KR102101806B1 - 항-인간-her3 항체 및 이의 용도 - Google Patents
항-인간-her3 항체 및 이의 용도 Download PDFInfo
- Publication number
- KR102101806B1 KR102101806B1 KR1020137033910A KR20137033910A KR102101806B1 KR 102101806 B1 KR102101806 B1 KR 102101806B1 KR 1020137033910 A KR1020137033910 A KR 1020137033910A KR 20137033910 A KR20137033910 A KR 20137033910A KR 102101806 B1 KR102101806 B1 KR 102101806B1
- Authority
- KR
- South Korea
- Prior art keywords
- antibody
- her3
- antibodies
- cancer
- cells
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/30—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants from tumour cells
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2863—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against receptors for growth factors, growth regulators
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/32—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against translation products of oncogenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/34—Identification of a linear epitope shorter than 20 amino acid residues or of a conformational epitope defined by amino acid residues
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/77—Internalization into the cell
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Molecular Biology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Oncology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Cell Biology (AREA)
- Engineering & Computer Science (AREA)
- Mycology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Peptides Or Proteins (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Description
도 1a는 선별된 모노클로날 항체의 HER3 vs HER2 및 Fc 결합을 나타낸다.
도 1b는 Ab6 항체에 관한, 본 발명의 정제된 마우스 IgG 항체의 마우스 HER3에 대한 반응성을 나타낸다.
도 2는 HER3 항원에 대한 정제된 mAb의 ELISA 결합 곡선(a) 및 제조(b)를 나타낸다.
도 3은 wt-, EGFR-, HER2-, HER3-, HER2/HER3- 및 EGFR/HER4-형질감염된 NIH 3T3 세포에 대한 정제된 마우스 IgG 4H9-B11, 9B4-D6, 9F7-F11, 11G10-D2, 12H8-B11, 14H1-H8, 15D4-F2 및 16D3-C1의 유동 세포분석 특이적 결합 프로파일(기하 평균)을 나타낸다. Px 항체는 음성 대조군이다. 경쟁자 항체 Ab6 및 U1-59가 표시되었다.
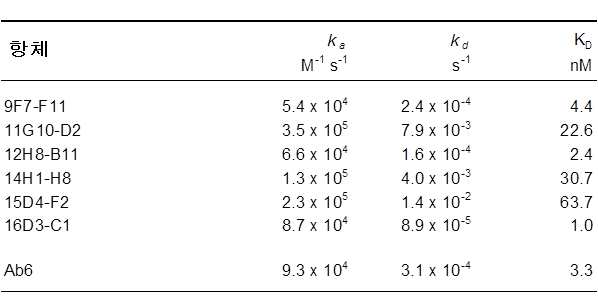
도 4는 mAb 16D3-C1, 9F7-F11 및 12H8-B11의 BIACORE 결합 동적특성을 나타낸다. 표 6은 Ab6 항체에 대한 본 발명의 정제된 마우스 항체 9F7-F11, 11G10-D2, 12H8-B11, 14H1-H8, 15D4-F2 및 16D3-C1의 친화도를 나타낸다.
도 5는 SKBR3 세포 상의 헤리굴린과 HER3-특이적 항체 16D3-C1, 9F7-F11 및 12H8-B11의 FACS 경쟁 실험을 나타낸다. HER3-특이적 양성 대조군 항체 A, B 및 C가 표시되었다.
도 6은 ELISA에 의해 측정된, 항-HER3 뮤린 mAb에 의해 처리된 HER2/HER3-형질감염된 NIH 3T3 세포의 전체 HER2 인산화를 나타낸다.
도 7은 ELISA에 의해 측정된, 항-HER3 뮤린 mAb에 의해 처리된 HER2/HER3-형질감염된 NIH 3T3 세포의 전체 HER3 인산화를 나타낸다.
