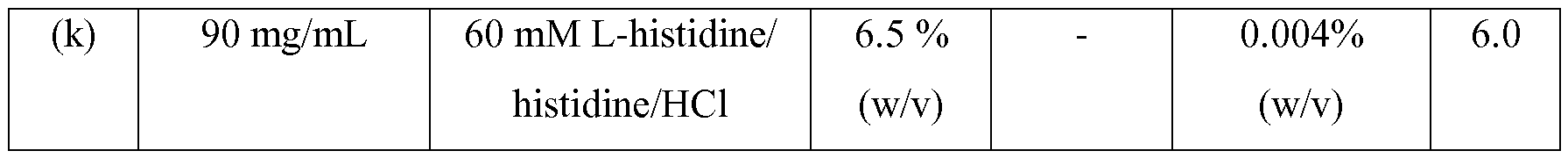WO2019106206A1 - Stable, low viscosity, high concentration liquid formulations of an anti-il-12/23p40 antibody - Google Patents
Stable, low viscosity, high concentration liquid formulations of an anti-il-12/23p40 antibody Download PDFInfo
- Publication number
- WO2019106206A1 WO2019106206A1 PCT/EP2018/083353 EP2018083353W WO2019106206A1 WO 2019106206 A1 WO2019106206 A1 WO 2019106206A1 EP 2018083353 W EP2018083353 W EP 2018083353W WO 2019106206 A1 WO2019106206 A1 WO 2019106206A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- pharmaceutical composition
- polysorbate
- antibody
- histidine buffer
- sucrose
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/24—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against cytokines, lymphokines or interferons
- C07K16/244—Interleukins [IL]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
- A61K39/39591—Stabilisation, fragmentation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Definitions
- the present invention relates to stable, low viscosity, high concentration liquid
- compositions of an anti-IL-l2/23p40 antibody comprising at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, and either i) 50-70 mM of a histidine buffer and no inorganic salt, or ii) 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt, or iii) 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- cytokine interleukin- 12 The heterodimeric cytokine interleukin- 12 (IL-12) consists of disulfide bonded, glycosylated polypeptide chains with a weight of 35 and 40 kD. IL-12 is synthesized and secreted by antigen presenting cells, including dendritic cells, monocytes, macrophages, B cells, Langerhans cells and keratinocytes as well as natural killer (NK) cells, and is involved in a variety of biological processes. It is also referred to as NK cell stimulatory factor (NKSF), T- cell stimulating factor, cytotoxic T-lymphocyte maturation factor, and EBV-transformed B- cell line factor (Curfs et al., 1997). IL-12 can bind to IL-12 receptor, which is expressed on the plasma membrane of cells (e.g.,
- T cells T cells, NK cell
- IL-12 binding to IL-12 receptor can stimulate the proliferation of pre-activated T cells and NK cells, enhance the cytolytic activity of cytotoxic T cells (CTL), NK cells and lymphokine activated killer (LAK) cells, induce production of gamma interferon (IFN-g) by T cells and NK cells, and induce differentiation of naive ThO cells into Thl cells that produce IFN-g and IL-2 (Trinchieri, 1995).
- CTL cytotoxic T cells
- LAK lymphokine activated killer
- IL-12 An especially key role of IL-12 is in the generation of cytolytic cells, such as NKs and CTLs, and in mounting a cellular immune response, such as a Thl cell mediated immune response, making IL-12 vital in the generation and regulation of both protective immunity, for example eradication of infections, and pathological immune responses, such as autoimmunity (Hendrzak and Brunda, 1995).
- cytolytic cells such as NKs and CTLs
- a cellular immune response such as a Thl cell mediated immune response
- the p40 protein subunit of IL-12 can form another cytokine, interleukin 23 (IL-23) by associating with another protein subunit, pl9 (Oppman et 1., 2000).
- IL-23 similarly to IL-12, can bind to a two-chain receptor complex, which mediates an intracellular signal.
- these two cytokines have distinct biological functions (Langrish wet al., 2005).
- IL-23 mediates IL-17 production by T-cells (Parham et al., 2002; Aggarwal et al., 2003).
- IL-12 and IL-23 can modulate immune responses to be enhanced, suppressed or prevented.
- abnormal IL-12 and IL-23 regulation is an important factor in many immune-mediated diseases, making IL-12 and IL- 23 promising targets in treatment.
- An anti-IL-l2/IL-23p40 antibody ustekinumab, also known as CNTO 1275 or by its trade name Stelara ® , is approved in Europe and the United States for the treatment of moderate to severe plaque psoriasis, psoriatic arthritis, and Crohn’s disease.
- Ustekinumab as Stelara ® is available as a solution for injection (45 and 90 mg) in vials or prefilled syringes and as a concentrate (130 mg) to make a solution for infusion (drip) into a vein.
- Stelara ® As Stelara ® , ustekinumab is formulated at 90 mg/ml with 0.48 mg/ml L-histidine, 0.74 mg/ml L-histidine hydrochloride monohydrate, 0.04 mg/ml polysorbate 80, and 76 mg/ml sucrose.
- ustekinumab is given by subcutaneous injection.
- the usual dose is 45 mg, whereas the dose in children depends on bodyweight.
- the first injection is followed by a further injection four weeks later, and then an injection every 12 weeks.
- Patients weighing over 100 kg should be given ustekinumab in 90-mg doses for psoriasis, and this should also be considered for psoriatic arthritis.
- treatment is started with ustekinumab concentrate.
- the infusion lasts at least one hour and the dose depends on the patient’s bodyweight.
- Eight weeks after the first infusion patients should receive 90 mg ustekinumab by subcutaneous injection. Patients then continue with ustekinumab given subcutaneously every 8 or 12 weeks depending on response to treatment.
- Subcutaneous administration of concentrated antibody solutions is preferable for patients with chronic diseases such as the ones that ustekinumab is approved for, because frequent dosing may be needed over their lifetimes.
- Subcutaneous injections can be self-administered by the patients using ready to use prefilled syringes or pens /auto-injectors and delivery devices. Such devices are advantageous for better patient comfort.
- the concentrated antibody drug solutions contained in these devices may provide the additional benefit of longer intervals between injections (and more effective pharmacokinetic profile for the antibody).
- not more than 1 mL of drug product solution can be administered as a single bolus dose via a prefilled syringe due to volume restrictions for dose administration in the subcutaneous space.
- the total volume and duration of administration is dictated by the concentration of the monoclonal antibody in the dosing solution.
- concentration of the monoclonal antibody in the dosing solution In order to achieve higher dose administration in smaller volumes, either for infusion or bolus administration, high concentrations of monoclonal antibodies in solution are required, as is the case with ustekinumab at 90 mg/ml.
- highly concentrated antibody solutions can demonstrate high viscosities, which is problematic because high viscosity may increase the injection time and pain at the site of injection, adversely affecting patient compliance.
- high viscosity requires the use of thicker needles, which makes the injection more painful, whereas low viscosity formulations can be injected using, e.g., 29G, 30G, 31G, or 34G needles or even thinner needles due to better syringeability,.
- High viscosity of a concentrated antibody solution also poses difficulties during bioprocessing of the drug substance. During downstream ultrafiltration and diafiltration steps, high viscosity of the antibody solution may increase the back pressure of the pump and decrease the transmembrane flux, which may increase the processing time, destabilize the drug substance and increase manufacturing costs (Tomar et al., 2016).
- anti-IL-l2/23p40 antibodies that are stable, low viscosity, high concentration liquid pharmaceutical compositions of an anti- IL-l2/23p40 antibody suitable for intravenous or subcutaneous administration in the treatment of immune-mediated diseases such as e.g. plaque psoriasis, psoriatic arthritis, and Crohn’s disease.
- a stable, high concentration liquid pharmaceutical composition of an anti-IL-l2/23p40 antibody comprising at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, and either i) 50-70 mM of histidine and no inorganic salt, or ii) 25-35 mM of histidine and 10-15 mM of an inorganic salt, or iii) 5-10 mM of histidine and 20-40 mM, preferably 25-30 mM, of an inorganic salt has a surprisingly low level of viscosity and is therefore particularly suitable for subcutaneous administration and for use in treating immune-mediated diseases in which IL12 and/or IL-23 play a role.
- a further advantage of the stable, high concentration liquid pharmaceutical composition according to the present invention is that bioprocessing of the formulations is made more convenient. Downstream ultrafiltration and diafiltration steps are made easier by decreased back pressure of the pump and increased transmembrane flux, which reduces the processing time, results in a more stable antibody solution, and decreases manufacturing costs.
- the present invention provides a stable, low viscosity, liquid pharmaceutical composition
- a stable, low viscosity, liquid pharmaceutical composition comprising
- the anti-IL-l2/23p40 antibody may be present in a concentration of from 80 mg/ml to 100 mg/ml, preferably in a concentration of 90 mg/ml.
- the sugar comprised in the composition is sucrose.
- the non-ionic surfactant is present in a concentration of 0.04 mg/1.
- the non-ionic surfactant can be polysorbate 20 or 80.
- the inorganic salt may be sodium chloride.
- the composition has a pH of 5.8-6.2.
- the pharmaceutical composition comprises
- the pharmaceutical composition comprises
- the pharmaceutical composition comprises
- the antibody may be one that substantially neutralizes at least one biological activity of at least one IL-12 and/or IL-23 protein, and preferably is ustekinumab.
- the pharmaceutical composition is suitable for administration by at least one mode selected from the group consisting of parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic, intrapericardiac, intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and transdermal.
- the mode of administration the pharmaceutical composition is suitable for is subcutaneous administration.
- the mode of administration the pharmaceutical composition is suitable for is intravenous administration.
- the pharmaceutical composition may be used in treating an immune-mediated disease in which IL-12 and/or IL-23 play a role.
- the immune-mediated disease is selected from the group of plaque psoriasis, psoriatic arthritis, Crohn’s disease, ankylosing spondylitis, spondyloarthritis, and ulcerative colitis.
- the present invention also relates to a prefilled syringe containing the pharmaceutical composition as defined herein.
- the syringe is suitable for containing or administering, as defined herein, the composition as defined herein.
- composition refers to any composition comprising a chemical substance or active ingredient which composition is intended for use in the medical cure, treatment, or prevention of disease and which is in such a form as to permit the active ingredient to be effective.
- a pharmaceutical composition does not contain excipients which are unacceptably toxic to a subject to which the composition is to be administered.
- the pharmaceutical compositions are sterile, i.e. aseptic and free from all living microorganisms and their spores.
- the pharmaceutical composition used in the present invention is liquid and stable.
- the pharmaceutically active agent e.g. the anti-IL-l2/23p40 antibody
- the liquid pharmaceutical composition used in the invention is at no point lyophilised, i.e. the production method does not contain a lyophilisation step and the composition is not lyophilised for storage.
- Liquid compositions can be stored in vials, IV bags, ampoules, cartridges, and prefilled or ready-to-use syringes.
- the liquid composition is an aqueous liquid composition.
- An aqueous liquid composition comprises water as solvent.
- a “stable" liquid composition is one in which the anti-IL-l2/23p40 antibody contained therein essentially retains its physical stability and/or chemical stability and/or biological activity upon storage for a certain period. Preferably, the composition essentially retains upon storage its physical and chemical stability, as well as its biological activity.
- Various analytical techniques for measuring protein stability are available in the art and are reviewed, for example, in Peptide and Protein Drug Delivery, 247-301, Vincent Lee Ed, Marcel Dekker, Inc, New York, New York, Pubs (1991) and Jones, Adv Drug Delivery Rev, 1993, 10:29-90. For example, stability can be measured at a selected temperature for a selected time period.
- Stability can be evaluated qualitatively and/or quantitatively in a variety of different ways, including evaluation of aggregate formation (for example using size exclusion chromatography, by measuring turbidity, and/or by visual inspection), by assessing charge heterogeneity using cation exchange chromatography or capillary zone
- electrophoresis amino-terminal or carboxy-terminal sequence analysis
- mass spectrometric analysis SDS-PAGE analysis to detect aggregated or fragmented molecules
- peptide map for example tryptic or LYS-C
- the pharmaceutical composition is stable at a temperature of about 40°C for at least 1 to 2 weeks, and/or is stable at a temperature of about 5°C for at least 9 months, and/or is stable at a temperature of about 25°C for at least two weeks to one month.
- the formulation is preferably stable following freezing (to, e.g., -20°C) and thawing of the formulation at 25°C as described in the examples herein, for example following 1, 2, 3, 4 or 5 cycles of freezing and thawing.
- the percentage of high molecular weight species relative to the total amount of the anti-lL- 12/23r40 antibody as measured by size exclusion chromatography is not more than 5%, preferably not more than 3% or 2%, more preferably not more than 1% and most preferably not more than 0.8% or 0.2% after storage of the composition at 5°C for 1 month.
- the percentage of fragments relative to the total amount of the anti-lL-l2/23p40 antibody as measured by size exclusion chromatography is not more than 5%, preferably not more than 3% or 2%, more preferably not more than 1% or 0.5% and most preferably not more than 0.4% or 0.2% after storage of the composition at 5°C for 1 month.
- low- viscosity is used herein to indicate that the viscosity of the pharmaceutical composition is less than about 20 cP, preferably below 15 cP, and most preferably below 10 cP at about 25° C. In some embodiments, the viscosity is less than about 10 cP, 9cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- measurements of viscosity typically are performed at shear rates from about 0.6 s' 1 to about 450 s"l, and preferably from about 2 s"l to about 400 s"l, when measured using a cone and plate viscometer.
- viscosity can be measured at shear rates from about 3 s"l to about 55,000 s"l, and preferably from about 20 s"l to about 2,000 s"l when measured using a micro fluidic viscometer.
- compositions of the invention have an osmolality of 250-350 mOsm/kg, preferably of 280-320 mOsm/kg.
- the term “immunoglobulin” is used herein in the broadest sense and includes full length antibodies, genetically engineered antibodies, recombinant antibodies, multivalent antibodies, monoclonal antibodies, polyclonal antibodies, bispecific antibodies, multispecific antibodies, chimeric antibodies, humanized antibodies, fully human antibodies, as well as fragments of such antibodies as long as they remain functional and exhibit the desired biological activity.
- The“biological activity” of an antibody refers to the ability of the antibody to bind to antigen and result in a biological response which can be measured in vitro or in vivo.
