KR101170925B1 - 암 치료용 키네신 방추 단백질 (ksp) 억제제로서의n-(1-(1-벤질-4-페닐-1h-이미다졸-2-일)-2,2-디메틸프로필)벤자미드 유도체 및 관련 화합물 - Google Patents
암 치료용 키네신 방추 단백질 (ksp) 억제제로서의n-(1-(1-벤질-4-페닐-1h-이미다졸-2-일)-2,2-디메틸프로필)벤자미드 유도체 및 관련 화합물 Download PDFInfo
- Publication number
- KR101170925B1 KR101170925B1 KR1020077000979A KR20077000979A KR101170925B1 KR 101170925 B1 KR101170925 B1 KR 101170925B1 KR 1020077000979 A KR1020077000979 A KR 1020077000979A KR 20077000979 A KR20077000979 A KR 20077000979A KR 101170925 B1 KR101170925 B1 KR 101170925B1
- Authority
- KR
- South Korea
- Prior art keywords
- imidazol
- benzyl
- phenyl
- propyl
- dimethylpropyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 *C(*)(c1nc(*)c(*)[n]1*)N(*)* Chemical compound *C(*)(c1nc(*)c(*)[n]1*)N(*)* 0.000 description 5
- ROGCPZWZLBDDHH-HNNXBMFYSA-N CC(C)(C)[C@H](c1nc(-c2ccccc2)c[nH]1)NC(OC(C)(C)C)=O Chemical compound CC(C)(C)[C@H](c1nc(-c2ccccc2)c[nH]1)NC(OC(C)(C)C)=O ROGCPZWZLBDDHH-HNNXBMFYSA-N 0.000 description 1
- HVXAJOGTXVOCBW-NDEPHWFRSA-N CC(C)(C)[C@H](c1nc(-c2ccccc2)c[n]1Cc1ccccc1)N(CCCN)C(c1ccccc1)=O Chemical compound CC(C)(C)[C@H](c1nc(-c2ccccc2)c[n]1Cc1ccccc1)N(CCCN)C(c1ccccc1)=O HVXAJOGTXVOCBW-NDEPHWFRSA-N 0.000 description 1
- WUPIMLLNSBEHCO-XCZPVHLTSA-N CC(C)(C)[C@H](c1nc(-c2ccccc2)c[n]1Cc1ccccc1)N(C[C@H](CN)O)C(c1ccccc1)=O Chemical compound CC(C)(C)[C@H](c1nc(-c2ccccc2)c[n]1Cc1ccccc1)N(C[C@H](CN)O)C(c1ccccc1)=O WUPIMLLNSBEHCO-XCZPVHLTSA-N 0.000 description 1
- NWSQACVSCHVQJT-GDLZYMKVSA-N CC(C)[C@H](c1nc(-c2ccccc2)c[n]1Cc1ccccc1)N(CCCN)C(c1ccc(C)cc1)=O Chemical compound CC(C)[C@H](c1nc(-c2ccccc2)c[n]1Cc1ccccc1)N(CCCN)C(c1ccc(C)cc1)=O NWSQACVSCHVQJT-GDLZYMKVSA-N 0.000 description 1
- OBKSTEOVEUUBBH-UHFFFAOYSA-N COC(C(CN(Cc1ccccc1)Cc1ccccc1)F)=O Chemical compound COC(C(CN(Cc1ccccc1)Cc1ccccc1)F)=O OBKSTEOVEUUBBH-UHFFFAOYSA-N 0.000 description 1
- FINHMQYKQOZUEZ-UHFFFAOYSA-N COC(CC(CO)N(Cc1ccccc1)Cc1ccccc1)=O Chemical compound COC(CC(CO)N(Cc1ccccc1)Cc1ccccc1)=O FINHMQYKQOZUEZ-UHFFFAOYSA-N 0.000 description 1
- XQYWBHMNEQPMHN-UHFFFAOYSA-N O=C(C(CCC1)N(Cc2ccccc2)C1=O)OC(c1ccccc1)=O Chemical compound O=C(C(CCC1)N(Cc2ccccc2)C1=O)OC(c1ccccc1)=O XQYWBHMNEQPMHN-UHFFFAOYSA-N 0.000 description 1
- LIGACIXOYTUXAW-UHFFFAOYSA-N O=C(CBr)c1ccccc1 Chemical compound O=C(CBr)c1ccccc1 LIGACIXOYTUXAW-UHFFFAOYSA-N 0.000 description 1
- ZZGOJAUAVFBXHX-UHFFFAOYSA-N O=C(CC(CF)N(C(c1c2cccc1)=O)C2=O)OCc1ccccc1 Chemical compound O=C(CC(CF)N(C(c1c2cccc1)=O)C2=O)OCc1ccccc1 ZZGOJAUAVFBXHX-UHFFFAOYSA-N 0.000 description 1
- KVUIKEFIXATPAO-UHFFFAOYSA-N O=C(CCC1)N(Cc2ccccc2)C1c1nc(-c2ccccc2)c[n]1Cc1cccc(Cl)c1 Chemical compound O=C(CCC1)N(Cc2ccccc2)C1c1nc(-c2ccccc2)c[n]1Cc1cccc(Cl)c1 KVUIKEFIXATPAO-UHFFFAOYSA-N 0.000 description 1
- WIEOGTQQBXPEBZ-UHFFFAOYSA-N O=CCCCc1ncc[nH]1 Chemical compound O=CCCCc1ncc[nH]1 WIEOGTQQBXPEBZ-UHFFFAOYSA-N 0.000 description 1
- MQGDIMKFAHSDQP-UHFFFAOYSA-N OC(C(CCC1)N(Cc2ccccc2)C1=O)=O Chemical compound OC(C(CCC1)N(Cc2ccccc2)C1=O)=O MQGDIMKFAHSDQP-UHFFFAOYSA-N 0.000 description 1
- XVPOTOFPQGIJRN-UHFFFAOYSA-N OC(CC(CF)N(C(c1c2cccc1)=O)C2=O)=O Chemical compound OC(CC(CF)N(C(c1c2cccc1)=O)C2=O)=O XVPOTOFPQGIJRN-UHFFFAOYSA-N 0.000 description 1
- AUMHFGPVVAUJHB-UHFFFAOYSA-N OC(CCCc1ncc[nH]1)=O Chemical compound OC(CCCc1ncc[nH]1)=O AUMHFGPVVAUJHB-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/04—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
- C07D233/20—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member with substituted hydrocarbon radicals, directly attached to ring carbon atoms
- C07D233/24—Radicals substituted by nitrogen atoms not forming part of a nitro radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D233/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings
- C07D233/54—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members
- C07D233/64—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, not condensed with other rings having two double bonds between ring members or between ring members and non-ring members with substituted hydrocarbon radicals attached to ring carbon atoms, e.g. histidine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/06—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/02—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings
- C07D405/12—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/06—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings
- C07D417/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/10—Spiro-condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
Abstract
Description
Claims (33)
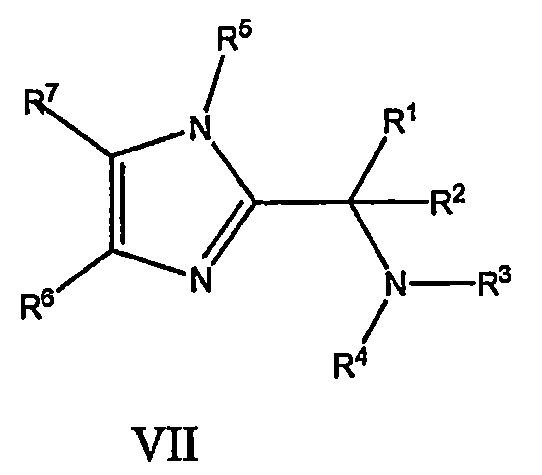
- 화학식 I의 화합물 또는 그것의 약학적으로 허용가능한 염:상기식에서,R1은 페닐, 메틸, iso-프로필, t-부틸, 1-메틸-n-프로프-1-일, 및 2-히드록시-이소-프로필로 이루어진 군으로부터 선택되고;R2는 수소, 메틸, 및 에틸로 이루어진 군으로부터 선택되고;R3는 -X-A이고, 여기서 X는 -C(O)-이고;A는 3-아미노-2-옥소-1(2H)-피리디닐메틸, 시아노메틸, (N,N-디메틸아미노)메틸, 에톡시메틸, p-플루오로페녹시메틸, 히드록시메틸, 1H-이미다졸-1-일메틸, 메톡시메틸, (N-메틸아미노)메틸, 메틸설포닐메틸, (5-메틸-1H-테트라졸-1-일)메틸, (5-메틸-2H-테트라졸-2-일)메틸, 모르폴린-4-일메틸, 1H-피라졸-1-일메틸, 1H-1,2,3-트리아졸-1-일메틸, 2H-1,2,3-트리아졸-2-일메틸, 1H-1,2,4-트리아졸-1-일메틸, 2H-1,2,4-트리아졸-2-일메틸, 4H-1,2,4-트리아졸-4-일메틸, 1H-테트라졸-1-일메틸, 1H-테트라졸-5-일메틸, 및 2H-테트라졸-2-일메틸, 이미다졸-4-일메틸, 1-메틸피라졸-3-일메틸, 피페리딘-4-일메틸, 트리플루오로메틸, 디메틸아미노에틸, 및 2-옥소-3-아미노피롤리딘-1-일메틸로 이루어진 군으로부터 선택되고;R4는 수소, 피페리딘-4-일, -(CH2)2-NH2, -CH2-아제티딘-3-일, -CH2-(2,5-디히드로피롤-3-일), -(CH2)3-이미다졸-1-일, -CH2-(1H-이미다졸-4-일), -CH2-피리딘-3-일, -CH2-(2-히드록시피리딘-4-일), -CH2-(6-히드록시피리딘-3-일), -CH2-모르폴린-2-일, -CH2-피롤리딘-3-일, -CH2-(3-플루오로피롤리딘-3-일), -CH2-(3-히드록시피롤리딘-3-일), -CH2-(4-플루오로피롤리딘-3-일), -CH2-(4-히드록시피롤리딘-3-일), -CH2-(2-히드록시메틸피롤리딘-3-일), -CH2-피페리딘-3-일, -CH2-[1H-(1,2,3-트리아졸-4-일)], -CH2CH(NH2)CH20H, -(CH2)3-OH, -(CH2)3-0(CO)-페닐, -(CH2)3-NH2, -(CH2)3-NHCH3, -(CH2)3-N(CH3)2, -(CH2)3-NHOCH3, -(CH2)3-NHS02CH3, -(CH2)3NH-(5-시아노피리딘-2-일), -(CH2)3NH-시클로프로필, -(CH2)3NH-시클로부틸, -(CH2)3-(1H-이미다졸-2-일), -(CH2)3-(2-히드록시에틸피페리딘-1-일), -(CH2)3NH(2-히드록시메틸페닐), -(CH2)3NH-(5-트리플루오로메틸피리딘-2-일), -(CH2)3NHCH2-시클로프로필, -(CH2)3NHCH2-{5-(피리딘-3-일옥시)-1H-인다졸-3-일}, -(CH2)3NHCH2-(5-메톡시-1H-인다졸-3-일), -(CH2)3NHCH2-(6-플루오로-1H-인다졸-3-일), -CH2CHOHCH2NH2, -CH2CH(CH2OH)CH2NH2, -CH2C(CH3)2CH2-N(CH3)2, -CH2C(CH3)2CH2-(4-메틸피페라진-1-일), -(CH2)2C(O)NH2, -(CH2)2CH(NH2)C(O)NH2, -(CH2)2CH(NH2)C(O)OH, -(CH2)2CH(NH2)CH2C(O)NH2, -(CH2)2CH(NH2)CH2OH, -(CH2)2CH(NH2)CH3, -(CH2)3NHC(O)CH2NH2, -(CH2)3NHC(O)CH(NH2)CH(CH3)2, -CH2CHFCH2NH2, -(CH2)2NHC(O)CH2NH2, -(CH2)3-NHCH2CH2OH, -(CH2)3-NHCH2CO2H, -(CH2)3-NHCH2CO2CH2CH3, -(CH2)3-N(CH2CH2OH)2, -(CH2)3-NHCH(CH2OH)2, -(CH2)3CH3, -(CH)2CH(NH2)CH2OH, -(CH2)2C(CH3)2NH2, -(CH2)2CH(NH2)CH2OCH3, -(CH2)2CH(NH2)CH2F, -CH2CHFCH(NH2)CH2OH, 및 -(CH2)2스피로시클로프로필-NH2로 이루어진 군으로부터 선택되고;R5는 L-A1이고, 여기서 L은 메틸렌이고 A1은 페닐, 6-아미노피리딘-2-일, 3-클로로페닐, 3-시아노페닐, 2,4-디플루오로페닐, 2,5-디플루오로페닐, 3,5-디플루오로페닐, 2-플루오로페닐, 3-플루오로페닐, 4-플루오로페닐, 3-히드록시페닐, 3-메톡시페닐, 1-(5-메틸)-이속사졸-3-일, 2-메틸페닐, 3-메틸페닐, 4-메틸페닐, 피리딘-2-일, 피리딘-3-일, 피리딘-4-일, 테트라히드로피란-4-일, 티아졸-4-일, 및 5-트리플루오로메틸푸란-2-일로 이루어진 군으로부터 선택되고; 및R6은 페닐, 피리디닐, 피라진일, 및 티아졸릴으로 이루어진 군으로부터 선택되며, 이들은 선택적으로 -(R8)m 로 치환될 수 있고, 이때 R8은 하기에서 정의된 바와 같고 m은 1 내지 3의 정수이고, 및R7 은 수소, 할로, 및 알킬로 이루어진 군으로부터 선택되고,R8은 시아노, 알킬, 알케닐, 알키닐, -CF3, 알콕시, 할로, 및 히드록시로 이루어진 군으로부터 선택된다.
- 제 1 항에 있어서, 화학식 II로 표시되는 화합물 또는 그것의 약학적으로 허용가능한 염인 것을 특징으로 하는 화합물:상기 식에서,A, R4, 및 R8은 청구항 1에 정의된 바와 같으며;Ar은 페닐, 피리디닐, 피라진일, 및 티아졸릴로 이루어진 군으로부터 선택되고;X1은 -C(O)- 이고;A2는 페닐, 6-아미노피리딘-2-일, 3-클로로페닐, 3-시아노페닐, 2,4-디플루오로페닐, 2,5-디플루오로페닐, 3,5-디플루오로페닐, 2-플루오로페닐, 3-플루오로페닐, 4-플루오로페닐, 3-히드록시페닐, 3-메톡시페닐, 1-(5-메틸)-이속사졸-3-일, 2-메틸페닐, 3-메틸페닐, 4-메틸페닐, 피리딘-2-일, 피리딘-3-일, 피리딘-4-일, 테트라히드로피란-4-일, 티아졸-4-일, 및 5-트리플루오로메틸푸란-2-일로 이루어진 군으로부터 선택되고;R12는 수소, 메틸, 및 에틸로 이루어진 군으로부터 선택되고;R13은 페닐, 메틸, iso-프로필, t-부틸, 1-메틸-n-프로프-1-일, 및 2-히드록시-iso-프로필로 이루어진 군으로부터 선택되고;R14는 수소 또는 C1 내지 C4 알킬이고;m1은 0 내지 2의 정수이며;n은 1이다.
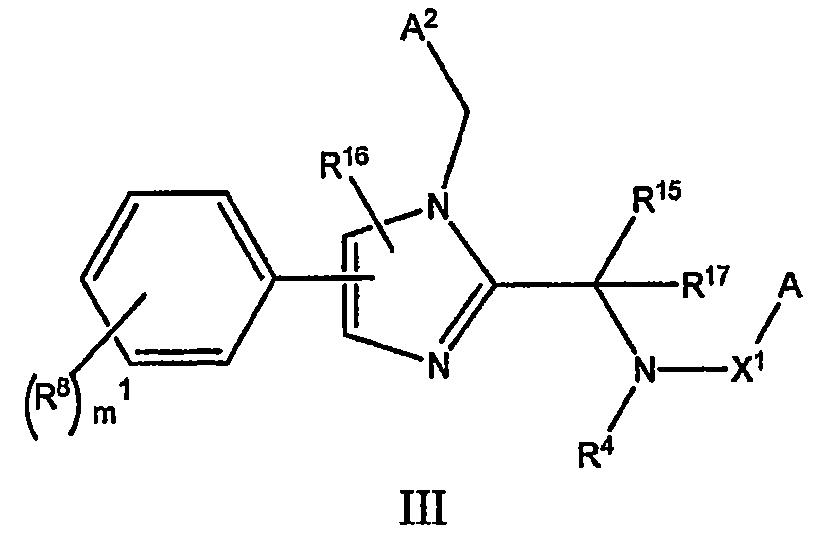
- 제 1 항에 있어서, 화학식 III로 표시되는 화합물, 또는 그것의 약학적으로 허용가능한 염인 것을 특징으로 하는 화합물:상기 식에서,A, R4, 및 R8은 청구항 1에 정의된 바와 같으며;X1은 -C(O)-이고;A2는 페닐, 6-아미노피리딘-2-일, 3-클로로페닐, 3-시아노페닐, 2,4-디플루오로페닐, 2,5-디플루오로페닐, 3,5-디플루오로페닐, 2-플루오로페닐, 3-플루오로페닐, 4-플루오로페닐, 3-히드록시페닐, 3-메톡시페닐, 1-(5-메틸)-이속사졸-3-일, 2-메틸페닐, 3-메틸페닐, 4-메틸페닐, 피리딘-2-일, 피리딘-3-일, 피리딘-4-일, 테트라히드로피란-4-일, 티아졸-4-일, 및 5-트리플루오로메틸푸란-2-일로 이루어진 군으로부터 선택되고;R15는 이소프로필, t-부틸, 또는 페닐로 이루어진 군으로부터 선택되고;R16은 수소 또는 메틸이고;R17은 수소 또는 메틸이며;m1은 0, 1 또는 2의 정수이다.
- 제 1 항에 있어서, 화학식 IV로 표시되는 것을 특징으로 하는 화합물, 또는 그것의 약학적으로 허용가능한 염인 것을 특징으로 하는 화합물:상기 식에서 X1은 -C(O)-이고;A, R4 및 R8은 청구항 1에 정의된 바와 같으며;A2는 페닐, 6-아미노피리딘-2-일, 3-클로로페닐, 3-시아노페닐, 2,4-디플루오로페닐, 2,5-디플루오로페닐, 3,5-디플루오로페닐, 2-플루오로페닐, 3-플루오로페닐, 4-플루오로페닐, 3-히드록시페닐, 3-메톡시페닐, 1-(5-메틸)-이속사졸-3-일, 2-메틸페닐, 3-메틸페닐, 4-메틸페닐, 피리딘-2-일, 피리딘-3-일, 피리딘-4-일, 테트라히드로피란-4-일, 티아졸-4-일, 및 5-트리플루오로메틸푸란-2-일로 이루어진 군으로부터 선택되고;R15는 이소프로필, t-부틸, 또는 페닐로 이루어진 군으로부터 선택되고;R16은 수소 또는 메틸이고;R17은 수소 또는 메틸이고;m1은 0, 1 또는 2의 정수이다.
