WO2022066883A1 - Extracellular vesicles comprising kras antigens and uses thereof - Google Patents
Extracellular vesicles comprising kras antigens and uses thereof Download PDFInfo
- Publication number
- WO2022066883A1 WO2022066883A1 PCT/US2021/051720 US2021051720W WO2022066883A1 WO 2022066883 A1 WO2022066883 A1 WO 2022066883A1 US 2021051720 W US2021051720 W US 2021051720W WO 2022066883 A1 WO2022066883 A1 WO 2022066883A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- amino acids
- aspects
- composition
- cell
- moiety
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/0005—Vertebrate antigens
- A61K39/0011—Cancer antigens
- A61K39/001154—Enzymes
- A61K39/001164—GTPases, e.g. Ras or Rho
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6901—Conjugates being cells, cell fragments, viruses, ghosts, red blood cells or viral vectors
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5063—Compounds of unknown constitution, e.g. material from plants or animals
- A61K9/5068—Cell membranes or bacterial membranes enclosing drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/555—Medicinal preparations containing antigens or antibodies characterised by a specific combination antigen/adjuvant
- A61K2039/55511—Organic adjuvants
- A61K2039/55555—Liposomes; Vesicles, e.g. nanoparticles; Spheres, e.g. nanospheres; Polymers
Definitions
- the present disclosure relates to engineered extracellular vesicles (EVs) (e.g., exosomes) comprising a KRAS antigen, and the use of such EVs to treat a disease or disorder, including cancer.
- EVs engineered extracellular vesicles
- the present disclosure also relates to methods of producing such EVs (e.g., exosomes).
- Kirsten rat sarcoma viral oncogene homology is a member of a superfamily of guanosine-5-triphosphatase (GTPase) proteins that also includes NRAS and HRAS. KRAS mutations have been implicated in many types of cancers, including more than 90% of pancreatic cancers, 35-45% of colorectal cancers, and approximately 25% of lung cancers. Zeitouni, D., et al., Cancers 8(4): 45 (2016); Tan, C., et al., World J Gastroenterol 18(37): 5171-5180 (2012); and Roman, M., et al., Molecular Cancer 17:33 (2016).
- composition comprising (i) an isolated extracellular vesicle (EV) comprising a KRAS antigen, and (ii) an adjuvant.
- EV extracellular vesicle
- adjuvant an adjuvant.
- the composition further comprises a T helper peptide.
- the KRAS antigen comprises an amino acid substitution selected from G12D, G12C, G12V, G13D, G12A, G12R, G12S, G13C, G13A, GBR, G13S, G13V, Q61H, Q61L, Q61R, Q61K, A18D, K117N, or any combination thereof corresponding to SEQ ID NO: 381.
- the amino acid substitution of the KRAS antigen is selected from G12D, G12C, G12V, G13D, and any combination thereof corresponding to SEQ ID NO: 381.
- the KRAS antigen comprises a natural peptide, synthetic peptide, or both.
- the KRAS antigen is less than about 50 amino acids in length, less than about 45 amino acids in length, less than about 40 amino acids in length, less than about 35 amino acids in length, less than about 30 amino acids in length, less than about 25 amino acids in length, less than about 20 amino acids in length, less than about 15 amino acids in length, or less than about 10 amino acids in length.
- the KRAS antigen is between about 12 to about 20 amino acids in length.
- the KRAS antigen comprises a concatemer of multiple KRAS antigens.
- the multiple KRAS antigens are separated by a spacer.
- the spacer comprises the amino acid sequence AAY, GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), or both.
- the KRAS antigen comprises or consists of an epitope of amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids in length.
- the KRAS antigen comprises or consists of an epitope of amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length.
- the KRAS antigen comprises or consists of an epitope of amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids,
- the KRAS antigen is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- T helper peptide is associated with the EV.
- the T helper peptide is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- the T helper peptide is not associated with the EV.
- the T helper peptide comprises a universal T helper peptide .
- the universal T helper peptide comprises a PADRE, tetanus toxin, diphtheria toxin, HBV peptide, measles peptide, or any combination thereof.
- the tetanus toxin is a tetanus toxin P2, tetanus toxin P30, or both.
- the diphtheria toxin is CRM-197.
- the HBV peptide is HbsAg.
- the T helper peptide is a KRAS peptide comprising an epitope for a CD4+ T cell ("KRAS CD4+ T cell peptide").
- the T helper peptide comprises a concatemer of multiple T helper peptides. In certain aspects, the multiple T helper peptides are separated by a spacer.
- the adjuvant is associated with the EV.
- the adjuvant is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- the adjuvant is not associated with the EV.
- the adjuvant comprises a STING agonist, TLR agonist, or both.
- the STING agonist comprises a cyclic dinucleotide STING agonist or a non-cyclic dinucleotide STING agonist.
- the TLR agonist comprises a TLR2 agonist (e.g.
- lipoteichoic acid atypical LPS, MALP-2 and MALP-404, OspA, porin, LcrV, lipomannan, GPI anchor, lysophosphatidylserine, lipophosphoglycan (LPG), glycophosphatidylinositol (GPI), zymosan, hsp60, gH/gL glycoprotein, hemagglutinin), a TLR3 agonist (e.g., double-stranded RNA, e.g., poly(I:C), ampligen, hiltonol, polyA:U), a TLR4 agonist (e.g.
- LPS lipopolysaccharides
- lipoteichoic acid [3-defensin 2, fibronectin EDA, HMGB 1, snapin, tenascin C, MPLA), a TLR5 agonist (e.g. , flagellin), a TLR6 agonist, a TLR7/8 agonist (e.g. , single- stranded RNA, , Poly GIO, Poly G3, Resiquimod, Imiquimod, 3M-052), aTLR9 agonist (e.g., unmethylated CpG DNA), or any combination thereof.
- TLR5 agonist e.g. , flagellin
- TLR6 agonist e.g. , TLR6 agonist
- TLR7/8 agonist e.g. , single- stranded RNA, , Poly GIO, Poly G3, Resiquimod, Imiquimod, 3M-052
- aTLR9 agonist e.g.,
- the composition comprising an EV, which comprises a KRAS antigen further comprises one or more additional moieties selected from an immune modulator, targeting moiety, or any combination thereof.
- the immune modulator and/or targeting moiety are associated with the EV.
- the immune modulator, targeting moiety, and/or anti -phagocytic moiety are: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- the immune modulator and/or targeting moiety are not associated with the EV.
- the immune modulator comprises (i) an inhibitor for a negative checkpoint regulator or an inhibitor for a binding partner of a negative checkpoint regulator (e.g., anti-CTLA4 antibody); (ii) an activator for a positive co-stimulatory molecule or an activator for a binding partner of a positive co-stimulatory molecule (e.g., CD40L); (iii) a cytokine or a binding partner of a cytokine; (iv) a protein that supports intracellular interactions required for germinal center responses; (v) a polynucleotide; or (vi) any combination thereof.
- a negative checkpoint regulator e.g., anti-CTLA4 antibody
- an activator for a positive co-stimulatory molecule or an activator for a binding partner of a positive co-stimulatory molecule e.g., CD40L
- a cytokine or a binding partner of a cytokine e.g., CD40L
- the targeting moiety binds to a marker expressed on an immune cell (e.g., dendritic cell, T cell, B cell, or any combination thereof).
- the marker expressed on an immune cell comprises a C-type lectin domain family 9 member A (Clec9a) protein, a dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN), CD207, CD40, Clec6, dendritic cell immunoreceptor (DCIR), DEC-205, lectin-like oxidized low-density lipoprotein receptor- 1 (LOX-1), MARCO, Clecl2a, DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin-1, macrophage mannose receptor (MMR), BDCA-1 (CD303, Clec4c), Dectin-2, Bst-2 (CD317), CD3, CD19, CDla, CDl l
- Clec9a C-
- the targeting moiety binds to a marker expressed in a lymphoid tissue of a subject suffering from a cancer.
- the lymphoid tissue comprises a tumor draining lymph node, sentinel lymph node, tumor tertiary lymph node, or any combination thereof.
- the targeting moiety comprises anti -fibronectin EDA & EDB domain antibodies; anti-ICAM-1; anti-CD20; anti-DC-LAMP; or any combination thereof.
- the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties is linked to the exterior surface and/or luminal surface of the EV by an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), or any combination thereof.
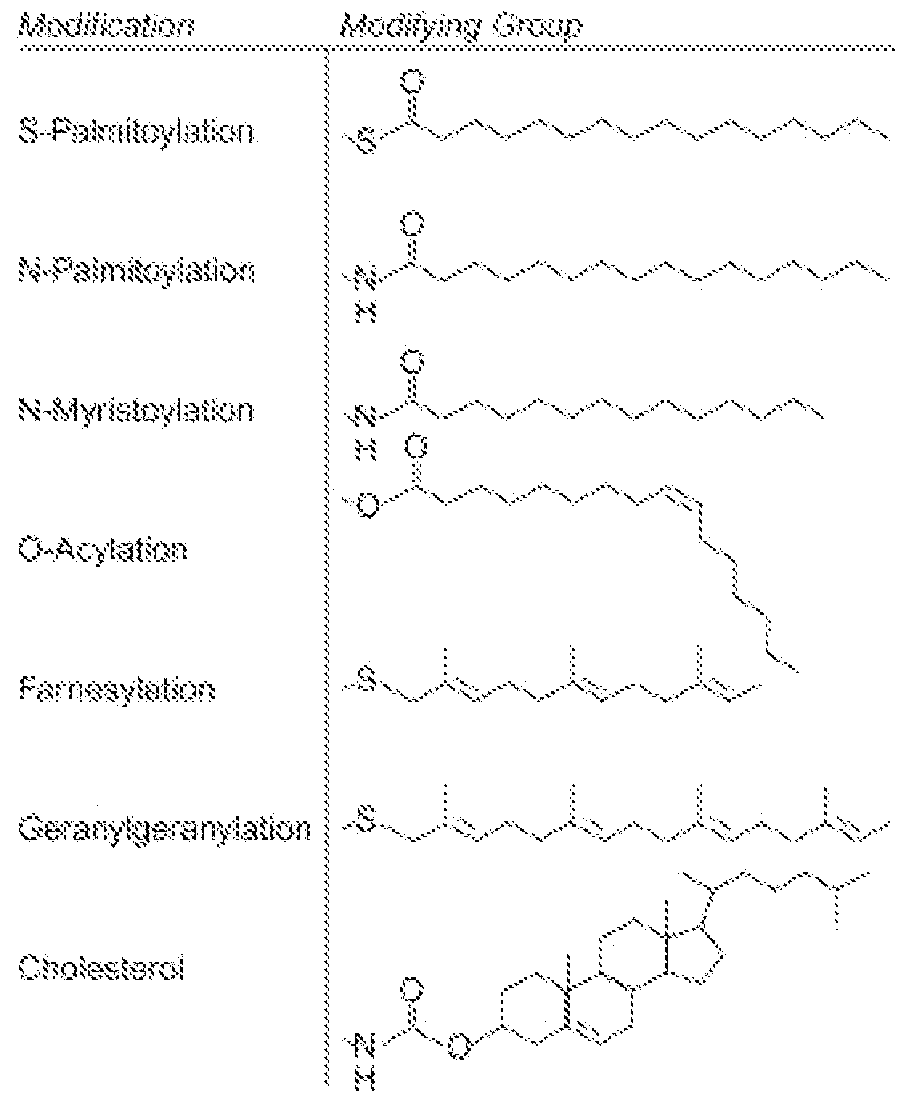
- the anchoring moiety comprises a cholesterol, fatty acid (e.g., palmitate), tocopherol (e.g., vitamin E), or any combination thereof.
- the chemical conjugation comprises a maleimide moiety, copper-free, biorthogonal click chemistry (e.g., azide/strained alkyne (DIFO, DBCO, BCN)), metal- catalyzed click chemistry (e.g., CUAAC, RUAAC), or any combination thereof.
- a composition disclosed herein i.e., comprising an EV, which comprises a KRAS antigen
- the KRAS antigen is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV;
- the T helper peptide is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV;
- the adjuvant is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV;
- the one or more additional moieties are linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; or (v) any combination thereof.
- the KRAS antigen is linked to the one or more scaffold moieties by a linker;
- the T helper peptide is linked to the one or more scaffold moieties by a linker;
- the adjuvant is linked to the one or more scaffold moieties by a linker;
- the one or more additional moieties are linked to the one or more scaffold moieties by a linker; or (v) any combination thereof.
- the linker is a polypeptide. In some aspects, the linker is a non-polypeptide moiety. In some aspects, the linker comprises a maleimide moiety. In some aspects, the linker comprises a cholesterol moiety. In some aspects, the linker comprises an ALFA-tag moiety.
- the one or more scaffold moieties comprise a Scaffold X, Scaffold Y, or both.
- the Scaffold X comprises a prostaglandin F2 receptor negative regulator (the PTGFRN protein); basigin (the BSG protein); immunoglobulin superfamily member 2 (the IGSF2 protein); immunoglobulin superfamily member 3 (the IGSF3 protein); immunoglobulin superfamily member 8 (the IGSF8 protein); integrin beta-1 (the ITGB1 protein); integrin alpha-4 (the ITGA4 protein); 4F2 cell-surface antigen heavy chain (the SLC3A2 protein); a class of ATP transporter proteins (the ATP1A1, ATP1A2, ATP1A3, ATP1A4, ATP1B3, ATP2B1, ATP2B2, ATP2B3, ATP2B4 proteins), or any combination thereof.
- the Scaffold X is PTGFRN or a fragment thereof.
- the Scaffold Y comprises myristoylated alanine rich Protein Kinase C substrate (the MARCKS protein); myristoylated alanine rich Protein Kinase C substrate like 1 (the MARCKSL1 protein); brain acid soluble protein 1 (the BASP 1 protein), and any combination thereof.
- the Scaffold Y is BASP 1 or a fragment thereof.
- the EV is an exosome.
- a pharmaceutical composition comprising any of the EVs described herein, and a pharmaceutically acceptable carrier. Also disclosed herein is a cell that produces any of the EVs of the present disclosure. Provided herein is a kit comprising the EV of the present disclosure. Present disclosure further provides an EV -drug conjugate comprising any of the EVs described in the present disclosure.
- Present disclosure provides a method of making EVs comprising culturing a cell describe herein under a suitable condition and obtaining the EV.
- method of making EVs comprising loading an EV that has been isolated from a producer cell with the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties disclosed herein.
- Present disclosure additionally provides methods of making a cancer vaccine comprising loading an EV with the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties disclosed herein.
- the EV is isolated from a producer cell prior to the loading of the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties.
- a vaccine produced by a method disclosed herein.
- Also provided herein is a method of inducing a vaccine in a subject afflicted with a cancer, comprising administering the vaccine of the present disclosure. Also provided herein is a method of preventing or treating a cancer in a subject in need thereof, comprising administering the composition or the vaccine described herein to the subject.
- the cancer comprises a colorectal cancer, lung cancer (e.g., non-small cell lung cancer (NSCLC)), pancreatic cancer (e.g., pancreatic ductal adenocarcinoma), leukemia, uterine cancer, ovarian cancer, bladder cancer, bile duct cancer, gastric cancer, stomach cancer, testicular cancer, esophageal cancer, cholangiocarcinoma, cervical cancer, acute myeloid leukemia (AML), diffuse large B-cell lymphoma (DLBC), sarcoma, melanoma, glioma (e.g., low-grade glioma, e.g., glioblastoma), mesothelioma, liver cancer, breast cancer (e.g., breast invasive carcinoma), renal carcinoma (e.g., papillary renal cell carcinoma (pRCC), and chromophobe renal cell carcinoma), head and neck cancer, prostate cancer, adenoid cyst
- FIG. 1 shows an exemplary EV (e.g., exosome) comprising (i) a KRAS antigen, (ii) a T helper peptide, (iii) an adjuvant, and (iv) a targeting moiety, one or more antigens.
- Certain aspects of the present disclosure are directed to an engineered extracellular vesicle (EV) (e.g., exosome) comprising a KRAS antigen.
- EV extracellular vesicle
- the present disclosure is directed to an engineered EV, e.g., exosome, comprising one or more payloads, wherein the one or more payloads can improve at least one property (e.g., such as those disclosed herein) of the EV, and uses thereof.
- the EVs (e.g., exosomes) disclosed herein are capable of targeting an immune cell (e.g., macrophage or dendritic cell) within the central nervous system of a subject.
- the one or more payloads that can be expressed in an EV (e.g., exosome) disclosed herein comprise an antigen (e.g., associated with a neurological disorder disclosed herein), an adjuvant, an immune modulator, or combinations thereof.
- the one or more payloads can be attached (or linked) to one or more scaffold moieties on the surface of EVs, e.g., exosomes, or on the luminal surface of EVs, e.g., exosomes.
- the EVs (e.g., exosomes) can further comprise a targeting moiety, which can also be attached (or linked) to one or more of the scaffold moieties disclosed herein.
- a targeting moiety which can also be attached (or linked) to one or more of the scaffold moieties disclosed herein.
- a or “an” entity refers to one or more of that entity; for example, “a nucleotide sequence,” is understood to represent one or more nucleotide sequences.
- the terms “a” (or “an”), “one or more,” and “at least one” can be used interchangeably herein.
- extracellular vesicle refers to a cell-derived vesicle comprising a membrane that encloses an internal space.
- Extracellular vesicles comprise all membranebound vesicles (e.g., exosomes, nanovesicles) that have a smaller diameter than the cell from which they are derived.
- extracellular vesicles range in diameter from 20 nm to 1000 nm, and can comprise various macromolecular payload either within the internal space (i.e., lumen), displayed on the external surface of the extracellular vesicle, and/or spanning the membrane.
- the payload can comprise nucleic acids, proteins, carbohydrates, lipids, small molecules, and/or combinations thereof.
- an extracellular vehicle comprises a scaffold moiety.
- extracellular vesicles include apoptotic bodies, fragments of cells, vesicles derived from cells by direct or indirect manipulation (e.g., by serial extrusion or treatment with alkaline solutions), vesiculated organelles, and vesicles produced by living cells (e.g., by direct plasma membrane budding or fusion of the late endosome with the plasma membrane).
- Extracellular vesicles can be derived from a living or dead organism, explanted tissues or organs, prokaryotic or eukaryotic cells, and/or cultured cells. In some aspects, the extracellular vesicles are produced by cells that express one or more transgene products.
- exosome refers to an extracellular vesicle with a diameter between 20-300 nm (e.g., between 40-200 nm). Exosomes comprise a membrane that encloses an internal space (i.e., lumen), and, in some aspects, can be generated from a cell (e.g., producer cell) by direct plasma membrane budding or by fusion of the late endosome or multi -vesicular body with the plasma membrane. In certain aspects, an exosome comprises a scaffold moiety. As described infra, exosome can be derived from a producer cell, and isolated from the producer cell based on its size, density, biochemical parameters, or a combination thereof. In some aspects, the EVs, e.g., exosomes, of the present disclosure are produced by cells that express one or more transgene products.
- the term "nanovesicle” refers to an extracellular vesicle with a diameter between 20-250 nm (e.g., between 30-150 nm) and is generated from a cell (e.g., producer cell) by direct or indirect manipulation such that the nanovesicle would not be produced by the cell without the manipulation.
- Appropriate manipulations of the cell to produce the nanovesicles include but are not limited to serial extrusion, treatment with alkaline solutions, sonication, or combinations thereof. In some aspects, production of nanovesicles can result in the destruction of the producer cell.
- population of nanovesicles described herein are substantially free of vesicles that are derived from cells by way of direct budding from the plasma membrane or fusion of the late endosome with the plasma membrane.
- a nanovesicle comprises a scaffold moiety. Nanovesicles, once derived from a producer cell, can be isolated from the producer cell based on its size, density, biochemical parameters, or a combination thereof.
- surface-engineered EVs e.g., exosomes
- EVs e.g., Scaffold X- engineered EVs, e.g., exosomes
- the term "surface-engineered EVs, e.g., exosomes” refers to an EV, e.g., exosome, with the membrane or the surface of the EV, e.g., exosome, modified in its composition so that the surface of the engineered EV, e.g., exosome, is different from that of the EV, e.g., exosome, prior to the modification or of the naturally occurring EV, e.g., exosome.
- the engineering can be on the surface of the EV, e.g., exosome, or in the membrane of the EV, e.g., exosome, so that the surface of the EV, e.g., exosome, is changed.
- the membrane is modified in its composition of a protein, a lipid, a small molecule, a carbohydrate, etc.
- the composition can be changed by a chemical, a physical, or a biological method or by being produced from a cell previously or concurrently modified by a chemical, a physical, or a biological method.
- the composition can be changed by a genetic engineering or by being produced from a cell previously modified by genetic engineering.
- a surface -engineered EV e.g., exosome
- comprises an exogenous protein i.e., a protein that the EV, e.g. , exosome, does not naturally express
- a fragment or variant thereof that can be exposed to the surface of the EV, e.g., exosome, or can be an anchoring point (attachment) for a moiety exposed on the surface of the EV, e.g., exosome.
- a surface-engineered EV e.g., exosome
- lumen-engineered exosome refers to an EV, e.g., exosome, with the membrane or the lumen of the EV, e.g., exosome, modified in its composition so that the lumen of the engineered EV, e.g., exosome, is different from that of the EV, e.g., exosome, prior to the modification or of the naturally occurring EV, e.g., exosome.
- the engineering can be directly in the lumen or in the membrane of the EV, e.g., exosome so that the lumen of the EV, e.g., exosome is changed.
- the membrane is modified in its composition of a protein, a lipid, a small molecule, a carbohydrate, etc. so that the lumen of the EV, e.g., exosome is modified.
- the composition can be changed by a chemical, a physical, or a biological method or by being produced from a cell previously modified by a chemical, a physical, or a biological method.
- the composition can be changed by a genetic engineering or by being produced from a cell previously modified by genetic engineering.
- a lumen-engineered exosome comprises an exogenous protein (i.e., a protein that the EV, e.g., exosome does not naturally express) or a fragment or variant thereof that can be exposed in the lumen of the EV, e.g., exosome or can be an anchoring point (attachment) for a moiety exposed on the inner layer ofthe EV, e.g., exosome.
- exogenous protein i.e., a protein that the EV, e.g., exosome does not naturally express
- a fragment or variant thereof that can be exposed in the lumen of the EV, e.g., exosome or can be an anchoring point (attachment) for a moiety exposed on the inner layer ofthe EV, e.g., exosome.
- a lumen-engineered EV e.g., exosome
- a lumen-engineered EV comprises a higher expression of a natural exosome protein (e.g., Scaffold X or Scaffold Y) or a fragment or variant thereof that can be exposed to the lumen of the exosome or can be an anchoring point (attachment) for a moiety exposed in the lumen of the exosome.
- modified when used in the context of EVs, e.g., exosomes described herein, refers to an alteration or engineering of an EV, e.g. , exosome and/or its producer cell, such that the modified EV, e.g., exosome is different from a naturally-occurring EV, e.g., exosome.
- a modified EV, e.g., exosome described herein comprises a membrane that differs in composition of a protein, a lipid, a small molecular, a carbohydrate, etc.
- exosome e.g., membrane comprises higher density or number of natural exosome proteins and/or membrane comprises proteins that are not naturally found in exosomes (e.g., antigen, adjuvant, and/or immune modulator).
- modifications to the membrane changes the exterior surface of the EV, e.g., exosome (e.g., surface-engineered EVs, e.g., exosomes described herein).
- such modifications to the membrane changes the lumen of the EV, e.g., exosome (e.g., lumen-engineered EVs, e.g., exosomes described herein).
- a scaffold moiety refers to a molecule that can be used to anchor a payload or any other compound of interest (e.g., antigen, adjuvant, and/or immune modulator) to the EV, e.g., exosome either on the luminal surface or on the exterior surface of the EV, e.g., exosome.
- a scaffold moiety comprises a synthetic molecule.
- a scaffold moiety comprises a non-polypeptide moiety.
- a scaffold moiety comprises a lipid, carbohydrate, or protein that naturally exists in the EV, e.g. , exosome.
- a scaffold moiety comprises a lipid, carbohydrate, or protein that does not naturally exist in the EV, e.g., exosome.
- a scaffold moiety is Scaffold X.
- a scaffold moiety is Scaffold Y.
- a scaffold moiety comprises both Scaffold X and Scaffold Y.
- Non-limiting examples of other scaffold moieties that can be used with the present disclosure include: aminopeptidase N (CD13); Neprilysin, AKA membrane metalloendopeptidase (MME); ectonucleotide pyrophosphatase/phosphodiesterase family member 1 (ENPP1); Neuropilin-1 (NRP1); CD9, CD63, CD81, PDGFR, GPI anchor proteins, lactadherin, LAMP2, and LAMP2B.
- Scaffold X refers to exosome proteins that have recently been identified on the surface of exosomes. See, e.g., U.S. Pat. No. 10,195,290, which is incorporated herein by reference in its entirety.
- Non-limiting examples of Scaffold X proteins include: prostaglandin F2 receptor negative regulator (“the PTGFRN protein”); basigin (“the BSG protein”); immunoglobulin superfamily member 2 (“the IGSF2 protein”); immunoglobulin superfamily member 3 (“the IGSF3 protein”); immunoglobulin superfamily member 8 (“the IGSF8 protein”); integrin beta-1 ("the ITGB1 protein); integrin alpha-4 (“the ITGA4 protein”); 4F2 cell-surface antigen heavy chain (“the SLC3A2 protein”); and a class of ATP transporter proteins ("the ATP1A1 protein,” “the ATP1A2 protein,” “the ATP1A3 protein,” “the ATP1A4 protein,” “the ATP1B3 protein,” “the ATP2B1 protein,” “the ATP2B2 protein,” “the ATP2B3 protein,” “the ATP2B protein”).
- a Scaffold X protein can be a whole protein or a fragment thereof (e.g., functional fragment, e.g., the smallest fragment that is capable of anchoring another moiety on the exterior surface or on the luminal surface of the EV, e.g., exosome).
- a Scaffold X can anchor a moiety (e.g., antigen, adjuvant, and/or immune modulator) to the external surface or the luminal surface of the exosome.
- Scaffold Y refers to exosome proteins that were newly identified within the lumen of exosomes. See, e.g., International Publ. No. WO/2019/099942 (or the U.S. equivalent US 2020/0347112); and WO 2020/101740, each of which is incorporated herein by reference in its entirety.
- Non-limiting examples of Scaffold Y proteins include: myristoylated alanine rich Protein Kinase C substrate ("the MARCKS protein”); myristoylated alanine rich Protein Kinase C substrate like 1 (“the MARCKSE1 protein”); and brain acid soluble protein 1 (“the BASP1 protein”).
- a Scaffold Y protein can be a whole protein or a fragment thereof (e.g. , functional fragment, e.g. , the smallest fragment that is capable of anchoring a moiety to the luminal surface of the exosome).
- a Scaffold Y can anchor a moiety (e.g., antigen, adjuvant, and/or immune modulator) to the luminal surface of the EV, e.g., exosome.
- fragment of a protein refers to an amino acid sequence of a protein that is shorter than the naturally-occurring sequence, N- and/or C-terminally deleted or any part of the protein deleted in comparison to the naturally occurring protein.
- functional fragment refers to a protein fragment that retains protein function. Accordingly, in some aspects, a functional fragment of a Scaffold X protein retains the ability to anchor a moiety on the luminal surface or on the exterior surface of the EV, e.g., exosome.
- a functional fragment of a Scaffold Y protein retains the ability to anchor a moiety on the luminal surface of the EV, e.g., exosome. Whether a fragment is a functional fragment can be assessed by any art known methods to determine the protein content of EVs, e.g., exosomes including Western Blots, FACS analysis and fusions of the fragments with autofluorescent proteins like, e.g., GFP.
- a functional fragment of a Scaffold X protein retains at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90% or at least about 100% of the ability, e.g., an ability to anchor a moiety, of the naturally occurring Scaffold X protein.
- a functional fragment of a Scaffold Y protein retains at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90% or at least about 100% of the ability, e.g., an ability to anchor another molecule, of the naturally occurring Scaffold Y protein.
- variant of a molecule refers to a molecule that shares certain structural and functional identities with another molecule upon comparison by a method known in the art.
- a variant of a protein can include a substitution, insertion, deletion, frameshift or rearrangement in another protein.
- a "conservative amino acid substitution” is one in which the amino acid residue is replaced with an amino acid residue having a similar side chain.
- Families of amino acid residues having similar side chains have been defined in the art, including basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine).
- basic side chains e
- a string of amino acids can be conservatively replaced with a structurally similar string that differs in order and/or composition of side chain family members.
- percent sequence identity or “percent identity” between two polynucleotide or polypeptide sequences refers to the number of identical matched positions shared by the sequences over a comparison window, taking into account additions or deletions (i.e., gaps) that must be introduced for optimal alignment of the two sequences.
- a matched position is any position where an identical nucleotide or amino acid is presented in both the target and reference sequence. Gaps presented in the target sequence are not counted since gaps are not nucleotides or amino acids. Likewise, gaps presented in the reference sequence are not counted since target sequence nucleotides or amino acids are counted, not nucleotides or amino acids from the reference sequence.
- the percentage of sequence identity is calculated by determining the number of positions at which the identical amino-acid residue or nucleic acid base occurs in both sequences to yield the number of matched positions, dividing the number of matched positions by the total number of positions in the window of comparison and multiplying the result by 100 to yield the percentage of sequence identity.
- the comparison of sequences and determination of percent sequence identity between two sequences can be accomplished using readily available software both for online use and for download. Suitable software programs are available from various sources, and for alignment of both protein and nucleotide sequences. One suitable program to determine percent sequence identity is bl2seq, part of the BLAST suite of programs available from the U.S.
- B12seq performs a comparison between two sequences using either the BLASTN or BLASTP algorithm.
- BLASTN is used to compare nucleic acid sequences
- BLASTP is used to compare amino acid sequences.
- Other suitable programs are, e.g., Needle, Stretcher, Water, or Matcher, part of the EMBOSS suite of bioinformatics programs and also available from the European Bioinformatics Institute (EBI) at www.ebi.ac.uk/Tools/psa.
- Different regions within a single polynucleotide or polypeptide target sequence that aligns with a polynucleotide or polypeptide reference sequence can each have their own percent sequence identity. It is noted that the percent sequence identity value is rounded to the nearest tenth. For example, 80.11, 80.12, 80.13, and 80.14 are rounded down to 80.1, while 80.15, 80.16, 80.17, 80.18, and 80.19 are rounded up to 80.2. It also is noted that the length value will always be an integer. [0054] One skilled in the art will appreciate that the generation of a sequence alignment for the calculation of a percent sequence identity is not limited to binary sequence-sequence comparisons exclusively driven by primary sequence data.
- Sequence alignments can be derived from multiple sequence alignments.
- One suitable program to generate multiple sequence alignments is ClustalW2, available from www.clustal.org.
- Another suitable program is MUSCLE, available from www.drive5.com/muscle/.
- ClustalW2 and MUSCLE are alternatively available, e.g., from the EBE
- sequence alignments can be generated by integrating sequence data with data from heterogeneous sources such as structural data (e.g., crystallographic protein structures), functional data (e.g., location of mutations), or phylogenetic data.
- a suitable program that integrates heterogeneous data to generate a multiple sequence alignment is T-Coffee, available at worldwideweb.tcoffee.org, and alternatively available, e.g., from the EBE It will also be appreciated that the final alignment used to calculate percent sequence identity can be curated either automatically or manually.
- variants can be generated to improve or alter the characteristics of the polypeptides. For instance, one or more amino acids can be deleted from the N-terminus or C-terminus of the secreted protein without substantial loss of biological function.
- interferon gamma exhibited up to ten times higher activity after deleting 8-10 amino acid residues from the carboxy terminus of this protein. (Dobeli et al., J. Biotechnology 7: 199-216 (1988), incorporated herein by reference in its entirety.)
- polypeptide variants include, e.g., modified polypeptides.
- Modifications include, e.g., conservative amino acid substitution, acetylation, acylation, ADP-ribosylation, amidation, covalent attachment of flavin, covalent attachment of a heme moiety, covalent attachment of a nucleotide or nucleotide derivative, covalent attachment of a lipid or lipid derivative, covalent attachment of phosphotidylinositol, covalent attachment of bioorthoganal functionalities (e.g., azide, alkyne, trans- cycloalkyne, tetrazine), cross-linking, cyclization, disulfide bond formation, demethylation, formation of covalent cross-links, formation of cysteine, formation of pyroglutamate, formylation, gammacarboxylation, glycosylation, GPI anchor formation, hydroxylation, iodination, methylation
- Scaffold X and/or Scaffold Y is modified at any convenient location.
- a molecule described herein e.g., antigen, adjuvant, immune modulator, targeting moiety, affinity ligand, and/or scaffold moiety
- an EV e.g., exosome
- the molecule is present in (e.g., in the lumen) or on (e.g., on the exterior surface and/or luminal surface) the EV.
- a molecule can be exogenously introduced into a producer cell or directly into an EV, such that the EV expresses the molecule of interest.
- a molecule of interest can be produced separately from an EV and then conjugated or linked to a moiety present in the EV, such that the EV expresses the molecule.
- an antigen e.g., KRAS antigen
- an affinity ligand disclosed herein can be fused to an affinity ligand disclosed herein.
- the antigen-affinity ligand fusion can be linked or conjugated to a scaffold moiety expressed on the surface of an EV via the affinity ligand. Additional disclosure relating to different methods of expressing a molecule of interest in or on an EV (e.g., exosome) is described elsewhere in the present disclosure.
- the term "linked to,” “fused,” or “conjugated to” are used interchangeably and refer to a covalent or non-covalent bond formed between a first moiety and a second moiety, e.g., Scaffold X and an antigen (or adjuvant or immune modulator), respectively, e.g., a scaffold moiety expressed in or on the extracellular vesicle and an antigen, e.g., Scaffold X (e.g., a PTGFRN protein), respectively, in the luminal surface of or on the external surface of the extracellular vesicle.
- a payload disclosed herein e.g.
- antigen, adjuvant, and/or immune modulator and/or a targeting moiety can be directly linked to the exterior surface and/or the luminal surface of an EV (e.g. , exosome).
- an EV e.g. , exosome
- directly linked refers to the process of linking (fusing or conjugating) a moiety (e.g., a payload and/or targeting moiety) to the surface of an EV (e.g., exosome) without the use of a scaffold moiety disclosed herein.
- fusion protein or “fusion molecule” (or derivatives thereof) refers to two or more proteins (or molecules) that are linked or conjugated to each other.
- a fusion protein that can be expressed in an EV e.g., exosome
- a payload e.g., antigen, adjuvant, and/or immune modulator
- a scaffold moiety e.g., Scaffold X and/or Scaffold Y.
- the payload (e.g., antigen, adjuvant, and/or immune modulator) is linked or conjugated to the scaffold moiety via an affinity ligand (e.g., those described herein).
- a fusion protein that can be expressed in an EV (e.g., exosome) useful for the present disclosure comprises (i) a targeting moiety and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y).
- the targeting moiety is linked or conjugated to the scaffold moiety via an affinity ligand (e.g., those described herein).
- EVs e.g., exosomes
- a first fusion protein comprises (i) a payload (e.g., antigen, adjuvant, and/or immune modulator) and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y), and wherein a second fusion protein comprises (i) a targeting moiety and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y)
- a payload e.g., antigen, adjuvant, and/or immune modulator
- a scaffold moiety e.g., Scaffold X and/or Scaffold Y
- a second fusion protein comprises (i) a targeting moiety and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y)
- encapsulated refers to a status or process of having a first moiety (e.g., antigen, adjuvant, or immune modulator) inside a second moiety (e.g., an EV, e.g., exosome) without chemically or physically linking the two moieties.
- a first moiety e.g., antigen, adjuvant, or immune modulator
- a second moiety e.g., an EV, e.g., exosome
- the term “encapsulated” can be used interchangeably with "in the lumen of”.
- Non-limiting examples of encapsulating a first moiety (e.g., antigen, adjuvant, or immune modulator) into a second moiety (e.g., EVs, e.g., exosomes) are disclosed elsewhere herein.
- the term "producer cell” refers to a cell used for generating an EV, e.g., exosome.
- a producer cell can be a cell cultured in vitro, or a cell in vivo.
- a producer cell includes, but are not limited to, a cell known to be effective in generating EVs, e.g, exosomes, e.g., HEK293 cells, Chinese hamster ovary (CHO) cells, mesenchymal stem cells (MSCs), BJ human foreskin fibroblast cells, fHDF fibroblast cells, AGE.HN® neuronal precursor cells, CAP® amniocyte cells, adipose mesenchymal stem cells, RPTEC/TERT1 cells.
- a producer cell is not a naturally-existing antigen-presenting cell (i.e., has been modified).
- a producer cell is not a naturally -existing dendritic cell, a naturally-existing B cell, a naturally-existing mast cell, a naturally-existing macrophage, a naturally-existing neutrophil, naturally-existing Kupffer-Browicz cell, cell derived from any of these cells, or any combination thereof. Additional disclosures relating to such producer cells are provided elsewhere in the present disclosure.
- the EVs, e.g., exosomes useful in the present disclosure do not carry an antigen on MHC class I or class II molecule (i.e., antigen is not presented on MHC class I or class II molecule) exposed on the surface of the EV, e.g., exosome, but instead can carry an antigen in the lumen of the EV, e.g., exosome, or on the surface of the EV, e.g., exosome, by attachment to Scaffold X and/or Scaffold Y.
- isolating or purifying as used herein is the process of removing, partially removing (e.g., a fraction) of the EVs from a sample containing producer cells.
- an isolated EV composition has no detectable undesired activity or, alternatively, the level or amount of the undesired activity is at or below an acceptable level or amount. In some aspects, an isolated EV composition has an amount and/or concentration of desired EVs at or above an acceptable amount and/or concentration. In some aspects, the isolated EV composition is enriched as compared to the starting material (e.g., producer cell preparations) from which the composition is obtained. This enrichment can be by 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, 99.99%, 99.999%, 99.9999%, or greater than 99.9999% as compared to the starting material.
- the starting material e.g., producer cell preparations
- isolated EV preparations are substantially free of residual biological products.
- the isolated EV preparations are 100% free, 99% free, 98% free, 97% free, 96% free, 95% free, 94% free, 93% free, 92% free, 91% free, or 90% free of any contaminating biological matter.
- Residual biological products can include abiotic materials (including chemicals) or unwanted nucleic acids, proteins, lipids, or metabolites.
- Substantially free of residual biological products can also mean that the EV composition contains no detectable producer cells and that only EVs are detectable.
- immune modulator refers to an agent (i.e., payload) that acts on a target (e.g. , a target cell) that is contacted with the extracellular vesicle, and regulates the immune system.
- a target e.g. , a target cell
- immune modulator that can be introduced into an EV (e.g., exosome) and/or a producer cell include agents such as, modulators of checkpoint inhibitors, ligands of checkpoint inhibitors, cytokines, derivatives thereof, or any combination thereof.
- the immune modulator can also include an agonist, an antagonist, an antibody, an antigen-binding fragment, a polynucleotide, such as siRNA, antisense oligonucleotide, a phosphorodiamidate morpholino oligomer (PMO), a peptide -conjugated phosphorodiamidate morpholino oligomer (PPMO), miRNA, IncRNA, mRNA DNA, or a small molecule.
- a polynucleotide such as siRNA, antisense oligonucleotide, a phosphorodiamidate morpholino oligomer (PMO), a peptide -conjugated phosphorodiamidate morpholino oligomer (PPMO), miRNA, IncRNA, mRNA DNA, or a small molecule.
- PMO phosphorodiamidate morpholino oligomer
- PPMO peptide -conjugated phosphorodiamidate morph
- the term “targeting moiety” can be used interchangeably with the term bio-distribution modifying agent.
- the targeting moiety alters the tropism of the EV (e.g., exosome) (“tropism moiety”).
- tropism moiety refers to a targeting moiety that when expressed on an EV (e.g., exosome) alters and/or enhances the natural movement of the EV.
- a tropism moiety can promote the EV to move towards a particular cell, tissues, or a stimuli.
- a tropism moiety can promote the EV to be taken up by a particular cell, tissue, or organ.
- targeting moiety encompasses tropism moieties and can be used interchangeably.
- the bio-distribution agent can be a biological molecule, such as a protein, a peptide, a lipid, or a carbohydrate, or a synthetic molecule.
- the bio-distribution modifying agent can be an antibody, a synthetic polymer (e.g. , PEG), a natural ligand (e.g., CD40L, albumin), a recombinant protein (e.g., XTEN), but not limited thereto.
- a synthetic polymer e.g. , PEG
- a natural ligand e.g., CD40L, albumin
- a recombinant protein e.g., XTEN
- the bio-distribution modifying agent is displayed on the surface of EVs (e.g., exosomes).
- the bio-distribution modifying agent can be displayed on the EV surface by being fused to a scaffold protein (e.g. , Scaffold X) (e.g. , as a genetically encoded fusion molecule).
- the bio-distribution modifying agent can be displayed on the EV surface by chemical reaction attaching the biodistribution modifying agent to an EV surface molecule.
- a non-limiting example is PEGylation.
- EVs disclosed herein e.g., exosomes
- Non-limiting examples of tropsim or targeting moiety that can be used with the present disclosure include a C-type lectin domain family 9 member A (Clec9a) protein, a dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN), CD207, CD40, Clec6, dendritic cell immunoreceptor (DCIR), DEC-205, lectin-like oxidized low-density lipoprotein receptor- 1 (LOX-1), MARCO, Clecl2a, DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin-1, macrophage mannose receptor (MMR), BDCA-1 (CD303, Clec4c), Dectin-2, Bst-2 (CD317), CD3, CD14, CD16, CD64, CD68, CD71, CCR5, or any combination thereof.
- the targeting moiety is Clec9a protein.
- the targeting moiety is Clec
- the term "payload” refers to an agent that acts on a target (e.g. , a target cell) that is contacted with the EV (e.g., exosome). Contacting can occur in vitro or in a subject.
- a target e.g. , a target cell
- the term payload can be used interchangeably with the terms "moiety,” “agents,” and “biologically active molecules.”
- payload Non-limiting examples of payload that can be included on the EV, e.g., exosome, are an antigen, an adjuvant, and/or an immune modulator.
- Payloads that can be introduced into an EV, e.g., exosome, and/or a producer cell include agents such as, nucleotides (e.g., nucleotides comprising a detectable moiety or a toxin or that disrupt transcription), nucleic acids (e.g.
- DNA or mRNA molecules that encode a polypeptide such as an enzyme, or RNA molecules that have regulatory function such as miRNA, dsDNA, IncRNA, siRNA, antisense oligonucleotide, a phosphorodiamidate morpholino oligomer (PMO), a peptide-conjugated phosphorodiamidate morpholino oligomer (PPMO), or combinations thereof), amino acids (e.g., amino acids comprising a detectable moiety or a toxin or that disrupt translation), polypeptides (e.g., enzymes), lipids, carbohydrates, peptides (e.g., cell penetrating peptides), and small molecules (e.g., small molecule drugs and toxins).
- PMO phosphorodiamidate morpholino oligomer
- PPMO peptide-conjugated phosphorodiamidate morpholino oligomer
- amino acids e.g., amino acids comprising a detectable
- a payload comprises an antigen.
- antigen refers to any agent that when introduced into a subject elicits an immune response (cellular or humoral) to itself.
- an antigen is associated with a neurological disorder. Additional disclosure relating to such antigens are provided elsewhere in the present disclosure.
- affinity ligand refers to a molecule that can selectively and preferentially bind to a specific marker, e.g., expressed on a target cell or on EVs, e.g, a scaffold moiety, e.g., PTGFRN on EVs.
- an affinity ligand comprises a peptide (e.g., linear peptide) or protein that can increase the binding of a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to a moiety on the surface of EVs, e.g., a scaffold moiety disclosed herein.
- a molecule of interest e.g., antigen, adjuvant, immune modulator, and/or targeting moiety
- affinity ligands that can be used with the present disclosure include an antibody, phage display peptide, fibronectin domain, camelid, VNAR, VHH domain, and combinations thereof.
- antibody encompasses an immunoglobulin whether natural or partly or wholly synthetically produced, and fragments thereof. The term also covers any protein having a binding domain that is homologous to an immunoglobulin binding domain. "Antibody” further includes a polypeptide comprising a framework region from an immunoglobulin gene or fragments thereof that specifically binds and recognizes an antigen.
- antibody is meant to include whole antibodies, polyclonal, monoclonal and recombinant antibodies, fragments thereof, and further includes single-chain antibodies, humanized antibodies, murine antibodies, chimeric, mouse-human, mouse-primate, primatehuman monoclonal antibodies, anti-idiotype antibodies, antibody fragments, such as, e.g., scFv, (scFv)2, Fab, Fab', and F(ab')2, F(abl)2, Fv, dAb, and Fd fragments, diabodies, and antibody-related polypeptides.
- Antibody includes bispecific antibodies and multispecific antibodies so long as they exhibit the desired biological activity or function.
- the terms "individual,” “subject,” “host,” and “patient,” are used interchangeably herein and refer to any mammalian subject for whom diagnosis, treatment, or therapy is desired, particularly humans.
- the compositions and methods described herein are applicable to both human therapy and veterinary applications.
- the subject is a mammal, and in some aspects, the subject is a human.
- a “mammalian subject” includes all mammals, including without limitation, humans, domestic animals (e.g., dogs, cats and the like), farm animals (e.g., cows, sheep, pigs, horses and the like) and laboratory animals (e.g., monkey, rats, mice, rabbits, guinea pigs and the like).
- the term "substantially free” means that the sample comprising EVs, e.g., exosomes, comprise less than about 10% of macromolecules by mass/volume (m/v) percentage concentration. Some fractions can contain less than about 0.001%, less than about 0.01%, less than about 0.05%, less than about 0.1%, less than about 0.2%, less than about 0.3%, less than about 0.4%, less than about 0.5%, less than about 0.6%, less than about 0.7%, less than about 0.8%, less than about 0.9%, less than about 1%, less than about 2%, less than about 3%, less than about 4%, less than about 5%, less than about 6%, less than about 7%, less than about 8%, less than about 9%, or less than about 10% (m/v) of macromolecules.
- macromolecule means nucleic acids, contaminant proteins, lipids, carbohydrates, metabolites, or a combination thereof.
- the term "conventional exosome protein” means a protein previously known to be enriched in exosomes, including but is not limited to CD9, CD63, CD81, PDGFR, GPI anchor proteins, lactadherin LAMP2, and LAMP2B, a fragment thereof, or a peptide that binds thereto.
- administering means to give a composition comprising an EV, e.g., exosome, disclosed herein to a subject via a pharmaceutically acceptable route.
