KR20110036075A - 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 - Google Patents
안정한 주입 가능 수중유형 도세타셀 나노에멀젼 Download PDFInfo
- Publication number
- KR20110036075A KR20110036075A KR1020117001821A KR20117001821A KR20110036075A KR 20110036075 A KR20110036075 A KR 20110036075A KR 1020117001821 A KR1020117001821 A KR 1020117001821A KR 20117001821 A KR20117001821 A KR 20117001821A KR 20110036075 A KR20110036075 A KR 20110036075A
- Authority
- KR
- South Korea
- Prior art keywords
- composition
- docetacell
- oil
- synthetic triglyceride
- triglyceride oil
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 239000007908 nanoemulsion Substances 0.000 title claims abstract description 73
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 title claims abstract description 24
- 239000000203 mixture Substances 0.000 claims abstract description 143
- 150000005846 sugar alcohols Polymers 0.000 claims abstract description 21
- 206010020751 Hypersensitivity Diseases 0.000 claims abstract description 9
- 206010016807 Fluid retention Diseases 0.000 claims abstract description 8
- 239000008215 water for injection Substances 0.000 claims abstract description 8
- 239000002577 cryoprotective agent Substances 0.000 claims abstract description 7
- 238000007911 parenteral administration Methods 0.000 claims abstract description 7
- 239000000839 emulsion Substances 0.000 claims description 42
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 claims description 37
- 239000008346 aqueous phase Substances 0.000 claims description 26
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 23
- 239000012071 phase Substances 0.000 claims description 23
- 238000000034 method Methods 0.000 claims description 19
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 16
- 230000008569 process Effects 0.000 claims description 16
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 claims description 15
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 claims description 14
- 239000002245 particle Substances 0.000 claims description 11
- 239000008344 egg yolk phospholipid Substances 0.000 claims description 9
- 238000004519 manufacturing process Methods 0.000 claims description 9
- 239000000243 solution Substances 0.000 claims description 9
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 claims description 8
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 claims description 8
- WGCNASOHLSPBMP-UHFFFAOYSA-N hydroxyacetaldehyde Natural products OCC=O WGCNASOHLSPBMP-UHFFFAOYSA-N 0.000 claims description 8
- 239000005635 Caprylic acid (CAS 124-07-2) Substances 0.000 claims description 7
- 229920002505 N-(Carbonyl-Methoxypolyethylene Glycol 2000)-1,2-Distearoyl-Sn-Glycero-3-Phosphoethanolamine Polymers 0.000 claims description 7
- 229930006000 Sucrose Natural products 0.000 claims description 7
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims description 7
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 7
- 239000000194 fatty acid Substances 0.000 claims description 7
- 229930195729 fatty acid Natural products 0.000 claims description 7
- 150000004665 fatty acids Chemical class 0.000 claims description 7
- 229940071238 n-(carbonyl-methoxypolyethylene glycol 2000)-1,2-distearoyl-sn-glycero-3-phosphoethanolamine Drugs 0.000 claims description 7
- 229960002446 octanoic acid Drugs 0.000 claims description 7
- 239000005720 sucrose Substances 0.000 claims description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 claims description 6
- BAECOWNUKCLBPZ-HIUWNOOHSA-N Triolein Natural products O([C@H](OCC(=O)CCCCCCC/C=C\CCCCCCCC)COC(=O)CCCCCCC/C=C\CCCCCCCC)C(=O)CCCCCCC/C=C\CCCCCCCC BAECOWNUKCLBPZ-HIUWNOOHSA-N 0.000 claims description 6
- PHYFQTYBJUILEZ-UHFFFAOYSA-N Trioleoylglycerol Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC(OC(=O)CCCCCCCC=CCCCCCCCC)COC(=O)CCCCCCCC=CCCCCCCCC PHYFQTYBJUILEZ-UHFFFAOYSA-N 0.000 claims description 6
- 238000002360 preparation method Methods 0.000 claims description 6
- PHYFQTYBJUILEZ-IUPFWZBJSA-N triolein Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC(OC(=O)CCCCCCC\C=C/CCCCCCCC)COC(=O)CCCCCCC\C=C/CCCCCCCC PHYFQTYBJUILEZ-IUPFWZBJSA-N 0.000 claims description 6
- 229940117972 triolein Drugs 0.000 claims description 6
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 claims description 5
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 claims description 5
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 claims description 5
- 238000004108 freeze drying Methods 0.000 claims description 5
- 229940057917 medium chain triglycerides Drugs 0.000 claims description 5
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 claims description 4
