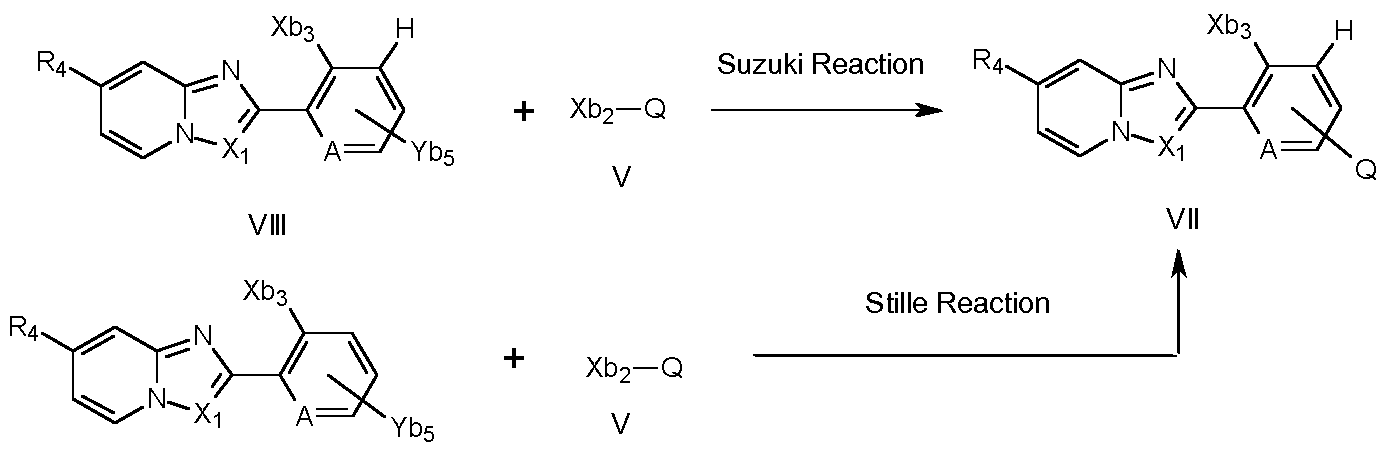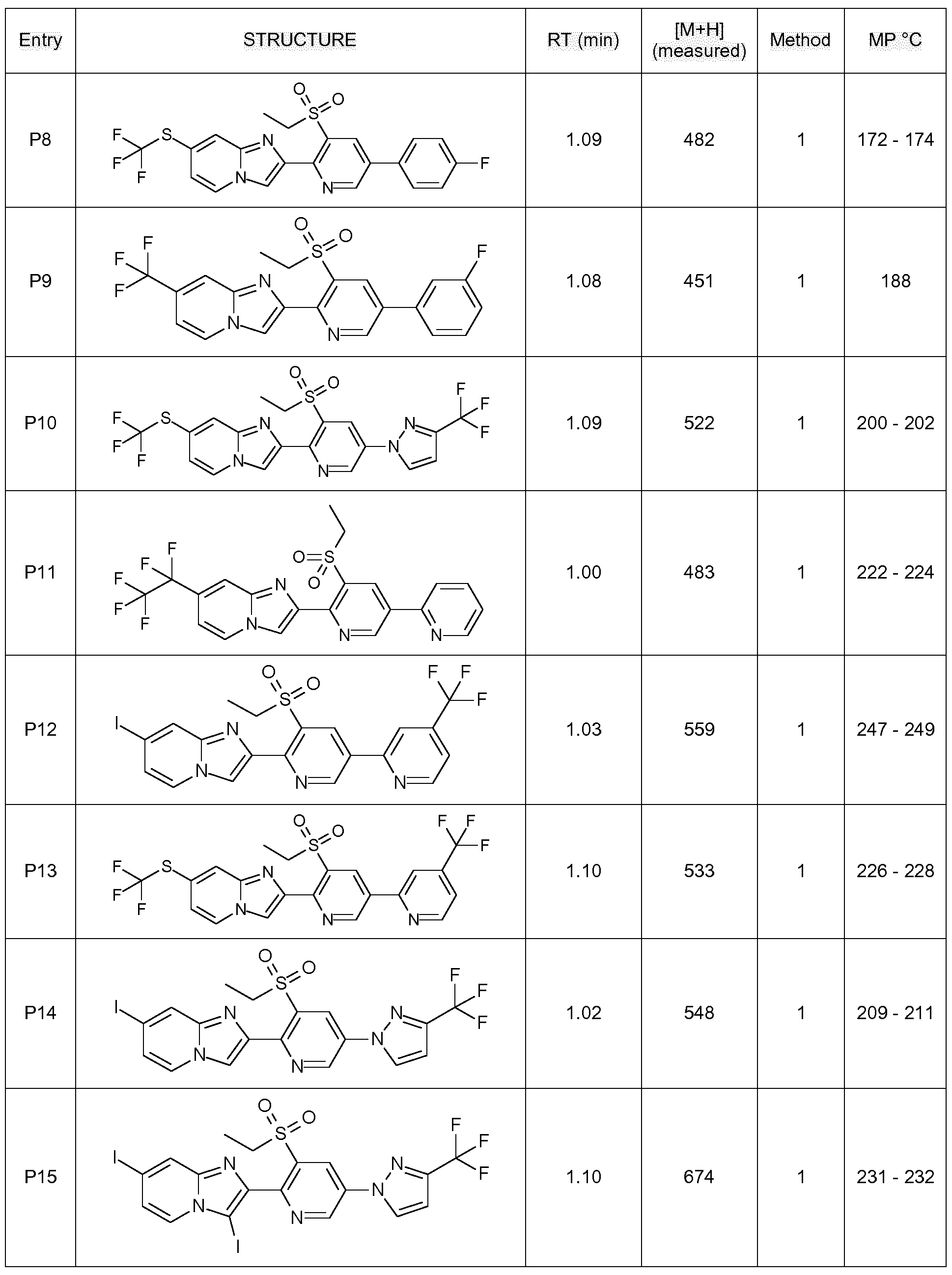WO2018095795A1 - Pesticidally active polycyclic derivatives with sulfur containing substituents - Google Patents
Pesticidally active polycyclic derivatives with sulfur containing substituents Download PDFInfo
- Publication number
- WO2018095795A1 WO2018095795A1 PCT/EP2017/079389 EP2017079389W WO2018095795A1 WO 2018095795 A1 WO2018095795 A1 WO 2018095795A1 EP 2017079389 W EP2017079389 W EP 2017079389W WO 2018095795 A1 WO2018095795 A1 WO 2018095795A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- spp
- formula
- compounds
- ccn
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC1(*)C=*C(c2c[n](ccc(*)c3)c3n2)=C(**)C=C1 Chemical compound CC1(*)C=*C(c2c[n](ccc(*)c3)c3n2)=C(**)C=C1 0.000 description 3
- ZCVCLTCAGVEJJX-UHFFFAOYSA-N CC(c1c(C)ccc(Cl)n1)=O Chemical compound CC(c1c(C)ccc(Cl)n1)=O ZCVCLTCAGVEJJX-UHFFFAOYSA-N 0.000 description 1
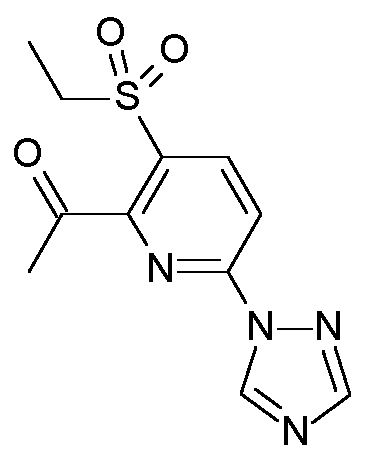
- GMQLHQTUKRNENK-UHFFFAOYSA-N CCS(c(cc1)c(C(C)=O)nc1-[n]1ncnc1)(=O)=O Chemical compound CCS(c(cc1)c(C(C)=O)nc1-[n]1ncnc1)(=O)=O GMQLHQTUKRNENK-UHFFFAOYSA-N 0.000 description 1
- BJLFUVJAUMROND-UHFFFAOYSA-N CCS(c1cc(-c2cc(C(F)(F)F)ccn2)cnc1C(C)=O)(=O)=O Chemical compound CCS(c1cc(-c2cc(C(F)(F)F)ccn2)cnc1C(C)=O)(=O)=O BJLFUVJAUMROND-UHFFFAOYSA-N 0.000 description 1
- BTEKHAKGAJLDAN-UHFFFAOYSA-N CCSc(cc1)c(C(Cl)=O)nc1Cl Chemical compound CCSc(cc1)c(C(Cl)=O)nc1Cl BTEKHAKGAJLDAN-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
Definitions
- the present invention relates to pesticidally active, in particular insecticidally active polycyclic derivatives containing sulfur substituents, to intermediates for the preparation of those compounds, to compositions comprising those compounds, and to their use for controlling animal pests (including arthropods and in particular insects or representatives of the order Acarina).
- Heterocyclic compounds with pesticidal action are known and described, for example, in
- the present invention accordingly relates to compounds of formula I,
- A is CH or N
- Q is phenyl which can be mono- or polysubstituted by substituents selected from the group consisting of halogen, cyano, Ci-C4alkyl, Ci-C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy, Ci-C4alkylsulfanyl, Ci- C4alkylsulfinyl, Ci-C4alkylsulfonyl, and Ci-C4haloalkylsulfanyl; or
- Q is pyridyl or pyrimidyl which can be mono- or polysubstituted by substituents selected from the group consisting of halogen, cyano, Ci-C4alkyl, Ci-C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci- C4haloalkylsulfanyl; or
- Q is pyrazolyl which is linked via a nitrogen atom to the ring which contains the substituent A, and which in turn can be substituted by halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl; or
- Q is triazolyl which is linked via a nitrogen atom to the ring which contains the substituent A, and which in turn can be substituted by halogen, cyano or Ci-C4haloalkyl; or
- X is S, SO or S0 2 ;
- R4 is halogen, Ci-C4haloalkyl, Ci-C4haloalkylsulfanyl, Ci-C4haloalkylsulfinyl, or Ci-C4haloalkylsulfonyl;
- R5 is Ci-C4alkyl or C3-C6cycloalkyl-Ci-C4alkyl;
- Xi is CR6, wherein R6 is hydrogen, Ci-C4alkyl or halogen;
- Compounds of formula I which have at least one basic centre can form, for example, acid addition salts, for example with strong inorganic acids such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, a phosphorus acid or a hydrohalic acid, with strong organic carboxylic acids, such as Ci-C4alkanecarboxylic acids which are unsubstituted or substituted, for example by halogen, for example acetic acid, such as saturated or unsaturated dicarboxylic acids, for example oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid or phthalic acid, such as hydroxycarboxylic acids, for example ascorbic acid, lactic acid, malic acid, tartaric acid or citric acid, or such as benzoic acid, or with organic sulfonic acids, such as Ci-C
- Compounds of formula I which have at least one acidic group can form, for example, salts with bases, for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, diethyl-, triethyl- or
- dimethylpropylamine or a mono-, di- or trihydroxy-lower-alkylamine, for example mono-, di- or triethanolamine.
- alkyl groups occurring in the definitions of the substituents can be straight-chain or branched and are, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, iso-butyl, tert-butyl, pentyl, hexyl, and their branched isomers.
- Alkylsulfanyl, alkylsulfinyl, alkylsulfonyl, alkoxy, alkenyl and alkynyl radicals are derived from the alkyl radicals mentioned.
- the alkenyl and alkynyl groups can be mono- or polyunsaturated.
- Ci-di-alkylamino is dimethylamino.
- Halogen is generally fluorine, chlorine, bromine or iodine. This also applies, correspondingly, to halogen in combination with other meanings, such as haloalkyl or halophenyl.
- HaloalkyI groups preferably have a chain length of from 1 to 6 carbon atoms.
- Haloalkyl is, for example, fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, 2,2,2- trifluoroethyl, 2-fluoroethyl, 2-chloroethyl, pentafluoroethyl, 1 , 1-difluoro-2,2,2-trichloroethyl, 2,2,3,3- tetrafluoroethyl and 2,2,2-trichloroethyl.
- Haloalkoxy groups preferably have a chain length of from 1 to 4 carbon atoms.
- Haloalkoxy is, for example, difluoromethoxy, trifluoromethoxy or 2,2,2-trifluoroethoxy.
- Haloalkylsulfanyl groups preferably have a chain length of from 1 to 4 carbon atoms.
- Haloalkylsulfanyl is, for example, difluoromethylsulfanyl, trifluoromethylsulfanyl or 2,2,2-trifluoroethylsulfanyl.
- Ci-C4haloalkylsulfinyl and Ci-C4haloalkylsulfonyl which may be, for example, trifluoromethylsulfinyl, trifluoromethylsulfonyl or 2,2,2-trifluoroethylsulfonyl.
- a triazolyl which is linked via a nitrogen atom to the ring which contains the substituent A is for example 1 ,2,4-triazol-1-yl, 1 ,2,4-triazol-4-yl, triazol-1-yl, or triazol-2-yl.
- Preferred is 1 ,2,4-triazol-1-yl.
- the cycloalkyl groups preferably have from 3 to 6 ring carbon atoms, for example cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
- mono- to polysubstituted in the definition of the substituents, means typically, depending on the chemical structure of the substituents, monosubstituted to three-times substituted, preferably monosubstituted to three-times substituted, more preferably mono-, or double- substituted.
- the compounds of formula I according to the invention also include hydrates which may be formed during the salt formation.
- a preferred group of compounds of formula I is represented by the compounds of formula 1-1
- F3 ⁇ 4, Rs, A and Xi are as defined under formula I above;
- X is S, SO or SO2; and
- Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci-
- C4haloalkyl Ci-C4haloalkoxy, Ci-C4alkoxy, Ci-C4alkylsulfanyl, Ci-C4alkylsulfinyl, Ci-C4alkylsulfonyl, and Ci-C4haloalkylsulfanyl.
- R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl
- X is preferably SO2
- Rs is preferably ethyl
- Xi is preferably CH .
- R4, Rs, and A are as defined under formula I above; and wherein Rx is independently selected from hydrogen or halogen.
- R4 is preferably Ci- C4haloalkyl and R5 is preferably ethyl.
- More highly preferred compounds of formula l-1a are those in which R4 is CF3, Rs is ethyl, A is nitrogen and Rx is hydrogen or halogen, preferably fluorine or chlorine, with the proviso that at least one Rx is halogen.
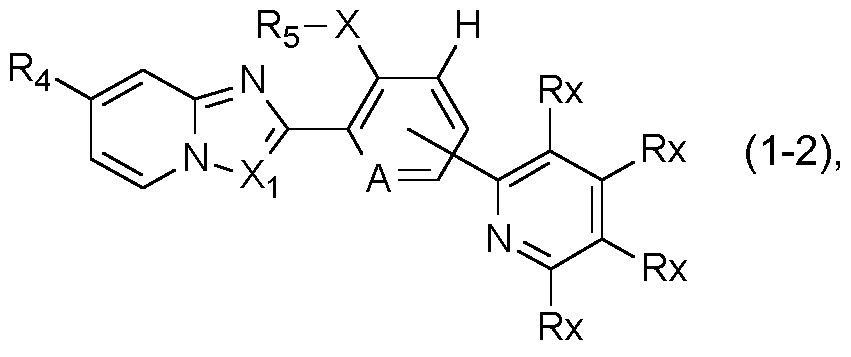
- a further preferred group of compounds of formula I is represented by the compounds of formula I-2:
- R4, Rs, A and Xi are as defined under formula I above; X is S, SO or SO2; and Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci-C4haloalkylsulfanyl.
- R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl
- X is preferably SO2
- Rs is preferably ethyl
- Xi is preferably CH.
- R4, Rs, and A are as defined under formula I above; and wherein Rx is independently hydrogen or halogen.
- R4 is preferably Ci- C4haloalkyl and Rs is preferably ethyl.
- More highly preferred compounds of formula 1-1 a are those in which R4 is CF3, Rs is ethyl A is nitrogen and Rx is hydrogen or halogen, preferably fluorine or chlorine, with the proviso that at least one Rx is halogen. Most preferably both Rx are halogen, in particular fluorine.
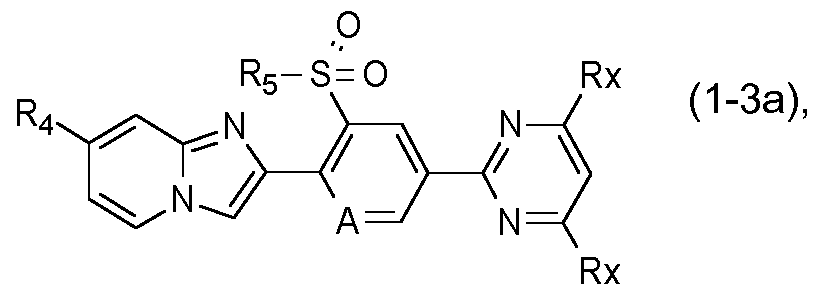
- a further preferred group of compounds of formula I is represented by the compounds of formula I-3:
- F3 ⁇ 4, Rs, A and Xi are as defined under formula I above;
- X is S, SO or SO2; and
- Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci-C4haloalkylsulfanyl.
- R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl
- X is preferably SO2
- Rs is preferably ethyl
- Xi is preferably CH.
- R4, Rs, and A are as defined under formula I above; and Rx is independently hydrogen or halogen.
- R4 is preferably Ci-C4haloalkyl and Rs is preferably ethyl, More highly preferred compounds of formula l-3a are those in which R4 is CF3, Rs is ethyl, A is nitrogen and Rx is hydrogen or halogen, preferably fluorine or chlorine, with the proviso that at least one Rx is halogen.
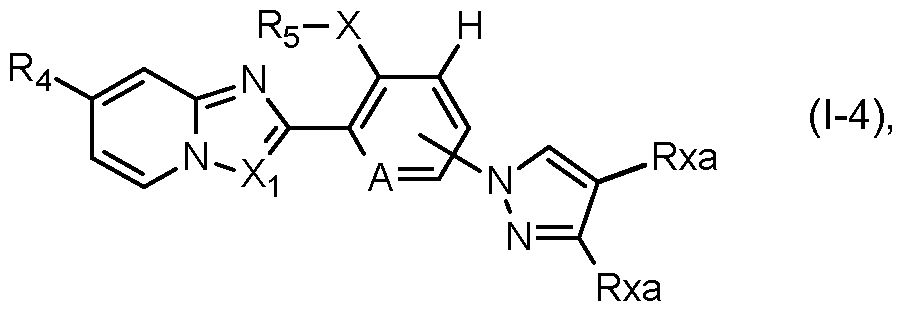
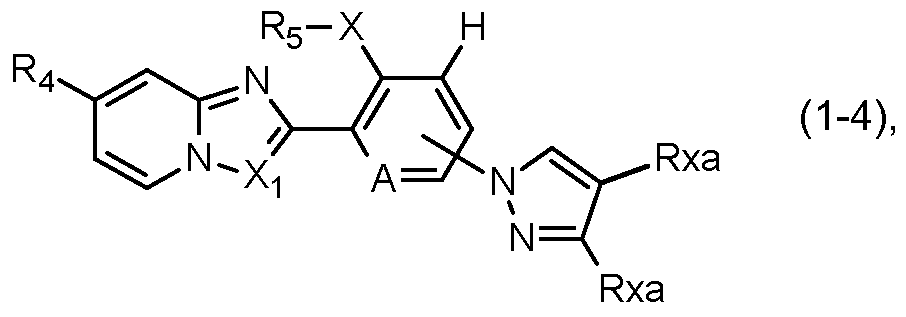
- a further preferred group of compounds of formula I is represented by the compounds of formula I-4:
- R4, Rs, A and Xi are as defined under formula I above; and wherein X is S, SO or SO2; and Rxa is hydrogen, halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl. ln this preferred group of compounds of formula I-4, F3 ⁇ 4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl, X is preferably SO2, Rs is preferably ethyl and Xi is preferably CH.
- R4, Rs, and A are as defined under formula I above; and wherein Rxa is hydrogen, cyano, or Ci-C4haloalkyl.
- R4 is preferably Ci-C4haloalkyl and Rs is preferably ethyl.
- More highly preferred compounds of formula l-4a are those in which R4 is CF3, Rs is ethyl, A is nitrogen and Rxa is hydrogen, cyano, or Ci-C4haloalkyl, preferably CF3 or cyano.
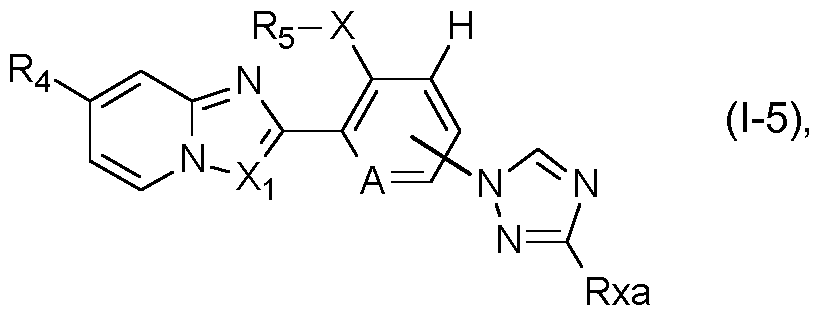
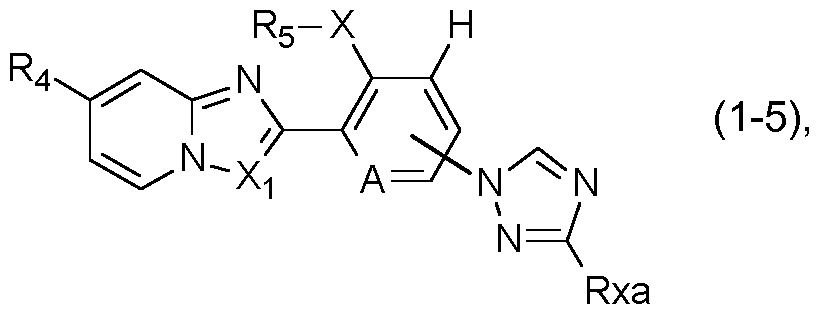
- a further preferred group of compounds of formula I is represented by the compounds of formula I-5:
- R4, Rs, A and Xi are as defined under formula I above; and wherein X is S, SO or SO2; and Rxa is hydrogen, halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl.
- R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl
- X is preferably SO2
- Rs is preferably ethyl
- Xi is preferably CH.
- R4, Rs, and A are as defined under formula I above; and wherein Rxa is hydrogen, cyano, or halogen.
- R4 is preferably Ci-C4haloalkyl and Rs is preferably ethyl.
- the process according to the invention for preparing compounds of formula I is carried out by methods known to those skilled in the art.
- the reaction can be catalyzed by a palladium based catalyst, for example fefra/c/ ' s(triphenylphosphine)-palladium or (1 ,1 'bis(diphenylphosphino)-ferrocene)dichloropalladium-dichloromethane (1 : 1 complex), in presence of a base, like sodium carbonate or cesium fluoride, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water or of dioxane and water, preferably under inert atmosphere.
- the reaction temperature can preferentially range from ambient temperature to the boiling point of the reaction mixture.
- Suzuki reactions are well known to those skilled in the art and have been reviewed, for example J.Orgmet. Chem. 576, 1999, 147-168.
- compounds of formula I can be prepared by a Stille reaction of compounds of formula 1Mb wherein Yb2 is a trialkyl tin derivative, preferably tri-n-butyl tin, with compounds of formula II.
- Such Stille reactions are usually carried out in the presence of a palladium catalyst, for example
- Compounds of formula I can also be prepared (as depicted in scheme 3) by a Suzuki reaction as described above, which involves reacting compounds of formula IV with compounds of formula V, wherein Xb2 can be a halogen, preferentially chlorine, bromine or iodine, or a sulfonate, like for example a trifluoromethanesulfonate and Yb3 can be a boron-derived functional group, as for example B(OH)2 or B(ORb2)2 wherein Rb2 can be a C1-C4alkyl group or the two groups ORb2 can form together with the boron atom a five membered ring, as for example a pinacol boronic ester.
- Xb2 can be a halogen, preferentially chlorine, bromine or iodine, or a sulfonate, like for example a trifluoromethanesulfonate
- Yb3 can be a boron
- A, Xi , R4, Rs, and X are as described in formula I.
- the reaction can be catalyzed by a palladium based catalyst, for example tetrakis(triphenylphosphine)-palladium, in presence of a base, like sodium carbonate, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water, preferably under inert atmosphere.
- the reaction temperature can preferentially range ambient temperature to the boiling point of the reaction mixture.
- compounds of formula I can be prepared by a Stille coupling (Scheme 3) of compounds of formula V with compounds of formula VI, wherein F3 ⁇ 4, Rs, Xi , A, X are as described above, and Yb4 is a trialkyl tin derivative, preferably tri-n-butyl tin, under conditions described as in scheme 1.
- Compounds of formula l-b1 may be prepared (scheme 4) by reacting a compound of the formula VII with a compound of the formula Villa, wherein A, R4, Rs, and Xi have the values defined in formula I and X is sulfur and M is a metal or non-metal cation.
- the cation M is assumed to be monovalent, but polyvalent cations associated with more than one S-Rs group can also be considered. Preferred cations are, for example lithium, sodium, potassium or cesium.
- Xb3 is a leaving group like, for example, fluorine, chlorine, bromine or iodine, or an aryl- or alkylsulfonate, but many other leaving groups could be considered (for example NO2).
- the reaction can be performed in a solvent, preferably aprotic, at temperatures below 0°C or up to boiling temperature of the reaction mixture.
- Compounds of formula VI I wherein Xb3 is a leaving group like, for example, fluorine, chlorine, bromine iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate, or any other similar leaving group, can be prepared (scheme 5) by reacting compounds of formula V with compounds of formula VI I I, wherein Xb3 can be a halogen, preferentially chlorine, bromine or iodine, or a sulfonate, like for example a trifluoromethanesulfonate, most preferably bromine or iodine and Yb5 can be a boron- derived functional group, as for example B(OH)2 or B(ORb4)2 wherein Rb4 can be a Ci-C4alkyl group or the two groups ORb4 can form together with the boron atom a five membered ring, as for example a pinacol boronic ester.
- reaction can be catalyzed by a palladium based catalyst, for example tetrakis(triphenylphosphine)- palladium, in presence of a base, like sodium carbonate, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water, preferably under inert atmosphere.
- a palladium based catalyst for example tetrakis(triphenylphosphine)- palladium
- a base like sodium carbonate
- solvent or a solvent mixture like, for example a mixture of 1 ,2-dimethoxyethane and water, preferably under inert atmosphere.
- the reaction temperature can preferentially range from ambient temperature to the boiling point of the reaction mixture.
- compounds of formula VII can be prepared from compounds of formula X, wherein A, Xi , R4 and Xb3 are as previously defined, and Yb6 is a trialkyl tin derivative, preferably tri- n-butyl tin, with compounds of formula V, under conditions described for those described for the chemistry illustrated in scheme 1 .
- compounds of formula VII can also be prepared by reacting compounds of formula X, wherein Xb3 and Xb4 are leaving groups, for example, fluorine, chlorine, bromine or iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate, or any other similar leaving group, with compounds of formula Ilia (Suzuki reaction) or Illb (Stille reaction).
- the chemistry is carried out analogously to that discussed for scheme 1.
- a further route to prepare compounds of formula II, respectively Il-c1 involves reaction of compounds of formula X with compounds of formula Villa as shown in scheme 7.
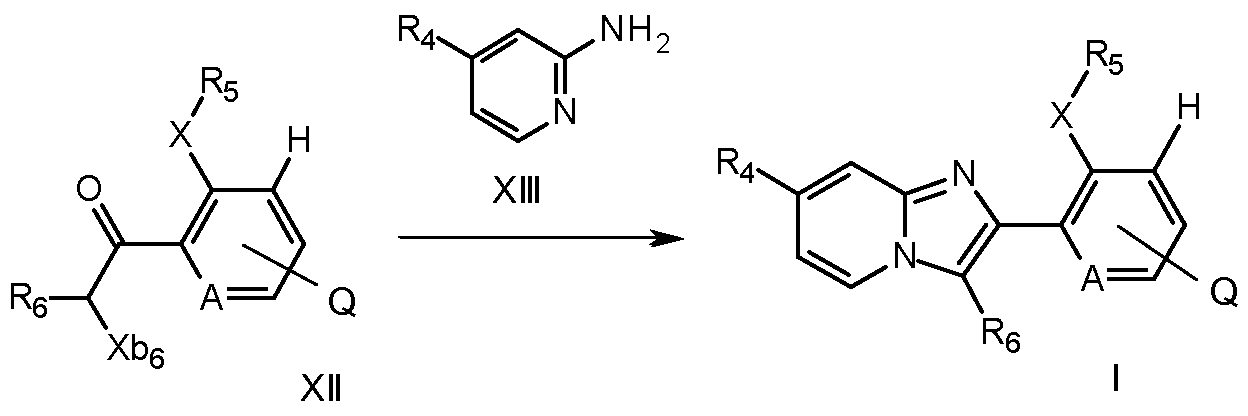
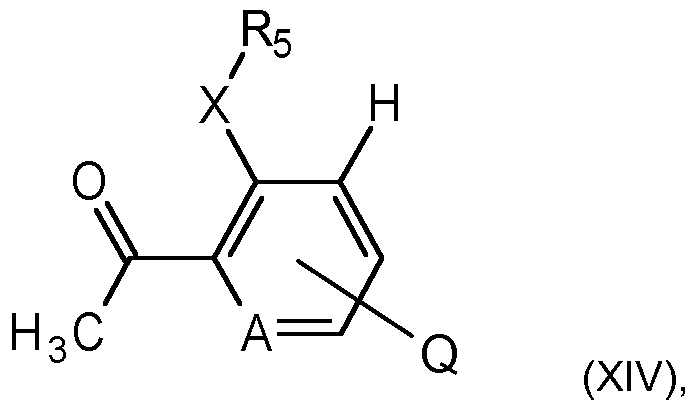
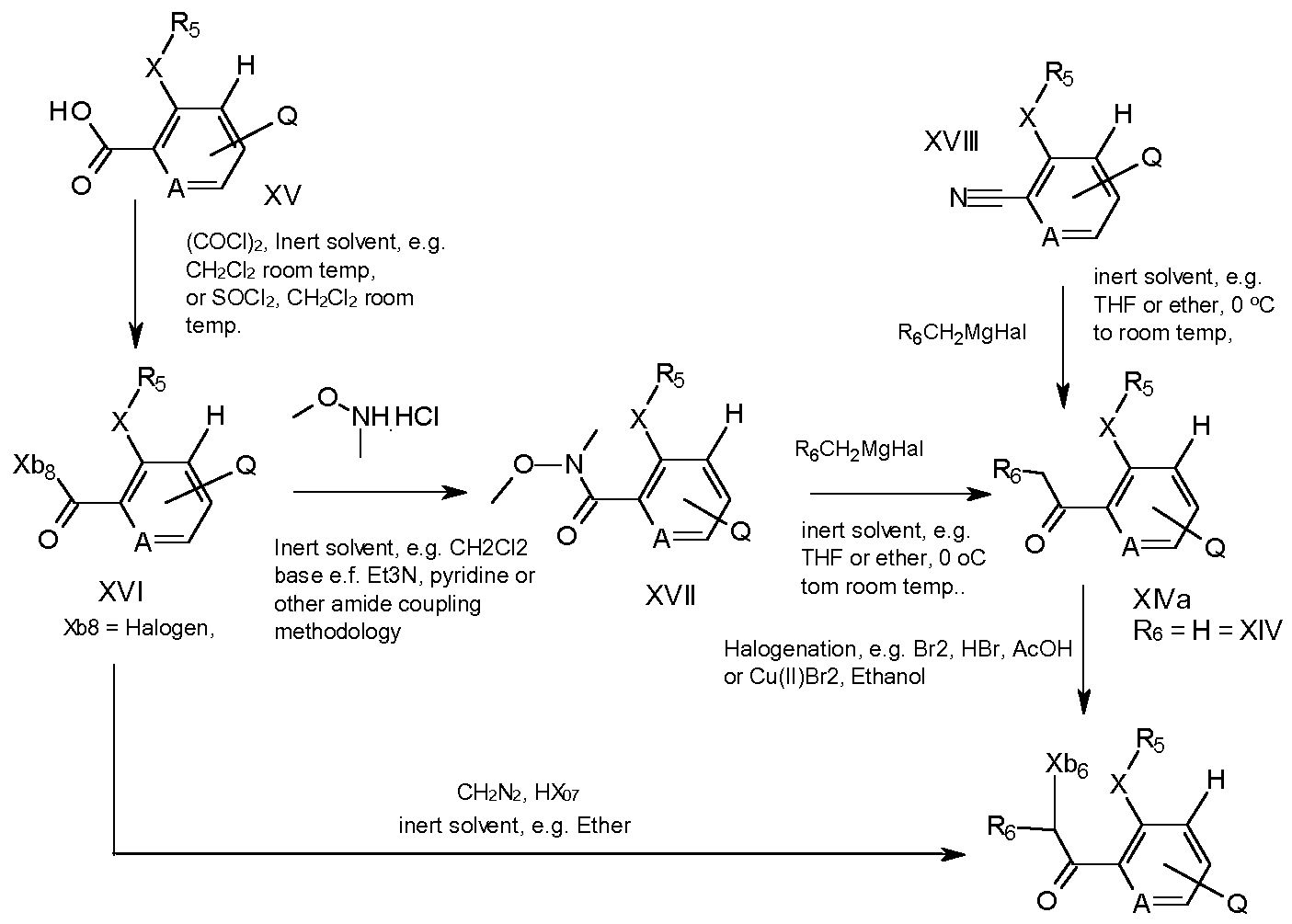
- a further process to prepare compounds of formula I involves reacting a compound of formula XIII with a compound of formula XIV
- the reaction is usually carried out in the presence of a base, for example potassium carbonate, cesium carbonate, or potassium phosphate, in an inert solvent, such as dioxane, optionally in the presence of water, with a palladium(O) catalyst, for example
- a base for example potassium carbonate, cesium carbonate, or potassium phosphate
- an inert solvent such as dioxane
- a palladium(O) catalyst for example
- Compounds of formula XlVa and XIV can be halogenated to compounds of formula XII, with for example mixtures of bromine and hydrobromic acid in acetic acid (as described in Phosphorus, Sulfur and Silicon and the Related Elements, 2013, 188(12), 1835-1844) or with, for example,
- compounds of formula XII where R6 is hydrogen can be prepared directly from compounds of formula XVI by treatment with diazomethane or trimethyl silyl diazomethane and subsequent treatment with an halogen acid, for example, hydrobromic acid or hydrochloric acid in an inert solvent such as diethyl ether.
- an halogen acid for example, hydrobromic acid or hydrochloric acid in an inert solvent such as diethyl ether.
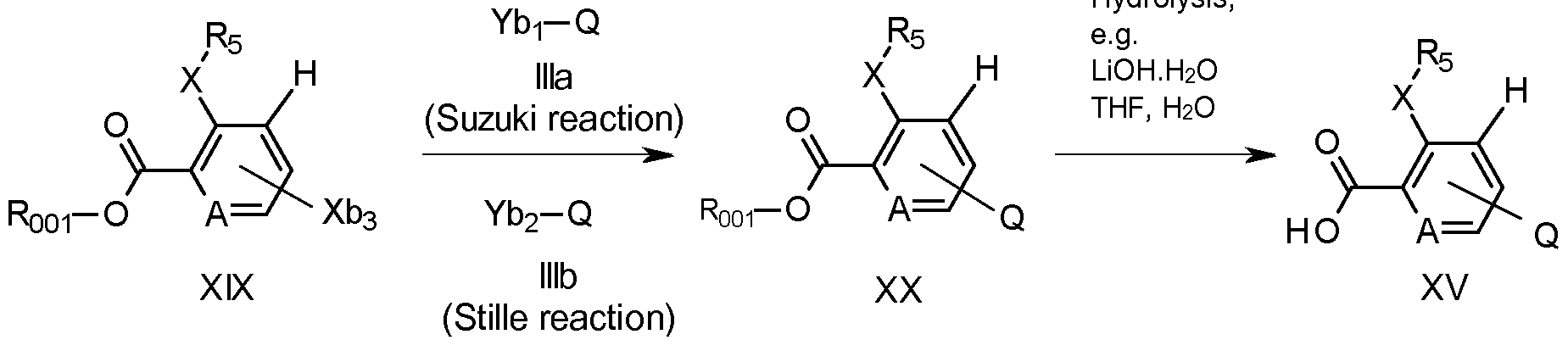
- Compounds of formula XV can be prepared (as shown in scheme 1 1 ) by ester hydrolysis of compounds of formula XX, wherein A, Xb3, and Rs are as previously defined, and R001 is Ci-C6alkyl, by methods known to those skilled in the art, for example by treatment with an alkaline earth metal base, such as lithium hydroxide, typically in water with sufficient miscible organic solvent, for example THF or acetone, to dissolve compounds of the formula XX.
- an alkaline earth metal base such as lithium hydroxide
- Compounds XX can be prepared by a Suzuki reaction, which involves for example, reacting compounds of formula XIX, wherein Xb3 is a leaving group like, for example, chlorine, bromine or iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate (especially preferred are those in which Xb1 is fluoro or bromo) with compounds of formula Ilia, wherein Ybi can be a boron-derived functional group, as for example B(OH)2 or B(ORbi)2 wherein Rbi can be a Ci-C4alkyl group or the two groups ORbi can form together with the boron atom a five membered ring, as for example a pinacol boronic ester.
- Xb3 is a leaving group like, for example, chlorine, bromine or iodine
- an aryl- or alkylsulfonate such as trifluoromethane
- the reaction can be catalyzed by a palladium based catalyst, for example fefra/c/ ' s(triphenylphosphine)-palladium or (1 , 1 'bis(diphenylphosphino)-ferrocene)dichloropalladium-dichloromethane (1 : 1 complex), in presence of a base, like sodium carbonate or cesium fluoride, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water or of dioxane and water, preferably under inert atmosphere.
- the reaction temperature can preferentially range from ambient temperature to the boiling point of the reaction mixture.
- Suzuki reactions are well known to those skilled in the art and have been reviewed, for example J.Orgmet. Chem. 576, 1999, 147-168. Scheme 1 1
- compounds of formula XX can be prepared by a Stille reaction of compounds of formula lllb wherein Yb2 is a trialkyl tin derivative, preferably tri-n-butyl tin, with compounds of formula XIX.
- Such Stille reactions are usually carried out in the presence of a palladium catalyst, for example ieira/f/s(triphenylphosphine)palladium(0), or (1 ,1 'bis(diphenylphosphino)-ferrocene)dichloropalladium- dichloromethane (1 : 1 complex), in an inert solvent such as DMF, acetonitrile, or dioxane, optionally in the presence of an additive, such as cesium fluoride, or lithium chloride, and optionally in the presence of a further catalyst, for example copper(l)iodide.
- a palladium catalyst for example ieira/f/s(triphenylphosphine)palladium(0), or (1 ,1 'bis(diphenylphosphino)-ferrocene)dichloropalladium- dichloromethane (1 : 1 complex
- an inert solvent such as DMF, acetonitrile, or dioxan
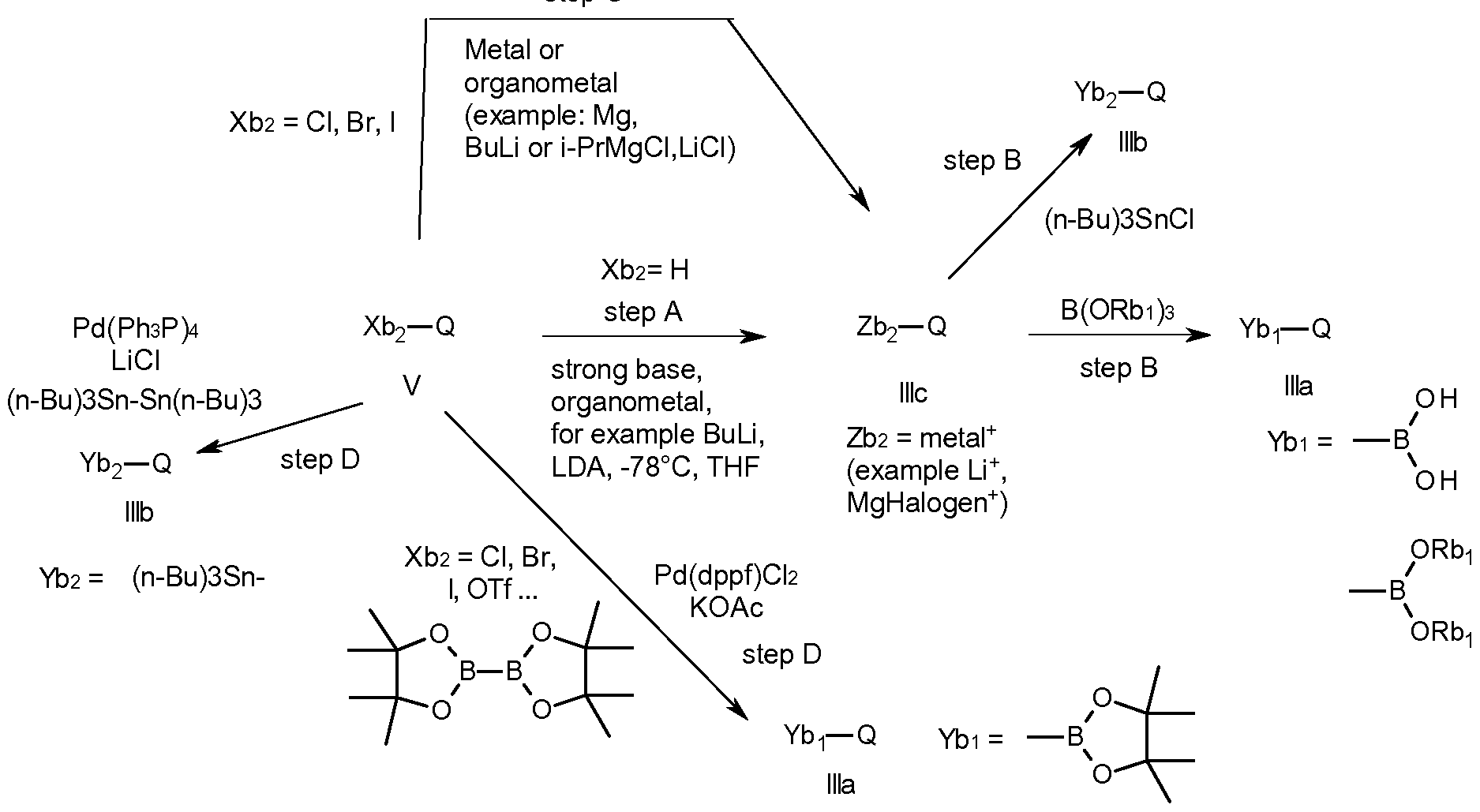
- a large number of compounds of the formula V are commercially available or can be prepared by those skilled in the art. Many chemical transformations, well known by those skilled in the art, can be used to access boronic acid derivatives of formula Ilia, starting from various and easily available starting materials, as for example, to cite only a few (scheme 14), hydrogen abstraction on a heteroaromatic compound of the formula V wherein Xb2 is hydrogen, with a strong base (step A), like butyl lithium or lithium diisopropylamide or (i-PrMgCI.
- LiCI Li + or MgCI + for example
- Zb ⁇ is a metal such as Li + or MgCI + for example
- a trialkylborate step B
- a tri-n-butyl tin chloride step B
- Another way to access an organometal intermediate of the formulae Ilia or lllb is from a compound of the formula 111 which is obtained by via metal-halogen exchange of compound of formula V with an organometallic species (step C), using for example butyl lithium or an organ magnesium compound, or direct metalation with a metal, like magnesium.
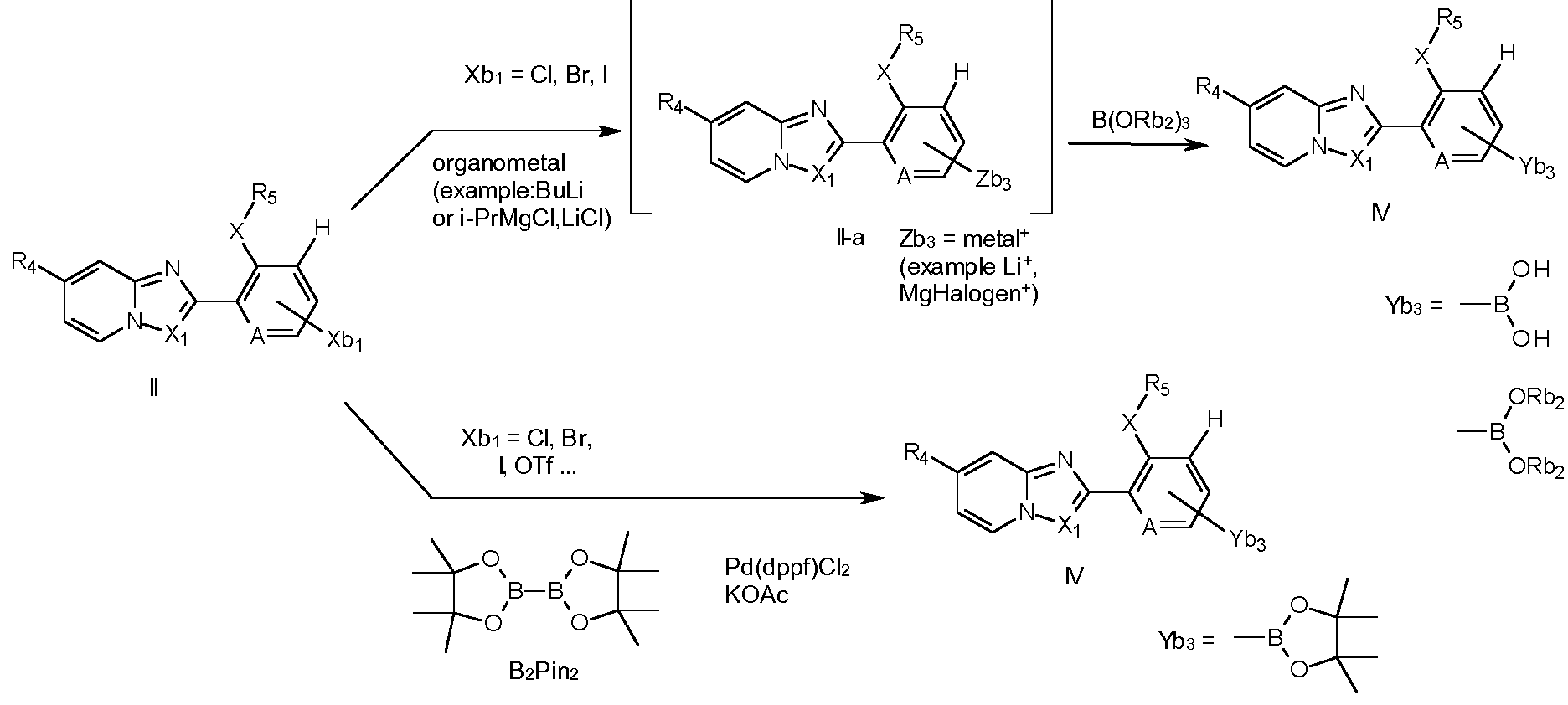
- Compounds of formula IV wherein A, X, Xi, F3 ⁇ 4 and R5 are as described in formula I, can be prepared from compounds of formula II (scheme 15), wherein A, X, Xi, R4 and R5 are as described in formula I.
