RU2002128352A - Производные 4,5-дигидро-1н-пиразола, обладающие cb1-антагонистической активностью - Google Patents
Производные 4,5-дигидро-1н-пиразола, обладающие cb1-антагонистической активностьюInfo
- Publication number
- RU2002128352A RU2002128352A RU2002128352/04A RU2002128352A RU2002128352A RU 2002128352 A RU2002128352 A RU 2002128352A RU 2002128352/04 A RU2002128352/04 A RU 2002128352/04A RU 2002128352 A RU2002128352 A RU 2002128352A RU 2002128352 A RU2002128352 A RU 2002128352A
- Authority
- RU
- Russia
- Prior art keywords
- compound
- formula
- iii
- group
- disease
- Prior art date
Links
- MCGBIXXDQFWVDW-UHFFFAOYSA-N 4,5-dihydro-1h-pyrazole Chemical class C1CC=NN1 MCGBIXXDQFWVDW-UHFFFAOYSA-N 0.000 title abstract 2
- 150000001875 compounds Chemical class 0.000 claims abstract 39
- 229910052739 hydrogen Inorganic materials 0.000 claims abstract 7
- 239000001257 hydrogen Substances 0.000 claims abstract 7
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims abstract 6
- 125000000217 alkyl group Chemical group 0.000 claims abstract 5
- 125000004076 pyridyl group Chemical group 0.000 claims abstract 5
- 125000001624 naphthyl group Chemical group 0.000 claims abstract 4
- 125000001544 thienyl group Chemical group 0.000 claims abstract 4
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 claims abstract 3
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 claims abstract 3
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 claims abstract 2
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims abstract 2
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims abstract 2
- 201000010099 disease Diseases 0.000 claims 5
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims 5
- 238000000034 method Methods 0.000 claims 5
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims 3
- 150000001412 amines Chemical class 0.000 claims 3
- 229930003827 cannabinoid Natural products 0.000 claims 3
- 239000003557 cannabinoid Substances 0.000 claims 3
- 238000006243 chemical reaction Methods 0.000 claims 3
- 229910052736 halogen Inorganic materials 0.000 claims 3
- 150000002367 halogens Chemical class 0.000 claims 3
- 150000002431 hydrogen Chemical class 0.000 claims 3
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims 3
- 230000005062 synaptic transmission Effects 0.000 claims 3
- -1 trifluoromethoxy, nitro, amino Chemical group 0.000 claims 3
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 claims 2
- 125000003545 alkoxy group Chemical group 0.000 claims 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims 2
- 125000003854 p-chlorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1Cl 0.000 claims 2
- 239000008194 pharmaceutical composition Substances 0.000 claims 2
- 125000006239 protecting group Chemical group 0.000 claims 2
- 150000003839 salts Chemical class 0.000 claims 2
- 125000001424 substituent group Chemical group 0.000 claims 2
- NWZSZGALRFJKBT-KNIFDHDWSA-N (2s)-2,6-diaminohexanoic acid;(2s)-2-hydroxybutanedioic acid Chemical compound OC(=O)[C@@H](O)CC(O)=O.NCCCC[C@H](N)C(O)=O NWZSZGALRFJKBT-KNIFDHDWSA-N 0.000 claims 1
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 claims 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 claims 1
- 208000024827 Alzheimer disease Diseases 0.000 claims 1
- 208000019901 Anxiety disease Diseases 0.000 claims 1
- 208000027559 Appetite disease Diseases 0.000 claims 1
- 208000024172 Cardiovascular disease Diseases 0.000 claims 1
- 206010012289 Dementia Diseases 0.000 claims 1
- 208000014094 Dystonic disease Diseases 0.000 claims 1
- 241001331845 Equus asinus x caballus Species 0.000 claims 1
- 208000019454 Feeding and Eating disease Diseases 0.000 claims 1
- 208000018522 Gastrointestinal disease Diseases 0.000 claims 1
- 208000023105 Huntington disease Diseases 0.000 claims 1
- 208000012902 Nervous system disease Diseases 0.000 claims 1
- 208000025966 Neurological disease Diseases 0.000 claims 1
- 208000008589 Obesity Diseases 0.000 claims 1
- 208000002193 Pain Diseases 0.000 claims 1
- 208000018737 Parkinson disease Diseases 0.000 claims 1
- 208000028017 Psychotic disease Diseases 0.000 claims 1
- 208000000323 Tourette Syndrome Diseases 0.000 claims 1
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 claims 1
- 125000002777 acetyl group Chemical group [H]C([H])([H])C(*)=O 0.000 claims 1
- 125000004390 alkyl sulfonyl group Chemical group 0.000 claims 1
- 125000003368 amide group Chemical group 0.000 claims 1
- 125000004397 aminosulfonyl group Chemical group NS(=O)(=O)* 0.000 claims 1
- 230000036506 anxiety Effects 0.000 claims 1
- 210000003169 central nervous system Anatomy 0.000 claims 1
- 239000003795 chemical substances by application Substances 0.000 claims 1
- 230000006735 deficit Effects 0.000 claims 1
- 208000010643 digestive system disease Diseases 0.000 claims 1
- 125000002147 dimethylamino group Chemical group [H]C([H])([H])N(*)C([H])([H])[H] 0.000 claims 1
- 208000010118 dystonia Diseases 0.000 claims 1
- 206010015037 epilepsy Diseases 0.000 claims 1
- 208000018685 gastrointestinal system disease Diseases 0.000 claims 1
- 125000005843 halogen group Chemical group 0.000 claims 1
- 230000002140 halogenating effect Effects 0.000 claims 1
- IKDUDTNKRLTJSI-UHFFFAOYSA-N hydrazine monohydrate Substances O.NN IKDUDTNKRLTJSI-UHFFFAOYSA-N 0.000 claims 1
- 208000028867 ischemia Diseases 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 230000001404 mediated effect Effects 0.000 claims 1
- 206010027175 memory impairment Diseases 0.000 claims 1
- 150000002730 mercury Chemical class 0.000 claims 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims 1
- 235000020824 obesity Nutrition 0.000 claims 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims 1
- 239000000651 prodrug Substances 0.000 claims 1
- 229940002612 prodrug Drugs 0.000 claims 1
