KR101281501B1 - 항인테그린 면역 컨쥬게이트, 방법 및 용도 - Google Patents
항인테그린 면역 컨쥬게이트, 방법 및 용도 Download PDFInfo
- Publication number
- KR101281501B1 KR101281501B1 KR1020077015643A KR20077015643A KR101281501B1 KR 101281501 B1 KR101281501 B1 KR 101281501B1 KR 1020077015643 A KR1020077015643 A KR 1020077015643A KR 20077015643 A KR20077015643 A KR 20077015643A KR 101281501 B1 KR101281501 B1 KR 101281501B1
- Authority
- KR
- South Korea
- Prior art keywords
- antibody
- cnto
- drug conjugate
- cancer
- human
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2839—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the integrin superfamily
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6801—Drug-antibody or immunoglobulin conjugates defined by the pharmacologically or therapeutically active agent
- A61K47/6803—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates
- A61K47/68033—Drugs conjugated to an antibody or immunoglobulin, e.g. cisplatin-antibody conjugates the drug being a maytansine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/68—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment
- A61K47/6835—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site
- A61K47/6849—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an antibody, an immunoglobulin or a fragment thereof, e.g. an Fc-fragment the modifying agent being an antibody or an immunoglobulin bearing at least one antigen-binding site the antibody targeting a receptor, a cell surface antigen or a cell surface determinant
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/08—Antiseborrheics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/10—Anti-acne agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/46—Hybrid immunoglobulins
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/20—Immunoglobulins specific features characterized by taxonomic origin
- C07K2317/21—Immunoglobulins specific features characterized by taxonomic origin from primates, e.g. man
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Immunology (AREA)
- Epidemiology (AREA)
- Dermatology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Ophthalmology & Optometry (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Rheumatology (AREA)
- Cardiology (AREA)
- Cell Biology (AREA)
- Heart & Thoracic Surgery (AREA)
- Oncology (AREA)
- Mycology (AREA)
- Microbiology (AREA)
- Urology & Nephrology (AREA)
- Pain & Pain Management (AREA)
