WO2018219773A1 - Fungicidal compositions - Google Patents
Fungicidal compositions Download PDFInfo
- Publication number
- WO2018219773A1 WO2018219773A1 PCT/EP2018/063641 EP2018063641W WO2018219773A1 WO 2018219773 A1 WO2018219773 A1 WO 2018219773A1 EP 2018063641 W EP2018063641 W EP 2018063641W WO 2018219773 A1 WO2018219773 A1 WO 2018219773A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- compound
- phenyl
- component
- trifluoromethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- UKQPJPIWKPHPKR-UHFFFAOYSA-N BCc(c(F)c1)ccc1-c1n[o]c(C(F)(F)F)n1 Chemical compound BCc(c(F)c1)ccc1-c1n[o]c(C(F)(F)F)n1 UKQPJPIWKPHPKR-UHFFFAOYSA-N 0.000 description 1
- 0 CCC(C(*)C1*)=CC=C1c1n[o]c(C(F)(F)F)n1 Chemical compound CCC(C(*)C1*)=CC=C1c1n[o]c(C(F)(F)F)n1 0.000 description 1
- HEETXOBWZIEQBI-UHFFFAOYSA-N O=C1N(Cc(cc2)ccc2-c2n[o]c(C(F)(F)F)n2)CCCC1 Chemical compound O=C1N(Cc(cc2)ccc2-c2n[o]c(C(F)(F)F)n2)CCCC1 HEETXOBWZIEQBI-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/82—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with three ring hetero atoms
Definitions
- the present invention relates to novel fungicidal compositions, to their use in agriculture or horticulture for controlling diseases caused by phytopathogens, especially phytopathogenic fungi, and to methods of controlling diseases on useful plants.
- oxadiazole derivatives are known as insecticidal and acaricidal agents, eg, from CN 1927860.
- WO 2013/064079, EP 0 276 432 and WO 2015/185485 describe the use of substituted oxadiazoles for combating phytopathogenic fungi.
- compositions comprising mixtures of different fungicidal compounds possessing different modes of action can address some of these needs (eg, by combining fungicides with differing spectrums of activity).
- fungicidal composition comprising a mixture of components (A) and (B) as active ingredients, wherein component (A) is a compound of formula (I):
- R hydrogen or fluoro
- a 1 is selected from -CH2-, -O- or -N(CH 3 )-;
- a 2 is selected from -CH2- or -0-;
- Z is optionally substituted by 1 or 2 methyl groups, and at least one of A 1 and A 2 is -CH2-
- a 3 is selected from -CH2- or -0-;
- a 4 is selected from -CH2- or -0-;
- Z 2 is optionally substituted by 1 or 2 methyl groups, and at least one of A 3 and A 4 is -CH2- and
- component (B) is a compound selected from the group consisting of: benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, sedaxane, bixafen, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, propiconazole, epoxiconazole, flutriafol, mefentrifluconazole, ipconazole, paclobutrazol, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, oxathiapi
- the weight ratio of component (A) to component (B) may preferably be from 100: 1 to 1 : 100, from 50:1 to 1 :50, from 20: 1 to 1 :40, from 15:1 to 1 :30, from 12:1 to 1 :25, from 10:1 to 1 :20, from 5: 1 and 1 : 15, from 3: 1 to 1 : 10 or from 2: 1 to 1 :5.
- a method of controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi, on useful plants or on propagation material thereof which comprises applying to the useful plants, the locus thereof or propagation material thereof a fungicidal composition according to the invention.
- fungicidal mixture compositions according to the invention may also include, inter alia, advantageous levels of biological activity for protecting plants against diseases that are caused by fungi or superior properties for use as agrochemical active ingredients (for example, greater biological activity, an advantageous spectrum of activity, an increased safety profile, improved physico-chemical properties, or increased biodegradability).
- the presence of one or more possible asymmetric carbon atoms in a compound of formula (I) means that the compounds may occur in optically isomeric forms, i.e., enantiomeric or diastereomeric forms. Also atropisomers may occur as a result of restricted rotation about a single bond.
- the present invention includes all those possible isomeric forms (e.g. geometric isomers) and mixtures thereof for a compound of formula (I).
- the present invention includes all possible tautomeric forms for a compound of formula (I), and also a racemic compound, i.e., a mixture of at least two enantiomers in a ratio of substantially 50:50.
- the compounds of formula (I) according to the invention are in free form, in oxidized form as a N-oxide or in salt form, e.g. an agronomically usable salt form.
- N-oxides are oxidized forms of tertiary amines or oxidized forms of nitrogen containing heteroaromatic compounds. They are described for instance in the book "Heterocyclic N-oxides" by A. Albini and S. Pietra, CRC Press, Boca Raton 1991.
- Preferred groups and values for the substituents R ⁇ R 2 , Z ⁇ Z 2 , Z 3 , A 1 , A 2 , A 3 and A 4 in the compounds of formula (I) are, in any combination thereof, as set out below.
- R and R 2 are both hydrogen;
- a 1 and A 2 are -CH2-, wherein Z 2 is optionally substituted by 1 or 2 methyl groups;
- component (A) is a compound selected from compound no. X.01 , X.02, X.03, X.04, X.05, X.06, X.07, X.08, X.09, X.10, X.1 1 , X.12, X.13, X.14, X.15, X.16, X.17, X.18 or X.19 as defined in the Table X below. More preferably, component (A) is a compound selected from compound no.
- component (A) is a compound selected from compound no. X.02, X.04, X.06, X.07 and X.19 as defined in the Table X below.
- component (B) is a compound selected from the group consisting of: benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, metalaxyl-M, fenpropidin, fenpropimorph, cyprodinil, spiroxamine, mancozeb, chlorothalonil, oxathiapiprolin, mandipropamid, fluazinam, fosetyl-aluminium, trinexapac-ethyl, acibenzolar-S-methyl, Timorex GoldTM (plant extract comprising tea tree oil), glyphosate
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N-methyl-formamidine.
- component (B) is a compound selected from the group consisting of benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine.
- component (B) compounds are referred to herein and above by a so-called "ISO common name” or another "common name” being used in individual cases or a trademark name.
- the component (B) compounds are known and are commercially available and/or can be prepared using procedures known in the art and/or procedures reported in the literature.
- component (A) is compound no.
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2-methyl-6-(1-methyl-2-propoxy-e
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-
- component (A) is compound no. X.03 [4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]morpholin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo
- component (A) is compound no. X.04 [4,4-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil
- component (A) is compound no. X.05 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-[5-[5-[5
- component (A) is compound no. X.06 [5,5-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3
- component (A) is compound no. X.08 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-brom
- component (A) is compound no. X.09 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo
- component (A) is compound no. X.10 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazinan-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-bromo-2-
- component (A) is compound no. X.1 1 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bro
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-bromo-2- methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyri
- component (A) is compound no. X.13 [1-methyl-3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]imidazolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-brom
- component (A) is compound no. X.14 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-3,3-dimethyl-piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil,
- component (A) is compound no. X.15 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bro
- component (A) is compound no. X.16 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-4,4-dimethyl-isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalon
- component (B) is a compound selected from of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2-methyl-6-(1-methyl-2-propoxy-
- component (A) is compound no. X.18 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-[5-fluoro-4-
- component (A) is compound no. X.19 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]azepan-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-bromo-2- methyl
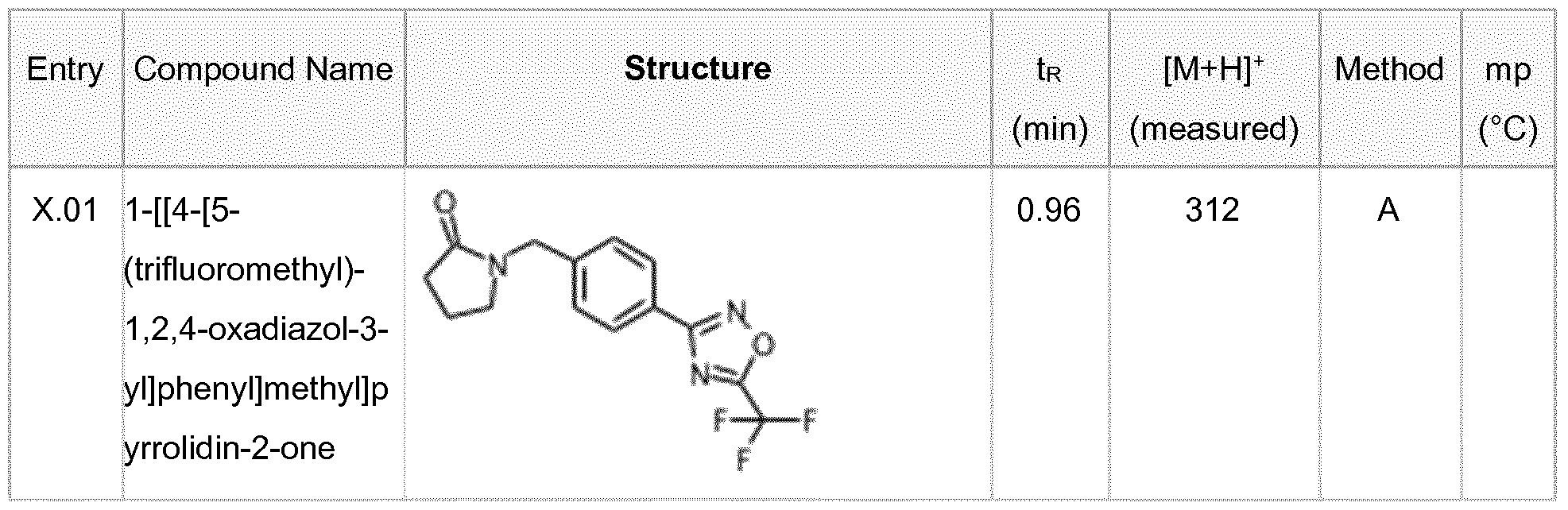
- component (A) is compound no. X.01 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15: 1 to 1 :30.
- component (A) is compound no. X.02 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.03 [4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]morpholin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected frombenzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.04 [4,4-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N-methyl- formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.05 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.06 [5,5-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.07 [3,3-dimethyl-1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.08 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.09 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.10 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazinan-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-nriethyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.1 1 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.12 [3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.13 [1-methyl-3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]imidazolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15: 1 to 1 :30.
- component (A) is compound no. X.14 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-3,3-dimethyl-piperidin-2- one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.15 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.16 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-4,4-dimethyl- isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2- propoxy-ethoxy)-3-pyridyl]-N-ethyl-N-methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15: 1 to 1 :30
- component (A) is compound no. X.17 [2-[[2,3-difluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.18 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15:1 to 1 :30.
- component (A) is compound no. X.19 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]azepan-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 15: 1 to 1 :30.