- 208000020016 psychiatric disease Diseases 0.000 claims 1
- 208000011580 syndromic disease Diseases 0.000 claims 1
- 125000005034 trifluormethylthio group Chemical group FC(S*)(F)F 0.000 claims 1
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims 1
- 125000001889 triflyl group Chemical group FC(F)(F)S(*)(=O)=O 0.000 claims 1
- 125000006274 (C1-C3)alkoxy group Chemical group 0.000 abstract 1
- 102000009132 CB1 Cannabinoid Receptor Human genes 0.000 abstract 1
- 108010073366 CB1 Cannabinoid Receptor Proteins 0.000 abstract 1
- 239000005557 antagonist Substances 0.000 abstract 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 abstract 1
- 125000004356 hydroxy functional group Chemical group O* 0.000 abstract 1
- 240000004308 marijuana Species 0.000 abstract 1
- 230000003389 potentiating effect Effects 0.000 abstract 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/06—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/04—Centrally acting analgesics, e.g. opioids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/08—Antiepileptics; Anticonvulsants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/18—Antipsychotics, i.e. neuroleptics; Drugs for mania or schizophrenia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/22—Anxiolytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/24—Antidepressants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/04—Anorexiants; Antiobesity agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/06—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D231/08—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with oxygen or sulfur atoms directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biomedical Technology (AREA)
- Neurosurgery (AREA)
- Neurology (AREA)
- Psychiatry (AREA)
- Pain & Pain Management (AREA)
- Heart & Thoracic Surgery (AREA)
- Psychology (AREA)
- Hematology (AREA)
- Obesity (AREA)
- Cardiology (AREA)
- Diabetes (AREA)
- Hospice & Palliative Care (AREA)
- Urology & Nephrology (AREA)
- Child & Adolescent Psychology (AREA)
- Vascular Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Claims (16)
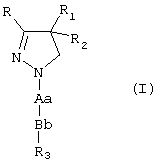
1. Соединение формулы (I)
где R и R1 могут быть одинаковыми или различными и обозначают фенил, тиенил или пиридил, причем указанные группы могут быть замещены 1, 2 или 3 заместителями Y, которые могут быть одинаковыми или различными, из группы, включающей С1-3-алкил или алкокси, гидрокси, галоген, трифторметил, трифторметилтио, трифторметокси, нитро, амино, моно- или диалкил (С1-2) амино, моно- или диалкил (Сl-2) амидо, (С1-3) алкилсульфонил, диметилсульфамидо, С1-3-алкоксикарбонил, карбоксил, трифторметилсульфонил, циано, карбамоил, сульфамоил и ацетил, или R и/или R1, обозначают нафтил;
R2 обозначает водород, гидрокси, C1-3-алкокси, ацетилокси или пропионилокси;
Аа обозначает одну из групп (i), (ii), (iii), (iv) или (v)
где R4 и R5 независимо друг от друга обозначают водород или C1-8 разветвленный или неразветвленный алкил, или С3-8 циклоалкил или R4 обозначает ацетамидо или диметиламино, или 2,2,2-трифторэтил, или фенил, или пиридил, при условии, что R6 обозначает водород, R6 обозначает водород или C1-3 неразветвленный алкил;
Bb обозначает сульфонил или карбонил;
R3 обозначает бензил, фенил, тиенил или пиридил, которые могут быть замещены 1, 2 или 3 заместителями Y, которые могут быть одинаковыми или различными, или R3 обозначает C1-8 разветвленный или неразветвленный алкил или С3-8 циклоалкил, или R3 обозначает нафтил,
и его таутомеры, пролекарства и соли.
2. Соединение формулы (I) по п.1, отличающееся тем, что R обозначает 4-хлорфенильную группу, R1 обозначает фенил, R2 обозначает водород, Аа обозначает группу (i), в которой R4 обозначает водород и R5 обозначает метил, Bb обозначает сульфонил и R3 обозначает 4-хлорфенил, и его соли.
3. Фармацевтическая композиция, включающая по меньшей мере одно соединение по п.1 в качестве активного компонента.
4. Способ получения фармацевтических композиций, отличающийся тем, что соединение по п.1 переводят в форму, подходящую для введения.
5. Способ получения соединения общей формулы I, отличающийся тем, что a) получают соединение, в котором R, R1-R3 и Bb имеют значения, указанные в п.1, и Аа обозначает группу формулы (i) или (ii) по п.1, посредством 1) реакции соединения формулы (II) с гидразином или гидратом гидразина с получением соединения формулы (III), которое подвергают реакции с соединением формулы (IVa) или (IVb) с получением соединения формулы (V), которое подвергают реакции с соединением формулы R3-SO2X или R3-COX, где Х обозначает галоген, или 2) реакции соединения формулы (III) с тиоизоцианатом формулы (VI) с получением соединения формулы (VII), которое подвергают реакции с амином в присутствии соли ртути (II), или 3) реакции соединения формулы (III) с соединением формулы (VIII) с получением соединения формулы (IX), которое подвергают реакции с галогенирующим агентом с получением соединения формулы (X), которое подвергают реакции с амином, или 4) реакции соединения формулы (III) с соединением формулы (XI) с получением соединения формулы (XII), которое подвергают реакции с амином, или b) получают соединение, в котором R, R1-R3 и Bb имеют значения, указанные в п.1, и Аа обозначает группу формулы (iii) или (iv) по п.1, посредством реакции соединения формулы (III) с соединением формулы (XIII) или (XIV), или с) получают соединение, в котором R, R1-R3 и Bb имеют значения, указанные в п.1, и Аа обозначает группу формулы (v) по п.1, посредством реакции соединения формулы (III) с соединением формулы (XV) или (XVI) с получением соединения формулы (XVII), от которого удаляют защитную группу с получением соединения формулы (V), которое подвергают реакции с соединением формулы R3-SO2Х или R3-COX, где Х обозначает галоген, или с соединением формулы R3-COOH.