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 claims description 4
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 claims description 4
- 239000005639 Lauric acid Substances 0.000 claims description 4
- 239000005642 Oleic acid Substances 0.000 claims description 4
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 claims description 4
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 claims description 4
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 claims description 4
- 239000008347 soybean phospholipid Substances 0.000 claims description 4
- 238000003756 stirring Methods 0.000 claims description 4
- TUNFSRHWOTWDNC-HKGQFRNVSA-N tetradecanoic acid Chemical compound CCCCCCCCCCCCC[14C](O)=O TUNFSRHWOTWDNC-HKGQFRNVSA-N 0.000 claims description 4
- 229940093609 tricaprylin Drugs 0.000 claims description 4
- VLPFTAMPNXLGLX-UHFFFAOYSA-N trioctanoin Chemical compound CCCCCCCC(=O)OCC(OC(=O)CCCCCCC)COC(=O)CCCCCCC VLPFTAMPNXLGLX-UHFFFAOYSA-N 0.000 claims description 4
- PZNPLUBHRSSFHT-RRHRGVEJSA-N 1-hexadecanoyl-2-octadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)O[C@@H](COP([O-])(=O)OCC[N+](C)(C)C)COC(=O)CCCCCCCCCCCCCCC PZNPLUBHRSSFHT-RRHRGVEJSA-N 0.000 claims description 3
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 claims description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 claims description 3
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 3
- 229930195725 Mannitol Natural products 0.000 claims description 3
- 239000002253 acid Substances 0.000 claims description 3
- 239000008101 lactose Substances 0.000 claims description 3
- 239000000594 mannitol Substances 0.000 claims description 3
- 235000010355 mannitol Nutrition 0.000 claims description 3
- 229910052757 nitrogen Inorganic materials 0.000 claims description 3
- LVNGJLRDBYCPGB-LDLOPFEMSA-N (R)-1,2-distearoylphosphatidylethanolamine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[NH3+])OC(=O)CCCCCCCCCCCCCCCCC LVNGJLRDBYCPGB-LDLOPFEMSA-N 0.000 abstract description 10
- 150000003626 triacylglycerols Chemical class 0.000 abstract description 5
- 239000003921 oil Substances 0.000 description 49
- 235000019198 oils Nutrition 0.000 description 49
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 30
- 238000009472 formulation Methods 0.000 description 28
- 150000003904 phospholipids Chemical class 0.000 description 14
- 239000003814 drug Substances 0.000 description 10
- 239000013022 formulation composition Substances 0.000 description 10
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 9
- 239000004530 micro-emulsion Substances 0.000 description 9
- 229940079593 drug Drugs 0.000 description 8
- 229940063683 taxotere Drugs 0.000 description 8
- 239000007788 liquid Substances 0.000 description 7
- KILNVBDSWZSGLL-KXQOOQHDSA-N 1,2-dihexadecanoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCC KILNVBDSWZSGLL-KXQOOQHDSA-N 0.000 description 6
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 6
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 description 6
- 235000011187 glycerol Nutrition 0.000 description 6
- 229920001223 polyethylene glycol Polymers 0.000 description 6
- 239000004094 surface-active agent Substances 0.000 description 6
- 241001465754 Metazoa Species 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 238000001035 drying Methods 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 150000004684 trihydrates Chemical class 0.000 description 5
- 206010028980 Neoplasm Diseases 0.000 description 4
- 229930012538 Paclitaxel Natural products 0.000 description 4
- 241000700159 Rattus Species 0.000 description 4
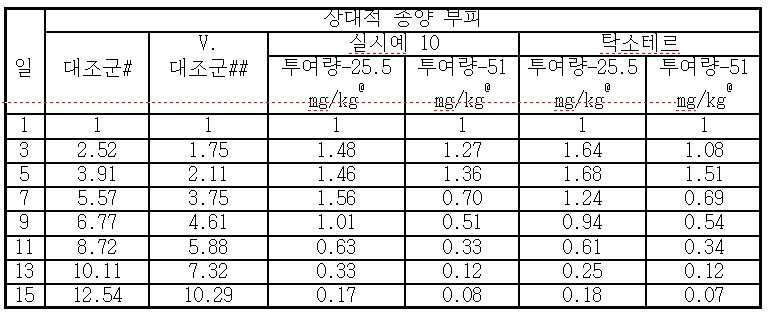
- 230000000259 anti-tumor effect Effects 0.000 description 4
- 230000000052 comparative effect Effects 0.000 description 4
- 239000006184 cosolvent Substances 0.000 description 4
- 238000001802 infusion Methods 0.000 description 4
- 229960001592 paclitaxel Drugs 0.000 description 4
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 4
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 description 4
- 229920000053 polysorbate 80 Polymers 0.000 description 4
- 229940068968 polysorbate 80 Drugs 0.000 description 4
- 239000002904 solvent Substances 0.000 description 4
- 241000894007 species Species 0.000 description 4
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 4
- 231100000419 toxicity Toxicity 0.000 description 4
- 230000001988 toxicity Effects 0.000 description 4
- 235000015112 vegetable and seed oil Nutrition 0.000 description 4