- compounds of formula II, wherein Xbi is chlorine, bromine or iodine can be treated with an organometallic species like, for example, butyl lithium or an organomagnesium compound, to generate an intermediate compound of the formula ll-a, wherein Zb3 is as defined in the scheme, via metal- halogen exchange.
- This reaction is preferentially performed in an anhydrous aprotic solvent, such as THF, at low temperature (between -120°C and 0°C), preferentially between -1 10°C and -60°C).
- an anhydrous aprotic solvent such as THF
- the intermediate organometal compound of formula ll-a is preferably directly converted into compound of formula IV by reaction with a boronate compound B(ORb2)3, wherein Rb2 is a C1-C4alkyl group.
- the boronic acid IV wherein Yb3 is -B(OH)2, or a dialkylboronate IV, wherein Yb3 is -B(ORb2)2, can be formed.
- R4 is Ci-C2haloalkyl or Ci-Chaloalkylsulfanyl, i.e. compounds of formula lac and lad respectively, can be prepared from compounds of formula lab, wherein in R4 is halogen, preferably bromine or iodine, by the same chemistry described for the preparation of XII lb, XI lie and XI I If . This is illustrated in scheme 18.
- the reaction can be performed with reagents like, for example, a peracid such as peracetic acid or m-chloroperbenzoic acid, or a hydroperoxide, such as for example, hydrogen peroxide or tert- butylhydroperoxide, or an inorganic oxidant, like a monoperoxo-disulfate salt or potassium permanganate.
- a peracid such as peracetic acid or m-chloroperbenzoic acid
- a hydroperoxide such as for example, hydrogen peroxide or tert- butylhydroperoxide
- an inorganic oxidant like a monoperoxo-disulfate salt or potassium permanganate.
- the reactants can be reacted in the presence of a base.
- suitable bases are alkali metal or alkaline earth metal hydroxides, alkali metal or alkaline earth metal hydrides, alkali metal or alkaline earth metal amides, alkali metal or alkaline earth metal alkoxides, alkali metal or alkaline earth metal acetates, alkali metal or alkaline earth metal carbonates, alkali metal or alkaline earth metal dialkylamides or alkali metal or alkaline earth metal alkylsilylamides, alkylamines, alkylenediamines, free or N-alkylated saturated or unsaturated cycloalkylamines, basic heterocycles, ammonium hydroxides and carbocyclic amines.
- sodium hydroxide sodium hydride, sodium amide, sodium methoxide, sodium acetate, sodium carbonate, potassium tert- butoxide, potassium hydroxide, potassium carbonate, potassium hydride, lithium diisopropylamide, potassium bis(trimethylsilyl)amide, calcium hydride, triethylamine, diisopropylethylamine,
- the reactants can be reacted with each other as such, i.e. without adding a solvent or diluent. In most cases, however, it is advantageous to add an inert solvent or diluent or a mixture of these. If the reaction is carried out in the presence of a base, bases which are employed in excess, such as triethylamine, pyridine, N-methylmorpholine or ⁇ , ⁇ -diethylaniline, may also act as solvents or diluents.
- the reaction is advantageously carried out in a temperature range from approximately -80°C to approximately +140°C, preferably from approximately -30°C to approximately +100°C, in many cases in the range between ambient temperature and approximately +80°C.
- a compound of formula I can be converted in a manner known per se into another compound of formula I by replacing one or more substituents of the starting compound of formula I in the customary manner by (an)other substituent(s) according to the invention.
- Salts of compounds of formula I can be prepared in a manner known per se.
- acid addition salts of compounds of formula I are obtained by treatment with a suitable acid or a suitable ion exchanger reagent and salts with bases are obtained by treatment with a suitable base or with a suitable ion exchanger reagent.
- Salts of compounds of formula I can be converted in the customary manner into the free compounds I, acid addition salts, for example, by treatment with a suitable basic compound or with a suitable ion exchanger reagent and salts with bases, for example, by treatment with a suitable acid or with a suitable ion exchanger reagent.
- Salts of compounds of formula I can be converted in a manner known per se into other salts of compounds of formula I, acid addition salts, for example, into other acid addition salts, for example by treatment of a salt of inorganic acid such as hydrochloride with a suitable metal salt such as a sodium, barium or silver salt, of an acid, for example with silver acetate, in a suitable solvent in which an inorganic salt which forms, for example silver chloride, is insoluble and thus precipitates from the reaction mixture.
- a salt of inorganic acid such as hydrochloride
- a suitable metal salt such as a sodium, barium or silver salt
- the compounds of formula I which have salt- forming properties, can be obtained in free form or in the form of salts.
- the compounds of formula I and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can be present in the form of one of the isomers which are possible or as a mixture of these, for example in the form of pure isomers, such as antipodes and/or diastereomers, or as isomer mixtures, such as enantiomer mixtures, for example racemates, diastereomer mixtures or racemate mixtures, depending on the number, absolute and relative configuration of asymmetric carbon atoms which occur in the molecule and/or depending on the configuration of non-aromatic double bonds which occur in the molecule; the invention relates to the pure isomers and also to all isomer mixtures which are possible and is to be understood in each case in this sense hereinabove and hereinbelow, even when stereochemical details are not mentioned specifically in each case.
- Diastereomer mixtures or racemate mixtures of compounds of formula I, in free form or in salt form, which can be obtained depending on which starting materials and procedures have been chosen can be separated in a known manner into the pure diasteromers or racemates on the basis of the physicochemical differences of the components, for example by fractional crystallization, distillation and/or chromatography.
- Enantiomer mixtures, such as racemates, which can be obtained in a similar manner can be resolved into the optical antipodes by known methods, for example by recrystallization from an optically active solvent, by chromatography on chiral adsorbents, for example high-performance liquid
- N-oxides can be prepared by reacting a compound of the formula I with a suitable oxidizing agent, for example the hbCh/urea adduct in the presence of an acid anhydride, e.g. trifluoroacetic anhydride.
- a suitable oxidizing agent for example the hbCh/urea adduct
- an acid anhydride e.g. trifluoroacetic anhydride.
- the compounds of formula I and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form.
- the compounds of formula I according to the invention are preventively and/or curatively valuable active ingredients in the field of pest control, even at low rates of application, which have a very favourable biocidal spectrum and are well tolerated by warm-blooded species, fish and plants.
- the active ingredients according to the invention act against all or individual developmental stages of normally sensitive, but also resistant, animal pests, such as insects or representatives of the order Acarina.
- insecticidal or acaricidal activity of the active ingredients according to the invention can manifest itself directly, i. e. in destruction of the pests, which takes place either immediately or only after some time has elapsed, for example during ecdysis, or indirectly, for example in a reduced oviposition and/or hatching rate.
- animal pests are:
- Hyalomma spp. Ixodes spp., Olygonychus spp, Ornithodoros spp., Polyphagotarsone latus,
- Panonychus spp. Phyllocoptruta oleivora, Phytonemus spp, Polyphagotarsonemus spp, Psoroptes spp., Rhipicephalus spp., Rhizoglyphus spp., Sarcoptes spp., Steneotarsonemus spp, Tarsonemus spp. and Tetranychus spp.;
- Haematopinus spp. Linognathus spp., Pediculus spp., Pemphigus spp. and Phylloxera spp.;
- Agriotes spp. Amphimallon majale, Anomala orientalis, Anthonomus spp., Aphodius spp, Astylus atromaculatus, Ataenius spp, Atomaria linearis, Chaetocnema tibialis, Cerotoma spp, Conoderus spp, Cosmopolites spp., Cotinis nitida, Curculio spp., Cyclocephala spp, Dermestes spp., Diabrotica spp., Diloboderus abderus, Epilachna spp., Eremnus spp., Heteronychus arator, Hypothenemus hampei, Lagria vilosa, Leptinotarsa decemLineata, Lissorhoptrus spp., Liogenys spp, Maecolaspis spp, Maladera castanea, Megas
- Acyrthosium pisum Adalges spp, Agalliana ensigera, Agonoscena targionii, Aleurodicus spp, Aleurocanthus spp, Aleurolobus barodensis, Aleurothrixus floccosus, Aleyrodes brassicae, Amarasca biguttula, Amritodus atkinsoni, Aonidiella spp., Aphididae, Aphis spp., Aspidiotus spp., Aulacorthum solani, Bactericera cockerelli, Bemisia spp, Brachycaudus spp, Brevicoryne brassicae, Cacopsylla spp, Cavariella aegopodii Scop., Ceroplaster spp., Chrysomphalus aonidium, Chrysomphalus dictyospermi, Cicadella spp, Cofana spec
- Macrosiphum spp. Mahanarva spp, Metcalfa pruinosa, Metopolophium dirhodum, Myndus crudus, Myzus spp., Neotoxoptera sp, Nephotettix spp., Nilaparvata spp., Nippolachnus piri Mats, Odonaspis ruthae, Oregma lanigera Zehnter, Parabemisia myricae, Paratrioza cockerelli, Parlatoria spp., Pemphigus spp., Peregrinus maidis, Perkinsiella spp, Phorodon humuli, Phylloxera spp, Planococcus spp., Pseudaulacaspis spp., Pseudococcus spp., Pseudatomoscelis seriatus, Psylla spp., Pulvinaria
- Coptotermes spp Corniternes cumulans, Incisitermes spp, Macrotermes spp, Mastotermes spp, Microtermes spp, Reticulitermes spp.; Solenopsis geminate
- Blatta spp. Blattella spp., Gryllotalpa spp., Leucophaea maderae, Locusta spp., Neocurtilla hexadactyla, Periplaneta spp. , Scapteriscus spp, and Schistocerca spp.;
- Thysanura for example, Lepisma saccharina.
- the active ingredients according to the invention can be used for controlling, i. e. containing or destroying, pests of the abovementioned type which occur in particular on plants, especially on useful plants and ornamentals in agriculture, in horticulture and in forests, or on organs, such as fruits, flowers, foliage, stalks, tubers or roots, of such plants, and in some cases even plant organs which are formed at a later point in time remain protected against these pests.
- Suitable target crops are, in particular, cereals, such as wheat, barley, rye, oats, rice, maize or sorghum; beet, such as sugar or fodder beet; fruit, for example pomaceous fruit, stone fruit or soft fruit, such as apples, pears, plums, peaches, almonds, cherries or berries, for example strawberries, raspberries or blackberries; leguminous crops, such as beans, lentils, peas or soya; oil crops, such as oilseed rape, mustard, poppies, olives, sunflowers, coconut, castor, cocoa or ground nuts; cucurbits, such as pumpkins, cucumbers or melons; fibre plants, such as cotton, flax, hemp or jute; citrus fruit, such as oranges, lemons, grapefruit or tangerines; vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes or bell peppers; Lauraceae, such as avocado, Cinnamonium or camphor; and also tobacco, nuts,
- the invention may be used on any of the following ornamental species: Ageratum spp., Alonsoa spp., Anemone spp., Anisodontea capsenisis, Anthemis spp., Antirrhinum spp., Aster spp., Begonia spp. (e.g. B. elatior, B. semperflorens, B. tubereux), Bougainvillea spp., Brachycome spp., Brassica spp.
- Ageratum spp. Ageratum spp., Alonsoa spp., Anemone spp., Anisodontea capsenisis, Anthemis spp., Antirrhinum spp., Aster spp., Begonia spp. (e.g. B. elatior, B. semperflorens, B. tubereux), Bougainvillea spp., Brachycome spp.
- Calceolaria spp. (ornamental), Calceolaria spp., Capsicum annuum, Catharanthus roseus, Canna spp., Centaurea spp., Chrysanthemum spp., Cineraria spp. (C. maritime), Coreopsis spp., Crassula coccinea, Cuphea ignea, Dahlia spp., Delphinium spp., Dicentra spectabilis, Dorotheantus spp., Eustoma grandiflorum, Forsythia spp., Fuchsia spp., Geranium gnaphalium, Gerbera spp.,
- Gomphrena globosa Heliotropium spp., Helianthus spp., Hibiscus spp., Hortensia spp., Hydrangea spp., Hypoestes phyllostachya, Impatiens spp. (/. Walleriana), Iresines spp., Kalanchoe spp., Lantana camara, Lavatera trimestris, Leonotis leonurus, Lilium spp., Mesembryanthemum spp., Mimulus spp., Monarda spp., Nemesia spp., Tagetes spp., Dianthus spp.
- Plecthranthus spp. Poinsettia spp., Parthenocissus spp. CP- quinquefolia, P. tricuspidata), Primula spp., Ranunculus spp., Rhododendron spp., Rosa spp. (rose), Rudbeckia spp., Saintpaulia spp., Sa/w ' a spp., Scaevola aemola, Schizanthus wisetonensis, Sedum spp., Solanum spp., Surfinia spp., Tagetes spp., Nicotinia spp., Verbena spp., Zinnia spp. and other bedding plants.
- the invention may be used on any of the following vegetable species: Allium spp. (A sativum, A., cepa, A. oschaninii, A. Porrum, A. ascalonicum, A. fistulosum), Anthriscus cerefolium, Apium graveolus, Asparagus officinalis, Beta vulgarus, Brassica spp. (B. Oleracea, B. Pekinensis, B. rapa), Capsicum annuum, Cicer arietinum, Cichorium endivia, Cichorum spp. (C. intybus, C. endivia), Citrillus lanatus, Cucumis spp. (C. sativus, C.
- Preferred ornamental species include African violet, Begonia, Dahlia, Gerbera, Hydrangea, Verbena, Rosa, Kalanchoe, Poinsettia, Aster, Centaurea, Coreopsis, Delphinium, Monarda, Phlox, Rudbeckia, Sedum, Petunia, Viola, Impatiens, Geranium, Chrysanthemum, Ranunculus, Fuchsia, Salvia, Hortensia, rosemary, sage, St. Johnswort, mint, sweet pepper, tomato and cucumber.
- the active ingredients according to the invention are especially suitable for controlling Aphis craccivora, Diabrotica balteata, Heliothis virescens, Myzus persicae, Plutella xylostella and
- the active ingredients according to the invention are further especially suitable for controlling Mamestra (preferably in vegetables), Cydia pomonella (preferably in apples), Empoasca(preferably in vegetables, vineyards), Leptinotarsa (preferably in potatos) and Chilo supressalis (preferably in rice).
- the invention may also relate to a method of controlling damage to plant and parts thereof by plant parasitic nematodes (Endoparasitic-, Semiendoparasitic- and Ectoparasitic nematodes), especially plant parasitic nematodes such as root knot nematodes, Meloidogyne hapla, Meloidogyne incognita, Meloidogyne javanica, Meloidogyne arenaria and other Meloidogyne species; cyst-forming nematodes, Globodera rostochiensis and other Globodera species; Heterodera avenae, Heterodera glycines, Heterodera schachtii, Heterodera trifolii, and other Heterodera species; Seed gall nematodes, Anguina species; Stem and foliar nematodes, Aphelenchoides species; Sting nematodes, Belonolai
- Needle nematodes Longidorus elongatus and other Longidorus species; Pin nematodes,
- Pratylenchus species Lesion nematodes, Pratylenchus neglectus, Pratylenchus penetrans,
- the compounds of the invention may also have activity against the molluscs.
- examples of which include, for example, Ampullariidae; Arion (A. ater, A. circumscriptus, A. hortensis, A. rufus);
- Bradybaenidae (Bradybaena fruticum); Cepaea (C. hortensis, C. Nemoralis); ochlodina; Deroceras (D. agrestis, D. empiricorum, D. laeve, D. reticulatum); Discus (D. rotundatus); Euomphalia; Galba (G. trunculata); Helicelia (H. itala, H. obvia); Helicidae Helicigona arbustorum); Helicodiscus; Helix (H. aperta); Limax (L. cinereoniger, L. flavus, L. marginatus, L. maximus, L. tenellus); Lymnaea; Milax (M. gagates, M. marginatus, M. sowerbyi); Opeas; Pomacea (P. canaticulata); Vallonia and Zanitoides.
- crops is to be understood as including also crop plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria, especially those of the genus Bacillus.
- Toxins that can be expressed by such transgenic plants include, for example, insecticidal proteins, for example insecticidal proteins from Bacillus cereus or Bacillus popilliae; or insecticidal proteins from Bacillus thuringiensis, such as ⁇ -endotoxins, e.g. CrylAb, CrylAc, Cryl F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), e.g. Vip1 , Vip2, Vip3 or Vip3A; or insecticidal proteins of bacteria colonising nematodes, for example Photorhabdus spp. or
- Xenorhabdus spp. such as Photorhabdus luminescens, Xenorhabdus nematophilus
- toxins produced by animals such as scorpion toxins, arachnid toxins, wasp toxins and other insect-specific neurotoxins
- toxins produced by fungi such as Streptomycetes toxins, plant lectins, such as pea lectins, barley lectins or snowdrop lectins
- agglutinins proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin, papain inhibitors
- ribosome-inactivating proteins (RIP) such as ricin, maize-RIP, abrin, luffin, saporin or bryodin
- steroid metabolism enzymes such as 3-hydroxysteroidoxidase, ecdysteroid-UDP-glycosyl-transferase, cholesterol oxidases, ecd
- ⁇ -endotoxins for example CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), for example Vip1 , Vip2, Vip3 or Vip3A, expressly also hybrid toxins, truncated toxins and modified toxins.
- Hybrid toxins are produced recombinantly by a new combination of different domains of those proteins (see, for example, WO 02/15701 ).
- Truncated toxins for example a truncated CrylAb, are known.
- modified toxins one or more amino acids of the naturally occurring toxin are replaced.
- preferably non-naturally present protease recognition sequences are inserted into the toxin, such as, for example, in the case of Cry3A055, a cathepsin-G-recognition sequence is inserted into a Cry3A toxin (see WO 03/018810).
- Examples of such toxins or transgenic plants capable of synthesising such toxins are disclosed, for example, in EP-A-0 374 753, WO 93/07278, WO 95/34656, EP-A-0 427 529, EP-A-451 878 and WO 03/052073.
- deoxyribonucleic acids and their preparation are known, for example, from WO 95/34656, EP-A-0 367 474, EP-A-0 401 979 and WO 90/13651.
- the toxin contained in the transgenic plants imparts to the plants tolerance to harmful insects.
- insects can occur in any taxonomic group of insects, but are especially commonly found in the beetles (Coleoptera), two-winged insects (Diptera) and moths (Lepidoptera).
- Transgenic plants containing one or more genes that code for an insecticidal resistance and express one or more toxins are known and some of them are commercially available. Examples of such plants are: YieldGard® (maize variety that expresses a CrylAb toxin); YieldGard Rootworm® (maize variety that expresses a Cry3Bb1 toxin); YieldGard Plus® (maize variety that expresses a CrylAb and a Cry3Bb1 toxin); Starlink® (maize variety that expresses a Cry9C toxin); Herculex I® (maize variety that expresses a Cry1 Fa2 toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylAc toxin); Bollgard I® (cotton variety that expresses a
- transgenic crops are:
- Bt11 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a truncated Cry1 Ab toxin. Bt1 1 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium.
- MIR604 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Maize which has been rendered insect-resistant by transgenic expression of a modified Cry3A toxin. This toxin is Cry3A055 modified by insertion of a cathepsin-G- protease recognition sequence. The preparation of such transgenic maize plants is described in WO 03/018810.
- MON 863 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1 150 Brussels, Belgium, registration number C/DE/02/9. MON 863 expresses a Cry3Bb1 toxin and has resistance to certain Coleoptera insects.
- NK603 x MON 810 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1 150 Brussels, Belgium, registration number C/GB/02/M3/03. Consists of conventionally bred hybrid maize varieties by crossing the genetically modified varieties NK603 and MON 810. NK603 * MON 810
- Lepidoptera include the European corn borer.
- crops is to be understood as including also crop plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so-called "pathogenesis-related proteins" (PRPs, see e.g. EP-A-0 392 225).
- PRPs pathogenesis-related proteins
- Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP-A-0 392 225, WO 95/33818 and EP-A-0 353 191.
- the methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
- Crops may also be modified for enhanced resistance to fungal (for example Fusarium, Anthracnose, or Phytophthora), bacterial (for example Pseudomonas) or viral (for example potato leafroll virus, tomato spotted wilt virus, cucumber mosaic virus) pathogens.
- fungal for example Fusarium, Anthracnose, or Phytophthora
- bacterial for example Pseudomonas
- viral for example potato leafroll virus, tomato spotted wilt virus, cucumber mosaic virus
- Crops also include those that have enhanced resistance to nematodes, such as the soybean cyst nematode.
- Crops that are tolerance to abiotic stress include those that have enhanced tolerance to drought, high salt, high temperature, chill, frost, or light radiation, for example through expression of NF-YB or other proteins known in the art.
- Antipathogenic substances which can be expressed by such transgenic plants include, for example, ion channel blockers, such as blockers for sodium and calcium channels, for example the viral KP1 , KP4 or KP6 toxins; stilbene synthases; bibenzyl synthases; chitinases; glucanases; the so-called "pathogenesis-related proteins" (PRPs; see e.g.
- EP-A-0 392 225 antipathogenic substances produced by microorganisms, for example peptide antibiotics or heterocyclic antibiotics (see e.g. WO 95/33818) or protein or polypeptide factors involved in plant pathogen defence (so-called “plant disease resistance genes", as described in WO 03/000906).
- compositions according to the invention are the protection of stored goods and store ambients and the protection of raw materials, such as wood, textiles, floor coverings or buildings, and also in the hygiene sector, especially the protection of humans, domestic animals and productive livestock against pests of the mentioned type.
- the present invention also provides a method for controlling pests (such as mosquitoes and other disease vectors; see also http://www.who.int/malaria/vector_control/irs/en/).
- the method for controlling pests comprises applying the compositions of the invention to the target pests, to their locus or to a surface or substrate by brushing, rolling, spraying, spreading or dipping.
- an IRS (indoor residual spraying) application of a surface such as a wall, ceiling or floor surface is contemplated by the method of the invention.
- the method for controlling such pests comprises applying a pesticidally effective amount of the compositions of the invention to the target pests, to their locus, or to a surface or substrate so as to provide effective residual pesticidal activity on the surface or substrate.
- a pesticidally effective amount of the compositions of the invention to the target pests, to their locus, or to a surface or substrate so as to provide effective residual pesticidal activity on the surface or substrate.
- Such application may be made by brushing, rolling, spraying, spreading or dipping the pesticidal composition of the invention.
- an IRS application of a surface such as a wall, ceiling or floor surface is contemplated by the method of the invention so as to provide effective residual pesticidal activity on the surface.
- it is contemplated to apply such compositions for residual control of pests on a substrate such as a fabric material in the form of (or which can be used in the manufacture of) netting, clothing, bedding, curtains and tents.
- Substrates including non-woven, fabrics or netting to be treated may be made of natural fibres such as cotton, raffia, jute, flax, sisal, hessian, or wool, or synthetic fibres such as polyamide, polyester, polypropylene, polyacrylonitrile or the like.
- the polyesters are particularly suitable.
- the methods of textile treatment are known, e.g. WO 2008/151984, WO 2003/034823, US 5631072, WO 2005/64072, WO2006/128870, EP 1724392, WO20051 13886 or WO 2007/090739.
- compositions according to the invention are the field of tree injection/trunk treatment for all ornamental trees as well all sort of fruit and nut trees.
- the compounds according to the present invention are especially suitable against wood-boring insects from the order Lepidoptera as mentioned above and from the order Coleoptera, especially against woodborers listed in the following tables A and B:
- Agrilus sayi Bayberry, Sweetfern
- Rhododendron Rhadodendron, Azalea, Laurel, Poplar, Willow, Mulberry
- Phloeotribus liminaris Peach, Cherry, Plum, Black cherry,
- the present invention may be also used to control any insect pests that may be present in turfgrass, including for example beetles, caterpillars, fire ants, ground pearls, millipedes, sow bugs, mites, mole crickets, scales, mealybugs ticks, spittlebugs, southern chinch bugs and white grubs.
- the present invention may be used to control insect pests at various stages of their life cycle, including eggs, larvae, nymphs and adults.
- the present invention may be used to control insect pests that feed on the roots of turfgrass including white grubs (such as Cyclocephala spp. (e.g. masked chafer, C. lurida),
- white grubs such as Cyclocephala spp. (e.g. masked chafer, C. lurida)
- Rhizotrogus spp. e.g. European chafer, R. majalis
- Cotinus spp. e.g. Green June beetle, C. nitida
- Popillia spp. e.g. Japanese beetle, P. japonica
- Phyllophaga spp. e.g. May/June beetle
- Ataenius spp. e.g. Black turfgrass ataenius, A. spretulus
- Maladera spp. e.g. Asiatic garden beetle, M.
- the present invention may also be used to control insect pests of turfgrass that are thatch dwelling, including armyworms (such as fall armyworm Spodoptera frugiperda, and common armyworm Pseudaletia unipuncta), cutworms, billbugs (Sphenophorus spp. , such as S. venatus verstitus and S. parvulus), and sod webworms (such as Crambus spp. and the tropical sod webworm, Herpetogramma phaeopteralis).
- armyworms such as fall armyworm Spodoptera frugiperda, and common armyworm Pseudaletia unipuncta
- cutworms billbugs
- Sphenophorus spp. such as S. venatus verstitus and S. parvulus
- sod webworms such as Crambus spp. and the tropical sod webworm, Herpetogramma phaeopteralis.
- the present invention may also be used to control insect pests of turfgrass that live above the ground and feed on the turfgrass leaves, including chinch bugs (such as southern chinch bugs, Blissus insularis), Bermudagrass mite (Eriophyes cynodoniensis), rhodesgrass mealybug (Antonina graminis), two-lined spittlebug (Propsapia bicincta), leafhoppers, cutworms (Noctuidae family), and greenbugs.
- the present invention may also be used to control other pests of turfgrass such as red imported fire ants (Solenopsis invicta) that create ant mounds in turf.
- compositions according to the invention are active against ectoparasites such as hard ticks, soft ticks, mange mites, harvest mites, flies (biting and licking), parasitic fly larvae, lice, hair lice, bird lice and fleas.
- ectoparasites such as hard ticks, soft ticks, mange mites, harvest mites, flies (biting and licking), parasitic fly larvae, lice, hair lice, bird lice and fleas.
- Anoplurida Haematopinus spp., Linognathus spp., Pediculus spp. and Phtirus spp., Solenopotes spp..
- Mallophagida Trimenopon spp., Menopon spp., Trinoton spp., Bovicola spp.,
- Nematocerina and Brachycerina for example Aedes spp., Anopheles spp., Culex spp., Simulium spp., Eusimulium spp., Phlebotomus spp., Lutzomyia spp., Culicoides spp., Chrysops spp., Hybomitra spp., Atylotus spp., Tabanus spp., Haematopota spp.,
- Philipomyia spp. Braula spp., Musca spp., Hydrotaea spp., Stomoxys spp., Haematobia spp., Morellia spp., Fannia spp., Glossina spp., Calliphora spp., Lucilia spp., Chrysomyia spp., Wohlfahrtia spp., Sarcophaga spp., Oestrus spp., Hypoderma spp., Gasterophilus spp., Hippobosca spp., Lipoptena spp. and Melophagus spp..
- Siphonaptrida for example Pulex spp., Ctenocephalides spp., Xenopsylla spp., Ceratophyllus spp..
- Heteropterida for example Cimex spp., Triatoma spp., Rhodnius spp., Panstrongylus spp..
- Actinedida Prostigmata
- Acaridida Acaridida
- Acarapis spp. Cheyletiella spp., Ornitrocheyletia spp., Myobia spp., Psorergatesspp., Demodex spp., Trombicula spp., Listrophorus spp., Acarus spp., Tyrophagus spp., Caloglyphus spp., Hypodectes spp.,
- Pterolichus spp. Psoroptes spp., Chorioptes spp., Otodectes spp., Sarcoptes spp., Notoedres spp., Knemidocoptes spp., Cytodites spp. and Laminosioptes spp..
- compositions according to the invention are also suitable for protecting against insect infestation in the case of materials such as wood, textiles, plastics, adhesives, glues, paints, paper and card, leather, floor coverings and buildings.
- compositions according to the invention can be used, for example, against the following pests: beetles such as Hylotrupes bajulus, Chlorophorus pilosis, Anobium punctatum, Xestobium
- rufovillosum Ptilinuspecticornis, Dendrobium pertinex, Ernobius mollis, Priobium carpini, Lyctus brunneus, Lyctus africanus, Lyctus planicollis, Lyctus linearis, Lyctus pubescens, Trogoxylon aequale, Minthesrugicollis, Xyleborus spec.,Tryptodendron spec, Apate monachus, Bostrychus capucins, Heterobostrychus brunneus, Sinoxylon spec, and Dinoderus minutus, and also hymenopterans such as Sirex juvencus, Urocerus gigas, Urocerus gigas taignus and Urocerus augur, and termites such as Kalotermes flavicollis, Cryptotermes brevis, Heterotermes indicola, Reticulitermes flavipes,
- Reticulitermes santonensis Reticulitermes lucifugus
- Mastotermes darwiniensis Zootermopsis nevadensis and Coptotermes formosanus
- bristletails such as Lepisma saccharina.
- the compounds according to the invention can be used as pesticidal agents in unmodified form, but they are generally formulated into compositions in various ways using formulation adjuvants, such as carriers, solvents and surface-active substances.
- formulation adjuvants such as carriers, solvents and surface-active substances.
- the formulations can be in various physical forms, e.g.
- Such formulations can either be used directly or diluted prior to use.
- the dilutions can be made, for example, with water, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
- the formulations can be prepared e.g. by mixing the active ingredient with the formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions.
- the active ingredients can also be formulated with other adjuvants, such as finely divided solids, mineral oils, oils of vegetable or animal origin, modified oils of vegetable or animal origin, organic solvents, water, surface-active substances or combinations thereof.
- the active ingredients can also be contained in very fine microcapsules.
- Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into the environment in controlled amounts (e.g. slow-release).
- Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight.
- the active ingredients can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution.
- the encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art.
- very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
- liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2-butanone, butylene carbonate, chlorobenzene, cyclohexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1 ,2-dichloropropane, diethanolamine, p- diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, A/,A/-dimethylformamide, dimethyl sulfoxide, 1 ,4- dioxane, di
- Suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montmorillonite, cottonseed husks, wheat flour, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar substances.
- a large number of surface-active substances can advantageously be used in both solid and liquid formulations, especially in those formulations which can be diluted with a carrier prior to use.
- Surface- active substances may be anionic, cationic, non-ionic or polymeric and they can be used as emulsifiers, wetting agents or suspending agents or for other purposes.
- Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol/alkylene oxide addition products, such as nonylphenol ethoxylate; alcohol/alkylene oxide addition products, such as tridecylalcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonat.es, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2- ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryltrimethylammonium chloride, polyethylene glycol est
- Further adjuvants that can be used in pesticidal formulations include crystallisation inhibitors, viscosity modifiers, suspending agents, dyes, anti-oxidants, foaming agents, light absorbers, mixing auxiliaries, antifoams, complexing agents, neutralising or pH-modifying substances and buffers, corrosion inhibitors, fragrances, wetting agents, take-up enhancers, micronutrients, plasticisers, glidants, lubricants, dispersants, thickeners, antifreezes, microbicides, and liquid and solid fertilisers.
- compositions according to the invention can include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives.
- the amount of oil additive in the composition according to the invention is generally from 0.01 to 10 %, based on the mixture to be applied.
- the oil additive can be added to a spray tank in the desired concentration after a spray mixture has been prepared.
- Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow.
- Preferred oil additives comprise alkyl esters of C8-C22 fatty acids, especially the methyl derivatives of C12-C18 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid (methyl laurate, methyl palmitate and methyl oleate, respectively).
- Many oil derivatives are known from the Compendium of Herbicide Adjuvants, 10 th Edition, Southern Illinois University, 2010.
- the inventive compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, of compounds of the present invention and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance. Whereas commercial products may preferably be formulated as concentrates, the end user will normally employ dilute formulations.
- the rates of application vary within wide limits and depend on the nature of the soil, the method of application, the crop plant, the pest to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- a general guideline compounds may be applied at a rate of from 1 to 2000 l/ha, especially from 10 to 1000 l/ha.
- Preferred formulations can have the following compositions (weight %):
- Emulsifiable concentrates are:
- active ingredient 1 to 95 %, preferably 60 to 90 %
- surface-active agent 1 to 30 %, preferably 5 to 20 %
- liquid carrier 1 to 80 %, preferably 1 to 35 %
- active ingredient 0.1 to 10 %, preferably 0.1 to 5 %
- solid carrier 99.9 to 90 %, preferably 99.9 to 99 %
- active ingredient 5 to 75 %, preferably 10 to 50 %
- surface-active agent 1 to 40 %, preferably 2 to 30 %
- active ingredient 0.5 to 90 %, preferably 1 to 80 %
- surface-active agent 0.5 to 20 %, preferably 1 to 15 %
- solid carrier 5 to 95 %, preferably 15 to 90 %
- active ingredient 0.1 to 30 %, preferably 0.1 to 15 %
- solid carrier 99.5 to 70 %, preferably 97 to 85 %
- active ingredients 25 % 50 % 75 % sodium lignosulfonate 5 % 5 % - sodium lauryl sulfate 3 % - 5 % sodium diisobutylnaphthalenesulfonate - 6 % 10 % phenol polyethylene glycol ether (7-8 mol of ethylene 2 %
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsions of any required dilution which can be used in plant protection, can be obtained from this concentrate by dilution with water.
- Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
- the combination is mixed and ground with the adjuvants, and the mixture is moistened with water.
- the mixture is extruded and then dried in a stream of air.
- the finely ground combination is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol.
- Non-dusty coated granules are obtained in this manner.
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- 28 parts of the combination are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8: 1 ).
- This mixture is emulsified in a mixture of 1 .2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51 .6 parts of water until the desired particle size is achieved.
- To this emulsion a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added. The mixture is agitated until the polymerization reaction is completed.
- the obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent.
- the capsule suspension formulation contains 28% of the active ingredients.
- the medium capsule diameter is 8-15 microns.
- the resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
- Formulation types include an emulsion concentrate (EC), a suspension concentrate (SC), a suspo- emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK), a dispersible concentrate (DC), a wettable powder (WP), a soluble granule (SG) or any technically feasible formulation in combination with agriculturally acceptable adjuvants.
- EC emulsion concentrate
- SC suspension concentrate
- SE suspo- emulsion
- CS capsule suspension
- WG water dispersible granule
- Mp melting point in °C. Free radicals represent methyl groups. H NMR and 9 F NMR measurements were recorded on a Brucker 400 MHz or 300 MHz spectrometer, chemical shifts are given in ppm relevant to a TMS standard. Spectra measured in deuterated solvents as indicated.
- Spectra were recorded on a Mass Spectrometer from Waters (SQD, SQDII or ZQ Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, Capillary: 3.00 kV, Cone range: 30-60 V, Extractor: 2.00 V, Source Temperature: 150°C, Desolvation Temperature: 350°C, Cone Gas Flow: 0 L/Hr, Desolvation Gas Flow: 650 L/Hr, Mass range: 100 to 900 Da) and an Acquity UPLC from Waters: Binary pump, heated column compartment and diode- array detector. Solvent degasser, binary pump, heated column compartment and diode-array detector.
- Spectra were recorded on a Mass Spectrometer from Waters (SQD or ZQ Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, Capillary: 3.00 kV, Cone range: 30-60 V, Extractor: 2.00 V, Source Temperature: 150°C, Desolvation Temperature: 350°C, Cone Gas Flow: 0 L/Hr, Desolvation Gas Flow: 650 L/Hr, Mass range: 100 to 900 Da) and an Acquity UPLC from Waters: Binary pump, heated column compartment and diode-array detector. Solvent degasser, binary pump, heated column compartment and diode-array detector.
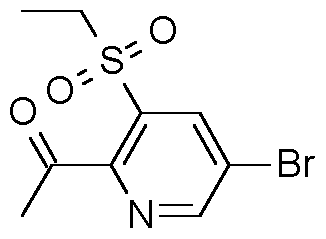
- Step 1 Preparation of 5-bromo-3-ethvsulfanyl-pyridine-2-carbonitrile
- Step 5 Preparation of 2-(5-bromo-3-ethylsulfonyl-2-pyridvn-7-(trifluoromethvnimidazo[1 ,2-alpyridine
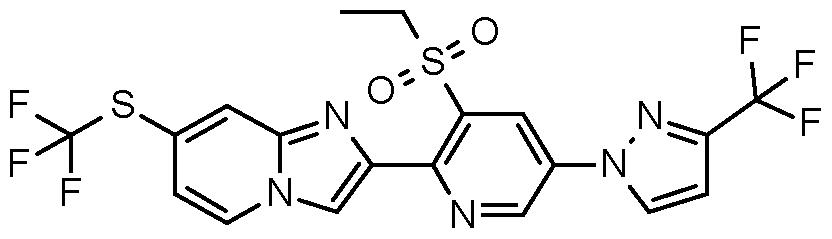
- Step 6 Preparation of 2-[5-(3,5-difluorophenyl)-3-ethylsulfonyl-2-pyridyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
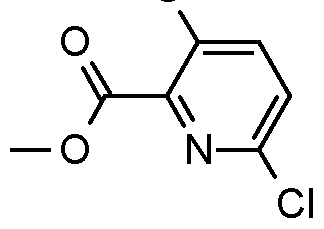
- Step 1 Preparation of methyl 3,6-dichloropyridine-2-carboxylate
- Step 2 Preparation of methyl 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylate
- Step 3 Preparation of methyl 3-ethylsulfanyl-6-[4-(trifluoromethyl)phenyllpyridine-2-carboxylate
- Step 4 Preparation of methyl 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyllpyridine-2-carboxylate
- Methyl 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]pyridine-2-carboxylate (3.64 g, 9.75 mmol) was dissolved in tetrahydrofuran (54.6 mL) and water (18.2 mL) . Then hydroxylithium hydrate (0.429 g, 10.2 mmol) was added at ambient temperature. Reaction mixture was stirred one night at ambient temperature.
- N-methoxymethanamine hydrochloride (1.1 g, 1 1 mmol) was solved with dichloromethane (62 mL) and TRIETHYLAMINE (3.8 g, 5.3 mL, 38 mmol) was added. Reaction mixture was cooled down at 0-5°C and 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]pyridine-2- carbonyl chloride previously prepared (4.1 g, 100, 1 1 mmol) was dissolved in 3 ml of dichloromethane and added slowly at 0°C. Reaction mixture was stirred 1 hour at 0°C.
- reaction mixture was warm up at ambient temperature and stirred 30'. Water was added in the reaction mixture, organic layer was separated and aqueous layer was extracted 2 times with dichloromethane. The combined organic layer was washed with water, dried over Na2S04, filtered and evaporated under vacuum. The crude was purified by flash chromatography on silica gel to give the title compound as a colorless oil.
- Step 7 Preparation of 1-[3-ethylsulfonyl-6-[4-(trifluoromethvnphenyll-2-pyridyllethanone
- the crude was slowly quenched with NH4CI sat aq (10 ml) and HCI 10% (5 ml) and resulting mixture was strongly stirred 15' at ambient temperature.
- the aqueous layer was extracted twice with ethyl acetate.
- the organic phase was then washed with 10 % HCI aq sol, water and brine, dried over Na2S04 anhydrous, filtered and concentrated.
- the crude was purified two times by flash
- Step 9 Preparation of 2-[3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyll-2-pyridyll-7-
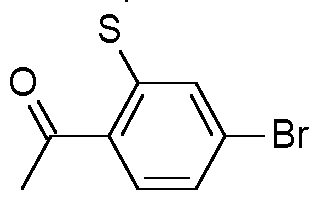
- Step 1 Preparation of 1-(4-bromo-2-ethylsulfanyl-phenyl)ethanone
- Step 3 Preparation of 1-[2-ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyllethanone
- Step 4 Preparation of 2-bromo-1-[2-ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyllethanone
- Step 5 Preparation of 2-[2-ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
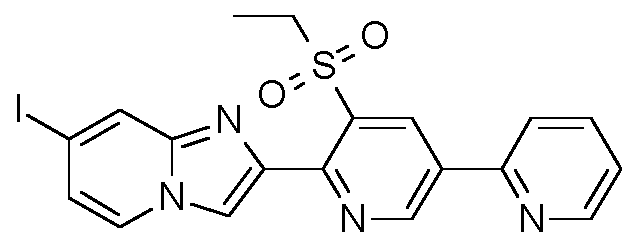
- Step 2 Preparation of 2-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyll-7-iodo-imidazo[1 ,2-alpyridine
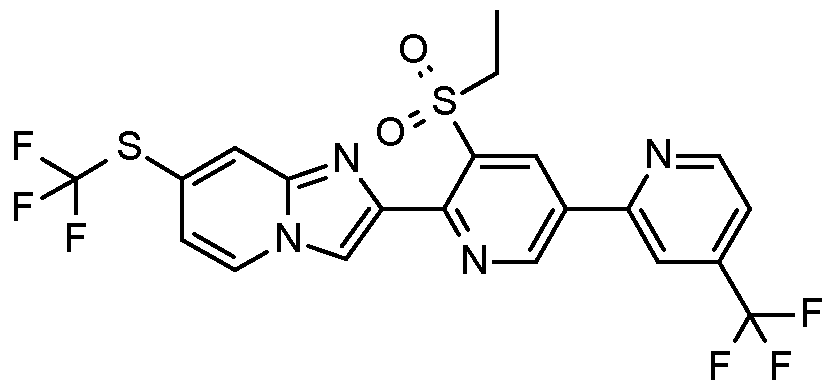
- Example H7 Preparation of 2-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyll-7-(1 , 1 ,2,2,2- pentafluoroethyl)imidazo[1 ,2-alpyridine
- Stepl Preparation of 1-[3-ethylsulfanyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyllethanone:
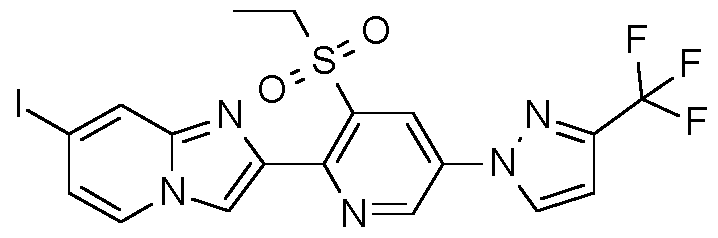
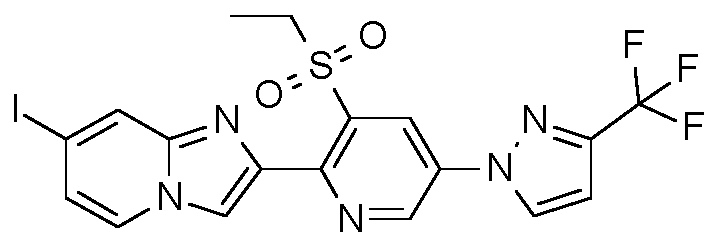
- Step 3 Preparation of 2-bromo-1-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyllethanone
- Step 3 Preparation of 2-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyll-7-iodo- imidazoM ,2-alpyridine:
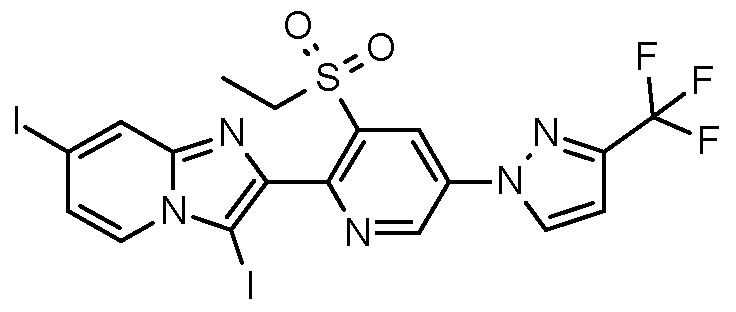
- Example H10 Preparation of 2-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyll-3,7-diiodo- imidazoM ,2-alpyridine.
- Step 1 Preparation of 1-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyllethanone
- Step 2 Preparation of 2-bromo-1-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyllethanone
- Step 3 Preparation of 2-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyll-7-iodo-imidazo[1 ,2- al pyridine
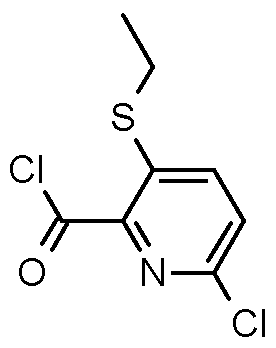
- Step 1 Preparation of 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylic acid
- Step 2 Preparaton of 6-chloro-3-ethylsulfanyl-pyridine-2-carbonyl chloride
- Step 3 Preparaton of 6-chloro-3-ethylsulfanyl-N-methoxy-N-methyl-pyridine-2-carboxamide
- Step 6 Preparation of 1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyllethanone
- Step 7 Preparation of tert-butyl-[1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyllvinyloxyl-dimethyl- silane
- Step 8 Preparation of 2-bromo-1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyllethanone
- reaction mixture was diluted with additional phosphate buffer (pH 7, 10 mL of a 0.5M solution) and ieri-butyl methyl ether (10 mL).
- the organic layer was separated, washed with water (3x), dried (Na2S04) and concentrated in vacuo.
- the obtained tan solid was suspended small amounts of TBME amnd filtered (3x). The remaining solid was then dissolved in chloroform, washed with water (2x) and brine, dried (Na2S04) to obtain the crude title compound which was used in the next step without further purification.
- Step 9 Preparation of 2-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyll-7-(trifluoromethyl)imidazo[1 ,2-
- a microwave vial was charged with a mixture of 2-bromo-1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2- pyridyljethanone (390 mg, 1 .08 mmol, 1.00 equiv.) and 4-(trifluoromethyl)pyridin-2-amine (194 mg, 1.19 mmol, 1.10 equiv.) in acetonitrile (4.0 mL).
- the vial was sealed and heated to 150 °C under microwave irradiation for 30 min.
- the obtained crude mixture was concentrated in vacuo and subjected to flash chromatography (silica gel, cyclohexane/ethyl acetate) to obtain the title compound as a white solid.
- compositions according to the invention can be broadened considerably, and adapted to prevailing circumstances, by adding other insecticidally, acaricidally and/or fungicidally active ingredients.
- the mixtures of the compounds of formula I with other insecticidally, acaricidally and/or fungicidally active ingredients may also have further surprising advantages which can also be described, in a wider sense, as synergistic activity. For example, better tolerance by plants, reduced phytotoxicity, insects can be controlled in their different development stages or better behaviour during their production, for example during grinding or mixing, during their storage or during their use.
- Suitable additions to active ingredients here are, for example, representatives of the following classes of active ingredients: organophosphorus compounds, nitrophenol derivatives, thioureas, juvenile hormones, formamidines, benzophenone derivatives, ureas, pyrrole derivatives, carbamates, pyrethroids, chlorinated hydrocarbons, acylureas, pyridylmethyleneamino derivatives, macrolides, neonicotinoids and Bacillus thuringiensis preparations.
