WO2013172554A1 - Polymer, preparation method thereof, and molded product comprising said polymer - Google Patents
Polymer, preparation method thereof, and molded product comprising said polymer Download PDFInfo
- Publication number
- WO2013172554A1 WO2013172554A1 PCT/KR2013/003030 KR2013003030W WO2013172554A1 WO 2013172554 A1 WO2013172554 A1 WO 2013172554A1 KR 2013003030 W KR2013003030 W KR 2013003030W WO 2013172554 A1 WO2013172554 A1 WO 2013172554A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- substituted
- unsubstituted
- organic group
- polymer
- same
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- QKTBDFBIQLOPPB-UHFFFAOYSA-N C[O]1C(C=C[I]=C=C2)=C2Oc2c1cccc2 Chemical compound C[O]1C(C=C[I]=C=C2)=C2Oc2c1cccc2 QKTBDFBIQLOPPB-UHFFFAOYSA-N 0.000 description 1
- DNMYITOWMOUSOJ-UHFFFAOYSA-N c(cc1)cc2c1nc(C=C=[I]C=C1)c1n2 Chemical compound c(cc1)cc2c1nc(C=C=[I]C=C1)c1n2 DNMYITOWMOUSOJ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/58—Other polymers having nitrogen in the main chain, with or without oxygen or carbon only
- B01D71/62—Polycondensates having nitrogen-containing heterocyclic rings in the main chain
- B01D71/64—Polyimides; Polyamide-imides; Polyester-imides; Polyamide acids or similar polyimide precursors
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/22—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols by diffusion
- B01D53/228—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols by diffusion characterised by specific membranes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/76—Macromolecular material not specifically provided for in a single one of groups B01D71/08 - B01D71/74
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/0683—Polycondensates containing six-membered rings, condensed with other rings, with nitrogen atoms as the only ring hetero atoms
- C08G73/0694—Polycondensates containing six-membered rings, condensed with other rings, with nitrogen atoms as the only ring hetero atoms with only two nitrogen atoms in the ring, e.g. polyquinoxalines
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1039—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors comprising halogen-containing substituents
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1042—Copolyimides derived from at least two different tetracarboxylic compounds or two different diamino compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/10—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
- C08G73/1075—Partially aromatic polyimides
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G73/00—Macromolecular compounds obtained by reactions forming a linkage containing nitrogen with or without oxygen or carbon in the main chain of the macromolecule, not provided for in groups C08G12/00 - C08G71/00
- C08G73/06—Polycondensates having nitrogen-containing heterocyclic rings in the main chain of the macromolecule
- C08G73/22—Polybenzoxazoles
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L79/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing nitrogen with or without oxygen or carbon only, not provided for in groups C08L61/00 - C08L77/00
- C08L79/04—Polycondensates having nitrogen-containing heterocyclic rings in the main chain; Polyhydrazides; Polyamide acids or similar polyimide precursors
- C08L79/08—Polyimides; Polyester-imides; Polyamide-imides; Polyamide acids or similar polyimide precursors
Definitions
- the present invention relates to a polymer, a method for producing the same, and a molded article including the polymer.
- One embodiment of the present invention is to provide a polymer having excellent low molecular permeability and selectivity, and excellent heat resistance, chemical resistance and solubility in a solvent.
- Another embodiment of the present invention is to provide a method for producing the polymer.
- Another embodiment of the present invention is to provide a molded article comprising the polymer.
- a polymer according to an aspect of the present invention includes a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof; A polyimide comprising a repeating unit represented by Formulas 3 and 4, a copolymer thereof, or a blend thereof; Or combinations thereof.
- Ar 1 is the same as or different from each other at each repeating unit, and each independently an aromatic ring group selected from a substituted or unsubstituted tetravalent C6 to C60 arylene group and a substituted or unsubstituted tetravalent C4 to C60 heterocyclic group,
- T is the same or different at each repeating unit and is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted group Tetravalent C6 to C40 aromatic organic group,
- Y is the same or different at each repeating unit and each independently is OH, SH or NH 2 ,
- n is an integer satisfying 10 ⁇ n ⁇ 400.
- the polymer derived from the polyamic acid or the polymer derived from the polyimide may have a free volume degree (FFV) of about 0.20 to about 0.35.
- the polymer derived from the polyamic acid or the polymer derived from the polyimide may have an interplanar distance by XRD measurement in the range of about 520 pm to about 850 pm.
- the polymer derived from the polyamic acid or the polymer derived from the polyimide may have a BET surface area of about 280 m 2 / g to about 600 m 2 / g.
- Ar 1 may be selected from those represented by the following formulas.
- W 1 and W 2 are the same or different and are each independently O, S, or C ( ⁇ O),
- Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
- Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
- T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
- R 1 to R 62 are the same or different and are each independently hydrogen or a substituted or unsubstituted C1 to C10 aliphatic organic group
- k1 to k3, k8 to k14, k24, k25, k49 to k54 and k59 to k62 are integers of 0 to 2
- k5, k15, k16, k19, k21 and k23 are integers of 0 or 1
- k4, k6, k7, k17, k18, k20, k22, k26 to k29, k31, k34 to k36, k38, k41, k44 to k46 and k55 to k58 are integers from 0 to 3,
- k30, k37, k42, k43, k47 and k48 are integers from 0 to 4,
- k32, k33, k39 and k40 are integers from 0 to 5.
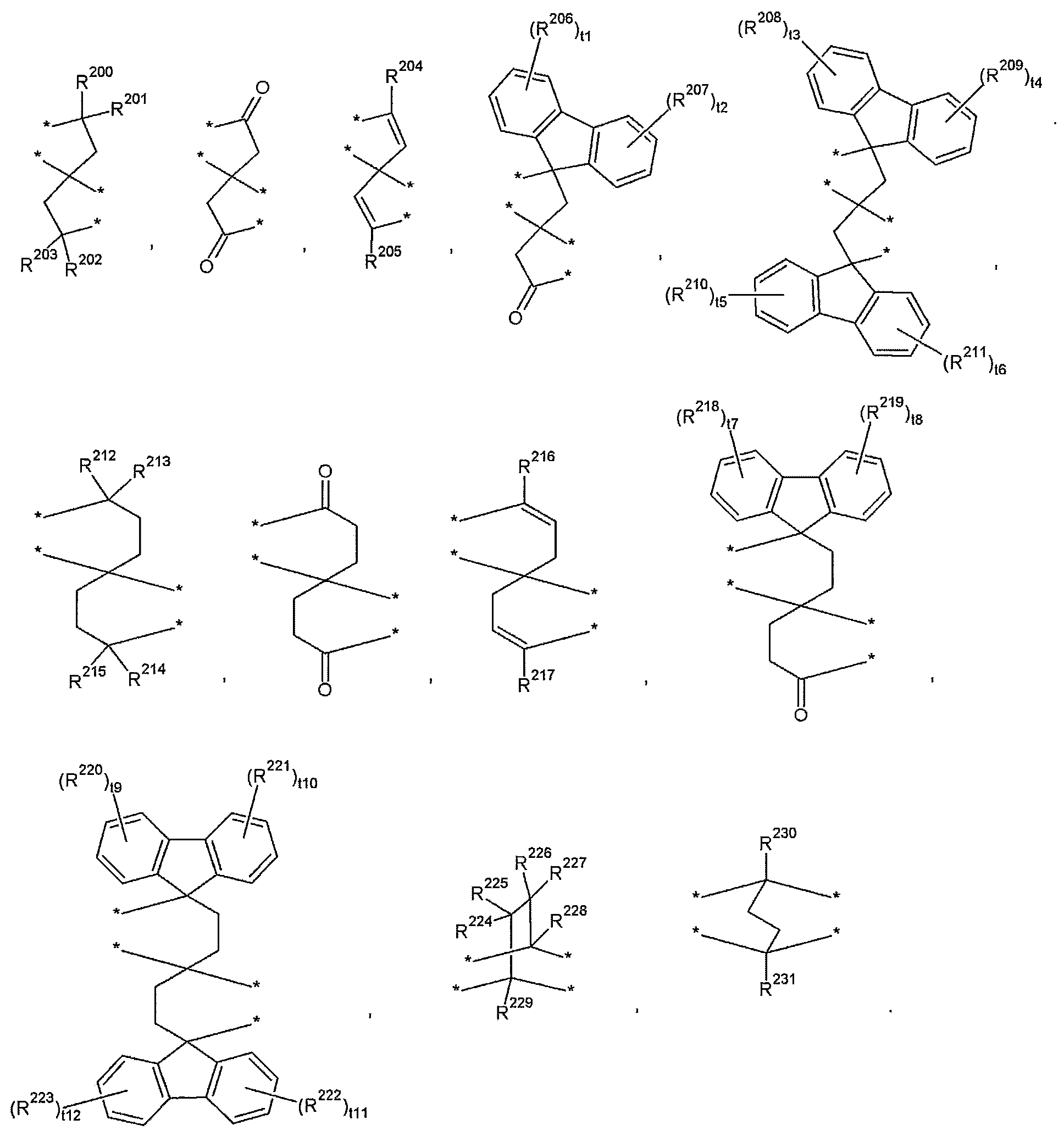
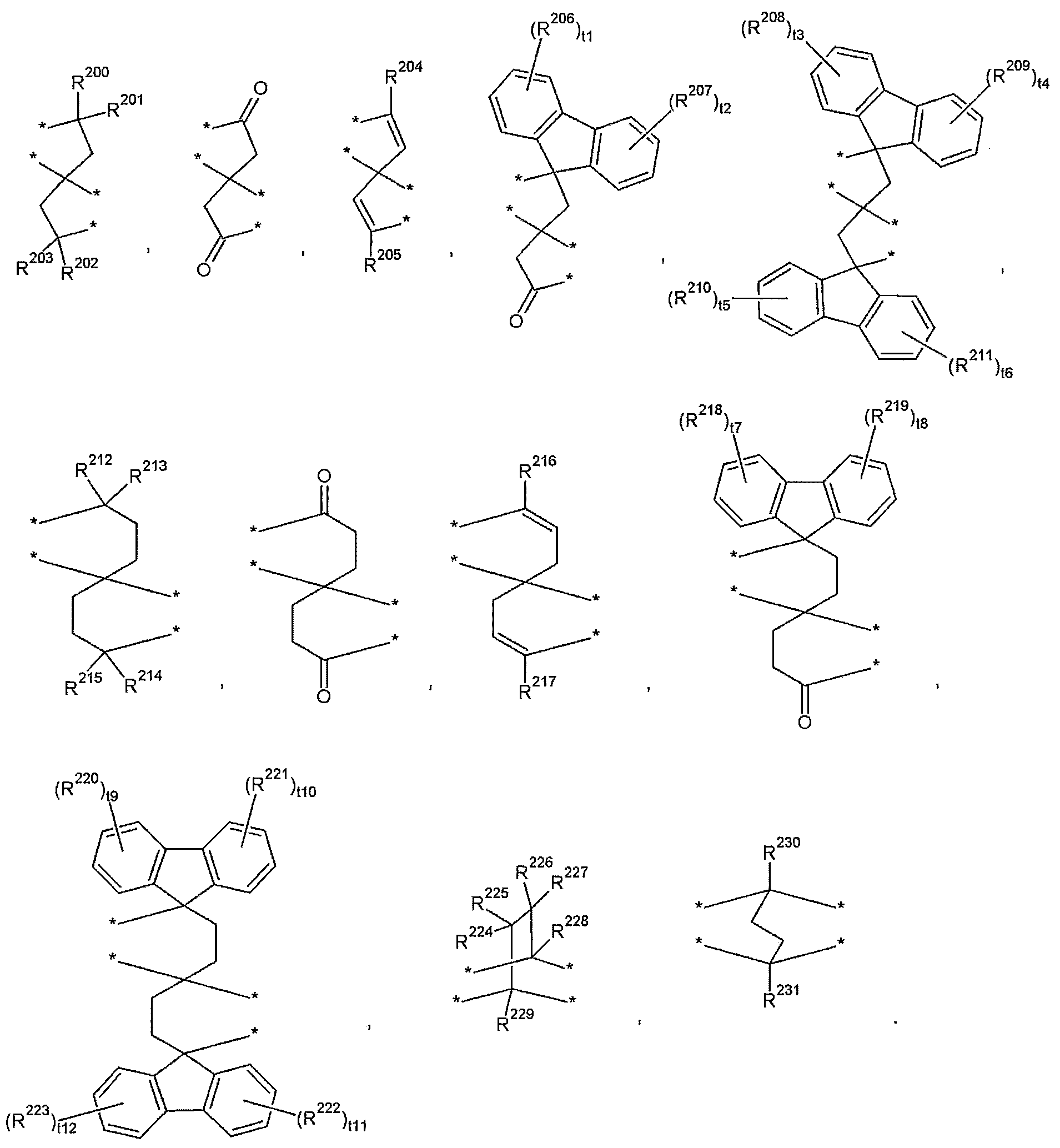
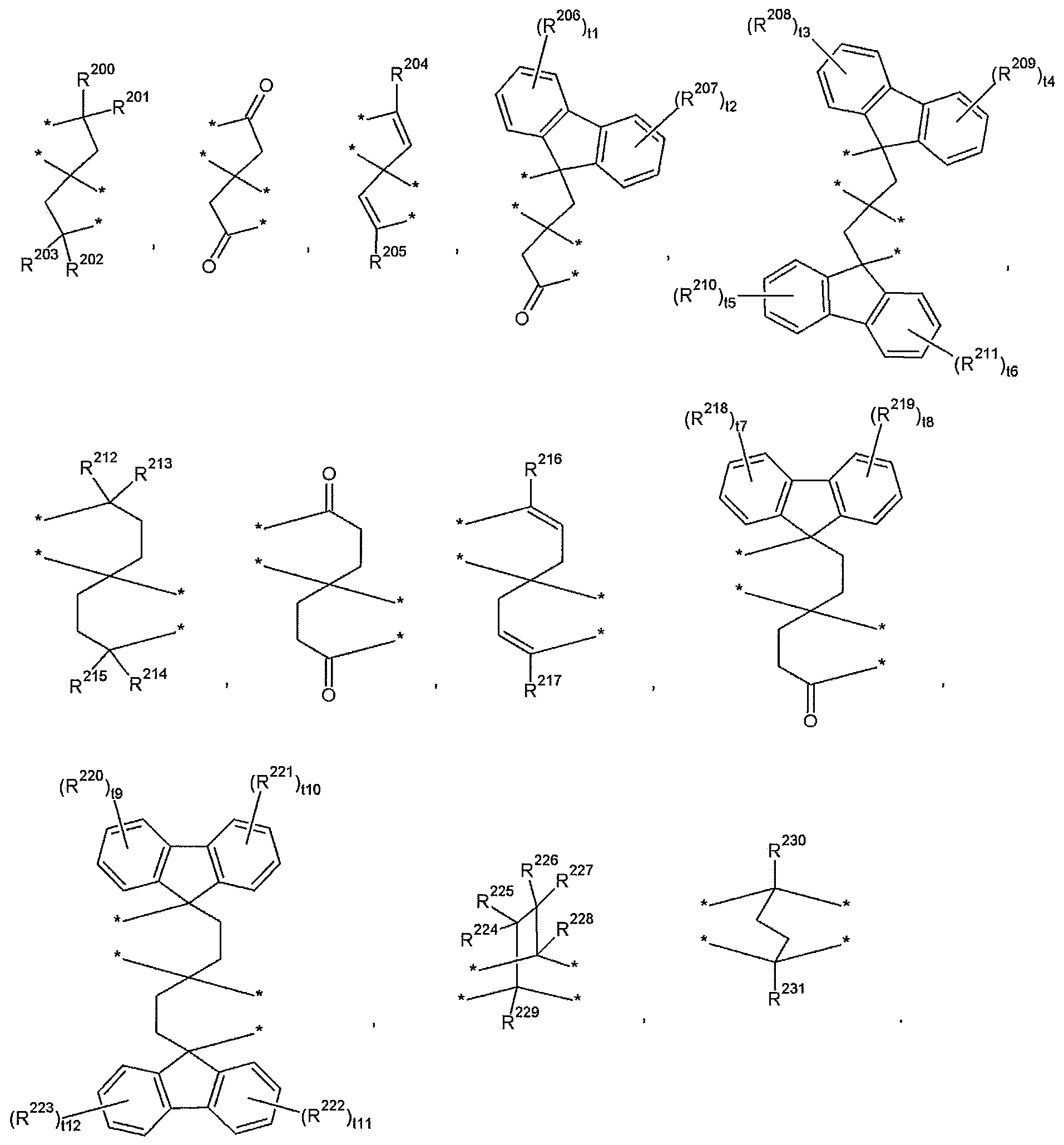
- T may be selected from those represented by the following formulas.
- R 200 to R 231 are the same or different from each other and are each independently hydrogen, a substituted or unsubstituted C1 to C30 aliphatic organic group, a substituted or unsubstituted C3 to C30 alicyclic organic group, or a substituted or unsubstituted divalent C6 To C30 aromatic organic group,
- t1 to t12 are the same or different and are each independently an integer of 0 to 4;
- T may be selected from those represented by the following formulas.
- Ar 1 may be selected from those represented by the following formulas.
- the molar ratio between each repeating unit in the copolymer of polyamic acid including the repeating units represented by Formula 1 and Formula 2 may be about 0.1: 9.9 to about 9.9: 0.1.
- the molar ratio between each repeating unit in the copolymer of the polyimide including the repeating units represented by Formulas 3 and 4 may be about 0.1: 9.9 to about 9.9: 0.1.
- the polymer derived from the polyamic acid and the polymer derived from the polyimide may include a compound or a copolymer thereof including a repeating unit represented by any one of Formulas 5 to 8.
- Ar 1, T and n are as described in each of Ar 1, T, and n in the above Chemical Formulas 1 to 16,
- Y '' is O or S.
- Ar 1 ′ may be selected from those represented by the following formulas.
- W 1 and W 2 are the same or different and are each independently O, S, or C ( ⁇ O),
- Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
- Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
- T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
- R 1 to R 62 are the same or different and are each independently hydrogen or a substituted or unsubstituted C1 to C10 aliphatic organic group, or a metal sulfonate group,
- k63, k69, k84 to k88, k92 to k96, k102 to k109, k116 and k119 are integers from 0 to 4,
- k67, k72, k73, k76, k79, k80, k82, k90, k98, k100, k101, k113, k114, k121 and k122 are integers from 0 to 2
- k70 is an integer of 0 or 1
- k89, k91, k97 and k99 are integers of 0-5.
- T in Ar 1 ′ is also the same as defined for T in Ar 1 , and examples and specific examples are also the same as those defined in T in Ar 1 .
- Ar 1 ′ may be selected from those represented by the following formulas.
- M is hydrogen or a metal, said metal being sodium, potassium, lithium, alloys thereof or a combination thereof.
- the polymer may have a weight average molecular weight (Mw) of about 10,000 g / mol to about 500,000 g / mol.
- a method of preparing a polymer wherein a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof is imidized to form a poly Obtaining a mead; And heat treating the polyimide.
- the heat treatment may be performed at a temperature increase rate of about 1 ° C / min to about 30 ° C / min to about 350 ° C to about 500 ° C, and at that temperature under an inert atmosphere for about 1 minute to about 12 hours.
- Method for producing a polymer according to another embodiment of the present invention is a step of heat-treating a polyimide comprising a repeating unit represented by the formula (3) and formula (4), a copolymer thereof, or a blend thereof It includes.
- Method for producing a polymer according to another embodiment of the present invention is a polyamic acid comprising a repeating unit represented by the formula (1) and formula (2), a copolymer thereof, or a polyamic acid comprising a blend thereof, and the formula Imidizing a polyamic acid in a compound including a polyimide comprising a repeating unit represented by Formula 3 and Formula 4, a copolymer thereof, or a polyimide including a blend thereof to obtain a polyimide; And heat treating the polyimide.
- Another aspect of the invention provides a molded article comprising the polymer.
- the molded article may be a gas separation membrane.
- the polymer according to the present invention is excellent in permeability and selectivity of low molecules, excellent in heat resistance, chemical resistance, solubility in solvents, and also excellent in mechanical strength.
- substituted or “substituted” means that the hydrogen atom in the compound or functional group is a C1 to C10 alkyl group, a C1 to C10 alkoxy group, a C1 to C10 haloalkyl group, a C1 to C10 haloalkoxy group and a C6 group.
- To C20 is substituted with one or more substituents selected from the group consisting of aromatic organic groups.