- Routes of administration can be intravenous, e.g., intravenous injection and intravenous infusion. Additional routes of administration include, e.g., subcutaneous, intramuscular, intrathecal, intravitreal, intracranial, oral, nasal, and pulmonary administration.
- Exosomes can also be directly administered to the target tissue, EVs, e.g., exosomes can be administered as part of a pharmaceutical composition comprising at least one excipient.
- an "immune response,” as used herein, refers to a biological response within a vertebrate against foreign agents or abnormal, e.g. , cancerous cells, which response protects the organism against these agents and diseases caused by them.
- An immune response is mediated by the action of one or more cells of the immune system (for example, a T lymphocyte, B lymphocyte, natural killer (NK) cell, macrophage, eosinophil, mast cell, dendritic cell or neutrophil) and soluble macromolecules produced by any of these cells or the liver (including antibodies, cytokines, and complement) that results in selective targeting, binding to, damage to, destruction of, and/or elimination from the vertebrate's body of invading pathogens, cells or tissues infected with pathogens, cancerous or other abnormal cells, or, in cases of autoimmunity or pathological inflammation, normal human cells or tissues.
- immune response comprises a cellular immune response, a humoral immune response, an innate cell immune response, or a combination thereof.
- An immune reaction includes, e.g., activation or inhibition of a T cell, e.g., an effector T cell, a Th cell, a CD4+ cell, a CD8+ T cell, or a Treg cell, or activation or inhibition of any other cell of the immune system, e.g., NK cell.
- an immune response can comprise a humoral immune response (e.g., mediated by B-cells), cellular immune response (e.g., mediated by T cells), or both humoral and cellular immune responses.
- an immune response is an "inhibitory" immune response.
- an “inhibitory” or “tolerogenic” immune response is an immune response that blocks or diminishes the effects of a stimulus (e.g., antigen).
- the inhibitory immune response comprises the production of inhibitory antibodies against the stimulus.
- the inhibitory immune response comprises the induction of tolerogenic cells, such as regulatory T cells (e.g., FoxP3+ regulatory CD4+ T cells).
- the inhibitory immune response comprises the production of tolerogenic cytokines/chemokines (e.g., IL-10 or TGF-J3).
- an immune response is a "stimulatory" immune response.
- a “stimulatory” immune response comprises an immune response that results in the generation of effectors cells (e.g., cytotoxic T lymphocytes) that can destroy and clear a target antigen (e.g., tumor antigen or viruses).
- a stimulatory immune response comprises the production of antibodies that can specifically bind and neutralize an antigen.
- cellular immune response can be used interchangeably with the term “cell-mediated immune response” and refers to an immune response that does not predominantly involve antibodies. Instead, a cellular immune response involves the activation of different immune cells (e.g., phagocytes and antigen-specific cytotoxic T-lymphocytes) that produce various effector molecules (e.g., cytokines, perforin, granzymes) upon activation (e.g., via antigen stimulation).
- effector molecules e.g., cytokines, perforin, granzymes
- the term “humoral immune response” refers to an immune response predominantly mediated by macromolecules found in extracellular fluids, such as secreted antibodies, complement proteins, and certain antimicrobial peptides.
- antibody-mediated immune response refers to an aspect of a humoral immune response that is mediated by antibodies.
- immune cells refers to any cells of the immune system that are involved in mediating an immune response.
- Non-limiting examples of immune cells include a T lymphocyte, B lymphocyte, natural killer (NK) cell, macrophage, eosinophil, mast cell, dendritic cell, neutrophil, or combination thereof.
- an immune cell expresses CD3.
- the CD3-expressing immune cells are T cells (e.g., CD4+ T cells or CD8+ T cells).
- an immune cell that can be targeted with a targeting moiety disclosed herein e.g. , anti-CD3 comprises a naive CD4+ T cell.
- an immune cell comprises a memory CD4+ T cell. In some aspects, an immune cell comprises an effector CD4+ T cell. In some aspects, an immune cell comprises a naive CD8+ T cell. In some aspects, an immune cell comprises a memory CD8+ T cell. In some aspects, an immune cell comprises an effector CD8+ T cell. In some aspects, an immune cell is a dendritic cell.
- a dendritic cell comprises a plasmacytoid dendritic cell (pDC), a conventional dendritic cell 1 (cDCl), a conventional dendritic cell 2 (cDC2), inflammatory monocyte derived dendritic cells, Langerhans cells, dermal dendritic cells, lysozyme-expressing dendritic cells (LysoDCs), Kupffer cells, or any combination thereof.
- an immune cell that an EV disclosed herein e.g. , exosomes
- an immune cell is a macrophage.
- the macrophage comprises Ml macrophages, M2 macrophages, or both.
- the macrophage is a microglia, meningeal macrophage, perivascular macrophage, choroid plexus macrophage, or combinations thereof.
- T cell refers to a type of lymphocyte that matures in the thymus. T cells play an important role in cell-mediated immunity and are distinguished from other lymphocytes, such as B cells, by the presence of a T-cell receptor on the cell surface. T-cells include all types of immune cells expressing CD3, including, but not limited to, T-helper cells (CD4+ cells), cytotoxic T-cells (CD8+ cells), natural killer T-cells, T-regulatory cells (Treg), T follicular helper (Tfh) cells, peripheral Tfh cells, mucosal-associated invariant T (MAIT) cells, and gamma-delta T cells.
- T-helper cells CD4+ cells
- CD8+ cells cytotoxic T-cells
- Treg T-regulatory cells
- Tfh T follicular helper
- MAIT mucosal-associated invariant T
- MAIT mucosal-associated invariant T
- a "naive" T cell refers to a mature T cell that remains immunologically undifferentiated (i. e. , not activated). Following positive and negative selection in the thymus, T cells emerge as either CD4+ or CD8+ naive T cells. In their naive state, T cells express L-selectin (CD62L+), IL-7 receptor-a (IL-7R-a), and CD132, but they do not express CD25, CD44, CD69, or CD45RO.
- immature can also refers to a T cell which exhibits a phenotype characteristic of either a naive T cell or an immature T cell, such as a TSCM cell or a TCM cell.
- an immature T cell can express one or more of L- selectin (CD62L+), IL-7Ra, CD132, CCR7, CD45RA, CD45RO, CD27, CD28, CD95, CXCR3, and LFA- 1.
- L- selectin CD62L+
- IL-7Ra IL-7Ra
- CD132 CCR7
- CD45RA CD45RO
- Naive or immature T cells can be contrasted with terminal differentiated effector T cells, such as TEM cells and TEFF cells.
- effector T cells refers to a T cell that can mediate the removal of a pathogen or cell without requiring further differentiation.
- effector T cells are distinguished from naive T cells and memory T cells, and these cells often have to differentiate and proliferate before becoming effector cells.
- memory T cells refer to a subset of T cells that have previously encountered and responded to their cognate antigen. In some aspects, the term is synonymous with "antigen-experienced" T cells. In some aspects, memory T cells can be effector memory T cells or central memory T cells. In some aspects, the memory T cells are tissue-resident memory T cells. As used herein, the term “tissue-resident memory T cells” or “TRM cells” refers to a lineage of T cells that occupies tissues (e.g., skin, lung, gastrointestinal tract) without recirculating.
- TRM cells are transcriptionally, phenotypically and functionally distinct from central memory and effector memory T cells which recirculate between blood, the T cell zones of secondary lymphoid organs, lymph and nonlymphoid tissues.
- One of the roles of TRM cells is to provide immune protection against infection in extralymphoid tissues.
- dendritic cells refers to a class of bone-marrow-derived immune cells that are capable of processing extracellular and intracellular proteins and to present antigens in the context of MHC molecules to prime naive T cells.
- dendritic cells can be divided into further subtypes, such as conventional dendritic cell 1 (cDCl), conventional dendritic cell 2 (cDC2), plasmacytoid dendritic cell (pDC), inflammatory monocyte derived dendritic cells, Langerhans cells, dermal dendritic cells, lysozyme-expressing dendritic cells (LysoDCs), Kupffer cells, and combinations thereof.
- human cDCl cells are CDlc" and CD141 + .
- human cDC2 cells are CDlc + and CD 141’.
- human pDC cells are CD123 + .
- mouse cDCl cells are XCR1 + , Clec9a + , and Sirpa".
- mouse cDC2 cells are CD8 + , CD1 lb + , Sirpa + , XCR1", and CDlc,b + .
- mouse pDC cells are CD137 + , XCR1", and Sirpa”.
- phenotypic markers for distinguishing the different DC subsets are known in the art. See, e.g. , Collin et al. , Immunology 154(1): 3-20 (2016).
- the different DC subsets can be distinguished based on their functional properties. For example, in certain aspects, pDCs produce large amounts of IFN-a, while cDCls and cDC2s produce inflammatory cytokines, such as IL-12, IL-6, and TNF-a.
- Other methods of distinguishing the different DC subsets are known in the art. See, e.g., U.S. Patent Nos. 8,426,565 B2 and 9,988,431, each of which is herein incorporated by reference in its entirety.
- macrophage refers to a mononuclear phagocyte characterized by the expression of at least CD 14 and lack of expression of dendritic cell markers. Macrophages can be typically divided into (i) classically-activated macrophages (“Ml macrophages”) and (ii) alternatively- activated macrophages (“M2 macrophages”). Martinez et al., Annu. Rev. Immunol. 27:451-483 (2009). Generally, Ml macrophages exhibit potent anti -microbial properties, reminiscent of type 1 T-helper lymphocyte (Thl) responses.
- Thl type 1 T-helper lymphocyte
- M2 macrophages promote type 2 T-helper lymphocyte (Th2)-like responses, secrete less pro-inflammatory cytokines, and assist resolution of inflammation by trophic factor synthesis and phagocytosis.
- Th2 macrophages can be further divided into three distinct subclasses, i.e., M2a, M2b, and M2c, defined by specific cytokine profiles. Mantovani et al., Trends Immunol. 25:677-686 (2004).
- M2 macrophages are generally characterized by low production of pro-inflammatory cytokines, such as IL- 12, and high production of anti-inflammatory cytokines such as IL- 10, M2b macrophages retain high levels of inflammatory cytokine production, such as TNF-a and IL-6. Mosser, J. Leukocyte Biol. 73:209-212 (2003).
- Macrophages can be polarized by their microenvironment to assume different phenotypes associated with different stages of inflammation and healing. Stout et al., J. Immunol. 175:342-349 (2005). Certain macrophages are indispensible for wound healing. They participate in the early stages of cell recruitment and of tissue defense, as well as the later stages of tissue homeostasis and repair. Pollard, Nature Rev. 9:259-270 (2009). Macrophages derived from peripheral blood monocytes have been used to treat refractory ulcers. Danon et al., Exp. Gerontol. 32:633-641 (1997); Zuloff-Shani et al., Transfus. Apher. Sci. 30: 163-167 (2004), each of which is incorporated herein by reference as if set forth in its entirety.
- immunoconjugate refers to a compound comprising a binding molecule (e.g., an antibody) and one or more moieties, e.g., therapeutic or diagnostic moieties, chemically conjugated to the binding molecule.
- a binding molecule e.g., an antibody
- moieties e.g., therapeutic or diagnostic moieties
- an immunoconjugate is defined by a generic formula: A- (L-M)n, wherein A is a binding molecule (e.g., an antibody), L is an optional linker, and M is a heterologous moiety which can be for example a therapeutic agent, a detectable label, etc., and n is an integer.
- multiple heterologous moieties can be chemically conjugated to the different attachment points in the same binding molecule (e.g., an antibody). In some aspects, multiple heterologous moieties can be concatenated and attached to an attachment point in the binding molecule (e.g., an antibody). In some aspects, multiple heterologous moieties (being the same or different) can be conjugated to the binding molecule (e.g., an antibody).
- Immunoconjugates can also be defined by the generic formula in reverse order. In some aspects, the immunoconjugate is an "antibody-Drug Conjugate" ("ADC").
- the term “immunoconjugate” is not limited to chemically or enzymatically conjugates molecules.
- the term “immunoconjugate” as used in the present disclosure also includes genetic fusions.
- the biologically active molecule is an immunoconjugate.
- the terms “antibodydrug conjugate” and “ADC” are used interchangeably and refer to an antibody linked, e.g., covalently, to a therapeutic agent (sometimes referred to herein as agent, drug, or active pharmaceutical ingredient) or agents.
- the biologically active molecule i.e., a payload
- the biologically active molecule is an antibody-drug conjugate.
- Treating refers to, e.g., the reduction in severity of a disease or condition; the reduction in the duration of a disease course; the amelioration or elimination of one or more symptoms associated with a disease or condition; the provision of beneficial effects to a subject with a disease or condition, without necessarily curing the disease or condition.
- the term also include prophylaxis or prevention of a disease or condition or its symptoms thereof.
- the term “treating” or “treatment” means inducing an immune response in a subject against an antigen.
- Prevent refers to decreasing or reducing the occurrence or severity of a particular outcome. In some aspects, preventing an outcome is achieved through prophylactic treatment.
- compositions comprising an EV (e.g., exosome), which has been modified to comprise a KRAS antigen.
- a composition provided herein further comprises an adjuvant, a T helper peptide, or both.
- the composition can further comprise one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator).
- an adjuvant, T helper peptide, and/or additional moieties of interest described herein are associated with the EV (e.g., exosome).
- a moiety of interest e.g., adjuvant, T helper peptide, targeting moiety, and/or immune modulator
- an EV e.g. , exosome
- the moiety of interest is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- an adjuvant, T helper peptide, and/or additional moieties of interest present in the compositions described herein are not associated with the EV (e.g., exosome).
- a moiety of interest e.g., adjuvant, T helper peptide, targeting moiety, and/or immune modulator
- the EV e.g., exosome
- Extracellular Vesicles e.g., Exosomes
- EVs e.g., exosomes
- EVs described herein are extracellular vesicles with a diameter between about 20-300 nm.
- an EV e.g.
- exosome of the present disclosure has a diameter between about 20-80 nm, between about 80-300 nm, between about 80-290 nm, between about 80-280 nm, between about 80-270 nm, between about 80-260 nm, between about 80-250 nm, between about 80-240 nm, between about 80-230 nm, between about 80-220 nm, between about 80-210 nm, between about 80-200 nm, between about 80-190 nm, between about 80-180 nm, between about 80-170 nm, between about 80-160 nm, between about 80-150 nm, between about 80-140 nm, between about 80-130 nm, between about 80-120 nm, between about 80-110 nm, between about 80-100 nm, between about 80-90 nm, between about 90-300 nm, between about 90-290 nm, between about 90-280 nm, between about 90-270 nm,
- an EV (e.g., exosome) comprises a bi-lipid membrane comprising an interior surface and an exterior surface.
- the interior surface faces the inner core (i.e., lumen) of the EV (e.g., exosome).
- the exterior surface can be in contact with the endosome, the multivesicular bodies, or the membrane/cytoplasm of a producer cell or a target cell.
- scaffold moieties are polypeptides ("exosome proteins").
- scaffold moieties are non-polypeptide moieties.
- exosome proteins include various membrane proteins, such as transmembrane proteins, integral proteins and peripheral proteins, enriched on the exosome membranes.
- a scaffold moiety (e.g., exosome protein) comprises Scaffold X.
- a scaffold moiety (e.g., exosome protein) comprises Scaffold Y.
- a scaffold moiety (e.g., exosome protein) comprises both a Scaffold X and a Scaffold Y.
- EVs e.g., exosomes
- a moiety of interest e.g. , KRAS antigen, adjuvant, T cell helper peptide, targeting moiety, and/or immune modulator
- an EV e.g. , exomsome
- a composition comprising the EV e.g., comprising a KRAS antigen
- an immune response e.g., CD8+ T cell response, CD4+ T cell response, B cell response, or any combination thereof
- an immune response e.g., CD8+ T cell response, CD4+ T cell response, B cell response, or any combination thereof
- EVs e.g., exosomes
- the EVs comprise one or more of the following properties: (i) flexibility of moiety display, (ii) diverse combinations of moieties of interest, (iii) enhanced cell-specific tropism, (iv) selectively promoting T-cell, B-cell, or Treg/tolerogenic immune responses, or (v) any combination thereof.
- EVs e.g., exosomes
- a moiety of interest e.g., KRAS antigen, adjuvant, T cell helper peptide, targeting moiety, and/or immune modulator
- a scaffold moiety e.g., Scaffold X and/or Scaffold Y
- a surface of the EV e.g., exterior surface and/or luminal surface
- iii can be expressed in the lumen of the EV, or (iv) combinations thereof.
- EV e.g., exosome
- a single EV e.g., exosome
- a moiety of interest e.g. , antigen
- rapidly attaching a moiety e.g., antigen of interest
- EVs that are useful for the present disclosure allow for the diverse combinations of different moieties of interest (e.g., KRAS antigen, adjuvant, T cell helper peptide, targeting moiety, and/or immune modulator).
- an EV e.g., exosome
- an EV that can be used with the present disclosure comprises: (i) a KRAS antigen and (ii) an adjuvant.
- the EV e.g., exosome
- the EV comprises: (i) a KRAS antigen, (ii) an adjuvant, and (iii) a T helper peptide.
- the EV e.g.
- exosome comprises: (i) a KRAS antigen, (ii) an adjuvant, (iii) a T helper peptide, and (iv) an additional moiety of interest (e.g., targeting moiety and/or immune modulator).
- the EV e.g., exosome
- the EV can comprise a single KRAS antigen, a single adjuvant, a single T helper peptide, and/or single additional moiety of interest (e.g. , targeting moiety and/or immune modulator).
- the EV e.g., exosome
- EVs e.g., exosomes
- the EVs can be engineered to exhibit enhanced cell-specific tropism.
- the EVs can be engineered to express on their exterior surface a targeting moiety (e.g. , antibodies and/or proteins) that can specifically bind to a marker on a specific cell.
- EVs (e.g. , exosomes) described herein can be engineered to induce certain types of immune responses (e.g., T cell, B cell, and/or Treg/tolerogenic immune responses). Additional disclosure relating to such properties are provided elsewhere in the present disclosure.
- B KRAS Antigen
- EVs e.g., exosomes
- KRAS antigen an antigen derived from a KRAS protein
- KRAS antigen does not comprise an antigen derived from a wild-type KRAS protein.
- Kirsten rat sarcoma viral oncogene homology is a member of a superfamily of guanosine-5-triphosphatase (GTPase) proteins that also includes NRAS and HRAS.
- GTPase guanosine-5-triphosphatase
- the primary role of the members of this superfamily is to transmit signals from upstream cell surface receptors (e.g., EGFR, FGFR, and ERBB2-4) to downstream proliferation and survival pathways such as RAF-MEK-ERK, PI3K-AKT- mTOR. and RALGDS-RA.
- KRAS mutations have been implicated in many types of cancers, including more than 90% of pancreatic cancers, 35-45% of colorectal cancers, and approximately 25% of lung cancers.
- KRAS is known in the art by various names. Such names include: KRAS Proto-Oncogene, GTPase; V-Ki-Ras2 Kirsten Rat Sarcoma 2 Viral Oncogene Homolog; GTPase KRas; C-Ki-Ras; K-Ras 2; KRAS2; RASK2; V-Ki-Ras2 Kirsten Rat Sarcoma Viral Oncogene Homolog; Kirsten Rat Sarcoma Viral Proto-Oncogene; Cellular Transforming Proto-Oncogene; Cellular C-Ki-Ras2 Proto-Oncogene; Transforming Protein P21; PR310 C-K-Ras Oncogene; C-Kirsten-Ras Protein; K-Ras P21 Protein; and Oncogene KRAS2.
- the sequence for human KRAS protein can be found under publicly available Accession Numbers: P01116 (canonical sequence), A8K8Z5, B0LPF9, P01118, and Q96D10, each of which is incorporated by reference herein in its entirety.
- P01116 canonical sequence
- A8K8Z5, B0LPF9, P01118, and Q96D10 is incorporated by reference herein in its entirety.
- Isoform 2A is the canonical sequence. It is also known as K-Ras4A.
- Isoform 2B (Accession Number: P01116- 2; also known as K-Ras4B; SEQ ID NO: 382) differs from the canonical sequence as follows: (i) 151-153: RVE GVD; and (ii) 165-189: QYRLKKISKEEKTPGCVKIKKCIIM (SEQ ID NO: 389) KHKEKMSKDGKKKKKKSKTKCVIM (SEQ ID NO: 390).
- Natural variants of the human KRAS gene product are known.
- natural variants of human KRAS protein can contain one or more amino acid substitutions selected from: K5E, K5N, G10GG, G10V, G12A, G12C, G12D, G12F, G12I, G12L, G12R, G12S, G12V, G13A, G13C, G13D, G13E, G13R, G13S, G13V, V14I, A18D, L19F, T20M, Q22E, Q22H, Q22K, Q22R, Q25H, N26Y, F28L, E31K, D33E, P34L, P34Q, P34R, I36M, R41K, D57N, T58I, A59T, G60D, G60R, G60S, G60V, Q61A, Q61H, Q61K, Q61L, Q61P, Q61R, E63K, S65N, R
- a KRAS antigen that is useful for the present disclosure does not comprise a wild-type KRAS protein (e.g., SEQ ID NO: 381 and 382, as shown in Table 1). Accordingly, a KRAS antigen that is useful for the present disclosure is derived from a KRAS mutant protein, such that the KRAS antigen comprises one or more amino acid mutations present in the KRAS mutant protein.
- KRAS mutant protein and “KRAS variant protein” can be used interchangeably and refer to a KRAS protein that differs in sequence from the wild-type KRAS protein (e.g., SEQ ID NO: 381 or 382). Unless indicated otherwise, the term "KRAS antigen" comprises both wild-type and KRAS mutant proteins.
- a KRAS antigen that is useful for the present disclosure can be derived from any KRAS mutant proteins known in the art, such as those described herein (e.g., comprising one or more of the amino acid substitutions described above).
- the KRAS antigen is derived from a KRAS mutant and comprises one or more of the following amino acid substitutions: G12D, G12C, G12V, G12A, G12R, G12S, G13C, G13D, G13A, G13R, G13S, G13V, Q61H, Q61L, Q61R, Q61K, A18D, K117N, or any combination thereof, as compared to the wild-type KRAS protein (SEQ ID NO: 381).
- the KRAS antigen comprises one or more amino acid substitutions selected from the group consisting ofG12D, G12C, G12V, G13D, and any combination thereof, as compared to the wild-type KRAS protein. (SEQ ID NO: 381).
- a KRAS antigen useful for the present disclosure comprises a natural peptide.
- the KRAS antigen comprises a synthetic peptide.
- the KRAS antigen comprises both a natural and a synthetic peptide.
- the KRAS antigen comprises or consists of an epitope within amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids in length.
- the KRAS antigen comprises an epitope within amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids, in length.
- the KRAS antigen consists of an epitope within amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids, in length.
- the KRAS antigen comprises or consists of an epitope within amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length.
- the KRAS antigen comprises an epitope within amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, , at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length.
- the KRAS antigen consists of an epitope within amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, , at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length.
- the KRAS antigen comprises or consists of an epitope within amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least
- the KRAS antigen comprises an epitope within amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least 37 amino
- the KRAS antigen consists of an epitope within amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least
- the overall length of a peptide can influence both the type (e.g. , CD8+ T cell response, CD4+ T cell response, and/or B cell response) and the magnitude of immune response induced against the antigen. Khan, M., et al., Set Rep 9(1): 13321 (Sep. 2019), which is incorporated herein by reference in its entirety.
- a KRAS antigen that is useful for the present disclosure is less than about 50 amino acids in length, less than about 45 amino acids in length, less than about 40 amino acids in length, less than about 35 amino acids in length, less than about 30 amino acids in length, less than about 25 amino acids in length, less than about 20 amino acids in length, less than about 15 amino acids in length, or less than about 10 amino acids in length.
- the KRAS antigen is between about 12 to about 20 amino acids in length.
- an EV (e.g., exosome) of the present disclosure comprises a single KRAS antigen.
- an EV (e.g., exosome) disclosed herein comprises multiple KRAS antigens (e.g., at least about 2, at least about 3, at least about 4, at least about 5, at least about 6, at least about 7, at least about 8, at least about 9, or at least about 10).
- each of the multiple KRAS antigens is different (e.g., each of the KRAS antigens comprise a different amino acid substitution, compared to a corresponding KRAS antigen derived from a wild-type KRAS protein).
- some or all of the multiple KRAS antigens are the same.
- a suitable spacer comprises the amino acid sequence selected from GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), AAY, or combinations thereof.
- an EV e.g., exosome
- the multiple KRAS antigens are separated by the GSGSG (SEQ ID NO: 391) spacer.
- the multiple KRAS antigens are separated by the AAY spacer.
- the multiple KRAS antigens are separated by any combination of the GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), and AAY spacers.
- the KRAS antigen can be encapsulated within the lumen of the EV.
- the KRAS antigen is linked to the exterior surface and/or luminal surface of the EVs (e.g., exosomes) using a scaffold moiety (e.g., Scaffold X and/or Scaffold Y).
- a KRAS antigen can be directly linked (i.e., without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g. , exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof.
- an antigen comprises a peptide, which has been modified to contain a N-terminal lysine.
- such a modification allows for the attachment of the antigen to a surface of the EV (e.g., exosome) with chemical conjugation.
- a surface of the EV e.g., exosome
- an azide or strained alkyne e.g., difluorinated cyclooctyne (DIFO), dibenzocyclooctyne (DBCO), or bicyclononyne (BCN)
- DIFO difluorinated cyclooctyne
- DBCO dibenzocyclooctyne
- BCN bicyclononyne
- the azide or strained alkyne e.g.
- the azide can be attached to the antigen (via the primary amine side chain on the N-terminal amino acid (e.g., lysine)), and the strained alkyne can be attached to a surface of the EV (e.g., exosome).
- modifying the antigen to comprise a N-terminal lysine can also be useful in linking the antigens to a surface of the EVs (e.g., exosomes) using anchoring moieties.
- the anchoring moiety e.g. , cholesterol, fatty acid, and/or vitamin E
- the antigens can be readily inserted into the membrane of the EVs (e.g., exosomes) via the anchoring moieties.
- the above described approaches to linking an antigen to the exterior surface and/or luminal surface of the EVs can also be performed by modifying one or more proteins on the EVs to contain unnatural amino acids with side chains to allow for the binding of molecules such as the azide, strained alkyne (e.g., difluorinated cyclooctyne (DIFO), dibenzocyclooctyne (DBCO), or bicyclononyne (BCN)) or combinations thereof.
- DIFO difluorinated cyclooctyne
- DBCO dibenzocyclooctyne
- BCN bicyclononyne
- CD4+ T cells play an important role in the induction of both T and B cell immune responses against many diseases and disorders. See, e.g., Borst, J., et al., Nat Rev Immunol 18(10): 635-647 (Oct. 2018); and Crotty, S., Nat Rev Immunol 15(3): 185-189 (Mar. 2015); both of which are herein incorporated by reference in their entireties.
- a composition comprising an EV (e.g., exosome) described herein can further comprise a T helper peptide.
- the T helper peptide can be associated with the EV (e.g., exosome), such that the T helper peptide is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- the T helper peptide is not associated with the EV.
- the relevant disclosures provided below can equally apply to both T helper peptides that are associated with the EV (e.g., exosomes) and those that are not.
- T helper peptide refers to any peptide that is capable of activating a CD4+ T cell, which can in turn provide the necessary help to induce the activation of both CD8+ T cells and B cells.
- T helper peptides include: a helper peptide derived from tubercle bacillus, a helper peptide derived from measles virus, a helper peptide derived from hepatitis B virus, a helper peptide derived from hepatitis C virus, a helper peptide derived from Chlamydia trachomatis, a helper peptide derived from P.
- helper peptides derived from cancer cells e.g., IMA-MMP-001 helper peptide, CEA-006 helper peptide, MMP-001 helper peptide, TGFBI-004 helper peptide, HER-2/neu(aa776- 790) helper peptide, AE36 helper peptide, AE37 helper peptide, MET-005 helper peptide, and BIR-002 helper peptide
- cancer cells e.g., IMA-MMP-001 helper peptide, CEA-006 helper peptide, MMP-001 helper peptide, TGFBI-004 helper peptide, HER-2/neu(aa776- 790) helper peptide, AE36 helper peptide, AE37 helper peptide, MET-005 helper peptide, and BIR-002 helper peptide
- universal T helper peptide e.g.,
- the T helper peptide that can be used with the present disclosure is a universal T helper peptide.
- the term "universal T helper peptide” refers to a T helper peptide that binds to a broad range of MHC class II haplotypes, and can thereby activate CD4+ T cells expressing those MHCs.
- the universal T helper peptide comprises a PADRE, tetanus toxin, diphtheria toxin, HBV peptide, measles peptide, or any combination thereof.
- the universal T helper peptide is PADRE (AKFVAAWTLKAAA) (SEQ ID NO: 395), or such as that described in Benmohamed et al., Immunology 106: 113-121 (2001), which is herein incorporated by reference in its entirety.
- the universal T helper peptide is a tetanus toxin (e.g. , QYIKANSKFIGITE; amino acid residues 830-843 of tetanus; SEQ ID NO: 396), or such as that described in Falugi et al. , Eur J Immunol 31: 3816-3824 (2001), which is herein incorporated by reference in its entirety.
- the universal T helper peptide is a diphtheria toxin (e.g. , QSIALSSLMVAQAIP; amino acid residues 356-370 of diphtheria toxin; SEQ ID NO: 397), or such as that described in Diethelm-Okita, et al., J Infec Dis 181: 1001-1009 (2000), which is herein incorporated by reference in its entirety.
- the diphtheria toxin comprises CRM 197.
- the universal T helper peptide is a HBV peptide, such as HbsAg.
- the universal T helper peptide is a peptide derived from a measles virus.
- an EV e.g., exosome
- EVs (e.g. , exosomes) described herein comprises a single T helper peptide.
- an EV (e.g. , exosome) disclosed herein comprises multiple multiple T helper peptides (e.g. , at least 2, 3, 4, 5, 6, 7, 8, 9, 10).
- each of the multiple T helper peptides is different.
- some or all of the multiple T helper peptides are the same.
- a T helper peptide can be encapsulated within the lumen of the EV.
- the T helper peptide is linked to the exterior surface and/or luminal surface of the EVs (e.g., exosomes) using a scaffold moiety (e.g., Scaffold X and/or Scaffold Y).
- the T helper peptide can be directly linked (i.e., without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g., exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof.
- anchoring moieties e.g., affinity agents
- chemical conjugation e.g., click chemistry
- Non-limiting examples of chemical conjugation includes: moiety, copper-free, biorthogonal click chemistry (e.g., azide/strained alkyne (DIFO, DBCO, BCN), metal -catalyzed click chemistry (e.g., CuAAC, RuAAC), or any combination thereof.
- a composition comprising an EV (e.g., exosome) described herein can further comprise an adjuvant.
- the adjuvant can be associated with the EV (e.g., exosome), such that the adjuvant is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- the adjuvant is not associated with the EV.
- the relevant disclosures provided below can equally apply to both adjuvants that are associated with the EV (e.g., exosomes) and those that are not.
- adjuvant refers to any substance that enhances the therapeutic effect of the EVs (e.g., exosomes) of the present disclosure (e.g., increasing an immune response to the KRAS antigen).
- EVs e.g., exsomes described herein comprising an adjuvant are capable of increasing an immune response, e.g., to an antigen, by at least about 5%, at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 100%, at least about 250%, at least about 500%, at least about 750%, at least about 1,000% or more or more, compared to a reference (e.g., corresponding EV without the adjuvant or a non-EV delivery vehicle comprising an antigen alone or in combination with the adjuvant).
- a reference e.g., corresponding EV without the adjuvant or a non-EV delivery vehicle comprising an antigen alone or in combination with the adjuvant.
- incorporating an adjuvant disclosed herein to an EV can increase an immune response, e.g., to an antigen, by at least about 1-fold, at least about 2-fold, at least about 3-fold, at least about 4-fold, at least about 5-fold, at least about 6-fold, at least about 7-fold, at least about 8-fold, at least about 9-fold, at least about 10-fold, at least about 20-fold, at least about 30-fold, at least about 40- fold, at least about 50-fold, at least about 60-fold, at least about 70-fold, at least about 80-fold, at least about 90-fold, at least about 100-fold, at least about 200-fold, at least about 300-fold, at least about 400-fold, at least about 500-fold, at least about 600-fold, at least about 700-fold, at least about 800-fold, at least about 900-fold, at least about 1,000-fold, at least about 2,000-fold, at least about 3,000-fold, at least about 4,000
- EVs (e.g., exosomes) described herein comprises a single adjuvant.
- an EV (e.g., exosome) disclosed herein comprises multiple adjuvants (e.g., at least about 2, at least about 3, at least about 4, at least about 5, at least about 6, at least about 7, at least about 8, at least about 9, or at least about 10).
- each of the multiple adjuvants is different.
- some or all of the multiple adjuvants are the same.
- an adjuvant can be encapsulated within the lumen of the EV.
- the adjuvant is linked to the exterior surface and/or luminal surface of the EVs (e.g. , exosomes) using a scaffold moiety (e.g. , Scaffold X and/or Scaffold Y).
- the adjuvant can be directly linked (i. e. , without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g., exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof. Additional disclosure relating to such approaches (including modifications that can be made to the adjuvants and or EVs for improved binding) to linking the adjuvants to the exterior surface and/or luminal surface of the EVs are provided elsewhere in the present disclosure.
- Non-limiting examples of adjuvants that can be used with the present disclosure include: Stimulator of Interferon Genes (STING) agonist, a toll-like receptor (TLR) agonist, an inflammatory mediator, RIG-I agonists, alpha-gal-cer (NKT agonist), heat shock proteins (e.g., HSP65 and HSP70), C- type lectin agonists (e.g., beta glucan (Dectin 1), chitin, and curdlan), and combinations thereof.
- STING Stimulator of Interferon Genes
- TLR toll-like receptor
- RIG-I agonists e.g., alpha-gal-cer
- HSP65 and HSP70 heat shock proteins
- C- type lectin agonists e.g., beta glucan (Dectin 1), chitin, and curdlan
- an adjuvant e.g., such as those disclosed herein
- an EV e.g.,
- the diversity of an immune response refers to enhancing the diversity of an immune response.
- the diversity of an immune response can be enhanced through epitope spreading (i.e., inducing and/or increasing an immune response (cellular and/or humoral immune response) against a greater number/variety of epitopes on an antigen).
- the diversity of an immune response can be enhanced through the production of different and/or multiple antibody isotypes (e.g., IgG, IgA, IgD, IgM, and/or IgE).
- an adjuvant can also help regulate the type of immune response induced by the EV (e.g., exosome).
- incorporating an adjuvant to an EV can help drive an immune response towards a more Thl phenotype.
- a "Thl" immune response is generally characterized by the production of IFN-y, which can activate the bactericidal activities of innate cells (e.g. , macrophages), help induce B cells to make opsonizing (marking for phagocytosis) and complement-fixing antibodies, and/or lead to cell-mediated immunity (i. e. , not mediated by antibodies).
- Thl responses are more effective against intracellular pathogens (viruses and bacteria that are inside host cells) and suppressing tumor growth.
- incorporating an adjuvant to an EV can help drive an immune response towards a more Th2 phenotype.
- a "Th2" immune response can be characterized by the release of certain cytokines, such as IL-5 (induces eosinophils in the clearance of parasites) and IL-4 (facilitates B cell isotype switching).
- cytokines such as IL-5 (induces eosinophils in the clearance of parasites) and IL-4 (facilitates B cell isotype switching).
- Th2 responses are more effective against extracellular bacteria, parasites including helminths and toxins.
- Thl7 immune response is mediated by Thl7 cells.
- Thl7 cells refer to a subset of CD4+ T cells characterized by the production of pro-inflammatory cytokines, such as IL- 17A, IL- 17F, IL-21 , IL-22, and granulocyte-macrophage colonystimulating factor (GM-CSF). Thl7 cells are generally thought to play an important role in host defense against infection, by recruiting neutrophils and macrophages to infected tissues.
- incorporating an adjuvant to an EV can help drive an immune response towards a more cellular immune response (e.g., T-cell mediated).
- incorporating an adjuvant to an EV can help drive an immune response towards a more humoral immune response (e.g., antibody -mediated).
- an adjuvant induces the activation of a cytosolic pattern recognition receptor.
- cytosolic pattern recognition receptor includes: stimulator of interferon genes (STING), retinoic acid-inducible gene I (RIG-1), Melanoma Differentiation-Associated protein 5 (MDA5), Nucleotide-binding oligomerization domain, Leucine rich Repeat and Pyrin domain containing (NLRP), inflammasomes, or combinations thereof.
- an adjuvant is a STING agonist.
- Stimulator of Interferon Genes (STING) is a cytosolic sensor of cyclic dinucleotides that is typically produced by bacteria.
- the STING agonist comprises a cyclic dinucleotide STING agonist or a non-cyclic dinucleotide STING agonist. As described herein, in some aspects, the STING agonist is loaded in the lumen of the EV (e.g., exosome).
- exoSTING EVs
- exoSTING EVs
- Non-limiting examples of exoSTING are provided in International Publication No. WO 2019183578A1, which is herein incorporated by reference in its entirety. Further disclosures of useful STING agonists are also provided throughout the present disclosure.
- Cyclic purine dinucleotides such as, but not limited to, cGMP, cyclic di-GMP (c-di-GMP), cAMP, cyclic di-AMP (c-di-AMP), cyclic-GMP-AMP (cGAMP), cyclic di-IMP (c-di-IMP), cyclic AMP- IMP (cAIMP), and any analogue thereof, are known to stimulate or enhance an immune or inflammation response in a patient.
- the CDNs can have 2'2', 2'3', 2'5', 3'3', or 3'5' bonds linking the cyclic dinucleotides, or any combination thereof.
- Cyclic purine dinucleotides can be modified via standard organic chemistry techniques to produce analogues of purine dinucleotides.
- Suitable purine dinucleotides include, but are not limited to, adenine, guanine, inosine, hypoxanthine, xanthine, isoguanine, or any other appropriate purine dinucleotide known in the art.
- the cyclic dinucleotides can be modified analogues. Any suitable modification known in the art can be used, including, but not limited to, phosphorothioate, biphosphorothioate, fluorinate, and difluorinate modifications.
- Non cyclic dinucleotide agonists can also be used, such as 5,6-Dimethylxanthenone-4-acetic acid (DMXAA), or any other non-cyclic dinucleotide agonist known in the art.
- DMXAA 5,6-Dimethylxanthenone-4-acetic acid
- Non-limiting examples of STING agonists that can be used with the present disclosure include: DMXAA, STING agonist- 1, ML RR-S2 CD A, ML RR-S2c-di-GMP, ML-RR-S2 cGAMP, 2'3'-c- di-AM(PS)2, 2'3'-cGAMP, 2'3'-cGAMPdFHS, 3'3'-cGAMP, 3'3'-cGAMPdFSH, cAIMP, cAIM(PS)2, 3'3'- cAIMP, 3'3'-cAIMPdFSH, 2'2'-cGAMP, 2'3'-cGAM(PS)2, 3'3'-cGAMP, and combinations thereof.
- Nonlimiting examples of the STING agonists can be found at US Patent No. 9,695,212, WO 2014/189805 Al, WO 2014/179335 Al, WO 2018/100558 Al, US Patent No. 10,011,630 B2, WO 2017/027646 Al, WO 2017/161349 Al, and WO 2016/096174 Al, each of which is incorporated by reference in its entirety.
- the STING agonist useful for the present disclosure comprises the compound or a pharmaceutically acceptable salt thereof. See WO 2016/096174 Al, which is incorporated herein by reference in its entirety.
- the STING agonist useful for the present disclosure comprises a compound described in WO 2014/093936, WO 2014/189805, WO 2015/077354, Cell reports 11, 1018-1030 (2015), WO 2013/185052, Sci. Transl. Med. 283,283ra52 (2015), WO 2014/189806, WO 2015/185565, WO 2014/179760, WO 2014/179335, WO 2015/017652, WO 2016/096577, WO 2016/120305, WO
- the STING agonist useful for the present disclosure is CL606, CL611, CL602, CL655, CL604, CL609, CL614, CL656, CL647, CL626, CL629, CL603, CL632, CL633, CL659, or a pharmaceutically acceptable salt thereof.
- the STING agonist useful for the present disclosure is CL606 or a pharmaceutically acceptable salt thereof.
- the STING agonist useful for the present disclosure is CL611 or a pharmaceutically acceptable salt thereof.
- the STING agonist useful for the present disclosure is CL602 or a pharmaceutically acceptable salt thereof.
- the STING agonist useful for the present disclosure is CL655 or a pharmaceutically acceptable salt thereof.
- the STING agonist useful for the present disclosure is CL604 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL609 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL614 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL656 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL647 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL626 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL629 or a pharmaceutically acceptable salt thereof.
- the STING agonist useful for the present disclosure is CL603 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL632 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL633 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL659 or a pharmaceutically acceptable salt thereof.
- STING agonists can be modified to allow for better expression of the agonists on the surface of the EV (e.g. , exterior and/or luminal surface of the EV, e.g. , exosome, (e.g. , linked to a scaffold moiety disclosed herein, e.g., Scaffold X and/or Scaffold Y)). Any of the modifications described above can be used.
- an adjuvant is a TLR agonist.
- TLR agonists include: TLR2 agonist (e.g., lipoteichoic acid, atypical LPS, MALP-2 and MALP-404, OspA, porin, LcrV, lipomannan, GPI anchor, lysophosphatidylserine, lipophosphoglycan (LPG), glycophosphatidylinositol (GPI), zymosan, hsp60, gH/gL glycoprotein, hemagglutinin), a TLR3 agonist (e.g., double-stranded RNA, e.g., poly(EC), ampligen, hiltonol, polyA:U), a TLR4 agonist (e.g., lipopolysaccharides (LPS), lipoteichoic acid, P-defensin 2, fibronectin EDA, HMGB1, snapin
- TLR2 agonist e.
- adjuvant comprises a saponin-based agent, e.g., Quil-A.
- TLR agonists can be found at WO2008115319A2, US20130202707A1, US20120219615A1, US20100029585A1, W02009030996A1, W02009088401A2, and WO2011044246A1, each of which are incorporated by reference in its entirety.
- the adjuvant is a TLR4 agonist.
- the TLR4 agonist comprises MPLA.
- the adjuvant is a TLR7/8 agonist.
- the TLR7/8 agonist comprises Resiquimod (R848).
- the TLR7/8 agonist comprises 3M-052.
- a composition comprising an EV (e.g., exosome) described herein can comprise one or more additional moieties of interest.
- the additional moiety of interest can be associated with the EV (e.g. , exosome), such that the additional moiety of interest is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
- the additional moiety of interest is not associated with the EV.
- the relevant disclosures provided below can equally apply to additional moieties of interest that are associated with the EV (e.g., exosomes) and those that are not.
- the additional moieties of interest can further enhance the therapeutic effects of the EVs (e.g., exosomes), e.g., when administered to a subject.
- the additional moiety of interest can help direct EV uptake (e.g., targeting moiety), activate, or block cellular pathways to enhance the combinatorial effects associated with the EVs (e.g., exosomes), e.g., when administered to a subject.
- EVs (e.g., exosomes) described herein comprises a single additional moiety of interest (e.g., targeting moiety or immune modulator).
- an EV (e.g., exosome) disclosed herein comprises multiple additional moieties of interest (e.g. , at least about 2, at least about 3, at least about 4, at least about 5, at least about 6, at least about 7, at least about 8, at least about 9, or at least about 10) (e.g. , both a targeting moiety and an immune modulator).
- each of the multiple adjuvants is different. In some aspects, some or all of the multiple adjuvants are the same.
- an adjuvant can be encapsulated within the lumen of the EV.
- the adjuvant is linked to the exterior surface and/or luminal surface of the EVs (e.g. , exosomes) using a scaffold moiety (e.g. , Scaffold X and/or Scaffold Y).
- the adjuvant can be directly linked (i. e. , without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g., exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof.
- the additional moiety of interest comprises a targeting moiety that can modify the distribution of the EVs in vivo or in vitro.
- the targeting moiety can be a biological molecule, such as a protein, a peptide, a lipid, or a synthetic molecule.
- a targeting moiety e.g., tropism moiety
- a targeting moiety of the present disclosure specifically binds to a marker (or target molecule) expressed on a cell or a population of cells.
- the marker is expressed on multiple cell types, e.g. , all antigen-presenting cells (e.g. , dendritic cells, macrophages, and B lymphocytes).
- the marker is expressed only on a specific population of cells (e.g., dendritic cells).
- a targeting moiety of the present disclosure specifically binds to a marker for a dendritic cell.
- the marker is expressed only on dendritic cells.
- dendritic cells comprise a progenitor (Pre) dendritic cells, inflammatory mono dendritic cells, plasmacytoid dendritic cell (pDC), a myeloid/conventional dendritic cell 1 (cDCl), a myeloid/conventional dendritic cell 2 (cDC2), inflammatory monocyte derived dendritic cells, Langerhans cells, dermal dendritic cells, lysozyme-expressing dendritic cells (LysoDCs), Kupffer cells, nonclassical monocytes, or any combination thereof. Markers that are expressed on these dendritic cells are known in the art. See, e.g., Collin et al., Immunology 154(l):3-20 (2018).
- the targeting moiety is a protein, wherein the protein is an antibody or a fragment thereof that can specifically bind to a marker selected from lymphocyte antigen 75 (DEC205), C-type lectin domain family 9 member A (CLEC9A), C-type lectin domain family 6 (CLEC6), ), C-type lectin domain family 4 member A (also known as DCIR or CLEC4A), Dendritic Cell- Specific Intercellular adhesion molecule -3 -Grabbing Non-integrin (also known as DC-SIGN or CD209), lectin-type oxidized LDL receptor 1 (LOX-1), macrophage receptor with collagenous structure (MARCO), C-type lectin domain family 12 member A (CLEC12A), C-type lectin domain family 10 member A (CLEC10A), DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin- 1, macrophage mannose
- a marker selected from lymphocyte antigen 75 (DEC205), C-
- a targeting moiety useful for the present disclosure is capable of binding a marker expressed on CD8+ dendritic cells and thereby, the target an EV (e.g., exosome) to the CD8+ dendritic cells for uptake.
- the targeting moiety binds to Clec9A, XCR1, CD 103, or combinations thereof.
- a targeting moiety disclosed herein can comprise a peptide, an antibody or an antigen binding fragment thereof, a chemical compound, or any combination thereof.
- the targeting moiety is an antibody or an antigen binding fragment thereof.
- a targeting moiety is a single-chain Fv antibody fragment.
- a targeting moiety is a single-chain F(ab) antibody fragment.
- a targeting moiety is a nanobody.
- a targeting moiety is a monobody.
- an EV e.g., exosome
- an EV can be surface engineered to adjust its properties, e.g., biodistribution, e.g., via incorporation of immuno-affinity ligands or cognate receptor ligands.