- component (A) is compound no. X.01 [1-[[4- [5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bro
- component (A) is compound no. X.02 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-[5-[5-[5-[
- component (A) is compound no. X.03 [4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]morpholin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-brovindiflupyr, fluxapyrox
- component (A) is compound no. X.04 [4,4-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chloro
- component (A) is compound no. X.05 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from the group consisting of benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[
- component (A) is compound no. X.06 [5,5-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N
- component (A) is compound no. X.07 [3,3-dimethyl-1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin- 2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5
- component (A) is compound no. X.08 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2- one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-[5-[5-flupyr, flux
- component (A) is compound no. X.09 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2- one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-bro
- component (A) is compound no. X.10 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazinan-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2
- component (A) is compound no. X.1 1 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2- one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5
- component (A) is compound no. X.12 [3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo
- component (A) is compound no. X.13 [1-methyl-3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]imidazolidin- 2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5-[5-[5-[5-[
- component (A) is compound no. X.14 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-3,3-dimethyl- piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalon
- component (A) is compound no. X.15 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2- one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[5
- component (A) is compound no. X.16 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-4,4-dimethyl- isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chloro
- component (A) is compound no. X.17 [2-[[2,3-difluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil,
- component (A) is compound no. X.18 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin- 3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'-[
- component (A) is compound no. X.19 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]azepan-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, fluxapyroxad, pydiflumetofen, isopyrazam, fluopyram, penthiopyrad, difenoconazole, cyproconazole, tebuconazole, hexaconazole, prothioconazole, mefentrifluconazole, azoxystrobin, trifloxystrobin, picoxystrobin, pyraclostrobin, fenpropidin, fenpropimorph, mancozeb, chlorothalonil, N'- [5-bromo-2-
- component (A) is compound no. X.01 [1-[[4- [5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5- bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N-methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10: 1 to 1 :10 (or even more preferably, 5:1 to 1 :5)
- component (A) is compound no. X.02 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1 to 1 :
- component (A) is compound no. X.03 [4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]morpholin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1 to 1 :5)
- component (A) is compound no. X.04 [4,4-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even
- component (A) is compound no. X.05 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1 to 1
- component (A) is compound no. X.06 [5,5-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more
- component (A) is compound no. X.07 [3,3-dimethyl-1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5
- component (A) is compound no. X.08 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:
- component (A) is compound no. X.09 [1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1
- component (A) is compound no. X.10 [2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazinan-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1 to 1
- component (A) is compound no. X.1 1 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]piperidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5
- component (A) is compound no. X.12 [3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]oxazolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1 to 1
- component (A) is compound no. X.13 [1-methyl-3-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]imidazolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:
- component (A) is compound no. X.14 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-3,3-dimethyl-piperidi one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even
- component (A) is compound no. X.15 [1-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]pyrrolidin-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5
- component (A) is compound no. X.16 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]-4,4-dimethyl- isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2- propoxy-ethoxy)-3-pyridyl]-N-ethyl-N-methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10: 1 to 1 :10
- component (A) is compound no. X.17 [2-[[2,3-difluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more
- component (A) is compound no. X.18 [2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]isoxazolidin-3-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably,
- component (A) is compound no. X.19 [1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yl]phenyl]methyl]azepan-2-one] or a salt, enantiomer, tautomer or N-oxide thereof
- component (B) is a compound selected from benzovindiflupyr, pydiflumetofen, difenoconazole, cyproconazole, hexaconazole, prothioconazole, azoxystrobin, fenpropidin, or N'-[5-bromo-2-methyl-6-(1-methyl-2-propoxy-ethoxy)-3-pyridyl]-N-ethyl-N- methyl-formamidine, wherein the weight ratio of component (A) to component (B) is from 10:1 to 1 :10 (or even more preferably, 5:1 to 1 :
- fungicide as used herein means a compound that controls, modifies, or prevents the growth of fungi.
- fungicidally effective amount means the quantity of such a compound or combination of such compounds that is capable of producing an effect on the growth of fungi. Controlling or modifying effects include all deviation from natural development, such as killing, retardation and the like, and prevention includes barrier or other defensive formation in or on a plant to prevent fungal infection.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- plant propagation material denotes all generative parts of a plant, for example seeds or vegetative parts of plants such as cuttings and tubers. It includes seeds in the strict sense, as well as roots, fruits, tubers, bulbs, rhizomes, and parts of plants.
- locus means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
- composition stands for the various mixtures or combinations of components (A) and (B) (including the above-defined embodiments), for example in a single “ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a "tank-mix", and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- the order of applying the components (A) and (B) is not essential for working the present invention.
- composition according to the invention is effective against harmful microorganisms, such as microorganisms, that cause phytopathogenic diseases, in particular against phytopathogenic fungi and bacteria.
- composition of the invention may be used to control plant diseases caused by a broad spectrum of fungal plant pathogens in the Basidiomycete, Ascomycete, Oomycete and/or Deuteromycete, Blasocladiomycete, Chrytidiomycete, Glomeromycete and/or Mucoromycete classes.
- the composition is effective in controlling a broad spectrum of plant diseases, such as foliar pathogens of ornamental, turf, vegetable, field, cereal, and fruit crops.
- pathogens may include: Oomycetes, including Phytophthora diseases such as those caused by Phytophthora capsici, Phytophthora infestans, Phytophthora sojae, Phytophthora fragariae, Phytophthora nicotianae, Phytophthora cinnamomi, Phytophthora citricola, Phytophthora citrophthora and Phytophthora erythroseptica; Pythium diseases such as those caused by Pythium aphanidermatum, Pythium arrhenomanes, Pythium graminicola, Pythium irregulare and Pythium ultimum; diseases caused by Peronosporales such as Peronospora destructor, Peronospora parasitica, Plasmopara viticola, Plasmopara halstedii, Pseudoperonospora cubensis, Albugo Candida, Scleroph
- Ascomycetes including blotch, spot, blast or blight diseases and/or rots for example those caused by Pleosporales such as Stemphylium solani, Stagonospora tainanensis, Spilocaea oleaginea, Setosphaeria turcica, Pyrenochaeta lycoperisici, Pleospora herbarum, Phoma destructiva, Phaeosphaeria herpotrichoides, Phaeocryptocus gaeumannii, Ophiosphaerella graminicola, Ophiobolus graminis, Leptosphaeria maculans, Hendersonia creberrima, Helminthosporium triticirepentis, Setosphaeria turcica, Drechslera glycines, Didymella bryoniae, Cycloconium oleagineum, Corynespora cassiicola, Cochliobolus sativus, Bi
- Valsa ceratosperma and others such as Actinothyrium graminis, Ascochyta pisi, Aspergillus flavus, Aspergillus fumigatus, Aspergillus nidulans, Asperisporium caricae, Blumeriella jaapii, Candida spp.
- Gerlachia nivale Gibberella fujikuroi
- Gibberella zeae Gibberella zeae
- Gliocladium spp. Myrothecium verrucaria, Nectria ramulariae, Trichoderma viride, Trichothecium roseum, and Verticillium theobromae;
- Basidiomycetes including smuts for example those caused by Ustilaginales such as Ustilaginoidea virens, Ustilago nuda, Ustilago tritici, Ustilago zeae, rusts for example those caused by Pucciniales such as Cerotelium fici, Chrysomyxa arctostaphyli, Coleosporium ipomoeae, Hemileia vastatrix, Puccinia arachidis, Puccinia cacabata, Puccinia graminis, Puccinia recondita, Puccinia sorghi, Puccinia hordei, Puccinia striiformis f.sp.
- Ustilaginales such as Ustilaginoidea virens, Ustilago nuda, Ustilago tritici, Ustilago zeae
- rusts for example those caused by Pucciniales such as Cerotelium fici, Chr
- Puccinia striiformis f.sp. Secalis Pucciniastrum coryli, or Uredinales such as Cronartium ribicola, Gymnosporangium juniperi-viginianae, Melampsora medusae, Phakopsora pachyrhizi, Phragmidium mucronatum, Physopella ampelosidis, Tranzschelia discolor and Uromyces viciae-fabae; and other rots and diseases such as those caused by Cryptococcus spp., Exobasidium vexans, Marasmiellus inoderma, Mycena spp., Sphacelotheca reiliana, Typhula ishikariensis, Urocystis agropyri, Itersonilia perplexans, Corticium invisum, Laetisaria fuciformis, Waitea circinata, Rhizoctonia solani, Tha
- Blastocladiomycetes such as Physoderma maydis
- Mucoromycetes such as Choanephora cucurbitarum.; Mucor spp.; Rhizopus arrhizus;
- compositions may also have activity against bacteria such as Erwinia amylovora, Erwinia caratovora, Xanthomonas campestris, Pseudomonas syringae, Strptomyces scabies and other related species as well as certain protozoa.
- bacteria such as Erwinia amylovora, Erwinia caratovora, Xanthomonas campestris, Pseudomonas syringae, Strptomyces scabies and other related species as well as certain protozoa.
- composition according to the invention is particularly effective against phytopathogenic fungi belonging to the following classes: Ascomycetes (e.g. Venturia, Podosphaera, Erysiphe, Monilinia, Mycosphaerella, Uncinula); Basidiomycetes (e.g. the genus Hemileia, Rhizoctonia, Phakopsora, Puccinia, Ustilago, Tilletia); Fungi imperfecti (also known as Deuteromycetes; e.g.
- Ascomycetes e.g. Venturia, Podosphaera, Erysiphe, Monilinia, Mycosphaerella, Uncinula
- Basidiomycetes e.g. the genus Hemileia, Rhizoctonia, Phakopsora, Puccinia, Ustilago, Tilletia
- Fungi imperfecti also known as Deuteromycetes; e.g.
- Botrytis Helminthosporium, Rhynchosporium, Fusarium, Septoria, Cercospora, Alternaria, Pyricularia and Pseudocercosporella); Oomycetes (e.g. Phytophthora, Peronospora, Pseudoperonospora, Albugo, Bremia, Pythium, Pseudosclerospora, Plasmopara).
- Crops of useful plants in which the composition according to the invention can be used include perennial and annual crops, such as berry plants for example blackberries, blueberries, cranberries, raspberries and strawberries; cereals for example barley, maize (corn), millet, oats, rice, rye, sorghum triticale and wheat; fibre plants for example cotton, flax, hemp, jute and sisal; field crops for example sugar and fodder beet, coffee, hops, mustard, oilseed rape (canola), poppy, sugar cane, sunflower, tea and tobacco; fruit trees for example apple, apricot, avocado, banana, cherry, citrus, nectarine, peach, pear and plum; grasses for example Bermuda grass, bluegrass, bentgrass, centipede grass, fescue, ryegrass, St.
- perennial and annual crops such as berry plants for example blackberries, blueberries, cranberries, raspberries and strawberries
- cereals for example barley, maize (corn), mille
- Augustine grass and Zoysia grass herbs such as basil, borage, chives, coriander, lavender, lovage, mint, oregano, parsley, rosemary, sage and thyme; legumes for example beans, lentils, peas and soya beans; nuts for example almond, cashew, ground nut, hazelnut, peanut, pecan, pistachio and walnut; palms for example oil palm; ornamentals for example flowers, shrubs and trees; other trees, for example cacao, coconut, olive and rubber; vegetables for example asparagus, aubergine, broccoli, cabbage, carrot, cucumber, garlic, lettuce, marrow, melon, okra, onion, pepper, potato, pumpkin, rhubarb, spinach and tomato; and vines for example grapes.
- herbs such as basil, borage, chives, coriander, lavender, lovage, mint, oregano, parsley, rosemary, sage and thyme
- legumes for example beans, lentils, peas and soya beans
- Crops are to be understood as being those which are naturally occurring, obtained by conventional methods of breeding, or obtained by genetic engineering. They include crops which contain so-called output traits (e.g. improved storage stability, higher nutritional value and improved flavour).
- output traits e.g. improved storage stability, higher nutritional value and improved flavour.
- Crops are to be understood as also including those crops which have been rendered tolerant to herbicides like bromoxynil or classes of herbicides such as ALS-, EPSPS-, GS-, HPPD- and PPO- inhibitors.
- herbicides like bromoxynil or classes of herbicides such as ALS-, EPSPS-, GS-, HPPD- and PPO- inhibitors.
- An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding is Clearfield® summer canola.
- crops that have been rendered tolerant to herbicides by genetic engineering methods include e.g. glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names Round upReady®, Herculex I® and LibertyLink®.
- Crops are also to be understood as being those which naturally are or have been rendered resistant to harmful insects. This includes plants transformed by the use of recombinant DNA techniques, for example, to be capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria. Examples of toxins which can be expressed include ⁇ -endotoxins, vegetative insecticidal proteins (Vip), insecticidal proteins of bacteria colonising nematodes, and toxins produced by scorpions, arachnids, wasps and fungi.
- Vip vegetative insecticidal proteins
- insecticidal proteins of bacteria colonising nematodes and toxins produced by scorpions, arachnids, wasps and fungi.
- An example of a crop that has been modified to express the Bacillus thuringiensis toxin is the Bt maize KnockOut® (Syngenta Seeds).
- An example of a crop comprising more than one gene that codes for insecticidal resistance and thus expresses more than one toxin is VipCot® (Syngenta Seeds).
- Crops or seed material thereof can also be resistant to multiple types of pests (so-called stacked transgenic events when created by genetic modification).
- a plant can have the ability to express an insecticidal protein while at the same time being herbicide tolerant, for example Herculex I® (Dow AgroSciences, Pioneer Hi-Bred International).
- the compounds of Formula (I) may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Phakopsora pachyrhizi) on soy bean plants.
- transgenic soybean plants expressing toxins for example insecticidal proteins such as delta-endotoxins, e.g. CrylAc (Cry1 Ac Bt protein).
- toxins for example insecticidal proteins such as delta-endotoxins, e.g. CrylAc (Cry1 Ac Bt protein).