- a "hetero ring group” means a substitution containing 1 to 3 heteroatoms selected from the group consisting of O, S, N, P, Si, and combinations thereof in one ring, or Unsubstituted C2 to C30 cycloalkyl group, substituted or unsubstituted C2 to C30 cycloalkenyl group, substituted or unsubstituted C2 to C30 cycloalkynyl group, or substituted or unsubstituted C2 to C30 heteroaryl group.
- an "aliphatic organic group” means a C1 to C30 alkyl group, a C2 to C30 alkenyl group, a C2 to C30 alkynyl group, a C1 to C30 alkylene group, a C2 to C30 alkenylene group, or a C2 to C30 group.
- An alkynylene group and specifically, a C1 to C15 alkyl group, a C2 to C15 alkenyl group, a C2 to C15 alkynyl group, a C1 to C15 alkylene group, a C2 to C15 alkenylene group, or a C2 to C15 alkynylene group, and Alicyclic organic group "means a C3 to C30 cycloalkyl group, a C3 to C30 cycloalkenyl group, a C3 to C30 cycloalkynyl group, a C3 to C30 cycloalkylene group, a C3 to C30 cycloalkenylene group, or a C3 to C30 cycloalkynylene group Specifically, C3 to C15 cycloalkyl group, C3 to C15 cycloalkenyl group, C3 to C15 cycloalkynyl group, C3 to C15 cycloalkylene group, C3
- “combination” means mixed or copolymerized.
- “copolymerization” means block copolymerization to random copolymerization, and “copolymer” means block copolymer to random copolymerization.
- a polymer according to an embodiment of the present invention includes a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof; A polyimide comprising a repeating unit represented by Formulas 3 and 4, a copolymer thereof, or a blend thereof; Or combinations thereof.
- Ar 1 is the same as or different from each other at each repeating unit, and each independently an aromatic ring group selected from a substituted or unsubstituted tetravalent C6 to C60 arylene group and a substituted or unsubstituted tetravalent C4 to C60 heterocyclic group,
- T is the same or different at each repeating unit and is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted group Tetravalent C6 to C40 aromatic organic group,
- Y is the same or different at each repeating unit and each independently is OH, SH or NH 2 ,
- n is an integer satisfying 10 ⁇ n ⁇ 400.
- the polyamic acid including the repeating units represented by Formulas 1 and 2 and the polyimide including the repeating units represented by Formulas 3 and 4 may be prepared according to a general method.
- the polyamic acid may be prepared by reacting a tetraamine anhydride with a diamine including an OH, SH, or NH 2 group present at an ortho position relative to an amine group.
- the polyimide can be prepared by thermal solution imidization or chemical imidization of the polyamic acid prepared as described above. The thermal solution imidization and chemical imidization will be described later.
- the polyamic acid is imidized and thermally converted through a manufacturing process to be described later, and the polyimide is thermally converted through a manufacturing process to be described later, and has a high degree of free polybenzoxazole, polybenzothiazole, polypyrrolone or It can be converted into a polymer comprising a combination of these.
- the polymer derived from the polyamic acid and the polymer derived from the polyimide include a rigid structure having excellent solubility in an organic solvent and a flexible structure, and at the same time, a rigid structure connected between the flexible structures. , Specifically, a rigid ladder structure. Accordingly, the polymer derived from the polyamic acid and the polymer derived from the polyimide include a repeating structure in which a rigid ladder structure is connected by a flexible hinge, and thus fine pores are formed while the refractive staircase is amorphously distributed. It may have, and also excellent mechanical strength and workability can be easily used to manufacture in the form of film, fiber, hollow fiber and the like.
- the polymer derived from the polyamic acid and the polymer derived from the polyimide may have a free volume (FFV) of about 0.20 to about 0.35, and the d-spacing by XRD is about 520 pm to about It can be in the range of 850 pm.
- FMV free volume
- the polymer derived from the polyamic acid and the polymer derived from the polyimide may have a Brunauer-Emmett-Teller (BET) surface area of about 280 m 2 / g to about 600 m 2 / g. Therefore, the polymer derived from the polyamic acid and the polymer derived from the polyimide can efficiently permeate or selectively separate low molecules.
- BET Brunauer-Emmett-Teller
- examples of Ar 1 may be selected from those represented by the following formulas, but are not limited thereto.
- W 1 and W 2 are the same or different and are each independently O, S, or C ( ⁇ O),
- Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
- Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
- T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
- R 1 to R 62 are the same or different and are each independently hydrogen or a substituted or unsubstituted C1 to C10 aliphatic organic group
- k1 to k3, k8 to k14, k24, k25, k49 to k54 and k59 to k62 are integers of 0 to 2
- k5, k15, k16, k19, k21 and k23 are integers of 0 or 1
- k4, k6, k7, k17, k18, k20, k22, k26 to k29, k31, k34 to k36, k38, k41, k44 to k46 and k55 to k58 are integers from 0 to 3,
- k30, k37, k42, k43, k47 and k48 are integers from 0 to 4,
- k32, k33, k39 and k40 are integers from 0 to 5.
- T examples of the T may be selected from those represented by the following formula, but are not limited thereto.
- R 200 to R 231 are the same or different from each other and are each independently hydrogen, a substituted or unsubstituted C1 to C30 aliphatic organic group, a substituted or unsubstituted C3 to C30 alicyclic organic group, or a substituted or unsubstituted divalent C6 To C30 aromatic organic group,
- t1 to t12 are the same or different and are each independently an integer of 0 to 4;
- T may be selected from those represented by the following formula, but is not limited thereto.
- the molar ratio between each repeating unit in the copolymer of polyamic acid including the repeating units represented by Formula 1 and Formula 2 is about 0.1: 9.9 to about 9.9: 0.1, specifically about 2: 8 to about 8: 2 More specifically, about 5: 5.
- the molar ratio between each repeating unit in the copolymer of the polyimide including the repeating units represented by Formula 3 and Formula 4 is about 0.1: 9.9 to about 9.9: 0.1, specifically about 2: 8 to about 8: 2, more specifically, about 5: 5.
- the copolymerization ratio affects the molecular structure and morphology of the prepared polymer, which is related to density, pore properties, heat resistance, tensile strength, shear force, and the like.
- the prepared polymer may efficiently permeate or selectively separate low molecules, and may have excellent heat resistance, chemical resistance, mechanical strength and processability.
- the polymer derived from the polyamic acid and the polymer derived from the polyimide may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
- Ar 1 , T and n are as described for Ar 1 , T and n of Formulas 1 to 4, respectively,
- Y '' is O or S.
- an example of Ar 1 ' may be selected from those represented by the following formulas, but is not limited thereto.
- W 1 and W 2 are the same or different and are each independently O, S, or C ( ⁇ O),
- Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
- Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
- T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
- R 63 to R 123 are the same or different and are each independently hydrogen, a substituted or unsubstituted C 1 to C 10 aliphatic organic group, or a metal sulfonate group,
- k63, k69, k84 to k88, k92 to k96, k102 to k109, k116 and k119 are integers from 0 to 4,
- k67, k72, k73, k76, k79, k80, k82, k90, k98, k100, k101, k113, k114, k121 and k122 are integers from 0 to 2
- k70 is an integer of 0 or 1
- k89, k91, k97 and k99 are integers of 0-5.
- Ar 1 ' may be selected from those represented by the following formula, but is not limited thereto.
- M is hydrogen or a metal, said metal being sodium, potassium, lithium, alloys thereof or a combination thereof.
- Polymers derived from polyamic acid and polymers derived from polyimide according to an embodiment of the present invention may be prepared using polyamic acid or polyimide that is soluble in a general organic solvent, and the polymer may be easily formed without defects or cracks. Since it can be coated, it is possible to simplify the manufacturing process to reduce the process cost and to form a large area.
- the polymer can control the pore size or distribution by adjusting the manufacturing process conditions. As a result, the polymer may be widely applied to various applications such as gas permeation, gas separation, steam separation, water purification, adsorbent, heat resistant fiber, thin film manufacturing, and the like.
- the polymer may be derived from a combination of the polyamic acid and the polyimide, in which case the polymer may include a polymer derived from the polyamic acid and a polymer derived from the polyimide.
- the description of the polyamic acid, the polyimide, the polymer derived from the polyamic acid and the polymer derived from the polyimide is as described above.
- the polymer including the polymer derived from the polyamic acid and the polymer derived from the polyimide may be a polymer having a weight ratio of about 0.1: 9.9 to about 9.9: 0.1 of the polymer: polyimide derived from the polyamic acid. 8: 2 to about 2: 8 by weight, more specifically about 5: 5 by weight.
- Such a polymer may have both the properties of the polymer derived from the polyamic acid described above and the properties of the polymer derived from polyimide. It can also have good dimensional stability and long term stability.
- the polymer derived from the polyamic acid and the polymer derived from the polyimide may each have a weight average molecular weight (Mw) of about 10,000 g / mol to about 500,000 g / mol.
- Mw weight average molecular weight
- the polymer derived from the polyamic acid and the polymer derived from the polyimide can be easily synthesized and have excellent mechanical strength and dimensional stability.
- Another embodiment of the present invention comprises the steps of imidating a polyamic acid comprising a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof, to obtain a polyimide; And it provides a method for producing a polymer comprising the step of heat-treating the polyimide.
- the polymer may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
- the imidization may be performed by a thermal imidization process, but is not limited thereto.
- the thermal imidization may be performed at about 150 ° C. to about 300 ° C. for about 30 minutes to about 2 hours under an inert atmosphere. If the temperature of imidation is less than the said range, the imidation of the polyamic acid which is a precursor is insignificant, On the contrary, even if it exceeds the said range, there is no big increase in effect and it is uneconomical.
- the imidation condition is Ar, which is a functional group of the polyamic acid.
- Ar which is a functional group of the polyamic acid.
- One It can adjust suitably according to the kind of T and Y.
- the polyimide When the polyimide is heat-treated, it is rearranged through a heat conversion reaction to obtain a polymer having picopores.
- the polymer having picopores has a reduced density compared to the polyimide, increased free volume and increased interplanar distance as picopores are well connected to each other. As a result, the polymer having the picopores can efficiently permeate or selectively separate low molecules.
- the polymer derived from the polyamic acid has a rigid structure, specifically rigid, which has excellent solubility in organic solvents and includes a flexible structure, and at the same time is connected between the flexible structures.
- a rigid structure specifically rigid, which has excellent solubility in organic solvents and includes a flexible structure, and at the same time is connected between the flexible structures.
- it can have fine pores, and can also have excellent workability and improve mechanical strength such as tensile strength, expansion rate, and the like.
- the heat treatment may be performed at a temperature rising rate of about 1 ° C / min to 30 ° C / min to about 350 ° C to about 500 ° C, and at that temperature under an inert atmosphere for about 1 minute to about 12 hours.
- the heat treatment may be performed at an elevated temperature rate of about 5 ° C./min to about 20 ° C./min to about 350 ° C. to about 450 ° C., and at that temperature for about 1 hour to about 6 hours under an inert atmosphere.
- the heat treatment may be performed at an elevated temperature rate of about 10 ° C./min to about 15 ° C./min to about 420 ° C. to about 450 ° C., and at that temperature for about 2 hours to about 5 hours under inert atmosphere. have.
- the heat conversion reaction may be sufficiently made.
- a polybenzoxazole, polybenzothiazole or polypyrrolone polymer including a repeating unit represented by Chemical Formulas 5 to 8 may be obtained.
- Preparation of such a polymer is carried out through the CO 2 or H 2 O removal reaction in the polyimide obtained by the imidization.
- Another embodiment of the present invention is to prepare a polymer comprising the step of heat-treating a polyimide comprising a repeating unit represented by the formula (3) and formula (4), a copolymer thereof, or a blend thereof Provide a method.
- the polymer may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
- the polyimide can be obtained by imidizing a polyamic acid including a repeating unit represented by the above formulas (1) and (2), for example, chemical imidation or thermal solution imidation.
- the chemical imidization can be accomplished by running the reaction at about 20 ° C. to about 180 ° C. for about 4 hours to about 24 hours. At this time, as a catalyst, acetic anhydride may be added for removal of pyridine and generated water. If the temperature of the chemical imidization is within the above range, the imidization of the polyamic acid can be sufficiently made.
- the chemical imidization may be performed after first protecting OH, SH and NH 2 , which are functional groups present at the ortho position of the amine group in the polyamic acid. Specifically, a protecting group may be introduced into OH, SH, and NH 2 , which are functional groups, and imidization may be performed to remove the protecting group.
- the protecting group may be trimethylchlorosilane ((CH 3 ) 3 SiCl), triethylchlorosilane ((C 2 H 5 ) 3 SiCl), tributylchlorosilane ((C 4 H 9 ) 3 SiCl), tribenzylchlorosilane Chlorosilanes such as ((C 6 H 5 ) 3 SiCl), triethoxychlorosilane ((OC 2 H 5 ) 3 SiCl), and hydrofuran, such as tetrahydrofurane (THF), may be used.
- tertiary amines such as trimethylamine, triethylamine, tripropylamine, pyridine and the like can be used.
- Diluted hydrochloric acid, sulfuric acid, nitric acid, acetic acid, etc. may be used as the material for removing the protecting group.
- Chemical imidization using a protecting group as described above may increase the yield and molecular weight of the polymer according to an embodiment of the present invention.
- the thermal solution imidization can be accomplished by running the reaction on the solution at about 100 ° C. to about 180 ° C. for about 2 hours to about 30 hours. If the temperature of the thermal solution imidization is within the above range, the imidization of the polyamic acid can be sufficiently achieved.
- the thermal solution imidization may be performed after first protecting OH, SH and NH 2 which are functional groups present at the ortho position of the polyamic acid amine group.
- a protecting group may be introduced into OH, SH, and NH 2 , which are functional groups, and imidization may be performed to remove the protecting group.
- chlorosilanes such as trimethylchlorosilane, triethylchlorosilane, tributylchlorosilane, tribenzylchlorosilane, triethoxychlorosilane, and hydrofuran, such as tetrahydrofuran, may be used.
- Tertiary amines such as triethylamine, tripropylamine, pyridine and the like can be used.
- Diluted hydrochloric acid, sulfuric acid, nitric acid, acetic acid, etc. may be used as a material for removing the protecting group.
- the thermal solution imidization may be performed using an azeotrope made by further adding benzenes such as benzene, toluene, xylene, cresol, aliphatic organic solvents such as hexane, cyclohexane, and the like.
- benzenes such as benzene, toluene, xylene, cresol, aliphatic organic solvents such as hexane, cyclohexane, and the like.
- Thermal solution imidization made by introducing a protecting group and using an azeotrope as described above may increase the yield and molecular weight of the polymer according to one embodiment of the present invention.
- the imidation condition is Ar, which is a functional group of the polyamic acid.
- Ar which is a functional group of the polyamic acid.
- One It can adjust suitably according to the kind of T and Y.
- Another embodiment of the present invention is a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a polyamic acid including a blend thereof, and represented by Formula 3 and Formula 4 Imidizing a polyamic acid in a compound including a polyimide comprising a repeating unit, a copolymer thereof, or a combination of polyimides including a blend thereof to obtain a polyimide; And it provides a method for producing a polymer comprising the step of heat-treating the polyimide.
- the polymer may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
- the polymer according to one embodiment of the present invention includes a rigid structure present in the polymer, so that it can withstand not only under mild conditions but also under long working hours, acidic conditions and harsh conditions such as high humidity and high temperature. That is, the polymer according to one embodiment of the present invention has excellent chemical stability, heat resistance, and mechanical properties.
- the polymer comprising a repeating unit represented by Formula 5 to Formula 8 or copolymers thereof are designed to have an appropriate weight average molecular weight in the manufacturing step, specifically, the weight average molecular weight is about 10,000 g / mol to about 500,000 g / mol. If their weight average molecular weight is within the above range, the physical properties of the polymer can be maintained excellent.
- the polymer according to an embodiment of the present invention is a polymer derived from polyamic acid or a polymer derived from polyimide, and has picopores. Since two or more picopores are connected to each other to form an hourglass shape, the picopores may have a high free volume to efficiently penetrate or selectively separate small molecules.
- the molded article may be a sheet, a film, a powder, a film, or a fiber, but is not limited thereto.
- the molded article may include a structure in which a rigid ladder structure is repeated in the form of being connected by a flexible hinge, and thus may have micropores, specifically dumbbell-shaped micropores, which are formed while the refractive staircase is amorphous. . Therefore, the molded article can efficiently penetrate or selectively separate low-molecules, and is excellent in heat resistance, surface hardness, and dimensional stability, and thus can be used in various technical fields requiring the above performance. Specifically, the molded article may be used as a gas separation membrane.
- Example and comparative example of this description are described.
- the following examples are merely examples of the present disclosure, and the present disclosure is not limited by the following examples.
- a polymer containing polybenzoxazole was prepared as shown in Scheme 1 below.
- the dissolved solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film.
- the thickness of the film thus prepared was 64 ⁇ m.
- the polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes. Subsequently, a gas separation membrane was prepared by performing a heat treatment to be heated to 425 ° C. and maintained for 2 hours to obtain a transparent pale brown TSBIDD-based polybenzoxazole film.
- the FTIR analysis, free volume, interplanar distance and BET surface area were measured for the polybenzoxazole film obtained according to the following method.
- FTIR Fourier transform infrared
- the density of the polymer is related to the free volume and affects the gas permeability.
- the film density was measured by buoyancy method using a Sartorius LA 310S analytical balance according to Equation 1 below.
- ⁇ P is the density of the polymer
- ⁇ W is the density of deionized water
- Wa is the weight of the polymer measured in air
- Ww is the weight of the polymer measured in deionized water.
- Free volume degrees (FFV, V f ) were calculated according to the following equation (2) from the data.
- V is the specific volume of the polymer
- Vw is the specific Van der Waals volume.
- the interplanar distance was calculated according to the Bragg's equation from the X-ray diffraction pattern results.
- the BET surface area was measured by Brunauer-Emmett-Teller using an isothermal curve of the relative pressure-nitrogen adsorption of materials measured at a temperature of 77K using a pore size and surface area analyzer (ASAP 2020, Micromeritics, USA). Calculated using the formula.
- the prepared polymer had a free volume of 0.26 and an interplanar distance of 642 pm.
- the BET surface area was 446 m 2 / g.
- Example 2 Except that the heat treatment for 30 minutes at 450 °C was carried out in the same manner as in Example 1 to prepare a polymer containing a polybenzoxazole, to obtain a polybenzoxazole film.
- the prepared polymer had a free volume of 0.26 and an interplanar distance of 619 pm.
- the BET surface area was 371 m 2 / g.
- the prepared solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film.
- the thickness of the film thus prepared was 126 mu m.
- the polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
- a gas separation membrane was prepared by performing a heat treatment to be heated to 425 ° C. and maintained for 2 hours to obtain a transparent pale brown TSBIDD-based polybenzoxazole film.
- the prepared polymer had a free volume of 0.26 and an interplanar distance of 634 pm.
- the BET surface area was 431 m 2 / g.
- a polymer containing polybenzoxazole was prepared as shown in Scheme 2 below.
- Heat treatment temperature 1.5 hours at 450 °C
- the prepared polymer had a free volume of 0.21 and an interplanar distance of 525 pm.
- the BET surface area was 282.5 m 2 / g.
- the prepared solution was cast on a glass plate and kept in a vacuum oven at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour to prepare a film.
- the thickness of the film thus prepared was 52 ⁇ m.
- the polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
- the prepared polymer had a free volume of 0.22 and an interplanar distance of 544 pm.
- a polymer containing polybenzoxazole was prepared as shown in Scheme 3 below.
- the polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
- the prepared polymer had a free volume of 0.18 and an interplanar distance of 526 pm.
- the BET surface area was 306 m 2 / g.
- the prepared solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film.
- the thickness of the film thus prepared was 93 ⁇ m.
- the polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
- the prepared polymer had a free volume of 0.25 and an interplanar distance of 565 pm.
- a polymer containing polybenzoxazole was prepared as shown in Scheme 4 below.
- the polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
- the distance between planes of the prepared polymer was 831pm.
- the BET surface area was 434 m 2 / g.
- a porous polymer having a trapezoidal structure was prepared as shown in Scheme 5 below.
- the reaction flask was placed in an oil bath and raised to 55 ° C., and maintained for about 23 hours. At this time, the stirring speed is 810 rpm.
- the reaction flask is cooled and then poured into 300 ml of water to obtain a crude polymer.
- the polymer is again dissolved in chloroform, and then immersed in methanol and dried again to obtain a PIM-1 polymer.
- a polymer solution having a concentration of 2 wt% was prepared using a chloroform solvent, purified using a 0.45 ⁇ m syringe filter, and then cast on a glass plate to volatilize the solvent slowly at room temperature for one day to obtain a polymer film.