- a full length antibody comprises an antigen-binding variable region of the light (V L ) and heavy chain (VH), a light chain constant region (CL) and heavy chain constant domains Cnl , CH2 and CH3.
- antibody fragment or“antigen-binding fragment” is used herein in the broadest sense and comprises a portion of a full length antibody, preferably comprising the antigen binding or variable region thereof.
- An antibody fragment retains the original specificity of the parent immunoglobulin.
- antibody fragments include, e.g., Fab, Fab', F(ab')2, and Fv fragments, diabodies, linear antibodies, single-chain antibody molecules, and multispecific antibodies formed from antibody fragment(s).
- the antibody fragment is a Fab fragment.
- A“monoclonal antibody” is an antibody that is specific for a single epitope of an antigen, i.e. directed against a single determinant on an antigen. Methods for producing monoclonal antibodies are known to the person skilled in the art.
- recombinant antibody refers to all antibodies prepared, expressed, created or isolated by recombinant means, such as antibodies isolated from a transgenic host cell, such as e.g. a NS0 or CHO cell, or from an animal transgenic for immunoglobulin genes, or antibodies expressed using recombinant expression vectors transfected into a host cell, such as e.g. SP 2/0 mouse myeloma cells.
- A“humanised antibody” is a human antibody wherein the antigen binding portion (CDR) is derived from non-human species, such as a mouse, and thus has a different specificity compared to the parent immunoglobulin.
- the CDR protein sequences can be modified to increase their similarities to antibody variants produced naturally in humans.
- anti-IL-l2/23p40 antibody refers to an antibody or antibody fragment, such as a Fab or a scFV fragment, which specifically interacts with the p40 protein subunit of IL-12 and/or IL-23 VEGF and neutralizes one or more of the biological activities of these cytokines, e.g.
- NK cells cytotoxic T cells
- LAK lymphokine activated killer cells
- IFN-g gamma interferon
- T cells and NK cells induce differentiation of naive ThO cells into Thl cells that produce IFN-g and IL-2, in mounting Thl cell mediated immune responses, or in mediating IL-17 production in T cells.
- IFN-g gamma interferon
- Anti-IL-l2/23p40 antibodies act, e.g., by interfering with the binding of IL-12 and/or IL-23 to their cellular receptors, and by interfering with the biological functions of IL-12 and/or IL- 23 after binding to their cellular receptor.
- Anti-ILl2/23p40 antibodies include, e.g., ustekinumab as described in, for example, WO 02/12500.
- the anti-ILl2/23p40 antibody or antigen-binding fragment thereof present in the pharmaceutical composition used in the present invention is ustekinumab or an antigen-binding fragment thereof.
- Ultrakinumab is a fully human IgGlK monoclonal antibody directed against the IL- 12/23r40 subunit having the heavy and light chain variable domain sequences as described in SEQ ID Nos. 7 and 8 of WO 02/12500 and Benson et al. (2011) MAbs 3(6): 535-545.
- the CAS number of ustekinumab is 815610-63-0.
- Ustekinumab binds with high specificity to the p40 protein subunit of interleukin (IL)-l2 and IL-23, thereby suppressing IL-12- and IL-23- mediated immune responses and inflammation It has been approved in Europe, the United States of America, and Canada for the treatment of moderate to severe plaque psoriasis, active psoriatic arthritis, and moderately to severely active Crohn’s disease. It is produced in a murine myeloma cell line using recombinant DNA technology as described in WO 02/12500 and Benson et al. (2011) MAbs 3(6): 535-545.
- the present commercial ustekinumab formulation contains L-histidine/histidine hydrochloride, sucrose, polysorbate 80, and water for injection and is supplied in a concentration of 90 mg/ml.
- the concentration of the anti-ILl2/23p40 antibody in the pharmaceutical compositions is at least 80 mg/ml, typically 80-100 mg/ml, preferably 85-95 mg/ml, most preferably 90 mg/ml.
- compositions of the invention permit the anti-ILl2/23p40 antibody comprised therein to exhibit its activity of neutralizing at least one of the biological functions of IL-12 and/or IL-23.
- sugar refers to an organic compound comprising only carbon, hydrogen, and oxygen, usually with a hydrogemoxygen atom ratio of 2: 1 and the empirical formula C m (H 2 0) n .
- the term“sugar” includes mono-, di-, oligo- and polysaccharides.
- carbohydrates include glucose, fructose, galactose, xylose, ribose, sucrose, mannose, lactose, maltose, trehalose, starch, and glycogen.
- sugar alcohols such as glycerol, mannitol, sorbitol, and xylitol
- sugar acids e.g.
- aldonic acids such as ascorbic acid, aldaric acids such as tartaric acid
- reducing sugars e.g. glucose, glyceraldehydes, galactose, lactose, and maltose
- amino sugars e.g. N-acetylglucosamine, galactosamine, glucosamine, and sialic acid
- sulfoquinovose a sulphonic acid derivative of glucose.
- the sugar of the invention is sucrose.
- A“surfactant” as used herein refers to an amphiphilic compound, i.e.
- A“non-ionic surfactant” has no charged groups in its head. The formation of insoluble particles during freeze/thaw cycles of antibody-containing compositions can be remarkably inhibited by addition of surfactants. Examples of“non-ionic surfactants” include e.g.
- polyoxyethylene glycol alkyl ethers such as octaethylene glycol monododecyl ether, pentaethylene glycol monododecyl ether; polyoxypropylene glycol alkyl ethers; glucoside alkyl ethers, such as decyl glucoside, lauryl glucoside, octyl glucoside; polyoxyethylene glycol octylphenol ethers, such as triton X-100; polyoxyethylene glycol alkylphenol ethers, such as nonoxynol- 9; glycerol alkyl esters, such as glyceryl laurate; polyoxyethylene glycol sorbitan alkyl esters, such as polysorbate; sorbitan alkyl esters, such as spans; cocamide MEA, cocamide DEA, dodecyldimethylamine oxide; block copolymers of polyethylene glycol and polypropylene glycol, such as polox
- Preferred non-ionic surfactants for use in the pharmaceutical compositions of the present invention are polysorbates such as polysorbate 20, 40, 60 or 80 or poloxamer 188, and especially polysorbate 20 (i.e. Tween 20) or polysorbate 80. Most preferred is polysorbate 80.
- the concentration of the non-ionic surfactant is in the range of 0.002 to 0.006% (w/v), preferably in the range of 0.0025 to 0.0055% (w/v), more preferably in the range of 0.003 to 0.005% (w/v) and most preferably it is 0.004% (w/v) relative to the total volume of the composition.
- the non-ionic surfactant is polysorbate 20 with a concentration in the range of 0.002 to 0.006% (w/v), more preferably in the range of 0.003 to 0.005% (w/v) and most preferably it is 0.004% (w/v), relative to the total volume of the composition.
- the non-ionic surfactant is polysorbate 80 with a concentration in the range of 0.002 to 0.006% (w/v), more preferably in the range of 0.003 to 0.005% (w/v) and most preferably it is 0.004% (w/v), relative to the total volume of the composition.
- the non-ionic surfactant is polysorbate 20 with a concentration of 0.004% (w/v), relative to the total volume of the composition.
- the non-ionic surfactant is polysorbate 80 with a concentration of 0.004% (w/v), relative to the total volume of the composition.
- A“buffer” is an aqueous solution consisting of a mixture of a weak acid and its conjugate base or vice versa which resists changes in its pH and therefore keeps the pH at a nearly constant value.
- the buffer of the present invention preferably has a pH in the range from about 5.5 to about 7.0, preferably from about 5.7 to about 6.8, more preferably from about 5.8 to 6.5 and most preferably has a pH of about 5.8 to 6.2, such as, most preferably, a pH of 6.0.
- the buffer used in the present invention is a "histidine-containing buffer” or“histidine buffer”, which terms are used interchangeably herein and refer to a buffer comprising histidine.
- histidine buffers include histidine chloride, histidine hydrochloride, histidine acetate, histidine phosphate, and histidine sulphate.
- the preferred histidine buffer of the invention further comprises L-histidine. Even more preferably, the histidine buffer of the invention comprises histidine hydrochloride, most preferably it comprises histidine hydrochloride and L-histidine.
- the histidine buffer or histidine hydrochloride buffer or histidine hydrochloride/L-histidine buffer has a pH in the range from about 5.5 to about 7.0, preferably from about 5.7 to about 6.8, more preferably from about 5.8 to 6.5 and most preferably has a pH of about 5.8 to 6.2, such as, most preferably, a pH of 6.0.
- the histidine buffer has a concentration in the range of 50 mM to 70 mM, preferably of 55 mM to 65 mM, and most preferably of 60 mM.
- the histidine buffer of this embodiment is histidine hydrochloride/L-histidine.
- the histidine buffer has a concentration in the range of 25 mM to 35 mM, preferably of 28 mM to 32 mM, and most preferably of 31 mM. Most preferably the histidine buffer of this embodiment is histidine hydrochloride/L- histidine. In a third particularly preferred embodiment, the histidine buffer has a concentration in the range of 5 mM to 10 mM, preferably of 6 mM to 8 mM, and most preferably of 6.6 mM. Most preferably the histidine buffer of this embodiment is histidine hydrochloride/L- histidine.
- the buffer is histidine hydrochloride/L-histidine with the concentrations specified above and with a pH of 6.0.
- compositions of the present invention may be prepared by dissolving L- histidine/histidine hydrochloride, the sugar, preferably sucrose, and, if present, the inorganic salt, preferably sodium chloride, in water before adding the non-ionic surfactant, preferably polysorbate 20 or polysorbate 80 and then adding the anti-IL-l2/23p40 antibody.
- an“inorganic salt” refers to a ionic compound which has osmoregulatory properties.
- An inorganic salt such as sodium chloride (NaCl) can dissociate in solution into its constituent ions, i.e. NaCl dissociates into Na + and Cl ions, which both affect the osmotic pressure, i.e. the osmolality, of the solution.
- Preferred inorganic salts for use in the pharmaceutical formulation of the present invention are potassium chloride, calcium chloride, sodium chloride, sodium phosphate, potassium phosphate and sodium bicarbonate.
- the inorganic salt is a sodium salt, more preferably it is sodium chloride (NaCl).
- the inorganic salt has a concentration in the range of 10 mM to 15 mM, preferably of 12 mM to 13 mM, and most preferably of 12.5 mM.
- the histidine buffer has a concentration of 25 to 35 mM.
- the inorganic salt of this embodiment is NaCl.
- the inorganic salt has a concentration in the range of 20 mM to 30 mM, preferably of 22.5 mM to 27.5 mM, and most preferably of 25 mM.
- the histidine buffer has a concentration of 5 to 10 mM.
- the inorganic salt of this embodiment is NaCl .
- compositions of the invention preferably have a pH in the range from about 5.5 to about 7.0, preferably from about 5.7 to about 6.8, more preferably from about 5.8 to 6.5 and most preferably has a pH of about 5.8 to 6.2, such as, most preferably, a pH of 6.0.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C
- the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25- 30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients , i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no iurther excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e.
- the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition used in the present invention may further contain diluents, solubilising agents, isotonising agents, excipients, pH- modifiers, soothing agents, buffers, sulphur-containing reducing agents, antioxidants or the like.
- the pharmaceutical composition is suitable for parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic,
- intrapericardiac intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and/or transdermal administration.
- the pharmaceutical administration is suitable for intravenous and/or subcutaneous administration.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL- 12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL- 12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti- IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti- IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80- 100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients , i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- an "immune-mediated disease in which IL-12 and/or IL-23 play a role” is a disease characterized by inappropriate or elevated immune functions mediated by IL-12 and/or IL- 23, e.g. via their activity in proliferation of pre-activated T cells and NK cells, in enhancing the cytolytic activity of cytotoxic T cells (CTL), NK cells and lymphokine activated killer (LAK) cells, in inducing production of gamma interferon (IFN-g) by T cells and NK cells, induce differentiation of naive ThO cells into Thl cells that produce IFN-g and IL-2, in mounting Thl cell mediated immune responses, or in mediating IL-17 production in T cells.
- CTL cytotoxic T cells
- LAK lymphokine activated killer
- immune-mediated disease in which IL-12 and/or IL-23 play a role are, for example, rheumatoid arthritis, juvenile rheumatoid arthritis, systemic onset juvenile rheumatoid arthritis, psoriatic arthritis, ankylosing spondilitis, gastric ulcer, seronegative arthropathies, osteoarthritis, osteolysis, aseptic loosening of orthopedic implants, inflammatory bowel disease, ulcerative colitis, systemic lupus erythematosus,
- antiphospholipid syndrome iridocyclitis/uveitis/optic neuritis, idiopathic pulmonary fibrosis, systemic vasculitis/ admirer's granulomatosis, sarcoidosis, orchitis/vasectomy reversal procedures, allergic/atopic diseases, asthma, allergic rhinitis, eczema, allergic contact dermatitis, allergic conjunctivitis, hypersensitivity pneumonitis, transplants, organ transplant rejection, graft- versus-host disease, systemic inflammatory response syndrome, sepsis syndrome, gram positive sepsis, gram negative sepsis, culture negative sepsis, fungal sepsis, neutropenic fever, urosepsis, meningococcemia, trauma/hemorrhage, bums, ionizing radiation exposure, acute pancreatitis, adult respiratory distress syndrome, rheumatoid arthritis, alcohol-induced hepatitis, chronic inflammatory pathologies,
- hemachromatosis alpha- 1 -antitrypsin deficiency, diabetic retinopathy, hashimoto's thyroiditis, osteoporosis, hypothalamic-pituitary-adrenal axis evaluation, primary biliary cirrhosis, thyroiditis, encephalomyelitis, cachexia, cystic fibrosis, neonatal chronic lung disease, chronic obstructive pulmonary disease (COPD), familial hematophagocytic lymphohistiocytosis, dermatologic conditions, psoriasis, alopecia, nephrotic syndrome, nephritis, glomerular nephritis, acute renal failure, hemodialysis, uremia, toxicity, preeclampsia, okt3 therapy, anti-cd3 therapy, cytokine therapy, chemotherapy, radiation therapy (e.g., including but not limited to, asthenia, anemia, cachexia, and the like
- the pharmaceutical composition may be used in the treatment of any such immune- mediated disease in which IL-12 and/or IL-23 play a role.