- 제 1 항에 있어서, 화학식 V로 표시되는 것을 특징으로 하는 화합물, 또는 그것의 약학적으로 허용가능한 염인 것을 특징으로 하는 화합물:상기 식에서 X1은 -C(O)-이고;A, R4 및 R8은 청구항 1에 정의된 바와 같으며;A2는 페닐, 6-아미노피리딘-2-일, 3-클로로페닐, 3-시아노페닐, 2,4-디플루오로페닐, 2,5-디플루오로페닐, 3,5-디플루오로페닐, 2-플루오로페닐, 3-플루오로페닐, 4-플루오로페닐, 3-히드록시페닐, 3-메톡시페닐, 1-(5-메틸)-이속사졸-3-일, 2-메틸페닐, 3-메틸페닐, 4-메틸페닐, 피리딘-2-일, 피리딘-3-일, 피리딘-4-일, 테트라히드로피란-4-일, 티아졸-4-일, 및 5-트리플루오로메틸푸란-2-일로 이루어진 군으로부터 선택되고;R15는 이소프로필, t-부틸, 또는 페닐로 이루어진 군으로부터 선택되고;R16은 수소 또는 메틸이고;R17은 수소 또는 메틸이고;m1은 0, 1 또는 2의 정수이다.
- 제 1 항에 있어서, 화학식 VI로 표시되는 것을 특징으로 하는 화합물, 또는 그것의 약학적으로 허용가능한 염인 것을 특징으로 하는 화합물:상기 식에서 X1은 -C(O)-이고;A, R4 및 R8은 청구항 1에 정의된 바와 같으며;A2는 페닐, 6-아미노피리딘-2-일, 3-클로로페닐, 3-시아노페닐, 2,4-디플루오로페닐, 2,5-디플루오로페닐, 3,5-디플루오로페닐, 2-플루오로페닐, 3-플루오로페닐, 4-플루오로페닐, 3-히드록시페닐, 3-메톡시페닐, 1-(5-메틸)-이속사졸-3-일, 2-메틸페닐, 3-메틸페닐, 4-메틸페닐, 피리딘-2-일, 피리딘-3-일, 피리딘-4-일, 테트라히드로피란-4-일, 티아졸-4-일, 및 5-트리플루오로메틸푸란-2-일로 이루어진 군으로부터 선택되고;R15는 이소프로필, t-부틸, 또는 페닐로 이루어진 군으로부터 선택되고;R16은 수소 또는 메틸이고;R17은 수소 또는 메틸이고;m1은 0, 1 또는 2의 정수이다.
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 제 1 항에 있어서, R6은 페닐, 3-브로모페닐, 3-클로로페닐, 4-시아노페닐, 2,5-디플루오로페닐, 3-플루오로페닐, 2-메톡시페닐, 3-메톡시페닐, 4-메톡시페닐, 4-메틸페닐, 피라진-2-일, 피리딘-2-일, 티아졸-2-일, 2-트리플루오로메틸페닐, 및 3-트리플루오로메틸페닐로 이루어진 군으로부터 선택되는 것을 특징으로 하는 화합물.
- 삭제
- 삭제
- 삭제
- 하기로 이루어진 군으로부터 선택되는 것을 특징으로 하는 화합물 및 그것의 약학적으로 허용가능한 염:N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]벤자미드;N-(3-아미노프로필)-N-[(R)-(1-벤질-4-페닐-1H-이미다졸-2-일)(페닐)메틸]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(R)-(1-벤질-4-페닐-1H-이미다졸-2-일)(페닐)메틸]벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일)-2-메틸프로필}-4-(트리플루오로메틸)벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2,4-디플루오로벤자미드;N-(3-아미노프로필)-4-메틸-N-{2-메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-메톡시페닐)-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-메톡시페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-메톡시페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-(트리플루오로메틸)벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-메톡시페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2,4-디플루오로벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-(트리플루오로메틸)벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2,4-디플루오로벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(4-메틸페닐)-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(4-메틸페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(4-메틸페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-(트리플루오로메틸)벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(4-메틸페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2,4-디플루오로벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3,5-디플루오로벤질)-4-페닐-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3,5-디플루오로벤질)-4-페닐-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4- 메틸-N-{3-[(메틸설포닐)아미노]프로필}벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-N-[(3R)-피페리딘-3-일메틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-N-[(3S)-피페리딘-3-일메틸]벤자미드;N-(3-아미노프로필)-4-메틸-N-{(1R)-2-메틸-1-[1-(4-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필)벤자미드;N-(3-아미노프로필)-N-{(1R)-2-메틸-1-[1-(4-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}벤자미드;N-(3-아미노프로필)-4-메틸-N-{(1R)-2-메틸-1-[1-(2-메틸벤질)-4-페닐-1H-이미다졸-2-일] 프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-2-메틸-1-[1-(2-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-N-[2,2-디메틸-3-(4-메틸피페라진-1-일)프로필]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-N-[2,2-디메틸-3-(4-메틸피페라진-1-일)프로필]-4-메틸벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸-N-[(3R)-피페리딘-3-일메틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸-N-[(3S)-피페리딘-3-일메틸]벤자미드;N-(3-아미노프로필)-N-((1R)-1-{1-벤질-4-[2-(트리플루오로메틸)페닐]-1H-이미다졸-2-일}-2-메틸프로필)벤자미드;N-(3-아미노프로필)-N-((1R)-1-{1-벤질-4-[2-(트리플루오로메틸)페닐]-1H-이미다졸-2-일)-2-메틸프로필)-4-메틸벤자미드;N-(3-아미노프로필)-N-((1R)-1-{1-벤질-4-[2-(트리플루오로메틸)페닐]-1H-이미다졸-2-일}-2-메틸프로필)-2,4-디플루오로벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-메톡시벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-3-클로로벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-클로로벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-tert-부틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-3,4-디클로로벤자미드;N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일]-2-메틸프로필)-N-{3-[(5-시아노피리딘-2-일)아미노]프로필}벤자미드;N-{(1R)-1-[1-벤질-4-(3-브로모페닐)-1H-이미다졸-2-일]-2-메틸프로필}-N-(3-[5-(트리플루오로메틸)피리딘-2-일]아미노}프로필)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-N-피페리딘-4-일벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸-N-피페리딘-4-일벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-2,4-디플루오로- N-피페리딘-4-일벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}시클로헥산카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-6-클로로니코틴아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{((1R)-1-[4-(3-클로로페닐)-1-(2,4-디플루오로벤질)-1H-이미다졸-2-일]-2,2-디메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[4-(3-클로로페닐)-1-(2,4-디플루오로벤질)-1H-이미다졸-2-일]-2,2-디메틸프로필}-4-메틸벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-N-[3-(디메틸아미노)-2,2-디메틸프로필]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-H-이미다졸-2-일)-2-메틸프로필]-N-[3-(디메틸아미노)-2,2-디메틸프로필]-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}-6-클로로니코틴아미드;N-(3-아미노프로필)-6-클로로-N-{(1R)-1-[4-(3-클로로페닐)-1-(3,5-디플루오로벤질)-1H-이미다졸-2-일]-2,2-디메틸프로필}니코틴아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-5-메틸-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-5-메틸-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-5-메틸-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-2,4-디플루오로벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-5-메틸-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-6-클로로니코틴아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-클로로벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-클로로-6-메틸이소니코틴아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-3-푸라미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-5-메틸-2H-이미다졸-4-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-메톡시벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}피리딘-2-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-1,5-디메틸-1H-피라졸-3-카르복사미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-1,2-디메틸프로필]벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-1,2-디메틸프로필]-4-메틸벤자미드;N-(2-아미노에틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸벤자미드;N-(2-아미노에틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-1,3-티아졸-4-카르복사미드;N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필)- N-(피롤리딘-3-일메틸)벤자미드;N-{(1R)-1-[4-(3-클로로페닐)-1-(3,5-디플루오로벤질)-1H-이미다졸-2-일]-2,2-디메틸프로필}-N-(피롤리딘-3-일메틸)벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}벤젠설폰아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸이속사졸-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필)이속사졸-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필)-5-메틸-1H-피라졸-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2,4-디메틸-1,3-티아졸-5-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}테트라히드로푸란-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}피페리딘-4-카르복사미드;2-아미노-N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-1,3-티아졸-4-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(1H-이미다졸-4-일메틸)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(2-히드록시피리딘-4-일)메틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(6-히드록시피리딘-3-일) 메틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(피리딘-3-일메틸)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[3-(시클로부틸아미노)프로필]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(피롤리딘-3-일메틸)벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-6-메틸니코틴아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{3-[(시클로프로필메틸)아미노]프로필}벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[3-({[5-(피리딘-3-일옥시)-1H-인다졸-3-일]메틸}아미노)프로필]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(5-메톡시-1H-인다졸-3-일)메틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(3-{[(6-플루오로-1H-인다졸-3-일)메틸]아미노}프로필)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[5-(피리딘-3-일옥시)-1H-인다졸-3-일]메틸)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(3-히드록시프로필) 벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3R)- 3,5-디아미노-5-옥소펜틸]벤자미드;N-{3-[(아미노아세틸)아미노]프로필}-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-{2-[(아미노아세틸)아미노]에틸}-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-1,5-디메틸-N-(피페리딘-3-일메틸)-1H-피라졸-3-카르복사미드;(2R)-N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}테트라히드로푸란-2-카르복사미드;(2S)-N-(3-아미노프로필)-N-((1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}테트라히드로푸란-2-카르복사미드;N-(3-아미노프로필)-N-((1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}피라진-2-카르복사미드;3-{벤조일[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]아미노)프로필 