13. Способ лечения заболеваний, опосредованных CB1-рецептором, включающий введение пациенту эффективного количества соединения по п.1.
14. Способ по п.13, отличающийся тем, что заболеванием являются психиатрические заболевания, такие как психоз, беспокойство, депрессия, дефицит внимания, нарушения памяти и расстройства аппетита, ожирение, неврологические заболевания, такие как болезнь Паркинсона, деменция, дистония, болезнь Альцгеймера, эпилепсия, болезнь Хантингтона, болезнь Туретта, ишемия, болевые синдромы и другие заболевания ЦНС, включающие каннабиноидную нейротрансмиссию.
15. Способ по п.13, отличающийся тем, что заболеванием является желудочно-кишечные расстройства, включающие каннабиноидную нейротрансмиссию.
16. Способ по п.13, отличающийся тем, что заболеванием является сердечно-сосудистые заболевания, включающие каннабиноидную нейротрансмиссию.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| NL1014728 | 2000-03-23 | ||
| EP00201032 | 2000-03-23 | ||
| NL1014728 | 2000-03-23 | ||
| EP00201032.0 | 2000-03-23 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| RU2002128352A true RU2002128352A (ru) | 2004-02-27 |
| RU2245878C2 RU2245878C2 (ru) | 2005-02-10 |
Family
ID=26072029
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| RU2002128352/04A RU2245878C2 (ru) | 2000-03-23 | 2001-03-22 | Производные 4,5-дигидро-1н-пиразола, обладающие cb1-антагонистической активностью |
Country Status (26)
| Country | Link |
|---|---|
| US (1) | US6476060B2 (ru) |
| EP (1) | EP1268435B1 (ru) |
| JP (1) | JP2004500401A (ru) |
| KR (1) | KR100800277B1 (ru) |
| CN (1) | CN1205188C (ru) |
| AT (1) | ATE346047T1 (ru) |
| AU (2) | AU4250101A (ru) |
| BR (1) | BR0109457A (ru) |
| CA (1) | CA2401832C (ru) |
| CY (1) | CY1105967T1 (ru) |
| DE (1) | DE60124685T2 (ru) |
| DK (1) | DK1268435T3 (ru) |
| DZ (1) | DZ3335A1 (ru) |
| ES (1) | ES2272449T3 (ru) |
| HK (1) | HK1052349B (ru) |
| HU (1) | HUP0204519A3 (ru) |
| IL (2) | IL151452A0 (ru) |
| MX (1) | MXPA02009258A (ru) |
| NO (1) | NO324173B1 (ru) |
| PL (1) | PL208098B1 (ru) |
| PT (1) | PT1268435E (ru) |
| RU (1) | RU2245878C2 (ru) |
| SI (1) | SI1268435T1 (ru) |
| SK (1) | SK287074B6 (ru) |
| UA (1) | UA74367C2 (ru) |
| WO (1) | WO2001070700A1 (ru) |
Families Citing this family (132)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100366614C (zh) * | 2001-03-22 | 2008-02-06 | 索尔瓦药物有限公司 | 具有cb1-拮抗活性的4,5-二氢-1h-吡唑衍生物 |
| US20050054730A1 (en) * | 2001-03-27 | 2005-03-10 | The Regents Of The University Of California | Compounds, compositions and treatment of oleoylethanolamide-like modulators of PPARalpha |
| TWI231757B (en) | 2001-09-21 | 2005-05-01 | Solvay Pharm Bv | 1H-Imidazole derivatives having CB1 agonistic, CB1 partial agonistic or CB1-antagonistic activity |
| PL368441A1 (en) * | 2001-09-21 | 2005-03-21 | Solvay Pharmaceuticals B.V. | Novel 4,5-dihydro-1h-pyrazole derivatives having cb1-antagonistic activity |
| HRP20030913A2 (en) * | 2001-09-21 | 2004-06-30 | Solvay Pharm Bv | 4,5-dihydro-1h-pyrazole derivatives having potent cb1-antagonistic activity |
| CA2478338A1 (en) | 2002-03-08 | 2003-09-18 | Signal Pharmaceuticals, Inc. | Combination therapy for treating, preventing or managing proliferative disorders and cancers |
| NZ534757A (en) | 2002-03-12 | 2006-07-28 | Merck & Co Inc | Substituted amides |
| US6825209B2 (en) | 2002-04-15 | 2004-11-30 | Research Triangle Institute | Compounds having unique CB1 receptor binding selectivity and methods for their production and use |
| GB0216700D0 (en) | 2002-07-18 | 2002-08-28 | Astrazeneca Ab | Process |
| ES2301833T3 (es) * | 2002-07-29 | 2008-07-01 | Hoffmann La Roche | Derivados de benzodioxol. |
| US7765162B2 (en) * | 2002-10-07 | 2010-07-27 | Mastercard International Incorporated | Method and system for conducting off-line and on-line pre-authorized payment transactions |
| JP2006506366A (ja) * | 2002-10-18 | 2006-02-23 | ファイザー・プロダクツ・インク | カンナビノイド受容体リガンドおよびその使用方法 |
| US7129239B2 (en) * | 2002-10-28 | 2006-10-31 | Pfizer Inc. | Purine compounds and uses thereof |
| US7247628B2 (en) * | 2002-12-12 | 2007-07-24 | Pfizer, Inc. | Cannabinoid receptor ligands and uses thereof |
| ATE450505T1 (de) | 2003-01-02 | 2009-12-15 | Hoffmann La Roche | Cb 1 rezeptor inversagonisten |
| ATE400571T1 (de) | 2003-01-02 | 2008-07-15 | Hoffmann La Roche | Pyrrolyl-thiazole und ihre verwendung als inverse agonisten des cb 1 rezeptors |
| US7772188B2 (en) | 2003-01-28 | 2010-08-10 | Ironwood Pharmaceuticals, Inc. | Methods and compositions for the treatment of gastrointestinal disorders |