- 239000008158 vegetable oil Substances 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- 239000004471 Glycine Substances 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- 241000699666 Mus <mouse, genus> Species 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- BCKXLBQYZLBQEK-KVVVOXFISA-M Sodium oleate Chemical compound [Na+].CCCCCCCC\C=C/CCCCCCCC([O-])=O BCKXLBQYZLBQEK-KVVVOXFISA-M 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 238000010790 dilution Methods 0.000 description 3
- 239000012895 dilution Substances 0.000 description 3
- 230000000694 effects Effects 0.000 description 3
- 239000003995 emulsifying agent Substances 0.000 description 3
- 235000021588 free fatty acids Nutrition 0.000 description 3
- 238000000265 homogenisation Methods 0.000 description 3
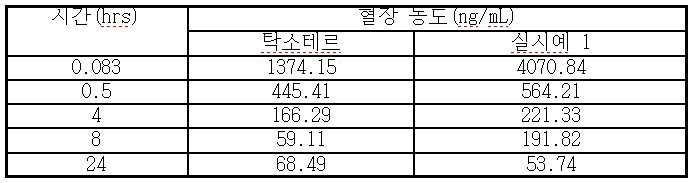
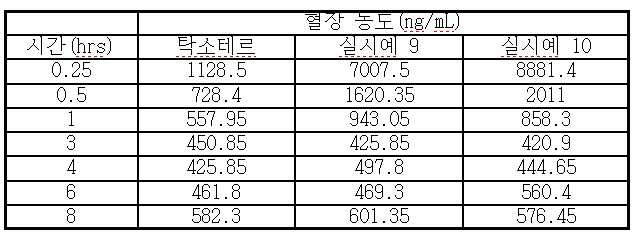
- 230000036470 plasma concentration Effects 0.000 description 3
- 238000001556 precipitation Methods 0.000 description 3
- 235000012424 soybean oil Nutrition 0.000 description 3
- 239000003549 soybean oil Substances 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- NRJAVPSFFCBXDT-HUESYALOSA-N 1,2-distearoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCCCCCC NRJAVPSFFCBXDT-HUESYALOSA-N 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- 231100000403 acute toxicity Toxicity 0.000 description 2
- 230000007059 acute toxicity Effects 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 239000000560 biocompatible material Substances 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 150000001875 compounds Chemical class 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 2
- 238000007865 diluting Methods 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 238000004128 high performance liquid chromatography Methods 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 2
- 239000000693 micelle Substances 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000000546 pharmaceutical excipient Substances 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 231100000279 safety data Toxicity 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- 239000003981 vehicle Substances 0.000 description 2
- LVNGJLRDBYCPGB-UHFFFAOYSA-N 1,2-distearoylphosphatidylethanolamine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(COP([O-])(=O)OCC[NH3+])OC(=O)CCCCCCCCCCCCCCCCC LVNGJLRDBYCPGB-UHFFFAOYSA-N 0.000 description 1
- BJQHLKABXJIVAM-BGYRXZFFSA-N 1-o-[(2r)-2-ethylhexyl] 2-o-[(2s)-2-ethylhexyl] benzene-1,2-dicarboxylate Chemical compound CCCC[C@H](CC)COC(=O)C1=CC=CC=C1C(=O)OC[C@H](CC)CCCC BJQHLKABXJIVAM-BGYRXZFFSA-N 0.000 description 1
- JKMHFZQWWAIEOD-UHFFFAOYSA-N 2-[4-(2-hydroxyethyl)piperazin-1-yl]ethanesulfonic acid Chemical compound OCC[NH+]1CCN(CCS([O-])(=O)=O)CC1 JKMHFZQWWAIEOD-UHFFFAOYSA-N 0.000 description 1
- VLHWNGXLXZPNOO-UHFFFAOYSA-N 2-[4-[2-(2,3-dihydro-1H-inden-2-ylamino)pyrimidin-5-yl]-3-(2-morpholin-4-ylethyl)pyrazol-1-yl]-1-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethanone Chemical compound C1C(CC2=CC=CC=C12)NC1=NC=C(C=N1)C=1C(=NN(C=1)CC(=O)N1CC2=C(CC1)NN=N2)CCN1CCOCC1 VLHWNGXLXZPNOO-UHFFFAOYSA-N 0.000 description 1
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 1
- 102000009027 Albumins Human genes 0.000 description 1
- 108010088751 Albumins Proteins 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 239000005632 Capric acid (CAS 334-48-5) Substances 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 229920002307 Dextran Polymers 0.000 description 1
- BJQHLKABXJIVAM-UHFFFAOYSA-N Diethylhexyl phthalate Natural products CCCCC(CC)COC(=O)C1=CC=CC=C1C(=O)OCC(CC)CCCC BJQHLKABXJIVAM-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 239000007995 HEPES buffer Substances 0.000 description 1
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 241000700157 Rattus norvegicus Species 0.000 description 1
- 238000011579 SCID mouse model Methods 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 239000002535 acidifier Substances 0.000 description 1
- 229940095602 acidifiers Drugs 0.000 description 1
- 231100000215 acute (single dose) toxicity testing Toxicity 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 239000000908 ammonium hydroxide Substances 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 239000007975 buffered saline Substances 0.000 description 1
- 208000035269 cancer or benign tumor Diseases 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- STORWMDPIHOSMF-UHFFFAOYSA-N decanoic acid;octanoic acid;propane-1,2,3-triol Chemical compound OCC(O)CO.CCCCCCCC(O)=O.CCCCCCCCCC(O)=O STORWMDPIHOSMF-UHFFFAOYSA-N 0.000 description 1
- PWEOPMBMTXREGV-UHFFFAOYSA-N decanoic acid;octanoic acid;propane-1,2-diol Chemical compound CC(O)CO.CCCCCCCC(O)=O.CCCCCCCC(O)=O.CCCCCCCCCC(O)=O.CCCCCCCCCC(O)=O PWEOPMBMTXREGV-UHFFFAOYSA-N 0.000 description 1