벤조에이트;N-{(1R)-1-[4-(3-클로로페닐)-1-(3,5-디플루오로벤질)-1H-이미다졸-2-일]-2,2-디메틸프로필}-1,5-디메틸-N-[(3R)-피롤리딘-3-일메틸]-1H-피라졸-3-카르복사미드;N-{(1R)-1-[4-(3-클로로페닐)-1-(3,5-디플루오로벤질)-1H-이미다졸-2-일]- 2,2-디메틸프로필)-1,5-디메틸-N-[(3,5)-피롤리딘-3-일메틸]-1H-피라졸-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필)-1-메틸-3-(트리플루오로메틸)-1H-피라졸-4-카르복사미드;N-(3-아미노프로필)-N-((1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-5-클로로-1-메틸-1H-피라졸-4-카르복사미드;N-(3-아미노프로필)-N-((1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-(디메틸아미노)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[3-(1H-이미다졸-2-일)프로필]벤자미드;1-벤질-6-(1-벤질-4-페닐-1H-이미다졸-2-일)피페리딘-2-온;1-벤질-6-[1-(3-클로로벤질)-4-페닐-1H-이미다졸-2-일]피페리딘-2-온;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{3-[(2- 히드록시에틸)아미노]프로필벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[3-(시클로프로필아미노)프로필]벤자미드;N-(3-아미노프로필)-N-{(1R)-2-메틸-1-[4-페닐-1-(1,3-티아졸-4-일메틸)-1H-이미다졸-2-일]프로필)벤자미드;N-(3-아미노프로필)-4-메틸-N-{(1R)-2-메틸-1-[4-페닐-1-(1,3-티아졸-4-일메틸)-1H-이미다졸-2-일]프로필}벤자미드;N-(3-아미노프로필)-2,4-디플루오로-N-{(1R)-2-메틸-1-[4-페닐-1-(1,3-티아졸-4-일메틸)-1H-이미다졸-2-일]프로필}벤자미드;N-(3-아미노프로필)-6-클로로-N-{(1R)-2-메틸-1-[4-페닐-1-(1,3 -티아졸-4-일메틸)-1H-이미다졸-2-일]프로필}니코틴아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-피리딘-2-일-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-피리딘-2-일-1H-이미다졸-2-일)-2,2-디메틸프로필]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-피리딘-2-일-1H-이미다졸-2-일)-2,2-디메틸프로필]-2,4-디플루오로벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-피리딘-2-일-1H-이미다졸-2-일)-2,2-디메틸프로필]-6-클로로니코틴아미드;N-(2-아미노-3-히드록시프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필] 벤자미드;N-(3-아미노프로필)-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-3-메틸부틸]벤자미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-3-메틸부틸]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(1R,2R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸부틸]벤자미드;N-(3-아미노프로필)-N-[(1R,2R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸부틸]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(1R,2S)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸부틸]벤자미드;N-(3-아미노프로필)-N-[(1R,2S)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸부틸]-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일)-2-메틸프로필}-5-메틸이속사졸-4-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필)-2-(4-플루오로페녹시)아세트아미드;N-(3-아미노프로필)-N-{((1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필)-2-(1H-이미다졸-1-일)아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-모르폴린-4-일아세트아미드;[(3-{벤조일[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필] 아미노}프로필)아미노]아세트산;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-히드록시아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-메톡시아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-1-에틸-3-메틸-1H-피라졸-5-카르복사미드;에틸 [(3-{벤조일[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]아미노}프로필)아미노]아세테이트;N-{3-[(2-아미노-3-메틸부타노일)아미노]프로필}-N-[(1R)-1-(1-벤질-4-페닐- 1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}테트라히드로-2H-피란-4-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-(메틸아미노)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-프로필벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-부틸벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-부틸- 1,5-디메틸-1H-피라졸-3-카르복사미드;[[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필](부틸)아미노](옥소)아세트산;[[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필](프로필)아미노](옥소)아세트산;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{3-[2-(히드록시메틸)피페리딘-1-일]프로필 벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{3-[bis(2-히드록시에틸)아미노]프로필}벤자미드;1-(2-아미노-1-페닐에틸)-6-(1-벤질-4-페닐-1H-이미다졸-2-일)피페리딘-2-온;6-(1-벤질-4-페닐-1H-이미다졸-2-일)피페리딘-2-온;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(3-{[2- 히드록시-1-(히드록시메틸)에틸]아미노}프로필)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(3-{[2- (히드록시메틸)페닐]아미노}프로필)벤자미드;N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-N- 피페리딘-4-일벤자미드;N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-N-(피페리딘-3-일메틸)벤자미드;2-(3-아미노-2-옥소피리딘-1(2H)-일)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]아세트아미드;N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-피롤리딘-3-일벤자미드;(1R,3S)-3-{[(2-아미노에틸)설포닐]아미노}-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]시클로헥산카르복사미드;(1R,3S)-3-[(아미노아세틸)아미노]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]시클로헥산카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-6-(트리플루오로메틸)니코틴아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;(3R)-N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}피페리딘-3-카르복사미드;(3S)-N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸- 2-일]-2-메틸프로필}피페리딘-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-(1H-1,2,4-트리아졸-1-일)아세트아미드;[(5R)-1-벤조일-5-(1-벤질-4-페닐-1H-이미다졸-2-일)피롤리딘-2-일]메틸아민;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2-(4H-1,2,4-트리아졸-4-일)아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필}-2,1,3-벤조티아디아졸-4-카르복사미드;N-[(2S)-3-아미노-2-히드록시프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(l 1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-N-[(2S)-모르폴린-2-일메틸]-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1, 5-디메틸-N-[(2R)-모르폴린-2-일메틸]-1H-피라졸-3-카르복사미드;N-[(2S)-3-아미노-2-히드록시프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(2S)-3-아미노-2-히드록시프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸 -2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-피롤리딘-3-일벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-벤질-4-(3-클로로페닐)-1H-이미다졸-2-일]-2-메틸프로필]-2-(1H-피라졸-1-일)아세트아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;메틸 3-아미노프로필 [(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필] 카르바메이트;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1H-테트라졸-1-일)아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1H-1,2,4-트리아졸-1-일)아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(4H-1,2,4-트리아졸-4-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시-N-[(2S)-모르폴린-2-일메틸]아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시- N-[(2R)-모르폴린-2-일메틸]아세트아미드;N-(3-아미노프로필)-N-[(1S)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-히드록시-2-메틸프로필] 벤자미드;N-[(1R)-1-(1-벤질-4-페닐-H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시-N-[(2S)-모르폴린-2-일메틸]아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시-N-[(2R)-모르폴린-2-일메틸]아세트아미드;(2S)-N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]테트라히드로푸란-2-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[4-(3-클로로페닐)-1-(3-시아노벤질)-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[4-(3-클로로페닐)-1-(3-시아노벤질)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[4-(3-클로로페닐)-1-(3-메틸벤질)-1H-이미다졸-2-일]-2-메틸프로필}-4-메틸벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[4-(3-클로로페닐)-1-(3-메틸벤질)-1H-이미다졸-2-일]-2-메틸프로필}벤자미드;에틸 3-아미노프로필[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]카르바메이트;네오펜틸 3-아미노프로필[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디 메틸프로필]카르바메이트;이소부틸 3-아미노프로필[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]카르바메이트;프로필 3-아미노프로필[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]카르바메이트;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-시아노아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시-N-[(3R)-피롤리딘-3-일메틸]아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시-N-[(3S)-피롤리딘-3-일메틸]아세트아미드;부틸 3-아미노프로필[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필] 카르바메이트;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시-N-[(3R)-피롤리딘-3-일메틸]아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시-N-[(3S)-피롤리딘-3-일메틸]아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3S)- 피롤리딘-3-일메틸] 벤자미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸 -2-일)-2,2-디메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-yl)-2,2-디메틸프로필]-2-(1H-테트라졸-1-일)아세트아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3S)- 3,4-디아미노-4-옥소부틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3R)- 3,4-디아미노-4-옥소부틸]벤자미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;(2S)-2-아미노-4-{벤조일[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]아미노}부탄산;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1H-테트라졸-5-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3S)-피 롤리딘-3 -일메틸]-2-(1 H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3S)- 피롤리딘-3-일메틸]-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-6-메틸-N-[(3S)-피롤리딘-3-일메틸]니코틴아미드;N-(아제티딘-3-일메틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}- N-(피롤리딘-3-일메틸)벤자미드;N-{(1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}-N-(피롤리딘-3-일메틸)벤자미드;N-[(2S)-3-아미노-2-히드록시프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[3-(메톡시아미노)프로필]벤자미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1H-테트라졸-1-일)아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}- 