KR20180069423A - 스파이로플루오렌 잔텐닐 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 - Google Patents
스파이로플루오렌 잔텐닐 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 Download PDFInfo
- Publication number
- KR20180069423A KR20180069423A KR1020160171598A KR20160171598A KR20180069423A KR 20180069423 A KR20180069423 A KR 20180069423A KR 1020160171598 A KR1020160171598 A KR 1020160171598A KR 20160171598 A KR20160171598 A KR 20160171598A KR 20180069423 A KR20180069423 A KR 20180069423A
- Authority
- KR
- South Korea
- Prior art keywords
- group
- substituted
- layer
- unsubstituted
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 150000001875 compounds Chemical class 0.000 claims description 89
- 239000010410 layer Substances 0.000 claims description 88
- 239000000463 material Substances 0.000 claims description 31
- 125000001072 heteroaryl group Chemical group 0.000 claims description 21
- 239000010409 thin film Substances 0.000 claims description 19
- 125000003118 aryl group Chemical group 0.000 claims description 18
- 125000001424 substituent group Chemical group 0.000 claims description 18
- 239000000203 mixture Substances 0.000 claims description 12
- 230000000903 blocking effect Effects 0.000 claims description 11
- 125000000217 alkyl group Chemical group 0.000 claims description 9
- 238000000034 method Methods 0.000 claims description 8
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 7
- 229910052805 deuterium Inorganic materials 0.000 claims description 7
- 229910052736 halogen Inorganic materials 0.000 claims description 7
- 150000002367 halogens Chemical class 0.000 claims description 7
- 229910052739 hydrogen Inorganic materials 0.000 claims description 7
- 150000002431 hydrogen Chemical class 0.000 claims description 7
- 239000001257 hydrogen Substances 0.000 claims description 7
- 150000002825 nitriles Chemical class 0.000 claims description 7
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 6
- 125000006749 (C6-C60) aryl group Chemical group 0.000 claims description 5
- -1 C1 to C12 alkoxy Chemical group 0.000 claims description 5
- 238000002347 injection Methods 0.000 claims description 5
- 239000007924 injection Substances 0.000 claims description 5
- 125000006539 C12 alkyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 claims description 4
- 125000002529 biphenylenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C12)* 0.000 claims description 4
- 229910052799 carbon Inorganic materials 0.000 claims description 4
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 claims description 4
- 229910052757 nitrogen Inorganic materials 0.000 claims description 4
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 claims description 4
- 125000005580 triphenylene group Chemical group 0.000 claims description 4
- 125000000732 arylene group Chemical group 0.000 claims description 3
- 125000005549 heteroarylene group Chemical group 0.000 claims description 3
- 239000002346 layers by function Substances 0.000 claims description 3
- 125000004642 (C1-C12) alkoxy group Chemical group 0.000 claims description 2
- ICPSWZFVWAPUKF-UHFFFAOYSA-N 1,1'-spirobi[fluorene] Chemical group C1=CC=C2C=C3C4(C=5C(C6=CC=CC=C6C=5)=CC=C4)C=CC=C3C2=C1 ICPSWZFVWAPUKF-UHFFFAOYSA-N 0.000 claims description 2
- YJTKZCDBKVTVBY-UHFFFAOYSA-N 1,3-Diphenylbenzene Chemical group C1=CC=CC=C1C1=CC=CC(C=2C=CC=CC=2)=C1 YJTKZCDBKVTVBY-UHFFFAOYSA-N 0.000 claims description 2
- IANQTJSKSUMEQM-UHFFFAOYSA-N 1-benzofuran Chemical group C1=CC=C2OC=CC2=C1 IANQTJSKSUMEQM-UHFFFAOYSA-N 0.000 claims description 2
- 125000002252 acyl group Chemical group 0.000 claims description 2
- 125000003545 alkoxy group Chemical group 0.000 claims description 2
- 125000006267 biphenyl group Chemical group 0.000 claims description 2
- TXCDCPKCNAJMEE-UHFFFAOYSA-N dibenzofuran Chemical group C1=CC=C2C3=CC=CC=C3OC2=C1 TXCDCPKCNAJMEE-UHFFFAOYSA-N 0.000 claims description 2
- IYYZUPMFVPLQIF-ALWQSETLSA-N dibenzothiophene Chemical group C1=CC=CC=2[34S]C3=C(C=21)C=CC=C3 IYYZUPMFVPLQIF-ALWQSETLSA-N 0.000 claims description 2
- 125000001181 organosilyl group Chemical group [SiH3]* 0.000 claims description 2
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 claims description 2
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 2
- 125000004076 pyridyl group Chemical group 0.000 claims description 2
- 125000003944 tolyl group Chemical group 0.000 claims description 2
- 125000004400 (C1-C12) alkyl group Chemical group 0.000 claims 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 claims 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 claims 1
- 150000002221 fluorine Chemical group 0.000 claims 1
- 150000002894 organic compounds Chemical class 0.000 abstract description 3
- 238000006243 chemical reaction Methods 0.000 description 32
- 239000007787 solid Substances 0.000 description 24
- 230000000052 comparative effect Effects 0.000 description 22
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 18
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 15
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 15
- NFHFRUOZVGFOOS-UHFFFAOYSA-N palladium;triphenylphosphane Chemical compound [Pd].C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 NFHFRUOZVGFOOS-UHFFFAOYSA-N 0.000 description 14
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 14
- 238000002360 preparation method Methods 0.000 description 14
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 10
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 10
- 239000012153 distilled water Substances 0.000 description 10
- 239000011541 reaction mixture Substances 0.000 description 9
- 238000010898 silica gel chromatography Methods 0.000 description 9
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 8
- 239000011368 organic material Substances 0.000 description 7
- 229910000027 potassium carbonate Inorganic materials 0.000 description 7
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 6
- SCVFZCLFOSHCOH-UHFFFAOYSA-M potassium acetate Chemical compound [K+].CC([O-])=O SCVFZCLFOSHCOH-UHFFFAOYSA-M 0.000 description 6
- 238000010992 reflux Methods 0.000 description 6
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 5
- 239000007864 aqueous solution Substances 0.000 description 5
- 239000011780 sodium chloride Substances 0.000 description 5
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 4
- 229940125904 compound 1 Drugs 0.000 description 4
- 238000005401 electroluminescence Methods 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 125000005842 heteroatom Chemical group 0.000 description 4
- 229940098779 methanesulfonic acid Drugs 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- TVIVIEFSHFOWTE-UHFFFAOYSA-K tri(quinolin-8-yloxy)alumane Chemical compound [Al+3].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 TVIVIEFSHFOWTE-UHFFFAOYSA-K 0.000 description 4