- 230000006866 deterioration Effects 0.000 description 1
- 239000008121 dextrose Substances 0.000 description 1
- 238000011038 discontinuous diafiltration by volume reduction Methods 0.000 description 1
- 229960003668 docetaxel Drugs 0.000 description 1
- 238000012377 drug delivery Methods 0.000 description 1
- 238000002296 dynamic light scattering Methods 0.000 description 1
- 230000032050 esterification Effects 0.000 description 1
- 238000005886 esterification reaction Methods 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000010408 film Substances 0.000 description 1
- 235000019253 formic acid Nutrition 0.000 description 1
- 238000007710 freezing Methods 0.000 description 1
- 230000008014 freezing Effects 0.000 description 1
- 125000005456 glyceride group Chemical group 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 238000009775 high-speed stirring Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- FZWBNHMXJMCXLU-BLAUPYHCSA-N isomaltotriose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C=O)O1 FZWBNHMXJMCXLU-BLAUPYHCSA-N 0.000 description 1
- 230000004576 lipid-binding Effects 0.000 description 1
- 230000004130 lipolysis Effects 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 239000004973 liquid crystal related substance Substances 0.000 description 1
- 239000008176 lyophilized powder Substances 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 238000012856 packing Methods 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 229940085991 phosphate ion Drugs 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 238000004094 preconcentration Methods 0.000 description 1
- 238000009101 premedication Methods 0.000 description 1
- 238000010926 purge Methods 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- NLJMYIDDQXHKNR-UHFFFAOYSA-K sodium citrate Chemical compound O.O.[Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O NLJMYIDDQXHKNR-UHFFFAOYSA-K 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 230000002269 spontaneous effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 229940124597 therapeutic agent Drugs 0.000 description 1
- 230000001225 therapeutic effect Effects 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 231100000041 toxicology testing Toxicity 0.000 description 1
- 150000005691 triesters Chemical class 0.000 description 1
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 1
- 235000013311 vegetables Nutrition 0.000 description 1
- 231100000925 very toxic Toxicity 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/337—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having four-membered rings, e.g. taxol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/107—Emulsions ; Emulsion preconcentrates; Micelles
- A61K9/1075—Microemulsions or submicron emulsions; Preconcentrates or solids thereof; Micelles, e.g. made of phospholipids or block copolymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/14—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles
- A61K9/19—Particulate form, e.g. powders, Processes for size reducing of pure drugs or the resulting products, Pure drug nanoparticles lyophilised, i.e. freeze-dried, solutions or dispersions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Dispersion Chemistry (AREA)
- Dermatology (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- Medicinal Preparation (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
Abstract
Description
Claims (35)
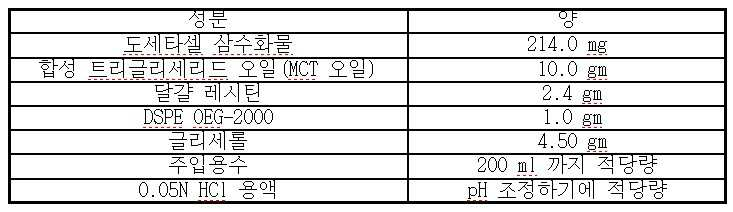
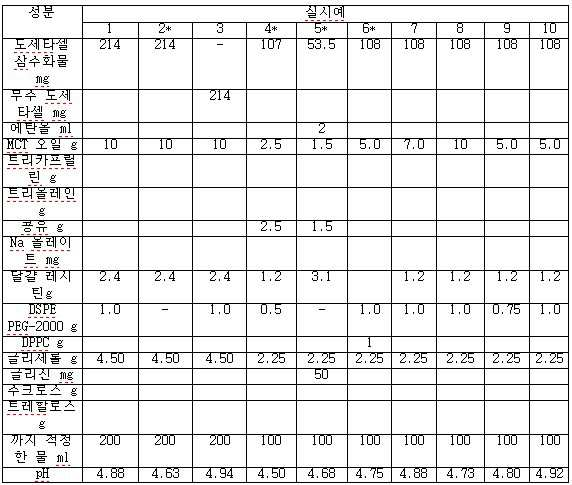
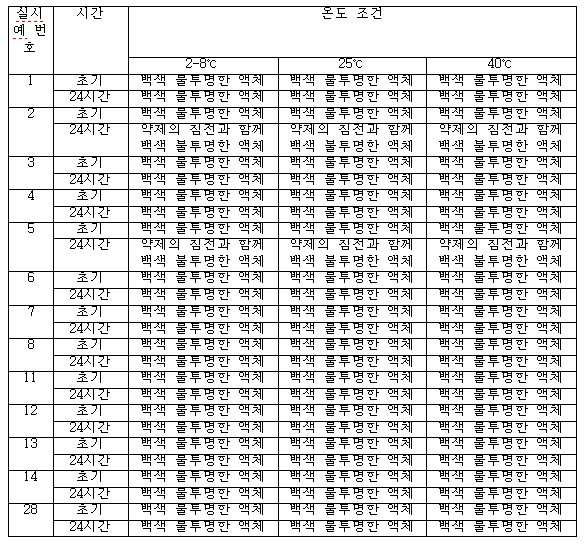
- 도세타셀, 합성 트리글리세리드 오일, N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민, 정제된 천연 포스파티드, 다가 알코올(polyhydric alcohol) 및 주입용수를 포함하는, 과민성 반응 및 체액 저류가 없는 pH 4.0 내지 5.5의 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 조성물.
- 제 1 항에 있어서, 상기 도세타셀은 상기 조성물의 0.05% 내지 2.0% w/v인 것인 조성물.
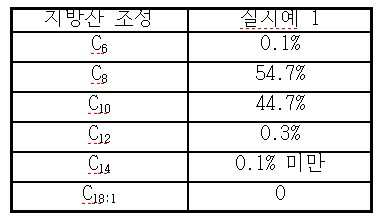
- 제 1 항에 있어서, 상기 합성 트리글리세리드 오일은 카프로산, 카프릴산, 카푸르산, 라우르산, 미리스트산, 올레산 및 이들의 혼합물로부터 선택된 지방산을 가지는 것인 조성물.
- 제 1 항에 있어서, 상기 합성 트리글리세리드 오일은 카프릴산을 85 내지 100 중량% 가지는 것인 조성물.
- 제 3 항에 있어서, 상기 합성 트리글리세리드 오일은 중간쇄 트리글리세리드, 트리카프릴린(Tricaprylin) 및 트리올레인(Triolein) 및 이들의 혼합물로부터 선택되는 것인 조성물.
- 제 1 항에 있어서, 상기 정제된 천연 포스파티드는 정제된 달걀 레시틴, 정제된 콩 레시틴 및 이들의 혼합물로부터 선택되는 것인 조성물.
- 제 1 항에 있어서, 상기 다가 알코올은 글리세롤, 프로필렌 글리콜 및 이들의 혼합물로부터 선택되는 것인 조성물.
- 제 1 항에 있어서, 합성 트리글리세리드 오일 대 도세타셀의 중량 비는 1:1 내지 100:1인 것인 조성물.
- 제 1 항에 있어서, 합성 트리글리세리드 오일 대 도세타셀의 중량 비는 10:1 내지 50:1인 것인 조성물.
- 제 1 항에 있어서, 합성 트리글리세리드 오일 대 N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민의 중량 비는 1:1 내지 100:1인 것인 조성물.