- the PIM-1 polymer film is dried in an oven at 70 ° C. to completely remove the solvent.
- the average thickness was 53 ⁇ m and the BET surface area was 850 m 2 / g.
- a porous polymer including polybenzoxazole was prepared as shown in Scheme 6 below.
- 2,2'-bis (3-amino-4-hydroxyphenyl) hexafluoropropane (bisAPAF, 3.663 g, 10 mmol) and NMP (15.06 mL) were placed in a 100 mL three neck flask with nitrogen gas and maintained at 0 ° C. Place in ice bath.
- NMP 15.06 mL
- a solution of propylene oxide (PO, 0.3 mL) and terephthaloyl chloride (TCL, 2.030 g, 10 mmol) mixed with NMP (8.35 mL) was placed in a flask and allowed to proceed for 2 hours.
- a viscous polyhydroxyamide solution is obtained.
- the solution is poured onto a glass plate, cast, and then placed in a vacuum oven for 1 hour at 100 ° C. and 10 hours at 200 ° C. to remove the solvent. After cooling slowly, a poly hydroxyamide precursor film is obtained.
- the poly hydroxy amide precursor film flows argon gas, placed in a furnace that increases by 5 ° C. per minute, increases the temperature to 350 ° C. and is maintained for one hour. After slowly cooling the temperature inside the furnace, a polybenzoxazole film is obtained.
- the average thickness of the polymer film was 40 ⁇ m, the density was 1.39 g / cm 3 , and the interplanar distance was 719 pm.
- a porous polymer including polybenzoxazole was prepared as shown in Scheme 7 below.
- 2,2'-bis (3-amino-4-hydroxyphenyl) hexafluoropropane (bisAPAF, 3.663 g, 10 mmol) and NMP (15.06 mL) were placed in a 100 mL three neck flask with nitrogen gas and maintained at 0 ° C. Place in ice bath.
- NMP 15.06 mL
- a solution of propylene oxide (PO, 0.3 mL) and para-phenylene (tedraloyl dichloride) (TPCL, 2.030 g, 10 mmol) mixed with NMP (8.35 mL) was added to the flask, followed by reaction for 2 hours. Allow this to proceed.
- a viscous polyhydroxyamide solution is obtained.
- the solution is poured onto a glass plate, cast, and then placed in a vacuum oven for 1 hour at 100 ° C. and 10 hours at 200 ° C. to remove the solvent. After cooling slowly, a poly hydroxyamide precursor film is obtained.
- the poly hydroxyamide precursor film is placed in a furnace with flowing argon gas and increased by 5 ° C. per minute, increased in temperature to 350 ° C. and maintained for one hour. After slowly cooling the temperature inside the furnace, a polybenzoxazole film is obtained.
- the average thickness of the polymer film was 42 ⁇ m, the density was 1.32 g / cm 3 , and the interplanar distance was 758 pm.
- the gas separation membranes prepared in Examples 1 to 8 generally have elongation percent at break (unit:%) at the gas separation membranes prepared in Comparative Examples 1 to 3. Better than This is because the polymers included in the gas separation membranes prepared in Examples 1 to 8 have a benzoxazole structure and a ladder-type structure at the same time to have a rigidity and flexibility to increase tensile strength.
- the polymers included in the gas separation membranes prepared in Examples 1 to 8 have a benzoxazole structure and a ladder structure at the same time, such as long working time, acidic conditions and high humidity, high temperature, as well as mild conditions. It can be seen that it can withstand harsh conditions.
- Test Example 2 Measurement of gas permeability and selectivity
- Gas permeability and selectivity were measured using a high-vacuum time-lag apparatus, the downstream volume was adjusted to 30 cm 3 , and the upstream and downstream pressures were 33 atm and 0.002 atm, respectively. Measurements were made using a Baratron transducer with full scale.
- V is the volume of the fixed downstream collector
- A is just area
- p 1 and p 2 are the pressures upstream and downstream, respectively.
- R , T and t are gas constant, temperature and time, respectively.
- the gas separation membranes prepared in Examples 1 to 8 generally have excellent gas permeability compared to the gas separation membranes prepared in Comparative Examples 2 and 3. In addition, it can be seen that the gas separation membranes prepared in Examples 1 to 8 have a higher level of gas selectivity than the gas separation membranes prepared in Comparative Examples 2 and 3.
- the gas separation membranes prepared in Examples 1 to 8 can efficiently separate a larger amount of gas than the gas separation membranes prepared in Comparative Examples 2 and 3.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Analytical Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Macromolecular Compounds Obtained By Forming Nitrogen-Containing Linkages In General (AREA)
Description
본 기재는 고분자, 이의 제조 방법 및 상기 고분자를 포함하는 성형품에 관한 것이다.The present invention relates to a polymer, a method for producing the same, and a molded article including the polymer.
경성 유기물질에서 기공을 통한 저분자 또는 이온의 확산은 본질적으로 서브나노 또는 나노 기술에 기초한 현상이다. 이러한 유기물질을 포함하는 막은 저분자 또는 이온을 선택적으로 용이하게 분리하기 위해 이용될 수 있으며, 이러한 기술은 화학물질의 제조공정, 에너지 전환, 에너지 저장, 유기 배터리, 연료 전지, 기체 분리 등 다양한 분야에서 다양한 목적을 위해 이용될 수 있다.The diffusion of small molecules or ions through pores in hard organic materials is essentially a phenomenon based on subnano or nanotechnology. Membranes containing such organic materials can be used to selectively separate low molecules or ions easily, and this technique can be used in various fields such as chemical manufacturing, energy conversion, energy storage, organic batteries, fuel cells, and gas separation. It can be used for a variety of purposes.
따라서 이에 대한 연구가 활발히 진행되고 있다. 그러나 상기와 같이 저분자 또는 이온을 선택적으로 용이하게 분리하면서, 동시에 내열성, 내화학성, 일반적인 용매에의 가용성 및 기계적 강도를 모두 갖춘 물질에 대한 개발이 이루어지지 못하고 있어 다양한 응용 분야에 적용되지 못하고 있는 실정이다.Therefore, research on this is being actively conducted. However, as described above, it is not easy to selectively separate low-molecules or ions, and at the same time, development of a material having both heat resistance, chemical resistance, solubility in general solvents, and mechanical strength has not been applied to various applications. to be.
본 발명의 일 구현예는 저분자의 투과도 및 선택도가 우수하고, 내열성, 내화학성 및 용매에의 가용성이 우수한 고분자를 제공하기 위한 것이다.One embodiment of the present invention is to provide a polymer having excellent low molecular permeability and selectivity, and excellent heat resistance, chemical resistance and solubility in a solvent.
본 발명의 다른 일 구현예는 상기 고분자의 제조 방법을 제공하기 위한 것이다.Another embodiment of the present invention is to provide a method for producing the polymer.
본 발명의 또 다른 일 구현예는 상기 고분자를 포함하는 성형품을 제공하기 위한 것이다.Another embodiment of the present invention is to provide a molded article comprising the polymer.
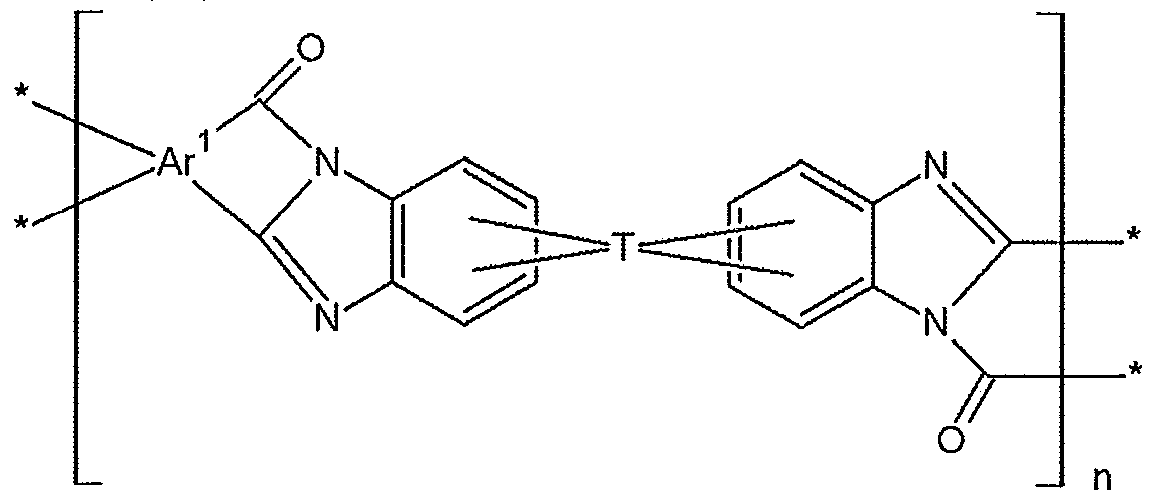
본 발명의 일 측면에 따른 고분자는 하기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리아믹산; 하기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리이미드; 또는 이들의 조합으로부터 유도된다.A polymer according to an aspect of the present invention includes a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof; A polyimide comprising a repeating unit represented by Formulas 3 and 4, a copolymer thereof, or a blend thereof; Or combinations thereof.
[규칙 제91조에 의한 정정 17.06.2013]
화학식 1
Formula 1
[규칙 제91조에 의한 정정 17.06.2013]
화학식 2
Formula 2
[규칙 제91조에 의한 정정 17.06.2013]
화학식 3
Formula 3
[규칙 제91조에 의한 정정 17.06.2013]
화학식 4
Formula 4
상기 화학식 1 내지 화학식 4에서,In Chemical Formulas 1 to 4,
Ar1은 각각의 반복단위에서 동일하거나 서로 상이하며 각각 독립적으로 치환 또는 비치환된 4가의 C6 내지 C60 아릴렌기 및 치환 또는 비치환된 4가의 C4 내지 C60 헤테로 고리기에서 선택되는 방향족 고리기이고, 상기 방향족 고리기는 단독으로 존재하거나; 2개 이상이 서로 접합되어 축합 고리를 형성하거나; 2개 이상이 단일결합, O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, C(=O)NH 또는 치환 또는 비치환된 4가의C1 내지 C30 지방족 유기기에 의해 연결되어 있고,Ar 1 is the same as or different from each other at each repeating unit, and each independently an aromatic ring group selected from a substituted or unsubstituted tetravalent C6 to C60 arylene group and a substituted or unsubstituted tetravalent C4 to C60 heterocyclic group, The aromatic ring groups are present alone; Two or more are joined to each other to form a condensed ring; Two or more single bonds, O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where 1 ≦ p ≦ 10) , (CF 2 ) q (where 1 ≦ q ≦ 10), C (CH 3 ) 2 , C (CF 3 ) 2 , C (═O) NH or a substituted or unsubstituted tetravalent C 1 to C 30 aliphatic organic group Connected by
T는 각각의 반복단위에서 동일하거나 서로 상이하며 각각 독립적으로 치환 또는 비치환된 4가의 C1 내지 C40 지방족 유기기, 치환 또는 비치환된 4가의 C3 내지 C40 지환족 유기기, 또는 치환 또는 비치환된 4가의 C6 내지 C40 방향족 유기기이고,T is the same or different at each repeating unit and is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted group Tetravalent C6 to C40 aromatic organic group,
Y는 각각의 반복단위에서 동일하거나 서로 상이하며 각각 독립적으로 OH, SH 또는 NH2이고,Y is the same or different at each repeating unit and each independently is OH, SH or NH 2 ,
n은 10≤n≤400을 만족하는 정수이다.n is an integer satisfying 10 ≦ n ≦ 400.
상기 폴리아믹산으로부터 유도된 고분자 또는 상기 폴리이미드로부터 유도된 고분자는 약 0.20 내지 약 0.35의 자유 체적도(FFV)를 가질 수 있다.The polymer derived from the polyamic acid or the polymer derived from the polyimide may have a free volume degree (FFV) of about 0.20 to about 0.35.
상기 폴리아믹산으로부터 유도된 고분자 또는 상기 폴리이미드로부터 유도된 고분자는 XRD 측정에 의한 면간 거리가 약 520 pm 내지 약 850 pm의 범위에 있을 수 있다.The polymer derived from the polyamic acid or the polymer derived from the polyimide may have an interplanar distance by XRD measurement in the range of about 520 pm to about 850 pm.
상기 폴리아믹산으로부터 유도된 고분자 또는 상기 폴리이미드로부터 유도된 고분자는 약 280 m2/g 내지 약 600 m2/g의 BET 표면적을 가질 수 있다.The polymer derived from the polyamic acid or the polymer derived from the polyimide may have a BET surface area of about 280 m 2 / g to about 600 m 2 / g.
상기 Ar1은 하기 식으로 표시된 것 중에서 선택될 수 있다.Ar 1 may be selected from those represented by the following formulas.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
X1 내지 X6은 동일하거나 서로 상이하며 각각 독립적으로 O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, 또는 C(=O)NH이고,X 1 to X 6 are the same or different from each other and each independently O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where , 1 ≦ p ≦ 10), (CF 2 ) q (where 1 ≦ q ≦ 10), C (CH 3 ) 2 , C (CF 3 ) 2 , or C (═O) NH,
W1 및 W2는 동일하거나 서로 상이하며 각각 독립적으로 O, S, 또는 C(=O)이고,W 1 and W 2 are the same or different and are each independently O, S, or C (═O),
Z1은 O, S, CR300R301 또는 NR302이고, 여기서 R300, R301 및 R302는 동일하거나 서로 상이하며 각각 독립적으로 수소 또는 C1 내지 C5 알킬기이고,Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
Z2 및 Z3는 동일하거나 서로 상이하며 각각 독립적으로 N 또는 CR303(여기서, R303은 수소 또는 C1 내지 C5 알킬기이다)이나 동시에 CR303은 아니고,Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
T는 각각 독립적으로 치환 또는 비치환된 4가의 C1 내지 C40 지방족 유기기, 치환 또는 비치환된 4가의 C3 내지 C40 지환족 유기기, 또는 치환 또는 비치환된 4가의 C6 내지 C40 방향족 유기기이고,T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
R1 내지 R62는 동일하거나 서로 상이하며 각각 독립적으로 수소, 또는 치환 또는 비치환된 C1 내지 C10 지방족 유기기이고,R 1 to R 62 are the same or different and are each independently hydrogen or a substituted or unsubstituted C1 to C10 aliphatic organic group,
k1 내지 k3, k8 내지 k14, k24, k25, k49 내지 k54 및 k59 내지 k62는 0 내지 2의 정수이고,k1 to k3, k8 to k14, k24, k25, k49 to k54 and k59 to k62 are integers of 0 to 2,
k5, k15, k16, k19, k21 및 k23은 0 또는 1의 정수이고,k5, k15, k16, k19, k21 and k23 are integers of 0 or 1,
k4, k6, k7, k17, k18, k20, k22, k26 내지 k29, k31, k34 내지 k36, k38, k41, k44 내지 k46 및 k55 내지 k58은 0 내지 3의 정수이고,k4, k6, k7, k17, k18, k20, k22, k26 to k29, k31, k34 to k36, k38, k41, k44 to k46 and k55 to k58 are integers from 0 to 3,
k30, k37, k42, k43, k47 및 k48은 0 내지 4의 정수이고,k30, k37, k42, k43, k47 and k48 are integers from 0 to 4,
k32, k33, k39 및 k40은 0 내지 5의 정수이다.k32, k33, k39 and k40 are integers from 0 to 5.
상기 T는 하기 식으로 표시된 것 중에서 선택될 수 있다.T may be selected from those represented by the following formulas.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
R200 내지 R231은 동일하거나 서로 상이하며 각각 독립적으로 수소, 치환 또는 비치환된 C1 내지 C30 지방족 유기기, 치환 또는 비치환된 C3 내지 C30 지환족 유기기, 또는 치환 또는 비치환된 2가의 C6 내지 C30 방향족 유기기이고,R 200 to R 231 are the same or different from each other and are each independently hydrogen, a substituted or unsubstituted C1 to C30 aliphatic organic group, a substituted or unsubstituted C3 to C30 alicyclic organic group, or a substituted or unsubstituted divalent C6 To C30 aromatic organic group,
t1 내지 t12는 동일하거나 서로 상이하며 각각 독립적으로 0 내지 4의 정수이다.t1 to t12 are the same or different and are each independently an integer of 0 to 4;
구체적으로는 상기 T는 하기 식으로 표시되는 것 중에서 선택될 수 있다.Specifically, T may be selected from those represented by the following formulas.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
구체적으로는 상기 Ar1은 하기 식으로 표시된 것 중에서 선택될 수 있다.Specifically, Ar 1 may be selected from those represented by the following formulas.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 고분자에서, 상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산의 공중합체에서의 각 반복단위 사이의 몰비는 약 0.1:9.9 내지 약 9.9:0.1일 수 있다.In the polymer, the molar ratio between each repeating unit in the copolymer of polyamic acid including the repeating units represented by Formula 1 and Formula 2 may be about 0.1: 9.9 to about 9.9: 0.1.
한편, 상기 고분자에서, 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드의 공중합체에서의 각 반복단위 사이의 몰비는 약 0.1:9.9 내지 약 9.9:0.1일 수 있다.Meanwhile, in the polymer, the molar ratio between each repeating unit in the copolymer of the polyimide including the repeating units represented by Formulas 3 and 4 may be about 0.1: 9.9 to about 9.9: 0.1.
상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자는 하기 화학식 5 내지 화학식 8 중 어느 하나로 표시되는 반복단위를 포함하는 화합물 또는 이들의 공중합체를 포함할 수 있다.The polymer derived from the polyamic acid and the polymer derived from the polyimide may include a compound or a copolymer thereof including a repeating unit represented by any one of Formulas 5 to 8.
[규칙 제91조에 의한 정정 17.06.2013]
화학식 5
Formula 5
[규칙 제91조에 의한 정정 17.06.2013]
화학식 6
Formula 6
[규칙 제91조에 의한 정정 17.06.2013]
화학식 7
Formula 7
[규칙 제91조에 의한 정정 17.06.2013]
화학식 8
Formula 8
상기 화학식 5 내지 화학식 8에서,In Chemical Formulas 5 to 8,
Ar1, T 및 n은 각각 상기 화학식 1 내지 화학식 16의 Ar1, T 및 n에서 설명된 바와 같고,Ar 1, T and n are as described in each of Ar 1, T, and n in the above Chemical Formulas 1 to 16,
Ar1'는 동일하거나 서로 상이하며 각각 독립적으로 치환 또는 비치환된 2가의 C6 내지 C60 아릴렌기 및 치환 또는 비치환된 2가의 C4 내지 C60 헤테로 고리기에서 선택되는 방향족 고리기이고, 상기 방향족 고리기는 단독으로 존재하거나; 2개 이상이 서로 접합되어 축합 고리를 형성하거나; 2개 이상이 단일결합, O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, C(=O)NH 또는 또는 치환 또는 비치환된 4가의C1 내지 C30 지방족 유기기에 의해 연결되어 있고,Ar 1 ′ is the same or different from each other and each independently an aromatic ring group selected from a substituted or unsubstituted divalent C6 to C60 arylene group and a substituted or unsubstituted divalent C4 to C60 heterocyclic group, wherein the aromatic ring group Exist alone; Two or more are joined to each other to form a condensed ring; Two or more single bonds, O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where 1 ≦ p ≦ 10) , (CF 2 ) q where 1 ≦ q ≦ 10, C (CH 3 ) 2 , C (CF 3 ) 2 , C (═O) NH or substituted or unsubstituted tetravalent C 1 to C 30 aliphatic organic Connected by groups,
Y''는 O 또는 S 이다.Y '' is O or S.
상기 Ar1, 및 T 의 예, 그리고 구체예에 대한 설명은 상술한 바와 같다.Examples of Ar 1 and T and descriptions of specific examples are as described above.
