WO2014131191A1 - A composition containing honokiol and/or magnolol and uses thereof - Google Patents
A composition containing honokiol and/or magnolol and uses thereof Download PDFInfo
- Publication number
- WO2014131191A1 WO2014131191A1 PCT/CN2013/072041 CN2013072041W WO2014131191A1 WO 2014131191 A1 WO2014131191 A1 WO 2014131191A1 CN 2013072041 W CN2013072041 W CN 2013072041W WO 2014131191 A1 WO2014131191 A1 WO 2014131191A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- acid
- carboxylic acid
- magnolol
- honokiol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/34—Alcohols
- A61K8/347—Phenols
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/57—Magnoliaceae (Magnolia family)
- A61K36/575—Magnolia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/362—Polycarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/365—Hydroxycarboxylic acids; Ketocarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/368—Carboxylic acids; Salts or anhydrides thereof with carboxyl groups directly bound to carbon atoms of aromatic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/72—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds
- A61K8/81—Cosmetics or similar toiletry preparations characterised by the composition containing organic macromolecular compounds obtained by reactions involving only carbon-to-carbon unsaturated bonds
- A61K8/8141—Compositions of homopolymers or copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by only one carboxyl radical, or of salts, anhydrides, esters, amides, imides or nitriles thereof; Compositions of derivatives of such polymers
- A61K8/8147—Homopolymers or copolymers of acids; Metal or ammonium salts thereof, e.g. crotonic acid, (meth)acrylic acid; Compositions of derivatives of such polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q17/00—Barrier preparations; Preparations brought into direct contact with the skin for affording protection against external influences, e.g. sunlight, X-rays or other harmful rays, corrosive materials, bacteria or insect stings
- A61Q17/005—Antimicrobial preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/10—Washing or bathing preparations
Definitions
- the present invention relates to compositions comprising antimicrobial actives. More specifically, the present invention relates to compositions comprising antimicrobial actives such as honokiol and/or magnolol and carboxylic acids for enhanced deposition of the antimicrobial actives on skin, and methods of the use of the personal care products comprising such composition.
- antimicrobial actives such as honokiol and/or magnolol and carboxylic acids
- Antimicrobial cleansing products have been widely used to clean skin surface in variety of forms. These antimicrobial cleansing products have been formulated with antimicrobial agents in combination with surfactants and aqueous systems to remove microorganisms such as bacteria, viruses, and fungi, during washing and to provide residual effectiveness against microorganisms upon rinsing off the cleansing products.
- antimicrobial cleansing compositions and methods of treatment that that are effective against pathogenic microorganisms using relatively lower amounts of antimicrobial actives.
- antimicrobial cleansing compositions that provide significantly enhanced deposition of such antimicrobial actives.
- the present invention provides antimicrobial cleansing compositions that improve the delivery of antimicrobial actives and provide an enhanced deposition of the antimicrobial actives from personal wash or rinse, resulting in an enhanced skin cleansing and disinfecting benefits.
- compositions that tend to have unexpected and beneficial properties associated therewith, including increased deposition of the antimicrobial actives upon application to a surface (such as skin), wherein the composition is adjusted to a pH from about 3.0 to 5.2.
- compositions comprising an antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and a carboxylic acid having water solubility greater than about 0.05g/100g.
- the present invention provides methods of cleansing skin comprising applying to skin a composition comprising an antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and a carboxylic acid having a water solubility greater than about 0.05g/100g.
- the term "antimicrobial active” refers to the ability of an agent or composition to beneficially control or kill pathogenic, spoilage, or otherwise harmful microorganisms, including, but not limited to bacteria, fungi, viruses, protozoa, yeasts, mold and mildew.
- the term "antimicrobial cleansing composition” refers to composition suitable for application to a surface for the purpose of removing microorganisms from the surface, as well as inhibiting, preventing and eliminating the process of the growth of the microorganism in the surface.
- Preferred embodiments of the present invention are cleansing compositions suitable for use on the mammalian skin.
- rinse-off indicates that the compositions of the present invention are used in a context whereby the composition is ultimately rinsed or washed from the treated surface, (e.g. skin or hard surfaces) either after or during the application of the product.
- These rinse-off compositions are to be distinguished from compositions which are applied to the skin and allowed to remain on the skin subsequent to application.
- leave-on refers generally to the compositions applied to the skin for controlling the growth and viability of the bacteria on the skin, such as lotions, creams, gel-creams, sticks, sprays, pastes, mousses and moisturizer.
- safe and effective amount refers to an amount of an active ingredient high enough to modify the condition to be treated or to deliver the desired skin benefit, but low enough to avoid serious side effects, at a reasonable benefit to risk ratio within the scope of sound medical judgment. What is a safe and effective amount of the active ingredient will vary with the specific active, the ability of the active to penetrate through the skin, the age, health condition, and skin condition of the user, and other like factors.
- pH shall include pH measurements as determined by ASTM method E70 - 07 Standard Test Method for pH of Aqueous Solutions with the Glass Electrode.
- compositions are weight percent of active/solids ingredient based on the total weight of composition.
- Applicants have recognized it is often difficult to formulate an antimicrobial cleansing product that deposits sufficient amount of antimicrobial agents on the skin during its use, especially upon rinsing or washing.
- the low deposition of the antimicrobial agents is caused by the interaction of the antimicrobial agents and the surfactants within the antimicrobial cleansing products.
- the antimicrobial agents are emulsified by the surfactant leaving only a small amount of antimicrobial agents available for deposition upon washing and rinsing.
- antimicrobial agents such as honokiol, magnolol
- carboxylic acids when combined with carboxylic acids can be formulated into cleansing formulations that tend to exhibit increased deposition of the antimicrobial actives upon rinsing or washing under controlled pH conditions.
- compositions including antimicrobial cleansing compositions, can be formulated by using relatively low concentrations of antimicrobial actives, such as honokiol and/or magnolol, which may be naturally derived and/or extracted, in combination with a carboxylic acid having a water solubility greater than about 0.05g/100g, such that the compositions tend to exhibit enhanced skin deposition of the antimicrobial actives.
- antimicrobial actives such as honokiol and/or magnolol, which may be naturally derived and/or extracted
- compositions comprising honokiol and/or magnolol in combination with the carboxylic acid tended to deposit an amount of antimicrobial active that was at least 100% more than the same composition without the carboxylic acid and as high as 200% to 300% or more than compositions in certain embodiments.
- other known antimicrobial phenolic actives tend not to exhibit such significant increase in deposition when combined with the carboxylic acid (e.g. compositions comprising thymol, as shown herein below having an enhanced deposition of less than 100%).
- the compositions of the present invention exhibit an enhanced deposition of the antimicrobial actives of greater than 50%, preferably greater than 100% as compared to the same composition without the carboxylic acid as described and illustrated in the Examples below.
- Honokiol and magnolol are biphenolic compounds in which honokiol is an isomer of magnolol. The chemical structures of honokiol and magnolol are illustrated below:
- honokiol and magnolol used independently or in their combinations, exhibit relatively high antimicrobial efficacy.
- honokiol and/or magnolol are highly effective against gram-positive bacteria such as Staphylococcus aureus, and fungi, including yeasts such as Candida albicans, as evidenced by the low Minimum Inhibitory Concentration (MIC) (i.e. 15ppm).
- MIC Minimum Inhibitory Concentration
- the MIC for a particular antimicrobial agent and a particular microbe is defined as the minimum concentration of that antimicrobial agent that must be present in an otherwise suitable growth medium for that microbe, in order to render the growth medium unsuitable for that microbe, i.e., the minimum concentration to inhibit growth of that microbe.
- Honokiol and magnolol from any suitable source can be used in the present invention.
- honokiol and magnolol can be isolated or purified from plants of the genus Magnolia (e.g., whole plant, flower, bark, seed cone extracts and the like) or other extracts that include such compounds by standard techniques.
- these compounds are commercially available or can be synthesized using conventional chemical synthesis techniques known to those of ordinary skill in the art.
- compositions comprise honokiol and/or magnolol which are naturally occurring and extracted from plants of the genus
- Magnolia In general, Magnolia is a large genus of about 210 flowering plant species in the subfamily Magnoliodieae of the family Magnoliaceae.
- Magnolia extract can be obtained from the species within the Magnoliaceae family. Non-limiting examples of these species include Magnolia acuminata, Magnolia ashei, Magnolia biondii, Magnolia cylindrica, Magnolia cambellii, Magnolia denudata.
- Active ingredients that have been identified in and isolated from Magnolia flower, bark, and seed cone extracts include magnolol,
- the present invention comprises honokiol and/or magnolol that is extracted from Magnolia officinalis, preferably from the bark thereof.
- Magnolia extracts containing magnolol and/or honokiol are commercially available from a variety of different sources.
- an extract of the bark of Magnolia officinalis which comprises both honokiol and magnolol, each in a concentration of about 45% (total concentration of honokiol and magnolol combined in the extract is 90%) is available from H- NSEA under the tradename "MagnoPro®.”
- Individual magnolol and honokiol compounds - isolated from the magnolia bark are available from H-NSEA under the tradenames "Magnolol 95% and "Honokiol 95% ", respectively.
- a person of ordinary skill in the art would be able to isolate Magnolia extract from the Magnolia flower, bark, or seed cone by using any suitable isolation and purification methods known in the art.
- Conventional methods include, but not limited to, direct extraction of material from the wood by grinding, macerating, pressing, squeezing, mashing, centrifuging, and/or processes such as cold percolation, agitation/distillation, microwave assisted extraction, sonication, supercritical/subcritical C0 2 compressed gas extraction with or without polar modifiers, pressurized solvent extraction, accelerated solvent extraction, pressurized or normal hot water extraction, surfactant assisted pressurized hot water extraction, oil extraction, membrane extraction, Soxhlet extraction, the gold finger distillation/extraction and/or processes disclosed, for example, in US Pat. Nos. 7442391 , 7473435, and 7537791 to Integrated Botanical Technologies, LLC, incorporated herein by reference, and the like, or by other methods such as solvent extraction, and the like.
- the composition of the present invention comprise from about 0.004% to about 1.25 % by weight of total honokiol or magnolol (or both if both are present) in the composition. In certain more preferred embodiments the compositions comprise from about 0.005% to about 1.0%, and more preferably from 0.01 wt % to about 0.75 wt % by weight honokiol and/or magnolol.
- the composition of the present invention comprise from about 0.001 % to about 5.0%, more preferably from 0.01% to about 2.5% and more preferably from about 0.02 % to about 2.0% carboxylic acid.
- Suitable carboxylic acids can be naturally derived or synthetically produced, having one or more carboxylic (COOH) groups.
- the pH of the composition should be below the pKa of the weaker carboxylic acid in the composition plus 1 (i.e. pH ⁇ p a+l).
- pKa value for a compound is the pH value at which the carboxylic acid is 50 percent dissociated or ionized to yield its conjugate base and a proton (or hydrated proton).
- the pH of the antimicrobial cleansing compositions is adjusted and preferably buffered to range from about 3.0 to about 5.2. In certain preferred embodiments, the pH is from about 3.5 to about 4.5.
- the solubility in water of the carboxylic acids of the present invention is greater than about 0.05g/100g. More preferably, the solubility in water of the carboxylic acids of the present invention is greater than about O. lg/lOOg. Most preferably, the solubility in water of the carboxylic acids of the present invention is greater than about 0.2g/100g.