- TX means "one compound selected from the group consisting of the compounds described in Table P of the present invention”: an adjuvant selected from the group of substances consisting of petroleum oils (628) + TX, an acaricide selected from the group of substances consisting of 1 , 1-bis(4-chlorophenyl)-2- ethoxyethanol (lUPAC name) (910) + TX,2,4-dichlorophenyl benzenesulfonate (lUPAC/Chemical Abstracts name) (1059) + TX,2-fluoro-A/-methyl-A/-1-naphthylacetamide (lUPAC name) (1295) + TX,4- chlorophenyl phenyl sulfone (lUPAC name) (981 ) + TX, abamectin (1 ) + TX, acequinocyl (3) + TX, acetoprole [CCN]
- a bactericide selected from the group of substances consisting of 1-hydroxy-1 /- -pyridine-2-thione (lUPAC name) (1222) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (lUPAC name) (748) + TX, 8-hydroxyquinoline sulfate (446) + TX, bronopol (97) + TX, copper dioctanoate (lUPAC name) (170) + TX, copper hydroxide (lUPAC name) (169) + TX, cresol [CCN] + TX, dichlorophen (232) + TX, dipyrithione (1 105) + TX, dodicin (1 1 12) + TX, fenaminosulf (1 144) + TX, formaldehyde (404) + TX, hydrargaphen [CCN] + TX, kasugamycin (483) + TX, kasugamycin hydrochloride hydrate (483) + TX, nickel
- a biological agent selected from the group of substances consisting of Adoxophyes orana GV (12) + TX, Agrobacterium radiobacter (13) + TX, Amblyseius spp. (19) + TX, Anagrapha falcifera NPV (28) + TX, Anagrus atomus (29) + TX, Aphelinus abdominalis (33) + TX, Aphidius colemani (34) + TX, Aphidoletes aphidimyza (35) + TX, Autographa californica NPV (38) + TX, Bacillus firmus (48) + TX, Bacillus sphaericus Neide (scientific name) (49) + TX, Bacillus thuringiensis Kirk (scientific name) (51 ) + TX, Bacillus thuringiensis subsp.
- a soil sterilant selected from the group of substances consisting of iodomethane (lUPAC name) (542) and methyl bromide (537) + TX,
- a chemosterilant selected from the group of substances consisting of apholate [CCN] + TX, bisazir [CCN] + TX, busulfan [CCN] + TX, diflubenzuron (250) + TX, dimatif [CCN] + TX, hemel [CCN] + TX, hempa [CCN] + TX, metepa [CCN] + TX, methiotepa [CCN] + TX, methyl apholate [CCN] + TX, morzid [CCN] + TX, penfluron [CCN] + TX, tepa [CCN] + TX, thiohempa [CCN] + TX, thiotepa [CCN] + TX, tretamine [CCN] and uredepa [CCN] + TX,
- an insect pheromone selected from the group of substances consisting of (E)-dec-5-en-1-yl acetate with (E)-dec-5-en-1-ol (lUPAC name) (222) + TX, (E)-tridec-4-en-1-yl acetate (lUPAC name) (829) + TX, (E)-6-methylhept-2-en-4-ol (lUPAC name) (541 ) + TX, (E,Z)-tetradeca-4, 10-dien-1-yl acetate (lUPAC name) (779) + TX, (Z)-dodec-7-en-1-yl acetate (lUPAC name) (285) + TX, (Z)-hexadec-l 1- enal (lUPAC name) (436) + TX, (Z)-hexadec-l 1-en-1-yl acetate (lUPAC name) (437) + TX, (Z)- hexade
- an insecticide selected from the group of substances consisting of 1-dichloro-1-nitroethane
- pentachlorophenyl laurate (lUPAC name) (623) + TX, permethrin (626) + TX, petroleum oils (628) + TX, PH 60-38 (development code) (1328) + TX, phenkapton (1330) + TX, phenothrin (630) + TX, phenthoate (631 ) + TX, phorate (636) + TX, phosalone (637) + TX, phosfolan (1338) + TX, phosmet (638) + TX, phosnichlor (1339) + TX, phosphamidon (639) + TX, phosphine (lUPAC name) (640) + TX, phoxim (642) + TX, phoxim-methyl (1340) + TX, pirimetaphos (1344) + TX, pirimicarb (651 ) + TX, pirimiphos-ethyl (1345) + TX,
- TX TX
- tralomethrin (812) + TX TX
- transfluthrin (813) + TX transpermethrin (1440) + TX
- triamiphos (1441 ) + TX triazamate (818) + TX
- triazophos (820) + TX triazuron + TX, trichlorfon (824) + TX,
- trichlormetaphos-3 [CCN] + TX, trichloronat (1452) + TX, trifenofos (1455) + TX, triflumuron (835) + TX, trimethacarb (840) + TX, triprene (1459) + TX, vamidothion (847) + TX, vaniliprole [CCN] + TX, veratridine (725) + TX, veratrine (725) + TX, XMC (853) + TX, xylylcarb (854) + TX, YI-5302
- a molluscicide selected from the group of substances consisting of bis(tributyltin) oxide (lUPAC name) (913) + TX, bromoacetamide [CCN] + TX, calcium arsenate [CCN] + TX, cloethocarb (999) + TX, copper acetoarsenite [CCN] + TX, copper sulfate (172) + TX, fentin (347) + TX, ferric phosphate
- a nematicide selected from the group of substances consisting of AKD-3088 (compound code) + TX, 1 ,2-dibromo-3-chloropropane (lUPAC/Chemical Abstracts name) (1045) + TX, 1 ,2-dichloropropane (lUPAC/ Chemical Abstracts name) (1062) + TX, 1 ,2-dichloropropane with 1 ,3-dichloropropene (lUPAC name) (1063) + TX, 1 ,3-dichloropropene (233) + TX, 3,4-dichlorotetrahydrothiophene 1 ,1- dioxide (lUPAC/Chemical Abstracts name) (1065) + TX,3-(4-chlorophenyl)-5-methylrhodanine (lUPAC name) (980) + TX, 5-methyl-6-thioxo-1 ,3,5-thiadiazinan-3-ylacetic acid (lUPAC name) (12
- a nitrification inhibitor selected from the group of substances consisting of potassium ethylxanthate [CCN] and nitrapyrin (580) + TX,
- a plant activator selected from the group of substances consisting of acibenzolar (6) + TX, acibenzolar-S-methyl (6) + TX, probenazole (658) and Reynoutria sachalinensis extract (720) + TX
- a rodenticide selected from the group of substances consisting of 2-isovalerylindan-1 ,3-dione (lUPAC name) (1246) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (lUPAC name) (748) + TX, alpha- chlorohydrin [CCN] + TX, aluminium phosphide (640) + TX, antu (880) + TX, arsenous oxide (882) + TX, barium carbonate (891 ) + TX, bisthiosemi (912) + TX, brodifacoum (89) + TX, bromadiolone (91 ) + TX, bromethalin (92) +
- a synergist selected from the group of substances consisting of 2-(2-butoxyethoxy)ethyl piperonylate (lUPAC name) (934) + TX, 5-(1 ,3-benzodioxol-5-yl)-3-hexylcyclohex-2-enone (lUPAC name) (903) + TX, farnesol with nerolidol (324) + TX, MB-599 (development code) (498) + TX, MGK 264
- development code (296) + TX, piperonyl butoxide (649) + TX, piprotal (1343) + TX, propyl isomer (1358) + TX, S421 (development code) (724) + TX, sesamex (1393) + TX, sesasmolin (1394) and sulfoxide (1406) + TX,
- an animal repellent selected from the group of substances consisting of anthraquinone (32) + TX, chloralose (127) + TX, copper naphthenate [CCN] + TX, copper oxychloride (171 ) + TX, diazinon (227) + TX, dicyclopentadiene (chemical name) (1069) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, methiocarb (530) + TX, pyridin-4-amine (lUPAC name) (23) + TX, thiram (804) + TX, trimethacarb (840) + TX, zinc naphthenate [CCN] and ziram (856) + TX,
- a virucide selected from the group of substances consisting of imanin [CCN] and ribavirin [CCN] + TX,
- a wound protectant selected from the group of substances consisting of mercuric oxide (512) + TX,octhilinone (590) and thiophanate-methyl (802) + TX, and biologically active compounds selected from the group consisting of azaconazole (60207-31-0] + TX, bitertanol [70585-36-3] + TX, bromuconazole [1 16255-48-2] + TX, cyproconazole [94361-06-5] + TX, difenoconazole [1 19446-68-3] + TX, diniconazole [83657-24-3] + TX, epoxiconazole [106325-08- 0] + TX, fenbuconazole [1 14369-43-6] + TX, fluquinconazole [136426-54-5] + TX, flusilazole [85509- 19-9] + TX, flutriafol [76674-21-0] + T
- Acinetobacter Iwoffii + TX Acremonium alternatum + TX + TX, Acremonium cephalosporium + TX + TX, Acremonium diospyri + TX, Acremonium obclavatum + TX, Adoxophyes orana granulovirus (AdoxGV) (Capex®) + TX, Agrobacterium radiobacter strain K84 (Galltrol-A®) + TX, Alternaria alternate + TX, Alternaria cassia + TX, Alternaria destruens (Smolder®) + TX,
- Ampelomyces quisqualis (AQ10®) + TX, Aspergillus flavus AF36 (AF36®) + TX, Aspergillus flavus NRRL 21882 (Aflaguard®) + TX, Aspergillus spp. + TX, Aureobasidium pullulans + TX, Azospirillum + TX, (MicroAZ® + TX, TAZO B®) + TX, Azotobacter + TX, Azotobacter chroocuccum (Azotomeal®) + TX, Azotobacter cysts (Bionatural Blooming Blossoms®) + TX, Bacillus amyloliquefaciens + TX,
- Bacillus cereus + TX Bacillus chitinosporus strain CM-1 + TX, Bacillus chitinosporus strain AQ746 + TX, Bacillus licheniformis strain HB-2 (BiostartTM Rhizoboost®) + TX, Bacillus licheniformis strain 3086 (EcoGuard® + TX, Green Releaf®) + TX, Bacillus circulans + TX, Bacillus firmus (BioSafe® + TX, BioNem-WP® + TX, VOTiVO®) + TX, Bacillus firmus strain 1-1582 + TX, Bacillus macerans + TX, Bacillus marismortui + TX, Bacillus megaterium + TX, Bacillus mycoides strain AQ726 + TX, Bacillus papillae (Milky Spore Powder®) + TX, Bacillus pumilus spp.
- amyloliquefaciens strain FZB24 (Taegro® + TX, Rhizopro®) + TX, Bacillus thuringiensis Cry 2Ae + TX, Bacillus thuringiensis Cry1 Ab + TX, Bacillus thuringiensis aizawai GC 91 (Agree®) + TX, Bacillus thuringiensis israelensis (BMP123® + TX, Aquabac® + TX, VectoBac®) + TX, Bacillus thuringiensis kurstaki
- aizawai (XenTari® + TX, DiPel®) + TX, bacteria spp. (GROWMEND® + TX, GROWSWEET® + TX, Shootup®) + TX, bacteriophage of Clavipacter michiganensis (AgriPhage®) + TX, Bakflor® + TX, Beauveria bassiana (Beaugenic® + TX, Brocaril WP®) + TX, Beauveria bassiana GHA (Mycotrol ES® + TX, Mycotrol O® + TX, BotaniGuard®) + TX, Beauveria brongniartii (Engerlingspilz® + TX,
- Bradyrhizobium japonicum (TerraMax®) + TX, Brevibacillus brevis + TX, Bacillus thuringiensis tenebrionis (Novodor®) + TX, BtBooster + TX, Burkholderia cepacia (Deny® + TX, Intercept® + TX, Blue Circle®) + TX, Burkholderia gladii + TX, Burkholderia gladioli + TX, Burkholderia spp.
- TX Canadian thistle fungus (CBH Canadian Bioherbicide®) + TX, Candida butyri + TX, Candida famata + TX, Candida fructus + TX, Candida glabrata + TX, Candida guilliermondii + TX, Candida melibiosica + TX, Candida oleophila strain O + TX, Candida parapsilosis + TX, Candida pelliculosa + TX, Candida pulcherrima + TX, Candida reuêtii + TX, Candida saitoana (Bio-Coat® + TX, Biocure®) + TX, Candida sake + TX, Candida spp.
- TX Cladosporium tenuissimum + TX, Clonostachys rosea (EndoFine®) + TX, Colletotrichum acutatum + TX, Coniothyrium minitans (Cotans WG®) + TX, Coniothyrium spp. + TX, Cryptococcus albidus (YIELDPLUS®) + TX, Cryptococcus humicola + TX, Cryptococcus infirmo-miniatus + TX,
- Cryptococcus laurentii + TX TX
- Cupriavidus campinensis + TX Cydia pomonella granulovirus (CYD-X®) + TX
- Drechslera hawaiinensis + TX Enterobacter cloacae + TX
- Enterobacteriaceae + TX Entomophtora virulenta (Vektor®) + TX
- Epicoccum nigrum + TX Epicoccum purpurascens + TX, Epicoccum
- TX Filobasidium floriforme + TX, Fusarium acuminatum + TX, Fusarium chlamydosporum + TX, Fusarium oxysporum (Fusaclean® / Biofox C®) + TX, Fusarium proliferatum + TX, Fusarium spp. + TX, Galactomyces geotrichum + TX, Gliocladium catenulatum (Primastop® + TX, Prestop®) + TX, Gliocladium roseum + TX, Gliocladium spp. (SoilGard®) + TX, Gliocladium virens (Soilgard®) + TX, Granulovirus
- Pseudomonas chlororaphis (AtEze®) + TX, Pseudomonas corrugate + TX, Pseudomonas fluorescens strain A506 (BlightBan A506®) + TX, Pseudomonas putida + TX, Pseudomonas reactans + TX, Pseudomonas spp.
- TX Scytalidium uredinicola + TX, Spodoptera exigua nuclear polyhedrosis virus (Spod-X® + TX, Spexit®) + TX, Serratia marcescens + TX, Serratia plymuthica + TX, Serratia spp. + TX, Sordaria fimicola + TX, Spodoptera littoralis nucleopolyhedrovirus (Littovir®) + TX,
- Trichoderma asperellum T34 Biocontrol®
- Trichoderma gamsii TX
- Trichoderma atroviride Plantmate®
- Trichoderma harzianum rifai Mycostar®
- Trichoderma harzianum T-22 Trianum- P® + TX, PlantShield HC® + TX, RootShield® + TX, Trianum-G®) + TX, Trichoderma harzianum T-39 (Trichodex®) + TX, Trichoderma inhamatum + TX, Trichoderma koningii + TX, Trichoderma spp.
- LC 52 (Sentinel®) + TX, Trichoderma lignorum + TX, Trichoderma longibrachiatum + TX, Trichoderma polysporum (Binab T®) + TX, Trichoderma taxi + TX, Trichoderma virens + TX, Trichoderma virens (formerly Gliocladium virens GL-21 ) (SoilGuard®) + TX, Trichoderma viride + TX, Trichoderma viride strain ICC 080 (Remedier®) + TX, Trichosporon pullulans + TX, Trichosporon spp. + TX,
- Trichothecium spp. + TX Trichothecium roseum + TX, Typhula phacorrhiza strain 94670 + TX,
- Plant extracts including: pine oil (Retenol®) + TX, azadirachtin (Plasma Neem Oil® + TX, AzaGuard® + TX, MeemAzal® + TX, Molt-X® + TX, Botanical IGR (Neemazad® + TX, Neemix®) + TX, canola oil (Lilly Miller Vegol®) + TX, Chenopodium ambrosioides near ambrosioides (Requiem®) + TX, Chrysanthemum extract (Crisant®) + TX, extract of neem oil (Trilogy®) + TX, essentials oils of
- pheromones including: blackheaded fireworm pheromone (3M Sprayable Blackheaded Fireworm Pheromone®) + TX, Codling Moth Pheromone (Paramount dispenser-(CM)/ Isomate C-Plus®) + TX, Grape Berry Moth Pheromone (3M MEC-GBM Sprayable Pheromone®) + TX, Leafroller pheromone (3M MEC - LR Sprayable Pheromone®) + TX, Muscamone (Snip7 Fly Bait® + TX, Starbar Premium Fly Bait®) + TX, Oriental Fruit Moth Pheromone (3M oriental fruit moth sprayable pheromone®) + TX, Peachtree Borer Pheromone (Isomate-P®) + TX, Tomato Pinworm Pheromone (3M Sprayable pheromone®) + TX, Entostat powder (extract from palm tree) (Exosex CM®) + TX, (E + ⁇ , ⁇ +
- Macrobials including: Aphelinus abdominalis + TX, Aphidius ervi (Aphelinus-System®) + TX,
- TX Coccidoxenoides perminutus (Planopar®) + TX, Coccophagus cowperi + TX, Coccophagus lycimnia + TX, Cotesia flavipes + TX, Cotesia plutellae + TX, Cryptolaemus montrouzieri (Cryptobug® + TX, Cryptoline®) + TX, Cybocephalus nipponicus + TX, Dacnusa sibirica + TX, Dacnusa sibirica
- Diglyphus isaea (Diminex®) + TX, Delphastus catalinae (Delphastus®) + TX, Delphastus pusillus + TX, Diachasmimorpha krausii + TX, Diachasmimorpha longicaudata + TX, Diaparsis jucunda + TX, Diaphorencyrtus aligarhensis + TX, Diglyphus isaea + TX, Diglyphus isaea (Miglyphus® + TX, Digline®) + TX, Dacnusa sibirica (DacDigline® + TX, Minex®) + TX, Diversinervus spp.
- TX Steinernematid spp. (Guardian Nematodes®) + TX, Stethorus punctillum (Stethorus®) + TX, Tamarixia radiate + TX, Tetrastichus setifer + TX, Thripobius semiluteus + TX, Torymus sinensis + TX, Trichogramma brassicae (Tricholine b®) + TX, Trichogramma brassicae (Tricho-Strip®) + TX, Trichogramma evanescens + TX, Trichogramma minutum + TX, Trichogramma ostriniae + TX, Trichogramma platneri + TX, Trichogramma pretiosum + TX, Xanthopimpla stemmator; and
- the designation is not a "common name”
- the nature of the designation used instead is given in round brackets for the particular compound; in that case, the lUPAC name, the lUPAC/Chemical Abstracts name, a "chemical name”, a “traditional name”, a “compound name” or a “development code” is used or, if neither one of those designations nor a "common name” is used, an "alternative name” is employed.
- "CAS Reg. No” means the Chemical Abstracts Registry Number.
- the active ingredient mixture of the compounds of formula I selected from Table P with active ingredients described above comprises a compound selected from Table P and an active ingredient as described above preferably in a mixing ratio of from 100: 1 to 1 :6000, especially from 50:1 to 1 :50, more especially in a ratio of from 20: 1 to 1 :20, even more especially from 10: 1 to 1 :10, very especially from 5: 1 and 1 :5, special preference being given to a ratio of from 2:1 to 1 :2, and a ratio of from 4: 1 to 2: 1 being likewise preferred, above all in a ratio of 1 : 1 , or 5: 1 , or 5:2, or 5:3, or 5:4, or 4:1 , or 4:2, or 4:3, or 3: 1 , or 3:2, or 2:1 , or 1 :5, or 2:5, or 3:5, or 4:5, or 1 :4, or 2:4, or 3:4, or 1 :3, or 2:3, or 1 :2, or 1 :600, or 1 :300, or 1
- the mixtures as described above can be used in a method for controlling pests, which comprises applying a composition comprising a mixture as described above to the pests or their environment, with the exception of a method for treatment of the human or animal body by surgery or therapy and diagnostic methods practised on the human or animal body.
- the mixtures comprising a compound of formula I selected from Table P and one or more active ingredients as described above can be applied, for example, in a single "ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a "tank-mix", and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- the order of applying the compounds of formula I selected from Table P and the active ingredients as described above is not essential for working the present invention.
- compositions according to the invention can also comprise further solid or liquid auxiliaries, such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematocides, plant activators, molluscicides or herbicides.
- auxiliaries such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematocides
- compositions according to the invention are prepared in a manner known per se, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries).
- auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries).
- compositions that is the methods of controlling pests of the abovementioned type, such as spraying, atomizing, dusting, brushing on, dressing, scattering or pouring - which are to be selected to suit the intended aims of the prevailing circumstances - and the use of the compositions for controlling pests of the abovementioned type are other subjects of the invention.
- Typical rates of concentration are between 0.1 and 1000 ppm, preferably between 0.1 and 500 ppm, of active ingredient.
- the rate of application per hectare is generally 1 to 2000 g of active ingredient per hectare, in particular 10 to 1000 g/ha, preferably 10 to 600 g/ha.
- a preferred method of application in the field of crop protection is application to the foliage of the plants (foliar application), it being possible to select frequency and rate of application to match the danger of infestation with the pest in question.
- the active ingredient can reach the plants via the root system (systemic action), by drenching the locus of the plants with a liquid composition or by incorporating the active ingredient in solid form into the locus of the plants, for example into the soil, for example in the form of granules (soil application). In the case of paddy rice crops, such granules can be metered into the flooded paddy-field.
- the compounds of the invention and compositions thereof are also be suitable for the protection of plant propagation material, for example seeds, such as fruit, tubers or kernels, or nursery plants, against pests of the abovementioned type.
- the propagation material can be treated with the compound prior to planting, for example seed can be treated prior to sowing.
- the compound can be applied to seed kernels (coating), either by soaking the kernels in a liquid composition or by applying a layer of a solid composition. It is also possible to apply the compositions when the propagation material is planted to the site of application, for example into the seed furrow during drilling.
- These treatment methods for plant propagation material and the plant propagation material thus treated are further subjects of the invention.
- Typical treatment rates would depend on the plant and pest/fungi to be controlled and are generally between 1 to 200 grams per 100 kg of seeds, preferably between 5 to 150 grams per 100 kg of seeds, such as between 10 to 100 grams per 100 kg of seeds.
- seed embraces seeds and plant propagules of all kinds including but not limited to true seeds, seed pieces, suckers, corns, bulbs, fruit, tubers, grains, rhizomes, cuttings, cut shoots and the like and means in a preferred embodiment true seeds.
- the present invention also comprises seeds coated or treated with or containing a compound of formula I.
- coated or treated with and/or containing generally signifies that the active ingredient is for the most part on the surface of the seed at the time of application, although a greater or lesser part of the ingredient may penetrate into the seed material, depending on the method of application.
- the seed product When the said seed product is (re)planted, it may absorb the active ingredient.
- the present invention makes available a plant propagation material adhered thereto with a compound of formula (I). Further, it is hereby made available, a composition comprising a plant propagation material treated with a compound of formula (I).
- Seed treatment comprises all suitable seed treatment techniques known in the art, such as seed dressing, seed coating, seed dusting, seed soaking and seed pelleting.
- the seed treatment application of the compound formula (I) can be carried out by any known methods, such as spraying or by dusting the seeds before sowing or during the sowing/planting of the seeds.
- Example B1 Diabrotica balteata (Corn root worm)
- Maize sprouts placed onto an agar layer in 24-well microtiter plates were treated with aqueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions by spraying. After drying, the plates were infested with L2 larvae (6 to 10 per well). The samples were assessed for mortality and growth inhibition in comparison to untreated samples 4 days after infestation.
- the following compounds gave an effect of at least 80% in at least one of the two categories (mortality or growth inhibition) at an application rate of 200 ppm:
- Example B2 Euschistus heros (Neotropical Brown Stink Bug)
- Soybean leaves on agar in 24-well microtiter plates were sprayed with aqueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions. After drying the leaves were infested with N2 nymphs. The samples were assessed for mortality and growth inhibition in comparison to untreated samples 5 days after infestation.
- Example B3 Frankliniella occidentalis (Western flower thrips)
- Sunflower leaf discs were placed on agar in 24-well microtiter plates and sprayed with aqueous test solutions prepared from 10 ⁇ 00 DMSO stock solutions. After drying the leaf discs were infested with a Frankliniella population of mixed ages. The samples were assessed for mortality 7 days after infestation.
- Example B4 Mvzus persicae (Green peach aphid)
- Sunflower leaf discs were placed onto agar in a 24-well microtiter plate and sprayed with aqueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions. After drying, the leaf discs were infested with an aphid population of mixed ages. The samples were assessed for mortality 6 days after infestation.
- Example B5 Plutella xylostella (Diamond back moth)
- 24-well microtiter plates with artificial diet were treated with aqueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions by pipetting. After drying, the plates were infested with L2 larvae (10 to 15 per well). The samples were assessed for mortality and growth inhibition in comparison to untreated samples 5 days after infestation.
- Example B6 Spodoptera littoralis (Egyptian cotton leaf worm)
- Cotton leaf discs were placed onto agar in 24-well microtiter plates and sprayed with aqueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions. After drying the leaf discs were infested with five L1 larvae. The samples were assessed for mortality, anti-feeding effect, and growth inhibition in comparison to untreated samples 3 days after infestation. Control of Spodoptera littoralis by a test sample is given when at least one of the categories mortality, anti-feedant effect, and growth inhibition is higher than the untreated sample.
- Example B7 Spodoptera littoralis (Egyptian cotton leaf worm)
- Test compounds were applied by pipette from 10 ⁇ 00 ppm DMSO stock solutions into 24-well plates and mixed with agar. Lettuce seeds were placed onto the agar and the multi well plate was closed by another plate which contained also agar. After 7 days the compound was absorbed by the roots and the lettuce grew into the lid plate. The lettuce leaves were then cut off into the lid plate. Spodoptera eggs were pipetted through a plastic stencil onto a humid gel blotting paper and the lid plate was closed with it. The samples were assessed for mortality, anti-feedant effect and growth inhibition in comparison to untreated samples 6 days after infestation.
- Example B8 Tetranychus urticae (Two-spotted spider mite)
- Bean leaf discs on agar in 24-well microtiter plates were sprayed with agueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions. After drying the leaf discs were infested with a mite population of mixed ages. The samples were assessed for mortality on mixed population (mobile stages) 8 days after infestation.
- Example B9 Aedes aegypti (Yellow fever mosguito)
- Test solutions at an application rate of 200ppm in ethanol, were applied to 12 well tissue culture plates. Once the deposits were dry, five, two to five day old adult female Aedes aegypti were added to each well, and sustained with a 10% sucrose solution in a cotton wool plug. Assessment of knockdown was made one hour after introduction, and mortality was assessed at 24 and 48 hours after introduction.
- Example B10 Anopheles stephensi (Indian malaria mosquito)
- Test solutions at an application rate of 200 ppm in ethanol, were applied to 12 well tissue culture plates. Once the deposits were dry, five, two to five day old adult female Anopheles stephensi were added to each well, and sustained with a 10% sucrose solution in a cotton wool plug. Assessment of knockdown was made one hour after introduction, and mortality was assessed at 24 and 48 hours after introduction.
- Example B1 1 Bemisia tabaci (Cotton white fly)
- Cotton leaf discs were placed on agar in 24-well microtiter plates and sprayed with aqueous test solutions prepared from 10 ⁇ 00 ppm DMSO stock solutions. After drying the leaf discs were infested with adult white flies. The samples were checked for mortality 6 days after incubation.
- Compounds were assessed for at least 80% mortality at an application rate of 200 ppm.
- Example B12 Mvzus persicae (Green peach aphid)
- Roots of pea seedlings infested with an aphid population of mixed ages were placed directly into aqueous test solutions prepared from 10 ⁇ 00 DMSO stock solutions. The samples were assessed for mortality 6 days after placing seedlings into test solutions.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Polycyclic compounds of formula (I), wherein the substituents are as defined in claim 1, and the agrochemically acceptable salts, stereoisomers, enantiomers, tautomers and N-oxides of those compounds, can be used as insecticides and can be prepared in a manner known per se.
Description
Pesticidally active polycyclic derivatives with sulfur containing substituents
The present invention relates to pesticidally active, in particular insecticidally active polycyclic derivatives containing sulfur substituents, to intermediates for the preparation of those compounds, to compositions comprising those compounds, and to their use for controlling animal pests (including arthropods and in particular insects or representatives of the order Acarina).
Heterocyclic compounds with pesticidal action are known and described, for example, in
WO 2013/191 1 13, WO 2015/000715 and WO 2015/087458.
There have now been found novel pesticidally active polycyclic ring derivatives with sulfur containing phenyl and pyridyl substituents.
The present invention accordingly relates to compounds of formula I,
wherein
A is CH or N;
Q is phenyl which can be mono- or polysubstituted by substituents selected from the group consisting of halogen, cyano, Ci-C4alkyl, Ci-C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy, Ci-C4alkylsulfanyl, Ci- C4alkylsulfinyl, Ci-C4alkylsulfonyl, and Ci-C4haloalkylsulfanyl; or
Q is pyridyl or pyrimidyl which can be mono- or polysubstituted by substituents selected from the group consisting of halogen, cyano, Ci-C4alkyl, Ci-C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci- C4haloalkylsulfanyl; or
Q is pyrazolyl which is linked via a nitrogen atom to the ring which contains the substituent A, and which in turn can be substituted by halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl; or
Q is triazolyl which is linked via a nitrogen atom to the ring which contains the substituent A, and which in turn can be substituted by halogen, cyano or Ci-C4haloalkyl; or
X is S, SO or S02;
R4 is halogen, Ci-C4haloalkyl, Ci-C4haloalkylsulfanyl, Ci-C4haloalkylsulfinyl, or Ci-C4haloalkylsulfonyl; R5 is Ci-C4alkyl or C3-C6cycloalkyl-Ci-C4alkyl; and
Xi is CR6, wherein R6 is hydrogen, Ci-C4alkyl or halogen;
and agrochemically acceptable salts, stereoisomers, enantiomers, tautomers and N-oxides of those compounds.
Compounds of formula I which have at least one basic centre can form, for example, acid addition salts, for example with strong inorganic acids such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, a phosphorus acid or a hydrohalic acid, with strong organic carboxylic acids, such as Ci-C4alkanecarboxylic acids which are unsubstituted or substituted, for example by halogen, for example acetic acid, such as saturated or unsaturated dicarboxylic acids, for example oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid or phthalic acid, such as hydroxycarboxylic acids, for example ascorbic acid, lactic acid, malic acid, tartaric acid or citric acid, or such as benzoic acid, or with organic sulfonic acids, such as Ci-C4alkane- or arylsulfonic acids which are unsubstituted or substituted, for example by halogen, for example methane- or p-toluenesulfonic acid. Compounds of formula I which have at least one acidic group can form, for example, salts with bases, for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, diethyl-, triethyl- or
dimethylpropylamine, or a mono-, di- or trihydroxy-lower-alkylamine, for example mono-, di- or triethanolamine.
The alkyl groups occurring in the definitions of the substituents can be straight-chain or branched and are, for example, methyl, ethyl, n-propyl, isopropyl, n-butyl, sec-butyl, iso-butyl, tert-butyl, pentyl, hexyl, and their branched isomers. Alkylsulfanyl, alkylsulfinyl, alkylsulfonyl, alkoxy, alkenyl and alkynyl radicals are derived from the alkyl radicals mentioned. The alkenyl and alkynyl groups can be mono- or polyunsaturated. Ci-di-alkylamino is dimethylamino.
Halogen is generally fluorine, chlorine, bromine or iodine. This also applies, correspondingly, to halogen in combination with other meanings, such as haloalkyl or halophenyl.
HaloalkyI groups preferably have a chain length of from 1 to 6 carbon atoms. Haloalkyl is, for example, fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, 2,2,2- trifluoroethyl, 2-fluoroethyl, 2-chloroethyl, pentafluoroethyl, 1 , 1-difluoro-2,2,2-trichloroethyl, 2,2,3,3- tetrafluoroethyl and 2,2,2-trichloroethyl.
Haloalkoxy groups preferably have a chain length of from 1 to 4 carbon atoms. Haloalkoxy is, for example, difluoromethoxy, trifluoromethoxy or 2,2,2-trifluoroethoxy. Haloalkylsulfanyl groups preferably have a chain length of from 1 to 4 carbon atoms. Haloalkylsulfanyl is, for example, difluoromethylsulfanyl, trifluoromethylsulfanyl or 2,2,2-trifluoroethylsulfanyl. Similar considerations apply to the radicals Ci-C4haloalkylsulfinyl and Ci-C4haloalkylsulfonyl, which may be, for example, trifluoromethylsulfinyl, trifluoromethylsulfonyl or 2,2,2-trifluoroethylsulfonyl.
A triazolyl which is linked via a nitrogen atom to the ring which contains the substituent A is for example 1 ,2,4-triazol-1-yl, 1 ,2,4-triazol-4-yl, triazol-1-yl, or triazol-2-yl. Preferred is 1 ,2,4-triazol-1-yl. The cycloalkyl groups preferably have from 3 to 6 ring carbon atoms, for example cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
In the context of this invention "mono- to polysubstituted" in the definition of the substituents, means typically, depending on the chemical structure of the substituents, monosubstituted to three-times substituted, preferably monosubstituted to three-times substituted, more preferably mono-, or double- substituted.
The compounds of formula I according to the invention also include hydrates which may be formed during the salt formation.
A preferred group of compounds of formula I is represented by the compounds of formula 1-1
wherein F¾, Rs, A and Xi are as defined under formula I above; X is S, SO or SO2; and Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci-
C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy, Ci-C4alkylsulfanyl, Ci-C4alkylsulfinyl, Ci-C4alkylsulfonyl, and Ci-C4haloalkylsulfanyl.
In this preferred group of compounds of formula 1-1 , R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl, X is preferably SO2, Rs is preferably ethyl and Xi is preferably CH .
More highly preferred compounds of formula 1-1 are represented by compounds of formula 1-1 a:
wherein R4, Rs, and A are as defined under formula I above; and wherein Rx is independently selected from hydrogen or halogen. In this preferred group of compounds of formula l-1a, R4 is preferably Ci- C4haloalkyl and R5 is preferably ethyl.
More highly preferred compounds of formula l-1a are those in which R4 is CF3, Rs is ethyl, A is nitrogen and Rx is hydrogen or halogen, preferably fluorine or chlorine, with the proviso that at least one Rx is halogen.
A further preferred group of compounds of formula I is represented by the compounds of formula I-2:
wherein R4, Rs, A and Xi are as defined under formula I above; X is S, SO or SO2; and Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci-C4haloalkylsulfanyl.
In this preferred group of compounds of formula I-2, R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl, X is preferably SO2, Rs is preferably ethyl and Xi is preferably CH.
More highly preferred compounds of formula I-2 are represented by compounds of formula l-2a:
wherein R4, Rs, and A are as defined under formula I above; and wherein Rx is independently hydrogen or halogen. In this preferred group of compounds of formula l-1a, R4 is preferably Ci- C4haloalkyl and Rs is preferably ethyl. More highly preferred compounds of formula 1-1 a are those in which R4 is CF3, Rs is ethyl A is nitrogen and Rx is hydrogen or halogen, preferably fluorine or chlorine, with the proviso that at least one Rx is halogen. Most preferably both Rx are halogen, in particular fluorine.
wherein F¾, Rs, A and Xi are as defined under formula I above; X is S, SO or SO2; and Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci-C4haloalkylsulfanyl.
In this preferred group of compounds of formula I-3, R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl, X is preferably SO2, Rs is preferably ethyl and Xi is preferably CH.
More highly preferred compounds of formula I-3 are represented by compounds of formula l-3a:
wherein R4, Rs, and A are as defined under formula I above; and Rx is independently hydrogen or halogen. In this preferred group of compounds of formula l-3a, R4 is preferably Ci-C4haloalkyl and Rs is preferably ethyl, More highly preferred compounds of formula l-3a are those in which R4 is CF3, Rs is ethyl, A is nitrogen and Rx is hydrogen or halogen, preferably fluorine or chlorine, with the proviso that at least one Rx is halogen.
A further preferred group of compounds of formula I is represented by the compounds of formula I-4:
wherein R4, Rs, A and Xi are as defined under formula I above; and wherein X is S, SO or SO2; and Rxa is hydrogen, halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl.
ln this preferred group of compounds of formula I-4, F¾ is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl, X is preferably SO2, Rs is preferably ethyl and Xi is preferably CH.
More highly preferred compounds of formula I-4 are represented by compounds of formula l-4a:
wherein R4, Rs, and A are as defined under formula I above; and wherein Rxa is hydrogen, cyano, or Ci-C4haloalkyl. In this preferred group of compounds of formula l-4a, R4 is preferably Ci-C4haloalkyl and Rs is preferably ethyl.
More highly preferred compounds of formula l-4a are those in which R4 is CF3, Rs is ethyl, A is nitrogen and Rxa is hydrogen, cyano, or Ci-C4haloalkyl, preferably CF3 or cyano.
A further preferred group of compounds of formula I is represented by the compounds of formula I-5:
wherein R4, Rs, A and Xi are as defined under formula I above; and wherein X is S, SO or SO2; and Rxa is hydrogen, halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl.
In this preferred group of compounds of formula I-5, R4 is preferably Ci-C4haloalkyl or Ci- C4haloalkylsulfanyl, X is preferably SO2, Rs is preferably ethyl and Xi is preferably CH.
More highly preferred compounds of formula I-5 are re resented by compounds of formula l-5a:
wherein R4, Rs, and A are as defined under formula I above; and wherein Rxa is hydrogen, cyano, or halogen. In this preferred group of compounds of formula l-5a, R4 is preferably Ci-C4haloalkyl and Rs is preferably ethyl.
The process according to the invention for preparing compounds of formula I is carried out by methods known to those skilled in the art. Compounds of formula I, wherein F¾, Rs, X, Xi , A and Q are as defined in formula I, can be prepared (as shown in scheme 1 ) by a Suzuki reaction, which involves for example, reacting compounds of formula II, wherein Xbi is a leaving group like, for example, chlorine, bromine or iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate with compounds of formula Ilia, wherein Ybi can be a boron-derived functional group, as for example B(OH)2 or B(ORbi)2 wherein Rbi can be a Ci-C4alkyl group or the two groups ORbi can form together with the boron atom a five membered ring, as for example a pinacol boronic ester. The reaction can be catalyzed by a palladium based catalyst, for example fefra/c/'s(triphenylphosphine)-palladium or (1 ,1 'bis(diphenylphosphino)-ferrocene)dichloropalladium-dichloromethane (1 : 1 complex), in presence of a base, like sodium carbonate or cesium fluoride, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water or of dioxane and water, preferably under inert atmosphere. The reaction temperature can preferentially range from ambient temperature to the boiling point of the reaction mixture. Such Suzuki reactions are well known to those skilled in the art and have been reviewed, for example J.Orgmet. Chem. 576, 1999, 147-168.
Scheme 1 :
II lb
(Stille Reaction)
Alternatively compounds of formula I can be prepared by a Stille reaction of compounds of formula 1Mb wherein Yb2 is a trialkyl tin derivative, preferably tri-n-butyl tin, with compounds of formula II. Such Stille reactions are usually carried out in the presence of a palladium catalyst, for example
ieira/f/s(triphenylphosphine)palladium(0), or (1 ,1 'bis(diphenylphosphino)-ferrocene)dichloropalladium- dichloromethane (1 :1 complex), in an inert solvent such as DMF, acetonitrile, or dioxane, optionally in the presence of an additive, such as cesium fluoride, or lithium chloride, and optionally in the presence of a further catalyst, for example copper(l)iodide. Such Stille couplings are also well known to those skilled in the art, and have been described in for example J. Org. Chem. , 2005, 70, 8601-8604, J. Org. Chem. , 2009 , 74, 5599-5602, and Angew. Chem. Int. Ed., 2004, 43, 1 132-1 136.
Compounds of formula I wherein Q is a nitrogen bearing hetereocyclic system, and X, Xi , R4, Rs and A are as defined in formula I, can be prepared from compounds of formula II, wherein X, Xi , R4, Rs and A are as defined in formula I, and Xbi is a leaving group such as chlorine, bromine or iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate by reacting the hetreocycle Q (which contains a an appropriate NH functionality), in the presence of a base, such as K2CO3 or CS2CO3, optionally in
the presence of a copper catalyst, for example copper (I) iodide in an inert solvent such as N-methyl pyrollidione or DMF at temperatures between 30-150°C. The reaction is illustrated for the heterocycle Het-1 in scheme 2, which gives compounds of formula laa, wherein Ai is CRxa or N, and F¾, Rs, X, Xi , A and Rxa are as previously defined.
Scheme 2
Compounds of formula I can also be prepared (as depicted in scheme 3) by a Suzuki reaction as described above, which involves reacting compounds of formula IV with compounds of formula V, wherein Xb2 can be a halogen, preferentially chlorine, bromine or iodine, or a sulfonate, like for example a trifluoromethanesulfonate and Yb3 can be a boron-derived functional group, as for example B(OH)2 or B(ORb2)2 wherein Rb2 can be a C1-C4alkyl group or the two groups ORb2 can form together with the boron atom a five membered ring, as for example a pinacol boronic ester. In formula IV, A, Xi , R4, Rs, and X are as described in formula I. The reaction can be catalyzed by a palladium based catalyst, for example tetrakis(triphenylphosphine)-palladium, in presence of a base, like sodium carbonate, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water, preferably under inert atmosphere. The reaction temperature can preferentially range ambient temperature to the boiling point of the reaction mixture.
Scheme 3
VI
In a similar manner, compounds of formula I can be prepared by a Stille coupling (Scheme 3) of compounds of formula V with compounds of formula VI, wherein F¾, Rs, Xi , A, X are as described above, and Yb4 is a trialkyl tin derivative, preferably tri-n-butyl tin, under conditions described as in scheme 1.
Compounds of formula l-b3, wherein A, F¾, Rs, Xi and Q have the values defined in formula I, and X is -SO2-, can be prepared by oxidation of compounds of formula l-b2, wherein A, R4, Rs, and Xi have the values defined in formula I, and X is -SO- (as shown in scheme 4) The reaction can be performed with reagents like, for example, a peracid such as peracetic acid or m-chloroperbenzoic acid, or a hydroperoxide, such as for example, hydrogen peroxide or tert-butylhydroperoxide, or an inorganic oxidant, like a monoperoxo-disulfate salt or potassium permanganate. In a similar way, compounds of formula l-b2, wherein A, R4, Rs, and Xi have the values defined in formula I, and X is -SO- can be prepared by oxidation of compounds of formula l-b1 , wherein A, R4, Rs, Xi and Q have the values defined in formula I, and X is -S-, under analogous conditions described above. These reactions can be performed in various organic or aqueous solvents compatible to these conditions, by temperatures from below 0°C up to the boiling point of the solvent system. The transformation of compounds of the formula 1-b1 into compounds of the formula 1-b2 and 1-b3 is represented in scheme 4. The reactions can occur in a stepwise fashion through compounds of formula l-b2. Those skilled in the art will appreciate that is therefore possible to control the reaction (depending on amount of oxidant added, the temperature, and time of reaction) to allow isolation of compounds of formula l-b2.
Compounds of formula l-b1 may be prepared (scheme 4) by reacting a compound of the formula VII with a compound of the formula Villa, wherein A, R4, Rs, and Xi have the values defined in formula I and X is sulfur and M is a metal or non-metal cation. In scheme 4, the cation M is assumed to be monovalent, but polyvalent cations associated with more than one S-Rs group can also be considered. Preferred cations are, for example lithium, sodium, potassium or cesium. For this transformation to work, Xb3 is a leaving group like, for example, fluorine, chlorine, bromine or iodine, or an aryl- or alkylsulfonate, but many other leaving groups could be considered (for example NO2). The
reaction can be performed in a solvent, preferably aprotic, at temperatures below 0°C or up to boiling temperature of the reaction mixture.
Scheme 4
Compounds of formula VI I , wherein Xb3 is a leaving group like, for example, fluorine, chlorine, bromine iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate, or any other similar leaving group, can be prepared (scheme 5) by reacting compounds of formula V with compounds of formula VI I I, wherein Xb3 can be a halogen, preferentially chlorine, bromine or iodine, or a sulfonate, like for example a trifluoromethanesulfonate, most preferably bromine or iodine and Yb5 can be a boron- derived functional group, as for example B(OH)2 or B(ORb4)2 wherein Rb4 can be a Ci-C4alkyl group or the two groups ORb4 can form together with the boron atom a five membered ring, as for example a pinacol boronic ester. In formula VI II and V, A, Xi , R4 and Q are as described in formula I. The reaction can be catalyzed by a palladium based catalyst, for example tetrakis(triphenylphosphine)- palladium, in presence of a base, like sodium carbonate, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water, preferably under inert atmosphere. The reaction temperature can preferentially range from ambient temperature to the boiling point of the reaction mixture. In a similar manner, compounds of formula VII can be prepared from compounds of formula X, wherein A, Xi , R4 and Xb3 are as previously defined, and Yb6 is a trialkyl tin derivative, preferably tri- n-butyl tin, with compounds of formula V, under conditions described for those described for the chemistry illustrated in scheme 1 .
Scheme 5
In an alternative way, depicted in scheme 6, compounds of formula VII can also be prepared by reacting compounds of formula X, wherein Xb3 and Xb4 are leaving groups, for example, fluorine, chlorine, bromine or iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate, or any other similar leaving group, with compounds of formula Ilia (Suzuki reaction) or Illb (Stille reaction). The chemistry is carried out analogously to that discussed for scheme 1.
Scheme 6
χ Illb VII
(Stille Reaction)
A further route to prepare compounds of formula II, respectively Il-c1 , involves reaction of compounds of formula X with compounds of formula Villa as shown in scheme 7.
Scheme 7:
II X = -S02- II X = -SO- ln scheme 7, compounds of formula X are reacted with compounds of formula Villa to give compounds of formula Il-c1. Oxidation according the conditions described in scheme 4 (which depending on conditions known to those skilled in the art) will generate compounds of the formulas II- c2 and Il-c3. It is particularly preferred to have compounds of formula X with Xb3 is fluorine or nitro in such reactions to allow selective introduction of the group -SRs.
Compounds of formula I can be also prepared according to the chemistry shown in scheme 8:
ln scheme 8, compounds of formula XIII, wherein F¾ is as described in formula I, are reacted with compounds of formula XII, wherein Xb6 is a halogen and Q, X, A, Rs and R6 are as defined above, in an inert solvent, for example ethanol or acetonitrile, optionally in the presence of a suitable base at temperatures between 80-150 0 C, to give compounds of formula I. The reaction may optionally be carried out in a microwave optionally in a micro wave, to give compounds of formula I. Such reactions are well described in the literature, for example WO 2012/49280 or WO 03/031587.
A further process to prepare compounds of formula I, involves reacting a compound of formula XIII with a compound of formula XIV
In the presence of a Lewis acid, such as Zinc(ll)iodide or Indium(lll) triflate, in an inert solvent such as chlorobenzene or 1 ,2,dichlorobenzene, with a catalytic copper(ll) salt, such as Cu(ll)acetate, under an oxygen or air atmosphere at temperatures between 100-180 °C, preferably 1 10-140 °C, to give compounds of formula I wherein R6 is hydrogen. Such reactions have previously been described in the literature (see Adv. Synth. Catal. 2013, 355, 1741 - 1747, and J. Org. Chem., 2013 , 78 , 12494- 12504). Halogenation of compounds of formula I, wherein R6 is hydrogen, with a halogenating agent such as N-chlorosuccinamide, N-bromosuccinamide, or N-iodosuccinamide, in a polar aprotic solvent such as acetonitrile or dimethylformamide, at ambient temperature, leads to compounds of formula l-u
wherein Q, Rs, R4, X, and A are as described in formula (I), and Xb7 is halogen. Compounds of formula l-u can be reacted with compounds R6-Yb7, wherein Yb7 is a boron-derived functional group, as for
example B(OH)2 or B(ORb4)2 wherein Rb4 can be a Ci-C4alkyl group or the two groups ORb4 can form together with the boron atom a five membered ring, as for example a pinacol boronic ester, in the presence of a palladium catalyst to give compounds of formula wherein R4, R5, R6, A, X and Q are as defined as in formula I. The reaction is usually carried out in the presence of a base, for example potassium carbonate, cesium carbonate, or potassium phosphate, in an inert solvent, such as dioxane, optionally in the presence of water, with a palladium(O) catalyst, for example
tetrakis(triphenylphosphine)palladium, at a temperature between 80-120°C. Such Suzuki reactions are well precedented in the literature, see for example Masuda, Naoyuki ef al, WO 2012133607. The chemistry is illustrated in scheme 9
Scheme 9
Compounds of formula XII and XIV can be prepared from compounds of formula XVI by, for example, the methods shown in scheme 10.