도 8은 HER2/HER3-형질감염된 NIH 3T3 세포에서 항-HER3 mAb에 의한 HER3/Y1196 HER2(a) 및 Y1262 HER3/Y1112 HER2(b)의 인산화의 억제를 나타낸다. GAPDH는 웨스턴 블롯에서 대조군으로 사용되었다.
도 9는 BxPC3 췌장 암종 세포에서 항-HER3 뮤린 mAb 16D3-C1 및 9F7-F11을 사용함에 따른 HER2/HER3 수용체 및 다운스트림(dowstream) PI3K/Akt 시그날링(signalling)의 인산화의 억제를 나타낸다.
도 10은 BxPC3 췌장 암종에서 HER3 내재화의 항체-유도 억제를 웨스턴 블롯에 의해 나타내고(a), HER3 내재화의 정량을 나타낸다(이미지 J 소프트웨어)(b).
도 11은 MTS 어세이에 의해 측정된, HER2/HER3-형질감염된 NIH 3T3 세포 및 종양 세포주의 뮤린 HER3-특이적 항체에 의한 증식 억제를 나타낸다.
도 12는 TR-FRET 분석에 의해 측정된, HER2/HER3-형질감염된 NIH 3T3 세포에서 항-HER3 mAb에 의한 HER2/HER3 헤테로다이머화의 억제를 나타낸다.
도 13은 16D3-C1 mAb에 의해 인식된 에피토프를 식별한다. (A) 항-HER3 항체 16D3-C1에 의해 인식된 영역의 스폿 분석, (B) 16D3-C1 mAb에 의해 인식된 영역의 알라스칸(Alascan) 분석, 및 (C) HER3 펩티드에 결합하는 16D3-C1의 픽셀 정량(이미지 J 소프트웨어).
도 14는 9F7-F11 mAb에 의해 인식된 에피토프를 식별한다. (A) 항-HER3 항체 9F7-F11에 의해 인식된 영역의 스폿 분석, (B) 9F7-F11 mAb에 의해 인식된 영역의 알라스칸 분석, 및 (C) HER3 펩티드에 결합하는 9F7-F11의 픽셀 정량(이미지 J 소프트웨어).
도 15는 비리간드 HER3 수용체의 결정학적 구조에서 mAb 16D3-C1 및 9F7-F11에 의해 인식된 스폿-기여 잔기(Spot-Contributing Residue)의 포지셔닝(pdb 1M6B)(좌측), 및 페르투주마브에 결합된 HER2 수용체의 결정학적 구조에서 상기 에피토프의 슈퍼포지션(우측)(pdb 1S78)을 나타낸다.
도 16은 HRG-중독된 HER2-비증폭/PIK3CA-wt/p53-mut 편평세포 A431 암 세포가 이종이식된 누드 마우스에서 mAb 16D3-C1 및 9F7-F11에 의한 종양 진행의 억제(A), 및 대응되는 카플란-마이어(Kaplan-Meier) 생존 곡선(B)을 나타낸다.
도 17은 HER2-비증폭/PIK3CA-wt/p53-wt 췌장 BxPC3 암 세포가 이종이식된 누드 마우스에서 mAb 9F7-F11 및 16D3-C1에 의한 종양 진행의 억제(A), 및 대응되는 카플란-마이어 생존 곡선(B)을 나타낸다.
도 18은 운반체- 또는 16D3-C1-처리된 마우스로부터의 추출된 BxPC3 이종이식체에서 전체 HER3 발현 및 Y1289-HER3의 인산화 수준을 나타낸다.
도 19는 HRG-중독된 HER2low 편평세포 A431 (A) 및 폐 A549 (B) 암 세포에서, 단독으로 또는 트라스투주마브와 조합하여 사용된 HER3-특이적 mAb 16D3-C1에 의한 종양 진행의 억제를 나타낸다.
