WO2023001229A1 - 嘧啶并环类衍生物及其制备方法和用途 - Google Patents
嘧啶并环类衍生物及其制备方法和用途 Download PDFInfo
- Publication number
- WO2023001229A1 WO2023001229A1 PCT/CN2022/107001 CN2022107001W WO2023001229A1 WO 2023001229 A1 WO2023001229 A1 WO 2023001229A1 CN 2022107001 W CN2022107001 W CN 2022107001W WO 2023001229 A1 WO2023001229 A1 WO 2023001229A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cycloalkyl
- alkyl
- pharmaceutically acceptable
- group
- tautomer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5383—1,4-Oxazines, e.g. morpholine ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D498/14—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/22—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains four or more hetero rings
Definitions
- the invention relates to a pyrimidocyclic derivative, a preparation method thereof, a pharmaceutical composition containing the derivative and its use as a therapeutic agent, especially as an SOS1 inhibitor.
- RAS genes are widely found in various eukaryotic organisms such as mammals, fruit flies, fungi, nematodes and yeasts, and have important physiological functions in various life systems.
- the mammalian RAS gene family has three members, namely H- RAS, K-RAS and N-RAS, each RAS gene has a similar structure, and all of them are composed of four exons, which are distributed on the DNA with a total length of about 30 kb.
- Their encoded products are monomeric globular proteins with a relative molecular mass of 21kDa.
- the activation and inactivation state of RAS protein has a significant impact on the life processes such as cell growth, differentiation, proliferation and apoptosis.
- This protein is a membrane-bound guanine nucleotide-binding protein with weak GTPase activity and regulates RAS through GTPase-activating proteins (GAPs) and guanine nucleotide exchange factors (GEFs) in normal physiological activities
- GAPs GTPase-activating proteins
- GEFs guanine nucleotide exchange factors
- the active state when the RAS protein combines with GTP to form RAS-GTP, it is the active state, and the GTPase activating protein can dephosphorylate RAS-GTP into RAS-GDP, and then inactivate it; the inactivated RAS-GDP is in the guanine Under the action of nucleotide exchange factors, it is transformed into active RAS-GTP, thereby activating a series of downstream pathways such as RAF/MER/ERK and PI3K/AKT/mTOR.
- the RAS gene is also closely related to various human diseases, especially in cancer.
- RAS is a frequently mutated oncogene, and the KRAS subtype gene mutation accounts for 86% of the total number of RAS gene mutations, and about 90% of pancreatic cancers. 30%-40% of colon cancer and 15-20% of lung cancer have different degrees of KRAS gene mutation.
- this target has always been the focus of drug research and development workers. Since the announcement of the clinical results of AMG-510, which directly acts on the KRAS-G12C target, research on KRAS inhibitors has set off a wave of upsurge at home and abroad.
- SOS Seon of sevenless homolog
- hSOS1 and hSOS2 SOS homologues
- hSOS1 and hSOS2 SOS homologues
- hSOS1 and hSOS2 SOS homologues
- hSOS1 and hSOS2 SOS homologues
- the hSOS1 protein is 150kDa in size and is a multi-structural protein domain consisting of 1333 amino acids, including an N-terminal protein domain (HD), multiple homology domains, a helical linker (HL), a RAS exchange sequence (REM) and a rich Proline C-terminal domain.
- HD N-terminal protein domain
- HL helical linker
- REM RAS exchange sequence
- RAS protein on hSOS1 There are two binding sites for RAS protein on hSOS1, namely the catalytic site and the allosteric site.
- the catalytic site binds to the RAS protein on the RAS-GDP complex to promote the exchange of guanine nucleotides
- the allosteric site binds The RAS protein on the RAS-GTP complex further enhances the catalytic effect, and then participates in and activates the signal transduction of RAS family proteins.
- SOS1 alterations in SOS1 have also been implicated in cancer.
- inherited SOS1 mutations have also been implicated in the pathogenesis of RAS disorders such as Noonan Syndrome (NS), Heart-Face-Skin Syndrome (CFC) and Type 1 Hereditary Gingival Fiber Tumor etc.
- SOS1 is also a GEF for activation of the GTPase RAC1 (Ras-associated C3 botulinum toxin substrate 1). Like RAS family proteins, RAC1 has been implicated in the pathogenesis of a variety of human cancers and other diseases.
- the object of the present invention is to provide a pyrimidocyclic derivative shown in general formula (I), or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof:
- W is selected from N or CH
- V is selected from N, -CR 2 ;
- L is selected from a bond or -CH2- ;
- X is selected from -O- or -N(R 3 )-;
- Y is selected from -C(O)- or -CH 2 -;
- Z is selected from -O- or -N(R 5 )-;
- Ring A is selected from C 6 -C 10 aryl, 5-10 membered heteroaryl, 9-10 membered bicyclic heterocyclyl or 9-10 membered fused ring;
- R 2 is selected from fluorine atom, hydroxyl group, cyano group, methoxyl group or amino group
- R 4 is selected from C 1 -C 6 alkyl, C 3 -C 6 cycloalkyl or 4-10 membered heterocyclic group; wherein said alkyl, cycloalkyl or heterocyclic group is optionally further replaced by one or more R A replaced;
- R 5 is selected from hydrogen atom, C 1 -C 6 alkyl or C 3 -C 6 cycloalkyl; wherein said alkyl or cycloalkyl is optionally further selected from one or more halogen, cyano, Substituents of hydroxy, amino, C 1 -C 3 alkoxy or C 1 -C 3 hydroxyalkyl;
- R 9 , R 10 and R 11 are each independently selected from hydrogen atom, alkyl, amino, cycloalkyl, heterocyclyl, aryl or heteroaryl, wherein the alkyl, cycloalkyl, heterocyclyl , aryl or heteroaryl are optionally further replaced by one or more selected from hydroxyl, halogen, nitro, amino, cyano, alkyl, alkoxy, cycloalkyl, heterocyclyl, aryl, heteroaryl , carboxyl or carboxylate substituents;
- n 1, 2 or 3.
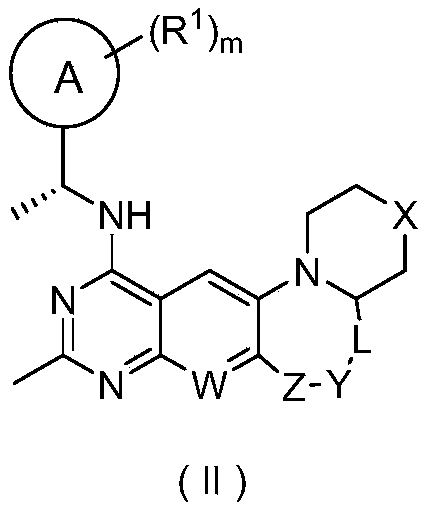
- the compound represented by general formula (I) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof is the compound represented by general formula (II) or its Stereoisomers, tautomers or pharmaceutically acceptable salts thereof:
- ring A, R 1 , W, X, Y, Z, L and m are as defined in general formula (I).
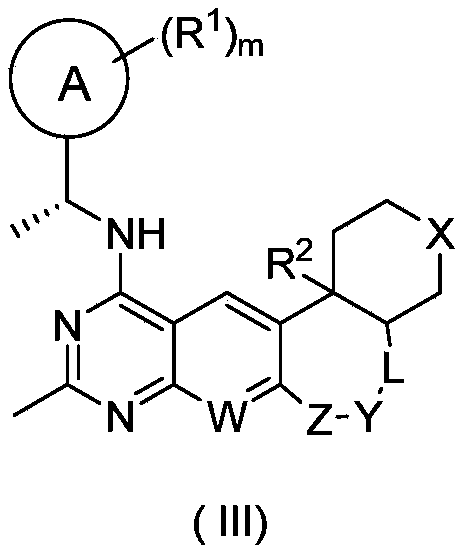
- the compound represented by general formula (I) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof is the compound represented by general formula (III) or its Stereoisomers, tautomers or pharmaceutically acceptable salts thereof:
- the compound represented by general formula (II) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof it is general formula (II-1), (II- 2), the compound shown in (II-3) or (II-4) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof:
- ring A, R 1 , R 5 , W, X and m are as defined in general formula (II).
- the compound represented by the general formula (III) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof it is the general formula (III-1), (III- 2), the compound represented by (III-3) or (III-4) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof:
- ring A, R 1 , R 2 , R 5 , W, X and m are as defined in general formula (III).
- X is selected from -N(R 3 )-;
- R 3 is selected from -C(O)R 4 ;
- R 4 is as described in the general formula (I).
- X is selected from -N(R 3 )-;
- R 3 is selected from -C(O)R 4 ;
- R4 is selected from methyl or cyclopropyl.
- Typical compounds of the invention include, but are not limited to:
- the present invention provides a pharmaceutical composition, which contains effective doses of general formula (I), (II), (III), (II-1), (II-2), (II) -3), (II-4), (III-1), (III-2), (III-3) or (III-4) compound or its stereoisomer, tautomer or Its pharmaceutically acceptable salt, and pharmaceutically acceptable carrier, excipient or their combination.
- the present invention provides a kind of general formula (I), (II), (III), (II-1), (II-2), (II-3), (II-4), (III-1), ( The compound described in III-2), (III-3) or (III-4) or its stereoisomer, tautomer or its pharmaceutically acceptable salt, or its pharmaceutical composition is used in the preparation of SOS1 inhibitor use in .
- the present invention also provides a general formula (I), (II), (III), (II-1), (II-2), (II-3), (II-4), (III-1), Compounds described in (III-2), (III-3) or (III-4) or their stereoisomers, tautomers or pharmaceutically acceptable salts thereof, or pharmaceutical compositions thereof are used in the preparation of Use in medicine for SOS1-mediated diseases, wherein the SOS1-mediated diseases are preferably RAS family protein signaling pathway-dependent cancers, cancers caused by SOS1 mutations, or hereditary diseases caused by SOS1 mutations; wherein The disease mediated by SOS1 is preferably lung cancer, pancreatic cancer, colon cancer, bladder cancer, prostate cancer, cholangiocarcinoma, gastric cancer, diffuse large B-cell lymphoma, neurofibroma, Noonan syndrome, heart face skin syndrome, hereditary gingival fibroma type I, embryonal rhabdomyosarcoma, Sertoli cell testicular tumor, or
- the present invention further provides a general formula (I), (II), (III), (II-1), (II-2), (II-3), (II-4), (III-1), Compounds described in (III-2), (III-3) or (III-4) or their stereoisomers, tautomers or pharmaceutically acceptable salts thereof, or pharmaceutical compositions thereof are used in the preparation and treatment of RAS Use in medicine for cancers related to family protein signaling pathway dependence, cancers caused by SOS1 mutations or hereditary diseases caused by SOS1 mutations.
- the present invention provides a kind of general formula (I), (II), (III), (II-1), (II-2), (II-3), (II-4), (III-1), ( The compound described in III-2), (III-3) or (III-4) or its stereoisomer, tautomer or pharmaceutically acceptable salt thereof, or its pharmaceutical composition in the preparation of the treatment of lung cancer, Pancreatic cancer, colon cancer, bladder cancer, prostate cancer, cholangiocarcinoma, gastric cancer, diffuse large B-cell lymphoma, neurofibroma, Noonan syndrome, cardiofacial skin syndrome, hereditary gingival fibroma type I, embryonal Use in medicine for rhabdomyosarcoma, Sertoli cell testicular tumor or cutaneous granulosa cell tumor.
- the pharmaceutical formulations of the present invention can be administered topically, orally, transdermally, rectally, vaginally, parenterally, intranasally, intrapulmonarily, intraocularly, intravenously, intramuscularly, intraarterially, intrathecally, intravesically, intradermally , intraperitoneally, subcutaneously, subkeratinally or by inhalation.
- the pharmaceutical composition containing the active ingredient may be in a form suitable for oral administration, such as tablets, troches, lozenges, aqueous or oily suspensions, dispersible powders or granules, emulsions, hard or soft capsules, or syrups or elixir. Tablets contain the active ingredient in admixture with non-toxic pharmaceutically acceptable excipients suitable for the manufacture of tablets.
- the formulations of the invention are suitably presented in unit dosage form and may be prepared by any of the methods well known in the art of pharmacy.