상기 Ar1'는 하기 식으로 표시된 것 중에서 선택될 수 있다.Ar 1 ′ may be selected from those represented by the following formulas.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
X1 내지 X6은 동일하거나 서로 상이하며 각각 독립적으로 O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, 또는 C(=O)NH이고,X 1 to X 6 are the same or different from each other and each independently O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where , 1 ≦ p ≦ 10), (CF 2 ) q (where 1 ≦ q ≦ 10), C (CH 3 ) 2 , C (CF 3 ) 2 , or C (═O) NH,
W1 및 W2는 동일하거나 서로 상이하며 각각 독립적으로 O, S, 또는 C(=O)이고,W 1 and W 2 are the same or different and are each independently O, S, or C (═O),
Z1은 O, S, CR300R301 또는 NR302이고, 여기서 R300, R301 및 R302는 동일하거나 서로 상이하며 각각 독립적으로 수소 또는 C1 내지 C5 알킬기이고,Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
Z2 및 Z3는 동일하거나 서로 상이하며 각각 독립적으로 N 또는 CR303(여기서, R303은 수소 또는 C1 내지 C5 알킬기이다)이나 동시에 CR303은 아니고,Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
T는 각각 독립적으로 치환 또는 비치환된 4가의 C1 내지 C40 지방족 유기기, 치환 또는 비치환된 4가의 C3 내지 C40 지환족 유기기, 또는 치환 또는 비치환된 4가의 C6 내지 C40 방향족 유기기이고,T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
R1 내지 R62는 동일하거나 서로 상이하며 각각 독립적으로 수소, 또는 치환 또는 비치환된 C1 내지 C10 지방족 유기기, 또는 금속 술포네이트기이고,R 1 to R 62 are the same or different and are each independently hydrogen or a substituted or unsubstituted C1 to C10 aliphatic organic group, or a metal sulfonate group,
k63, k69, k84 내지 k88, k92 내지 k96, k102 내지 k109, k116 및 k119는 0 내지 4의 정수이고,k63, k69, k84 to k88, k92 to k96, k102 to k109, k116 and k119 are integers from 0 to 4,
k64 내지 k66, k68, k71, k74, k75, k77, k78, k81, k83, k110 내지 k112, k115, k117, k118, k120 및 k123은 0 내지 3의 정수이고,k64 to k66, k68, k71, k74, k75, k77, k78, k81, k83, k110 to k112, k115, k117, k118, k120 and k123 are integers from 0 to 3,
k67, k72, k73, k76, k79, k80, k82, k90, k98, k100, k101, k113, k114, k121 및 k122는 0 내지 2의 정수이고,k67, k72, k73, k76, k79, k80, k82, k90, k98, k100, k101, k113, k114, k121 and k122 are integers from 0 to 2,
k70은 0 또는 1의 정수이고,k70 is an integer of 0 or 1,
k89, k91, k97 및 k99는 0 내지 5의 정수이다.k89, k91, k97 and k99 are integers of 0-5.
전술한 바와 같이, 상기 Ar1'에서의 T 역시 상기 Ar1에서 T에 대해 정의한 바와 같고, 그 예 및 구체적인 예 또한 Ar1에서 T에 대해 정의한 것과 같다.As described above, T in Ar 1 ′ is also the same as defined for T in Ar 1 , and examples and specific examples are also the same as those defined in T in Ar 1 .
구체적으로는 상기 Ar1' 는 하기 식으로 표시된 것 중에서 선택될 수 있다.Specifically, Ar 1 ′ may be selected from those represented by the following formulas.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
M은 수소 또는 금속이고, 상기 금속은 나트륨, 칼륨, 리튬, 이들의 합금 또는 이들의 조합이다.M is hydrogen or a metal, said metal being sodium, potassium, lithium, alloys thereof or a combination thereof.
상기 고분자는 약 10,000 g/mol 내지 약 500,000 g/mol의 중량평균 분자량(Mw)을 가질 수 있다.The polymer may have a weight average molecular weight (Mw) of about 10,000 g / mol to about 500,000 g / mol.
본 발명의 다른 일 구현예에 따른 고분자의 제조 방법은 상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리아믹산을 이미드화하여 폴리이미드를 얻는 단계; 및 상기 폴리이미드를 열처리하는 단계를 포함한다.According to another aspect of the present invention, there is provided a method of preparing a polymer, wherein a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof is imidized to form a poly Obtaining a mead; And heat treating the polyimide.
상기 열처리는 약 1 ℃/min 내지 약 30 ℃/min의 승온 속도로 약 350℃ 내지 약 500℃까지 승온하고, 그 온도로 비활성 분위기 하에서 약 1분 내지 약 12시간 동안 수행할 수 있다.The heat treatment may be performed at a temperature increase rate of about 1 ° C / min to about 30 ° C / min to about 350 ° C to about 500 ° C, and at that temperature under an inert atmosphere for about 1 minute to about 12 hours.
본 발명의 또 다른 일 구현예에 따른 고분자의 제조 방법은 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리이미드를 열처리하는 단계를 포함한다.Method for producing a polymer according to another embodiment of the present invention is a step of heat-treating a polyimide comprising a repeating unit represented by the formula (3) and formula (4), a copolymer thereof, or a blend thereof It includes.
상기 열처리에 대한 설명은 상술한 바와 같다.Description of the heat treatment is as described above.
본 발명의 또 다른 일 구현예에 따른 고분자의 제조 방법은 상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리아믹산, 그리고 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리이미드의 조합을 포함하는 화합물 중 폴리아믹산을 이미드화하여 폴리이미드를 얻는 단계; 및 상기 폴리이미드를 열처리하는 단계를 포함한다.Method for producing a polymer according to another embodiment of the present invention is a polyamic acid comprising a repeating unit represented by the formula (1) and formula (2), a copolymer thereof, or a polyamic acid comprising a blend thereof, and the formula Imidizing a polyamic acid in a compound including a polyimide comprising a repeating unit represented by Formula 3 and Formula 4, a copolymer thereof, or a polyimide including a blend thereof to obtain a polyimide; And heat treating the polyimide.
상기 열처리에 대한 설명은 상술한 바와 같다.Description of the heat treatment is as described above.
본 발명의 또 다른 일 측면은 상기 고분자를 포함하는 성형품을 제공한다.Another aspect of the invention provides a molded article comprising the polymer.
상기 성형품은 기체 분리막일 수 있다.The molded article may be a gas separation membrane.
기타 본 발명의 측면들의 구체적인 사항은 이하의 상세한 설명에 포함되어 있다.Other details of aspects of the invention are included in the following detailed description.
본 발명에 따른 고분자는 저분자의 투과도 및 선택도가 우수하고, 내열성, 내화학성,용매에의 가용성이 우수하며 기계적 강도 또한 우수하다.The polymer according to the present invention is excellent in permeability and selectivity of low molecules, excellent in heat resistance, chemical resistance, solubility in solvents, and also excellent in mechanical strength.
이하, 본 발명의 구현예를 상세히 설명하기로 한다. 다만, 이는 예시로서 제시되는 것으로, 이에 의해 본 발명이 제한되지는 않으며 본 발명은 후술할 청구항의 범주에 의해 정의될 뿐이다.Hereinafter, embodiments of the present invention will be described in detail. However, this is presented as an example, by which the present invention is not limited and the present invention is defined only by the scope of the claims to be described later.
본 명세서에서 별도의 정의가 없는 한, "치환" 또는 "치환된"이란 화합물 또는 작용기 중의 수소 원자가 C1 내지 C10 알킬기, C1 내지 C10 알콕시기, C1 내지 C10 할로알킬기, C1 내지 C10 할로알콕시기 및 C6 내지 C20 방향족 유기기로 이루어진 군에서 선택되는 1종 이상의 치환기로 치환된 것을 의미한다.Unless otherwise defined herein, the term "substituted" or "substituted" means that the hydrogen atom in the compound or functional group is a C1 to C10 alkyl group, a C1 to C10 alkoxy group, a C1 to C10 haloalkyl group, a C1 to C10 haloalkoxy group and a C6 group. To C20 is substituted with one or more substituents selected from the group consisting of aromatic organic groups.
본 명세서에서 별도의 정의가 없는 한, "헤테로 고리기"란 O, S, N, P, Si 및 이들의 조합으로 이루어진 군에서 선택되는 헤테로 원자를 하나의 고리 내에 1 내지 3 개 함유하는 치환 또는 비치환된 C2 내지 C30의 사이클로알킬기, 치환 또는 비치환된 C2 내지 C30의 사이클로알케닐기, 치환 또는 비치환된 C2 내지 C30의 사이클로알키닐기, 또는 치환 또는 비치환된 C2 내지 C30 헤테로아릴기를 의미한다.Unless otherwise defined herein, a "hetero ring group" means a substitution containing 1 to 3 heteroatoms selected from the group consisting of O, S, N, P, Si, and combinations thereof in one ring, or Unsubstituted C2 to C30 cycloalkyl group, substituted or unsubstituted C2 to C30 cycloalkenyl group, substituted or unsubstituted C2 to C30 cycloalkynyl group, or substituted or unsubstituted C2 to C30 heteroaryl group. .
본 명세서에서 별도의 정의가 없는 한, "지방족 유기기"란 C1 내지 C30 알킬기, C2 내지 C30 알케닐기, C2 내지 C30 알키닐기, C1 내지 C30 알킬렌기, C2 내지 C30 알케닐렌기, 또는 C2 내지 C30 알키닐렌기를 의미하고, 구체적으로는 C1 내지 C15 알킬기, C2 내지 C15 알케닐기, C2 내지 C15 알키닐기, C1 내지 C15 알킬렌기, C2 내지 C15 알케닐렌기, 또는 C2 내지 C15 알키닐렌기를 의미하고, "지환족 유기기"란 C3 내지 C30 사이클로알킬기, C3 내지 C30 사이클로알케닐기, C3 내지 C30 사이클로알키닐기, C3 내지 C30 사이클로알킬렌기, C3 내지 C30 사이클로알케닐렌기, 또는 C3 내지 C30 사이클로알키닐렌기를 의미하고, 구체적으로는 C3 내지 C15 사이클로알킬기, C3 내지 C15 사이클로알케닐기, C3 내지 C15 사이클로알키닐기, C3 내지 C15 사이클로알킬렌기, C3 내지 C15 사이클로알케닐렌기, 또는 C3 내지 C15 사이클로알키닐렌기를 의미하고, "방향족 유기기"란 C6 내지 C30 아릴기 또는 C6 내지 C30 아릴렌기를 의미하고, 구체적으로는 C6 내지 C16 아릴기 또는 C6 내지 C16 아릴렌기를 의미한다.Unless otherwise defined herein, an "aliphatic organic group" means a C1 to C30 alkyl group, a C2 to C30 alkenyl group, a C2 to C30 alkynyl group, a C1 to C30 alkylene group, a C2 to C30 alkenylene group, or a C2 to C30 group. An alkynylene group, and specifically, a C1 to C15 alkyl group, a C2 to C15 alkenyl group, a C2 to C15 alkynyl group, a C1 to C15 alkylene group, a C2 to C15 alkenylene group, or a C2 to C15 alkynylene group, and Alicyclic organic group "means a C3 to C30 cycloalkyl group, a C3 to C30 cycloalkenyl group, a C3 to C30 cycloalkynyl group, a C3 to C30 cycloalkylene group, a C3 to C30 cycloalkenylene group, or a C3 to C30 cycloalkynylene group Specifically, C3 to C15 cycloalkyl group, C3 to C15 cycloalkenyl group, C3 to C15 cycloalkynyl group, C3 to C15 cycloalkylene group, C3 to C15 cycloalkenylene group, or By C3 to C15 cycloalkynylene group, "aromatic organic group" means a C6 to C30 aryl group or a C6 to C30 arylene group, and specifically means a C6 to C16 aryl group or a C6 to C16 arylene group.
본 명세서에서 별도의 정의가 없는 한, "조합"이란 혼합 또는 공중합을 의미한다. 또한 "공중합"이란 블록 공중합 내지 랜덤 공중합을 의미하고, "공중합체"란 블록 공중합체 내지 랜덤 공중합체를 의미한다.Unless otherwise defined herein, “combination” means mixed or copolymerized. In addition, "copolymerization" means block copolymerization to random copolymerization, and "copolymer" means block copolymer to random copolymerization.
또한 본 명세서에서 "*"는 동일하거나 상이한 원자 또는 화학식과 연결되는 부분을 의미한다.In addition, in this specification, "*" means the part connected with the same or different atom or formula.
본 발명의 일 구현예에 따른 고분자는 하기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리아믹산; 하기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리이미드; 또는 이들의 조합으로부터 유도된다.A polymer according to an embodiment of the present invention includes a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof; A polyimide comprising a repeating unit represented by Formulas 3 and 4, a copolymer thereof, or a blend thereof; Or combinations thereof.
[화학식 1][Formula 1]
[규칙 제91조에 의한 정정 17.06.2013]
[화학식 2][Formula 2]
[규칙 제91조에 의한 정정 17.06.2013]
[화학식 3][Formula 3]
[규칙 제91조에 의한 정정 17.06.2013]
[화학식 4][Formula 4]
[규칙 제91조에 의한 정정 17.06.2013]
상기 화학식 1 내지 화학식 4에서,In Chemical Formulas 1 to 4,
Ar1은 각각의 반복단위에서 동일하거나 서로 상이하며 각각 독립적으로 치환 또는 비치환된 4가의 C6 내지 C60 아릴렌기 및 치환 또는 비치환된 4가의 C4 내지 C60 헤테로 고리기에서 선택되는 방향족 고리기이고, 상기 방향족 고리기는 단독으로 존재하거나; 2개 이상이 서로 접합되어 축합 고리를 형성하거나; 2개 이상이 단일결합, O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, C(=O)NH 또는 치환 또는 비치환된 4가의C1 내지 C30 지방족 유기기에 의해 연결되어 있고,Ar 1 is the same as or different from each other at each repeating unit, and each independently an aromatic ring group selected from a substituted or unsubstituted tetravalent C6 to C60 arylene group and a substituted or unsubstituted tetravalent C4 to C60 heterocyclic group, The aromatic ring groups are present alone; Two or more are joined to each other to form a condensed ring; Two or more single bonds, O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where 1 ≦ p ≦ 10) , (CF 2 ) q (where 1 ≦ q ≦ 10), C (CH 3 ) 2 , C (CF 3 ) 2 , C (═O) NH or a substituted or unsubstituted tetravalent C 1 to C 30 aliphatic organic group Connected by
T는 각각의 반복단위에서 동일하거나 서로 상이하며 각각 독립적으로 치환 또는 비치환된 4가의 C1 내지 C40 지방족 유기기, 치환 또는 비치환된 4가의 C3 내지 C40 지환족 유기기, 또는 치환 또는 비치환된 4가의 C6 내지 C40 방향족 유기기이고,T is the same or different at each repeating unit and is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted group Tetravalent C6 to C40 aromatic organic group,
Y는 각각의 반복단위에서 동일하거나 서로 상이하며 각각 독립적으로 OH, SH 또는 NH2이고,Y is the same or different at each repeating unit and each independently is OH, SH or NH 2 ,
n은 10≤n≤400을 만족하는 정수이다.n is an integer satisfying 10 ≦ n ≦ 400.
상기 화학식 1 및 2로 표시되는 반복단위를 포함하는 폴리아믹산 및 상기 화학식 3 및 4로 표시되는 반복단위를 포함하는 폴리이미드는 일반적인 방법에 따라 제조할 수 있다. 일 예로, 상기 폴리아믹산은 아민기에 대하여 오르쏘 위치에 존재하는 OH, SH 또는 NH2기를 포함하는 디아민과 테트라카르복실산 무수물을 반응시켜 제조할 수 있다. 또한 상기 폴리이미드는 상기와 같이 제조한 폴리아믹산을 열적 용액 이미드화 또는 화학적 이미드화함으로써 제조할 수 있다. 상기 열적 용액 이미드화 및 화학적 이미드화에 대하여는 후술한다.The polyamic acid including the repeating units represented by Formulas 1 and 2 and the polyimide including the repeating units represented by Formulas 3 and 4 may be prepared according to a general method. For example, the polyamic acid may be prepared by reacting a tetraamine anhydride with a diamine including an OH, SH, or NH 2 group present at an ortho position relative to an amine group. In addition, the polyimide can be prepared by thermal solution imidization or chemical imidization of the polyamic acid prepared as described above. The thermal solution imidization and chemical imidization will be described later.
상기 폴리아믹산은 후술할 제조공정을 통해 이미드화 및 열전환되고, 상기 폴리이미드는 후술할 제조공정을 통해 열전환되어, 높은 자유 체적도를 갖는 폴리벤조옥사졸, 폴리벤조티아졸, 폴리피롤론 또는 이들의 조합을 포함하는 고분자로 전환될 수 있다.The polyamic acid is imidized and thermally converted through a manufacturing process to be described later, and the polyimide is thermally converted through a manufacturing process to be described later, and has a high degree of free polybenzoxazole, polybenzothiazole, polypyrrolone or It can be converted into a polymer comprising a combination of these.
상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자는 유기 용매에의 용해성이 우수하고 유연한 구조(flexible structure)를 포함하고, 동시에 상기 유연한 구조를 사이에 두고 연결되는 강직한 구조(rigid structure), 구체적으로는 강직한 사다리형 구조를 포함한다. 이에 따라 상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자는 강직한 사다리형 구조가 유연한 경첩으로 연결되는 형태로 반복되는 구조를 포함함으로써, 굴절 가능한 계단이 무정형으로 분포되면서 형성되는 미세 기공을 가질 수 있으며, 또한 기계적 강도 및 가공성이 우수하므로 필름, 섬유, 중공사 등의 형태로 제조하는데 용이하게 사용될 수 있다.The polymer derived from the polyamic acid and the polymer derived from the polyimide include a rigid structure having excellent solubility in an organic solvent and a flexible structure, and at the same time, a rigid structure connected between the flexible structures. , Specifically, a rigid ladder structure. Accordingly, the polymer derived from the polyamic acid and the polymer derived from the polyimide include a repeating structure in which a rigid ladder structure is connected by a flexible hinge, and thus fine pores are formed while the refractive staircase is amorphously distributed. It may have, and also excellent mechanical strength and workability can be easily used to manufacture in the form of film, fiber, hollow fiber and the like.
상기 폴리아믹산으로부터 유도되는 고분자 및 상기 폴리이미드로부터 유도되는 고분자는 약 0.20 내지 약 0.35의 자유 체적도(FFV)를 가질 수 있고, XRD 측정에 의한 면간 거리(d-spacing)가 약 520 pm 내지 약 850 pm의 범위에 있을 수 있다. 이로써 상기 폴리아믹산으로부터 유도되는 고분자 및 상기 폴리이미드로부터 유도되는 고분자는 저분자를 용이하게 투과 내지 분리할 수 있다. The polymer derived from the polyamic acid and the polymer derived from the polyimide may have a free volume (FFV) of about 0.20 to about 0.35, and the d-spacing by XRD is about 520 pm to about It can be in the range of 850 pm. As a result, the polymer derived from the polyamic acid and the polymer derived from the polyimide can easily permeate or separate low molecules.
상기 폴리아믹산으로부터 유도되는 고분자 및 상기 폴리이미드로부터 유도되는 고분자는 약 280 m2/g 내지 약 600 m2/g 의 BET(Brunauer-Emmett-Teller) 표면적을 가질 수 있다. 따라서 상기 폴리아믹산으로부터 유도되는 고분자 및 상기 폴리이미드로부터 유도되는 고분자는 효율적으로 저분자를 투과시키거나 선택적으로 분리할 수 있다.The polymer derived from the polyamic acid and the polymer derived from the polyimide may have a Brunauer-Emmett-Teller (BET) surface area of about 280 m 2 / g to about 600 m 2 / g. Therefore, the polymer derived from the polyamic acid and the polymer derived from the polyimide can efficiently permeate or selectively separate low molecules.
상기 화학식 1 내지 화학식 4에서, Ar1의 예는 하기 식으로 표시된 것 중에서 선택될 수 있으나, 이에 한정되는 것은 아니다.In Chemical Formulas 1 to 4, examples of Ar 1 may be selected from those represented by the following formulas, but are not limited thereto.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
X1 내지 X6은 동일하거나 서로 상이하며 각각 독립적으로 O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, 또는 C(=O)NH이고,X 1 to X 6 are the same or different from each other and each independently O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where , 1 ≦ p ≦ 10), (CF 2 ) q (where 1 ≦ q ≦ 10), C (CH 3 ) 2 , C (CF 3 ) 2 , or C (═O) NH,
W1 및 W2는 동일하거나 서로 상이하며 각각 독립적으로 O, S, 또는 C(=O)이고,W 1 and W 2 are the same or different and are each independently O, S, or C (═O),
Z1은 O, S, CR300R301 또는 NR302이고, 여기서 R300, R301 및 R302는 동일하거나 서로 상이하며 각각 독립적으로 수소 또는 C1 내지 C5 알킬기이고,Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
Z2 및 Z3는 동일하거나 서로 상이하며 각각 독립적으로 N 또는 CR303(여기서, R303은 수소 또는 C1 내지 C5 알킬기이다)이나 동시에 CR303은 아니고,Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
T는 각각 독립적으로 치환 또는 비치환된 4가의 C1 내지 C40 지방족 유기기, 치환 또는 비치환된 4가의 C3 내지 C40 지환족 유기기, 또는 치환 또는 비치환된 4가의 C6 내지 C40 방향족 유기기이고,T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
R1 내지 R62는 동일하거나 서로 상이하며 각각 독립적으로 수소, 또는 치환 또는 비치환된 C1 내지 C10 지방족 유기기이고,R 1 to R 62 are the same or different and are each independently hydrogen or a substituted or unsubstituted C1 to C10 aliphatic organic group,
k1 내지 k3, k8 내지 k14, k24, k25, k49 내지 k54 및 k59 내지 k62는 0 내지 2의 정수이고,k1 to k3, k8 to k14, k24, k25, k49 to k54 and k59 to k62 are integers of 0 to 2,
k5, k15, k16, k19, k21 및 k23은 0 또는 1의 정수이고,k5, k15, k16, k19, k21 and k23 are integers of 0 or 1,
k4, k6, k7, k17, k18, k20, k22, k26 내지 k29, k31, k34 내지 k36, k38, k41, k44 내지 k46 및 k55 내지 k58은 0 내지 3의 정수이고,k4, k6, k7, k17, k18, k20, k22, k26 to k29, k31, k34 to k36, k38, k41, k44 to k46 and k55 to k58 are integers from 0 to 3,
k30, k37, k42, k43, k47 및 k48은 0 내지 4의 정수이고,k30, k37, k42, k43, k47 and k48 are integers from 0 to 4,
k32, k33, k39 및 k40은 0 내지 5의 정수이다.k32, k33, k39 and k40 are integers from 0 to 5.
