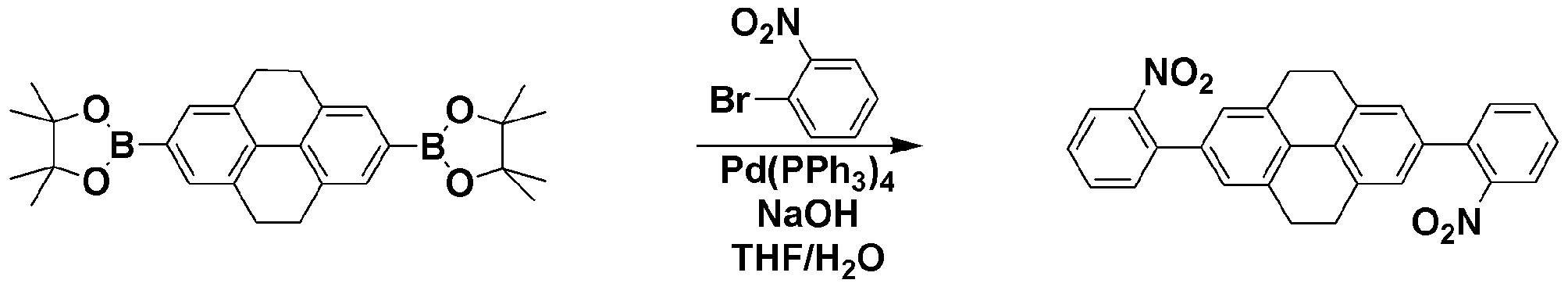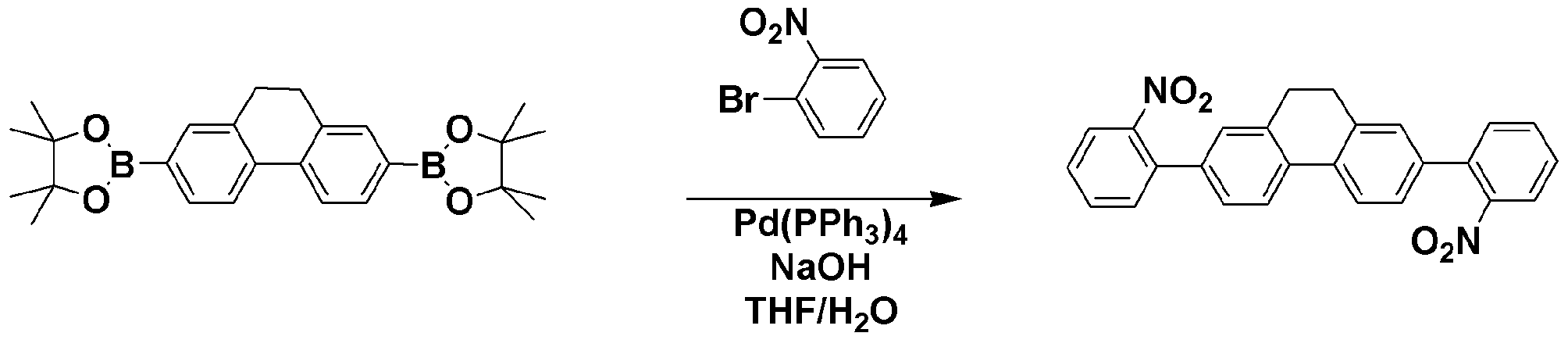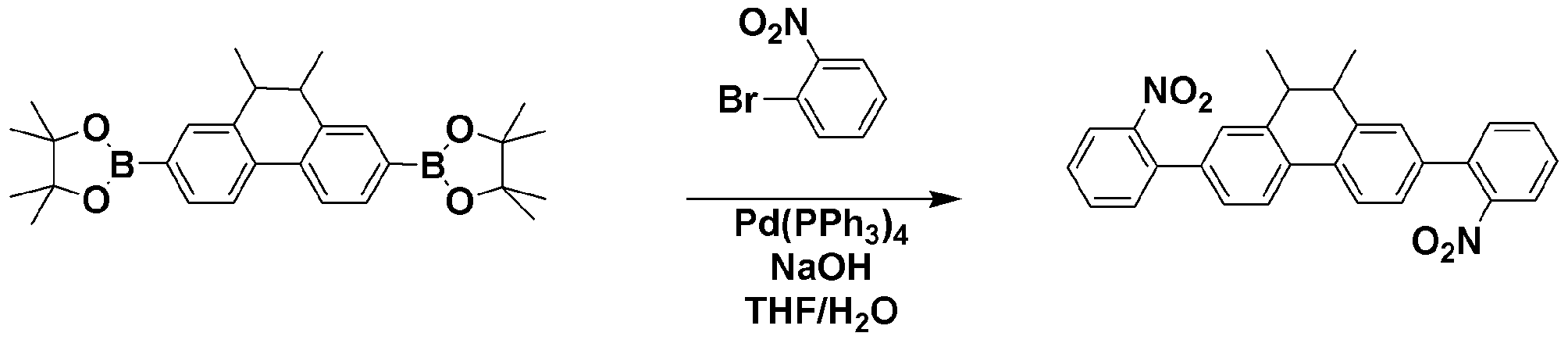WO2013100507A1 - Novel compound and organic electroluminescence device including same - Google Patents
Novel compound and organic electroluminescence device including same Download PDFInfo
- Publication number
- WO2013100507A1 WO2013100507A1 PCT/KR2012/011341 KR2012011341W WO2013100507A1 WO 2013100507 A1 WO2013100507 A1 WO 2013100507A1 KR 2012011341 W KR2012011341 W KR 2012011341W WO 2013100507 A1 WO2013100507 A1 WO 2013100507A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- spc
- synthesis
- formula
- mol
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 Cc(cc12)c(C)cc1-c1cc(*)c(*3CC3)c(*)c1C(*)C2I Chemical compound Cc(cc12)c(C)cc1-c1cc(*)c(*3CC3)c(*)c1C(*)C2I 0.000 description 4
- INJYATWDDJSOIP-UHFFFAOYSA-N Brc1nc(-c2ccccc2)cc(-c2ccccc2)c1 Chemical compound Brc1nc(-c2ccccc2)cc(-c2ccccc2)c1 INJYATWDDJSOIP-UHFFFAOYSA-N 0.000 description 1
- OQHHZGQGRJLQKO-UHFFFAOYSA-N C(C1)c(cc(c2ccccc2[nH]2)c2c2CCc3c4)c2-c3c1c1c4c2ccccc2[nH]1 Chemical compound C(C1)c(cc(c2ccccc2[nH]2)c2c2CCc3c4)c2-c3c1c1c4c2ccccc2[nH]1 OQHHZGQGRJLQKO-UHFFFAOYSA-N 0.000 description 1
- SKXKNDKNBXDPMG-UHFFFAOYSA-N C1c(cc(c2ccccc2[n]2-c3nc(-c4ccccc4)cc(-c4ccccc4)c3)c2c2CC3)c2-c2c3cc(c(cccc3)c3[n]3-c4cc(-c5ccccc5)cc(-c5ccccc5)n4)c3c2C1 Chemical compound C1c(cc(c2ccccc2[n]2-c3nc(-c4ccccc4)cc(-c4ccccc4)c3)c2c2CC3)c2-c2c3cc(c(cccc3)c3[n]3-c4cc(-c5ccccc5)cc(-c5ccccc5)n4)c3c2C1 SKXKNDKNBXDPMG-UHFFFAOYSA-N 0.000 description 1
- BDJDWGLGOKIDMY-UHFFFAOYSA-N c(cc1)cc2c1/C1=N/C(c3ccccc3C3=NC(c4c5cccc4)N4C5=NC5c6ccccc66)N3[U+]4N5/C6=N\C2N1 Chemical compound c(cc1)cc2c1/C1=N/C(c3ccccc3C3=NC(c4c5cccc4)N4C5=NC5c6ccccc66)N3[U+]4N5/C6=N\C2N1 BDJDWGLGOKIDMY-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/10—Apparatus or processes specially adapted to the manufacture of electroluminescent light sources
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/653—Aromatic compounds comprising a hetero atom comprising only oxygen as heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
Definitions
- the present invention relates to a novel compound and an organic electroluminescent device comprising the same, and more particularly to a compound used in the organic material layer of the organic electroluminescent device.
- the organic electroluminescent device when a voltage is applied between two electrodes, holes are injected from the anode and electrons are injected into the organic material layer from the cathode. When the injected holes and the electrons meet to form an exciton, the formed excitons fall to the ground state. The light will shine.
- the material used as the organic material layer may be classified into a light emitting material, a hole injection material, a hole transport material, an electron transport material, an electron injection material and the like according to a function.
- the light emitting material may be classified into blue, green, and red light emitting materials and yellow and orange light emitting materials for realizing natural colors according to light emitting colors.
- a host / dopant system may be used as a light emitting material.
- the dopant material may be divided into a fluorescent dopant using an organic material and a phosphorescent dopant using a metal complex compound containing heavy atoms such as Ir and Pt.
- a metal complex compound containing heavy atoms such as Ir and Pt.
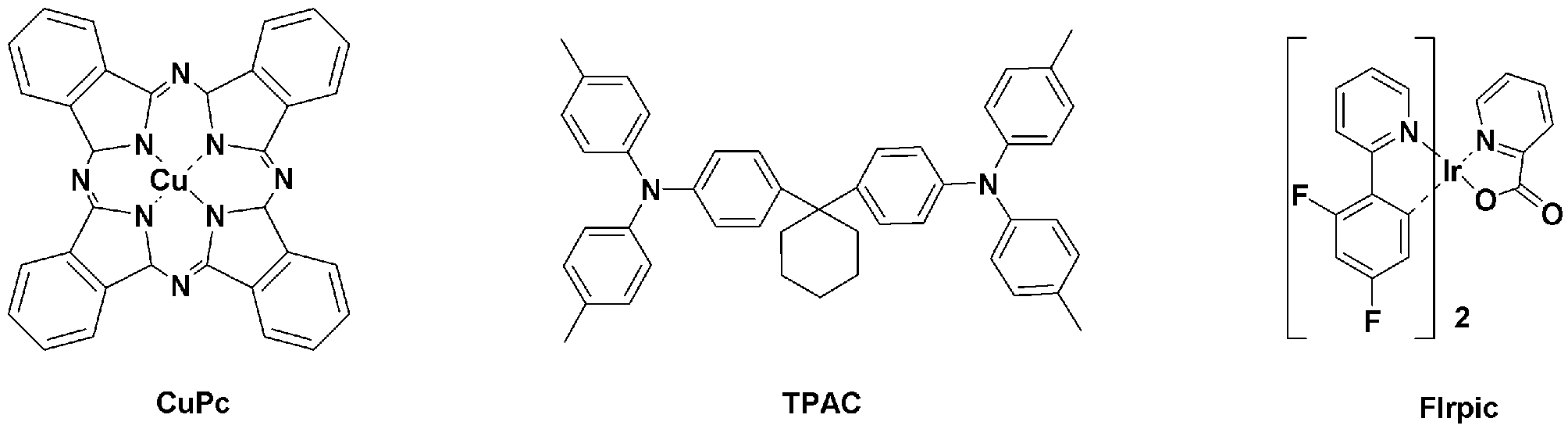
- NPB, BCP, Alq 3 and the like are used as materials for the hole blocking layer and the electron transport layer, and anthracene derivatives are used as the light emitting material.
- metal complex compounds including Ir such as Firpic, Ir (ppy) 3 , and (acac) Ir (btp) 2 among the light emitting materials are phosphorescent dopant materials of blue, green, and red, and CBP is used as a phosphorescent host material.
- JP Laid-Open No. 2004-0094842 discloses a technique of using a compound having a nitrogen-containing heterocyclic group bonded to an arylcarbazolyl group or carbazolylalkylene group as a phosphorescent host material.
- the conventional luminescent materials have good luminescence properties, but the glass transition temperature is low and the thermal stability is not very good. Thus, the luminescent materials are not satisfactory in terms of the lifespan of the organic EL device. Therefore, there is a demand for development of a light emitting material having excellent performance.
- an object of the present invention is to provide a novel compound and an organic electroluminescent device using the compound which can improve the efficiency, lifespan and stability of the organic electroluminescent device.
- the present invention provides a compound represented by the following formula (1).
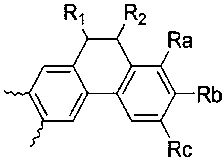
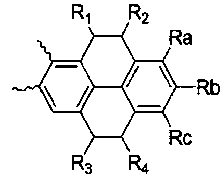
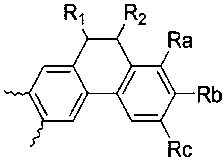
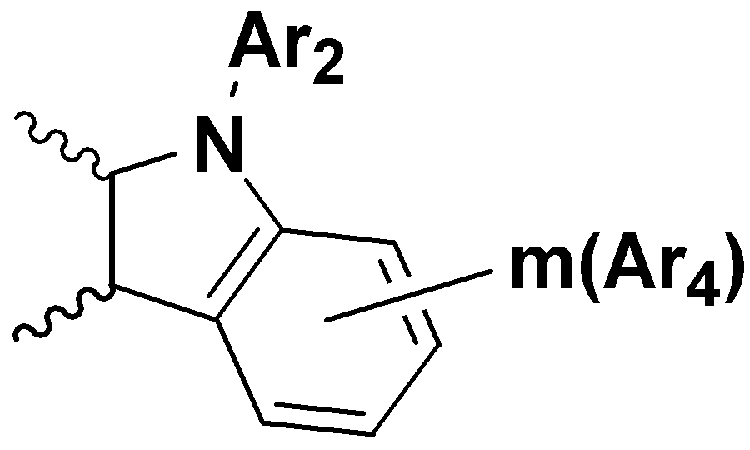
- A is , And Selected from the group consisting of, R a and R b or R b and R c form a condensed ring represented by the following formula (2), and when R a and R b form a condensed ring, R c is hydrogen and R When b and R c form a condensed ring, R a is hydrogen,
- Ar 1 to Ar 4 are each independently a C 1 to C 40 alkyl group, C 3 to C 40 cycloalkyl group, C 3 to C 40 heterocycloalkyl group, C 6 to C 60 aryl group, C 5 Is selected from the group consisting of -C 60 heteroaryl group, C 1 -C 40 alkyloxy group, C 6 -C 60 aryloxy group, and C 6 -C 60 arylamine group,
- R 1 to R 4 are each independently hydrogen, halogen, C 1 -C 40 alkyl group, C 3 -C 40 cycloalkyl group, C 3 -C 40 heterocycloalkyl group, C 6 -C 60 aryl Group, C 5 ⁇ C 60 heteroaryl group, C 1 ⁇ C 40 alkyloxy group, C 6 ⁇ C 60 aryloxy group and C 6 ⁇ C 60 arylamine group,
- N and m are each an integer of 0 to 4.
- the alkyl group, the cycloalkyl group, the heterocycloalkyl group, the aryl group, the heteroaryl group, the alkyloxy group, the aryloxy group and the arylamine group of Ar 1 to Ar 4 are each independently hydrogen, an alkyl group of C 1 to C 40 , aryloxy group of C 2 ⁇ C 40 alkenyl group, C 2 ⁇ C 40 alkynyl group, C 6 ⁇ C 40 aryl group, C 5 ⁇ C 40 heteroaryl group, C 6 ⁇ C 40 of, C 1 ⁇ C 40 alkyloxy group, C 6 ⁇ C 40 arylamino group, C 6 ⁇ C 40 diarylamino group, C 6 ⁇ C 40 arylalkyl group, C 3 ⁇ C 40 cycloalkyl group, C 6 ⁇ C 40 It may be substituted with one or more substituents selected from the group consisting of an arylsilyl group and a C 3 to C 40 heterocycloalkyl group
- Alkyl of the present invention means a straight chain or branched saturated hydrocarbon having 1 to 40 carbon atoms, and examples thereof include methyl, ethyl, propyl, isobutyl, sec-butyl, pentyl, iso-amyl, hexyl and the like.
- Aryl of the present invention means an aromatic moiety having 6 to 60 carbon atoms combined with a single ring or two or more rings, and two or more rings may be in the form of a simple attachment or condensation with each other.
- Heteroaryl of the present invention means a monoheterocyclic or polyheterocyclic aromatic moiety having 5 to 60 nuclear atoms, wherein at least one carbon in the ring, preferably 1 to 3 carbons is N, O, S or Se Mean substituted with a hetero atom such as.
- the heteroaryl may be in a form in which two or more rings are simply attached or condensed with each other, and a condensed form with an aryl group may also be included.
- the condensed ring of the present invention means a condensed aliphatic ring, a condensed aromatic ring, a condensed heteroaliphatic ring, a condensed heteroaromatic ring or a combination thereof.
- the present invention in the organic electroluminescent device comprising an anode, a cathode and one or more organic material layer interposed between the anode and the cathode, at least one of the organic material layer comprises a compound represented by the formula (1) It provides an organic electroluminescent device characterized in that the organic material layer.
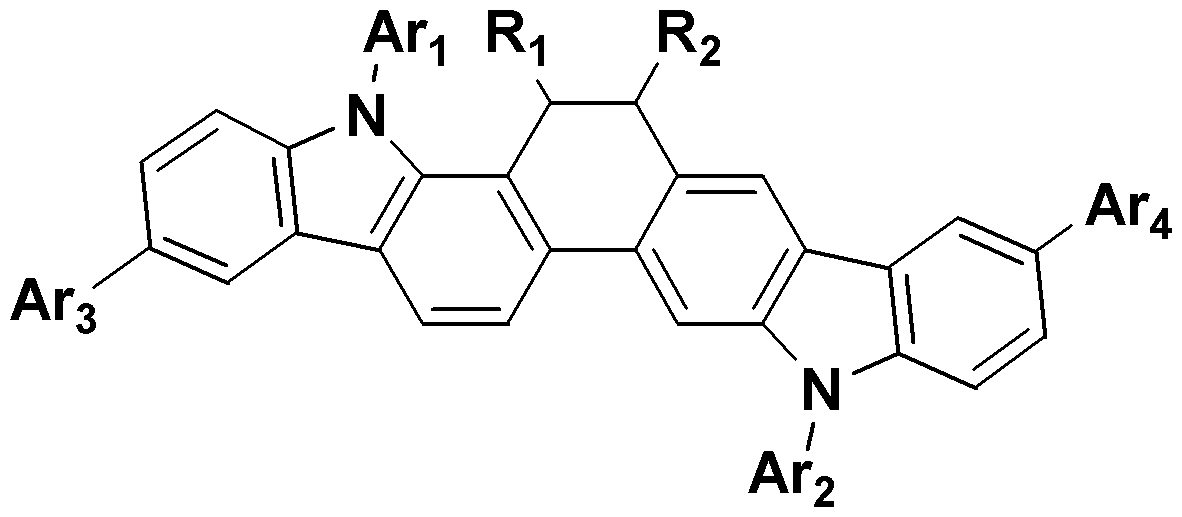
- the novel compound according to the present invention forms a basic skeleton by binding an indole group to the sock end of tetrahydropyrene or tetrahydrophenanthrene, and is represented by Formula 1 as a compound in which various substituents are bonded.
- a compound represented by Formula 1 of the present invention is a conventional organic light emitting device material (for example, by adjusting the energy level by combining a variety of substituents (R 1 to R 4 and Ar 1 to Ar 4 in Formula 1) It has a higher molecular weight than CBP (4,4-dicarbazolybiphenyl)) and is characterized by a wide energy bandgap.
- the compound represented by the formula (1) of the present invention in which various substituents are introduced has a significant increase in molecular weight, thereby improving the glass transition temperature, thereby having a high thermal stability compared to conventional materials.
- the compound represented by Chemical Formula 1 of the present invention when used as a material of an organic electroluminescent device, not only phosphorescence properties of the device, but also electron and / or hole transporting ability, luminous efficiency, driving voltage, and lifetime characteristics may be improved.
- the compound represented by Formula 1 of the present invention may be used as a material of the organic material layer of the organic electroluminescent device, preferably a light emitting layer, a hole transporting layer or an electron transporting layer, more preferably a host material of the light emitting layer.
- the compound represented by the formula (1) of the present invention is preferably selected from the group consisting of the compound represented by the following formula (3 to 6).
- Ar 1 to Ar 4 and R 1 to R 4 are the same as those described for Formula 1 above.
- Ar 1 to Ar 4 of the compounds represented by Formulas 3 to 6 are each independently hydrogen, an alkyl group of C 1 to C 40 , C 6 in consideration of the lifespan, luminous efficiency, driving voltage, etc. of the organic EL device. It is preferably selected from the group consisting of an aryl group of ⁇ C 60 and a heteroaryl group of C 5 ⁇ C 60 , hydrogen, methyl, phenyl, pyridine, pyrimidine, 1, 3,5-triazine, naphthalene, quinoline, 1,10-phenanthroline, acenaphthylene, biphenyl more preferably selected from the group consisting of biphenyl), fluorine and 9H-carbazole.
- R 1 to R 4 of the compounds represented by Formulas 3 to 6 are each independently hydrogen or methyl.