- the pharmaceutical composition is preferably for use in the treatment of plaque psoriasis, psoriatic arthritis, Crohn’s disease, ankylosing spondylitis, spondyloarthritis, and ulcerative colitis.
- the plaque psoriasis is preferably moderate to severe plaque psoriasis.
- the psoriatic arthritis is preferably active psoriatic arthritis.
- the Crohn’s disease is preferably moderately to severely active Crohn’s disease.
- the pharmaceutical composition is preferably given by subcutaneous injection.
- the preferred dose is 45 mg of the anti-IL-l2/23p40 antibody, whereas the preferred dose in children depends on bodyweight.
- the first injection is followed by a further injection four weeks later, and then an injection every 12 weeks.
- the pharmaceutical composition is preferably given in 90 mg doses for psoriasis, and this could also be done for patients weighing over lOOkg having psoriatic arthritis.
- treatment is preferably started with an intravenous infusion of an anti-lLl2/23p40 antibody prepared from concentrate that lasts at least one hour, wherein the administered dose preferably depends on the patient’s bodyweight.
- an intravenous infusion of an anti-lLl2/23p40 antibody prepared from concentrate that lasts at least one hour, wherein the administered dose preferably depends on the patient’s bodyweight.
- eight weeks after the first infusion patients then receive the pharmaceutical compositions at a dose of 90 mg anti-lLl2/23p40 antibody by subcutaneous injection.
- patients then continue receiving subcutaneously administered pharmaceutical composition every 8 or 12 weeks depending on response to treatment.
- the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-lL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-lL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-lL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL- 12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL- 12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80- 100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients , i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- compositions of the present invention can be supplied in sealed and sterilized plastic, glass, or other suitable containers having a defined volume such as vials, ampoules or syringes or a large volume such as bottles.
- liquid pharmaceutical composition is supplied in a prefilled syringe.
- a “ready-to-use syringe” or“prefilled syringe” is a syringe which is supplied in a filled state, i.e. the pharmaceutical composition to be administered is already present in the syringe and ready for administration.
- Prefilled syringes have many benefits compared to separately provided syringe and vial, such as improved convenience, affordability, accuracy, sterility, and safety.
- prefilled syringes results in greater dose precision, in a reduction of the potential for needle sticks injuries that can occur while drawing medication from vials, in pre-measured dosage reducing dosing errors due to the need to reconstituting and/or drawing medication into a syringe, and in less overfilling of the syringe helping to reduce costs by minimising drug waste.
- the prefilled syringe containing the pharmaceutical composition is suitable for parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic, intrapericardiac, intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and/or transdermal administration.
- compositions are suitable for intravenous and/or subcutaneous
- the prefilled syringe containing the pharmaceutical composition suitable for one of the above-specified administrations is suitable for use in the above-specified treatment of an immune-mediated disease in which IL-12 and/or IL-23 play a role.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP,
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP,
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL- 12/23r40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL- 12/23r40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10- 15 mM of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e.
- the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
- the pharmaceutical composition has a pH of 5.8-6.2.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0.
- the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
- the final ustekinumab concentration of all samples was 10 mg/mL.
- the different ustekinumab formulations were stored at 5 °C for up to 6 months, or at 25°C / 60% relative humidity or 40°C / 75% relative humidity for up to 3 months. Additionally, ustekinumab in the different formulations was stressed by five freeze/ thaw cycles or by shaking samples at 600rpm, 25°C / 60% relative humidity.
- Ustekinumab in the Stelara ® formulation buffer system was included in the stability program as control sample (a). Table 1 : Detailed information of all formulations used in this study.
- Capillary isoelectric focusing is a method used for the determination of the identity and purity of Ustekinumab.
- cIEF is a quantitative high-resolution analytical technique that allows the separation of proteins, protein glycoforms and other protein isoforms, differing in their net-charge based on their isoelectric point (pi). Modifications like deamidation or N- terminal pyroglutamic acid formation, leading to a change of net charge can be detected.
- the cIEF separation consists of two steps, focusing and mobilization.
- the capillary is filled with the sample.
- the cIEF sample is a mixture of ampholytes, stabilizers, pi markers and the protein sample. Focusing is performed by submerging one capillary end in anolyte (H+) and one capillary end in catholyte (OFF) and applying voltage.
- the cathodic stabilizer (Arg) migrates toward the cathodic side of the capillary
- the anodic stabilizer (IAA) migrates toward the anodic side of the capillary.
- a pH gradient is set up in the capillary and the proteins align according to their pi.
- a large amount of cathodic stabilizer in cIEF is used to fill the outlet side of the capillary. This forces the ampholytes and protein sam-ple to focus before the detection window and ensures their detection during mobilization.
- the catholyte is replaced against a vial with chemical mobilizer (HAc) and the actual voltage is increased from 25 kV to 30 kV.
- HAc chemical mobilizer
- hydronium ions (H+) are introduced from the anolyte into the capillary, while acetate ions are introduced at the cathodic side.
- the pH gradient is titrated from basic to acidic and the protein sample bands are detected at 280 nm as they obtain a positive charge and migrate toward the cathode.
- Anodic Stabilizer 200 mM Iminodiacetic Acid in DDI water
- FTIR Fourier transform infrared
- SDS-cGE electrophoresis
- LMW low molecular weight compounds
- Capillary electrophoresis is an analytical technique that separates ions based on their electrophoretic mobility with the use of an applied voltage.
- the electrophoretic mobility is dependent upon the charge of the molecule, the viscosity, and the atom's radius.
- the rate at which the particle moves is directly proportional to the applied electric field. Neutral species are not affected, only ions move with the electric field.
- Capillary gel electrophoresis uses separation based on the difference in solute size as the particles migrate through the gel. Gels are useful because they minimize solute diffusion that causes zone broadening, prevent the capillary walls from absorbing the solute, and limit the heat transfer by slowing down the molecules.
- the strongly anionic detergent sodium dodecyl sulphate (SDS) is used in combination with heat to denature the proteins before they are loaded onto the gel. The denatured proteins are enclosed by SDS, become negatively charged, and exhibit a consistent charge-to-mass ratio regard-less of protein type.
- Corrected area (capillary length to detector x area) / retention time
- LMWS low molecular weight species
- Size exclusion chromatography uses porous particles to separate molecules of different sizes. It is generally used to separate biological molecules according to their molecular weight and shape in dilute solution.
- the stationary phase consists of spherical porous particles of carefully controlled pore size through which biomolecules diffuse to different extents based on differences in their molecular sizes. Small molecules diffuse freely into the pores and their movement through the column is retarded, whereas large molecules are unable to enter the pores and are therefore eluted earlier. Hence, molecules are separated in order of decreasing molecular mass, with the largest molecules eluting from the column first.
- the protein samples of the stability study were loaded onto a TSKgel G3000SWXL, (Tosoh, 300 x 7.8 mm, 5 pm) column to detect high molecular weight- and low molecular weight species of ustekinumab.
- the protein was eluted by isocratic elution using 0.1 M sodium phosphate (pH 6.0) with 0.2 M sodium chloride at a flow rate of 0.5 mL/min at 25°C. Eluted species were detected at a wavelength of 214 nm and displayed on a graph showing the concentration of the eluted species vs. time.
- the elution profile showed a main peak with the non-aggregated protein, peaks of the protein representing higher molecular weight forms of the protein and peaks showing lower molecular weight species (fragments). The area of all peaks was determined.
- Table 7 shows the percentage of peak area for high molecular weight species (HMWS) and the low molecular weight species (LMWS) in relation to the total peak area of the eluted species for the samples of Table 1.
- Two dilutions of a sample aliquot of each formulation were prepared by diluting the sample with mobile phase to a concentration of 1.0 mg/mL. Each dilution was examined as a single measurement. For this purpose 20 pL equal to 20 pg were applied on the column.
- Table 7 Overview of High molecular weight- and Low molecular weight species species determined by SEC for all formulations at time point 1 month and tO at the incubation temperatures 5°C, 25 °C/ 60 % r.H. and 40 °C/ 75 % r.H.
- the sample was diluted with 50 mM Tris(hydroxymethyl)-aminomethane to a final ustekinumab concentration of 0.125 mg/mL for analysis of the peptides.
- 11 oxidated aminoacids in ustekinumab could be identified after 1 month storage at 40 °C/ 75 % r.H.
- the iLiteTM IL-23 cells is a stably transfected DT40 cell line expressing the IL-23 receptor and IL-12 receptor beta chains together with a STAT5 expression vector, a STAT5 responsive Firefly luciferase (FL) reporter, and Renilla luciferase (RL) under a constitutive promoter for normalization.
- IL-23 treatment of the IL-23 responsive iLITETM cells induces expression of FL in a dose-dependent manner.
- Sequential measurement of the second reporter gene construct RL in the same well using the Dual GloTM system from Promega allows normalization of cell number and serum matrix effects. Ustekinumab neutralizes the p40 subunit IL-23.
- the relative biological activity of ustekinumab in all formulations was measured by its inhibition of IL-23 induced Firefly Luciferase activation in the iLITE IL-23 cells.
- a dilution series of every sample starting from dilution 0 (375 pg/mL) down to 0.96 ng/pL (dilution 8) in eight steps was performed. Two independent dilutions were used for each formulation in this assay.
- the Firefly luciferase- and Renilla luciferase activity was determined using a Promega GloMax Multi luminometer. The calculation of the relative potency of the samples was done with the PLA 2.1 software from Stegmann Systems. The normalized“fold induction of FL activity” values were used for the potency calculation.
- Assay medium RPMI 1640 (61870-010, Life Technologies) + 10 % FBS (10500-064, Life Technologies)
- the potency of ustekinumab at tO is comparable for all formulations.
- the potency decreases after one month storage at 40°C/ 75 % relative humidity in all formulations at comparable rates.
- the viscosity is determined in the undiluted ustekinumab formulations using a Kinexus ultra plus cone-plate rheometer from Malvern.
- Ustekinumab at a target concentration of 90 mg/ml is formulated in the following solutions:
- the different ustekinumab formulations are stored at 5 °C for up to 12 months, at 25°C / 60% relative humidity for 6 months and at 40 °C / 75% relative humidity for up to 3 months. Additionally, ustekinumab in the different formulations is stressed by five freeze/ thaw cycles or by shaking the samples at 600 rpm, 25 °C / 60% relative humidity for three months.
- Ustekinumab in the Stelara ® formulation buffer system is included in the stability program as control sample (h) according to tablel3. Table 13: Detailed information of all formulations used in this study.
- the samples according to table 13 is analyzed by visual inspection for changes in appearance and color, by capillary isoelectric focusing (cIEF) for detection of modifications leading to charge heterogeneities like deamination or N-terminal pyroglutamic acid formation, by Fourier transform infrared (FTIR) spectroscopy for secondary structure, by sodium dodecyl sulfate capillary gel electrophoresis (SDS-cGE) under non-reducing conditions for the presence of fragments and sample purity, by size exclusion
- cIEF capillary isoelectric focusing
- FTIR Fourier transform infrared
- SDS-cGE sodium dodecyl sulfate capillary gel electrophoresis
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Microbiology (AREA)
- Molecular Biology (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Mycology (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Preparation (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
The present invention relates to stable, low viscosity, high concentration liquid pharmaceutical compositions of an anti-IL-12/23p40 antibody comprising at least 80 mg/ml of the anti-IL-12/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, and either i) 50-70 mM of a histidine buffer and no inorganic salt, or ii) 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt, or iii) 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
Description
Stable, low viscosity, high concentration liquid formulations of an anti-IL-l2/23p40 antibody
FIELD OF THE INVENTION
The present invention relates to stable, low viscosity, high concentration liquid
pharmaceutical compositions of an anti-IL-l2/23p40 antibody comprising at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, and either i) 50-70 mM of a histidine buffer and no inorganic salt, or ii) 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt, or iii) 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. BACKGROUND OF THE INVENTION
The heterodimeric cytokine interleukin- 12 (IL-12) consists of disulfide bonded, glycosylated polypeptide chains with a weight of 35 and 40 kD. IL-12 is synthesized and secreted by antigen presenting cells, including dendritic cells, monocytes, macrophages, B cells, Langerhans cells and keratinocytes as well as natural killer (NK) cells, and is involved in a variety of biological processes. It is also referred to as NK cell stimulatory factor (NKSF), T- cell stimulating factor, cytotoxic T-lymphocyte maturation factor, and EBV-transformed B- cell line factor (Curfs et al., 1997). IL-12 can bind to IL-12 receptor, which is expressed on the plasma membrane of cells (e.g.,
T cells, NK cell), and thereby alter, e.g. initiate or prevent, these cells’ biological processes. IL-12 binding to IL-12 receptor can stimulate the proliferation of pre-activated T cells and NK cells, enhance the cytolytic activity of cytotoxic T cells (CTL), NK cells and lymphokine activated killer (LAK) cells, induce production of gamma interferon (IFN-g) by T cells and
NK cells, and induce differentiation of naive ThO cells into Thl cells that produce IFN-g and IL-2 (Trinchieri, 1995). An especially key role of IL-12 is in the generation of cytolytic cells, such as NKs and CTLs, and in mounting a cellular immune response, such as a Thl cell mediated immune response, making IL-12 vital in the generation and regulation of both protective immunity, for example eradication of infections, and pathological immune responses, such as autoimmunity (Hendrzak and Brunda, 1995).
The p40 protein subunit of IL-12 can form another cytokine, interleukin 23 (IL-23) by associating with another protein subunit, pl9 (Oppman et 1., 2000). IL-23, similarly to IL-12, can bind to a two-chain receptor complex, which mediates an intracellular signal. Despite a shared protein subunit between IL-23 and IL-12, as well as a corresponding shared receptor subunit between IL-23 receptor and IL-12 receptor, these two cytokines have distinct biological functions (Langrish wet al., 2005). IL-23 mediates IL-17 production by T-cells (Parham et al., 2002; Aggarwal et al., 2003).
As a result, manipulating the activity of IL-12 and IL-23 in vivo can modulate immune responses to be enhanced, suppressed or prevented. Indeed, abnormal IL-12 and IL-23 regulation is an important factor in many immune-mediated diseases, making IL-12 and IL- 23 promising targets in treatment.
An anti-IL-l2/IL-23p40 antibody, ustekinumab, also known as CNTO 1275 or by its trade name Stelara®, is approved in Europe and the United States for the treatment of moderate to severe plaque psoriasis, psoriatic arthritis, and Crohn’s disease. Ustekinumab as Stelara® is available as a solution for injection (45 and 90 mg) in vials or prefilled syringes and as a concentrate (130 mg) to make a solution for infusion (drip) into a vein. As Stelara®, ustekinumab is formulated at 90 mg/ml with 0.48 mg/ml L-histidine, 0.74 mg/ml L-histidine hydrochloride monohydrate, 0.04 mg/ml polysorbate 80, and 76 mg/ml sucrose.
In plaque psoriasis and psoriatic arthritis, ustekinumab is given by subcutaneous injection. For adults, the usual dose is 45 mg, whereas the dose in children depends on bodyweight.
The first injection is followed by a further injection four weeks later, and then an injection every 12 weeks. Patients weighing over 100 kg should be given ustekinumab in 90-mg doses for psoriasis, and this should also be considered for psoriatic arthritis. In Crohn’s disease, treatment is started with ustekinumab concentrate. The infusion lasts at least one hour and
the dose depends on the patient’s bodyweight. Eight weeks after the first infusion patients should receive 90 mg ustekinumab by subcutaneous injection. Patients then continue with ustekinumab given subcutaneously every 8 or 12 weeks depending on response to treatment.
Subcutaneous administration of concentrated antibody solutions is preferable for patients with chronic diseases such as the ones that ustekinumab is approved for, because frequent dosing may be needed over their lifetimes. Subcutaneous injections can be self-administered by the patients using ready to use prefilled syringes or pens /auto-injectors and delivery devices. Such devices are advantageous for better patient comfort. Moreover, the concentrated antibody drug solutions contained in these devices may provide the additional benefit of longer intervals between injections (and more effective pharmacokinetic profile for the antibody). Typically, not more than 1 mL of drug product solution can be administered as a single bolus dose via a prefilled syringe due to volume restrictions for dose administration in the subcutaneous space. However, the total volume and duration of administration is dictated by the concentration of the monoclonal antibody in the dosing solution. In order to achieve higher dose administration in smaller volumes, either for infusion or bolus administration, high concentrations of monoclonal antibodies in solution are required, as is the case with ustekinumab at 90 mg/ml. However, such highly concentrated antibody solutions can demonstrate high viscosities, which is problematic because high viscosity may increase the injection time and pain at the site of injection, adversely affecting patient compliance. Similarly, high viscosity requires the use of thicker needles, which makes the injection more painful, whereas low viscosity formulations can be injected using, e.g., 29G, 30G, 31G, or 34G needles or even thinner needles due to better syringeability,. High viscosity of a concentrated antibody solution also poses difficulties during bioprocessing of the drug substance. During downstream ultrafiltration and diafiltration steps, high viscosity of the antibody solution may increase the back pressure of the pump and decrease the transmembrane flux, which may increase the processing time, destabilize the drug substance and increase manufacturing costs (Tomar et al., 2016).
Similarly, filling a viscous formulation into ready-to-use syringes may be problematic.
Various strategies to lower the viscosity of high concentration antibody solutions have been reported, recent strategies including the addition of caffeine (WO 2015/196187) or n-acetyl arginine, n-acetyl lysine, n-acetyl histidine, n-acetyl proline, and mixtures thereof (WO 2016/065181). However, due to the small number of high concentration antibody formulations on the market, information on how to achieve a stable, low viscosity, high
concentration solution for any given antibody of choice is limited, and determination thereof remains not trivial.
Accordingly, there is a need for alternative formulations of anti-IL-l2/23p40 antibodies that are stable, low viscosity, high concentration liquid pharmaceutical compositions of an anti- IL-l2/23p40 antibody suitable for intravenous or subcutaneous administration in the treatment of immune-mediated diseases such as e.g. plaque psoriasis, psoriatic arthritis, and Crohn’s disease.
SUMMARY OF THE INVENTION
The inventors found that a stable, high concentration liquid pharmaceutical composition of an anti-IL-l2/23p40 antibody comprising at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, and either i) 50-70 mM of histidine and no inorganic salt, or ii) 25-35 mM of histidine and 10-15 mM of an inorganic salt, or iii) 5-10 mM of histidine and 20-40 mM, preferably 25-30 mM, of an inorganic salt has a surprisingly low level of viscosity and is therefore particularly suitable for subcutaneous administration and for use in treating immune-mediated diseases in which IL12 and/or IL-23 play a role.
A further advantage of the stable, high concentration liquid pharmaceutical composition according to the present invention is that bioprocessing of the formulations is made more convenient. Downstream ultrafiltration and diafiltration steps are made easier by decreased back pressure of the pump and increased transmembrane flux, which reduces the processing time, results in a more stable antibody solution, and decreases manufacturing costs.
The object of the present invention is solved by the subject matter of the independent claims. Preferred embodiments are apparent from the dependent claims.
Accordingly, in one embodiment the present invention provides a stable, low viscosity, liquid pharmaceutical composition comprising
a) at least 80 mg/ml of an anti-IL-l2/23p40 antibody
b) between 65 to 70 mg/ml sugar
c) between 0.02-0.06 mg/ml of a non-ionic surfactant; and
i) 50-70 mM of histidine and no inorganic salt, or
ii) 25-35 mM of histidine buffer and 10-15 mM of an inorganic salt, or
iii) 5-10 mM of histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
The anti-IL-l2/23p40 antibody may be present in a concentration of from 80 mg/ml to 100 mg/ml, preferably in a concentration of 90 mg/ml.
In one embodiment, the sugar comprised in the composition is sucrose.
Preferably, the non-ionic surfactant is present in a concentration of 0.04 mg/1.
The non-ionic surfactant can be polysorbate 20 or 80.
The inorganic salt may be sodium chloride.
In one embodiment, the composition has a pH of 5.8-6.2.
In a preferred embodiment, the pharmaceutical composition comprises
a) 90 mg/ml of the anti-IL-l2/23p40 antibody
b) 65 mg/ml sucrose
c) 0.04 mg/ml polysorbate 80; and
d) 60 mM histidine buffer and no inorganic salt,
and has a pH of 6.0.
In another preferred embodiment, the pharmaceutical composition comprises
a) 90 mg/ml of the anti-IL-l2/23p40 antibody
b) 70 mg/ml sucrose
c) 0.04 mg/ml polysorbate 80; and
d) 31 mM histidine buffer and 12.5 mM sodium chloride,
and has a pH of 6.0.
In a third preferred embodiment, the pharmaceutical composition comprises
a) 90 mg/ml of the anti-IL-l2/23p40 antibody
b) 70 mg/ml sucrose
c) 0.04 mg/ml polysorbate 80; and
d) 6.6 mM histidine buffer and 25 mM sodium chloride,
and has a pH of 6.0.
The antibody may be one that substantially neutralizes at least one biological activity of at least one IL-12 and/or IL-23 protein, and preferably is ustekinumab.
In an embodiment, the pharmaceutical composition is suitable for administration by at least one mode selected from the group consisting of parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic, intrapericardiac, intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and transdermal. In a preferred embodiment, the mode of administration the pharmaceutical composition is suitable for is subcutaneous administration.
In a preferred embodiment, the mode of administration the pharmaceutical composition is suitable for is intravenous administration.
The pharmaceutical composition may be used in treating an immune-mediated disease in which IL-12 and/or IL-23 play a role.
Preferably, the immune-mediated disease is selected from the group of plaque psoriasis, psoriatic arthritis, Crohn’s disease, ankylosing spondylitis, spondyloarthritis, and ulcerative colitis.
The present invention also relates to a prefilled syringe containing the pharmaceutical composition as defined herein.
In an embodiment, the syringe is suitable for containing or administering, as defined herein, the composition as defined herein.
DETAILED DESCRIPTION OF THE INVENTION
The present invention, as illustratively described in the following, may suitably be practiced in the absence of any element or elements, limitation or limitations, not specifically disclosed herein.
The present invention will be described with respect to particular embodiments, but the invention is not limited thereto, but only by the claims. Where the term“comprising” is used in the present description and claims, it does not exclude other elements. For the purposes of the present invention, the term“consisting of’ is considered to be a preferred embodiment of the term“comprising". If hereinafter a group is defined to comprise at least a certain number of embodiments, this is also to be understood to disclose a group which preferably consists only of these embodiments.
Where an indefinite or definite article is used when referring to a singular noun, e.g.“a”,
“an” or“the”, this includes a plural of that noun unless something else is specifically stated.
The term“pharmaceutical composition” as used herein refers to any composition comprising a chemical substance or active ingredient which composition is intended for use in the medical cure, treatment, or prevention of disease and which is in such a form as to permit the active ingredient to be effective. In particular, a pharmaceutical composition does not contain excipients which are unacceptably toxic to a subject to which the composition is to be administered. The pharmaceutical compositions are sterile, i.e. aseptic and free from all living microorganisms and their spores. The pharmaceutical composition used in the present invention is liquid and stable.
In a“liquid composition”, the pharmaceutically active agent, e.g. the anti-IL-l2/23p40 antibody, can be combined with a variety of excipients to ensure a stable active medication following storage. The liquid pharmaceutical composition used in the invention is at no point lyophilised, i.e. the production method does not contain a lyophilisation step and the composition is not lyophilised for storage. Liquid compositions can be stored in vials, IV bags, ampoules, cartridges, and prefilled or ready-to-use syringes.
Preferably, the liquid composition is an aqueous liquid composition. An aqueous liquid composition comprises water as solvent.
A "stable" liquid composition is one in which the anti-IL-l2/23p40 antibody contained therein essentially retains its physical stability and/or chemical stability and/or biological activity upon storage for a certain period. Preferably, the composition essentially retains upon storage its physical and chemical stability, as well as its biological activity. Various analytical techniques for measuring protein stability are available in the art and are reviewed, for example, in Peptide and Protein Drug Delivery, 247-301, Vincent Lee Ed, Marcel Dekker, Inc, New York, New York, Pubs (1991) and Jones, Adv Drug Delivery Rev, 1993, 10:29-90. For example, stability can be measured at a selected temperature for a selected time period. Stability can be evaluated qualitatively and/or quantitatively in a variety of different ways, including evaluation of aggregate formation (for example using size exclusion chromatography, by measuring turbidity, and/or by visual inspection), by assessing charge heterogeneity using cation exchange chromatography or capillary zone
electrophoresis, amino-terminal or carboxy-terminal sequence analysis, mass spectrometric analysis, SDS-PAGE analysis to detect aggregated or fragmented molecules, peptide map (for example tryptic or LYS-C) analysis, evaluating biological activity or binding of the antagonist, etc.
Preferably, the pharmaceutical composition is stable at a temperature of about 40°C for at least 1 to 2 weeks, and/or is stable at a temperature of about 5°C for at least 9 months, and/or is stable at a temperature of about 25°C for at least two weeks to one month. Furthermore, the formulation is preferably stable following freezing (to, e.g., -20°C) and thawing of the formulation at 25°C as described in the examples herein, for example following 1, 2, 3, 4 or 5 cycles of freezing and thawing.
For example, in the pharmaceutical composition used in the present invention, the percentage of high molecular weight species relative to the total amount of the anti-lL- 12/23r40 antibody as measured by size exclusion chromatography is not more than 5%, preferably not more than 3% or 2%, more preferably not more than 1% and most preferably not more than 0.8% or 0.2% after storage of the composition at 5°C for 1 month.
Further, in the pharmaceutical composition used in the present invention, the percentage of fragments relative to the total amount of the anti-lL-l2/23p40 antibody as measured by size
exclusion chromatography is not more than 5%, preferably not more than 3% or 2%, more preferably not more than 1% or 0.5% and most preferably not more than 0.4% or 0.2% after storage of the composition at 5°C for 1 month.
The term“low- viscosity” is used herein to indicate that the viscosity of the pharmaceutical composition is less than about 20 cP, preferably below 15 cP, and most preferably below 10 cP at about 25° C. In some embodiments, the viscosity is less than about 10 cP, 9cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C. For liquid, high concentration antibody containing pharmaceutical compositions, measurements of viscosity typically are performed at shear rates from about 0.6 s' 1 to about 450 s"l, and preferably from about 2 s"l to about 400 s"l, when measured using a cone and plate viscometer. Alternatively, viscosity can be measured at shear rates from about 3 s"l to about 55,000 s"l, and preferably from about 20 s"l to about 2,000 s"l when measured using a micro fluidic viscometer.
The compositions of the invention have an osmolality of 250-350 mOsm/kg, preferably of 280-320 mOsm/kg.
The term„antibody” or“immunoglobulin” is used herein in the broadest sense and includes full length antibodies, genetically engineered antibodies, recombinant antibodies, multivalent antibodies, monoclonal antibodies, polyclonal antibodies, bispecific antibodies, multispecific antibodies, chimeric antibodies, humanized antibodies, fully human antibodies, as well as fragments of such antibodies as long as they remain functional and exhibit the desired biological activity. The“biological activity” of an antibody refers to the ability of the antibody to bind to antigen and result in a biological response which can be measured in vitro or in vivo.
A full length antibody comprises an antigen-binding variable region of the light (VL) and heavy chain (VH), a light chain constant region (CL) and heavy chain constant domains Cnl , CH2 and CH3.
The term“antibody fragment” or“antigen-binding fragment” is used herein in the broadest sense and comprises a portion of a full length antibody, preferably comprising the antigen binding or variable region thereof. An antibody fragment retains the original specificity of the parent immunoglobulin. Examples of antibody fragments include, e.g., Fab, Fab', F(ab')2, and Fv fragments, diabodies, linear antibodies, single-chain antibody molecules, and
multispecific antibodies formed from antibody fragment(s). Preferably, the antibody fragment is a Fab fragment.
A“monoclonal antibody” is an antibody that is specific for a single epitope of an antigen, i.e. directed against a single determinant on an antigen. Methods for producing monoclonal antibodies are known to the person skilled in the art.
The term“recombinant antibody” refers to all antibodies prepared, expressed, created or isolated by recombinant means, such as antibodies isolated from a transgenic host cell, such as e.g. a NS0 or CHO cell, or from an animal transgenic for immunoglobulin genes, or antibodies expressed using recombinant expression vectors transfected into a host cell, such as e.g. SP 2/0 mouse myeloma cells.