Scheme 10
XII
ln scheme 10, an acyl halide of formula XVI (easily prepared from compounds of formula XV by methods known to those skilled in the art) is converted to a Weinreb amide XVII upon reaction with Λ/,Ο-Dimethylhydroxylamine by methods described for example in C. Ferri, "Reaktionen der
Organischen Synthese", Georg Thieme Verlag, Stuttgart, 1978, page 223ff. The Weinreb amide of formula XVII is then reacted with a Grignard reagent of formula ReChbMgHal according to the method of Weinreb (Tetrahedron Letters 1981 , 22, 3815-3818) to give compounds of formula XlVa and XIV. Compounds of formula XlVa and XIV can also be prepared by treatment of nitrile compounds of formula XVIII, wherein Q, X, Rs, and A are as described in formula I, with a Grignard reagent of formula ReChbMgHal, followed by acidic hydrolysis (as described in C. Ferri, "Reaktionen der Organischen Synthese", Georg Thieme Verlag, Stuttgart, 1978, page 223ff.).
Compounds of formula XlVa and XIV can be halogenated to compounds of formula XII, with for example mixtures of bromine and hydrobromic acid in acetic acid (as described in Phosphorus, Sulfur and Silicon and the Related Elements, 2013, 188(12), 1835-1844) or with, for example,
copper(ll)bromide in an inert solvent, for example chloroform, ethyl acetate and the like, as described in J Med. Chem., 2013, 56(1 ), 84-96. Alternatively, compounds of formula XII where R6 is hydrogen, can be prepared directly from compounds of formula XVI by treatment with diazomethane or trimethyl silyl diazomethane and subsequent treatment with an halogen acid, for example, hydrobromic acid or hydrochloric acid in an inert solvent such as diethyl ether. Such procedures are well known in the literature, for example see Eu. J. Med. Chem., 1987, 22(5), 457-62 and WO 2009010455.
Compounds of formula XV can be prepared (as shown in scheme 1 1 ) by ester hydrolysis of compounds of formula XX, wherein A, Xb3, and Rs are as previously defined, and R001 is Ci-C6alkyl, by methods known to those skilled in the art, for example by treatment with an alkaline earth metal base, such as lithium hydroxide, typically in water with sufficient miscible organic solvent, for example THF or acetone, to dissolve compounds of the formula XX. Compounds XX can be prepared by a Suzuki reaction, which involves for example, reacting compounds of formula XIX, wherein Xb3 is a leaving group like, for example, chlorine, bromine or iodine, or an aryl- or alkylsulfonate such as trifluoromethanesulfonate (especially preferred are those in which Xb1 is fluoro or bromo) with compounds of formula Ilia, wherein Ybi can be a boron-derived functional group, as for example B(OH)2 or B(ORbi)2 wherein Rbi can be a Ci-C4alkyl group or the two groups ORbi can form together with the boron atom a five membered ring, as for example a pinacol boronic ester. The reaction can be catalyzed by a palladium based catalyst, for example fefra/c/'s(triphenylphosphine)-palladium or (1 , 1 'bis(diphenylphosphino)-ferrocene)dichloropalladium-dichloromethane (1 : 1 complex), in presence of a base, like sodium carbonate or cesium fluoride, in a solvent or a solvent mixture, like, for example a mixture of 1 ,2-dimethoxyethane and water or of dioxane and water, preferably under inert atmosphere. The reaction temperature can preferentially range from ambient temperature to the boiling point of the reaction mixture. Such Suzuki reactions are well known to those skilled in the art and have been reviewed, for example J.Orgmet. Chem. 576, 1999, 147-168.
Scheme 1 1
H drol sis
Alternatively compounds of formula XX can be prepared by a Stille reaction of compounds of formula lllb wherein Yb2 is a trialkyl tin derivative, preferably tri-n-butyl tin, with compounds of formula XIX. Such Stille reactions are usually carried out in the presence of a palladium catalyst, for example ieira/f/s(triphenylphosphine)palladium(0), or (1 ,1 'bis(diphenylphosphino)-ferrocene)dichloropalladium- dichloromethane (1 : 1 complex), in an inert solvent such as DMF, acetonitrile, or dioxane, optionally in the presence of an additive, such as cesium fluoride, or lithium chloride, and optionally in the presence of a further catalyst, for example copper(l)iodide. Such Stille couplings are also well known to those skilled in the art, and have been described in for example J. Org. Chem. , 2005, 70, 8601-8604, J. Org. Chem. , 2009 , 74, 5599-5602, and Angew. Chem. Int. Ed., 2004, 43, 1 132-1 136.
In a very similar manner compounds of formula Xa can similarly be prepared as shown in scheme 12, using analogous procedures and strategies to those described in scheme 8.
The intermediates required to synthesize compounds of formula XXI, can be obtained analogously to the chemistry shown in scheme 10, and illustrated here again in scheme 13.
Scheme 13
e.g.
0 °C
temp,
A large number of compounds of the formula V are commercially available or can be prepared by those skilled in the art. Many chemical transformations, well known by those skilled in the art, can be used to access boronic acid derivatives of formula Ilia, starting from various and easily available starting materials, as for example, to cite only a few (scheme 14), hydrogen abstraction on a heteroaromatic compound of the formula V wherein Xb2 is hydrogen, with a strong base (step A), like butyl lithium or lithium diisopropylamide or (i-PrMgCI. LiCI), followed by reaction of the metalated intermediate of the formula 111 , wherein Zb∑ is a metal such as Li+ or MgCI+ for example, with, for example, a trialkylborate (step B), or a tri-n-butyl tin chloride (step B). Another way to access an organometal intermediate of the formulae Ilia or lllb is from a compound of the formula 111 which is obtained by via metal-halogen exchange of compound of formula V with an organometallic species (step C), using for example butyl lithium or an organ magnesium compound, or direct metalation with a metal, like magnesium.
Introduction of a pinacolborate functional group via a palladium catalyzed reaction with bispinacol diborane, or hexa-n-butyldistannane, on a compound of the formula V, wherein Xb2 c, is another common strategy (scheme 14, step D). In the compounds of formula Ilia, and lllb within scheme 14, Q has the values defined for the formula I. A person skilled in the art will be able to select an adequate preparation method to access compounds of formula Ilia and lllb depending on the values of Q.
Scheme 14
step C
Compounds of formula IV, wherein A, X, Xi, F¾ and R5 are as described in formula I, can be prepared from compounds of formula II (scheme 15), wherein A, X, Xi, R4 and R5 are as described in formula I. Indeed, compounds of formula II, wherein Xbi is chlorine, bromine or iodine, can be treated with an organometallic species like, for example, butyl lithium or an organomagnesium compound, to generate an intermediate compound of the formula ll-a, wherein Zb3 is as defined in the scheme, via metal- halogen exchange. This reaction is preferentially performed in an anhydrous aprotic solvent, such as THF, at low temperature (between -120°C and 0°C), preferentially between -1 10°C and -60°C). The intermediate organometal compound of formula ll-a is preferably directly converted into compound of formula IV by reaction with a boronate compound B(ORb2)3, wherein Rb2 is a C1-C4alkyl group.
Depending on nature of the boronate, the reaction treatment conditions and the workup conditions, the boronic acid IV, wherein Yb3 is -B(OH)2, or a dialkylboronate IV, wherein Yb3 is -B(ORb2)2, can be formed. Scheme 15
Introduction of a pinacolborate functional group via a palladium catalyzed reaction with bispinacol diborane on compound of the formula II, wherein Xb1 is chlorine, bromine, iodine or triflate, is another common strategy (scheme 15). In the compounds of formula II within scheme 15, A, F¾, Rs, X, and Xi, have the values defined for the formula I, and Xb1 is chlorine, bromine, fluorine, iodine or triflate. A person skilled in the art will be able to select an adequate preparation method to access compounds of formula Ma from II depending on the values A, F¾, Rs, X, and Xi.
In a similar fashion to the chemistry shown in scheme 15, compounds of formula VIII can be obtained from compounds of formula X (scheme 16).
Scheme 16
The very similar preparation methods described in schemes 15 and 16 may be applied for the synthesis of intermediates of the formula IX and VI, but in this case instead of using boronic compounds e.g. of formula B(ORb2)3, those skilled in the art would know to use a tin compound of formula (n-butyl)3SnCI (as described as for example in Eu. J. Chem., 4098-4104, 20, 2014) or instead of bispinacol diborane, the use of hexabutylditin (as described in for example Eur. Pat. Appl., 2749561 , 2014). This is illustrated for compound VI in scheme 17.
)3Sn-
Compounds of formula XIII wherein R4 is Ci-C2haloalkyl, Ci haloalkylsulfanyl, Ci haloalkylsulfinyl, Ci haloalkylsulfonyl, or C3-C6cycloalkyl can be prepared as shown in scheme 17.
Scheme 17.
R2-B(OH)2 (Suzuki)
or R2Sn(n-Bu)3 (Stille)
Nal/ HI
R2-B(OH)2 (Suzuki)
or R2Sn(n-Bu)3 (Stille)
Xllla Xllld Xllle
f¾ = CFs = Xlllb Xlllf n = 1 = Xlllg
f¾ = CF2CF3 = Xlllc n = 2 = Xlllh As shown in scheme 20, reaction of the known compound Xll la with (Phen)CuCF3 or
(Phen)CuCF2CF3 in an inert solvent, such as DMF or NMP, at temperatures between 50-120 °C leads to compounds of formula Xll lb and Xl l lc, respectively. Such reactions are well precedented in the literature, see for example, Angew. Chem. Int. Ed. 2011, 50, 3793 and Org. Lett. 2014, 16, 1744 (R4 is
CF3), and Angew. Chem. Int. Ed. 2012, 51, 536 (F is CF2CF3). Compounds of formula Xllla can be converted to compounds of formula Xllld by treatment with hydroiodic acid, optionaly in the presence of sodium iodide, according to those skilled in the art and as described for example in Bio.
Med. Chem., 15(4), 1586-1605, 2007. Reaction of compounds of formula Xllld with (bpy)CuSCF3 in an inert solvent, such as DMF or NMP, at temperatures between 50-120 °C leads to compounds of formula XI I I . Such reactions are precedented in the literature, for example in Angew. Chem. Int. Ed. 2013, 52, 1548-1552. Compounds of formula XI I If can be further oxidized to compounds of formula XII Ig and XI 11 h by oxidation, for example with MCPBA or other methods known to those skilled in the art. Compounds of formula I where R4 is Ci-C2haloalkyl or Ci-Chaloalkylsulfanyl, i.e. compounds of formula lac and lad respectively, can be prepared from compounds of formula lab, wherein in R4 is halogen, preferably bromine or iodine, by the same chemistry described for the preparation of XII lb, XI lie and XI I If . This is illustrated in scheme 18.
, X = S, SO, or S02 X = S, SO, or SO2
n = or
Compounds of formula lae, wherein A, R5, Xi and Q have the values defined in formula I, X is -S-, -SO-, or -SO2-, and n is 1 or 2, can be prepared by oxidation of compounds of formula lad, wherein A, R5, Q and Xi have the values defined in formula I, and X is -S-, -SO-, or -SO2-. (as shown in scheme 18). The reaction can be performed with reagents like, for example, a peracid such as peracetic acid or m-chloroperbenzoic acid, or a hydroperoxide, such as for example, hydrogen peroxide or tert- butylhydroperoxide, or an inorganic oxidant, like a monoperoxo-disulfate salt or potassium permanganate. A person skilled in the art will realize that by varying the number of equivalents of the
oxidising agent, all combinations of compounds of formula lae with n is 1 or 2, and X is -S-, -SO-, or - SO2- can be obtained.
The reactants can be reacted in the presence of a base. Examples of suitable bases are alkali metal or alkaline earth metal hydroxides, alkali metal or alkaline earth metal hydrides, alkali metal or alkaline earth metal amides, alkali metal or alkaline earth metal alkoxides, alkali metal or alkaline earth metal acetates, alkali metal or alkaline earth metal carbonates, alkali metal or alkaline earth metal dialkylamides or alkali metal or alkaline earth metal alkylsilylamides, alkylamines, alkylenediamines, free or N-alkylated saturated or unsaturated cycloalkylamines, basic heterocycles, ammonium hydroxides and carbocyclic amines. Examples which may be mentioned are sodium hydroxide, sodium hydride, sodium amide, sodium methoxide, sodium acetate, sodium carbonate, potassium tert- butoxide, potassium hydroxide, potassium carbonate, potassium hydride, lithium diisopropylamide, potassium bis(trimethylsilyl)amide, calcium hydride, triethylamine, diisopropylethylamine,
triethylenediamine, cyclohexylamine, N-cyclohexyl-N,N-dimethylamine, Ν,Ν-diethylaniline, pyridine, 4- (N,N-dimethylamino)pyridine, quinuclidine, N-methylmorpholine, benzyltrimethylammonium hydroxide and 1 ,8-diazabicyclo[5.4.0]undec-7-ene (DBU).
The reactants can be reacted with each other as such, i.e. without adding a solvent or diluent. In most cases, however, it is advantageous to add an inert solvent or diluent or a mixture of these. If the reaction is carried out in the presence of a base, bases which are employed in excess, such as triethylamine, pyridine, N-methylmorpholine or Ν,Ν-diethylaniline, may also act as solvents or diluents.
The reaction is advantageously carried out in a temperature range from approximately -80°C to approximately +140°C, preferably from approximately -30°C to approximately +100°C, in many cases in the range between ambient temperature and approximately +80°C.
A compound of formula I can be converted in a manner known per se into another compound of formula I by replacing one or more substituents of the starting compound of formula I in the customary manner by (an)other substituent(s) according to the invention.
Depending on the choice of the reaction conditions and starting materials which are suitable in each case, it is possible, for example, in one reaction step only to replace one substituent by another substituent according to the invention, or a plurality of substituents can be replaced by other substituents according to the invention in the same reaction step.
Salts of compounds of formula I can be prepared in a manner known per se. Thus, for example, acid addition salts of compounds of formula I are obtained by treatment with a suitable acid or a suitable
ion exchanger reagent and salts with bases are obtained by treatment with a suitable base or with a suitable ion exchanger reagent.
Salts of compounds of formula I can be converted in the customary manner into the free compounds I, acid addition salts, for example, by treatment with a suitable basic compound or with a suitable ion exchanger reagent and salts with bases, for example, by treatment with a suitable acid or with a suitable ion exchanger reagent.
Salts of compounds of formula I can be converted in a manner known per se into other salts of compounds of formula I, acid addition salts, for example, into other acid addition salts, for example by treatment of a salt of inorganic acid such as hydrochloride with a suitable metal salt such as a sodium, barium or silver salt, of an acid, for example with silver acetate, in a suitable solvent in which an inorganic salt which forms, for example silver chloride, is insoluble and thus precipitates from the reaction mixture.
Depending on the procedure or the reaction conditions, the compounds of formula I, which have salt- forming properties, can be obtained in free form or in the form of salts.
The compounds of formula I and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can be present in the form of one of the isomers which are possible or as a mixture of these, for example in the form of pure isomers, such as antipodes and/or diastereomers, or as isomer mixtures, such as enantiomer mixtures, for example racemates, diastereomer mixtures or racemate mixtures, depending on the number, absolute and relative configuration of asymmetric carbon atoms which occur in the molecule and/or depending on the configuration of non-aromatic double bonds which occur in the molecule; the invention relates to the pure isomers and also to all isomer mixtures which are possible and is to be understood in each case in this sense hereinabove and hereinbelow, even when stereochemical details are not mentioned specifically in each case.
Diastereomer mixtures or racemate mixtures of compounds of formula I, in free form or in salt form, which can be obtained depending on which starting materials and procedures have been chosen can be separated in a known manner into the pure diasteromers or racemates on the basis of the physicochemical differences of the components, for example by fractional crystallization, distillation and/or chromatography. Enantiomer mixtures, such as racemates, which can be obtained in a similar manner can be resolved into the optical antipodes by known methods, for example by recrystallization from an optically active solvent, by chromatography on chiral adsorbents, for example high-performance liquid
chromatography (HPLC) on acetyl cellulose, with the aid of suitable microorganisms, by cleavage with
specific, immobilized enzymes, via the formation of inclusion compounds, for example using chiral crown ethers, where only one enantiomer is complexed, or by conversion into diastereomeric salts, for example by reacting a basic end-product racemate with an optically active acid, such as a carboxylic acid, for example camphor, tartaric or malic acid, or sulfonic acid, for example camphorsulfonic acid, and separating the diastereomer mixture which can be obtained in this manner, for example by fractional crystallization based on their differing solubilities, to give the diastereomers, from which the desired enantiomer can be set free by the action of suitable agents, for example basic agents.
Pure diastereomers or enantiomers can be obtained according to the invention not only by separating suitable isomer mixtures, but also by generally known methods of diastereoselective or
enantioselective synthesis, for example by carrying out the process according to the invention with starting materials of a suitable stereochemistry.
N-oxides can be prepared by reacting a compound of the formula I with a suitable oxidizing agent, for example the hbCh/urea adduct in the presence of an acid anhydride, e.g. trifluoroacetic anhydride. Such oxidations are known from the literature, for example from J. Med. Chem., 32 (12), 2561-73, 1989 or WO 00/15615.
It is advantageous to isolate or synthesize in each case the biologically more effective isomer, for example enantiomer or diastereomer, or isomer mixture, for example enantiomer mixture or diastereomer mixture, if the individual components have a different biological activity.
The compounds of formula I and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form. The compounds of formula I according to the invention are preventively and/or curatively valuable active ingredients in the field of pest control, even at low rates of application, which have a very favourable biocidal spectrum and are well tolerated by warm-blooded species, fish and plants. The active ingredients according to the invention act against all or individual developmental stages of normally sensitive, but also resistant, animal pests, such as insects or representatives of the order Acarina. The insecticidal or acaricidal activity of the active ingredients according to the invention can manifest itself directly, i. e. in destruction of the pests, which takes place either immediately or only after some time has elapsed, for example during ecdysis, or indirectly, for example in a reduced oviposition and/or hatching rate. Examples of the abovementioned animal pests are:
from the order Acarina, for example,
Acalitus spp, Aculus spp, Acaricalus spp, Aceria spp, Acarus siro, Amblyomma spp., Argas spp., Boophilus spp., Brevipalpus spp., Bryobia spp, Calipitrimerus spp., Chorioptes spp., Dermanyssus
gallinae, Dermatophagoides spp, Eotetranychus spp, Eriophyes spp., Hemitarsonemus spp,
Hyalomma spp., Ixodes spp., Olygonychus spp, Ornithodoros spp., Polyphagotarsone latus,
Panonychus spp., Phyllocoptruta oleivora, Phytonemus spp, Polyphagotarsonemus spp, Psoroptes spp., Rhipicephalus spp., Rhizoglyphus spp., Sarcoptes spp., Steneotarsonemus spp, Tarsonemus spp. and Tetranychus spp.;
from the order Anoplura, for example,
Haematopinus spp., Linognathus spp., Pediculus spp., Pemphigus spp. and Phylloxera spp.;
from the order Coleoptera, for example,
Agriotes spp., Amphimallon majale, Anomala orientalis, Anthonomus spp., Aphodius spp, Astylus atromaculatus, Ataenius spp, Atomaria linearis, Chaetocnema tibialis, Cerotoma spp, Conoderus spp, Cosmopolites spp., Cotinis nitida, Curculio spp., Cyclocephala spp, Dermestes spp., Diabrotica spp., Diloboderus abderus, Epilachna spp., Eremnus spp., Heteronychus arator, Hypothenemus hampei, Lagria vilosa, Leptinotarsa decemLineata, Lissorhoptrus spp., Liogenys spp, Maecolaspis spp, Maladera castanea, Megascelis spp, Melighetes aeneus, Melolontha spp., Myochrous armatus, Orycaephilus spp., Otiorhynchus spp., Phyllophaga spp, Phlyctinus spp., Popillia spp., Psylliodes spp., Rhyssomatus aubtilis, Rhizopertha spp., Scarabeidae, Sitophilus spp., Sitotroga spp., Somaticus spp, Sphenophorus spp, Sternechus subsignatus, Tenebrio spp., Tribolium spp. and Trogoderma spp.; from the order Diptera, for example,
Aedes spp., Anopheles spp, Antherigona soccata,Bactrocea oleae, Bibio hortulanus, Bradysia spp, Calliphora erythrocephala, Ceratitis spp., Chrysomyia spp., Culex spp., Cuterebra spp., Dacus spp., Delia spp, Drosophila melanogaster, Fannia spp., Gastrophilus spp., Geomyza tripunctata, Glossina spp., Hypoderma spp., Hyppobosca spp., Liriomyza spp., Lucilia spp., Melanagromyza spp., Musca spp., Oestrus spp., Orseolia spp., Oscinella frit, Pegomyia hyoscyami, Phorbia spp., Rhagoletis spp, Rivelia quadrifasciata, Scatella spp, Sciara spp., Stomoxys spp., Tabanus spp., Tannia spp. and Tipula spp.;
from the order Hemiptera, for example,
Acanthocoris scabrator, Acrosternum spp, Adelphocoris lineolatus, Amblypelta nitida, Bathycoelia thalassina, Blissus spp, Cimex spp., Clavigralla tomentosicollis, Creontiades spp, Distantiella theobroma, Dichelops furcatus, Dysdercus spp., Edessa spp, Euchistus spp., Eurydema pulchrum, Eurygaster spp., Halyomorpha halys, Horcias nobilellus, Leptocorisa spp., Lygus spp, Margarodes spp, Murgantia histrionic, Neomegalotomus spp, Nesidiocoris tenuis, Nezara spp., Nysius simulans, Oebalus insularis, Piesma spp., Piezodorus spp, Rhodnius spp., Sahlbergella singularis, Scaptocoris castanea, Scotinophara spp., Thyanta spp , Triatoma spp., Vatiga illudens;
Acyrthosium pisum, Adalges spp, Agalliana ensigera, Agonoscena targionii, Aleurodicus spp, Aleurocanthus spp, Aleurolobus barodensis, Aleurothrixus floccosus, Aleyrodes brassicae, Amarasca biguttula, Amritodus atkinsoni, Aonidiella spp., Aphididae, Aphis spp., Aspidiotus spp., Aulacorthum solani, Bactericera cockerelli, Bemisia spp, Brachycaudus spp, Brevicoryne brassicae, Cacopsylla spp, Cavariella aegopodii Scop., Ceroplaster spp., Chrysomphalus aonidium, Chrysomphalus
dictyospermi, Cicadella spp, Cofana spectra, Cryptomyzus spp, Cicadulina spp, Coccus hesperidum, Dalbulus maidis, Dialeurodes spp, Diaphorina citri, Diuraphis noxia, Dysaphis spp, Empoasca spp., Eriosoma larigerum, Erythroneura spp., Gascardia spp., Glycaspis brimblecombei, Hyadaphis pseudobrassicae, Hyalopterus spp, Hyperomyzus pallidus, Idioscopus clypealis, Jacobiasca lybica, Laodelphax spp., Lecanium corni, Lepidosaphes spp., Lopaphis erysimi, Lyogenys maidis,
Macrosiphum spp., Mahanarva spp, Metcalfa pruinosa, Metopolophium dirhodum, Myndus crudus, Myzus spp., Neotoxoptera sp, Nephotettix spp., Nilaparvata spp., Nippolachnus piri Mats, Odonaspis ruthae, Oregma lanigera Zehnter, Parabemisia myricae, Paratrioza cockerelli, Parlatoria spp., Pemphigus spp., Peregrinus maidis, Perkinsiella spp, Phorodon humuli, Phylloxera spp, Planococcus spp., Pseudaulacaspis spp., Pseudococcus spp., Pseudatomoscelis seriatus, Psylla spp., Pulvinaria aethiopica, Quadraspidiotus spp., Quesada gigas, Recilia dorsalis, Rhopalosiphum spp., Saissetia spp., Scaphoideus spp., Schizaphis spp., Sitobion spp., Sogatella furcifera, Spissistilus festinus, Tarophagus Proserpina, Toxoptera spp, Trialeurodes spp, Tridiscus sporoboli, Trionymus spp, Trioza erytreae , Unaspis citri, Zygina flammigera, Zyginidia scutellaris, ;
from the order Hymenoptera, for example,
Acromyrmex, Arge spp, Atta spp., Cephus spp., Diprion spp., Diprionidae, Gilpinia polytoma, Hoplo- campa spp., Lasius spp., Monomorium pharaonis, Neodiprion spp., Pogonomyrmex spp, Slenopsis invicta, Solenopsis spp. and Vespa spp.;
from the order Isoptera, for example,
Coptotermes spp, Corniternes cumulans, Incisitermes spp, Macrotermes spp, Mastotermes spp, Microtermes spp, Reticulitermes spp.; Solenopsis geminate
from the order Lepidoptera, for example,
Acleris spp., Adoxophyes spp., Aegeria spp., Agrotis spp., Alabama argillaceae, Amylois spp., Anticarsia gemmatalis, Archips spp., Argyresthia spp, Argyrotaenia spp., Autographa spp., Bucculatrix thurberiella, Busseola fusca, Cadra cautella, Carposina nipponensis, Chilo spp., Choristoneura spp., Chrysoteuchia topiaria, Clysia ambiguella, Cnaphalocrocis spp., Cnephasia spp., Cochylis spp., Coleophora spp., Colias lesbia, Cosmophila flava, Crambus spp, Crocidolomia binotalis, Cryptophlebia leucotreta, Cydalima perspectalis, Cydia spp., Diaphania perspectalis, Diatraea spp., Diparopsis castanea, Earias spp., Eldana saccharina, Ephestia spp., Epinotia spp, Estigmene acrea, Etiella zinckinella, Eucosma spp., Eupoecilia ambiguella, Euproctis spp., Euxoa spp., Feltia jaculiferia, Gra- pholita spp., Hedya nubiferana, Heliothis spp., Hellula undalis, Herpetogramma spp, Hyphantria cunea, Keiferia lycopersicella, Lasmopalpus lignosellus, Leucoptera scitella, Lithocollethis spp., Lobesia botrana, Loxostege bifidalis, Lymantria spp., Lyonetia spp., Malacosoma spp., Mamestra brassicae, Manduca sexta, Mythimna spp, Noctua spp, Operophtera spp., Orniodes indica, Ostrinia nubilalis, Pammene spp., Pandemis spp., Panolis flammea, Papaipema nebris, Pectinophora gossypi- ela, Perileucoptera coffeella, Pseudaletia unipuncta, Phthorimaea operculella, Pieris rapae, Pieris spp., Plutella xylostella, Prays spp., Pseudoplusia spp, Rachiplusia nu, Richia albicosta, Scirpophaga
spp., Sesamia spp., Sparganothis spp., Spodoptera spp., Sylepta derogate, Synanthedon spp., Thaumetopoea spp., Tortrix spp., Trichoplusia ni, Tuta absoluta, and Yponomeuta spp.;
from the order Mallophaga, for example,
Damalinea spp. and Trichodectes spp.;
from the order Orthoptera, for example,
Blatta spp., Blattella spp., Gryllotalpa spp., Leucophaea maderae, Locusta spp., Neocurtilla hexadactyla, Periplaneta spp. , Scapteriscus spp, and Schistocerca spp.;
from the order Psocoptera, for example,
Liposcelis spp.;
from the order Siphonaptera, for example,
Ceratophyllus spp., Ctenocephalides spp. and Xenopsylla cheopis; from the order Thysanoptera, for example,
Calliothrips phaseoli, Frankliniella spp., Heliothrips spp, Hercinothrips spp., Parthenothrips spp, Scirtothrips aurantii, Sericothrips variabilis, Taeniothrips spp., Thrips spp;
from the order Thysanura, for example, Lepisma saccharina.
The active ingredients according to the invention can be used for controlling, i. e. containing or destroying, pests of the abovementioned type which occur in particular on plants, especially on useful plants and ornamentals in agriculture, in horticulture and in forests, or on organs, such as fruits, flowers, foliage, stalks, tubers or roots, of such plants, and in some cases even plant organs which are formed at a later point in time remain protected against these pests.
Suitable target crops are, in particular, cereals, such as wheat, barley, rye, oats, rice, maize or sorghum; beet, such as sugar or fodder beet; fruit, for example pomaceous fruit, stone fruit or soft fruit, such as apples, pears, plums, peaches, almonds, cherries or berries, for example strawberries, raspberries or blackberries; leguminous crops, such as beans, lentils, peas or soya; oil crops, such as oilseed rape, mustard, poppies, olives, sunflowers, coconut, castor, cocoa or ground nuts; cucurbits, such as pumpkins, cucumbers or melons; fibre plants, such as cotton, flax, hemp or jute; citrus fruit, such as oranges, lemons, grapefruit or tangerines; vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes or bell peppers; Lauraceae, such as avocado, Cinnamonium or camphor; and also tobacco, nuts, coffee, eggplants, sugarcane, tea, pepper, grapevines, hops, the plantain family and latex plants. The compositions and/or methods of the present invention may be also used on any ornamental and/or vegetable crops, including flowers, shrubs, broad-leaved trees and evergreens.
For example the invention may be used on any of the following ornamental species: Ageratum spp., Alonsoa spp., Anemone spp., Anisodontea capsenisis, Anthemis spp., Antirrhinum spp., Aster spp.,
Begonia spp. (e.g. B. elatior, B. semperflorens, B. tubereux), Bougainvillea spp., Brachycome spp., Brassica spp. (ornamental), Calceolaria spp., Capsicum annuum, Catharanthus roseus, Canna spp., Centaurea spp., Chrysanthemum spp., Cineraria spp. (C. maritime), Coreopsis spp., Crassula coccinea, Cuphea ignea, Dahlia spp., Delphinium spp., Dicentra spectabilis, Dorotheantus spp., Eustoma grandiflorum, Forsythia spp., Fuchsia spp., Geranium gnaphalium, Gerbera spp.,
Gomphrena globosa, Heliotropium spp., Helianthus spp., Hibiscus spp., Hortensia spp., Hydrangea spp., Hypoestes phyllostachya, Impatiens spp. (/. Walleriana), Iresines spp., Kalanchoe spp., Lantana camara, Lavatera trimestris, Leonotis leonurus, Lilium spp., Mesembryanthemum spp., Mimulus spp., Monarda spp., Nemesia spp., Tagetes spp., Dianthus spp. (carnation), Canna spp., Oxalis spp., Be//;'s spp., Pelargonium spp. CP. peltatum, P. Zonale), Viola spp. (pansy), Petunia spp., PWox spp.,
Plecthranthus spp., Poinsettia spp., Parthenocissus spp. CP- quinquefolia, P. tricuspidata), Primula spp., Ranunculus spp., Rhododendron spp., Rosa spp. (rose), Rudbeckia spp., Saintpaulia spp., Sa/w'a spp., Scaevola aemola, Schizanthus wisetonensis, Sedum spp., Solanum spp., Surfinia spp., Tagetes spp., Nicotinia spp., Verbena spp., Zinnia spp. and other bedding plants.
For example the invention may be used on any of the following vegetable species: Allium spp. (A sativum, A., cepa, A. oschaninii, A. Porrum, A. ascalonicum, A. fistulosum), Anthriscus cerefolium, Apium graveolus, Asparagus officinalis, Beta vulgarus, Brassica spp. (B. Oleracea, B. Pekinensis, B. rapa), Capsicum annuum, Cicer arietinum, Cichorium endivia, Cichorum spp. (C. intybus, C. endivia), Citrillus lanatus, Cucumis spp. (C. sativus, C. melo), Cucurbita spp. (C. pepo, C. maxima), Cyanara spp. (C. scolymus, C. cardunculus), Daucus carota, Foeniculum vulgare, Hypericum spp., Lactuca sativa, Lycopersicon spp. (L esculentum, L. lycopersicum), Mentha spp., Ocimum basilicum, Petroselinum crispum, Phaseolus spp. (P. vulgaris, P. coccineus), Pisum sativum, Raphanus sativus, Rheum rhaponticum, Rosemarinus spp., Salvia spp., Scorzonera hispanica, Solanum melongena, Spinacea oleracea, Valerianella spp. (V. locusta, V. eriocarpa) and V/c/a /aba.
Preferred ornamental species include African violet, Begonia, Dahlia, Gerbera, Hydrangea, Verbena, Rosa, Kalanchoe, Poinsettia, Aster, Centaurea, Coreopsis, Delphinium, Monarda, Phlox, Rudbeckia, Sedum, Petunia, Viola, Impatiens, Geranium, Chrysanthemum, Ranunculus, Fuchsia, Salvia, Hortensia, rosemary, sage, St. Johnswort, mint, sweet pepper, tomato and cucumber. The active ingredients according to the invention are especially suitable for controlling Aphis craccivora, Diabrotica balteata, Heliothis virescens, Myzus persicae, Plutella xylostella and
Spodoptera littoralis in cotton, vegetable, maize, rice and soya crops. The active ingredients according to the invention are further especially suitable for controlling Mamestra (preferably in vegetables), Cydia pomonella (preferably in apples), Empoasca(preferably in vegetables, vineyards), Leptinotarsa (preferably in potatos) and Chilo supressalis (preferably in rice).
In a further aspect, the invention may also relate to a method of controlling damage to plant and parts thereof by plant parasitic nematodes (Endoparasitic-, Semiendoparasitic- and Ectoparasitic
nematodes), especially plant parasitic nematodes such as root knot nematodes, Meloidogyne hapla, Meloidogyne incognita, Meloidogyne javanica, Meloidogyne arenaria and other Meloidogyne species; cyst-forming nematodes, Globodera rostochiensis and other Globodera species; Heterodera avenae, Heterodera glycines, Heterodera schachtii, Heterodera trifolii, and other Heterodera species; Seed gall nematodes, Anguina species; Stem and foliar nematodes, Aphelenchoides species; Sting nematodes, Belonolaimus longicaudatus and other Belonolaimus species; Pine nematodes, Bursaphelenchus xylophilus and other Bursaphelenchus species; Ring nematodes, Criconema species, Criconemella species, Criconemoides species, Mesocriconema species; Stem and bulb nematodes, Ditylenchus destructor, Ditylenchus dipsaci and other Ditylenchus species; Awl nematodes, Dolichodorus species; Spiral nematodes, Heliocotylenchus multicinctus and other Helicotylenchus species; Sheath and sheathoid nematodes, Hemicycliophora species and Hemicriconemoides species; Hirshmanniella species; Lance nematodes, Hoploaimus species; false rootknot nematodes, Nacobbus species;
Needle nematodes, Longidorus elongatus and other Longidorus species; Pin nematodes,
Pratylenchus species; Lesion nematodes, Pratylenchus neglectus, Pratylenchus penetrans,
Pratylenchus curvitatus, Pratylenchus goodeyi and other Pratylenchus species; Burrowing nematodes, Radopholus similis and other Radopholus species; Reniform nematodes, Rotylenchus robustus, Rotylenchus reniformis and other Rotylenchus species; Scutellonema species; Stubby root nematodes, Trichodorus primitivus and other Trichodorus species, Paratrichodorus species; Stunt nematodes, Tylenchorhynchus claytoni, Tylenchorhynchus dubius and other Tylenchorhynchus species; Citrus nematodes, Tylenchulus species; Dagger nematodes, Xiphinema species; and other plant parasitic nematode species, such as Subanguina spp., Hypsoperine spp., Macroposthonia spp., Melinius spp., Punctodera spp., and Quinisulcius spp..
The compounds of the invention may also have activity against the molluscs. Examples of which include, for example, Ampullariidae; Arion (A. ater, A. circumscriptus, A. hortensis, A. rufus);
Bradybaenidae (Bradybaena fruticum); Cepaea (C. hortensis, C. Nemoralis); ochlodina; Deroceras (D. agrestis, D. empiricorum, D. laeve, D. reticulatum); Discus (D. rotundatus); Euomphalia; Galba (G. trunculata); Helicelia (H. itala, H. obvia); Helicidae Helicigona arbustorum); Helicodiscus; Helix (H. aperta); Limax (L. cinereoniger, L. flavus, L. marginatus, L. maximus, L. tenellus); Lymnaea; Milax (M. gagates, M. marginatus, M. sowerbyi); Opeas; Pomacea (P. canaticulata); Vallonia and Zanitoides.
The term "crops" is to be understood as including also crop plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria, especially those of the genus Bacillus.
Toxins that can be expressed by such transgenic plants include, for example, insecticidal proteins, for example insecticidal proteins from Bacillus cereus or Bacillus popilliae; or insecticidal proteins from
Bacillus thuringiensis, such as δ-endotoxins, e.g. CrylAb, CrylAc, Cryl F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), e.g. Vip1 , Vip2, Vip3 or Vip3A; or insecticidal proteins of bacteria colonising nematodes, for example Photorhabdus spp. or
Xenorhabdus spp., such as Photorhabdus luminescens, Xenorhabdus nematophilus; toxins produced by animals, such as scorpion toxins, arachnid toxins, wasp toxins and other insect-specific neurotoxins; toxins produced by fungi, such as Streptomycetes toxins, plant lectins, such as pea lectins, barley lectins or snowdrop lectins; agglutinins; proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin, papain inhibitors; ribosome-inactivating proteins (RIP), such as ricin, maize-RIP, abrin, luffin, saporin or bryodin; steroid metabolism enzymes, such as 3-hydroxysteroidoxidase, ecdysteroid-UDP-glycosyl-transferase, cholesterol oxidases, ecdysone inhibitors, HMG-COA-reductase, ion channel blockers, such as blockers of sodium or calcium channels, juvenile hormone esterase, diuretic hormone receptors, stilbene synthase, bibenzyl synthase, chitinases and glucanases.
In the context of the present invention there are to be understood by δ-endotoxins, for example CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), for example Vip1 , Vip2, Vip3 or Vip3A, expressly also hybrid toxins, truncated toxins and modified toxins. Hybrid toxins are produced recombinantly by a new combination of different domains of those proteins (see, for example, WO 02/15701 ). Truncated toxins, for example a truncated CrylAb, are known. In the case of modified toxins, one or more amino acids of the naturally occurring toxin are replaced. In such amino acid replacements, preferably non-naturally present protease recognition sequences are inserted into the toxin, such as, for example, in the case of Cry3A055, a cathepsin-G-recognition sequence is inserted into a Cry3A toxin (see WO 03/018810). Examples of such toxins or transgenic plants capable of synthesising such toxins are disclosed, for example, in EP-A-0 374 753, WO 93/07278, WO 95/34656, EP-A-0 427 529, EP-A-451 878 and WO 03/052073.
The processes for the preparation of such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above. Cryl-type
deoxyribonucleic acids and their preparation are known, for example, from WO 95/34656, EP-A-0 367 474, EP-A-0 401 979 and WO 90/13651.
The toxin contained in the transgenic plants imparts to the plants tolerance to harmful insects. Such insects can occur in any taxonomic group of insects, but are especially commonly found in the beetles (Coleoptera), two-winged insects (Diptera) and moths (Lepidoptera).
Transgenic plants containing one or more genes that code for an insecticidal resistance and express one or more toxins are known and some of them are commercially available. Examples of such plants are: YieldGard® (maize variety that expresses a CrylAb toxin); YieldGard Rootworm® (maize variety that expresses a Cry3Bb1 toxin); YieldGard Plus® (maize variety that expresses a CrylAb and a Cry3Bb1 toxin); Starlink® (maize variety that expresses a Cry9C toxin); Herculex I® (maize variety
that expresses a Cry1 Fa2 toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylAc toxin); Bollgard I® (cotton variety that expresses a CrylAc toxin); Bollgard II® (cotton variety that expresses a CrylAc and a Cry2Ab toxin); VipCot® (cotton variety that expresses a Vip3A and a Cry1 Ab toxin); NewLeaf® (potato variety that expresses a Cry3A toxin); NatureGard®, Agrisure® GT Advantage (GA21 glyphosate-tolerant trait), Agrisure® CB Advantage (Bt1 1 corn borer (CB) trait) and Protecta®.
Further examples of such transgenic crops are:
1. Bt11 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a truncated Cry1 Ab toxin. Bt1 1 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium.
2. Bt176 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a CrylAb toxin. Bt176 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium.
3. MIR604 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Maize which has been rendered insect-resistant by transgenic expression of a modified Cry3A toxin. This toxin is Cry3A055 modified by insertion of a cathepsin-G- protease recognition sequence. The preparation of such transgenic maize plants is described in WO 03/018810.
4. MON 863 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1 150 Brussels, Belgium, registration number C/DE/02/9. MON 863 expresses a Cry3Bb1 toxin and has resistance to certain Coleoptera insects.
5. IPC 531 Cotton from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1 150 Brussels, Belgium, registration number C/ES/96/02.
6. 1507 Maize from Pioneer Overseas Corporation, Avenue Tedesco, 7 B-1 160 Brussels, Belgium, registration number C/NL/00/10. Genetically modified maize for the expression of the protein Cryl F for achieving resistance to certain Lepidoptera insects and of the PAT protein for achieving tolerance to the herbicide glufosinate ammonium.
7. NK603 x MON 810 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1 150 Brussels, Belgium, registration number C/GB/02/M3/03. Consists of conventionally bred hybrid maize varieties by crossing the genetically modified varieties NK603 and MON 810. NK603 * MON 810
Maize transgenically expresses the protein CP4 EPSPS, obtained from Agrobacterium sp. strain CP4, which imparts tolerance to the herbicide Roundup® (contains glyphosate), and also a CrylAb toxin
obtained from Bacillus thuringiensis subsp. kurstaki which brings about tolerance to certain
Lepidoptera, include the European corn borer.
Transgenic crops of insect-resistant plants are also described in BATS (Zentrum fur Biosicherheit und Nachhaltigkeit, Zentrum BATS, Clarastrasse 13, 4058 Basel, Switzerland) Report 2003,
(http://bats.ch).
The term "crops" is to be understood as including also crop plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so-called "pathogenesis-related proteins" (PRPs, see e.g. EP-A-0 392 225). Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP-A-0 392 225, WO 95/33818 and EP-A-0 353 191. The methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above. Crops may also be modified for enhanced resistance to fungal (for example Fusarium, Anthracnose, or Phytophthora), bacterial (for example Pseudomonas) or viral (for example potato leafroll virus, tomato spotted wilt virus, cucumber mosaic virus) pathogens.
Crops also include those that have enhanced resistance to nematodes, such as the soybean cyst nematode.
Crops that are tolerance to abiotic stress include those that have enhanced tolerance to drought, high salt, high temperature, chill, frost, or light radiation, for example through expression of NF-YB or other proteins known in the art. Antipathogenic substances which can be expressed by such transgenic plants include, for example, ion channel blockers, such as blockers for sodium and calcium channels, for example the viral KP1 , KP4 or KP6 toxins; stilbene synthases; bibenzyl synthases; chitinases; glucanases; the so-called "pathogenesis-related proteins" (PRPs; see e.g. EP-A-0 392 225); antipathogenic substances produced by microorganisms, for example peptide antibiotics or heterocyclic antibiotics (see e.g. WO 95/33818) or protein or polypeptide factors involved in plant pathogen defence (so-called "plant disease resistance genes", as described in WO 03/000906).
Further areas of use of the compositions according to the invention are the protection of stored goods and store ambients and the protection of raw materials, such as wood, textiles, floor coverings or buildings, and also in the hygiene sector, especially the protection of humans, domestic animals and productive livestock against pests of the mentioned type.
The present invention also provides a method for controlling pests (such as mosquitoes and other disease vectors; see also http://www.who.int/malaria/vector_control/irs/en/). In one embodiment, the
method for controlling pests comprises applying the compositions of the invention to the target pests, to their locus or to a surface or substrate by brushing, rolling, spraying, spreading or dipping. By way of example, an IRS (indoor residual spraying) application of a surface such as a wall, ceiling or floor surface is contemplated by the method of the invention. In another embodiment, it is contemplated to apply such compositions to a substrate such as non-woven or a fabric material in the form of (or which can be used in the manufacture of) netting, clothing, bedding, curtains and tents.
In one embodiment, the method for controlling such pests comprises applying a pesticidally effective amount of the compositions of the invention to the target pests, to their locus, or to a surface or substrate so as to provide effective residual pesticidal activity on the surface or substrate. Such application may be made by brushing, rolling, spraying, spreading or dipping the pesticidal composition of the invention. By way of example, an IRS application of a surface such as a wall, ceiling or floor surface is contemplated by the method of the invention so as to provide effective residual pesticidal activity on the surface. In another embodiment, it is contemplated to apply such compositions for residual control of pests on a substrate such as a fabric material in the form of (or which can be used in the manufacture of) netting, clothing, bedding, curtains and tents.
Substrates including non-woven, fabrics or netting to be treated may be made of natural fibres such as cotton, raffia, jute, flax, sisal, hessian, or wool, or synthetic fibres such as polyamide, polyester, polypropylene, polyacrylonitrile or the like. The polyesters are particularly suitable. The methods of textile treatment are known, e.g. WO 2008/151984, WO 2003/034823, US 5631072, WO 2005/64072, WO2006/128870, EP 1724392, WO20051 13886 or WO 2007/090739.
Further areas of use of the compositions according to the invention are the field of tree injection/trunk treatment for all ornamental trees as well all sort of fruit and nut trees.
In the field of tree injection/trunk treatment, the compounds according to the present invention are especially suitable against wood-boring insects from the order Lepidoptera as mentioned above and from the order Coleoptera, especially against woodborers listed in the following tables A and B:
Table A. Examples of exotic woodborers of economic importance.
Family Species Host or Crop Infested
Buprestidae Agrilus planipennis Ash
Cerambycidae Anoplura glabripennis Hardwoods
Xylosandrus crassiusculus Hardwoods
Scolytidae
X. mutilatus Hardwoods
Tomicus piniperda Conifers
Table B. Examples of native woodborers of economic importance.