- this may include transgenic soybean plants comprising event MON87701 (see U.S. Patent No. 8,049,071 and related applications and patents, as well as WO 2014/170327 A1 (eg, see paragraph [008] reference to Intacta RR2 PROTM soybean)), event MON87751 (US. Patent Application Publication No. 2014/0373191 ) or event DAS- 81419 (U.S. Patent No. 8632978 and related applications and patents).
- event MON87701 see U.S. Patent No. 8,049,071 and related applications and patents, as well as WO 2014/170327 A1 (eg, see paragraph [008] reference to In
- transgenic soybean plants may comprise event SYHT0H2 - HPPD tolerance (U.S. Patent Application Publication No. 2014/0201860 and related applications and patents), event MON89788 - glyphosate tolerance (U.S. Pat. No. 7,632,985 and related applications and patents), event MON87708
- event DAS-40278-9 - tolerance to 2,4- dichlorophenoxyacetic acid and aryloxyphenoxypropionate see WO 201 1/022469, WO 201 1/022470, WO 201 1/022471 , and related applications and patents
- event 127 - ALS tolerance WO 2010/080829 and related applications and patents
- event GTS 40-3-2 - glyphosate tolerance event DAS-68416-4- 2,4-dichlorophenoxyacetic acid and glufosinate tolerance
- event FG72 - glyphosate and isoxaflutole tolerance event BPS-CV127-9 - ALS tolerance and GU262 - glufosinate tolerance or event SYHT04R
- the compounds of Formula (I) may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Phakopsora pachyrhizi) on soy bean plants.
- phytopathogenic diseases especially phytopathogenic fungi (such as Phakopsora pachyrhizi)
- soy bean plants there are known in the scientific literature certain Elite soybean plant varieties where R-gene stacks, conferring a degree of immunity or resistance to specific Phakopsora pachyrhizi, have been been introgressed in the plant genome, see for example: “Fighting Asian Soybean Rust", Langenbach C, ef al, Front Plant Science 7(797) 2016).
- An elite plant is any plant from an elite line, such that an elite plant is a representative plant from an elite variety.
- elite soybean varieties that are commercially available to farmers or soybean breeders include: AG00802, A0868, AG0902, A1923, AG2403, A2824, A3704, A4324, A5404, AG5903, AG6202 AG0934; AG1435; AG2031 ; AG2035; AG2433; AG2733; AG2933; AG3334; AG3832; AG4135; AG4632; AG4934; AG5831 ; AG6534; and AG7231 (Asgrow Seeds, Des Moines, Iowa, USA); BPR0144RR, BPR 4077NRR and BPR 4390NRR (Bio Plant Research, Camp Point, III., USA); DKB17-51 and DKB37-51 (DeKalb Genetics, DeKalb, III., USA); DP 4546 RR, and DP 7870 RR (Delta & Pine Land Company, Lubbock,
- the compounds of Formula (I) are used to control Phakopsora pachyrhizi, (including fungicidally-resistant strains thereof, as outlined below) on Elite soybean plant varieties where R-gene stacks, conferring a degree of immunity or resistance to specific Phakopsora pachyrhizi, have been been introgressed in the plant genome. Numerous benefits may be expected to ensue from said use, e.g. improved biological activity, an advantageous or broader spectrum of activity (inc.
- Phakopsora pachyrhizi sensitive and resistant strains of Phakopsora pachyrhizi
- an increased safety profile improved crop tolerance, synergistic interactions or potentiating properties, improved onset of action or a longer lasting residual activity, a reduction in the number of applications and/or a reduction in the application rate of the compounds and compositions required for effective control of the phytopathogen (Phakopsora pachyrhizi), thereby enabling beneficial resistance-management practices, reduced environmental impact and reduced operator exposure.
- fungicidal compositions according to the present invention comprising a compound of Formula (I) when used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (such as Phakopsora pachyrhizi) on soy bean plants (in particular any of the transgenic soybean plants as described above), may display a synergistic interaction between the active ingredients.
- phytopathogenic diseases especially phytopathogenic fungi (such as Phakopsora pachyrhizi) on soy bean plants (in particular any of the transgenic soybean plants as described above)
- soy bean plants in particular any of the transgenic soybean plants as described above
- the compounds of Formula (I) may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi (in particular, Phakopsora pachyrhizi) on soybean plants.
- the compounds of Formula (I) are used to control Phakopsora pachyrhizi which are resistant to one or more fungicides from any of the following fungicidal MoA classes: sterol demethylation-inhibitors (DMI), quinone-outside- inhibitors (Qol) and succinate dehydrogenase inhibitors (SDHI).
- DMI sterol demethylation-inhibitors
- Qol quinone-outside- inhibitors
- SDHI succinate dehydrogenase inhibitors
- the compounds of formula (I) according to the invention may be present in a reversible equilibrium with the corresponding covalently hydrated forms (ie, the compounds of formula (l-la) and formula (l-lla) as shown below, which may exist in tautomeric form as the compounds of formula (l-lb) and formula (l-llb)) at the CF3-oxadiazole motif.
- This dynamic equilibrium may be important for the biological activity of the compounds of Formula (I).
- Z Z 2 , Z 3 , A 1 , A 2 , A 3 , A 4 , R , and R 2 apply generally to the compounds of Formula (l-la), Formula (l-lla), Formula (l-lb), and Formula (l-llb), as well as to the specific disclosures of combinations Z Z 2 , Z 3 , A 1 , A 2 , A 3 , A 4 , R , and R 2 as represented in the compounds X.01 to X.19 described in Table T1 (below).
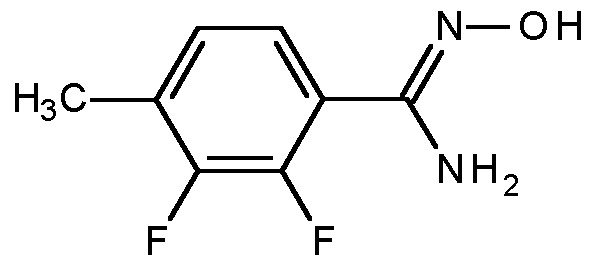
- Compounds of formula (I) can be prepared from compounds of formula (III), wherein X is F, CI, Br, I, OH, or OSCteMe, via treatment with compounds of formula (II), in the presence of a base (eg, lithium hexamethyldisilylazide, n-butyllithium, CS2CO3, CsF, K2CO3 or NaH) in a suitable solvent (eg, dimethylformamide, acetone or tetrahydrofuran) at temperatures between -78°C and 25°C. Additionally, some reactions might only proceed at higher temperatures and/or in the presence of a catalyst system such as Cul/dimethylethylenediamine or Pd(OAc)2/Xantphos.
- a base eg, lithium hexamethyldisilylazide, n-butyllithium, CS2CO3, CsF, K2CO3 or NaH
- a suitable solvent eg, dimethylformamide, acetone
- compounds of formula (I), or compounds of formula (VIII), can be prepared from compounds of formula (IV) or (IVa), respectively, via reactions with trifluoroacetic acid, trifluoroacetic ester (including trifluoroacetic methyl ester, trifluoroacetic ethyl ester), trifluoroacetic anhydride, or trifluoroacetyl halide (including trifluoroacetyl fluoride, trifluoroacetyl chloride, and trifluoroacetyl bromide) in the presence of a base (eg, triethylamine, pyridine or 4-dimethylaminopyridine) in a suitable solvent, (eg, ethyl acetate, tetrahydrofuran, 2-methyltetrahydrofuran, or ethanol), at temperatures between 25°C and 75°C.
- a base eg, triethylamine, pyridine or 4-dimethylaminopyridine
- V Compounds of formula (V) are commercially available or can be prepared from compounds of formula (VI), wherein V is formyl, CI, Br, or I, via metal-promoted reactions with a suitable cyanide reagent, such as acetone cyanohydrin, dimethylmalononitrile, K4[Fe(CN)6] , Zn(CN)2, NaCN, or CuCN, in a suitable solvent (eg, dimethylformamide or N-methylpyrrolidone) at elevated temperatures between 80°C and 120°C, optionally in the presence of a metal catalyst (eg, Pd or Ni) or a Grignard or organolithium reagent.
- a metal catalyst eg, Pd or Ni
- organolithium reagent for related examples, see Reeves, J. T. ei al J.
- compounds of formula (V) can be prepared from compounds of formula (VI), wherein V is NH2, via radical-nucleophilic aromatic substitution reactions in the presence of a nitrite source (eg, NaN02 or /so-amylnitrite), an acid (eg, hydrochloric acid or HBF4), and a copper source (eg, CuCN) in an acceptable solvent system, such as aqueous acetonitrile, at suitable temperatures between 0°C to 100°C.
- a nitrite source eg, NaN02 or /so-amylnitrite
- an acid eg, hydrochloric acid or HBF4
- a copper source eg, CuCN
- Compounds of formula (VI), wherein V is CI, Br or I, or CN can be prepared from compounds of formula (VII), wherein X is F, CI, Br, I, OH, or OS02Me, via treatment with compounds of formula (II), in the presence of a base (eg, lithium hexamethyldisilylazide, n-butyllithium, CS2CO3, CsF, K2CO3 or NaH) in a suitable solvent (eg, dimethylformamide, acetone or tetrahydrofuran) at temperatures between - 78°C and 25°C.
- a base eg, lithium hexamethyldisilylazide, n-butyllithium, CS2CO3, CsF, K2CO3 or NaH
- a suitable solvent eg, dimethylformamide, acetone or tetrahydrofuran
- Compounds of formula (III) wherein X is CI, Br, or I can be prepared from compounds of formula (VIII) by treatment with a halogen source [eg, N-bromosuccimide (NBS), N-chlorosuccimide (NCS), or N-chlorosuccimide (NIS)] and a radical initiator (eg, (PhC02)2 or azobisisobutyronitrile (AIBN)) in a suitable solvent, such as tetrachloromethane, at temperatures between 55°C and 100°C, optionally in the presence of ultraviolet light.
- a halogen source eg, N-bromosuccimide (NBS), N-chlorosuccimide (NCS), or N-chlorosuccimide (NIS)
- a radical initiator eg, (PhC02)2 or azobisisobutyronitrile (AIBN)
- suitable solvent such as tetrachloromethane
- compositions of this invention can be mixed with one or more further pesticides including further fungicides, insecticides, nematocides, bactericides, acaricides, growth regulators, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants or other biologically active compounds to form a multi-component pesticide giving an even broader spectrum of agricultural protection.
- further pesticides including further fungicides, insecticides, nematocides, bactericides, acaricides, growth regulators, chemosterilants, semiochemicals, repellents, attractants, pheromones, feeding stimulants or other biologically active compounds to form a multi-component pesticide giving an even broader spectrum of agricultural protection.
- Fungicides such as etridiazole, fluazinam, benalaxyl, benalaxyl-M (kiralaxyl), furalaxyl, metalaxyl, metalaxyl-M (mefenoxam), dodicin, N'-(2,5-dimethyl-4-phenoxy-phenyl)-N-ethyl-N-methyl- formamidine, N'-[4-(4,5-dichloro-thiazol-2-yloxy)-2,5-dimethyl-phenyl]-N-ethyl-N-methyl-formamidine, N'-[4-[[3-[(4-chlorophenyl)methyl]-1 , 2, 4-th iadiazol-5-yl]oxy]-2,5-dimethyl-phenyl]-N-ethyl-N-m ethyl- formamidine, ethirimol, 3'-chloro-2-methoxy-N
- Insecticides such as abamectin, acephate, acetamiprid, amidoflumet (S-1955), avermectin, azadirachtin, azinphos-methyl, bifenthrin, bifenazate, buprofezin, carbofuran, cartap, chlorantraniliprole (DPX-E2Y45), chlorfenapyr, chlorfluazuron, chlorpyrifos, chlorpyrifos-methyl, chromafenozide, clothianidin, cyflumetofen, cyfluthrin, beta-cyfluthrin, cyhalothrin, lambda-cyhalothrin, cypermethrin, cyromazine, deltamethrin, diafenthiuron, diazinon, dieldrin, diflubenzuron, dimefluthrin, dimethoate, dinote
- Bactericides such as streptomycin
- Acaricides such as amitraz, chinomethionat, chlorobenzilate, cyenopyrafen, cyhexatin, dicofol, dienochlor, etoxazole, fenazaquin, fenbutatin oxide, fenpropathrin, fenpyroximate, hexythiazox, propargite, pyridaben and tebufenpyrad; and
- Biological agents such as Bacillus thuringiensis, Bacillus thuringiensis delta endotoxin, baculovirus, and entomopathogenic bacteria, virus and fungi.