A“humanised antibody” is a human antibody wherein the antigen binding portion (CDR) is derived from non-human species, such as a mouse, and thus has a different specificity compared to the parent immunoglobulin. The CDR protein sequences can be modified to increase their similarities to antibody variants produced naturally in humans.
The term“anti-IL-l2/23p40 antibody” refers to an antibody or antibody fragment, such as a Fab or a scFV fragment, which specifically interacts with the p40 protein subunit of IL-12 and/or IL-23 VEGF and neutralizes one or more of the biological activities of these cytokines, e.g. their activity in proliferation of pre-activated T cells and NK cells, in enhancing the cytolytic activity of cytotoxic T cells (CTL), NK cells and lymphokine activated killer (LAK) cells, in inducing production of gamma interferon (IFN-g) by T cells and NK cells, induce differentiation of naive ThO cells into Thl cells that produce IFN-g and IL-2, in mounting Thl cell mediated immune responses, or in mediating IL-17 production in T cells. It is intended to include both anti-IL-l2/23p40 antibodies and antigen-binding fragments thereof.
Anti-IL-l2/23p40 antibodies act, e.g., by interfering with the binding of IL-12 and/or IL-23 to their cellular receptors, and by interfering with the biological functions of IL-12 and/or IL- 23 after binding to their cellular receptor. Anti-ILl2/23p40 antibodies include, e.g., ustekinumab as described in, for example, WO 02/12500. Preferably, the anti-ILl2/23p40
antibody or antigen-binding fragment thereof present in the pharmaceutical composition used in the present invention is ustekinumab or an antigen-binding fragment thereof.
“Ustekinumab” is a fully human IgGlK monoclonal antibody directed against the IL- 12/23r40 subunit having the heavy and light chain variable domain sequences as described in SEQ ID Nos. 7 and 8 of WO 02/12500 and Benson et al. (2011) MAbs 3(6): 535-545. The CAS number of ustekinumab is 815610-63-0. Ustekinumab binds with high specificity to the p40 protein subunit of interleukin (IL)-l2 and IL-23, thereby suppressing IL-12- and IL-23- mediated immune responses and inflammation It has been approved in Europe, the United States of America, and Canada for the treatment of moderate to severe plaque psoriasis, active psoriatic arthritis, and moderately to severely active Crohn’s disease. It is produced in a murine myeloma cell line using recombinant DNA technology as described in WO 02/12500 and Benson et al. (2011) MAbs 3(6): 535-545. The present commercial ustekinumab formulation contains L-histidine/histidine hydrochloride, sucrose, polysorbate 80, and water for injection and is supplied in a concentration of 90 mg/ml.
The concentration of the anti-ILl2/23p40 antibody in the pharmaceutical compositions is at least 80 mg/ml, typically 80-100 mg/ml, preferably 85-95 mg/ml, most preferably 90 mg/ml.
The pharmaceutical compositions of the invention permit the anti-ILl2/23p40 antibody comprised therein to exhibit its activity of neutralizing at least one of the biological functions of IL-12 and/or IL-23.
The term“sugar” refers to an organic compound comprising only carbon, hydrogen, and oxygen, usually with a hydrogemoxygen atom ratio of 2: 1 and the empirical formula Cm(H20)n. The term“sugar” includes mono-, di-, oligo- and polysaccharides. Examples of carbohydrates include glucose, fructose, galactose, xylose, ribose, sucrose, mannose, lactose, maltose, trehalose, starch, and glycogen. Various other forms of sugars are known, e.g., sugar alcohols such as glycerol, mannitol, sorbitol, and xylitol; sugar acids, e.g. aldonic acids such as ascorbic acid, aldaric acids such as tartaric acid; reducing sugars, e.g. glucose, glyceraldehydes, galactose, lactose, and maltose; amino sugars, e.g. N-acetylglucosamine, galactosamine, glucosamine, and sialic acid; or sulfoquinovose, a sulphonic acid derivative of glucose. Most preferably, the sugar of the invention is sucrose.
A“surfactant” as used herein refers to an amphiphilic compound, i.e. a compound containing both hydrophobic groups and hydrophilic groups that lowers the surface tension (or interfacial tension) between two liquids or between a liquid and a solid. A“non-ionic surfactant” has no charged groups in its head. The formation of insoluble particles during freeze/thaw cycles of antibody-containing compositions can be remarkably inhibited by addition of surfactants. Examples of“non-ionic surfactants” include e.g. polyoxyethylene glycol alkyl ethers, such as octaethylene glycol monododecyl ether, pentaethylene glycol monododecyl ether; polyoxypropylene glycol alkyl ethers; glucoside alkyl ethers, such as decyl glucoside, lauryl glucoside, octyl glucoside; polyoxyethylene glycol octylphenol ethers, such as triton X-100; polyoxyethylene glycol alkylphenol ethers, such as nonoxynol- 9; glycerol alkyl esters, such as glyceryl laurate; polyoxyethylene glycol sorbitan alkyl esters, such as polysorbate; sorbitan alkyl esters, such as spans; cocamide MEA, cocamide DEA, dodecyldimethylamine oxide; block copolymers of polyethylene glycol and polypropylene glycol, such as poloxamers; and polyethoxylated tallow amine (POEA). The pharmaceutical compositions of the present invention can contain one or more of these surfactants in combination.
Preferred non-ionic surfactants for use in the pharmaceutical compositions of the present invention are polysorbates such as polysorbate 20, 40, 60 or 80 or poloxamer 188, and especially polysorbate 20 (i.e. Tween 20) or polysorbate 80. Most preferred is polysorbate 80.
The concentration of the non-ionic surfactant is in the range of 0.002 to 0.006% (w/v), preferably in the range of 0.0025 to 0.0055% (w/v), more preferably in the range of 0.003 to 0.005% (w/v) and most preferably it is 0.004% (w/v) relative to the total volume of the composition.
In a preferred embodiment, the non-ionic surfactant is polysorbate 20 with a concentration in the range of 0.002 to 0.006% (w/v), more preferably in the range of 0.003 to 0.005% (w/v) and most preferably it is 0.004% (w/v), relative to the total volume of the composition.
In another preferred embodiment, the non-ionic surfactant is polysorbate 80 with a concentration in the range of 0.002 to 0.006% (w/v), more preferably in the range of 0.003 to 0.005% (w/v) and most preferably it is 0.004% (w/v), relative to the total volume of the composition.
In a particularly preferred embodiment the non-ionic surfactant is polysorbate 20 with a concentration of 0.004% (w/v), relative to the total volume of the composition. In another particularly preferred embodiment the non-ionic surfactant is polysorbate 80 with a concentration of 0.004% (w/v), relative to the total volume of the composition.
A“buffer” is an aqueous solution consisting of a mixture of a weak acid and its conjugate base or vice versa which resists changes in its pH and therefore keeps the pH at a nearly constant value. The buffer of the present invention preferably has a pH in the range from about 5.5 to about 7.0, preferably from about 5.7 to about 6.8, more preferably from about 5.8 to 6.5 and most preferably has a pH of about 5.8 to 6.2, such as, most preferably, a pH of 6.0. The buffer used in the present invention is a "histidine-containing buffer" or“histidine buffer”, which terms are used interchangeably herein and refer to a buffer comprising histidine. Examples of histidine buffers include histidine chloride, histidine hydrochloride, histidine acetate, histidine phosphate, and histidine sulphate. The preferred histidine buffer of the invention further comprises L-histidine. Even more preferably, the histidine buffer of the invention comprises histidine hydrochloride, most preferably it comprises histidine hydrochloride and L-histidine. Preferably, the histidine buffer or histidine hydrochloride buffer or histidine hydrochloride/L-histidine buffer has a pH in the range from about 5.5 to about 7.0, preferably from about 5.7 to about 6.8, more preferably from about 5.8 to 6.5 and most preferably has a pH of about 5.8 to 6.2, such as, most preferably, a pH of 6.0. ln a particularly preferred embodiment, the histidine buffer has a concentration in the range of 50 mM to 70 mM, preferably of 55 mM to 65 mM, and most preferably of 60 mM. Most preferably the histidine buffer of this embodiment is histidine hydrochloride/L-histidine. ln another particularly preferred embodiment, the histidine buffer has a concentration in the range of 25 mM to 35 mM, preferably of 28 mM to 32 mM, and most preferably of 31 mM. Most preferably the histidine buffer of this embodiment is histidine hydrochloride/L- histidine.
In a third particularly preferred embodiment, the histidine buffer has a concentration in the range of 5 mM to 10 mM, preferably of 6 mM to 8 mM, and most preferably of 6.6 mM. Most preferably the histidine buffer of this embodiment is histidine hydrochloride/L- histidine.
In another particular preferred embodiment the buffer is histidine hydrochloride/L-histidine with the concentrations specified above and with a pH of 6.0.
The pharmaceutical compositions of the present invention may be prepared by dissolving L- histidine/histidine hydrochloride, the sugar, preferably sucrose, and, if present, the inorganic salt, preferably sodium chloride, in water before adding the non-ionic surfactant, preferably polysorbate 20 or polysorbate 80 and then adding the anti-IL-l2/23p40 antibody.
Herein, an“inorganic salt” refers to a ionic compound which has osmoregulatory properties. An inorganic salt such as sodium chloride (NaCl) can dissociate in solution into its constituent ions, i.e. NaCl dissociates into Na+ and Cl ions, which both affect the osmotic pressure, i.e. the osmolality, of the solution. Preferred inorganic salts for use in the pharmaceutical formulation of the present invention are potassium chloride, calcium chloride, sodium chloride, sodium phosphate, potassium phosphate and sodium bicarbonate. Preferably the inorganic salt is a sodium salt, more preferably it is sodium chloride (NaCl).
In an embodiment, the inorganic salt has a concentration in the range of 10 mM to 15 mM, preferably of 12 mM to 13 mM, and most preferably of 12.5 mM. In this embodiment, the histidine buffer has a concentration of 25 to 35 mM. Most preferably, the inorganic salt of this embodiment is NaCl.
In another embodiment, the inorganic salt has a concentration in the range of 20 mM to 30 mM, preferably of 22.5 mM to 27.5 mM, and most preferably of 25 mM. In this embodiment, the histidine buffer has a concentration of 5 to 10 mM. Most preferably, the inorganic salt of this embodiment is NaCl .
The compositions of the invention preferably have a pH in the range from about 5.5 to about 7.0, preferably from about 5.7 to about 6.8, more preferably from about 5.8 to 6.5 and most preferably has a pH of about 5.8 to 6.2, such as, most preferably, a pH of 6.0.
In an embodiment the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2.
In an embodiment the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2.
Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical
composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25- 30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the anti- IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04
mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer,
water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no iurther excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2.
Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
Unless otherwise specified, the pharmaceutical composition used in the present invention may further contain diluents, solubilising agents, isotonising agents, excipients, pH- modifiers, soothing agents, buffers, sulphur-containing reducing agents, antioxidants or the like.
In an embodiment, the pharmaceutical composition is suitable for parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic,
intrapericardiac, intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and/or transdermal administration. Preferably, the pharmaceutical administration is suitable for intravenous and/or subcutaneous administration.
In an embodiment the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL- 12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-
12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti- IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP,
and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti- IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80- 100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0.
Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less
than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e.
the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a
pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition suitable for one of the above specified administrations comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
An "immune-mediated disease in which IL-12 and/or IL-23 play a role" is a disease characterized by inappropriate or elevated immune functions mediated by IL-12 and/or IL- 23, e.g. via their activity in proliferation of pre-activated T cells and NK cells, in enhancing the cytolytic activity of cytotoxic T cells (CTL), NK cells and lymphokine activated killer (LAK) cells, in inducing production of gamma interferon (IFN-g) by T cells and NK cells, induce differentiation of naive ThO cells into Thl cells that produce IFN-g and IL-2, in mounting Thl cell mediated immune responses, or in mediating IL-17 production in T cells. Examples of immune-mediated disease in which IL-12 and/or IL-23 play a role are, for example, rheumatoid arthritis, juvenile rheumatoid arthritis, systemic onset juvenile rheumatoid arthritis, psoriatic arthritis, ankylosing spondilitis, gastric ulcer, seronegative arthropathies, osteoarthritis, osteolysis, aseptic loosening of orthopedic implants, inflammatory bowel disease, ulcerative colitis, systemic lupus erythematosus,
antiphospholipid syndrome, iridocyclitis/uveitis/optic neuritis, idiopathic pulmonary fibrosis, systemic vasculitis/wegener's granulomatosis, sarcoidosis, orchitis/vasectomy reversal procedures, allergic/atopic diseases, asthma, allergic rhinitis, eczema, allergic contact dermatitis, allergic conjunctivitis, hypersensitivity pneumonitis, transplants, organ transplant rejection, graft- versus-host disease, systemic inflammatory response syndrome, sepsis syndrome, gram positive sepsis, gram negative sepsis, culture negative sepsis, fungal sepsis, neutropenic fever, urosepsis, meningococcemia, trauma/hemorrhage, bums, ionizing radiation exposure, acute pancreatitis, adult respiratory distress syndrome, rheumatoid
arthritis, alcohol-induced hepatitis, chronic inflammatory pathologies, sarcoidosis, Crohn's pathology, sickle cell anemia, diabetes, nephrosis, atopic diseases, hypersensitity reactions, allergic rhinitis, hay fever, perennial rhinitis, conjunctivitis, endometriosis, asthma, urticaria, systemic anaphalaxis, dermatitis, pernicious anemia, hemolytic disesease, thrombocytopenia, graft rejection of any organ or tissue, kidney translplant rejection, heart transplant rejection, liver transplant rejection, pancreas transplant rejection, lung transplant rejection, bone marrow transplant (BMT) rejection, skin allograft rejection, cartilage transplant rejection, bone graft rejection, small bowel transplant rejection, fetal thymus implant rejection, parathyroid transplant rejection, xenograft rejection of any organ or tissue, allograft rejection, anti-receptor hypersensitivity reactions, Graves disease, Raynaud's disease, type B insulin-resistant diabetes, asthma, myasthenia gravis, antibody-meditated cytotoxicity, type III hypersensitivity reactions, POEMS syndrome (polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, and skin changes syndrome), polyneuropathy, organomegaly, endocrinopathy, monoclonal gammopathy, skin changes syndrome, antiphospholipid syndrome, pemphigus, scleroderma, mixed connective tissue disease, idiopathic Addison's disease, diabetes mellitus, chronic active hepatitis, primary billiary cirrhosis, vitiligo, vasculitis, post-MI cardiotomy syndrome, type IV hypersensitivity, contact dermatitis, hypersensitivity pneumonitis, allograft rejection, granulomas due to intracellular organisms, drug sensitivity, metabolic/idiopathic, Wilson's disease,
hemachromatosis, alpha- 1 -antitrypsin deficiency, diabetic retinopathy, hashimoto's thyroiditis, osteoporosis, hypothalamic-pituitary-adrenal axis evaluation, primary biliary cirrhosis, thyroiditis, encephalomyelitis, cachexia, cystic fibrosis, neonatal chronic lung disease, chronic obstructive pulmonary disease (COPD), familial hematophagocytic lymphohistiocytosis, dermatologic conditions, psoriasis, alopecia, nephrotic syndrome, nephritis, glomerular nephritis, acute renal failure, hemodialysis, uremia, toxicity, preeclampsia, okt3 therapy, anti-cd3 therapy, cytokine therapy, chemotherapy, radiation therapy (e.g., including but not limited to, asthenia, anemia, cachexia, and the like), chronic salicylate intoxication, and the like. See, e.g., the Merck Manual, l2th- l7th Editions, Merck & Company, Rahway, NJ (1972, 1977, 1982, 1987, 1992, 1999), Pharmacotherapy
Handbook, Wells et al, eds., Second Edition, Appleton and Lange, Stamford, Conn. (1998, 2000). The pharmaceutical composition may be used in the treatment of any such immune- mediated disease in which IL-12 and/or IL-23 play a role.