- Physical Education & Sports Medicine (AREA)
- Hematology (AREA)
- Vascular Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
Description
| 화합물 | 약물 | 메이탄시놀 활성화제 |
Mab 활성화제 | 생성물 중의 디술파이에 인접하는 메틸기 (Mab측:약물측) |
| CNTO 364 | DM1 | N-메틸, N-(1-디티오메틸-3-카복시-프로필)알라닌 | SPP 또는 SSNPP | 1:0 |
| CNTO 365 | DM4 | N-메틸, N-(1-디티오메틸-2-메틸-4-카복시-n-부틸)알라닌 | SPDB 또는 SSNPB | 0:2 |
| CNTO 366 | DM4 | N-메틸, N-(1-디티오메틸-2-메틸-4-카복시-n-부틸)알라닌 | SPP 또는 SSNPP | 1:2 |
| F105-DM1 | DM1 | N-메틸, N-(1-디티오메틸-3-카복시-프로필)알라닌 | SPP 또는 SSNPP | 1:0 |
| F105-DM4 | DM4 | N-메틸, N-(1-디티오메틸-2-메틸-4-카복시-n-부틸)알라닌 | SPDB 또는 SSNPB | 0:2 |
| F105-DM4 | DM4 | N-메틸, N-(1-디티오메틸-2-메틸-4-카복시-n-부틸)알라닌 | SPP 또는 SSNPP | 1:2 |
| 화합물 | HT29 | A549 | A2780 |
| CNTO 95 | 0.14 | 0.18 | 0.17 |
| CNTO 364 | 0.19 | 0.27 | 0.27 |
| CNTO 365 | 0.21 | 0.34 | 0.27 |
| CNTO 366 | 0.29 | 0.42 | 0.30 |
| EC50 (㎍/ml) | ||
| 면역 컨쥬게이트 | HT29 | A549 |
| CNTO 364 | 1.0 | 1.2 |
| CNTO 365 | 0.24 | 0.3 |
| CNTO 366 | 1.0 | 1.5 |
| 그룹 | N | CNTO 364 (mg/kg) |
투여일 |
| 1) PBS | 9 | 0 | 7, 14, 21, 28 및 35 |
| 2) F105-DM1 | 9 | 25 | 7 및 14 |
| 3) CNTO 364 | 9 | 3 | 7, 14, 21, 28 및 35 |
| 4) CNTO 364 | 9 | 6 | 7, 14, 21, 28 및 35 |
| 5) CNTO 364 | 9 | 10 | 7, 14, 21, 28 및 35 |
| 6) CNTO 364 | 9 | 15 | 7, 14 및 35 |
| 7) CNTO 364 | 9 | 25 | 7 및 14 |
| 그룹 | 약물 | 17일째 및 29일째의 투약량 |
| 1 | PBS | 사용되지 않음 |
| 2 | F105-DM1 | 15 mg/kg |
| 3 | CNTO 364 | 15 mg/kg |
| 4 | CNTO 95 단독 | 15 mg/kg |
| 5 | CNTO 95 + 메이탄신 | 15 mg/kg + 260 ㎍/kg |
| 6 | 메이탄신 단독 | 260 ㎍/kg |
| 그룹 번호 | 투여된 Ab 컨쥬게이트 (mg/kg) |
DMx의 용량 ㎍/kg |
| 1. PBS | 0 | 0 |
| 2. F105-SPP-DM1 | 11.5 | DM1: 175 |
| 3. F105-SPP-DM1 | 23 | DM1: 350 |
| 4. F105-SSNPB-DM4 | 10 | DM4: 175 |
| 5. F105-SSNPB-DM4 | 20 | DM4: 350 |
| 6. F105-SSNPP-DM4 | 10.5 | DM4: 175 |
| 7. F105-SSNPP-DM4 | 21 | DM4: 350 |
| 8. CNTO 364 | 10 | DM1: 175 |
| 9. CNTO 364 | 20 | DM1: 350 |
| 10. CNTO 365 | 10 | DM4: 175 |
| 11. CNTO 365 | 20 | DM4: 350 |
| 12. CNTO 366 | 12 | DM4: 175 |
| 13. CNTO 366 | 24 | DM4: 350 |
Claims (36)
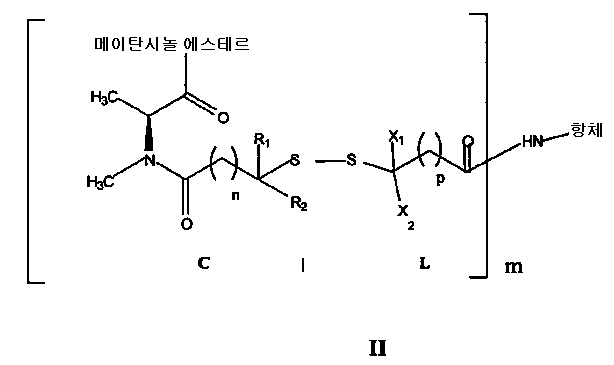
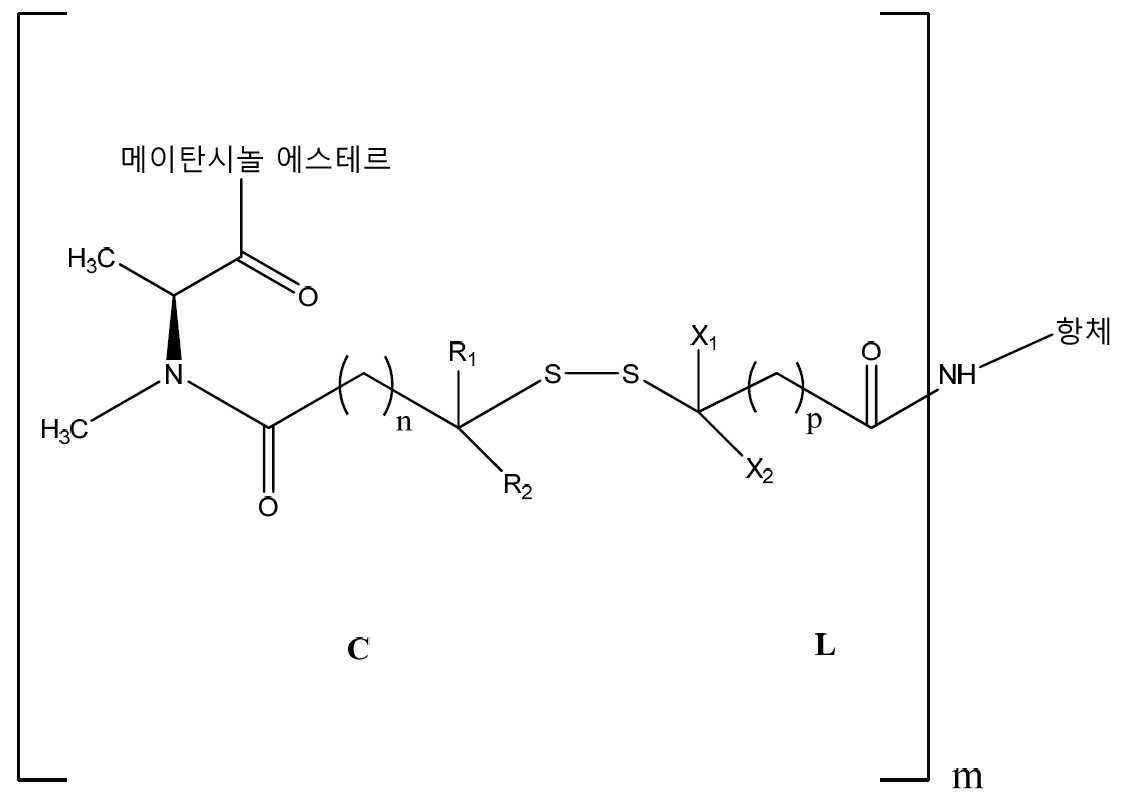
- 하기 화학식의 항체-약물 컨쥬게이트, 및 이의 약제학적으로 허용가능한 염 및 에스테르:상기 식에서,항체는 인간 알파V 인테그린 서브유닛 특이적인 항체로서, 상기 알파V 인테그린 서브유닛을 발현하는 세포에 의해 내재화될 수 있으며, (i) 서열번호 1, 2 및 3에 기재된 CNTO 95 항체의 모든 중쇄 상보성 결정 영역(CDR) 아미노산 서열, 및 (ii) 서열번호 4, 5 및 6에 기재된 CNTO 95 항체의 모든 경쇄 CDR 아미노산 서열을 포함하고;메이탄시놀은 C-3에서 에스테르화되고;R1 및 R2는 각각 Me이고;X1 및 X2는 각각 H이며;n은 2이고;p는 1 내지 5이며;m은 1 내지 10이다.