- the carboxylic acid may be an aromatic acid.
- suitable aromatic carboxylic acids include, but not limited to benzoic acid, salicylic acid, O-phthalic acid, Quinoline-2-carboxylic acid, ferulic acid, and niacin, derivatives thereof, and combinations of two or more thereof, and the like.
- the aromatic carboxylic acid is selected from the group consisting of benzoic acid, salicylic acid, and combinations thereof.
- the aromatic carboxylic acid is benzoic acid.
- the carboxylic acid may be an aliphatic acid.
- suitable aliphatic carboxylic acids include, but not limited to citric acid, malic acid, succinic acid, poly acrylic acid, derivatives thereof, and combinations of two or more thereof, and the like.
- compositions of this invention may be formulated into a wide variety of product types that include but are not limited to liquid cleansers, creamy cleansers, gel cleansers, soaps and makeup removers, gels, sticks, sprays, solid bars, shampoos, pastes, foams, powders, mousses, shaving creams, wipes, patches, nail lacquers, wound dressing and adhesive bandages, hydrogels, films and make-up such as foundations, mascaras, and lipsticks.
- product types may comprise several types of carriers including, but not limited to solutions, emulsions (e.g., microemulsions and nanoemulsions), gels, and solids.
- Other carriers include solvents, which can include, but are not limited to water, acetone, alcohols, such as isopropanol and ethanol, ethylene glycol, glycerin, dimethylformamide, tetrahydrofuran, dimethylsulfoxide, sorbitol and ethers and ester of sorbitol.
- solvents can include, but are not limited to water, acetone, alcohols, such as isopropanol and ethanol, ethylene glycol, glycerin, dimethylformamide, tetrahydrofuran, dimethylsulfoxide, sorbitol and ethers and ester of sorbitol.
- solvents can include, but are not limited to water, acetone, alcohols, such as isopropanol and ethanol, ethylene glycol, glycerin, dimethylformamide, tetrahydrofuran, dimethylsulfoxide, sorbitol and ethers and ester of sorbitol
- compositions useful in the present invention may include formulations suitable for administering to the target tissues, such as human skin.
- the composition further comprises a carrier, preferably a cosmetically/dermatologicaily acceptable carrier.
- a cosmetically/dermatologicaily acceptable carrier means a carrier that is suitable for use in contact with the skin without undue toxicity, incompatibility, instability, irritation, allergic response, and the like.
- the compositions can be formulated as solutions. Solutions typically include an aqueous or organic solvent (e.g., from about 50% to about 99.99% or from about 90% to about 99% of a cosmetically acceptable aqueous or organic solvent).
- compositions of the present invention are aqueous solutions comprising from about 50% to about 99% by weight of water.
- compositions of the present invention may be formulated as a solution comprising a surfactant.
- the surfactant can be any of the anionic surfactants, nonionic surfactants, amphoteric surfactants, zwitterionic surfactants cationic surfactants and mixtures clearly as are well known in the art.
- anionic surfactants include, but are certainly not limited to aliphatic sulphate, aliphatic sulfonate (e.g., Cs to C 22 sulfonate or disu'fonate), aromatic sulfonate (e.g., alkyl benzene sulfonate), alkyl sulfoccinates, alkyl and acyl taurates, alkyl and acyl sarcosinates, sulfoacetates, alkyl phosphates, carboxylates, isethionates, etc.
- aliphatic sulphate e.g., Cs to C 22 sulfonate or disu'fonate
- aromatic sulfonate e.g., alkyl benzene sulfonate
- alkyl sulfoccinates alkyl and acyl taurates
- alkyl and acyl sarcosinates alky
- zwitterionic surfactants are exemplified by those which can be broadly described as derivatives of aliphatic quaternary ammonium, phosphonium, and sulfonium compounds, in which the aliphatic radicals can be straight or branched chain, and wherein one of the aliphatic substituents contains from about 8 to about 18 carbon atoms and one contains an anionic group, e.g., carboxy, sulfonate, sulfate, phosphate, or phosphonate.
- amphoteric detergents which may be used in this invention include at least one acid group. This may be a carboxylic or a sulphonic acid group. They include quaternary nitrogen and therefore are quaternary amido acids. They should generally include an alkyl or alkenyl group of 7 to 18 carbon atoms.
- nonionic which may be used includes in particular the reaction products of compounds having a hydrophobic group and a reactive hydrogen atom, for example aliphatic alcohols, acids, amides or alkyl phenols with alkylene oxides, especially ethylene oxide either alone or with propylene oxide.
- Specific nonionic detergent compounds are alkyl (C 6 -C 2 2) phenols-ethylene oxide condensates, the condensation products of aliphatic (Ce-C ) primary or secondary linear or branched alcohols with ethylene oxide, and products made by condensation of ethylene oxide with the reaction products of propylene oxide and ethylenediamine.
- Other so-called nonionic detergent compounds include long chain tertiary amine oxides, long chain tertiary phosphine oxides and dialkyl sulphoxides.
- compositions useful in the present invention may be formulated as a solution comprising an emollient.
- Such compositions preferably contain from about 2% to about 50% of an emollient(s).
- emollients refer to materials used for the prevention or relief of dryness, as well as for the protection of the skin.
- suitable emollients are known and may be used herein. Sagarin, Cosmetics, Science and Technology, 2nd Edition, Vol. 1, pp. 32 43 (1972) and the International Cosmetic Ingredient Dictionary and Handbook, eds. Wenninger and McEwen, pp.
- a lotion can be made from such a solution.
- Lotions typically comprise from about 1% to about 20% (e.g., from about 5% to about 10%) of an emollient(s) and from about 50% to about 90% (e.g., from about 60% to about 80%) of water.
- compositions of the present invention may be of varying phase compositions, but are preferably aqueous solutions or otherwise include an exterior aqueous phase (e.g., aqueous phase is the most exterior phase of the composition).
- compositions of the present invention may be formulated to be oil-in-water emulsions that are shelf-stable in that the emulsion does not lose phase stability or "break" when kept at standard conditions (22 degrees Celsius, 50% relative humidity) for a week or more after it is made.
- the composition of the present invention may comprise an additional benefit agent.
- benefit agent refers to an element, an ion, a compound, or other chemical moiety that provides a cosmetic or therapeutic effect on the skin.
- benefit agents include small molecules, peptides, proteins, nucleic acid materials, minerals and plant extracts.
- the benefit agents useful herein may be categorized by their therapeutic benefit or their postulated mode of action. However, it is to be understood that the benefit agents useful herein may, in some circumstances, provide more than one therapeutic benefit or operate via greater than one mode of action. Therefore, the particular classifications provided herein are made for the sake of convenience and are not intended to limit the benefit agents to the particular application(s) listed.
- suitable benefit agents include those that provide benefits to the skin, such as, but not limited to, depigmentation agents; reflectants; amino acids and their derivatives; antimicrobial agents; allergy inhibitors; anti-aging agents; anti-wrinkling agents, antiseptics; analgesics; shine-control agents; antipruritics; local anesthetics; anti-hair loss agents; hair growth promoting agents; hair growth inhibitor agents, antihistamines; antiinfectives; antiinflammatory agents; anticholinergics; vasoconstrictors; vasodilators; wound healing promoters; peptides, polypeptides and proteins; deodorants and anti-perspirants; medicament agents; skin firming agents, vitamins; skin lightening agents; skin darkening agents;
- antifungals depilating agents; counterirritants; hemorrhoidals; insecticides; enzymes for exfoliation or other functional benefits; enzyme inhibitors; poison ivy products; poison oak products; burn products; anti-diaper rash agents; prickly heat agents; vitamins; herbal extracts; vitamin A and its derivatives; flavonoids; sensates; anti-oxidants; hair lighteners; sunscreens; anti-edema agents, neo-collagen enhancers, film-forming polymers, chelating agents; anti- dandruff/sebhorreic dermatitis/psoriasis agents; keratolyses; and mixtures thereof.
- the compositions produced via the present invention are preferably used as or in personal care products for treating or cleansing at least a portion of a human body.
- personal care products include various products suitable for application to the skin, hair, oral and/or perineal region of the body, such as shampoos, hand, face, and/or body washes, bath additives, gels, lotions, creams, and the like.
- shampoos, hand, face, and/or body washes, bath additives, gels, lotions, creams, and the like As discussed above, applicants have discovered unexpectedly that the instant methods provide personal care products having reduced irritation to the skin and/or eyes and, in certain embodiments one or more of desirable properties such as flash foaming characteristics, rheology, and functionality, even at high surfactant concentrations.
- Such products may further include a substrate onto which a composition is applied for use on the body. Examples of suitable substrates include a wipe, pouf, sponge, and the like as well as absorbent articles, such as a bandage, sanitary napkin, t
- compositions produced via the present invention are preferably a rinse-off product in the form of a gel or a liquid composition that can be applied in liquid or gel form on the skin and is rinsed-off upon washing yet provides antimicrobial effect on the skin.
- the present invention provides methods of treating and/or cleansing the human body comprising contacting at least a portion of the body with a composition of the present invention.
- Certain preferred methods comprising contacting mammalian skin, hair and/or vaginal region with a composition of the present invention to cleanse such region and/or treat such region for any of a variety of conditions including, but not limited to wrinkles, dermatitis, dryness, muscle pain, itch, and the like. Any of a variety of actives or benefit agents known in the art for treating such conditions may be used in the present invention.
- the cleansing methods of the present invention may further comprise any of a variety of additional, optional steps associated conventionally with cleansing hair and skin including, for example, lathering, rinsing steps, and the like.
- Example 1 Minimum Inhibitory Concentration (MIC) Measurement for Magnolia Bark PE.
- the 96-well plate micro-dilution broth method as described below was used to measure the Minimum Inhibitory Concentration (MIC) value of Magnolia Bark PE (90% magnolol + honokiol) against Staphylococcus aureus ATCC 6538.
- 1.1 96 Well Plate(s) were prepared with an antimicrobial stock solution (e.g. Magnolia Bark PE) at a starting concentration that is at least 10 folds of the concentration needed to be inhibitory against the target bacterium or panel of bacteria to be inoculated (e.g. Staphylococcus aureus ATCC 6538.)
- an antimicrobial stock solution e.g. Magnolia Bark PE
- a starting concentration that is at least 10 folds of the concentration needed to be inhibitory against the target bacterium or panel of bacteria to be inoculated (e.g. Staphylococcus aureus ATCC 6538.)
- At least three to five well-isolated colonies of the same morphological type are selected from an agar plate culture. The top of each colony is touched with a wire loop and the growth was transferred to a tube containing 4 to 5 mL of a suitable broth medium, such as tryptic soy broth.
- a suitable broth medium such as tryptic soy broth.
- the broth culture is incubated at 35 °C until it reaches or exceeds the turbidity of the 0.5 McFarland standards (usually 2 to 6 hours). 2.1.3 The turbidity of the actively growing broth culture is adjusted with sterile broth to obtain turbidity comparable to that of a 0.5 McFarland standard. This results in a suspension containing approximately 1 to 2 x 10 8 CFU/mL (for most commonly used bacteria).
- the adjusted inoculum suspension is diluted in the broth so that, after inoculation, each well contained approximately 5 x 10 4 CFU/mL.