The pharmaceutical composition is preferably for use in the treatment of plaque psoriasis, psoriatic arthritis, Crohn’s disease, ankylosing spondylitis, spondyloarthritis, and ulcerative
colitis. The plaque psoriasis is preferably moderate to severe plaque psoriasis. The psoriatic arthritis is preferably active psoriatic arthritis. The Crohn’s disease is preferably moderately to severely active Crohn’s disease.
In plaque psoriasis and psoriatic arthritis treatment, the pharmaceutical composition is preferably given by subcutaneous injection. For adults, the preferred dose is 45 mg of the anti-IL-l2/23p40 antibody, whereas the preferred dose in children depends on bodyweight. Preferably, the first injection is followed by a further injection four weeks later, and then an injection every 12 weeks. For patients weighing over 100 kg, the pharmaceutical composition is preferably given in 90 mg doses for psoriasis, and this could also be done for patients weighing over lOOkg having psoriatic arthritis. ln Crohn’s disease treatment, treatment is preferably started with an intravenous infusion of an anti-lLl2/23p40 antibody prepared from concentrate that lasts at least one hour, wherein the administered dose preferably depends on the patient’s bodyweight. Preferably, eight weeks after the first infusion, patients then receive the pharmaceutical compositions at a dose of 90 mg anti-lLl2/23p40 antibody by subcutaneous injection. Preferably, patients then continue receiving subcutaneously administered pharmaceutical composition every 8 or 12 weeks depending on response to treatment. ln an embodiment the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-lL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C. ln an embodiment the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-lL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-lL-l2/23p40 antibody,
between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL- 12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-
12/23r40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02- 0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80- 100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15
cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic
surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition for use in one of the above-specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water.
Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0.
Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition for use in one of the above-specified treatments comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no iurther excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium
chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition for use in one of the above- specified treatments comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml
polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
Pharmaceutical compositions of the present invention can be supplied in sealed and sterilized plastic, glass, or other suitable containers having a defined volume such as vials, ampoules or syringes or a large volume such as bottles.
It is preferred that the liquid pharmaceutical composition is supplied in a prefilled syringe. A “ready-to-use syringe” or“prefilled syringe” is a syringe which is supplied in a filled state, i.e. the pharmaceutical composition to be administered is already present in the syringe and ready for administration. Prefilled syringes have many benefits compared to separately provided syringe and vial, such as improved convenience, affordability, accuracy, sterility, and safety. The use of prefilled syringes results in greater dose precision, in a reduction of the potential for needle sticks injuries that can occur while drawing medication from vials, in pre-measured dosage reducing dosing errors due to the need to reconstituting and/or drawing medication into a syringe, and in less overfilling of the syringe helping to reduce costs by minimising drug waste.
Preferably, the prefilled syringe containing the pharmaceutical composition is suitable for parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic, intrapericardiac, intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and/or transdermal administration. Most preferably, the prefilled syringe containing the
pharmaceutical administration is suitable for intravenous and/or subcutaneous
administration.
Preferably, the prefilled syringe containing the pharmaceutical composition suitable for one of the above-specified administrations is suitable for use in the above-specified treatment of an immune-mediated disease in which IL-12 and/or IL-23 play a role.
In an embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2.
Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP,
6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP,
7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04
mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL-l2/23p40 antibody, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water.
Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20
cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL- 12/23r40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml of the anti-IL-l2/23p40 antibody, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the anti-IL-l2/23p40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of the anti-IL- 12/23r40 antibody, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer and no inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In an embodiment the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of the ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 50-70 mM of a histidine buffer, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the
pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer and no inorganic salt, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, water and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 60 mM of a histidine buffer, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer and 10- 15 mM of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab, between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non-ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, water, and no further excipients, i.e. the pharmaceutical composition consists of at least 80 mg/ml, preferably 80-100 mg/ml, of ustekinumab,
between 65 to 70 mg/ml of a sugar, preferably sucrose, between 0.02-0.06 mg/ml of a non ionic surfactant, preferably polysorbate 20 or polysorbate 80, between 25-35 mM of a histidine buffer, 10-15 mM of an inorganic salt, and water. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of the ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer and 12.5 mM sodium chloride, and has a pH of 6.0.
Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, water, and no further excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 31 mM of a histidine buffer, 12.5 mM sodium chloride, and water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt. Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In another embodiment, the pharmaceutical composition contained in the prefilled syringe comprises at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, water, and no other excipients, i.e. the
pharmaceutical composition consists of at least 80 mg/ml of ustekinumab, between 65 to 70 mg/ml of a sugar, between 0.02-0.06 mg/ml of a non-ionic surfactant, between 5-10 mM of a histidine buffer, 20-40 mM, preferably 25-30 mM, of an inorganic salt, and water.
Preferably, the pharmaceutical composition has a pH of 5.8-6.2. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer and 25 mM sodium chloride, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
In a preferred embodiment, the pharmaceutical composition contained in the prefilled syringe comprises 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and no other excipients, i.e. the pharmaceutical composition consists of 90 mg/ml, of ustekinumab, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80, 6.6 mM of a histidine buffer, 25 mM sodium chloride, water, and has a pH of 6.0. Preferably, the viscosity of the pharmaceutical composition is less than about 20 cP, more preferably less than about 15 cP, and most preferably less than about 10 cP, 9 cP, 8 cP, 7 cP, 6 cP, 5 cP, 4 cP, 3 cP, 2 cP, or about 1 cP at about 25° C.
While the invention has been illustrated and described in detail in the drawings and foregoing description, such illustration and description are to be considered illustrative or exemplary and not restrictive. The invention is not limited to the disclosed embodiments. Other variations to the disclosed embodiments can be understood and effected by those skilled in the art in practicing a claimed invention, from a study of the drawings, the disclosure, and the dependent claims.
The detailed description is merely exemplary in nature and is not intended to limit application and uses. The following examples further illustrate the present invention without, however, limiting the scope of the invention thereto. Various changes and modifications can
be made by those skilled in the art on the basis of the description of the invention, and such changes and modifications are also included in the present invention.
EXAMPLES
1. Sample preparation
Ustekinumab from the EU marketed Stelara® (90 mg/ml) was transferred into 4 different formulations containing:
(a) 6.6 mM histidine/ histidine chloride, 76 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(b) 25 mM NaCl, 6.6 mM histidine/ histidine chloride, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(c) 12.5 mM NaCl, 31 mM histidine/ histidine chloride, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(d) 60 mM histidine/ histidine chloride, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80.
The final ustekinumab concentration of all samples was 10 mg/mL. The different ustekinumab formulations were stored at 5 °C for up to 6 months, or at 25°C / 60% relative humidity or 40°C / 75% relative humidity for up to 3 months. Additionally, ustekinumab in the different formulations was stressed by five freeze/ thaw cycles or by shaking samples at 600rpm, 25°C / 60% relative humidity. Ustekinumab in the Stelara® formulation buffer system was included in the stability program as control sample (a). Table 1 : Detailed information of all formulations used in this study.
Afterwards the samples according to table 1 were analyzed by visual inspection for changes in appearance and color, by capillary isoelectric focusing (cIEF) for detection of modifications leading to charge heterogeneities like deamination or N-terminal pyroglutamic acid formation, by Fourier transform infrared (FTIR) spectroscopy for secondary structure, by sodium dodecyl sulfate capillary gel electrophoresis (SDS-cGE) under non-reducing conditions for the presence of fragments and sample purity, by size exclusion
chromatography (SEC) for the presence of high molecular weight species (HMWS), by liquid chromatography coupled to mass spectrometry (LC-MS) peptide mapping after trypsin digestion for chemical modification, by iLITE™ IL-23 assay for potency, and by UV-Vis spectroscopy at 280 nm and 330 nm for protein concentration,
2. Analysis of appearance /color by visual inspection Each sample vial was visually checked for appearance and color, recording any changes such as e.g. color changes or visible particle formation.
In all formulations (a - d) no visible changes in color could be detected after 3 months storage at all temperatures (5 °C, 25 °C/ 60 % relative humidity, 40 °C/ 75 % relative humidity). The solutions were always identified as clear, no visible particles could be identified. This was also demonstrated for all formulations after freeze/ thaw and shake stress.
3. Analysis of modifications by cIEF
Capillary isoelectric focusing (cIEF) is a method used for the determination of the identity and purity of Ustekinumab. cIEF is a quantitative high-resolution analytical technique that allows the separation of proteins, protein glycoforms and other protein isoforms, differing in their net-charge based on their isoelectric point (pi). Modifications like deamidation or N- terminal pyroglutamic acid formation, leading to a change of net charge can be detected.
The cIEF separation consists of two steps, focusing and mobilization. At the start of the analysis the capillary is filled with the sample. The cIEF sample is a mixture of ampholytes, stabilizers, pi markers and the protein sample. Focusing is performed by submerging one capillary end in anolyte (H+) and one capillary end in catholyte (OFF) and applying voltage. During focusing, the cathodic stabilizer (Arg) migrates toward the cathodic side of the capillary, and the anodic stabilizer (IAA) migrates toward the anodic side of the capillary. A pH gradient is set up in the capillary and the proteins align according to their pi. A large amount of cathodic stabilizer in cIEF is used to fill the outlet side of the capillary. This forces the ampholytes and protein sam-ple to focus before the detection window and ensures their detection during mobilization. For mobilization the catholyte is replaced against a vial with chemical mobilizer (HAc) and the actual voltage is increased from 25 kV to 30 kV. During mobilization, hydronium ions (H+) are introduced from the anolyte into the capillary, while acetate ions are introduced at the cathodic side. As a result, the pH gradient is titrated from basic to acidic and the protein sample bands are detected at 280 nm as they obtain a positive charge and migrate toward the cathode.
Equipment:
PA800 plus Pharmaceutical Analysis System, Beckman Coulter
Neutral Capillary, 50 pm i.d., PN 477441, Beckman Coulter, cutted to 30.2 cm
Anolyte: 200 mM Phosphoric Acid in DDI water
Catholyte: 300 mM Sodium Hydroxide in DDI water
Chemical Mobilizer: 350 mM Acetic Acid in DDI water
Cathodic Stabilizer: 350 mM L-arginine in DDI water
Anodic Stabilizer: 200 mM Iminodiacetic Acid in DDI water
Urea Solution: 4.3 M Urea in DDI water
3M Urea - cIEF Gel: 1.8 g urea, solved and filled to 10 mL with cIEF Gel (477497, Beckman Coulter)
Table 2: Separation method
15 pL of each sample were diluted from 10.0 mg/mL with ultrapure water to a concentration of 7.5 mg/mL. The samples were analyzed as single measurement. The relative areas of 6 peaks detectable in all formulations were analyzed.
All results were comparable for all tested formulations (see table 3) during one month incubation at 40 °C/ 75 % relative humidity. In all formulations there was a clear trend to a decrease of the main peak (peak 3) with increasing peaks 4 - 6 which is based on the generation of acidic species of ustekinumab like deamidations. Basic species decreased at 40°C/ 75 r.H after one month in all formulations in a comparable way due to ustekinumab fragmentation and a shift to acidic pi e.g. due to deamidations. 1 month storage at 5 °C and 25 °C showed no significant alteration in the peak profile in all formulations (a - d).
Table 3: Peaks
4. Analysis of secondary structure by FTIR
Fourier transform infrared (FTIR) spectroscopy provides information about the secondary structure content of proteins and works by excitation of a sample with infrared radiation and detection which wavelengths of the spectrum are absorbed by the protein. Regions of Amide I and Amide II in ustekinumab were analyzed with regard to changes in the secondary structure components alpha-helixes and beta-sheet formation. Beta-sheet- and alpha-helix- contents were calculated by using the second derivative spectra correlated to known structures of proteins.
20 pL of each of the samples containing a concentration of 10.0 mg/mL were injected into a Tensor 27 (Broker Optics, Ettlingen, Germany) and analyzed for a single measurement.
Also the corresponding formulation buffers (without Ustekinumab) were analyzed because of their absorption in the wavelength range 2000 - 1000 cm 1 which interferes with the analyzed Amide I absorption range (1700 - 1600 cm 1). A buffer subtraction was performed for every formulation (a - d), the second derivatives were calculated and the average spectra
were generated. The secondary structures were calculated by OPUS 6.5 software (Broker Optics, Ettlingen, Germany)
Table 4: Overview of the alpha-helix- and beta-sheet content
In all formulations (a - d) no clear trends in FTIR could be detected after 2 months storage at all temperatures (5 °C, 25 °C/ 60 % relative humidity, 40 °C/ 75 % relative humidity). The beta-sheet content was unchanged to tO in all formulations. Alpha-helix content was identified at low level in all samples during 2 months stability.
5. Analysis of sample purity by non-reduced SDS-cGE
This test method describes the analysis of ustekinumab samples by capillary gel
electrophoresis (SDS-cGE) under non reducing conditions with respect to protein purity, especially low molecular weight compounds (LMW).