- 제1항에 있어서, 항체가 인간 알파V 인테그린으로의 결합에 대하여 모노클로널 항체 CNTO 95 또는 이의 단편과 경쟁하는 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제1항에 있어서, 항체가 모노클로널 항체 CNTO 95인 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제2항에 있어서, 항체가 인간 알파V베타3 인테그린을 발현하는 생세포로의 결합에 대하여 CNTO 95와 경쟁하는 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제1항에 있어서, 항체가 서열번호 9의 아미노산 서열 내의 에피토프에 특이적인 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제1항에 있어서, 항체가 인간, 인간화 항체 또는 키메라 항체인 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제1항에 있어서, m이 3 또는 4인 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제1항에 있어서, X1 및 X2가 각각 H이고, p가 2인 것을 특징으로 하는 항체-약물 컨쥬게이트.
- 제8항에 있어서, 항체가 모노클로널 항체 CNTO 95인 것을 특징으로 하는 항체-약물 컨쥬게이트.
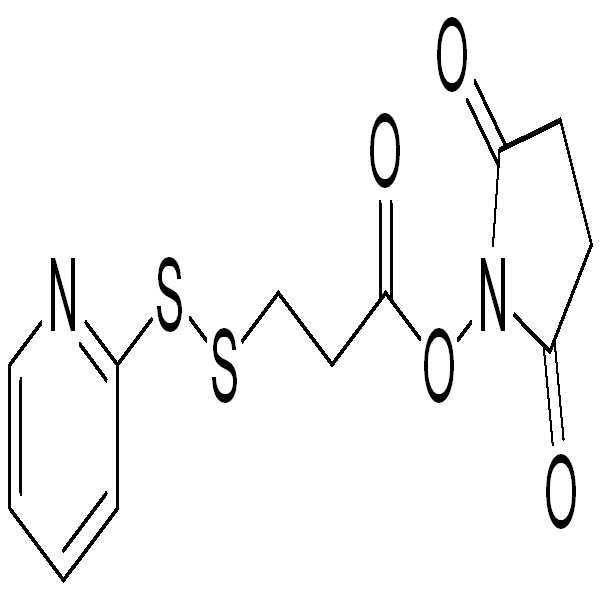
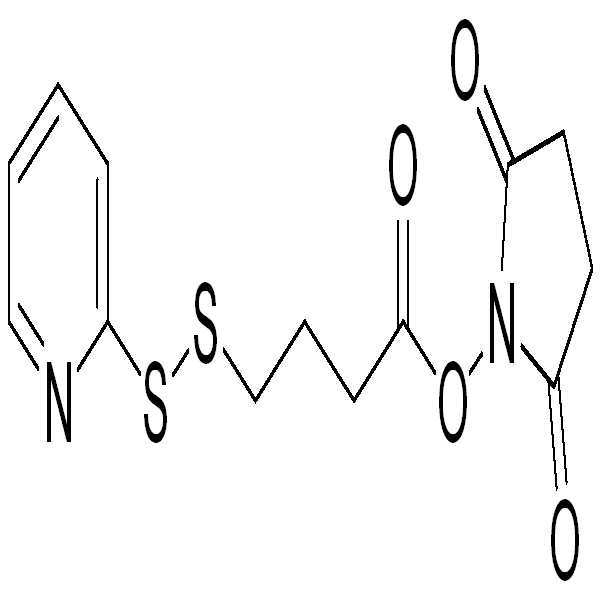
- 제10항에 있어서, 이작용성 커플링 시약이 (2-피리딜)-3-디티오프로판산 N-하이드록시 숙신이미드 에스테르(SPDP), 또는 (2-피리딜)-4-디티오부탄산 N-하이드록시 숙신이미드 에스테르(SPDB)인 것을 특징으로 하는 방법.
- 제1항 내지 제9항 중 어느 한 항의 항체-약물 컨쥬게이트, 및 약제학적으로 허용가능한 담체, 희석제 또는 부형제를 포함하는, 암 치료용 약제학적 조성물.
- 암 치료용 약제학적 조성물의 제조를 위해 제1항 내지 제9항 중 어느 한 항의 항체-약물 컨쥬게이트를 사용하는 방법.
- 제1항의 항체-약물 컨쥬게이트의 치료적 유효량을 포함하는, 유방 선암, 폐 선암, 췌장 선암, 결장 선암, 신장 세포암, 또는 위 선암 중에서 선택된 암 치료용 약제.
- 제14항에 있어서, 암이 두경부 편평상피암, 식도 편평상피암, 폐 편평상피암, 피부 편평상피암, 또는 자궁경부 편평상피암인 것을 특징으로 하는 약제.