Family Species Host or Crop Infested
Agrilus anxius Birch
Agrilus politus Willow, Maple
Agrilus sayi Bayberry, Sweetfern
Agrilus vittaticolllis Apple, Pear, Cranberry,
Serviceberry, Hawthorn
Chrysobothris femorata Apple, Apricot, Beech, Boxelder,
Buprestidae Cherry, Chestnut, Currant, Elm,
Hawthorn, Hackberry, Hickory, Horsechestnut, Linden, Maple, Mountain-ash, Oak, Pecan, Pear, Peach, Persimmon, Plum, Poplar, Quince, Redbud, Serviceberry, Sycamore, Walnut, Willow
Texania campestris Basswood, Beech, Maple, Oak,
Sycamore, Willow, Yellow-poplar
Goes pulverulentus Beech, Elm, Nuttall, Willow, Black oak, Cherrybark oak, Water oak, Sycamore
Goes tigrinus Oak
Cerambycidae Neoclytus acuminatus Ash, Hickory, Oak, Walnut, Birch,
Beech, Maple, Eastern
hophornbeam, Dogwood,
Persimmon, Redbud, Holly, Hackberry, Black locust,
Honeylocust, Yellow-poplar, Chestnut, Osage-orange, Sassafras,
Family Species Host or Crop Infested
Lilac, Mountain-mahogany, Pear, Cherry, Plum, Peach, Apple, Elm, Basswood, Sweetgum
Neoptychodes trilineatus Fig, Alder, Mulberry, Willow, Netleaf hackberry
Oberea ocellata Sumac, Apple, Peach, Plum, Pear,
Currant, Blackberry
Oberea tripunctata Dogwood, Viburnum, Elm,
Sourwood, Blueberry,
Rhododendron, Azalea, Laurel, Poplar, Willow, Mulberry
Oncideres cingulata Hickory, Pecan, Persimmon, Elm,
Sourwood, Basswood, Honeylocust, Dogwood, Eucalyptus, Oak, Hackberry, Maple, Fruit trees
Saperda calcarata Poplar
Strophiona nitens Chestnut, Oak, Hickory, Walnut,
Beech, Maple
Corthylus columbianus Maple, Oak, Yellow-poplar, Beech,
Boxelder, Sycamore, Birch, Basswood, Chestnut, Elm
Dendroctonus frontalis Pine
Dryocoetes betulae Birch, Sweetgum, Wild cherry,
Scolytidae Beech, Pear
Monarthrum fasciatum Oak, Maple, Birch, Chestnut,
Sweetgum, Blackgum, Poplar, Hickory, Mimosa, Apple, Peach, Pine
Phloeotribus liminaris Peach, Cherry, Plum, Black cherry,
Elm, Mulberry, Mountain-ash
Family Species Host or Crop Infested
Pseudopityophthorus pruinosus Oak, American beech, Black cherry,
Chickasaw plum, Chestnut, Maple,
Hickory, Hornbeam, Hophornbeam
Paranthrene simulans Oak, American chestnut
Sannina uroceriformis Persimmon
Synanthedon exitiosa Peach, Plum, Nectarine, Cherry,
Apricot, Almond, Black cherry
Synanthedon pictipes Peach, Plum, Cherry, Beach, Black
Cherry
Sesiidae
Synanthedon rubrofascia Tupelo
Synanthedon scitula Dogwood, Pecan, Hickory, Oak,
Chestnut, Beech, Birch, Black cherry,
Elm, Mountain-ash, Viburnum,
Willow, Apple, Loquat, Ninebark,
Bayberry
Vitacea polistiformis Grape
The present invention may be also used to control any insect pests that may be present in turfgrass, including for example beetles, caterpillars, fire ants, ground pearls, millipedes, sow bugs, mites, mole crickets, scales, mealybugs ticks, spittlebugs, southern chinch bugs and white grubs. The present invention may be used to control insect pests at various stages of their life cycle, including eggs, larvae, nymphs and adults.
In particular, the present invention may be used to control insect pests that feed on the roots of turfgrass including white grubs (such as Cyclocephala spp. (e.g. masked chafer, C. lurida),
Rhizotrogus spp. (e.g. European chafer, R. majalis), Cotinus spp. (e.g. Green June beetle, C. nitida), Popillia spp. (e.g. Japanese beetle, P. japonica), Phyllophaga spp. (e.g. May/June beetle), Ataenius spp. (e.g. Black turfgrass ataenius, A. spretulus), Maladera spp. (e.g. Asiatic garden beetle, M. castanea) and Tomarus spp.), ground pearls (Margarodes spp.), mole crickets (tawny, southern, and short-winged; Scapteriscus spp., Gryllotalpa africana) and leatherjackets (European crane fly, Tipula spp.).
The present invention may also be used to control insect pests of turfgrass that are thatch dwelling, including armyworms (such as fall armyworm Spodoptera frugiperda, and common armyworm Pseudaletia unipuncta), cutworms, billbugs (Sphenophorus spp. , such as S. venatus verstitus and S. parvulus), and sod webworms (such as Crambus spp. and the tropical sod webworm, Herpetogramma phaeopteralis).
The present invention may also be used to control insect pests of turfgrass that live above the ground and feed on the turfgrass leaves, including chinch bugs (such as southern chinch bugs, Blissus insularis), Bermudagrass mite (Eriophyes cynodoniensis), rhodesgrass mealybug (Antonina graminis), two-lined spittlebug (Propsapia bicincta), leafhoppers, cutworms (Noctuidae family), and greenbugs. The present invention may also be used to control other pests of turfgrass such as red imported fire ants (Solenopsis invicta) that create ant mounds in turf.
In the hygiene sector, the compositions according to the invention are active against ectoparasites such as hard ticks, soft ticks, mange mites, harvest mites, flies (biting and licking), parasitic fly larvae, lice, hair lice, bird lice and fleas.
Examples of such parasites are:
Of the order Anoplurida: Haematopinus spp., Linognathus spp., Pediculus spp. and Phtirus spp., Solenopotes spp.. Of the order Mallophagida: Trimenopon spp., Menopon spp., Trinoton spp., Bovicola spp.,
Werneckiella spp., Lepikentron spp., Damalina spp., Trichodectes spp. and Felicola spp..
Of the order Diptera and the suborders Nematocerina and Brachycerina, for example Aedes spp., Anopheles spp., Culex spp., Simulium spp., Eusimulium spp., Phlebotomus spp., Lutzomyia spp., Culicoides spp., Chrysops spp., Hybomitra spp., Atylotus spp., Tabanus spp., Haematopota spp.,
Philipomyia spp., Braula spp., Musca spp., Hydrotaea spp., Stomoxys spp., Haematobia spp., Morellia spp., Fannia spp., Glossina spp., Calliphora spp., Lucilia spp., Chrysomyia spp., Wohlfahrtia spp., Sarcophaga spp., Oestrus spp., Hypoderma spp., Gasterophilus spp., Hippobosca spp., Lipoptena spp. and Melophagus spp..
Of the order Siphonapterida, for example Pulex spp., Ctenocephalides spp., Xenopsylla spp., Ceratophyllus spp..
Of the order Heteropterida, for example Cimex spp., Triatoma spp., Rhodnius spp., Panstrongylus spp..
Of the order Blattarida, for example Blatta orientalis, Periplaneta americana, Blattelagermanica and Supella spp..
Of the subclass Acaria (Acarida) and the orders Meta- and Meso-stigmata, for example Argas spp., Ornithodorus spp., Otobius spp., Ixodes spp., Amblyomma spp., Boophilus spp., Dermacentor spp., Haemophysalis spp., Hyalomma spp., Rhipicephalus spp., Dermanyssus spp., Raillietia spp., Pneumonyssus spp., Sternostoma spp. and Varroa spp..
Of the orders Actinedida (Prostigmata) and Acaridida (Astigmata), for example Acarapis spp., Cheyletiella spp., Ornithocheyletia spp., Myobia spp., Psorergatesspp., Demodex spp., Trombicula spp., Listrophorus spp., Acarus spp., Tyrophagus spp., Caloglyphus spp., Hypodectes spp.,
Pterolichus spp., Psoroptes spp., Chorioptes spp., Otodectes spp., Sarcoptes spp., Notoedres spp., Knemidocoptes spp., Cytodites spp. and Laminosioptes spp..
The compositions according to the invention are also suitable for protecting against insect infestation in the case of materials such as wood, textiles, plastics, adhesives, glues, paints, paper and card, leather, floor coverings and buildings.
The compositions according to the invention can be used, for example, against the following pests: beetles such as Hylotrupes bajulus, Chlorophorus pilosis, Anobium punctatum, Xestobium
rufovillosum, Ptilinuspecticornis, Dendrobium pertinex, Ernobius mollis, Priobium carpini, Lyctus brunneus, Lyctus africanus, Lyctus planicollis, Lyctus linearis, Lyctus pubescens, Trogoxylon aequale, Minthesrugicollis, Xyleborus spec.,Tryptodendron spec, Apate monachus, Bostrychus capucins, Heterobostrychus brunneus, Sinoxylon spec, and Dinoderus minutus, and also hymenopterans such as Sirex juvencus, Urocerus gigas, Urocerus gigas taignus and Urocerus augur, and termites such as Kalotermes flavicollis, Cryptotermes brevis, Heterotermes indicola, Reticulitermes flavipes,
Reticulitermes santonensis, Reticulitermes lucifugus, Mastotermes darwiniensis, Zootermopsis nevadensis and Coptotermes formosanus, and bristletails such as Lepisma saccharina.
The compounds according to the invention can be used as pesticidal agents in unmodified form, but they are generally formulated into compositions in various ways using formulation adjuvants, such as carriers, solvents and surface-active substances. The formulations can be in various physical forms, e.g. in the form of dusting powders, gels, wettable powders, water-dispersible granules, water- dispersible tablets, effervescent pellets, emulsifiable concentrates, microemulsifiable concentrates, oil- in-water emulsions, oil-flowables, aqueous dispersions, oily dispersions, suspo-emulsions, capsule suspensions, emulsifiable granules, soluble liquids, water-soluble concentrates (with water or a water- miscible organic solvent as carrier), impregnated polymer films or in other forms known e.g. from the Manual on Development and Use of FAO and WHO Specifications for Pesticides, United Nations, First Edition, Second Revision (2010). Such formulations can either be used directly or diluted prior to use.
The dilutions can be made, for example, with water, liquid fertilisers, micronutrients, biological organisms, oil or solvents.
The formulations can be prepared e.g. by mixing the active ingredient with the formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions. The active ingredients can also be formulated with other adjuvants, such as finely divided solids, mineral oils, oils of vegetable or animal origin, modified oils of vegetable or animal origin, organic solvents, water, surface-active substances or combinations thereof.
The active ingredients can also be contained in very fine microcapsules. Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into the environment in controlled amounts (e.g. slow-release). Microcapsules usually have a diameter of from 0.1 to 500 microns. They contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight. The active ingredients can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution. The encapsulating membranes can comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art. Alternatively, very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
The formulation adjuvants that are suitable for the preparation of the compositions according to the invention are known per se. As liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2-butanone, butylene carbonate, chlorobenzene, cyclohexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1 ,2-dichloropropane, diethanolamine, p- diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, A/,A/-dimethylformamide, dimethyl sulfoxide, 1 ,4- dioxane, dipropylene glycol, dipropylene glycol methyl ether, dipropylene glycol dibenzoate, diproxitol, alkylpyrrolidone, ethyl acetate, 2-ethylhexanol, ethylene carbonate, 1 ,1 , 1-trichloroethane, 2- heptanone, alpha-pinene, d-limonene, ethyl lactate, ethylene glycol, ethylene glycol butyl ether, ethylene glycol methyl ether, gamma-butyrolactone, glycerol, glycerol acetate, glycerol diacetate, glycerol triacetate, hexadecane, hexylene glycol, isoamyl acetate, isobornyl acetate, isooctane, isophorone, isopropylbenzene, isopropyl myristate, lactic acid, laurylamine, mesityl oxide, methoxy- propanol, methyl isoamyl ketone, methyl isobutyl ketone, methyl laurate, methyl octanoate, methyl oleate, methylene chloride, m-xylene, n-hexane, n-octylamine, octadecanoic acid, octylamine acetate, oleic acid, oleylamine, o-xylene, phenol, polyethylene glycol, propionic acid, propyl lactate, propylene carbonate, propylene glycol, propylene glycol methyl ether, p-xylene, toluene, triethyl phosphate, triethylene glycol, xylenesulfonic acid, paraffin, mineral oil, trichloroethylene, perchloroethylene, ethyl acetate, amyl acetate, butyl acetate, propylene glycol methyl ether, diethylene glycol methyl ether,
methanol, ethanol, isopropanol, and alcohols of higher molecular weight, such as amyl alcohol, tetrahydrofurfuryl alcohol, hexanol, octanol, ethylene glycol, propylene glycol, glycerol, A/-methyl-2- pyrrolidone and the like.
Suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montmorillonite, cottonseed husks, wheat flour, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar substances.
A large number of surface-active substances can advantageously be used in both solid and liquid formulations, especially in those formulations which can be diluted with a carrier prior to use. Surface- active substances may be anionic, cationic, non-ionic or polymeric and they can be used as emulsifiers, wetting agents or suspending agents or for other purposes. Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol/alkylene oxide addition products, such as nonylphenol ethoxylate; alcohol/alkylene oxide addition products, such as tridecylalcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonat.es, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2- ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryltrimethylammonium chloride, polyethylene glycol esters of fatty acids, such as polyethylene glycol stearate; block copolymers of ethylene oxide and propylene oxide; and salts of mono- and di- alkylphosphate esters; and also further substances described e.g. in McCutcheon's Detergents and Emulsifiers Annual, MC Publishing Corp., Ridgewood New Jersey (1981 ).
Further adjuvants that can be used in pesticidal formulations include crystallisation inhibitors, viscosity modifiers, suspending agents, dyes, anti-oxidants, foaming agents, light absorbers, mixing auxiliaries, antifoams, complexing agents, neutralising or pH-modifying substances and buffers, corrosion inhibitors, fragrances, wetting agents, take-up enhancers, micronutrients, plasticisers, glidants, lubricants, dispersants, thickeners, antifreezes, microbicides, and liquid and solid fertilisers.
The compositions according to the invention can include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives. The amount of oil additive in the composition according to the invention is generally from 0.01 to 10 %, based on the mixture to be applied. For example, the oil additive can be added to a spray tank in the desired concentration after a spray mixture has been prepared. Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow. Preferred oil additives comprise alkyl esters of C8-C22 fatty acids, especially the methyl derivatives of C12-C18 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid (methyl laurate, methyl palmitate and methyl oleate, respectively). Many oil derivatives are known from the Compendium of Herbicide Adjuvants, 10th Edition, Southern Illinois University, 2010.
The inventive compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, of compounds of the present invention and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance. Whereas commercial products may preferably be formulated as concentrates, the end user will normally employ dilute formulations.
The rates of application vary within wide limits and depend on the nature of the soil, the method of application, the crop plant, the pest to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop. As a general guideline compounds may be applied at a rate of from 1 to 2000 l/ha, especially from 10 to 1000 l/ha.
Preferred formulations can have the following compositions (weight %):
Emulsifiable concentrates:
active ingredient: 1 to 95 %, preferably 60 to 90 %
surface-active agent: 1 to 30 %, preferably 5 to 20 %
liquid carrier: 1 to 80 %, preferably 1 to 35 %
Dusts:
active ingredient: 0.1 to 10 %, preferably 0.1 to 5 %
solid carrier: 99.9 to 90 %, preferably 99.9 to 99 %
Suspension concentrates:
active ingredient: 5 to 75 %, preferably 10 to 50 %
water: 94 to 24 %, preferably 88 to 30 %
surface-active agent: 1 to 40 %, preferably 2 to 30 %
Wettable powders:
active ingredient: 0.5 to 90 %, preferably 1 to 80 %
surface-active agent: 0.5 to 20 %, preferably 1 to 15 %
solid carrier: 5 to 95 %, preferably 15 to 90 %
Granules:
active ingredient: 0.1 to 30 %, preferably 0.1 to 15 %
solid carrier: 99.5 to 70 %, preferably 97 to 85 %
The following Examples further illustrate, but do not limit, the invention.
Wettable powders a) b) c)
active ingredients 25 % 50 % 75 %
sodium lignosulfonate 5 % 5 % - sodium lauryl sulfate 3 % - 5 % sodium diisobutylnaphthalenesulfonate - 6 % 10 % phenol polyethylene glycol ether (7-8 mol of ethylene 2 %
oxide)
highly dispersed silicic acid 5 % 10 % 10 %
Kaolin 62 % 27 % -
The combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
The combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
Emulsions of any required dilution, which can be used in plant protection, can be obtained from this concentrate by dilution with water.
Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such powders can also be used for dry dressings for seed.
Extruder granules
Active ingredients 15 %
sodium lignosulfonate 2 %
carboxymethylcellulose 1 %
Kaolin 82 %
The combination is mixed and ground with the adjuvants, and the mixture is moistened with water. The mixture is extruded and then dried in a stream of air.
The finely ground combination is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol. Non-dusty coated granules are obtained in this manner.
The finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
The finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water. Using such dilutions, living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
Slow Release Capsule Suspension
28 parts of the combination are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8: 1 ). This mixture is emulsified in a mixture of 1 .2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51 .6 parts of water until the desired particle size is achieved. To this emulsion a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added. The mixture is agitated until the polymerization reaction is completed. The obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent. The capsule suspension formulation contains 28% of the active ingredients. The medium capsule diameter is 8-15 microns. The resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
Formulation types include an emulsion concentrate (EC), a suspension concentrate (SC), a suspo- emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK), a dispersible concentrate (DC), a wettable powder (WP), a soluble granule (SG) or any technically feasible formulation in combination with agriculturally acceptable adjuvants.
Preparatory Examples:
"Mp" means melting point in °C. Free radicals represent methyl groups. H NMR and 9F NMR measurements were recorded on a Brucker 400 MHz or 300 MHz spectrometer, chemical shifts are given in ppm relevant to a TMS standard. Spectra measured in deuterated solvents as indicated.
LCMS Methods:
Method 1 :
Spectra were recorded on a Mass Spectrometer from Waters (SQD, SQDII or ZQ Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, Capillary: 3.00 kV, Cone range: 30-60 V, Extractor: 2.00 V, Source Temperature: 150°C, Desolvation Temperature: 350°C, Cone Gas Flow: 0 L/Hr, Desolvation Gas Flow: 650 L/Hr, Mass range: 100 to 900 Da) and an Acquity UPLC from Waters: Binary pump, heated column compartment and diode- array detector. Solvent degasser, binary pump, heated column compartment and diode-array detector. Column: Waters UPLC HSS T3 , 1 .8 mm, 30 x 2.1 mm, Temp: 60 °C, DAD Wavelength range (nm): 210 to 500, Solvent Gradient: A = water + 5% MeOH + 0.05 % HCOOH, B= Acetonitrile + 0.05 % HCOOH, gradient: 10-100% B in 1.2 min; Flow (ml/min) 0.85
Method 2 - Standard long:
Spectra were recorded on a Mass Spectrometer from Waters (SQD or ZQ Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, Capillary: 3.00
kV, Cone range: 30-60 V, Extractor: 2.00 V, Source Temperature: 150°C, Desolvation Temperature: 350°C, Cone Gas Flow: 0 L/Hr, Desolvation Gas Flow: 650 L/Hr, Mass range: 100 to 900 Da) and an Acquity UPLC from Waters: Binary pump, heated column compartment and diode-array detector. Solvent degasser, binary pump, heated column compartment and diode-array detector. Column: Waters UPLC HSS T3 , 1.8 μηη, 30 x 2.1 mm, Temp: 60 °C, DAD wavelength range (nm): 210 to 500, Solvent Gradient: A = water + 5% MeOH + 0.05 % HCOOH, B= acetonitrile + 0.05 % HCOOH:
gradient: gradient: 0 min 0% B, 100%A; 2.7-3. Omin 100% B; Flow (ml/min) 0.85
Mass Spectroscopy Method MS (ESI-MS)
LC-20AD Mass Spectrometer from Shimadzu (Single quadrupole mass spectrometer)
Instrument Parameters:
lonisation method: Electrospray
Polarity: positive and negative ions
Capillary (kV) 1.50
Cone (V) unknown
Extractor (V) 5.00
Source Temperature (°C) 200
Desolvation Temperature (°C) 250
Cone gas Flow (l/Hr) 90
Desolvation gas Flow (l/Hr) 90
Mass range:50 to 1000 Da
Example H1 : Preparation of 2-[5-(3,5-difluorophenyl)-3-ethylsulfonyl-2-pyridyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
Step 1 : Preparation of 5-bromo-3-ethvsulfanyl-pyridine-2-carbonitrile
A sample of 5-bromo-3-nitro-pyridine-2-carbonitrile (5.7 g, 25 mmol, CAS: 573675-25-9) was dissolved in DMF(100 ml) and EtSNa (2.2 g, 26.25 mmol) was added when the temperature was dropped to -50 °C. After the mixture was stirred at r.t for 16 h, it was poured into the dilute hydrochloric acid and
extracted with ethyl acetate three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated under vacuum. The crude product was purified by column chromatography on silica gel to give the title compound.
H NMR (400Mz, CDCIs) δ (ppm): 1.42 (t, 3 H), 3.07 (q, 2 H), 7.81 (d, 1 H), 8.48 (d, 1 H); ESI-MS(-): 187 (M-1 ).
Step 2: Preparation of 1-(5-bromo-3-ethylsulfanyl-2-pyridyl)ethanone
To a solution of 5-bromo-3-ethylsulfanyl-pyridine-2-carbonitrile (22 g, 90 mmol) in 300 mL of dry tetrahydrofuran at -10 °C was added Ch MgBr (90 mL of a 3 M solution in hexane, 270 mmol) under an nitrogen atmosphere. After stirring for 30 min at -10 °C, the mixture was stirred at r.t for 2 h. Then, the mixture was poured into dilute hydrochloric acid and extracted with ethyl acetate three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated under vacuum. The crude product was purified by column chromatography on silica gel to give 1-(5-bromo-3-ethylsulfanyl- 2-pyridyl)ethanone.
Ή NMR (400Mz, CDCI3) δ (ppm): 1.40 (t, 3H), 2.65 (s, 3H), 2.88(q, 2H), 7.74 (s, 1 H), 8.38 (s, 1 H); ESI-MS(+): 261 (M+1 ).
A solution of 1-(5-bromo-3-ethylsulfanyl-2-pyridyl)ethanone (4 mmol, 1.04 g) and mCPBA (2.06 g, 12 mmol) in 20 ml of DCM was stirred at ambient temperature for 4 h. Then the mixture was poured into a saturated solution of NaHCCh and Na2S03 in water, and extracted with ethyl acetate three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated under vacuum. The crude product was purified by column chromatography on silica gel to give 1-(5-bromo-3-ethylsulfonyl- 2-pyridyl) ethanone.
Ή NMR (400Mz, CDCI3) δ (ppm): 1.34 (t, 3 H), 2.66 (s, 3 H), 3.58 (q, 2 H), 8.47(s, 1 H), 8.83 (s, 1
1-(5-bromo-3-ethylsulfonyl-2-pyridyl)ethanone (10 mmol, 2.92 g) and CuBr2 (15 mmol, 3.36 g) in 5 ml of Ch CN and 5 ml of CHC were stirred at sealed tube 140 °C for 7 h. Then the mixture was concentrated under vacuum. The crude product was purified by column chromatography on silica gel to give 2-bromo-1-(5-bromo-3-ethylsulfonyl-2-pyridyl)ethanone.
Ή NMR (400Mz, CDCI3) δ: 1.34 (t, 3H), 3.60 (q, 2H), 4.68 (s, 2H), 8.54 (s, 1 H), 8.87 (s, 1 H).
Step 5 : Preparation of 2-(5-bromo-3-ethylsulfonyl-2-pyridvn-7-(trifluoromethvnimidazo[1 ,2-alpyridine
In MW vial, 4-(trifluoromethyl)pyridin-2-amine (283 mg, 1.7491 mmol) and 2-bromo-1-(5-bromo-3- ethylsulfonyl-2-pyridyl)ethanone(649 mg, 1.7491 mmol) were dissolved in acetonitrile (14 ml) and heated in microwave reactor for 2 hours at 150 °C. Solvent was evaporated under pressure, residue was dissolved in dichloromethane and saturated solution of NaHCCh was added. The aqueous layer was extracted with dichloromethane and then washed with saturated solution of NaCI. The combined organic layer were dried with Na2S04, filtered and evaporated. The crude was purified by flash chromatography on silica gel to give the title compound as a yellow solid.
LC-MS (method 1 ): (M+H+) 434/436; Rt: 1 .01 min
Ή NMR (400 MHz, CHLOROFORM-d) δ ppm 1.41 (t, J=7.52 Hz, 3 H) 4.05 (q, J=7.46 Hz, 2 H) 7.08 (dd, J=7.15, 1.65 Hz, 1 H) 7.99 (s, 1 H) 8.32 (d, J=7.34 Hz, 1 H) 8.34 (s, 1 H) 8.71 (d, J=2.20 Hz, 1 H) 8.96 (d, J=2.20 Hz, 1 H).
Step 6: Preparation of 2-[5-(3,5-difluorophenyl)-3-ethylsulfonyl-2-pyridyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
ln a supelco vial, 2-(5-bromo-3-ethylsulfonyl-2-pyridyl)-7-(trifluoromethyl)imidazo[1 ,2-a]p (100 mg, 0.2303 mmol) , (3,5-difluorophenyl)boronic acid (43 mg, 0.2764 mmol) and potassium carbonate (95 mg, 0.6909 mmol) were dissolved in 1 ,4-dioxane (2.5 ml). Argon was blew into reaction mixture for 5 minutes. Then tetrakis(triphenylphosphine)palladium (26 mg, 0.02303 mmol) was added and the vial was degassed with argon, closed and stirred one night at 95°C. LC-MS analysis showed the formation of desired product. The reaction mixture was cooled down at ambient temperature, quenched with water (4 ml) and extracted with ethyl acetate (4 x 4 ml). The combined organic layers were washed with saturated solution of NaHCCh and saturated solution of NaCI, dried with Na2S04, filtered and evaporated. The crude was purified by flash chromatography on silica gel to give the title compound as an orange solid.
Mpt. 213 - 214 °C
LC-MS (method 1 ): (M+H+) 468; Rt: 1.10 min
Ή NMR (400 MHz, CHLOROFORM-d) δ ppm 1.42 (t, J=7.34 Hz, 3 H) 4.05 (q, J=7.46 Hz, 2 H) 6.98 (tt, J=8.67, 2.34 Hz, 1 H) 7.09 (dd, J=6.97, 1.83 Hz, 1 H) 7.27 (dd, J=7.70, 2.20 Hz, 2 H) 8.01 (s, 1 H) 8.34 (d, J=7.34 Hz, 1 H) 8.41 (s, 1 H) 8.72 (d, J=2.20 Hz, 1 H) 9.10 (d, J=2.20 Hz, 1 H).
Example H2: Preparation of 2-[5-(4-chlorophenyl)-3-ethylsulfonyl-2-pyridyll-7-
(trifluoromethyl)imidazo[1 ,2-alpyridine
2-(5-bromo-3-ethylsulfonyl-2-pyridyl)-7-(trifluoromethyl)imidazo[1 ,2-a]pyridine (0.1 g, 0.2303 mmol) ,(4-chlorophenyl)boronic acid (0.04549 g, 0.2764 mmol) ,disodium carbonic acid (0.2879 mL, 0.5758 mmol), 1 , 1-dimethoxyethane (4 mL) were mixed in a vial and argon was bubbled within 5min through the mixture. Then tetrakis(triphenylphosphine)palladium (0.05323 g, 0.04606 mmol) was added and the now pale brown mixture was stirred one night at 95°C. LC-MS analysis showed the formation of desired product. The reaction mixture was cooled down at ambient temperature, diluted with 10ml water, and extracted with ethyl acetate. The combined organic layers were washed with water and brine, dried over Na2S04 and concentrated under vacuum at 45°C. The crude was purified by flash chromatography on silica gel to give the title compound as an orange solid.
Mpt. 178 - 180°C
LC-MS (method 1 ): (M+H+) 466; Rt: 1.1 1 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.35 - 1.40 (m, 3 H) 3.98 (q, J=7.46 Hz, 2 H) 7.05 (dd, J=7.15, 1.65 Hz, 1 H) 7.51 - 7.54 (m, 2 H) 7.63 - 7.67 (m, 2 H) 7.98 (s, 1 H) 8.31 (d, J=6.97 Hz, 1 H) 8.36 (s, 1 H) 8.70 (d, J=2.20 Hz, 1 H) 9.08 (d, J=2.20 Hz, 1 H)
Example H3: Preparation of 2-[3-ethylsulfonyl-5-[4-(trifluoromethyl)phenyll-2-pyridyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
2-(5-bromo-3-ethylsulfonyl-2-pyridyl)-7-(trifluoromethyl)imidazo[1 ,2-a]pyridine (0.06 g, 0.1382 mmol), ([4-(trifluoromethyl)phenyl]boronic acid (0.03149 g, 0.1658 mmol), carbonic acid (0.1727 mL, 0.3455 mmol), 1 , 1-dimethoxyethane (2 mL) were mixed in a vial and argon was bubbled within 5min through the mixture. Then tetrakis(triphenylphosphine)palladium (0.03194 g, 0.02764 mmol) was added and the now pale brown mixture was stirred one night at 95°C. LC-MS analysis showed the formation of desired product. The reaction mixture was cooled down at ambient temperature, diluted with 10ml water, and extracted with ethyl acetate. The combined organic layers were washed with water and brine, dried over Na2S04 and concentrated under vacuum at 45°C. The crude was purified by flash chromatography on silica gel to give the title compound as an orange solid.
Mpt. 95 - 98 °C
LC-MS (method 1 ): (M+H+) 500; Rt: 1.12 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.39 (t, J=7.52 Hz, 3 H) 4.01 (q, J=7.34 Hz, 2 H) 7.06 (dd, J=6.97, 1.83 Hz, 1 H) 7.82 (s, 4 H) 7.98 (s, 1 H) 8.31 (d, J=7.34 Hz, 1 H) 8.38 (s, 1 H) 8.76 (d, J=2.20 Hz, 1 H) 9.12 (d, J=2.20 Hz, 1 H)
Example H4: Preparation of 2-[3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyll-2-pyridyll-7- (trifluorometh l)imidazo[1 ,2-alpyridine
To a solution of 3,6-dichloropyridine-2-carboxylic acid (76.8 g, 0.4 mol) in methanol (500 mL) was added SOCI2 (150 ml) dropwise at ambient temperature. The reaction mixture was stirred at ambient temperature for 3 hours. After this time, the reaction mixture was poured into water and extracted with ethyl acetate three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated in vacuo to give the title compound.
ESI-MS(+): 228 (M + Na)+.
Ή-NMR (400 MHz, DMSO-d6): 5ppm 3.90 (s, 3 H), 7.80 (d, J=8.8 Hz,1 H), 8.20 (d, J=8.8 Hz,1 H).
Step 2: Preparation of methyl 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylate
To a solution of methyl 3,6-dichloropyridine-2-carboxylate (16 g, 77.6 mmol) in DMF (150 mL) was added sodium ethanethiolate (7.2 g, 85.8 mmol) at 0 °C. After the addition, the reaction mixture was stirred at ambient temperature for 30 min. LCMS analysis after this time showed reaction completion. The reaction mixture was poured into water, and precipitate formed filtered and dried under an infrared oven to afford the title compound as white solid.
ESI-MS(+): 254 (M + Na)+.
Ή-NMR (400 MHz, CDCI3): 5ppm 1.38 (t, 3 H), 2.92 (q, 2 H), 3.98 (s, 3H), 7.40 (d, J=8.8 Hz, 1 H), 7.66 (d, J=8.8 Hz,1 H)-
Step 3: Preparation of methyl 3-ethylsulfanyl-6-[4-(trifluoromethyl)phenyllpyridine-2-carboxylate
In a three neck flask under argon, methyl 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylate (0.3 g, 1.2948 mmol), [4-(trifluoromethyl)phenyl]boronic acid (0.31969 g, 1.6832 mmol) , potassium carbonate (0.53683 g, 3.8843 mmol) were dissolved in 1 ,4-dioxane (7.5 mL). The resulting mixture was flushed
10' with argon and tetrakis(triphenylphosphine)palladium (0.14962 g, 0.12948 mmol) was added. Reaction mixture was stirred one night at 95°C. LC-MS analysis showed the mass of the desired product and a bit of starting material. Reaction mixture was quenched with water at ambient temperature and ethyl acetate was added. The aqueous layer was extracted 3 times with ethyl acetate. The combined organic layer was washed with NaHCCh sat sol and brine, dried over Na2S04, filtered and evaporated under vacuum at 45°C. The crude was purified by flash chromatography on silica gel to give the title compound as a yellow solid.
LC-MS (method 1 ): (M+H+) 342; Rt: 1.18 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.45 (t, J=7.52 Hz, 3 H) 3.03 (q, J=7.34 Hz, 2 H) 4.07 (s, 3 H) 7.76 (d, J=8.07 Hz, 2 H) 7.81 - 7.88 (m, 2 H) 8.16 (d, J=8.07 Hz, 2 H)
Step 4: Preparation of methyl 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyllpyridine-2-carboxylate
In a three neck flask under argon, methyl 3-ethylsulfanyl-6-[4-(trifluoromethyl)phenyl]pyridine-2- carboxylate (3.35 g, 9.81 mmol) was dissolved in dichloromethane (67.0 mL) and cooled down at 0°C Then 3-CHLOROPEROXYBENZOIC ACID (5.08 g, 20.6 mmol) was added and reaction was stirred 30' at 0°C then warmed up at ambient temperature and stirred 3 hours. LC-MS analysis showed the mass of desired product. Reaction mixture was quenched with NaOH 1 M (10 ml) and sodium thiosulfate sol (5ml). The aqueous layer was extracted 3 times with dichloromethane. The combined organic layer was washed with NaOH 1 M (2 times), brine, dried over Na2S04, filtered and evaporated under vacuum to give the title product.
LC-MS (method 1 ): (M+H+) 374; Rt: 1.08 min
1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 1.37 (t, J=7.34 Hz, 3 H) 3.53 (q, J=7.34 Hz, 2 H) 4.07 (s, 3 H) 7.78 (d, J=8.07 Hz, 2 H) 8.03 (d, J=8.44 Hz, 1 H) 8.21 (d, J=8.44 Hz, 2 H) 8.42 (d, J=8.44 Hz, 1 H)
Methyl 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]pyridine-2-carboxylate (3.64 g, 9.75 mmol) was dissolved in tetrahydrofuran (54.6 mL) and water (18.2 mL) . Then hydroxylithium hydrate (0.429 g, 10.2 mmol) was added at ambient temperature. Reaction mixture was stirred one night at ambient temperature.
LC-MS analysis showed the formation of desired product. Tetrahydrofuran was evaporated and 9.75 mL of HCI 1 N was added to the residue until pH 1. Then aqueous layer was extracted 3 times with ethylacetate. The organic layer was dried over Na2S04, filtered and evaporated under vacuum to give the title compound.
LC-MS (method 1 ): (M+H+) 358; Rt: 0.91 min
Ή NMR (400 MHz, DMSO-c/e) δ ppm 1.23 (t, J=7.34 Hz, 3 H) 3.58 (q, J=7.58 Hz, 2 H) 7.96 (d, J=8.07 Hz, 2 H) 8.41 (d, J=8.07 Hz, 2 H) 8.45 - 8.52 (m, 2 H) 14.30 (br. s., 1 H)
A sample of 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]pyridine-2-carboxylic acid (3.9 g, 1 1 mmol) was dissolved in dichloromethane (59 mL) and dimethylformamide (2 drops) was added. Then oxalyl dichloride (1.2 mL, 14 mmol) was added (formation of gas ) and reaction mixture was stirred at ambient temperature until the gas evolution was stopped. Sample of reaction mixture was taken and quenched with methanol. LC-MS analysis showed the formation of the intermediate 3-ethylsulfonyl-6- [4-(trifluoromethyl)phenyl]pyridine-2-carbonyl chloride. The solvent was removed by evaporation. In a three necks flasks, under argon, N-methoxymethanamine hydrochloride (1.1 g, 1 1 mmol) was solved with dichloromethane (62 mL) and TRIETHYLAMINE (3.8 g, 5.3 mL, 38 mmol) was added. Reaction mixture was cooled down at 0-5°C and 3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]pyridine-2- carbonyl chloride previously prepared (4.1 g, 100, 1 1 mmol) was dissolved in 3 ml of dichloromethane
and added slowly at 0°C. Reaction mixture was stirred 1 hour at 0°C. Reaction mixture was warm up at ambient temperature and stirred 30'. Water was added in the reaction mixture, organic layer was separated and aqueous layer was extracted 2 times with dichloromethane. The combined organic layer was washed with water, dried over Na2S04, filtered and evaporated under vacuum. The crude was purified by flash chromatography on silica gel to give the title compound as a colorless oil.
LC-MS (method 1 ): (M+H+) 500; Rt: 1 .21 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.33 (t, J=7.34 Hz, 3 H) 3.43 (s, 3 H) 3.45 - 3.51 (q, 2 H) 3.62 (s, 3 H) 7.78 (d, J=8.44 Hz, 2 H) 7.99 (d, J=8.44 Hz, 1 H) 8.24 (d, J=8.07 Hz, 2 H) 8.35 - 8.41 (m, 1 H)
Step 7: Preparation of 1-[3-ethylsulfonyl-6-[4-(trifluoromethvnphenyll-2-pyridyllethanone
In a three neck flask under argon, bromo(methyl)magnesium (6.8 mL, 9.5 mmol) was added in toluene (48 mL). Then, the solution was cooled down at 0°C and 3-ethylsulfonyl-N-methoxy-N-methyl-6-[4- (trifluoromethyl)phenyl]pyridine-2-carboxamide (3.2 g, 8.0 mmol) dissolved in 10 ml of Toluene was added dropwise. Reaction mixture was stirred 1 h at 0°C and then 1 h at ambient temperature. A white precipitate was formed in the reaction mixture. LC-MS analysis showed the formation of the desired product. The crude was slowly quenched with NH4CI sat aq (10 ml) and HCI 10% (5 ml) and resulting mixture was strongly stirred 15' at ambient temperature. The aqueous layer was extracted twice with ethyl acetate. The organic phase was then washed with 10 % HCI aq sol, water and brine, dried over Na2S04 anhydrous, filtered and concentrated. The crude was purified two times by flash
chromatography on silica gel to give the title compound as a mixture. This mixture was used for the next step without further purification.
LC-MS (method 1 ): (M+H+) 358; Rt: 1.1 1 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.37 (t, J=7.34 Hz, 3 H) 2.80 (s, 3 H) 3.59 (q, J=7.46 Hz, 2 H) 7.80 (d, J=8.07 Hz, 2 H) 8.02 (d, J=8.07 Hz, 1 H) 8.22 (d, J=8.07 Hz, 2 H) 8.44 (d, J=8.44 Hz, 1 H) Step 8 : Preparation of 2-bromo-1-[3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyll-2-pyridyllethanone
ln microwave vial, 1-[3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]-2-pyridyl]ethanone (1 g, 2.798 mmol), dibromocopper (1.250 g, 5.596 mmol) , acetonitrile (7 mL) and chloroform (7 mL) were mixed together and the resulting mixture was stirred 55' at 140°C under microwave.LC-MS analysis showed the formation of desired product. The reaction mixture was dissolved with dichloromethane, NaHCCh sat sol (30 mL) and NhUOH 1 M (30 mL). The aqueous layer was extracted 2 times with
dichloromethane. The combined organic layers were dried over Na2S04, filtered and evaporated under vacuum.
Obtained: 1.2 g, brown oil, mixture containing desired product
The crude was purified by flash chromatography on silica gel to give the title compound as a white solid.
LC-MS (method 1 ): (M+H+) 436; Rt: 1.15 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.39 (t, J=7.52 Hz, 3 H) 3.61 (q, J=7.34 Hz, 2 H) 4.82 (s, 2 H) 7.81 (d, J=8.44 Hz, 2 H) 8.09 (d, J=8.07 Hz, 1 H) 8.21 (d, J=8.44 Hz, 2 H) 8.50 (d, J=8.44 Hz, 1 H)
Step 9: Preparation of 2-[3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyll-2-pyridyll-7-
(trifluoromethyl)imidazo[1 ,2-alpyridine
In a microwave vial, 2-bromo-1-[3-ethylsulfonyl-6-[4-(trifluoromethyl)phenyl]-2-pyridyl]ethanone (0.215 g, 0.4929 mmol) and 4-(trifluoromethyl)pyridin-2-amine (95 mg, 0.591 mmol) were dissolved in acetonitrile (3.2 mL) . The vial was closed and stirred 1 h30 at 150°C under microwave conditions. LC- MS analysis showed the mixture of desired product and stating material. Reaction mixture was evaporated. The rest was dissolved in dichloromethane and washed with NaHCChsat sol. The organic layer was washed with brine, dried over Na2S04, filtered and evaporated under vacuum. The crude was purified by flash chromatography on silica gel to give the title compound.
Mpt. 230 - 231 °C
LC-MS (method 1 ): (M+H+) 500; Rt: 1.21 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.41 (t, J=7.52 Hz, 3 H) 4.10 (q, J=7.34 Hz, 2 H) 7.06 (dd, J=7.15, 1.65 Hz, 1 H) 7.78 (d, J=8.07 Hz, 2 H) 7.95 (d, J=8.44 Hz, 1 H) 7.98 (s, 1 H) 8.24 (d, J=8.07 Hz, 2 H) 8.32 (d, J=6.97 Hz, 1 H) 8.35 (s, 1 H) 8.63 (d, J=8.44 Hz, 1 H)
Example H5: Preparation of 2-[2-ethylsulfonyl-4-[3-(trifluoromethvnpyrazol-1-yllphenyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
Step 1 : Preparation of 1-(4-bromo-2-ethylsulfanyl-phenyl)ethanone
1-(4-bromo-2-fluoro-phenyl)ethanone (8.5 g, 39 mmol, CAS: [625446-22-2]) was dissolved in tetrahydrofuran (260 mL) under argon and cooled to -10°C. Then sodium thioethanolate (4.4 g, 47 mmol) was added portionally and some crystal of 18-Crown-6 was also added. Reaction mixture was stirred 1 hour at -10°C and one night at ambient temperature. After one night the LC-MS analysis showed the reaction was not completed, sodium thioethanolate (2g, 21 .3 mmol) was added at -10°C and reaction was stirred 1 h at ambient temperature .NhUCI sat sol was added in the reaction mixture (100 ml) followed by water (100 ml) and ethyl acetate (100 ml). The aqueous layer was extracted 2 times with ethyl acetate (200 ml). The organic layer was washed with water and brine, dried over Na2S04, filtered and evaporated under vacuum. The crude was purified by flash chromatography on silica gel to give the title compound as a beige solid.
LC-MS (method 1 ): (M+H+) 261 ; Rt: 1.03 min
1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 1.38 (t, J=7.52 Hz, 3 H) 2.59 (s, 3 H) 2.92 (q, J=7.46 Hz, 2 H) 7.28 - 7.33 (m, 1 H) 7.46 (d, J=1.83 Hz, 1 H) 7.65 (d, J=8.07 Hz, 1 H)
Step 2: Preparation of 1-[2-ethylsulfanyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyllethanone
In a vial, copper(l)iodide (0.038 g, 0.193 mmol) N,N'-dimethylethane-1 ,2-diamine (0.034015 g, 0.0415 mL, 0.38586 mmol) and potassium carbonate(0.108 g, 0.77 mmol) were added to a solution of 3- (trifluoromethyl)-I H-pyrazole (0.578 g, 4.2445 mmol) and 1-(4-bromo-2-ethylsulfanyl-phenyl)ethanone (1 g, 3.8586 mmol) in dimethylformamide (3.6 g, 3.8590 mL, 50 mmol).
The resulting mixture was stirred at 120°C under an argon atmosphere for 12h. LC-MS analysis showed the desired mass and reaction was not completed. N,N'-dimethylethane-1 ,2-diamine (0.034 g, 0.0415 mL, 0.38586 mmol) , copper(l)iodide (0.038 g, 0.193 mmol) and potassium carbonate (0.108 g, 0.77 mmol) were added and the resulting mixture was stirred one night more at 120°C. Reaction was stopped, cooled, filtered and evaporated. The crude was purified by flash chromatography on silica gel to give the title compound which was used without further purification for the next step.
LC-MS (method 1 ): (M+H+) 315; Rt: 1.08 min
1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 1.43 (t, J=7.34 Hz, 3 H) 2.65 (s, 3 H) 3.02 (q, J=7.58 Hz, 2 H) 6.77 (d, J=2.57 Hz, 1 H) 7.46 (dd, J=8.44, 2.20 Hz, 1 H) 7.76 (d, J=2.20 Hz, 1 H) 7.93 (d, J=8.44 Hz, 1 H) 8.02 (dd, J=2.57, 0.73 Hz, 1 H)
Step 3: Preparation of 1-[2-ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyllethanone
In a three neck flask under argon, 1-[2-ethylsulfanyl-4-[3-(trifluoromethyl)pyrazol-1-yl]phenyl]ethanone (1.02 g, 2.92 mmol) was dissolved in dichloromethane (20.4 mL) and cooled down at 0°C. Then 3- chlorobenzenecarboperoxoic acid (1.51 g, 6.13 mmol) was added and reaction was stirred 30' at 0°C then warmed up at ambient temperature and stirred one night. LC-MS analysis showed the mass of the desired product. Reaction mixture was quenched with NaOH 1 M (10 ml) and sodium thiosulfate sol ( 5ml). The aqueous layer was extracted 3 times with dichloromethane. The combined organic layer was washed with NaOH 1 M, HCI 1 M (remove amine from last step), brine, dried over Na2S04, filtered and evaporated under vacuum to give the title compound as a yellow oil.
LC-MS (method 1 ): (M+H+) 347; Rt: 0.94 min
1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 1.36 (t, J=7.34 Hz, 3 H) 2.68 (s, 3 H) 3.47 (q, J=7.34 Hz, 2 H) 6.81 (d, J=2.57 Hz, 1 H) 7.61 (d, J=8.44 Hz, 1 H) 8.08 - 8.1 1 (m, 1 H) 8.16 (dd, J=8.44, 2.20 Hz, 1 H) 8.30 (d, J=2.20 Hz, 1 H)
Step 4: Preparation of 2-bromo-1-[2-ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyllethanone
1-[2-ethylsulfonyl-4 3-(trifluoromethyl)pyrazol-1-yl]phenyl]ethanone (0.9 g, 2.599 mmol) was solved in chloroform (4.5 mL) and ethyl acetate (4.5 mL) in microwave vial and dibromocopper (1.161 g, 5.198 mmol) was added and the reaction mixture was stirred in the microwave for 50' at 140°C. LC-MS analysis showed the mass of desired product and starting material. Reaction mixture was dissolved in ethyl acetate, dried over Na2S04, filtered and evaporated under vacuum. The crude was purified by flash chromatography on silica gel and then by reverse phase to give the title compound.
LC-MS (method 1 ): (M+H+) 425/427; Rt: 1.01 min
1 H NMR (400 MHz, CHLOROFORM-d) δ ppm 1.38 (t, J=7.52 Hz, 3 H) 3.38 (q, J=7.34 Hz, 2 H) 4.52 (s, 2 H) 6.85 (d, J=2.93 Hz, 1 H) 7.72 (d, J=8.44 Hz, 1 H) 8.14 (dd, J=2.57, 0.73 Hz, 1 H) 8.19 (dd, J=8.44, 2.20 Hz, 1 H) 8.34 (d, J=2.20 Hz, 1 H)
Step 5: Preparation of 2-[2-ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yllphenyll-7- (trifluoromethyl)imidazo[1 ,2-alpyridine
In a microwave vial, 4-(trifluoromethyl)pyridin-2-amine (0.07625 g, 0.4704 mmol) and 2-bromo-1-[2- ethylsulfonyl-4-[3-(trifluoromethyl)pyrazol-1-yl]phenyl]ethanone (0.2 g, 0.4704 mmol) were dissolved in acetonitrile (4 mL) . The resulting mixture was stirred 2 hours at 150°C. LC-MS analysis showed the mass of desired product and a bit of starting material. Reaction mixture was dissolved in
dichloromethane and evaporated under vacuum. The crude was purified by flash chromatography on silica gel to give the title compound as a beige solid.
Mpt. 77 - 79 °C
LC-MS (method 1 ): (M+H+) 489; Rt: 1.08 min
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.25 (t, J=7.34 Hz, 3 H) 3.36 (q, J=7.34 Hz, 2 H) 6.85 (d, J=2.57 Hz, 1 H) 7.10 (dd, J=7.15, 1.65 Hz, 1 H) 8.00 (s, 1 H) 8.02 (s, 1 H) 8.18 (d, J=1 .47 Hz, 1 H) 8.23 (dd, J=8.44, 2.20 Hz, 1 H) 8.31 (s, 1 H) 8.34 (d, J=7.34 Hz, 1 H) 8.54 (d, J=2.57 Hz, 1 H)-
Example H6: Preparation of 2-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyll-7-iodo-imidazo[1 ,2-alpyridine
A sample of tri-n-butyl(2-pyridyl)stannane (2.35 g, 6.4 mmol, CAS [17997-47-6]) was added to a mixture of 1-(5-bromo-3-ethylsulfonyl-2-pyridyl)ethanone (1.24 g, 4.2 mmol, example H1 , step 3), Cul (122 mg, 0.64 mmol), PdCI2(PPh3)2(295 mg, 0.42 mmol) in 20 ml of 1 ,4-dioxane. The mixture was then refluxed for 2 hr and concentrated in vacuo. The crude product was purified by column chromatography on silica gel to give the title compound.