- the exact inoculum volume delivered to the wells must be known to make this calculation. For example, since the volume of medium in the well was 0.1 mL and the inoculum volume is 0.005 mL, then the 0.5 McFarland suspension (1 x 10 8 CFU/mL) is diluted 1 : 10 to yield 10 7 CFU/mL. When 0.005 mL of this suspension is inoculated into the broth, the final test concentration of bacteria would be approximately 5 x 10 4 CFU/well in the micro-dilution method.
- each tray is sealed in a plastic bag, or with plastic tape, or with a tight-fitting plastic cover before it is incubated to minimize evaporation.
- ation for the Micro-dilution Method The inoculated micro-dilution trays are incubated at 35 °C for 16 to 20 hours in an ambient air incubator. To maintain the same incubation temperature for all cultures, no more than four micro-dilution trays are stacked at the same time.
- the MIC is the lowest concentration of antimicrobial agent that completely inhibits growth of the organism in the micro-dilution wells as detected by the unaided eye. Viewing devices intended to facilitate reading micro-dilution tests and recording of results is used as long as the device has been validated for this application.
- the amount of growth in the wells containing the antimicrobial are compared with the amount of growth in the growth-control wells or tubes (no antimicrobial) used in each set of tests when determining the growth end points. For a test to be considered valid, acceptable growth (definite turbidity) must occur in the positive control well.
- the Minimum Inhibitory Concentration (MIC) of Magnolia Bark PE is measured to be 60ppm against Staphylococcus aureus, as determined by the 96-well plate micro-dilution broth method as described above.
- test samples were made in accord with the procedure described below.
- Step I In a manufacturing vessel of 1 kg size, mix deionized water and sodium laureth sulfate (3EO) together at a temperature in the range from about 40 to 60°C until dissolved. Add cocoamidopropyl betaine to the above aqueous solution under constant stirring to obtain a fully uniform mixture. Add sodium lauroamphoacetate and citric acid until uniform mixture formed. Maintained the vessel at 60°C.
- Step II Cool the mixture from Step I to room temperature under constant stirring. Add antimicrobial active (Magnolia extract with combined honokiol and magnolol, magnolol, or honokiol) and Benzoic acid (when present) with continuous stirring until fully dissolved and uniform.
- Step III Adjust viscosity with sodium chloride, and adjust pH with citric acid or hydrochloric acid to below 4.5. Add remaining water and/or sodium chloride to complete the sample preparation.
- Example 3 Enhanced Deposition on Skin:
- test subjects Female subjects (20-35 year old, with non-dry and non-sensitive skin type) were selected and screened by anhydrous alcohol scrubbed on forearm; those showing skin redness were excluded. Once selected, the test subjects used no leave on product two days prior to the test day
- the concentration detected by HPLC is converted into total amount collected in ⁇ g by using the concentration multiply volume and then the deposition per cm 2 was obtained by the total amount collected divide test area.
- the % of enhanced deposition is calculated by Mean of extract base + acid) divided by Mean of extract base.
- enhanced deposition is defined as the average increase of residue collected from the phenol extract/ carboxylic acid complex compared to the residue collected from the phenol extract.
- Mean is defined as the average residue collected from the subjects and STD is standard deviation calculated from the group of data.
- the P value is associated with a test statistic. It is "the probability, if the test statistic really were distributed as it would be under the null hypothesis, of observing a test statistic [as extreme as, or more extreme than] the one actually observed.” The smaller the P value, the more strongly the test rejects the null hypothesis, that is, the hypothesis being tested. A p-value of 0 .05 or less rejects the null hypothesis "at the 5% level” that is, the statistical assumptions used imply that only 5% of the time would the supposed statistical process produce a finding this extreme if the null hypothesis were true. 5% and 10% are common significance levels to which p-values are compared. This is further described in John A. Rice (2006), Mathematical Statistics and Data Analysis, Third Edition, Duxbury Advanced.
- Salicylic Acid Hebei Jingye Salicylic Acid
- HPLC analysis was conducted as described above on either the total Magnolia Bark Extract component (Examples A and A') or the individual component Honokiol and Magnolol, respectively.
- the composition prepared in accordance with this ( ⁇ - ⁇ ') yielded a significant enhancement with respect to the amount of antimicrobial agent, either combined (Magnolia Bark Extract) or individually (Honokiol and Magnolol), deposited on the skin surface.
- the HPLC spectra included peaks that corresponded to the Honokiol enhancement and peaks that corresponded to the Magnolol enhancement.
- the enhancement due to Honokiol and enhancement due to Magnolol can be separately determined.
- Table 4 shows the combined enhancement of Honokiol and Magnolol
- Table 4 B shows the data corresponding to the separate enhancement due to Honokiol and Magnolol by spectral analysis
- Table 4 and Table 4B were derived from the same experimental Examples.
- the HPLC spectra included peaks that corresponded to the Honokiol enhancement and peaks that corresponded to the Magnolol enhancement.
- Table 5 shows the combined enhancement of Honokiol and Magnolol while Table 5 B shows the data corresponding to the separate enhancement due to Honokiol and Magnolol by spectral analysis.
- Table 5 and Table 5B were derived from the same experimental Examples.
- Example 5 As shown in Table 6 and Table 7, the composition prepared in accordance with these examples (C through D) yielded a significant enhancement with respect to the amount of antimicrobial agent, Honokiol and Magnolol deposited on the skin surface.
- Example 5 As shown in Table 6 and Table 7, the composition prepared in accordance with these examples (C through D) yielded a significant enhancement with respect to the amount of antimicrobial agent, Honokiol and Magnolol deposited on the skin surface.
- Example 5 Example 5
- Table 8A Deposition Enhancement of 0.5% of Thymol by 1.5% of Salicylic Acid, pH Adjust to 3.9 - 4.2.
- Table 8B Deposition Enhancement of 0.5% of Thymol by 1.5% of Benzoic Acid, pH Adjust to 3.9 - 4.2.
- composition prepared in accordance with the examples (E through E") yielded a lower degree of enhancement with respect to the amount of antimicrobial agent (thymol) deposited on the skin surface compared to Examples A through D.
- Example 7 Enhanced Deposition on Dead Swine Skin:
- the dead swine skin model has shown to be comparable to human skin and is considered to be a substitute for the human skin test.
- Example F Magnolia extract base + Magnolia extract base
- Magnolia extract base 1.0% 4- + 1.0% p-phthalic acid methoxybenzoic acid
- Table 14 Deposition Enhancement of 0.5% of Magnolia extract by 1.25% of Quinoline 2-carboxyIic acid on dead swine skin, pH-3.9-4.3
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Birds (AREA)
- Emergency Medicine (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Biotechnology (AREA)
- Dermatology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Botany (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Alternative & Traditional Medicine (AREA)
- Medical Informatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Cosmetics (AREA)
Abstract
Provided are compositions comprising an amtimicrobial active comprising honokiol and/or magnolol and a carboxylic acid. Also provided are methods of use thereof.
Description
A COMPOSITION CONTAINING HONO IOL AND/OR MAGNOLOL AND USES
THEREOF
FIELD OF INVENTION
The present invention relates to compositions comprising antimicrobial actives. More specifically, the present invention relates to compositions comprising antimicrobial actives such as honokiol and/or magnolol and carboxylic acids for enhanced deposition of the antimicrobial actives on skin, and methods of the use of the personal care products comprising such composition.
DESCRIPTION OF RELATED ART
Antimicrobial cleansing products have been widely used to clean skin surface in variety of forms. These antimicrobial cleansing products have been formulated with antimicrobial agents in combination with surfactants and aqueous systems to remove microorganisms such as bacteria, viruses, and fungi, during washing and to provide residual effectiveness against microorganisms upon rinsing off the cleansing products.
While antimicrobials are generally effective against various pathogenic
microorganisms, certain strains of pathogenic microorganisms have become increasingly resistant to one or more antimicrobials, thereby rendering the standard courses of treatment ineffective. Accordingly, higher doses of antimicrobial treatments are required to achieve efficacy, which can result in undesirable side effects and toxicity, both biologically (in vivo) and environmentally.
Therefore, it would be desirable to have antimicrobial cleansing compositions and methods of treatment that that are effective against pathogenic microorganisms using relatively lower amounts of antimicrobial actives. In addition, it would be desirable to have antimicrobial cleansing compositions that provide significantly enhanced deposition of such antimicrobial actives.
The present invention provides antimicrobial cleansing compositions that improve the delivery of antimicrobial actives and provide an enhanced deposition of the antimicrobial actives from personal wash or rinse, resulting in an enhanced skin cleansing and disinfecting benefits.
SUMMARY OF THE INVENTION
Applicants have discovered that combinations of antimicrobial actives selected from the group consisting of honokiol, magnolol, and combinations thereof can be combined with carboxylic acids to make compositions that tend to have unexpected and beneficial properties associated therewith, including increased deposition of the antimicrobial actives upon application to a surface (such as skin), wherein the composition is adjusted to a pH from about 3.0 to 5.2.
Accordingly, one aspect the present invention provides compositions comprising an antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and a carboxylic acid having water solubility greater than about 0.05g/100g.
According to another aspect, the present invention provides methods of cleansing skin comprising applying to skin a composition comprising an antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and a carboxylic acid having a water solubility greater than about 0.05g/100g.
DESCRIPTION OF THE INVENTION
As used herein, the term "antimicrobial active" refers to the ability of an agent or composition to beneficially control or kill pathogenic, spoilage, or otherwise harmful microorganisms, including, but not limited to bacteria, fungi, viruses, protozoa, yeasts, mold and mildew.
As used herein, the term "antimicrobial cleansing composition" refers to composition suitable for application to a surface for the purpose of removing microorganisms from the surface, as well as inhibiting, preventing and eliminating the process of the growth of the microorganism in the surface. Preferred embodiments of the present invention are cleansing compositions suitable for use on the mammalian skin.
As used herein, the term "rinse-off' as used herein indicates that the compositions of the present invention are used in a context whereby the composition is ultimately rinsed or washed from the treated surface, (e.g. skin or hard surfaces) either after or during the application of the product. These rinse-off compositions are to be distinguished from compositions which are applied to the skin and allowed to remain on the skin subsequent to application.
As used herein, the term "leave-on" refers generally to the compositions applied to the skin for controlling the growth and viability of the bacteria on the skin, such as lotions, creams, gel-creams, sticks, sprays, pastes, mousses and moisturizer.
As used herein, the term "safe and effective amount" refers to an amount of an active ingredient high enough to modify the condition to be treated or to deliver the desired skin benefit, but low enough to avoid serious side effects, at a reasonable benefit to risk ratio within the scope of sound medical judgment. What is a safe and effective amount of the active ingredient will vary with the specific active, the ability of the active to penetrate through the skin, the age, health condition, and skin condition of the user, and other like factors.
As used herein, the term "pH" shall include pH measurements as determined by ASTM method E70 - 07 Standard Test Method for pH of Aqueous Solutions with the Glass Electrode.
As used herein, unless otherwise specified, all percentages of ingredients in
compositions are weight percent of active/solids ingredient based on the total weight of composition.
Applicants have recognized it is often difficult to formulate an antimicrobial cleansing product that deposits sufficient amount of antimicrobial agents on the skin during its use, especially upon rinsing or washing. The low deposition of the antimicrobial agents is caused by the interaction of the antimicrobial agents and the surfactants within the antimicrobial cleansing products. Generally, the antimicrobial agents are emulsified by the surfactant leaving only a small amount of antimicrobial agents available for deposition upon washing and rinsing.