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 238000004020 luminiscence type Methods 0.000 description 3
- 239000012299 nitrogen atmosphere Substances 0.000 description 3
- 235000011056 potassium acetate Nutrition 0.000 description 3
- MTCARZDHUIEYMB-UHFFFAOYSA-N 2-bromofluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC(Br)=CC=C3C2=C1 MTCARZDHUIEYMB-UHFFFAOYSA-N 0.000 description 2
- DDGPPAMADXTGTN-UHFFFAOYSA-N 2-chloro-4,6-diphenyl-1,3,5-triazine Chemical compound N=1C(Cl)=NC(C=2C=CC=CC=2)=NC=1C1=CC=CC=C1 DDGPPAMADXTGTN-UHFFFAOYSA-N 0.000 description 2
- UJOBWOGCFQCDNV-UHFFFAOYSA-N 9H-carbazole Chemical compound C1=CC=C2C3=CC=CC=C3NC2=C1 UJOBWOGCFQCDNV-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- IZYQYWHHPQRWFX-UHFFFAOYSA-N [3-(4,6-diphenyl-1,3,5-triazin-2-yl)phenyl]boronic acid Chemical compound OB(O)C1=CC=CC(C=2N=C(N=C(N=2)C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 IZYQYWHHPQRWFX-UHFFFAOYSA-N 0.000 description 2
- 238000004140 cleaning Methods 0.000 description 2
- 239000012043 crude product Substances 0.000 description 2
- NXQGGXCHGDYOHB-UHFFFAOYSA-L cyclopenta-1,4-dien-1-yl(diphenyl)phosphane;dichloropalladium;iron(2+) Chemical compound [Fe+2].Cl[Pd]Cl.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1.[CH-]1C=CC(P(C=2C=CC=CC=2)C=2C=CC=CC=2)=C1 NXQGGXCHGDYOHB-UHFFFAOYSA-L 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 230000005525 hole transport Effects 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- IBHBKWKFFTZAHE-UHFFFAOYSA-N n-[4-[4-(n-naphthalen-1-ylanilino)phenyl]phenyl]-n-phenylnaphthalen-1-amine Chemical compound C1=CC=CC=C1N(C=1C2=CC=CC=C2C=CC=1)C1=CC=C(C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C3=CC=CC=C3C=CC=2)C=C1 IBHBKWKFFTZAHE-UHFFFAOYSA-N 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- HXITXNWTGFUOAU-UHFFFAOYSA-N phenylboronic acid Chemical compound OB(O)C1=CC=CC=C1 HXITXNWTGFUOAU-UHFFFAOYSA-N 0.000 description 2
- 238000000926 separation method Methods 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000000859 sublimation Methods 0.000 description 2
- 230000008022 sublimation Effects 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 125000006710 (C2-C12) alkenyl group Chemical group 0.000 description 1
- 125000006750 (C7-C60) arylalkyl group Chemical group 0.000 description 1
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 1
- HIHYAKDOWUFGBK-UHFFFAOYSA-N 1,3-dibromo-5-phenylbenzene Chemical group BrC1=CC(Br)=CC(C=2C=CC=CC=2)=C1 HIHYAKDOWUFGBK-UHFFFAOYSA-N 0.000 description 1
- KQZLRWGGWXJPOS-NLFPWZOASA-N 1-[(1R)-1-(2,4-dichlorophenyl)ethyl]-6-[(4S,5R)-4-[(2S)-2-(hydroxymethyl)pyrrolidin-1-yl]-5-methylcyclohexen-1-yl]pyrazolo[3,4-b]pyrazine-3-carbonitrile Chemical compound ClC1=C(C=CC(=C1)Cl)[C@@H](C)N1N=C(C=2C1=NC(=CN=2)C1=CC[C@@H]([C@@H](C1)C)N1[C@@H](CCC1)CO)C#N KQZLRWGGWXJPOS-NLFPWZOASA-N 0.000 description 1
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical group C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- VADKRMSMGWJZCF-UHFFFAOYSA-N 2-bromophenol Chemical compound OC1=CC=CC=C1Br VADKRMSMGWJZCF-UHFFFAOYSA-N 0.000 description 1
- JHUUPUMBZGWODW-UHFFFAOYSA-N 3,6-dihydro-1,2-dioxine Chemical compound C1OOCC=C1 JHUUPUMBZGWODW-UHFFFAOYSA-N 0.000 description 1
- YLZOPXRUQYQQID-UHFFFAOYSA-N 3-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)-1-[4-[2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidin-5-yl]piperazin-1-yl]propan-1-one Chemical compound N1N=NC=2CN(CCC=21)CCC(=O)N1CCN(CC1)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F YLZOPXRUQYQQID-UHFFFAOYSA-N 0.000 description 1
- MNOJRWOWILAHAV-UHFFFAOYSA-N 3-bromophenol Chemical compound OC1=CC=CC(Br)=C1 MNOJRWOWILAHAV-UHFFFAOYSA-N 0.000 description 1
- GZFGOTFRPZRKDS-UHFFFAOYSA-N 4-bromophenol Chemical compound OC1=CC=C(Br)C=C1 GZFGOTFRPZRKDS-UHFFFAOYSA-N 0.000 description 1
- 0 C*=*C=C(C1(c2ccccc22)c3ccccc3Oc3c1cccc3)C2=C Chemical compound C*=*C=C(C1(c2ccccc22)c3ccccc3Oc3c1cccc3)C2=C 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000004984 aromatic diamines Chemical class 0.000 description 1
- 125000004429 atom Chemical group 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 229940125877 compound 31 Drugs 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- 125000004093 cyano group Chemical group *C#N 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 125000000753 cycloalkyl group Chemical group 0.000 description 1
- JCWIWBWXCVGEAN-UHFFFAOYSA-L cyclopentyl(diphenyl)phosphane;dichloropalladium;iron Chemical compound [Fe].Cl[Pd]Cl.[CH]1[CH][CH][CH][C]1P(C=1C=CC=CC=1)C1=CC=CC=C1.[CH]1[CH][CH][CH][C]1P(C=1C=CC=CC=1)C1=CC=CC=C1 JCWIWBWXCVGEAN-UHFFFAOYSA-L 0.000 description 1
- 239000002019 doping agent Substances 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- YLQWCDOCJODRMT-UHFFFAOYSA-N fluoren-9-one Chemical compound C1=CC=C2C(=O)C3=CC=CC=C3C2=C1 YLQWCDOCJODRMT-UHFFFAOYSA-N 0.000 description 1
- 150000002220 fluorenes Chemical group 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000005283 ground state Effects 0.000 description 1
- MILUBEOXRNEUHS-UHFFFAOYSA-N iridium(3+) Chemical class [Ir+3] MILUBEOXRNEUHS-UHFFFAOYSA-N 0.000 description 1
- 125000005647 linker group Chemical group 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 125000002560 nitrile group Chemical group 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- PIBWKRNGBLPSSY-UHFFFAOYSA-L palladium(II) chloride Chemical compound Cl[Pd]Cl PIBWKRNGBLPSSY-UHFFFAOYSA-L 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/04—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D251/00—Heterocyclic compounds containing 1,3,5-triazine rings
- C07D251/02—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings
- C07D251/12—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D251/14—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom
- C07D251/24—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom to three ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/96—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings spiro-condensed with carbocyclic rings or ring systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/10—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H01L51/0067—
-
- H01L51/0073—
-
- H01L51/50—
-
- H01L51/5012—
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1059—Heterocyclic compounds characterised by ligands containing three nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1088—Heterocyclic compounds characterised by ligands containing oxygen as the only heteroatom
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
도 2 는 실시예 1에서 제조된 화합물 1의 DSC 측정 결과이다.