ESI-MS(+): 313(M+Na)
Ή-NMR (400Mz, CDCIs) δ (ppm): 1.37 (t, 3 H), 2.75 (s, 3 H), 3.60 (q, 2 H), 7.38(s, 1 H), 7.85(s, 2 H), 8.77(s, 1 H), 8.94 (s, 1 H), 9.42(s, 1 H).
Step 2: Preparation of 2-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyll-7-iodo-imidazo[1 ,2-alpyridine
A mixture of 1-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyl]ethanone (499 mg, 1.72 mmol), 4-iodopyridin-2- amine (490 mg, 2.23 mmol), Cu(OAc)2.H2O(40 mg,0.2 mmol), Znl2(65 mg, 0.2 mmol) and o- phenanthroline(36 mg, 0.2 mmol) in 10 ml of 1 , 2-dichlorobenzene was stirred at 135°C for 16 h. Then the reaction mixture was purified by column chromatography on silica gel to give the title compound. Mpt. 230 - 232 °C
LC-MS (method 1 ): (M+H+) 491 ; Rt: 0.88 min
Ή-NMR (400Mz, CDCI3) δ (ppm): 1.34(t, 3 H), 3.91 (q, 2 H), 7.08(d, 1 H), 7.35(t, 1 H), 7.85(m, 2 H), 7.93(d, 1 H), 8.08(s, 1 H), 8.76(d, 1 H), 9.07(s, 1 H), 9.50(s, 1 H).
Example H7: Preparation of 2-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyll-7-(1 , 1 ,2,2,2- pentafluoroethyl)imidazo[1 ,2-alpyridine
A solution of compound 2-[3-ethylsulfonyl-5-(2-pyridyl)-2-pyridyl]-7-iodo-imidazo[1 ,2-a]pyridine (70 mg, 0.14 mmol) and (1 , 1 ,2,2,2-pentafluoroethyl)(1 , 10-phenanthroline- κΝ1 , KN10)-copper (104 mg, 0.28 mmol, purchased from Aspira scientific) in 4 ml of NMP was stirred in a microwave 1 10 °C for 2 hr. The mixture was cooled and purified by column chromatography on silica gel to give the title compound.
Mpt. 222 - 224 °C
LC-MS (method 1 ): (M+H+) 483; Rt: 1.00 min
Ή-NMR (400Mz, DMSO-de) δ (ppm): 1.27(t, 3 H), 4.24(q, 2 H), 7.24(d, 1 H), 7.48(m, 1 H), 7.99(m, 1 H), 8.15(s, 1 H), 8.24(d, 1 H), 8.64(s, 1 H), 8.77(d, 1 H), 8.92(s, 1 H), 9.10(s, 1 H), 9.55(s, 1 H ). 9F-NMR (400Mz,DMSO-d6):-81.45(s, 3F), -1 1 1 .88(s, 2F);
Example H8: Preparation of 2-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyll-7-iodo- imidazoM ,2-alpyridine:
F
In a supelco vial, copper(l)iodide (0.1 10 g, 0.561 mmol) N,N'-dimethylethane-1 ,2-diamine (0.0989 g, 1.12 mmol) and potassium carbonate (0.313 g, 2.24 mmol) were added to a solution of 3-
(trifluoromethyl)-l h-pyrazole (0.840 g, 6.17 mmol) and 1-(5-bromo-3-ethylsulfanyl-2-pyridyl)ethanone (1.46 g, 5.61 mmol, prepared as described in step 2, example H1 ) in dimethylformamide (6 mL). The resulting mixture was stirred at 120°C under an argon atmosphere for 48h. Aqueous work-up and purification of the crude product by Combi flash chromatography with a column of 24 g and a gradient cyclohexane 0-30 % ethylacetate gave the title compound as a white solid.
LCMS (method 1 ); Rt= 1.09 min, [M+H] 316.
Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm: 1.40 (t, J=7.52 Hz, 3 H); 4.00 (q, J=7.46 Hz, 2 H); 7.97 (s, 1 H); 8.36 (s, 1 H); 8.69 (d, J=2.20 Hz, 1 H); 8.95 (d, J=2.20 Hz, 1 H); 9.17 (s, 1 H). Step 2: 1-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyllethanone:
A solution of 1 3-ethylsulfanyl-5-[3-(trifluoromethyl)pyrazol-1-yl]-2-pyridyl]ethanone (1.36 g, 3.88 mmol) in dichloromethane (27ml_) was cooled to 0°C and treated portionwise with meta- chloroperoxybenzoic acid (1.79 g, 7.76 mmol) . The reaction mixture was stirred for 30min at 0°C then warmed up to ambient temperature and stirred for a further 3 hours. Reaction mixture was quenched with NaOH 1 M ( 10 ml ) and sodium thiosulfate sol ( 5ml). The aqueous layer was extracted 3 times with dichloromethane. The combined organic layer was washed successively with NaOH 1 M, and brine, dried over Na2S04, filtered and concentrated in vacuo his gave the title compound as a yellow solid.
Mpt: 82-84°C.
LCMS (method 1 ); Rt= 0.96 min, [M+H] 348. Ή NMR (400 MHz, CHLOROFORM-c/) δ ppm 1.40 (t, J=7.52 Hz, 3 H) 2.76 (s, 3 H) 3.68 (q, J=7.34 Hz, 2 H) 6.87 (d, J=2.57 Hz, 1 H) 8.15 (dd, J=2.75, 0.92 Hz, 1 H) 8.68 (d, J=2.57 Hz, 1 H) 9.27 (d, J=2.57 Hz, 1 H).
Step 3: Preparation of 2-bromo-1-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyllethanone
To a solution of 1-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yl]-2-pyridyl]ethanone (837 mg, 2.41 mmol) dissolved in 4 ml_ 4 mL chloroform/ethyl acetate, was added copper bromide (967 mg, 4.33 mmol). The mixture was stirred at 130 °C for 1.5 h. The mixture was then filtered through celite, and the filtrate was evaporated to dryness. The residue was purified by chromatography (petroleum ether/ethyl acetate=6: 1 ) on silica to give the title compound.
Step 3: Preparation of 2-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyll-7-iodo- imidazoM ,2-alpyridine:
ln a MW vial, 2-bromo-1-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yl]-2-pyridyl]eth (938 mg, 1.1 mmol, wt: 50 %) and 4-iodopyridin-2-amine (266 mg, 1.21 mmol) were added to acetonitrile (8 ml_),and the mixture was stirred at 140 °C under microwave conditions for 2 hr. The mixture was evaporated to dryness and the residue wpurified by chromatography (petroleum ether/ethyl acetate=1/3) on silica to give the title compound as brown solid.
Mpt. 209 - 211 °C
LCMS (method 1 ); Rt= 1.02 min, [M+H] 548.
Ή-NMR (400MHz, de-DMSO): δ (ppm): 9.46 (s, 1 H), 9.01 (d, J=1.2Hz, 1 H), 8.77 (d, J=2Hz, 1 H), 8.48 (s, 1 H), 8.44 (d, J=6.8Hz, 1 H), 8.14 (s, 1 H), 7.25 (d, J=7.2Hz, 1 H), 7.17 (d, J=2.4Hz, 1 H), 4.26 (q, J=7.6 Hz, 2 H), 1.28 (t, J=7.6Hz, 3 H).
9F-NMR (400MHz, de-DMSO) δ (ppm): -62.98 (s, 3F).
Example H9: Preparation of 2-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyll-7-
(trifluoromethylsulfanyl)imidazo[1 ,2-alpyridine
To a solution of 2-bromo-1-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yl]-2-pyridyl]ethanone (400 mg, 0.73 mmol) in 18 ml_ of acetonitrile, was added
,1 , 1-trifluoromethanethio lato- S)-copper (467 mg, 1.46 mmol, purchased from Aspira Scientific), and the mixture was stirred at
70 °C for 2 h under nitrogen atmosphere. The mixture was cooled to rt, evaporated to dryness. The residue was purified by chromatography (petroleum ether/ethyl acetate=1/3) on silica to get the desire product A18-5 as white solid.
Mpt. 229-231 °C
LCMS (method 1 ); Rt= 1.04 min, [M+H] 523.
Ή-NMR (400MHz, de-DMSO): δ (ppm): 9.48 (s, 1 H), 9.02 (s, 1 H), 8.80 (s, 1 H), 8.77-8.75 (m, 1 H), 8.63 (s, 1 H), 8.18 (d, J=3.2 Hz, 1 H), 7.22-7.18 (m, 2 H), 4.27 (q, J=7.6 Hz, 2 H), 1.28 (t, J=7.6 Hz, 3 H).
9F-NMR (300MHz, d6-DMSO) δ -39.44 ppm (s, 3F), -58.81 ppm (s, 3F).
Example H10: Preparation of 2-[3-ethylsulfonyl-5-[3-(trifluoromethyl)pyrazol-1-yll-2-pyridyll-3,7-diiodo- imidazoM ,2-alpyridine.
A solution of 1-[5-[3-(trifluoromethyl)pyrazol-1-yl]-2-pyridyl]ethanone (207 mg, 0.941 mmol, prepared as described in step 2, example H1 1 ) in 3 mL of 1 ,2-dichlorobenzene was tretated with 4-iodo-pyridin-2- amine (200 mg, 0.784 mmol), cupric acetate monohydrate (25 mg, 0.125 mmol), zinc iodide (50 mg, 0.1568 mmol) and 1 , 10-phenanthroline (28 mg, 0.1568 mmol) and the mixture stirred at 130 °C for 24 hr. The mixture was evaporated to dryness, and the residue purified by chromatography eluting with petroleum ether/ethyl acetate, to give the title compound as a white solid.
Mpt. 231 - 232 °C
LCMS (method 1 ); Rt= 1.10 min, [M+H] 674.
Ή NMR (400MHz, CDCIs): δ (ppm): 9.38(d, J=2 Hz, 1 H), 8.77 (d, J=2.4Hz, 1 H), 8.16 (d, J=2.8 Hz, 1 H), 8.03-7.98 (m, 2 H), 7.24-7.22 (m, 1 H), 6.85 (d, J=2.4 Hz, 1 H), 3.99 (q, J=7.6 Hz, 2 H), 1.29 (t, J=7.6 Hz, 3 H). HPLC: 95.6%.
9F NMR (400MHz, CDCI3) δ (ppm): -60.88 (s, 3F).
Example H11 : Preparation of 2-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyll-7-iodo- imidazoM ,2-alpyridine
Step 1 : Preparation of 1-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyllethanone
To a stirred solution of 1-(5-bromo-3-ethylsulfonyl-2-pyridyl)ethanone (582 mg, 2 mmol, prepared as described in step 3, example H1 ), tributyl-[4-(trifluoromethyl)-2-pyridyl]stannane
(874 mg, 4 mmol, CAS [1334675-40-9]) in dioxane (35 mL) was added Cul (57 mg, 0.3 mmol), PdCl2(PPh3)2 (140 mg, 0.2 mmol). The reaction system was refluxed under an nitrogen atmosphere at 120 °C for 24 hr. After cooling to ambient temperature, the reaction mixture was filtered and
concentrated in vacuo. The crude product was purified by column chromatography on silica gel (eluting with petroleum: EtOAc = 4: 1 ) to give the title compound.
HNMR (400 MHz, CDCh): δ (ppm) 1 .41 (t, 3 H), 2.76 (s, 3 H), 3.63 (q, 2 H), 7.79 (s, 1 H), 8.04 (s, 1 H), 8.97 (d, 2 H), 8.47 (d, 1 H).
9FNMR (376 MHz, CDCh): δ (ppm) -63.59 (s, 3 F).
Step 2: Preparation of 2-bromo-1-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyllethanone
To a stirred solution of 1-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyl]-2-pyridyl]ethanone (358 mg, 1 mmol) in CH3CN (5 ml) and CHCI3 (5 ml) was added CuBr2 (446 mg, 2 mmol). The reaction system was refluxed in a sealed tube at 130 °C for 4 hr. After cooling to ambient temperature, the reaction mixture was filtered and concentrated in vacuo. The crude product was purified by column
chromatography on silica gel (eluting with petroleum: EtOAc = 8:1 ) to give the tile compound.
H-NMR (400 MHz, CDCh): δ (ppm) 1.40 (t, 3 H), 3.64 (q, 2 H), 4.77 (s, 2 H), 7.63 (d, 1 H), 8.04 (s, 1 H), 8.96 (d, 1 H), 9.01 (s, 1 H), 9.48 (s, 1 H).
9FNMR (376 MHz, CDCh): δ (ppm) -63.76 (s, 3 F).
Step 3: Preparation of 2-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyll-2-pyridyll-7-iodo-imidazo[1 ,2- al pyridine
A mixture of 2-bromo-1-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyl]-2-pyridyl]ethanone (196 mg, 0.45 mmol) and 4-iodopyridin-2-amine (198 mg, 0.9 mmol) in 5 ml CH3CN was stirred under microwave conditions at 135 °C for 2 hr. After cooling to ambient temperature, the reaction mixture was filtered and concentrated in vacuo. The crude product was purified by column chromatography on silica gel (eluting with petroleum: EtOAc = 4: 1 ) to give the title compound as a white solid.
Mpt. 247-249 °C
LCMS (method 1 ); Rt= 1.03 min, [M+H] 559.
H-NMR (400 MHz, DMSO-de): δ (ppm) 1.29 (t, 3 H), 4.29 (q, 2 H), 7.28 (d, 1 H), 7.88 (d, 1 H), 8.16 (s, 1 H), 8.48 (d, 1 H), 8.55 (s, 1 H), 8.64 (s, 1 H), 9.06 (d, 1 H), 9.17 (s, 1 H), 9.69 (s, 1 H).
9FNMR (376 MHz, CDCh): δ (ppm) -60.91 (s, 3 F); ESI-MS: 559 (M+H).
Example H12: Preparation of 2-[3-ethylsulfonyl-5-[4-(trifluoromethvn-2-pyridyll-2-pyridyll-7- (trifluoromethylsulfanyl)imidazo[1 ,2-alpyridine
A solution of (2,2'-bipyridine-KA/' ,κΛ/' )(1 , 1 , 1 -trifiuoromethanethiolato-KS)-copper (160 mg, 0.5 mmol) and 2-[3-ethylsulfonyl-5-[4-(trifluoromethyl)-2-pyridyl]-2-pyridyl]-7-iodo-imidazo[1 ,2-a]pyridine (1 12 mg, 0.2mmol) in 20 ml of Ch CN was refluxed for 8 hr under nitrogen. The reaction mixture was removed from the oil bath and allowed to cool and then filtered through S1O2, eluted with diethyl ether, washed with brine, and concentrated in vacuo. The residue was purified by silica gel column chromatography (eluting with petroleum: EtOAc = 4: 1 ) to give the title compound as a white solid Mpt. 226-228 °C
LCMS (method 1 ); Rt= 1.10 min, [M+H] 533.
Ή-NMR (400 MHz, CDCh): δ (ppm) 1 .43 (t, 3 H), 4.04 (q, 2 H), 7.10 (d, 1 H), 7.61 (d, 1 H), 8.06 (d, 1 H), 8.08 (s, 1 H), 8.25 (d, 1 H), 8.40 (s, 1 H), 8.97 (s, 1 H), 9.16 (s, 1 H), 9.57 (s, 1 H);
9F-NMR (376 MHz, CDCh): δ (ppm) -60.91 (s, 3 F), -40.10 (s, 3F).
Example H13: Preparation of 2-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyll-7- (trifluorometh l)imidazo[1 ,2-alpyridine
Step 1 : Preparation of 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylic acid
CI
To a stirred solution of methyl 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylate (1 1.55 g, 50 mmol, prepared as described in step 2, example H4) in THF (50 mL) was added NaOH (8 g, 200 mmol) andhbO (150 ml). The reaction system was stirred at ambient temperature for 4 hr. The pH value was adjusted to 2 with HCI and the reaction mixture extracted with ethyl acetate three times. The combined organic layers were dried over anhydrous sodium sulfate. After filtration and concentration in vacuo, the title compound was obtained as a white solid.
ESI-MS(-): 216(M-H).
HNMR (400 MHz, DMSO-c/6): δ ppm 1.22 (t, 3 H), 2.98 (q, 2 H), 7.60 (d, 1 H), 7.92 (d, 1 H), 13.5 (bs, 1 H).
Step 2: Preparaton of 6-chloro-3-ethylsulfanyl-pyridine-2-carbonyl chloride
To a stirred solution of methyl 6-chloro-3-ethylsulfanyl-pyridine-2-carboxylate (4.36 g, 20 mmol) in CH2CI2 (60 mL) was added oxalyl chloride (5.08 g, 40 mmol) and a drop of DMF. The reaction system was stirred at ambient temperature for 24 h. Then the reaction mixture was concentrated in vacuo to give the crude title compound which was used in the next step without further purification.
Step 3: Preparaton of 6-chloro-3-ethylsulfanyl-N-methoxy-N-methyl-pyridine-2-carboxamide
To a stirred solution of 6-chloro-3-ethylsulfanyl-pyridine-2-carbonyl chloride (4.7 g, 20 mmol) in anhydrous THF (100 mL) was added N-methoxymethylamine (2.48 g, 40 mmol) Et3N (15 ml) at 0 °C and the reaction mixtures was stirred at ambient temperature for 16 hr. The reaction mixture was poured into water, and extracted with ethyl acetate three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated in vacuo. The crude product was purified by column chromatography on silica gel (eluting with petroleum: EtOAc = 4: 1 ) to give the title compound as a white solid.
ESI-MS(+): 283(M+Na).
HNMR (400 MHz, DMSO-c/6): 5ppm 1.22 (t, 3 H), 2.87 (q, 2 H), 3.31 (s, 3 H) 3.54 (s, 3 H), 7.26 (d, 1 H), 7.67 (d, 1 H).
Step 4: Preparation of 1-(6-chloro-3-ethylsulfanyl-2-pyridyl)ethanone
To a solution of 6-chloro-3-ethylsulfanyl-N-methoxy-N-methyl-pyridine-2-carboxamide (2.6 g, 10 mmol) in 30 mL of dry tetrahydrofurane at 0 °C was added Ch MgBr (10 mL of a 3 M solution in hexane, 30 mmol) under an nitrogen atmosphere. After stirring for 30 min at 0 °C, the mixture was stirred at ambient temperature for 16 hr. The mixture was then poured into dilute hydrochloric acid and extracted with ethyl acetate three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated in vacuo. The crude product was purified by column chromatography on silica gel (eluting with petroleum: EtOAc = 4: 1 ) to give the title compound.
ESI-MS(+): 247 (M +CH3OH)+.
Ή-NMR (400 MHz, CDCh): δ ppm 1.39 (t, 3 H), 2.69 (q, 2 H), 2.90 (s, 3 H), 7.39 (d, 1 H), 7.65 (d, 1 H).
Step 5: Preparation of 1-(6-chloro-3-ethylsulfonyl-2-pyridyl)ethanone
A solution of 1-(6-chloro-3-ethylsulfanyl-2-pyridyl)ethanone (1.075 g, 5 mmol) in 40 ml of CH2CI2 was treated with m-Chloroperbenzoic acid (2.58 g, 15 mmol) at ambient temperature and allowed to stir at this temperature for 2 hr. The mixture was then poured into a saturated aqueous solution of NaHCCh and Na2S03 i, and extracted with CH2CI2 three times. The combined organic layers were dried over sodium sulfate, filtered and concentrated in vacuo.TUe crude product was purified by column chromatography on silica gel (eluting with petroleum: EtOAc = 2:1 ) to give the title compound.
ESI-MS(+): 270 (M +Na)+.
Ή-NMR (400 MHz, CDCh): δ (ppm) 1.33 (t, 3 H), 2.68 (s, 3 H), 3.52 (q, 2 H), 7.56 (d, 1 H), 8.27 (d, 1
H); Step 6: Preparation of 1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyllethanone
To a solution of 1-(6-chloro-3-ethylsulfonyl-2-pyridyl)ethanone (2.00 g, 8.10 mmol, 1.00 equiv.) in acetonitrile (15 mL) was sequentially added 1 ,2,4-triazole (670 mg, 9.70 mmol, 1.20 equiv.) and K2CO3 (3.30 g, 24.0 mmol, 3.00 equiv.). the obtained reaction mixture was heated to 80 °C under microwave irradiation for 15 min. After cooling to ambient temperature, the reaction mixtutre was filtered and the filtrate concentrated in vacuo. The crude product was purified by column chromatography on silica gel to give the title compound.
LCMS (method 1 ); Rt= 0.71 min, [M+H+] 281.
Step 7: Preparation of tert-butyl-[1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyllvinyloxyl-dimethyl- silane
To a solution of 1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyl]ethanone (5.90 g, 21.0 mmol, 1.00 equiv.) in dichloromethane (74 mL) was added trimethylamine (4.70 mL, 3.41 g, 33.7 mmol, 1.60 equiv.) followed by [tert-butyl(dimethyl)silyl] trifluoromethanesulfonate (6.41 mL, 7.38 g, 27.4 mmol, 1.30 equiv.) at ambient temperature. The obtained reaction mixture was heated under reflux overnight. After cooling to ambient temperature, the volatiles were removed in vacuo and the residual was dissolved in dichloromethane (40 mL), washed with water (3x) and brine. Drying (Na2S04) and concentration in vacuo furnished the crude material which was purified by flash chromatography (silica gel, cyclohexane / Et3N / EtOAc) to deliver the desired product as an oil.
LCMS (method 1 ); Rt= 1 .13 min, [M+H+] 395.
To a solution of of tert-butyl-[1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyl]vinyloxy]-dimethyl-silane (660 mg, 1.67 mmol, 1.00 equiv.) in THF (23 mL) was added phosphate buffer (pH 7, 3.8 mL of a 0.5M solution) and the obtained mixture was cooled to 0 °C. At this temperature, N-bromosuccinimide (304 mg, 1.67 mmol, 1.00 equiv.) was added inone portion. After stirring for at 0 °C for 3 min, the reaction mixture was diluted with additional phosphate buffer (pH 7, 10 mL of a 0.5M solution) and ieri-butyl methyl ether (10 mL). The organic layer was separated, washed with water (3x), dried (Na2S04) and concentrated in vacuo. The obtained tan solid was suspended small amounts of TBME amnd filtered (3x). The remaining solid was then dissolved in chloroform, washed with water (2x) and brine, dried (Na2S04) to obtain the crude title compound which was used in the next step without further purification.
LCMS (method 1 ); Rt= 0.78 min, [M+H+] 359.
Step 9: Preparation of 2-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2-pyridyll-7-(trifluoromethyl)imidazo[1 ,2-
A microwave vial was charged with a mixture of 2-bromo-1-[3-ethylsulfonyl-6-(1 ,2,4-triazol-1-yl)-2- pyridyljethanone (390 mg, 1 .08 mmol, 1.00 equiv.) and 4-(trifluoromethyl)pyridin-2-amine (194 mg, 1.19 mmol, 1.10 equiv.) in acetonitrile (4.0 mL). The vial was sealed and heated to 150 °C under microwave irradiation for 30 min. The obtained crude mixture was concentrated in vacuo and subjected to flash chromatography (silica gel, cyclohexane/ethyl acetate) to obtain the title compound as a white solid.
LCMS (method 1 ); Rt= 0.93 min, [M+H+] 423.
Table P: Examples of compounds of formula (I).
All other compounds listed in the tables above can be prepared in analogous methods to those described here in the experiment, and using methods known to those skilled in the art. The activity of the compositions according to the invention can be broadened considerably, and adapted to prevailing circumstances, by adding other insecticidally, acaricidally and/or fungicidally active ingredients. The mixtures of the compounds of formula I with other insecticidally, acaricidally and/or fungicidally active ingredients may also have further surprising advantages which can also be described, in a wider sense, as synergistic activity. For example, better tolerance by plants, reduced phytotoxicity, insects can be controlled in their different development stages or better behaviour during their production, for example during grinding or mixing, during their storage or during their use.
Suitable additions to active ingredients here are, for example, representatives of the following classes of active ingredients: organophosphorus compounds, nitrophenol derivatives, thioureas, juvenile hormones, formamidines, benzophenone derivatives, ureas, pyrrole derivatives, carbamates, pyrethroids, chlorinated hydrocarbons, acylureas, pyridylmethyleneamino derivatives, macrolides, neonicotinoids and Bacillus thuringiensis preparations.
The following mixtures of the compounds of formula I with active ingredients are preferred (the abbreviation "TX" means "one compound selected from the group consisting of the compounds described in Table P of the present invention"): an adjuvant selected from the group of substances consisting of petroleum oils (628) + TX,
an acaricide selected from the group of substances consisting of 1 , 1-bis(4-chlorophenyl)-2- ethoxyethanol (lUPAC name) (910) + TX,2,4-dichlorophenyl benzenesulfonate (lUPAC/Chemical Abstracts name) (1059) + TX,2-fluoro-A/-methyl-A/-1-naphthylacetamide (lUPAC name) (1295) + TX,4- chlorophenyl phenyl sulfone (lUPAC name) (981 ) + TX, abamectin (1 ) + TX, acequinocyl (3) + TX, acetoprole [CCN] + TX, acrinathrin (9) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, alpha- cypermethrin (202) + TX, amidithion (870) + TX, amidoflumet [CCN] + TX, amidothioate (872) + TX, amiton (875) + TX, amiton hydrogen oxalate (875) + TX, amitraz (24) + TX, aramite (881 ) + TX, arsenous oxide (882) + TX, AVI 382 (compound code) + TX, AZ 60541 (compound code) + TX, azinphos-ethyl (44) + TX, azinphos-methyl (45) + TX, azobenzene (lUPAC name) (888) + TX, azocyclotin (46) + TX, azothoate (889) + TX, benomyl (62) + TX, benoxafos [CCN] + TX, benzoximate (71 ) + TX, benzyl benzoate (lUPAC name) [CCN] + TX, bifenazate (74) + TX, bifenthrin (76) + TX, binapacryl (907) + TX, brofenvalerate + TX, bromocyclen (918) + TX, bromophos (920) + TX, bromophos-ethyl (921 ) + TX, bromopropylate (94) + TX, buprofezin (99) + TX, butocarboxim (103) + TX, butoxycarboxim (104) + TX, butylpyridaben + TX, calcium polysulfide (lUPAC name) (1 1 1 ) + TX, camphechlor (941 ) + TX, carbanolate (943) + TX, carbaryl (1 15) + TX, carbofuran (1 18) + TX, carbophenothion (947) + TX, CGA 50'439 (development code) (125) + TX, chinomethionat (126) + TX, chlorbenside (959) + TX, chlordimeform (964) + TX, chlordimeform hydrochloride (964) + TX, chlorfenapyr (130) + TX, chlorfenethol (968) + TX, chlorfenson (970) + TX, chlorfensulfide (971 ) + TX, chlorfenvinphos (131 ) + TX, chlorobenzilate (975) + TX, chloromebuform (977) + TX, chloromethiuron (978) + TX, chloropropylate (983) + TX, chlorpyrifos (145) + TX, chlorpyrifos-methyl (146) + TX, chlorthiophos (994) + TX, cinerin I (696) + TX, cinerin II (696) + TX, cinerins (696) + TX, clofentezine (158) + TX, closantel [CCN] + TX, coumaphos (174) + TX, crotamiton [CCN] + TX, crotoxyphos (1010) + TX, cufraneb (1013) + TX, cyanthoate (1020) + TX, cyflumetofen (CAS Reg. No.: 400882-07- 7) + TX, cyhalothrin (196) + TX, cyhexatin (199) + TX, cypermethrin (201 ) + TX, DCPM (1032) + TX, DDT (219) + TX, demephion (1037) + TX, demephion-0 (1037) + TX, demephion-S (1037) + TX, demeton (1038) + TX, demeton-methyl (224) + TX, demeton-0 (1038) + TX, demeton-O-methyl (224) + TX, demeton-S (1038) + TX, demeton-S-methyl (224) + TX, demeton-S-methylsulfon (1039) + TX, diafenthiuron (226) + TX, dialifos (1042) + TX, diazinon (227) + TX, dichlofluanid (230) + TX, dichlorvos (236) + TX, dicliphos + TX, dicofol (242) + TX, dicrotophos (243) + TX, dienochlor (1071 ) + TX, dimefox (1081 ) + TX, dimethoate (262) + TX, dinacti (653) + TX, dinex (1089) + TX, dinex- diclexine (1089) + TX, dinobuton (269) + TX, dinocap (270) + TX, dinocap-4 [CCN] + TX, dinocap-6 [CCN] + TX, dinocton (1090) + TX, dinopenton (1092) + TX, dinosulfon (1097) + TX, dinoterbon (1098) + TX, dioxathion (1 102) + TX, diphenyl sulfone (lUPAC name) (1 103) + TX, disulfiram [CCN] + TX, disulfoton (278) + TX, DNOC (282) + TX, dofenapyn (1 1 13) + TX, doramectin [CCN] + TX, endosulfan (294) + TX, endothion (1 121 ) + TX, EPN (297) + TX, eprinomectin [CCN] + TX, ethion (309) + TX, ethoate-m ethyl (1 134) + TX, etoxazole (320) + TX, etrimfos (1 142) + TX, fenazaflor (1 147) + TX, fenazaquin (328) + TX, fenbutatin oxide (330) + TX, fenothiocarb (337) + TX, fenpropathrin (342) + TX, fenpyrad + TX, fenpyroximate (345) + TX, fenson (1 157) + TX, fentrifanil (1 161 ) + TX, fenvalerate
(349) + TX, fipronil (354) + TX, fluacrypyrim (360) + TX, fluazuron (1 166) + TX, flubenzimine (1 167) + TX, flucycloxuron (366) + TX, flucythrinate (367) + TX, fluenetil (1 169) + TX, flufenoxuron (370) + TX, flumethrin (372) + TX, fluorbenside (1 174) + TX, fluvalinate (1 184) + TX, FMC 1 137 (development code) (1 185) + TX, formetanate (405) + TX, formetanate hydrochloride (405) + TX, formothion (1 192) + TX, formparanate (1 193) + TX, gamma-HCH (430) + TX, glyodin (1205) + TX, halfenprox (424) + TX, heptenophos (432) + TX, hexadecyl cyclopropanecarboxylate (lUPAC/Chemical Abstracts name) (1216) + TX, hexythiazox (441 ) + TX, iodomethane (lUPAC name) (542) + TX, isocarbophos (473) + TX, isopropyl 0-(methoxyaminothiophosphoryl)salicylate (lUPAC name) (473) + TX, ivermectin [CCN] + TX, jasmolin I (696) + TX, jasmolin II (696) + TX, jodfenphos (1248) + TX, lindane (430) + TX, lufenuron (490) + TX, malathion (492) + TX, malonoben (1254) + TX, mecarbam (502) + TX, mephosfolan (1261 ) + TX, mesulfen [CCN] + TX, methacrifos (1266) + TX, methamidophos (527) + TX, methidathion (529) + TX, methiocarb (530) + TX, methomyl (531 ) + TX, methyl bromide (537) + TX, metolcarb (550) + TX, mevinphos (556) + TX, mexacarbate (1290) + TX, milbemectin (557) + TX, milbemycin oxime [CCN] + TX, mipafox (1293) + TX, monocrotophos (561 ) + TX, morphothion (1300) + TX, moxidectin [CCN] + TX, naled (567) + TX, NC-184 (compound code) + TX, NC-512 (compound code) + TX, nifluridide (1309) + TX, nikkomycins [CCN] + TX, nitrilacarb (1313) + TX, nitrilacarb 1 : 1 zinc chloride complex (1313) + TX, NNI-0101 (compound code) + TX, NNI-0250 (compound code) + TX, omethoate (594) + TX, oxamyl (602) + TX, oxydeprofos (1324) + TX, oxydisulfoton (1325) + TX, pp'-DDT (219) + TX, parathion (615) + TX, permethrin (626) + TX, petroleum oils (628) + TX, phenkapton (1330) + TX, phenthoate (631 ) + TX, phorate (636) + TX, phosalone (637) + TX, phosfolan (1338) + TX, phosmet (638) + TX, phosphamidon (639) + TX, phoxim (642) + TX, pirimiphos-methyl (652) + TX, polychloroterpenes (traditional name) (1347) + TX, polynactins (653) + TX, proclonol (1350) + TX, profenofos (662) + TX, promacyl (1354) + TX, propargite (671 ) + TX, propetamphos (673) + TX, propoxur (678) + TX, prothidathion (1360) + TX, prothoate (1362) + TX, pyrethrin I (696) + TX, pyrethrin II (696) + TX, pyrethrins (696) + TX, pyridaben (699) + TX, pyridaphenthion (701 ) + TX, pyrimidifen (706) + TX, pyrimitate (1370) + TX, quinalphos (71 1 ) + TX, quintiofos (1381 ) + TX, R-1492 (development code) (1382) + TX, RA-17 (development code) (1383) + TX, rotenone (722) + TX, schradan (1389) + TX, sebufos + TX, selamectin [CCN] + TX, SI-0009 (compound code) + TX, sophamide (1402) + TX, spirodiclofen (738) + TX, spiromesifen (739) + TX, SSI-121 (development code) (1404) + TX, sulfiram [CCN] + TX, sulfluramid (750) + TX, sulfotep (753) + TX, sulfur (754) + TX, SZI-121 (development code) (757) + TX, tau-fluvalinate (398) + TX, tebufenpyrad (763) + TX, TEPP (1417) + TX, terbam + TX, tetrachlorvinphos (777) + TX, tetradifon (786) + TX, tetranactin (653) + TX, tetrasul (1425) + TX, thiafenox + TX, thiocarboxime (1431 ) + TX, thiofanox (800) + TX, thiometon (801 ) + TX, thioquinox (1436) + TX, thuringiensin [CCN] + TX, triamiphos (1441 ) + TX, triarathene (1443) + TX, triazophos (820) + TX, triazuron + TX, trichlorfon
(824) + TX, trifenofos (1455) + TX, trinactin (653) + TX, vamidothion (847) + TX, vaniliprole [CCN] and YI-5302 (compound code) + TX,
an algicide selected from the group of substances consisting of bethoxazin [CCN] + TX, copper dioctanoate (lUPAC name) (170) + TX, copper sulfate (172) + TX, cybutryne [CCN] + TX, dichlone (1052) + TX, dichlorophen (232) + TX, endothal (295) + TX, fentin (347) + TX, hydrated lime [CCN] + TX, nabam (566) + TX, quinoclamine (714) + TX, quinonamid (1379) + TX, simazine (730) + TX, triphenyltin acetate (lUPAC name) (347) and triphenyltin hydroxide (lUPAC name) (347) + TX, an anthelmintic selected from the group of substances consisting of abamectin (1 ) + TX, crufomate (101 1 ) + TX, doramectin [CCN] + TX, emamectin (291 ) + TX, emamectin benzoate (291 ) + TX, eprinomectin [CCN] + TX, ivermectin [CCN] + TX, milbemycin oxime [CCN] + TX, moxidectin [CCN] + TX, piperazine [CCN] + TX, selamectin [CCN] + TX, spinosad (737) and thiophanate (1435) + TX, an avicide selected from the group of substances consisting of chloralose (127) + TX, endrin (1 122) + TX, fenthion (346) + TX, pyridin-4-amine (lUPAC name) (23) and strychnine (745) + TX,
a bactericide selected from the group of substances consisting of 1-hydroxy-1 /- -pyridine-2-thione (lUPAC name) (1222) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (lUPAC name) (748) + TX, 8-hydroxyquinoline sulfate (446) + TX, bronopol (97) + TX, copper dioctanoate (lUPAC name) (170) + TX, copper hydroxide (lUPAC name) (169) + TX, cresol [CCN] + TX, dichlorophen (232) + TX, dipyrithione (1 105) + TX, dodicin (1 1 12) + TX, fenaminosulf (1 144) + TX, formaldehyde (404) + TX, hydrargaphen [CCN] + TX, kasugamycin (483) + TX, kasugamycin hydrochloride hydrate (483) + TX, nickel bis(dimethyldithiocarbamate) (lUPAC name) (1308) + TX, nitrapyrin (580) + TX, octhilinone (590) + TX, oxolinic acid (606) + TX, oxytetracycline (61 1 ) + TX, potassium hydroxyquinoline sulfate (446) + TX, probenazole (658) + TX, streptomycin (744) + TX, streptomycin sesquisulfate (744) + TX, tecloftalam (766) + TX, and thiomersal [CCN] + TX,
a biological agent selected from the group of substances consisting of Adoxophyes orana GV (12) + TX, Agrobacterium radiobacter (13) + TX, Amblyseius spp. (19) + TX, Anagrapha falcifera NPV (28) + TX, Anagrus atomus (29) + TX, Aphelinus abdominalis (33) + TX, Aphidius colemani (34) + TX, Aphidoletes aphidimyza (35) + TX, Autographa californica NPV (38) + TX, Bacillus firmus (48) + TX, Bacillus sphaericus Neide (scientific name) (49) + TX, Bacillus thuringiensis Berliner (scientific name) (51 ) + TX, Bacillus thuringiensis subsp. aizawai (scientific name) (51 ) + TX, Bacillus thuringiensis subsp. israelensis (scientific name) (51 ) + TX, Bacillus thuringiensis subsp. japonensis (scientific name) (51 ) + TX, Bacillus thuringiensis subsp. kurstaki (scientific name) (51 ) + TX, Bacillus thurin- giensis subsp. tenebrionis (scientific name) (51 ) + TX, Beauveria bassiana (53) + TX, Beauveria brongniartii (54) + TX, Chrysoperla carnea (151 ) + TX, Cryptolaemus montrouzieri (178) + TX, Cydia pomonella GV (191 ) + TX, Dacnusa sibirica (212) + TX, Diglyphus isaea (254) + TX, Encarsia formosa (scientific name) (293) + TX, Eretmocerus eremicus (300) + TX, Helicoverpa zea NPV (431 ) + TX, Heterorhabditis bacteriophora and H. megidis (433) + TX, Hippodamia convergens (442) + TX, Leptomastix dactylopii (488) + TX, Macrolophus caliginosus (491 ) + TX, Mamestra brassicae NPV (494) + TX, Metaphycus helvolus (522) + TX, Metarhizium anisopliae var. acridum (scientific name) (523) + TX, Metarhizium anisopliae var. anisopliae (scientific name) (523) + TX, Neodiprion sertifer NPV and N. lecontei NPV (575) + TX, Orius spp. (596) + TX, Paecilomyces fumosoroseus (613) +
TX, Phytoseiulus persimilis (644) + TX, Spodoptera exigua multicapsid nuclear polyhedrosis virus (scientific name) (741 ) + TX, Steinemema bibionis (742) + TX, Steinemema carpocapsae (742) + TX, Steinemema feltiae (742) + TX, Steinemema glaseri (742) + TX, Steinemema riobrave (742) + TX, Steinemema riobravis (742) + TX, Steinemema scapterisci (742) + TX, Steinemema spp. (742) + TX, Trichogramma spp. (826) + TX, Typhlodromus occidentalis (844) and Verticillium lecanii (848) + TX,
a soil sterilant selected from the group of substances consisting of iodomethane (lUPAC name) (542) and methyl bromide (537) + TX,
a chemosterilant selected from the group of substances consisting of apholate [CCN] + TX, bisazir [CCN] + TX, busulfan [CCN] + TX, diflubenzuron (250) + TX, dimatif [CCN] + TX, hemel [CCN] + TX, hempa [CCN] + TX, metepa [CCN] + TX, methiotepa [CCN] + TX, methyl apholate [CCN] + TX, morzid [CCN] + TX, penfluron [CCN] + TX, tepa [CCN] + TX, thiohempa [CCN] + TX, thiotepa [CCN] + TX, tretamine [CCN] and uredepa [CCN] + TX,
an insect pheromone selected from the group of substances consisting of (E)-dec-5-en-1-yl acetate with (E)-dec-5-en-1-ol (lUPAC name) (222) + TX, (E)-tridec-4-en-1-yl acetate (lUPAC name) (829) + TX, (E)-6-methylhept-2-en-4-ol (lUPAC name) (541 ) + TX, (E,Z)-tetradeca-4, 10-dien-1-yl acetate (lUPAC name) (779) + TX, (Z)-dodec-7-en-1-yl acetate (lUPAC name) (285) + TX, (Z)-hexadec-l 1- enal (lUPAC name) (436) + TX, (Z)-hexadec-l 1-en-1-yl acetate (lUPAC name) (437) + TX, (Z)- hexadec-13-en-1 1-yn-1-yl acetate (lUPAC name) (438) + TX, (Z)-icos-13-en-10-one (lUPAC name) (448) + TX, (Z)-tetradec-7-en-1-al (lUPAC name) (782) + TX, (Z)-tetradec-9-en-1-ol (lUPAC name) (783) + TX, (Z)-tetradec-9-en-1-yl acetate (lUPAC name) (784) + TX,(7E,9Z)-dodeca-7,9-dien-1-yl acetate (lUPAC name) (283) + TX, (9Z, 1 1 E)-tetradeca-9, 1 1-dien-1-yl acetate (lUPAC name) (780) + TX, (9Z, 12E)-tetradeca-9, 12-dien-1-yl acetate (lUPAC name) (781 ) + TX, 14-methyloctadec-1-ene (lUPAC name) (545) + TX, 4-methylnonan-5-ol with 4-methylnonan-5-one (lUPAC name) (544) + TX, alpha-multistriatin [CCN] + TX, brevicomin [CCN] + TX, codlelure [CCN] + TX, codlemone (167) + TX, cuelure (179) + TX, disparlure (277) + TX, dodec-8-en-1-yl acetate (lUPAC name) (286) + TX, dodec-9-en-1-yl acetate (lUPAC name) (287) + TX, dodeca-8 + TX, 10-dien-1-yl acetate (lUPAC name) (284) + TX, dominicalure [CCN] + TX, ethyl 4-methyloctanoate (lUPAC name) (317) + TX, eugenol [CCN] + TX, frontalin [CCN] + TX, gossyplure (420) + TX, grandlure (421 ) + TX, grandlure I (421 ) + TX, grandlure II (421 ) + TX, grandlure III (421 ) + TX, grandlure IV (421 ) + TX, hexalure
[CCN] + TX, ipsdienol [CCN] + TX, ipsenol [CCN] + TX, japonilure (481 ) + TX, lineatin [CCN] + TX, litlure [CCN] + TX, looplure [CCN] + TX, medlure [CCN] + TX, megatomoic acid [CCN] + TX, methyl eugenol (540) + TX, muscalure (563) + TX, octadeca-2, 13-dien-1-yl acetate (lUPAC name) (588) + TX,octadeca-3, 13-dien-1-yl acetate (lUPAC name) (589) + TX, orfralure [CCN] + TX, oryctalure (317) + TX, ostramone [CCN] + TX, siglure [CCN] + TX, sordidin (736) + TX,sulcatol [CCN] + TX, tetradec- 1 1-en-1-yl acetate (lUPAC name) (785) + TX, trimedlure (839) + TX, trimedlure A (839) + TX, trimedlure Bi (839) + TX, trimedlure B2 (839) + TX, trimedlure C (839) and trunc-call [CCN] + TX,
an insect repellent selected from the group of substances consisting of 2-(octylthio)ethanol (lUPAC name) (591 ) + TX, butopyronoxyl (933) + TX, butoxy(polypropylene glycol) (936) + TX, dibutyl adipate (lUPAC name) (1046) + TX, dibutyl phthalate (1047) + TX, dibutyl succinate (lUPAC name) (1048) + TX, diethyltoluamide [CCN] + TX, dimethyl carbate [CCN] + TX, dimethyl phthalate [CCN] + TX, ethyl hexanediol (1 137) + TX, hexamide [CCN] + TX, methoquin-butyl (1276) + TX, methylneodecanamide [CCN] + TX, oxamate [CCN] and picaridin [CCN] + TX,
an insecticide selected from the group of substances consisting of 1-dichloro-1-nitroethane
(lUPAC/Chemical Abstracts name) (1058) + TX, 1 , 1-dichloro-2,2-bis(4-ethylphenyl)ethane (lUPAC name) (1056), + TX,1 ,2-dichloropropane (lUPAC/Chemical Abstracts name) (1062) + TX, 1 ,2- dichloropropane with 1 ,3-dichloropropene (lUPAC name) (1063) + TX, 1-bromo-2-chloroethane (lUPAC/Chemical Abstracts name) (916) + TX,2,2,2-trichloro-1-(3,4-dichlorophenyl)ethyl acetate (lUPAC name) (1451 ) + TX, 2,2-dichlorovinyl 2-ethylsulfinylethyl methyl phosphate (lUPAC name) (1066) + TX, 2-(1 ,3-dithiolan-2-yl)phenyl dimethylcarbamate (lUPAC/ Chemical Abstracts name) (1 109) + TX, 2-(2-butoxyethoxy)ethyl thiocyanate (lUPAC/Chemical Abstracts name) (935) + TX,2- (4,5-dimethyl-1 ,3-dioxolan-2-yl)phenyl methylcarbamate (lUPAC/ Chemical Abstracts name) (1084) + TX,2-(4-chloro-3,5-xylyloxy)ethanol (lUPAC name) (986) + TX, 2-chlorovinyl diethyl phosphate (lUPAC name) (984) + TX, 2-imidazolidone (lUPAC name) (1225) + TX,2-isovalerylindan-1 ,3-dione (lUPAC name) (1246) + TX, 2-methyl(prop-2-ynyl)aminophenyl methylcarbamate (lUPAC name) (1284) + TX, 2-thiocyanatoethyl laurate (lUPAC name) (1433) + TX, 3-bromo-1-chloroprop-1-ene (lUPAC name) (917) + TX, 3-methyl-1-phenylpyrazol-5-yl dimethylcarbamate (lUPAC name) (1283) + TX, 4- methyl(prop-2-ynyl)amino-3,5-xylyl methylcarbamate (lUPAC name) (1285) + TX, 5,5-dimethyl-3- oxocyclohex-1-enyl dimethylcarbamate (lUPAC name) (1085) + TX, abamectin (1 ) + TX, acephate (2) + TX, acetamiprid (4) + TX, acethion [CCN] + TX, acetoprole [CCN] + TX, acrinathrin (9) + TX, acrylonitrile (lUPAC name) (861 ) + TX, alanycarb (15) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, aldrin (864) + TX, allethrin (17) + TX, allosamidin [CCN] + TX, allyxycarb (866) + TX, alpha- cypermethrin (202) + TX, alpha-ecdysone [CCN] + TX, aluminium phosphide (640) + TX, amidithion (870) + TX, amidothioate (872) + TX, aminocarb (873) + TX, amiton (875) + TX, amiton hydrogen oxalate (875) + TX, amitraz (24) + TX, anabasine (877) + TX, athidathion (883) + TX, AVI 382 (compound code) + TX, AZ 60541 (compound code) + TX, azadirachtin (41 ) + TX, azamethiphos (42) + TX, azinphos-ethyl (44) + TX, azinphos-methyl (45) + TX, azothoate (889) + TX, Bacillus thuringiensis delta endotoxins (52) + TX, barium hexafluorosilicate [CCN] + TX, barium polysulfide (lUPAC/Chemical Abstracts name) (892) + TX, barthrin [CCN] + TX, Bayer 22/190 (development code) (893) + TX, Bayer 22408 (development code) (894) + TX, bendiocarb (58) + TX, benfuracarb (60) + TX, bensultap (66) + TX, beta-cyfluthrin (194) + TX, beta-cypermethrin (203) + TX, bifenthrin (76) + TX, bioallethrin (78) + TX, bioallethrin S-cyclopentenyl isomer (79) + TX, bioethanomethrin [CCN] + TX, biopermethrin (908) + TX, bioresmethrin (80) + TX, bis(2-chloroethyl) ether (lUPAC name) (909) + TX, bistrifluron (83) + TX, borax (86) + TX, brofenvalerate + TX, bromfenvinfos (914) + TX, bromocyclen (918) + TX, bromo-DDT [CCN] + TX, bromophos (920) + TX, bromophos-ethyl (921 )
+ TX, bufencarb (924) + TX, buprofezin (99) + TX, butacarb (926) + TX, butathiofos (927) + TX, butocarboxim (103) + TX, butonate (932) + TX, butoxycarboxim (104) + TX, butylpyridaben + TX, cadusafos (109) + TX, calcium arsenate [CCN] + TX, calcium cyanide (444) + TX, calcium polysulfide (lUPAC name) (1 1 1 ) + TX, camphechlor (941 ) + TX, carbanolate (943) + TX, carbaryl (1 15) + TX, carbofuran (1 18) + TX, carbon disulfide (lUPAC/Chemical Abstracts name) (945) + TX, carbon tetrachloride (lUPAC name) (946) + TX, carbophenothion (947) + TX, carbosulfan (1 19) + TX, cartap (123) + TX, cartap hydrochloride (123) + TX, cevadine (725) + TX, chlorbicyclen (960) + TX, chlordane (128) + TX, chlordecone (963) + TX, chlordimeform (964) + TX, chlordimeform
hydrochloride (964) + TX, chlorethoxyfos (129) + TX, chlorfenapyr (130) + TX, chlorfenvinphos (131 ) + TX, chlorfluazuron (132) + TX, chlormephos (136) + TX, chloroform [CCN] + TX, chloropicrin (141 ) + TX, chlorphoxim (989) + TX, chlorprazophos (990) + TX, chlorpyrifos (145) + TX, chlorpyrifos-methyl (146) + TX, chlorthiophos (994) + TX, chromafenozide (150) + TX, cinerin I (696) + TX, cinerin II (696) + TX, cinerins (696) + TX, cis-resmethrin + TX, cismethrin (80) + TX, clocythrin + TX, cloethocarb (999) + TX, closantel [CCN] + TX, clothianidin (165) + TX, copper acetoarsenite [CCN] + TX, copper arsenate [CCN] + TX, copper oleate [CCN] + TX, coumaphos (174) + TX, coumithoate (1006) + TX, crotamiton [CCN] + TX, crotoxyphos (1010) + TX, crufomate (101 1 ) + TX, cryolite (177) + TX, CS 708 (development code) (1012) + TX, cyanofenphos (1019) + TX, cyanophos (184) + TX, cyanthoate (1020) + TX, cyclethrin [CCN] + TX, cycloprothrin (188) + TX, cyfluthrin (193) + TX, cyhalothrin (196) + TX, cypermethrin (201 ) + TX, cyphenothrin (206) + TX, cyromazine (209) + TX, cythioate [CCN] + TX, c/-limonene [CCN] + TX, c/-tetramethrin (788) + TX, DAEP (1031 ) + TX, dazomet (216) + TX, DDT