| US7329658B2 (en) * | 2003-02-06 | 2008-02-12 | Pfizer Inc | Cannabinoid receptor ligands and uses thereof |
| GB0302673D0 (en) | 2003-02-06 | 2003-03-12 | Astrazeneca Ab | Pharmaceutical formulations |
| US7176210B2 (en) * | 2003-02-10 | 2007-02-13 | Pfizer Inc. | Cannabinoid receptor ligands and uses thereof |
| US7268133B2 (en) * | 2003-04-23 | 2007-09-11 | Pfizer, Inc. Patent Department | Cannabinoid receptor ligands and uses thereof |
| US20040214856A1 (en) * | 2003-04-23 | 2004-10-28 | Pfizer Inc | Cannabinoid receptor ligands and uses thereof |
| US7141669B2 (en) * | 2003-04-23 | 2006-11-28 | Pfizer Inc. | Cannabiniod receptor ligands and uses thereof |
| US7145012B2 (en) | 2003-04-23 | 2006-12-05 | Pfizer Inc. | Cannabinoid receptor ligands and uses thereof |
| MXPA05011922A (es) * | 2003-05-07 | 2006-02-17 | Pfizer Prod Inc | Ligandos del receptor de cannabinoides y sus usos. |
| US7232823B2 (en) | 2003-06-09 | 2007-06-19 | Pfizer, Inc. | Cannabinoid receptor ligands and uses thereof |
| GB0314049D0 (en) * | 2003-06-18 | 2003-07-23 | Astrazeneca Ab | Therapeutic agents |
| US20040259887A1 (en) * | 2003-06-18 | 2004-12-23 | Pfizer Inc | Cannabinoid receptor ligands and uses thereof |
| GB0314057D0 (en) * | 2003-06-18 | 2003-07-23 | Astrazeneca Ab | Therapeutic agents |
| US20060135523A1 (en) * | 2003-06-18 | 2006-06-22 | Astrazeneca Ab | 2-substituted 5,6-diaryl-pyrazine derivatives as cb1 modulator |
| GB0314261D0 (en) * | 2003-06-19 | 2003-07-23 | Astrazeneca Ab | Therapeutic agents |
| ATE390922T1 (de) | 2003-06-20 | 2008-04-15 | Hoffmann La Roche | 2-aminobenzothiazole als cb1 rezeptor inverse agonisten |
| AR045533A1 (es) * | 2003-09-02 | 2005-11-02 | Solvay Pharm Gmbh | Uso de un compuesto antagonista de receptor de cb1, composicion farmaceutica y metodo de tratamiento y/o profilaxis de enfermedades relacionadas con dicho receptor de cb1 |
| RU2006110545A (ru) * | 2003-09-02 | 2007-10-10 | Зольвай Фармасьютикалз Гмбх (De) | Новое медицинское применение избирательных антагонистов св1-рецептора |
| US20050239859A2 (en) * | 2003-09-03 | 2005-10-27 | Solvay Pharmaceuticals Gmbh | Novel medical uses of 4,5-dihydro-1h-pyrazole derivatives having cb1- antagonistic activity |
| TW200528102A (en) * | 2003-10-24 | 2005-09-01 | Solvay Pharm Gmbh | Novel medical combination treatment of obesity involving 4,5-dihydro-1h-pyrazole derivatives having cb1-antagonistic activity |
| US20050143441A1 (en) * | 2003-10-27 | 2005-06-30 | Jochen Antel | Novel medical combination treatment of obesity involving 4,5-dihydro-1H-pyrazole derivatives having CB1-antagonistic activity |
| US20050124660A1 (en) * | 2003-10-27 | 2005-06-09 | Jochen Antel | Novel medical uses of compounds showing CB1-antagonistic activity and combination treatment involving said compounds |
| GB0327331D0 (en) * | 2003-11-25 | 2003-12-31 | Astrazeneca Ab | Therapeutic agents |
| CN100509808C (zh) | 2003-12-08 | 2009-07-08 | 霍夫曼-拉罗奇有限公司 | 新型噻唑衍生物 |
| BRPI0507120A (pt) | 2004-01-28 | 2007-06-19 | Hoffmann La Roche | compostos, processo para a manufatura dos mesmos, composições farmacêuticas que os compreendem, método para o tratamento e/ou profilaxia de enfermidades que estão associadas com a modulação de receptores de cb1 e sua utilização |
| US7745476B2 (en) | 2004-01-30 | 2010-06-29 | Solvay Pharmaceuticals B.V. | 1,3,5-trisubstituted 4,5-dihydro-1H-pyrazole derivatives having CB1-antagonistic activity |
| DE602005008555D1 (de) * | 2004-01-30 | 2008-09-11 | Solvay Pharm Bv | 1,3,5-trisubstituierte 4,5-dihydro-1h-pyrazol-derivative mit cb1-antagonistischer aktivität |
| TW200533657A (en) | 2004-02-17 | 2005-10-16 | Esteve Labor Dr | Substituted pyrazoline compounds, their preparation and use as medicaments |
| GB0403780D0 (en) * | 2004-02-20 | 2004-03-24 | Astrazeneca Ab | Therapeutic agents |
| EP1734963A4 (en) | 2004-04-02 | 2008-06-18 | Merck & Co Inc | METHOD FOR TREATING PEOPLE WITH METABOLIC AND ANTHROPOMETRIC DISORDER |
| JP4177435B2 (ja) | 2004-04-03 | 2008-11-05 | アストラゼネカ アクチボラグ | 治療薬 |
| ATE432275T1 (de) | 2004-05-10 | 2009-06-15 | Hoffmann La Roche | Pyrrol- bzw. imidazolamide zur behandlung von obesitas |
| ITMI20041033A1 (it) | 2004-05-24 | 2004-08-24 | Neuroscienze S C A R L | Composti farmaceutici |
| ITMI20041032A1 (it) * | 2004-05-24 | 2004-08-24 | Neuroscienze S C A R L | Compositi farmaceutici |
| TW200602314A (en) | 2004-05-28 | 2006-01-16 | Tanabe Seiyaku Co | A novel pyrrolidine compound and a process for preparing the same |