상기 T의 예는 하기 식으로 표시된 것 중에서 선택될 수 있으나, 이에 한정되는 것은 아니다.Examples of the T may be selected from those represented by the following formula, but are not limited thereto.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
R200 내지 R231은 동일하거나 서로 상이하며 각각 독립적으로 수소, 치환 또는 비치환된 C1 내지 C30 지방족 유기기, 치환 또는 비치환된 C3 내지 C30 지환족 유기기, 또는 치환 또는 비치환된 2가의 C6 내지 C30 방향족 유기기이고,R 200 to R 231 are the same or different from each other and are each independently hydrogen, a substituted or unsubstituted C1 to C30 aliphatic organic group, a substituted or unsubstituted C3 to C30 alicyclic organic group, or a substituted or unsubstituted divalent C6 To C30 aromatic organic group,
t1 내지 t12는 동일하거나 서로 상이하며 각각 독립적으로 0 내지 4의 정수이다.t1 to t12 are the same or different and are each independently an integer of 0 to 4;
구체적으로는 상기 T의 예는 하기 식으로 표시된 것 중에서 선택될 수 있으나, 이에 한정되는 것은 아니다.Specifically, the example of T may be selected from those represented by the following formula, but is not limited thereto.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 화학식 1 내지 화학식 4에서, Ar1의 구체적인 예는 하기 식으로 표시된 것 중에서 선택될 수 있으나, 이에 한정되는 것은 아니다.In Chemical Formulas 1 to 4, specific examples of Ar 1 may be selected from those represented by the following formulas, but are not limited thereto.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 화학식 1 내지 화학식 4에서, T의 예 및 구체적인 예는 상기 Ar1을 설명할 때 기재한 T의 예 및 구체적인 예로 언급된 것과 동일하다.In Formulas 1 to 4, examples and specific examples of T are the same as those mentioned in Examples and specific examples of T described when describing Ar 1 .
상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산의 공중합체에서의 각 반복단위 사이의 몰비; 또는 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드의 공중합체에서의 각 반복단위 사이의 몰비를 조절하여, 제조된 고분자의 물성 제어가 가능하다.Molar ratio between each repeating unit in the copolymer of polyamic acid including the repeating units represented by Formula 1 and Formula 2; Alternatively, by controlling the molar ratio between the repeating units in the copolymer of the polyimide including the repeating units represented by Formulas 3 and 4, it is possible to control the physical properties of the prepared polymer.
상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산의 공중합체에서의 각 반복단위 사이의 몰비는 약 0.1:9.9 내지 약 9.9:0.1, 구체적으로는 약 2:8 내지 약 8:2, 더욱 구체적으로는 약 5:5로 조절할 수 있다.The molar ratio between each repeating unit in the copolymer of polyamic acid including the repeating units represented by Formula 1 and Formula 2 is about 0.1: 9.9 to about 9.9: 0.1, specifically about 2: 8 to about 8: 2 More specifically, about 5: 5.
또한 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드의 공중합체에서의 각 반복단위 사이의 몰비는 약 0.1:9.9 내지 약 9.9:0.1, 구체적으로는 약 2:8 내지 약 8:2, 더욱 구체적으로는 약 5:5로 조절할 수 있다.In addition, the molar ratio between each repeating unit in the copolymer of the polyimide including the repeating units represented by Formula 3 and Formula 4 is about 0.1: 9.9 to about 9.9: 0.1, specifically about 2: 8 to about 8: 2, more specifically, about 5: 5.
이러한 공중합비는 제조된 고분자의 분자구조 배열 및 모폴로지에 영향을 주는데, 이러한 분자구조 배열 및 모폴로지 변화는 밀도, 기공 특성, 내열성, 인장 강도, 전단력 등과 관련되어 있다. 상기 몰비 내지 공중합비가 상기 범위 내인 경우, 제조된 고분자는 저분자를 효율적으로 투과시키거나 선택적으로 분리할 수 있고, 우수한 내열성, 내화학성, 기계적 강도 및 가공성을 가질 수 있다.The copolymerization ratio affects the molecular structure and morphology of the prepared polymer, which is related to density, pore properties, heat resistance, tensile strength, shear force, and the like. When the molar ratio to the copolymerization ratio is within the above range, the prepared polymer may efficiently permeate or selectively separate low molecules, and may have excellent heat resistance, chemical resistance, mechanical strength and processability.
상기 폴리아믹산으로부터 유도되는 고분자 및 상기 폴리이미드로부터 유도되는 고분자는 하기 화학식 5 내지 화학식 8 중 어느 하나로 표시되는 반복단위를 포함하는 화합물 또는 이들의 공중합체를 포함할 수 있으나, 이에 한정되는 것은 아니다.The polymer derived from the polyamic acid and the polymer derived from the polyimide may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
[화학식 5][Formula 5]
[규칙 제91조에 의한 정정 17.06.2013]
[화학식 6][Formula 6]
[규칙 제91조에 의한 정정 17.06.2013]
[화학식 7][Formula 7]
[규칙 제91조에 의한 정정 17.06.2013]
[화학식 8][Formula 8]
[규칙 제91조에 의한 정정 17.06.2013]
상기 화학식 5 내지 화학식 8에서, Ar1, T 및 n은, 각각 상기 화학식 1 내지 화학식 4의 Ar1, T 및 n에 대해 설명된 바와 같고,In Formulas 5 to 8, Ar 1 , T and n are as described for Ar 1 , T and n of Formulas 1 to 4, respectively,
Ar1'는 동일하거나 서로 상이하며, 각각 독립적으로 치환 또는 비치환된 2가의 C6 내지 C60 아릴렌기, 및 치환 또는 비치환된 2가의 C4 내지 C60 헤테로 고리기에서 선택되는 방향족 고리기이고, 상기 방향족 고리기는 단독으로 존재하거나; 2개 이상이 서로 접합되어 축합 고리를 형성하거나; 2개 이상이 단일결합, O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, C(=O)NH 또는 또는 치환 또는 비치환된 4가의C1 내지 C30 지방족 유기기에 의해 연결되어 있고,Ar 1 ′ is the same or different from each other, and is each independently an aromatic ring group selected from a substituted or unsubstituted divalent C6 to C60 arylene group, and a substituted or unsubstituted divalent C4 to C60 heterocyclic group, and the aromatic The ring group is present alone; Two or more are joined to each other to form a condensed ring; Two or more single bonds, O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where 1 ≦ p ≦ 10) , (CF 2 ) q where 1 ≦ q ≦ 10, C (CH 3 ) 2 , C (CF 3 ) 2 , C (═O) NH or substituted or unsubstituted tetravalent C 1 to C 30 aliphatic organic Connected by groups,
Y''는 O 또는 S 이다.Y '' is O or S.
상기 화학식 5 내지 화학식 8에서, Ar1, 및 T의 예, 그리고 이들에 대한 구체적인 예는 각각 상기 화학식 1 내지 화학식 4의 Ar1 및 T의 예 및 구체적인 예로 언급된 것과 동일하다.In the above Chemical Formulas 5 to 8, examples of Ar 1, and T, and specific examples of them are the same as each of the formulas (1) to the examples and mentioned specific examples of Ar 1 and T in the formula (4).
상기 화학식 5 내지 화학식 8에서, Ar1'의 예는 하기 식으로 표시된 것 중에서 선택될 수 있으나, 이에 한정되는 것은 아니다.In Formulas 5 to 8, an example of Ar 1 ' may be selected from those represented by the following formulas, but is not limited thereto.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
X1 내지 X6은 동일하거나 서로 상이하며 각각 독립적으로 O, S, C(=O), CH(OH), S(=O)2, Si(CH3)2, (CH2)p(여기서, 1≤p≤10), (CF2)q(여기서, 1≤q≤10), C(CH3)2, C(CF3)2, 또는 C(=O)NH이고,X 1 to X 6 are the same or different from each other and each independently O, S, C (= 0), CH (OH), S (= 0) 2 , Si (CH 3 ) 2 , (CH 2 ) p (where , 1 ≦ p ≦ 10), (CF 2 ) q (where 1 ≦ q ≦ 10), C (CH 3 ) 2 , C (CF 3 ) 2 , or C (═O) NH,
W1 및 W2는 동일하거나 서로 상이하며 각각 독립적으로 O, S, 또는 C(=O)이고,W 1 and W 2 are the same or different and are each independently O, S, or C (═O),
Z1은 O, S, CR300R301 또는 NR302이고, 여기서 R300, R301 및 R302는 동일하거나 서로 상이하며 각각 독립적으로 수소 또는 C1 내지 C5 알킬기이고,Z 1 is O, S, CR 300 R 301 or NR 302 , wherein R 300 , R 301 and R 302 are the same or different from each other and are each independently hydrogen or a C1 to C5 alkyl group,
Z2 및 Z3는 동일하거나 서로 상이하며 각각 독립적으로 N 또는 CR303(여기서, R303은 수소 또는 C1 내지 C5 알킬기이다)이나 동시에 CR303은 아니고,Z 2 and Z 3 are the same or different from each other and independently of each other N or CR 303 (wherein R 303 is hydrogen or a C1 to C5 alkyl group) but not CR 303 at the same time,
T는 각각 독립적으로 치환 또는 비치환된 4가의 C1 내지 C40 지방족 유기기, 치환 또는 비치환된 4가의 C3 내지 C40 지환족 유기기, 또는 치환 또는 비치환된 4가의 C6 내지 C40 방향족 유기기이고,T is each independently a substituted or unsubstituted tetravalent C1 to C40 aliphatic organic group, a substituted or unsubstituted tetravalent C3 to C40 alicyclic organic group, or a substituted or unsubstituted tetravalent C6 to C40 aromatic organic group,
R63 내지 R123은 동일하거나 서로 상이하며 각각 독립적으로 수소, 치환 또는 비치환된 C1 내지 C10 지방족 유기기, 또는 금속 술포네이트기이고,R 63 to R 123 are the same or different and are each independently hydrogen, a substituted or unsubstituted C 1 to C 10 aliphatic organic group, or a metal sulfonate group,
k63, k69, k84 내지 k88, k92 내지 k96, k102 내지 k109, k116 및 k119는 0 내지 4의 정수이고,k63, k69, k84 to k88, k92 to k96, k102 to k109, k116 and k119 are integers from 0 to 4,
k64 내지 k66, k68, k71, k74, k75, k77, k78, k81, k83, k110 내지 k112, k115, k117, k118, k120 및 k123은 0 내지 3의 정수이고,k64 to k66, k68, k71, k74, k75, k77, k78, k81, k83, k110 to k112, k115, k117, k118, k120 and k123 are integers from 0 to 3,
k67, k72, k73, k76, k79, k80, k82, k90, k98, k100, k101, k113, k114, k121 및 k122는 0 내지 2의 정수이고,k67, k72, k73, k76, k79, k80, k82, k90, k98, k100, k101, k113, k114, k121 and k122 are integers from 0 to 2,
k70은 0 또는 1의 정수이고,k70 is an integer of 0 or 1,
k89, k91, k97 및 k99는 0 내지 5의 정수이다.k89, k91, k97 and k99 are integers of 0-5.
구체적으로는 상기 Ar1'의 예는 하기 식으로 표시된 것 중에서 선택될 수 있으나, 이에 한정되는 것은 아니다.Specifically, the example of Ar 1 ' may be selected from those represented by the following formula, but is not limited thereto.
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 식에서,Where
M은 수소 또는 금속이고, 상기 금속은 나트륨, 칼륨, 리튬, 이들의 합금 또는 이들의 조합이다.M is hydrogen or a metal, said metal being sodium, potassium, lithium, alloys thereof or a combination thereof.
본 발명의 일 구현예에 따른 폴리아믹산으로부터 유도된 고분자 및 폴리이미드로부터 유도된 고분자는 일반적인 유기 용매에 가용성인 폴리아믹산 내지 폴리이미드를 이용하여 제조될 수 있고, 상기 고분자는 결함이나 크랙없이 용이하게 코팅될 수 있으므로, 제조 공정을 단순화하여 공정 비용을 감소시킬 수 있고 대면적으로 형성할 수 있다. 또한 상기 고분자는 제조 공정 조건을 조절함으로써 기공 크기 내지 분포를 조절할 수 있다. 이로써 상기 고분자는 기체 투과, 기체 분리, 증기 분리, 물 정제, 흡착제, 내열성 섬유, 박막 제조 분야 등과 같은 다양한 응용 분야에 폭넓게 적용될 수 있다.Polymers derived from polyamic acid and polymers derived from polyimide according to an embodiment of the present invention may be prepared using polyamic acid or polyimide that is soluble in a general organic solvent, and the polymer may be easily formed without defects or cracks. Since it can be coated, it is possible to simplify the manufacturing process to reduce the process cost and to form a large area. In addition, the polymer can control the pore size or distribution by adjusting the manufacturing process conditions. As a result, the polymer may be widely applied to various applications such as gas permeation, gas separation, steam separation, water purification, adsorbent, heat resistant fiber, thin film manufacturing, and the like.
본 발명의 다른 일 구현예에 따르면 고분자는 상기 폴리아믹산 및 상기 폴리이미드의 조합으로부터 유도될 수 있고, 이 경우 상기 고분자는 상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자를 포함할 수 있다. 이하에서 달리 설명하지 않는 한, 상기 폴리아믹산, 상기 폴리이미드, 상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자에 대한 설명은 상술한 바와 같다.According to another embodiment of the present invention, the polymer may be derived from a combination of the polyamic acid and the polyimide, in which case the polymer may include a polymer derived from the polyamic acid and a polymer derived from the polyimide. have. Unless otherwise described below, the description of the polyamic acid, the polyimide, the polymer derived from the polyamic acid and the polymer derived from the polyimide is as described above.
상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자를 포함하는 고분자는 폴리아믹산으로부터 유도된 고분자:폴리이미드로부터 유도된 고분자를 약 0.1:9.9 내지 약 9.9:0.1의 중량비, 구체적으로는 약 8:2 내지 약 2:8의 중량비, 더욱 구체적으로는 약 5:5 중량비로 포함할 수 있다. 이러한 고분자는 상술한 폴리아믹산으로부터 유도된 고분자의 특성 및 폴리이미드로부터 유도된 고분자의 특성을 모두 가질 수 있다. 또한 우수한 치수 안정성 및 장기 안정성을 가질 수 있다.The polymer including the polymer derived from the polyamic acid and the polymer derived from the polyimide may be a polymer having a weight ratio of about 0.1: 9.9 to about 9.9: 0.1 of the polymer: polyimide derived from the polyamic acid. 8: 2 to about 2: 8 by weight, more specifically about 5: 5 by weight. Such a polymer may have both the properties of the polymer derived from the polyamic acid described above and the properties of the polymer derived from polyimide. It can also have good dimensional stability and long term stability.
상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자는 각각 약 10,000 g/mol 내지 약 500,000 g/mol의 중량평균 분자량(Mw)을 가질 수 있다. 이 경우, 상기 폴리아믹산으로부터 유도된 고분자 및 상기 폴리이미드로부터 유도된 고분자는 용이하게 합성될 수 있고, 우수한 기계적 강도 및 치수 안정성을 가질 수 있다.The polymer derived from the polyamic acid and the polymer derived from the polyimide may each have a weight average molecular weight (Mw) of about 10,000 g / mol to about 500,000 g / mol. In this case, the polymer derived from the polyamic acid and the polymer derived from the polyimide can be easily synthesized and have excellent mechanical strength and dimensional stability.
본 발명의 다른 일 구현예는 상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리아믹산을 이미드화하여 폴리이미드를 얻는 단계; 및 상기 폴리이미드를 열처리하는 단계를 포함하는 고분자의 제조 방법을 제공한다. 상기 고분자는 상기 화학식 5 내지 화학식 8 중 어느 하나로 표시되는 반복단위를 포함하는 화합물 또는 이들의 공중합체를 포함할 수 있으나, 이에 한정되는 것은 아니다.Another embodiment of the present invention comprises the steps of imidating a polyamic acid comprising a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a blend thereof, to obtain a polyimide; And it provides a method for producing a polymer comprising the step of heat-treating the polyimide. The polymer may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
상기 고분자의 제조 방법에서, 상기 이미드화는 열적 이미드화 공정으로 이루어질 수 있으나, 이에 한정되는 것은 아니다.In the method of preparing the polymer, the imidization may be performed by a thermal imidization process, but is not limited thereto.
상기 열적 이미드화는 비활성 분위기 하에 약 150℃ 내지 약 300℃에서 약 30 분 내지 약 2시간 동안 수행할 수 있다. 이미드화의 온도가 상기 범위 미만이면 전구체인 폴리아믹산의 이미드화가 미미하고, 이와 반대로 상기 범위를 초과하더라도 효과상의 큰 증가가 없어 비경제적이다.The thermal imidization may be performed at about 150 ° C. to about 300 ° C. for about 30 minutes to about 2 hours under an inert atmosphere. If the temperature of imidation is less than the said range, the imidation of the polyamic acid which is a precursor is insignificant, On the contrary, even if it exceeds the said range, there is no big increase in effect and it is uneconomical.
상기 이미드화의 조건은 상기 폴리아믹산의 작용기인 Ar1, T 및 Y의 종류에 따라 적절히 조절할 수 있다.The imidation condition is Ar, which is a functional group of the polyamic acid.One, It can adjust suitably according to the kind of T and Y.
상기 폴리이미드를 열처리하면, 열전환 반응을 통해 재배열되어 피코기공을 가지는 고분자를 얻을 수 있다. 상기 피코기공을 가지는 고분자는 상기 폴리이미드에 비해 감소된 밀도, 피코 기공이 서로 잘 연결됨에 따라 증가된 자유 체적도 및 증가된 면간 거리를 가진다. 이로써 상기 피코기공을 가지는 고분자는 효율적으로 저분자를 투과시키거나 선택적으로 분리할 수 있다.When the polyimide is heat-treated, it is rearranged through a heat conversion reaction to obtain a polymer having picopores. The polymer having picopores has a reduced density compared to the polyimide, increased free volume and increased interplanar distance as picopores are well connected to each other. As a result, the polymer having the picopores can efficiently permeate or selectively separate low molecules.
또한, 상기 폴리아믹산으로부터 유도된 고분자는 유기 용매에의 용해성이 우수하고 유연한 구조(flexible structure)를 포함하고, 동시에 상기 유연한 구조를 사이에 두고 연결되는 강직한 구조(rigid structure), 구체적으로는 강직한 사다리형 구조를 포함함으로써, 미세 기공을 가질 수 있으며, 또한 가공성이 우수하고 인장강도, 팽창률 등과 같은 기계적 강도를 개선할 수 있다.In addition, the polymer derived from the polyamic acid has a rigid structure, specifically rigid, which has excellent solubility in organic solvents and includes a flexible structure, and at the same time is connected between the flexible structures. By including one ladder structure, it can have fine pores, and can also have excellent workability and improve mechanical strength such as tensile strength, expansion rate, and the like.