(219) + TX, decarbofuran (1034) + TX, deltamethrin (223) + TX, demephion (1037) + TX, demephion- O (1037) + TX, demephion-S (1037) + TX, demeton (1038) + TX, demeton-methyl (224) + TX, demeton-0 (1038) + TX, demeton-O-methyl (224) + TX, demeton-S (1038) + TX, demeton-S-methyl (224) + TX, demeton-S-methylsulphon (1039) + TX, diafenthiuron (226) + TX, dialifos (1042) + TX, diamidafos (1044) + TX, diazinon (227) + TX, dicapthon (1050) + TX, dichlofenthion (1051 ) + TX, dichlorvos (236) + TX, dicliphos + TX, dicresyl [CCN] + TX, dicrotophos (243) + TX, dicyclanil (244) + TX, dieldrin (1070) + TX, diethyl 5-methylpyrazol-3-yl phosphate (lUPAC name) (1076) + TX, diflubenzuron (250) + TX, dilor [CCN] + TX, dimefluthrin [CCN] + TX, dimefox (1081 ) + TX, dimetan (1085) + TX, dimethoate (262) + TX, dimethrin (1083) + TX, dimethylvinphos (265) + TX, dimetilan (1086) + TX, dinex (1089) + TX, dinex-diclexine (1089) + TX, dinoprop (1093) + TX, dinosam (1094) + TX, dinoseb (1095) + TX, dinotefuran (271 ) + TX, diofenolan (1099) + TX, dioxabenzofos (1 100) + TX, dioxacarb (1 101 ) + TX, dioxathion (1 102) + TX, disulfoton (278) + TX, dithicrofos (1 108) + TX, DNOC (282) + TX, doramectin [CCN] + TX, DSP (1 1 15) + TX, ecdysterone [CCN] + TX, El 1642
(development code) (1 1 18) + TX, emamectin (291 ) + TX, emamectin benzoate (291 ) + TX, EMPC (1 120) + TX, empenthrin (292) + TX, endosulfan (294) + TX, endothion (1 121 ) + TX, endrin (1 122) + TX, EPBP (1 123) + TX, EPN (297) + TX, epofenonane (1 124) + TX, eprinomectin [CCN] + TX, esfenvalerate (302) + TX, etaphos [CCN] + TX, ethiofencarb (308) + TX, ethion (309) + TX, ethiprole (310) + TX, ethoate-m ethyl (1 134) + TX, ethoprophos (312) + TX, ethyl formate (lUPAC name) [CCN]
+ TX, ethyl-DDD (1056) + TX, ethylene dibromide (316) + TX, ethylene dichloride (chemical name) (1 136) + TX, ethylene oxide [CCN] + TX, etofenprox (319) + TX, etrimfos (1 142) + TX, EXD (1 143) + TX, famphur (323) + TX, fenamiphos (326) + TX, fenazaflor (1 147) + TX, fenchlorphos (1 148) + TX, fenethacarb (1 149) + TX, fenfluthrin (1 150) + TX, fenitrothion (335) + TX, fenobucarb (336) + TX, fenoxacrim (1 153) + TX, fenoxycarb (340) + TX, fenpirithrin (1 155) + TX, fenpropathrin (342) + TX, fenpyrad + TX, fensulfothion (1 158) + TX, fenthion (346) + TX, fenthion-ethyl [CCN] + TX, fenvalerate (349) + TX, fipronil (354) + TX, flonicamid (358) + TX, flubendiamide (CAS. Reg. No.: 272451-65-7) + TX, flucofuron (1 168) + TX, flucycloxuron (366) + TX, flucythrinate (367) + TX, fluenetil (1 169) + TX, flufenerim [CCN] + TX, flufenoxuron (370) + TX, flufenprox (1 171 ) + TX, flumethrin (372) + TX, fluvalinate (1 184) + TX, FMC 1 137 (development code) (1 185) + TX, fonofos (1 191 ) + TX, formetanate (405) + TX, formetanate hydrochloride (405) + TX, formothion (1 192) + TX, formparanate (1 193) + TX, fosmethilan (1 194) + TX, fospirate (1 195) + TX, fosthiazate (408) + TX, fosthietan (1 196) + TX, furathiocarb (412) + TX, furethrin (1200) + TX, gamma-cyhalothrin (197) + TX, gamma-HCH (430) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, GY-81 (development code) (423) + TX, halfenprox (424) + TX, halofenozide (425) + TX, HCH (430) + TX, HEOD (1070) + TX, heptachlor
(121 1 ) + TX, heptenophos (432) + TX, heterophos [CCN] + TX, hexaflumuron (439) + TX, HHDN (864) + TX, hydramethylnon (443) + TX, hydrogen cyanide (444) + TX, hydroprene (445) + TX, hyquincarb (1223) + TX, imidacloprid (458) + TX, imiprothrin (460) + TX, indoxacarb (465) + TX, iodomethane (lUPAC name) (542) + TX, IPSP (1229) + TX, isazofos (1231 ) + TX, isobenzan (1232) + TX, isocarbophos (473) + TX, isodrin (1235) + TX, isofenphos (1236) + TX, isolane (1237) + TX, isoprocarb (472) + TX, isopropyl 0-(methoxyaminothiophosphoryl)salicylate (lUPAC name) (473) + TX, isoprothiolane (474) + TX, isothioate (1244) + TX, isoxathion (480) + TX, ivermectin [CCN] + TX, jasmolin I (696) + TX, jasmolin II (696) + TX, jodfenphos (1248) + TX, juvenile hormone I [CCN] + TX, juvenile hormone II [CCN] + TX, juvenile hormone III [CCN] + TX, kelevan (1249) + TX, kinoprene (484) + TX, lambda-cyhalothrin (198) + TX, lead arsenate [CCN] + TX, lepimectin (CCN) + TX, leptophos (1250) + TX, lindane (430) + TX, lirimfos (1251 ) + TX, lufenuron (490) + TX, lythidathion (1253) + TX, m-cumenyl methylcarbamate (lUPAC name) (1014) + TX, magnesium phosphide (lUPAC name) (640) + TX, malathion (492) + TX, malonoben (1254) + TX, mazidox (1255) + TX, mecarbam (502) + TX, mecarphon (1258) + TX, menazon (1260) + TX, mephosfolan (1261 ) + TX, mercurous chloride (513) + TX, mesulfenfos (1263) + TX, metaflumizone (CCN) + TX, metam (519) + TX, metam- potassium (519) + TX, metam-sodium (519) + TX, methacrifos (1266) + TX, methamidophos (527) + TX, methanesulfonyl fluoride (lUPAC/Chemical Abstracts name) (1268) + TX, methidathion (529) + TX, methiocarb (530) + TX, methocrotophos (1273) + TX, methomyl (531 ) + TX, methoprene (532) + TX, methoquin-butyl (1276) + TX, methothrin (533) + TX, methoxychlor (534) + TX, methoxyfenozide (535) + TX, methyl bromide (537) + TX, methyl isothiocyanate (543) + TX, methylchloroform [CCN] + TX, methylene chloride [CCN] + TX, metofluthrin [CCN] + TX, metolcarb (550) + TX, metoxadiazone (1288) + TX, mevinphos (556) + TX, mexacarbate (1290) + TX, milbemectin (557) + TX, milbemycin oxime [CCN] + TX, mipafox (1293) + TX, mirex (1294) + TX, monocrotophos (561 ) + TX, morphothion
(1300) + TX, moxidectin [CCN] + TX, naftalofos [CCN] + TX, naled (567) + TX, naphthalene (lUPAC/Chemical Abstracts name) (1303) + TX, NC-170 (development code) (1306) + TX, NC-184 (compound code) + TX, nicotine (578) + TX, nicotine sulfate (578) + TX, nifluridide (1309) + TX, nitenpyram (579) + TX, nithiazine (131 1 ) + TX, nitrilacarb (1313) + TX, nitrilacarb 1 : 1 zinc chloride complex (1313) + TX, NNI-0101 (compound code) + TX, NNI-0250 (compound code) + TX, nornicotine (traditional name) (1319) + TX, novaluron (585) + TX, noviflumuron (586) + TX, 0-5- dichloro-4-iodophenyl O-ethyl ethylphosphonothioate (lUPAC name) (1057) + TX, 0,0-diethyl 0-4- methyl-2-oxo-2A -chromen-7-yl phosphorothioate (lUPAC name) (1074) + TX, 0,0-diethyl O-6-methyl- 2-propylpyrimidin-4-yl phosphorothioate (lUPAC name) (1075) + TX, 0,0, 0',Ο'-tetrapropyl dithiopyrophosphate (lUPAC name) (1424) + TX, oleic acid (lUPAC name) (593) + TX, omethoate (594) + TX, oxamyl (602) + TX, oxydemeton-methyl (609) + TX, oxydeprofos (1324) + TX, oxydisulfoton (1325) + TX, pp'-DDT (219) + TX, para-dichlorobenzene [CCN] + TX, parathion (615) + TX, parath ion-methyl (616) + TX, penfluron [CCN] + TX, pentachlorophenol (623) + TX,
pentachlorophenyl laurate (lUPAC name) (623) + TX, permethrin (626) + TX, petroleum oils (628) + TX, PH 60-38 (development code) (1328) + TX, phenkapton (1330) + TX, phenothrin (630) + TX, phenthoate (631 ) + TX, phorate (636) + TX, phosalone (637) + TX, phosfolan (1338) + TX, phosmet (638) + TX, phosnichlor (1339) + TX, phosphamidon (639) + TX, phosphine (lUPAC name) (640) + TX, phoxim (642) + TX, phoxim-methyl (1340) + TX, pirimetaphos (1344) + TX, pirimicarb (651 ) + TX, pirimiphos-ethyl (1345) + TX, pirimiphos-methyl (652) + TX, polychlorodicyclopentadiene isomers (lUPAC name) (1346) + TX, polychloroterpenes (traditional name) (1347) + TX, potassium arsenite [CCN] + TX, potassium thiocyanate [CCN] + TX, prallethrin (655) + TX, precocene I [CCN] + TX, precocene II [CCN] + TX, precocene III [CCN] + TX, primidophos (1349) + TX, profenofos (662) + TX, profluthrin [CCN] + TX, promacyl (1354) + TX, promecarb (1355) + TX, propaphos (1356) + TX, propetamphos (673) + TX, propoxur (678) + TX, prothidathion (1360) + TX, prothiofos (686) + TX, prothoate (1362) + TX, protrifenbute [CCN] + TX, pymetrozine (688) + TX, pyraclofos (689) + TX, pyrazophos (693) + TX, pyresmethrin (1367) + TX, pyrethrin I (696) + TX, pyrethrin II (696) + TX, pyrethrins (696) + TX, pyridaben (699) + TX, pyridalyl (700) + TX, pyridaphenthion (701 ) + TX, pyrimidifen (706) + TX, pyrimitate (1370) + TX, pyriproxyfen (708) + TX, quassia [CCN] + TX, quinalphos (71 1 ) + TX, quinalphos-methyl (1376) + TX, quinothion (1380) + TX, quintiofos (1381 ) + TX, R-1492 (development code) (1382) + TX, rafoxanide [CCN] + TX, resmethrin (719) + TX, rotenone (722) + TX, RU 15525 (development code) (723) + TX, RU 25475 (development code) (1386) + TX, ryania (1387) + TX, ryanodine (traditional name) (1387) + TX, sabadilla (725) + TX, schradan (1389) + TX, sebufos + TX, selamectin [CCN] + TX, SI-0009 (compound code) + TX, Sl- 0205 (compound code) + TX, SI-0404 (compound code) + TX, SI-0405 (compound code) + TX, silafluofen (728) + TX, SN 72129 (development code) (1397) + TX, sodium arsenite [CCN] + TX, sodium cyanide (444) + TX, sodium fluoride (lUPAC/Chemical Abstracts name) (1399) + TX, sodium hexafluorosilicate (1400) + TX, sodium pentachlorophenoxide (623) + TX, sodium selenate (lUPAC name) (1401 ) + TX, sodium thiocyanate [CCN] + TX, sophamide (1402) + TX, spinosad (737) + TX,
spiromesifen (739) + TX, spirotetrmat (CCN) + TX, sulcofuron (746) + TX, sulcofuron-sodium (746) + TX, sulfluramid (750) + TX, sulfotep (753) + TX, sulfuryl fluoride (756) + TX, sulprofos (1408) + TX, tar oils (758) + TX, tau-fluvalinate (398) + TX, tazimcarb (1412) + TX, TDE (1414) + TX, tebufenozide (762) + TX, tebufenpyrad (763) + TX, tebupirimfos (764) + TX, teflubenzuron (768) + TX, tefluthrin (769) + TX, temephos (770) + TX, TEPP (1417) + TX, terallethrin (1418) + TX, terbam + TX, terbufos (773) + TX, tetrachloroethane [CCN] + TX, tetrachlorvinphos (777) + TX, tetramethrin (787) + TX, theta-cypermethrin (204) + TX, thiacloprid (791 ) + TX, thiafenox + TX, thiamethoxam (792) + TX, thicrofos (1428) + TX, thiocarboxime (1431 ) + TX, thiocyclam (798) + TX, thiocyclam hydrogen oxalate (798) + TX, thiodicarb (799) + TX, thiofanox (800) + TX, thiometon (801 ) + TX, thionazin (1434) + TX, thiosultap (803) + TX, thiosultap-sodium (803) + TX, thuringiensin [CCN] + TX, tolfenpyrad (809) +
TX, tralomethrin (812) + TX, transfluthrin (813) + TX, transpermethrin (1440) + TX, triamiphos (1441 ) + TX, triazamate (818) + TX, triazophos (820) + TX, triazuron + TX, trichlorfon (824) + TX,
trichlormetaphos-3 [CCN] + TX, trichloronat (1452) + TX, trifenofos (1455) + TX, triflumuron (835) + TX, trimethacarb (840) + TX, triprene (1459) + TX, vamidothion (847) + TX, vaniliprole [CCN] + TX, veratridine (725) + TX, veratrine (725) + TX, XMC (853) + TX, xylylcarb (854) + TX, YI-5302
(compound code) + TX, zeta-cypermethrin (205) + TX, zetamethrin + TX, zinc phosphide (640) + TX, zolaprofos (1469) and ZXI 8901 (development code) (858) + TX,cyantraniliprole [736994-63-19 + TX, chlorantraniliprole [500008-45-7] + TX, cyenopyrafen [560121-52-0] + TX, cyflumetofen [400882-07-7] + TX, pyrifluquinazon [337458-27-2] + TX, spinetoram [187166-40-1 + 187166-15-0] + TX, spirotetramat [203313-25-1] + TX, sulfoxaflor [946578-00-3] + TX, flufiprole [704886-18-0] + TX, meperfluthrin [915288-13-0] + TX, tetramethylfluthrin [84937-88-2] + TX, triflumezopyrim (disclosed in WO 2012/0921 15) + TX,
a molluscicide selected from the group of substances consisting of bis(tributyltin) oxide (lUPAC name) (913) + TX, bromoacetamide [CCN] + TX, calcium arsenate [CCN] + TX, cloethocarb (999) + TX, copper acetoarsenite [CCN] + TX, copper sulfate (172) + TX, fentin (347) + TX, ferric phosphate
(lUPAC name) (352) + TX, metaldehyde (518) + TX, methiocarb (530) + TX, niclosamide (576) + TX, niclosamide-olamine (576) + TX, pentachlorophenol (623) + TX, sodium pentachlorophenoxide (623) + TX, tazimcarb (1412) + TX, thiodicarb (799) + TX, tributyltin oxide (913) + TX, trifenmorph (1454) + TX, trimethacarb (840) + TX, triphenyltin acetate (lUPAC name) (347) and triphenyltin hydroxide (lUPAC name) (347) + TX, pyriprole [394730-71-3] + TX,
a nematicide selected from the group of substances consisting of AKD-3088 (compound code) + TX, 1 ,2-dibromo-3-chloropropane (lUPAC/Chemical Abstracts name) (1045) + TX, 1 ,2-dichloropropane (lUPAC/ Chemical Abstracts name) (1062) + TX, 1 ,2-dichloropropane with 1 ,3-dichloropropene (lUPAC name) (1063) + TX, 1 ,3-dichloropropene (233) + TX, 3,4-dichlorotetrahydrothiophene 1 ,1- dioxide (lUPAC/Chemical Abstracts name) (1065) + TX,3-(4-chlorophenyl)-5-methylrhodanine (lUPAC name) (980) + TX, 5-methyl-6-thioxo-1 ,3,5-thiadiazinan-3-ylacetic acid (lUPAC name) (1286) + TX, 6- isopentenylaminopurine (210) + TX, abamectin (1 ) + TX, acetoprole [CCN] + TX, alanycarb (15) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, AZ 60541 (compound code) + TX, benclothiaz [CCN] + TX,
benomyl (62) + TX, butylpyridaben + TX, cadusafos (109) + TX, carbofuran (1 18) + TX, carbon disulfide (945) + TX, carbosulfan (1 19) + TX, chloropicrin (141 ) + TX, chlorpyrifos (145) + TX, cloethocarb (999) + TX, cytokinins (210) + TX, dazomet (216) + TX, DBCP (1045) + TX, DCIP (218) + TX, diamidafos (1044) + TX, dichlofenthion (1051 ) + TX, dicliphos + TX, dimethoate (262) + TX, doramectin [CCN] + TX, emamectin (291 ) + TX, emamectin benzoate (291 ) + TX, eprinomectin
[CCN] + TX, ethoprophos (312) + TX, ethylene dibromide (316) + TX, fenamiphos (326) + TX, fenpyrad + TX, fensulfothion (1 158) + TX, fosthiazate (408) + TX, fosthietan (1 196) + TX, furfural [CCN] + TX, GY-81 (development code) (423) + TX, heterophos [CCN] + TX, iodomethane (lUPAC name) (542) + TX, isamidofos (1230) + TX, isazofos (1231 ) + TX, ivermectin [CCN] + TX, kinetin (210) + TX, mecarphon (1258) + TX, metam (519) + TX, metam-potassium (519) + TX, metam- sodium (519) + TX, methyl bromide (537) + TX, methyl isothiocyanate (543) + TX, milbemycin oxime [CCN] + TX, moxidectin [CCN] + TX, Myrothecium verrucaria composition (565) + TX, NC-184 (compound code) + TX, oxamyl (602) + TX, phorate (636) + TX, phosphamidon (639) + TX, phosphocarb [CCN] + TX, sebufos + TX, selamectin [CCN] + TX, spinosad (737) + TX, terbam + TX, terbufos (773) + TX, tetrachlorothiophene (lUPAC/ Chemical Abstracts name) (1422) + TX, thiafenox + TX, thionazin (1434) + TX, triazophos (820) + TX, triazuron + TX, xylenols [CCN] + TX, YI-5302 (compound code) and zeatin (210) + TX,fluensulfone [318290-98-1] + TX,
a nitrification inhibitor selected from the group of substances consisting of potassium ethylxanthate [CCN] and nitrapyrin (580) + TX,
a plant activator selected from the group of substances consisting of acibenzolar (6) + TX, acibenzolar-S-methyl (6) + TX, probenazole (658) and Reynoutria sachalinensis extract (720) + TX, a rodenticide selected from the group of substances consisting of 2-isovalerylindan-1 ,3-dione (lUPAC name) (1246) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (lUPAC name) (748) + TX, alpha- chlorohydrin [CCN] + TX, aluminium phosphide (640) + TX, antu (880) + TX, arsenous oxide (882) + TX, barium carbonate (891 ) + TX, bisthiosemi (912) + TX, brodifacoum (89) + TX, bromadiolone (91 ) + TX, bromethalin (92) + TX, calcium cyanide (444) + TX, chloralose (127) + TX, chlorophacinone (140) + TX, cholecalciferol (850) + TX, coumachlor (1004) + TX, coumafuryl (1005) + TX, coumatetralyl (175) + TX, crimidine (1009) + TX, difenacoum (246) + TX, difethialone (249) + TX, diphacinone (273) + TX, ergocalciferol (301 ) + TX, flocoumafen (357) + TX, fluoroacetamide (379) + TX, flupropadine (1 183) + TX, flupropadine hydrochloride (1 183) + TX, gamma-HCH (430) + TX, HCH (430) + TX, hydrogen cyanide (444) + TX, iodomethane (lUPAC name) (542) + TX, lindane (430) + TX, magnesium phosphide (lUPAC name) (640) + TX, methyl bromide (537) + TX, norbormide (1318) + TX, phosacetim (1336) + TX, phosphine (lUPAC name) (640) + TX, phosphorus [CCN] + TX, pindone (1341 ) + TX, potassium arsenite [CCN] + TX, pyrinuron (1371 ) + TX, scilliroside (1390) + TX, sodium arsenite [CCN] + TX, sodium cyanide (444) + TX, sodium fluoroacetate (735) + TX, strychnine (745) + TX, thallium sulfate [CCN] + TX, warfarin (851 ) and zinc phosphide (640) + TX,
a synergist selected from the group of substances consisting of 2-(2-butoxyethoxy)ethyl piperonylate (lUPAC name) (934) + TX, 5-(1 ,3-benzodioxol-5-yl)-3-hexylcyclohex-2-enone (lUPAC name) (903) +
TX, farnesol with nerolidol (324) + TX, MB-599 (development code) (498) + TX, MGK 264
(development code) (296) + TX, piperonyl butoxide (649) + TX, piprotal (1343) + TX, propyl isomer (1358) + TX, S421 (development code) (724) + TX, sesamex (1393) + TX, sesasmolin (1394) and sulfoxide (1406) + TX,
an animal repellent selected from the group of substances consisting of anthraquinone (32) + TX, chloralose (127) + TX, copper naphthenate [CCN] + TX, copper oxychloride (171 ) + TX, diazinon (227) + TX, dicyclopentadiene (chemical name) (1069) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, methiocarb (530) + TX, pyridin-4-amine (lUPAC name) (23) + TX, thiram (804) + TX, trimethacarb (840) + TX, zinc naphthenate [CCN] and ziram (856) + TX,
a virucide selected from the group of substances consisting of imanin [CCN] and ribavirin [CCN] + TX,
a wound protectant selected from the group of substances consisting of mercuric oxide (512) + TX,octhilinone (590) and thiophanate-methyl (802) + TX, and biologically active compounds selected from the group consisting of azaconazole (60207-31-0] + TX, bitertanol [70585-36-3] + TX, bromuconazole [1 16255-48-2] + TX, cyproconazole [94361-06-5] + TX, difenoconazole [1 19446-68-3] + TX, diniconazole [83657-24-3] + TX, epoxiconazole [106325-08- 0] + TX, fenbuconazole [1 14369-43-6] + TX, fluquinconazole [136426-54-5] + TX, flusilazole [85509- 19-9] + TX, flutriafol [76674-21-0] + TX, hexaconazole [79983-71-4] + TX, imazalil [35554-44-0] + TX, imibenconazole [86598-92-7] + TX, ipconazole [125225-28-7] + TX, metconazole [1251 16-23-6] + TX, myclobutanil [88671-89-0] + TX, pefurazoate [101903-30-4] + TX, penconazole [66246-88-6] + TX, prothioconazole [178928-70-6] + TX, pyrifenox [88283-41-4] + TX, prochloraz [67747-09-5] + TX, propiconazole [60207-90-1] + TX, simeconazole [149508-90-7] + TX, tebuconazole [107534-96-3] + TX, tetraconazole [1 12281-77-3] + TX, triadimefon [43121-43-3] + TX, triadimenol [55219-65-3] + TX, triflumizole [99387-89-0] + TX, triticonazole [131983-72-7] + TX, ancymidol [12771-68-5] + TX, fenarimol [60168-88-9] + TX, nuarimol [63284-71-9] + TX, bupirimate [41483-43-6] + TX, dimethirimol [5221-53-4] + TX, ethirimol [23947-60-6] + TX, dodemorph [1593-77-7] + TX, fenpropidine [67306-00- 7] + TX, fenpropimorph [67564-91-4] + TX, spiroxamine [1 18134-30-8] + TX, tridemorph [81412-43-3] + TX, cyprodinil [121552-61-2] + TX, mepanipyrim [1 10235-47-7] + TX, pyrimethanil [531 12-28-0] + TX, fenpiclonil [74738-17-3] + TX, fludioxonil [131341-86-1] + TX, benalaxyl [71626-1 1-4] + TX, furalaxyl [57646-30-7] + TX, metalaxyl [57837-19-1] + TX, R-metalaxyl [70630-17-0] + TX, ofurace [58810-48-3] + TX, oxadixyl [77732-09-3] + TX, benomyl [17804-35-2] + TX, carbendazim [10605-21- 7] + TX, debacarb [62732-91-6] + TX, fuberidazole [3878-19-1] + TX, thiabendazole [148-79-8] + TX, chlozolinate [84332-86-5] + TX, dichlozoline [24201-58-9] + TX, iprodione [36734-19-7] + TX, myclozoline [54864-61-8] + TX, procymidone [32809-16-8] + TX, vinclozoline [50471-44-8] + TX, boscalid [188425-85-6] + TX, carboxin [5234-68-4] + TX, fenfuram [24691-80-3] + TX, flutolanil
[66332-96-5] + TX, mepronil [55814-41-0] + TX, oxycarboxin [5259-88-1] + TX, penthiopyrad [183675- 82-3] + TX, thifluzamide [130000-40-7] + TX, guazatine [108173-90-6] + TX, dodine [2439-10-3] [1 12-
65-2] (free base) + TX, iminoctadine [13516-27-3] + TX, azoxystrobin [131860-33-8] + TX, dimoxystrobin [149961-52-4] + TX, enestroburin {Proc. BCPC, Int. Congr., Glasgow, 2003, 1 , 93} + TX, fluoxastrobin [361377-29-9] + TX, kresoxim-methyl [143390-89-0] + TX, metominostrobin [133408- 50-1] + TX, trifloxystrobin [141517-21-7] + TX, orysastrobin [248593-16-0] + TX, picoxystrobin
[1 17428-22-5] + TX, pyraclostrobin [175013-18-0] + TX, ferbam [14484-64-1] + TX, mancozeb [8018- 01-7] + TX, maneb [12427-38-2] + TX, metiram [9006-42-2] + TX, propineb [12071-83-9] + TX, thiram [137-26-8] + TX, zineb [12122-67-7] + TX, ziram [137-30-4] + TX, captafol [2425-06-1] + TX, captan [133-06-2] + TX, dichlofluanid [1085-98-9] + TX, fluoroimide [41205-21-4] + TX, folpet [133-07-3 ] + TX, tolylfluanid [731-27-1] + TX, bordeaux mixture [801 1-63-0] + TX, copperhydroxid [20427-59-2] + TX, copperoxychlorid [1332-40-7] + TX, coppersulfat [7758-98-7] + TX, copperoxid [1317-39-1] + TX, mancopper [53988-93-5] + TX, oxine-copper [10380-28-6] + TX, dinocap [131-72-6] + TX, nitrothal- isopropyl [10552-74-6] + TX, edifenphos [17109-49-8] + TX, iprobenphos [26087-47-8] + TX, isoprothiolane [50512-35-1] + TX, phosdiphen [36519-00-3] + TX, pyrazophos [13457-18-6] + TX, tolclofos-methyl [57018-04-9] + TX, acibenzolar-S-methyl [135158-54-2] + TX, anilazine [101-05-3] + TX, benthiavalicarb [413615-35-7] + TX, blasticidin-S [2079-00-7] + TX, chinomethionat [2439-01-2] + TX, chloroneb [2675-77-6] + TX, chlorothalonil [1897-45-6] + TX, cyflufenamid [180409-60-3] + TX,cymoxanil [57966-95-7] + TX, dichlone [117-80-6] + TX, diclocymet [139920-32-4] + TX, diclomezine [62865-36-5] + TX, dicloran [99-30-9] + TX, diethofencarb [87130-20-9] + TX, dimetho- morph [110488-70-5] + TX, SYP-LI90 (Flumorph) [211867-47-9] + TX, dithianon [3347-22-6] + TX, ethaboxam [162650-77-3] + TX, etridiazole [2593-15-9] + TX, famoxadone [131807-57-3] +
T fenamidone [161326-34-7] + TX, fenoxanil [115852-48-7] + TX, fentin [668-34-8] + TX, ferimzone [89269-64-7] + TX, fluazinam [79622-59-6] + TX, fluopicolide [2391 10-15-7] + TX, flusulfamide
[106917-52-6] + TX, fenhexamid [126833-17-8] + TX, fosetyl-aluminium [39148-24-8] + TX, hymexazol [10004-44-1] + TX, iprovalicarb [140923-17-7] + TX, IKF-916 (Cyazofamid) [120116-88-3] + TX, kasugamycin [6980-18-3] + TX, methasulfocarb [66952-49-6] + TX, metrafenone [220899-03-6] + TX, pencycuron [66063-05-6] + TX, phthalide [27355-22-2] + TX, polyoxins [1 1 1 13-80-7] + TX, probenazole [27605-76-1] + TX, propamocarb [25606-41-1] + TX, proquinazid [189278-12-4] + TX, pyroquilon [57369-32-1] + TX, quinoxyfen [124495-18-7] + TX, quintozene [82-68-8] + TX, sulfur
[7704-34-9] + TX, tiadinil [223580-51-6] + TX, triazoxide [72459-58-6] + TX, tricyclazole [41814-78-2] + TX, triforine [26644-46-2] + TX, validamycin [37248-47-8] + TX, zoxamide (RH7281 ) [156052-68-5] + TX,mandipropamid [374726-62-2] + TXjsopyrazam [881685-58-1] + TX, sedaxane [874967-67-6] + TX, 3-difluoromethyl-1-methyl-1 H-pyrazole-4-carboxylic acid (9-dichloromethylene-1 ,2,3,4-tetrahydro- 1 ,4-methano-naphthalen-5-yl)-amide (dislosed in WO 2007/048556) + TX, 3-difluoromethyl-1-methyl- 1 H-pyrazole-4-carboxylic acid (3',4',5'-trifluoro-biphenyl-2-yl)-amide (disclosed in WO 2006/087343) + TX,[(3S,4R,4aR,6S,6aS, 12R, 12aS, 12bS)-3-[(cyclopropylcarbonyl)oxy]- 1 ,3,4,4a,5,6,6a, 12,12a, 12b- decahydro-6, 12-dihydroxy-4,6a,12b-trimethyl-1 1-ox^
e]pyran-4-yl]methyl-cyclopropanecarboxylate [915972-17-7] + TX and 1 ,3,5-trimethyl-N-(2-methyl-1- oxopropyl)-N-[3-(2-methylpropyl)-4-[2,2,2-trifl^
pyrazole-4-carboxamide [926914-55-8] + TX; lancotrione [1486617-21-3] + TX, florpyrauxifen [943832- 81-3] ] + TX, ipfentrifluconazole[1417782-08-1] + TX, mefentrifluconazole [1417782-03-6] + TX, quinofumelin [861647-84-9] + TX, chloroprallethrin [399572-87-3] + TX, cyhalodiamide [1262605-53- 7] ] + TX, fluazaindolizine [1254304-22-7] + TX, fluxametamide [928783-29-3] + TX, epsilon- metofluthrin [240494-71-7] + TX, epsilon-momfluorothrin [1065124-65-3] + TX, pydiflumetofen
[1228284-64-7] + TX, kappa-bifenthrin [439680-76-9] + TX, broflanilide [1207727-04-5] + TX, dicloromezotiaz [1263629-39-5] + TX, dipymetitrone [161 14-35-5] + TX, pyraziflumid [942515-63-1] + TX, kappa-tefluthrin [391634-71-2] + TX, fenpicoxamid [517875-34-2] + TX; fluindapyr [1383809-87-7] + TX; alpha-bromadiolone [28772-56-7] + TX; flupyrimin [1689566-03-7] + TX; benzpyrimoxan
[1449021-97-9] + TX; acynonapyr [1332838-17-1] + TX; inpyrfluxam [1352994-67-2] + TX, isoflucypram [1255734-28-1] + TX; rescalure [64309-03-1] + TX; aminopyrifen [1531626-08-0] + TX; tyclopyrazoflor [1477919-27-9] + TX; and spiropidion [1229023-00-0] + TX; and
microbials including: Acinetobacter Iwoffii + TX, Acremonium alternatum + TX + TX, Acremonium cephalosporium + TX + TX, Acremonium diospyri + TX, Acremonium obclavatum + TX, Adoxophyes orana granulovirus (AdoxGV) (Capex®) + TX, Agrobacterium radiobacter strain K84 (Galltrol-A®) + TX, Alternaria alternate + TX, Alternaria cassia + TX, Alternaria destruens (Smolder®) + TX,
Ampelomyces quisqualis (AQ10®) + TX, Aspergillus flavus AF36 (AF36®) + TX, Aspergillus flavus NRRL 21882 (Aflaguard®) + TX, Aspergillus spp. + TX, Aureobasidium pullulans + TX, Azospirillum + TX, (MicroAZ® + TX, TAZO B®) + TX, Azotobacter + TX, Azotobacter chroocuccum (Azotomeal®) + TX, Azotobacter cysts (Bionatural Blooming Blossoms®) + TX, Bacillus amyloliquefaciens + TX,
Bacillus cereus + TX, Bacillus chitinosporus strain CM-1 + TX, Bacillus chitinosporus strain AQ746 + TX, Bacillus licheniformis strain HB-2 (Biostart™ Rhizoboost®) + TX, Bacillus licheniformis strain 3086 (EcoGuard® + TX, Green Releaf®) + TX, Bacillus circulans + TX, Bacillus firmus (BioSafe® + TX, BioNem-WP® + TX, VOTiVO®) + TX, Bacillus firmus strain 1-1582 + TX, Bacillus macerans + TX, Bacillus marismortui + TX, Bacillus megaterium + TX, Bacillus mycoides strain AQ726 + TX, Bacillus papillae (Milky Spore Powder®) + TX, Bacillus pumilus spp. + TX, Bacillus pumilus strain GB34 (Yield Shield®) + TX, Bacillus pumilus strain AQ717 + TX, Bacillus pumilus strain QST 2808 (Sonata® + TX, Ballad Plus®) + TX, Bacillus spahericus (VectoLex®) + TX, Bacillus spp. + TX, Bacillus spp. strain AQ175 + TX, Bacillus spp. strain AQ177 + TX, Bacillus spp. strain AQ178 + TX, Bacillus subtilis strain QST 713 (CEASE® + TX, Serenade® + TX, Rhapsody®) + TX, Bacillus subtilis strain QST 714
(JAZZ®) + TX, Bacillus subtilis strain AQ153 + TX, Bacillus subtilis strain AQ743 + TX, Bacillus subtilis strain QST3002 + TX, Bacillus subtilis strain QST3004 + TX, Bacillus subtilis var. amyloliquefaciens strain FZB24 (Taegro® + TX, Rhizopro®) + TX, Bacillus thuringiensis Cry 2Ae + TX, Bacillus thuringiensis Cry1 Ab + TX, Bacillus thuringiensis aizawai GC 91 (Agree®) + TX, Bacillus thuringiensis israelensis (BMP123® + TX, Aquabac® + TX, VectoBac®) + TX, Bacillus thuringiensis kurstaki
(Javelin® + TX, Deliver® + TX, CryMax® + TX, Bonide® + TX, Scutella WP® + TX, Turilav WP ® + TX, Astuto® + TX, Dipel WP® + TX, Biobit® + TX, Foray®) + TX, Bacillus thuringiensis kurstaki BMP 123 (Baritone®) + TX, Bacillus thuringiensis kurstaki HD-1 (Bioprotec-CAF / 3P®) + TX, Bacillus
thuringiensis strain BD#32 + TX, Bacillus thuringiensis strain AQ52 + TX, Bacillus thuringiensis var. aizawai (XenTari® + TX, DiPel®) + TX, bacteria spp. (GROWMEND® + TX, GROWSWEET® + TX, Shootup®) + TX, bacteriophage of Clavipacter michiganensis (AgriPhage®) + TX, Bakflor® + TX, Beauveria bassiana (Beaugenic® + TX, Brocaril WP®) + TX, Beauveria bassiana GHA (Mycotrol ES® + TX, Mycotrol O® + TX, BotaniGuard®) + TX, Beauveria brongniartii (Engerlingspilz® + TX,
Schweizer Beauveria® + TX, Melocont®) + TX, Beauveria spp. + TX, Botrytis cineria + TX,
Bradyrhizobium japonicum (TerraMax®) + TX, Brevibacillus brevis + TX, Bacillus thuringiensis tenebrionis (Novodor®) + TX, BtBooster + TX, Burkholderia cepacia (Deny® + TX, Intercept® + TX, Blue Circle®) + TX, Burkholderia gladii + TX, Burkholderia gladioli + TX, Burkholderia spp. + TX, Canadian thistle fungus (CBH Canadian Bioherbicide®) + TX, Candida butyri + TX, Candida famata + TX, Candida fructus + TX, Candida glabrata + TX, Candida guilliermondii + TX, Candida melibiosica + TX, Candida oleophila strain O + TX, Candida parapsilosis + TX, Candida pelliculosa + TX, Candida pulcherrima + TX, Candida reukaufii + TX, Candida saitoana (Bio-Coat® + TX, Biocure®) + TX, Candida sake + TX, Candida spp. + TX, Candida tenius + TX, Cedecea dravisae + TX, Cellulomonas flavigena + TX, Chaetomium cochliodes (Nova-Cide®) + TX, Chaetomium globosum (Nova-Cide®) + TX, Chromobacterium subtsugae strain PRAA4-1T (Grandevo®) + TX, Cladosporium cladosporioides + TX, Cladosporium oxysporum + TX, Cladosporium chlorocephalum + TX, Cladosporium spp. + TX, Cladosporium tenuissimum + TX, Clonostachys rosea (EndoFine®) + TX, Colletotrichum acutatum + TX, Coniothyrium minitans (Cotans WG®) + TX, Coniothyrium spp. + TX, Cryptococcus albidus (YIELDPLUS®) + TX, Cryptococcus humicola + TX, Cryptococcus infirmo-miniatus + TX,
Cryptococcus laurentii + TX, Cryptophlebia leucotreta granulovirus (Cryptex®) + TX, Cupriavidus campinensis + TX, Cydia pomonella granulovirus (CYD-X®) + TX, Cydia pomonella granulovirus (Madex® + TX, Madex Plus® + TX, Madex Max/ Carpovirusine®) + TX, Cylindrobasidium laeve (Stumpout®) + TX, Cylindrocladium + TX, Debaryomyces hansenii + TX, Drechslera hawaiinensis + TX, Enterobacter cloacae + TX, Enterobacteriaceae + TX, Entomophtora virulenta (Vektor®) + TX, Epicoccum nigrum + TX, Epicoccum purpurascens + TX, Epicoccum spp. + TX, Filobasidium floriforme + TX, Fusarium acuminatum + TX, Fusarium chlamydosporum + TX, Fusarium oxysporum (Fusaclean® / Biofox C®) + TX, Fusarium proliferatum + TX, Fusarium spp. + TX, Galactomyces geotrichum + TX, Gliocladium catenulatum (Primastop® + TX, Prestop®) + TX, Gliocladium roseum + TX, Gliocladium spp. (SoilGard®) + TX, Gliocladium virens (Soilgard®) + TX, Granulovirus
(Granupom®) + TX, Halobacillus halophilus + TX, Halobacillus litoralis + TX, Halobacillus trueperi + TX, Halomonas spp. + TX, Halomonas subglaciescola + TX, Halovibrio variabilis + TX, Hanseniaspora uvarum + TX, Helicoverpa armigera nucleopolyhedrovirus (Helicovex®) + TX, Helicoverpa zea nuclear polyhedrosis virus (Gemstar®) + TX, Isoflavone - formononetin (Myconate®) + TX, Kloeckera apiculata + TX, Kloeckera spp. + TX, Lagenidium giganteum (Laginex®) + TX, Lecanicillium longisporum (Vertiblast®) + TX, Lecanicillium muscarium (Vertikil®) + TX, Lymantria Dispar nucleopolyhedrosis virus (Disparvirus®) + TX, Marinococcus halophilus + TX, Meira geulakonigii + TX, Metarhizium anisopliae (Met52®) + TX, Metarhizium anisopliae (Destruxin WP®) + TX, Metschnikowia
fruticola (Shemer®) + TX, Metschnikowia pulcherrima + TX, Microdochium dimerum (Antibot®) + TX, Micromonospora coerulea + TX, Microsphaeropsis ochracea + TX, Muscodor albus 620 (Muscudor®) + TX, Muscodor roseus strain A3-5 + TX, Mycorrhizae spp. (AMykor® + TX, Root Maximizer®) + TX, Myrothecium verrucaria strain AARC-0255 (DiTera®) + TX, BROS PLUS® + TX, Ophiostoma piliferum strain D97 (Sylvanex®) + TX, Paecilomyces farinosus + TX, Paecilomyces fumosoroseus (PFR-97® + TX, PreFeRal®) + TX, Paecilomyces linacinus (Biostat WP®) + TX, Paecilomyces lilacinus strain 251 (MeloCon WG®) + TX, Paenibacillus polymyxa + TX, Pantoea agglomerans (BlightBan C9-1®) + TX, Pantoea spp. + TX, Pasteuria spp. (Econem®) + TX, Pasteuria nishizawae + TX, Penicillium aurantiogriseum + TX, Penicillium billai (Jumpstart® + TX, TagTeam®) + TX, Penicillium
brevicompactum + TX, Penicillium frequentans + TX, Penicillium griseofulvum + TX, Penicillium purpurogenum + TX, Penicillium spp. + TX, Penicillium viridicatum + TX, Phlebiopsis gigantean (Rotstop®) + TX, phosphate solubilizing bacteria (Phosphomeal®) + TX, Phytophthora cryptogea + TX, Phytophthora palmivora (Devine®) + TX, Pichia anomala + TX, Pichia guilermondii + TX, Pichia membranaefaciens + TX, Pichia onychis + TX, Pichia stipites + TX, Pseudomonas aeruginosa + TX, Pseudomonas aureofasciens (Spot-Less Biofungicide®) + TX, Pseudomonas cepacia + TX,
Pseudomonas chlororaphis (AtEze®) + TX, Pseudomonas corrugate + TX, Pseudomonas fluorescens strain A506 (BlightBan A506®) + TX, Pseudomonas putida + TX, Pseudomonas reactans + TX, Pseudomonas spp. + TX, Pseudomonas syringae (Bio-Save®) + TX, Pseudomonas viridiflava + TX, Pseudomons fluorescens (Zequanox®) + TX, Pseudozyma flocculosa strain PF-A22 UL (Sporodex L®) + TX, Puccinia canaliculata + TX, Puccinia thlaspeos (Wood Warrior®) + TX, Pythium
paroecandrum + TX, Pythium oligandrum (Polygandron® + TX, Polyversum®) + TX, Pythium periplocum + TX, Rhanella aquatilis + TX, Rhanella spp. + TX, Rhizobia (Dormal® + TX, Vault®) + TX, Rhizoctonia + TX, Rhodococcus globerulus strain AQ719 + TX, Rhodosporidium diobovatum + TX, Rhodosporidium toruloides + TX, Rhodotorula spp. + TX, Rhodotorula glutinis + TX, Rhodotorula graminis + TX, Rhodotorula mucilagnosa + TX, Rhodotorula rubra + TX, Saccharomyces cerevisiae + TX, Salinococcus roseus + TX, Sclerotinia minor + TX, Sclerotinia minor (SARRITOR®) + TX, Scytalidium spp. + TX, Scytalidium uredinicola + TX, Spodoptera exigua nuclear polyhedrosis virus (Spod-X® + TX, Spexit®) + TX, Serratia marcescens + TX, Serratia plymuthica + TX, Serratia spp. + TX, Sordaria fimicola + TX, Spodoptera littoralis nucleopolyhedrovirus (Littovir®) + TX,
Sporobolomyces roseus + TX, Stenotrophomonas maltophilia + TX, Streptomyces ahygroscopicus + TX, Streptomyces albaduncus + TX, Streptomyces exfoliates + TX, Streptomyces galbus + TX, Streptomyces griseoplanus + TX, Streptomyces griseoviridis (Mycostop®) + TX, Streptomyces lydicus (Actinovate®) + TX, Streptomyces lydicus WYEC-108 (ActinoGrow®) + TX, Streptomyces violaceus + TX, Tilletiopsis minor + TX, Tilletiopsis spp. + TX, Trichoderma asperellum (T34 Biocontrol®) + TX, Trichoderma gamsii (Tenet®) + TX, Trichoderma atroviride (Plantmate®) + TX, Trichoderma hamatum TH 382 + TX, Trichoderma harzianum rifai (Mycostar®) + TX, Trichoderma harzianum T-22 (Trianum- P® + TX, PlantShield HC® + TX, RootShield® + TX, Trianum-G®) + TX, Trichoderma harzianum T-39 (Trichodex®) + TX, Trichoderma inhamatum + TX, Trichoderma koningii + TX, Trichoderma spp. LC
52 (Sentinel®) + TX, Trichoderma lignorum + TX, Trichoderma longibrachiatum + TX, Trichoderma polysporum (Binab T®) + TX, Trichoderma taxi + TX, Trichoderma virens + TX, Trichoderma virens (formerly Gliocladium virens GL-21 ) (SoilGuard®) + TX, Trichoderma viride + TX, Trichoderma viride strain ICC 080 (Remedier®) + TX, Trichosporon pullulans + TX, Trichosporon spp. + TX,
Trichothecium spp. + TX, Trichothecium roseum + TX, Typhula phacorrhiza strain 94670 + TX,
Typhula phacorrhiza strain 94671 + TX, Ulocladium atrum + TX, Ulocladium oudemansii (Botry-Zen®) + TX, Ustilago maydis + TX, various bacteria and supplementary micronutrients (Natural II®) + TX, various fungi (Millennium Microbes®) + TX, Verticillium chlamydosporium + TX, Verticillium lecanii (Mycotal® + TX, Vertalec®) + TX, Vip3Aa20 (VIPtera®) + TX, Virgibaclillus marismortui + TX, Xanthomonas campestris pv. Poae (Camperico®) + TX, Xenorhabdus bovienii + TX, Xenorhabdus nematophilus; and
Plant extracts including: pine oil (Retenol®) + TX, azadirachtin (Plasma Neem Oil® + TX, AzaGuard® + TX, MeemAzal® + TX, Molt-X® + TX, Botanical IGR (Neemazad® + TX, Neemix®) + TX, canola oil (Lilly Miller Vegol®) + TX, Chenopodium ambrosioides near ambrosioides (Requiem®) + TX, Chrysanthemum extract (Crisant®) + TX, extract of neem oil (Trilogy®) + TX, essentials oils of
Labiatae (Botania®) + TX, extracts of clove rosemary peppermint and thyme oil (Garden insect killer®) + TX, Glycinebetaine (Greenstim®) + TX, garlic + TX, lemongrass oil (GreenMatch®) + TX, neem oil + TX, Nepeta cataria (Catnip oil) + TX, Nepeta catarina + TX, nicotine + TX, oregano oil (MossBuster®) + TX, Pedaliaceae oil (Nematon®) + TX, pyrethrum + TX, Quillaja saponaria (NemaQ®) + TX, Reynoutria sachalinensis (Regalia® + TX, Sakalia®) + TX, rotenone (Eco Roten®) + TX, Rutaceae plant extract (Soleo®) + TX, soybean oil (Ortho ecosense®) + TX, tea tree oil (Timorex Gold®) + TX, thymus oil + TX, AGNIQUE® MMF + TX, BugOil® + TX, mixture of rosemary sesame pepermint thyme and cinnamon extracts (EF 300®) + TX, mixture of clove rosemary and peppermint extract (EF 400®) + TX, mixture of clove pepermint garlic oil and mint (Soil Shot®) + TX, kaolin (Screen®) + TX, storage glucam of brown algae (Laminarin®); and
pheromones including: blackheaded fireworm pheromone (3M Sprayable Blackheaded Fireworm Pheromone®) + TX, Codling Moth Pheromone (Paramount dispenser-(CM)/ Isomate C-Plus®) + TX, Grape Berry Moth Pheromone (3M MEC-GBM Sprayable Pheromone®) + TX, Leafroller pheromone (3M MEC - LR Sprayable Pheromone®) + TX, Muscamone (Snip7 Fly Bait® + TX, Starbar Premium Fly Bait®) + TX, Oriental Fruit Moth Pheromone (3M oriental fruit moth sprayable pheromone®) + TX, Peachtree Borer Pheromone (Isomate-P®) + TX, Tomato Pinworm Pheromone (3M Sprayable pheromone®) + TX, Entostat powder (extract from palm tree) (Exosex CM®) + TX, (E + ΤΧ,Ζ + ΤΧ,Ζ)- 3 + TX,8 + TX,1 1 Tetradecatrienyl acetate + TX, (Z + ΤΧ,Ζ + TX,E)-7 + TX, 1 1 + TX,13- Hexadecatrienal + TX, (E + TX,Z)-7 + TX,9-Dodecadien-1-yl acetate + TX, 2-Methyl-1-butanol + TX, Calcium acetate + TX, Scenturion® + TX, Biolure® + TX, Check-Mate® + TX, Lavandulyl senecioate; and
Macrobials including: Aphelinus abdominalis + TX, Aphidius ervi (Aphelinus-System®) + TX,
Acerophagus papaya + TX, Adalia bipunctata (Adalia-System®) + TX, Adalia bipunctata (Adaline®) +
TX, Adalia bipunctata (Aphidalia®) + TX, Ageniaspis citricola + TX, Ageniaspis fuscicollis + TX, Amblyseius andersoni (Anderline® + TX, Andersoni-System®) + TX, Amblyseius californicus (Amblyline® + TX, Spical®) + TX, Amblyseius cucumeris (Thripex® + TX, Bugline cucumeris®) + TX, Amblyseius fallacis (Fallacis®) + TX, Amblyseius swirskii (Bugline swirskii® + TX, Swirskii-Mite®) + TX, Amblyseius womersleyi (WomerMite®) + TX, Amitus hesperidum + TX, Anagrus atomus + TX, Anagyrus fusciventris + TX, Anagyrus kamali + TX, Anagyrus loecki + TX, Anagyrus pseudococci (Citripar®) + TX, Anicetus benefices + TX, Anisopteromalus calandrae + TX, Anthocoris nemoralis (Anthocoris-System®) + TX, Aphelinus abdominalis (Apheline® + TX, Aphiline®) + TX, Aphelinus asychis + TX, Aphidius colemani (Aphipar®) + TX, Aphidius ervi (Ervipar®) + TX, Aphidius gifuensis + TX, Aphidius matricariae (Aphipar-M®) + TX, Aphidoletes aphidimyza (Aphidend®) + TX, Aphidoletes aphidimyza (Aphidoline®) + TX, Aphytis lingnanensis + TX, Aphytis melinus + TX, Aprostocetus hagenowii + TX, Atheta coriaria (Staphyline®) + TX, Bombus spp. + TX, Bombus terrestris (Natupol Beehive®) + TX, Bombus terrestris (Beeline® + TX, Tripol®) + TX, Cephalonomia stephanoderis + TX, Chilocorus nigritus + TX, Chrysoperla carnea (Chrysoline®) + TX, Chrysoperla carnea
(Chrysopa®) + TX, Chrysoperla rufilabris + TX, Cirrospilus ingenuus + TX, Cirrospilus quadristriatus + TX, Citrostichus phyllocnistoides + TX, Closterocerus chamaeleon + TX, Closterocerus spp. + TX, Coccidoxenoides perminutus (Planopar®) + TX, Coccophagus cowperi + TX, Coccophagus lycimnia + TX, Cotesia flavipes + TX, Cotesia plutellae + TX, Cryptolaemus montrouzieri (Cryptobug® + TX, Cryptoline®) + TX, Cybocephalus nipponicus + TX, Dacnusa sibirica + TX, Dacnusa sibirica