| US20060025448A1 (en) | 2004-07-22 | 2006-02-02 | Cadila Healthcare Limited | Hair growth stimulators |
| CA2581596A1 (en) * | 2004-09-29 | 2006-04-13 | Schering Corporation | Combinations of substituted azetidonones and cb1 antagonists |
| WO2006045799A2 (en) * | 2004-10-25 | 2006-05-04 | Solvay Pharmaceuticals Gmbh | Pharmaceutical compositions comprising cb1 cannabinoid receptor antagonists and potassium channel openers for the treatment of diabetes mellitus type i, obesity and related conditions |
| JP2008517959A (ja) | 2004-10-27 | 2008-05-29 | エフ.ホフマン−ラ ロシュ アーゲー | 新規なインドール又はベンゾイミダゾール誘導体 |
| JP4675969B2 (ja) | 2004-11-09 | 2011-04-27 | エフ.ホフマン−ラ ロシュ アーゲー | ジベンゾスベロン誘導体 |
| WO2006060186A2 (en) * | 2004-11-30 | 2006-06-08 | Bayer Pharmaceuticals Corporation | Pyrazole derivatives for the treatment of dementia and related disorders |
| WO2006060192A2 (en) * | 2004-11-30 | 2006-06-08 | Bayer Pharmaceuticals Corporation | Pyrazole derivatives |
| WO2006060201A2 (en) * | 2004-11-30 | 2006-06-08 | Bayer Pharmaceuticals Corporation | Pyrazole derivatives for the treatment of psychiatric disorders |
| WO2006060211A2 (en) * | 2004-11-30 | 2006-06-08 | Bayer Pharmaceuticals Corporation | Pyrazole derivatives for the treatment of sexual dysfunction |
| ES2326280T3 (es) | 2005-04-06 | 2009-10-06 | F. Hoffmann-La Roche Ag | Derivados de piridin-3-carboxamida como agonistas inversos de cb1. |
| JP2008542255A (ja) * | 2005-05-27 | 2008-11-27 | ファイザー・プロダクツ・インク | 肥満症の治療または体重減量の維持のためのカンナビノイド−1受容体アンタゴニストおよびミクロソームトリグリセリド輸送タンパク質阻害物質の併用 |
| JP2008545739A (ja) | 2005-06-02 | 2008-12-18 | グレンマーク・ファーマシューティカルズ・エスエー | 新規なカンナビノイド受容体リガンド、それらを含む薬剤組成物、およびそれらの調製方法 |
| US7923465B2 (en) | 2005-06-02 | 2011-04-12 | Glenmark Pharmaceuticals S.A. | Cannabinoid receptor ligands, pharmaceutical compositions containing them, and process for their preparation |
| ES2326460B1 (es) * | 2005-07-15 | 2010-04-19 | Laboratorios Del Dr. Esteve, S.A. | Sales de amonio cuaternario de compuestos de pirazolina sustituidos, su preparacion y uso como medicamentos. |
| WO2007009704A2 (en) * | 2005-07-15 | 2007-01-25 | Laboratorios Del Dr. Esteve, S.A. | Use of substituted pyrazoline compounds for the treatment of food disorders, including obesity or metabolic syndrome in patients with developed diabetes |
| EP1749820A1 (en) * | 2005-07-15 | 2007-02-07 | Laboratorios Del Dr. Esteve, S.A. | Salts of substituted pyrazoline compounds, their preparation and use as medicaments |
| EP1910300A2 (en) * | 2005-07-15 | 2008-04-16 | Laboratorios del Dr. Esteve S.A. | Prodrugs of pyrazoline compounds, their preparation and use as medicaments |
| EP1749526A1 (en) * | 2005-07-15 | 2007-02-07 | Laboratorios Del Dr. Esteve, S.A. | Use of substituted pyrazoline compounds for the treatment of food disorders, including obesity or metabolic syndrome in patients with developed diabetes |
| EP1743890A1 (en) * | 2005-07-15 | 2007-01-17 | Laboratorios Del Dr. Esteve, S.A. | 4,5-Dihydro-1H-pyrazole derivatives, their preparation and use as medicaments |
| EP1749821A1 (en) * | 2005-07-15 | 2007-02-07 | Laboratorios Del Dr. Esteve, S.A. | Quaternary ammonium salts of substituted pyrazoline compounds, their preparation and use as medicaments |
| EP1743892A1 (en) | 2005-07-15 | 2007-01-17 | Laboratorios del Dr. Esteve S.A. | Substituted pyrazoline compounds, their preparation and use as medicaments |
| ES2330992B1 (es) * | 2005-07-15 | 2010-07-06 | Laboratorios Del Dr. Esteve, S.A. | Compuestos de pirazolina heterociclilsustituidos, su preparacion y uso como medicamentos. |
| US20070191357A1 (en) * | 2005-08-17 | 2007-08-16 | Jochen Antel | Methods of using potassium channel inhibiting compounds |
| WO2007040238A1 (ja) | 2005-10-04 | 2007-04-12 | Mitsubishi Tanabe Pharma Corporation | 光学活性4-ヒドロキシ-1,2,3,4-テトラヒドロキノリン化合物の製法 |
| AR056560A1 (es) | 2005-10-06 | 2007-10-10 | Astrazeneca Ab | Pirrolopiridinonas como moduladores cb1 |
| AU2006328483A1 (en) * | 2005-12-20 | 2007-06-28 | Solvay Pharmaceuticals B.V. | 4,5-dihydro- (1H)-pyrazole derivatives as cannabinoid CB1 receptor modulators |
| US20070254863A1 (en) * | 2006-04-27 | 2007-11-01 | Jochen Antel | Use of CBx cannabinoid receptor modulators as potassium channel modulators |
| US7763607B2 (en) | 2006-04-27 | 2010-07-27 | Solvay Pharmaceuticals Gmbh | Pharmaceutical compositions comprising CBx cannabinoid receptor modulators and potassium channel modulators |
| AU2007245733A1 (en) * | 2006-04-27 | 2007-11-08 | Solvay Pharmaceuticals Gmbh | Pharmaceutical compositions comprising CBX cannabinoid receptor modulators and Potassium channel modulators |