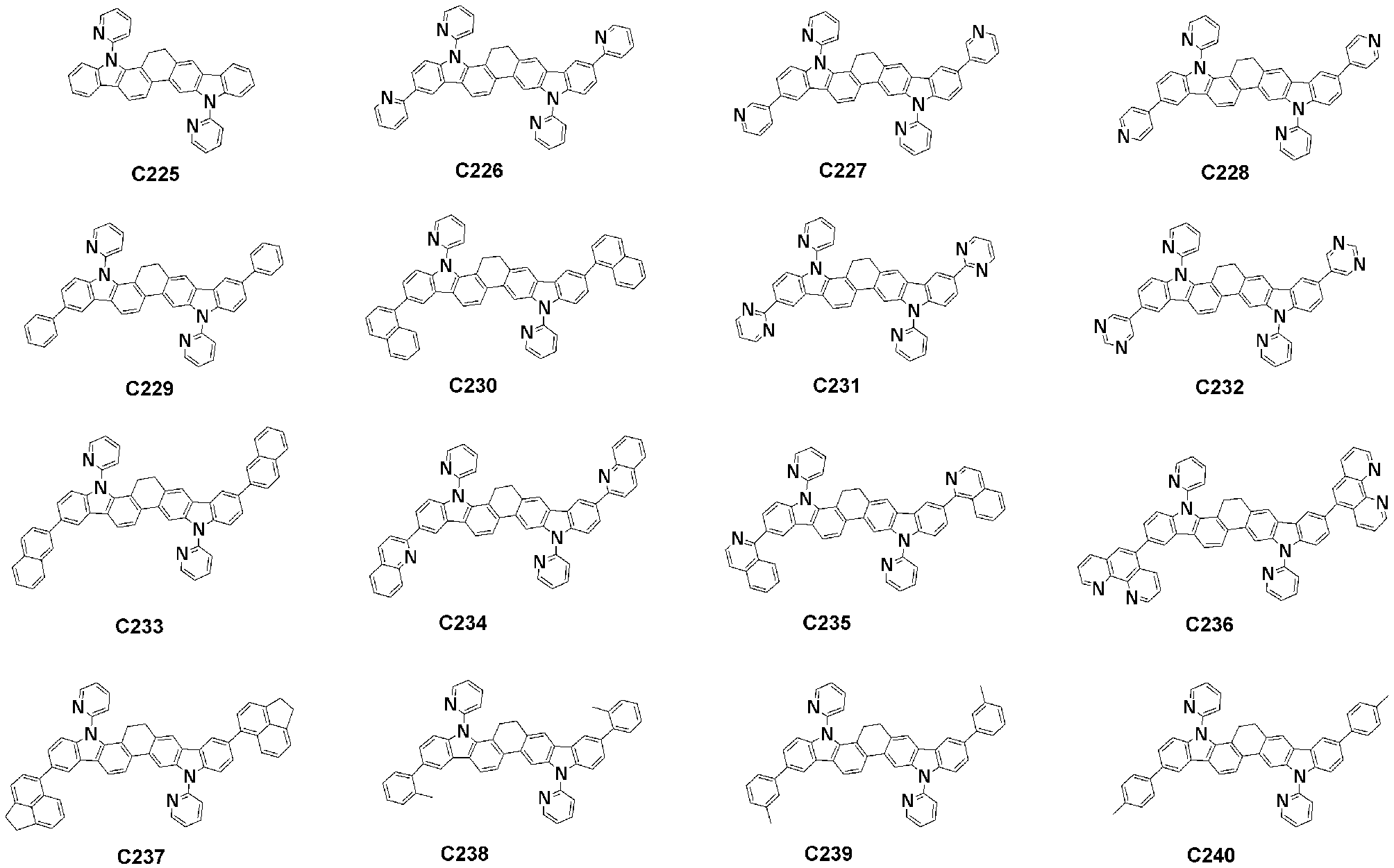
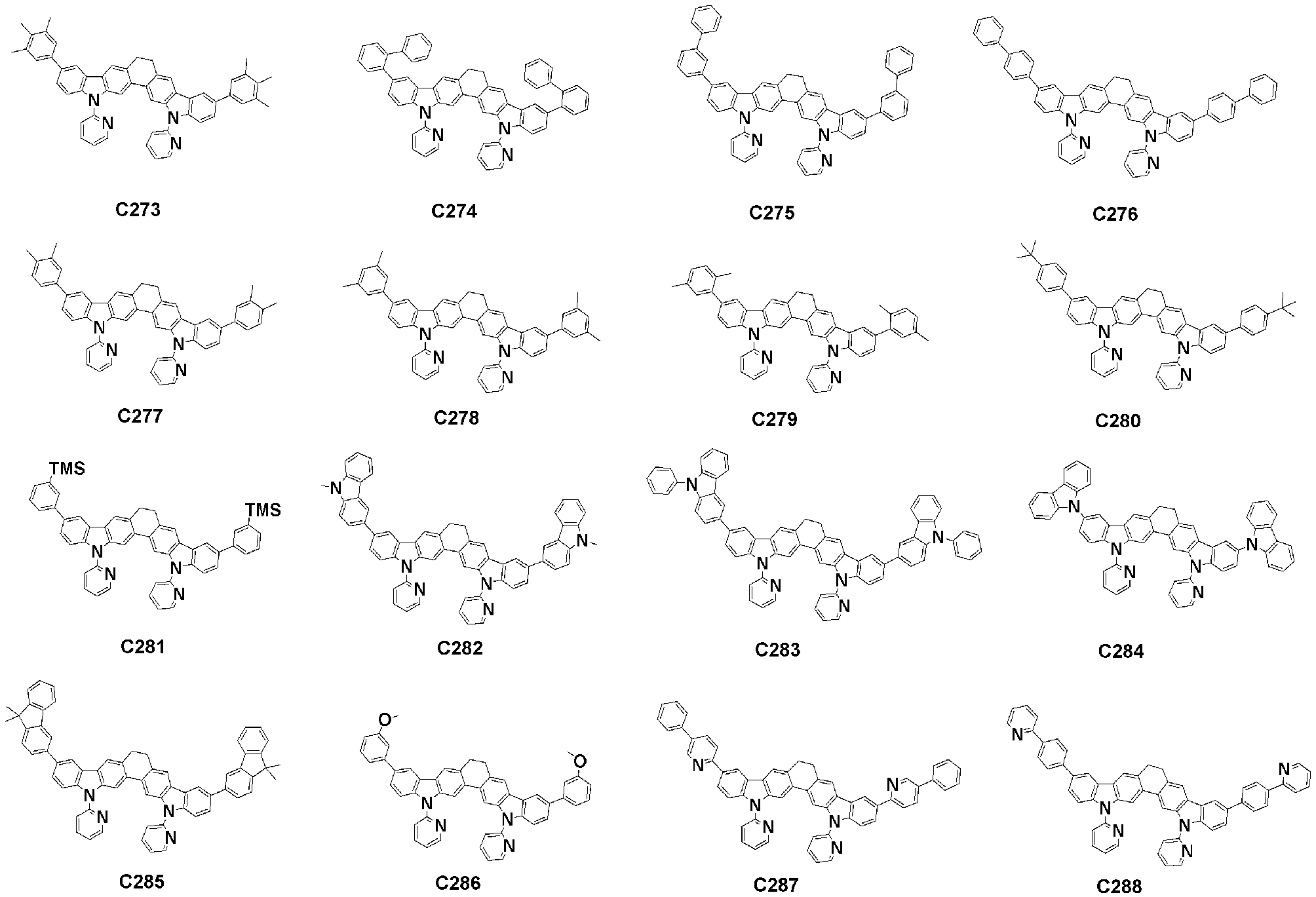
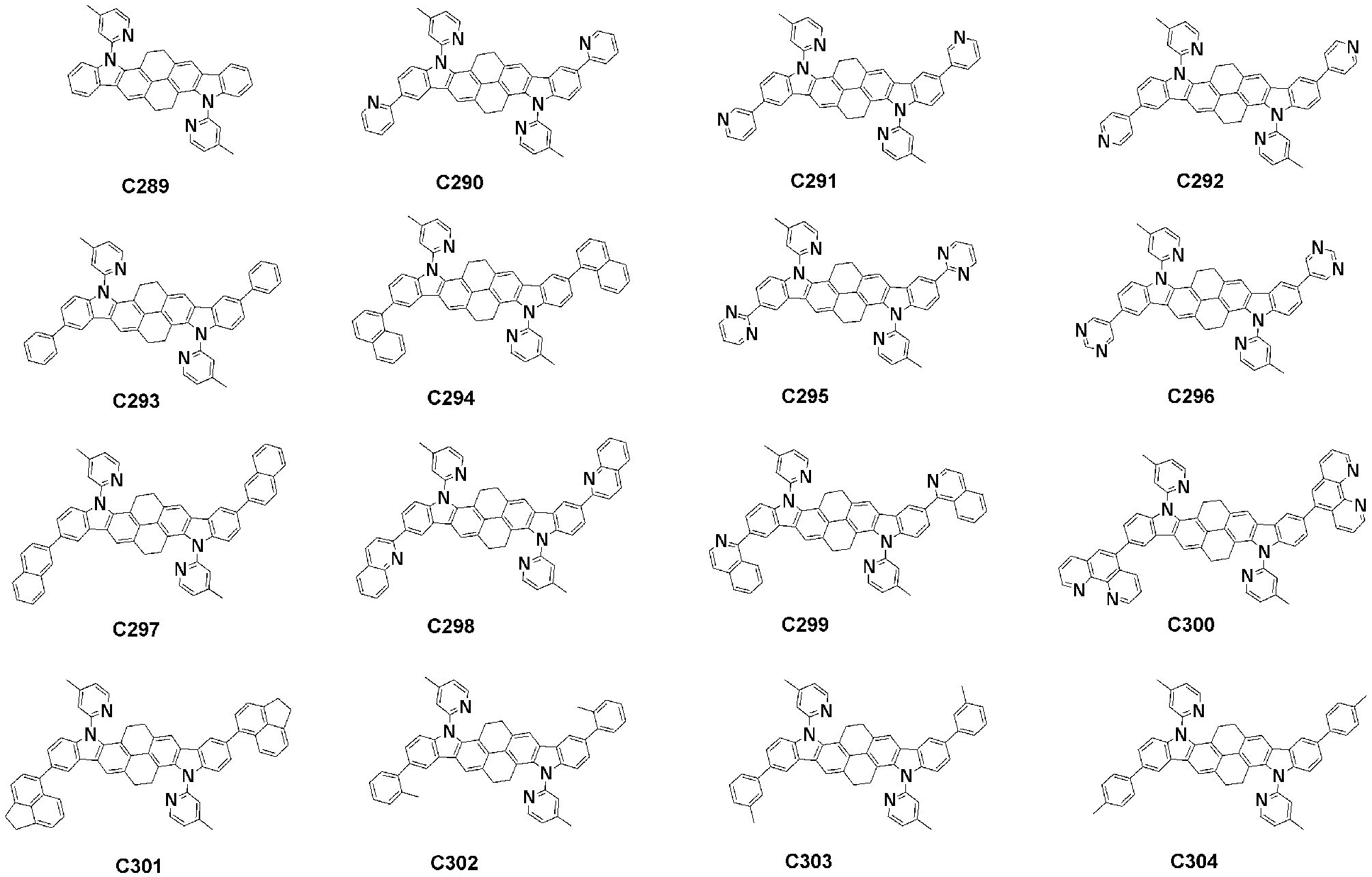
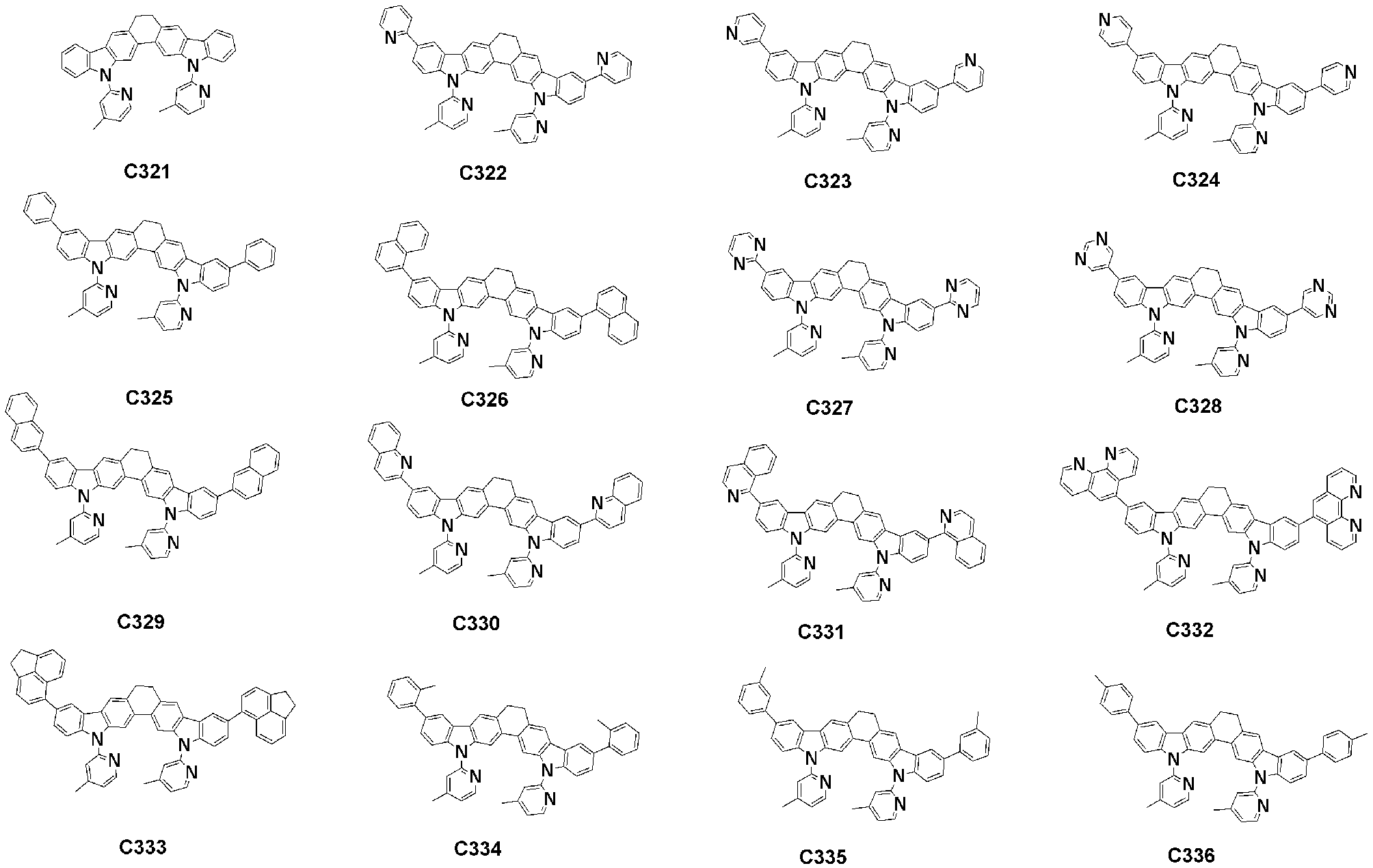
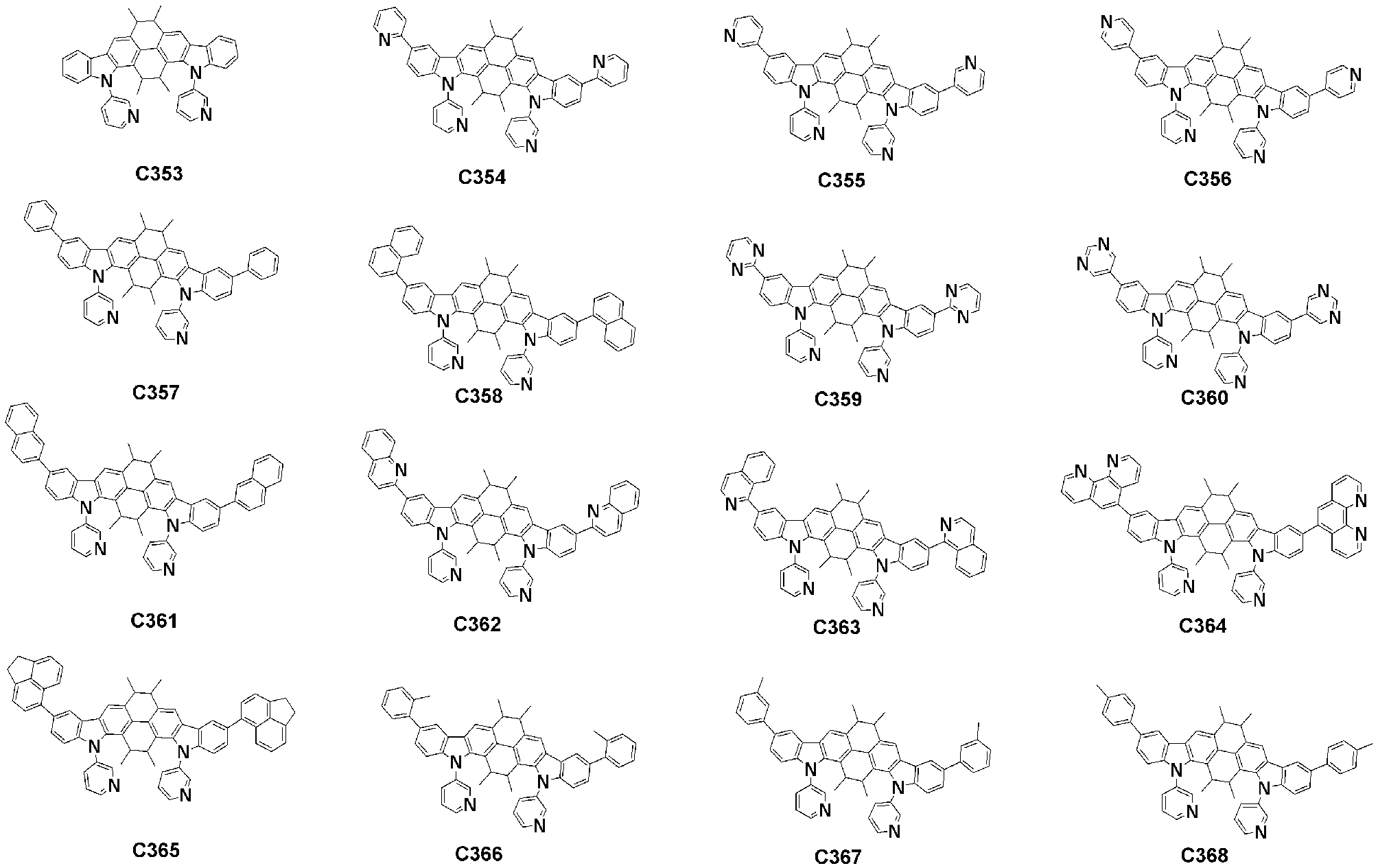
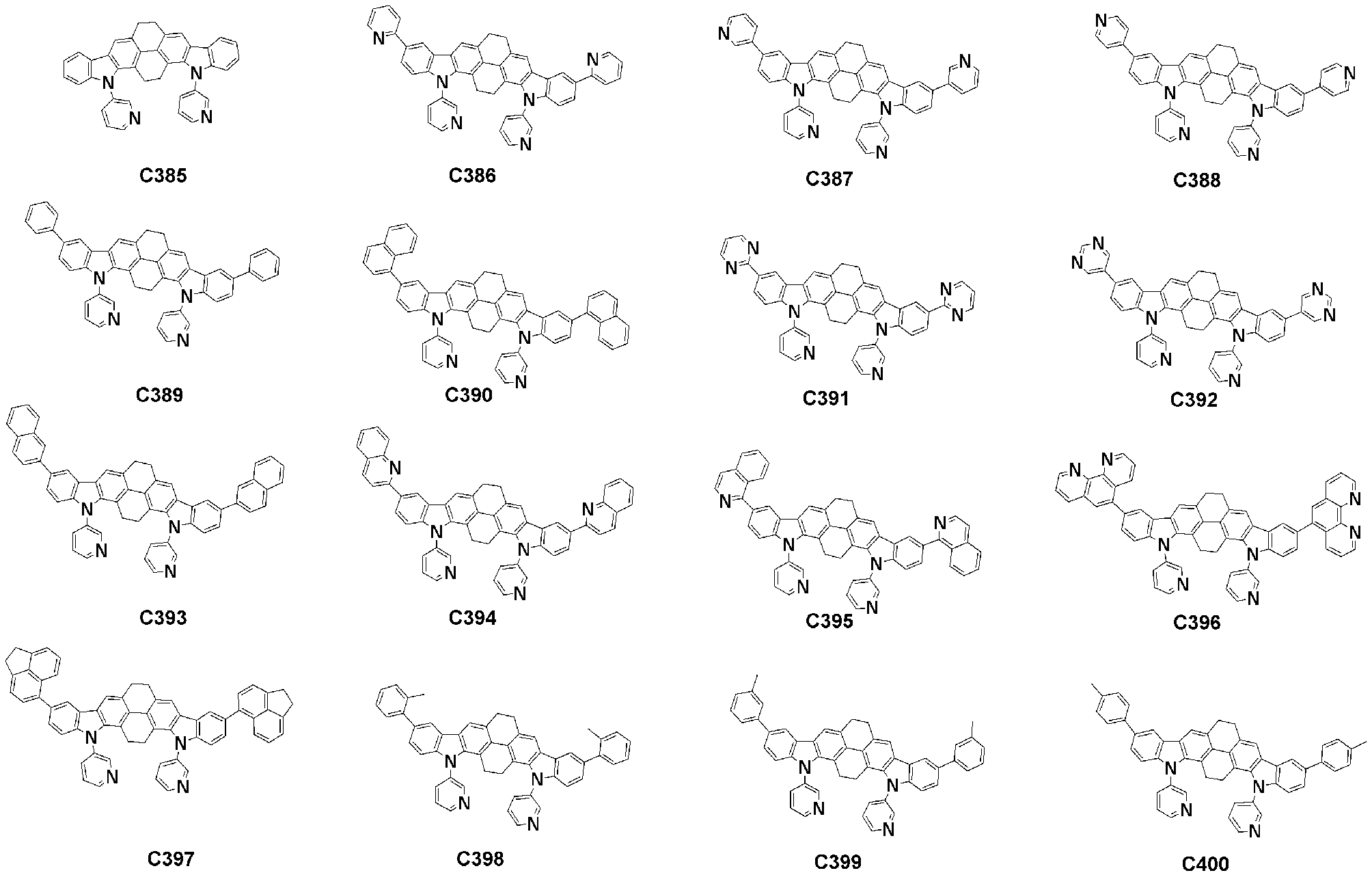
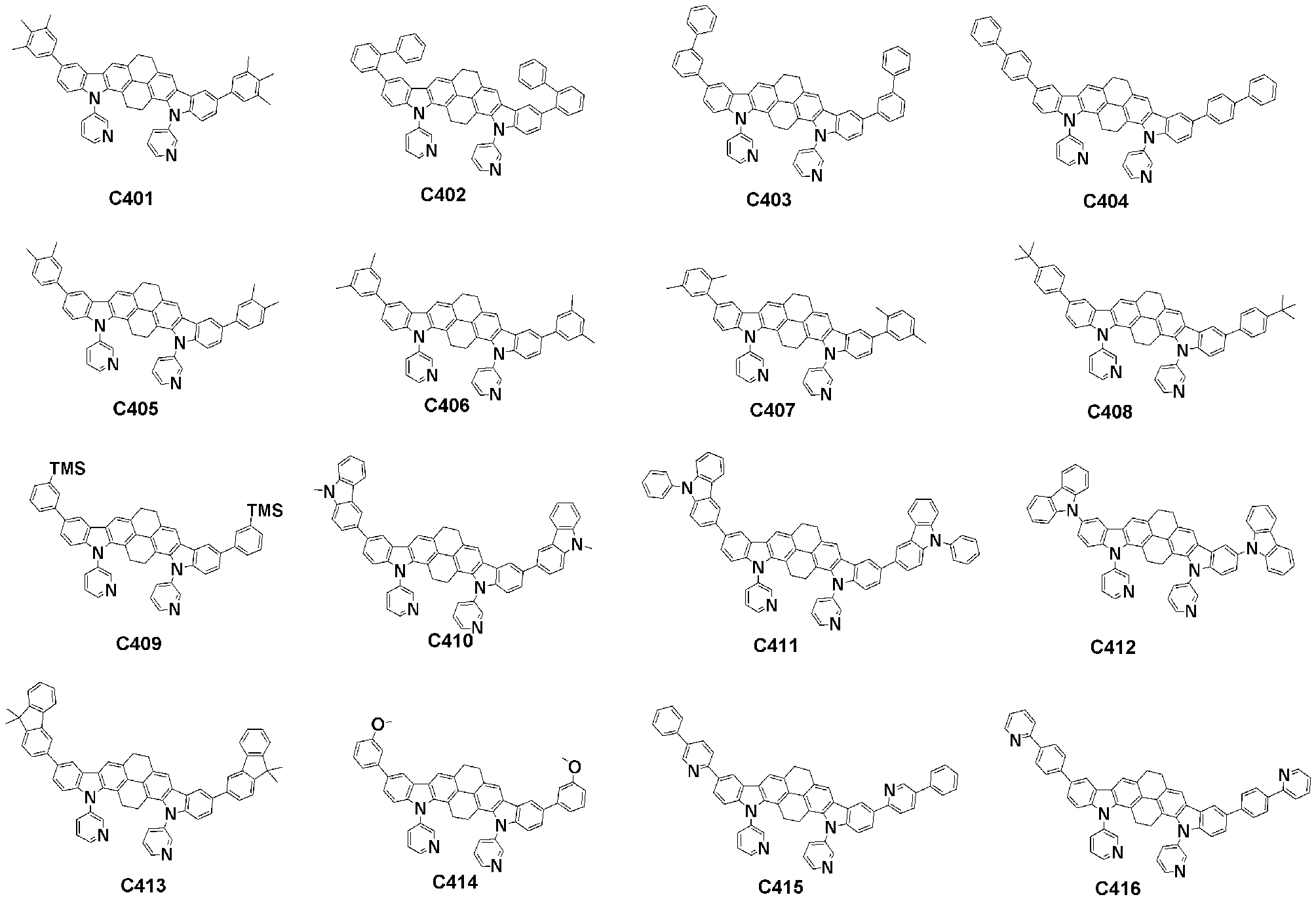
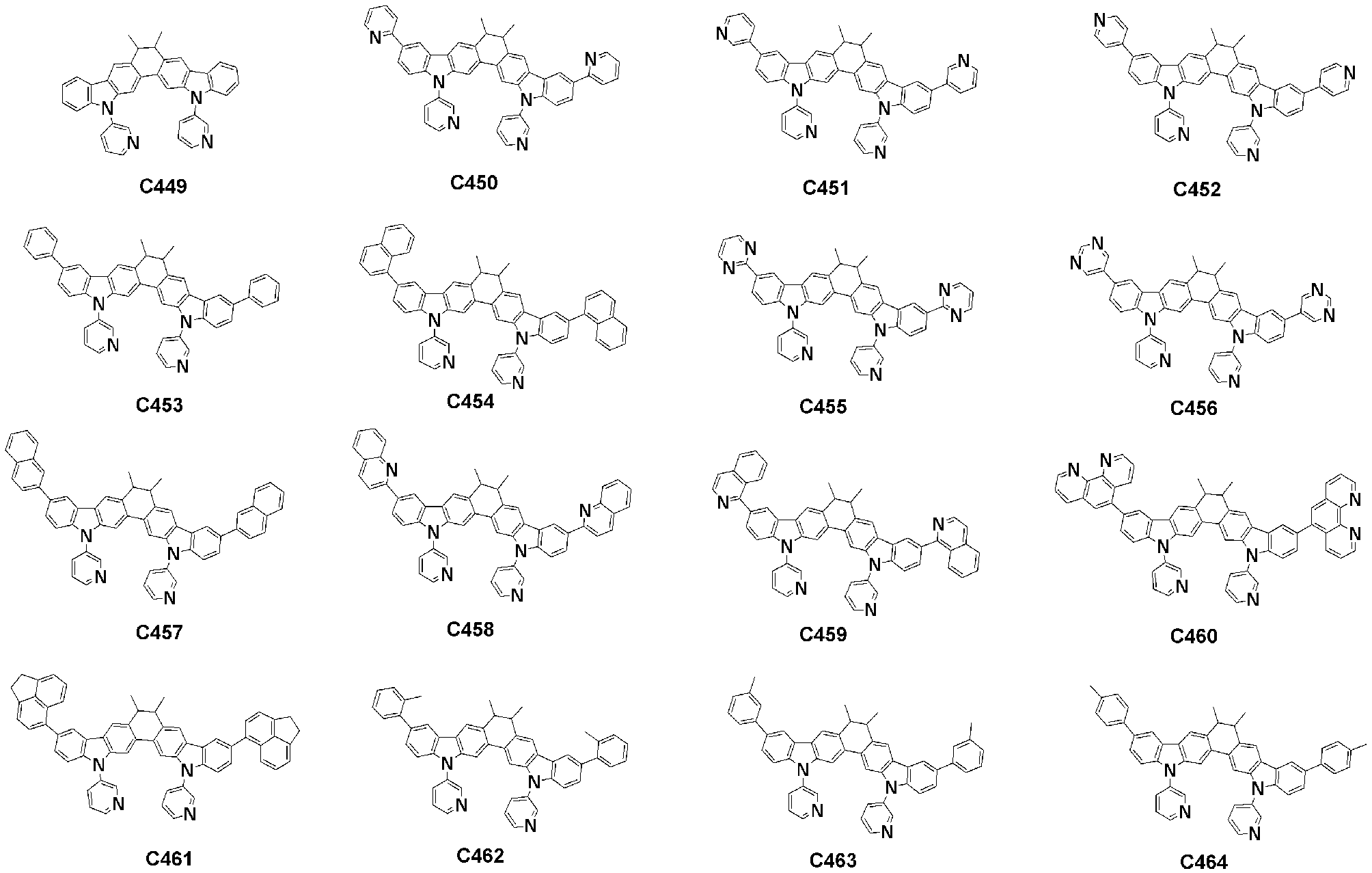
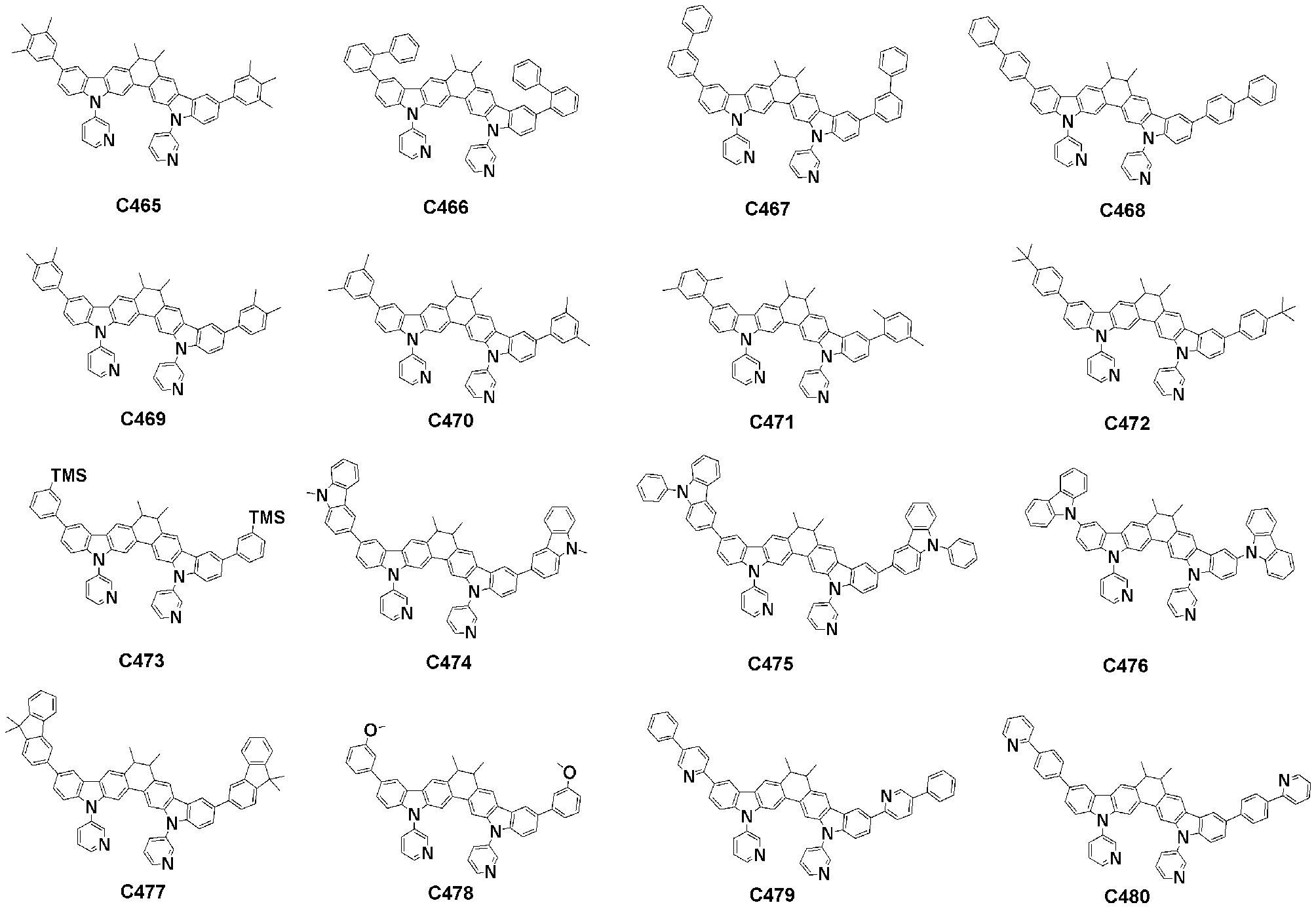
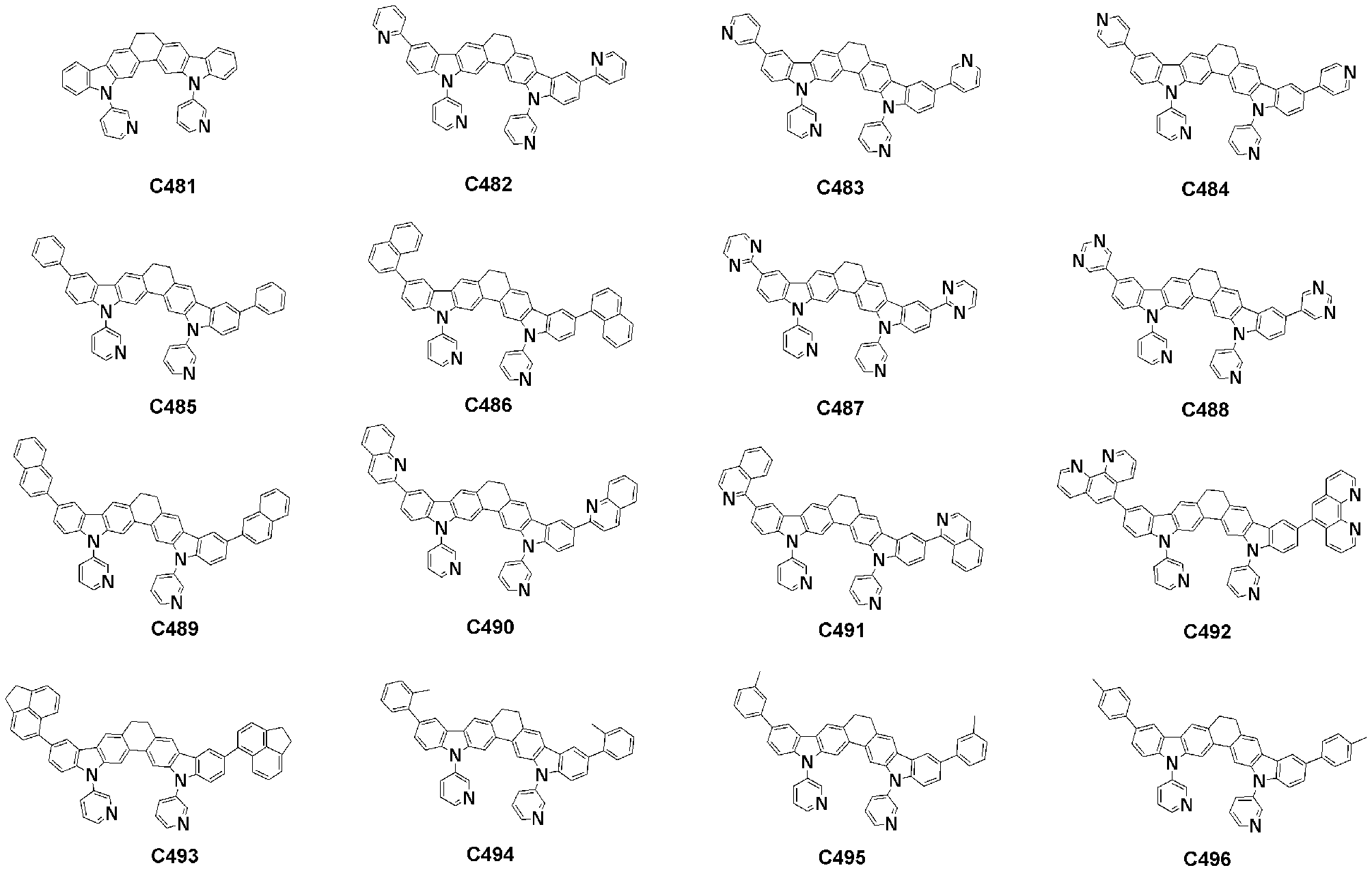
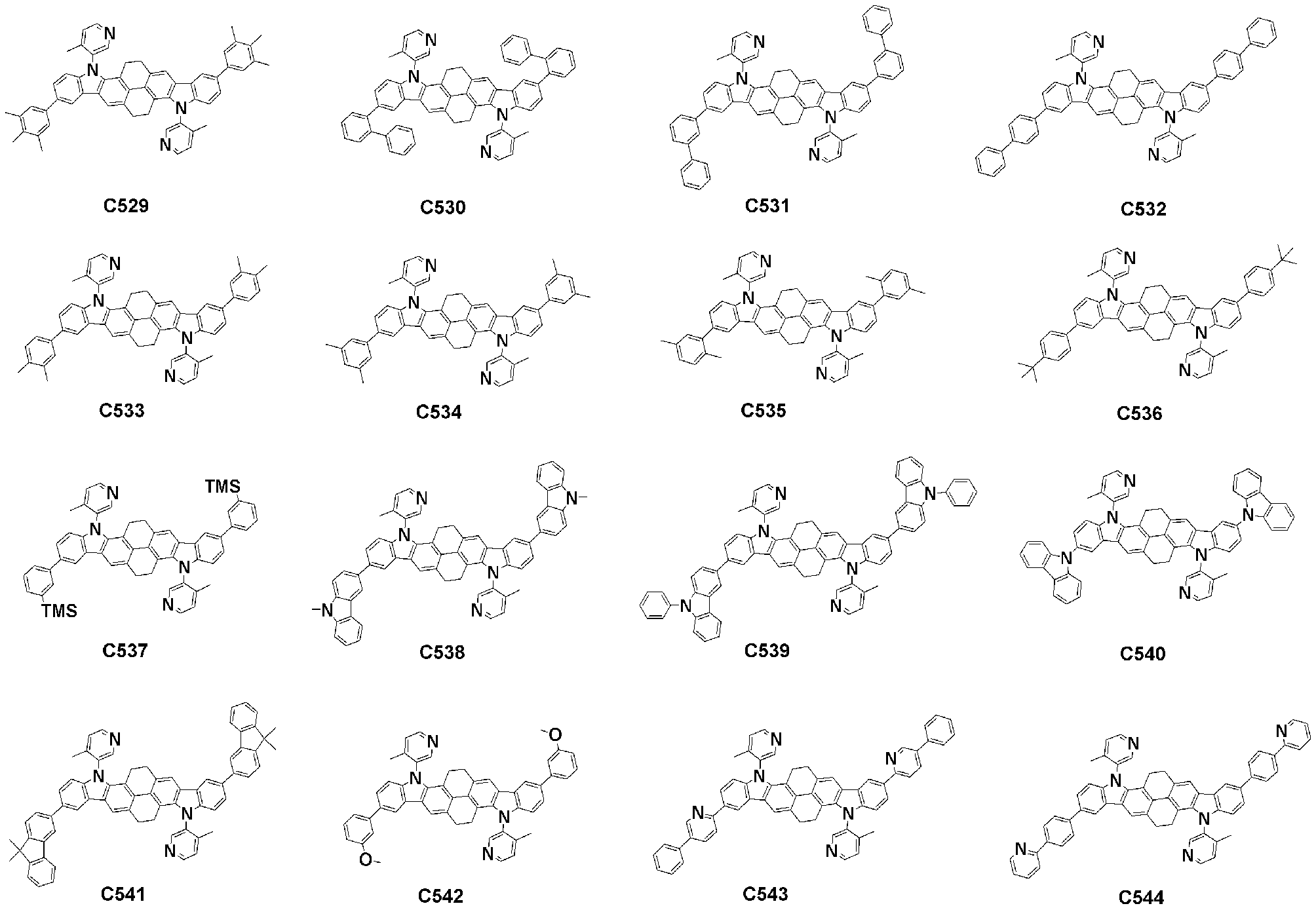
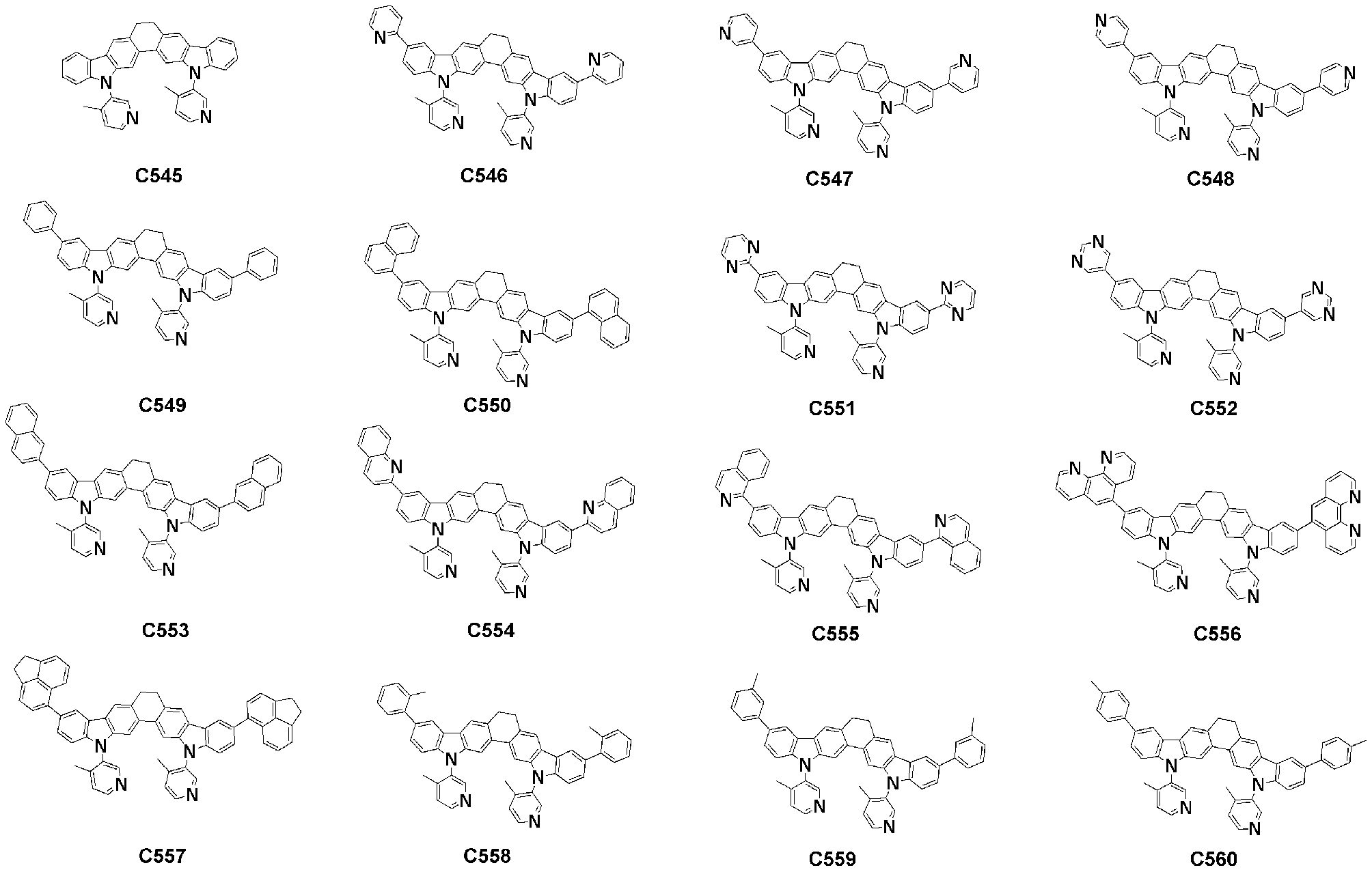
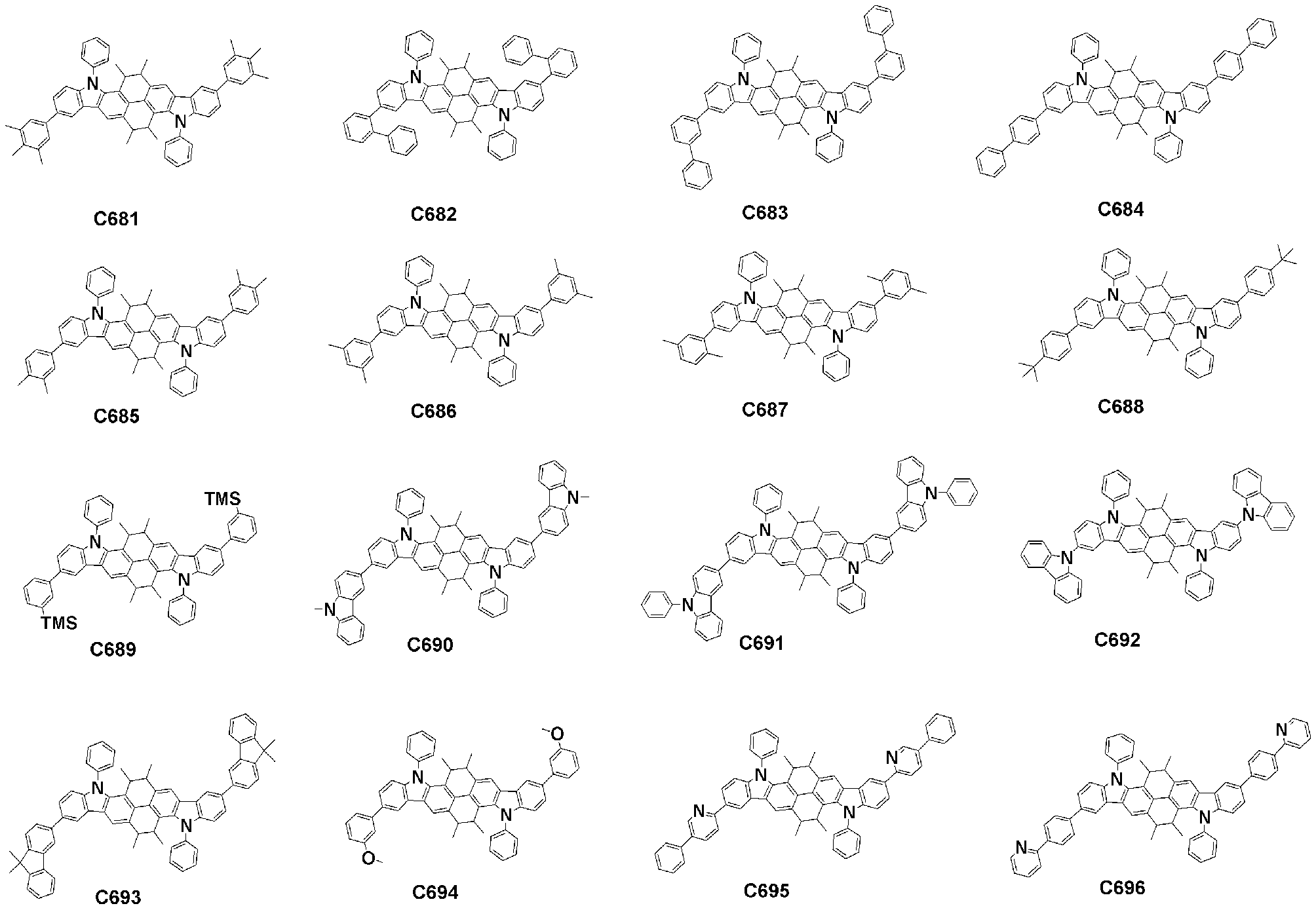
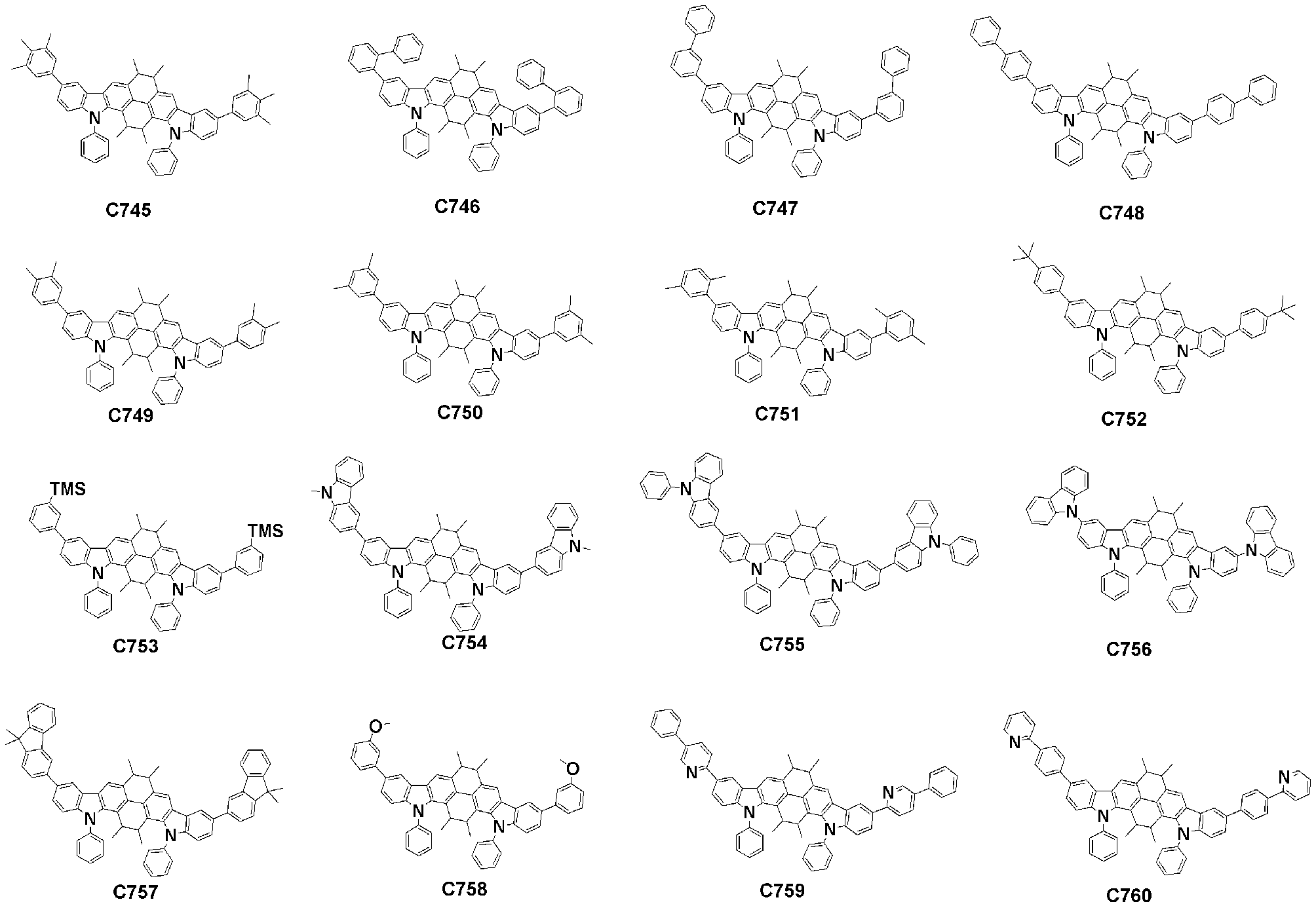
- Specific examples of the compound represented by Formula 1 of the present invention include, but are not limited to, the following compounds (C1-C760).
- Such a compound represented by Formula 1 of the present invention can be synthesized in various ways with reference to the synthesis process of the following examples.
- the present invention provides an organic electroluminescent device comprising an anode, a cathode, and one or more organic material layers interposed between the anode and the cathode, wherein at least one of the one or more organic material layers is represented by Formula 1 above.
- the organic material layer including the compound represented by Formula 1 of the present invention may be any one or more of a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer and an electron injection layer. Specifically, the organic material layer is preferably a light emitting layer.
- the light emitting layer of the organic EL device according to the present invention may contain a host material (preferably a phosphorescent host material), in which case, the compound represented by the formula (1) can be used as the host material.
- a host material preferably a phosphorescent host material
- the compound represented by the formula (1) can be used as the host material.
- the hole transporting ability is increased to increase the bonding force between the holes and the electrons in the light emitting layer, so that the organic light emitting layer has excellent efficiency (emission efficiency and power efficiency), lifetime, luminance and driving voltage.
- An electroluminescent device can be provided.
- the structure of the organic electroluminescent device of the present invention is not particularly limited, but may be formed of a structure in which a substrate, an anode, a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer and a cathode are sequentially stacked.
- the electron injection layer may be further stacked on the electron transport layer.
- the organic electroluminescent device according to the present invention may have a structure in which an anode, one or more organic material layers, and a cathode are sequentially stacked, as well as a structure in which an insulating layer or an adhesive layer is inserted at an interface between the electrode and the organic material layer.
- the material that can be used as the anode included in the organic electroluminescent device of the present invention is not particularly limited, but non-limiting examples include metals such as vanadium, chromium, copper, zinc, gold or alloys thereof; Metal oxides such as zinc oxide, indium oxide, indium tin oxide (ITO), indium zinc oxide (IZO); Combinations of metals and oxides such as ZnO: Al, SnO 2 : Sb; Conductive polymers such as polythiophene, poly (3-methylthiophene), poly [3,4- (ethylene-1,2-dioxy) thiophene] (PEDT), polypyrrole, polyaniline; And carbon black and the like can be used.
- metals such as vanadium, chromium, copper, zinc, gold or alloys thereof
- Metal oxides such as zinc oxide, indium oxide, indium tin oxide (ITO), indium zinc oxide (IZO); Combinations of metals and oxides such as ZnO: Al, SnO 2
- the material that can be used as the cathode included in the organic electroluminescent device of the present invention is not particularly limited, but non-limiting examples include magnesium, calcium, sodium, potassium, titanium, indium, yttrium, lithium, gadolinium, aluminum, silver, tin Metals such as lead or alloys thereof; And multilayer structure materials such as LiF / Al, LiO 2 / Al, and the like.
- the organic material layer included in the organic electroluminescent device of the present invention is in the art, except that the compound represented by Formula 1 is used in any one of a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer and an electron injection layer It may be made of known materials.
- the material usable as the substrate included in the organic electroluminescent device of the present invention is not particularly limited, but non-limiting examples may be used a silicon wafer, quartz, glass plate, metal plate, plastic film and sheet.
- Such an organic electroluminescent device of the present invention can be produced by a method known in the art.
- the light emitting layer included in the organic material layer can be produced by a vacuum deposition method or a solution coating method.
- the solution coating method include, but are not limited to, spin coating, dip coating, doctor blading, inkjet printing, or thermal transfer.