상기 열처리는 약 1℃/min 내지 30℃/min의 승온 속도로 약 350℃ 내지 약 500℃까지 승온하고, 그 온도로 비활성 분위기 하에서 약 1분 내지 약 12시간 동안 수행할 수 있다. 구체적으로는 상기 열처리는 약 5 ℃/min 내지 약 20 ℃/min의 승온 속도로 약 350℃ 내지 약 450℃까지 승온하고, 그 온도로 비활성 분위기 하에서 약 1시간 내지 약 6시간 동안 수행할 수 있다. 더욱 구체적으로는 상기 열처리는 약 10 ℃/min 내지 약 15 ℃/min의 승온 속도로 약 420℃ 내지 약 450℃까지 승온하고, 그 온도로 비활성 분위기 하에서 약 2시간 내지 약 5시간 동안 수행할 수 있다. 열처리가 상기 조건 범위 내에서 이루어지는 경우 열전환 반응이 충분히 이루어질 수 있다.The heat treatment may be performed at a temperature rising rate of about 1 ° C / min to 30 ° C / min to about 350 ° C to about 500 ° C, and at that temperature under an inert atmosphere for about 1 minute to about 12 hours. Specifically, the heat treatment may be performed at an elevated temperature rate of about 5 ° C./min to about 20 ° C./min to about 350 ° C. to about 450 ° C., and at that temperature for about 1 hour to about 6 hours under an inert atmosphere. . More specifically, the heat treatment may be performed at an elevated temperature rate of about 10 ° C./min to about 15 ° C./min to about 420 ° C. to about 450 ° C., and at that temperature for about 2 hours to about 5 hours under inert atmosphere. have. When the heat treatment is made within the above condition range, the heat conversion reaction may be sufficiently made.
이러한 열처리를 통해, 상기 화학식 5 내지 화학식 8로 표시되는 반복단위를 포함하는 폴리벤조옥사졸, 폴리벤조티아졸 또는 폴리피롤론 고분자를 얻을 수 있다. 이와 같은 고분자의 제조는 상기 이미드화로 얻어진 폴리이미드 내의 CO2 또는 H2O 제거반응을 통해 이루어진다.Through such heat treatment, a polybenzoxazole, polybenzothiazole or polypyrrolone polymer including a repeating unit represented by Chemical Formulas 5 to 8 may be obtained. Preparation of such a polymer is carried out through the CO 2 or H 2 O removal reaction in the polyimide obtained by the imidization.
본 발명의 또 다른 일 구현예는 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리이미드를 열처리하는 단계를 포함하는 고분자의 제조 방법을 제공한다. 상기 고분자는 상기 화학식 5 내지 화학식 8 중 어느 하나로 표시되는 반복단위를 포함하는 화합물 또는 이들의 공중합체를 포함할 수 있으나, 이에 한정되는 것은 아니다.Another embodiment of the present invention is to prepare a polymer comprising the step of heat-treating a polyimide comprising a repeating unit represented by the formula (3) and formula (4), a copolymer thereof, or a blend thereof Provide a method. The polymer may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
이하에서 달리 설명하지 않는 한, 상기 열처리, 상기 열전환 및 상기 재배열에 대한 설명은 상술한 바와 같다.Unless otherwise described below, the description of the heat treatment, the heat conversion and the rearrangement is as described above.
상기 폴리이미드는, 상기 화학식 1 및 2로 표시되는 반복단위를 포함하는 폴리아믹산을 이미드화, 예컨대 화학적 이미드화 또는 열적 용액 이미드화 함으로써 얻을 수 있다.The polyimide can be obtained by imidizing a polyamic acid including a repeating unit represented by the above formulas (1) and (2), for example, chemical imidation or thermal solution imidation.
상기 화학적 이미드화는 약 20℃ 내지 약 180℃에서 약 4 시간 내지 약 24 시간 동안 반응을 진행시킴으로써 이루어질 수 있다. 이때 촉매로 피리딘과 생성된 물의 제거를 위한 아세틱 무수물을 첨가할 수 있다. 화학적 이미드화의 온도가 상기 범위 이내이면 폴리아믹산의 이미드화가 충분히 이루어질 수 있다.The chemical imidization can be accomplished by running the reaction at about 20 ° C. to about 180 ° C. for about 4 hours to about 24 hours. At this time, as a catalyst, acetic anhydride may be added for removal of pyridine and generated water. If the temperature of the chemical imidization is within the above range, the imidization of the polyamic acid can be sufficiently made.
상기 화학적 이미드화는 먼저 상기 폴리아믹산에서 아민기의 오르쏘 위치에 존재하는 작용기인 OH, SH 및 NH2를 보호한 후, 이루어질 수 있다. 구체적으로는 상기 작용기인 OH, SH 및 NH2에 보호기를 도입하고, 이미드화를 진행한 후, 보호기를 제거하는 방법으로 이루어질 수 있다. 상기 보호기로는 트리메틸클로로실란((CH3)3SiCl), 트리에틸클로로실란((C2H5)3SiCl), 트리부틸클로로실란((C4H9)3SiCl), 트리벤질클로로실란((C6H5)3SiCl), 트리에톡시클로로실란((OC2H5)3SiCl) 등과 같은 클로로실란, 테트라하이드로퓨란(tetrahydrofurane, THF)과 같은 하이드로퓨란을 사용할 수 있고, 이때 염기로서 트리메틸아민, 트리에틸아민, 트리프로필아민, 피리딘 등과 같은 3차 아민을 사용할 수 있다. 또한 상기 보호기를 제거하는 물질로는 희석된 염산, 황산, 질산, 아세트산 등을 사용할 수 있다. 상기와 같이 보호기를 사용한 화학적 이미드화는 본 발명의 일 구현예에 따른 고분자의 수율 및 분자량을 증가시킬 수 있다.The chemical imidization may be performed after first protecting OH, SH and NH 2 , which are functional groups present at the ortho position of the amine group in the polyamic acid. Specifically, a protecting group may be introduced into OH, SH, and NH 2 , which are functional groups, and imidization may be performed to remove the protecting group. The protecting group may be trimethylchlorosilane ((CH 3 ) 3 SiCl), triethylchlorosilane ((C 2 H 5 ) 3 SiCl), tributylchlorosilane ((C 4 H 9 ) 3 SiCl), tribenzylchlorosilane Chlorosilanes such as ((C 6 H 5 ) 3 SiCl), triethoxychlorosilane ((OC 2 H 5 ) 3 SiCl), and hydrofuran, such as tetrahydrofurane (THF), may be used. As such, tertiary amines such as trimethylamine, triethylamine, tripropylamine, pyridine and the like can be used. Diluted hydrochloric acid, sulfuric acid, nitric acid, acetic acid, etc. may be used as the material for removing the protecting group. Chemical imidization using a protecting group as described above may increase the yield and molecular weight of the polymer according to an embodiment of the present invention.
상기 열적 용액 이미드화는 용액 상에서 약 100℃ 내지 약 180℃에서 약 2 시간 내지 약 30 시간 동안 반응을 진행시킴으로써 이루어질 수 있다. 열적 용액 이미드화의 온도가 상기 범위 이내이면 폴리아믹산의 이미드화가 충분히 이루어질 수 있다.The thermal solution imidization can be accomplished by running the reaction on the solution at about 100 ° C. to about 180 ° C. for about 2 hours to about 30 hours. If the temperature of the thermal solution imidization is within the above range, the imidization of the polyamic acid can be sufficiently achieved.
상기 열적 용액 이미드화는 먼저 상기 폴리아믹산에사 아민기의 오르쏘 위치에 존재하는 작용기인 OH, SH 및 NH2를 보호한 후, 이루어질 수 있다. 구체적으로는 상기 작용기인 OH, SH 및 NH2에 보호기를 도입하고, 이미드화를 진행한 후, 보호기를 제거하는 방법으로 이루어질 수 있다. 상기 보호기로는 트리메틸클로로실란, 트리에틸클로로실란, 트리부틸클로로실란, 트리벤질클로로실란, 트리에톡시클로로실란 등과 같은 클로로실란, 테트라하이드로퓨란과 같은 하이드로퓨란을 사용할 수 있고, 이때 염기로서 트리메틸아민, 트리에틸아민, 트리프로필아민, 피리딘 등과 같은 3차 아민을 사용할 수 있다. 상기 보호기를 제거하는 물질로는 희석된 염산, 황산, 질산, 아세트산 등을 사용할 수 있다.The thermal solution imidization may be performed after first protecting OH, SH and NH 2 which are functional groups present at the ortho position of the polyamic acid amine group. Specifically, a protecting group may be introduced into OH, SH, and NH 2 , which are functional groups, and imidization may be performed to remove the protecting group. As the protecting group, chlorosilanes such as trimethylchlorosilane, triethylchlorosilane, tributylchlorosilane, tribenzylchlorosilane, triethoxychlorosilane, and hydrofuran, such as tetrahydrofuran, may be used. Tertiary amines such as triethylamine, tripropylamine, pyridine and the like can be used. Diluted hydrochloric acid, sulfuric acid, nitric acid, acetic acid, etc. may be used as a material for removing the protecting group.
또한 상기 열적 용액 이미드화는 벤젠, 톨루엔, 자일렌, 크레졸 등의 벤젠류, 헥산, 시클로헥산 등의 지방족 유기 용매류 등을 더 첨가하여 이루어진 공비혼합물을 이용하여 이루어질 수 있다.In addition, the thermal solution imidization may be performed using an azeotrope made by further adding benzenes such as benzene, toluene, xylene, cresol, aliphatic organic solvents such as hexane, cyclohexane, and the like.
상기와 같이 보호기를 도입하고, 공비혼합물을 사용함으로써 이루어진 열적 용액 이미드화는 본 발명의 일 구현예에 따른 고분자의 수율 및 분자량을 증가시킬 수 있다.Thermal solution imidization made by introducing a protecting group and using an azeotrope as described above may increase the yield and molecular weight of the polymer according to one embodiment of the present invention.
상기 이미드화의 조건은 상기 폴리아믹산의 작용기인 Ar1, T 및 Y의 종류에 따라 적절히 조절할 수 있다.The imidation condition is Ar, which is a functional group of the polyamic acid.One, It can adjust suitably according to the kind of T and Y.
본 발명의 또 다른 일 구현예는 상기 화학식 1 및 화학식 2로 표시되는 반복단위를 포함하는 폴리아믹산, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리아믹산, 그리고 상기 화학식 3 및 화학식 4로 표시되는 반복단위를 포함하는 폴리이미드, 이들의 공중합체, 또는 이들의 블렌드를 포함하는 폴리이미드의 조합을 포함하는 화합물 중 폴리아믹산을 이미드화하여 폴리이미드를 얻는 단계; 및 상기 폴리이미드를 열처리하는 단계를 포함하는 고분자의 제조 방법을 제공한다. 상기 고분자는 상기 화학식 5 내지 화학식 8 중 어느 하나로 표시되는 반복단위를 포함하는 화합물 또는 이들의 공중합체를 포함할 수 있으나, 이에 한정되는 것은 아니다.Another embodiment of the present invention is a polyamic acid including a repeating unit represented by Formula 1 and Formula 2, a copolymer thereof, or a polyamic acid including a blend thereof, and represented by Formula 3 and Formula 4 Imidizing a polyamic acid in a compound including a polyimide comprising a repeating unit, a copolymer thereof, or a combination of polyimides including a blend thereof to obtain a polyimide; And it provides a method for producing a polymer comprising the step of heat-treating the polyimide. The polymer may include a compound including a repeating unit represented by any one of Formulas 5 to 8 or a copolymer thereof, but is not limited thereto.
이하에서 달리 설명하지 않는 한, 상기 이미드화, 상기 열처리, 상기 열전환 및 상기 재배열에 대한 설명은 상술한 바와 같다.Unless otherwise described below, the description of the imidization, the heat treatment, the heat conversion and the rearrangement is as described above.
상기 고분자의 제조 공정에서, 화학 구조 내 Ar1, T 및 Y의 특성을 고려하여 고분자 설계를 조절함으로써, 기공 크기 내지 분포 등 관련 특성을 제어할 수 있다.In the manufacturing process of the polymer, by adjusting the polymer design in consideration of the properties of Ar 1 , T and Y in the chemical structure, it is possible to control the relevant properties such as pore size or distribution.
본 발명의 일 구현예에 따른 고분자는 고분자 내에 존재하는 강직한 구조를 포함함으로써, 온화한 조건에서뿐만 아니라 긴 작업 시간, 산성 조건 및 고습, 고온과 같은 가혹한 조건하에서도 견딜 수 있다. 즉 본 발명의 일 구현예에 따른 고분자는 화학적 안정성, 내열성 및 기계적 물성이 우수하다.The polymer according to one embodiment of the present invention includes a rigid structure present in the polymer, so that it can withstand not only under mild conditions but also under long working hours, acidic conditions and harsh conditions such as high humidity and high temperature. That is, the polymer according to one embodiment of the present invention has excellent chemical stability, heat resistance, and mechanical properties.
이때 상기 화학식 5 내지 화학식 8로 표시되는 반복단위를 포함하는 고분자 또는 이들의 공중합체는 제조 단계에서 적절한 중량평균 분자량을 갖도록 설계하며, 구체적으로는 중량평균 분자량이 약 10,000 g/mol 내지 약 500,000 g/mol이 되도록 한다. 이들의 중량평균 분자량이 상기 범위 내인 경우, 고분자의 물성이 우수하게 유지될 수 있다.At this time, the polymer comprising a repeating unit represented by Formula 5 to Formula 8 or copolymers thereof are designed to have an appropriate weight average molecular weight in the manufacturing step, specifically, the weight average molecular weight is about 10,000 g / mol to about 500,000 g / mol. If their weight average molecular weight is within the above range, the physical properties of the polymer can be maintained excellent.
본 발명의 일 구현예에 따른 고분자는 폴리아믹산으로부터 유도된 고분자 또는 폴리이미드로부터 유도된 고분자로서, 피코기공을 가진다. 상기 피코기공은 2개 이상이 서로 연결되어 모래시계 모양(hourglass shaped)으로 형성됨으로써, 높은 자유 체적도를 가져 저분자를 효율적으로 투과시키거나 선택적으로 분리할 수 있다. The polymer according to an embodiment of the present invention is a polymer derived from polyamic acid or a polymer derived from polyimide, and has picopores. Since two or more picopores are connected to each other to form an hourglass shape, the picopores may have a high free volume to efficiently penetrate or selectively separate small molecules.
본 발명의 또 다른 일 구현예는 상기 고분자를 포함하는 성형품(article)을 제공한다. 상기 성형품은 시트, 필름, 파우더, 막 또는 파이버(fiber)일 수 있으나, 이에 한정되는 것은 아니다.Yet another embodiment of the present invention provides an article including the polymer. The molded article may be a sheet, a film, a powder, a film, or a fiber, but is not limited thereto.
상기 성형품은 내부에 강직한 사다리형 구조가 유연한 경첩으로 연결되는 형태로 반복되는 구조를 포함함으로써, 굴절 가능한 계단이 무정형으로 분포되면서 형성되는 미세 기공, 구체적으로는 아령 모양의 미세 기공을 가질 수 있다. 따라서 상기 성형품은 저분자를 효율적으로 투과시키거나 선택적으로 분리할 수 있고, 내열성, 표면 경도 및 치수 안정성이 우수하므로, 상기 성능을 필요로 하는 다양한 기술분야에 이용될 수 있다. 구체적으로는 상기 성형품은 기체 분리막으로 사용될 수 있다.The molded article may include a structure in which a rigid ladder structure is repeated in the form of being connected by a flexible hinge, and thus may have micropores, specifically dumbbell-shaped micropores, which are formed while the refractive staircase is amorphous. . Therefore, the molded article can efficiently penetrate or selectively separate low-molecules, and is excellent in heat resistance, surface hardness, and dimensional stability, and thus can be used in various technical fields requiring the above performance. Specifically, the molded article may be used as a gas separation membrane.
이하 본 기재의 실시예 및 비교예를 기재한다. 그러나 하기 실시예는 본 기재의 일 실시예일 뿐이며, 본 기재가 하기 실시예에 의해 한정되는 것은 아니다.Hereinafter, the Example and comparative example of this description are described. However, the following examples are merely examples of the present disclosure, and the present disclosure is not limited by the following examples.
실시예Example : 고분자 합성 및 이를 이용한 기체 분리막의 제조: Synthesis of Polymer and Preparation of Gas Separation Membrane Using the Same
실시예 1Example 1
하기 반응식 1로 표시되는 바에 따라 폴리벤조옥사졸을 포함하는 고분자를 제조하였다.A polymer containing polybenzoxazole was prepared as shown in Scheme 1 below.
[반응식 1]Scheme 1
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
비스페놀 A(bisphenol A) 100g을 메탄 술폰산(methane sulfonic acid) 4.5g과 혼합한 후, 140℃에서 5시간 동안 반응시킨다. 이어서, 2 리터(liter)의 증류수에서 침전 분리하여 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-6,6'-디올(3,3,3',3'-tetramethyl-1,1'-spirobi(indane)-6,6'-diol)을 제조한다.100 g of bisphenol A is mixed with 4.5 g of methane sulfonic acid, and then reacted at 140 ° C. for 5 hours. Then, precipitated in 2 liters of distilled water to separate 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (indan) -6,6'-diol (3,3,3' , 3'-tetramethyl-1,1'-spirobi (indane) -6,6'-diol) is prepared.
이어서, 상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-6,6'-디올을 질산 360 ml 에 넣고 5시간 동안 반응시켜 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디니트로-6,6'-디올(3,3,3',3'-tetramethyl-1,1'-spirobi(indane)-5,5'-dinitro-6,6'-diol)을 제조하였다.Subsequently, 3,3,3 ', 3'-tetramethyl-1,1'-spirobibi (Indan) -6,6'-diol prepared above was added to 360 ml of nitric acid and reacted for 5 hours to give 3,3, 3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-dinitro-6,6'-diol (3,3,3', 3'-tetramethyl-1,1 '-spirobi (indane) -5,5'-dinitro-6,6'-diol) was prepared.
이어서, 상기 제조한 3,3,3',3'-테트라메틸 -1,1'-스피로비(인단)-5,5'-디니트로-6,6'-디올을 Pd/C 2.5g과 함께 에탄올 330ml에 넣고 환류(reflux)하면서 NH2NH2H2O 66 ml와 10시간 동안 교반하여 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(3,3,3',3'-tetramethyl-1,1'-spirobi(indane)-5,5'-diamino-6,6'-diol) 2.85g을 제조하였다.Subsequently, the 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (indan) -5,5'-dinitro-6,6'-diol prepared above with 2.5 g of Pd / C Put together 330ml of ethanol and reflux while stirring with 66ml of NH 2 NH 2 H 2 O for 10 hours, 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5 , 5'-diamino-6,6'-diol (3,3,3 ', 3'-tetramethyl-1,1'-spirobi (indane) -5,5'-diamino-6,6'-diol) 2.85 g was prepared.
상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 3,3',4,4'-헥사플루오로프로필리덴 디프탈산 이무수물(3,3',4,4'-hexafluoropropylidene diphthalic dianhydride, 6FDA) 4.442 g과 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared above was 3,3 N-methylpyrrolidinone (NMP) with 4.442 g of ', 4,4'-hexafluoropropylidene diphthalic dianhydride (3,3', 4,4'-hexafluoropropylidene diphthalic dianhydride, 6FDA) The solution was reacted for 4 hours in 31.3 g of a solution to prepare a light yellow solution.
이어서, 여기에 피리딘(pyridine) 40 ml와 오르토-자일렌(o-xylene)을 첨가한 후, 환류(reflux)하면서 6시간 동안 반응시켜 노란색 용액을 얻었다. 이어서, 이를 증류수에 침적하고 세척 건조하여 얻어진 6FDA-TSBIDD 폴리이미드를 다시 N-메틸피롤리돈(NMP)에 20 중량%가 되도록 용해시킨다. Subsequently, 40 ml of pyridine and ortho-xylene were added thereto, followed by reaction for 6 hours while refluxing to obtain a yellow solution. Subsequently, the 6FDA-TSBIDD polyimide obtained by immersing it in distilled water and washing and drying was dissolved again in N-methylpyrrolidone (NMP) to 20% by weight.
이어서, 상기 용해된 용액을 유리판 위에 캐스팅하고 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1시간씩 유지하여 필름을 제조하였다. 상기 제조한 필름의 두께는 64 ㎛이었다.Subsequently, the dissolved solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film. The thickness of the film thus prepared was 64 μm.
상기 제조한 폴리이미드 필름을 머플로(muffle furnace)에 넣고 분당 10℃/min의 속도로 300℃까지 가열하고, 300℃에서 30분 동안 유지하였다. 이어서, 425℃까지 가열하고 2 시간 동안 유지하는 열처리를 수행하여 투명한 옅은 갈색의 TSBIDD-계(TSBIDD-based) 폴리벤조옥사졸 필름을 수득함으로써, 기체 분리막을 제조하였다. The polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes. Subsequently, a gas separation membrane was prepared by performing a heat treatment to be heated to 425 ° C. and maintained for 2 hours to obtain a transparent pale brown TSBIDD-based polybenzoxazole film.