- TX represents a compound (according to the definition of component (A) of the compositions of the present invention) selected from compound no. X.01 , X.02, X.03, X.04, X.05, X.06, X.07, X.08, X.09, X.10, X.1 1 , X.12, X.13, X.14, X.15, X.16, X.17, X.18 or X.19, as defined in the Table X above):
- an adjuvant selected from the group of substances consisting of petroleum oils (alternative name) (628) + TX,
- an acaricide selected from the group of substances consisting of 1 , 1-bis(4-chlorophenyl)-2- ethoxyethanol (lUPAC name) (910) + TX, 2,4-dichlorophenyl benzenesulfonate (lUPAC/Chemical Abstracts name) (1059) + TX, 2-fluoro-A/-methyl-A/-1-naphthylacetamide (lUPAC name) (1295) + TX, 4- chlorophenyl phenyl sulfone (lUPAC name) (981 ) + TX, abamectin (1 ) + TX, acequinocyl (3) + TX, acetoprole [CCN] + TX, acrinathrin (9) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, alpha- cypermethrin (202) + TX, amidithion (870) + TX, amido
- an anthelmintic selected from the group of substances consisting of abamectin (1 ) + TX, crufomate (101 1 ) + TX, doramectin (alternative name) [CCN] + TX, emamectin (291 ) + TX, emamectin benzoate (291 ) + TX, eprinomectin (alternative name) [CCN] + TX, ivermectin (alternative name) [CCN] + TX, milbemycin oxime (alternative name) [CCN] + TX, moxidectin (alternative name) [CCN] + TX, piperazine [CCN] + TX, selamectin (alternative name) [CCN] + TX, spinosad (737) and thiophanate (1435) + TX,
- an avicide selected from the group of substances consisting of chloralose (127) + TX, endrin (1 122) + TX, fenthion (346) + TX, pyridin-4-amine (lUPAC name) (23) and strychnine (745) + TX, a bactericide selected from the group of substances consisting of 1 -hydroxy- 1 - -pyridi ne-2- thione (lUPAC name) (1222) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (lUPAC name) (748) + TX, 8-hydroxyquinoline sulfate (446) + TX, bronopol (97) + TX, copper dioctanoate (lUPAC name) (170) + TX, copper hydroxide (lUPAC name) (169) + TX, cresol [CCN] + TX, dichlorophen (232) + TX, dipyrithione (1 105) + TX, do
- a biological agent selected from the group of substances consisting of Adoxophyes orana GV (alternative name) (12) + TX, Agrobacterium radiobacter (alternative name) (13) + TX, Amblyseius spp. (alternative name) (19) + TX, Anagrapha falcifera NPV (alternative name) (28) + TX, Anagrus atomus (alternative name) (29) + TX, Aphelinus abdominalis (alternative name) (33) + TX, Aphidius colemani (alternative name) (34) + TX, Aphidoletes aphidimyza (alternative name) (35) + TX, Autographa californica NPV (alternative name) (38) + TX, Bacillus firmus (alternative name) (48) + TX, Bacillus sphaericus Neide (scientific name) (49) + TX, Bacillus thuringiensis Hopkins (scientific name) (
- a soil sterilant selected from the group of substances consisting of iodomethane (lUPAC name) (542) and methyl bromide (537) + TX,
- a chemosterilant selected from the group of substances consisting of apholate [CCN] + TX, bisazir (alternative name) [CCN] + TX, busulfan (alternative name) [CCN] + TX, diflubenzuron (250) + TX, dimatif (alternative name) [CCN] + TX, hemel [CCN] + TX, hempa [CCN] + TX, metepa [CCN] + TX, methiotepa [CCN] + TX, methyl apholate [CCN] + TX, morzid [CCN] + TX, penfluron (alternative name) [CCN] + TX, tepa [CCN] + TX, thiohempa (alternative name) [CCN] + TX, thiotepa (alternative name) [CCN] + TX, tretamine (alternative name) [CCN] and
- an insect pheromone selected from the group of substances consisting of (E)-dec-5-en-1-yl acetate with (E)-dec-5-en-1-ol (lUPAC name) (222) + TX, (E)-tridec-4-en-1-yl acetate (lUPAC name) (829) + TX, (E)-6-methylhept-2-en-4-ol (lUPAC name) (541 ) + TX, (E,Z)-tetradeca-4, 10-dien-1-yl acetate (lUPAC name) (779) + TX, (Z)-dodec-7-en-1-yl acetate (lUPAC name) (285) + TX, (Z)-hexadec- 1 1-enal (lUPAC name) (436) + TX, (Z)-hexadec-l 1-en-1-yl acetate (lUPAC name) (437) + TX, (Z)- hexadec
- an insect repellent selected from the group of substances consisting of 2-(octylthio)ethanol (lUPAC name) (591 ) + TX, butopyronoxyl (933) + TX, butoxy(polypropylene glycol) (936) + TX, dibutyl adipate (lUPAC name) (1046) + TX, dibutyl phthalate (1047) + TX, dibutyl succinate (lUPAC name) (1048) + TX, diethylamide [CCN] + TX, dimethyl carbate [CCN] + TX, dimethyl phthalate [CCN] + TX, ethyl hexanediol (1137) + TX, hexamide [CCN] + TX, methoquin-butyl (1276) + TX, methylneodecanamide [CCN] + TX, oxamate [CCN] and picaridin [CCN] + TX,
- an insecticide selected from the group of substances consisting of 1-dichloro-1-nitroethane
- a molluscicide selected from the group of substances consisting of bis(tributyltin) oxide (lUPAC name) (913) + TX, bromoacetamide [CCN] + TX, calcium arsenate [CCN] + TX, cloethocarb (999) + TX, copper acetoarsenite [CCN] + TX, copper sulfate (172) + TX, fentin (347) + TX, ferric phosphate (lUPAC name) (352) + TX, metaldehyde (518) + TX, methiocarb (530) + TX, niclosamide (576) + TX, niclosamide-olamine (576) + TX, pentachlorophenol (623) + TX, sodium pentachlorophenoxide (623) + TX, tazimcarb (1412) + TX, thiodicarb (799) + TX, tributyltin oxide (913)
- a nematicide selected from the group of substances consisting of AKD-3088 (compound code) + TX, 1 ,2-dibromo-3-chloropropane (lUPAC/Chemical Abstracts name) (1045) + TX, 1 ,2- dichloropropane (lUPAC/ Chemical Abstracts name) (1062) + TX, 1 ,2-dichloropropane with 1 ,3- dichloropropene (lUPAC name) (1063) + TX, 1 ,3-dichloropropene (233) + TX, 3,4- dichlorotetrahydrothiophene 1 , 1-dioxide (lUPAC/Chemical Abstracts name) (1065) + TX, 3-(4- chlorophenyl)-5-methylrhodanine (lUPAC name) (980) + TX, 5-methyl-6-thioxo-1 ,3,5-thiadiazinan-3- ylacetic acid (lUPAC name) (1286)
- a nitrification inhibitor selected from the group of substances consisting of potassium ethylxanthate [CCN] and nitrapyrin (580) + TX,
- a plant activator selected from the group of substances consisting of acibenzolar (6) + TX, acibenzolar-S-methyl (6) + TX, probenazole (658) and Reynoutria sachalinensis extract (alternative name) (720) + TX,
- a rodenticide selected from the group of substances consisting of 2-isovalerylindan-1 ,3-dione (lUPAC name) (1246) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (lUPAC name) (748) + TX, alpha-chlorohydrin [CCN] + TX, aluminium phosphide (640) + TX, antu (880) + TX, arsenous oxide (882) + TX, barium carbonate (891 ) + TX, bisthiosemi (912) + TX, brodifacoum (89) + TX, bromadiolone (91 ) + TX, bromethalin (92) + TX, calcium cyanide (444) + TX, chloralose (127) + TX, chlorophacinone (140) + TX, cholecalciferol (alternative name) (850) + TX, coumachlor (1004) + TX, co
- a synergist selected from the group of substances consisting of 2-(2-butoxyethoxy)ethyl piperonylate (lUPAC name) (934) + TX, 5-(1 ,3-benzodioxol-5-yl)-3-hexylcyclohex-2-enone (lUPAC name) (903) + TX, farnesol with nerolidol (alternative name) (324) + TX, MB-599 (development code) (498) + TX, MGK 264 (development code) (296) + TX, piperonyl butoxide (649) + TX, piprotal (1343) + TX, propyl isomer (1358) + TX, S421 (development code) (724) + TX, sesamex (1393) + TX, sesasmolin (1394) and sulfoxide (1406) + TX,
- an animal repellent selected from the group of substances consisting of anthraquinone (32) + TX, chloralose (127) + TX, copper naphthenate [CCN] + TX, copper oxychloride (171 ) + TX, diazinon (227) + TX, dicyclopentadiene (chemical name) (1069) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, methiocarb (530) + TX, pyridin-4-amine (lUPAC name) (23) + TX, thiram (804) + TX, trimethacarb (840) + TX, zinc naphthenate [CCN] and ziram (856) + TX,
- a virucide selected from the group of substances consisting of imanin (alternative name) [CCN] and ribavirin (alternative name) [CCN] + TX,
- a wound protectant selected from the group of substances consisting of mercuric oxide (512) + TX, octhilinone (590) and thiophanate-methyl (802) + TX,
- the mixtures of compounds of formula (I) (selected from Table X (above)) with active ingredients described above comprise a compound selected from Table X (above) and an active ingredient as described above preferably in a mixing ratio of from 100: 1 to 1 : 100, especially from 50:1 to 1 :50, more especially in a ratio of from 20:1 to 1 :20, even more especially from 10:1 to 1 :10, very especially from 5:1 and 1 :5, special preference being given to a ratio of from 2:1 to 1 :2, and a ratio of from 4:1 to 2:1 being likewise preferred, above all in a ratio of 1 : 1 , or 5: 1 , or 5:2, or 5:3, or 5:4, or 4:1 , or 4:2, or 4:3, or 3: 1 , or 3:2, or 2:1 , or 1 :5, or 2:5, or 3:5, or 4:5, or 1 :4, or 2:4, or 3:4, or 1 :3, or 2:3, or 1
- mixture compositions as described above can be used in a method for controlling pests, which comprises applying a composition comprising a mixture as described above to the pests or their environment.
- the mixtures comprising a compound of formula (I) selected from Table X (above) and one or more active ingredients as described above can be applied, for example, in a single "ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a "tank-mix", and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- the order of applying the compounds of formula (I) selected from Table X (above) and the active ingredients as described above is not essential for working the present invention.
- the compositions of the present invention may also be used in crop enhancement. According to the present invention, 'crop enhancement' means an improvement in plant vigour, an improvement in plant quality, improved tolerance to stress factors, and/or improved input use efficiency.
- an 'improvement in plant vigour' means that certain traits are improved qualitatively or quantitatively when compared with the same trait in a control plant which has been grown under the same conditions in the absence of the method of the invention.
- Such traits include, but are not limited to, early and/or improved germination, improved emergence, the ability to use less seeds, increased root growth, a more developed root system, increased root nodulation, increased shoot growth, increased tillering, stronger tillers, more productive tillers, increased or improved plant stand, less plant verse (lodging), an increase and/or improvement in plant height, an increase in plant weight (fresh or dry), bigger leaf blades, greener leaf colour, increased pigment content, increased photosynthetic activity, earlier flowering, longer panicles, early grain maturity, increased seed, fruit or pod size, increased pod or ear number, increased seed number per pod or ear, increased seed mass, enhanced seed filling, less dead basal leaves, delay of senescence, improved vitality of the plant, increased levels of amino acids in storage tissues and/or
- an 'improvement in plant quality' means that certain traits are improved qualitatively or quantitatively when compared with the same trait in a control plant which has been grown under the same conditions in the absence of the method of the invention.
- Such traits include, but are not limited to, improved visual appearance of the plant, reduced ethylene (reduced production and/or inhibition of reception), improved quality of harvested material, e.g. seeds, fruits, leaves, vegetables (such improved quality may manifest as improved visual appearance of the harvested material), improved carbohydrate content (e.g.