(Minusa®) + TX, Diglyphus isaea (Diminex®) + TX, Delphastus catalinae (Delphastus®) + TX, Delphastus pusillus + TX, Diachasmimorpha krausii + TX, Diachasmimorpha longicaudata + TX, Diaparsis jucunda + TX, Diaphorencyrtus aligarhensis + TX, Diglyphus isaea + TX, Diglyphus isaea (Miglyphus® + TX, Digline®) + TX, Dacnusa sibirica (DacDigline® + TX, Minex®) + TX, Diversinervus spp. + TX, Encarsia citrina + TX, Encarsia formosa (Encarsia max® + TX, Encarline® + TX, En- Strip®) + TX, Eretmocerus eremicus (Enermix®) + TX, Encarsia guadeloupae + TX, Encarsia haitiensis + TX, Episyrphus balteatus (Syrphidend®) + TX, Eretmoceris siphonini + TX, Eretmocerus californicus + TX, Eretmocerus eremicus (Ercal® + TX, Eretline e®) + TX, Eretmocerus eremicus (Bemimix®) + TX, Eretmocerus hayati + TX, Eretmocerus mundus (Bemipar® + TX, Eretline m®) + TX, Eretmocerus siphonini + TX, Exochomus quadripustulatus + TX, Feltiella acarisuga (Spidend®) + TX, Feltiella acarisuga (Feltiline®) + TX, Fopius arisanus + TX, Fopius ceratitivorus + TX,
Formononetin (Wirless Beehome®) + TX, Franklinothrips vespiformis (Vespop®) + TX, Galendromus occidentalis + TX, Goniozus legneri + TX, Habrobracon hebetor + TX, Harmonia axyridis
(HarmoBeetle®) + TX, Heterorhabditis spp. (Lawn Patrol®) + TX, Heterorhabditis bacteriophora (NemaShield HB® + TX, Nemaseek® + TX, Terranem-Nam® + TX, Terranem® + TX, Larvanem® + TX, B-Green® + TX, NemAttack ® + TX, Nematop®) + TX, Heterorhabditis megidis (Nemasys H® + TX, BioNem H® + TX, Exhibitline hm® + TX, Larvanem-M®) + TX, Hippodamia convergens + TX, Hypoaspis aculeifer (Aculeifer-System® + TX, Entomite-A®) + TX, Hypoaspis miles (Hypoline m® + TX, Entomite-M®) + TX, Lbalia leucospoides + TX, Lecanoideus floccissimus + TX, Lemophagus
errabundus + TX, Leptomastidea abnormis + TX, Leptomastix dactylopii (Leptopar®) + TX,
Leptomastix epona + TX, Lindorus lophanthae + TX, Lipolexis oregmae + TX, Lucilia caesar
(Natufly®) + TX, Lysiphlebus testaceipes + TX, Macrolophus caliginosus (Mirical-N® + TX, Macroline c® + TX, Mirical®) + TX, Mesoseiulus longipes + TX, Metaphycus flavus + TX, Metaphycus lounsburyi + TX, Micromus angulatus (Milacewing®) + TX, Microterys flavus + TX, Muscidifurax raptorellus and Spalangia cameroni (Biopar®) + TX, Neodryinus typhlocybae + TX, Neoseiulus californicus + TX, Neoseiulus cucumeris (THRYPEX®) + TX, Neoseiulus fallacis + TX, Nesideocoris tenuis
(NesidioBug® + TX, Nesibug®) + TX, Ophyra aenescens (Biofly®) + TX, Onus insidiosus (Thripor-I® + TX, Oriline i®) + TX, Onus laevigatus (Thripor-L® + TX, Oriline I®) + TX, Orius majusculus (Oriline m®) + TX, Orius strigicollis (Thripor-S®) + TX, Pauesia juniperorum + TX, Pediobius foveolatus + TX, Phasmarhabditis hermaphrodita (Nemaslug®) + TX, Phymastichus coffea + TX, Phytoseiulus macropilus + TX, Phytoseiulus persimilis (Spidex® + TX, Phytoline p®) + TX, Podisus maculiventris (Podisus®) + TX, Pseudacteon curvatus + TX, Pseudacteon obtusus + TX, Pseudacteon tricuspis + TX, Pseudaphycus maculipennis + TX, Pseudleptomastix mexicana + TX, Psyllaephagus pilosus + TX, Psyttalia concolor (complex) + TX, Quadrastichus spp. + TX, Rhyzobius lophanthae + TX, Rodolia cardinalis + TX, Rumina decollate + TX, Semielacher petiolatus + TX, Sitobion avenae (Ervibank®) + TX, Steinemema carpocapsae (Nematac C® + TX, Millenium® + TX, BioNem C® + TX, NemAttack® + TX, Nemastar® + TX, Capsanem®) + TX, Steinemema feltiae (NemaShield® + TX, Nemasys F® + TX, BioNem F® + TX, Steinernema-System® + TX, NemAttack® + TX, Nemaplus® + TX, Exhibitline sf® + TX, Scia-rid® + TX, Entonem®) + TX, Steinemema kraussei (Nemasys L® + TX, BioNem L® + TX, Exhibitline srb®) + TX, Steinemema riobrave (BioVector® + TX, BioVektor®) + TX, Steinemema scapterisci (Nematac S®) + TX, Steinemema spp. + TX, Steinernematid spp. (Guardian Nematodes®) + TX, Stethorus punctillum (Stethorus®) + TX, Tamarixia radiate + TX, Tetrastichus setifer + TX, Thripobius semiluteus + TX, Torymus sinensis + TX, Trichogramma brassicae (Tricholine b®) + TX, Trichogramma brassicae (Tricho-Strip®) + TX, Trichogramma evanescens + TX, Trichogramma minutum + TX, Trichogramma ostriniae + TX, Trichogramma platneri + TX, Trichogramma pretiosum + TX, Xanthopimpla stemmator; and
other biologicals including: abscisic acid + TX, bioSea® + TX, Chondrostereum purpureum (Chontrol Paste®) + TX, Colletotrichum gloeosporioides (Collego®) + TX, Copper Octanoate (Cueva®) + TX, Delta traps (Trapline d®) + TX, Erwinia amylovora (Harpin) (ProAct® + TX, Ni-HIBIT Gold CST®) + TX, Ferri-phosphate (Ferramol®) + TX, Funnel traps (Trapline y®) + TX, Gallex® + TX, Grower's Secret® + TX, Homo-brassonolide + TX, Iron Phosphate (Lilly Miller Worry Free Ferramol Slug & Snail Bait®) + TX, MCP hail trap (Trapline f®) + TX, Microctonus hyperodae + TX, Mycoleptodiscus terrestris (Des-X®) + TX, BioGain® + TX, Aminomite® + TX, Zenox® + TX, Pheromone trap (Thripline ams®) + TX, potassium bicarbonate (MilStop®) + TX, potassium salts of fatty acids (Sanova®) + TX, potassium silicate solution (Sil-Matrix®) + TX, potassium iodide + potassiumthiocyanate (Enzicur®) + TX, SuffOil-X® + TX, Spider venom + TX, Nosema locustae (Semaspore Organic Grasshopper
Control®) + TX, Sticky traps (Trapline YF® + TX, Rebell Amarillo®) + TX and Traps (Takitrapline y + b®) + TX.
The references in brackets behind the active ingredients, e.g. [3878-19-1] refer to the Chemical Abstracts Registry number. The above described mixing partners are known. Where the active ingredients are included in "The Pesticide Manual" [The Pesticide Manual - A World Compendium; Thirteenth Edition; Editor: C. D. S. TomLin; The British Crop Protection Council], they are described therein under the entry number given in round brackets hereinabove for the particular compound; for example, the compound "abamectin" is described under entry number (1 ). Where "[CCN]" is added hereinabove to the particular compound, the compound in question is included in the "Compendium of Pesticide Common Names", which is accessible on the internet [A. Wood; Compendium of Pesticide Common Names, Copyright © 1995-2004]; for example, the compound "acetoprole" is described under the internet address http://www.alanwood.net/pesticides/acetoprole.html. Most of the active ingredients described above are referred to hereinabove by a so-called "common name", the relevant "ISO common name" or another "common name" being used in individual cases. If the designation is not a "common name", the nature of the designation used instead is given in round brackets for the particular compound; in that case, the lUPAC name, the lUPAC/Chemical Abstracts name, a "chemical name", a "traditional name", a "compound name" or a "development code" is used or, if neither one of those designations nor a "common name" is used, an "alternative name" is employed. "CAS Reg. No" means the Chemical Abstracts Registry Number.
The active ingredient mixture of the compounds of formula I selected from Table P with active ingredients described above comprises a compound selected from Table P and an active ingredient as described above preferably in a mixing ratio of from 100: 1 to 1 :6000, especially from 50:1 to 1 :50, more especially in a ratio of from 20: 1 to 1 :20, even more especially from 10: 1 to 1 :10, very especially from 5: 1 and 1 :5, special preference being given to a ratio of from 2:1 to 1 :2, and a ratio of from 4: 1 to 2: 1 being likewise preferred, above all in a ratio of 1 : 1 , or 5: 1 , or 5:2, or 5:3, or 5:4, or 4:1 , or 4:2, or 4:3, or 3: 1 , or 3:2, or 2:1 , or 1 :5, or 2:5, or 3:5, or 4:5, or 1 :4, or 2:4, or 3:4, or 1 :3, or 2:3, or 1 :2, or 1 :600, or 1 :300, or 1 :150, or 1 :35, or 2:35, or 4:35, or 1 :75, or 2:75, or 4:75, or 1 :6000, or 1 :3000, or 1 : 1500, or 1 :350, or 2:350, or 4:350, or 1 :750, or 2:750, or 4:750. Those mixing ratios are by weight.
The mixtures as described above can be used in a method for controlling pests, which comprises applying a composition comprising a mixture as described above to the pests or their environment, with the exception of a method for treatment of the human or animal body by surgery or therapy and diagnostic methods practised on the human or animal body.
The mixtures comprising a compound of formula I selected from Table P and one or more active ingredients as described above can be applied, for example, in a single "ready-mix" form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a "tank-mix", and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days. The order of applying the compounds of formula I selected from Table P and the active ingredients as described above is not essential for working the present invention.
The compositions according to the invention can also comprise further solid or liquid auxiliaries, such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematocides, plant activators, molluscicides or herbicides. The compositions according to the invention are prepared in a manner known per se, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries). These processes for the preparation of the compositions and the use of the compounds I for the preparation of these compositions are also a subject of the invention.
The application methods for the compositions, that is the methods of controlling pests of the abovementioned type, such as spraying, atomizing, dusting, brushing on, dressing, scattering or pouring - which are to be selected to suit the intended aims of the prevailing circumstances - and the use of the compositions for controlling pests of the abovementioned type are other subjects of the invention. Typical rates of concentration are between 0.1 and 1000 ppm, preferably between 0.1 and 500 ppm, of active ingredient. The rate of application per hectare is generally 1 to 2000 g of active ingredient per hectare, in particular 10 to 1000 g/ha, preferably 10 to 600 g/ha. A preferred method of application in the field of crop protection is application to the foliage of the plants (foliar application), it being possible to select frequency and rate of application to match the danger of infestation with the pest in question. Alternatively, the active ingredient can reach the plants via the root system (systemic action), by drenching the locus of the plants with a liquid composition or by incorporating the active ingredient in solid form into the locus of the plants, for example into the soil, for example in the form of granules (soil application). In the case of paddy rice crops, such granules can be metered into the flooded paddy-field.
The compounds of the invention and compositions thereof are also be suitable for the protection of plant propagation material, for example seeds, such as fruit, tubers or kernels, or nursery plants, against pests of the abovementioned type. The propagation material can be treated with the compound prior to planting, for example seed can be treated prior to sowing. Alternatively, the compound can be applied to seed kernels (coating), either by soaking the kernels in a liquid composition or by applying a layer of a solid composition. It is also possible to apply the compositions when the propagation material is planted to the site of application, for example into the seed furrow during drilling. These treatment methods for plant propagation material and the plant propagation material thus treated are further subjects of the invention. Typical treatment rates would depend on the plant and pest/fungi to be controlled and are generally between 1 to 200 grams per 100 kg of seeds, preferably between 5 to 150 grams per 100 kg of seeds, such as between 10 to 100 grams per 100 kg of seeds.
The term seed embraces seeds and plant propagules of all kinds including but not limited to true seeds, seed pieces, suckers, corns, bulbs, fruit, tubers, grains, rhizomes, cuttings, cut shoots and the like and means in a preferred embodiment true seeds.
The present invention also comprises seeds coated or treated with or containing a compound of formula I. The term "coated or treated with and/or containing" generally signifies that the active ingredient is for the most part on the surface of the seed at the time of application, although a greater or lesser part of the ingredient may penetrate into the seed material, depending on the method of application. When the said seed product is (re)planted, it may absorb the active ingredient. In an embodiment, the present invention makes available a plant propagation material adhered thereto with a compound of formula (I). Further, it is hereby made available, a composition comprising a plant propagation material treated with a compound of formula (I).
Seed treatment comprises all suitable seed treatment techniques known in the art, such as seed dressing, seed coating, seed dusting, seed soaking and seed pelleting. The seed treatment application of the compound formula (I) can be carried out by any known methods, such as spraying or by dusting the seeds before sowing or during the sowing/planting of the seeds.
Biological Examples:
Example B1 : Diabrotica balteata (Corn root worm)
Maize sprouts placed onto an agar layer in 24-well microtiter plates were treated with aqueous test solutions prepared from 10Ό00 ppm DMSO stock solutions by spraying. After drying, the plates were infested with L2 larvae (6 to 10 per well). The samples were assessed for mortality and growth inhibition in comparison to untreated samples 4 days after infestation.
The following compounds gave an effect of at least 80% in at least one of the two categories (mortality or growth inhibition) at an application rate of 200 ppm:
P1 , P2, P3, P4, P5, P6, P7, P8, P9, P10, P1 1 , P12, P13, P14, P16, P17, P18, P20, P21 , P22, P23, P24 and P25. Example B2: Euschistus heros (Neotropical Brown Stink Bug)
Soybean leaves on agar in 24-well microtiter plates were sprayed with aqueous test solutions prepared from 10Ό00 ppm DMSO stock solutions. After drying the leaves were infested with N2 nymphs. The samples were assessed for mortality and growth inhibition in comparison to untreated samples 5 days after infestation.
The following compounds gave an effect of at least 80% in at least one of the two categories (mortality or growth inhibition) at an application rate of 200 ppm:
P3, P5, P8, P9, P10, P1 1 , P13, P14, P22, P23 and P25
Example B3: Frankliniella occidentalis (Western flower thrips)
Sunflower leaf discs were placed on agar in 24-well microtiter plates and sprayed with aqueous test solutions prepared from 10Ό00 DMSO stock solutions. After drying the leaf discs were infested with a Frankliniella population of mixed ages. The samples were assessed for mortality 7 days after infestation.
The following compounds resulted in at least 80% mortality at an application rate of 200 ppm:
P1 , P3 and P4
Example B4: Mvzus persicae (Green peach aphid)
Sunflower leaf discs were placed onto agar in a 24-well microtiter plate and sprayed with aqueous test solutions prepared from 10Ό00 ppm DMSO stock solutions. After drying, the leaf discs were infested with an aphid population of mixed ages. The samples were assessed for mortality 6 days after infestation.
The following compounds resulted in at least 80% mortality at an application rate of 200 ppm:
P1 , P3, P5, P8, P9, P15, P16, P17, P22, P23 and P25.
Example B5: Plutella xylostella (Diamond back moth)
24-well microtiter plates with artificial diet were treated with aqueous test solutions prepared from 10Ό00 ppm DMSO stock solutions by pipetting. After drying, the plates were infested with L2 larvae (10 to 15 per well). The samples were assessed for mortality and growth inhibition in comparison to untreated samples 5 days after infestation.
The following compounds gave an effect of at least 80% in at least one of the two categories (mortality or growth inhibition) at an application rate of 200 ppm:
P1 , P2, P3, P4, P5, P6, P7, P8, P9, P10, P1 1 , P12, P13, P14, P15, P16, P17, P18, P21 , P22, P23, P24, P25 and P26.
Example B6: Spodoptera littoralis (Egyptian cotton leaf worm)
Cotton leaf discs were placed onto agar in 24-well microtiter plates and sprayed with aqueous test solutions prepared from 10Ό00 ppm DMSO stock solutions. After drying the leaf discs were infested with five L1 larvae. The samples were assessed for mortality, anti-feeding effect, and growth inhibition in comparison to untreated samples 3 days after infestation. Control of Spodoptera littoralis by a test sample is given when at least one of the categories mortality, anti-feedant effect, and growth inhibition is higher than the untreated sample.
The following compounds resulted in at least 80% control at an application rate of 200 ppm:
P1 , P2, P3, P4, P5, P6, P7, P8, P9, P10, P1 1 , P12, P13, P14, P16, P17, P18, P21 , P22, P23, P24, P25 and P26
Example B7: Spodoptera littoralis (Egyptian cotton leaf worm)
Test compounds were applied by pipette from 10Ό00 ppm DMSO stock solutions into 24-well plates and mixed with agar. Lettuce seeds were placed onto the agar and the multi well plate was closed by another plate which contained also agar. After 7 days the compound was absorbed by the roots and the lettuce grew into the lid plate. The lettuce leaves were then cut off into the lid plate. Spodoptera eggs were pipetted through a plastic stencil onto a humid gel blotting paper and the lid plate was closed with it. The samples were assessed for mortality, anti-feedant effect and growth inhibition in comparison to untreated samples 6 days after infestation.
The following compounds gave an effect of at least 80% in at least one of the three categories (mortality, anti-feeding, or growth inhibition) at a test rate of 12.5 ppm:
P1 , P3, P8, P1 1 and P22.
Example B8: Tetranychus urticae (Two-spotted spider mite)
Bean leaf discs on agar in 24-well microtiter plates were sprayed with agueous test solutions prepared from 10Ό00 ppm DMSO stock solutions. After drying the leaf discs were infested with a mite population of mixed ages. The samples were assessed for mortality on mixed population (mobile stages) 8 days after infestation.
The following compounds resulted in at least 80% mortality at an application rate of 200 ppm:
P5 and P6. Example B9: Aedes aegypti (Yellow fever mosguito)
Test solutions, at an application rate of 200ppm in ethanol, were applied to 12 well tissue culture plates. Once the deposits were dry, five, two to five day old adult female Aedes aegypti were added to each well, and sustained with a 10% sucrose solution in a cotton wool plug. Assessment of knockdown was made one hour after introduction, and mortality was assessed at 24 and 48 hours after introduction.
The following compounds gave at least 80% control of Aedes aegypti after 48h and/or 24h:
P7, P9, P10, P18, P22, P23 and P25.
Example B10: Anopheles stephensi (Indian malaria mosquito)
Test solutions, at an application rate of 200 ppm in ethanol, were applied to 12 well tissue culture plates. Once the deposits were dry, five, two to five day old adult female Anopheles stephensi were added to each well, and sustained with a 10% sucrose solution in a cotton wool plug. Assessment of knockdown was made one hour after introduction, and mortality was assessed at 24 and 48 hours after introduction.
The following compounds gave at least 80% control of Anopheles stephensi after 48h and/or 24h: P7, P9, P10, P22, P23 and P25. Example B1 1 : Bemisia tabaci (Cotton white fly)
Cotton leaf discs were placed on agar in 24-well microtiter plates and sprayed with aqueous test solutions prepared from 10Ό00 ppm DMSO stock solutions. After drying the leaf discs were infested with adult white flies. The samples were checked for mortality 6 days after incubation.
Compounds were assessed for at least 80% mortality at an application rate of 200 ppm.
Example B12: Mvzus persicae (Green peach aphid)
Roots of pea seedlings infested with an aphid population of mixed ages were placed directly into aqueous test solutions prepared from 10Ό00 DMSO stock solutions. The samples were assessed for mortality 6 days after placing seedlings into test solutions.
Compounds were assessed for at least 80% mortality at a test rate of 24 ppm.
Claims
1. A compound of formula I,
wherein
A is CH or N;
Q is phenyl which can be mono- or polysubstituted by substituents selected from the group consisting of halogen, cyano, Ci-C4alkyl, Ci-C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy, Ci-C4alkylsulfanyl, Ci- C4alkylsulfinyl, Ci-C4alkylsulfonyl and Ci-C4haloalkylsulfanyl; or
Q is pyridyl or pyrimidyl which can be mono- or polysubstituted by substituents selected from the group consisting of halogen, cyano, Ci-C4alkyl, Ci-C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci- C4haloalkylsulfanyl; or
Q is pyrazolyl which is linked via a nitrogen atom to the ring which contains the substituent A, and which in turn can be substituted by halogen, cyano, Ci-C4alkyl or Ci-C4haloalkyl; or
Q is triazolyl which is linked via a nitrogen atom to the ring which contains the substituent A, and which in turn can be substituted by halogen, cyano or Ci-C4haloalkyl; or
X is S, SO or S02;
R4 is halogen, Ci-C4haloalkyl, Ci-C4haloalkylsulfanyl, Ci-C4haloalkylsulfinyl, or Ci-C4haloalkylsulfonyl; R5 is Ci-C4alkyl or C3-C6cycloalkyl-Ci-C4alkyl; and
Xi is CR6, wherein R6 is hydrogen, Ci-C4alkyl or halogen;
and agrochemically acceptable salts, stereoisomers, enantiomers, tautomers and N-oxides of those compounds.
2. A compound of formula I according to claim 1 represented by the compounds of formula 1-1
wherein R4, R5, A and Xi are as defined under formula I in claim 1 ;
X is S, SO or S02; and
Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy, Ci-C4alkylsulfanyl, Ci-C4alkylsulfinyl, Ci-C4alkylsulfonyl and Ci-C4haloalkylsulfanyl.
3. A compound of formula I according to claim 1 represented by compounds of formula 1-1 a:
wherein R4, R5, and A are as defined under formula I in claim 1 ; and
Rx is independently selected from hydrogen or halogen.
4. A compound of formula I according to claim 1 represented by the compounds of formula I-2:
wherein R4, R5, A and Xi are as defined under formula I in claim 1 ;
X is S, SO or S02; and
Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci-C4haloalkylsulfanyl.
5. A compound of formula I according to claim 1 represented by compounds of formula l-2a
wherein R4, R5, and A are as defined under formula I in claim 1 ; and
Rx is independently hydrogen or halogen.
wherein R4, R5, A and Xi are as defined under formula I in claim 1 ;
X is S, SO or S02; and
Rx is independently selected from the group consisting of hydrogen, halogen, cyano, Ci-C4alkyl, Ci- C4haloalkyl, Ci-C4haloalkoxy, Ci-C4alkoxy and Ci-C4haloalkylsulfanyl.
7. A compound of formula I according to claim 1 represented by compounds of formula l-3a
wherein R4, R5, and A are as defined under formula I in claim 1 ; and
Rx is independently hydrogen or halogen.
8. A compound of formula I according to claim 1 represented by the compounds of formula I-4:
wherein R4, R5, A and Xi are as defined under formula I in claim 1 ;
X is S, SO or S02; and
Rxa is hydrogen, cyano or Ci-C4haloalkyl.
(1-4a),
wherein R4, R5, and A are as defined under formula I in claim 1 ; and
Rxa is hydrogen, cyano or Ci-C4haloalkyl.
10. A compound of formula I according to claim 1 represented by the compounds of formula I-5:
wherein R4, R5, A and Xi are as defined under formula I in claim 1 ;
Rxa is hydrogen, cyano or halogen,
1 1. A compound of formula I according to claim 1 represented by compounds of formula l-5a
wherein R4, R5, and A are as defined under formula I in claim 1 ; and
Rxa is hydrogen, cyano or halogen.
12. A pesticidal composition, which comprises at least one compound of formula I according to claim 1 or, where appropriate, a tautomer thereof, in each case in free form or in agrochemically utilizable salt form, as active ingredient and at least one auxiliary.
13. A method for controlling pests, which comprises applying a composition according to claim 12 to the pests or their environment with the exception of a method for treatment of the human or animal body by surgery or therapy and diagnostic methods practised on the human or animal body.
14. A method for the protection of plant propagation material from the attack by pests, which comprises treating the propagation material or the site, where the propagation material is planted, with a composition according to claim 12.
15. Plant propagation material treated in accordance with the method described in claim 14.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP16200143 | 2016-11-23 | ||
| EP16200143.2 | 2016-11-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018095795A1 true WO2018095795A1 (en) | 2018-05-31 |
Family
ID=57391849
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2017/079389 Ceased WO2018095795A1 (en) | 2016-11-23 | 2017-11-16 | Pesticidally active polycyclic derivatives with sulfur containing substituents |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2018095795A1 (en) |
Cited By (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN112824402A (en) * | 2019-11-21 | 2021-05-21 | 浙江省化工研究院有限公司 | Benzimidazole derivative, preparation method and application thereof |
| WO2021204577A1 (en) | 2020-04-06 | 2021-10-14 | Basf Se | Imidazo-pyrimidone compounds as pesticides |
| WO2022128524A1 (en) | 2020-12-14 | 2022-06-23 | Basf Se | Sulfoximine pesticides |
| WO2023088718A1 (en) | 2021-11-19 | 2023-05-25 | Basf Se | Bicyclic compounds for the control of invertebrate pests |
| JP2023182586A (en) * | 2018-09-12 | 2023-12-26 | 日本化薬株式会社 | pest control agent |
| WO2024175559A1 (en) | 2023-02-23 | 2024-08-29 | Boehringer Ingelheim Vetmedica Gmbh | Tetrazole-substituted ethyl-sulfone/sulfoximine-pyridine derivatives |
| WO2024189139A1 (en) | 2023-03-14 | 2024-09-19 | Syngenta Crop Protection Ag | Control of pests resistant to insecticides |
| WO2024251913A1 (en) | 2023-06-06 | 2024-12-12 | Syngenta Crop Protection Ag | Synthesis of enantioenriched 2-cyano pyridyl sulfoxides |
| US12435083B2 (en) | 2021-09-13 | 2025-10-07 | Boehringer Ingelheim Vetmedica Gmbh | Cyclopropyl-(hetero)aryl-substituted ethylsulphonyl-pyridine derivatives |
| WO2025229111A1 (en) | 2024-05-01 | 2025-11-06 | Basf Se | Pyrimidinone derivatives as pesticidal compounds |
Citations (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| US5631072A (en) | 1995-03-10 | 1997-05-20 | Avondale Incorporated | Method and means for increasing efficacy and wash durability of insecticide treated fabric |
| WO2000015615A1 (en) | 1998-09-15 | 2000-03-23 | Syngenta Participations Ag | Pyridine ketones useful as herbicides |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003000906A2 (en) | 2001-06-22 | 2003-01-03 | Syngenta Participations Ag | Plant disease resistance genes |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003031587A2 (en) | 2001-10-09 | 2003-04-17 | The Regents Of The University Of California | Use of stat-6 inhibitors as therapeutic agents |
| WO2003034823A1 (en) | 2001-10-25 | 2003-05-01 | Siamdutch Mosquito Netting Company Limited | Treatment of fabric materials with an insecticide |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2005064072A2 (en) | 2003-12-22 | 2005-07-14 | Basf Aktiengesellschaft | Composition for the impregnation of fibers, fabrics and nettings imparting a protective activity against pests |
| WO2005113886A1 (en) | 2004-05-12 | 2005-12-01 | Basf Aktiengesellschaft | Method for the treatment of flexible substrates |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| EP1724392A2 (en) | 2005-05-04 | 2006-11-22 | Fritz Blanke Gmbh & Co. Kg | Process for the microbicidal finishing of textile surfaces |
| WO2006128870A2 (en) | 2005-06-03 | 2006-12-07 | Basf Aktiengesellschaft | Composition for the impregnation of fibers, fabrics and nettings imparting a protective activity against pests |
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2007090739A1 (en) | 2006-02-03 | 2007-08-16 | Basf Se | Process for treating substrates |
| WO2008151984A1 (en) | 2007-06-12 | 2008-12-18 | Basf Se | Aqueous formulation and process for the impregnation of non-living-materials imparting a protective activity against pests |
| WO2009010455A2 (en) | 2007-07-13 | 2009-01-22 | Addex Pharma S.A. | Pyrazole derivatives as modulators of metabotropic glutamate receptors |
| WO2012049280A1 (en) | 2010-10-15 | 2012-04-19 | Xolution Gmbh | A method for producing filled and reclosable pressure vessels |
| WO2012092115A1 (en) | 2010-12-29 | 2012-07-05 | E. I. Du Pont De Nemours And Company | Mesoionic pyrido [1,2 -a] pyrimidine pesticides |
| WO2012133607A1 (en) | 2011-03-31 | 2012-10-04 | アステラス製薬株式会社 | Pyrazole compound |
| WO2013191113A1 (en) | 2012-06-18 | 2013-12-27 | 住友化学株式会社 | Fused heterocyclic compound |
| EP2749561A1 (en) | 2012-12-27 | 2014-07-02 | Kyoto University | Radioactive quinolinone derivative and pharmaceutical drug comprising the same |
| WO2015000715A1 (en) | 2013-07-02 | 2015-01-08 | Syngenta Participations Ag | Pesticidally active bi- or tricyclic heterocycles with sulfur containing substituents |
| WO2015087458A1 (en) | 2013-12-13 | 2015-06-18 | 住友化学株式会社 | Fused heterocyclic compound and pest control application thereof |
| WO2016041819A1 (en) * | 2014-09-16 | 2016-03-24 | Syngenta Participations Ag | Pesticidally active tetracyclic derivatives with sulphur containing substituents |
| WO2016046071A1 (en) * | 2014-09-25 | 2016-03-31 | Syngenta Participations Ag | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
| WO2016058928A1 (en) * | 2014-10-14 | 2016-04-21 | Syngenta Participations Ag | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
| WO2016071214A1 (en) * | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Pesticidally active polycyclic derivatives with sulfur containing substituents |
-
2017
- 2017-11-16 WO PCT/EP2017/079389 patent/WO2018095795A1/en not_active Ceased
Patent Citations (39)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| US5631072A (en) | 1995-03-10 | 1997-05-20 | Avondale Incorporated | Method and means for increasing efficacy and wash durability of insecticide treated fabric |
| WO2000015615A1 (en) | 1998-09-15 | 2000-03-23 | Syngenta Participations Ag | Pyridine ketones useful as herbicides |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003000906A2 (en) | 2001-06-22 | 2003-01-03 | Syngenta Participations Ag | Plant disease resistance genes |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003031587A2 (en) | 2001-10-09 | 2003-04-17 | The Regents Of The University Of California | Use of stat-6 inhibitors as therapeutic agents |
| WO2003034823A1 (en) | 2001-10-25 | 2003-05-01 | Siamdutch Mosquito Netting Company Limited | Treatment of fabric materials with an insecticide |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2005064072A2 (en) | 2003-12-22 | 2005-07-14 | Basf Aktiengesellschaft | Composition for the impregnation of fibers, fabrics and nettings imparting a protective activity against pests |
| WO2005113886A1 (en) | 2004-05-12 | 2005-12-01 | Basf Aktiengesellschaft | Method for the treatment of flexible substrates |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| EP1724392A2 (en) | 2005-05-04 | 2006-11-22 | Fritz Blanke Gmbh & Co. Kg | Process for the microbicidal finishing of textile surfaces |
| WO2006128870A2 (en) | 2005-06-03 | 2006-12-07 | Basf Aktiengesellschaft | Composition for the impregnation of fibers, fabrics and nettings imparting a protective activity against pests |
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2007090739A1 (en) | 2006-02-03 | 2007-08-16 | Basf Se | Process for treating substrates |
| WO2008151984A1 (en) | 2007-06-12 | 2008-12-18 | Basf Se | Aqueous formulation and process for the impregnation of non-living-materials imparting a protective activity against pests |
| WO2009010455A2 (en) | 2007-07-13 | 2009-01-22 | Addex Pharma S.A. | Pyrazole derivatives as modulators of metabotropic glutamate receptors |
| WO2012049280A1 (en) | 2010-10-15 | 2012-04-19 | Xolution Gmbh | A method for producing filled and reclosable pressure vessels |
| WO2012092115A1 (en) | 2010-12-29 | 2012-07-05 | E. I. Du Pont De Nemours And Company | Mesoionic pyrido [1,2 -a] pyrimidine pesticides |
| WO2012133607A1 (en) | 2011-03-31 | 2012-10-04 | アステラス製薬株式会社 | Pyrazole compound |
| WO2013191113A1 (en) | 2012-06-18 | 2013-12-27 | 住友化学株式会社 | Fused heterocyclic compound |
| EP2749561A1 (en) | 2012-12-27 | 2014-07-02 | Kyoto University | Radioactive quinolinone derivative and pharmaceutical drug comprising the same |
| WO2015000715A1 (en) | 2013-07-02 | 2015-01-08 | Syngenta Participations Ag | Pesticidally active bi- or tricyclic heterocycles with sulfur containing substituents |
| WO2015087458A1 (en) | 2013-12-13 | 2015-06-18 | 住友化学株式会社 | Fused heterocyclic compound and pest control application thereof |
| WO2016041819A1 (en) * | 2014-09-16 | 2016-03-24 | Syngenta Participations Ag | Pesticidally active tetracyclic derivatives with sulphur containing substituents |
| WO2016046071A1 (en) * | 2014-09-25 | 2016-03-31 | Syngenta Participations Ag | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
| WO2016058928A1 (en) * | 2014-10-14 | 2016-04-21 | Syngenta Participations Ag | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
| WO2016071214A1 (en) * | 2014-11-07 | 2016-05-12 | Syngenta Participations Ag | Pesticidally active polycyclic derivatives with sulfur containing substituents |
Non-Patent Citations (22)
| Title |
|---|
| "Compendium of Herbicide Adjuvants, 10th ed.", 2010, SOUTHERN ILLINOIS UNIVERSITY |
| "Proc. BCPC", INT. CONGR., vol. 1, 2003, pages 93 |
| A. WOOD, COMPENDIUM OF PESTICIDE COMMON NAMES, 1995 |
| ADV. SYNTH. CATAL., vol. 355, 2013, pages 1741 - 1747 |
| ANGEW. CHEM. INT. ED., vol. 43, 2004, pages 1132 - 1136 |
| ANGEW. CHEM. INT. ED., vol. 50, 2011, pages 3793 |
| ANGEW. CHEM. INT. ED., vol. 51, 2012, pages 536 |
| ANGEW. CHEM. INT. ED., vol. 52, 2013, pages 1548 - 1552 |
| BIO. MED.CHEM., vol. 15, no. 4, 2007, pages 1586 - 1605 |
| C. D. S. TOMLIN: "The Pesticide Manual - A World Compendium, 13th ed", THE BRITISH CROP PROTECTION COUNCIL |
| C. FERRI: "Reaktionen der Organischen Synthese", 1978, GEORG THIEME VERLAG, pages: 223ff |
| EU. J. CHEM., vol. 20, 2014, pages 4098 - 4104 |
| EU. J. MED. CHEM., vol. 22, no. 5, 1987, pages 457 - 62 |
| J. MED. CHEM., vol. 32, no. 12, 1989, pages 2561 - 73 |
| J. MED. CHEM., vol. 56, no. 1, 2013, pages 84 - 96 |
| J. ORG. CHEM., vol. 70, 2005, pages 8601 - 8604 |
| J. ORG. CHEM., vol. 74, 2009, pages 5599 - 5602 |
| J. ORG. CHEM., vol. 78, 2013, pages 12494 - 12504 |
| J.ORGMET. CHEM., vol. 576, 1999, pages 147 - 168 |
| ORG. LETT., vol. 16, 2014, pages 1744 |
| PHOSPHORUS, SULFUR AND SILICON AND THE RELATED ELEMENTS, vol. 188, no. 12, 2013, pages 1835 - 1844 |
| TETRAHEDRON LETTERS, vol. 22, 1981, pages 3815 - 3818 |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2023182586A (en) * | 2018-09-12 | 2023-12-26 | 日本化薬株式会社 | pest control agent |
| JP7682234B2 (en) | 2018-09-12 | 2025-05-23 | 日本化薬株式会社 | Pest control agents |
| CN112824402A (en) * | 2019-11-21 | 2021-05-21 | 浙江省化工研究院有限公司 | Benzimidazole derivative, preparation method and application thereof |
| CN112824402B (en) * | 2019-11-21 | 2023-02-28 | 浙江省化工研究院有限公司 | A class of benzimidazole derivatives, its preparation method and application |
| WO2021204577A1 (en) | 2020-04-06 | 2021-10-14 | Basf Se | Imidazo-pyrimidone compounds as pesticides |
| WO2022128524A1 (en) | 2020-12-14 | 2022-06-23 | Basf Se | Sulfoximine pesticides |
| US12435083B2 (en) | 2021-09-13 | 2025-10-07 | Boehringer Ingelheim Vetmedica Gmbh | Cyclopropyl-(hetero)aryl-substituted ethylsulphonyl-pyridine derivatives |
| WO2023088718A1 (en) | 2021-11-19 | 2023-05-25 | Basf Se | Bicyclic compounds for the control of invertebrate pests |
| WO2024175559A1 (en) | 2023-02-23 | 2024-08-29 | Boehringer Ingelheim Vetmedica Gmbh | Tetrazole-substituted ethyl-sulfone/sulfoximine-pyridine derivatives |
| WO2024189139A1 (en) | 2023-03-14 | 2024-09-19 | Syngenta Crop Protection Ag | Control of pests resistant to insecticides |
| WO2024251913A1 (en) | 2023-06-06 | 2024-12-12 | Syngenta Crop Protection Ag | Synthesis of enantioenriched 2-cyano pyridyl sulfoxides |
| WO2025229111A1 (en) | 2024-05-01 | 2025-11-06 | Basf Se | Pyrimidinone derivatives as pesticidal compounds |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3214939B1 (en) | Pesticidally active polycyclic derivatives with sulfur containing substituents | |
| EP3240788B1 (en) | Pesticidally active tetracyclic derivatives with sulfur containing substituents | |
| EP3166941B1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| EP3268365B1 (en) | Pesticidally active tetracyclic derivatives with sulfur containing substituents | |
| EP3317276B1 (en) | Pesticidally active polycyclic derivatives with sulfur containing substituents | |
| WO2016116338A1 (en) | Pesticidally active polycyclic derivatives with sulfur containing substituents | |
| WO2016026848A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| EP3207044A1 (en) | Pesticidally active tetracyclic heterocyclic derivatives with sulphur containing substituents | |
| EP3177620A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| EP3240786A1 (en) | Pesticidally active polycyclic derivatives with sulfur containing substituents | |
| EP3554242A1 (en) | Pesticidally active heterocyclic derivatives with sulfur containing substituents | |
| WO2017134066A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| EP3585770A1 (en) | Pesticidally active heterocyclic derivatives with sulfur containing substituents | |
| WO2017133994A1 (en) | Pesticidally active derivatives with sulfur and cyclopropyl containing substituents | |
| WO2018206348A1 (en) | Imidazopyrimidine derivatives with sulfur containing phenyl and pyridyl substituents | |
| US10512265B2 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| WO2018095795A1 (en) | Pesticidally active polycyclic derivatives with sulfur containing substituents | |
| EP3245192A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| WO2018099812A1 (en) | Pesticidally active heterocyclic derivatives with sulfur containing substituents | |
| WO2019008115A1 (en) | Pesticidally active heterocyclic derivatives with sulfur containing substituents | |
| WO2016012395A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| WO2019053182A1 (en) | Pesticidally active heterocyclic derivatives with sulfur containing substituents | |
| EP3353160A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents | |
| WO2018215304A1 (en) | Tetracyclic pyridazine sulphur containing compounds and their use as pesticides | |
| WO2018091389A1 (en) | Pesticidally active heterocyclic derivatives with sulphur containing substituents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17803872 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17803872 Country of ref document: EP Kind code of ref document: A1 |