Applicants have discovered that antimicrobial agents such as honokiol, magnolol, when combined with carboxylic acids can be formulated into cleansing formulations that tend to exhibit increased deposition of the antimicrobial actives upon rinsing or washing under controlled pH conditions.
Applicants have further discovered that compositions, including antimicrobial cleansing compositions, can be formulated by using relatively low concentrations of antimicrobial actives, such as honokiol and/or magnolol, which may be naturally derived and/or extracted, in combination with a carboxylic acid having a water solubility greater than about 0.05g/100g, such that the compositions tend to exhibit enhanced skin deposition of the antimicrobial actives. For example, as shown in the Examples herein, applicants have discovered that compositions comprising honokiol and/or magnolol in combination with the carboxylic acid tended to deposit an amount of antimicrobial active that was at least 100% more than the same composition without the carboxylic acid and as high as 200% to 300% or more than compositions in certain embodiments. In addition, applicants have discovered that other known antimicrobial phenolic actives tend not to exhibit such significant increase in deposition when combined with the carboxylic acid (e.g. compositions comprising thymol, as shown herein below having an enhanced deposition of less than 100%). Accordingly, in certain embodiments, the compositions of the present invention exhibit an enhanced deposition of the antimicrobial actives of greater than 50%, preferably greater than 100% as compared to the same composition without the carboxylic acid as described and illustrated in the Examples below.
Honokiol and magnolol are biphenolic compounds in which honokiol is an isomer of magnolol. The chemical structures of honokiol and magnolol are illustrated below:
Applicants have recognized that honokiol and magnolol, used independently or in their combinations, exhibit relatively high antimicrobial efficacy. For instance, honokiol and/or magnolol are highly effective against gram-positive bacteria such as Staphylococcus aureus, and fungi, including yeasts such as Candida albicans, as evidenced by the low Minimum Inhibitory Concentration (MIC) (i.e. 15ppm). The MIC for a particular antimicrobial agent and a particular microbe is defined as the minimum concentration of that antimicrobial agent that must be present in an otherwise suitable growth medium for that microbe, in order to render the growth medium unsuitable for that microbe, i.e., the minimum concentration to inhibit growth of that microbe.
Honokiol and magnolol from any suitable source can be used in the present invention. For example, honokiol and magnolol can be isolated or purified from plants of the genus Magnolia (e.g., whole plant, flower, bark, seed cone extracts and the like) or other extracts that include such compounds by standard techniques. Alternatively, these compounds are
commercially available or can be synthesized using conventional chemical synthesis techniques known to those of ordinary skill in the art.
In certain preferred embodiments of the invention, the compositions comprise honokiol and/or magnolol which are naturally occurring and extracted from plants of the genus
Magnolia. In general, Magnolia is a large genus of about 210 flowering plant species in the subfamily Magnoliodieae of the family Magnoliaceae. Magnolia extract can be obtained from the species within the Magnoliaceae family. Non-limiting examples of these species include Magnolia acuminata, Magnolia ashei, Magnolia biondii, Magnolia cylindrica, Magnolia cambellii, Magnolia denudata. Magnolia fraseri, Magnolia grandiflora, Magnolia hypoleuca, Magnolia kobus, Magnolia hliiflora, Magnolia loegneri, Magnolia macrophylla, Magnolia officinalis, Magnolia pyramidata, Magnolia sargentiana. Magnolia seiboldii, Magnolia soulangiana, Magnolia sprengeri, Magnolia stellata. Magnolia tripetala, Magnolia virginiana, Magnolia zenii, and Michelia figo. Active ingredients that have been identified in and isolated from Magnolia flower, bark, and seed cone extracts include magnolol,
dihydroxydihdromagnolol, honokiol, and dihydrohonokiol, Isomagnolol, 4,4 -Diallyl-3,2 ,6 - trimethoxybiphenyl ether, 6-O-methylhonokiol, 4,4 -Diallyl-2,3 -dihydroxybiphenyl ether, Bornylmagnolol. In certain preferred embodiments, the present invention comprises honokiol and/or magnolol that is extracted from Magnolia officinalis, preferably from the bark thereof.
Magnolia extracts containing magnolol and/or honokiol are commercially available from a variety of different sources. For example, an extract of the bark of Magnolia officinalis which comprises both honokiol and magnolol, each in a concentration of about 45% (total concentration of honokiol and magnolol combined in the extract is 90%), is available from H- NSEA under the tradename "MagnoPro®." Individual magnolol and honokiol compounds - isolated from the magnolia bark are available from H-NSEA under the tradenames "Magnolol 95% and "Honokiol 95% ", respectively.
Alternatively, a person of ordinary skill in the art would be able to isolate Magnolia extract from the Magnolia flower, bark, or seed cone by using any suitable isolation and purification methods known in the art. Conventional methods include, but not limited to, direct
extraction of material from the wood by grinding, macerating, pressing, squeezing, mashing, centrifuging, and/or processes such as cold percolation, agitation/distillation, microwave assisted extraction, sonication, supercritical/subcritical C02 compressed gas extraction with or without polar modifiers, pressurized solvent extraction, accelerated solvent extraction, pressurized or normal hot water extraction, surfactant assisted pressurized hot water extraction, oil extraction, membrane extraction, Soxhlet extraction, the gold finger distillation/extraction and/or processes disclosed, for example, in US Pat. Nos. 7442391 , 7473435, and 7537791 to Integrated Botanical Technologies, LLC, incorporated herein by reference, and the like, or by other methods such as solvent extraction, and the like.
According to certain embodiments, the composition of the present invention comprise from about 0.004% to about 1.25 % by weight of total honokiol or magnolol (or both if both are present) in the composition. In certain more preferred embodiments the compositions comprise from about 0.005% to about 1.0%, and more preferably from 0.01 wt % to about 0.75 wt % by weight honokiol and/or magnolol.
According to certain embodiments, the composition of the present invention comprise from about 0.001 % to about 5.0%, more preferably from 0.01% to about 2.5% and more preferably from about 0.02 % to about 2.0% carboxylic acid. Suitable carboxylic acids can be naturally derived or synthetically produced, having one or more carboxylic (COOH) groups.
According to certain embodiments, the pH of the composition should be below the pKa of the weaker carboxylic acid in the composition plus 1 (i.e. pH<p a+l). By standard definitions, pKa value for a compound is the pH value at which the carboxylic acid is 50 percent dissociated or ionized to yield its conjugate base and a proton (or hydrated proton). Without being limited by theory, when the pH of the formulation is below the pKa +1 of the carboxylic acid it is believed that the carboxylic acid will exist more than 9% in its un- ionized/protonated form, which will facilitate the acid form complexes with the biphenolic compounds such as honokiol or magnolol in the magnolia extract and enhance the biphenolic compounds deposited on the skin. Further, a low pH is preferred to suppress ionization and enhance the penetration of the acid into the stratum corneum. Accordingly, in certain
embodiments of the present invention, the pH of the antimicrobial cleansing compositions is adjusted and preferably buffered to range from about 3.0 to about 5.2. In certain preferred embodiments, the pH is from about 3.5 to about 4.5.
Preferably, the solubility in water of the carboxylic acids of the present invention is greater than about 0.05g/100g. More preferably, the solubility in water of the carboxylic acids of the present invention is greater than about O. lg/lOOg. Most preferably, the solubility in water of the carboxylic acids of the present invention is greater than about 0.2g/100g.
The carboxylic acid may be an aromatic acid. Examples of suitable aromatic carboxylic acids include, but not limited to benzoic acid, salicylic acid, O-phthalic acid, Quinoline-2-carboxylic acid, ferulic acid, and niacin, derivatives thereof, and combinations of two or more thereof, and the like. In certain preferred embodiments, the aromatic carboxylic acid is selected from the group consisting of benzoic acid, salicylic acid, and combinations thereof. In certain more preferred embodiments, the aromatic carboxylic acid is benzoic acid.
Alternatively, the carboxylic acid may be an aliphatic acid. Examples of suitable aliphatic carboxylic acids include, but not limited to citric acid, malic acid, succinic acid, poly acrylic acid, derivatives thereof, and combinations of two or more thereof, and the like.
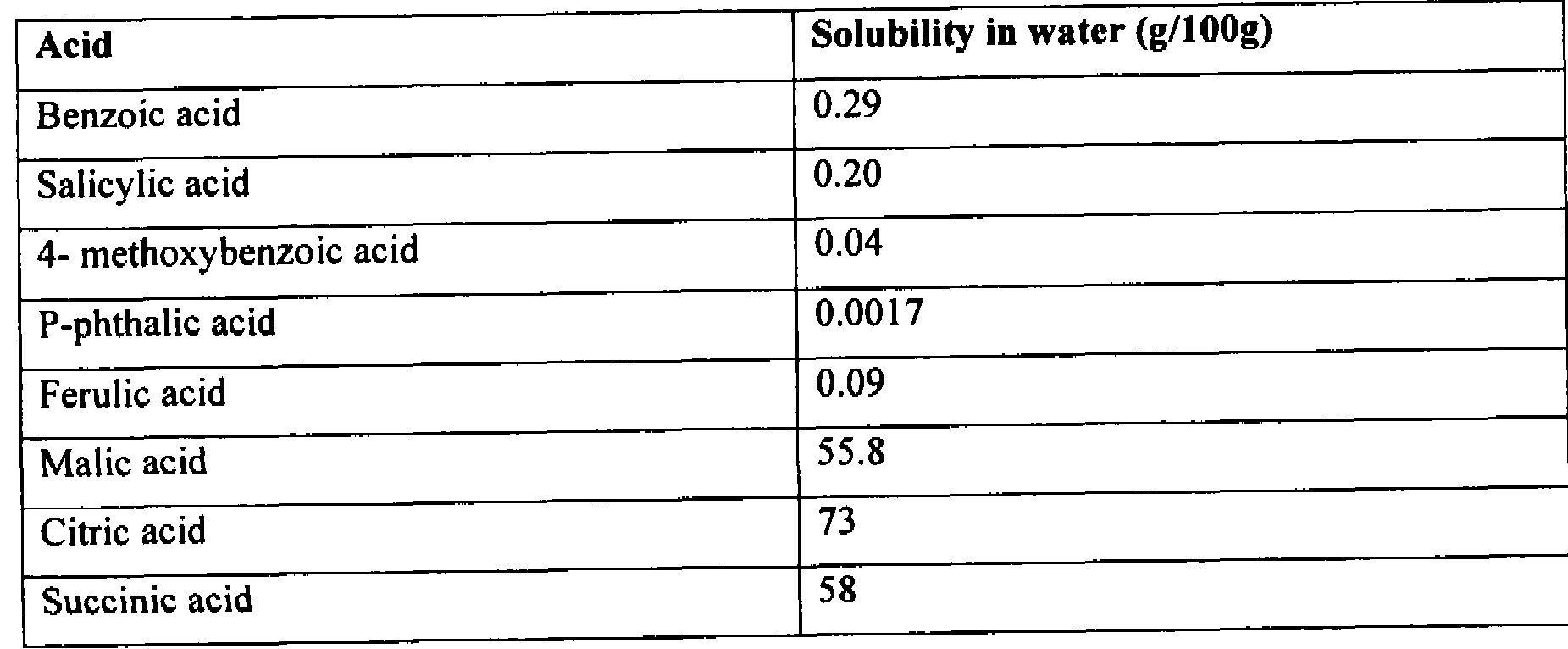
Table 1 Solubility of Various Acids at Ambient Temperature
O-Phthalic acid 0.6
Quinoline-2-carboxylic acid 1.4
Niacin 1.8
The compositions of this invention may be formulated into a wide variety of product types that include but are not limited to liquid cleansers, creamy cleansers, gel cleansers, soaps and makeup removers, gels, sticks, sprays, solid bars, shampoos, pastes, foams, powders, mousses, shaving creams, wipes, patches, nail lacquers, wound dressing and adhesive bandages, hydrogels, films and make-up such as foundations, mascaras, and lipsticks. These product types may comprise several types of carriers including, but not limited to solutions, emulsions (e.g., microemulsions and nanoemulsions), gels, and solids. Other carriers include solvents, which can include, but are not limited to water, acetone, alcohols, such as isopropanol and ethanol, ethylene glycol, glycerin, dimethylformamide, tetrahydrofuran, dimethylsulfoxide, sorbitol and ethers and ester of sorbitol. In an embodiment of the invention, water and alcohols are the preferred carriers. Other carriers can be formulated by those of ordinary skill in the art.