- TPCA-1 (3.4 g, 8.86 mmol), 1-bromobenzene (4.17 g, 26.56 mmol), Cu powder (0.11 g, 1.77 mmol), K 2 CO 3 (2.44 g, 17.71) prepared in Preparation Example 1 under nitrogen stream. mmol), Na 2 SO 4 (2.52 g, 17.71 mmol) and nitrobenzene (100 ml) were mixed and stirred at 190 ° C. for 12 hours.
- TPCA-2 (3.75 g, 6.92 mmol), iodobenzene (4.24 g, 20.76 mmol), Cu powder (0.09 g, 1.38 mmol), K 2 CO 3 (1.91 g, 13.84 mmol) prepared in Preparation Example 2 under a nitrogen stream.
- Na 2 SO 4 (1.97 g, 13.84 mmol)
- nitrobenzene 80 ml
- nitrobenzene was removed, the organic layer was separated with methylene chloride, and water was removed using MgSO 4 . After removing the solvent of the organic layer was purified by column chromatography to give the intermediate compound TPCA-2-Ph (2.74 g, yield 57%).
- the compound synthesized in Synthesis Example 1-30 was subjected to high purity sublimation purification by a method known in the art, and then a green organic EL device was manufactured according to the following procedure.
- an ITO Indium tin oxide
- a solvent such as isopropyl alcohol, acetone, methanol, etc.
- UV OZONE cleaner Power sonic 405, Hwasin Tech
- An organic electroluminescent device was manufactured in the same manner as in Example 1 except for using the following CBP instead of the compound SPC-1 as a light emitting host material when forming the light emitting layer.
- Example 1-30 and Comparative Example 1 For each organic electroluminescent device produced in Example 1-30 and Comparative Example 1, the driving voltage, current efficiency and emission peak at a current density of 10 mA / cm 2 were measured, and the results are shown in Table 1 below.
- Example 1 Device Host Drive voltage (V) Emission Peak (nm) Current efficiency (cd / A)
- SPC-1 6.78 515 42.4 Example 2
- SPC-2 6.81 518 41.1 Example 3
- SPC-4 6.81 515 41.0 Example 5
- SPC-5 6.81 518 41.3 Example 6
- SPC-6 6.77 516 39.4 Example 7
- Example 8 SPC-8 6.80 515 41.1
- Example 9 SPC-9 6.79 518 40.8
- Example 10 SPC-10 6.85 516 41.0
- Example 11 SPC-11 6.77 515 42.0
- Example 12 SPC-12 6.79 518 41.3
- Example 13 SPC-13 6.82 517 41.1
- Example 14 SPC-14 6.83 518 40.8
- Example 15 SPC-15 6.81 516 41.0 Example 16
- SPC-16 6.79 516 41.3 Example 17
- SPC-17 6.87 517 39.4 Example 18
- the compound synthesized in Synthesis Example 21-30 was subjected to high purity sublimation purification by a method known in the art, and then a blue organic EL device was manufactured according to the following procedure.
- an ITO Indium tin oxide
- a solvent such as isopropyl alcohol, acetone, methanol, etc.
- UV OZONE cleaner Power sonic 405
- a blue organic electroluminescent device was manufactured in the same manner as in Example 31, except that CBP used in Comparative Example 1 was used instead of Compound SPC-21 as a light emitting host material when forming the emission layer.
- Example 31 SPC-21 7.55 471 5.99
- Example 32 SPC-22 7.67 472 5.85
- Example 33 SPC-23 7.22 472 6.34
- Example 34 SPC-24 7.12 473 6.90
- Example 35 SPC-25 7.00 474 6.34
- Example 36 SPC-26 7.29 475 6.55
- Example 37 SPC-27 7.30 471 6.94
- Example 38 SPC-28 7.24 472 6.25
- Example 39 SPC-29 7.15 473 6.47
- Example 40 SPC-30 7.23 473 6.94 Comparative Example 2 CBP 7.80 474 5.80
- the efficiency (light emitting efficiency and power efficiency), lifetime, luminance, driving voltage, etc. of the organic light emitting device are improved compared to the conventional light emitting material. You can. Therefore, the present invention can improve the performance and lifespan of a full color organic electroluminescent panel.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Manufacturing & Machinery (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
본 발명은 신규 화합물 및 이를 포함하는 유기 전계 발광 소자에 관한 것으로, 보다 구체적으로는 유기 전계 발광 소자의 유기물층에 사용되는 화합물에 관한 것이다.The present invention relates to a novel compound and an organic electroluminescent device comprising the same, and more particularly to a compound used in the organic material layer of the organic electroluminescent device.
유기 전계 발광 소자는 두 전극 사이에 전압을 걸어주면 양극에서 정공이, 음극에서 전자가 유기물층으로 주입되어, 주입된 정공과 전자가 만나 엑시톤(exciton)이 형성되며, 형성된 엑시톤이 바닥상태로 떨어질 때 빛이 나게 된다. 유기물층으로 사용되는 물질은 기능에 따라, 발광 물질, 정공주입 물질, 정공수송 물질, 전자수송 물질, 전자주입 물질 등으로 분류될 수 있다.In the organic electroluminescent device, when a voltage is applied between two electrodes, holes are injected from the anode and electrons are injected into the organic material layer from the cathode. When the injected holes and the electrons meet to form an exciton, the formed excitons fall to the ground state. The light will shine. The material used as the organic material layer may be classified into a light emitting material, a hole injection material, a hole transport material, an electron transport material, an electron injection material and the like according to a function.
발광 물질은 발광색에 따라 청색, 녹색, 적색의 발광 물질과 천연색을 구현하기 위한 노란색 및 주황색의 발광 물질로 구분될 수 있다. 또한, 색순도의 증가와 에너지 전이를 통해 발광 효율을 증가시키기 위하여, 발광 물질로 호스트/도판트 계를 사용할 수 있다.The light emitting material may be classified into blue, green, and red light emitting materials and yellow and orange light emitting materials for realizing natural colors according to light emitting colors. In addition, in order to increase luminous efficiency through increasing color purity and energy transfer, a host / dopant system may be used as a light emitting material.