상기 수득한 폴리벤조옥사졸 필름에 대해 하기 방법에 따라FTIR 분석, 자유체적도, 면간 거리 및 BET 표면적을 측정하였다. The FTIR analysis, free volume, interplanar distance and BET surface area were measured for the polybenzoxazole film obtained according to the following method.
<FTIR 분석>FTIR analysis
FTIR(fourier transform infrared(FTIR)) 스펙트럼은 적외선 분광기(infrared microspectrometer)(IlluminatIR, SensIR Technologies, Danbury, CT, USA)를 사용하여 측정하였다.Fourier transform infrared (FTIR) spectra were measured using an infrared microspectrometer (IlluminatIR, SensIR Technologies, Danbury, CT, USA).
<자유체적도 측정><Free volume measurement>
고분자의 밀도는 자유체적도와 관련이 있고, 기체 투과도에 영향을 미친다. 막의 밀도를 하기 수학식 1에 따라 사르토리우스 LA 310S 정밀저울(Sartorius LA 310S analytical balance)을 이용하여 부력법(buoyancy method)으로 측정하였다.The density of the polymer is related to the free volume and affects the gas permeability. The film density was measured by buoyancy method using a Sartorius LA 310S analytical balance according to Equation 1 below.
[수학식 1][Equation 1]
상기 수학식 1에서,In Equation 1,
ρP는 고분자의 밀도이고,ρ P is the density of the polymer,
ρ W 는 탈이온수의 밀도이고,ρ W is the density of deionized water,
Wa 는 공기 중에서 측정한 고분자의 무게이고, Wa is the weight of the polymer measured in air,
Ww 는 탈이온수에서 측정한 고분자의 무게이다. Ww is the weight of the polymer measured in deionized water.
자유 체적도(FFV, V f )는 상기 데이터로부터 하기 수학식 2에 따라 계산했다.Free volume degrees (FFV, V f ) were calculated according to the following equation (2) from the data.
[수학식 2][Equation 2]
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 수학식 2에서,In Equation 2,
V는 고분자의 비부피(specific volume)이고,V is the specific volume of the polymer,
Vw는 반데르발스 비부피(specific Van der Waals volume)이다.Vw is the specific Van der Waals volume.
<면간 거리 측정><Measurement between planes>
면간 거리는 X-선 회절 패턴 결과로부터 브래그 식(Bragg's equation)에 따라 계산하였다.The interplanar distance was calculated according to the Bragg's equation from the X-ray diffraction pattern results.
<BET 표면적 측정><BET surface area measurement>
BET 표면적은 표면적 및 기공크기 측정기(pore size and surface area analyzer, ASAP 2020, Micromeritics, USA)를 이용하여 77K의 온도에서 측정한 재료의 상대압력-질소 흡착량의 등온 곡선을 Brunauer-Emmett-Teller의 수식을 이용하여 계산하였다.The BET surface area was measured by Brunauer-Emmett-Teller using an isothermal curve of the relative pressure-nitrogen adsorption of materials measured at a temperature of 77K using a pore size and surface area analyzer (ASAP 2020, Micromeritics, USA). Calculated using the formula.
FTIR 분석 결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.26, 면간 거리는 642 pm였다. BET 표면적은 446 m2/g였다.The FTIR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which were polybenzoxazole characteristic bands that were not present in the polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.26 and an interplanar distance of 642 pm. The BET surface area was 446 m 2 / g.
실시예 2Example 2
450℃에서 30분시간 동안 열처리 한 것을 제외하고는 상기 실시예 1과 동일하게 실시하여 폴리벤조옥사졸을 포함하는 고분자를 제조하고, 이를 이용하여 폴리벤조옥사졸 필름을 얻었다. FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.26, 면간 거리는 619 pm였다. BET 표면적은 371 m2/g였다. Except that the heat treatment for 30 minutes at 450 ℃ was carried out in the same manner as in Example 1 to prepare a polymer containing a polybenzoxazole, to obtain a polybenzoxazole film. The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.26 and an interplanar distance of 619 pm. The BET surface area was 371 m 2 / g.
실시예 3Example 3
상기 제조한 3,3,3', 3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 3,3',4,4'-헥사플루오로프로필리덴 디프탈산 이무수물(3,3',4,4'-hexafluoropropylidene diphthalic dianhydride, 6FDA) 4.442 g과 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4 시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared above was 3,3 N-methylpyrrolidinone (NMP) with 4.442 g of ', 4,4'-hexafluoropropylidene diphthalic dianhydride (3,3', 4,4'-hexafluoropropylidene diphthalic dianhydride, 6FDA) The solution was reacted for 4 hours in 31.3 g of a solution to prepare a pale yellow solution.
제조된 용액을 유리판 위에 캐스팅하고 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1시간씩 유지하여 필름을 제조하였다. 상기 제조한 필름의 두께는 126 ㎛이었다. The prepared solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film. The thickness of the film thus prepared was 126 mu m.
상기 제조한 폴리이미드 필름을 머플로(muffle furnace)에 넣고 분당 10℃/min의 속도로 300℃까지 가열하고, 300℃에서 30분 동안 유지하였다.The polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
이어서, 425℃까지 가열하고 2 시간 동안 유지하는 열처리를 수행하여 투명한 옅은 갈색의 TSBIDD-계(TSBIDD-based) 폴리벤조옥사졸 필름을 수득함으로써, 기체 분리막을 제조하였다.Subsequently, a gas separation membrane was prepared by performing a heat treatment to be heated to 425 ° C. and maintained for 2 hours to obtain a transparent pale brown TSBIDD-based polybenzoxazole film.
FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.26, 면간 거리는 634 pm였다. BET 표면적은 431 m2/g였다.The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.26 and an interplanar distance of 634 pm. The BET surface area was 431 m 2 / g.
실시예 4Example 4
하기 반응식 2로 표시되는 바에 따라 폴리벤조옥사졸을 포함하는 고분자를 제조하였다.A polymer containing polybenzoxazole was prepared as shown in Scheme 2 below.
[반응식 2]Scheme 2
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 파이로멜리틱 이무수물 (pyromelitic, PMDA) 2.181g과 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4 시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared as above was pyromelli A light yellow solution was prepared by reacting with 2.181 g of tic dianhydride (PMDA) in 31.3 g of N-methylpyrrolidinone (NMP) solution for 4 hours.
이어서, 여기에 피리딘(pyridine) 40 ml와 오르토-자일렌(o-xylene)을 첨가한 후, 환류(reflux)하면서 6 시간 동안 반응시켜 노란색 용액을 얻었다. 이어서, 이를 증류수에 침적하고 세척 건조하여 얻어진 PMDA-TSBIDD 폴리이미드를 다시 N-메틸피롤리돈(NMP)에 20 중량%가 되도록 용해시킨다. 이어서, 상기 용해된 용액을 유리판 위에 캐스팅하고 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1시간씩 유지하여 필름을 제조하여, 기체 분리막을 얻었다. 상기 제조한 필름의 두께는 45 ㎛이었다.Subsequently, 40 ml of pyridine and ortho-xylene were added thereto, followed by reaction under reflux for 6 hours to obtain a yellow solution. Subsequently, PMDA-TSBIDD polyimide obtained by immersing it in distilled water and washing and drying is dissolved again in N-methylpyrrolidone (NMP) to 20% by weight. Subsequently, the dissolved solution was cast on a glass plate and maintained at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film to obtain a gas separation membrane. The thickness of the film thus prepared was 45 μm.
열처리 온도: 450℃에서 1.5시간Heat treatment temperature: 1.5 hours at 450 ℃
FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.21, 면간 거리는 525 pm였다. BET 표면적은 282.5 m2/g였다.The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.21 and an interplanar distance of 525 pm. The BET surface area was 282.5 m 2 / g.
실시예 5Example 5
상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 파이로멜리틱 이무수물 (pyromelitic, PMDA) 2.181g과 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4 시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared as above was pyromelli A light yellow solution was prepared by reacting with 2.181 g of tic dianhydride (PMDA) in 31.3 g of N-methylpyrrolidinone (NMP) solution for 4 hours.
제조된 용액을 유리판 위에 캐스팅하고, 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1시간씩 유지하여 필름을 제조하였다. 상기 제조한 필름의 두께는 52 ㎛이었다. The prepared solution was cast on a glass plate and kept in a vacuum oven at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour to prepare a film. The thickness of the film thus prepared was 52 μm.
상기 제조한 폴리이미드 필름을 머플로(muffle furnace)에 넣고 분당 10℃/min의 속도로 300℃까지 가열하고, 300℃에서 30분 동안 유지하였다.The polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
이어서, 450℃까지 가열하고 1.5 시간 동안 유지하는 열처리를 수행하여 투명한 옅은 갈색의 TSBIDD-계(TSBIDD-based) 폴리벤조옥사졸 필름을 제조하였다.Subsequently, heat treatment was performed to heat to 450 ° C. and maintained for 1.5 hours to prepare a transparent pale brown TSBIDD-based polybenzoxazole film.
FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.22, 면간 거리는 544 pm였다.The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.22 and an interplanar distance of 544 pm.
실시예 6Example 6
하기 반응식 3으로 표시되는 바에 따라 폴리벤조옥사졸을 포함하는 고분자를 제조하였다.A polymer containing polybenzoxazole was prepared as shown in Scheme 3 below.
[반응식 3]Scheme 3
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 3,3',4,4'-바이페닐 테트라카르복실산 이무수물( 3,3',4,4'-Biphenyltetracarboxylic Dianhydride, BP) 2.942 g과 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4 시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared above was 3,3 N-methylpyrrolidinone (NMP) solution with 2.942 g of ', 4,4'-biphenyl tetracarboxylic dianhydride (3,3', 4,4'-Biphenyltetracarboxylic Dianhydride, BP) 31.3 The reaction was carried out for 4 hours at g to prepare a pale yellow solution.
이어서, 여기에 피리딘(pyridine) 40 ml와 오르토-자일렌(o-xylene)을 첨가한 후, 환류(reflux)하면서 6 시간 동안 반응시켜 노란색 용액을 얻었다. 이어서, 이를 증류수에 침적하고 세척 건조하여 얻어진 BPDA-TSBIDD 폴리이미드를 다시 N-메틸피롤리돈(NMP)에 20 중량%가 되도록 용해시킨다. 이어서, 상기 용해된 용액을 유리판 위에 캐스팅하고 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1시간씩 유지하여 필름을 제조하였다. 상기 제조한 필름의 두께는 61㎛이었다.Subsequently, 40 ml of pyridine and ortho-xylene were added thereto, followed by reaction under reflux for 6 hours to obtain a yellow solution. Subsequently, the BPDA-TSBIDD polyimide obtained by immersion in distilled water, washed and dried is dissolved again in N-methylpyrrolidone (NMP) to 20% by weight. Subsequently, the dissolved solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film. The thickness of the film thus prepared was 61 μm.
상기 제조한 폴리이미드 필름을 머플로(muffle furnace)에 넣고 분당 10℃/min의 속도로 300℃까지 가열하고, 300℃에서 30분 동안 유지하였다.The polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
이어서, 500℃까지 가열하고 15분 동안 유지하는 열처리를 수행하여 투명한 옅은 갈색의 TSBIDD-계(TSBIDD-based) 폴리벤조옥사졸 필름을 제조하였다.Subsequently, heat treatment was performed to heat to 500 ° C. and maintained for 15 minutes to prepare a transparent pale brown TSBIDD-based polybenzoxazole film.
FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.18, 면간 거리는 526 pm였다. BET 표면적은 306 m2/g였다. The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.18 and an interplanar distance of 526 pm. The BET surface area was 306 m 2 / g.
실시예 7Example 7
상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 3,3',4,4'-바이페닐 테트라카르복실산 이무수물( 3,3',4,4'-Biphenyltetracarboxylic Dianhydride, BPDA) 2.942 g 과 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4 시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared above was 3,3 N-methylpyrrolidinone (NMP) solution with 2.942 g of ', 4,4'-biphenyl tetracarboxylic dianhydride (3,3', 4,4'-Biphenyltetracarboxylic Dianhydride, BPDA) The reaction was carried out for 4 hours at g to prepare a pale yellow solution.
제조된 용액을 유리판 위에 캐스팅하고 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1 시간씩 유지하여 필름을 제조하였다. 상기 제조한 필름의 두께는 93 ㎛이었다. The prepared solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film. The thickness of the film thus prepared was 93 μm.
상기 제조한 폴리이미드 필름을 머플로(muffle furnace)에 넣고 분당 10℃/min의 속도로 300℃까지 가열하고, 300℃에서 30 분 동안 유지하였다.The polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
이어서, 500℃까지 가열하고 15 분 동안 유지하는 열처리를 수행하여 투명한 옅은 갈색의 TSBIDD-계(TSBIDD-based) 폴리벤조옥사졸 필름을 제조하였다.Subsequently, heat treatment was performed to heat to 500 ° C. and maintained for 15 minutes to prepare a transparent pale brown TSBIDD-based polybenzoxazole film.
FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 자유체적도는 0.25, 면간 거리는 565 pm였다. The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the prepared polymer had a free volume of 0.25 and an interplanar distance of 565 pm.
실시예 8Example 8
하기 반응식 4로 표시되는 바에 따라 폴리벤조옥사졸을 포함하는 고분자를 제조하였다.A polymer containing polybenzoxazole was prepared as shown in Scheme 4 below.
[반응식 4]Scheme 4
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
상기 제조한 3,3,3',3'-테트라메틸-1,1'-스피로비(인단)-5,5'-디아미노-6,6'-디올(TSBIDD) 3.384 g을 파이로멜리틱 이무수물 (pyromelitic, PMDA) 1.09g과 3,3',4,4'-헥사플루오로프로필리덴 디프탈산 이무수물(3,3',4,4'-hexafluoropropylidene diphthalic dianhydride, 6FDA) 2.221 g 함께 N-메틸피롤리돈(N-methylpyrrolidinone, NMP) 용액 31.3 g에서 4 시간 동안 반응시켜 연한 노란색 용액을 제조하였다.3.384 g of 3,3,3 ', 3'-tetramethyl-1,1'-spirobi (Indan) -5,5'-diamino-6,6'-diol (TSBIDD) prepared as above was pyromelli With 1.09g of pyromelitic (PMDA) and 2.221g of 3,3 ', 4,4'-hexafluoropropylidene diphthalic dianhydride (3,3', 4,4'-hexafluoropropylidene diphthalic dianhydride, 6FDA) A light yellow solution was prepared by reacting N-methylpyrrolidinone (N-methylpyrrolidinone, NMP) solution for 3 hours in 31.3 g.
이어서, 여기에 피리딘(pyridine) 40 ml와 오르토-자일렌(o-xylene)을 첨가한 후, 환류(reflux)하면서 6 시간 동안 반응시켜 노란색 용액을 얻었다. 이어서, 이를 증류수에 침적하고 세척 건조하여 얻어진 6FDA/PMDA-TSBIDD 폴리이미드를 다시 N-메틸피롤리돈(NMP)에 20 중량%가 되도록 용해시킨다. 이어서, 상기 용해된 용액을 유리판 위에 캐스팅하고 진공 오븐 내에서 100℃, 150℃, 200℃ 및 250℃에서 각 1시간씩 유지하여 필름을 제조하였다. 상기 제조한 필름의 두께는 46㎛이었다.Subsequently, 40 ml of pyridine and ortho-xylene were added thereto, followed by reaction under reflux for 6 hours to obtain a yellow solution. Subsequently, the 6FDA / PMDA-TSBIDD polyimide obtained by immersing it in distilled water and washing and drying was dissolved again in N-methylpyrrolidone (NMP) to 20% by weight. Subsequently, the dissolved solution was cast on a glass plate and kept at 100 ° C., 150 ° C., 200 ° C. and 250 ° C. for 1 hour in a vacuum oven to prepare a film. The thickness of the film thus prepared was 46 μm.
상기 제조한 폴리이미드 필름을 머플로(muffle furnace)에 넣고 분당 10℃/min의 속도로 300℃까지 가열하고, 300℃에서 30 분 동안 유지하였다.The polyimide film prepared above was placed in a muffle furnace and heated to 300 ° C. at a rate of 10 ° C./min per minute, and maintained at 300 ° C. for 30 minutes.
이어서, 450℃까지 가열하고 1시간 동안 유지하는 열처리를 수행하여 투명한 옅은 갈색의 TSBIDD-계(TSBIDD-based) 폴리벤조옥사졸 필름을 제조하였다.Subsequently, heat treatment was performed to heat to 450 ° C. and maintained for 1 hour to prepare a transparent pale brown TSBIDD-based polybenzoxazole film.
FT-IR 분석결과 폴리하이드록시이미드에서는 존재하지 않았던 폴리벤조옥사졸 특성밴드인 1,553 cm-1, 1,480 cm-1(C=N) 및 1,058 cm-1(C-O)의 밴드가 확인되었다. 또한 제조된 고분자의 면간 거리는 831pm였다. BET 표면적은 434 m2/g였다.The FT-IR analysis revealed bands of 1,553 cm -1 , 1,480 cm -1 (C = N) and 1,058 cm -1 (CO), which are polybenzoxazole characteristic bands that were not present in polyhydroxyimide. In addition, the distance between planes of the prepared polymer was 831pm. The BET surface area was 434 m 2 / g.
비교예 1: 고분자 합성 및 이를 이용한 기체 분리막 제조Comparative Example 1: Polymer Synthesis and Gas Separation Membrane Preparation Using the Same
하기 반응식 5로 표시되는 바에 따라 사다리꼴 구조의 다공성 고분자를 제조하였다.A porous polymer having a trapezoidal structure was prepared as shown in Scheme 5 below.
[반응식 5]Scheme 5
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
500ml 플라스크에 3,3,3',3'-테트라메틸-1,1'-스파이로비스인단-5,5',6,6'-테트롤(3,3,3',3'-tetramethyl-1,1'-spirobisindane-5,5',6,6'-tetrol(TTSBI)) 10.213 g(30mmol)과 1,4-다이시아노테트라플루오로벤젠 (1,4-dicyanotetrafluorobenzene (DCTB)) 6.003g (30mmol), 탄산칼륨 8.292g(60mmol)을 디메틸포마이드 (DMF) 200 ml 용매 하에 상온에서 약 20분 격렬히 교반하였다. 3,3,3 ', 3'-tetramethyl-1,1'-spirobisindane-5,5', 6,6'-tetrol (3,3,3 ', 3'-tetramethyl) in 500 ml flask -1,1'-spirobisindane-5,5 ', 6,6'-tetrol (TTSBI)) 10.213 g (30 mmol) and 1,4-dicyanotetrafluorobenzene (1,4-dicyanotetrafluorobenzene (DCTB)) 6.003 g (30 mmol) and 8.292 g (60 mmol) of potassium carbonate were vigorously stirred at room temperature for about 20 minutes under 200 ml of dimethylformamide (DMF).
반응 플라스크를 오일 베스에 넣고 55℃까지 올린 후, 약 23 시간 동안 유지시켰다. 이때 교반 속도는 810 rpm이다. The reaction flask was placed in an oil bath and raised to 55 ° C., and maintained for about 23 hours. At this time, the stirring speed is 810 rpm.
반응 플라스크를 식힌 후 300 ml 물에 부어 정제되지 않은 고분자를 얻는다. 이 고분자를 다시 클로로포름에 녹힌 후 메탄올에 다시 침적을 잡고 건조시켜 PIM-1 고분자를 얻는다. The reaction flask is cooled and then poured into 300 ml of water to obtain a crude polymer. The polymer is again dissolved in chloroform, and then immersed in methanol and dried again to obtain a PIM-1 polymer.
2 wt% 농도의 고분자 용액을 클로로포름 용매를 이용하여 제조한 후, 0.45 ㎛ 실린지 필터를 이용하여 정제한 뒤 유리판에 캐스팅하여 하루 동아 천천히 상온에서 용매를 휘발시켜 고분자 필름을 얻어낸다. PIM-1고분자 필름을 70℃ 오븐에서 건조하여 용매를 완벽히 제거한다. 평균 두께는 53 ㎛였으며, BET 표면적은 850m2/g 였다.A polymer solution having a concentration of 2 wt% was prepared using a chloroform solvent, purified using a 0.45 μm syringe filter, and then cast on a glass plate to volatilize the solvent slowly at room temperature for one day to obtain a polymer film. The PIM-1 polymer film is dried in an oven at 70 ° C. to completely remove the solvent. The average thickness was 53 μm and the BET surface area was 850 m 2 / g.