- the amount of active ingredient which can be combined with a carrier material to produce a single dosage form will vary depending upon the host treated and the particular mode of administration.
- the amount of active ingredient which can be combined with a carrier material to produce a single dosage form generally refers to that amount of the compound which produces a therapeutic effect.
- Dosage forms for the topical or transdermal administration of a compound of this invention may include powders, sprays, ointments, pastes, creams, lotions, gels, solutions, patches and inhalants.
- the active compound may be mixed under sterile conditions with a pharmaceutically acceptable carrier, in admixture with any preservatives, buffers or propellants which may be required.
- the compound of the present invention when administered to humans and animals in the form of medicine, the compound can be provided alone or in the form of a pharmaceutical composition containing Active ingredient, such as 0.1% to 99.5% (more preferably, 0.5% to 90%) active ingredient.
- Active ingredient such as 0.1% to 99.5% (more preferably, 0.5% to 90%) active ingredient.
- Examples of pharmaceutically acceptable carriers include, but are not limited to: (1) sugars, such as lactose, glucose, and sucrose; (2) starches, such as corn starch and potato starch; (3) cellulose and its derivatives, such as carboxy Sodium methylcellulose, ethylcellulose and cellulose acetate; (4) powdered gum tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients such as cocoa butter and Suppository waxes; (9) oils such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, corn oil, and soybean oil; (10) glycols such as propylene glycol; (11) polyols such as glycerin, sorbitol , mannitol and polyethylene glycol; (12) esters such as ethyl oleate and ethyl laurate; (13) agar; (14) buffers such as magnesium hydroxide and aluminum hydroxide; (15) seaweed
- antioxidants examples include, but are not limited to: (1) water-soluble antioxidants, such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite, and the like; ( 2) Oil-soluble antioxidants, such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluene (BHT), lecithin, propyl gallate, alpha-tocopherol and the like; and (3) metal chelating agents such as citric acid, ethylenediaminetetraacetic acid (EDTA), sorbitol, tartaric acid, phosphoric acid, and the like.
- water-soluble antioxidants such as ascorbic acid, cysteine hydrochloride, sodium bisulfate, sodium metabisulfite, sodium sulfite, and the like
- Oil-soluble antioxidants such as ascorbyl palmitate, butylated hydroxyanisole (BHA), butylated hydroxytoluen
- Solid dosage forms may include one or more pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or the following Any one of: (1) fillers or bulking agents, such as starch, lactose, sucrose, glucose, mannitol and/or silicic acid; (2) binders, such as carboxymethylcellulose, alginate, Gelatin, polyvinylpyrrolidone, sucrose, and/or gum arabic; (3) humectants such as glycerin; (4) disintegrants such as agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates and sodium carbonate; (5) dissolution retarders, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as cetyl alcohol and glycerol monostearate; (8) absorption (9) lubricants
- fillers or bulking agents such as starch, lactose, sucrose, glucose, mannito
- Liquid dosage forms can include pharmaceutically acceptable emulsions, microemulsions, solutions, suspensions, syrups and elixirs.
- liquid dosage forms may contain inert diluents commonly used in the art, such as water or other solvents; solubilizers and emulsifiers, such as ethanol, isopropanol, ethyl carbonate, ethyl acetate, benzene Methanol, benzyl benzoate, propylene glycol, 1,3-butanediol, oils (especially cottonseed oil, peanut oil, corn oil, germ oil, olive oil, castor oil, and sesame oil), glycerin, THF, polyethylene glycol Fatty acid esters of diols and sorbitan, and mixtures thereof.
- inert diluents commonly used in the art, such as water or other solvents
- solubilizers and emulsifiers such as ethanol, isopropanol
- Suspensions in addition to the active compounds, may contain suspending agents such as ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum hydroxide oxide, bentonite, agar and gum tragacanth and mixtures thereof.
- suspending agents such as ethoxylated isostearyl alcohols, polyoxyethylene sorbitol and sorbitan esters, microcrystalline cellulose, aluminum hydroxide oxide, bentonite, agar and gum tragacanth and mixtures thereof.
- Ointments, pastes, creams and gels may contain, in addition to the active compounds, excipients such as animal and vegetable fats, oils, waxes, paraffins, starches, tragacanth, cellulose derivatives, polyesters, etc. Glycol, polysiloxane, bentonite, silicic acid, talc and zinc oxide or mixtures thereof.
- Powders and sprays can contain, in addition to the active compounds, excipients such as lactose, talc, silicic acid, aluminum hydroxide, calcium silicates and polyamide powder, or mixtures of these substances.
- the sprays can contain other customary propellants, such as chlorofluorohydrocarbons, and volatile unsubstituted hydrocarbons, such as butane and propane.
- “Bond” means that the indicated substituent does not exist, and the two end portions of the substituent are directly connected to form a bond.
- Alkyl when used as a group or a part of a group refers to a C 1 -C 20 straight chain or branched aliphatic hydrocarbon group. It is preferably C 1 -C 10 alkyl, more preferably C 1 -C 6 alkyl.
- alkyl groups include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, sec-butyl, n-pentyl, 1,1-di Methylpropyl, 1,2-dimethylpropyl, 2,2-dimethylpropyl, 1-ethylpropyl, 2-methylbutyl, 3-methylbutyl, n-hexyl, 1 -Ethyl-2-methylpropyl, 1,1,2-trimethylpropyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 2,2-dimethyl Butyl, 1,3-dimethylbutyl, 2-ethylbutyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 2,3-dimethylbutyl Wait. Alkyl groups can be substituted or unsubstituted.
- Alkenyl means an alkyl group as defined above consisting of at least two carbon atoms and at least one carbon-carbon double bond, representative examples include but are not limited to ethenyl, 1-propenyl, 2-propenyl, 1-, 2- or 3-butenyl, etc. Alkenyl groups can be optionally substituted or unsubstituted.
- Alkynyl refers to an aliphatic hydrocarbon group containing a carbon-carbon triple bond, which may be straight or branched. Preference is given to C 2 -C 10 alkynyl, more preferably C 2 -C 6 alkynyl, most preferably C 2 -C 4 alkynyl. Examples of alkynyl groups include, but are not limited to, ethynyl, 1-propynyl, 2-propynyl, 1-, 2-, or 3-butynyl, and the like. Alkynyl groups can be substituted or unsubstituted.
- Cycloalkyl refers to saturated or partially saturated monocyclic, fused, bridged and spiro carbocyclic rings. It is preferably a C 3 -C 12 cycloalkyl group, more preferably a C 3 -C 8 cycloalkyl group, and most preferably a C 3 -C 6 cycloalkyl group.
- Examples of monocyclic cycloalkyls include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclopentenyl, cyclohexyl, cyclohexenyl, cyclohexadienyl, cycloheptyl, cycloheptyl Alkenyl, cyclooctyl, etc., preferably cyclopropyl, cyclohexenyl. Cycloalkyl groups can be optionally substituted or unsubstituted.
- “Spirocycloalkyl” refers to a polycyclic group with 5 to 18 members, two or more ring structures, and one carbon atom (called a spiro atom) shared between the single rings.
- the ring contains one or more Aromatic systems with double bonds, but none of the rings have fully conjugated ⁇ electrons. Preferably it is 6 to 14 yuan, more preferably 7 to 10 yuan.
- the spiro cycloalkyl group is divided into single spiro, double spiro or polyspiro cycloalkyl, preferably single spiro and double spiro cycloalkyl, preferably 4-membered/5-membered, 4-membered Yuan/6 yuan, 5 yuan/5 yuan or 5 yuan/6 yuan.
- spirocycloalkyl include, but are not limited to: spiro[4.5]decyl, spiro[4.4]nonyl, spiro[3.5]nonyl, spiro[2.4]heptyl.
- “Fused cycloalkyl” refers to a 5 to 18-membered, full-carbon polycyclic group containing two or more ring structures sharing a pair of carbon atoms with each other, and one or more rings may contain one or more double bonds, Aromatic systems where none of the rings have fully conjugated pi-electrons, preferably 6 to 12, more preferably 7 to 10 membered. According to the number of rings, it can be divided into bicyclic, tricyclic, tetracyclic or polycyclic condensed cycloalkyl groups, preferably bicyclic or tricyclic, more preferably 5-membered/5-membered or 5-membered/6-membered bicycloalkyl groups.
- fused cycloalkyl include, but are not limited to: bicyclo[3.1.0]hexyl, bicyclo[3.2.0]hept-1-enyl, bicyclo[3.2.0]heptyl, Decalinyl or tetrahydrophenanthrenyl.
- “Bridged cycloalkyl” refers to a 5- to 18-membered full-carbon polycyclic group that contains two or more ring structures and shares two carbon atoms that are not directly connected to each other.
- One or more rings may contain one or more Aromatic systems with multiple double bonds but none of the rings having fully conjugated pi electrons are preferably 6 to 12 membered, more preferably 7 to 10 membered. Preferably it is 6 to 14 yuan, more preferably 7 to 10 yuan.
- bridged cycloalkyl include, but are not limited to: (1s,4s)-bicyclo[2.2.1]heptyl, bicyclo[3.2.1]octyl, (1s,5s)-bis Cyclo[3.3.1]nonyl, bicyclo[2.2.2]octyl, (1r,5r)-bicyclo[3.3.2]decyl.
- Heterocyclyl “heterocyclic” or “heterocyclic” are used interchangeably in this application and all refer to non-aromatic heterocyclic groups in which one or more atoms forming the ring are heteroatoms, such as oxygen, Nitrogen, sulfur atoms, etc., including monocyclic rings, condensed rings, bridged rings, and spiro rings. It preferably has a 5 to 7 membered monocyclic ring or a 7 to 10 membered bi- or tricyclic ring, which may contain 1, 2 or 3 atoms selected from nitrogen, oxygen and/or sulfur.
- heterocyclyl examples include, but are not limited to, morpholinyl, oxetanyl, thiomorpholinyl, tetrahydropyranyl, 1,1-dioxothiomorpholinyl, piperidinyl , 2-oxopiperidinyl, pyrrolidinyl, 2-oxopyrrolidinyl, piperazin-2-one, 8-oxa-3-aza-bicyclo[3.2.1]octyl and piperazinyl .
- a heterocyclyl group can be substituted or unsubstituted.
- “Spiroheterocyclic group” refers to a polycyclic group with 5 to 18 members, two or more ring structures, and one atom shared between the single rings.
- the ring contains one or more double bonds, but no Aromatic systems with a ring having fully conjugated ⁇ -electrons, in which one or more ring atoms are selected from nitrogen, oxygen, or heteroatoms of S(O) r (where r is selected from 0, 1 or 2), and the remaining ring atoms are carbon.
- it is 6 to 14 yuan, more preferably 7 to 10 yuan.
- the spirocycloalkyl group can be divided into single spiroheterocyclyl, double spiroheterocyclyl or polyspiroheterocyclyl, preferably single spiroheterocyclyl and double spiroheterocyclyl. More preferably, it is a 4-membered/4-membered, 4-membered/5-membered, 4-membered/6-membered, 5-membered/5-membered or 5-membered/6-membered monospiro heterocyclic group.
- spiroheterocyclyl include, but are not limited to: 1,7-dioxaspiro[4.5]decyl, 2-oxa-7-azaspiro[4.4]nonyl, 7-oxaspiro[4.4]nonyl, Heteraspiro[3.5]nonyl and 5-oxaspiro[2.4]heptyl.
- “Fused heterocyclic group” refers to an all-carbon polycyclic group containing two or more ring structures that share a pair of atoms with each other.
- One or more rings may contain one or more double bonds, but none of the rings has a complete Conjugated ⁇ -electron aromatic systems in which one or more ring atoms are heteroatoms selected from nitrogen, oxygen or S(O) r (where r is selected from 0, 1 or 2) and the remaining ring atoms are carbon.
- it is 6 to 14 yuan, more preferably 7 to 10 yuan.
- bicyclic, tricyclic, tetracyclic or polycyclic fused heterocyclic groups preferably bicyclic or tricyclic, more preferably 5-membered/5-membered or 5-membered/6-membered bicyclic fused heterocyclic groups.