도판트 물질은 유기 물질을 사용하는 형광 도판트와 Ir, Pt 등의 중원자(heavy atoms)가 포함된 금속 착체 화합물을 사용하는 인광 도판트로 나눌 수 있다. 인광 도판트의 개발은 이론적으로 형광 도판트에 비해 4배까지 발광 효율을 향상시킬 수 있어 인광 도판트 뿐만 아니라 인광 호스트에 대해서도 연구되고 있다.The dopant material may be divided into a fluorescent dopant using an organic material and a phosphorescent dopant using a metal complex compound containing heavy atoms such as Ir and Pt. The development of phosphorescent dopants is theoretically able to improve luminous efficiency up to four times as compared to fluorescent dopants, so that not only phosphorescent dopants but also phosphorescent hosts are being studied.
현재까지 정공 수송층. 정공 차단층 및 전자 수송층의 물질로는 NPB, BCP, Alq3 등이 사용되고 있으며, 발광 물질로는 안트라센 유도체들이 사용되고 있다. 또한, 발광 물질 중 Firpic, Ir(ppy)3, (acac)Ir(btp)2 등과 같은 Ir을 포함하는 금속 착체 화합물은 blue, green, red의 인광 도판트 재료로, CBP는 인광 호스트 재료로 사용되고 있다. 이외에도 공개특허공보 제2004-0094842호에는 아릴카바졸릴기 또는 카바졸릴알킬렌기에 질소 함유 헤테로환기가 결합한 화합물을 인광 호스트 재료로 사용하는 기술이 개시되어 있다.Hole transport layer to date. NPB, BCP, Alq 3 and the like are used as materials for the hole blocking layer and the electron transport layer, and anthracene derivatives are used as the light emitting material. In addition, metal complex compounds including Ir such as Firpic, Ir (ppy) 3 , and (acac) Ir (btp) 2 among the light emitting materials are phosphorescent dopant materials of blue, green, and red, and CBP is used as a phosphorescent host material. have. In addition, Japanese Patent Laid-Open No. 2004-0094842 discloses a technique of using a compound having a nitrogen-containing heterocyclic group bonded to an arylcarbazolyl group or carbazolylalkylene group as a phosphorescent host material.
그러나 종래의 발광 물질들은 발광 특성이 양호하나, 유리전이온도가 낮고 열적 안정성이 매우 좋지 않아 유기 전계 발광 소자의 수명 측면에서 만족할만한 수준이 되지 못하고 있다. 따라서, 성능이 우수한 발광 물질의 개발이 요구되고 있다.However, the conventional luminescent materials have good luminescence properties, but the glass transition temperature is low and the thermal stability is not very good. Thus, the luminescent materials are not satisfactory in terms of the lifespan of the organic EL device. Therefore, there is a demand for development of a light emitting material having excellent performance.
본 발명은 상기한 문제점을 해결하기 위해, 유기 전계 발광 소자의 효율, 수명 및 안정성 등을 향상시킬 수 있는 신규 화합물 및 상기 화합물을 이용한 유기 전계 발광 소자를 제공하는 것을 목적으로 한다.In order to solve the above problems, an object of the present invention is to provide a novel compound and an organic electroluminescent device using the compound which can improve the efficiency, lifespan and stability of the organic electroluminescent device.
상기한 목적을 달성하기 위해, 본 발명은 하기 화학식 1로 표시되는 화합물을 제공한다.In order to achieve the above object, the present invention provides a compound represented by the following formula (1).
[화학식 1] [Formula 1]
상기 화학식 1에서,In Chemical Formula 1,
A는, , 및 로 이루어진 군에서 선택되고, Ra와 Rb 또는 Rb와 Rc 는 하기 화학식 2로 표시된 축합고리를 형성하며, Ra와 Rb가 축합고리를 형성할 때, Rc는 수소이고, Rb와 Rc가 축합고리를 형성할 때, Ra는 수소이며,A is , And Selected from the group consisting of, R a and R b or R b and R c form a condensed ring represented by the following formula (2), and when R a and R b form a condensed ring, R c is hydrogen and R When b and R c form a condensed ring, R a is hydrogen,
[화학식 2][Formula 2]
상기 Ar1 내지 Ar4는, 각각 독립적으로, C1~C40의 알킬기, C3~C40의 시클로알킬기, C3~C40의 헤테로시클로알킬기, C6~C60의 아릴기, C5~C60의 헤테로아릴기, C1~C40의 알킬옥시기, C6~C60의 아릴옥시기 및 C6~C60의 아릴아민기로 이루어진 군에서 선택되고,Ar 1 to Ar 4 are each independently a C 1 to C 40 alkyl group, C 3 to C 40 cycloalkyl group, C 3 to C 40 heterocycloalkyl group, C 6 to C 60 aryl group, C 5 Is selected from the group consisting of -C 60 heteroaryl group, C 1 -C 40 alkyloxy group, C 6 -C 60 aryloxy group, and C 6 -C 60 arylamine group,
상기 R1 내지 R4는, 각각 독립적으로, 수소, 할로겐, C1~C40의 알킬기, C3~C40의 시클로알킬기, C3~C40의 헤테로시클로알킬기, C6~C60의 아릴기, C5~C60의 헤테로아릴기, C1~C40의 알킬옥시기, C6~C60의 아릴옥시기 및 C6~C60의 아릴아민기로 이루어진 군에서 선택되며,R 1 to R 4 are each independently hydrogen, halogen, C 1 -C 40 alkyl group, C 3 -C 40 cycloalkyl group, C 3 -C 40 heterocycloalkyl group, C 6 -C 60 aryl Group, C 5 ~ C 60 heteroaryl group, C 1 ~ C 40 alkyloxy group, C 6 ~ C 60 aryloxy group and C 6 ~ C 60 arylamine group,
상기 n, m은 각각 0 내지 4의 정수이다N and m are each an integer of 0 to 4
여기서, 상기 Ar1 내지 Ar4의 알킬기, 시클로알킬기, 헤테로시클로알킬기, 아릴기, 헤테로아릴기, 알킬옥시기, 아릴옥시기 및 아릴아민기는 각각 독립적으로, 수소, C1~C40의 알킬기, C2~C40의 알케닐기, C2~C40의 알키닐기, C6~C40의 아릴기, C5~C40의 헤테로아릴기, C6~C40의 아릴옥시기, C1~C40의 알킬옥시기, C6~C40의 아릴아미노기, C6~C40의 디아릴아미노기, C6~C40의 아릴알킬기, C3~C40의 시클로알킬기, C6~C40의 아릴실릴기 및 C3~C40의 헤테로시클로알킬기로 이루어진 군에서 선택된 1종 이상의 치환기로 치환될 수 있다.Here, the alkyl group, the cycloalkyl group, the heterocycloalkyl group, the aryl group, the heteroaryl group, the alkyloxy group, the aryloxy group and the arylamine group of Ar 1 to Ar 4 are each independently hydrogen, an alkyl group of C 1 to C 40 , aryloxy group of C 2 ~ C 40 alkenyl group, C 2 ~ C 40 alkynyl group, C 6 ~ C 40 aryl group, C 5 ~ C 40 heteroaryl group, C 6 ~ C 40 of, C 1 ~ C 40 alkyloxy group, C 6 ~ C 40 arylamino group, C 6 ~ C 40 diarylamino group, C 6 ~ C 40 arylalkyl group, C 3 ~ C 40 cycloalkyl group, C 6 ~ C 40 It may be substituted with one or more substituents selected from the group consisting of an arylsilyl group and a C 3 to C 40 heterocycloalkyl group.