| 화합물 번호 | 1H NMR (400 MHz, THF-d8): δ |
MS/
Q- TOF (M+) |
| 1 | 8.81(m, 4H), 8.66(d, 1H), 8.20(dd, 1H), 7.96(d, 2H), 7.68~7.58(m, 6H), 7.44(t, 2H), 7.36(d, 1H), 7.29~7.23(m, 5H), 6.84(t, 1H), 6.62(d, 1H), 6.47(d, 1H) | 563 |
| 2 | 8.75~8.70(m, 4H), 8.59~8.58(d, 1H), 8.12~8.10(d, 1H), 7.84~7.16(m, 20H), 6.79~6.76(t, 1H), 6.56~6.54(d, 1H), 6.39(d, 1H) | 639 |
| 3 | 8.99(s, 2H), 8.39(d, 2H), 8.14(d, 1H), 7.90~7.77(m, 8H), 7.49~7.45(m, 6H), 7.29~7.11(m, 9H), 6.95(t, 1H) | 663 |
| 4 | 8.24(d, 1H), 7.77~7.75(d, 6H), 7.45~7.28(m, 15H), 7.18~7.09(m, 10H), 6.95(t, 1H) | 715 |
| 5 | 8.84(d, 3H), 7.77(d, 2H), 7.60(s, 2H), 7.47~7.28(m, 19H), 7.18~7.09(m, 6H), 6.95(t, 1H) | 715 |
| 6 | 8.78~8.14(d, 5H), 7.77(d, 1H), 7.65(d, 1H), 7.45~7.09(m, 16H), 6.95(t, 1H), 2.24(s, 3H) | 577 |
| 7 | 8.80~8.14(d, 5H), 7.77(d, 2H), 7.45~7.15(m, 14H), 6.97(d, 1H), 6.73(d, 1H), 6.64(s, 1H), 2.24(s, 3H) | 577 |
| 8 | 8.78~8.14(d, 5H), 7.77~7.75(d, 2H), 7.67(s, 1H), 7.50~7.31(m, 6H), 7.31~7.28(m, 4H), 7.18~7.09(m, 5H), 6.95(t, 1H), 0.15(s, 9H) | 635 |
| 9 | 8.81~8.14(d, 5H), 7.77(d, 1H), 7.45~7.06(m, 17H), 6.95(t, 1H), 2.49(s, 3H) | 577 |
| 10 | 8.80~8.14(m, 5H), 7.77(d, 2H), 7.45~7.15(m, 15H), 6.96~6.90(m, 2H), 2.24(s, 3H) | 577 |
| 11 | 8.18~8.14(d, 5H), 7.69(d, 2H), 7.48~7.41(m, 6H), 7.31~7.29(m, 5H), 7.15~7.09(m, 4H), 6.95(t, 1H), 1.25(s, 18H) | 675 |
| 12 | 8.18~8.14(d, 5H), 7.83~7.77(d, 2H), 7.67(s, 1H), 7.53(d, 1H), 7.45~7.29(m, 14H), 7.18~7.09(m, 5H), 6.95(t, 1H) | 639 |
| 13 | 8.18~8.14(d, 5H), 7.77(d, 2H), 7.45~7.28(m, 17H), 7.18~7.15(m, 4H), 6.95(d, 1H) | 639 |
| 14 | 8.18~8.14(d, 5H), 7.77(d, 1H), 7.69(d, 2H), 7.53(d, 1H), 7.45~7.11(m, 19H), 6.95(t, 1H) | 639 |
| 15 | 8.18~8.14(d, 5H), 7.90~7.77(m, 5H), 7.65~7.63(m, 2H), 7.49~7.15(m, 18H), 6.98(d, 1H) | 689 |
| 16 | 8.18~8.14(d, 5H), 7.77~7.72(d, 2H), 7.45~7.28(m, 10H), 7.18~7.09(m, 6H), 6.95(t, 1H), 2.52(m, 2H), 1.49(m, 2H), 1.21~1.19(m, 6H), 0.78(m, 3H) | 647 |
| 17 | 8.18(d, 4H), 7.77~7.75(d, 4H), 7.45~7.09(m, 19H), 6.95(d, 2H) | 639 |
| 18 | 8.18~8.14(m, 5H), 7.77(dd, 2H), 7.6(s, 1H), 7.47~7.11(m, 19H), 6.95(m, 2H) | 639 |
| 19 | 8.18(dd, 4H), 7.77~7.75(m, 4H), 7.45~7.09(m, 19H), 6.95(m, 2H) | 639 |
| 20 | 8.18(dd, 4H), 7.77~7.75(m, 4H), 7.45~7.41(m, 6H), 7.31~7.28(m, 5H), 7.18~7.11(m, 12H), 6.95(m, 2H) | 715 |
| 21 | 8.18~8.14(m, 5H), 7.77(dd, 2H), 7.6(s, 1H), 7.47~7.28(m, 13H), 7.18~7.09(m, 10H), 6.95(m, 2H) | 715 |
| 22 | 8.18(dd, 4H), 7.77~7.75(m, 4H), 7.6(s, 1H), 7.47~7.11(m, 22H), 6.95(m, 2H) | 715 |
| 23 | 8.18(dd, 4H), 7.77~7.75(m, 4H), 7.41~7.7.28(m, 13H), 7.18~7.09(m, 10H), 6.95(m, 1H) | 715 |
| 24 | 8.18(dd, 4H), 7.77~7.75(m, 6H), 7.41~7.09(m, 21H), 6.95(m, 2H) | 715 |
| 25 | 8.18(dd, 4H), 7.77~7.75(m, 6H), 7.41~7.11(m, 21H), 6.95(m, 2H) | 715 |
| 26 | 8.18(dd, 4H), 7.77~7.75(m, 4H), 7.45~7.41(m, 6H), 7.31~7.28(m, 5H), 7.18~7.11(m, 16H), 6.95(m, 2H) | 791 |