- fused heterocyclyl include, but are not limited to: octahydropyrrolo[3,4-c]pyrrolyl, octahydro-1H-isoindolyl, 3-azabicyclo[3.1. 0]hexyl, octahydrobenzo[b][1,4]dioxine or
- “Bridged heterocyclic group” refers to a polycyclic group with 5 to 14 members, 5 to 18 members, containing two or more ring structures, sharing two atoms that are not directly connected to each other, and one or more rings can be Aromatic systems containing one or more double bonds, but none of the rings have fully conjugated ⁇ -electrons, wherein one or more ring atoms are selected from nitrogen, oxygen or S(O) r (where r is selected from 0, 1 or 2), the remaining ring atoms are carbon. Preferably it is 6 to 14 yuan, more preferably 7 to 10 yuan.
- bridged heterocyclyl include, but are not limited to: 2-azabicyclo[2.2.1]heptyl, 2-azabicyclo[2.2.2]octyl and 2-azabicyclo[2.2.2]octyl Cyclo[3.3.2]decyl.
- Aryl means a carbocyclic aromatic system containing one or two rings, wherein the rings may be joined together in a fused fashion.
- aryl includes monocyclic or bicyclic aryl groups, such as phenyl, naphthyl, tetrahydronaphthyl aromatic groups. Preferred aryl groups are C 6 -C 10 aryl groups, more preferred aryl groups are phenyl and naphthyl, most preferably naphthyl.
- Aryl groups can be substituted or unsubstituted.
- Heteroaryl refers to an aromatic 5 to 6 membered monocyclic ring or 8 to 10 membered bicyclic ring, which may contain 1 to 4 atoms selected from nitrogen, oxygen and/or sulfur. Bicyclic heteroaryl is preferred.
- heteroaryl examples include, but are not limited to, furyl, pyridyl, 2-oxo-1,2-dihydropyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, Thienyl, isoxazolyl, oxazolyl, oxadiazolyl, imidazolyl, pyrrolyl, pyrazolyl, triazolyl, tetrazolyl, thiazolyl, isothiazolyl, 1,2,3-thiazolyl Adiazolyl, benzodioxolyl, benzothienyl, benzimidazolyl, indolyl, isoindolyl, 1,3-dioxo-isoindolyl, quinolinyl , Indazolyl, benzisothiazolyl, benzoxazolyl, benzisoxazolyl, benzothiazolyl.
- Heteroaryl groups can be substituted or unsubstituted.
- “Fused ring” refers to a polycyclic group in which two or more ring structures share a pair of atoms with each other, one or more rings may contain one or more double bonds, but at least one ring does not have complete conjugation Aromatic system of ⁇ electrons in which the ring atoms are selected from 0, one or more heteroatoms selected from nitrogen, oxygen or S(O) r (where r is selected from 0, 1 or 2), and the remaining ring atoms are carbon .
- the fused ring preferably includes a bicyclic or tricyclic fused ring, wherein the bicyclic fused ring is preferably a fused ring of an aryl or heteroaryl group and a monocyclic heterocyclic group or a monocyclic cycloalkyl group. Preferably it is 7 to 14 yuan, more preferably 8 to 10 yuan. Examples of "fused rings" include, but are not limited to:
- Alkoxy refers to a group of (alkyl-O-). Wherein, alkyl refers to relevant definitions herein. C 1 -C 6 alkoxy is preferred. Examples include, but are not limited to: methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, and the like.
- Haloalkyl means an alkyl group optionally further substituted with one or more halogens, wherein alkyl is as defined herein.
- Hydroalkyl means an alkyl group optionally further substituted with one or more hydroxy groups, wherein alkyl is as defined herein.
- Hydromethyl refers to a group in which the methyl group is optionally further substituted with one or more hydroxy groups.
- Haloalkoxy refers to a group in which the alkyl group of (alkyl-O-) is optionally further substituted by one or more halogens, wherein alkoxy is as defined herein.
- Haldroxy means an -OH group.
- Halogen refers to fluorine, chlorine, bromine and iodine.
- Amino refers to -NH2 .
- Cyano refers to -CN.
- Niro refers to -NO2 .
- Benzyl means -CH2 -phenyl.
- Carboxy refers to -C(O)OH.
- Carboxylate group refers to -C(O)O-alkyl or -C(O)O-cycloalkyl, wherein the definitions of alkyl and cycloalkyl are as above.
- DMSO dimethylsulfoxide
- Ts refers to p-toluenesulfonyl.
- T3P refers to propylphosphoric anhydride.
- DPPA diphenylphosphoryl azide
- DEA diethylamine
- X-PHOS Pd G2 refers to chloro(2-dicyclohexylphosphino-2',4',6'-triisopropyl-1,1'-biphenyl)[2-(2'-amino- 1,1'-biphenyl)]palladium(II).
- Pd(dppf)Cl 2 refers to [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride.
- Substituted means that one or more hydrogen atoms in a group, preferably up to 5, more preferably 1 to 3 hydrogen atoms are independently substituted by the corresponding number of substituents. It goes without saying that substituents are only in their possible chemical positions and that a person skilled in the art can determine (by experiment or theory) possible or impossible substitutions without undue effort. For example, an amino or hydroxyl group with free hydrogen may be unstable when bonded to a carbon atom with an unsaturated (eg, ethylenic) bond.
- R 9 , R 10 and R 11 are each independently selected from hydrogen atom, alkyl, amino, cycloalkyl, heterocyclyl, aryl or heteroaryl, wherein the alkyl, cycloalkyl, heterocyclyl , aryl or heteroaryl are optionally further replaced by one or more selected from hydroxyl, halogen, nitro, amino, cyano, alkyl, alkoxy, cycloalkyl, heterocyclyl, aryl, heteroaryl , carboxyl or carboxylate substituents;
- r 0, 1 or 2.
- the compounds of the present invention may contain asymmetric centers or chiral centers and thus exist in different stereoisomeric forms. All stereoisomeric forms of the compounds of the present invention are contemplated, including but not limited to diastereomers, enantiomers and atropisomers (atropisomers) and geometric (conformational) isomers and Mixtures thereof, such as racemic mixtures, are within the scope of the present invention.
- structures depicted herein also include all isomeric (eg, diastereoisomers, enantiomers, and atropisomers) and geometric (conformational) isomeric forms of such structures; e.g. , the R and S configurations of each asymmetric center, (Z) and (E) double bond isomers, and (Z) and (E) conformational isomers.
- isomeric e.g. diastereoisomers, enantiomers, and atropisomers
- geometric (conformational) isomeric forms of such structures e.g. , the R and S configurations of each asymmetric center, (Z) and (E) double bond isomers, and (Z) and (E) conformational isomers.
- the individual stereoisomers of the compounds of the present invention and the pair Mixtures of enantiomers, diastereoisomers and geometric (conformational) isomers are within the scope of the present invention.
- “Pharmaceutically acceptable salt” refers to certain salts of the above compounds that can maintain their original biological activity and are suitable for medical use.
- the pharmaceutically acceptable salt of the compound represented by formula (I) may be a metal salt or an amine salt with a suitable acid.
- “Pharmaceutical composition” means a mixture containing one or more compounds described herein, or a physiologically acceptable salt or prodrug thereof, and other chemical components, as well as other components such as physiologically acceptable carriers and excipients. Forming agent.
- the purpose of the pharmaceutical composition is to promote the administration to the organism, facilitate the absorption of the active ingredient and thus exert biological activity.
- the mass spectrum is measured by LC/MS instrument, and the ionization method can be ESI or APCI.
- the thin-layer chromatography silica gel plate uses Yantai Huanghai HSGF254 or Qingdao GF254 silica gel plate.
- the specification of the silica gel plate used in thin-layer chromatography (TLC) is 0.15mm-0.2mm, and the specification of thin-layer chromatography separation and purification products is 0.4mm. ⁇ 0.5mm.
- CD 3 OD deuterated methanol.
- the solution in the reaction refers to an aqueous solution.
- reaction solution was lowered to room temperature, concentrated under reduced pressure, added water (10 mL), extracted with dichloromethane (20 mL ⁇ 3), combined the organic phases, dried over anhydrous sodium sulfate, filtered, and concentrated under reduced pressure, and the obtained residue was washed with silica gel Separation and purification by column chromatography (eluent: system B) to obtain 9-methyl-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4, 5] [1,4]oxazino[3,2-g]quinazolin-11-ol 1e (256 mg, 0.94 mmol), yield 70%.
- reaction solution was lowered to room temperature, concentrated under reduced pressure, dichloromethane (20 mL) was added to the obtained residue, the pH was adjusted to alkaline with saturated sodium bicarbonate solution, the insoluble matter was removed by filtration, the layers were separated, the organic phase was collected, and the anhydrous Dry over sodium sulfate, filter, and concentrate under reduced pressure.
- Test example 1 the test that the compound of the present invention blocks the combination of SOS1 and KRAS G12C protein
- the following method is used to determine the ability of the compound of the present invention to block the interaction between SOS1 and KRAS G12C protein under in vitro conditions.
- This method uses the KRAS-G12C/SOS1 BINDING ASSAY KITS kit (Cat. No. 63ADK000CB16PEG) from Cisbio.
- Kit instructions for detailed experimental operations, please refer to the kit instructions.
- the experimental procedure is briefly described as follows: use diluent buffer (Product No. 62DLBDDF) to prepare Tag1-SOS1 and Tag2-KRAS-G12C proteins at a working solution concentration of 5X for later use.
- the test compound was dissolved in DMSO to prepare a 10 mM stock solution, and then diluted with diluent buffer for use.
- the percentage inhibition rate of the test compound at each concentration was calculated, and the nonlinear regression analysis was carried out with the concentration of the test compound on the value-inhibition rate by GraphPad Prism 5 software , to obtain the IC50 value of the compound.
- Test example 2 compound of the present invention is to OCI-AML5 cell proliferation inhibitory assay
- OCI-AML5 cells containing the SOS1N233Y mutation
- MEM ⁇ medium containing 10% fetal bovine serum, 100 U penicillin and 100 ⁇ g/mL streptomycin.
- cell viability through Luminescent Cell Viability Assay Kit was used for determination.
- test compound is first dissolved in DMSO to prepare a 10mM stock solution, and then diluted with medium to prepare a test sample.
- concentration of the compound ranges from 10000nM to 0.15nM .
- Cells in the logarithmic growth phase were seeded into 96-well cell culture plates at a density of 1000 cells per well, cultured overnight at 37°C in a 5% CO2 incubator, and then continued to culture for 120 hours after adding the test compound.
- the compound of the present invention inhibits the IC50 value of OCI-AML5 cell proliferation
- the compound of the present invention has a good inhibitory effect on OCI-AML5 cell proliferation inhibition.
- Test example 3 the assay of compound of the present invention to p-ERK1/2 inhibitory activity in DLD-1 cell
- the following method is used to determine the inhibitory activity of the compounds of the present invention on p-ERK1/2 in DLD-1 cells.
- This method uses the Advanced phospho-ERK1/2 (Thr202/tyr204) kit (Cat. No. 64AERPEH) from Cisbio, and the detailed experimental operation can refer to the kit instruction manual.
- DLD-1 cells (containing the KRAS G13D mutation) were purchased from the Cell Resource Center, Shanghai Institutes for Biological Sciences, Chinese Academy of Sciences.
- DLD-1 cells were cultured in RPMI 1640 complete medium containing 10% fetal bovine serum, 100 U penicillin, 100 ⁇ g/mL streptomycin and 1 mM Sodium Pyruvate. DLD-1 cells were plated in 96-well plates at a rate of 30,000 per well, the medium was complete medium, and cultured overnight at 37° C. in a 5% CO2 incubator.
- test compound was dissolved in DMSO to prepare a 10mM stock solution, then diluted with RPMI 1640 basal medium, and 90uL of RPMI 1640 basal medium containing the corresponding concentration of the test compound was added to each well, and the final concentration of the test compound in the reaction system was The concentration range is 10000nM-0.15nM, and cultured in a cell incubator for 3 hours and 40 minutes. Subsequently, 10 uL of hEGF prepared with RPMI 1640 basal medium (purchased from Roche, product number 11376454001) was added to make the final concentration 5 nM, and cultured in an incubator for 20 minutes.