- 제 1 항에 있어서, 합성 트리글리세리드 오일 대 N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민의 중량 비는 5:1 내지 20:1인 것인 조성물.
- 제 1 항에 있어서, 합성 트리글리세리드 오일 대 정제된 천연 포스파티드의 중량 비는 4:1 내지 40:1인 것인 조성물.
- 제 1 항에 있어서, 합성 트리글리세리드 오일 대 정제된 천연 포스파티드의 중량 비는 7:1 내지 20:1인 것인 조성물.
- 제 1 항에 있어서, 상기 다가 알코올의 함량은 상기 조성물의 0.5 내지 3% w/v인 것인 조성물.
- 다음 단계를 포함하는, 제 1 항의 도세타셀 나노에멀젼 조성물의 제조 방법:
i) 도세타셀을 합성 트리글리세리드 오일에 용해시키고, 초음파 처리하거나(sonication) 가열하여 투명한 용액을 얻어 오일 상을 형성하고;
ii) 다가 알코올을 주입용수(water for injection)에 용해시켜 수성 상을 형성하며;
iii) N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민을 단계 i)의 오일 상 또는 단계 ii)의 수성 상에 분산시키거나, 일부는 단계 i)의 오일 상에 및 일부는 단계 ii)의 수성 상에 분산시키고;
iv) 정제된 천연 포스파티드를 단계 ii)에서 제조된 수성 상에 분산시키며;
v) 오일 상을 수성 상에 교반 하에 첨가하여 조(coarse) 에멀젼을 얻고;
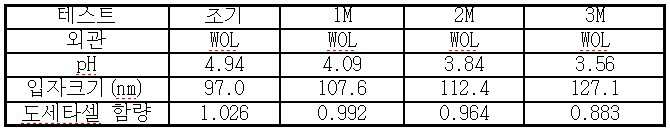
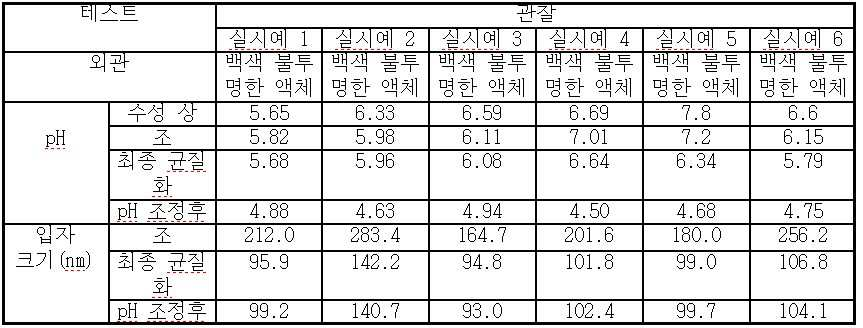
vi) 조 에멀젼을 균질화시켜 200 nm 미만, 바람직하게는 100 nm 미만의 평균 입자 크기를 수득하며;
vii) 수득된 에멀젼의 pH를 단계 v) 또는 단계 vi)에서 4.0 내지 5.5로 조정하고;
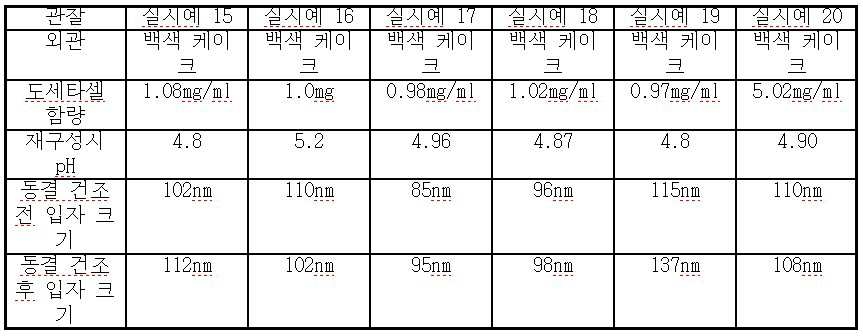
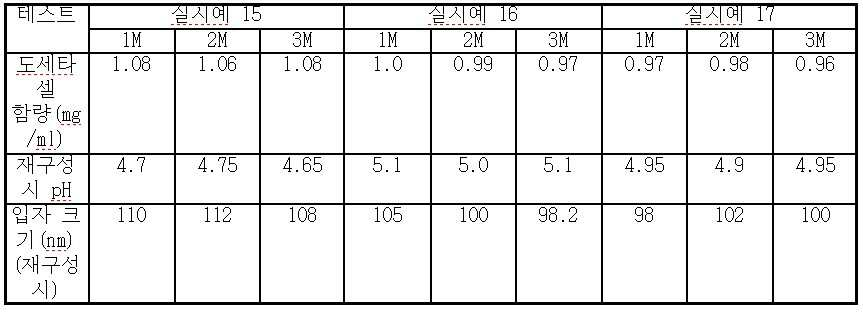
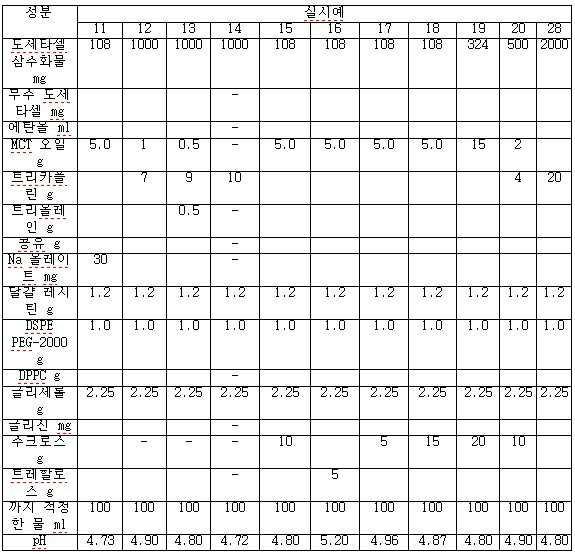
viii) 단계 vii)의 종료시 수득되는 나노에멀젼을 0.2 μ 필터를 통해 무균적으로 여과하여 질소 하에 바이알에 채운다. - 도세타셀, 합성 트리글리세리드 오일, N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민, 정제된 천연 포스파티드, 다가 알코올(polyhydric alcohol), 및 수크로스, 트레할로스, 만니톨, 락토스 또는 이들의 혼합물로부터 선택된 동결 방지제를 포함하고, 과민성 반응 및 체액 저류가 없으며, 재구성(reconstitution) 시, pH 4.0 내지 5.5인, 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 조성물을 형성하는 비경구 투여용 동결 건조 조성물.
