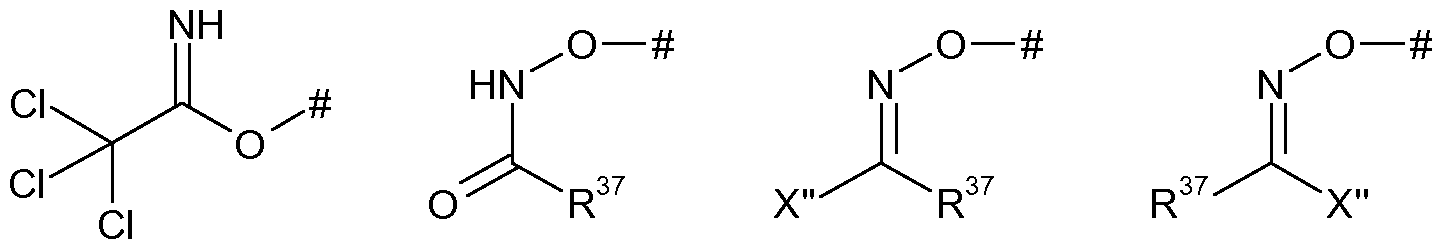WO2013092460A1 - Cyclic bisoxime microbicides - Google Patents
Cyclic bisoxime microbicides Download PDFInfo
- Publication number
- WO2013092460A1 WO2013092460A1 PCT/EP2012/075712 EP2012075712W WO2013092460A1 WO 2013092460 A1 WO2013092460 A1 WO 2013092460A1 EP 2012075712 W EP2012075712 W EP 2012075712W WO 2013092460 A1 WO2013092460 A1 WO 2013092460A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- phenyl
- halogen
- alkoxy
- optionally substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C(*)=*C(*)=NC(*)=* Chemical compound *C(*)=*C(*)=NC(*)=* 0.000 description 2
- YTQNUVLMVCPPFF-HJWRWDBZSA-N CCOC(C1=O)=CCC/C1=C/N(C)C Chemical compound CCOC(C1=O)=CCC/C1=C/N(C)C YTQNUVLMVCPPFF-HJWRWDBZSA-N 0.000 description 1
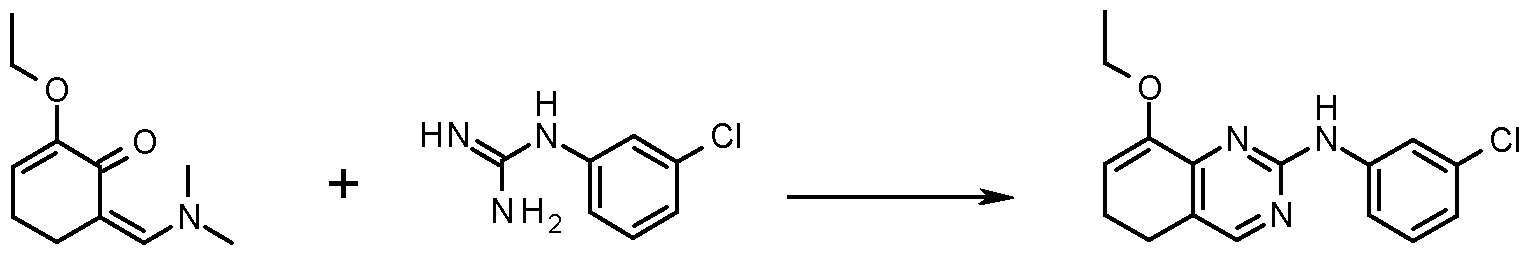
- HVDSRKTZCIDEAO-UHFFFAOYSA-N CCOC1=CCCc2c1nc(Nc1cccc(Cl)c1)nc2 Chemical compound CCOC1=CCCc2c1nc(Nc1cccc(Cl)c1)nc2 HVDSRKTZCIDEAO-UHFFFAOYSA-N 0.000 description 1
- UPQQXPKAYZYUKO-UHFFFAOYSA-N N=C(C(Cl)(Cl)Cl)O Chemical compound N=C(C(Cl)(Cl)Cl)O UPQQXPKAYZYUKO-UHFFFAOYSA-N 0.000 description 1
- DWLMIHRZURMFAQ-UHFFFAOYSA-N NC(Nc1cccc(Cl)c1)=N Chemical compound NC(Nc1cccc(Cl)c1)=N DWLMIHRZURMFAQ-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/70—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings condensed with carbocyclic rings or ring systems
- C07D239/72—Quinazolines; Hydrogenated quinazolines
- C07D239/74—Quinazolines; Hydrogenated quinazolines with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, attached to ring carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the present invention relates to novel microbiocidally active, in particular fungicidally active, cyclic bisoxime derivatives. It further relates to intermediates used in the preparation of these compounds, to compositions which comprise these compounds and to their use in agriculture or horticulture for controlling or preventing infestation of plants by phytopathogenic
- microorganisms preferably fungi.
- Fungicidally active bisoximes are described in WO08074418.
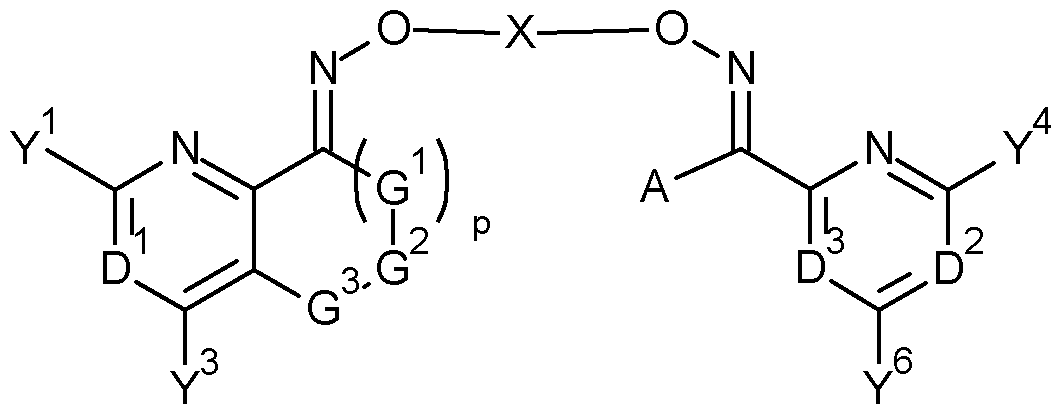
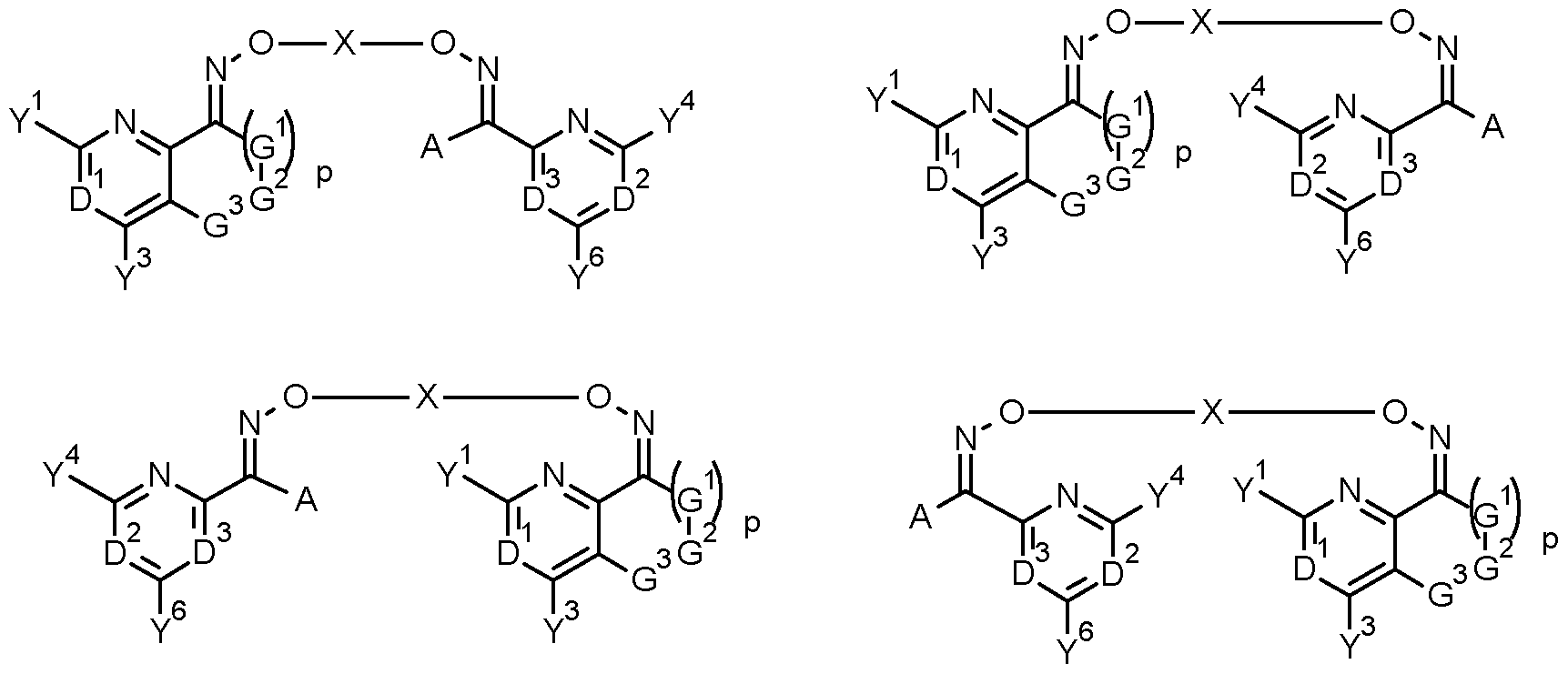
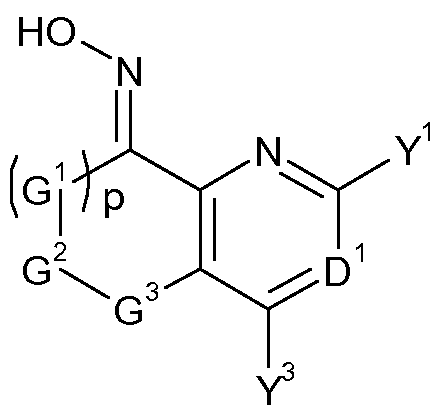
- the present invention accordingly relates to bisoxime derivatives of formula (I)
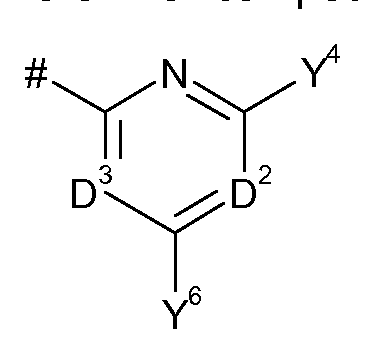
- D 1 represents N or C-Y 2 ;
- D 2 represents N or C-Y 5 ;
- D 3 represents N or C-Y 7 ;
- D 1 , D 2 or D 3 is N;
- A represents hydrogen, halogen, CN, OH, SH, Ci-C 8 alkylthio, Ci-C 8 alkylsulphinyl, d-C 8 alkylsulphonyl, NH 2 , Ci-Cio alkyl, C 3 -C 8 cycloalkyl, C 2 -C 8 alkenyl, C 2 -C 8 alkynyl, (R 10 O)carbonyl(Ci- C4-alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, d-C 4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C 3 -C 6 cycloal
- a and Y 7 together may be -(G 4 ) q -G 5 -G 6 -;
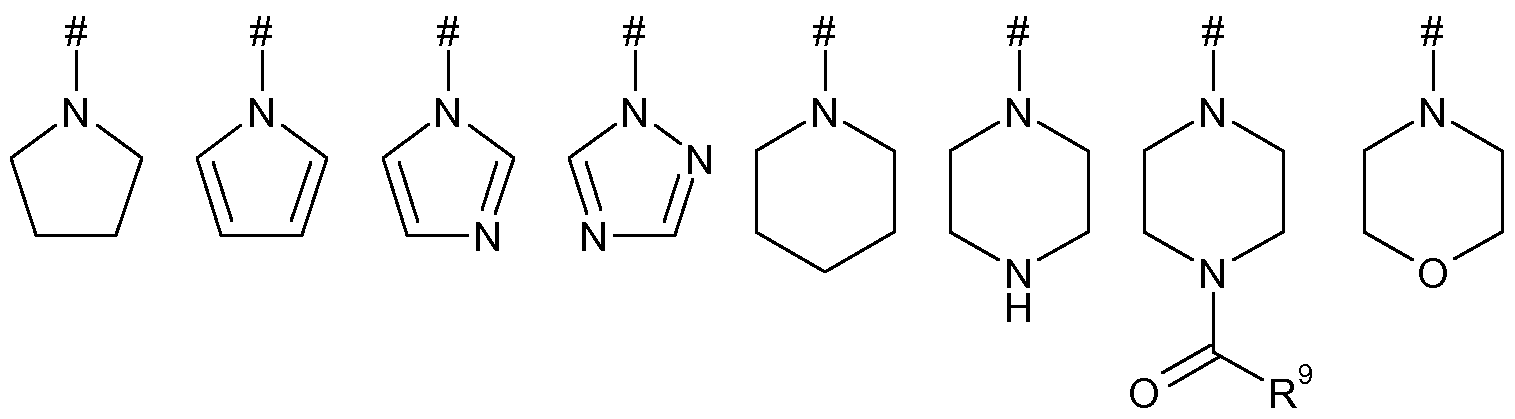
- X represents X-2, X-3, X-4 or X-5: #— ⁇ — ⁇ —# #— z— z— z— # #— z— z— z— # #— z— z— z— #
- each R 1 and R 2 independently of one another represent hydrogen, halogen, OH, d-C 4 alkyl,
- R 1 and R 2 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyi group;
- each R 3 and R 4 independently of one another represent hydrogen, halogen, C1-C4 alkyl or C1-C4 haloalkyi;
- each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, OH, Ci- C 4 alkyl, C1-C4 haloalkyi, C 3 -C 6 cycloalkyi, C 3 -C 6 halocycloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, d-C 4 alkoxy and C1-C4 haloalkoxy;
- R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyi group;
- groupings X-2, X-3, X-4 and X-5 may contain at most one ring which contains either only one of the radicals Z 1 to Z 14 or two radicals Z 1 to Z 14 or three radicals Z 1 to Z 14 or four radicals Z 1 to Z 14 as ring members; and wherein radicals Z 1 , Z 2 , Z 3 , Z 5 , Z 6 , Z 9 , Z 10 and Z 14 are not substituted by OH;
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, halogen, CN, N0 2 , Ci-C 8 alkyl, Ci-C 4 -alkoxy-Ci-C 4 -alkyl, Ci-C 4 -alkoxy-Ci-C 4 -alkoxy-Ci-C 4 -alkyl, C 3 -C 8 cycloalkyi, C 2 -C 8 alkenyl, C 2 -C 8 alkynyl, phenyl, pyridyl, pyrimidinyl, COR 9 , OR 10 , SH, Ci-C 8 alkylthio, Ci-C 8 alkylsulphinyl, C r C 8 alkylsulphonyl, N(R n ) 2 , C0 2 R 10 , 0(CO)R 9 , CON(R n ) 2 , NR
- Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from 0, S, N and N(R n ), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 , or Y 6 and Y 7 is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, d-C 4 haloal
- each R 9 independently of one another represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, C 2 - C 8 alkenyl, C 2 -C 8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy;
- each R 10 independently of one another represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, C 3 -C 8 alkenyl, C 3 -C 8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups
- halogen independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy, Ci-C 4 haloalkoxy and CrC 4 -alkoxy-d-C 4 -alkyl;
- each R 11 independently of one another represents hydrogen, OH, d-C 8 alkyl, Ci-C 8 alkoxy, Ci-C 8 -alkoxy-Ci-C 4 -alkyl, C 3 -C 8 alkenyl, C 3 -C 8 alkynyl, COR 9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, d-C 4 alkyl, d-C 4 haloalkyi, C C 4 alkoxy, d-C 4 haloalkoxy, Ci-C 4 alkylthio, Ci-C 4 alkylsulphinyl and d-C 4 alkylsulphonyl;
- radicals R 11 wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different;
- G 1 , G 2 , G 4 and G 5 independently of one another represent -C(R 12 R 13 )-, 0, N(R 14 ) or S;
- G 3 and G 6 independently of one another represent -C(R 12 R 13 )-, 0, N(R 14 ) or S;
- each R 12 and R 13 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci- C 4 haloalkyl, Ci-C 4 alkoxy or Ci-C 4 haloalkoxy;
- R 14 represents hydrogen, OH, Ci-C 4 alkyl, Ci-C 4 alkoxy, C 3 -C 6 cycloalkyl, Ci-C 8 alkylcarbonyl or Ci-C 8 haloalkylcarbonyl;
- p and q are each independently 0, 1 or 2;
- Halogen either as a lone substituent or in combination with another substituent (e.g.
- haloalkyl is generally fluorine, chlorine, bromine or iodine, and usually fluorine, chlorine or bromine.
- Each alkyl moiety (including the alkyl moiety of alkoxy, alkylthio, etc.) is a straight or branched chain and, depending on the number of carbon atoms it contains, is, for example, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, /so-propyl, sec-butyl, /so-butyl, tert-butyl, neo-pentyl, n-heptyl or 1,3-dimethylbutyl, and usually methyl or ethyl.
- alkenyl and alkynyl groups can be mono- or di-unsaturated and are examples thereof are derived from the above mentioned alkyl groups.
- Haloalkyl moieties are alkyl moieties which are substituted by one or more of the same or different halogen atoms and are, for example, monofluoromethyl, difluoromethyl,
- trifluoromethyl monochloromethyl, dichloromethyl, trichloromethyl, 2,2,2-trifluoroethyl, 2,2- difluoroethyl, 2-fluoroethyl, 1,1 -difluoroethyl, 1-fluoroethyl, 2-chloroethyl, pentafluoroethyl, 1,1- difluoro-2,2,2-trichloroethyl, 2,2,3,3-tetrafluoroethyl and 2,2,2 -trichloroethyl, and typically trichloromethyl, difluorochloromethyl, difluoromethyl, trifluoromethyl and dichlorofluoromethyl.
- Alkoxy is, for example, methoxy, ethoxy, propoxy, / ' so-propoxy, n-butoxy, /so-butoxy, sec- butoxy and tert-butoxy, and usually methoxy or ethoxy.
- Haloalkoxy is, for example, fluoromethoxy, difluoromethoxy, trifluoromethoxy, 2,2,2- trifluoroethoxy, 1,1,2,2-tetrafluoroethoxy, 2-fluoroethoxy, 2-chloroethoxy, 2,2-difluoroethoxy and 2,2,2-trichloroethoxy, and usually difluoromethoxy, 2-chloroethoxy and trifluoromethoxy.
- Alkylthio is, for example, methylthio, ethylthio, propylthio, /so-propylthio, n-butylthio, iso- butylthio, sec-butylthio or tert- butylthio, and usually methylthio or ethylthio.
- Alkylsulphonyl is, for example, methylsulphonyl, ethylsulphonyl, propylsulphonyl, iso- propylsulphonyl, n-butylsulphonyl, / ' so-butylsulphonyl, sec-butylsulphonyl or tert-butylsulphonyl, and usually methylsulphonyl or ethylsulphonyl.
- Alkylsulphinyl is, for example, methylsulphinyl, ethylsulphinyl, propylsulphinyl, iso- propylsulphinyl, n-butylsulphinyl, /so-butylsulphinyl, sec-butylsulphinyl or tert-butylsulphinyl, and usually methylsulphinyl or ethylsulphinyl
- Cycloalkyl may be saturated or partially unsaturated, preferably fully saturated, and is, for example, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
- Alkoxyalkyl is, for example, methoxymethyl, methoxyethyl, ethoxymethyl, ethoxyethyl, n- propoxymethyl, n-propoxyethyl, /so-propoxymethyl or /so-propoxyethyl.
- Aryl includes phenyl, naphthyl, anthracyl, fluorenyl and indanyl, but is usually phenyl.
- Carbocycle includes cycloalkyl groups and aryl groups.
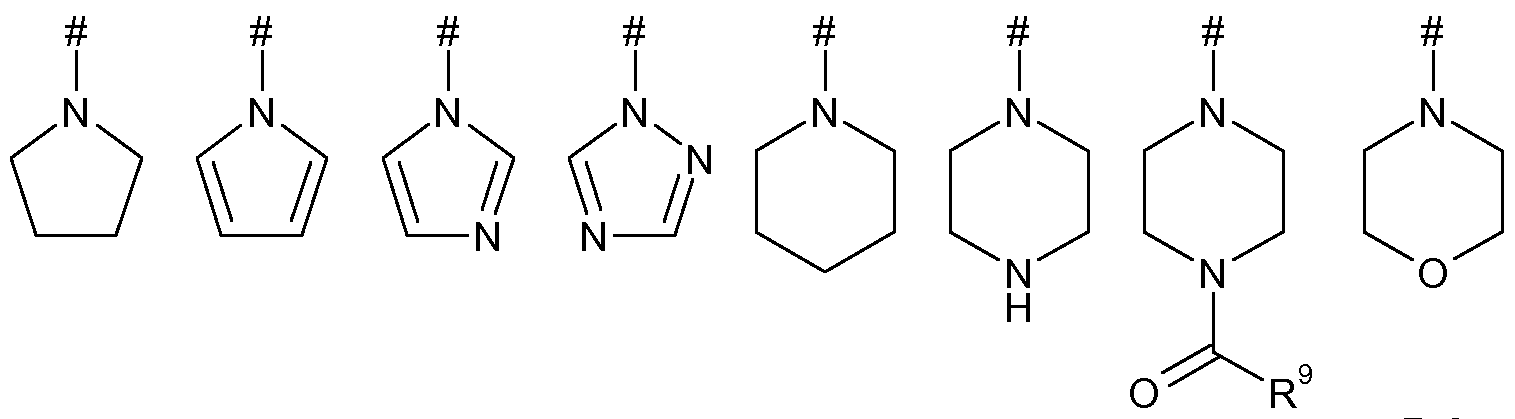
- Heterocycloalkyl is a non-aromatic ring that may be saturated or partially unsaturated, preferably fully saturated, containing carbon atoms as ring members and at least one heteroatom selected from 0, S and N as ring members.
- Examples include oxiranyl, oxetanyl, tetrahydrofuranyl, tetrahydropyranyl, 1,3-dioxolanyl, 1,4-dioxanyl, aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, oxazinanyl, morpholinyl, thiomorpholinyl, imidazolidinyl, pyrazolidinyl and piperazinyl, preferably morpholinyl, pyrrolidinyl, piperdinyl and piperazinyl, more preferably morpholinyl and pyrollidinyl.
- Heteroaryl is, for example, a monovalent monocyclic or bicyclic aromatic hydrocarbon radical.
- monocyclic groups include pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, pyrrolyl, pyrazolyl, imidazolyl, triazolyl, tetrazolyl, furanyl, thiophenyl, oxazolyl, isoxazolyl, oxadiazolyl, thiazolyl, isothiazolyl, and thiadiazolyl.
- bicyclic groups include quinolinyl, cinnolinyl, quinoxalinyl, benzimidazolyl, benzothiophenyl, and benzothiadiazolyl.
- Monocyclic heteroaryl groups are preferred, preferably pyridyl, pyrrolyl, imidazolyl and triazolyl, e.g. 1,2,4 triazolyl, pyridyl and imidazolyl being most preferred.
- heterocycle and “heterocyclic ring” are used interchangeably and are defined to include heterocycloalkyl and heteroaryl groups. Any reference herein to a heterocycle or heterocyclic ring preferably refers to the specific examples given under the definition of heteroaryl and heterocycloalkyl above, and are preferably morpholinyl, pyrrolidinyl, piperdinyl, piperazinyl pyridyl, pyrrolyl, imidazolyl and triazolyl, e.g. 1,2,4 triazolyl, more preferably morpholinyl, pyrollidinyl, pyridyl and imidazolyl.
- a moiety is indicated as being (optionally) substituted, e.g. alkyl, this includes those moieties where they are part of a larger group, e.g. the alkyl in the alkylthio group.
- a moiety is indicated as being optionally substituted by one or more other groups, preferably there are one to five optional substituents, more preferably one to three optional substituents.
- D 1 represents N or C-Y 2 ;
- D 2 represents N or C-Y 5 ;
- D 3 represents N or C-Y 7 ;
- D 1 , D 2 or D 3 is N;
- D 1 represents C-Y 2
- D 2 represents N
- D 3 represents C-Y 7 .
- D 1 represents C-Y 2
- D 2 represents C-Y 5
- D 3 represents N.
- D 1 represents N
- D 2 represents C-Y 5
- D 3 represents C-Y 7 .
- D 1 represents N
- D 2 represents N
- D 3 represents C-Y 7 .
- D 1 represents N
- D 2 represents C-Y 5
- D 3 represents N
- A represents hydrogen, halogen, CN, OH, SH, Ci-C 8 alkylthio, Ci-C 8 alkylsulphinyl, d-C 8 alkylsulphonyl, NH 2 , Ci-Cio alkyl, C 3 -C 8 cycloalkyl, C 2 -C 8 alkenyl, C 2 -C 8 alkynyl, (R 10 O)carbonyl(Ci- C 4 -alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, d-C 4 haloalkyl, Ci-C 4 alkoxy
- a and Y 7 together may be -(G 4 ) q -G 5 -G 6 -.
- A represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, phenyl or pyridyl, wherein the alkyl, cycloalkyi, phenyl and pyridyl are optionally substituted by one or more groups, e.g.
- one to five groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, OH, C1-C4 alkoxy, C1-C4 haloalkoxy and C 3 -C 6 cycloalkyi.
- A represents hydrogen, C1-C4 alkyl, phenyl or pyridyl, wherein alkyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, OH, C1-C4 alkoxy and d-C 4 haloalkoxy, and wherein phenyl and pyridyl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, OH, d-C 4 alkoxy, CrC 4 haloalkoxy and C 3 -C 6 cycloalkyi.
- A represents hydrogen, C1-C4 alkyl, phenyl or pyridin-2-yl, wherein the phenyl and pyridin-2-yl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, methyl, halomethyl, methoxy and
- A represents C1-C4 alkyl.
- compounds A represents pyridyl, optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C4-alkyl, C1-C4- haloalkyl, d-C 4 -alkoxy, d-C 4 -haloalkoxy, C 3 -C 6 cycloalkyi and a 5 or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms.
- the heterocycle is preferably one as defined herein, preferably morpholinyl, pyrrolidinyl, piperdinyl, piperazinyl, pyridyl, pyrrolyl, imidazolyl or triazolyl, e.g. 1,2,4 triazolyl, more preferably morpholinyl, pyrollidinyl, pyridyl or imidazolyl.
- this preferred group of compounds A preferably represents pyridin-2-yl, optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, d- C 4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy, C1-C4 haloalkoxy, C 3 -C 6 cycloalkyi and a 5 or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms.
- the heterocycle is preferably one as defined herein, preferably morpholinyl, pyrrolidinyl, piperdinyl, piperazinyl, pyridyl, pyrrolyl, imidazolyl or triazolyl, e.g. 1,2,4 triazolyl, more preferably morpholinyl, pyrollidinyl, pyridyl or imidazolyl.
- A represents hydrogen, C1-C4 alkyl, C 2 -C 4 alkenyl, phenyl or pyridyl, wherein the alkyl, alkenyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from d-C 4 alkyi, Ci-C 4 haloalkyi, halogen, CN, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy.
- A represents hydrogen, halogen, CN, OH, SH, Ci-Cs alkylthio, Ci-C 8 alkylsulphinyl, Ci-C 8 alkylsulphonyl, NH 2 , C1-C10 alkyi, C 3 -C 8 cycloalkyl, C 2 -C 8 alkenyl, C 2 -C 8 alkynyl, (d-C 4 alkyloxycarbonyl) Ci-C 4 alkyi, (Ci-C 4 alkyl)0 2 C, phenyl or pyridyl, wherein the alkyi, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyi, Ci-C 4 haloalkyi, Ci-C 4 alkoxy, d-
- A represents hydrogen, (Ci-C 4 alkyl)0 2 C, C1-C10 alkyi, phenyl or pyridyl, wherein the alkyi, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, d-C 4 alkyi, Ci-C 4 haloalkyi, C 3 -C 6 cycloalkyl and a 5- or 6-membered heterocycle containing one to three nitrogen atoms.
- X represents X-2, X-3, X-4 or X-5:
- X represents X-3 or X-5. More preferably X represents X-3.
- Z 1 , Z 2 , Z 3 , Z 5 , Z 6 , Z 7 , Z 8 , Z 9 , Z 10 , Z 11 , Z 13 and Z 14 independently of one another represent methylene, halomethylene, CH(CH 3 ) or C(CH 3 ) 2 .
- Z 1 , Z 2 , Z 3 , Z 5 , Z 6 , Z 7 , Z 8 , Z 9 , Z 10 , Z 11 , Z 13 and Z 14 represent methylene or halomethylene.
- Z 1 , Z 2 , Z 3 , Z 5 , Z 6 , Z 7 , Z 8 , Z 9 , Z 10 , Z 11 , Z 13 and Z 14 represent methylene.
- Each R 1 and R 2 independently of one another represent hydrogen, halogen, OH, Ci-C 4 alkyl, C1-C4 haloalkyi, C 3 -C 6 cycloalkyi, C 3 -C 6 halocycloalkyi, C1-C4 alkylthio, C1-C4 alkylsulphinyl, C1-C4 alkylsulphonyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups, e.g.
- R 1 and R 2 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyi group.
- Each R 3 and R 4 independently of one another represent hydrogen, halogen, C1-C4 alkyl or C1-C4 haloalkyi.
- each R 3 and R 4 independently of one another represent hydrogen, halogen, methyl or halomethyl.
- Each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, OH, Ci- C 4 alkyl, C1-C4 haloalkyi, C 3 -C 6 cycloalkyi, C 3 -C 6 halocycloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups, e.g.
- R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyi group.
- each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, OH, C1-C4 alkyl, C1-C4 haloalkyi, C 3 -C 6 cycloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, d-C 4 alkyl, C1-C4 haloalkyi, d-C 4 alkoxy and d-C 4 haloalkoxy; or R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyi group.
- each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, OH, d-C 4 alkyl, d-C 4 haloalkyi, phenyl or CN wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy; or R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group optionally substituted by halogen.
- each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, d-C 4 alkyl, C1-C4 haloalkyi or phenyl, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy; or R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group optionally substituted by halogen.
- Y 1 , Y 2 , ⁇ 3 , ⁇ 4 , ⁇ 5 , Y 6 and ⁇ 7 independently of one another represent hydrogen, halogen, CN, N0 2 , Ci-C 8 alkyl, Ci-C 4 -alkoxy-Ci-C 4 -alkyl, Ci-C 4 -alkoxy-Ci-C 4 -alkoxy-Ci-C 4 -alkyl, C 3 -C 8 cycloalkyi, C 2 -C 8 alkenyl, C 2 -C 8 alkynyl, phenyl, pyridyl, pyrimidinyl, COR 9 , OR 10 , SH, Ci-C 8 alkylthio, Ci-C 8 alkylsulphinyl, d-C 8 alkylsulphonyl, N(R n ) 2 , C0 2 R 10 , 0(CO)R 9 , CON(R n ) 2 , NR
- Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from 0, S, N and N(R n ), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, d-C 4 haloalkyi
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one represent hydrogen, halogen, OH, CN, Ci-C 8 alkyl, Ci-C 8 haloalkyi, Ci-C 8 alkoxy, Ci-C 8 haloalkoxy, Ci-C 8 alkylthio, C 3 -C 8 cycloalkyi, phenyl, pyridyl, N(R n ) 2 , or NR n COR 9 , wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and d-C 4 haloalkoxy;
- Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from N and N(R n ) 2 and wherein the ring formed by Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy;
- each R 9 independently represents d-C 8 alkyl or Ci-C 8 haloalkyi
- each R 11 independently of one another represents hydrogen, Ci-C 8 alkyl, COR 9 or phenyl, wherein the phenyl is optionally substituted by one or more group independently selected from halogen, CN, NH 2 , N0 2 , OH, d-C 4 alkyl, d-C 4 haloalkyi, d-C 4 alkoxy, d-C 4 haloalkoxy, Ci-C 4 alkylthio, Ci-C 4 alkylsulphinyl and d-C 4 alkylsulphonyl; wherein when two radicals R are attached to the same nitrogen atom, these radicals can be identical or different;
- cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl.
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, CN, OH, halogen, d-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy, d-C 4 haloalkoxy, C 3 -C 6 cycloalkyl, Ci-C 4 alkylthio, N(R n ) 2 , NR n COR 9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, d-C 4 alkyl, Ci-C 4 haloalkyi, d- C 4 alkoxy and d-C 4 haloalkoxy;
- each R 9 independently represents d-C 4 alkyl or Ci-C 4 haloalkyi
- each R 11 independently of one another represents hydrogen, Ci-Cs alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, d-C 4 alkyl, d-C 4 haloalkyi, d-C 4 alkoxy and d-C 4 haloalkoxy; wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different;
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, CN, OH, halogen, d-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy, d-C 4 haloalkoxy, C 3 -C 6 cycloalkyl, d-C 4 alkylthio, N(R n ) 2 , NR n COR 9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
- each R 9 independently represents d-C 4 alkyl or Ci-C 4 haloalkyi; wherein each R independently of one another represents hydrogen, Ci-C 8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy.
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, CN, OH, halogen, d-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy, d-C 4 haloalkoxy, C 3 -C 6 cycloalkyi, d-C 4 alkylthio, N(R n ) 2 , NR n COR 9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
- each R 9 independently represents d-C 4 alkyl or Ci-C 4 haloalkyi
- each R 11 independently of one another represents hydrogen, Ci-C 8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy.
- Each R 9 independently of one another represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, C 2 - C 8 alkenyl, C 2 -C 8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups, e.g.
- one to five groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, d-C 4 haloalkyi, Ci- C 4 alkoxy and Ci-C 4 haloalkoxy.
- each R 9 independently of one another represent Ci-Cs alkyl or d-C 8 haloalkyi.
- each R 9 independently of one another represent d-C 4 alkyl or Ci-C 4 haloalkyi.
- Each R 10 independently of one another represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, C 3 -C 8 alkenyl, C 3 -C 8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups, e.g.
- one to five groups independently selected from halogen, CN, NH 2 , N0 2 , OH, d-C 4 alkyl, Ci-C 4 haloalkyi, d-C 4 alkoxy, Ci-C 4 -haloalkoxy and Ci-C 4 -alkoxy-Ci-C 4 -alkyl.
- Each R 11 independently of one another represents hydrogen, OH, Ci-C 8 alkyl, Ci-C 8 alkoxy, Ci-C 8 -alkoxy-Ci-C 4 -alkyl, C 3 -C 8 alkenyl, C 3 -C 8 alkynyl, COR 9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, d-C 4 alkyl, d-C 4 haloalkyi, d-C 4 alkoxy, d-C 4 haloalkoxy, Ci-C 4 alkylthio, Ci-C 4 alkylsulphinyl and d-C 4 alkylsulphonyl;
- radicals R 11 when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different; wherein when two radicals R are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C 4 alkoxy or Ci-C 4 haloalkoxy;
- each R 11 independently of one another represents hydrogen, d-C 8 alkyl, COR 9 or phenyl, wherein the phenyl is optionally substituted by one or more group independently selected from halogen, CN, NH 2 , N0 2 , OH, d-C 4 alkyl, d-C 4 haloalkyi, C C 4 alkoxy, d-C 4 haloalkoxy, Ci-C 4 alkylthio, Ci-C 4 alkylsulphinyl and d-C 4 alkylsulphonyl;
- radicals R 11 wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different;
- cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl.
- each R 11 independently of one another represents hydrogen, d-C 8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy; wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different; wherein when two radicals R are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C 4 alkoxy or Ci-C 4 haloalkoxy.
- each R 11 independently of one another represents hydrogen, Ci-Cs alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, d-C 4 alkoxy and Ci-C 4 haloalkoxy.
- G 1 , G 2 , G 4 and G 5 independently of one another represent -C(R 12 R 13 )- , 0, N(R 14 ) or S;
- G 3 and G 6 independently of one another represent -C(R 12 R 13 )-, 0, N(R 14 ) or S;
- G 1 , G 2 , G 4 and G 5 independently of one another represent -C(R 12 R 13 )- ;
- G 3 and G 6 independently of one another represent -C(R 12 R 13 )-, 0, N(R 14 ) or S;
- G 1 , G 2 , G 4 and G 5 independently of one another represent -C(R 12 R 13 )-;
- G 3 and G 6 independently of one another represent -C(R 12 R 13 )-, 0, N(R 14 ) or S.
- G 1 , G 2 , G 3 , G 4 , G 5 and G 6 independently of one another represent - C(R 12 R 13 )-.
- G 1 , G 2 , G 3 , G 4 , G 5 and G 6 represent methylene.
- Each R 12 and R 13 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci-C 4 haloalkyi, d-C 4 alkoxy or Ci-C 4 haloalkoxy.
- R 14 represents hydrogen, OH, d-C 4 alkyl, Ci-C 4 alkoxy, d-C 6 -cycloalkyl, Ci-Cs alkylcarbonyl or Ci-Q haloalkylcarbonyl.
- p and q are each independently of one another 0, 1 or 2.
- p and q are each independently of one another 1 or 2.
- D 1 represents N or C-Y 2 ;
- D 2 represents N or C-Y 5 ;
- D 3 represents N or C-Y 7 ;
- D 1 , D 2 or D 3 is N;
- A represents hydrogen, halogen, CN, OH, SH, Ci-C 8 alkylthio, Ci-C 8 alkylsulphinyl, d-C 8 alkylsulphonyl, NH 2 , Ci-Cio alkyl, C 3 -C 8 cycloalkyl, C 2 -C 8 alkenyl, C 2 -C 8 alkynyl, (R 10 O)carbonyl(Ci- C4-alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci- haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C 3
- a and Y 7 together may be -(G 4 ) q -G 5 -G 6 -.
- X represents X-2, X-3, X-4 or X-5:
- each R 1 and R 2 independently of one another represent hydrogen, halogen, OH, Ci-d alkyl,
- C1-C4 haloalkyl C 3 -C 6 cycloalkyl, C 3 -C 6 halocycloalkyl, Ci-C 4 alkylthio, C1-C4 alkylsulphinyl, Ci-C 4 alkylsulphonyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyl, Ci-d alkoxy and C1-C4 haloalkoxy;
- R 1 and R 2 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyl group or a C 3 -C 6 halocycloalkyl group;
- each R 3 and R 4 independently of one another represent hydrogen, halogen, d-C 4 alkyl or C1-C4 haloalkyl;
- each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, OH, Ci- C 4 alkyl, Ci-d haloalkyl, C 3 -C 6 cycloalkyl, C 3 -C 6 halocycloalkyl, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, d-d alkyl, Ci- haloalkyl, Ci-d alkoxy and Ci-d haloalkoxy;
- R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyl group or a C 3 -C 6 halocycloalkyl group; wherein the groupings X-2, X-3, X-4 and X-5 may contain at most one ring which contains either only one of the radicals Z 1 to Z 14 or two radicals Z 1 to Z 14 or three radicals Z 1 to Z 14 or four radicals Z 1 to Z 14 as ring members; and wherein radicals Z 1 , Z 2 , Z 3 , Z 5 , Z 6 , Z 9 , Z 10 and Z 14 are not substituted by OH;
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, halogen, CN,
- Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from 0, S, N and N(R n ), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, C C 4 alkyl, C r C 4 haloalkyi, C
- each R 9 independently of one another represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, C 2 - C 8 alkenyl, C 2 -C 8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy;
- each R 10 independently of one another represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, C 3 -C 8 alkenyl, C 3 -C 8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups
- halogen independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 -haloalkyl, Ci-C 4 alkoxy, Ci-C 4 haloalkoxy and d-C 4 -alkoxy-Ci-C 4 -alkyl;
- each R 11 independently of one another represents hydrogen, OH, Ci-C 8 alkyl, Ci-C 8 alkoxy, Ci-C 8 -alkoxy-Ci-C 4 -alkyl, C 3 -C 8 alkenyl, C 3 -C 8 alkynyl, COR 9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, C C 4 alkoxy, C C 4 haloalkoxy, Ci-C 4 alkylthio, Ci-C 4 alkylsulphinyl and d-C 4 alkylsulphonyl;
- radicals R 11 wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different;
- cycle formed is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy;
- G 1 , G 2 , G 4 and G 5 independently of one another represent -C(R 12 R 13 )-;
- G 3 and G 6 independently of one another represent -C(R 12 R 13 )-, 0, N(R 14 ) or S;
- each R 12 and R 13 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci- C 4 haloalkyi, Ci-C 4 alkoxy or Ci-C 4 haloalkoxy;
- R 14 represents hydrogen, OH, d-C 4 alkyl, Ci-C 4 alkoxy, C 3 -C 6 cycloalkyi, Ci-C 8 alkylcarbonyl or Ci-C 8 haloalkylcarbonyl;
- p and q are each independently 0, 1 or 2.
- A represents hydrogen, Ci-C 8 alkyl, C 3 -C 8 cycloalkyi, phenyl or pyridyl, wherein the alkyl, cycloalkyi, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, OH, d-C 4 alkoxy, Ci-C 4 haloalkoxy and C 3 -C 6 cycloalkyi;
- X represents X-3
- Z 3 and Z 5 independently of one another represent methylene or halomethylene
- each R 3 and R 4 independently of one another represent hydrogen, halogen, methyl or halomethyl
- each R 5 , R 6 , R 7 and R 8 independently of one another represent hydrogen, halogen, OH, Ci- C 4 alkyl, Ci-C 4 haloalkyi, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy;
- R 5 and R 6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyl group or a C 3 -C 6 halocycloalkyl group;
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one represent hydrogen, halogen, OH, CN, d-
- Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from N and N(R n ) 2 and wherein the ring formed by Y 1 and Y 2 , Y 2 and Y 3 , Y 4 and Y 5 , Y 5 and Y 6 or Y 6 and Y 7 is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy and C1-C4 haloalkoxy;
- each R 9 independently represents Ci-C 8 alkyl or Ci-C 8 haloalkyi
- each R 11 independently of one another represents hydrogen, Ci-C 8 alkyl, COR 9 or phenyl, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy, C1-C4 haloalkoxy, C1-C4 alkylthio, C1-C4 alkylsulphinyl and C1-C4 alkylsulphonyl;
- radicals R 11 wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different;
- cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl;
- G 1 , G 2 , G 3 , G 4 , G 5 and G 6 independently of one another represent -C(R 12 R 13 )-;
- A represents hydrogen, Ci-C 4 alkyl, phenyl or pyridyl, wherein alkyl is optionally substituted by one or more groups independently selected from halogen, OH, C1-C4 alkoxy and C1-C4 haloalkoxy, and wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyl, OH, d-C 4 alkoxy, Ci-C 4 haloalkoxy and C 3 -C 6 cycloalkyi;
- X represents X-3
- Z 3 and Z 5 represent methylene
- each R 3 and R 4 independently of one another represent hydrogen, halogen, methyl or halomethyl
- each R 5 and R 6 independently of one another represent hydrogen, halogen, OH, d-C 4 alkyl, Ci-C 4 haloalkyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy; or R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyl group;
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, CN, OH, halogen, Ci-C 4 alkyl, Ci-C 4 haloalkyl, Ci-C 4 alkoxy, Ci-C 4 haloalkoxy, Ci-C 4 alkylthio, C 3 -C 6 cycloalkyi, N(R n ) 2 , NR n COR 9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyl, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy;
- each R 9 independently represents d-C 4 alkyl or Ci-C 4 haloalkyl
- each R 11 independently of one another represents hydrogen, Ci-C 8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, Ci-C 4 alkyl, d-C 4 haloalkyl, C C 4 alkoxy and C r C 4 haloalkoxy;
- radicals R 11 wherein when two radicals R 11 are attached to the same nitrogen atom, these radicals can be identical or different;
- G 1 , G 2 , G 3 , G 4 , G 5 and G 6 represent methylene; p and q are each independently 0, 1 or 2.
- A represents hydrogen, d-C 4 alkyl, phenyl or pyridin-2-yl wherein phenyl and pyridin-2-yl are optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy;
- X represents X-3
- Z 3 and Z 5 represent methylene
- each R 3 and R 4 independently of one another represent hydrogen, halogen, methyl or halomethyl
- each R 5 and R 6 independently of one another represent hydrogen, halogen, OH, d-C 4 alkyl,
- Ci-C 4 haloalkyl, phenyl or CN wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy; or R 5 and R 6 together with the carbon atom to which they are attached may form a C 3 -C 6 cycloalkyi group or a C 3 -C 6 halocycloalkyl group;
- Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 and Y 7 independently of one another represent hydrogen, CN, OH, halogen, Ci-C 4 alkyl, Ci-C 4 haloalkyl, Ci-C 4 alkoxy, Ci-C 4 haloalkoxy, d-C 4 alkylthio, C 3 -C 6 cycloalkyi, N(R n ) 2 , NR n COR 9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
- each R 9 independently represents d-C 4 alkyl or Ci-C 4 haloalkyl
- each R 11 independently of one another represents hydrogen, d-C 8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyl, Ci-C 4 alkoxy and Ci-C 4 haloalkoxy;
- G 1 , G 2 , G 3 , G 4 , G 5 and G 6 represent methylene
- p and q are each independently 0, 1 or 2.
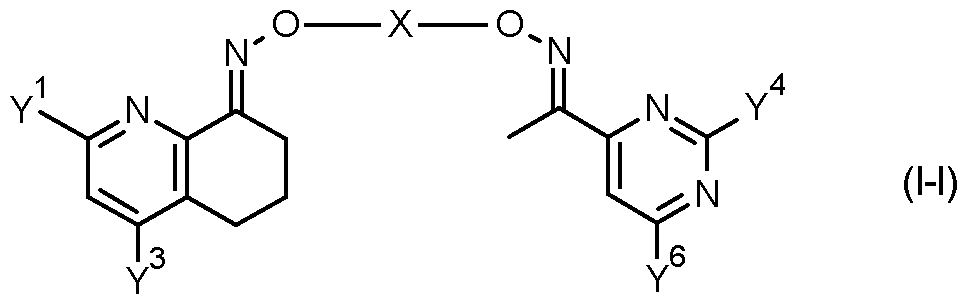
- p is 1 and -G ⁇ G ⁇ G 3 - represent -CH 2 -CH 2 -CH 2 -.
- a and Y 7 together represent -CH 2 -CH 2 -CH 2 -.
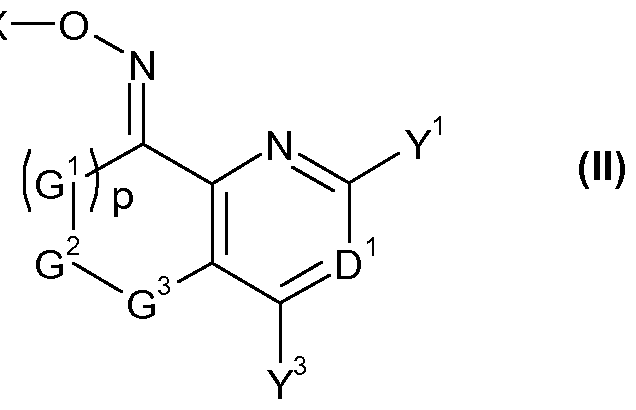
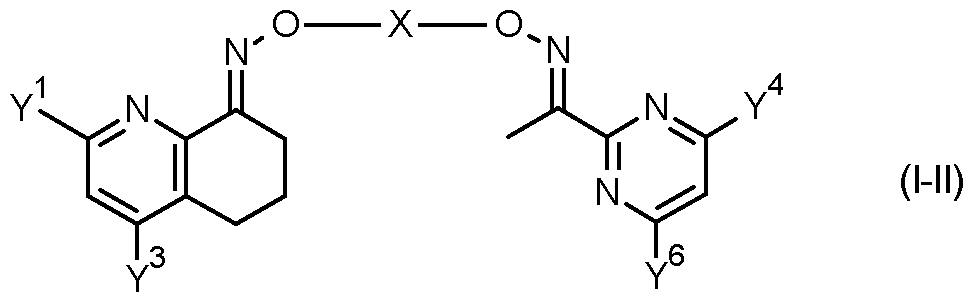
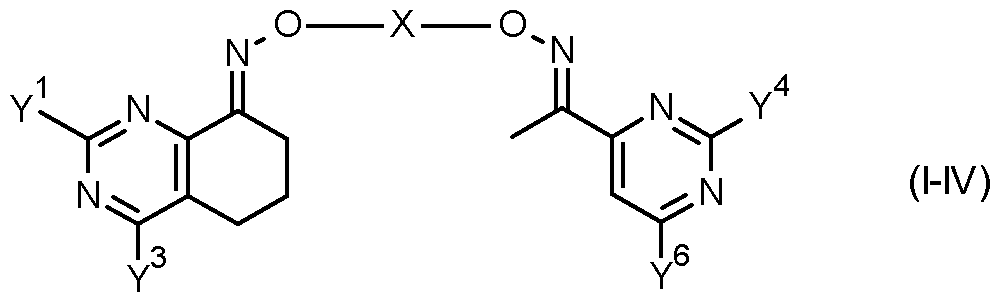
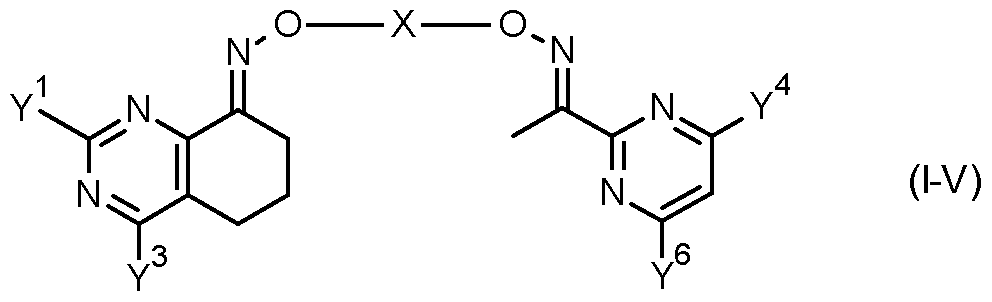
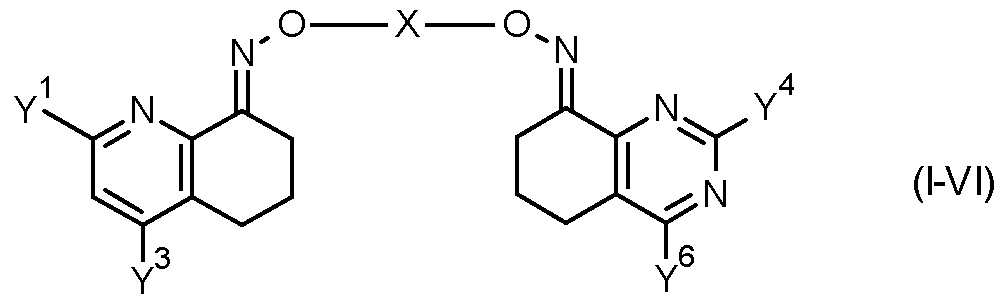
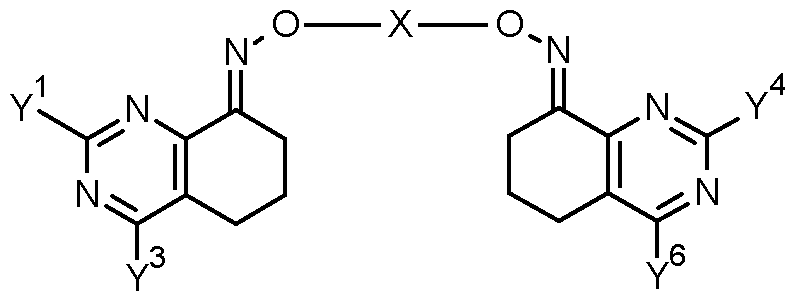
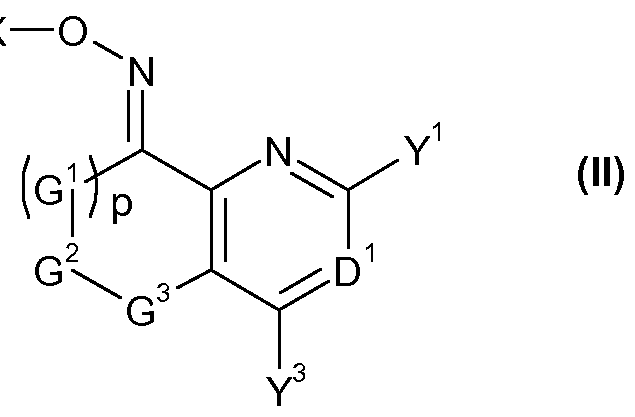
- the invention provides a compound of formula (II) FT -X
- R represents -ONH 2 , halogen, -0-S0 2 -R 2B or one of the groups LG, C-l, C-2A or C-2B:
- R 28 represents C1-C4 alkyl, C1-C4 haloalkyi or phenyl, wherein the phenyl is optionally substituted by one or two substituents independently selected from methyl, trihalomethyl, N0 2 , CN, C1-C7 alkoxycarbonyl;
- X" represents halogen
- R 37 represents either A as defined herein for compounds of formula (I) or the group R '
- R 27 represents -ONH 2 , -0-S0 2 -R 28 or one of the groups LG, C-l or C-2.
- R 27 represents -ONH 2 , tosylate, mesylate, triflate or one of the groups LG, C-l or C-2.
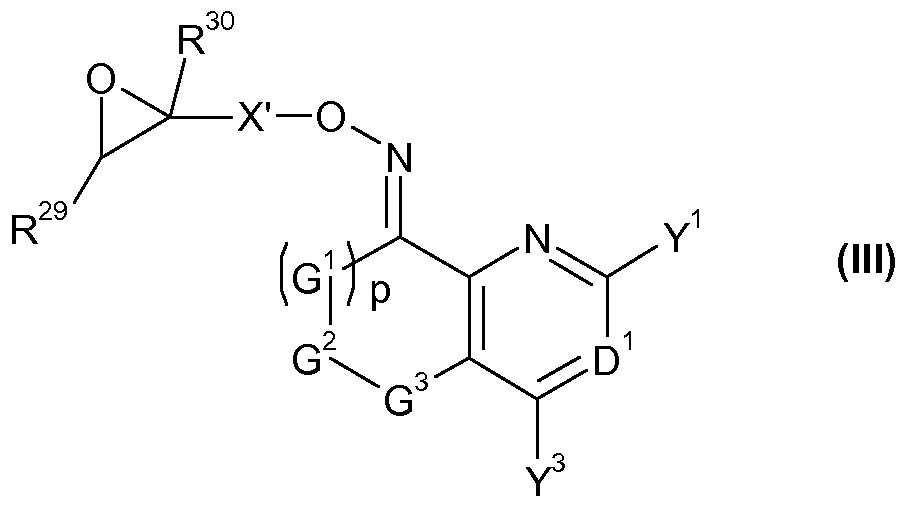
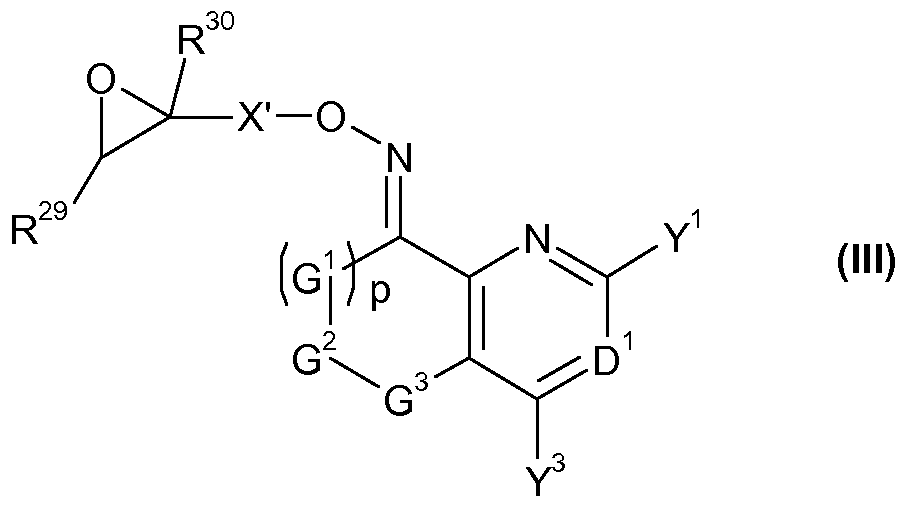
- X' represents one of the groupings X'-l, X'-2 or X'-3:
- R 29 and R 30 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci-C 4 haloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkyoxy; and
- G 1 , G 2 , G 3 , D 1 , Y 1 , Y 3 and p are as defined for a compound of formula (I);
- R 29 and R 30 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci-C 4 haloalkyi, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and
- At least one of Y 4 , Y 5 or Y 6 represents pyrimidinyl, Ci-Cs haloalkylthio, Ci-Cs haloalkylsulphinyl or Ci-Cs haloalkylsulphonyl ,wherein the pyrimidinyl is optionally substituted by one or more groups independently selected from halogen, CN, NH 2 , N0 2 , OH, C C 4 alkyl, C C 4 haloalkyi, C C 4 alkoxy, d-C 4 haloalkoxy, d-C 4 alkylthio, Ci-C 4 alkylsulphinyl and Ci-C 4 alkylsulphonyl.
- G 3 represents N(R 14 ) wherein R 14 represents
- X' represents X'-l.
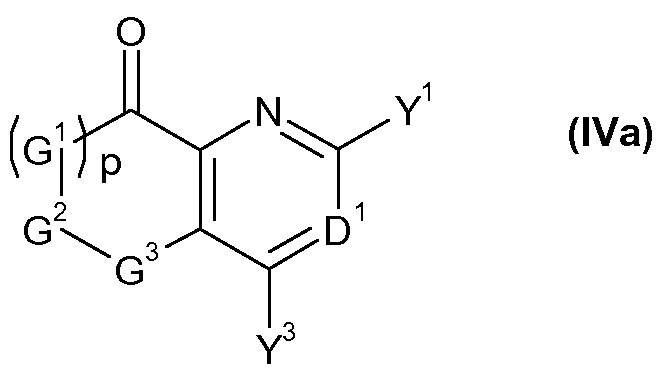
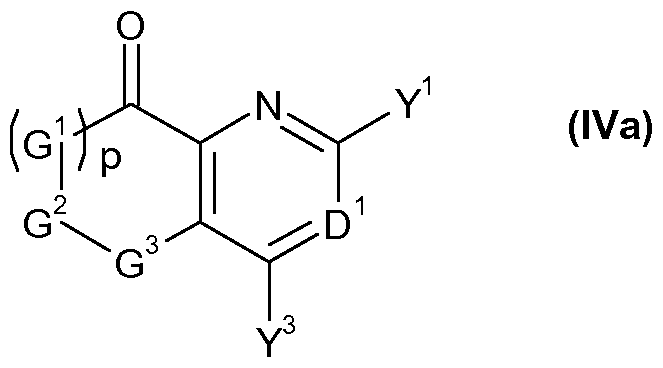
- G 1 , G 2 , G 3 , p, D 1 , Y 1 and Y 3 are as defined for a compound of formula I, or a salt or N-oxide thereof.
- the preferred definitions of G 1 , G 2 , G 3 , p, D 1 , Y 1 and Y 3 are as defined in respect of compounds of formula (I) above also apply to compounds of formula (IVa).
- Y 6 is Ci-C 6 alkyl, halogen, NH 2 , CrC 6 haloalkyl, Ci-Cs haloalkoxy, or Ci-C 6 haloalkoxy;
- Y 7 and Y 8 independently of one another represent hydrogen, halogen, CN, N0 2 , CrC 8 alkyl, Ci-C 4 -alkoxy-Ci-C 4 -alkyl, Ci-C 4 -alkoxy-Ci-C 4 -alkoxy-Ci-C 4 -alkyl, C 3 -C 8 cycloalkyi, C 2 -C 8 alkenyl, C 2 - C 8 alkynyl, phenyl, pyridyl, COR 13 , OR 22 , SH, Ci-C 8 alkylthio, Ci-C 8 alkylsulphinyl, Ci-C 8 alkylsulphonyl, N(R 23 ) 2 , C0 2 R 22 , 0(CO)R 13 , CON(R 23 ) 2 , NR 23 COR 13 or CR 13 N-OR 22 , wherein the alkyl, alkoxy, cycloalkyi, alkenyl,
- G 1 and G 2 independently represent -C(R 24 )(R 25 )-;
- G 3 represents -C(R 24 )(R 25 )-, O, N(R 26 ) or S;
- each R 24 and R 25 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci- C 4 haloalkyl, Ci-C 4 alkoxy, or d-C 4 haloalkyl;
- R 26 is hydrogen, OH, d-C 4 alkyl or Ci-C 4 alkoxy; p is 0, 1 or 2. Preferably, p is 1 or 2.
- Y 6 is hydrogen, methyl or halomethyl.
- Y 6 is methyl or halomethyl.
- the invention provides a compound of formula XVI.
- G 1 , G 2 , G 3 , p, D 1 , Y 1 and Y 3 are as defined herein for a compound of formula I, and wherein R 38 is hydrogen, CO-CH 3 , CO-CH 2 CH 3 , CO-CH 2 CH 2 CH 3 , CO-CH(CH 3 ) 2 , CO-CF 3 , CO- CF 2 CF 3 , -CH 3 , -CH 2 CH 3 , -CH 2 CH 2 CH 3 , -CH(CH 3 ) 2 , or a salt or N-oxide thereof.
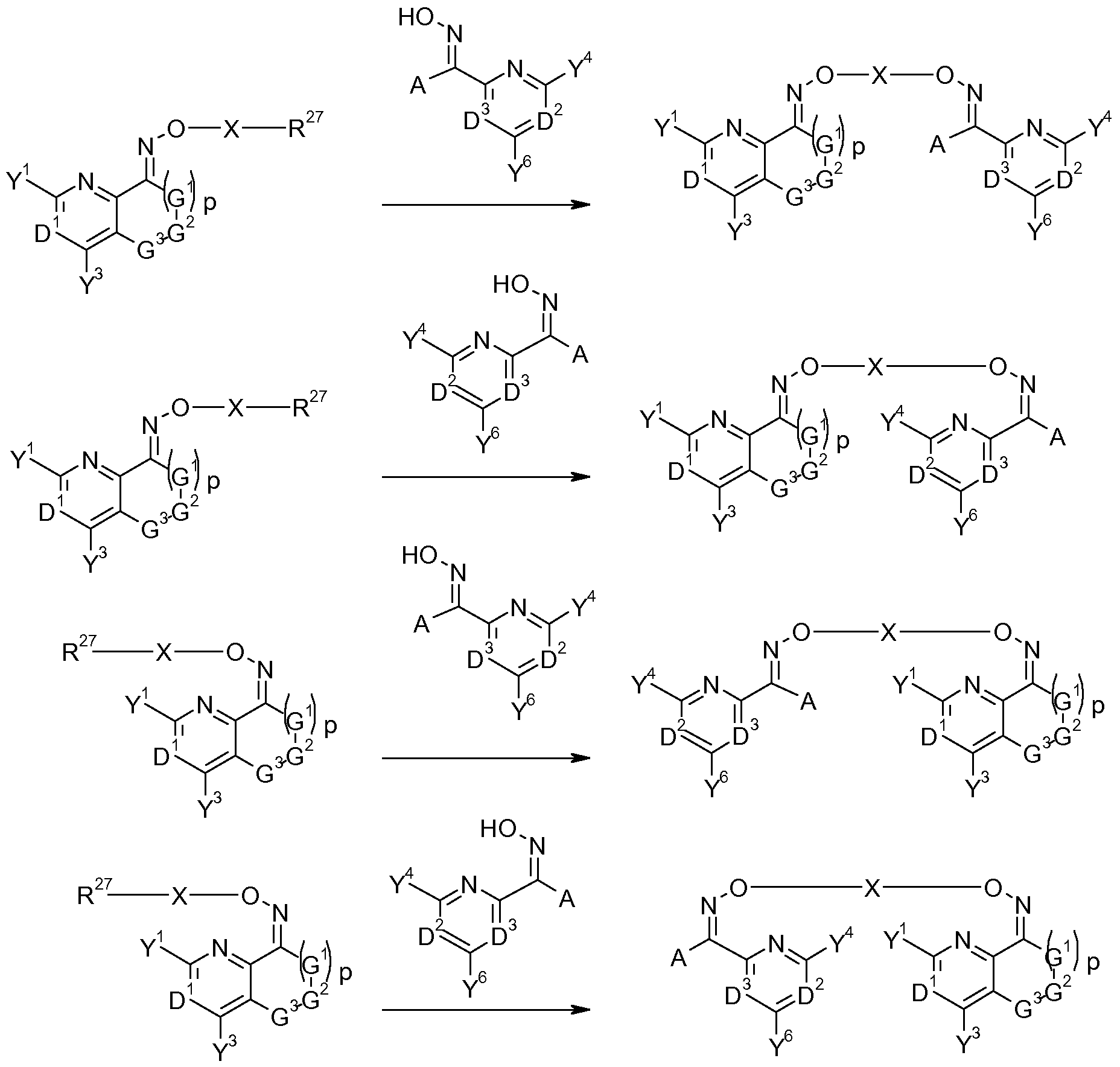
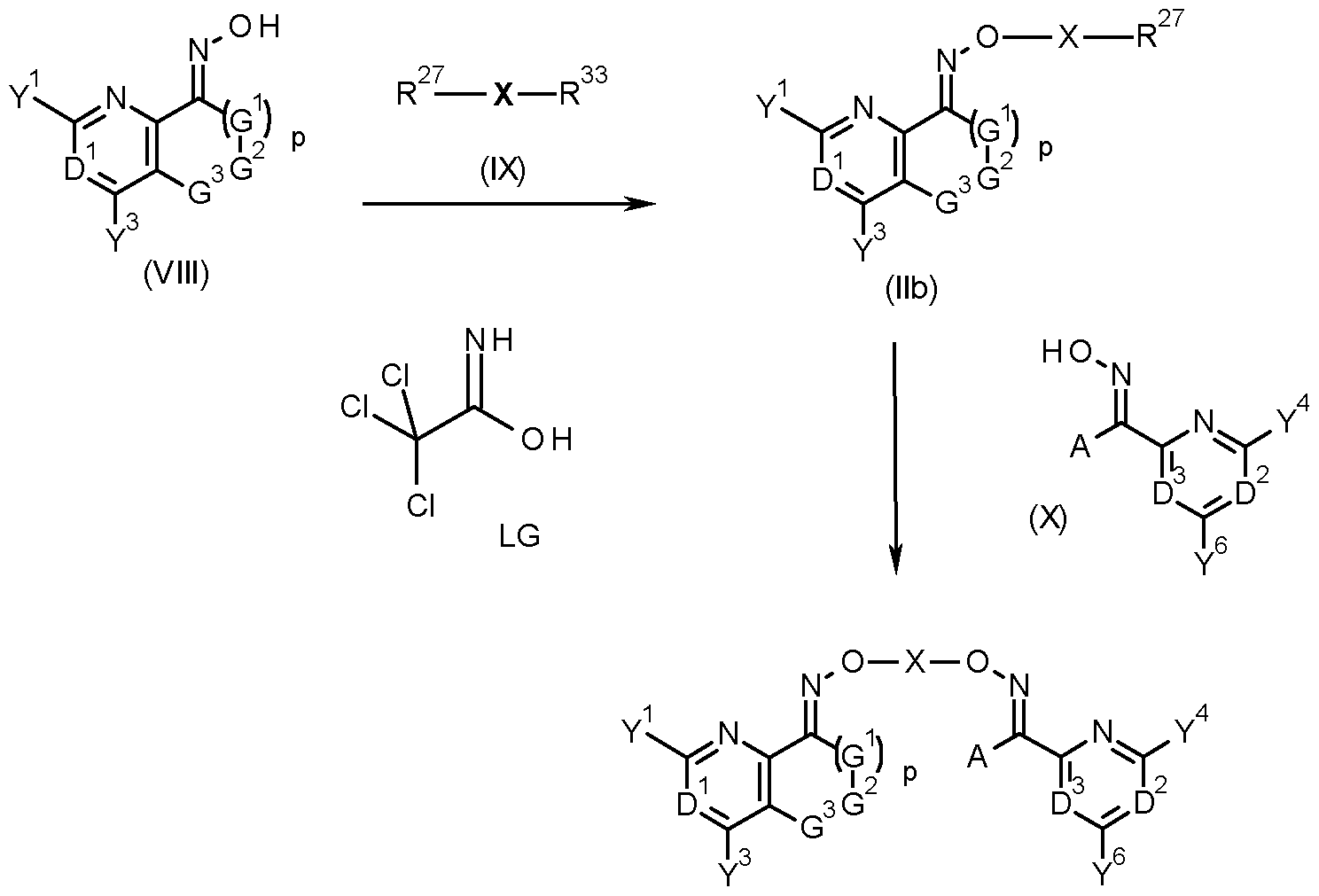
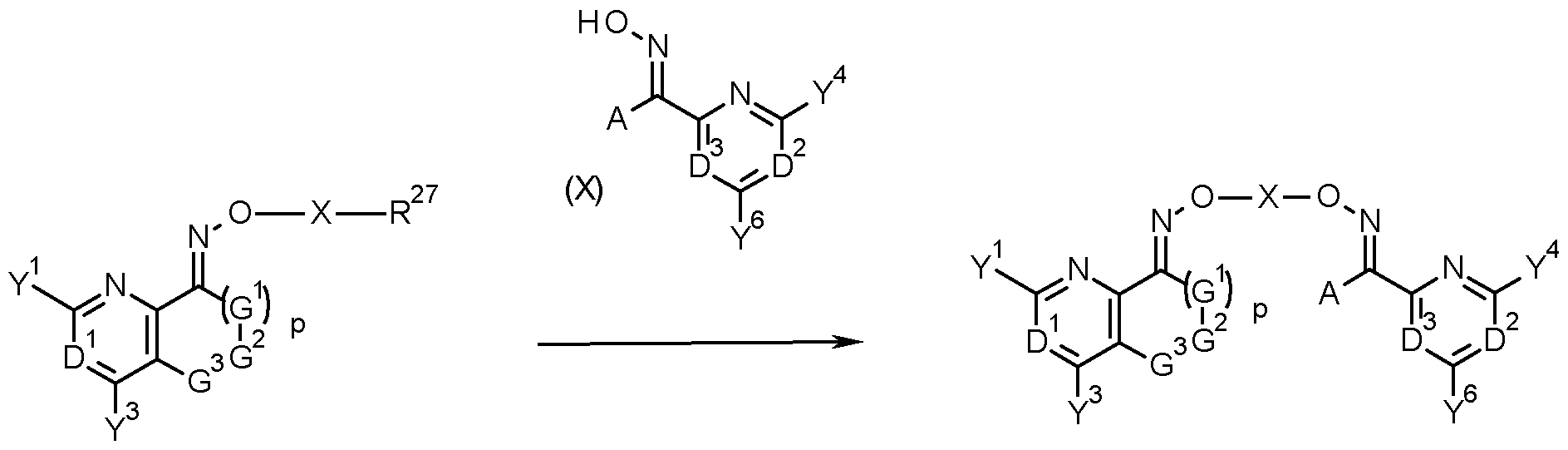
- the invention provides a process for the production of a compound of formula (I) comprising reacting a compound of formula (lib) with a compound of formula (X) as shown in scheme X
- R represents Ci-C 4 alkyl, d-C 4 haloalkyl or phenyl, wherein the phenyl is optionally substituted by one or two substituents independently selected from methyl, trihalomethyl, N0 2 , CN, C1-C7 alkoxycarbonyl; and
- the compounds of formula (I) may exist as different geometric or optical isomers or in different tautomeric forms. These may be separated and isolated by well-known (usually chromatographic) techniques, and all such isomers and tautomers and mixtures thereof in all proportions as well as isotopic forms, such as deuterated compounds, are part of the present invention.
- the carbon-nitrogen double bonds of the compound of formula (I) allow the four cis/trans isomers shown below:
- the present invention includes each of these isomers.
- the invention may provide a compound of formula (I) as just one of these isomers or as a mixture of one or more isomers in any ratio.
- the invention also includes the corresponding isomers of the intermediates described herein, e.g. compounds (II), (III) and (VIII).
- a reaction scheme depicts synthesis of one geometric isomer
- the scheme also includes synthesis of the other geometric isomers where possible.
- Scheme X shown above encompasses the reactions:
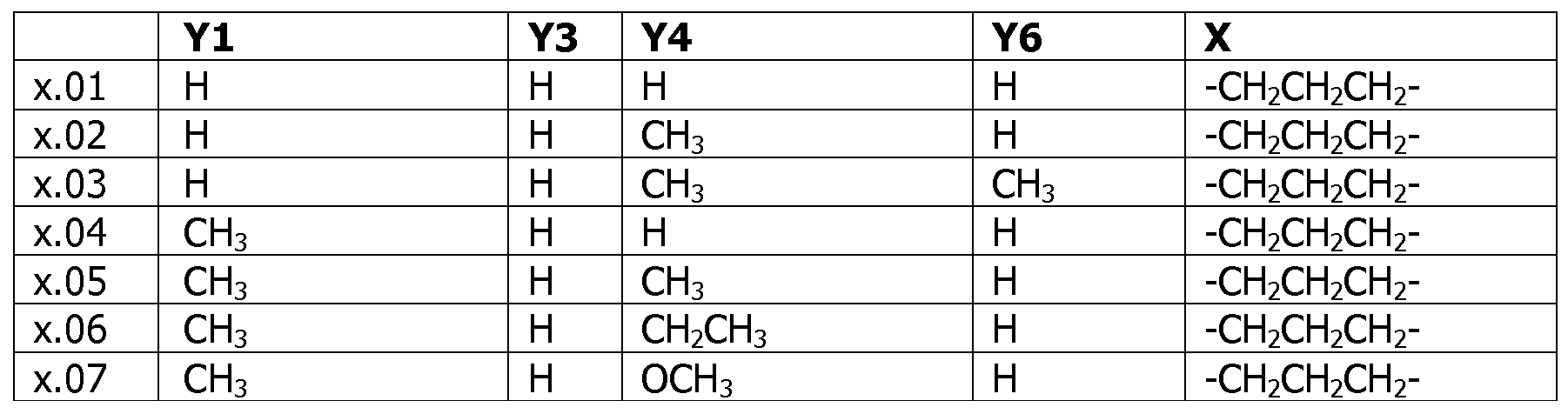
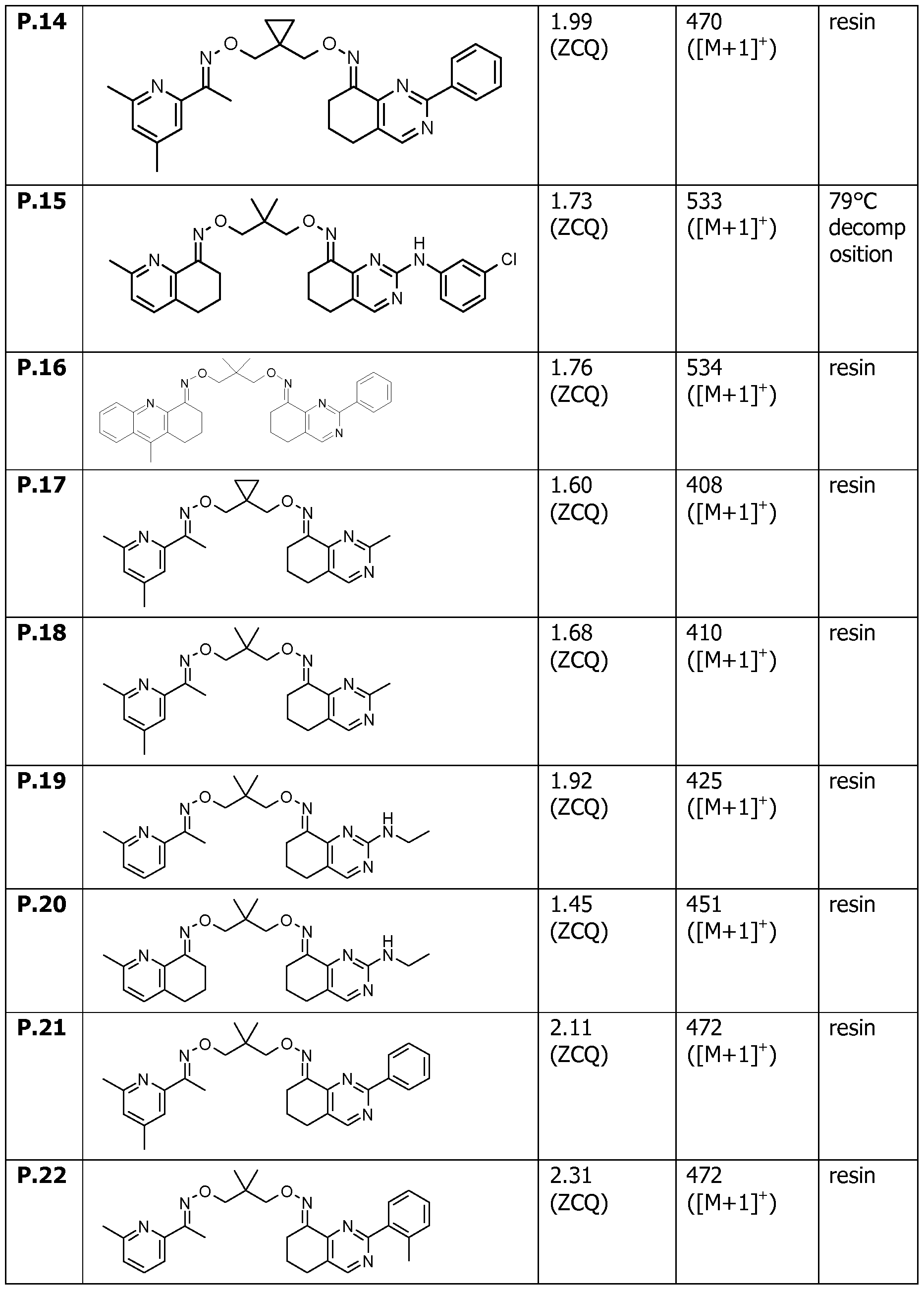
- Table X represents Table 1 (when X is 1), Table 2 (when X is 2), Table 3 (when X is 3), Table 4 (when X is 4), Table 5 (when X is 5), Table 6 (when X is 6), Table 7 (when X is 7).
- Y 1 , Y 3 , Y 4 , Y 6 and X have the specific meanings given in the table.
- Table 7 This table discloses com ounds 1.01 to 1.192 of the formula (I-VII)
- Y 1 , Y 3 , Y 4 , Y 6 and X have the specific meanings given in the table.
- the compounds in Tables 1 to 7 include all isomers, tautomers and mixtures thereof, including the cis/trans isomers shown above.
- the compounds of the invention may be made by a variety of methods, illustrated in schemes 1-8.
- the compounds depicted in the schemes also indicate any isomers and tautomers, in particular the geometric isomers arising from the oxime and oxime ether moieties.
- Compounds of formula (I) may be prepared by reacting a compound of formula (Ha), that is a compound of formula (II) wherein R is -ONH 2 , with a compound of formula (VI), wherein A, D 2 , D 3 , Y 4 and Y 6 are as defined herein for compounds of formula (I), and T 1 and T 2 are Ci-C 8 alkoxy, or T 1 and T 2 together with the carbon they are attached to form a carbonyl group or an acetal or ketal function of the form C(0-Ci-C 6 -alkylidene-0) whereby the alkylidene fragment may optionally be mono- to tetra-substituted by d-C 6 alkyl, as seen in scheme 1.
- Hydroxylamine derivatives of formula (Ila) may be made by reacting compounds of formula (IV), wherein G 1 , G 2 , G 3 , p, D 1 , Y 1 and Y 3 are as defined herein for compounds of formula (I), and T 1 and T 2 are Ci-Cs alkoxy, or T 1 and T 2 together with the carbon they are attached to form a carbonyl group or an acetal or ketal function of the form C(0-Ci-C 6 - alkylidene-O) whereby the alkylidene fragment may optionally be mono- to tetra -substituted by Ci-C 6 alkyl, with a bishydroxylamine derivative of formula (V), wherein X is as defined herein for a compound of formula (I) and R 31 and R 32 are either hydrogen or suitable protecting groups such as tert-butyloxycarbonyl (BOC), allyloxycarbonyl, fluorenylmethyloxycarbonyl (FMOC), formyl,
- R 31 and R 32 are hydrogen, in order to optimize the yield of compound (Ila) an excess of intermediate (V) over intermediate (IV) may preferably be used. If R 31 or R 32 is not hydrogen, the hydroxylamine derivative may be deprotected using techniques well known to the person skilled in the art. Examples can be found in Greene, T. W., Wuts, P. G. N., Protective Groups in Organic Synthesis, John Wiley & Sons, Inc, 2006.
- compounds of formula (I) may be prepared by reacting a compound of formula (IV) and a compound of formula (VI) in the presence of a compound of formula (VII), wherein X is as defined herein for compounds of formula (I).
- Compounds of formula (IV) and (VI) are described under Scheme 1.
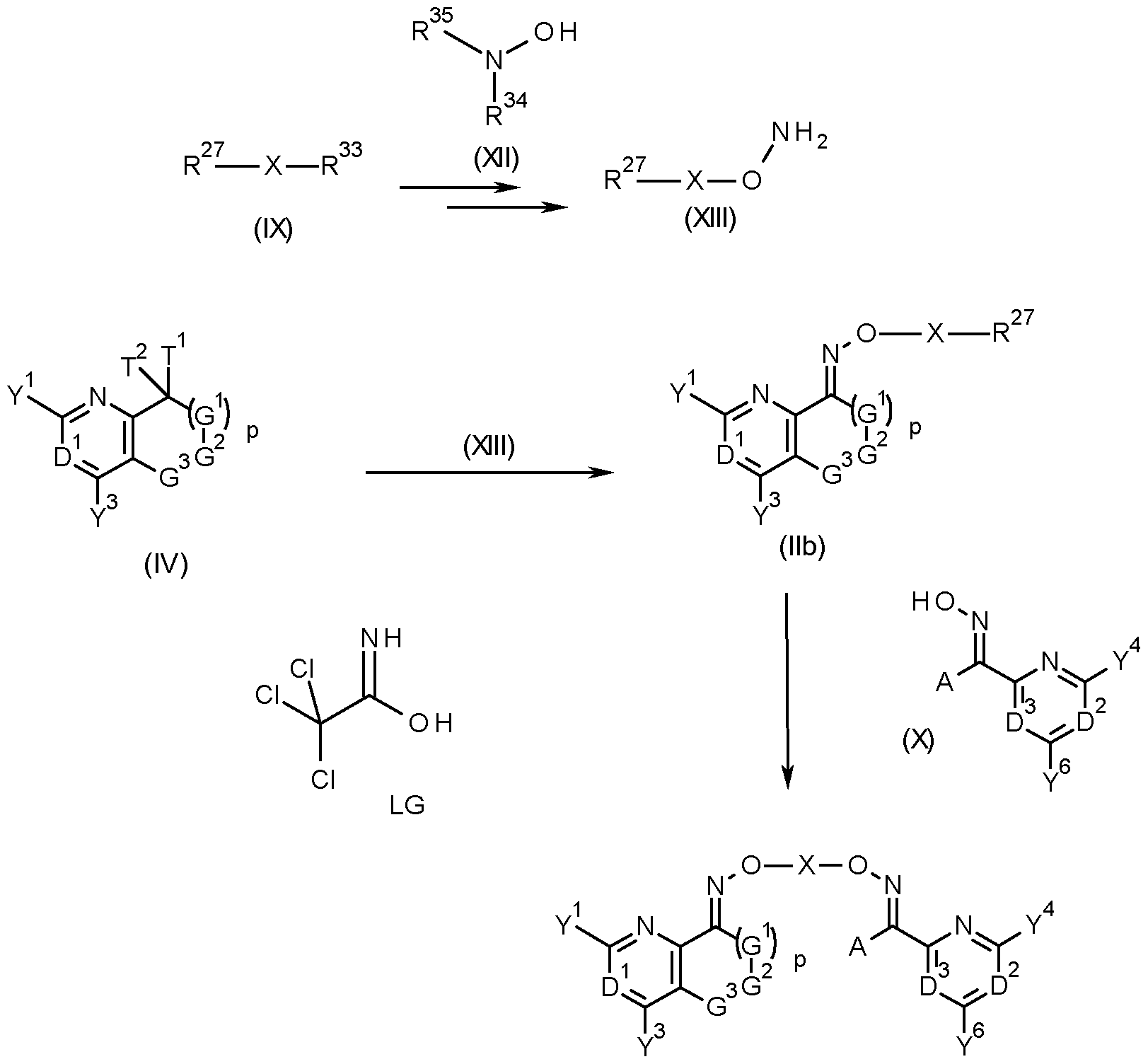
- compounds of formula (I) can be obtained by reacting a compound of formula (lib), that is a compound of formula (II) wherein R 27 is a halogen, in particular chlorine, bromine or iodine, or a sulfonic acid ester group, such as mesylate, tosylate, triflate, a phenylsulfonic acid ester, a nitro-phenylsulfonic acid ester, a nonafluorobutylsulfonic acid ester or LG with a compound of formula (X), wherein A, D 2 , D 3 , Y 4 and Y 6 are as defined herein for compounds of formula (I) (scheme 3).
- a compound of formula (lib) that is a compound of formula (II) wherein R 27 is a halogen, in particular chlorine, bromine or iodine, or a sulfonic acid ester group, such as mesylate, tosylate, triflate, a
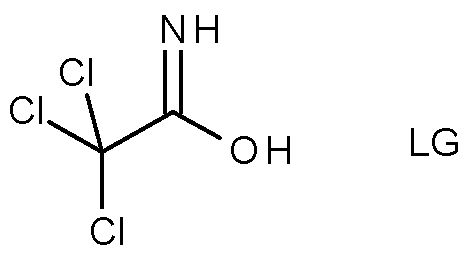
- Compounds of formula (lib) may be obtained by reacting an oxime of formula (VIII) with a compound of formula (IX), wherein R 27 is as defined herein for compounds of formula (lib) and R 33 is a halogen, in particular chlorine, bromine or iodine, a sulfonic acid ester group, or the group LG (scheme 3).
- R 27 and R 33 may be the same or different.
- R 33 is a better leaving group under the conditions of the reaction, such as tosylate or bromine when R is chlorine.
- an excess of the compound of formula (IX) relative to the oxime (VIII) would be used in the reaction, especially when R 27 and R 33 are the same.
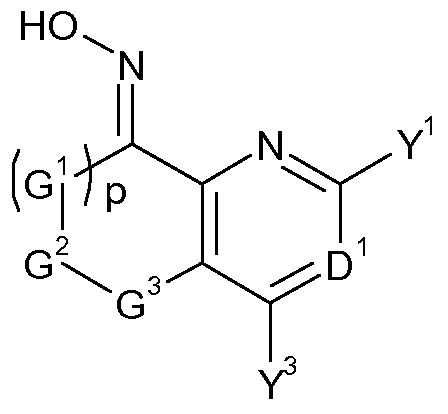
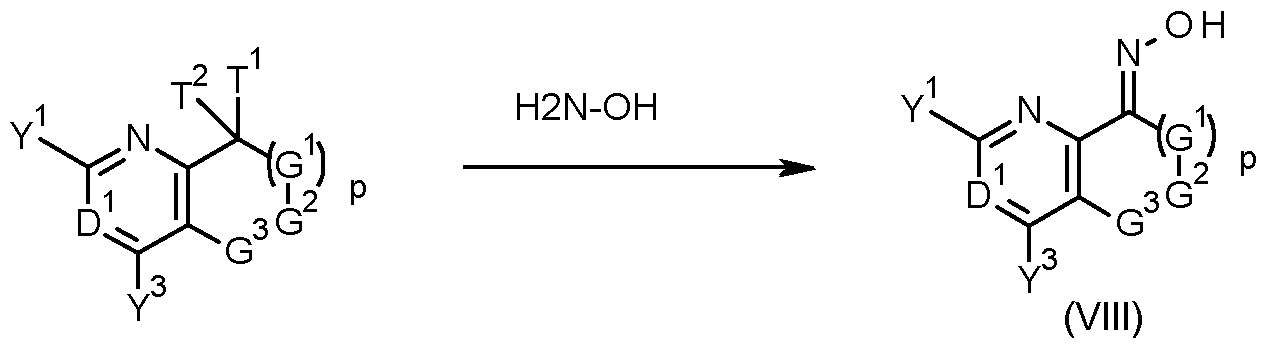
- Oximes of formula (VIII) may be obtained by a condensation reaction, whereby a compound of formula (IV), wherein G 1 , G 2 , G 3 , p, D 1 , Y 1 and Y 3 are as defined herein for compounds of formula (I) and T 1 and T 2 are Ci-C 8 alkoxy, or T 1 and T 2 together with the carbon they are attached to form a carbonyl group or an acetal or ketal function of the form C(0-Ci-C 6 - alkylidene-O) whereby the alkylidene fragment may optionally be mono- to tetra -substituted by Ci-C 6 -alkyl, is reacted with hydroxylamine, or, alternatively, with a salt of hydroxylamine.
- condensation processes is given below.
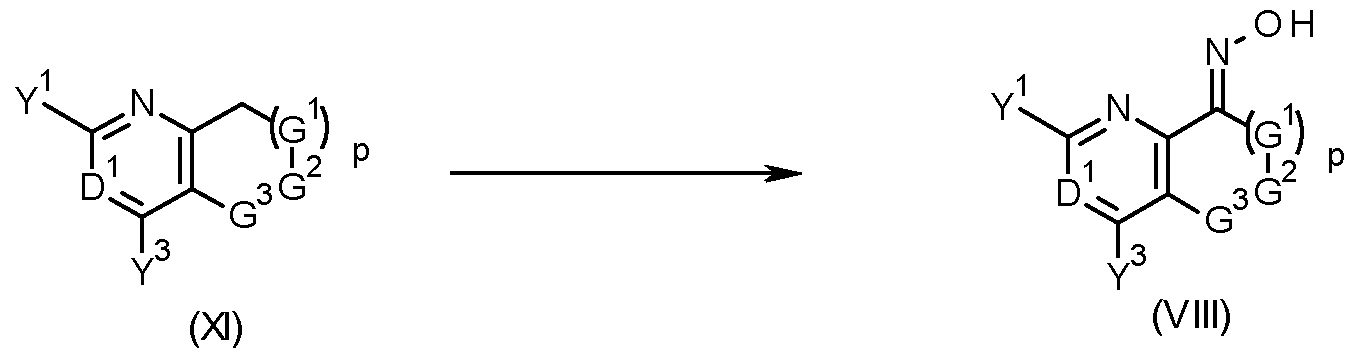
- oximes of formula (VIII) can be obtained by a nitrosation reaction of compounds of formula (XI), wherein G 1 , G 2 , G 3 , p, D 1 , Y 1 and Y 3 are as defined herein for compounds of formula (I), with base and an alkyl nitrite, as seen in scheme 5.
- Typical bases include lithium diisopropyl amide (LDA), lithium hexamethyldisilazane, n-butyl lithium, s-butyl lithium, tert-butyl lithium, sodium tert-butylate or potassium tert-butylate .
- Typical alkyl nitrites include isopentyl nitrite and tert-butyl nitrite.
- the compound of formula (XI), the alkyl nitrite or the base can be used in different stoichiometric amounts, with each reagent possibly being in excess with respect to the others.
- such reactions are carried out under nonaqueous conditions in an inert solvent such as hexane, heptanes, cyclohexane, toluene or ethers such as THF or tert-butyl methyl ether.
- the reaction may be performed at temperatures ranging from -80 to 250°C, preferably between -50 and 120°C.
- Such reactions can lead to a mixture of the E- and the Z-oxime (ether) product, or the product may also be exclusively either the E- or the Z-oxime (ether).
- the compounds of (lib) can be formed by reacting a hydroxylamine derivative of formula (XIII), wherein R 27 halogen, with a compound of formula (IV), as seen in scheme 6.
- Compounds of formula (XIII) can be made by alkylating a hydroxylamine derivative of formula (XII), wherein R 34 and R 35 , either independently of each other, or together with each other and the nitrogen atom to which they are attached, are protecting groups, such as tert- butoxy carbonyl, acetyl, benzyl, or phthalyl, with the alkylating agent (IX), wherein R is halogen and R 33 is halogen, in particular chloro, bromo or iodo, a sulfonic acid ester group, or LG (scheme 6). Typical conditions for such an alkylation reaction may be found below.
- the protecting groups or group can then be removed using techniques well known to a person skilled in the art, examples of which can be found in Greene, T. W., Wuts, P. G. N., Protective Groups in Organic Synthesis, John Wiley & Sons, Inc, 2006.
- Z 4 , Z 8 and Z 13 represent C(OH)R 30 and Z 5
- Z 9 and Z 14 represent CHR 29 wherein R 29 and R 30 independently of one another represent hydrogen, halogen, Ci-C 4 alkyl, Ci-C 4 haloalkyl, phenyl or CN, wherein phenyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, Ci-C 4 alkyl, Ci-C 4 haloalkyi, Ci-C 4 alkoxy and Ci-C 4 haloalkyoxy, can be made by reacting compounds of formula (III) with compounds of formula (X) (scheme 7).
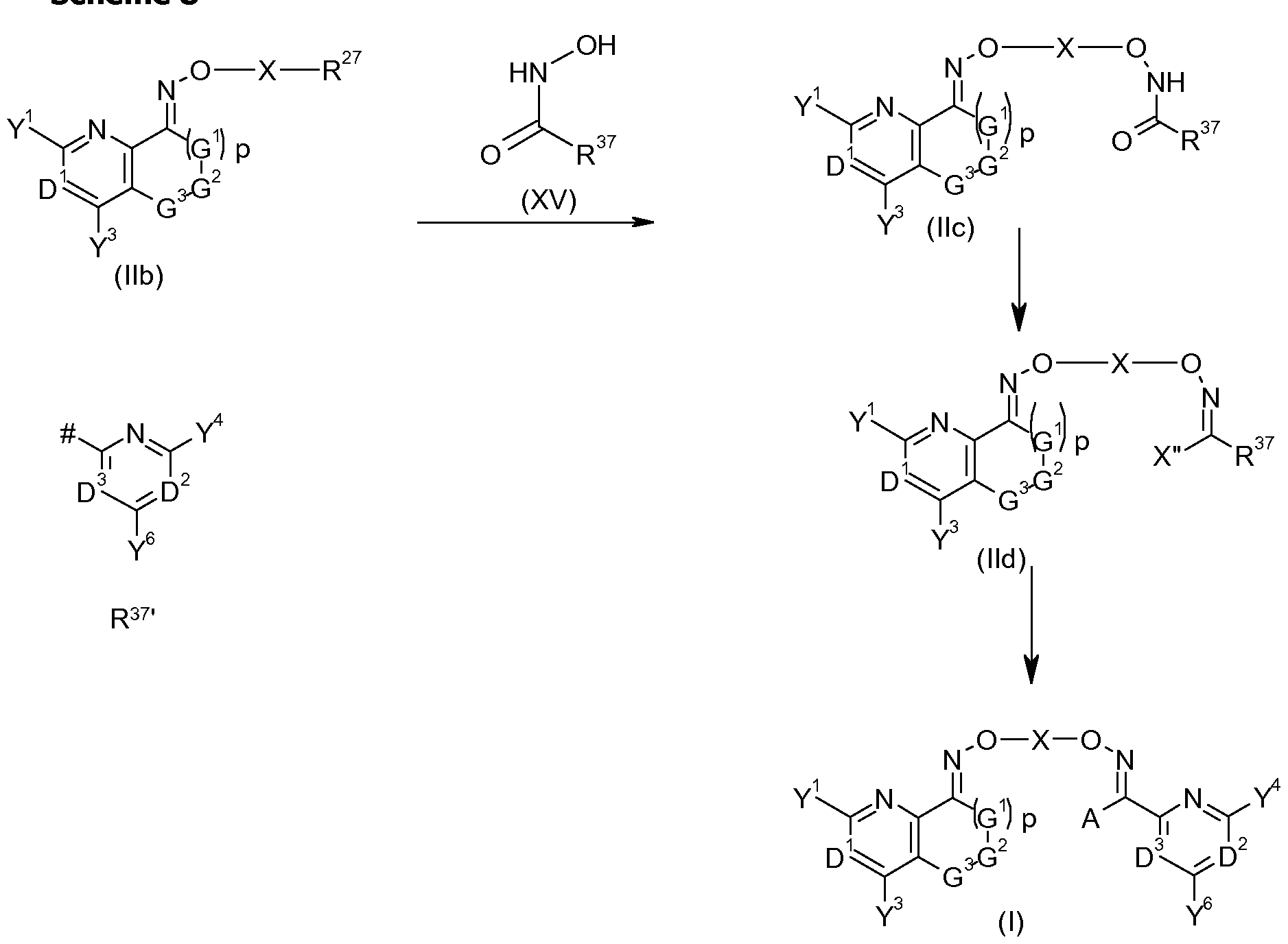
- Compounds of formula (I) may be formed from compounds of formula (lid), that is a compound of formula (II) wherein R 27 represents C-2B, as seen in scheme 8. This can be done by displacing the group X" with a suitable derivative of the group A or the fragment R 37 using one of several techniques well known to the person skilled in the art, including coupling reactions such as Suzuki (Suzuki-Miyaura) couplings and Stille couplings.
- the Suzuki coupling comprises the reaction between an organoboron compound, such as the boronic acid derivative of A or R 37 or their esters and a halide of formula (lid) to give compounds of formula (I).
- organoboron compound such as the boronic acid derivative of A or R 37 or their esters
- a halide of formula (lid) to give compounds of formula (I).
- the reaction may be done in the presence of a palladium catalyst such as Pd(PPh 3 ) 4 , Pd(OAc) 2 , Pd(dppf)CI 2 and a base such as Na 2 C0 3 , Ba(OH) 2 , K 3 P0 4 , Cs 2 C0 3 , K 2 C0 3 , KF, NaOH or alkali alcoholates, such as potassium tert-butoxide or sodium ethoxide.
- Typical catalyst loadings are in the range of 0.01 to 10 mol%.
- Preferred solvents for such cross coupling reactions include ethers such as THF or dimethoxyethane, acetonitrile, DMF, NMP, benzene or toluene or a mixture of such solvents. Such solvents can also be used together with water.
- the preferred temperature range for carrying out such reactions is between 0°C and 180°C.
- the Stille coupling comprises the use of an organotin compound, such as the tributylstannane derivative of A or R 37 and a halide of formula (lid) to give compounds of formula (I).
- the reaction can be done in the presence of a palladium catalyst such as Pd(PPh 3 ) 4 , Pd 2 (dba) 3 .CHCI 3 with or with an added ligand such as P(2-furyl) 3 or Pd(OAc) 2 , Pd(dppf)CI 2 ,
- a palladium catalyst such as Pd(PPh 3 ) 4 , Pd 2 (dba) 3 .CHCI 3 with or with an added ligand such as P(2-furyl) 3 or Pd(OAc) 2 , Pd(dppf)CI 2 ,
- Pd(MeCN) 2 CI 2 Typical catalyst loadings are in the range of 0.01 to 10 mol%.
- Preferred solvents for such cross coupling reactions include ethers such as THF or dimethoxyethane, acetonitrile, DMF, NMP, benzene or toluene. Such solvents can also be used together with water.
- the preferred temperature range for carrying out such reactions is between 0°C and 180°C.
- Compounds of formula (lid) can be formed from compounds of formula (lie), that is a compound of formula (II) wherein R 27 represents C-l, by the action of a halogenating agent.
- the reaction can be performed using an excess of either the halogenating agent, equimolar amounts of halogenating agent and the hydroxamic acid ester (XV), or with an excess of the hydroxamic acid ester (lie). Preferentially it is carried out with an excess of halogenating agent over the hydroxamic acid ester (lie).
- Typical halogenating agents include CCI 4 or CBr 4 along with and a phosphine such as triphenyl phosphine or tributylphosphine.
- Other typical halogenating agents include Et 2 NSF 3 , (MeOCH 2 CH 2 ) 2 NSF 3 (Deoxo-Fluor), morpholinotrifluorosulfurane and SF 4 , SOCI 2 , COCI 2 , PCI 5 , PCI 3 , PBr 3 or POCI 3 , or a mixture of PCI 5 and POCI 3 .
- Typical conditions include the use of a sub- stoichiometric, equimolar or excess amount of PCI 5 in POCI 3 relative to the compound of formula (lie), where POCI 3 itself may be present in an equimolar amount or alternatively, be used in a sub-stoichiometric amount or excess relative to the compound of formula (lie).
- halogenation of hydroxamic acid ester of formula (lie) can be done without a solvent in certain cases or, preferentially, in the presence of a solvent or mixture of solvents. Any organic solvent that is inert under the specific reaction conditions can be chosen.
- Preferred solvents include the following, without limiting the selection: aliphatic or aromatic hydrocarbons that may optionally be substituted by one or several halogen atoms such as pentane, hexanes, heptanes, cyclohexane, petroleum ether, benzene, toluene, xylene, chlorobenzene, dichlorobenzenes, dichloromethane, chloroform, 1,2-dichloroethane, carbon tetrachloride, ethers such as diethylether, diisopropyl ether, dibutyl ether, tert-butyl methyl ether, tetrahydrofuran, 1,4- dioxane, dimethoxyethane, triethylene glycol dimethyl ether (methyltriglyme), or acetonitrile, propionitrile, benzonitrile or a substituted benzonitrile.