- the compounds of the present invention inhibit the IC50 value of ERK phosphorylation in DLD-1 cells
- the compound of the present invention has a better inhibitory effect on ERK phosphorylation in DLD-1 cells.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明涉及一种嘧啶并环类衍生物、其制备方法及含有该衍生物的药物组合物在医药上的应用。具体而言,本发明涉及一种通式(I)所示的取代的嘧啶并环类衍生物、其制备方法及其可药用的盐,以及它们作为治疗剂,特别是SOS1抑制剂的用途,其中通式(I)中的各取代基的定义与说明书中的定义相同。
Description
本发明涉及一种嘧啶并环类衍生物、其制备方法及含有该衍生物的药物组合物以及其作为治疗剂特别是作为SOS1抑制剂的用途。
RAS基因广泛存在于各种真核生物如哺乳类、果蝇、真菌、线虫及酵母中,在各种生命体系中具有重要的生理功能,哺乳动物的RAS基因家族有三个成员,分别是H-RAS、K-RAS和N-RAS,各种RAS基因具有相似的结构,均由四个外显子组成,分布于全长约30kb的DNA上。它们的编码产物为相对分子质量21kDa的单体球状蛋白质。RAS蛋白的激活和非激活状态对细胞生长、分化、增殖和凋亡等生命进程有着重大的影响。该蛋白是一种膜结合的鸟嘌呤核苷酸结合蛋白,具有弱的GTP酶活性,在正常生理活动中,通过GTP酶活化蛋白(GAPs)和鸟嘌呤核苷酸交换因子(GEFs)调节RAS的活性状态,当RAS蛋白与GTP结合形成RAS-GTP时为激活状态,GTP酶活化蛋白可以使RAS-GTP去磷酸化转变成RAS-GDP,进而失活;失活的RAS-GDP在鸟嘌呤核苷酸交换因子作用下又转变成有活性的RAS-GTP,从而激活RAF/MER/ERK和PI3K/AKT/mTOR等一系列下游通路。
RAS基因同时也与人类的各种疾病密切相关,尤其在癌症方面,RAS是经常出现突变的致癌基因,其中KRAS亚型基因突变占到RAS基因突变总数的86%,约90%的胰腺癌,30%-40%的结肠癌、15-20%的肺癌中均出现不同程度的KRAS基因突变。鉴于KRAS基因突变的普遍性,该靶点一直是药物研发工作者关注的方向。从直接作用于KRAS-G12C靶点的AMG-510临床结果的公布开始,KRAS抑制剂的研究在国内外掀起一股热潮。SOS(Son of sevenless homolog)蛋白最初是在果蝇研究中被发现,是由SOS基因编码的鸟苷释放蛋白。人类有2种SOS同源体,hSOS1和hSOS2,二者都是鸟嘌呤核苷酸交换因子家族的成员,具有70%的同源性,尽管它们在结构和序列上高度相似,但各自的生理功能存在一定差异。hSOS1蛋白大小为150kDa,是由1333个氨基酸组成的多结构蛋白域,包含N端蛋白结构域(HD)、多个同源结构域、螺旋接头(HL)、RAS交换序列(REM)和富含脯氨酸的C端结构域。hSOS1上有2个与RAS蛋白的结合位点,分别是催化位点和变构位点,催化位点结合RAS-GDP复合物上的RAS蛋白促进鸟嘌呤核苷酸交换,变构位点结合RAS-GTP复合物上的RAS蛋白进一步增强催化作用,进而参与并激活RAS家族蛋白的信号转导。有研究表明,对SOS1的抑制不仅能够对野生型KRAS细胞中的RAS-RAF-MEK-ERK通路产生完全抑制,在突变的KRAS细胞系中,也能导致磷酸-ERK的活性降低50%。因此,对SOS1的抑制也能够降低RAS的活性,从而治疗由RAS基因突变或RAS蛋白过度激活导致的各种癌症,包括胰腺癌、结直肠癌、非小细胞肺癌等。
此外,癌症中亦牵涉SOS1的改变。研究表明,SOS1突变发现于胚胎性横纹肌肉瘤、塞尔托利细胞睾丸瘤、皮肤颗粒细胞肿瘤和肺腺癌中。同时,有研究已描述了在膀胱癌和前列腺癌中SOS1的过表达。除了癌症之外,遗传性SOS1突变也牵涉在RAS病的发病机理中, 所述RAS病变如像努南综合征(NS)、心-面-皮肤综合征(CFC)和1型遗传性牙龈纤维瘤病等。
SOS1也是用于激活GTP酶RAC1(Ras相关的C3肉毒杆菌毒素底物1)的GEF。与RAS家族蛋白一样,RAC1与多种人类癌症和其他疾病的发病机理有关。
市场上还未有选择性地针对于SOS1的药物上市,但已经公布了一系列相关专利,其中包括BI公司的WO2018115380A1,WO2019122129A1,Bayer公司的WO2019201848A1,Revolution公司的WO2020180768A1,WO2020180770A1等,目前处于临床试验阶段的药物为BI-1701963。但这些对于抗肿瘤研究是远远不够的,仍有必要研究和开发新的选择性SOS1激酶抑制剂,来解决未满足的医疗需求。
发明内容
本发明的目的在于提供一种通式(I)所示的嘧啶并环类衍生物,或其立体异构体、互变异构体或其可药用的盐:
其中:
W选自N或CH;
V选自N、-CR
2;
L选自键或-CH
2-;
X选自-O-或-N(R
3)-;
Y选自-C(O)-或-CH
2-;
Z选自-O-或-N(R
5)-;
环A选自C
6-C
10芳基、5-10元杂芳基、9-10元双环杂环基或9-10元稠合环;
R
1相同或不同,各自独立地选自氢原子、卤素、氰基、氨基、硝基、C
1-C
6烷基、C
3-C
6环烷基、4-11元杂环基、C
6-C
10芳基或5-10元杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自卤素、氰基、羟基、氨基、烷氧基、=O、-C(O)R
6、-C(O)OR
6、-NHC(O)R
6、-NHC(O)OR
6、-NR
7R
8、-C(O)NR
7R
8、-CH
2NR
7R
8、-CH
2NHC(O)OR
6或-SO
2R
6的取代基所取代;
R
2选自氟原子、羟基、氰基、甲氧基或氨基;
R
3选自氢原子、C
1-C
6烷基、C
3-C
6环烷基或-C(O)R
4;其中所述的烷基或环烷基任选进一步被一个或多个选自卤素、氰基、羟基、氨基、C
1-C
3烷氧基、C
1-C
3羟基烷基、4-11元杂环基所取代;其中所述的4-11元杂环基任选进一步被C
1-C
6烷基、C
3-C
6环烷基、烷氧基、=O、 -C(O)R
6、-C(O)OR
6、-NHC(O)R
6、-NHC(O)OR
6、-NR
7R
8、-C(O)NR
7R
8、-CH
2NR
7R
8、-CH
2NHC(O)OR
6或-SO
2R
6的取代基所取代;
R
4选自C
1-C
6烷基、C
3-C
6环烷基或4-10元杂环基;其中所述的烷基、环烷基或杂环基任选进一步被一个或多个R
A取代;
R
A各自独立地选自卤素、氰基、羟基、氨基、C
1-C
3烷氧基、C
1-C
3羟基烷基、C
1-C
3卤代烷基或4-11元杂环基;其中所述的4-11元杂环基任选进一步被C
1-C
6烷基、C
3-C
6环烷基、烷氧基、=O、-C(O)R
6、-C(O)OR
6、-NHC(O)R
6、-NHC(O)OR
6、-NR
7R
8、-C(O)NR
7R
8、-CH
2NR
7R
8、-CH
2NHC(O)OR
6或-SO
2R
6的取代基所取代;
R
5选自于氢原子、C
1-C
6烷基或C
3-C
6环烷基;其中所述的烷基或环烷基任选进一步被一个或多个选自卤素、氰基、羟基、氨基、C
1-C
3烷氧基或C
1-C
3羟基烷基的取代基所取代;
R
6各自独立地选自氢原子、烷基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、卤代烷基、卤代烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R
9、-C(O)OR
9、-OC(O)R
9、-NR
10R
11、-C(O)NR
10R
11、-SO
2NR
10R
11或-NR
10C(O)R
11的取代基所取代;
R
7和R
8各自独立地选自氢原子、羟基、卤素、烷基、烷氧基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、烷氧基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R
9、-C(O)OR
9、-OC(O)R
9、-NR
10R
11、-C(O)NR
10R
11、-SO
2NR
10R
11或-NR
10C(O)R
11的取代基所取代;
或者,R
7和R
8与它们相连接的原子一起形成一个4~8元杂环基,其中4~8元杂环基内含有一个或多个N、O、S或SO
2,并且所述的4~8元杂环基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R
9、-C(O)OR
9、-OC(O)R
9、-NR
10R
11、-C(O)NR
10R
11、-SO
2NR
10R
11或-NR
10C(O)R
11的取代基所取代;
R
9、R
10和R
11各自独立地选自氢原子、烷基、氨基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氨基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、羧基或羧酸酯基的取代基所取代;
m为1、2或3。
在本发明的优选方案中,通式(I)所示的化合物或其立体异构体、互变异构体或其可药用的盐,其为通式(II)所示的化合物或其立体异构体、互变异构体或其可药用的盐:
其中环A、R
1、W、X、Y、Z、L和m的定义如通式(I)中所述。
在本发明的优选方案中,通式(I)所示的化合物或其立体异构体、互变异构体或其可药用的盐,其为通式(III)所示的化合物或其立体异构体、互变异构体或其可药用的盐:
其中环A、R
1、R
2、W、X、Y、Z、L和m的定义如通式(I)中所述。
在本发明的优选方案中,通式(II)所示的化合物或其立体异构体、互变异构体或其可药用的盐,其为通式(II-1)、(II-2)、(II-3)或(II-4)所示的化合物或其立体异构体、互变异构体或其可药用的盐:
其中:环A、R
1、R
5、W、X和m的定义如通式(II)中所述。
在本发明的优选方案中,通式(III)所示的化合物或其立体异构体、互变异构体或其可药用的盐,其为通式(III-1)、(III-2)、(III-3)或(III-4)所示的化合物或其立体异构体、互变异构体或其可药用的盐:
其中:环A、R
1、R
2、R
5、W、X和m的定义如通式(III)中所述。
在本发明的优选方案中,通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,
其中:R
1和m的定义如通式(I)中所述。
在本发明的优选方案中,通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,
在本发明的优选方案中,通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中X为-O-。
在本发明的优选方案中,通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中:
X选自-N(R
3)-;
R
3选自-C(O)R
4;
R
4的定义如通式(I)中所述。
在本发明的优选方案中,通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中:
X选自-N(R
3)-;
R
3选自-C(O)R
4;
R
4选自甲基或环丙基。
在本发明的优选方案中,通式(II-2)、(II-4)、(III-2)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中R
5选自甲基。
本发明的典型化合物包括,但不限于:
注:如果在画出的结构和给出的该结构的名称之间有差异,则画出的结构将给予更大的权重。
更进一步,本发明提供一种药物组合物,所述的药物组合物含有有效剂量的通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,及可药用的载体、赋形剂或它们的组合。
本发明提供一种通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,或其药物组合物在制备SOS1抑制剂中的用途。
本发明还提供一种通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,或其药物组合物在制备治疗由SOS1介导的疾病的药物中的用途,其中所述的由SOS1介导的疾病优选为RAS家族蛋白信号传导通路依赖性相关的癌症、SOS1突变导致的癌症或SOS1突变导致的遗传性疾病;其中所述的由SOS1介导的疾病优选为肺癌、胰腺癌、结肠癌、膀胱癌、前列腺癌、胆管癌、胃癌、弥漫性大B细胞淋巴瘤、神经纤维瘤、努南综合征、心面皮肤综合征、Ⅰ型遗传性齿龈纤维瘤、胚胎性横纹肌肉瘤、塞尔托利细胞睾丸瘤或皮肤颗粒细胞瘤。
本发明进一步提供一种通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,或其药物组合物在制备治疗RAS家族蛋白信号传导通路依赖性相关的癌症、SOS1突变导致的癌症或SOS1突变导致的遗传性疾病的药物中的用途。