본 발명의 알킬은 탄소수 1 내지 40의 직쇄 또는 측쇄의 포화 탄화수소를 의미하며, 그 예로는 메틸, 에틸, 프로필, 이소부틸, sec-부틸, 펜틸, iso-아밀, 헥실 등을 들 수 있다.Alkyl of the present invention means a straight chain or branched saturated hydrocarbon having 1 to 40 carbon atoms, and examples thereof include methyl, ethyl, propyl, isobutyl, sec-butyl, pentyl, iso-amyl, hexyl and the like.
본 발명의 아릴은 단독 고리 또는 2 이상의 고리가 조합된 탄소수 6 내지 60의 방향족 부위를 의미하며, 2 이상의 고리가 서로 단순 부착되거나 축합된 형태일 수 있다.Aryl of the present invention means an aromatic moiety having 6 to 60 carbon atoms combined with a single ring or two or more rings, and two or more rings may be in the form of a simple attachment or condensation with each other.
본 발명의 헤테로아릴은 핵원자수 5 내지 60의 모노헤테로사이클릭 또는 폴리헤테로사이클릭 방향족 부위를 의미하며, 고리 중 하나 이상의 탄소, 바람직하게는 1 내지 3개의 탄소가 N, O, S 또는 Se와 같은 헤테로원자로 치환된 것을 의미한다. 여기서, 헤테로아릴은 2 이상의 고리가 서로 단순 부착되거나 축합된 형태일 수 있으며, 아릴기와의 축합된 형태도 포함될 수 있다.Heteroaryl of the present invention means a monoheterocyclic or polyheterocyclic aromatic moiety having 5 to 60 nuclear atoms, wherein at least one carbon in the ring, preferably 1 to 3 carbons is N, O, S or Se Mean substituted with a hetero atom such as. Here, the heteroaryl may be in a form in which two or more rings are simply attached or condensed with each other, and a condensed form with an aryl group may also be included.
본 발명의 축합고리는 축합 지방족 고리, 축합 방향족 고리, 축합 헤테로지방족 고리, 축합 헤테로방향족 고리 또는 이들의 조합된 형태를 의미한다.The condensed ring of the present invention means a condensed aliphatic ring, a condensed aromatic ring, a condensed heteroaliphatic ring, a condensed heteroaromatic ring or a combination thereof.
한편, 본 발명은, 양극, 음극 및 상기 양극과 음극 사이에 개재(介在)된 1층 이상의 유기물층을 포함하는 유기 전계 발광 소자에 있어서, 상기 유기물층 중 적어도 하나는 상기 화학식 1로 표시되는 화합물을 포함하는 유기물층인 것을 특징으로 하는 유기 전계 발광 소자를 제공한다.On the other hand, the present invention, in the organic electroluminescent device comprising an anode, a cathode and one or more organic material layer interposed between the anode and the cathode, at least one of the organic material layer comprises a compound represented by the formula (1) It provides an organic electroluminescent device characterized in that the organic material layer.
이하, 본 발명을 상세히 설명한다.Hereinafter, the present invention will be described in detail.
1. 신규 화합물1. New Compound
본 발명에 따른 신규 화합물은 테트라하이드로파이렌 또는 테트라하이드로페난트렌의 양말단에 인돌기가 결합되어 기본 골격을 이루며, 다양한 치환체가 결합된 화합물로 상기 화학식 1로 표시된다. 이러한, 본 발명의 화학식 1로 표시되는 화합물은 다양한 치환체(상기 화학식 1에서 R1 내지 R4 및 Ar1 내지 Ar4)가 결합되어 에너지 레벨이 조절됨으로써 종래의 유기 발광 소자용 재료(예를 들어, CBP(4,4-dicarbazolybiphenyl))보다 높은 분자량을 가지며, 넓은 에너지 밴드갭을 나타내는 것이 특징이다.The novel compound according to the present invention forms a basic skeleton by binding an indole group to the sock end of tetrahydropyrene or tetrahydrophenanthrene, and is represented by Formula 1 as a compound in which various substituents are bonded. Such a compound represented by Formula 1 of the present invention is a conventional organic light emitting device material (for example, by adjusting the energy level by combining a variety of substituents (R 1 to R 4 and Ar 1 to Ar 4 in Formula 1) It has a higher molecular weight than CBP (4,4-dicarbazolybiphenyl)) and is characterized by a wide energy bandgap.
또한, 다양한 치환체가 도입된 본 발명의 화학식 1로 표시되는 화합물은 분자량이 유의적으로 증대됨으로써, 유리전이온도가 향상되고 이로 인해 종래의 재료들에 비해 높은 열적 안정성을 가질 수 있다.In addition, the compound represented by the formula (1) of the present invention in which various substituents are introduced has a significant increase in molecular weight, thereby improving the glass transition temperature, thereby having a high thermal stability compared to conventional materials.
따라서, 본 발명의 화학식 1로 표시되는 화합물을 유기 전계 발광 소자의 재료로 사용할 경우 소자의 인광특성뿐만 아니라, 전자 및/또는 정공 수송 능력, 발광효율, 구동전압, 수명 특성 등이 개선될 수 있다. 이때, 본 발명의 화학식 1로 표시되는 화합물은 유기 전계 발광 소자의 유기물층의 재료, 바람직하게는 발광층, 정공수송층 또는 전자수송층의 재료, 보다 바람직하게는 발광층의 호스트 재료로 사용될 수 있다.Therefore, when the compound represented by Chemical Formula 1 of the present invention is used as a material of an organic electroluminescent device, not only phosphorescence properties of the device, but also electron and / or hole transporting ability, luminous efficiency, driving voltage, and lifetime characteristics may be improved. . In this case, the compound represented by Formula 1 of the present invention may be used as a material of the organic material layer of the organic electroluminescent device, preferably a light emitting layer, a hole transporting layer or an electron transporting layer, more preferably a host material of the light emitting layer.
이러한 본 발명의 화학식 1로 표시되는 화합물은 하기 화학식 3 내지 6으로 표시되는 화합물로 이루어진 군에서 선택되는 것이 바람직하다.The compound represented by the formula (1) of the present invention is preferably selected from the group consisting of the compound represented by the following formula (3 to 6).
[화학식 3] [Formula 3]
[화학식 4][Formula 4]
[화학식 5][Formula 5]
[화학식 6][Formula 6]
상기 화학식 3 내지 6에서, Ar1 내지 Ar4 및 R1 내지 R4에 대해서는 상기 화학식 1에 대해서 설명한 바와 동일하다.In Formulas 3 to 6, Ar 1 to Ar 4 and R 1 to R 4 are the same as those described for Formula 1 above.
여기서, 유기 전계 발광 소자의 수명, 발광효율, 구동전압 등을 고려할 때 상기 화학식 3 내지 6으로 표시되는 화합물의 Ar1 내지 Ar4는 각각 독립적으로, 수소, C1~C40의 알킬기, C6~C60의 아릴기 및 C5~C60의 헤테로아릴기로 이루어진 군에서 선택되는 것이 바람직하며, 수소, 메틸(methyl), 페닐(phenyl), 피리딘(pyridine), 피리미딘(pyrimidine), 1,3,5-트리아진(1,3,5- triazine), 나프탈렌(naphthalene), 퀴놀린(quinoline), 1,10-페난트롤린(1, 10-phenanthroline), 아세나프탈렌(acenaphthylene), 바이페닐(biphenyl), 플루오렌(fluorine) 및 9H-카바졸(9H- carbazole)로 이루어진 군에서 선택되는 것이 더욱 바람직하다.Here, Ar 1 to Ar 4 of the compounds represented by Formulas 3 to 6 are each independently hydrogen, an alkyl group of C 1 to C 40 , C 6 in consideration of the lifespan, luminous efficiency, driving voltage, etc. of the organic EL device. It is preferably selected from the group consisting of an aryl group of ~ C 60 and a heteroaryl group of C 5 ~ C 60 , hydrogen, methyl, phenyl, pyridine, pyrimidine, 1, 3,5-triazine, naphthalene, quinoline, 1,10-phenanthroline, acenaphthylene, biphenyl more preferably selected from the group consisting of biphenyl), fluorine and 9H-carbazole.
또한, 상기 화학식 3 내지 6으로 표시되는 화합물의 R1 내지 R4는 각각 독립적으로, 수소 또는 메틸인 것이 바람직하다.In addition, R 1 to R 4 of the compounds represented by Formulas 3 to 6 are each independently hydrogen or methyl.
이러한 본 발명의 화학식 1로 표시되는 화합물의 구체적인 예로 하기 화합물들(C1-C760)을 들 수 있지만, 이에 한정되는 것은 아니다.Specific examples of the compound represented by Formula 1 of the present invention include, but are not limited to, the following compounds (C1-C760).
이와 같은 본 발명의 화학식 1로 표시되는 화합물은 하기 실시예의 합성과정을 참고하여 다양하게 합성할 수 있다.Such a compound represented by Formula 1 of the present invention can be synthesized in various ways with reference to the synthesis process of the following examples.
2. 유기 전계 발광 소자2. Organic electroluminescent device
본 발명은 양극, 음극, 및 상기 양극과 음극 사이에 개재(介在)된 1층 이상의 유기물층을 포함하는 유기 전계 발광 소자를 제공하는데, 이때, 상기 1층 이상의 유기물층 중 적어도 하나는 상기에서 설명한 화학식 1로 표시되는 화합물, 바람직하게는, 상기 화학식 3 내지 6으로 표시되는 화합물을 포함하는 유기물층인 것을 특징으로 한다. 이때, 상기 화학식 3 내지 6으로 표시되는 화합물은 단독 또는 2종 이상 포함될 수 있다.The present invention provides an organic electroluminescent device comprising an anode, a cathode, and one or more organic material layers interposed between the anode and the cathode, wherein at least one of the one or more organic material layers is represented by Formula 1 above. Compounds represented by, preferably, characterized in that the organic material layer containing a compound represented by the formula (3 to 6). In this case, the compounds represented by Formulas 3 to 6 may be included alone or two or more.
본 발명의 화학식 1로 표시되는 화합물을 포함하는 유기물층은 정공주입층, 정공수송층, 발광층, 전자수송층 및 전자주입층 중 어느 하나 이상일 수 있다. 구체적으로, 상기 유기물층은 발광층인 것이 바람직하다.The organic material layer including the compound represented by Formula 1 of the present invention may be any one or more of a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer and an electron injection layer. Specifically, the organic material layer is preferably a light emitting layer.
본 발명에 따른 유기 전계 발광 소자의 발광층은 호스트 재료(바람직하게는, 인광 호스트 재료)를 함유할 수 있는데, 이때, 호스트 재료로 상기 화학식 1로 표시되는 화합물을 사용할 수 있는 것이다. 이와 같이 발광층이 상기 화학식 1로 표시되는 화합물을 함유할 경우 정공 수송 능력이 증가되어 발광층에서 정공과 전자의 결합력이 높아지기 때문에 효율(발광효율 및 전력효율), 수명, 휘도 및 구동전압 등이 우수한 유기 전계 발광 소자를 제공할 수 있다.The light emitting layer of the organic EL device according to the present invention may contain a host material (preferably a phosphorescent host material), in which case, the compound represented by the formula (1) can be used as the host material. As such, when the light emitting layer contains the compound represented by Chemical Formula 1, the hole transporting ability is increased to increase the bonding force between the holes and the electrons in the light emitting layer, so that the organic light emitting layer has excellent efficiency (emission efficiency and power efficiency), lifetime, luminance and driving voltage. An electroluminescent device can be provided.
이러한 본 발명의 유기 전계 발광 소자의 구조는 특별히 한정되지 않으나, 비제한적인 예로 기판, 양극, 정공주입층, 정공수송층, 발광층, 전자수송층 및 음극이 순차적으로 적층된 구조로 이루어질 수 있다. 여기서, 전자수송층 위에는 전자주입층이 추가로 적층될 수도 있다. 또한, 본 발명에 따른 유기 전계 발광 소자는 양극, 1층 이상의 유기물층 및 음극이 순차적으로 적층된 구조뿐만 아니라, 전극과 유기물층 계면에 절연층 또는 접착층이 삽입된 구조로 이루어질 수 있다.The structure of the organic electroluminescent device of the present invention is not particularly limited, but may be formed of a structure in which a substrate, an anode, a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer and a cathode are sequentially stacked. Here, the electron injection layer may be further stacked on the electron transport layer. In addition, the organic electroluminescent device according to the present invention may have a structure in which an anode, one or more organic material layers, and a cathode are sequentially stacked, as well as a structure in which an insulating layer or an adhesive layer is inserted at an interface between the electrode and the organic material layer.
한편, 본 발명의 유기 전계 발광 소자에 포함되는 양극으로 사용 가능한 물질은 특별히 한정되지 않으나, 비제한적인 예로 바나듐, 크롬, 구리, 아연, 금과 같은 금속 또는 이들의 합금; 아연산화물, 인듐산화물, 인듐주석산화물(ITO), 인듐아연산화물(IZO)과 같은 금속 산화물; ZnO:Al, SnO2:Sb와 같은 금속과 산화물의 조합; 폴리티오펜, 폴리(3-메틸티오펜), 폴리[3,4-(에틸렌-1,2-디옥시)티오펜](PEDT), 폴리피롤, 폴리아닐린과 같은 전도성 고분자; 및 카본블랙 등을 사용할 수 있다.On the other hand, the material that can be used as the anode included in the organic electroluminescent device of the present invention is not particularly limited, but non-limiting examples include metals such as vanadium, chromium, copper, zinc, gold or alloys thereof; Metal oxides such as zinc oxide, indium oxide, indium tin oxide (ITO), indium zinc oxide (IZO); Combinations of metals and oxides such as ZnO: Al, SnO 2 : Sb; Conductive polymers such as polythiophene, poly (3-methylthiophene), poly [3,4- (ethylene-1,2-dioxy) thiophene] (PEDT), polypyrrole, polyaniline; And carbon black and the like can be used.
또한, 본 발명의 유기 전계 발광 소자에 포함되는 음극으로 사용 가능한 물질은 특별히 한정되지 않으나, 비제한적인 예로 마그네슘, 칼슘, 나트륨, 칼륨, 타이타늄, 인듐, 이트륨, 리튬, 가돌리늄, 알루미늄, 은, 주석, 납과 같은 금속 또는 이들의 합금; 및 LiF/Al, LiO2/Al과 같은 다층 구조 물질 등을 사용할 수 있다.In addition, the material that can be used as the cathode included in the organic electroluminescent device of the present invention is not particularly limited, but non-limiting examples include magnesium, calcium, sodium, potassium, titanium, indium, yttrium, lithium, gadolinium, aluminum, silver, tin Metals such as lead or alloys thereof; And multilayer structure materials such as LiF / Al, LiO 2 / Al, and the like.
또, 본 발명의 유기 전계 발광 소자에 포함되는 유기물층은 상기 화학식 1로 표시되는 화합물을 정공주입층, 정공수송층, 발광층, 전자수송층 및 전자주입층 중 어느 하나에 사용하는 것을 제외하고는 당업계에 공지된 물질로 이루어질 수 있다.In addition, the organic material layer included in the organic electroluminescent device of the present invention is in the art, except that the compound represented by Formula 1 is used in any one of a hole injection layer, a hole transport layer, a light emitting layer, an electron transport layer and an electron injection layer It may be made of known materials.
본 발명의 유기 전계 발광 소자에 포함되는 기판으로 사용 가능한 물질은 특별히 한정되지 않으나, 비제한적인 예로 실리콘 웨이퍼, 석영, 유리판, 금속판, 플라스틱 필름 및 시트 등이 사용될 수 있다. The material usable as the substrate included in the organic electroluminescent device of the present invention is not particularly limited, but non-limiting examples may be used a silicon wafer, quartz, glass plate, metal plate, plastic film and sheet.
이와 같은 본 발명의 유기 전계 발광 소자는 당업계에 공지된 방법으로 제조할 수 있다. 한편, 유기물층에 포함되는 발광층은 진공 증착법이나 용액 도포법으로 제조할 수 있다. 여기서, 용액 도포법의 예로는 스핀 코팅, 딥코팅, 닥터 블레이딩, 잉크젯 프린팅 또는 열 전사법 등이 있으나, 이들에 한정되지 않는다.Such an organic electroluminescent device of the present invention can be produced by a method known in the art. On the other hand, the light emitting layer included in the organic material layer can be produced by a vacuum deposition method or a solution coating method. Here, examples of the solution coating method include, but are not limited to, spin coating, dip coating, doctor blading, inkjet printing, or thermal transfer.
이하 본 발명을 실시예를 통하여 상세히 설명하면 다음과 같다. 단, 하기 실시예는 본 발명을 예시하는 것일 뿐 본 발명이 하기 실시예에 의해 한정되는 것은 아니다.Hereinafter, the present invention will be described in detail with reference to the following Examples. However, the following examples are merely to illustrate the present invention and the present invention is not limited by the following examples.
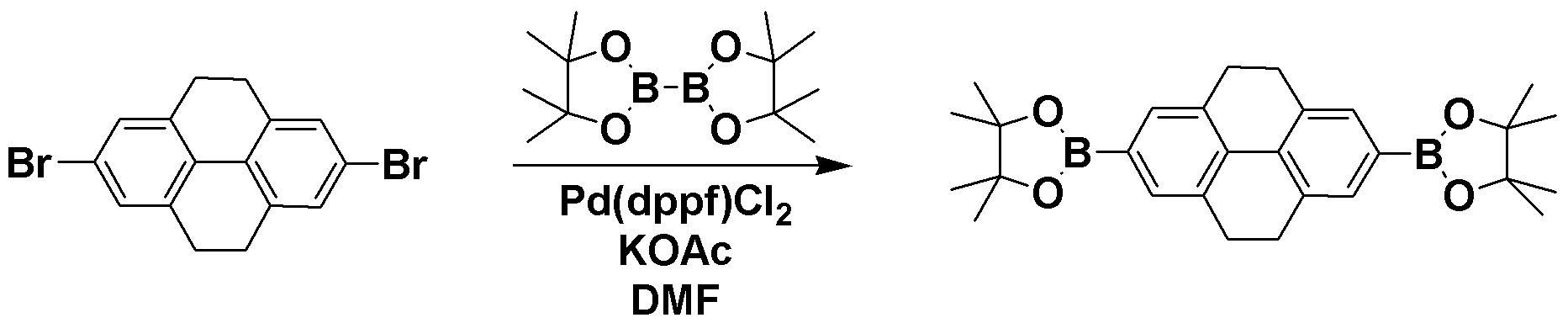
[준비예 1] TPCA-1 및 TPCB-1의 합성 Preparation Example 1 Synthesis of TPCA-1 and TPCB-1
<단계 1> 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4,5,9,10-tetrahydropyrene의 합성Step 1 Synthesis of 2,7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -4,5,9,10-tetrahydropyrene
질소 기류 하에서 2,7-dibromo-4,5,9,10-tetrahydropyrene 23.3g (0.064 mol)과 4,4,4',4',5,5,5',5'-octamethyl-2,2'-bi(1,3,2-dioxaborolane) 48.58g (0.191 mol), Pd(dppf)Cl2 5.2g (5 mol %), KOAc 37.55g (0.383 mol), DMF 900ml를 넣고 130℃에서 12h 교반한 후 반응을 종결시키고, 에틸아세테이트로 추출하여 MgSO4로 수분을 제거하였다. 용매를 제거한 반응물을 컬럼크로마토그래피를 이용하여 목적 화합물인 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4,5,9,10-tetrahydropyrene 11.73g (yield : 40 %)을 획득하였다. 23.3 g (0.064 mol) of 2,7-dibromo-4,5,9,10-tetrahydropyrene and 4,4,4 ', 4', 5,5,5 ', 5'-octamethyl-2,2 under nitrogen stream '-bi (1,3,2-dioxaborolane) 48.58g (0.191 mol), Pd (dppf) Cl 2 5.2g (5 mol%), KOAc 37.55g (0.383 mol), DMF 900ml was added and stirred for 12h at 130 ℃ After completion of the reaction, the mixture was extracted with ethyl acetate and water was removed with MgSO 4 . The reaction product from which the solvent was removed was subjected to column chromatography using 2,7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -4,5,9,10 as a target compound. 11.73 g (yield: 40%) of -tetrahydropyrene were obtained.