Claims (15)
- HER-3의 세포외 도메인에 특이적으로 결합하고, 인간 HER-3의 세포외 도메인과의 결합에 대해 CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체와 경쟁하는 분리된 모노클로날 항체로,
상기 분리된 모노클로날 항체가 CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체의 가변 경쇄(VL: variable light chain)의 CDR을 포함하는 VL, 및 CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체의 가변 중쇄(VH: variable heavy chain)의 CDR을 포함하는 VH를 포함하는 것을 특징으로 하는, 분리된 모노클로날 항체. - 제1항에 있어서,
상기 항체는 뮤린 항체, 키메라 항체, 인간화 항체 및 인간 항체로 이루어진 군에서 선택되는 것을 특징으로 하는, 분리된 모노클로날 항체. - 제1항에 있어서,
CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체의 VL 및 CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체의 VH를 포함하는 것을 특징으로 하는, 분리된 모노클로날 항체. - 제3항에 있어서,
CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체의 가변 도메인을 포함하는 모노클로날 키메라 항체인 것을 특징으로 하는, 분리된 모노클로날 항체. - 제1항에 있어서,
CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 항체의 CDR을 포함하는 모노클로날 인간화 항체인 것을 특징으로 하는, 분리된 모노클로날 항체. - 제1항에 있어서,
CNCM-I-4486으로 기탁된 하이브리도마로부터 획득할 수 있는 뮤린 모노클로날 항체(16D3-C1)인 것을 특징으로 하는, 분리된 모노클로날 항체. - Fv, Fab, F(ab')2, Fab', dsFv, scFv, sc(Fv)2 및 디아바디로 이루어진 군에서 선택되는, 제1항에 따른 분리된 모노클로날 항체의 항체 단편.
- 제1항에 따른 분리된 모노클로날 항체의 VH 도메인 또는 제1항에 따른 분리된 모노클로날 항체의 VL 도메인을 인코딩하는 핵산.
- 제8항에 따른 핵산을 포함하는 벡터.
- 제8항에 따른 핵산에 의해 형질감염, 감염 또는 형질전환된 숙주 세포.
- 제9항에 따른 벡터에 의해 형질감염, 감염 또는 형질전환된 숙주 세포.
- 제1항에 따른 분리된 모노클로날 항체를 포함하는 암 치료용 약학조성물.
- 제7항에 따른 항체 단편을 포함하는 암 치료용 약학조성물.
- 제1항에 있어서,
암 치료에 사용되는 것을 특징으로 하는, 분리된 모노클로날 항체. - 제7항에 있어서,
암 치료에 사용되는 것을 특징으로 하는, 항체 단편.
Applications Claiming Priority (8)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP11305607.1 | 2011-05-19 | ||
| EP11305607 | 2011-05-19 | ||
| US201161499948P | 2011-06-22 | 2011-06-22 | |
| US61/499,948 | 2011-06-22 | ||
| US201161502932P | 2011-06-30 | 2011-06-30 | |
| US61/507,932 | 2011-06-30 | ||
| US61/502,932 | 2011-06-30 | ||
| PCT/EP2012/059402 WO2012156532A1 (en) | 2011-05-19 | 2012-05-21 | Anti-human-her3 antibodies and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20140041572A KR20140041572A (ko) | 2014-04-04 |
| KR102101806B1 true KR102101806B1 (ko) | 2020-04-20 |
Family
ID=47176333
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020137033910A Expired - Fee Related KR102101806B1 (ko) | 2011-05-19 | 2012-05-21 | 항-인간-her3 항체 및 이의 용도 |
Country Status (7)
| Country | Link |
|---|---|
| US (1) | US9127065B2 (ko) |
| EP (1) | EP2710040B1 (ko) |
| JP (1) | JP2014516960A (ko) |
| KR (1) | KR102101806B1 (ko) |
| CN (1) | CN103890010B (ko) |
| ES (1) | ES2643694T3 (ko) |
| WO (1) | WO2012156532A1 (ko) |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2511761A (en) * | 2013-03-11 | 2014-09-17 | Cancer Rec Tech Ltd | Methods for detecting molecules in a sample |
| EP3052657A2 (en) * | 2013-09-30 | 2016-08-10 | Daiichi Sankyo Company, Limited | Nucleic acid biomarker and use thereof |
| ES2865196T3 (es) | 2013-11-07 | 2021-10-15 | Inst Nat Sante Rech Med | Anticuerpos anti-HER3 humana alostéricos de neuregulina |
| AU2017201075B2 (en) * | 2014-07-17 | 2019-03-07 | The Trustees Of The University Of Pennsylvania | Identification of immunogenic MHC Class II peptides for immune-based therapy |
| EP3169354A4 (en) * | 2014-07-17 | 2018-06-13 | Brian J. Czerniecki | Identification of immunogenic mhc class ii peptides for immune-based therapy |
| WO2017014810A1 (en) * | 2015-07-17 | 2017-01-26 | Czerniecki Brian J | Identification of immunogenic mhc class ii peptides for immune-based therapy |
| CA2959716A1 (en) | 2014-09-08 | 2016-03-17 | Yeda Research And Development Co. Ltd. | Anti-her3 antibodies and uses of same |
| AU2015313811A1 (en) | 2014-09-08 | 2017-04-06 | Yeda Research And Development Co. Ltd. | Compositions and methods for treating cancer resistant to a tyrosine kinase inhibitor (TKI) |
| EP3091033A1 (en) * | 2015-05-06 | 2016-11-09 | Gamamabs Pharma | Anti-human-her3 antibodies and uses thereof |
| EA201792560A1 (ru) * | 2015-05-28 | 2018-06-29 | Нанобиотикс | Наночастицы для применения в качестве терапевтической вакцины |
| WO2017072150A1 (en) * | 2015-10-27 | 2017-05-04 | INSERM (Institut National de la Santé et de la Recherche Médicale) | Anti-nrg1 (heregulin) antibodies and uses thereof |
| CN108602890A (zh) | 2015-12-11 | 2018-09-28 | 瑞泽恩制药公司 | 用于减少或预防对egfr和/或erbb3阻滞剂具有抗性的肿瘤生长的方法 |
| US10093731B2 (en) | 2017-02-24 | 2018-10-09 | Kindred Biosciences, Inc. | Anti-IL31 antibodies for veterinary use |
| CA3072267A1 (en) * | 2017-08-09 | 2019-02-14 | University Of Saskatchewan | Her3 binding agents and uses thereof |
| WO2019185164A1 (en) | 2018-03-29 | 2019-10-03 | Hummingbird Bioscience Holdings Pte. Ltd. | Her3 antigen-binding molecules |
| WO2019241893A2 (en) * | 2018-06-22 | 2019-12-26 | Crd Pharmaceuticals Inc | Anti-her3 antibody and uses thereof |
| CN110724194B (zh) | 2018-07-17 | 2021-03-19 | 上海生物制品研究所有限责任公司 | 抗her3人源化单克隆抗体及其制剂 |
| JP6655673B2 (ja) * | 2018-07-20 | 2020-02-26 | アンスティチュ ナショナル ドゥ ラ サンテ エ ドゥ ラ ルシェルシュ メディカル | ニューレグリンに対して非競合的でアロステリックな抗ヒトher3抗体及びその使用 |
| CN120239709A (zh) * | 2022-10-28 | 2025-07-01 | 翰森生物有限责任公司 | 抗体、其抗原结合片段及其药物用途 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007077028A2 (en) * | 2005-12-30 | 2007-07-12 | U3 Pharma Ag | Antibodies directed to her-3 and uses thereof |
| US20090291085A1 (en) | 2007-02-16 | 2009-11-26 | Merrimack Pharmaceuticals, Inc. | Antibodies against erbb3 and uses thereof |
Family Cites Families (61)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4179337A (en) | 1973-07-20 | 1979-12-18 | Davis Frank F | Non-immunogenic polypeptides |
| JPS6023084B2 (ja) | 1979-07-11 | 1985-06-05 | 味の素株式会社 | 代用血液 |
| US4640835A (en) | 1981-10-30 | 1987-02-03 | Nippon Chemiphar Company, Ltd. | Plasminogen activator derivatives |
| US4486414A (en) | 1983-03-21 | 1984-12-04 | Arizona Board Of Reagents | Dolastatins A and B cell growth inhibitory substances |
| US4816567A (en) | 1983-04-08 | 1989-03-28 | Genentech, Inc. | Recombinant immunoglobin preparations |
| US4496689A (en) | 1983-12-27 | 1985-01-29 | Miles Laboratories, Inc. | Covalently attached complex of alpha-1-proteinase inhibitor with a water soluble polymer |
| EP0173494A3 (en) | 1984-08-27 | 1987-11-25 | The Board Of Trustees Of The Leland Stanford Junior University | Chimeric receptors by dna splicing and expression |
| DE3675588D1 (de) | 1985-06-19 | 1990-12-20 | Ajinomoto Kk | Haemoglobin, das an ein poly(alkenylenoxid) gebunden ist. |
| WO1987002671A1 (en) | 1985-11-01 | 1987-05-07 | International Genetic Engineering, Inc. | Modular assembly of antibody genes, antibodies prepared thereby and use |
| WO1987005330A1 (en) | 1986-03-07 | 1987-09-11 | Michel Louis Eugene Bergh | Method for enhancing glycoprotein stability |
| GB8607679D0 (en) | 1986-03-27 | 1986-04-30 | Winter G P | Recombinant dna product |
| US5225539A (en) | 1986-03-27 | 1993-07-06 | Medical Research Council | Recombinant altered antibodies and methods of making altered antibodies |
| US4861719A (en) | 1986-04-25 | 1989-08-29 | Fred Hutchinson Cancer Research Center | DNA constructs for retrovirus packaging cell lines |
| US4791192A (en) | 1986-06-26 | 1988-12-13 | Takeda Chemical Industries, Ltd. | Chemically modified protein with polyethyleneglycol |
| US4816444A (en) | 1987-07-10 | 1989-03-28 | Arizona Board Of Regents, Arizona State University | Cell growth inhibitory substance |
| US5202238A (en) | 1987-10-27 | 1993-04-13 | Oncogen | Production of chimeric antibodies by homologous recombination |
| US5204244A (en) | 1987-10-27 | 1993-04-20 | Oncogen | Production of chimeric antibodies by homologous recombination |
| US5278056A (en) | 1988-02-05 | 1994-01-11 | The Trustees Of Columbia University In The City Of New York | Retroviral packaging cell lines and process of using same |
| DE68921982T4 (de) | 1988-06-14 | 1996-04-25 | Cetus Oncology Corp | Kupplungsmittel und sterisch gehinderte, mit disulfid gebundene konjugate daraus. |
| US5076973A (en) | 1988-10-24 | 1991-12-31 | Arizona Board Of Regents | Synthesis of dolastatin 3 |
| US5530101A (en) | 1988-12-28 | 1996-06-25 | Protein Design Labs, Inc. | Humanized immunoglobulins |
| US4978744A (en) | 1989-01-27 | 1990-12-18 | Arizona Board Of Regents | Synthesis of dolastatin 10 |
| US4879278A (en) | 1989-05-16 | 1989-11-07 | Arizona Board Of Regents | Isolation and structural elucidation of the cytostatic linear depsipeptide dolastatin 15 |
| US4986988A (en) | 1989-05-18 | 1991-01-22 | Arizona Board Of Regents | Isolation and structural elucidation of the cytostatic linear depsipeptides dolastatin 13 and dehydrodolastatin 13 |
| US5138036A (en) | 1989-11-13 | 1992-08-11 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Isolation and structural elucidation of the cytostatic cyclodepsipeptide dolastatin 14 |
| GB8928874D0 (en) | 1989-12-21 | 1990-02-28 | Celltech Ltd | Humanised antibodies |
| US5859205A (en) | 1989-12-21 | 1999-01-12 | Celltech Limited | Humanised antibodies |
| US5670488A (en) | 1992-12-03 | 1997-09-23 | Genzyme Corporation | Adenovirus vector for gene therapy |
| EP0519596B1 (en) | 1991-05-17 | 2005-02-23 | Merck & Co. Inc. | A method for reducing the immunogenicity of antibody variable domains |
| ES2136092T3 (es) | 1991-09-23 | 1999-11-16 | Medical Res Council | Procedimientos para la produccion de anticuerpos humanizados. |
| US5639641A (en) | 1992-09-09 | 1997-06-17 | Immunogen Inc. | Resurfacing of rodent antibodies |
| US6034065A (en) | 1992-12-03 | 2000-03-07 | Arizona Board Of Regents | Elucidation and synthesis of antineoplastic tetrapeptide phenethylamides of dolastatin 10 |
| US5635483A (en) | 1992-12-03 | 1997-06-03 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Tumor inhibiting tetrapeptide bearing modified phenethyl amides |
| US5410024A (en) | 1993-01-21 | 1995-04-25 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide amides |
| US5780588A (en) | 1993-01-26 | 1998-07-14 | Arizona Board Of Regents | Elucidation and synthesis of selected pentapeptides |
| CA2156725A1 (en) | 1993-02-22 | 1994-09-01 | Warren S. Pear | Production of high titer helper-free retroviruses by transient transfection |
| FR2712812B1 (fr) | 1993-11-23 | 1996-02-09 | Centre Nat Rech Scient | Composition pour la production de produits thérapeutiques in vivo. |
| KR100261941B1 (ko) | 1994-07-13 | 2000-07-15 | 나가야마 오사무 | 사람의 인터루킨-8에 대한 재구성 사람항체 |
| US5530097A (en) | 1994-08-01 | 1996-06-25 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory peptide amides |
| US5521284A (en) | 1994-08-01 | 1996-05-28 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide amides and esters |
| US5504191A (en) | 1994-08-01 | 1996-04-02 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide methyl esters |
| US5554725A (en) | 1994-09-14 | 1996-09-10 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Synthesis of dolastatin 15 |
| US5599902A (en) | 1994-11-10 | 1997-02-04 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Cancer inhibitory peptides |
| US5663149A (en) | 1994-12-13 | 1997-09-02 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Human cancer inhibitory pentapeptide heterocyclic and halophenyl amides |
| IL116816A (en) | 1995-01-20 | 2003-05-29 | Rhone Poulenc Rorer Sa | Cell for the production of a defective recombinant adenovirus or an adeno-associated virus and the various uses thereof |
| WO1997010354A1 (fr) | 1995-09-11 | 1997-03-20 | Kyowa Hakko Kogyo Co., Ltd. | Anticorps de la chaine alpha du recepteur de l'interleukine 5 humaine |
| US6013516A (en) | 1995-10-06 | 2000-01-11 | The Salk Institute For Biological Studies | Vector and method of use for nucleic acid delivery to non-dividing cells |
| US6130237A (en) | 1996-09-12 | 2000-10-10 | Cancer Research Campaign Technology Limited | Condensed N-aclyindoles as antitumor agents |
| US6239104B1 (en) | 1997-02-25 | 2001-05-29 | Arizona Board Of Regents | Isolation and structural elucidation of the cytostatic linear and cyclo-depsipeptides dolastatin 16, dolastatin 17, and dolastatin 18 |
| CA2287084A1 (en) | 1997-04-10 | 1998-10-15 | Royal Netherlands Academy Of Arts And Sciences | Diagnosis method and reagents |
| US6323315B1 (en) | 1999-09-10 | 2001-11-27 | Basf Aktiengesellschaft | Dolastatin peptides |
| US6884869B2 (en) | 2001-04-30 | 2005-04-26 | Seattle Genetics, Inc. | Pentapeptide compounds and uses related thereto |
| US20030083263A1 (en) | 2001-04-30 | 2003-05-01 | Svetlana Doronina | Pentapeptide compounds and uses related thereto |
| EP1283053A1 (en) * | 2001-08-09 | 2003-02-12 | Max-Planck-Gesellschaft zur Förderung der Wissenschaften e.V. | Inhibitors of HER3 activity |