- 제 16 항에 있어서, 상기 도세타셀은 동결 건조 전 0.05% 내지 2.0% w/v인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 상기 합성 트리글리세리드 오일은 카프로산, 카프릴산, 카푸르산, 라우르산, 미리스트산, 올레산 및 이들의 혼합물로부터 선택된 지방산을 가지는 것인 동결 건조 조성물.
- 제 16 항에 있어서, 상기 합성 트리글리세리드 오일은 카프릴산을 85 내지 100 중량% 가지는 것인 동결 건조 조성물.
- 제 18 항에 있어서, 상기 합성 트리글리세리드 오일은 중간쇄 트리글리세리드, 트리카프릴린(Tricaprylin) 및 트리올레인(Triolein) 및 이들의 혼합물로부터 선택되는 것인 동결 건조 조성물.
- 제 16 항에 있어서, 상기 정제된 천연 포스파티드는 정제된 달걀 레시틴, 정제된 콩 레시틴 및 이들의 혼합물로부터 선택되는 것인 동결 건조 조성물.
- 제 16 항에 있어서, 상기 다가 알코올은 글리세롤, 프로필렌 글리콜 및 이들의 혼합물로부터 선택되는 것인 동결 건조 조성물.
- 제 16 항에 있어서, 합성 트리글리세리드 오일 대 도세타셀의 중량 비는 1:1 내지 100:1인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 합성 트리글리세리드 오일 대 도세타셀의 중량 비는 10:1 내지 50:1인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 합성 트리글리세리드 오일 대 N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민의 중량 비는 1:1 내지 100:1인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 합성 트리글리세리드 오일 대 N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민의 중량 비는 5:1 내지 20:1인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 합성 트리글리세리드 오일 대 정제된 천연 포스파티드의 중량 비는 4:1 내지 40:1인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 합성 트리글리세리드 오일 대 정제된 천연 포스파티드의 중량 비는 7:1 내지 20:1인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 상기 다가 알코올 함량이 0.5 내지 3 중량%인 것인 동결 건조 조성물.
- 제 16 항에 있어서, 상기 수크로스의 함량은 20 중량% 이하인 것인 동결 건조 조성물.
- 다음 단계를 포함하는, 제 16 항의 동결 건조 조성물의 제조 방법:
i) 도세타셀을 합성 트리글리세리드 오일에 용해시키고, 초음파 처리하거나(sonication) 가열하여 투명한 용액을 얻어 오일 상을 형성하고;
ii) 다가 알코올 및 동결 방지제를 주입용수에 용해시켜 수성 상을 형성하며;
iii) N-(카보닐-메톡시폴리에틸렌 글리콜 2000)-1,2-디스테아로일-sn-글리세로-3-포스포에탄올아민을 단계 i)의 오일 상 또는 단계 ii)의 수성 상에 분산시키거나, 일부는 단계 i)의 오일 상에 및 일부는 단계 ii)의 수성 상에 분산시키고;
iv) 정제된 천연 포스파티드를 단계 ii)에서 제조된 수성 상에 분산시키며;
v) 오일 상을 수성 상에 교반 하에 첨가하여 조(coarse) 에멀젼을 얻고;
vi) 조 에멀젼을 균질화시켜 200 nm 미만, 바람직하게는 100 nm 미만의 평균 입자 크기를 수득하며;
vii) 수득된 에멀젼의 pH를 단계 v) 또는 단계 vi)에서 4.0 내지 5.5로 조정하고;
viii) 단계 vii)의 종료시 수득되는 나노에멀젼을 0.2 μ 필터를 통해 무균적으로 여과하여 바이알에 채우고 동결 건조시킨다. - 실질적으로 본 명세서(text) 및 실시예에 기술된 바와 같은, pH 4.0 내지 5.5의, 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 조성물.
- 실질적으로 본 명세서 및 실시예에 기술된 바와 같은, 도세타셀 나노에멀젼 조성물의 제조 방법.
- 실질적으로 본 명세서 및 실시예에 기술된 바와 같은, 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 조성물을 형성하는 비경구 투여용 동결 건조 조성물.