- 제1항의 항체-약물 컨쥬게이트의 치료적 유효량을 포함하는, 암의 전이 확산 치료 또는 예방용 약제.
- 인간 알파V베타3 인테그린을 발현하는 생세포에 대한 CNTO 95의 결합을 방지하는 제1항의 항체-약물 컨쥬게이트를 암 세포 증식을 억제하는데 유효한 양으로 포함하는, 암 세포 증식 억제용 약제.
- 제17항에 있어서, 약제가 정맥내로 투여되는 것을 특징으로 하는 약제.
- 제18항에 있어서, 항체-약물 컨쥬게이트가 0.05 mg/체중 kg 내지 12.0 mg/체중 kg의 양으로 투여되는 것을 특징으로 하는 약제.
- 인간 알파V베타3 인테그린을 발현하는 생세포에 대한 CNTO 95의 결합을 방지하는 제1항의 항체-약물 컨쥬게이트를 혈관신생을 억제하는데 유효한 양으로 포함하는,암 전이, 혈관종, 혈관섬유종, 당뇨병성 망막증, 미숙아 망막증, 혈관 신생 녹내장, 혈관 형성에 의해 유발되는 각막 질환, 퇴행기 황반, 황반 변성, 익상편, 망막 변성, 수정체후 섬유증식증, 과립성 결막염, 건선, 모세혈관 확장증, 화농성 육아종, 지루성 피부염, 좌창 및 관절염으로 구성되는 그룹 중에서 선택되는 혈관 신생 의존성 질환에 걸린 포유동물의 혈관신생 억제용 약제.
- 인간 알파V베타3 인테그린을 발현하는 생세포에 대한 CNTO 95의 결합을 방지하는 제1항의 항체-약물 컨쥬게이트를 염증성 질환을 개선하는데 유효한 양으로 포함하는, 류머티스 관절염, 황반 변성, 건선 및 당뇨병성 망막증으로 구성되는 그룹 중에서 선택되는 염증성 질환 개선용 약제.
- 제20항에 있어서, 항체-약물 컨쥬게이트가 건선, 정맥성 궤양, 좌창, 주사, 사마귀, 습진, 혈관종, 및 림프관 형성으로 구성되는 그룹 중에서 선택되는 혈관신생 피부 질환을 치료하는 것을 특징으로 하는 약제.
- 제20항에 있어서, 항체-약물 컨쥬게이트가 각막 또는 망막 신생혈관을 수반하는 질환을 치료하는 것을 특징으로 하는 약제.
- 제14항 내지 제23항 중 어느 한 항에 있어서, 항체-약물 컨쥬게이트가 제2의 치료제 또는 예방제 또는 모댈러티(modality)와 병용하여 투여되는 것을 특징으로 하는 약제.
- 제1항 내지 제9항 중 어느 한 항의 항체-약물 컨쥬게이트 및 용기(container)를 포함하고, 추가로 상기 항체-약물 컨쥬게이트가 알파V 인테그린 서브유닛을 발현하는 세포를 특징으로 하는 질환을 치료하는데 사용될 수 있음을 표시하는 패키지 인서트 또는 라벨을 포함하는 제품.
- 제25항에 있어서, 패키지 인서트가, 상기 항체-약물 컨쥬게이트가 암을 치료하는데 사용될 수 있음을 표시하는 것을 특징으로 하는 제품.
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US63444504P | 2004-12-09 | 2004-12-09 | |
| US60/634,445 | 2004-12-09 | ||
| PCT/US2005/043250 WO2006062779A2 (en) | 2004-12-09 | 2005-11-30 | Anti-integrin immunoconjugates, methods and uses |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20070089220A KR20070089220A (ko) | 2007-08-30 |
| KR101281501B1 true KR101281501B1 (ko) | 2013-07-15 |
Family
ID=36578407
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020077015643A Expired - Fee Related KR101281501B1 (ko) | 2004-12-09 | 2005-11-30 | 항인테그린 면역 컨쥬게이트, 방법 및 용도 |
Country Status (24)
| Country | Link |
|---|---|
| US (2) | US8603483B2 (ko) |
| EP (1) | EP1819359B1 (ko) |
| JP (1) | JP5421532B2 (ko) |
| KR (1) | KR101281501B1 (ko) |
| CN (1) | CN101193916B (ko) |
| AR (1) | AR052041A1 (ko) |
| AU (1) | AU2005314392B2 (ko) |
| BR (1) | BRPI0515745A (ko) |
| CA (1) | CA2591148A1 (ko) |
| DK (1) | DK1819359T3 (ko) |
| EA (1) | EA013323B1 (ko) |
| ES (1) | ES2539126T3 (ko) |
| HU (1) | HUE025449T2 (ko) |
| IL (1) | IL183695A0 (ko) |
| MX (1) | MX2007007011A (ko) |
| NO (1) | NO20073493L (ko) |
| NZ (1) | NZ555601A (ko) |
| PL (1) | PL1819359T3 (ko) |
| PT (1) | PT1819359E (ko) |
| SG (1) | SG158119A1 (ko) |
| TW (1) | TWI400087B (ko) |
| UA (1) | UA94388C2 (ko) |
| WO (1) | WO2006062779A2 (ko) |
| ZA (1) | ZA200705556B (ko) |
Families Citing this family (67)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2325344B1 (es) * | 2004-11-02 | 2010-06-09 | Univ Madrid Autonoma | Inhibidores de angiogenesis multifuncionales y multivalentes. |
| EA013323B1 (ru) * | 2004-12-09 | 2010-04-30 | Сентокор, Инк. | Иммуноконъюгаты против интегрина, способы и варианты применения |
| CA2617637C (en) | 2005-08-02 | 2017-07-18 | Xbiotech Inc. | Diagnosis, treatment, and prevention of vascular disorders using il-1.alpha. autoantibodies |
| CA2646611A1 (en) | 2006-03-21 | 2008-05-22 | Genentech, Inc. | Combinatorial therapy |
| CN103524619B (zh) * | 2006-08-03 | 2016-10-05 | 阿斯利康(瑞典)有限公司 | 针对αVβ6的抗体及其应用 |
| WO2008042611A2 (en) * | 2006-09-29 | 2008-04-10 | Centocor, Inc. | Method of using il6 antagonists with mitoxantrone for prostate cancer |
| US20090175784A1 (en) * | 2007-05-11 | 2009-07-09 | Joshua Goldstein | Anti-Alpha V Immunoliposome Composition, Methods, and Uses |