- a plant with improved quality may have an increase in any of the aforementioned traits or any combination or two or more of the aforementioned traits.
- an 'improved tolerance to stress factors' means that certain traits are improved qualitatively or quantitatively when compared with the same trait in a control plant which has been grown under the same conditions in the absence of the method of the invention.
- Such traits include, but are not limited to, an increased tolerance and/or resistance to abiotic stress factors which cause sub-optimal growing conditions such as drought (e.g. any stress which leads to a lack of water content in plants, a lack of water uptake potential or a reduction in the water supply to plants), cold exposure, heat exposure, osmotic stress, UV stress, flooding, increased salinity (e.g. in the soil), increased mineral exposure, ozone exposure, high light exposure and/or limited availability of nutrients (e.g.
- a plant with improved tolerance to stress factors may have an increase in any of the aforementioned traits or any combination or two or more of the aforementioned traits. In the case of drought and nutrient stress, such improved tolerances may be due to, for example, more efficient uptake, use or retention of water and nutrients.
- an 'improved input use efficiency' means that the plants are able to grow more effectively using given levels of inputs compared to the grown of control plants which are grown under the same conditions in the absence of the method of the invention.
- the inputs include, but are not limited to fertiliser (such as nitrogen, phosphorous, potassium, micronutrients), light and water.
- a plant with improved input use efficiency may have an improved use of any of the aforementioned inputs or any combination of two or more of the aforementioned inputs.
- crop enhancements of the present invention include a decrease in plant height, or reduction in tillering, which are beneficial features in crops or conditions where it is desirable to have less biomass and fewer tillers. Any or all of the above crop enhancements may lead to an improved yield by improving e.g. plant physiology, plant growth and development and/or plant architecture.
- 'yield' includes, but is not limited to, (i) an increase in biomass production, grain yield, starch content, oil content and/or protein content, which may result from (a) an increase in the amount produced by the plant per se or (b) an improved ability to harvest plant matter, (ii) an improvement in the composition of the harvested material (e.g.
- Improved sugar acid ratios means that, where it is possible to take a quantitative measurement, the yield of a product of the respective plant is increased by a measurable amount over the yield of the same product of the plant produced under the same conditions, but without application of the present invention. According to the present invention, it is preferred that the yield be increased by at least 0.5%, more preferred at least 1 %, even more preferred at least 2%, still more preferred at least 4% , preferably 5% or even more.
- any or all of the above crop enhancements may also lead to an improved utilisation of land, i.e. land which was previously unavailable or sub-optimal for cultivation may become available.
- land i.e. land which was previously unavailable or sub-optimal for cultivation
- plants which show an increased ability to survive in drought conditions may be able to be cultivated in areas of sub-optimal rainfall, e.g. perhaps on the fringe of a desert or even the desert itself.
- crop enhancements are made in the substantial absence of pressure from pests and/or diseases and/or abiotic stress.
- improvements in plant vigour, stress tolerance, quality and/or yield are made in the substantial absence of pressure from pests and/or diseases.
- pests and/or diseases may be controlled by a pesticidal treatment that is applied prior to, or at the same time as, the method of the present invention.
- improvements in plant vigour, stress tolerance, quality and/or yield are made in the absence of pest and/or disease pressure.
- improvements in plant vigour, quality and/or yield are made in the absence, or substantial absence, of abiotic stress.
- compositions of the present invention may also be used in the field of protecting storage goods against attack of fungi.
- the term "storage goods” is understood to denote natural substances of vegetable and/or animal origin and their processed forms, which have been taken from the natural life cycle and for which long-term protection is desired.
- Storage goods of vegetable origin such as plants or parts thereof, for example stalks, leafs, tubers, seeds, fruits or grains, can be protected in the freshly harvested state or in processed form, such as pre-dried, moistened, comminuted, ground, pressed or roasted.
- timber whether in the form of crude timber, such as construction timber, electricity pylons and barriers, or in the form of finished articles, such as furniture or objects made from wood.
- Storage goods of animal origin are hides, leather, furs, hairs and the like.
- the composition according the present invention can prevent disadvantageous effects such as decay, discoloration or mold.
- storage goods is understood to denote natural substances of vegetable origin and/or their processed forms, more preferably fruits and their processed forms, such as pomes, stone fruits, soft fruits and citrus fruits and their processed forms.
- “storage goods” is understood to denote wood.
- a further aspect of the present invention is a method of protecting storage goods, which comprises applying to the storage goods a composition according to the invention.
- composition of the present invention may also be used in the field of protecting technical material against attack of fungi.
- the term "technical material” includes paper; carpets; constructions; cooling and heating systems; wall-boards; ventilation and air conditioning systems and the like; preferably “technical material” is understood to denote wall-boards.
- the composition according the present invention can prevent disadvantageous effects such as decay, discoloration or mold.
- composition according to the invention is generally formulated in various ways using formulation adjuvants, such as carriers, solvents and surface-active substances.
- the formulations can be in various physical forms, e.g. in the form of dusting powders, gels, wettable powders, water- dispersible granules, water-dispersible tablets, effervescent pellets, emulsifiable concentrates, micro- emulsifiable concentrates, oil-in-water emulsions, oil-flowables, aqueous dispersions, oily dispersions, suspo-emulsions, capsule suspensions, emulsifiable granules, soluble liquids, water-soluble concentrates (with water or a water-miscible organic solvent as carrier), impregnated polymer films or in other forms known e.g.
- Such formulations can either be used directly or diluted prior to use.
- the dilutions can be made, for example, with water, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
- the formulations can be prepared e.g. by mixing the active ingredient with the formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions.
- the active ingredients can also be formulated with other adjuvants, such as finely divided solids, mineral oils, oils of vegetable or animal origin, modified oils of vegetable or animal origin, organic solvents, water, surface-active substances or combinations thereof.
- the active ingredients can also be contained in microcapsules.
- Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into the environment in controlled amounts (e.g. slow-release).
- Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight.
- the active ingredients can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution.
- the encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art.
- very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
- liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2-butanone, butylene carbonate, chlorobenzene, cyclohexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1 ,2-dichloropropane, diethanolamine, p- diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, A/,A/-dimethylformamide, dimethyl sulfoxide, 1 ,4- dioxane, di
- Suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montmorillonite, cottonseed husks, wheat flour, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar substances.
- a large number of surface-active substances can advantageously be used in both solid and liquid formulations, especially in those formulations which can be diluted with a carrier prior to use.
- Surface-active substances may be anionic, cationic, non-ionic or polymeric and they can be used as emulsifiers, wetting agents or suspending agents or for other purposes.
- Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol/alkylene oxide addition products, such as nonylphenol ethoxylate; alcohol/alkylene oxide addition products, such as tridecylalcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonat.es, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2- ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryltrimethylammonium chloride, polyethylene glycol est
- Further adjuvants that can be used in pesticidal formulations include crystallisation inhibitors, viscosity modifiers, suspending agents, dyes, anti-oxidants, foaming agents, light absorbers, mixing auxiliaries, antifoams, complexing agents, neutralising or pH-modifying substances and buffers, corrosion inhibitors, fragrances, wetting agents, take-up enhancers, micronutrients, plasticisers, glidants, lubricants, dispersants, thickeners, antifreezes, microbicides, and liquid and solid fertilisers.
- the formulations according to the invention can include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives.
- the amount of oil additive in the formulation according to the invention is generally from 0.01 to 10 %, based on the mixture to be applied.
- the oil additive can be added to a spray tank in the desired concentration after a spray mixture has been prepared.
- Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow.
- Preferred oil additives comprise alkyl esters of C8-C22 fatty acids, especially the methyl derivatives of C12-C18 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid (methyl laurate, methyl palmitate and methyl oleate, respectively).
- Many oil derivatives are known from the Compendium of Herbicide Adjuvants, 10 th Edition, Southern Illinois University, 2010.
- the formulations generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, of compounds of component (A) and component (B) and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- the rates of application vary within wide limits and depend on the nature of the soil, the method of application, the crop plant, the pest to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- a general guideline compounds may be applied at a rate of from 1 to 2000 l/ha, especially from 10 to 1000 l/ha.
- Certain mixture compositions comprising a compound of formula (I) described above may show a synergistic effect. This occurs whenever the action of an active ingredient combination is greater than the sum of the actions of the individual components.
- the action to be expected E for a given active ingredient combination obeys the so-called COLBY formula and can be calculated as follows (COLBY, S.R. "Calculating synergistic and antagonistic responses of herbicide combination". Weeds, Vol. 15, pages 20-22; 1967):
- synergism corresponds to a positive value for the difference of (O-E).
- expected activity said difference (O-E) is zero.
- a negative value of said difference (O-E) signals a loss of activity compared to the expected activity.
- composition according to the invention may also have further surprising advantageous properties.
- advantageous properties are: more advantageous degradability; improved toxicological and/or ecotoxicological behaviour; or improved characteristics of the useful plants including: emergence, crop yields, more developed root system, tillering increase, increase in plant height, bigger leaf blade, less dead basal leaves, stronger tillers, greener leaf colour, less fertilizers needed, less seeds needed, more productive tillers, earlier flowering, early grain maturity, less plant verse (lodging), increased shoot growth, improved plant vigor, and early germination.
- composition according to the invention can be applied to the phytopathogenic microorganisms, the useful plants, the locus thereof, the propagation material thereof, storage goods or technical materials threatened by microorganism attack.
- composition according to the invention may be applied before or after infection of the useful plants, the propagation material thereof, storage goods or technical materials by the microorganisms.
- compositions according to the invention to be applied will depend on various factors, such as the compounds employed; the subject of the treatment, such as, for example plants, soil or seeds; the type of treatment, such as, for example spraying, dusting or seed dressing; the purpose of the treatment, such as, for example prophylactic or therapeutic; the type of fungi to be controlled or the application time.
- component (A) When applied to the useful plants component (A) is typically applied at a rate of 5 to 2000 g a.i./ha, particularly 10 to 1000 g a.i./ha, e.g. 50, 75, 100 or 200 g a.i./ha, typically in association with 1 to 5000 g a.i./ha, particularly 2 to 2000 g a.i./ha, e.g. 100, 250, 500, 800, 1000, 1500 g a.i./ha of component (B).
- the application rates of the composition according to the invention depend on the type of effect desired, and typically range from 20 to 4000 g of total composition per hectare.
- composition according to the invention When the composition according to the invention is used for treating seed, rates of 0.001 to 50 g of a compound of component (A) per kg of seed, preferably from 0.01 to 10g per kg of seed, and 0.001 to 50 g of a compound of component (B), per kg of seed, preferably from 0.01 to 10g per kg of seed, are generally sufficient.
- LC/MS Liquid Chromatography Mass Spectrometry and the description of the apparatus and the method (Methods A and B) is as follows: The description of the LC/MS apparatus and the method A is:
- Type of column Waters ACQUITY UPLC HSS T3; Column length: 30 mm; Internal diameter of column: 2.1 mm; Particle Size: 1.8 micron; Temperature: 60°C.
- Type of column Waters ACQUITY UPLC HSS T3; Column length: 30 mm; Internal diameter of column: 2.1 mm; Particle Size: 1 .8 micron; Temperature: 60°C.
- enantiomerically pure final compounds may be obtained from racemic materials as appropriate via standard physical separation techniques, such as reverse phase chiral chromatography, or through stereoselective synthetic techniques, eg, by using chiral starting materials.
- active ingredients [components (A) and (B)] 25 % 50 % 75 %
- the active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
- active ingredients [components (A) and (B)] 25 % 50 % 75 %
- the active ingredient is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsions of any required dilution which can be used in plant protection, can be obtained from this concentrate by dilution with water.
- Dusts a) b) c) active ingredients [components (A) and (B)] 5 % 6 % 4 % talcum 95 %
- Ready-for-use dusts are obtained by mixing the active ingredient with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
- the active ingredient is mixed and ground with the adjuvants, and the mixture is moistened with water.
- the mixture is extruded and then dried in a stream of air.
- polyethylene glycol (mol. wt. 200)
- the finely ground active ingredient is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner. Suspension concentrate
- nonylphenol polyethylene glycol ether (15 mol of ethylene oxide) 6 %
- the finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- Silicone oil (in the form of a 75 % emulsion in water) 0.2 %
- the finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- This mixture is emulsified in a mixture of 1.2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51.6 parts of water until the desired particle size is achieved.
- a mixture of 2.8 parts 1 ,6- diaminohexane in 5.3 parts of water is added.
- the mixture is agitated until the polymerization reaction is completed.
- the obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent.
- the capsule suspension formulation contains 28% of the active ingredients.
- the medium capsule diameter is 8-15 microns.
- the resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
- Timorex GoldTM active ingredient tea tree (Melaleuca alternifolia) oil
- Stockton Group http://www.stockt.on- ag.com/products/timorex-gold/).
- Example 1 This example illustrates the preparation 2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]isoxazolidin- -one (Compound X.05 of Table T1 )
- Step 1 Preparation of N'-hvdroxy-4-methyl-benzamidine
- hydroxylamine hydrochloride 41.1 g, 0.58 mol
- potassium carbonate 65.4 g, 0.47 mol
- 8-hydroxyquinoline 0.22 g, 1.5 mmol
- the reaction mixture was heated at 80°C for 4 hours.
- the mixture was cooled to room temperature and diluted with 2N HCI until pH 8 and volatiles were then removed under reduced pressure.
- the contents were filtered, washed with water, and dried under vacuum to afford the title compound.
- LC/MS (Method A) retention time 0.23 minutes, 151 .0 (M+H).
- Step 3b Preparation of 3-[4-(bromomethyl)phenyll-5-(trifluoromethvn-1 ,2,4-oxadiazole from 3-[4-
- Step 4 Preparation of 2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yllphenyllmethyllisoxazolidin-3-one
- a solution of 3-[4-(bromomethyl)phenyl]-5-(trifluoromethyl)-1 ,2,4-oxadiazole 150 mg, 0.46 mmol
- isoxazolidin-3-one 61 mg, 0.7 mmol
- potassium carbonate 130 mg, 0.93 mmol
- acetonitrile 4.7 mL
- Example 2 This example illustrates the preparation of 1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]piperidin-2-one (Compound X.02 of Table T1 )
- Step 1 4-[(2-oxo-1-piperidvDmethvHbenzonitrile
- Step 3 4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yllphenyllmethyllmorpholin-3-one
- tetrahydrofuran 2 mL
- trifluoroacetic anhydride 0.27 mL, 1.94 mmol
- Example 3 This example illustrates the preparation of 4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]morpholin-3 )
- Step 3 4-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yllphenyllmethyllmorpholin-3-one
- Example 4 This example illustrates the preparation of 5,5-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4- oxadiazol-3-yl]phenyl]methyl] of Table T1 )
- Step 3 5,5-dimethyl-2-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yllphenyllmethyllisoxazolidin-3-one
- pyridine 0.044 mL, 5.41 mmol
- CFsCOCI 0.530 g, 3.97 mmol
- Example 5 This example illustrates the preparation 2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]phenyl]methyl]-4,4-dimethyl-isoxazolidin-3-one (Compound X.16 of Table T1 )
- Step 2 Preparation of 3-(2-fluoro-4-methyl-phenyl)-5-(trifluoromethyl)-1 ,2,4-oxadiazole
- Step 3b Preparation of 3-[4-(bromomethvn-2-fluoro-phenyll-5-(trifluoromethvn-1 ,2,4-oxadiazole from 3-
- Step 4 Preparation of2-[[3-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yllphenyllmethyll-4,4- dimethyl-isoxazolidin-3-one
- Example 6 This example illustrates the preparation of 1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol- 3-yl]phenyl]methyl]pyrrolidin-2-one (Compound X.08 of Table T1 )
- Step 4 Preparation of 1-[[2-fluoro-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3-yllphenyllmethyllpyrrolidin-2- one
- Step 1 Preparation of 2,3-difluoro-N'-hvdroxy-4-methyl-benzamidine
- Step 3 Preparation of 3-[4-(bromomethyl)-2,3-difluoro-phenyll-5-(trifluoromethyl)-1 ,2,4-oxadiazole
- the mixture was cooled to 25°C, diluted with dichloromethane, water, and the layers were separated.
- the succinimide by-product was filtered off and volatiles were removed under reduced pressure.
- the resultant crude residue was purified by flash chromatography over silica gel (cyclohexane:EtOAc eluent gradient 100:0 to 4: 1 ) to afford 4.8 g of the title compound as a white solid.
- Example 8 This example illustrates the preparation 1-[[4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl] phenyl] methyl]azepan-2-one (Compound X.19 of Table T1 )
- enantiomerically pure final compounds may be obtained from racemic materials as appropriate via standard physical separation techniques, such as reverse phase chiral chromatography, or through stereoselective synthetic techniques, (eg, by using chiral starting materials).
- Table T1 Melting point (mp) data and/or retention times (T.R) for compounds X.01 to X.19 according to
- Leaf disks or leaf segments of various plant species are cut from plants grown in a greenhouse.
- the cut leaf disks or segments are placed in multiwell plates (24-well format) onto water agar.
- the leaf disks are sprayed with a test solution before (preventative) or after (curative) inoculation.
- Compounds to be tested are prepared as DMSO solutions (max. 10 mg/mL) which are diluted to the appropriate concentration with 0.025% Tween20 just before spraying.
- the inoculated leaf disks or segments are incubated under defined conditions (temperature, relative humidity, light, etc.) according to the respective test system.
- a single evaluation of disease level is carried out 3 to 14 days after inoculation, depending on the pathosystem. Percent disease control relative to the untreated check leaf disks or segments is then calculated.
- General examples of liquid culture tests in well plates :
- Mycelia fragments or conidia suspensions of a fungus prepared either freshly from liquid cultures of the fungus or from cryogenic storage, are directly mixed into nutrient broth.
- DMSO solutions of the test compound (max. 10 mg/mL) are diluted with 0.025% Tween20 by a factor of 50 and 10 ⁇ of this solution is pipetted into a microtiter plate (96-well format).
- the nutrient broth containing the fungal spores/mycelia fragments is then added to give an end concentration of the tested compound.
- the test plates are incubated in the dark at 24°C and 96% relative humidity. The inhibition of fungal growth is determined photometrically after 2 to 7 days, depending on the pathosystem, and percent antifungal activity relative to the untreated check is calculated.
- Example A1 Fungicidal activity against Puccinia recondita f. sp. tritici I wheat / leaf disc preventative (Brown rust)
- Wheat leaf segments cv. Kanzler were placed on agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf disks were inoculated with a spore suspension of the fungus 1 day after application.
- the inoculated leaf segments were incubated at 19 C and 75% relative humidity (rh) under a light regime of 12 hours light / 12 hours darkness in a climate cabinet and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (7 to 9 days after application).
- Wheat leaf segments cv. Kanzler are placed on agar in multiwell plates (24-well format). The leaf segments are then inoculated with a spore suspension of the fungus. Plates were stored in darkness at 19°C and 75% relative humidity. The formulated test compound diluted in water was applied 1 day after inoculation. The leaf segments were incubated at 19°C and 75% relative humidity under a light regime of 12 hours light / 12 hours darkness in a climate cabinet and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (6 to 8 days after application).
- the following compounds at 200 ppm in the applied formulation give at least 80% disease control in this test when compared to untreated control leaf disks under the same conditions, which show extensive disease development.
- Example A3 Fungicidal activity against Phakopsora pachyrhizil soybean / leaf disc preventative (Asian soybean rust) Soybean leaf disks are placed on water agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water. One day after application leaf discs are inoculated by spraying a spore suspension on the lower leaf surface. After an incubation period in a climate cabinet of 24-36 hours in darkness at 20°C and 75% relative humidity leaf disc are kept at 20°C with 12 h light/day and 75% rh. The activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (12 to 14 days after application).
- the following compounds at 200 ppm in the applied formulation give at least 80% disease control in this test when compared to untreated control leaf disks under the same conditions, which show extensive disease development.
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB - potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24 C and the inhibition of growth is measured photometrically 3 to 4 days after application.
- nutrient broth PDB - potato dextrose broth
- the following compounds at 20 ppm in the applied formulation give at least 80% disease control in this test when compared to untreated control under the same conditions, which show extensive disease development.
- Example B1a Preventative activity against Phakopsora pachyrhizi on soybean 4-Week old soybean plants are sprayed in a spray chamber with a tank-mix of formulated test compounds diluted in water.
- Leaf disks are cut from treated plants and placed on agar into 24-well plates one day after application.
- Leaf disks are inoculated by spraying them with a spore suspension on their lower leaf surface. After an incubation period in a climate cabinet of 24-36 hours in darkness at 20° C and 75% relative humidity, the leaf disks are then kept at 20° C with 12 hours light/day and 75% relative humidity. The percentage leaf disk area covered by disease is assessed when an appropriate level of disease appears on untreated check plants (10 - 14 days after application).
- Soybean leaf disks are placed on water agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water.
- leaf discs are inoculated by spraying a spore suspension on the lower leaf surface.
- leaf disc After an incubation period in a climate cabinet of 24-36 hours in darkness at 20°C and 75% relative humidity leaf disc are kept at 20°C with 12 hours light/day and 75% relative humidity.
- the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (12 to 14 days after application).
- Example B2 Uncinula necator l grape / preventive (Powdery mildew on grape)
- Example B3a Glomerella lagenarium (Colletotrichum lagenarium) I cucumber
- Example B3b Glomerella lagenarium (Colletotrichum lagenarium) I cucumber
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB - potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24 °C and the inhibition of growth is measured photometrically 3 to 4 days after application.
- nutrient broth PDB - potato dextrose broth
- Example B4a Puccinia recondita / wheat / preventive (Brown rust on wheat)
- Example B4b Puccinia recondita / wheat / preventive (Brown rust on wheat
- Wheat leaf segments cv. Kanzler were placed on agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf disks were inoculated with a spore suspension of the fungus 1 day after application.
- the inoculated leaf segments were incubated at 19 ° C and 75% relative humidity under a light regime of 12 hours light / 12 hours darkness in a climate cabinet and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (7 to 9 days after application).