- 실질적으로 본 명세서 및 실시예에 기술된 바와 같은, 동결 건조 조성물의 제조 방법.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN1565MU2008 | 2008-07-23 | ||
| IN1565/MUM/2008 | 2008-07-23 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20110036075A true KR20110036075A (ko) | 2011-04-06 |
Family
ID=41527728
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020117001821A Ceased KR20110036075A (ko) | 2008-07-23 | 2009-07-22 | 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 |
Country Status (13)
| Country | Link |
|---|---|
| US (1) | US20110275705A1 (ko) |
| EP (1) | EP2317978A2 (ko) |
| JP (1) | JP5635504B2 (ko) |
| KR (1) | KR20110036075A (ko) |
| CN (1) | CN102105134B (ko) |
| AU (1) | AU2009280803B2 (ko) |
| BR (1) | BRPI0916535A2 (ko) |
| CA (1) | CA2731353A1 (ko) |
| EA (1) | EA201100069A1 (ko) |
| MX (1) | MX2011000795A (ko) |
| NZ (1) | NZ590730A (ko) |
| WO (1) | WO2010018596A2 (ko) |
| ZA (1) | ZA201100465B (ko) |
Families Citing this family (30)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5591434B2 (ja) | 2002-12-20 | 2014-09-17 | ゼリス ファーマシューティカルズ インコーポレイテッド | 皮内注射方法 |
| TWI438009B (zh) * | 2010-02-19 | 2014-05-21 | Teikoku Pharma Usa Inc | 紫杉烷前-乳劑調配物及其製造與使用之方法 |
| AU2011248449B2 (en) | 2010-05-03 | 2016-09-15 | Teikoku Pharma Usa, Inc. | Non-aqueous taxane pro-emulsion formulations and methods of making and using the same |
| CN102309445B (zh) * | 2010-07-06 | 2013-03-27 | 上海现代药物制剂工程研究中心有限公司 | 多西他赛静脉注射组合物及其制备方法 |
| US20120046225A1 (en) | 2010-07-19 | 2012-02-23 | The Regents Of The University Of Colorado, A Body Corporate | Stable glucagon formulations for the treatment of hypoglycemia |
| US9963368B2 (en) | 2010-11-15 | 2018-05-08 | Archer Daniels Midland Company | Microemulsions and uses thereof in dispersing catalysts |
| JP6030565B2 (ja) * | 2010-12-10 | 2016-11-24 | エヌエス テクノロジーズ プロプライエタリー リミテッドNs Technologies Pty Ltd | ミニエマルション、同ミニエマルションを形成するための方法および医薬品の製造における同ミニエマルションの使用 |
| AU2012225268B2 (en) | 2011-03-10 | 2016-10-20 | Xeris Pharmaceuticals, Inc. | Stable formulations for parenteral injection of peptide drugs |
| KR102117544B1 (ko) * | 2011-05-10 | 2020-06-02 | 아처 다니엘 미드랜드 캄파니 | 바이오계 화합물을 갖는 분산제 |
| WO2012156999A1 (en) * | 2011-05-19 | 2012-11-22 | Manu Chaudhary | Ready to use docetaxel formulation |
| CN103930096B (zh) | 2011-10-31 | 2017-05-31 | Xeris药物公司 | 用于治疗糖尿病的制剂 |
| US9125805B2 (en) | 2012-06-27 | 2015-09-08 | Xeris Pharmaceuticals, Inc. | Stable formulations for parenteral injection of small molecule drugs |
| JO3685B1 (ar) | 2012-10-01 | 2020-08-27 | Teikoku Pharma Usa Inc | صيغ التشتيت الجسيمي للتاكسين غير المائي وطرق استخدامها |
| HK1211203A1 (en) * | 2012-10-16 | 2016-05-20 | Inspyr Therapeutics, Inc. | Injectable cancer compositions |
| US9018162B2 (en) | 2013-02-06 | 2015-04-28 | Xeris Pharmaceuticals, Inc. | Methods for rapidly treating severe hypoglycemia |
| CN103315978B (zh) * | 2013-07-12 | 2014-12-03 | 上海市第八人民医院 | 多西紫杉醇干酏剂及其制备方法和应用 |
| EP3024441A4 (en) * | 2013-07-25 | 2017-04-19 | Nemucore Medical Innovations, Inc. | Nanoemulsions of hydrophobic platinum derivative |
| WO2015106094A1 (en) * | 2014-01-10 | 2015-07-16 | Atossa Genetics Inc. | Transpapillary methods and compositions for diagnosing and treating breast conditions |
| WO2016022831A1 (en) | 2014-08-06 | 2016-02-11 | Xeris Pharmaceuticals, Inc. | Syringes, kits, and methods for intracutaneous and/or subcutaneous injection of pastes |
| CN104626418B (zh) * | 2015-01-27 | 2016-10-05 | 天津现代职业技术学院 | 异型橡胶管用无硅脂型脱模剂 |
| CN106176599A (zh) * | 2015-05-06 | 2016-12-07 | 江苏天士力帝益药业有限公司 | 一种卡巴他赛脂肪乳注射剂及其制备方法 |
| US9649364B2 (en) | 2015-09-25 | 2017-05-16 | Xeris Pharmaceuticals, Inc. | Methods for producing stable therapeutic formulations in aprotic polar solvents |
| US11590205B2 (en) | 2015-09-25 | 2023-02-28 | Xeris Pharmaceuticals, Inc. | Methods for producing stable therapeutic glucagon formulations in aprotic polar solvents |
| CN109562065A (zh) * | 2016-06-07 | 2019-04-02 | 塔佳吉尼克斯公司 | 靶向癌细胞和癌干细胞的紫杉烷类药物的纳米乳剂组合物及其使用方法 |
| US10864280B2 (en) * | 2016-06-09 | 2020-12-15 | Der-Yang Tien | Nanodroplet compositions for the efficient delivery of anti-cancer agents |
| JP7097593B2 (ja) * | 2017-03-31 | 2022-07-08 | テクノガード株式会社 | 薬物を保持した脂肪粒子を含む非水系組成物およびその製造方法 |
| EP3630061B1 (en) | 2017-06-02 | 2024-04-03 | Xeris Pharmaceuticals, Inc. | Precipitation resistant small molecule drug formulations |
| CN111920782B (zh) * | 2019-05-13 | 2025-11-18 | 中国医学科学院药物研究所 | 复合脂质纳米囊组合物及其制备方法和应用 |
| WO2023048758A1 (en) * | 2021-09-22 | 2023-03-30 | Hdt Bio Corp. | Dried nanoparticle compositions |
| WO2024206239A1 (en) * | 2023-03-24 | 2024-10-03 | Insitu Biologics, Inc. | Sustained release cancer therapeutics formulations |