| 27 | 8.18~8.14(m, 5H), 7.77(dd, 2H), 7.6(s, 3H), 7.47~7.28(m, 19H), 7.18~7.11(m, 6H), 6.95(d, 2H) | 791 |
| 28 | 8.18(dd, 4H), 7.77~7.75(m, 4H), 7.6(m, 1H), 7.47~7.28(m, 14H), 7.18~7.11(m, 12H), 6.95(m, 2H) | 791 |
| 29 | 8.18(dd, 4H), 7.77(dd, 2H), 7.56(s, 3H), 7.41~7.28(m, 16H), 7.18~7.09(m, 6H), 6.95(m, 2H) | 715 |
| 30 | 8.4(dd, 1H), 8.18(dd, 4H), 8.07(d, 2H), 7.77(dd, 2H), 7.62(m, 1H), 7.41~7.09(m, 19H), 6.95~6.9(m, 3H) | 716 |
| 31 | 9.14(s, 1H), 8.6(dd, 1H), 8.32(m, 1H), 8.18(dd, 4H), 7.77(dd, 2H), 7.56(s, 3H), 7.47~7.09(m, 18H), 6.95(m, 2H) | 716 |
| 32 | 8.65(d, 2H), 8.18(dd, 4H), 7.89(d, 2H), 7.77(dd, 2H), 7.56(s, 3H), 7.41~7.09(m, 17H), 6.95(m, 2H) | 716 |
| 33 | 8.18(dd, 4H), 7.83(d, 2H), 7.77(dd, 2H), 7.67(d, 2H), 7.53(dd, 2H), 7.45~7.28(m, 11H), 7.18~7.09(m, 6H), 6.95(m, 2H), 1.62(s, 6H) | 755 |
| 34 | 8.18(dd, 4H), 7.83(d, 2H), 7.77(dd, 2H), 7.67(d, 2H), 7.53~7.09(m, 19H), 6.95(m, 2H), 2.11(m, 2H), 1.86(m, 2H), 1.46~1.36(m, 4H) | 781 |
| 35 | 8.38(d, 4H), 8.03~7.97(m, 4H), 7.87(d, 2H), 7.73~7.15(m, 31H) | 880 |
| 36 | 8.38(d, 4H), 8.03~7.97(m, 4H), 7.87(d, 2H), 7.73(d, 2H), 7.65~7.61(m, 6H), 7.51~7.48(m, 5H), 7.38~7.29(m, 6H), 7.15(d, 2H), 1.97(s, 4H), 1.41~1.29(m, 16H), 0.98(s, 6H) | 896 |
| 37 | 8.38(d, 4H), 8.03~7.97(m, 4H), 7.87~7.85(m, 4H), 7.73~7.26(m, 25H), 7.15(s, 2H) | 878 |
| 38 | 8.38(d, 4H), 8.03~7.85(m, 7H), 7.73~7.26(m, 26H), 7.15(s, 2H) | 878 |
| 39 | 8.38(d, 4H), 8.05(d, 1H), 7.97(d, 2H), 7.76(s, 3H), 7.65~7.29(m, 17H), 7.18~7.15(m, 3H), 3.24(s, 3H), 2.63(s, 3H) | 744 |
| 40 | 8.38(d, 4H), 7.97~7.95(m, 3H), 7.85(s, 1H), 7.65~7.29(m, 15H), 7.18~7.15(m, 2H) | 563 |
| 41 | 8.38(s, 2H), 7.97~7.95(m, 5H), 7.85(s, 1H), 7.65~7.48(m, 12H), 7.48~7.31 (m, 7H), 7.18~7.15(m, 2H) | 639 |
| 42 | 9.19(s, 2H), 8.59(s, 2H), 8.10~7.95(m, 9H), 7.85(s, 1H), 7.69~7.65(m, 6H), 7.48~7.29(m, 7H), 7.18~7.15(m, 2H) | 663 |
| 43 | 7.97~7.95(m, 7H), 7.85(s, 1H), 7.65~7.48(m, 14H), 7.38~7.29(m, 9H), 7.18~7.15(m, 2H) | 715 |
| 44 | 8.34(d, 2H), 7.97~7.95(m, 3H), 7.85~7.80(m, 3H), 7.67~7.48(m, 18H), 7.38~7.29(m, 5H), 7.18~7.15(m, 2H) | 715 |
| 45 | 8.38(d, 4H), 7.97~7.95(m, 2H), 7.85(s, 2H), 7.65~7.28(m, 14H), 7.18~7.15(m, 2H), 2.44(s, 3H) | 577 |
| 46 | 8.38(d, 4H), 7.97~7.95(m, 3H), 7.85(s, 1H), 7.65~7.38(m, 12H), 7.18~7.15(m, 2H), 6.93(d, 1H), 6.84(s, 1H), 2.44(s, 3H) | 577 |
| 47 | 8.38(d, 4H), 7.97~7.95(m, 3H), 7.87~7.85(m, 2H), 7.70~7.61(m, 6H), 7.51~7.48(m, 3H), 7.38~7.29(m, 4H), 7.18~7.15(m, 2H), 0.35(s, 9H) | 635 |
| 48 | 8.38(d, 4H), 7.97~7.95(m, 2H), 7.85(s, 1H), 7.65~7.15(m, 17H) 2.69(s, 3H) | 577 |
| 49 | 8.38(d, 4H), 7.97~7.95(m, 3H), 7.85(s, 1H), 7.65~7.31(m, 13H), 7.18~7.10(m, 3H), 2.44(s, 3H) | 577 |
| 50 | 8.38(d, 4H), 7.97~7.85(m, 4H), 7.68~7.61(m, 6H), 7.51~7.48(m, 4H), 7.38~7.29(m, 4H), 7.18~7.15(m, 2H), 1.45(s, 9H) | 619 |