| WO2008141276A1 (en) * | 2007-05-11 | 2008-11-20 | Centocor, Inc. | Anti-alpha-v immunoliposome composition, methods and uses |
| HUE027911T2 (en) | 2007-09-26 | 2016-11-28 | Genentech Inc | New antibodies |
| CN104524590B (zh) | 2008-04-30 | 2019-06-21 | 伊缪诺金公司 | 交联剂和它们的用途 |
| EP3085773B1 (en) | 2008-05-30 | 2020-03-18 | XBiotech, Inc | Uses of il-1 alpha antibodies |
| EP2315747B1 (en) * | 2008-07-21 | 2017-12-06 | The Brigham and Women's Hospital, Inc. | Methods and compositions relating to synthetic beta-1,6 glucosamine oligosaccharides |
| US8242074B2 (en) | 2008-09-12 | 2012-08-14 | Xbiotech, Inc. | Modulation of the amount or function of pathogenic CD14+CD16+ monocytes |
| CN102216779B (zh) * | 2008-10-13 | 2013-11-27 | 泽普塔根股份公司 | 免疫缀合物的制备方法及其应用 |
| CN102365297B (zh) | 2009-03-25 | 2014-10-29 | 霍夫曼-拉罗奇有限公司 | 新型抗-α5β1抗体及其应用 |
| KR102444399B1 (ko) | 2009-06-03 | 2022-09-16 | 이뮤노젠 아이엔씨 | 메이탄시노이드의 제조방법 |
| FR2947269B1 (fr) * | 2009-06-29 | 2013-01-18 | Sanofi Aventis | Nouveaux composes anticancereux |
| CA2676946A1 (en) * | 2009-08-28 | 2011-02-28 | Lucie Peduto | Adam12 inhibitors and their use against inflammation-induced fibrosis |
| EP2486023A4 (en) | 2009-10-06 | 2014-05-07 | Immunogen Inc | EFFICIENT CONJUGATES AND HYDROPHILIC BINDER |
| KR20130009760A (ko) | 2010-02-10 | 2013-01-23 | 이뮤노젠 아이엔씨 | Cd20 항체 및 이의 용도 |
| WO2011162933A1 (en) * | 2010-06-04 | 2011-12-29 | Immunogen, Inc. | Anti-integrin immunoconjugate dosing regimens |
| ES2703774T3 (es) | 2010-06-18 | 2019-03-12 | Xbiotech Inc | Tratamiento para la artritis |
| KR102314003B1 (ko) * | 2010-08-23 | 2021-10-19 | 얀센 바이오테크 인코포레이티드 | 종양성 질병들에 대한 치료 |
| HUE039375T2 (hu) * | 2010-10-29 | 2018-12-28 | Perseus Proteomics Inc | Magas internalizálási kapacitású anti-CDH3 antitest |
| CN107537040A (zh) | 2011-03-29 | 2018-01-05 | 伊缪诺金公司 | 通过一步法制备类美登素抗体缀合物 |
| SG193996A1 (en) | 2011-03-29 | 2013-11-29 | Immunogen Inc | Preparation of maytansinoid antibody conjugates by a one-step process |
| US9724409B2 (en) | 2011-04-01 | 2017-08-08 | Xbiotech, Inc. | Treatment of inflammatory skin disease |
| AR086363A1 (es) | 2011-04-21 | 2013-12-11 | Bayer Ip Gmbh | Conjugados de principio activo-ligante (adc) y el uso de los mismos |
| CN103648523A (zh) * | 2011-05-09 | 2014-03-19 | 弗吉尼亚大学专利基金会 | 用于治疗癌症的组合物和方法 |
| US9809649B2 (en) | 2011-09-23 | 2017-11-07 | Xbiotech, Inc. | Cachexia treatment |
| WO2013110578A1 (en) * | 2012-01-27 | 2013-08-01 | F. Hoffmann-La Roche Ag | Integrin antagonist conjugates for targeted delivery to cells expressing alpha-v-beta-3 |
| EP2814509B1 (en) * | 2012-02-17 | 2018-05-16 | Seattle Genetics, Inc. | Antibodies to integrin v 6 and use of same to treat cancer |
| US9545441B2 (en) | 2012-09-18 | 2017-01-17 | Xbiotech, Inc. | Treatment of diabetes |
| PT3486248T (pt) | 2012-09-26 | 2021-07-12 | Immunogen Inc | Métodos melhorados para a acilação de maitansinol |
| SG11201502429YA (en) | 2012-10-04 | 2015-04-29 | Immunogen Inc | Use of a pvdf membrane to purify cell-binding agent cytotoxic agent conjugates |
| US9180203B2 (en) * | 2012-10-23 | 2015-11-10 | The Johns Hopkins University | Self-assembling drug amphiphiles and methods for synthesis and use |
| CA2898239A1 (en) | 2013-01-23 | 2014-07-31 | Syddansk Universitet | Mfap4 binding antibodies blocking the interaction between mfap4 and integrin receptors |
| EP2777714A1 (en) | 2013-03-15 | 2014-09-17 | NBE-Therapeutics LLC | Method of producing an immunoligand/payload conjugate by means of a sequence-specific transpeptidase enzyme |