| ES2389569T3 (es) * | 2006-05-05 | 2012-10-29 | Jenrin Discovery | Antagonistas/agonistas inversos de receptores de cannabinoides |
| US7629346B2 (en) | 2006-06-19 | 2009-12-08 | Hoffmann-La Roche Inc. | Pyrazinecarboxamide derivatives as CB1 antagonists |
| US7964728B2 (en) * | 2006-07-06 | 2011-06-21 | Solvay Pharmaceuticals B.V. | Azaindole derivatives with a combination of partial nicotinic acetyl-choline receptor agonism and dopamine reuptake inhibition |
| JP2010500300A (ja) | 2006-08-08 | 2010-01-07 | サノフィ−アベンティス | アリールアミノアリール−アルキル−置換イミダゾリジン−2,4−ジオン、それらの製造法、それらの化合物を含有する薬剤、およびそれらの使用 |
| US7781593B2 (en) | 2006-09-14 | 2010-08-24 | Hoffmann-La Roche Inc. | 5-phenyl-nicotinamide derivatives |
| AR062907A1 (es) * | 2006-09-22 | 2008-12-17 | Solvay Pharm Bv | Derivados de sulfonilpirazol y sulfonilpirazolincarboxamidina como antagonistas de 5-ht6 |
| NZ578063A (en) * | 2006-12-18 | 2011-09-30 | 7Tm Pharma As | Cannabinoid CB1 receptor modulators |
| US8969514B2 (en) | 2007-06-04 | 2015-03-03 | Synergy Pharmaceuticals, Inc. | Agonists of guanylate cyclase useful for the treatment of hypercholesterolemia, atherosclerosis, coronary heart disease, gallstone, obesity and other cardiovascular diseases |
| WO2008151257A2 (en) | 2007-06-04 | 2008-12-11 | Synergy Pharmaceuticals Inc. | Agonists of guanylate cyclase useful for the treatment of gastrointestinal disorders, inflammation, cancer and other disorders |
| EP2025674A1 (de) | 2007-08-15 | 2009-02-18 | sanofi-aventis | Substituierte Tetrahydronaphthaline, Verfahren zu ihrer Herstellung und ihre Verwendung als Arzneimittel |
| US7655685B2 (en) * | 2007-11-02 | 2010-02-02 | Jenrin Discovery, Inc. | Cannabinoid receptor antagonists/inverse agonists useful for treating metabolic disorders, including obesity and diabetes |
| WO2010003624A2 (en) | 2008-07-09 | 2010-01-14 | Sanofi-Aventis | Heterocyclic compounds, processes for their preparation, medicaments comprising these compounds, and the use thereof |
| CA2730603C (en) | 2008-07-16 | 2019-09-24 | Synergy Pharmaceuticals Inc. | Agonists of guanylate cyclase useful for the treatment of gastrointestinal disorders, inflammation, cancer and other disorders |
| EP2151234A1 (en) * | 2008-07-28 | 2010-02-10 | Laboratorios Del. Dr. Esteve, S.A. | Pharmaceutical formulation comprising a CB1-receptor compound in a solid solution and/or solid dispersion |
| AR072539A1 (es) * | 2008-08-01 | 2010-09-01 | Solvay Pharm Bv | Sintesis de derivados de 3,4-diaril -4,5-dihidro -(1h) - pirazol -1- carboxamida |
| CA2741125A1 (en) | 2008-10-22 | 2010-04-29 | Merck Sharp & Dohme Corp. | Novel cyclic benzimidazole derivatives useful anti-diabetic agents |
| WO2010051206A1 (en) | 2008-10-31 | 2010-05-06 | Merck Sharp & Dohme Corp. | Novel cyclic benzimidazole derivatives useful anti-diabetic agents |
| WO2010068601A1 (en) | 2008-12-08 | 2010-06-17 | Sanofi-Aventis | A crystalline heteroaromatic fluoroglycoside hydrate, processes for making, methods of use and pharmaceutical compositions thereof |
| WO2010079241A1 (es) | 2009-01-12 | 2010-07-15 | Fundacion Hospital Nacional De Paraplejicos Para La Investigacion Y La Integracion | Uso de antagonistas y/o agonistas inversos de los receptores cb1 para la preparación de medicamentos que incrementen la excitabilidad de las motoneuronas |
| KR20120060207A (ko) | 2009-08-26 | 2012-06-11 | 사노피 | 신규한 결정성 헤테로방향족 플루오로글리코시드 수화물, 이들 화합물을 포함하는 약제 및 이들의 용도 |
| WO2011044370A1 (en) | 2009-10-07 | 2011-04-14 | Jenrin Discovery | Cannabinoid receptor antagonists/inverse agonists useful for treating metabolic disorders, including obesity and diabetes |
| US8895596B2 (en) | 2010-02-25 | 2014-11-25 | Merck Sharp & Dohme Corp | Cyclic benzimidazole derivatives useful as anti-diabetic agents |
| WO2011157827A1 (de) | 2010-06-18 | 2011-12-22 | Sanofi | Azolopyridin-3-on-derivate als inhibitoren von lipasen und phospholipasen |
| US9616097B2 (en) | 2010-09-15 | 2017-04-11 | Synergy Pharmaceuticals, Inc. | Formulations of guanylate cyclase C agonists and methods of use |
| US8410107B2 (en) | 2010-10-15 | 2013-04-02 | Hoffmann-La Roche Inc. | N-pyridin-3-yl or N-pyrazin-2-yl carboxamides |
| WO2012068529A2 (en) | 2010-11-18 | 2012-05-24 | Jenrin Discovery | Cannabinoid receptor antagonists-inverse agonists useful for treating metabolic disorders, including obesity and diabetes |
| US8669254B2 (en) | 2010-12-15 | 2014-03-11 | Hoffman-La Roche Inc. | Pyridine, pyridazine, pyrimidine or pyrazine carboxamides as HDL-cholesterol raising agents |
| BR112013021236B1 (pt) | 2011-02-25 | 2021-05-25 | Merck Sharp & Dohme Corp | composto derivado de benzimidazol, e, composição |
| WO2012120057A1 (de) | 2011-03-08 | 2012-09-13 | Sanofi | Neue substituierte phenyl-oxathiazinderivate, verfahren zu deren herstellung, diese verbindungen enthaltende arzneimittel und deren verwendung |
| WO2012120055A1 (de) | 2011-03-08 | 2012-09-13 | Sanofi | Di- und trisubstituierte oxathiazinderivate, verfahren zu deren herstellung, ihre verwendung als medikament sowie sie enthaltendes arzneimittel und deren verwendung |
| EP2683702B1 (de) | 2011-03-08 | 2014-12-24 | Sanofi | Neue substituierte phenyl-oxathiazinderivate, verfahren zu deren herstellung, diese verbindungen enthaltende arzneimittel und deren verwendung |
| EP2683701B1 (de) | 2011-03-08 | 2014-12-24 | Sanofi | Mit benzyl- oder heteromethylengruppen substituierte oxathiazinderivate, verfahren zu deren herstellung, ihre verwendung als medikament sowie sie enthaltendes arzneimittel und deren verwendung |
| US8828995B2 (en) | 2011-03-08 | 2014-09-09 | Sanofi | Branched oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120056A1 (de) | 2011-03-08 | 2012-09-13 | Sanofi | Tetrasubstituierte oxathiazinderivate, verfahren zu deren herstellung, ihre verwendung als medikament sowie sie enthaltendes arzneimittel und deren verwendung |
| EP2766349B1 (de) | 2011-03-08 | 2016-06-01 | Sanofi | Mit carbozyklen oder heterozyklen substituierte oxathiazinderivate, verfahren zu deren herstellung, diese verbindungen enthaltende arzneimittel und deren verwendung |
| US8828994B2 (en) | 2011-03-08 | 2014-09-09 | Sanofi | Di- and tri-substituted oxathiazine derivatives, method for the production thereof, use thereof as medicine and drug containing said derivatives and use thereof |
| WO2012120051A1 (de) | 2011-03-08 | 2012-09-13 | Sanofi | Mit adamantan- oder noradamantan substituierte benzyl-oxathiazinderivate, diese verbindungen enthaltende arzneimittel und deren verwendung |
| US8680131B2 (en) | 2012-07-25 | 2014-03-25 | Jenrin Discovery, Inc. | Cannabinoid receptor antagonists/inverse agonists useful for treating disease conditions, including metabolic disorders and cancers |
| NZ703992A (en) | 2012-08-01 | 2018-04-27 | Lewis And Clark Pharmaceuticals Inc | N-alkyl 2-(disubstituted)alkynyladenosine-5’-uronamides as a2a agonists |
| US9527875B2 (en) | 2012-08-02 | 2016-12-27 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| US11155521B2 (en) | 2012-11-13 | 2021-10-26 | The United States Of America As Represented By The Secretary, Department Of Health And Human Services | Cannabinoid receptor mediating compounds |
| CA2889697C (en) | 2012-11-13 | 2023-03-14 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Cannabinoid receptor mediating compounds |
| MX2015010935A (es) | 2013-02-22 | 2015-10-29 | Merck Sharp & Dohme | Compuestos biciclicos antidiabeticos. |
| US9650375B2 (en) | 2013-03-14 | 2017-05-16 | Merck Sharp & Dohme Corp. | Indole derivatives useful as anti-diabetic agents |
| EP2970384A1 (en) | 2013-03-15 | 2016-01-20 | Synergy Pharmaceuticals Inc. | Agonists of guanylate cyclase and their uses |
| WO2014151200A2 (en) | 2013-03-15 | 2014-09-25 | Synergy Pharmaceuticals Inc. | Compositions useful for the treatment of gastrointestinal disorders |
| AU2014274812B2 (en) | 2013-06-05 | 2018-09-27 | Bausch Health Ireland Limited | Ultra-pure agonists of guanylate cyclase C, method of making and using same |
| WO2015051496A1 (en) | 2013-10-08 | 2015-04-16 | Merck Sharp & Dohme Corp. | Antidiabetic tricyclic compounds |
| CA2948349C (en) | 2014-05-09 | 2023-03-28 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | Pyrazole derivatives and their use as cannabinoid receptor mediators |
| EP3109237A1 (en) | 2015-06-22 | 2016-12-28 | AnaMar AB | Novel 5-ht2 antagonists |
| EP3551176A4 (en) | 2016-12-06 | 2020-06-24 | Merck Sharp & Dohme Corp. | Antidiabetic heterocyclic compounds |
| WO2022245627A1 (en) | 2021-05-17 | 2022-11-24 | The United States Of America, As Represented By The Secretary, Department Of Health And Human Services | A facile and odor-free approach to convert sulfonyl urea derivatives to chalcogenide sulfonyl urea derivatives |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5591764A (en) * | 1988-09-27 | 1997-01-07 | E. I. Du Pont De Nemours And Company | N-acylated pyrazolines |
| FR2692575B1 (fr) * | 1992-06-23 | 1995-06-30 | Sanofi Elf | Nouveaux derives du pyrazole, procede pour leur preparation et compositions pharmaceutiques les contenant. |