本发明提供一种通式(I)、(II)、(III)、(II-1)、(II-2)、(II-3)、(II-4)、(III-1)、(III-2)、(III-3)或(III-4)所述的化合物或其立体异构体、互变异构体或其可药用的盐,或其药物组合物在制备治疗肺癌、胰腺癌、结肠癌、膀胱癌、前列腺癌、胆管癌、胃癌、弥漫性大B细胞淋巴瘤、神经纤维瘤、努南综合征、心面皮肤综合征、Ⅰ型遗传性齿龈纤维瘤、胚胎性横纹肌肉瘤、塞尔托利细胞睾丸瘤或皮肤颗粒细胞瘤的药物中的用途。
本发明的药物制剂可以经局部、口服、经皮、经直肠、经阴道、非经肠、鼻内、肺内、眼内、静脉内、肌肉内、动脉内、鞘内、囊内、皮内、腹膜内、皮下、角质层下或者通过吸入进行给药。含活性成分的药物组合物可以是适用于口服的形式,例如片剂、糖锭剂、锭剂、水或油混悬液、可分散粉末或颗粒、乳液、硬或软胶囊,或糖浆剂或酏剂。片剂含有活性成分和用于混合的适宜制备片剂的无毒的可药用的赋形剂。
本发明的制剂适合以单位计量的形式存在,并且所述制剂可借由在制药技术中所众所周知的任何方法进行制备。能够通过与载体物质进行组合,从而产生单一剂型的活性成分的量可以依据所治疗的宿主及特定给药模式而变化。能够通过与载体物质进行组合从而产生单一剂型的活性成分的量通常指的是能够产生治疗效果的化合物的量。
用于本发明化合物的局部或者透皮给药的剂型可包括粉末、喷雾剂、软膏剂、糊剂、乳膏剂、洗剂、凝胶剂、溶液、贴片及吸入剂。活性化合物可在无菌条件下与药学上可接受的载剂进行混合,并且其可与可能需要的任何防腐剂、缓冲剂或者推进剂进行混合。
当本发明的化合物以药物的形式对人类及动物进行给药时,所述化合物可进行单独提供或者以药物组合物的形式提供,所述药物组合物含有与药学上可接受的载剂进行组合的活性成分,例如0.1%至99.5%(更优选地,0.5%至90%)的活性成分。
药学上可接受的载剂的实例包括但不限于:(1)糖,例如乳糖、葡萄糖及蔗糖;(2)淀粉,例如玉米淀粉及马铃薯淀粉;(3)纤维素及其衍生物,例如羧甲基纤维素钠、乙基纤维素及乙 酸纤维素;(4)粉末状黄蓍胶;(5)麦芽;(6)明胶;(7)滑石;(8)赋形剂,例如可可脂及栓剂蜡;(9)油,例如花生油、棉籽油、红花油、芝麻油、橄榄油、玉米油及大豆油;(10)二醇,例如丙二醇;(11)多元醇,例如甘油、山梨糖醇、甘露糖醇及聚乙二醇;(12)酯,例如油酸乙酯及月桂酸乙酯;(13)琼脂;(14)缓冲剂,例如氢氧化镁及氢氧化铝;(15)海藻酸;(16)无热原水;(17)等渗盐水;(18)林格氏溶液(Ringer's solution);(19)乙醇;(20)磷酸盐缓冲溶液;(21)环糊精,例如连接于纳米粒子的靶向配体,例如AccurinsTM;及(22)用于药物制剂中的其它无毒兼容物质,例如聚合物基组合物。
药学上可接受的抗氧化剂的实例包括但不限于:(1)水溶性抗氧化剂,例如抗坏血酸、半胱胺酸盐酸盐、硫酸氢钠、偏亚硫酸氢钠、亚硫酸钠及其类似物;(2)油溶性抗氧化剂,例如抗坏血酸棕榈酸酯、丁基化羟基苯甲醚(BHA)、丁基化羟基甲苯(BHT)、卵磷脂、五倍子酸丙酯、α-生育酚及其类似物;及(3)金属螯合剂,例如柠檬酸、乙二胺四乙酸(EDTA)、山梨糖醇、酒石酸、磷酸及其类似物。固体剂型(例如胶囊、锭剂丸剂、糖衣锭、粉末、颗粒剂及其类似物)可包括一种或者多种药学上可接受的载剂,例如柠檬酸钠或者磷酸二钙,和/或以下任意其中之一:(1)填充剂或增量剂,例如淀粉、乳糖、蔗糖、葡萄糖、甘露糖醇及/或者硅酸;(2)黏合剂,例如羧甲基纤维素、海藻酸盐、明胶、聚乙烯吡咯啶酮、蔗糖和/或阿拉伯胶;(3)保湿剂,例如甘油;(4)崩解剂,例如琼脂、碳酸钙、马铃薯或木薯淀粉、海藻酸、某些硅酸盐及碳酸钠;(5)溶解阻滞剂,例如石蜡;(6)吸收加速剂,例如四级铵化合物;(7)湿润剂,例如十六醇及甘油单硬脂酸酯;(8)吸收剂,例如高岭土及膨润土;(9)润滑剂,例如滑石、硬脂酸钙、硬脂酸镁、固体聚乙二醇、月桂基硫酸钠及其混合物;和(10)着色剂。液体剂型可包括药学上可接受的乳液、微乳液、溶液、悬浮液、糖浆及酏剂。除活性成分之外,液体剂型可含有通常用于本技术领域中的惰性稀释剂,例如水或其它溶剂;增溶剂及乳化剂,例如乙醇、异丙醇、碳酸乙酯、乙酸乙脂、苯甲醇、苯甲酸苯甲酯、丙二醇、1,3-丁二醇、油(特别是棉籽油、花生油、玉米油、胚芽油、橄榄油、蓖麻油、及芝麻油)、甘油、四氢呋喃甲醇、聚乙二醇以及脱水山梨醇的脂肪酸酯、及其混合物。
除活性化合物之外,悬浮液也可含有悬浮剂,例如乙氧基化异硬脂醇、聚氧化乙烯山梨糖醇及脱水山梨醇酯、微晶纤维素、氢氧化铝氧化物、膨润土、琼脂及黄蓍胶及其混合物。
除活性化合物之外,软膏剂、糊剂、乳膏剂以及凝胶剂也可含有赋形剂,例如动物脂肪及植物脂肪、油、蜡、石蜡、淀粉、黄蓍胶、纤维素衍生物、聚乙二醇、聚硅氧、膨润土、硅酸、滑石及氧化锌或者其混合物。
除活性化合物之外,粉末及喷雾剂也可以含有赋形剂,例如乳糖、滑石、硅酸、氢氧化铝、硅酸钙及聚酰胺粉末或者上述这些物质的混合物。所述喷雾剂可以含有其它的常用推进剂,例如氯氟烃、以及挥发性的未被取代的烃,例如丁烷及丙烷。
发明的详细说明
除非有相反陈述,否则本发明在说明书和权利要求书中所使用的部分术语定义如下:
“键”是指标示的取代基不存在,该取代基的两端部分直接连接成键。
“烷基”当作一基团或一基团的一部分时是指包括C
1-C
20直链或者带有支链的脂肪烃基团。 优选为C
1-C
10烷基,更优选为C
1-C
6烷基。烷基基团的实施例包括但不限于甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基、1,1-二甲基丙基、1,2-二甲基丙基、2,2-二甲基丙基、1-乙基丙基、2-甲基丁基、3-甲基丁基、正己基、1-乙基-2-甲基丙基、1,1,2-三甲基丙基、1,1-二甲基丁基、1,2-二甲基丁基、2,2-二甲基丁基、1,3-二甲基丁基、2-乙基丁基、2-甲基戊基、3-甲基戊基、4-甲基戊基、2,3-二甲基丁基等。烷基可以是取代或未取代的。
“烯基”指由至少两个碳原子和至少一个碳-碳双键组成的如上定义的烷基,代表性实例包括但不限于乙烯基、1-丙烯基、2-丙烯基、1-,2-或3-丁烯基等。烯基可以是任选取代的或未取代的。
“炔基”是指含有一个碳碳三键的脂肪烃基团,可为直链也可以带有支链。优先选择的是C
2-C
10的炔基,更优选C
2-C
6炔基,最优选C
2-C
4炔基。炔基基团的实施例包括,但不限于乙炔基、1-丙炔基、2-丙炔基、1-、2-或3-丁炔基等。炔基可以是取代或未取代的。
“环烷基”是指饱和或部分饱和的单环、稠环、桥环和螺环的碳环。优选为C
3-C
12环烷基,更优选为C
3-C
8环烷基,最优选为C
3-C
6环烷基。单环环烷基的实施例包括但不限于环丙基、环丁基、环戊基、环戊烯基、环己基、环己烯基、环己二烯基、环庚基、环庚三烯基、环辛基等,优选环丙基、环己烯基。环烷基可以是任选取代的或未取代的。
“螺环烷基”指5至18元,两个或两个以上环状结构,且单环之间彼此共用一个碳原子(称螺原子)的多环基团,环内含有1个或多个双键,但没有一个环具有完全共轭的π电子的芳香系统。优选为6至14元,更优选为7至10元。根据环与环之间共用螺原子的数目将螺环烷基分为单螺、双螺或多螺环烷基,优选为单螺和双螺环烷基,优选为4元/5元、4元/6元、5元/5元或5元/6元。“螺环烷基”的非限制性实施例包括但不限于:螺[4.5]癸基、螺[4.4]壬基、螺[3.5]壬基、螺[2.4]庚基。
“稠环烷基”指5至18元,含有两个或两个以上环状结构彼此共用一对碳原子的全碳多环基团,一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子的芳香系统,优选为6至12元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环稠环烷基,优选为双环或三环,更优选为5元/5元或5元/6元双环烷基。“稠环烷基”的非限制性实施例包括但不限于:二环[3.1.0]己基、二环[3.2.0]庚-1-烯基、二环[3.2.0]庚基、十氢化萘基或十四氢菲基。
“桥环烷基”指5至18元,含有两个或两个以上环状结构,彼此共用两个不直接相连接碳原子的全碳多环基团,一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子的芳香系统,优选为6至12元,更优选为7至10元。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环桥环烷基,优选为双环、三环或四环,更有选为双环或三环。“桥环烷基”的非限制性实施例包括但不限于:(1s,4s)-二环[2.2.1]庚基、二环[3.2.1]辛基、(1s,5s)-二环[3.3.1]壬基、二环[2.2.2]辛基、(1r,5r)-二环[3.3.2]癸基。
“杂环基”、“杂环”或“杂环的”在本申请中可交换使用,都是指非芳香性杂环基,其中一个 或多个成环的原子是杂原子,如氧、氮、硫原子等,包括单环、稠环、桥环和螺环。优选具有5至7元单环或7至10元双或三环,其可以包含1,2或3个选自氮、氧和/或硫中的原子。“杂环基”的实例包括但不限于吗啉基,氧杂环丁烷基,硫代吗啉基,四氢吡喃基,1,1-二氧代硫代吗啉基,哌啶基,2-氧代哌啶基,吡咯烷基,2-氧代吡咯烷基,哌嗪-2-酮,8-氧杂-3-氮杂-双环[3.2.1]辛基和哌嗪基。杂环基可以是取代或未取代的。
“螺杂环基”指5至18元,两个或两个以上环状结构,且单环之间彼此共用一个原子的多环基团,环内含有1个或多个双键,但没有一个环具有完全共轭的π电子的芳香系统,其中一个或多个环原子选自氮、氧或S(O)
r(其中r选自0、1或2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据环与环之间共用螺原子的数目将螺环烷基分为单螺杂环基、双螺杂环基或多螺杂环基,优选为单螺杂环基和双螺杂环基。更优选为4元/4元、4元/5元、4元/6元、5元/5元或5元/6元单螺杂环基。“螺杂环基”的非限制性实施例包括但不限于:1,7-二氧杂螺[4.5]癸基、2-氧杂-7-氮杂螺[4.4]壬基、7-氧杂螺[3.5]壬基和5-氧杂螺[2.4]庚基。
“稠杂环基”指含有两个或两个以上环状结构彼此共用一对原子的全碳多环基团,一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子的芳香系统,其中一个或多个环原子选自氮、氧或S(O)
r(其中r选自0、1或2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环稠杂环基,优选为双环或三环,更优选为5元/5元或5元/6元双环稠杂环基。“稠杂环基”的非限制性实施例包括但不限于:八氢吡咯并[3,4-c]吡咯基、八氢-1H-异吲哚基,3-氮杂二环[3.1.0]己基,八氢苯并[b][1,4]二噁英(dioxine)或
“桥杂环基”指5至14元,5至18元,含有两个或两个以上环状结构,彼此共用两个不直接相连接的原子的多环基团,一个或多个环可以含有一个或多个双键,但没有一个环具有完全共轭的π电子的芳香系统,其中一个或多个环原子选自氮、氧或S(O)
r(其中r选自0、1或2)的杂原子,其余环原子为碳。优选为6至14元,更优选为7至10元。根据组成环的数目可以分为双环、三环、四环或多环桥杂环基,优选为双环、三环或四环,更有选为双环或三环。“桥杂环基”的非限制性实施例包括但不限于:2-氮杂二环[2.2.1]庚基,2-氮杂二环[2.2.2]辛基和2-氮杂二环[3.3.2]癸基。
“芳基”是指含有一个或者两个环的碳环芳香系统,其中所述环可以以稠合的方式连接在一起。术语“芳基”包括单环或双环的芳基,比如苯基、萘基、四氢萘基的芳香基团。优选芳基为C
6-C
10芳基,更优选芳基为苯基和萘基,最优选为萘基。芳基可以是取代或未取代的。
“杂芳基”是指芳香族5至6元单环或8至10元双环,其可以包含1至4个选自氮、氧和/或硫中的原子。优选为双环杂芳基,“杂芳基”的实施例包括但不限于呋喃基,吡啶基,2-氧代-1,2-二氢吡啶基,哒嗪基,嘧啶基,吡嗪基,噻吩基,异噁唑基,噁唑基,噁二唑基,咪唑基,吡咯基,吡唑基,三唑基,四氮唑基,噻唑基,异噻唑基,1,2,3-噻二唑基,苯并间二氧 杂环戊烯基,苯并噻吩基、苯并咪唑基,吲哚基,异吲哚基,1,3-二氧代-异吲哚基,喹啉基,吲唑基,苯并异噻唑基,苯并噁唑基、苯并异噁唑基、苯并噻唑基。
杂芳基可以是取代或未取代的。
“稠合环”是指两个或两个以上环状结构彼此共用一对原子的多环基团,一个或多个环可以含有一个或多个双键,但至少一个环不具有完全共轭的π电子的芳香系统,其中环原子选自0个、一个或多个选自氮、氧或S(O)
r(其中r选自0、1或2)的杂原子,其余环原子为碳。稠合环优选包括双环或三环的稠合环,其中双环稠合环优选为芳基或杂芳基与单环杂环基或单环环烷基的稠合环。优选为7至14元,更优选为8至10元。“稠合环”的实施例包括但不限于:
“烷氧基”是指(烷基-O-)的基团。其中,烷基见本文有关定义。C
1-C
6的烷氧基为优先选择。其实例包括,但不限于:甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、异丁氧基、叔丁氧基等。