The compositions useful in the present invention may include formulations suitable for administering to the target tissues, such as human skin. In one embodiment, the composition further comprises a carrier, preferably a cosmetically/dermatologicaily acceptable carrier. As used herein, the term "cosmetically/dermatologicaily acceptable carrier" means a carrier that is suitable for use in contact with the skin without undue toxicity, incompatibility, instability, irritation, allergic response, and the like. The compositions can be formulated as solutions. Solutions typically include an aqueous or organic solvent (e.g., from about 50% to about 99.99% or from about 90% to about 99% of a cosmetically acceptable aqueous or organic solvent). Examples of suitable organic solvents include: polyglycerols, propylene glycol, polyethylene glycol (200, 600), polypropylene glycol (425, 2025), glycerol, 1,2,4-butanetriol, sorbitol esters, 1,2,6-hexanetrioI, ethanol, and mixtures thereof. In certain preferred
embodiments, the compositions of the present invention are aqueous solutions comprising from about 50% to about 99% by weight of water.
According to certain embodiments, compositions of the present invention may be formulated as a solution comprising a surfactant. The surfactant can be any of the anionic surfactants, nonionic surfactants, amphoteric surfactants, zwitterionic surfactants cationic surfactants and mixtures clearly as are well known in the art.
Examples of anionic surfactants include, but are certainly not limited to aliphatic sulphate, aliphatic sulfonate (e.g., Cs to C22 sulfonate or disu'fonate), aromatic sulfonate (e.g., alkyl benzene sulfonate), alkyl sulfoccinates, alkyl and acyl taurates, alkyl and acyl sarcosinates, sulfoacetates, alkyl phosphates, carboxylates, isethionates, etc.
Examples of zwitterionic surfactants are exemplified by those which can be broadly described as derivatives of aliphatic quaternary ammonium, phosphonium, and sulfonium compounds, in which the aliphatic radicals can be straight or branched chain, and wherein one of the aliphatic substituents contains from about 8 to about 18 carbon atoms and one contains an anionic group, e.g., carboxy, sulfonate, sulfate, phosphate, or phosphonate.
Examples of amphoteric detergents which may be used in this invention include at least one acid group. This may be a carboxylic or a sulphonic acid group. They include quaternary nitrogen and therefore are quaternary amido acids. They should generally include an alkyl or alkenyl group of 7 to 18 carbon atoms.
Examples of nonionic which may be used includes in particular the reaction products of compounds having a hydrophobic group and a reactive hydrogen atom, for example aliphatic alcohols, acids, amides or alkyl phenols with alkylene oxides, especially ethylene oxide either alone or with propylene oxide. Specific nonionic detergent compounds are alkyl (C6-C22) phenols-ethylene oxide condensates, the condensation products of aliphatic (Ce-C ) primary or secondary linear or branched alcohols with ethylene oxide, and products made by condensation of ethylene oxide with the reaction products of propylene oxide and
ethylenediamine. Other so-called nonionic detergent compounds include long chain tertiary amine oxides, long chain tertiary phosphine oxides and dialkyl sulphoxides.
According to certain embodiments, compositions useful in the present invention may be formulated as a solution comprising an emollient. Such compositions preferably contain from about 2% to about 50% of an emollient(s). As used herein, "emollients" refer to materials used for the prevention or relief of dryness, as well as for the protection of the skin. A wide variety of suitable emollients are known and may be used herein. Sagarin, Cosmetics, Science and Technology, 2nd Edition, Vol. 1, pp. 32 43 (1972) and the International Cosmetic Ingredient Dictionary and Handbook, eds. Wenninger and McEwen, pp. 1656 61, 1626, and 1654 55 (The Cosmetic, Toiletry, and Fragrance Assoc., Washington, D.C., 7.sup.th Edition, 1997) (hereinafter "ICI Handbook") contains numerous examples of suitable materials. A lotion can be made from such a solution. Lotions typically comprise from about 1% to about 20% (e.g., from about 5% to about 10%) of an emollient(s) and from about 50% to about 90% (e.g., from about 60% to about 80%) of water.
The present compositions may be of varying phase compositions, but are preferably aqueous solutions or otherwise include an exterior aqueous phase (e.g., aqueous phase is the most exterior phase of the composition). As such, compositions of the present invention may be formulated to be oil-in-water emulsions that are shelf-stable in that the emulsion does not lose phase stability or "break" when kept at standard conditions (22 degrees Celsius, 50% relative humidity) for a week or more after it is made.
In certain embodiments, the composition of the present invention may comprise an additional benefit agent. As used herein, "benefit agent" refers to an element, an ion, a compound, or other chemical moiety that provides a cosmetic or therapeutic effect on the skin. Examples of benefit agents include small molecules, peptides, proteins, nucleic acid materials, minerals and plant extracts.
The benefit agents useful herein may be categorized by their therapeutic benefit or their postulated mode of action. However, it is to be understood that the benefit agents useful
herein may, in some circumstances, provide more than one therapeutic benefit or operate via greater than one mode of action. Therefore, the particular classifications provided herein are made for the sake of convenience and are not intended to limit the benefit agents to the particular application(s) listed.
Examples of suitable benefit agents include those that provide benefits to the skin, such as, but not limited to, depigmentation agents; reflectants; amino acids and their derivatives; antimicrobial agents; allergy inhibitors; anti-aging agents; anti-wrinkling agents, antiseptics; analgesics; shine-control agents; antipruritics; local anesthetics; anti-hair loss agents; hair growth promoting agents; hair growth inhibitor agents, antihistamines; antiinfectives; antiinflammatory agents; anticholinergics; vasoconstrictors; vasodilators; wound healing promoters; peptides, polypeptides and proteins; deodorants and anti-perspirants; medicament agents; skin firming agents, vitamins; skin lightening agents; skin darkening agents;
antifungals; depilating agents; counterirritants; hemorrhoidals; insecticides; enzymes for exfoliation or other functional benefits; enzyme inhibitors; poison ivy products; poison oak products; burn products; anti-diaper rash agents; prickly heat agents; vitamins; herbal extracts; vitamin A and its derivatives; flavonoids; sensates; anti-oxidants; hair lighteners; sunscreens; anti-edema agents, neo-collagen enhancers, film-forming polymers, chelating agents; anti- dandruff/sebhorreic dermatitis/psoriasis agents; keratolyses; and mixtures thereof.
In certain embodiments, the compositions produced via the present invention are preferably used as or in personal care products for treating or cleansing at least a portion of a human body. Examples of certain preferred personal care products include various products suitable for application to the skin, hair, oral and/or perineal region of the body, such as shampoos, hand, face, and/or body washes, bath additives, gels, lotions, creams, and the like. As discussed above, applicants have discovered unexpectedly that the instant methods provide personal care products having reduced irritation to the skin and/or eyes and, in certain embodiments one or more of desirable properties such as flash foaming characteristics, rheology, and functionality, even at high surfactant concentrations. Such products may further include a substrate onto which a composition is applied for use on the body. Examples of
suitable substrates include a wipe, pouf, sponge, and the like as well as absorbent articles, such as a bandage, sanitary napkin, tampon, and the like.
In one embodiment, the compositions produced via the present invention are preferably a rinse-off product in the form of a gel or a liquid composition that can be applied in liquid or gel form on the skin and is rinsed-off upon washing yet provides antimicrobial effect on the skin.
The present invention provides methods of treating and/or cleansing the human body comprising contacting at least a portion of the body with a composition of the present invention. Certain preferred methods comprising contacting mammalian skin, hair and/or vaginal region with a composition of the present invention to cleanse such region and/or treat such region for any of a variety of conditions including, but not limited to wrinkles, dermatitis, dryness, muscle pain, itch, and the like. Any of a variety of actives or benefit agents known in the art for treating such conditions may be used in the present invention.
The cleansing methods of the present invention may further comprise any of a variety of additional, optional steps associated conventionally with cleansing hair and skin including, for example, lathering, rinsing steps, and the like.
While the following examples demonstrate certain embodiments of the invention, they are not to be interpreted as limiting the scope of the invention, but rather as contributing to a complete description of the invention.
EXAMPLES
The following example illustrates the preparation and efficacy of antimicrobial cleansing composition in accord with certain embodiments of the present invention.
Example 1 : Minimum Inhibitory Concentration (MIC) Measurement for Magnolia Bark PE.
The 96-well plate micro-dilution broth method as described below was used to measure the Minimum Inhibitory Concentration (MIC) value of Magnolia Bark PE (90% magnolol + honokiol) against Staphylococcus aureus ATCC 6538.
1. Preparation of the 96 Well Plate(s)
1.1 96 Well Plate(s) were prepared with an antimicrobial stock solution (e.g. Magnolia Bark PE) at a starting concentration that is at least 10 folds of the concentration needed to be inhibitory against the target bacterium or panel of bacteria to be inoculated (e.g. Staphylococcus aureus ATCC 6538.)
1.2 Dispense O.lmL of sterile media in all 96 wells.
1.3 Dispense O.lmL of additional sterile media into the first two lanes.
1.4 Dispense O. lmL of the stock solution prepared in 1.1 into the wells of lane 3 and mix thoroughly.
1.5 Draw O. lmL of the contents of lane 3 and dispense it into the wells of lane 4 and mix thoroughly.
1.6 Repeat the dilution through the remaining lanes and discard the O. lmL removed from lane 12.
1.7 A series of 1 :2 dilutions of the antimicrobial under evaluation are prepared, with the lane 3 being the most concentrated (1/2 the concentration of the starting stock solution) and lane 12 being the least concentrated.
2. Inoculum Preparation
2.1 A standardized inoculum was prepared as described in the following:
2.1.1 At least three to five well-isolated colonies of the same morphological type are selected from an agar plate culture. The top of each colony is touched with a wire loop and the growth was transferred to a tube containing 4 to 5 mL of a suitable broth medium, such as tryptic soy broth.
2.1.2 The broth culture is incubated at 35 °C until it reaches or exceeds the turbidity of the 0.5 McFarland standards (usually 2 to 6 hours).
2.1.3 The turbidity of the actively growing broth culture is adjusted with sterile broth to obtain turbidity comparable to that of a 0.5 McFarland standard. This results in a suspension containing approximately 1 to 2 x 108 CFU/mL (for most commonly used bacteria).