| 51 | 8.38(d, 4H), 8.03~7.95(m, 3H), 7.87~7.85(m, 2H), 7.73(s, 1H), 7.65~7.48(m, 13H), 7.38~7.29(m, 4H), 7.18~7.15(m, 2H) | 639 |
| 52 | 8.38(d, 4H), 7.97 ~ 7.95(m, 3H), 7.85(s, 1H), 7.65 ~ 7.48(m, 16H), 7.38 ~ 7.35(m, 3H), 7.18 ~ 7.15(m, 2H) | 639 |
| 53 | 8.38(d, 4H), 7.97 ~ 7.85(m, 5H), 7.73(d, 1H), 7.65 ~ 7.31(m, 17H), 7.18 ~ 7.15(m, 2H) | 639 |
| 54 | 8.38(d, 4H), 8.11 ~ 7.95(m, 6H), 7.85(s, 2H), 7.69 ~ 7.48(m, 17H), 7.18(d, 2H) | 689 |
| 55 | 8.38(d, 4H), 7.97 ~ 7.92(m, 3H), 7.85(s, 1H), 7.61 ~ 7.48(m, 9H), 7.38 ~ 7.29(m, 5H), 7.18 ~ 7.15(m, 2H), 2.72(t, 2H), 1.69(m, 2H), 1.41 ~ 1.39(m, 6H), 0.98(t, 3H) | 647 |
| 56 | 8.38(d, 4H), 7.97 ~ 7.95(m, 4H), 7.85(s, 1H), 7.61 ~ 7.31(m, 18H), 7.18 ~ 7.15(m, 2H) | 639 |
| 57 | 8.38 ~ 8.34(m, 5H), 7.97(d, 2H), 7.85(s, 1H), 7.81(s, 1H), 7.67 ~ 7.31(m, 18H), 7.18 ~ 7.15(m, 2H) | 639 |
| 58 | 8.38(d, 4H), 7.97 ~ 7.95(m, 3H), 7.85(s, 1H), 7.61 ~ 7.29(m, 19H), 7.18 ~ 7.15(m, 2H) | 639 |
| 59 | 8.38(d, 4H), 7.97 ~ 7.95(m, 4H), 7.85(s, 1H), 7.66 ~ 7.61(m, 7H), 7.51 ~ 7.48(m, 4H), 7.38 ~ 7.31(m, 11H), 7.18 ~ 7.15(m, 2H) | 715 |
| 60 | 8.38(m, 5H), 7.97(d, 2H), 7.85(s, 1H), 7.81(s, 1H), 7.67 ~ 7.48(m, 13H), 7.38 ~ 7.31(m, 9H), 7.18 ~ 7.15(m, 2H) | 715 |
| 61 | 8.38(d, 4H), 7.97 ~ 7.95(m, 4H), 7.85(s, 1H), 7.81(s, 1H), 7.67 ~ 7.31(m, 21H), 7.18 ~ 7.15(m, 2H) | 715 |
| 62 | 8.38(d, 4H), 7.97 ~ 7.95(m, 4H), 7.85(s, 1H), 7.65 ~ 7.48(m, 13H), 7.38 ~ 7.31(m, 9H), 7.18 ~ 7.15(m, 2H) | 715 |
| 63 | 8.38(d, 4H), 7.97 ~ 7.95(m, 6H), 7.85(s, 1H), 7.65 ~ 7.38(m, 20H), 7.18 ~ 7.15(m, 2H) | 715 |
| 64 | 8.38(d, 4H), 7.97 ~ 7.95(m, 6H), 7.85(s, 1H), 7.65 ~ 7.48(m, 15H), 7.38 ~ 7.29(m, 5H), 7.18 ~ 7.15(m, 2H) | 715 |
| 65 | 8.38(d, 4H), 7.97 ~ 7.95(m, 4H), 7.66 ~ 7.61(m, 7H), 7.51 ~ 7.48(m, 4H), 7.38 ~ 7.29(m, 16H), 7.18 ~ 7.15(m, 2H) | 791 |
| 66 | 8.38 ~ 8.34(m, 5H), 7.97(d, 1H), 7.85(s, 1H), 7.81(m, 3H), 7.67 ~ 7.48(m, 20H), 7.38 ~ 7.29(m, 5H), 7.18 ~ 7.15(m, 2H) | 791 |
| 67 | 8.38(d, 4H), 7.97 ~ 7.95(m, 4H), 7.85(s, 1H), 7.81(s, 1H), 7.67 ~ 7.48(m, 14H), 7.38 ~ 7.29(m, 11H), 7.18 ~ 7.15(m, 2H) | 791 |
| 68 | 8.38(d, 4H), 7.97(d, 2H), 7.85(s, 1H), 7.76(s, 3H), 7.66 ~ 7.48(m, 16H), 7.38 ~ 7.29(m, 5H), 7.18 ~ 7.15(m, 2H) | 715 |
| 69 | .40(d, 1H), 8.18(d, 4H), 8.07(s, 2H), 7.77(d, 2H), 7.65~7.62(d, 2H), 7.46~7.11(m, 18H), 6.98~6.90(m, 3H) | 716 |
| 70 | 9.14(s, 1H), 8.60(d, 1H), 8.32(d, 1H), 8.18(d, 4H), 7.77(d, 2H), 7.45(s, 1H), 7.41~7.11(m, 20H), 6.98~6.95(m, 2H) | 716 |
| 71 | 8.65(d, 2H), 8.18(d, 4H), 7.89(d, 2H), 7.77(d, 2H), 7.65(s, 1H), 7.56(s, 3H), 7.46~7.11(m, 16H), 6.98~6.95(m, 2H) | 716 |
| 72 | 8.18(d, 4H), 7.83~7.77(m, 4H), 7.67~7.65(m, 3H), 7.53~7.28(m, 13H), 7.18~7.11(m, 4H), 6.98~6.95(m, 2H), 1.62(s, 6H) | 755 |
| 73 | 8.18(d, 4H), 7.83~7.77(m, 4H), 7.67~7.65(m, 3H), 7.53~7.11(m, 18H), 6.98~6.95(m, 2H), 2.11~1.86(m, 4H), 1.46~1.36(m, 4H) | 781 |