“卤代烷基”是指烷基任选进一步被一个或多个卤素所取代的基团,其中烷基见本文有关定义。
“羟烷基”是指烷基任选进一步被一个或多个羟基所取代的基团,其中烷基见本文有关定义。
“羟甲基”指甲基任选进一步被一个或多个羟基所取代的基团。
“卤代烷氧基”是指(烷基-O-)的烷基任选进一步被一个或多个卤素所取代的基团,其中烷氧基见本文有关定义。
“羟基”指-OH基团。
“卤素”是指氟、氯、溴和碘。
“氨基”指-NH
2。
“氰基”指-CN。
“硝基”指-NO
2。
“苄基”指-CH
2-苯基。
“羧基”指-C(O)OH。
“羧酸酯基”指-C(O)O-烷基或-C(O)O-环烷基,其中烷基、环烷基的定义如上所述。
“DMSO”指二甲基亚砜。
“BOC”指叔丁氧基羰基。
“Ts”指对甲苯磺酰基。
“T3P”指丙基磷酸酐。
“DPPA”指叠氮磷酸二苯酯。
“DEA”指二乙胺。
“X-PHOS Pd G2”指氯(2-二环己基膦基-2',4',6'-三异丙基-1,1'-联苯基)[2-(2'-氨基-1,1'-联苯)]钯(II)。
“RuPhos Pd G3”指甲磺酸(2-二环己基膦基-2',6'-二异丙氧基-1,1'-联苯基)(2-氨基-1,1'-联苯-2-基)钯(II)。
“cataCXium A Pd-G3”指甲磺酸[正丁基二(1-金刚烷基)膦](2-氨基-1,1'-联苯-2-基)钯。
“Pd(dppf)Cl
2”指[1,1'-双(二苯基膦基)二茂铁]二氯化钯。
“取代的”指基团中的一个或多个氢原子,优选为最多5个,更优选为1~3个氢原子彼此独立地被相应数目的取代基取代。不言而喻,取代基仅处在它们的可能的化学位置,本领域技术人员能够在不付出过多努力的情况下确定(通过实验或理论)可能或不可能的取代。例如,具有游离氢的氨基或羟基与具有不饱和(如烯属)键的碳原子结合时可能是不稳定的。
本说明书所述的“取代”或“取代的”,如无特别指出,均是指基团可被一个或多个选自以下的基团取代:烷基、烯基、炔基、烷氧基、烷硫基、烷基氨基、卤素、疏基、羟基、硝基、氰基、环烷基、杂环基、芳基、杂芳基、环烷氧基、杂环烷氧基、环烷硫基、杂环烷硫基、氨基、卤代烷基、羟烷基、羧基、羧酸酯基、=O、-C(O)R
6、-C(O)OR
6、-NHC(O)R
6、-NHC(O)OR
6、-NR
7R
8、-C(O)NR
7R
8、-CH
2NHC(O)OR
6、-CH
2NR
7R
8或-S(O)
rR
6的取代基所取代;
R
6选自氢原子、烷基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、卤代烷基、卤代烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R
9、-C(O)OR
9、-OC(O)R
9、-NR
10R
11、-C(O)NR
10R
11、-SO
2NR
10R
11或-NR
10C(O)R
11的取代基所取代;
R
7和R
8各自独立地选自氢原子、羟基、卤素、烷基、烷氧基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、烷氧基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R
9、-C(O)OR
9、-OC(O)R
9、-NR
10R
11、-C(O)NR
10R
11、-SO
2NR
10R
11或-NR
10C(O)R
11的取代基所取代;
或者,R
7和R
8与它们相连接的原子一起形成一个4~8元杂环基,其中4~8元杂环基内含有一个或多个N、O或S(O)
r,并且所述的4~8元杂环基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R
9、-C(O)OR
9、-OC(O)R
9、-NR
10R
11、-C(O)NR
10R
11、-SO
2NR
10R
11或-NR
10C(O)R
11的取代基所取代;
R
9、R
10和R
11各自独立地选自氢原子、烷基、氨基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氨基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、羧基或羧酸酯基的取代基所取代;
r为0、1或2。
本发明化合物可以含有不对称中心或手性中心,因此以不同的立体异构体形式存在。所 预期的是,本发明化合物的所有立体异构体形式,包括但不限于非对映异构体、对映异构体和阻转异构体(atropisomer)和几何(构象)异构体及它们的混合物,如外消旋体混合物,均在本发明的范围内。
除非另外指出,本发明描述的结构还包括此结构的所有异构体(如,非对映异构体、对映异构体和阻转异构体和几何(构象)异构体形式;例如,各不对称中心的R和S构型,(Z)和(E)双键异构体,以及(Z)和(E)构象异构体。因此本发明化合物的单个立体异构体以及对映体混合物、非对映异构体混合物和几何(构象)异构体混合物均在本发明范围内。
“可药用的盐”是指上述化合物能保持原有生物活性并且适合于医药用途的某些盐类。式(I)所表示的化合物的可药用的盐可以为金属盐、与合适的酸形成的胺盐。
“药物组合物”表示含有一种或多种本文所述化合物或其生理学上可药用的盐或前体药物与其他化学组分的混合物,以及其他组分例如生理学可药用的载体和赋形剂。药物组合物的目的是促进对生物体的给药,利于活性成分的吸收进而发挥生物活性。
以下结合实施例用于进一步描述本发明,但这些实施例并非限制着本发明的范围。
实施例
实施例给出了式(I)所表示的代表性化合物的制备及相关结构鉴定数据。必须说明,下述实施例是用于说明本发明而不是对本发明的限制。
1HNMR图谱是用Bruker仪器(400MHz)测定而得,化学位移用ppm表示。使用四甲基硅烷内标准(0.00ppm)。
1HNMR的表示方法:s=单峰,d=双重峰,t=三重峰,m=多重峰,br=变宽的,dd=双重峰的双重峰,dt=三重峰的双重峰。若提供偶合常数时,其单位为Hz。
质谱是用LC/MS仪测定得到,离子化方式可为ESI或APCI。
薄层层析硅胶板使用烟台黄海HSGF254或青岛GF254硅胶板,薄层色谱法(TLC)使用的硅胶板采用的规格是0.15mm~0.2mm,薄层层析分离纯化产品采用的规格是0.4mm~0.5mm。
柱层析一般使用烟台黄海硅胶200~300目硅胶为载体。
在下列实例中,除非另有指明,所有温度为摄氏温度,除非另有指明,各种起始原料和试剂来自市售或者是根据已知的方法合成,市售原料和试剂均不经进一步纯化直接使用,除非另有指明,市售厂家包括但不限于上海皓鸿生物医药科技有限公司,上海韶远试剂有限公司,上海毕得医药科技有限公司,萨恩化学技术(上海)有限公司和上海凌凯医药科技有限公司等。
CD
3OD:氘代甲醇。
CDCl
3:氘代氯仿。
DMSO-d
6:氘代二甲基亚砜。
实施例中无特殊说明,反应中的溶液是指水溶液。
对化合物进行纯化,采用柱层析和薄层色谱法的洗脱剂体系,其中该体系选自:A:石油 醚和乙酸乙酯体系;B:二氯甲烷和甲醇体系;C:二氯甲烷和乙酸乙酯体系,D:二氯甲烷和乙醇体系,其中溶剂的体积比根据化合物的极性不同而不同,也可以加入少量的酸性或碱性试剂进行条件,如醋酸或三乙胺等。
室温:20℃~30℃。
实施例1
N-((R)-1-(3-amino-5-(trifluoromethyl)phenyl)ethyl)-9-methyl-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4,5][1,4]oxazino[3,2-g]quinazolin-11-amine
N-((R)-1-(3-氨基-5-(三氟甲基)苯基)乙基)-9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺
第一步
methyl 8-nitro-1,2,4a,5-tetrahydro-4H-benzo[b][1,4]oxazino[4,3-d][1,4]oxazine-9-carboxylate8-硝基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯
于50mL双口瓶中,依次加入4,5-二氟-2-硝基苯甲酸甲酯1a(1.5g,6.9mmol),吗啡啉-3-基-甲醇盐酸盐(1g,8.5mmol),二甲亚砜(15mL),氢氧化钾(1.5g,27.6mmol),置换氮气,室温下反应2小时,升温至60℃继续反应4小时。将反应液冷却至室温,加入二氯甲烷(80mL),以水(20mL×3),饱和食盐水(20mL×1)洗涤,有机相以无水硫酸钠干燥,过滤,减压 浓缩,得到的残余物用硅胶柱层析(洗脱剂:A体系)分离纯化,得到8-硝基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯1b(396mg,1.9mmol),产率20%。
第二步
methyl 8-amino-1,2,4a,5-tetrahydro-4H-benzo[b][1,4]oxazino[4,3-d][1,4]oxazine-9-carboxylate
8-氨基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯
于50mL双口瓶中,依次加入8-硝基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯1b(396mg,1.9mmol),甲醇(20mL),钯碳(132mg,1.1mmol),置换氢气,氢气氛下室温反应4小时。反应液过滤,滤饼以二氯甲烷和甲醇的混合溶剂(V:V=10:1,10mL×3)洗涤,滤液减压浓缩,得到8-氨基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯1c(355mg,1.5mmol),产率95%。
MS m/z(ESI):265.2[M+1]
+
第三步
methyl 8-acetimidamido-1,2,4a,5-tetrahydro-4H-benzo[b][1,4]oxazino[4,3-d][1,4]oxazine-9-carboxylate
8-乙酰亚胺基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯
于50mL双口瓶中,依次加入8-氨基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯1c(355mg,1.5mmol),乙腈(20mL),盐酸的1,4-二氧六环溶液(4M,6mL),升温至60℃下反应3小时。反应液减压浓缩,得粗品8-乙酰亚胺基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯1d(413mg),直接进行下步反应。
MS m/z(ESI):306.2[M+1]
+
第四步
9-methyl-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4,5][1,4]oxazino[3,2-g]quinazolin-11-ol
9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-醇
于50mL单口瓶中,依次加入粗品8-乙酰亚胺基-1,2,4a,5-四氢-4H-苯并[b][1,4]恶嗪并[4,3-d][1,4]恶嗪-9-羧酸甲酯1d(413mg,1.4mmol),乙醇(20mL),碳酸氢钠(454mg,5.2mmol),升温至80℃反应1小时。反应液降至室温,减压浓缩,加入水(10mL),以二氯甲烷萃取(20mL×3),合并有机相,以无水硫酸钠干燥,过滤,减压浓缩,得到的残余物用硅胶柱层析(洗脱剂:B体系)分离纯化,得到9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-醇1e(256mg,0.94mmol),产率70%。
MS m/z(ESI):274.1[M+1]
+
第五步
9-methyl-N-((R)-1-(3-nitro-5-(trifluoromethyl)phenyl)ethyl)-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4,5][1,4]oxazino[3,2-g]quinazolin-11-amine
9-甲基-N-((R)-1-(3-硝基-5-(三氟甲基)苯基)乙基)-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺
于25mL单口瓶中,依次加入9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并 [3,2-g]喹唑啉-11-醇1e(50mg,0.18mmol),(R)-1-(3-硝基-5-(三氟甲基)苯基)乙-1-胺盐酸盐1f(74mg,0.27mmol,根据公开专利WO 2018115380制备),BOP(161mg,0.36mmol),二甲亚砜(5mL)和DBU(98mg,0.64mmol),室温下搅拌5天。反应液中加入水(5mL),以二氯甲烷萃取(15mL×3),合并有机相,以无水硫酸钠干燥,过滤,减压浓缩,得到的残余物用硅胶柱层析(洗脱剂:A体系)分离纯化,得到9-甲基-N-((R)-1-(3-硝基-5-(三氟甲基)苯基)乙基)-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺1g(56mg,0.11mmol),产率63%。
MS m/z(ESI):490.2[M+1]
+
第六步
N-((R)-1-(3-amino-5-(trifluoromethyl)phenyl)ethyl)-9-methyl-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4,5][1,4]oxazino[3,2-g]quinazolin-11-amine
N-((R)-1-(3-氨基-5-(三氟甲基)苯基)乙基)-9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺
于25mL单口瓶中,依次加入9-甲基-N-((R)-1-(3-硝基-5-(三氟甲基)苯基)乙基)-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺1g(56mg,0.11mmol),乙醇(10mL),铁粉(22.8mg,0.41mmol),浓盐酸(0.5mL),回流反应2小时。反应液降至室温,减压浓缩,所得的残余物中加入二氯甲烷(20mL),用饱和碳酸氢钠溶液调节pH至碱性,过滤除去不溶物,分液,收集有机相,以无水硫酸钠干燥,过滤,减压浓缩,得到的残余物用硅胶柱层析(洗脱剂:B体系)分离纯化,得到N-((R)-1-(3-氨基-5-(三氟甲基)苯基)乙基)-9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺1(32mg,0.07mmol),产率62%。
MS m/z(ESI):460.2[M+1]
+
1H NMR(400MHz,DMSO-d
6)δ8.05(s,1H),7.57(d,J=5.2Hz,1H),6.85-6.83(m,3H),6.69(s,1H),5.58-5.54(m,3H),4.33(d,J=10.7Hz,1H),4.09(d,J=11.5Hz,1H),3.98-3.90(m,2H),3.83(t,J=10.6Hz,1H),3.68(t,J=11.9Hz,1H),3.22-3.19(m,2H),2.84-2.81(m,1H),2.33(s,3H),1.54(d,J=6.9Hz,3H)ppm.
实施例2和实施例3
(R)-N-((R)-1-(3-amino-5-(trifluoromethyl)phenyl)ethyl)-9-methyl-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4,5][1,4]oxazino[3,2-g]quinazolin-11-amine 2
(R)-N-((R)-1-(3-氨基-5-(三氟甲基)苯基)乙基)-9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺2
(S)-N-((R)-1-(3-amino-5-(trifluoromethyl)phenyl)ethyl)-9-methyl-1,2,4a,5-tetrahydro-4H-[1,4]oxazino[4',3':4,5][1,4]oxazino[3,2-g]quinazolin-11-amine 3
(S)-N-((R)-1-(3-氨基-5-(三氟甲基)苯基)乙基)-9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺3
将N-((R)-1-(3-氨基-5-(三氟甲基)苯基)乙基)-9-甲基-1,2,4a,5-四氢-4H-[1,4]恶嗪并[4',3':4,5][1,4]恶嗪并[3,2-g]喹唑啉-11-胺1(98mg)通过SFC手性拆分(柱型号:Superchiral S-OJ(Chiralway),2.1cm I.D.*25cm Length,5um;流动相:hexane/EtOH/DEA=70/30/0.05(v/v/v);流速:15mL/min;检测波长:250nm;柱温:35℃)纯化后,得到单一构型化合物(较短保留时间)和单一构型化合物(较长保留时间)。
单一构型化合物(较短保留时间):
MS m/z(ESI):460.2[M+1]
+
36mg,0.08mmol;保留时间:4.398分钟,手性纯度99%ee。
1H NMR(400MHz,DMSO-d
6)δ7.89(d,J=8.2Hz,1H),7.55(s,1H),6.84(d,J=8.1Hz,2H),6.83(s,1H),6.68(s,1H),5.57-5.53(m,3H),4.32(d,J=10.7Hz,1H),4.08(d,J=11.4Hz,1H),3.96-3.90(m,2H),3.85(d,J=12.4Hz,1H),3.69(t,J=11.4Hz,1H),3.20-3.16(m,2H),2.80(t,J=11.7Hz,1H),2.31(s,3H),1.54(d,J=7.0Hz,3H)ppm.
单一构型化合物(较长保留时间):
MS m/z(ESI):460.2[M+1]
+
48mg,0.10μmol;保留时间:4.838分钟,手性纯度97%ee。
1H NMR(400MHz,DMSO-d
6)δ8.51(s,2H),7.72(s,1H),6.95(s,1H),6.86(d,J=9.4Hz,2H),6.72(s,1H),5.65-5.59(m,3H),4.38(d,J=10.8Hz,1H),4.15-4.05(m,1H),4.00(t,J=9.9Hz,1H),3.93(d,J=10.4Hz,1H),3.85(d,J=12.4Hz,1H),3.67(t,J=11.5Hz,1H),3.20(t,J=10.6Hz,1H),2.91-2.87(s,1H),2.44(s,3H),1.58(d,J=7.0Hz,3H)ppm.
生物学评价
测试例1、本发明化合物阻断SOS1与KRAS G12C蛋白结合的测试
以下方法用于测定本发明化合物在体外条件下阻断SOS1与KRAS G12C蛋白相互作用的能力。本方法使用Cisbio公司的KRAS-G12C/SOS1 BINDING ASSAY KITS试剂盒(货号 63ADK000CB16PEG),详细实验操作可参考试剂盒说明书。
将实验流程简述如下:使用diluent buffer(货号62DLBDDF)配置Tag1-SOS1和Tag2-KRAS-G12C蛋白为5X的工作液浓度备用。受试化合物溶解于DMSO中制备为10mM贮存液,随后使用diluent buffer进行稀释备用。首先向孔中加入2μL受试化合物(反应体系终浓度为10000nM-0.1nM),随后加入4μL Tag1-SOS1 5X的工作液和4μL Tag2-KRAS-G12C 5X的工作液,离心并混匀,静置15分钟;随后加入10μL预混匀的anti-Tag1-Tb
3+和anti-Tag2-XL665,室温下孵育2小时;最后使用酶标仪以TF-FRET模式上测定在304nM的激发波长下,各孔发射波长为620nM和665nM的荧光强度,并计算各孔665/620的荧光强度比值。通过与对照组(0.1%DMSO)的荧光强度比值进行比较,计算受试化合物在各浓度下的百分比抑制率,并通过GraphPad Prism 5软件以受试化合物浓度对数值-抑制率进行非线性回归分析,获得化合物的IC
50值。
表1.本发明化合物对SOS1与KRAS G12C蛋白相互作用抑制的IC
50值
| 化合物编号 | IC 50/(nM) |
| 1 | 41.8 |
结论:从表1可以看出,本发明化合物对SOS1与KRAS G12C蛋白相互作用具有较强阻断作用。
测试例2、本发明化合物对OCI-AML5细胞增殖抑制测定
以下方法用于测定本发明化合物对OCI-AML5细胞增殖的影响。OCI-AML5细胞(含有SOS1N233Y突变)购于南京科佰生物科技有限公司,培养于含10%胎牛血清、100U青霉素和100μg/mL链霉素的MEMα培养基中。细胞活力通过
Luminescent Cell Viability Assay试剂盒(Promega,货号G7573)进行测定。
实验方法按照试剂盒说明书的步骤操作,简述如下:受试化合物首先溶解于DMSO中制备为10mM贮存液,随后以培养基进行稀释,配制成测试样品,化合物的终浓度范围在10000nM-0.15nM。将处于对数生长期的细胞以1000个细胞每孔的密度接种至96孔细胞培养板中,在37℃,5%CO
2培养箱中培养过夜,随后加入受试化合物后继续培养120小时。培养结束后,向每孔加入50uL体积的CellTiter-Glo检测液,震荡5分钟后静置10分钟,随后在酶标仪上使用Luminescence模式读取样品各孔发光值。通过与对照组(0.3%DMSO)的数值进行比较计算化合物在各浓度点的百分比抑制率,之后在GraphPad Prism 5软件中以化合物浓度对数-抑制率进行非线性回归分析,获得化合物抑制细胞增殖的IC
50值,结果见下表2。
表2.本发明化合物对OCI-AML5细胞增殖抑制的IC
50值
| 化合物编号 | IC 50/(nM) |
| 1 | 41.2 |
结论:本发明化合物对OCI-AML5细胞增殖抑制具有较好抑制作用。
测试例3、本发明化合物对DLD-1细胞中p-ERK1/2抑制活性的测定
以下方法用于测定本发明化合物对DLD-1细胞中p-ERK1/2抑制活性。本方法使用Cisbio公司的Advanced phospho-ERK1/2(Thr202/tyr204)试剂盒(货号64AERPEH),详细实验操作可参考试剂盒说明书。DLD-1细胞(含有KRAS G13D突变)购于中国科学院上海生命科学研究院细胞资源中心。
将实验流程简述如下:DLD-1细胞培养于含10%胎牛血清、100U青霉素,100μg/mL链霉素和1mM Sodium Pyruvate的RPMI 1640完全培养基中。DLD-1细胞按每孔30000个铺于96孔板中,培养基为完全培养基,在37℃,5%CO2培养箱内培养过夜。将受试化合物溶解于DMSO中制备为10mM贮存液,随后使用RPMI 1640基础培养基进行稀释,每孔加入90uL含对应浓度受试化合物的RPMI 1640基础培养基,受试化合物在反应体系中的终浓度范围为10000nM-0.15nM,置于细胞培养箱培养3小时40分钟。随后加入10uL用RPMI 1640基础培养基配制的hEGF(购自Roche,货号11376454001),使其终浓度为5nM,置于培养箱培养20分钟。弃去细胞上清,使用冰浴的PBS清洗细胞,之后每孔加入45μl的1×cell phospho/total protein lysis buffer(Advanced phospho-ERK1/2试剂盒组分)进行裂解,96孔板置于冰上裂解半小时,随后参照Advanced phospho-ERK1/2(Thr202/tyr204)试剂盒说明书检测裂解液。最后在酶标仪以TF-FRET模式上测定在304nM的激发波长下,各孔发射波长为620nM和665nM的荧光强度,并计算各孔665/620的荧光强度比值。通过与对照组(0.1%DMSO)的荧光强度比值进行比较,计算受试化合物在各浓度下的百分比抑制率,并通过GraphPad Prism 5软件以受试化合物浓度对数值-抑制率进行非线性回归分析,获得化合物的IC
50值,结果见下表3。
表3.本发明化合物对DLD-1细胞ERK磷酸化抑制的IC
50值
| 化合物编号 | IC 50/(nM) |
| 1 | 44.2 |
| 2和3中保留时间较短的化合物 | 43.8 |
| 2和3中保留时间较长的化合物 | 21.2 |
结论:本发明化合物对DLD-1细胞ERK磷酸化具有较好的抑制作用。
Claims (16)
- 一种通式(I)所示的化合物或其立体异构体、互变异构体或其可药用的盐:其中:W选自N或CH;V选自N、-CR 2;L选自键或-CH 2-;X选自-O-或-N(R 3)-;Y选自-C(O)-或-CH 2-;Z选自-O-或-N(R 5)-;环A选自C 6-C 10芳基、5-10元杂芳基、9-10元双环杂环基或9-10元稠合环;R 1相同或不同,各自独立地选自氢原子、卤素、氰基、氨基、硝基、C 1-C 6烷基、C 3-C 6环烷基、4-11元杂环基、C 6-C 10芳基或5-10元杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自卤素、氰基、羟基、氨基、烷氧基、=O、-C(O)R 6、-C(O)OR 6、-NHC(O)R 6、-NHC(O)OR 6、-NR 7R 8、-C(O)NR 7R 8、-CH 2NR 7R 8、-CH 2NHC(O)OR 6或-SO 2R 6的取代基所取代;R 2选自氟原子、羟基、氰基、甲氧基或氨基;R 3选自氢原子、C 1-C 6烷基、C 3-C 6环烷基或-C(O)R 4;其中所述的烷基或环烷基任选进一步被一个或多个选自卤素、氰基、羟基、氨基、C 1-C 3烷氧基、C 1-C 3羟基烷基、4-11元杂环基所取代;其中所述的4-11元杂环基任选进一步被C 1-C 6烷基、C 3-C 6环烷基、烷氧基、=O、-C(O)R 6、-C(O)OR 6、-NHC(O)R 6、-NHC(O)OR 6、-NR 7R 8、-C(O)NR 7R 8、-CH 2NR 7R 8、-CH 2NHC(O)OR 6或-SO 2R 6的取代基所取代;R 4选自C 1-C 6烷基、C 3-C 6环烷基或4-10元杂环基;其中所述的烷基、环烷基或杂环基任选进一步被一个或多个R A取代;R A各自独立地选自卤素、氰基、羟基、氨基、C 1-C 3烷氧基、C 1-C 3羟基烷基、C 1-C 3卤代烷基或4-11元杂环基;其中所述的4-11元杂环基任选进一步被C 1-C 6烷基、C 3-C 6环烷基、烷氧基、=O、-C(O)R 6、-C(O)OR 6、-NHC(O)R 6、-NHC(O)OR 6、-NR 7R 8、-C(O)NR 7R 8、-CH 2NR 7R 8、-CH 2NHC(O)OR 6或-SO 2R 6的取代基所取代;R 5选自于氢原子、C 1-C 6烷基或C 3-C 6环烷基;其中所述的烷基或环烷基任选进一步被一个或多个选自卤素、氰基、羟基、氨基、C 1-C 3烷氧基或C 1-C 3羟基烷基的取代基所取代;R 6各自独立地选自氢原子、烷基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、卤代烷基、卤代烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R 9、-C(O)OR 9、-OC(O)R 9、-NR 10R 11、-C(O)NR 10R 11、-SO 2NR 10R 11或-NR 10C(O)R 11的取代基所取代;R 7和R 8各自独立地选自氢原子、羟基、卤素、烷基、烷氧基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、烷氧基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R 9、-C(O)OR 9、-OC(O)R 9、-NR 10R 11、-C(O)NR 10R 11、-SO 2NR 10R 11或-NR 10C(O)R 11的取代基所取代;或者,R 7和R 8与它们相连接的原子一起形成一个4~8元杂环基,其中4~8元杂环基内含有一个或多个N、O、S或SO 2,并且所述的4~8元杂环基任选进一步被一个或多个选自羟基、卤素、硝基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、=O、-C(O)R 9、-C(O)OR 9、-OC(O)R 9、-NR 10R 11、-C(O)NR 10R 11、-SO 2NR 10R 11或-NR 10C(O)R 11的取代基所取代;R 9、R 10和R 11各自独立地选自氢原子、烷基、氨基、环烷基、杂环基、芳基或杂芳基,其中所述的烷基、环烷基、杂环基、芳基或杂芳基任选进一步被一个或多个选自羟基、卤素、硝基、氨基、氰基、烷基、烷氧基、环烷基、杂环基、芳基、杂芳基、羧基或羧酸酯基的取代基所取代;m为1、2或3。
- 根据权利要求1-6任一项所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中X为-O-。
- 根据权利要求1-6任一项所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中:X选自-N(R 3)-;R 3选自-C(O)R 4;R 4的定义如权利要求1中所述。
- 根据权利要求9所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中R 4选自甲基或环丙基。
- 根据权利要求4或5所述的化合物或其立体异构体、互变异构体或其可药用的盐,其中R 5为甲基。
- 一种药物组合物,所述的药物组合物含有有效剂量的根据权利要求1~12中任何一项所述的化合物或其立体异构体、互变异构体或其可药用的盐,及可药用的载体、赋形剂或它们的组合物。
- 根据权利要求1~12中任何一项所述的化合物或其立体异构体、互变异构体或其可药用的盐,或根据权利要求13所述的药物组合物在制备SOS1抑制剂中的用途。
- 根据权利要求1~12中任何一项所述的化合物或其立体异构体、互变异构体或其可药用的盐,或根据权利要求13所述的药物组合物在制备治疗由SOS1介导的疾病的药物中的用途,其中所述的由SOS1介导的疾病优选为RAS家族蛋白信号传导通路依赖性相关的癌症、SOS1突变导致的癌症或SOS1突变导致的遗传性疾病。
- 根据权利要求15所述的用途,其中所述的由SOS1介导的疾病选自肺癌、胰腺癌、结肠癌、膀胱癌、前列腺癌、胆管癌、胃癌、弥漫性大B细胞淋巴瘤、神经纤维瘤、努南综合征、心面皮肤综合征、Ⅰ型遗传性齿龈纤维瘤、胚胎性横纹肌肉瘤、塞尔托利细胞睾丸瘤或皮肤颗粒细胞瘤。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202280049444.7A CN117769556A (zh) | 2021-07-23 | 2022-07-21 | 嘧啶并环类衍生物及其制备方法和用途 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110836112 | 2021-07-23 | ||
| CN202110836112.0 | 2021-07-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023001229A1 true WO2023001229A1 (zh) | 2023-01-26 |
Family
ID=84979705
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2022/107001 Ceased WO2023001229A1 (zh) | 2021-07-23 | 2022-07-21 | 嘧啶并环类衍生物及其制备方法和用途 |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN117769556A (zh) |
| WO (1) | WO2023001229A1 (zh) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114685531A (zh) * | 2020-12-25 | 2022-07-01 | 武汉誉祥医药科技有限公司 | 四并环化合物及其药物组合物和应用 |
| WO2023165438A1 (zh) * | 2022-03-03 | 2023-09-07 | 浙江海正药业股份有限公司 | 三环类衍生物及其制备方法和用途 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110167928A (zh) * | 2016-12-22 | 2019-08-23 | 勃林格殷格翰国际有限公司 | 作为sos1抑制剂的新型经苄基氨基取代的喹唑啉和衍生物 |
| WO2022135610A1 (zh) * | 2020-12-25 | 2022-06-30 | 武汉誉祥医药科技有限公司 | 四并环化合物及其药物组合物和应用 |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN115490699B (zh) * | 2021-06-19 | 2024-08-09 | 成都海博为药业有限公司 | 一种稠环化合物及其药物组合物和用途 |
-
2022
- 2022-07-21 WO PCT/CN2022/107001 patent/WO2023001229A1/zh not_active Ceased
- 2022-07-21 CN CN202280049444.7A patent/CN117769556A/zh active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110167928A (zh) * | 2016-12-22 | 2019-08-23 | 勃林格殷格翰国际有限公司 | 作为sos1抑制剂的新型经苄基氨基取代的喹唑啉和衍生物 |
| WO2022135610A1 (zh) * | 2020-12-25 | 2022-06-30 | 武汉誉祥医药科技有限公司 | 四并环化合物及其药物组合物和应用 |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114685531A (zh) * | 2020-12-25 | 2022-07-01 | 武汉誉祥医药科技有限公司 | 四并环化合物及其药物组合物和应用 |
| CN114685531B (zh) * | 2020-12-25 | 2024-10-22 | 武汉誉祥医药科技有限公司 | 四并环化合物及其药物组合物和应用 |
| WO2023165438A1 (zh) * | 2022-03-03 | 2023-09-07 | 浙江海正药业股份有限公司 | 三环类衍生物及其制备方法和用途 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN117769556A (zh) | 2024-03-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN116113632B (zh) | 杂环类衍生物、其制备方法及其医药上的用途 | |
| CN116249683B (zh) | 氘甲基取代吡嗪并吡嗪并喹啉酮类衍生物、其制备方法及其在医药上的应用 | |
| WO2023025116A1 (zh) | 杂环类衍生物、其制备方法及其医药上的用途 | |
| CN116162099A (zh) | 杂环类衍生物及其制备方法和用途 | |
| CN116514846A (zh) | 杂环类衍生物、其制备方法及其医药上的用途 | |
| CN116332938A (zh) | 稠合三环类衍生物及其制备方法和用途 | |
| WO2022007841A1 (zh) | 一种egfr抑制剂、其制备方法和在药学上的应用 | |
| CN115403575A (zh) | 杂芳环类衍生物及其制备方法和用途 | |
| CN115557949A (zh) | 四环类衍生物、其制备方法及其在医药上的应用 | |
| CN115304602A (zh) | 吡嗪并吡嗪并萘啶二酮类衍生物、其制备方法及其医药上的用途 | |
| CN117466917A (zh) | 杂环类衍生物、其制备方法及其医药上的用途 | |
| CN116390923B (zh) | 杂环类衍生物及其制备方法和用途 | |
| EA027533B1 (ru) | Конденсированные тетра- или пентациклические дигидродиазепинокарбазолоны в качестве ингибиторов parps | |
| CN116113416B (zh) | 四环类衍生物、其制备方法和其医药上的用途 | |
| CN116157401B (zh) | 杂环类衍生物及其制备方法和用途 | |
| JP6731917B2 (ja) | Dpp−4阻害剤としてのベンゼン縮合6員環誘導体およびその使用 | |
| JP2020527173A (ja) | ヒストン脱アセチル化酵素1および/または2(hdac1−2)の選択的阻害剤としての新規なヘテロアリールアミド誘導体 | |
| CN116157400B (zh) | 杂环类衍生物及其制备方法和用途 | |
| KR20240021239A (ko) | Cdk 키나아제 억제제로 사용되는 화합물 및 이의 용도 | |
| WO2023001229A1 (zh) | 嘧啶并环类衍生物及其制备方法和用途 | |
| WO2014090398A1 (en) | Use of maleimide derivatives for preventing and treating leukemia | |
| WO2022198905A1 (zh) | 三环类衍生物及其制备方法和用途 | |
| CN111499613B (zh) | N-甲酰胺衍生物、其制备方法及其在医药上的用途 | |
| CN113166148B (zh) | 作为cdk-hdac双通路抑制剂的杂环化合物 | |
| CN113527298B (zh) | 大环内酰胺类衍生物、及其制备方法和用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22845393 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280049444.7 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22845393 Country of ref document: EP Kind code of ref document: A1 |