2-메톡시-N-[(3S)-피롤리딘-3-일메틸]아세트아미드;N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}-2-메톡시-N-[(3R)-피롤리딘-3-일메틸]아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필} 벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}-2-메톡시아세트아미드;2-히드록시-N-{(1R)-1-[1-(3-히드록시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필)-N-(피롤리딘-3-일메틸)아세트아미드;N-{(1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}-2-메톡시-N-[(3S)-피롤리딘-3-일메틸]아세트아미드;N-((1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}-2-메톡시-N-[(3R)-피롤리딘-3-일메틸] 아세트아미드;N-(3-아미노프로필)-N-{(1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}-2-메톡시아세트아미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-(3-아미노-2-히드록시프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1 H-테트라졸-1-일)아세트아미드;N-(3-아미노-2-히드록시프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2- 일)-2,2-디메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,,5-디메틸-N-[(35)-피롤리딘-3-일메틸]-1H-피라졸-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필} 벤자미드;N-(아제티딘-3-일메틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-(아제티딘-3-일메틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-히드록시아세트아미드;N-(아제티딘-3-일메틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1 H-1,2, 3-트리아졸-1-일)아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(2H-1,2,3 -트리아졸-2-일)아세트아미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1 H-테트라졸-1-일)아세트아미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸- 2-일)-2,2-디메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1H-1,2,3-트리아졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3-플루오로피롤리딘-3-일)메틸]아민;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S)- 3-히드록시피롤리딘-3-일]메틸)-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-([(3R)- 3-히드록시피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3-히드록시피롤리딘-3-일)메틸]-2-메톡시아세트아미드;N-(아제티딘-3-일메틸)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-6-메틸니코틴아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3S)- 3-플루오로피롤리딘-3-일]메틸}-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R)-3-플루오로피롤리딘-3-일]메틸)-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3-플루오로피롤리딘-3-일)메틸]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S)- 3-플루오로피롤리딘-3-일]메틸)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R)- 3-플루오로피롤리딘-3-일]메틸}벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3-플루오로피롤리딘-3-일)메틸]벤자미드;N-(3-아미노프로필)-2-히드록시-N-{(1R)-1-[1-(3-히드록시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}아세트아미드;N-{(1R)-1-[1-(3-히드록시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}-N-(피롤리딘-3 -일메틸)벤자미드;N-(3-아미노프로필)-N-{((1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}-1, 5-디메틸-1H-피라졸-3-카르복사미드;N-((1R)-2,2-디메틸-1-[1-(3-메틸벤질)-4-페닐-1H-이미다졸-2-일]프로필}-1,5-디메틸-N-(피롤리딘-3-일메틸)-1H-피라졸-3-카르복사미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-히드록시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-메톡시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}벤자미드;N-(3-아미노프로필)-2-메톡시-N-{(1R)-1-[1-(3-메톡시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-시아노벤질)-4-페닐- IH-이미다졸-2-일 l-2,2-디메틸프로필}-2-히드록시아세트아미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-메톡시벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(3S)-3-아미노-4-메톡시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]벤자미드;N-(3-아미노프로필)-N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필l-1,5-디메틸-1H-피라졸-3-카르복사미드;N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필}- 1,5-디메틸-N-(피롤리딘-3-일메틸)-1H-피라졸-3-카르복사미드;N-{(1R)-1-[1-(3-시아노벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸프로필)-2-히드록시-N-[(3S)-피롤리딘-3-일메틸]아세트아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1 H-1,2,3-트리아졸-1-일)아세트아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(2H-1,2,3-트리아졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{(3S)- 3-히드록시피롤리딘-3-일]메틸)벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필)-N-{[(3R)- 3-히드록시피롤리딘-3-일]메틸}벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3-히드 록시피롤리딘-3-일)메틸]벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-에톡시- N-[(3S)-피롤리딘-3-일메틸]아세트아미드;N-(3-아미노프로필)-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-에톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S)-3-히드록시피롤리딘-3-일]메틸)-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R)- 3-히드록시피롤리딘-3-일]메틸}-1, 5-디메틸-1H-피라졸-3-카르복사미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-[(3- 히드록시피롤리딘-3-일)메틸]-1,5-디메틸-1H-피라졸-3-카르복사미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1S)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-히드록시-2-메틸프로필]-2-(1H-테트라졸-1-일)아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-[(1S)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-히드록시-2-메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-{(1R)-1-[1-벤질-4-(3-플루오로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}-2-(1 H-테트라졸-1-일)아세트아미드;N-[(2S)-3-아미노-2-플루오로프로필]-N-{(1R)-1-[1-벤질-4-(3-플루오로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필)-2-(2H-테트라졸-2-일)아세트아미드;N-[(2S)-3-아미노-2-(히드록시메틸)프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이 미다졸-2-일)-2,2-디메틸프로필]-N'-메틸에탄디아미드;N-[(2R)-3-아미노-2-(히드록시메틸)프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N'-메틸에탄디아미드;N-[(3R)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(3R)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(1H-테트라졸-1-일)아세트아미드;N-[(3R)-3-아미노-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2yl)-2,2-디메틸프로필]-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S)- 3-플루오로피롤리딘-3-일]메틸)-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R)- 3-플루오로피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R,4R)-4-히드록시피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3 S,4S)-4-히드록시피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3 R,4R)-4-히드록시피롤리딘-3-일]메틸}벤자미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S,4 S)-4-히드록시피롤리딘-3-일]메틸}벤자미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-{(1R)-1-[1-벤질-4-(3-플루오로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}-2-(1H-테트라졸-1-일)아세트아미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-{(1R)-1-[1-벤질-4-(3-플루오로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}-2-(2H-테트라졸-2-일)아세트아미드;N-[(3S)-3-아미노-4-메톡시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(3S)-3-아미노-4-히드록시부틸]-N-{(1R)-1-[1-벤질-4-(3-플루오로페닐)-1H-이미다졸-2-일]-2,2-디메틸프로필}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S)-3-플루오로피롤리딘-3-일]메틸}-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R)-3-플루오로피롤리딘-3-일]메틸}-2-(1H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3 S,4S)-4-히드록시피롤리딘-3-일]메틸}-2-(1 H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3S,4S)-4-히드록시피롤리딘-3-일]메틸}-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-([(3R,4R)-4-히드록시피롤리딘-3-일]메틸)-2-(1H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N- {[(3R,4R)-4-히드록시피롤리딘-3-일]메틸}-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(2S,3S)-2-(히드록시메틸)피롤리딘-3-일]메틸}-2-(2H-테트라졸-2-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(2S,3S)-2-(히드록시메틸)피롤리딘-3-일]메틸}-2-(1H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(2S,3S)-2-(히드록시메틸)피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(2S,3 S)-2-(히드록시메틸)피롤리딘-3-일]메틸}벤자미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(5-메틸-2H-테트라졸-2-일)아세트아미드;N-[(2R)-3-아미노-2-플루오로프로필]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-(5-메틸-1H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R)- 3-히드록시피롤리딘-3-일]메틸}-2-(1H-테트라졸-1-일)아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필)-N-{[(3S)- 3-히드록시피롤리딘-3-일] 메틸}-2-(1 H-테트라졸-1-일)아세트아미드;(5S)-3-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1-옥 사-3,7-디아자스피로 [4.4]노난-2-온;(5R)-3-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-1-옥사-3,7-디아자스피로 [4.4]노난-2-온;N-[(2S,3R)-3-아미노-2-플루오로-4-히드록시부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(3S)-3-아미노-4-플루오로부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(3R)-3-아미노-4-플루오로부틸]-N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R,4S)-4-히드록시피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3 S,4R)-4-히드록시피롤리딘-3 -일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-{[(3R,4 S)-4-플루오로피롤리딘-3-일]메틸}-2-메톡시아세트아미드;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸프로필]-N-(2,5- 디히드로-1H-피롤-3-일메틸)-2-메톡시아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-4-피라진-2-일-IH-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-(R)-1-[1-벤질-4-(3-메톡시-페닐)- 1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-에톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-((R)-1-[1-벤질-4-(3-메톡시-페닐)- I H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-[(R)-1-(1-벤질-4-피라진-2-일-1H-이미다졸-2-일)-2,2-디메틸-프로필]-아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-4-피라진-2-일-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-에톡시-아세트아미드;N-((R)-3 -아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-에톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-[(R)-2,2-디메틸-1-(4-페닐-1-피리딘-3-일메틸-1H-이미다졸-2-일)-프로필)-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-[(R)-2,2-디메틸-1-(4-페닐-1-피리딘-3-일메틸-1H-이미다졸-2-일)-프로필]-2-메톡시-아세트아미드;N-((S)-3-아미노-4-메톡시-부틸)-N-{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-벤질-4-(4-시아노-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-벤질-4-(4-메톡시-페닐)- 1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-(2-플루오로-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-(3-플루오로-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-(3-클로로-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-((R)-1-[1-(3-시아노-벤질)-4-페닐- H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-2,2-디메틸-1-[4-페닐-1-(테트라히드로-피란-4-일메틸)-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-{(R)-1-[1-벤질-4-(2-메톡시-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-{(R)-1-[1-벤질-4-(3-트리플루오로메틸-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필 )-아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)- {(R)-1-[1-벤질-4-(3-브로모-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-벤질-4-(2-메톡시-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-벤질-4-(3-트리플루오로메틸- 페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-벤질-4-(3-브로모-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-4-p-톨릴-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-[(R)-2,2-디메틸-1-(4-페닐-1-피리딘-4-일메틸-1H-이미다졸-2-일)-프로필]-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-[(R)-2,2-디메틸-1-(4-페닐-1-피리딘-4-일메틸-1H-이미다졸-2-일)-프로필]-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-2,2-디메틸-1-[1-(5-메틸-이속사졸-3 -일메틸)-4-페닐-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-{(R)-2,2-디메틸-1-[1-(5-메틸-이속사졸-3-일메틸)-4-페닐-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-[(R)-2,2-디메틸-1-(4-페닐-1-피리딘- 2-일메틸-1H-이미다졸-2-일)-프로필]-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-[(R)-2,2-디메틸-1-(4-페닐-1-피리딘-2-일메틸-1H-이미다졸-2-일)-프로필]-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-(3-아미노-3-메틸-부틸)-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-[(R)-1-(1- 벤질-5-클로로-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-5-클로로-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((R)-3-아미노-2-플루오로-프로필)-[(R)-1-(1-벤질-4-티아졸-2-일-1H-이미다졸-2-일)-2,2-디메틸-프로필]-아미드;N-((S)-3-아미노-2-플루오로-프로필)-N-{(R)-2,2-디메틸-1-[4-페닐-1-(5-트리플루오로메틸-푸란-2-일메틸)-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-2-플루오로-프로필)-N-{(R)-2,2-디메틸-1-[1-(5-메틸-이속사졸-3-일메틸)-4-페닐-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산((S)-3-아미노-2-플루오로-프로필)-{(R)-2,2-디메틸-1-[1-(5-메틸-이속사졸-3-일메틸)-4-페닐-1H-이미다졸-2-일]-프로필}-아미드;(S)-테트라히드로-푸란-2-카르복시산 ((S)-3-아미노-2-플루오로-프로필)-{(R)-2,2-디메틸-1-[4-페닐-1-(5-트리플루오로메틸-푸란-2-일메틸)-1H-이미다졸-2-일]-프로필 -아미드;N-((S)-3-아미노-4-메톡시-부틸)-N-((R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로- 페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-히드록시-부틸)-N-((R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(2,5-디플루오로-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-(R)-1-[1-(2,5-디플루오로-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-1-[1-(4-플루오로-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-2-메톡시-N-{(R)-1-[1-(3-메톡시-벤질)-4-페닐-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-2,2-디메틸-1-[1-(2-메틸-벤질)-4-페닐-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-2-플루오로-프로필)-N-{(R)-2,2-디메틸-1-[1-(3-메틸-벤질)-4-페닐-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-2-플루오로-프로필)-N-{(R)-2,2-디메틸-1-[1-(3-메틸-벤질)-4-페닐-1H-이미다졸-2-일]-프로필}-2-메톡시-아세트아미드;N-(3-아미노-프로필)-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-아세트아미드;N-((S)-3-아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((S)-3-아미노-2-플루오로-프로필)-[(R)-1-(1-벤질-1H-이미다졸-2-일)-2,3-디메틸-프로필]-아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-2-플루오로-프로필)-N-[(R)-1-(1-벤질-4-티아졸-2-일-1H-이미다졸-2-일)-2,3-디메틸-프로필]-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-[2-(1-아미노-시클로프로필)-에틸]-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 [2-(1-아미노-시클로프로필)-에틸]-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-아미드;[2-(1-아미노-시클로프로필)-에틸]-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2,2-디메틸-프로필]-카르밤산 메틸 에스테르;N-[2-(1-아미노-시클로프로필)-에틸]-N{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 [2-(1-아미노-시클로프로필)-에틸]-{(R)-1-[1-벤질-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;N-(3-아미노-프로필)-N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-((R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-(3-아미노-프로필)-N-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-1-메틸-에틸}-2-메톡시-아세트아미드;N-(3-아미노-프로필)-N-((R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산((S)-3-아미노-4-플루오로-부틸)-((R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-((R)-1-[1-벤질-5-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-메톡시-부틸)-N-{(R)-1-[4-(3-클로로-페닐)-1-(3-플루오로- 벤질)-1H-이미다졸-2-일]-2-메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-벤질-4-(3-클로로-페닐)-1H-이미다졸-2-일]-2-메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[4-(3-클로로-페닐)-1-(3-플루오로벤질)-1H-이미다졸-2-일]-2-메틸-프로필}-2-메톡시-아세트아미드;N-(3-아미노-4-플루오로-부틸)-N-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-1-메틸-에틸}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로- 페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((S)-3-아미노-2-플루오로-프로필)-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;(R)-9-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-6-옥사-2,9-디아자-스피로[4.5]데칸-8-온;(3-아미노-4-플루오로-부틸)-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페 닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-카르밤산 메틸 에스테르;N-((S)-3-아미노-4-플루오로-부틸)-N-((R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-포름아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-포름아미드;N-(3-디메틸아미노-프로필)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-부틸)-N-((R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)- H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-부틸)-N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(6-아미노-피리딘-2-일메틸)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3-아미노-4-플루오로-부틸)-N-{(R)-1-[1-(6-아미노-피리딘-2-일메틸)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-((R)-1-[4-(3-플루오로-페닐)-1-(3-히드록시-벤질)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((R)-3 -아미노-4-플루오로-부틸)-N-{(R)-1-[4-(3-플루오로-페닐)-1-(3-히 드록시- 벤질)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-[(R)-1-(1-벤질-4-티아졸-2-일-1H-이미다졸-2-일)-2,2-디메틸-프로필]-2-메톡시-아세트아미드;N-{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-N-(3-메틸아미노-프로필)-아세트아미드;N-(3-아미노-프로필)-N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아세트아미드;(3-아미노-프로필)-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-카르밤산 메틸 에스테르;(S)-테트라히드로-푸란-2-카르복시산 (3-아미노-프로필)-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;3-[{(R)-1-[1-(3-플루오로-벤질)-4-(3-플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-(2-메톡시-아세틸)-아미노]-프로피온아미드;N-(3-아미노-프로필)-N-((R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메탄설포닐-아세트아미드;N-((S)-3-아미노-4-메톡시-부틸)-N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;(S)-테트라히드로-푸란-2-카르복시산 ((S)-3-아미노-4-플루오로-부틸)-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필아민;(S)-테트라히드로-푸란-2-카르복시산 ((S)-3-아미노-4-메톡시-부틸)-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아미드;N-((R)-3-아미노-부틸)-N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-((S)-3-아미노-부틸)-N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-아세트아미드;N-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-2-메톡시-N-(H-[1 ,2,3]트리아졸-4-일메틸)-아세트아미드;N-((S)-3-아미노-4-플루오로-부틸)-N-(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-아세트아미드;((S)-3-아미노-4-플루오로-부틸)-{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-카르밤산 메틸 에스테르;{(R)-1-[1-벤질-4-(2,5-디플루오로-페닐)-1H-이미다졸-2-일]-2,2-디메틸-프로필}-카르밤산 tert-부틸 에스테르;N-[(1R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-4-메틸벤자미드;N-(3-아미노프로필)-N-[(R)-1-(1-벤질-4-페닐-1H-이미다졸-2-일)-2-메틸프로필]-2-(4-플루오로페녹시)아세트아미드.
- 치료적 유효량의 제 1 항의 화합물 및 약학적으로 허용가능한 담체를 포함하는 암 치료용 약학적 조성물.
- 삭제
- 제 23 항에 있어서, 이리노테칸, 토포테칸, 젬시타빈, 이마티니브, 트라스츄마브, 5-플루오로우라실, 류코보린, 카르보플라틴, 시스플라틴, 도세탁셀, 파클리탁셀, 테자시타빈, 시클로포스파미드, 빈카 알칼로이드, 안트라사이클린, 리튜시마브, 및 트라스츄마브로 이루어진 군으로부터 선택되는 적어도 하나의 암 치료용 첨가제를 더 포함하는 것을 특징으로 하는 조성물.
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US58092704P | 2004-06-18 | 2004-06-18 | |
| US60/580,927 | 2004-06-18 | ||
| PCT/US2005/022062 WO2006002236A1 (en) | 2004-06-18 | 2005-06-20 | N- (1- (1-benzyl -4-phenyl-1h-imidazol-2-yl) -2,2-dymethylpropyl) benzamide derivatives and related compounds as kinesin spindle protein (ksp) inhibitors for the treatment of cancer |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20070106961A KR20070106961A (ko) | 2007-11-06 |
| KR101170925B1 true KR101170925B1 (ko) | 2012-08-07 |
Family
ID=34980173