1H-NMR : δ 1.24 (s, 24H), 3.14 (m, 8H), 7.27 (m, 4H) 1 H-NMR: δ 1.24 (s, 24H), 3.14 (m, 8H), 7.27 (m, 4H)
<단계 2> 2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene의 합성Step 2 Synthesis of 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene
질소 기류 하에서 8g (39.6 mmol)의 1-bromo-2-nitrobenzene, 9.07g (19.8 mmol)의 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4,5,9,10-tetrahydropyrene, 4.75g (118.8 mmol)의 NaOH과 200ml/100ml의 THF/H2O를 넣고 교반하였다. 40℃에서 1.15g (5 mol%)의 Pd(PPh3)4를 넣고 80℃에서 12시간 동안 교반하였다. 반응 종결 후 메틸렌클로라이드로 추출하고 MgSO4를 넣고 필터하였다. 필터된 유기층의 용매를 제거한 후 컬럼크로마토그래피를 이용하여 목적 화합물인 2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene 6.13g (yield: 69%)을 획득하였다.8 g (39.6 mmol) of 1-bromo-2-nitrobenzene, 9.07 g (19.8 mmol) of 2,7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2- under nitrogen stream yl) -4,5,9,10-tetrahydropyrene, 4.75 g (118.8 mmol) of NaOH and 200 ml / 100 ml of THF / H 2 O were added and stirred. 1.15 g (5 mol%) of Pd (PPh 3 ) 4 was added at 40 ° C. and stirred at 80 ° C. for 12 hours. After completion of the reaction, the mixture was extracted with methylene chloride, MgSO 4 was added and filtered. After removing the solvent of the filtered organic layer, 6.13g (yield: 69%) of the target compound, 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene, was obtained by using column chromatography.
1H-NMR : δ 3.16 (m, 8H), 7.67 (m, 4H), 7.91 (m. 8H) 1 H-NMR: δ 3.16 (m, 8H), 7.67 (m, 4H), 7.91 (m. 8H)
<단계 3> TPCA-1 및 TPCB-1의 합성Step 3 Synthesis of TPCA-1 and TPCB-1
질소 기류 하에서 2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene 4.28g (9.55 mmol)과 triphenylphosphine 12.52g (47.72 mmol), 1,2-dichlorobenzene 50ml를 넣은 후 12시간 교반하였다. 반응 종료 후 1,2-dichlorobenzene를 제거하고 디클로로메탄으로 추출하였다. 추출한 유기층을 MgSO4로 물을 제거하고, 컬럼크로마토그래피를 이용하여 목적 화합물인 TPCA-1 1.58g (yield : 43 %)과 TPCB-1 1.50g (yield : 41 %)을 획득하였다.Under nitrogen stream, add 4.28 g (9.55 mmol) of 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene, 12.52 g (47.72 mmol) of triphenylphosphine, and 50 ml of 1,2-dichlorobenzene, and then stir for 12 hours. It was. After the reaction was completed, 1,2-dichlorobenzene was removed and extracted with dichloromethane. Water was removed from the extracted organic layer with MgSO 4 , and TPCA-1 1.58 g (yield: 43%) and TPCB-1 1.50 g (yield: 41%) were obtained by using column chromatography.
TPCA-1에 대한 1H-NMR: δ 3.10 (m, 8H), 7.48 (m, 2H), 7.62 (m, 2H), 7.67 (m, 2H), 7.91 (m. 4H), 10.42(s, 2H) 1 H-NMR for TPCA-1: δ 3.10 (m, 8H), 7.48 (m, 2H), 7.62 (m, 2H), 7.67 (m, 2H), 7.91 (m. 4H), 10.42 (s, 2H)
TPCB-1에 대한 1H-NMR: δ 3.11 (m, 8H), 7.45 (m, 2H), 7.58 (m, 4H), 7.98 (m. 4H), 10.40(s, 2H) 1 H-NMR for TPCB-1: δ 3.11 (m, 8H), 7.45 (m, 2H), 7.58 (m, 4H), 7.98 (m. 4H), 10.40 (s, 2H)
[준비예 2] TPCA-2 및 TPCB-2의 합성 Preparation Example 2 Synthesis of TPCA-2 and TPCB-2
<단계 1> 2,7-bis(5-bromo-2-nitrophenyl)-4,5,9,10-tetrahydropyrene의 합성Step 1 Synthesis of 2,7-bis (5-bromo-2-nitrophenyl) -4,5,9,10-tetrahydropyrene
1-bromo-2-nitrobenzene 대신 2,4-dibromo-1-nitrobenzene을 사용하는 것을 제외하고는 준비예 1의 <단계 2>와 동일한 과정을 수행하여 2,7-bis(5-bromo-2-nitrophenyl)-4,5,9,10-tetrahydropyrene을 얻었다.Except for using 2,4-dibromo-1-nitrobenzene instead of 1-bromo-2-nitrobenzene was carried out the same procedure as in <Step 2> of Preparation Example 1 to 2,7-bis (5-bromo-2- nitrophenyl) -4,5,9,10-tetrahydropyrene was obtained.
1H-NMR : δ 3.16 (m, 8H), 7.67 (m, 6H), 7.91 (m. 2H) , 8.12 (m. 2H) 1 H-NMR: δ 3.16 (m, 8H), 7.67 (m, 6H), 7.91 (m. 2H), 8.12 (m. 2H)
<단계 2> TPCA-2 및 TPCB-2의 합성Step 2 Synthesis of TPCA-2 and TPCB-2
2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene 대신 상기 <단계 1>의 2,7-bis(5-bromo-2-nitrophenyl)-4,5,9,10-tetrahydropyrene을 사용하는 것을 제외하고는 준비예 1의 <단계 3>와 동일한 과정을 수행하여 TPCA-2과 TPCB-2를 얻었다.2,7-bis (5-bromo-2-nitrophenyl) -4,5,9,10- of <Step 1> instead of 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene TPCA-2 and TPCB-2 were obtained by the same procedure as <Step 3> of Preparation Example 1, except that tetrahydropyrene was used.
TPCA-2에 대한 1H-NMR: δ 3.05 (m, 8H), 7.51 (m, 4H), 7.90 (m. 4H) , 10.22 (s, 2H) 1 H-NMR for TPCA-2: δ 3.05 (m, 8H), 7.51 (m, 4H), 7.90 (m. 4H), 10.22 (s, 2H)
TPCB-2에 대한 1H-NMR: δ 3.02 (m, 8H), 7.50 (m, 4H), 7.95 (m. 4H) , 10.20 (s, 2H) 1 H-NMR for TPCB-2: δ 3.02 (m, 8H), 7.50 (m, 4H), 7.95 (m. 4H), 10.20 (s, 2H)
[준비예 3] DPCA-1 및 DPCB-1의 합성 Preparation Example 3 Synthesis of DPCA-1 and DPCB-1
<단계 1> 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9,10-dihydrophenanthrene의 합성Step 1 Synthesis of 2,7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -9,10-dihydrophenanthrene
2,7-dibromo-4,5,9,10-tetrahydropyrene 대신 2,7-dibromo-9,10-dihydrophenanthrene를 사용하는 것을 제외하고는 준비예 1의 <단계 1>과 동일한 과정을 수행하여 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9,10-dihydrophenanthrene 을 얻었다.Except for using 2,7-dibromo-9,10-dihydrophenanthrene instead of 2,7-dibromo-4,5,9,10-tetrahydropyrene, the same procedure as in <Step 1> of Preparation Example 1 was performed. 7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -9,10-dihydrophenanthrene was obtained.
1H-NMR : δ 1.23 (s, 24H), 3.13 (m, 4H), 7.37 (m, 2H), 7.73 (m, 4H) 1 H-NMR: δ 1.23 (s, 24H), 3.13 (m, 4H), 7.37 (m, 2H), 7.73 (m, 4H)
<단계 2> 2,7-bis(2-nitrophenyl)-9,10-dihydrophenanthrene의 합성Step 2 Synthesis of 2,7-bis (2-nitrophenyl) -9,10-dihydrophenanthrene
2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4,5,9,10-tetrahydropyrene 대신 상기 <단계 1>에서 얻은 2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-9,10-dihydrophenanthrene 을 사용하는 것을 제외하고는 준비예 1의 <단계 2>와 동일한 과정을 수행하여 2,7-bis(2-nitrophenyl)-9,10-dihydrophenanthrene 을 얻었다.2,7-bis obtained in <Step 1> instead of 2,7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -4,5,9,10-tetrahydropyrene Perform the same procedure as in <Step 2> of Preparation Example 1, except using bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -9,10-dihydrophenanthrene To 2,7-bis (2-nitrophenyl) -9,10-dihydrophenanthrene.
1H-NMR : δ 3.16 (m, 4H), 7.05 (d, 2H), 7.67 (t, 2H), 7.91 (m, 10H) 1 H-NMR: δ 3.16 (m, 4H), 7.05 (d, 2H), 7.67 (t, 2H), 7.91 (m, 10H)
<단계 3> DPCA-1 및 DPCB-1의 합성Step 3 Synthesis of DPCA-1 and DPCB-1
2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene 대신 상기 <단계 2>에서 2,7-2,7-bis(2-nitrophenyl)-9,10-dihydrophenanthrene 을 사용하는 것을 제외하고는 준비예 1의 <단계 3>와 동일한 과정을 수행하여 DPCA-1과 DPCB-1를 얻었다.2,7-2,7-bis (2-nitrophenyl) -9,10-dihydrophenanthrene in <Step 2> instead of 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene Except for this, the same procedure as in <Step 3> of Preparation Example 1 was performed to obtain DPCA-1 and DPCB-1.
DPCA-1에 대한 1H-NMR: δ 3.00 (m, 4H), 7.30 (t, 2H), 7.51 (m, 6H), 8.09 (m, 4H) , 10.20 (s, 2H) 1 H-NMR for DPCA-1: δ 3.00 (m, 4H), 7.30 (t, 2H), 7.51 (m, 6H), 8.09 (m, 4H), 10.20 (s, 2H)
DPCB-1에 대한 1H-NMR: δ 3.04 (m, 4H), 7.34 (t, 2H), 7.57 (m, 6H), 8.06 (m, 4H) , 10.23 (s, 2H) 1 H-NMR for DPCB-1: δ 3.04 (m, 4H), 7.34 (t, 2H), 7.57 (m, 6H), 8.06 (m, 4H), 10.23 (s, 2H)
[준비예 4] DPCA-2 및 DPCB-2의 합성 Preparation Example 4 Synthesis of DPCA-2 and DPCB-2
<단계 1> 2,7-bis(5-bromo-2-nitrophenyl)-9,10-dihydrophenanthrene의 합성Step 1 Synthesis of 2,7-bis (5-bromo-2-nitrophenyl) -9,10-dihydrophenanthrene
1-bromo-2-nitrobenzene 대신 2,4-dibromo-1-nitrobenzene을 사용하는 것을 제외하고는 준비예 3의 <단계 2>와 동일한 과정을 수행하여 2,7-bis(5-bromo-2-nitrophenyl)-9,10-dihydrophenanthrene 을 얻었다.Except for using 2,4-dibromo-1-nitrobenzene instead of 1-bromo-2-nitrobenzene was carried out the same procedure as in <Step 2> of Preparation Example 3 to 2,7-bis (5-bromo-2- nitrophenyl) -9,10-dihydrophenanthrene was obtained.
1H-NMR : δ 3.06 (m, 4H), 7.07 (d, 2H), 7.81 (m, 8H) , 8.10 (d, 2H) 1 H-NMR: δ 3.06 (m, 4H), 7.07 (d, 2H), 7.81 (m, 8H), 8.10 (d, 2H)
<단계 2> DPCA-2 및 DPCB-2의 합성Step 2 Synthesis of DPCA-2 and DPCB-2
2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene 대신 상기 <단계 1>에서 2,7-bis(5-bromo-2-nitrophenyl)-9,10-dihydrophenanthrene을 사용하는 것을 제외하고는 준비예 1의 <단계 3>와 동일한 과정을 수행하여 DPCA-2과 DPCB-2를 얻었다.Using 2,7-bis (5-bromo-2-nitrophenyl) -9,10-dihydrophenanthrene in <Step 1> instead of 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene Except for the same procedure as in <Step 3> of Preparation Example 1 to obtain DPCA-2 and DPCB-2.
DPCA-2에 대한 1H-NMR: δ 3.05 (m, 4H), 7.51 (m, 6H), 8.09 (m, 4H) , 10.32 (s, 2H) 1 H-NMR for DPCA-2: δ 3.05 (m, 4H), 7.51 (m, 6H), 8.09 (m, 4H), 10.32 (s, 2H)
DPCB-2에 대한 1H-NMR: δ 3.02 (m, 4H), 7.50 (m, 6H), 8.05 (m, 4H) , 10.30 (s, 2H) 1 H-NMR for DPCB-2: δ 3.02 (m, 4H), 7.50 (m, 6H), 8.05 (m, 4H), 10.30 (s, 2H)
[준비예 5] mDPCB-1의 합성 Preparation Example 5 Synthesis of mDPCB-1
<단계 1> 2,2'-(9,10-dimethyl-9,10-dihydrophenanthrene-2,7-diyl)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)의 합성<Step 1> Synthesis of 2,2 '-(9,10-dimethyl-9,10-dihydrophenanthrene-2,7-diyl) bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolane)
2,7-dibromo-4,5,9,10-tetrahydropyrene 대신 2,7-dibromo-9,10-dimethyl-9,10-dihydrophenanthrene를 사용하는 것을 제외하고는 준비예 1의 <단계 1>과 동일한 과정을 수행하여 2,2'-(9,10-dimethyl-9,10-dihydrophenanthrene-2,7-diyl)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane) 을 얻었다.Same as <Step 1> of Preparation Example 1, except that 2,7-dibromo-9,10-dimethyl-9,10-dihydrophenanthrene was used instead of 2,7-dibromo-4,5,9,10-tetrahydropyrene The procedure was followed to obtain 2,2 '-(9,10-dimethyl-9,10-dihydrophenanthrene-2,7-diyl) bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolane) .
1H-NMR : δ 1.25 (s, 24H), 1.28 (s, 6H), 3.13 (m, 2H), 7.35 (m, 2H), 7.72 (m, 4H) 1 H-NMR: δ 1.25 (s, 24H), 1.28 (s, 6H), 3.13 (m, 2H), 7.35 (m, 2H), 7.72 (m, 4H)
<단계 2> 9,10-dimethyl-2,7-bis(2-nitrophenyl)-9,10-dihydrophenanthrene의 합성Step 2 Synthesis of 9,10-dimethyl-2,7-bis (2-nitrophenyl) -9,10-dihydrophenanthrene
2,7-bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-4,5,9,10-tetrahydropyrene 대신 상기 <단계 1>에서 얻은 2,2'-(9,10-dimethyl-9,10-dihydrophenanthrene-2,7-diyl)bis(4,4,5,5-tetramethyl-1,3,2-dioxaborolane)을 사용하는 것을 제외하고는 준비예 1의 <단계 2>와 동일한 과정을 수행하여 9,10-dimethyl-2,7-bis(2-nitrophenyl)-9,10-dihydrophenanthrene 을 얻었다.2,2 'obtained in <Step 1> instead of 2,7-bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl) -4,5,9,10-tetrahydropyrene Preparation Example 1 except that-(9,10-dimethyl-9,10-dihydrophenanthrene-2,7-diyl) bis (4,4,5,5-tetramethyl-1,3,2-dioxaborolane) was used 9,10-dimethyl-2,7-bis (2-nitrophenyl) -9,10-dihydrophenanthrene was obtained by the same process as in <Step 2>.
1H-NMR : δ 1.25 (s, 6H), 3.32 (m, 2H), 7.01 (m, 2H), 7.72 (m, 4H), 8.02 (m, 8H) 1 H-NMR: δ 1.25 (s, 6H), 3.32 (m, 2H), 7.01 (m, 2H), 7.72 (m, 4H), 8.02 (m, 8H)
<단계 3> mDPCB-1의 합성Step 3 Synthesis of mDPCB-1
2,7-bis(2-nitrophenyl)-4,5,9,10-tetrahydropyrene 대신 상기 <단계 2>에서 9,10-dimethyl-2,7-bis(2-nitrophenyl)-9,10-dihydrophenanthrene을 사용하는 것을 제외하고는 준비예 1의 <단계 3>와 동일한 과정을 수행하여 mDPCB-1를 얻었다.9,10-dimethyl-2,7-bis (2-nitrophenyl) -9,10-dihydrophenanthrene in <Step 2> instead of 2,7-bis (2-nitrophenyl) -4,5,9,10-tetrahydropyrene Except for using the same procedure as in <Step 3> of Preparation Example 1 to obtain mDPCB-1.
1H-NMR: δ 1.24 (s, 6H), 3.36 (m, 2H), 7.31 (m, 2H), 7.72 (m, 6H), 8.12 (m, 4H), 10.33 (s, 2H) 1 H-NMR: δ 1.24 (s, 6H), 3.36 (m, 2H), 7.31 (m, 2H), 7.72 (m, 6H), 8.12 (m, 4H), 10.33 (s, 2H)
[합성예 1] SPC-1의 합성Synthesis Example 1 Synthesis of SPC-1
질소 기류 하에서 준비예 1에서 제조한 TPCA-1 (3.4g, 8.86mmol), 1-bromobenzene (4.17g, 26.56mmol), Cu powder(0.11g, 1.77mmol), K2CO3(2.44g, 17.71mmol), Na2SO4(2.52g, 17.71mmol) 및 nitrobenzene(100 ml)를 혼합하고 190℃에서 12시간 동안 교반하였다. TPCA-1 (3.4 g, 8.86 mmol), 1-bromobenzene (4.17 g, 26.56 mmol), Cu powder (0.11 g, 1.77 mmol), K 2 CO 3 (2.44 g, 17.71) prepared in Preparation Example 1 under nitrogen stream. mmol), Na 2 SO 4 (2.52 g, 17.71 mmol) and nitrobenzene (100 ml) were mixed and stirred at 190 ° C. for 12 hours.
반응이 종결된 후 nitrobenzene을 제거하고 메틸렌클로라이드로 유기층을 분리한 다음 MgSO4를 사용하여 물을 제거하였다. 유기층의 용매를 제거한 후 컬럼크로마토그래피로 정제하여 목적 화합물인 SPC-1 (3.28g, 수율 69%)을 얻었다. After completion of the reaction, nitrobenzene was removed, the organic layer was separated with methylene chloride, and water was removed using MgSO 4 . After removing the solvent of the organic layer was purified by column chromatography to give the title compound SPC-1 (3.28g, 69% yield).
GC-Mass (이론치: 536.66 g/mol, 측정치: 536 g/mol)GC-Mass (Theoretical value: 536.66 g / mol, Measured value: 536 g / mol)
[합성예 2] SPC-2의 합성Synthesis Example 2 Synthesis of SPC-2
1-bromobenzene 대신 2-bromopyridine을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-2 (3.09 g, 수율 65%)을 얻었다.Except for using 2-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 1 to obtain the target compound SPC-2 (3.09 g, yield 65%).
GC-Mass (이론치: 538.64 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.64 g / mol, Measured value: 538 g / mol)
[합성예 3] SPC-3의 합성Synthesis Example 3 Synthesis of SPC-3
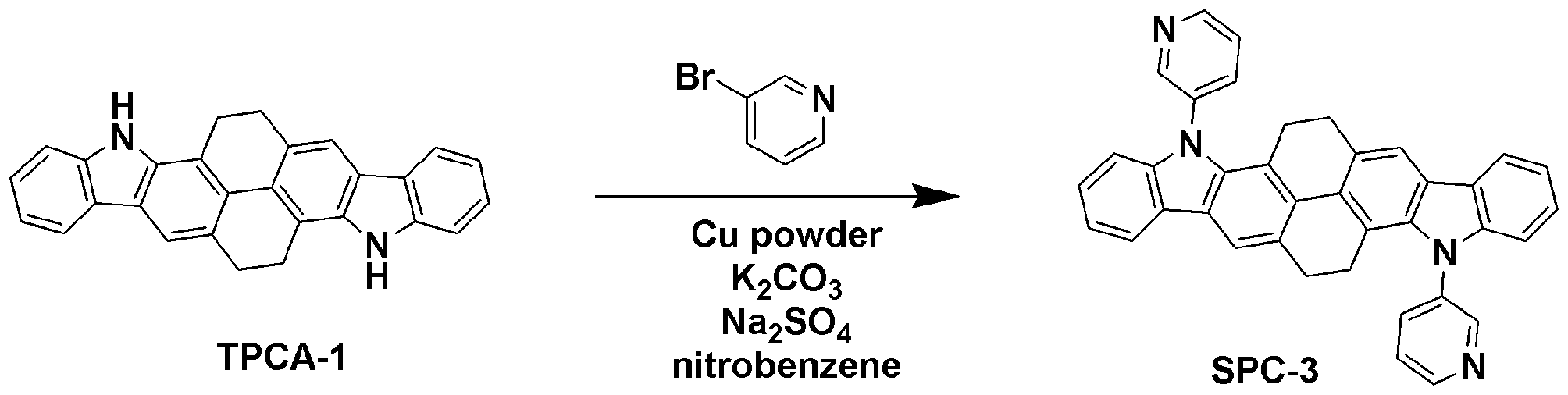
1-bromobenzene 대신 3-bromopyridine을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-3 (2.85 g, 수율 60%)을 얻었다.Except for using 3-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 1 to obtain the target compound SPC-3 (2.85 g, yield 60%).
GC-Mass (이론치: 538.64 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.64 g / mol, Measured value: 538 g / mol)
[합성예 4] SPC-4의 합성Synthesis Example 4 Synthesis of SPC-4
1-bromobenzene 대신 4-bromopyridine을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-4 (3.10 g, 수율 65%)을 얻었다.Except for using 4-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 1 to obtain the target compound SPC-4 (3.10 g, yield 65%).
GC-Mass (이론치: 538.64 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.64 g / mol, Measured value: 538 g / mol)
[합성예 5] SPC-5의 합성Synthesis Example 5 Synthesis of SPC-5
1-bromobenzene 대신 2-bromo-4,6-diphenylpyridine을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-5 (4.67 g, 수율 55%)을 얻었다.Except for using 2-bromo-4,6-diphenylpyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 1 to obtain the target compound SPC-5 (4.67 g, 55% yield).
GC-Mass (이론치: 843.02 g/mol, 측정치: 842 g/mol)GC-Mass (Theoretical value: 843.02 g / mol, Measured value: 842 g / mol)
[합성예 6] SPC-6의 합성Synthesis Example 6 Synthesis of SPC-6
1-bromobenzene 대신 2-bromo-4,6-diphenylpyrimidine을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-6 (4.60 g, 수율 54%)을 얻었다.Except for using 2-bromo-4,6-diphenylpyrimidine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 1 to obtain the target compound SPC-6 (4.60 g, yield 54%).
GC-Mass (이론치: 845.00 g/mol, 측정치: 844 g/mol)GC-Mass (Theoretical value: 845.00 g / mol, Measured value: 844 g / mol)
[합성예 7] SPC-7의 합성Synthesis Example 7 Synthesis of SPC-7
1-bromobenzene 대신 2-bromo-4,6-diphenyl-1,3,5-triazine을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-7 (4.80 g, 수율 57%)을 얻었다.Except for using 2-bromo-4,6-diphenyl-1,3,5-triazine instead of 1-bromobenzene, the same procedure as in Synthesis Example 1 was carried out to obtain the target compound SPC-7 (4.80 g, yield 57% )
GC-Mass (이론치: 846.98 g/mol, 측정치: 846 g/mol)GC-Mass (Theoretical value: 846.98 g / mol, Measured value: 846 g / mol)
[합성예 8] SPC-8의 합성Synthesis Example 8 Synthesis of SPC-8
TPCA-1 대신 준비예 1 에서 제조한 TPCB-1을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-8 (3.27 g, 수율 69%)을 얻었다.Except for using the TPCB-1 prepared in Preparation Example 1 instead of TPCA-1 was carried out in the same manner as in Synthesis Example 1 to obtain the target compound SPC-8 (3.27 g, 69% yield).
GC-Mass (이론치: 536.66 g/mol, 측정치: 536 g/mol)GC-Mass (Theoretical value: 536.66 g / mol, Measured value: 536 g / mol)
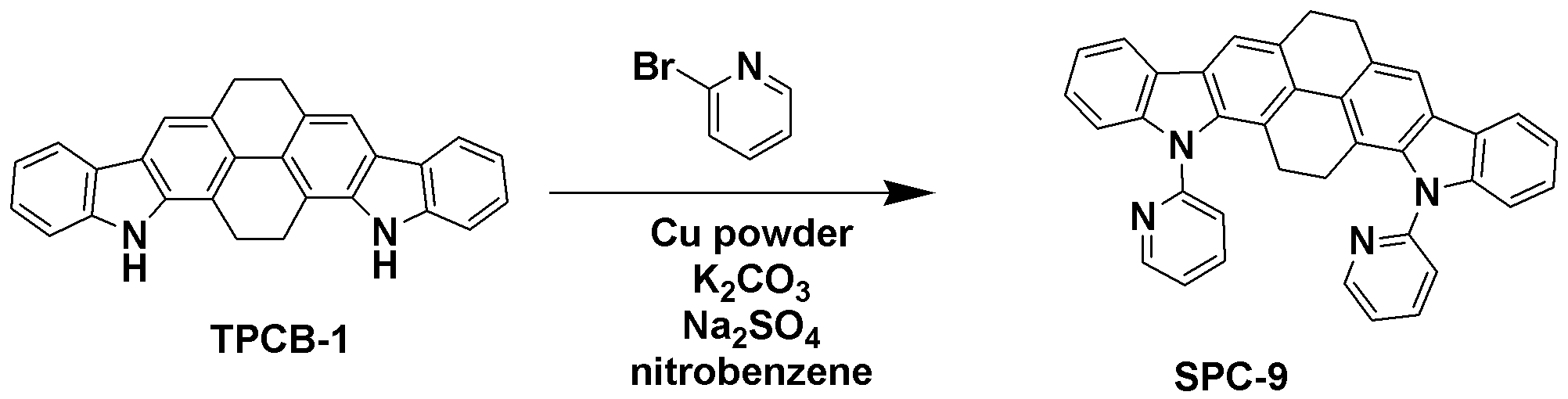
[합성예 9] SPC-9의 합성Synthesis Example 9 Synthesis of SPC-9
1-bromobenzene 대신 2-bromopyridine을 사용하는 것을 제외하고는 합성예 8와 동일한 과정을 수행하여 목적 화합물인 SPC-9 (2.86 g, 수율 60%)을 얻었다.Except for using 2-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 8 to obtain the target compound SPC-9 (2.86 g, yield 60%).
GC-Mass (이론치: 538.64 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.64 g / mol, Measured value: 538 g / mol)
[합성예 10] SPC-10의 합성Synthesis Example 10 Synthesis of SPC-10
1-bromobenzene 대신 3-bromopyridine을 사용하는 것을 제외하고는 합성예 8와 동일한 과정을 수행하여 목적 화합물인 SPC-10 (2.83 g, 수율 59%)을 얻었다.Except for using 3-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 8 to obtain the target compound SPC-10 (2.83 g, 59% yield).
GC-Mass (이론치: 538.64 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.64 g / mol, Measured value: 538 g / mol)
[합성예 11] SPC-11의 합성Synthesis Example 11 Synthesis of SPC-11
1-bromobenzene 대신 4-bromopyridine을 사용하는 것을 제외하고는 합성예 8와 동일한 과정을 수행하여 목적 화합물인 SPC-11 (2.80 g, 수율 58%)을 얻었다.Except for using 4-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 8 to obtain the target compound SPC-11 (2.80 g, yield 58%).
GC-Mass (이론치: 538.64 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.64 g / mol, Measured value: 538 g / mol)
[합성예 12] SPC-12의 합성Synthesis Example 12 Synthesis of SPC-12
TPCA-1 대신 준비예 3에서 제조한 DPCA-1을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-12 (3.77 g, 수율 71%)을 얻었다.Except for using DPCA-1 prepared in Preparation Example 3 instead of TPCA-1 to obtain the target compound SPC-12 (3.77 g, yield 71%) was carried out in the same manner as in Synthesis Example 1.
GC-Mass (이론치: 510.63 g/mol, 측정치: 510 g/mol)GC-Mass (Theoretical value: 510.63 g / mol, Measured value: 510 g / mol)
[합성예 13] SPC-13의 합성Synthesis Example 13 Synthesis of SPC-13
1-bromobenzene 대신 2-bromopyridine을 사용하는 것을 제외하고는 합성예 12과 동일한 과정을 수행하여 목적 화합물인 SPC-13 (3.58 g, 수율 68%)을 얻었다.Except for using 2-bromopyridine instead of 1-bromobenzene was carried out in the same manner as in Synthesis Example 12 to obtain the target compound SPC-13 (3.58 g, yield 68%).
GC-Mass (이론치: 512.60 g/mol, 측정치: 512 g/mol)GC-Mass (Theoretical value: 512.60 g / mol, Measured value: 512 g / mol)
[합성예 14] SPC-14의 합성Synthesis Example 14 Synthesis of SPC-14
1-bromobenzene 대신 2-bromo-4,6-diphenylpyridine을 사용하는 것을 제외하고는 합성예 12과 동일한 과정을 수행하여 목적 화합물인 SPC-14 (4.96 g, 수율 70%)을 얻었다.Except for using 2-bromo-4,6-diphenylpyridine instead of 1-bromobenzene was carried out in the same manner as in Synthesis Example 12 to obtain the target compound SPC-14 (4.96 g, yield 70%).
GC-Mass (이론치: 816.99 g/mol, 측정치: 816 g/mol)GC-Mass (Theoretical value: 816.99 g / mol, Measured value: 816 g / mol)
[합성예 15] SPC-15의 합성Synthesis Example 15 Synthesis of SPC-15
1-bromobenzene 대신 2-bromo-4,6-diphenylpyrimidine을 사용하는 것을 제외하고는 합성예 12과 동일한 과정을 수행하여 목적 화합물인 SPC-15 (4.90 g, 수율 69%)을 얻었다.Except for using 2-bromo-4,6-diphenylpyrimidine instead of 1-bromobenzene was carried out in the same manner as in Synthesis Example 12 to obtain the target compound SPC-15 (4.90 g, 69% yield).
GC-Mass (이론치: 818.96 g/mol, 측정치: 818 g/mol)GC-Mass (Theoretical value: 818.96 g / mol, Measured value: 818 g / mol)
[합성예 16] SPC-16의 합성Synthesis Example 16 Synthesis of SPC-16
1-bromobenzene 대신 2-bromo-4,6-diphenyl-1,3,5-triazine을 사용하는 것을 제외하고는 합성예 12와 동일한 과정을 수행하여 목적 화합물인 SPC-16 (4.05 g, 수율 57%)을 얻었다.Except for using 2-bromo-4,6-diphenyl-1,3,5-triazine instead of 1-bromobenzene, the same procedure as in Synthesis Example 12 was carried out to obtain the target compound SPC-16 (4.05 g, yield 57% )
GC-Mass (이론치: 820.94 g/mol, 측정치: 820 g/mol)GC-Mass (Theoretical value: 820.94 g / mol, Measured value: 820 g / mol)
[합성예 17] SPC-17의 합성Synthesis Example 17 Synthesis of SPC-17
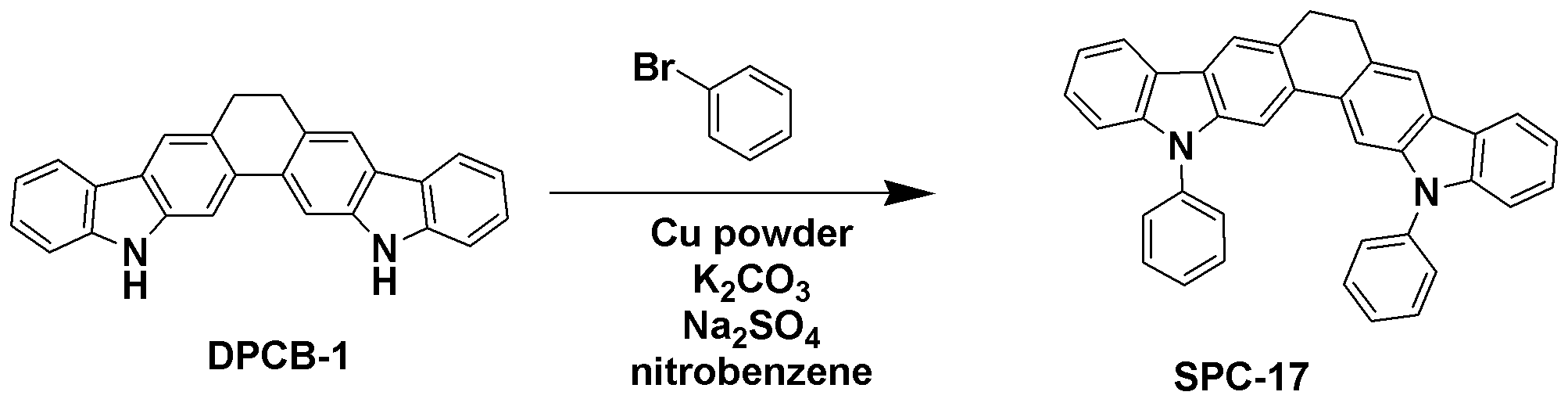
TPCA-1 대신 준비예 3에서 제조한 또 다른 화합물 DPCB-1을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-17 (3.00 g, 수율 68%)을 얻었다.Except for using another compound DPCB-1 prepared in Preparation Example 3 instead of TPCA-1 to the same procedure as in Synthesis Example 1 to obtain the target compound SPC-17 (3.00 g, yield 68%).
GC-Mass (이론치: 510.63 g/mol, 측정치: 510 g/mol)GC-Mass (Theoretical value: 510.63 g / mol, Measured value: 510 g / mol)
[합성예 18] SPC-18의 합성Synthesis Example 18 Synthesis of SPC-18
1-bromobenzene 대신 2-bromopyridine을 사용하는 것을 제외하고는 합성예 17와 동일한 과정을 수행하여 목적 화합물인 SPC-18 (2.66 g, 수율 61%)을 얻었다.Except for using 2-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 17 to obtain the target compound SPC-18 (2.66 g, 61% yield).
GC-Mass (이론치: 512.60 g/mol, 측정치: 512 g/mol)GC-Mass (Theoretical value: 512.60 g / mol, Measured value: 512 g / mol)
[합성예 19] SPC-19의 합성Synthesis Example 19 Synthesis of SPC-19
1-bromobenzene 대신 3-bromopyridine을 사용하는 것을 제외하고는 합성예 17와 동일한 과정을 수행하여 목적 화합물인 SPC-19 (2.64 g, 수율 60%)을 얻었다.Except for using 3-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 17 to obtain the target compound SPC-19 (2.64 g, yield 60%).
GC-Mass (이론치: 512.60 g/mol, 측정치: 512 g/mol)GC-Mass (Theoretical value: 512.60 g / mol, Measured value: 512 g / mol)
[합성예 20] SPC-20의 합성Synthesis Example 20 Synthesis of SPC-20
1-bromobenzene 대신 4-bromopyridine을 사용하는 것을 제외하고는 합성예 17와 동일한 과정을 수행하여 목적 화합물인 SPC-20 (2.65 g, 수율 60%)을 얻었다.Except for using 4-bromopyridine instead of 1-bromobenzene was carried out the same procedure as in Synthesis Example 17 to obtain the target compound SPC-20 (2.65 g, yield 60%).
GC-Mass (이론치: 512.60 g/mol, 측정치: 512 g/mol)GC-Mass (Theoretical value: 512.60 g / mol, Measured value: 512 g / mol)
[합성예 21] SPC-21의 합성Synthesis Example 21 Synthesis of SPC-21
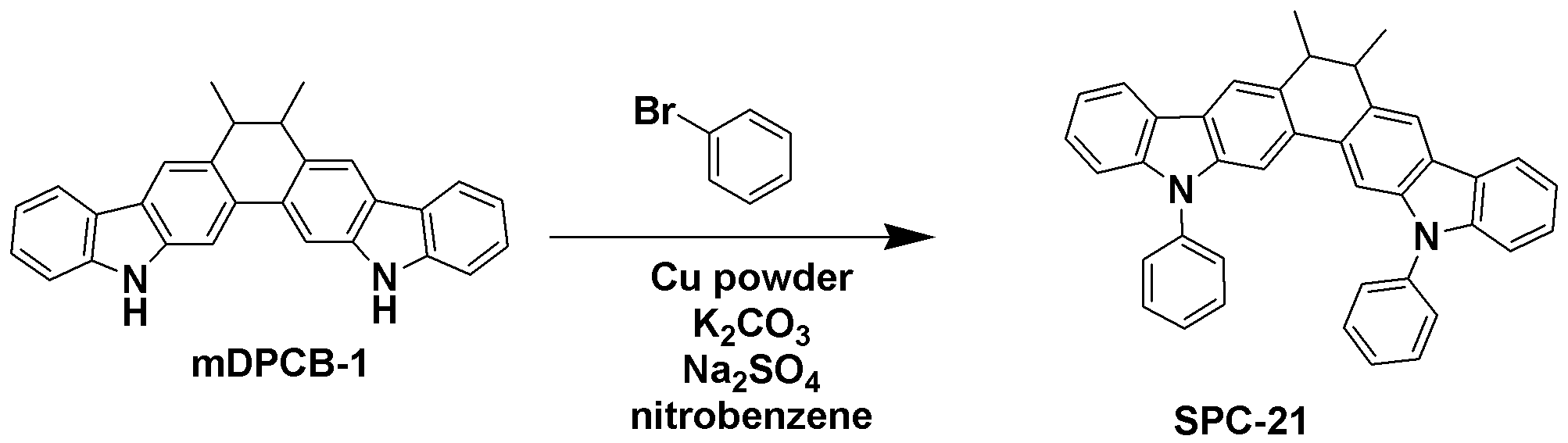
TPCA-1 대신 준비예 5에서 제조한 mDPCB-1을 사용하는 것을 제외하고는 합성예 1과 동일한 과정을 수행하여 목적 화합물인 SPC-21 (3.26 g, 수율 70%)을 얻었다.Except for using mDPCB-1 prepared in Preparation Example 5 instead of TPCA-1 was carried out in the same manner as in Synthesis Example 1 to obtain the target compound SPC-21 (3.26 g, yield 70%).
GC-Mass (이론치: 538.68 g/mol, 측정치: 538 g/mol)GC-Mass (Theoretical value: 538.68 g / mol, Measured value: 538 g / mol)
[합성예 22] SPC-22의 합성Synthesis Example 22 Synthesis of SPC-22
1-bromobenzene 대신 2-bromopyridine을 사용하는 것을 제외하고는 합성예 21과 동일한 과정을 수행하여 목적 화합물인 SPC-22 (2.85 g, 수율 61%)을 얻었다.Except for using 2-bromopyridine instead of 1-bromobenzene was carried out in the same manner as in Synthesis Example 21 to obtain the target compound SPC-22 (2.85 g, 61% yield).
GC-Mass (이론치: 540.66 g/mol, 측정치: 540 g/mol)GC-Mass (Theoretical value: 540.66 g / mol, Measured value: 540 g / mol)
[합성예 23] SPC-23의 합성Synthesis Example 23 Synthesis of SPC-23
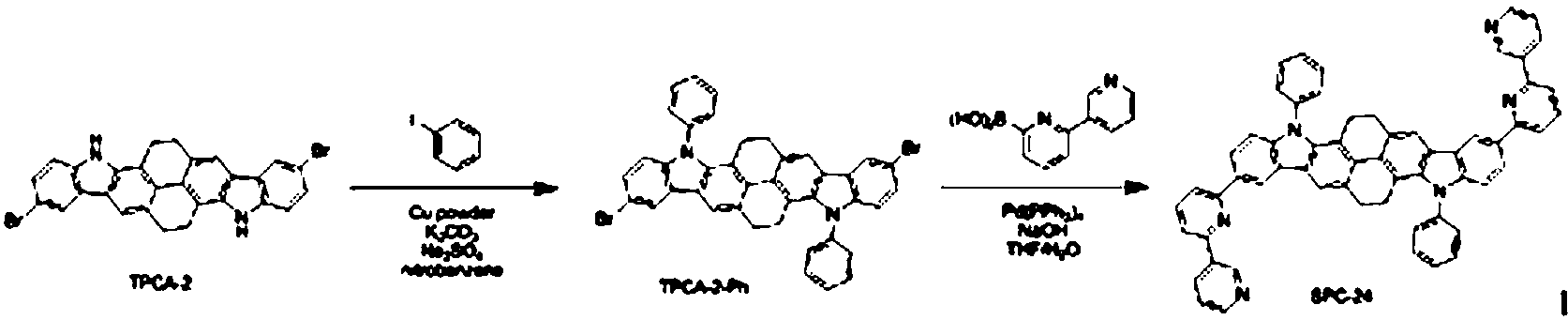
질소 기류 하에서 준비예 2에서 제조한 TPCA-2(3.75 g, 6.92 mmol), iodobenzene(4.24 g, 20.76 mmol), Cu powder(0.09 g, 1.38 mmol), K2CO3(1.91 g, 13.84 mmol), Na2SO4(1.97 g, 13.84 mmol) 및 nitrobenzene(80 ml)을 혼합하고 190℃에서 12시간 동안 교반하였다. 반응이 종결된 후 nitrobenzene을 제거하고 메틸렌클로라이드로 유기층을 분리한 다음 MgSO4를 사용하여 물을 제거하였다. 유기층의 용매를 제거한 후 컬럼 크로마토그래피로 정제하여 중간 화합물인 TPCA-2-Ph (2.74 g, 수율 57%)을 얻었다. TPCA-2 (3.75 g, 6.92 mmol), iodobenzene (4.24 g, 20.76 mmol), Cu powder (0.09 g, 1.38 mmol), K 2 CO 3 (1.91 g, 13.84 mmol) prepared in Preparation Example 2 under a nitrogen stream. , Na 2 SO 4 (1.97 g, 13.84 mmol) and nitrobenzene (80 ml) were mixed and stirred at 190 ° C. for 12 hours. After completion of the reaction, nitrobenzene was removed, the organic layer was separated with methylene chloride, and water was removed using MgSO 4 . After removing the solvent of the organic layer was purified by column chromatography to give the intermediate compound TPCA-2-Ph (2.74 g, yield 57%).
질소 기류 하에서, 얻어진 상기 중간 화합물인 TPCA-2-Ph (2.74 g, 3.94 mmol), pyridin-2-ylboronic acid (1.16 g, 9.47 mmol), NaOH(0.95 g, 23.67 mmol) 및 THF/H2O(100 ml/50 ml)를 혼합하여 교반한 다음, 40℃에서 0.46g (5 mol%)의 Pd(PPh3)4를 넣고 80℃에서 12시간 동안 교반하였다. 반응이 종결된 후 메틸렌클로라이드로 추출하고 MgSO4를 넣고 여과하였다. 얻어진 유기층의 용매를 제거한 후 컬럼 크로마토그래피로 정제하여 목적 화합물인 SPC-23 (2.27 g, 수율 83%)을 얻었다. Under nitrogen stream, the obtained intermediate compound TPCA-2-Ph (2.74 g, 3.94 mmol), pyridin-2-ylboronic acid (1.16 g, 9.47 mmol), NaOH (0.95 g, 23.67 mmol) and THF / H 2 O (100 ml / 50 ml) was mixed and stirred, and 0.46 g (5 mol%) of Pd (PPh 3 ) 4 was added at 40 ° C. and stirred at 80 ° C. for 12 hours. After the reaction was terminated and extracted with methylene chloride, MgSO 4 was added and filtered. The solvent of the obtained organic layer was removed, and then purified by column chromatography to obtain SPC-23 (2.27 g, yield 83%) as a target compound.
GC-Mass (이론치: 690.83 g/mol, 측정치: 690 g/mol)GC-Mass (Theoretical value: 690.83 g / mol, Measured value: 690 g / mol)
[합성예 24] SPC-24의 합성Synthesis Example 24 Synthesis of SPC-24
합성예 23과 동일한 과정을 수행하되, pyridin-2-ylboronic acid 대신 2,3'-bipyridin-6-ylboronic acid를 사용하여 목적 화합물인 SPC-24 (2.53 g, 수율 76%)을 얻었다.The same procedure as in Synthesis Example 23 was performed, but SPC-24 (2.53 g, yield 76%) was obtained using 2,3'-bipyridin-6-ylboronic acid instead of pyridin-2-ylboronic acid.
