WO2008018177A1 - Process for production of photosemiconductor particles - Google Patents
Process for production of photosemiconductor particles Download PDFInfo
- Publication number
- WO2008018177A1 WO2008018177A1 PCT/JP2007/000853 JP2007000853W WO2008018177A1 WO 2008018177 A1 WO2008018177 A1 WO 2008018177A1 JP 2007000853 W JP2007000853 W JP 2007000853W WO 2008018177 A1 WO2008018177 A1 WO 2008018177A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- optical semiconductor
- ceramic
- producing
- substrate
- water
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J35/00—Catalysts, in general, characterised by their form or physical properties
- B01J35/30—Catalysts, in general, characterised by their form or physical properties characterised by their physical properties
- B01J35/39—Photocatalytic properties
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J21/00—Catalysts comprising the elements, oxides, or hydroxides of magnesium, boron, aluminium, carbon, silicon, titanium, zirconium, or hafnium
- B01J21/06—Silicon, titanium, zirconium or hafnium; Oxides or hydroxides thereof
- B01J21/063—Titanium; Oxides or hydroxides thereof
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J37/00—Processes, in general, for preparing catalysts; Processes, in general, for activation of catalysts
- B01J37/02—Impregnation, coating or precipitation
- B01J37/03—Precipitation; Co-precipitation
- B01J37/031—Precipitation
Definitions
- the present invention relates to a base that is a metal compound optical semiconductor, and a method for producing optical semiconductor particles comprising ceramics immobilized on at least a part of the surface of the base.
- Metal compound optical semiconductors such as titania and zinc oxide exhibit the property of absorbing light having energy corresponding to the bandwidth. For this reason, application development is being attempted as an ultraviolet shielding agent in the fields of cosmetics and resin materials. Focusing on the high reactivity of holes and electrons generated in metal compound photo-semiconductors that absorb light, the photocatalyst is applied to environmental purification such as water purification, antifouling, antibacterial, deodorization, and air purification. Attempts have also been made.
- the metal compound optical semiconductor may be used as it is, but improvement may be achieved by immobilizing different compounds on the surface.
- metal oxide particles whose surface is coated with dense silica see Patent Document 1
- pigments whose surface is coated with a silicon compound and an aluminum compound see Patent Document 2
- silica hydrate coating layer Titanium oxide photocatalyst for basic gas removal see Patent Document 3
- high-activity photocatalyst coated with a silicon oxide film substantially free of pores see Patent Document 4
- partially coated with calcium phosphate Photocatalysts see Patent Document 5).
- a method of immobilizing different compounds on the surface of a metal compound optical semiconductor includes a gas phase method in which a gaseous raw material is reacted with a metal compound optical semiconductor (see Patent Document 6), or treatment in a solvent.
- a liquid phase method There is a liquid phase method.
- the liquid phase method is excellent in that heat removal can be easily performed and the amount of raw materials to be reacted can be easily controlled.
- liquid phase methods include a method in which silicate and sulfuric acid are dropped while heating in a basic aqueous solution (see Patent Document 7), water is adsorbed in advance on the surface of an optical semiconductor in an organic solvent, and a metal alkoxy A method of hydrolyzing a metal on the surface of an optical semiconductor (see Patent Document 8), a production method of coating with silicate in an acidic aqueous medium having a pH of 5 or less (see Patent Document 4), Na, , Cl, Ca, P, Mg, etc. (see Patent Document 5).
- Examples of simplifying solid-liquid separation using a flocculant in a method for producing a metal compound optical semiconductor include polybasic mineral acids, organic acid or phenols having two or more hydroxyl groups and / or hydroxyl groups, and those A method of treating with at least one of the above salts and at least one organic polymer flocculant (see Patent Document 9), and metatitanic acid generated by hydrolysis of titanium sulfate is aggregated with the polymer flocculant A method (see Patent Document 10) is disclosed.
- Patent Documents 9 and 10 in a method for producing optical semiconductor particles comprising a base that is a metal compound optical semiconductor and ceramics immobilized on at least a part of the surface of the base, There is no mention of the use of chemicals.
- Patent Document 1 Japanese Patent No. 3570730
- Patent Document 2 Japanese Patent No. 539263
- Patent Document 3 Japanese Patent Application Laid-Open No. 2002_159865
- Patent Document 4 Japanese Patent Application No. P CT / J P 2006/302542
- Patent Document 5 Japanese Patent No. 3275032
- Patent Document 6 Japanese Patent No. 1 895322
- Patent Document 7 Japanese Patent Laid-Open No. 02-296726
- Patent Document 8 Japanese Patent No. 2832342
- Patent Document 9 Japanese Patent Laid-Open No. 53-39296
- Patent Document 10 Japanese Patent No. 3554803
- the present invention provides a production method in which solid-liquid separation can be easily performed when producing optical semiconductor particles by a liquid phase method, and the operation of the obtained optical semiconductor particles as an optical semiconductor is not affected.
- the challenge is to provide a method.
- the present inventors diligently studied using a slurry containing a substrate having a ceramic surface fixed thereto.
- the flocculant is added to solid-liquid separate the particles containing the substrate in the agglomerated state, and the resulting solid is heated to decompose the flocculant and from the substrate having ceramics immobilized on the surface.
- the optical semiconductor particles can be easily produced.
- the inventors have found that the obtained optical semiconductor particles exhibit the same performance as the optical semiconductor particles produced without using a flocculant, and have completed the invention. That is, according to the present invention, the following method for producing optical semiconductor particles is provided.
- a base that is a metal compound optical semiconductor, and at least one of the surfaces of the base A method for producing optical semiconductor particles comprising ceramics fixed to a part, wherein the ceramics is a compound different from the substrate, and comprising the following steps (A) to (D) :
- the step (E) is based on the flow of the water-containing solvent, and the flow rate of the liquid in the portion separating the solid and the washing waste liquid is determined by the horizontal vector component and the vertical direction.
- washing is performed by controlling the supply amount of the hydrous solvent so that the rising rate of the liquid as a vector component in the vertical direction is slower than the settling rate of the solids.
- the polymer flocculant is a polymer compound containing one or more partial structures selected from the group consisting of acrylamide or a modified product thereof as a repeating unit.
- the step (A) force is selected from the group consisting of a hydrous solvent containing the substrate and a silicate, a hydrous solvent containing the silicate and the substrate, and a hydrous solvent containing the substrate and a hydrous solvent containing the silicate.
- the silicon oxide is fixed in a film form on at least a part of the surface of the substrate, and in pore size distribution measurement in a region of 20 to 500 angstroms by a nitrogen adsorption method,
- the amount of silicon supported per 1 m 2 of the surface area of the optical semiconductor particles is 0.1 mg or more and 2. Omg or less, according to any one of [9] to [1 1] Method for producing optical semiconductor particles.
- the step (A) is a step of mixing a hydrous solvent containing at least one or more water-soluble salts of magnesium and aluminum with a hydrous solvent containing the substrate and the alkali metal salt.
- a semiconductor substrate which is a metal compound optical semiconductor, and optical semiconductor particles comprising ceramics fixed to at least a part of the surface of the substrate, by a liquid phase method
- liquid phase method it is possible to provide a production method in which liquid separation can be easily performed and the obtained optical semiconductor particles do not affect the action as an optical semiconductor.
- FIG. 1 is a diagram showing an I og differential pore volume distribution curve (solid line) of optical semiconductor particle 1 and an I og differential pore volume distribution curve (dotted line) of titanium dioxide ST_01 corresponding to the substrate.
- FIG. 3 A diagram showing the I og differential pore volume distribution curve (solid line) of photo-semiconductor particle 6 and the I og differential pore volume distribution curve (dotted line) of titanium dioxide P _ 2 5 corresponding to the substrate. is there.
- the optical semiconductor particles produced in the present invention are composed of a base that is a metal compound optical semiconductor and ceramics immobilized on at least a part of the surface of the base.
- the photosemiconductor particles may be independently configured as described above, and all the particles may have the same configuration, or may be a mixture of two or more differently configured particles. Good. Further, two or more kinds of ceramics may be fixed to one substrate.
- the term “consisting of” does not exclude the case where other components are included, and includes both the case where only the mentioned components are included and the case where other components are also included. Shall mean.
- the substrate is not particularly limited as long as it is a metal compound optical semiconductor. Examples include titanium oxide, zinc oxide, tungsten oxide, and strontium titanate.
- the substrate preferably has a crystallite diameter derived from X-ray diffraction (X R D) in the range of 1 to 500 nm.
- X R D X-ray diffraction
- the shape is not particularly limited, and examples thereof include a spherical shape, a spheroid shape, a column shape, a geometric shape, and a flake shape.
- a substrate having ceramics immobilized on at least a part of its surface can be used as the substrate of the present invention. In many cases, only one type of substrate is used, but a mixture of two or more types may be used.
- titanium oxide is preferable because it is excellent in photocatalytic activity and is excellent in safety and stability.
- examples of titanium oxide include amorphous, anatase type, rutile type, and wurtzite type titanium dioxide, and among these, anatase type, rutile type, or a mixture thereof, which is superior in photocatalytic activity, is more suitable. preferable. These may contain a small amount of amorphous material.
- one or more transition metals are added to the metal compound optical semiconductor. Addition or support, Metal compound semiconductor with one or more typical elements of Group 14, 15 and / or Group 16 added or supported, Optical semiconductor composed of two or more metal compounds A mixture of two or more kinds of metal compound semiconductors may be used.
- the ceramic is not particularly limited as long as it is a compound different from the base constituting the optical semiconductor particles. Therefore, it may be a metal compound optical semiconductor.
- the ceramic when the substrate is titanium oxide, the ceramic may be zinc oxide.
- the ceramic is preferably an oxide of Si, AI, Zr, or Ce, or a calcium compound.
- the oxide may include a hydroxyl group, adsorbed water, etc., and may be a single oxide, a binary complex oxide, or a multicomponent complex oxide.
- Other metal elements may be included as an ion or a metal.
- the ceramic constituting the optical semiconductor particles provided by the production method of the present invention is a fired ceramic.
- Calcium compounds include calcium phosphates or silicates, such as hydroxyapatite, fluorideapatites, and hydroxyapatites containing other metal ions. And calcium phosphate salt having a mineral composition known as Calcium silicate.
- an oxide of Si is more preferable.
- the Si oxide is immobilized on the surface of the substrate, the heat resistance of the substrate is improved. This allows the flocculant to be decomposed at higher temperatures.
- the ceramic raw material forms a ceramic or a precursor thereof on the surface of the substrate by causing hydrolysis, condensation, precipitation or the like when mixed with the substrate and the water-containing solvent in the step (A). It is a substance. Ceramic materials may be used alone or in combination of two or more. For example, metal halides, nitrates or sulfates, metal alkoxides, metal complexes, metal oxoacid salts And the like can be mentioned as raw materials used alone. Examples of the metal oxo acid salt include sodium silicate and potassium silicate as Si oxo acid salt. Examples of the raw material used in two or more types include a combination of a metal-soluble salt and a soluble oxoacid or a salt thereof.
- an insoluble or hardly soluble oxoacid salt of the metal is precipitated on the surface of the substrate to become a ceramic or a precursor thereof.
- a phosphate compound is precipitated.
- the ceramic is silicon oxide
- examples of the raw material include silicic acid, silicate, silicate ester, silicon halide, organohydridopolysiloxane.
- a precipitating agent or a catalyst may be used for forming ceramics or a precursor thereof.
- a ceramic precursor is a compound that changes into a ceramic by a physical or chemical treatment.
- the ceramic precursor may be converted into ceramics, or a process of converting into ceramics may be provided separately.
- those converted into ceramics include those that become ceramics by oxidation, hydrolysis, condensation reaction, etc. in the heat treatment in step (D).
- An example of such a precursor is aluminum hydroxide.
- Aluminum hydroxide is a compound that is formed on the surface of a substrate using aluminum alkoxide as a raw material and can be converted to alumina in the step (D).
- the water-containing solvent is water or a mixed solvent mainly containing water.
- the water-containing solvent may include a water-soluble organic solvent having 1 to 4 carbon atoms among organic solvents classified into alcohols, ketones, ethers, or esters.
- Specific examples of the water-containing solvent include water, water and methyl alcohol, water and ethyl alcohol, water and isopropanol, and the like. Of these, water is preferred.
- These water or mixed solvents may be used alone or in combination of two or more.
- a substrate, ceramics or a raw material thereof, and water-containing there is no restriction
- a method of mixing a ceramic or its raw material after mixing the substrate with a hydrous solvent (b) a mixed solution of mixing the hydrous solvent and the substrate, and a mixed solution of mixing the hydrous solvent and ceramics or its raw material
- E When the ceramic is formed by reacting two or more kinds of raw materials, the mixture containing the hydrous solvent, the substrate, and the first raw material of the ceramic is mixed.
- the mixing is not particularly limited as long as the water-containing solvent can exist as a liquid, and may be performed at room temperature or appropriately under cooling, heating, and pressurizing conditions. Further, the mixed solution thus prepared may be appropriately aged.
- the mixed solution prepared in the step (A) includes a substrate having ceramics or a precursor thereof immobilized on at least a part of the surface and water.
- the mixed liquid does not necessarily need to be composed only of these.
- the concentration of the substrate relative to the total weight of the mixed solution prepared in step (A) (hereinafter referred to as slurry concentration as appropriate) is preferably 0.5 wt% or more and 40 wt% or less. More preferably, it is more than 20% by weight.
- slurry concentration is equal to or higher than the lower limit, productivity is increased, and when the slurry concentration is lower than the upper limit, the specific gravity of the liquid can be prevented from becoming excessive, and economic efficiency can be ensured.
- the force average particle size is 1
- the average particle diameter is preferably 0 m or less, and more preferably 3 m or less.
- the solid matter is easily agglomerated by agglomerating the fine particles.
- the feature of the present invention is that it can be collected in the stool.
- the concentration of water in the mixed solution prepared in the step (A) is preferably 60% by weight or more, more preferably 80% by weight or more, when expressed as the weight of water with respect to the total weight of the mixed solution.
- the flocculant is easy to dissolve, and the agglomeration effect is manifested.
- the water contained in the mixed solution may be finally in the above range. Therefore, a preparation method that uses only water as the water-containing solvent and falls within the above range upon completion of mixing may be used.
- water may be added for adjustment.
- the flocculant may not be effective due to the salting-out action, so even if the water concentration is within this range, the water content can be increased by decanting. Substitution of the solvent may be performed.
- step (A) for example, a hydrous solvent and a silicate containing a substrate that is a metal compound optical semiconductor, a hydrous solvent containing a silicate and the substrate, and a hydrous solvent containing the substrate and a hydrous solvent containing a silicate, Mix at least one set. Then, the pH of the mixed solution containing both the substrate and silicate is maintained at 5 or less and mixed.
- the ceramic contains silicon oxide.
- the pH of the hydrous solvent is constantly measured, and adjusted by adding an acid and a base as appropriate. It doesn't matter how. However, it is easy to neutralize the total amount of the base components contained in the silicate used for the production, and to make a sufficient amount of acid present in the water-containing solvent beforehand so that the pH is 5 or less.
- the silicate a salt of silicic acid and / or its oligomer may be used, and two or more kinds may be mixed and used.
- Sodium and potassium salts are preferred because they are easily available industrially, and the dissolution step can be omitted, so that a sodium silicate aqueous solution (JISK 1408 "water glass") is further used.
- a sodium silicate aqueous solution JISK 1408 "water glass"
- Any acid can be used, but a mineral acid such as hydrochloric acid, nitric acid, or sulfuric acid is preferably used.
- the acids may be used alone or in combination of two or more.
- the base does not need to be used separately when using the above-described method in which a sufficient amount of acid to have a pH of 5 or less is previously present in the aqueous solvent. However, when using a base, any base can be used. Of these, metal hydroxides such as hydroxy hydroxide and sodium hydroxide are preferably used.
- step (A) for example, a compound containing calcium and phosphorus is formed on the surface of the substrate using a simulated body fluid.
- the specific mixing procedure is as follows. First, a simulated body fluid is prepared, and then the substrate is mixed. In this example, an apatite, which is a kind of calcium phosphate, is formed as ceramics.
- the simulated body fluid refers to sodium chloride, potassium chloride, sodium bicarbonate, dipotassium hydrogen phosphate, magnesium chloride, calcium chloride, sodium sulfate, or sodium fluoride dissolved in water.
- an acid base such as namine hydrochloride.
- N a CI, N a H C0 3, N a 2 H P0 4, N a H 2 P0 4, KC I, KH C0 3, K 2 H P0 4, KH 2 P0 4, M g CI 2, C a CI 2 , N a 2 S0 4, that the N a F any compound selected from the like are dissolved in water.
- a compound containing calcium and a compound containing phosphorus are added. If necessary, HCI, (CH 2 OH) 3 CN H 2 or the like can be added.
- the composition of the simulated body fluid is as follows: N a +: 1 20 to 160 mM, K +: 1 to 20 mM, C a 2+ : 0.5 to 50 mM, H C0 3 _: 0.5 to 30 mM, HP 0 4 2 _: “! ⁇ 20 mM is preferred.
- Aqueous solution of 4 Can be exemplified as one of the preferred simulated body fluid compositions.
- a water-containing solvent containing one or more water-soluble salts of magnesium and aluminum is mixed with a water-containing medium containing the base and an alkali metal salt.
- a water-containing medium containing the base and an alkali metal salt for example, magnesium and aluminum nitrates are used as water-soluble salts, and sodium carbonate is used as an alkali metal salt.
- sodium carbonate becomes a precipitant, and magnesium and aluminum hydroxides are formed on the surface of the substrate.
- Step (B) is a step of adding an aggregating agent to the mixed liquid obtained in step (A) and stirring.
- the substrate on which the ceramic or its precursor is immobilized on at least a part of the surface is agglomerated by the aggregating agent.
- a solid body containing the substrate and the flocculant having ceramics or a precursor thereof immobilized on at least a part of the surface is formed.
- the flocculant added in the step (B) is preferably a water-soluble flocculant.
- water-soluble flocculants polymer flocculants are preferred. Examples thereof include polymers such as acrylamide, acrylic acid, and methacrylic acid, and those obtained by modifying at least a part of functional groups of these polymers.
- any type of anionic, nonionic, cationic, or amphoteric can be used, and any type of polymer flocculant can be selected depending on the pH of the added mixture.
- an inorganic salt may remain when the flocculant is decomposed in step (D), and further washing may be required.
- ion pairs include sodium salts, potassium salts, chlorides, sulfates, and the like. From this point, a nonionic polymer flocculant is preferable. However, as is apparent from the amount of the flocculant used for the optical semiconductor particles, the amount of the remaining inorganic salt is not large, so that types other than the nonionic type can be used.
- polyacrylamide containing one or more compounds selected from the group consisting of acrylamide or a modified product thereof as a repeating unit.
- a system flocculant can be illustrated. Specifically, polyacrylamide, which is a polymer of acrylic amide, modified acrylic amide polymer, copolymer of acrylic acid and acrylic amide, copolymer of acrylic amide and modified acrylic amide, acrylic amide And a copolymer of dimethylaminoethyl methacrylate.
- the anionicity or cationicity of the flocculant is adjusted by post-processing the polymer or by the copolymerization ratio.
- the modified acrylic amide is the substitution of the hydrogen atom of the amide group of the acrylamide with an alkyl group or an alkyl group having an ammonium salt / sulfonic acid group / hydroxyl group and a hydrophilic substituent. Examples are given.
- the form of the flocculant is not particularly limited, and any of powder, dispersion, base, emulsion or solution can be used.
- the addition method is not particularly limited, and any known addition method may be used.
- the amount of the flocculant used is set to 0. 0 based on the weight of the substrate contained in the mixed solution.
- the amount is preferably 1% by weight or more and 3% by weight or less, more preferably 0.5% by weight or more and 1.5% by weight or less.
- the amount is not less than the above lower limit value, a sufficient agglomeration effect can be obtained, and when the amount is not more than the above upper limit value, the decomposition in the step (D) can be prevented from taking too much time and the productivity can be improved. Become. In addition, the effect of simplifying solid-liquid separation can be obtained.
- the solid obtained in the step (B) is recovered from the water-containing solvent.
- the solid matter recovered from the mixed solution contains a substrate having a ceramic or its precursor immobilized on at least a part of the surface and a flocculant. When recovered, it need not be composed of only these, and may contain a hydrous solvent, a neutralized salt, or the like.
- the method for recovering the solid matter is not particularly limited. Known methods for separating solids and liquids may be applied, such as natural filtration by the weight of the water-containing medium, vacuum filtration, pressure filtration, centrifugation, and decantation.
- the solid collected in the step (C) is heated to decompose the flocculant.
- the optical semiconductor particles of the present invention can be obtained.
- the flocculant is not limited to the case where it is completely removed, and a small amount of insoluble matter may remain.
- the flocculant must be decomposed.
- the solid is heated in the temperature range. At low temperatures, the flocculant tends to be insufficiently decomposed, and it takes a long time to decompose. When the temperature is high, the flocculant decomposes quickly, but the photo-semiconductor particles are easily deteriorated by heating.
- the heating temperature is preferably 400 ° C. or more and 100 ° C. or less, and more preferably 500 ° C. or more and 80 ° C. or less.
- the heating time must be selected depending on the heating temperature, and is not particularly limited. For example, low temperature processing takes a long time, and high temperature processing takes a short time. In the case of heating at 600 ° C., 0.5 hours or more and 24 hours or less are preferable, and 1 hour or more and 18 hours or less are more preferable.
- the decomposition of the flocculant is based on an oxidative decomposition reaction, the decomposition can be promoted by increasing the oxygen partial pressure in the atmosphere.
- the non-volatile compound when a non-volatile compound is by-produced or a non-volatile compound is used, the non-volatile compound may be incorporated into the optical semiconductor particles as an impurity. In this case, it is necessary to clean the optical semiconductor particles.
- a method for producing a non-volatile compound as a by-product for example, base particles are dispersed in a nitric acid aqueous solution, and sodium silicate is mixed therewith, and the silicon compound is immobilized on the surface of the base.
- sodium nitrate formed as a neutralized salt corresponds to a nonvolatile compound.
- a base particle is mixed in a solution in which calcium chloride and phosphate are dissolved as a ceramic raw material, and the compound of calcium and phosphorus is immobilized on the surface.
- a method for preparing a mixed solution containing the substrate particles In this method, the remaining amount of calcium chloride and phosphate used corresponds to the nonvolatile compound.
- the step (A) is a method using a strong sulfuric acid, a method using a sulfate as a by-product or using, or a method using a substrate, ceramics or a raw material of ceramics containing a sulfur compound.
- Sulfur compounds such as so 4 minutes and SO 3 minutes may be incorporated into the optical semiconductor particles and colored.
- the content of the sulfur compound is 0.5% by weight as the weight of S atoms with respect to the weight of the metal compound photo semiconductor contained in the photo semiconductor particles. In the following, it is desirable that the content be 0.3% by weight or less, more preferably 0.2% by weight or less. Therefore, cleaning may be necessary to adjust the sulfur compound content.
- step (A) When cleaning is required, when preparing the mixed solution in step (A), a method of removing impurities from the mixed solution using an ion exchange resin, or collecting solids in step (C) In this case, washing may be carried out by a method of passing a water-containing solvent through a cake made of solid matter formed on a filter, etc., independently of steps (A) to (D), You may implement as a manufacturing method including the process (E) which wash
- Process (E) includes process (B) and
- washing solvent used for washing
- the cleaning solvent is a hydrous solvent mainly composed of water.
- the washing solvent may include a water-soluble organic solvent having 1 to 4 carbon atoms among organic solvents classified into alcohols, ketones, ethers, or esters.
- the solvent used for washing is preferably water.
- the cleaning solvent may be adjusted to be acidic or basic in order to enhance the removal efficiency of impurities.
- the removal efficiency of cationic impurities such as alkali metals and alkaline earth metals can be enhanced.
- the basicity with alkali metal hydroxide or ammonia the removal efficiency of anionic impurities such as nitrate, chloride and sulfate can be enhanced.
- the acid basicity of the washing solvent may be carried out only in either one, or the acidic and basic can be used in combination. Swing between acidic and basic In some cases, it may be washed.
- the pH of the washing solvent adjusted to be acidic or basic may be adjusted to any pH as long as the flocculant is not decomposed and exhibits a coagulation effect.
- the pH is preferably in the range of 1 to 10, more preferably pH 2 to 8.
- the washing method includes, for example, a method in which the addition of the decant and the washing solvent is alternately repeated one or more times, a desalting treatment by ion exchange, a washing method in which redispersion in the washing solvent and filtration are alternately repeated one or more times, etc. It can be carried out by any known cleaning method. A flow cleaning in which the solvent is successively replaced is preferable, and an upward flow is more preferable. This is because it is possible to utilize the point that the solid matter (hereinafter referred to as “floc” where appropriate) in which coarse particles are formed by the effect of the flocculant exhibits a large sedimentation rate.
- Examples of such an apparatus include an apparatus in which a liquid mixture containing optical semiconductor particles and impurities is placed in a cylindrical container, water is supplied from the bottom of the container, and discharged from the top.
- Known devices such as thickeners and screw decanters can also be used.
- a flocculant may be added to re-form the flocs when the flocs break and fine particles are produced during the cleaning process.
- the flow rate of the liquid in the portion separating the floc and the washing solvent is divided into a horizontal vector component and a vertical vector component.
- cleaning is performed by controlling the supply amount of the cleaning solvent so that the rising speed of the liquid as a vector component in the vertical direction is lower than the sedimentation speed of the hook. If the ascending speed exceeds the settling speed of the floc, the floc flows out from the washing device and cannot be washed.
- the ascending speed is greater than 0 and equal to or less than the sedimentation speed of the floc.
- the rising flow rate is preferably 1% or more and 50% or less, and more preferably 3% or more and 25% or less, based on the floc sedimentation rate (100%).
- the photo-semiconductor particles produced in the present invention are photocatalysts having an Si oxide immobilized on the surface, the following can be exemplified.
- a photocatalyst comprising: a substrate that is a metal compound photo-semiconductor; a silicon oxide film that substantially does not have pores covering the substrate;
- alkali metal examples include lithium, sodium, potassium, rubidium, cesium, and francium. These alkali metals may contain only 1 type, and may contain 2 or more types.
- the metal strength metal content is preferably from 10 ppm to 1 000 ppm, more preferably from 10 ppm to 500 ppm. When the alkali metal content is within the above range, heat resistance is excellent, which is preferable. When an alkali metal is contained, a silicon oxide film with improved photocatalytic activity is formed.
- the amount of silicon supported is a calculated value calculated from the amount of silicon contained in the photocatalyst having the Si oxide immobilized on the surface and the surface area.
- the silicon loading is preferably 0.1 2 mg or more and 1.5 mg or less, more preferably 0.1 6 mg or more and 1. Omg or less.
- the amount of silicon supported is not less than the above lower limit, the activation effect by the silicon oxide film is improved, and a sufficient photolytic activity effect is obtained. Further, when the amount of silicon supported is not more than the above upper limit value, a sufficient proportion of the substrate in the photocatalyst can be secured, and good photolytic activity can be obtained.
- the sulfur compound is preferably 0.5% by weight or less, more preferably 0.3% by weight or less, and most preferably it is not contained.
- the presence or absence of pores derived from the silicon oxide film is determined by determining the pore volume distribution of the photo-semiconductor particles in which the ceramic is silicon oxide and the pores of the particles corresponding to the substrate of the photo-semiconductor particles. It can be determined by comparing the volume distribution. Specifically, it can be determined by the following methods (1) to (4).
- the photolytic activity may be difficult to improve.
- the reason for this is not always clear, but the following reasons are presumed.
- light scattering and reflection on the silicon oxide film is likely to occur due to the presence of the pores, the amount of ultraviolet light reaching the substrate having photocatalytic activity is reduced, and the amount of holes and electrons generated by photocatalytic excitation is reduced.
- the one with pores is decomposed from the substrate with photocatalytic activity as a result of the thickness of the silicon oxide film being increased by the volume of the pores compared to the one without pores. It is presumed that sufficient photodegradation activity cannot be obtained because the physical distance from the target organic substance becomes large.
- Examples of the photocatalyst in which the calcium compound produced in the present invention is immobilized on the surface include the following.
- the substrate is described as “titanium oxide particles” having excellent photocatalytic activity.
- the calcium phosphate apatite is immobilized on at least a part of the surface.
- Titanium oxide particles
- the calcium compound is effective as an adsorbent or a spacer.
- a photocatalyst has excellent adsorption performance, and deterioration of an organic base material (a base material made of an organic substance such as a resin) hardly occurs.
- “deterioration of the organic base material” means that when the photocatalyst is added to the organic base material, the organic base material is decomposed and deteriorated by the decomposing power of the photocatalyst.
- the spacer effect of calcium compounds in the photocatalysts of (X) to (z) This is due to the structure in which the child does not directly contact the organic substrate. Therefore, it is difficult for the organic base material to be decomposed while maintaining the decomposing power of the photocatalyst.
- the amount of the calcium compound is preferably 0.5% by weight or more and 20% by weight or less, more preferably 3% by weight or more and 10% by weight or less, as the weight percent of Ca ions with respect to the total weight of the photocatalyst. .
- the amount of the calcium compound is not less than the above lower limit, the effect as an adsorbent or a spacer can be sufficiently obtained.
- the amount of the calcium compound is not more than the above upper limit value, the effect of suppressing deterioration of the organic base material can be obtained while maintaining the photocatalytic activity.
- the calcium compound may be of a single composition, but may be a mixture or composite of two or more compounds.
- calcium phosphate when calcium phosphate is immobilized, calcium carbonate generated based on carbon dioxide in the air may be immobilized together, or other inorganic ions may be included.
- the optical semiconductor particles obtained in the present invention have no difference in performance as compared with those produced without using a flocculant. Therefore, there are no particular restrictions on applications. For example, it can be blended as a UV shielding agent or pigment in the same manner as known ones in applications such as cosmetics, sunscreen creams, synthetic resins and paints. Further, as a photocatalyst, it can be used for a photocatalyst-containing body exhibiting functions such as air purification, water purification, deodorization, antibacterial, antifouling, and self-cleaning.
- the photocatalyst-containing body includes an inorganic molded body containing a photocatalyst, an organic resin molded body containing a photocatalyst, a material having a photocatalytic coating film (the substrate may be either an inorganic compound or an organic compound), a photocatalyst paint,
- photocatalyst sprays, etc. which can be used in the following application fields and products: Ceramic filters and glass filters (air conditioners, refrigerators, humidifiers, dehumidifiers, cookers, air purifiers, dust collectors , Wastewater treatment equipment, etc.), paving asphalt, structural materials for RC buildings, humidity control building materials such as diatomaceous earth or zeolite, ceiling materials (rock wool, gypsum, etc.), tile joint materials, concrete blocks and concrete Rocking block, tile (exterior, paving, flooring, etc.), tile, sizing Materials, sealing materials, plastering materials such as building rubber, building stones, road stones such as paving
- the alkali metal content was measured using a fluorescent X-ray analyzer (LAB CENTER XRE-1700, Shimadzu Corporation). The amount detected in this measurement was quantified using an atomic absorption photometer (Z-5000, Hitachi, Ltd.).
- Quantification was performed using a fluorescent X-ray analyzer (LAB CENTER XRE-1 700, Shimadzu Corporation).
- X-ray diffraction (XRD) measurements were performed and calculated using the Sierra suite.
- polymer flocculant Acofloc registered trademark of Mitsui Chemicals Aqua Polymer
- N 21 0 Mitsubishi Chemicals Aqua Polymer Co., Ltd., nonionic, modified acrylic amide polymer
- a liquid mixture 2 was obtained in the same manner as in Example 1 (1_A).
- the entire amount of the coarse particle liquid 2 was transferred to a glass 50 Om L beaker and allowed to stand for 30 minutes, and then the supernatant liquid was decanted off. Next, washing was performed by repeating the following (a) to (d) five times.
- This photo-semiconductor particle 2 is a white powder, with a silicon content of 5.7 wt%, a specific surface area of 195.99 m 2 / g, a silicon loading of 1/29 m 2 , and a sodium content of 1 It was 50 p pm.
- liquid A was maintained at 35 ° C. and stirred
- liquid B was added dropwise at 3 ml / min to obtain liquid mixture C.
- the pH of mixture C at the end of dropping was 3.9.
- the mixture C was stirred for 16 hours while maintaining the temperature at 35 ° C. to obtain a mixture 3.
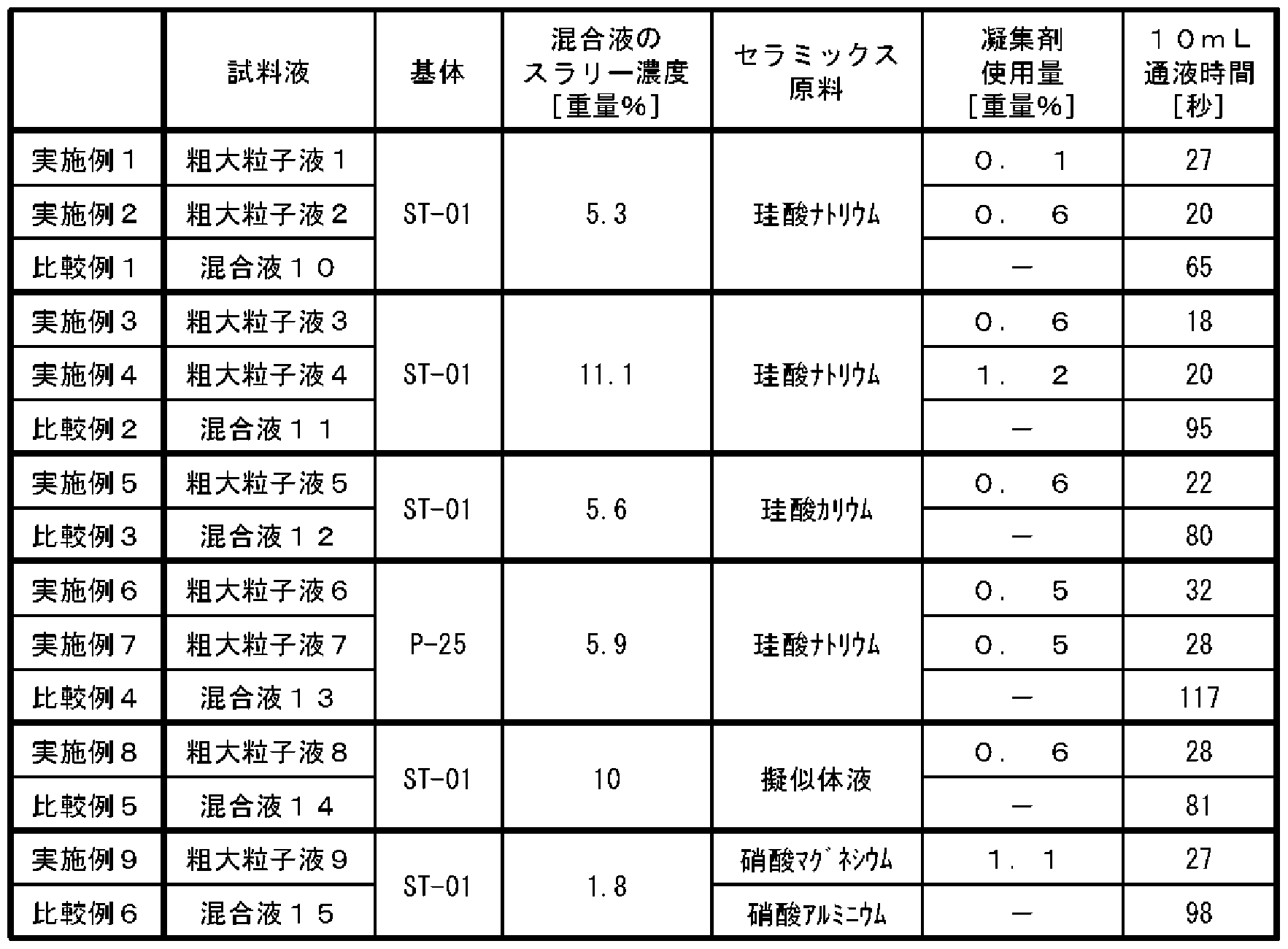
- the slurry concentration of mixture 3 is 11.1% by weight.
- Example 1 except that 1/3 of the mixture 3 was used instead of the mixture 1 and that 21 8 mg of the commercially available flocculant N 2120 was used and was added in 23 portions.
- coarse particle liquid 3 was obtained.
- Coarse particle liquid 3 was stirred for 30 minutes and left to stand for 30 minutes.
- White floc settled on the bottom of the flask, and the supernatant liquid was clear.
- the amount of the polymer flocculant used corresponds to 0.6% by weight.
- the flocs settled when stirring was stopped the length at which the boundary between the sludge and the supernatant liquid descended in 1 minute was measured to determine the floc sedimentation rate. As a result, the sedimentation rate of floc was 3.7 cm / min.
- the coarse particle solution 3 was transferred to a glass 50 OmL beaker and allowed to stand for 30 minutes. Thereafter, the supernatant was decanted off. Next, glass column tube (inner diameter 9 Omm, length 300 mm, lower end; 2-way cock included, upper end; 4 mm inner diameter glass tube can be held at any height and sealed except for inside the glass tube. ) was fixed vertically, the bottom end cock was closed, the top end plug was opened, and the contents of the beaker were transferred into the column. After closing the stopper at the upper end and opening the cock at the lower end, pure water was supplied from the lower end at a rate of 1 OmL per minute for 5 hours, and drainage was discharged through the glass pipe at the upper end.
- the height of the glass tube was adjusted so that the liquid volume inside the column tube was 40 Om L. Note that the rising velocity of the liquid in the column tube, calculated from the supply rate of pure water and the inner diameter of the column tube, is 0.16 cm / min, which corresponds to 4% of the floc sedimentation rate.
- the supply of pure water was stopped, and the contents of the column tube were filtered under reduced pressure to recover the solid matter.
- the collected white solid was placed in a magnetic dish and heated in an air atmosphere at 120 ° C for 3 hours using an electric furnace. The temperature was continuously raised to 600 ° C, and then kept at 600 ° C for 3 hours. After heating 42.5 g of photo-semiconductor particles 3 were obtained by standing until the temperature reached room temperature.
- This optical semiconductor particle 3 is a white powder having a silicon content of 6.1% by weight, a specific surface area of 209.8 m 2 / g, a silicon loading of 0.29 mg per 1 m 2 of surface area, and a sodium content of 1 It was 70 ppm.
- Example 1 Same as Example 1 (1 _B) except that the mixture 4 was used instead of the mixture 1 and that 435 mg of the polymer flocculant N 210 was added in 36 portions. Coarse particle liquid 4 was obtained. The amount of polymer flocculant used is equivalent to 1.2% by weight.
- This photo-semiconductor particle 4 is a white powder with a silicon content of 6.4% by weight, a specific surface area of 212.3 m 2 / g, and a silicon load of 0.30 mg per 1 m 2 of surface area, a sodium content. Was 200 p pm.
- Titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, specific surface area 300 m 2 / g, crystallite diameter 6 nm, N a content 1 400 p pm) 20. 4 g was dispersed to prepare a solution A.
- a beaker 88.2 g of water and an aqueous solution of potassium silicate (manufactured by Wako Pure Chemical Industries, Ltd., Si 0 2 content 28% by weight) 1 1.7 g were added and stirred to make B solution.
- the liquid A was kept at 25 ° C.
- Example 1 Same as Example 1 (1 _B), except that mixed solution 5 was used instead of mixed solution 1 and that 109 mg of polymer flocculant N 2120 was added in 10 batches. Thus, a coarse particle liquid 5 was obtained.
- the amount of the polymer flocculant used corresponds to 0.6% by weight.
- This optical semiconductor particle 5 is a white powder, silicon content 4.9% by weight, specific surface area 193.9 m 2 / g, silicon loading 0.25 mg per 1 m 2 surface area, sodium content 80 p pm, potassium content was 100 ppm.
- Liquid A 100 g of water in a beaker and sodium silicate aqueous solution (SiO 2 content 29 wt%, Na 2 O content 9.5 wt 0 / o, JISK 1 408 "Water Glass No. 3") 2.4 g was added and stirred to make B liquid. Liquid A was kept at 35 ° C.
- liquid B was added dropwise at 2 m I / min to obtain liquid mixture C.
- the pH of the mixed solution C at the end of dropping was 2.9.
- the mixture C was stirred for 72 hours while maintaining the temperature at 35 ° C. to obtain a mixture 6.
- the slurry concentration of the mixed solution 6 is 5.9% by weight.
- photo-semiconductor particles 6 were obtained in the same manner as in Example 2 (2-C) except that the coarse particle solution 6 was used instead of the coarse particle solution 2.
- This photo-semiconductor particle 6 is a white powder, with a silicon content of 1.3% by weight, a specific surface area of 52.9 m2 / g, a silicon loading of 0 25 mg per 1 m2 of surface area, and a sodium content of 40 ppm. Met.
- a liquid mixture 7 was obtained in the same manner as in Example 6 (6-A).
- Example 6 In the same manner as in Example 6 (6-B), a coarse particle liquid 7 was obtained. After the coarse particle liquid 7 was left stirring for 30 minutes, a white floc had settled at the bottom of the flask, and the supernatant liquid was transparent. The amount of polymer flocculant used corresponds to 0.5% by weight.
- the coarse particle liquid 7 was filtered under reduced pressure to collect a solid.
- the collected white solid was put in a magnetic dish and heated at 120 ° C. for 3 hours in an air atmosphere using an electric furnace. The temperature was continuously raised to 600 ° C, and then kept at 600 ° C for 3 hours. After heating, the mixture was allowed to stand until the room temperature was reached, and 29.9 g of photosemiconductor particles 7 were obtained.
- This photo-semiconductor particle 7 is a white powder with a silicon content of 1.3% by weight, a specific surface area of 54.2 m 2 / g, a silicon loading of 0.24 mg per 1 m 2 of surface area, and a sodium content of 820 ppm. there were.
- Example 1 (1 -B), except that the mixture 8 was used instead of the mixture 1 and that the commercially available polymer flocculant N 2120 was added in 57 mg, powdered in 5 portions. In the same manner, coarse particle liquid 8 was obtained. After the coarse particle liquid 8 was stopped stirring and allowed to stand for 30 minutes, white floc settled on the bottom of the flask, and the supernatant liquid was transparent. The amount of polymer flocculant used corresponds to 0.6% by weight.
- the coarse particle liquid 8 was transferred to a glass 20 OmL beaker and allowed to stand for 30 minutes, and then the supernatant liquid was decanted off. Next, the following (a) to (d) were repeated 5 times for washing.
- titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, specific surface area 300 m 2 / g, Crystallite diameter 6 nm, Na content 1 400 p pm) 1 5.37 g was dispersed to prepare X solution. At this time, the pH of the solution X was 10.9. Next, 280 g of water, 6.39 g of magnesium nitrate hexahydrate, and 3.09 g of aluminum nitrate nonahydrate were placed in a beaker and stirred to make Y solution.
- the Y solution was added dropwise at 1 Om I / min while stirring the X solution to obtain a mixed solution Z.
- the pH of the mixed solution Z at the end of dropping was 9.2.
- the mixture Z was stirred for 20 hours while maintaining the temperature at 64 ° C. to obtain a mixture 9.
- the slurry concentration of the mixed solution 9 is 1.8% by weight.
- the coarse particle solution 9 was obtained in the same manner as in Example 1 (1_B) except that the charged particles were different.
- the amount of polymer flocculant used is equivalent to 1.1% by weight.
- photo-semiconductor particles 9 15.1 g was obtained in the same manner as in Example 2 (2-C) except that coarse particle liquid 9 was used instead of coarse particle liquid 2.
- This light half Conductor particle 9 was a white powder and contained 2.0% by weight of Mg and 1.4% by weight of AI by ICP spectroscopy.
- Example 2 In the same manner as in Example 1, a mixed solution 10 was obtained.
- the mixture solution 10 remained cloudy even after stirring was stopped and left for 30 minutes.
- photo semiconductor particles 10 were obtained from the total amount of the mixed solution 10. These optical semiconductor particles 10 are white powder, silicon content 5.4% by weight, specific surface area 190.4 m 2 / g, silicon loading per 1 m 2 surface area 0.28 mg, sodium content 1 It was 50 p pm.
- mixture 1 One third of the mixture 3 was divided into mixture 1 1.
- the mixture 11 remained cloudy even after stirring was stopped and left for 30 minutes.
- the mixed liquid 11 was used in the same manner as in Example 1 (1-C) to obtain 40.9 g of the optical semiconductor particles 11.
- This photo-semiconductor particle 11 is a white powder, with a silicon content of 6.2% by weight, a specific surface area of 218.6 m 2 / g, a silicon loading per 1 m 2 of surface area of 0.28 mg, and a sodium content of 1 80 p pm.
- optical semiconductor particles 1 2 19.5 g was obtained in the same manner as in Example 1 (1-C) using the mixed liquid 12 instead of the mixed liquid 1.
- the optical semiconductor particles 1 2 is a white powder powder, silicon content 4.8 wt%, specific surface area 1 86. 0 m 2 / g, silicon supported amount per surface area of 1 m 2 is 0. 26 mg, sodium content 60 It was ppm and the rhodium content was 90 p pm.
- the mixed liquid 13 was used in the same manner as in Example 1 (1-C) to obtain 21.1 g of the optical semiconductor particles 13.
- This photo-semiconductor particle 13 is a white powder with a silicon content of 1.4% by weight, a specific surface area of 56.4 m 2 / g, a silicon loading per 1 m 2 of surface area of 0.22 mg, and a sodium content of 40 p. It was pm.
- the mixed solution 14 was put into 10 centrifuge tubes at a rate of 1 O g, and each of the following (a) to (d) was repeated 5 times for washing.
- This photo-semiconductor particle 13 was a white powder and contained 4.1% by weight of Ca and 1.8% by weight of P by fluorescent X-ray analysis.
- a liquid mixture 15 was obtained in the same manner as Example 9 (9-A). Mixture 15 remained cloudy even after stirring was stopped and left for 30 minutes.
- the liquid passing speed in the filtration treatment was measured by the following method. Coarse particle liquid 1-9 and mixed liquid 10-15 are diluted with water, part of the supernatant liquid is removed, or a centrifuge (Hokusan, H-18) is used. The slurry concentration was adjusted to 5 wt% by either method of treating at 3000 rpm for 3 minutes to settle the solids and then decanting off a portion of the supernatant.
- a glass filter holder for vacuum filtration (Advantech Toyo Co., Ltd., K GS-47, funnel volume 1 1 OmL, effective filtration area 9.6 cm 2 ) with a membrane filter inside, 2 Om L inside
- a vacuum filtration device was prepared in combination with a vacuum container equipped with a measuring cylinder.
- the photo-semiconductor particles 1 to 15 were suspended in a methylene blue aqueous solution, irradiated with light, and the methylene blue concentration in the liquid was quantified by spectroscopic analysis to test the photolysis activity.
- the test operation method is as follows.
- Standard suspension cell made of quartz (Tosoh_ Quartz Co., Ltd., outer dimensions 1 2.5 X 1 2.5 X 45 mm optical path width 10 mm, optical path length 10 mm, volume 4.5 cc) and stirred with a magnetic stirrer.
- light was irradiated for 5 minutes from the outside / lateral direction of the spectroscopic cell. Light irradiation was performed through a quartz filter container filled with pure water using the light source device S X_UI 15 1 XQ (Usio Electric Co., Ltd., 15 OW Xenon short clamp) as a light source.
- the amount of irradiation light was 5.0 mW / cm 2 when the highest luminance part was measured with an ultraviolet illuminance meter UVD-3650 PD (Usio Electric Co., Ltd., test wavelength 36 5 nm). After irradiation, the suspension in the spectroscopic cell was collected and used as a sample after light irradiation.
- Membrane filter 1 (Toyo Roshi Kaisha, Ltd., DIMSIC—13 HP) was attached to an all plastic 5 cc syringe. The sample suspensions before and after the light irradiation were put into each, and extruded with Biston to remove the photo semiconductor particles. At that time, discard the first half of the filtrate and collect the latter half of the filtrate in a semi-micro mouth type disposable cell for analysis of visible light (made of polystyrene, optical path width 4 mm, optical path length 1 Omm, volume 1.5 cc). did.
- the absorbance at a wavelength of 680 nanometers was measured, and the methylene blue concentration was calculated.
- the reason for discarding the first half of the filtrate is to avoid the influence of the phenomenon that the concentration of methylene blue is adsorbed on the membrane filter attached to the syringe and the concentration changes at the beginning of the flow. This is because, if about L is passed, the adsorption is saturated and the concentration of the sample solution can be measured correctly.
- the photodegradation activity is based on the initial concentration of methylene blue as the methylene blue concentration from the methylene blue concentration before the light irradiation, and as the methylene blue adsorption rate before the light irradiation as the methylene blue concentration after the light irradiation as the methylene blue decomposition rate. It is shown in Table 2.
- Photo-semiconductor particles 1-7, 10-1-13, and titanium oxide ST-01 and P_2 in the desorption process under liquid nitrogen (7 7 K) using autosoap (manufactured by Cantachrome) 5 nitrogen adsorption isotherms were measured. Titanium oxide was used after baking at 200 ° C. for 3 hours and drying.
- the production method according to the present invention facilitates solid-liquid separation when producing optical semiconductor particles in which ceramics are immobilized on the surface of a metal compound optical semiconductor and the performance is modified. Does not affect the performance. Therefore, it can be used for the production of optical semiconductor particles used as cosmetics, pigments, photocatalysts and the like.
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Catalysts (AREA)
- Inorganic Compounds Of Heavy Metals (AREA)
Abstract
Description
明 細 書 Specification
光半導体粒子の製造方法 Method for producing optical semiconductor particles
技術分野 Technical field
[0001 ] 本発明は、 金属化合物光半導体である基体と、 該基体の表面の少なくとも 一部に固定化されたセラミックスからなる光半導体粒子の製造方法に関する 背景技術 TECHNICAL FIELD [0001] The present invention relates to a base that is a metal compound optical semiconductor, and a method for producing optical semiconductor particles comprising ceramics immobilized on at least a part of the surface of the base.
[0002] チタニア、 酸化亜鉛などの金属化合物光半導体は、 そのバンド幅に相当す るエネルギーを有する光を吸収する性質を示す。 そのため、 化粧料や樹脂顏 料などの分野で紫外線遮蔽剤として応用展開が図られている。 また、 光を吸 収した金属化合物光半導体に生じる正孔と電子の高い反応性に着目し、 「光 触媒」 として、 水質浄化、 防汚、 抗菌、 脱臭、 大気浄化などの環境浄化へ応 用することも試みられている。 [0002] Metal compound optical semiconductors such as titania and zinc oxide exhibit the property of absorbing light having energy corresponding to the bandwidth. For this reason, application development is being attempted as an ultraviolet shielding agent in the fields of cosmetics and resin materials. Focusing on the high reactivity of holes and electrons generated in metal compound photo-semiconductors that absorb light, the photocatalyst is applied to environmental purification such as water purification, antifouling, antibacterial, deodorization, and air purification. Attempts have also been made.
[0003] これらの用途に金属化合物光半導体を用いる場合、 金属化合物光半導体を そのまま利用する場合もあるが、 その表面に異なる化合物を固定化して改良 を図ることもある。 例えば、 表面を緻密なシリカにより被覆された金属酸化 物粒子 (特許文献 1を参照) 、 表面を珪素化合物とアルミ化合物で多層被覆 した顔料 (特許文献 2を参照) 、 シリカ水和物の被覆層を有する塩基性ガス 除去用酸化チタン光触媒 (特許文献 3を参照) 、 実質的に細孔を有さない酸 化珪素膜で被覆した高活性光触媒 (特許文献 4を参照) 、 リン酸カルシウム で一部被覆された光触媒 (特許文献 5を参照) が挙げられる。 [0003] When a metal compound optical semiconductor is used for these applications, the metal compound optical semiconductor may be used as it is, but improvement may be achieved by immobilizing different compounds on the surface. For example, metal oxide particles whose surface is coated with dense silica (see Patent Document 1), pigments whose surface is coated with a silicon compound and an aluminum compound (see Patent Document 2), silica hydrate coating layer Titanium oxide photocatalyst for basic gas removal (see Patent Document 3), high-activity photocatalyst coated with a silicon oxide film substantially free of pores (see Patent Document 4), partially coated with calcium phosphate Photocatalysts (see Patent Document 5).
[0004] 金属化合物光半導体の表面に異なる化合物を固定化する方法は、 金属化合 物光半導体に、 ガス状の原料を反応させる気相法 (特許文献 6を参照) や、 溶媒中で処理する液相法がある。 このうち液相法は除熱が容易に行える点や 反応させる原料の量を制御しやすい点で優れている。 液相法の例としては、 塩基性水溶液中で加熱しながら珪酸塩と硫酸を滴下する方法 (特許文献 7を 参照) 、 有機溶媒中で光半導体の表面に予め水を吸着させ、 金属アルコキシ ドを光半導体の表面で加水分解する方法 (特許文献 8を参照) 、 p H 5以下 の酸性水系媒体中で珪酸塩を用いて被覆する製造方法 (特許文献 4を参照) 、 N a、 :、 C l、 C a、 P、 M g等のイオンを含む擬似体液に浸漬する方 法 (特許文献 5を参照) が挙げられる。 [0004] A method of immobilizing different compounds on the surface of a metal compound optical semiconductor includes a gas phase method in which a gaseous raw material is reacted with a metal compound optical semiconductor (see Patent Document 6), or treatment in a solvent. There is a liquid phase method. Among these, the liquid phase method is excellent in that heat removal can be easily performed and the amount of raw materials to be reacted can be easily controlled. Examples of liquid phase methods include a method in which silicate and sulfuric acid are dropped while heating in a basic aqueous solution (see Patent Document 7), water is adsorbed in advance on the surface of an optical semiconductor in an organic solvent, and a metal alkoxy A method of hydrolyzing a metal on the surface of an optical semiconductor (see Patent Document 8), a production method of coating with silicate in an acidic aqueous medium having a pH of 5 or less (see Patent Document 4), Na, , Cl, Ca, P, Mg, etc. (see Patent Document 5).
凝集剤を用いて固液分離を簡便にすることは公知であり、 工業的には廃水 や汚水の処理に利用されている。 凝集剤の使用により、 液中に分散した微細 粒子をつなぎ合わせて、 粗大粒子、 いわゆるフロックが形成される。 このた め、 凝集剤の効果として、 固形物が分離しやすくなる。 金属化合物光半導体 の製造方法において、 凝集剤を用いて固液分離を簡便にする例としては、 多 塩基鉱酸、 力ルポキシル基および/または水酸基を 2以上有する有機酸また はフエノール類、 並びにそれらの塩の少なくとも 1種と、 有機高分子凝集剤 の少なくとも 1種とで処理する方法 (特許文献 9を参照) 、 および硫酸チタ ンの加水分解で生じたメタチタン酸を高分子凝集剤で凝集する方法 (特許文 献 1 0を参照) が開示されている。 し力、し、 特許文献 9および 1 0には、 金 属化合物光半導体である基体と、 該基体の表面の少なくとも一部に固定化さ れたセラミックスからなる光半導体粒子の製造方法において、 凝集剤を用い ることに関しては、 何ら記載がない。 It is known to simplify solid-liquid separation using a flocculant, and it is industrially used to treat wastewater and sewage. By using the flocculant, fine particles dispersed in the liquid are joined together to form coarse particles, so-called flocs. For this reason, the solid matter is easily separated as an effect of the flocculant. Examples of simplifying solid-liquid separation using a flocculant in a method for producing a metal compound optical semiconductor include polybasic mineral acids, organic acid or phenols having two or more hydroxyl groups and / or hydroxyl groups, and those A method of treating with at least one of the above salts and at least one organic polymer flocculant (see Patent Document 9), and metatitanic acid generated by hydrolysis of titanium sulfate is aggregated with the polymer flocculant A method (see Patent Document 10) is disclosed. In Patent Documents 9 and 10, in a method for producing optical semiconductor particles comprising a base that is a metal compound optical semiconductor and ceramics immobilized on at least a part of the surface of the base, There is no mention of the use of chemicals.
特許文献 1 :特許 3570730号公報 Patent Document 1: Japanese Patent No. 3570730
特許文献 2:特許 1 539263号公報 Patent Document 2: Japanese Patent No. 539263
特許文献 3:特開 2002 _ 1 59865号公報 Patent Document 3: Japanese Patent Application Laid-Open No. 2002_159865
特許文献 4:特願 P CT/J P 2006/302542号 Patent Document 4: Japanese Patent Application No. P CT / J P 2006/302542
特許文献 5:特許 3275032号公報 Patent Document 5: Japanese Patent No. 3275032
特許文献 6:特許 1 895322号公報 Patent Document 6: Japanese Patent No. 1 895322
特許文献 7:特開平 02— 296726号公報 Patent Document 7: Japanese Patent Laid-Open No. 02-296726
特許文献 8:特許 2832342号公報 Patent Document 8: Japanese Patent No. 2832342
特許文献 9:特開昭 53— 39296号公報 Patent Document 9: Japanese Patent Laid-Open No. 53-39296
特許文献 10:特許 3554803号公報 Patent Document 10: Japanese Patent No. 3554803
発明の開示 [0006] 光半導体粒子を紫外線遮蔽剤として利用する分野では、 可視光に対して透 明であり、 且つ、 紫外線を吸収する材料が求められている。 そこで、 金属化 合物光半導体の粒子径を可視光の波長の半分以下にした光半導体粒子が提案 されている。 また、 光半導体粒子を光触媒として利用する分野では、 脱臭や 大気浄化などの効果が高い点を特長とする比表面積の大きい材料が提案され ている。 いずれも従来のものよりも粒子径を微細にすることで、 可視光に対 する透明性、 あるいは大きな比表面積を達成している。 しかしながら、 微細 な粒子を液相法で製造する場合、 取り扱う光半導体粒子が微細な粒子を含む ために、 以下の点 (a ) または (b ) が問題となる : Disclosure of the invention [0006] In the field of utilizing optical semiconductor particles as an ultraviolet shielding agent, a material that is transparent to visible light and absorbs ultraviolet rays is required. Therefore, optical semiconductor particles have been proposed in which the particle diameter of the metal compound optical semiconductor is less than half the wavelength of visible light. In the field where photo semiconductor particles are used as a photocatalyst, a material having a large specific surface area, which is characterized by high effects such as deodorization and air purification, has been proposed. In both cases, by making the particle diameter finer than the conventional one, transparency to visible light or a large specific surface area is achieved. However, when producing fine particles by the liquid phase method, the following points (a) or (b) are problematic because the handled optical semiconductor particles contain fine particles:
( a ) ろ過で光半導体粒子を回収する場合、 フィルターの通液速度が小さい ことにより、 処理に時間がかかる、 (a) When collecting optical semiconductor particles by filtration, the processing speed is long due to the low flow rate of the filter.
( b ) 光半導体粒子の回収をシックナ一で行う場合、 すなわち光半導体粒子 の沈降によって濃縮分離をする場合、 沈降に要する時間が長いことにより、 処理に時間がかかる。 (b) When recovery of the photo semiconductor particles is performed with a thickener, that is, when concentration separation is performed by sedimentation of the photo semiconductor particles, the time required for the settling is long, so that the processing takes time.
その結果、 光半導体粒子を溶媒から簡便に回収することが出来ないという問 題がある。 As a result, there is a problem that the optical semiconductor particles cannot be easily recovered from the solvent.
[0007] そこで本発明は、 光半導体粒子を、 液相法で製造するに際して、 固液分離 を簡便に実施でき、 且つ、 得られた前記光半導体粒子の光半導体としての作 用に影響しない製造方法を提供することを課題としている。 [0007] Therefore, the present invention provides a production method in which solid-liquid separation can be easily performed when producing optical semiconductor particles by a liquid phase method, and the operation of the obtained optical semiconductor particles as an optical semiconductor is not affected. The challenge is to provide a method.
[0008] 本発明者らは、 前記の課題を解決するため、 表面にセラミックスを固定化 した基体を含むスラリーを用いて鋭意検討した。 その結果、 凝集剤を加えて 前記基体を含有する粒子を凝集状態で固液分離し、 得られた固形物を加熱す ることにより、 凝集剤を分解し、 表面にセラミックスを固定化した基体から なる光半導体粒子を簡便に製造できること見出した。 それと共に、 得られた 光半導体粒子が、 凝集剤を用いないで製造した光半導体粒子と同等の性能を 示すことを見出し、 発明を完成させるに至った。 すなわち、 本発明によれば 、 以下の光半導体粒子の製造方法が提供される。 [0008] In order to solve the above-mentioned problems, the present inventors diligently studied using a slurry containing a substrate having a ceramic surface fixed thereto. As a result, the flocculant is added to solid-liquid separate the particles containing the substrate in the agglomerated state, and the resulting solid is heated to decompose the flocculant and from the substrate having ceramics immobilized on the surface. It has been found that the optical semiconductor particles can be easily produced. At the same time, the inventors have found that the obtained optical semiconductor particles exhibit the same performance as the optical semiconductor particles produced without using a flocculant, and have completed the invention. That is, according to the present invention, the following method for producing optical semiconductor particles is provided.
[0009] [ 1 ] 金属化合物光半導体である基体と、 前記基体の表面の少なくとも一 部に固定化されたセラミックスからなる光半導体粒子の製造方法であって、 前記セラミックスが前記基体と異なる化合物であり、 且つ、 下記の工程 (A ) 〜 (D) からなる光半導体粒子の製造方法: [1] A base that is a metal compound optical semiconductor, and at least one of the surfaces of the base A method for producing optical semiconductor particles comprising ceramics fixed to a part, wherein the ceramics is a compound different from the substrate, and comprising the following steps (A) to (D) :
(A) 前記基体、 前記セラミックスまたはその原料、 および含水溶媒を混合 して、 表面の少なくとも一部に前記セラミックスまたはその前駆体が固定化 された前記基体および水を含む混合液を調製する工程、 (A) a step of mixing the substrate, the ceramic or its raw material, and a water-containing solvent to prepare a mixed solution containing the substrate and water on which the ceramic or its precursor is immobilized on at least a part of the surface;
(B) 前記混合液に凝集剤を添加し、 攪拌し、 表面の少なくとも一部に前記 セラミックスまたはその前駆体が固定化された前記基体および凝集剤を含む 固形物を得る工程、 (B) adding a flocculant to the mixed solution, stirring, and obtaining a solid body containing the base and the flocculant on which the ceramic or its precursor is fixed at least on a part of the surface;
(C) 前記固形物を、 前記含水溶媒から回収する工程、 (C) recovering the solid from the water-containing solvent,
(D) 前記固形物を加熱することにより凝集剤を分解して除去し、 光半導体 粒子を得る工程。 (D) A step of decomposing and removing the flocculant by heating the solid to obtain photo-semiconductor particles.
[2] 前記工程 (B) と (C) の間、 あるいは前記工程 (C) と (D) の 間に、 更に、 下記の工程 (E) を含む、 [1 ] に記載の光半導体粒子の製造 方法: [2] Between the steps (B) and (C), or between the steps (C) and (D), further comprising the following step (E): Production method:
(E) 表面の少なくとも一部に前記セラミックスまたはその前駆体が固定化 された前記基体および凝集剤を含む固形物を洗浄する工程。 (E) A step of washing the solid body containing the substrate and the flocculant on which the ceramic or its precursor is immobilized on at least a part of the surface.
[3] 前記工程 (E) が含水溶媒の流通によるものであって、 前記固形物 と洗浄排液とを分離する部分の液体の流速を、 水平方向のべク トル成分と垂 直方向のべク トル成分に分けた場合に、 液体の垂直方向のべク トル成分とし ての上昇速度が、 該固形物の沈降速度よりも遅くなるように含水溶媒の供給 量を制御して洗浄する、 [2] に記載の光半導体粒子の製造方法。 [3] The step (E) is based on the flow of the water-containing solvent, and the flow rate of the liquid in the portion separating the solid and the washing waste liquid is determined by the horizontal vector component and the vertical direction. When divided into vector components, washing is performed by controlling the supply amount of the hydrous solvent so that the rising rate of the liquid as a vector component in the vertical direction is slower than the settling rate of the solids. [2] The method for producing an optical semiconductor particle according to [2].
[4] 前記凝集剤が高分子凝集剤である、 [1 ] または [2] に記載の光 半導体粒子の製造方法。 [4] The method for producing optical semiconductor particles according to [1] or [2], wherein the flocculant is a polymer flocculant.
[5] 前記高分子凝集剤がノニオン型である、 [4] に記載の光半導体粒 子の製造方法。 [5] The method for producing an optical semiconductor particle according to [4], wherein the polymer flocculant is a nonionic type.
[6] 前記高分子凝集剤が、 アクリルアミ ドまたはその変性体からなる群 から選ばれる 1種以上の部分構造を繰り返し単位として含む高分子化合物で ある、 [4] に記載の光半導体粒子の製造方法。 [6] The polymer flocculant is a polymer compound containing one or more partial structures selected from the group consisting of acrylamide or a modified product thereof as a repeating unit. The method for producing an optical semiconductor particle according to [4].
[7] 前記工程 (D) の加熱温度が、 400°C以上、 1 000°C以下であ る、 [1 ] 、 [2] 、 または [4] のいずれか一項に記載の光半導体粒子の 製造方法 [7] The photo-semiconductor particle according to any one of [1], [2], or [4], wherein the heating temperature in the step (D) is 400 ° C. or higher and 1 000 ° C. or lower. The manufacturing method
[8] 前記基体が酸化チタンからなる [1 ] 、 [2] 、 [4] 、 または [ 7 ] のいずれか一項に記載の光半導体粒子の製造方法。 [8] The method for producing optical semiconductor particles according to any one of [1], [2], [4], and [7], wherein the substrate is made of titanium oxide.
[9] 前記セラミックスが酸化珪素からなる [1 ] または [8] に記載の 光半導体粒子の製造方法。 [9] The method for producing optical semiconductor particles according to [1] or [8], wherein the ceramic is made of silicon oxide.
[1 0] 前記工程 (A) 力 前記基体を含む含水溶媒と珪酸塩、 珪酸塩を 含む含水溶媒と前記基体、 および前記基体を含む含水溶媒と珪酸塩を含む含 水溶媒からなる群より選ばれる少なくとも一組を混合し、 かつ前記基体およ び珪酸塩の両方を含む混合液の p Hを 5以下に維持して混合する工程である 、 [9] に記載の光半導体粒子の製造方法。 [10] The step (A) force is selected from the group consisting of a hydrous solvent containing the substrate and a silicate, a hydrous solvent containing the silicate and the substrate, and a hydrous solvent containing the substrate and a hydrous solvent containing the silicate. The method for producing optical semiconductor particles according to [9], wherein at least one set is mixed and the pH of the mixed solution containing both the substrate and the silicate is maintained at 5 or less. .
[1 1 ] 前記酸化珪素が、 前記基体の表面の少なくとも一部に膜状に固定 化されているものであって、 窒素吸着法による 20〜500オングストロ一 ムの領域の細孔径分布測定において、 細孔がない酸化珪素膜である、 [1 0 ] に記載の光半導体粒子の製造方法。 [1 1] The silicon oxide is fixed in a film form on at least a part of the surface of the substrate, and in pore size distribution measurement in a region of 20 to 500 angstroms by a nitrogen adsorption method, The method for producing optical semiconductor particles according to [1 0], which is a silicon oxide film having no pores.
[1 2] 光半導体粒子の表面積 1 m 2当りの珪素担持量が、 0. 1 0 m g以 上、 2. Omg以下である、 [9] 乃至 [1 1 ] のいずれか一項に記載の光 半導体粒子の製造方法。 [1 2] The amount of silicon supported per 1 m 2 of the surface area of the optical semiconductor particles is 0.1 mg or more and 2. Omg or less, according to any one of [9] to [1 1] Method for producing optical semiconductor particles.
[1 3] 光半導体粒子がアルカリ金属を 1 p pm以上、 l O O O p pm以 下含有する、 [1 1 ] に記載の光半導体粒子の製造方法。 [1 3] The method for producing an optical semiconductor particle according to [1 1], wherein the optical semiconductor particle contains an alkali metal in an amount of 1 ppm or more and 1 O 2 O O ppm or less.
[1 4] 前記酸化珪素がアル力リ金属を含む、 [1 3] に記載の光半導体 粒子の製造方法。 [1 4] The method for producing optical semiconductor particles according to [1 3], wherein the silicon oxide contains an alkali metal.
[1 5] 前記セラミックスが、 カルシウムとリンの化合物からなる [8] に記載の光半導体粒子の製造方法。 [15] The method for producing optical semiconductor particles according to [8], wherein the ceramic is composed of a compound of calcium and phosphorus.
[1 6] 前記セラミックスが、 ァパタイ トである [1 5] に記載の光半導 体粒子の製造方法。 [1 7] 前記工程 (A) 力 擬似体液を用いてカルシウムおよびリンを含 む化合物を前記基体の表面に形成する工程である、 [1 5] に記載の光半導 体粒子の製造方法。 [16] The method for producing an optical semiconductor particle according to [15], wherein the ceramic is a parameter. [17] The method for producing optical semiconductor particles according to [15], wherein the step (A) is a step of forming a compound containing calcium and phosphorus on the surface of the substrate using a force simulated body fluid.
[1 8] 前記セラミックスが、 マグネシウムおよびアルミニウムの少なく ともいずれか 1種を構成成分とする酸化物からなるセラミックスである、 [ 8] に記載の光半導体粒子の製造方法。 [18] The method for producing optical semiconductor particles according to [8], wherein the ceramic is a ceramic made of an oxide containing at least one of magnesium and aluminum as a constituent component.
[1 9] 前記セラミックスが、 マグネシウムおよびアルミニウムの酸化物 の混合物、 またはマグネシウムおよびアルミニウムの複合酸化物からなるセ ラミックスである、 [1 8] に記載の光半導体粒子の製造方法。 [19] The method for producing optical semiconductor particles according to [18], wherein the ceramic is a mixture of an oxide of magnesium and aluminum, or a ceramic composed of a composite oxide of magnesium and aluminum.
[20] 前記工程 (A) 、 前記基体とアルカリ金属塩とを含む含水溶媒 に、 マグネシウムおよびアルミニウムの水溶性の塩を少なくとも 1種以上含 む含水溶媒を混合する工程である、 [1 8] に記載の光半導体粒子の製造方 法。 [20] The step (A) is a step of mixing a hydrous solvent containing at least one or more water-soluble salts of magnesium and aluminum with a hydrous solvent containing the substrate and the alkali metal salt. [18] A method for producing the optical semiconductor particles described in 1.
[21 ] 光半導体粒子が光触媒である [9] 、 [1 5] 、 または [1 8] のいずれか一項に記載の光半導体粒子の製造方法。 [21] The method for producing optical semiconductor particles according to any one of [9], [15], and [18], wherein the optical semiconductor particles are a photocatalyst.
[0010] 本発明によれば、 金属化合物光半導体である基体と、 該基体の表面の少な くとも一部に固定化されたセラミックスからなる光半導体粒子を、 液相法で 製造するに際して、 固液分離を簡便に実施でき、 且つ、 得られた前記光半導 体粒子の光半導体としての作用に影響しない製造方法を提供することができ る。 [0010] According to the present invention, when producing a semiconductor substrate, which is a metal compound optical semiconductor, and optical semiconductor particles comprising ceramics fixed to at least a part of the surface of the substrate, by a liquid phase method, It is possible to provide a production method in which liquid separation can be easily performed and the obtained optical semiconductor particles do not affect the action as an optical semiconductor.
図面の簡単な説明 Brief Description of Drawings
[0011] 上述した目的、 およびその他の目的、 特徴および利点は、 以下に述べる好 適な実施の形態、 およびそれに付随する以下の画面によってさらに明らかに なる。 [0011] The above-described object and other objects, features, and advantages will be further clarified by a preferred embodiment described below and the following screens that accompany it.
[図 1]光半導体粒子 1の I o g微分細孔容積分布曲線 (実線) と、 その基体に 該当する二酸化チタン S T_01の I o g微分細孔容積分布曲線 (点線) と を示す図である。 FIG. 1 is a diagram showing an I og differential pore volume distribution curve (solid line) of optical semiconductor particle 1 and an I og differential pore volume distribution curve (dotted line) of titanium dioxide ST_01 corresponding to the substrate.
[図 2]光半導体粒子 5の I o g微分細孔容積分布曲線 (実線) と、 その基体に 該当する二酸化チタン S T _ 0 1の I o g微分細孔容積分布曲線 (点線) と を示す図である。 [Figure 2] Iog differential pore volume distribution curve (solid line) of photo-semiconductor particle 5 and its substrate It is a figure which shows Iog differential pore volume distribution curve (dotted line) of applicable titanium dioxide ST_01.
[図 3]光半導体粒子 6の I o g微分細孔容積分布曲線 (実線) と、 その基体に 該当する二酸化チタン P _ 2 5の I o g微分細孔容積分布曲線 (点線) とを 示す図である。 [Fig. 3] A diagram showing the I og differential pore volume distribution curve (solid line) of photo-semiconductor particle 6 and the I og differential pore volume distribution curve (dotted line) of titanium dioxide P _ 2 5 corresponding to the substrate. is there.
発明を実施するための最良の形態 BEST MODE FOR CARRYING OUT THE INVENTION
[0012] 本発明で製造される光半導体粒子は、 金属化合物光半導体である基体と、 該基体の表面の少なくとも一部に固定化されたセラミックスからなる。 そし て、 前記光半導体粒子は、 それぞれ独立して前記の構成であればよく、 すべ ての粒子が同じ構成であつてもよいし、 2種以上の異なる構成の粒子の混合 物であってもよい。 また、 一つの基体に 2種以上のセラミックスが固定化さ れていてもよい。 また、 ここでいう 「からなる」 の用語は、 他の成分が含ま れる場合を排除するものでなく、 言及した構成成分のみが含まれる場合、 お よびさらにその他の成分も含まれる場合の両方を意味するものとする。 The optical semiconductor particles produced in the present invention are composed of a base that is a metal compound optical semiconductor and ceramics immobilized on at least a part of the surface of the base. The photosemiconductor particles may be independently configured as described above, and all the particles may have the same configuration, or may be a mixture of two or more differently configured particles. Good. Further, two or more kinds of ceramics may be fixed to one substrate. In addition, the term “consisting of” does not exclude the case where other components are included, and includes both the case where only the mentioned components are included and the case where other components are also included. Shall mean.
[0013] 基体は、 金属化合物光半導体であれば、 特に制限はない。 例えば、 酸化チ タン、 酸化亜鉛、 酸化タングステン、 チタン酸ストロンチウムなどが挙げら れる。 基体は、 X線回折 (X R D ) から導かれる結晶子径が、 1〜5 0 0 n mの範囲にあるものが好ましい。 また、 形状にも特に限定はなく、 例えば、 球状、 回転楕円体状、 柱状、 幾何学形状、 薄片状等が挙げられる。 さらに、 表面の少なくとも一部にセラミックスが固定化された基体を、 本発明の基体 として用いることも可能である。 なお、 基体は、 1種のみを用いることが多 いが、 2種以上の混合物を用いてもよい。 [0013] The substrate is not particularly limited as long as it is a metal compound optical semiconductor. Examples include titanium oxide, zinc oxide, tungsten oxide, and strontium titanate. The substrate preferably has a crystallite diameter derived from X-ray diffraction (X R D) in the range of 1 to 500 nm. The shape is not particularly limited, and examples thereof include a spherical shape, a spheroid shape, a column shape, a geometric shape, and a flake shape. Furthermore, a substrate having ceramics immobilized on at least a part of its surface can be used as the substrate of the present invention. In many cases, only one type of substrate is used, but a mixture of two or more types may be used.
[0014] 光触媒の製造方法として本発明を実施する場合には、 光触媒能に優れてお り、 安全性や安定性にも優れる酸化チタンが好ましい。 酸化チタンとしては 、 例えば、 非晶質、 アナターゼ型、 ルチル型、 ブルツカイ ト型の二酸化チタ ンがあり、 このうちでは、 光触媒能に優れているアナターゼ型、 ルチル型、 または、 これらの混合物がより好ましい。 また、 これらに非晶質が少量含ま れていてもよい。 基体として、 金属化合物光半導体に 1種以上の遷移金属を 添加または担持したもの、 金属化合物半導体に 1 4族、 1 5族、 および/ま たは 1 6族の典型元素を 1種以上添加あるいは担持したもの、 2種以上の金 属化合物からなる光半導体、 2種以上の金属化合物半導体の混合物等を使用 してもよい。 [0014] When the present invention is carried out as a method for producing a photocatalyst, titanium oxide is preferable because it is excellent in photocatalytic activity and is excellent in safety and stability. Examples of titanium oxide include amorphous, anatase type, rutile type, and wurtzite type titanium dioxide, and among these, anatase type, rutile type, or a mixture thereof, which is superior in photocatalytic activity, is more suitable. preferable. These may contain a small amount of amorphous material. As a substrate, one or more transition metals are added to the metal compound optical semiconductor. Addition or support, Metal compound semiconductor with one or more typical elements of Group 14, 15 and / or Group 16 added or supported, Optical semiconductor composed of two or more metal compounds A mixture of two or more kinds of metal compound semiconductors may be used.
[0015] セラミックスは、 光半導体粒子を構成する基体と異なる化合物であれば、 特に制限はない。 したがって、 金属化合物光半導体であっても構わない。 例 えば、 基体が酸化チタンである場合に、 セラミックスが酸化亜鉛であっても 構わない。 化粧料、 顔料、 または光触媒の製造方法として本発明を実施する 場合には、 セラミックスは、 S i、 A I、 Z r、 C eの酸化物、 またはカル シゥム化合物が好ましい。 酸化物は、 水酸基、 吸着水などを含むものであつ ても良く、 単一酸化物、 二元系複合酸化物、 または多元系複合酸化物であつ てもよい。 そして前記の金属に限定されず、 他の金属元素を、 イオンまたは 金属として含むものであってもよい。 なお本発明の製造方法では、 工程 (D ) において、 凝集剤を分解するために加熱処理を施す。 したがって、 本発明 の製造方法で提供される光半導体粒子を構成するセラミックスは、 焼成され たセラミックスである。 [0015] The ceramic is not particularly limited as long as it is a compound different from the base constituting the optical semiconductor particles. Therefore, it may be a metal compound optical semiconductor. For example, when the substrate is titanium oxide, the ceramic may be zinc oxide. When the present invention is carried out as a method for producing cosmetics, pigments, or photocatalysts, the ceramic is preferably an oxide of Si, AI, Zr, or Ce, or a calcium compound. The oxide may include a hydroxyl group, adsorbed water, etc., and may be a single oxide, a binary complex oxide, or a multicomponent complex oxide. And it is not limited to the said metal, Other metal elements may be included as an ion or a metal. In the production method of the present invention, in the step (D), heat treatment is performed to decompose the flocculant. Therefore, the ceramic constituting the optical semiconductor particles provided by the production method of the present invention is a fired ceramic.
[001 6] カルシウム化合物としては、 カルシウムのリン酸塩または珪酸塩が挙げら れ、 ヒドロキシァパタイ ト、 フッ化ァパタイ ト、 他の金属イオンを含有する ヒドロキシァパタイ ト等、 一般に 『ァパタイ ト』 として知られる鉱物組成を 持つリン酸カルシウム塩、 並びに珪酸カルシウムが例示される。 [001 6] Calcium compounds include calcium phosphates or silicates, such as hydroxyapatite, fluorideapatites, and hydroxyapatites containing other metal ions. And calcium phosphate salt having a mineral composition known as Calcium silicate.
[001 7] 上記の酸化物または化合物のうち、 S iの酸化物がより好ましい。 S iの 酸化物を基体の表面に固定化すると、 該基体の耐熱性が向上する。 これによ り、 凝集剤の分解をより高い温度で実施できるようになる。 [001 7] Of the above oxides or compounds, an oxide of Si is more preferable. When the Si oxide is immobilized on the surface of the substrate, the heat resistance of the substrate is improved. This allows the flocculant to be decomposed at higher temperatures.
[0018] セラミックスの原料とは、 工程 (A ) において、 基体および含水溶媒と混 合した際に、 加水分解、 縮合、 析出等を起こして、 基体の表面にセラミック スまたはその前駆体を形成する物質のことである。 セラミックスの原料は 1 種単独または 2種以上を用いても構わない。 例えば、 金属のハロゲン化物、 硝酸塩、 または硫酸塩、 金属のアルコキシド、 金属錯体、 金属のォキソ酸塩 等が、 1種単独で用いる原料として挙げられる。 金属のォキソ酸塩としては 、 例えば S iのォキソ酸塩として、 ケィ酸ナトリウム、 ケィ酸カリウム等が 挙げられる。 また、 2種以上で用いられる原料として、 例えば、 金属の可溶 性の塩と可溶性のォキソ酸またはその塩の組み合わせ等が挙げられる。 前記 の組み合わせを原料として用いる場合は、 基体の表面に不溶性あるいは難溶 性の該金属のォキソ酸塩が析出して、 セラミックスまたはその前駆体となる 。 例えば、 塩化カルシウムとリン酸ナトリウムを原料とした場合、 リン酸力 ルシゥム化合物が析出する。 セラミックスが酸化珪素である場合、 珪酸、 珪 酸塩、 珪酸エステル、 ハロゲン化珪素、 オルガノハイ ドロジヱンポリシロキ サン等が原料として例示される。 また、 セラミックスまたはその前駆体の形 成のため、 沈殿剤や触媒を使用してもよい。 [0018] The ceramic raw material forms a ceramic or a precursor thereof on the surface of the substrate by causing hydrolysis, condensation, precipitation or the like when mixed with the substrate and the water-containing solvent in the step (A). It is a substance. Ceramic materials may be used alone or in combination of two or more. For example, metal halides, nitrates or sulfates, metal alkoxides, metal complexes, metal oxoacid salts And the like can be mentioned as raw materials used alone. Examples of the metal oxo acid salt include sodium silicate and potassium silicate as Si oxo acid salt. Examples of the raw material used in two or more types include a combination of a metal-soluble salt and a soluble oxoacid or a salt thereof. When the above combination is used as a raw material, an insoluble or hardly soluble oxoacid salt of the metal is precipitated on the surface of the substrate to become a ceramic or a precursor thereof. For example, when calcium chloride and sodium phosphate are used as raw materials, a phosphate compound is precipitated. When the ceramic is silicon oxide, examples of the raw material include silicic acid, silicate, silicate ester, silicon halide, organohydridopolysiloxane. In addition, a precipitating agent or a catalyst may be used for forming ceramics or a precursor thereof.
[0019] セラミックスの前駆体とは、 物理的あるいは化学的な処理によってセラミ ックスに変化する化合物のことである。 セラミックスの前駆体は、 本発明の 工程の中で、 セラミックスに変換されてもよいし、 別途セラミックスに変換 する工程を設けても構わない。 本発明の工程の中でセラミックスに変換され るものとしては、 工程 (D ) における加熱処理において、 酸化、 加水分解、 縮合反応等によりセラミックスになるものがある。 このような前駆体は、 例 えば、 水酸化アルミニウムが挙げられる。 水酸化アルミニウムは、 原料とし てアルミニウムのアルコキシドを用いて基体の表面に形成され、 工程 (D ) でアルミナに変換可能な化合物である。 [0019] A ceramic precursor is a compound that changes into a ceramic by a physical or chemical treatment. In the process of the present invention, the ceramic precursor may be converted into ceramics, or a process of converting into ceramics may be provided separately. Among the steps of the present invention, those converted into ceramics include those that become ceramics by oxidation, hydrolysis, condensation reaction, etc. in the heat treatment in step (D). An example of such a precursor is aluminum hydroxide. Aluminum hydroxide is a compound that is formed on the surface of a substrate using aluminum alkoxide as a raw material and can be converted to alumina in the step (D).
[0020] 含水溶媒とは、 水、 または主として水を含む混合溶媒である。 含水溶媒は 、 アルコール類、 ケトン類、 エーテル類、 またはエステル類に分類される有 機溶媒のうち、 炭素数 1以上 4以下で、 且つ水溶性の有機溶媒を含んでも構 わない。 含水溶媒を具体的に例示するとすれば、 水、 水とメチルアルコール 、 水とエチルアルコール、 水とイソプロパノール等の混合溶媒が挙げられる 。 これらの中では水が好ましい。 また、 これらの水または混合溶媒は、 1種 単独または 2種以上組み合わせて用いてもよい。 [0020] The water-containing solvent is water or a mixed solvent mainly containing water. The water-containing solvent may include a water-soluble organic solvent having 1 to 4 carbon atoms among organic solvents classified into alcohols, ketones, ethers, or esters. Specific examples of the water-containing solvent include water, water and methyl alcohol, water and ethyl alcohol, water and isopropanol, and the like. Of these, water is preferred. These water or mixed solvents may be used alone or in combination of two or more.
[0021 ] 工程 (A ) において、 基体、 セラミックスまたはその原料、 および含水溶 媒を混合する方法に特に制限はない。 例えば、 (a ) 含水溶媒に基体を混合 した後に、 セラミックスまたはその原料を混合する方法、 (b ) 含水溶媒と 基体を混合した混合液と、 含水溶媒とセラミツクスまたはその原料とを混合 した混合液との二つを混合する方法、 (c ) 含水溶媒に、 セラミックスまた はその原料を混合した後に、 基体を混合する方法、 (d ) 基体、 セラミック スまたはその原料、 および含水溶媒を含む混合液の p Hを 5以下に維持して 混合する方法、 (e ) セラミックスが二種以上の原料を反応させて形成され る場合において、 含水溶媒、 基体、 およびセラミックスの第一原料を混合し た混合液と、 含水溶媒とセラミックスの第二原料とを混合した混合液との二 つを混合する方法、 などが挙げられる。 混合は含水溶媒が液体として存在で きる条件であれば特に限定されず、 常温でも良いし、 冷却、 加熱、 加圧条件 で適宜行なってもよい。 また、 こうして調製された混合液は、 適宜、 熟成を 施しても構わない。 [0021] In the step (A), a substrate, ceramics or a raw material thereof, and water-containing There is no restriction | limiting in particular in the method of mixing a medium. For example, (a) a method of mixing a ceramic or its raw material after mixing the substrate with a hydrous solvent, (b) a mixed solution of mixing the hydrous solvent and the substrate, and a mixed solution of mixing the hydrous solvent and ceramics or its raw material (C) A method of mixing a ceramic or its raw material in a water-containing solvent and then mixing the substrate, (d) A mixed solution containing the substrate, the ceramic or its raw material, and a water-containing solvent (E) When the ceramic is formed by reacting two or more kinds of raw materials, the mixture containing the hydrous solvent, the substrate, and the first raw material of the ceramic is mixed. And a method of mixing two of a liquid and a mixed liquid obtained by mixing a hydrous solvent and a second ceramic raw material. The mixing is not particularly limited as long as the water-containing solvent can exist as a liquid, and may be performed at room temperature or appropriately under cooling, heating, and pressurizing conditions. Further, the mixed solution thus prepared may be appropriately aged.
[0022] 工程 (A ) で調製される混合液は、 表面の少なくとも一部にセラミックス またはその前駆体が固定化された基体および水を含むものである。 混合液は 必ずしもこれらのみで構成される必要はなく、 未反応のセラミックス原料、 セラミックス前駆体形成時に生じる副生成物、 アルコール類、 ケトン類、 ェ 一テル類、 またはエステル類に分類される有機溶媒のうち、 炭素数 1以上 4 以下で、 且つ水溶性の有機溶媒などを含んでも構わない。 [0022] The mixed solution prepared in the step (A) includes a substrate having ceramics or a precursor thereof immobilized on at least a part of the surface and water. The mixed liquid does not necessarily need to be composed only of these. Organic solvents classified as unreacted ceramic raw materials, by-products generated during the formation of the ceramic precursor, alcohols, ketones, ethers, or esters. Among them, it may contain a water-soluble organic solvent having 1 to 4 carbon atoms.
[0023] 工程 (A ) で調製される混合液の全重量に対する基体の濃度 (以下適宜、 スラリー濃度、 と表記する) は、 0 . 5重量%以上、 4 0重量%以下が好ま しく、 5重量%以上 2 0重量%以下がより好ましい。 スラリ一濃度が上記下 限値以上であると生産性が高くなり、 また上記上限値以下であると液の比重 が過大となることを防止でき、 経済性を担保できる。 [0023] The concentration of the substrate relative to the total weight of the mixed solution prepared in step (A) (hereinafter referred to as slurry concentration as appropriate) is preferably 0.5 wt% or more and 40 wt% or less. More preferably, it is more than 20% by weight. When the slurry concentration is equal to or higher than the lower limit, productivity is increased, and when the slurry concentration is lower than the upper limit, the specific gravity of the liquid can be prevented from becoming excessive, and economic efficiency can be ensured.
[0024] 工程 (A ) で得られる、 表面の少なくとも一部にセラミックスまたはその 前駆体が固定化された基体の、 混合液中での分散粒子径は、 特に制限は無い 力 平均粒子径が 1 0 m以下であることが好ましく、 平均粒子径が 3 m 以下であることがより好ましい。 この場合、 微粒子を凝集させて固形物を簡 便に回収できるという、 本発明の特徴を利かせる。 There is no particular limitation on the dispersed particle size of the substrate obtained by the step (A), on which the ceramic or its precursor is immobilized on at least a part of the surface. The force average particle size is 1 The average particle diameter is preferably 0 m or less, and more preferably 3 m or less. In this case, the solid matter is easily agglomerated by agglomerating the fine particles. The feature of the present invention is that it can be collected in the stool.
[0025] 工程 (A ) で調製される混合液中の水の濃度は、 混合液の全重量に対する 水の重量として示すと、 6 0重量%以上が好ましく、 8 0重量%以上がより 好ましい。 かかる範囲内において、 凝集剤が溶解しやすく、 凝集効果が発現 しゃすくなる。 混合液に含まれる水は、 最終的に前記の範囲にあればよい。 したがって、 含水溶媒として水のみを用いて、 混合完了時に前記範囲に入る 調製方法でも構わない。 また、 有機溶媒中で光半導体粒子を形成した後、 水 を加えて調整しても構わない。 さらに、 混合液が中和塩等の無機塩類を含む 場合には、 塩析作用により凝集剤が効果を示さないことがあるので、 水の濃 度が該範囲にある場合でも、 デカント等によって含水溶媒の置換を行っても よい。 [0025] The concentration of water in the mixed solution prepared in the step (A) is preferably 60% by weight or more, more preferably 80% by weight or more, when expressed as the weight of water with respect to the total weight of the mixed solution. Within such a range, the flocculant is easy to dissolve, and the agglomeration effect is manifested. The water contained in the mixed solution may be finally in the above range. Therefore, a preparation method that uses only water as the water-containing solvent and falls within the above range upon completion of mixing may be used. Moreover, after forming the optical semiconductor particles in an organic solvent, water may be added for adjustment. Furthermore, when the mixed solution contains inorganic salts such as neutralized salts, the flocculant may not be effective due to the salting-out action, so even if the water concentration is within this range, the water content can be increased by decanting. Substitution of the solvent may be performed.
[0026] 以下、 工程 (A ) における混合方法の具体例を示す。 [0026] Specific examples of the mixing method in the step (A) are shown below.
[0027] ( 1 . 基体、 セラミックスまたはその原料、 および含水溶媒を含む混合液 の P Hを 5以下に維持して混合する方法) [0027] (1. Method of mixing while maintaining pH of 5 or less in mixed liquid containing substrate, ceramic or raw material, and water-containing solvent)
工程 (A ) において、 例えば、 金属化合物光半導体である基体を含む含水 溶媒と珪酸塩、 珪酸塩を含む含水溶媒と前記基体、 および前記基体を含む含 水溶媒と珪酸塩を含む含水溶媒、 の少なくともいずれか一組を混合する。 そ して、 基体および珪酸塩の両方を含む混合液の p Hを 5以下に維持して混合 する。 In step (A), for example, a hydrous solvent and a silicate containing a substrate that is a metal compound optical semiconductor, a hydrous solvent containing a silicate and the substrate, and a hydrous solvent containing the substrate and a hydrous solvent containing a silicate, Mix at least one set. Then, the pH of the mixed solution containing both the substrate and silicate is maintained at 5 or less and mixed.
[0028] この例では、 セラミックスが酸化珪素を含む。 ここで、 p Hを 5以下に維 持する方法としては、 基体、 珪酸塩、 および含水溶媒の混合を行う際、 含水 溶媒の p Hを常時測定し、 適宜、 酸および塩基を加えて調整する方法でも構 わない。 しかし、 製造に用いる珪酸塩に含まれる塩基成分の総量を中和した 上で p H 5以下となるに十分な量の酸を予め含水溶媒中に存在させておく方 法が簡便である。 この場合に、 珪酸塩としては、 珪酸および/またはそのォ リゴマーの塩を用い、 2種以上を混合して用いてもよい。 ナトリウム塩およ びカリウム塩は、 工業的に入手容易である点から好ましく、 溶解工程を省略 できるので珪酸ナトリゥム水溶液 ( J I S K 1 4 0 8 "水ガラス") がさら に好ましい。 酸は、 どのような酸でも使用可能であるが、 塩酸, 硝酸、 また は硫酸といった鉱酸が好適に用いられる。 酸は、 1種単独または 2種以上を 混合して用いてもよい。 塩基は、 p H 5以下となるのに十分な量の酸を予め 含水溶媒中に存在させておく前述した方法を用いる場合には、 特に別途用い る必要は無い。 しかしながら、 塩基を用いる場合は、 どのような塩基でも使 用可能である。 なかでも、 水酸化力リゥム、 水酸化ナトリゥム等のアル力リ 金属水酸化物が好適に用いられる。 [0028] In this example, the ceramic contains silicon oxide. Here, as a method of maintaining the pH at 5 or less, when mixing the substrate, the silicate, and the hydrous solvent, the pH of the hydrous solvent is constantly measured, and adjusted by adding an acid and a base as appropriate. It doesn't matter how. However, it is easy to neutralize the total amount of the base components contained in the silicate used for the production, and to make a sufficient amount of acid present in the water-containing solvent beforehand so that the pH is 5 or less. In this case, as the silicate, a salt of silicic acid and / or its oligomer may be used, and two or more kinds may be mixed and used. Sodium and potassium salts are preferred because they are easily available industrially, and the dissolution step can be omitted, so that a sodium silicate aqueous solution (JISK 1408 "water glass") is further used. Is preferable. Any acid can be used, but a mineral acid such as hydrochloric acid, nitric acid, or sulfuric acid is preferably used. The acids may be used alone or in combination of two or more. The base does not need to be used separately when using the above-described method in which a sufficient amount of acid to have a pH of 5 or less is previously present in the aqueous solvent. However, when using a base, any base can be used. Of these, metal hydroxides such as hydroxy hydroxide and sodium hydroxide are preferably used.
[0029] (2. 含水溶媒にセラミックスまたはその原料を混合した後に、 基体を混 合する方法) [0029] (2. Method of mixing substrate after mixing ceramic or raw material in water-containing solvent)
工程 (A) において、 例えば、 擬似体液を用いてカルシウムおよびリンを 含む化合物を前記基体の表面に形成する。 具体的な混合手順は、 まず、 擬似 体液を調製し、 次いで基体を混合する。 この例では、 セラミックスとして、 カルシウムのリン酸塩の一種であるアパタイ トが形成される。 In step (A), for example, a compound containing calcium and phosphorus is formed on the surface of the substrate using a simulated body fluid. The specific mixing procedure is as follows. First, a simulated body fluid is prepared, and then the substrate is mixed. In this example, an apatite, which is a kind of calcium phosphate, is formed as ceramics.
[0030] ここで、 擬似体液とは、 塩化ナトリウム、 塩化カリウム、 炭酸水素ナトリ ゥム、 リン酸水素二カリウム、 塩化マグネシウム、 塩化カルシウム、 硫酸ナ トリウム、 あるいはフッ化ナトリウムなどを水に溶解して調製されるもので あり、 塩酸ゃァミン等の酸塩基によって p Hを 7〜8に調整された溶液を意 味する。 例えば、 擬似体液としては、 N a C I、 N a H C03、 N a 2H P04 、 N a H2 P04、 KC I、 KH C03、 K2H P04、 KH2P04、 M g C I 2 、 C a C I 2、 N a 2S04、 N a Fなどから選ばれる任意の化合物を水に溶解 させたものが挙げられる。 さらに、 カルシウムを含む化合物およびリンを含 む化合物を添加する。 また、 必要に応じて H C I、 (CH2OH) 3CN H2等 を添加することも可能である。 [0030] Here, the simulated body fluid refers to sodium chloride, potassium chloride, sodium bicarbonate, dipotassium hydrogen phosphate, magnesium chloride, calcium chloride, sodium sulfate, or sodium fluoride dissolved in water. This means a solution prepared by adjusting the pH to 7 to 8 with an acid base such as namine hydrochloride. For example, as the simulated body fluid, N a CI, N a H C0 3, N a 2 H P0 4, N a H 2 P0 4, KC I, KH C0 3, K 2 H P0 4, KH 2 P0 4, M g CI 2, C a CI 2 , N a 2 S0 4, that the N a F any compound selected from the like are dissolved in water. In addition, a compound containing calcium and a compound containing phosphorus are added. If necessary, HCI, (CH 2 OH) 3 CN H 2 or the like can be added.
[0031] 擬似体液の組成は、 N a+ : 1 20〜1 60mM、 K+: 1〜20mM、 C a 2+: 0. 5〜50mM、 H C03_ : 0. 5〜30mM、 H P 04 2_: "!〜 20mMが好ましい。 例えば、 ナトリウムイオン濃度 1 47mM、 カリウム イオン濃度 5mM、 カルシウムイオン濃度 7. 5mM、 炭酸水素イオン 4. 2mM、 並びにリン酸水素イオン 4. 2mMの組成で、 p H 7. 4の水溶液 が好適な擬似体液組成のうちの一つとして例示できる。 [0031] The composition of the simulated body fluid is as follows: N a +: 1 20 to 160 mM, K +: 1 to 20 mM, C a 2+ : 0.5 to 50 mM, H C0 3 _: 0.5 to 30 mM, HP 0 4 2 _: “! ~ 20 mM is preferred. For example, sodium ion concentration 1 47 mM, potassium ion concentration 5 mM, calcium ion concentration 7.5 mM, bicarbonate ion 4.2 mM, and hydrogen phosphate ion 4.2 mM, pH 7. Aqueous solution of 4 Can be exemplified as one of the preferred simulated body fluid compositions.
[0032] ( 3 . 含水溶媒に基体を混合した後に、 セラミックスまたはその原料を混合 する方法) [0032] (3. Method of mixing ceramic or its raw material after mixing substrate with hydrous solvent)
工程 (A ) において、 例えば、 前記基体とアルカリ金属塩とを含む含水溶 媒に、 マグネシウムとアルミニウムの水溶性の塩を 1種以上含む含水溶媒を 混合する。 具体的には、 例えば、 マグネシウムとアルミニウムの硝酸塩を水 溶性の塩として用い、 炭酸ナトリウムをアルカリ金属塩として用いる。 その 結果、 炭酸ナトリウムが沈殿剤となり、 マグネシウムとアルミニウムの水酸 化物が前記基体の表面に形成される。 In the step (A), for example, a water-containing solvent containing one or more water-soluble salts of magnesium and aluminum is mixed with a water-containing medium containing the base and an alkali metal salt. Specifically, for example, magnesium and aluminum nitrates are used as water-soluble salts, and sodium carbonate is used as an alkali metal salt. As a result, sodium carbonate becomes a precipitant, and magnesium and aluminum hydroxides are formed on the surface of the substrate.
[0033] 工程 (B ) は、 工程 (A ) で得られた混合液に凝集剤を添加し、 攪拌する 工程である。 この工程において、 表面の少なくとも一部にセラミックスまた はその前駆体が固定化された基体が、 凝集剤によって凝集される。 その結果 、 表面の少なくとも一部にセラミックスまたはその前駆体が固定化された前 記基体および凝集剤を含む固形物が形成される。 [0033] Step (B) is a step of adding an aggregating agent to the mixed liquid obtained in step (A) and stirring. In this step, the substrate on which the ceramic or its precursor is immobilized on at least a part of the surface is agglomerated by the aggregating agent. As a result, a solid body containing the substrate and the flocculant having ceramics or a precursor thereof immobilized on at least a part of the surface is formed.
[0034] 工程 (B ) で添加する凝集剤は、 好ましくは水溶性の凝集剤である。 水溶 性の凝集剤のうち、 高分子凝集剤が好ましい。 例えば、 アクリルアミ ド、 ァ クリル酸、 メタクリル酸等の重合体やこれら重合体の官能基の少なくとも一 部を変性したものが挙げられる。 高分子凝集剤であれば、 ァニオン型、 ノニ オン型、 カチオン型、 または両性型のいずれの型でも使用でき、 添加する混 合液の p Hによって任意の型の高分子凝集剤を選択できる。 ノニオン型以外 の高分子凝集剤は、 イオン対の部分構造を含むので、 工程 (D ) で凝集剤を 分解した際に無機塩が残留し、 更に洗浄が必要となる場合がある。 イオン対 としては、 ナトリウム塩、 カリウム塩、 塩化物、 硫酸塩等が挙げられる。 こ の点から、 ノニオン型の高分子凝集剤が好ましい。 しかしながら、 光半導体 粒子に対する凝集剤の使用量から明白なように、 残留する無機塩の量は多く ないので、 ノニオン型以外の型も利用可能である。 [0034] The flocculant added in the step (B) is preferably a water-soluble flocculant. Of the water-soluble flocculants, polymer flocculants are preferred. Examples thereof include polymers such as acrylamide, acrylic acid, and methacrylic acid, and those obtained by modifying at least a part of functional groups of these polymers. As long as it is a polymer flocculant, any type of anionic, nonionic, cationic, or amphoteric can be used, and any type of polymer flocculant can be selected depending on the pH of the added mixture. Since polymer flocculants other than the nonionic type contain a partial structure of ion pairs, an inorganic salt may remain when the flocculant is decomposed in step (D), and further washing may be required. Examples of ion pairs include sodium salts, potassium salts, chlorides, sulfates, and the like. From this point, a nonionic polymer flocculant is preferable. However, as is apparent from the amount of the flocculant used for the optical semiconductor particles, the amount of the remaining inorganic salt is not large, so that types other than the nonionic type can be used.
[0035] 高分子凝集剤としては、 ァクリルアミ ドまたはその変性体からなる群から 選ばれる 1種以上の化合物を、 繰り返し単位として含むポリアクリルアミ ド 系凝集剤が例示できる。 具体的には、 アクリルアミ ドの重合体であるポリア クリルアミ ド、 変性アクリルアミ ドポリマ一、 アクリル酸とアクリルアミ ド の共重合体、 アクリルアミ ドと変性アクリルアミ ドの共重合体、 アクリルァ ミ ドとジメチルアミノエチルメタクリレートとの共重合体などがある。 凝集 剤のァニオン性、 あるいはカチオン性は、 ポリマ一を後加工して、 あるいは 、 共重合の比率で調整される。 ここで変性アクリルアミ ドとは、 アクリルァ ミ ドのアミ ド基部分の水素原子を、 アルキル基、 またはアンモニゥム塩/ス ルホン酸基/水酸基といつた親水性置換基を持つたアルキル基で置換したも のが例示される。 凝集剤の形態には、 特に制限はなく、 粉末、 分散液、 ベー スト、 ェマルジヨン、 または溶液のいずれのものも利用できる。 添加方法に も特に制限はなく、 公知の添加方法であればいずれの方法で行ってもよい。 [0035] As the polymer flocculant, a polyacrylamide containing one or more compounds selected from the group consisting of acrylamide or a modified product thereof as a repeating unit. A system flocculant can be illustrated. Specifically, polyacrylamide, which is a polymer of acrylic amide, modified acrylic amide polymer, copolymer of acrylic acid and acrylic amide, copolymer of acrylic amide and modified acrylic amide, acrylic amide And a copolymer of dimethylaminoethyl methacrylate. The anionicity or cationicity of the flocculant is adjusted by post-processing the polymer or by the copolymerization ratio. Here, the modified acrylic amide is the substitution of the hydrogen atom of the amide group of the acrylamide with an alkyl group or an alkyl group having an ammonium salt / sulfonic acid group / hydroxyl group and a hydrophilic substituent. Examples are given. The form of the flocculant is not particularly limited, and any of powder, dispersion, base, emulsion or solution can be used. The addition method is not particularly limited, and any known addition method may be used.
[0036] 凝集剤の使用量は、 前記混合液に含まれる前記基体の重量に対して、 0 . [0036] The amount of the flocculant used is set to 0. 0 based on the weight of the substrate contained in the mixed solution.
1重量%以上、 3重量%以下が好ましく、 0 . 5重量%以上 1 . 5重量%以 下がより好ましい。 上記下限値以上であると、 十分な凝集効果が得られ、 上 記上限値以下であると、 工程 (D ) での分解に時間がかかりすぎることが防 止され、 生産性が良好なものとなる。 また、 固液分離を簡便にする効果が得 られる。 It is preferably 1% by weight or more and 3% by weight or less, more preferably 0.5% by weight or more and 1.5% by weight or less. When the amount is not less than the above lower limit value, a sufficient agglomeration effect can be obtained, and when the amount is not more than the above upper limit value, the decomposition in the step (D) can be prevented from taking too much time and the productivity can be improved. Become. In addition, the effect of simplifying solid-liquid separation can be obtained.
[0037] 工程 (C ) において、 工程 (B ) で得られた固形物を含水溶媒から回収す る。 混合液から回収する固形物は、 表面の少なくとも一部にセラミックスま たはその前駆体が固定化された基体および凝集剤を含むものである。 回収し た際に、 これらのみで構成されている必要はなく、 含水溶媒や中和塩などを 含んでいてもよい。 また、 固形物の回収方法は、 特に限定されない。 含水溶 媒の自重による自然濾過、 減圧濾過、 加圧濾過、 遠心分離、 デカンテーショ ン等、 固体と液体を分離する公知の方法を適用してよい。 [0037] In the step (C), the solid obtained in the step (B) is recovered from the water-containing solvent. The solid matter recovered from the mixed solution contains a substrate having a ceramic or its precursor immobilized on at least a part of the surface and a flocculant. When recovered, it need not be composed of only these, and may contain a hydrous solvent, a neutralized salt, or the like. Further, the method for recovering the solid matter is not particularly limited. Known methods for separating solids and liquids may be applied, such as natural filtration by the weight of the water-containing medium, vacuum filtration, pressure filtration, centrifugation, and decantation.
[0038] 工程 (D ) において、 工程 (C ) で回収された固形物を加熱し、 凝集剤を 分解する。 分解した凝集剤を除去することにより、 本発明の光半導体粒子が 得られる。 ここで、 凝集剤は完全に除去される場合に限定されず、 少量の無 機物等が残留していてもよい。 凝集剤を分解するには、 凝集剤の分解が起こ る温度域で固形物を加熱する。 低温では、 凝集剤の分解が不十分になりやす く、 また、 分解に長時間を要してしまう。 高温であると、 凝集剤の分解は速 くなるが、 光半導体粒子が加熱によって劣化しやすい。 そのため、 加熱温度 は、 4 0 0 °C以上 1 0 0 0 °C以下が好ましく、 5 0 0 °C以上 8 0 0 °C以下が より好ましい。 加熱時間は、 加熱温度によって選択する必要があり、 特に限 定されない。 例えば、 低温処理では長時間を要し、 高温処理では短時間で十 分となる。 6 0 0 °C加熱の場合、 0 . 5時間以上、 2 4時間以下が好ましく 、 1時間以上、 1 8時間以下がより好ましい。 また、 凝集剤の分解が酸化分 解反応に基づく場合は、 雰囲気中の酸素分圧を高めることで、 分解を促進す ることもできる。 [0038] In the step (D), the solid collected in the step (C) is heated to decompose the flocculant. By removing the decomposed flocculant, the optical semiconductor particles of the present invention can be obtained. Here, the flocculant is not limited to the case where it is completely removed, and a small amount of insoluble matter may remain. To decompose the flocculant, the flocculant must be decomposed. The solid is heated in the temperature range. At low temperatures, the flocculant tends to be insufficiently decomposed, and it takes a long time to decompose. When the temperature is high, the flocculant decomposes quickly, but the photo-semiconductor particles are easily deteriorated by heating. Therefore, the heating temperature is preferably 400 ° C. or more and 100 ° C. or less, and more preferably 500 ° C. or more and 80 ° C. or less. The heating time must be selected depending on the heating temperature, and is not particularly limited. For example, low temperature processing takes a long time, and high temperature processing takes a short time. In the case of heating at 600 ° C., 0.5 hours or more and 24 hours or less are preferable, and 1 hour or more and 18 hours or less are more preferable. Moreover, when the decomposition of the flocculant is based on an oxidative decomposition reaction, the decomposition can be promoted by increasing the oxygen partial pressure in the atmosphere.
[0039] 工程 (A ) において、 不揮発性の化合物が副生され、 または不揮発性の化 合物を使用する場合、 不純物として該不揮発性の化合物が光半導体粒子に取 り込まれることがある。 この場合には、 光半導体粒子の洗浄が必要となる。 不揮発性の化合物が副生される方法には、 例えば、 硝酸酸性の水溶液に基体 粒子を分散し、 これにケィ酸ナトリウムを混合して、 該基体の表面に珪素化 合物が固定化された基体粒子を含む混合液を調製する方法がある。 この方法 においては、 中和塩として生じた硝酸ナトリウムが、 不揮発性の化合物に該 当する。 また、 不揮発性の化合物を使用する方法には、 セラミックスの原料 として塩化カルシウムとリン酸塩を溶解した液に、 基体粒子を混合して、 表 面にカルシウムとリンとの化合物が固定化された該基体粒子を含む混合液を 調製する方法がある。 この方法においては、 使用した塩化カルシウムやリン 酸塩の残存分が不揮発性の化合物に該当する。 [0039] In the step (A), when a non-volatile compound is by-produced or a non-volatile compound is used, the non-volatile compound may be incorporated into the optical semiconductor particles as an impurity. In this case, it is necessary to clean the optical semiconductor particles. As a method for producing a non-volatile compound as a by-product, for example, base particles are dispersed in a nitric acid aqueous solution, and sodium silicate is mixed therewith, and the silicon compound is immobilized on the surface of the base. There is a method for preparing a mixed solution containing base particles. In this method, sodium nitrate formed as a neutralized salt corresponds to a nonvolatile compound. In addition, in the method using a non-volatile compound, a base particle is mixed in a solution in which calcium chloride and phosphate are dissolved as a ceramic raw material, and the compound of calcium and phosphorus is immobilized on the surface. There is a method for preparing a mixed solution containing the substrate particles. In this method, the remaining amount of calcium chloride and phosphate used corresponds to the nonvolatile compound.
[0040] また、 工程 (A ) 力 硫酸を使用する方法、 硫酸塩を副生または使用する 方法、 または、 基体、 セラミックスまたはセラミックスの原料として、 硫黄 化合物を含有するものを使用する方法であると、 光半導体粒子中に s o 4分、 S O 3分等の硫黄化合物が取り込まれ、 着色することがある。 この着色を抑制 または回避するために、 硫黄化合物の含有量を、 前記光半導体粒子中に含ま れる金属化合物光半導体の重量に対する S原子の重量として、 0 . 5重量% 以下、 好ましくは 0 . 3重量%以下、 より好ましくは 0 . 2重量%以下とす ることが望ましい。 したがって、 硫黄化合物の含有量を調整するために、 洗 浄が必要となる場合もある。 [0040] In addition, the step (A) is a method using a strong sulfuric acid, a method using a sulfate as a by-product or using, or a method using a substrate, ceramics or a raw material of ceramics containing a sulfur compound. Sulfur compounds such as so 4 minutes and SO 3 minutes may be incorporated into the optical semiconductor particles and colored. In order to suppress or avoid this coloring, the content of the sulfur compound is 0.5% by weight as the weight of S atoms with respect to the weight of the metal compound photo semiconductor contained in the photo semiconductor particles. In the following, it is desirable that the content be 0.3% by weight or less, more preferably 0.2% by weight or less. Therefore, cleaning may be necessary to adjust the sulfur compound content.
[0041 ] 洗浄が必要とされる場合、 工程 (A ) で混合液を調製する際に、 前記混合 液からイオン交換樹脂を用いて不純物を除去する方法、 または工程 (C ) で 固形物を回収する際に、 フィルタ一上に形成された固形物からなるケーキに 含水溶媒を通液する方法、 などによって洗浄を実施してもよいし、 工程 (A ) 〜 (D ) とは独立して、 表面の少なくとも一部にセラミックスまたはその 前駆体が固定化された前記基体、 および凝集剤を含む固形物を洗浄する工程 ( E ) を含む製造方法として実施してもよい。 工程 (E ) は、 工程 (B ) と[0041] When cleaning is required, when preparing the mixed solution in step (A), a method of removing impurities from the mixed solution using an ion exchange resin, or collecting solids in step (C) In this case, washing may be carried out by a method of passing a water-containing solvent through a cake made of solid matter formed on a filter, etc., independently of steps (A) to (D), You may implement as a manufacturing method including the process (E) which wash | cleans the solid substance containing the said base | substrate with which ceramics or its precursor was fix | immobilized to at least one part of the surface, and a flocculant. Process (E) includes process (B) and
( C ) の間、 あるいは工程 (C ) と (D ) の間の、 いずれかであれば、 表面 の少なくとも一部にセラミックスまたはその前駆体が固定化された前記基体 、 および凝集剤を含む固形物と、 洗浄に用いる溶媒 (以下適宜 「洗浄溶媒」 と表記) とを容易に分離でき、 簡便に洗浄できるので好ましい。 洗浄は工程(C), or between steps (C) and (D), if the ceramic substrate or its precursor is immobilized on at least a part of the surface, and a solid containing a flocculant And the solvent used for washing (hereinafter referred to as “washing solvent” where appropriate) can be easily separated, and can be easily washed. Cleaning is a process
( D ) の後に実施することも可能であるが、 付随して、 固形物を洗浄溶媒か ら回収する工程や固形物を乾燥する工程が必要となる。 Although it can be carried out after (D), a process for recovering the solid from the washing solvent and a process for drying the solid are necessary.
[0042] 洗浄溶媒は、 主として水からなる含水溶媒である。 洗浄溶媒は、 アルコ一 ル類、 ケトン類、 エーテル類、 あるいはエステル類に分類される有機溶媒の うち、 炭素数 1以上 4以下で、 且つ水溶性の有機溶媒を含んでも構わない。 工程 (A ) が無機塩などの水溶性不純物を副生する工程である場合、 洗浄に 用いる溶媒は水が好ましい。 [0042] The cleaning solvent is a hydrous solvent mainly composed of water. The washing solvent may include a water-soluble organic solvent having 1 to 4 carbon atoms among organic solvents classified into alcohols, ketones, ethers, or esters. When the step (A) is a step of by-producing water-soluble impurities such as inorganic salts, the solvent used for washing is preferably water.
[0043] また、 前記洗浄溶媒は、 不純物の除去効率を強化するために、 酸性や塩基 性に調整してもよい。 例えば、 鉱酸ゃ有機酸で酸性に調整すれば、 アルカリ 金属やアル力リ土類金属等、 陽イオン性の不純物の除去効率を強化できる。 また、 アルカリ金属水酸化物やアンモニアで塩基性に調整すれば、 硝酸ィォ ン、 塩素イオン、 硫酸イオン等、 陰イオン性の不純物の除去効率を強化でき る。 そして、 洗浄溶媒の酸塩基性は、 どちらか一方のみを実施してもよいし 、 酸性と塩基性とを併用することもできる。 酸性と塩基性との間でスイング させて洗浄する場合もある。 酸性または塩基性に調整した洗浄溶媒の p Hは 、 凝集剤が分解せず、 且つ凝集効果を示す範囲であればいずれの p Hに調整 しても構わない。 ノニオン型のポリアクリルアミ ド系高分子凝集剤を用いる 場合は、 p H 1〜 1 0の範囲が好ましく、 p H 2〜8がより好ましい。 [0043] The cleaning solvent may be adjusted to be acidic or basic in order to enhance the removal efficiency of impurities. For example, by adjusting the acidity with mineral acid or organic acid, the removal efficiency of cationic impurities such as alkali metals and alkaline earth metals can be enhanced. In addition, by adjusting the basicity with alkali metal hydroxide or ammonia, the removal efficiency of anionic impurities such as nitrate, chloride and sulfate can be enhanced. And the acid basicity of the washing solvent may be carried out only in either one, or the acidic and basic can be used in combination. Swing between acidic and basic In some cases, it may be washed. The pH of the washing solvent adjusted to be acidic or basic may be adjusted to any pH as long as the flocculant is not decomposed and exhibits a coagulation effect. When a nonionic polyacrylamide polymer flocculant is used, the pH is preferably in the range of 1 to 10, more preferably pH 2 to 8.
[0044] 洗浄方法は、 デカントと前記洗浄溶媒の添加を 1回以上交互に繰り返す方 法、 イオン交換による脱塩処理、 洗浄溶媒への再分散とろ過を 1回以上交互 に繰り返す洗浄方法等、 公知の洗浄方法のいずかで実施できる。 逐次連続的 に溶媒置換する流通洗浄が好ましく、 上昇流によるものがさらに好ましい。 凝集剤の効果によって粗大粒子を形成した前記固形物 (以下、 適宜 「フロッ ク」 と表記) が大きな沈降速度を示す点を利用できるからである。 このよう な装置としては、 筒状の容器に光半導体粒子や不純物を含む混合液を入れ、 水を容器の底部から供給し、 上部から排出する装置が例示できる。 シックナ —やスクリューデカンター等の公知装置も利用可能である。 また、 洗浄の途 中でフロックが壊れて微細粒子が生じた場合に、 フロックを再形成させるた めに、 凝集剤を追加しても構わない。 [0044] The washing method includes, for example, a method in which the addition of the decant and the washing solvent is alternately repeated one or more times, a desalting treatment by ion exchange, a washing method in which redispersion in the washing solvent and filtration are alternately repeated one or more times, etc. It can be carried out by any known cleaning method. A flow cleaning in which the solvent is successively replaced is preferable, and an upward flow is more preferable. This is because it is possible to utilize the point that the solid matter (hereinafter referred to as “floc” where appropriate) in which coarse particles are formed by the effect of the flocculant exhibits a large sedimentation rate. Examples of such an apparatus include an apparatus in which a liquid mixture containing optical semiconductor particles and impurities is placed in a cylindrical container, water is supplied from the bottom of the container, and discharged from the top. Known devices such as thickeners and screw decanters can also be used. In addition, a flocculant may be added to re-form the flocs when the flocs break and fine particles are produced during the cleaning process.
[0045] 含水溶媒の上昇流による洗浄を実施する際には、 前記フロックと洗浄溶媒 とを分離する部分の液体の流速を、 水平方向のべク トル成分と垂直方向のベ ク トル成分に分けた場合に、 液体の垂直方向のべク トル成分としての上昇速 度が、 該フ口ックの沈降速度以下になるように洗浄溶媒の供給量を制御して 洗浄する。 前記上昇速度がフロックの沈降速度を越えていると、 洗浄装置か ら、 該フロックが流出してしまい洗浄することが出来ない。 上昇速度を 0に すること、 すなわち、 水平方向にのみ洗浄溶媒を流して層流で洗浄すること も、 可能ではあるが、 装置が複雑になる。 したがって、 前記上昇速度は、 0 よりも大きく、 前記フロックの沈降速度以下であることが好ましい。 [0045] When washing with an upward flow of a hydrous solvent, the flow rate of the liquid in the portion separating the floc and the washing solvent is divided into a horizontal vector component and a vertical vector component. In this case, cleaning is performed by controlling the supply amount of the cleaning solvent so that the rising speed of the liquid as a vector component in the vertical direction is lower than the sedimentation speed of the hook. If the ascending speed exceeds the settling speed of the floc, the floc flows out from the washing device and cannot be washed. Although it is possible to set the ascending speed to 0, that is, to wash in a laminar flow with a washing solvent flowing only in the horizontal direction, the apparatus becomes complicated. Therefore, it is preferable that the rising speed is greater than 0 and equal to or less than the sedimentation speed of the floc.
[0046] フロックの沈殿の下部から洗浄溶媒を供給すると、 沈殿層が洗浄溶媒に対 する抵抗となるので、 上昇流速にバラツキが生じて、 局部的な上昇流が生じ ることがある。 また、 設備配管や送液ポンプ中などで発生した気泡が洗浄容 器に供給され、 気泡に同伴して、 フロックが洗浄容器内を上昇することもあ る。 そのため、 上昇流速が該フロックの沈降速度に近似していると、 洗浄は 短時間で処理できるが、 前記固形物の損失量が多くなる。 一方、 上昇流速が 過小であると、 フロックの損失は抑制できるが、 洗浄に時間を要する。 そこ で、 前記上昇流速は、 フロックの沈降速度を基準 (1 00%) として、 1 % 以上、 50%以下が好ましく、 3%以上 25%以下がより好ましい。 [0046] When the washing solvent is supplied from the lower part of the floc precipitation, the precipitation layer becomes a resistance to the washing solvent, so that the ascending flow rate varies, and a local upflow may occur. In addition, bubbles generated in equipment piping and liquid pumps are supplied to the cleaning container, and the flock may rise in the cleaning container with the bubbles. The Therefore, if the rising flow rate approximates the floc sedimentation rate, washing can be processed in a short time, but the loss of the solid matter increases. On the other hand, if the ascending flow rate is too small, the loss of flocs can be suppressed, but it takes time for cleaning. Therefore, the rising flow rate is preferably 1% or more and 50% or less, and more preferably 3% or more and 25% or less, based on the floc sedimentation rate (100%).
[0047] 本発明で製造される光半導体粒子が、 S iの酸化物を表面に固定化した光 触媒である場合、 以下のものが例示できる。 [0047] When the photo-semiconductor particles produced in the present invention are photocatalysts having an Si oxide immobilized on the surface, the following can be exemplified.
(a) 金属化合物光半導体である基体と、 該基体を被覆する実質的に細孔を 有しない酸化珪素膜と、 を含有する光触媒、 (a) a photocatalyst comprising: a substrate that is a metal compound photo-semiconductor; a silicon oxide film that substantially does not have pores covering the substrate;
(b) 前記酸化珪素膜が焼成された膜である前記 (a) の光触媒、 (b) The photocatalyst of (a), wherein the silicon oxide film is a fired film,
( c) 前記酸化珪素膜が、 400°C以上、 1 000°C以下の温度で焼成して 得られる焼成膜である、 前記 (b) に記載の光触媒、 (c) The photocatalyst according to (b), wherein the silicon oxide film is a fired film obtained by firing at a temperature of 400 ° C or higher and 1000 ° C or lower.
(d) アルカリ金属含有量が 1 p pm以上、 1 000 p pm以下である前記 (a) から (c) の光触媒、 (d) the photocatalyst according to (a) to (c), wherein the alkali metal content is 1 ppm or more and 1 000 ppm or less,
(e) 窒素吸着法による 20〜 500オングストロームの領域の細孔径分布 測定において、 酸化珪素膜由来の細孔がない前記 (a) から (d) の光触媒 (e) In the measurement of the pore size distribution in the region of 20 to 500 angstroms by the nitrogen adsorption method, the photocatalyst according to (a) to (d) above, which has no pores derived from a silicon oxide film
( f ) 表面積 1 m2当りの珪素担持量が 0. 1 0mg以上、 2. 0 m g以下で ある前記 (a) から (e) の光触媒、 (f) The photocatalyst according to any one of (a) to (e) above, wherein the supported amount of silicon per 1 m 2 of surface area is from 0.1 mg to 2.0 mg.
(g) 酸化珪素にアルカリ金属が含まれている前記 (a) から ( f ) の光触 媒、 (g) The photocatalyst of (a) to (f) above, wherein silicon oxide contains an alkali metal,
(h) 硫黄化合物の含有量が、 前記光触媒の重量に対する硫黄原子として 0 . 5重量%以下である前記 (a) から (g) の光触媒。 (h) The photocatalyst according to any one of (a) to (g), wherein the content of the sulfur compound is 0.5% by weight or less as a sulfur atom with respect to the weight of the photocatalyst.
[0048] 前記 (a) から (h) の光触媒についてより詳細に述べる。 例えば、 以下 のものが光触媒活性に優れる。 [0048] The photocatalysts (a) to (h) will be described in more detail. For example, the following are excellent in photocatalytic activity.
アルカリ金属としては、 リチウム、 ナトリウム、 カリウム、 ルビジウム、 セシウム、 フランシウムが挙げられる。 これらのアルカリ金属は 1種のみを 含んでもよいし、 2種以上含んでいても構わない。 アル力リ金属含有量は、 好ましくは 1 0 p pm〜 1 000 p pmであり、 より好ましくは 1 0 p pm〜500 p pmである。 アルカリ金属含有量が上 記範囲内であると、 耐熱性にも優れるので好ましい。 アルカリ金属が含まれ ていると、 光触媒活性が向上された酸化珪素膜が形成される。 Examples of the alkali metal include lithium, sodium, potassium, rubidium, cesium, and francium. These alkali metals may contain only 1 type, and may contain 2 or more types. The metal strength metal content is preferably from 10 ppm to 1 000 ppm, more preferably from 10 ppm to 500 ppm. When the alkali metal content is within the above range, heat resistance is excellent, which is preferable. When an alkali metal is contained, a silicon oxide film with improved photocatalytic activity is formed.
[0049] 珪素担持量は、 S iの酸化物を表面に固定化した光触媒が含有する珪素量 と、 表面積から算出される計算値である。 珪素担持量は、 好ましくは 0. 1 2mg以上、 1. 5m g以下、 より好ましくは 0. 1 6mg以上、 1. Omg 以下である。 珪素担持量が上記下限値以上であると、 酸化珪素膜による活性 効果が向上し、 十分な光分解活性の効果が得られる。 また、 珪素担持量が上 記上限値以下であると、 光触媒に占める十分な基体の割合を確保でき、 良好 な光分解活性が得られる。 [0049] The amount of silicon supported is a calculated value calculated from the amount of silicon contained in the photocatalyst having the Si oxide immobilized on the surface and the surface area. The silicon loading is preferably 0.1 2 mg or more and 1.5 mg or less, more preferably 0.1 6 mg or more and 1. Omg or less. When the amount of silicon supported is not less than the above lower limit, the activation effect by the silicon oxide film is improved, and a sufficient photolytic activity effect is obtained. Further, when the amount of silicon supported is not more than the above upper limit value, a sufficient proportion of the substrate in the photocatalyst can be secured, and good photolytic activity can be obtained.
[0050] 硫黄化合物は、 光触媒の着色防止の観点から、 好ましくは 0. 5重量%以 下、 より好ましくは 0. 3重量%以下であり、 最も好ましいのは、 含まれな いことである。 [0050] From the viewpoint of preventing coloring of the photocatalyst, the sulfur compound is preferably 0.5% by weight or less, more preferably 0.3% by weight or less, and most preferably it is not contained.
[0051] 本発明において、 酸化珪素膜由来の細孔の有無は、 前記セラミックスが酸 化珪素である前記光半導体粒子の細孔容積分布と、 光半導体粒子の基体に該 当する粒子の細孔容積分布とを比較して判定できるものである。 具体的には 以下の (1 ) 〜 (4) の手法によって判定することができる。 [0051] In the present invention, the presence or absence of pores derived from the silicon oxide film is determined by determining the pore volume distribution of the photo-semiconductor particles in which the ceramic is silicon oxide and the pores of the particles corresponding to the substrate of the photo-semiconductor particles. It can be determined by comparing the volume distribution. Specifically, it can be determined by the following methods (1) to (4).
( 1 ) 基体の脱着過程での窒素吸着等温線を測定する、 (1) Measure the nitrogen adsorption isotherm during the substrate desorption process,
( 2 ) 光半導体粒子の脱着過程での窒素吸着等温線を測定する (2) Measure the nitrogen adsorption isotherm during the desorption process of optical semiconductor particles
(3) B J H (B a r r e t t— J o y n e r— H a l e n d a) 法で、 前 記二つの窒素吸着等温線を解析して、 20〜500オングストロームの領域 の I o g微分細孔容積分布曲線を求める。 (3) Analyze the two nitrogen adsorption isotherms by the BJH (Ba r rt t — J o y ner — H a len d a) method to obtain an I o g differential pore volume distribution curve in the region of 20 to 500 angstroms.
(4) 二つの I o g微分細孔容積分布曲線を比較し、 光半導体粒子の I o g 微分細孔容積が、 基体の I o g微分細孔容積よりも 0. 1 m I / g以上大き い領域が存在しない場合には、 光半導体粒子に細孔が実質的にないと判定し 、 0. 1 m l /g以上大きい領域が存在する場合には、 酸化珪素膜に細孔が 有ると判定する。 なお、 0 . 1 m l / g以上とするのは、 窒素吸着法による細孔分布測定で は、 l o g微分細孔容積値で約 0 . 1 m I / g幅の測定誤差が生じることが 多いためである。 (4) Comparing two I og differential pore volume distribution curves, the region where the I og differential pore volume of the photo-semiconductor particle is 0.1 m I / g or more larger than the I og differential pore volume of the substrate In the absence of pores, it is determined that the optical semiconductor particles are substantially free of pores, and in the case where a region larger than 0.1 ml / g exists, it is determined that the silicon oxide film has pores. Note that 0.1 ml / g or more is often caused by a measurement error of approximately 0.1 m I / g width in the log differential pore volume value in the pore distribution measurement by the nitrogen adsorption method. It is.
[0052] ここで、 酸化珪素膜に細孔が存在する場合、 光分解活性が向上し難い場合 がある。 この理由は必ずしも明らかではないが、 以下のような理由が推察さ れる。 すなわち、 細孔の存在によって酸化珪素膜での光の散乱や反射が起こ りやすくなり、 光触媒活性を有する基体に到達する紫外線の光量が減少し、 光触媒励起による正孔と電子の生成量が減少することによるものと推察され る。 また、 同じ酸化珪素量で被覆した場合、 細孔有りのものは、 細孔無しの ものに比べ、 細孔の容積の分だけ酸化珪素膜の厚さが増す結果、 光触媒活性 を有する基体と分解対象物である有機物との物理的距離が大きくなるため、 充分な光分解活性が得られないものと推察される。 Here, when the silicon oxide film has pores, the photolytic activity may be difficult to improve. The reason for this is not always clear, but the following reasons are presumed. In other words, light scattering and reflection on the silicon oxide film is likely to occur due to the presence of the pores, the amount of ultraviolet light reaching the substrate having photocatalytic activity is reduced, and the amount of holes and electrons generated by photocatalytic excitation is reduced. This is presumed to be due to Also, when coated with the same amount of silicon oxide, the one with pores is decomposed from the substrate with photocatalytic activity as a result of the thickness of the silicon oxide film being increased by the volume of the pores compared to the one without pores. It is presumed that sufficient photodegradation activity cannot be obtained because the physical distance from the target organic substance becomes large.
[0053] 本発明で製造されるカルシウム化合物を表面に固定化した光触媒としては 、 以下のものが例示できる。 なお、 ここでは基体が、 光触媒活性に優れる 「 酸化チタン粒子」 であるものを記載している。 [0053] Examples of the photocatalyst in which the calcium compound produced in the present invention is immobilized on the surface include the following. Here, the substrate is described as “titanium oxide particles” having excellent photocatalytic activity.
( χ ) 表面の少なくとも一部に、 カルシウムのリン酸塩が固定化された酸化 チタン粒子、 (χ) Titanium oxide particles with calcium phosphate immobilized on at least a part of the surface,
( y ) 表面の少なくとも一部に、 カルシウムのリン酸塩からなるアパタイ ト が固定化され (y) The calcium phosphate apatite is immobilized on at least a part of the surface.
た酸化チタン粒子、 Titanium oxide particles,
( z ) 表面の少なくとも一部に、 カルシウムの珪酸塩が固定化された酸化チ タン粒子。 (z) Titanium oxide particles in which calcium silicate is immobilized on at least a part of the surface.
[0054] 前記 (X ) 〜 (z ) の光触媒は、 カルシウム化合物が、 吸着剤、 あるいは スぺーサ一として効果を示す。 かかる光触媒は、 吸着性能に優れ、 および有 機基材 (樹脂等の有機物からなる基材) の劣化が起こりにくい。 ここで 「有 機基材の劣化」 とは、 有機基材に光触媒を添加した場合に、 光触媒の分解力 によって有機基材が分解されて劣化することである。 これら (X ) 〜 (z ) の光触媒における、 カルシウム化合物のスぺーサー効果とは、 酸化チタン粒 子が直接有機基材に接触しない構造によるものである。 したがって、 光触媒 の分解力を保ちつつ、 有機基材が分解されにくい。 [0054] In the photocatalysts (X) to (z), the calcium compound is effective as an adsorbent or a spacer. Such a photocatalyst has excellent adsorption performance, and deterioration of an organic base material (a base material made of an organic substance such as a resin) hardly occurs. Here, “deterioration of the organic base material” means that when the photocatalyst is added to the organic base material, the organic base material is decomposed and deteriorated by the decomposing power of the photocatalyst. The spacer effect of calcium compounds in the photocatalysts of (X) to (z) This is due to the structure in which the child does not directly contact the organic substrate. Therefore, it is difficult for the organic base material to be decomposed while maintaining the decomposing power of the photocatalyst.
[0055] カルシウム化合物の量は、 光触媒全体の重量に対する C aイオンの重量% として、 0 . 5重量%以上、 2 0重量%以下が好ましく、 3重量%以上、 1 0重量%以下がより好ましい。 カルシウム化合物の量が上記下限値以上であ ると、 吸着剤あるいはスぺ一サ一としての効果が十分に得られる。 カルシゥ ム化合物の量が上記上限値以下であると、 光触媒活性を保ちつつ、 有機基材 劣化抑制の効果を得ることができる。 [0055] The amount of the calcium compound is preferably 0.5% by weight or more and 20% by weight or less, more preferably 3% by weight or more and 10% by weight or less, as the weight percent of Ca ions with respect to the total weight of the photocatalyst. . When the amount of the calcium compound is not less than the above lower limit, the effect as an adsorbent or a spacer can be sufficiently obtained. When the amount of the calcium compound is not more than the above upper limit value, the effect of suppressing deterioration of the organic base material can be obtained while maintaining the photocatalytic activity.
[0056] また、 カルシウム化合物は、 単一組成のものである場合もあるが、 2種以 上の化合物の混合物や複合物となる場合もある。 例えば、 リン酸カルシウム を固定化する際に、 空気中の二酸化炭素に基づいて生じた炭酸カルシウムが 併せて固定化されることがあり、 または他の無機イオンが含まれることもあ る。 [0056] The calcium compound may be of a single composition, but may be a mixture or composite of two or more compounds. For example, when calcium phosphate is immobilized, calcium carbonate generated based on carbon dioxide in the air may be immobilized together, or other inorganic ions may be included.
[0057] 本発明で得られる光半導体粒子は凝集剤を用いないで製造されたものと比 較して、 性能に差異はない。 したがって、 特に用途の制限は無い。 例えば、 化粧料、 日焼け止めクリーム、 合成樹脂、 塗料などの用途に、 公知のものと 同様に、 紫外線遮蔽剤あるいは顔料として配合できる。 また、 光触媒として 、 大気浄化、 水質浄化、 消臭、 抗菌、 防汚、 セルフクリーニング等の機能を 示す光触媒含有体に用いることが可能である。 [0057] The optical semiconductor particles obtained in the present invention have no difference in performance as compared with those produced without using a flocculant. Therefore, there are no particular restrictions on applications. For example, it can be blended as a UV shielding agent or pigment in the same manner as known ones in applications such as cosmetics, sunscreen creams, synthetic resins and paints. Further, as a photocatalyst, it can be used for a photocatalyst-containing body exhibiting functions such as air purification, water purification, deodorization, antibacterial, antifouling, and self-cleaning.
[0058] 光触媒含有体には、 光触媒を含有する無機成形体、 光触媒を含有する有機 樹脂成形体、 光触媒塗膜を有する材料 (基材は無機化合物または有機化合物 のいずれでもよい) 、 光触媒塗料、 光触媒噴霧液等があり、 次のような応用 分野や応用製品に用いることができる :セラミックスフィルタ一およびガラ スフィルター (エアコン、 冷蔵庫、 加湿器、 除湿器、 調理器、 空気清浄機、 集塵装置、 廃水処理装置等に用いる) 、 舗装用アスファルト, R C建造物の 構造材料、 珪藻土またはゼォライ トといった調湿建材、 天井材 (ロックウー ル系、 石膏系など) 、 タイル目地材、 コンクリートブロックやインタ一ロッ キングブロック、 タイル (外装用、 舗装用、 床材用など) 、 瓦、 サイジング 材、 シーリング材、 建築用ゴム等の左官材料、 建築用石材、 敷石などの道路 用石材、 灯簧、 庭石、 墓石、 岩風呂、 陶磁器、 食器、 窓ガラス、 車両用ガラ ス (自動車、 電車、 航空機、 船舶など) 、 ランプカバー (蛍光灯、 トンネル 内照明、 道路灯、 街灯、 へッドライ トなど) 、 太陽電池パネルカバーガラス 、 眼鏡、 家具用ガラス (食器棚ガラス戸やガラス製テーブルなど) 、 ガラス 製調度類 (花瓶など) 、 鏡 (道路鏡、 自動車ミラー、 洗面台、 歯科用など) 、 水質浄化用ガラスビーズ、 外装材 (建築用、 自動車、 電車、 航空機、 船舶 など) 、 金属屋根、 サッシ、 金網、 アンテナ、 水栓金具、 成形用型、 ハンド ル、 ハンドルカバ一、 シフトノブ、 ダッシュポ一ド、 バンパー、 電車のつり 革、 網棚、 内張り、 メータ一パネル、 ヘルメットシールド、 ドアノブ、 フロ —リング材、 畳、 ブラインド、 口一ルスクリーン、 家具、 化粧板、 すだれ、 浴室用部材、 手すり、 テーブルクロス、 壁紙、 電化製品 (冷蔵庫、 調理器、 手乾燥器、 パソコン、 マウス、 キーボードなど) 、 合成樹脂成形体、 光触媒 含有マスターバッチ、 繊維原糸、 原料布 (織布ゃ不織布など) 、 衣料品 (上 着、 ズボン、 肌着、 靴下など) 、 寝具類 (シーツ、 ふとん、 毛布など) 、 力 —テン、 のれん、 力一ペット、 マスク、 ハンカチ、 タオル、 座席シート表面 生地、 シートカバ一、 車両マット、 テント、 ロープ、 網、 ソファ、 書道用紙 、 障子紙、 新聞用紙、 非塗工印刷用紙 (上級印刷紙、 中級印刷紙、 下級印刷 紙、 薄葉印刷紙) 、 塗工印刷用紙 (アート紙、 コート紙、 軽量コート紙など ) 、 特殊印刷用紙 (色上質紙、 その他特殊印刷用紙) 、 情報用紙 (コピー用 紙、 ノーカーボンペーパーなど) 、 包装用紙 (未晒し包装紙、 晒し包装紙な ど) 、 衛生用紙 (ティシュペーパー、 ちり紙など) 、 雑種紙 (工業用雑種紙 、 家庭用雑種紙) 、 人工観葉植物、 医療用器具、 燃料電池用電極触媒、 色素 增感太陽電池。 [0058] The photocatalyst-containing body includes an inorganic molded body containing a photocatalyst, an organic resin molded body containing a photocatalyst, a material having a photocatalytic coating film (the substrate may be either an inorganic compound or an organic compound), a photocatalyst paint, There are photocatalyst sprays, etc., which can be used in the following application fields and products: Ceramic filters and glass filters (air conditioners, refrigerators, humidifiers, dehumidifiers, cookers, air purifiers, dust collectors , Wastewater treatment equipment, etc.), paving asphalt, structural materials for RC buildings, humidity control building materials such as diatomaceous earth or zeolite, ceiling materials (rock wool, gypsum, etc.), tile joint materials, concrete blocks and concrete Rocking block, tile (exterior, paving, flooring, etc.), tile, sizing Materials, sealing materials, plastering materials such as building rubber, building stones, road stones such as paving stones, lanterns, garden stones, tombstones, rock baths, ceramics, tableware, window glass, vehicle glass (automobiles, trains, Aircraft, ships, etc.), lamp covers (fluorescent lights, lighting in tunnels, road lights, street lights, headlights, etc.), solar panel cover glass, glasses, furniture glass (such as cupboard glass doors and glass tables), Glass furniture (vases, etc.), mirrors (road mirrors, automobile mirrors, washstands, dental etc.), glass beads for water purification, exterior materials (for construction, automobiles, trains, aircraft, ships, etc.), metal roofs, Sash, wire mesh, antenna, faucet, mold for molding, handle, handle cover, shift knob, dash pad, bumper, train suspension leather, net shelf Liner, Meter panel, Helmet shield, Door knob, Flooring material, Tatami mat, Blind, Mouth screen, Furniture, Decorative plate, Sandwich, Bathroom material, Handrail, Tablecloth, Wallpaper, Electrical appliance (Refrigerator, Cooker , Hand dryers, personal computers, mice, keyboards, etc.), synthetic resin moldings, photocatalyst-containing masterbatches, fiber yarns, raw fabrics (woven fabrics, non-woven fabrics, etc.), clothing (overwear, pants, underwear, socks, etc.) , Bedding (sheets, futons, blankets, etc.), force—ten, goodwill, force pet, mask, handkerchief, towel, seat seat surface fabric, seat cover, vehicle mat, tent, rope, net, sofa, calligraphy paper, Shoji paper, newsprint, uncoated printing paper (high-grade printing paper, intermediate printing paper, low-grade printing paper, thin-leaf printing paper), Coated printing paper (art paper, coated paper, lightweight coated paper, etc.), special printing paper (color fine paper, other special printing paper), information paper (copy paper, carbonless paper, etc.), packaging paper (unexposed packaging) Paper, bleached wrapping paper, etc.), sanitary paper (tissue paper, dust paper, etc.), hybrid paper (industrial hybrid paper, household hybrid paper), artificial foliage plant, medical instrument, fuel cell electrode catalyst, dye sensitization Solar cell.
実施例 Example
以下、 本発明を実施例および比較例を用いて更に詳述するが、 本発明はこ れによって限定されるものではない。 なお、 以下、 本発明の工程 (B ) に相 当する工程を経て調製した、 表面の少なくとも一部にセラミックスまたはそ の前駆体が固定化された基体および凝集剤を含む固形物と含水溶媒とからな るスラリーを 「粗大粒子液」 という。 そして、 実施例 1〜7、 および比較例 1〜 4において、 光半導体粒子のアル力リ金属含有量を記載していないもの については、 アル力リ金属が検出できない量であることを意味するものとす る。 Hereinafter, the present invention will be described in more detail with reference to Examples and Comparative Examples, but the present invention is not limited thereto. Hereinafter, at least a part of the surface prepared through a process corresponding to the process (B) of the present invention, ceramics or the like. A slurry composed of a substrate on which the precursor is immobilized and a solid containing a flocculant and a water-containing solvent is referred to as a “coarse particle liquid”. And in Examples 1 to 7 and Comparative Examples 1 to 4, those that do not describe the content of Al-strand metal in the optical semiconductor particles mean that the amount of Al-strand metal cannot be detected. Suppose that
[0060] はじめに、 実施例および比較例で用いた分析方法について説明する。 [0060] First, analysis methods used in Examples and Comparative Examples will be described.
( i ) アル力リ金属含有量 (i) Al strength metal content
アルカリ金属含有量は、 蛍光 X線分析器 (LAB CEN T ER XRE - 1 700, 島津製作所) を用いて測定した。 また、 本測定で検出されたも のに関して、 原子吸光光度計 (Z— 5000, 日立製作所) を用いて定量し た。 The alkali metal content was measured using a fluorescent X-ray analyzer (LAB CENTER XRE-1700, Shimadzu Corporation). The amount detected in this measurement was quantified using an atomic absorption photometer (Z-5000, Hitachi, Ltd.).
( i i ) 珪素含有量、 Ca含有量、 および P含有量 (i i) Silicon content, Ca content, and P content
蛍光 X線分析器 (LAB CEN T ER XRE- 1 700, 島津製作所 ) を用いて定量した。 Quantification was performed using a fluorescent X-ray analyzer (LAB CENTER XRE-1 700, Shimadzu Corporation).
( i i i ) Mgおよび A I含有量 (i i i) Mg and A I content
I CP発光分光器 (V I S TA_PRO、 S I I社) を用いて定量した。 ( i v ) 比表面積 Quantification was performed using an I CP emission spectrometer (V I S TA_PRO, S I I). (i v) Specific surface area
B E T法比表面積測定装置 (F l owS o r b 2 2300、 島津製作 所) により測定した。 It was measured by a BET method specific surface area measuring apparatus (FlowSorb 2 2300, Shimadzu Corporation).
(V) 結晶子径 (V) Crystallite diameter
X線回折 (XRD) 測定を行い、 シエラ一式を用いて算出した。 X-ray diffraction (XRD) measurements were performed and calculated using the Sierra suite.
[0061] (実施例 1 ) [0061] (Example 1)
(1 -A ;混合液 1の調製) (1 -A; Preparation of mixture 1)
ガラスフラスコに水 200 gと 1 mo I /Lの硝酸水溶液 81. 7 gを加 え、 二酸化チタン (S T_01、 石原産業株式会社、 水分量 9重量%、 比表 面積 300m2/g、 結晶子径 6 nm、 N a含有量 1 400 p pm) 24. 5 gを分散させて、 A液とした。 ビーカ一内に水 1 00 gと珪酸ナトリウム水 溶液 (S i 02含有量29. 1重量%、 N a20含有量 9. 5重量%、 J I S K 1 408〃水ガラス 3号") 1 3. 5 gを加え、 攪拌し B液とした。 A液 を 35°Cに保持し、 攪拌しているところに、 B液を 2m I /分で滴下し、 混 合液 Cを得た。 滴下終了時の混合液 Cの p Hは 2. 8であった。 混合液 Cを 35°Cに保持したまま 1 6時間攪拌し、 混合液 1 とした。 なお、 混合液 1の スラリー濃度は 5. 3重量%である。 Add 200 g of water and 81.7 g of 1 mo I / L nitric acid aqueous solution to a glass flask. Titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, ratio table area 300 m 2 / g, crystallite (Diameter 6 nm, Na content 1 400 ppm) 24.5 g was dispersed to prepare Liquid A. Water 1 00 g sodium silicate water solution beaker in one (S i 0 2 content of 29.1 wt%, N a 2 0 content of 9.5 wt%, JIS K 1 408 water-quenched glass No. 3 ") 1 Add 3 g and stir to make liquid B. Keep liquid A at 35 ° C and stir while liquid B at 2 m I / min. The mixture was added dropwise to obtain a mixture C. The pH of the mixture C at the end of the addition was 2.8, and the mixture C was stirred for 16 hours while maintaining the temperature at 35 ° C. The slurry concentration of Mixture 1 is 5.3% by weight.
[0062] (1 -B ;粗大粒子液 1の調製) [0062] (1-B; Preparation of coarse particle liquid 1)
混合液 1の全量に対して、 室温で攪拌しながら、 市販の高分子凝集剤アコ フロック (三井化学アクアポリマー登録商標) N 21 0 (三井化学アクアポ リマ一社製、 ノニオン型、 変性アクリルアミ ドボリマ一) 26mgを粉末の ままで、 3回に分けて投入した。 なお、 高分子凝集剤の使用量は、 0. 1重 量%に相当する。 攪拌を続けていると徐々に粗大粒子が形成され、 高分子凝 集剤の投入完了より 1 5分間攪拌することで、 粗大粒子液 1を得た。 粗大粒 子液 1は、 攪拌を止めて 30分放置した後には、 フラスコ底部に白色のフロ ックが沈降しており、 上澄み液はわずかに白濁していた。 Commercially available polymer flocculant Acofloc (registered trademark of Mitsui Chemicals Aqua Polymer) N 21 0 (Mitsui Chemicals Aqua Polymer Co., Ltd., nonionic, modified acrylic amide polymer) 1) 26mg of powder was added in three divided doses. The amount of the polymer flocculant used corresponds to 0.1% by weight. When stirring was continued, coarse particles were gradually formed, and the coarse particle liquid 1 was obtained by stirring for 15 minutes after completion of the addition of the polymer coagulant. In coarse particle solution 1, after stirring was stopped and allowed to stand for 30 minutes, a white floc settled on the bottom of the flask, and the supernatant liquid was slightly cloudy.
[0063] (1 -C ;光半導体粒子 1の調製) [0063] (1-C; Preparation of optical semiconductor particle 1)
粗大粒子液 1の全量から、 減圧ろ過によって固形物を回収し、 500mL の純水への再分散、 並びに減圧ろ過を 4回交互に繰り返して洗浄を行った。 次に洗浄した固形物を、 磁性皿に入れ、 電気炉を用いて、 空気雰囲気下、 1 20°Cで 3時間加熱した。 続けて 600°Cに昇温した後、 3時間 600°Cを 保持した。 加熱後、 室温になるまで放置して、 光半導体粒子 1を 23. 4 g 得た。 この光半導体粒子 1は、 白色粉末であり、 珪素含有量は 5. 7重量% であり、 比表面積は 1 87. 1 m2/gであった。 よって、 光半導体粒子 1の 表面積 1 m2当りの珪素担持量は 0. 31 mgであった。 また光半導体粒子 1 はナトリウム含有量 1 60 p pmであった。 From the total amount of the coarse particle liquid 1, solids were recovered by vacuum filtration, and washed by repeating redispersion in 500 mL of pure water and vacuum filtration four times alternately. The washed solid was then placed in a magnetic dish and heated at 120 ° C for 3 hours in an air atmosphere using an electric furnace. The temperature was continuously raised to 600 ° C, and then kept at 600 ° C for 3 hours. After heating, the mixture was allowed to stand at room temperature to obtain 23.4 g of photo semiconductor particles 1. This optical semiconductor particle 1 was a white powder, the silicon content was 5.7 wt%, and the specific surface area was 187.1 m2 / g. Therefore, the amount of silicon supported per 1 m 2 of the surface area of the optical semiconductor particle 1 was 0.31 mg. The photo-semiconductor particle 1 had a sodium content of 1 60 ppm.
[0064] (実施例 2) [0064] (Example 2)
(2-A ;混合液 2の調製) (2-A; Preparation of mixture 2)
実施例 1 (1 _A) と同様にして、 混合液 2を得た。 A liquid mixture 2 was obtained in the same manner as in Example 1 (1_A).
[0065] (2-B ;粗大粒子液 2の調製) 市販の凝集剤 N 21 0を 1 30mg用いて、 1 2回に分けて投入した以外 は、 混合液 2を用いて実施例 1 (1 _B) と同様にして、 粗大粒子液 2を得 た。 粗大粒子液 2は、 攪拌を止めて 30分放置した後には、 フラスコ底部に 白色のフロックが沈降しており、 上澄み液は透明だった。 なお、 高分子凝集 剤の使用量は、 0. 6重量%に相当する。 [0065] (2-B; Preparation of coarse particle liquid 2) A coarse particle liquid 2 was obtained in the same manner as in Example 1 (1_B) using the mixed liquid 2 except that 1 30 mg of the commercially available flocculant N 210 was used and charged in 12 portions. After the coarse particle liquid 2 was left stirring for 30 minutes, white floc settled at the bottom of the flask, and the supernatant liquid was clear. The amount of the polymer flocculant used corresponds to 0.6% by weight.
[0066] (2-C ;光半導体粒子 2の調製) [0066] (2-C; Preparation of optical semiconductor particle 2)
粗大粒子液 2の全量をガラス製 50 Om Lビーカーに移し、 30分放置し た後、 上澄み液をデカント除去した。 次に、 以下の (a) 〜 (d) を 5回繰 り返して洗浄を行った。 The entire amount of the coarse particle liquid 2 was transferred to a glass 50 Om L beaker and allowed to stand for 30 minutes, and then the supernatant liquid was decanted off. Next, washing was performed by repeating the following (a) to (d) five times.
(a) 純水 33 Om Lをビーカ一に注いだ、 (a) Pure water 33 Om L was poured into the beaker.
(b) ガラス棒で 1分間攪拌した、 (b) Stir with a glass rod for 1 minute,
(c) 30分間、 放置した、 (c) Left for 30 minutes,
(d) 上澄み液をデカント除去した。 (d) The supernatant was decanted off.
そして、 ビーカ一の内容物を、 減圧ろ過して固形物を回収した。 回収した 白色固形物を、 磁性皿に入れ、 電気炉を用いて、 空気雰囲気下、 1 20°Cで 3時間加熱した。 続けて 600°Cに昇温した後、 3時間 600°Cを保持した 。 加熱後、 室温になるまで放置して、 光半導体粒子 2を 24. 1 g得た。 こ の光半導体粒子 2は、 白色粉末であり、 珪素含有量 5. 7重量%、 比表面積 1 95. 9m2/g、 表面積 1 m2当りの珪素担持量 0. 29mg、 ナトリウ ム含有量 1 50 p pmであった。 The contents of the beaker were filtered under reduced pressure to recover the solid matter. The collected white solid was placed in a magnetic dish and heated at 120 ° C for 3 hours in an air atmosphere using an electric furnace. The temperature was continuously raised to 600 ° C, and then kept at 600 ° C for 3 hours. After heating, the mixture was allowed to stand at room temperature to obtain 24.1 g of photo semiconductor particles 2. This photo-semiconductor particle 2 is a white powder, with a silicon content of 5.7 wt%, a specific surface area of 195.99 m 2 / g, a silicon loading of 1/29 m 2 , and a sodium content of 1 It was 50 p pm.
[0067] (実施例 3) [0067] (Example 3)
(3-A ;混合液 3の調製) (3-A; Preparation of mixture 3)
容積 1. 5 Lのガラス製セパラブルフラスコに、 水 555 gと 60%硝酸 22. 7 gを加え、 二酸化チタン (S T_01、 石原産業株式会社、 水分量 9重量%、 比表面積 300m2/g、 結晶子径 6 nm, N a含有量 1 400 p pm) 1 22. O gを分散させて、 A液とした。 ビーカ一内に水 232. 4 gと珪酸ナトリウム水溶液 (S i 02含有量29重量%、 N a20含有量 9. 5重量0 /0、 J I S K 1 408〃水ガラス 3号") 67. 6 gを加え、 攪拌し B液とした。 A液を 35 °Cに保持し、 攪拌しているところに、 B液を 3 m l /分で滴下し、 混合液 Cを得た。 滴下終了時の混合液 Cの p Hは 3. 9であ つた。 混合液 Cを 35°Cに保持したまま 1 6時間攪拌し、 混合液 3とした。 なお、 混合液 3のスラリー濃度は 1 1. 1重量%である。 Add 555 g of water and 22.7 g of 60% nitric acid to a glass separable flask with a volume of 1.5 L. Titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, specific surface area 300 m 2 / g (Crystallite diameter 6 nm, Na content 1 400 ppm) 1 22. Og was dispersed to prepare a solution A. Beaker of water 232. 4 g sodium silicate solution in one (S i 0 2 content of 29 wt%, N a 2 0 content of 9.5 wt 0/0, JISK 1 408〃 water glass No. 3 ") 67. Add 6 g and stir B liquid was used. While liquid A was maintained at 35 ° C. and stirred, liquid B was added dropwise at 3 ml / min to obtain liquid mixture C. The pH of mixture C at the end of dropping was 3.9. The mixture C was stirred for 16 hours while maintaining the temperature at 35 ° C. to obtain a mixture 3. The slurry concentration of mixture 3 is 11.1% by weight.
[0068] (3-B ;粗大粒子液 3の調製) [0068] (3-B; Preparation of coarse particle liquid 3)
混合液 1の代わりに混合液 3の 3分の 1量を用いたことと、 市販の凝集剤 N 21 0を 21 8mg用いて、 23回に分けて投入した事が異なる以外は、 実施例 1 (1 _B) と同様にして、 粗大粒子液 3を得た。 粗大粒子液 3は、 攪拌を止めて 30分放置した後には、 フラスコ底部に白色のフロックが沈降 しており、 上澄み液は透明だった。 なお、 高分子凝集剤の使用量は、 0. 6 重量%に相当する。 また、 攪拌を止めてフロックが沈降する際に、 スラッジ と上澄み液の境が 1分間に下降する長さを計測し、 フロックの沈降速度を求 めた。 その結果、 フロックの沈降速度は、 3. 7 cm/分であった。 Example 1 except that 1/3 of the mixture 3 was used instead of the mixture 1 and that 21 8 mg of the commercially available flocculant N 2120 was used and was added in 23 portions. In the same manner as (1 _B), coarse particle liquid 3 was obtained. Coarse particle liquid 3 was stirred for 30 minutes and left to stand for 30 minutes. White floc settled on the bottom of the flask, and the supernatant liquid was clear. The amount of the polymer flocculant used corresponds to 0.6% by weight. In addition, when the flocs settled when stirring was stopped, the length at which the boundary between the sludge and the supernatant liquid descended in 1 minute was measured to determine the floc sedimentation rate. As a result, the sedimentation rate of floc was 3.7 cm / min.
[0069] (3-C ;光半導体粒子 3の調製) [0069] (3-C; Preparation of optical semiconductor particle 3)
粗大粒子液 3をガラス製 50 OmLビーカ一に移し、 30分放置した。 そ の後、 上澄み液をデカント除去した。 次に、 ガラスカラム管 (内径 9 Omm 、 長さ 300mm、 下端; 2方コック付属、 上端; 内径 4 mmのガラス管を 任意の高さに保持可能で且つガラス管内を除き密封可能な栓が付属) を垂直 に固定し、 下端のコックを閉じた状態で、 上端の栓を開放して、 ビーカ一の 内容物をカラム内に移した。 そして、 上端の栓を塞ぎ、 下端のコックを開い た後に、 下端より純水を毎分 1 OmLの速度で 5時間供給して、 上端のガラ ス管を介して排水を流出させた。 その際、 カラム管内部の液量が 40 Om L となるように、 ガラス管の高さを調整した。 なお、 純水の供給速度とカラム 管の内径から算出した、 カラム管内の液の上昇流速は 0. 1 6 cm/分であ り、 前記のフロック沈降速度の 4%に相当する。 5時間後、 純水の供給を停 止し、 カラム管の内容物を減圧ろ過して、 固形物を回収した。 回収した白色 固形物を磁性皿に入れ、 電気炉を用いて、 空気雰囲気下 1 20°Cで 3時間加 熱した。 続けて 600°Cに昇温した後、 3時間 600°Cを保持した。 加熱後 、 室温になるまで放置して、 光半導体粒子 3を 42. 5 g得た。 この光半導 体粒子 3は、 白色粉末であり、 珪素含有量 6. 1重量%、 比表面積 209. 8m2/g、 表面積 1 m2当りの珪素担持量 0. 29mg、 ナトリウム含有量 は 1 70 p p mであった。 The coarse particle solution 3 was transferred to a glass 50 OmL beaker and allowed to stand for 30 minutes. Thereafter, the supernatant was decanted off. Next, glass column tube (inner diameter 9 Omm, length 300 mm, lower end; 2-way cock included, upper end; 4 mm inner diameter glass tube can be held at any height and sealed except for inside the glass tube. ) Was fixed vertically, the bottom end cock was closed, the top end plug was opened, and the contents of the beaker were transferred into the column. After closing the stopper at the upper end and opening the cock at the lower end, pure water was supplied from the lower end at a rate of 1 OmL per minute for 5 hours, and drainage was discharged through the glass pipe at the upper end. At that time, the height of the glass tube was adjusted so that the liquid volume inside the column tube was 40 Om L. Note that the rising velocity of the liquid in the column tube, calculated from the supply rate of pure water and the inner diameter of the column tube, is 0.16 cm / min, which corresponds to 4% of the floc sedimentation rate. After 5 hours, the supply of pure water was stopped, and the contents of the column tube were filtered under reduced pressure to recover the solid matter. The collected white solid was placed in a magnetic dish and heated in an air atmosphere at 120 ° C for 3 hours using an electric furnace. The temperature was continuously raised to 600 ° C, and then kept at 600 ° C for 3 hours. After heating 42.5 g of photo-semiconductor particles 3 were obtained by standing until the temperature reached room temperature. This optical semiconductor particle 3 is a white powder having a silicon content of 6.1% by weight, a specific surface area of 209.8 m 2 / g, a silicon loading of 0.29 mg per 1 m 2 of surface area, and a sodium content of 1 It was 70 ppm.
[0070] (実施例 4) [0070] (Example 4)
(4-A ;混合液 4の調製) (4-A; Preparation of mixture 4)
混合液 3の 3分の 1量を取り分けて、 混合液 4とした。 One third of the mixture 3 was divided into 4 mixture.
[0071 ] (4-B ;粗大粒子液 4の調製) [0071] (4-B; Preparation of coarse particle liquid 4)
混合液 1の代わりに混合液 4を用いたことと、 435 m gの高分子凝集剤 N 21 0を 36回に分けて投入した事が異なる以外は、 実施例 1 ( 1 _B) と同様にして、 粗大粒子液 4を得た。 なお、 高分子凝集剤の使用量は、 1. 2重量%に相当する。 Same as Example 1 (1 _B) except that the mixture 4 was used instead of the mixture 1 and that 435 mg of the polymer flocculant N 210 was added in 36 portions. Coarse particle liquid 4 was obtained. The amount of polymer flocculant used is equivalent to 1.2% by weight.
[0072] (4-C ;光半導体粒子 4の調製) [0072] (4-C; Preparation of optical semiconductor particles 4)
粗大粒子液 3の代わりに粗大粒子液 4を用いたことと、 600 °Cを 5時間 保持したことが異なる以外は、 実施例 3 (3-C) と同様にして、 光半導体 粒子 4を 41. 9 g得た。 この光半導体粒子 4は、 白色粉末であり、 珪素含 有量 6. 4重量%、 比表面積 21 2. 3m2/g、 表面積 1 m 2当りの珪素担 持量は 0. 30mg、 ナトリウム含有量は 200 p pmであった。 In the same manner as in Example 3 (3-C), except that the coarse particle liquid 4 was used instead of the coarse particle liquid 3, and that 600 ° C. was maintained for 5 hours, the optical semiconductor particles 4 were changed to 41 9 g was obtained. This photo-semiconductor particle 4 is a white powder with a silicon content of 6.4% by weight, a specific surface area of 212.3 m 2 / g, and a silicon load of 0.30 mg per 1 m 2 of surface area, a sodium content. Was 200 p pm.
[0073] (実施例 5) [0073] (Example 5)
(5-A ;混合液 5の調製) (5-A; Preparation of mixture 5)
ガラスフラスコに水 21 0 gと 60%硝酸 2. 8 gを加え、 二酸化チタン (S T_01、 石原産業株式会社、 水分量 9重量%、 比表面積 300 m2/ g 、 結晶子径 6 nm、 N a含有量 1 400 p pm) 20. 4 gを分散させて、 A液とした。 ビーカー内に水 88. 2 gと珪酸カリゥム水溶液 (和光純薬ェ 業製, S i 02含有量 28重量%) 1 1. 7 gを加え、 攪拌し B液とした。 A 液を 25°Cに保持し、 攪拌しているところに、 B液を 2m I /分で滴下し、 混合液 Cを得た。 滴下終了時の混合液 Cの p Hは 2. 9であった。 混合液 C を 25°Cに保持したまま 1 6時間攪拌し、 混合液 5とした。 なお、 混合液 5 のスラリー濃度は、 5. 6重量%である。 Add 210 g of water and 2.8 g of 60% nitric acid to a glass flask. Titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, specific surface area 300 m 2 / g, crystallite diameter 6 nm, N a content 1 400 p pm) 20. 4 g was dispersed to prepare a solution A. In a beaker, 88.2 g of water and an aqueous solution of potassium silicate (manufactured by Wako Pure Chemical Industries, Ltd., Si 0 2 content 28% by weight) 1 1.7 g were added and stirred to make B solution. The liquid A was kept at 25 ° C. and stirred while the liquid B was added dropwise at 2 m I / min to obtain a liquid mixture C. The pH of mixture C at the end of the addition was 2.9. The mixture C was stirred for 16 hours while maintaining the temperature at 25 ° C. to obtain a mixture 5. Mixture 5 The slurry concentration of 5.6% by weight.
[0074] (5-B ;粗大粒子液 5の調製) [0074] (5-B; Preparation of coarse particle liquid 5)
混合液 1の代わりに混合液 5を用いた事と、 1 09 m gの高分子凝集剤 N 21 0を 1 0回に分けて投入した事が異なる以外は、 実施例 1 (1 _B) と 同様にして、 粗大粒子液 5を得た。 なお、 高分子凝集剤の使用量は、 0. 6 重量%に相当する。 Same as Example 1 (1 _B), except that mixed solution 5 was used instead of mixed solution 1 and that 109 mg of polymer flocculant N 2120 was added in 10 batches. Thus, a coarse particle liquid 5 was obtained. The amount of the polymer flocculant used corresponds to 0.6% by weight.
(5-C ;光半導体粒子 5の調製) (5-C; Preparation of photo-semiconductor particles 5)
粗大粒子液 2の代わりに粗大粒子液 5を用いたことが異なる以外は、 実施 例 2 (2-C) と同様にして、 光半導体粒子 5を 20. 6 g得た。 この光半 導体粒子 5は、 白色粉末であり、 珪素含有量 4. 9重量%、 比表面積 1 93 . 9m2/g、 表面積 1 m2当りの珪素担持量 0. 25mg、 ナトリウム含有 量 80 p pm, カリウム含有量 1 00 p p mであった。 In the same manner as in Example 2 (2-C) except that the coarse particle liquid 5 was used instead of the coarse particle liquid 2, 20.6 g of the photo semiconductor particles 5 was obtained. This optical semiconductor particle 5 is a white powder, silicon content 4.9% by weight, specific surface area 193.9 m 2 / g, silicon loading 0.25 mg per 1 m 2 surface area, sodium content 80 p pm, potassium content was 100 ppm.
[0075] (実施例 6) [0075] (Example 6)
(6-A ;混合液 6の調製) (6-A; Preparation of mixture 6)
ガラスフラスコに水 230 gと 1 mo I /Lの塩酸水溶液 25. 2 gを加 え、 二酸化チタン (P_25、 日本ァエロジル株式会社、 アナタ一ゼ:ルチ ル比が 8 : 2の混合体、 純度 99. 5%、 比表面積 50 m2/ g, アルカリ金 属が検出されない) 22. 4 gを分散させて、 A液とした。 ビーカ一内に水 1 00 gと珪酸ナトリゥム水溶液 ( S i O 2含有量 29重量%、 N a 2 O含有 量 9. 5重量0 /o、 J I S K 1 408"水ガラス 3号") 2. 4 gを加え、 攪 拌し B液とした。 A液を 35 °Cに保持し、 攪拌しているところに、 B液を 2 m I /分で滴下し、 混合液 Cを得た。 滴下終了時の混合液 Cの p Hは 2. 9 であった。 混合液 Cを 35°Cに保持したまま 72時間攪拌し、 混合液 6とし た。 なお、 混合液 6のスラリー濃度は、 5. 9重量%である。 Add 230 g of water and 25.2 g of 1 mo I / L hydrochloric acid solution to a glass flask and mix with titanium dioxide (P_25, Nippon Aerosil Co., Ltd., ANATAZE: rutile ratio 8: 2, purity 99 (5%, specific surface area 50 m 2 / g, no alkali metal detected) 22.4 g was dispersed to prepare Liquid A. 100 g of water in a beaker and sodium silicate aqueous solution (SiO 2 content 29 wt%, Na 2 O content 9.5 wt 0 / o, JISK 1 408 "Water Glass No. 3") 2.4 g was added and stirred to make B liquid. Liquid A was kept at 35 ° C. and stirred while liquid B was added dropwise at 2 m I / min to obtain liquid mixture C. The pH of the mixed solution C at the end of dropping was 2.9. The mixture C was stirred for 72 hours while maintaining the temperature at 35 ° C. to obtain a mixture 6. The slurry concentration of the mixed solution 6 is 5.9% by weight.
[0076] (6-B ;粗大粒子液 6の調製) [0076] (6-B; Preparation of coarse particle liquid 6)
混合液 1の代わりに混合液 6を用いた事と、 市販の高分子凝集剤 N 21 0の 代わりにアコフロック (三井化学アクアポリマー登録商標) N 1 00S (三 井化学アクアポリマー社製、 ノニオン型、 ポリアクリルアミ ド) を 1 1 5m g用いて、 1 0回に分けて投入した事が異なる以外は、 実施例 1 (1—B) と同様にして、 粗大粒子液 6を得た。 粗大粒子液 6は、 攪拌を止めて 30分 放置した後には、 フラスコ底部に白色のフロックが沈降しており、 上澄み液 は透明だった。 なお、 高分子凝集剤の使用量は、 0. 5重量%に相当する。Mixture 6 was used in place of Mixture 1, and Acofloc (registered trademark of Mitsui Chemicals Aqua Polymer) N 1 00S (Nonion type, Mitsui Chemicals Aqua Polymer Co., Ltd.) instead of the commercially available polymer flocculant N 2100 Polyacrylamide) 1 1 5m The coarse particle liquid 6 was obtained in the same manner as in Example 1 (1-B), except that it was added in 10 batches. In the coarse particle liquid 6, after stirring was stopped for 30 minutes, a white floc settled on the bottom of the flask, and the supernatant liquid was transparent. The amount of polymer flocculant used corresponds to 0.5% by weight.
(6-C ;光半導体粒子 6の調製) (6-C; Preparation of optical semiconductor particles 6)
粗大粒子液 2の代わりに粗大粒子液 6を用いたことが異なる以外は、 実施例 2 (2-C) と同様にして、 光半導体粒子 6を 22. 5 g得た。 この光半導 体粒子 6は、 白色粉末であり、 珪素含有量 1. 3重量%、 比表面積 52. 9 m2/g、 表面積 1 m2当りの珪素担持量は 0 25mg、 ナトリウム含有量 は 40 p p mであった。 22.5 g of photo-semiconductor particles 6 were obtained in the same manner as in Example 2 (2-C) except that the coarse particle solution 6 was used instead of the coarse particle solution 2. This photo-semiconductor particle 6 is a white powder, with a silicon content of 1.3% by weight, a specific surface area of 52.9 m2 / g, a silicon loading of 0 25 mg per 1 m2 of surface area, and a sodium content of 40 ppm. Met.
[0077] (実施例 7 ) [0077] (Example 7)
(7-A ;混合液 7の調製) (7-A; Preparation of mixture 7)
実施例 6 (6-A) と同様にして、 混合液 7を得た。 A liquid mixture 7 was obtained in the same manner as in Example 6 (6-A).
(7-B ;粗大粒子液 7の調製) (7-B; Preparation of coarse particle liquid 7)
実施例 6 (6-B) と同様にして、 粗大粒子液 7を得た。 粗大粒子液 7は 、 攪拌を止めて 30分放置した後には、 フラスコ底部に白色のフロックが沈 降しており、 上澄み液は透明だった。 なお、 高分子凝集剤の使用量は、 0. 5重量%に相当する。 In the same manner as in Example 6 (6-B), a coarse particle liquid 7 was obtained. After the coarse particle liquid 7 was left stirring for 30 minutes, a white floc had settled at the bottom of the flask, and the supernatant liquid was transparent. The amount of polymer flocculant used corresponds to 0.5% by weight.
[0078] (7-C ;光半導体粒子 7の調製) [0078] (7-C; Preparation of optical semiconductor particles 7)
粗大粒子液 7を、 減圧ろ過して固形物を回収した。 回収した白色固形物を 、 磁性皿に入れ、 電気炉を用いて、 空気雰囲気下、 1 20°Cで 3時間加熱し た。 続けて 600°Cに昇温した後、 3時間 600°Cを保持した。 加熱後、 室 温になるまで放置して、 光半導体粒子 7を 22. 9 g得た。 この光半導体粒 子 7は、 白色粉末であり、 珪素含有量 1. 3重量%、 比表面積 54. 2m2/ g、 表面積 1 m2当りの珪素担持量 0. 24mg、 ナトリウム含有量 820 p p mであった。 The coarse particle liquid 7 was filtered under reduced pressure to collect a solid. The collected white solid was put in a magnetic dish and heated at 120 ° C. for 3 hours in an air atmosphere using an electric furnace. The temperature was continuously raised to 600 ° C, and then kept at 600 ° C for 3 hours. After heating, the mixture was allowed to stand until the room temperature was reached, and 29.9 g of photosemiconductor particles 7 were obtained. This photo-semiconductor particle 7 is a white powder with a silicon content of 1.3% by weight, a specific surface area of 54.2 m 2 / g, a silicon loading of 0.24 mg per 1 m 2 of surface area, and a sodium content of 820 ppm. there were.
[0079] (実施例 8) [0079] (Example 8)
(8-A ;混合液 8の調製) 擬似体液として、 塩化ナトリゥム、 炭酸水素ナトリゥム、 塩化力リゥム、 リン酸水素ニナトリウム、 塩化カルシウム、 塩酸、 並びに水を用いて、 N a イオン 1 47mM、 Kイオン 5mM、 Caイオン 7. 5mM、 炭酸水 素イオン 4. 2mM、 リン酸水素イオン 5mMの組成で、 p H 7. 4の 水溶液を調製した。 そして、 この擬似体液 2000 gに、 二酸化チタン (S T_01、 石原産業株式会社、 水分量 9重量%、 比表面積 3 O Om 2/ g、 結晶子径 6 nm) 1 1. O gを加え、 50 °Cで 8時間攪拌し続けた。 この段 階でのスラリー濃度は、 0. 5重量%である。 次に、 加熱と攪拌を停止し、 室温で 1 8時間静置したところ、 白色沈殿が底に生じ、 上澄み液は僅かに濁 つていた。 上澄み液をデカント除去した後、 沈殿物を純水で希釈して、 全量 を 1 00 gにした。 これに超音波照射と 1時間の攪拌処理を行い、 沈殿を再 分散させて混合液 8とした。 なお、 混合液 8のスラリー濃度は、 1 0. 0重 量%である。 (8-A; Preparation of mixture 8) Using simulated body fluids such as sodium chloride, sodium bicarbonate, lithium chloride, disodium hydrogen phosphate, calcium chloride, hydrochloric acid, and water, Na ion 147 mM, K ion 5 mM, Ca ion 7.5 mM, carbonated water An aqueous solution of pH 7.4 was prepared with a composition of 4.2 mM elementary ions and 5 mM hydrogen phosphate ions. Titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, specific surface area 3 O Om 2 / g, crystallite diameter 6 nm) is added to 2000 g of this simulated body fluid. Stirring was continued at ° C for 8 hours. The slurry concentration at this stage is 0.5% by weight. Next, heating and stirring were stopped, and the mixture was allowed to stand at room temperature for 18 hours. As a result, a white precipitate was formed at the bottom, and the supernatant liquid was slightly turbid. After decanting off the supernatant, the precipitate was diluted with pure water to a total volume of 100 g. This was subjected to ultrasonic irradiation and stirring treatment for 1 hour, and the precipitate was redispersed to obtain a mixed solution 8. The slurry concentration of the mixed solution 8 is 10.0% by weight.
(8-B ;粗大粒子液 8の調製) (8-B; Preparation of coarse particle liquid 8)
混合液 1の代わりに混合液 8を用いた事と、 市販の高分子凝集剤 N 21 0 を 57mg、 粉末のまま 5回に分けて投入した事が異なる以外は、 実施例 1 (1 -B) と同様にして、 粗大粒子液 8を得た。 粗大粒子液 8は、 攪拌を止 めて、 30分放置した後には、 フラスコ底部に白色のフロックが沈降してお り、 上澄み液は透明だった。 なお、 高分子凝集剤の使用量は、 0. 6重量% に相当する。 Example 1 (1 -B), except that the mixture 8 was used instead of the mixture 1 and that the commercially available polymer flocculant N 2120 was added in 57 mg, powdered in 5 portions. In the same manner, coarse particle liquid 8 was obtained. After the coarse particle liquid 8 was stopped stirring and allowed to stand for 30 minutes, white floc settled on the bottom of the flask, and the supernatant liquid was transparent. The amount of polymer flocculant used corresponds to 0.6% by weight.
(8-C ;光半導体粒子 8の調製) (8-C; Preparation of optical semiconductor particles 8)
粗大粒子液 8をガラス製 20 OmLビーカ一に移し、 30分放置した後、 上澄み液をデカント除去した。 次に、 以下の (a) 〜 (d) を 5回繰り返し て洗浄を行った。 The coarse particle liquid 8 was transferred to a glass 20 OmL beaker and allowed to stand for 30 minutes, and then the supernatant liquid was decanted off. Next, the following (a) to (d) were repeated 5 times for washing.
(a) 純水 1 3 Om Lをビーカ一に注いだ、 (a) Pure water 1 3 Om L was poured into a beaker,
(b) ガラス棒で 5分間攪拌した、 (b) stirred for 5 minutes with a glass rod,
(c) 30分間、 放置した、 (c) Left for 30 minutes,
(d) 上澄み液をデカント除去した。 そして、 ビーカ一の内容物を、 減圧ろ過して固形物を回収した。 回収した 白色固形物を、 磁性皿に入れ、 電気炉を用いて、 空気雰囲気下、 1 20°Cで 3時間加熱した。 続けて 450°Cに昇温した後、 1 2時間 450°Cを保持し た。 加熱後、 室温になるまで放置して、 光半導体粒子 8を 1 0. 9 g得た。 この光半導体粒子 8は、 白色粉末であり、 蛍光 X線分析より Caを 4. 4重 量0 /o、 Pを 1. 9重量%含んでいた。 (d) The supernatant was decanted off. The contents of the beaker were filtered under reduced pressure to recover the solid matter. The collected white solid was placed in a magnetic dish and heated at 120 ° C for 3 hours in an air atmosphere using an electric furnace. Subsequently, the temperature was raised to 450 ° C, and then maintained at 450 ° C for 12 hours. After heating, the mixture was allowed to stand at room temperature to obtain 10.9 g of photo semiconductor particles 8. This photo-semiconductor particle 8 was a white powder, and contained 4.4 weight of Ca ( 0 / o) and 1.9 weight% of P by fluorescent X-ray analysis.
[0081] (実施例 9) [Example 9]
(9-A ;混合液 9の調整) (9-A; adjustment of mixture 9)
ガラスフラスコに水 450 gを入れ、 炭酸ナトリウム十水和物 1 6. 5 g を溶解した後、 二酸化チタン (S T_01、 石原産業株式会社、 水分量 9重 量%、 比表面積 300m2/g、 結晶子径 6 nm, N a含有量 1 400 p pm ) 1 5. 37 gを分散させて、 X液とした。 この時、 X液の p Hは 1 0. 9 であった。 次にビーカ一に、 水 280 g、 硝酸マグネシゥム六水和物 6. 3 9 g、 並びに硝酸アルミニウム九水和物 3. 09 gを入れ、 攪拌して Y液と した。 続いて、 X液を攪拌しているところに、 Y液を 1 Om I /分で滴下し 、 混合液 Zを得た。 滴下終了時の混合液 Zの p Hは 9. 2であった。 混合液 Zを 64°Cに保持したまま 20時間攪拌し、 混合液 9とした。 なお、 混合液 9のスラリー濃度は、 1. 8重量%である。 After putting 450 g of water into a glass flask and dissolving 16.5 g of sodium carbonate decahydrate, titanium dioxide (ST_01, Ishihara Sangyo Co., Ltd., water content 9% by weight, specific surface area 300 m 2 / g, Crystallite diameter 6 nm, Na content 1 400 p pm) 1 5.37 g was dispersed to prepare X solution. At this time, the pH of the solution X was 10.9. Next, 280 g of water, 6.39 g of magnesium nitrate hexahydrate, and 3.09 g of aluminum nitrate nonahydrate were placed in a beaker and stirred to make Y solution. Subsequently, the Y solution was added dropwise at 1 Om I / min while stirring the X solution to obtain a mixed solution Z. The pH of the mixed solution Z at the end of dropping was 9.2. The mixture Z was stirred for 20 hours while maintaining the temperature at 64 ° C. to obtain a mixture 9. The slurry concentration of the mixed solution 9 is 1.8% by weight.
[0082] (9-B ;粗大粒子液 9の調製) [0082] (9-B; Preparation of coarse particle liquid 9)
混合液 1の代わりに混合液 9を用いたことと、 1 50 m gの高分子凝集剤 N 21 0を 1 4回に分けて投入した事が異なる以外は、 凝集剤を 1 6回に分 けて投入した事が異なる以外は、 実施例 1 (1 _B) と同様にして、 粗大粒 子液 9を得た。 粗大粒子液 9は、 攪拌を止めて 30分放置した後には、 フラ スコ底部に白色のフロックが沈降しており、 上澄み液は少し白く濁っていた 。 なお、 高分子凝集剤の使用量は、 1. 1重量%に相当する。 Divide the flocculant into 16 times, except that the mixture 9 was used instead of the liquid mixture 1 and that 150 mg of the polymer flocculant N2120 was added in 14 portions. The coarse particle solution 9 was obtained in the same manner as in Example 1 (1_B) except that the charged particles were different. In the coarse particle liquid 9, after stirring was stopped and left for 30 minutes, a white floc settled on the bottom of the flask, and the supernatant liquid was slightly white and cloudy. The amount of polymer flocculant used is equivalent to 1.1% by weight.
[0083] (9-C ;光半導体粒子 9の調製) [0083] (9-C; Preparation of optical semiconductor particles 9)
粗大粒子液 2の代わりに粗大粒子液 9を用いたことが異なる以外は、 実施 例 2 (2-C) と同様にして、 光半導体粒子 9を 1 5. 1 g得た。 この光半 導体粒子 9は、 白色粉末であり、 I CP分光分析より Mgを 2. 0重量%、 A I を 1. 4重量%含んでいた。 15.1 g of photo-semiconductor particles 9 was obtained in the same manner as in Example 2 (2-C) except that coarse particle liquid 9 was used instead of coarse particle liquid 2. This light half Conductor particle 9 was a white powder and contained 2.0% by weight of Mg and 1.4% by weight of AI by ICP spectroscopy.
[0084] (比較例 1 ) [0084] (Comparative Example 1)
( 1 0-A ;混合液 1 0の調製) (10-A; Preparation of mixed solution 10)
実施例 1 と同様にして、 混合液 1 0を得た。 混合液 1 0は、 攪拌を止めて 30分放置した後にも、 上澄み液は白濁したままだった。 In the same manner as in Example 1, a mixed solution 10 was obtained. The mixture solution 10 remained cloudy even after stirring was stopped and left for 30 minutes.
( 1 0-B ;粗大粒子液 1 0の調製) (1 0-B; Preparation of coarse particle liquid 10)
比較のために、 凝集剤を用いなかったので、 粗大粒子液 1 0は調製しなか つた。 For comparison, no flocculant was used, so the coarse particle liquid 10 was not prepared.
(1 o-c;光半導体粒子 1 0の調製) (1 o-c; Preparation of optical semiconductor particles 10)
混合液 1 0の全量から、 実施例 1 (1 _C) と同様にして、 光半導体粒子 1 0を24. O g得た。 この光半導体粒子 1 0は白色粉末であり、 珪素含有 量 5. 4重量%、 比表面積 1 90. 4m2/g、 表面積 1 m 2当りの珪素担持 量は 0. 28mg、 ナトリウム含有量は 1 50 p pmであった。 In the same manner as in Example 1 (1_C), 24.Og of photo semiconductor particles 10 was obtained from the total amount of the mixed solution 10. These optical semiconductor particles 10 are white powder, silicon content 5.4% by weight, specific surface area 190.4 m 2 / g, silicon loading per 1 m 2 surface area 0.28 mg, sodium content 1 It was 50 p pm.
[0085] (比較例 2) [0085] (Comparative Example 2)
(1 1 -A ;混合液 1 1の調製) (1 1 -A; Preparation of mixture 1 1)
混合液 3の 3分の 1量を取り分けて、 混合液 1 1 とした。 混合液 1 1は、 攪拌を止めて 30分放置した後にも、 上澄み液は白濁したままだった。 One third of the mixture 3 was divided into mixture 1 1. The mixture 11 remained cloudy even after stirring was stopped and left for 30 minutes.
(1 1 -B ;粗大粒子液 1 1の調製) (1 1 -B; Preparation of coarse particle liquid 1 1)
比較のために、 凝集剤を用いなかったので、 粗大粒子液 1 1は調製しなか つた。 For comparison, no flocculant was used, so the coarse particle liquid 11 was not prepared.
(1 1 -c;光半導体粒子 1 1の調製) (1 1 -c; Preparation of optical semiconductor particles 1 1)
混合液 1の代わりに、 混合液 1 1を用いて、 実施例 1 (1—C) と同様に して、 光半導体粒子 1 1を 40. 9 g得た。 この光半導体粒子 1 1は白色粉 末であり、 珪素含有量 6. 2重量%、 比表面積 21 8. 6m2/g、 表面積 1 m2当りの珪素担持量は 0. 28mg、 ナトリウム含有量は 1 80 p pmであ つた。 Instead of the mixed liquid 1, the mixed liquid 11 was used in the same manner as in Example 1 (1-C) to obtain 40.9 g of the optical semiconductor particles 11. This photo-semiconductor particle 11 is a white powder, with a silicon content of 6.2% by weight, a specific surface area of 218.6 m 2 / g, a silicon loading per 1 m 2 of surface area of 0.28 mg, and a sodium content of 1 80 p pm.
[0086] (比較例 3) ( 1 2-A ;混合液 1 2の調製) [0086] (Comparative Example 3) (1 2-A; Preparation of liquid mixture 1 2)
実施例 5 (5-A) と同様にして、 混合液 1 2を得た。 混合液 1 2は、 攪 拌を止めて 30分放置した後にも、 上澄み液は白濁したままだった。 In the same manner as in Example 5 (5-A), a mixed liquid 12 was obtained. Mixture 1 and 2 remained cloudy even after stirring was stopped for 30 minutes.
(1 2-B ;粗大粒子液 1 2の調製) (1 2-B; Preparation of coarse particle liquid 12)
比較のために、 凝集剤を用いなかったので、 粗大粒子液 1 2は調製しなか つた。 For comparison, no flocculant was used, so the coarse particle liquid 12 was not prepared.
(1 2-C ;光半導体粒子 1 2の調製) (1 2-C; Preparation of optical semiconductor particles 12)
混合液 1の代わりに、 混合液 1 2を用いて、 実施例 1 (1—C) と同様に して、 光半導体粒子 1 2を 1 9. 5 g得た。 この光半導体粒子 1 2は白色粉 末であり、 珪素含有量 4. 8重量%、 比表面積 1 86. 0m2/g、 表面積 1 m2当りの珪素担持量は 0. 26mg、 ナトリウム含有量 60 p p m、 力リウ ム含有量 90 p pmであった。 19.5 g of the optical semiconductor particles 1 2 was obtained in the same manner as in Example 1 (1-C) using the mixed liquid 12 instead of the mixed liquid 1. The optical semiconductor particles 1 2 is a white powder powder, silicon content 4.8 wt%, specific surface area 1 86. 0 m 2 / g, silicon supported amount per surface area of 1 m 2 is 0. 26 mg, sodium content 60 It was ppm and the rhodium content was 90 p pm.
[0087] (比較例 4) [0087] (Comparative Example 4)
( 1 3-A ;混合液 1 3の調製) (1 3-A; Preparation of liquid mixture 1 3)
実施例 6 (6-A) と同様にして、 混合液 1 3を得た。 混合液 1 3は、 攪 拌を止めて 30分放置した後にも、 上澄み液は白濁したままだった。 In the same manner as in Example 6 (6-A), a mixed solution 13 was obtained. Mixture 13 remained cloudy even after stirring was stopped and left for 30 minutes.
( 1 3-B ;粗大粒子液 1 3の調製) (1 3-B; Preparation of coarse particle liquid 1 3)
比較のために、 凝集剤を用いなかったので、 粗大粒子液 1 3は調製しなか つた。 For comparison, no flocculant was used, so the coarse particle liquid 13 was not prepared.
(1 3-C ;光半導体粒子 1 3の調製) (1 3-C; Preparation of optical semiconductor particles 1 3)
混合液 1の代わりに、 混合液 1 3を用いて、 実施例 1 (1—C) と同様に して、 光半導体粒子 1 3を 21. 1 g得た。 この光半導体粒子 1 3は白色粉 末であり、 珪素含有量 1. 4重量%、 比表面積 56. 4m2/g、 表面積 1 m 2当りの珪素担持量は 0. 22mg、 ナトリウム含有量 40 p pmであった。 Instead of the mixed liquid 1, the mixed liquid 13 was used in the same manner as in Example 1 (1-C) to obtain 21.1 g of the optical semiconductor particles 13. This photo-semiconductor particle 13 is a white powder with a silicon content of 1.4% by weight, a specific surface area of 56.4 m 2 / g, a silicon loading per 1 m 2 of surface area of 0.22 mg, and a sodium content of 40 p. It was pm.
[0088] (比較例 5) [0088] (Comparative Example 5)
( 1 4-A ;混合液 1 4の調製) (14-A; Preparation of mixture 14)
実施例 8 (8-A) と同様にして、 混合液 1 4を得た。 混合液 1 4は、 攪 拌を止めて 30分放置した後にも、 上澄み液は白濁したままだった。 (1 4-B ;粗大粒子液 1 4の調製) In the same manner as in Example 8 (8-A), a mixed solution 14 was obtained. Mixture 14 remained cloudy even after stirring was stopped for 30 minutes. (1 4-B; Preparation of coarse particle liquid 14)
比較のために、 凝集剤を用いなかったので、 粗大粒子液 1 4は調製しなか つた。 For comparison, no flocculant was used, so coarse particle liquid 14 was not prepared.
(1 4-C ;光半導体粒子 1 4の調製) (1 4-C; Preparation of photo-semiconductor particles 14)
混合液 1 4を、 1 0本の遠沈管に 1 O gずつ入れて、 各々、 以下の (a) 〜 (d) を 5回繰り返して洗浄を行った。 The mixed solution 14 was put into 10 centrifuge tubes at a rate of 1 O g, and each of the following (a) to (d) was repeated 5 times for washing.
(a) 遠心分離器 (コクサン社製、 H— 1 8) で、 1 0分間、 3000 r p mで処理して、 固形物を沈降させた、 (a) In a centrifuge (Kokusan Co., Ltd., H-1 8), it was treated at 3000 rpm for 10 minutes to precipitate solids.
(b) 上澄み液をデカント除去した、 (b) The supernatant was decanted away.
( c) 純水を 1 0 g加えた、 (c) 10 g of pure water was added,
(d) 超音波照射と振盪により、 固形物を分散させた。 (d) The solid was dispersed by ultrasonic irradiation and shaking.
次に、 1 0本の遠沈管の内容物を、 減圧ろ過して固形物を回収した。 回収 した白色固形物を、 磁性皿に入れ、 電気炉を用いて、 空気雰囲気下、 1 20 °Cで 3時間加熱した。 続けて 450°Cに昇温した後、 1 2時間 450°Cを保 持した。 加熱後、 室温になるまで放置して、 光半導体粒子 1 3を 1 0. 9 g 得た。 この光半導体粒子 1 3は、 白色粉末であり、 蛍光 X線分析より C aを 4. 1重量%、 Pを 1. 8重量%含んでいた。 Next, the contents of the 10 centrifuge tubes were filtered under reduced pressure to recover the solid matter. The recovered white solid was placed in a magnetic dish and heated at 120 ° C. for 3 hours in an air atmosphere using an electric furnace. The temperature was continuously raised to 450 ° C, and then maintained at 450 ° C for 12 hours. After heating, it was allowed to stand until it reached room temperature to obtain 10.9 g of optical semiconductor particles 13. This photo-semiconductor particle 13 was a white powder and contained 4.1% by weight of Ca and 1.8% by weight of P by fluorescent X-ray analysis.
(比較例 6) (Comparative Example 6)
( 1 5-A ;混合液 1 5) の調製) (Preparation of 1 5-A; Mixture 1 5)
実施例 9 (9-A) と同様にして、 混合液 1 5を得た。 混合液 1 5は、 攪 拌を止めて 30分放置した後にも、 上澄み液は白濁したままだった。 A liquid mixture 15 was obtained in the same manner as Example 9 (9-A). Mixture 15 remained cloudy even after stirring was stopped and left for 30 minutes.
( 1 5-B ;粗大粒子液 1 5の調製) (1 5-B; Preparation of coarse particle liquid 15)
比較のために、 凝集剤を用いなかったので、 粗大粒子液 1 5は調製しなか つた。 For comparison, no flocculant was used, so the coarse particle liquid 15 was not prepared.
(1 5-C ;光半導体粒子 1 5の調製) (1 5-C; Preparation of photo semiconductor particles 15)
粗大粒子液 1の代わりに、 混合液 1 5を全量用いて、 実施例 1 ( 1 _ C) と同様にして、 光半導体粒子 1 5を 1 3. 8 g得た。 この光半導体粒子 1 5 は白色粉末であり、 I CP分光分析より Mgを 2. 1重量%、 八 1 を1. 4重 量0 /o含んでいた。 Instead of the coarse particle liquid 1, 13.8 g of the photo-semiconductor particles 15 were obtained in the same manner as in Example 1 (1_C) by using the whole amount of the mixed liquid 15. This photo-semiconductor particle 15 is a white powder. Based on ICP spectroscopic analysis, Mg is 2.1% by weight, and 8 is 1.4. Contained 0 / o quantity.
[0090] (実施例 1 0) [0090] (Example 1 0)
< 調製品の評価 > <Evaluation of preparation>
( 1. 通液速度の測定) (1. Measurement of flow rate)
ろ過処理における通液速度を以下の方法で測定した。 粗大粒子液 1〜9並 びに混合液 1 0〜1 5を、 水で希釈すること、 上澄み液を一部除去すること 、 あるいは遠心分離器 (コクサン社製、 H— 1 8) で、 1 0分間、 3000 r pmで処理して固形物を沈降させた後に上澄み液の一部をデカント除去す ることのいずれかの方法によって、 スラリー濃度を 5重量%に調整した。 ガ ラス製減圧ろ過用フィルタ一ホルダ一 (アドバンテック東洋株式会社製、 K GS— 47、 ファンネル容積 1 1 OmL、 有効ろ過面積 9. 6 cm2) にメン プレンフィルタ一を挟み、 内部に 2 Om Lメスシリンダ一を設置した減圧容 器と組み合わせて、 減圧ろ過器具を準備した。 その際、 ろ液がメスシリンダ —に溜まるように、 ホルダ一ベースの足がメスシリンダ一の口に刺さつた状 態になるように設置した。 次にァスピレーターで減圧容器を減圧し、 フィル ター上部に、 試料液を 30mL注いだ。 そして、 ろ液が 5mL溜まった時点 を開始時刻として、 ろ液が 1 5 mL溜まるまでの時間、 すなわち 1 OmL通 液する時間を計測した。 測定結果は、 各試料液の調製における、 基体、 混合 液のスラリー濃度、 セラミックス原料、 並びに凝集剤使用量と併せて、 表 1 に示した。 The liquid passing speed in the filtration treatment was measured by the following method. Coarse particle liquid 1-9 and mixed liquid 10-15 are diluted with water, part of the supernatant liquid is removed, or a centrifuge (Hokusan, H-18) is used. The slurry concentration was adjusted to 5 wt% by either method of treating at 3000 rpm for 3 minutes to settle the solids and then decanting off a portion of the supernatant. A glass filter holder for vacuum filtration (Advantech Toyo Co., Ltd., K GS-47, funnel volume 1 1 OmL, effective filtration area 9.6 cm 2 ) with a membrane filter inside, 2 Om L inside A vacuum filtration device was prepared in combination with a vacuum container equipped with a measuring cylinder. At that time, it was installed so that the foot of the base of the holder pierced the mouth of the graduated cylinder so that the filtrate collected in the graduated cylinder. Next, the vacuum vessel was depressurized with an aspirator, and 30 mL of the sample solution was poured onto the top of the filter. Then, the time until 5 mL of filtrate was accumulated, that is, the time for 1 OmL to flow, was measured with the time when 5 mL of filtrate was accumulated as the start time. The measurement results are shown in Table 1 together with the substrate, the slurry concentration of the mixed solution, the ceramic raw material, and the amount of coagulant used in the preparation of each sample solution.
[0091] [0091]
ほ 1] 1
(2. 光半導体粒子の光分解性能評価) (2. Photodegradation performance evaluation of optical semiconductor particles)
光半導体粒子 1〜 1 5を、 メチレンブルー水溶液に懸濁させて光照射を行 し、、 液中のメチレンブルー濃度を分光分析で定量することにより、 光分解活 性を試験した。 試験操作方法は、 次のとおりである。 The photo-semiconductor particles 1 to 15 were suspended in a methylene blue aqueous solution, irradiated with light, and the methylene blue concentration in the liquid was quantified by spectroscopic analysis to test the photolysis activity. The test operation method is as follows.
(光半導体粒子懸濁液の調製) (Preparation of optical semiconductor particle suspension)
あらかじめフッ素樹脂製攪拌子を入れた 1 O O c cポリエチレン製広口び んに、 濃度 40 X 1 0-6mo I / Lのメチレンブル一水溶液を 45 g量り こんだ。 次に、 マグネチックスターラ一による攪拌下、 1 Omgの光半導体 粒子を加えた。 そして、 5分間激しく攪拌した後に、 液が飛び散らない程度 に攪拌強度を調整し、 攪拌を継続した。 45 g of methylene blue aqueous solution with a concentration of 40 X 10 0-6 mo I / L was weighed into a 1 O O c c polyethylene wide-mouthed bottle containing a fluororesin stirrer in advance. Next, 1 Omg of photo-semiconductor particles was added while stirring with a magnetic stirrer. Then, after stirring vigorously for 5 minutes, the stirring intensity was adjusted to such an extent that the liquid did not scatter and stirring was continued.
(予備吸着処理) (Preliminary adsorption treatment)
光半導体粒子を加え終わった瞬間を起点として、 60分間、 光照射せずに 、 攪拌し続けた。 60分経過後、 懸濁液を 3. O c c採取し、 光照射前サン プルとした。 (光分解処理) Starting from the moment the photo semiconductor particles were added, the stirring was continued for 60 minutes without light irradiation. After 60 minutes, 3. O cc of the suspension was collected and used as a sample before irradiation. (Photolytic treatment)
予備吸着処理後の懸濁液を 3. 5 c c抜き出し、 あらかじめフッ素樹脂製 攪拌子を入れた石英製標準分光セル (東ソ_ 'クォーツ株式会社、 外寸 1 2 . 5 X 1 2. 5 X 45 mm 光路幅 1 0 m m、 光路長 1 0 m m、 容積 4. 5 c c) に入れ、 マグネチックスターラーで攪拌した。 次に、 分光セルの外部 /横方向から光を 5分間照射した。 光照射は、 光源装置 S X_ U I 1 5 1 X Q (ゥシォ電機株式会社、 1 5 OWクセノンショートァ一クランプ) を光源 として、 純水を満たした石英製フィルタ一容器越しに行った。 照射光量は、 最高輝度の部分を、 紫外線照度計 U V D— 3 6 5 P D (ゥシォ電機株式会社 、 試験波長 3 6 5 n m ) で計測して、 5. 0 mW/ c m 2であった。 照射後、 分光セル内の懸濁液を回収し、 光照射後サンプルとした。 Standard suspension cell made of quartz (Tosoh_ Quartz Co., Ltd., outer dimensions 1 2.5 X 1 2.5 X 45 mm optical path width 10 mm, optical path length 10 mm, volume 4.5 cc) and stirred with a magnetic stirrer. Next, light was irradiated for 5 minutes from the outside / lateral direction of the spectroscopic cell. Light irradiation was performed through a quartz filter container filled with pure water using the light source device S X_UI 15 1 XQ (Usio Electric Co., Ltd., 15 OW Xenon short clamp) as a light source. The amount of irradiation light was 5.0 mW / cm 2 when the highest luminance part was measured with an ultraviolet illuminance meter UVD-3650 PD (Usio Electric Co., Ltd., test wavelength 36 5 nm). After irradiation, the suspension in the spectroscopic cell was collected and used as a sample after light irradiation.
[0093] (メチレンブルーの定量) [0093] (Quantification of methylene blue)
オールプラスチックス製 5 c cシリンジにメンブレンフィルタ一 (東洋濾 紙株式会社、 D I SM I C— 1 3 H P) を装着した。 これに、 光照射前後の サンプル懸濁液をそれぞれ入れ、 ビストンで押出して光半導体粒子を除去し た。 その際、 前半量のろ液は廃棄し、 後半量のろ液を、 可視光分析用セミマ イク口型ディスポセル (ポリスチレン製、 光路幅 4mm、 光路長 1 Omm、 容積 1 . 5 c c) に採取した。 そして、 紫外可視分光光度計 (U V— 2 55 0、 島津製作所) を使用して、 波長 6 80ナノメートルの吸光度を測定し、 メチレンブルー濃度を算定した。 なお、 前半量のろ液を廃棄するのは、 シリ ンジに装着したメンブレンフィルタ一に、 通液初期にメチレンブル一が吸着 して濃度が変化する現象の影響を回避するためであり、 0. 5m L程度通液 すれば、 吸着が飽和してサンプル液の濃度が正しく測定できるからである。 光分解活性は、 メチレンブルーの初濃度を基準として光照射前のメチレン ブル一濃度からメチレンブル一吸着率として、 そして光照射前のメチレンブ ルー濃度を基準として、 光照射後のメチレンブルー濃度からメチレンブルー 分解率として表 2に示した。 Membrane filter 1 (Toyo Roshi Kaisha, Ltd., DIMSIC—13 HP) was attached to an all plastic 5 cc syringe. The sample suspensions before and after the light irradiation were put into each, and extruded with Biston to remove the photo semiconductor particles. At that time, discard the first half of the filtrate and collect the latter half of the filtrate in a semi-micro mouth type disposable cell for analysis of visible light (made of polystyrene, optical path width 4 mm, optical path length 1 Omm, volume 1.5 cc). did. Then, using a UV-visible spectrophotometer (U V-2550, Shimadzu Corporation), the absorbance at a wavelength of 680 nanometers was measured, and the methylene blue concentration was calculated. The reason for discarding the first half of the filtrate is to avoid the influence of the phenomenon that the concentration of methylene blue is adsorbed on the membrane filter attached to the syringe and the concentration changes at the beginning of the flow. This is because, if about L is passed, the adsorption is saturated and the concentration of the sample solution can be measured correctly. The photodegradation activity is based on the initial concentration of methylene blue as the methylene blue concentration from the methylene blue concentration before the light irradiation, and as the methylene blue adsorption rate before the light irradiation as the methylene blue concentration after the light irradiation as the methylene blue decomposition rate. It is shown in Table 2.
[0094] (3. 細孔分布測定による酸化珪素膜由来の細孔有無の判定) オートソープ (カンタクローム社製) を使用し、 液体窒素下 (7 7 K ) に おける脱着過程での光半導体粒子 1〜7、 1 0〜1 3、 並びに酸化チタン S T - 0 1および P _ 2 5の窒素吸着等温線を測定した。 なお、 酸化チタンは 、 2 0 0 °C、 3時間焼成処理を施して、 乾燥したものを使用した。 [0094] (3. Determination of presence or absence of pores derived from silicon oxide film by pore distribution measurement) Photo-semiconductor particles 1-7, 10-1-13, and titanium oxide ST-01 and P_2 in the desorption process under liquid nitrogen (7 7 K) using autosoap (manufactured by Cantachrome) 5 nitrogen adsorption isotherms were measured. Titanium oxide was used after baking at 200 ° C. for 3 hours and drying.
まず、 前処理として 1 0 0 °Cでの真空脱気を行い、 次に各サンプルの窒素 吸着等温線を測定し、 結果を B J H法で解析することにより、 I o g微分細 孔容積分布曲線を求めた。 次に、 酸化珪素膜由来の細孔の有無を判定した。 具体的には、 光半導体粒子 1〜5は S T— 0 1 と、 光半導体粒子 6と 7は P 一 2 5と、 I o g微分細孔容積分布曲線を比較して、 酸化珪素膜由来の細孔 の有無を判定した。 図 1〜3に I o g微分細孔容積分布曲線を示す。 判定の 結果は、 表 2に示した。 First, as a pretreatment, vacuum degassing at 100 ° C was performed.Next, the nitrogen adsorption isotherm of each sample was measured, and the results were analyzed by the BJH method to obtain an Iog differential pore volume distribution curve. Asked. Next, the presence or absence of pores derived from the silicon oxide film was determined. Specifically, the photo-semiconductor particles 1 to 5 are ST-01, and the photo-semiconductor particles 6 and 7 are P 1 25, and the Iog differential pore volume distribution curves are compared. The presence or absence of holes was determined. Figures 1-3 show I og differential pore volume distribution curves. The results are shown in Table 2.
[0095] [表 2] [0095] [Table 2]
[0096] 表 1より、 凝集剤を用いた実施例 1〜 9においては、 凝集剤を用いない比 較例 1〜6と比較して、 通液時間が短くなつていることがわかる。 したがつ て、 本実施例の方法を用いた場合、 光半導体粒子の回収を短時間で行うこと ができる。 また、 表 2より、 凝集剤の有無に関わらず、 実施例および比較例 において同等の光半導体粒子の光分解性能が得られていることがわかる。 以 上より、 本発明の製造方法では、 固液分離が簡便になると共に、 光半導体粒 子の光半導体としての性能に影響を与えないことを示した。 [0096] From Table 1, in Examples 1 to 9 using the flocculant, the ratio without using the flocculant Compared with Comparative Examples 1 to 6, it can be seen that the passing time is shorter. Therefore, when the method of this example is used, the photo semiconductor particles can be collected in a short time. Table 2 also shows that the same photodegradation performance of the photo-semiconductor particles is obtained in the examples and comparative examples regardless of the presence or absence of the flocculant. From the above, it has been shown that the production method of the present invention facilitates solid-liquid separation and does not affect the performance of optical semiconductor particles as an optical semiconductor.
産業上の利用可能性 Industrial applicability
本発明の製造方法は、 金属化合物光半導体の表面にセラミックスを固定化 して性能改変を施した光半導体粒子を製造する場合に、 固液分離を簡便にす ると共に、 光半導体粒子の光半導体としての性能に影響を与えない。 そのた め、 化粧料、 顔料、 および光触媒等として用いられる光半導体粒子の製造に 利用できる。 The production method according to the present invention facilitates solid-liquid separation when producing optical semiconductor particles in which ceramics are immobilized on the surface of a metal compound optical semiconductor and the performance is modified. Does not affect the performance. Therefore, it can be used for the production of optical semiconductor particles used as cosmetics, pigments, photocatalysts and the like.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2006218631A JP2009256111A (en) | 2006-08-10 | 2006-08-10 | Method for producing photosemiconductor particle |
| JP2006-218631 | 2006-08-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2008018177A1 true WO2008018177A1 (en) | 2008-02-14 |
Family
ID=39032718
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/JP2007/000853 Ceased WO2008018177A1 (en) | 2006-08-10 | 2007-08-08 | Process for production of photosemiconductor particles |
Country Status (3)
| Country | Link |
|---|---|
| JP (1) | JP2009256111A (en) |
| TW (1) | TW200829334A (en) |
| WO (1) | WO2008018177A1 (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| TWI467784B (en) * | 2011-12-21 | 2015-01-01 | Univ Nat Cheng Kung | Solar cell |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH09206602A (en) * | 1995-02-03 | 1997-08-12 | Akira Fujishima | Fixing of photocatalyst |
| JP2001269573A (en) * | 2000-03-24 | 2001-10-02 | Titan Kogyo Kk | Photocatalyst particles, method for manufacturing the same and use of photocatalyst particles |
| JP2002136880A (en) * | 2000-11-01 | 2002-05-14 | Kanagawa Acad Of Sci & Technol | Photocatalyst and environmental purification method using the same |
| JP2003080078A (en) * | 2001-09-07 | 2003-03-18 | National Institute Of Advanced Industrial & Technology | Photoactive compounds and uses thereof |
| JP2003095805A (en) * | 2001-09-27 | 2003-04-03 | National Institute Of Advanced Industrial & Technology | Antibacterial material and antibacterial product using the same |
| JP2003327869A (en) * | 2002-05-13 | 2003-11-19 | Yoshiyuki Nagae | Coating, paint and method for producing coating |
| JP2004243307A (en) * | 2002-06-27 | 2004-09-02 | Showa Denko Kk | High activity photocatalyst particle, manufacturing method therefor and usage thereof |
| JP2005334737A (en) * | 2004-05-25 | 2005-12-08 | Futaba Shoji Kk | Magnetic adsorbent, photocatalyst supporting adsorbent, magnetic photocatalyst, photocatalyst supporting magnetic adsorbent, and harmful substance decomposing treatment method |
-
2006
- 2006-08-10 JP JP2006218631A patent/JP2009256111A/en active Pending
-
2007
- 2007-08-08 WO PCT/JP2007/000853 patent/WO2008018177A1/en not_active Ceased
- 2007-08-10 TW TW096129588A patent/TW200829334A/en unknown
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH09206602A (en) * | 1995-02-03 | 1997-08-12 | Akira Fujishima | Fixing of photocatalyst |
| JP2001269573A (en) * | 2000-03-24 | 2001-10-02 | Titan Kogyo Kk | Photocatalyst particles, method for manufacturing the same and use of photocatalyst particles |
| JP2002136880A (en) * | 2000-11-01 | 2002-05-14 | Kanagawa Acad Of Sci & Technol | Photocatalyst and environmental purification method using the same |
| JP2003080078A (en) * | 2001-09-07 | 2003-03-18 | National Institute Of Advanced Industrial & Technology | Photoactive compounds and uses thereof |
| JP2003095805A (en) * | 2001-09-27 | 2003-04-03 | National Institute Of Advanced Industrial & Technology | Antibacterial material and antibacterial product using the same |
| JP2003327869A (en) * | 2002-05-13 | 2003-11-19 | Yoshiyuki Nagae | Coating, paint and method for producing coating |
| JP2004243307A (en) * | 2002-06-27 | 2004-09-02 | Showa Denko Kk | High activity photocatalyst particle, manufacturing method therefor and usage thereof |
| JP2005334737A (en) * | 2004-05-25 | 2005-12-08 | Futaba Shoji Kk | Magnetic adsorbent, photocatalyst supporting adsorbent, magnetic photocatalyst, photocatalyst supporting magnetic adsorbent, and harmful substance decomposing treatment method |
Also Published As
| Publication number | Publication date |
|---|---|
| TW200829334A (en) | 2008-07-16 |
| JP2009256111A (en) | 2009-11-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102573926B (en) | Method for inactivating virus and article provided with antiviral properties | |
| Hassani et al. | Heterogeneous photocatalytic ozonation of ciprofloxacin using synthesized titanium dioxide nanoparticles on a montmorillonite support: parametric studies, mechanistic analysis and intermediates identification | |
| Stathatos et al. | TiO2/palygorskite composite nanocrystalline films prepared by surfactant templating route: Synergistic effect to the photocatalytic degradation of an azo-dye in water | |
| da Silva Lopes et al. | Modification of kaolinite from Pará/Brazil region applied in the anionic dye photocatalytic discoloration | |
| JPH09165218A (en) | Nanodispersible titanium dioxide, process for its production and its use | |
| Eddy et al. | Synthesis and photocatalytic activity of silica-based sand quartz as the supporting TiO2 photocatalyst | |
| UA104911C2 (en) | Stable nano titania sols and process for their producing | |
| WO2007125998A1 (en) | Photocatalyst, process for producing the photocatalyst, and photocatalyst coating agent, photocatalyst dispersion, and photocatalyst body using the photocatalyst | |
| JP2000053421A (en) | Titanium oxide sol and its preparation | |
| WO2007105705A1 (en) | Visible light-responsive photocatalyst, method for producing same, photocatalyst coating agent using same, and photocatalyst dispersion | |
| ES2385714T3 (en) | Titanium oxide product, method to prepare it and its use as a photocatalyst | |
| JP2004243307A (en) | High activity photocatalyst particle, manufacturing method therefor and usage thereof | |
| JP4686536B2 (en) | Photocatalyst, method for producing the same, dispersion containing photocatalyst, and photocatalyst coating composition | |
| WO2012067590A1 (en) | Coating of tio2 rutile nanoparticles in a suspension with hydrated sio2 and ai2o3 | |
| JP4540971B2 (en) | Neutral titanium oxide sol and method for producing the same | |
| KR20110064130A (en) | Phosphor-Titanium Dioxide Composite Nanoparticles Useful as Photocatalyst and Method for Preparing the Same | |
| JP4319184B2 (en) | PHOTOCATALYST, ITS MANUFACTURING METHOD, AND ARTICLE USING PHOTOCATALYST | |
| WO2008018177A1 (en) | Process for production of photosemiconductor particles | |
| Podbršček et al. | The preparation of zinc silicate/ZnO particles and their use as an efficient UV absorber | |
| JP2004161592A (en) | Anatase type titania-silica composite and its production method | |
| CN101374768B (en) | Method for recovering organic/inorganic elements-doped metal oxides from hydrolyzable metal compounds added as coagulants simultaneously with sewage treatment | |
| JP4522082B2 (en) | Photocatalyst liquid composition and photocatalyst formed using the same | |
| JP4316064B2 (en) | Method for producing titanium oxide photocatalyst fine particles | |
| US20090156394A1 (en) | Inorganic sintered material containing photocatalyst covered with silicon oxide film | |
| WO2008018178A1 (en) | Photocatalyst, method for producing the same, photocatalyst dispersion containing photocatalyst, and photocatalyst coating composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 07790341 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| NENP | Non-entry into the national phase |
Ref country code: RU |
|
| NENP | Non-entry into the national phase |
Ref country code: JP |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 07790341 Country of ref document: EP Kind code of ref document: A1 |