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020077000979A Expired - Fee Related KR101170925B1 (ko) | 2004-06-18 | 2005-06-20 | 암 치료용 키네신 방추 단백질 (ksp) 억제제로서의n-(1-(1-벤질-4-페닐-1h-이미다졸-2-일)-2,2-디메틸프로필)벤자미드 유도체 및 관련 화합물 |
Country Status (21)
| Country | Link |
|---|---|
| US (3) | US7576221B2 (ko) |
| EP (1) | EP1765789B1 (ko) |
| JP (2) | JP4836280B2 (ko) |
| KR (1) | KR101170925B1 (ko) |
| CN (1) | CN1993331A (ko) |
| AU (1) | AU2005258135B2 (ko) |
| BR (1) | BRPI0510929A (ko) |
| CA (1) | CA2571002A1 (ko) |
| EC (1) | ECSP077183A (ko) |
| ES (1) | ES2409345T3 (ko) |
| IL (1) | IL180147A0 (ko) |
| MA (1) | MA28739B1 (ko) |
| MX (1) | MXPA06014909A (ko) |
| NO (1) | NO20070343L (ko) |
| NZ (1) | NZ552510A (ko) |
| RU (1) | RU2427572C2 (ko) |
| SG (1) | SG153851A1 (ko) |
| TN (1) | TNSN06419A1 (ko) |
| TW (1) | TW200612958A (ko) |
| WO (1) | WO2006002236A1 (ko) |
| ZA (1) | ZA200700481B (ko) |
Families Citing this family (32)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101170925B1 (ko) * | 2004-06-18 | 2012-08-07 | 노바티스 백신즈 앤드 다이아그노스틱스 인코포레이티드 | 암 치료용 키네신 방추 단백질 (ksp) 억제제로서의n-(1-(1-벤질-4-페닐-1h-이미다졸-2-일)-2,2-디메틸프로필)벤자미드 유도체 및 관련 화합물 |
| UY29070A1 (es) | 2004-08-18 | 2006-03-31 | Astrazeneca Ab | Enantiómeros de heterocíclicos fusionados y sus usos |
| WO2007011647A2 (en) * | 2005-07-15 | 2007-01-25 | Kalypsys, Inc. | Inhibitors of mitotic kinesin ksp |
| TW200800951A (en) * | 2005-08-09 | 2008-01-01 | Novartis Ag | Substituted imidazole compounds as KSP inhibitors |
| US8119655B2 (en) | 2005-10-07 | 2012-02-21 | Takeda Pharmaceutical Company Limited | Kinase inhibitors |
| SI2010528T1 (en) | 2006-04-19 | 2018-02-28 | Novartis Ag | 6-O-substituted benzoxazole and benzothiazole compounds and procedures for inhibiting CSF-1R signaling |
| TWI386405B (zh) * | 2006-09-05 | 2013-02-21 | 咪唑衍生物 | |
| EA200970361A1 (ru) | 2006-10-09 | 2010-02-26 | Такеда Фармасьютикал Компани Лимитед | Ингибиторы киназы |
| MX2009005071A (es) | 2006-11-13 | 2009-05-25 | Novartis Ag | Compuestos de pirazol y triazol sustituidos como inhibidores de ksp. |
| GEP20125415B (en) * | 2007-01-05 | 2012-03-12 | Novartis Ag | Imidazole derivatives as kinesin spindle protein inhibitors (eg-5) |
| US8252832B2 (en) | 2007-12-14 | 2012-08-28 | Novartis Ag | Kinesin inhibitors as cancer therapeutics |
| WO2010084186A1 (en) | 2009-01-26 | 2010-07-29 | Novartis Ag | Salts and polymorphs of a kinesin inhibitor compound |
| US8748626B2 (en) | 2010-04-15 | 2014-06-10 | Novartis Ag | Oxazole and thiazole compounds as KSP inhibitors |
| EP2558450A1 (en) * | 2010-04-15 | 2013-02-20 | Novartis AG | Triazole compounds as ksp inhibitors |
| DE102010025663A1 (de) | 2010-06-30 | 2012-01-05 | Karl-Heinz Glüsenkamp | Neue beta-Aminoaldehyd-Derivate, Verfahren zu ihrer Herstellung und ihre chemische Verwendung als reaktive Intermediate |
| US8809372B2 (en) | 2011-09-30 | 2014-08-19 | Asana Biosciences, Llc | Pyridine derivatives |
| US9498540B2 (en) | 2013-03-15 | 2016-11-22 | Novartis Ag | Cell proliferation inhibitors and conjugates thereof |
| CN105451773A (zh) * | 2013-03-15 | 2016-03-30 | 诺华股份有限公司 | 细胞增殖抑制剂及其缀合物 |
| KR102329024B1 (ko) * | 2013-12-23 | 2021-11-19 | 바이엘 파마 악티엔게젤샤프트 | 키네신 스핀들 단백질(ksp)과의 항체 약물 접합체 (adcs) |
| US10786578B2 (en) | 2014-08-05 | 2020-09-29 | Novartis Ag | CKIT antibody drug conjugates |
| JP6743015B2 (ja) * | 2014-12-15 | 2020-08-19 | バイエル ファーマ アクチエンゲゼルシャフト | Ksp阻害剤の脱グリコシル化抗tweakr抗体との抗体薬物複合体(adc類) |
| IS2977B (is) | 2015-02-23 | 2017-07-15 | Actavis Group Ptc Ehf. | Aðferð til framleiðslu á milliefnum sem eru nytsamleg við nýsmíði á elúxadólíni |
| WO2016207089A1 (de) | 2015-06-22 | 2016-12-29 | Bayer Pharma Aktiengesellschaft | Binder-wirkstoff-konjugate (adcs) und binder-prodrug-konjugate (apdcs) mit enzymatisch spaltbaren gruppen |
| CA2990411A1 (en) | 2015-06-23 | 2016-12-29 | Bayer Pharma Aktiengesellschaft | Antibody drug conjugates of kinesin spindel protein (ksp) inhibitors with anti-b7h3-antibodies |
| JP2018525334A (ja) | 2015-06-23 | 2018-09-06 | バイエル ファーマ アクチエンゲゼルシャフト | キネシンスピンドルタンパク質(ksp)阻害剤の抗tweakr抗体との抗体薬物複合体 |
| US10973923B2 (en) * | 2015-06-23 | 2021-04-13 | Bayer Pharma Aktiengesellschaft | Site specific homogeneous with KSP inhibitors |
| WO2017216028A1 (en) | 2016-06-15 | 2017-12-21 | Bayer Pharma Aktiengesellschaft | Specific antibody-drug-conjugates (adcs) with ksp inhibitors and anti-cd123-antibodies |
| WO2018114578A1 (de) | 2016-12-21 | 2018-06-28 | Bayer Pharma Aktiengesellschaft | Binder-wirkstoff-konjugate (adcs) mit enzymatisch spaltbaren gruppen |
| CA3047522A1 (en) | 2016-12-21 | 2018-06-28 | Bayer Pharma Aktiengesellschaft | Specific antibody drug conjugates (adcs) having ksp inhibitors |
| WO2019243159A1 (de) | 2018-06-18 | 2019-12-26 | Bayer Aktiengesellschaft | Gegen cxcr5 gerichtete binder-wirkstoff-konjugate mit enzymatisch spaltbaren linkern und verbessertem wirkungsprofil |
| KR102875569B1 (ko) * | 2018-12-20 | 2025-10-23 | 암젠 인크 | Kif18a 억제제 |
| CA3230123A1 (en) | 2021-08-26 | 2023-03-02 | Derek A. Cogan | Spiro indoline inhibitors of kif18a |
Family Cites Families (65)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE2550959C3 (de) * | 1975-11-13 | 1980-12-04 | Hoechst Ag, 6000 Frankfurt | Tetrazolyl-imidazole und Tetrazolyl--benzimidazole, Verfahren zu deren Herstellung und diese enthaltende Arzneimittel |
| JP2001505585A (ja) | 1996-12-16 | 2001-04-24 | 藤沢薬品工業株式会社 | 新規アミド化合物およびそれらの一酸化窒素シンターゼ阻害剤としての用途 |
| US6117940A (en) | 1997-10-17 | 2000-09-12 | Mjalli; Adnan M. M. | Amino-ketone solid support templates |
| TR200100300T2 (tr) * | 1998-08-04 | 2001-07-23 | Astrazeneca Ab | Sitokinlerin üretiminde engelleyici olarak kullanışlı amid türevleri |
| JP2002525358A (ja) * | 1998-09-25 | 2002-08-13 | アストラゼネカ アクチボラグ | ベンズアミド誘導体及びサイトカイン阻害剤としてのその使用 |
| CA2369549A1 (en) | 1999-04-02 | 2000-10-12 | Robert W. Desimone | Aryl and heteroaryl fused aminoalkyl-imidazole derivatives and their use as antidiabetics |
| TWI292316B (en) * | 1999-10-11 | 2008-01-11 | Sod Conseils Rech Applic | Pharmaceutical composition of thiazole derivatives intended to inhibit mao and/or lipidic peroxidation and/or to act as modulators of sodium channels and the use thereof |
| US20040132788A1 (en) | 1999-10-11 | 2004-07-08 | Chabrier De Lassauniere Pierre-Etienne | Derivatives of heterocycles with 5 members, their preparation and their use as medicaments |
| US7291641B2 (en) * | 1999-10-11 | 2007-11-06 | Societe De Conseils De Recherches Et D'applications Scientifiques (S.C.R.A.S.) | Derivatives of heterocycles with 5 members, their preparation and their use as medicaments |
| US6545004B1 (en) | 1999-10-27 | 2003-04-08 | Cytokinetics, Inc. | Methods and compositions utilizing quinazolinones |
| NZ518480A (en) | 1999-10-27 | 2004-02-27 | Cytokinetics Inc | Methods and compositions utilizing quinazolinones |
| US6620882B1 (en) | 2000-05-02 | 2003-09-16 | Advanced Syntech, Llc | Solid support template for preparation of highly functionalized heterocycle compounds |
| AU2001264872A1 (en) * | 2000-06-05 | 2001-12-17 | Ortho-Mcneil Pharmaceutical, Inc. | Method for synthesis of substituted azole libraries |
| US6683191B2 (en) * | 2000-06-05 | 2004-01-27 | Ortho-Mcneil Pharmaceuticals, Inc. | Method for synthesis of substituted azole libraries |
| ES2282275T3 (es) * | 2000-08-01 | 2007-10-16 | Societe De Conseils De Recherches Et D'applications Scientifiques (S.C.R.A.S.) | Derivados de imidazolilo. |
| US20030055037A1 (en) | 2000-10-06 | 2003-03-20 | Delombaert Stephane | Benzimidazole and indole derivatives as CRF receptor modulators |
| JP2002190437A (ja) | 2000-12-20 | 2002-07-05 | Advantest Corp | 電子ビーム露光装置及び校正方法 |
| US20040132830A1 (en) | 2001-01-19 | 2004-07-08 | Finer Jeffrey T | Triphenylmethane kinesin inhibitors |
| WO2002057244A1 (en) | 2001-01-19 | 2002-07-25 | Cytokinetics, Inc. | Phenothiazine kinesin inhibitors |
| US6610723B2 (en) * | 2001-01-29 | 2003-08-26 | Hoffmann-La Roche Inc. | Imidazole derivatives |
| US7049303B2 (en) | 2001-11-07 | 2006-05-23 | Medical Research Council | Inhibition of viruses |
| JP2005511581A (ja) | 2001-11-07 | 2005-04-28 | メルク エンド カムパニー インコーポレーテッド | 有糸分裂キネシン阻害剤 |
| WO2003043995A1 (en) | 2001-11-20 | 2003-05-30 | Cytokinetics, Inc. | Process for the racemization of chiral quinazolinones |
| DE60232994D1 (de) | 2001-12-06 | 2009-08-27 | Merck & Co Inc | Inhibitoren von mitotischem kinesin |
| EP1463733B1 (en) | 2001-12-06 | 2007-09-05 | Merck & Co., Inc. | Mitotic kinesin inhibitors |
| ATE424388T1 (de) | 2001-12-06 | 2009-03-15 | Merck & Co Inc | Mitotische kinesinhemmer |
| US7244723B2 (en) | 2001-12-06 | 2007-07-17 | Merck & Co., Inc. | Substituted furopyrimidinones as a mitotic kinesin inhibitors |
| ATE447577T1 (de) * | 2001-12-06 | 2009-11-15 | Merck & Co Inc | Mitotische kinesin-hemmer |
| EP1465888A2 (en) | 2002-01-10 | 2004-10-13 | Neurogen Corporation | Melanin concentrating hormone receptor ligands: substituted benzoimidazole analogues |
| WO2003105855A1 (en) | 2002-01-11 | 2003-12-24 | Merck & Co., Inc. | Mitotic kinesin inhibitors |
| JP2005529076A (ja) | 2002-02-15 | 2005-09-29 | サイトキネティクス・インコーポレーテッド | キナゾリノンの合成 |
| ATE448207T1 (de) | 2002-03-08 | 2009-11-15 | Merck & Co Inc | Mitotische kinesin-hemmer |
| DE10211770A1 (de) | 2002-03-14 | 2003-10-02 | Boehringer Ingelheim Pharma | Neue substituierte Piperidine, diese Verbindungen enthaltende Arzneimittel und Verfahren zu ihrer Herstellung |
| US7026312B2 (en) | 2002-03-14 | 2006-04-11 | Boehringer Ingelheim Pharma Gmbh & Co. Kg | Substituted piperidines, pharmaceutical compositions containing these compounds, their use and processes for the preparation thereof |
| AU2003245259A1 (en) * | 2002-05-02 | 2003-11-17 | Neurogen Corporation | Substituted imidazole derivatives: gabaa receptor ligands |
| AU2003301864A1 (en) * | 2002-05-08 | 2004-06-07 | Neurogen Corporation | Substituted imidazolylmethyl pyridine and pyrazine derivatives and their use as gabaa receptor ligands |
| AU2003223786A1 (en) * | 2002-05-09 | 2003-11-11 | Cytokinetics, Inc. | Compounds, methods and compositions |
| WO2003097053A1 (en) | 2002-05-09 | 2003-11-27 | Cytokinetics, Inc. | Compounds, compositions, and methods |
| EP1507534A4 (en) | 2002-05-10 | 2006-11-08 | Cytokinetics Inc | COMPOUNDS, COMPOSITIONS AND METHODS |
| ATE333455T1 (de) * | 2002-05-17 | 2006-08-15 | Neurogen Corp | Substituierte, ringkondensierte imidazolderivate: gabaa-rezeptorliganden |
| AU2003265242A1 (en) | 2002-05-23 | 2003-12-22 | Cytokinetics, Inc. | Compounds, compositions, and methods |
| CA2483627A1 (en) | 2002-05-23 | 2003-12-04 | Merck & Co., Inc. | Mitotic kinesin inhibitors |
| JP4463679B2 (ja) | 2002-06-14 | 2010-05-19 | メルク エンド カムパニー インコーポレーテッド | 有糸分裂キネシン阻害剤 |
| WO2004001489A1 (ja) | 2002-06-19 | 2003-12-31 | Mitsubishi Denki Kabushiki Kaisha | 光変調器 |
| US20060134767A1 (en) | 2002-07-08 | 2006-06-22 | Buser-Doepner Carolyn A | Mitotic kinesin binding site |
| RU2233736C2 (ru) | 2002-07-11 | 2004-08-10 | Раховский Вадим Израилович | Нанометрическое позиционирующее устройство |
| WO2004006865A2 (en) | 2002-07-17 | 2004-01-22 | Cytokinetics, Inc. | Compounds, compositions, and methods |
| AU2003256805A1 (en) | 2002-07-23 | 2004-02-09 | Cytokinetics, Inc. | Compounds compositions and methods |
| KR100492252B1 (ko) | 2002-08-09 | 2005-05-30 | 한국화학연구원 | 이미다졸을 포함하는 이차아민으로 치환된 벤조피란유도체 및 그의 제조방법 |
| JP2005536553A (ja) | 2002-08-21 | 2005-12-02 | サイトキネティクス・インコーポレーテッド | 化合物、組成物および方法 |
| JP2005539062A (ja) | 2002-09-13 | 2005-12-22 | サイトキネティクス・インコーポレーテッド | 化合物、組成物および方法 |
| JP4547271B2 (ja) | 2002-09-18 | 2010-09-22 | ファイザー・プロダクツ・インク | トランスフォーミング増殖因子(tgf)阻害剤としての新規イミダゾール化合物 |
| US7557115B2 (en) * | 2002-09-30 | 2009-07-07 | Cytokinetics, Inc. | Compounds, compositions, and methods |
| CN100579579C (zh) * | 2002-10-01 | 2010-01-13 | 诺华疫苗和诊断公司 | 抗癌及抗感染性疾病组合物及其使用方法 |
| MXPA05003830A (es) * | 2002-10-11 | 2005-06-23 | Cytokinetics Inc | Compuestos, composiciones y metodos. |
| WO2004041808A1 (en) * | 2002-11-08 | 2004-05-21 | Neurogen Corporation | 4-imidazol-1-ylmethyl-pyrimidine derivatives_as ligands for gabaa receptors |
| US20040186151A1 (en) | 2003-02-12 | 2004-09-23 | Mjalli Adnan M.M. | Substituted azole derivatives as therapeutic agents |
| US7662581B1 (en) * | 2003-12-18 | 2010-02-16 | Novartis Vaccines And Diagnostics, Inc. | Eg5 co-crystals |
| JP2007517905A (ja) | 2004-01-16 | 2007-07-05 | ワイス | アゾールを含有するベータアミロイド産生のヘテロ環式スルホンアミド阻害剤 |
| AU2005214349B2 (en) * | 2004-02-12 | 2011-11-03 | Transtech Pharma, Inc. | Substituted azole derivatives, compositions, and methods of use |
| PT1732926E (pt) * | 2004-04-06 | 2009-04-03 | Novartis Vaccines & Diagnostic | Inibidores de cinesinas mitóticas |
| US20080132549A1 (en) | 2004-04-14 | 2008-06-05 | Pfizer Inc. | Sulphur-Linked Imidazone Compounds for the Treatment of Hiv/Aids |
| TW200540155A (en) | 2004-04-28 | 2005-12-16 | Takeda Pharmaceutical | Fused quinoline derivative and use thereof |
| US7618981B2 (en) | 2004-05-06 | 2009-11-17 | Cytokinetics, Inc. | Imidazopyridinyl-benzamide anti-cancer agents |
| KR101170925B1 (ko) * | 2004-06-18 | 2012-08-07 | 노바티스 백신즈 앤드 다이아그노스틱스 인코포레이티드 | 암 치료용 키네신 방추 단백질 (ksp) 억제제로서의n-(1-(1-벤질-4-페닐-1h-이미다졸-2-일)-2,2-디메틸프로필)벤자미드 유도체 및 관련 화합물 |
-
2005
- 2005-06-20 KR KR1020077000979A patent/KR101170925B1/ko not_active Expired - Fee Related
- 2005-06-20 EP EP05760871A patent/EP1765789B1/en not_active Expired - Lifetime
- 2005-06-20 CN CNA2005800262037A patent/CN1993331A/zh active Pending
- 2005-06-20 SG SG200904294-6A patent/SG153851A1/en unknown
- 2005-06-20 MX MXPA06014909A patent/MXPA06014909A/es active IP Right Grant
- 2005-06-20 TW TW094120475A patent/TW200612958A/zh unknown
- 2005-06-20 RU RU2007101612/04A patent/RU2427572C2/ru not_active IP Right Cessation
- 2005-06-20 JP JP2007516842A patent/JP4836280B2/ja not_active Expired - Fee Related
- 2005-06-20 NZ NZ552510A patent/NZ552510A/en not_active IP Right Cessation
- 2005-06-20 AU AU2005258135A patent/AU2005258135B2/en not_active Ceased
- 2005-06-20 US US11/158,574 patent/US7576221B2/en not_active Expired - Fee Related
- 2005-06-20 WO PCT/US2005/022062 patent/WO2006002236A1/en not_active Ceased
- 2005-06-20 BR BRPI0510929-9A patent/BRPI0510929A/pt not_active IP Right Cessation
- 2005-06-20 ES ES05760871T patent/ES2409345T3/es not_active Expired - Lifetime
- 2005-06-20 CA CA002571002A patent/CA2571002A1/en not_active Abandoned
-
2006
- 2006-12-18 IL IL180147A patent/IL180147A0/en unknown
- 2006-12-18 TN TNP2006000419A patent/TNSN06419A1/en unknown
-
2007
- 2007-01-16 ZA ZA200700481A patent/ZA200700481B/xx unknown
- 2007-01-17 MA MA29617A patent/MA28739B1/fr unknown
- 2007-01-18 NO NO20070343A patent/NO20070343L/no not_active Application Discontinuation
- 2007-01-18 EC EC2007007183A patent/ECSP077183A/es unknown
-
2009
- 2009-06-05 US US12/455,798 patent/US8318791B2/en not_active Expired - Fee Related
-
2011
- 2011-08-18 JP JP2011179247A patent/JP2011256197A/ja not_active Withdrawn
-
2012
- 2012-09-13 US US13/613,977 patent/US8735599B2/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| TNSN06419A1 (en) | 2008-02-22 |
| IL180147A0 (en) | 2007-06-03 |
| JP2011256197A (ja) | 2011-12-22 |
| AU2005258135A1 (en) | 2006-01-05 |
| CA2571002A1 (en) | 2006-01-05 |
| ECSP077183A (es) | 2007-02-28 |
| US20130017196A1 (en) | 2013-01-17 |
| US7576221B2 (en) | 2009-08-18 |
| EP1765789B1 (en) | 2013-02-27 |
| WO2006002236A1 (en) | 2006-01-05 |
| NO20070343L (no) | 2007-03-05 |
| US8735599B2 (en) | 2014-05-27 |
| NZ552510A (en) | 2010-12-24 |
| TW200612958A (en) | 2006-05-01 |
| MA28739B1 (fr) | 2007-07-02 |
| ES2409345T3 (es) | 2013-06-26 |
| EP1765789A1 (en) | 2007-03-28 |
| BRPI0510929A (pt) | 2007-07-17 |
| AU2005258135B2 (en) | 2011-09-22 |
| RU2007101612A (ru) | 2008-08-10 |
| JP4836280B2 (ja) | 2011-12-14 |
| MXPA06014909A (es) | 2007-02-28 |
| US8318791B2 (en) | 2012-11-27 |
| US20090258016A1 (en) | 2009-10-15 |
| JP2008503501A (ja) | 2008-02-07 |
| SG153851A1 (en) | 2009-07-29 |
| US20060009472A1 (en) | 2006-01-12 |
| KR20070106961A (ko) | 2007-11-06 |
| RU2427572C2 (ru) | 2011-08-27 |
| ZA200700481B (en) | 2008-10-29 |
| CN1993331A (zh) | 2007-07-04 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101170925B1 (ko) | 암 치료용 키네신 방추 단백질 (ksp) 억제제로서의n-(1-(1-벤질-4-페닐-1h-이미다졸-2-일)-2,2-디메틸프로필)벤자미드 유도체 및 관련 화합물 | |
| AU2008205169B2 (en) | Imidazole derivatives as kinesin spindle protein inhibitors (Eg-5) | |
| AU2007323998B2 (en) | Substituted pyrazole and triazole compounds as KSP inhibitors | |
| JP4975745B2 (ja) | Kspインヒビターとしての置換イミダゾール化合物 | |
| HK1111995B (en) | Substituted imidazole compounds as ksp inhibitors |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| A201 | Request for examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| R17-X000 | Change to representative recorded |
St.27 status event code: A-3-3-R10-R17-oth-X000 |
|
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| LAPS | Lapse due to unpaid annual fee | ||
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20150731 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20150731 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |