WO2022226539A1 - Methods of administering antibodies against sars-cov-2 spike protein - Google Patents
Methods of administering antibodies against sars-cov-2 spike protein Download PDFInfo
- Publication number
- WO2022226539A1 WO2022226539A1 PCT/US2022/071875 US2022071875W WO2022226539A1 WO 2022226539 A1 WO2022226539 A1 WO 2022226539A1 US 2022071875 W US2022071875 W US 2022071875W WO 2022226539 A1 WO2022226539 A1 WO 2022226539A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- amino acid
- acid sequence
- antibody
- antigen binding
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C07K16/104—
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
- A61K2039/507—Comprising a combination of two or more separate antibodies
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/54—Medicinal preparations containing antigens or antibodies characterised by the route of administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/545—Medicinal preparations containing antigens or antibodies characterised by the dose, timing or administration schedule
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/33—Crossreactivity, e.g. for species or epitope, or lack of said crossreactivity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Definitions
- SARS-CoV-2 severe acute respiratory syndrome coronavirus 2
- SARS-CoV-2 genomic RNA contains a large viral replicase gene, genes encoding non-structural proteins at its 5’ end, and a region encoding four major structural and multiple accessory proteins at the 3’ end.
- Structural proteins include Spike or Surface glycoprotein (S), Membrane protein (M), Envelope protein (E) and Nucleocapsid protein (N) [Fehr & Perlman 2015].
- the membrane surface glycoprotein S consists of two subunits, S1 and S2, that mediate viral binding to the host receptor ACE2 and fusion with the host cell membrane, respectively.
- the S1 subunit contains the receptor binding domain (RBD) that directly interacts with ACE2 and is a target of multiple neutralizing antibodies currently in clinical trials [Lan et al. 2020, Robbiani et al. 2020].
- RBD receptor binding domain
- the vast majority of the ongoing efforts are all targeting the S protein.
- Both passive (therapeutic antibodies) and active (vaccine) approaches directed at S protein are expected to promote virus neutralization, that is, inhibition of viral entry into healthy cells.
- Variants of Interests which are defined as having specific genetic markers that are predicted to affect transmission, diagnostics, therapeutics, or immune escape. Data also demonstrates these variants of interest were the cause of an increased proportion of cases or outbreak, but they are of limited prevalence in the US or other countries (https://www.cdc.gov/coronavirus/2019-ncov/cases- updates/variant-surveillance/variant-info.html#Concern). [0007] Studies are described herein that elucidate the memory B cell antibody response in convalescent patients, using an approach that enables the generation of large, stable hybridoma libraries from primary human B cells.
- Hybridoma libraries were generated from the memory B cells of convalescent COVID-19 blood donors who were eligible to donate convalescent plasma based upon their high titer of IgG antibodies against the SARS-CoV-2 virus. Monoclonal antibodies derived from those libraries were selected on the basis of their selective binding to one of multiple SARS-CoV-2 proteins used as targets in both cell-based and soluble protein-based screens. Characterization of these antibodies revealed broad responses to diverse viral antigens. Fewer than half of the antibodies were directed at S protein, while the remainder were directed at other viral proteins including N and ORF-encoded proteins.
- anti-Spike antibodies were directed at highly diverse SARS-CoV-2 antigens, they were generally characterized as having variable levels of somatic hypermutation (SHM) and a diversity of VH and VL gene usage.
- Functional properties of anti-Spike antibodies were successfully confirmed against reference strains (e.g., USA/WA_CDC-WA1/2020), as well as multiple variants including the CDC variants of concern, in series of tests ranging from in vitro neutralization of both pseudovirus and live virus isolates to in vivo neutralization activity in a hamster model of COVID-19.
- Three anti-Spike antibodies were identified that when mixed together in a cocktail exhibited combinatorial effects against those variants.
- kits for treating a SARS-CoV-2 infection in an immunocmpromised subject comprising administering to the subject at least first and send antibodies or antigen binding fragmetns thereof that specifically bind to distinct epitopes of a Spike protein a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
- the antibodies disclosed herein are especially effective for treating immunocompromised individuals due to synergistic effects and their ability to fix complement.
- the antibodies are able to effectively fix complement due to their specific binding sites on the Spike protein and orientation of binding.
- the methods provided herein are effective for viral clearance in immunocompromised patients.
- the methods provided herein are effective for treatment of individual who have decreased adaptive immune system function.
- Also provided herein are methods of treating a SARS-CoV-2 infection in a subject comprising administering about 200 mg, about 400 mg, about 600 mg, or about 800 mg of one, two, three, or four antibodies or antigen binding fragments provided herein.
- FIG. 1 depicts the breadth of antibodies isolated against a range of SARS-CoV-2 viral proteins.
- FIG. 1 depicts the breadth of antibodies isolated against a range of SARS-CoV-2 viral proteins.
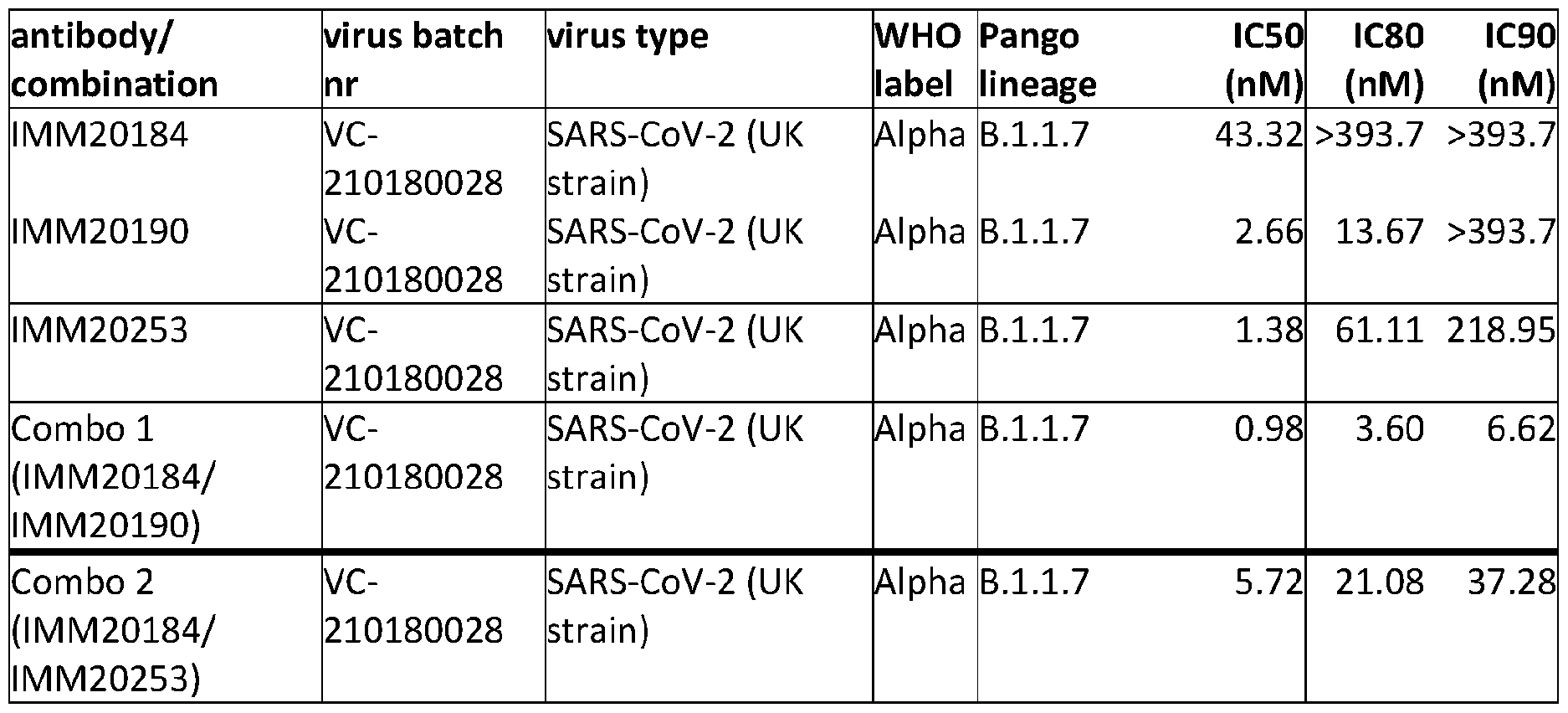
- FIGS. 3A-3D depict in vitro pseudovirus neutralization activity of PR193_00018 (IMM20184), PR194_00232 (IMM20190), PR200_00622 (IMM20253), and PR190_00255 (IMM20279) against pseudoviruses expressing four different variations of Spike.
- FIGS. 3A-3D depict in vitro pseudovirus neutralization activity of PR193_00018 (IMM20184), PR194_00232 (IMM20190), PR200_00622 (IMM20253), and PR190_00255 (IMM20279) against pseudoviruses expressing four different variations of Spike.
- FIGS. 5A-5C depict in vitro neutralization activity of identified anti-Spike antibodies, as noted, against pseudovirus expressing the Spike protein from either the U.K.
- FIGS. 6A-6C depict the concentration-dependent ability of the triple antibody cocktail (IMM20184/IMM20190/IMM20253) and the two antibody cocktail (IMM20184/IMM20253) to neutralize pseudoviruses expressing the reference (WA1/2020), alpha/UK, beta/S. African, gamma/Brazilian, or Epsilon/Californian Spike proteins.
- FIGS. 7A-7C depict the combinatorial activity of the IMM20184 / IMM20190 / IMM20253 triple combination against pseudoviruses expressing the Spike protein from the reference strain (WA_CDC-WA1/2020), U.K.
- FIG. 8 depicts the crystal structure of the SARS-CoV-2 RBD (RCSB PDB: 7A97) represented in cartoon with the epitopes of IMM20184, IMM20190, and IMM20253 depicted in black spheres.
- FIG. 10 depicts in vitro neutralization activity triple antibody cocktail (IMM20184 / IMM20190 / IMM20253) against pseudovirus expressing either the WA1/2020 (REF), B1.617(L452R, E484Q), B.1.617.2 (delta), or lambda (C.37) Spike proteins.
- FIG. 10 depicts in vitro neutralization activity triple antibody cocktail (IMM20184 / IMM20190 / IMM20253) against pseudovirus expressing the Spike protein from the reference strain (WA_CDC-WA1/2020), D614G, B.1.1.7 (alpha/U.K.), B.1.351 (beta/S.
- FIGS. 11A-11D depict in vitro neutralization activity of the triple (IMM20184 / IMM20190 / IMM20253) and double (IMM20184 / IMM20253) combinations of antibodies against three live virus strains as measured by plaque forming assays; the reference strain (WA_CDC-WA1/2020)(FIG. 11A), U.K.
- FIG. 12 depicts lung titer per gram tissue, as measured by plaque forming units, as an assessment of in vivo activity of the identified anti-Spike antibody PR194_00232, alone and in combination with either PR193_00018 or PR200_00622, against the live virus isolate SAR-CoV-2/human/USA/WA_CDC-WA1/2020, when dosed in the prophylactic setting.
- FIG. 12 depicts lung titer per gram tissue, as measured by plaque forming units, as an assessment of in vivo activity of the identified anti-Spike antibody PR194_00232, alone and in combination with either PR193_00018 or PR200_00622, against the live virus isolate SAR-CoV-2/human/USA/WA_CDC-WA1/2020, when dosed in the prophylactic setting.
- FIG. 13 depicts lung titer per gram tissue, as measured by plaque forming units, as an assessment of in vivo activity of various combinations of anti-Spike antibodies, dosed in a therapeutic setting, in the hamster model of COVID-19. Error bars represent the median +/- 95% CI.
- FIG. 14 depicts lung titer per gram tissue, measured by TCID50 assays, as an assessment of in vivo activity of the triple combination ofIMM20184/IMM20190/IMM20253, at two different ratios, dosed in a therapeutic setting, in hamster model of COVID-19. Error bars represent the median +/- interquartile range.
- FIG. 14 depicts lung titer per gram tissue, measured by TCID50 assays, as an assessment of in vivo activity of the triple combination ofIMM20184/IMM20190/IMM20253, at two different ratios, dosed in a therapeutic setting, in hamster model of COVID-19
- FIGS. 16A-16B depict lung titer per gram of tissue, as measured by plaque forming units. Hamsters infected with either the (FIG. 16A) WA1/2020 or (FIG. 16B) Beta isolate were treated in prophylactic setting with
- FIGS. 18A-18C depict the ability of IMM20184, IMM20190, and IMM20253 to bind to the isolated RBD and intact trimer of SARS-CoV-2 reference strain (WA1/2020) as measured by surface plasmon resonance.
- FIGS. 18A-18C depict the ability of IMM20184, IMM20190, and IMM20253 to bind to the isolated RBD and intact trimer of SARS-CoV-2 reference strain (WA1/2020) as measured by surface plasmon resonance.
- FIGS. 20A-20C depict in vitro complement fixation activity of PR193_00018 (IMM20184), PR194_00190 (IMM20190), PR200_00253 (IMM20253), the two antibody cocktail of IMM20184/IMM20253, and the triple antibody cocktail (IMM20184/IMM20190/IMM20253) relative to control antibodies when assessed at defined (FIG. 20A-20B) concentrations and in a concentration-dependent (FIG. 20C) manners.
- FIG. 21 depicts in vitro phagocytosis activity of IMM20184, IMM20190, IMM20253, the IMM20184/IMM20253 two -antibody cocktail, and IMM-BCP-01 (three antibody cocktail) relative to isotype control antibodies when assessed across a range of concentrations.
- FIG. 22 depicts antibody-dependent cellular cytotoxicity activity of IMM20184, IMM20190, IMM20253, the two antibody cocktail of IMM20184/IMM20253, and the triple antibody cocktail (IMM20184/IMM20190/IMM20253; IMM-BCP-01) relative to isotype control antibodies when assessed in a concentration-dependent manner.
- FIG. 23A-23B depict time-dependent conformational change in Spike protein, as measured by dynamic light scattering, upon binding of IMM20253 or IMM20190.
- FIG. 24 depicts time-dependent protease digestion of Spike and Spike in complex with ACE2, IMM20253, or IMM20190.
- FIGS. 25A-25C depict the combinatorial neutralization activity of IMM20253 in combination with IMM20184, REGN987, or REGN933 against pseudoviruses expressing the Spike protein from the Delta Plus (B.1.617.2 ay1/2) variant. Dark grey area represent regions of synergy. Bottom portion of the figure lists the overall HSA scores for each combination.
- FIG. 25A-25C depict the combinatorial neutralization activity of IMM20253 in combination with IMM20184, REGN987, or REGN933 against pseudoviruses expressing the Spike protein from the Delta Plus (B.1.617.2 ay1/2) variant. Dark grey area represent regions of synergy. Bottom portion of the figure
- FIGS. 27A-27D depict the in vitro binding activity of individual antibodies (FIG. 27A) IMM20184, (FIG. 27B) IMM20190, (FIG. 27C) IMM20253, and (FIG. 27D) IMM20279 to the SARS-CoV-2 Spike-RBD Omicron variant relative to the reference strain.
- FIG. 27A depicts the in vitro binding activity of individual antibodies (FIG. 27A) IMM20184, (FIG. 27B) IMM20190, (FIG. 27C) IMM20253, and (FIG. 27D) IMM20279 to the SARS-CoV-2 Spike-RBD Omicron variant relative to the reference strain.
- FIG. 28 depicts the in vitro neutralization activity of the IMM20253 antibody against pseudovirus expressing the Spike protein from the reference strain (SARS-CoV- 2/human/USA/WA CDC-WA1/2020), DG14G (SARS-CoV-2/human/Germany/BavPat 1/2020), B.1.351 (beta/S. African), B.1.617.2 Ay.2 (Delta Plus) and B.1.1.529 (Omicron) variants of SARS-CoV-2. [0039] FIG.
- FIGS. 30A-30B depict the 3 Fab fragments of IMM20190 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL.
- SARS-CoV-2 trimer is depicted in light grey cartoon.
- IMM20190 Fab fragments are depicted in black cartoon.
- FIG. 30A is the side view and FIG. 30B is the top view.
- FIGS. 31A-31B depict the 3 Fab fragments of IMM20184 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL.
- SARS-CoV-2 trimer is depicted in light grey cartoon.
- IMM20184 Fab fragments are depicted in black cartoon.
- FIG. 31A is the side view and FIG. 31B is the top view.
- FIG. 32A-32B depict a single Fab fragment of IMM20253 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL.
- SARS-CoV-2 trimer is depicted in light grey cartoon.
- IMM20253 Fab fragments are depicted in black cartoon.
- FIG. 32A is the side view and FIG. 32B is the top view. [0043] FIG.
- FIG. 33 depicts a 3.9 Angstrom structure of the variable domains of IMM20184 and IMM20253 bound to an isolated receptor binding domain (RBD) corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC-WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL.
- RBD receptor binding domain
- SARS-CoV-2 trimer is depicted in light grey cartoon.
- IMM20184 variable domains are depicted in black cartoon.
- IMM20253 variable domains are depicted in dark grey cartoon.
- RBD residues predicted to be within 4 Angstroms of IMM20184 are visualized in black spheres.
- RBD residues predicted to be within 4 Angstroms of IMM20253 are depicted in dark grey spheres.
- FIGS. 35A-35B depict an isolated receptor binding domain (RBD) corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC-WA1/2020) of SARS-CoV-2 with (FIG. 34A) predicted IMM20184 contact residues depicted in black spheres and (FIG. 34B) predicted IMM20253 contact residues depicted in dark grey spheres.
- FIGS. 35A-35B depict the 3 Fab fragments of IMM20279 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL. SARS-CoV-2 trimer is depicted in light grey cartoon.
- FIG. 35A depicts lung titers of hamsters dosed prophylactically with IMM20253, IMM20253/IMM20184 or IMM20253/IMM20279 one day prior to inoculation with Omicron BA.1.
- FIG. 36 depicts lung titers of hamsters dosed prophylactically with IMM20253, IMM20253/IMM20184 or IMM20253/IMM20279 one day prior to inoculation with Omicron BA.1.
- Methods of treating the severe acute respiratory syndrome coronavirus 2 (SARS- CoV-2) virus are described herein.
- the methods comprise treating an immunocompromised subject.
- the subject has a high risk for contracting SARS-CoV2.
- the subject has a suppressed immune system.
- the individual is being treated with an immunosuppressive agent.
- These antibodies may be used to neutralize SARS-CoV-2 by preventing the virus from infecting new host cells. Therefore, the inventions disclosed here also relate to pharmaceutical compositions that contain one or more antibodies of the invention, as well as relate to methods of preventing or treating a SARS-CoV-2 infection in a subject in need thereof. Accordingly, the inventions disclosed herein also relate to methods of administering antibody compositions of the invention to a subject in need thereof. [0048] In some embodiments, the antibodies, compositions, and kits provided herein are especially effective for treating and/or preventing SARS-CoV-2 due to particular novel properties.
- antibodies that bind to the Spike protein at multiple different locations are provided herein. This is beneficial because SARS-CoV-2 variants may have one or more mutations in the Spike protein to evade the immune system. Thus providing multiple antibodies that bind to multiple different locations in the Spike protein allows binding and neutralization of such variants. [0049] Moreover, some of the antibodies provided herein bind to an ACE2 binding site in the Spike protein, while others bind outside the ACE2 binding site. Without being bound by theory, providing multiple antibodies, some of which target the ACE2 binding site and some of which target regions outside of the ACE2 binding site may combine to provide more effective treatment for SARS-CoV-2.
- a method of treating or preventing SARS-CoV-2 comprising administering multiple antibodies that bind to non- overlapping epitopes on the Spike protein.
- the method comprises administering an antibody that binds to an ACE2 binding site of the Spike protein and an antibody that binds to an epitope outside of the ACE2 binding site of the Spike protein.
- the method comprises administering an antibody that neutralizes SARS- CoV-2 through an ACE2-dependent mechanism and an antibody that neutralizes SARS-CoV- 2 through an ACE2-independent mechanism.
- dose refers to the amount of a single agent (such as an antibody).
- total dose is used to refer to a total amount of multiple agents that are delivered as a combination therapy (for example, an antibody cocktail).
- an antibody cocktail comprising three antibodies each delivered at a dose of 200 mg is considered to be administered at a total dose of 600 mg.
- the multiple agents in such a combination therapy can be administered in the same or different compositions and simultaneously or sequentially.
- the method comprises administering one antibody provided herein. In some embodiments, the method comprises administering two or more antibodies provided herein. In some embodiments, the method comprises administering three or more, or four or more antibodies provided herein. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the methods provided herein comprise treating an immunocompromised subject with one or more antibodies provided herein (such as two, three, or four antibodies).
- the antibodies provided herein are especially advantageous for treatment of immunocompromised individuals because they facilitate viral clearance from the subject.
- the antibodies are able to engage in complement fixation.
- the antibodies provided herein bind to a Spike protein in an orientation that allows for complement fixation.
- the antibodies bind to a Spike protein in an orientation that provides synergy between the Fc and the antigen binding domain.
- the antibodies effectively neutralize SARS-CoV-2.
- the subject is administered one, two, three, or four antibodies provided herein to treat a SARS-CoV-2 infection.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- the immunocompromised subject has an impaired immune system.
- the immunocompromised subject has decreased resistance to an infection, such as SARS-CoV-2 infection.
- the subject has one or more markers of low immune function.
- the number or activity of T, B, and/or NK cells in the subject is below normal.
- one or more functional markers of immune function is altered in the immunocompromised subject.
- the subject is administered one, two, three, or four antibodies or antigen binding fragments thereof provided herein to treat a SARS-CoV-2 infection.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- the subject has an immunodeficiency disease.
- the immunodeficiency disease is a primary immunodeficiency disease.
- the immunodeficiency results in decreased antibody production and/or decrease of amount or activity of one or more types of immune cells.
- the immunodeficiency disease is an acquired immunodeficiency condition.
- the acquired immunodeficiency condition is caused by another disease or a treatment of another disease.
- the subject has received a solid organ transplant and being treated with one or more immunosuppressants.
- the subject has received a bone marrow transplant.
- the subject is a cancer patient undergoing active chemotherapy.
- the subject is undergoing treatment with an EGFR inhibitor, an ALK inhibitor, a ROS1 inhibitor, a BRAF inhibitor, a RET inhibitor, a MET inhibitor, a NTRK inhibitor, a BTK inhibitor, a PI3K inhibitor, or a BCL-2 inhibitor and or radiation therapy.
- the subject is on dialysis.
- th e subject has end stage renal disease and is on dialysis.
- the individual has multiple sclerosis and is receiving anti-CD20 therapy.
- the subject has an inflammatory disease.
- the subject has an inflammatory disease and is being treated with a maintenance therapy.
- the subject has an inflammatory disease and is being treated with an immunosuppressant maintenance therapy.
- the inflammatory disease is arthritis, psoriasis, or Crohn’s disease.
- the subject has rheumatoid arthritis.
- the inflammatory disease is an auto inflammatory disease.
- the inflammatory disease is selected from the group consisting of ankylosing spondylitis arthritis, osteoarthritis, rheumatoid arthritis (RA), psoriatic arthritis, asthma, atherosclerosis, Crohn's disease, colitis, dermatitis, diverticulitis, fibromyalgia, hepatitis, irritable bowel syndrome (IBS), systemic lupus erythematous (SLE), nephritis, Parkinson's disease, ulcerative colitis, Cryopyrin-Associated Periodic Syndromes (CAPS) including Familial Cold Autoinflammatory Syndrome and Muckle-Wells Syndrome, Systemic Juvenile Idiopathic Arthritis, giant cell arteritis, Paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (a-HUS), vasculitis.
- ankylosing spondylitis arthritis rheumatoid arthritis (RA), psoriatic arthritis, asthma, athe
- the subject is receiving steroids, sulfasalazine, methotrexate, mycophenolate mofetil (IMPDH inhibitor), azathioprine, Anti-IL-1 biologics (anakinra (Kineret®), canakinumab (Ilaris®), rilonacept (Arcalyst®), a TNF ⁇ inhibitors [ Infliximab (Remicade®), Adalimumab (Humira®), Golimumab (Simponi®), Etanercept (Enbrel®), Certolizumab (Cimzia®)], Anti-IL-6 Biologics: Tocilizumab (Actemra®), Sarilumab (Kevzara®); Complement inhibitors: Eculizumab, Anti-CD20 Biologics: Rituximab (Rituxan), BLyS inhibitor (Belimumab (Benlysta®)), Calcineurin inhibitor (cyclospor
- the antibodies or antigen binding fragments thereof that bind to a Spike protein comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is administered one, two, three, or four antibodies or antigen binding fragments thereof provided herein to treat a SARS-CoV-2 infection.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- provided herein are methods of treating a subject who is at high risk for contracting SARS-CoV-2 comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject.
- the subject has a high risk for contracting severe SARS-CoV-2.
- the subject is immunocompromised.
- the subject has one or more risk factors for contracting SARS-CoV-2.
- the subject has one or more risk factors such as, old age, serious heart or lung conditions, weakened immune system, obesity, or diabetes.
- the individual is obese.
- the individual is pregnant.
- the subject has a high risk for contracting other serious respiratory illnesses, such as the flu.
- the individual is age 65 or older.
- the individual has COPD, lung cancer, cystic fibrosis, pulmonary fibrosis, asthma, pulmonary hypertension, pulmonary embolism, cardiomyopathy, congenital heart disease, heart failure, coronary artery disease, Type 1 diabetes, Type 2 diabetes, a high body mass index, HIV/AIDS, chronic kidney or liver diseases, sickle cell or anemia.
- the subject has chronic kidney disease, hypertension, severe neurodevelopment disorders, dementia, Alzheimers disease, multiple co- morbidities, and/or is a nursing home resident.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- provided herein is a method of treating a SARS-CoV-2 infection in a subject that has cancer comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject.
- the subject is being treated with more chemotherapeutic agents.
- the subject has recently received treatment with one or more chemotherapeutic agents. In some embodiments, the subject is receiving or has recently received radiation therapy.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- provided herein is a method of treating a SARS-CoV-2 infection in a subject that has received a solid organ transplant comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject.
- the subject is on one or more immunosuppressive drugs.
- the subject has recently received one or more immunosuppressive drugs.
- the subject is receiving or has recently received an antirejection drug.
- the subject has received a calcineurin inhibitor, an antiproliferative agent, a mTOR inhibitor, and/or a steroid.
- the subject is receiving or has recently received corticosteroid treatment.
- the subject is being treated or has recently received treatment with an IMPDH inhibitor.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject, wherein the antibodies or antigen binding fragments thereof neutralize the SARS-CoV-2 virus.
- the antibodies block viral infection of human cells.
- the antibodies are able to clear SARS-CoV2 viral particles.
- viral clearance is observed within 28 days, or within 12 weeks of administering the antibodies.
- viral load is decreased upon administration of the antibodies or antigen binding fragments thereof.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- Also provided herein are method of treating subjects with moderate or mild SARS- CoV-2 infections comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject.
- the subject does not have severe COVID-19, is not at increased risk of severe COVID-19, and does not have an active infection.
- the subject does not have cancer (basal cell carcinoma and prostate carcinoma in situ [Gleason ⁇ 6] are acceptable), chronic kidney disease, chronic obstructive pulmonary disease, heart condition (congestive heart failure II, III and IV as per New York Heart Association: coronary disease and any other cardiac condition that imposes high risk of developing severe COVID-19), immunocompromised state from solid organ transplant, sickle cell disease, or other condition, autoimmune disease, use of immunosuppressants (including high doses of systemic corticosteroids), type 1 or type 2 diabetes mellitus, current or prior history of smoking or vaping any product, including nicotine or THC.
- cancer basic cell carcinoma and prostate carcinoma in situ [Gleason ⁇ 6] are acceptable
- chronic kidney disease chronic obstructive pulmonary disease
- heart condition congestive heart failure II, III and IV as per New York Heart Association: coronary disease and any other cardiac
- the individual has not received a previous therapy to treat SARS-CoV-2.
- the antibodies comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
- the method comprises administering a single dose of the antibody or antigen binding fragment thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein.
- the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1.
- the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibody or antigen binding fragment thereof.
- a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein.
- the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1.
- the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibody or antigen binding fragment thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 mg of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein.
- the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1.
- the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibody or antigen binding fragment thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein.
- the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1. In some embodiments, the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibody or antigen binding fragment thereof.
- kits for treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 400 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 800 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 1200 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- kits for treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 1600 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0071] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 600 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 mg of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 1200 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- kits for treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 1800 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 2400 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 800 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- kits for treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 mg of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 1600 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0077] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 2400 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein.
- a total dose of 3200 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject.
- the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1.
- the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- the first, second, and third antibodies are administered at a ratio of about 1:1:1.
- the antibodies or antigen binding fragments thereof are administered in a flat dose fashion.
- the first second, and third antibodies are administered at a total dose of about 600 mg to about 2400 mg.
- the antibodies are delivered at a total dose of about 600 mg.
- the antibodies are delivered at a dose about 200 mg each (e.g.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- kits for treating a SARS-CoV-2 infection in a subject comprising administering to the subject at least first, second, and third antibodies or antigen binding active fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 1200 mg.
- the antibodies are administered at a dose of about 400 mg each (e.g. 1200 mg total).
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1.
- the subject is immunocompromised.
- the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
- the administration is intravenous administration.
- Provided herein are methods of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first, second, and third antibodies or antigen binding active fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 1800 mg.
- the antibodies are administered at a dose of about 600 mg each (e.g. 1800 mg total).
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1.
- the subject is immunocompromised.
- kits for treating a SARS-CoV-2 infection in a subject comprising administering to the subject at least first, second, and third antibodies or antigen binding active fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 2400 mg.
- the antibodies are administered at a dose of about 800 mg each (e.g. 2400 mg total).
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1.
- the subject is immunocompromised.
- methods of treating a SARS-CoV-2 infection in a subject comprising administering to the subject at least first, second, and third antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 3 mg/kg to about 6 mg/kg each. In some embodiments, the antibodies or antigen binding fragments thereof are administered at a total dose of about 3 mg/kg each.
- the antibodies or antigen binding fragments thereof are administered at a total dose of about 6 mg/kg each.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1.
- the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1.
- the subject is immunocompromised.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid
- the first, second, and third antibodies are administered at a dose of about 200 mg each.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 compris
- the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 400 mg each.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61,
- the first, second, and third antibodies or antigen binding fragments thereof are delivered at a dose of about 600 mg each.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61,
- the first, second, and third antibodies or antigen binding fragments thereof are delivered at a dose of about 800 mg each.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. [0089] In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 as a monotherapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 as a combination therapy.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 in combination with one or more additional antibodies provided herein.
- the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 is administered simultaneously with one or more additional antibodies provided herein.
- the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 is administered sequentially with one or more additional antibodies provided herein.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCD
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0091] In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 as a monotherapy.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 is administered simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 is administered sequentially with one or more additional antibodies provided herein.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72).
- VH variable heavy chain
- VL variable light chain
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the individual is immunocompromised.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing an infection with an Omicron variant of SARS-CoV-2 in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72
- the method further comprises administering a second antibody.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 as a monotherapy.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 is administered simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 is administered sequentially with one or more additional antibodies provided herein.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the individual is immunocompromised.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114).
- VH variable heavy chain
- VL variable light chain
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing an infection with an Omicron variant of SARS-CoV-2 a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114).
- VH variable heavy chain
- VL variable light chain
- the method further comprises administering a second antibody.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 as a monotherapy.
- the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 is administered simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 is administered sequentially with one or more additional antibodies provided herein. In some embodiments, the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a second antibody comprising a first antibody comprising a VH compris
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of three antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of Y
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 600 mg, about 1200 mg, about 1800 mg, or about 2400 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of three antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 600 mg, about 1200 mg, about 1800 mg, or about 2400 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of three antibodies that bind to a Spike protein comprising a first antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFT
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 600 mg, about 1200 mg, about 1800 mg, or about 2400 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously.
- a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of four antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 800 mg, about 1600 mg, about 2400 mg, or about 3200 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously.
- the method comprises administering one or more of IMM20184, IMM20190, IMM20253, and/or IMM20279 and an additional agent selected from the group consisting of casirivimab, imdevimab, bamlanivimab, etesevimab and sotrovimab.
- an additional agent selected from the group consisting of casirivimab, imdevimab, bamlanivimab, etesevimab and sotrovimab.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering two or more antibodies provided herein to the subject.
- the SARS-CoV-2 infection is caused by SARS-CoV-2 variant.
- the SARS-CoV-2 is the Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gammma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Episolon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2.
- the variant has one or more mutations in a Spike protein.
- 3 or more, 4 or more, 5 or more, or 6 or more antibodies provided herein are administered to the subject.
- the antibodies comprise a HCVR comprising three heavy chain complementarity determining regions (CDRs), (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 1, and a LCVR comprising three light chain complementarity determining regions (CDRs) (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 2; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 3, and
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering two or antibodies provided herein.
- the Omicron variant includes a combination of mutations in the Spike protein. In some embodiments, the mutations include a large number of changes that form a ring around the ACE2 binding site.
- an advantage of the present methods is the ability to target epitopes within and outside of the ACE2 binding site such that the treatment is effective in variants, such as Omicron which harbor mutations within the Spike protein including in the ACE2 binding site.
- variants such as Omicron which harbor mutations within the Spike protein including in the ACE2 binding site.
- the combinations of antibodies provided herein are especially effective for treating variants such as Omicron because they bind to non-overlapping epitopes.
- the Omicron variant has one or more modifications that make the Spike protein more susceptible to cleavage and/or cause pre-cleavage of the Spike protein.
- the antibodies provided herein are able to bind to pre-cleaved Spike proteins, or Spike proteins that are more susceptible to cleavage with high affinity.
- provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering two or antibodies provided herein.
- the Omicron variant includes a combination of mutations in the Spike protein.
- the mutations include a large number of changes that form a ring around the ACE2 binding site.
- an advantage of the present methods is the ability to target epitopes within and outside of the ACE2 binding site such that the treatment is effective in variants, such as Omicron which harbor mutations within the Spike protein including in the ACE2 binding site.
- the combinations of antibodies provided herein are especially effective for treating variants such as Delta because they bind to non-overlapping epitopes.
- the Delta variant has one or more modifications that make the Spike protein more susceptible to cleavage and/or cause pre-cleavage of the Spike protein.
- the antibodies provided herein, for example IMM20253 are able to bind to pre-cleaved Spike proteins, or Spike proteins that are more susceptible to cleavage with high affinity.
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. [0112] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering antibody IMM20184 to the subject.
- the antibody comprises a HCVR comprising three heavy chain complementarity determining regions (CDRs), (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 1, and a LCVR comprising three light chain complementarity determining regions (CDRs) (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 2.
- CDRs heavy chain complementarity determining regions
- LCDR1, LCDR2 and LCDR3 three light chain complementarity determining regions
- the antibody comprise a HCDR1, a HCDR2, and anHCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2.
- the method comprises administering 2 or more antibodies.
- IMM20184 targets an epitope outside the ACE2 binding site and operates via an ACE2 dependent mechanism of action.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody IMM20253 to the subject.
- the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6.
- the antibody comprises a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6.
- IMM20253 targets an epitope outside the ACE2 binding site and operates via an ACE2 independent mechanism of action.
- IMM20253 has high affinity for virus comprising pre-cleaved Spike proteins or Spike proteins that are more susceptible to cleavage, such as Omicron.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody IMM20253 to the subject.
- the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6.
- the antibody comprises a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6.
- IMM20253 targets an epitope outside the ACE2 binding site and operates via an ACE2 independent mechanism of action. In some embodiments, IMM20253 targets an epitope outside the ACE2 binding site and operates via an ACE2 independent mechanism of action. In some embodiments, IMM20253 has high affinity for virus comprising pre-cleaved Spike proteins or Spike proteins that are more susceptible to cleavage, such as Delta. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody MM20190 to the subject.
- the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4.
- the antibody comprises a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4.
- IMM20190 targets an epitope within the ACE2 binding site and operates via an ACE2 dependent mechanism of action.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody MM20190 to the subject.
- the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4.
- the antibody comprises a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4.
- IMM20190 targets an epitope within the ACE2 binding site and operates via an ACE2 dependent mechanism of action.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184 and IMM20253 to the subject.
- the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 and ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6.
- the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 and ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184 and IMM20253 to the subject.
- the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 and ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6.
- the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 and ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6.
- the subject is immunocompromised.
- the subject is receiving an immunosuppressant.
- the subject has received a solid organ transplant or has a chronic inflammatory disease.
- the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- provided herein is a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184, IMM20253, and IMM20190 to the subject.
- the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6; and iii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the LCVR
- the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and a LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6, and iii) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4.
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0120] In some embodiments, provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184, IMM20253, and IMM20190 to the subject.
- the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6; and iii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the LCVR
- the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and a LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6, and iii) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4.
- the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0121] ”
- Subjects refers to any person who has been infected with, or has the potential to be infected with the SARS-CoV-2 virus. In some embodiments of the invention those subjects may be of high risk for contracting the virus as a result of being immunocompromised through genetic mutation or drug treatment.
- subjects may be being treated with immunosuppressive medications as a result of being a solid organ transplant recipient or having a chronic inflammatory disease (e.g. rheumatoid arthritis, psoriasis, chrohn’s disease). They may be being treated with chemotherapeutic, radiation, or targeted agents that suppress immune function for treatment of diseases such as cancer. Subjects may also have conditions that place them into high-risk categories for developing severe COVID-19, such as diabetes, chronic pulmonary conditions, chronic cardiovascular conditions, obesity, or pregnancy. [0122] "Preventing" a disease refers to inhibiting the full development of a disease. Other terms, such as “prophylaxis”, are also understood to refer to the concept of preventing a disease.
- Treating refers to a therapeutic intervention that ameliorates, (i.e., reduces the severity), a sign or symptom of a disease or pathological condition after it has begun to develop.
- the antibodies or antigen-binding fragments thereof contained in the pharmaceutical composition treat or prevent the a SARS-CoV-2 infection by neutralizing SARS-CoV-2 virus and/or a SARS-CoV-2 variant.
- SARS-CoV-2 variants that are responsive to a method of treatment of the invention include the: Alpha (U.K./B.1.1.7), Beta (South African/B.1.351), Gamma (Brazil/P.1), Delta (India/B.1.617.2), B.1.617 (L452R/E484Q) and Epsilon (California/B.1.429/427).
- SARS-CoV-2 variants that are responsive to a method of treatment of the invention include the CDC Variants of Interest, including the Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV- 2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS- CoV-2.
- Alpha U.K./B.1.1.7
- Beta
- an effective amount of an antibody or antibody composition of the invention to prevent or treat SARS-CoV-2 infection does not result in complete protection from a SARS-CoV-2 disease but results in a lower titer or reduced number of SARS-CoV-2 viruses compared to an untreated subject.
- the effective amount results in a 0.5-fold, 1-fold, 2-fold, 4-fold, 6-fold, 8-fold, 10-fold, 15- fold, 20-fold, 25-fold, 50-fold, 75-fold, 100-fold, 125-fold, 150-fold, 175-fold, 200-fold, 300- fold, 400-fold, 500-fold, 750-fold, or 1,000-fold or greater reduction in titer of SARS-CoV-2 virus relative to an untreated subject.
- the effective amount results in a reduction in titer of SARS-CoV-2 virus relative to an untreated subject of approximately 1 log or more, approximately 2 logs or more, approximately 3 logs or more, approximately 4 logs or more, approximately 5 logs or more, approximately 6 logs or more, approximately 7 logs or more, approximately 8 logs or more, approximately 9 logs or more, approximately 10 logs or more, 1 to 5 logs, 2 to 10 logs, 2 to 5 logs, or 2 to 10 logs.
- Antibodies of the invention are typically monoclonal antibodies, meaning an antibody is produced by a single clonal B-lymphocyte population, a clonal hybridoma cell population, or a clonal population of cells into which the genes of a single antibody, or portions thereof, have been transfected.
- Monoclonal antibodies are produced by methods known to those of skill in the art, for instance by making hybrid antibody-forming cells from a fusion of myeloma cells with immune lymphocyte cells.
- An antibody of the invention may also be an "antigen-binding fragment".
- An antigen-binding fragment refers to a polypeptide fragment of an immunoglobulin or antibody that binds antigen or competes with intact antibody (i.e., with the intact antibody from which they were derived) for antigen binding (i.e., specific binding to an epitope of SARS-CoV-2).
- fragment of an antibody molecule includes antigen-binding fragments of antibodies, for example, an antibody light chain variable domain (VL), an antibody heavy chain variable domain (VH), a single chain antibody (scFv), a F(ab')2 fragment, a Fab fragment, an Fd fragment, an Fv fragment, and a single domain antibody fragment (DAb). Fragments can be obtained, e.g., via chemical or enzymatic treatment of an intact or complete antibody or antibody chain or by recombinant means.
- VL antibody light chain variable domain
- VH antibody heavy chain variable domain
- scFv single chain antibody
- F(ab')2 fragment fragment
- Fab fragment fragment
- Fd fragment fragment
- Fv fragment single domain antibody fragment
- DAb single domain antibody fragment
- immunoglobulin variants that are considered antibodies according to the invention include single-domain antibodies (such as VH domain antibodies), Fab fragments, Fab' fragments, F(ab)'2 fragments, single chain Fv proteins ("scFv”), and disulfide stabilized Fv proteins ("dsFv").
- a VH single-domain antibody is an immunoglobulin fragment consisting of a heavy chain variable domain.
- An Fab fragment contains a monovalent antigen-binding immunoglobulin fragment, which can be produced by digestion of whole antibody with the enzyme papain to yield an intact light chain and a portion of one heavy chain.
- an Fab' fragment also contains a monovalent antigen-binding immunoglobulin fragment, which can be produced by digestion of whole antibody with the enzyme pepsin, followed by reduction, to yield an intact light chain and a portion of the heavy chain. Two Fab' fragments are obtained per immunoglobulin molecule.
- a (Fab')2 fragment is a dimer of two Fab' fragments, that can be obtained by treating whole antibody with the enzyme pepsin without subsequent reduction, so Fab' monomers remain held together by two disulfide bonds.
- An Fv fragment is a genetically engineered fragment containing the variable region of the light chain and the variable region of the heavy chain expressed as two chains.
- a single chain (“sc”) antibody such as scFv fragment
- scFv fragment is a genetically engineered molecule containing the VL region of a light chain, the VH region of a heavy chain, linked by a suitable polypeptide linker as a genetically fused single chain molecule.
- a dimer of a single chain antibody such as a scFV2 antibody, is a dimer of a scFV, and may also be known as a "miniantibody”.
- a dsFvs variant also contains a VL region of an immunoglobulin and a VH region, but the chains have been mutated to introduce a disulfide bond to stabilize the association of the chains.
- An antibody of the invention is typically a "human” antibody, which may also be referred to as a "fully human” antibody.
- An antibody which possess a human framework regions and CDRs from a human immunoglobulin is generally be considered to be a human or humanized antibody.
- a human or humanized antibody may contain the framework and the CDRs of an antibody from the same originating human heavy chain, or human light chain amino acid sequence, or both.
- the framework regions may originate from one human antibody, and be engineered to include CDRs from a different human antibody.
- An antibody according to the invention may also comprise a "tagged" immunoglobulin CH3 domain to facilitate detection of the biologic against a background of endogenous antibodies. More particularly, a tagged CH3 domain is a heterogeneous antibody epitope that has been incorporated into one or more of the AB, EF, or CD structural loops of a human IgG-derived CH3 domain.
- CH3 tag may be incorporated into the structural context of an IgG1 subclass antibody, other human IgG subclasses, including IgG2, IgG3, and IgG4.
- Epitope-tagged CH3 domains also referred to as "CH3 scaffolds” can be incorporated into any antibody of the invention having a heavy chain constant region, generally in the form of an immunoglobulin Fc portion. Examples of CH3 scaffold tags, and methods for incorporating them into antibodies are disclosed in International Patent Application No. PCT/US2019/032780.
- Antibodies used to detect epitope tagged CH3 scaffolds, and antibodies of the invention, that comprise epitope tagged CH3 scaffolds are generally referred to herein as "detector antibodies”.
- Some antibodies of the invention may be described as an "isolated” antibody or “isolated” antigen-binding fragment thereof.
- An isolated antibody or antigen-binding fragment thereof has been substantially separated or purified away from other biological components environment, such as a cell, proteins and organelles.
- an isolated antibody or antigen-binding fragment thereof is prepared by a process involving at least one purification step, although the term "isolated” is not intended to refer to a complete absence of such biological molecules or to an absence of water, buffers, or salts or to components of a pharmaceutical formulation that includes an antibody or antigen-binding fragment of the invention.
- antibody or antigen-binding fragment thereof is specific for the Spike protein (S) of SARS-CoV-2.
- an antibody or antigen- binding fragment thereof may, in one embodiment, bind the S1 subunit of the Spike protein, while in another embodiment, an antibody or antigen-binding fragment thereof binds the S2 subunit.
- some antibodies or antigen-binding fragments thereof bind to the receptor-binding domain (RBD) of the S protein, while other antibodies of the invention bind to non-RBD epitopes of the S protein.
- RBD-binding antibodies or antigen-binding fragments thereof of the invention may, in certain embodiments, bind a soluble form of the RBD of the S protein.
- Some antibodies or antigen-binding fragments of the invention bind to sites on the RBD that are resistant to mutational drift.
- an antibody or antigen-binding fragment thereof binds the RBD at, or in close proximity of such a site described by Greaney t al. (2021) as the “the E465 patch”.
- Greaney t al. (2021) as the “the E465 patch”.
- binding of an antibody or antigen-binding fragment of the invention to a highly-conserved site of the SARS-CoV-2 Spike protein is not affected by point mutations in the Spike protein associated with variants of SARS-Cov-2, Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV- 2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B
- an isolated antibody or antigen-binding fragment thereof contains a heavy chain variable region (HCVR) and a light chain variable region (LCVR) that is described in Table 1.
- HCVR heavy chain variable region
- LCVR light chain variable region
- an antibody comprising a HCDR1, a HCDR2, a HCDR3, a LCDR1, a LCDR2, and a LCDR3 of a heavy chain variable region (HCVR) and a light chain variable region (LCVR) that is described in Table 1.
- an antibody comprising a HCDR1, a HCDR2, a HCDR3, a LCDR1, a LCDR2, and a LCDR3 set forth in Table 1.
- the CDRs are determined using Kabat, Chothia, or contact systems. In some embodiments, the CDRs are determined using the system described in North et al. J.M.B 406(8):228-56 (2011) as set forth in Table 1. Table 1 Description SEQ Sequence Name Binds Amino acid sequence ID P _ ( I P _ ( IMM200184) ASWVFGGGTKLTVL Description SEQ Sequence Name Binds Amino acid sequence ID No.
- CD-4I2X A WEIDGSERQNGKTT e b s C e C e b s C e C e b s C e C e b s C e C e b s C e C e b s C e C d e b s C d e C epitope with 233 Description SEQ Sequence Name Binds Amino acid sequence ID No.
- an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGA
- the CDRs are defined according to North et al.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 1.
- the VL comprises the amino acid sequence set forth in SEQ ID NO: 2.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 1 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 2.
- the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 2.
- the antibody comprises one or more CDRs or variable region sequences of IMM20184 (PR193_00018_HC). In some embodiments, the antibody binds to an epitope on the Spike protein outside of the ACE2 binding site. In some embodiments, the antibody neutralizes SARS-CoV-2 through an ACE2-dependent mechanism. In some embodiments, IMM20184 contacts one or more amino acids in the Spike protein selected from the group consisting of N370, A372, F374, K378, S383, and P384. In some embodiments, IMM20184 binds to a conserved epitope.
- IMM20184 binds to an epitope of the Spike protein comprising one or more of N370, A372, F374, K378, S383, and P384.
- VH variable heavy chain
- VL variable light chain
- the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the amino acid sequence
- the CDRs are defined according to North et al.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 3.
- the VL comprises the amino acid sequence set forth in SEQ ID NO: 4.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 3 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 4.
- the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 4.
- the antibody comprises one or more CDRs or variable region sequences of IMM20190 (PR194_00232_HC). In some embodiments, the antibody binds to an epitope of the Spike protein in the ACE2 binding site and operates through an ACE2-dependent mechanism. In some embodiments, IMM20190 contacts one or more amino acids of the Spike protein selected from the group consisting of K417, D420, L455, F456, N460, Y473, N487, Y489, N501, and Y505. In some embodiments, IMM20190 binds to a non-conserved epitope.
- IMM20190 binds to an epitope of the Spike protein comprising one or more of K417, D420, L455, F456, N460, Y473, N487, Y489, N501, and Y505.
- an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72).
- VH variable heavy chain
- VL variable light chain
- the CDRs are defined according to North et al.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 5.
- the VL comprises the amino acid sequence set forth in SEQ ID NO: 6.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 5 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 6.
- the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 6.
- the antibody comprises one or more CDR sequences or variable region sequences of IMM20253 (PR200_00622_HC).
- the antibody binds to a Spike protein with a mutation in the protease cleavage site.
- the antibody binds with high affinity to pre-cleaved Spike proteins (i.e. Spike proteins that are cleaved prior to binding to the host cell surface).
- the antibody binds with high affinity to Spike proteins with mutations that make a protease site more readily cleavable.
- the antibody binds to a Spike protein at an epitope outside of the ACE2 binding site and operates through an ACE2 independent mechanism.
- the antibody causes a confirmation change in the Spike protein. In some embodiments, the antibody makes the Spike protein more susceptible to cleavage. In some embodiments IMM20253 contacts one or more amino acid of the Spike protein selected from the group consisting of K356 and R466. In some embodiments, IMM20253 binds to a conserved epitope. In some embodiments, IMM20253 binds to an epitope of the Spike protein comprising K356 and/or R466.
- an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114).
- VH variable heavy chain
- VL variable light chain
- the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTY
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 19.
- the CDRs are defined according to North et al.
- the VL comprises the amino acid sequence set forth in SEQ ID NO: 20.
- the VH comprises the amino acid sequence set forth in SEQ ID NO: 19 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 20.
- the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 19, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 20.
- the antibody comprises one or more CDR sequences or variable region sequences of IMM20279 (PR199_00255_HC).
- the antibody binds to an epitope of the Spike protein outside of the ACE2 binding site.
- the antibody comprising one or more CDR sequences or variable region sequences of IMM20279 cross-reacts with an antibody comprising one or more CDR sequences or variable region sequences of IMM20184.
- an isolated antibody or antigen-binding fragment thereof binds a SARS-CoV-2 Spike protein, and contains one of the following combinations of a heavy chain variable region (HCVR) and a light chain variable region (LCVR): a HCVR comprising three heavy chain complementarity determining regions (CDRs), (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 1, and a LCVR comprising three light chain complementarity determining regions (CDRs) (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 2; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3),
- antibodies of the invention bind to highly-conserved epitopes, and, thus, are not affected by point mutations in the Spike protein associated with variants of SARS-Cov-2.
- a nonlimiting list of examples of antibodies or antigen-binding fragments of the invention which bind equivalently to the SARS-CoV-2 reference isolate, USA/WA_CDC-WA1/2020, and each of the aforementioned variant isolates include: An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 1 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 2 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 3 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 4 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 5 or fragment thereof, and a LCVR
- the foregoing antibodies of the invention neutralize SARS-CoV-2 and SARS- CoV-2 variants, including variants Alpha (U.K./B.1.1.7), Beta (South African/B.1.351), Gamma (Brazil/P.1), Delta (India/B.1.617.2), B.1.617 (L452R/E484Q) and Epsilon (California/B.1.429/427), either alone, or in combination.
- the antibodies neutralize the Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV- 2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2.
- certain antibodies of the invention bind the SARS-CoV-2 RBD at epitopes, which contain residues that are conserved among variants. In some embodiments, binding to such conserved residues prevents viral escape. In some embodiments, binding to such conserved residues allows binding across variants. Accordingly, a RBD amino acid substitution at a non-conserved or poorly-conserved residue position will have limited or no impact on binding of an antibody of the invention. Such non conserved or poorly-conserved substitutions in the RBD are commonly associated with SARS-CoV-2 variants.
- an RBD epitope of some antibodies of the invention may contain at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 or more residues at RBD positions 356 (K), 370 (N), 372 (A), 374 (F), 378 (K), 384 (P), 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 466 (R), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y).
- an antibody of the invention may bind an RBD epitope in which the binding energy is relatively higher at residues at 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y).
- another antibody of the invention may bind an RBD epitope in which the binding energy is relatively higher at residues at 370 (N), 372 (A), 374 (F), 378 (K), 384 (P).
- yet another antibody of the invention may bind an RBD epitope in which the binding energy is relatively higher at residues at 356 (K) and 466 (R).
- the antibody contacts one or more residues at RBD positions 356 (K), 370 (N), 372 (A), 374 (F), 378 (K), 384 (P), 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 466 (R), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y).
- the antibody contacts one or more of 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y).
- the antibody contacts residues at 370 (N), 372 (A), 374 (F), 378 (K), 384 (P).
- the epitope is identified using alanine scanning.
- the antibody binds the SARS-CoV-2 RBD at epitopes, which contain residues that are not conserved.
- a composition i.e., a mixture of two or more of: An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 1 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 2 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 3 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 4 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 5 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 6 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 19 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 20 or fragment thereof; and An antibody with a HCVR based on the amino acid sequence set forth in S
- the antibodies neutralize the Omicron variant.
- the antibodies neutralize Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2.
- an antibody of the invention alters the conformation of Spike protein upon binding of the antibody to the Spike protein. More particularly, in certain embodiments, an antibody of the invention binds a Spike protein epitope located on the outside face of the RBD domain of the SARS-CoV-2 RBD. In such embodiments, the epitope may be in close proximity to the N-terminal domain of a second Spike protein within the Spike trimer, and binding of the antibody inactivates the virus’ ability to bind cells. Consequently, certain antibodies of the invention neutralize SARS-CoV-2 by the foregoing mechanism.
- binding of such antibodies to the outer face of the RBD imparts its intrinsic neutralization through induction of a conformational change in the Spike protein that prevents virus uptake into the cells without competing for ACE2 binding.
- An example of an embodiment of the invention that alters conformation of the Spike protein upon binding is an antibody with a heavy chain variable region (HCVR) or fragment thereof and/or a light chain variable region (LCVR) or a fragment thereof derived from an antibody described herein as IMM20253, which contains HCVR and LCVR with amino acid sequences of SEQ ID NOS. 5 and 6, respectively.
- an antibody of the invention that alters the conformation of Spike protein upon binding of the antibody to the Spike protein as part of its intrinsic neutralization activity acts additively, and more preferentially synergistically, with other antibodies that compete for ACE2 binding as part of their intrinsic neutralization mechanism.
- An example of an embodiment of the invention that alters conformation of the Spike protein upon binding and acts additively, and more preferentially synergistically, with antibodies that block ACE2 binding is an antibody with a heavy chain variable region (HCVR) or fragment thereof and/or a light chain variable region (LCVR) or a fragment thereof derived from an antibody described herein as IMM20253, which contains HCVR and LCVR with amino acid sequences of SEQ ID NOS.
- HCVR heavy chain variable region
- LCVR light chain variable region
- an isolated antibody or antigen-binding fragment thereof of the invention may contain a CH3 scaffold "epitope tag", comprising at least one modification of the wild-type amino acid sequence of the CH3 domain derived from an immunoglobulin Fc region. Accordingly, any of the aforementioned antibodies may have been engineered to contain a CH3 scaffold.
- the CH3 scaffold of such an isolated antibody or antigen-binding fragment thereof may possess at least one modification of the wild-type sequence occurs within the AB, EF, or CD loops of the CH3 scaffold, including an amino acid substitution, deletion or insertion, for example.
- the epitope tag amino acid sequence contains a sequence derived from SIRP ⁇ or Sip.
- he epitope tag amino acid sequence contains a sequence derived from a constant light chain of an antibody. More particularly, the antibody epitope amino acid sequence of an isolated antibody or antigen-binding fragment thereof that contains a CH3 scaffold with an amino acid sequence set forth in SEQ ID Nos. 3-30, SEQ ID Nos. 33-57, or SEQ ID Nos. 60-67 of International Patent Application No. PCT/US2019/032780.
- Compositions [0145] Also provided herein are compositions comprising one or more antibodies provided herein. In some embodiments, provided herein a composition comprising two or more antibodies that bind to a Spike protein, wherein the antibodies bind to different epitopes.
- the composition comprises an antibody that operates in an ACE2 independent mechanism and an antibody that operates through an ACE2 dependent mechanism.
- the multiple antibodies in the composition act synergistically to treat a SARS-CoV-2 infection in a subject.
- the presence of multiple antibodies binding to different epitopes of a SARS-CoV-2 protein allows treatment of variants with mutations in one or more SARS-CoV-2 proteins, such as the Spike protein.
- a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGASWV (SEQ ID NO: 60).
- VH variable heavy chain
- VL variable light chain
- the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 compris
- the CDRs are defined according to North et al.
- a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the amino acid sequence QKYNSAPGLT (SEQ ID NO: 66).
- VH variable heavy chain
- VL variable light chain
- the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO:
- the CDRs are defined according to North et al.
- a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72).
- the CDRs are defined according to North et al.
- a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114).
- VH variable heavy chain
- VL variable light chain
- the CDRs are defined according to North et al.
- a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67),
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109),
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109),
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HC
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF
- composition comprising a first antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 67), a HCDR2 comprising the
- composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF
- compositions of the invention contain one or more isolated antibodies or antigen-binding fragments thereof of the invention and a pharmaceutically acceptable carrier or diluent.
- the pharmaceutical composition contains only one of the Spike-binding antibodies described herein.
- Other pharmaceutical composition of the invention contain a mixture of different Spike-binding antibodies which are described herein, such as, for example, at least 2, at least 3, at least 4, or at least 5, or at least 6 or more Spike-binding antibodies.
- the antibodies neutralize Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS- CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2.
- a pharmaceutical composition of the invention further contains a second therapeutic agent.
- a pharmaceutical composition of the invention may also contain an anti-inflammatory agent or an antiviral agent.
- the second agent is an antibody.
- the second agent is casirivimab (REGN10933).
- the second agent is imdevimab (REGN10987).
- the second agent is a combination of the neutralizing antibodies, casirivimab and imdevimab (REGEN-COV, previously known as REGN-COV-2) (ClinicalTrials.gov number, NCT04452318 and NCT04425629).
- the two antibodies can simultaneously bind to two independent epitopes on the RBD (Hansen J. et.al., Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail. 369(6506):1010-1014 (2020); Baum A. et.al, Antibody cocktail to SARS-CoV-2 Spike protein prevents rapid mutational escape seen with individual antibodies, Science 369(6506):1014-1018 (2020)).
- the second agent is bamlanivimab (LY3819253).
- the second agent is etesevimab (LY3832479).
- the second agent is a combination of the neutralizing antibodies, bamlanivimab and etesevimab (ClinicalTrials.gov number, NCT04427501, Dougan M. et.al., Bamlanivimab plus Etesevimab in Mild or Moderate Covid-19. N Engl J med 385(15):1382-1392 (2021) ).
- the second agent is sotrovimab (ClinicalTrials.gov number, NCT04545060, Gupta A. et.al., Early Treatment for Covid-19 with SARS-CoV-2 Neutralizing antibody sotrovimab. N Engl J med 385:1941-1950 (2021)).
- kits for treating or preventing a SARS-CoV-2 infection in a subject comprising one or more antibodies provided herein.
- the kit comprises instructions for use according to the methods provided herein.
- the kit comprises instructions for treating an immunocompromised subject. In some embodiments, the kit provides instructions for treating a subject who is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the kit provides instructions for administration of the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg . In some embodiments, the kit provides instructions for In some embodiments, the kit comprises two antibodies that bind to a Spike protein. In some embodiments, the kit comprises three or more antibodies that bind to a Spike protein. In some embodiments, the kit comprises four or more antibodies that bind to a Spike protein.
- the anti-Spike antibodies are in different compositions in the kit. In some embodiments, the anti-Spike antibodies are in the same composition. [0165] In some embodiments, the kit comprises one or more of IMM20184, IMM20190, IMM20253, and/or IMM20279 and an additional agent. In some embodiments, the kit comprises one or more of IMM20184, IMM20190, IMM20253, and/or IMM20279 and an additional agent selected from the group consisting of casirivimab, imdevimab, bamlanivimab, etesevimab and sotrovimab.
- the kit comprises an anti- Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGASWV (SEQ ID NO: 60).
- VH variable heavy chain
- VL variable light chain
- the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55);
- the CDRs are defined according to North et al.
- the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- the kit comprises an anti-Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the
- the CDRs are defined according to North et al.
- the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- the kit comprises an anti-Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and
- the CDRs are defined according to North et al.
- the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- the kit comprises an anti-Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a
- the CDRs are defined according to North et al.
- the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- the kit comprises an anti-Spike antibody comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising
- each antibody is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 55), a
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 55), a
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 67),
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of three anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of three anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of three anti-Spike antibodies comprising a first antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDG
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
- the kit comprises a combination of four anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid
- the kit comprises a combination of casirivimab and imdevimab (REGEN-COV, previously known as REGN-COV-2).
- the kit comprises a combination of bamlanivimab and etesevimab.
- the kit comprises sotrovimab.
- each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
- each antibody is provided in a separate composition.
- each antibody is provided in the same composition.
- a method of treating a SARS-CoV-2 infection in an immunocompromised subject comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
- Embodiment 2 Embodiment 2.
- a method of treating a SARS-CoV-2 infection in an immunocompromised subject comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 independent mechanism and the second antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
- a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 independent mechanism and the second antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism.
- Embodiment 5 The method of any one of embodiments 1-4, further comprising administering a third antibody or antigen binding fragment thereof that specifically binds to the Spike protein of SARS-CoV-2 at an epitope distinct from the first and second antibodies or antigen binding fragments thereof. 6.
- Embodiment 3 wherein the third antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
- Embodiment 7 The method of embodiment 5 or embodiment 6, wherein the third antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism.
- Embodiment 8. The method of any one of embodiments 5-7, wherein each of the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 600 mg to about 2400 mg.
- Embodiment 9 The method of embodiment 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 600 mg.
- Embodiment 11. The method of embodiment 8, wherein first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 1200 mg.
- Embodiment 12. The method of embodiment 11, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 400 mg each.
- Embodiment 13 The method of embodiment 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 1800 mg.
- Embodiment 14. The method of embodiment 13, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 600 mg each.
- Embodiment 15 The method of embodiment 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 2400 mg.
- Embodiment 16 The method of embodiment 15, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 800 mg each.
- Embodiment 17. The method of any one of embodiments 5-8, wherein the first, second and third antibodies or antigen binding fragments thereof are administered at a dose of about 3 mg/kg, or about 6 mg/kg each.
- a method of treating a SARS-CoV-2 infection in an immunocompromised subject comprising administering to the subject an antibody or antigen binding fragment that binds to the Spike protein of SARS-CoV-2, wherein the antibody or antigen binding fragment comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 2; a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 4; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 5, and
- Embodiment 19 The method of embodiment 18, wherein the antibody or antigen binding fragment thereof comprises a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 2; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 4; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 6; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 7, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 8; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 9, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 10; a HCVR comprising the amino acid sequence set forth in SEQ ID NO
- Embodiment 20 The method of embodiment 18, wherein the antibody or antigen binding fragment thereof comprises a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60; a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino
- Embodiment 21 The method of any one of embodiments 18-20, comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a V
- Embodiment 22 The method of any one of embodiments 5-20 comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL compris
- Embodiment 23 The method of embodiment 18 comprising administering to the subject a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising
- Embodiment 24 The method of any one of embodiments 1-23, wherein the subject has a high risk for contracting SARS-CoV-2.
- Embodiment 25 The method of any one of embodiments 1-24, wherein the subject is being treated with an immunosuppressant.
- Embodiment 26 The method of embodiment 25, wherein the immunosuppressant is a corticosteroid.
- Embodiment 27 The method of any one of embodiments 1-26, wherein the subject has received a solid organ transplant or has a chronic inflammatory disease.
- Embodiment 28 The method of embodiment 27, wherein the subject has a chronic inflammatory disease selected from the group consisting of rheumatoid arthritis, psoriasis, and Crohn’s disease.
- Embodiment 29 The method of any one of embodiments 1-24, wherein the subject is being treated with a chemotherapeutic agent or radiation.
- Embodiment 30 The method of any one of embodiments 1-29, wherein the subject is at risk for developing severe COVID-19.
- Embodiment 31 The method of any one of embodiments 1-24 or 30, wherein the subject has diabetes, a chronic pulmonary condition, a chronic cardiovascular condition, obesity, or is pregnant.
- Embodiment 32 The method of any one of embodiments 1-31, wherein the viral load in the subject is decreased upon treatment.
- Embodiment 33 The method of any one of embodiments 1-32, wherein administration of the antibody or antigen-binding fragment thereof results in viral clearance in the subject.
- Embodiment 34 The method of any one of embodiments 1-32, wherein administration of the antibody or antigen-binding fragment thereof results in viral clearance in the subject.
- Embodiment 35 The method of any one of embodiments 1-17 and 21-34, wherein the ratio between the first and the second antibodies or antigen binding fragments thereof is about 1 : 1.
- Embodiment 36 The method of any one of embodiments 1-17 and 22-34, wherein the ratios between the first, second, and third antibodies or antigen binding fragments thereof are about 1 : 1 : 1.
- Embodiment 37 The method of embodiment 23, wherein the ratio between the first, second, third, and fourth antibodies or antigen binding fragments thereof is about 1 : 1 : 1: 1.
- Embodiment 38 The method of embodiment 38.
- the antibody or antigen binding fragment therefor is an Fc IgG1, IgG2, IgG3, IgG4, IgM, IgD, IgA1, IgA2 or IgE isotype.
- Embodiment 39. The method of embodiment 38, wherein the antibody is an IgG1 isotype.
- Embodiment 40 The method of embodiment 39, wherein the IgG1 is a G1m1 or nG1m1 allotype.
- Embodiment 41 The method of any one of embodiment 1-40, wherein the antibody is a fully human antibody.
- Embodiment 42. The method of any one of embodiments 1-41, wherein the antibody is a full length antibody.
- Embodiment 46 The method of embodiment 45, wherein the SARS-CoV-2 variant is the U.K. (B.1.1.7) variant of SARS-CoV-2, the South African (B.1.351) variant of SARS-CoV-2, the California (B.1.429) variant of SARS-CoV-2, the California (B.1.427) variant of SARS- CoV-2, the Brazilian (P.1) variant of SARS-CoV-2, the New York (B.1.526) variant of SARS-CoV-2, the New York (B.1.526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2 or the Omicron (B.1.1.529) variant.
- the SARS-CoV-2 variant is the U.K. (B.1.1.7) variant of SARS-CoV-2, the South African (B.1.351) variant of SARS-CoV-2, the California (B.1.429) variant
- Embodiment 47 The method of any one of embodiments 1-46, wherein the antibody or antigen binding fragment thereof treats SARS-CoV-2 variant and nonvariant infections with about equivalent efficacies.
- Embodiment 48 The method of any one of embodiments 1-47, wherein the administering of the antibody or antigen binding fragment thereof is intravenous.
- Embodiment 49 The method of any one of embodiments 1-17 and 22-48, wherein the first, second, and third antibodies or antigen binding fragments are administered sequentially or simultaneously.
- Embodiment 50 The method of any one of embodiments 1-17 and 22-48, wherein the first, second, and third antibodies or antigen binding fragments are administered sequentially or simultaneously.
- Example 1 Evaluation of the breadth of patients’ humoral responses against SARS-CoV-2. The overall spectrum of the productive antibody response to SARS-CoV-2 was examined using an automated, high-throughput hybridoma library generation and screening platform [Puligedda et al.
- Hybridomas stably expressing human mAbs were generated by electrofusion of expanded B-cells to the B5-6T myeloma cell line, which expresses an ectopic human telomerase gene that stabilizes human chromosomes in the hybrid cells created.
- Fused hybridomas were plated into 96-well plates in growth medium with HAT selection of stable hybridomas for 7 days. After 7 days, growth media were switched to media with HT for stable selected hybridoma growth.
- Hybridomas were cultured in a 37°C incubator for 14-21 days during which time they were imaged for monoclonality and monitored for isotype-and sub-class-specific Ig secretion. Supernatants from monoclonal wells expressing measurable levels of Ig were cherry-picked and submitted for target-based screening. [0182] Screening Assays For Antiviral Antibodies. The naturally occurring human antibodies (IgM, IgG, and IgA isotypes) secreted by those hybridomas were screened for reactivity against a panel of SARS-CoV-2 proteins. Antibody screening assays were developed for three SARS-CoV-2 structural proteins (S, N, M) and a panel of accessory ORF proteins of SARS-CoV-2.
- the screening assays included a rapid and sensitive homogeneous time resolved fluorescence (hTRF) assay that used soluble recombinant viral proteins, as well as a selective, cell-based flow cytometry assay that allowed probing of antibodies to transiently transfected viral antigens expressed within the context of human cells.
- hTRF homogeneous time resolved fluorescence
- Viral protein expression in the cell-based assay was additionally confirmed by Western blot.
- the localization of the C-terminus truncated Spike protein (S ⁇ 19aa) was confirmed to be on the surface of the transfected cells.
- terbium labeled anti-human IgG (H+L) (Cisbio, custom label) donor and AF488-labeled anti-HIS (Cell Signaling, Cat # 14930S) acceptor antibodies was used to screen patient-derived antibodies for their binding to recombinantly produced SARS-CoV-2 antigens, and more specifically to the SARS-CoV-2 Spike protein.
- This recombinant target panel consisted of a full-length S (FL, trimer-stabilized, LakePharma) and two truncated S protein domains, S1 (GenScript, Cat # Z03485-1) and RBD (aa 319-591, LakePharma).
- S1 GenScript, Cat # Z03485-1
- RBD aa 319-591, LakePharma
- Commercially available antibodies specific for the individual structural viral proteins SARS-CoV/SARS-CoV-2 Spike S1 (RBD) chimeric mAb (Sino Biological, Cat # 40150-D001), SARS CoV-2 Nucleocapsid human chimeric mAb (GenScript, Cat # A02039-100 served as controls.
- Assay background was determined by averaging the signal of wells containing only the donor and acceptor cocktail.
- Hybridoma supernatants exhibiting signals greater than 2-fold over background were reported as positive HITs and are submitted for Ig sequence analysis.
- Flow cytometry-based cellular screens for antiviral antibodies SARS-CoV-2 antigen sequences were cloned into pcDNA3.4 plasmids and transfected into 293F cells utilizing the Expi293 Expression System (Life Technologies, Inc.) per manufacturer’s instructions. Cells transiently expressing SARS-CoV-2 S (S ⁇ 19aa) were used for screening. Optimal protein expression was achieved three days post-transfection for S proteins.
- the cell suspensions were dispensed into 384-well plates, followed by the addition of hybridoma supernatant at a 1:10 final dilution. The reaction was allowed to incubate for 90 minutes at room temperature. Cells for each assay were fixed with a final concentration of 1% paraformaldehyde and analyzed with IntelliCyte iQue Screener (IntelliCyte Corporation). Positive binding gates for detection of each secondary antibody were established using cells plus secondary antibody cocktail as a negative control. Binding of hybridoma supernatant antibodies to specific SARS-CoV-2 proteins, and more specifically the S ⁇ 19a, were quantified as percent positive relative to the secondary only control.
- Example 2 Immunoglobulin gene usage in convalescent COVID-19 patients. Ig gene usage in memory B cells of six COVID-19 patients was evaluated using an NGS analysis of identified and sequenced 134 hybridoma hits. RNA isolation and Next Generation Sequencing (NGS). Hybridoma RNA was isolated using Invitrogen RNAqueous-96 Total RNA Isolation Kit (Cat. #AM1920). Isolated RNA samples were submitted to iRepertoire (Huntsville, AL) for NGS. Hybridoma-derived RNA samples were sequenced using the Illumina MiSeq system at iRepertoire (Huntsville, AL).
- Immunoglobulin sequences containing CDR1, 2 and 3 and framework regions were amplified using IgG and IgA-specific mixes for IgH, and kappa and lambda- specific primers for IgL.
- IgM-expressing hybridoma samples, from which IgG or IgA heavy chains were not amplified using this approach, were sequenced using the iRepertoire iPair system. Final sequences were exported using iPair software.
- Immunoglobulin sequences were analyzed for predicted CDR sequences, % identity to appropriate germlines, isotype of the constant regions and read counts.
- Antibody- containing supernatants or purified antibodies were advanced to downstream assays. If multiple heavy or light chain sequences were detected within one well, their CDRs were aligned and compared for potential PCR errors. In cases where multiple sequences within a well were different, i.e., originated from separate clones, all potential combinations of light and heavy chains were recombinantly produced and tested in downstream assays. Wells that yielded a single HC/LC pair were advanced to recombinant expression and downstream assays. 5’ fragments of the constant regions were sequenced to identify the isotype of the antibody and compared to the experimentally identified isotype of hybridoma supernatants.
- the resulting isotype of the heavy or light chain was assigned based on two or more positive readings from experimental (ex. ELISA and FACS) assays and sequencing.
- a combined analysis of Ig isotype and their level of SHM of virus-specific antibodies revealed several key properties of the productive antiviral response.
- Purified recombinant antibodies were assessed, in the HTRF assay described above, using either soluble RBD or S1 domains containing mutations found in naturally occurring viral variants, as well as mutations predicted to decrease binding of neutralizing antibodies.
- Table 3 depicts the binding of identified anti-Spike antibodies, expressed as fold-binding over background. As anticipated, binding of some antibodies, such as PR201_00151 and PR194_00232, are negatively affected by specific mutations within the Spike domain (e.g. K417N).
- binding by antibodies like PR199_00255, PR193_00018, and PR200_00622 are unaffected by the range of mutations analyzed, including variants containing single point mutations, as well as multiple point mutations that mimic the naturally occurring Spike proteins found on the B.1.1.7 and B.1.351 virus isolates.
- a subset of antibodies, such as PR199_00255 and PR200_00622 also bind to Spike protein found on SARS-CoV-1, suggesting that they bind to a highly conserved epitope.
- Binding of the antibodies PR913_00018, PR194_00232, and PR200_00622 were further characterized in the HTRF assay to determine EC50 of binding to a wide range of single and multi-point mutations, including to the B.1.1.7, B.1.429, P.1, and B.1.351 variants.
- PR194_00232 is the most sensitive of the three antibodies to mutational drift within the Spike protein. Mutations, such as K417N and N501Y, mutations known to exist in naturally occurring variants, and consistent with the alanine scanning data, significantly decrease the ability of PR194_00232 to bind the Spike protein. Whereas other naturally occurring mutations, such as L452R, E484Q, or D614G do not impact binding relative to binding to the Washington reference Spike protein. In contrast, binding of PR193_0018 and PR200_00622 are not significantly impacted by any of the single point mutations tested.
- binding by PR193_0018 and PR200_00622 to specific variant Spike proteins appears to be modestly enhanced over the reference Spike.
- Similar observations were made when binding of PR193_0018, PR194_00232, and PR200_00622 was tested against RBD domain proteins containing the full complement of mutations known to exist in four different variants. Of particular interest is the binding to RBDs corresponding to the South African (B.1.351) and U.K. (B.1.1.7) variants. As predicted by the K417N and N501Y single point mutation data, binding of PR194_00232 to both the U.K. and S.A. variants is weaker than observed against the reference strain Spike protein.
- Identified anti-Spike antibodies bind to non-overlapping epitopes within the RBD of SARS-Co-V-2 Spike.
- Purified recombinant forms of the PR193_00018, PR194_00232, and PR200_00622 failed to compete with each other for binding to the SARS-CoV-2 RBD when assessed via biolayer interferometry on an Octet QKe instrument.
- These data suggested the three antibodies bind to non-overlapping epitopes on the SARS- CoV-2 RBD.
- Alanine scanning of the RBD was performed to identify residues critical for binding of each of the antibodies. Consistent with each of the antibodies binding to non- overlapping epitopes, non-overlapping sets of residues were identified as being critical for binding of the antibodies (Table 5).
- Identified anti-Spike antibodies neutralize SARS-CoV-2 pseudovirus.
- the functional consequence of antibodies binding to Spike was assessed in assays using pseudotyped replication-incompetent lentivirus which were used to infect HEK293 cells overexpressing Angiotensin converting enzyme 2 (ACE2).
- Spike-expressing pseudovirus was generated with System Bioscience’s pPACK-SPIKE packaging system (System Biosciences, Cat #CVD19-500A-1) as per manufacturer’s protocol. Briefly, 8 x 10 6 293TN Producer cells (System Biosciences, LV900A-1) were plated in T150 flasks overnight.
- Plasmids encoding lentiviral packaging proteins and Spike were added 1 mL of plain DMEM for each T150 being transfected.
- 55 mL of PureFection reagent (System Biosciences; Cat #LV750A-1) was added to each 1 mL tube, vortexed for 10 seconds, and incubated at room temperature for 15 minutes.
- the plasmid and PureFection mixture were added to a T150 flask containing 293TN cells and placed in a 37°C incubator containing 5% CO 2 for 48 hours.
- Pseudovirus-containing supernatants were harvested at 48 hours and passed through a 0.45 micron PVDF filter to remove cellular debris.
- Pseudoviruses were produced with the pPACK-SPIKE packaging kit, 10 4 ACE2- 293T cells were plated in the inner 60 wells of a white opaque 96 well flat bottom plate (Corning; Cat #3917) in 100 ⁇ L of ACE2-293T media overnight in a 37°C incubator containing 5% CO2. To determine infectivity of each lot of pseudovirus, pseudovirus- containing supernatants were thawed from -80°C and two-fold dilutions were performed. 100 mL of pseudovirus at various dilutions was added to ACE2-293T cells.
- RVP right atrial pressure
- 384 well plate 384 well plate
- Media containing the indicated amount of antibody was added to each well, resulting in a final volume of 100 mL per well (96 well plate) or 25 mL per well (384 well plate).
- the antibody/RVP mixture was pre-incubated for 1 hour in a 37°C incubator containing 5% CO 2 .
- ACE2-293T target cells were added to each well (2 x 10 4 cells in 100 mL for a 96 well plate or 0.9 x 10 4 cells for a 384 well plate) and incubated for 72 hours.
- PR194_00232 antibody that exhibited potent neutralization against pseudovirus expressing both the reference (SARS-CoV-2/human/USA/WA_CDC- WA1/2020) and D614G (SARS-CoV-2/human/Germany/BavPat 1/2020) were also assessed in neutralization assays using live virus of each of the isolates (FIGSs 4A and 4B).
- Full dose response of purified antibodies confirmed strong neutralizing activity by antibodies such as PR194_00232.
- the D614G variant is a widespread mutation [Plante et al. 2020] found in a number of different isolates.
- Pseudovirus particles expressing Spike proteins that mimic the U.K. (B.1.1.7) and South African (B.1.351) isolates are neutralized by antibodies which retain binding to the mutated Spikes (Tables 3 &4). This is exemplified by the antibodies PR193_00018 and PR200_00622 (FIGS. 5A-5C).
- Example 7 A cocktail of three anti-SARS-CoV-2 anti-Spike antibodies elicit combinatorial effects.
- antibodies selective for non-overlapping epitopes on the SARS-CoV-2 Spike protein were assessed in pair-wise, and three-way combinations to identify additive, or preferably synergistic, neutralization.
- the antibodies PR194_00232 (IMM20190), PR193_00018 (IMM20184), and PR200_00622 (IMM20253) were assessed in combinations to evaluate the combinatorial impact on neutralization of pseudovirus expressing a range of different variations of the Spike protein (FIGS. 6A-6C).
- IC50 values for the triple combination and double combination comprising IMM20184 and IMM20253 were determined using RVPs, as described above.
- 6AA-6C the triple combination of antibodies neutralized pseudoviruses corresponding to the USA/WA_CDC-WA1/2020 (reference sequence) and the CDC variants of concern (alpha/U.K./B.1.1.7, beta/South African/B.1.351, gamma/Brazil/P.1 and epsilon/California/B.1.429/427).
- the IC 50 s for neutralization of the reference and California variants were not determined due to the potency exhibited against those variants; concentrations sufficiently low enough to obtain below 50% neutralization were not tested.
- the observed combination response matrix of pseudovirus neutralization was used as input for the online SynergyFinder platform (4), where quadruplicate data points were input separately.
- the highest single agent (HSA) reference model was applied, which quantifies synergy as the excess over the maximum response of a single drug in the combination.
- Synergy between antibodies in each combination is reported as an overall synergy score (the average of observed synergy across the dose combination matrix) as well as a peak HSA score (the highest synergy score calculated across the dose combination matrix).
- Synergy scores of less than -10, between -10 and 10, and greater than 10 indicate antagonistic, additive, and synergistic antibody combinations, respectively.
- the residues critical for binding of IMM20184, IMM20190, and IMM20253 are spatially distinct from the residues mutated in the RBD of the B.1.617.2 (delta), B.1.617.3 (kappa), and C.37 (lambda) variants.
- B.1.617.2 delta
- B.1.617.3 kappa
- C.37 lambda
- IMM20190 binds to a large epitope that encompasses two distinct regions on the RBD (FIG. 2).
- K417 and N501 residues known to be mutated in different variants, represent residues in each of the two binding sites. Mutation of one of those sites (N501Y), as observed in the alpha/B.1.1.7 variant, is sufficient to maintain IMM20190 activity (FIGS. 5A-5C) and provides for improved synergy with IMM20184/IMM20253.
- the delta plus/B.1.617.2.ay1/2 variant contains a K417N mutation.
- Example 9 Triple combination is active against live virus
- neutralization assays on four different live virus variants under BSL3 conditions: USA/WA_CDC-WA1/2020 (reference sequence), Germany/BavPat 1/2020 (D614G), UK (B.1.1.7), and South African (B.1.351).
- PRA pseudovirus neutralization assays
- Assays using the Germany/BavPat1/2020 strain were performed in a manner sufficient to assess combinatorial effects via the HSA algorithm. Data demonstrated an overall additive effect, with peak HSA scores reaching levels of synergy (FIG. 7B)
- Example 10 Example 10.
- Abs neutralize virus in a hamster model of SARS-CoV-2 Antibodies capable of neutralizing live virus in vitro were assessed for the ability to neutralize virus in vivo using a hamster model of SARS-CoV-2 infection.
- Hamsters treated with increasing doses of PR194_00232 were infected with SARS-CoV-2 (SARS-CoV- 2/human/USA/WA_CDC-WA1/2020) and viral load in the lungs were assessed at Day 4 post inoculation of the virus using standard tissue culture infectivity assays and plaque counting.
- PR194_00232 was able to neutralize virus, relative to no-treatment controls, in a dose- dependent manner when dosed in the prophylactic setting (FIG. 12).
- IMM-BCP-01 is able to clear virus from the lungs of hamsters infected with variants of concern. Hamsters were dosed prophylactically with IMM-BCP-01 one day prior to inoculation with either the Alpha (FIG. 16A) or Beta (FIG.
- IMM-BCP-01 at the lowest doses tested provided levels of viral clearance that match, or exceed, those obtained by antibodies with demonstrated clinical efficacy.
- increased doses of IMM-BCP-01 led to a dose- dependent improvement in viral clearance, with > four-log clearance of WA1/2020 and 2.5 log clearance of Beta at the highest doses tested. All doses tested represent clinically relevant doses.
- Robust activity of the cocktail was observed regardless of inoculation titer used (FIG. 17).
- IMM20184, IMM20190, and IMM20253 bind to both isolated RBD and Spike trimer when assessed by surface plasmon resonance (FIGS. 18A-18C).
- IMM20184 can bind avidly to the Spike trimer, as signified by the decrease off-rate (Table 7), and crosslink two Spike monomers.
- IMM20190 Consistent with its epitope overlapping with the ACE2 binding site, IMM20190 is able to compete binding of ACE2 to isolated RBD protein from the REF and Alpha variant (FIG. 19A and FIG. 19B), but its ability to compete ACE2 binding to the Beta variant is decreased (FIG.
- IMM20184 effectively competes binding of ACE2 to all three isolated RBD (FIGS. 19A – 19C), despite binding to an epitope that is outside of known ACE2 binding site (FIG. 2). Despite exhibiting neutralization activity against both pseudovirus and live virus, IMM20253 is unable to effectively compete for ACE2 binding (FIGS. 19A-19C). This suggests the neutralization is due to a mechanism distinct from direct ACE2 competition. [0223] IMM20184, IMM20190 and IMM20253 were assessed for the ability to fix complement using standard assays [Nikitin et al., 2019]. As depicted in FIGS.
- IMM-BCP-01 exhibited enhanced activity, over a wider concentration range, as compared to any of the individual antibodies and the IMM20184/IMM20253 two antibody combination.
- IMM-BCP-01 induces a more robust antibody-dependent cellular cytotoxicity than any of the individual component antibodies when assessed in vitro using Promega’s ADCC Reporter Bioassay and S-expressing CHO-K1 target cells at a 2:1 effector:target cell ratio and manufacturer protocols.
- the IMM-BCP-01 cocktail robustly induces multiple effector functions in a manner that is enhanced by the presence of two or more of its constituent antibodies as compared to the individual antibodies alone.
- IMM20253 may neutralize SARS-CoV-2 through a mechanism that alters Spike protein conformation. As depicted in FIG. 2, IMM20253 binds to an epitope that is on the outside face of the RBD domain of the SARS-CoV-2 RBD. When in the closed conformation, the epitope is in close proximity to the N-terminal domain of a second Spike protein within the Spike trimer.
- Nb nanobodies
- Biochemical characterization the Class III Nb demonstrates that binding of the Nb to the Spike protein induces a conformational change to the post-fusion conformation. This presumably inactivates the virus’ ability to bind cells and provides a mechanism for the neutralization observed in vitro.
- IMM20253 is unable to directly compete for ACE2 binding (FIGS. 20A-20C), but is able to neutralize both reference and alpha variant live virus as a single agent when measured as a function of virus internalization (Table 9). Together, these data suggest that IMM20253 binding to the outer face of the RBD may impart its intrinsic neutralization through induction of a conformational change in the Spike protein that prevents virus uptake into the cells without competing for ACE2 binding.
- IMM20253 appears to be approximately 30-times more potent against the alpha strain than the delta strain. This may be a function of the internalization kinetics of the two different variants. This difference may also underlie the strong synergy observed between the IMM20184/IMM20190/IMM20253 in the context of neutralizing the alpha variant (FIGS. 7A-7C). It should be noted that like Greaney et al [Greany e al], Sun et al describe the region around the IMM20253 epitope as being of therapeutic interest, one to which antibodies are not known to exist [Greany et al] and one that is going to be difficult for antibodies to access [Sun et al].
- IMM20190/Spike trimer complex maintains a similar hydrodynamic radius throughout the two hour incubation.
- Binding of either IMM20253, IMM20184, or the combination of the two antibodies (IMM20253/IMM20184) induces combinatorial effects, preferably synergy, when combined with the ACE2-competitive antibody IMM20190.
- Combinations of IMM20253, IMM20184, or IMM20253/IMM20184 induces synergy with other ACE2-competing enzymes.
- IMM20253, IMM20184, or IMM20253/IMM20184 combines with one or more of the following antibodies to induce combinatorial, preferably synergistic, viral neutralization in vitro and promote in vivo viral clearance.
- Examples of antibodies that combine to induce the combinatorial effect with IMM20253, IMM20184 or IMM20253/IMM20184 include, but are not limited to, sotrovimab, casirivimab, imdevimab, bamlanivimab, etesevimab, tixagevimab, cilgavimab, ADG2, ADG10, ADG20, ADG30, and CR3022.
- IMM20253 works in combination with IMM20184, as well as in-house generated versions of REGN987 (imdevimab) and REGN933 (casirivimab) when assayed against B.1.617.2 ay2 pseudovirus.
- Example 14 IMM20253 and IMM20279 and exhibit potent binding to the Spike protein of the Omicron variant. Individual antibodies were assessed, in the HTRF assay described above, using the full length Spike protein and soluble RBD domains of the Spike protein containing mutations found in the Omicron variant. FIGS.
- FIG. 27A-27D depict the binding of the individual antibodies to the Spike-RBD of the Omicron variant, expressed as percentage binding, relative to the binding to the reference strain.
- Binding of the antibodies IMM20190 and IMM20184 are negatively affected by the mutations within the Spike domain of the Omicron variant.
- binding by the IMM20253 (FIG. 27C) and IMM20279 (FIG. 27D) antibodies are unaffected by the range of mutations within the Spike protein of the Omicron variant.
- the binding affinity of the IMM20279 antibody to the RBD as well as the full-length Spike protein is comparable to that of the reference strain (FIG. 27D).
- IMM20253 antibody neutralizes SARS-CoV-2 pseudovirus expressing the Omicron variant.
- the functional consequence of the IMM20253 antibody binding to Spike was assessed. Pseudovirus infection and neutralization assays were performed by using standard methods described above.
- the IMM20253 antibody exhibited potent neutralization against pseudovirus expressing the Spike protein from the B.1.1.529 (Omicron) variant (FIG. 28).
- the neutralization activity was comparable to the activity against pseudovirus expressing the Spike protein from the reference strain (SARS-CoV- 2/human/USA/WA_CDC-WA1/2020), D614G (SARS-CoV-2/human/Germany/BavPat 1/2020), B.1.351 (beta/S.
- IMM20253 exhibited equivalent neutralization of psuedovirus expressing the spike of either the BA.1 or BA.2 sub-lineage of the Omicron variant (FIG. 29).
- IMM20184 and IMM20279 act to enhance IMM20253 activity in vivo. Hamsters were dosed prophylactically with IMM20253 (0.3 mg, 1 mg, or 3 mg), IMM20253/IMM20184 (0.5 mg each or 1.5 mg each), or IMM20253/IMM20279 (1.5 mg each) one day prior to inoculation with the Omicron BA.1 (FIG.
- IMM20253 exhibited a dose-dependent decrease in lung titer. While IMM20184 showed a substantial decrease in in vitro binding of omicron Spike protein relative to REF (FIG. 27A) and IMM20279 also showed decreased binding of omicron Spike protein relative to REF (FIG. 27D), the combinations of the combination of IMM20253/IMM20184 and IMM20253/IMM20279 exhibits more robust clearance of Omicron BA.1 in vivo.
- Example 17 CryoEM structures of IMM20184, IMM20290, IMM20253, and IMM20279 in complex with the SARS-CoV-2 spike protein.
- the structures of antibodies in complex with the intact trimer of the SARS-CoV-2 trimer were solved by cryoEM to a resolution of approximately 7 Angstroms (FIGS 30A-30B, FIGS. 31A-31B, FIGS. 32A-32B, and FIGS. 34A-34B).
- IMM20279 binds to a site that appears to overlap with the binding site of IMM20184.
- Three Fabs of IMM20190 bind simultaneously to the trimer in a manner consistent with the Class 1 binders as defined by the CoVIC consortium and induce all three RBDs to adopt the up conformation (FIGS. 30A-30B).
- Three Fabs of IMM20184 bind simultaneously to the spike trimer, perpendicular to the vertical axis of the spike. Binding appears to induce a large conformational change in the spike trimer, particularly in the RBD domains.
- IMM20279 binds to similar regions on RBD as compared to IMM20184, but does so by binding from a different angle of attack. It binds at approximately a 45 degree angle to the vertical axis of the spike as compared to the 90 degree angle adopted by IMM20184. [0236] Only a single FAb of IMM20253 is ever observed to bind to the intact trimer (FIGS. 32A-32B). Binding results in loss of density in the cryoEM structure associated with the RBD and N-terminal domain of spike monomer that is not bound by the Fab fragment.
- the structures of the ternary complex are in agreement with those obtained with the intact trimer. Residues within 4 Angstroms of the variable domains of each Fab are depicted in spheres. Those residues include all residues identified by alanine scanning. Residues buried by IMM20253 interaction with the RBD are conserved in the Omicron variant and provide additional basis for continued activity of this antibody against the variant.
- Example 18 Phase 1 Study to Evaluate the Safety, Pharmacokinetics, and Viral Clearance of Single Ascending Doses of IMM-BCP-01 Administered Intravenously in Adults with Mild to Moderate COVID
- This is a randomized, double-blind, placebo-controlled Phase 1 study to evaluate the safety, PK, and viral clearance of single ascending doses of IV IMM-BCP-01 in subjects with mild to moderate COVID-19 caused by infection with SARS-CoV-2 and/or its variants.
- the primary objective of this study is to evaluate the safety and tolerability of intravenous (IV) IMM-BCP-01 in subjects with mild to moderate COVID-19 through Day 28.
- the secondary objectives of the study are to: Determine PK and evaluate viral clearance after single ascending doses of IV IMM-BCP-01 in subjects with mild to moderate COVID-19 through Day 28. Evaluate the safety and tolerability, determine PK, and evaluate viral clearance of single ascending doses of IV IMM-BCP-01 in subjects with mild to moderate COVID-19 through Week 12.
- the study consists of a screening period of up to 36 hours, a treatment period of one day, two further clinic visits, four visits that take place either in the clinic or at the subject's home, six virtual visits (telephone or video), and an end-of-study visit 12 weeks (+/- 5 days) after study drug dosing. The total duration of a subject's participation is approximately 90 days.
- Subjects presenting at the clinic with signs and symptoms of mild to moderate COVID-19 and agreeing to participate in the study are screened, and if deemed eligible for the study, are randomized (2:1) to receive a single IV dose of IMM-BCP-01 or placebo on Day 1.
- Subjects at risk of severe disease and those who have been vaccinated against COVID-19 within 6 weeks prior to screening or who have received monoclonal antibodies against SARS-CoV-2 and/or COVID-19 convalescent plasma at any time are not eligible.
- Subjects are randomized to receive IMM-BCP-01 or placebo.
- Table 10 Phase 1 Trial Design - - Drug: Placebo Placebo matching single dose of IMM-BCP- - Primary Outcome Measure is: 1.
- TEAEs Treatment Emergent Adverse Events
- TEAEs include clinical laboratory values, standard 12-lead ECGs, vital signs, pulse oximetry Secondary Outcome Measures are: 1. Incidence and severity of Treatment Emergent Adverse Events (TEAEs) (Time Frame: up to 12 weeks) 2. PK parameters measured by maximum observed concentration, time to maximum observed concentration, terminal elimination half-life, clearance, volume of distribution (Time frame: up to 28 days) 3. PK parameters measured by maximum observed concentration, time to maximum observed concentration, terminal elimination half-life, clearance, volume of distribution (Time frame: up to 12 weeks) 4. Anti-drug antibody change from baseline (Time frame: 28 days) 5.
- Anti-drug antibody change from baseline (Time frame: 12 weeks) 6. Viral clearance change from baseline (Time frame: 28 days) 7. Viral clearance change from baseline (Time frame: 12 weeks)
- Inclusion Criteria are: 1. Male or female 18 to 50 years of age, inclusive, at the time of signing the informed consent. 2. Subjects must have mild to moderate COVID-19 with symptom onset within 5 days prior to study drug administration (see Appendix Error! Reference source not found. for Food and Drug Administration [FDA] severity guidance). Subjects whose symptoms began >5 days (i.e. ⁇ 120 hours) prior to dosing or whose time of symptom onset cannot be accurately assessed are not eligible. 3.
- Subjects must have at least 2 of the following COVID-19 symptoms: fever, cough, sore throat, rhinorrhea, malaise, headache, muscle pain, nausea, vomiting, diarrhea, and loss of taste or smell, or other symptoms that the Principal Investigator judges to be referrable to COVID-19. 4. Subjects must be able to maintain oxygen saturation (SpO 2 ) ⁇ 94% on room air (no supplemental oxygen). 5. Body mass index ⁇ 18.0 and ⁇ 30.0 kg/m 2 . 6. Body weight ⁇ 40 kg at screening. 7. Sexually active subjects of reproductive potential must agree to use highly effective contraception from signing of the informed consent through 90 days after infusion of the study drug (see Section Error! Reference source not found.). 8.
- Subjects must have been in generally good health, as judged by the Principal Investigator, prior to onset of current COVID-19 illness, with no clinically significant medical history. 10. Subjects must be without clinically significant abnormalities as assessed by review of medical and surgical history, physical examination, vital signs measurement, ECG, and laboratory evaluations conducted at screening. Exclusion criteria are: 1. Has one or more symptoms suggestive of more severe illness with COVID-19 (see Appendix Error! Reference source not found. for FDA severity guidance) and/or requires hospitalization. 2. Is asymptomatic at screening or randomization, regardless of a positive COVID- 19 test. 3.
- Is at increased risk of severe COVID-19 for any reason including but not limited to: cancer (basal cell carcinoma and prostate carcinoma in situ [Gleason ⁇ 6] are acceptable), chronic kidney disease, chronic obstructive pulmonary disease, heart condition (congestive heart failure II, III and IV as per New York Heart Association: coronary disease and any other cardiac condition that imposes high risk of developing severe COVID-19), immunocompromised state from solid organ transplant, sickle cell disease, or other condition, autoimmune disease, use of immunosuppressants (including high doses of systemic corticosteroids), type 1 or type 2 diabetes mellitus,current or prior history of smoking or vaping any product, including nicotine or THC. 4. Has any active infection, other than the underlying COVID-19. 5.
- cancer basic cell carcinoma and prostate carcinoma in situ [Gleason ⁇ 6] are acceptable
- chronic kidney disease chronic obstructive pulmonary disease
- heart condition congestive heart failure II, III and IV as per New York Heart Association: coronary disease and any other cardiac condition
- History or suspicion of excessive alcohol use (defined as drinking on average 14 drinks a week for males and 7 drinks a week for females) or of binge drinking (defined as 4 drinks on any day for males and 3 drinks on any day for females, for 5 or more days in the past month) 13. History of substance abuse or current use of any drugs of abuse 14.
- Antibody-dependent cellular cytotoxicity-mediating antibodies from an HIV-1 vaccine efficacy trial target multiple epitopes and preferentially use the VH1 gene family.
- the coronavirus is mutating — does it matter? Nature.2020;585.
- Puligedda R.D., Kouiavskaia, D., Adekar, S.P., Sharma, R., Devi Kattala, C., Rezapkin, G., Bidzhieva, B., Dessain, S.K, and Chumakov, K. Human monoclonal antibodies that neutralize vaccine and wild-type poliovirus strains. Antiviral Res. 2014;108: 36–43.
- TNT003 an inhibitor of the serine protease C1s, prevents complement activation induced by cold agglutinins.
Landscapes
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Provided herein are methods for administering antibodies to treat SARS-CoV-2 infections in a subject. Also provided herein are compositions comprising one or more antibodies, methods of treatment comprising administering one or more antibodies, and kits comprising one or more antibodies.
Description
Methods of Administering Antibodies Against SARs-CoV-2 Spike Protein Cross-reference to related applications [0001] This application claims the priority benefit of U.S. Provisional Application No. 63/178,848 filed April 23, 2021, U.S. Provisional Application No. 63/220,881 filed July 12, 2021, U.S. Provisional Application No. 63/236,479 filed August 24, 2021, U.S. Provisional Application No. 63/270,665 filed October 22, 2021, U.S. Provisional Application No. 63/284,963 filed December 1, 2021, and PCT/US2022/070026 filed Janury 4, 2022, the disclosure of each of which is incorporated herein by reference in its entirety. Sequence listing [0002] The content of the following submission on ASCII text file is incorporated herein by reference in its entirety: a computer readable form (CRF) of the Sequence Listing (file name: 224192000240SEQLIST.TXT, date recorded: April 20, 2022, size: 98,706 bytes). Field of the Invention [0003] The present invention relates to methods of administering antibodies used for treatment of SARS-CoV-2 infections. Background [0004] Since its emergence in Wuhan, China in late 2019 [Zhou et al. 2020], the prolonged spread of the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) virus has resulted in one of the most devastating global health challenges of the last century [Nature Editorial 2020, Morens & Fauci 2020, Chakraborty & Maity 2020]. With greater than 289 million confirmed cases and over 5.43 million deaths world-wide, the virus continues to pose an extraordinary challenge to the scientific community, consequently becoming an unprecedented socio-economic disaster and burdening healthcare systems around the world. Infection with SARS-CoV-2 results in a myriad of pathologies [Berlin et al. 2020] collectively referred to as COVID-19 [3]. While a majority of individuals who become infected with the virus are capable of generating a productive anti-viral response, for many their anti-viral humoral response will not be sufficient to shield them from a potentially deadly infection. Therefore, as the global community braces for the next Spike in infection and mortality rates, the urgency to develop effective therapeutics recapitulating the productive anti-viral response to combat the swelling health crisis has never been greater.
[0005] By the end of October 2020, a mere 11 months after the virus was first identified, the Clinicaltrials.gov database listed more than 3500 distinct clinical trial activities directed at patients infected with SARS-CoV-2. There is significant diversity among these efforts, from the assessment of existing drugs to the use of convalescent plasma from recovered patients, to the use of specific vaccines and antibodies directed at the viral S protein. It is not yet apparent that there will be a single approach that will prove to be uniformly effective at preventing viral infections or accelerating viral clearance in all groups of COVID-19 patients. [0006] SARS-CoV-2 genomic RNA contains a large viral replicase gene, genes encoding non-structural proteins at its 5’ end, and a region encoding four major structural and multiple accessory proteins at the 3’ end. Structural proteins include Spike or Surface glycoprotein (S), Membrane protein (M), Envelope protein (E) and Nucleocapsid protein (N) [Fehr & Perlman 2015]. The membrane surface glycoprotein S consists of two subunits, S1 and S2, that mediate viral binding to the host receptor ACE2 and fusion with the host cell membrane, respectively. The S1 subunit contains the receptor binding domain (RBD) that directly interacts with ACE2 and is a target of multiple neutralizing antibodies currently in clinical trials [Lan et al. 2020, Robbiani et al. 2020]. The vast majority of the ongoing efforts are all targeting the S protein. Both passive (therapeutic antibodies) and active (vaccine) approaches directed at S protein are expected to promote virus neutralization, that is, inhibition of viral entry into healthy cells. Unfortunately, mutations in S protein have arisen, including ones the Center for Disease Control (CDC) has identified in a series of viral variants for which there is evidence of an increase in transmissibility, increased levels of hospitalization and/or deaths, significant reduction in neutralization by antibodies generated during previous infections or vaccinations, reduced effectiveness of treatments or vaccines, or diagnostic detection failures. These virus variants are considered Variants of Concern include Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV- 2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS- CoV-2. Additionally, the CDC has identified Variants of Interests, which are defined as having specific genetic markers that are predicted to affect transmission, diagnostics, therapeutics, or immune escape. Data also demonstrates these variants of interest were the cause of an increased proportion of cases or outbreak, but they are of limited prevalence in
the US or other countries (https://www.cdc.gov/coronavirus/2019-ncov/cases- updates/variant-surveillance/variant-info.html#Concern). [0007] Studies are described herein that elucidate the memory B cell antibody response in convalescent patients, using an approach that enables the generation of large, stable hybridoma libraries from primary human B cells. This approach was previously used to identify a panel of monoclonal antibodies from convalescent patients infected with natural polio virus (PV), oral PV-vaccinated and inactivated PV-boosted healthy subjects [Puligedda et al. 2014, Puligedda et al. 2017, Puligedda et al. 2020], and, most recently, an anti-amyloid antibody with the anti-biofilm activity [Tursi et al. 2020] from a hybridoma library generated with memory B cells from an Alzheimer’s Disease patient [Levites et al. 2015]. [0008] Hybridoma libraries were generated from the memory B cells of convalescent COVID-19 blood donors who were eligible to donate convalescent plasma based upon their high titer of IgG antibodies against the SARS-CoV-2 virus. Monoclonal antibodies derived from those libraries were selected on the basis of their selective binding to one of multiple SARS-CoV-2 proteins used as targets in both cell-based and soluble protein-based screens. Characterization of these antibodies revealed broad responses to diverse viral antigens. Fewer than half of the antibodies were directed at S protein, while the remainder were directed at other viral proteins including N and ORF-encoded proteins. Even though the antibodies were directed at highly diverse SARS-CoV-2 antigens, they were generally characterized as having variable levels of somatic hypermutation (SHM) and a diversity of VH and VL gene usage. Functional properties of anti-Spike antibodies were successfully confirmed against reference strains (e.g., USA/WA_CDC-WA1/2020), as well as multiple variants including the CDC variants of concern, in series of tests ranging from in vitro neutralization of both pseudovirus and live virus isolates to in vivo neutralization activity in a hamster model of COVID-19. Three anti-Spike antibodies were identified that when mixed together in a cocktail exhibited combinatorial effects against those variants. These studies indicate that an unbiased interrogation of COVID-19 patient B cell repertoires is an effective approach to identifying specific antiviral antibodies and antibody mixtures with the desired binding and functional properties. Antibodies identified and characterized in this manner could be recombinantly produced to yield therapeutic or prophylactic products to address the COVID-19 pandemic. The rapidity with which antibodies from convalescent patients can be identified and characterized suggests that this platform could be a useful component of a rapid response to future pandemics.
Summary of the Invention [0009] Provided herein is are methods of treating a SARS-CoV-2 infection in an immunocmpromised subject comprising administering to the subject at least first and send antibodies or antigen binding fragmetns thereof that specifically bind to distinct epitopes of a Spike protein a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein. In some embodiments, the antibodies disclosed herein are especially effective for treating immunocompromised individuals due to synergistic effects and their ability to fix complement. In some embodiments, the antibodies are able to effectively fix complement due to their specific binding sites on the Spike protein and orientation of binding. In some embodiments, the methods provided herein are effective for viral clearance in immunocompromised patients. In some embodiments, the methods provided herein are effective for treatment of individual who have decreased adaptive immune system function. [0010] Also provided herein are methods of treating a SARS-CoV-2 infection in a subject comprising administering about 200 mg, about 400 mg, about 600 mg, or about 800 mg of one, two, three, or four antibodies or antigen binding fragments provided herein. Brief Description of the Drawings [0011] FIG. 1 depicts the breadth of antibodies isolated against a range of SARS-CoV-2 viral proteins. [0012] FIG. 2 depicts the epitopes for IMM20190 (black sphere, left panel), IMM20184 (light gray, left panel), and IMM20253 (dark gray, right panel), as determined by alanine scanning. ACE2 binding site and two other residues [N343 represents a site of potential glycosylation and E465 (Greaney et al 2021)]. [0013] FIGS. 3A-3D depict in vitro pseudovirus neutralization activity of PR193_00018 (IMM20184), PR194_00232 (IMM20190), PR200_00622 (IMM20253), and PR190_00255 (IMM20279) against pseudoviruses expressing four different variations of Spike. [0014] FIGS. 4A-B depict in vitro neutralizing activity of the identified anti-Spike antibody PR194_00232 against (FIG. 4A) the live virus isolate SAR-CoV-2/human/USA/WA_CDC- WA1/2020, which contains a reference Spike protein. (FIG.4B) depicts in vitro neutralizing activity of the identified anti-Spike antibody PR194_00232 against the live virus isolate
SAR-CoV-2/human/Germany/BavPAt 1/2020, which contains a mutated Spike protein (D614G). [0015] FIGS. 5A-5C. depict in vitro neutralization activity of identified anti-Spike antibodies, as noted, against pseudovirus expressing the Spike protein from either the U.K. (B.1.1.7) or South African (B.1.351) variant of SARS-CoV-2 [0016] FIGS. 6A-6C depict the concentration-dependent ability of the triple antibody cocktail (IMM20184/IMM20190/IMM20253) and the two antibody cocktail (IMM20184/IMM20253) to neutralize pseudoviruses expressing the reference (WA1/2020), alpha/UK, beta/S. African, gamma/Brazilian, or Epsilon/Californian Spike proteins. [0017] FIGS. 7A-7C depict the combinatorial activity of the IMM20184 / IMM20190 / IMM20253 triple combination against pseudoviruses expressing the Spike protein from the reference strain (WA_CDC-WA1/2020), U.K. (B.1.1.7/alpha) ,South African (B.1.351/beta) , Californian (B.1.429/epsilon), or Brazilian (P.1/P.2/gamma) variants of SARS-CoV-2 (FIG. 7A)and the live virus BavPat1/2020 that contains the D614G mutation (FIG. 7B). Dark gray area represent regions of synergy. Bottom portion of the figure lists the overall and peak HSA scores against each strain. [0018] FIG. 8 depicts the crystal structure of the SARS-CoV-2 RBD (RCSB PDB: 7A97) represented in cartoon with the epitopes of IMM20184, IMM20190, and IMM20253 depicted in black spheres. The locations of residues L452, T478, and E484, which are mutated in B.1.617.1/kappa (L452R E484Q) and B.1.617.2/delta (L452R T478K), are depicted in gray spheres. The residues mutated in the delta and kappa SARS-CoV-2 variants lie outside of the IMM-20184, IMM20190, and IMM20253 epitopes. [0019] FIG. 9 depicts the in vitro neutralization activity of triple antibody cocktail (IMM20184 / IMM20190 / IMM20253) against pseudovirus expressing either the WA1/2020 (REF), B1.617(L452R, E484Q), B.1.617.2 (delta), or lambda (C.37) Spike proteins. [0020] FIG. 10 depicts in vitro neutralization activity triple antibody cocktail (IMM20184 / IMM20190 / IMM20253) against pseudovirus expressing the Spike protein from the reference strain (WA_CDC-WA1/2020), D614G, B.1.1.7 (alpha/U.K.), B.1.351 (beta/S. African), P.1/P.2 (gamma/Brazilian), B.1.429 (Epsilon/Californian), B.1.617.1 (L452/E484Q), B.1.617.2 (delta/India), B.1.617.2 Ay.2 (Delta Plus), or C.37 (lambda) variants of SARS-CoV-2. [0021] FIGS. 11A-11D depict in vitro neutralization activity of the triple (IMM20184 / IMM20190 / IMM20253) and double (IMM20184 / IMM20253) combinations of antibodies against three live virus strains as measured by plaque forming assays; the reference strain
(WA_CDC-WA1/2020)(FIG. 11A), U.K. (B.1.1.7/alpha) (FIG. 11B), South African (B.1.351/beta) (FIG. 11C). [0022] FIG. 12 depicts lung titer per gram tissue, as measured by plaque forming units, as an assessment of in vivo activity of the identified anti-Spike antibody PR194_00232, alone and in combination with either PR193_00018 or PR200_00622, against the live virus isolate SAR-CoV-2/human/USA/WA_CDC-WA1/2020, when dosed in the prophylactic setting. [0023] FIG. 13 depicts lung titer per gram tissue, as measured by plaque forming units, as an assessment of in vivo activity of various combinations of anti-Spike antibodies, dosed in a therapeutic setting, in the hamster model of COVID-19. Error bars represent the median +/- 95% CI. [0024] FIG. 14 depicts lung titer per gram tissue, measured by TCID50 assays, as an assessment of in vivo activity of the triple combination ofIMM20184/IMM20190/IMM20253, at two different ratios, dosed in a therapeutic setting, in hamster model of COVID-19. Error bars represent the median +/- interquartile range. [0025] FIG. 15 depicts correlation of viral lung titer per gram tissue, as measured by TCID50 assays, plotted against serum concentration of triple antibody cocktail (IMM20184/190/253, 1:1:1 ratio) at day 4 post administration of antibodies in a therapeutic setting. Antibody cocktail was administered at three dose levels, 0.1 mg each, 0.2 mg each, 0.3 mg each. [0026] FIGS. 16A-16B depict lung titer per gram of tissue, as measured by plaque forming units. Hamsters infected with either the (FIG. 16A) WA1/2020 or (FIG. 16B) Beta isolate were treated in prophylactic setting with a dose response of triple antibody cocktail (IMM20184/IMM20190/IMM20253, 1:1:1). [0027] FIG. 17 depicts anti-viral activity of a triple antibody cocktail (IMM20184/190/253, 1:1:1 ratio), dosed at 0.1mg each, into Syrian hamsters that were inoculated with either 3.3x104 or 3.3x105 virus. Viral titers in lungs were assessed four days post-inoculation via TCID50 assays. Antibody was administered in a therapeutic setting. [0028] FIGS. 18A-18C depict the ability of IMM20184, IMM20190, and IMM20253 to bind to the isolated RBD and intact trimer of SARS-CoV-2 reference strain (WA1/2020) as measured by surface plasmon resonance. [0029] FIGS. 19A-19C depict the ability of IMM20184, IMM20190, and IMM20253 to block binding of ACE2 to isolated RBD corresponding to WA1/2020 (FIG. 19A), Alpha (FIG. 19B), Beta viral isolate (FIG. 19C).
[0030] FIGS. 20A-20C depict in vitro complement fixation activity of PR193_00018 (IMM20184), PR194_00190 (IMM20190), PR200_00253 (IMM20253), the two antibody cocktail of IMM20184/IMM20253, and the triple antibody cocktail (IMM20184/IMM20190/IMM20253) relative to control antibodies when assessed at defined (FIG. 20A-20B) concentrations and in a concentration-dependent (FIG. 20C) manners. [0031] FIG. 21 depicts in vitro phagocytosis activity of IMM20184, IMM20190, IMM20253, the IMM20184/IMM20253 two -antibody cocktail, and IMM-BCP-01 (three antibody cocktail) relative to isotype control antibodies when assessed across a range of concentrations. [0032] FIG. 22 depicts antibody-dependent cellular cytotoxicity activity of IMM20184, IMM20190, IMM20253, the two antibody cocktail of IMM20184/IMM20253, and the triple antibody cocktail (IMM20184/IMM20190/IMM20253; IMM-BCP-01) relative to isotype control antibodies when assessed in a concentration-dependent manner. [0033] FIGS. 23A-23B depict time-dependent conformational change in Spike protein, as measured by dynamic light scattering, upon binding of IMM20253 or IMM20190. [0034] FIG. 24 depicts time-dependent protease digestion of Spike and Spike in complex with ACE2, IMM20253, or IMM20190. [0035] FIGS. 25A-25C depict the combinatorial neutralization activity of IMM20253 in combination with IMM20184, REGN987, or REGN933 against pseudoviruses expressing the Spike protein from the Delta Plus (B.1.617.2 ay1/2) variant. Dark grey area represent regions of synergy. Bottom portion of the figure lists the overall HSA scores for each combination. [0036] FIG. 26 depicts the combinatorial neutralization activity of IMM20253 in combination with S309 against pseudoviruses expressing the Spike protein from the Delta Plus (B.1.617.2 ay1/2) variant. Grey areas represent areas of antagonism between the two antibodies. Bottom portion of the figure lists the overall HSA score for the combination. [0037] FIGS. 27A-27D depict the in vitro binding activity of individual antibodies (FIG. 27A) IMM20184, (FIG. 27B) IMM20190, (FIG. 27C) IMM20253, and (FIG. 27D) IMM20279 to the SARS-CoV-2 Spike-RBD Omicron variant relative to the reference strain. [0038] FIG. 28 depicts the in vitro neutralization activity of the IMM20253 antibody against pseudovirus expressing the Spike protein from the reference strain (SARS-CoV- 2/human/USA/WA CDC-WA1/2020), DG14G (SARS-CoV-2/human/Germany/BavPat 1/2020), B.1.351 (beta/S. African), B.1.617.2 Ay.2 (Delta Plus) and B.1.1.529 (Omicron) variants of SARS-CoV-2.
[0039] FIG. 29 depicts the in vitro neutralization activity of the IMM20253 antibody against pseudovirus expressing the Spike protein from the BA.1 and BA.2 sub-lineages of the B.1.1.529 (Omicron) variant of SARS-CoV-2. [0040] FIGS. 30A-30B depict the 3 Fab fragments of IMM20190 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL. SARS-CoV-2 trimer is depicted in light grey cartoon. IMM20190 Fab fragments are depicted in black cartoon. FIG. 30A is the side view and FIG. 30B is the top view. [0041] FIGS. 31A-31B depict the 3 Fab fragments of IMM20184 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL. SARS-CoV-2 trimer is depicted in light grey cartoon. IMM20184 Fab fragments are depicted in black cartoon. FIG. 31A is the side view and FIG. 31B is the top view. [0042] FIGS. 32A-32B depict a single Fab fragment of IMM20253 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL. SARS-CoV-2 trimer is depicted in light grey cartoon. IMM20253 Fab fragments are depicted in black cartoon. FIG. 32A is the side view and FIG. 32B is the top view. [0043] FIG. 33 depicts a 3.9 Angstrom structure of the variable domains of IMM20184 and IMM20253 bound to an isolated receptor binding domain (RBD) corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC-WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL. SARS-CoV-2 trimer is depicted in light grey cartoon. IMM20184 variable domains are depicted in black cartoon. IMM20253 variable domains are depicted in dark grey cartoon. RBD residues predicted to be within 4 Angstroms of IMM20184 are visualized in black spheres. RBD residues predicted to be within 4 Angstroms of IMM20253 are depicted in dark grey spheres. [0044] FIG. 34A-34B depict an isolated receptor binding domain (RBD) corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC-WA1/2020) of SARS-CoV-2 with (FIG. 34A) predicted IMM20184 contact residues depicted in black spheres and (FIG. 34B) predicted IMM20253 contact residues depicted in dark grey spheres. [0045] FIGS. 35A-35B depict the 3 Fab fragments of IMM20279 bound to an intact spike trimer corresponding to the reference strain (SARS-CoV-2/human/USA/WA CDC- WA1/2020) of SARS-CoV-2 solved by cryoEM and visualized using PyMOL. SARS-CoV-2
trimer is depicted in light grey cartoon. IMM20190 Fab fragments are depicted in black cartoon. (FIG. 35A) side view and (FIG. 35B) top view. [0046] FIG. 36 depicts lung titers of hamsters dosed prophylactically with IMM20253, IMM20253/IMM20184 or IMM20253/IMM20279 one day prior to inoculation with Omicron BA.1. Detailed Description [0047] Methods of treating the severe acute respiratory syndrome coronavirus 2 (SARS- CoV-2) virus are described herein. In some embodiments, the methods comprise treating an immunocompromised subject. In some embodiments, the subject has a high risk for contracting SARS-CoV2. In some embodiments, the subject has a suppressed immune system. In some embodiments, the individual is being treated with an immunosuppressive agent. These antibodies may be used to neutralize SARS-CoV-2 by preventing the virus from infecting new host cells. Therefore, the inventions disclosed here also relate to pharmaceutical compositions that contain one or more antibodies of the invention, as well as relate to methods of preventing or treating a SARS-CoV-2 infection in a subject in need thereof. Accordingly, the inventions disclosed herein also relate to methods of administering antibody compositions of the invention to a subject in need thereof. [0048] In some embodiments, the antibodies, compositions, and kits provided herein are especially effective for treating and/or preventing SARS-CoV-2 due to particular novel properties. In some embodiments, provided herein are antibodies that bind to the Spike protein at multiple different locations, for example at multiple non-overlapping epitopes. This is beneficial because SARS-CoV-2 variants may have one or more mutations in the Spike protein to evade the immune system. Thus providing multiple antibodies that bind to multiple different locations in the Spike protein allows binding and neutralization of such variants. [0049] Moreover, some of the antibodies provided herein bind to an ACE2 binding site in the Spike protein, while others bind outside the ACE2 binding site. Without being bound by theory, providing multiple antibodies, some of which target the ACE2 binding site and some of which target regions outside of the ACE2 binding site may combine to provide more effective treatment for SARS-CoV-2. [0050] Accordingly, in some embodiments, provided herein is a method of treating or preventing SARS-CoV-2 comprising administering multiple antibodies that bind to non-
overlapping epitopes on the Spike protein. In some embodiments, the method comprises administering an antibody that binds to an ACE2 binding site of the Spike protein and an antibody that binds to an epitope outside of the ACE2 binding site of the Spike protein. In some embodiments, the method comprises administering an antibody that neutralizes SARS- CoV-2 through an ACE2-dependent mechanism and an antibody that neutralizes SARS-CoV- 2 through an ACE2-independent mechanism. [0051] As used herein “dose” or “dosage” refers to the amount of a single agent (such as an antibody). The term “total dose” is used to refer to a total amount of multiple agents that are delivered as a combination therapy (for example, an antibody cocktail). By way of example, an antibody cocktail comprising three antibodies each delivered at a dose of 200 mg is considered to be administered at a total dose of 600 mg. The multiple agents in such a combination therapy can be administered in the same or different compositions and simultaneously or sequentially. Methods of Treatment [0052] Provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering an antibody or antigen binding fragment thereof provided herein to the subject. In some embodiments, the method comprises administering one antibody provided herein. In some embodiments, the method comprises administering two or more antibodies provided herein. In some embodiments, the method comprises administering three or more, or four or more antibodies provided herein. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. [0053] In some embodiments, the methods provided herein comprise treating an immunocompromised subject with one or more antibodies provided herein (such as two, three, or four antibodies). In certain embodiments, the antibodies provided herein are especially advantageous for treatment of immunocompromised individuals because they facilitate viral clearance from the subject. In some embodiments, the antibodies are able to engage in complement fixation. In some embodiments, the antibodies provided herein bind to a Spike protein in an orientation that allows for complement fixation. In some embodiments, the antibodies bind to a Spike protein in an orientation that provides synergy
between the Fc and the antigen binding domain. In some embodiments, the antibodies effectively neutralize SARS-CoV-2. In some embodiments, the subject is administered one, two, three, or four antibodies provided herein to treat a SARS-CoV-2 infection. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. [0054] In some embodiments, the immunocompromised subject has an impaired immune system. In some embodiments, the immunocompromised subject has decreased resistance to an infection, such as SARS-CoV-2 infection. In some embodiments, the subject has one or more markers of low immune function. In some embodiments, the number or activity of T, B, and/or NK cells in the subject is below normal. In some embodiments, one or more functional markers of immune function is altered in the immunocompromised subject. In some embodiments, the subject is administered one, two, three, or four antibodies or antigen binding fragments thereof provided herein to treat a SARS-CoV-2 infection. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. [0055] In some embodiments, the subject has an immunodeficiency disease. In some embodiments, the immunodeficiency disease is a primary immunodeficiency disease. In some embodiments, the immunodeficiency results in decreased antibody production and/or decrease of amount or activity of one or more types of immune cells. [0056] In some embodiments, the immunodeficiency disease is an acquired immunodeficiency condition. In some embodiments, the acquired immunodeficiency condition is caused by another disease or a treatment of another disease. In some embodiments, the subject has received a solid organ transplant and being treated with one or more immunosuppressants. In some embodiments, the subject has received a bone marrow transplant. In some embodiments, the subject is a cancer patient undergoing active chemotherapy. In some embodiments, the subject is undergoing treatment with an EGFR inhibitor, an ALK inhibitor, a ROS1 inhibitor, a BRAF inhibitor, a RET inhibitor, a MET inhibitor, a NTRK inhibitor, a BTK inhibitor, a PI3K inhibitor, or a BCL-2 inhibitor and or
radiation therapy. In some embodiments, the subject is on dialysis. In some embodiments, th e subject has end stage renal disease and is on dialysis. In some embodiments, the individual has multiple sclerosis and is receiving anti-CD20 therapy. [0057] In some embodiments, the subject has an inflammatory disease. In some embodiments, the subject has an inflammatory disease and is being treated with a maintenance therapy. In some embodiments, the subject has an inflammatory disease and is being treated with an immunosuppressant maintenance therapy. In some embodiments, the inflammatory disease is arthritis, psoriasis, or Crohn’s disease. In some embodiments, the subject has rheumatoid arthritis. In some embodiments, the inflammatory disease is an auto inflammatory disease. In some embodiments, the inflammatory disease is selected from the group consisting of ankylosing spondylitis arthritis, osteoarthritis, rheumatoid arthritis (RA), psoriatic arthritis, asthma, atherosclerosis, Crohn's disease, colitis, dermatitis, diverticulitis, fibromyalgia, hepatitis, irritable bowel syndrome (IBS), systemic lupus erythematous (SLE), nephritis, Parkinson's disease, ulcerative colitis, Cryopyrin-Associated Periodic Syndromes (CAPS) including Familial Cold Autoinflammatory Syndrome and Muckle-Wells Syndrome, Systemic Juvenile Idiopathic Arthritis, giant cell arteritis, Paroxysmal nocturnal hemoglobinuria (PNH), atypical hemolytic uremic syndrome (a-HUS), vasculitis. In some embodiments, the subject is receiving steroids, sulfasalazine, methotrexate, mycophenolate mofetil (IMPDH inhibitor), azathioprine, Anti-IL-1 biologics (anakinra (Kineret®), canakinumab (Ilaris®), rilonacept (Arcalyst®), a TNFα inhibitors [ Infliximab (Remicade®), Adalimumab (Humira®), Golimumab (Simponi®), Etanercept (Enbrel®), Certolizumab (Cimzia®)], Anti-IL-6 Biologics: Tocilizumab (Actemra®), Sarilumab (Kevzara®); Complement inhibitors: Eculizumab, Anti-CD20 Biologics: Rituximab (Rituxan), BLyS inhibitor (Belimumab (Benlysta®)), Calcineurin inhibitor (cyclosporin®) CTLA-4/CD28 modulator; Abatacept (Orencia®), an anti-IL-17 biologics; Secukinumab (Cosentyx®), Ixekizumab (Taltz®), Brodalumab (Siliq®), a JAK inhibitors:Tofacitinib (Xeljanz®), Upadacitinib (Rinvoq®), Baricitinib (Olumiant®) . In some embodiments, the antibodies or antigen binding fragments thereof that bind to a Spike protein comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the subject is administered one, two, three, or four antibodies or antigen binding fragments thereof provided herein to treat a SARS-CoV-2 infection. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each.
[0058] In some embodiments, provided herein are methods of treating a subject who is at high risk for contracting SARS-CoV-2 comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject. In some embodiments, the subject has a high risk for contracting severe SARS-CoV-2. In some embodiments, the subject is immunocompromised. In some embodiments, the subject has one or more risk factors for contracting SARS-CoV-2. In some embodiments, the subject has one or more risk factors such as, old age, serious heart or lung conditions, weakened immune system, obesity, or diabetes. In some embodiments, the individual is obese. In some embodiments, the individual is pregnant. In some embodiments, the subject has a high risk for contracting other serious respiratory illnesses, such as the flu. In some embodiments, the individual is age 65 or older. In some embodiments, the individual has COPD, lung cancer, cystic fibrosis, pulmonary fibrosis, asthma, pulmonary hypertension, pulmonary embolism, cardiomyopathy, congenital heart disease, heart failure, coronary artery disease, Type 1 diabetes, Type 2 diabetes, a high body mass index, HIV/AIDS, chronic kidney or liver diseases, sickle cell or anemia. In some embodiments, the subject has chronic kidney disease, hypertension, severe neurodevelopment disorders, dementia, Alzheimers disease, multiple co- morbidities, and/or is a nursing home resident. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. [0059] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject that has cancer comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject. In some embodiments, the subject is being treated with more chemotherapeutic agents. In some embodiments, the subject has recently received treatment with one or more chemotherapeutic agents. In some embodiments, the subject is receiving or has recently received radiation therapy. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. [0060] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject that has received a solid organ transplant comprising administering one,
two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject. In some embodiments, the subject is on one or more immunosuppressive drugs. In some embodiments, the subject has recently received one or more immunosuppressive drugs. In some embodiments, the subject is receiving or has recently received an antirejection drug. In some embodiments, the subject has received a calcineurin inhibitor, an antiproliferative agent, a mTOR inhibitor, and/or a steroid. In some embodiments, the subject is receiving or has recently received corticosteroid treatment. In some embodiments, the subject is being treated or has recently received treatment with an IMPDH inhibitor. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. [0061] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject, wherein the antibodies or antigen binding fragments thereof neutralize the SARS-CoV-2 virus. In some embodiments, the antibodies block viral infection of human cells. In some embodiments, the antibodies are able to clear SARS-CoV2 viral particles. In some embodiments, viral clearance is observed within 28 days, or within 12 weeks of administering the antibodies. In some embodiments, viral load is decreased upon administration of the antibodies or antigen binding fragments thereof. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the subject is immunocompromised. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. [0062] Also provided herein are method of treating subjects with moderate or mild SARS- CoV-2 infections comprising administering one, two, three, or four antibodies or antigen binding fragments thereof provided herein to the subject. In some embodiments, the subject does not have severe COVID-19, is not at increased risk of severe COVID-19, and does not have an active infection. In some embodiments, the subject does not have cancer (basal cell carcinoma and prostate carcinoma in situ [Gleason ≤ 6] are acceptable), chronic kidney disease, chronic obstructive pulmonary disease, heart condition (congestive heart failure II, III and IV as per New York Heart Association: coronary disease and any other cardiac condition that imposes high risk of developing severe COVID-19), immunocompromised
state from solid organ transplant, sickle cell disease, or other condition, autoimmune disease, use of immunosuppressants (including high doses of systemic corticosteroids), type 1 or type 2 diabetes mellitus, current or prior history of smoking or vaping any product, including nicotine or THC. In some embodiments, the individual has not received a previous therapy to treat SARS-CoV-2. In some embodiments, the antibodies comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the subject is immunocompromised. In some embodiments, the one, two, three, or four antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg each. In some embodiments, the method comprises administering a single dose of the antibody or antigen binding fragment thereof. [0063] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein. In some embodiments, the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1. In some embodiments, the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibody or antigen binding fragment thereof. [0064] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein. In some embodiments, the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1. In some embodiments, the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibody or antigen binding fragment thereof.
[0065] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 mg of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein. In some embodiments, the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1. In some embodiments, the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1 In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibody or antigen binding fragment thereof. [0066] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of an antibody or antigen binding fragment thereof provided herein that binds to a SARS-CoV-2 Spike protein. In some embodiments, the antibody or antigen binding fragment thereof comprises CDR sequence of an antibody disclosed in Table 1. In some embodiments, the antibody or antigen binding fragment thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibody or antigen binding fragment thereof. [0067] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 400 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof.
[0068] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 800 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0069] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 1200 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0070] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of two antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 1600 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid
organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0071] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 600 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0072] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 mg of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 1200 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0073] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 1800 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table
1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0074] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of three antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 2400 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0075] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 200 mg of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 800 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0076] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 400 mg of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 1600 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table
1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0077] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 600 of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 2400 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0078] In some aspects, provided herein are method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject about 800 mg of four antibodies or antigen binding fragments thereof provided herein that bind to a SARS-CoV-2 Spike protein. In some embodiments, a total dose of 3200 mg of anti-Spike antibodies or antigen binding fragments thereof is administered to the subject. In some embodiments, the antibodies or antigen binding fragments thereof comprise CDR sequence of antibodies disclosed in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprises the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0079] Also provided herein are methods of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first, second, and third antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2. In some embodiments, the first, second, and third antibodies are administered
at a ratio of about 1:1:1. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a flat dose fashion. In some embodiments, the first second, and third antibodies are administered at a total dose of about 600 mg to about 2400 mg. In some embodiments, the antibodies are delivered at a total dose of about 600 mg. In some embodiments the antibodies are delivered at a dose about 200 mg each (e.g. 600 mg total). In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. [0080] Provided herein are methods of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first, second, and third antibodies or antigen binding active fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 1200 mg. In some embodiments, the antibodies are administered at a dose of about 400 mg each (e.g. 1200 mg total). In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1. In some embodiments, the subject is immunocompromised. In some embodiments, the method comprises administering a single dose of the antibodies or antigen binding fragments thereof. In some embodiments, the administration is intravenous administration. [0081] Provided herein are methods of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first, second, and third antibodies or antigen binding active fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 1800 mg. In some embodiments, the antibodies are administered at a dose of about 600 mg each (e.g. 1800 mg total). In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184,
IMM20253, and IMM 20190 as set forth in Table 1. In some embodiments, the subject is immunocompromised. [0082] Provided herein are methods of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first, second, and third antibodies or antigen binding active fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 2400 mg. In some embodiments, the antibodies are administered at a dose of about 800 mg each (e.g. 2400 mg total). In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1. In some embodiments, the subject is immunocompromised. [0083] Provided herein are methods of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first, second, and third antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the antibodies or antigen binding fragments thereof are administered at a total dose of about 3 mg/kg to about 6 mg/kg each. In some embodiments, the antibodies or antigen binding fragments thereof are administered at a total dose of about 3 mg/kg each. In some embodiments, the antibodies or antigen binding fragments thereof are administered at a total dose of about 6 mg/kg each. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, IMM 20190 and/or IMM20279 set forth in Table 1. In some embodiments, the antibodies or antigen binding fragments thereof comprise the CDR sequences of IMM20184, IMM20253, and IMM 20190 as set forth in Table 1. In some embodiments, the subject is immunocompromised. [0084] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof
comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, wherein the first, second and third antibodies are administered at a total dose of about 600 mg. In some embodiments, the first, second, and third antibodies are administered at a dose of about 200 mg each. [0085] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, wherein the first, second and third antibodies or antigen binding fragments thereof are administered at a
total dose of about 1200 mg. In some embodiments, the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 400 mg each. [0086] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, wherein the first, second and third antibodies or antigen binding fragments thereof are delivered at a total dose of about 1800 mg. In some embodiments, the first, second, and third antibodies or antigen binding fragments thereof are delivered at a dose of about 600 mg each. [0087] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3
comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, wherein the first, second and third antibodies or antigen binding fragments thereof are delivered at a total dose of about 2400 mg. In some embodiments, the first, second, and third antibodies or antigen binding fragments thereof are delivered at a dose of about 800 mg each. [0088] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGASWV (SEQ ID NO: 60). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. [0089] In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 as a monotherapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or
more CDR sequences and/or VH and VL sequences of IMM20184 is administered simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20184 is administered sequentially with one or more additional antibodies provided herein. In some aspects, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0090] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the amino acid sequence QKYNSAPGLT (SEQ ID NO: 66). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0091] In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 as a monotherapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 is administered
simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20190 is administered sequentially with one or more additional antibodies provided herein. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0092] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the individual is immunocompromised. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0093] In some embodiments, provided herein is a method of treating or preventing an infection with an Omicron variant of SARS-CoV-2 in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the method further comprises administering a second antibody. In some
embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0094] In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 as a monotherapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 is administered simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20253 is administered sequentially with one or more additional antibodies provided herein. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the individual is immunocompromised. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0095] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an
immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0096] In some embodiments, provided herein is a method of treating or preventing an infection with an Omicron variant of SARS-CoV-2 a subject comprising administering an antibody that binds to a Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the method further comprises administering a second antibody. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0097] In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 as a monotherapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 as a combination therapy. In some embodiments, the method comprises administering an antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 in combination with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 is administered simultaneously with one or more additional antibodies provided herein. In some embodiments, the antibody comprising one or more CDR sequences and/or VH and VL sequences of IMM20279 is administered sequentially with one or more additional antibodies provided herein. In some embodiments, the subject is immunocompromised. In some
embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0098] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0099] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of
QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69), and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0100] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen
binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0101] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0102] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID
NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0103] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of two antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease.
[0104] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of three antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 600 mg, about 1200 mg, about 1800 mg, or about 2400 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously. [0105] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of three
antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 600 mg, about 1200 mg, about 1800 mg, or about 2400 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously. [0106] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of three antibodies that bind to a Spike protein comprising a first antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid
sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 600 mg, about 1200 mg, about 1800 mg, or about 2400 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously. [0107] In some embodiments, provided herein is a method of treating or preventing a SARS-CoV-2 infection in a subject comprising administering a combination of four antibodies that bind to a Spike protein comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID
NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a fourth antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, a total dose of about 800 mg, about 1600 mg, about 2400 mg, or about 3200 mg is administered. In some embodiments, the antibodies or antigen binding fragments thereof are administered in a single composition. In some embodiments, the antibodies or antigen binding fragments are administered intravenously.
[0108] In some embodiments, the method comprises administering one or more of IMM20184, IMM20190, IMM20253, and/or IMM20279 and an additional agent selected from the group consisting of casirivimab, imdevimab, bamlanivimab, etesevimab and sotrovimab. [0109] Also provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering two or more antibodies provided herein to the subject. In some embodiments, the SARS-CoV-2 infection is caused by SARS-CoV-2 variant. In some embodiments, the SARS-CoV-2 is the Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gammma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Episolon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2. In some embodiments, the variant has one or more mutations in a Spike protein. In some embodiments, 3 or more, 4 or more, 5 or more, or 6 or more antibodies provided herein are administered to the subject. In some embodiments, the antibodies comprise a HCVR comprising three heavy chain complementarity determining regions (CDRs), (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 1, and a LCVR comprising three light chain complementarity determining regions (CDRs) (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 2; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 19, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 20; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 7, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID
NO: 8; or a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 23, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 24. ). In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, each antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0110] In some embodiments, provided herein is a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering two or antibodies provided herein. In some embodiments, the Omicron variant includes a combination of mutations in the Spike protein. In some embodiments, the mutations include a large number of changes that form a ring around the ACE2 binding site. Accordingly, in one aspect an advantage of the present methods is the ability to target epitopes within and outside of the ACE2 binding site such that the treatment is effective in variants, such as Omicron which harbor mutations within the Spike protein including in the ACE2 binding site. In some embodiments, the combinations of antibodies provided herein are especially effective for treating variants such as Omicron because they bind to non-overlapping epitopes. In some embodiments, the Omicron variant has one or more modifications that make the Spike protein more susceptible to cleavage and/or cause pre-cleavage of the Spike protein. In some embodiments, the antibodies provided herein, for example IMM20253, are able to bind to pre-cleaved Spike proteins, or Spike proteins that are more susceptible to cleavage with high affinity. [0111] In some embodiments, provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering two or antibodies provided herein. In some embodiments, the Omicron variant includes a combination of mutations in the Spike protein. In some embodiments, the mutations include a large number of changes that form a ring around the ACE2 binding site. Accordingly, in one aspect an advantage of the present methods is the ability to target epitopes within and outside of the ACE2 binding site such that the treatment is effective in variants, such as Omicron which harbor mutations within the Spike protein including in the ACE2 binding site. In some embodiments, the combinations of antibodies provided herein are especially effective for treating variants such as Delta because they bind to non-overlapping epitopes. In some embodiments, the Delta variant has one or
more modifications that make the Spike protein more susceptible to cleavage and/or cause pre-cleavage of the Spike protein. In some embodiments, the antibodies provided herein, for example IMM20253, are able to bind to pre-cleaved Spike proteins, or Spike proteins that are more susceptible to cleavage with high affinity. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. [0112] In some embodiments, provided herein is a method of treating a SARS-CoV-2 infection in a subject comprising administering antibody IMM20184 to the subject. In some embodiments the antibody comprises a HCVR comprising three heavy chain complementarity determining regions (CDRs), (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 1, and a LCVR comprising three light chain complementarity determining regions (CDRs) (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 2. In some embodiments, the antibody comprise a HCDR1, a HCDR2, and anHCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2. In some embodiments, the method comprises administering 2 or more antibodies. In some embodiments, IMM20184 targets an epitope outside the ACE2 binding site and operates via an ACE2 dependent mechanism of action. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0113] In some embodiments, provided herein is a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody IMM20253 to the subject. In some embodiments the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6. In some embodiments, the antibody comprises a HCDR1, a HCDR2 and a HCDR3 of the HCVR
amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6. In some embodiments, IMM20253 targets an epitope outside the ACE2 binding site and operates via an ACE2 independent mechanism of action. In some embodiments, IMM20253 has high affinity for virus comprising pre-cleaved Spike proteins or Spike proteins that are more susceptible to cleavage, such as Omicron. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0114] In some embodiments, provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody IMM20253 to the subject. In some embodiments the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6. In some embodiments, the antibody comprises a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6. In some embodiments, IMM20253 targets an epitope outside the ACE2 binding site and operates via an ACE2 independent mechanism of action. In some embodiments, IMM20253 targets an epitope outside the ACE2 binding site and operates via an ACE2 independent mechanism of action. In some embodiments, IMM20253 has high affinity for virus comprising pre-cleaved Spike proteins or Spike proteins that are more susceptible to cleavage, such as Delta. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0115] In some embodiments, provided herein is a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody MM20190 to the subject. In some embodiments, the antibody comprises a HCVR comprising three CDRs, (HCDR1,
HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4. In some embodiments, the antibody comprises a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4. In some embodiments, IMM20190, targets an epitope within the ACE2 binding site and operates via an ACE2 dependent mechanism of action. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0116] In some embodiments, provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody MM20190 to the subject. In some embodiments, the antibody comprises a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4. In some embodiments, the antibody comprises a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4. In some embodiments, IMM20190, targets an epitope within the ACE2 binding site and operates via an ACE2 dependent mechanism of action. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0117] In some embodiments, provided herein is a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184 and IMM20253 to the subject. In some embodiments, the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising
three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 and ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6. In some embodiments, the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 and ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0118] In some embodiments, provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184 and IMM20253 to the subject. In some embodiments, the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 and ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6. In some embodiments, the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 and ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an
immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0119] In some embodiments, provided herein is a method of treating an Omicron variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184, IMM20253, and IMM20190 to the subject. In some embodiments, the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6; and iii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4. In some embodiments, the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and a LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6, and iii) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
[0120] In some embodiments, provided herein is a method of treating a Delta variant of SARS-CoV-2 in a subject comprising administering antibody IMM20184, IMM20253, and IMM20190 to the subject. In some embodiments, the method comprises administering i) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6 ii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6; and iii) an antibody comprising a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4. In some embodiments, the method comprises administering i) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:1 and a LCDR1, a LCDR2, and a LCRD3 of the LCVR amino acid sequence set forth in SEQ ID NO: 2 ii) an antibody comprising a HCDR1, a HCDR2 and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO:5 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:6, and iii) an antibody comprising a HCDR1, a HCDR2, and a HCDR3 of the HCVR amino acid sequence set forth in SEQ ID NO: 3 and a LCDR1, a LCDR2, and a LCDR3 of the LCVR amino acid sequence set forth in SEQ ID NO:4. In some embodiments, the subject is immunocompromised. In some embodiments, the subject is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the antibody or antigen binding fragment thereof is administered at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0121] ”Subjects” refers to any person who has been infected with, or has the potential to be infected with the SARS-CoV-2 virus. In some embodiments of the invention those subjects may be of high risk for contracting the virus as a result of being immunocompromised through genetic mutation or drug treatment. For example, subjects may be being treated with immunosuppressive medications as a result of being a solid organ transplant recipient or having a chronic inflammatory disease (e.g. rheumatoid arthritis,
psoriasis, chrohn’s disease). They may be being treated with chemotherapeutic, radiation, or targeted agents that suppress immune function for treatment of diseases such as cancer. Subjects may also have conditions that place them into high-risk categories for developing severe COVID-19, such as diabetes, chronic pulmonary conditions, chronic cardiovascular conditions, obesity, or pregnancy. [0122] "Preventing" a disease refers to inhibiting the full development of a disease. Other terms, such as "prophylaxis", are also understood to refer to the concept of preventing a disease. [0123] "Treating" refers to a therapeutic intervention that ameliorates, (i.e., reduces the severity), a sign or symptom of a disease or pathological condition after it has begun to develop. In some embodiments of the invention, the antibodies or antigen-binding fragments thereof contained in the pharmaceutical composition treat or prevent the a SARS-CoV-2 infection by neutralizing SARS-CoV-2 virus and/or a SARS-CoV-2 variant. Examples of SARS-CoV-2 variants that are responsive to a method of treatment of the invention include the: Alpha (U.K./B.1.1.7), Beta (South African/B.1.351), Gamma (Brazil/P.1), Delta (India/B.1.617.2), B.1.617 (L452R/E484Q) and Epsilon (California/B.1.429/427). Other examples of SARS-CoV-2 variants that are responsive to a method of treatment of the invention include the CDC Variants of Interest, including the Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV- 2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS- CoV-2. [0124] In certain embodiments, an effective amount of an antibody or antibody composition of the invention to prevent or treat SARS-CoV-2 infection does not result in complete protection from a SARS-CoV-2 disease but results in a lower titer or reduced number of SARS-CoV-2 viruses compared to an untreated subject. In certain embodiments, the effective amount results in a 0.5-fold, 1-fold, 2-fold, 4-fold, 6-fold, 8-fold, 10-fold, 15- fold, 20-fold, 25-fold, 50-fold, 75-fold, 100-fold, 125-fold, 150-fold, 175-fold, 200-fold, 300- fold, 400-fold, 500-fold, 750-fold, or 1,000-fold or greater reduction in titer of SARS-CoV-2 virus relative to an untreated subject. In some embodiments, the effective amount results in a
reduction in titer of SARS-CoV-2 virus relative to an untreated subject of approximately 1 log or more, approximately 2 logs or more, approximately 3 logs or more, approximately 4 logs or more, approximately 5 logs or more, approximately 6 logs or more, approximately 7 logs or more, approximately 8 logs or more, approximately 9 logs or more, approximately 10 logs or more, 1 to 5 logs, 2 to 10 logs, 2 to 5 logs, or 2 to 10 logs. Benefits of a reduction in the titer, number or total burden of SARS-CoV-2 virus include, but are not limited to, less severe symptoms of the infection, fewer symptoms of the infection, reduction in the length of the disease associated with the infection, reduction in the length of viral shedding, and prevention of the onset or diminution of severity of diseases occurring secondary to SARS- CoV-2 virus infections. Antibodies [0125] Antibodies of the invention are typically monoclonal antibodies, meaning an antibody is produced by a single clonal B-lymphocyte population, a clonal hybridoma cell population, or a clonal population of cells into which the genes of a single antibody, or portions thereof, have been transfected. Monoclonal antibodies are produced by methods known to those of skill in the art, for instance by making hybrid antibody-forming cells from a fusion of myeloma cells with immune lymphocyte cells. [0126] An antibody of the invention may also be an "antigen-binding fragment". An antigen-binding fragment refers to a polypeptide fragment of an immunoglobulin or antibody that binds antigen or competes with intact antibody (i.e., with the intact antibody from which they were derived) for antigen binding (i.e., specific binding to an epitope of SARS-CoV-2). As used herein, the term "fragment" of an antibody molecule includes antigen-binding fragments of antibodies, for example, an antibody light chain variable domain (VL), an antibody heavy chain variable domain (VH), a single chain antibody (scFv), a F(ab')2 fragment, a Fab fragment, an Fd fragment, an Fv fragment, and a single domain antibody fragment (DAb). Fragments can be obtained, e.g., via chemical or enzymatic treatment of an intact or complete antibody or antibody chain or by recombinant means. Examples of immunoglobulin variants that are considered antibodies according to the invention include single-domain antibodies (such as VH domain antibodies), Fab fragments, Fab' fragments, F(ab)'2 fragments, single chain Fv proteins ("scFv"), and disulfide stabilized Fv proteins ("dsFv"). A VH single-domain antibody is an immunoglobulin fragment consisting of a heavy chain variable domain. An Fab fragment contains a monovalent antigen-binding
immunoglobulin fragment, which can be produced by digestion of whole antibody with the enzyme papain to yield an intact light chain and a portion of one heavy chain. Similarly, an Fab' fragment also contains a monovalent antigen-binding immunoglobulin fragment, which can be produced by digestion of whole antibody with the enzyme pepsin, followed by reduction, to yield an intact light chain and a portion of the heavy chain. Two Fab' fragments are obtained per immunoglobulin molecule. A (Fab')2 fragment is a dimer of two Fab' fragments, that can be obtained by treating whole antibody with the enzyme pepsin without subsequent reduction, so Fab' monomers remain held together by two disulfide bonds. An Fv fragment is a genetically engineered fragment containing the variable region of the light chain and the variable region of the heavy chain expressed as two chains. A single chain ("sc") antibody, such as scFv fragment, is a genetically engineered molecule containing the VL region of a light chain, the VH region of a heavy chain, linked by a suitable polypeptide linker as a genetically fused single chain molecule. A dimer of a single chain antibody, such as a scFV2 antibody, is a dimer of a scFV, and may also be known as a "miniantibody". A dsFvs variant also contains a VL region of an immunoglobulin and a VH region, but the chains have been mutated to introduce a disulfide bond to stabilize the association of the chains. [0127] One of skill in the art will realize that conservative variants of the antibodies can be produced. Such conservative variants employed in antibody fragments, such as dsFv fragments or in scFv fragments, will retain critical amino acid residues necessary for correct folding and stabilizing between the VH and the VL regions, and will retain the charge characteristics of the residues in order to preserve the low pI and low toxicity of the molecules. [0128] An antibody of the invention is typically a "human" antibody, which may also be referred to as a "fully human" antibody. An antibody which possess a human framework regions and CDRs from a human immunoglobulin is generally be considered to be a human or humanized antibody. For example, a human or humanized antibody may contain the framework and the CDRs of an antibody from the same originating human heavy chain, or human light chain amino acid sequence, or both. Alternatively, the framework regions may originate from one human antibody, and be engineered to include CDRs from a different human antibody.
[0129] An antibody according to the invention may also comprise a "tagged" immunoglobulin CH3 domain to facilitate detection of the biologic against a background of endogenous antibodies. More particularly, a tagged CH3 domain is a heterogeneous antibody epitope that has been incorporated into one or more of the AB, EF, or CD structural loops of a human IgG-derived CH3 domain. For example a CH3 tag may be incorporated into the structural context of an IgG1 subclass antibody, other human IgG subclasses, including IgG2, IgG3, and IgG4. Epitope-tagged CH3 domains, also referred to as "CH3 scaffolds" can be incorporated into any antibody of the invention having a heavy chain constant region, generally in the form of an immunoglobulin Fc portion. Examples of CH3 scaffold tags, and methods for incorporating them into antibodies are disclosed in International Patent Application No. PCT/US2019/032780. Antibodies used to detect epitope tagged CH3 scaffolds, and antibodies of the invention, that comprise epitope tagged CH3 scaffolds, are generally referred to herein as "detector antibodies". [0130] Some antibodies of the invention may be described as an "isolated" antibody or "isolated" antigen-binding fragment thereof. An isolated antibody or antigen-binding fragment thereof has been substantially separated or purified away from other biological components environment, such as a cell, proteins and organelles. Ordinarily, an isolated antibody or antigen-binding fragment thereof is prepared by a process involving at least one purification step, although the term "isolated" is not intended to refer to a complete absence of such biological molecules or to an absence of water, buffers, or salts or to components of a pharmaceutical formulation that includes an antibody or antigen-binding fragment of the invention. [0131] In some embodiments of the invention, antibody or antigen-binding fragment thereof is specific for the Spike protein (S) of SARS-CoV-2. Thus, an antibody or antigen- binding fragment thereof may, in one embodiment, bind the S1 subunit of the Spike protein, while in another embodiment, an antibody or antigen-binding fragment thereof binds the S2 subunit. Moreover, some antibodies or antigen-binding fragments thereof bind to the receptor-binding domain (RBD) of the S protein, while other antibodies of the invention bind to non-RBD epitopes of the S protein. RBD-binding antibodies or antigen-binding fragments thereof of the invention may, in certain embodiments, bind a soluble form of the RBD of the S protein. Some antibodies or antigen-binding fragments of the invention bind to sites on the RBD that are resistant to mutational drift. For example, in some embodiments, an antibody
or antigen-binding fragment thereof binds the RBD at, or in close proximity of such a site described by Greaney t al. (2021) as the “the E465 patch”. [0132] Indeed, among the antibodies of the invention that bind the Spike protein, there are some that bind to highly-conserved epitopes. Accordingly, binding of an antibody or antigen-binding fragment of the invention to a highly-conserved site of the SARS-CoV-2 Spike protein is not affected by point mutations in the Spike protein associated with variants of SARS-Cov-2, Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV- 2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2. [0133] In certain embodiments of the invention, an isolated antibody or antigen-binding fragment thereof contains a heavy chain variable region (HCVR) and a light chain variable region (LCVR) that is described in Table 1. In some embodiments, provided herein is an antibody comprising a HCDR1, a HCDR2, a HCDR3, a LCDR1, a LCDR2, and a LCDR3 of a heavy chain variable region (HCVR) and a light chain variable region (LCVR) that is described in Table 1. In some embodiments, provided herein is an antibody comprising a HCDR1, a HCDR2, a HCDR3, a LCDR1, a LCDR2, and a LCDR3 set forth in Table 1. In some embodiments, the CDRs are determined using Kabat, Chothia, or contact systems. In some embodiments, the CDRs are determined using the system described in North et al. J.M.B 406(8):228-56 (2011) as set forth in Table 1. Table 1 Description SEQ Sequence Name Binds Amino acid sequence ID P _ ( I P _ (
IMM200184) ASWVFGGGTKLTVL
Description SEQ Sequence Name Binds Amino acid sequence ID No. VH of IMM20190 EVQLVESGGGLVQPGGSLRLSCSAS P _ ( I P _ ( I P _ ( I P _ ( I P _ P _ P _ P _ P
_a_HC RBD GGIIPIFGTANYAQKFQGRVTITADE
Description SEQ Sequence Name Binds Amino acid sequence ID No. STSTAYMELSSLRSEDTAVYYCASD P _ P _ P _ P _ P _ P _ P _ P _ ( I
S
Description SEQ Sequence Name Binds Amino acid sequence ID No. VL of IMM20279 DIQMTQSPSSLSASVGERATITCRAS P _ ( I P _ P _ P _ P _ P _ P _ P _ P L
LFTFGPGTKVDIK
Description SEQ Sequence Name Binds Amino acid sequence ID No. QVQLQESGPGLVKPSETLSLTCAVY P _ P _ P _ P _ P _ P _ P _ P _ P _
b_λ_HC VGRIKSKTDGGTIDYAAPMKGRLII
Description SEQ Sequence Name Binds Amino acid sequence ID No. SRDDSKNMLYLQMSSLKTDDTAV P _ P _ P _ P _ P _ P _ P _ P _ P
_ _LC S SDVGSYNLVSWYQQHPGKAPKLMI 46
Description SEQ Sequence Name Binds Amino acid sequence ID No. Non- YEGNKRPSGVSNRFSGSKSGNTASL P _ P _ P _ P _ P _ P _ P _ P _
TVL
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR193 00018 HCDR1 _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _
69
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR200 00622 LCDR1 _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P
_ _a_HC 87
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR196 00413 LCDR1 TSSQSVLYSSNNKNFLA _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _
109
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR199 00255 HCDR2 _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P
_ _LC 130
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR194 00068 LCDR2 YQDTKRPS _ P _ P _ P _ P _ P L P L P L P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P
_ _HC 152
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR210 01524 HCDR3 ARDGQWLRILDY _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P
_ _κ_LC 174
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR197 00705 HCDR1 _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P
_ _LC 196
Description SEQ Sequence Name Binds Amino acid sequence ID No. PR194 00292 LCDR2 YEGTKRPS _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ P _ C l w s C
epitope 218
Description SEQ Sequence Name Binds Amino acid sequence ID No. CD-4I2X:A WEIDGSERQNGKTT e b s C e C e b s C e C e b s C e C e b s C e C e b s C e C e b s C e C d e b s C d e C
epitope with 233
Description SEQ Sequence Name Binds Amino acid sequence ID No. border s C e C e b s C e C e b s C e C e b s C e C e b s C e C e b s C e E e b s E e E e b s E
epitope 248
Description SEQ Sequence Name Binds Amino acid sequence ID No. EF-4I2X:E WELDRWDVKTT e b s E e E e b s E e
[0134] In some embodiments, provided herein is an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGASWV (SEQ ID NO: 60). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 1. In some embodiments, the VL comprises the amino acid sequence set forth in SEQ ID NO: 2. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 1 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 2. In some embodiments, the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 2. In some embodiments the antibody comprises one or more CDRs or variable region sequences of IMM20184 (PR193_00018_HC). In some embodiments, the antibody binds to an epitope on the Spike protein outside of the ACE2 binding site. In some embodiments, the antibody neutralizes SARS-CoV-2 through an ACE2-dependent mechanism. In some embodiments, IMM20184 contacts one or more amino acids in the
Spike protein selected from the group consisting of N370, A372, F374, K378, S383, and P384. In some embodiments, IMM20184 binds to a conserved epitope. In some embodiments, IMM20184 binds to an epitope of the Spike protein comprising one or more of N370, A372, F374, K378, S383, and P384. [0135] In some embodiments, provided herein is an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the amino acid sequence QKYNSAPGLT (SEQ ID NO: 66). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 3. In some embodiments, the VL comprises the amino acid sequence set forth in SEQ ID NO: 4. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 3 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 4. In some embodiments, the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 4. In some embodiments, the antibody comprises one or more CDRs or variable region sequences of IMM20190 (PR194_00232_HC). In some embodiments, the antibody binds to an epitope of the Spike protein in the ACE2 binding site and operates through an ACE2-dependent mechanism. In some embodiments, IMM20190 contacts one or more amino acids of the Spike protein selected from the group consisting of K417, D420, L455, F456, N460, Y473, N487, Y489, N501, and Y505. In some embodiments, IMM20190 binds to a non-conserved epitope. In some embodiments, IMM20190 binds to an epitope of the Spike protein comprising one or more of K417, D420, L455, F456, N460, Y473, N487, Y489, N501, and Y505. [0136] In some embodiments, provided herein is an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL),
wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 5. In some embodiments, the VL comprises the amino acid sequence set forth in SEQ ID NO: 6. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 5 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 6. In some embodiments, the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 6. In some embodiments, the antibody comprises one or more CDR sequences or variable region sequences of IMM20253 (PR200_00622_HC). In some embodiments, the antibody binds to a Spike protein with a mutation in the protease cleavage site. In some embodiments, the antibody binds with high affinity to pre-cleaved Spike proteins (i.e. Spike proteins that are cleaved prior to binding to the host cell surface). In some embodiments, the antibody binds with high affinity to Spike proteins with mutations that make a protease site more readily cleavable. In some embodiments, the antibody binds to a Spike protein at an epitope outside of the ACE2 binding site and operates through an ACE2 independent mechanism. In some embodiments, the antibody causes a confirmation change in the Spike protein. In some embodiments, the antibody makes the Spike protein more susceptible to cleavage. In some embodiments IMM20253 contacts one or more amino acid of the Spike protein selected from the group consisting of K356 and R466. In some embodiments, IMM20253 binds to a conserved epitope. In some embodiments, IMM20253 binds to an epitope of the Spike protein comprising K356 and/or R466. [0137] In some embodiments, provided herein is an antibody that binds to a SARS-CoV- 2 Spike protein comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence
VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 19. In some embodiments, the CDRs are defined according to North et al. In some embodiments, the VL comprises the amino acid sequence set forth in SEQ ID NO: 20. In some embodiments, the VH comprises the amino acid sequence set forth in SEQ ID NO: 19 and the VL comprises the amino acid sequence set forth in SEQ ID NO: 20. In some embodiments, the antibody comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 19, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 20. In some embodiments, the antibody comprises one or more CDR sequences or variable region sequences of IMM20279 (PR199_00255_HC). In some embodiments, the antibody binds to an epitope of the Spike protein outside of the ACE2 binding site. In some embodiments, the antibody comprising one or more CDR sequences or variable region sequences of IMM20279 cross-reacts with an antibody comprising one or more CDR sequences or variable region sequences of IMM20184. In some embodiments of the invention, an isolated antibody or antigen-binding fragment thereof binds a SARS-CoV-2 Spike protein, and contains one of the following combinations of a heavy chain variable region (HCVR) and a light chain variable region (LCVR): a HCVR comprising three heavy chain complementarity determining regions (CDRs), (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 1, and a LCVR comprising three light chain complementarity determining regions (CDRs) (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 2; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 3, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 4; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 19, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 20; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth
in SEQ ID NO: 5, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 6; a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 7, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 8; or a HCVR comprising three CDRs, (HCDR1, HCDR2 and HCDR3), wherein the amino acid sequence of the HCVR is set forth in SEQ ID NO: 23, and a LCVR comprising three CDRs, (LCDR1, LCDR2 and LCDR3), wherein the amino acid sequence of the LCVR is set forth in SEQ ID NO: 24. [0138] As described above, some antibodies of the invention bind to highly-conserved epitopes, and, thus, are not affected by point mutations in the Spike protein associated with variants of SARS-Cov-2. A nonlimiting list of examples of antibodies or antigen-binding fragments of the invention which bind equivalently to the SARS-CoV-2 reference isolate, USA/WA_CDC-WA1/2020, and each of the aforementioned variant isolates, include: An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 1 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 2 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 3 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 4 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 5 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 6 or fragment thereof; and An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 19 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 20 or fragment thereof. [0139] The foregoing antibodies of the invention neutralize SARS-CoV-2 and SARS- CoV-2 variants, including variants Alpha (U.K./B.1.1.7), Beta (South African/B.1.351), Gamma (Brazil/P.1), Delta (India/B.1.617.2), B.1.617 (L452R/E484Q) and Epsilon (California/B.1.429/427), either alone, or in combination. In some embodiments, the antibodies neutralize the Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV- 2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda
(C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2. More particularly, certain antibodies of the invention bind the SARS-CoV-2 RBD at epitopes, which contain residues that are conserved among variants. In some embodiments, binding to such conserved residues prevents viral escape. In some embodiments, binding to such conserved residues allows binding across variants. Accordingly, a RBD amino acid substitution at a non-conserved or poorly-conserved residue position will have limited or no impact on binding of an antibody of the invention. Such non conserved or poorly-conserved substitutions in the RBD are commonly associated with SARS-CoV-2 variants. Thus, an RBD epitope of some antibodies of the invention may contain at least 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 or more residues at RBD positions 356 (K), 370 (N), 372 (A), 374 (F), 378 (K), 384 (P), 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 466 (R), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y). For example, an antibody of the invention may bind an RBD epitope in which the binding energy is relatively higher at residues at 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y). Likewise, another antibody of the invention may bind an RBD epitope in which the binding energy is relatively higher at residues at 370 (N), 372 (A), 374 (F), 378 (K), 384 (P). And yet another antibody of the invention may bind an RBD epitope in which the binding energy is relatively higher at residues at 356 (K) and 466 (R). In some embodiments, the antibody contacts one or more residues at RBD positions 356 (K), 370 (N), 372 (A), 374 (F), 378 (K), 384 (P), 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 466 (R), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y). In some embodiments, the antibody contacts one or more of 417 (K), 420 (D), 455 (L), 456 (F), 460 (N), 473 (Y), 487 (N), 489 (Y), 501 (N), and 505 (Y). In some embodiments, the antibody contacts residues at 370 (N), 372 (A), 374 (F), 378 (K), 384 (P). In some embodiments, the epitope is identified using alanine scanning. [0140] In some embodiments, the antibody binds the SARS-CoV-2 RBD at epitopes, which contain residues that are not conserved. [0141] In certain embodiments of the invention, for example, a composition (i.e., a mixture) of two or more of: An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 1 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 2 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 3 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 4 or fragment thereof; An antibody with a
HCVR based on the amino acid sequence set forth in SEQ ID NO: 5 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 6 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 19 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 20 or fragment thereof; and An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 21 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 22 or fragment thereof, additively or synergistically neutralizes SARS-CoV-2, or a variant thereof, such as the Alpha (U.K./B.1.1.7), Beta (South African/B.1.351), Gamma (Brazil/P.1), Delta (India/B.1.617.2), B.1.617 (L452R/E484Q) and Epsilon (California/B.1.429/427). In some embodiments, the antibodies neutralize the Omicron variant. In some embodiments, the antibodies neutralize Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS-CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2. [0142] In some embodiments, an antibody of the invention alters the conformation of Spike protein upon binding of the antibody to the Spike protein. More particularly, in certain embodiments, an antibody of the invention binds a Spike protein epitope located on the outside face of the RBD domain of the SARS-CoV-2 RBD. In such embodiments, the epitope may be in close proximity to the N-terminal domain of a second Spike protein within the Spike trimer, and binding of the antibody inactivates the virus’ ability to bind cells. Consequently, certain antibodies of the invention neutralize SARS-CoV-2 by the foregoing mechanism. More specifically, binding of such antibodies to the outer face of the RBD imparts its intrinsic neutralization through induction of a conformational change in the Spike protein that prevents virus uptake into the cells without competing for ACE2 binding. An example of an embodiment of the invention that alters conformation of the Spike protein upon binding is an antibody with a heavy chain variable region (HCVR) or fragment thereof and/or a light chain variable region (LCVR) or a fragment thereof derived from an antibody described herein as IMM20253, which contains HCVR and LCVR with amino acid sequences of SEQ ID NOS. 5 and 6, respectively.
[0143] In certain embodiments, an antibody of the invention that alters the conformation of Spike protein upon binding of the antibody to the Spike protein as part of its intrinsic neutralization activity, acts additively, and more preferentially synergistically, with other antibodies that compete for ACE2 binding as part of their intrinsic neutralization mechanism. An example of an embodiment of the invention that alters conformation of the Spike protein upon binding and acts additively, and more preferentially synergistically, with antibodies that block ACE2 binding, is an antibody with a heavy chain variable region (HCVR) or fragment thereof and/or a light chain variable region (LCVR) or a fragment thereof derived from an antibody described herein as IMM20253, which contains HCVR and LCVR with amino acid sequences of SEQ ID NOS. 5 and 6, respectively. [0144] As described above, an isolated antibody or antigen-binding fragment thereof of the invention may contain a CH3 scaffold "epitope tag", comprising at least one modification of the wild-type amino acid sequence of the CH3 domain derived from an immunoglobulin Fc region. Accordingly, any of the aforementioned antibodies may have been engineered to contain a CH3 scaffold. The CH3 scaffold of such an isolated antibody or antigen-binding fragment thereof may possess at least one modification of the wild-type sequence occurs within the AB, EF, or CD loops of the CH3 scaffold, including an amino acid substitution, deletion or insertion, for example. In certain embodiments, the epitope tag amino acid sequence contains a sequence derived from SIRPα or Sip. Alternatively, he epitope tag amino acid sequence contains a sequence derived from a constant light chain of an antibody. More particularly, the antibody epitope amino acid sequence of an isolated antibody or antigen-binding fragment thereof that contains a CH3 scaffold with an amino acid sequence set forth in SEQ ID Nos. 3-30, SEQ ID Nos. 33-57, or SEQ ID Nos. 60-67 of International Patent Application No. PCT/US2019/032780. Compositions [0145] Also provided herein are compositions comprising one or more antibodies provided herein. In some embodiments, provided herein a composition comprising two or more antibodies that bind to a Spike protein, wherein the antibodies bind to different epitopes. In some embodiments, the composition comprises an antibody that operates in an ACE2 independent mechanism and an antibody that operates through an ACE2 dependent mechanism. In some embodiments, the multiple antibodies in the composition act synergistically to treat a SARS-CoV-2 infection in a subject. In some embodiments, the
presence of multiple antibodies binding to different epitopes of a SARS-CoV-2 protein allows treatment of variants with mutations in one or more SARS-CoV-2 proteins, such as the Spike protein. [0146] In some embodiments, provided herein is a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGASWV (SEQ ID NO: 60). In some embodiments, the CDRs are defined according to North et al. [0147] In some embodiments, provided herein is a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the amino acid sequence QKYNSAPGLT (SEQ ID NO: 66). In some embodiments, the CDRs are defined according to North et al. [0148] In some embodiments, provided herein is a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the CDRs are defined according to North et al.
[0149] In some embodiments, provided herein is a composition comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the CDRs are defined according to North et al. [0150] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66). [0151] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody
comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69), and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). [0152] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). [0153] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO:
68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). [0154] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). [0155] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of
RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). [0156] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). [0157] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino
acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). [0158] In some embodiments, provided herein is a composition comprising a first antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of
YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). [0159] In some embodiments, provided herein is a composition comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a fourth antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). [0160] Pharmaceutical compositions of the invention contain one or more isolated antibodies or antigen-binding fragments thereof of the invention and a pharmaceutically
acceptable carrier or diluent. In one embodiment of the invention, the pharmaceutical composition contains only one of the Spike-binding antibodies described herein. Other pharmaceutical composition of the invention contain a mixture of different Spike-binding antibodies which are described herein, such as, for example, at least 2, at least 3, at least 4, or at least 5, or at least 6 or more Spike-binding antibodies. For example, a pharmaceutical composition of two or more of: An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 1 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 2 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 3 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 4 or fragment thereof; An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 5 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 6 or fragment thereof; an antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 19 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 20 or fragment thereof, and An antibody with a HCVR based on the amino acid sequence set forth in SEQ ID NO: 21 or fragment thereof, and a LCVR based on the amino acid sequence set forth in SEQ ID NO: 22 or fragment thereof, additively or synergistically neutralizes SARS-CoV-2, or a variant thereof, such as the Alpha (U.K./B.1.1.7), Beta (South African/B.1.351), Gamma (Brazil/P.1), Delta (India/B.1.617.2), B.1.617 (L452R/E484Q) and Epsilon (California/B.1.429/427). In some embodiments, the antibodies neutralize Alpha (U.K./B.1.1.7) variant of SARS-CoV-2, the Beta (South African/B.1.351) variant of SARS- CoV-2, the Gamma (Brazilian/P.1) variant of SARS-CoV-2, the Delta (India/B.1.617.2), B.1.617 (L452R/E484Q )), the Epsilon (California/B.1.429/427, New York/B.1.526/526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2, the lambda (C.37) variant of SARS-CoV-2, or the Omicron (B.1.1.529) variant of SARS-CoV-2. [0161] In some embodiments a pharmaceutical composition of the invention further contains a second therapeutic agent. For example, a pharmaceutical composition of the invention may also contain an anti-inflammatory agent or an antiviral agent. [0162] In some embodiments, the second agent is an antibody. In some aspects, the second agent is casirivimab (REGN10933). In some aspects, the second agent is imdevimab (REGN10987). In some aspects, the second agent is a combination of the neutralizing
antibodies, casirivimab and imdevimab (REGEN-COV, previously known as REGN-COV-2) (ClinicalTrials.gov number, NCT04452318 and NCT04425629). The two antibodies can simultaneously bind to two independent epitopes on the RBD (Hansen J. et.al., Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail. 369(6506):1010-1014 (2020); Baum A. et.al, Antibody cocktail to SARS-CoV-2 Spike protein prevents rapid mutational escape seen with individual antibodies, Science 369(6506):1014-1018 (2020)). In some aspects, the second agent is bamlanivimab (LY3819253). In some aspects, the second agent is etesevimab (LY3832479). In some aspects, the second agent is a combination of the neutralizing antibodies, bamlanivimab and etesevimab (ClinicalTrials.gov number, NCT04427501, Dougan M. et.al., Bamlanivimab plus Etesevimab in Mild or Moderate Covid-19. N Engl J med 385(15):1382-1392 (2021) ). In some aspects, the second agent is sotrovimab (ClinicalTrials.gov number, NCT04545060, Gupta A. et.al., Early Treatment for Covid-19 with SARS-CoV-2 Neutralizing antibody sotrovimab. N Engl J med 385:1941-1950 (2021)). [0163] As stated above, antibodies and pharmaceutical compositions of the invention can be used in methods for preventing or treating, a SARS-CoV-2 infection in subject. Accordingly, antibodies and pharmaceutical compositions of the invention are useful for treating the disease caused by SARS-CoV-2, commonly referred to as COVID-19, or simply COVID. A pharmaceutical composition of the invention is typically administered to subject in need thereof by injecting the composition into the body of the subject subcutaneously, intravenously or intramuscularly. Kits [0164] Also, provided herein are kits for treating or preventing a SARS-CoV-2 infection in a subject comprising one or more antibodies provided herein. In some embodiments, the kit comprises instructions for use according to the methods provided herein. In some embodiments, the kit comprises instructions for treating an immunocompromised subject. In some embodiments, the kit provides instructions for treating a subject who is receiving an immunosuppressant. In some embodiments the subject has received a solid organ transplant or has a chronic inflammatory disease. In some embodiments, the kit provides instructions for administration of the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg .In some embodiments, the kit provides instructions for In some embodiments, the kit comprises two antibodies that bind to a Spike
protein. In some embodiments, the kit comprises three or more antibodies that bind to a Spike protein. In some embodiments, the kit comprises four or more antibodies that bind to a Spike protein. In some embodiments, the anti-Spike antibodies are in different compositions in the kit. In some embodiments, the anti-Spike antibodies are in the same composition. [0165] In some embodiments, the kit comprises one or more of IMM20184, IMM20190, IMM20253, and/or IMM20279 and an additional agent. In some embodiments, the kit comprises one or more of IMM20184, IMM20190, IMM20253, and/or IMM20279 and an additional agent selected from the group consisting of casirivimab, imdevimab, bamlanivimab, etesevimab and sotrovimab. In some embodiments, the kit comprises an anti- Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTFSSFWMS (SEQ ID NO: 55); a HCDR2 comprising the amino acid sequence TIREDGSEKYYVD (SEQ ID NO: 56); and a HCDR3 comprising the amino acid sequence ARSKWLRGSFDY (SEQ ID NO: 57); wherein the antibody comprises a LCDR1 comprising the amino acid sequence TRRSGSIASNYVQ (SEQ ID NO: 58); a LCDR2 comprising the amino acid sequence YEDNQRPS (SEQ ID NO: 59); and a LCDR3 comprising the amino acid sequence QSYDSSNPPGASWV (SEQ ID NO: 60). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0166] In some embodiments, the kit comprises an anti-Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence SASGFTVSSNYMS (SEQ ID NO: 61); a HCDR2 comprising the amino acid sequence VIYAGGSTF (SEQ ID NO: 62); and a HCDR3 comprising the amino acid sequence ARDRGGYLDY (SEQ ID NO: 63); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQGISNYLA (SEQ ID NO: 64); a LCDR2 comprising the amino acid sequence YAASTLQS (SEQ ID NO: 65); and a LCDR3 comprising the amino acid sequence QKYNSAPGLT (SEQ ID NO: 66). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg.
[0167] In some embodiments, the kit comprises an anti-Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence TASGFTFSTYGMH (SEQ ID NO: 67); a HCDR2 comprising the amino acid sequence VISYDGSSKH (SEQ ID NO: 68); and a HCDR3 comprising the amino acid sequence ARDGQPPGWGNYFDY (SEQ ID NO: 69); wherein the antibody comprises a LCDR1 comprising the amino acid sequence GGNGIGSKSVY (SEQ ID NO: 70); a LCDR2 comprising the amino acid sequence YDDSDRPS (SEQ ID NO: 71); and a LCDR3 comprising the amino acid sequence QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0168] In some embodiments, the kit comprises an anti-Spike antibody comprising a variable heavy chain (VH) and a variable light chain (VL), wherein the antibody comprises a HCDR1 comprising the amino acid sequence AASGFTFSTYGMH (SEQ ID NO: 109); a HCDR2 comprising the amino acid sequence VIWYNGINKH (SEQ ID NO: 110); and a HCDR3 comprising the amino acid sequence ARDWGTLTTLFDF (SEQ ID NO: 111); wherein the antibody comprises a LCDR1 comprising the amino acid sequence RASQSISSHLN (SEQ ID NO: 112); a LCDR2 comprising the amino acid sequence YGASSLQS (SEQ ID NO: 113); and a LCDR3 comprising the amino acid sequence QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, the CDRs are defined according to North et al. In some embodiments, the kit comprises a composition comprising the antibody or antigen binding fragment for administration at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. [0169] In some embodiments, the kit comprises an anti-Spike antibody comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody
comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66). In some embodiments, each antibody is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0170] In some embodiments, the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69), and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
[0171] In some embodiments, the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0172] In some embodiments, the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the
amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0173] In some embodiments, the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0174] In some embodiments, the kit comprises a combination of two anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a
LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0175] In some embodiments, the kit comprises a combination of three anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3
comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0176] In some embodiments, the kit comprises a combination of three anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition.
[0177] In some embodiments, the kit comprises a combination of three anti-Spike antibodies comprising a first antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a second antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114). In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. [0178] In some embodiments, the kit comprises a combination of four anti-Spike antibodies comprising a first antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of SASGFTFSSFWMS (SEQ ID NO: 55), a HCDR2 comprising the amino acid sequence of TIREDGSEKYYVD (SEQ ID NO: 56), and a HCDR3 comprising the amino acid sequence of ARSKWLRGSFDY (SEQ ID NO: 57); and a VL comprising a LCDR1 comprising the amino acid sequence of TRRSGSIASNYVQ (SEQ ID NO: 58), a LCDR2 comprising the amino acid sequence of YEDNQRPS (SEQ ID NO: 59), and a
LCDR3 comprising the amino acid sequence of QSYDSSNPPGASWV (SEQ ID NO: 60); a second antibody comprising the amino acid sequence of SASGFTVSSNYMS (SEQ ID NO: 61), a HCDR2 comprising the amino acid sequence of VIYAGGSTF (SEQ ID NO: 62), and a HCDR3 comprising the amino acid sequence of ARDRGGYLDY (SEQ ID NO: 63); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQGISNYLA (SEQ ID NO: 64), a LCDR2 comprising the amino acid sequence of YAASTLQS (SEQ ID NO: 65), and a LCDR3 comprising the amino acid sequence of QKYNSAPGLT (SEQ ID NO: 66); a third antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of TASGFTFSTYGMH (SEQ ID NO: 67), a HCDR2 comprising the amino acid sequence of VISYDGSSKH (SEQ ID NO: 68), and a HCDR3 comprising the amino acid sequence of ARDGQPPGWGNYFDY (SEQ ID NO: 69); and a VL comprising a LCDR1 comprising the amino acid sequence of GGNGIGSKSVY (SEQ ID NO: 70), a LCDR2 comprising the amino acid sequence of YDDSDRPS (SEQ ID NO: 71), and a LCDR3 comprising the amino acid sequence of QVWDSSSDPWV (SEQ ID NO: 72); and a fourth antibody comprising a VH comprising a HCDR1 comprising the amino acid sequence of AASGFTFSTYGMH (SEQ ID NO: 109), a HCDR2 comprising the amino acid sequence of VIWYNGINKH (SEQ ID NO: 110), and a HCDR3 comprising the amino acid sequence of ARDWGTLTTLFDF (SEQ ID NO: 111); and a VL comprising a LCDR1 comprising the amino acid sequence of RASQSISSHLN (SEQ ID NO: 112), a LCDR2 comprising the amino acid sequence of YGASSLQS (SEQ ID NO: 113), and a LCDR3 comprising the amino acid sequence of QQSYSTPPWT (SEQ ID NO: 114).In some embodiments, the kit comprises an anti-Spike antibody provided in Table 1 and one or more additional antibodies. In some embodiments, the kit comprises a combination of casirivimab and imdevimab (REGEN-COV, previously known as REGN-COV-2). In some embodiments, the kit comprises a combination of bamlanivimab and etesevimab. In some embodiments, the kit comprises sotrovimab. In some embodiments, each antibody or antigen binding fragment thereof is provided in a composition suitable for administering the antibody or antigen binding fragment thereof at a dose of about 200 mg, about 400 mg, about 600 mg, or about 800 mg. In some embodiments, each antibody is provided in a separate composition. In some embodiments, each antibody is provided in the same composition. Embodiments Embodiment 1. A method of treating a SARS-CoV-2 infection in an immunocompromised subject, comprising administering to the subject at least first and second antibodies or antigen
binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein. Embodiment 2. A method of treating a SARS-CoV-2 infection in an immunocompromised subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 independent mechanism and the second antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism. Embodiment 3. A method of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein. Embodiment 4. A method of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 independent mechanism and the second antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism. Embodiment 5. The method of any one of embodiments 1-4, further comprising administering a third antibody or antigen binding fragment thereof that specifically binds to the Spike protein of SARS-CoV-2 at an epitope distinct from the first and second antibodies or antigen binding fragments thereof.
6. The method of embodiment 3, wherein the third antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein. Embodiment 7. The method of embodiment 5 or embodiment 6, wherein the third antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism. Embodiment 8. The method of any one of embodiments 5-7, wherein each of the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 600 mg to about 2400 mg. Embodiment 9. The method of embodiment 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 600 mg. Embodiment 10. The method of embodiment 9, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg each. Embodiment 11. The method of embodiment 8, wherein first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 1200 mg. Embodiment 12. The method of embodiment 11, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 400 mg each. Embodiment 13. The method of embodiment 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 1800 mg. Embodiment 14. The method of embodiment 13, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 600 mg each.
Embodiment 15. The method of embodiment 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 2400 mg. Embodiment 16. The method of embodiment 15, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 800 mg each. Embodiment 17. The method of any one of embodiments 5-8, wherein the first, second and third antibodies or antigen binding fragments thereof are administered at a dose of about 3 mg/kg, or about 6 mg/kg each. Embodiment 18. A method of treating a SARS-CoV-2 infection in an immunocompromised subject, comprising administering to the subject an antibody or antigen binding fragment that binds to the Spike protein of SARS-CoV-2, wherein the antibody or antigen binding fragment comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 2; a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 4; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 6; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 7, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 8; a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region comprising the amino acid sequence set forth in SEQ ID NO: 9, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 10;
a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 11, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 12; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 13, a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 14; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 15, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO:16 a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 17, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO:18; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 19, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 20; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 21, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 22; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 23, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 24; or a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 25 and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 26. Embodiment 19. The method of embodiment 18, wherein the antibody or antigen binding fragment thereof comprises a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 2; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 4; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 6;
a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 7, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 8; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 9, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 10; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 11, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 12; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 13, a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 14; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 15, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 16; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: SEQ ID NO:17, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 18; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 19, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 20; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 21, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 22; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 23, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 24; or a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 25 and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 26. Embodiment 20. The method of embodiment 18, wherein the antibody or antigen binding fragment thereof comprises a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60; a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66;
a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72; or a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114. Embodiment 21. The method of any one of embodiments 18-20, comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66; b) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and
a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72; c) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114; d) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72; e) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid
sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114; or f) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114. Embodiment 22. The method of any one of embodiments 5-20 comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid
sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72; b) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114; or c) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2
comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114. Embodiment 23. The method of embodiment 18 comprising administering to the subject a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, and a fourth antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the
amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114. Embodiment 24. The method of any one of embodiments 1-23, wherein the subject has a high risk for contracting SARS-CoV-2. Embodiment 25. The method of any one of embodiments 1-24, wherein the subject is being treated with an immunosuppressant. Embodiment 26. The method of embodiment 25, wherein the immunosuppressant is a corticosteroid. Embodiment 27. The method of any one of embodiments 1-26, wherein the subject has received a solid organ transplant or has a chronic inflammatory disease. Embodiment 28. The method of embodiment 27, wherein the subject has a chronic inflammatory disease selected from the group consisting of rheumatoid arthritis, psoriasis, and Crohn’s disease. Embodiment 29. The method of any one of embodiments 1-24, wherein the subject is being treated with a chemotherapeutic agent or radiation. Embodiment 30. The method of any one of embodiments 1-29, wherein the subject is at risk for developing severe COVID-19. Embodiment 31. The method of any one of embodiments 1-24 or 30, wherein the subject has diabetes, a chronic pulmonary condition, a chronic cardiovascular condition, obesity, or is pregnant. Embodiment 32. The method of any one of embodiments 1-31, wherein the viral load in the subject is decreased upon treatment. Embodiment 33. The method of any one of embodiments 1-32, wherein administration of the antibody or antigen-binding fragment thereof results in viral clearance in the subject.
Embodiment 34. The methods of any one of embodiments 1-33, wherein the method results in viral neutralization. Embodiment 35. The method of any one of embodiments 1-17 and 21-34, wherein the ratio between the first and the second antibodies or antigen binding fragments thereof is about 1 : 1. Embodiment 36. The method of any one of embodiments 1-17 and 22-34, wherein the ratios between the first, second, and third antibodies or antigen binding fragments thereof are about 1 : 1 : 1. Embodiment 37. The method of embodiment 23, wherein the ratio between the first, second, third, and fourth antibodies or antigen binding fragments thereof is about 1 : 1 : 1: 1. Embodiment 38. The method of any one of embodiments 1-37, wherein the antibody or antigen binding fragment therefor is an Fc IgG1, IgG2, IgG3, IgG4, IgM, IgD, IgA1, IgA2 or IgE isotype. Embodiment 39. The method of embodiment 38, wherein the antibody is an IgG1 isotype. Embodiment 40. The method of embodiment 39, wherein the IgG1 is a G1m1 or nG1m1 allotype. Embodiment 41. The method of any one of embodiment 1-40, wherein the antibody is a fully human antibody. Embodiment 42. The method of any one of embodiments 1-41, wherein the antibody is a full length antibody. Embodiment 43. The method of any one of embodiments 1-42, wherein binding of the antibody or antigen-binding fragment thereof: inhibits binding of a SARS-CoV-2 virus to a host ACE2 receptor; fixes complement to a SARS-CoV-2 virus;
induces phagocytosis of a SARS-CoV-2 virus; or any combination thereof. Embodiment 44. The method of any one of embodiments 1-43, wherein the binding of the antibody or antigen-binding fragment thereof neutralizes a SARS-CoV-2 virus by blocking binding of the receptor binding domain (RBD) of the virus with an ACE2 receptor. Embodiment 45. The method of any one of embodiments 1-44, wherein the SARS-CoV-2 infection is caused by a SARS-CoV-2 variant. Embodiment 46. The method of embodiment 45, wherein the SARS-CoV-2 variant is the U.K. (B.1.1.7) variant of SARS-CoV-2, the South African (B.1.351) variant of SARS-CoV-2, the California (B.1.429) variant of SARS-CoV-2, the California (B.1.427) variant of SARS- CoV-2, the Brazilian (P.1) variant of SARS-CoV-2, the New York (B.1.526) variant of SARS-CoV-2, the New York (B.1.526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2 or the Omicron (B.1.1.529) variant. Embodiment 47. The method of any one of embodiments 1-46, wherein the antibody or antigen binding fragment thereof treats SARS-CoV-2 variant and nonvariant infections with about equivalent efficacies. Embodiment 48. The method of any one of embodiments 1-47, wherein the administering of the antibody or antigen binding fragment thereof is intravenous. Embodiment 49. The method of any one of embodiments 1-17 and 22-48, wherein the first, second, and third antibodies or antigen binding fragments are administered sequentially or simultaneously. Embodiment 50. The method of any one of embodiments 1-17 and 22-49, further comprising combining each of a composition comprising the first antibody or antigen biding fragment thereof, a composition comprising the second antibody or antigen binding fragment thereof, and a composition comprising the third antibody or antigen binding fragment thereof prior to administration to the subject.
Embodiment 51. The method of any one of embodiments 1-50, wherein the antibody or antigen binding fragment thereof is recombinant. Examples [0179] Example 1. Evaluation of the breadth of patients’ humoral responses against SARS-CoV-2. The overall spectrum of the productive antibody response to SARS-CoV-2 was examined using an automated, high-throughput hybridoma library generation and screening platform [Puligedda et al. 2014] after isolating memory B cells acquired from blood samples of COVID-19 convalescent patients who demonstrated a high antibody titer to N and/or S proteins. [0180] Collection of patient samples. Blood samples were drawn from convalescing COVID-19 patient volunteers deemed eligible for donating convalescent plasma as set forth in the U.S. Food and Drug Administration (FDA)’s Recommendations [FDA 2020]. Patients displayed no PCR-detectable viremia and maximal IgG (2880) titer of class switched, virus- specific antibodies. [0181] Generation of hybridoma libraries. Hybridomas were generated from the memory B cells isolated from the donors by following protocols for isolating and expanding primary B cells as well as electrofusion methods described in U.S. patents [Dessain & Weinberg 2002, Dessain & Adekar 2009]. Hybridomas stably expressing human mAbs were generated by electrofusion of expanded B-cells to the B5-6T myeloma cell line, which expresses an ectopic human telomerase gene that stabilizes human chromosomes in the hybrid cells created. Fused hybridomas were plated into 96-well plates in growth medium with HAT selection of stable hybridomas for 7 days. After 7 days, growth media were switched to media with HT for stable selected hybridoma growth. Hybridomas were cultured in a 37°C incubator for 14-21 days during which time they were imaged for monoclonality and monitored for isotype-and sub-class-specific Ig secretion. Supernatants from monoclonal wells expressing measurable levels of Ig were cherry-picked and submitted for target-based screening. [0182] Screening Assays For Antiviral Antibodies. The naturally occurring human antibodies (IgM, IgG, and IgA isotypes) secreted by those hybridomas were screened for reactivity against a panel of SARS-CoV-2 proteins. Antibody screening assays were
developed for three SARS-CoV-2 structural proteins (S, N, M) and a panel of accessory ORF proteins of SARS-CoV-2. In addition to the reference Spike (S) protein corresponding to that found on the USA/WA_CDC-WA1/2020 virus, counterscreening was carried out on Spike proteins, or Spike protein domains, corresponding to the Spike proteins present on the surface of viral variants. The breadth of variants tested are described in Table 2. Table 2. SARS-CoV-2 Spike Screening Proteins SARS-CoV-2 Viral Tar et Assa S stem Variants Tested 453F, . n 4K, partial Y144, 69/70, R,
[0183] The screening assays included a rapid and sensitive homogeneous time resolved fluorescence (hTRF) assay that used soluble recombinant viral proteins, as well as a selective, cell-based flow cytometry assay that allowed probing of antibodies to transiently transfected viral antigens expressed within the context of human cells. Viral protein expression in the cell-based assay was additionally confirmed by Western blot. For cell-based assays, using commercially available controls, the localization of the C-terminus truncated Spike protein (SΔ19aa) was confirmed to be on the surface of the transfected cells. Commercially available antibodies specific for SARS-CoV-2 targets, and more specifically the Spike protein, demonstrated selective and saturable binding in both assays. Screening led to the isolation of antibodies specific for eight different viral proteins (FIG. 1). [0184] HTRF Screening Assays. A homogeneous time-resolved fluorescence (hTRF) assay [Degorce et al. 2009] comprised of terbium labeled anti-human IgG (H+L) (Cisbio, custom label) donor and AF488-labeled anti-HIS (Cell Signaling, Cat # 14930S) acceptor antibodies was used to screen patient-derived antibodies for their binding to recombinantly produced SARS-CoV-2 antigens, and more specifically to the SARS-CoV-2 Spike protein. The assay, adapted for high-throughput screening, was optimized so that a number of
recombinant, HIS-tagged SARS-CoV-2 target proteins could be substituted interchangeably. This recombinant target panel consisted of a full-length S (FL, trimer-stabilized, LakePharma) and two truncated S protein domains, S1 (GenScript, Cat # Z03485-1) and RBD (aa 319-591, LakePharma). Commercially available antibodies specific for the individual structural viral proteins, SARS-CoV/SARS-CoV-2 Spike S1 (RBD) chimeric mAb (Sino Biological, Cat # 40150-D001), SARS CoV-2 Nucleocapsid human chimeric mAb (GenScript, Cat # A02039-100 served as controls. Assay background was determined by averaging the signal of wells containing only the donor and acceptor cocktail. Hybridoma supernatants exhibiting signals greater than 2-fold over background were reported as positive HITs and are submitted for Ig sequence analysis. [0185] Flow cytometry-based cellular screens for antiviral antibodies. SARS-CoV-2 antigen sequences were cloned into pcDNA3.4 plasmids and transfected into 293F cells utilizing the Expi293 Expression System (Life Technologies, Inc.) per manufacturer’s instructions. Cells transiently expressing SARS-CoV-2 S (SΔ19aa) were used for screening. Optimal protein expression was achieved three days post-transfection for S proteins. [0186] For the cell-surface binding assays cells were incubated with LIVE/DEAD Cell Stain (Life Technologies, Inc.) per the manufacturer’s instructions. Live cells expressing SΔ19aa were suspended in QSol Buffer (IntelliCyte Corporation) to which a cocktail of Fc-specific secondary antibodies was added: AF647 goat anti-human IgG (Jackson ImmunoResearch, Inc.), AF488 goat anti-human IgA (Jackson ImmunoResearch, Inc.) and BV650 mouse anti-human IgM (BioLegend, Inc.). [0187] For each assay, the cell suspensions were dispensed into 384-well plates, followed by the addition of hybridoma supernatant at a 1:10 final dilution. The reaction was allowed to incubate for 90 minutes at room temperature. Cells for each assay were fixed with a final concentration of 1% paraformaldehyde and analyzed with IntelliCyte iQue Screener (IntelliCyte Corporation). Positive binding gates for detection of each secondary antibody were established using cells plus secondary antibody cocktail as a negative control. Binding of hybridoma supernatant antibodies to specific SARS-CoV-2 proteins, and more specifically the SΔ19a, were quantified as percent positive relative to the secondary only control. To calculate percent positive, live events that shifted into detection channels were divided by the total live events.
[0188] Example 2. Immunoglobulin gene usage in convalescent COVID-19 patients. Ig gene usage in memory B cells of six COVID-19 patients was evaluated using an NGS analysis of identified and sequenced 134 hybridoma hits. RNA isolation and Next Generation Sequencing (NGS). Hybridoma RNA was isolated using Invitrogen RNAqueous-96 Total RNA Isolation Kit (Cat. #AM1920). Isolated RNA samples were submitted to iRepertoire (Huntsville, AL) for NGS. Hybridoma-derived RNA samples were sequenced using the Illumina MiSeq system at iRepertoire (Huntsville, AL). Sequencing runs were performed using the MiSeq Nano Kit V2 following bead-based cleanup of RNA. Immunoglobulin sequences containing CDR1, 2 and 3 and framework regions were amplified using IgG and IgA-specific mixes for IgH, and kappa and lambda- specific primers for IgL. IgM-expressing hybridoma samples, from which IgG or IgA heavy chains were not amplified using this approach, were sequenced using the iRepertoire iPair system. Final sequences were exported using iPair software. [0189] Immunoglobulin sequences were analyzed for predicted CDR sequences, % identity to appropriate germlines, isotype of the constant regions and read counts. Sequence pairing was performed based on the read count information. In the event more than one LC:HC pair was discovered in a single well, each LC:HC combination was analyzed as a separate antibody. In all cases, sequencing method resulted in 5’ truncation in the VH and VL regions, the germline sequence for the closest related V gene was used to generate a full- length version of each gene. The gene sequences were codon optimized to create expression constructs for recombinant production and testing of antibodies. Final sequences were translated and analyzed for potential stop codons and frame shifts. [0190] Example 3. Recombinant production of antibodies. Heavy and light chain pairs from 103 clones, from which productive immunoglobulin RNA sequences were determined, were expressed as recombinant antibodies. [0191] Production of paired light and heavy chains. Variable domains yielding productive uninterrupted protein sequences were analyzed for number of reads and the degree of somatic hypermutations (SHM) in comparison to the closest immunoglobulin germline. Hybridoma hit sequences with at least one chain that had more than 2% of SHM were advanced to HC/LC pairing and the recombinant production of antibodies. Immunoglobulin expression fragments were cloned into the pcDNA3.4-based vectors and expressed in 293F
cells. Affinity and binding pattern of recombinant antibodies were compared to the original antibody-containing hybridoma supernatants in BLI, HTRF and cell-based assay. Antibody- containing supernatants or purified antibodies were advanced to downstream assays. If multiple heavy or light chain sequences were detected within one well, their CDRs were aligned and compared for potential PCR errors. In cases where multiple sequences within a well were different, i.e., originated from separate clones, all potential combinations of light and heavy chains were recombinantly produced and tested in downstream assays. Wells that yielded a single HC/LC pair were advanced to recombinant expression and downstream assays. 5’ fragments of the constant regions were sequenced to identify the isotype of the antibody and compared to the experimentally identified isotype of hybridoma supernatants. The resulting isotype of the heavy or light chain was assigned based on two or more positive readings from experimental (ex. ELISA and FACS) assays and sequencing. [0192] A combined analysis of Ig isotype and their level of SHM of virus-specific antibodies revealed several key properties of the productive antiviral response. First, among all “mutated” immunoglobulins that had more than 2% of their nucleotide sequence deviated from the closest germline, there was an unusually high (26.4%) proportion of mutated IgMs, having a mean SHM rate of 5.73%. The functional basis of this phenomenon is not known, but one could speculate that these IgMs came from non-switched memory B cells that had undergone affinity maturation. Second, a subset of such somatically hypermutated IgMs recognized full-length Spike, but not the soluble RBD or S1 subunit of Spike protein. And third, while the predominant isotype among class-switched antibodies was, as expected, IgG it was possible to capture a panel of fairly mutated virus-specific IgAs. It is plausible that these antibodies play a major role in mucosal neutralization of the incoming virus and may be of particular use for prophylaxis of viral infection and for vaccine design. [0193] Example 4. Identified anti-Spike antibodies from convalescent COVID-19 patients exhibit potent binding to Spike proteins mimicking a range of different viral variants. Purified recombinant antibodies were assessed, in the HTRF assay described above, using either soluble RBD or S1 domains containing mutations found in naturally occurring viral variants, as well as mutations predicted to decrease binding of neutralizing antibodies. Table 3 depicts the binding of identified anti-Spike antibodies, expressed as fold-binding over background. As anticipated, binding of some antibodies, such as PR201_00151 and PR194_00232, are negatively affected by specific mutations within the Spike domain (e.g.
K417N). In contrast, binding by antibodies like PR199_00255, PR193_00018, and PR200_00622 are unaffected by the range of mutations analyzed, including variants containing single point mutations, as well as multiple point mutations that mimic the naturally occurring Spike proteins found on the B.1.1.7 and B.1.351 virus isolates. A subset of antibodies, such as PR199_00255 and PR200_00622, also bind to Spike protein found on SARS-CoV-1, suggesting that they bind to a highly conserved epitope. [0194] Binding of the antibodies PR913_00018, PR194_00232, and PR200_00622 were further characterized in the HTRF assay to determine EC50 of binding to a wide range of single and multi-point mutations, including to the B.1.1.7, B.1.429, P.1, and B.1.351 variants. Each of the antibodies bound to the Wuhan/Washington reference Spike RBD protein with EC50s in the HTRF assay of between 45 – 68 pM. Mutations that mapped to the ACE2 binding site on the RBD appear to disrupt the binding affinity of PR199_00232, whereas no mutation, or combination of mutations, tested disrupted the ability of either PR193_00018 or PR200_00622 to bind to the RBD. The ability of these two antibodies to bind to the wide range of variants suggests that their epitopes are maintained even in the face of the viral drift that has occurred to date. [0195] The impact of mutational drift on the binding of each of PR193_0018, PR194_00232, and PR200_00622 antibodies was evaluated using a series of Spike proteins containing either single point mutations or complex point mutations that recapitulate the spectrum of mutations in naturally occurring variants that have emerged across the globe. PR194_00232 is the most sensitive of the three antibodies to mutational drift within the Spike protein. Mutations, such as K417N and N501Y, mutations known to exist in naturally occurring variants, and consistent with the alanine scanning data, significantly decrease the ability of PR194_00232 to bind the Spike protein. Whereas other naturally occurring mutations, such as L452R, E484Q, or D614G do not impact binding relative to binding to the Washington reference Spike protein. In contrast, binding of PR193_0018 and PR200_00622 are not significantly impacted by any of the single point mutations tested. In fact, binding by PR193_0018 and PR200_00622 to specific variant Spike proteins (e.g., E484K and E484Q) appears to be modestly enhanced over the reference Spike. Similar observations were made when binding of PR193_0018, PR194_00232, and PR200_00622 was tested against RBD domain proteins containing the full complement of mutations known to exist in four different variants. Of particular interest is the binding to RBDs corresponding to the South African
(B.1.351) and U.K. (B.1.1.7) variants. As predicted by the K417N and N501Y single point mutation data, binding of PR194_00232 to both the U.K. and S.A. variants is weaker than observed against the reference strain Spike protein.
Table 3. Binding of Antibodies to Variant Forms of Recombinant Spike Protein
DHV6 A222 N K, A Y G P 3 7 0 P 4 0 0 P 4 3 2 P 4 7 2 P 4 6 3 P 4 4 4 P 6 2 41 P 7 5 6 P 9 7 2 P 0 8 6 P 1_
9 7 9 6 9 8 0 151
Antibodies were evaluated for ability to bind to recombinant Spike protein in an HTRF assay, binding was expressed as a fold over background. Single point mutations (except D614G) were expressed in the context of soluble RBD domain, all others were expressed in context of soluble S1 domain. No binding was anticipated to RBD domain constructs by antibodies that bind to epitopes outside the RBD domain. Table 4. EC50 of Binding to Variant Forms of Recombinant Spike Protein. EC50 (pM) HTRF assay (relative to RBD) PR194_00232 PR200_00622 PR193_00018 Mutation Location IMM20190 IMM20253 IMM20184 REF RBD 68.2 52.7 44.8 N439K RBD 68.9 53.4 51.0 E406Q RBD 67.7 40.6 26.1 K417N RBD >500 67.4 43.7 N501Y RBD 279 58.5 39.8 Y453F RBD 64.6 62.6 49.4 A352S RBD 21.9 31.7 26.3 A475V RBD 72.4 33.0 27.9 E484Q RBD 52.4 23.8 21.6 E484K RBD 33.4 31.6 20.5 L452R RBD 31.8 34.0 43.6 K444R RBD 25.8 26.1 19.4 F486S RBD 33.4 25.9 22.1 K417N, E484K, RBD Inactive 19.7 14.5 N501Y K417T, E484K, RBD Inactive 23.6 18.3 N501Y REF S1 56.0 49.1 31.8 D614G S1 62.7 62.4 39.7 A222S, S1 49.1 44.9 26.9 D614G K417N, E484K, S1 230 24.8 26.7 N501Y, D614G ∆HV69/70, N501Y, S1 396 21.6 14.3 D614G ∆H69/70, ∆Y144, S1, RBD >500 62.4 45.5 N501Y, A570D,
D614G, P681H ∆H69/70, Y453F, S1, RBD 47.2 66.4 31.6 D614G [0196] Example 5. Identified anti-Spike antibodies bind to non-overlapping epitopes within the RBD of SARS-Co-V-2 Spike. Purified recombinant forms of the PR193_00018, PR194_00232, and PR200_00622, failed to compete with each other for binding to the SARS-CoV-2 RBD when assessed via biolayer interferometry on an Octet QKe instrument. These data suggested the three antibodies bind to non-overlapping epitopes on the SARS- CoV-2 RBD. Alanine scanning of the RBD was performed to identify residues critical for binding of each of the antibodies. Consistent with each of the antibodies binding to non- overlapping epitopes, non-overlapping sets of residues were identified as being critical for binding of the antibodies (Table 5). Those residues map to unique regions of three dimensional structure of the RBD, consistent with the ability of the antibodies to fail to compete with each other for binding (FIG. 2) Table 5. Identification of critical residues for Ab binding. M b n K N A F K S P K D L F N R Y N
Y489A 3.1 (4) 73 (6) 65 (12) 99.3 (21) 73.9 (1)
N501A 12.9 (1) 93.4 (5) 80.1 (14) 94.1 (8) 84.7 (1) Y
Mean binding reactivities (and ranges) are listed for all identified critical residues. Critical residues for Ab binding (boxed) were residues whose mutations were negative for binding to test Abs , but positive for binding to control Ab [0197] Example 6. Identified anti-Spike antibodies neutralize SARS-CoV-2 pseudovirus. The functional consequence of antibodies binding to Spike was assessed in assays using pseudotyped replication-incompetent lentivirus which were used to infect HEK293 cells overexpressing Angiotensin converting enzyme 2 (ACE2). Spike-expressing pseudovirus was generated with System Bioscience’s pPACK-SPIKE packaging system (System Biosciences, Cat #CVD19-500A-1) as per manufacturer’s protocol. Briefly, 8 x 106293TN Producer cells (System Biosciences, LV900A-1) were plated in T150 flasks overnight. Plasmids encoding lentiviral packaging proteins and Spike were added 1 mL of plain DMEM for each T150 being transfected. 55 mL of PureFection reagent (System Biosciences; Cat #LV750A-1) was added to each 1 mL tube, vortexed for 10 seconds, and incubated at room temperature for 15 minutes. The plasmid and PureFection mixture were added to a T150 flask containing 293TN cells and placed in a 37°C incubator containing 5% CO2 for 48 hours. Pseudovirus-containing supernatants were harvested at 48 hours and passed through a 0.45 micron PVDF filter to remove cellular debris. 5x PEG-it Virus Precipitation Solution (System Biosciences, Cat #LV810A-1) was added to supernatants and incubated 4°C overnight. Pseudovirus-containing supernatants with 1x PEG-it Virus Precipitation Solution were then spun at 1500 x g for 30 minutes. Pseudovirus-containing pellets were resuspended in plain DMEM to achieve at 10x concentration and frozen at -80°C in single use aliquots. [0198] Pseudovirus infection and neutralization assays were performed by using standard methods. Pseudoviruses were produced with the pPACK-SPIKE packaging kit, 104 ACE2- 293T cells were plated in the inner 60 wells of a white opaque 96 well flat bottom plate (Corning; Cat #3917) in 100 µL of ACE2-293T media overnight in a 37°C incubator containing 5% CO2. To determine infectivity of each lot of pseudovirus, pseudovirus- containing supernatants were thawed from -80°C and two-fold dilutions were performed. 100 mL of pseudovirus at various dilutions was added to ACE2-293T cells. To test neutralization activity of antibodies, indicated antibody concentrations were pre-incubated with pseudovirus for 1 hour in a 37°C incubator containing 5% CO2. Then, 100 µL of
antibody/pseudovirus mixture was added to ACE2-293T cells. After 72 hours, cells and media were equilibrated to room temperature for 20 minutes. 100 mL of media was removed and replaced with 100 µL of Bright-Glo Luciferase Assay Reagent (Promega, Cat #E2620). Luminescence was measured on the EnSpire Plate Reader (PerkinElmer). [0199] Initial testing with pseudovirus was performed using antibody-containing supernatants, 3-4 log dilutions of 26 unique anti-Spike antibodies were tested for their ability to block infection in comparison to an anti-RSV negative control and a commercially available anti-Spike positive control. [0200] Neutralization activity of a subset of antibodies (FIGS. 3A-3D) was confirmed using SARS-CoV-2 luciferase reporter virus particles (RVP’s) (Integral Molecular) based on the manufacturer’s instructions. In brief, RVP’s were thawed for 2-3 minutes in a 37°C water bath. The recommended amount of RVP’s was added to the inner wells of a white opaque 96 well plate (Corning; Cat #3917) or 384 well plate (Greiner Bio-One; Cat #781080). Media containing the indicated amount of antibody was added to each well, resulting in a final volume of 100 mL per well (96 well plate) or 25 mL per well (384 well plate). The antibody/RVP mixture was pre-incubated for 1 hour in a 37°C incubator containing 5% CO2. ACE2-293T target cells were added to each well (2 x 104 cells in 100 mL for a 96 well plate or 0.9 x 104 cells for a 384 well plate) and incubated for 72 hours. Media was removed from all wells, equal volumes of PBS and Renilla-Glo Luciferase Assay Reagent (Promega; Cat #E2720) were added to each well (60 mL total for a 96 well plate or 30 mL total for a 384 well plate). After 10 minutes, luminescence was measured on the EnSpire Plate Reader (PerkinElmer). Percent neutralization was calculated with the following equation: [(RLU of Virus + cells) – (RLU of Experimental Sample)] / [(RLU of Virus + cells) – (RLU of cells only)]. [0201] The PR194_00232 antibody that exhibited potent neutralization against pseudovirus expressing both the reference (SARS-CoV-2/human/USA/WA_CDC- WA1/2020) and D614G (SARS-CoV-2/human/Germany/BavPat 1/2020) were also assessed in neutralization assays using live virus of each of the isolates (FIGSs 4A and 4B). [0202] Full dose response of purified antibodies confirmed strong neutralizing activity by antibodies such as PR194_00232. The D614G variant is a widespread mutation [Plante et al. 2020] found in a number of different isolates. Antibodies such as PR194_00232, that bound
to the soluble protein and neutralized pseudovirus expressing the D614G Spike, were also able to neutralize the live virus with comparable IC50 values (FIG. 4B). [0203] Pseudovirus particles expressing Spike proteins that mimic the U.K. (B.1.1.7) and South African (B.1.351) isolates are neutralized by antibodies which retain binding to the mutated Spikes (Tables 3 &4). This is exemplified by the antibodies PR193_00018 and PR200_00622 (FIGS. 5A-5C). In contrast, antibodies that lose binding affinity for the mutated Spikes have a decreased neutralization potency; PR193_00232 retains neutralization activity against the UK variant pseudovirus but is unable to effectively neutralize the S. African pseudovirus. [0204] Example 7. A cocktail of three anti-SARS-CoV-2 anti-Spike antibodies elicit combinatorial effects. With the objective of producing an antibody composition that is able to neutralize both current, and future, variants, including, but not limited to, CDC Variants of Concern/Interest (alpha/B.1.1.7; beta/B.1.351; gamma/P.1; delta/B.1.617.2; epsilon/B.1.429/427), antibodies selective for non-overlapping epitopes on the SARS-CoV-2 Spike protein were assessed in pair-wise, and three-way combinations to identify additive, or preferably synergistic, neutralization. [0205] More specifically, the antibodies PR194_00232 (IMM20190), PR193_00018 (IMM20184), and PR200_00622 (IMM20253) were assessed in combinations to evaluate the combinatorial impact on neutralization of pseudovirus expressing a range of different variations of the Spike protein (FIGS. 6A-6C). IC50 values for the triple combination and double combination comprising IMM20184 and IMM20253 were determined using RVPs, as described above. [0206] As depicted in FIGS. 6AA-6C the triple combination of antibodies neutralized pseudoviruses corresponding to the USA/WA_CDC-WA1/2020 (reference sequence) and the CDC variants of concern (alpha/U.K./B.1.1.7, beta/South African/B.1.351, gamma/Brazil/P.1 and epsilon/California/B.1.429/427). In these pseudovirus assays, the IC50s for neutralization of the reference and California variants were not determined due to the potency exhibited against those variants; concentrations sufficiently low enough to obtain below 50% neutralization were not tested. Neutralization of the U.K., Brazilian, and South African were achieved with IC50s of 0.7 nM, 31 nM, and 23 nM, respectively, in these assays. The impact of IMM20190 on neutralization of the reference, CA, and UK variants can be observed by
comparing the IC50s achieved with the double combination of IMM20184/IMM20253 against the same pseudoviruses. In the absence of IMM20190, the reference California and U.K. variant pseudoviruses are neutralized with higher IC50s, corresponding to 37 nM, 15 nM, and 25 nM respectively. In the case of the S. African and Brazilian, the lack of IMM20190 does not dramatically alter the IC50s, consistent with IMM20184/IMM20253 providing the majority of the neutralization activity. [0207] To determine combinatorial impact of the antibodies pseudovirus neutralization experiments were set up as described above, except that for combinations of two antibodies, one test article was titrated in the background of each concentration in a serial dilution of the other test article. Single antibody titrations were included as controls. For combinations of three antibodies, one test article was titrated in the background of each concentration in a serial dilution of a 1:1 mixture of the other two test articles. To evaluate antibody synergy in the combinations, the observed combination response matrix of pseudovirus neutralization was used as input for the online SynergyFinder platform (4), where quadruplicate data points were input separately. The highest single agent (HSA) reference model was applied, which quantifies synergy as the excess over the maximum response of a single drug in the combination. Synergy between antibodies in each combination is reported as an overall synergy score (the average of observed synergy across the dose combination matrix) as well as a peak HSA score (the highest synergy score calculated across the dose combination matrix). Synergy scores of less than -10, between -10 and 10, and greater than 10 indicate antagonistic, additive, and synergistic antibody combinations, respectively. While peak HSA scores report on synergy at the most optimal combination concentrations, the overall synergy score is less affected by outlier data points. [0208] As depicted in FIGS. 7A-7C, the triple antibody combination of IMM20184/IMM20190/IMM20253, also known as IMM-BCP-01, synergized with each other to neutralize pseudovirus expressing the WA1/2020 reference, alpha/UK/B.1.1.7, Beta/S.African/B.1.351, Gamma/Brazil/P.1, and epsilon/Californian/B.1.429 spike proteins. Peak synergy scores ranged from 19.8 against the gamma variant to 61.1 against the alpha variant. Synergy was maintained against the alpha/UK variant across the entire concentration range tested. Overall additivity was observed against the other pseudoviruses. Synergy was observed against the live virus isolate Germany BavPat 1/2020 at defined ratios, with overall additivity being observed across the concentrations tested (FIG. 7B).
[0209] Example 8. Predicted activity against new variants. As the SARS-CoV-2 virus continues to mutate it will be imperative to predict, and confirm, the ability of antibodies to neutralize newly arising variants. As depicted in FIG. 8 the residues critical for binding of IMM20184, IMM20190, and IMM20253 are spatially distinct from the residues mutated in the RBD of the B.1.617.2 (delta), B.1.617.3 (kappa), and C.37 (lambda) variants. Based upon the location of the L452R T478K mutations in the B.1.617.2 variant, the L452R E484Q mutations in the B.1.617.3 variant, and the L452Q F490S mutations in the lambda variant, it was predicted that the triple antibody combination would be able to neutralize those variants in a manner at least equivalent to the reference WA1/2020 strain. [0210] Pseudovirus neutralization assays were performed with RVPs, as described above, that express the B.1.617.2, B.1.617.3, or C.37 variants. Consistent with the prediction, IMM- BCP-01 neutralized the pseudoviruses in a manner that was at least equivalent to the reference WA1/2020 variant (Table 6). Neutralization curves (FIG. 9). [0211] Activity of the triple antibody cocktail against the B.1.617.2 and B.1.617.3 variants is depicted in the context of other variants in FIG. 10. Consistent with the known epitopes of the antibodies, and positions of RBD-localized mutations in the different variants, the triple antibody cocktail exhibits neutralizing activity across all variants tested. [0212] IMM20190 binds to a large epitope that encompasses two distinct regions on the RBD (FIG. 2). K417 and N501, residues known to be mutated in different variants, represent residues in each of the two binding sites. Mutation of one of those sites (N501Y), as observed in the alpha/B.1.1.7 variant, is sufficient to maintain IMM20190 activity (FIGS. 5A-5C) and provides for improved synergy with IMM20184/IMM20253. The delta plus/B.1.617.2.ay1/2 variant contains a K417N mutation. As predicted by our understanding of the IMM20190 epitope, and borne out by the results depicted in Table 6, the K417N mutation causes a partial loss of neutralization potential, the magnitude of the effect is in line with that observed against the alpha. Mutation of both K417 and N501, as observed in the beta/B.1.351 and gamma/P.1 variants, more fully abrogates IMM20190 activity, and neutralization is achieved through the IMM20184/IMM20253 antibodies. Those are reflected in the results depicted FIG. 10 and the IC50/90 values derived from those data (Table 6). Table 6. IC50 & IC90 Values (nM) for IMM-BCP-01 Against Reference and Variants Pseudovirus Live Virus IC50 IC90 IC50 IC90
REF (WA1/2020) 1.0 5.2 1.1 3.4 D614G 0.9 4.2 NT NT Alpha 3.4 16.1 3.1 8.7 Beta 13.5 87.4 7.4 41.7 Gamma 24.8 129 NT NT Delta 0.4 2.3 NT NT Delta plus 3.0 15.2 NT NT Epsilon 0.6 3.4 NT NT Lambda 0.4 1.5 NT NT Kappa (L452/E484Q/D614G) 1.0 5.7 NT NT NT = Not Tested [0213] The broad panel of neutralization data, combined with our understanding of the IMM-BCP-01 epitopes, provides us with confidence on predicting the impact of any newly emerging variants on the efficacy of the IMM-BCP-01 cocktail. [0214] Example 9. Triple combination is active against live virus To confirm results obtained with pseudovirus testing we performed neutralization assays on four different live virus variants under BSL3 conditions: USA/WA_CDC-WA1/2020 (reference sequence), Germany/BavPat 1/2020 (D614G), UK (B.1.1.7), and South African (B.1.351). In all cases, neutralization of the live virus recapitulated data obtained in pseudovirus neutralization assays (FIGS. 11A-11D and Table 6). Assays using the Germany/BavPat1/2020 strain were performed in a manner sufficient to assess combinatorial effects via the HSA algorithm. Data demonstrated an overall additive effect, with peak HSA scores reaching levels of synergy (FIG. 7B) [0215] Example 10. Abs neutralize virus in a hamster model of SARS-CoV-2. Antibodies capable of neutralizing live virus in vitro were assessed for the ability to neutralize virus in vivo using a hamster model of SARS-CoV-2 infection. Hamsters treated with increasing doses of PR194_00232 were infected with SARS-CoV-2 (SARS-CoV- 2/human/USA/WA_CDC-WA1/2020) and viral load in the lungs were assessed at Day 4 post inoculation of the virus using standard tissue culture infectivity assays and plaque counting. PR194_00232 was able to neutralize virus, relative to no-treatment controls, in a dose- dependent manner when dosed in the prophylactic setting (FIG. 12). Consistent with PR193_00018 and PR200_00622 working combinatorially with PR194_00232 to clear the virus, combinations consisting of PR194_00232/PR193_0018 and
PR194_00232/PR200_00622 appear more effective at clearing the virus than PR194_00232 alone. [0216] When dosed in the therapeutic setting (FIG. 13), a roughly 3-log clearance of virus, relative to no treatment, was observed in 5 out of 6 animals treated with this antibody cocktail. In contrast, other dose groups exhibited a more variable response with only three animals in any other group ever reaching maximal clearance. These data suggest that optimal viral clearance requires all three antibodies to be part of the cocktail. [0217] In a follow-up study, the triple combination dosed at equimolar ratios (0.25 mg each) resulted in a statistically significant decrease in viral load in the lungs at day 4 post- inoculation, as measured with TCID50 assays (FIG. 14). To that end, in the same study doses of IMM20184 and IMM20253 were decreased to 0.125 mg each and we observed an increase in the median level of viral load in the lung compared to the cohort treated with the 1:1:1 ratio. These data provide additional support that IMM20184 and IMM20253 are contributing to in vivo efficacy, even in the context of the USA/WA_CDC-WA1/2020 viral isolate which is highly sensitive to neutralization by IMM20190 in vitro. [0218] Variability observed in the efficacy of IMM-BCP-01 in various studies led to retrospective correlation of systemic exposure post-intraperitoneal injection with overall viral clearance (FIG. 15). Syrian golden Hamsters were inoculated with 3.3x105 TCID50 doses of WA1/2020 live virus in the nasal turbinates (1.65 x 105 per nare). IMM-BCP-01 was administered i.p. 6 hours post-inoculation at three different dose levels. Dose levels were 0.1mg each (0.3mg total), 0.2mg each (0.6 mg total), or 0.3 mg each (0.9mg total). Animals were euthanized at Day 4 and viral titers in lung determined by TCID50 assays. Levels of IgG in serum at time of euthanasia were determined by anti-huIgG ELISA to correlate IgG exposure with overall viral clearance. Data support the idea that i.p. injections led to variable levels of exposure and correlated with viral clearance. Serum levels of approximately 3 – 5 ug/mL at day 4 were sufficient to achieve viral clearance and were obtained by all three dose levels tested. [0219] IMM-BCP-01 is able to clear virus from the lungs of hamsters infected with variants of concern. Hamsters were dosed prophylactically with IMM-BCP-01 one day prior to inoculation with either the Alpha (FIG. 16A) or Beta (FIG. 16B) variant and lung titers were determined on day 4 post-inoculation. IMM-BCP-01 at the lowest doses tested
provided levels of viral clearance that match, or exceed, those obtained by antibodies with demonstrated clinical efficacy. In addition, increased doses of IMM-BCP-01 led to a dose- dependent improvement in viral clearance, with > four-log clearance of WA1/2020 and 2.5 log clearance of Beta at the highest doses tested. All doses tested represent clinically relevant doses. [0220] Robust activity of the cocktail was observed regardless of inoculation titer used (FIG. 17). Syrian hamsters were inoculated with either 3.3x105 TCID50 doses of WA1/2020 live virus in the nasal turbinates (1.65 x 105 per nare) or 3.3x104 TCID50 doses of WA1/2020 live virus in the nasal turbinates (1.65 x 104 per nare). Animals were then treated 6 hours post-inoculation with either vehicle or IMM-BCP-01 at 0.1mg each dose level. Robust viral clearance was observed, relative to the vehicle control, for both inoculation levels. Table 7. Binding Constants of IMM20184, IMM20190, & IMM20253 for Isolated RBD and Trimer Ligand Analyte Kon (1/ms) Koff (1/s) KD (M) Trimer 3.12E+04 2.29E-04 7.35E-09 IMM20184 RBD 5.95E+04 1.31E-03 2.20E-09 Trimer 2.95E+04 1.95E-04 6.59E-09 IMM20190 RBD 2.02E+05 3.76E-04 1.87E-09 Trimer 8.13E+04 1.17E-04 1.44E-09 IMM20253 RBD 1.39E+06 2.18E-04 1.57E-10 Table 8. Phagocytosis Scores of Various anti-RBD Abs Score at 0.6 IMM # Target nM IMM20184 Spike (RBD) 56.6 IMM20190 Spike (RBD) 48.1 IMM20198 Spike (RBD) 37.9 IMM20242 Spike (RBD) 1.26 IMM20253 Spike (RBD) 49.5 IMM20254 Spike (RBD) 48.9 IMM20279 Spike (RBD) 43.3 Positive COM00035 50.1 Control Negative Anti-RSV 1.38 Control [0221] Example 11. Antibodies Elicit Mechanisms that Could Enhance Viral Neutralization and Clearance. Each of the antibodies comprising IMM-BCP-01 (IMM20184,
IMM20190, and IMM20253) bind to both isolated RBD and Spike trimer when assessed by surface plasmon resonance (FIGS. 18A-18C). These data suggest that IMM20184 can bind avidly to the Spike trimer, as signified by the decrease off-rate (Table 7), and crosslink two Spike monomers. [0222] Consistent with its epitope overlapping with the ACE2 binding site, IMM20190 is able to compete binding of ACE2 to isolated RBD protein from the REF and Alpha variant (FIG. 19A and FIG. 19B), but its ability to compete ACE2 binding to the Beta variant is decreased (FIG. 19C), consistent with its known decreased binding and neutralization potency. IMM20184 effectively competes binding of ACE2 to all three isolated RBD (FIGS. 19A – 19C), despite binding to an epitope that is outside of known ACE2 binding site (FIG. 2). Despite exhibiting neutralization activity against both pseudovirus and live virus, IMM20253 is unable to effectively compete for ACE2 binding (FIGS. 19A-19C). This suggests the neutralization is due to a mechanism distinct from direct ACE2 competition. [0223] IMM20184, IMM20190 and IMM20253 were assessed for the ability to fix complement using standard assays [Nikitin et al., 2019]. As depicted in FIGS. 20A-20B, all three antibodies comprising IMM-BCP-01 were able to fix complement, albeit to different levels when assessed at defined concentrations. All three antibodies demonstrated a preference for fixing complement upon binding to the stabilized trimer as compared to the isolated RBD. When assessed in a concentration-dependent manner (FIG. 20C), the combination of IMM20184 and IMM20253 induced an enhanced level of complement fixation beyond what was observed by either antibody alone. That activity was further enhanced by addition of IMM20190 to the antibody cocktail. These data suggesting the antibodies comprising IMM-BCP-01 elicit a combinatorial, or synergistic, activation of complement fixation upon binding Spike. [0224] Select antibodies were also assessed for the ability to phagocytosis at a defined concentration and using methods described previously [Shi et al., 2014]. As listed in Table 8, antibodies elicited a range of phagocytosis scores, with higher values representing a more robust ability to induce phagocytosis. Each of the antibodies comprising IMM-BCP-01 was further evaluated in the ability to induce phagocytosis in a concentration-dependent manner against the full-length trimer, both as individual antibodies and as cocktails of two or three antibodies (FIG. 21). All three antibodies induce phagocytosis of the full-length trimer in concentration-dependent manners, with IMM20253 inducing the most robust activation of
phagocytosis as assessed in vitro. IMM-BCP-01 exhibited enhanced activity, over a wider concentration range, as compared to any of the individual antibodies and the IMM20184/IMM20253 two antibody combination. [0225] IMM-BCP-01 induces a more robust antibody-dependent cellular cytotoxicity than any of the individual component antibodies when assessed in vitro using Promega’s ADCC Reporter Bioassay and S-expressing CHO-K1 target cells at a 2:1 effector:target cell ratio and manufacturer protocols. [0226] Taken together these data suggest that the IMM-BCP-01 cocktail robustly induces multiple effector functions in a manner that is enhanced by the presence of two or more of its constituent antibodies as compared to the individual antibodies alone. [0227] Example 12. IMM20253 may neutralize SARS-CoV-2 through a mechanism that alters Spike protein conformation. As depicted in FIG. 2, IMM20253 binds to an epitope that is on the outside face of the RBD domain of the SARS-CoV-2 RBD. When in the closed conformation, the epitope is in close proximity to the N-terminal domain of a second Spike protein within the Spike trimer. A recent paper [Sun et al, 2021] describes the isolation of nanobodies (Nb), defined as class III Nb, that bind to epitopes that appear to overlap with the IMM20253 epitope. Biochemical characterization the Class III Nb demonstrates that binding of the Nb to the Spike protein induces a conformational change to the post-fusion conformation. This presumably inactivates the virus’ ability to bind cells and provides a mechanism for the neutralization observed in vitro. Like the Class III Nbs, IMM20253 is unable to directly compete for ACE2 binding (FIGS. 20A-20C), but is able to neutralize both reference and alpha variant live virus as a single agent when measured as a function of virus internalization (Table 9). Together, these data suggest that IMM20253 binding to the outer face of the RBD may impart its intrinsic neutralization through induction of a conformational change in the Spike protein that prevents virus uptake into the cells without competing for ACE2 binding. Interestingly, IMM20253 appears to be approximately 30-times more potent against the alpha strain than the delta strain. This may be a function of the internalization kinetics of the two different variants. This difference may also underlie the strong synergy observed between the IMM20184/IMM20190/IMM20253 in the context of neutralizing the alpha variant (FIGS. 7A-7C). It should be noted that like Greaney et al [Greany e al], Sun et al describe the region around the IMM20253 epitope as being of therapeutic interest, one to which antibodies are not known to exist [Greany et al] and one that is going to be difficult for
antibodies to access [Sun et al]. Consistent with these assertions, the CoVIC consortium analyzed over 300 antibodies that bind to the RBD domain of the SARS-CoV-2 Spike protein and failed to identify an antibody that bound to the IMM20253 epitope. These points all highlight the uniqueness of the IMM20253 antibody [Hastie et al]. [0228] As outlined in FIGS. 23A-23B, binding of IMM20253, but not IMM20190, induces a time-dependent conformational changed in the trimeric Spike protein. Dynamic light scattering (DLS) analysis performed at t = 0 and t = 2 hours, demonstrates that IMM20253 binding results in the complex adopting a conformation with a smaller hydrodynamic radius. In contrast, the IMM20190/Spike trimer complex maintains a similar hydrodynamic radius throughout the two hour incubation. [0229] The conformational change induced by IMM20253 results in an increased protease sensitivity of the Spike protein (FIG. 24). Cleavage of Spike, when incubated in the presence of thrombin protease, is observed at t = 1 hour. Similar results are observed when Spike is complexed with either ACE2 or IMM20190. In contrast, proteolytic cleavage of Spike, and release of S2, is observed within 15 minutes when Spike is complexed with the IMM20253. Without being bound by theory, this data suggests that the IMM20253 antibody is particularly effective against variants that have pre-cleaved Spike proteins and/or variants that are more susceptible to cleavage. Table 9. Neutralization capacity of all single, double, and triple combinations of IMM20184, IMM20190, and IMM20253 in a virus internalization assay. an 90 co M) IM .7 IM .7 IM 95 Co 62 (I IM Co 28 (I
IMM20253)
Combo 3 VC- SARS-CoV-2 (UK Alpha B.1.1.7 0.85 8.49 16.62 (I IM Co 80 (I IM IM 94 IM 06 IM 21 Co 50 (I IM Co 50 (I IM Co 34 (I IM Co 81 (I IM
[0230] Example 13. IMM20253, alone or in combination with IMM20184, induces synergy with other ACE2-competitive antibodies. Binding of either IMM20253, IMM20184, or the combination of the two antibodies (IMM20253/IMM20184), induces combinatorial effects, preferably synergy, when combined with the ACE2-competitive antibody IMM20190. Combinations of IMM20253, IMM20184, or IMM20253/IMM20184, induces synergy with other ACE2-competing enzymes. IMM20253, IMM20184, or IMM20253/IMM20184 combines with one or more of the following antibodies to induce combinatorial, preferably synergistic, viral neutralization in vitro and promote in vivo viral clearance. Examples of antibodies that combine to induce the combinatorial effect with IMM20253, IMM20184 or IMM20253/IMM20184 include, but are not limited to, sotrovimab, casirivimab, imdevimab, bamlanivimab, etesevimab, tixagevimab, cilgavimab, ADG2, ADG10, ADG20, ADG30, and CR3022. As depicted in FIGS. 25A-25C, IMM20253 works in combination with IMM20184, as well as in-house generated versions of REGN987 (imdevimab) and REGN933 (casirivimab) when assayed against B.1.617.2 ay2 pseudovirus.
Under the conditions tested all three combinations displayed synergistic neutralization at defined concentrations achievable in vivo (HSA score >10). Under the conditions tested, the IMM20253/IMM20184 combination achieved an overall HSA score indicative of synergy (12.5). IMM20253/REGN933 achieved an overall HSA score of 9.5, suggestive of additivity (HSA score between -10 and 10). The IMM20253/REGN933 combination demonstrated an overall HSA score of 5.6, consistent with additive activity under the conditions tested. All three antibodies (IMM20184, REGN933 and REGN987) block ACE2 binding. In contrast, IMM20253 in combination with in-house generated S309 (sotrovimab) resulted in strong antagonism across all concentrations tested (FIG. 26). S309 is thought to work through a non- ACE2 dependent neutralization mechanism. This suggests that IMM20253’s non-ACE2 dependent neutralization mechanism may enhance neutralization in combination with antibodies that work via an ACE2-dependent neutralization mechanism, but may antagonize other non-ACE2 dependent mechanisms of neutralization. [0231] Example 14. IMM20253 and IMM20279 and exhibit potent binding to the Spike protein of the Omicron variant. Individual antibodies were assessed, in the HTRF assay described above, using the full length Spike protein and soluble RBD domains of the Spike protein containing mutations found in the Omicron variant. FIGS. 27A-27D depict the binding of the individual antibodies to the Spike-RBD of the Omicron variant, expressed as percentage binding, relative to the binding to the reference strain. Binding of the antibodies IMM20190 and IMM20184 (FIGS. 27A and 27B), are negatively affected by the mutations within the Spike domain of the Omicron variant. In contrast, binding by the IMM20253 (FIG. 27C) and IMM20279 (FIG. 27D) antibodies are unaffected by the range of mutations within the Spike protein of the Omicron variant. The binding affinity of the IMM20279 antibody to the RBD as well as the full-length Spike protein is comparable to that of the reference strain (FIG. 27D). [0232] Example 15. IMM20253 antibody neutralizes SARS-CoV-2 pseudovirus expressing the Omicron variant. The functional consequence of the IMM20253 antibody binding to Spike was assessed. Pseudovirus infection and neutralization assays were performed by using standard methods described above. The IMM20253 antibody exhibited potent neutralization against pseudovirus expressing the Spike protein from the B.1.1.529 (Omicron) variant (FIG. 28). The neutralization activity was comparable to the activity against pseudovirus expressing the Spike protein from the reference strain (SARS-CoV-
2/human/USA/WA_CDC-WA1/2020), D614G (SARS-CoV-2/human/Germany/BavPat 1/2020), B.1.351 (beta/S. African), B.1.617.2 Ay.2 (Delta Plus) variants of SARS-CoV-2 (FIG. 28). IMM20253 exhibited equivalent neutralization of psuedovirus expressing the spike of either the BA.1 or BA.2 sub-lineage of the Omicron variant (FIG. 29). [0233] Example 16. IMM20184 and IMM20279 act to enhance IMM20253 activity in vivo. Hamsters were dosed prophylactically with IMM20253 (0.3 mg, 1 mg, or 3 mg), IMM20253/IMM20184 (0.5 mg each or 1.5 mg each), or IMM20253/IMM20279 (1.5 mg each) one day prior to inoculation with the Omicron BA.1 (FIG. 36) variant and lung titers were determined on day 4 post-inoculation by plaque forming assays. IMM20253 exhibited a dose-dependent decrease in lung titer. While IMM20184 showed a substantial decrease in in vitro binding of omicron Spike protein relative to REF (FIG. 27A) and IMM20279 also showed decreased binding of omicron Spike protein relative to REF (FIG. 27D), the combinations of the combination of IMM20253/IMM20184 and IMM20253/IMM20279 exhibits more robust clearance of Omicron BA.1 in vivo. These data suggest that the combination of IMM20253 and IMM20184 and the combination of IMM20253 and IMM20279 act combinatorially in vivo to clear the virus (FIG. 36). [0234] Example 17. CryoEM structures of IMM20184, IMM20290, IMM20253, and IMM20279 in complex with the SARS-CoV-2 spike protein. The structures of antibodies in complex with the intact trimer of the SARS-CoV-2 trimer were solved by cryoEM to a resolution of approximately 7 Angstroms (FIGS 30A-30B, FIGS. 31A-31B, FIGS. 32A-32B, and FIGS. 34A-34B). The cryoEM structures corroborated the binding sites for IMM20184, IMM20190, and IMM20253 that were determined by alanine scanning mutagenesis (Table 5). IMM20279 binds to a site that appears to overlap with the binding site of IMM20184. [0235] Three Fabs of IMM20190 bind simultaneously to the trimer in a manner consistent with the Class 1 binders as defined by the CoVIC consortium and induce all three RBDs to adopt the up conformation (FIGS. 30A-30B). Three Fabs of IMM20184 bind simultaneously to the spike trimer, perpendicular to the vertical axis of the spike. Binding appears to induce a large conformational change in the spike trimer, particularly in the RBD domains. A similar reorganization of the spike trimer is observed in the cryoEM structure obtained for the IMM20279/Spike trimer complex (FIGS. 35A-35B). IMM20279 binds to similar regions on RBD as compared to IMM20184, but does so by binding from a different
angle of attack. It binds at approximately a 45 degree angle to the vertical axis of the spike as compared to the 90 degree angle adopted by IMM20184. [0236] Only a single FAb of IMM20253 is ever observed to bind to the intact trimer (FIGS. 32A-32B). Binding results in loss of density in the cryoEM structure associated with the RBD and N-terminal domain of spike monomer that is not bound by the Fab fragment. This loss of density is consistent with destabilization of those structures upon Fab binding. Consistent with destabilization of the trimer, monomeric S protein is observed in the cryoEM imaging. Increasing the ratio of Ab:Spike from 3:1 to 6:1 results in almost complete loss of intact spike. These data provide structural evidence that support the biochemical data demonstrating increased protease sensitivity upon IMM20253, but not IMM20190 binding. Destabilization of the spike may provide the basis for neutralization through a non-ACE2 competitive mechanism. [0237] Higher resolution cryoEM structures (approximately 3. 9 Angstroms) of IMM20184 and IMM20253 Fab fragments in a trimeric complex with an isolated RBD were solved to better define the epitopes of those two antibodies. As depicted in FIG. 33 and FIGS. 34A-34B the structures of the ternary complex are in agreement with those obtained with the intact trimer. Residues within 4 Angstroms of the variable domains of each Fab are depicted in spheres. Those residues include all residues identified by alanine scanning. Residues buried by IMM20253 interaction with the RBD are conserved in the Omicron variant and provide additional basis for continued activity of this antibody against the variant. [0238] Example 18 Phase 1 Study to Evaluate the Safety, Pharmacokinetics, and Viral Clearance of Single Ascending Doses of IMM-BCP-01 Administered Intravenously in Adults with Mild to Moderate COVID [0239] This is a randomized, double-blind, placebo-controlled Phase 1 study to evaluate the safety, PK, and viral clearance of single ascending doses of IV IMM-BCP-01 in subjects with mild to moderate COVID-19 caused by infection with SARS-CoV-2 and/or its variants. [0240] The primary objective of this study is to evaluate the safety and tolerability of intravenous (IV) IMM-BCP-01 in subjects with mild to moderate COVID-19 through Day 28.