2.2 Within 15 minutes of preparation, the adjusted inoculum suspension is diluted in the broth so that, after inoculation, each well contained approximately 5 x 104 CFU/mL. For micro-dilution tests the exact inoculum volume delivered to the wells must be known to make this calculation. For example, since the volume of medium in the well was 0.1 mL and the inoculum volume is 0.005 mL, then the 0.5 McFarland suspension (1 x 108 CFU/mL) is diluted 1 : 10 to yield 107 CFU/mL. When 0.005 mL of this suspension is inoculated into the broth, the final test concentration of bacteria would be approximately 5 x 104 CFU/well in the micro-dilution method.
2.3 Inoculating the Broth in Micro-dilution Trays. Within 15 minutes after the inoculum has been standardized as described above, dispense 0.005mL of inoculum into all wells of lanes 2 through 12 (lane 1 is the sterile control) of the micro-dilution tray (use of a pippeter capable of delivering 0.005mL is essential as this volume that does not exceed 5% of the volume in the well, making the addition negligible in regards to dilution factors); after inoculation the wells are thoroughly mixed (if mixing was by pipette pumping, moved from low concentration to high).
2.4 Perform a purity check of the inoculum suspension by sub-culturing an aliquot onto a nonselective agar plate for simultaneous incubation.
2.5 To prevent drying, each tray is sealed in a plastic bag, or with plastic tape, or with a tight-fitting plastic cover before it is incubated to minimize evaporation. ation for the Micro-dilution Method
The inoculated micro-dilution trays are incubated at 35 °C for 16 to 20 hours in an ambient air incubator. To maintain the same incubation temperature for all cultures, no more than four micro-dilution trays are stacked at the same time.
4. Determining MIC End Points
The MIC is the lowest concentration of antimicrobial agent that completely inhibits growth of the organism in the micro-dilution wells as detected by the unaided eye. Viewing devices intended to facilitate reading micro-dilution tests and recording of results is used as long as the device has been validated for this application. The amount of growth in the wells containing the antimicrobial are compared with the amount of growth in the growth-control wells or tubes (no antimicrobial) used in each set of tests when determining the growth end points. For a test to be considered valid, acceptable growth (definite turbidity) must occur in the positive control well.
The Minimum Inhibitory Concentration (MIC) of Magnolia Bark PE is measured to be 60ppm against Staphylococcus aureus, as determined by the 96-well plate micro-dilution broth method as described above.
Example 2: Preparation of Various Test Samples
The test samples were made in accord with the procedure described below.
Step I - In a manufacturing vessel of 1 kg size, mix deionized water and sodium laureth sulfate (3EO) together at a temperature in the range from about 40 to 60°C until dissolved. Add cocoamidopropyl betaine to the above aqueous solution under constant stirring to obtain a fully uniform mixture. Add sodium lauroamphoacetate and citric acid until uniform mixture formed. Maintained the vessel at 60°C.
Step II- Cool the mixture from Step I to room temperature under constant stirring. Add antimicrobial active (Magnolia extract with combined honokiol and magnolol, magnolol, or honokiol) and Benzoic acid (when present) with continuous stirring until fully dissolved and uniform.
Step III- Adjust viscosity with sodium chloride, and adjust pH with citric acid or hydrochloric acid to below 4.5. Add remaining water and/or sodium chloride to complete the sample preparation.
Example 3: Enhanced Deposition on Skin:
The efficacy of the present invention to enhance the deposition of antimicrobial actives was demonstrated using the test procedure described in the protocol below:
Subject Recruitment:
Female subjects (20-35 year old, with non-dry and non-sensitive skin type) were selected and screened by anhydrous alcohol scrubbed on forearm; those showing skin redness were excluded. Once selected, the test subjects used no leave on product two days prior to the test day
Residue Test Protocol A
1 . Mark subject's left and right forearm with a rectangular test area 2cm from the wrist and label the rectangles as L and R, respectively.
2. Adjust the tap water to temperature at approximate 37°C and flow rate at lL/min.
3. Wash subject's left and right forearms with fragrance & residue free shower gel/soap and rinse each arm for about 1 min with tap water to remove the residue. UV lamp & Image Station is used to detect any remaining residue on the forearm.
4. Wet the forearm with tap water and applied 0.00625 g/cm2 test sample as described in Tables 4 through 21 on the marked test areas (L and R) with a syringe. Rub the test area back and forth to distribute the lather until uniform and yielded a foam lasting 30 to 60 seconds.
5. Allow the foam lather of the sample to stay on the forearm for 40 to 60 S followed by a rinse, without rubbing, with 1 L of water using a nozzle (flow rate ~ 1 L/min, at a distance of ~ 1 cm from the forearm) at 37 °C for 40 to 60s. The forearm must be maintained perpendicular to the water flow to minimize slip angle.
6. Repeat Steps 4 and 5 for each test area
7. Pat dry the test areas with non-fragrant paper wipes, rinsed each test area with anhydrous alcohol by standing burette in 60 sec while rubbing the test area with constant force with each sample collection. Collect residue removed from test area using alcohol followed by HPLC analysis and quantification of such collected residue. The residue collected is analyzed by HPLC in conditions as follows:
Mobile phase:
A: Deion water + 0.1% TFA (Trifiuoroacetic acid)
B: ACN (Acetonitrile) +0.1% TFA
Mobile Phase Gradient:
Time (min) A% B%
0.0 50 50
3.0 50 50
10.0 5 95
20.0 5 95
25.0 50 50
30.0 50 50
Total analysis time: 30 min
Column: Waters C18 RP (Reversal Phase) column
Column temperature: 40°C (if available)
Detector:
Waters Fluorescence detector,
Ex (excitation spectrum): 288nm
Em (emission spectrum): 350nm
The concentration detected by HPLC is converted into total amount collected in μg by using the concentration multiply volume and then the deposition per cm2 was obtained by the
total amount collected divide test area. The % of enhanced deposition is calculated by Mean of extract base + acid) divided by Mean of extract base. As used herein, enhanced deposition is defined as the average increase of residue collected from the phenol extract/ carboxylic acid complex compared to the residue collected from the phenol extract.
Mean is defined as the average residue collected from the subjects and STD is standard deviation calculated from the group of data.
The P value, by definition, is associated with a test statistic. It is "the probability, if the test statistic really were distributed as it would be under the null hypothesis, of observing a test statistic [as extreme as, or more extreme than] the one actually observed." The smaller the P value, the more strongly the test rejects the null hypothesis, that is, the hypothesis being tested. A p-value of 0 .05 or less rejects the null hypothesis "at the 5% level" that is, the statistical assumptions used imply that only 5% of the time would the supposed statistical process produce a finding this extreme if the null hypothesis were true. 5% and 10% are common significance levels to which p-values are compared. This is further described in John A. Rice (2006), Mathematical Statistics and Data Analysis, Third Edition, Duxbury Advanced.
Table 2: The materials utilized (and the active weight % of solids therein if less than 100%)
City, Jiangsu, Chloride USP
China)
Benzoic Acid Merck Benzoic Acid.
Salicylic Acid Hebei Jingye Salicylic Acid
Pingshan
Magnolia Bark Extract H-NSEA MagnoPro® 90% honokiol and magnolol
Magnolol H-NSEA Magnolol 95% 95% Magnolol
Honokiol H-NSEA Honokiol 95% 95% Honokiol
Water d ionized (DI) water
*Other materials utilized in the present in application (if not specified) are from Aladdin Reagent or Sino Reagent Company.
Example 4
The following Examples A through D' were made in accord with the procedure described below. The sources for the materials utilized (and the active weight % of solids therein) in the following examples are listed in Table 3 and Table 3A.
Table 3 Examples A, A', B and B'
1889/50 Sodium
(Cognis) Lauroamphoacetate 1 1 1 1
Citric Acid,
Anhydrous Fine
Citric Acid
Granu!ar,USPl N
(Roche) 0.01 0.01 0.01 0.01
Benzoic Acid Benzoic Acid 1.0 1.5
Magnolia Bark
Extract Magnolia
(combined Officinalis Bark
Magnolol and Extract
Honokiol) (90%) 0.5 0.5 0.2 0.2
Sodium Chloride Sodium Chloride 1.0 1.0
TOTAL 100.00 100.00 100.00 100.00
Table 3A: Examples C, C, D, D'
Anhydrous Fine
Citric Acid
Granular,USPlN
(Roche) 0.01 0.01 0.01 0.01
Benzoic Acid Benzoic Acid 1.0 1.0
Magnolia
Officinalis Bark
Maenolol 95%
Extract Magnolol
95% 0.474 0.474
Magnolia
Officinalis Bark
Honokiol 95%
Extract Honokiol
95% 0.474 0.474
Sodium Chloride Sodium Chloride 1.0 0.5 1.0 0.5
TOTAL 100.00 100.00 100.00 100.00
Table 4 Deposition Enhancement of 0.5% Magnolia Bark Extract by 1.0 % Benzoic acid pH Adjust to 3.9 - 4.2.
Mean 0.14 0.61
STD 0.03 0.30
Average increase 42%
P Value 0.01 1
Table 4B Deposition enhancement of the individual component of Honokiol and
Magnolol in Example A' compared with Example A
As shown in Table 4 and Table 4B, HPLC analysis was conducted as described above on either the total Magnolia Bark Extract component (Examples A and A') or the individual component Honokiol and Magnolol, respectively. The composition prepared in accordance with this (Α-Α') yielded a significant enhancement with respect to the amount of antimicrobial agent, either combined (Magnolia Bark Extract) or individually (Honokiol and Magnolol), deposited on the skin surface.
The HPLC spectra included peaks that corresponded to the Honokiol enhancement and peaks that corresponded to the Magnolol enhancement. Thus the enhancement due to Honokiol and enhancement due to Magnolol can be separately determined. Table 4 shows the combined enhancement of Honokiol and Magnolol, while Table 4 B shows the data corresponding to the separate enhancement due to Honokiol and Magnolol by spectral analysis Table 4 and Table 4B were derived from the same experimental Examples.
Table 5 Deposition enhancement of 0.2% Magnolia Bark Extract by 1.5% Benzoic acid, PH Adjust to 3.9 - 4.2.
Table 5B Deposition enhancement of the individual component of Honokiol and
Magnolol has been detected in example B' compared with example B
Example B Example B
Example B' Example B'
Base
1.5%BA 1.5%BA
Base
Magnolol Honokiol
1 0.04 0.26 0.06 0.21
2 0.02 0.10 0.02 0.08
3 0.06 0.13 0.05 0.1 1
4 0.03 0.14 0.03 0.1 1
5 0.1 1 0.19 0.09 0.16
6 0.04 0.12 0.04 0.10
7 0.03 0.12 0.03 0.10
8 0.03 0.08 0.03 0.08
Mean 0.044 0.14 0.044 0.12
STD 0.03 0.06 0.02 0.05
Average
222% 173%
increase
P Value 0.001 0.000
As shown in Table 5 and Table 5B, HPLC analysis was conducted as described above on either the total Magnolia Bark Extract component (Examples B and B') or the individual component Magnolol and Honokiol, respectively. The composition prepared in accordance with this (Β-Β') yielded a significant enhancement with respect to the amount of antimicrobial agent, either combined (Magnolia Bark Extract) or individually (Honokiol and Magnolol), deposited on the skin surface.
The HPLC spectra included peaks that corresponded to the Honokiol enhancement and peaks that corresponded to the Magnolol enhancement. Table 5 shows the combined enhancement of Honokiol and Magnolol while Table 5 B shows the data corresponding to the separate enhancement due to Honokiol and Magnolol by spectral analysis. Table 5 and Table 5B were derived from the same experimental Examples.
Table 6 Deposition enhancement of 0.474% Magnolia Bark Extract Magnolol 95% by 1.0% Benzoic acid (pH = 4.1)
Example C
Example C
Magnolol Base + BA 1.0%
Magnolol Base
1 0.215 0.555
2 0.175 0.51
0.47 1.13
3
4 0.125 0.25
5 0.18 0.58
6 0.09 0.355
Mean 0.21 0.56
STD 0.14 0.31
Average
169%
increase
P Value 0.004
Table 7 Deposition enhancement of 0.474% Magnolia Bark Extract Honokiol 95% by
1.0% Benzoic acid (pH
As shown in Table 6 and Table 7, the composition prepared in accordance with these examples (C through D) yielded a significant enhancement with respect to the amount of antimicrobial agent, Honokiol and Magnolol deposited on the skin surface.
Example 5
The following Examples E through E" were made in accord with the procedure described below. The sources for the materials utilized (and the active weight % of solids therein) in the following examples are listed in Table 8.
Table 8 Examples E, E' and E"
Table 8A: Deposition Enhancement of 0.5% of Thymol by 1.5% of Salicylic Acid, pH Adjust to 3.9 - 4.2.
STD 0.02 0.02
Average increase 29%
P Value 0. 002
Table 8B: Deposition Enhancement of 0.5% of Thymol by 1.5% of Benzoic Acid, pH Adjust to 3.9 - 4.2.
As shown in Tables 8A and 8B, the composition prepared in accordance with the examples (E through E") yielded a lower degree of enhancement with respect to the amount of antimicrobial agent (thymol) deposited on the skin surface compared to Examples A through D.
Example 6:
The following Examples F-F5 were made in accord with the procedure described below. The sources for the materials utilized (and the active weight % of solids therein if less than 100%) in the following examples are listed in Table 9.
Table 9:
Inventiv Inventiv Compar Compa Compar Compare e e ative rativc ative tive
Example Exampl Example Exampl Exampl Example
Trade name INCI Name F e Fl F2 e F3 e F4 F5
Water Purified Water 74.7 72.7 73.7 73.2 73.7 73.2
Texapon N70 Sodium Laureth
LS-J Sulfate (3EO) 16 16 16 16 16 16
Tego Betaine
F50J Cocoami Betaine 8 8 8 8 8 8
DEHYTON KE 1889/50 Sodium
(Cognis) Lauroamphoacetate 1 1 1 1 1 1
Magnolia
Magnolia Officinalis
Bark Extract
Bark Extract
(90%)
0.5 0.5 0.5 0.5 0.5 0.5
O-phthalic
O-phthalic acid
acid
1.5
Ferulic acid Ferulic acid
1.0
Niacin Niacin 1.0
4-
4-methoxy benzoic
methoxybenz
acid
oic acid 1.0
p-phthalic
p-phthalic acid
acid 1.0
Sodium
Sodium Chloride
Chloride 0.3 0.3 0.3 0.3 0.3 0.3
TOTAL 100.00 100.00 100.00 100.00 100.00 100.00
Example 7: Enhanced Deposition on Dead Swine Skin:
The following examples were tested by the methods as described above but on a dead swine skin model. The dead swine skin model has shown to be comparable to human skin and is considered to be a substitute for the human skin test.
Table 10: Deposition Enhancement of 0.5% of Magnolia extract by 1.5%Opbthalic acid on dead swine skin, pH=3.9-4.3
Table 11 : Deposition Enhancement of 0.5% of Magnolia extract by 1.0% of Ferulic acid and 1.0% of Niacin on dead swine skin, pH=3.9-4.3
Table 12: Deposition Enhancement of 0.5% of Magnolia extract by 1.0% of 4- methoxybenzoic acid and p-phthalic acid on dead swine skin, pH=3.9-4.3
Example F4 Example FS
Example F Magnolia extract base + Magnolia extract base
Magnolia extract base 1.0% 4- + 1.0% p-phthalic acid methoxybenzoic acid
10"6 g/cm2 10-6 g/cm2 10~6 g
1 0.2575 0.2175 0.13
2 0.26 0.1825 0.265
3 0.175 0.1925 0.2725
4 0.1025 0.29 0.1975
5 0.1775 0.2225 0.1575
6 0.12 0.1975 0.2075
Mean 0.18 0.22 0.21
STD 0.07 0.04 0.06
Average increase 22%* 18%*
P Value 0.40 0.56
• Statistically no difference due to P value > 0.05
As shown in Tables 10- 12, HPLC analysis was conducted as described above on the Magnolia Bark Extract component with various acids (O-phthalic acid, Ferulic acid, Niacin, 4- methoxybenzoic acid and p-phthalic acid) to demonstrate that water solubility of the carboxylic acid is critical to the amount of the deposition. The composition prepared in accordance with the examples (Fl through F3) yielded a significant enhancement with respect to the amount of antimicrobial agent deposited on the skin surface. In contrary, the composition prepared in accordance with examples (F4 through F5) showed no statically significant difference in the amount of deposition.
Example 8:
The following Examples G were made in accord with the procedure described below. The sources for the materials utilized (and the active weight % of solids therein) in the following examples are listed in Table 13.
Table 13
13 072041
Table 14: Deposition Enhancement of 0.5% of Magnolia extract by 1.25% of Quinoline 2-carboxyIic acid on dead swine skin, pH-3.9-4.3
As shown in Table 14, HPLC analysis was conducted as described above on the Magnolia Bark Extract component with quinoline-2-carboxylic acid. The composition
prepared in accordance with example (G-G') yielded a significant enhancement with respect to the amount of antimicrobial agent deposited on the skin surface.
Example 9:
The following Examples H-J were made in accord with the procedure described below. The sources for the materials utilized (and the active weight % of solids therein) in the following examples are listed in Table 15.
Table 15
Table 16: Deposition Enhancement of 0.5% of Magnolia extract by 1.5% of Citric acid on human skin, pH=3.9-4.3
Table 17: Deposition Enhancement of 0.5% of Magnolia extract by 1.5% of Malic acid on human skin, pH=3.9-4.3
Table 18: Deposition Enhancement of 0.5% of Magnolia extract by 1.5% of Succinic acid on dead swine skin, pH=3.9-4.3
As shown in Tables 16- 18, HPLC analysis was conducted as described above on the Magnolia Bark Extract component with various acids (citric acid, malic acid and succinic acid). The composition prepared in accordance with the examples (H through J) yielded a significant enhancement with respect to the amount of antimicrobial agent deposited on the skin surface.
Example 10
The following Examples K-M were made in accord with the procedure described below. The sources for the materials utilized (and the active weight % of solids therein) in the following examples are listed in Table 19.
Table 20: Deposition Enhancement of 0.5% of Magnolia extract by 1.5% of Poly acrylic acid on dead swine skin, pH=3.9-4.3
Mean 0.16 0.46
STD 0.06 0.12
Average increase 196%
P Value 0.0003
As shown in Table 20, HPLC analysis was conducted as described above on the Magnolia Bark Extract component with Poly acrylic acid. The composition prepared in accordance with the example K yielded a significant enhancement with respect to the amount of antimicrobial agent deposited on the skin surface.
Table 21: Deposition Enhancement of 0.5% of Magnolia extract by 1.5% of benzoic acid on human skin, pH= 53
• Statistically no difference due to P value > 0.05
As shown in Table 21 , HPLC analysis was conducted as described above on the Magnolia Bark Extract component with Benzoic acid at pH 5.3. The composition prepared in
accordance with the example M-M' showed no statically significant enhancement with respect to the amount of antimicrobial agent deposited on the skin surface.
Claims
1. A composition comprising an antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and a carboxylic acid, said composition having a pH of from about 3.0 to about 5.2.
2. The composition of claim 1 wherein said antimicrobial active comprises both honokiol and magnolol.
3. The composition of claim 1 wherein said antimicrobial active is extracted from a plant of the genus Magnolia.
4. The composition of claim 3 wherein the antimicrobial active is an extract of
Magnolia officinalis bark.
5. The composition of claim 1 comprising from about 0.004 wt % to about
1.25% wt of the antimicrobial active.
6. The composition of claim 5 comprising from about 0.01 wt % to about 1.25 wt% of the antimicrobial active.
7. The composition of claim 1 comprising from about 0.001 wt % to about 5.0 wt % of said carboxylic acid.
8. The composition of claim 7 comprising from about 0.1 wt % to about 2.5 wt
% of said carboxylic acid.
9. The composition of claim 1 wherein said carboxylic acid has a water
solubility of greater than 0.05g/100g.
10. The composition of claim 1 wherein said carboxylic acid has a water solubility of greater than O. l g/l OOg.
1 1 . The composition of claim 1 wherein said carboxylic acid has a water solubility of greater than 0.2g/ t00g.
12. The composition of claim 1 wherein said carboxylic acid is an aromatic acid.
13. The composition of claim 1 wherein said carboxylic acid i ; an aliphatic acid.
14. The composition of claim 12 wherein said aromatic carboxylic acid is selected from the group consisting of benzoic acid, salicylic acid, O-phthalic acid, Quinotinc- 2-carboxylic acid , 4 methoxybenzoic acid, ferulic acid, niacin, and
combinations thereof.
1 5. The composition of claim 13 wherein said aliphatic carboxylic acid is selected from the group consisting of citric acid, malic acid, succinic acid, poly acrylic acid, and combinations thereof.
1 6. A composition comprising from about 0.004 wt % to about 1.25 wt% of
antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and from about 0.001 wt % to about 5.0 wt % of aromatic carboxylic acid selected from the group consisting of benzoic acid, salicylic acid, O-phthalic acid, Quinoline-2-carboxylic acid, ferulic acid, niacin, and combinations thereof, said composition having a pll of from about 3.0 to about 5.2.
17. A composition comprising from about 0.01 wt % to about 1 .25% wt of
antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and from about 0.1 wt % to about 2.5 wt % of aromatic carboxylic acid selected from the group consisting of benzoic acid, salicylic acid, O-phthalic acid, Quinoline-2-carboxylie acid, ferulic acid, niacin,
and combinations thereof, said composition having a H of from about 3.0 to about 5.2.
18. A composition comprising from about 0.004 wt% to about 1.25 wt% wt of
antimicrobial active selected from the group consisting of honokiol, magnolol, and combinations thereof, and from about 0.001 wt % to about 5.0 wt % of aliphatic carboxylic acid selected from the group consisting of citric acid, malic acid, succinic acid, poly acrylic acid, and combinations thereof, said composition having a pH of from about 3.0 to about 5.2.
1 9. A composition comprising from about 0.01 wt % to about 1 .25 wt% of
antimicrobial active selected from the group consisting of honokiol. magnolol, and combinations thereof, and from about 0.1 wt % to about 2.5 wt % of aliphatic carboxylic acid selected from the group consisting of citric acid, malic acid, succinic acid, poly acrylic acid, and combinations thereof, said composition having a pi I of from about 3.0 to about 5.2.
20. The composition of claim I wherein said composition is in the form of a rinse-off product.
21. The composition of claim 1 wherein said composition is in the form of a leave-on product.
22. The composition of claim 1 wherein said composition is in the form of a solution, suspension, lotion, cream, serum, gel, stick, spray, ointment, liquid wash, soap bar, shampoo, hair conditioner, paste, foam, powder, mousse, shaving cream, hydrogel, or film-forming product.