| 74 | 8.18(d, 4H), 7.83~7.77(m, 4H), 7.67~7.65(m, 3H), 7.53~6.95(m, 30H) | 880 |
| 75 | 8.18(d, 4H), 7.83~7.77(m, 4H), 7.67~7.65(m, 3H), 7.53(d, 2H), 7.46~7.41(m, 7H), 7.31~7.28(m, 4H), 7.18~7.11(m, 5H), 6.98~6.95(m, 2H), 1.77(m, 4H), 1.21~1.19(m, 16H), 0.78(m, 6H) | 896 |
| 76 | 8.18(d, 4H), 7.83~7.77(m, 4H), 7.67~7.65(m, 5H), 7.53~6.95(m, 26H) | 878 |
| 77 | 8.18(d, 4H), 7.83~7.65(m, 8H), 7.45~6.95(m, 27H) | 878 |
| 78 | 8.18(d, 4H), 7.85(d, 1H), 7.77(d, 2H), 7.65(s, 1H), 7.56(s, 3H), 7.46~7.11(m, 16H), 6.98~6.95(m, 3H), 3.04(s, 3H), 2.43(s, 3H) | 744 |
| 79 | 8.18~8.14(m, 5H), 7.83~7.77(m, 2H), 7.67~7.60(m, 2H), 7.53~7.31(m, 17H), 7.18~7.09(m, 4H), 6.98~6.95(m, 2H) | 715 |
| 80 | 8.18(d, 4H), 7.77(d, 2H), 7.45~6.95(m, 19H) | 563 |
| 81 | 8.18(d, 2H), 7.77~7.75(m, 4H), 7.45~7.28(m, 13H), 7.18~6.95(m, 10H) | 639 |
| 82 | 8.18~8.14(m, 5H), 7.77(d, 2H), 7.60(s, 1H), 7.47~6.95(m, 21H) | 639 |
| 83 | 8.18(d, 4H), 7.77~7.75(m, 4H), 7.46~6.95(m, 21H) | 639 |
| 84 | 8.18(d, 4H), 7.83~7.77(m, 2H), 7.67(s, 1H), 7.53~7.28(m, 15H), 7.18~6.95(m, 7H) | 639 |
| 85 | 9.14(s, 1H), 8.60(d, 1H), 8.32(d, 1H), 8.18(d, 4H), 7.77(d, 2H), 7.56~6.95(m, 23H) | 716 |
| 86 | 8.65(d, 2H), 8.18(d, 4H), 7.89(d, 2H), 7.77(d, 2H), 7.56(s, 3H), 7.46~6.95(m, 19H) | 716 |
| 87 | 8.18(d, 4H), 7.85(d, 1H), 7.77(d, 2H), 7.56(s, 3H), 7.46~6.95(m, 20H), 3.04(s, 3H), 2.43(s, 3H) | 744 |
| 88 | 8.18(d, 4H), 7.83(d, 2H), 7.67(s, 2H), 7.53~7.31(m, 19H), 7.12~6.95(m, 6H) | 715 |
| 89 | 9.20(s, 1H), 8.80~8.60(m, 6H), 7.70(d, 1H), 7.61~7.20(m, 18H), 7.10~7.05(m, 2H) | 640 |
| 90 | 8.95(d, 1H), 8.50(s, 1H), 8.30~8.25(m, 3H), 7.85(d, 2H), 7.70~7.20(m, 20H), 7.05(m, 1H) | 640 |
| 91 | 8.90(d, 1H), 8.51~8.40(m, 6H), 7.80~7.21(m, 19H), 7.05(m, 2H) | 640 |
| 92 | 9.21(s, 1H), 8.61(m, 6H), 7.69~7.25(m, 18H), 7.10(m, 3H) | 640 |
| 93 | 8.95(d, 1H), 8.70~8.50(m, 3H), 8.30(m, 3H), 7.80~7.60(m, 3H), 7.51~7.19(m, 16H), 7.08(m, 2H) | 640 |
| 94 | 8.96(d, 1H), 8.57(s, 1H), 8.30(m, 5H), 7.80~7.19(m, 19H), 7.05(m, 2H) | 640 |
| 95 | 9.20(s, 1H), 8.58(d, 2H), 8.06(d, 1H), 7.85(d, 2H), 7.77(d, 1H), 7.60~7.03(m, 21H) | 640 |
| 96 | 9.01(d, 1H), 8.50(s, 1H), 8.48(d, 2H), 8.30(d, 2H), 7.77(d, 2H), 7.60~7.05(m, 20H) | 640 |
| 97 | 8.60(m, 5H), 8.20(m, 4H), 7.55~7.40(m, 14H), 7.30~7.22(m, 5H), 7.05(m, 1H) | 638 |
| 98 | 8.58(m, 5H), 7.90~7.87(m, 5H), 7.55~7.45(m, 13H), 7.30(m, 3H), 7.01(m, 2H) | 638 |
| 99 | 8.50(m, 6H), 7.95(m, 4H), 7.60~7.40(m, 12H), 7.31~7.05(m, 8H) | 638 |
| 100 | 8.48(m, 3H), 7.90~7.80(m, 6H), 7.61~7.05(m, 21H) | 638 |
| 샘플 No. | 전자수송 화합물 No. |
정공차단 화합물 No. |
전압 OP. V |
효율 [cd/A] |
발광피크 [nm] |
수명 [LT97] |
| 비교예 1 | Alq3 | - | 6.7 | 4.2 | 461 | 20 |
| 비교예 2 | b | - | 6.2 | 4.5 | 461 | 43 |
| 비교예 3 | c | - | 6.3 | 4.6 | 462 | 45 |