- EVs e.g., exosomes
- described herein can be engineered to direct the EVs to a particular tissue within a subject.
- the EVs (e.g., exosomes) comprising a KRAS antigen can be modified to specifically target tissues where T cell priming would likely occur.
- Nonlimiting examples of such tissues include: tumor draining lymph nodes, sentinel lymph nodes, tumor tertiary lymph nodes, or any combination thereof.
- EVs e.g., exosomes
- an immune response against the KRAS antigen could be increased compared to a corresponding EV (e.g., exosome) without the targeting/tropism moiety disclosed herein.
- targeting moieties that could be used include: anti-fibronectin EDA & EDB domain antibodies; anti-ICAM-1; anti-CD20; anti-DC-LAMP; or any combination thereof.
- EVs e.g., exosomes
- a tropism moiety can increase uptake of the EV, e.g., an exosome, by a cell.
- the tropism moiety that can increase uptake of the EV, e.g., an exosome, by a cell comprises a lymphocyte antigen 75 (also known as DEC205 or CD205), C-type lectin domain family 9 member A (CLEC9A), C-type lectin domain family 6 (CLEC6), C-type lectin domain family 4 member A (also known as DCIR or CLEC4A), Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin (also known as DC-SIGN or CD209), lectin-type oxidized LDL receptor l(LOX-l), macrophage receptor with collagenous structure (MARCO), C-type lectin domain family 12 member A (CLEC12A), C-type lectin domain family 10 member A (CLEC10A), DC-asialogly
- an EV, e.g., exosome, of the present disclosure can comprise a tissue or cell-specific target ligand, which increases EV, e.g., exosome, tropism to a specific central nervous system tissue or cell.
- the cell is a glial cell.
- the glial cell is an oligodendrocyte, an astrocyte, an ependymal cell, a microglia cell, a Schwann cell, a satellite glial cell, an olfactory ensheathing cell, or a combination thereof.
- the cell is a neural stem cell.
- the cell-specific target ligand which increases EV, e.g., exosome, tropism to a Schwann cells, binds to a Schwann cell surface marker, such as Myelin Basic Protein (MBP), Myelin Protein Zero (P0), P75NTR, NCAM, PMP22, or any combination thereof.
- a Schwann cell surface marker such as Myelin Basic Protein (MBP), Myelin Protein Zero (P0), P75NTR, NCAM, PMP22, or any combination thereof.
- MBP Myelin Basic Protein
- P0 Myelin Protein Zero
- P75NTR e.g., NCAM
- PMP22 binds to a Schwann cell surface marker, such as Myelin Basic Protein (MBP), Myelin Protein Zero (P0), P75NTR, NCAM, PMP22, or any combination thereof.
- the cell-specific tropism moiety comprises an antibody or an antigen-binding portion thereof, an apta
- the tropism moiety is linked, e.g., chemically linked via a maleimide moiety, to a scaffold moiety, e.g., a Scaffold X protein or a fragment thereof, on the exterior surface of the EV, e.g., exosome.
- a scaffold moiety e.g., a Scaffold X protein or a fragment thereof
- Tropism can be further improved by the attachment of a half-life extension moiety (e.g., albumin or PEG), or any combination thereof to the external surface of an EV, e.g., exosome of the present disclosure.
- a half-life extension moiety e.g., albumin or PEG
- Pharmacokinetics, biodistribution, and in particular, tropism and retention in the desired tissue or anatomical location can also be accomplish by selecting the appropriate administration route (e.g., intrathecal administration or intraocular administration to improve tropism to the central nervous system).
- administration route e.g., intrathecal administration or intraocular administration to improve tropism to the central nervous system.
- Surface antigens useful in the present disclosure include, but are not limited to, antigens that label a cell as a "self cell.
- the surface antigen is selected from CD47, CD24, a fragment thereof, and any combination thereof.
- the surface antigen comprises CD24, e.g., human CD24.
- the surface antigen comprises a fragment of CD24, e.g., human CD24.
- the EV, e.g., exosome is modified to express CD47 or a fragment thereof on the exterior surface of the EV, e.g., exosome.
- the additional moiety of interest comprises an immune modulator (e.g., along with an antigen, adjuvant, and/or additional moiety of interest described herein).
- a base EV e.g. , exosome
- the immune modulator e.g., alone or in combination with the adjuvant and/or additional moiety of interest described herein, such that the immune modulator is present in the EV prior to the addition of the antigen.
- the immune modulator can be introduced into a producer cell when producing the base EVs (e.g., exosomes).
- the immune modulator can be added to the EVs (e.g., exosomes) after being isolated from the producer cells. In such aspects, the immune modulator can be added to the isolated EVs (e.g., exosomes) before adding the antigen. In some aspects, the immune modulator is added to the EV after adding the antigen. In some aspects, the immune modulator is added to the EV together with the antigen.
- an immune modualtor comprises an inhibitor for a negative checkpoint regulator or an inhibitor for a binding partner of a negative checkpoint regulator.
- the negative checkpoint regulator comprises cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1), lymphocyte -activated gene 3 (LAG-3), T-cell immunoglobulin mucin-containing protein 3 (TIM-3), B and T lymphocyte attenuator (BTLA), T cell immunoreceptor with Ig and ITIM domains (TIGIT), V-domain Ig suppressor of T cell activation (VISTA), adenosine A2a receptor (A2aR), killer cell immunoglobulin like receptor (KIR), indoleamine 2,3 -dioxygenase (IDO), CD20, CD39, CD73, or any combination thereof.
- CTLA-4 cytotoxic T-lymphocyte-associated protein 4
- PD-1 programmed cell death protein 1
- LAG-3 lymphocyte -activated gene 3
- TIM-3
- the immunomodulating component is a protein, a peptide, a glycolipid, or a glycoprotein.
- an antigen can be linked to an exterior surface and/or luminal surface of the EVs using an anchoring moiety or any other coupling strategy described herein or known in the art.
- the coupling strategy comprises: an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), split intein, SpyTag/SpyCatcher, ALFA-tag, Streptavidin/Avitag, Sortase, SNAP-tag, ProA/Fc-binding peptide, or any combinations thereof.
- the antigen, adjuvant, and/or one or more additional moieties of interest is linked directly to the anchoring moiety or via a linker.
- the antigen, adjuvant, and/or one or more additional moieties of interest can be attached to an anchoring moiety or linker combination via reaction between a "reactive group” (RG; e.g., amine, thiol, hydroxy, carboxylic acid, or azide) with a "reactive moiety” (RM; e.g., maleimide, succinate, NHS).
- RG reactive group
- RM reactive moiety
- an anchoring moiety can be chemically conjugated to the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) to enhance its hydrophobic character.
- additional moieties of interest e.g., targeting moiety and/or immune modulator
- the antigen, adjuvant, and/or one or more additional moieties of interest are modified, such that they include a free amine group at the N-terminus (e.g. , comprising a N-terminal lysine).
- the anchoring moiety is conjugated to the free amine group at the N-terminus either directly or via one or more linkers.
- anchoring moieties that can be used to link an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) to the exterior surface and/or luminal surface of the EV (e.g., targeting moiety and/or immune modulator) to the exterior surface and/or luminal surface of the EV (e.g.
- exosome comprises: a sterol (e.g., cholesterol), GM1, a lipid (e.g., fatty acid (e.g., palmitate), ionizable lipid, glycerophospholipid, sphingolipid), a vitamin (e.g., tocopherol (e.g., vitamin E)), vitamin A, vitamin D, vitamin K), a small molecule, a peptide (e.g., cell penetrating peptide), including any derivatives thereof, or a combination thereof.
- the anchoring moiety is a lipid.
- a lipid anchoring moiety can be any lipid known in the art, e.g., palmitic acid or glycosylphosphatidylinositols.
- suitable lipids are described in Fahy et al., Biochim Biophys Acta 1811(11): 637-647 (Nov. 2011), which is incorporated herein by reference in its entirety.
- the lipid is a fatty acid, phosphatide, phospholipid (e.g., phosphatidyl choline, phosphatidyl serine, or phosphatidyl ethanolamine), or analogue thereof (e.g.
- the lipid comprises an ionizable lipid. In some aspects, the lipid comprises a glycerophospholipid. In some aspects, the lipid comprises a sphingolipid.
- anchoring moieties are chemically attached.
- an anchoring moiety can be attached to an an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) enzymatically.
- an anchoring moiety of the present disclosure can comprise two or more types of anchoring moieties disclosed herein.
- an anchoring moiety can comprise two lipids, e.g., a phospholipids and a fatty acid, or two phospholipids, or two fatty acids, or a lipid and a vitamin, or cholesterol and a vitamin.
- the anchoring moiety useful for the present disclosure comprises a sterol, steroid, hopanoid, hydroxysteroid, secosteroid, or analog thereof with lipophilic properties.
- the anchoring moiety comprises a sterol, such as a phytosterol, mycosterol, or zoosterol.
- exemplary zoosterols include cholesterol and 24S-hydroxycholesterol;
- exemplary phytosterols include ergosterol (mycosterol), campesterol, sitosterol, and stigmasterol.
- the sterol is selected from ergosterol, 7-dehydrocholesterol, cholesterol, 24S-hydroxycholesterol, lanosterol, cycloartenol, fucosterol, saringosterol, campesterol, [3-sitosterol, sitostanol, coprostanol, avenasterol, or stigmasterol.
- Sterols can be found either as free sterols, acylated (sterol esters), alkylated (steryl alkyl ethers), sulfated (sterol sulfate), or linked to a glycoside moiety (steryl glycosides), which can be itself acylated (acylated sterol glycosides).
- the anchoring moiety is a cholesterol.
- the anchoring moiety comprises a steroid.
- the steroid is selected from dihydrotestosterone, uvaol, hecigenin, diosgenin, progesterone, or cortisol.
- the anchoring moiety is a fatty acid.
- the fatty acid is a short-chain, medium -chain, or long-chain fatty acid.
- the fatty acid is a saturated fatty acid.
- the fatty acid is an unsaturated fatty acid.
- the fatty acid is a monounsaturated fatty acid.
- the fatty acid is a polyunsaturated fatty acid, such as an omega- 3 or omega-6 fatty acid.
- the anchoring moiety comprises a phospholipid.
- Phospholipids are a class of lipids that are a major component of all cell membranes. They can form lipid bilayers because of their amphiphilic characteristic.
- the structure of the phospholipid molecule generally consists of two hydrophobic fatty acid "tails" and a hydrophilic "head” consisting of a phosphate group.
- a phospholipid can be a lipid according to the following formula: in which R p represents a phospholipid moiety and Ri and R2 represent fatty acid moieties with or without unsaturation that can be the same or different.
- an antigen, adjuvant, and/or one or more additional moieties of interest are provided.
- an EV described herein can comprise multiple coupling strategies described herein (e.g., a cell penetrating peptide and a linker).
- the coupling strategy comprises: an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), split intein, SpyTag/SpyCatcher, ALFA-tag, Streptavidin/Avitag, Sortase, SNAP -tag, ProA/Fc-binding peptide, or any combinations thereof.
- CPP cell penetrating peptide
- split intein SpyTag/SpyCatcher
- ALFA-tag Streptavidin/Avitag
- Sortase SNAP -tag
- ProA/Fc-binding peptide ProA/Fc-binding peptide, or any combinations thereof.
- one of the functions of a linker combination is to provide the optimal spacing between the anchoring moiety and the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator).
- Linkers can be susceptible to cleavage ("cleavable linker”) thereby facilitating release of the biologically active molecule.
- a linker combination disclosed herein can comprise a cleavable linker.
- Such cleavable linkers can be susceptible, for example, to acid-induced cleavage, photoinduced cleavage, peptidase -induced cleavage, esterase-induced cleavage, and disulfide bond cleavage, at conditions under which the biologically active molecule remains active.
- linkers can be substantially resistant to cleavage ("non-cleavable linker").
- the cleavable linker comprises a spacer.
- the spacer is PEG.
- a linker combination comprises at least 2, at least 3, at least 4, at least 5, or at least 6 or more different linkers disclosed herein.
- linkers in a linker combination can be linked by an ester linkage (e.g., phosphodiester or phosphorothioate ester).
- the linker is direct bond between an anchoring moiety and an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator).
- the linker combination comprises a "non-cleavable liker.”
- Non-cleavable linkers are any chemical moiety capable of linking two or more components of an EV (e.g., exosome) disclosed herein, such as an an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator), in a stable, covalent manner and does not fall off under the categories listed above for cleavable linkers.
- non-cleavable linkers are substantially resistant to acid- induced cleavage, photo-induced cleavage, peptidase-induced cleavage, esterase-induced cleavage and disulfide bond cleavage.
- non-cleavable refers to the ability of the chemical bond in the linker or adjoining to the linker to withstand cleavage induced by an acid, photolabile -cleaving agent, a peptidase, an esterase, or a chemical or physiological compound that cleaves a disulfide bond, at conditions under which a cyclic dinucleotide and/or the antibody does not lose its activity.
- the biologically active molecule is attached to the linker via another linker, e.g., a self-immolative linker.
- the linker combination comprises a non-cleavable linker comprising, e.g., tetraethylene glycol (TEG), hexaethylene glycol (HEG), polyethylene glycol (PEG), succinimide, thiosuccinimide, non-peptidyl amide, or any combination thereof.
- the non-cleavable linker comprises a spacer unit to link the biologically active molecule to the non-cleavable linker.
- one or more non-cleavable linkers comprise smaller units (e.g., HEG, TEG, glycerol, C2 to C12 alkyl, and the like) linked together.
- the linkage is an ester linkage (e.g., phosphodiester or phosphorothioate ester) or other linkage.
- the linker combination comprises a non-cleavable linker, wherein the non- cleavable linker comprises a polyethylene glycol (PEG) characterized by a formula R 3 -(O-CH2-CH2) n - or R 3 -(0-CH2-CH2)n-O- with R 3 being hydrogen, methyl or ethyl and n having a value from 2 to 200.
- the linker comprises a spacer, wherein the spacer is PEG.
- the PEG linker is an oligo-ethylene glycol, e.g., diethylene glycol, triethylene glycol, tetra ethylene glycol (TEG), pentaethylene glycol, or a hexaethylene glycol (HEG) linker.
- TEG tetra ethylene glycol
- HOG hexaethylene glycol
- n has a value of 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17,18,19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114
- n is between 2 and 10, between 10 and 20, between 20 and 30, between 30 and 40, between 40 and 50, between 50 and 60, between 60 and 70, between 70 and 80, between 80 and 90, between 90 and 100, between 100 and 110, between 110 and 120, between 120 and 130, between 130 and 140, between 140 and 150, between 150 and 160, between 160 and 170, between 170 and 180, between 180 and 190, or between 190 and 200.
- n has a value from 3 to 200, from 3 to 20, from 10 to 30, or from 9 to 45.
- the PEG is a branched PEG.
- Branched PEGs have three to ten PEG chains emanating from a central core group.
- the PEG moiety is a monodisperse polyethylene glycol.
- a monodisperse polyethylene glycol is a PEG that has a single, defined chain length and molecular weight. mdPEGs are typically generated by separation from the polymerization mixture by chromatography. In certain formulae, a monodisperse PEG moiety is assigned the abbreviation mdPEG.
- the PEG is a Star PEG.
- Star PEGs have 10 to 100 PEG chains emanating from a central core group.
- the PEG is a Comb PEGs.
- Comb PEGs have multiple PEG chains normally grafted onto a polymer backbone.
- the PEG has a molar mass between 100 g/mol and 3000 g/mol, particularly between 100 g/mol and 2500 g/mol, more particularly of approx. 100 g/mol to 2000 g/mol. In certain aspects, the PEG has a molar mass between 200 g/mol and 3000 g/mol, particularly between 300 g/mol and 2500 g/mol, more particularly of approx. 400 g/mol to 2000 g/mol.
- the PEG is PEGwo, PEG200, PEG300, PEG400, PEG500, PEGgoo, PEG700, PEGsoo, PEG900, PEG1000, PEGnoo, PEG1200, PEG1300, PEG1400, PEG1500, PEGigoo, PEG1700, PEGisoo, PEG1900, PEG2000, PEG2100, PEG2200, PEG2300, PEG2400, PEG2500, PEGigoo, PEG1700, PEGigoo, PEG1900, PEG2000, PEG2100, PEG2200, PEG2300, PEG2400, PEG2500, PEG2600, PEG2700, PEG2800, PEG2900, 0 rPEG 3 ooo.
- the PEG is PEG400-
- the PEG is PEG2ooo-
- a linker combination of the present disclosure can comprise several PEG linkers, e.g., a cleavable linker flanked by PEG, HEG, or TEG linkers.
- the linker combination comprises (HEG)n and/or (TEG)n, wherein n is an integer between 1 and 50, and each unit is connected, e.g., via a phosphate ester linker, a phosphorothioate ester linkage, or a combination thereof.
- the linker combination comprises a non-cleavable linker comprising a glycerol unit or a polyglycerol (PG) described by the formula ((R3 — O — (CH2 — CHOH — CEEC n — ) with R3 being hydrogen, methyl or ethyl, and n having a value from 3 to 200.
- n has a value from 3 to 20.
- n has a value from 10 to 30.
- the PG linker is a diglycerol, triglycerol, tetraglycerol (TG), pentaglycerol, or a hexaglycerol (HG) linker.
- the linker combination comprises (glycerol)n, and/or (HG)n and/or (TG)n, wherein n is an integer between 1 and 50, and each unit is connected, e.g., via a phosphate ester linker, a phosphorothioate ester linkage, or a combination thereof.
- the linker combination comprises at least one aliphatic (alkyl) linker, e.g., propyl, butyl, hexyl , or C2-C12 alkyl, such as C2-C10 alkyl or C2-C6 alkyl.
- alkyl e.g., propyl, butyl, hexyl , or C2-C12 alkyl, such as C2-C10 alkyl or C2-C6 alkyl.
- cleavable linker refers to a linker comprising at least one linkage or chemical bond that can be broken or cleaved.
- cleave refers to the breaking of one or more chemical bonds in a relatively large molecule in a manner that produces two or more relatively smaller molecules.
- Cleavage can be mediated, e.g., by a nuclease, peptidase, protease, phosphatase, oxidase, or reductase, for example, or by specific physicochemical conditions, e.g., redox environment, pH, presence of reactive oxygen species, or specific wavelengths of light.
- a nuclease e.g., a nuclease, peptidase, protease, phosphatase, oxidase, or reductase
- specific physicochemical conditions e.g., redox environment, pH, presence of reactive oxygen species, or specific wavelengths of light.
- cleavable refers, e.g., to rapidly degradable linkers, such as, e.g., phosphodiester and disulfides, while the term “non-cleavable” refers, e.g., to more stable linkages, such as, e.g., nuclease-resistant phosphorothioates.
- the cleavable linker is a dinucleotide, trinucleotide, tetranucleotide linker, a disulfide, an imine, a silyl ether, carbonate, a thioketal, a val-cit dipeptide, Vai-Ala dipeptide, Ala-Ala-Asn tripeptide, poly-arginine, phosphodiesters, acid-labile, or any combination thereof.
- the cleavable linker comprises valine-alanine-p-aminobenzylcarbamate, valine-citrulline-p-aminobenzylcarbamate, Ala-Ala-Asn-p-aminobenzylcarbamate.
- the linker combination comprises a redox cleavable linker.
- cleavable linker is a redox cleavable linking group that is cleaved upon reduction or upon oxidation.
- the redox cleavable linker contains a disulfide bond, i.e., it is a disulfide cleavable linker.
- Redox cleavable linkers can be reduced, e.g., by intracellular mercaptans, oxidases, or reductases.
- the linker combination can comprise a cleavable linker which can be cleaved by a reactive oxygen species (ROS), such as superoxide (Of) or hydrogen peroxide (H2O2), generated, e.g., by inflammation processes such as activated neutrophils.
- ROS reactive oxygen species
- the ROS cleavable linker is a thioketal cleavable linker. See, e.g., U.S. Pat. 8,354,455B2, which is herein incorporated by reference in its entirety.
- the linker is an "acid labile linker" comprising an acid cleavable linking group, which is a linking group that is selectively cleaved under acidic conditions (pH ⁇ 7).
- the acid cleavable linking group is cleaved in an acidic environment, e.g., about 6.0, 5.5, 5.0 or less. In some aspects, the pH is about 6.5 or less.
- the linker is cleaved by an agent such as an enzyme that can act as a general acid, e.g., a peptidase (which can be substrate specific) or a phosphatase.
- an enzyme such as a general acid, e.g., a peptidase (which can be substrate specific) or a phosphatase.
- certain low pH organelles such as endosomes and lysosomes, can provide a cleaving environment to the acid cleavable linking group.
- pH of human serum is 7.4, the average pH in cells is slightly lower, ranging from about 7.1 to 7.3. Endosomes also have an acidic pH, ranging from 5.5 to 6.0, and lysosomes are about 5.0 at an even more acidic pH. Accordingly, pH dependent cleavable linkers are sometimes called endosomically labile linkers in the art.
- the linker comprises a low pH-labile hydrazone bond, silyl ether, carbonate, or combinations thereof.
- acid-labile bonds have been extensively used in the field of conjugates, e.g., antibody-drug conjugates. See, for example, Zhou et al, Biomacromolecules 2011, 12, 1460-7; Yuan et al, Acta Biomater. 2008, 4, 1024-37; Zhang et al, Acta Biomater. 2007, 6, 838-50; Yang et al, J. Pharmacol. Exp. Ther. 2007, 321, 462-8; Reddy et al, Cancer Chemother. Pharmacol. 2006, 58, 229-36; Doronina et al, Nature Biotechnol. 2003, 21, 778-84.
- the linker combination can comprise a linker cleavable by intracellular or extracellular enzymes, e.g., proteases, esterases, nucleases, amidades, or reductase.
- enzymes e.g., proteases, esterases, nucleases, amidades, or reductase.
- the range of enzymes that can cleave a specific linker in a linker combination depends on the specific bonds and chemical structure of the linker.
- peptidic linkers can be cleaved, e.g., by peptidades, linkers containing ester linkages can be cleaved, e.g., by esterases; linkers containing amide linkages can be cleaved, e.g., by amidades; linkers containing disulfide linkages can be cleaved, e.g., by reductases; etc.
- the linker combination comprises a protease cleavable linker, i.e., a linker that can be cleaved by an endogenous protease. Only certain peptides are readily cleaved inside or outside cells. See, e.g., Trout et al., 79 Proc. Natl. Acad. Sci. USA, 626-629 (1982) and Umemoto et al. 43 Int. J. Cancer, 677-684 (1989).
- a protease cleavable linker i.e., a linker that can be cleaved by an endogenous protease. Only certain peptides are readily cleaved inside or outside cells. See, e.g., Trout et al., 79 Proc. Natl. Acad. Sci. USA, 626-629 (1982) and Umemoto et al. 43 Int. J. Cancer, 677-684 (1989).
- Cleavable linkers can contain cleavable sites composed of a-amino acid units and peptidic bonds, which chemically are amide bonds between the carboxylate of one amino acid and the amino group of a second amino acid.
- Other amide bonds such as the bond between a carboxylate and the [3-amino acid group of lysine, are understood not to be peptidic bonds and are considered non-cleavable.
- ester cleavable linkers are cleaved by esterases ("esterase cleavable linkers"). Only certain esters can be cleaved by esterases and amidases present inside or outside of cells. Esters are formed by the condensation of a carboxylic acid and an alcohol. Simple esters are esters produced with simple alcohols, such as aliphatic alcohols, and small cyclic and small aromatic alcohols. Examples of ester-based cleavable linking groups include, but are not limited to, esters of alkylene, alkenylene and alkynylene groups. The ester cleavable linking group has the general formula -C (O) O- or -OC (O)-.
- a linker combination can includes a phosphate-based cleavable linking group is cleaved by an agent that degrades or hydrolyzes phosphate groups.
- an agent that cleaves intracellular phosphate groups is an enzyme such as intracellular phosphatase, nuclease, or phosphodiesterase .
- the combination linker comprises a photoactivated cleavable linker, e.g., a nitrobenzyl linker or a linker comprising a nitrobenzyl reactive group.
- the linker combination comprises a self-immolative linker
- the self-immolative linker in the EV (e.g., exosome) of the present disclosure undergoes 1,4 elimination after the enzymatic cleavage of the protease-cleavable linker.
- the self-immolative linker in the EV (e.g., exosome) of the present disclosure undergoes 1,6 elimination after the enzymatic cleavage of the protease-cleavable linker.
- the self-immolative linker is, e.g., a p-aminobenzyl (pAB) derivative, such as a p-aminobenzyl carbamate (pABC), a p-amino benzyl ether (PABE), a p-amino benzyl carbonate, or a combination thereof.
- pAB p-aminobenzyl
- PABE p-amino benzyl ether
- EVs e.g., exosomes
- EVs can accommodate large numbers of molecules attached to their surface, e.g., on the order of thousands to tens of thousands of molecules per EV (e.g., exosome).
- EV (e.g., exosome)-drug conjugates thus represent a platform to deliver a high concentration of therapeutic compound to discrete cell types, while at the same time limiting overall systemic exposure to the compound, which in turn reduces off-target toxicity.
- the present disclosure provide EVs, e.g., exosomes, that have been engineered by reacting a first molecular entity comprising a free thiol group with a second molecular entity comprising a maleimide group, wherein the maleimide moiety covalently links the first molecular entity with the second molecular entity via a maleimide moiety.
- Non-limiting examples of biologically active molecules that can attached to an EV (e.g., exosome) via a maleimide moiety include agents such as, nucleotides (e.g., nucleotides comprising a detectable moiety or a toxin or that disrupt transcription), nucleic acids (e.g., DNA or mRNA molecules that encode a polypeptide such as an enzyme, or RNA molecules that have regulatory function such as miRNA, dsDNA, IncRNA, or siRNA), morpholino, amino acids (e.g., amino acids comprising a detectable moiety or a toxin that disrupt translation), polypeptides (e.g., enzymes), lipids, carbohydrates, small molecules (e.g., small molecule drugs and toxins), antigens (e.g., vaccine antigens), adjuvants (e.g., vaccine adjuvants), etc.
- nucleotides e.g., nucleotides comprising a detectable moiety or
- an EV (e.g., exosome) of the present disclosure can comprise more than one type of biologically active molecule.
- biologically active molecules can be, e.g., small molecules such as cyclic dinucleotides, toxins such as auristatins (e.g., monoethyl auristatin E, MMAE), antibodies (e.g., naked antibodies or antibody -drug conjugates), STING agonists, tolerizing agents, antisense oligonucleotides, PROTACs, morpholines, lysophosphatidic acid receptor antagonists (e.g., LPA1 antagonists) or any combinations thereof.
- auristatins e.g., monoethyl auristatin E, MMAE
- antibodies e.g., naked antibodies or antibody -drug conjugates
- STING agonists e.g., tolerizing agents
- antisense oligonucleotides e.g.,
- an EV (e.g., exosome) of the present disclosure can comprise, e.g., a vaccine antigen and optionally a vaccine adjuvant.
- an EV (e.g., exosome) of the present disclosure can comprise a therapeutic payload (e.g., a STING or one payload disclosed below) and a targeting moiety and/or a tropism moiety.
- EVs e.g., exosomes
- EVs can comprise one or more affinity ligands that link or conjugate a molecule of interest (e.g. , antigen, adjuvant, immune modulator, and/or targeting moiety) to the EVs (e.g., to the exterior surface or on the luminal surface) or to a target cell.
- a molecule of interest e.g. , antigen, adjuvant, immune modulator, and/or targeting moiety
- an affinity ligand disclosed herein has one or more of the following properties: (i) derived from a synthetic library, (ii) sub-nanomolar affinity for a scaffold moiety (e.g., Scaffold X) with emphasis on slow off rate, (iii) binds epitope on membrane-distal IgV domain of a scaffold moiety (e.g., Scaffold X), (iv) free of disulfide linkages, (v) free of N-linked glycosylation sites, (vi) less than 20 amino acids in length, (vii) monomeric, (viii) electroneutral at physiological pH, (ix) hydrophilic, (x) resistant to protease digestion, (xi) amenable to expression in prokaryotic and eukaryotic hosts, (xii) can accommodate N- or C-terminus fusion, (xiii) nonimmunogenic, (xiv) can contain a tag for purification and/or separation, e.g.,
- an affinity ligand disclosed herein can specifically bind (e.g. , with high affinity) to a moiety expressed on the surface of an EV (e.g. , exosome).
- an affinity ligand specifically binds to a scaffold moiety expressed on the surface of an EV.
- an affinity ligand specifically binds to any moiety expressed on the surface of an EV (e.g., cholesterol).
- an affinity ligand disclosed herein can specifically bind (e.g., with high affinity) to a moiety expressed on a target cell. Non-limiting examples of such affinity ligands are provided throughout the present disclosure.
- an affinity ligand useful for the present disclosure can be engineered to express one or more tags.
- tags can be useful in the purification and/or separation of an agent that is conjugated to the affinity ligand.
- an EV e.g., exosome
- an affinity ligand fusion which comprises a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) and a tag.
- the tag can be used to purify and/or separate the EV from a sample comprising the EV.
- a tag of an affinity ligand fusion described above is present between the affinity ligand and the molecule of interest.
- the tag of an affinity ligand fusion described above can be present at an end (e.g., N-terminus) of the molecule of interest, as long as the tag does not interfere with the activity of the molecule of interest. Any tags useful in the art for purifying and/or separating an agent from a sample can be used in the present disclosure.
- Non-limiting examples of such tags include polyhistidine tags, polyarginine tags, glutathione-S-transferase (GST), maltose binding protein (MBP), S-tag, influenza virus HA tag, thioredoxin, staphylococcal protein A tag, FLAGTM epitope, AviTag epitope (for subsequent biotinylation), c-myc epitope, and combinations thereof. See, e.g., U.S. Pat. No. 7,655,413, which is herein incorporated by reference in its entirety.
- a molecule of interest can be expressed on the surface of an EV (e.g., exosome) via a scaffold moiety.
- the molecule of interest can be linked or conjugated to the scaffold moiety via an affinity ligand.
- an affinity ligand can be fused to a molecule of interest (e.g. , antigen, adjuvant, immune modulator, and/or targeting moiety), and then the molecule of interest can be conjugated to a moiety expressed on the surface of an EV (e.g., scaffold moiety) via the affinity ligand.
- the affinity ligand increases the binding of the molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to the moiety on the EV (e.g. , scaffold moiety).
- the binding of the molecule of interest to the moiety on the EV (e.g., scaffold moiety) is increased by at least about one-fold, at least about two-fold, at least about three -fold, at least about four-fold, at least about five-fold, at least about six-fold, at least about seven-fold, at least about eight-fold, at least about nine-fold, at least about 10-fold, at least about 20- fold, at least about 30-fold, at least about 40-fold, at least about 50-fold, at least about 60-fold, at least about 70-fold, at least about 80-fold, at least about 90-fold, at least about 100-fold, at least about 200-fold, at least about 300-fold, at least about 400-fold, at least about 500-fold, at
- an affinity ligand that can be used with the present disclosure comprises a linear peptide.
- an affinity ligand comprises at least about two, at least about three, at least about four, at least about five, at least about seven, at least about eight, at least about nine, at least about 10, at least about 11, at least about 12, at least about 13, at least about 14, at least about 15, at least about 16, at least about 17, at least about 18, at least about 19, at least about 20, at least about 25, at least about 30, at least about 35, at least about 40, at least about 45, at least about 50, at least about 55, at least about 60, at least about 65, at least about 70, at least about 75, at least about 80, at least about 85, at least about 90, at least about 95, or at least about 100 amino acids.
- an affinity ligand e.g., disclosed herein
- link or conjugate a molecule of interest e.g., antigen, adjuvant, immune modulator, and/or targeting moiety
- a moiety expressed on the surface of an EV e.g. , scaffold moiety
- an affinity ligand disclosed herein can allow for increased expression of the molecule of interest on the surface of an EV (e.g. , exterior surface).
- a fusion protein comprising (i) a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety), (ii) an affinity ligand, and (iii) a scaffold moiety is present in the EV (e.g., exterior surface) at a higher density compared to a reference (e.g., corresponding fusion protein without the affinity ligand).
- a molecule of interest e.g., antigen, adjuvant, immune modulator, and/or targeting moiety
- an affinity ligand e.g., an affinity ligand
- a scaffold moiety e.g., exterior surface
- the density of the fusion protein on the surface of the exosome is increased by at least about one-fold, at least about two-fold, at least about three -fold, at least about fourfold, at least about five-fold, at least about six-fold, at least about seven-fold, at least about eight-fold, at least about nine-fold, at least about 10-fold, at least about 20-fold, at least about 30-fold, at least about 40- fold, at least about 50-fold, at least about 60-fold, at least about 70-fold, at least about 80-fold, at least about 90-fold, at least about 100-fold, at least about 200-fold, at least about 300-fold, at least about 400-fold, at least about 500-fold, at least about 600-fold, at least about 700-fold, at least about 800-fold, at least about 900-fold, at least about 1,000-fold, at least about 2,000-fold, at least about 3,000-fold, at least about 4,000- fold, at least about 5,000-fold, at least about 6,000-fold
- an improved binding of a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to a moiety expressed on the surface of an EV (e.g., scaffold moiety) can reduce the time required to produce an EV (e.g., exosome) disclosed herein.
- an affinity ligand disclosed herein can reduce the time required for producing an engineered EV (e.g., exosome) disclosed herein (e.g., comprising a molecule of interest and a scaffold moiety).
- the time required to produce an engineered EV is reduced by at least about 5%, at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, or at least about 90% or more, compared to a reference (e.g., time required to produce the corresponding EV without the affinity ligand).
- an affinity ligand useful for the present disclosure comprises a cleavage site, such as a protease (e.g., thrombin) cleavage site.
- a protease e.g., thrombin
- an EV e.g., exosome
- an affinity ligand disclosed herein can be used in combination with other EVs (e.g., exosomes) disclosed herein.
- EVs e.g., exosomes
- EVs can be produced from a cell grown in vitro or a body fluid of a subject.
- various producer cells e.g., HEK293 cells, CHO cells, and MSCs
- a producer cell is not a naturally- existing dendritic cell, macrophage, B cell, mast cell, neutrophil, Kupffer-Browicz cell, cell derived from any of these cells, or any combination thereof (i.e., non-naturally existing producer cell).
- non-naturally existing producer cell refers to a producer cell (e.g.
- the non-naturally existing producer cell has been modified to express one or more payloads disclosed herein (e.g., antigen, immune modulator, and/or adjuvant.
- the non-naturally existing producer cell has been modified to express one or more targeting moieties disclosed herein.
- the non-naturally existing producer cell has been modified to express one or more scaffold moieties disclosed herein (e.g. , Scaffold X and/or Scaffold Y).
- the producer cell can be genetically modified to comprise one or more exogenous sequences (e.g., encoding an antigen, adjuvant, immune modulator, and/or targeting moiety) to produce the EVs (e.g., exosomes) described herein.
- the genetically-modified producer cell can contain the exogenous sequence by transient or stable transformation.
- the exogenous sequence can be transformed as a plasmid.
- the exogenous sequences can be stably integrated into a genomic sequence of the producer cell, at a targeted site or in a random site.
- a stable cell line is generated for production of lumen-engineered EVs (e.g., exosomes).
- compositions comprising an EV, e.g., exosome, of the present disclosure having the desired degree of purity, and a pharmaceutically acceptable carrier or excipient, in a form suitable for administration to a subject.
- Pharmaceutically acceptable excipients or carriers can be determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions comprising a plurality of extracellular vesicles. (See, e.g., Remington's Pharmaceutical Sciences, Mack Publishing Co., Easton, Pa. 21st ed. (2005)).
- the pharmaceutical compositions are generally formulated sterile and in full compliance with all Good Manufacturing Practice (GMP) regulations of the U.S. Food and Drug Administration.
- GMP Good Manufacturing Practice
- a pharmaceutical composition comprises one or more therapeutic agents and an exosome described herein.
- the EVs, e.g., exosomes are co-administered with of one or more additional therapeutic agents, in a pharmaceutically acceptable carrier.
- the pharmaceutical composition comprising the EV, e.g., exosome is administered prior to administration of the additional therapeutic agents.
- the pharmaceutical composition comprising the EV, e.g., exosome is administered after the administration of the additional therapeutic agents.
- the pharmaceutical composition comprising the EV, e.g., exosome is administered concurrently with the additional therapeutic agents.
- Acceptable carriers, excipients, or stabilizers are nontoxic to recipients (e.g., animals or humans) at the dosages and concentrations employed, and include buffers such as phosphate, citrate, and other organic acids; antioxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine,
- Examples of carriers or diluents include, but are not limited to, water, saline, Ringer's solutions, dextrose solution, and 5% human serum albumin.
- the use of such media and compounds for pharmaceutically active substances is well known in the art. Except insofar as any conventional media or compound is incompatible with the extracellular vesicles described herein, use thereof in the compositions is contemplated. Supplementary therapeutic agents can also be incorporated into the compositions.
- a pharmaceutical composition is formulated to be compatible with its intended route of administration.
- the EVs can be administered by parenteral, topical, intravenous, oral, subcutaneous, intra-arterial, intradermal, transdermal, rectal, intracranial, intraperitoneal, intranasal, intrathecal, intramuscular route or as inhalants.
- the pharmaceutical composition comprising exosomes is administered intravenously, e.g. by injection.
- the EVs, e.g., exosomes can optionally be administered in combination with other therapeutic agents that are at least partly effective in treating the disease, disorder or condition for which the EVs, e.g., exosomes, are intended.
- Solutions or suspensions can include the following components: a sterile diluent such as water, saline solution, fixed oils, polyethylene glycols, glycerin, propylene glycol or other synthetic solvents; antibacterial compounds such as benzyl alcohol or methyl parabens; antioxidants such as ascorbic acid or sodium bisulfite; chelating compounds such as ethylenediaminetetraacetic acid (EDTA); buffers such as acetates, citrates or phosphates, and compounds for the adjustment of tonicity such as sodium chloride or dextrose.
- the pH can be adjusted with acids or bases, such as hydrochloric acid or sodium hydroxide.
- the preparation can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic.
- compositions suitable for injectable use include sterile aqueous solutions (if water soluble) or dispersions and sterile powders.
- suitable carriers include physiological saline, bacteriostatic water, Cremophor ELTM (BASF, Parsippany, N.J.) or phosphate buffered saline (PBS).
- the composition is generally sterile and fluid to the extent that easy syringeability exists.
- the carrier can be a solvent or dispersion medium containing, e.g., water, ethanol, polyol (e.g., glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof.
- the proper fluidity can be maintained, e.g., by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants.
- Prevention of the action of microorganisms can be achieved by various antibacterial and antifungal compounds, e.g., parabens, chlorobutanol, phenol, ascorbic acid, thimerosal, and the like.
- isotonic compounds e.g., sugars, polyalcohols such as mannitol, sorbitol, and sodium chloride can be added to the composition.
- Prolonged absorption of the injectable compositions can be brought about by including in the composition a compound which delays absorption, e.g., aluminum monostearate and gelatin.
- Sterile injectable solutions can be prepared by incorporating the EVs, e.g., exosomes, in an effective amount and in an appropriate solvent with one or a combination of ingredients enumerated herein, as desired.
- dispersions are prepared by incorporating the EVs, e.g., exosomes, into a sterile vehicle that contains a basic dispersion medium and any desired other ingredients.
- methods of preparation are vacuum drying and freeze-drying that yields a powder of the active ingredient plus any additional desired ingredient from a previously sterile-fdtered solution thereof.
- the EVs e.g., exosomes
- compositions comprising exosomes can also be by transmucosal means.
- penetrants appropriate to the barrier to be permeated are used in the formulation.
- penetrants are generally known in the art, and include, e.g., for transmucosal administration, detergents, bile salts, and fusidic acid derivatives.
- Transmucosal administration can be accomplished through the use of, e.g., nasal sprays.
- the pharmaceutical composition comprising exosomes is administered intravenously into a subject that would benefit from the pharmaceutical composition.
- the composition is administered to the lymphatic system, e.g., by intralymphatic injection or by intranodal injection (see e.g., Senti etal., PNAS 105( 46): 17908 (2008)), or by intramuscular injection, by intrathecal administration, by subcutaneous administration, by direct injection into the thymus, or into the liver.
- the pharmaceutical composition comprising exosomes is administered as a liquid suspension.
- the pharmaceutical composition is administered as a formulation that is capable of forming a depot following administration.
- the depot slowly releases the EVs, e.g., exosomes, into circulation, or remains in depot form.
- compositions are highly purified to be free of contaminants, are biocompatible and not toxic, and are suited to administration to a subject. If water is a constituent of the carrier, the water is highly purified and processed to be free of contaminants, e.g., endotoxins.
- the pharmaceutically-acceptable carrier can be lactose, dextrose, sucrose, sorbitol, mannitol, starch, gum acacia, calcium phosphate, alginates, gelatin, calcium silicate, micro-crystalline cellulose, polyvinylpyrrolidone, cellulose, water, syrup, methyl cellulose, methylhydroxy benzoate, propylhydroxy benzoate, talc, magnesium stearate, and/or mineral oil, but is not limited thereto.
- the pharmaceutical composition can further include a lubricant, a wetting agent, a sweetener, a flavor enhancer, an emulsifying agent, a suspension agent, and/or a preservative.
- compositions described herein comprise the EVs, e.g., exosomes, described herein and optionally a pharmaceutically active or therapeutic agent.
- the therapeutic agent can be a biological agent, a small molecule agent, or a nucleic acid agent.
- Dosage forms are provided that comprise a pharmaceutical composition comprising the EVs, e.g., exosomes, described herein.
- the dosage form is formulated as a liquid suspension for intravenous injection.
- the preparation of exosomes is subjected to radiation, e.g., X rays, gamma rays, beta particles, alpha particles, neutrons, protons, elemental nuclei, UV rays in order to damage residual replication-competent nucleic acids.
- radiation e.g., X rays, gamma rays, beta particles, alpha particles, neutrons, protons, elemental nuclei, UV rays in order to damage residual replication-competent nucleic acids.
- the preparation of exosomes is subjected to gamma irradiation using an irradiation dose of more than 1, 5, 10, 15, 20, 25, 30, 35, 40, 50, 60, 70, 80, 90, 100, or more than 100 kGy.
- the preparation of exosomes is subjected to X-ray irradiation using an irradiation dose of more than 0.1, 0.5, 1, 5, 10, 15, 20, 25, 30, 35, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, 500, 600, 700, 800, 900, 1000, 2000, 3000, 4000, 5000, 6000, 7000, 8000, 9000, 10000, or greater than 10000 mSv.
- kits comprising one or more EVs (e.g., exosomes) described herein.
- kits comprising one or more containers fdled with one or more of the ingredients of the pharmaceutical compositions described herein.
- the kit comprises a first container and a second container, wherein the first container comprises a base EV (e.g., exosome) described herein (e.g., comprising an adjuvant), and the second container comprises an antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator), and optional an instruction for use.
- a base EV e.g., exosome
- the second container comprises an antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator), and optional an instruction for use.
- combining the first and second container results in the antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator) to link to a surface of the EV.
- additional moieties of interest e.g., additional adjuvant, targeting moiety, or immune modulator
- kits comprising one or more EVs (e.g., exosomes) described herein.
- kits comprising one or more containers filled with one or more of the ingredients of the pharmaceutical compositions described herein.
- the kit comprises a first container and a second container, wherein the first container comprises a base EV (e.g., exosome) described herein (e.g., comprising an adjuvant), and the second container comprises an antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator), and optional an instruction for use.
- a base EV e.g., exosome
- the second container comprises an antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator), and optional an instruction for use.
- combining the first and second container results in the antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator) to link to a surface of the EV.
- additional moieties of interest e.g., additional adjuvant, targeting moiety, or immune modulator
- the present disclosure is also directed to methods of producing exosomes described herein.
- the method comprises: obtaining the EV, e.g., exosome, from a producer cell, wherein the producer cell contains two or more components of the EV, e.g., exosome (e.g., (i) therapeutic molecule and adjuvant, (ii) therapeutic molecule and immune modulator, or (iii) therapeutic molecule, adjuvant, and immune modulator); and optionally isolating the obtained EV, e.g., exosome.
- exosome e.g., (i) therapeutic molecule and adjuvant, (ii) therapeutic molecule and immune modulator, or (iii) therapeutic molecule, adjuvant, and immune modulator
- optionally isolating the obtained EV e.g., exosome.
- the method comprises: modifying a producer cell by introducing two or more components of an exosome disclosed herein (e.g., (i) therapeutic molecule and adjuvant, (ii) therapeutic molecule and immune modulator, or (iii) therapeutic molecule, adjuvant, and immune modulator); obtaining the EV, e.g., exosome from the modified producer cell; and optionally isolating the obtained EV, e.g. , exosome.
- an exosome disclosed herein e.g., (i) therapeutic molecule and adjuvant, (ii) therapeutic molecule and immune modulator, or (iii) therapeutic molecule, adjuvant, and immune modulator
- obtaining the EV e.g., exosome from the modified producer cell
- optionally isolating the obtained EV e.g. , exosome.
- the method comprises: obtaining an exosome from a producer cell; isolating the obtained exosome; and modifying the isolated exosome (e.g., by inserting multiple exogenous biologically active molecules, e.g., therapeutic molecules, adjuvants, immune modulators, and/or targeting moieties).
- the method further comprises formulating the isolated exosome into a pharmaceutical composition.
- a method of producing an exosome comprises modifying a producer cell with multiple (e.g., two or more) exogenous biologically active molecules described herein (e.g., therapeutic molecule, adjuvant, immune modulator, and/or targeting moiety).
- a producer cell disclosed herein can be further modified with a scaffold moiety disclosed herein (e.g., Scaffold X or Scaffold Y).
- the producer cell can be a mammalian cell line, a plant cell line, an insect cell line, a fungi cell line, or a prokaryotic cell line.
- the producer cell is a mammalian cell line.
- mammalian cell lines include: a human embryonic kidney (HEK) cell line, a Chinese hamster ovary (CHO) cell line, an HT-1080 cell line, a HeLa cell line, a PERC-6 cell line, a CEVEC cell line, a fibroblast cell line, an amniocyte cell line, an epithelial cell line, a mesenchymal stem cell (MSC) cell line, and combinations thereof.
- the mammalian cell line comprises HEK- 293 cells, BJ human foreskin fibroblast cells, fHDF fibroblast cells, AGE.HN® neuronal precursor cells, CAP® amniocyte cells, adipose mesenchymal stem cells, RPTEC/TERT1 cells, or combinations thereof.
- the producer cell is a primary cell.
- the primary cell can be a primary mammalian cell, a primary plant cell, a primary insect cell, a primary fungi cell, or a primary prokaryotic cell.
- the producer cell is not an immune cell, such as an antigen presenting cell, a T cell, a B cell, a natural killer cell (NK cell), a macrophage, a T helper cell, or a regulatory T cell (Treg cell).
- the producer cell is not an antigen presenting cell (e.g., dendritic cells, macrophages, B cells, mast cells, neutrophils, Kupffer-Browicz cell, or a cell derived from any such cells).
- the multiple exogenous biologically active molecules used to modify a producer cell can be a transgene or mRNA, and introduced into the producer cell by transfection, viral transduction, electroporation, extrusion, sonication, cell fusion, or other methods that are known to the skilled in the art.
- the multiple exogenous biologically active molecules are introduced to the producer cell by transfection.
- the multiple exogenous biologically active molecules can be introduced into suitable producer cells using synthetic macromolecules, such as cationic lipids and polymers (Papapetrou et al., Gene Therapy 12: SI 18-S130 (2005)).
- the cationic lipids form complexes with the multiple exogenous biologically active molecules through charge interactions.
- the positively charged complexes bind to the negatively charged cell surface and are taken up by the cell by endocytosis.
- a cationic polymer can be used to transfect producer cells.
- the cationic polymer is polyethylenimine (PEI).
- chemicals such as calcium phosphate, cyclodextrin, or polybrene, can be used to introduce the multiple exogenous biologically active molecules to the producer cells.
- the multiple exogenous biologically active molecules can also be introduced into a producer cell using a physical method such as particle-mediated transfection, "gene gun”, biolistics, or particle bombardment technology (Papapetrou et al., Gene Therapy 12: SI 18- S130 (2005)).
- a reporter gene such as, for example, beta-galactosidase, chloramphenicol acetyltransferase, luciferase, or green fluorescent protein can be used to assess the transfection efficiency of the producer cell.
- the multiple exogenous biologically active molecules are introduced to the producer cell by viral transduction.
- viruses can be used as gene transfer vehicles, including moloney murine leukemia virus (MMLV), adenovirus, adeno-associated virus (AAV), herpes simplex virus (HSV), lentiviruses, and spumaviruses.
- the viral mediated gene transfer vehicles comprise vectors based on DNA viruses, such as adenovirus, adeno-associated virus and herpes virus, as well as retroviral based vectors.
- the multiple exogenous biologically active molecules are introduced to the producer cell by electroporation. Electroporation creates transient pores in the cell membrane, allowing for the introduction of various molecules into the cell.
- DNA and RNA as well as polypeptides and non-polypeptide therapeutic agents can be introduced into the producer cell by electroporation.
- the multiple exogenous biologically active molecules are introduced to the producer cell by microinjection.
- a glass micropipette can be used to inject the multiple exogenous biologically active molecules into the producer cell at the microscopic level.
- the multiple exogenous biologically active molecules are introduced to the producer cell by extrusion.
- the multiple exogenous biologically active molecules are introduced to the producer cell by sonication.
- the producer cell is exposed to high intensity sound waves, causing transient disruption of the cell membrane allowing loading of the multiple exogenous biologically active molecules.
- the multiple exogenous biologically active molecules are introduced to the producer cell by cell fusion. In some aspects, the multiple exogenous biologically active molecules are introduced by electrical cell fusion. In some aspects, polyethylene glycol (PEG) is used to fuse the producer cells. In further aspects, sendai virus is used to fuse the producer cells. [0265] In some aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by hypotonic lysis. In such aspects, the producer cell can be exposed to low ionic strength buffer causing them to burst allowing loading of the one or more moieties. In some aspects, controlled dialysis against a hypotonic solution can be used to swell the producer cell and to create pores in the producer cell membrane. The producer cell is subsequently exposed to conditions that allow resealing of the membrane.
- hypotonic lysis controlled dialysis against a hypotonic solution can be used to swell the producer cell and to create pores in the producer cell membrane. The producer cell is subsequently exposed to conditions that allow resealing
- the multiple exogenous biologically active molecules are introduced to the producer cell by detergent treatment.
- producer cell is treated with a mild detergent which transiently compromises the producer cell membrane by creating pores allowing loading of the multiple exogenous biologically active molecules. After producer cells are loaded, the detergent is washed away thereby re sealing the membrane.
- the multiple exogenous biologically active molecules are introduced to the producer cell by receptor mediated endocytosis.
- producer cells have a surface receptor which, upon binding of the multiple exogenous biologically active molecules, induces internalization of the receptor and the associated molecules.
- the multiple exogenous biologically active molecules are introduced to the producer cell by fdtration.
- the producer cells and the multiple exogenous biologically active molecules can be forced through a filter of pore size smaller than the producer cell causing transient disruption of the producer cell membrane and allowing the multiple exogenous biologically active molecules to enter the producer cell.
- the producer cell is subjected to several freeze thaw cycles, resulting in cell membrane disruption allowing loading of the multiple exogenous biologically active molecules.
- a method of producing an exosome comprises modifying the isolated exosome by directly introducing the multiple exogenous biologically active molecules into the EVs.
- the multiple exogenous biologically active molecules comprise a therapeutic molecule (e.g., an antigen), adjuvant, immune modulator, targeting moieties, or combinations thereof.
- an isolated exosome can be further modified by directly introducing a scaffold moiety disclosed herein (e.g., Scaffold X or Scaffold Y) using any of the methods disclosed herein for introducing the multiple exogenous biologically active molecules into the EV, e.g., exosome.
- the multiple exogenous biologically active molecules are introduced to the exosome by transfection.
- the multiple exogenous biologically active molecules can be introduced into the EV using synthetic macromolecules such as cationic lipids and polymers (Papapetrou et al., Gene Therapy 12: S118-S130 (2005)).
- chemicals such as calcium phosphate, cyclodextrin, or polybrene, can be used to introduce the multiple exogenous biologically active molecules to the EV.
- the multiple exogenous biologically active molecules are introduced to the EV by electroporation.
- exosomes are exposed to an electrical field which causes transient holes in the EV membrane, allowing loading of the multiple exogenous biologically active molecules.
- the multiple exogenous biologically active molecules are introduced to the EV by microinjection.
- a glass micropipette can be used to inject the multiple exogenous biologically active molecules directly into the EV at the microscopic level.
- the multiple exogenous biologically active molecules are introduced to the EV by extrusion.
- the multiple exogenous biologically active molecules are introduced to the EV by sonication.
- EVs are exposed to high intensity sound waves, causing transient disruption of the EV membrane allowing loading of the multiple exogenous biologically active molecules.
- multiple exogenous biologically active molecules can be conjugated to the surface of the EV. Conjugation can be achieved chemically or enzymatically, by methods known in the art.
- the EV comprises multiple (e.g., two or more) exogenous biologically active molecules that are chemically conjugated. Chemical conjugation can be accomplished by covalent bonding of the multiple exogenous biologically active molecules to another molecule, with or without use of a linker.
- polypeptides are conjugated to the EV.
- non-polypeptides such as lipids, carbohydrates, nucleic acids, and small molecules, are conjugated to the EV.
- the multiple exogenous biologically active molecules are introduced to the EV by hypotonic lysis.
- the EVs can be exposed to low ionic strength buffer causing them to burst allowing loading of the multiple exogenous biologically active molecules.
- controlled dialysis against a hypotonic solution can be used to swell the EV and to create pores in the EV membrane. The EV is subsequently exposed to conditions that allow resealing of the membrane.
- the multiple exogenous biologically active molecules are introduced to the EV by detergent treatment.
- extracellular vesicles are treated with a mild detergent which transiently compromises the EV membrane by creating pores allowing loading of the multiple exogenous biologically active molecules. After EVs are loaded, the detergent is washed away thereby resealing the membrane.
- the multiple exogenous biologically active molecules are introduced to the EV by receptor mediated endocytosis.
- EVs have a surface receptor which, upon binding of the multiple exogenous biologically active molecules, induces internalization of the receptor and the associated molecules.
- the multiple exogenous biologically active molecules are introduced to the EV by mechanical firing.
- extracellular vesicles can be bombarded with multiple exogenous biologically active molecules attached to a heavy or charged particle such as gold microcarriers.
- the particle can be mechanically or electrically accelerated such that it traverses the EV membrane.
- extracellular vesicles are subjected to several freeze thaw cycles, resulting in EV membrane disruption allowing loading of the multiple exogenous biologically active molecules.
- methods of producing EVs disclosed herein comprises isolating the EV from the producer cells.
- the EVs released by the producer cell into the cell culture medium it is contemplated that all known manners of isolation of EVs are deemed suitable for use herein.
- physical properties of EVs can be employed to separate them from a medium or other source material, including separation on the basis of electrical charge (e.g., electrophoretic separation), size (e.g., filtration, molecular sieving, etc. ), density (e.g. , regular or gradient centrifugation), Svedberg constant (e.g. , sedimentation with or without external force, etc.).
- isolation can be based on one or more biological properties, and include methods that can employ surface markers (e.g., for precipitation, reversible binding to solid phase, FACS separation, specific ligand binding, non-specific ligand binding, affinity purification etc.).
- surface markers e.g., for precipitation, reversible binding to solid phase, FACS separation, specific ligand binding, non-specific ligand binding, affinity purification etc.
- Isolation and enrichment can be done in a general and non-selective manner, typically including serial centrifugation.
- isolation and enrichment can be done in a more specific and selective manner, such as using EV or producer cell-specific surface markers.
- specific surface markers can be used in immunoprecipitation, FACS sorting, affinity purification, and magnetic separation with bead-bound ligands.
- size exclusion chromatography can be utilized to isolate the EVs. Size exclusion chromatography techniques are known in the art. Exemplary, non-limiting techniques are provided herein.
- a void volume fraction is isolated and comprises the EVs of interest.
- the EVs can be further isolated after chromatographic separation by centrifugation techniques (of one or more chromatography fractions), as is generally known in the art.
- density gradient centrifugation can be utilized to further isolate the extracellular vesicles.
- the producer cell-derived EVs can be separated from non-producer cell-derived EVs by immunosorbent capture using an antigen antibody specific for the producer cell.
- the isolation of EVs can involve combinations of methods that include, but are not limited to, differential centrifugation, size -based membrane filtration, immunoprecipitation, FACS sorting, and magnetic separation.
- Present disclosure also provides methods of preventing and/or treating a disease or disorder in a subject in need thereof, comprising administering to the subject an EV (e.g., exosome) comprising a KRAS antigen (or a composition comprising the EV) to the subject.
- an EV e.g., exosome
- the disease or disorder that can treated with the present disclosure comprises a cancer, particularly those associated with a KRAS mutant. Examples of such KRAS mutants are provided elsewhere in the present disclosure.
- the EVs (e.g., exosomes) described herein are capable of treating all cancers associated with a KRAS mutation.
- EVs e.g., exosomes
- EVs can be readily modified to comprise a KRAS antigen derived from the KRAS mutant protein associated with a particular cancer.
- such EVs e.g., exosomes
- the cancer is associated with a solid tumor. In some aspects, the cancer is associated with a liquid tumor.
- solid tumor refers to an abnormal mass of tissue that does not contain cysts or liquid areas, and generally occur in the bones, muscles, and organs.
- liquid tumor refers to tumors that occur in body fluids (e.g., blood and bone marrow).
- Non-limiting examples of cancers that can be treated with the present disclosure include a colorectal cancer, lung cancer (e.g., non-small cell lung cancer (NSCLC)), pancreatic cancer (e.g., pancreatic ductal adenocarcinoma), leukemia, uterine cancer, ovarian cancer, bladder cancer, bile duct cancer, gastric cancer, stomach cancer, testicular cancer, esophageal cancer, cholangiocarcinoma, cervical cancer, acute myeloid leukemia (AML), diffuse large B-cell lymphoma (DLBC), sarcoma, melanoma, glioma (e.g., low-grade glioma, e.g., glioblastoma), mesothelioma, liver cancer, breast cancer (e.g., breast invasive carcinoma), renal carcinoma (e.g., papillary renal cell carcinoma (pRCC), and chromophobe renal cell carcinoma), head
- EVs e.g., exosome
- the cancer being treated is characterized by infiltration of leukocytes (T- cells, B-cells, macrophages, dendritic cells, monocytes) into the tumor microenvironment, or so-called "hot tumors” or "inflammatory tumors".
- the cancer being treated is characterized by low levels or undetectable levels of leukocyte infiltration into the tumor microenvironment, or so-called “cold tumors” or “non-inflammatory tumors” .
- an EV is administered in an amount and for a time sufficient to convert a "cold tumor” into a "hot tumor", i.e. , said administering results in the infiltration of leukocytes (such as T-cells) into the tumor microenvironment.
- cancer comprises bladder cancer, cervical cancer, renal cell cancer, testicular cancer, colorectal cancer, lung cancer, head and neck cancer, and ovarian, lymphoma, liver cancer, glioblastoma, melanoma, myeloma, leukemia, pancreatic cancers, or combinations thereof.
- distal tumor or “distant tumor” refers to a tumor that has spread from the original (or primary) tumor to distant organs or distant tissues, e.g. , lymph nodes.
- the EVs of the disclosure treats a tumor after the metastatic spread.
- administering an EV, e.g., exosome, disclosed herein e.g., comprising a
- KRAS antigen, adjuvant, and T helper peptide inhibits and/or reduces growth of a tumor in a subject.
- the growth of a tumor e.g. , tumor volume or weight
- the growth of a tumor is reduced by at least about 5%, at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, or about 100% compared to a reference (e.g., tumor volume in a corresponding subject after administration of an EV, e.g., exosome, without the ASO).
- EVs e.g., exosomes
- fibrosis that can be treated include liver fibrosis (NASH), cirrhosis, pulmonary fibrosis, cystic fibrosis, chronic ulcerative colitis/IBD, bladder fibrosis, kidney fibrosis, CAPS (Muckle-Wells syndrome), atrial fibrosis, endomyocardial fibrosis, old myocardial infarction, glial scar, arterial stiffness, arthrofibrosis, Crohn's disease, Dupuytren's contracture, keloid fibrosis, mediastinal fibrosis, myelofibrosis, Peyronie's disease, nephrogenic systemic fibrosis, progressive massive fibrosis, retroperitoneal fibrosis, scleroderma/systemic sclerosis, adhesive capsulitis, neurofibromatosis
- NASH liver fibrosis
- pulmonary fibrosis pulmonary fibrosis
- EVs e.g., exosomes
- the EVs e.g., exosomes
- the EVs are administered intravenously to the circulatory system of the subject.
- the EVs are infused in suitable liquid and administered into a vein of the subject.
- the EVs are administered intra-arterially to the circulatory system of the subject.
- the EVs e.g., exosomes
- the EVs are infused in suitable liquid and administered into an artery of the subject.
- the EVs are administered to the subject by intranasal administration.
- the EVs e.g., exosomes
- the EVs e.g., exosomes
- intranasal administration can allow for the effective delivery of an EV (e.g., exosome) disclosed herein to the gastrointestinal tissues.
- Such EVs (e.g., exosomes) delivered to the gastrointestinal tissues could be useful in providing protection against various gut- associated pathogens.
- the EVs are administered to the subject by intraperitoneal administration.
- the EVs e.g., exosomes
- the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to the lymphatics.
- the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to the thymus, spleen, and/or bone marrow.
- the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to one or more lymph nodes. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to one or more of the cervical lymph node, the inguinal lymph node, the mediastinal lymph node, or the sternal lymph node. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to the pancreas.
- EVs e.g., exosomes
- parenteral topical, oral, subcutaneous, intradermal, transdermal, rectal, intraperitoneal, intramuscular, sublingual, or combinations thereof.
- EVs e.g., exosomes
- a subject in combination with one or more additional agents, e.g., adjuvants.
- the one or more additional agents and the EVs e.g., exosomes
- the one or more additional agents and the EVs are administered sequentially.
- the EVs e.g., exosomes
- the EVs are administered to the subject prior to administering the one or more additional agents.
- the EVs e.g., exosome
- agents refers to any agents that can be used in combination with the present EVs, e.g., in treating an infectious disease or disorder disclosed herein).
- the one or more additional agents that can be used in combination with the EVs (e.g., exosomes) of the present disclosure include a payload (e.g., antigen, adjuvant, and/or immune modulator) which is not expressed in an EV (e.g., exosome).
- a treatment method disclosed herein can comprise administering to a subject in need thereof (i) an antigen-expressing EV (e.g., exosome) and (ii) an antigen that is the same or different from that expressed in the EV (e.g., soluble antigen).
- an antigen-expressing EV e.g., exosome
- an antigen that is the same or different from that expressed in the EV e.g., soluble antigen
- a subject that can be treated with the present disclosure is a human.
- a subject is a non-human mammal (e.g., non-human primates, dogs, cats, guinea pigs, rabbits, rats, mice, horses, cattle, chickens, birds, and bears).
- the EVs e.g., exosomes
- the practice of the present disclosure will employ, unless otherwise indicated, conventional techniques of cell biology, cell culture, molecular biology, transgenic biology, microbiology, recombinant DNA, and immunology, which are within the skill of the art.
- Example 1 Construction ofEVs (e.g., Exosomes) Comprising a KRAS Antigen
- an engineered EV comprising a KRAS antigen
- the KRAS antigen will be linked to either the external surface and/or the luminal surface of the EVs (e.g., exosome) using the various methods disclosed herein (e.g., scaffold moieties, e.g., PTGFRN or BASP1).
- multiple KRAS antigens will be added to the EVs, e.g., either as separate KRAS antigens or as a peptide concatemer separated by a linker.
- the EVs e.g., exosomes
- the EVs will further comprise an adjuvant, T helper peptide, and/or any of the additional moieties of interest described herein (e.g., targeting moiety and an immune modulator).
- the EVs e.g., exosomes
- the EVs will be tested in an animal model to assess the ability of the vaccines to prevent and/or treat a disease or disorder.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Public Health (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Cell Biology (AREA)
- Immunology (AREA)
- Mycology (AREA)
- Oncology (AREA)
- Microbiology (AREA)
- Virology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Biophysics (AREA)
- General Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Botany (AREA)
- Zoology (AREA)
- Hematology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
The present disclosure relates to extracellular vesicles (EVs), e.g., exosomes, comprising a KRAS antigen. In some aspects, the EVs further comprise an adjuvant, T helper peptide, targeting moiety, and/or immune modulator. Also provided herein are methods for producing such EVs and using such EVs to treat a disease or disorder associated with KRAS mutant proteins.
Description
EXTRACELLULAR VESICLES COMPRISING KRAS ANTIGENS AND USES THEREOF
CROSS-REFERENCE TO RELATED APPLICATION
[0001] This PCT application claims the priority benefit of U.S. Provisional Application No. 63/082,433, filed on September 23, 2020, which is herein incorporated by reference in its entirety.
REFERENCE TO SEQUENCE LISTING SUBMITTED ELECTRONICALLY VIA EFS-WEB
[0002] The content of the electronically submitted sequence listing (Name: 4000_121PC01_Seqlisting_ST25.txt, Size: 650,900 bytes; and Date of Creation: September 23, 2021) submitted in this application is incorporated herein by reference in its entirety.
FIELD OF DISCLOSURE
[0003] The present disclosure relates to engineered extracellular vesicles (EVs) (e.g., exosomes) comprising a KRAS antigen, and the use of such EVs to treat a disease or disorder, including cancer. The present disclosure also relates to methods of producing such EVs (e.g., exosomes).
BACKGROUND
[0004] Kirsten rat sarcoma viral oncogene homology (KRAS) is a member of a superfamily of guanosine-5-triphosphatase (GTPase) proteins that also includes NRAS and HRAS. KRAS mutations have been implicated in many types of cancers, including more than 90% of pancreatic cancers, 35-45% of colorectal cancers, and approximately 25% of lung cancers. Zeitouni, D., et al., Cancers 8(4): 45 (2016); Tan, C., et al., World J Gastroenterol 18(37): 5171-5180 (2012); and Roman, M., et al., Molecular Cancer 17:33 (2018). To date, there are few therapeutic options for most KRAS mutant cancers beyond radiation and non-specific cytotoxic chemotherapies. Accordingly, patients with KRAS mutant cancer often have very poor prognosis. Accordingly, there is a need for new and more effective treatment options for cancers associated with a KRAS mutation.
SUMMARY OF DISCLOSURE
[0005] Provided herein is a composition comprising (i) an isolated extracellular vesicle (EV) comprising a KRAS antigen, and (ii) an adjuvant. In some aspects, the composition further comprises a T helper peptide.
[0006] In some aspects, the KRAS antigen comprises an amino acid substitution selected from G12D, G12C, G12V, G13D, G12A, G12R, G12S, G13C, G13A, GBR, G13S, G13V, Q61H, Q61L, Q61R, Q61K, A18D, K117N, or any combination thereof corresponding to SEQ ID NO: 381. In certain aspects, the amino acid substitution of the KRAS antigen is selected from G12D, G12C, G12V, G13D, and any combination thereof corresponding to SEQ ID NO: 381.
[0007] In some aspects, the KRAS antigen comprises a natural peptide, synthetic peptide, or both. In some aspects, the KRAS antigen is less than about 50 amino acids in length, less than about 45 amino acids in length, less than about 40 amino acids in length, less than about 35 amino acids in length, less than about 30 amino acids in length, less than about 25 amino acids in length, less than about 20 amino acids in length, less than about 15 amino acids in length, or less than about 10 amino acids in length. In certain aspects, the KRAS antigen is between about 12 to about 20 amino acids in length.
[0008] In some aspects, the KRAS antigen comprises a concatemer of multiple KRAS antigens. In certain aspects, the multiple KRAS antigens are separated by a spacer. In certain aspects, the spacer comprises the amino acid sequence AAY, GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), or both. [0009] In some aspects, the KRAS antigen comprises or consists of an epitope of amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids in length. In certain aspects, the KRAS antigen comprises or consists of an epitope of amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length. In some aspects, the KRAS antigen comprises or consists of an epitope of amino acids 97 to 137
of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least 37 amino acids, at least 38 amino acids, at least 39 amino acids, or all 40 amino acids in length.
[0010] In some aspects, the KRAS antigen is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof.
[0011] In some aspects, T helper peptide is associated with the EV. In some aspects, the T helper peptide is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In certain aspects, the T helper peptide is not associated with the EV.
[0012] In some aspects, the T helper peptide comprises a universal T helper peptide . In some aspects, the universal T helper peptide comprises a PADRE, tetanus toxin, diphtheria toxin, HBV peptide, measles peptide, or any combination thereof. In some aspects, the tetanus toxin is a tetanus toxin P2, tetanus toxin P30, or both. In certain aspects, the diphtheria toxin is CRM-197. In some aspects, the HBV peptide is HbsAg. In some aspects, the T helper peptide is a KRAS peptide comprising an epitope for a CD4+ T cell ("KRAS CD4+ T cell peptide"). In some aspects, the T helper peptide comprises a concatemer of multiple T helper peptides. In certain aspects, the multiple T helper peptides are separated by a spacer.
[0013] In some aspects, the adjuvant is associated with the EV. In some aspects, the adjuvant is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In some aspects, the adjuvant is not associated with the EV.
[0014] In some aspects, the adjuvant comprises a STING agonist, TLR agonist, or both. In certain aspects, the STING agonist comprises a cyclic dinucleotide STING agonist or a non-cyclic dinucleotide STING agonist. In some aspects, the TLR agonist comprises a TLR2 agonist (e.g. , lipoteichoic acid, atypical LPS, MALP-2 and MALP-404, OspA, porin, LcrV, lipomannan, GPI anchor, lysophosphatidylserine, lipophosphoglycan (LPG), glycophosphatidylinositol (GPI), zymosan, hsp60, gH/gL glycoprotein, hemagglutinin), a TLR3 agonist (e.g., double-stranded RNA, e.g., poly(I:C), ampligen, hiltonol, polyA:U), a TLR4 agonist (e.g. , lipopolysaccharides (LPS), lipoteichoic acid, [3-defensin 2, fibronectin EDA, HMGB 1, snapin, tenascin C, MPLA), a TLR5 agonist (e.g. , flagellin), a TLR6 agonist, a TLR7/8 agonist (e.g. , single-
stranded RNA, , Poly GIO, Poly G3, Resiquimod, Imiquimod, 3M-052), aTLR9 agonist (e.g., unmethylated CpG DNA), or any combination thereof.
[0015] In some aspects, the composition comprising an EV, which comprises a KRAS antigen, further comprises one or more additional moieties selected from an immune modulator, targeting moiety, or any combination thereof. In certain aspects, the immune modulator and/or targeting moiety are associated with the EV. In some aspects, the immune modulator, targeting moiety, and/or anti -phagocytic moiety are: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In some aspects, the immune modulator and/or targeting moiety are not associated with the EV.
[0016] In some aspects, the immune modulator comprises (i) an inhibitor for a negative checkpoint regulator or an inhibitor for a binding partner of a negative checkpoint regulator (e.g., anti-CTLA4 antibody); (ii) an activator for a positive co-stimulatory molecule or an activator for a binding partner of a positive co-stimulatory molecule (e.g., CD40L); (iii) a cytokine or a binding partner of a cytokine; (iv) a protein that supports intracellular interactions required for germinal center responses; (v) a polynucleotide; or (vi) any combination thereof.
[0017] In some aspects, the targeting moiety binds to a marker expressed on an immune cell (e.g., dendritic cell, T cell, B cell, or any combination thereof). In some aspects, the marker expressed on an immune cell comprises a C-type lectin domain family 9 member A (Clec9a) protein, a dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN), CD207, CD40, Clec6, dendritic cell immunoreceptor (DCIR), DEC-205, lectin-like oxidized low-density lipoprotein receptor- 1 (LOX-1), MARCO, Clecl2a, DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin-1, macrophage mannose receptor (MMR), BDCA-1 (CD303, Clec4c), Dectin-2, Bst-2 (CD317), CD3, CD19, CDla, CDl lc, Langeran, CD103, XCR1, BDCA-3 (CD141), or any combination thereof.
[0018] In some aspects, the targeting moiety binds to a marker expressed in a lymphoid tissue of a subject suffering from a cancer. In some aspects, the lymphoid tissue comprises a tumor draining lymph node, sentinel lymph node, tumor tertiary lymph node, or any combination thereof. In certain aspects, the targeting moiety comprises anti -fibronectin EDA & EDB domain antibodies; anti-ICAM-1; anti-CD20; anti-DC-LAMP; or any combination thereof.
[0019] In some aspects, the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties is linked to the exterior surface and/or luminal surface of the EV by an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), or any combination thereof. In some aspects, the anchoring moiety comprises a cholesterol, fatty acid (e.g., palmitate), tocopherol (e.g., vitamin E), or any combination thereof. In certain aspects, the chemical conjugation comprises a maleimide moiety, copper-free, biorthogonal click chemistry (e.g., azide/strained alkyne (DIFO, DBCO, BCN)), metal- catalyzed click chemistry (e.g., CUAAC, RUAAC), or any combination thereof.
[0020] In some aspects, a composition disclosed herein (i.e., comprising an EV, which comprises a KRAS antigen) further comprises one or more scaffold moieties. In certain aspects, (i) the KRAS antigen is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; (ii) the T helper peptide is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; (iii) the adjuvant is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; (iv) the one or more additional moieties are linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; or (v) any combination thereof. In some aspects, (i) the KRAS antigen is linked to the one or more scaffold moieties by a linker; (ii) the T helper peptide is linked to the one or more scaffold moieties by a linker; (iii) the adjuvant is linked to the one or more scaffold moieties by a linker; (iv) the one or more additional moieties are linked to the one or more scaffold moieties by a linker; or (v) any combination thereof.
[0021] In some aspects, the linker is a polypeptide. In some aspects, the linker is a non-polypeptide moiety. In some aspects, the linker comprises a maleimide moiety. In some aspects, the linker comprises a cholesterol moiety. In some aspects, the linker comprises an ALFA-tag moiety.
[0022] In some aspects, the one or more scaffold moieties comprise a Scaffold X, Scaffold Y, or both. In certain aspects, the Scaffold X comprises a prostaglandin F2 receptor negative regulator (the PTGFRN protein); basigin (the BSG protein); immunoglobulin superfamily member 2 (the IGSF2 protein); immunoglobulin superfamily member 3 (the IGSF3 protein); immunoglobulin superfamily member 8 (the IGSF8 protein); integrin beta-1 (the ITGB1 protein); integrin alpha-4 (the ITGA4 protein); 4F2 cell-surface antigen heavy chain (the SLC3A2 protein); a class of ATP transporter proteins (the ATP1A1, ATP1A2, ATP1A3, ATP1A4, ATP1B3, ATP2B1, ATP2B2, ATP2B3, ATP2B4 proteins), or any combination thereof. In some aspects, the Scaffold X is PTGFRN or a fragment thereof. In some aspects, the Scaffold Y comprises myristoylated alanine rich Protein Kinase C substrate (the MARCKS protein); myristoylated alanine rich Protein Kinase C substrate like 1 (the MARCKSL1 protein); brain acid soluble protein 1 (the BASP 1 protein), and any combination thereof. In some aspects, the Scaffold Y is BASP 1 or a fragment thereof.
[0023] In some aspects, the EV is an exosome.
[0024] Also disclosed herein is a pharmaceutical composition comprising any of the EVs described herein, and a pharmaceutically acceptable carrier. Also disclosed herein is a cell that produces any of the EVs of the present disclosure. Provided herein is a kit comprising the EV of the present disclosure. Present disclosure further provides an EV -drug conjugate comprising any of the EVs described in the present disclosure.
[0025] Present disclosure provides a method of making EVs comprising culturing a cell describe herein under a suitable condition and obtaining the EV. Provided herein is method of making EVs comprising loading an EV that has been isolated from a producer cell with the KRAS antigen, T helper
peptide, adjuvant, and/or one or more additional moieties disclosed herein. Present disclosure additionally provides methods of making a cancer vaccine comprising loading an EV with the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties disclosed herein. In some aspects, the EV is isolated from a producer cell prior to the loading of the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties.
[0026] Provided herein is a vaccine produced by a method disclosed herein.
[0027] Provided herein is a method of inducing an immune response in a subject in need thereof, comprising administering to the subject any one of the compositions described herein.
[0028] Also provided herein is a method of inducing a vaccine in a subject afflicted with a cancer, comprising administering the vaccine of the present disclosure. Also provided herein is a method of preventing or treating a cancer in a subject in need thereof, comprising administering the composition or the vaccine described herein to the subject. In some aspects, the cancer comprises a colorectal cancer, lung cancer (e.g., non-small cell lung cancer (NSCLC)), pancreatic cancer (e.g., pancreatic ductal adenocarcinoma), leukemia, uterine cancer, ovarian cancer, bladder cancer, bile duct cancer, gastric cancer, stomach cancer, testicular cancer, esophageal cancer, cholangiocarcinoma, cervical cancer, acute myeloid leukemia (AML), diffuse large B-cell lymphoma (DLBC), sarcoma, melanoma, glioma (e.g., low-grade glioma, e.g., glioblastoma), mesothelioma, liver cancer, breast cancer (e.g., breast invasive carcinoma), renal carcinoma (e.g., papillary renal cell carcinoma (pRCC), and chromophobe renal cell carcinoma), head and neck cancer, prostate cancer, adenoid cystic carcinoma (ACC), thymoma cancer, thyroid cancer, clear cell renal cell carcinoma (CCRCC), neuroendocrine neoplasm (e.g., pheochromocytoma/paraganglioma), uveal melanoma, multiple myeloma, or any combination thereof.
BRIEF DESCRIPTION OF FIGURES
[0029] FIG. 1 shows an exemplary EV (e.g., exosome) comprising (i) a KRAS antigen, (ii) a T helper peptide, (iii) an adjuvant, and (iv) a targeting moiety, one or more antigens.
DETAILED DESCRIPTION OF DISCLOSURE
[0030] Certain aspects of the present disclosure are directed to an engineered extracellular vesicle (EV) (e.g., exosome) comprising a KRAS antigen.
[0031] The present disclosure is directed to an engineered EV, e.g., exosome, comprising one or more payloads, wherein the one or more payloads can improve at least one property (e.g., such as those disclosed herein) of the EV, and uses thereof. For example, in some aspects, the EVs (e.g., exosomes) disclosed herein are capable of targeting an immune cell (e.g., macrophage or dendritic cell) within the central nervous system of a subject. In some aspects, the one or more payloads that can be expressed in an
EV (e.g., exosome) disclosed herein comprise an antigen (e.g., associated with a neurological disorder disclosed herein), an adjuvant, an immune modulator, or combinations thereof. In some aspects, the one or more payloads can be attached (or linked) to one or more scaffold moieties on the surface of EVs, e.g., exosomes, or on the luminal surface of EVs, e.g., exosomes. In some aspects, the EVs (e.g., exosomes) can further comprise a targeting moiety, which can also be attached (or linked) to one or more of the scaffold moieties disclosed herein. Non-limiting examples of the various aspects are shown in the present disclosure.
I. Definitions
[0032] In order that the present description can be more readily understood, certain terms are first defined. Additional definitions are set forth throughout the detailed description.
[0033] It is to be noted that the term "a" or "an" entity refers to one or more of that entity; for example, "a nucleotide sequence," is understood to represent one or more nucleotide sequences. As such, the terms "a" (or "an"), "one or more," and "at least one" can be used interchangeably herein.
[0034] Furthermore, "and/or" where used herein is to be taken as specific disclosure of each of the two specified features or components with or without the other. Thus, the term "and/or" as used in a phrase such as "A and/or B" herein is intended to include "A and B," "A or B," "A" (alone), and "B" (alone). Likewise, the term "and/or" as used in a phrase such as "A, B, and/or C" is intended to encompass each of the following aspects: A, B, and C; A, B, or C; A or C; A or B; B or C; A and C; A and B; B and C; A (alone); B (alone); and C (alone).
[0035] It is understood that wherever aspects are described herein with the language "comprising," otherwise analogous aspects described in terms of "consisting of" and/or "consisting essentially of" are also provided.
[0036] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure is related. For example, the Concise Dictionary of Biomedicine and Molecular Biology, Juo, Pei-Show, 2nd ed., 2002, CRC Press; The Dictionary of Cell and Molecular Biology, 3rd ed., 1999, Academic Press; and the Oxford Dictionary Of Biochemistry And Molecular Biology, Revised, 2000, Oxford University Press, provide one of skill with a general dictionary of many of the terms used in this disclosure.
[0037] Units, prefixes, and symbols are denoted in their Systeme International de Unites (SI) accepted form. Numeric ranges are inclusive of the numbers defining the range. Unless otherwise indicated, nucleotide sequences are written left to right in 5' to 3' orientation. Amino acid sequences are written left to right in amino to carboxy orientation. The headings provided herein are not limitations of the various aspects of the disclosure, which can be had by reference to the specification as a whole. Accordingly, the terms defined immediately below are more fully defined by reference to the specification in its entirety.
[0038] The term "about" is used herein to mean approximately, roughly, around, or in the regions of. When the term "about" is used in conjunction with a numerical range, it modifies that range by extending the boundaries above and below the numerical values set forth. In general, the term "about" can modify a numerical value above and below the stated value by a variance of, e.g., 10 percent, up or down (higher or lower).
[0039] As used herein, the term "extracellular vesicle" or "EV" refers to a cell-derived vesicle comprising a membrane that encloses an internal space. Extracellular vesicles comprise all membranebound vesicles (e.g., exosomes, nanovesicles) that have a smaller diameter than the cell from which they are derived. In some aspects, extracellular vesicles range in diameter from 20 nm to 1000 nm, and can comprise various macromolecular payload either within the internal space (i.e., lumen), displayed on the external surface of the extracellular vesicle, and/or spanning the membrane. In some aspects, the payload can comprise nucleic acids, proteins, carbohydrates, lipids, small molecules, and/or combinations thereof. In certain aspects, an extracellular vehicle comprises a scaffold moiety. By way of example and without limitation, extracellular vesicles include apoptotic bodies, fragments of cells, vesicles derived from cells by direct or indirect manipulation (e.g., by serial extrusion or treatment with alkaline solutions), vesiculated organelles, and vesicles produced by living cells (e.g., by direct plasma membrane budding or fusion of the late endosome with the plasma membrane). Extracellular vesicles can be derived from a living or dead organism, explanted tissues or organs, prokaryotic or eukaryotic cells, and/or cultured cells. In some aspects, the extracellular vesicles are produced by cells that express one or more transgene products.
[0040] As used herein, the term "exosome" refers to an extracellular vesicle with a diameter between 20-300 nm (e.g., between 40-200 nm). Exosomes comprise a membrane that encloses an internal space (i.e., lumen), and, in some aspects, can be generated from a cell (e.g., producer cell) by direct plasma membrane budding or by fusion of the late endosome or multi -vesicular body with the plasma membrane. In certain aspects, an exosome comprises a scaffold moiety. As described infra, exosome can be derived from a producer cell, and isolated from the producer cell based on its size, density, biochemical parameters, or a combination thereof. In some aspects, the EVs, e.g., exosomes, of the present disclosure are produced by cells that express one or more transgene products.
[0041] As used herein, the term "nanovesicle" refers to an extracellular vesicle with a diameter between 20-250 nm (e.g., between 30-150 nm) and is generated from a cell (e.g., producer cell) by direct or indirect manipulation such that the nanovesicle would not be produced by the cell without the manipulation. Appropriate manipulations of the cell to produce the nanovesicles include but are not limited to serial extrusion, treatment with alkaline solutions, sonication, or combinations thereof. In some aspects, production of nanovesicles can result in the destruction of the producer cell. In some aspects, population of nanovesicles described herein are substantially free of vesicles that are derived from cells by way of direct budding from the plasma membrane or fusion of the late endosome with the plasma membrane. In certain
aspects, a nanovesicle comprises a scaffold moiety. Nanovesicles, once derived from a producer cell, can be isolated from the producer cell based on its size, density, biochemical parameters, or a combination thereof.
[0042] As used herein the term "surface-engineered EVs, e.g., exosomes" (e.g., Scaffold X- engineered EVs, e.g., exosomes) refers to an EV, e.g., exosome, with the membrane or the surface of the EV, e.g., exosome, modified in its composition so that the surface of the engineered EV, e.g., exosome, is different from that of the EV, e.g., exosome, prior to the modification or of the naturally occurring EV, e.g., exosome. The engineering can be on the surface of the EV, e.g., exosome, or in the membrane of the EV, e.g., exosome, so that the surface of the EV, e.g., exosome, is changed. For example, the membrane is modified in its composition of a protein, a lipid, a small molecule, a carbohydrate, etc. The composition can be changed by a chemical, a physical, or a biological method or by being produced from a cell previously or concurrently modified by a chemical, a physical, or a biological method. Specifically, the composition can be changed by a genetic engineering or by being produced from a cell previously modified by genetic engineering. In some aspects, a surface -engineered EV, e.g., exosome, comprises an exogenous protein (i.e., a protein that the EV, e.g. , exosome, does not naturally express) or a fragment or variant thereof that can be exposed to the surface of the EV, e.g., exosome, or can be an anchoring point (attachment) for a moiety exposed on the surface of the EV, e.g., exosome. In some aspects, a surface-engineered EV, e.g., exosome, comprises a higher expression (e.g., higher number) of a natural exosome protein (e.g., Scaffold X) or a fragment or variant thereof that can be exposed to the surface of the EV, e.g., exosome, or can be an anchoring point (attachment) for a moiety exposed on the surface of the EV, e.g., exosome.
[0043] As used herein the term "lumen-engineered exosome" (e.g., Scaffold Y-engineered exosome) refers to an EV, e.g., exosome, with the membrane or the lumen of the EV, e.g., exosome, modified in its composition so that the lumen of the engineered EV, e.g., exosome, is different from that of the EV, e.g., exosome, prior to the modification or of the naturally occurring EV, e.g., exosome. The engineering can be directly in the lumen or in the membrane of the EV, e.g., exosome so that the lumen of the EV, e.g., exosome is changed. For example, the membrane is modified in its composition of a protein, a lipid, a small molecule, a carbohydrate, etc. so that the lumen of the EV, e.g., exosome is modified. The composition can be changed by a chemical, a physical, or a biological method or by being produced from a cell previously modified by a chemical, a physical, or a biological method. Specifically, the composition can be changed by a genetic engineering or by being produced from a cell previously modified by genetic engineering. In some aspects, a lumen-engineered exosome comprises an exogenous protein (i.e., a protein that the EV, e.g., exosome does not naturally express) or a fragment or variant thereof that can be exposed in the lumen of the EV, e.g., exosome or can be an anchoring point (attachment) for a moiety exposed on the inner layer ofthe EV, e.g., exosome. In some aspects, a lumen-engineered EV, e.g., exosome, comprises a higher expression of a natural exosome protein (e.g., Scaffold X or Scaffold Y) or a fragment or variant
thereof that can be exposed to the lumen of the exosome or can be an anchoring point (attachment) for a moiety exposed in the lumen of the exosome.
[0044] The term "modified," when used in the context of EVs, e.g., exosomes described herein, refers to an alteration or engineering of an EV, e.g. , exosome and/or its producer cell, such that the modified EV, e.g., exosome is different from a naturally-occurring EV, e.g., exosome. In some aspects, a modified EV, e.g., exosome described herein comprises a membrane that differs in composition of a protein, a lipid, a small molecular, a carbohydrate, etc. compared to the membrane of a naturally-occurring EV, e.g., exosome (e.g., membrane comprises higher density or number of natural exosome proteins and/or membrane comprises proteins that are not naturally found in exosomes (e.g., antigen, adjuvant, and/or immune modulator). In certain aspects, such modifications to the membrane changes the exterior surface of the EV, e.g., exosome (e.g., surface-engineered EVs, e.g., exosomes described herein). In certain aspects, such modifications to the membrane changes the lumen of the EV, e.g., exosome (e.g., lumen-engineered EVs, e.g., exosomes described herein).
[0045] As used herein, the term "scaffold moiety" refers to a molecule that can be used to anchor a payload or any other compound of interest (e.g., antigen, adjuvant, and/or immune modulator) to the EV, e.g., exosome either on the luminal surface or on the exterior surface of the EV, e.g., exosome. In certain aspects, a scaffold moiety comprises a synthetic molecule. In some aspects, a scaffold moiety comprises a non-polypeptide moiety. In some aspects, a scaffold moiety comprises a lipid, carbohydrate, or protein that naturally exists in the EV, e.g. , exosome. In some aspects, a scaffold moiety comprises a lipid, carbohydrate, or protein that does not naturally exist in the EV, e.g., exosome. In certain aspects, a scaffold moiety is Scaffold X. In some aspects, a scaffold moiety is Scaffold Y. In further aspects, a scaffold moiety comprises both Scaffold X and Scaffold Y. Non-limiting examples of other scaffold moieties that can be used with the present disclosure include: aminopeptidase N (CD13); Neprilysin, AKA membrane metalloendopeptidase (MME); ectonucleotide pyrophosphatase/phosphodiesterase family member 1 (ENPP1); Neuropilin-1 (NRP1); CD9, CD63, CD81, PDGFR, GPI anchor proteins, lactadherin, LAMP2, and LAMP2B.
[0046] As used herein, the term "Scaffold X" refers to exosome proteins that have recently been identified on the surface of exosomes. See, e.g., U.S. Pat. No. 10,195,290, which is incorporated herein by reference in its entirety. Non-limiting examples of Scaffold X proteins include: prostaglandin F2 receptor negative regulator ("the PTGFRN protein"); basigin ("the BSG protein"); immunoglobulin superfamily member 2 ("the IGSF2 protein"); immunoglobulin superfamily member 3 ("the IGSF3 protein"); immunoglobulin superfamily member 8 ("the IGSF8 protein"); integrin beta-1 ("the ITGB1 protein); integrin alpha-4 ("the ITGA4 protein"); 4F2 cell-surface antigen heavy chain ("the SLC3A2 protein"); and a class of ATP transporter proteins ("the ATP1A1 protein," "the ATP1A2 protein," "the ATP1A3 protein," "the ATP1A4 protein," "the ATP1B3 protein," "the ATP2B1 protein," "the ATP2B2 protein," "the ATP2B3 protein," "the ATP2B protein"). In some aspects, a Scaffold X protein can be a whole protein or a fragment
thereof (e.g., functional fragment, e.g., the smallest fragment that is capable of anchoring another moiety on the exterior surface or on the luminal surface of the EV, e.g., exosome). In some aspects, a Scaffold X can anchor a moiety (e.g., antigen, adjuvant, and/or immune modulator) to the external surface or the luminal surface of the exosome.
[0047] As used herein, the term "Scaffold Y" refers to exosome proteins that were newly identified within the lumen of exosomes. See, e.g., International Publ. No. WO/2019/099942 (or the U.S. equivalent US 2020/0347112); and WO 2020/101740, each of which is incorporated herein by reference in its entirety. Non-limiting examples of Scaffold Y proteins include: myristoylated alanine rich Protein Kinase C substrate ("the MARCKS protein"); myristoylated alanine rich Protein Kinase C substrate like 1 ("the MARCKSE1 protein"); and brain acid soluble protein 1 ("the BASP1 protein"). In some aspects, a Scaffold Y protein can be a whole protein or a fragment thereof (e.g. , functional fragment, e.g. , the smallest fragment that is capable of anchoring a moiety to the luminal surface of the exosome). In some aspects, a Scaffold Y can anchor a moiety (e.g., antigen, adjuvant, and/or immune modulator) to the luminal surface of the EV, e.g., exosome. [0048] As used herein, the term "fragment" of a protein (e.g., therapeutic protein, Scaffold X, or Scaffold Y) refers to an amino acid sequence of a protein that is shorter than the naturally-occurring sequence, N- and/or C-terminally deleted or any part of the protein deleted in comparison to the naturally occurring protein. As used herein, the term "functional fragment" refers to a protein fragment that retains protein function. Accordingly, in some aspects, a functional fragment of a Scaffold X protein retains the ability to anchor a moiety on the luminal surface or on the exterior surface of the EV, e.g., exosome. Similarly, in certain aspects, a functional fragment of a Scaffold Y protein retains the ability to anchor a moiety on the luminal surface of the EV, e.g., exosome. Whether a fragment is a functional fragment can be assessed by any art known methods to determine the protein content of EVs, e.g., exosomes including Western Blots, FACS analysis and fusions of the fragments with autofluorescent proteins like, e.g., GFP. In certain aspects, a functional fragment of a Scaffold X protein retains at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90% or at least about 100% of the ability, e.g., an ability to anchor a moiety, of the naturally occurring Scaffold X protein. In some aspects, a functional fragment of a Scaffold Y protein retains at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90% or at least about 100% of the ability, e.g., an ability to anchor another molecule, of the naturally occurring Scaffold Y protein.
[0049] As used herein, the term "variant" of a molecule (e.g. , functional molecule, antigen, Scaffold X and/or Scaffold Y) refers to a molecule that shares certain structural and functional identities with another molecule upon comparison by a method known in the art. For example, a variant of a protein can include a substitution, insertion, deletion, frameshift or rearrangement in another protein.