| EP2968593B1 (en) | 2013-03-15 | 2024-11-20 | Regeneron Pharmaceuticals, Inc. | Biologically active molecules, conjugates thereof, and therapeutic uses |
| EP3021942A4 (en) * | 2013-07-19 | 2017-04-19 | The Regents of The University of California | Milk fat globule epidermal growth factor 8 regulates fatty acid uptake |
| JP6608823B2 (ja) | 2013-08-26 | 2019-11-20 | レゲネロン ファーマシューティカルス,インコーポレーテッド | マクロライドジアステレオマーを含む医薬組成物、その合成方法、及び治療上の使用 |
| KR102329024B1 (ko) | 2013-12-23 | 2021-11-19 | 바이엘 파마 악티엔게젤샤프트 | 키네신 스핀들 단백질(ksp)과의 항체 약물 접합체 (adcs) |
| CN109147874A (zh) * | 2014-09-02 | 2019-01-04 | 伊缪诺金公司 | 用于配制抗体药物缀合物组合物的方法 |
| AU2015349985A1 (en) | 2014-11-19 | 2017-05-25 | Immunogen, Inc. | Process for preparing cell-binding agent-cytotoxic agent conjugates |
| BR112017011773A2 (pt) | 2014-12-04 | 2018-02-20 | Celgene Corporation | composto isolado, conjugados isolados, composiçôes farmacêuticas, e métodos de tratamento de um distúrbio em um mamífero |
| MX389728B (es) | 2014-12-23 | 2025-03-20 | Nbe Therapeutics Ag | Conjugados de farmacos proteinas de union que comprenden derivados de antraciclina. |
| EP3273998B1 (en) | 2015-03-27 | 2019-09-04 | Regeneron Pharmaceuticals, Inc. | Maytansinoid derivatives, conjugates thereof, and methods of use |
| WO2016207089A1 (de) | 2015-06-22 | 2016-12-29 | Bayer Pharma Aktiengesellschaft | Binder-wirkstoff-konjugate (adcs) und binder-prodrug-konjugate (apdcs) mit enzymatisch spaltbaren gruppen |
| WO2017060322A2 (en) | 2015-10-10 | 2017-04-13 | Bayer Pharma Aktiengesellschaft | Ptefb-inhibitor-adc |
| CA3013125A1 (en) | 2016-02-05 | 2017-08-10 | Immunogen, Inc. | Efficient process for preparing cell-binding agent-cytotoxic agent conjugates |
| GB201602356D0 (en) * | 2016-02-10 | 2016-03-23 | Medimmune Ltd | Pyrrolobenzodiazepine Conjugates |
| KR20180123047A (ko) | 2016-03-24 | 2018-11-14 | 바이엘 파마 악티엔게젤샤프트 | 효소적으로 절단가능한 기를 갖는 세포독성 활성제의 전구약물 |
| WO2017216028A1 (en) | 2016-06-15 | 2017-12-21 | Bayer Pharma Aktiengesellschaft | Specific antibody-drug-conjugates (adcs) with ksp inhibitors and anti-cd123-antibodies |
| US12059472B2 (en) | 2016-12-21 | 2024-08-13 | Bayer Aktiengesellschaft | Prodrugs of cytotoxic active agents having enzymatically cleavable groups |
| WO2018114578A1 (de) | 2016-12-21 | 2018-06-28 | Bayer Pharma Aktiengesellschaft | Binder-wirkstoff-konjugate (adcs) mit enzymatisch spaltbaren gruppen |
| CA3047522A1 (en) | 2016-12-21 | 2018-06-28 | Bayer Pharma Aktiengesellschaft | Specific antibody drug conjugates (adcs) having ksp inhibitors |
| SG11201907159SA (en) | 2017-02-16 | 2019-09-27 | Xbiotech Inc | Treatment of hidradenitis suppurativa |
| US10639382B2 (en) | 2017-04-13 | 2020-05-05 | National Guard Health Affairs | Method for treating leukemia |
| EP3630185A4 (en) * | 2017-05-30 | 2020-06-17 | The Board of Trustees of the Leland Stanford Junior University | Treatment of neuroinflammatory disease |
| WO2018227018A1 (en) * | 2017-06-07 | 2018-12-13 | Silverback Therapeutics, Inc. | Antibody conjugates of immune-modulatory compounds and uses thereof |
| US20200046737A1 (en) | 2018-08-09 | 2020-02-13 | Notable Labs, Inc. | Methods for treating cancer, and compositions therefor |
| JP7675060B2 (ja) | 2019-07-10 | 2025-05-12 | サイブレクサ 3,インコーポレイテッド | 治療薬としての微小管標的化剤のペプチドコンジュゲート |
| CA3146560A1 (en) | 2019-07-10 | 2021-01-14 | Cybrexa 2, Inc. | Peptide conjugates of cytotoxins as therapeutics |
| CA3095676A1 (en) | 2020-10-07 | 2022-04-07 | Xbiotech Inc. | True human antibody specific for interleukin 1 alpha (il-1a) |
