甘氨酸-盐酸(PH3.0)按1mL/min的流速洗脱,收集目的峰;洗脱的蛋白调节PH至10.0后,过Hitrap Q column(Cytiva,17115401),收集目的蛋白,10K超滤管浓缩,换buffer为1×PBS,并使用BCA法定量。图5中A为抗体PAF022的protein A亲和层析纯化图,图5中B为Hitrap Q column纯化图,图5中C为Hitrap Q column纯化后取样SDS-PAGE电泳图。 The expression method of the trivalent single-domain antibody is the same as that of the monovalent single-domain antibody. After two steps of purification, a high-purity trivalent single-domain antibody can be obtained. The purification method is as follows: After expressing the trivalent single-domain antibody, add an appropriate amount of pre-cooled Lysis buffer (1×PBS, 1mM DTT, 1mM EDTA), ultrasonicate (400W, 3s on/3s off, 15min) and then centrifuge at a high speed , load the supernatant to the pretreated HiTrap TM MabSelect SuRe Column (Cytiva, 11003493), equilibrate with 1×PBS until the A280 baseline goes flat to zero, wash with 100mM glycine-hydrochloric acid (PH3.0) at a flow rate of 1mL/min Remove and collect the target peak; after adjusting the pH of the eluted protein to 10.0, pass through the Hitrap Q column (Cytiva, 17115401) to collect the target protein, concentrate in a 10K ultrafiltration tube, change the buffer to 1×PBS, and quantify using the BCA method. A in Figure 5 is the protein A affinity chromatography purification diagram of the antibody PAF022, B in Figure 5 is the purification diagram of the Hitrap Q column, and C in Figure 5 is the SDS-PAGE electrophoresis diagram of the Hitrap Q column after purification.
3)三价单域抗体亲和力测试3) Trivalent single domain antibody affinity test
与单价单域抗体亲和力测试不同,三价单域抗体与人源IL17A、IL17A/F和IL17F的亲和力测试时,首先将His标签捕获试剂盒内的Anti-His耦联到CM5芯片上,用于捕获带有His标签的人源IL17A(Acro Biosystems,ILA-H82Q1)、IL17A/F(Acro Biosystems,ILF-H52W6)和IL17F(Sino Biological,11855-H07H),三价单域抗体作为流动相进行测试。检测结果见表9。Different from the affinity test of monovalent single-domain antibodies, when testing the affinity of trivalent single-domain antibodies with human IL17A, IL17A/F and IL17F, the Anti-His in the His tag capture kit was first coupled to the CM5 chip for Capture human IL17A (Acro Biosystems, ILA-H82Q1), IL17A/F (Acro Biosystems, ILF-H52W6) and IL17F (Sino Biological, 11855-H07H) with His-tagged, trivalent single domain antibody as mobile phase for testing . The test results are shown in Table 9.
表9三价抗体亲和力测试Table 9 Trivalent antibody affinity test
4)三价单域抗体热稳定性4) Thermal stability of trivalent single domain antibody
为了检测所制备的三价单域抗体的热稳定性,本发明使用差示扫描量热仪(TA Instruments,Discovery DSC2500/250/25)对所制备的对照抗体MS3091(US 2014/0314743 A1)、PAF022、PAF025、PAF041、PA002、PA026和PA154共七个三价抗体进行了热稳定性测试。具体方法是,取20μL待测样品(2mg/mL)加入样品盘,密封,放入仪器,按1℃/分钟的速度从40℃加热至95℃,记录样品盘和参比盘之间的热流变化。PA022的T m 为76.7℃,图6为PA022的热流变化曲线。阳性对照的T m值是82.0℃,PAF025和PAF041分别为79.4℃和78.6℃,PA002为76.0℃,都大于65℃,热稳定性好。 In order to detect the thermal stability of the prepared trivalent single domain antibody, the present invention uses a differential scanning calorimeter (TA Instruments, Discovery DSC2500/250/25) to test the prepared control antibody MS3091 (US 2014/0314743 A1), A total of seven trivalent antibodies, PAF022, PAF025, PAF041, PA002, PA026 and PA154, were tested for thermal stability. The specific method is to take 20 μL of the sample to be tested (2mg/mL) and add it to the sample plate, seal it, put it into the instrument, heat it from 40°C to 95°C at a speed of 1°C/min, and record the heat flow between the sample plate and the reference plate Variety. The T m of PA022 is 76.7°C, and Figure 6 shows the heat flow curve of PA022. The T m value of the positive control is 82.0°C, PAF025 and PAF041 are 79.4°C and 78.6°C, respectively, and PA002 is 76.0°C, all of which are greater than 65°C, showing good thermal stability.
5)三价单域抗体细胞功能评价5) Cell function evaluation of trivalent single domain antibody
IL17可以刺激表皮细胞(Hs27细胞系,人类表皮纤维细胞,ATCC#CRL-1634)分泌GROα,如果加入IL17的抗体,通过与IL17蛋白结合,阻断了IL17与细胞膜表面受体的作用,从而达到抑制表皮细胞分泌GROα的效果,通过检测GROα分泌量,可以得出候选抗体的IC50值。IL17 can stimulate epidermal cells (Hs27 cell line, human epidermal fibroblasts, ATCC#CRL-1634) to secrete GROα. If IL17 antibody is added, it will block the interaction between IL17 and cell membrane surface receptors by binding to IL17 protein, thereby achieving Inhibiting the effect of epidermal cells secreting GROα, by detecting the amount of GROα secretion, the IC50 value of the candidate antibody can be obtained.
本发明对三价抗体活性进行了测试,部分结果见图7、表10,所测试的6个三价抗体,其对IL17A的最大抑制率都大于83%;抗体PAF022、PAF025和PAF041的IC50都在0.03nM附近,与MS3091的IC50值0.04nM相比,活性可比,抗体PA002、PA026和PA154的IC50则比MS3091低10-20倍。所测试的6个三价抗体,其对IL17A/F的最大抑制率都大于90%,IC50都在0.6-2.7nM之间,与MS3091仅3-5倍差别。The present invention has tested the activity of trivalent antibodies, and some results are shown in Figure 7 and Table 10. The maximum inhibitory rates of the six tested trivalent antibodies to IL17A were all greater than 83%; the IC50 of antibodies PAF022, PAF025 and PAF041 were all In the vicinity of 0.03nM, compared with the IC50 value of MS3091 of 0.04nM, the activity was comparable, and the IC50 of antibodies PA002, PA026 and PA154 were 10-20 times lower than MS3091. The six trivalent antibodies tested had a maximum inhibitory rate of more than 90% on IL17A/F, and their IC50 were all between 0.6-2.7nM, which was only 3-5 times different from MS3091.
表10三价候选抗体对IL17A、IL17A/F中和能力Table 10 Trivalent Candidate Antibodies Neutralizing Ability to IL17A and IL17A/F
虽然以上描述了本发明的具体实施方式,但是本领域的技术人员应当理解,这些仅是举例说明,在不背离本发明的原理和实质的前提下,可以对这些实施方式做出多种变更或修改。因此,本发明的保护范围由所附权利要求书限定。Although the specific implementations of the present invention have been described above, those skilled in the art should understand that these are only examples, and various changes or changes can be made to these implementations without departing from the principle and essence of the present invention. Revise. Accordingly, the protection scope of the present invention is defined by the appended claims.
Claims (25)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2021/118023 WO2023035272A1 (en) | 2021-09-13 | 2021-09-13 | Il17 antibody, preparation method therefor and application thereof |
| CN202180100166.9A CN117642423A (en) | 2021-09-13 | 2021-09-13 | An IL17 antibody and its preparation method and application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2021/118023 WO2023035272A1 (en) | 2021-09-13 | 2021-09-13 | Il17 antibody, preparation method therefor and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023035272A1 true WO2023035272A1 (en) | 2023-03-16 |
Family
ID=85506977
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2021/118023 Ceased WO2023035272A1 (en) | 2021-09-13 | 2021-09-13 | Il17 antibody, preparation method therefor and application thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN117642423A (en) |
| WO (1) | WO2023035272A1 (en) |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN117843803A (en) * | 2023-12-29 | 2024-04-09 | 北京贝来药业有限公司 | Novel tandem single domain antibodies and their use in the treatment of disease |
| WO2025049345A1 (en) | 2023-08-25 | 2025-03-06 | Proteologix Us Inc. | Anti-il-13 multispecific antibody constructs and uses thereof |
| WO2025190431A1 (en) * | 2024-03-12 | 2025-09-18 | 北京贝来药业有限公司 | Pharmaceutical use of stem cell modified based on single-domain antibody genes for various diseases |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118206654B (en) * | 2024-03-05 | 2024-09-20 | 北京贝来药业有限公司 | Novel antibodies for the treatment of diseases, products and uses thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103717618A (en) * | 2011-05-05 | 2014-04-09 | 默克专利股份有限公司 | Amino acid sequences directed against il-17a, il-17f and/or il17-a/f and polypeptides comprising the same |
| CN105073775A (en) * | 2013-02-08 | 2015-11-18 | 诺华股份有限公司 | Anti-il-17a antibodies and their use in treating autoimmune and inflammatory disorders |
| CN108473568A (en) * | 2015-12-18 | 2018-08-31 | Ucb生物制药私人有限公司 | Multispecific antibody molecules specific for TNF-α, IL-17A and IL-17F |
| CN110003329A (en) * | 2019-04-12 | 2019-07-12 | 深圳普瑞金生物药业有限公司 | Polypeptide, IL17A/F single domain antibody, nucleotide sequence and kit |
| CN111699006A (en) * | 2018-02-12 | 2020-09-22 | 上海原能细胞医学技术有限公司 | IL17 antibodies and uses thereof |
-
2021
- 2021-09-13 WO PCT/CN2021/118023 patent/WO2023035272A1/en not_active Ceased
- 2021-09-13 CN CN202180100166.9A patent/CN117642423A/en active Pending
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103717618A (en) * | 2011-05-05 | 2014-04-09 | 默克专利股份有限公司 | Amino acid sequences directed against il-17a, il-17f and/or il17-a/f and polypeptides comprising the same |
| CN105073775A (en) * | 2013-02-08 | 2015-11-18 | 诺华股份有限公司 | Anti-il-17a antibodies and their use in treating autoimmune and inflammatory disorders |
| CN108473568A (en) * | 2015-12-18 | 2018-08-31 | Ucb生物制药私人有限公司 | Multispecific antibody molecules specific for TNF-α, IL-17A and IL-17F |
| CN111699006A (en) * | 2018-02-12 | 2020-09-22 | 上海原能细胞医学技术有限公司 | IL17 antibodies and uses thereof |
| CN110003329A (en) * | 2019-04-12 | 2019-07-12 | 深圳普瑞金生物药业有限公司 | Polypeptide, IL17A/F single domain antibody, nucleotide sequence and kit |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025049345A1 (en) | 2023-08-25 | 2025-03-06 | Proteologix Us Inc. | Anti-il-13 multispecific antibody constructs and uses thereof |
| CN117843803A (en) * | 2023-12-29 | 2024-04-09 | 北京贝来药业有限公司 | Novel tandem single domain antibodies and their use in the treatment of disease |
| WO2025190431A1 (en) * | 2024-03-12 | 2025-09-18 | 北京贝来药业有限公司 | Pharmaceutical use of stem cell modified based on single-domain antibody genes for various diseases |
Also Published As
| Publication number | Publication date |
|---|---|
| CN117642423A (en) | 2024-03-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109937212B (en) | B7-H3 antibody, antigen binding fragment thereof and medical application thereof | |
| WO2023035272A1 (en) | Il17 antibody, preparation method therefor and application thereof | |
| US20240368264A1 (en) | Anti-TSLP nanobodies and their applications | |
| CN106749667B (en) | A nanobody against carcinoembryonic antigen and its application | |
| CN106946989B (en) | anti-CEA antigen VHH domain and bispecific antibody containing same | |
| WO2022095926A1 (en) | Antibody targeting interleukin 36r, preparation method therefor, and application thereof | |
| WO2016173558A1 (en) | Preparation and use of anti-norovirus gii.4 type murine monoclonal antibody | |
| KR20180118671A (en) | Anti-TNF [alpha] antibodies and their functional fragments | |
| CN108178799B (en) | A kind of nano antibody against CA125 carbohydrate antigen and its application | |
| TW202321300A (en) | Il-11 humanized antibody and uses thereof | |
| CN117304315B (en) | anti-IL-6 nanobody and application thereof in IL-6 related diseases | |
| WO2019015282A1 (en) | Antibody targeting interleukin 17a and preparation method and application thereof | |
| CN119708223B (en) | A monoclonal antibody that recognizes recombinant EGF-CRM197 vaccine and its preparation method | |
| CN113234166A (en) | AFP-resistant nano antibody 1C5 and application thereof | |
| CN117700557A (en) | Antibody or antigen binding fragment specifically binding to folate receptor alpha | |
| TW202212359A (en) | Antibody that binds to human IL-33, its preparation method and use | |
| KR20180117108A (en) | Anti-TNF [alpha] antibodies and their functional fragments | |
| CN107827981B (en) | Anti-Pseudomonas exotoxin A nanobody and its application | |
| CN109206515B (en) | Fully human anti-human interleukin 17A antibody and application thereof | |
| WO2022127842A1 (en) | Bispecific antibody targeting il-17a and il-36r and application thereof | |
| CN113924315B (en) | anti-beta-NGF nano antibody and application thereof | |
| CN114702573A (en) | Hepatitis B virus surface S protein high-affinity nano antibody and application thereof | |
| CN118638235B (en) | Nanometer antibody targeting TfR1 and application thereof | |
| CN118725117B (en) | A nanoantibody targeting CD36 and its application | |
| CN120399065B (en) | Anti-IL-17A nanobody, pharmaceutical compositions, methods and uses |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21956450 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202180100166.9 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 21956450 Country of ref document: EP Kind code of ref document: A1 |