GC-Mass (이론치: 845.00 g/mol, 측정치: 844 g/mol)GC-Mass (Theoretical value: 845.00 g / mol, Measured value: 844 g / mol)
[합성예 25] SPC-25의 합성Synthesis Example 25 Synthesis of SPC-25
합성예 23과 동일한 과정을 수행하되, pyridin-2-ylboronic acid 대신 phenylboronic acid를 사용하여 목적 화합물인 SPC-25 (2.37 g, 수율 71%)을 얻었다.The same procedure as in Synthesis Example 23 was performed, but phenylboronic acid was used instead of pyridin-2-ylboronic acid to obtain the target compound SPC-25 (2.37 g, 71% yield).
GC-Mass (이론치: 688.86 g/mol, 측정치: 688 g/mol)GC-Mass (Theoretical value: 688.86 g / mol, Measured value: 688 g / mol)
[합성예 26] SPC-26의 합성Synthesis Example 26 Synthesis of SPC-26
합성예 23과 동일한 과정을 수행하되, TPCA-2 대신 준비예 2에서 제조한 TPCB-2를 사용하여 목적 화합물인 SPC-26 (2.56 g, 수율 77%)을 얻었다.The same procedure as in Synthesis Example 23 was performed, but SPC-26 (2.56 g, yield 77%) was obtained using TPCB-2 prepared in Preparation Example 2 instead of TPCA-2.
GC-Mass (이론치: 690.83 g/mol, 측정치: 690 g/mol)GC-Mass (Theoretical value: 690.83 g / mol, Measured value: 690 g / mol)
[합성예 27] SPC-27의 합성Synthesis Example 27 Synthesis of SPC-27
합성예 25와 동일한 과정을 수행하되, TPCA-2 대신 준비예 2에서 제조한 TPCB-2를 사용하여 목적 화합물인 SPC-27 (2.33 g, 수율 70%)을 얻었다.The same procedure as in Synthesis Example 25 was carried out, but SPC-27 (2.33 g, yield 70%) was obtained using TPCB-2 prepared in Preparation Example 2 instead of TPCA-2.
GC-Mass (이론치: 688.86 g/mol, 측정치: 688 g/mol)GC-Mass (Theoretical value: 688.86 g / mol, Measured value: 688 g / mol)
[합성예 28] SPC-28의 합성Synthesis Example 28 Synthesis of SPC-28
합성예 24와 동일한 과정을 수행하되, TPCA-2 대신 준비예 2에서 제조한 TPCB-2를 사용하여 목적 화합물인 SPC-28 (2.50 g, 수율 75%)을 얻었다.The same procedure as in Synthesis Example 24 was performed, but SPC-28 (2.50 g, yield 75%) was obtained using TPCB-2 prepared in Preparation Example 2 instead of TPCA-2.
GC-Mass (이론치: 845.00 g/mol, 측정치: 844 g/mol)GC-Mass (Theoretical value: 845.00 g / mol, Measured value: 844 g / mol)
[합성예 29] SPC-29의 합성Synthesis Example 29 Synthesis of SPC-29
합성예 23과 동일한 과정을 수행하되, TPCA-2 대신 준비예 4에서 제조한 DPCB-2를 사용하고, iodobenzene 대신 2-bromo-4,6-diphenyl-1,3,5-triazine를 사용하여 목적 화합물인 SPC-29 (2.25 g, 수율 66%)을 얻었다.Perform the same process as in Synthesis Example 23, using DPCB-2 prepared in Preparation Example 4 instead of TPCA-2, and using 2-bromo-4,6-diphenyl-1,3,5-triazine instead of iodobenzene SPC-29 (2.25 g, yield 66%) was obtained.
GC-Mass (이론치: 975.11 g/mol, 측정치: 974 g/mol)GC-Mass (Theoretical value: 975.11 g / mol, Measured value: 974 g / mol)
[합성예 30] SPC-30의 합성Synthesis Example 30 Synthesis of SPC-30
합성예 29과 동일한 과정을 수행하되, pyridin-2-ylboronic acid 대신 phenylboronic acid를 사용하여 목적 화합물인 SPC-30 (2.20 g, 수율 64%)을 얻었다.The same procedure as in Synthesis Example 29 was performed, but phenylboronic acid was used instead of pyridin-2-ylboronic acid to obtain SPC-30 (2.20 g, 64% yield) as a target compound.
GC-Mass (이론치: 973.13 g/mol, 측정치: 972 g/mol)GC-Mass (Theoretical value: 973.13 g / mol, Measured value: 972 g / mol)
[실시예 1 ~ 30] 녹색 유기 전계 발광 소자의 제작Examples 1 to 30 Fabrication of Green Organic Electroluminescent Devices
합성예 1-30에서 합성한 화합물을 당업계에 공지된 방법으로 고순도 승화정제를 한 후 아래의 과정에 따라 녹색 유기 전계 발광 소자를 제작하였다.The compound synthesized in Synthesis Example 1-30 was subjected to high purity sublimation purification by a method known in the art, and then a green organic EL device was manufactured according to the following procedure.
먼저, ITO (Indium tin oxide)가 1500Å 두께로 박막 코팅된 유리 기판을 증류수로 초음파 세척하였다. 증류수 세척이 끝나면 이소프로필 알코올, 아세톤, 메탄올 등의 용제로 초음파 세척을 하고 건조시킨 후 UV OZONE 세정기 (Power sonic 405, 화신테크)로 이송시킨 다음 UV를 이용하여 상기 기판을 5분간 세정하고 진공 증착기로 기판을 이송하였다.First, an ITO (Indium tin oxide) was ultrasonically cleaned with distilled water to a glass substrate coated with a thin film of 1500Å thickness. After washing the distilled water, ultrasonic cleaning with a solvent such as isopropyl alcohol, acetone, methanol, etc., dried and transferred to a UV OZONE cleaner (Power sonic 405, Hwasin Tech), the substrate is cleaned by UV for 5 minutes and vacuum evaporator The substrate was transferred to.
이렇게 준비된 ITO 투명 전극 위에 m-MTDATA (60 nm)/TCTA (80 nm)/화합물 SPC-1 ~ SPC-30 각각 적용 + 10 % Ir(ppy)3 (300nm)/BCP (10 nm)/Alq3 (30 nm)/LiF (1 nm)/Al (200 nm) 순으로 적층하여 유기 전계 발광 소자를 제작하였다.M-MTDATA (60 nm) / TCTA (80 nm) / Compound SPC-1 to SPC-30 applied on the ITO transparent electrode thus prepared + 10% Ir (ppy) 3 (300 nm) / BCP (10 nm) / Alq 3 (30 nm) / LiF (1 nm) / Al (200 nm) were stacked to fabricate an organic EL device.
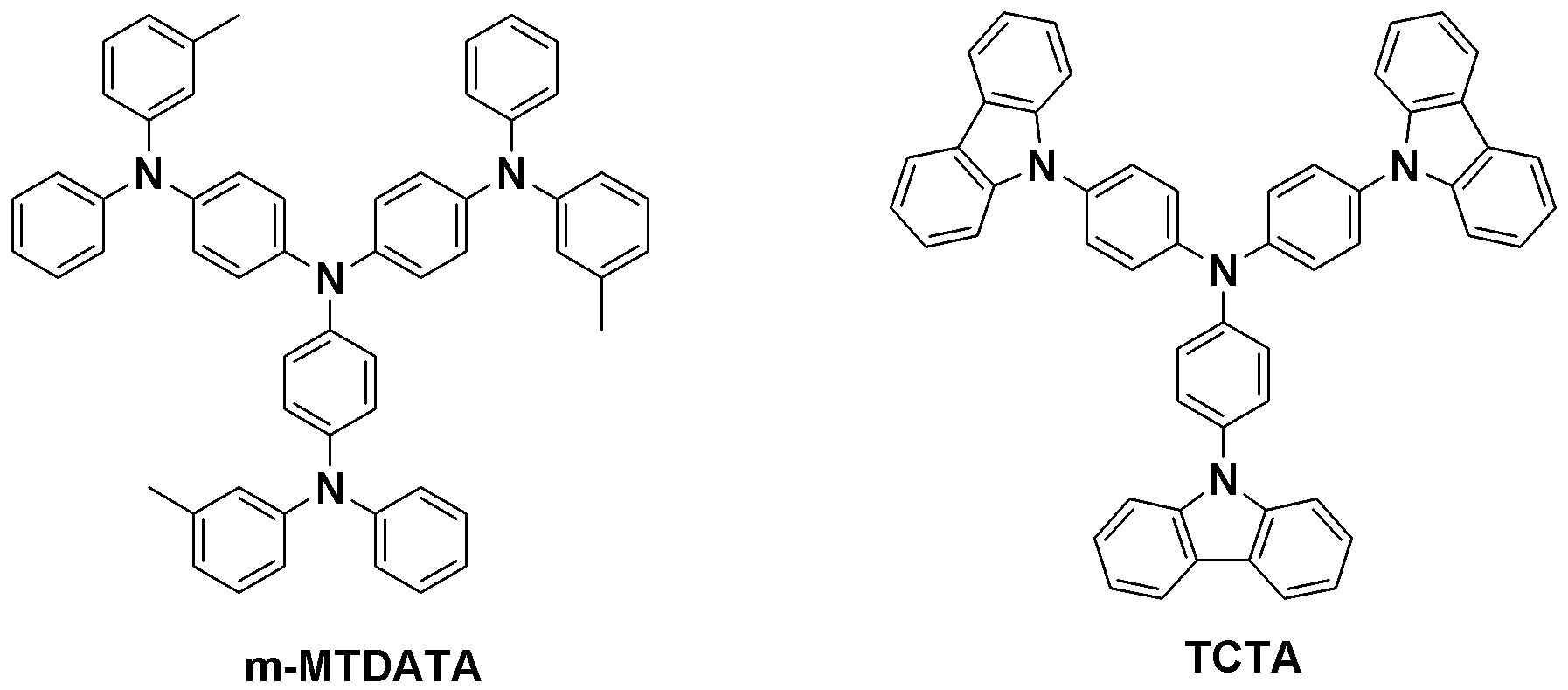
사용된 m-MTDATA, TCTA, Ir(ppy)3 및 BCP의 구조는 하기와 같다.The structures of m-MTDATA, TCTA, Ir (ppy) 3 and BCP used are as follows.
[비교예 1]Comparative Example 1
발광층 형성시 발광 호스트 물질로서 화합물 SPC-1 대신 하기와 같은 CBP를 사용하는 것을 제외하고는 실시예 1과 동일한 과정으로 유기 전계 발광 소자를 제작하였다.An organic electroluminescent device was manufactured in the same manner as in Example 1 except for using the following CBP instead of the compound SPC-1 as a light emitting host material when forming the light emitting layer.
[평가예 1][Evaluation Example 1]
실시예 1-30 및 비교예 1에서 제작한 각각의 유기 전계 발광 소자에 대하여 전류밀도10mA/㎠에서의 구동전압, 전류효율 및 발광 피크를 측정하고, 그 결과를 하기 표 1에 나타내었다.For each organic electroluminescent device produced in Example 1-30 and Comparative Example 1, the driving voltage, current efficiency and emission peak at a current density of 10 mA / cm 2 were measured, and the results are shown in Table 1 below.
표 1
상기 표 1에 나타낸 바와 같이, 본 발명에 따른 화합물(SPC-1~SPC-30)을 녹색 유기 전계 발광 소자의 발광층으로 사용하였을 경우(실시예 1 ~ 30) 종래 CBP를 사용한 녹색 유기 전계 발광 소자(비교예 1)와 발광파장은 유사하고, 효율 및 구동전압 면에서는 보다 우수한 성능을 나타내는 것을 확인할 수 있었다.As shown in Table 1 above, when the compounds (SPC-1 to SPC-30) according to the present invention were used as the light emitting layer of the green organic electroluminescent device (Examples 1 to 30), the green organic electroluminescent device using the conventional CBP It was confirmed that (Comparative Example 1) and the light emission wavelength were similar, and showed better performance in terms of efficiency and driving voltage.
[실시예 31 ~ 40] 청색 유기 전계 발광 소자의 제작Examples 31 to 40 Fabrication of Blue Organic Electroluminescent Devices
합성예 21-30에서 합성한 화합물을 당업계에 공지된 방법으로 고순도 승화정제를 한 후 아래의 과정에 따라 청색 유기 전계 발광 소자를 제작하였다.The compound synthesized in Synthesis Example 21-30 was subjected to high purity sublimation purification by a method known in the art, and then a blue organic EL device was manufactured according to the following procedure.
먼저, ITO (Indium tin oxide)가 1500Å 두께로 박막 코팅된 유리 기판을 증류수로 초음파 세척하였다. 증류수 세척이 끝나면 이소프로필 알코올, 아세톤, 메탄올 등의 용제로 초음파 세척을 하고 건조시킨 후 UV OZONE 세정기 (Power sonic 405)로 이송시킨 다음 UV를 이용하여 상기 기판을 5분간 세정하고 진공 증착기로 기판을 이송하였다.First, an ITO (Indium tin oxide) was ultrasonically cleaned with distilled water to a glass substrate coated with a thin film of 1500Å thickness. After washing the distilled water, ultrasonic cleaning with a solvent such as isopropyl alcohol, acetone, methanol, etc., dried and transferred to a UV OZONE cleaner (Power sonic 405), the substrate is cleaned using UV for 5 minutes and the substrate is vacuum-deposited. Transferred.
이렇게 준비된 ITO 투명 전극 위에 CuPc (10 nm)/ TPAC (30 nm)/ 화합물 SPC-21~SPC-30 각각 적용 + 7 % Flrpic (30nm)/ Alq3 (30 nm)/ LiF (0.2 nm)/Al (150 nm) 순으로 적층하여 유기 전계 발광 소자를 제작하였다.CuPc (10 nm) / TPAC (30 nm) / Compound SPC-21 ~ SPC-30 respectively applied on the prepared ITO transparent electrode + 7% Flrpic (30 nm) / Alq 3 (30 nm) / LiF (0.2 nm) / Al (150 nm) were laminated in order to produce an organic EL device.
사용된 CuPc, TPAC, Flrpic의 구조는 하기와 같다.The structures of CuPc, TPAC, and Flrpic used are as follows.
[비교예 2]Comparative Example 2
발광층 형성시 발광 호스트 물질로서 화합물 SPC-21 대신 비교예 1에서 사용된 CBP를 사용하는 것을 제외하고는 실시예 31과 동일한 과정으로 청색 유기 전계 발광 소자를 제작하였다.A blue organic electroluminescent device was manufactured in the same manner as in Example 31, except that CBP used in Comparative Example 1 was used instead of Compound SPC-21 as a light emitting host material when forming the emission layer.
[평가예 2][Evaluation Example 2]
실시예 31-40 및 비교예 2에서 제작한 각각의 청색 유기 전계 발광 소자에 대하여 전류밀도 10mA/㎠에서의 구동전압, 전류효율 및 발광 피크를 측정하고, 그 결과를 하기 표 2에 나타내었다.For each of the blue organic electroluminescent devices fabricated in Examples 31-40 and Comparative Example 2, the driving voltage, current efficiency, and emission peak at a current density of 10 mA / cm 2 were measured, and the results are shown in Table 2 below.
표 2
상기 표 2에 나타낸 바와 같이, 본 발명에 따른 화합물(SPC-21~SPC-30)을 청색 유기 전계 발광 소자의 발광층으로 사용하였을 경우(실시예 31 ~ 40) 종래 CBP를 사용한 청색 유기 전계 발광 소자(비교예 2)보다 전류효율 및 구동전압 면에서 우수한 성능을 나타내는 것을 확인할 수 있었다.As shown in Table 2, when the compound (SPC-21 ~ SPC-30) according to the present invention is used as a light emitting layer of the blue organic electroluminescent device (Examples 31 to 40) Blue organic electroluminescent device using a conventional CBP From Comparative Example 2, it was confirmed that the device exhibited better performance in terms of current efficiency and driving voltage.
본 발명의 화학식 1로 표시되는 화합물을 유기 전계 발광 소자의 발광 물질로 사용할 경우, 종래의 발광 물질에 비해 유기 전계 발광 소자의 효율(발광 효율 및 전력 효율), 수명, 휘도 및 구동전압 등을 향상시킬 수 있다. 따라서, 본 발명은 풀 칼라 유기 전계 발광 패널의 성능 및 수명을 향상시킬 수 있다.When the compound represented by Chemical Formula 1 of the present invention is used as a light emitting material of an organic light emitting device, the efficiency (light emitting efficiency and power efficiency), lifetime, luminance, driving voltage, etc. of the organic light emitting device are improved compared to the conventional light emitting material. You can. Therefore, the present invention can improve the performance and lifespan of a full color organic electroluminescent panel.
Claims (8)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2011-0144208 | 2011-12-28 | ||
| KR1020110144208A KR101376874B1 (en) | 2011-12-28 | 2011-12-28 | Novel compounds and organic electro luminescence device using the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013100507A1 true WO2013100507A1 (en) | 2013-07-04 |
Family
ID=48697882
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/KR2012/011341 Ceased WO2013100507A1 (en) | 2011-12-28 | 2012-12-24 | Novel compound and organic electroluminescence device including same |
Country Status (2)
| Country | Link |
|---|---|
| KR (1) | KR101376874B1 (en) |
| WO (1) | WO2013100507A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014094954A1 (en) * | 2012-12-18 | 2014-06-26 | Merck Patent Gmbh | Indenophenanthrene based compounds |
| CN106977519A (en) * | 2017-03-13 | 2017-07-25 | 江南大学 | A kind of blue emission luminous organic material and preparation method thereof |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20140034711A (en) * | 2012-09-12 | 2014-03-20 | 에스에프씨 주식회사 | Heterocyclic com pounds and organic light-emitting diode including the same |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004113468A1 (en) * | 2003-06-26 | 2004-12-29 | Covion Organic Semiconductors Gmbh | Novel materials for electroluminescence |
| JP2009194042A (en) * | 2008-02-13 | 2009-08-27 | Toyo Ink Mfg Co Ltd | Charge transport material for organic electroluminescence device containing carbazolyl group and use thereof |
| JP2010205986A (en) * | 2009-03-04 | 2010-09-16 | Mitsui Chemicals Inc | Organic transistor |
| KR20110041726A (en) * | 2009-10-16 | 2011-04-22 | 에스에프씨 주식회사 | Aromatic Compounds and Organic Electroluminescent Devices Using the Same |
Family Cites Families (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE4431039A1 (en) | 1994-09-01 | 1996-03-07 | Hoechst Ag | Poly (4,5,9,10-tetrahydropyrene-2,7-diyl) derivatives and their use as electroluminescent materials |
| EP2555270B1 (en) | 2010-03-31 | 2015-05-13 | Idemitsu Kosan Co., Ltd. | Material for organic electroluminescence element, and organic electroluminescence element using same |
-
2011
- 2011-12-28 KR KR1020110144208A patent/KR101376874B1/en not_active Expired - Fee Related
-
2012
- 2012-12-24 WO PCT/KR2012/011341 patent/WO2013100507A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004113468A1 (en) * | 2003-06-26 | 2004-12-29 | Covion Organic Semiconductors Gmbh | Novel materials for electroluminescence |
| JP2009194042A (en) * | 2008-02-13 | 2009-08-27 | Toyo Ink Mfg Co Ltd | Charge transport material for organic electroluminescence device containing carbazolyl group and use thereof |
| JP2010205986A (en) * | 2009-03-04 | 2010-09-16 | Mitsui Chemicals Inc | Organic transistor |
| KR20110041726A (en) * | 2009-10-16 | 2011-04-22 | 에스에프씨 주식회사 | Aromatic Compounds and Organic Electroluminescent Devices Using the Same |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014094954A1 (en) * | 2012-12-18 | 2014-06-26 | Merck Patent Gmbh | Indenophenanthrene based compounds |
| CN106977519A (en) * | 2017-03-13 | 2017-07-25 | 江南大学 | A kind of blue emission luminous organic material and preparation method thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20130075895A (en) | 2013-07-08 |
| KR101376874B1 (en) | 2014-03-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2016105161A2 (en) | Organic compound and organic electroluminescent element comprising same | |
| WO2015060684A2 (en) | Organic compound and organic electroluminescent device comprising same | |
| WO2013100497A1 (en) | Organic light-emitting compound and organic electroluminescent device using same | |
| WO2017003009A1 (en) | Organic light-emitting compound and organic electroluminescent device using same | |
| WO2013168927A2 (en) | Novel compound and organic electroluminescent device comprising same | |
| WO2017209488A1 (en) | Organic compound and organic electroluminescent device comprising same | |
| WO2020022860A1 (en) | Novel compound and organic light-emitting device using same | |
| WO2018230782A1 (en) | Organic compound and organic electroluminescent device comprising same | |
| WO2014098447A1 (en) | Organic compound and organic electroluminescent element comprising same | |
| WO2015133804A1 (en) | Organic compound and organic electroluminescent device comprising same | |
| WO2013191355A1 (en) | Novel compound and organic electroluminescent element comprising same | |
| WO2022031036A1 (en) | Organic light emitting device | |
| WO2020218680A1 (en) | Organic compound and organic electroluminescent diode using same | |
| WO2017111389A1 (en) | Organic compound and organic electroluminescent diode including same | |
| WO2015046982A2 (en) | Organic compound and organic electroluminescent element comprising same | |
| WO2022139455A1 (en) | Organic compound and organic electroluminescent device using same | |
| WO2015099477A2 (en) | Organic compound and organic light-emitting diode device using same | |
| WO2018186551A1 (en) | Organic compound and organic electroluminescent element comprising same | |
| WO2013100507A1 (en) | Novel compound and organic electroluminescence device including same | |
| WO2017105063A1 (en) | Organic compound and organic electroluminescent device comprising same | |
| WO2020130613A1 (en) | Organic compound and organic electroluminescent device comprising same | |
| WO2020111663A1 (en) | Organic compound and organic electroluminescent device using same | |
| WO2024144241A1 (en) | Organic compound and organic electroluminescent device using same | |
| WO2016105072A2 (en) | Organic compound and organic electroluminescent element comprising same | |
| WO2019203430A1 (en) | Organic light emitting compound and organic electroluminescent device using same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12862209 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 32PN | Ep: public notification in the ep bulletin as address of the adressee cannot be established |
Free format text: NOTING OF LOSS OF RIGHTS PURSUANT TO RULE 112(1) EPC (EPO FORM 1205A DATED 04/11/2014) |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12862209 Country of ref document: EP Kind code of ref document: A1 |