Capillary electrophoresis is an analytical technique that separates ions based on their electrophoretic mobility with the use of an applied voltage. The electrophoretic mobility is dependent upon the charge of the molecule, the viscosity, and the atom's radius. The rate at which the particle moves is directly proportional to the applied electric field. Neutral species are not affected, only ions move with the electric field.
Capillary gel electrophoresis uses separation based on the difference in solute size as the particles migrate through the gel. Gels are useful because they minimize solute diffusion that causes zone broadening, prevent the capillary walls from absorbing the solute, and limit the
heat transfer by slowing down the molecules. The strongly anionic detergent sodium dodecyl sulphate (SDS) is used in combination with heat to denature the proteins before they are loaded onto the gel. The denatured proteins are enclosed by SDS, become negatively charged, and exhibit a consistent charge-to-mass ratio regard-less of protein type.
4.5 mΐ of each sample was diluted with 19.5 mΐ 50 mM acetate buffer pH 4.0 and 6 mΐ 5 % SDS stock solution and further incubated 10 minutes at 65 °C. The vials were shortly centrifuged for 20 seconds to collect all liquid from the inner surfaces of the reaction tube. 50 mM Acetate buffer pH 4 0:
286 mΐ 100 % acetic acid were add to 80 mL ultrapure water. pH 4.0 was adjusted using 1 N NaOH. The solution was further filled with ultrapure water to a final volume of 100 mL.
Electrophoretic conditions:
System: CE PA800plus (Beckmann Coulter)
Software 32 Karat V 10.1 for data aquistion (Beckman Coulter)
Software Chromeleon 7.2 for data evaluation and data processing (Thermo Scientific) Cartridge Assembly Kit (144738, Beckman Coulter)
PA 800plus SDS-MW Analysis Kit (390953, Beckman Coulter)
Separation polarity: Reverse (negative at the inlet)
lnjection: Hydrodynamic (pressure)
Capillary temperature: 20 °C
Detection wavelength: 214 nm Table 5: Method
All samples were analyzed as a single measurement. The relative areas of peaks for the monomeric form and low molecular weight (LMW) species of ustekinumab were analyzed. The peak areas were corrected by following formula:
Corrected area = (capillary length to detector x area) / retention time
Table 6: Overview of low molecular weight species determined by non-reduced SDS-cGE
The generation of low molecular weight species (LMWS) of ustekinumab determined by non-reduced SDS-cGE was comparable for the incubation parameters 5 °C, 25 °C/ 60 % r.H., 40 °C/ 75 % r.H. and freeze/ thaw stress within the different formulations (a-d).
With increasing temperature the amounts of LMWS increased over time. There was no clear trend within 2 months incubation comparing the different formulations. Also freeze/ thaw processes led to a comparable amount of LMWS for the different formulations, unchanged compared to the starting material (tO).
6. Analysis of HMWS by SEC
Size exclusion chromatography (SEC) uses porous particles to separate molecules of different sizes. It is generally used to separate biological molecules according to their
molecular weight and shape in dilute solution. The stationary phase consists of spherical porous particles of carefully controlled pore size through which biomolecules diffuse to different extents based on differences in their molecular sizes. Small molecules diffuse freely into the pores and their movement through the column is retarded, whereas large molecules are unable to enter the pores and are therefore eluted earlier. Hence, molecules are separated in order of decreasing molecular mass, with the largest molecules eluting from the column first.
The protein samples of the stability study were loaded onto a TSKgel G3000SWXL, (Tosoh, 300 x 7.8 mm, 5 pm) column to detect high molecular weight- and low molecular weight species of ustekinumab.
The protein was eluted by isocratic elution using 0.1 M sodium phosphate (pH 6.0) with 0.2 M sodium chloride at a flow rate of 0.5 mL/min at 25°C. Eluted species were detected at a wavelength of 214 nm and displayed on a graph showing the concentration of the eluted species vs. time. The elution profile showed a main peak with the non-aggregated protein, peaks of the protein representing higher molecular weight forms of the protein and peaks showing lower molecular weight species (fragments). The area of all peaks was determined. Table 7 shows the percentage of peak area for high molecular weight species (HMWS) and the low molecular weight species (LMWS) in relation to the total peak area of the eluted species for the samples of Table 1.
Two dilutions of a sample aliquot of each formulation were prepared by diluting the sample with mobile phase to a concentration of 1.0 mg/mL. Each dilution was examined as a single measurement. For this purpose 20 pL equal to 20 pg were applied on the column.
Table 7: Overview of High molecular weight- and Low molecular weight species species determined by SEC for all formulations at time point 1 month and tO at the incubation temperatures 5°C, 25 °C/ 60 % r.H. and 40 °C/ 75 % r.H.
After one month incubation at all temperatures the results were comparable for all four formulations. Whereas the generation of high molecular weight species was still comparable to the starting material in the formulations after one month at all tested temperatures the generation of low molecular weight species was identified as temperature dependent. With increase of the temperature the amount of fragments increased in all formulations in a comparable way. After one month incubation at 40 °C/ 75 % relative humidity formulation (b) showed the lowest generation of LMWS compared to the other tested formulations.
7. Analysis of chemical modifications by reduced LC-MS peptide mapping By reduced peptide mapping the purity of ustekinumab with regard to asparagine-/ glutamine
deamidation and methionine-/ tryptophane oxidation was analyzed after digestion with trypsin and liquid chromatography coupled to mass spectrometry (LC-MS). After reduction the protein was submitted to enzymatic cleavage with trypsin. The resulting peptides were analyzed by RP-UPLC-MS. During chromatography the peptides were eluted by changing the mobile phase from highly polar (formic acid in water) to less polar (formic acid in acetonitrile) and analyzed by mass spectrometry (VION IMS QTOF). The peptide data were processed and compared with the theoretical protein sequence coverage information and detect modifications.
Samples shown in Table 1 were analyzed as single measurement after 1 month incubation at 40 °C/ 75 % relative humidity and compared to the starting material tO.
Samples were diluted with denaturation buffer (50 mM Tris(hydroxymethyl)aminomethane in water, pH adjusted with HC1 to pH 7.2) to a ustekinumab concentration of 1.5 mg/mL.
66.5 mΐ of the diluted samples were mixed with 13.5 mΐ 100 mM L-methionine (149 mg L- methionine solved in 10 mL denaturation buffer) and 10 mΐ of 0.5 % RapiGest (from Waters, solved in 50 mM Tris(hydroxymethyl)aminomethane) and incubated for 5 minutes at 95 °C.
4.5 mΐ of 0.02 M DTT (dissolved in 50 mM Tris(hydroxymethyl)-aminomethane) were added for reduction and the samples were incubated for 30 minutes at 37°C. The final DTT concentration is 1 mM. For ustekinumab digestion 5 mΐ of a 1 mg/mL Trypsin solution (solved in 50 mM acetic acid) were added and the solution was incubated for further 3 hours at 37°C. The reaction was stopped with 20 mΐ of 5 % (v/v) formic acid and an incubation for 30 minutes at 37°C. 24 mΐ of 20 % (v/v) acetonitrile/ 33.4 mM L-methionine/ 1 % (w/v) formic acid were added to the pellet (after centrifugation for 5 minutes at 13.400 rpm). The solution was now incubated for further 15 minutes at 37 °C, centrifuged for 5 minutes at 13.400 rpm and the supernatant was combined with the supernatant of the centrifugation step before.
The sample was diluted with 50 mM Tris(hydroxymethyl)-aminomethane to a final ustekinumab concentration of 0.125 mg/mL for analysis of the peptides.
UPLC Parameters:
The digested protein samples from the syringes were loaded onto an ACQU1TY UPLC-CSH C-18 column from Waters, 100 mm x 2.1 mm, 1.7 pm. 0.25 pg of the digested samples were eluted at 65 °C with a gradient of eluent A (water), eluent B (acetonitrile), eluent C (1 % (w/v) formic acid) according to the following Table 8:
Table 8: Overview of eluent gradient
Method parameters for mass spectrometry:
Ionisation type: ESI
Polarity: Positive
Analyser mode: Sensitivity
Experiment type: MS
Start Mass: 50 m/z
End Mass: 2000 m/z
Cone Gas Flow: 30 L/h
Desolvation Gas Flow: 1000 L/h
Source Temperature: 120 °C
Scan Time: 0.5 s
Desolvation Temperature: 450 °C
Capillary Voltage: 3.0 kV
Cone Voltage: 40 V
Low CE: 6.0 eV
High CE Ramp Start: 25 eV
High CE Ramp End: 45 eV
Acquisition Start Time: 0.00 min
Acquisition End Time: 44.00 min
LockSpray Profile
Reference Compound: Leucine Enkephalin
MS Lock mass: 556.2766 m/z
Scan Time: 0.5 s
Interval: 30 s
Come voltage: 30 V
Capillary voltage: 3.0 kV
Attenuation: 80 %
Analyzer Mode: Sensitivity
Flow rate: 5.0 mΐ/min Table 9: Instrument Parameter:
Quadrupole RF profile: Manual
Source offset: 80 V
Step Wave 1 offset: 20 V
Step wave 2 offset: 30 V
11 oxidated aminoacids in ustekinumab could be identified after 1 month storage at 40 °C/ 75 % r.H. (light chain: methionine (1 :Tl) AA 4, tryptophane (1 :T3) AA 32; heavy chain: methionine (2:T20) AA 254, tryptophane (2:T22) AA 279, tryptophane (2:T25) AA 315, methionine (2:T5) AA 40; methionine (2:T6) AA 51, tryptophane (2:T6) AA 47, methionine (2:T7) AA 70, tryptophane (2:T8) AA 82, methionine (2:T9) AA 93).
All these oxidation sites were summed up for total oxidation of ustekinumab.
10 deamidation sites were detected during incubation at 40 °C / 75 % r.H. up to 1 month (light chain: glutamine (1 :T5) AA 55, glutamine (1 :T9) AA 124; heavy chain: glutamine (2:Tl) AA 3+6, asparagine (2:T22) AA 278+288, asparagine (2:T25) AA 317, asparagine (2:T35) AA 363, asparagine (2:T36) AA 386 +391, glutamine (2:T5) AA 39, glutamine (2:T7) AA 65+67, asparagine (2:T8) AA 83).
All these deamidation sites were summed up for total deamidation of ustekinumab.
Table 10: Overview of oxidated species and deamidation determined via reduced peptide mapping for all formulations at time point tO and one month at the incubation temperature 40°C/ 75 % r.H.
Incubation of the formulations a - d at 40 °C/ 75 % relative humidity for one month led to an increase of both oxidation and deamidation compared to the starting material. Although all formulations were evaluated with an acceptable range especially formulation (d) seems to be superior compared to the others with regard to these protein degradation. 8. Analysis of potency by iLITE™ IL-23 assay
The iLite™ IL-23 cells is a stably transfected DT40 cell line expressing the IL-23 receptor and IL-12 receptor beta chains together with a STAT5 expression vector, a STAT5 responsive Firefly luciferase (FL) reporter, and Renilla luciferase (RL) under a constitutive promoter for normalization. IL-23 treatment of the IL-23 responsive iLITETM cells induces expression of FL in a dose-dependent manner. Sequential measurement of the second reporter gene construct RL in the same well using the Dual Glo™ system from Promega allows normalization of cell number and serum matrix effects. Ustekinumab neutralizes the p40 subunit IL-23.
The relative biological activity of ustekinumab in all formulations was measured by its inhibition of IL-23 induced Firefly Luciferase activation in the iLITE IL-23 cells. For assay performance, a dilution series of every sample starting from dilution 0 (375 pg/mL) down to 0.96 ng/pL (dilution 8) in eight steps was performed. Two independent dilutions were used
for each formulation in this assay.
The Firefly luciferase- and Renilla luciferase activity was determined using a Promega GloMax Multi luminometer. The calculation of the relative potency of the samples was done with the PLA 2.1 software from Stegmann Systems. The normalized“fold induction of FL activity” values were used for the potency calculation.
Further Reagents & Solutions:
Assay medium: RPMI 1640 (61870-010, Life Technologies) + 10 % FBS (10500-064, Life Technologies)
Dual-Glo Luciferase Assay System (E2940, Promega)
Cells: iLite IL-23 Assay ready cells (BM4023, Biomonitor)
Recombinant human IL-23 (1290-IL/CF, R&D Systems) Table 11 : Potency of all formulations after one month incubation compared to the starting material determined via iLite™ IL-23 assay
The potency of ustekinumab at tO is comparable for all formulations. The potency decreases after one month storage at 40°C/ 75 % relative humidity in all formulations at comparable rates.
9. Analysis of protein content by UV-Vis
The concentration of ustekinumab in the samples was measured spectrophotometrically (at 280 nm and 330 nm). Potential light scattering (at 330 nm) was subtracted from the absorbance reading at 280 nm prior to total protein concentration calculation. The experimentally determined specific extinction coefficient was 1.595 Lg-lcm-l.
Table 12: Protein concentrations of all formulations determined by UV280
There was no significant change in protein concentration (spectrophotometric quantification at 280nm; n =3; corrected for light scattering at 330nm) detectable in all samples during the complete stability program including thermal stability, freeze/ thaw processes and shaking stress, respectively.
10. Determination of viscosity
Ustekinumab from the EU marketed Stelara® (90 mg/ml) was reformulated into 3 different formulations containing each 90 mg/mL ± 4 mg/mL ustekinumab:
(e) 6.6 mM histidine/ histidine chloride, 76 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(f) 25.0 mM NaCl, 6.6 mM histidine/ histidine chloride, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(g) 60 mM histidine/ histidine chloride, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80
The viscosity is determined in the undiluted ustekinumab formulations using a Kinexus ultra plus cone-plate rheometer from Malvern.
Upper geometry: Cone Plate CP1/20 = 1° angle, 20 mm diameter
Lower geometry: Plate PU61 = 61 mm diameter
11. Stability study of ustekinumab at 90 mg/mL in different formulations
Ustekinumab at a target concentration of 90 mg/ml is formulated in the following solutions:
(h) 6.6 mM histidine/ histidine chloride, 76 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(i) 25.0 mM NaCl, 6.6 mM histidine/ histidine chloride, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(j) 12.5 mM NaCl, 31 mM histidine/ histidine chloride, 70 mg/ml sucrose, 0.04 mg/ml polysorbate 80
(k) 60 mM histidine/ histidine chloride, 65 mg/ml sucrose, 0.04 mg/ml polysorbate 80.
The different ustekinumab formulations are stored at 5 °C for up to 12 months, at 25°C / 60% relative humidity for 6 months and at 40 °C / 75% relative humidity for up to 3 months. Additionally, ustekinumab in the different formulations is stressed by five freeze/ thaw cycles or by shaking the samples at 600 rpm, 25 °C / 60% relative humidity for three months. Ustekinumab in the Stelara® formulation buffer system is included in the stability program as control sample (h) according to tablel3. Table 13: Detailed information of all formulations used in this study.
Afterwards the samples according to table 13 is analyzed by visual inspection for changes in appearance and color, by capillary isoelectric focusing (cIEF) for detection of modifications leading to charge heterogeneities like deamination or N-terminal pyroglutamic acid formation, by Fourier transform infrared (FTIR) spectroscopy for secondary structure, by sodium dodecyl sulfate capillary gel electrophoresis (SDS-cGE) under non-reducing conditions for the presence of fragments and sample purity, by size exclusion
chromatography (SEC) for the presence of high molecular weight species (HMWS) and low molecular weight species, by liquid chromatography coupled to mass spectrometry (LC-MS) peptide mapping after trypsin digestion for chemical modification, by iLITE™ IL-23 assay for potency, and by UV-Vis spectroscopy at 280 nm for protein concentration.
Claims
1. A stable, low viscosity, liquid pharmaceutical composition comprising
a) at least 80 mg/ml of an anti-IL-l2/23p40 antibody
b) between 65 to 70 mg/ml sugar
c) between 0.02-0.06 mg/ml of a non-ionic surfactant; and
i) 50-70 mM of a histidine buffer and no inorganic salt, or
ii) 25-35 mM of a histidine buffer and 10-15 mM of an inorganic salt, or
iii) 5-10 mM of a histidine buffer and 20-40 mM, preferably 25-30 mM, of an inorganic salt.
2. The pharmaceutical composition according to claim 1, wherein the anti-IL-l2/23p40 antibody is present in a concentration of from 80 mg/ml to 100 mg/ml.
3. The pharmaceutical composition according to claim 2, wherein the anti-IL-l2/23p40 antibody is present in a concentration of 90 mg/ml.
4. The pharmaceutical composition according to any one of the preceding claims, wherein the sugar is sucrose.
5. The pharmaceutical composition according any one of the preceding claims, wherein the non-ionic surfactant is present in a concentration of 0.04 mg/1.
6. The pharmaceutical composition according to any one of the preceding claims, wherein the non-ionic surfactant is polysorbate 20 or 80.
7. The pharmaceutical composition according to any one the preceding claims, wherein the inorganic salt is sodium chloride. 8. The pharmaceutical composition according to anyone of the preceding claims, wherein the composition has a pH of 5.
8-6.2.
9. The pharmaceutical composition according to any one of the preceding claims, comprising
a) 90 mg/ml of the anti-IL-l2/23p40 antibody
b) 65 mg/ml sucrose
c) 0.04 mg/ml polysorbate 80; and
d) 60 mM histidine buffer and no inorganic salt,
and wherein the composition has a pH of 6.0.
10. The pharmaceutical composition according to any one of claims 1-8, comprising a) 90 mg/ml of the anti-IL-l2/23p40 antibody
b) 70 mg/ml sucrose
c) 0.04 mg/ml polysorbate 80; and
d) 31 mM histidine buffer and 12.5 mM sodium chloride,
and wherein the composition has a pH of 6.0.
11. The pharmaceutical composition according to any one of claims 1-8, comprising a) 90 mg/ml of the anti-IL-l2/23p40 antibody
b) 70 mg/ml sucrose
c) 0.04 mg/ml polysorbate 80; and
d) 6.6 mM histidine buffer and 25 mM sodium chloride,
and wherein the composition has a pH of 6.0.
12. The pharmaceutical composition according to any one of the preceding claims, wherein the antibody substantially neutralizes at least one biological activity of at least one IL-12 and/or IL-23 protein.
13. The pharmaceutical composition according to any one of the preceding claims, wherein the antibody is ustekinumab.
14. The pharmaceutical composition according to any one of the preceding claims, wherein the composition is suitable for administration by at least one mode selected from the group consisting of parenteral, subcutaneous, intramuscular, intravenous, intraarticular, intrabronchial, intraabdominal, intracapsular, intracartileginous, intracavitary, intracelial, intracerebellar, intracerebroventicular, intracolic, intracervical, intragastric, intrahepatic, intramyocardial, intraosteal, intrapelvic, intrapericardiac, intraperitoneal, intrapleural, intraprostatic, intrapulmonary, intrarectal, intrarenal, intraretinal, intraspinal, intrasynovial, intrathorachic, intraunterine, intravesical, bolud, vaginal, rectal, buccal, sublingual, intranasal, and transdermal.
15. The pharmaceutical composition according to claim 14, wherein the composition is suitable for subcutaneous administration.
16. The pharmaceutical composition according to claim 14, wherein the composition is suitable for intravenous administration.
17. The pharmaceutical composition of any one of the preceding claims, for use in treating an immune-mediated disease in which IL-12 and/or IL-23 play a role.
18. The pharmaceutical composition for use according to claim 16, wherein the immune- mediated disease is selected from the group consisting of plaque psoriasis, psoriatic arthritis, Crohn’s disease, ankylosing spondylitis, spondyloarthritis, and ulcerative colitis.
19. A prefilled syringe containing the pharmaceutical composition as defined in any one of the preceding claims.
20. The prefilled syringe according to claim 18, wherein the syringe is suitable for containing or administering according to any one of claims 14-16 the composition according to any of claims 1-18.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17205000.7 | 2017-12-01 | ||
| EP17205000 | 2017-12-01 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2019106206A1 true WO2019106206A1 (en) | 2019-06-06 |
Family
ID=60629441
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2018/083353 Ceased WO2019106206A1 (en) | 2017-12-01 | 2018-12-03 | Stable, low viscosity, high concentration liquid formulations of an anti-il-12/23p40 antibody |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2019106206A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3714902A1 (en) * | 2020-03-13 | 2020-09-30 | LEK Pharmaceuticals d.d. | Stabilization of pharmaceutical compositions comprising polysorbate |
| EP4045085A4 (en) * | 2019-10-18 | 2023-11-15 | Janssen Biotech, Inc. | Safe and effective method of treating ulcerative colitis with anti-il12/il23 antibody |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002012500A2 (en) | 2000-08-07 | 2002-02-14 | Centocor, Inc. | Anti-il-12 antibodies, compositions, methods and uses |
| WO2007076062A2 (en) * | 2005-12-21 | 2007-07-05 | Wyeth | Protein formulations with reduced viscosity and uses thereof |
| WO2009114040A2 (en) * | 2007-09-28 | 2009-09-17 | Centocor Ortho Biotech Inc. | Anti-il-12/23p40 antibodies, epitopes, formulations, compositions, methods and uses |
| WO2015196187A1 (en) | 2014-06-20 | 2015-12-23 | Reform Biologics, Llc | Viscosity-reducing excipient compounds for protein formulations |
| WO2016065181A1 (en) | 2014-10-23 | 2016-04-28 | Amgen Inc. | Reducing viscosity of pharmaceutical formulations |
-
2018
- 2018-12-03 WO PCT/EP2018/083353 patent/WO2019106206A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002012500A2 (en) | 2000-08-07 | 2002-02-14 | Centocor, Inc. | Anti-il-12 antibodies, compositions, methods and uses |
| WO2007076062A2 (en) * | 2005-12-21 | 2007-07-05 | Wyeth | Protein formulations with reduced viscosity and uses thereof |
| WO2009114040A2 (en) * | 2007-09-28 | 2009-09-17 | Centocor Ortho Biotech Inc. | Anti-il-12/23p40 antibodies, epitopes, formulations, compositions, methods and uses |
| WO2015196187A1 (en) | 2014-06-20 | 2015-12-23 | Reform Biologics, Llc | Viscosity-reducing excipient compounds for protein formulations |
| WO2016065181A1 (en) | 2014-10-23 | 2016-04-28 | Amgen Inc. | Reducing viscosity of pharmaceutical formulations |
Non-Patent Citations (11)
| Title |
|---|
| "Merck Manual", 1972, MERCK & COMPANY |
| "Peptide and Protein Drug Delivery", 1991, MARCEL DEKKER, INC, pages: 247 - 301 |
| "Pharmacotherapy Handbook", 1998, APPLETON AND LANGE |
| BENSON ET AL., MABS, vol. 3, no. 6, 2011, pages 535 - 545 |
| CHEN BEI ET AL: "Influence of histidine on the stability and physical properties of a fully human antibody in aqueous and solid forms", PHARMACEUTICAL RESEARCH, SPRINGER NEW YORK LLC, US, vol. 20, no. 12, 1 December 2003 (2003-12-01), pages 1952 - 1960, XP002386671, ISSN: 0724-8741, DOI: 10.1023/B:PHAM.0000008042.15988.C0 * |
| DING C ET AL: "ABT-874, a fully human monoclonal anti-IL-12/IL-23 antibody for the potential treatment of autoimmune diseases", CURRENT OPINION IN INVESTIGATIONAL DR, CURRENT DRUGS, LONDON, GB, vol. 9, no. 5, 1 January 2008 (2008-01-01), pages 515 - 522, XP008134761, ISSN: 0967-8298 * |
| JACQUELINE M. BENSON ET AL: "Discovery and mechanism of ustekinumab: A human monoclonal antibody targeting interleukin-12 and interleukin-23 for treatment of immune-mediated disorders", MABS, vol. 3, no. 6, 1 November 2011 (2011-11-01), pages 535 - 545, XP055133436, ISSN: 1942-0862, DOI: 10.4161/mabs.3.6.17815 * |
| JANSSEN: "STELARA 45 mg Injektionslösung in einer Fertigspritze", 1 July 2011 (2011-07-01), pages 1 - 5, XP055467870, Retrieved from the Internet <URL:https://www.praxis-kiedrowski.de/files/fachinformation_stelara.pdf> [retrieved on 20180417] * |
| JONES, ADV DRUG DELIVERY REV, vol. 10, 1993, pages 29 - 90 |
| LIMA XINAIDA TALIGARE ET AL: "Briakinumab", EXPERT OPINION ON BIOLOGICAL THE, INFORMA HEALTHCARE, UK, vol. 9, no. 8, 1 August 2009 (2009-08-01), pages 1107 - 1113, XP009176839, ISSN: 1744-7682, DOI: 10.1517/14712590903092188 * |
| WANG WEI ED - BLANCO-PRIETO MARIA J ET AL: "Instability, stabilization, and formulation of liquid protein pharmaceuticals", INTERNATIONAL JOURNAL OF PHARMACEUTICS, ELSEVIER, NL, vol. 185, no. 2, 20 August 1999 (1999-08-20), pages 129 - 188, XP002323952, ISSN: 0378-5173, DOI: 10.1016/S0378-5173(99)00152-0 * |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP4045085A4 (en) * | 2019-10-18 | 2023-11-15 | Janssen Biotech, Inc. | Safe and effective method of treating ulcerative colitis with anti-il12/il23 antibody |
| EP4424381A3 (en) * | 2019-10-18 | 2024-11-13 | Janssen Biotech, Inc. | Safe and effective method of treating ulcerative colitis with anti-il12/il23 antibody |
| EP3714902A1 (en) * | 2020-03-13 | 2020-09-30 | LEK Pharmaceuticals d.d. | Stabilization of pharmaceutical compositions comprising polysorbate |
| WO2021180981A1 (en) * | 2020-03-13 | 2021-09-16 | Lek Pharmaceuticals D.D. | Stabilization of pharmaceutical compositions comprising polysorbate |
| US20230137216A1 (en) * | 2020-03-13 | 2023-05-04 | Lek Pharmaceuticals D.D. | Stabilization of Pharmaceutical Compositions Comprising Polysorbate |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109078182B (en) | Antibody preparation | |
| RU2664736C2 (en) | Pharmaceutical composition containing adalimumab | |
| ES2732861T3 (en) | Solution formulations of engine-manipulated anti-IL-23p19 antibodies | |
| JP2020500195A (en) | Aflibercept preparation and its use | |
| US20180008707A1 (en) | Stable liquid formulation for monoclonal antibodies | |
| TWI682788B (en) | Liquid formulation comprising gm-csf neutralizing compound | |
| CN110167531B (en) | Stable liquid formulations | |
| US20230173069A1 (en) | Formulation comprising anti-il-23p19 antibody, method for preparing same and use thereof | |
| KR102106914B1 (en) | Liquid formulation comprising GM-CSF neutralizing compound | |
| JP2017105754A (en) | Low viscosity antibody composition | |
| CN111356471A (en) | Abutip formulation comprising lysine salt as tonicity modifier and use thereof | |
| KR20240109250A (en) | Pharmaceutical preparations containing anti-OX40 monoclonal antibodies | |
| WO2019106206A1 (en) | Stable, low viscosity, high concentration liquid formulations of an anti-il-12/23p40 antibody | |
| WO2021147854A1 (en) | Recombinant fully human anti-tigit monoclonal antibody preparations, preparation method therefor and use thereof | |
| JP2024527517A (en) | Anti-PD1 antibody preparation | |
| RU2745814C1 (en) | Aqueous pharmaceutical composition of levilimab and the use thereof | |
| TW202227130A (en) | Stable pharmaceutical formulation, vial, cartridge, pre-filled syringe and auto-injector comprising the same | |
| WO2020259593A1 (en) | Preparations containing anti-lag-3 antibody, and preparation method therefor and use thereof | |
| CN119095618B (en) | Compositions and methods for treating macrophage activation syndrome | |
| KR20230032347A (en) | Stable pharmaceutical formulation | |
| CN121490074A (en) | Compositions and methods for treating macrophage activation syndrome | |
| HK40000750A (en) | Antibody formulations | |
| HK1261758A1 (en) | Antibody formulations | |
| HK40079581A (en) | Preparation comprising anti-il-23p19 antibody, preparation method therefor and use thereof | |
| HK40000750B (en) | Antibody formulations |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18815598 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 18815598 Country of ref document: EP Kind code of ref document: A1 |