[0050] A "conservative amino acid substitution" is one in which the amino acid residue is replaced with an amino acid residue having a similar side chain. Families of amino acid residues having similar side
chains have been defined in the art, including basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine). Thus, if an amino acid in a polypeptide is replaced with another amino acid from the same side chain family, the substitution is considered to be conservative. In some aspects, a string of amino acids can be conservatively replaced with a structurally similar string that differs in order and/or composition of side chain family members.
[0051] The term "percent sequence identity" or "percent identity" between two polynucleotide or polypeptide sequences refers to the number of identical matched positions shared by the sequences over a comparison window, taking into account additions or deletions (i.e., gaps) that must be introduced for optimal alignment of the two sequences. A matched position is any position where an identical nucleotide or amino acid is presented in both the target and reference sequence. Gaps presented in the target sequence are not counted since gaps are not nucleotides or amino acids. Likewise, gaps presented in the reference sequence are not counted since target sequence nucleotides or amino acids are counted, not nucleotides or amino acids from the reference sequence.
[0052] The percentage of sequence identity is calculated by determining the number of positions at which the identical amino-acid residue or nucleic acid base occurs in both sequences to yield the number of matched positions, dividing the number of matched positions by the total number of positions in the window of comparison and multiplying the result by 100 to yield the percentage of sequence identity. The comparison of sequences and determination of percent sequence identity between two sequences can be accomplished using readily available software both for online use and for download. Suitable software programs are available from various sources, and for alignment of both protein and nucleotide sequences. One suitable program to determine percent sequence identity is bl2seq, part of the BLAST suite of programs available from the U.S. government's National Center for Biotechnology Information BLAST web site (blast.ncbi.nlm.nih.gov). B12seq performs a comparison between two sequences using either the BLASTN or BLASTP algorithm. BLASTN is used to compare nucleic acid sequences, while BLASTP is used to compare amino acid sequences. Other suitable programs are, e.g., Needle, Stretcher, Water, or Matcher, part of the EMBOSS suite of bioinformatics programs and also available from the European Bioinformatics Institute (EBI) at www.ebi.ac.uk/Tools/psa.
[0053] Different regions within a single polynucleotide or polypeptide target sequence that aligns with a polynucleotide or polypeptide reference sequence can each have their own percent sequence identity. It is noted that the percent sequence identity value is rounded to the nearest tenth. For example, 80.11, 80.12, 80.13, and 80.14 are rounded down to 80.1, while 80.15, 80.16, 80.17, 80.18, and 80.19 are rounded up to 80.2. It also is noted that the length value will always be an integer.
[0054] One skilled in the art will appreciate that the generation of a sequence alignment for the calculation of a percent sequence identity is not limited to binary sequence-sequence comparisons exclusively driven by primary sequence data. Sequence alignments can be derived from multiple sequence alignments. One suitable program to generate multiple sequence alignments is ClustalW2, available from www.clustal.org. Another suitable program is MUSCLE, available from www.drive5.com/muscle/. ClustalW2 and MUSCLE are alternatively available, e.g., from the EBE
[0055] It will also be appreciated that sequence alignments can be generated by integrating sequence data with data from heterogeneous sources such as structural data (e.g., crystallographic protein structures), functional data (e.g., location of mutations), or phylogenetic data. A suitable program that integrates heterogeneous data to generate a multiple sequence alignment is T-Coffee, available at worldwideweb.tcoffee.org, and alternatively available, e.g., from the EBE It will also be appreciated that the final alignment used to calculate percent sequence identity can be curated either automatically or manually.
[0056] Using known methods of protein engineering and recombinant DNA technology, variants can be generated to improve or alter the characteristics of the polypeptides. For instance, one or more amino acids can be deleted from the N-terminus or C-terminus of the secreted protein without substantial loss of biological function. Ron et al., J. Biol. Chem. 268: 2984-2988 (1993), incorporated herein by reference in its entirety, reported variant KGF proteins having heparin binding activity even after deleting 3, 8, or 27 amino-terminal amino acid residues. Similarly, interferon gamma exhibited up to ten times higher activity after deleting 8-10 amino acid residues from the carboxy terminus of this protein. (Dobeli et al., J. Biotechnology 7: 199-216 (1988), incorporated herein by reference in its entirety.)
[0057] Moreover, ample evidence demonstrates that variants often retain a biological activity similar to that of the naturally occurring protein. For example, Gayle and coworkers (J. Biol. Chem 268:22105- 22111 (1993), incorporated herein by reference in its entirety) conducted extensive mutational analysis of human cytokine IL- la. They used random mutagenesis to generate over 3,500 individual IL- la mutants that averaged 2.5 amino acid changes per variant over the entire length of the molecule. Multiple mutations were examined at every possible amino acid position. The investigators found that "[m]ost of the molecule could be altered with little effect on either [binding or biological activity]." (See Abstract.) In fact, only 23 unique amino acid sequences, out of more than 3,500 nucleotide sequences examined, produced a protein that significantly differed in activity from wild-type.
[0058] As stated above, polypeptide variants include, e.g., modified polypeptides. Modifications include, e.g., conservative amino acid substitution, acetylation, acylation, ADP-ribosylation, amidation, covalent attachment of flavin, covalent attachment of a heme moiety, covalent attachment of a nucleotide or nucleotide derivative, covalent attachment of a lipid or lipid derivative, covalent attachment of phosphotidylinositol, covalent attachment of bioorthoganal functionalities (e.g., azide, alkyne, trans-
cycloalkyne, tetrazine), cross-linking, cyclization, disulfide bond formation, demethylation, formation of covalent cross-links, formation of cysteine, formation of pyroglutamate, formylation, gammacarboxylation, glycosylation, GPI anchor formation, hydroxylation, iodination, methylation, myristoylation, oxidation, pegylation (Mei et al., Blood 7./ <5:270-79 (2010), which is incorporated herein by reference in its entirety), proteolytic processing, phosphorylation, prenylation, racemization, selenoylation, sulfation, transfer-RNA mediated addition of amino acids to proteins such as arginylation, and ubiquitination. In some aspects, Scaffold X and/or Scaffold Y is modified at any convenient location. [0059] As used herein, when a molecule described herein (e.g., antigen, adjuvant, immune modulator, targeting moiety, affinity ligand, and/or scaffold moiety) is "expressed," "added," or "loaded" in an EV (e.g., exosome), it means that the molecule is present in (e.g., in the lumen) or on (e.g., on the exterior surface and/or luminal surface) the EV. In some aspects, a molecule can be exogenously introduced into a producer cell or directly into an EV, such that the EV expresses the molecule of interest. In some aspects, a molecule of interest can be produced separately from an EV and then conjugated or linked to a moiety present in the EV, such that the EV expresses the molecule. For instance, in some aspects, an antigen (e.g., KRAS antigen) can be fused to an affinity ligand disclosed herein. Then, the antigen-affinity ligand fusion can be linked or conjugated to a scaffold moiety expressed on the surface of an EV via the affinity ligand. Additional disclosure relating to different methods of expressing a molecule of interest in or on an EV (e.g., exosome) is described elsewhere in the present disclosure.
[0060] As used herein the term "linked to," "fused," or "conjugated to" are used interchangeably and refer to a covalent or non-covalent bond formed between a first moiety and a second moiety, e.g., Scaffold X and an antigen (or adjuvant or immune modulator), respectively, e.g., a scaffold moiety expressed in or on the extracellular vesicle and an antigen, e.g., Scaffold X (e.g., a PTGFRN protein), respectively, in the luminal surface of or on the external surface of the extracellular vesicle. In some aspects, a payload disclosed herein (e.g. , antigen, adjuvant, and/or immune modulator) and/or a targeting moiety can be directly linked to the exterior surface and/or the luminal surface of an EV (e.g. , exosome). As used herein, the term "directly linked," "directly fused," or "directly conjugated to" refer to the process of linking (fusing or conjugating) a moiety (e.g., a payload and/or targeting moiety) to the surface of an EV (e.g., exosome) without the use of a scaffold moiety disclosed herein.
[0061] As used herein, the term "fusion protein" or "fusion molecule" (or derivatives thereof) refers to two or more proteins (or molecules) that are linked or conjugated to each other. For instance, in some aspects, a fusion protein that can be expressed in an EV (e.g., exosome) disclosed herein comprises (i) a payload (e.g., antigen, adjuvant, and/or immune modulator) and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y). In some aspects, the payload (e.g., antigen, adjuvant, and/or immune modulator) is linked or conjugated to the scaffold moiety via an affinity ligand (e.g., those described herein). In some aspects, a fusion protein that can be expressed in an EV (e.g., exosome) useful for the present disclosure
comprises (i) a targeting moiety and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y). In some aspects, the targeting moiety is linked or conjugated to the scaffold moiety via an affinity ligand (e.g., those described herein). As described herein, in some aspects, EVs (e.g., exosomes) of the present disclosure can express multiple fusion proteins, wherein a first fusion protein comprises (i) a payload (e.g., antigen, adjuvant, and/or immune modulator) and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y), and wherein a second fusion protein comprises (i) a targeting moiety and (ii) a scaffold moiety (e.g., Scaffold X and/or Scaffold Y)
[0062] The term "encapsulated", or grammatically different forms of the term (e.g., encapsulation, or encapsulating) refers to a status or process of having a first moiety (e.g., antigen, adjuvant, or immune modulator) inside a second moiety (e.g., an EV, e.g., exosome) without chemically or physically linking the two moieties. In some aspects, the term "encapsulated" can be used interchangeably with "in the lumen of". Non-limiting examples of encapsulating a first moiety (e.g., antigen, adjuvant, or immune modulator) into a second moiety (e.g., EVs, e.g., exosomes) are disclosed elsewhere herein.
[0063] As used herein, the term "producer cell" refers to a cell used for generating an EV, e.g., exosome. A producer cell can be a cell cultured in vitro, or a cell in vivo. A producer cell includes, but are not limited to, a cell known to be effective in generating EVs, e.g, exosomes, e.g., HEK293 cells, Chinese hamster ovary (CHO) cells, mesenchymal stem cells (MSCs), BJ human foreskin fibroblast cells, fHDF fibroblast cells, AGE.HN® neuronal precursor cells, CAP® amniocyte cells, adipose mesenchymal stem cells, RPTEC/TERT1 cells. In certain aspects, a producer cell is not a naturally-existing antigen-presenting cell (i.e., has been modified). In some aspects, a producer cell is not a naturally -existing dendritic cell, a naturally-existing B cell, a naturally-existing mast cell, a naturally-existing macrophage, a naturally- existing neutrophil, naturally-existing Kupffer-Browicz cell, cell derived from any of these cells, or any combination thereof. Additional disclosures relating to such producer cells are provided elsewhere in the present disclosure. In some aspects, the EVs, e.g., exosomes useful in the present disclosure do not carry an antigen on MHC class I or class II molecule (i.e., antigen is not presented on MHC class I or class II molecule) exposed on the surface of the EV, e.g., exosome, but instead can carry an antigen in the lumen of the EV, e.g., exosome, or on the surface of the EV, e.g., exosome, by attachment to Scaffold X and/or Scaffold Y.
[0064] As used herein, the terms "isolate," "isolated," and "isolating" or "purify," "purified," and "purifying" as well as "extracted" and "extracting" are used interchangeably and refer to the state of a preparation (e.g. , a plurality of known or unknown amount and/or concentration) of desired EVs, that have undergone one or more processes of purification, e.g., a selection or an enrichment of the desired EV preparation. In some aspects, isolating or purifying as used herein is the process of removing, partially removing (e.g., a fraction) of the EVs from a sample containing producer cells. In some aspects, an isolated EV composition has no detectable undesired activity or, alternatively, the level or amount of the undesired
activity is at or below an acceptable level or amount. In some aspects, an isolated EV composition has an amount and/or concentration of desired EVs at or above an acceptable amount and/or concentration. In some aspects, the isolated EV composition is enriched as compared to the starting material (e.g., producer cell preparations) from which the composition is obtained. This enrichment can be by 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, 99.99%, 99.999%, 99.9999%, or greater than 99.9999% as compared to the starting material. In some aspects, isolated EV preparations are substantially free of residual biological products. In some aspects, the isolated EV preparations are 100% free, 99% free, 98% free, 97% free, 96% free, 95% free, 94% free, 93% free, 92% free, 91% free, or 90% free of any contaminating biological matter. Residual biological products can include abiotic materials (including chemicals) or unwanted nucleic acids, proteins, lipids, or metabolites. Substantially free of residual biological products can also mean that the EV composition contains no detectable producer cells and that only EVs are detectable.
[0065] As used herein, the term "immune modulator" refers to an agent (i.e., payload) that acts on a target (e.g. , a target cell) that is contacted with the extracellular vesicle, and regulates the immune system. Non-limiting examples of immune modulator that can be introduced into an EV (e.g., exosome) and/or a producer cell include agents such as, modulators of checkpoint inhibitors, ligands of checkpoint inhibitors, cytokines, derivatives thereof, or any combination thereof. The immune modulator can also include an agonist, an antagonist, an antibody, an antigen-binding fragment, a polynucleotide, such as siRNA, antisense oligonucleotide, a phosphorodiamidate morpholino oligomer (PMO), a peptide -conjugated phosphorodiamidate morpholino oligomer (PPMO), miRNA, IncRNA, mRNA DNA, or a small molecule. Unless indicated otherwise, in some aspects, the terms immune modulator and adjuvants can be used interchangeably. Additional examples of suitable adjuvants/immune modulators are provided elsewhere in the present disclosure.
[0066] As used herein, the term a "bio-distribution modifying agent," which refers to an agent that can modify the distribution of extracellular vesicles (e.g., exosomes, nanovesicles) in vivo or in vitro (e.g., in a mixed culture of cells of different varieties). In some aspects, the term "targeting moiety" can be used interchangeably with the term bio-distribution modifying agent. In some aspects, the targeting moiety alters the tropism of the EV (e.g., exosome) (“tropism moiety”). As used herein, the term "tropism moiety" refers to a targeting moiety that when expressed on an EV (e.g., exosome) alters and/or enhances the natural movement of the EV. For example, in some aspects, a tropism moiety can promote the EV to move towards a particular cell, tissues, or a stimuli. In some aspects, a tropism moiety can promote the EV to be taken up by a particular cell, tissue, or organ. Unless indicated otherwise, the term "targeting moiety," as used herein, encompasses tropism moieties and can be used interchangeably. The bio-distribution agent can be a biological molecule, such as a protein, a peptide, a lipid, or a carbohydrate, or a synthetic molecule. For example, the bio-distribution modifying agent can be an antibody, a synthetic polymer (e.g. , PEG), a natural
ligand (e.g., CD40L, albumin), a recombinant protein (e.g., XTEN), but not limited thereto. Non-limiting examples of such moieties are provided elsewhere in the present disclosure.
[0067] In certain aspects, the bio-distribution modifying agent is displayed on the surface of EVs (e.g., exosomes). The bio-distribution modifying agent can be displayed on the EV surface by being fused to a scaffold protein (e.g. , Scaffold X) (e.g. , as a genetically encoded fusion molecule). In some aspects, the bio-distribution modifying agent can be displayed on the EV surface by chemical reaction attaching the biodistribution modifying agent to an EV surface molecule. A non-limiting example is PEGylation. In some aspects, EVs disclosed herein (e.g., exosomes) can further comprise a bio-distribution modifying agent (in addition to an antigen, adjuvant, or immune modulator).
[0068] Non-limiting examples of tropsim or targeting moiety that can be used with the present disclosure include a C-type lectin domain family 9 member A (Clec9a) protein, a dendritic cell-specific intercellular adhesion molecule-3-grabbing non-integrin (DC-SIGN), CD207, CD40, Clec6, dendritic cell immunoreceptor (DCIR), DEC-205, lectin-like oxidized low-density lipoprotein receptor- 1 (LOX-1), MARCO, Clecl2a, DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin-1, macrophage mannose receptor (MMR), BDCA-1 (CD303, Clec4c), Dectin-2, Bst-2 (CD317), CD3, CD14, CD16, CD64, CD68, CD71, CCR5, or any combination thereof. In certain aspects, the targeting moiety is Clec9a protein. In some aspects, the targeting moiety is CD14, CD16, CD64, CD68, CD71, CCR5, or any combination thereof.
[0069] As used herein, the term "payload" refers to an agent that acts on a target (e.g. , a target cell) that is contacted with the EV (e.g., exosome). Contacting can occur in vitro or in a subject. In some aspects, unless indicated otherwise, the term payload can be used interchangeably with the terms "moiety," "agents," and "biologically active molecules." Non-limiting examples of payload that can be included on the EV, e.g., exosome, are an antigen, an adjuvant, and/or an immune modulator. Payloads that can be introduced into an EV, e.g., exosome, and/or a producer cell include agents such as, nucleotides (e.g., nucleotides comprising a detectable moiety or a toxin or that disrupt transcription), nucleic acids (e.g. , DNA or mRNA molecules that encode a polypeptide such as an enzyme, or RNA molecules that have regulatory function such as miRNA, dsDNA, IncRNA, siRNA, antisense oligonucleotide, a phosphorodiamidate morpholino oligomer (PMO), a peptide-conjugated phosphorodiamidate morpholino oligomer (PPMO), or combinations thereof), amino acids (e.g., amino acids comprising a detectable moiety or a toxin or that disrupt translation), polypeptides (e.g., enzymes), lipids, carbohydrates, peptides (e.g., cell penetrating peptides), and small molecules (e.g., small molecule drugs and toxins). In certain aspects, a payload comprises an antigen. As used herein, the term "antigen" refers to any agent that when introduced into a subject elicits an immune response (cellular or humoral) to itself. As will be apparent from the present disclosure, in some aspects, an antigen is associated with a neurological disorder. Additional disclosure relating to such antigens are provided elsewhere in the present disclosure.
[0070] As used herein, the term "affinity ligand" refers to a molecule that can selectively and preferentially bind to a specific marker, e.g., expressed on a target cell or on EVs, e.g, a scaffold moiety, e.g., PTGFRN on EVs. As described herein, in some aspects, an affinity ligand comprises a peptide (e.g., linear peptide) or protein that can increase the binding of a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to a moiety on the surface of EVs, e.g., a scaffold moiety disclosed herein. Non-limiting examples of affinity ligands that can be used with the present disclosure include an antibody, phage display peptide, fibronectin domain, camelid, VNAR, VHH domain, and combinations thereof.
[0071] As used herein, the term "antibody" encompasses an immunoglobulin whether natural or partly or wholly synthetically produced, and fragments thereof. The term also covers any protein having a binding domain that is homologous to an immunoglobulin binding domain. "Antibody" further includes a polypeptide comprising a framework region from an immunoglobulin gene or fragments thereof that specifically binds and recognizes an antigen. Use of the term antibody is meant to include whole antibodies, polyclonal, monoclonal and recombinant antibodies, fragments thereof, and further includes single-chain antibodies, humanized antibodies, murine antibodies, chimeric, mouse-human, mouse-primate, primatehuman monoclonal antibodies, anti-idiotype antibodies, antibody fragments, such as, e.g., scFv, (scFv)2, Fab, Fab', and F(ab')2, F(abl)2, Fv, dAb, and Fd fragments, diabodies, and antibody-related polypeptides. Antibody includes bispecific antibodies and multispecific antibodies so long as they exhibit the desired biological activity or function.
[0072] The terms "individual," "subject," "host," and "patient," are used interchangeably herein and refer to any mammalian subject for whom diagnosis, treatment, or therapy is desired, particularly humans. The compositions and methods described herein are applicable to both human therapy and veterinary applications. In some aspects, the subject is a mammal, and in some aspects, the subject is a human. As used herein, a "mammalian subject" includes all mammals, including without limitation, humans, domestic animals (e.g., dogs, cats and the like), farm animals (e.g., cows, sheep, pigs, horses and the like) and laboratory animals (e.g., monkey, rats, mice, rabbits, guinea pigs and the like).
[0073] As used herein, the term "substantially free" means that the sample comprising EVs, e.g., exosomes, comprise less than about 10% of macromolecules by mass/volume (m/v) percentage concentration. Some fractions can contain less than about 0.001%, less than about 0.01%, less than about 0.05%, less than about 0.1%, less than about 0.2%, less than about 0.3%, less than about 0.4%, less than about 0.5%, less than about 0.6%, less than about 0.7%, less than about 0.8%, less than about 0.9%, less than about 1%, less than about 2%, less than about 3%, less than about 4%, less than about 5%, less than about 6%, less than about 7%, less than about 8%, less than about 9%, or less than about 10% (m/v) of macromolecules.
[0074] As used herein, the term "macromolecule" means nucleic acids, contaminant proteins, lipids, carbohydrates, metabolites, or a combination thereof.
[0075] As used herein, the term "conventional exosome protein" means a protein previously known to be enriched in exosomes, including but is not limited to CD9, CD63, CD81, PDGFR, GPI anchor proteins, lactadherin LAMP2, and LAMP2B, a fragment thereof, or a peptide that binds thereto.
[0076] "Administering," as used herein, means to give a composition comprising an EV, e.g., exosome, disclosed herein to a subject via a pharmaceutically acceptable route. Routes of administration can be intravenous, e.g., intravenous injection and intravenous infusion. Additional routes of administration include, e.g., subcutaneous, intramuscular, intrathecal, intravitreal, intracranial, oral, nasal, and pulmonary administration. Exosomes can also be directly administered to the target tissue, EVs, e.g., exosomes can be administered as part of a pharmaceutical composition comprising at least one excipient.
[0077] An "immune response," as used herein, refers to a biological response within a vertebrate against foreign agents or abnormal, e.g. , cancerous cells, which response protects the organism against these agents and diseases caused by them. An immune response is mediated by the action of one or more cells of the immune system (for example, a T lymphocyte, B lymphocyte, natural killer (NK) cell, macrophage, eosinophil, mast cell, dendritic cell or neutrophil) and soluble macromolecules produced by any of these cells or the liver (including antibodies, cytokines, and complement) that results in selective targeting, binding to, damage to, destruction of, and/or elimination from the vertebrate's body of invading pathogens, cells or tissues infected with pathogens, cancerous or other abnormal cells, or, in cases of autoimmunity or pathological inflammation, normal human cells or tissues. As used herein, immune response comprises a cellular immune response, a humoral immune response, an innate cell immune response, or a combination thereof. An immune reaction includes, e.g., activation or inhibition of a T cell, e.g., an effector T cell, a Th cell, a CD4+ cell, a CD8+ T cell, or a Treg cell, or activation or inhibition of any other cell of the immune system, e.g., NK cell. Accordingly an immune response can comprise a humoral immune response (e.g., mediated by B-cells), cellular immune response (e.g., mediated by T cells), or both humoral and cellular immune responses. In some aspects, an immune response is an "inhibitory" immune response. An "inhibitory" or "tolerogenic" immune response is an immune response that blocks or diminishes the effects of a stimulus (e.g., antigen). In certain aspects, the inhibitory immune response comprises the production of inhibitory antibodies against the stimulus. In some aspects, the inhibitory immune response comprises the induction of tolerogenic cells, such as regulatory T cells (e.g., FoxP3+ regulatory CD4+ T cells). In some aspects, the inhibitory immune response comprises the production of tolerogenic cytokines/chemokines (e.g., IL-10 or TGF-J3). In some aspects, an immune response is a "stimulatory" immune response. A "stimulatory" immune response comprises an immune response that results in the generation of effectors cells (e.g., cytotoxic T lymphocytes) that can destroy and clear a target antigen (e.g., tumor antigen or
viruses). In some aspects, a stimulatory immune response comprises the production of antibodies that can specifically bind and neutralize an antigen.
[0078] As used herein, the term "cellular immune response" can be used interchangeably with the term "cell-mediated immune response" and refers to an immune response that does not predominantly involve antibodies. Instead, a cellular immune response involves the activation of different immune cells (e.g., phagocytes and antigen-specific cytotoxic T-lymphocytes) that produce various effector molecules (e.g., cytokines, perforin, granzymes) upon activation (e.g., via antigen stimulation). As used herein, the term "humoral immune response" refers to an immune response predominantly mediated by macromolecules found in extracellular fluids, such as secreted antibodies, complement proteins, and certain antimicrobial peptides. The term "antibody-mediated immune response" refers to an aspect of a humoral immune response that is mediated by antibodies.
[0079] As used herein, the term "immune cells" refers to any cells of the immune system that are involved in mediating an immune response. Non-limiting examples of immune cells include a T lymphocyte, B lymphocyte, natural killer (NK) cell, macrophage, eosinophil, mast cell, dendritic cell, neutrophil, or combination thereof. In some aspects, an immune cell expresses CD3. In certain aspects, the CD3-expressing immune cells are T cells (e.g., CD4+ T cells or CD8+ T cells). In some aspects, an immune cell that can be targeted with a targeting moiety disclosed herein (e.g. , anti-CD3) comprises a naive CD4+ T cell. In some aspects, an immune cell comprises a memory CD4+ T cell. In some aspects, an immune cell comprises an effector CD4+ T cell. In some aspects, an immune cell comprises a naive CD8+ T cell. In some aspects, an immune cell comprises a memory CD8+ T cell. In some aspects, an immune cell comprises an effector CD8+ T cell. In some aspects, an immune cell is a dendritic cell. In certain aspects, a dendritic cell comprises a plasmacytoid dendritic cell (pDC), a conventional dendritic cell 1 (cDCl), a conventional dendritic cell 2 (cDC2), inflammatory monocyte derived dendritic cells, Langerhans cells, dermal dendritic cells, lysozyme-expressing dendritic cells (LysoDCs), Kupffer cells, or any combination thereof. Accordingly, in certain aspects, an immune cell that an EV disclosed herein (e.g. , exosomes) can specifically target includes a conventional dendritic cell 1 (cDCl) and/or plasmacytoid dendritic cells (pDC). In some aspects, an immune cell is a macrophage. In some aspects, the macrophage comprises Ml macrophages, M2 macrophages, or both. In certain aspects, the macrophage is a microglia, meningeal macrophage, perivascular macrophage, choroid plexus macrophage, or combinations thereof.
[0080] As used herein, the term "T cell" or "T-cell" refers to a type of lymphocyte that matures in the thymus. T cells play an important role in cell-mediated immunity and are distinguished from other lymphocytes, such as B cells, by the presence of a T-cell receptor on the cell surface. T-cells include all types of immune cells expressing CD3, including, but not limited to, T-helper cells (CD4+ cells), cytotoxic T-cells (CD8+ cells), natural killer T-cells, T-regulatory cells (Treg), T follicular helper (Tfh) cells, peripheral Tfh cells, mucosal-associated invariant T (MAIT) cells, and gamma-delta T cells.
[0081] A "naive" T cell refers to a mature T cell that remains immunologically undifferentiated (i. e. , not activated). Following positive and negative selection in the thymus, T cells emerge as either CD4+ or CD8+ naive T cells. In their naive state, T cells express L-selectin (CD62L+), IL-7 receptor-a (IL-7R-a), and CD132, but they do not express CD25, CD44, CD69, or CD45RO. As used herein, "immature" can also refers to a T cell which exhibits a phenotype characteristic of either a naive T cell or an immature T cell, such as a TSCM cell or a TCM cell. For example, an immature T cell can express one or more of L- selectin (CD62L+), IL-7Ra, CD132, CCR7, CD45RA, CD45RO, CD27, CD28, CD95, CXCR3, and LFA- 1. Naive or immature T cells can be contrasted with terminal differentiated effector T cells, such as TEM cells and TEFF cells.
[0082] As used herein, the term "effector" T cells or "TEFF" cells refers to a T cell that can mediate the removal of a pathogen or cell without requiring further differentiation. Thus, effector T cells are distinguished from naive T cells and memory T cells, and these cells often have to differentiate and proliferate before becoming effector cells.
[0083] As used herein, the term "memory" T cells refer to a subset of T cells that have previously encountered and responded to their cognate antigen. In some aspects, the term is synonymous with "antigen-experienced" T cells. In some aspects, memory T cells can be effector memory T cells or central memory T cells. In some aspects, the memory T cells are tissue-resident memory T cells. As used herein, the term "tissue-resident memory T cells" or "TRM cells" refers to a lineage of T cells that occupies tissues (e.g., skin, lung, gastrointestinal tract) without recirculating. TRM cells are transcriptionally, phenotypically and functionally distinct from central memory and effector memory T cells which recirculate between blood, the T cell zones of secondary lymphoid organs, lymph and nonlymphoid tissues. One of the roles of TRM cells is to provide immune protection against infection in extralymphoid tissues.
[0084] As used herein, the term "dendritic cells" or "DCs" refers to a class of bone-marrow-derived immune cells that are capable of processing extracellular and intracellular proteins and to present antigens in the context of MHC molecules to prime naive T cells. In some aspects, dendritic cells can be divided into further subtypes, such as conventional dendritic cell 1 (cDCl), conventional dendritic cell 2 (cDC2), plasmacytoid dendritic cell (pDC), inflammatory monocyte derived dendritic cells, Langerhans cells, dermal dendritic cells, lysozyme-expressing dendritic cells (LysoDCs), Kupffer cells, and combinations thereof. In certain aspects, the different DC subsets can be distinguished based on their phenotypic expression. For example, in some aspects, human cDCl cells are CDlc" and CD141+. In some aspects, human cDC2 cells are CDlc+ and CD 141’. In some aspects, human pDC cells are CD123+. In some aspects, mouse cDCl cells are XCR1+, Clec9a+, and Sirpa". In some aspects, mouse cDC2 cells are CD8+, CD1 lb+, Sirpa+, XCR1", and CDlc,b+. In some aspects, mouse pDC cells are CD137+, XCR1", and Sirpa". Other phenotypic markers for distinguishing the different DC subsets are known in the art. See, e.g. , Collin et al. , Immunology 154(1): 3-20 (2018). In some aspects, the different DC subsets can be distinguished based on
their functional properties. For example, in certain aspects, pDCs produce large amounts of IFN-a, while cDCls and cDC2s produce inflammatory cytokines, such as IL-12, IL-6, and TNF-a. Other methods of distinguishing the different DC subsets are known in the art. See, e.g., U.S. Patent Nos. 8,426,565 B2 and 9,988,431, each of which is herein incorporated by reference in its entirety.
[0085] As used herein, the term "macrophage" refers to a mononuclear phagocyte characterized by the expression of at least CD 14 and lack of expression of dendritic cell markers. Macrophages can be typically divided into (i) classically-activated macrophages ("Ml macrophages") and (ii) alternatively- activated macrophages ("M2 macrophages"). Martinez et al., Annu. Rev. Immunol. 27:451-483 (2009). Generally, Ml macrophages exhibit potent anti -microbial properties, reminiscent of type 1 T-helper lymphocyte (Thl) responses. In contrast, M2 macrophages promote type 2 T-helper lymphocyte (Th2)-like responses, secrete less pro-inflammatory cytokines, and assist resolution of inflammation by trophic factor synthesis and phagocytosis. Mosser et al., Nature Rev. 8:958-969 (2008). M2 macrophages can be further divided into three distinct subclasses, i.e., M2a, M2b, and M2c, defined by specific cytokine profiles. Mantovani et al., Trends Immunol. 25:677-686 (2004). While M2 macrophages are generally characterized by low production of pro-inflammatory cytokines, such as IL- 12, and high production of anti-inflammatory cytokines such as IL- 10, M2b macrophages retain high levels of inflammatory cytokine production, such as TNF-a and IL-6. Mosser, J. Leukocyte Biol. 73:209-212 (2003).
[0086] Macrophages can be polarized by their microenvironment to assume different phenotypes associated with different stages of inflammation and healing. Stout et al., J. Immunol. 175:342-349 (2005). Certain macrophages are indispensible for wound healing. They participate in the early stages of cell recruitment and of tissue defense, as well as the later stages of tissue homeostasis and repair. Pollard, Nature Rev. 9:259-270 (2009). Macrophages derived from peripheral blood monocytes have been used to treat refractory ulcers. Danon et al., Exp. Gerontol. 32:633-641 (1997); Zuloff-Shani et al., Transfus. Apher. Sci. 30: 163-167 (2004), each of which is incorporated herein by reference as if set forth in its entirety.
[0087] The term "immunoconjugate," as used herein, refers to a compound comprising a binding molecule (e.g., an antibody) and one or more moieties, e.g., therapeutic or diagnostic moieties, chemically conjugated to the binding molecule. In general, an immunoconjugate is defined by a generic formula: A- (L-M)n, wherein A is a binding molecule (e.g., an antibody), L is an optional linker, and M is a heterologous moiety which can be for example a therapeutic agent, a detectable label, etc., and n is an integer. In some aspects, multiple heterologous moieties can be chemically conjugated to the different attachment points in the same binding molecule (e.g., an antibody). In some aspects, multiple heterologous moieties can be concatenated and attached to an attachment point in the binding molecule (e.g., an antibody). In some aspects, multiple heterologous moieties (being the same or different) can be conjugated to the binding molecule (e.g., an antibody).
[0088] Immunoconjugates can also be defined by the generic formula in reverse order. In some aspects, the immunoconjugate is an "antibody-Drug Conjugate" ("ADC"). In the context of the present disclosure, the term "immunoconjugate" is not limited to chemically or enzymatically conjugates molecules. The term "immunoconjugate" as used in the present disclosure also includes genetic fusions. In some aspects of the present disclosure, the biologically active molecule is an immunoconjugate. The terms "antibodydrug conjugate" and "ADC" are used interchangeably and refer to an antibody linked, e.g., covalently, to a therapeutic agent (sometimes referred to herein as agent, drug, or active pharmaceutical ingredient) or agents. In some aspects of the present disclosure, the biologically active molecule (i.e., a payload) is an antibody-drug conjugate.
[0089] "Treat," "treatment," or "treating," as used herein refers to, e.g., the reduction in severity of a disease or condition; the reduction in the duration of a disease course; the amelioration or elimination of one or more symptoms associated with a disease or condition; the provision of beneficial effects to a subject with a disease or condition, without necessarily curing the disease or condition. The term also include prophylaxis or prevention of a disease or condition or its symptoms thereof. In one aspect, the term "treating" or "treatment" means inducing an immune response in a subject against an antigen.
[0090] "Prevent" or "preventing," as used herein, refers to decreasing or reducing the occurrence or severity of a particular outcome. In some aspects, preventing an outcome is achieved through prophylactic treatment.
IL Compositions of the Present Disclosure
[0091] Disclosed herein are compositions comprising an EV (e.g., exosome), which has been modified to comprise a KRAS antigen. In some aspects, a composition provided herein further comprises an adjuvant, a T helper peptide, or both. In certain aspects, the composition can further comprise one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator). In some aspects, an adjuvant, T helper peptide, and/or additional moieties of interest described herein (e.g., targeting moiety and/or immune modulator) are associated with the EV (e.g., exosome). As used herein, a moiety of interest (e.g., adjuvant, T helper peptide, targeting moiety, and/or immune modulator) is "associated with" an EV (e.g. , exosome), where the moiety of interest is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In certain aspects, an adjuvant, T helper peptide, and/or additional moieties of interest present in the compositions described herein are not associated with the EV (e.g., exosome). As used herein, a moiety of interest (e.g., adjuvant, T helper peptide, targeting moiety, and/or immune modulator) is "not associated with" the EV (e.g., exosome), where the moiety of interest and the EV exist in the composition independent of one another (e.g., in separate compartments of the composition).
IE A, Extracellular Vesicles (e.g., Exosomes)
[0092] EVs (e.g., exosomes) described herein are extracellular vesicles with a diameter between about 20-300 nm. In certain aspects, an EV (e.g. , exosome) of the present disclosure has a diameter between about 20-80 nm, between about 80-300 nm, between about 80-290 nm, between about 80-280 nm, between about 80-270 nm, between about 80-260 nm, between about 80-250 nm, between about 80-240 nm, between about 80-230 nm, between about 80-220 nm, between about 80-210 nm, between about 80-200 nm, between about 80-190 nm, between about 80-180 nm, between about 80-170 nm, between about 80-160 nm, between about 80-150 nm, between about 80-140 nm, between about 80-130 nm, between about 80-120 nm, between about 80-110 nm, between about 80-100 nm, between about 80-90 nm, between about 90-300 nm, between about 90-290 nm, between about 90-280 nm, between about 90-270 nm, between about 90-260 nm, between about 90-250 nm, between about 90-240 nm, between about 90-230 nm, between about 90-220 nm, between about 90-210 nm, between about 90-200 nm, between about 90-190 nm, between about 90-180 nm, between about 90-170 nm, between about 90-160 nm, between about 90-150 nm, between about 90-140 nm, between about 90-130 nm, between about 90-120 nm, between about 90-110 nm, between about 90-100 nm, between about 100-300 nm, between about 110-290 nm, between about 120-280 nm, between about 130-270 nm, between about 140-260 nm, between about 150-250 nm, between about 160-240 nm, between about 170- 230 nm, between about 180-220 nm, or between about 190-210 nm. The size of the EV, e.g., exosome, described herein can be measured according to different methods known in the art, such as those described elsewhere in the present disclosure.
[0093] In some aspects, an EV (e.g., exosome) comprises a bi-lipid membrane comprising an interior surface and an exterior surface. In certain aspects, the interior surface faces the inner core (i.e., lumen) of the EV (e.g., exosome). In certain aspects, the exterior surface can be in contact with the endosome, the multivesicular bodies, or the membrane/cytoplasm of a producer cell or a target cell.
[0094] In some aspects, the bi-lipid membrane of an EV (e.g., exosome) useful for the present disclosure comprises one or more scaffold moieties, which are capable of linking, e.g., KRAS antigen, adjuvant, and/or T helper peptide, to the EV (e.g., exosome) (e.g., either on the luminal surface or on the exterior surface). In certain aspects, scaffold moieties are polypeptides ("exosome proteins"). In some aspects, scaffold moieties are non-polypeptide moieties. In some aspects, exosome proteins include various membrane proteins, such as transmembrane proteins, integral proteins and peripheral proteins, enriched on the exosome membranes. They can include various CD proteins, transporters, integrins, lectins, and cadherins. In certain aspects, a scaffold moiety (e.g., exosome protein) comprises Scaffold X. In some aspects, a scaffold moiety (e.g., exosome protein) comprises Scaffold Y. In some aspects, a scaffold moiety (e.g., exosome protein) comprises both a Scaffold X and a Scaffold Y.
[0095] In some aspects, EVs (e.g., exosomes) described herein are capable of delivering a moiety of interest (e.g. , KRAS antigen, adjuvant, T cell helper peptide, targeting moiety, and/or immune modulator) to a target and thereby, induce an effect on the target. For instance, in some aspects, an EV (e.g. , exomsome)
(or a composition comprising the EV) described herein (e.g., comprising a KRAS antigen) is capable of inducing an immune response (e.g., CD8+ T cell response, CD4+ T cell response, B cell response, or any combination thereof) against the KRAS antigen after contacting with a target. Contacting can occur in vitro or in vivo (e.g., in a subject that received an administration of the EV or a composition comprising the EV). [0096] Additionally, as is apparent from the present disclosure, EVs (e.g., exosomes) described herein differ from other vaccine platforms in that the EVs comprise one or more of the following properties: (i) flexibility of moiety display, (ii) diverse combinations of moieties of interest, (iii) enhanced cell-specific tropism, (iv) selectively promoting T-cell, B-cell, or Treg/tolerogenic immune responses, or (v) any combination thereof.
[0097] In some aspects, EVs (e.g., exosomes) described herein provide flexibility of moiety display. For instance, a moiety of interest (e.g., KRAS antigen, adjuvant, T cell helper peptide, targeting moiety, and/or immune modulator) (i) can be directly linked to a surface of the EV (e.g., exterior surface and/or luminal surface), (ii) can be attached to a scaffold moiety (e.g., Scaffold X and/or Scaffold Y) and then linked to a surface of the EV (e.g., exterior surface and/or luminal surface), (iii) can be expressed in the lumen of the EV, or (iv) combinations thereof. Such flexibility, which can allow for rapid modifcations of the EVs to comprise different moieties of interest) can be useful in developing EV (e.g., exosome)-based vaccines for treating the diseases and disorders described herein. For instance, a single EV (e.g., exosome) can be used in treating a wide range of diseases or disorders by simply "plugging" a moiety of interest (e.g. , antigen) into the EVs or rapidly attaching a moiety (e.g., antigen of interest) as a "clip-on" attachment to the EVs). Methods of producing such modular or "plug and play" EVs are provided elsewhere in the present disclosure
[0098] In some aspects, EVs (e.g., exosomes) that are useful for the present disclosure allow for the diverse combinations of different moieties of interest (e.g., KRAS antigen, adjuvant, T cell helper peptide, targeting moiety, and/or immune modulator). For instance, in some aspects, an EV (e.g., exosome) that can be used with the present disclosure comprises: (i) a KRAS antigen and (ii) an adjuvant. In some aspects, the EV (e.g., exosome) comprises: (i) a KRAS antigen, (ii) an adjuvant, and (iii) a T helper peptide. In some aspects, the EV (e.g. , exosome) comprises: (i) a KRAS antigen, (ii) an adjuvant, (iii) a T helper peptide, and (iv) an additional moiety of interest (e.g., targeting moiety and/or immune modulator). Moreover, in some of these aspects, the EV (e.g., exosome) can comprise a single KRAS antigen, a single adjuvant, a single T helper peptide, and/or single additional moiety of interest (e.g. , targeting moiety and/or immune modulator). In some aspects, the EV (e.g., exosome) can comprise multiple KRAS antigens, multiple adjuvants, multiple T helper peptides, and/or multiple additional moieties of interest.
[0099] In some aspects, EVs (e.g., exosomes) described herein can be engineered to exhibit enhanced cell-specific tropism. For instance, the EVs can be engineered to express on their exterior surface a targeting moiety (e.g. , antibodies and/or proteins) that can specifically bind to a marker on a specific cell.
In some aspects, EVs (e.g. , exosomes) described herein can be engineered to induce certain types of immune responses (e.g., T cell, B cell, and/or Treg/tolerogenic immune responses). Additional disclosure relating to such properties are provided elsewhere in the present disclosure. B, KRAS Antigen
[0100] As described herein, EVs (e.g., exosomes) of the present disclosure comprises an antigen derived from a KRAS protein (referred to herein as "KRAS antigen"). As used herein, the term "KRAS antigen" does not comprise an antigen derived from a wild-type KRAS protein.
[0101] Kirsten rat sarcoma viral oncogene homology (KRAS) is a member of a superfamily of guanosine-5-triphosphatase (GTPase) proteins that also includes NRAS and HRAS. The primary role of the members of this superfamily is to transmit signals from upstream cell surface receptors (e.g., EGFR, FGFR, and ERBB2-4) to downstream proliferation and survival pathways such as RAF-MEK-ERK, PI3K-AKT- mTOR. and RALGDS-RA. Adderley, H., et al., EBioMedicine 41:711-716 (2019). KRAS mutations have been implicated in many types of cancers, including more than 90% of pancreatic cancers, 35-45% of colorectal cancers, and approximately 25% of lung cancers. Zeitouni, D., et al., Cancers 8(4): 45 (2016); Tan, C., et al., World J Gastroenterol 18(37): 5171-5180 (2012); and Roman, M., et al., Molecular Cancer 17:33 (2018).
[0102] KRAS is known in the art by various names. Such names include: KRAS Proto-Oncogene, GTPase; V-Ki-Ras2 Kirsten Rat Sarcoma 2 Viral Oncogene Homolog; GTPase KRas; C-Ki-Ras; K-Ras 2; KRAS2; RASK2; V-Ki-Ras2 Kirsten Rat Sarcoma Viral Oncogene Homolog; Kirsten Rat Sarcoma Viral Proto-Oncogene; Cellular Transforming Proto-Oncogene; Cellular C-Ki-Ras2 Proto-Oncogene; Transforming Protein P21; PR310 C-K-Ras Oncogene; C-Kirsten-Ras Protein; K-Ras P21 Protein; and Oncogene KRAS2.
[0103] The sequence for human KRAS protein can be found under publicly available Accession Numbers: P01116 (canonical sequence), A8K8Z5, B0LPF9, P01118, and Q96D10, each of which is incorporated by reference herein in its entirety. There are two isoforms of the human wild-type KRAS protein (P01116), resulting from alternative splicing. Isoform 2A (Accession Number: P01116-1; SEQ ID NO: 381) is the canonical sequence. It is also known as K-Ras4A. Isoform 2B (Accession Number: P01116- 2; also known as K-Ras4B; SEQ ID NO: 382) differs from the canonical sequence as follows: (i) 151-153: RVE GVD; and (ii) 165-189: QYRLKKISKEEKTPGCVKIKKCIIM (SEQ ID NO: 389) KHKEKMSKDGKKKKKKSKTKCVIM (SEQ ID NO: 390).
[0104] Natural variants of the human KRAS gene product are known. For example, natural variants of human KRAS protein can contain one or more amino acid substitutions selected from: K5E, K5N, G10GG, G10V, G12A, G12C, G12D, G12F, G12I, G12L, G12R, G12S, G12V, G13A, G13C, G13D, G13E, G13R, G13S, G13V, V14I, A18D, L19F, T20M, Q22E, Q22H, Q22K, Q22R, Q25H, N26Y, F28L, E31K, D33E, P34L, P34Q, P34R, I36M, R41K, D57N, T58I, A59T, G60D, G60R, G60S, G60V, Q61A, Q61H, Q61K, Q61L, Q61P, Q61R, E63K, S65N, R68S, Y71H, T74A, L79I, R97I, Q99E, Ml 1 IL, KI 17N, KI 17R, D119G, S122F, T144P, A146P, A146T, A146V, K147E, K147T, R149K, L159S, I163S, R164Q, I183N, I84M, or combinations thereof. Natural variants that are specific to KRAS protein Isoform 2B contain one or more amino acid substitutions selected from: V152G, D153V, F156I, F156L, or combinations thereof.
[0105] As described herein, a KRAS antigen that is useful for the present disclosure does not comprise a wild-type KRAS protein (e.g., SEQ ID NO: 381 and 382, as shown in Table 1). Accordingly, a KRAS antigen that is useful for the present disclosure is derived from a KRAS mutant protein, such that the KRAS antigen comprises one or more amino acid mutations present in the KRAS mutant protein. As used herein, the terms "KRAS mutant protein" and "KRAS variant protein" can be used interchangeably and refer to a KRAS protein that differs in sequence from the wild-type KRAS protein (e.g., SEQ ID NO: 381 or 382). Unless indicated otherwise, the term "KRAS antigen" comprises both wild-type and KRAS mutant proteins.
[0106] In some aspects, a KRAS antigen that is useful for the present disclosure can be derived from any KRAS mutant proteins known in the art, such as those described herein (e.g., comprising one or more of the amino acid substitutions described above). In some aspects, the KRAS antigen is derived from a KRAS mutant and comprises one or more of the following amino acid substitutions: G12D, G12C, G12V, G12A, G12R, G12S, G13C, G13D, G13A, G13R, G13S, G13V, Q61H, Q61L, Q61R, Q61K, A18D, K117N, or any combination thereof, as compared to the wild-type KRAS protein (SEQ ID NO: 381). In some aspects, the KRAS antigen comprises one or more amino acid substitutions selected from the group
consisting ofG12D, G12C, G12V, G13D, and any combination thereof, as compared to the wild-type KRAS protein. (SEQ ID NO: 381).
[0107] In some aspects, a KRAS antigen useful for the present disclosure comprises a natural peptide. In some aspects, the KRAS antigen comprises a synthetic peptide. In certain aspects, the KRAS antigen comprises both a natural and a synthetic peptide.
[0108] In some aspects, the KRAS antigen comprises or consists of an epitope within amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids in length. In some aspects, the KRAS antigen comprises an epitope within amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids, in length. In some aspects, the KRAS antigen consists of an epitope within amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids, in length.
[0109] In some aspects, the KRAS antigen comprises or consists of an epitope within amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22
amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length. In some aspects, the KRAS antigen comprises an epitope within amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, , at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length. In some aspects, the KRAS antigen consists of an epitope within amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, , at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids in length.
[0110] In some aspects, the KRAS antigen comprises or consists of an epitope within amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least 37 amino acids, at least 38 amino acids, at least 39 amino acids, or all 40 amino acids in length. In some aspects, the KRAS antigen comprises an epitope within amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids,
at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least 37 amino acids, at least 38 amino acids, at least 39 amino acids, or all 40 amino acids in length. In some aspects, the KRAS antigen consists of an epitope within amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least 37 amino acids, at least 38 amino acids, at least 39 amino acids, or all 40 amino acids in length.
[oni] As will be apparent to those skilled in the arts, the overall length of a peptide can influence both the type (e.g. , CD8+ T cell response, CD4+ T cell response, and/or B cell response) and the magnitude of immune response induced against the antigen. Khan, M., et al., Set Rep 9(1): 13321 (Sep. 2019), which is incorporated herein by reference in its entirety. Accordingly, in some aspects, a KRAS antigen that is useful for the present disclosure is less than about 50 amino acids in length, less than about 45 amino acids in length, less than about 40 amino acids in length, less than about 35 amino acids in length, less than about 30 amino acids in length, less than about 25 amino acids in length, less than about 20 amino acids in length, less than about 15 amino acids in length, or less than about 10 amino acids in length. In certain aspets, the KRAS antigen is between about 12 to about 20 amino acids in length.
[0112] As described herein, in some aspects, an EV (e.g., exosome) of the present disclosure comprises a single KRAS antigen. In some aspects, an EV (e.g., exosome) disclosed herein comprises multiple KRAS antigens (e.g., at least about 2, at least about 3, at least about 4, at least about 5, at least about 6, at least about 7, at least about 8, at least about 9, or at least about 10). In certain aspects, each of the multiple KRAS antigens is different (e.g., each of the KRAS antigens comprise a different amino acid substitution, compared to a corresponding KRAS antigen derived from a wild-type KRAS protein). In some aspects, some or all of the multiple KRAS antigens are the same.
[0113] In some aspects, where multiple KRAS antigens are involved, some or all of the multiple KRAS antigens can be joined to form a cancetmer of multiple KRAS antigens. In certain aspects, some or all of the multiple KRAS antigens can be separated by a spacer. In constructing such a KRAS antigens, any suitable spacers known in the art can be used. In some aspects, a suitable spacer comprises the amino acid
sequence selected from GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), AAY, or combinations thereof. For instance, in certain aspects, an EV (e.g., exosome) disclosed herein comprises multiple KRAS antigens, wherein the multiple KRAS antigens are separated by the GPGPG spacer. In some aspects, the multiple KRAS antigens are separated by the GSGSG (SEQ ID NO: 391) spacer. In some aspects, the multiple KRAS antigens are separated by the AAY spacer. In certain aspects, the multiple KRAS antigens are separated by any combination of the GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), and AAY spacers.
[0114] As described herein, in some aspects, the KRAS antigen can be encapsulated within the lumen of the EV. In some aspects, the KRAS antigen is linked to the exterior surface and/or luminal surface of the EVs (e.g., exosomes) using a scaffold moiety (e.g., Scaffold X and/or Scaffold Y). In some aspects, a KRAS antigen can be directly linked (i.e., without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g. , exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof. To improve the attachment of the antigens to a surface of the EVs (e.g., exosomes) using such methods, the antigens described herein can be further modified. In certain aspects, an antigen comprises a peptide, which has been modified to contain a N-terminal lysine.
[0115] In some aspects, such a modification allows for the attachment of the antigen to a surface of the EV (e.g., exosome) with chemical conjugation. For example, to enable click chemistry conjugation, an azide or strained alkyne (e.g., difluorinated cyclooctyne (DIFO), dibenzocyclooctyne (DBCO), or bicyclononyne (BCN)) would have to be attached (or linked) to the EV (e.g. , exosome) and/or to the antigen. In certain aspects, the azide or strained alkyne (e.g. , DIFO, DBCO, or BCN) conjugates to the to the primary amine side chain on the N-terminal amino acid (e.g., lysine) of the antigen, which in turn can interact with the azide that can be attached to a surface of the EV. In some aspects, the azide can be attached to the antigen (via the primary amine side chain on the N-terminal amino acid (e.g., lysine)), and the strained alkyne can be attached to a surface of the EV (e.g., exosome).
[0116] In some aspects, modifying the antigen to comprise a N-terminal lysine can also be useful in linking the antigens to a surface of the EVs (e.g., exosomes) using anchoring moieties. In such aspects, the anchoring moiety (e.g. , cholesterol, fatty acid, and/or vitamin E) can attach to the N-terminal lysine via the primary amine side chain. Once attached, the antigens can be readily inserted into the membrane of the EVs (e.g., exosomes) via the anchoring moieties.
[0117] As will be apparent to those skilled in the arts, the above described approaches to linking an antigen to the exterior surface and/or luminal surface of the EVs (e.g., exosome) can also be performed by modifying one or more proteins on the EVs to contain unnatural amino acids with side chains to allow for the binding of molecules such as the azide, strained alkyne (e.g., difluorinated cyclooctyne (DIFO), dibenzocyclooctyne (DBCO), or bicyclononyne (BCN)) or combinations thereof. Additional disclosure
regarding such approaches to linking an antigen to a surface of the EVs (e.g., exosomes) are provided elsewhere in the present disclosure.
II. C. T Helper Peptides
[0118] CD4+ T cells play an important role in the induction of both T and B cell immune responses against many diseases and disorders. See, e.g., Borst, J., et al., Nat Rev Immunol 18(10): 635-647 (Oct. 2018); and Crotty, S., Nat Rev Immunol 15(3): 185-189 (Mar. 2015); both of which are herein incorporated by reference in their entireties.
[0119] Accordingly, in some aspects, a composition comprising an EV (e.g., exosome) described herein can further comprise a T helper peptide. As described herein, the T helper peptide can be associated with the EV (e.g., exosome), such that the T helper peptide is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In certain aspects, the T helper peptide is not associated with the EV. As will be apparent to those skilled in the art, the relevant disclosures provided below can equally apply to both T helper peptides that are associated with the EV (e.g., exosomes) and those that are not.
[0120] As used herein, the term "T helper peptide" refers to any peptide that is capable of activating a CD4+ T cell, which can in turn provide the necessary help to induce the activation of both CD8+ T cells and B cells. Non-limiting examples of such T helper peptides include: a helper peptide derived from tubercle bacillus, a helper peptide derived from measles virus, a helper peptide derived from hepatitis B virus, a helper peptide derived from hepatitis C virus, a helper peptide derived from Chlamydia trachomatis, a helper peptide derived from P. falciparum sporozoite, a helper peptide derived from keyhole limpet haemocyanin, a helper peptide derived from tetanus toxin, a helper peptide derived from pertussis toxin, a helper peptide derived from diphtheria toxin, helper peptides derived from cancer cells (e.g., IMA-MMP-001 helper peptide, CEA-006 helper peptide, MMP-001 helper peptide, TGFBI-004 helper peptide, HER-2/neu(aa776- 790) helper peptide, AE36 helper peptide, AE37 helper peptide, MET-005 helper peptide, and BIR-002 helper peptide), and universal T helper peptide (e.g., PADRE), and altered peptides thereof.
[0121] In some aspects, the T helper peptide that can be used with the present disclosure is a universal T helper peptide. As used herein, the term "universal T helper peptide" refers to a T helper peptide that binds to a broad range of MHC class II haplotypes, and can thereby activate CD4+ T cells expressing those MHCs. In some aspects, the universal T helper peptide comprises a PADRE, tetanus toxin, diphtheria toxin, HBV peptide, measles peptide, or any combination thereof. In certain aspects, the universal T helper peptide is PADRE (AKFVAAWTLKAAA) (SEQ ID NO: 395), or such as that described in Benmohamed et al., Immunology 106: 113-121 (2001), which is herein incorporated by reference in its entirety. In some aspects, the universal T helper peptide is a tetanus toxin (e.g. , QYIKANSKFIGITE; amino acid residues 830-843 of tetanus; SEQ ID NO: 396), or such as that described in Falugi et al. , Eur J Immunol 31: 3816-3824 (2001), which is herein incorporated by reference in its entirety. In some aspects, the
universal T helper peptide is a diphtheria toxin (e.g. , QSIALSSLMVAQAIP; amino acid residues 356-370 of diphtheria toxin; SEQ ID NO: 397), or such as that described in Diethelm-Okita, et al., J Infec Dis 181: 1001-1009 (2000), which is herein incorporated by reference in its entirety. In certain aspects, the diphtheria toxin comprises CRM 197. In some aspects, the universal T helper peptide is a HBV peptide, such as HbsAg. In some aspects, the universal T helper peptide is a peptide derived from a measles virus. In some aspects, an EV (e.g., exosome) comprises a T helper peptide, wherein the T helper peptide comprises a KRAS CD4+ T cell peptide antigen.
[0122] In some aspects, EVs (e.g. , exosomes) described herein comprises a single T helper peptide. In some aspects, an EV (e.g. , exosome) disclosed herein comprises multiple multiple T helper peptides (e.g. , at least 2, 3, 4, 5, 6, 7, 8, 9, 10). In certain aspects, each of the multiple T helper peptides is different. In some aspects, some or all of the multiple T helper peptides are the same.
[0123] As described herein, in some aspects, a T helper peptide can be encapsulated within the lumen of the EV. In some aspects, the T helper peptide is linked to the exterior surface and/or luminal surface of the EVs (e.g., exosomes) using a scaffold moiety (e.g., Scaffold X and/or Scaffold Y). In some aspects, the T helper peptide can be directly linked (i.e., without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g., exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof. Non-limiting examples of chemical conjugation includes: moiety, copper-free, biorthogonal click chemistry (e.g., azide/strained alkyne (DIFO, DBCO, BCN), metal -catalyzed click chemistry (e.g., CuAAC, RuAAC), or any combination thereof. Additional disclosure relating to such approaches (including modifications that can be made to the T helper peptides and or EVs for improved binding) to linking the T helper peptides to the exterior surface and/or luminal surface of the EVs are provided elsewhere in the present disclosure (see, e.g., section II.B) P, Adjuvants
[0124] In some aspects, a composition comprising an EV (e.g., exosome) described herein (i.e., comprising a KRAS antigen) can further comprise an adjuvant. As described herein, the adjuvant can be associated with the EV (e.g., exosome), such that the adjuvant is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In certain aspects, the adjuvant is not associated with the EV. As will be apparent to those skilled in the art, the relevant disclosures provided below can equally apply to both adjuvants that are associated with the EV (e.g., exosomes) and those that are not.
[0125] As used herein, the term "adjuvant" refers to any substance that enhances the therapeutic effect of the EVs (e.g., exosomes) of the present disclosure (e.g., increasing an immune response to the KRAS antigen). Accordingly, EVs (e.g., exsomes) described herein comprising an adjuvant are capable of increasing an immune response, e.g., to an antigen, by at least about 5%, at least about 10%, at least about
20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, at least about 100%, at least about 250%, at least about 500%, at least about 750%, at least about 1,000% or more or more, compared to a reference (e.g., corresponding EV without the adjuvant or a non-EV delivery vehicle comprising an antigen alone or in combination with the adjuvant). In some aspects, incorporating an adjuvant disclosed herein to an EV (e.g. , exosome) can increase an immune response, e.g., to an antigen, by at least about 1-fold, at least about 2-fold, at least about 3-fold, at least about 4-fold, at least about 5-fold, at least about 6-fold, at least about 7-fold, at least about 8-fold, at least about 9-fold, at least about 10-fold, at least about 20-fold, at least about 30-fold, at least about 40- fold, at least about 50-fold, at least about 60-fold, at least about 70-fold, at least about 80-fold, at least about 90-fold, at least about 100-fold, at least about 200-fold, at least about 300-fold, at least about 400-fold, at least about 500-fold, at least about 600-fold, at least about 700-fold, at least about 800-fold, at least about 900-fold, at least about 1,000-fold, at least about 2,000-fold, at least about 3,000-fold, at least about 4,000- fold, at least about 5,000-fold, at least about 6,000-fold, at least about 7,000-fold, at least about 8,000-fold, at least about 9,000-fold, at least about 10,000-fold or more, compared to a reference (e.g., corresponding EV comprising the antigen alone or a non-EV delivery vehicle comprising an antigen alone or in combination with the adjuvant).
[0126] In some aspects, EVs (e.g., exosomes) described herein comprises a single adjuvant. In some aspects, an EV (e.g., exosome) disclosed herein comprises multiple adjuvants (e.g., at least about 2, at least about 3, at least about 4, at least about 5, at least about 6, at least about 7, at least about 8, at least about 9, or at least about 10). In certain aspects, each of the multiple adjuvants is different. In some aspects, some or all of the multiple adjuvants are the same.
[0127] As described herein, in some aspects, an adjuvant can be encapsulated within the lumen of the EV. In some aspects, the adjuvant is linked to the exterior surface and/or luminal surface of the EVs (e.g. , exosomes) using a scaffold moiety (e.g. , Scaffold X and/or Scaffold Y). In some aspects, the adjuvant can be directly linked (i. e. , without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g., exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof. Additional disclosure relating to such approaches (including modifications that can be made to the adjuvants and or EVs for improved binding) to linking the adjuvants to the exterior surface and/or luminal surface of the EVs are provided elsewhere in the present disclosure.
[0128] Non-limiting examples of adjuvants that can be used with the present disclosure include: Stimulator of Interferon Genes (STING) agonist, a toll-like receptor (TLR) agonist, an inflammatory mediator, RIG-I agonists, alpha-gal-cer (NKT agonist), heat shock proteins (e.g., HSP65 and HSP70), C- type lectin agonists (e.g., beta glucan (Dectin 1), chitin, and curdlan), and combinations thereof.
[0129] In some aspects, incorporating an adjuvant (e.g., such as those disclosed herein) to an EV (e.g., exosome) can broaden an immune response induced by the EV. As used herein, to "broaden an immune response" refers to enhancing the diversity of an immune response. In some aspects, the diversity of an immune response can be enhanced through epitope spreading (i.e., inducing and/or increasing an immune response (cellular and/or humoral immune response) against a greater number/variety of epitopes on an antigen). In some aspects, the diversity of an immune response can be enhanced through the production of different and/or multiple antibody isotypes (e.g., IgG, IgA, IgD, IgM, and/or IgE).
[0130] In some aspects, an adjuvant (e.g., such as those disclosed herein) can also help regulate the type of immune response induced by the EV (e.g., exosome). For example, in some aspects, incorporating an adjuvant to an EV (e.g., exosome) can help drive an immune response towards a more Thl phenotype. As used herein, a "Thl" immune response is generally characterized by the production of IFN-y, which can activate the bactericidal activities of innate cells (e.g. , macrophages), help induce B cells to make opsonizing (marking for phagocytosis) and complement-fixing antibodies, and/or lead to cell-mediated immunity (i. e. , not mediated by antibodies). In general, Thl responses are more effective against intracellular pathogens (viruses and bacteria that are inside host cells) and suppressing tumor growth.
[0131] In some aspects, incorporating an adjuvant to an EV (e.g., exosome) can help drive an immune response towards a more Th2 phenotype. As used herein, a "Th2" immune response can be characterized by the release of certain cytokines, such as IL-5 (induces eosinophils in the clearance of parasites) and IL-4 (facilitates B cell isotype switching). In general, Th2 responses are more effective against extracellular bacteria, parasites including helminths and toxins.
[0132] In some aspects, incorporating an adjuvant to an EV (e.g., exosome) can help drive an immune response towards a more Thl7 phenotype. As used herein, a "Thl7" immune response is mediated by Thl7 cells. As used herein, "Thl7 cells" refer to a subset of CD4+ T cells characterized by the production of pro-inflammatory cytokines, such as IL- 17A, IL- 17F, IL-21 , IL-22, and granulocyte-macrophage colonystimulating factor (GM-CSF). Thl7 cells are generally thought to play an important role in host defense against infection, by recruiting neutrophils and macrophages to infected tissues.
[0133] In some aspects, incorporating an adjuvant to an EV (e.g., exosome) can help drive an immune response towards a more cellular immune response (e.g., T-cell mediated). In some aspects, incorporating an adjuvant to an EV (e.g., exosome) can help drive an immune response towards a more humoral immune response (e.g., antibody -mediated).
[0134] In some aspects, an adjuvant induces the activation of a cytosolic pattern recognition receptor. Non-limiting examples of cytosolic pattern recognition receptor includes: stimulator of interferon genes (STING), retinoic acid-inducible gene I (RIG-1), Melanoma Differentiation-Associated protein 5 (MDA5), Nucleotide-binding oligomerization domain, Leucine rich Repeat and Pyrin domain containing (NLRP), inflammasomes, or combinations thereof. In certain aspects, an adjuvant is a STING agonist.
Stimulator of Interferon Genes (STING) is a cytosolic sensor of cyclic dinucleotides that is typically produced by bacteria. Upon activation, it leads to the production of type I interferons (e.g., IFN-a (alpha), IFN-J3 (beta), IFN-K (kappa), IFN-5 (delta), IFN-a (epsilon), IFN-r (tau), IFN-co (omega), and IFN- (zeta, also known as limitin)) and initiates an immune response. In certain aspects, the STING agonist comprises a cyclic dinucleotide STING agonist or a non-cyclic dinucleotide STING agonist. As described herein, in some aspects, the STING agonist is loaded in the lumen of the EV (e.g., exosome). In some aspects, such EVs (e.g., exosomes) are referred to herein as "exoSTING." Non-limiting examples of exoSTING are provided in International Publication No. WO 2019183578A1, which is herein incorporated by reference in its entirety. Further disclosures of useful STING agonists are also provided throughout the present disclosure.
[0135] Cyclic purine dinucleotides such as, but not limited to, cGMP, cyclic di-GMP (c-di-GMP), cAMP, cyclic di-AMP (c-di-AMP), cyclic-GMP-AMP (cGAMP), cyclic di-IMP (c-di-IMP), cyclic AMP- IMP (cAIMP), and any analogue thereof, are known to stimulate or enhance an immune or inflammation response in a patient. The CDNs can have 2'2', 2'3', 2'5', 3'3', or 3'5' bonds linking the cyclic dinucleotides, or any combination thereof.
[0136] Cyclic purine dinucleotides can be modified via standard organic chemistry techniques to produce analogues of purine dinucleotides. Suitable purine dinucleotides include, but are not limited to, adenine, guanine, inosine, hypoxanthine, xanthine, isoguanine, or any other appropriate purine dinucleotide known in the art. The cyclic dinucleotides can be modified analogues. Any suitable modification known in the art can be used, including, but not limited to, phosphorothioate, biphosphorothioate, fluorinate, and difluorinate modifications.
[0137] Non cyclic dinucleotide agonists can also be used, such as 5,6-Dimethylxanthenone-4-acetic acid (DMXAA), or any other non-cyclic dinucleotide agonist known in the art.
[0138] Non-limiting examples of STING agonists that can be used with the present disclosure include: DMXAA, STING agonist- 1, ML RR-S2 CD A, ML RR-S2c-di-GMP, ML-RR-S2 cGAMP, 2'3'-c- di-AM(PS)2, 2'3'-cGAMP, 2'3'-cGAMPdFHS, 3'3'-cGAMP, 3'3'-cGAMPdFSH, cAIMP, cAIM(PS)2, 3'3'- cAIMP, 3'3'-cAIMPdFSH, 2'2'-cGAMP, 2'3'-cGAM(PS)2, 3'3'-cGAMP, and combinations thereof. Nonlimiting examples of the STING agonists can be found at US Patent No. 9,695,212, WO 2014/189805 Al, WO 2014/179335 Al, WO 2018/100558 Al, US Patent No. 10,011,630 B2, WO 2017/027646 Al, WO 2017/161349 Al, and WO 2016/096174 Al, each of which is incorporated by reference in its entirety.
[0139] In some aspects, the STING agonist useful for the present disclosure comprises the compound or a pharmaceutically acceptable salt thereof. See WO 2016/096174 Al, which is incorporated herein by reference in its entirety.
[0140] In some aspects, the STING agonist useful for the present disclosure comprises a compound described in WO 2014/093936, WO 2014/189805, WO 2015/077354, Cell reports 11, 1018-1030 (2015),
WO 2013/185052, Sci. Transl. Med. 283,283ra52 (2015), WO 2014/189806, WO 2015/185565, WO 2014/179760, WO 2014/179335, WO 2015/017652, WO 2016/096577, WO 2016/120305, WO
2016/145102, WO 2017/027646, WO 2017/075477, WO 2017/027645, WO 2018/100558, WO
2017/175147, and/or WO 2017/175156, the contents of which are incorporated herein by reference in their entireties.
[0141] In some aspects, the STING agonist useful for the present disclosure is CL606, CL611, CL602, CL655, CL604, CL609, CL614, CL656, CL647, CL626, CL629, CL603, CL632, CL633, CL659, or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL606 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL611 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL602 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL655 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL604 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL609 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL614 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL656 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL647 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL626 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL629 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL603 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL632 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL633 or a pharmaceutically acceptable salt thereof. In some aspects, the STING agonist useful for the present disclosure is CL659 or a pharmaceutically acceptable salt thereof.
[0142] In some aspects, STING agonists can be modified to allow for better expression of the agonists on the surface of the EV (e.g. , exterior and/or luminal surface of the EV, e.g. , exosome, (e.g. , linked to a scaffold moiety disclosed herein, e.g., Scaffold X and/or Scaffold Y)). Any of the modifications described above can be used.
[0143] In some aspects, an adjuvant is a TLR agonist. Non-limiting examples of TLR agonists include: TLR2 agonist (e.g., lipoteichoic acid, atypical LPS, MALP-2 and MALP-404, OspA, porin, LcrV, lipomannan, GPI anchor, lysophosphatidylserine, lipophosphoglycan (LPG), glycophosphatidylinositol (GPI), zymosan, hsp60, gH/gL glycoprotein, hemagglutinin), a TLR3 agonist (e.g., double-stranded RNA, e.g., poly(EC), ampligen, hiltonol, polyA:U), a TLR4 agonist (e.g., lipopolysaccharides (LPS), lipoteichoic
acid, P-defensin 2, fibronectin EDA, HMGB1, snapin, tenascin C, MPLA), a TLR5 agonist (e.g., flagellin), a TLR6 agonist, a TLR7/8 agonist (e.g., single-stranded RNA, Poly GIO, Poly G3, Resiquimod (R848), Imiquimod, 3M-052), a TLR9 agonist (e.g., unmethylated CpG DNA), and combinations thereof. In some aspects, adjuvant comprises a saponin-based agent, e.g., Quil-A. Non-limiting examples of TLR agonists can be found at WO2008115319A2, US20130202707A1, US20120219615A1, US20100029585A1, W02009030996A1, W02009088401A2, and WO2011044246A1, each of which are incorporated by reference in its entirety. In some aspects, the adjuvant is a TLR4 agonist. In some aspects, the TLR4 agonist comprises MPLA. In some aspects, the adjuvant is a TLR7/8 agonist. In some aspects, the TLR7/8 agonist comprises Resiquimod (R848). In some aspects, the TLR7/8 agonist comprises 3M-052.
II. E, Additional Moi eties of Interest
[0144] In some aspects, a composition comprising an EV (e.g., exosome) described herein (i.e., comprising a KRAS antigen, e.g., in combination with an adjuvant and/or T helper peptide) can comprise one or more additional moieties of interest. As described herein, the additional moiety of interest can be associated with the EV (e.g. , exosome), such that the additional moiety of interest is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. In certain aspects, the additional moiety of interest is not associated with the EV. As will be apparent to those skilled in the art, the relevant disclosures provided below can equally apply to additional moieties of interest that are associated with the EV (e.g., exosomes) and those that are not.
[0145] In some aspects, the additional moieties of interest can further enhance the therapeutic effects of the EVs (e.g., exosomes), e.g., when administered to a subject. For instance, the additional moiety of interest can help direct EV uptake (e.g., targeting moiety), activate, or block cellular pathways to enhance the combinatorial effects associated with the EVs (e.g., exosomes), e.g., when administered to a subject.
[0146] In some aspects, EVs (e.g., exosomes) described herein comprises a single additional moiety of interest (e.g., targeting moiety or immune modulator). In some aspects, an EV (e.g., exosome) disclosed herein comprises multiple additional moieties of interest (e.g. , at least about 2, at least about 3, at least about 4, at least about 5, at least about 6, at least about 7, at least about 8, at least about 9, or at least about 10) (e.g. , both a targeting moiety and an immune modulator). In certain aspects, each of the multiple adjuvants is different. In some aspects, some or all of the multiple adjuvants are the same.
[0147] As described herein, in some aspects, an adjuvant can be encapsulated within the lumen of the EV. In some aspects, the adjuvant is linked to the exterior surface and/or luminal surface of the EVs (e.g. , exosomes) using a scaffold moiety (e.g. , Scaffold X and/or Scaffold Y). In some aspects, the adjuvant can be directly linked (i. e. , without the use of a scaffold moiety) to the exterior surface and/or luminal surface of EVs (e.g., exosomes) by various methods, including, but not limited to, anchoring moieties, affinity agents, chemical conjugation (e.g., click chemistry), or combinations thereof. Additional disclosure relating to such approaches (including modifications that can be made to the adjuvants and or EVs for
improved binding) to linking the adjuvants to the exterior surface and/or luminal surface of the EVs are provided elsewhere in the present disclosure (see, e.g., section II. B).
[0148] In some aspects, the additional moiety of interest comprises a targeting moiety that can modify the distribution of the EVs in vivo or in vitro. In some aspects, the targeting moiety can be a biological molecule, such as a protein, a peptide, a lipid, or a synthetic molecule.
[0149] In some aspects, a targeting moiety (e.g., tropism moiety) of the present disclosure specifically binds to a marker (or target molecule) expressed on a cell or a population of cells. In certain aspects, the marker is expressed on multiple cell types, e.g. , all antigen-presenting cells (e.g. , dendritic cells, macrophages, and B lymphocytes). In some aspects, the marker is expressed only on a specific population of cells (e.g., dendritic cells).
[0150] In some aspects, a targeting moiety of the present disclosure specifically binds to a marker for a dendritic cell. In certain aspects, the marker is expressed only on dendritic cells. In some aspects, dendritic cells comprise a progenitor (Pre) dendritic cells, inflammatory mono dendritic cells, plasmacytoid dendritic cell (pDC), a myeloid/conventional dendritic cell 1 (cDCl), a myeloid/conventional dendritic cell 2 (cDC2), inflammatory monocyte derived dendritic cells, Langerhans cells, dermal dendritic cells, lysozyme-expressing dendritic cells (LysoDCs), Kupffer cells, nonclassical monocytes, or any combination thereof. Markers that are expressed on these dendritic cells are known in the art. See, e.g., Collin et al., Immunology 154(l):3-20 (2018).
[0151] In some aspects, the targeting moiety (e.g., tropism moiety) is a protein, wherein the protein is an antibody or a fragment thereof that can specifically bind to a marker selected from lymphocyte antigen 75 (DEC205), C-type lectin domain family 9 member A (CLEC9A), C-type lectin domain family 6 (CLEC6), ), C-type lectin domain family 4 member A (also known as DCIR or CLEC4A), Dendritic Cell- Specific Intercellular adhesion molecule -3 -Grabbing Non-integrin (also known as DC-SIGN or CD209), lectin-type oxidized LDL receptor 1 (LOX-1), macrophage receptor with collagenous structure (MARCO), C-type lectin domain family 12 member A (CLEC12A), C-type lectin domain family 10 member A (CLEC10A), DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin- 1, macrophage mannose receptor (MMR), BDCA-2 (CD303, CLEC4C), Dectin-2, Bst-2 (CD317), Langerin, CD206, CDl lb, CDl lc, CD103, CD123, CD304, XCR1, AXL, Siglec 6, CD209, SIRPA, CX3CR1, GPR182, CD14, CD16, CD32, CD34, CD38, CD10, or any combination thereof.
[0152] In some aspects, a targeting moiety useful for the present disclosure is capable of binding a marker expressed on CD8+ dendritic cells and thereby, the target an EV (e.g., exosome) to the CD8+ dendritic cells for uptake. In certain aspects, the targeting moiety binds to Clec9A, XCR1, CD 103, or combinations thereof.
[0153] As described supra, a targeting moiety disclosed herein can comprise a peptide, an antibody or an antigen binding fragment thereof, a chemical compound, or any combination thereof.
[0154] In some aspects, the targeting moiety is an antibody or an antigen binding fragment thereof. In certain aspects, a targeting moiety is a single-chain Fv antibody fragment. In certain aspects, a targeting moiety is a single-chain F(ab) antibody fragment. In certain aspects, a targeting moiety is a nanobody. In certain aspects, a targeting moiety is a monobody.
[0155] In some aspects, an EV, e.g., exosome, disclosed herein can be surface engineered to adjust its properties, e.g., biodistribution, e.g., via incorporation of immuno-affinity ligands or cognate receptor ligands. For example, EVs (e.g., exosomes) described herein can be engineered to direct the EVs to a particular tissue within a subject. For instance, in cancer subjects, the EVs (e.g., exosomes) comprising a KRAS antigen can be modified to specifically target tissues where T cell priming would likely occur. Nonlimiting examples of such tissues include: tumor draining lymph nodes, sentinel lymph nodes, tumor tertiary lymph nodes, or any combination thereof. Not to be bound by any one theory, by modifying the EVs (e.g., exosomes) to specifically target such tissues, an immune response against the KRAS antigen could be increased compared to a corresponding EV (e.g., exosome) without the targeting/tropism moiety disclosed herein. Non-limiting examples of targeting moieties that could be used include: anti-fibronectin EDA & EDB domain antibodies; anti-ICAM-1; anti-CD20; anti-DC-LAMP; or any combination thereof.
[0156] EVs, e.g., exosomes, exhibit preferential uptake in discrete cell types and tissues, and their tropism can be directed by adding proteins to their surface that interact with receptors on the surface of target cells.
[0157] In some aspects, a tropism moiety can increase uptake of the EV, e.g., an exosome, by a cell. In some aspects, the tropism moiety that can increase uptake of the EV, e.g., an exosome, by a cell comprises a lymphocyte antigen 75 (also known as DEC205 or CD205), C-type lectin domain family 9 member A (CLEC9A), C-type lectin domain family 6 (CLEC6), C-type lectin domain family 4 member A (also known as DCIR or CLEC4A), Dendritic Cell-Specific Intercellular adhesion molecule-3-Grabbing Non-integrin (also known as DC-SIGN or CD209), lectin-type oxidized LDL receptor l(LOX-l), macrophage receptor with collagenous structure (MARCO), C-type lectin domain family 12 member A (CLEC12A), C-type lectin domain family 10 member A (CLEC10A), DC-asialoglycoprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin- 1, macrophage mannose receptor (MMR), BDCA-2 (CD303, CLEC4C), Dectin-2, BST-2 (CD317), Langerin, CD206, CDl lb, CDl lc, CD123, CD304, XCR1, AXL, SIGLEC 6, CD209, SIRPA, CX3CR1, GPR182, CD14, CD16, CD32, CD34, CD38, CD10, anti-CD3 antibody, or any combination thereof.
[0158] In some aspects, when tropism to the central nervous system is desired, an EV, e.g., exosome, of the present disclosure can comprise a tissue or cell-specific target ligand, which increases EV, e.g., exosome, tropism to a specific central nervous system tissue or cell. In some aspects, the cell is a glial cell. In some aspects, the glial cell is an oligodendrocyte, an astrocyte, an ependymal cell, a microglia cell, a Schwann cell, a satellite glial cell, an olfactory ensheathing cell, or a combination thereof. In some aspects,
the cell is a neural stem cell. In some aspects, the cell-specific target ligand, which increases EV, e.g., exosome, tropism to a Schwann cells, binds to a Schwann cell surface marker, such as Myelin Basic Protein (MBP), Myelin Protein Zero (P0), P75NTR, NCAM, PMP22, or any combination thereof. In some aspects, the cell-specific tropism moiety comprises an antibody or an antigen-binding portion thereof, an aptamer, or an agonist or antagonist of a receptor expressed on the surface of the Schwann cell.
[0159] In certain aspects, the tropism moiety is linked, e.g., chemically linked via a maleimide moiety, to a scaffold moiety, e.g., a Scaffold X protein or a fragment thereof, on the exterior surface of the EV, e.g., exosome. Tropism can be further improved by the attachment of a half-life extension moiety (e.g., albumin or PEG), or any combination thereof to the external surface of an EV, e.g., exosome of the present disclosure.
[0160] Pharmacokinetics, biodistribution, and in particular, tropism and retention in the desired tissue or anatomical location can also be accomplish by selecting the appropriate administration route (e.g., intrathecal administration or intraocular administration to improve tropism to the central nervous system).
[0161] Surface antigens useful in the present disclosure include, but are not limited to, antigens that label a cell as a "self cell. In some aspects, the surface antigen is selected from CD47, CD24, a fragment thereof, and any combination thereof. In certain aspects, the surface antigen comprises CD24, e.g., human CD24. In some aspects, the surface antigen comprises a fragment of CD24, e.g., human CD24. In certain aspects, the EV, e.g., exosome, is modified to express CD47 or a fragment thereof on the exterior surface of the EV, e.g., exosome.
[0162] In some aspects, the additional moiety of interest comprises an immune modulator (e.g., along with an antigen, adjuvant, and/or additional moiety of interest described herein). Accordingly, in certain aspects, a base EV (e.g. , exosome) that can be used to produce or manufacture an EV-based vaccine described herein comprises the immune modulator (e.g., alone or in combination with the adjuvant and/or additional moiety of interest described herein), such that the immune modulator is present in the EV prior to the addition of the antigen. For instance, in some aspects, the immune modulator can be introduced into a producer cell when producing the base EVs (e.g., exosomes). In some aspects, the immune modulator can be added to the EVs (e.g., exosomes) after being isolated from the producer cells. In such aspects, the immune modulator can be added to the isolated EVs (e.g., exosomes) before adding the antigen. In some aspects, the immune modulator is added to the EV after adding the antigen. In some aspects, the immune modulator is added to the EV together with the antigen.
[0163] In some aspects, an immune modualtor comprises an inhibitor for a negative checkpoint regulator or an inhibitor for a binding partner of a negative checkpoint regulator. In certain aspects, the negative checkpoint regulator comprises cytotoxic T-lymphocyte-associated protein 4 (CTLA-4), programmed cell death protein 1 (PD-1), lymphocyte -activated gene 3 (LAG-3), T-cell immunoglobulin mucin-containing protein 3 (TIM-3), B and T lymphocyte attenuator (BTLA), T cell immunoreceptor with
Ig and ITIM domains (TIGIT), V-domain Ig suppressor of T cell activation (VISTA), adenosine A2a receptor (A2aR), killer cell immunoglobulin like receptor (KIR), indoleamine 2,3 -dioxygenase (IDO), CD20, CD39, CD73, or any combination thereof.
[0164] In some aspects, the immunomodulating component is a protein, a peptide, a glycolipid, or a glycoprotein.
ILF, Anchoring moieties (AM)
[0165] As described herein, in some aspects, an antigen (KRAS), adjuvant, and/or one or more additional moieties of interest (e.g. , targeting moiety and/or immune modulator) can be linked to an exterior surface and/or luminal surface of the EVs using an anchoring moiety or any other coupling strategy described herein or known in the art. In some aspects, the coupling strategy comprises: an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), split intein, SpyTag/SpyCatcher, ALFA-tag, Streptavidin/Avitag, Sortase, SNAP-tag, ProA/Fc-binding peptide, or any combinations thereof. In some aspects, the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) is linked directly to the anchoring moiety or via a linker. In some aspects, the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) can be attached to an anchoring moiety or linker combination via reaction between a "reactive group" (RG; e.g., amine, thiol, hydroxy, carboxylic acid, or azide) with a "reactive moiety" (RM; e.g., maleimide, succinate, NHS).
[0166] In some aspect, an anchoring moiety can be chemically conjugated to the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) to enhance its hydrophobic character. Non-limiting examples of such anchoring moieties are provided further below.
[0167] As described elsewhere in the present disclosure, in some aspects, the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) are modified, such that they include a free amine group at the N-terminus (e.g. , comprising a N-terminal lysine). In some aspects, the anchoring moiety is conjugated to the free amine group at the N-terminus either directly or via one or more linkers.
III.A.1. Exemplary Anchoring moieties
[0168] In some aspects, anchoring moieties that can be used to link an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) to the exterior surface and/or luminal surface of the EV (e.g. , exosome) comprises: a sterol (e.g., cholesterol), GM1, a lipid (e.g., fatty acid (e.g., palmitate), ionizable lipid, glycerophospholipid, sphingolipid), a vitamin (e.g., tocopherol (e.g., vitamin E)), vitamin A, vitamin D, vitamin K), a small molecule, a peptide (e.g., cell penetrating peptide), including any derivatives thereof, or a combination thereof.
[0169] In some aspects, the anchoring moiety is a lipid. A lipid anchoring moiety can be any lipid known in the art, e.g., palmitic acid or glycosylphosphatidylinositols. Non-limiting examples of other suitable lipids are described in Fahy et al., Biochim Biophys Acta 1811(11): 637-647 (Nov. 2011), which is incorporated herein by reference in its entirety. In some aspects, the lipid is a fatty acid, phosphatide, phospholipid (e.g., phosphatidyl choline, phosphatidyl serine, or phosphatidyl ethanolamine), or analogue thereof (e.g. phophatidylcholine, lecithin, phosphatidylethanolamine, cephalin, or phosphatidylserine or analogue or portion thereof, such as a partially hydrolyzed portion thereof). In some aspects, the lipid comprises an ionizable lipid. In some aspects, the lipid comprises a glycerophospholipid. In some aspects, the lipid comprises a sphingolipid.
[0170] Generally, anchoring moieties are chemically attached. However, an anchoring moiety can be attached to an an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator) enzymatically.
[0171] Some types of membrane anchors that can be used to practice the methods of the present disclosure are presented in the following table:
[0172] In some aspects, an anchoring moiety of the present disclosure can comprise two or more types of anchoring moieties disclosed herein. For example, in some aspects, an anchoring moiety can comprise two lipids, e.g., a phospholipids and a fatty acid, or two phospholipids, or two fatty acids, or a lipid and a vitamin, or cholesterol and a vitamin.
[0173] In some aspects, the anchoring moiety useful for the present disclosure comprises a sterol, steroid, hopanoid, hydroxysteroid, secosteroid, or analog thereof with lipophilic properties. In some aspects, the anchoring moiety comprises a sterol, such as a phytosterol, mycosterol, or zoosterol. Exemplary
zoosterols include cholesterol and 24S-hydroxycholesterol; exemplary phytosterols include ergosterol (mycosterol), campesterol, sitosterol, and stigmasterol. In some aspects, the sterol is selected from ergosterol, 7-dehydrocholesterol, cholesterol, 24S-hydroxycholesterol, lanosterol, cycloartenol, fucosterol, saringosterol, campesterol, [3-sitosterol, sitostanol, coprostanol, avenasterol, or stigmasterol. Sterols can be found either as free sterols, acylated (sterol esters), alkylated (steryl alkyl ethers), sulfated (sterol sulfate), or linked to a glycoside moiety (steryl glycosides), which can be itself acylated (acylated sterol glycosides). In some aspects, the anchoring moiety is a cholesterol.
[0174] In some aspects, the anchoring moiety comprises a steroid. In some aspects, the steroid is selected from dihydrotestosterone, uvaol, hecigenin, diosgenin, progesterone, or cortisol.
[0175] In some aspects, the anchoring moiety is a fatty acid. In some aspects, the fatty acid is a short-chain, medium -chain, or long-chain fatty acid. In some aspects, the fatty acid is a saturated fatty acid. In some aspects, the fatty acid is an unsaturated fatty acid. In some aspects, the fatty acid is a monounsaturated fatty acid. In some aspects, the fatty acid is a polyunsaturated fatty acid, such as an omega- 3 or omega-6 fatty acid.
[0176] In some aspects, the anchoring moiety comprises a phospholipid. Phospholipids are a class of lipids that are a major component of all cell membranes. They can form lipid bilayers because of their amphiphilic characteristic. The structure of the phospholipid molecule generally consists of two hydrophobic fatty acid "tails" and a hydrophilic "head" consisting of a phosphate group. For example, a phospholipid can be a lipid according to the following formula:
in which Rp represents a phospholipid moiety and Ri and R2 represent fatty acid moieties with or without unsaturation that can be the same or different.
[0177] In some aspects, an antigen, adjuvant, and/or one or more additional moieties of interest
(e.g., targeting moiety and/or immune modulator) is linked to an anchoring moiety disclosed herein via a linker combination, which can comprise any combination of cleavable and/or non-cleavable linkers. Accordingly, in some aspects, an EV described herein can comprise multiple coupling strategies described herein (e.g., a cell penetrating peptide and a linker). In some aspects, the coupling strategy comprises: an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), split intein, SpyTag/SpyCatcher, ALFA-tag, Streptavidin/Avitag, Sortase, SNAP -tag, ProA/Fc-binding peptide, or any combinations thereof. Not to be bound by any one theory, one of the functions of a linker combination is to
provide the optimal spacing between the anchoring moiety and the antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator).
[0178] Linkers can be susceptible to cleavage ("cleavable linker") thereby facilitating release of the biologically active molecule. Thus, in some aspects, a linker combination disclosed herein can comprise a cleavable linker. Such cleavable linkers can be susceptible, for example, to acid-induced cleavage, photoinduced cleavage, peptidase -induced cleavage, esterase-induced cleavage, and disulfide bond cleavage, at conditions under which the biologically active molecule remains active. Alternatively, linkers can be substantially resistant to cleavage ("non-cleavable linker"). In some aspects, the cleavable linker comprises a spacer. In some aspects the spacer is PEG.
[0179] In some aspects, a linker combination comprises at least 2, at least 3, at least 4, at least 5, or at least 6 or more different linkers disclosed herein. In some aspects, linkers in a linker combination can be linked by an ester linkage (e.g., phosphodiester or phosphorothioate ester).
[0180] In some aspects, the linker is direct bond between an anchoring moiety and an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator).
[0181] In some aspects, the linker combination comprises a "non-cleavable liker." Non-cleavable linkers are any chemical moiety capable of linking two or more components of an EV (e.g., exosome) disclosed herein, such as an an antigen, adjuvant, and/or one or more additional moieties of interest (e.g., targeting moiety and/or immune modulator), in a stable, covalent manner and does not fall off under the categories listed above for cleavable linkers. Thus, non-cleavable linkers are substantially resistant to acid- induced cleavage, photo-induced cleavage, peptidase-induced cleavage, esterase-induced cleavage and disulfide bond cleavage.
[0182] Furthermore, non-cleavable refers to the ability of the chemical bond in the linker or adjoining to the linker to withstand cleavage induced by an acid, photolabile -cleaving agent, a peptidase, an esterase, or a chemical or physiological compound that cleaves a disulfide bond, at conditions under which a cyclic dinucleotide and/or the antibody does not lose its activity. In some aspects, the biologically active molecule is attached to the linker via another linker, e.g., a self-immolative linker.
[0183] In some aspects, the linker combination comprises a non-cleavable linker comprising, e.g., tetraethylene glycol (TEG), hexaethylene glycol (HEG), polyethylene glycol (PEG), succinimide, thiosuccinimide, non-peptidyl amide, or any combination thereof. In some aspects, the non-cleavable linker comprises a spacer unit to link the biologically active molecule to the non-cleavable linker.
[0184] In some aspects, one or more non-cleavable linkers comprise smaller units (e.g., HEG, TEG, glycerol, C2 to C12 alkyl, and the like) linked together. In one aspect, the linkage is an ester linkage (e.g., phosphodiester or phosphorothioate ester) or other linkage.
[0185] In some aspects, the linker combination comprises a non-cleavable linker, wherein the non- cleavable linker comprises a polyethylene glycol (PEG) characterized by a formula R3-(O-CH2-CH2)n- or R3-(0-CH2-CH2)n-O- with R3 being hydrogen, methyl or ethyl and n having a value from 2 to 200. In some aspects, the linker comprises a spacer, wherein the spacer is PEG.
[0186] In some aspects, the PEG linker is an oligo-ethylene glycol, e.g., diethylene glycol, triethylene glycol, tetra ethylene glycol (TEG), pentaethylene glycol, or a hexaethylene glycol (HEG) linker. [0187] In some aspects, n has a value of 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17,18,19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 121, 122, 123, 124,
125, 126, 127, 128, 129, 130, 131, 132, 133, 134, 135, 136, 137, 138, 139, 140, 141, 142, 143, 144, 145,
146, 147, 148, 149, 150, 151, 152, 153, 154, 155, 156, 157, 158, 159, 160, 161, 162, 163, 164, 165, 166,
167, 168, 169, 170, 171, 172, 173, 174, 175, 176, 177, 178, 179, 189, 181, 182, 183, 184, 185, 186, 187,
188, 189, 190, 191, 192, 193, 194, 195, 196, 197, 198, 199, or 200.
[0188] In some aspects, n is between 2 and 10, between 10 and 20, between 20 and 30, between 30 and 40, between 40 and 50, between 50 and 60, between 60 and 70, between 70 and 80, between 80 and 90, between 90 and 100, between 100 and 110, between 110 and 120, between 120 and 130, between 130 and 140, between 140 and 150, between 150 and 160, between 160 and 170, between 170 and 180, between 180 and 190, or between 190 and 200.
[0189] In some specific aspects, n has a value from 3 to 200, from 3 to 20, from 10 to 30, or from 9 to 45.
[0190] In some aspects, the PEG is a branched PEG. Branched PEGs have three to ten PEG chains emanating from a central core group.
[0191] In some aspects, the PEG moiety is a monodisperse polyethylene glycol. In the context of the present disclosure, a monodisperse polyethylene glycol (mdPEG) is a PEG that has a single, defined chain length and molecular weight. mdPEGs are typically generated by separation from the polymerization mixture by chromatography. In certain formulae, a monodisperse PEG moiety is assigned the abbreviation mdPEG.
[0192] In some aspects, the PEG is a Star PEG. Star PEGs have 10 to 100 PEG chains emanating from a central core group.
[0193] In some aspects, the PEG is a Comb PEGs. Comb PEGs have multiple PEG chains normally grafted onto a polymer backbone.
[0194] In certain aspects, the PEG has a molar mass between 100 g/mol and 3000 g/mol, particularly between 100 g/mol and 2500 g/mol, more particularly of approx. 100 g/mol to 2000 g/mol. In certain
aspects, the PEG has a molar mass between 200 g/mol and 3000 g/mol, particularly between 300 g/mol and 2500 g/mol, more particularly of approx. 400 g/mol to 2000 g/mol.
[0195] In some aspects, the PEG is PEGwo, PEG200, PEG300, PEG400, PEG500, PEGgoo, PEG700, PEGsoo, PEG900, PEG1000, PEGnoo, PEG1200, PEG1300, PEG1400, PEG1500, PEGigoo, PEG1700, PEGisoo, PEG1900, PEG2000, PEG2100, PEG2200, PEG2300, PEG2400, PEG2500, PEGigoo, PEG1700, PEGigoo, PEG1900, PEG2000, PEG2100, PEG2200, PEG2300, PEG2400, PEG2500, PEG2600, PEG2700, PEG2800, PEG2900, 0rPEG3ooo. In one particular aspect, the PEG is PEG400- In another particular aspect, the PEG is PEG2ooo-
[0196] In some aspects, a linker combination of the present disclosure can comprise several PEG linkers, e.g., a cleavable linker flanked by PEG, HEG, or TEG linkers.
[0197] In some aspects, the linker combination comprises (HEG)n and/or (TEG)n, wherein n is an integer between 1 and 50, and each unit is connected, e.g., via a phosphate ester linker, a phosphorothioate ester linkage, or a combination thereof.
[0198] In some aspects, the linker combination comprises a non-cleavable linker comprising a glycerol unit or a polyglycerol (PG) described by the formula ((R3 — O — (CH2 — CHOH — CEEC n — ) with R3 being hydrogen, methyl or ethyl, and n having a value from 3 to 200. In some aspects, n has a value from 3 to 20. In some aspects, n has a value from 10 to 30.
[0199] In some aspects, the PG linker is a diglycerol, triglycerol, tetraglycerol (TG), pentaglycerol, or a hexaglycerol (HG) linker.
[0200] In some aspects, the linker combination comprises (glycerol)n, and/or (HG)n and/or (TG)n, wherein n is an integer between 1 and 50, and each unit is connected, e.g., via a phosphate ester linker, a phosphorothioate ester linkage, or a combination thereof.
[0201] In some aspects, the linker combination comprises at least one aliphatic (alkyl) linker, e.g., propyl, butyl, hexyl , or C2-C12 alkyl, such as C2-C10 alkyl or C2-C6 alkyl.
[0202] In some aspects, different components of an EV (e.g. , exosome) disclosed herein (e.g. , an antigen, adjuvant, and/or additional moiety of interest (e.g., targeting moiety and/or immune modulator)) can be linker by a cleavable linker. The term cleavable linker refers to a linker comprising at least one linkage or chemical bond that can be broken or cleaved. As used herein, the term cleave refers to the breaking of one or more chemical bonds in a relatively large molecule in a manner that produces two or more relatively smaller molecules. Cleavage can be mediated, e.g., by a nuclease, peptidase, protease, phosphatase, oxidase, or reductase, for example, or by specific physicochemical conditions, e.g., redox environment, pH, presence of reactive oxygen species, or specific wavelengths of light.
[0203] In some aspects, the term "cleavable," as used herein, refers, e.g., to rapidly degradable linkers, such as, e.g., phosphodiester and disulfides, while the term "non-cleavable" refers, e.g., to more stable linkages, such as, e.g., nuclease-resistant phosphorothioates.
[0204] In some aspects, the cleavable linker is a dinucleotide, trinucleotide, tetranucleotide linker, a disulfide, an imine, a silyl ether, carbonate, a thioketal, a val-cit dipeptide, Vai-Ala dipeptide, Ala-Ala-Asn tripeptide, poly-arginine, phosphodiesters, acid-labile, or any combination thereof.
[0205] In some aspects, the cleavable linker comprises valine-alanine-p-aminobenzylcarbamate, valine-citrulline-p-aminobenzylcarbamate, Ala-Ala-Asn-p-aminobenzylcarbamate.
[0206] In some aspects, the linker combination comprises a redox cleavable linker. As a nonlimiting example, one type of cleavable linker is a redox cleavable linking group that is cleaved upon reduction or upon oxidation.
[0207] In some aspects, the redox cleavable linker contains a disulfide bond, i.e., it is a disulfide cleavable linker.
[0208] Redox cleavable linkers can be reduced, e.g., by intracellular mercaptans, oxidases, or reductases.
[0209] In some aspects, the linker combination can comprise a cleavable linker which can be cleaved by a reactive oxygen species (ROS), such as superoxide (Of) or hydrogen peroxide (H2O2), generated, e.g., by inflammation processes such as activated neutrophils. In some aspects, the ROS cleavable linker is a thioketal cleavable linker. See, e.g., U.S. Pat. 8,354,455B2, which is herein incorporated by reference in its entirety.
[0210] In some aspects, the linker is an "acid labile linker" comprising an acid cleavable linking group, which is a linking group that is selectively cleaved under acidic conditions (pH<7).
[0211] As a non-limiting example, the acid cleavable linking group is cleaved in an acidic environment, e.g., about 6.0, 5.5, 5.0 or less. In some aspects, the pH is about 6.5 or less. In some aspects, the linker is cleaved by an agent such as an enzyme that can act as a general acid, e.g., a peptidase (which can be substrate specific) or a phosphatase. Within cells, certain low pH organelles, such as endosomes and lysosomes, can provide a cleaving environment to the acid cleavable linking group. Although the pH of human serum is 7.4, the average pH in cells is slightly lower, ranging from about 7.1 to 7.3. Endosomes also have an acidic pH, ranging from 5.5 to 6.0, and lysosomes are about 5.0 at an even more acidic pH. Accordingly, pH dependent cleavable linkers are sometimes called endosomically labile linkers in the art.
[0212] In some aspects, the linker comprises a low pH-labile hydrazone bond, silyl ether, carbonate, or combinations thereof. Such acid-labile bonds have been extensively used in the field of conjugates, e.g., antibody-drug conjugates. See, for example, Zhou et al, Biomacromolecules 2011, 12, 1460-7; Yuan et al, Acta Biomater. 2008, 4, 1024-37; Zhang et al, Acta Biomater. 2007, 6, 838-50; Yang et al, J. Pharmacol. Exp. Ther. 2007, 321, 462-8; Reddy et al, Cancer Chemother. Pharmacol. 2006, 58, 229-36; Doronina et al, Nature Biotechnol. 2003, 21, 778-84.
[0213] Further examples can be found in U.S. Pat. Nos. 9,790,494B2 and 8,137,695B2, the contents of which are incorporated herein by reference in their entireties.
[0214] In some aspects, the linker combination can comprise a linker cleavable by intracellular or extracellular enzymes, e.g., proteases, esterases, nucleases, amidades, or reductase. The range of enzymes that can cleave a specific linker in a linker combination depends on the specific bonds and chemical structure of the linker. Accordingly, peptidic linkers can be cleaved, e.g., by peptidades, linkers containing ester linkages can be cleaved, e.g., by esterases; linkers containing amide linkages can be cleaved, e.g., by amidades; linkers containing disulfide linkages can be cleaved, e.g., by reductases; etc.
[0215] In some aspects, the linker combination comprises a protease cleavable linker, i.e., a linker that can be cleaved by an endogenous protease. Only certain peptides are readily cleaved inside or outside cells. See, e.g., Trout et al., 79 Proc. Natl. Acad. Sci. USA, 626-629 (1982) and Umemoto et al. 43 Int. J. Cancer, 677-684 (1989). Cleavable linkers can contain cleavable sites composed of a-amino acid units and peptidic bonds, which chemically are amide bonds between the carboxylate of one amino acid and the amino group of a second amino acid. Other amide bonds, such as the bond between a carboxylate and the [3-amino acid group of lysine, are understood not to be peptidic bonds and are considered non-cleavable.
[0216] Some linkers are cleaved by esterases ("esterase cleavable linkers"). Only certain esters can be cleaved by esterases and amidases present inside or outside of cells. Esters are formed by the condensation of a carboxylic acid and an alcohol. Simple esters are esters produced with simple alcohols, such as aliphatic alcohols, and small cyclic and small aromatic alcohols. Examples of ester-based cleavable linking groups include, but are not limited to, esters of alkylene, alkenylene and alkynylene groups. The ester cleavable linking group has the general formula -C (O) O- or -OC (O)-.
[0217] In some aspects, a linker combination can includes a phosphate-based cleavable linking group is cleaved by an agent that degrades or hydrolyzes phosphate groups. An example of an agent that cleaves intracellular phosphate groups is an enzyme such as intracellular phosphatase, nuclease, or phosphodiesterase .
[0218] In some aspects, the combination linker comprises a photoactivated cleavable linker, e.g., a nitrobenzyl linker or a linker comprising a nitrobenzyl reactive group.
[0219] In some aspects, the linker combination comprises a self-immolative linker In some aspects, the self-immolative linker in the EV (e.g., exosome) of the present disclosure undergoes 1,4 elimination after the enzymatic cleavage of the protease-cleavable linker. In some aspects, the self-immolative linker in the EV (e.g., exosome) of the present disclosure undergoes 1,6 elimination after the enzymatic cleavage of the protease-cleavable linker. In some aspects, the self-immolative linker is, e.g., a p-aminobenzyl (pAB) derivative, such as a p-aminobenzyl carbamate (pABC), a p-amino benzyl ether (PABE), a p-amino benzyl carbonate, or a combination thereof.
II.F Conjugated EVs (e.g., Exosomes)
[0220] Unlike antibodies, EVs (e.g., exosomes) can accommodate large numbers of molecules attached to their surface, e.g., on the order of thousands to tens of thousands of molecules per EV (e.g.,
exosome). EV (e.g., exosome)-drug conjugates thus represent a platform to deliver a high concentration of therapeutic compound to discrete cell types, while at the same time limiting overall systemic exposure to the compound, which in turn reduces off-target toxicity.
[0221] The present disclosure provide EVs, e.g., exosomes, that have been engineered by reacting a first molecular entity comprising a free thiol group with a second molecular entity comprising a maleimide group, wherein the maleimide moiety covalently links the first molecular entity with the second molecular entity via a maleimide moiety.
[0222] Non-limiting examples of biologically active molecules that can attached to an EV (e.g., exosome) via a maleimide moiety include agents such as, nucleotides (e.g., nucleotides comprising a detectable moiety or a toxin or that disrupt transcription), nucleic acids (e.g., DNA or mRNA molecules that encode a polypeptide such as an enzyme, or RNA molecules that have regulatory function such as miRNA, dsDNA, IncRNA, or siRNA), morpholino, amino acids (e.g., amino acids comprising a detectable moiety or a toxin that disrupt translation), polypeptides (e.g., enzymes), lipids, carbohydrates, small molecules (e.g., small molecule drugs and toxins), antigens (e.g., vaccine antigens), adjuvants (e.g., vaccine adjuvants), etc.
[0223] In some aspects, an EV (e.g., exosome) of the present disclosure can comprise more than one type of biologically active molecule. In some aspects, biologically active molecules can be, e.g., small molecules such as cyclic dinucleotides, toxins such as auristatins (e.g., monoethyl auristatin E, MMAE), antibodies (e.g., naked antibodies or antibody -drug conjugates), STING agonists, tolerizing agents, antisense oligonucleotides, PROTACs, morpholines, lysophosphatidic acid receptor antagonists (e.g., LPA1 antagonists) or any combinations thereof. In some aspects, an EV (e.g., exosome) of the present disclosure can comprise, e.g., a vaccine antigen and optionally a vaccine adjuvant. In some aspects, an EV (e.g., exosome) of the present disclosure can comprise a therapeutic payload (e.g., a STING or one payload disclosed below) and a targeting moiety and/or a tropism moiety.
[0224] EVs (e.g., exosomes) disclosed herein can comprise one or more affinity ligands that link or conjugate a molecule of interest (e.g. , antigen, adjuvant, immune modulator, and/or targeting moiety) to the EVs (e.g., to the exterior surface or on the luminal surface) or to a target cell. In some aspects, an affinity ligand disclosed herein has one or more of the following properties: (i) derived from a synthetic library, (ii) sub-nanomolar affinity for a scaffold moiety (e.g., Scaffold X) with emphasis on slow off rate, (iii) binds epitope on membrane-distal IgV domain of a scaffold moiety (e.g., Scaffold X), (iv) free of disulfide linkages, (v) free of N-linked glycosylation sites, (vi) less than 20 amino acids in length, (vii) monomeric, (viii) electroneutral at physiological pH, (ix) hydrophilic, (x) resistant to protease digestion, (xi) amenable to expression in prokaryotic and eukaryotic hosts, (xii) can accommodate N- or C-terminus fusion, (xiii) nonimmunogenic, (xiv) can contain a tag for purification and/or separation, e.g., of an EV, and (xv) combinations thereof. As described herein, in some aspects, an affinity ligand disclosed herein can
specifically bind (e.g. , with high affinity) to a moiety expressed on the surface of an EV (e.g. , exosome). In certain aspects, an affinity ligand specifically binds to a scaffold moiety expressed on the surface of an EV. In some aspects, an affinity ligand specifically binds to any moiety expressed on the surface of an EV (e.g., cholesterol). In some aspects, an affinity ligand disclosed herein can specifically bind (e.g., with high affinity) to a moiety expressed on a target cell. Non-limiting examples of such affinity ligands are provided throughout the present disclosure.
[0225] As described above, an affinity ligand useful for the present disclosure can be engineered to express one or more tags. In some aspects, such tags can be useful in the purification and/or separation of an agent that is conjugated to the affinity ligand. For example, in some aspects, an EV (e.g., exosome) comprises a scaffold moiety that is conjugated to an affinity ligand fusion, which comprises a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) and a tag. In such aspects, the tag can be used to purify and/or separate the EV from a sample comprising the EV. In some aspects, a tag of an affinity ligand fusion described above is present between the affinity ligand and the molecule of interest. In some aspects, the tag of an affinity ligand fusion described above can be present at an end (e.g., N-terminus) of the molecule of interest, as long as the tag does not interfere with the activity of the molecule of interest. Any tags useful in the art for purifying and/or separating an agent from a sample can be used in the present disclosure. Non-limiting examples of such tags include polyhistidine tags, polyarginine tags, glutathione-S-transferase (GST), maltose binding protein (MBP), S-tag, influenza virus HA tag, thioredoxin, staphylococcal protein A tag, FLAG™ epitope, AviTag epitope (for subsequent biotinylation), c-myc epitope, and combinations thereof. See, e.g., U.S. Pat. No. 7,655,413, which is herein incorporated by reference in its entirety.
[0226] As described herein, in some aspects, a molecule of interest can be expressed on the surface of an EV (e.g., exosome) via a scaffold moiety. In some of these aspects, the molecule of interest can be linked or conjugated to the scaffold moiety via an affinity ligand. For instance, as described herein, in certain aspects, an affinity ligand can be fused to a molecule of interest (e.g. , antigen, adjuvant, immune modulator, and/or targeting moiety), and then the molecule of interest can be conjugated to a moiety expressed on the surface of an EV (e.g., scaffold moiety) via the affinity ligand. In some aspects, the affinity ligand increases the binding of the molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to the moiety on the EV (e.g. , scaffold moiety). In certain aspects, the binding of the molecule of interest to the moiety on the EV (e.g., scaffold moiety) is increased by at least about one-fold, at least about two-fold, at least about three -fold, at least about four-fold, at least about five-fold, at least about six-fold, at least about seven-fold, at least about eight-fold, at least about nine-fold, at least about 10-fold, at least about 20- fold, at least about 30-fold, at least about 40-fold, at least about 50-fold, at least about 60-fold, at least about 70-fold, at least about 80-fold, at least about 90-fold, at least about 100-fold, at least about 200-fold, at least about 300-fold, at least about 400-fold, at least about 500-fold, at least about 600-fold, at least about 700-
fold, at least about 800-fold, at least about 900-fold, at least about 1,000-fold, at least about 2,000-fold, at least about 3,000-fold, at least about 4,000-fold, at least about 5,000-fold, at least about 6,000-fold, at least about 7,000-fold, at least about 8,000-fold, at least about 9,000-fold, at least about 10,000-fold or more, compared to a reference (e.g., binding of the molecule of interest to the moiety on the EV (e.g., scaffold moiety) without the use of an affinity ligand).
[0227] In some aspects, an affinity ligand that can be used with the present disclosure comprises a linear peptide. In certain aspects, an affinity ligand comprises at least about two, at least about three, at least about four, at least about five, at least about seven, at least about eight, at least about nine, at least about 10, at least about 11, at least about 12, at least about 13, at least about 14, at least about 15, at least about 16, at least about 17, at least about 18, at least about 19, at least about 20, at least about 25, at least about 30, at least about 35, at least about 40, at least about 45, at least about 50, at least about 55, at least about 60, at least about 65, at least about 70, at least about 75, at least about 80, at least about 85, at least about 90, at least about 95, or at least about 100 amino acids.
[0228] Not to be bound by any one theory, using an affinity ligand (e.g., disclosed herein) to link or conjugate a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to a moiety expressed on the surface of an EV (e.g. , scaffold moiety) can improve one or more properties of the EV (e.g., exosome) disclosed herein. For instance, in some aspects, by increasing the binding of a molecule of interest to a moiety on the EVs (e.g., scaffold moiety), an affinity ligand disclosed herein can allow for increased expression of the molecule of interest on the surface of an EV (e.g. , exterior surface). Accordingly, in certain aspects, a fusion protein comprising (i) a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety), (ii) an affinity ligand, and (iii) a scaffold moiety is present in the EV (e.g., exterior surface) at a higher density compared to a reference (e.g., corresponding fusion protein without the affinity ligand). In some aspects, the density of the fusion protein on the surface of the exosome is increased by at least about one-fold, at least about two-fold, at least about three -fold, at least about fourfold, at least about five-fold, at least about six-fold, at least about seven-fold, at least about eight-fold, at least about nine-fold, at least about 10-fold, at least about 20-fold, at least about 30-fold, at least about 40- fold, at least about 50-fold, at least about 60-fold, at least about 70-fold, at least about 80-fold, at least about 90-fold, at least about 100-fold, at least about 200-fold, at least about 300-fold, at least about 400-fold, at least about 500-fold, at least about 600-fold, at least about 700-fold, at least about 800-fold, at least about 900-fold, at least about 1,000-fold, at least about 2,000-fold, at least about 3,000-fold, at least about 4,000- fold, at least about 5,000-fold, at least about 6,000-fold, at least about 7,000-fold, at least about 8,000-fold, at least about 9,000-fold, at least about 10,000-fold or more, compared to the reference.
[0229] In some aspects, an improved binding of a molecule of interest (e.g., antigen, adjuvant, immune modulator, and/or targeting moiety) to a moiety expressed on the surface of an EV (e.g., scaffold moiety) can reduce the time required to produce an EV (e.g., exosome) disclosed herein. Accordingly, in
some aspects, an affinity ligand disclosed herein can reduce the time required for producing an engineered EV (e.g., exosome) disclosed herein (e.g., comprising a molecule of interest and a scaffold moiety). In certain aspects, the time required to produce an engineered EV (e.g., exosome) is reduced by at least about 5%, at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, or at least about 90% or more, compared to a reference (e.g., time required to produce the corresponding EV without the affinity ligand).
[0230] In some aspects, an affinity ligand useful for the present disclosure comprises a cleavage site, such as a protease (e.g., thrombin) cleavage site.
[0231] A non-limiting example of an EV (e.g., exosome) comprising an affinity ligand is described below. It will be apparent to those skilled in the art that an affinity ligand disclosed herein can be used in combination with other EVs (e.g., exosomes) disclosed herein.
III. Producer Cell for Production of Engineered EVs, e.g., Exosomes
[0232] EVs, e.g., exosomes, of the present disclosure can be produced from a cell grown in vitro or a body fluid of a subject. When exosomes are produced from in vitro cell culture, various producer cells, e.g., HEK293 cells, CHO cells, and MSCs, can be used. In certain aspects, a producer cell is not a naturally- existing dendritic cell, macrophage, B cell, mast cell, neutrophil, Kupffer-Browicz cell, cell derived from any of these cells, or any combination thereof (i.e., non-naturally existing producer cell). As used herein, the term "non-naturally existing producer cell" refers to a producer cell (e.g. , dendritic cell, macrophage, B cell, mast cell, neutrophil, Kupffer-Browicz cell, cell derived from any of these cells, or any combination thereof) that has been modified, such that the producer cell differs in structure and/or function compared to the naturally-existing counterpart. As described further below, in some aspects, the non-naturally existing producer cell has been modified to express one or more payloads disclosed herein (e.g., antigen, immune modulator, and/or adjuvant. In certain aspects, the non-naturally existing producer cell has been modified to express one or more targeting moieties disclosed herein. In some aspects, the non-naturally existing producer cell has been modified to express one or more scaffold moieties disclosed herein (e.g. , Scaffold X and/or Scaffold Y).
[0233] The producer cell can be genetically modified to comprise one or more exogenous sequences (e.g., encoding an antigen, adjuvant, immune modulator, and/or targeting moiety) to produce the EVs (e.g., exosomes) described herein. The genetically-modified producer cell can contain the exogenous sequence by transient or stable transformation. The exogenous sequence can be transformed as a plasmid. The exogenous sequences can be stably integrated into a genomic sequence of the producer cell, at a targeted site or in a random site. In some aspects, a stable cell line is generated for production of lumen-engineered EVs (e.g., exosomes).
IV. Pharmaceutical Compositions
[0234] Provided herein are pharmaceutical compositions comprising an EV, e.g., exosome, of the present disclosure having the desired degree of purity, and a pharmaceutically acceptable carrier or excipient, in a form suitable for administration to a subject. Pharmaceutically acceptable excipients or carriers can be determined in part by the particular composition being administered, as well as by the particular method used to administer the composition. Accordingly, there is a wide variety of suitable formulations of pharmaceutical compositions comprising a plurality of extracellular vesicles. (See, e.g., Remington's Pharmaceutical Sciences, Mack Publishing Co., Easton, Pa. 21st ed. (2005)). The pharmaceutical compositions are generally formulated sterile and in full compliance with all Good Manufacturing Practice (GMP) regulations of the U.S. Food and Drug Administration.
[0235] In some aspects, a pharmaceutical composition comprises one or more therapeutic agents and an exosome described herein. In certain aspects, the EVs, e.g., exosomes, are co-administered with of one or more additional therapeutic agents, in a pharmaceutically acceptable carrier. In some aspects, the pharmaceutical composition comprising the EV, e.g., exosome is administered prior to administration of the additional therapeutic agents. In some aspects, the pharmaceutical composition comprising the EV, e.g., exosome is administered after the administration of the additional therapeutic agents. In further aspects, the pharmaceutical composition comprising the EV, e.g., exosome is administered concurrently with the additional therapeutic agents.
[0236] Acceptable carriers, excipients, or stabilizers are nontoxic to recipients (e.g., animals or humans) at the dosages and concentrations employed, and include buffers such as phosphate, citrate, and other organic acids; antioxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, histidine, arginine, or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugars such as sucrose, mannitol, trehalose or sorbitol; salt-forming counter-ions such as sodium; metal complexes (e.g., Zn-protein complexes); and/or non-ionic surfactants such as TWEEN™, PLURONICS™ or polyethylene glycol (PEG).
[0237] Examples of carriers or diluents include, but are not limited to, water, saline, Ringer's solutions, dextrose solution, and 5% human serum albumin. The use of such media and compounds for pharmaceutically active substances is well known in the art. Except insofar as any conventional media or compound is incompatible with the extracellular vesicles described herein, use thereof in the compositions is contemplated. Supplementary therapeutic agents can also be incorporated into the compositions.
Typically, a pharmaceutical composition is formulated to be compatible with its intended route of administration. The EVs, e.g., exosomes, can be administered by parenteral, topical, intravenous, oral, subcutaneous, intra-arterial, intradermal, transdermal, rectal, intracranial, intraperitoneal, intranasal, intrathecal, intramuscular route or as inhalants. In certain aspects, the pharmaceutical composition comprising exosomes is administered intravenously, e.g. by injection. The EVs, e.g., exosomes, can optionally be administered in combination with other therapeutic agents that are at least partly effective in treating the disease, disorder or condition for which the EVs, e.g., exosomes, are intended.
[0238] Solutions or suspensions can include the following components: a sterile diluent such as water, saline solution, fixed oils, polyethylene glycols, glycerin, propylene glycol or other synthetic solvents; antibacterial compounds such as benzyl alcohol or methyl parabens; antioxidants such as ascorbic acid or sodium bisulfite; chelating compounds such as ethylenediaminetetraacetic acid (EDTA); buffers such as acetates, citrates or phosphates, and compounds for the adjustment of tonicity such as sodium chloride or dextrose. The pH can be adjusted with acids or bases, such as hydrochloric acid or sodium hydroxide. The preparation can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic.
[0239] Pharmaceutical compositions suitable for injectable use include sterile aqueous solutions (if water soluble) or dispersions and sterile powders. For intravenous administration, suitable carriers include physiological saline, bacteriostatic water, Cremophor EL™ (BASF, Parsippany, N.J.) or phosphate buffered saline (PBS). The composition is generally sterile and fluid to the extent that easy syringeability exists. The carrier can be a solvent or dispersion medium containing, e.g., water, ethanol, polyol (e.g., glycerol, propylene glycol, and liquid polyethylene glycol, and the like), and suitable mixtures thereof. The proper fluidity can be maintained, e.g., by the use of a coating such as lecithin, by the maintenance of the required particle size in the case of dispersion and by the use of surfactants. Prevention of the action of microorganisms can be achieved by various antibacterial and antifungal compounds, e.g., parabens, chlorobutanol, phenol, ascorbic acid, thimerosal, and the like. If desired, isotonic compounds, e.g., sugars, polyalcohols such as mannitol, sorbitol, and sodium chloride can be added to the composition. Prolonged absorption of the injectable compositions can be brought about by including in the composition a compound which delays absorption, e.g., aluminum monostearate and gelatin.
[0240] Sterile injectable solutions can be prepared by incorporating the EVs, e.g., exosomes, in an effective amount and in an appropriate solvent with one or a combination of ingredients enumerated herein, as desired. Generally, dispersions are prepared by incorporating the EVs, e.g., exosomes, into a sterile vehicle that contains a basic dispersion medium and any desired other ingredients. In the case of sterile powders for the preparation of sterile injectable solutions, methods of preparation are vacuum drying and freeze-drying that yields a powder of the active ingredient plus any additional desired ingredient from a previously sterile-fdtered solution thereof. The EVs, e.g., exosomes, can be administered in the form of a
depot injection or implant preparation which can be formulated in such a manner to permit a sustained or pulsatile release of the EV, e.g., exosomes.
[0241] Systemic administration of compositions comprising exosomes can also be by transmucosal means. For transmucosal administration, penetrants appropriate to the barrier to be permeated are used in the formulation. Such penetrants are generally known in the art, and include, e.g., for transmucosal administration, detergents, bile salts, and fusidic acid derivatives. Transmucosal administration can be accomplished through the use of, e.g., nasal sprays.
[0242] In certain aspects, the pharmaceutical composition comprising exosomes is administered intravenously into a subject that would benefit from the pharmaceutical composition. In some aspects, the composition is administered to the lymphatic system, e.g., by intralymphatic injection or by intranodal injection (see e.g., Senti etal., PNAS 105( 46): 17908 (2008)), or by intramuscular injection, by intrathecal administration, by subcutaneous administration, by direct injection into the thymus, or into the liver.
[0243] In certain aspects, the pharmaceutical composition comprising exosomes is administered as a liquid suspension. In certain aspects, the pharmaceutical composition is administered as a formulation that is capable of forming a depot following administration. In certain preferred aspects, the depot slowly releases the EVs, e.g., exosomes, into circulation, or remains in depot form.
[0244] Typically, pharmaceutically-acceptable compositions are highly purified to be free of contaminants, are biocompatible and not toxic, and are suited to administration to a subject. If water is a constituent of the carrier, the water is highly purified and processed to be free of contaminants, e.g., endotoxins.
[0245] The pharmaceutically-acceptable carrier can be lactose, dextrose, sucrose, sorbitol, mannitol, starch, gum acacia, calcium phosphate, alginates, gelatin, calcium silicate, micro-crystalline cellulose, polyvinylpyrrolidone, cellulose, water, syrup, methyl cellulose, methylhydroxy benzoate, propylhydroxy benzoate, talc, magnesium stearate, and/or mineral oil, but is not limited thereto. The pharmaceutical composition can further include a lubricant, a wetting agent, a sweetener, a flavor enhancer, an emulsifying agent, a suspension agent, and/or a preservative.
[0246] The pharmaceutical compositions described herein comprise the EVs, e.g., exosomes, described herein and optionally a pharmaceutically active or therapeutic agent. The therapeutic agent can be a biological agent, a small molecule agent, or a nucleic acid agent.
[0247] Dosage forms are provided that comprise a pharmaceutical composition comprising the EVs, e.g., exosomes, described herein. In some aspects, the dosage form is formulated as a liquid suspension for intravenous injection.
[0248] In certain aspects, the preparation of exosomes is subjected to radiation, e.g., X rays, gamma rays, beta particles, alpha particles, neutrons, protons, elemental nuclei, UV rays in order to damage residual replication-competent nucleic acids.
[0249] In certain aspects, the preparation of exosomes is subjected to gamma irradiation using an irradiation dose of more than 1, 5, 10, 15, 20, 25, 30, 35, 40, 50, 60, 70, 80, 90, 100, or more than 100 kGy. [0250] In certain aspects, the preparation of exosomes is subjected to X-ray irradiation using an irradiation dose of more than 0.1, 0.5, 1, 5, 10, 15, 20, 25, 30, 35, 40, 50, 60, 70, 80, 90, 100, 200, 300, 400, 500, 600, 700, 800, 900, 1000, 2000, 3000, 4000, 5000, 6000, 7000, 8000, 9000, 10000, or greater than 10000 mSv.
V. Kits
[0251] Also provided herein are kits comprising one or more EVs (e.g., exosomes) described herein.
In some aspects, provided herein is a kit comprising one or more containers fdled with one or more of the ingredients of the pharmaceutical compositions described herein. In certain aspects, the kit comprises a first container and a second container, wherein the first container comprises a base EV (e.g., exosome) described herein (e.g., comprising an adjuvant), and the second container comprises an antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator), and optional an instruction for use. As is apparent from the disclosure, in some aspects, combining the first and second container results in the antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator) to link to a surface of the EV.
VI. Vaccines
[0252] Also provided herein are kits comprising one or more EVs (e.g., exosomes) described herein.
In some aspects, provided herein is a kit comprising one or more containers filled with one or more of the ingredients of the pharmaceutical compositions described herein. In certain aspects, the kit comprises a first container and a second container, wherein the first container comprises a base EV (e.g., exosome) described herein (e.g., comprising an adjuvant), and the second container comprises an antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator), and optional an instruction for use. As is apparent from the disclosure, in some aspects, combining the first and second container results in the antigen of interest (or any additional moieties of interest, e.g., additional adjuvant, targeting moiety, or immune modulator) to link to a surface of the EV.
VII. Methods of Producing EVs (e.g., Exosomes)
[0253] In some aspects, the present disclosure is also directed to methods of producing exosomes described herein. In some aspects, the method comprises: obtaining the EV, e.g., exosome, from a producer cell, wherein the producer cell contains two or more components of the EV, e.g., exosome (e.g., (i) therapeutic molecule and adjuvant, (ii) therapeutic molecule and immune modulator, or (iii) therapeutic molecule, adjuvant, and immune modulator); and optionally isolating the obtained EV, e.g., exosome. In
some aspects, the method comprises: modifying a producer cell by introducing two or more components of an exosome disclosed herein (e.g., (i) therapeutic molecule and adjuvant, (ii) therapeutic molecule and immune modulator, or (iii) therapeutic molecule, adjuvant, and immune modulator); obtaining the EV, e.g., exosome from the modified producer cell; and optionally isolating the obtained EV, e.g. , exosome. In further aspects, the method comprises: obtaining an exosome from a producer cell; isolating the obtained exosome; and modifying the isolated exosome (e.g., by inserting multiple exogenous biologically active molecules, e.g., therapeutic molecules, adjuvants, immune modulators, and/or targeting moieties). In certain aspects, the method further comprises formulating the isolated exosome into a pharmaceutical composition.
VILA, Methods of Modifying a Producer Cell
[0254] As described supra, in some aspects, a method of producing an exosome comprises modifying a producer cell with multiple (e.g., two or more) exogenous biologically active molecules described herein (e.g., therapeutic molecule, adjuvant, immune modulator, and/or targeting moiety). In some aspects, a producer cell disclosed herein can be further modified with a scaffold moiety disclosed herein (e.g., Scaffold X or Scaffold Y).
[0255] In some aspects, the producer cell can be a mammalian cell line, a plant cell line, an insect cell line, a fungi cell line, or a prokaryotic cell line. In certain aspects, the producer cell is a mammalian cell line. Non-limiting examples of mammalian cell lines include: a human embryonic kidney (HEK) cell line, a Chinese hamster ovary (CHO) cell line, an HT-1080 cell line, a HeLa cell line, a PERC-6 cell line, a CEVEC cell line, a fibroblast cell line, an amniocyte cell line, an epithelial cell line, a mesenchymal stem cell (MSC) cell line, and combinations thereof. In certain aspects, the mammalian cell line comprises HEK- 293 cells, BJ human foreskin fibroblast cells, fHDF fibroblast cells, AGE.HN® neuronal precursor cells, CAP® amniocyte cells, adipose mesenchymal stem cells, RPTEC/TERT1 cells, or combinations thereof. In some aspects, the producer cell is a primary cell. In certain aspects, the primary cell can be a primary mammalian cell, a primary plant cell, a primary insect cell, a primary fungi cell, or a primary prokaryotic cell.
[0256] In some aspects, the producer cell is not an immune cell, such as an antigen presenting cell, a T cell, a B cell, a natural killer cell (NK cell), a macrophage, a T helper cell, or a regulatory T cell (Treg cell). In some aspects, the producer cell is not an antigen presenting cell (e.g., dendritic cells, macrophages, B cells, mast cells, neutrophils, Kupffer-Browicz cell, or a cell derived from any such cells).
[0257] In some aspects, the multiple exogenous biologically active molecules used to modify a producer cell can be a transgene or mRNA, and introduced into the producer cell by transfection, viral transduction, electroporation, extrusion, sonication, cell fusion, or other methods that are known to the skilled in the art.
[0258] In some aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by transfection. In some aspects, the multiple exogenous biologically active molecules can be
introduced into suitable producer cells using synthetic macromolecules, such as cationic lipids and polymers (Papapetrou et al., Gene Therapy 12: SI 18-S130 (2005)). In some aspects, the cationic lipids form complexes with the multiple exogenous biologically active molecules through charge interactions. In some of these aspects, the positively charged complexes bind to the negatively charged cell surface and are taken up by the cell by endocytosis. In some some aspects, a cationic polymer can be used to transfect producer cells. In some of these aspects, the cationic polymer is polyethylenimine (PEI). In certain aspects, chemicals such as calcium phosphate, cyclodextrin, or polybrene, can be used to introduce the multiple exogenous biologically active molecules to the producer cells. The multiple exogenous biologically active molecules can also be introduced into a producer cell using a physical method such as particle-mediated transfection, "gene gun", biolistics, or particle bombardment technology (Papapetrou et al., Gene Therapy 12: SI 18- S130 (2005)). A reporter gene such as, for example, beta-galactosidase, chloramphenicol acetyltransferase, luciferase, or green fluorescent protein can be used to assess the transfection efficiency of the producer cell. [0259] In certain aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by viral transduction. A number of viruses can be used as gene transfer vehicles, including moloney murine leukemia virus (MMLV), adenovirus, adeno-associated virus (AAV), herpes simplex virus (HSV), lentiviruses, and spumaviruses. The viral mediated gene transfer vehicles comprise vectors based on DNA viruses, such as adenovirus, adeno-associated virus and herpes virus, as well as retroviral based vectors.
[0260] In certain aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by electroporation. Electroporation creates transient pores in the cell membrane, allowing for the introduction of various molecules into the cell. In some aspects, DNA and RNA as well as polypeptides and non-polypeptide therapeutic agents can be introduced into the producer cell by electroporation.
[0261] In certain aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by microinjection. In some aspects, a glass micropipette can be used to inject the multiple exogenous biologically active molecules into the producer cell at the microscopic level.
[0262] In certain aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by extrusion.
[0263] In certain aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by sonication. In some aspects, the producer cell is exposed to high intensity sound waves, causing transient disruption of the cell membrane allowing loading of the multiple exogenous biologically active molecules.
[0264] In certain aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by cell fusion. In some aspects, the multiple exogenous biologically active molecules are introduced by electrical cell fusion. In some aspects, polyethylene glycol (PEG) is used to fuse the producer cells. In further aspects, sendai virus is used to fuse the producer cells.
[0265] In some aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by hypotonic lysis. In such aspects, the producer cell can be exposed to low ionic strength buffer causing them to burst allowing loading of the one or more moieties. In some aspects, controlled dialysis against a hypotonic solution can be used to swell the producer cell and to create pores in the producer cell membrane. The producer cell is subsequently exposed to conditions that allow resealing of the membrane.
[0266] In some aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by detergent treatment. In certain aspects, producer cell is treated with a mild detergent which transiently compromises the producer cell membrane by creating pores allowing loading of the multiple exogenous biologically active molecules. After producer cells are loaded, the detergent is washed away thereby re sealing the membrane.
[0267] In some aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by receptor mediated endocytosis. In certain aspects, producer cells have a surface receptor which, upon binding of the multiple exogenous biologically active molecules, induces internalization of the receptor and the associated molecules.
[0268] In some aspects, the multiple exogenous biologically active molecules are introduced to the producer cell by fdtration. In certain aspects, the producer cells and the multiple exogenous biologically active molecules can be forced through a filter of pore size smaller than the producer cell causing transient disruption of the producer cell membrane and allowing the multiple exogenous biologically active molecules to enter the producer cell.
[0269] In some aspects, the producer cell is subjected to several freeze thaw cycles, resulting in cell membrane disruption allowing loading of the multiple exogenous biologically active molecules.
VII, B, Methods of Modifying an Exosome
[0270] In some aspects, a method of producing an exosome comprises modifying the isolated exosome by directly introducing the multiple exogenous biologically active molecules into the EVs. In certain aspects, the multiple exogenous biologically active molecules comprise a therapeutic molecule (e.g., an antigen), adjuvant, immune modulator, targeting moieties, or combinations thereof. In some aspects, an isolated exosome can be further modified by directly introducing a scaffold moiety disclosed herein (e.g., Scaffold X or Scaffold Y) using any of the methods disclosed herein for introducing the multiple exogenous biologically active molecules into the EV, e.g., exosome.
[0271] In certain aspects, the multiple exogenous biologically active molecules are introduced to the exosome by transfection. In some aspects, the multiple exogenous biologically active molecules can be introduced into the EV using synthetic macromolecules such as cationic lipids and polymers (Papapetrou et al., Gene Therapy 12: S118-S130 (2005)). In certain aspects, chemicals such as calcium phosphate,
cyclodextrin, or polybrene, can be used to introduce the multiple exogenous biologically active molecules to the EV.
[0272] In certain aspects, the multiple exogenous biologically active molecules are introduced to the EV by electroporation. In some aspects, exosomes are exposed to an electrical field which causes transient holes in the EV membrane, allowing loading of the multiple exogenous biologically active molecules.
[0273] In certain aspects, the multiple exogenous biologically active molecules are introduced to the EV by microinjection. In some aspects, a glass micropipette can be used to inject the multiple exogenous biologically active molecules directly into the EV at the microscopic level.
[0274] In certain aspects, the multiple exogenous biologically active molecules are introduced to the EV by extrusion.
[0275] In certain aspects, the multiple exogenous biologically active molecules are introduced to the EV by sonication. In some aspects, EVs are exposed to high intensity sound waves, causing transient disruption of the EV membrane allowing loading of the multiple exogenous biologically active molecules. [0276] In some aspects, multiple exogenous biologically active molecules can be conjugated to the surface of the EV. Conjugation can be achieved chemically or enzymatically, by methods known in the art. [0277] In some aspects, the EV comprises multiple (e.g., two or more) exogenous biologically active molecules that are chemically conjugated. Chemical conjugation can be accomplished by covalent bonding of the multiple exogenous biologically active molecules to another molecule, with or without use of a linker. The formation of such conjugates is within the skill of artisans and various techniques are known for accomplishing the conjugation, with the choice of the particular technique being guided by the materials to be conjugated. In certain aspects, polypeptides are conjugated to the EV. In some aspects, non-polypeptides, such as lipids, carbohydrates, nucleic acids, and small molecules, are conjugated to the EV.
[0278] In some aspects, the multiple exogenous biologically active molecules are introduced to the EV by hypotonic lysis. In such aspects, the EVs can be exposed to low ionic strength buffer causing them to burst allowing loading of the multiple exogenous biologically active molecules. In some aspects, controlled dialysis against a hypotonic solution can be used to swell the EV and to create pores in the EV membrane. The EV is subsequently exposed to conditions that allow resealing of the membrane.
[0279] In some aspects, the multiple exogenous biologically active molecules are introduced to the EV by detergent treatment. In certain aspects, extracellular vesicles are treated with a mild detergent which transiently compromises the EV membrane by creating pores allowing loading of the multiple exogenous biologically active molecules. After EVs are loaded, the detergent is washed away thereby resealing the membrane.
[0280] In some aspects, the multiple exogenous biologically active molecules are introduced to the EV by receptor mediated endocytosis. In certain aspects, EVs have a surface receptor which, upon binding
of the multiple exogenous biologically active molecules, induces internalization of the receptor and the associated molecules.
[0281] In some aspects, the multiple exogenous biologically active molecules are introduced to the EV by mechanical firing. In certain aspects, extracellular vesicles can be bombarded with multiple exogenous biologically active molecules attached to a heavy or charged particle such as gold microcarriers. In some of these aspects, the particle can be mechanically or electrically accelerated such that it traverses the EV membrane.
[0282] In some aspects, extracellular vesicles are subjected to several freeze thaw cycles, resulting in EV membrane disruption allowing loading of the multiple exogenous biologically active molecules.
VII.C. Methods of Isolating an EV, e.g., Exosome
[0283] In some aspects, methods of producing EVs disclosed herein comprises isolating the EV from the producer cells. In certain aspects, the EVs released by the producer cell into the cell culture medium. It is contemplated that all known manners of isolation of EVs are deemed suitable for use herein. For example, physical properties of EVs can be employed to separate them from a medium or other source material, including separation on the basis of electrical charge (e.g., electrophoretic separation), size (e.g., filtration, molecular sieving, etc. ), density (e.g. , regular or gradient centrifugation), Svedberg constant (e.g. , sedimentation with or without external force, etc.). Alternatively, or additionally, isolation can be based on one or more biological properties, and include methods that can employ surface markers (e.g., for precipitation, reversible binding to solid phase, FACS separation, specific ligand binding, non-specific ligand binding, affinity purification etc.).
[0284] Isolation and enrichment can be done in a general and non-selective manner, typically including serial centrifugation. Alternatively, isolation and enrichment can be done in a more specific and selective manner, such as using EV or producer cell-specific surface markers. For example, specific surface markers can be used in immunoprecipitation, FACS sorting, affinity purification, and magnetic separation with bead-bound ligands.
[0285] In some aspects, size exclusion chromatography can be utilized to isolate the EVs. Size exclusion chromatography techniques are known in the art. Exemplary, non-limiting techniques are provided herein. In some aspects, a void volume fraction is isolated and comprises the EVs of interest. Further, in some aspects, the EVs can be further isolated after chromatographic separation by centrifugation techniques (of one or more chromatography fractions), as is generally known in the art. In some aspects, for example, density gradient centrifugation can be utilized to further isolate the extracellular vesicles. In certain aspects, it can be desirable to further separate the producer cell-derived EVs from EVs of other origin. For example, the producer cell-derived EVs can be separated from non-producer cell-derived EVs by immunosorbent capture using an antigen antibody specific for the producer cell.
[0286] In some aspects, the isolation of EVs can involve combinations of methods that include, but are not limited to, differential centrifugation, size -based membrane filtration, immunoprecipitation, FACS sorting, and magnetic separation.
VIII. Methods of Treatment
[0287] Present disclosure also provides methods of preventing and/or treating a disease or disorder in a subject in need thereof, comprising administering to the subject an EV (e.g., exosome) comprising a KRAS antigen (or a composition comprising the EV) to the subject. In certain aspects, the disease or disorder that can treated with the present disclosure comprises a cancer, particularly those associated with a KRAS mutant. Examples of such KRAS mutants are provided elsewhere in the present disclosure. As is apparent from the present disclosure, the EVs (e.g., exosomes) described herein are capable of treating all cancers associated with a KRAS mutation. For instance, using the methods described herein, EVs (e.g., exosomes) can be readily modified to comprise a KRAS antigen derived from the KRAS mutant protein associated with a particular cancer. When administered, such EVs (e.g., exosomes) would be capable of inducing an anti -tumor immune response (i.e., specifically targeting KRAS proteins with the particular mutation) in the subject and thereby, treat the cancer.
[0288] In some aspects, the cancer is associated with a solid tumor. In some aspects, the cancer is associated with a liquid tumor. As used herein, the term "solid tumor" refers to an abnormal mass of tissue that does not contain cysts or liquid areas, and generally occur in the bones, muscles, and organs. As used herein, the term "liquid tumor" refers to tumors that occur in body fluids (e.g., blood and bone marrow).
[0289] Non-limiting examples of cancers that can be treated with the present disclosure include a colorectal cancer, lung cancer (e.g., non-small cell lung cancer (NSCLC)), pancreatic cancer (e.g., pancreatic ductal adenocarcinoma), leukemia, uterine cancer, ovarian cancer, bladder cancer, bile duct cancer, gastric cancer, stomach cancer, testicular cancer, esophageal cancer, cholangiocarcinoma, cervical cancer, acute myeloid leukemia (AML), diffuse large B-cell lymphoma (DLBC), sarcoma, melanoma, glioma (e.g., low-grade glioma, e.g., glioblastoma), mesothelioma, liver cancer, breast cancer (e.g., breast invasive carcinoma), renal carcinoma (e.g., papillary renal cell carcinoma (pRCC), and chromophobe renal cell carcinoma), head and neck cancer, prostate cancer, adenoid cystic carcinoma (ACC), thymoma cancer, thyroid cancer, clear cell renal cell carcinoma (CCRCC), neuroendocrine neoplasm (e.g., pheochromocytoma/paraganglioma), uveal melanoma, multiple myeloma, or any combination thereof.
[0290] When administered to a subject with a cancer, in certain aspects, EVs (e.g., exosome) of the present disclosure can up-regulate an immune response and enhance the tumor targeting of the subject’s immune system. In some aspects, the cancer being treated is characterized by infiltration of leukocytes (T- cells, B-cells, macrophages, dendritic cells, monocytes) into the tumor microenvironment, or so-called "hot tumors" or "inflammatory tumors". In some aspects, the cancer being treated is characterized by low levels or undetectable levels of leukocyte infiltration into the tumor microenvironment, or so-called "cold tumors"
or "non-inflammatory tumors" . In some aspects, an EV is administered in an amount and for a time sufficient to convert a "cold tumor" into a "hot tumor", i.e. , said administering results in the infiltration of leukocytes (such as T-cells) into the tumor microenvironment. In certain aspects, cancer comprises bladder cancer, cervical cancer, renal cell cancer, testicular cancer, colorectal cancer, lung cancer, head and neck cancer, and ovarian, lymphoma, liver cancer, glioblastoma, melanoma, myeloma, leukemia, pancreatic cancers, or combinations thereof. In other term, "distal tumor" or "distant tumor" refers to a tumor that has spread from the original (or primary) tumor to distant organs or distant tissues, e.g. , lymph nodes. In some aspects, the EVs of the disclosure treats a tumor after the metastatic spread.
[0291] In some aspects, administering an EV, e.g., exosome, disclosed herein (e.g., comprising a
KRAS antigen, adjuvant, and T helper peptide) inhibits and/or reduces growth of a tumor in a subject. In some aspects, the growth of a tumor (e.g. , tumor volume or weight) is reduced by at least about 5%, at least about 10%, at least about 20%, at least about 30%, at least about 40%, at least about 50%, at least about 60%, at least about 70%, at least about 80%, at least about 90%, or about 100% compared to a reference (e.g., tumor volume in a corresponding subject after administration of an EV, e.g., exosome, without the ASO).
[0292] In some aspects, EVs (e.g., exosomes) disclosed herein can be used to treat a fibrosis. Nonlimiting examples of fibrosis that can be treated include liver fibrosis (NASH), cirrhosis, pulmonary fibrosis, cystic fibrosis, chronic ulcerative colitis/IBD, bladder fibrosis, kidney fibrosis, CAPS (Muckle-Wells syndrome), atrial fibrosis, endomyocardial fibrosis, old myocardial infarction, glial scar, arterial stiffness, arthrofibrosis, Crohn's disease, Dupuytren's contracture, keloid fibrosis, mediastinal fibrosis, myelofibrosis, Peyronie's disease, nephrogenic systemic fibrosis, progressive massive fibrosis, retroperitoneal fibrosis, scleroderma/systemic sclerosis, adhesive capsulitis, neurofibromatosis type 1 (NFl), or any combination thereof. In certain aspects, the fibrosis is associated with a cancer (e.g., pancreatic ductal adenocarcinoma (PDAC)).
[0293] EVs (e.g., exosomes) of the present disclosure can be administered to a subject by any useful method and/or route known in the art. In some aspects, the EVs (e.g., exosomes) are administered intravenously to the circulatory system of the subject. In some aspects, the EVs (e.g., exosomes) are infused in suitable liquid and administered into a vein of the subject.
[0294] In some aspects, the EVs (e.g., exosomes) are administered intra-arterially to the circulatory system of the subject. In some aspects, the EVs (e.g., exosomes) are infused in suitable liquid and administered into an artery of the subject.
[0295] In some aspects, the EVs (e.g., exosomes) are administered to the subject by intranasal administration. In some aspects, the EVs (e.g., exosomes) can be insufflated through the nose in a form of either topical administration or systemic administration. In certain aspects, the EVs (e.g., exosomes) are administered as nasal spray. In some aspects, intranasal administration can allow for the effective delivery
of an EV (e.g., exosome) disclosed herein to the gastrointestinal tissues. Such EVs (e.g., exosomes) delivered to the gastrointestinal tissues could be useful in providing protection against various gut- associated pathogens.
[0296] In some aspects, the EVs (e.g., exosomes) are administered to the subject by intraperitoneal administration. In some aspects, the EVs (e.g., exosomes) are infused in suitable liquid and injected into the peritoneum of the subject. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to the lymphatics. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to the thymus, spleen, and/or bone marrow. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to one or more lymph nodes. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to one or more of the cervical lymph node, the inguinal lymph node, the mediastinal lymph node, or the sternal lymph node. In some aspects, the intraperitoneal administration results in distribution of the EVs (e.g., exosomes) to the pancreas.
[0297] Non-limiting examples of other routes of administration that can be used to administer the
EVs (e.g., exosomes) disclosed herein include parenteral, topical, oral, subcutaneous, intradermal, transdermal, rectal, intraperitoneal, intramuscular, sublingual, or combinations thereof.
[0298] As disclosed herein, in some aspects, EVs (e.g., exosomes) disclosed herein can be administered to a subject in combination with one or more additional agents, e.g., adjuvants. In certain aspects, the one or more additional agents and the EVs (e.g., exosomes) are administered concurrently. In some aspects, the one or more additional agents and the EVs (e.g., exosomes) are administered sequentially. In some aspects, the EVs (e.g., exosomes) are administered to the subject prior to administering the one or more additional agents. In certain aspects, the EVs (e.g., exosome) are administered to the subject after administering the one or more additional agents. As used herein, the term " agents" refers to any agents that can be used in combination with the present EVs, e.g., in treating an infectious disease or disorder disclosed herein). In some aspects, the one or more additional agents that can be used in combination with the EVs (e.g., exosomes) of the present disclosure include a payload (e.g., antigen, adjuvant, and/or immune modulator) which is not expressed in an EV (e.g., exosome). For instance, a treatment method disclosed herein can comprise administering to a subject in need thereof (i) an antigen-expressing EV (e.g., exosome) and (ii) an antigen that is the same or different from that expressed in the EV (e.g., soluble antigen).
[0299] In some aspects, a subject that can be treated with the present disclosure is a human. In some aspects, a subject is a non-human mammal (e.g., non-human primates, dogs, cats, guinea pigs, rabbits, rats, mice, horses, cattle, chickens, birds, and bears). Accordingly, in some aspects, the EVs (e.g., exosomes) disclosed herein can be used to improve the health of an animal (i.e., non-human mammal)
[0300] The practice of the present disclosure will employ, unless otherwise indicated, conventional techniques of cell biology, cell culture, molecular biology, transgenic biology, microbiology, recombinant DNA, and immunology, which are within the skill of the art. Such techniques are explained fully in the literature. See, for example, Sambrook et al., ed. (1989) Molecular Cloning A Laboratory Manual (2nd ed.; Cold Spring Harbor Laboratory Press); Sambrook et al., ed. (1992) Molecular Cloning: A Laboratory Manual, (Cold Springs Harbor Laboratory, NY); D. N. Glover ed., (1985) DNA Cloning, Volumes I and II; Gait, ed. (1984) Oligonucleotide Synthesis; Mullis et al. U.S. Pat. No. 4,683,195; Hames and Higgins, eds. (1984) Nucleic Acid Hybridization; Hames and Higgins, eds. (1984) Transcription And Translation; Freshney (1987) Culture Of Animal Cells (Alan R. Liss, Inc.); Immobilized Cells And Enzymes (IRL Press) (1986); Perbal (1984) A Practical Guide To Molecular Cloning; the treatise, Methods In Enzymology (Academic Press, Inc., N.Y.); Miller and Calos eds. (1987) Gene Transfer Vectors For Mammalian Cells, (Cold Spring Harbor Laboratory); Wu et al., eds., Methods In Enzymology, Vols. 154 and 155; Mayer and Walker, eds. (1987) Immunochemical Methods In Cell And Molecular Biology (Academic Press, London); Weir and Blackwell, eds., (1986) Handbook Of Experimental Immunology, Volumes I-IV; Manipulating the Mouse Embryo, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., (1986); Crooke, Antisense drug Technology: Principles, Strategies and Applications, 2nd Ed. CRC Press (2007) and in Ausubel et al. (1989) Current Protocols in Molecular Biology (John Wiley and Sons, Baltimore, Md.).
[0301] The following examples are offered by way of illustration and not by way of limitation.
EXAMPLES
Example 1: Construction ofEVs (e.g., Exosomes) Comprising a KRAS Antigen
[0302] To assess the therapeutic capability of the EVs (e.g., exosomes) described herein, an engineered EV (e.g., exosomes) comprising a KRAS antigen will be constructed. As shown in FIG. 1A, in some aspects, the KRAS antigen will be linked to either the external surface and/or the luminal surface of the EVs (e.g., exosome) using the various methods disclosed herein (e.g., scaffold moieties, e.g., PTGFRN or BASP1). In some aspects, multiple KRAS antigens will be added to the EVs, e.g., either as separate KRAS antigens or as a peptide concatemer separated by a linker. As described herein, in some aspects, the EVs (e.g., exosomes) will further comprise an adjuvant, T helper peptide, and/or any of the additional moieties of interest described herein (e.g., targeting moiety and an immune modulator).
[0303] In some aspects, once constructed, the EVs (e.g., exosomes) will be tested in an animal model to assess the ability of the vaccines to prevent and/or treat a disease or disorder.
INCORPORATION BY REFERENCE
[0304] All publications, patents, patent applications and other documents cited in this application are hereby incorporated by reference in their entireties for all purposes to the same extent as if each individual publication, patent, patent application or other document were individually indicated to be incorporated by reference for all purposes.
EQUIVALENTS
[0305] While various specific aspects have been illustrated and described, the above specification is not restrictive. It will be appreciated that various changes can be made without departing from the spirit and scope of the disclosure(s). Many variations will become apparent to those skilled in the art upon review of this specification.
Claims
1. A composition comprising (i) an isolated extracellular vesicle (EV) comprising a KRAS antigen, and (ii) an adjuvant.
2. The composition of claim 1, which further comprises a T helper peptide.
3. The composition of claim 1 or 2, wherein the KRAS antigen comprises an amino acid substitution selected from G12D, G12C, G12V, G13D, G12A, G12R, G12S, G13C, G13A, GBR, G13S, G13V, Q61H, Q61L, Q61R, Q61K, A18D, KI 17N, or any combination thereof corresponding to SEQ ID NO: 381.
4. The composition of claim 3, wherein the amino acid substitution of the KRAS antigen is selected from G12D, G12C, G12V, G13D, and any combination thereof corresponding to SEQ ID NO: 381.
5. The composition of any one of claims 1 to 4, wherein the KRAS antigen comprises a natural peptide, synthetic peptide, or both.
6. The composition of any one of claims 1 to 5, wherein the KRAS antigen is less than about 50 amino acids in length, less than about 45 amino acids in length, less than about 40 amino acids in length, less than about 35 amino acids in length, less than about 30 amino acids in length, less than about 25 amino acids in length, less than about 20 amino acids in length, less than about 15 amino acids in length, or less than about 10 amino acids in length.
7. The composition of claim 6, wherein the KRAS antigen is between about 12 to about 20 amino acids in length.
8. The composition of any one of claim 1 to 7, wherein the KRAS antigen comprises a concatemer of multiple KRAS antigens.
9. The composition of claim 8, wherein the multiple KRAS antigens are separated by a spacer.
10. The composition of claim 9, wherein the spacer comprises the amino acid sequence AAY, GPGPG (SEQ ID NO: 391), GSGSG (SEQ ID NO: 392), or both.
11. The composition of any one of claims 1 to 10, wherein the KRAS antigen comprises or consists of an epitope of amino acids 1 to 32 or 97 to 137 of SEQ ID NO: 381, wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at
least 16 amino acids, at least 17 amino acids, at least 18 ammo acids, at least 19 ammo acids, at least 20 ammo acids, at least 21 ammo acids, at least 22 ammo acids, at least 23 ammo acids, at least 24 ammo acids, at least 25 ammo acids, at least 26 ammo acids, at least 27 ammo acids, at least 28 amino acids, at least 29 amino acids, or at least 30 amino acids in length.
The composition of claim 11, wherein the KRAS antigen comprises or consists of an epitope of amino acids 1 to 32 of SEQ ID NO: 381 (SEQ ID NO: 393), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids in length, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, or all 32 amino acids.
The composition of claim 11, wherein the KRAS antigen comprises or consists of an epitope of amino acids 97 to 137 of SEQ ID NO: 381 (SEQ ID NO: 394), wherein the epitope has at least 3 amino acids, at least 4 amino acids, at least 5 amino acids, at least 6 amino acids, at least 7 amino acids, at least 8 amino acids, at least 9 amino acids, at least 10 amino acids, at least 11 amino acids, at least 12 amino acids, at least 13 amino acids, at least 14 amino acids, at least 15 amino acids, at least 16 amino acids, at least 17 amino acids, at least 18 amino acids, at least 19 amino acids, at least 20 amino acids, at least 21 amino acids, at least 22 amino acids, at least 23 amino acids, at least 24 amino acids, at least 25 amino acids, at least 26 amino acids, at least 27 amino acids, at least 28 amino acids, at least 29 amino acids, at least 30 amino acids, at least 31 amino acids, at least 32 amino acids, at least 33 amino acids, at least 34 amino acids, at least 35 amino acids, at least 36 amino acids, at least 37 amino acids, at least 38 amino acids, at least 39 amino acids, or all
40 amino acids in length. The composition of any one of claims 1 to 13, wherein the KRAS antigen is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. The composition of any one of claims 2 to 14, wherein the T helper peptide is associated with the EV.
- 70 - The composition of claim 15, wherein the T helper peptide is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. The composition of any one of claims 2 to 14, wherein the T helper peptide is not associated with the EV. The composition of any one of claims 2 to 17, wherein the T helper peptide comprises a universal T helper peptide. The composition of claim 18, wherein the universal T helper peptide comprises a PADRE, tetanus toxin, diphtheria toxin, HBV peptide, measles peptide, or any combination thereof. The composition of claim 19, wherein the tetanus toxin is a tetanus toxin P2, tetanus toxin P30, or both. The composition of claim 19, wherein the diphtheria toxic is CRM- 197. The composition of claim 19, wherein the HBV peptide is HbsAg. The composition of any one of claims 2 to 17, wherein the T helper peptide is a KRAS peptide comprising an epitope for a CD4+ T cell ("KRAS CD4+ T cell peptide"). The composition of any one of claims 2 to 23, wherein the T helper peptide comprises a concatemer of multiple T helper peptides. The composition of claim 24, wherein the multiple T helper peptides are separated by a spacer. The composition of any one of claims 1 to 25, wherein the adjuvant is associated with the EV. The composition of claim 26, wherein the adjuvant is: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. The composition of any one of claims 1 to 25, wherein the adjuvant is not associated with the EV. The composition of any one of claims 1 to 28, wherein the adjuvant comprises a STING agonist, TLR agonist, or both. The composition of claim 29, wherein the STING agonist comprises a cyclic dinucleotide STING agonist or a non-cyclic dinucleotide STING agonist.
- 71 - The composition of claim 29, wherein the TLR agonist comprises a TLR2 agonist (e.g. , lipoteichoic acid, atypical LPS, MALP-2 and MALP-404, OspA, porin, LcrV, lipomannan, GPI anchor, lysophosphatidylserine, lipophosphoglycan (LPG), glycophosphatidylinositol (GPI), zymosan, hsp60, gH/gL glycoprotein, hemagglutinin), a TLR3 agonist (e.g., double -stranded RNA, e.g., poly(I:C), ampligen, hiltonol, polyA:U), a TLR4 agonist (e.g., lipopolysaccharides (LPS), lipoteichoic acid, P-defensin 2, fibronectin EDA, HMGB1, snapin, tenascin C, MPLA), a TLR5 agonist (e.g., flagellin), a TLR6 agonist, a TLR7/8 agonist (e.g., single -stranded RNA, Poly GIO, Poly G3, Resiquimod, Imiquimod, 3M-052), a TLR9 agonist (e.g., unmethylated CpG DNA), or any combination thereof. The composition of any one of claims 1 to 31, which further comprises one or more additional moieties selected from an immune modulator, targeting moiety, or any combination thereof. The composition of claim 32, wherein the immune modulator and/or targeting moiety are associated with the EV. The composition of claim 33, wherein the immune modulator, targeting moiety, and/or antiphagocytic moiety are: (i) linked to the exterior surface of the EV, (ii) linked to the luminal surface of the EV, (iii) in the lumen of the EV, or (iv) any combination thereof. The composition of claim 32, wherein the immune modulator and/or targeting moiety are not associated with the EV. The composition of any one of claims 32 to 35, wherein the immune modulator comprises (i) an inhibitor for a negative checkpoint regulator or an inhibitor for a binding partner of a negative checkpoint regulator (e.g., anti-CTLA4 antibody); (ii) an activator for a positive co-stimulatory molecule or an activator for a binding partner of a positive co-stimulatory molecule (e.g., CD40L); (iii) a cytokine or a binding partner of a cytokine; (iv) a protein that supports intracellular interactions required for germinal center responses; (v) a polynucleotide; or (vi) any combination thereof. The composition of any one of claims 32 to 36, wherein the targeting moiety binds to a marker expressed on an immune cell (e.g., dendritic cell, T cell, B cell, or any combination thereof). The composition of claim 37, wherein the marker expressed on an immune cell comprises a C-type lectin domain family 9 member A (Clec9a) protein, a dendritic cell-specific intercellular adhesion molecule -3 -grabbing non-integrin (DC-SIGN), CD207, CD40, Clec6, dendritic cell immunoreceptor (DCIR), DEC-205, lectin-like oxidized low-density lipoprotein receptor- 1 (LOX-
- 72 -
1), MARCO, Clecl2a, DC-asialogly coprotein receptor (DC-ASGPR), DC immunoreceptor 2 (DCIR2), Dectin-1, macrophage mannose receptor (MMR), BDCA-1 (CD303, Clec4c), Dectin-2, Bst-2 (CD317), CD3, CD19, CDla, CDl lc, Langeran, CD103, XCR1, BDCA-3 (CD141), or any combination thereof. The composition of claim 32 to 36, wherein the targeting moiety binds to a marker expressed in a lymphoid tissue of a subject suffering from a cancer. The composition of claim 39, the lymphoid tissue comprises a tumor draining lymph node, sentinel lymph node, tumor tertiary lymph node, or any combination thereof. The composition of claim 40, wherein the targeting moiety comprises anti-fibronectin EDA & EDB domain antibodies; anti-ICAM-1; anti-CD20; anti-DC-LAMP; or any combination thereof. The composition of any one of claims 1 to 41, wherein the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties is linked to the exterior surface and/or luminal surface of the EV by an anchoring moiety, affinity agent, chemical conjugation, cell penetrating peptide (CPP), or any combination thereof. The composition of claim 42, wherein the anchoring moiety comprises a cholesterol, fatty acid (e.g. , palmitate), tocopherol (e.g., vitamin E), or any combination thereof. The composition of claim 42, wherein the chemical conjugation comprises a maleimide moiety, copper-free, biorthogonal click chemistry (e.g., azide/strained alkyne (DIFO, DBCO, BCN)), metal-catalyzed click chemistry (e.g., CUAAC, RUAAC), or any combination thereof. The composition of any one of claims 1 to 44, which further comprises one or more scaffold moieties. The composition of claim 45, wherein: (i) the KRAS antigen is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; (ii) the T helper peptide is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; (iii) the adjuvant is linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; (iv) the one or more additional moieties are linked to the one or more scaffold moieties on the exterior surface and/or luminal surface of the EV; or (v) any combination thereof. . The composition of claim 46, wherein: (i) the KRAS antigen is linked to the one or more scaffold moieties by a linker; (ii) the T helper peptide is linked to the one or more scaffold moieties by a linker; (iii) the adjuvant is linked to the one or more scaffold moieties by a linker; (iv) the one or
- 73 - more additional moieties are linked to the one or more scaffold moieties by a linker; or (v) any combination thereof. The composition of claim 47, wherein the linker is a polypeptide. The composition of claim 47, wherein the linker is a non-polypeptide moiety. The composition of any one of claims 47 to 49, wherein the linker comprises a maleimide moiety. The composition of any one of claims 47 to 50, wherein the linker comprises a cholesterol moiety. The composition of any one of claims 45 to 51, wherein the one or more scaffold moieties comprise a Scaffold X, Scaffold Y, or both. The composition of claim 52, wherein the Scaffold X comprises a prostaglandin F2 receptor negative regulator (the PTGFRN protein); basigin (the BSG protein); immunoglobulin superfamily member 2 (the IGSF2 protein); immunoglobulin superfamily member 3 (the IGSF3 protein); immunoglobulin superfamily member 8 (the IGSF8 protein); integrin beta-1 (the ITGB1 protein); integrin alpha-4 (the ITGA4 protein); 4F2 cell-surface antigen heavy chain (the SLC3A2 protein); a class of ATP transporter proteins (the ATP1A1, ATP1A2, ATP1A3, ATP1A4, ATP1B3, ATP2B1, ATP2B2, ATP2B3, ATP2B4 proteins), or any combination thereof. The composition of claim 52 or 53, wherein the Scaffold X is PTGFRN or a fragment thereof. The composition of any one of claims 52 to 54, wherein the Scaffold Y comprises myristoylated alanine rich Protein Kinase C substrate (the MARCKS protein); myristoylated alanine rich Protein Kinase C substrate like 1 (the MARCKSL1 protein); brain acid soluble protein 1 (the BASP1 protein), and any combination thereof. The composition of any one of claims 52 to 55, wherein the Scaffold Y is BASP1 or a fragment thereof. The composition of any one of claims 1 to 56, wherein the EV is an exosome. A pharmaceutical composition comprising the EV of any one of claims 1 to 57 and a pharmaceutically acceptable carrier. A cell that produces the EV of any one of claims 1 to 57. A kit comprising the EV of any one of claims 1 to 57 and instructions for use.
An EV-drug conjugate comprising the EV of any one of claims 1 to 57. A method of making EVs comprising culturing the cell of claim 59 under a suitable condition and obtaining the EVs. A method of making EVs comprising loading an EV that has been isolated from a producer cell with the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties of any one of claims 1 to 57. A method of making a cancer vaccine comprising loading an EV with the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties of any one of claims 1 to 57. The method of claim 64, wherein the EV is isolated from a producer cell prior to the loading of the KRAS antigen, T helper peptide, adjuvant, and/or one or more additional moieties. A vaccine produced by the method of claim 64 or 65. A method of inducing an immune response in a subject in need thereof, comprising administering the composition of any one of claims 1 to 57 to the subject. A method of inducing a vaccine response in a subject afflicted with a cancer, comprising administering the vaccine of claim 66. A method of preventing or treating a cancer in a subject in need thereof, comprising administering the composition of any one of claims 1 to 57 or the vaccine of claim 66 to the subject. The method of claim 69, wherein the cancer comprises a colorectal cancer, lung cancer (e.g., nonsmall cell lung cancer (NSCLC)), pancreatic cancer (e.g., pancreatic ductal adenocarcinoma), leukemia, uterine cancer, ovarian cancer, bladder cancer, bile duct cancer, gastric cancer, stomach cancer, testicular cancer, esophageal cancer, cholangiocarcinoma, cervical cancer, acute myeloid leukemia (AML), diffuse large B-cell lymphoma (DLBC), sarcoma, melanoma, glioma (e.g., low- grade glioma, e.g., glioblastoma), mesothelioma, liver cancer, breast cancer (e.g., breast invasive carcinoma), renal carcinoma (e.g., papillary renal cell carcinoma (pRCC), and chromophobe renal cell carcinoma), head and neck cancer, prostate cancer, adenoid cystic carcinoma (ACC), thymoma cancer, thyroid cancer, clear cell renal cell carcinoma (CCRCC), neuroendocrine neoplasm (e.g., pheochromocytoma/paraganglioma), uveal melanoma, multiple myeloma, or any combination thereof.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202063082433P | 2020-09-23 | 2020-09-23 | |
| US63/082,433 | 2020-09-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022066883A1 true WO2022066883A1 (en) | 2022-03-31 |
Family
ID=78179536
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2021/051720 Ceased WO2022066883A1 (en) | 2020-09-23 | 2021-09-23 | Extracellular vesicles comprising kras antigens and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2022066883A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025080869A1 (en) * | 2023-10-11 | 2025-04-17 | The Regents Of The University Of California | Pancreatic cancer immunotherapy using cationic lipid and polymer nanocarriers for co-delivery of kras neoantigens and sting and/or other immunomodulators to boost the cancer immunity cycle and treat liver metastasis |
Citations (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4683195A (en) | 1986-01-30 | 1987-07-28 | Cetus Corporation | Process for amplifying, detecting, and/or-cloning nucleic acid sequences |
| WO2008115319A2 (en) | 2007-02-07 | 2008-09-25 | Regents Of The University Of California | Conjugates of synthetic tlr agonists and uses therefor |
| WO2009030996A1 (en) | 2007-09-05 | 2009-03-12 | Coley Pharmaceutical Group, Inc. | Triazole compounds as toll-like receptor (tlr) agonists |
| WO2009088401A2 (en) | 2007-09-24 | 2009-07-16 | Government Of The Usa, As Represented By The Secretary, Dept. Of Health And Human Services | Immunostimulatory combinations of tlr ligands and methods of use |
| US7655413B2 (en) | 2003-06-26 | 2010-02-02 | Lifesensors, Inc. | Methods and compositions for enhanced protein expression and purification |
| US20100029585A1 (en) | 2008-08-01 | 2010-02-04 | Howbert J Jeffry | Toll-like receptor agonist formulations and their use |
| WO2011044246A1 (en) | 2009-10-06 | 2011-04-14 | Cleveland Biolabs, Inc. | Use of toll-like receptor and agonist for treating cancer |
| US8137695B2 (en) | 2006-08-18 | 2012-03-20 | Arrowhead Madison Inc. | Polyconjugates for in vivo delivery of polynucleotides |
| US20120219615A1 (en) | 2010-10-01 | 2012-08-30 | The Trustees Of The University Of Pennsylvania | Therapeutic Use of a TLR Agonist and Combination Therapy |
| US8354455B2 (en) | 2004-08-26 | 2013-01-15 | Piramal Enterprises Limited and Apparao Satyam | Prodrugs containing novel bio-cleavable linkers |
| US8426565B2 (en) | 2007-08-30 | 2013-04-23 | Walter And Eliza Hall Institute Of Medical Research | Dendritic cell marker and uses thereof |
| US20130202707A1 (en) | 2008-02-13 | 2013-08-08 | Dana-Farber Cancer Institute, Inc. | Controlled Delivery of TLR Agonists in Structural Polymeric Devices |
| WO2013185052A1 (en) | 2012-06-08 | 2013-12-12 | Aduro Biotech | Compostions and methods for cancer immunotherapy |
| WO2014093936A1 (en) | 2012-12-13 | 2014-06-19 | Aduro Biotech, Inc. | Compositions comprising cyclic purine dinucleotides having defined stereochemistries and methods for their preparation and use |
| WO2014179760A1 (en) | 2013-05-03 | 2014-11-06 | The Regents Of The University Of California | Cyclic di-nucleotide induction of type i interferon |
| WO2014179335A1 (en) | 2013-04-29 | 2014-11-06 | Memorial Sloan Kettering Cancer Center | Compositions and methods for altering second messenger signaling |
| WO2014189805A1 (en) | 2013-05-18 | 2014-11-27 | Auro Biotech, Inc. | Compositions and methods for activating "stimulator of interferon gene"-dependent signalling |
| WO2014189806A1 (en) | 2013-05-18 | 2014-11-27 | Aduro Biotech, Inc. | Compositions and methods for inhibiting "stimulator of interferon gene" dependent signalling |
| WO2015017652A1 (en) | 2013-07-31 | 2015-02-05 | Memorial Sloan-Kettering Cancer Center | Sting crystals and modulators |
| WO2015077354A1 (en) | 2013-11-19 | 2015-05-28 | The University Of Chicago | Use of sting agonist as cancer treatment |
| WO2015185565A1 (en) | 2014-06-04 | 2015-12-10 | Glaxosmithkline Intellectual Property Development Limited | Cyclic di-nucleotides as modulators of sting |
| WO2016096577A1 (en) | 2014-12-16 | 2016-06-23 | Invivogen | Combined use of a chemotherapeutic agent and a cyclic dinucleotide for cancer treatment |
| WO2016096174A1 (en) | 2014-12-16 | 2016-06-23 | Invivogen | Fluorinated cyclic dinucleotides for cytokine induction |
| WO2016120305A1 (en) | 2015-01-29 | 2016-08-04 | Glaxosmithkline Intellectual Property Development Limited | Cyclic dinucleotides useful for the treatment of inter alia cancer |
| WO2016145102A1 (en) | 2015-03-10 | 2016-09-15 | Aduro Biotech, Inc. | Compositions and methods for activating "stimulator of interferon gene" -dependent signalling |
| WO2017027645A1 (en) | 2015-08-13 | 2017-02-16 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| WO2017075477A1 (en) | 2015-10-28 | 2017-05-04 | Aduro Biotech, Inc. | Compositions and methods for activating "stimulator of interferon gene"-dependent signalling |
| WO2017161349A1 (en) | 2016-03-18 | 2017-09-21 | Immune Sensor, Llc | Cyclic di-nucleotide compounds and methods of use |
| WO2017175156A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2017175147A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| US9790494B2 (en) | 2012-09-14 | 2017-10-17 | Translate Bio Ma, Inc. | Multimeric oligonucleotide compounds having non-nucleotide based cleavable linkers |
| WO2018100558A2 (en) | 2016-12-01 | 2018-06-07 | Takeda Pharmaceutical Company Limited | Cyclic dinucleotide |
| WO2018145020A1 (en) * | 2017-02-03 | 2018-08-09 | The Medical College Of Wisconsin, Inc. | Kras peptide vaccine compositions and method of use |
| US10195290B1 (en) | 2017-08-25 | 2019-02-05 | Codiak Biosciences, Inc. | Preparation of therapeutic exosomes using membrane proteins |
| WO2019099942A1 (en) | 2017-11-17 | 2019-05-23 | Codiak Biosciences, Inc. | Compositions of engineered exosomes and methods of loading luminal exosomes payloads |
| WO2019183578A1 (en) | 2018-03-23 | 2019-09-26 | Codiak Biosciences, Inc. | Extracellular vesicles comprising sting-agonist |
| WO2020101740A1 (en) | 2018-11-16 | 2020-05-22 | Codiak Biosciences, Inc. | Engineered extracellular vesicles and uses thereof |
| WO2020191361A2 (en) * | 2019-03-21 | 2020-09-24 | Codiak Biosciences, Inc. | Extracellular vesicles for vaccine delivery |
-
2021
- 2021-09-23 WO PCT/US2021/051720 patent/WO2022066883A1/en not_active Ceased
Patent Citations (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4683195B1 (en) | 1986-01-30 | 1990-11-27 | Cetus Corp | |
| US4683195A (en) | 1986-01-30 | 1987-07-28 | Cetus Corporation | Process for amplifying, detecting, and/or-cloning nucleic acid sequences |
| US7655413B2 (en) | 2003-06-26 | 2010-02-02 | Lifesensors, Inc. | Methods and compositions for enhanced protein expression and purification |
| US8354455B2 (en) | 2004-08-26 | 2013-01-15 | Piramal Enterprises Limited and Apparao Satyam | Prodrugs containing novel bio-cleavable linkers |
| US8137695B2 (en) | 2006-08-18 | 2012-03-20 | Arrowhead Madison Inc. | Polyconjugates for in vivo delivery of polynucleotides |
| WO2008115319A2 (en) | 2007-02-07 | 2008-09-25 | Regents Of The University Of California | Conjugates of synthetic tlr agonists and uses therefor |
| US8426565B2 (en) | 2007-08-30 | 2013-04-23 | Walter And Eliza Hall Institute Of Medical Research | Dendritic cell marker and uses thereof |
| US9988431B2 (en) | 2007-08-30 | 2018-06-05 | The Walter And Eliza Hall Institute Of Medical Research | Dendritic cell marker and uses thereof |
| WO2009030996A1 (en) | 2007-09-05 | 2009-03-12 | Coley Pharmaceutical Group, Inc. | Triazole compounds as toll-like receptor (tlr) agonists |
| WO2009088401A2 (en) | 2007-09-24 | 2009-07-16 | Government Of The Usa, As Represented By The Secretary, Dept. Of Health And Human Services | Immunostimulatory combinations of tlr ligands and methods of use |
| US20130202707A1 (en) | 2008-02-13 | 2013-08-08 | Dana-Farber Cancer Institute, Inc. | Controlled Delivery of TLR Agonists in Structural Polymeric Devices |
| US20100029585A1 (en) | 2008-08-01 | 2010-02-04 | Howbert J Jeffry | Toll-like receptor agonist formulations and their use |
| WO2011044246A1 (en) | 2009-10-06 | 2011-04-14 | Cleveland Biolabs, Inc. | Use of toll-like receptor and agonist for treating cancer |
| US20120219615A1 (en) | 2010-10-01 | 2012-08-30 | The Trustees Of The University Of Pennsylvania | Therapeutic Use of a TLR Agonist and Combination Therapy |
| WO2013185052A1 (en) | 2012-06-08 | 2013-12-12 | Aduro Biotech | Compostions and methods for cancer immunotherapy |
| US9790494B2 (en) | 2012-09-14 | 2017-10-17 | Translate Bio Ma, Inc. | Multimeric oligonucleotide compounds having non-nucleotide based cleavable linkers |
| US9695212B2 (en) | 2012-12-13 | 2017-07-04 | Aduro Biotech, Inc. | Compositions comprising cyclic purine dinucleotides having defined stereochemistries and methods for their preparation and use |
| WO2014093936A1 (en) | 2012-12-13 | 2014-06-19 | Aduro Biotech, Inc. | Compositions comprising cyclic purine dinucleotides having defined stereochemistries and methods for their preparation and use |
| WO2014179335A1 (en) | 2013-04-29 | 2014-11-06 | Memorial Sloan Kettering Cancer Center | Compositions and methods for altering second messenger signaling |
| WO2014179760A1 (en) | 2013-05-03 | 2014-11-06 | The Regents Of The University Of California | Cyclic di-nucleotide induction of type i interferon |
| WO2014189805A1 (en) | 2013-05-18 | 2014-11-27 | Auro Biotech, Inc. | Compositions and methods for activating "stimulator of interferon gene"-dependent signalling |
| WO2014189806A1 (en) | 2013-05-18 | 2014-11-27 | Aduro Biotech, Inc. | Compositions and methods for inhibiting "stimulator of interferon gene" dependent signalling |
| WO2015017652A1 (en) | 2013-07-31 | 2015-02-05 | Memorial Sloan-Kettering Cancer Center | Sting crystals and modulators |
| WO2015077354A1 (en) | 2013-11-19 | 2015-05-28 | The University Of Chicago | Use of sting agonist as cancer treatment |
| WO2015185565A1 (en) | 2014-06-04 | 2015-12-10 | Glaxosmithkline Intellectual Property Development Limited | Cyclic di-nucleotides as modulators of sting |
| WO2016096577A1 (en) | 2014-12-16 | 2016-06-23 | Invivogen | Combined use of a chemotherapeutic agent and a cyclic dinucleotide for cancer treatment |
| WO2016096174A1 (en) | 2014-12-16 | 2016-06-23 | Invivogen | Fluorinated cyclic dinucleotides for cytokine induction |
| US10011630B2 (en) | 2014-12-16 | 2018-07-03 | Invivogen | Cyclic dinucleotides for cytokine induction |
| WO2016120305A1 (en) | 2015-01-29 | 2016-08-04 | Glaxosmithkline Intellectual Property Development Limited | Cyclic dinucleotides useful for the treatment of inter alia cancer |
| WO2016145102A1 (en) | 2015-03-10 | 2016-09-15 | Aduro Biotech, Inc. | Compositions and methods for activating "stimulator of interferon gene" -dependent signalling |
| WO2017027646A1 (en) | 2015-08-13 | 2017-02-16 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| WO2017027645A1 (en) | 2015-08-13 | 2017-02-16 | Merck Sharp & Dohme Corp. | Cyclic di-nucleotide compounds as sting agonists |
| WO2017075477A1 (en) | 2015-10-28 | 2017-05-04 | Aduro Biotech, Inc. | Compositions and methods for activating "stimulator of interferon gene"-dependent signalling |
| WO2017161349A1 (en) | 2016-03-18 | 2017-09-21 | Immune Sensor, Llc | Cyclic di-nucleotide compounds and methods of use |
| WO2017175147A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2017175156A1 (en) | 2016-04-07 | 2017-10-12 | Glaxosmithkline Intellectual Property Development Limited | Heterocyclic amides useful as protein modulators |
| WO2018100558A2 (en) | 2016-12-01 | 2018-06-07 | Takeda Pharmaceutical Company Limited | Cyclic dinucleotide |
| WO2018145020A1 (en) * | 2017-02-03 | 2018-08-09 | The Medical College Of Wisconsin, Inc. | Kras peptide vaccine compositions and method of use |
| US10195290B1 (en) | 2017-08-25 | 2019-02-05 | Codiak Biosciences, Inc. | Preparation of therapeutic exosomes using membrane proteins |
| WO2019099942A1 (en) | 2017-11-17 | 2019-05-23 | Codiak Biosciences, Inc. | Compositions of engineered exosomes and methods of loading luminal exosomes payloads |
| US20200347112A1 (en) | 2017-11-17 | 2020-11-05 | Codiak Biosciences, Inc. | Compositions of engineered exosomes and methods of loading luminal exosomes pay-loads |
| WO2019183578A1 (en) | 2018-03-23 | 2019-09-26 | Codiak Biosciences, Inc. | Extracellular vesicles comprising sting-agonist |
| WO2020101740A1 (en) | 2018-11-16 | 2020-05-22 | Codiak Biosciences, Inc. | Engineered extracellular vesicles and uses thereof |
| WO2020191361A2 (en) * | 2019-03-21 | 2020-09-24 | Codiak Biosciences, Inc. | Extracellular vesicles for vaccine delivery |
Non-Patent Citations (46)
| Title |
|---|
| "Crooke, Antisense drug Technology: Principles, Strategies and Applications", vol. 154,155, 2007, CRC PRESS |
| "DNA Cloning", vol. 1,2, 1985 |
| "Handbook Of Experimental Immunology", vol. I-IV, 1986, COLD SPRING HARBOR LABORATORY PRESS, article "Manipulating the Mouse Embryo" |
| "Molecular Cloning: A Laboratory Manual", 1992, COLD SPRINGS HARBOR LABORATORY |
| "The Dictionary of Cell and Molecular Biology", 1999, ACADEMIC PRESS |
| ADDERLEY, H. ET AL., EBIOMEDICINE, vol. 41, 2019, pages 711 - 716 |
| AUSUBEL ET AL.: "Current Protocols in Molecular Biology", 1989, COLD SPRING HARBOR LABORATORY PRESS |
| BENMOHAMED ET AL., IMMUNOLOGY, vol. 106, 2001, pages 113 - 121 |
| BORST, J. ET AL., NAT REV IMMUNOL, vol. 18, no. 10, October 2018 (2018-10-01), pages 635 - 647 |
| CELL REPORTS, vol. 11, 2015, pages 1018 - 1030 |
| COLLIN ET AL., IMMUNOLOGY, vol. 154, no. 1, 2018, pages 3 - 20 |
| CROTTY, S., NAT REV 7/M/MMMO/, vol. 15, no. 3, March 2015 (2015-03-01), pages 185 - 189 |
| DANON ET AL., EXP. GERONTOL., vol. 32, 1997, pages 633 - 641 |
| DIETHELM-OKITA ET AL., J INFEC DIS, vol. 181, 2000, pages 1001 - 1009 |
| DOBELI ET AL., J. BIOTECHNOLOGY, vol. 7, 1988, pages 199 - 216 |
| DORONINA ET AL., NATURE BIOTECHNOL, vol. 21, 2003, pages 778 - 84 |
| FAHY ET AL., BIOCHIM BIOPHYS ACTA, vol. 1811, no. 11, November 2011 (2011-11-01), pages 637 - 647 |
| FALUGI ET AL., EUR J IMMUNOL, vol. 31, 2001, pages 3816 - 3824 |
| FRESHNEY: "Immunochemical Methods In Cell And Molecular Biology", 1987, COLD SPRING HARBOR LABORATORY |
| GAYLE, J. BIOL. CHEM, vol. 268, 1993, pages 22105 - 22111 |
| JUO, PEI-SHOW: "the Concise Dictionary of Biomedicine and Molecular Biology", 2002, CRC PRESS |
| KHAN, M. ET AL., SCI REP, vol. 9, no. 1, September 2019 (2019-09-01), pages 13321 |
| MANTOVANI ET AL., TRENDS IMMUNOL, vol. 25, 2004, pages 677 - 686 |
| MARTINEZ ET AL., ANNU. REV. IMMUNOL., vol. 27, 2009, pages 451 - 483 |
| MEI ET AL., BLOOD, vol. 116, 2010, pages 270 - 79 |
| MOSSER ET AL., NATURE REV, vol. 8, 2008, pages 958 - 969 |
| MOSSER, J., LEUKOCYTE BIOL, vol. 73, 2003, pages 209 - 212 |
| PAPAPETROU ET AL., GENE THERAPY, vol. 12, 2005, pages S118 - S130 |
| PERBAL: "A Practical Guide To Molecular Cloning", 1984, ACADEMIC PRESS, INC., article "Methods In Enzymology" |
| POLLARD, NATURE REV, vol. 9, 2009, pages 259 - 270 |
| REDDY ET AL., CANCER CHEMOTHER. PHARMACOL., vol. 58, 2006, pages 229 - 36 |
| ROMAN, M. ET AL., MOLECULAR CANCER, vol. 17, 2018, pages 33 |
| RON ET AL., J. BIOL. CHEM., vol. 268, 1993, pages 2984 - 2988 |
| ROSS N ET AL: "Induction of Potent Systemic and Tissue Resident Immune Responses by ExoVACC<TM>: A Novel Exosome-Based Vaccine Platform", MOLECULAR THERAPY, vol. 28, no. 4, Suppl. 1, 66, 28 April 2020 (2020-04-28), 23rd Annual Meeting of the American Society for Gene and Cell Therapy; virtual; 12-15 May 2020, pages 34, XP055880679, ISSN: 1525-0016, DOI: 10.1016/j.ymthe.2020.04.019 * |
| SENTI ET AL., PNAS, vol. 105, no. 46, 2008, pages 17908 |
| SOOS T ET AL: "EXOVACC (TM): A Novel Exosome-Based Vaccine Platform That Induces Robust, Tunable Cellular and Humoral Immune Responses in Animal Models", MOLECULAR THERAPY, vol. 29, no. 4, Suppl. 1, 721, 27 April 2021 (2021-04-27), 24th Annual Meeting of the American Society of Gene and Cell Therapy; 11-14 May 2021, pages 350 - 351, XP055880686, ISSN: 1525-0016 * |
| STOUT ET AL., J. IMMUNOL., vol. 175, 2005, pages 342 - 349 |
| TAN, C. ET AL., WORLD J GASTROENTEROL, vol. 18, no. 37, 2012, pages 5171 - 5180 |
| TROUT ET AL., PROC. NATL. ACAD. SCI. USA, vol. 79, 1982, pages 626 - 629 |
| UMEMOTO ET AL., INT. J. CANCER, vol. 43, 1989, pages 677 - 684 |
| YANG ET AL., J. PHARMACOL. EXP. THER., vol. 321, 2007, pages 462 - 8 |
| YUAN ET AL., ACTA BIOMATER, vol. 4, 2008, pages 1024 - 37 |
| ZEITOUNI, D. ET AL., CANCERS, vol. 8, no. 4, 2016, pages 45 |
| ZHANG ET AL., ACTA BIOMATER, vol. 6, 2007, pages 838 - 50 |
| ZHOU ET AL., BIOMACROMOLECULES, vol. 12, 2011, pages 1460 - 7 |
| ZULOFF-SHANI ET AL., TRANSFUS. APHER. SCI., vol. 30, 2004, pages 163 - 167 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025080869A1 (en) * | 2023-10-11 | 2025-04-17 | The Regents Of The University Of California | Pancreatic cancer immunotherapy using cationic lipid and polymer nanocarriers for co-delivery of kras neoantigens and sting and/or other immunomodulators to boost the cancer immunity cycle and treat liver metastasis |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US11992527B2 (en) | Extracellular vesicles for vaccine delivery | |
| US20240108747A1 (en) | Extracellular vesicle conjugates and uses thereof | |
| US20240082389A1 (en) | Methods of producing extracellular vesicles | |
| US20250115904A1 (en) | Extracellular vesicle-aso constructs targeting stat6 | |
| US20220251200A1 (en) | Extracellular vesicles targeting t cells and uses thereof | |
| US20240099973A1 (en) | Extracellular vesicle compositions | |
| WO2020041720A1 (en) | Extracellular vesicles targeting dendritic cells and uses thereof | |
| US20230114434A1 (en) | Extracellular vesicles for treating neurological disorders | |
| US20250114445A1 (en) | Extracellular vesicles for therapy | |
| US20230002764A1 (en) | Extracellular vesicle-aso constructs targeting cebp/beta | |
| US20230193274A1 (en) | Extracellular vesicles with stat3-antisense oligonucleotides | |
| US20240189430A1 (en) | Extracellular vesicle linked to a biologically active molecule via an optimized linker and an anchoring moiety | |
| WO2022076596A1 (en) | Extracellular vesicle-aso constructs targeting stat6 | |
| WO2022066883A1 (en) | Extracellular vesicles comprising kras antigens and uses thereof | |
| US20230220068A1 (en) | Anti-transferrin extracellular vesicles | |
| US20260041764A1 (en) | Extracellular vesicles for vaccine delivery | |
| US20250382613A1 (en) | Methods of using extracellular vesicle-aso targeting stat6 | |
| US20250381292A1 (en) | Methods of administering a sting agonist | |
| WO2024118592A2 (en) | Methods of using extracellular vesicle-aso targeting stat6 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 21791549 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 21791549 Country of ref document: EP Kind code of ref document: A1 |