Family Cites Families (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5478860A (en) | 1993-06-04 | 1995-12-26 | Inex Pharmaceuticals Corp. | Stable microemulsions for hydrophobic compound delivery |
| GB9715759D0 (en) * | 1997-07-26 | 1997-10-01 | Danbiosyst Uk | New emulsion formulations |
| TW422706B (en) * | 1999-07-01 | 2001-02-21 | Wang Iz Rung | An oil-in-water emulsion type of paclitaxel |
| MXPA04012567A (es) * | 2002-06-11 | 2005-04-19 | Ethypharm Sa | Nanocapsulas lipidicas encubiertas, metodos para la preparacion de las mismas y uso de las mismas como un portador para principios activos. |
| US8557861B2 (en) * | 2004-09-28 | 2013-10-15 | Mast Therapeutics, Inc. | Low oil emulsion compositions for delivering taxoids and other insoluble drugs |
| TWI376239B (en) * | 2006-02-01 | 2012-11-11 | Andrew Xian Chen | Vitamin e succinate stabilized pharmaceutical compositions, methods for the preparation and the use thereof |
| WO2008058366A1 (en) * | 2006-09-28 | 2008-05-22 | Université de Montréal | Oil-in-water emulsions, methods of use thereof, methods of preparation thereof and kits thereof |
| WO2008042841A2 (en) * | 2006-10-02 | 2008-04-10 | Dr. Reddy's Laboratories Limited | Docetaxel compositions |
| US20080234376A1 (en) * | 2007-03-21 | 2008-09-25 | Taiwan Liposome Company (A Taiwan Corporation) | Emulsion composition comprising prostaglandin e1 |
| CN100569294C (zh) * | 2007-04-13 | 2009-12-16 | 西安力邦制药有限公司 | 一种静脉注射用高稳定长循环脂肪乳载药制剂 |
-
2009
- 2009-07-22 KR KR1020117001821A patent/KR20110036075A/ko not_active Ceased
- 2009-07-22 US US13/055,613 patent/US20110275705A1/en not_active Abandoned
- 2009-07-22 AU AU2009280803A patent/AU2009280803B2/en not_active Ceased
- 2009-07-22 EA EA201100069A patent/EA201100069A1/ru unknown
- 2009-07-22 CA CA2731353A patent/CA2731353A1/en not_active Abandoned
- 2009-07-22 CN CN2009801287862A patent/CN102105134B/zh not_active Expired - Fee Related
- 2009-07-22 EP EP09768245A patent/EP2317978A2/en not_active Withdrawn
- 2009-07-22 MX MX2011000795A patent/MX2011000795A/es unknown
- 2009-07-22 NZ NZ590730A patent/NZ590730A/xx not_active IP Right Cessation
- 2009-07-22 JP JP2011519283A patent/JP5635504B2/ja not_active Expired - Fee Related
- 2009-07-22 BR BRPI0916535A patent/BRPI0916535A2/pt not_active Application Discontinuation
- 2009-07-22 WO PCT/IN2009/000416 patent/WO2010018596A2/en not_active Ceased
-
2011
- 2011-01-18 ZA ZA2011/00465A patent/ZA201100465B/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| EP2317978A2 (en) | 2011-05-11 |
| AU2009280803A1 (en) | 2010-02-18 |
| WO2010018596A2 (en) | 2010-02-18 |
| CN102105134B (zh) | 2013-08-14 |
| WO2010018596A3 (en) | 2010-06-24 |
| NZ590730A (en) | 2012-10-26 |
| MX2011000795A (es) | 2011-03-29 |
| JP2011529042A (ja) | 2011-12-01 |
| US20110275705A1 (en) | 2011-11-10 |
| ZA201100465B (en) | 2012-02-29 |
| EA201100069A1 (ru) | 2011-10-31 |
| BRPI0916535A2 (pt) | 2015-11-10 |
| CN102105134A (zh) | 2011-06-22 |
| JP5635504B2 (ja) | 2014-12-03 |
| CA2731353A1 (en) | 2010-02-18 |
| AU2009280803B2 (en) | 2013-10-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20110036075A (ko) | 안정한 주입 가능 수중유형 도세타셀 나노에멀젼 | |
| US9339553B2 (en) | Liquid compositions of insoluble drugs and preparation methods thereof | |
| US6245349B1 (en) | Drug delivery compositions suitable for intravenous injection | |
| US20190201338A1 (en) | Compositions for Nanoemulsion Delivery Systems | |
| KR101505419B1 (ko) | 나노분산액 | |
| AU2002312777A1 (en) | Method and composition for solubilising a biologically active compound with low water solubility | |
| EP1389089A2 (en) | Method and composition for solubilising a biologically active compound with low water solubility | |
| KR102656823B1 (ko) | 주입액 또는 주사액으로서 및 주입용 농축액의 정맥내 투여를 위한 개선된 레보시멘단 제형 | |
| JP5574965B2 (ja) | 新規タキソイドに基づく組成物 | |
| JP4929158B2 (ja) | 難水溶性薬物を含有する医薬組成物 | |
| JP2005225818A (ja) | パクリタキセル又はドセタキセルの医薬組成物 | |
| WO2012029456A1 (ja) | 難容性薬物含有水中油型乳化組成物及びその製造方法 | |
| US20250099479A1 (en) | Stable antiemetic emulsions | |
| JP2022514991A (ja) | 安定な麻酔薬製剤および関連する剤形 | |
| HK1154502B (en) | Preparation method of drug loaded emulsion | |
| HK1154502A1 (en) | Preparation method of drug loaded emulsion |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20110124 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20130729 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20140924 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20150305 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20140924 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |