WO2008130949A2 - Modulating nurr1 expression in a cell - Google Patents
Modulating nurr1 expression in a cell Download PDFInfo
- Publication number
- WO2008130949A2 WO2008130949A2 PCT/US2008/060392 US2008060392W WO2008130949A2 WO 2008130949 A2 WO2008130949 A2 WO 2008130949A2 US 2008060392 W US2008060392 W US 2008060392W WO 2008130949 A2 WO2008130949 A2 WO 2008130949A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- nurrl
- seq
- nucleic acid
- vector
- expression
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70567—Nuclear receptors, e.g. retinoic acid receptor [RAR], RXR, nuclear orphan receptors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
- C12N15/113—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing
- C12N15/1138—Non-coding nucleic acids modulating the expression of genes, e.g. antisense oligonucleotides; Antisense DNA or RNA; Triplex- forming oligonucleotides; Catalytic nucleic acids, e.g. ribozymes; Nucleic acids used in co-suppression or gene silencing against receptors or cell surface proteins
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2217/00—Genetically modified animals
- A01K2217/05—Animals comprising random inserted nucleic acids (transgenic)
- A01K2217/054—Animals comprising random inserted nucleic acids (transgenic) inducing loss of function
- A01K2217/058—Animals comprising random inserted nucleic acids (transgenic) inducing loss of function due to expression of inhibitory nucleic acid, e.g. siRNA, antisense
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2227/00—Animals characterised by species
- A01K2227/10—Mammal
- A01K2227/105—Murine
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2267/00—Animals characterised by purpose
- A01K2267/03—Animal model, e.g. for test or diseases
- A01K2267/035—Animal model for multifactorial diseases
- A01K2267/0356—Animal model for processes and diseases of the central nervous system, e.g. stress, learning, schizophrenia, pain, epilepsy
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/11—Antisense
- C12N2310/111—Antisense spanning the whole gene, or a large part of it
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2310/00—Structure or type of the nucleic acid
- C12N2310/10—Type of nucleic acid
- C12N2310/12—Type of nucleic acid catalytic nucleic acids, e.g. ribozymes
- C12N2310/121—Hammerhead
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2799/00—Uses of viruses
- C12N2799/02—Uses of viruses as vector
- C12N2799/021—Uses of viruses as vector for the expression of a heterologous nucleic acid
- C12N2799/025—Uses of viruses as vector for the expression of a heterologous nucleic acid where the vector is derived from a parvovirus
Definitions
- the invention relates generally to the fields of molecular biology, gene therapy, and medicine. More particularly, the invention relates to methods and compositions for modulating Nurrl expression in a cell. BACKGROUND
- Nurrl is a transcription factor that belongs to the nuclear receptor superfamily. It has been demonstrated that Nurrl is essential for the development of ventral dopaminergic neurons. The role of Nurrl in adult animals is unknown, however, because Nurrl knock-out mice die after birth. Nurrl is suspected of playing a role in Parkinson's disease because a mutation in a familial form of Parkinson's disease was found in the Nurrl gene. Nurrl is also suspected of playing a role in learning and memory. Because to dopaminergic system is involved in drug addiction, it is possible that Nurrl plays a role in this disorder as well. A means for studying the role of Nurrl in the adult brain (e.g. maintenance of the adult dopaminergic phenotype), and in several neurological diseases, is thus highly desirable.
- the invention relates to compositions and methods for modulating Nurrl expression in a cell, including a cell in a subject. Somatic knock-outs of Nurrl in the adult rat midbrain were performed using viral-delivered ribozymes, and overexpression of Nurrl in the adult rat brain was achieved using a recombinant Adeno- Associated Virus (AAV) vector encoding Nurrl.
- AAV Adeno- Associated Virus
- the invention features an isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of : SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3.
- a vector having at least one isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of : SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO: 3.
- the vector can be a recombinant AAV vector, e.g., a serotype 5 recombinant AAV vector.
- the invention also features a recombinant AAV vector including a nucleotide sequence encoding Nurrl .
- the invention in another aspect, relates to a method for modulating Nurrl expression in a eukaryotic cell.
- the method includes the steps of: a) providing at least one isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3; and b) contacting the cell with the at least one isolated nucleic acid under conditions in which the nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 can be expressed.
- Modulating Nurrl expression in a eukaryotic cell can include decreasing Nurrl expression in the eukaryotic cell.
- the invention features a method of expressing Nurrl in the hippocampus of a subject.
- the method includes the steps of: a) providing the subject; and cO administering to the subject at least one isolated nucleic acid that encodes Nurrl protein.
- the at least one isolated nucleic acid can be within a vector, e.g., within a recombinant AAV vector.
- the recombinant AAV vector can be serotype 5, for example.
- Another method within the invention includes a step of administering to a cell a recombinant AAV virion including: (a) a nucleotide sequence that when expressed modulates Nurrl expression in the cell, the nucleotide sequence interposed between a first AAV inverted terminal repeat and second AAV inverted terminal repeat: and (b) at least one AAV serotype 5 capsid protein.
- the nucleotide sequence can be one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3, and a nucleic acid that encodes Nurrl protein.
- nucleic acid means a chain of two or more nucleotides such as RNA (ribonucleic acid) and DNA (deoxyribonucleic acid).
- a “purified” nucleic acid molecule is one that has been substantially separated or isolated away from other nucleic acid sequences in a cell or organism in which the nucleic acid naturally occurs (e.g., 30, 40, 50, 60, 70, 80, 90, 95, 96, 97, 98, 99, 100% free of contaminants).
- the term includes, e.g., a recombinant nucleic acid molecule incorporated into a vector, a plasmid, a virus, or a genome of a prokaryote or eukaryote.
- a recombinant nucleic acid molecule incorporated into a vector, a plasmid, a virus, or a genome of a prokaryote or eukaryote.
- “Nurrl gene” or “Nurrl nucleic acid” is meant a native Nurrl - encoding nucleic acid sequence, genomic sequences from which Nurrl cDNA can be transcribed, and/or allelic variants and homologues of the foregoing.
- the terms encompass double-stranded DNA, single-stranded DNA, and RNA.
- protein or “polypeptide” are used synonymously to mean any peptide-linked chain of amino acids, regardless of length or post-translational modification, e.g., glycosylation or phosphorylation.
- Neuronal protein an expression product of a Nurrl gene: or a protein that shares at least 65% (but preferably 75, 80, 85, 90, 95, 96, 97, 98, or 99%) amino acid sequence identity with Nurrl (e.g., accession number NM 019328 in rat) and displays a functional activity of Nurrl .
- a "functional activity" of a protein is any activity associated with the physiological function of the protein.
- Nurrl can mean a native Nurrl -encoding nucleic acid sequence, genomic sequences from which Nurrl cDNA can be transcribed, and/or allelic variants and homologues of the foregoing, as well as an expression product of a Nurrl gene, or a protein that shares at least 65% (but preferably 75, 80, 85, 90, 95, 96, 97, 98, or 99%) amino acid sequence identity with Nurrl )e.g., accession number NM Ol 9328 in rat) and displays a functional activity of Nurrl.
- nucleic acid molecule or polypeptide when referring to a nucleic acid molecule or polypeptide, term “native” refers to a naturally-occurring (e.g., a wild-type; "WT”) nucleic acid or polypeptide.
- WT wild-type
- hybridization or “hybridizing” is meant the pairing of complementary RNA and DNA strands, or the pairing of complementary DNA single strands.
- sequence identity means the percentage of identical subunits at corresponding positions in two sequences when the two sequences are aligned to maximize subunit matching, i.e., taking into account gaps and insertions. Sequence identity can be measured using sequence analysis software (e.g., Sequence Analysis Software Package from Accelrys CGC, San Diego, CA).
- vector refers to a nucleic acid molecule capable of transporting another nucleic acid to which it has been linked.
- An example of a type of vector is an episome, i.e., a nucleic acid capable of extra-chromosomal replication.
- Another example of a vector is a viral vector (e.g., recombinant AAV vector).
- Vectors capable of directing the expression of genes to which they are operatively linked are referred to herein as "expression vectors.”
- a first nucleic acid sequence is "operably" linked with a second nucleic acid sequence when the first nucleic acid sequence is place in a functional relationship with the second nucleic acid sequence.
- a promoter is operable linked to a coding sequence if the promoter affects the transcription or expression of the coding sequence.
- operably linked nucleic acid sequences are contiguous and, where necessary to join two protein coding regions, in reading frame.
- expression control sequence refers to a nucleic acid that regulates the replication, transcription and translation of a coding sequence in a recipient cell.
- expression control sequences include promoter sequences, polyadenylation (pA) signals, introns, transcription termination sequences, enhancers, upstream regulatory domains, origins of replication, and internal ribosome entry sites ("IRES").
- promoter is used herein to refer to a DNA regulatory sequence to which RNA polymerase binds, initiating transcription of a downstream (3' direction) coding sequence.
- pseudotyped is meant a nucleic acid or genome derived from a first AAV serotype that is encapsidated or packaged by an AAV capsid containing at least one AAV capsid protein of a second serotype.
- AAV inverted terminal repeats AAV terminal repeats, “ITRs” and “TRs” are meant those sequences required in cis for replication and packaging of the AAV virion including any fragments or derivatives of an ITR which retain activity of a full-length or WT ITR.
- rAAV vector and “recombinant AAV vector” refer to a recombinant nucleic acid derived from an AAV serotype, including without limitation, AAVl, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, etc.
- rAAV vectors can have one or more of the AAV WT genes deleted in Whole or in part, preferably the rep and/or cap genes, but retain functional flanking ITR sequences.
- a “recombinant AAV virion” or “rAAV virion” is defined herein as an infectious, replication-defective virus composed of and AAV protein shell encapsulating a heterologous nucleotide sequence that is flanked on both sides by AAV ITRs.
- FIG. 1 is a graph showing relative abundance of the Nurrl mRNA in four rats infected with the AAV5-Rzl844 virus. Rats were infected with the virus in the ipsilateral side, the contralateral side being the uninfected side.
- FIG. 2 is a graph showing relative abundance of the Nurrl mRNA in eight rats infected with the AAV5-Rzl844 virus. Rats were infected with the virus in the ipsilateral side, the contralateral side being the uninfected side.
- FIG. 3 shows AAVl, AAV2 and AAV5 transduction in the substantia nigra of adult rats.
- Panels A, D, G show a montage of rostral to caudal coronal sections to illustrate the extent of expression of GFP in the substantia nigra with AAVl , AA V2 or AAV5.
- Panels C-O high magnification of the transduced areas on B, G, L. J.
- the dotted line indicates that the number of cells and the mean estimated volume counted for AAVl were done for the entire midbrain, whereas for AA V2 and AAV5, cells in the substantia nigra were counted. Number of cells transduced with each virus pseudotype estimated from stereological counts. The dots individual animals.
- AAVl shows a higher transduction rate through the entire midbrain area (A, B, P, Q).
- AA V2 number of cells and volume
- SN pars compacta of the substantia nigra
- AAV5 transduces the entire SN (K, L, P, Q).
- FIG. 4 is a photograph of an electrophoretic gel showing cleavage of Nurrl mRNA by ten ribozymes. Of these, three were found to cleave the in vitro transcribed full- length Nurl mRNA. The three ribozymes that cut are indicated on the top: Nurrl 1363 hh, Nurrl 1884 hh, andNurrl 1869hh. In the control lane the labeled IVR mRNA was incubated with no ribozymes under the same conditions. Arrow on the far left indicates the size of the full-length Nurrl mRNA. The arrows after the third lane show the size of the cleaved fragments. The larger cleaved fragments for 1844 and 1869 barely resolved in this gel (they can be appreciated just underneath the full length band). Only the smaller fragments of the mRNA are marked by arrows (arrows 1844 and 1869).
- FIG. 5 is a graph showing the effects of Nurrl ribozymes in the transcriptional activity of endogenous Nurrl.
- Transient transfection in PC 12 cells of the reporter 3x NBRE tk luciferase plus control expression vector (endo) or ribozyme (Rz) (1363, 1844 or 1869).
- the vectors were transfected at a 1 :2 molar ratio of reporter DNA to ribozyme, as indicated.
- Two of the ribozymes caused an increase in the transcriptional activation of the reporter gene. Only Rz 1844hh decreases Nurrl transactivation by almost half- fold.
- the invention encompasses compositions and methods relating to the use of rAAV-based vectors encoding Nurrl and rAAV-based vectors encoding ribozymes against Nurrl for modulating Nurrl expression in a eukaryotic cell.
- the below described preferred embodiments illustrate adaptations of these compositions and methods. Nonetheless, from the description of these embodiments, other aspects of the invention can be made and/or practiced based on the description provided below.
- the invention provides nucleic acids for modulating Nurrl expression in a cell.
- a typical nucleic acid as described herein for modulating Nurrl in a cell is an isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of : SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3.
- the sequences of SEQ ID :1, SEQ ID NO:2, and SEQ ID NO:3 are ribozyme sequences that decreased or prevented Nurrl expression in rodents as described in the Examples section below.
- Additional nucleic acids of the invention are variants of SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 that differ from SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 in one or more bases and that substantially maintain a functional activity (e.g., decreasing or preventing Nurrl expression in a cell) of SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3.
- Nucleic acids for modulating Nurrl expression in a cell can be delivered to a cell or a subject by any suitable route, but are typically delivered by a viral vector (e.g., recombinant AAV vector). Modulating Nurrl Expression In A Cell
- a typical method includes the steps of: a) providing at least one isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 and b) contacting the cell with the at least one isolated nucleic acid under conditions in which the nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 can be expressed.
- modulating Nurrl expression involves decreasing or preventing Nurrl expression in the eukaryotic cell, hi the Examples described herein, ribozyme molecules designed to catalytically cleave Nurrl mRNA transcripts (e.g. SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3) were used to decrease translation of Nurrl mRNA and expression of Nurrl protein (see, e.g. Lewin AS, and Hauswirth WW, Trends MoI Med 7:221-228, 2001; and Lewin et al, Nat. Med 6:967-971, 1998).
- SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 were used to decrease translation of Nurrl mRNA and expression of Nurrl protein (see, e.g. Lewin AS, and Hauswirth WW, Trends MoI Med 7:221-228, 2001; and Lewin et al, Nat. Med 6:967-971, 1998).
- ribozymes that cleave mRNA at site-specific recognition sequences can be used to destroy Nurrl mRNAs
- the use of hammerhead ribozymes may be preferred some embodiments.
- Hammerhead ribozymes cleave mRNAs at locations dictated flanking regions that form complementary base pairs with the target mRNA.
- the construction and production of hammerhead ribozymes is well known in the art and are described more fully in Vazquez- Tello et al., Nucleic Acid Res. 30(7): 1606-1612, 2002; Canny et al., J. Am Chem Soc.
- Ribozymes as described herein can be delivered to a cell by any suitable means, such as a vector (e.g., recombinant AAV vector).
- a vector e.g., recombinant AAV vector.
- Methods of increasing Nurrl expression in a cell are also encompassed by the invention. Such methods typically include delivering to the cell a nucleic acid that encodes Nurrl protein.
- a method of expressing Nurrl in the hippocampus of a subject includes the steps of providing the subject and administering to the subject at least one isolated nucleic acid that encodes Nurrl protein. rAAV Vectors And Virions
- the nucleic acids of the invention may be incorporated into vectors and/or virions in order to facilitate their introduction into a cell.
- rAAV vectors useful in the invention are recombinant nucleic acid constructs that include (1) a heterologous sequence to be expressed (e.g., Nurrl, ribozymes against Nurrl, etc.) and (2) viral sequences that facilitate expression of the heterologous sequence.
- the viral sequences may include those sequences of AAV that are required in cis for replication and packaging (e.g., functional TRs) of the DNA into a virion, hi addition to containing a heterologous sequence (e.g., Nurrl .
- rAAV vectors may also contain marker or reporter genes.
- Useful rAAV vectors have one or more of the AAV WT genes deleted in whole or in par, but retain functional flanking TR sequences.
- the AAV TRs may be of any serotype (e.g., derived from serotype 2, serotype 5, etc.) suitable for a particular application.
- the nucleic acids and vectors of the invention may be incorporated into a rAAV virion in order to facilitate introduction of the nucleic acid or vector into a cell.
- the capsid proteins of AAV compose the exterior, non-nucleic acid portion of the virion and are encoded by the AAV cap gene.
- the cap gene encodes three viral coat proteins, VPl, VP2 and VP3, which are required for virion assembly.
- the construction and use of rAAV virions has been described. See, e.g., U.S. Pat. Nos. 5,173,414, 5,139,941, 5,863,541, and 5,869,305, 6,057,152, 6,376,237; Rabinowitz et al., J. Virol.
- rAAV virions useful in the invention include those derived from a number of AAV serotypes including 1, 2, 3, 4, 5, 6, and 7.
- rAAV virions that have serotype 2 capsid proteins may be particularly useful as the experiments reported herein show they induce significantly higher cellular expression of ribozymes against Nurrl as well as higher cellular expression of nurrl than do rAAV virions having only serotype 2 capsids. Construction and use of AAV vectors and AAV proteins of different serotypes, including mosaic rAAV, are discussed in Chao et al., MoI. Ther.
- Pseudotyped vectors of the invention include AAV vectors of a given serotype (e.g., AA V2) pseudotyped with a capsid gene derived from a serotype other than the given serotype (e.g., AAVl, AAV3, AAV4, AA V5, AAV6 or AAV 7 capsids).
- AAV vectors of a given serotype e.g., AA V2
- a capsid gene derived from a serotype other than the given serotype e.g., AAVl, AAV3, AAV4, AA V5, AAV6 or AAV 7 capsids.
- An example of a pseudotyped vector that can be used in methods and compositions as described herein is an AA V2 vector encoding Nurrl or ribozymes against Nurrl pseudotyped with a capsid gene derived from AAV serotype 5.
- AA V2 vector encoding Nurrl or ribozymes against nurrl is pseudotyped with a capsid gene derived from AAV serotype 9 or serotype 10.
- the serotype of the vector and of the capsid protein(s) typically depends on the heterologous sequence to be delivered, as well as the type of cell or tissue into which the heterologous sequence is to be delivered. For example, when delivering a heterologous sequence to cells of the substantia nigra, AA V2 is preferred. As another example, when delivering a heterologous sequence to cells of the ventral tegmantal area, AAV5 is preferred.
- AAV virions that have mutations within the virion capsid may be used to infect particular cell types more effectively than non-mutated capsid virions.
- suitable AAV mutants may have ligand insertion mutations for the facilitation of targeting AAV to specific cell types.
- the construction and characterization of AAV capsid mutants including insertion mutants, alanine screening mutants, and epitope tag mutants is described in Wu et al., J. Virol. 74:8635-45, 2000.
- Other rAAV virions that can be used in methods of the invention include those capsid hybrids that are mosaic capsids, as well as those that are generated by molecular breeding of viruses as well as by exon shuffling.
- compositions described above may be administered to subjects including mammals (e.g., rodents, human beings) in any suitable formulation by any suitable method.
- mammals e.g., rodents, human beings
- rAAV virions i.e., particles
- target tissue e.g., substantia nigra
- IV intravenous
- IP intraperitoneal
- a conventional syringe and needle can be used to inject an rAAV virion suspension into a subject.
- injection can be in situ (i.e., to as particular tissue or location on a tissue), IM, IV, IP, or by another parenteral route.
- Parenteral administration of virions by injection can be performed, for example, by bolus injection of continuous infusion.
- Formulations for injection may be presented in unit dosage form, for example, in ampoules or in multi-dose containers, with an added preservative.
- the compositions may take such forms as suspensions, solutions or emulsions in oily or aqueous vehicles, and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents.
- the rAAV virions may be in powder form (e.g., lyophilized) for constitution with a suitable vehicle, for example, sterile pyrogen-free water, before use.
- the virions of the invention can be mixed with a carrier or excipient.
- Carriers and excipients that might be used include saline (especially sterilized, pyrogen-free saline) saline buffers (for example, citrate buffer, phosphate buffer, acetate buffer, and bicarbonate buffer), amino acids, urea, alcohols, ascorbic acid, phospholipids, proteins (for example, serum albumin), EDTA, sodium chloride, liposomes, mannitol, sorbitol, glycerol, and mannitol (which increases the spread of viral transduction).
- USP grade carriers and excipients are particularly preferred for delivery of virions to human subjects. Methods for making such formulations are well known and can be found in, for example, Remington's Pharmaceutical Sciences.
- the virions can also be formulated as a depot preparation. Such long acting formulations may be administered by implantation (for example subcutaneously or intramuscularly) or by IM injection.
- the virions may be formulated with suitable polymeric or hydrophobic materials (for example as an emulsion in an acceptable oil) or ion exchange resins, or as sparingly soluble derivatives.
- rAAV vectors may be administered to a subject using a variety of methods.
- rAAV vectors may be directly introduced into a subject by peritoneal administration (e.g., IP injection, oral administration), as well as parenteral administration (e.g., IV injection, IM injection, and in situ injection into target tissue).
- parenteral administration e.g., IV injection, IM injection, and in situ injection into target tissue.
- Methods and formulations for parenteral administration described above for rAAV virions may be used to administer rAAV vectors.
- Ex vivo delivery of cells transduced with rAAV vectors or virions is also provided _for within the invention.
- Ex vivo gene delivery may be used to transplant rAAV- transduced host cells back into the host.
- ex vivo stem cell e.g., mesenchymal stem cell
- a suitable ex vivo protocol may include several steps.
- a segment of target_tissue e.g., substantia nigra
- rAAV virions may be used to transduce a Nurrl -encoding nucleic acid into the host's cells.
- These genetically modified cells may then be transplanted back into the host.
- Several approaches may be used for the reintroduction of cells into the host, including intravenous injection, intraperitoneal injection, or in situ injection into target tissue.
- Microencapsulation of cells transduced or infected with rAAV modified ex vivo is another technique that may be used within the invention.
- Autologous and allogeneic cell transplantation may be used according to the invention. Effective Doses
- compositions described above are preferably administered to a subject (e.g., a mammal) in an effective amount, that is, and amount capable of producing a desirable result in a treated subject (e.g., decreasing or increasing Nurrl expression in a target tissue in the subject).
- a therapeutically effective amount can be determined as described below.
- Toxicity and therapeutic efficacy of the compositions utilized in methods of the invention can be determined by standard pharmaceutical procedures, using either cells in culture or experimental animals to determine the LD so (dose lethal to 50% of the population).
- the dose ration between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio LDso/EDso.
- Those compositions that exhibit large therapeutic indices are preferred. While those that exhibit toxic side effects may be used, care should be taken to design a delivery system that minimizes the potential damage of such side effects.
- the dosage of preferred compositions lies preferably within a range that includes an EDso with little or no toxicity. The dosage may vary within this range depending upon the dosage form employed and the route of administration utilized.
- dosage for any one subject depends on may factors, including the subject's size, body surface area, age, the particular composition to be administered, time and route of administration, general health, and other drugs being administered concurrently.
- Example 1 - Somatic knockdown of Nurrl an approach to evaluate its function in the dopaminergic system in the adult rat brain.
- Ribozymes against the mRNA of Nurrl were designed and delivered via rAAV, resulting in a somatic knock-out of Nurrl.
- rAAV ribozymes against the mRNA of Nurrl
- a decrease in Nurrl expression in the rats infected with rAAV expressing the ribozymes was detected.
- the ribozymes described herein can be used to investigate the physiological role of Nurrl in the mesencephalic dopaminergic system, for example.
- RNA is stored at -80 0 C.
- Example 2 Knock-down of Nurrl expression in the normal adult substantia nigra to determine its role in the maintenance of the mature midbrain dopagminergic phenotype.
- Example 3 Determination of the impact of Nurrl knock-down on the mature nigrostriatal dopagminergic system.
- a full-length rat Nurrl gene was cloned into a vector containing AA V5 TRs and this vector was encapsidated into virions with AAV5 capsid proteins. This virus was expressed in adult rat brain.
- a vector containing AA V2 TRs and Nurrl gene is constructed for encapsidation by capsid proteins of other AAV seroptypes (e.g., AAVl, AAV5, AA V9, AAVlO, or combinations thereof).
- AAV seroptypes e.g., AAVl, AAV5, AA V9, AAVlO, or combinations thereof.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Genetics & Genomics (AREA)
- Engineering & Computer Science (AREA)
- Organic Chemistry (AREA)
- Biomedical Technology (AREA)
- Molecular Biology (AREA)
- Zoology (AREA)
- General Engineering & Computer Science (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Physics & Mathematics (AREA)
- Biotechnology (AREA)
- Cell Biology (AREA)
- Immunology (AREA)
- High Energy & Nuclear Physics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Toxicology (AREA)
- Plant Pathology (AREA)
- Medicinal Chemistry (AREA)
- Microbiology (AREA)
- Gastroenterology & Hepatology (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
[0068] The invention relates to compositions and methods for modulating Nurrl expression in a cell, including a cell in a subject. Somatic knock-outs of Nurrl in the adult rat midbrain were performed using viral-delivered ribozymes, and over expression of Nurrl in the adult rat brain was achieved using a recombinant Adeno-Associated Virus (AAV) vector encoding Nurrl.
Description
MODULATING NURRl EXPRESSION IN A CELL
RELATED APPLICATIONS
[001] This application claims priority to U. S. Serial No. 60/912,002 filed April 16, 2007, which is incorporated herein in its entirety.
FIELD OF THE INVENTION
[002] The invention relates generally to the fields of molecular biology, gene therapy, and medicine. More particularly, the invention relates to methods and compositions for modulating Nurrl expression in a cell. BACKGROUND
[003] Nurrl is a transcription factor that belongs to the nuclear receptor superfamily. It has been demonstrated that Nurrl is essential for the development of ventral dopaminergic neurons. The role of Nurrl in adult animals is unknown, however, because Nurrl knock-out mice die after birth. Nurrl is suspected of playing a role in Parkinson's disease because a mutation in a familial form of Parkinson's disease was found in the Nurrl gene. Nurrl is also suspected of playing a role in learning and memory. Because to dopaminergic system is involved in drug addiction, it is possible that Nurrl plays a role in this disorder as well. A means for studying the role of Nurrl in the adult brain (e.g. maintenance of the adult dopaminergic phenotype), and in several neurological diseases, is thus highly desirable. SUMMARY
[004] The invention relates to compositions and methods for modulating Nurrl expression in a cell, including a cell in a subject. Somatic knock-outs of Nurrl in the adult rat midbrain were performed using viral-delivered ribozymes, and overexpression of Nurrl in the adult rat brain was achieved using a recombinant Adeno- Associated Virus (AAV) vector encoding Nurrl.
[005] Accordingly, the invention features an isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of : SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3.
[006] Also within the invention is a vector having at least one isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of : SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO: 3. The vector can be a recombinant AAV vector, e.g., a serotype 5 recombinant AAV vector.
[007] The invention also features a recombinant AAV vector including a nucleotide sequence encoding Nurrl .
[008] In another aspect, the invention relates to a method for modulating Nurrl expression in a eukaryotic cell. The method includes the steps of: a) providing at least one isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3; and b) contacting the cell with the at least one isolated nucleic acid under conditions in which the nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 can be expressed. Modulating Nurrl expression in a eukaryotic cell can include decreasing Nurrl expression in the eukaryotic cell.
[009] In still another aspect, the invention features a method of expressing Nurrl in the hippocampus of a subject. The method includes the steps of: a) providing the subject; and cO administering to the subject at least one isolated nucleic acid that encodes Nurrl protein. The at least one isolated nucleic acid can be within a vector, e.g., within a recombinant AAV vector. The recombinant AAV vector can be serotype 5, for example.
[0010] Another method within the invention includes a step of administering to a cell a recombinant AAV virion including: (a) a nucleotide sequence that when expressed modulates Nurrl expression in the cell, the nucleotide sequence interposed between a first AAV inverted terminal repeat and second AAV inverted terminal repeat: and (b) at least one AAV serotype 5 capsid protein. The nucleotide sequence can be one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3, and a nucleic acid that encodes Nurrl protein.
[0011] Unless otherwise defined, all technical terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs.
[0012] As used herein, a "nucleic acid," "nucleic acid molecule," "nucleod\tide sequence" or "polynucleotide" means a chain of two or more nucleotides such as RNA (ribonucleic acid) and DNA (deoxyribonucleic acid). A "purified" nucleic acid molecule is one that has been substantially separated or isolated away from other nucleic acid sequences in a cell or organism in which the nucleic acid naturally occurs (e.g., 30, 40, 50, 60, 70, 80, 90, 95, 96, 97, 98, 99, 100% free of contaminants). The term includes, e.g., a recombinant nucleic acid molecule incorporated into a vector, a plasmid, a virus, or a genome of a prokaryote or eukaryote.
[0013] By the term "Nurrl gene," or "Nurrl nucleic acid" is meant a native Nurrl - encoding nucleic acid sequence, genomic sequences from which Nurrl cDNA can be transcribed, and/or allelic variants and homologues of the foregoing. The terms encompass double-stranded DNA, single-stranded DNA, and RNA.
[0014] As used herein, "protein" or "polypeptide" are used synonymously to mean any peptide-linked chain of amino acids, regardless of length or post-translational modification, e.g., glycosylation or phosphorylation.
[0015] By the tern "Nurrl protein" is meant an expression product of a Nurrl gene: or a protein that shares at least 65% (but preferably 75, 80, 85, 90, 95, 96, 97, 98, or 99%) amino acid sequence identity with Nurrl (e.g., accession number NM 019328 in rat) and displays a functional activity of Nurrl . A "functional activity" of a protein is any activity associated with the physiological function of the protein.
[0016] As used herein, "Nurrl" can mean a native Nurrl -encoding nucleic acid sequence, genomic sequences from which Nurrl cDNA can be transcribed, and/or allelic variants and homologues of the foregoing, as well as an expression product of a Nurrl gene, or a protein that shares at least 65% (but preferably 75, 80, 85, 90, 95, 96, 97, 98, or 99%) amino acid sequence identity with Nurrl )e.g., accession number NM Ol 9328 in rat) and displays a functional activity of Nurrl.
[0017] When referring to a nucleic acid molecule or polypeptide, term "native" refers to a naturally-occurring (e.g., a wild-type; "WT") nucleic acid or polypeptide.
[0018] By the terms "hybridization" or "hybridizing" is meant the pairing of complementary RNA and DNA strands, or the pairing of complementary DNA single strands.
[0019] As used herein, "sequence identity" means the percentage of identical subunits at corresponding positions in two sequences when the two sequences are aligned to maximize subunit matching, i.e., taking into account gaps and insertions. Sequence identity can be measured using sequence analysis software (e.g., Sequence Analysis Software Package from Accelrys CGC, San Diego, CA).
[0020] As used herein, the term "vector" refers to a nucleic acid molecule capable of transporting another nucleic acid to which it has been linked. An example of a type of vector is an episome, i.e., a nucleic acid capable of extra-chromosomal replication. Another example of a vector is a viral vector (e.g., recombinant AAV vector). Vectors capable of directing the expression of genes to which they are operatively linked are referred to herein as "expression vectors."
[0021] A first nucleic acid sequence is "operably" linked with a second nucleic acid sequence when the first nucleic acid sequence is place in a functional relationship with the second nucleic acid sequence. For instance, a promoter is operable linked to a coding sequence if the promoter affects the transcription or expression of the coding sequence. Generally, operably linked nucleic acid sequences are contiguous and, where necessary to join two protein coding regions, in reading frame.
[0022] As used herein, the phrase "expression control sequence" refers to a nucleic acid that regulates the replication, transcription and translation of a coding sequence in a recipient cell. Examples of expression control sequences include promoter sequences, polyadenylation (pA) signals, introns, transcription termination sequences, enhancers, upstream regulatory domains, origins of replication, and internal ribosome entry sites ("IRES"). The term "promoter" is used herein to refer to a DNA regulatory sequence to which RNA polymerase binds, initiating transcription of a downstream (3' direction) coding sequence.
[0023] By the term "pseudotyped" is meant a nucleic acid or genome derived from a first AAV serotype that is encapsidated or packaged by an AAV capsid containing at least one AAV capsid protein of a second serotype. By "AAV inverted terminal repeats", "AAV terminal repeats, "ITRs" and "TRs" are meant those sequences required in cis for replication and packaging of the AAV virion including any fragments or derivatives of an ITR which retain activity of a full-length or WT ITR.
[0024] As used herein, the terms "rAAV vector" and "recombinant AAV vector" refer to a recombinant nucleic acid derived from an AAV serotype, including without limitation, AAVl, AAV2, AAV3, AAV4, AAV5, AAV6, AAV7, etc. rAAV vectors can have one or more of the AAV WT genes deleted in Whole or in part, preferably the rep and/or cap genes, but retain functional flanking ITR sequences. A "recombinant AAV virion" or "rAAV virion" is defined herein as an infectious, replication-defective virus composed of and AAV protein shell encapsulating a heterologous nucleotide sequence that is flanked on both sides by AAV ITRs.
[0025] Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present invention, suitable methods and materials are described below. All publications, patent applications, patents, and other references mentioned herein are incorporated by reference in their entirety. In the case of
conflict, the present specification, including definitions with control, hi addition, the particular embodiments discussed below are illustrative only and not intended to be limiting.
BRIEF DESCRIPTION OF THE DRAWINGS
[0026] FIG. 1 is a graph showing relative abundance of the Nurrl mRNA in four rats infected with the AAV5-Rzl844 virus. Rats were infected with the virus in the ipsilateral side, the contralateral side being the uninfected side.
[0027] FIG. 2 is a graph showing relative abundance of the Nurrl mRNA in eight rats infected with the AAV5-Rzl844 virus. Rats were infected with the virus in the ipsilateral side, the contralateral side being the uninfected side.
[0028] FIG. 3 shows AAVl, AAV2 and AAV5 transduction in the substantia nigra of adult rats. Panels A, D, G, show a montage of rostral to caudal coronal sections to illustrate the extent of expression of GFP in the substantia nigra with AAVl , AA V2 or AAV5. Panels, NeuroN (red) and GFP (green merged fluorescence to demonstrate the spread and number of cells transduced with each psuedotype. Panels C-O. high magnification of the transduced areas on B, G, L. J. The dotted line indicates that the number of cells and the mean estimated volume counted for AAVl were done for the entire midbrain, whereas for AA V2 and AAV5, cells in the substantia nigra were counted. Number of cells transduced with each virus pseudotype estimated from stereological counts. The dots individual animals. AAVl shows a higher transduction rate through the entire midbrain area (A, B, P, Q). AA V2 (number of cells and volume) is limited to the pars compacta of the substantia nigra (SN) (F, G, P, Q). AAV5 transduces the entire SN (K, L, P, Q). AAVl, AAV2 and AAV5 transduce TH+neurons (D, I, N) with similar efficiency (E, J, O). There are no significant differences in the number and volume of spread between AA V2 and AAV5 within the pars compacta (P,
Q)-
[0029] FIG. 4 is a photograph of an electrophoretic gel showing cleavage of Nurrl mRNA by ten ribozymes. Of these, three were found to cleave the in vitro transcribed full- length Nurl mRNA. The three ribozymes that cut are indicated on the top: Nurrl 1363 hh, Nurrl 1884 hh, andNurrl 1869hh. In the control lane the labeled IVR mRNA was incubated with no ribozymes under the same conditions. Arrow on the far left indicates the size of the full-length Nurrl mRNA. The arrows after the third lane show the size of the cleaved fragments. The larger cleaved fragments for 1844 and 1869 barely resolved in this gel (they
can be appreciated just underneath the full length band). Only the smaller fragments of the mRNA are marked by arrows (arrows 1844 and 1869).
[0030] FIG. 5 is a graph showing the effects of Nurrl ribozymes in the transcriptional activity of endogenous Nurrl. Transient transfection in PC 12 cells of the reporter 3x NBRE tk luciferase plus control expression vector (endo) or ribozyme (Rz) (1363, 1844 or 1869). The vectors were transfected at a 1 :2 molar ratio of reporter DNA to ribozyme, as indicated. Two of the ribozymes caused an increase in the transcriptional activation of the reporter gene. Only Rz 1844hh decreases Nurrl transactivation by almost half- fold.
DETAILED DESCRIPTION
[0031] The invention encompasses compositions and methods relating to the use of rAAV-based vectors encoding Nurrl and rAAV-based vectors encoding ribozymes against Nurrl for modulating Nurrl expression in a eukaryotic cell. The below described preferred embodiments illustrate adaptations of these compositions and methods. Nonetheless, from the description of these embodiments, other aspects of the invention can be made and/or practiced based on the description provided below. Biological Methods
[0032] Methods involving conventional molecular biology techniques are described herein. Such techniques are generally known in the art and are described in detail in methodology treatises such as Molecular Cloning: A Laboratory Manual, 3rd ed., vol. 1-3, ed. Sambrook et al., Cold Spring harbor Laboratory Press, Cold Spring Harbor, N. Y., 2001; and Current Protocols in Molecular Biology, ed. Ausubel et al., Greene Publishing and Wiley- Interscience, New York, 1992 (with periodic updates). Methods for chemical synthesis of nucleic acids are discussed, for example, in Beaucage and Carruthers, Tetra. Letts. 22:1859- 1862, 1981, and Matteucci et al., Am. Chem. Soc. 103:3185, 1981. Chemical synthesis of nucleic acids can be performed, for example, on commercial automated oligonucleotide synthesizers. Methods of designing, testing and using ribozymes are described in detail in methodology treatises such as Ribozymes and siRNA Protocols (Methods in Molecular Biology), 2nd ed., ed. Mouldy Sioud, Humana Press, Totowa, NJ, 2004; and Antisense and Ribozyme Methodology: Laboratory Companion, ed. Ian Gibson, Wiley- VCH Press, New York NY, 2003. Conventional methods of gene transfer and gene therapy can also be adapted for use in the present invention. See. e.g., Gene Therapy Protocols (Methods in Molecular
Medicine), ed. P.D. Robbins, Humana Press, 1997; and Retro-vectors for Human Gene Therapy, ed. CP. Hodgson, Springer Verlag, 1996. Compositions for Modulating Nurrl Expression In A Cell
[0033] The invention provides nucleic acids for modulating Nurrl expression in a cell. A typical nucleic acid as described herein for modulating Nurrl in a cell is an isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of : SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3. The sequences of SEQ ID :1, SEQ ID NO:2, and SEQ ID NO:3 are ribozyme sequences that decreased or prevented Nurrl expression in rodents as described in the Examples section below. Additional nucleic acids of the invention are variants of SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 that differ from SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 in one or more bases and that substantially maintain a functional activity (e.g., decreasing or preventing Nurrl expression in a cell) of SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3. Nucleic acids for modulating Nurrl expression in a cell can be delivered to a cell or a subject by any suitable route, but are typically delivered by a viral vector (e.g., recombinant AAV vector). Modulating Nurrl Expression In A Cell
[0034] Another aspect of the invention relates to methods for modulating Nurrl expression in a eukaryotic cell. A typical method includes the steps of: a) providing at least one isolated nucleic acid including a nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 and b) contacting the cell with the at least one isolated nucleic acid under conditions in which the nucleotide sequence having at least 80% sequence identity with one of: SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3 can be expressed. In such a method, modulating Nurrl expression involves decreasing or preventing Nurrl expression in the eukaryotic cell, hi the Examples described herein, ribozyme molecules designed to catalytically cleave Nurrl mRNA transcripts (e.g. SEQ ID NO:1, SEQ ID NO:2, and SEQ ID NO:3) were used to decrease translation of Nurrl mRNA and expression of Nurrl protein (see, e.g. Lewin AS, and Hauswirth WW, Trends MoI Med 7:221-228, 2001; and Lewin et al, Nat. Med 6:967-971, 1998). While ribozymes that cleave mRNA at site-specific recognition sequences can be used to destroy Nurrl mRNAs, the use of hammerhead ribozymes may be preferred some embodiments. Hammerhead ribozymes cleave mRNAs at locations dictated flanking regions that form complementary base pairs with the target mRNA. The construction and production of hammerhead ribozymes is well known in the art and are described more fully in Vazquez-
Tello et al., Nucleic Acid Res. 30(7): 1606-1612, 2002; Canny et al., J. Am Chem Soc. 126(35): 10848-10849, 2004; and Citti L, and Rainaldi G., Curr Gene Ther 5(1):11-24, 2005. Ribozymes as described herein can be delivered to a cell by any suitable means, such as a vector (e.g., recombinant AAV vector).
[0035] Methods of increasing Nurrl expression in a cell are also encompassed by the invention. Such methods typically include delivering to the cell a nucleic acid that encodes Nurrl protein. For example, a method of expressing Nurrl in the hippocampus of a subject includes the steps of providing the subject and administering to the subject at least one isolated nucleic acid that encodes Nurrl protein. rAAV Vectors And Virions
[0036] The nucleic acids of the invention may be incorporated into vectors and/or virions in order to facilitate their introduction into a cell. rAAV vectors useful in the invention are recombinant nucleic acid constructs that include (1) a heterologous sequence to be expressed (e.g., Nurrl, ribozymes against Nurrl, etc.) and (2) viral sequences that facilitate expression of the heterologous sequence. The viral sequences may include those sequences of AAV that are required in cis for replication and packaging (e.g., functional TRs) of the DNA into a virion, hi addition to containing a heterologous sequence (e.g., Nurrl . ribozymes against Nurrl, etc.), such rAAV vectors may also contain marker or reporter genes. Useful rAAV vectors have one or more of the AAV WT genes deleted in whole or in par, but retain functional flanking TR sequences. The AAV TRs may be of any serotype (e.g., derived from serotype 2, serotype 5, etc.) suitable for a particular application.
[0037] The nucleic acids and vectors of the invention may be incorporated into a rAAV virion in order to facilitate introduction of the nucleic acid or vector into a cell. The capsid proteins of AAV compose the exterior, non-nucleic acid portion of the virion and are encoded by the AAV cap gene. The cap gene encodes three viral coat proteins, VPl, VP2 and VP3, which are required for virion assembly. The construction and use of rAAV virions has been described. See, e.g., U.S. Pat. Nos. 5,173,414, 5,139,941, 5,863,541, and 5,869,305, 6,057,152, 6,376,237; Rabinowitz et al., J. Virol. 76:791-801, 2002; Bowles et al., J. Virol. 77:423-432, 2003, Kohlbrenner et al., MoI Ther. 12(6):1217-1225, 2005; Muzycka N. and Waπϊngton KH Jr., Human Gene Ther. 16(4):408-416, 2005; and Burger et al., MoI Ther. 10(2)302-317, 2004.
[0038] rAAV virions useful in the invention include those derived from a number of AAV serotypes including 1, 2, 3, 4, 5, 6, and 7. For targeting cells in the substantia nigra, for
example, rAAV virions that have serotype 2 capsid proteins may be particularly useful as the experiments reported herein show they induce significantly higher cellular expression of ribozymes against Nurrl as well as higher cellular expression of nurrl than do rAAV virions having only serotype 2 capsids. Construction and use of AAV vectors and AAV proteins of different serotypes, including mosaic rAAV, are discussed in Chao et al., MoI. Ther. 2:619- 623, 2000, Davidson et al., PNAS 97:3428-3432, 2000; Xiao et al., J. Virol. 72:2224-2232, 1998: Halbert et al., J. Virol. 74:1524-1532, 2000; Halbert et al., J. Virol. 75:6615-6624, 2001; and Auricchio et al., Hum. Molec. Genet. 10:3075-3081, 2001.
[0039] Also useful in the compositions and methods described herein are pseudotyped rAAV. Pseudotyped vectors of the invention include AAV vectors of a given serotype (e.g., AA V2) pseudotyped with a capsid gene derived from a serotype other than the given serotype (e.g., AAVl, AAV3, AAV4, AA V5, AAV6 or AAV 7 capsids). An example of a pseudotyped vector that can be used in methods and compositions as described herein is an AA V2 vector encoding Nurrl or ribozymes against Nurrl pseudotyped with a capsid gene derived from AAV serotype 5. As another example of a pseudotyped vector, and AA V2 vector encoding Nurrl or ribozymes against nurrl is pseudotyped with a capsid gene derived from AAV serotype 9 or serotype 10. In compositions and methods described herein, the serotype of the vector and of the capsid protein(s) typically depends on the heterologous sequence to be delivered, as well as the type of cell or tissue into which the heterologous sequence is to be delivered. For example, when delivering a heterologous sequence to cells of the substantia nigra, AA V2 is preferred. As another example, when delivering a heterologous sequence to cells of the ventral tegmantal area, AAV5 is preferred. Techniques involving the construction and use of pseudotyped rAAV virions are known in the art and are described Duan et al., J. Virol., 75:7662-7671, 2001 Halbert et al., J. Virol, 74:7524-1532, 2000; Zolotukhin et al., Methods, 28:158-167, 2002 and Auricchio et al., Hum. Molec. Genet, 10:3075-3081, 2001.
[0040] AAV virions that have mutations within the virion capsid may be used to infect particular cell types more effectively than non-mutated capsid virions. For example, suitable AAV mutants may have ligand insertion mutations for the facilitation of targeting AAV to specific cell types. The construction and characterization of AAV capsid mutants including insertion mutants, alanine screening mutants, and epitope tag mutants is described in Wu et al., J. Virol. 74:8635-45, 2000. Other rAAV virions that can be used in methods of the invention include those capsid hybrids that are mosaic capsids, as well as those that are
generated by molecular breeding of viruses as well as by exon shuffling. See Soong et al., Nat. Genet. 25:436-439, 2000; Kilman and Stemmer Nat. Biotechnol. 19:423-428, 2001; and Rabinowitz et al., J. Virol. 78(9):4421-4432, 2004. Administration of Compositions
[0041] The compositions described above may be administered to subjects including mammals (e.g., rodents, human beings) in any suitable formulation by any suitable method. For example, rAAV virions (i.e., particles) having a nucleotide sequence that, when expressed modulated Nurrl expression in a cell, may be directly introduced into a subject, including by intravenous (IV) injection, intraperitoneal (IP) injection, or in situ Injection into target tissue (e.g., substantia nigra). For example, a conventional syringe and needle can be used to inject an rAAV virion suspension into a subject. Depending on the desired route of administration, injection can be in situ (i.e., to as particular tissue or location on a tissue), IM, IV, IP, or by another parenteral route. Parenteral administration of virions by injection can be performed, for example, by bolus injection of continuous infusion. Formulations for injection may be presented in unit dosage form, for example, in ampoules or in multi-dose containers, with an added preservative. The compositions may take such forms as suspensions, solutions or emulsions in oily or aqueous vehicles, and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents. Alternatively, the rAAV virions may be in powder form (e.g., lyophilized) for constitution with a suitable vehicle, for example, sterile pyrogen-free water, before use.
[0042] To facilitate delivery of the rAAV virions to a subject, the virions of the invention can be mixed with a carrier or excipient. Carriers and excipients that might be used include saline (especially sterilized, pyrogen-free saline) saline buffers (for example, citrate buffer, phosphate buffer, acetate buffer, and bicarbonate buffer), amino acids, urea, alcohols, ascorbic acid, phospholipids, proteins ( for example, serum albumin), EDTA, sodium chloride, liposomes, mannitol, sorbitol, glycerol, and mannitol (which increases the spread of viral transduction). USP grade carriers and excipients are particularly preferred for delivery of virions to human subjects. Methods for making such formulations are well known and can be found in, for example, Remington's Pharmaceutical Sciences.
[0043] In addition to the formulations described preciously, the virions can also be formulated as a depot preparation. Such long acting formulations may be administered by implantation (for example subcutaneously or intramuscularly) or by IM injection. Thus, for example, the virions may be formulated with suitable polymeric or hydrophobic materials
(for example as an emulsion in an acceptable oil) or ion exchange resins, or as sparingly soluble derivatives.
[0044] Similarly, rAAV vectors may be administered to a subject using a variety of methods. rAAV vectors may be directly introduced into a subject by peritoneal administration (e.g., IP injection, oral administration), as well as parenteral administration (e.g., IV injection, IM injection, and in situ injection into target tissue). Methods and formulations for parenteral administration described above for rAAV virions may be used to administer rAAV vectors.
[0045] Ex vivo delivery of cells transduced with rAAV vectors or virions is also provided _for within the invention. Ex vivo gene delivery may be used to transplant rAAV- transduced host cells back into the host. Similarly, ex vivo stem cell (e.g., mesenchymal stem cell) therapy may be used to transplant rAAV vector-transduced host cells back into the host. A suitable ex vivo protocol may include several steps. A segment of target_tissue (e.g., substantia nigra) may be harvested from the host and rAAV virions may be used to transduce a Nurrl -encoding nucleic acid into the host's cells. These genetically modified cells may then be transplanted back into the host. Several approaches may be used for the reintroduction of cells into the host, including intravenous injection, intraperitoneal injection, or in situ injection into target tissue. Microencapsulation of cells transduced or infected with rAAV modified ex vivo is another technique that may be used within the invention. Autologous and allogeneic cell transplantation may be used according to the invention. Effective Doses
[0046] The compositions described above are preferably administered to a subject (e.g., a mammal) in an effective amount, that is, and amount capable of producing a desirable result in a treated subject (e.g., decreasing or increasing Nurrl expression in a target tissue in the subject). Such a therapeutically effective amount can be determined as described below.
[0047] Toxicity and therapeutic efficacy of the compositions utilized in methods of the invention can be determined by standard pharmaceutical procedures, using either cells in culture or experimental animals to determine the LD so (dose lethal to 50% of the population). The dose ration between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio LDso/EDso. Those compositions that exhibit large therapeutic indices are preferred. While those that exhibit toxic side effects may be used, care should be taken to design a delivery system that minimizes the potential damage of such side effects. The dosage of preferred compositions lies preferably within a range that includes an EDso with
little or no toxicity. The dosage may vary within this range depending upon the dosage form employed and the route of administration utilized.
[0048] As is well known in the medical and veterinary arts, dosage for any one subject depends on may factors, including the subject's size, body surface area, age, the particular composition to be administered, time and route of administration, general health, and other drugs being administered concurrently.
EXAMPLES
[0049] Example 1 - Somatic knockdown of Nurrl: an approach to evaluate its function in the dopaminergic system in the adult rat brain.
[0050] Ribozymes against the mRNA of Nurrl were designed and delivered via rAAV, resulting in a somatic knock-out of Nurrl. By semi-quantitative RT-PCR, a decrease in Nurrl expression in the rats infected with rAAV expressing the ribozymes was detected. The ribozymes described herein can be used to investigate the physiological role of Nurrl in the mesencephalic dopaminergic system, for example.
[0051] In the Year 1 and 2 experiments described below, Spraque-Dawley rats were injected unilaterally in SN with Nurrl 1844-hh ribozyme; the uninjected side served as a positive control for Nurrl normal expression. In addition, another set of control animals were injected with AAV5-GFP vector unilaterally to show that injection of AAV 5 alone doesn't have any effect on Nurrl expression. Recombinant AAV5-ribozymes were produced and the injections and titer were performed as described previously (Kirik et al., Proc Natl Aca Sci USA 99:4708-4713, 2002). The expression of Nurrl mRNA and protein was assessed by semi-quantitative RT-PCR and immunohistochemistry (IHC), respectively, essentially as previously described (Ojeda et al., J. Neurosci Res. 73:686-697, 2003). Long-term efficacy of the ribozyme are evaluated at 6 months and 1 year after the injection.
In vitro assays
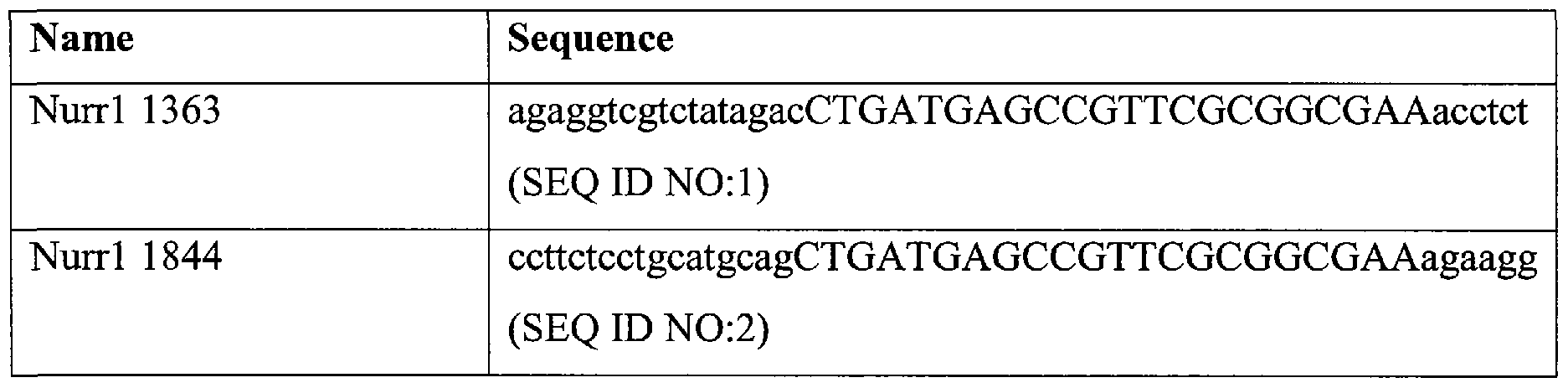
Table I: Designed ribozymes:
* Capital letters represent the sequences that constitute the structure of the ribozyme, and lower-case letters represent the sequences that will specifically bind Nurrl.
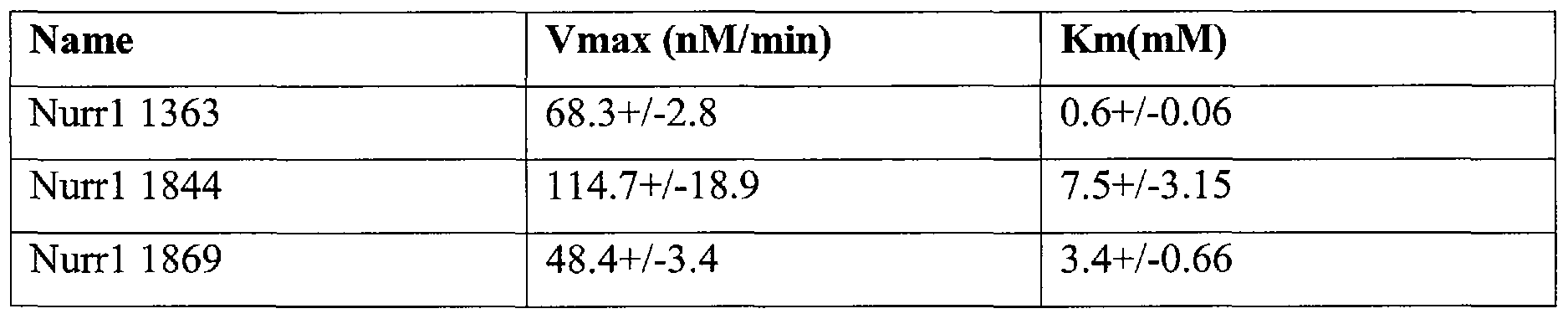
Table II: Kinetic parameters
In vivo assay
Designed AAV Vectors
AAV5-Rzl363
AAV5-Rzl844
AAV5-Rzl869
AAV5-Nurrl-GFP
AAV5-Nurrl
AAV5-ΔEndo
AAV5-UF11
[0052] For every experiment male Sprague Dawley rats were used. The AAVs were delivered directly on the Substantia nigra of the rats using a 10 ul Hamilton syringe mounted in a sterotaxis apparatus. After the injection, rats were maintained for 4 to 8 weeks in the Animal Service Unit of the Catholic University of Chile prior to the dissection of the Substantia nigra and total RNA extraction.
[0053] Summary of the experiments by year (Years 1 and 2 have been performed)
Year l :
Number of infected animals: 7
Weight: 180-20O g.
Coordinates of infection: AP: -0,53 L: -0,20 DV: -0,70
Time before dissection: 4 weeks
Number of analyzed animals: 7
Method of analysis: Semi-Quantitative RT-PCR (Aliaga, E., Rage, F., Bustos, G. & Tapia-
Arancibia, L. NeuroReport 9, 1959-1962 (1998)
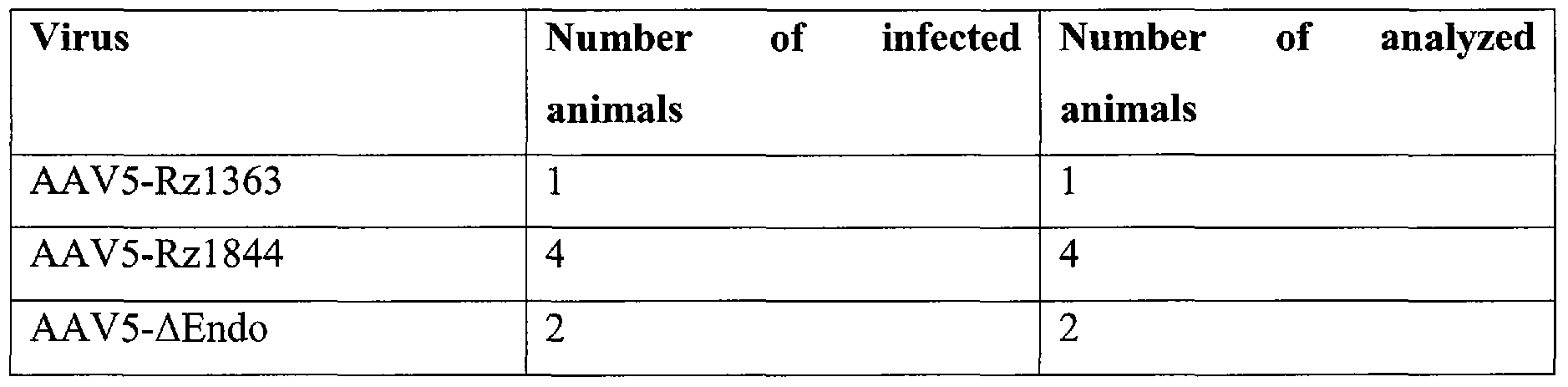
Table III
[0054] Referring to FIG. 1, of the 4 rats infected with the AAV5-Rzl844 virus, 2 of them showed a decrease (75%) in the relative abundance of the Nurrl mRNA when comparing with the uninfected side (contralateral side). Both rats infected with the AAV5- ΔEndo virus shown a decrease of the 35% in the relative abundance of the Nurrl mRNA. This decrease is smaller than the one produced by the AAV5-Rzl844 virus.
Year 2:
Number of infected animals: 28
Weight: 180-20Og.
Coordinates of infection: AP: -0,53 L: -0,20 DV: -0,70
Time before dissection: 4 weeks
Number of analyzed animals: 21
[0055] Method of analysis: Semi-Quantitative Real Time RT-PCR (Pfaffl, M. Nucleic Acids Research 29, 2002-2007 (2001))
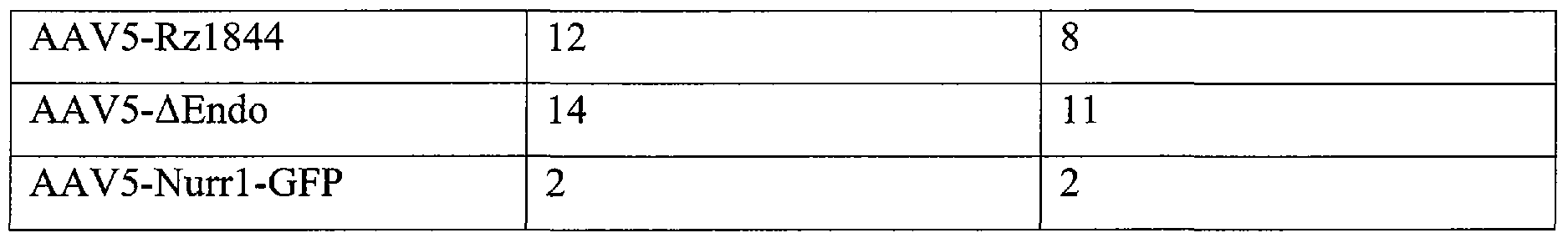
Table IV
[0056] Referring to FIG. 2, of the 8 rats infected with the AAV5-Rzl844 virus, 2 of them showed a decrease (54%) in the relative abundance of the Nurrl mRNA when comparing to the contralateral side. On average, rats infected with the AAV5-ΔEndo virus shown and 89% of abundance of the Nurrl mRNA.
Year 3:
(To be performed)
Number of infected animals: 16
Weight: 180-20O g.
Coordinates of infection: AP: -0,53 L: -0,20 DV: -0,70
Time before dissection: 8 weeks
Number of analyzed animals:
Table V
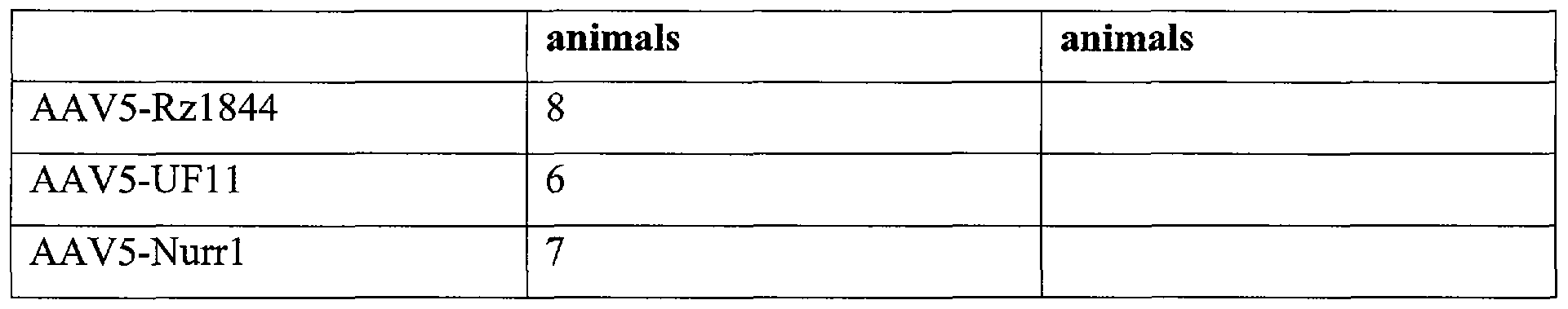
Virus Number of infected Number of analyzed animals animals
AAV5-Rzl844 8
AAV5-Nurrl 8
Year 4:
(To be performed)
Number of infected animals: 21
Weight: 180-20O g.
Coordinates of infection: AP: -0,53 L: -0,15 DV: -0,65
Time before dissection: 8 weeks
Number of analyzed animals:
Table VI
Following dissection and RNA extraction, total RNA is stored at -800C.
[0057] Example 2 - Knock-down of Nurrl expression in the normal adult substantia nigra to determine its role in the maintenance of the mature midbrain dopagminergic phenotype.
[0058] Several studies have shown that Nurrl regulates the expression of TH and the dopamine transporter, two proteins directly implicated in the maintenance of dopamine (DA) extracellular levels in the ST. The GDNF receptor cRet, important in axonal sprouting of DA neurons has also been shown to be regulated by Nurrl . More importantly, two mutations in the Nurrl gene found in familial cases of Parkinson's disease resulted in a marked decrease of Nurrl and TH expression. Furthermore, in studies of aged post mortem human brain, a parallel loss of TH and Nurrl immunoreactivity have been observed in melanin-containing neurons. In view of the above evidence, we hypothesize that Nurrl maintains functional DA extracellular levels by regulating the transcription of genes of the mature dopagminergic phenotype in the adult neurons.
[0059] The effects of abolishing Nurrl expression in a region-specific manner in the adult rat were investigated in order to learn the role of Nurrl in the maintenance of the mature mesencephalic dopagminergic phenotype. Recombinant AAV was used to deliver specific ribozymes against Nurrl directly in the SN of adult rats. Knowledge of the molecular signature of adult neurons in the substania nigra can potentially shed light on the selective vulnerability of these dopagminergic neurons to Parkinson's disease.
[0060] Because AA V5 (serotype 5) has been shown to work optimally in various regions of the brain including the SN (Fig. 3), this serotype was used in the experiments described in this example. Using AAV5, the levels of gene expression within the cell were much higher than with AA V2, enabling the delivery of more copies of a ribozyme per cell and more successfully reducing Nurrl mRNA levels.
[0061] Ten ribozymes targeted against various regions of the rat Nurrl mRNA were used. Three of these ribozymes were found to cut the full-length Nurrl mRNA in vitro: Nurrl-1363hh, Nurrl-1844hh and Nurrl-1869hh (Fig. 4). This was the first step in ribozyme selection, since full-length mRNA contains secondary structure that might hinder ribozyme activity. Once ribozymes that were accessible to the mRNA were selected, the kinetic properties of these ribozymes were analyzed. Studies relating to the experiments described herein have shown that ribozymes with fast kinetics work best in vivo. Ribozymes Nurrl - 1363, Nurrl-1844hh and Nurrl-1869hh cut the target RNA sequence in less than five minutes. The multiturnover kinetic analysis indicates that Nurrl-1363hh, Nurrl-1844hh and Nurrl -1869hh have a fast turnover rate (Table II). These three fast ribozymes were cloned in AAV5 to test their effects in cell lines (Fig 5).PC12 cells, which express endogenous Nurrl were transfected with a reporter plasmid containing three copies of Nurrl DNA binding sequence NBRE2, and a minimal promoter directing the expression of the luciferase gene, and each of the AA V5 Nurrl ribozyme vectors. Only Nurrl -1844hh decreased Nurrl -dependent transcription by -40% when compared to the basal transcriptional regulation of endogenous Nurrl on reporter gene expression alone (endo). Surprisingly the other two ribozymes had a rebound effect on Nurrl regulation and caused an increase in transcription of the reporter gene.
[0062] Example 3 - Determination of the impact of Nurrl knock-down on the mature nigrostriatal dopagminergic system.
[0063] Integrity and functionality of the nigro-striatal dopagminergic system are evaluated in rats with decreased/null Nurrl expression as follows:
Behavioral analysis. Animals are injected unilaterally with Nurrl -1844hh so that asymmetries in motor behavior can be identified. Asymmetries in the control of voluntary movements are estimated by the rotation test after apomorphine-HCl or D-amphetamine sulfate injection and by the cylinder test as previously described (Kirik et al., Proc Natl Acad Sci USA 99:4708-4713, 2002).
[0064] Neurochemical analysis. In vivo microdialysis and DA quantification are performed essentially as described by Andres et al., Brain Res 797:255-266, 1998.
[0065] Histology. It has been reported that Nurrl -null mice do not express TH, DAT, Ret and VMAT2 in ventral dopagminergic neuron precursors. Other genes specific of this phenotype, e.g., Ptx3, are expressed initially during development and then they are lost
together with the loss of the dopagminergic neurons in the mutant mice. The expression of TH, DAT, Ptx3, Ret and VMAT2 is examined by IHC coupled to densitometry analysis in SN and striatum of control and AAV5 ribozyme-injected rats.
Example 4 - Overexpression of Nurrl
[0066] A full-length rat Nurrl gene was cloned into a vector containing AA V5 TRs and this vector was encapsidated into virions with AAV5 capsid proteins. This virus was expressed in adult rat brain. A vector containing AA V2 TRs and Nurrl gene is constructed for encapsidation by capsid proteins of other AAV seroptypes (e.g., AAVl, AAV5, AA V9, AAVlO, or combinations thereof). Other Embodiments
[0067] It is to be understood that while the invention has been described in conjunction with the detailed description thereof, the foregoing description is intended to illustrate and not limit the scope of the invention, which is defined by the scope of the appended claims. Other aspects, advantages, and modifications are within the scope of the following claims.
Claims
1. An isolated nucleic acid comprising a nucleotide sequence having at least 80% sequence identity with a sequence selected from the group consisting of: SEQ ID NO: 1 , SEQ ID NO:2 and SEQ ID NO:3.
2. A vector comprising at least one isolated nucleic acid comprising a nucleotide sequence having at least 80% sequence identity with a sequence selected from the group consisting of SEQ ID NO:1, SEQ ID NO:2 and SEQ ID NO:3.
3. The vector of claim 2, wherein the vector is a recombinant AAV vector.
4. The vector of claim 3, wherein the recombinant AAV vector is serotype 5.
5. A recombinant AAV vector comprising a nucleotide sequence encoding Nurrl .
6. A method for modulating Nurrl expression in a eukaryotic cell, the method comprising the steps of: a) providing at least one isolated nucleic acid comprising a nucleotide sequence having at least 80% sequence identity with a sequence selected from the group consisting of: SEQ ID NO:1, SEQ ID NO:2 and SEQ ID NO:3 and b) contacting the cell with the at least one isolated nucleic acid under conditions in which the nucleotide sequence having at least 80% sequence identity with a sequence selected from the group consisting of: : SEQ ID NO:1, SEQ ID NO:2 and SEQ ID NO:3 can be expressed.
7. The method of claim 6, wherein modulating Nurrl expression in a eukaryotic cell comprises decreasing Nurrl expression in the eukaryotic cell.
8. A method of expressing Nurrl in the hippocampus of a subject, the method comprising the steps of: a) providing the subject; and b) administering to the subject at least one isolated nucleic acid that encodes Nurrl protein.
9. The method of claim 8, wherein the at least one isolated nucleic acid is comprised within a vector.
10. The method of claim 9, wherein the at least one isolated nucleic acid is comprised within a recombinant AAV vector.
11. The method of claim 10, wherein the recombinant AAV vector is serotype 5.
12. A method comprising a step of administering to a cell a recombinant AAV virion comprising: (a) a nucleotide sequence that when expressed modulates Nurrl expression in the cell, the nucleotide sequence interposed between a first AAV inverted terminal repeat and second AAV inverted terminal repeat; and (b) at least one AAV serotype 5 capsid protein.
13. The method of claim 12, wherein the nucleotide sequence is selected from the group consisting of: SEQ ID NO:1, SEQ ID NO:2 and SEQ ID NO:3, and a nucleic acid that encodes Nurrl protein.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US91200207P | 2007-04-16 | 2007-04-16 | |
| US60/912,002 | 2007-04-16 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2008130949A2 true WO2008130949A2 (en) | 2008-10-30 |
| WO2008130949A3 WO2008130949A3 (en) | 2009-04-16 |
Family
ID=39876148
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2008/060392 Ceased WO2008130949A2 (en) | 2007-04-16 | 2008-04-16 | Modulating nurr1 expression in a cell |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2008130949A2 (en) |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1183389A2 (en) * | 1999-04-30 | 2002-03-06 | University of Florida | Adeno-associated virus-delivered ribozyme compositions and methods of use |
| JP2004514407A (en) * | 2000-04-28 | 2004-05-20 | ザ・トラステイーズ・オブ・ザ・ユニバーシテイ・オブ・ペンシルベニア | AAV5 capsid pseudotyped in heterologous capsid and recombinant AAV vector comprising AAV5 vector |
| US20020049151A1 (en) * | 2000-05-12 | 2002-04-25 | Evelyn Murphy | Therapeutic approaches to diseases by suppression of the NURR subfamily of nuclear transcription factors |
-
2008
- 2008-04-16 WO PCT/US2008/060392 patent/WO2008130949A2/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| WO2008130949A3 (en) | 2009-04-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7504967B2 (en) | MeCP2 expression cassette | |
| Klein et al. | Dose and promoter effects of adeno-associated viral vector for green fluorescent protein expression in the rat brain | |
| US7238674B2 (en) | Methods for delivering DNA to muscle cells using recombinant adeno-associated virus vectors | |
| KR102629418B1 (en) | Optimized CLN1 gene and expression cassette and uses thereof | |
| US20230270884A1 (en) | Compositions useful for treatment of charcot-marie-tooth disease | |
| JP2025120285A (en) | Gene therapy for ciliopathy | |
| CN110325219A (en) | Intrathecal delivery of recombinant adeno-associated viruses encoding methyl-CpG binding protein 2 | |
| KR20250102136A (en) | Nucleic acid regulatory elements and methods of use for gene expression in the central nervous system | |
| WO2021207415A1 (en) | CpG-FREE ITRs FOR AAV GENE THERAPY | |
| CN116390773A (en) | Viral particles for the treatment of tauopathies such as Alzheimer's disease by gene therapy | |
| US20240376493A1 (en) | Optimized ap4m1 polyneucleotides and expression cassettes and their use | |
| WO2024079661A1 (en) | Atp7b gene therapy | |
| US20240401078A1 (en) | Compositions useful for treatment of charcot-marie-tooth disease | |
| WO2008130949A2 (en) | Modulating nurr1 expression in a cell | |
| EP4185700A1 (en) | Optimized slc13a5 genes and expression cassettes and their use | |
| AU2021252515A1 (en) | Adeno-associated virus compositions for IDS gene transfer and methods of use thereof | |
| WO2020223215A1 (en) | Optimized sumf1 genes and expression cassettes and their use | |
| WO2025076463A1 (en) | Gene therapy for lafora disease | |
| WO2024257061A1 (en) | A hybrid dual aav vector system with splice enhancer elements for expression of large genes | |
| TW202434729A (en) | Synthetic nucleic acids including astrocyte-directed promoter constructs and methods of using the same | |
| CN116670159A (en) | Compositions and their use for the treatment of Angelman syndrome | |
| BR102020023425A2 (en) | Biological vector for the treatment of neurodegenerative diseases based on gene therapy | |
| JP2022506066A (en) | Optimized FIG4 genes and expression cassettes and their use | |
| CA2978917A1 (en) | Recombinant glut1 adeno-associated viral vector constructs and related methods for restoring glut1 expression | |
| BR112017019294B1 (en) | RECOMBINANT ADENO-ASSOCIATED VECTOR (RAAV), COMPOSITION COMPRISING THE SAME, AND KIT |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 08745900 Country of ref document: EP Kind code of ref document: A2 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 08745900 Country of ref document: EP Kind code of ref document: A2 |