| CA3095675A1 (en) | 2020-10-07 | 2022-04-07 | Xbiotech Inc. | True human antibody specific for interleukin 1 alpha (il-1a) |
| EP4545563A1 (en) * | 2022-06-27 | 2025-04-30 | Foreseen Technology (Beijing) Co., Ltd. | Antibodies targeting itga2 and antibody-drug conjugates comprising same |
| CN115350187B (zh) * | 2022-08-30 | 2023-08-29 | 中山大学 | 异丙托溴铵在抑制肺癌骨转移中的应用 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002012501A2 (en) | 2000-08-07 | 2002-02-14 | Centocor, Inc. | Anti-dual integrin antibodies, compositions, methods and uses |
| US20030103985A1 (en) | 2001-05-18 | 2003-06-05 | Boehringer Ingelheim International Gmbh | Cytotoxic CD44 antibody immunoconjugates |
Family Cites Families (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3896111A (en) | 1973-02-20 | 1975-07-22 | Research Corp | Ansa macrolides |
| SE430062B (sv) | 1977-03-04 | 1983-10-17 | Pharmacia Fine Chemicals Ab | Kopplings- eller tioleringsreagens |
| US4151042A (en) | 1977-03-31 | 1979-04-24 | Takeda Chemical Industries, Ltd. | Method for producing maytansinol and its derivatives |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| CA2026147C (en) | 1989-10-25 | 2006-02-07 | Ravi J. Chari | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| US5965132A (en) | 1992-03-05 | 1999-10-12 | Board Of Regents, The University Of Texas System | Methods and compositions for targeting the vasculature of solid tumors |
| ES2193143T3 (es) | 1992-03-05 | 2003-11-01 | Univ Texas | Uso de inmunoconjugados para la diagnosis y/o terapia de tumores vascularizaos. |
| JP3161490B2 (ja) | 1993-07-30 | 2001-04-25 | 松下電器産業株式会社 | 金型装置 |
| US6160099A (en) | 1998-11-24 | 2000-12-12 | Jonak; Zdenka Ludmila | Anti-human αv β3 and αv β5 antibodies |
| GB9909392D0 (en) * | 1999-04-24 | 1999-06-23 | Imp Cancer Res Tech | Treatment, imaging and diagnosis of disease |
| PT2283866E (pt) | 1999-06-25 | 2015-07-20 | Genentech Inc | Métodos de tratamento utilizando conjugados de anticorpo anti-erbb-maitansinóide |
| US7163681B2 (en) | 2000-08-07 | 2007-01-16 | Centocor, Inc. | Anti-integrin antibodies, compositions, methods and uses |
| AR030612A1 (es) | 2000-09-12 | 2003-08-27 | Smithkline Beecham Corp | Procedimiento e intermedios |
| EP1389090A2 (en) * | 2001-04-26 | 2004-02-18 | Board of Regents, The University of Texas System | Diagnostic imaging compositions, their methods of synthesis and use |
| EP1258255A1 (en) * | 2001-05-18 | 2002-11-20 | Boehringer Ingelheim International GmbH | Conjugates of an antibody to CD44 and a maytansinoid |
| US6441163B1 (en) * | 2001-05-31 | 2002-08-27 | Immunogen, Inc. | Methods for preparation of cytotoxic conjugates of maytansinoids and cell binding agents |
| AU2003228998A1 (en) | 2002-05-13 | 2003-12-02 | Smithkline Beecham Corporation | Process for preparing maytansinol |
| ATE499116T1 (de) | 2002-08-16 | 2011-03-15 | Immunogen Inc | Vernetzer mit hoher reaktivität und löslichkeit und ihre verwendung bei der herstellung von konjugaten für die gezielte abgabe von kleinmolekularen arzneimitteln |
| EP2281577B1 (en) * | 2003-05-14 | 2016-11-16 | ImmunoGen, Inc. | Drug conjugate composition |
| CA2525130C (en) | 2003-05-20 | 2014-04-15 | Immunogen, Inc. | Improved cytotoxic agents comprising new maytansinoids |
| US7276497B2 (en) * | 2003-05-20 | 2007-10-02 | Immunogen Inc. | Cytotoxic agents comprising new maytansinoids |
| EA013323B1 (ru) * | 2004-12-09 | 2010-04-30 | Сентокор, Инк. | Иммуноконъюгаты против интегрина, способы и варианты применения |
-
2005
- 2005-11-30 EA EA200701250A patent/EA013323B1/ru not_active IP Right Cessation
- 2005-11-30 DK DK05852484T patent/DK1819359T3/en active