| EP2353611B1 (en) | 2002-07-31 | 2015-05-13 | Seattle Genetics, Inc. | Drug conjugates and their use for treating cancer, an autoimmune disease or an infectious disease |
| BRPI0510716A (pt) | 2004-05-05 | 2007-11-20 | Merrimack Pharmaceuticals Inc | uso de um agente de ligação bi-especìfico, agente de ligação bi-especìfico, composição de um agente de ligação bi-especìfico, e, kit |
| EP1850874B1 (en) * | 2005-02-23 | 2013-10-16 | Genentech, Inc. | Extending time to disease progression or survival in ovarian cancer patients using pertuzumab |
| JP2008531557A (ja) | 2005-02-23 | 2008-08-14 | メリマック ファーマシューティカルズ インコーポレーティッド | 生物活性を調節するための二重特異性結合剤 |
| AU2008100624B4 (en) * | 2008-07-07 | 2009-08-06 | Bailey Enterprises Australia | Multifunction. Decorative Water Tank II |
| ES2572728T3 (es) | 2009-03-20 | 2016-06-02 | F. Hoffmann-La Roche Ag | Anticuerpos anti-HER biespecíficos |
| AU2010242914B2 (en) * | 2009-04-29 | 2014-11-13 | Trellis Bioscience, Llc | Improved antibodies immunoreactive with heregulin-coupled HER3 |
-
2012
- 2012-05-21 KR KR1020137033910A patent/KR102101806B1/ko not_active Expired - Fee Related
- 2012-05-21 CN CN201280023353.2A patent/CN103890010B/zh active Active
- 2012-05-21 JP JP2014510836A patent/JP2014516960A/ja active Pending
- 2012-05-21 US US14/118,747 patent/US9127065B2/en active Active
- 2012-05-21 ES ES12723857.4T patent/ES2643694T3/es active Active
- 2012-05-21 EP EP12723857.4A patent/EP2710040B1/en active Active
- 2012-05-21 WO PCT/EP2012/059402 patent/WO2012156532A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2007077028A2 (en) * | 2005-12-30 | 2007-07-12 | U3 Pharma Ag | Antibodies directed to her-3 and uses thereof |
| US20090291085A1 (en) | 2007-02-16 | 2009-11-26 | Merrimack Pharmaceuticals, Inc. | Antibodies against erbb3 and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| US20140112931A1 (en) | 2014-04-24 |
| JP2014516960A (ja) | 2014-07-17 |
| WO2012156532A1 (en) | 2012-11-22 |
| ES2643694T3 (es) | 2017-11-23 |
| US9127065B2 (en) | 2015-09-08 |
| EP2710040B1 (en) | 2017-07-12 |
| EP2710040A1 (en) | 2014-03-26 |
| KR20140041572A (ko) | 2014-04-04 |
| CN103890010B (zh) | 2017-04-19 |
| CN103890010A (zh) | 2014-06-25 |
| WO2012156532A8 (en) | 2014-07-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR102101806B1 (ko) | 항-인간-her3 항체 및 이의 용도 | |
| US9409988B2 (en) | Anti-Axl antibodies and uses thereof | |
| EP2723376B1 (en) | Anti-axl antibodies and uses thereof | |
| US11001634B2 (en) | Anti-human-HER3 antibodies and uses thereof | |
| JP6449876B2 (ja) | ニューレグリンに対して非競合的でアロステリックな抗ヒトher3抗体及びその使用 | |
| JP6655673B2 (ja) | ニューレグリンに対して非競合的でアロステリックな抗ヒトher3抗体及びその使用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P18-X000 | Priority claim added or amended |
St.27 status event code: A-2-2-P10-P18-nap-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20250411 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| R18 | Changes to party contact information recorded |
Free format text: ST27 STATUS EVENT CODE: A-5-5-R10-R18-OTH-X000 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20250411 |