| RU2149479C1 (ru) * | 1999-03-23 | 2000-05-20 | Научно-исследовательский институт электронных приборов | Способ сборки вакуумной оболочки рентгеновского электронно-оптического преобразователя |
-
2001
- 2001-03-22 PT PT01915398T patent/PT1268435E/pt unknown
- 2001-03-22 EP EP01915398A patent/EP1268435B1/en not_active Expired - Lifetime
- 2001-03-22 PL PL358101A patent/PL208098B1/pl not_active IP Right Cessation
- 2001-03-22 CN CNB01806910XA patent/CN1205188C/zh not_active Expired - Fee Related
- 2001-03-22 RU RU2002128352/04A patent/RU2245878C2/ru not_active IP Right Cessation
- 2001-03-22 AU AU4250101A patent/AU4250101A/xx active Pending
- 2001-03-22 MX MXPA02009258A patent/MXPA02009258A/es active IP Right Grant
- 2001-03-22 KR KR1020027012531A patent/KR100800277B1/ko not_active Expired - Fee Related
- 2001-03-22 WO PCT/EP2001/003247 patent/WO2001070700A1/en not_active Ceased
- 2001-03-22 IL IL15145201A patent/IL151452A0/xx active IP Right Grant
- 2001-03-22 AT AT01915398T patent/ATE346047T1/de active
- 2001-03-22 ES ES01915398T patent/ES2272449T3/es not_active Expired - Lifetime
- 2001-03-22 DZ DZ013335A patent/DZ3335A1/fr active
- 2001-03-22 UA UA2002108258A patent/UA74367C2/ru unknown
- 2001-03-22 DE DE60124685T patent/DE60124685T2/de not_active Expired - Lifetime
- 2001-03-22 HU HU0204519A patent/HUP0204519A3/hu unknown
- 2001-03-22 SI SI200130668T patent/SI1268435T1/sl unknown
- 2001-03-22 AU AU2001242501A patent/AU2001242501B2/en not_active Ceased
- 2001-03-22 CA CA002401832A patent/CA2401832C/en not_active Expired - Fee Related
- 2001-03-22 HK HK03104555.6A patent/HK1052349B/zh not_active IP Right Cessation
- 2001-03-22 BR BR0109457-2A patent/BR0109457A/pt not_active Application Discontinuation
- 2001-03-22 DK DK01915398T patent/DK1268435T3/da active
- 2001-03-22 JP JP2001568910A patent/JP2004500401A/ja not_active Withdrawn
- 2001-03-22 SK SK1352-2002A patent/SK287074B6/sk not_active IP Right Cessation
- 2001-03-23 US US09/814,694 patent/US6476060B2/en not_active Expired - Lifetime
-
2002
- 2002-08-22 IL IL151452A patent/IL151452A/en not_active IP Right Cessation
- 2002-09-20 NO NO20024531A patent/NO324173B1/no not_active IP Right Cessation
-
2007
- 2007-01-30 CY CY20071100124T patent/CY1105967T1/el unknown
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| RU2002128352A (ru) | Производные 4,5-дигидро-1н-пиразола, обладающие cb1-антагонистической активностью | |
| JP4313672B2 (ja) | 強力なcb1−拮抗活性を有する4,5−ジヒドロ−1h−ピラゾール誘導体 | |
| ES2341540T3 (es) | Derivados de 1h-1,2,4-triazol-3-carboxamida como ligandos de receptores de canabinoide cb1. | |
| ES2311972T3 (es) | Derivados 1,3,5-trisubstituidos de 4,5-dihidro-1h-pirazol que tienen actividad antagonista de cb1. | |
| KR100846614B1 (ko) | Cb1-길항 활성을 갖는 4,5-디하이드로-1h-피라졸 유도체 | |
| RU2004107131A (ru) | Новые производные 4,5-дигидро-1h-пиразола, имеющие cb1- антагонистическую активность | |
| AU2002333853A1 (en) | 4,5-dihydro-1H-pyrazole derivatives having potent CB1-antagonistic activity | |
| RU2004114263A (ru) | Производные тиазола, имеющие антагонистичекую агонистическую или частично агонистическую св1-активность | |
| CA2457444A1 (en) | 1h-imidazole derivatives having cb1 agonistic, cb1 partial agonistic or cb1- antagonistic activity | |
| EP3409658A1 (en) | Tetrahydronaphthalene derivative | |
| CN106715395B (zh) | 作为taar调节剂的6-氨基-5,6,7,8-四氢萘-2-基或3-氨基色满-7-基衍生物 | |
| JPS6039069B2 (ja) | アミン誘導体 | |
| FR2728571A1 (fr) | Nouveaux ethers d'oximes tricycliques, leurs procedes de preparation et les compositions pharmaceutiques qui les contiennent | |
| RU2006141835A (ru) | Тетразамещенные производные имидазола в качестве модуляторов рецепторов св1 каннабиноидов с высокой селективностью к подтипам рецепторов св1/св2 | |
| RU2006110543A (ru) | Применение производных 4,5-дигидро-1н-пиразолов в качестве антагонистов св1 | |
| PT869952E (pt) | Derivados de 5-naftalen-1-il-1,3-dioxanos processo para a sua preparacao e sua aplicacao em terapeutica | |
| JPWO2022094400A5 (ru) | ||
| HK1117512A (en) | 4,5-dihydro-1h-pyrazole derivatives having potent cb1-antagonistic activity | |
| JP2004175720A (ja) | アミジン類、アミドオキシム類、1H−ピラゾロ〔1,5−b〕〔1,2,4〕トリアゾール類の合成方法 | |
| HK1068253A (en) | 4,5-dihydro-1h-pyrazole derivatives having potent cb1-antagonistic acitivty | |
| HK1099688B (en) | 1,3,5-trisubstituted 4,5-dihydro-1h-pyrazole derivatives having cb1-antagonistic activity |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| MM4A | The patent is invalid due to non-payment of fees |
Effective date: 20120323 |