- 2005-11-30 HU HUE05852484A patent/HUE025449T2/en unknown
- 2005-11-30 US US11/290,249 patent/US8603483B2/en not_active Expired - Fee Related
- 2005-11-30 CA CA002591148A patent/CA2591148A1/en not_active Abandoned
- 2005-11-30 ES ES05852484.4T patent/ES2539126T3/es not_active Expired - Lifetime
- 2005-11-30 CN CN2005800478135A patent/CN101193916B/zh not_active Expired - Fee Related
- 2005-11-30 SG SG200908161-3A patent/SG158119A1/en unknown
- 2005-11-30 WO PCT/US2005/043250 patent/WO2006062779A2/en not_active Ceased
- 2005-11-30 PL PL05852484T patent/PL1819359T3/pl unknown
- 2005-11-30 BR BRPI0515745-5A patent/BRPI0515745A/pt not_active IP Right Cessation
- 2005-11-30 PT PT58524844T patent/PT1819359E/pt unknown
- 2005-11-30 KR KR1020077015643A patent/KR101281501B1/ko not_active Expired - Fee Related
- 2005-11-30 MX MX2007007011A patent/MX2007007011A/es active IP Right Grant
- 2005-11-30 UA UAA200706239A patent/UA94388C2/ru unknown
- 2005-11-30 EP EP05852484.4A patent/EP1819359B1/en not_active Expired - Lifetime
- 2005-11-30 NZ NZ555601A patent/NZ555601A/en not_active IP Right Cessation
- 2005-11-30 JP JP2007545515A patent/JP5421532B2/ja not_active Expired - Fee Related
- 2005-11-30 AU AU2005314392A patent/AU2005314392B2/en not_active Ceased
- 2005-12-07 AR ARP050105122A patent/AR052041A1/es not_active Application Discontinuation
- 2005-12-08 TW TW094143287A patent/TWI400087B/zh not_active IP Right Cessation
-
2007
- 2007-06-05 IL IL183695A patent/IL183695A0/en unknown
- 2007-07-06 NO NO20073493A patent/NO20073493L/no not_active Application Discontinuation
- 2007-07-06 ZA ZA200705556A patent/ZA200705556B/xx unknown
-
2013
- 2013-12-04 US US14/096,767 patent/US20140093523A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2002012501A2 (en) | 2000-08-07 | 2002-02-14 | Centocor, Inc. | Anti-dual integrin antibodies, compositions, methods and uses |
| US20030103985A1 (en) | 2001-05-18 | 2003-06-05 | Boehringer Ingelheim International Gmbh | Cytotoxic CD44 antibody immunoconjugates |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101281501B1 (ko) | 항인테그린 면역 컨쥬게이트, 방법 및 용도 | |
| JP7641323B2 (ja) | 治療抗体およびその使用 | |
| US20210221907A1 (en) | Antibodies specific to trophoblast antigen 2 (trop2) | |
| JP7575102B2 (ja) | Muc18に特異的な抗体 | |
| CN109219618B (zh) | 针对表皮生长因子受体变体iii和cd3的单特异性和双特异性抗体及其用途 | |
| JP7137474B2 (ja) | NaPi2b標的化抗体-薬物コンジュゲート及びその使用方法 | |
| JP6333882B2 (ja) | 抗体−薬剤コンジュゲート | |
| TWI731856B (zh) | 抗c-Met抗體和抗c-Met抗體-細胞毒性藥物偶聯物及其醫藥用途 | |
| US12378310B2 (en) | Antibodies specific to folate receptor alpha | |
| WO2021067820A1 (en) | Formulation of antibody-drug conjugate | |
| US12364770B2 (en) | Antibodies specific to MUC18 | |
| WO2023124963A1 (zh) | 一种可逆反应减少的抗体-药物偶联物,其制备方法及应用 | |
| TW202133885A (zh) | 利用與191p4d12蛋白結合之抗體藥物結合物(adc)治療癌症 | |
| CN117295770A (zh) | 包含抗p-钙粘蛋白抗体的抗体缀合物及其用途 | |
| CA3195865A1 (en) | Anti-cspg4 binding agents, conjugates thereof and methods of using the same | |
| KR20230125129A (ko) | 항체-약물 접합체 | |
| HK1107001B (en) | Anti-integrin immunoconjugates, methods of their production and their use | |
| TW202342539A (zh) | 一種靶向ror1的結合分子及其應用 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-3-3-R10-R18-oth-X000 |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| FPAY | Annual fee payment |
Payment date: 20160527 Year of fee payment: 4 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| LAPS | Lapse due to unpaid annual fee | ||
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20170628 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20170628 |