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| BR112019024993-1A BR112019024993B1 (en) | 2017-06-02 | 2018-05-24 | FUNGICIDE COMPOSITIONS AND METHOD OF CONTROL OR PREVENTION OF PHYTOPATHOGENIC DISEASES |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17174299 | 2017-06-02 | ||
| EP17174299.2 | 2017-06-02 | ||
| EP17178132 | 2017-06-27 | ||
| EP17178132.1 | 2017-06-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018219773A1 true WO2018219773A1 (en) | 2018-12-06 |
Family
ID=62244503
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2018/063641 Ceased WO2018219773A1 (en) | 2017-06-02 | 2018-05-24 | Fungicidal compositions |
Country Status (2)
| Country | Link |
|---|---|
| UY (1) | UY37754A (en) |
| WO (1) | WO2018219773A1 (en) |
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020120204A3 (en) * | 2018-12-11 | 2020-07-23 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from septoria tritici and puccinia spp. in cereals by compositions comprising mefentrifluconazole |
| WO2020120206A3 (en) * | 2018-12-11 | 2020-07-23 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from phakopsora spp. in soybeans by compositions comprising mefentrifluconazole |
| WO2020120205A3 (en) * | 2018-12-11 | 2020-07-23 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from uncinula necator, plasmopara viticola and gloeosporium ampelophagum in grapes by compositions comprising mefentrifluconazole |
| CN113748105A (en) * | 2019-04-18 | 2021-12-03 | 先正达农作物保护股份公司 | Process for the preparation of microbicidal oxadiazole derivatives |
| CN113767906A (en) * | 2021-08-17 | 2021-12-10 | 陕西美邦药业集团股份有限公司 | Bactericidal composition for preventing and treating bacterial angular leaf spot of muskmelon |
| WO2021251274A1 (en) * | 2020-06-08 | 2021-12-16 | 日本曹達株式会社 | Method for reducing or preventing effect of non-biological stress on plant |
| WO2022117653A1 (en) | 2020-12-02 | 2022-06-09 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2022117650A1 (en) | 2020-12-02 | 2022-06-09 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2022190090A1 (en) * | 2021-03-11 | 2022-09-15 | Stockton (Israel) Ltd | Combinations of fludioxonil and tea tree oil for controlling fungal plant pathogens |
| EP4091451A1 (en) | 2021-05-17 | 2022-11-23 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| EP4094579A1 (en) | 2021-05-28 | 2022-11-30 | Basf Se | Pesticidal mixtures comprising metyltetraprole |
| CN116239587A (en) * | 2021-12-07 | 2023-06-09 | 浙江省化工研究院有限公司 | A class of acyl imide derivatives containing trifluoromethyl oxadiazole, its preparation method and application |
| WO2024068655A1 (en) | 2022-09-28 | 2024-04-04 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2024068656A1 (en) | 2022-09-28 | 2024-04-04 | Syngenta Crop Protection Ag | Fungicidal compositions |
Citations (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0276432A2 (en) | 1986-12-12 | 1988-08-03 | Ciba-Geigy Ag | Pesticides |
| WO2003028729A2 (en) | 2001-10-03 | 2003-04-10 | Pharmacia Corporation | Prodrugs of substituted polycyclic compounds useful for selective inhibition of the coagulation cascade |
| WO2003035617A2 (en) | 2001-10-23 | 2003-05-01 | Dow Agrosciences Llc Patent Department | Derivatives of uk-2a |
| WO2004087646A2 (en) | 2003-04-03 | 2004-10-14 | Merck Patent Gmbh | Pyrrolidino-1,2-dicarboxy-1-(phenylamide)-2-(4-(3-oxo-morpholino-4-yl)-phenylamide) derivatives and related compounds for use as inhibitors of coagulation factor xa in the treatment of thrombo-embolic diseases |
| WO2005070917A1 (en) | 2004-01-23 | 2005-08-04 | Sankyo Agro Company, Limited | 3-(dihydro(tetrahydro)isoquinolin-1-yl)quinolines |
| WO2005121104A1 (en) | 2004-06-09 | 2005-12-22 | Sumitomo Chemical Company, Limited | Pyridazine compound and use thereof |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| CN1927860A (en) | 2005-09-08 | 2007-03-14 | 国家南方农药创制中心江苏基地 | Pyrazolecarboxamide compounds, intermediate thereof and pest control chemicals with the same as active component |
| WO2007031513A1 (en) | 2005-09-13 | 2007-03-22 | Bayer Cropscience Ag | Pesticide thiazolyloxy substituted phenylamidine derivatives |
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2007072999A1 (en) | 2005-12-22 | 2007-06-28 | Nihon Nohyaku Co., Ltd | Pyrazinecarboxamide derivatives and plant disease controlling agents containing the same |
| US20070155739A1 (en) | 2005-12-30 | 2007-07-05 | Alantos Pharmaceuticals, Inc. | Substituted bis-amide metalloprotease inhibitors |
| WO2007129454A1 (en) | 2006-05-08 | 2007-11-15 | Kumiai Chemical Industry Co., Ltd. | 1,2-benzisothiazole derivative, and agricultural or horticultural plant disease-controlling agent |
| US20080196127A1 (en) | 2005-04-11 | 2008-08-14 | Bayer Bioscience N.V. | Elite Event A5547-127 and Methods and Kits For Identifying Such Event in Biological Samples |
| US20080312082A1 (en) | 2006-10-31 | 2008-12-18 | Kinney Anthony J | Soybean event dp-305423-1 and compositions and methods for the identification and/or detection thereof |
| US20080320616A1 (en) | 2005-04-08 | 2008-12-25 | Bayer Bioscience N.V. | Elite Event A2407-12 and Methods and Kits for Identifying Such Event in Biological Samples |
| WO2009022746A1 (en) | 2007-08-10 | 2009-02-19 | Nippon Soda Co., Ltd. | Nitrogen-containing heterocyclic compound and pest control agent |
| US7632985B2 (en) | 2005-05-27 | 2009-12-15 | Monsanto Technology Llc | Soybean event MON89788 and methods for detection thereof |
| WO2010000841A1 (en) | 2008-07-04 | 2010-01-07 | Bayer Cropscience Sa | Fungicide hydroximoyl-tetrazole derivatives |
| WO2010045251A2 (en) | 2008-10-17 | 2010-04-22 | Xenon Pharmaceuticals, Inc. | Spiro-oxindole compounds and their use as therapeutic agents |
| WO2010080829A1 (en) | 2009-01-07 | 2010-07-15 | Basf Agrochemical Products B.V. | Soybean event 127 and methods related thereto |
| US20100184079A1 (en) | 2006-06-28 | 2010-07-22 | Pioneer Hi-Bred International, Inc. | Soybean event 3560.4.3.5 and compositions and methods for the identification and detection thereof |
| WO2010093059A1 (en) | 2009-02-16 | 2010-08-19 | 住友化学株式会社 | Method for producing phenylacetamide compound |
| WO2010130767A2 (en) | 2009-05-15 | 2010-11-18 | Bayer Cropscience Ag | Fungicide pyrazole carboxamides derivatives |
| WO2010146031A2 (en) | 2009-06-16 | 2010-12-23 | Basf Se | Fungicidal mixtures |
| WO2011022471A1 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Detection of aad-1 event das-40278-9 |
| WO2011022470A1 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Control of aad-1 monocot volunteers in fields of dicot crops |
| WO2011022469A2 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Aad-1 event das-40278-9, related transgenic corn lines, and event-specific identification thereof |
| US20110067134A1 (en) | 2009-09-17 | 2011-03-17 | Brinker Ronald J | Soybean transgenic event mon 87708 and methods of use thereof |
| WO2011081174A1 (en) | 2010-01-04 | 2011-07-07 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and agricultural/horticultural germicide |
| US8049071B2 (en) | 2007-11-15 | 2011-11-01 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event MON87701 and methods for detection thereof |
| WO2011138281A2 (en) | 2010-05-06 | 2011-11-10 | Bayer Cropscience Ag | Process for the preparation of dithiine tetracarboxydiimides |
| WO2011162397A1 (en) | 2010-06-24 | 2011-12-29 | Sumitomo Chemical Company, Limited | Plant disease control composition and method of controlling plant disease |
| WO2012020774A1 (en) | 2010-08-10 | 2012-02-16 | 住友化学株式会社 | Plant disease control composition and application for same |
| WO2012025557A1 (en) | 2010-08-25 | 2012-03-01 | Bayer Cropscience Ag | Heteroarylpiperidine and -piperazine derivatives as fungicides |
| WO2012031061A2 (en) | 2010-09-01 | 2012-03-08 | E. I. Du Pont De Nemours And Company | Fungicidal pyrazoles and their mixtures |
| WO2012084812A1 (en) | 2010-12-20 | 2012-06-28 | Isagro Ricerca S.R.L. | Aminoindanes amides having a high fungicidal activity and their phytosanitary compositions |
| WO2012092115A1 (en) | 2010-12-29 | 2012-07-05 | E. I. Du Pont De Nemours And Company | Mesoionic pyrido [1,2 -a] pyrimidine pesticides |
| WO2013024082A1 (en) | 2011-08-15 | 2013-02-21 | Basf Se | Fungicidal substituted 1-{2-cyclyloxy-2-[2-halo-4-(4-halogen-phenoxy)-phenyl]-ethyl}-1h-[1,2,4]triazole compounds |
| WO2013064079A1 (en) | 2011-11-02 | 2013-05-10 | 中国中化股份有限公司 | Use of compound of pyrazole amides as agricultural fungicide |
| WO2013066838A1 (en) | 2011-10-31 | 2013-05-10 | Glaxosmithkline Llc | Compounds and methods |
| WO2013132253A1 (en) | 2012-03-07 | 2013-09-12 | Institute Of Cancer Research: Royal Cancer Hospital (The) | 3-aryl-5-substituted-isoquinolin-1-one compounds and their therapeutic use |
| WO2013162072A1 (en) | 2012-04-27 | 2013-10-31 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and its use as pesticides |
| US8632978B2 (en) | 2011-07-26 | 2014-01-21 | Dow Agrosciences, Llc. | Soybean event pDAB9582.814.19.1 detection method |
| WO2014013842A1 (en) | 2012-07-20 | 2014-01-23 | 住友化学株式会社 | Composition for controlling plant disease and application therefor |
| WO2014051165A1 (en) | 2012-09-28 | 2014-04-03 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and their use as pesticides |
| WO2014095675A1 (en) | 2012-12-19 | 2014-06-26 | Bayer Cropscience Ag | Difluoromethyl-nicotinic-indanyl carboxamides as fungicides |
| US20140201860A1 (en) | 2010-12-15 | 2014-07-17 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2014170327A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Method for combating pests |
| US20140373191A1 (en) | 2013-06-14 | 2014-12-18 | Monsanto Technology, Llc | Soybean transgenic event mon87751 and methods for detection and use thereof |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2016139189A1 (en) | 2015-03-05 | 2016-09-09 | Bayer Cropscience Aktiengesellschaft | Fungicidally active compound combinations |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017085100A1 (en) * | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017093348A1 (en) * | 2015-12-02 | 2017-06-08 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
-
2018
- 2018-05-24 WO PCT/EP2018/063641 patent/WO2018219773A1/en not_active Ceased
- 2018-05-31 UY UY0001037754A patent/UY37754A/en not_active Application Discontinuation
Patent Citations (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0276432A2 (en) | 1986-12-12 | 1988-08-03 | Ciba-Geigy Ag | Pesticides |
| WO2003028729A2 (en) | 2001-10-03 | 2003-04-10 | Pharmacia Corporation | Prodrugs of substituted polycyclic compounds useful for selective inhibition of the coagulation cascade |
| WO2003035617A2 (en) | 2001-10-23 | 2003-05-01 | Dow Agrosciences Llc Patent Department | Derivatives of uk-2a |
| WO2004087646A2 (en) | 2003-04-03 | 2004-10-14 | Merck Patent Gmbh | Pyrrolidino-1,2-dicarboxy-1-(phenylamide)-2-(4-(3-oxo-morpholino-4-yl)-phenylamide) derivatives and related compounds for use as inhibitors of coagulation factor xa in the treatment of thrombo-embolic diseases |
| WO2005070917A1 (en) | 2004-01-23 | 2005-08-04 | Sankyo Agro Company, Limited | 3-(dihydro(tetrahydro)isoquinolin-1-yl)quinolines |
| WO2005121104A1 (en) | 2004-06-09 | 2005-12-22 | Sumitomo Chemical Company, Limited | Pyridazine compound and use thereof |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| US20080320616A1 (en) | 2005-04-08 | 2008-12-25 | Bayer Bioscience N.V. | Elite Event A2407-12 and Methods and Kits for Identifying Such Event in Biological Samples |
| US20080196127A1 (en) | 2005-04-11 | 2008-08-14 | Bayer Bioscience N.V. | Elite Event A5547-127 and Methods and Kits For Identifying Such Event in Biological Samples |
| US7632985B2 (en) | 2005-05-27 | 2009-12-15 | Monsanto Technology Llc | Soybean event MON89788 and methods for detection thereof |
| CN1927860A (en) | 2005-09-08 | 2007-03-14 | 国家南方农药创制中心江苏基地 | Pyrazolecarboxamide compounds, intermediate thereof and pest control chemicals with the same as active component |
| WO2007031513A1 (en) | 2005-09-13 | 2007-03-22 | Bayer Cropscience Ag | Pesticide thiazolyloxy substituted phenylamidine derivatives |
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2007072999A1 (en) | 2005-12-22 | 2007-06-28 | Nihon Nohyaku Co., Ltd | Pyrazinecarboxamide derivatives and plant disease controlling agents containing the same |
| US20070155739A1 (en) | 2005-12-30 | 2007-07-05 | Alantos Pharmaceuticals, Inc. | Substituted bis-amide metalloprotease inhibitors |
| WO2007129454A1 (en) | 2006-05-08 | 2007-11-15 | Kumiai Chemical Industry Co., Ltd. | 1,2-benzisothiazole derivative, and agricultural or horticultural plant disease-controlling agent |
| US20100184079A1 (en) | 2006-06-28 | 2010-07-22 | Pioneer Hi-Bred International, Inc. | Soybean event 3560.4.3.5 and compositions and methods for the identification and detection thereof |
| US20080312082A1 (en) | 2006-10-31 | 2008-12-18 | Kinney Anthony J | Soybean event dp-305423-1 and compositions and methods for the identification and/or detection thereof |
| WO2009022746A1 (en) | 2007-08-10 | 2009-02-19 | Nippon Soda Co., Ltd. | Nitrogen-containing heterocyclic compound and pest control agent |
| US8049071B2 (en) | 2007-11-15 | 2011-11-01 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event MON87701 and methods for detection thereof |
| WO2010000841A1 (en) | 2008-07-04 | 2010-01-07 | Bayer Cropscience Sa | Fungicide hydroximoyl-tetrazole derivatives |
| WO2010045251A2 (en) | 2008-10-17 | 2010-04-22 | Xenon Pharmaceuticals, Inc. | Spiro-oxindole compounds and their use as therapeutic agents |
| WO2010080829A1 (en) | 2009-01-07 | 2010-07-15 | Basf Agrochemical Products B.V. | Soybean event 127 and methods related thereto |
| WO2010093059A1 (en) | 2009-02-16 | 2010-08-19 | 住友化学株式会社 | Method for producing phenylacetamide compound |
| WO2010130767A2 (en) | 2009-05-15 | 2010-11-18 | Bayer Cropscience Ag | Fungicide pyrazole carboxamides derivatives |
| WO2010146031A2 (en) | 2009-06-16 | 2010-12-23 | Basf Se | Fungicidal mixtures |
| WO2011022471A1 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Detection of aad-1 event das-40278-9 |
| WO2011022470A1 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Control of aad-1 monocot volunteers in fields of dicot crops |
| WO2011022469A2 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Aad-1 event das-40278-9, related transgenic corn lines, and event-specific identification thereof |
| US20110067134A1 (en) | 2009-09-17 | 2011-03-17 | Brinker Ronald J | Soybean transgenic event mon 87708 and methods of use thereof |
| WO2011081174A1 (en) | 2010-01-04 | 2011-07-07 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and agricultural/horticultural germicide |
| WO2011138281A2 (en) | 2010-05-06 | 2011-11-10 | Bayer Cropscience Ag | Process for the preparation of dithiine tetracarboxydiimides |
| WO2011162397A1 (en) | 2010-06-24 | 2011-12-29 | Sumitomo Chemical Company, Limited | Plant disease control composition and method of controlling plant disease |
| WO2012020774A1 (en) | 2010-08-10 | 2012-02-16 | 住友化学株式会社 | Plant disease control composition and application for same |
| WO2012025557A1 (en) | 2010-08-25 | 2012-03-01 | Bayer Cropscience Ag | Heteroarylpiperidine and -piperazine derivatives as fungicides |
| WO2012031061A2 (en) | 2010-09-01 | 2012-03-08 | E. I. Du Pont De Nemours And Company | Fungicidal pyrazoles and their mixtures |
| US20140201860A1 (en) | 2010-12-15 | 2014-07-17 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2012084812A1 (en) | 2010-12-20 | 2012-06-28 | Isagro Ricerca S.R.L. | Aminoindanes amides having a high fungicidal activity and their phytosanitary compositions |
| WO2012092115A1 (en) | 2010-12-29 | 2012-07-05 | E. I. Du Pont De Nemours And Company | Mesoionic pyrido [1,2 -a] pyrimidine pesticides |
| US8632978B2 (en) | 2011-07-26 | 2014-01-21 | Dow Agrosciences, Llc. | Soybean event pDAB9582.814.19.1 detection method |
| WO2013024082A1 (en) | 2011-08-15 | 2013-02-21 | Basf Se | Fungicidal substituted 1-{2-cyclyloxy-2-[2-halo-4-(4-halogen-phenoxy)-phenyl]-ethyl}-1h-[1,2,4]triazole compounds |
| WO2013066838A1 (en) | 2011-10-31 | 2013-05-10 | Glaxosmithkline Llc | Compounds and methods |
| WO2013064079A1 (en) | 2011-11-02 | 2013-05-10 | 中国中化股份有限公司 | Use of compound of pyrazole amides as agricultural fungicide |
| WO2013132253A1 (en) | 2012-03-07 | 2013-09-12 | Institute Of Cancer Research: Royal Cancer Hospital (The) | 3-aryl-5-substituted-isoquinolin-1-one compounds and their therapeutic use |
| WO2013162072A1 (en) | 2012-04-27 | 2013-10-31 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and its use as pesticides |
| WO2014013842A1 (en) | 2012-07-20 | 2014-01-23 | 住友化学株式会社 | Composition for controlling plant disease and application therefor |
| WO2014051165A1 (en) | 2012-09-28 | 2014-04-03 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and their use as pesticides |
| WO2014095675A1 (en) | 2012-12-19 | 2014-06-26 | Bayer Cropscience Ag | Difluoromethyl-nicotinic-indanyl carboxamides as fungicides |
| WO2014170327A1 (en) | 2013-04-19 | 2014-10-23 | Bayer Cropscience Ag | Method for combating pests |
| US20140373191A1 (en) | 2013-06-14 | 2014-12-18 | Monsanto Technology, Llc | Soybean transgenic event mon87751 and methods for detection and use thereof |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2016139189A1 (en) | 2015-03-05 | 2016-09-09 | Bayer Cropscience Aktiengesellschaft | Fungicidally active compound combinations |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017085100A1 (en) * | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017093348A1 (en) * | 2015-12-02 | 2017-06-08 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
Non-Patent Citations (20)
| Title |
|---|
| "Compendium of Herbicide Adjuvants", 2010, SOUTHERN ILLINOIS UNIVERSITY |
| "McCutcheon's Detergents and Emulsifiers Annual", 1981, MC PUBLISHING CORP. |
| "The Pesticide Manual'' [The Pesticide Manual - A World Compendium", THE BRITISH CROP PROTECTION COUNCIL |
| A. WOOD, COMPENDIUM OF PESTICIDE COMMON NAMES, 1995 |
| COLBY, S.R.: "Calculating synergistic and antagonistic responses of herbicide combination", WEEDS, vol. 15, 1967, pages 20 - 22, XP001112961 |
| GARCIA, M. ET AL., ORG. BIOMOL. CHEM., vol. 11, 2004, pages 1633 |
| GUYONNER, M.; BAUDOIN, O., ORG. LETT., vol. 14, 2012, pages 398 |
| KITAMURA, S. ET AL., CHEM. PHARM. BULL., vol. 49, 2001, pages 268 |
| KLOSOWSKI AC ET AL.: "Competitive fitness of Phakopsora pachyrhizi isolates with mutations in the CYP51 and CYTB genes", PHYTOPATHOLOGY, vol. 106, 2016, pages 1278 - 1284 |
| KLOSOWSKI AC ET AL.: "Detection of the F129L mutation in the cytochrome b gene in Phakopsora pachyrhizi", PEST MANAG SCI, vol. 72, 2016, pages 1211 - 1215 |
| KOMPELLA, A. ET AL., ORG. PROC. RES. DEV., vol. 16, 2012, pages 1794 |
| LANGENBACH C ET AL.: "Fighting Asian Soybean Rust", FRONT PLANT SCIENCE, vol. 7, no. 797, 2016 |
| LIU, S. ET AL., SYNTHESIS, vol. 14, 2001, pages 2078 |
| PHILIPPS, D. P. ET AL., TETRAHEDRON LETT., vol. 50, 2009, pages 7293 |
| PROC. BCPC, INT. CONGR., vol. 1, 2003, pages 93 |
| REEVES, J. T. ET AL., J. AM. CHEM. SOC., vol. 137, 2015, pages 9481 - 9488 |
| SCHMITZ HK ET AL.: "Sensitivity of Phakopsora pachyrhizi towards quinone-outside-inhibitors and demethylation-inhibitors, and corresponding resistance mechanisms", PEST MANAG SCI, vol. 70, 2014, pages 378 - 388 |
| SIMÕES K ET AL.: "First detection of a SDH variant with reduced SDHI sensitivity in Phakopsora pachyrhizl", J PLANT DIS PROT, vol. 125, 2018, pages 21 - 2 |
| USHIJIMA, S.; TOGO, H., SYNLETT, 2010, pages 1067 |
| WEN, Q. ET AL., TET. LETT., vol. 55, 2014, pages 1271 |
Cited By (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN113163765B (en) * | 2018-12-11 | 2023-04-25 | 巴斯夫农业公司 | Method for controlling phytopathogenic fungi in grapes by a composition comprising penoxsulam |
| WO2020120206A3 (en) * | 2018-12-11 | 2020-07-23 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from phakopsora spp. in soybeans by compositions comprising mefentrifluconazole |
| WO2020120205A3 (en) * | 2018-12-11 | 2020-07-23 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from uncinula necator, plasmopara viticola and gloeosporium ampelophagum in grapes by compositions comprising mefentrifluconazole |
| CN113163765A (en) * | 2018-12-11 | 2021-07-23 | 巴斯夫农业公司 | Method for controlling phytopathogenic fungi selected from the group consisting of erysiphe necator, monascus viticola and botrytis cinerea in grapes by means of a composition comprising chlorofluoromethrizole |
| US20220007646A1 (en) * | 2018-12-11 | 2022-01-13 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from uncinula necator, plasmopara viticola and gloeosporium ampelophagum in grapes by compositions comprising mefentrifluconazole |
| WO2020120204A3 (en) * | 2018-12-11 | 2020-07-23 | BASF Agro B.V. | Method to control a phythopatogenic fungi selected from septoria tritici and puccinia spp. in cereals by compositions comprising mefentrifluconazole |
| CN113748105A (en) * | 2019-04-18 | 2021-12-03 | 先正达农作物保护股份公司 | Process for the preparation of microbicidal oxadiazole derivatives |
| CN113748105B (en) * | 2019-04-18 | 2024-06-11 | 先正达农作物保护股份公司 | Process for the preparation of microbicidal oxadiazole derivatives |
| WO2021251274A1 (en) * | 2020-06-08 | 2021-12-16 | 日本曹達株式会社 | Method for reducing or preventing effect of non-biological stress on plant |
| WO2022117653A1 (en) | 2020-12-02 | 2022-06-09 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2022117650A1 (en) | 2020-12-02 | 2022-06-09 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2022190090A1 (en) * | 2021-03-11 | 2022-09-15 | Stockton (Israel) Ltd | Combinations of fludioxonil and tea tree oil for controlling fungal plant pathogens |
| EP4091451A1 (en) | 2021-05-17 | 2022-11-23 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| EP4094579A1 (en) | 2021-05-28 | 2022-11-30 | Basf Se | Pesticidal mixtures comprising metyltetraprole |
| CN113767906B (en) * | 2021-08-17 | 2024-02-27 | 陕西美邦药业集团股份有限公司 | Sterilization composition for preventing and treating muskmelon bacterial angular leaf spot bacteria |
| CN113767906A (en) * | 2021-08-17 | 2021-12-10 | 陕西美邦药业集团股份有限公司 | Bactericidal composition for preventing and treating bacterial angular leaf spot of muskmelon |
| CN116239587A (en) * | 2021-12-07 | 2023-06-09 | 浙江省化工研究院有限公司 | A class of acyl imide derivatives containing trifluoromethyl oxadiazole, its preparation method and application |
| WO2024068655A1 (en) | 2022-09-28 | 2024-04-04 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2024068656A1 (en) | 2022-09-28 | 2024-04-04 | Syngenta Crop Protection Ag | Fungicidal compositions |
Also Published As
| Publication number | Publication date |
|---|---|
| UY37754A (en) | 2019-01-02 |
| BR112019024993A2 (en) | 2020-06-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12201113B2 (en) | Fungicidal compositions | |
| US11974572B2 (en) | Fungicidal compositions | |
| US11154058B2 (en) | Fungicidal compositions | |
| US10631539B2 (en) | Fungicidal compositions | |
| EP3644743B1 (en) | Fungicidal compositions | |
| US10966425B2 (en) | Fungicidal compositions | |
| WO2018219773A1 (en) | Fungicidal compositions | |
| EP3582616B1 (en) | Fungicidal compositions | |
| WO2018177880A1 (en) | Fungicidal compositions | |
| WO2020064696A1 (en) | Fungicidal compositions | |
| WO2017067837A1 (en) | Microbiocidal phenylamidine derivatives | |
| BR112019024993B1 (en) | FUNGICIDE COMPOSITIONS AND METHOD OF CONTROL OR PREVENTION OF PHYTOPATHOGENIC DISEASES |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 18726998 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112019024993 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 112019024993 Country of ref document: BR Kind code of ref document: A2 Effective date: 20191127 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 18726998 Country of ref document: EP Kind code of ref document: A1 |