[0241] The secondary objectives of the study are to: Determine PK and evaluate viral clearance after single ascending doses of IV IMM-BCP-01 in subjects with mild to moderate COVID-19 through Day 28. Evaluate the safety and tolerability, determine PK, and evaluate viral clearance of single ascending doses of IV IMM-BCP-01 in subjects with mild to moderate COVID-19 through Week 12. [0242] The study consists of a screening period of up to 36 hours, a treatment period of one day, two further clinic visits, four visits that take place either in the clinic or at the subject's home, six virtual visits (telephone or video), and an end-of-study visit 12 weeks (+/- 5 days) after study drug dosing. The total duration of a subject's participation is approximately 90 days. [0243] Subjects presenting at the clinic with signs and symptoms of mild to moderate COVID-19 and agreeing to participate in the study are screened, and if deemed eligible for the study, are randomized (2:1) to receive a single IV dose of IMM-BCP-01 or placebo on Day 1. Subjects at risk of severe disease and those who have been vaccinated against COVID-19 within 6 weeks prior to screening or who have received monoclonal antibodies against SARS-CoV-2 and/or COVID-19 convalescent plasma at any time are not eligible. Subjects are randomized to receive IMM-BCP-01 or placebo. Table 10 Phase 1 Trial Design - -
Drug: Placebo
Placebo matching single dose of IMM-BCP- -
Primary Outcome Measure is: 1. Incidence and severity of Treatment Emergent Adverse Events (TEAEs) (Time Frame: up to 28 days) a. TEAEs include clinical laboratory values, standard 12-lead ECGs, vital signs, pulse oximetry Secondary Outcome Measures are: 1. Incidence and severity of Treatment Emergent Adverse Events (TEAEs) (Time Frame: up to 12 weeks) 2. PK parameters measured by maximum observed concentration, time to maximum observed concentration, terminal elimination half-life, clearance, volume of distribution (Time frame: up to 28 days) 3. PK parameters measured by maximum observed concentration, time to maximum observed concentration, terminal elimination half-life, clearance, volume of distribution (Time frame: up to 12 weeks) 4. Anti-drug antibody change from baseline (Time frame: 28 days) 5. Anti-drug antibody change from baseline (Time frame: 12 weeks) 6. Viral clearance change from baseline (Time frame: 28 days) 7. Viral clearance change from baseline (Time frame: 12 weeks) Inclusion Criteria are: 1. Male or female 18 to 50 years of age, inclusive, at the time of signing the informed consent.
2. Subjects must have mild to moderate COVID-19 with symptom onset within 5 days prior to study drug administration (see Appendix Error! Reference source not found. for Food and Drug Administration [FDA] severity guidance). Subjects whose symptoms began >5 days (i.e. ˃120 hours) prior to dosing or whose time of symptom onset cannot be accurately assessed are not eligible. 3. Subjects must have at least 2 of the following COVID-19 symptoms: fever, cough, sore throat, rhinorrhea, malaise, headache, muscle pain, nausea, vomiting, diarrhea, and loss of taste or smell, or other symptoms that the Principal Investigator judges to be referrable to COVID-19. 4. Subjects must be able to maintain oxygen saturation (SpO2) ≥ 94% on room air (no supplemental oxygen). 5. Body mass index ≥ 18.0 and ≤ 30.0 kg/m2. 6. Body weight ≥ 40 kg at screening. 7. Sexually active subjects of reproductive potential must agree to use highly effective contraception from signing of the informed consent through 90 days after infusion of the study drug (see Section Error! Reference source not found.). 8. Males must agree not to donate sperm from dosing until 90 days after administration of the study drug. 9. Subjects must have been in generally good health, as judged by the Principal Investigator, prior to onset of current COVID-19 illness, with no clinically significant medical history. 10. Subjects must be without clinically significant abnormalities as assessed by review of medical and surgical history, physical examination, vital signs measurement, ECG, and laboratory evaluations conducted at screening. Exclusion criteria are: 1. Has one or more symptoms suggestive of more severe illness with COVID-19 (see Appendix Error! Reference source not found. for FDA severity guidance) and/or requires hospitalization.
2. Is asymptomatic at screening or randomization, regardless of a positive COVID- 19 test. 3. Is at increased risk of severe COVID-19 for any reason including but not limited to: cancer (basal cell carcinoma and prostate carcinoma in situ [Gleason ≤ 6] are acceptable), chronic kidney disease, chronic obstructive pulmonary disease, heart condition (congestive heart failure II, III and IV as per New York Heart Association: coronary disease and any other cardiac condition that imposes high risk of developing severe COVID-19), immunocompromised state from solid organ transplant, sickle cell disease, or other condition, autoimmune disease, use of immunosuppressants (including high doses of systemic corticosteroids), type 1 or type 2 diabetes mellitus,current or prior history of smoking or vaping any product, including nicotine or THC. 4. Has any active infection, other than the underlying COVID-19. 5. Has been admitted to a hospital within 3 months prior to randomization (except for planned admissions for minor procedures). 6. Has been hospitalized due to COVID-19 at any time. 7. Has participated or is participating in a clinical research study currently or within 3 months or less than 5 half-lives of the investigational product (whichever is longer) prior to the screening visit. 8. Has received monoclonal antibodies against SARS-CoV-2 and/or COVID-19 convalescent plasma. 9. Is anticipated to be treated with any approved or investigational drug or agent against SARS-CoV-2 (other than the study drug) during the study including antiviral drug(s), antibodies, or convalescent plasma. 10. Has received any COVID-19 directed treatment in the 3 months prior to the screening visit including but not limited to: Intravenous immunoglobulin, Approved drugs or products used off label for treatment of COVID-19, Other experimental interventions.
11. Has received a COVID-19 vaccination within 6 weeks of screening. Subjects vaccinated ≥ 6 weeks prior to screening and who otherwise meet inclusion/exclusion criteria are eligible to participate. 12. History or suspicion of excessive alcohol use (defined as drinking on average 14 drinks a week for males and 7 drinks a week for females) or of binge drinking (defined as 4 drinks on any day for males and 3 drinks on any day for females, for 5 or more days in the past month) 13. History of substance abuse or current use of any drugs of abuse 14. Any other condition or prior therapy which the Principal Investigator feels may jeopardize the safety of the subject or the objectives of the study. References Berlin DA, Gulick RM, Martinez FJ. Severe Covid-19. N Engl J Med. 2020 [cited 5 Nov 2020]. doi:10.1056/nejmcp2009575 Bonsignori, M., Pollara, J., Moody, M.A., Alpert, M.D., Chen, X., Hwang, K.-K., Gilbert, P.B., Huang, Y., Gurley, T.C., Kozink, D.M., et al. (2012). Antibody-dependent cellular cytotoxicity-mediating antibodies from an HIV-1 vaccine efficacy trial target multiple epitopes and preferentially use the VH1 gene family. J. Virol. 86, 11521–11532. Braun J, Loyal L, Frentsch M, Wendisch D, Georg P, Kurth F, et al. SARS-CoV-2-reactive T cells in healthy donors and patients with COVID-19. Nature. 2020. doi:10.1038/s41586-020- 2598-9 Callaway BE. The coronavirus is mutating — does it matter? Nature.2020;585. Available: https://www.nature.com/articles/d41586-020-02544-6 Cao Y, Su B, Guo X, Sun W, Deng Y, Bao L, et al. Potent neutralizing antibodies against SARS-CoV-2 identified by high-throughput single-cell sequencing of convalescent patients’ B cells. Cell. 2020. doi:10.1016/j.cell.2020.05.025 Chakraborty I, Maity P. COVID-19 outbreak: Migration, effects on society, global environment and prevention. Sci Total Environ. 2020;728: 138882. doi:10.1016/j.scitotenv.2020.138882 Dessain, S.K., and Chumakov, K. (2014). Human monoclonal antibodies that neutralize vaccine and wild-type poliovirus strains. Antiviral Res. 108, 36–43.
Degorce, F., Card, A., Soh, S., Trinquet, E., Knapik, G.P., Xie, B. (2009) HTRF: A technology tailored for drug discovery - A review of theoretical aspects and recent applications. Current Chemical Genomics, 22–32. Dessain, S.K.., and Weinberg, R.A. (2002). US7491530B2. Dessain, S.K., and Adekar, S.P. (2009). US8999707B2. Editorial. Progress report on the coronavirus pandemic. Nature. 2020;584: 325-325. Available: https://media.nature.com/original/magazine-assets/d41586-020-02414-1/d41586- 020-02414-1.pdf FDA (2020). Guidance for Industry: FDA’s Recommendations for Investigational COVID-19 Conavalescent Plasma. Fehr, A.R., and Perlman, S. (2015). Coronaviruses: An overview of their replication and pathogenesis. In Coronaviruses: Methods and Protocols, (Springer New York), pp. 1–23. Ferretti AP, Kula T, Wang Y, Nguyen DMV, Weinheimer A, Dunlap GS, et al. Unbiased Screens Show CD8+ T Cells of COVID-19 Patients Recognize Shared Epitopes in SARS- CoV-2 that Largely Reside outside the Spike Protein. Immunity. 2020 [cited 5 Nov 2020]. doi:10.1016/j.immuni.2020.10.006 Greany AJ, Loes AN, Crawfor KHD, Starr TN, Malone KD, Chu HY, and Bloom JD. Comprehensive mapping of mutations in the SARS-CoV-2 receptor binding domain that affect recognition by polyclonal human plasma antibodies. Cell Host Microbe. 2021; 29: 463- 476 Grifoni A, Weiskopf D, Ramirez SI, Mateus J, Dan JM, Moderbacher CR, et al. Targets of T Cell Responses to SARS-CoV-2 Coronavirus in Humans with COVID-19 Disease and Unexposed Individuals. Cell. 2020;181: 1489-1501.e15. doi:10.1016/j.cell.2020.05.015 Hastie KM, Li H, Bedinger D, Schendel SL, Dennison SM, Li K, et al Defining Variant- resistant Epitopes Targeted by SARS-CoV-2 Antibodies: A Global Consortium Study. Science. 2021. doi10.1126/science.abh2315 Jacobsen, H.J., Poulsen, T.T., Dahlman, A., Kjær, I., Koefoed, K., Sen, J.W., Weilguny, D., Bjerregaard, B., Andersen, C.R., Horak, I.D., et al. (2015). Pan-HER, an antibody mixture simultaneously targeting EGFR, HER2, and HER3, effectively overcomes tumor heterogeneity and plasticity. Clin. Cancer Res. 21, 4110–4122. Korber B, Fischer WM, Gnanakaran S, Yoon H, Theiler J, Abfalterer W, et al. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID- 19 Virus. Cell. 2020;182: 812-827.e19. doi:10.1016/j.cell.2020.06.043
Lan, J., Ge, J., Yu, J., Shan, S., Zhou, H., Fan, S., Zhang, Q., Shi, X., Wang, Q., Zhang, L., et al. (2020). Structure of the SARS-CoV-2 Spike receptor-binding domain bound to the ACE2 receptor. Nature 581, 215–220. Levites Y, O’Nuallain B, Puligedda RD, Ondrejcak T, Adekar SP, Chen C, et al. A human monoclonal IgG that binds Aβ assemblies and diverse amyloids exhibits anti-amyloid activities in vitro and in vivo. J Neurosci. 2015;35: 6265–6276. doi:10.1523/JNEUROSCI.5109-14.2015 Li Q, Wu J, Nie J, Zhang L, Hao H, Liu S, et al. The Impact of Mutations in SARS-CoV-2 Spike on Viral Infectivity and Antigenicity. Cell. 2020;182: 1284-1294.e9. doi:10.1016/j.cell.2020.07.012 Morens DM, Fauci AS. Emerging Pandemic Diseases: How We Got to COVID-19. Cell. Cell Press; 2020. pp. 1077–1092. doi:10.1016/j.cell.2020.08.021 Nikitin, P.A., Rose, E.L., Byun, T.S., Parry, G.C., and Panicker, S. (2019). C1s Inhibition by BIVV009 (Sutimlimab) Prevents Complement-Enhanced Activation of Autoimmune Human B Cells In Vitro. J. Immunol. 202, 1200–1209. Puligedda, R.D., Kouiavskaia, D., Adekar, S.P., Sharma, R., Devi Kattala, C., Rezapkin, G., Bidzhieva, B., Dessain, S.K, and Chumakov, K. Human monoclonal antibodies that neutralize vaccine and wild-type poliovirus strains. Antiviral Res. 2014;108: 36–43. Puligedda, R.D., Sharma, R., Al-Saleem, F.H., Kouiavskaia, D., Velu, A.B., Kattala, C.D., Prendergast, G.C., Lynch, D.R., Chumakov, K., and Dessain, S.K. (2019). Capture and display of antibodies secreted by hybridoma cells enables fluorescent on-cell screening. MAbs 11, 546–558. Puligedda, R.D., Vigdorovich, V., Kouiavskaia, D., Kattala, C.D., Zhao, J., Al-Saleem, F.H., Chumakov, K., Sather, D.N., and Dessain, S.K. (2020). Human IgA Monoclonal Antibodies That Neutralize Poliovirus, Produced by Hybridomas and Recombinant Expression. Antibodies 9, 5. Robbiani, D.F., Gaebler, C., Muecksch, F., Lorenzi, J.C.C., Wang, Z., Cho, A., Agudelo, M., Barnes, C.O., Finkin, S., Hagglof, T., et al. (2020). Convergent Antibody Responses to SARS-CoV-2 Infection in Convalescent Individuals. BioRxiv 2020.05.13.092619. Shi, J., Rose, E.L., Singh, A., Hussain, S., Stagliano, N.E., Parry, G.C., and Panicker, S. (2014). TNT003, an inhibitor of the serine protease C1s, prevents complement activation induced by cold agglutinins. Blood 123, 4015–4022.
Stamper CT, Dugan HL, Li L, Asby NW, Halfmann PJ, Guthmiller JJ, et al. Distinct B cell subsets give rise to antigen-specific antibody responses against SARS-CoV-2. Res SquareResearch Sq. 2020. Available: https://europepmc.org/articles/PMC7523131 Sun D, Sang Z, Kim YJ, Xiang Y, Cohen T, Belford AK, Huet A, Conway JF, Sun J, Taylor DJ, Scheidman-Duhovny D, Zhang C, Huang W, Shi Y. Potent neutralizing nanobodies resist convergent circulating variants of SARS-CoV-2 by targeting diverse and conserved epitopes. Nat. Comm. 12, 4676. Tursi, S.A., Puligedda, R.D., Szabo, P., Nicastro, L.K., Miller, A.L., Qiu, C., Gallucci, S., Relkin, N.R., Buttaro, B.A., Dessain, S.K., et al. (2020). Salmonella Typhimurium biofilm disruption by a human antibody that binds a pan-amyloid epitope on curli. Nat. Commun. 11. Von Holle, T.A., and Anthony Moody, M. (2019). Influenza and antibody-dependent cellular cytotoxicity. Front. Immunol. 10, 1–8. WHO Coronavirus Disease (COVID-19) Dashboard | WHO Coronavirus Disease (COVID- 19) Dashboard. [cited 9 Nov 2020]. Available: https://covid19.who.int/?gclid=Cj0KCQiA7qP9BRCLARIsABDaZzhj4p4n5emnuHTO24B5 Wi-N10YYvhUbGPsXFht_Cmcim1HjG7fLuV0aAn_iEALw_wcB Zhou, P., Yang, X. Lou, Wang, X.G., Hu, B., Zhang, L., Zhang, W., Si, H.R., Zhu, Y., Li, B., Huang, C.L., et al. (2020). A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature 579, 270–273.
Claims
CLAIMS 1. A method of treating a SARS-CoV-2 infection in an immunocompromised subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV- 2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
2. A method of treating a SARS-CoV-2 infection in an immunocompromised subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV- 2, wherein the first antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 independent mechanism and the second antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism.
3. A method of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof binds to an epitope in an ACE2 receptor binding site of the Spike protein and the second antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
4. A method of treating a SARS-CoV-2 infection in a subject, comprising administering to the subject at least first and second antibodies or antigen binding fragments thereof that specifically bind to distinct epitopes of a Spike protein of SARS-CoV-2, wherein the first antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 independent mechanism and the second antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism.
5. The method of any one of claims 1-4, further comprising administering a third antibody or antigen binding fragment thereof that specifically binds to the Spike protein of SARS-CoV-2 at an epitope distinct from the first and second antibodies or antigen binding fragments thereof.
6. The method of claim 3, wherein the third antibody or antigen binding fragment thereof binds to an epitope outside of the ACE2 receptor binding site of the Spike protein.
7. The method of claim 5 or claim 6, wherein the third antibody or antigen binding fragment thereof neutralizes SARS-CoV-2 via an ACE2 dependent mechanism.
8. The method of any one of claims 5-7, wherein each of the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 600 mg to about 2400 mg.
9. The method of claim 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 600 mg.
10. The method of claim 9, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 200 mg each.
11. The method of claim 8, wherein first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 1200 mg.
12. The method of claim 11, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 400 mg each.
13. The method of claim 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 1800 mg.
14. The method of claim 13, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 600 mg each.
15. The method of claim 8, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a total dose of about 2400 mg.
16. The method of claim 15, wherein the first, second, and third antibodies or antigen binding fragments thereof are administered at a dose of about 800 mg each.
17. The method of any one of claims 5-8, wherein the first, second and third antibodies or antigen binding fragments thereof are administered at a dose of about 3 mg/kg, or about 6 mg/kg each.
18. A method of treating a SARS-CoV-2 infection in an immunocompromised subject, comprising administering to the subject an antibody or antigen binding fragment that binds to the Spike protein of SARS-CoV-2, wherein the antibody or antigen binding fragment comprises a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region (HCVR) comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 2; a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 4; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 6; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 7, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 8; a HCDR1, HCDR2, and HCDR3 of a heavy chain variable region comprising the amino acid sequence set forth in SEQ ID NO: 9, and a LCDR1, LCDR2, and LCDR3 of a light chain variable region (LCVR) comprising the amino acid sequence is set forth in SEQ ID NO: 10; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 11, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 12;
a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 13, a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 14; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 15, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO:16 a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 17, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO:18; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 19, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 20; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 21, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 22; a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 23, and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 24; or a HCDR1, HCDR2, and HCDR3 of a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 25 and a LCDR1, LCDR2, and LCDR3 of a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 26.
19. The method of claim 18, wherein the antibody or antigen binding fragment thereof comprises a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 1, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 2; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 3, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 4; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 5, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 6; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 7, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 8; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 9, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 10;
a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 11, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 12; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 13, a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 14; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 15, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 16; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: SEQ ID NO:17, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 18; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 19, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 20; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 21, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 22; a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 23, and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 24; or a HCVR comprising the amino acid sequence set forth in SEQ ID NO: 25 and a LCVR comprising the amino acid sequence is set forth in SEQ ID NO: 26.
20. The method of claim 18, wherein the antibody or antigen binding fragment thereof comprises a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60; a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66; a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72; or
a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114.
21. The method of any one of claims 18-20, comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66; b) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72;
c) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114; d) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72; e) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the
amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114; or f) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114.
22. The method of any one of claims 5-20 comprising administering to the subject a) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72;
b) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114; or c) a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, and a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, and a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the
amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114.
23. The method of claim 18 comprising administering to the subject a first antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:55, a HCDR2 comprising the amino acid sequence of SEQ ID NO:56, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:57; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:58, a LCDR2 comprising the amino acid sequence of SEQ ID NO:59, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:60, a second antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:61, a HCDR2 comprising the amino acid sequence of SEQ ID NO:62, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:63; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:64 a LCDR2 comprising the amino acid sequence of SEQ ID NO:65, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:66, a third antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:67, a HCDR2 comprising the amino acid sequence of SEQ ID NO:68, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:69; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:70, a LCDR2 comprising the amino acid sequence of SEQ ID NO:71, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:72, and a fourth antibody or antigen binding fragment thereof comprising a VH comprising a HCDR1 comprising the amino acid sequence of SEQ ID NO:109, a HCDR2 comprising the amino acid sequence of SEQ ID NO:110, and a HCDR3 comprising the amino acid sequence of SEQ ID NO:111; and a VL comprising a LCDR1 comprising the amino acid sequence of SEQ ID NO:112, a LCDR2 comprising the amino acid sequence of SEQ ID NO:113, and a LCDR3 comprising the amino acid sequence of SEQ ID NO:114.
24. The method of any one of claims 1-23, wherein the subject has a high risk for contracting SARS-CoV-2.
25. The method of any one of claims 1-24, wherein the subject is being treated with an immunosuppressant.
26. The method of claim 25, wherein the immunosuppressant is a corticosteroid.
27. The method of any one of claims 1-26, wherein the subject has received a solid organ transplant or has a chronic inflammatory disease.
28. The method of claim 27, wherein the subject has a chronic inflammatory disease selected from the group consisting of rheumatoid arthritis, psoriasis, and Crohn’s disease.
29. The method of any one of claims 1-24, wherein the subject is being treated with a chemotherapeutic agent or radiation.
30. The method of any one of claims 1-29, wherein the subject is at risk for developing severe COVID-19.
31. The method of any one of claims 1-24 or 30, wherein the subject has diabetes, a chronic pulmonary condition, a chronic cardiovascular condition, obesity, or is pregnant.
32. The method of any one of claims 1-31, wherein the viral load in the subject is decreased upon treatment.
33. The method of any one of claims 1-32, wherein administration of the antibody or antigen-binding fragment thereof results in viral clearance in the subject.
34. The methods of any one of claims 1-33, wherein the method results in viral neutralization.
35. The method of any one of claims 1-17 and 21-34, wherein the ratio between the first and the second antibodies or antigen binding fragments thereof is about 1 : 1.
36. The method of any one of claims 1-17 and 22-34, wherein the ratios between the first, second, and third antibodies or antigen binding fragments thereof are about 1 : 1 : 1.
37. The method of claim 23, wherein the ratio between the first, second, third, and fourth antibodies or antigen binding fragments thereof is about 1 : 1 : 1: 1.
38. The method of any one of claims 1-37, wherein the antibody or antigen binding fragment therefor is an Fc IgG1, IgG2, IgG3, IgG4, IgM, IgD, IgA1, IgA2 or IgE isotype.
39. The method of claim 38, wherein the antibody is an IgG1 isotype.
40. The method of claim 39, wherein the IgG1 is a G1m1 or nG1m1 allotype.
41. The method of any one of claims 1-40, wherein the antibody is a fully human antibody.
42. The method of any one of claims 1-41, wherein the antibody is a full length antibody.
43. The method of any one of claims 1-42, wherein binding of the antibody or antigen- binding fragment thereof: inhibits binding of a SARS-CoV-2 virus to a host ACE2 receptor; fixes complement to a SARS-CoV-2 virus; induces phagocytosis of a SARS-CoV-2 virus; or any combination thereof.
44. The method of any one of claims 1-43, wherein the binding of the antibody or antigen-binding fragment thereof neutralizes a SARS-CoV-2 virus by blocking binding of the receptor binding domain (RBD) of the virus with an ACE2 receptor.
45. The method of any one of claims 1-44, wherein the SARS-CoV-2 infection is caused by a SARS-CoV-2 variant.
46. The method of claim 45, wherein the SARS-CoV-2 variant is the U.K. (B.1.1.7) variant of SARS-CoV-2, the South African (B.1.351) variant of SARS-CoV-2, the California (B.1.429) variant of SARS-CoV-2, the California (B.1.427) variant of SARS-CoV-2, the Brazilian (P.1) variant of SARS-CoV-2, the New York (B.1.526) variant of SARS-CoV-2,
the New York (B.1.526.1) variant of SARS-CoV-2, the UK/Nigeria (B.1.525) variant of SARS-CoV-2, the Brazilian (P.2) variant of SARS-CoV-2 or the Omicron (B.1.1.529) variant.
47. The method of any one of claims 1-46, wherein the antibody or antigen binding fragment thereof treats SARS-CoV-2 variant and nonvariant infections with about equivalent efficacies.
48. The method of any one of claims 1-47, wherein the administering of the antibody or antigen binding fragment thereof is intravenous.
49. The method of any one of claims 1-17 and 22-48, wherein the first, second, and third antibodies or antigen binding fragments are administered sequentially or simultaneously.
50. The method of any one of claims 1-17 and 22-49, further comprising combining each of a composition comprising the first antibody or antigen biding fragment thereof, a composition comprising the second antibody or antigen binding fragment thereof, and a composition comprising the third antibody or antigen binding fragment thereof prior to administration to the subject.
51. The method of any one of claims 1-50, wherein the antibody or antigen binding fragment thereof is recombinant.
Applications Claiming Priority (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202163178848P | 2021-04-23 | 2021-04-23 | |
| US63/178,848 | 2021-04-23 | ||
| US202163220881P | 2021-07-12 | 2021-07-12 | |
| US63/220,881 | 2021-07-12 | ||
| US202163236479P | 2021-08-24 | 2021-08-24 | |
| US63/236,479 | 2021-08-24 | ||
| US202163270665P | 2021-10-22 | 2021-10-22 | |
| US63/270,665 | 2021-10-22 | ||
| US202163284963P | 2021-12-01 | 2021-12-01 | |
| US63/284,963 | 2021-12-01 | ||
| PCT/US2022/070026 WO2022150809A1 (en) | 2021-01-05 | 2022-01-04 | Antibody cocktail against sars-cov-2 spike protein |
| USPCT/US2022/070026 | 2022-01-04 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022226539A1 true WO2022226539A1 (en) | 2022-10-27 |
Family
ID=83722707
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2022/071875 Ceased WO2022226539A1 (en) | 2021-04-23 | 2022-04-22 | Methods of administering antibodies against sars-cov-2 spike protein |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2022226539A1 (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060246071A1 (en) * | 2004-12-21 | 2006-11-02 | Larry Green | Antibodies directed to angiopoietin-2 and uses thereof |
| US20140017244A1 (en) * | 2012-07-13 | 2014-01-16 | Roche Glycart Ag | Bispecific Anti-VEGF/Anti-ANG-2 Antibodies and their use in the Treatment of Ocular Vascular Diseases |
| US20170137522A1 (en) * | 2009-11-24 | 2017-05-18 | Medimmune Limited | Targeted binding agents against b7-h1 |
| US20200239550A1 (en) * | 2017-10-13 | 2020-07-30 | Adimab, Llc | Anti-respiratory syncytial virus antibodies, methods of their generation and use |
| CN111592595A (en) * | 2020-04-27 | 2020-08-28 | 南京医科大学 | Neutralizing antibody against novel coronavirus SARS-Cov-2 and application thereof |
-
2022
- 2022-04-22 WO PCT/US2022/071875 patent/WO2022226539A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060246071A1 (en) * | 2004-12-21 | 2006-11-02 | Larry Green | Antibodies directed to angiopoietin-2 and uses thereof |
| US20170137522A1 (en) * | 2009-11-24 | 2017-05-18 | Medimmune Limited | Targeted binding agents against b7-h1 |
| US20140017244A1 (en) * | 2012-07-13 | 2014-01-16 | Roche Glycart Ag | Bispecific Anti-VEGF/Anti-ANG-2 Antibodies and their use in the Treatment of Ocular Vascular Diseases |
| US20200239550A1 (en) * | 2017-10-13 | 2020-07-30 | Adimab, Llc | Anti-respiratory syncytial virus antibodies, methods of their generation and use |
| CN111592595A (en) * | 2020-04-27 | 2020-08-28 | 南京医科大学 | Neutralizing antibody against novel coronavirus SARS-Cov-2 and application thereof |
Non-Patent Citations (1)
| Title |
|---|
| HUANG ET AL.: "Breadth and function of antibody response to acute SARS-CoV-2 infection in humans", PLOS PATHOG, vol. 17, no. 2, 26 February 2021 (2021-02-26), pages 1 - 26, XP055983158 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12216120B2 (en) | Human monoclonal antibodies to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) | |
| US20240150439A1 (en) | Neutralizing Anti-SARS-CoV-2 Antibodies and Methods of Use Thereof | |
| CA2703667C (en) | Anti-rsv g protein antibodies | |
| US12410241B2 (en) | Human monoclonal antibodies to enterovirus D68 | |
| EP3134111B1 (en) | Middle east respiratory syndrome coronavirus neutralizing antibodies and methods of use thereof | |
| CN1914226B (en) | Antibodies against SARS-COV and methods of use thereof | |
| CN104892753B (en) | Antibody neutralizing human infection with H7N9 influenza A virus and application thereof | |
| US20240101646A1 (en) | Sars-cov-2 coronavirus antibodies and uses thereof | |
| KR20230127305A (en) | Neutralizing monoclonal antibody to COVID-19 | |
| KR20160117608A (en) | Antibody molecules to dengue virus and uses thereof | |
| CA3250276A1 (en) | Antigen binding molecules targeting sars-cov-2 | |
| US20240067704A1 (en) | Antibody cocktail against sars-cov-2 spike protein | |
| AU2015217149A1 (en) | Novel full spectrum anti-dengue antibody | |
| US12534517B2 (en) | Highly specific zika neutralizing human antibodies | |
| JP2025507595A (en) | Antibodies capable of binding to the spike protein of coronavirus SARS-CoV-2 | |
| WO2022226539A1 (en) | Methods of administering antibodies against sars-cov-2 spike protein | |
| US20240209065A1 (en) | Secretory iga antibodies against covid infection | |
| US20250304659A1 (en) | HUMAN MONOCLONAL ANTIBODIES TO SEVERE ACUTE RESPIRATORY SYNDROME CORONAVIRUS 1 (SARS-CoV-1) | |
| HK40113917A (en) | Human monoclonal antibodies to severe acute respiratory syndrome coronavirus 2 (sars-cov-2) | |
| WO2024196463A2 (en) | Broadly neutralizing human monoclonal antibodies that target the sars-cov-2 receptor binding domain (rbd) | |
| HK40080049A (en) | Human monoclonal antibodies to severe acute respiratory syndrome coronavirus 2 (sars-cov-2) | |
| HK40080049B (en) | Human monoclonal antibodies to severe acute respiratory syndrome coronavirus 2 (sars-cov-2) |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22792715 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 22792715 Country of ref document: EP Kind code of ref document: A1 |