23. The composition of claim 1 wherein said antimicrobial cleansing composition exhibits an enhanced deposition of the antimicrobial actives greater than 50% as measured by residue test protocol A.
24. The composition of claim I wherein said antimicrobial cleansing composition exhibits an enhanced deposition of the antimicrobial actives greater than 100% as measured by residue test protocol A.
25. A method of cleansing skin comprising applying to skin in need of cleansing a composition of claim 1.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2013/072041 WO2014131191A1 (en) | 2013-03-01 | 2013-03-01 | A composition containing honokiol and/or magnolol and uses thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2013/072041 WO2014131191A1 (en) | 2013-03-01 | 2013-03-01 | A composition containing honokiol and/or magnolol and uses thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014131191A1 true WO2014131191A1 (en) | 2014-09-04 |
Family
ID=51427488
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2013/072041 Ceased WO2014131191A1 (en) | 2013-03-01 | 2013-03-01 | A composition containing honokiol and/or magnolol and uses thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2014131191A1 (en) |
Cited By (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017083363A1 (en) | 2015-11-09 | 2017-05-18 | Unigen, Inc. | Natural preservatives and antimicrobial agents, including compositions thereof |
| WO2018098160A1 (en) * | 2016-11-23 | 2018-05-31 | Gojo Industries, Inc. | Topical cleansing composition with prebiotic/probiotic additive |
| WO2018098156A1 (en) * | 2016-11-23 | 2018-05-31 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| CN109640930A (en) * | 2016-08-19 | 2019-04-16 | 荷兰联合利华有限公司 | Antimicrobial compositions |
| WO2019115136A1 (en) | 2017-12-15 | 2019-06-20 | Unilever N.V. | Topical composition comprising antimicrobial lipid |
| US10617621B2 (en) | 2016-08-19 | 2020-04-14 | Conopco, Inc. | Antimicrobial composition |
| WO2020083737A1 (en) * | 2018-10-24 | 2020-04-30 | Unilever N.V. | A topical composition |
| US10806769B2 (en) | 2016-03-31 | 2020-10-20 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| US10874700B2 (en) | 2016-03-31 | 2020-12-29 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| US20220087914A1 (en) * | 2018-12-18 | 2022-03-24 | Conopco, Inc., D/B/A Unilever | An antimicrobial composition |
| EP4039783A1 (en) * | 2021-02-05 | 2022-08-10 | ATP Cosmetic GmbH | Mild antimicrobial foam soap comprising malic acid and levulinic acid |
| CN115227592A (en) * | 2022-05-07 | 2022-10-25 | 彭氏(惠州)实业发展有限公司 | Bacteriostatic composition and application thereof |
| EP3727300B1 (en) | 2017-12-22 | 2022-10-26 | Unilever Global IP Limited | An antimicrobial composition |
| US11564879B2 (en) | 2016-11-23 | 2023-01-31 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| CN117323319A (en) * | 2022-06-27 | 2024-01-02 | 广西大学 | Traditional Chinese medicine monomer composition for preventing and controlling necrotic enteritis of chickens |
| US12539324B2 (en) | 2016-03-31 | 2026-02-03 | Gojo Industries, Inc. | Antimicrobial peptide stimulating sanitizing composition |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006069209A1 (en) * | 2004-12-22 | 2006-06-29 | Colgate-Palmolive Company | Antibacterial and anti-inflammatory oral care composition |
| WO2008130752A2 (en) * | 2007-04-19 | 2008-10-30 | Mary Kay Inc. | Magnolia extract containing compositions |
| CN102046158A (en) * | 2008-05-30 | 2011-05-04 | Wm.雷格利Jr.公司 | Oral compositions comprising enhanced antibacterial combination of antioxidants and magnolia bark extract |
| WO2011106492A1 (en) * | 2010-02-24 | 2011-09-01 | Colgate-Palmolive Company | Oral care compositions |
| WO2011106493A2 (en) * | 2010-02-24 | 2011-09-01 | Colgate-Palmolive Company | Oral care compositions |
-
2013
- 2013-03-01 WO PCT/CN2013/072041 patent/WO2014131191A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006069209A1 (en) * | 2004-12-22 | 2006-06-29 | Colgate-Palmolive Company | Antibacterial and anti-inflammatory oral care composition |
| WO2008130752A2 (en) * | 2007-04-19 | 2008-10-30 | Mary Kay Inc. | Magnolia extract containing compositions |
| CN102046158A (en) * | 2008-05-30 | 2011-05-04 | Wm.雷格利Jr.公司 | Oral compositions comprising enhanced antibacterial combination of antioxidants and magnolia bark extract |
| WO2011106492A1 (en) * | 2010-02-24 | 2011-09-01 | Colgate-Palmolive Company | Oral care compositions |
| WO2011106493A2 (en) * | 2010-02-24 | 2011-09-01 | Colgate-Palmolive Company | Oral care compositions |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2017083363A1 (en) | 2015-11-09 | 2017-05-18 | Unigen, Inc. | Natural preservatives and antimicrobial agents, including compositions thereof |
| US10780173B2 (en) * | 2015-11-09 | 2020-09-22 | Unigen, Inc. | Natural preservatives and antimicrobial agents, including compositions thereof |
| US12539324B2 (en) | 2016-03-31 | 2026-02-03 | Gojo Industries, Inc. | Antimicrobial peptide stimulating sanitizing composition |
| US11998575B2 (en) | 2016-03-31 | 2024-06-04 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| US11633451B2 (en) | 2016-03-31 | 2023-04-25 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| US10874700B2 (en) | 2016-03-31 | 2020-12-29 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| US10806769B2 (en) | 2016-03-31 | 2020-10-20 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| US10617621B2 (en) | 2016-08-19 | 2020-04-14 | Conopco, Inc. | Antimicrobial composition |
| JP2019524778A (en) * | 2016-08-19 | 2019-09-05 | ユニリーバー・ナームローゼ・ベンノートシヤープ | Antibacterial composition |
| US20190201311A1 (en) * | 2016-08-19 | 2019-07-04 | Conopco, Inc., D/B/A Unilever | An antimicrobial composition |
| CN109640930B (en) * | 2016-08-19 | 2021-11-26 | 联合利华知识产权控股有限公司 | Antimicrobial compositions |
| JP7009444B2 (en) | 2016-08-19 | 2022-01-25 | ユニリーバー・ナームローゼ・ベンノートシヤープ | Antibacterial composition |
| US11246817B2 (en) * | 2016-08-19 | 2022-02-15 | Conopco, Inc. | Antimicrobial composition |
| CN109640930A (en) * | 2016-08-19 | 2019-04-16 | 荷兰联合利华有限公司 | Antimicrobial compositions |
| US11564879B2 (en) | 2016-11-23 | 2023-01-31 | Gojo Industries, Inc. | Sanitizer composition with probiotic/prebiotic active ingredient |
| WO2018098156A1 (en) * | 2016-11-23 | 2018-05-31 | Gojo Industries, Inc. | Antimicrobial peptide stimulating cleansing composition |
| WO2018098160A1 (en) * | 2016-11-23 | 2018-05-31 | Gojo Industries, Inc. | Topical cleansing composition with prebiotic/probiotic additive |
| JP2021506753A (en) * | 2017-12-15 | 2021-02-22 | ユニリーバー・ナームローゼ・ベンノートシヤープ | Topical composition containing antibacterial lipids |
| JP7301832B2 (en) | 2017-12-15 | 2023-07-03 | ユニリーバー・アイピー・ホールディングス・ベスローテン・ヴェンノーツハップ | Topical composition containing antimicrobial lipid |
| WO2019115136A1 (en) | 2017-12-15 | 2019-06-20 | Unilever N.V. | Topical composition comprising antimicrobial lipid |
| EP3727300B1 (en) | 2017-12-22 | 2022-10-26 | Unilever Global IP Limited | An antimicrobial composition |
| WO2020083737A1 (en) * | 2018-10-24 | 2020-04-30 | Unilever N.V. | A topical composition |
| US20220087914A1 (en) * | 2018-12-18 | 2022-03-24 | Conopco, Inc., D/B/A Unilever | An antimicrobial composition |
| US12311044B2 (en) * | 2018-12-18 | 2025-05-27 | Conopco, Inc. | Antimicrobial composition |
| EP4039783A1 (en) * | 2021-02-05 | 2022-08-10 | ATP Cosmetic GmbH | Mild antimicrobial foam soap comprising malic acid and levulinic acid |
| CN115227592A (en) * | 2022-05-07 | 2022-10-25 | 彭氏(惠州)实业发展有限公司 | Bacteriostatic composition and application thereof |
| CN115227592B (en) * | 2022-05-07 | 2024-07-19 | 彭氏(惠州)实业发展有限公司 | Bacteriostatic agent composition and application thereof |
| CN117323319A (en) * | 2022-06-27 | 2024-01-02 | 广西大学 | Traditional Chinese medicine monomer composition for preventing and controlling necrotic enteritis of chickens |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2014131191A1 (en) | A composition containing honokiol and/or magnolol and uses thereof | |
| US12083085B2 (en) | Antiseptic, antiseborrheic, exfoliating composition to remove or prevent acne | |
| EP1725210A1 (en) | Substances with a probiotic action used in deodorants | |
| CN110035797B (en) | Synergistic herbal composition with prebiotic properties for the treatment of acne | |
| CN102123765A (en) | Skin lightening composition comprising CO2 extract | |
| JP5765744B2 (en) | Preventive or therapeutic agent for atopic dermatitis, and external preparation | |
| AU2018382466A1 (en) | Propanediol monoacetate mononitrate | |
| JP2003104886A (en) | Skin external preparation composition | |
| BRPI1101172A2 (en) | deodorant and antiperspirant composition, process for producing deodorant and antiperspirant composition and method for the prevention and / or reduction of perspiration odors | |
| KR20150116504A (en) | A cosmetics composition containing tea tree and solt | |
| JP2002145793A (en) | Skin care preparation | |
| CN112423727A (en) | Anti-dandruff composition | |
| JP5000964B2 (en) | Testosterone 5α-reductase activity inhibitor, androgen receptor antagonist, use thereof, and method for suppressing androgen activity expression | |
| JP2001131025A (en) | Cosmetic for scalp and hair | |
| WO2023126222A1 (en) | Antimicrobial system with beta carboline alkaloid and phenolic acid and compositions comprising them | |
| EP4456722A1 (en) | Antimicrobial system with indole alkaloid and phenolic acid and compositions comprising them | |
| KR100858196B1 (en) | Skin composition containing nutmeg active ingredient | |
| EP4456723A1 (en) | Antimicrobial system with beta carboline alkaloid and indole alkaloid and compositions comprising them | |
| BR112020022532B1 (en) | ANTI-DANDRUFF COMPOSITION | |
| JP2024542694A (en) | Antibacterial Composition | |
| JP2003335660A (en) | Lipase activity inhibitor and cosmetic containing the same | |
| KR20200026114A (en) | Topical compositions comprising pichia anomala and a soy product | |
| JP2000256142A (en) | 5alpha-reductase-inhibiting composition | |
| JP2003081735A (en) | Cosmetic |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13876170 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13876170 Country of ref document: EP Kind code of ref document: A1 |