비교예 2Comparative Example 2
하기 반응식 6으로 표시되는 바에 따라 폴리벤조옥사졸을 포함하는 다공성 고분자를 제조하였다.A porous polymer including polybenzoxazole was prepared as shown in Scheme 6 below.
[반응식 6]Scheme 6
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
2,2'-비스(3-아미노-4-하이드록시페닐)헥사플루오로프로판(bisAPAF, 3.663g, 10mmol)과 NMP(15.06mL)를 질소 기체가 흐르는 100mL 삼구 플라스크에 넣고 0℃로 유지된 아이스배스 안에 둔다. 순서대로 프로필렌 옥사이드(PO, 0.3mL)와 테레프탈로일 클로라이드(TCL, 2.030g, 10mmol)가 NMP(8.35mL)와 혼합된 용액을 플라스크에 넣고 2시간 동안 반응이 진행되도록 둔다. 2,2'-bis (3-amino-4-hydroxyphenyl) hexafluoropropane (bisAPAF, 3.663 g, 10 mmol) and NMP (15.06 mL) were placed in a 100 mL three neck flask with nitrogen gas and maintained at 0 ° C. Place in ice bath. In this order, a solution of propylene oxide (PO, 0.3 mL) and terephthaloyl chloride (TCL, 2.030 g, 10 mmol) mixed with NMP (8.35 mL) was placed in a flask and allowed to proceed for 2 hours.
상온에서 12 시간 동안 교반하면, 점성을 가진 폴리 하이드록시아마이드 용액을 얻게 된다. 이 용액을 유리판 위에 부어 캐스팅한 후, 100℃에서 1 시간, 200℃에서 10 시간 동안 진공 상태의 오븐 안에 두어 용매가 제거되도록 한다. 천천히 식힌 후 폴리 하이드록시아마이드 전구체 필름을 얻는다. After stirring for 12 hours at room temperature, a viscous polyhydroxyamide solution is obtained. The solution is poured onto a glass plate, cast, and then placed in a vacuum oven for 1 hour at 100 ° C. and 10 hours at 200 ° C. to remove the solvent. After cooling slowly, a poly hydroxyamide precursor film is obtained.
폴리 하이드록시 아마이드 전구체 필름은 아르곤 기체가 흐르며, 분당 5℃씩 증가하는 화로에 두어 350℃까지 온도를 증가시킨 뒤 한 시간 동안 유지시킨다. 화로 내부의 온도를 천천히 식힌 후 폴리벤조옥사졸 필름을 얻어낸다. 고분자 필름의 평균 두께는 40㎛였으며, 밀도는 1.39g/cm3, 면간거리는 719 pm였다.The poly hydroxy amide precursor film flows argon gas, placed in a furnace that increases by 5 ° C. per minute, increases the temperature to 350 ° C. and is maintained for one hour. After slowly cooling the temperature inside the furnace, a polybenzoxazole film is obtained. The average thickness of the polymer film was 40 μm, the density was 1.39 g / cm 3 , and the interplanar distance was 719 pm.
비교예 3Comparative Example 3
하기 반응식 7로 표시되는 바에 따라 폴리벤조옥사졸을 포함하는 다공성 고분자를 제조하였다.A porous polymer including polybenzoxazole was prepared as shown in Scheme 7 below.
[반응식 7]Scheme 7
[규칙 제91조에 의한 정정 17.06.2013]
[Revision 17.06.2013 under Rule 91]
2,2'-비스(3-아미노-4-하이드록시페닐)헥사플루오로프로판(bisAPAF, 3.663g, 10mmol)과 NMP(15.06mL)를 질소 기체가 흐르는 100mL 삼구 플라스크에 넣고 0℃로 유지된 아이스배스 안에 둔다. 순서대로 프로필렌 옥사이드(PO, 0.3mL)와 파라-페닐렌(테드랄로일 디클로라이드)(TPCL, 2.030g, 10mmol)이 NMP(8.35mL)와 혼합된 용액을 플라스크에 넣고, 2 시간 동안 반응이 진행되도록 둔다. 2,2'-bis (3-amino-4-hydroxyphenyl) hexafluoropropane (bisAPAF, 3.663 g, 10 mmol) and NMP (15.06 mL) were placed in a 100 mL three neck flask with nitrogen gas and maintained at 0 ° C. Place in ice bath. In this order, a solution of propylene oxide (PO, 0.3 mL) and para-phenylene (tedraloyl dichloride) (TPCL, 2.030 g, 10 mmol) mixed with NMP (8.35 mL) was added to the flask, followed by reaction for 2 hours. Allow this to proceed.
상온에서 12 시간 동안 교반하면, 점성을 가진 폴리 하이드록시아마이드 용액을 얻게 된다. 이 용액을 유리판 위에 부어 캐스팅한 후, 100℃에서 1 시간, 200℃에서 10 시간 동안 진공 상태의 오븐 안에 두어 용매가 제거되도록 한다. 천천히 식힌 후 폴리 하이드록시아마이드 전구체 필름을 얻는다. After stirring for 12 hours at room temperature, a viscous polyhydroxyamide solution is obtained. The solution is poured onto a glass plate, cast, and then placed in a vacuum oven for 1 hour at 100 ° C. and 10 hours at 200 ° C. to remove the solvent. After cooling slowly, a poly hydroxyamide precursor film is obtained.
폴리 하이드록시아마이드 전구체 필름은 아르곤 기체가 흐르며, 분당 5℃씩 증가하는 화로에 두어 350℃까지 온도를 증가시킨 뒤 한 시간 동안 유지시킨다. 화로 내부의 온도를 천천히 식힌 후 폴리벤조옥사졸 필름을 얻어낸다. 고분자 필름의 평균 두께는 42 ㎛였으며, 밀도는 1.32 g/cm3, 면간거리는 758 pm였다.The poly hydroxyamide precursor film is placed in a furnace with flowing argon gas and increased by 5 ° C. per minute, increased in temperature to 350 ° C. and maintained for one hour. After slowly cooling the temperature inside the furnace, a polybenzoxazole film is obtained. The average thickness of the polymer film was 42 μm, the density was 1.32 g / cm 3 , and the interplanar distance was 758 pm.
시험예 1: 기계적 특성 평가Test Example 1: Mechanical Properties Evaluation
실시예 1 내지 실시예 9 및 비교예 1 내지 비교예 3에서 제조된 필름을 기체 분리막으로 사용하여, 상기 기체 분리막의 기계적인 특성을 AGS-J 500N(shimadzu)를 이용하여 25℃에서 측정하였다. 각각의 샘플마다 4 개씩의 시편에 대하여 시험을 실시하였다. 평균에 대한 표준편차는 ±10%였다. 그 결과를 하기 표 1에 나타낸다.Using the films prepared in Examples 1 to 9 and Comparative Examples 1 to 3 as a gas separation membrane, the mechanical properties of the gas separation membrane was measured at 25 ° C using AGS-J 500N (shimadzu). Four specimens were tested for each sample. The standard deviation for the mean was ± 10%. The results are shown in Table 1 below.
표 1
상기 표 1 나타난 바와 같이, 실시예 1 내지 실시예 8에서 제조된 기체 분리막은 대체적으로 파괴점에서의 연신율(elongation percent at break, 단위: %)이 비교예 1 내지 비교예 3에서 제조된 기체 분리막보다 우수하다. 이는 실시예 1 내지 실시예 8에서 제조된 기체 분리막에 포함되는 고분자가 벤조옥사졸 구조와 사다리형의 구조를 동시에 가져 강직하면서도 인장강도가 증가할 수 있는 유연성을 동시에 포함하는 특성을 가지기 때문이다.As shown in Table 1, the gas separation membranes prepared in Examples 1 to 8 generally have elongation percent at break (unit:%) at the gas separation membranes prepared in Comparative Examples 1 to 3. Better than This is because the polymers included in the gas separation membranes prepared in Examples 1 to 8 have a benzoxazole structure and a ladder-type structure at the same time to have a rigidity and flexibility to increase tensile strength.
상기 시험 결과로부터, 실시예 1 내지 실시예 8에서 제조된 기체 분리막에 포함된 고분자는 벤조옥사졸 구조와 사다리형 구조를 동시에 가짐으로써 온화한 조건에서뿐만 아니라 긴 작업 시간, 산성 조건 및 고습, 고온과 같은 가혹한 조건하에서도 견딜 수 있음을 확인할 수 있다. From the test results, the polymers included in the gas separation membranes prepared in Examples 1 to 8 have a benzoxazole structure and a ladder structure at the same time, such as long working time, acidic conditions and high humidity, high temperature, as well as mild conditions. It can be seen that it can withstand harsh conditions.
시험예 2: 기체 투과도(permeability) 및 선택도(selectivity) 측정Test Example 2: Measurement of gas permeability and selectivity
실시예 1 내지 8 및 비교예 2 내지 비교예 3에서 제조한 기체 분리막에 대하여, 기체 투과도 및 선택도를 알아보기 위해 하기와 같은 시험을 실시했다. 그 결과를 하기 표 2 및 표 3에 나타낸다.The gas separation membranes prepared in Examples 1 to 8 and Comparative Examples 2 to 3 were subjected to the following tests in order to determine gas permeability and selectivity. The results are shown in Tables 2 and 3 below.
고진공 시간지연 장치(high-vacuum time-lag apparatus)를 이용하여 기체 투과도 및 선택도를 측정하였으며, 다운스트림 부피는 30 cm3로 조정하였고, 업스트림 압력 및 다운스트림 압력은 각각 33 atm 및 0.002 atm의 풀스케일(full scale)을 가진 바라트론 트랜스듀서(Baratron transducer)를 이용하여 측정하였다.Gas permeability and selectivity were measured using a high-vacuum time-lag apparatus, the downstream volume was adjusted to 30 cm 3 , and the upstream and downstream pressures were 33 atm and 0.002 atm, respectively. Measurements were made using a Baratron transducer with full scale.
모든 순수 기체 투과도 측정은 27℃에서 5번 이상 실시하였다. 투과도의 평균값에 대한 표준편차는 ±2% 이내였으며, 샘플들의 재현 가능성은 ±5% 이내로 우수하였다. 기체 분리막의 유효 면적은 4.00 cm2였다.All pure gas permeability measurements were performed 5 times or more at 27 ° C. The standard deviation for the mean value of permeability was within ± 2% and the reproducibility of the samples was excellent within ± 5%. The effective area of the gas separation membrane was 4.00 cm 2 .
순수 기체들에 대하여, 고정 압력에서 투과된 부피 또는 고정 수집 부피에서 투과압력의 증가 속도를 측정할 수 있다. 주입압력 p 1 이 대기압 이상인 경우 투과압력 p 2 는 매우 작은 값(< 2 Torr)을 가진다. 투과 면에서의 압력이 p 2 대 시간으로 기록되면서 측정되는 동안, 고분자막을 통한 기체 분자의 투과도의 근사값을 구할 수 있다. A 분자의 기체 투과율 P A 는 평형 상태에서 다운스트림 압력이 고정 투과 부피를 증가시키는 속도로부터 하기 수학식 3에 따라 계산될 수 있다. For pure gases, the rate of increase of the permeation pressure in the volume permeated at a fixed pressure or in a fixed collection volume can be measured. Injection pressurep One Permeation pressure above this atmospheric pressurep 2 Has a very small value (<2 Torr). Pressure on the permeate sidep 2 While measured while recording in large time, an approximation of the permeability of gas molecules through the polymer membrane can be obtained. Gas Permeability of A MoleculesP A Can be calculated according to the following equation from the rate at which the downstream pressure increases the fixed permeate volume at equilibrium.
[수학식 3][Equation 3]
상기 수학식 3에서,In Equation 3,
V 는 고정 다운스트림 수집기의 부피이고, V is the volume of the fixed downstream collector,
l 은 막 두께이고, l is the film thickness,
A 는 막 면적이고, A is just area,
p 1 및 p 2는 각각 업스트림 및 다운스트림에서의 압력이고, p 1 and p 2 are the pressures upstream and downstream, respectively,
R, T 및 t는 각각 기체상수, 온도 및 시간이다. R , T and t are gas constant, temperature and time, respectively.
표 2
표 3
상기 표 2 및 표 3에 나타난 바와 같이, 실시예 1 내지 8에서 제조한 기체 분리막은 대체로 비교예 2 및 3에서 제조한 기체 분리막에 비해서 우수한 기체 투과도를 가지고 있다. 또한, 실시예 1 내지 8에서 제조한 기체 분리막은 비교예 2및 3에서 제조한 기체 분리막보다 우수한 수준의 기체 선택도를 가지는 것을 확인할 수 있다.As shown in Table 2 and Table 3, the gas separation membranes prepared in Examples 1 to 8 generally have excellent gas permeability compared to the gas separation membranes prepared in Comparative Examples 2 and 3. In addition, it can be seen that the gas separation membranes prepared in Examples 1 to 8 have a higher level of gas selectivity than the gas separation membranes prepared in Comparative Examples 2 and 3.
이로써, 실시예 1 내지 8에서 제조한 기체 분리막이 비교예 2 및 비교예 3에서 제조한 기체 분리막보다 많은 양의 기체를 효율적으로 분리할 수 있음을 확인할 수 있다.Thus, it can be seen that the gas separation membranes prepared in Examples 1 to 8 can efficiently separate a larger amount of gas than the gas separation membranes prepared in Comparative Examples 2 and 3.
이상을 통해 본 발명의 구체적인 실시예에 대하여 설명하였지만, 본 발명은 이에 한정되는 것이 아니고 특허청구범위와 발명의 상세한 설명 및 첨부한 도면의 범위 안에서 여러 가지로 변형하여 실시하는 것이 가능하고 이 또한 본 발명의 범위에 속하는 것은 당연하다.Although specific embodiments of the present invention have been described above, the present invention is not limited thereto, and various modifications and changes can be made within the scope of the claims and the detailed description of the invention and the accompanying drawings. Naturally, it belongs to the scope of the invention.
Claims (30)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120050945A KR101404160B1 (en) | 2012-05-14 | 2012-05-14 | Polymer, method of preparing the same, and article including the polymer |
| KR10-2012-0050945 | 2012-05-14 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013172554A1 true WO2013172554A1 (en) | 2013-11-21 |
Family
ID=49583929
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2013/003030 Ceased WO2013172554A1 (en) | 2012-05-14 | 2013-04-11 | Polymer, preparation method thereof, and molded product comprising said polymer |
Country Status (2)
| Country | Link |
|---|---|
| KR (1) | KR101404160B1 (en) |
| WO (1) | WO2013172554A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107406591A (en) * | 2015-03-17 | 2017-11-28 | 陶氏环球技术有限责任公司 | Intrinsically Microporous Polymers |
| CN115477757A (en) * | 2021-05-31 | 2022-12-16 | 中国科学院宁波材料技术与工程研究所 | Thermally rearranged polymer based on 2,2',3,3'-biphenyltetracarboxylic dianhydride and its preparation method and application |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102763902B1 (en) * | 2022-02-15 | 2025-02-07 | 한국화학연구원 | Anhydride compound, polyimide-based polymer compound prepared therefrom, and asymmetric hollow fiber membrane for gas separation comprising the same |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06254367A (en) * | 1993-03-02 | 1994-09-13 | Ube Ind Ltd | Asymmetric hollow fiber polyimide gas separation membrane |
| JPH11342322A (en) * | 1998-06-02 | 1999-12-14 | Nitto Denko Corp | Conductive polyaniline polyimide composition and gas separation membrane comprising the same |
| KR100997531B1 (en) * | 2008-05-19 | 2010-11-30 | 한양대학교 산학협력단 | Hollow fiber, dope solution composition for hollow fiber formation and hollow fiber manufacturing method using the same |
| KR20110136745A (en) * | 2010-06-14 | 2011-12-21 | 한양대학교 산학협력단 | Separator for lithium secondary battery and manufacturing method thereof |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TW293832B (en) * | 1995-10-13 | 1996-12-21 | Du Pont | Polyimide alignment film from 2,2-bis(3,4-dicarboxyphenyl)-hexafluoropropane dianhydride and ortho-substituted aromatic diamines for active matrix liquid crystal displays |
-
2012
- 2012-05-14 KR KR1020120050945A patent/KR101404160B1/en not_active Expired - Fee Related
-
2013
- 2013-04-11 WO PCT/KR2013/003030 patent/WO2013172554A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH06254367A (en) * | 1993-03-02 | 1994-09-13 | Ube Ind Ltd | Asymmetric hollow fiber polyimide gas separation membrane |
| JPH11342322A (en) * | 1998-06-02 | 1999-12-14 | Nitto Denko Corp | Conductive polyaniline polyimide composition and gas separation membrane comprising the same |
| KR100997531B1 (en) * | 2008-05-19 | 2010-11-30 | 한양대학교 산학협력단 | Hollow fiber, dope solution composition for hollow fiber formation and hollow fiber manufacturing method using the same |
| KR20110136745A (en) * | 2010-06-14 | 2011-12-21 | 한양대학교 산학협력단 | Separator for lithium secondary battery and manufacturing method thereof |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN107406591A (en) * | 2015-03-17 | 2017-11-28 | 陶氏环球技术有限责任公司 | Intrinsically Microporous Polymers |
| US9920168B2 (en) * | 2015-03-17 | 2018-03-20 | Dow Global Technologies Llc | Polymers of intrinsic microporosity |
| CN107406591B (en) * | 2015-03-17 | 2020-03-24 | 陶氏环球技术有限责任公司 | Inherently Microporous Polymer |
| CN115477757A (en) * | 2021-05-31 | 2022-12-16 | 中国科学院宁波材料技术与工程研究所 | Thermally rearranged polymer based on 2,2',3,3'-biphenyltetracarboxylic dianhydride and its preparation method and application |
| CN115477757B (en) * | 2021-05-31 | 2024-04-05 | 中国科学院宁波材料技术与工程研究所 | Thermally rearranged polymer based on 2,2',3,3'-biphenyltetracarboxylic dianhydride and its preparation method and application |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20130127197A (en) | 2013-11-22 |
| KR101404160B1 (en) | 2014-06-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2010041909A2 (en) | Polymer and preparation method thereof | |
| WO2015183056A1 (en) | Polyimide-based liquid and polyimide-based film produced using same | |
| WO2009142434A2 (en) | Hollow fiber, dope solution composition for forming a hollow fiber, and method for manufacturing a hollow fiber using the same | |
| WO2018038436A1 (en) | Diamine compound and method for producing same | |
| WO2017111300A1 (en) | Polyamic acid solution to which diamine monomer having novel structure is applied, and polyimide film comprising same | |
| WO2009142433A2 (en) | Hollow fiber, dope solution composition for forming a hollow fiber, and method for manufacturing a hollow fiber using the same | |
| WO2014104557A1 (en) | Novel polyamideimide having low thermal expansion coefficient | |
| WO2011122842A2 (en) | Polyimide film | |
| WO2016032299A1 (en) | Polyimide preparation method using monomer salt | |
| WO2017047917A1 (en) | Modified polyimide and curable resin composition containing same | |
| WO2019235712A1 (en) | Siloxane compound and polyimide precursor composition comprising same | |
| WO2015072692A1 (en) | Thermally rearranged poly(benzoxazole-imide) copolymer having cross-linked structure, gas separation membrane comprising same and preparation method therefor | |
| WO2013172554A1 (en) | Polymer, preparation method thereof, and molded product comprising said polymer | |
| WO2020209625A1 (en) | Polyamide-imide block copolymer, method for preparing same, and polyamide-imide film comprising same | |
| WO2015182925A1 (en) | Novel diamine synthesis, and liquid crystal alignment agent using same | |
| WO2020159086A1 (en) | Polyamide resin film and resin laminate using same | |
| WO2021060752A1 (en) | Polyimide-based film having excellent surface evenness and method for producing same | |
| WO2018147617A1 (en) | Polyamide-imide film and method for producing same | |
| WO2018147606A1 (en) | Polyamide-imide film and method for preparing same | |
| WO2015072694A1 (en) | Flue gas separation membrane comprising thermally rearranged poly(benzoxazole-imide) copolymer having cross-linked structure, and preparation method therefor | |
| WO2020105933A1 (en) | Liquid crystal alignment agent composition, method for preparing liquid crystal alignment film by using same, and liquid crystal alignment film and liquid crystal display device which use same | |
| WO2022145950A1 (en) | Precursor, polymer, optical film comprising polymer, and display device comprising optical film | |
| WO2020159035A1 (en) | Polyimide film, flexible substrate using same, and flexible display comprising flexible substrate | |
| WO2020036443A1 (en) | Low-friction resin composite | |
| WO2020130552A1 (en) | Diamine compound, polyimide precursor using same, and polyimide film |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13790153 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13790153 Country of ref document: EP Kind code of ref document: A1 |