- halogen atoms such as pentane,
- catalysts to facilitate this type of reaction comprising the use of catalysts such as dimethylformamide, diethylformamide and formylpiperidine.
- reaction temperature can be varied over a wide range. Typical reaction temperatures vary between - 100°C and 250°C. Preferentially, the temperature range is between 0°C and 100°C. On some occasions, the reaction may be carried out under reflux.
- This transformation can also optionally be carried out under ultrasonication.
- Typical bases include the following without limiting the selction: Triethylamine, tripropylamine, tributylamine, di-isopropyl-ethylamine, ⁇ , ⁇ -dimethyl-cyclohexylamine, N-methyl- dicyclohexylamine, ⁇ , ⁇ -dimethyl-aniline, N,N-d iethyl-aniline, ⁇ , ⁇ -dimethyl-benzylamine, N,N- diethyl-benzylamine, pyridine, 2-methyl-pyridine, 3-methyl-pyridine, 4-methyl-pyridine, 2,6- dimethyl-pyridine, 2,4,6-trimethyl-pyridine, 4-dimethylamino-pyridine, N-methyl-piperidine, N- ethy
- Compounds of formula (lie) can be formed by reacting compounds of formula (lib) with a hydroxamic acid derivative of formula (XV) wherein R 37 is as defined herein for compounds of formula (lid). This transformation is an alkylation of the hydroxamic acid derivative (XV) or its salt generated in situ by the action of a base.
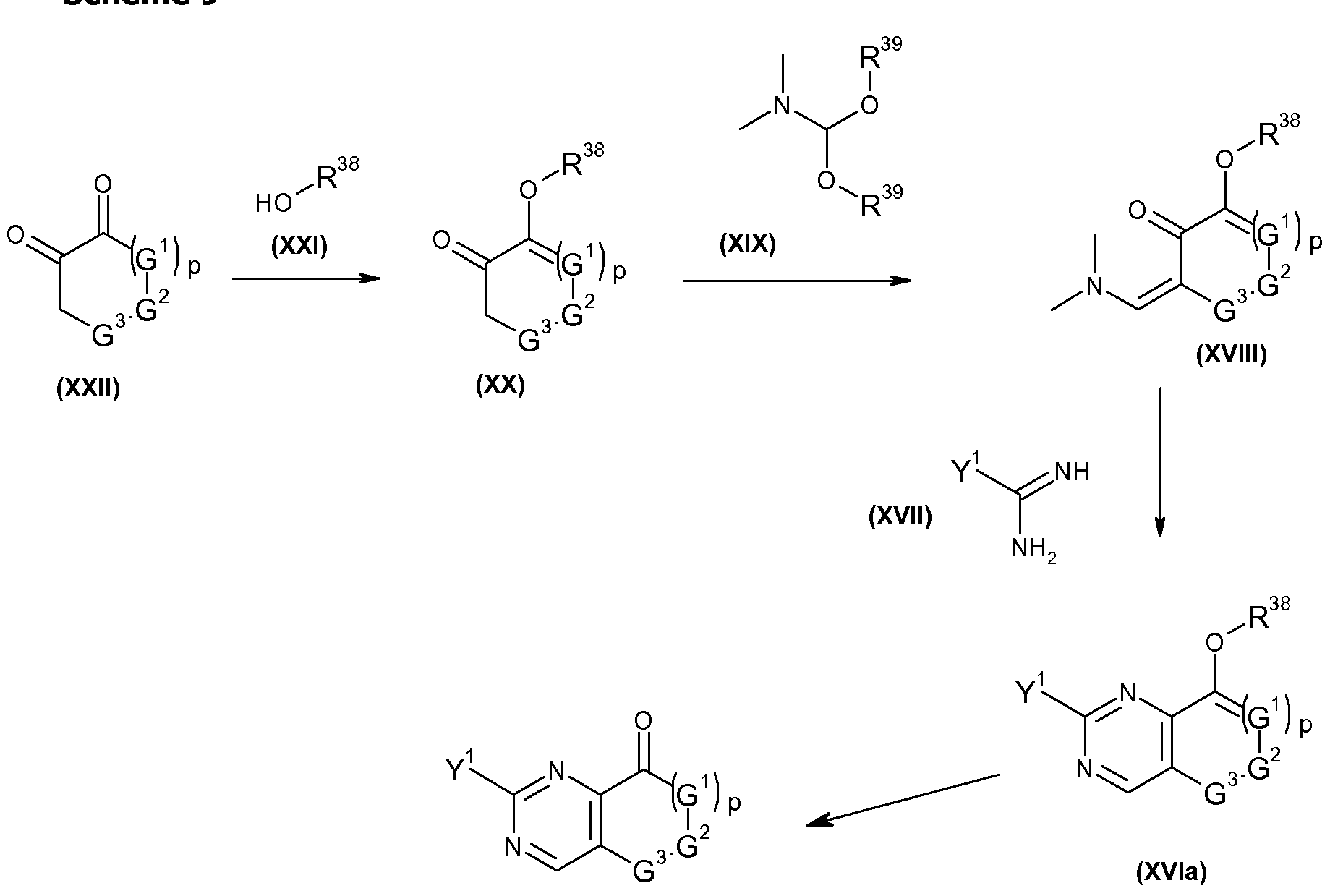
- Ketones of formula (IVa 7 ), that is a compound of formula (IVa) wherein D 1 is N may be obtained by acidic hydrolysis methods, well known to the person skilled in the art from compounds of formula (XVIa), that is a compound of formula (XVI) wherein D 1 is N.
- Enolethers of formula (XVIa) can be obtained by reacting enaminones of formula (XVIII) wherein G 1 , G 2 , G 3 and p are as described herein for compounds of formula (I) and R 38 represents hydrogen, CO-CH 3 , CO-CH 2 CH 3 , CO-CH 2 CH 2 CH 3 , CO-CH(CH 3 ) 2 , CO-CF 3 , CO-CF 2 CF 3 , CH 3 , CH 2 CH 3 , CH 2 CH 2 CH 3 or CH(CH 3 ) 2 with amidines or guanidines of formula (XVII) wherein Y 1 is as described herein for compounds of formula (I).
- condensation reactions can be performed using a number of conditions well known to the person skilled in the art. Such reactions are carried out in the presence of a base preferentially sodium or potassium alcoholate in ethanol or methanol. Specific reaction conditions may be found in Synthesis, 1012-1018, 1996; J. Heterocycl. Chem. 20, 649-653, 1983 or in WO2009010488.
- Enaminones of formula (XVIII) can be obtained by reacting ketone of formula (XX) wherein G 1 , G 2 , G 3 and p are as described herein for compounds of formula (I) and R 38 represents hydrogen, CO-CH 3 , CO-CH 2 CH 3 , CO-CH 2 CH 2 CH 3 , CO-CH(CH 3 ) 2 , CO-CF 3 , CO-CF 2 CF 3 , CH 3 , CH 2 CH 3 , CH 2 CH 2 CH 3 or CH(CH 3 ) 2 with ⁇ , ⁇ -dimethylformamide dialkyl acetal of formula (XIX) wherein R 39 represents d-C 4 alkyl.
- condensation reactions are well known to the person skilled in the art.
- the reaction is carried out in an inert solvent or without solvent at temperatures ranging from 0°C to 250°C preferably between 50 and 160°C.
- Specific reaction conditions may be found in Synthesis, (97), 3397, 1964; Synthesis, (97), 3407, 1964; Journal of Med. Chem., Vol. 52, No. 16, 5152-5163; Tetrahedron Lett, (27), 2567, 1986; Tetrahedron Lett, (50), 2255-2264, 1994; Synthetic Commun., (28), 10, 1743-1753, 1998 or in Chem. Ber., (104), 2975, 1971.
- Enol ethers of formula (XX) can be obtained by reacting ketones of formula (XXII) wherein G 1 , G 2 , G 3 and p are as described herein for compounds of formula (I) with an alcohol of formula (XXI) wherein R 38 represents hydrogen, CO-CH 3 , CO-CH 2 CH 3 , CO-CH 2 CH 2 CH 3 , CO- CH(CH 3 ) 2 , CO-CF 3 , CO-CF 2 CF 3 , CH 3 , CH 2 CH 3 , CH 2 CH 2 CH 3 or CH(CH 3 ) 2 .
- Such reactions can be affected using a number of conditions well known to the person skilled in the art. Specific reaction conditions may be found in WO2004104007.
- the reaction may be performed in the presence or absence of an inert organic or inorganic solvent, or in the presence of a mixture of such solvents. Preferentially, it is performed in the presence of one or more solvents.
- Preferred solvents include the following aliphatic or aromatic hydrocarbons, which may optionally be substituted by one or more halogen atoms, such as pentane, hexanes, heptanes, cyclohexane, petroleum ether, benzene, toluene, xylene, chlorobenzene, dichlorobenzenes, dichloromethane, chloroform, 1,2-dichloroethane or carbon tetrachloride, ethers such as diethylether, diisopropyl ether, tert-butyl methyl ether,
- ketones such as acetone, methyl ethyl ketone, methyl isopropyl ketone or methyl isobutyl ketone
- acids and ester such as acetic acid, ethyl acetate or methyl acetate
- aprotic polar solvents such as acetonitrile, pripionitril, dimethyl formamide, dimethyl acetamide, N-methyl-pyrrolidone, dimethyl sulfoxide, sulfolane, DMPU, or pyridine and picolines.
- solvents includes water and alcohols such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol, tert- butanol, pentanol, isopentanol, hexanol, trifluorethanol, ethylene glycol or methoxyethanol.
- alcohols such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol, tert- butanol, pentanol, isopentanol, hexanol, trifluorethanol, ethylene glycol or methoxyethanol.
- the reaction may be performed between -20°C and 250°C, preferentially between 0°C and
- reaction mixture may be heated to reflux.
- compounds can be used in the form of the free compound, or, alternatively, they can be used in the form of a salt such as the acetate, trifluoroacetate, propionate, benzoate, oxalate, methylsolfonate, phenylsulfonate, p-tolylsulfonate,
- the reaction can be carried out in the absence of an acid using the free compounds.
- reaction may be performed in the presence of an acid in catalytic,
- Acids that could be used include acetic acid, propionic acid, oxalic acid, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, hydroiodic acid, methansulfonic acid, para-toluenesulfonic acid, sulphuric acid, sodium hydrogensulphate and phosphoric acid.
- the reaction can optionally be carried out in a water-free solvent system in the presence of a drying agent, such as sodium or magnesium sulphate, potassium carbonate or molecular sieves.
- the condensation reaction can lead to a mixture of the E- and the Z- oxime (ether) product.
- the condensation product may also be exclusively either the E- or the Z- oxime (ether).
- Condensations can be performed under reduced pressure, normal pressure or increased pressure. Preferentially the reaction is performed under normal pressure.
- the reaction may be performed in the absence or presence of a solvent or a mixture of solvents.
- Preferential solvents include the following aliphatic or aromatic hydrocarbons that may optionally be substituted by one or more halogen atoms such as pentane, hexanes, heptanes, cyclohexane, petroleum ether, benzene, toluene, xylene, chlorobenzene, dichlorobenzenes, dichloromethane, chloroform, 1,2-dichloroethanev or carbon tetrachloride, ethers such as diethyl ether, diisopropyl ether, tert-butyl-methyl ether, tetrahydrofuran, 1,4-dioxane, dimethoxyethane or diglycol dimethyl ether, ketones such as acetone, methyl ethyl ketone, methyl isopropyl ketone or methyl isobutyl
- the selction of solvents includes also water and alcohols such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol, tert-butanol, pentanol, isopentanol, hexanol, trifluorethanol, ethylene glycol or methoxyethanol.
- alcohols such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol, tert-butanol, pentanol, isopentanol, hexanol, trifluorethanol, ethylene glycol or methoxyethanol.
- the reaction may be performed in a biphasic system comprising an organic solvent that is not miscible with water, such as toluene, dichloromethane, dichloro-ethylene, and an aqueous solvent, such as water.
- a phase-transfer catalyst such as tetra-n-butylammonium bromide (TBAB), Tetradecyldimethylbenzylammonium chloride (TDMBAC), N-Benzyltrimethylammonium hydroxide, along with aqueous sodium or potassium hydroxide in stoichiometric amounts.
- TBAB tetra-n-butylammonium bromide
- TDMBAC Tetradecyldimethylbenzylammonium chloride
- N-Benzyltrimethylammonium hydroxide N-Benzyltrimethylammonium hydroxide
- the reaction may be carried out at temperatures varying from -100°C and 250°C.
- the temperature range is between 0°C and 100°C.
- an organic or inorganic base may be present such as alkali- and earth alkali acetates, amides, carbonates, hydrogencarbonates, hydrides, hydroxides or alcoholates such as sodium, potassium, caesium or calcium acetate, sodium, potassium, caesium or calcium carbonate, sodium, potassium, caesium or calcium hydrogencarbonate, sodium, potassium, caesium or calcium hydride, sodium, potassium, caesium or calcium amide, sodium, potassium, caesium or calcium hydroxide, sodium, potassium, caesium or calcium methanolate, sodium, potassium, caesium or calcium ethanolate, sodium, potassium, caesium or calcium n-, i-, s- or t- butanolate, triethylamine, tripropylamine, tributylamine, di-isopropyl-ethylamine, N,N-dimethyl- cyclohexylamine, N-methyl-dicyclohexyl
- the alkylation can be performed under reduced pressure, normal pressure or increased pressure. Preferentially the reaction is performed under normal pressure.
- steps 1) to 19) may be required to be purified using, for example, chromatography, crystallisation or other purification techniques well known to the person skilled in the art.
- the compounds of formula (I) to formula (XXII) and, where appropriate, the tautomers thereof, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form.
- the invention therefore also relates to a method of controlling or preventing infestation of useful plants by phytopathogenic microorganisms, wherein a compound of formula (I) is applied as active ingredient to the plants, to parts thereof or the locus thereof.
- the compounds of formula (I) according to the invention are distinguished by excellent activity at low rates of application, by being well tolerated by plants and by being environmentally safe. They have very useful curative, preventive and systemic properties and are used for protecting numerous useful plants.
- the compounds of formula (I) can be used to inhibit or destroy the diseases that occur on plants or parts of plants (fruit, blossoms, leaves, stems, tubers, roots) of different crops of useful plants, while at the same time protecting also those parts of the plants that grow later e.g. from phytopathogenic microorganisms.
- compounds of formula (I) as dressing agents for the treatment of plant propagation material, in particular of seeds (fruit, tubers, grains) and plant cuttings (e.g. rice), for the protection against fungal infections as well as against phytopathogenic fungi occurring in the soil.
- the compounds of formula (I) according to the invention may be used for controlling fungi in related areas, for example in the protection of technical materials, including wood and wood related technical products, in food storage or in hygiene management.
- the compounds of formula (I) are, for example, effective against the phytopathogenic fungi of the following classes: Fungi imperfecti (e.g. Botrytis, Pyricularia, Helminthosporium,
- Basidiomycetes e.g. Rhizoctonia, Hemileia, Puccinia. Additionally, they are also effective against the Ascomycetes classes (e.g. Venturia and Erysiphe, Podosphaera, Monilinia, Uncinula) and of the Oomycetes classes (e.g.
- useful plants to be protected typically comprise the following species of plants: cereal (wheat, barley, rye, oat, rice, maize, sorghum and related species); beet (sugar beet and fodder beet); pomes, drupes and soft fruit (apples, pears, plums, peaches, almonds, cherries, strawberries, raspberries and blackberries); leguminous plants (beans, lentils, peas, soybeans); oil plants (rape, mustard, poppy, olives, sunflowers, coconut, castor oil plants, cocoa beans, groundnuts); cucumber plants (pumpkins, cucumbers, melons); fibre plants (cotton, flax, hemp, jute); citrus fruit
- useful plants is to be understood as including also useful plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides (such as, for example, HPPD inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, EPSPS (5-enol-pyrovyl-shikimate-3-phosphate-synthase) inhibitors, GS (glutamine synthetase) inhibitors or PPO (protoporphyrinogen-oxidase) inhibitors) as a result of conventional methods of breeding or genetic engineering.
- herbicides like bromoxynil or classes of herbicides
- EPSPS (5-enol-pyrovyl-shikimate-3-phosphate-synthase) inhibitors
- GS glutamine synthetase
- PPO protoporphyrinogen-oxidase
- Crola Clearfield® summer rape
- crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names
- useful plants is to be understood as including also useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin- producing bacteria, especially those of the genus Bacillus.
- YieldGard® (maize variety that expresses a CrylA(b) toxin); YieldGard Rootworm® (maize variety that expresses a CrylllB(bl) toxin); YieldGard Plus® (maize variety that expresses a CrylA(b) and a CrylllB(bl) toxin); Starlink® (maize variety that expresses a Cry9(c) toxin); Herculex I® (maize variety that expresses a CryIF(a2) toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylA(c) toxin);
- Bollgard I® (cotton variety that expresses a CrylA(c) toxin); Bollgard II® (cotton variety that expresses a CrylA(c) and a CryllA(b) toxin); VIPCOT® (cotton variety that expresses a VIP toxin); NewLeaf® (potato variety that expresses a CrylllA toxin); NatureGard® Agrisure® GT Advantage (GA21 glyphosate-tolerant trait), Agrisure® CB Advantage (Btll corn borer (CB) trait), Agrisure® RW (corn rootworm trait) and Protecta®.
- useful plants is to be understood as including also useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so- called “pathogenesis-related proteins” (PRPs, see e.g. EP-A-0 392 225).
- PRPs pathogenesis-related proteins
- Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP-A-0 392 225, WO 95/33818, and EP-A-0 353 191.
- the methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
- locus of a useful plant as used herein is intended to embrace the place on which the useful plants are growing, where the plant propagation materials of the useful plants are sown or where the plant propagation materials of the useful plants will be placed into the soil.
- An example for such a locus is a field, on which crop plants are growing.
- plant propagation material is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes. There may be mentioned for example seeds (in the strict sense), roots, fruits, tubers, bulbs, rhizomes and parts of plants. Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion. Preferably "plant propagation material” is understood to denote seeds.
- the compounds of formula (I) can be used in unmodified form or, preferably, together with carriers and adjuvants conventionally employed in the art of formulation.
- the invention also relates to compositions for controlling and protecting against phytopathogenic microorganisms, comprising a compound of formula (I) and an inert carrier, and to a method of controlling or preventing infestation of useful plants by phytopathogenic microorganisms, wherein a composition, comprising a compound of formula (I) as acitve ingredient and an inert carrier, is applied to the plants, to parts thereof or the locus thereof.
- compounds of formula (I) and inert carriers are conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g. in polymeric substances.
- the methods of application such as spraying, atomising, dusting, scattering, coating or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances.
- the compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
- Suitable carriers and adjuvants can be solid or liquid and are substances useful in formulation technology, e.g. natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders or fertilizers. Such carriers are for example described in WO 97/33890.
- the compounds of formula (I) or compositions, comprising a compound of formula (I) as active ingredient and an inert carrier can be applied to the locus of the plant or plant to be treated, simultaneously or in succession with further compounds.
- further compounds can be e.g. fertilizers or micronutrient donors or other preparations which influence the growth of plants. They can also be selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
- a preferred method of applying a compound of formula (I), or a composition, comprising a compound of formula (I) as active ingredient and an inert carrier is foliar application.
- the frequency of application and the rate of application will depend on the risk of infestation by the corresponding pathogen.
- the compounds of formula (I) may also penetrate the plant through the roots via the soil (systemic action) by drenching the locus of the plant with a liquid formulation, or by applying the compounds in solid form to the soil, e.g. in granular form (soil application). In crops of water rice such granulates can be applied to the flooded rice field.
- the compounds of formula (I) may also be applied to seeds (coating) by impregnating the seeds or tubers either with a liquid formulation of the fungicide or coating them with a solid formulation.
- a formulation i.e. a composition comprising the compound of formula (I) and, if desired, a solid or liquid adjuvant, is prepared in a known manner, typically by intimately mixing and/or grinding the compound with extenders, for example solvents, solid carriers and, optionally, surface-active compounds (surfactants).
- extenders for example solvents, solid carriers and, optionally, surface-active compounds (surfactants).
- the agrochemical formulations will usually contain from 0.1 to 99% by weight, preferably from 0.1 to 95% by weight, of the compound of formula (I), 99.9 to 1% by weight, preferably 99.8 to 5% by weight, of a solid or liquid adjuvant, and from 0 to 25% by weight, preferably from 0.1 to 25% by weight, of a surfactant.
- Advantageous rates of application are normally from 5g to 2kg of active ingredient (a.i.) per hectare (ha), preferably from lOg to 1kg a.i. /ha, most preferably from 20g to 600g a.i./ha.
- convenient rates of application are from lOmg to lg of active substance per kg of seeds.
- the rate of application for the desired action can be determined by experiments. It depends for example on the type of action, the developmental stage of the useful plant, and on the application (location, timing, application method) and can, owing to these parameters, vary within wide limits.
- the compounds of formula (I), or a pharmaceutical salt thereof, described above may also have an advantageous spectrum of activity for the treatment and/or prevention of microbial infection in an animal.
- Animal can be any animal, for example, insect, mammal, reptile, fish, amphibian, preferably mammal, most preferably human.
- Treatment means the use on an animal which has microbial infection in order to reduce or slow or stop the increase or spread of the infection, or to reduce the infection or to cure the infection.
- prevention means the use on an animal which has no apparent signs of microbial infection in order to prevent any future infection, or to reduce or slow the increase or spread of any future infection.
- a compound of formula (I) in the manufacture of a medicament for use in the treatment and/or prevention of microbial infection in an animal.
- a compound of formula (I) as a pharmaceutical agent.
- a compound of formula (I) as an antimicrobial agent in the treatment of an animal.
- a pharmaceutical composition comprising as an active ingredient a compound of formula (I), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable diluent or carrier. This composition can be used for the treatment and/or prevention of antimicrobial infection in an animal.
- This pharmaceutical composition can be in a form suitable for oral administration, such as tablet, lozenges, hard capsules, aqueous suspensions, oily suspensions, emulsions dispersible powders, dispersible granules, syrups and elixirs.
- this pharmaceutical composition can be in a form suitable for topical application, such as a spray, a cream or lotion.
- this pharmaceutical composition can be in a form suitable for parenteral administration, for example injection.
- parenteral administration for example injection.
- composition can be in inhalable form, such as an aerosol spray.
- the compounds of formula (I) may be effective against various microbial species able to cause a microbial infection in an animal.
- microbial species are those causing Aspergillosis such as Aspergillus fumigatus, A. flavus, A. terms, A. nidulans and A. niger, those causing Blastomycosis such as Blastomyces dermatitidis; those causing Candidiasis such as Candida albicans, C. glabrata, C. tropicalis, C. parapsilosis, C. krusei and C.
- Aspergillosis such as Aspergillus fumigatus, A. flavus, A. terms, A. nidulans and A. niger
- Blastomycosis such as Blastomyces dermatitidis
- Candidiasis such as Candida albicans, C. glabrata, C. tropicalis, C. parapsilosis, C.
- Microsporum Spp Trichophyton Spp, Epidermophyton Spp, Mucor Spp, Sporothorix Spp, Phialophora Spp, Cladosporium Spp, Petriellidium spp, Paracoccidioides Spp and Histoplasma Spp.
- compositions of this invention may contain other compounds having biological activity, for example micronutrients or compounds having fungicidal activity or which possess plant growth regulating, herbicidal, insecticidal, nematicidal or acaricidal activity.
- the compound of formula I (herein after abbreviated by the term "TX” thus means a compound encompassed by the compounds of formula I, or preferably the term “TX” refers to a compound selected from the Tables 1-7 or Table 9) may be the sole active ingredient of the composition or it may be admixed with one or more additional active ingredients such as a pesticide (insect, acarine, mollusc and nematode pesticide), fungicide, synergist, herbicide, safener or plant growth regulator where appropriate.
- a pesticide insect, acarine, mollusc and nematode pesticide
- fungicide fungicide
- synergist synergist
- herbicide herbicide
- safener plant growth regulator
- An additional active ingredient may: provide a composition having a broader spectrum of activity or increased persistence at a locus; provide a composition demonstrating better plant/crop tolerance by reducing phytotoxicity; provide a composition controlling insects in their different development stages; synergise the activity or complement the activity (for example by increasing the speed of effect or overcoming repellency) of the TX; or help to overcome or prevent the development of resistance to individual components.
- suitable pesticides include the following:
- a) Pyrethroids such as permethrin, cypermethrin, fenvalerate, esfenvalerate, deltamethrin, cyhalothrin (in particular lambda-cyhalothrin), bifenthrin, fenpropathrin, cyfluthrin, tefluthrin, fish safe pyrethroids (for example ethofenprox), natural pyrethrin, tetramethrin, s-bioallethrin, fenfluthrin, prallethrin or 5-benzyl-3-furylmethyl-(E)-(lR,3S)-2,2-dimethyl- 3-(2-oxothiolan-3-ylidenemethyl)cyclopropane carboxylate;
- Organophosphates such as, profenofos, sulprofos, acephate, methyl parathion, azinphos-methyl, demeton-s-methyl, heptenophos, thiometon, fenamiphos, monocrotophos, profenofos, triazophos, methamidophos, dimethoate, phosphamidon, malathion, chlorpyrifos, phosalone, terbufos, fensulfothion, fonofos, phorate, phoxim, pirimiphos-methyl,
- Carbamates including aryl carbamates
- pirimicarb triazamate
- cloethocarb carbofuran
- furathiocarb furathiocarb
- ethiofencarb aldicarb
- thiofurox carbosulfan
- bendiocarb fenobucarb
- propoxur methomyl or oxamyl
- Benzoyl ureas such as diflubenzuron, triflumuron, hexaflumuron, flufenoxuron or chlorfluazuron;
- Organic tin compounds such as cyhexatin, fenbutatin oxide or azocyclotin;
- Macrolides such as avermectins or milbemycins, for example abamectin, emamectin benzoate, ivermectin, milbemycin, or spinosad, spinetoram or azadirachtin;
- Organochlorine compounds such as endosulfan, benzene hexachloride, DDT, chlordane or dieldrin;
- Amidines such as chlordimeform or amitraz
- Fumigant agents such as chloropicrin, dichloropropane, methyl bromide or metam
- Neonicotinoid compounds such as imidacloprid, thiacloprid, acetamiprid, clothianidin, nitenpyram, dinotefuran or thiamethoxam;
- Diacyl hydrazines such as tebufenozide, chromafenozide or methoxyfenozide;
- Diphenyl ethers such as diofenolan or pyriproxifen
- pesticides having particular targets may be employed in the composition, if appropriate for the intended utility of the composition.
- selective insecticides for particular crops for example stemborer specific insecticides (such as cartap) or hopper specific insecticides (such as buprofezin) for use in rice may be employed.
- insecticides or acaricides specific for particular insect species/stages may also be included in the compositions (for example acaricidal ovo-larvicides, such as clofentezine, flubenzimine, hexythiazox or tetradifon; acaricidal motilicides, such as dicofol or propargite; acaricides, such as bromopropylate or chlorobenzilate; or growth regulators, such as hydramethylnon, cyromazine, methoprene, chlorfluazuron or diflubenzuron).
- acaricidal ovo-larvicides such as clofentezine, flubenzimine, hexythiazox or tetradifon
- acaricidal motilicides such as dicofol or propargite
- acaricides such as bromopropylate or chlorobenzilate
- growth regulators such
- TX refers to a compound covered by the compounds of formula I or preferably the term “TX” refers to a compound selected from the Tables 1 to 9 and the following List shows specific examples of mixtures comprising the component TX and the component (B):
- an adjuvant selected from the group of substances consisting of petroleum oils (alternative name) (628) + TX,
- an acaricide selected from the group of substances consisting of l,l-bis(4-chlorophenyl)-2- ethoxyethanol (IUPAC name) (910) + TX, 2,4-dichlorophenyl benzenesulfonate (IUPAC/Chemical Abstracts name) (1059) + TX, 2-fluoro-/V-methyl-/V-l-naphthylacetamide (IUPAC name) (1295) + TX, 4-chlorophenyl phenyl sulfone (IUPAC name) (981) + TX, abamectin (1) + TX, acequinocyl (3) + TX, acetoprole [CCN] + TX, acrinathrin (9) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, alpha-cypermethrin (202) + TX, amidithion (870) + TX, amidoflumet [CCN
- polychloroterpenes (traditional name) (1347) + TX, polynactins (alternative name) (653) + TX, proclonol (1350) + TX, profenofos (662) + TX, promacyl (1354) + TX, propargite (671) + TX, propetamphos (673) + TX, propoxur (678) + TX, prothidathion (1360) + TX, prothoate (1362) + TX, pyrethrin I (696) + TX, pyrethrin II (696) + TX, pyrethrins (696) + TX, pyridaben (699) + TX, pyridaphenthion (701) + TX, pyrimidifen (706) + TX, pyrimitate (1370) + TX, quinalphos (711) + TX, quintiofos (1381) + TX, R-1492 (development code
- development code (development code) (1383) + TX, rotenone (722) + TX, schradan (1389) + TX, sebufos (alternative name) + TX, selamectin (alternative name) [CCN] + TX, SI-0009 (compound code) + TX, sophamide (1402) + TX, spirodiclofen (738) + TX, spiromesifen (739) + TX, SSI-121 (development code) (1404) + TX, sulfiram (alternative name) [CCN] + TX, sulfluramid (750) + TX, sulfotep (753) + TX, sulphur (754) + TX, SZI-121 (development code) (757) + TX, tau- fluvalinate (398) + TX, tebufenpyrad (763) + TX, TEPP (1417) + TX, terbam
- an algicide selected from the group of substances consisting of bethoxazin [CCN] + TX, copper dioctanoate (IUPAC name) (170) + TX, copper sulfate (172) + TX, cybutryne [CCN] + TX, dichlone (1052) + TX, dichlorophen (232) + TX, endothal (295) + TX, fentin (347) + TX, hydrated lime [CCN] + TX, nabam (566) + TX, quinoclamine (714) + TX, quinonamid (1379) + TX, simazine (730) + TX, triphenyltin acetate (IUPAC name) (347) and triphenyltin hydroxide (IUPAC name) (347) + TX,
- an anthelmintic selected from the group of substances consisting of abamectin (1) + TX, crufomate (1011) + TX, doramectin (alternative name) [CCN] + TX, emamectin (291) + TX, emamectin benzoate (291) + TX, eprinomectin (alternative name) [CCN] + TX, ivermectin (alternative name) [CCN] + TX, milbemycin oxime (alternative name) [CCN] + TX, moxidectin (alternative name) [CCN] + TX, piperazine [CCN] + TX, selamectin (alternative name) [CCN] + TX, spinosad (737) and thiophanate (1435) + TX,
- an avicide selected from the group of substances consisting of chloralose (127) + TX, endrin (1122) + TX, fenthion (346) + TX, pyridin-4-amine (IUPAC name) (23) and strychnine (745) + TX,
- a bactericide selected from the group of substances consisting of l-hydroxy-lH-pyridine-2- thione (IUPAC name) (1222) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (IUPAC name) (748) + TX, 8-hydroxyquinoline sulfate (446) + TX, bronopol (97) + TX, copper dioctanoate (IUPAC name) (170) + TX, copper hydroxide (IUPAC name) (169) + TX, cresol [CCN] + TX, dichlorophen (232) + TX, dipyrithione (1105) + TX, dodicin (1112) + TX, fenaminosulf (1144) + TX, formaldehyde (404) + TX, hydrargaphen (alternative name) [CCN] + TX, kasugamycin (483) + TX, kasugamycin hydrochloride hydrate (483) + TX
- a biological agent selected from the group of substances consisting of Adoxophyes orana GV (alternative name) (12) + TX, Agrobacterium radiobacter (alternative name) (13) + TX, Amblyseius spp. (alternative name) (19) + TX, Anagrapha falcifera NPV (alternative name) (28) + TX, Anagrus atomus (alternative name) (29) + TX, Aphelinus abdominalis (alternative name) (33) + TX, Aphidius colemani (alternative name) (34) + TX, Aphidoletes aphidimyza (alternative name) (35) + TX, Autographa californica NPV (alternative name) (38) + TX, Bacillus firmus (alternative name) (48) + TX, Bacillus sphaericus Neide (scientific name) (49) + TX, Bacillus thuringiensis Hopkins (scientific name) (5
- israeltaki (scientific name) (51) + TX, Bacillus thuringiensis subsp. tenebrionis (scientific name) (51) + TX, Beauveria bassiana (alternative name) (53) + TX, Beauveria brongniartii (alternative name) (54) + TX, Chrysoperla carnea (alternative name) (151) + TX, Cryptolaemus
- montrouzieri (alternative name) (178) + TX, Cydia pomoneiia GV (alternative name) (191) + TX, Dacnusa sibirica (alternative name) (212) + TX, Diglyphus isaea (alternative name) (254) + TX, Encarsia formosa (scientific name) (293) + TX, Eretmocerus eremicus (alternative name) (300) + TX, Helicoverpa zea NPV (alternative name) (431) + TX, Heterorhabditis bacteriophora and H.
- a soil sterilant selected from the group of substances consisting of iodomethane (IUPAC name) (542) and methyl bromide (537) + TX,
- a chemosterilant selected from the group of substances consisting of apholate [CCN] + TX, bisazir (alternative name) [CCN] + TX, busulfan (alternative name) [CCN] + TX, diflubenzuron (250) + TX, dimatif (alternative name) [CCN] + TX, hemel [CCN] + TX, hempa [CCN] + TX, metepa [CCN] + TX, methiotepa [CCN] + TX, methyl apholate [CCN] + TX, morzid [CCN] + TX, penfluron (alternative name) [CCN] + TX, tepa [CCN] + TX, thiohempa (alternative name) [CCN] + TX, thiotepa (alternative name) [CCN] + TX, tretamine (alternative name) [CCN] and
- an insect pheromone selected from the group of substances consisting of (£)-dec-5-en-l-yl acetate with (£)-dec-5-en-l-ol (IUPAC name) (222) + TX, (£)-tridec-4-en-l-yl acetate (IUPAC name) (829) + TX, (£)-6-methylhept-2-en-4-ol (IUPAC name) (541) + TX, (E,Z)-tetradeca-4, 10- dien-l-yl acetate (IUPAC name) (779) + TX, (Z)-dodec-7-en-l-yl acetate (IUPAC name) (285) + TX, (Z)-hexadec-ll-enal (IUPAC name) (436) + TX, (Z)-hexadec-ll-en-l-yl acetate (IUPAC name) (437) + TX, (Z)-hexadec-13-en
- an insecticide selected from the group of substances consisting of 1-dichloro-l-nitroethane (IUPAC/Chemical Abstracts name) (1058) + TX, l,l-dichloro-2,2-bis(4-ethylphenyl)ethane (IUPAC name) (1056), + TX, 1,2-dichloropropane (IUPAC/Chemical Abstracts name) (1062) + TX, 1,2-dichloropropane with 1,3-dichloropropene (IUPAC name) (1063) + TX, l-bromo-2- chloroethane (IUPAC/Chemical Abstracts name) (916) + TX, 2,2,2-trichloro-l-(3,4-dichloro- phenyl)ethyl acetate (IUPAC name) (1451) + TX, 2,2-dichlorovinyl 2-ethylsulphinylethyl methyl phosphate (IUPAC name) (1066) + TX, 2-
- ethylphosphonothioate (1057) + TX, 0,0-diethyl 0-4-methyl-2-oxo-2H-chromen- 7-yl phosphorothioate (IUPAC name) (1074) + TX, ⁇ , ⁇ -diethyl 0-6-methyl-2-propylpyrimidin-4- yl phosphorothioate (IUPAC name) (1075) + TX, ⁇ , ⁇ , ⁇ ', O'-tetrapropyl dithiopyrophosphate (IUPAC name) (1424) + TX, oleic acid (IUPAC name) (593) + TX, omethoate (594) + TX, oxamyl (602) + TX, oxydemeton-methyl (609) + TX, oxydeprofos (1324) + TX, oxydisulfoton (1325) + TX, pp'-DDT (219) + TX, para-dichlorobenzen
- TX sodium hexafluorosilicate (1400) + TX, sodium pentachlorophenoxide (623) + TX, sodium selenate (IUPAC name) (1401) + TX, sodium thiocyanate [CCN] + TX, sophamide (1402) + TX, spinosad (737) + TX, spiromesifen (739) + TX, spirotetrmat (CCN) + TX, sulcofuron (746) + TX, sulcofuron-sodium (746) + TX, sulfluramid (750) + TX, sulfotep (753) + TX, sulphuryl fluoride (756) + TX, sulprofos (1408) + TX, tar oils (alternative name) (758) + TX, tau-fluvalinate (398) + TX, tazimcarb
- teflubenzuron (768) + TX, tefluthrin (769) + TX, temephos (770) + TX, TEPP (1417) + TX, terallethrin (1418) + TX, terbam (alternative name) + TX, terbufos (773) + TX,
- a molluscicide selected from the group of substances consisting of bis(tributyltin) oxide (IUPAC name) (913) + TX, bromoacetamide [CCN] + TX, calcium arsenate [CCN] + TX, cloethocarb (999) + TX, copper acetoarsenite [CCN] + TX, copper sulfate (172) + TX, fentin (347) + TX, ferric phosphate (IUPAC name) (352) + TX, metaldehyde (518) + TX, methiocarb (530) + TX, niclosamide (576) + TX, niclosamide-olamine (576) + TX, pentachlorophenol (623) + TX, sodium pentachlorophenoxide (623) + TX, tazimcarb (1412) + TX, thiodicarb (799) + TX, tributyltin oxide (913) + T
- a nematicide selected from the group of substances consisting of AKD-3088 (compound code) + TX, l,2-dibromo-3-chloropropane (IUPAC/Chemical Abstracts name) (1045) + TX, 1,2- dichloropropane (IUPAC/ Chemical Abstracts name) (1062) + TX, 1,2-dichloropropane with 1,3- dichloropropene (IUPAC name) (1063) + TX, 1,3-dichloropropene (233) + TX, 3,4- dichlorotetrahydrothiophene 1,1-dioxide (IUPAC/Chemical Abstracts name) (1065) + TX, 3-(4- chlorophenyl)-5-methylrhodanine (IUPAC name) (980) + TX, 5-methyl-6-thioxo-l,3,5- thiadiazinan-3-ylacetic acid (IUPAC name) (1286) + TX, 6-isopentenylaminopurine (
- a nitrification inhibitor selected from the group of substances consisting of potassium ethylxanthate [CCN] and nitrapyrin (580) + TX,
- a plant activator selected from the group of substances consisting of acibenzolar (6) + TX, acibenzolar-S-methyl (6) + TX, probenazole (658) and Reynoutria sachalinensis extract
- a rodenticide selected from the group of substances consisting of 2-isovalerylindan-l,3- dione (IUPAC name) (1246) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (IUPAC name) (748) + TX, alpha-chlorohydrin [CCN] + TX, aluminium phosphide (640) + TX, antu (880) + TX, arsenous oxide (882) + TX, barium carbonate (891) + TX, bisthiosemi (912) + TX, brodifacoum (89) + TX, bromadiolone (91) + TX, bromethalin (92) + TX, calcium cyanide (444) + TX, chloralose (127) + TX, chlorophacinone (140) + TX, cholecalciferol (alternative name) (850) + TX, coumachlor (1004) + TX, coumafuryl (
- a synergist selected from the group of substances consisting of 2-(2-butoxyethoxy)ethyl piperonylate (IUPAC name) (934) + TX, 5-(l,3-benzodioxol-5-yl)-3-hexylcyclohex-2-enone (IUPAC name) (903) + TX, farnesol with nerolidol (alternative name) (324) + TX, MB-599 (development code) (498) + TX, MGK 264 (development code) (296) + TX, piperonyl butoxide (649) + TX, piprotal (1343) + TX, propyl isomer (1358) + TX, S421 (development code) (724) + TX, sesamex (1393) + TX, sesasmolin (1394) and sulfoxide (1406) + TX,
- an animal repellent selected from the group of substances consisting of anthraquinone (32)
- TX chloralose (127) + TX, copper naphthenate [CCN] + TX, copper oxychloride (171) + TX, diazinon (227) + TX, dicyclopentadiene (chemical name) (1069) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, methiocarb (530) + TX, pyridin-4-amine (IUPAC name) (23) + TX, thiram (804) + TX, trimethacarb (840) + TX, zinc naphthenate [CCN] and ziram (856) + TX, a virucide selected from the group of substances consisting of imanin (alternative name)
- a wound protectant selected from the group of substances consisting of mercuric oxide (512) + TX, octhilinone (590) and thiophanate-methyl (802) + TX,
- azaconazole 60207-31-0] + TX, bitertanol [70585-36-3] + TX, bromuconazole [116255-48-2] + TX, cyproconazole [94361-06-5] + TX, difenoconazole [119446-68-3] + TX, diniconazole [83657-24- 3] + TX, epoxiconazole [106325-08-0] + TX, fenbuconazole [114369-43-6] + TX,
- TX isoprothiolane [50512-35-1] + TX, phosdiphen [36519-00-3] + TX, pyrazophos [13457-18-6] + TX, tolclofos-methyl [57018-04-9] + TX, acibenzolar-S-methyl [135158-54-2] + TX, anilazine [101-05-3] + TX, benthiavalicarb [413615-35-7] + TX, blasticidin-S [2079-00-7] + TX, chinomethionat [2439-01-2] + TX, chloroneb [2675-77-6] + TX, chlorothalonil [1897-45-6] + TX, cyflufenamid [180409-60-3] + TX, cymoxanil [57966-95-7] + TX, dichlone [117-80-6] + TX, diclocymet [139920-32-4] + TX
- the components (B) are known.
- the references in brackets behind the active ingredients, e.g. [3878-19-1] refer to the Chemical Abstracts Registry number.
- the above described mixing partners are known.
- the active ingredients are included in "The Pesticide Manual” [The Pesticide Manual - A World Compendium; Thirteenth Edition; Editor: C. D. S. TomLin; The British Crop Protection Council]
- they are described therein under the entry number given in round brackets hereinabove for the particular compound; for example, the compound “abamectin” is described under entry number (1).
- [CCN] is added hereinabove to the particular compound, the compound in question is included in the "Compendium of Pesticide Common Names", which is accessible on the internet under the internet address
- the compound of the formula I is preferably a compound selected from the Tables 1-7 or Table 9; more preferably Table 9;
- the mixing ratios can vary over a large range and are, preferably 100: 1 to 1:6000, especially 50: 1 to 1:50, more especially 20: 1 to 1:20, even more especially 10: 1 to 1: 10.
- Those mixing ratios are understood to include, on the one hand, ratios by weight and also, on other hand, molar ratios.
- mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient” relates to the respective mixture of TX with the mixing partner).
- Some mixtures may comprise active ingredients which have significantly different physical, chemical or biological properties such that they do not easily lend themselves to the same conventional formulation type.
- other formulation types may be prepared.
- one active ingredient is a water insoluble solid and the other a water insoluble liquid
- the resultant composition is a suspoemulsion (SE) formulation.
- the mixtures comprising a TX selected from Tables 1-7 or Table 9 and one or more active ingredients as described above can be applied, for example, in a single "ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a "tank-mix", and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- the order of applying the compounds of formula I selected from Tables 1-7 or Table 9 and the active ingredients as described above is not essential for working the present invention.
- Step B) A 50 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 2-methyl-5,6,7,8-tetrahydro-quinolin-4-ol (4.00 g) in phosphorus oxide chloride (18.3 mL) under an argon atmosphere. The resulting colourless solution was stirred at 100°C for 3.5 hours, after which time TLC indicated that no starting material was remaining. The solvent was removed in vacuo and hot water (40-50°C) was added carefully and slowly to the residue to hydrolyse the remaining phosphorus oxide chloride. Under cooling with an ice- water cooling bath, the pH was adjusted to 12 by the addition of 4 M aqueous NaOH.
- Step C A 25 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 4-chloro-2-methyl-5,6,7,8-tetrahydroquinoline (560 mg) in acetic anhydride (0.49 mL). Under stirring, benzaldehyde (0.34 mL) was added and the resulting yellow solution was stirred under heating to reflux for 3.5 hours. Following the course of the reaction by TLC indicated that starting material was consumed by this time. The resulting brown solution was cooled to room temperature. Crushed ice was added and the pH was adjusted to 10 using a small amount of 2 M aqueous NaOH. Extraction was carried out using ethyl acetate (2x20 mL).
- Step A A 5 mL microwave tube was charged with a solution of 4-chloro-2-methyl-5,6,7,8- tetrahydroquinoline (500 mg) in 1,2-dichloroethane (2.50 mL). Under stirring, trimethylboroxine (380 mg), potassium carbonate (647 mg) and dichloro[l,l'-bis(diphenylphosphino)ferrocene] palladium(II) dichloromethane adduct (101 mg) were added and the resulting red suspension was degassed under argon for 5 minutes. The reaction mixture was subjected to microwave irradiation at 120°C for 0.5 hour.
- Step C A 25 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 2,4-dimethyl-5,6,7,8-tetrahydro-quinoline-l-oxide (334 mg) in
- Step D A 25 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 2,4-dimethyl-5,6,7,8-tetrahydro-quinolin-8-ol (226 mg) in chloroform (2.00 mL). Under stirring, manganese(IV) oxide (443 mg) was added and the resulting black suspension was stirred under heating to reflux for 18 hours, after which time TLC indicated that no starting material was remaining. The resulting black material was purified by chromatography on silica gel (eluent: heptane / ethyl acetate gradient from 1:1 to 1:2 (v:v)). This gave the tittle compound (78 mg) as an orange gum.
- Step A) 9-Methyl-l,2,3,4-tetrahydroacridine In a round-bottom flask, cyclohexanone (6.1 mL, 58 mmol) was heated at 90°C, and 2-aminoacetophenone hydrochloride (10 g; 58 mmol) was added by small fractions. The bottom flask was then equipped with a condenser, and the crude heterogeneous mixture was further heated overnight at 110°C. After cooling to room temperature, the red-orange solid was dissolved in ethanol/HCI (12 N) [95/5 v/v]. The solution was then neutralized with an aqueous NaOH solution.
- Step B) N xide-9-methyl-l,2,3,4-tetrahydroacridine A dichloromethane solution (300 mL) of 3-chloroperbenzoic acid (26 g, 105 mmol) was slowly added to a dichloromethane solution (100 mL) of 9-methyl-l,2,3,4-tetrahydroacridine (10.2 g, 52 mmol) at 0°C. The mixture was stirred for 4 h at room temperature and quenched with an aqueous NaOH solution. The organic layers were further washed with water (5 x 100 mL) and dried over MgS0 4 , and the solvent was removed under reduced pressure giving desired product as a brownish solid.
- the crude solid was dissolved in methanol (50 mL) and saponified by an aqueous K 2 C0 3 solution (2 M, 150 mL); a brown solid precipitated.
- the methanol was removed under reduced pressure, and the product was extracted with dichloromethane (2 x 150 mL).
- the combined organic layers were washed with brine (2 x 50 mL), dried over magnesium sulfate, and evaporated to dryness.
- the desired product was recovered as a brown solid (9.4 g, 84%).
- Step D) 9-methyl-2,3-dihydro-lH-acridin-4-one To a dichloromethane solution (300 mL) of
- HP 1100 HPLC from Agilent solvent degasser, binary pump, heated column compartment and diode-array detector.
- HP 1100 HPLC from Agilent solvent degasser, quaternary pump (ZCQ) / binary pump (ZDQ), heated column compartment and diode-array detector.
- Type of column Waters ACQUITY UPLC HSS T3; Column length: 30 mm; Internal diameter of column: 2.1 mm; Particle Size: 1.8 micron; Temperature: 60°C.
- Ionization method Electrospray; Polarity: positive and negative ions; Capillary (kV): 3.00; Cone (V): 30.00; Extractor (V): 2.00; Source Temperature (°C): 100; Desolvation Temperature (°C): 250; Cone Gas Flow (L/Hr): 50; Desolvation Gas Flow (L/Hr): 400; Mass range: 100 to 900 Da
- Solvent degasser binary pump, heated column compartment and diode-array detector; Column: Phenomenex Gemini C18, 3 - m, 30 x 3 mm; Temp: 60°C; DAD Wavelength range (nm): 210 to 500;
- Solvent degasser binary pump, heated column compartment and diode-array detector.
- Wheat leaf segments cultivated variety (cv) Kanzler were placed on agar in 24-well plates and sprayed with formulated test compound diluted in water at an application rate of 200ppm.
- the leaf disks were inoculated with a spore suspension of the fungus 1 day after application.
- the inoculated leaf segments were incubated at 19°C and 75% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (7 - 9 days after application).
- the following compounds gave at least 80% control of Puccinia recondita f. sp. tritici: P.01, P.02, P.04, P.05, P.06, P.08, P.09. P.20, P.23, P.27, P.28, P.29, P.30.
- Wheat leaf segments cv Kanzler were placed on agar in 24-well plates. The leaf segments were inoculated with a spore suspension of the fungus. The plates were stored in darkness at 19°C and 75% relative humidity. The formulated test compound diluted in water was applied at an application rate of 200ppm 1 day after inoculation. The leaf segments were incubated at 19°C and 75% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (6 - 8 days after application). The following compounds gave at least 80% control of Puccinia recondita f. sp. tritici: P.01, P.02, P.04, P.05, P.06, P.08, P.09, P.11. P.20, P.23, P.27, P.28, P.29.
- Phaeosphaeria nodorum (Seotoria nodorum) / wheat / leaf disc preventative (Glume blotch):
- Wheat leaf segments cv Kanzler were placed on agar in a 24-well plate and sprayed with formulated test compound diluted in water at an application rate of 200ppm.
- the leaf disks are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated test leaf disks are incubated at 20°C and 75% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (5 - 7 days after application).
- Phaeosphaeria nodorum The following compounds gave at least 80% control of Phaeosphaeria nodorum: P.01, P.02, P.04, P.05, P.06, P.08, P.09, P.ll, P.14, P.16, P.17, P.18P.19, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30.
- Barley leaf segments cv Hasso are placed on agar in a 24-well plate and sprayed with formulated test compound diluted in water at an application rate of 200ppm.
- the leaf segments are inoculated with a spore suspension of the fungus two days after application of the test solution.
- the inoculated leaf segments are incubated at 20°C and 65% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (5 - 7 days after application).
- Tomato leaf disks cv Baby are placed on agar in 24-well plates (24-well format) and sprayed with the formulated test compound diluted in water at an application rate of 200ppm.
- the leaf disks are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated leaf disks are incubated at 23°C/21°C (day/night) and 80% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check disk leaf disks (5 - 7 days after application).
- the following compounds gave at least 80% control of Alternaria solani: P.08,P.29.
- Mycelia fragments and oospores of a newly grown liquid culture of the fungus were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96 well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal mycelia/spore mixture was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 2-3 days after application. The following compounds gave at least 80% control of Pythium ultimum: P.08,P.27, P.28.
- Botryotinia fuckeliana Botryotis cinerea I liquid culture (Gray mould):
- Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (Vogels broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 3-4 days after application.
- the following compounds gave at least 80% control of Botryotinia fuckeliana: P.01, P.02, P.03, P.04, P.05, P.06, P.07, P.09, P.10, P. ll, P.12, P.14, P.15. P.18, P.19, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30, P.31.
- Glomerella lagenarium (Colletotrichum lagenarium) / liquid culture (Anthracnose) :
- Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth is measured photometrically 3-4 days after application. The following compounds gave at least 80% control of Glomerella lagenarium: P.01, P.02, P.03, P.04, P.06, P.08, P.09, P.ll, P.14, P.15. P.22, P.23, P.27, P.28, P.29, P.30.
- Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates are incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application.
- the following compounds gave at least 80% control of Mycosphaerella arachidis: P.01, P.02, P.03, P.04, P.05, P.06, P.08, P.09, P.10, P. ll, P.12, P.14, P.15. P.18, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.31.
- Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application.
- the following compounds gave at least 80% control of Mycosphaerella graminicola: P.01, P.02, P.03, P.04, P.06, P.08, P.09, P.10, P.ll, P.12, P.14, P.15. P.18, P.19, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30, P.31.
- Gaeumannomyces graminis I liquid culture (Take-all of cereals):
- Mycelial fragments of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96- well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores is added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application. The following compounds gave at least 80% control of Gaeumannomyces graminis: P.01, P.02, P.03, P.08, P.09, P. ll, P.14, P.15., P.20, P.21, P.22, P.23, P.27, P.31.
- Thanatephorus cucumeris (Rhizoctonia solani) I liquid culture (foot rot, dampinq-off): Mycelia fragments of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of the test compounds into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal material was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 3-4 days after application. The following compounds gave at least 80% control of Thanatephorus cucumeris: P.01, P.02, P.04, P.06, P.08, P.09, P.14, P.15.
- Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application. The following compounds gave at least 80% control of Monographella nivalis: P.01, P.02, P.04, P.06, P.08, P.09. P.21, P.22, P.23,.28, P.29.
- Wheat leaf segments cv. Kanzler were placed on agar in a 24-well plate and sprayed with the formulated test compound diluted in water at an application rate of 200ppm.
- the leaf disks were inoculated by shaking powdery mildew infected plants above the test plates 1 day after application.
- the inoculated leaf disks were incubated at 20°C and 60% relative humidity under a light regime of 24 h darkness followed by 12h/12h (dark/light) in a climate chamber and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check leaf segments (6 - 8 days after application).
- Mycelia fragments of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate at an application rate of 200ppm (96-well format) the nutrient broth containing the fungal material is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 3-4 days after application. The following compounds gave at least 80% control of : _Sclerotinia sclerotiorum: P.04, P.06, P.09, P.15. P.23, P.27.
- Rice leaf segments cv. Ballila are placed on agar in a multiwell plate (24-well format) and sprayed with the formulated test compound diluted in water at an application rate of 200ppm.
- the leaf segments are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated leaf segments are incubated at 22°C and 80% rh under a light regime of 24 h darkness followed by 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (5 - 7 days after application).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
The invention relates to compounds of formula (I) wherein A, X, Y1, Y3, Y4, Y6, D1, D2, D3, G1, G2, G3 and p are as defined in the claims. The invention further provides intermediates used in the preparation of these compounds, to compositions which comprise these compounds and to their use in agriculture or horticulture for controlling or preventing infestation of plants by phytopathogenic microorganisms, preferably fungi.
Description
CYCLIC BISOXIME MICROBICIDES
The present invention relates to novel microbiocidally active, in particular fungicidally active, cyclic bisoxime derivatives. It further relates to intermediates used in the preparation of these compounds, to compositions which comprise these compounds and to their use in agriculture or horticulture for controlling or preventing infestation of plants by phytopathogenic
microorganisms, preferably fungi.
Fungicidally active bisoximes are described in WO08074418.
Surprisingly, it has been found that novel cyclic bisoxime derivatives have microbiocidal activity.
The present invention accordingly relates to bisoxime derivatives of formula (I)
(I)
wherein
D1 represents N or C-Y2;
D2 represents N or C-Y5;
D3 represents N or C-Y7;
wherein at least one of D1, D2 or D3 is N;
wherein both D2 and D3 cannot be N;
A represents hydrogen, halogen, CN, OH, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, d-C8 alkylsulphonyl, NH2, Ci-Cio alkyl, C3-C8 cycloalkyl, C2-C8 alkenyl, C2-C8 alkynyl, (R10O)carbonyl(Ci- C4-alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C3-C6 cycloalkyl and a 5- or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms;
or if D3 is C-Y7 then A and Y7 together may be -(G4)q-G5-G6-;
X represents X-2, X-3, X-4 or X-5:
#—∑—∑—# #— z— z— z— # #— z— z— z— z— #
X-2 X-3 X-4
#_z12_zli_z12_z13_zli_#
X-5
Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 independently of one another represent CRJR2, C=CR3R4 or C=0;
Z4 and Z12 independently of one another represent C=CR3R4, CR5R6, SiR7R8 or C=0;
each R1 and R2 independently of one another represent hydrogen, halogen, OH, d-C4 alkyl,
Ci-C4 haloalkyi, C3-C6 cycloalkyi, C3-C6 halocycloalkyi, C1-C4 alkylthio, C1-C4 alkylsulphinyl, C1-C4 alkylsulphonyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, d-C4 alkoxy and C1-C4 haloalkoxy;
or R1 and R2 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyi group;
each R3 and R4 independently of one another represent hydrogen, halogen, C1-C4 alkyl or C1-C4 haloalkyi;
each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, Ci- C4 alkyl, C1-C4 haloalkyi, C3-C6 cycloalkyi, C3-C6 halocycloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, d-C4 alkoxy and C1-C4 haloalkoxy;
or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyi group;
wherein the groupings X-2, X-3, X-4 and X-5 may contain at most one ring which contains either only one of the radicals Z1 to Z14 or two radicals Z1 to Z14 or three radicals Z1 to Z14 or four radicals Z1 to Z14 as ring members; and wherein radicals Z1, Z2, Z3, Z5, Z6, Z9, Z10 and Z14 are not substituted by OH;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one another represent hydrogen, halogen, CN, N02, Ci-C8 alkyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy-Ci-C4-alkyl, C3-C8 cycloalkyi, C2-C8 alkenyl, C2-C8 alkynyl, phenyl, pyridyl, pyrimidinyl, COR9, OR10, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, CrC8 alkylsulphonyl, N(Rn)2, C02R10, 0(CO)R9, CON(Rn)2, NRnCOR9 or CR9N- OR10, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, pyrimidinyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-
C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and Ci-C4 alkylsulphonyl;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from 0, S, N and N(Rn), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6, or Y6 and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyi, d-C4 alkoxy and CrC4 haloalkoxy;
each R9 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C2- C8 alkenyl, C2-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
each R10 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C3-C8 alkenyl, C3-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups
independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, Ci-C4 haloalkoxy and CrC4-alkoxy-d-C4-alkyl;
each R11 independently of one another represents hydrogen, OH, d-C8 alkyl, Ci-C8 alkoxy, Ci-C8-alkoxy-Ci-C4-alkyl, C3-C8 alkenyl, C3-C8 alkynyl, COR9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, d-C4 alkyl, d-C4 haloalkyi, C C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and d-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5, B-6, B-7 or B-8:
B-1 B-2 B-3 B-4 B-5 B-6 B-7 B-8 wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
G1, G2, G4 and G5 independently of one another represent -C(R12R13)-, 0, N(R14) or S;
G3 and G6 independently of one another represent -C(R12R13)-, 0, N(R14) or S;
or G1 and G2, or G2 and G3, or G1 and G1, or G4 and G5, or G5 and G6, or G4 and G4 together represent -CR12=CR13-;
each R12 and R13 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci- C4 haloalkyl, Ci-C4 alkoxy or Ci-C4 haloalkoxy;
R14 represents hydrogen, OH, Ci-C4 alkyl, Ci-C4 alkoxy, C3-C6 cycloalkyl, Ci-C8 alkylcarbonyl or Ci-C8 haloalkylcarbonyl; and
p and q are each independently 0, 1 or 2;
or a salt or an N-oxide thereof.
Halogen, either as a lone substituent or in combination with another substituent (e.g.
haloalkyl) is generally fluorine, chlorine, bromine or iodine, and usually fluorine, chlorine or bromine.
Each alkyl moiety (including the alkyl moiety of alkoxy, alkylthio, etc.) is a straight or branched chain and, depending on the number of carbon atoms it contains, is, for example, methyl, ethyl, n-propyl, n-butyl, n-pentyl, n-hexyl, /so-propyl, sec-butyl, /so-butyl, tert-butyl, neo-pentyl, n-heptyl or 1,3-dimethylbutyl, and usually methyl or ethyl.
The alkenyl and alkynyl groups can be mono- or di-unsaturated and are examples thereof are derived from the above mentioned alkyl groups.
Haloalkyl moieties are alkyl moieties which are substituted by one or more of the same or different halogen atoms and are, for example, monofluoromethyl, difluoromethyl,
trifluoromethyl, monochloromethyl, dichloromethyl, trichloromethyl, 2,2,2-trifluoroethyl, 2,2- difluoroethyl, 2-fluoroethyl, 1,1 -difluoroethyl, 1-fluoroethyl, 2-chloroethyl, pentafluoroethyl, 1,1-
difluoro-2,2,2-trichloroethyl, 2,2,3,3-tetrafluoroethyl and 2,2,2 -trichloroethyl, and typically trichloromethyl, difluorochloromethyl, difluoromethyl, trifluoromethyl and dichlorofluoromethyl.
Alkoxy is, for example, methoxy, ethoxy, propoxy, /'so-propoxy, n-butoxy, /so-butoxy, sec- butoxy and tert-butoxy, and usually methoxy or ethoxy.
Haloalkoxy is, for example, fluoromethoxy, difluoromethoxy, trifluoromethoxy, 2,2,2- trifluoroethoxy, 1,1,2,2-tetrafluoroethoxy, 2-fluoroethoxy, 2-chloroethoxy, 2,2-difluoroethoxy and 2,2,2-trichloroethoxy, and usually difluoromethoxy, 2-chloroethoxy and trifluoromethoxy.
Alkylthio is, for example, methylthio, ethylthio, propylthio, /so-propylthio, n-butylthio, iso- butylthio, sec-butylthio or tert- butylthio, and usually methylthio or ethylthio.
Alkylsulphonyl is, for example, methylsulphonyl, ethylsulphonyl, propylsulphonyl, iso- propylsulphonyl, n-butylsulphonyl, /'so-butylsulphonyl, sec-butylsulphonyl or tert-butylsulphonyl, and usually methylsulphonyl or ethylsulphonyl.
Alkylsulphinyl is, for example, methylsulphinyl, ethylsulphinyl, propylsulphinyl, iso- propylsulphinyl, n-butylsulphinyl, /so-butylsulphinyl, sec-butylsulphinyl or tert-butylsulphinyl, and usually methylsulphinyl or ethylsulphinyl
Cycloalkyl may be saturated or partially unsaturated, preferably fully saturated, and is, for example, cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl.
Alkoxyalkyl is, for example, methoxymethyl, methoxyethyl, ethoxymethyl, ethoxyethyl, n- propoxymethyl, n-propoxyethyl, /so-propoxymethyl or /so-propoxyethyl.
Aryl includes phenyl, naphthyl, anthracyl, fluorenyl and indanyl, but is usually phenyl.
Carbocycle includes cycloalkyl groups and aryl groups.
Heterocycloalkyl is a non-aromatic ring that may be saturated or partially unsaturated, preferably fully saturated, containing carbon atoms as ring members and at least one heteroatom selected from 0, S and N as ring members. Examples include oxiranyl, oxetanyl, tetrahydrofuranyl, tetrahydropyranyl, 1,3-dioxolanyl, 1,4-dioxanyl, aziridinyl, azetidinyl, pyrrolidinyl, piperidinyl, oxazinanyl, morpholinyl, thiomorpholinyl, imidazolidinyl, pyrazolidinyl and piperazinyl, preferably morpholinyl, pyrrolidinyl, piperdinyl and piperazinyl, more preferably morpholinyl and pyrollidinyl.
Heteroaryl is, for example, a monovalent monocyclic or bicyclic aromatic hydrocarbon radical. Examples of monocyclic groups include pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, pyrrolyl, pyrazolyl, imidazolyl, triazolyl, tetrazolyl, furanyl, thiophenyl, oxazolyl, isoxazolyl, oxadiazolyl, thiazolyl, isothiazolyl, and thiadiazolyl. Examples of bicyclic groups include quinolinyl, cinnolinyl, quinoxalinyl, benzimidazolyl, benzothiophenyl, and benzothiadiazolyl.
Monocyclic heteroaryl groups are preferred, preferably pyridyl, pyrrolyl, imidazolyl and triazolyl, e.g. 1,2,4 triazolyl, pyridyl and imidazolyl being most preferred.
The terms "heterocycle" and "heterocyclic ring" are used interchangeably and are defined to include heterocycloalkyl and heteroaryl groups. Any reference herein to a heterocycle or heterocyclic ring preferably refers to the specific examples given under the definition of heteroaryl and heterocycloalkyl above, and are preferably morpholinyl, pyrrolidinyl, piperdinyl, piperazinyl pyridyl, pyrrolyl, imidazolyl and triazolyl, e.g. 1,2,4 triazolyl, more preferably morpholinyl, pyrollidinyl, pyridyl and imidazolyl.
Where a moiety is indicated as being (optionally) substituted, e.g. alkyl, this includes those moieties where they are part of a larger group, e.g. the alkyl in the alkylthio group. Where a moiety is indicated as being optionally substituted by one or more other groups, preferably there are one to five optional substituents, more preferably one to three optional substituents.
The following substituents definitions, including preferred definitions, may be combined in any combination:
D1 represents N or C-Y2;
D2 represents N or C-Y5;
D3 represents N or C-Y7;
wherein at least one of D1, D2 or D3 is N;
wherein both D2 and D3 cannot be N.
In one group of compounds, D1 represents C-Y2, D2 represents N and D3 represents C-Y7.
In one group of compounds, D1 represents C-Y2, D2 represents C-Y5 and D3 represents N.
In one group of compounds, D1 represents N, D2 represents C-Y5 and D3 represents C-Y7.
In one group of compounds, D1 represents N, D2 represents N and D3 represents C-Y7.
In one group of compounds, D1 represents N, D2 represents C-Y5 and D3 represents N. A represents hydrogen, halogen, CN, OH, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, d-C8 alkylsulphonyl, NH2, Ci-Cio alkyl, C3-C8 cycloalkyl, C2-C8 alkenyl, C2-C8 alkynyl, (R10O)carbonyl(Ci- C4-alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyl, Ci-C4 alkoxy, Ci-C4 haloalkoxy, C3-C6 cycloalkyl and a 5- or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms;
or if D3 is C-Y7 then A and Y7 together may be -(G4)q-G5-G6-.
Preferably A represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, phenyl or pyridyl, wherein the alkyl, cycloalkyi, phenyl and pyridyl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, OH, C1-C4 alkoxy, C1-C4 haloalkoxy and C3-C6 cycloalkyi.
More preferably A represents hydrogen, C1-C4 alkyl, phenyl or pyridyl, wherein alkyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, OH, C1-C4 alkoxy and d-C4 haloalkoxy, and wherein phenyl and pyridyl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, OH, d-C4 alkoxy, CrC4 haloalkoxy and C3-C6 cycloalkyi.
Even more preferably A represents hydrogen, C1-C4 alkyl, phenyl or pyridin-2-yl, wherein the phenyl and pyridin-2-yl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, methyl, halomethyl, methoxy and
halomethoxy.
Yet more preferably, A represents C1-C4 alkyl.
In one preferred group of compounds A represents pyridyl, optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4-alkyl, C1-C4- haloalkyl, d-C4-alkoxy, d-C4-haloalkoxy, C3-C6 cycloalkyi and a 5 or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms. The heterocycle is preferably one as defined herein, preferably morpholinyl, pyrrolidinyl, piperdinyl, piperazinyl, pyridyl, pyrrolyl, imidazolyl or triazolyl, e.g. 1,2,4 triazolyl, more preferably morpholinyl, pyrollidinyl, pyridyl or imidazolyl.
In this preferred group of compounds A preferably represents pyridin-2-yl, optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, d- C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy, C1-C4 haloalkoxy, C3-C6 cycloalkyi and a 5 or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms. The heterocycle is preferably one as defined herein, preferably morpholinyl, pyrrolidinyl, piperdinyl, piperazinyl, pyridyl, pyrrolyl, imidazolyl or triazolyl, e.g. 1,2,4 triazolyl, more preferably morpholinyl, pyrollidinyl, pyridyl or imidazolyl.
In another group of compounds A represents hydrogen, C1-C4 alkyl, C2-C4 alkenyl, phenyl or pyridyl, wherein the alkyl, alkenyl, phenyl and pyridyl are optionally substituted by one or more
groups independently selected from d-C4 alkyi, Ci-C4 haloalkyi, halogen, CN, Ci-C4 alkoxy and Ci-C4 haloalkoxy.
In another preferred group of compounds, A represents hydrogen, halogen, CN, OH, SH, Ci-Cs alkylthio, Ci-C8 alkylsulphinyl, Ci-C8 alkylsulphonyl, NH2, C1-C10 alkyi, C3-C8 cycloalkyl, C2-C8 alkenyl, C2-C8 alkynyl, (d-C4 alkyloxycarbonyl) Ci-C4 alkyi, (Ci-C4 alkyl)02C, phenyl or pyridyl, wherein the alkyi, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyi, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, C3-C6 cycloalkyl and a 5- or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms.
In another preferred group of compounds, A represents hydrogen, (Ci-C4 alkyl)02C, C1-C10 alkyi, phenyl or pyridyl, wherein the alkyi, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, d-C4 alkyi, Ci-C4 haloalkyi, C3-C6 cycloalkyl and a 5- or 6-membered heterocycle containing one to three nitrogen atoms.
X represents X-2, X-3, X-4 or X-5:
#— z— z— # #— z— z— z— # #— z— z— z— z— #
X-2 X-3 X-4
#_z12_zli_z12_z13_zlf_#
X-5
Preferably X represents X-3 or X-5. More preferably X represents X-3.
Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 independently of one another represent CR R2, C=0 or C=CR3R4.
Preferably Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 independently of one another represent methylene, halomethylene, CH(CH3) or C(CH3)2.
More preferably Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 represent methylene or halomethylene.
Most preferably Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 represent methylene.
Z4 and Z12 independently of one another represent CR5R6, SiR7R8, C=0 or C=CR3R4.
Preferably Z4 and Z12 independently of one another represent CR5R6, SiR7R8 or C=CR3R4. More preferably Z4 and Z12 independently of one another represent CR5R6 or C=CR3R4. Most preferably Z4 and Z12 independently of one another represent CR5R6.
Each R1 and R2 independently of one another represent hydrogen, halogen, OH, Ci-C4 alkyl, C1-C4 haloalkyi, C3-C6 cycloalkyi, C3-C6 halocycloalkyi, C1-C4 alkylthio, C1-C4 alkylsulphinyl, C1-C4 alkylsulphonyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, C1-C4 alkyl, d-C4 haloalkyi, C1-C4 alkoxy and d-C4 haloalkoxy; or R1 and R2 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyi group.
Each R3 and R4 independently of one another represent hydrogen, halogen, C1-C4 alkyl or C1-C4 haloalkyi.
Preferably each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl.
Each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, Ci- C4 alkyl, C1-C4 haloalkyi, C3-C6 cycloalkyi, C3-C6 halocycloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy and C1-C4 haloalkoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyi group.
Preferably, each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, C1-C4 alkyl, C1-C4 haloalkyi, C3-C6 cycloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, d-C4 alkyl, C1-C4 haloalkyi, d-C4 alkoxy and d-C4 haloalkoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyi group.
More preferably, each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, d-C4 alkyl, d-C4 haloalkyi, phenyl or CN wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group optionally substituted by halogen.
More preferably still, each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, d-C4 alkyl, C1-C4 haloalkyi or phenyl, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group optionally substituted by halogen.
Y1, Y2, Υ3, Υ4, Υ5, Y6and Υ7 independently of one another represent hydrogen, halogen, CN, N02, Ci-C8 alkyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy-Ci-C4-alkyl, C3-C8 cycloalkyi, C2-C8 alkenyl, C2-C8 alkynyl, phenyl, pyridyl, pyrimidinyl, COR9, OR10, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, d-C8 alkylsulphonyl, N(Rn)2, C02R10, 0(CO)R9, CON(Rn)2, NRnCOR9 or CR9N- OR10, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, pyrimidinyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci- C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and Ci-C4 alkylsulphonyl;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from 0, S, N and N(Rn), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyi, d-C4 alkoxy and d-C4 haloalkoxy.
Preferably, Y1, Y2, Y3, Y4, Y5, Y6 and Y7 independently of one represent hydrogen, halogen, OH, CN, Ci-C8 alkyl, Ci-C8 haloalkyi, Ci-C8 alkoxy, Ci-C8 haloalkoxy, Ci-C8 alkylthio, C3-C8 cycloalkyi, phenyl, pyridyl, N(Rn)2, or NRnCOR9, wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and d-C4 haloalkoxy;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from N and N(Rn)2 and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
wherein each R9 independently represents d-C8 alkyl or Ci-C8 haloalkyi;
wherein each R11 independently of one another represents hydrogen, Ci-C8 alkyl, COR9 or phenyl, wherein the phenyl is optionally substituted by one or more group independently selected from halogen, CN, NH2, N02, OH, d-C4 alkyl, d-C4 haloalkyi, d-C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and d-C4 alkylsulphonyl;
wherein when two radicals R are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5;
wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl.
More preferably, Y1, Y2, Y3, Y4, Y5, Y6 and Y7 independently of one another represent hydrogen, CN, OH, halogen, d-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, C3-C6 cycloalkyl, Ci-C4 alkylthio, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, d-C4 alkyl, Ci-C4 haloalkyi, d- C4 alkoxy and d-C4 haloalkoxy;
wherein each R9 independently represents d-C4 alkyl or Ci-C4 haloalkyi;
wherein each R11 independently of one another represents hydrogen, Ci-Cs alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, d-C4 alkyl, d-C4 haloalkyi, d-C4 alkoxy and d-C4 haloalkoxy; wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5 wherein the cycle formed is optionally substituted by one or more independently selected from halogen, methyl and halomethyl.
Even more preferably, Y1, Y2, Y3, Y4, Y5, Y6 and Y7 independently of one another represent hydrogen, CN, OH, halogen, d-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, C3-C6 cycloalkyl, d-C4 alkylthio, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
wherein each R9 independently represents d-C4 alkyl or Ci-C4 haloalkyi;
wherein each R independently of one another represents hydrogen, Ci-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy.
Yet more preferably, Y1, Y2, Y3, Y4, Y5, Y6 and Y7 independently of one another represent hydrogen, CN, OH, halogen, d-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, C3-C6 cycloalkyi, d-C4 alkylthio, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
wherein each R9 independently represents d-C4 alkyl or Ci-C4 haloalkyi;
wherein each R11 independently of one another represents hydrogen, Ci-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy.
Each R9 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C2- C8 alkenyl, C2-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyi, Ci- C4 alkoxy and Ci-C4 haloalkoxy.
Preferably each R9 independently of one another represent Ci-Cs alkyl or d-C8 haloalkyi.
More preferably each R9 independently of one another represent d-C4 alkyl or Ci-C4 haloalkyi.
Each R10 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C3-C8 alkenyl, C3-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, NH2, N02, OH, d-C4 alkyl, Ci-C4 haloalkyi, d-C4alkoxy, Ci-C4-haloalkoxy and Ci-C4-alkoxy-Ci-C4-alkyl.
Each R11 independently of one another represents hydrogen, OH, Ci-C8 alkyl, Ci-C8 alkoxy, Ci-C8-alkoxy-Ci-C4-alkyl, C3-C8 alkenyl, C3-C8 alkynyl, COR9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, d-C4 alkyl, d-C4 haloalkyi, d-C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and d-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5, B-6, B-7 or B-8:
B-1 B-2 B-3 B-4 B-5 B-6 B-7 B-8 wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy.
Preferably, each R11 independently of one another represents hydrogen, d-C8 alkyl, COR9 or phenyl, wherein the phenyl is optionally substituted by one or more group independently selected from halogen, CN, NH2, N02, OH, d-C4 alkyl, d-C4 haloalkyi, C C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and d-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5;
wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl.
More preferably, each R11 independently of one another represents hydrogen, d-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy; wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy.
More preferably still, each R11 independently of one another represents hydrogen, Ci-Cs alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, d-C4 alkoxy and Ci-C4 haloalkoxy.
G1, G2, G4 and G5 independently of one another represent -C(R12R13)- , 0, N(R14) or S;
G3 and G6 independently of one another represent -C(R12R13)-, 0, N(R14) or S;
or G1 and G2, or G2 and G3, or G1 and G1, or G4 and G5, or G5 and G6, or G4 and G4 together represent -CR12=CR13-. For the avoidance of doubt, when p is 2, G1 and G1 as well as G2 and G3 may represent CR12=CR13 such that the ring contains two double bond moieties. Likewise, when q is 2, G4 and G4 as well as G5 and G6 may represent CR12=CR13.
Preferably G1, G2, G4 and G5 independently of one another represent -C(R12R13)- ;
G3 and G6 independently of one another represent -C(R12R13)-, 0, N(R14) or S;
or G1 and G2, or G2 and G3, or G1 and G1, or G4 and G5, or G5 and G6, or G4 and G4 together represent -CR12=CR13-.
More preferably, G1, G2, G4 and G5 independently of one another represent -C(R12R13)-;
G3 and G6 independently of one another represent -C(R12R13)-, 0, N(R14) or S.
More preferably again, G1, G2, G3, G4, G5 and G6 independently of one another represent - C(R12R13)-.
Even more preferably G1, G2, G3, G4, G5 and G6 represent methylene.
Each R12 and R13 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci-C4 haloalkyi, d-C4 alkoxy or Ci-C4 haloalkoxy.
R14 represents hydrogen, OH, d-C4 alkyl, Ci-C4 alkoxy, d-C6-cycloalkyl, Ci-Cs alkylcarbonyl or Ci-Q haloalkylcarbonyl.
p and q are each independently of one another 0, 1 or 2.
Preferably p and q are each independently of one another 1 or 2.
More preferably p and q are 1.
In one group of compounds, D1 represents N or C-Y2;
D2 represents N or C-Y5;
D3 represents N or C-Y7;
wherein at least one of D1, D2 or D3 is N;
wherein both D2 and D3 cannot be N;
A represents hydrogen, halogen, CN, OH, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, d-C8 alkylsulphonyl, NH2, Ci-Cio alkyl, C3-C8 cycloalkyl, C2-C8 alkenyl, C2-C8 alkynyl, (R10O)carbonyl(Ci- C4-alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyl, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci- haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C3-C6 cycloalkyl and a 5- or 6-membered heterocycle containing one to three heteroatoms independently selected from 0, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms;
or if D3 is C-Y7 then A and Y7 together may be -(G4)q-G5-G6-.
X represents X-2, X-3, X-4 or X-5:
#—∑—∑—# #— z— z— z— # #— z— z— z— z— #
X-2 X-3 X-4
#_z12_zU_z12_z13_zli_#
X-5
Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 independently of one another represent CRJR2, C=CR3R4 or C=0;
Z4 and Z12 independently of one another represent C=CR3R4, CR5R6, SiR7R8 or C=0;
each R1 and R2 independently of one another represent hydrogen, halogen, OH, Ci-d alkyl,
C1-C4 haloalkyl, C3-C6 cycloalkyl, C3-C6 halocycloalkyl, Ci-C4 alkylthio, C1-C4 alkylsulphinyl, Ci-C4 alkylsulphonyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyl, Ci-d alkoxy and C1-C4 haloalkoxy;
or R1 and R2 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyl group or a C3-C6 halocycloalkyl group;
each R3 and R4 independently of one another represent hydrogen, halogen, d-C4 alkyl or C1-C4 haloalkyl;
each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, Ci- C4 alkyl, Ci-d haloalkyl, C3-C6 cycloalkyl, C3-C6 halocycloalkyl, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, d-d alkyl, Ci- haloalkyl, Ci-d alkoxy and Ci-d haloalkoxy;
or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyl group or a C3-C6 halocycloalkyl group;
wherein the groupings X-2, X-3, X-4 and X-5 may contain at most one ring which contains either only one of the radicals Z1 to Z14 or two radicals Z1 to Z14 or three radicals Z1 to Z14 or four radicals Z1 to Z14 as ring members; and wherein radicals Z1, Z2, Z3, Z5, Z6, Z9, Z10 and Z14 are not substituted by OH;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one another represent hydrogen, halogen, CN,
N02, Ci-C8 alkyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy-Ci-C4-alkyl, C3-C8 cycloalkyi, C2-C8 alkenyl, C2-C8 alkynyl, phenyl, pyridyl, pyrimidinyl, COR9, OR10, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, CrC8 alkylsulphonyl, N(Rn)2, C02R10, 0(CO)R9, CON(Rn)2, NRnCOR9 or CR9N- OR10, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, pyrimidinyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci- C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, d-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and Ci-C4 alkylsulphonyl;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from 0, S, N and N(Rn), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C C4 alkyl, CrC4 haloalkyi, C C4 alkoxy and d-C4 haloalkoxy;
each R9 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C2- C8 alkenyl, C2-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
each R10 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C3-C8 alkenyl, C3-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups
independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4-haloalkyl, Ci-C4 alkoxy, Ci-C4 haloalkoxy and d-C4-alkoxy-Ci-C4-alkyl;
each R11 independently of one another represents hydrogen, OH, Ci-C8 alkyl, Ci-C8 alkoxy, Ci-C8-alkoxy-Ci-C4-alkyl, C3-C8 alkenyl, C3-C8 alkynyl, COR9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently
selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, C C4 alkoxy, C C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and d-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5, B-6, B-7 or B-8;
wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
G1, G2, G4 and G5 independently of one another represent -C(R12R13)-;
G3 and G6 independently of one another represent -C(R12R13)-, 0, N(R14) or S;
or G1 and G2, or G2 and G3, or G1 and G1, or G4 and G5, or G5 and G6, or G4 and G4 together represent -CR12=CR13-;
each R12 and R13 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci- C4 haloalkyi, Ci-C4 alkoxy or Ci-C4 haloalkoxy;
R14 represents hydrogen, OH, d-C4 alkyl, Ci-C4 alkoxy, C3-C6 cycloalkyi, Ci-C8 alkylcarbonyl or Ci-C8 haloalkylcarbonyl; and
p and q are each independently 0, 1 or 2.
In another group of compounds, A represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, phenyl or pyridyl, wherein the alkyl, cycloalkyi, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, OH, d-C4 alkoxy, Ci-C4 haloalkoxy and C3-C6 cycloalkyi;
X represents X-3;
Z3 and Z5 independently of one another represent methylene or halomethylene;
Z4 represent C=CR3R4, CR5R6 or SiR7R8;
each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl;
each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, Ci- C4 alkyl, Ci-C4 haloalkyi, phenyl or CN, wherein the phenyl is optionally substituted by one or
more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy;
or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyl group or a C3-C6 halocycloalkyl group;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one represent hydrogen, halogen, OH, CN, d-
C8 alkyl, Ci-C8 haloalkyi, Ci-C8 alkoxy, d-C8 haloalkoxy, Ci-C8 alkylthio, C3-C8 cycloalkyl, phenyl, pyridyl, N(Rn)2, or NRnCOR9, wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy and C1-C4 haloalkoxy;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from N and N(Rn)2 and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy and C1-C4 haloalkoxy;
each R9 independently represents Ci-C8 alkyl or Ci-C8 haloalkyi;
each R11 independently of one another represents hydrogen, Ci-C8 alkyl, COR9 or phenyl, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy, C1-C4 haloalkoxy, C1-C4 alkylthio, C1-C4 alkylsulphinyl and C1-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or C1-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-l, B-2, B-3, B-4, B-5;
wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl;
G1, G2, G3, G4 , G5 and G6 independently of one another represent -C(R12R13)-;
p and q are each independently 0, 1 or 2.
In another group of compounds, A represents hydrogen, Ci-C4 alkyl, phenyl or pyridyl, wherein alkyl is optionally substituted by one or more groups independently selected from halogen, OH, C1-C4 alkoxy and C1-C4 haloalkoxy, and wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyl, OH, d-C4 alkoxy, Ci-C4 haloalkoxy and C3-C6 cycloalkyi;
X represents X-3;
Z3 and Z5 represent methylene;
Z4 represents C=CR3R4 or CR5R6 ;
each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl;
each R5 and R6 independently of one another represent hydrogen, halogen, OH, d-C4 alkyl, Ci-C4 haloalkyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyl group;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one another represent hydrogen, CN, OH, halogen, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy, Ci-C4 haloalkoxy, Ci-C4 alkylthio, C3-C6 cycloalkyi, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
each R9 independently represents d-C4 alkyl or Ci-C4 haloalkyl;
each R11 independently of one another represents hydrogen, Ci-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyl, C C4 alkoxy and CrC4 haloalkoxy;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5 wherein the cycle formed is optionally substituted by one or more independently selected from halogen, methyl and halomethyl;
G1, G2, G3, G4, G5 and G6 represent methylene;
p and q are each independently 0, 1 or 2.
In a further group of compounds, A represents hydrogen, d-C4 alkyl, phenyl or pyridin-2-yl wherein phenyl and pyridin-2-yl are optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy;
X represents X-3;
Z3 and Z5 represent methylene;
Z4 represents C=CR3R4 or CR5R6 ;
each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl;
each R5 and R6 independently of one another represent hydrogen, halogen, OH, d-C4 alkyl,
Ci-C4 haloalkyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyl group;
Y1, Y2, Y3, Y4, Y5, Y6 and Y7 independently of one another represent hydrogen, CN, OH, halogen, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy, Ci-C4 haloalkoxy, d-C4 alkylthio, C3-C6 cycloalkyi, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
each R9 independently represents d-C4 alkyl or Ci-C4 haloalkyl;
each R11 independently of one another represents hydrogen, d-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
G1, G2, G3, G4, G5 and G6 represent methylene;
p and q are each independently 0, 1 or 2.
In one group of compounds, p is 1 and -G^G^G3- represent -CH2-CH2-CH2-.
In another group of compounds, A and Y7 together represent -CH2-CH2-CH2-.
Certain intermediates that can be used to prepare compounds of formula (I) are novel and as such also form part of the present invention.
(LG) (C-1 ) (C-2A) (C-2B)
R28 represents C1-C4 alkyl, C1-C4 haloalkyi or phenyl, wherein the phenyl is optionally substituted by one or two substituents independently selected from methyl, trihalomethyl, N02, CN, C1-C7 alkoxycarbonyl;
X" represents halogen;
R37 represents either A as defined herein for compounds of formula (I) or the group R'
R37'
wherein X, G1, G2, G3, D1, D2, D3, Y1, Y3, Y4, Y6 and p are as defined for the compound of formula (I);
or a salt or N-oxide thereof.
The preferred definitions of A, X, G1, G2, G3, D1, D2, D3, Y1, Y3, Y4, Y6 and p defined in respect of compounds of formula (I) also apply to compounds of formula (II).
Preferably R27 represents -ONH2, -0-S02-R28 or one of the groups LG, C-l or C-2.
Even more preferably R27 represents -ONH2, tosylate, mesylate, triflate or one of the groups LG, C-l or C-2.
wherein X' represents one of the groupings X'-l, X'-2 or X'-3:
#— z— # #— z— z— # #—∑—∑—∑—#
X'-1 X'-2 X'-3
wherein Z3, Z6, Z7, Z10, Z11 and Z12 are as defined for a compound of formula (I);
R29 and R30 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci-C4 haloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkyoxy; and
G1, G2, G3, D1, Y1, Y3 and p are as defined for a compound of formula (I);
or a salt or N-oxide thereof.
Preferably, R29and R30 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci-C4 haloalkyi, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and
halomethoxy.
In one group of compounds of formula (III) at least one of Y4, Y5 or Y6 represents pyrimidinyl, Ci-Cs haloalkylthio, Ci-Cs haloalkylsulphinyl or Ci-Cs haloalkylsulphonyl ,wherein the pyrimidinyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C C4 alkyl, C C4 haloalkyi, C C4 alkoxy, d-C4 haloalkoxy, d-C4 alkylthio, Ci-C4 alkylsulphinyl and Ci-C4 alkylsulphonyl.
In another group of compounds of formula (III) G3 represents N(R14) wherein R14 represents
C3-C6 cycloalkyl.
The preferred definitions of Z5, Z6, Z9, Z13, Z14, D1, G1, G2, G3, Y1, Y3 and p defined in respect of compounds of formula (I) above also apply to compounds of formula (III).
Preferably X' represents X'-l.
(VIII)
wherein G1, G2, G3, D1, Y1, Y3 and p are as defined for a compound of formula (I);
or a salt or N-oxide thereof.
The preferred definitions of G1, G2, G3, D1, Y1, Y3 and p defined in respect of compounds of formula (I) above also apply to compounds of formula (III).
In a further aspect the invention provides a compound of formula IVa
wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined for a compound of formula I, or a salt or N-oxide thereof. The preferred definitions of G1, G2, G3, p, D1, Y1 and Y3 are as defined in respect of compounds of formula (I) above also apply to compounds of formula (IVa).
In one preferred group of compounds of formula IVa, Y6 is Ci-C6 alkyl, halogen, NH2, CrC6 haloalkyl, Ci-Cs haloalkoxy, or Ci-C6 haloalkoxy;
Y7 and Y8 independently of one another represent hydrogen, halogen, CN, N02, CrC8 alkyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy-Ci-C4-alkyl, C3-C8 cycloalkyi, C2-C8 alkenyl, C2- C8 alkynyl, phenyl, pyridyl, COR13, OR22, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, Ci-C8 alkylsulphonyl, N(R23)2, C02R22, 0(CO)R13, CON(R23)2, NR23COR13 or CR13N-OR22, wherein the alkyl, alkoxy, cycloalkyi, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy and d-C4 haloalkoxy;
G1 and G2 independently represent -C(R24)(R25)-;
G3 represents -C(R24)(R25)-, O, N(R26) or S;
each R24 and R25 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci- C4 haloalkyl, Ci-C4 alkoxy, or d-C4 haloalkyl;
R26 is hydrogen, OH, d-C4 alkyl or Ci-C4 alkoxy;
p is 0, 1 or 2. Preferably, p is 1 or 2.
In another group of preferred compounds of formula IVa, Y6 is hydrogen, methyl or halomethyl.
In another group of preferred compounds of formula IVa, Y6 is methyl or halomethyl.
In a further aspect the invention provides a compound of formula XVI.
wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined herein for a compound of formula I, and wherein R38 is hydrogen, CO-CH3, CO-CH2CH3, CO-CH2CH2CH3, CO-CH(CH3)2, CO-CF3, CO- CF2CF3, -CH3, -CH2CH3, -CH2CH2CH3, -CH(CH3)2, or a salt or N-oxide thereof.
The preferred definitions of G1, G2, G3, D1, Y1, Y3 and p defined in respect of compounds of formula (I) above also apply to compounds of formula (III).
In a further aspect the invention provides a process for the production of a compound of formula (I) comprising reacting a compound of formula (lib) with a compound of formula (X) as shown in scheme X
Scheme X
(lib) (I) wherein R represents halogen, -0-S02-R or group LG:
R represents Ci-C4 alkyl, d-C4 haloalkyl or phenyl, wherein the phenyl is optionally substituted by one or two substituents independently selected from methyl, trihalomethyl, N02, CN, C1-C7 alkoxycarbonyl; and
X, G1, G2, G3, Y1, Y3, Y4, Y6, D1, D2, D3, p and A are as defined for the compound of formula (I).
The preferred definitions of X, G1, G2, G3, Y1, Y3, Y4, Y6, D1, D2, D3, p and A defined in respect of compounds of formula (I) above also apply to compound (lib) and (X).
The compounds of formula (I) may exist as different geometric or optical isomers or in different tautomeric forms. These may be separated and isolated by well-known (usually chromatographic) techniques, and all such isomers and tautomers and mixtures thereof in all proportions as well as isotopic forms, such as deuterated compounds, are part of the present invention. In particular, the carbon-nitrogen double bonds of the compound of formula (I) allow the four cis/trans isomers shown below:
The present invention includes each of these isomers. The invention may provide a compound of formula (I) as just one of these isomers or as a mixture of one or more isomers in any ratio. Likewise, the invention also includes the corresponding isomers of the intermediates described herein, e.g. compounds (II), (III) and (VIII). In addition, where a reaction scheme depicts synthesis of one geometric isomer, the scheme also includes synthesis of the other geometric isomers where possible. For example Scheme X shown above encompasses the reactions:
The compounds in tables 1 to 7 illustrate compounds of formula (I).
Table X represents Table 1 (when X is 1), Table 2 (when X is 2), Table 3 (when X is 3), Table 4 (when X is 4), Table 5 (when X is 5), Table 6 (when X is 6), Table 7 (when X is 7).
x.08 CH3 H OCH2CH3 H -CH2CH2CH2- x.09 CH3 H CH(CH3)2 H -CH2CH2CH2- x. lO CH3 H cyclopropyl H -CH2CH2CH2- x. ll CH3 H SCH3 H -CH2CH2CH2-
X.12 CH3 H Phenyl H -CH2CH2CH2-
X.13 CH3 H 2-CH3-phenyl H -CH2CH2CH2-
X.14 CH3 H NH-CH2CH3 H -CH2CH2CH2-
X.15 CH3 H NH-(phenyl) H -CH2CH2CH2-
X.16 CH3 H NH-(3-CI-phenyl) H -CH2CH2CH2-
X.17 CH3 H CH3 CH3 -CH2CH2CH2-
X.18 CH3 H CH2CH3 CH2CH3 -CH2CH2CH2-
X.19 CH3 H OCH3 OCH3 -CH2CH2CH2- x.20 CH3 H OCH2CH3 OCH2CH3 -CH2CH2CH2- x.21 CH2CH3 H H H -CH2CH2CH2- x.22 CH2CH3 H CH3 H -CH2CH2CH2- x.23 CH2CH3 H CH2CH3 H -CH2CH2CH2- x.24 CH2CH3 H OCH3 H -CH2CH2CH2- x.25 CH2CH3 H OCH2CH3 H -CH2CH2CH2- x.26 CH2CH3 H CH(CH3)2 H -CH2CH2CH2- x.27 CH2CH3 H cyclopropyl H -CH2CH2CH2- x.28 CH2CH3 H SCH3 H -CH2CH2CH2- x.29 CH2CH3 H CH3 CH3 -CH2CH2CH2- x.30 OCH3 H H H -CH2CH2CH2- x.31 OCH3 H CH3 H -CH2CH2CH2- x.32 OCH3 H CH2CH3 H -CH2CH2CH2- x.33 OCH3 H OCH3 H -CH2CH2CH2- x.34 OCH3 H OCH2CH3 H -CH2CH2CH2- x.35 OCH3 H CH(CH3)2 H -CH2CH2CH2- x.36 OCH3 H cyclopropyl H -CH2CH2CH2- x.37 OCH3 H SCH3 H -CH2CH2CH2- x.38 OCH3 H CH3 CH3 -CH2CH2CH2- x.39 SCH3 H H H -CH2CH2CH2- x.40 SCH3 H CH3 H -CH2CH2CH2- x.41 SCH3 H CH2CH3 H -CH2CH2CH2- x.42 SCH3 H OCH3 H -CH2CH2CH2- x.43 SCH3 H OCH2CH3 H -CH2CH2CH2- x.44 SCH3 H CH(CH3)2 H -CH2CH2CH2- x.45 SCH3 H cyclopropyl H -CH2CH2CH2- x.46 SCH3 H SCH3 H -CH2CH2CH2- x.47 SCH3 H CH3 CH3 -CH2CH2CH2- x.48 phenyl H CH3 H -CH2CH2CH2- x.49 phenyl H CH2CH3 H -CH2CH2CH2- x.50 phenyl H OCH3 H -CH2CH2CH2- x.51 phenyl H CH3 CH3 -CH2CH2CH2- x.52 phenyl H phenyl H -CH2CH2CH2- x.53 2-CH3-phenyl H CH3 H -CH2CH2CH2- x.54 2-CH3-phenyl H CH3 CH3 -CH2CH2CH2- x.55 2-CH3-phenyl H 2-CH3-phenyl H -CH2CH2CH2-
x.56 NH-CH2CH3 H CH3 H -CH2CH2CH2- x.57 NH-CH2CH3 H CH3 CH3 -CH2CH2CH2- x.58 NH-CH2CH3 H NH-CH2CH3 H -CH2CH2CH2- x.59 NH-(phenyl) H CH3 H -CH2CH2CH2- x.60 NH-(phenyl) H CH3 CH3 -CH2CH2CH2- x.61 NH-(phenyl) H NH-(phenyl) H -CH2CH2CH2- x.62 NH-(3-CI-phenyl) H CH3 H -CH2CH2CH2- x.63 NH-(3-CI-phenyl) H CH3 CH3 -CH2CH2CH2- x.64 NH-(3-CI-phenyl) H NH-(3-CI-phenyl) H -CH2CH2CH2- x.65 H H H H -CH2-C(CH3)2-CH2- x.66 H H CH3 H -CH2-C(CH3)2-CH2- x.67 H H CH3 CH3 -CH2-C(CH3)2-CH2- x.68 CH3 H H H -CH2-C(CH3)2-CH2- x.69 CH3 H CH3 H -CH2-C(CH3)2-CH2- x.70 CH3 H CH2CH3 H -CH2-C(CH3)2-CH2- x.71 CH3 H OCH3 H -CH2-C(CH3)2-CH2- x.72 CH3 H OCH2CH3 H -CH2-C(CH3)2-CH2- x.73 CH3 H CH(CH3)2 H -CH2-C(CH3)2-CH2- x.74 CH3 H cyclopropyl H -CH2-C(CH3)2-CH2- x.75 CH3 H SCH3 H -CH2-C(CH3)2-CH2- x.76 CH3 H Phenyl H -CH2-C(CH3)2-CH2- x.77 CH3 H 2-CH3-phenyl H -CH2-C(CH3)2-CH2- x.78 CH3 H NH-CH2CH3 H -CH2-C(CH3)2-CH2- x.79 CH3 H NH-(phenyl) H -CH2-C(CH3)2-CH2- x.80 CH3 H NH-(3-CI-phenyl) H -CH2-C(CH3)2-CH2- x.81 CH3 H CH3 CH3 -CH2-C(CH3)2-CH2- x.82 CH3 H CH2CH3 CH2CH3 -CH2-C(CH3)2-CH2- x.83 CH3 H OCH3 OCH3 -CH2-C(CH3)2-CH2- x.84 CH3 H OCH2CH3 OCH2CH3 -CH2-C(CH3)2-CH2- x.85 CH2CH3 H H H -CH2-C(CH3)2-CH2- x.86 CH2CH3 H CH3 H -CH2-C(CH3)2-CH2- x.87 CH2CH3 H CH2CH3 H -CH2-C(CH3)2-CH2- x.88 CH2CH3 H OCH3 H -CH2-C(CH3)2-CH2- x.89 CH2CH3 H OCH2CH3 H -CH2-C(CH3)2-CH2- x.90 CH2CH3 H CH(CH3)2 H -CH2-C(CH3)2-CH2- x.91 CH2CH3 H cyclopropyl H -CH2-C(CH3)2-CH2- x.92 CH2CH3 H SCH3 H -CH2-C(CH3)2-CH2- x.93 CH2CH3 H CH3 CH3 -CH2-C(CH3)2-CH2- x.94 OCH3 H H H -CH2-C(CH3)2-CH2- x.95 OCH3 H CH3 H -CH2-C(CH3)2-CH2- x.96 OCH3 H CH2CH3 H -CH2-C(CH3)2-CH2- x.97 OCH3 H OCH3 H -CH2-C(CH3)2-CH2- x.98 OCH3 H OCH2CH3 H -CH2-C(CH3)2-CH2- x.99 OCH3 H CH(CH3)2 H -CH2-C(CH3)2-CH2-
X.100 OCH3 H cyclopropyl H -CH2-C(CH3)2-CH2- x. lOl OCH3 H SCH3 H -CH2-C(CH3)2-CH2- x.102 OCH3 H CH3 CH3 -CH2-C(CH3)2-CH2-
X.103 SCH3 H H H -CH2-C(CH3)2-CH2-
X.104 SCH3 H CH3 H -CH2-C(CH3)2-CH2-
X.105 SCH3 H CH2CH3 H -CH2-C(CH3)2-CH2- x.106 SCH3 H OCH3 H -CH2-C(CH3)2-CH2-
X.107 SCH3 H OCH2CH3 H -CH2-C(CH3)2-CH2- x.108 SCH3 H CH(CH3)2 H -CH2-C(CH3)2-CH2-
X.109 SCH3 H cyclopropyl H -CH2-C(CH3)2-CH2- x. llO SCH3 H SCH3 H -CH2-C(CH3)2-CH2- x. lll SCH3 H CH3 CH3 -CH2-C(CH3)2-CH2- x.112 phenyl H CH3 H -CH2-C(CH3)2-CH2- x.113 phenyl H CH2CH3 H -CH2-C(CH3)2-CH2- x.114 phenyl H OCH3 H -CH2-C(CH3)2-CH2- x.115 phenyl H CH3 CH3 -CH2-C(CH3)2-CH2- x.116 phenyl H phenyl H -CH2-C(CH3)2-CH2- x.117 2-CH3-phenyl H CH3 H -CH2-C(CH3)2-CH2- x.118 2-CH3-phenyl H CH3 CH3 -CH2-C(CH3)2-CH2- x.119 2-CH3-phenyl H 2-CH3-phenyl H -CH2-C(CH3)2-CH2- x.120 NH-CH2CH3 H CH3 H -CH2-C(CH3)2-CH2- x.121 NH-CH2CH3 H CH3 CH3 -CH2-C(CH3)2-CH2- x.122 NH-CH2CH3 H NH-CH2CH3 H -CH2-C(CH3)2-CH2- x.123 NH-(phenyl) H CH3 H -CH2-C(CH3)2-CH2-
X.124 NH-(phenyl) H CH3 CH3 -CH2-C(CH3)2-CH2- x.125 NH-(phenyl) H NH-(phenyl) H -CH2-C(CH3)2-CH2-
X.126 NH-(3-CI-phenyl) H CH3 H -CH2-C(CH3)2-CH2-
X.127 NH-(3-CI-phenyl) H CH3 CH3 -CH2-C(CH3)2-CH2-
X.128 NH-(3-CI-phenyl) H NH-(3-CI-phenyl) H -CH2-C(CH3)2-CH2- x.129 H H H H
X.130 H H CH3 H -C Hp -^ -^- C Hp -
X.131 H H CH3 CH3 -C Hp -^ -^- C Hp -
X.132 CH3 H H H -CHp-^_^--CHp- x.133 CH3 H CH3 H -C H2 -^^* C H2 ~ x.134 CH3 H CH2CH3 H -C Hp -^ -^- C Hp - x.135 CH3 H OCH3 H -CH -^_^--CH -
X.136 CH3 H OCH2CH3 H -CHp-^^^CHp- x.137 CH3 H CH(CH3)2 H -C Hp -^ -^- C Hp -
X.138 CH3 H cyclopropyl H -C H2 -^^* C H2 ~ x.139 CH3 H SCH3 H
X.140 CH3 H Phenyl H -CHp-^_^--CHp-
X.141 CH3 H 2-CH3-phenyl H -CHp-^^^CHp-
X.142 CH3 H NH-CH2CH3 H -C Hp -^ -^- C Hp -
X.143 CH3 H NH-(phenyl) H -C Hp -^ -^- C Hp -
X.144 CH3 H NH-(3-CI-phenyl) H -CHp-^^^CHp-
X.145 CH3 H CH3 CH3 -C H2 -^^* C H2 ~
X.146 CH3 H CH2CH3 CH2CH3 -C Hp -^ -^- C Hp -
X.147 CH3 H OCH3 OCH3 -CHp-^_^--CHp-
X.148 CH3 H OCH2CH3 OCH2CH3 -CHp-^^^CHp-
X.149 CH2CH3 H H H -C Hp -^ -^- C Hp -
X.150 CH2CH3 H CH3 H -C Hp -^ -^- C Hp -
X.151 CH2CH3 H CH2CH3 H -CHp-^^^CHp-
X.152 CH2CH3 H OCH3 H -C H2 -^^* C H2 ~ x.153 CH2CH3 H OCH2CH3 H -C Hp -^ -^- C Hp - x.154 CH2CH3 H CH(CH3)2 H -CHp-^_^--CHp- x.155 CH2CH3 H cyclopropyl H -CHp-^^^CHp-
X.177 phenyl H CH2CH3 H
X.178 phenyl H OCH3 H -CHp-^_^--CHp-
X.179 phenyl H CH3 CH3 -CHp-^^^CHp- x.180 phenyl H phenyl H -C Hp -^ -^- C Hp - x.181 2-CH3-phenyl H CH3 H -C Hp -^ -^- C Hp -
X.182 2-CH3-phenyl H CH3 CH3 -CHp-^^^CHp-
X.183 2-CH3-phenyl H 2-CH3-phenyl H -C H2 -^^* C H2 ~
X.184 NH-CH2CH3 H CH3 H -C Hp -^ -^- C Hp -
X.185 NH-CH2CH3 H CH3 CH3 -CHp-^_^--CHp- x.186 NH-CH2CH3 H NH-CH2CH3 H -CHp-^^^CHp-
X.187 NH-(phenyl) H CH3 H -C Hp -^ -^- C Hp - x.188 NH-(phenyl) H CH3 CH3 -C Hp -^ -^- C Hp -
X.189 NH-(phenyl) H NH-(phenyl) H -CHp-^^^CHp-
X.190 NH-(3-CI-phenyl) H CH3 H -C H2 -^^* C H2 ~
X.191 NH-(3-CI-phenyl) H CH3 CH3 -C Hp -^ -^- C Hp -
X.192 NH-(3-CI-phenyl) H NH-(3-CI-phenyl) H -CHp-^_^--CHp-
Table 1: This table discloses com ounds 1.01 to 1.192 of the formula (I-I)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table. Table 2: This table discloses com ounds 1.01 to 1.192 of the formula (I-II)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table. Table 3: This table discloses com ounds 1.01 to 1.192 of the formula (I-III)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table. Table 4: This table discloses com ounds 1.01 to 1.192 of the formula (I-IV)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table. Table 5: This table discloses com ounds 1.01 to 1.192 of the formula (I-V)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table.
Table 6: This table discloses compounds 1.01 to 1.192 of the formula (I-VI)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table.
Table 7: This table discloses com ounds 1.01 to 1.192 of the formula (I-VII)
Wherein Y1, Y3, Y4, Y6 and X have the specific meanings given in the table.
The compounds in Tables 1 to 7 include all isomers, tautomers and mixtures thereof, including the cis/trans isomers shown above.
The compounds of the invention may be made by a variety of methods, illustrated in schemes 1-8. The compounds depicted in the schemes also indicate any isomers and tautomers, in particular the geometric isomers arising from the oxime and oxime ether moieties.
Scheme 1
(I)
1) Compounds of formula (I) may be prepared by reacting a compound of formula (Ha), that is a compound of formula (II) wherein R is -ONH2, with a compound of formula (VI), wherein A, D2, D3, Y4 and Y6 are as defined herein for compounds of formula (I), and T1 and T2 are Ci-C8 alkoxy, or T1 and T2 together with the carbon they are attached to form a carbonyl group or an acetal or ketal function of the form C(0-Ci-C6-alkylidene-0) whereby the alkylidene fragment may optionally be mono- to tetra-substituted by d-C6 alkyl, as seen in scheme 1.
A general description of condensation reactions is given below, and typical reaction conditions for this type of reaction may be found in Journal of Organic Chemistry, 52(22), 4978- 84; 1987; Chemical & Pharmaceutical Bulletin, 51(2), 138-151; 2003; Organic Letters, 10(2), 285-288; 2008; Journal of the American Chemical Society, 130(12), 4196-4201; 2008;
Chemistry & Biology, 9(1), 113-129; 2002; Organic Preparations and Procedures International, 32(2), 153-159; 2000; Scientia Pharmaceutica, 66(1), 9-21; 1998, Journal of Medicinal
Chemistry, 49(17), 5177-5186; 2006, Journal of Agricultural and Food Chemistry, 38(3), 839-44; 1990; Tetrahedron: Asymmetry, 8(2), 253-263; 1997; Journal of Medicinal Chemistry, 44(21), 3339-3342; 2001; Bioorganic & Medicinal Chemistry Letters, 12(3), 341-344; 2002; US
2007032470; WO 07/058504; Journal of Organic Chemistry, 73(5), 2007-2010; 2008; Bioorganic & Medicinal Chemistry Letters, 19(10), 2683-2687; 2009; and Bioorganic & Medicinal Chemistry Letters, 19(10), 2654-2660; 2009.
2) Hydroxylamine derivatives of formula (Ila) may be made by reacting compounds of formula (IV), wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined herein for compounds of formula (I), and T1 and T2 are Ci-Cs alkoxy, or T1 and T2 together with the carbon they are attached to form a carbonyl group or an acetal or ketal function of the form C(0-Ci-C6- alkylidene-O) whereby the alkylidene fragment may optionally be mono- to tetra -substituted by Ci-C6 alkyl, with a bishydroxylamine derivative of formula (V), wherein X is as defined herein for a compound of formula (I) and R31 and R32 are either hydrogen or suitable protecting groups such as tert-butyloxycarbonyl (BOC), allyloxycarbonyl, fluorenylmethyloxycarbonyl (FMOC), formyl, acetyl, propionyl, trifluoroacetyl, benzoyl, substituted benzoyl, STABASE, Si(0-Ci-C8- alkyl)3, bis-Si(0-Ci-C8-alkyl)3, bis-benzyl, substituted bis-benzyl, bis-allyl, substituted bis-allyl, bis Ci-Cs-alkoxy-alkyl, N-phenylmethylene, substituted N-phenylmethylene, trityl, benzhydryl, substituted benzhydryl, or R31 and R32 together with the nitrogen atom to which they are attached may form a phthalyl group (scheme 1). General conditions for this type of condensation reaction can be found below.
When R31 and R32 are hydrogen, in order to optimize the yield of compound (Ila) an excess of intermediate (V) over intermediate (IV) may preferably be used. If R31 or R32 is not hydrogen, the hydroxylamine derivative may be deprotected using techniques well known to the person skilled in the art. Examples can be found in Greene, T. W., Wuts, P. G. N., Protective Groups in Organic Synthesis, John Wiley & Sons, Inc, 2006.
Monoprotection of bis-hydroxylamines has been described in Tetrahedron (1997), 53(15),
5485-5492. It is to be understood that methods used to obtain mono-protected diamines can be used in an analogous way to obtain mono-protected bis-hydroxylamine derivatives. Typical conditions for this type of reaction can be found in Synthetic Communications (2007), 37(5), 737-742; Organic Preparations and Procedures International (2009), 41(4), 301-307;
Tetrahedron: Asymmetry (2003), 14(11), 1559-1563; Bulletin of the Korean Chemical Society (1994), 15(12), 1025-7; Synthesis (1990), (4), 366-8; and Synthesis (1984), (12), 1032-3.
Bishydroxylamine derivatives are known in the literature. A description of their preparation can be found in WO 08/074418; Inorganic Chemistry Communications (2009), 12(3), 234-236; WO 99/49314; Synthesis (1997), (1), 38-40; and Gazzetta Chimica Italiana (1954), 84 915-20.
Scheme 2
(IV) (VII) (I)
3) Alternatively, as seen in scheme 2, compounds of formula (I) may be prepared by reacting a compound of formula (IV) and a compound of formula (VI) in the presence of a compound of formula (VII), wherein X is as defined herein for compounds of formula (I). Compounds of formula (IV) and (VI) are described under Scheme 1.
Typical reaction conditions for condensation reactions are seen below, and typical conditions for this particular condensation reaction are seen in the following references: Nature Chemical Biology, 5(6), 407-413; 2009; Acta Crystallographica, Section E: Structure Reports Online, E65(7), ol657; 2009; Acta Crystallographica, Section E: Structure Reports Online, E64(8), ol405, ol405/l-ol405/7; 2008; Acta Crystallographica, Section E: Structure Reports Online, E64(7), ol324, ol324/l-ol324/6; 2008; Acta Crystallographica, Section E: Structure Reports Online, E63(10), o4080, So4080/l-So4080/7; 2007; Synthetic Communications, 33(4), 543-546; 2003.
Scheme 3
4) Alternatively, compounds of formula (I) can be obtained by reacting a compound of formula (lib), that is a compound of formula (II) wherein R27 is a halogen, in particular chlorine, bromine or iodine, or a sulfonic acid ester group, such as mesylate, tosylate, triflate, a phenylsulfonic acid ester, a nitro-phenylsulfonic acid ester, a nonafluorobutylsulfonic acid ester or LG with a compound of formula (X), wherein A, D2, D3, Y4 and Y6 are as defined herein for compounds of formula (I) (scheme 3).
Typical reaction conditions for alkylation reactions such as this may be found below. These are further illustrated in Chinese Journal of Chemistry, 27(1), 33-42; 2009; WO 09/049846;
Journal of Antibiotics, 61(10), 603-614; 2008; Bioorganic & Medicinal Chemistry Letters, 18(24), 6471-6475; 2008; Journal of Medicinal Chemistry, 51(15), 4601-4608; 2008; WO 06/123145, Archiv der Pharmazie (Weinheim, Germany), 340(4), 202-208; 2007; Synthetic Communications, 37(7), 1155-1165; 2007; Russian Journal of Organic Chemistry, 42(5), 735-738; 2006;
Bioinorganic Chemistry and Applications, 1(3-4), 299-308; 2003; Synthetic Communications, 28(14), 2621-2633; 1998; Synthetic Communications, 19(18), 3129-38; 1989.
5) Compounds of formula (lib) may be obtained by reacting an oxime of formula (VIII) with a compound of formula (IX), wherein R27 is as defined herein for compounds of formula (lib) and R33 is a halogen, in particular chlorine, bromine or iodine, a sulfonic acid ester group, or the group LG (scheme 3). R27and R33 may be the same or different. Preferentially, R33 is a better
leaving group under the conditions of the reaction, such as tosylate or bromine when R is chlorine. Preferentially, an excess of the compound of formula (IX) relative to the oxime (VIII) would be used in the reaction, especially when R27 and R33 are the same.
Typical reaction conditions for alkylation reactions such as this can be found below, and are further illustrated in Journal of Agricultural and Food Chemistry (2008), 56(23), 11376-1139, Farmaco (2003), 58(9), 707-714; 1985; Journal of Heterocyclic Chemistry (1979), 16(7), 1459- 67; WO 08/074418; Journal of Medicinal Chemistry (2008), 51(20), 6421-6431; Synthetic Communications (2007), 37(7), 1155-1165; Bioorganic & Medicinal Chemistry (2007), 15(13), 4520-4527; Journal of Medicinal Chemistry (2006), 49(15), 4638-4649; and Synlett (2001), (Spec. Issue), 931-936.
Scheme 4
(IV)
6) Oximes of formula (VIII) may be obtained by a condensation reaction, whereby a compound of formula (IV), wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined herein for compounds of formula (I) and T1 and T2 are Ci-C8 alkoxy, or T1 and T2 together with the carbon they are attached to form a carbonyl group or an acetal or ketal function of the form C(0-Ci-C6- alkylidene-O) whereby the alkylidene fragment may optionally be mono- to tetra -substituted by Ci-C6-alkyl, is reacted with hydroxylamine, or, alternatively, with a salt of hydroxylamine. A more detailed description of condensation processes is given below.
Related references include the following: Journal of Heterocyclic Chemistry, 46(1), 116-118; 2009; Journal of Medicinal Chemistry, 20(5), 718-21; 1977; Journal of Organic Chemistry, 73(11), 4017-4026; 2008; EJEAFChe, Electronic Journal of Environmental, Agricultural and Food Chemistry, 5(5), 1515-1521; 2006; Advanced Synthesis & Catalysis, 346(13-15), 1798-1811; 2004.
Some compounds of formula (IV) are known and their preparation has been published or they are available commercially. A few typical examples are given in Table 8 together with the
corresponding CAS numbers. Analogous protocols to those used to prepare the following compounds can be used to prepare other compounds of formula (IV).
Table 8
1196151-83-3 31170-78-2 41043-15-6
9568-10-7
14428-47-8 01622-35-9
N H2
0 o 0
122910-29-6 844891-39-0
149194-86-5
N H2
89967-17-9
O
857613-10-6
149194-90-1
1105664-58-1 1105662-77-8
Scheme 5
7) Alternatively, oximes of formula (VIII) can be obtained by a nitrosation reaction of compounds of formula (XI), wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined herein for compounds of formula (I), with base and an alkyl nitrite, as seen in scheme 5. Typical bases include lithium diisopropyl amide (LDA), lithium hexamethyldisilazane, n-butyl lithium, s-butyl lithium, tert-butyl lithium, sodium tert-butylate or potassium tert-butylate . Typical alkyl nitrites include isopentyl nitrite and tert-butyl nitrite. The compound of formula (XI), the alkyl nitrite or the base can be used in different stoichiometric amounts, with each reagent possibly being in excess with respect to the others. Preferentially, such reactions are carried out under nonaqueous conditions in an inert solvent such as hexane, heptanes, cyclohexane, toluene or ethers such as THF or tert-butyl methyl ether. The reaction may be performed at temperatures ranging from -80 to 250°C, preferably between -50 and 120°C.
Such reactions can lead to a mixture of the E- and the Z-oxime (ether) product, or the product may also be exclusively either the E- or the Z-oxime (ether).
A large number of these types of transformations are known in the art. Typical reaction conditions for this type of reaction may be found in Crawford, Jason B.; Chen, Gang; Gauthier, David; Wilson, Trevor; Carpenter, Bryon; Baird, Ian R.; McEachern, Ernie; Kaller, Alan; Harwig, Curtis; Atsma, Bern; Skerlj, Renato T.; Bridger, Gary 1, Organic Process Research &
Development (2008), 12(5), 823-830, McEachern, E. 1; Yang, W.; Chen, G.; Skerlj, R. T.;
Bridger, G. 1, Synthetic Communications (2003), 33(20), 3497-350; and Bark, Thomas;
Thummel, Randolph P., Inorganic Chemistry (2005), 44(24), 8733-8739.
Scheme 6
8) An alternative route to compounds of formula (I) is shown in Scheme 6. As in scheme 3, the compound of formula (I) is obtained by the reacting a compound of formula (lib) with a compound of formula (X) as an alkylation reaction. Typical conditions for this type of reaction are described below.
9) The compounds of (lib) can be formed by reacting a hydroxylamine derivative of formula (XIII), wherein R27 halogen, with a compound of formula (IV), as seen in scheme 6.
Compounds of formula (IV) are described above.
Typical reaction conditions for this type of condensation reaction may be found below, and are further illustrated in Angewandte Chemie, International Edition (2006), 45(32), 5307-5311.
10) Compounds of formula (XIII) can be made by alkylating a hydroxylamine derivative of formula (XII), wherein R34 and R35, either independently of each other, or together with each other and the nitrogen atom to which they are attached, are protecting groups, such as tert-
butoxy carbonyl, acetyl, benzyl, or phthalyl, with the alkylating agent (IX), wherein R is halogen and R33 is halogen, in particular chloro, bromo or iodo, a sulfonic acid ester group, or LG (scheme 6). Typical conditions for such an alkylation reaction may be found below. The protecting groups or group can then be removed using techniques well known to a person skilled in the art, examples of which can be found in Greene, T. W., Wuts, P. G. N., Protective Groups in Organic Synthesis, John Wiley & Sons, Inc, 2006.
Scheme 7
(la)
11) Compounds of formula (la), that is compounds of formula I wherein X is X-3, X-4 or X-5 wherein Z3, Z6, Z7, Z10, Z11 and Z12 are as defined herein for compounds of formula (I)
(represented in scheme 7 as X'), i.e.:
6 7 10 11 1 ?
#—∑—# #—∑—∑—# #— z— z— z— #
X'-1 X'-2 X'-3
and Z4, Z8 and Z13 represent C(OH)R30 and Z5, Z9 and Z14 represent CHR29 wherein R29 and R30 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci-C4 haloalkyl,
phenyl or CN, wherein phenyl is optionally substituted by one or more groups, e.g. one to five groups, independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkyoxy, can be made by reacting compounds of formula (III) with compounds of formula (X) (scheme 7).
Typical conditions for an alkylation such as this are described below, and a further illustrated in Synthesis, (13), 2055-2064; 2008; Russian Journal of Organic Chemistry, 43(2), 181-183; 2007; Russian Journal of Organic Chemistry 43(3), 449-453; 2007; and Journal of Molecular Catalysis B: Enzymatic, 11(4-6), 255-263; 2001.
Compounds of formula (la) are especially useful as intermediates to a number of other compounds, wherein the hydroxy group formed by the opening of the epoxide is transformed into other functional groups, for example carbonyl, fluorine or chlorine. Such transformations can be effected using a number of conditions well known to the person skilled in the art.
12) Compounds of formula (III) can be obtained by the alkylation of oximes of formula (VIII) with epoxides of formula (XIV) wherein X', R29 and R30 are as defined above for compounds of formula (III) and R36 is halogen. Such alkylation processes are described in more detail below.
Relevant references include the following: Synthetic Communications, 37(7), 1155-1165; 2007; Molecules, 10(7), 747-754; 2005; Molecules, 10(11), 1399-1408; 2005; European Journal of Medicinal Chemistry, 40(12), 1351-1358; 2005; Organic Preparations and Procedures International, 30(2), 195-202; 1998; WO 08/074418; and Journal fuer Praktische
Chemie/Chemiker-Zeitung (1993), 335(7), 623-7.
A large number of compounds of formula (XIV) are commercially available or their preparation is to be found in the literature. Commercially available compounds (XIV) include epichlorohydrin, 2(S)-epichlorohydrin, 2(R)-epichlorohydrin, 2-methyl-epichlorohydrin, epibromohydrin.
Scheme 8
13) Compounds of formula (I) may be formed from compounds of formula (lid), that is a compound of formula (II) wherein R27 represents C-2B, as seen in scheme 8. This can be done by displacing the group X" with a suitable derivative of the group A or the fragment R37 using one of several techniques well known to the person skilled in the art, including coupling reactions such as Suzuki (Suzuki-Miyaura) couplings and Stille couplings.
The Suzuki coupling comprises the reaction between an organoboron compound, such as the boronic acid derivative of A or R37 or their esters and a halide of formula (lid) to give compounds of formula (I).
The reaction may be done in the presence of a palladium catalyst such as Pd(PPh3)4, Pd(OAc)2, Pd(dppf)CI2 and a base such as Na2C03, Ba(OH)2, K3P04, Cs2C03, K2C03, KF, NaOH or alkali alcoholates, such as potassium tert-butoxide or sodium ethoxide. Typical catalyst loadings are in the range of 0.01 to 10 mol%. Preferred solvents for such cross coupling reactions include ethers such as THF or dimethoxyethane, acetonitrile, DMF, NMP, benzene or toluene or a mixture of such solvents. Such solvents can also be used together with water. The preferred temperature range for carrying out such reactions is between 0°C and 180°C.
The Stille coupling comprises the use of an organotin compound, such as the tributylstannane derivative of A or R37 and a halide of formula (lid) to give compounds of formula (I).
The reaction can be done in the presence of a palladium catalyst such as Pd(PPh3)4, Pd2(dba)3.CHCI3 with or with an added ligand such as P(2-furyl)3 or Pd(OAc)2, Pd(dppf)CI2,
Pd(MeCN)2CI2. Typical catalyst loadings are in the range of 0.01 to 10 mol%. Preferred solvents for such cross coupling reactions include ethers such as THF or dimethoxyethane, acetonitrile, DMF, NMP, benzene or toluene. Such solvents can also be used together with water. The preferred temperature range for carrying out such reactions is between 0°C and 180°C.
Typical reaction conditions for these types of reaction may be found in Bioorganic and
Medicinal Chemistry Letters 19(18), 5339-5345; 2009; Canadian Journal of Chemistry, 85(11), 913-922; 2007; Journal of Organic Chemistry, 72(13), 4892-4899; 2007; Tetrahedron Letters, 43(40), 7189-7191; 2002; Synlett, (10), 1557-1558; 2001; EP 792870; WO 95/20569.
14) Compounds of formula (lid) can be formed from compounds of formula (lie), that is a compound of formula (II) wherein R27 represents C-l, by the action of a halogenating agent.
The reaction can be performed using an excess of either the halogenating agent, equimolar amounts of halogenating agent and the hydroxamic acid ester (XV), or with an excess of the hydroxamic acid ester (lie). Preferentially it is carried out with an excess of halogenating agent over the hydroxamic acid ester (lie).
Typical halogenating agents include CCI4 or CBr4 along with and a phosphine such as triphenyl phosphine or tributylphosphine. Other typical halogenating agents include Et2NSF3, (MeOCH2CH2)2NSF3 (Deoxo-Fluor), morpholinotrifluorosulfurane and SF4, SOCI2, COCI2, PCI5, PCI3, PBr3 or POCI3, or a mixture of PCI5 and POCI3. Typical conditions include the use of a sub- stoichiometric, equimolar or excess amount of PCI5 in POCI3 relative to the compound of formula (lie), where POCI3 itself may be present in an equimolar amount or alternatively, be used in a sub-stoichiometric amount or excess relative to the compound of formula (lie).
The halogenation of hydroxamic acid ester of formula (lie) can be done without a solvent in certain cases or, preferentially, in the presence of a solvent or mixture of solvents. Any organic solvent that is inert under the specific reaction conditions can be chosen. Preferred solvents include the following, without limiting the selection: aliphatic or aromatic hydrocarbons that may optionally be substituted by one or several halogen atoms such as pentane, hexanes, heptanes, cyclohexane, petroleum ether, benzene, toluene, xylene, chlorobenzene, dichlorobenzenes, dichloromethane, chloroform, 1,2-dichloroethane, carbon tetrachloride, ethers such as
diethylether, diisopropyl ether, dibutyl ether, tert-butyl methyl ether, tetrahydrofuran, 1,4- dioxane, dimethoxyethane, triethylene glycol dimethyl ether (methyltriglyme), or acetonitrile, propionitrile, benzonitrile or a substituted benzonitrile.
The use of catalysts to facilitate this type of reaction is established, comprising the use of catalysts such as dimethylformamide, diethylformamide and formylpiperidine. The
transformation can also be done without a reaction catalyst.
Depending on the properties of the starting materials the reaction temperature can be varied over a wide range. Typical reaction temperatures vary between - 100°C and 250°C. Preferentially, the temperature range is between 0°C and 100°C. On some occasions, the reaction may be carried out under reflux.
This transformation can also optionally be carried out under ultrasonication.
The use of organic bases in this type of reaction is well exemplified in the literature. The amount of base can be stoichiometric, sub- or super-stoichiometric. Typically an excess of base is used. Typical bases include the following without limiting the selction: Triethylamine, tripropylamine, tributylamine, di-isopropyl-ethylamine, Ν,Ν-dimethyl-cyclohexylamine, N-methyl- dicyclohexylamine, Ν,Ν-dimethyl-aniline, N,N-d iethyl-aniline, Ν,Ν-dimethyl-benzylamine, N,N- diethyl-benzylamine, pyridine, 2-methyl-pyridine, 3-methyl-pyridine, 4-methyl-pyridine, 2,6- dimethyl-pyridine, 2,4,6-trimethyl-pyridine, 4-dimethylamino-pyridine, N-methyl-piperidine, N- ethyl-piperidine, N-methyl-morpholine, N-ethyl-morpholine, Ν,Ν'-dimethyl-piperazine. The transformation can also be done in the absence of bases
Typical reaction protocols for this type of reaction may be found in Bioorganic & Medicinal Chemistry Letters (2009), 19(18), 5339-5345; Journal of Medicinal Chemistry, 50(14), 3314- 3321; 2007; Journal of Organic Chemistry, 69(8), 2741-2749; 2004; WO 01/025206; Australian Journal of Chemistry, 52(8), 807-811; 1999; Indian Journal of Chemistry, Section B: Organic Chemistry Including Medicinal Chemistry, 23B(8), 728-32; 1984 .
15) Compounds of formula (lie) can be formed by reacting compounds of formula (lib) with a hydroxamic acid derivative of formula (XV) wherein R37 is as defined herein for compounds of formula (lid). This transformation is an alkylation of the hydroxamic acid derivative (XV) or its salt generated in situ by the action of a base. Typical conditions for alkylation reaction such as this are seen below, and are also described in the following references: WO 09/036020; Journal of the Chemical Society, Perkin Transactions 2, (10), 1728- 1739; 2002; Journal of Medicinal Chemistry, 42(1), 153-163; 1999; Journal of Medicinal Chemistry, 34(1), 57-65; 1991; Synthetic Communications, 19(3-4), 339-44; 1989; Journal of
Organic Chemistry, 54(14), 3394-403; 1989; Tetrahedron, 43(11), 2577-92; 1987; Journal of Organic Chemistry, 51(26), 5047-50; 1986.
Scheme 9
(IVa1)
16) Ketones of formula (IVa7), that is a compound of formula (IVa) wherein D1 is N, may be obtained by acidic hydrolysis methods, well known to the person skilled in the art from compounds of formula (XVIa), that is a compound of formula (XVI) wherein D1 is N.
17) Enolethers of formula (XVIa) can be obtained by reacting enaminones of formula (XVIII) wherein G1, G2, G3 and p are as described herein for compounds of formula (I) and R38 represents hydrogen, CO-CH3, CO-CH2CH3, CO-CH2CH2CH3, CO-CH(CH3)2, CO-CF3, CO-CF2CF3, CH3, CH2CH3, CH2CH2CH3 or CH(CH3)2 with amidines or guanidines of formula (XVII) wherein Y1 is as described herein for compounds of formula (I). Such condensation reactions can be performed using a number of conditions well known to the person skilled in the art. Such reactions are carried out in the presence of a base preferentially sodium or potassium alcoholate in ethanol or methanol. Specific reaction conditions may be found in Synthesis, 1012-1018, 1996; J. Heterocycl. Chem. 20, 649-653, 1983 or in WO2009010488.
18) Enaminones of formula (XVIII) can be obtained by reacting ketone of formula (XX) wherein G1, G2, G3 and p are as described herein for compounds of formula (I) and R38 represents hydrogen, CO-CH3, CO-CH2CH3, CO-CH2CH2CH3, CO-CH(CH3)2, CO-CF3, CO-CF2CF3, CH3, CH2CH3, CH2CH2CH3 or CH(CH3)2 with Ν,Ν-dimethylformamide dialkyl acetal of formula (XIX) wherein R39 represents d-C4 alkyl. Such condensation reactions are well known to the person skilled in the art. The reaction is carried out in an inert solvent or without solvent at temperatures ranging from 0°C to 250°C preferably between 50 and 160°C. Specific reaction conditions may be found in Synthesis, (97), 3397, 1964; Synthesis, (97), 3407, 1964; Journal of Med. Chem., Vol. 52, No. 16, 5152-5163; Tetrahedron Lett, (27), 2567, 1986; Tetrahedron Lett, (50), 2255-2264, 1994; Synthetic Commun., (28), 10, 1743-1753, 1998 or in Chem. Ber., (104), 2975, 1971.
19) Enol ethers of formula (XX) can be obtained by reacting ketones of formula (XXII) wherein G1, G2, G3 and p are as described herein for compounds of formula (I) with an alcohol of formula (XXI) wherein R38 represents hydrogen, CO-CH3, CO-CH2CH3, CO-CH2CH2CH3, CO- CH(CH3)2, CO-CF3, CO-CF2CF3, CH3, CH2CH3, CH2CH2CH3 or CH(CH3)2. Such reactions can be affected using a number of conditions well known to the person skilled in the art. Specific reaction conditions may be found in WO2004104007.
Typical conditions for condensation reactions:
This applies to procedures 1, 2, 3, 6 and 9.
Different stoichiometric set-ups may be used for these reactions, depending on the properties of reactants and product. An excess of the electrophile, the nudeophile, or equimolar amounts may be chosen. Preferentially equimolar amounts of electrophilic and nucleophilic compounds are used.
The reaction may be performed in the presence or absence of an inert organic or inorganic solvent, or in the presence of a mixture of such solvents. Preferentially, it is performed in the presence of one or more solvents. Preferred solvents include the following aliphatic or aromatic hydrocarbons, which may optionally be substituted by one or more halogen atoms, such as pentane, hexanes, heptanes, cyclohexane, petroleum ether, benzene, toluene, xylene, chlorobenzene, dichlorobenzenes, dichloromethane, chloroform, 1,2-dichloroethane or carbon tetrachloride, ethers such as diethylether, diisopropyl ether, tert-butyl methyl ether,
tetrahydrofuran, 1,4-dioxane, dimethoxyethane or diglycol dimethyl ether, ketones such as acetone, methyl ethyl ketone, methyl isopropyl ketone or methyl isobutyl ketone, acids and ester such as acetic acid, ethyl acetate or methyl acetate, aprotic polar solvents such as
acetonitrile, pripionitril, dimethyl formamide, dimethyl acetamide, N-methyl-pyrrolidone, dimethyl sulfoxide, sulfolane, DMPU, or pyridine and picolines. The selection of solvents includes water and alcohols such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol, tert- butanol, pentanol, isopentanol, hexanol, trifluorethanol, ethylene glycol or methoxyethanol.
The reaction may be performed between -20°C and 250°C, preferentially between 0°C and
100°C. In some cases the reaction mixture may be heated to reflux.
Where appropriate, compounds can be used in the form of the free compound, or, alternatively, they can be used in the form of a salt such as the acetate, trifluoroacetate, propionate, benzoate, oxalate, methylsolfonate, phenylsulfonate, p-tolylsulfonate,
trifluormethylsulfonate, fluoride, chloride, bromide, iodide, sulphate, hydrogensulphate or nitrate, including bis-salts if appropriate.
The reaction can be carried out in the absence of an acid using the free compounds.
Alternatively, the reaction may be performed in the presence of an acid in catalytic,
stoichiometric or excess amounts. Acids that could be used include acetic acid, propionic acid, oxalic acid, trifluoroacetic acid, hydrochloric acid, hydrobromic acid, hydroiodic acid, methansulfonic acid, para-toluenesulfonic acid, sulphuric acid, sodium hydrogensulphate and phosphoric acid. The reaction can optionally be carried out in a water-free solvent system in the presence of a drying agent, such as sodium or magnesium sulphate, potassium carbonate or molecular sieves.
If the two substituents at the carbon atom of the oxime or oxime ether function are different from each other, the condensation reaction can lead to a mixture of the E- and the Z- oxime (ether) product. The condensation product may also be exclusively either the E- or the Z- oxime (ether).
Condensations can be performed under reduced pressure, normal pressure or increased pressure. Preferentially the reaction is performed under normal pressure.
Typical conditions for alkylation reactions:
This applies to procedures 4, 5, 8, 10, 11, 12 and 15.
Different stoichiometric set-ups may be used for these reactions, depending on the properties of reactants and product. An excess of the electrophile, the nucleophile, or neither may be chosen. Usually, it is preferable that equimolar amounts of electrophilic and nucleophilic compounds are used.
The reaction may be performed in the absence or presence of a solvent or a mixture of solvents. Preferential solvents include the following aliphatic or aromatic hydrocarbons that may
optionally be substituted by one or more halogen atoms such as pentane, hexanes, heptanes, cyclohexane, petroleum ether, benzene, toluene, xylene, chlorobenzene, dichlorobenzenes, dichloromethane, chloroform, 1,2-dichloroethanev or carbon tetrachloride, ethers such as diethyl ether, diisopropyl ether, tert-butyl-methyl ether, tetrahydrofuran, 1,4-dioxane, dimethoxyethane or diglycol dimethyl ether, ketones such as acetone, methyl ethyl ketone, methyl isopropyl ketone or methyl isobutyl ketone, acids and ester such as acetic acid, ethyl acetate or methyl acetate, aprotic polar solvents such as acetonitrile, pripionitrile, dimethyl formamide, dimethyl acetamide, N-methyl-pyrrolidone, dimethyl sulfoxide, sulfolane, DMPU, or pyridine and picolines. The selction of solvents includes also water and alcohols such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol, tert-butanol, pentanol, isopentanol, hexanol, trifluorethanol, ethylene glycol or methoxyethanol.
The reaction may be performed in a biphasic system comprising an organic solvent that is not miscible with water, such as toluene, dichloromethane, dichloro-ethylene, and an aqueous solvent, such as water. Such a reaction would be performed in the presence of a phase-transfer catalyst, such as tetra-n-butylammonium bromide (TBAB), Tetradecyldimethylbenzylammonium chloride (TDMBAC), N-Benzyltrimethylammonium hydroxide, along with aqueous sodium or potassium hydroxide in stoichiometric amounts. The biphasic reaction may be performed with or without ultrasonication.
The reaction may be carried out at temperatures varying from -100°C and 250°C.
Preferentially, the temperature range is between 0°C and 100°C.
Optionally, an organic or inorganic base may be present such as alkali- and earth alkali acetates, amides, carbonates, hydrogencarbonates, hydrides, hydroxides or alcoholates such as sodium, potassium, caesium or calcium acetate, sodium, potassium, caesium or calcium carbonate, sodium, potassium, caesium or calcium hydrogencarbonate, sodium, potassium, caesium or calcium hydride, sodium, potassium, caesium or calcium amide, sodium, potassium, caesium or calcium hydroxide, sodium, potassium, caesium or calcium methanolate, sodium, potassium, caesium or calcium ethanolate, sodium, potassium, caesium or calcium n-, i-, s- or t- butanolate, triethylamine, tripropylamine, tributylamine, di-isopropyl-ethylamine, N,N-dimethyl- cyclohexylamine, N-methyl-dicyclohexylamine, N,N-dimethyl-aniline, Ν,Ν-diethyl-aniline, N,N- dimethyl-benzylamine, Ν,Ν-diethyl-benzylamine, pyridine, 2-methyl-pyridine, 3-methyl-pyridine, 4-methyl-pyridine, 2,6-dimethyl-pyridine, 2,4,6-trimethyl-pyridine, 4-dimethylamino-pyridine, N- methyl-piperidine, N-ethyl-piperidine, N-methyl-morpholine, N-ethyl-morpholine, N,N'-dimethyl- piperazine, l,4-Diazabicyclo[2.2.2]octane (DABCO), l,8-Diaza-7-bicyclo[5.4.0]undecene (DBU),
l,5-Diazabicyclo[4.3.0]non-5-ene (DBN), l-tert-Butyl-2,2,2-tri(l-pyrrolidinyl)phosphazene (BTPP), l-tert-Butyl-2,2,2-tris(dimethylamino)phosphazene, sodium hexamethyldisilazane, potassium hexamethyldisilazane, lithium diisopropylamide, ethyl magnesium chloride, isopropylmagnesium chloride.
The alkylation can be performed under reduced pressure, normal pressure or increased pressure. Preferentially the reaction is performed under normal pressure.
The products of steps 1) to 19) may be required to be purified using, for example, chromatography, crystallisation or other purification techniques well known to the person skilled in the art.
The compounds of formula (I) to formula (XXII) and, where appropriate, the tautomers thereof, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form.
It has now been found that the compounds of formula (I) according to the invention have, for practical purposes, a very advantageous spectrum of activities for protecting useful plants against diseases that are caused by phytopathogenic microorganisams, such as fungi, bacteria or viruses.
The invention therefore also relates to a method of controlling or preventing infestation of useful plants by phytopathogenic microorganisms, wherein a compound of formula (I) is applied as active ingredient to the plants, to parts thereof or the locus thereof. The compounds of formula (I) according to the invention are distinguished by excellent activity at low rates of application, by being well tolerated by plants and by being environmentally safe. They have very useful curative, preventive and systemic properties and are used for protecting numerous useful plants. The compounds of formula (I) can be used to inhibit or destroy the diseases that occur on plants or parts of plants (fruit, blossoms, leaves, stems, tubers, roots) of different crops of useful plants, while at the same time protecting also those parts of the plants that grow later e.g. from phytopathogenic microorganisms.
It is also possible to use compounds of formula (I) as dressing agents for the treatment of plant propagation material, in particular of seeds (fruit, tubers, grains) and plant cuttings (e.g. rice), for the protection against fungal infections as well as against phytopathogenic fungi occurring in the soil.
Furthermore the compounds of formula (I) according to the invention may be used for controlling fungi in related areas, for example in the protection of technical materials, including wood and wood related technical products, in food storage or in hygiene management.
The compounds of formula (I) are, for example, effective against the phytopathogenic fungi of the following classes: Fungi imperfecti (e.g. Botrytis, Pyricularia, Helminthosporium,
Fusarium, Septoria, Cercospora and Alternaria) and Basidiomycetes (e.g. Rhizoctonia, Hemileia, Puccinia). Additionally, they are also effective against the Ascomycetes classes (e.g. Venturia and Erysiphe, Podosphaera, Monilinia, Uncinula) and of the Oomycetes classes (e.g.
Phytophthora, Pythium, Plasmopara). Within the scope of the invention, useful plants to be protected typically comprise the following species of plants: cereal (wheat, barley, rye, oat, rice, maize, sorghum and related species); beet (sugar beet and fodder beet); pomes, drupes and soft fruit (apples, pears, plums, peaches, almonds, cherries, strawberries, raspberries and blackberries); leguminous plants (beans, lentils, peas, soybeans); oil plants (rape, mustard, poppy, olives, sunflowers, coconut, castor oil plants, cocoa beans, groundnuts); cucumber plants (pumpkins, cucumbers, melons); fibre plants (cotton, flax, hemp, jute); citrus fruit
(oranges, lemons, grapefruit, mandarins); vegetables (spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes, paprika); lauraceae (avocado, cinnamomum, camphor) or plants such as tobacco, nuts, coffee, eggplants, sugar cane, tea, pepper, vines, hops, bananas and natural rubber plants, as well as ornamentals.
The term "useful plants" is to be understood as including also useful plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides (such as, for example, HPPD inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, EPSPS (5-enol-pyrovyl-shikimate-3-phosphate-synthase) inhibitors, GS (glutamine synthetase) inhibitors or PPO (protoporphyrinogen-oxidase) inhibitors) as a result of conventional methods of breeding or genetic engineering. An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding (mutagenesis) is
Clearfield® summer rape (Canola). Examples of crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names
RoundupReady® , Herculex I® and LibertyLink®.
The term "useful plants" is to be understood as including also useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of
synthesising one or more selectively acting toxins, such as are known, for example, from toxin- producing bacteria, especially those of the genus Bacillus.
Examples of such plants are: YieldGard® (maize variety that expresses a CrylA(b) toxin); YieldGard Rootworm® (maize variety that expresses a CrylllB(bl) toxin); YieldGard Plus® (maize variety that expresses a CrylA(b) and a CrylllB(bl) toxin); Starlink® (maize variety that expresses a Cry9(c) toxin); Herculex I® (maize variety that expresses a CryIF(a2) toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylA(c) toxin);
Bollgard I® (cotton variety that expresses a CrylA(c) toxin); Bollgard II® (cotton variety that expresses a CrylA(c) and a CryllA(b) toxin); VIPCOT® (cotton variety that expresses a VIP toxin); NewLeaf® (potato variety that expresses a CrylllA toxin); NatureGard® Agrisure® GT Advantage (GA21 glyphosate-tolerant trait), Agrisure® CB Advantage (Btll corn borer (CB) trait), Agrisure® RW (corn rootworm trait) and Protecta®.
The term "useful plants" is to be understood as including also useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising antipathogenic substances having a selective action, such as, for example, the so- called "pathogenesis-related proteins" (PRPs, see e.g. EP-A-0 392 225). Examples of such antipathogenic substances and transgenic plants capable of synthesising such antipathogenic substances are known, for example, from EP-A-0 392 225, WO 95/33818, and EP-A-0 353 191. The methods of producing such transgenic plants are generally known to the person skilled in the art and are described, for example, in the publications mentioned above.
The term "locus" of a useful plant as used herein is intended to embrace the place on which the useful plants are growing, where the plant propagation materials of the useful plants are sown or where the plant propagation materials of the useful plants will be placed into the soil. An example for such a locus is a field, on which crop plants are growing.
The term "plant propagation material" is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes. There may be mentioned for example seeds (in the strict sense), roots, fruits, tubers, bulbs, rhizomes and parts of plants. Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants may be protected before transplantation by a total or partial treatment by immersion. Preferably "plant propagation material" is understood to denote seeds.
The compounds of formula (I) can be used in unmodified form or, preferably, together with carriers and adjuvants conventionally employed in the art of formulation.
Therefore the invention also relates to compositions for controlling and protecting against phytopathogenic microorganisms, comprising a compound of formula (I) and an inert carrier, and to a method of controlling or preventing infestation of useful plants by phytopathogenic microorganisms, wherein a composition, comprising a compound of formula (I) as acitve ingredient and an inert carrier, is applied to the plants, to parts thereof or the locus thereof.
To this end compounds of formula (I) and inert carriers are conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g. in polymeric substances. As with the type of the compositions, the methods of application, such as spraying, atomising, dusting, scattering, coating or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances. The compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
Suitable carriers and adjuvants (auxiliaries) can be solid or liquid and are substances useful in formulation technology, e.g. natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders or fertilizers. Such carriers are for example described in WO 97/33890.
The compounds of formula (I) or compositions, comprising a compound of formula (I) as active ingredient and an inert carrier, can be applied to the locus of the plant or plant to be treated, simultaneously or in succession with further compounds. These further compounds can be e.g. fertilizers or micronutrient donors or other preparations which influence the growth of plants. They can also be selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
A preferred method of applying a compound of formula (I), or a composition, comprising a compound of formula (I) as active ingredient and an inert carrier, is foliar application. The frequency of application and the rate of application will depend on the risk of infestation by the corresponding pathogen. However, the compounds of formula (I) may also penetrate the plant through the roots via the soil (systemic action) by drenching the locus of the plant with a liquid
formulation, or by applying the compounds in solid form to the soil, e.g. in granular form (soil application). In crops of water rice such granulates can be applied to the flooded rice field. The compounds of formula (I) may also be applied to seeds (coating) by impregnating the seeds or tubers either with a liquid formulation of the fungicide or coating them with a solid formulation.
A formulation, i.e. a composition comprising the compound of formula (I) and, if desired, a solid or liquid adjuvant, is prepared in a known manner, typically by intimately mixing and/or grinding the compound with extenders, for example solvents, solid carriers and, optionally, surface-active compounds (surfactants).
The agrochemical formulations will usually contain from 0.1 to 99% by weight, preferably from 0.1 to 95% by weight, of the compound of formula (I), 99.9 to 1% by weight, preferably 99.8 to 5% by weight, of a solid or liquid adjuvant, and from 0 to 25% by weight, preferably from 0.1 to 25% by weight, of a surfactant.
Whereas it is preferred to formulate commercial products as concentrates, the end user will normally use dilute formulations.
Advantageous rates of application are normally from 5g to 2kg of active ingredient (a.i.) per hectare (ha), preferably from lOg to 1kg a.i. /ha, most preferably from 20g to 600g a.i./ha. When used as seed drenching agent, convenient rates of application are from lOmg to lg of active substance per kg of seeds. The rate of application for the desired action can be determined by experiments. It depends for example on the type of action, the developmental stage of the useful plant, and on the the application (location, timing, application method) and can, owing to these parameters, vary within wide limits.
The compounds of formula (I), or a pharmaceutical salt thereof, described above may also have an advantageous spectrum of activity for the treatment and/or prevention of microbial infection in an animal. "Animal" can be any animal, for example, insect, mammal, reptile, fish, amphibian, preferably mammal, most preferably human. "Treatment" means the use on an animal which has microbial infection in order to reduce or slow or stop the increase or spread of the infection, or to reduce the infection or to cure the infection. "Prevention" means the use on an animal which has no apparent signs of microbial infection in order to prevent any future infection, or to reduce or slow the increase or spread of any future infection.
According to the present invention there is provided the use of a compound of formula (I) in the manufacture of a medicament for use in the treatment and/or prevention of microbial infection in an animal. There is also provided the use of a compound of formula (I) as a pharmaceutical agent. There is also provided the use of a compound of formula (I) as an
antimicrobial agent in the treatment of an animal. According to the present invention there is also provided a pharmaceutical composition comprising as an active ingredient a compound of formula (I), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable diluent or carrier. This composition can be used for the treatment and/or prevention of antimicrobial infection in an animal. This pharmaceutical composition can be in a form suitable for oral administration, such as tablet, lozenges, hard capsules, aqueous suspensions, oily suspensions, emulsions dispersible powders, dispersible granules, syrups and elixirs.
Alternatively this pharmaceutical composition can be in a form suitable for topical application, such as a spray, a cream or lotion. Alternatively this pharmaceutical composition can be in a form suitable for parenteral administration, for example injection. Alternatively this
pharmaceutical composition can be in inhalable form, such as an aerosol spray.
The compounds of formula (I) may be effective against various microbial species able to cause a microbial infection in an animal. Examples of such microbial species are those causing Aspergillosis such as Aspergillus fumigatus, A. flavus, A. terms, A. nidulans and A. niger, those causing Blastomycosis such as Blastomyces dermatitidis; those causing Candidiasis such as Candida albicans, C. glabrata, C. tropicalis, C. parapsilosis, C. krusei and C. lusitaniae; those causing Coccidioidomycosis such as Coccidioides immitis; those causing Cryptococcosis such as Cryptococcus neoformans; those causing Histoplasmosis such as Histoplasma capsulatum and those causing Zygomycosis such as Absidia corymbifera, Rhizomucor pusillus and Rhizopus arrhizus. Further examples are Fusarium Spp such as Fusarium oxysporum and Fusarium soiani and Scedosporium Spp such as Scedosporium apiospermum and Scedosporium prolificans. Still further examples are Microsporum Spp, Trichophyton Spp, Epidermophyton Spp, Mucor Spp, Sporothorix Spp, Phialophora Spp, Cladosporium Spp, Petriellidium spp, Paracoccidioides Spp and Histoplasma Spp.
The compositions of this invention may contain other compounds having biological activity, for example micronutrients or compounds having fungicidal activity or which possess plant growth regulating, herbicidal, insecticidal, nematicidal or acaricidal activity.
The compound of formula I (herein after abbreviated by the term "TX" thus means a compound encompassed by the compounds of formula I, or preferably the term "TX" refers to a compound selected from the Tables 1-7 or Table 9) may be the sole active ingredient of the composition or it may be admixed with one or more additional active ingredients such as a pesticide (insect, acarine, mollusc and nematode pesticide), fungicide, synergist, herbicide, safener or plant growth regulator where appropriate. The activity of the compositions according
to the invention may thereby be broadened considerably and may have surprising advantages which can also be described, in a wider sense, as synergistic activity. An additional active ingredient may: provide a composition having a broader spectrum of activity or increased persistence at a locus; provide a composition demonstrating better plant/crop tolerance by reducing phytotoxicity; provide a composition controlling insects in their different development stages; synergise the activity or complement the activity (for example by increasing the speed of effect or overcoming repellency) of the TX; or help to overcome or prevent the development of resistance to individual components. The particular additional active ingredient will depend upon the intended utility of the composition. Examples of suitable pesticides include the following:
a) Pyrethroids, such as permethrin, cypermethrin, fenvalerate, esfenvalerate, deltamethrin, cyhalothrin (in particular lambda-cyhalothrin), bifenthrin, fenpropathrin, cyfluthrin, tefluthrin, fish safe pyrethroids (for example ethofenprox), natural pyrethrin, tetramethrin, s-bioallethrin, fenfluthrin, prallethrin or 5-benzyl-3-furylmethyl-(E)-(lR,3S)-2,2-dimethyl- 3-(2-oxothiolan-3-ylidenemethyl)cyclopropane carboxylate;
b) Organophosphates, such as, profenofos, sulprofos, acephate, methyl parathion, azinphos-methyl, demeton-s-methyl, heptenophos, thiometon, fenamiphos, monocrotophos, profenofos, triazophos, methamidophos, dimethoate, phosphamidon, malathion, chlorpyrifos, phosalone, terbufos, fensulfothion, fonofos, phorate, phoxim, pirimiphos-methyl,
pirimiphos-ethyl, fenitrothion, fosthiazate or diazinon;
c) Carbamates (including aryl carbamates), such as pirimicarb, triazamate, cloethocarb, carbofuran, furathiocarb, ethiofencarb, aldicarb, thiofurox, carbosulfan, bendiocarb, fenobucarb, propoxur, methomyl or oxamyl;
d) Benzoyl ureas, such as diflubenzuron, triflumuron, hexaflumuron, flufenoxuron or chlorfluazuron;
e) Organic tin compounds, such as cyhexatin, fenbutatin oxide or azocyclotin;
f) Pyrazoles, such as tebufenpyrad and fenpyroximate;
g) Macrolides, such as avermectins or milbemycins, for example abamectin, emamectin benzoate, ivermectin, milbemycin, or spinosad, spinetoram or azadirachtin;
h) Hormones or pheromones;
i) Organochlorine compounds such as endosulfan, benzene hexachloride, DDT, chlordane or dieldrin;
j) Amidines, such as chlordimeform or amitraz;
k) Fumigant agents, such as chloropicrin, dichloropropane, methyl bromide or metam;
I) Neonicotinoid compounds such as imidacloprid, thiacloprid, acetamiprid, clothianidin, nitenpyram, dinotefuran or thiamethoxam;
m) Diacyl hydrazines, such as tebufenozide, chromafenozide or methoxyfenozide;
n) Diphenyl ethers, such as diofenolan or pyriproxifen;
o) Indoxacarb;
p) Chlorfenapyr;
q) Pymetrozine or pyrifluquinazon;
r) Spirotetramat, spirodiclofen or spiromesifen;
s) Flubendiamide, chloranthraliniprole, or cyanthraniliprole;
t) Cyenopyrafen or cyflumetofen; or
u) Sulfoxaflor.
In addition to the major chemical classes of pesticide listed above, other pesticides having particular targets may be employed in the composition, if appropriate for the intended utility of the composition. For instance, selective insecticides for particular crops, for example stemborer specific insecticides (such as cartap) or hopper specific insecticides (such as buprofezin) for use in rice may be employed. Alternatively insecticides or acaricides specific for particular insect species/stages may also be included in the compositions (for example acaricidal ovo-larvicides, such as clofentezine, flubenzimine, hexythiazox or tetradifon; acaricidal motilicides, such as dicofol or propargite; acaricides, such as bromopropylate or chlorobenzilate; or growth regulators, such as hydramethylnon, cyromazine, methoprene, chlorfluazuron or diflubenzuron).
The following mixtures of the compounds of formula I with active ingredients are preferred, wherein, preferably, the term "TX" refers to a compound covered by the compounds of formula I or preferably the term "TX" refers to a compound selected from the Tables 1 to 9 and the following List shows specific examples of mixtures comprising the component TX and the component (B):
an adjuvant selected from the group of substances consisting of petroleum oils (alternative name) (628) + TX,
an acaricide selected from the group of substances consisting of l,l-bis(4-chlorophenyl)-2- ethoxyethanol (IUPAC name) (910) + TX, 2,4-dichlorophenyl benzenesulfonate (IUPAC/Chemical Abstracts name) (1059) + TX, 2-fluoro-/V-methyl-/V-l-naphthylacetamide (IUPAC name) (1295) + TX, 4-chlorophenyl phenyl sulfone (IUPAC name) (981) + TX, abamectin (1) + TX, acequinocyl (3) + TX, acetoprole [CCN] + TX, acrinathrin (9) + TX, aldicarb (16) + TX,
aldoxycarb (863) + TX, alpha-cypermethrin (202) + TX, amidithion (870) + TX, amidoflumet [CCN] + TX, amidothioate (872) + TX, amiton (875) + TX, amiton hydrogen oxalate (875) + TX, amitraz (24) + TX, aramite (881) + TX, arsenous oxide (882) + TX, AVI 382 (compound code) + TX, AZ 60541 (compound code) + TX, azinphos-ethyl (44) + TX, azinphos-methyl (45) + TX, azobenzene (IUPAC name) (888) + TX, azocyclotin (46) + TX, azothoate (889) + TX, benomyl (62) + TX, benoxafos (alternative name) [CCN] + TX, benzoximate (71) + TX, benzyl benzoate (IUPAC name) [CCN] + TX, bifenazate (74) + TX, bifenthrin (76) + TX, binapacryl (907) + TX, brofenvalerate (alternative name) + TX, bromocyclen (918) + TX, bromophos (920) + TX, bromophos-ethyl (921) + TX, bromopropylate (94) + TX, buprofezin (99) + TX, butocarboxim (103) + TX, butoxycarboxim (104) + TX, butylpyridaben (alternative name) + TX, calcium polysulfide (IUPAC name) (111) + TX, camphechlor (941) + TX, carbanolate (943) + TX, carbaryl (115) + TX, carbofuran (118) + TX, carbophenothion (947) + TX, CGA 50'439
(development code) (125) + TX, chinomethionat (126) + TX, chlorbenside (959) + TX, chlordimeform (964) + TX, chlordimeform hydrochloride (964) + TX, chlorfenapyr (130) + TX, chlorfenethol (968) + TX, chlorfenson (970) + TX, chlorfensulphide (971) + TX, chlorfenvinphos (131) + TX, chlorobenzilate (975) + TX, chloromebuform (977) + TX, chloromethiuron (978) + TX, chloropropylate (983) + TX, chlorpyrifos (145) + TX, chlorpyrifos-methyl (146) + TX, chlorthiophos (994) + TX, cinerin I (696) + TX, cinerin II (696) + TX, cinerins (696) + TX, clofentezine (158) + TX, closantel (alternative name) [CCN] + TX, coumaphos (174) + TX, crotamiton (alternative name) [CCN] + TX, crotoxyphos (1010) + TX, cufraneb (1013) + TX, cyanthoate (1020) + TX, cyflumetofen (CAS Reg. No.: 400882-07-7) + TX, cyhalothrin (196) + TX, cyhexatin (199) + TX, cypermethrin (201) + TX, DCPM (1032) + TX, DDT (219) + TX, demephion (1037) + TX, demephion-0 (1037) + TX, demephion-S (1037) + TX, demeton (1038) + TX, demeton-methyl (224) + TX, demeton-0 (1038) + TX, demeton-O-methyl (224) + TX, demeton-S (1038) + TX, demeton-S-methyl (224) + TX, demeton-S-methylsulphon (1039) + TX, diafenthiuron (226) + TX, dialifos (1042) + TX, diazinon (227) + TX, dichlofluanid (230) + TX, dichlorvos (236) + TX, dicliphos (alternative name) + TX, dicofol (242) + TX, dicrotophos (243) + TX, dienochlor (1071) + TX, dimefox (1081) + TX, dimethoate (262) + TX, dinactin (alternative name) (653) + TX, dinex (1089) + TX, dinex-diclexine (1089) + TX, dinobuton (269) + TX, dinocap (270) + TX, dinocap-4 [CCN] + TX, dinocap-6 [CCN] + TX, dinocton (1090) + TX, dinopenton (1092) + TX, dinosulfon (1097) + TX, dinoterbon (1098) + TX, dioxathion (1102) + TX, diphenyl sulfone (IUPAC name) (1103) + TX, disulfiram (alternative name) [CCN] + TX, disulfoton (278) + TX, DNOC (282) + TX, dofenapyn (1113) + TX, doramectin
(alternative name) [CCN] + TX, endosulfan (294) + TX, endothion (1121) + TX, EPN (297) + TX, eprinomectin (alternative name) [CCN] + TX, ethion (309) + TX, ethoate-methyl (1134) + TX, etoxazole (320) + TX, etrimfos (1142) + TX, fenazaflor (1147) + TX, fenazaquin (328) + TX, fenbutatin oxide (330) + TX, fenothiocarb (337) + TX, fenpropathrin (342) + TX, fenpyrad (alternative name) + TX, fenpyroximate (345) + TX, fenson (1157) + TX, fentrifanil (1161) + TX, fenvalerate (349) + TX, fipronil (354) + TX, fluacrypyrim (360) + TX, fluazuron (1166) + TX, flubenzimine (1167) + TX, flucycloxuron (366) + TX, flucythrinate (367) + TX, fluenetil (1169) + TX, flufenoxuron (370) + TX, flumethrin (372) + TX, fluorbenside (1174) + TX, fluvalinate (1184) + TX, FMC 1137 (development code) (1185) + TX, formetanate (405) + TX, formetanate hydrochloride (405) + TX, formothion (1192) + TX, formparanate (1193) + TX, gamma-HCH (430) + TX, glyodin (1205) + TX, halfenprox (424) + TX, heptenophos (432) + TX, hexadecyl cyclopropanecarboxylate (IUPAC/Chemical Abstracts name) (1216) + TX, hexythiazox (441) + TX, iodomethane (IUPAC name) (542) + TX, isocarbophos (alternative name) (473) + TX, isopropyl 0-(methoxyaminothiophosphoryl)salicylate (IUPAC name) (473) + TX, ivermectin (alternative name) [CCN] + TX, jasmolin I (696) + TX, jasmolin II (696) + TX, jodfenphos (1248) + TX, lindane (430) + TX, lufenuron (490) + TX, malathion (492) + TX, malonoben (1254) + TX, mecarbam (502) + TX, mephosfolan (1261) + TX, mesulfen
(alternative name) [CCN] + TX, methacrifos (1266) + TX, methamidophos (527) + TX, methidathion (529) + TX, methiocarb (530) + TX, methomyl (531) + TX, methyl bromide (537) + TX, metolcarb (550) + TX, mevinphos (556) + TX, mexacarbate (1290) + TX, milbemectin (557) + TX, milbemycin oxime (alternative name) [CCN] + TX, mipafox (1293) + TX, monocrotophos (561) + TX, morphothion (1300) + TX, moxidectin (alternative name) [CCN] + TX, naled (567) + TX, NC-184 (compound code) + TX, NC-512 (compound code) + TX, nifluridide (1309) + TX, nikkomycins (alternative name) [CCN] + TX, nitrilacarb (1313) + TX, nitrilacarb 1:1 zinc chloride complex (1313) + TX, NNI-0101 (compound code) + TX, NNI-0250 (compound code) + TX, omethoate (594) + TX, oxamyl (602) + TX, oxydeprofos (1324) + TX, oxydisulfoton (1325) + TX, pp'-DDT (219) + TX, parathion (615) + TX, permethrin (626) + TX, petroleum oils (alternative name) (628) + TX, phenkapton (1330) + TX, phenthoate (631) + TX, phorate (636) + TX, phosalone (637) + TX, phosfolan (1338) + TX, phosmet (638) + TX, phosphamidon (639) + TX, phoxim (642) + TX, pirimiphos-methyl (652) + TX,
polychloroterpenes (traditional name) (1347) + TX, polynactins (alternative name) (653) + TX, proclonol (1350) + TX, profenofos (662) + TX, promacyl (1354) + TX, propargite (671) + TX, propetamphos (673) + TX, propoxur (678) + TX, prothidathion (1360) + TX, prothoate (1362)
+ TX, pyrethrin I (696) + TX, pyrethrin II (696) + TX, pyrethrins (696) + TX, pyridaben (699) + TX, pyridaphenthion (701) + TX, pyrimidifen (706) + TX, pyrimitate (1370) + TX, quinalphos (711) + TX, quintiofos (1381) + TX, R-1492 (development code) (1382) + TX, RA-17
(development code) (1383) + TX, rotenone (722) + TX, schradan (1389) + TX, sebufos (alternative name) + TX, selamectin (alternative name) [CCN] + TX, SI-0009 (compound code) + TX, sophamide (1402) + TX, spirodiclofen (738) + TX, spiromesifen (739) + TX, SSI-121 (development code) (1404) + TX, sulfiram (alternative name) [CCN] + TX, sulfluramid (750) + TX, sulfotep (753) + TX, sulphur (754) + TX, SZI-121 (development code) (757) + TX, tau- fluvalinate (398) + TX, tebufenpyrad (763) + TX, TEPP (1417) + TX, terbam (alternative name) + TX, tetrachlorvinphos (777) + TX, tetradifon (786) + TX, tetranactin (alternative name) (653) + TX, tetrasul (1425) + TX, thiafenox (alternative name) + TX, thiocarboxime (1431) + TX, thiofanox (800) + TX, thiometon (801) + TX, thioquinox (1436) + TX, thuringiensin (alternative name) [CCN] + TX, triamiphos (1441) + TX, triarathene (1443) + TX, triazophos (820) + TX, triazuron (alternative name) + TX, trichlorfon (824) + TX, trifenofos (1455) + TX, trinactin (alternative name) (653) + TX, vamidothion (847) + TX, vaniliprole [CCN] and YI-5302
(compound code) + TX,
an algicide selected from the group of substances consisting of bethoxazin [CCN] + TX, copper dioctanoate (IUPAC name) (170) + TX, copper sulfate (172) + TX, cybutryne [CCN] + TX, dichlone (1052) + TX, dichlorophen (232) + TX, endothal (295) + TX, fentin (347) + TX, hydrated lime [CCN] + TX, nabam (566) + TX, quinoclamine (714) + TX, quinonamid (1379) + TX, simazine (730) + TX, triphenyltin acetate (IUPAC name) (347) and triphenyltin hydroxide (IUPAC name) (347) + TX,
an anthelmintic selected from the group of substances consisting of abamectin (1) + TX, crufomate (1011) + TX, doramectin (alternative name) [CCN] + TX, emamectin (291) + TX, emamectin benzoate (291) + TX, eprinomectin (alternative name) [CCN] + TX, ivermectin (alternative name) [CCN] + TX, milbemycin oxime (alternative name) [CCN] + TX, moxidectin (alternative name) [CCN] + TX, piperazine [CCN] + TX, selamectin (alternative name) [CCN] + TX, spinosad (737) and thiophanate (1435) + TX,
an avicide selected from the group of substances consisting of chloralose (127) + TX, endrin (1122) + TX, fenthion (346) + TX, pyridin-4-amine (IUPAC name) (23) and strychnine (745) + TX,
a bactericide selected from the group of substances consisting of l-hydroxy-lH-pyridine-2- thione (IUPAC name) (1222) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (IUPAC name)
(748) + TX, 8-hydroxyquinoline sulfate (446) + TX, bronopol (97) + TX, copper dioctanoate (IUPAC name) (170) + TX, copper hydroxide (IUPAC name) (169) + TX, cresol [CCN] + TX, dichlorophen (232) + TX, dipyrithione (1105) + TX, dodicin (1112) + TX, fenaminosulf (1144) + TX, formaldehyde (404) + TX, hydrargaphen (alternative name) [CCN] + TX, kasugamycin (483) + TX, kasugamycin hydrochloride hydrate (483) + TX, nickel bis(dimethyldithiocarbamate) (IUPAC name) (1308) + TX, nitrapyrin (580) + TX, octhilinone (590) + TX, oxolinic acid (606) + TX, oxytetracycline (611) + TX, potassium hydroxyquinoline sulfate (446) + TX, probenazole (658) + TX, streptomycin (744) + TX, streptomycin sesquisulfate (744) + TX, tecloftalam (766) + TX, and thiomersal (alternative name) [CCN] + TX,
a biological agent selected from the group of substances consisting of Adoxophyes orana GV (alternative name) (12) + TX, Agrobacterium radiobacter (alternative name) (13) + TX, Amblyseius spp. (alternative name) (19) + TX, Anagrapha falcifera NPV (alternative name) (28) + TX, Anagrus atomus (alternative name) (29) + TX, Aphelinus abdominalis (alternative name) (33) + TX, Aphidius colemani (alternative name) (34) + TX, Aphidoletes aphidimyza (alternative name) (35) + TX, Autographa californica NPV (alternative name) (38) + TX, Bacillus firmus (alternative name) (48) + TX, Bacillus sphaericus Neide (scientific name) (49) + TX, Bacillus thuringiensis Berliner (scientific name) (51) + TX, Bacillus thuringiensis subsp. aizawai (scientific name) (51) + TX, Bacillus thuringiensis subsp. israelensis (scientific name) (51) + TX, Bacillus thuringiensis subsp. japonensis (scientific name) (51) + TX, Bacillus thuringiensis subsp.
kurstaki (scientific name) (51) + TX, Bacillus thuringiensis subsp. tenebrionis (scientific name) (51) + TX, Beauveria bassiana (alternative name) (53) + TX, Beauveria brongniartii (alternative name) (54) + TX, Chrysoperla carnea (alternative name) (151) + TX, Cryptolaemus
montrouzieri (alternative name) (178) + TX, Cydia pomoneiia GV (alternative name) (191) + TX, Dacnusa sibirica (alternative name) (212) + TX, Diglyphus isaea (alternative name) (254) + TX, Encarsia formosa (scientific name) (293) + TX, Eretmocerus eremicus (alternative name) (300) + TX, Helicoverpa zea NPV (alternative name) (431) + TX, Heterorhabditis bacteriophora and H. megidis (alternative name) (433) + TX, Hippodamia con vergens (alternative name) (442) + TX, Leptomastix dactylopii (alternative name) (488) + TX, Macrolophus caliginosus (alternative name) (491) + TX, Mamestra brassicae NPV (alternative name) (494) + TX, Metaphycus helvolus (alternative name) (522) + TX, Metarhizium anisopliae var. acridum (scientific name) (523) + TX, Metarhizium anisopliae var. anisopliae (scientific name) (523) + TX, Neodiprion sertifer NPV and N. lecontei NPV (alternative name) (575) + TX, Orius spp. (alternative name) (596) + TX, Paecilomyces fumosoroseus (alternative name) (613) + TX, Phytoseiulus persimilis
(alternative name) (644) + TX, Spodoptera exigua multicapsid nuclear polyhedrosis virus (scientific name) (741) + TX, Steinernema bibionis (alternative name) (742) + TX, Steinernema carpocapsae (alternative name) (742) + TX, Steinernema feltiae (alternative name) (742) + TX, Steinernema glaseri (alternative name) (742) + TX, Steinernema riobrave (alternative name) (742) + TX, Steinernema riobravis (alternative name) (742) + TX, Steinernema scapterisci (alternative name) (742) + TX, Steinernema spp. (alternative name) (742) + TX, Trichogramma spp. (alternative name) (826) + TX, Typhlodromus occidentalis (alternative name) (844) and Verticillium lecanii (alternative name) (848) + TX,
a soil sterilant selected from the group of substances consisting of iodomethane (IUPAC name) (542) and methyl bromide (537) + TX,
a chemosterilant selected from the group of substances consisting of apholate [CCN] + TX, bisazir (alternative name) [CCN] + TX, busulfan (alternative name) [CCN] + TX, diflubenzuron (250) + TX, dimatif (alternative name) [CCN] + TX, hemel [CCN] + TX, hempa [CCN] + TX, metepa [CCN] + TX, methiotepa [CCN] + TX, methyl apholate [CCN] + TX, morzid [CCN] + TX, penfluron (alternative name) [CCN] + TX, tepa [CCN] + TX, thiohempa (alternative name) [CCN] + TX, thiotepa (alternative name) [CCN] + TX, tretamine (alternative name) [CCN] and uredepa (alternative name) [CCN] + TX,
an insect pheromone selected from the group of substances consisting of (£)-dec-5-en-l-yl acetate with (£)-dec-5-en-l-ol (IUPAC name) (222) + TX, (£)-tridec-4-en-l-yl acetate (IUPAC name) (829) + TX, (£)-6-methylhept-2-en-4-ol (IUPAC name) (541) + TX, (E,Z)-tetradeca-4, 10- dien-l-yl acetate (IUPAC name) (779) + TX, (Z)-dodec-7-en-l-yl acetate (IUPAC name) (285) + TX, (Z)-hexadec-ll-enal (IUPAC name) (436) + TX, (Z)-hexadec-ll-en-l-yl acetate (IUPAC name) (437) + TX, (Z)-hexadec-13-en-ll-yn-l-yl acetate (IUPAC name) (438) + TX, (Z)-icos- 13-en-10-one (IUPAC name) (448) + TX, (Z)-tetradec-7-en-l-al (IUPAC name) (782) + TX, (Z)- tetradec-9-en-l-ol (IUPAC name) (783) + TX, (Z)-tetradec-9-en-l-yl acetate (IUPAC name) (784) + TX, (7E,9Z)-dodeca-7,9-dien-l-yl acetate (IUPAC name) (283) + TX, (9Z,11£> tetradeca-9,ll-dien-l-yl acetate (IUPAC name) (780) + TX, (9Z,12E)-tetradeca-9,12-dien-l-yl acetate (IUPAC name) (781) + TX, 14-methyloctadec-l-ene (IUPAC name) (545) + TX, 4- methylnonan-5-ol with 4-methylnonan-5-one (IUPAC name) (544) + TX, alpha-multistriatin (alternative name) [CCN] + TX, brevicomin (alternative name) [CCN] + TX, codlelure
(alternative name) [CCN] + TX, codlemone (alternative name) (167) + TX, cuelure (alternative name) (179) + TX, disparlure (277) + TX, dodec-8-en-l-yl acetate (IUPAC name) (286) + TX, dodec-9-en-l-yl acetate (IUPAC name) (287) + TX, dodeca-8 + TX, 10-dien-l-yl acetate (IUPAC
name) (284) + TX, dominicalure (alternative name) [CCN] + TX, ethyl 4-methyloctanoate (IUPAC name) (317) + TX, eugenol (alternative name) [CCN] + TX, frontalin (alternative name) [CCN] + TX, gossyplure (alternative name) (420) + TX, grandlure (421) + TX, grandlure I (alternative name) (421) + TX, grandlure II (alternative name) (421) + TX, grandlure III (alternative name) (421) + TX, grandlure IV (alternative name) (421) + TX, hexalure [CCN] + TX, ipsdienol (alternative name) [CCN] + TX, ipsenol (alternative name) [CCN] + TX, japonilure (alternative name) (481) + TX, lineatin (alternative name) [CCN] + TX, litlure (alternative name) [CCN] + TX, looplure (alternative name) [CCN] + TX, medlure [CCN] + TX, megatomoic acid (alternative name) [CCN] + TX, methyl eugenol (alternative name) (540) + TX, muscalure (563) + TX, octadeca-2,13-dien-l-yl acetate (IUPAC name) (588) + TX, octadeca-3,13-dien-l-yl acetate (IUPAC name) (589) + TX, orfralure (alternative name) [CCN] + TX, oryctalure
(alternative name) (317) + TX, ostramone (alternative name) [CCN] + TX, siglure [CCN] + TX, sordidin (alternative name) (736) + TX, sulcatol (alternative name) [CCN] + TX, tetradec-ll-en- 1-yl acetate (IUPAC name) (785) + TX, trimedlure (839) + TX, trimedlure A (alternative name) (839) + TX, trimedlure Bi (alternative name) (839) + TX, trimedlure B2 (alternative name) (839) + TX, trimedlure C (alternative name) (839) and trunc-call (alternative name) [CCN] + TX, an insect repellent selected from the group of substances consisting of 2-(octylthio)ethanol (IUPAC name) (591) + TX, butopyronoxyl (933) + TX, butoxy(polypropylene glycol) (936) + TX, dibutyl adipate (IUPAC name) (1046) + TX, dibutyl phthalate (1047) + TX, dibutyl succinate (IUPAC name) (1048) + TX, diethyltoluamide [CCN] + TX, dimethyl carbate [CCN] + TX, dimethyl phthalate [CCN] + TX, ethyl hexanediol (1137) + TX, hexamide [CCN] + TX, methoquin-butyl (1276) + TX, methylneodecanamide [CCN] + TX, oxamate [CCN] and picaridin [CCN] + TX,
an insecticide selected from the group of substances consisting of 1-dichloro-l-nitroethane (IUPAC/Chemical Abstracts name) (1058) + TX, l,l-dichloro-2,2-bis(4-ethylphenyl)ethane (IUPAC name) (1056), + TX, 1,2-dichloropropane (IUPAC/Chemical Abstracts name) (1062) + TX, 1,2-dichloropropane with 1,3-dichloropropene (IUPAC name) (1063) + TX, l-bromo-2- chloroethane (IUPAC/Chemical Abstracts name) (916) + TX, 2,2,2-trichloro-l-(3,4-dichloro- phenyl)ethyl acetate (IUPAC name) (1451) + TX, 2,2-dichlorovinyl 2-ethylsulphinylethyl methyl phosphate (IUPAC name) (1066) + TX, 2-(l,3-dithiolan-2-yl)phenyl dimethylcarbamate (IUPAC/ Chemical Abstracts name) (1109) + TX, 2-(2-butoxyethoxy)ethyl thiocyanate (IUPAC/Chemical Abstracts name) (935) + TX, 2-(4,5-dimethyl-l,3-dioxolan-2-yl)phenyl methylcarbamate (IUPAC/ Chemical Abstracts name) (1084) + TX, 2-(4-chloro-3,5-xylyloxy)ethanol (IUPAC name)
(986) + TX, 2-chlorovinyl diethyl phosphate (IUPAC name) (984) + TX, 2-imidazolidone (IUPAC name) (1225) + TX, 2-isovalerylindan-l,3-dione (IUPAC name) (1246) + TX, 2-methyl(prop-2- ynyl)aminophenyl methylcarbamate (IUPAC name) (1284) + TX, 2-thiocyanatoethyl laurate (IUPAC name) (1433) + TX, 3-bromo-l-chloroprop-l-ene (IUPAC name) (917) + TX, 3-methyl- l-phenylpyrazol-5-yl dimethylcarbamate (IUPAC name) (1283) + TX, 4-methyl(prop-2- ynyl)amino-3,5-xylyl methylcarbamate (IUPAC name) (1285) + TX, 5,5-dimethyl-3-oxocyclohex- 1-enyl dimethylcarbamate (IUPAC name) (1085) + TX, abamectin (1) + TX, acephate (2) + TX, acetamiprid (4) + TX, acethion (alternative name) [CCN] + TX, acetoprole [CCN] + TX, acrinathrin (9) + TX, acrylonitrile (IUPAC name) (861) + TX, alanycarb (15) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, aldrin (864) + TX, allethrin (17) + TX, allosamidin (alternative name) [CCN] + TX, allyxycarb (866) + TX, alpha-cypermethrin (202) + TX, alpha-ecdysone (alternative name) [CCN] + TX, aluminium phosphide (640) + TX, amidithion (870) + TX, amidothioate (872) + TX, aminocarb (873) + TX, amiton (875) + TX, amiton hydrogen oxalate (875) + TX, amitraz (24) + TX, anabasine (877) + TX, athidathion (883) + TX, AVI 382 (compound code) + TX, AZ 60541 (compound code) + TX, azadirachtin (alternative name) (41) + TX, azamethiphos (42) + TX, azinphos-ethyl (44) + TX, azinphos-methyl (45) + TX, azothoate (889) + TX, Bacillus thuringiensis delta endotoxins (alternative name) (52) + TX, barium hexafluorosilicate (alternative name) [CCN] + TX, barium polysulfide (IUPAC/Chemical Abstracts name) (892) + TX, barthrin [CCN] + TX, Bayer 22/190 (development code) (893) + TX, Bayer 22408 (development code) (894) + TX, bendiocarb (58) + TX, benfuracarb (60) + TX, bensultap (66) + TX, beta-cyfluthrin (194) + TX, beta-cypermethrin (203) + TX, bifenthrin (76) + TX, bioallethrin (78) + TX, bioallethrin S-cyclopentenyl isomer (alternative name) (79) + TX, bioethanomethrin [CCN] + TX, biopermethrin (908) + TX, bioresmethrin (80) + TX, bis(2- chloroethyl) ether (IUPAC name) (909) + TX, bistrifluron (83) + TX, borax (86) + TX, brofenvalerate (alternative name) + TX, bromfenvinfos (914) + TX, bromocyclen (918) + TX, bromo-DDT (alternative name) [CCN] + TX, bromophos (920) + TX, bromophos-ethyl (921) + TX, bufencarb (924) + TX, buprofezin (99) + TX, butacarb (926) + TX, butathiofos (927) + TX, butocarboxim (103) + TX, butonate (932) + TX, butoxycarboxim (104) + TX, butylpyridaben (alternative name) + TX, cadusafos (109) + TX, calcium arsenate [CCN] + TX, calcium cyanide (444) + TX, calcium polysulfide (IUPAC name) (111) + TX, camphechlor (941) + TX, carbanolate (943) + TX, carbaryl (115) + TX, carbofuran (118) + TX, carbon disulfide
(IUPAC/Chemical Abstracts name) (945) + TX, carbon tetrachloride (IUPAC name) (946) + TX, carbophenothion (947) + TX, carbosulfan (119) + TX, cartap (123) + TX, cartap hydrochloride
(123) + TX, cevadine (alternative name) (725) + TX, chlorbicyclen (960) + TX, chlordane (128) + TX, chlordecone (963) + TX, chlordimeform (964) + TX, chlordimeform hydrochloride (964) + TX, chlorethoxyfos (129) + TX, chlorfenapyr (130) + TX, chlorfenvinphos (131) + TX, chlorfluazuron (132) + TX, chlormephos (136) + TX, chloroform [CCN] + TX, chloropicrin (141) + TX, chlorphoxim (989) + TX, chlorprazophos (990) + TX, chlorpyrifos (145) + TX, chlorpyrifos-methyl (146) + TX, chlorthiophos (994) + TX, chromafenozide (150) + TX, cinerin I (696) + TX, cinerin II (696) + TX, cinerins (696) + TX, cis-resmethrin (alternative name) + TX, cismethrin (80) + TX, clocythrin (alternative name) + TX, cloethocarb (999) + TX, closantel (alternative name) [CCN] + TX, clothianidin (165) + TX, copper acetoarsenite [CCN] + TX, copper arsenate [CCN] + TX, copper oleate [CCN] + TX, coumaphos (174) + TX, coumithoate (1006) + TX, crotamiton (alternative name) [CCN] + TX, crotoxyphos (1010) + TX, crufomate (1011) + TX, cryolite (alternative name) (177) + TX, CS 708 (development code) (1012) + TX, cyanofenphos (1019) + TX, cyanophos (184) + TX, cyanthoate (1020) + TX, cyclethrin [CCN] + TX, cycloprothrin (188) + TX, cyfluthrin (193) + TX, cyhalothrin (196) + TX, cypermethrin (201) + TX, cyphenothrin (206) + TX, cyromazine (209) + TX, cythioate (alternative name) [CCN] + TX, Gf-limonene (alternative name) [CCN] + TX, Gf-tetramethrin (alternative name) (788) + TX, DAEP (1031) + TX, dazomet (216) + TX, DDT (219) + TX, decarbofuran (1034) + TX, deltamethrin (223) + TX, demephion (1037) + TX, demephion-0 (1037) + TX, demephion-S (1037) + TX, demeton (1038) + TX, demeton-methyl (224) + TX, demeton-0 (1038) + TX, demeton-O-methyl (224) + TX, demeton-S (1038) + TX, demeton-S-methyl (224) + TX, demeton-S-methylsulphon (1039) + TX, diafenthiuron (226) + TX, dialifos (1042) + TX, diamidafos (1044) + TX, diazinon (227) + TX, dicapthon (1050) + TX, dichlofenthion (1051) + TX, dichlorvos (236) + TX, dicliphos (alternative name) + TX, dicresyl (alternative name) [CCN] + TX, dicrotophos (243) + TX, dicyclanil (244) + TX, dieldrin (1070) + TX, diethyl 5- methylpyrazol-3-yl phosphate (IUPAC name) (1076) + TX, diflubenzuron (250) + TX, dilor (alternative name) [CCN] + TX, dimefluthrin [CCN] + TX, dimefox (1081) + TX, dimetan (1085) + TX, dimethoate (262) + TX, dimethrin (1083) + TX, dimethylvinphos (265) + TX, dimetilan (1086) + TX, dinex (1089) + TX, dinex-diclexine (1089) + TX, dinoprop (1093) + TX, dinosam (1094) + TX, dinoseb (1095) + TX, dinotefuran (271) + TX, diofenolan (1099) + TX, dioxabenzofos (1100) + TX, dioxacarb (1101) + TX, dioxathion (1102) + TX, disulfoton (278) + TX, dithicrofos (1108) + TX, DNOC (282) + TX, doramectin (alternative name) [CCN] + TX, DSP (1115) + TX, ecdysterone (alternative name) [CCN] + TX, EI 1642 (development code) (1118) + TX, emamectin (291) + TX, emamectin benzoate (291) + TX, EMPC (1120) + TX, empenthrin
(292) + TX, endosulfan (294) + TX, endothion (1121) + TX, endrin (1122) + TX, EPBP (1123) + TX, EPN (297) + TX, epofenonane (1124) + TX, eprinomectin (alternative name) [CCN] + TX, esfenvalerate (302) + TX, etaphos (alternative name) [CCN] + TX, ethiofencarb (308) + TX, ethion (309) + TX, ethiprole (310) + TX, ethoate-methyl (1134) + TX, ethoprophos (312) + TX, ethyl formate (IUPAC name) [CCN] + TX, ethyl-DDD (alternative name) (1056) + TX, ethylene dibromide (316) + TX, ethylene dichloride (chemical name) (1136) + TX, ethylene oxide [CCN] + TX, etofenprox (319) + TX, etrimfos (1142) + TX, EXD (1143) + TX, famphur (323) + TX, fenamiphos (326) + TX, fenazaflor (1147) + TX, fenchlorphos (1148) + TX, fenethacarb (1149) + TX, fenfluthrin (1150) + TX, fenitrothion (335) + TX, fenobucarb (336) + TX, fenoxacrim (1153) + TX, fenoxycarb (340) + TX, fenpirithrin (1155) + TX, fenpropathrin (342) + TX, fenpyrad (alternative name) + TX, fensulfothion (1158) + TX, fenthion (346) + TX, fenthion- ethyl [CCN] + TX, fenvalerate (349) + TX, fipronil (354) + TX, flonicamid (358) + TX, flubendiamide (CAS. Reg. No.: 272451-65-7) + TX, flucofuron (1168) + TX, flucycloxuron (366) + TX, flucythrinate (367) + TX, fluenetil (1169) + TX, flufenerim [CCN] + TX, flufenoxuron (370) + TX, flufenprox (1171) + TX, flumethrin (372) + TX, fluvalinate (1184) + TX, FMC 1137 (development code) (1185) + TX, fonofos (1191) + TX, formetanate (405) + TX, formetanate hydrochloride (405) + TX, formothion (1192) + TX, formparanate (1193) + TX, fosmethilan (1194) + TX, fospirate (1195) + TX, fosthiazate (408) + TX, fosthietan (1196) + TX, furathiocarb (412) + TX, furethrin (1200) + TX, gamma-cyhalothrin (197) + TX, gamma-HCH (430) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, GY-81 (development code) (423) + TX, halfenprox (424) + TX, halofenozide (425) + TX, HCH (430) + TX, HEOD (1070) + TX, heptachlor (1211) + TX, heptenophos (432) + TX, heterophos [CCN] + TX, hexaflumuron (439) + TX, HHDN (864) + TX, hydramethylnon (443) + TX, hydrogen cyanide (444) + TX, hydroprene (445) + TX, hyquincarb (1223) + TX, imidacloprid (458) + TX, imiprothrin (460) + TX, indoxacarb (465) + TX, iodomethane (IUPAC name) (542) + TX, IPSP (1229) + TX, isazofos (1231) + TX, isobenzan (1232) + TX, isocarbophos (alternative name) (473) + TX, isodrin (1235) + TX, isofenphos (1236) + TX, isolane (1237) + TX, isoprocarb (472) + TX, isopropyl O- (methoxyaminothiophosphoryl)salicylate (IUPAC name) (473) + TX, isoprothiolane (474) + TX, isothioate (1244) + TX, isoxathion (480) + TX, ivermectin (alternative name) [CCN] + TX, jasmolin I (696) + TX, jasmolin II (696) + TX, jodfenphos (1248) + TX, juvenile hormone I (alternative name) [CCN] + TX, juvenile hormone II (alternative name) [CCN] + TX, juvenile hormone III (alternative name) [CCN] + TX, kelevan (1249) + TX, kinoprene (484) + TX, lambda-cyhalothrin (198) + TX, lead arsenate [CCN] + TX, lepimectin (CCN) + TX, leptophos
(1250) + TX, lindane (430) + TX, lirimfos (1251) + TX, lufenuron (490) + TX, lythidathion (1253) + TX, m-cumenyl methylcarbamate (IUPAC name) (1014) + TX, magnesium phosphide (IUPAC name) (640) + TX, malathion (492) + TX, malonoben (1254) + TX, mazidox (1255) + TX, mecarbam (502) + TX, mecarphon (1258) + TX, menazon (1260) + TX, mephosfolan (1261) + TX, mercurous chloride (513) + TX, mesulfenfos (1263) + TX, metaflumizone (CCN) + TX, metam (519) + TX, metam-potassium (alternative name) (519) + TX, metam-sodium (519) + TX, methacrifos (1266) + TX, methamidophos (527) + TX, methanesulphonyl fluoride (IUPAC/Chemical Abstracts name) (1268) + TX, methidathion (529) + TX, methiocarb (530) + TX, methocrotophos (1273) + TX, methomyl (531) + TX, methoprene (532) + TX, methoquin- butyl (1276) + TX, methothrin (alternative name) (533) + TX, methoxychlor (534) + TX, methoxyfenozide (535) + TX, methyl bromide (537) + TX, methyl isothiocyanate (543) + TX, methylchloroform (alternative name) [CCN] + TX, methylene chloride [CCN] + TX, metofluthrin [CCN] + TX, metolcarb (550) + TX, metoxadiazone (1288) + TX, mevinphos (556) + TX, mexacarbate (1290) + TX, milbemectin (557) + TX, milbemycin oxime (alternative name) [CCN] + TX, mipafox (1293) + TX, mirex (1294) + TX, monocrotophos (561) + TX, morphothion (1300) + TX, moxidectin (alternative name) [CCN] + TX, naftalofos (alternative name) [CCN] + TX, naled (567) + TX, naphthalene (IUPAC/Chemical Abstracts name) (1303) + TX, NC-170 (development code) (1306) + TX, NC-184 (compound code) + TX, nicotine (578) + TX, nicotine sulfate (578) + TX, nifluridide (1309) + TX, nitenpyram (579) + TX, nithiazine (1311) + TX, nitrilacarb (1313) + TX, nitrilacarb 1:1 zinc chloride complex (1313) + TX, NNI-0101 (compound code) + TX, NNI-0250 (compound code) + TX, nornicotine (traditional name) (1319) + TX, novaluron (585) + TX, noviflumuron (586) + TX, 0-5-dichloro-4-iodophenyl O-ethyl
ethylphosphonothioate (IUPAC name) (1057) + TX, 0,0-diethyl 0-4-methyl-2-oxo-2H-chromen- 7-yl phosphorothioate (IUPAC name) (1074) + TX, Ο,Ο-diethyl 0-6-methyl-2-propylpyrimidin-4- yl phosphorothioate (IUPAC name) (1075) + TX, Ο,Ο,Ο', O'-tetrapropyl dithiopyrophosphate (IUPAC name) (1424) + TX, oleic acid (IUPAC name) (593) + TX, omethoate (594) + TX, oxamyl (602) + TX, oxydemeton-methyl (609) + TX, oxydeprofos (1324) + TX, oxydisulfoton (1325) + TX, pp'-DDT (219) + TX, para-dichlorobenzene [CCN] + TX, parathion (615) + TX, pa rath ion-methyl (616) + TX, penfluron (alternative name) [CCN] + TX, pentachlorophenol (623) + TX, pentachlorophenyl laurate (IUPAC name) (623) + TX, permethrin (626) + TX, petroleum oils (alternative name) (628) + TX, PH 60-38 (development code) (1328) + TX, phenkapton (1330) + TX, phenothrin (630) + TX, phenthoate (631) + TX, phorate (636) + TX, phosalone (637) + TX, phosfolan (1338) + TX, phosmet (638) + TX, phosnichlor (1339) + TX,
phosphamidon (639) + TX, phosphine (IUPAC name) (640) + TX, phoxim (642) + TX, phoxim- methyl (1340) + TX, pirimetaphos (1344) + TX, pirimicarb (651) + TX, pirimiphos-ethyl (1345) + TX, pirimiphos-methyl (652) + TX, polychlorodicyclopentadiene isomers (IUPAC name) (1346) + TX, polychloroterpenes (traditional name) (1347) + TX, potassium arsenite [CCN] + TX, potassium thiocyanate [CCN] + TX, prallethrin (655) + TX, precocene I (alternative name)
[CCN] + TX, precocene II (alternative name) [CCN] + TX, precocene III (alternative name) [CCN] + TX, primidophos (1349) + TX, profenofos (662) + TX, profluthrin [CCN] + TX, promacyl (1354) + TX, promecarb (1355) + TX, propaphos (1356) + TX, propetamphos (673) + TX, propoxur (678) + TX, prothidathion (1360) + TX, prothiofos (686) + TX, prothoate (1362) + TX, protrifenbute [CCN] + TX, pymetrozine (688) + TX, pyraclofos (689) + TX, pyrazophos (693) + TX, pyresmethrin (1367) + TX, pyrethrin I (696) + TX, pyrethrin II (696) + TX, pyrethrins (696) + TX, pyridaben (699) + TX, pyridalyl (700) + TX, pyridaphenthion (701) + TX, pyrimidifen (706) + TX, pyrimitate (1370) + TX, pyriproxyfen (708) + TX, quassia (alternative name) [CCN] + TX, quinalphos (711) + TX, quinalphos-methyl (1376) + TX, quinothion (1380) + TX, quintiofos (1381) + TX, R-1492 (development code) (1382) + TX, rafoxanide (alternative name) [CCN] + TX, resmethrin (719) + TX, rotenone (722) + TX, RU 15525 (development code) (723) + TX, RU 25475 (development code) (1386) + TX, ryania (alternative name) (1387) + TX, ryanodine (traditional name) (1387) + TX, sabadilla (alternative name) (725) + TX, schradan (1389) + TX, sebufos (alternative name) + TX, selamectin (alternative name) [CCN] + TX, SI-0009 (compound code) + TX, SI-0205 (compound code) + TX, SI-0404 (compound code) + TX, SI-0405 (compound code) + TX, silafluofen (728) + TX, SN 72129 (development code) (1397) + TX, sodium arsenite [CCN] + TX, sodium cyanide (444) + TX, sodium fluoride
(IUPAC/Chemical Abstracts name) (1399) + TX, sodium hexafluorosilicate (1400) + TX, sodium pentachlorophenoxide (623) + TX, sodium selenate (IUPAC name) (1401) + TX, sodium thiocyanate [CCN] + TX, sophamide (1402) + TX, spinosad (737) + TX, spiromesifen (739) + TX, spirotetrmat (CCN) + TX, sulcofuron (746) + TX, sulcofuron-sodium (746) + TX, sulfluramid (750) + TX, sulfotep (753) + TX, sulphuryl fluoride (756) + TX, sulprofos (1408) + TX, tar oils (alternative name) (758) + TX, tau-fluvalinate (398) + TX, tazimcarb (1412) + TX, TDE (1414) + TX, tebufenozide (762) + TX, tebufenpyrad (763) + TX, tebupirimfos (764) + TX,
teflubenzuron (768) + TX, tefluthrin (769) + TX, temephos (770) + TX, TEPP (1417) + TX, terallethrin (1418) + TX, terbam (alternative name) + TX, terbufos (773) + TX,
tetrachloroethane [CCN] + TX, tetrachlorvinphos (777) + TX, tetramethrin (787) + TX, theta- cypermethrin (204) + TX, thiacloprid (791) + TX, thiafenox (alternative name) + TX,
thiamethoxam (792) + TX, thicrofos (1428) + TX, thiocarboxime (1431) + TX, thiocyclam (798) + TX, thiocyclam hydrogen oxalate (798) + TX, thiodicarb (799) + TX, thiofanox (800) + TX, thiometon (801) + TX, thionazin (1434) + TX, thiosultap (803) + TX, thiosultap-sodium (803) + TX, thuringiensin (alternative name) [CCN] + TX, tolfenpyrad (809) + TX, tralomethrin (812) + TX, transfluthrin (813) + TX, transpermethrin (1440) + TX, triamiphos (1441) + TX, triazamate (818) + TX, triazophos (820) + TX, triazuron (alternative name) + TX, trichlorfon (824) + TX, trichlormetaphos-3 (alternative name) [CCN] + TX, trichloronat (1452) + TX, trifenofos (1455) + TX, triflumuron (835) + TX, trimethacarb (840) + TX, triprene (1459) + TX, vamidothion (847) + TX, vaniliprole [CCN] + TX, veratridine (alternative name) (725) + TX, veratrine (alternative name) (725) + TX, XMC (853) + TX, xylylcarb (854) + TX, YI-5302 (compound code) + TX, zeta-cypermethrin (205) + TX, zetamethrin (alternative name) + TX, zinc phosphide (640) + TX, zolaprofos (1469) and ZXI 8901 (development code) (858) + TX, cyantraniliprole [736994-63-19] + TX, chlorantraniliprole [500008-45-7] + TX, cyenopyrafen [560121-52-0] + TX, cyflumetofen [400882-07-7] + TX, pyrifluquinazon [337458-27-2] + TX, spinetoram [187166-40-1 + 187166-15-0] + TX, spirotetramat [203313-25-1] + TX, sulfoxaflor [946578-00-3] + TX, flufiprole [704886-18-0] + TX, meperfluthrin [915288-13-0] + TX, tetramethylfluthrin [84937-88-2] + TX,
a molluscicide selected from the group of substances consisting of bis(tributyltin) oxide (IUPAC name) (913) + TX, bromoacetamide [CCN] + TX, calcium arsenate [CCN] + TX, cloethocarb (999) + TX, copper acetoarsenite [CCN] + TX, copper sulfate (172) + TX, fentin (347) + TX, ferric phosphate (IUPAC name) (352) + TX, metaldehyde (518) + TX, methiocarb (530) + TX, niclosamide (576) + TX, niclosamide-olamine (576) + TX, pentachlorophenol (623) + TX, sodium pentachlorophenoxide (623) + TX, tazimcarb (1412) + TX, thiodicarb (799) + TX, tributyltin oxide (913) + TX, trifenmorph (1454) + TX, trimethacarb (840) + TX, triphenyltin acetate (IUPAC name) (347) and triphenyltin hydroxide (IUPAC name) (347) + TX, pyriprole [394730-71-3] + TX,
a nematicide selected from the group of substances consisting of AKD-3088 (compound code) + TX, l,2-dibromo-3-chloropropane (IUPAC/Chemical Abstracts name) (1045) + TX, 1,2- dichloropropane (IUPAC/ Chemical Abstracts name) (1062) + TX, 1,2-dichloropropane with 1,3- dichloropropene (IUPAC name) (1063) + TX, 1,3-dichloropropene (233) + TX, 3,4- dichlorotetrahydrothiophene 1,1-dioxide (IUPAC/Chemical Abstracts name) (1065) + TX, 3-(4- chlorophenyl)-5-methylrhodanine (IUPAC name) (980) + TX, 5-methyl-6-thioxo-l,3,5- thiadiazinan-3-ylacetic acid (IUPAC name) (1286) + TX, 6-isopentenylaminopurine (alternative
name) (210) + TX, abamectin (1) + TX, acetoprole [CCN] + TX, alanycarb (15) + TX, aldicarb (16) + TX, aldoxycarb (863) + TX, AZ 60541 (compound code) + TX, benclothiaz [CCN] + TX, benomyl (62) + TX, butylpyridaben (alternative name) + TX, cadusafos (109) + TX, carbofuran (118) + TX, carbon disulfide (945) + TX, carbosulfan (119) + TX, chloropicrin (141) + TX, chlorpyrifos (145) + TX, cloethocarb (999) + TX, cytokinins (alternative name) (210) + TX, dazomet (216) + TX, DBCP (1045) + TX, DCIP (218) + TX, diamidafos (1044) + TX, dichlofenthion (1051) + TX, dicliphos (alternative name) + TX, dimethoate (262) + TX, doramectin (alternative name) [CCN] + TX, emamectin (291) + TX, emamectin benzoate (291) + TX, eprinomectin (alternative name) [CCN] + TX, ethoprophos (312) + TX, ethylene dibromide (316) + TX, fenamiphos (326) + TX, fenpyrad (alternative name) + TX, fensulfothion (1158) + TX, fosthiazate (408) + TX, fosthietan (1196) + TX, furfural (alternative name) [CCN] + TX, GY-81 (development code) (423) + TX, heterophos [CCN] + TX, iodomethane (IUPAC name) (542) + TX, isamidofos (1230) + TX, isazofos (1231) + TX, ivermectin (alternative name) [CCN] + TX, kinetin (alternative name) (210) + TX, mecarphon (1258) + TX, metam (519) + TX, metam-potassium (alternative name) (519) + TX, metam-sodium (519) + TX, methyl bromide (537) + TX, methyl isothiocyanate (543) + TX, milbemycin oxime (alternative name) [CCN] + TX, moxidectin (alternative name) [CCN] + TX, Myrothecium verrucaria composition (alternative name) (565) + TX, NC-184 (compound code) + TX, oxamyl (602) + TX, phorate (636) + TX, phosphamidon (639) + TX, phosphocarb [CCN] + TX, sebufos (alternative name) + TX, selamectin (alternative name) [CCN] + TX, spinosad (737) + TX, terbam (alternative name) + TX, terbufos (773) + TX, tetrachlorothiophene (IUPAC/ Chemical Abstracts name) (1422) + TX, thiafenox (alternative name) + TX, thionazin (1434) + TX, triazophos (820) + TX, triazuron (alternative name) + TX, xylenols [CCN] + TX, YI-5302 (compound code) and zeatin (alternative name) (210) + TX, fluensulfone [318290-98-1] + TX,
a nitrification inhibitor selected from the group of substances consisting of potassium ethylxanthate [CCN] and nitrapyrin (580) + TX,
a plant activator selected from the group of substances consisting of acibenzolar (6) + TX, acibenzolar-S-methyl (6) + TX, probenazole (658) and Reynoutria sachalinensis extract
(alternative name) (720) + TX,
a rodenticide selected from the group of substances consisting of 2-isovalerylindan-l,3- dione (IUPAC name) (1246) + TX, 4-(quinoxalin-2-ylamino)benzenesulfonamide (IUPAC name) (748) + TX, alpha-chlorohydrin [CCN] + TX, aluminium phosphide (640) + TX, antu (880) + TX, arsenous oxide (882) + TX, barium carbonate (891) + TX, bisthiosemi (912) + TX, brodifacoum
(89) + TX, bromadiolone (91) + TX, bromethalin (92) + TX, calcium cyanide (444) + TX, chloralose (127) + TX, chlorophacinone (140) + TX, cholecalciferol (alternative name) (850) + TX, coumachlor (1004) + TX, coumafuryl (1005) + TX, coumatetralyl (175) + TX, crimidine (1009) + TX, difenacoum (246) + TX, difethialone (249) + TX, diphacinone (273) + TX, ergocalciferol (301) + TX, flocoumafen (357) + TX, fluoroacetamide (379) + TX, flupropadine (1183) + TX, flupropadine hydrochloride (1183) + TX, gamma-HCH (430) + TX, HCH (430) + TX, hydrogen cyanide (444) + TX, iodomethane (IUPAC name) (542) + TX, lindane (430) + TX, magnesium phosphide (IUPAC name) (640) + TX, methyl bromide (537) + TX, norbormide (1318) + TX, phosacetim (1336) + TX, phosphine (IUPAC name) (640) + TX, phosphorus [CCN] + TX, pindone (1341) + TX, potassium arsenite [CCN] + TX, pyrinuron (1371) + TX, scilliroside (1390) + TX, sodium arsenite [CCN] + TX, sodium cyanide (444) + TX, sodium fluoroacetate (735) + TX, strychnine (745) + TX, thallium sulfate [CCN] + TX, warfarin (851) and zinc phosphide (640) + TX,
a synergist selected from the group of substances consisting of 2-(2-butoxyethoxy)ethyl piperonylate (IUPAC name) (934) + TX, 5-(l,3-benzodioxol-5-yl)-3-hexylcyclohex-2-enone (IUPAC name) (903) + TX, farnesol with nerolidol (alternative name) (324) + TX, MB-599 (development code) (498) + TX, MGK 264 (development code) (296) + TX, piperonyl butoxide (649) + TX, piprotal (1343) + TX, propyl isomer (1358) + TX, S421 (development code) (724) + TX, sesamex (1393) + TX, sesasmolin (1394) and sulfoxide (1406) + TX,
an animal repellent selected from the group of substances consisting of anthraquinone (32)
+ TX, chloralose (127) + TX, copper naphthenate [CCN] + TX, copper oxychloride (171) + TX, diazinon (227) + TX, dicyclopentadiene (chemical name) (1069) + TX, guazatine (422) + TX, guazatine acetates (422) + TX, methiocarb (530) + TX, pyridin-4-amine (IUPAC name) (23) + TX, thiram (804) + TX, trimethacarb (840) + TX, zinc naphthenate [CCN] and ziram (856) + TX, a virucide selected from the group of substances consisting of imanin (alternative name)
[CCN] and ribavirin (alternative name) [CCN] + TX,
a wound protectant selected from the group of substances consisting of mercuric oxide (512) + TX, octhilinone (590) and thiophanate-methyl (802) + TX,
and biologically active compounds selected from the group consisting of azaconazole (60207-31-0] + TX, bitertanol [70585-36-3] + TX, bromuconazole [116255-48-2] + TX, cyproconazole [94361-06-5] + TX, difenoconazole [119446-68-3] + TX, diniconazole [83657-24- 3] + TX, epoxiconazole [106325-08-0] + TX, fenbuconazole [114369-43-6] + TX,
fluquinconazole [136426-54-5] + TX, flusilazole [85509-19-9] + TX, flutriafol [76674-21-0] +
TX, hexaconazole [79983-71-4] + TX, imazalil [35554-44-0] + TX, imibenconazole [86598-92-7] + TX, ipconazole [125225-28-7] + TX, metconazole [125116-23-6] + TX, myclobutanil [88671- 89-0] + TX, pefurazoate [101903-30-4] + TX, penconazole [66246-88-6] + TX, prothioconazole [178928-70-6] + TX, pyrifenox [88283-41-4] + TX, prochloraz [67747-09-5] + TX,
propiconazole [60207-90-1] + TX, simeconazole [149508-90-7] + TX, tebuconazole [107534-96- 3] + TX, tetraconazole [112281-77-3] + TX, triadimefon [43121-43-3] + TX, triadimenol
[55219-65-3] + TX, triflumizole [99387-89-0] + TX, triticonazole [131983-72-7] + TX, ancymidol [12771-68-5] + TX, fenarimol [60168-88-9] + TX, nuarimol [63284-71-9] + TX, bupirimate
[41483-43-6] + TX, dimethirimol [5221-53-4] + TX, ethirimol [23947-60-6] + TX, dodemorph [1593-77-7] + TX, fenpropidine [67306-00-7] + TX, fenpropimorph [67564-91-4] + TX, spiroxamine [118134-30-8] + TX, tridemorph [81412-43-3] + TX, cyprodinil [121552-61-2] + TX, mepanipyrim [110235-47-7] + TX, pyrimethanil [53112-28-0] + TX, fenpiclonil [74738-17-3] + TX, fludioxonil [131341-86-1] + TX, benalaxyl [71626-11-4] + TX, furalaxyl [57646-30-7] + TX, metalaxyl [57837-19-1] + TX, R-metalaxyl [70630-17-0] + TX, ofurace [58810-48-3] + TX, oxadixyl [77732-09-3] + TX, benomyl [17804-35-2] + TX, carbendazim [10605-21-7] + TX, debacarb [62732-91-6] + TX, fuberidazole [3878-19-1] + TX, thiabendazole [148-79-8] + TX, chlozolinate [84332-86-5] + TX, dichlozoline [24201-58-9] + TX, iprodione [36734-19-7] + TX, myclozoline [54864-61-8] + TX, procymidone [32809-16-8] + TX, vinclozoline [50471-44-8] + TX, boscalid [188425-85-6] + TX, carboxin [5234-68-4] + TX, fenfuram [24691-80-3] + TX, flutolanil [66332-96-5] + TX, mepronil [55814-41-0] + TX, oxycarboxin [5259-88-1] + TX, penthiopyrad [183675-82-3] + TX, thifluzamide [130000-40-7] + TX, guazatine [108173-90-6] + TX, dodine [2439-10-3] [112-65-2] (free base) + TX, iminoctadine [13516-27-3] + TX, azoxystrobin [131860-33-8] + TX, dimoxystrobin [149961-52-4] + TX, enestroburin {Proc.
BCPC, Int. Congr., Glasgow, 2003, 1, 93} + TX, fluoxastrobin [361377-29-9] + TX, kresoxim- methyl [143390-89-0] + TX, metominostrobin [133408-50-1] + TX, trifloxystrobin [141517-21- 7] + TX, orysastrobin [248593-16-0] + TX, picoxystrobin [117428-22-5] + TX, pyraclostrobin [175013-18-0] + TX, ferbam [14484-64-1] + TX, mancozeb [8018-01-7] + TX, maneb [12427- 38-2] + TX, metiram [9006-42-2] + TX, propineb [12071-83-9] + TX, thiram [137-26-8] + TX, zineb [12122-67-7] + TX, ziram [137-30-4] + TX, captafol [2425-06-1] + TX, captan [133-06-2] + TX, dichlofluanid [1085-98-9] + TX, fluoroimide [41205-21-4] + TX, folpet [133-07-3 ] + TX, tolylfluanid [731-27-1] + TX, bordeaux mixture [8011-63-0] + TX, copperhydroxid [20427-59-2] + TX, copperoxychlorid [1332-40-7] + TX, coppersulfat [7758-98-7] + TX, copperoxid [1317-39- 1] + TX, mancopper [53988-93-5] + TX, oxine-copper [10380-28-6] + TX, dinocap [131-72-6]
+ TX, nitrothal-isopropyl [10552-74-6] + TX, edifenphos [17109-49-8] + TX, iprobenphos
[26087-47-8] + TX, isoprothiolane [50512-35-1] + TX, phosdiphen [36519-00-3] + TX, pyrazophos [13457-18-6] + TX, tolclofos-methyl [57018-04-9] + TX, acibenzolar-S-methyl [135158-54-2] + TX, anilazine [101-05-3] + TX, benthiavalicarb [413615-35-7] + TX, blasticidin-S [2079-00-7] + TX, chinomethionat [2439-01-2] + TX, chloroneb [2675-77-6] + TX, chlorothalonil [1897-45-6] + TX, cyflufenamid [180409-60-3] + TX, cymoxanil [57966-95-7] + TX, dichlone [117-80-6] + TX, diclocymet [139920-32-4] + TX, diclomezine [62865-36-5] + TX, dicloran [99-30-9] + TX, diethofencarb [87130-20-9] + TX, dimethomorph [110488-70-5] + TX, SYP-LI90 (Flumorph) [211867-47-9] + TX, dithianon [3347-22-6] + TX, ethaboxam [162650-77 3] + TX, etridiazole [2593-15-9] + TX, famoxadone [131807-57-3] + TX, fenamidone [161326- 34-7] + TX, fenoxanil [115852-48-7] + TX, fentin [668-34-8] + TX, ferimzone [89269-64-7] + TX, fluazinam [79622-59-6] + TX, fluopicolide [239110-15-7] + TX, flusulfamide [106917-52-6] + TX, fenhexamid [126833-17-8] + TX, fosetyl-aluminium [39148-24-8] + TX, hymexazol
[10004-44-1] + TX, iprovalicarb [140923-17-7] + TX, IKF-916 (Cyazofamid) [120116-88-3] + TX, kasugamycin [6980-18-3] + TX, methasulfocarb [66952-49-6] + TX, metrafenone [220899- 03-6] + TX, pencycuron [66063-05-6] + TX, phthalide [27355-22-2] + TX, polyoxins [11113-80 7] + TX, probenazole [27605-76-1] + TX, propamocarb [25606-41-1] + TX, proquinazid
[189278-12-4] + TX, pyroquilon [57369-32-1] + TX, quinoxyfen [124495-18-7] + TX, quintozene [82-68-8] + TX, sulphur [7704-34-9] + TX, tiadinil [223580-51-6] + TX, triazoxide [72459-58-6] + TX, tricyclazole [41814-78-2] + TX, triforine [26644-46-2] + TX, validamycin [37248-47-8] + TX, zoxamide (RH7281) [156052-68-5] + TX, mandipropamid [374726-62-2] + TX, isopyrazam [881685-58-1] + TX, sedaxane [874967-67-6] + TX, 3-difluoromethyl-l-methyl- lH-pyrazole-4-carboxylic acid (9-dichloromethylene-l,2,3,4-tetrahydro-l,4-methano-naphthalen 5-yl)-amide (dislosed in WO 2007/048556) + TX, 3-difluoromethyl-l-methyl-lH-pyrazole-4- carboxylic acid [2-(2,4-dichlorophenyl)-2-methoxy-l-methyl-ethyl]-amide (disclosed in WO 2008/148570) + TX, l-[4-[4-[(5S)5-(2,6-difluorophenyl)-4,5-dihydro-l,2-oxazol-3-yl]-l,3- thiazol-2-yl]piperidin-l-yl]-2-[5-methyl-3-(trifluoromethyl)-lH-pyrazol-l-yl]ethanone + TX, l-[4- [4-[5-(2,6-difluorophenyl)-4,5-dihydro-l,2-oxazol-3-yl]-l,3-thiazol-2-yl]piperidin-l-yl]-2-[5- methyl-3-(trifluoromethyl)-lH-pyrazol-l-yl]ethanone [1003318-67-9], both disclosed in WO 2010/123791, WO 2008/013925, WO 2008/013622 and WO 2011/051243 page 20) +TX, S)-[3- (4-Chloro-2-fluoro-phenyl)-5 -(2,4-difluoro-phenyl)-isoxazol-4-y l]-pyridin-3-yl-methanol + TX, 3 (4-Chloro-2-fluoro-phenyl)-5 -(2,4-difluoro-phenyl)-isoxazol-4-y l]-pyridin-3-yl-methanol + TX, 3 difluoromethyl-l-methyl-lH-pyrazole-4-carboxylic acid (3',4',5'-trifluoro-biphenyl-2-yl)-amide
(disclosed in WO 2006/087343) + TX, 3-(difluoromethyl)-N-methoxy-l-methyl-N-[l-methyl-2- (2,4,6-trichlorophenyl)ethyl]-lH-Pyrazole-4-carboxamide + TX, 4-[(5S)-5-(3,5-dichlorophenyl)-5- (trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-N-(thietan-3-yl)benzamide (WO2011/104089) + TX, 4-[(5R)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-N-(th
yl)benzamide (WO2011/104089) + TX, 4-[(5S)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H- isoxazol-3-yl]-2-methyl-N-(cis-l-oxo-thietan-3-yl)benzamide (WO2011/104089) + TX, 4-[(5Λ)-5- (3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isox^^
yl)benzamide (WO2011/104089) + TX, 4-[(5S)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H- isoxazol-3-yl]-2-methyl-N-(trans-l-oxo-thietan-3-yl)benzamide (WO2011/104089) + TX, 4-
[(5/?)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-N-(tran
thietan-3-yl)benzamide (WO2011/104089) + TX, 4-[(5S)-5-(3,5-dichlorophenyl)-5-
(trifluoromethyl)-4H-isoxazol-3-yl]-N-(l,l-dioxothietan-3-yl)-2-methyl-benzamide
(WO2011/104089) + TX, 4-[(5/?)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-N-
(l,l-dioxothietan-3-yl)-2-methyl-benzamide (WO2011/104089) + TX, 4-[(5S)-5-(3,5- dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-N-[2-oxo-2-(2,2,2- trifluoroethylamino)ethyl]benzamide (WO2011/104089) + TX, 4-[(5R)-5-(3,5-dichlorophenyl)-5-
(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-N-[2-oxo-2-(2,2,2- trifluoroethylamino)ethyl]benzamide (WO2011/104089) + TX, Penflufen [494793-67-8] + TX, 5- [(5S)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-(l,2,4-triazol-l- yl)benzonitrile (WO2007/075459) + TX, 5-[(5R)-5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H- isoxazol-3-yl]-2-(l,2,4-triazol-l-yl)benzonitrile (WO2007/075459) +TX.
The components (B) are known. The references in brackets behind the active ingredients, e.g. [3878-19-1] refer to the Chemical Abstracts Registry number. The above described mixing partners are known. Where the active ingredients are included in "The Pesticide Manual" [The Pesticide Manual - A World Compendium; Thirteenth Edition; Editor: C. D. S. TomLin; The British Crop Protection Council], they are described therein under the entry number given in round brackets hereinabove for the particular compound; for example, the compound "abamectin" is described under entry number (1). Where "[CCN]" is added hereinabove to the particular compound, the compound in question is included in the "Compendium of Pesticide Common Names", which is accessible on the internet under the internet address
http://www.alanwood.net/pesticides/ [A. Wood; Compendium of Pesticide Common Names, Copyright © 1995-2012]; or preferably one of the further pesticides listed below.
In the above different lists of active ingredients to be mixed with a TX, the compound of the formula I is preferably a compound selected from the Tables 1-7 or Table 9; more preferably Table 9;
and even more preferably a compound selected from P.01, P.02, P.03, P.04, P.05, P.06, P.08, P.09, P.10, P.ll, P.12, P.14, P.15, P.16, P.17 or P.18.
In the above-mentioned mixtures of compounds of formula I, in particular a compound selected from said Tables 1-7 or Table 9, with other insecticides, fungicides, herbicides, safeners, adjuvants and the like, the mixing ratios can vary over a large range and are, preferably 100: 1 to 1:6000, especially 50: 1 to 1:50, more especially 20: 1 to 1:20, even more especially 10: 1 to 1: 10. Those mixing ratios are understood to include, on the one hand, ratios by weight and also, on other hand, molar ratios.
The mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient" relates to the respective mixture of TX with the mixing partner).
Some mixtures may comprise active ingredients which have significantly different physical, chemical or biological properties such that they do not easily lend themselves to the same conventional formulation type. In these circumstances other formulation types may be prepared. For example, where one active ingredient is a water insoluble solid and the other a water insoluble liquid, it may nevertheless be possible to disperse each active ingredient in the same continuous aqueous phase by dispersing the solid active ingredient as a suspension (using a preparation analogous to that of an SC) but dispersing the liquid active ingredient as an emulsion (using a preparation analogous to that of an EW). The resultant composition is a suspoemulsion (SE) formulation.
The mixtures comprising a TX selected from Tables 1-7 or Table 9 and one or more active ingredients as described above can be applied, for example, in a single "ready-mix" form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a "tank-mix", and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days. The order of applying the compounds of formula I selected from Tables 1-7 or Table 9 and the active ingredients as described above is not essential for working the present invention.
The following non-limiting Examples illustrate the above-described invention in greater detail without limiting it. Those skilled in the art will promptly recognize appropriate variations
from the procedures both as to reactants and as to reaction conditions and techniques. All references mentioned herein are incorporated by reference in their entirety.
Preparatory examples:
Pre aration of compound P.2
A 15 mL flask was charged with a solution of 2-methyl-6,7-dihydro-5H-quinolin-8-one 0-(3- aminooxy-propyl)-E-oxime (64 mg; 0.23 mmol) in methanol (10 mL). Under stirring l-(4,6- diethoxy-pyrimidin-2-yl)-ethanone (49 mg; 0.23 mmol) was added. The resulting solution was stirred at room temperature for 17 hours. TLC analysis of an aliquot of the reaction mixture showed that all of the starting materials was consumed. After evaporation of the solvent in vacuo, the title compound was recovered as a yellowish gum (106 mg, 98%). No further purification was required.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: cyclohexane/ ethylacetate 1: 1 (v:v); Rf of title compound = 0.59, Rf oxime starting material = 0.11.
LC-MS (Method ZMD): Rt = 1.72 min. MS: (M++l)=470
JH NMR (200 MHz, CDCI3) 6(ppm): 7.34 (d, 1H), 7.04 (d, 1H), 5.94 (s, 1H), 4.34-4.46 (q, 4H), 4.26 (s, 2H), 4.22 (s, 2H), 2.87 (t, 2H), 2.71 (m, 2H), 2.59 (s, 3H), 2.30 (s, 3H), 1.84 (m, 2H), 1.37-1.42 (t, 6H), 1.10 (s, 6H).
Preparation of 2-Methyl-6,7-dihydro-5H-quinolin-8-one-Q-(3-aminooxy-2,2-dimethyl-propyl>
E-oxime
A solution of hydroxylamine-0-[3-(aminooxy)-2,2-dimethylpropyl] hydrochloride (1:2) (21.7 g) in absolute ethanol (300 mL) was charged to a 500 mL reaction flask which had previously been placed under an atmosphere of argon. Under stirring, p-toluenesulphonic acid (1.2 g) was added, followed by the dropwise addition of 2-methyl-6,7-dihydro-5H-quinolin-8-one (6.77 g)
dissolved in absolute ethanol (30 ml_). The resulting yellow solution was stirred at room temperature for 1.5 hours. Following the course of the reaction by TLC indicated that no starting materials were left at this point in time. The ethanol was removed in vacuo. Aqueous sodium bicarbonate solution (150 mL) was then added. Extraction was carried out using ethyl acetate (2x100 mL). The combined organic layers were dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a beige oil (10.9 g). This raw material was purified by chromatography on silica gel (eluent: heptane/ ethyl acetate 2: 1 (v:v) with 1 vol-% of triethylamine). This was followed by RP-HPLC chromatography (Separation Laboratory: Method 10-40). The title compound (6.00 g) was obtained in the form of a light yellow gum.
LC-MS (Method ZMD): UV Detection: 220 nm; Rt = 0.83 min.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptane/ ethyl acetate 1:2 (v:v); Rf of title compound = 0.11.
A solution of hydroxylamine-0-[3-(aminooxy)-2,2-dimethylpropyl] hydrochloride (1:2) (100 mg; 0.48 mmol) in ethanol (18 mL) was charged to a 25 mL reaction flask. Under stirring 2- methyl-6,7-dihydro-5H-quinazolin-8-one (157 mg; 0.97 mmol) and and potassium carbonate (73 mg; 0.53 mmol) were added. The resulting suspension was stirred at room temperature for 15 hours, poured onto water (100 mL) and extracted with ethyl acetate (3x80 mL). The combined organic layers were dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a beige oil. This raw material was purified by chromatography (combiflash RF, 0-70% ethylacetate in cyclohexane) to give the title compound (116 mg, 57% yield) as brown crystals (decomposition from 180°C). LC-MS (Method ZCQ): Rt = 1.64 min. MS: ([M+l]+)=423.
A 10 mL flask was charged with a solution of 2-methyl-6,7-dihydro-5H-quinolin-8-one 0-(3- aminooxy-propyl)-E-oxime (101 mg; 0.36 mmol) in methanol (8 mL). Under stirring 2(3-Chloro- phenylamino)-6,7-dihydro-5H-quinazolin-8-one (100 mg; 0.36 mmol) was added. The resulting
solution was stirred at room temperature for 18 hours. After evaporation of the solvent in vacuo, the crude was purified via chromatography (combiflash RF, 0-60% ethylacetate in cyclohexane) to give 2-(3-chlorophenylamino)-6,7-dihydro-5-quinazolin-8-one 0-(2,2-dimethyl-3-(2-methyl- 6,7-dihydro-5H-quinolin-(8E)-ylideneaminooxy)-propyl)-oxime (compound P.15) (0.17g, 87% yield) as a yellow solid decomposition from 79°C. LC-MS (Method ZCQ) Rt = 1.73 min., MS: (M++l) = 533.
Preparation of 2(3-Chloro-phenylamino)-6,7-dihydro-5H-quinazolin-8-one
In a 25 mL single-necked round -bottomed flask equipped with a condenser, (3-chloro- phenyl)-(8-ethoxy-5,6-dihydro-quinazolin-2-yl)-amine (0.39 g; 1.31 mmol) was dissolved in a mixture of acetic acid (7.0 mL) and water (0.70 mL). Stirring was continued under heating to reflux for 1 hour. TLC indicated that starting materials were consumed. The mixture was cooled to room temperature, then water and saturated NaHC03 were added. Extraction was carried out using dichloromethane (3x20 mL). The combined organic phases were washed with brine and then dried over sodium sulphate, filtered and the solvent was removed in vacuo to give an oil. The crude was purified via chromatography (combiflash RF, 0-80% ethylacetate in cyclohexane) to give 2(3-Chloro-phenylamino)-6,7-dihydro-5H-quinazolin-8-one (0.21g, 58% of theory) as a white solid MP. 220-222°C. LC-MS (Method ZCQ) Rt = 1.68 min. , MS: (M++l) = 274. JH-NMR (CDCI3, 400 MHz): 2.23 (m, 2H); 2.84 (m, 2H); 2.94 (m, 2H); 7.05 (m, 1H); 7.24 (m, 1H); 7.22 (m, 2H); 7.83 (d, 1H); 8.68 (s, 1H). TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptane/ ethyl acetate 1: 1 (v:v); Rf of title compound = 0.56.
Preparation of (3-chloro- henyl>(8-ethoxy-5,6-dihydro-quinazolin-2-yn-amine
In a 25mL single-necked round-bottomed flask equipped with a condenser, a mixture of 6- [l-dimethylamino-meth-(E)-ylidene]-2-ethoxy-cyclohex-2-enone (WO 2004/104007) (2.60 g; 13.32 mmol), N-(3-chloro-phenyl)guanidine carbonate (4.01 g; 17.31 mmol) and potassium
carbonate (2.02 g; 14.65 mmol) in absolute ethanol (50 mL) was stirred at reflux temperature over night for 17 hours. The reaction mixture was evaporated to dryness, taken up in water (80 mL), extracted with dichloromethane (3x 50 mL). Combined organic layers were dried over sodium sulphate, filtered and the solvent was removed in vacuo . The crude was purified via chromatography (combiflash RF, 0-80% ethylacetate in cyclohexane) to give (3-chloro-phenyl)- (8-ethoxy-5,6-dihydro-quinazolin-2-yl)-amine (0.39g, 10% of theory) as a white solid melting MP. 113-116°C. LC-MS (Method ZCQ) Rt = 1.97 min., MS: (M++l) = 302.
The following examples illustrate the preparation of useful intermediates:
2-Methyl-6,7-Dihydro-5H-quinolin-8-one-(E>oxime
A 250 mL single-necked round-bottomed flask equipped with a condenser was charged with a solution of 2-methyl-6,7-dihydro-5H-quinolin-8-one (7.00 g) (CA Registry Number: 849643-01- 2) in absolute ethanol (70 mL). Under stirring, hydroxylamine-hydrochloride (4.50 g) was added, followed by portionwise addition of a solution of NaOH (8.70 g) in water (14.00 mL). Stirring was continued under heating to reflux for 6.0 hours. Following the course of the reaction by TLC indicated that starting materials were consumed by this time. The suspension was cooled to room temperature. Under stirring and cooling with an ice-water cooling bath, water (10 mL) was added and the pH was adjusted to 6 by the addition of 6 M aqueous HCI. Extraction was carried out using ethyl acetate (2x100 mL). The combined organic phases were washed with brine and then dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a yellow solid (7.65 g).
Analytical data for the title compound:
LC-MS (Method ZMD) UV Detection: 220 nm; Rt = 0.20 , MS: (M++l) = 177, (M+ +23) = 179; melting point = 177-181°C.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: ethyl acetate/triethylamine 10: 1 (v:v); Rf of title compound = 0.26, Rf of the ketone starting material = 0.46.
CA Registry Number: 849643-01-2
U.S. Pat. Appl. Publ. (2005), 75 pp., Cont.-in-part of U.S. Ser. No. 437,807. CODEN:
■2 HCI CA Registry Number: 1034433-68-5
PCT Int. Appl. (2008), 187 pp. CODEN: PIXXD2 WO 2008074418 A2 20080626
E CA Registry Number: 23089-39-6
CA Registry Number: 18103-88-3
Talanta (1969), 16(3), 448-52; DE 2447258 (19760408); Journal of Heterocyclic Chemistry (1968), 5(2), 161-4.
Preparation of 4-chloro-2-methyl-6,7-dihydro-8(5H)-quinolinone
Step A) 4-Hydroxy-2-methylquinoline (10.0 g) (CA Registry Number: 607-67-0) was charged to a reactor containing absolute ethanol (90.0 mL) under a nitrogen atmosphere. Under stirring, a suspension of Raney nickel (2.0 g) in absolute ethanol (10.0 mL) was added to the reaction mixture. The nitrogen atmosphere was then replaced by hydrogen. The reaction mixture was stirred at 75°C for 22 hours under a 100 bar hydrogen atmosphere, at which time analysis of the reaction mixture by TLC indicated that starting material was consumed. The catalyst was filtered off and the solvent was removed in vacuo to give a white solid (8.35 g). The compound was used as such for the next step.
LC-MS (ZMD): UV Detection: 220 nm; Rt = 0.40 min. MS: (M++l) 164; melting point = 237-
240°C.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: dichloromethane / methanol 9: 1 (v:v); Rf of title compound = 0.22, Rf of quinoline starting material = 0.34.
Step B) A 50 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 2-methyl-5,6,7,8-tetrahydro-quinolin-4-ol (4.00 g) in phosphorus oxide chloride (18.3 mL) under an argon atmosphere. The resulting colourless solution was stirred at 100°C for 3.5 hours, after which time TLC indicated that no starting material was remaining. The solvent was removed in vacuo and hot water (40-50°C) was added carefully and slowly to the residue to hydrolyse the remaining phosphorus oxide chloride. Under cooling with an ice- water cooling bath, the pH was adjusted to 12 by the addition of 4 M aqueous NaOH. The resulting solution was extracted using chloroform (2x50 mL). The combined organic layers were washed with brine (25 mL) and then dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a light yellow oil (4.21 g). The compound was used as such for the next step.
LC-MS (ZMD): UV Detection: 220 nm; Rt = 0.87 min. MS: (M++ l) = 182.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptane / ethyl acetate 1 :4 (v:v); Rf of title compound = 0.40, Rf of quinolinol starting material = 0.
Step C) A 25 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 4-chloro-2-methyl-5,6,7,8-tetrahydroquinoline (560 mg) in acetic anhydride (0.49 mL). Under stirring, benzaldehyde (0.34 mL) was added and the resulting yellow solution was stirred under heating to reflux for 3.5 hours. Following the course of the reaction by TLC indicated that starting material was consumed by this time. The resulting brown solution was cooled to room temperature. Crushed ice was added and the pH was adjusted to 10 using a small amount of 2 M aqueous NaOH. Extraction was carried out using ethyl acetate (2x20 mL). The combined organic phases were dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a brown gum (750 mg). The crude material was purified by chromatography on silica gel (eluent: heptane / ethyl acetate 98:2 (v:v)). This gave the title compound (263 mg) as a yellow oil.
LC-MS (Method ZMD) UV Detection: 220 nm; Rt = 2.23 , MS: (M++ l) = 270.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptanes / ethyl acetate 9: 1 (v:v); Rf of title compound = 0.44, Rf of the chloroquinoline starting material = 0.09.
Step D) A 25 mL single-necked round-bottom flask was charged with a solution of 8- benzylidene-4-chloro-2-methyl-5,6,7,8-tetrahydro-quinoline (263 mg) in dichloromethane (2.0 mL) / methanol (3.8 mL). Under stirring and cooling to -78°C with a dry ice-acetone cooling bath, ozone was passed through the reaction mixture for 3 minutes until a light blue color was observed. Then dimethyl sulfide (2.0 mL) was added at -78°C. Then, the reaction mixture was allowed to reach room temperature and was stirred for 4 hours. Solvents were removed in vacuo, then the resulting orange gum was taken up in diethyl ether and 1 M aqueous HCI (5 mL) was added. Extraction of the acidic by-products was carried out using diethyl ether (2x20 mL). Crushed ice was added to the aqueous layer and the pH was adjusted to 10 by the addition of 2 M aqueous NaOH. The resulting solution was extracted using chloroform (2x20 mL). The combined organic layers were dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a yellow solid (96 mg).
Analytical data for the title compound:
LC-MS (Method ZMD) UV Detection: 220 nm; Rt = 1.28 , MS: (M++l) = 196.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptanes / ethyl acetate 4: 1 (v:v); Rf of title compound = 0.04, Rf of the benzylidene starting material = 0.59.
Preparation of 2,4-dimethyl-6,7-dihydro-8(5H)-quinolinone
Step A) A 5 mL microwave tube was charged with a solution of 4-chloro-2-methyl-5,6,7,8- tetrahydroquinoline (500 mg) in 1,2-dichloroethane (2.50 mL). Under stirring, trimethylboroxine (380 mg), potassium carbonate (647 mg) and dichloro[l,l'-bis(diphenylphosphino)ferrocene] palladium(II) dichloromethane adduct (101 mg) were added and the resulting red suspension was degassed under argon for 5 minutes. The reaction mixture was subjected to microwave irradiation at 120°C for 0.5 hour. After addition of new portions of trimethylboroxine (2x380 mg) and catalyst (101 mg), the reaction mixture was again subjected to microwave irradiation at 120°C for 2x0.5 hour. Following the course of the reaction by TLC indicated that starting material was consumed by this time. The resulting brown material was purified by
chromatography on silica gel (eluent: heptane / ethyl acetate 4: 1 (v:v)). This gave the tittle compound (345 mg) as a light brown oil.
LC-MS (Method ZMD) UV Detection: 220 nm; Rt = 0.75 , MS: (M++ l) = 162.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptane / ethyl acetate 1:2 (v:v); Rf of title compound = 0.28, Rf of the chloroquinoline starting material = 0.40.
Step B) 2,4-Dimethyl-5,6,7,8-tetrahydro-quinoline (150 mg) was charged to a 10 mL single- necked round-bottom flask containing chloroform (1.50 mL). Under stirring and cooling with an ice-water cooling bath, 3-chloroperbenzoic acid (344 mg) was added portion wise. The resulting orange solution was stirred at room temperature for 5 hours, at which time analysis of the reaction mixture by TLC indicated that starting material was consumed. Under cooling with an ice-water cooling bath, the pH was adjusted to pH 12 by the addition of 4 M aqueous NaOH (2.0 mL). The resulting solution was extracted using chloroform (3x10 mL). The combined organic layers were washed with brine (10 mL) and then dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a light orange gum (180 mg). This intermediate was used as such for the following step.
LC-MS (ZMD): UV Detection: 220 nm; Rt = 1.26 min. MS: (M++ l) 178.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptane / ethyl acetate 1:2 (v:v); Rf of title compound = 0, Rf of quinoline starting material = 0.28.
Step C) A 25 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 2,4-dimethyl-5,6,7,8-tetrahydro-quinoline-l-oxide (334 mg) in
dichloromethane (2.00 mL) under an argon atmosphere. Under stirring and cooling with an ice- water cooling bath, trifluoroacetic anhydride (2.66 mL) was added dropwise and the resulting orange solution was stirred under heating to reflux for 22 hours. Following the course of the reaction by TLC indicated that starting material was consumed by this time. The resulting brown solution was cooled to room temperature. Crushed ice was added and the pH was adjusted to 12 using 2 M aqueous NaOH (5 mL). Extraction was carried out using dichloromethane (3x10 mL). The combined organic phases were dried over sodium sulphate, filtered and the solvent was removed in vacuo to give a dark brown gum (226 mg). Th is intermediate was used without further purification in the next step.
LC-MS (Method ZMD) UV Detection: 220 nm; Rt = 0.25 , MS: (M++ l) = 178, (M+ -18) =
160.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptanes / ethyl acetate 1:4 (v:v); Rf of title compound = 0.08, Rf of the quinoline-oxide starting material = 0.
Step D) A 25 mL single-necked round-bottom flask equipped with a condenser was charged with a solution of 2,4-dimethyl-5,6,7,8-tetrahydro-quinolin-8-ol (226 mg) in chloroform (2.00 mL). Under stirring, manganese(IV) oxide (443 mg) was added and the resulting black suspension was stirred under heating to reflux for 18 hours, after which time TLC indicated that no starting material was remaining. The resulting black material was purified by chromatography on silica gel (eluent: heptane / ethyl acetate gradient from 1:1 to 1:2 (v:v)). This gave the tittle compound (78 mg) as an orange gum.
Analytical data for the title compound:
LC-MS (ZMD): UV Detection: 220 nm; Rt = 0.34 min. MS: (M++l) = 176.
TLC: Plates: Merck DC-Plates, silica gel F254, saturated atmosphere in developing tank, UV detection, eluent: heptane / ethyl acetate 1:4 (v:v); Rf of title compound = 0.11, Rf of quinolinol starting material = 0.08.
Preparation of 9-methyl-2,3-dihydro-lH-acridin-4-one
Step A) 9-Methyl-l,2,3,4-tetrahydroacridine: In a round-bottom flask, cyclohexanone (6.1 mL, 58 mmol) was heated at 90°C, and 2-aminoacetophenone hydrochloride (10 g; 58 mmol) was added by small fractions. The bottom flask was then equipped with a condenser, and the crude heterogeneous mixture was further heated overnight at 110°C. After cooling to room temperature, the red-orange solid was dissolved in ethanol/HCI (12 N) [95/5 v/v]. The solution was then neutralized with an aqueous NaOH solution. Ethanol was evaporated, and the product extracted with diethyl ether (2 x 100 mL). The combined organic layers were washed with water (2 x 100 mL), dried over magnesium sulfate and filtered, and the solvent was removed under reduced pressure. The desired product was finally obtained as a brown-yellow solid (10.2 g, 89%). *H NMR (200.131 MHz; CDCI3) 6(ppm): 7.94 (dd, 3J = 8.3 Hz and 4J = 1.1 Hz, 1H), 7.87 (dd, 3J = 8.3 Hz and 4J = 1.3 Hz, 1H), 7.55 (ddd, 3J = 8.3 Hz, 3J = 8.3 Hz and 4J = 1.3 Hz, 1H), 7.38 (ddd, 3J = 8.3 Hz, 3J = 8.3 Hz and 4J = 1.1 Hz, 1H), 3.07 (t br, 3J = 6.7 Hz, 2H), 2.79 (t br, 3J = 6.1 Hz, 2H), 2.43 (s, 3H), 1.86 (m, 2 x 2H). 13C NMR (50.332 MHz, CDCI3) 6(ppm): 157.8,
145.5, 140.6, 128.0, 126.4, 128.6, 127.6, 124.8, 122.9, 34.2, 26.5, 22.8, 22.4, 12.9. HRMS (EI) m/z: calcd for [M]+ (found): 197.1204 (197.1198). Anal. Calcd for C14H15N (found): C 85.24 (85.25); H 7.66 (7.72); N 7.10 (6.78).
Step B) N xide-9-methyl-l,2,3,4-tetrahydroacridine: A dichloromethane solution (300 mL) of 3-chloroperbenzoic acid (26 g, 105 mmol) was slowly added to a dichloromethane solution (100 mL) of 9-methyl-l,2,3,4-tetrahydroacridine (10.2 g, 52 mmol) at 0°C. The mixture was stirred for 4 h at room temperature and quenched with an aqueous NaOH solution. The organic layers were further washed with water (5 x 100 mL) and dried over MgS04, and the solvent was removed under reduced pressure giving desired product as a brownish solid. (10.83 g, 98%). H NMR (200.131 MHz, CDCI3) 6(ppm): 8.77 (dd, 3 J = 8.5 Hz and J = 1.2 Hz, 1H), 7.97 (dd, 3 J = 8.5 Hz and 4J = 0.9 Hz, 1H), 7.70-7.50 (m, 2x lH), 3.19 (t, 3J = 6.1 Hz, 2H), 2.85 (t, 3J = 6.2 Hz, 2H), 2.51 (s, 3H), 1.88 (m, 2 x 2H). 13C NMR (50.332 MHz, CDCI3) 6(ppm): 146.7, 139.1, 131.6, 129.9, 127.7, 129.0, 127.3, 123.9, 119.6, 27.1, 26.6, 22.0, 21.4, 13.4. HRMS (EI) calcd for [M]+ (found): 213.1154 (213.1159).
Step C) 9-Methyl-l,2,3,4-tetrahydroacridin-4-ol: In a two-neck round-bottom flask equipped with a reflux condenser, /V-oxide-9-methyl-l,2,3,4-tetrahydroacridine (11.2 g, 52 mmol) was dissolved in dichloromethane (250 mL). Trifluoroacetic anhydride (17 mL, 120 mmol) was slowly added at room temperature (the reaction is exothermic). The solution was stirred for 5 h, and the solvent was evaporated. The crude solid was dissolved in methanol (50 mL) and saponified by an aqueous K2C03 solution (2 M, 150 mL); a brown solid precipitated. The methanol was removed under reduced pressure, and the product was extracted with dichloromethane (2 x 150 mL). The combined organic layers were washed with brine (2 x 50 mL), dried over magnesium sulfate, and evaporated to dryness. The desired product was recovered as a brown solid (9.4 g, 84%). JH NMR (200.131 MHz, CDCI3) 6(ppm): 7.96 (d, 3 J = 8.3 Hz, 1H), 7.91 (d, 3 J = 8.4 Hz, 1H), 7.58 (dd, 3 J = 8.3 Hz and 3 J = 8.1 Hz, 1H), 7.45 (dd, 3 J = 8.1 Hz and 3 J = 8.4 Hz, 1H), 4.95 (s br, 1H), 4.76 (dd, 3 J = 10.3 Hz and 3 J = 10.0 Hz, 1H), 2.89 (m, 2H), 2.54 (s, 3H), 2.40-1.92 (2xm, 2x lH), 1.82 (m, 2H). 13C NMR (50.332 MHz, CDCI3) 6(ppm): 159.2, 145.3, 142.0, 127.7, 127.3, 129.2, 128.5, 126.0, 123.5, 70.2, 30.3, 26.7, 19.6, 13.8. HRMS (EI) calcd for [M]+ (found): 213.1153 (213.1154).
Step D) 9-methyl-2,3-dihydro-lH-acridin-4-one: To a dichloromethane solution (300 mL) of
9-Methyl-l,2,3,4-tetrahydroacridin-4-ol (9.4 g, 44 mmol) was added Mn02 (23 g, 264 mmol) at room temperature, and the heterogeneous solution was allowed to stir for 2 days. After filtration over Celite, the solvent was evaporated. The crude dark solid was purified by column
chromatography (neutral alumina, dichloromethane as eluant). After evaporation of the solvent, the title compound was recovered as a brownish solid (5.41 g, 58%). H NMR (200.131 MHz, CDCI3) 6(ppm): 8.31 (dd, 3J = 8.1 Hz and 4J = 0.8 Hz, 1H), 7.94 (dd, 3J = 8.0 Hz and 4J = 1.4 Hz, 1H), 7.67-7.51 (m, 2H), 3.08 (t, 3 J = 6.1 Hz, 2H), 2.82 (t, 3J = 6.4 Hz, 2H), 2.60 (s, 3H), 2.22 (m, 2H). 13C NMR (50.332 MHz, CDCI3) 6(ppm): 198.2, 148.4, 146.9, 143.6, 134.1, 129.4, 132.4, 129.6, 128.9, 123.8, 40.2, 27.2, 22.4, 14.5. HRMS (EI) m/z calcd for [M]+ (found): 211.0997 (211.0989). Anal. Calcd for C14H13NO (found): C 79.59 (79.72); H 6.20 (6.28); N 6.63 (6.10).
Step A) 8-Ethoxy-2-methyl-5,6-dihydro-quinazoline: In a 25ml_ single-necked round- bottomed flask equipped with a condenser, a mixture of 6-[l-dimethylamino-meth-(E)-ylidene]- 2-ethoxy-cyclohex-2-enone (WO 2004/104007) (2.80 g; 14.3 mmol), acetamidine hydrochloride (1.70 g; 17.9 mmol) and potassium carbonate (2.73 g; 19.7 mmol) in absolute ethanol (50 mL) was stirred at reflux temperature over night for 16 hours. The reaction mixture was cooled to room temperature, poured onto water (400 mL) and extracted with dichloromethane (3x100 mL). The crude reaction mixture is purified via chromatography (combiflash RF, 70-100% ethylacetate in cydohexane) to give 8-Ethoxy-2-methyl-5,6-dihydro-quinazoline (1.47 g, 54% of theory) as brown oil. LC-MS (Method ZCQ) Rt = 1.21 min. , MS: (M++l) = 191. JH-NMR (CDCI3, 400 MHz): 1.49 (t, 3H); 2.41 (m, 2H); 2.74 (t, 2H); 2.75 (s, 3H); 3.99 (q, 2H); 5.46 (t,lH); 8.47 (s, 1H).
Step B) 2-Methyl-6,7-dihydro-5H-quinazolin-8-one: In a 25mL single-necked round- bottomed flask equipped with a condenser 8-Ethoxy-2-methyl-5,6-dihydro-quinazoline (1.30 g; 6.80 mmol) was dissolved in a mixture of acetic acid (31 mL) and water (3.1 mL). Stirring was continued under heating to reflux over night for 20 hours. TLC indicated that starting materials were consumed by this time. The mixture was cooled to room temperature, water and saturated NaHC03 was added. Extraction was carried out using dichloromethane (3x100 mL). The combined organic phases were washed with brine and then dried over sodium sulphate, filtered and the solvent was removed in vacuo. The crude is purified via chromatography (combiflash RF, 0-10% methanol in dichloromethane) to give 2-Methyl-6,7-dihydro-5H-quinazolin-8-one (764 mg; 69% of theory)as a brown oil. LC-MS (Method ZCQ) Rt = 0.91 min. , MS: (M++l) =
163. ^-NMR (CDCI3, 400 MHz): 2.24 (m, 2H); 2.83 (m, 2H); 2.84 (s, 3H); 3.01 (m, 2H); 8.85 (s, 1H).
Step A) 8-Ethoxy-2-phenyl-5,6-dihydro-quinazoline: In a 25mL single-necked round- bottomed flask equipped with a condenser, a mixture of 6-[l-dimethylamino-meth-(E)-ylidene]- 2-ethoxy-cyclohex-2-enone (WO 2004/104007) (0.51 g; 2.6mmol) and Benzamidine (0.38 g; 3.2mmol) in absolute ethanol (9 mL) is stirred at reflux temperature over night for 17 hours. The reaction mixture is evaporated to dryness, taken up in dichloromethane and purified via chromatography (combiflash RF, 0-40% ethylacetate in cyclohexane) to give 8-ethoxy-2-phenyl- 5,6-dihydro-quinazoline (0.50g, 63% of theory) as a white solid melting MP. 101-103°C. LC-MS (Method ZCQ) Rt = 1.84 min. , MS: (M++l) = 253. JH-NMR (CDCI3, 400 MHz): 1.48 (t, 3H); 2.45 (m, 2H); 2.81 (t, 2H); 4.02 (q, 2H); 5.52 (t,lH); 7.47 (m, 3H); 8.46 (m, 2H); 8.51 (s, 1H).
Step B) 2-Phenyl-6,7-dihydro-5H-quinazolin-8-one: In a 25ml_ single-necked round- bottomed flask equipped with a condenser, 8-ethoxy-2-phenyl-5,6-dihydro-quinazoline (0.45 g; 1.18 mmol) was dissolved in a mixture of acetic acid (9.5 mL) and water (0.95 mL). Stirring was continued under heating to reflux for 15 minutes. TLC indicated that starting materials were consumed by this time. The mixture was cooled to room temperature, water and saturated NaHC03 was added. Extraction was carried out using dichloromethane (3x20 mL). The combined organic phases were washed with brine and then dried over sodium sulphate, filtered and the solvent was removed in vacuo to give 2-Phenyl-6,7-dihydro-5H-quinazolin-8-one (380 mg; 95% of theory)as a beige solid MP. 154-156°C. LC-MS (Method ZCQ) Rt = 1.49 min. , MS: (M++l) = 225. l-NMR (CDCI3, 400 MHz): 2.25 (m, 2H); 2.86 (m, 2H); 3.03 (m, 2H); 7.51 (m, 3H); 8.52 (m, 2H); 8.94 (s, 1H).
Table 9: Physical data of compounds of formula (I):
Structure RT (mins) Molecular mp
(method) ion C°C)
P.l 1.40 442.49 resin
(UPLCl) ([M+l]+)
P.2 1.61 470 resin
(UPLCl) ([M+l]+)
1.17 470.49
(ZMD) ([M+1D
P.3 1.50 504.46 resin
(UPLCl) ([M+l]+)
P.4 . O^ /\ ,os 1.39 408 resin
(ZCQ) ([M+l]+)
P.5 1.24 382 oil
N' ^^^^ XN
(ZCQ) ([M+l]+)
LC-method used
Method A
Autopurification System from Waters: 2767 sample Manager, 2489 UV/Visible Detector, 2545 Quaternary Gradient Module.
Column: Phenomenex Synergi C18 Reversed Phase, 4 m particle size, 80 A, 75 x 30.00 mm,
100 mg of product dissolve in DMF injected
DAD Wavelength (nm): 220 and 254
Solvent Gradient:
A = water (Fluka Analytical)
B= Acetonitrile for praep. HPLC (Fluka Analytical)
Time A% B% Flow (mL/min)
0.00 90.0 10.0 50.00
0.01 90.0 10.0 50.00
6.00 60.0 40.0 50.00
7.90 60.0 40.0 50.00
8.00 0.0 100.0 50.00
8.90 0.0 100.0 50.00
9.00 90.0 10.0 50.00
9.50 90.0 10.0 50.00
9.55 90.0 10.0 50.00
Method ZMD
ZMD Mass Spectrometer from Waters (Single quadrupole mass spectrometer)
Instrument Parameter:
Ionisation method: Electrospray
Polarity: positive ions
Capillary (kV) 3.80, Cone (V), Extractor (V) 3.00, Source Temperature (°C) 150, Desolvation Temperature (°C) 350, Cone Gas Flow (L/Hr) OFF, Desolvation Gas Flow (L/Hr) 600
Mass range: 100 to 900 Da
HP 1100 HPLC from Agilent: solvent degasser, binary pump, heated column compartment and diode-array detector.
Column: Phenomenex Gemini C18, 3 mm particle size, 110 A 30 x 3 mm,
Temp: 60°C
DAD Wavelength range (nm): 200 to 500
Solvent Gradient:
A = water + 0.05 % HCOOH
B= Acetonitril/Methanol (4: 1, v:v) + 0.04 % HCOOH
Time A% B% Flow (mL/min)
0.00 95.0 5.0 1.700
2.00 0.0 100.0 1.700
2.80 0.0 100.0 1.700
2.90 95.0 5.0 1.700
3.00 95.0 5.0 1.700
Method ZCQ
ZQ Mass Spectrometer from Waters (Single quadrupole mass spectrometer)
Instrument Parameter:
Ionisation method: Electrospray
Polarity: positive ions
Capillary (kV) 3.00, Cone (V) 30.00, Extractor (V) 2.00, Source Temperature (°C) 100, Desolvation Temperature (°C) 250, Cone Gas Flow (L/Hr) 50, Desolvation Gas Flow (L/Hr) 400 Mass range: 100 to 900 Da
HP 1100 HPLC from Agilent: solvent degasser, quaternary pump (ZCQ) / binary pump (ZDQ), heated column compartment and diode-array detector.
Column: Phenomenex Gemini C18, 3 mm particle size, 110 A, 30 x 3 mm,
Temp: 60°C
DAD Wavelength range (nm): 200 to 500
Solvent Gradient:
A = water + 0.05 % HCOOH
B= Acetonitril/Methanol (4: 1, v:v) + 0.04 % HCOOH
Time A% B% Flow (mL/min)
0.00 95.0 5.0 1.700
2.00 0.0 100.0 1.700
2.80 0.0 100.0 1.700
2.90 95.0 5.0 1.700
3.00 95.0 5.0 1.700
Method UPLC1
ACQUITY SQD Mass Spectrometer from Waters (Single quadrupole mass spectrometer)
Ionisation method: Electrospray
Polarity: positive ions
Capillary (kV) 3.00, Cone (V) 20.00, Extractor (V) 3.00, Source Temperature (°C) 150, Desolvation Temperature (°C) 400, Cone Gas Flow (L/Hr) 60, Desolvation Gas Flow (L/Hr) 700 Mass range: 100 to 800 Da
DAD Wavelength range (nm): 210 to 400
Method Waters ACQUITY UPLC with the following HPLC gradient conditions
(Solvent A: Water/Methanol 9: 1,0.1% formic acid and Solvent B: Acetonitrile,0.1% formic acid)
Time (minutes) A (%) B (%) Flow rate (mL/min)
0 100 0 0.75
2.5 0 100 0.75
2.8 0 100 0.75
3.0 100 0 0.75
Type of column: Waters ACQUITY UPLC HSS T3; Column length: 30 mm; Internal diameter of column: 2.1 mm; Particle Size: 1.8 micron; Temperature: 60°C.
OA_3min_30V
ZQ Mass Spectrometer from Waters (Single quadrupole mass spectrometer):
Ionization method: Electrospray; Polarity: positive and negative ions; Capillary (kV): 3.00; Cone (V): 30.00; Extractor (V): 2.00; Source Temperature (°C): 100; Desolvation Temperature (°C): 250; Cone Gas Flow (L/Hr): 50; Desolvation Gas Flow (L/Hr): 400; Mass range: 100 to 900 Da
HP 1100 HPLC from Agilent:
Solvent degasser, binary pump, heated column compartment and diode-array detector; Column: Phenomenex Gemini C18, 3 - m, 30 x 3 mm; Temp: 60°C; DAD Wavelength range (nm): 210 to 500;
Solvent Gradient:
A = water + 5% MeOH + 0.05 % HCOOH
B= Acetonitrile + 0.05 % HCOOH
Time A% B% Flow (mL/min)
0.00 100 0 1.700
2.00 0 100 1.700
2.80 0 100 1.700
2.90 100 0 1.700
00 100 0 1.700
OA_Standard
ZQ Mass Spectrometer from Waters (Single quadrupole mass spectrometer)
Instrument Parameter:
Ionization method: Electrospray
Polarity: positive and negative ions
Capillary: 3.00 kV
Cone: 30 V
Extractor: 2.00 V
Source Temperature: 150°C,
Desolvation Temperature: 350C
Cone Gas Flow: 50 L/Hr
Desolvation Gas Flow: 400 L/Hr
Mass range: 100 to 900 Da
Acquity UPLC from Waters:
Binary pump, heated column compartment and diode-array detector.
Solvent degasser, binary pump, heated column compartment and diode-array detector.
Column: Waters UPLC HSS T3 , 1.8 - m, 30 x 2.1 mm,
Temp: 60 °C
DAD Wavelength range (nm): 210 to 500
Solvent Gradient:
A = H20 + 5% MeOH + 0.05 % HCOOH
B= Acetonitril + 0.05 % HCOOH
Time A% B% Flow (mL/min)
0.00 90 10 0.85
2.70 0 100.0 0.85
3.00 0 100.0 0.85
Biological examples:
Puccinia recondite f. sp. tri'ti'ci I wheat / leaf disc preventative (Brown rust):
Wheat leaf segments cultivated variety (cv) Kanzler were placed on agar in 24-well plates and sprayed with formulated test compound diluted in water at an application rate of 200ppm.
The leaf disks were inoculated with a spore suspension of the fungus 1 day after application. The inoculated leaf segments were incubated at 19°C and 75% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (7 - 9 days after application). The following compounds gave at least 80% control of Puccinia recondita f. sp. tritici: P.01, P.02, P.04, P.05, P.06, P.08, P.09. P.20, P.23, P.27, P.28, P.29, P.30.
Puccinia recondita f. sp. tritici I wheat / leaf disc curative ("Brown rust):
Wheat leaf segments cv Kanzler were placed on agar in 24-well plates. The leaf segments were inoculated with a spore suspension of the fungus. The plates were stored in darkness at 19°C and 75% relative humidity. The formulated test compound diluted in water was applied at an application rate of 200ppm 1 day after inoculation. The leaf segments were incubated at 19°C and 75% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (6 - 8 days after application). The following compounds gave at least 80% control of Puccinia recondita f. sp. tritici: P.01, P.02, P.04, P.05, P.06, P.08, P.09, P.11. P.20, P.23, P.27, P.28, P.29.
Phaeosphaeria nodorum (Seotoria nodorum) / wheat / leaf disc preventative (Glume blotch):
Wheat leaf segments cv Kanzler were placed on agar in a 24-well plate and sprayed with formulated test compound diluted in water at an application rate of 200ppm. The leaf disks are inoculated with a spore suspension of the fungus 2 days after application. The inoculated test leaf disks are incubated at 20°C and 75% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (5 - 7 days after application). The following compounds gave at least 80% control of Phaeosphaeria nodorum: P.01, P.02, P.04, P.05, P.06, P.08, P.09, P.ll, P.14, P.16, P.17, P.18P.19, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30.
Pyrenophora teres / barley / leaf disc preventative (Net blotch):
Barley leaf segments cv Hasso are placed on agar in a 24-well plate and sprayed with formulated test compound diluted in water at an application rate of 200ppm. The leaf segments are inoculated with a spore suspension of the fungus two days after application of the test solution. The inoculated leaf segments are incubated at 20°C and 65% relative humidity under a
light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (5 - 7 days after application). The following compounds gave at least 80% control of Pyrenophora teres: P.01, P.02, P.04, P.05, P.06, P.08, P.09, P.ll, P.14, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30.
Alternaria solani I tomato / leaf disc (early blight):
Tomato leaf disks cv Baby are placed on agar in 24-well plates (24-well format) and sprayed with the formulated test compound diluted in water at an application rate of 200ppm. The leaf disks are inoculated with a spore suspension of the fungus 2 days after application. The inoculated leaf disks are incubated at 23°C/21°C (day/night) and 80% relative humidity under a light regime of 12/12 h (light/dark) in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check disk leaf disks (5 - 7 days after application). The following compounds gave at least 80% control of Alternaria solani: P.08,P.29.
Pythium ultimum I liquid culture (seedling damping off):
Mycelia fragments and oospores of a newly grown liquid culture of the fungus were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96 well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal mycelia/spore mixture was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 2-3 days after application. The following compounds gave at least 80% control of Pythium ultimum: P.08,P.27, P.28.
Botryotinia fuckeliana (Botrytis cinerea) I liquid culture (Gray mould):
Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (Vogels broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 3-4 days after application. The following compounds gave at least 80% control of Botryotinia fuckeliana: P.01, P.02, P.03, P.04, P.05, P.06, P.07, P.09, P.10, P. ll, P.12, P.14, P.15. P.18, P.19, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30, P.31.
Glomerella lagenarium (Colletotrichum lagenarium) / liquid culture (Anthracnose) :
Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was
added. The test plates were incubated at 24°C and the inhibition of growth is measured photometrically 3-4 days after application. The following compounds gave at least 80% control of Glomerella lagenarium: P.01, P.02, P.03, P.04, P.06, P.08, P.09, P.ll, P.14, P.15. P.22, P.23, P.27, P.28, P.29, P.30.
Mycosphaerella arachidis CCercospora arachidicola) I liquid culture (early leaf spot):
Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates are incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application. The following compounds gave at least 80% control of Mycosphaerella arachidis: P.01, P.02, P.03, P.04, P.05, P.06, P.08, P.09, P.10, P. ll, P.12, P.14, P.15. P.18, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.31.
Mycosphaerella graminicola (Septoria tritici) I liquid culture (Septoria blotch):
Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application. The following compounds gave at least 80% control of Mycosphaerella graminicola: P.01, P.02, P.03, P.04, P.06, P.08, P.09, P.10, P.ll, P.12, P.14, P.15. P.18, P.19, P.20, P.21, P.22, P.23, P.27, P.28, P.29, P.30, P.31.
Gaeumannomyces graminis I liquid culture (Take-all of cereals):
Mycelial fragments of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96- well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores is added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application. The following compounds gave at least 80% control of Gaeumannomyces graminis: P.01, P.02, P.03, P.08, P.09, P. ll, P.14, P.15., P.20, P.21, P.22, P.23, P.27, P.31.
Thanatephorus cucumeris (Rhizoctonia solani) I liquid culture (foot rot, dampinq-off): Mycelia fragments of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of the test compounds into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal material was added. The test plates were incubated at 24°C and the
inhibition of growth was determined photometrically 3-4 days after application. The following compounds gave at least 80% control of Thanatephorus cucumeris: P.01, P.02, P.04, P.06, P.08, P.09, P.14, P.15.
Monographella nivalis (Microdochium nivale) I liquid culture (foot rot cereals'):
Conidia of the fungus from cryogenic storage were directly mixed into nutrient broth (PDB potato dextrose broth). After placing a DMSO solution of test compound into a 96-well microtiter plate at an application rate of 200ppm, the nutrient broth containing the fungal spores was added. The test plates were incubated at 24°C and the inhibition of growth was determined photometrically 4-5 days after application. The following compounds gave at least 80% control of Monographella nivalis: P.01, P.02, P.04, P.06, P.08, P.09. P.21, P.22, P.23,.28, P.29.
Blumeria graminisf. so. tritici (Erysiphe graminis f. sp. tritici) I wheat / leaf disc preventative (Powdery mildew on wheat):
Wheat leaf segments cv. Kanzler were placed on agar in a 24-well plate and sprayed with the formulated test compound diluted in water at an application rate of 200ppm. The leaf disks were inoculated by shaking powdery mildew infected plants above the test plates 1 day after application. The inoculated leaf disks were incubated at 20°C and 60% relative humidity under a light regime of 24 h darkness followed by 12h/12h (dark/light) in a climate chamber and the activity of a compound was assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check leaf segments (6 - 8 days after application). The following compounds gave at least 80% control of Blumeria graminis: P.01, P.02, P.04, P.05, P.06, P.08, P.09, P. ll. P.20, P.22, P.23, P.27, P.28, P.29, P.30, P.31.
Sclerotinia sclerotiorum / liquid culture (cottony rot)
Mycelia fragments of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate at an application rate of 200ppm (96-well format) the nutrient broth containing the fungal material is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 3-4 days after application. The following compounds gave at least 80% control of :_Sclerotinia sclerotiorum: P.04, P.06, P.09, P.15. P.23, P.27.
Magnaporthe grisea (Pyricularia oryzae) I rice / leaf disc preventative (Rice Blast)
Rice leaf segments cv. Ballila are placed on agar in a multiwell plate (24-well format) and sprayed with the formulated test compound diluted in water at an application rate of 200ppm. The leaf segments are inoculated with a spore suspension of the fungus 2 days after application. The inoculated leaf segments are incubated at 22°C and 80% rh under a light regime of 24 h
darkness followed by 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (5 - 7 days after application). The following compounds gave at least 80% control of_Magnaporthe grisea: P.04, P.05, P.06, P.08, P.09, P.ll, P.17. P.19, P.20, P.22, P.23, P.27, P.30.
Claims
Claims:
1. A compound of formula (I)
D1 represents N or C-Y2;
D2 represents N or C-Y5;
D3 represents N or C-Y7;
wherein at least one of D1, D2 or D3 is N;
wherein both D2 and D3 cannot be N;
A represents hydrogen, halogen, CN, OH, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, Ci-C8 alkylsulphonyl, NH2, Ci-Cio alkyl, C3-C8 cycloalkyi, C2-C8 alkenyl, C2-C8 alkynyl, (R10O)carbonyl(Ci- C4-alkyl), phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, d-C4 haloalkyl, Ci-C4 alkoxy, Ci-C4 haloalkoxy, C3-C6 cycloalkyi and a 5- or 6-membered heterocycle containing one to three heteroatoms independently selected from O, S and N, providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms;
or if D3 is C-Y7 then A and Y7 together may be -(G4)q-G5-G6-;
X represents X-2, X-3, X-4 or X-5:
#—∑—∑—# #— z— z— z— # #— z— z— z— z— #
X-2 X-3 X-4
#_z12_zU_z12_z13_zli_#
X-5
Z1, Z2, Z3, Z5, Z6, Z7, Z8, Z9, Z10, Z11, Z13 and Z14 independently of one another represent CR R2, C=CR3R4 or C=0;
Z4 and Z12 independently of one another represent C=CR3R4, CR5R6, SiR7R8 or C=0;
each R1 and R2 independently of one another represent hydrogen, halogen, OH, d-C4 alkyl, C1-C4 haloalkyi, C3-C6 cycloalkyi, C3-C6 halocycloalkyl, C1-C4 alkylthio, C1-C4 alkylsulphinyl, C1-C4 alkylsulphonyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, d-C4 alkoxy and C1-C4 haloalkoxy;
or R1 and R2 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyl group;
each R3 and R4 independently of one another represent hydrogen, halogen, C1-C4 alkyl or C1-C4 haloalkyi;
each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, Ci-
C4 alkyl, C1-C4 haloalkyi, C3-C6 cycloalkyi, C3-C6 halocycloalkyl, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, d-C4 alkoxy and C1-C4 haloalkoxy;
or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyl group;
wherein the groupings X-2, X-3, X-4 and X-5 may contain at most one ring which contains either only one of the radicals Z1 to Z14 or two radicals Z1 to Z14 or three radicals Z1 to Z14 or four radicals Z1 to Z14 as ring members; and wherein radicals Z1, Z2, Z3, Z5, Z6, Z9, Z10 and Z14 are not substituted by OH;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one another represent hydrogen, halogen, CN,
N02, Ci-C8 alkyl, Ci-C4-alkoxy-Ci-C4-alkyl, Ci-C4-alkoxy-Ci-C4-alkoxy-Ci-C4-alkyl, C3-C8 cycloalkyi, C2-C8 alkenyl, C2-C8 alkynyl, phenyl, pyridyl, pyrimidinyl, COR9, OR10, SH, Ci-C8 alkylthio, Ci-C8 alkylsulphinyl, CrC8 alkylsulphonyl, N(Rn)2, C02R10, 0(CO)R9, CON(Rn)2, NRnCOR9 or CR9N- OR10, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, pyrimidinyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci- C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy, Ci-C4 haloalkoxy, Ci-C4 alkylthio, Ci-C4 alkylsulphinyl and C1-C4 alkylsulphonyl;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from O, S, N and N(Rn), providing that the heterocycle does not contain adjacent oxygen atoms, adjacent sulphur atoms, or adjacent sulphur and oxygen atoms, and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6
and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy and C1-C4 haloalkoxy;
each R9 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C2- C8 alkenyl, C2-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy and C1-C4 haloalkoxy;
each R10 independently of one another represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, C3-C8 alkenyl, C3-C8 alkynyl, benzyl, phenyl or pyridyl, wherein the alkyl, cycloalkyi, alkenyl, alkynyl, phenyl, benzyl and pyridyl are optionally substituted by one or more groups
independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, d-C4-haloalkyl, C1-C4 alkoxy, Ci-C4 haloalkoxy and Ci-C4-alkoxy-Ci-C4-alkyl;
each R11 independently of one another represents hydrogen, OH, d-C8 alkyl, Ci-C8 alkoxy, Ci-C8-alkoxy-Ci-C4-alkyl, C3-C8 alkenyl, C3-C8 alkynyl, COR9 or phenyl, wherein the alkyl, alkoxy, alkenyl, alkynyl and phenyl are optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C1-C4 alkylthio, C1-C4 alkylsulphinyl and C1-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or C1-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5, B-6, B-7 or B-8:
B-1 B-2 B-3 B-4 B-5 B-6 B-7 B-8 wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy and C1-C4 haloalkoxy;
G1, G2, G4 and G5 independently of one another represent -C(R12R13)-, 0, N(R14) or S;
G3 and G6 independently of one another represent -C(R12R13)-, O, N(R14) or S;
or G1 and G2, or G2 and G3, or G1 and G1, or G4 and G5, or G5 and G6, or G4 and G4 together represent -CR12=CR13-;
each R12 and R13 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci-
C4 haloalkyi, Ci-C4 alkoxy or Ci-C4 haloalkoxy;
R14 represents hydrogen, OH, d-C4 alkyl, Ci-C4 alkoxy, C3-C6 cycloalkyi, Ci-C8 alkylcarbonyl or Ci-Cs haloalkylcarbonyl; and
p and q are each independently 0, 1 or 2;
or a salt or an N-oxide thereof.
2. A compound according to claim 1, wherein
A represents hydrogen, Ci-C8 alkyl, C3-C8 cycloalkyi, phenyl or pyridyl, wherein the alkyl, cycloalkyi, phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, OH, Ci-C4 alkoxy, d-C4 haloalkoxy and C3-C6 cycloalkyi;
X represents X-3;
Z3 and Z5 independently of one another represent methylene or halomethylene;
Z4 represent C=CR3R4, CR5R6 or SiR7R8;
each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl;
each R5, R6, R7 and R8 independently of one another represent hydrogen, halogen, OH, d- C4 alkyl, Ci-C4 haloalkyi, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy;
or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyi group or a C3-C6 halocycloalkyl group;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one represent hydrogen, halogen, OH, CN, d- C8 alkyl, Ci-Cs haloalkyi, Ci-Cs alkoxy, Ci-Cs haloalkoxy, Ci-Cs alkylthio, C3-C8 cycloalkyi, phenyl, pyridyl, N(Rn)2, or NRnCOR9, wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
or independently Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 together with the fragment of the ring to which they are attached may form a partially or fully unsaturated 5- to 7-membered carbocyclic ring or a 5- to 7-membered heterocyclic ring containing one to three heteroatoms independently selected from N and N(Rn)2 and wherein the ring formed by Y1 and Y2, Y2 and Y3, Y4 and Y5, Y5 and Y6 or Y6 and Y7 is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, Ci-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy and Ci-C4 haloalkoxy;
each R9 independently represents d-C8 alkyl or Ci-C8 haloalkyl;
each R11 independently of one another represents hydrogen, Ci-C8 alkyl, COR9 or phenyl, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C C4 alkyl, C C4 haloalkyl, d-C4 alkoxy, d-C4 haloalkoxy, d-C4 alkylthio, Ci-C4 alkylsulphinyl and Ci-C4 alkylsulphonyl;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or Ci-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5;
wherein the cycle formed is optionally substituted by one or more groups independently selected from halogen, methyl and halomethyl;
G1, G2, G3, G4, G5 and G6 independently of one another represent -C(R12R13)-;
p and q are each independently 0, 1 or 2. 3. A compound according to claim 1, wherein
A represents hydrogen, Ci-C4 alkyl, phenyl or pyridyl, wherein alkyl is optionally substituted by one or more groups independently selected from halogen, OH, d-C4 alkoxy and Ci-C4 haloalkoxy, and wherein phenyl and pyridyl are optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyl, OH, Ci-C4 alkoxy, d-C4 haloalkoxy and C3-C6 cycloalkyl;
X represents X-3;
Z3 and Z5 represent methylene;
Z4 represents C=CR3R4 or CR5R6 ;
each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl;
each R5 and R6 independently of one another represent hydrogen, halogen, OH, C1-C4 alkyl, C1-C4 haloalkyi, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyl group or a C3-C6 halocycloalkyl group;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one another represent hydrogen, CN, OH, halogen, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy, C1-C4 haloalkoxy, C3-C6 cycloalkyl, d-C4 alkylthio, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy and C1-C4 haloalkoxy;
each R9 independently represents d-C4 alkyl or C1-C4 haloalkyi;
each R11 independently of one another represents hydrogen, Ci-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, NH2, N02, OH, C1-C4 alkyl, C1-C4 haloalkyi, C1-C4 alkoxy and C1-C4 haloalkoxy;
wherein when two radicals R11 are attached to the same nitrogen atom, these radicals can be identical or different;
wherein when two radicals R11 are attached to the same nitrogen atom, both of these radicals cannot be OH, d-C4 alkoxy or C1-C4 haloalkoxy;
and wherein when two radicals R11 are attached to the same nitrogen atom, these two radicals together with the nitrogen atom to which they are attached may form a cycle B-1, B-2, B-3, B-4, B-5 wherein the cycle formed is optionally substituted by one or more independently selected from halogen, methyl and halomethyl;
G1, G2, G3, G4, G5 and G6 represent methylene;
p and q are each independently 0, 1 or 2.
4. A compound according to claim 1, wherein
A represents hydrogen, C1-C4 alkyl, phenyl or pyridin-2-yl wherein phenyl and pyridin-2-yl are optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy and halomethoxy;
X represents X-3;
Z3 and Z5 represent methylene;
Z4 represents C=CR3R4 or CR5R6 ;
each R3 and R4 independently of one another represent hydrogen, halogen, methyl or halomethyl;
each R5 and R6 independently of one another represent hydrogen, halogen, OH, d-C4 alkyl, C1-C4 haloalkyl, phenyl or CN, wherein the phenyl is optionally substituted by one or more groups independently selected from halogen, CN, methyl, halomethyl, methoxy or halomethoxy; or R5 and R6 together with the carbon atom to which they are attached may form a C3-C6 cycloalkyl group or a C3-C6 halocycloalkyl group;
Y1, Y2, Y3, Y4, Y5, Y6and Y7 independently of one another represent hydrogen, CN, OH, halogen, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C3-C6 cycloalkyl, d-C4 alkylthio, N(Rn)2, NRnCOR9 or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected selected from halogen, methyl, CN, methoxy, halomethyl and halomethoxy;
each R9 independently represents d-C4 alkyl or C1-C4 haloalkyl;
each R11 independently of one another represents hydrogen, Ci-C8 alkyl or phenyl, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, C1-C4 alkyl, Ci-C4 haloalkyl, Ci-C4 alkoxy and G-C4 haloalkoxy;
G1, G2, G3, G4, G5 and G6 represent methylene;
p and q are each independently 0, 1 or 2.
5. A compound according to any one of the preceding claims, wherein G1, G2 and G3 represent methylene.
6. A compound according to any one of the preceding claims, wherein p is 1.
8. A compound according to any one of claims 1-6 wherein A represents C1-C4 alkyl.
9. A compound according to any one of claims 1-8 wherein p is 1 and -G^G^G3- represents -CH2-CH2-CH2-.
A compound of formula (II)
(LG) (C-1 ) (C-2A) (C-2B)
R28 represents Ci-C4 alkyl, d-C4 haloalkyi or phenyl, wherein the phenyl is optionally substituted by one or two substituents independently selected from methyl, trihalomethyl, N02, CN, C1-C7 alkoxycarbonyl;
X" represents halogen;
R37 represents either A as defined herein for compounds of formula (I) in any one of claims 1 to 9 or the group R37
wherein X, G1, G2, G3, D1, D2, D3, Y1, Y3, Y4, Y6 and p are as defined for the compound of formula (I) in any one of claims 1 to 9;
or a salt or N-oxide thereof, or;
wherein X' represents one of the groupings X'-l, X'-2 or X'-3
wherein Z3, Z6, Z7, Z10, Z11 and Z12 are as defined for a compound of formula (I) in any one of claims 1 to 9;
R29 and R30 independently of one another represent hydrogen, halogen, Ci-C4 alkyl, Ci-C4 haloalkyi, phenyl or CN, wherein phenyl is optionally substituted by one or more groups independently selected from halogen, CN, Ci-C4 alkyl, Ci-C4 haloalkyi, d-C4 alkoxy and Ci-C4 haloalkyoxy; and
G1, G2, G3, D1, Y1, Y3 and p are as defined for a compound of formula (I) in any one of claims 1 to 9; or a salt or N-oxide thereof;
or a compound of formula (VIII)
(VIM)
wherein G1, G2, G3, D1, Y1, Y3 and p are as defined for a compound of formula (I) in any one of claims 1 to 9;
or a salt or N-oxide thereof, or;
wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined for a compound of formula I in any one of claims 1 to 9;
or a salt or N-oxide thereof, or;
a compound of formula XVI
wherein G1, G2, G3, p, D1, Y1 and Y3 are as defined herein for a compound of formula I in any one of claims 1 to 9;
R38 is hydrogen, CO-CH3, CO-CH2CH3, CO-CH2CH2CH3, CO-CH(CH3)2, CO-CF3, CO-CF2CF3, CH3, CH2CH3, CH2CH2CH3 or CH(CH3)2; or a salt or N-oxide thereof.
11. A process for the production of a compound of formula (I) as defined in any one of claims 1 to 9 comprising reacting a compound of formula (lib) with a compound of formula (X) as shown in scheme X
Scheme X
R represents Ci-C4-alkyl, Ci-C4-haloalkyl or phenyl, wherein the phenyl is optionally substituted by one or two substituents independently selected from methyl, trihalomethyl, N02, CN, C1-C7 alkoxycarbonyl; and
X, G1, G2, G3, Y1, Y3, Y4, Y6, D1, D2, D3, p and A are as defined for the compound of formula
(I) in any one of claims 1 to 9.
12. A fungicidal composition comprising a fungicidally effective amount of a compound of formula (I) as defined in any one of claims 1 to 9, optionally comprising an additional active ingredient.
13. A method of controlling phytopathogenic diseases on useful plants or on propagation material thereof, which comprises applying to the useful plants, the locus thereof or propagation material thereof a fungicidally effective amount of a compound of formula (I) as defined in any one of claims 1 to 9.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP11194620 | 2011-12-20 | ||
| EP11194620.8 | 2011-12-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013092460A1 true WO2013092460A1 (en) | 2013-06-27 |
Family
ID=47471802
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2012/075712 Ceased WO2013092460A1 (en) | 2011-12-20 | 2012-12-17 | Cyclic bisoxime microbicides |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2013092460A1 (en) |
Cited By (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9266886B2 (en) | 2014-02-03 | 2016-02-23 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9481674B1 (en) | 2016-06-10 | 2016-11-01 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9663515B2 (en) | 2014-11-05 | 2017-05-30 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9745253B2 (en) | 2015-03-13 | 2017-08-29 | Forma Therapeutics, Inc. | Alpha-cinnamide compounds and compositions as HDAC8 inhibitors |
| US9796710B2 (en) | 2014-10-14 | 2017-10-24 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9845308B2 (en) | 2014-11-05 | 2017-12-19 | Vitae Pharmaceuticals, Inc. | Isoindoline inhibitors of ROR-gamma |
| US10029995B2 (en) | 2015-09-03 | 2018-07-24 | Forma Therapeutics, Inc. | [6,6] fused bicyclic HDAC8 inhibitors |
| US10301261B2 (en) | 2015-08-05 | 2019-05-28 | Vitae Pharmaceuticals, Llc | Substituted indoles as modulators of ROR-gamma |
| US10829481B2 (en) | 2016-01-29 | 2020-11-10 | Vitae Pharmaceuticals, Llc | Benzimidazole derivatives as modulators of ROR-gamma |
| US10913739B2 (en) | 2017-07-24 | 2021-02-09 | Vitae Pharmaceuticals, LLC (121374) | Inhibitors of RORγ |
| US11008340B2 (en) | 2015-11-20 | 2021-05-18 | Vitae Pharmaceuticals, Llc | Modulators of ROR-gamma |
| US11186573B2 (en) | 2017-07-24 | 2021-11-30 | Vitae Pharmaceuticals, Llc | Inhibitors of ROR gamma |
Citations (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US437807A (en) | 1890-10-07 | Carl wustemann | ||
| DE2447258A1 (en) | 1974-10-03 | 1976-04-08 | Bayer Ag | Pyridyl alkyl-amidine derivs - hypoglycemics which influence flat metabolism for use in diabetes, hyperlipemia and adiposis |
| US3991065A (en) * | 1974-03-05 | 1976-11-09 | John Wyeth & Brother Limited | Tetrahydroquinoline derivatives |
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1995020569A1 (en) | 1994-01-27 | 1995-08-03 | Ciba-Geigy Ag | Process for the preparation of arylacetic ester derivatives via palladium-catalyzed cross coupling reaction |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| EP0792870A1 (en) | 1996-03-01 | 1997-09-03 | Hoechst Schering AgrEvo GmbH | Process for the preparation of ketoximethers by coupling with boronic acid derivatives |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO1999049314A1 (en) | 1998-03-27 | 1999-09-30 | The Regents Of The University Of California | Pharmacophore recombination for the identification of small molecule drug lead compounds |
| WO2001012604A1 (en) * | 1999-08-18 | 2001-02-22 | Aventis Cropscience Gmbh | Fungicides |
| WO2001025206A1 (en) | 1999-10-07 | 2001-04-12 | Aventis Cropscience Sa | Method for preparing hydroximic compounds for use as fungicides |
| WO2004104007A1 (en) | 2003-05-22 | 2004-12-02 | Pharmacia Italia S.P.A. | Pyrazolo-quinazoline derivatives,process for their preparation and their use as kinase inhibitors |
| US20050075366A1 (en) | 2002-05-21 | 2005-04-07 | Heidelbaugh Todd M. | 4-(2-Methyl-5,6,7,8-tetrahydro-quinolin-7-ylmethyl)-1,3-dihydro-imidazole-2-thione as specific alpha2B agonist and methods of using the same |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2006123145A1 (en) | 2005-05-18 | 2006-11-23 | F2G Ltd | Antifungal agents |
| US20070032470A1 (en) | 2005-08-04 | 2007-02-08 | Yong-Jin Wu | Novel phenylcarboxyamides as beta-secretase inhibitors |
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2007058504A1 (en) | 2005-11-21 | 2007-05-24 | Lg Life Sciences, Ltd. | Novel compounds as agonist for ppar gamma and ppar alpha, method for preparation of the same, and pharmaceutical composition containing the same |
| WO2007075459A2 (en) | 2005-12-16 | 2007-07-05 | E. I. Du Pont De Nemours And Company | 5-aryl isoxazolines for controlling invertebrate pests |
| WO2008013622A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2008074418A2 (en) | 2006-12-19 | 2008-06-26 | Bayer Cropscience Aktengesellschaft | Bisoximes as fungicides and arthropodicides |
| WO2008148570A1 (en) | 2007-06-08 | 2008-12-11 | Syngenta Participations Ag | Pyrazole carboxylic acid amides useful as microbiocides |
| WO2009010488A1 (en) | 2007-07-16 | 2009-01-22 | Novartis Ag | Heterocyclic compounds useful as mk2 inhibitors |
| WO2009036020A1 (en) | 2007-09-10 | 2009-03-19 | Curis, Inc. | Mek inhibitors containing a zinc binding moiety |
| WO2009049846A1 (en) | 2007-10-16 | 2009-04-23 | Syngenta Participations Ag | Insecticidal compounds |
| WO2010123791A1 (en) | 2009-04-22 | 2010-10-28 | E. I. Du Pont De Nemours And Company | Solid forms of an azocyclic amide |
| WO2011051243A1 (en) | 2009-10-29 | 2011-05-05 | Bayer Cropscience Ag | Active compound combinations |
| WO2011104089A1 (en) | 2010-02-25 | 2011-09-01 | Syngenta Participations Ag | Process for the preparation of isoxazoline derivatives |
| WO2012001040A1 (en) * | 2010-07-02 | 2012-01-05 | Syngenta Participations Ag | Novel microbiocidal dioxime ether derivatives |
-
2012
- 2012-12-17 WO PCT/EP2012/075712 patent/WO2013092460A1/en not_active Ceased
Patent Citations (31)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US437807A (en) | 1890-10-07 | Carl wustemann | ||
| US3991065A (en) * | 1974-03-05 | 1976-11-09 | John Wyeth & Brother Limited | Tetrahydroquinoline derivatives |
| DE2447258A1 (en) | 1974-10-03 | 1976-04-08 | Bayer Ag | Pyridyl alkyl-amidine derivs - hypoglycemics which influence flat metabolism for use in diabetes, hyperlipemia and adiposis |
| EP0353191A2 (en) | 1988-07-29 | 1990-01-31 | Ciba-Geigy Ag | DNA sequences encoding polypeptides having beta-1,3-glucanase activity |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| WO1995020569A1 (en) | 1994-01-27 | 1995-08-03 | Ciba-Geigy Ag | Process for the preparation of arylacetic ester derivatives via palladium-catalyzed cross coupling reaction |
| WO1995033818A2 (en) | 1994-06-08 | 1995-12-14 | Ciba-Geigy Ag | Genes for the synthesis of antipathogenic substances |
| EP0792870A1 (en) | 1996-03-01 | 1997-09-03 | Hoechst Schering AgrEvo GmbH | Process for the preparation of ketoximethers by coupling with boronic acid derivatives |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO1999049314A1 (en) | 1998-03-27 | 1999-09-30 | The Regents Of The University Of California | Pharmacophore recombination for the identification of small molecule drug lead compounds |
| WO2001012604A1 (en) * | 1999-08-18 | 2001-02-22 | Aventis Cropscience Gmbh | Fungicides |
| WO2001025206A1 (en) | 1999-10-07 | 2001-04-12 | Aventis Cropscience Sa | Method for preparing hydroximic compounds for use as fungicides |
| US20050075366A1 (en) | 2002-05-21 | 2005-04-07 | Heidelbaugh Todd M. | 4-(2-Methyl-5,6,7,8-tetrahydro-quinolin-7-ylmethyl)-1,3-dihydro-imidazole-2-thione as specific alpha2B agonist and methods of using the same |
| WO2004104007A1 (en) | 2003-05-22 | 2004-12-02 | Pharmacia Italia S.P.A. | Pyrazolo-quinazoline derivatives,process for their preparation and their use as kinase inhibitors |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2006123145A1 (en) | 2005-05-18 | 2006-11-23 | F2G Ltd | Antifungal agents |
| US20070032470A1 (en) | 2005-08-04 | 2007-02-08 | Yong-Jin Wu | Novel phenylcarboxyamides as beta-secretase inhibitors |
| WO2007048556A1 (en) | 2005-10-25 | 2007-05-03 | Syngenta Participations Ag | Heterocyclic amide derivatives useful as microbiocides |
| WO2007058504A1 (en) | 2005-11-21 | 2007-05-24 | Lg Life Sciences, Ltd. | Novel compounds as agonist for ppar gamma and ppar alpha, method for preparation of the same, and pharmaceutical composition containing the same |
| WO2007075459A2 (en) | 2005-12-16 | 2007-07-05 | E. I. Du Pont De Nemours And Company | 5-aryl isoxazolines for controlling invertebrate pests |
| WO2008013622A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2008013925A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2008074418A2 (en) | 2006-12-19 | 2008-06-26 | Bayer Cropscience Aktengesellschaft | Bisoximes as fungicides and arthropodicides |
| WO2008148570A1 (en) | 2007-06-08 | 2008-12-11 | Syngenta Participations Ag | Pyrazole carboxylic acid amides useful as microbiocides |
| WO2009010488A1 (en) | 2007-07-16 | 2009-01-22 | Novartis Ag | Heterocyclic compounds useful as mk2 inhibitors |
| WO2009036020A1 (en) | 2007-09-10 | 2009-03-19 | Curis, Inc. | Mek inhibitors containing a zinc binding moiety |
| WO2009049846A1 (en) | 2007-10-16 | 2009-04-23 | Syngenta Participations Ag | Insecticidal compounds |
| WO2010123791A1 (en) | 2009-04-22 | 2010-10-28 | E. I. Du Pont De Nemours And Company | Solid forms of an azocyclic amide |
| WO2011051243A1 (en) | 2009-10-29 | 2011-05-05 | Bayer Cropscience Ag | Active compound combinations |
| WO2011104089A1 (en) | 2010-02-25 | 2011-09-01 | Syngenta Participations Ag | Process for the preparation of isoxazoline derivatives |
| WO2012001040A1 (en) * | 2010-07-02 | 2012-01-05 | Syngenta Participations Ag | Novel microbiocidal dioxime ether derivatives |
Non-Patent Citations (101)
| Title |
|---|
| "EJEAFChe", ELECTRONIC JOURNAL OF ENVIRONMENTAL, AGRICULTURAL AND FOOD CHEMISTRY, vol. 5, no. 5, 2006, pages 1515 - 1521 |
| A. WOOD, COMPENDIUM OF PESTICIDE COMMON NAMES, 1995, Retrieved from the Internet <URL:http://www.alanwood.net/pesticides> |
| ACTA CRYSTALLOGRAPHICA, SECTION E: STRUCTURE REPORTS ONLINE, vol. E63, no. 10, 2007, pages 04080,S04080,1 - S04080,1 |
| ACTA CRYSTALLOGRAPHICA, SECTION E: STRUCTURE REPORTS ONLINE, vol. E64, no. 7, 2008, pages 01324,01324,1 - 01324,6 |
| ACTA CRYSTALLOGRAPHICA, SECTION E: STRUCTURE REPORTS ONLINE, vol. E64, no. 8, 2008, pages 01405,01405,1 - 01405,7 |
| ACTA CRYSTALLOGRAPHICA, SECTION E: STRUCTURE REPORTS ONLINE, vol. E65, no. 7, 2009, pages 01657 |
| ADVANCED SYNTHESIS & CATALYSIS, vol. 346, no. 13-15, 2004, pages 1798 - 1811 |
| ANGEWANDTE CHEMIE, vol. 45, no. 32, 2006, pages 5307 - 5311 |
| ANU NAIK ET AL: "Iron(ii)-bis(isonitrile) complexes: novel catalysts in asymmetric transfer hydrogenations of aromatic and heteroaromatic ketones", CHEMICAL COMMUNICATIONS, vol. 46, no. 25, 1 January 2010 (2010-01-01), pages 4475, XP055021267, ISSN: 1359-7345, DOI: 10.1039/c0cc00508h * |
| ARCHIV DER PHARMAZIE (WEINHEIM, GERMANY, vol. 340, no. 4, 2007, pages 202 - 208 |
| AUSTRALIAN JOURNAL OF CHEMISTRY, vol. 52, no. 8, 1999, pages 807 - 811 |
| BARK, THOMAS; THUMMEL, RANDOLPH P., INORGANIC CHEMISTRY, vol. 44, no. 24, 2005, pages 8733 - 8739 |
| BIOINORGANIC CHEMISTRY AND APPLICATIONS, vol. 1, no. 3-4, 2003, pages 299 - 308 |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 12, no. 3, 2002, pages 341 - 344 |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 18, no. 24, 2008, pages 6471 - 6475 |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 19, no. 10, 2009, pages 2654 - 2660 |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 19, no. 10, 2009, pages 2683 - 2687 |
| BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 19, no. 18, 2009, pages 5339 - 5345 |
| BIOORGANIC & MEDICINAL CHEMISTRY, vol. 15, no. 13, 2007, pages 4520 - 4527 |
| BIOORGANIC AND MEDICINAL CHEMISTRY LETTERS, vol. 19, no. 18, 2009, pages 5339 - 5345 |
| BULLETIN OF THE KOREAN CHEMICAL SOCIETY, vol. 15, no. 12, 1994, pages 1025 - 7 |
| C. D. S. TOMLIN: "The Pesticide Manual - A World Compendium; Thirteenth Edition;", THE BRITISH CROP PROTECTION COUNCIL |
| CANADIAN JOURNAL OF CHEMISTRY, vol. 85, no. 11, 2007, pages 913 - 922 |
| CHEM. BER., 1971, pages 2975 |
| CHEMICAL & PHARMACEUTICAL BULLETIN, vol. 51, no. 2, 2003, pages 138 - 151 |
| CHEMISTRY & BIOLOGY, vol. 9, no. 1, 2002, pages 113 - 129 |
| CHINESE JOURNAL OF CHEMISTRY, vol. 27, no. 1, 2009, pages 33 - 42 |
| CRAWFORD, JASON B.; CHEN, GANG; GAUTHIER, DAVID; WILSON, TREVOR; CARPENTER, BRYON; BAIRD, IAN R.; MCEACHERN, ERNIE; KALLER, ALAN;, ORGANIC PROCESS RESEARCH & DEVELOPMENT, vol. 12, no. 5, 2008, pages 823 - 830 |
| E. J. MCEACHERN ET AL: "Convenient Synthesis of 5,6,7,8-Tetrahydroquinolin-8-ylamine and 6,7-Dihydro-5 H -quinolin-8-one", SYNTHETIC COMMUNICATIONS, vol. 33, no. 20, 1 October 2003 (2003-10-01), pages 3497 - 3502, XP055021280, ISSN: 0039-7911, DOI: 10.1081/SCC-120024729 * |
| EUROPEAN JOURNAL OF MEDICINAL CHEMISTRY, vol. 40, no. 12, 2005, pages 1351 - 1358 |
| FARMACO, vol. 58, no. 9, 1985, pages 707 - 714 |
| GAZZETTA CHIMICA ITALIANA, vol. 84, 1954, pages 915 - 20 |
| GREENE, T. W.; WUTS, P. G. N.: "Protective Groups in Organic Synthesis", 2006, JOHN WILEY & SONS, INC |
| INDIAN JOURNAL OF CHEMISTRY, SECTION B: ORGANIC CHEMISTRY INCLUDING MEDICINAL CHEMISTRY, vol. 23B, no. 8, 1984, pages 728 - 32 |
| INORGANIC CHEMISTRY COMMUNICATIONS, vol. 12, no. 3, 2009, pages 234 - 236 |
| J. HETEROCYCL. CHEM., vol. 20, 1983, pages 649 - 653 |
| JOURNAL FUER PRAKTISCHE CHEMIE/CHEMIKER-ZEITUNG, vol. 335, no. 7, 1993, pages 623 - 7 |
| JOURNAL OF AGRICULTURAL AND FOOD CHEMISTRY, vol. 38, no. 3, 1990, pages 839 - 44 |
| JOURNAL OF AGRICULTURAL AND FOOD CHEMISTRY, vol. 56, no. 23, 2008, pages 11376 - 1139 |
| JOURNAL OF ANTIBIOTICS, vol. 61, no. 10, 2008, pages 603 - 614 |
| JOURNAL OF HETEROCYCLIC CHEMISTRY, vol. 16, no. 7, 1979, pages 1459 - 67 |
| JOURNAL OF HETEROCYCLIC CHEMISTRY, vol. 46, no. 1, 2009, pages 116 - 118 |
| JOURNAL OF HETEROCYCLIC CHEMISTRY, vol. 5, no. 2, 1968, pages 161 - 4 |
| JOURNAL OF MED. CHEM., vol. 52, no. 16, pages 5152 - 5163 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 20, no. 5, 1977, pages 718 - 21 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 34, no. 1, 1991, pages 57 - 65 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 42, no. 1, 1999, pages 153 - 163 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 44, no. 21, 2001, pages 3339 - 3342 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 49, no. 15, 2006, pages 4638 - 4649 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 49, no. 17, 2006, pages 5177 - 5186 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 50, no. 14, 2007, pages 3314 - 3321 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 51, no. 15, 2008, pages 4601 - 4608 |
| JOURNAL OF MEDICINAL CHEMISTRY, vol. 51, no. 20, 2008, pages 6421 - 6431 |
| JOURNAL OF MOLECULAR CATALYSIS B: ENZYMATIC, vol. 11, no. 4-6, 2001, pages 255 - 263 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 51, no. 26, 1986, pages 5047 - 50 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 52, no. 22, 1987, pages 4978 - 84 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 54, no. 14, 1989, pages 3394 - 403 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 69, no. 8, 2004, pages 2741 - 2749 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 72, no. 13, 2007, pages 4892 - 4899 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 73, no. 11, 2008, pages 4017 - 4026 |
| JOURNAL OF ORGANIC CHEMISTRY, vol. 73, no. 5, 2008, pages 2007 - 2010 |
| JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 130, no. 12, 2008, pages 4196 - 4201 |
| JOURNAL OF THE CHEMICAL SOCIETY, PERKIN TRANSACTIONS, vol. 2, no. 10, 2002, pages 1728 - 1739 |
| MCEACHERN, E. J.; YANG, W.; CHEN, G.; SKERLJ, R. T.; BRIDGER, G. J., SYNTHETIC COMMUNICATIONS, vol. 33, no. 20, 2003, pages 3497 - 350 |
| MOLECULES, vol. 10, no. 11, 2005, pages 1399 - 1408 |
| MOLECULES, vol. 10, no. 7, 2005, pages 747 - 754 |
| NATURE CHEMICAL BIOLOGY, vol. 5, no. 6, 2009, pages 407 - 413 |
| ORGANIC LETTERS, vol. 10, no. 2, 2008, pages 285 - 288 |
| ORGANIC PREPARATIONS AND PROCEDURES INTERNATIONAL, vol. 30, no. 2, 1998, pages 195 - 202 |
| ORGANIC PREPARATIONS AND PROCEDURES INTERNATIONAL, vol. 32, no. 2, 2000, pages 153 - 159 |
| ORGANIC PREPARATIONS AND PROCEDURES INTERNATIONAL, vol. 41, no. 4, 2009, pages 301 - 307 |
| PROC. BCPC, INT. CONGR., vol. 1, 2003, pages 93 |
| RUSSIAN JOURNAL OF ORGANIC CHEMISTRY, vol. 42, no. 5, 2006, pages 735 - 738 |
| RUSSIAN JOURNAL OF ORGANIC CHEMISTRY, vol. 43, 2007, pages 181 - 183 |
| RUSSIAN JOURNAL OF ORGANIC CHEMISTRY, vol. 43, no. 3, 2007, pages 449 - 453 |
| SCIENTIA PHARMACEUTICA, vol. 66, no. 1, 1998, pages 9 - 21 |
| SYNLETT, 2001, pages 1557 - 1558 |
| SYNLETT, 2001, pages 931 - 936 |
| SYNTHESIS, 1964, pages 3397 |
| SYNTHESIS, 1964, pages 3407 |
| SYNTHESIS, 1984, pages 1032 - 3 |
| SYNTHESIS, 1990, pages 366 - 8 |
| SYNTHESIS, 1996, pages 1012 - 1018 |
| SYNTHESIS, 1997, pages 38 - 40 |
| SYNTHESIS, 2008, pages 2055 - 2064 |
| SYNTHETIC COMMUN., vol. 10, 1998, pages 1743 - 1753 |
| SYNTHETIC COMMUNICATIONS, vol. 19, no. 18, 1989, pages 3129 - 38 |
| SYNTHETIC COMMUNICATIONS, vol. 19, no. 3-4, 1989, pages 339 - 44 |
| SYNTHETIC COMMUNICATIONS, vol. 28, no. 14, 1998, pages 2621 - 2633 |
| SYNTHETIC COMMUNICATIONS, vol. 33, no. 4, 2003, pages 543 - 546 |
| SYNTHETIC COMMUNICATIONS, vol. 37, no. 5, 2007, pages 737 - 742 |
| SYNTHETIC COMMUNICATIONS, vol. 37, no. 7, 2007, pages 1155 - 1165 |
| TAKAO SAKAMOTO ET AL: "Studies on Pyrimidine Derivatives. XXXIII. Synthesis of Alkyl Pyrimidinyl Ketones by Means of Nitrosation of Alkylpyrimidines", CHEM. PHARM. BULL, vol. 31, no. 1983, 1 December 1983 (1983-12-01), pages 4554 - 4560, XP055021271 * |
| TALANTA, vol. 16, no. 3, 1969, pages 448 - 52 |
| TETRAHEDRON LETT, 1986, pages 2567 |
| TETRAHEDRON LETT, 1994, pages 2255 - 2264 |
| TETRAHEDRON LETTERS, vol. 43, no. 40, 2002, pages 7189 - 7191 |
| TETRAHEDRON, vol. 43, no. 11, 1987, pages 2577 - 92 |
| TETRAHEDRON, vol. 53, no. 15, 1997, pages 5485 - 5492 |
| TETRAHEDRON: ASYMMETRY, vol. 14, no. 11, 2003, pages 1559 - 1563 |
| TETRAHEDRON: ASYMMETRY, vol. 8, no. 2, 1997, pages 253 - 263 |
Cited By (27)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9266886B2 (en) | 2014-02-03 | 2016-02-23 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US11535614B2 (en) | 2014-02-03 | 2022-12-27 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9624217B2 (en) | 2014-02-03 | 2017-04-18 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10807980B2 (en) | 2014-02-03 | 2020-10-20 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10399976B2 (en) | 2014-02-03 | 2019-09-03 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10047085B2 (en) | 2014-02-03 | 2018-08-14 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US9796710B2 (en) | 2014-10-14 | 2017-10-24 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10087184B2 (en) | 2014-10-14 | 2018-10-02 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of RORγ |
| US9845308B2 (en) | 2014-11-05 | 2017-12-19 | Vitae Pharmaceuticals, Inc. | Isoindoline inhibitors of ROR-gamma |
| US9663515B2 (en) | 2014-11-05 | 2017-05-30 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US11001583B2 (en) | 2014-11-05 | 2021-05-11 | Vitae Pharmaceuticals, Llc | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US11919839B2 (en) | 2015-03-13 | 2024-03-05 | Valo Health, Inc. | Alpha-cinnamide compounds and compositions as HDAC8 inhibitors |
| US10266487B2 (en) | 2015-03-13 | 2019-04-23 | Forma Therapeutics, Inc. | Alpha-cinnamide compounds and compositions as HDAC8 inhibitors |
| US10988441B2 (en) | 2015-03-13 | 2021-04-27 | Valo Early Discovery, Inc. | Alpha-cinnamide compounds and compositions as HDAC8 inhibitors |
| US9745253B2 (en) | 2015-03-13 | 2017-08-29 | Forma Therapeutics, Inc. | Alpha-cinnamide compounds and compositions as HDAC8 inhibitors |
| US10508077B2 (en) | 2015-03-13 | 2019-12-17 | Forma Therapeutics, Inc. | Alpha-cinnamide compounds and compositions as HDAC8 inhibitors |
| US10301261B2 (en) | 2015-08-05 | 2019-05-28 | Vitae Pharmaceuticals, Llc | Substituted indoles as modulators of ROR-gamma |
| US10829448B2 (en) | 2015-08-05 | 2020-11-10 | Vitae Pharmaceuticals, Llc | Substituted benzoimidazoles as modulators of ROR-γ |
| US10370343B2 (en) | 2015-09-03 | 2019-08-06 | Forma Therapeutics, Inc. | [6,6] Fused bicyclic HDAC8 inhibitors |
| US10829460B2 (en) | 2015-09-03 | 2020-11-10 | Valo Early Discovery, Inc. | [6,6] fused bicyclic HDAC8 inhibitors |
| US11414392B2 (en) | 2015-09-03 | 2022-08-16 | Valo Health, Inc. | [6,6] fused bicyclic HDAC8 inhibitors |
| US10029995B2 (en) | 2015-09-03 | 2018-07-24 | Forma Therapeutics, Inc. | [6,6] fused bicyclic HDAC8 inhibitors |
| US11008340B2 (en) | 2015-11-20 | 2021-05-18 | Vitae Pharmaceuticals, Llc | Modulators of ROR-gamma |
| US10829481B2 (en) | 2016-01-29 | 2020-11-10 | Vitae Pharmaceuticals, Llc | Benzimidazole derivatives as modulators of ROR-gamma |
| US9481674B1 (en) | 2016-06-10 | 2016-11-01 | Vitae Pharmaceuticals, Inc. | Dihydropyrrolopyridine inhibitors of ROR-gamma |
| US10913739B2 (en) | 2017-07-24 | 2021-02-09 | Vitae Pharmaceuticals, LLC (121374) | Inhibitors of RORγ |
| US11186573B2 (en) | 2017-07-24 | 2021-11-30 | Vitae Pharmaceuticals, Llc | Inhibitors of ROR gamma |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20110046088A1 (en) | Iminipyridine derivatives and their uses as microbiocides | |
| US10040806B2 (en) | Microbiocidally active benzoxaboroles | |
| WO2015055764A1 (en) | 3-methanimidamid-pyridine derivatives as fungicides | |
| WO2013092460A1 (en) | Cyclic bisoxime microbicides | |
| EP2951167A1 (en) | Novel microbiocides | |
| WO2012066122A1 (en) | 2 - (pyridin- 2 -yl) -quinazoline derivatives and their use as microbicides | |
| WO2014173880A1 (en) | Novel microbiocides | |
| WO2017067837A1 (en) | Microbiocidal phenylamidine derivatives | |
| WO2012069601A1 (en) | Substituted quinazolines as fungicides | |
| GB2503789A (en) | Quinazoline derivatives as antifungal agents | |
| WO2015003991A1 (en) | Novel microbiocides | |
| WO2014167133A1 (en) | Fungicides comprising boron | |
| US20140309244A1 (en) | Novel microbiocides | |
| EP3027592B1 (en) | Novel microbiocides | |
| EP2595985A1 (en) | Isoxazole, isothiazole, furane and thiophene compounds as microbicides | |
| WO2012117021A2 (en) | Novel microbiocidal oxime ethers | |
| WO2012062844A1 (en) | Novel microbiocides | |
| US20130210836A1 (en) | Microbicides | |
| WO2013026900A1 (en) | Pyridine derivatives as microbiocides | |
| WO2014147009A1 (en) | Novel microbiocides | |
| WO2012143395A1 (en) | 4,5-dihydro-isoxazole derivatives as fungicides | |
| WO2013139822A1 (en) | Novel bisoxime microbiocides | |
| WO2016038143A1 (en) | Microbiocidal 4-(imidazo[4,5-c]pyridin-2-yl)-1,2,5-oxadiazol-3- amine compounds having an oxime group in position 7 | |
| WO2012056003A1 (en) | Novel microbicides | |
| WO2015011194A1 (en) | Novel microbiocides |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12809240 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12809240 Country of ref document: EP Kind code of ref document: A1 |