| 비교예 4 | d | - | 6.6 | 4.3 | 462 | 22 |
| 비교예 5 | e | - | 6.6 | 4.4 | 461 | 24 |
| 실시예 1 | 1 | - | 5.4 | 4.9 | 461 | 65 |
| 실시예 2 | 2 | - | 4.8 | 5.3 | 461 | 85 |
| 실시예 3 | 12 | - | 5.8 | 4.7 | 462 | 56 |
| 실시예 4 | 18 | 5.3 | 5.1 | 462 | 66 | |
| 실시예 5 | 22 | - | 4.9 | 4.7 | 461 | 68 |
| 실시예 6 | 25 | - | 5.1 | 5.3 | 462 | 77 |
| 실시예 7 | 29 | 5.4 | 5.0 | 461 | 67 | |
| 실시예 8 | 31 | - | 5.1 | 5.4 | 461 | 77 |
| 실시예 9 | 38 | - | 5.8 | 4.7 | 461 | 54 |
| 실시예 10 | 41 | - | 5.2 | 5.4 | 462 | 79 |
| 실시예 11 | 49 | - | 5.7 | 4.7 | 462 | 53 |
| 실시예 12 | 57 | - | 5.4 | 5.0 | 462 | 68 |
| 실시예 13 | 58 | - | 5.1 | 5.4 | 462 | 79 |
| 실시예 14 | 70 | - | 5.0 | 5.3 | 462 | 78 |
| 실시예 15 | 79 | - | 5.8 | 4.7 | 461 | 56 |
| 실시예 16 | 80 | - | 5.7 | 4.7 | 461 | 55 |
| 실시예 17 | 82 | - | 5.1 | 5.3 | 462 | 81 |
| 실시예 18 | 97 | - | 5.1 | 5.3 | 461 | 80 |
| 비교예 6 | b | BCP | 6.0 | 6.0 | 462 | 88 |
| 실시예 19 | 31 | 90 | 4.8 | 6.5 | 462 | 102 |
| 실시예 20 | 31 | 92 | 4.7 | 6.4 | 462 | 110 |
| 실시예 21 | 31 | 95 | 4.8 | 6.5 | 462 | 107 |
Claims (9)
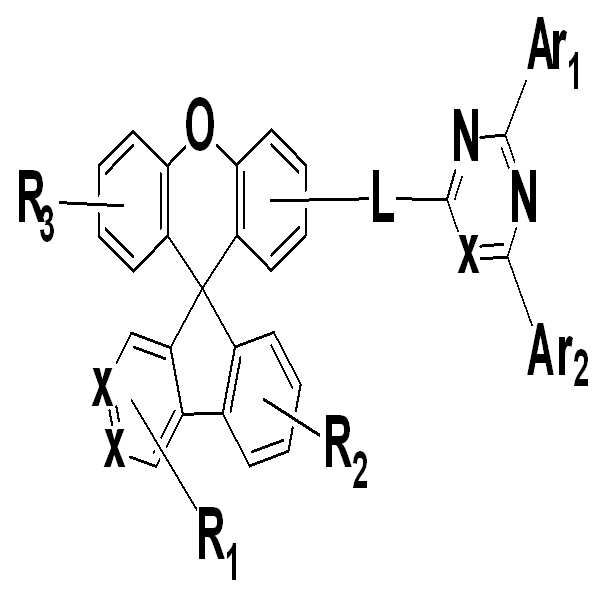
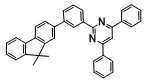
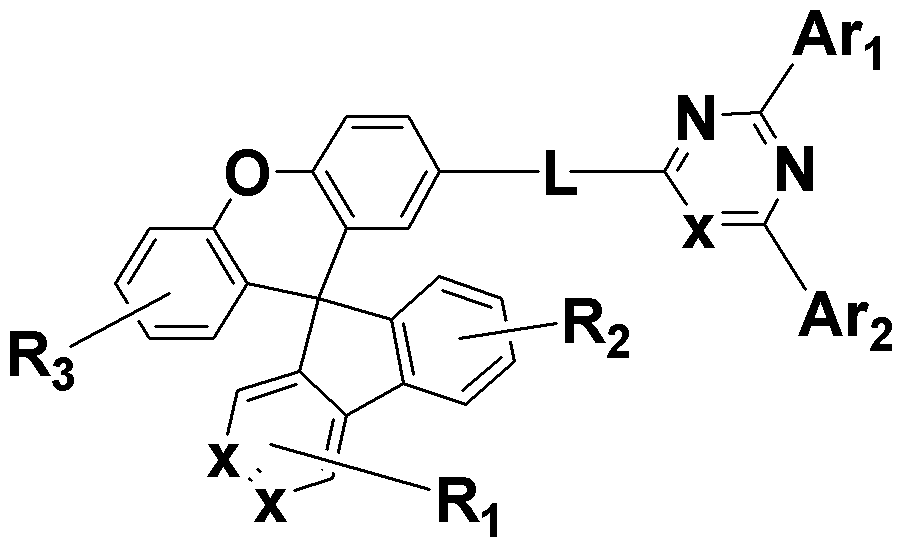
- 하기 화학식 a로 표시되는 스파이로플루오렌 잔텐닐 유도체 화합물:
<화학식 a>
상기 식에서,
R1 , 내지 R3 은, 각각 독립적으로, 수소, 중수소, 할로겐, C1~C12의 알킬기, C1~C12의 알콕시, 아미노, 니트릴, C2~C12의 아실, C3~C12의 실릴기, 치환 또는 비치환된 C6-C60의 아릴기, 또는 치환 또는 비치환된 C3~C60의 헤테로아릴기이고, 상기 R1 , 내지 R3 이 치환된 경우의 치환기는 C1~C30의 알킬기, C6~C30의 아릴기, C5~C30의 헤테로 아릴기 및 이들의 조합으로 이루어진 군으로부터 선택된 적어도 하나이고,
L은 단일결합, 치환 또는 비치환된 C6~C30의 아릴렌기, 치환 또는 비치환된 C5~C30의 헤테로아릴렌기 및 이들의 조합으로 이루어진 군으로부터 선택된 하나이고, 상기 L이 치환된 경우의 치환기는 C6~C30의 아릴기, C5~C30의 헤테로 아릴기이고,
상기 x 는, 상기 화학식 1 중 복수 개 포함되어 서로 동일하거나, 상이하고, 각각 독립적으로, 탄소 또는 질소이고,
Ar1 및 Ar2 는, 각각 독립적으로, 치환 또는 비치환된 C6-C60의 아릴기, 치환 또는 비치환된 C3-C60의 헤테로아릴기이고, 상기 Ar1 및 Ar2가 치환된 경우의 치환기는 C1~C30의 알킬기, C6~C30의 아릴기, C5~C30의 헤테로 아릴기 및 이들의 조합으로 이루어진 군으로부터 선택된 적어도 하나이다.
- 제1항 또는 제2항에 있어서,
상기 L은, 단일결합, 치환 또는 비치환된 페닐렌기, 치환 또는 비치환된 비페닐렌기, 치환 또는 비치환된 트리페닐렌기, 치환기를 갖는 플루오렌기 또는 스파이로플루오렌기이고,
상기 L의 정의 중 상기 페닐렌기, 상기 비페닐렌기 및 상기 트리페닐렌기가 치환된 경우의 치환기는 C6~C30의 아릴기, C5~C30의 헤테로 아릴기 및 이들의 조합으로 이루어진 군으로부터 선택된 적어도 하나이고,
상기 치환기를 갖는 플루오렌기의 상기 치환기는 수소, 중수소, 할로겐, C1~C12의 알킬기, C1~C12의 알콕시, 아미노, 니트릴, C2~C12의 아실, C3~C12의 실릴기, 치환 또는 비치환된 C6-C60의 아릴기, 치환 또는 비치환된 C3~C60의 헤테로아릴기 및 이들의 조합으로 이루어진 군으로부터 선택된 적어도 하나이고,
스파이로플루오렌 잔텐닐 유도체 화합물:
- 제1항 또는 제2항에 있어서,
상기 Ar1 및 상기 Ar2는, 각각 독립적으로, 페닐기, 톨릴기, 1-나프틸기, 2-나프틸기, 비페닐기, 페난트렌기, 플루오렌기, 스파이로플루오렌기, 벤조티오펜기, 벤조퓨란기, 디벤조퓨란기, 디벤조티오펜기, 터페닐기, 피리딜기 또는 페닐피리딜기인
스파이로플루오렌 잔텐닐 유도체 화합물.
- 제1항 또는 제2항에 있어서,
상기 스파이로플루오렌 잔텐닐 유도체 화합물은 유기전기발광소자용 재료 중 발광층, 정공차단층 전자수송층 또는 전자주입층 물질로 사용되는
스파이로플루오렌 잔텐닐 유도체 화합물.
- 음극과 양극 사이에 적어도 하나의 유기 박막층이 협지되어 있는 유기 전계 발광 소자에 있어서, 상기 유기 박막층은 적어도 하나의 발광층을 포함하는 다층 구조이고, 상기 발광층 또는 상기 발광층 이외의 상기 유기 박막층 내의 적어도 하나의 층이 제1항 또는 제2항에 따른 상기 스파이로플루오렌 잔텐닐 유도체 화합물을 단독 또는 2종 이상의 혼합물을 포함하는 유기 전계 발광 소자.
- 제8항에 있어서,
상기 유기 박막층은 음극과 발광층 사이에 개재되며, 전자주입층, 전자수송층, 전자주입 기능 및 전자수송 기능을 동시에 갖는 기능층, 버퍼층 및 정공차단층 중 적어도 하나를 포함한 전자 수송 영역을 포함하는
유기 전계 발광 소자.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020160171598A KR20180069423A (ko) | 2016-12-15 | 2016-12-15 | 스파이로플루오렌 잔텐닐 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| CN201711279160.4A CN108218842A (zh) | 2016-12-15 | 2017-12-06 | 螺芴氧杂蒽基衍生物化合物及包含其的有机电致发光器件 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020160171598A KR20180069423A (ko) | 2016-12-15 | 2016-12-15 | 스파이로플루오렌 잔텐닐 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20180069423A true KR20180069423A (ko) | 2018-06-25 |
Family
ID=62653958
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020160171598A Ceased KR20180069423A (ko) | 2016-12-15 | 2016-12-15 | 스파이로플루오렌 잔텐닐 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 |
Country Status (2)
| Country | Link |
|---|---|
| KR (1) | KR20180069423A (ko) |
| CN (1) | CN108218842A (ko) |
Cited By (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20190106801A (ko) * | 2018-03-09 | 2019-09-18 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 발광 소자 |
| WO2020022811A1 (ko) * | 2018-07-25 | 2020-01-30 | 주식회사 엘지화학 | 유기발광소자 |
| WO2020111762A1 (ko) * | 2018-11-27 | 2020-06-04 | 주식회사 엘지화학 | 유기 발광 소자 |
| KR20200090674A (ko) * | 2017-06-30 | 2020-07-29 | 주식회사 엘지화학 | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20210047251A (ko) * | 2019-10-21 | 2021-04-29 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기발광 소자 |
| WO2021080253A1 (ko) * | 2019-10-21 | 2021-04-29 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기발광 소자 |
| WO2021246698A1 (ko) * | 2020-06-05 | 2021-12-09 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물, 이를 이용한 유기전기소자 및 그 전자 장치 |
| WO2024117767A1 (ko) * | 2022-12-02 | 2024-06-06 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기 발광 소자 |
| US12202838B2 (en) | 2018-11-19 | 2025-01-21 | Lg Chem, Ltd. | Heterocyclic compound and organic light emitting device comprising same |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101755986B1 (ko) * | 2016-02-23 | 2017-07-07 | 주식회사 엘지화학 | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| CN115884652A (zh) * | 2022-12-09 | 2023-03-31 | 湖北长江新型显示产业创新中心有限公司 | 一种基于螺芴氧杂蒽的有机发光器件和面板 |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101440082A (zh) * | 2008-12-12 | 2009-05-27 | 南京邮电大学 | 螺芴氧杂蒽材料及其制备和应用方法 |
| KR102357439B1 (ko) * | 2012-02-14 | 2022-02-08 | 메르크 파텐트 게엠베하 | 유기 전계발광 소자용 스피로비플루오렌 화합물 |
| CN103193755A (zh) * | 2013-03-28 | 2013-07-10 | 哈尔滨工程大学 | 螺芴氧杂蒽酚化合物及其制备方法 |
| KR102285381B1 (ko) * | 2014-10-22 | 2021-08-04 | 삼성디스플레이 주식회사 | 화합물 및 이를 포함한 유기 발광 소자 |
| KR101755986B1 (ko) * | 2016-02-23 | 2017-07-07 | 주식회사 엘지화학 | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
-
2016
- 2016-12-15 KR KR1020160171598A patent/KR20180069423A/ko not_active Ceased
-
2017
- 2017-12-06 CN CN201711279160.4A patent/CN108218842A/zh active Pending
Cited By (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20200090674A (ko) * | 2017-06-30 | 2020-07-29 | 주식회사 엘지화학 | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20190106801A (ko) * | 2018-03-09 | 2019-09-18 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 발광 소자 |
| US12004423B2 (en) | 2018-07-25 | 2024-06-04 | Lg Chem, Ltd. | Organic light emitting device |
| WO2020022811A1 (ko) * | 2018-07-25 | 2020-01-30 | 주식회사 엘지화학 | 유기발광소자 |
| US12202838B2 (en) | 2018-11-19 | 2025-01-21 | Lg Chem, Ltd. | Heterocyclic compound and organic light emitting device comprising same |
| WO2020111762A1 (ko) * | 2018-11-27 | 2020-06-04 | 주식회사 엘지화학 | 유기 발광 소자 |
| CN112585775A (zh) * | 2018-11-27 | 2021-03-30 | 株式会社Lg化学 | 有机发光器件 |
| US12120949B2 (en) | 2018-11-27 | 2024-10-15 | Lg Chem, Ltd. | Organic light emitting diode |
| WO2021080253A1 (ko) * | 2019-10-21 | 2021-04-29 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기발광 소자 |
| CN114174286B (zh) * | 2019-10-21 | 2024-03-05 | 株式会社Lg化学 | 新型化合物及包含其的有机发光器件 |
| CN114174286A (zh) * | 2019-10-21 | 2022-03-11 | 株式会社Lg化学 | 新型化合物及包含其的有机发光器件 |
| KR20210047251A (ko) * | 2019-10-21 | 2021-04-29 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기발광 소자 |
| WO2021246698A1 (ko) * | 2020-06-05 | 2021-12-09 | 덕산네오룩스 주식회사 | 유기전기 소자용 화합물, 이를 이용한 유기전기소자 및 그 전자 장치 |
| WO2024117767A1 (ko) * | 2022-12-02 | 2024-06-06 | 주식회사 엘지화학 | 신규한 화합물 및 이를 이용한 유기 발광 소자 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN108218842A (zh) | 2018-06-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20180069423A (ko) | 스파이로플루오렌 잔텐닐 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR101719173B1 (ko) | 유기 전계 발광 소자 | |
| KR101910498B1 (ko) | 신규한 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR101763995B1 (ko) | 방향족 아민 유도체, 유기 전기발광 소자 및 전자 기기 | |
| JP5723764B2 (ja) | 有機電界発光素子 | |
| TWI471405B (zh) | A phosphorescent element material, and an organic electroluminescent device using the same | |
| KR101897632B1 (ko) | 디아릴플루오렌 아민 유도체 유기화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR101529878B1 (ko) | 유기 전기 발광 조성물 및 이를 포함하는 유기 전기 발광 소자 | |
| KR20190050525A (ko) | 2,3-치환된 나프틸아민 유도체 유기발광 화합물 및 유기 전계 발광 소자 | |
| KR102001425B1 (ko) | 3-디벤조퓨란닐 트리아진 유도체 유기화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR101513696B1 (ko) | 새로운 바이카바졸계 유기전계발광소자용 화합물 및 그를 포함하는 유기전계발광소자 | |
| KR102263822B1 (ko) | 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR20170039020A (ko) | 유기 전계발광 화합물 및 유기 전계발광 소자 | |
| KR20200107782A (ko) | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 | |
| WO2021085460A1 (ja) | ピロメテンホウ素錯体を含む発光素子材料、発光素子、表示装置および照明装置 | |
| CN101679857B (zh) | 发光元件材料和发光元件 | |
| KR101764907B1 (ko) | 2-펜안트렌아민 카바졸 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR101764908B1 (ko) | 2-펜안트렌 카바졸 유도체 화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR20210036253A (ko) | 화합물 및 이를 포함하는 유기 발광 소자 | |
| KR101939152B1 (ko) | 플루오렌닐 트리아진 유도체 유기화합물 및 이를 포함하는 유기 전계 발광 소자 | |
| KR20170060831A (ko) | 유기전계발광소자용 화합물 및 그를 포함하는 유기전계발광소자 | |
| KR101815653B1 (ko) | 아자보라디벤조크라이센 유도체 유기 발광 화합물, 잉크 조성물 및 유기 발광 소자 | |
| KR102107086B1 (ko) | 신규한 화합물 및 이를 포함하는 유기발광 소자 | |
| CN107915722B (zh) | 化合物及包含其的有机发光元件 | |
| KR20200095710A (ko) | 유기 전기 발광 조성물 및 이를 포함하는 유기 전기 발광 소자 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20161215 |
|
| PA0201 | Request for examination | ||
| N231 | Notification of change of applicant | ||
| PN2301 | Change of applicant |
Patent event date: 20170403 Comment text: Notification of Change of Applicant Patent event code: PN23011R01D |
|
| PG1501 | Laying open of application | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20180820 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20181101 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20180820 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |