JP6638192B2 - Aluminum alloy processing material and method of manufacturing the same - Google Patents
Aluminum alloy processing material and method of manufacturing the same Download PDFInfo
- Publication number
- JP6638192B2 JP6638192B2 JP2015031364A JP2015031364A JP6638192B2 JP 6638192 B2 JP6638192 B2 JP 6638192B2 JP 2015031364 A JP2015031364 A JP 2015031364A JP 2015031364 A JP2015031364 A JP 2015031364A JP 6638192 B2 JP6638192 B2 JP 6638192B2
- Authority
- JP
- Japan
- Prior art keywords
- mass
- less
- aluminum alloy
- present
- treatment
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22C—ALLOYS
- C22C21/00—Alloys based on aluminium
- C22C21/10—Alloys based on aluminium with zinc as the next major constituent
-
- C—CHEMISTRY; METALLURGY
- C22—METALLURGY; FERROUS OR NON-FERROUS ALLOYS; TREATMENT OF ALLOYS OR NON-FERROUS METALS
- C22F—CHANGING THE PHYSICAL STRUCTURE OF NON-FERROUS METALS AND NON-FERROUS ALLOYS
- C22F1/00—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working
- C22F1/04—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working of aluminium or alloys based thereon
- C22F1/053—Changing the physical structure of non-ferrous metals or alloys by heat treatment or by hot or cold working of aluminium or alloys based thereon of alloys with zinc as the next major constituent
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Mechanical Engineering (AREA)
- Metallurgy (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Thermal Sciences (AREA)
- Crystallography & Structural Chemistry (AREA)
- Extrusion Of Metal (AREA)
Description
本発明は、アルミニウム合金加工材及びその製造方法に関する。 The present invention relates to a processed aluminum alloy material and a method for producing the same.
アルミニウム合金は、アルミニウムを主成分とする合金である。アルミニウム(Al)は比較的軽い金属であるが、純度の高いアルミニウムは非常に軟らかいため、Cu(銅)、Mn(マンガン)、Si(ケイ素)、Mg(マグネシウム)、Zn(亜鉛)、Ni(ニッケル)等の添加物によって合金にすることで、所望の強度、靱性、延性等の特性の向上が図られる。 The aluminum alloy is an alloy containing aluminum as a main component. Aluminum (Al) is a relatively light metal, but high-purity aluminum is very soft, so Cu (copper), Mn (manganese), Si (silicon), Mg (magnesium), Zn (zinc), Ni ( By alloying with an additive such as nickel), desired properties such as strength, toughness, and ductility can be improved.
アルミニウム合金の中でも、いわゆる7000番台のAl‐Zn‐Mg‐Cu系合金は強度の高い合金であり、アルミニウム合金の軽さと強度を生かして、種々の分野に利用されている。 Among aluminum alloys, the so-called 7000-series Al-Zn-Mg-Cu alloy is a high strength alloy, and is utilized in various fields by making use of the lightness and strength of the aluminum alloy.
特許文献1には、靱性と静的機械的特性とをバランスよく得ることを目的としたアルミニウム合金から構成される航空機用の製品が開示されている。この文献の技術では、靱性、静的機械的特性、耐食性、破断伸びをバランスよく得ることを目的として、添加元素の濃度等を調整することを開示している。特許文献1では、アルミニウム合金の添加元素について、Mg/Cu<2.4等の条件を満たすことで、靱性が向上することを開示している。 Patent Literature 1 discloses an aircraft product made of an aluminum alloy for the purpose of obtaining a good balance between toughness and static mechanical properties. The technique disclosed in this document discloses adjusting the concentration of an additive element and the like for the purpose of obtaining a good balance of toughness, static mechanical properties, corrosion resistance, and elongation at break. Patent Literature 1 discloses that toughness is improved by satisfying a condition such as Mg / Cu <2.4 as an additive element of an aluminum alloy.
しかし一方で、アルミニウム合金が応用される分野によっては、より高い強度の製品が求められることがある。例えば、軽量かつ耐衝撃性の高い製品が求められることがある。
そこで、本発明は、高強度のアルミニウム合金加工材及びその製造方法を提供することを主な目的とする。
However, depending on the field to which the aluminum alloy is applied, a product having higher strength may be required. For example, a product that is lightweight and has high impact resistance may be required.
Therefore, an object of the present invention is to provide a high-strength aluminum alloy processed material and a method for manufacturing the same.
本発明によれば、Zn;9質量%以上11質量%以下、Mg;2.0質量%以上2.8質量%未満、Cu;0.8質量%を超えて1.2質量%以下、Zr;0.08質量%以上0.25質量%以下、Cr;0.02質量%以上0.30質量%以下を含有し、
残部がAlと不可避的不純物からなり、前記Mg,Cuの含有量は、Mg/Cu≧2.5の関係を満たし、Zr、Crの含有量の合計が、0.12質量%以上0.42質量%以下であり、塑性加工組織を有することを特徴とするアルミニウム合金加工材が提供される。
According to the present invention, Zn: 9% to 11% by mass, Mg: 2.0% to less than 2.8% by mass, Cu: more than 0.8% by mass to 1.2% by mass, Zr 0.08% by mass or less and 0.25% by mass or less; Cr; 0.02% by mass or more and 0.30% by mass or less;
The balance consists of Al and unavoidable impurities, the content of Mg and Cu satisfies the relationship of Mg / Cu ≧ 2.5, and the total content of Zr and Cr is 0.12% by mass or more and 0.42% or more. An aluminum alloy processed material, which is not more than mass% and has a plastic working structure.
本発明の一態様によれば、上記のアルミニウム合金加工材において、更にMn;0.05質量%以上0.32質量%以下を含有し、Zr、Cr、Mnの含有量の合計が、0.12質量%以上0.42質量%以下とすることを特徴とする。 According to one embodiment of the present invention, the processed aluminum alloy material further contains Mn; 0.05 mass% or more and 0.32 mass% or less, and the total content of Zr, Cr, and Mn is 0. It is not less than 12% by mass and not more than 0.42% by mass.
また本発明によれば、Zn;9質量%以上11質量%以下、Mg;2.00以上2.80質量%未満、Cu;0.8質量%を超えて1.2質量%以下、Zr;0.08質量%以上0.25質量%以下、Cr;0.02質量%以上0.30質量%以下を含有し、残部がAlと不可避的不純物からなり、前記Mg,Cuの含有量は、Mg/Cu≧2.5の関係を満たし、Zr、Crの含有量の合計が、0.12質量%以上0.42質量%以下であり、塑性加工、溶体化処理、焼き入れ処理及び時効処理を順次施すことを特徴とするアルミニウム合金加工材の製造方法が提供される。 Further, according to the present invention, Zn: 9 mass% to 11 mass%, Mg: 2.00 to less than 2.80 mass%, Cu: more than 0.8 mass% to 1.2 mass%, Zr; 0.08% by mass or more and 0.25% by mass or less, Cr; containing 0.02% by mass or more and 0.30% by mass or less, and the balance consists of Al and inevitable impurities. The relationship of Mg / Cu ≧ 2.5 is satisfied, and the total content of Zr and Cr is 0.12% by mass or more and 0.42% by mass or less, plastic working, solution treatment, quenching treatment and aging treatment Are provided sequentially, and a method for producing a processed aluminum alloy material is provided.
本発明の一態様によれば、上記のアルミニウム合金加工材の製造方法において、更にMn;0.05質量%以上0.32質量%以下を含有し、Zr、Cr、Mnの含有量の合計が、0.12質量%以上0.42質量%以下とすることを特徴とする。 According to one embodiment of the present invention, the method for producing a processed aluminum alloy material further includes Mn; 0.05 mass% or more and 0.32 mass% or less, and the total content of Zr, Cr, and Mn is reduced. , 0.12% by mass or more and 0.42% by mass or less.
本発明の一態様によれば、上記のアルミニウム合金加工材の製造方法において、塑性加工として、押出比が5から100、鋳塊温度が300℃から450℃の熱間押出し加工、440℃から470℃に保持する溶体化処理、450℃から100℃の範囲を1000℃/分以上の冷却速度で冷却する焼き入れ処理、100℃から180℃で10時間から30時間の時効処理を順次施すことを特徴とする。 According to one aspect of the present invention, in the above-described method for manufacturing a processed aluminum alloy material, as the plastic working, an extrusion ratio is 5 to 100, and an ingot temperature is 300 to 450 ° C., and hot extrusion is performed. C., a quenching treatment for cooling the range of 450 ° C. to 100 ° C. at a cooling rate of 1000 ° C./min or more, and an aging treatment for 10 hours to 30 hours at 100 ° C. to 180 ° C. Features.
以下に、本発明の実施形態を説明する。 Hereinafter, embodiments of the present invention will be described.
<アルミニウム合金加工材>
本発明の一実施形態は、Zn;9質量%以上11質量%以下、Mg;2.0質量%以上2.8質量%未満、Cu;0.8質量%を超えて1.2質量%以下、Zr;0.08質量%以上0.25質量%以下、Cr;0.02質量%以上0.30質量%以下を含有し、残部がAlと不可避的不純物からなり、前記Mg,Cuの含有量は、Mg/Cu≧2.5の関係を満たし、Zr、Crの含有量の合計が、0.12質量%以上0.42質量%以下であり、塑性加工組織を有することを特徴とするアルミニウム合金加工材である。本実施形態のアルミニウム合金加工材は、高強度のアルミニウム合金加工材である。
<Aluminum alloy processed material>
One embodiment of the present invention is Zn: 9% by mass or more and 11% by mass or less, Mg: 2.0% by mass or more and less than 2.8% by mass, Cu: more than 0.8% by mass and 1.2% by mass or less. , Zr: 0.08% by mass or more and 0.25% by mass or less, Cr: 0.02% by mass or more and 0.30% by mass or less, with the balance being Al and inevitable impurities, containing Mg and Cu. The amount satisfies the relationship of Mg / Cu ≧ 2.5, the sum of the contents of Zr and Cr is 0.12% by mass or more and 0.42% by mass or less, and has a plastic working structure. Aluminum alloy processed material. The aluminum alloy processed material of the present embodiment is a high-strength aluminum alloy processed material.
(Zn、Mg、Cu)
本実施形態のアルミニウム合金加工材は、Zn(亜鉛)の含有量が、9質量%以上11質量%以下である。
(Zn, Mg, Cu)
The processed aluminum alloy material of the present embodiment has a Zn (zinc) content of 9% by mass or more and 11% by mass or less.
本実施形態のアルミニウム合金加工材は、Mg(マグネシウム)の含有量が、2.0質量%以上2.8質量%以下である。より好ましくは、Mgの含有量が、2.2質量%以上2.8質量%以下である。 The aluminum alloy processed material of the present embodiment has a Mg (magnesium) content of 2.0% by mass to 2.8% by mass. More preferably, the content of Mg is 2.2 mass% or more and 2.8 mass% or less.
本実施形態のアルミニウム合金加工材は、Cu(銅)の含有量が、0.8質量%を超えて1.2質量%以下である。 The aluminum alloy processed material of the present embodiment has a Cu (copper) content of more than 0.8% by mass and not more than 1.2% by mass.
上記の元素を含有するアルミニウム合金を、溶体化後に時効処理することで、ZnとMgがZn‐Mg系析出物(MgZn2等の化合物)を形成する。また、Mg、Cuは、Al‐Cu‐Mg系析出物(Al2CuMg等の化合物)を形成する。これらの析出物による析出強化がアルミニウム合金の強度に寄与する。 An aluminum alloy containing the above elements, by aging treatment after solution treatment, Zn and Mg to form Zn-Mg-based precipitates (compounds such as MgZn 2). Mg and Cu form Al-Cu-Mg-based precipitates (compounds such as Al 2 CuMg). Precipitation strengthening by these precipitates contributes to the strength of the aluminum alloy.
上記の元素の含有量が、Znが9質量%未満、Mgが2.0質量%未満、Cuが0.8質量%以下であると、下限範囲外では十分に析出強化できず、アルミニウム合金が所望の強度に満たない。
上記の元素の含有量が、Znが11質量%より多く、Mgが2.8質量%より多く、Cuが1.2質量%より多いと、鋳造時に形成された晶出物が粗大化し、均質化処理や溶体化処理で十分に固溶させることができず、熱処理後に残存する晶出物への応力集中で破断の起点となり、アルミニウム合金の伸びを低下させる。
If the content of the above elements is less than 9% by mass of Zn, less than 2.0% by mass of Mg, and 0.8% by mass or less of Cu, precipitation strengthening cannot be sufficiently performed outside the lower limit range, and the aluminum alloy has Less than desired strength.
When the content of the above elements is more than 11% by mass of Zn, more than 2.8% by mass of Mg, and more than 1.2% by mass of Cu, a crystallized substance formed at the time of casting becomes coarse and homogeneous. A solid solution cannot be sufficiently formed by the solution treatment or the solution treatment, and the concentration of stress on the crystallized material remaining after the heat treatment becomes a starting point of fracture, thereby reducing the elongation of the aluminum alloy.
なお、Zn、Mg、Cuは上記の通り、強度に寄与する元素であるが、Zn‐Mg系析出物による強化が、Al合金の強度を最も高めることが確認された。
ただし、合金成分については、鋳造性や添加量当たりの強度の寄与も考慮する必要がある。Znは密度が高く、過剰に添加すると比強度を低下させる。よって、Zn量は鋳造性及び比強度の観点から、11質量%以下とすることが好ましい。
As described above, Zn, Mg, and Cu are elements that contribute to the strength, but it has been confirmed that strengthening by a Zn—Mg-based precipitate increases the strength of the Al alloy most.
However, regarding the alloy components, it is necessary to consider the contribution of castability and strength per amount of addition. Zn has a high density, and when added excessively, it lowers the specific strength. Therefore, the amount of Zn is preferably 11% by mass or less from the viewpoint of castability and specific strength.
MgとCuの含有量は、3.2質量%≦Mg+Cu≦4.2質量%であることが好ましい。
Mg+Cuの含有量が3.2質量%未満であるとMg‐Cu系析出物の量が不十分である。Mg+Cuの含有量が4.2質量%を超えると粗大晶出物が形成され、溶体化処理で固溶できない晶出物が熱処理後に存在するため、応力集中による破断の起点となり、アルミニウム合金の伸びが低下する。
The content of Mg and Cu is preferably 3.2% by mass ≦ Mg + Cu ≦ 4.2% by mass.
When the content of Mg + Cu is less than 3.2% by mass, the amount of the Mg-Cu-based precipitate is insufficient. If the content of Mg + Cu exceeds 4.2% by mass, coarse crystals are formed, and crystals that cannot be dissolved in the solution treatment are present after the heat treatment. Decrease.
更に、Mg/Cuで表されるMgとCuの比は2.5以上である。より好ましくは、Mg/Cuが2.5以上3.5以下である。
主な強化相であるZn‐Mg析出相で強化し、さらにAl‐Cu‐Mg析出相で強化することで、強度の高いアルミニウム合金が得られる。また、Mg/Cuが2.5未満では、Al‐Cu‐Mg系析出物の形成にMgが消費され、より強度に有効に作用するZn‐Mg系析出物の析出量が相対的に小さくなる。
Further, the ratio of Mg to Cu represented by Mg / Cu is 2.5 or more. More preferably, Mg / Cu is 2.5 or more and 3.5 or less.
By strengthening with the Zn-Mg precipitation phase, which is the main strengthening phase, and further strengthening with the Al-Cu-Mg precipitation phase, an aluminum alloy with high strength can be obtained. If Mg / Cu is less than 2.5, Mg is consumed in forming Al-Cu-Mg-based precipitates, and the amount of Zn-Mg-based precipitates acting more effectively on the strength becomes relatively small. .
(Zr、Cr、Mn)
本実施形態のアルミニウム合金加工材は、Zr(ジルコニウム)の含有量が、0.08質量%から0.25質量%である。
本実施形態のアルミニウム合金加工材は、Cr(クロム)の含有量が、0.02質量%以上0.30質量%以下である。
本実施形態のアルミニウム合金加工材は、更にMn;0.05質量%以上0.32質量%以下を含有させても良い。Zr、Cr、Mnの含有量の合計は、0.12質量%以上0.42質量%以下である
(Zr, Cr, Mn)
The processed aluminum alloy material of the present embodiment has a Zr (zirconium) content of 0.08% by mass to 0.25% by mass.
The processed aluminum alloy material of the present embodiment has a Cr (chromium) content of 0.02% by mass or more and 0.30% by mass or less.
The processed aluminum alloy material of the present embodiment may further contain Mn; 0.05% by mass or more and 0.32% by mass or less. The total content of Zr, Cr, and Mn is 0.12% by mass or more and 0.42% by mass or less.
上記の元素を含有するアルミニウム合金では、均質化処理時に、Al‐Zr系の分散粒子、及び又はAl‐Cr系の分散粒子、Al‐Mn系の分散粒子が形成され、結晶粒界の移動を抑制し、再結晶化を抑制する、いわゆるピン止め効果が生じる。これにより、再結晶化を抑制し、塑性加工時に形成された加工組織を、溶体化処理後も維持させることで、アルミニウム合金の強度に寄与する。
Zr、Cr、Mnの含有量の合計は、0.42質量%より大きくなると焼入れ感受性が高まり、T6強度が低下する。特に、押出断面積が大きいものは冷却速度が遅くなるため、強度が得られなくなる。
In an aluminum alloy containing the above elements, during homogenization treatment, Al-Zr-based dispersed particles, and / or Al-Cr-based dispersed particles, and Al-Mn-based dispersed particles are formed, and the movement of crystal grain boundaries is suppressed. In other words, a so-called pinning effect is suppressed. Thereby, recrystallization is suppressed and the processed structure formed at the time of plastic working is maintained after the solution treatment, thereby contributing to the strength of the aluminum alloy.
If the total content of Zr, Cr, and Mn is more than 0.42% by mass, quenching sensitivity increases and T6 strength decreases. In particular, when the extrusion cross-sectional area is large, the cooling rate becomes slow, so that the strength cannot be obtained.
上記の元素の含有量が、Zrが、0.08質量%未満であり、Crが、0.02質量%未満であり、Mnが0.05質量%未満であると、ピン止め効果が十分に得られず、強度に寄与できない。上記の元素の含有量が、Zrが、0.20質量%より多く、Crが、0.30質量%より多いと、鋳造時に上記の粗大な晶出物が形成され、アルミニウム合金の伸びが低下する。
また、Al‐Cr系の分散粒子及びAl‐Mn系の分散粒子は耐応力腐食割れ性を高める効果があるが、Crが、0.02質量%未満、Mnが0.05質量%未満であると、この効果が十分に得られない。この効果はAl‐Cr系分散粒子及びAl‐Mn系の分散粒子が、水素原子を補足することに起因すると考えられる。
When the content of the above elements is such that Zr is less than 0.08% by mass, Cr is less than 0.02% by mass, and Mn is less than 0.05% by mass, the pinning effect is sufficient. It cannot be obtained and cannot contribute to strength. If the content of the above elements is such that Zr is more than 0.20% by mass and Cr is more than 0.30% by mass, the above coarse crystals are formed during casting, and the elongation of the aluminum alloy is reduced. I do.
Al-Cr-based dispersed particles and Al-Mn-based dispersed particles have an effect of enhancing stress corrosion cracking resistance, but Cr is less than 0.02% by mass and Mn is less than 0.05% by mass. This effect cannot be obtained sufficiently. This effect is considered to be caused by the Al-Cr-based dispersed particles and the Al-Mn-based dispersed particles capturing hydrogen atoms.
本実施形態のアルミニウム合金加工材は、上記の元素以外の残部がAlと不可避的不純物からなる。アルミニウム合金中には、アルミニウム地金などに由来する不可避不純物として、その他の元素が含まれる。不可避的不純物の含有量は、Siが0.15質量%以下、Feが0.20質量%以下、更に好ましくは双方ともに0.10質量%以下であれば、本発明の効果を妨げることがなく好ましい。
TiとBは、鋳塊の割れ防止のために鋳造組織の微細剤として添加しても良い。
The remainder of the aluminum alloy processed material of the present embodiment other than the above-mentioned elements consists of Al and inevitable impurities. The aluminum alloy contains other elements as inevitable impurities derived from aluminum ingots and the like. If the content of the unavoidable impurities is 0.15% by mass or less for Si and 0.20% by mass or less for Fe, and more preferably 0.10% by mass or less for both, the effect of the present invention is not hindered. preferable.
Ti and B may be added as fine agents in the cast structure to prevent cracking of the ingot.
<アルミニウム合金加工材の製造方法>
本発明の一実施形態は、Zn;9質量%以上11質量%以下、Mg;2.0質量%以上2.8質量%未満、Cu;0.8質量%を超えて1.2質量%以下、Zr;0.08質量%以上0.25質量%以下、Cr;0.02質量%以上0.30質量%以下を含有し、
残部がAlと不可避的不純物からなり、前記Mg,Cuの含有量は、Mg/Cu≧2.5の関係を満たし、Zr、Crの含有量の合計が、0.12質量%以上0.42質量%以下であり、塑性加工、溶体化処理、焼き入れ処理及び時効処理を順次施すことを特徴とするアルミニウム合金加工材の製造方法である。
<Production method of aluminum alloy processed material>
One embodiment of the present invention is Zn: 9% by mass or more and 11% by mass or less, Mg: 2.0% by mass or more and less than 2.8% by mass, Cu: more than 0.8% by mass and 1.2% by mass or less. , Zr; 0.08% by mass or more and 0.25% by mass or less; Cr; 0.02% by mass or more and 0.30% by mass or less;
The balance consists of Al and unavoidable impurities, the content of Mg and Cu satisfies the relationship of Mg / Cu ≧ 2.5, and the total content of Zr and Cr is 0.12% by mass or more and 0.42% or more. Mass% or less, and is a method for producing an aluminum alloy processed material, wherein plastic working, solution treatment, quenching, and aging are sequentially performed.
本実施形態のアルミニウム合金加工材の製造方法によれば、上記の組成であり、かつ塑性加工、溶体化処理、焼き入れ処理及び時効処理が順次施されることにより、高強度のアルミニウム合金加工材を製造することができる。 According to the method for manufacturing an aluminum alloy processed material of the present embodiment, a high-strength aluminum alloy processed material having the above-described composition and being subjected to plastic working, solution treatment, quenching, and aging sequentially. Can be manufactured.
(塑性加工)
塑性加工としては、圧延加工、鍛造、押出加工、引き抜き加工等が挙げられる。塑性加工は、加工組織形成のため、熱間加工であることが好ましい。最も好ましい塑性加工は、押出比が5から100であり、鋳塊温度が300℃から450℃の条件の熱間押出加工である。これは、加工組織を熱処理後まで残存させるために適した条件であるという理由からである。
また、押出加工の後に、所定の形状や寸法にするために、引き抜き加工等の塑性加工や切削加工を行っても良い。
(Plastic processing)
Examples of the plastic working include rolling, forging, extrusion, and drawing. The plastic working is preferably hot working in order to form a working structure. The most preferred plastic working is hot extrusion working at an extrusion ratio of 5 to 100 and an ingot temperature of 300 to 450 ° C. This is because the conditions are suitable for allowing the processed structure to remain until after the heat treatment.
Further, after the extrusion, plastic processing such as drawing or cutting may be performed in order to obtain a predetermined shape or size.
なお、塑性加工の前に、鋳造時に偏析した晶出物等を均質化するため、並びに、Al‐Zr系、Al‐Cr系及びAl‐Mn系の析出物を形成させるための均質化処理(HO処理)を施すのがより好ましい。この処理が塑性加工性及び加工組織の形成に影響を与えるためである。
保持条件を460℃(450℃から470℃)で24時間とすることで、Zn、Mg、Cuなどの溶出元素を均質化させ、後の熱処理で十分な強度を得ることができる。一方で、保持温度を高め過ぎると過度に溶ける可能性がある。また、昇温速度は、50℃/h以下がより好ましい。昇温速度が速いとAl‐Zr系、Al‐Cr系、Al‐Mn系析出物の粒子間隔が広くなり、溶体化処理時に再結晶化し易くなる。すなわち、溶体化処理後に加工組織を維持するために昇温速度を制御している。
Before the plastic working, a homogenization treatment for homogenizing the crystallized substances and the like segregated at the time of casting and for forming Al-Zr-based, Al-Cr-based and Al-Mn-based precipitates ( (HO treatment) is more preferable. This is because this treatment affects the plastic workability and the formation of the processed structure.
By maintaining the holding conditions at 460 ° C. (450 ° C. to 470 ° C.) for 24 hours, elution elements such as Zn, Mg, and Cu can be homogenized, and sufficient strength can be obtained by a subsequent heat treatment. On the other hand, if the holding temperature is too high, there is a possibility that the resin is excessively melted. Further, the heating rate is more preferably 50 ° C./h or less. If the heating rate is high, the particle spacing of the Al-Zr-based, Al-Cr-based, and Al-Mn-based precipitates is widened, and it is easy to recrystallize during the solution treatment. That is, the heating rate is controlled in order to maintain the processed structure after the solution treatment.
(溶体化処理)
塑性加工の後で、溶体化処理を行う。溶体化処理では、塑性加工材を、高温に保持することにより、晶出物を母相中に固溶させる。
溶体化処理時の保持温度は、約440℃〜約470℃の範囲であることが好ましい。保持温度が高すぎると局部溶融が発生し、塑性加工材の強度が低下する。一方で保持温度が低いと、Zn、Mg、Cuが十分に固溶できないため、後の時効処理で十分な析出強化が得られない。
(Solution treatment)
After the plastic working, a solution treatment is performed. In the solution treatment, the crystallized material is dissolved in the parent phase by maintaining the plastic work material at a high temperature.
The holding temperature during the solution treatment is preferably in the range of about 440C to about 470C. If the holding temperature is too high, local melting occurs and the strength of the plastically processed material decreases. On the other hand, if the holding temperature is low, Zn, Mg, and Cu cannot be sufficiently solid-dissolved, so that sufficient precipitation strengthening cannot be obtained by the subsequent aging treatment.
(焼き入れ処理)
溶体化処理の後で、焼き入れ処理を行う。溶体化処理した塑性加工材を常温まで急冷することにより、母相中に固溶した晶出物が析出するのを抑制し、過飽和状態を維持する。
焼き入れ処理は、例えば、水冷で室温まで速やかに冷却する。冷却速度は450℃から100℃の範囲を1000℃/分以上の速度で冷却し、その後室温で2日間自然時効することが好ましい。このような条件で焼き入れ処理を行うことにより、良好な過飽和固溶体とすることができる。
(Hardening process)
After the solution treatment, a quenching treatment is performed. By quenching the solution-processed plastic work material to room temperature, precipitation of crystallized solid solution in the mother phase is suppressed, and a supersaturated state is maintained.
In the quenching process, for example, water is used to quickly cool to room temperature. The cooling rate is preferably from 450 ° C. to 100 ° C. at a rate of 1000 ° C./min or more, followed by natural aging at room temperature for 2 days. By performing the quenching treatment under such conditions, a good supersaturated solid solution can be obtained.
(時効処理)
焼き入れ処理の後、時効処理を行う。時効処理として、焼き入れ後の塑性加工材を、約100℃〜約180℃に加熱保持することにより、母相中に過飽和状態であったZn、Mg、Cuを、Zn‐Mg系化合物及びAl‐Mg‐Cu系化合物として微細均一に析出させ、塑性加工材の強度を向上させる。
溶体化処理や焼き入れ処理を行っていないと母相中に過飽和に固溶しているZn、Mg、Cu量が不足し、時効処理の際に析出する化合物の量が減少し、強度の向上が不足する。
(Aging treatment)
After the quenching process, an aging process is performed. As an aging treatment, the super-saturated Zn, Mg, and Cu in the mother phase are removed by heating and holding the quenched plastic work material at about 100 ° C. to about 180 ° C. -Precipitating finely and uniformly as an Mg-Cu-based compound to improve the strength of the plastically processed material.
If the solution treatment or quenching treatment is not performed, the amount of Zn, Mg, and Cu that are supersaturated in the mother phase will be insufficient, and the amount of the compound that precipitates during the aging treatment will decrease, and the strength will improve. Run out.
最も高い強度を得るための時効処理条件(T6)は、例えば、100℃から180℃で10時間から30時間、より好ましくは、110℃から120℃で12時間から25時間である。その他の耐食性を重視する場合は、T73、T74,T76の熱処理を選択できる。 The aging treatment condition (T6) for obtaining the highest strength is, for example, 10 to 30 hours at 100 to 180 ° C, more preferably 12 to 25 hours at 110 to 120 ° C. When other corrosion resistance is emphasized, the heat treatment of T73, T74, and T76 can be selected.
本発明の一実施形態では、上記のアルミニウム合金加工材の製造方法において、塑性加工として、押出比が5から100、鋳塊温度が300℃から450℃の熱間押出し加工、440℃から470℃に保持する溶体化処理、450℃から100℃の範囲を1000℃/分以上の冷却速度で冷却する焼き入れ処理、100℃から180℃で10時間から30時間の時効処理を順次施す。 In one embodiment of the present invention, in the above-described method for producing a processed aluminum alloy material, as the plastic working, an extrusion ratio of 5 to 100 and an ingot temperature of 300 to 450 ° C. are hot extruded, and 440 to 470 ° C. , A quenching process of cooling a range of 450 ° C. to 100 ° C. at a cooling rate of 1000 ° C./min or more, and an aging process at 100 ° C. to 180 ° C. for 10 hours to 30 hours.
本発明の一実施形態は、Zn;9質量%以上11質量%以下、Mg;2.0質量%以上2.8質量%未満、Cu;0.8質量%を超えて1.2質量%以下、Zr;0.08質量%以上0.25質量%以下、Cr;0.02質量%以上0.30質量%以下を含有し、更にMn;0.05質量%以上0.32質量%以下を含有し、残部がAlと不可避的不純物からなり、前記Mg,Cuの含有量は、Mg/Cu≧2.5の関係を満たし、Zr、Cr、Mnの含有量の合計が、0.12質量%以上0.42質量%以下であり、塑性加工、溶体化処理、焼き入れ処理及び時効処理を順次施すことを特徴とするアルミニウム合金加工材の製造方法である。 One embodiment of the present invention is as follows: Zn: 9 mass% to 11 mass%, Mg: 2.0 mass% to less than 2.8 mass%, Cu; more than 0.8 mass% to 1.2 mass% or less. , Zr: 0.08% by mass to 0.25% by mass, Cr: 0.02% by mass to 0.30% by mass, and Mn: 0.05% by mass to 0.32% by mass. Contained, with the balance being Al and unavoidable impurities, the content of Mg and Cu satisfying the relationship Mg / Cu ≧ 2.5, and the total content of Zr, Cr and Mn is 0.12 mass%. % Or more and not more than 0.42% by mass, and is a method for producing an aluminum alloy processed material, wherein plastic working, solution treatment, quenching and aging are sequentially performed.
この製造方法では、Mn(マンガン)を0.05質量%以上0.32質量%以下含有することで、耐応力腐食割れ性を高める効果がある。MnもAl‐Cr系の分散粒子と同様の効果が得られるため、上述の特性をより向上させることができる。
上記の元素を含有するアルミニウム合金では、均質化処理時に、Al‐Zr系の分散粒子、および又はAl‐Cr系の分散粒子、Al−Mn系の分散粒子が形成され、結晶粒界の移動を抑制し、再結晶化を抑制する、いわゆるピン止め効果が生じる。これにより、再結晶化を抑制し、塑性加工時に形成された加工組織を、溶体化処理後も維持させることで、アルミニウム合金の強度に寄与する。Zr、Cr、Mnの含有量の合計は、0.42%より、大きくなると焼入れ感受性が高まり、T6強度が低下する。特に、押出断面積が大きいものは冷却速度が遅くなるため、強度が得られなくなる。
In this manufacturing method, by containing Mn (manganese) in an amount of 0.05% by mass or more and 0.32% by mass or less, there is an effect of enhancing stress corrosion cracking resistance. Since Mn also has the same effect as Al-Cr-based dispersed particles, the above-described characteristics can be further improved.
In the aluminum alloy containing the above elements, at the time of the homogenization treatment, Al-Zr-based dispersed particles, and / or Al-Cr-based dispersed particles, and Al-Mn-based dispersed particles are formed, and the movement of crystal grain boundaries is suppressed. In other words, a so-called pinning effect is suppressed. Thereby, recrystallization is suppressed and the processed structure formed at the time of plastic working is maintained after the solution treatment, thereby contributing to the strength of the aluminum alloy. When the total content of Zr, Cr, and Mn is more than 0.42%, the quenching sensitivity increases and the T6 strength decreases. In particular, when the extrusion cross-sectional area is large, the cooling rate becomes slow, so that the strength cannot be obtained.
また本発明の一実施形態では、上記のアルミニウム合金加工材において、塑性加工として、押出比が5から100、鋳塊温度が300℃から450℃の熱間押出し加工、440から470℃に保持する溶体化処理、450℃から100℃の範囲を1000℃/分以上の冷却速度で冷却する焼き入れ処理、100℃から180℃で10時間から30時間の時効処理が順次施されている。 In one embodiment of the present invention, in the above-mentioned aluminum alloy processed material, as the plastic working, the extrusion ratio is 5 to 100, and the ingot temperature is 300 to 450 ° C., and the hot extruding process is maintained at 440 to 470 ° C. A solution treatment, a quenching treatment for cooling a range of 450 ° C. to 100 ° C. at a cooling rate of 1000 ° C./min or more, and an aging treatment at 100 ° C. to 180 ° C. for 10 hours to 30 hours are sequentially performed.
また、本発明の一実施形態では、アルミニウム合金加工材の主な結晶粒組織は、繊維状組織等の加工組織である。熱処理等を行うと再結晶化がおこり、外周部の組織に再結晶組織が生成する場合があるが、再結晶の厚さを1.0mm以下に制御することが好ましい。 Further, in one embodiment of the present invention, the main crystal grain structure of the aluminum alloy processed material is a processed structure such as a fibrous structure. When heat treatment or the like is performed, recrystallization occurs, and a recrystallized structure may be generated in the structure of the outer peripheral portion.
上記の実施形態に係るアルミニウム合金加工材は、スポ‐ツ等のレジャー用品、産業機器、自動車用部品等の構成部材等として利用することができる。 The aluminum alloy processed material according to the above embodiment can be used as leisure articles such as sports, industrial equipment, components for automobile parts and the like.
以下に、本発明について実施例を用いて説明するが、本発明はこれらの実施例に限定されるものではない。 Hereinafter, the present invention will be described using examples, but the present invention is not limited to these examples.
(アルミニウム合金試験材の作製)
下記[表1]のA〜Jの成分の鋳塊を連続鋳造にて得た。鋳塊の直径は254mmである。これらの鋳塊をHO処理(460℃、24時間、昇温速度は50℃/h)した後、800mmの長さに切断した。
(Production of aluminum alloy test material)
Ingots of the components A to J in Table 1 below were obtained by continuous casting. The diameter of the ingot is 254 mm. These ingots were subjected to HO treatment (460 ° C., 24 hours, heating rate 50 ° C./h), and then cut into 800 mm lengths.
上記のA〜Jの成分の各鋳塊を押出加工することにより供試材を得た。
押出材の直径は25mmで2本立、間接押出で実施した。押出条件は、押出比は52、鋳塊温度380℃、押出速度は1m/minであった。
A test material was obtained by extruding each of the ingots of the above components A to J.
The diameter of the extruded material was 25 mm, and two pieces were extruded, and indirect extrusion was performed. The extrusion conditions were an extrusion ratio of 52, an ingot temperature of 380 ° C., and an extrusion speed of 1 m / min.
次いで溶体化処理を行った。溶体化温度は460℃で2時間であった。その後、速やかに水冷し、室温で3日間自然時効した(焼き入れ処理)。その後、120℃、24時間で人工時効した。 Next, a solution treatment was performed. The solution temperature was 460 ° C. for 2 hours. Thereafter, the mixture was quickly cooled with water and naturally aged at room temperature for 3 days (quenching treatment). Thereafter, artificial aging was performed at 120 ° C. for 24 hours.
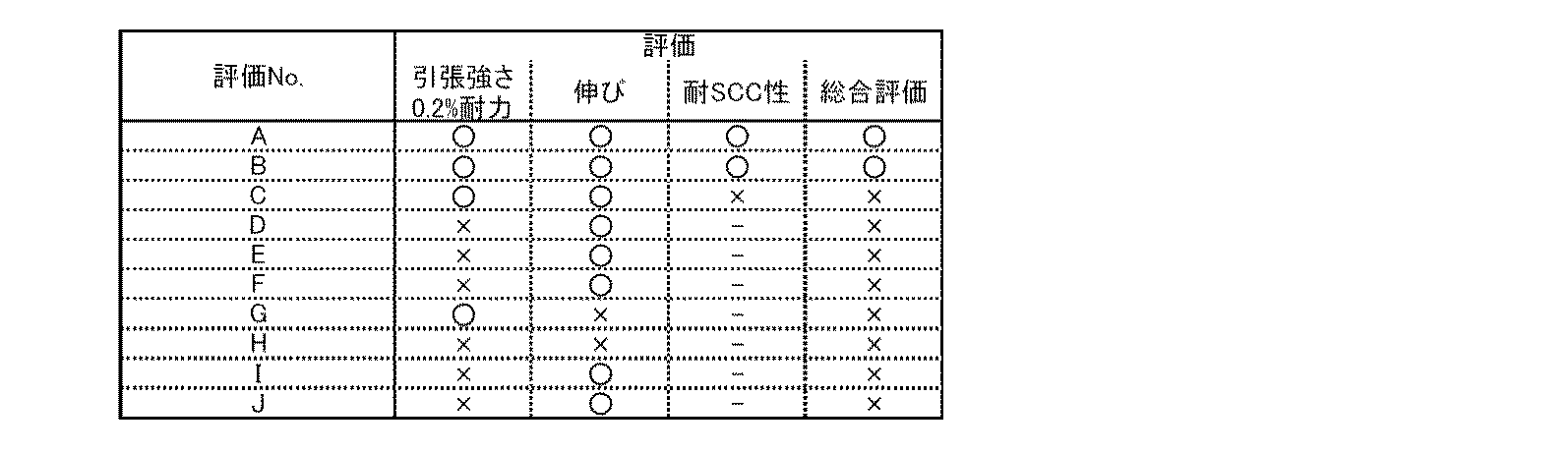
表1から理解されるように、実施例A及びBの組成が、本発明の規定を満足するものである。実施例CからJの組成は、本発明の規定する範囲外のものである。 As understood from Table 1, the compositions of Examples A and B satisfy the requirements of the present invention. The compositions of Examples C to J are outside the range defined by the present invention.
(アルミニウム合金試験材の特性評価)
アルミニウム合金試験材の強度を、引張試験により評価した。引張試験はJIS14A号の引張試験片に加工して実施した。試験片の平行部形状は径10mm、長さ60mmとし、標点間距離は50mmとした。引張試験はJIS2201に準拠して実施した。
引張試験の結果を[表2]に示す。
(Characteristic evaluation of aluminum alloy test material)
The strength of the aluminum alloy test material was evaluated by a tensile test. The tensile test was performed by processing a tensile test piece according to JIS 14A. The parallel part shape of the test piece was 10 mm in diameter and 60 mm in length, and the distance between gauge points was 50 mm. The tensile test was performed according to JIS2201.
The results of the tensile test are shown in [Table 2].
押出材の外周部に生成した再結晶組織は押出LT断面を研磨後に、改良タッカー液でエッチングし、実体顕微鏡で観察し、その厚さを測定した。 The recrystallized structure formed on the outer peripheral portion of the extruded material was etched with a modified Tucker solution after polishing the extruded LT cross section, observed with a stereoscopic microscope, and its thickness was measured.
引張試験では、引張強さ、0.2%耐力、破断伸びを測定した。引張試験では、引張強さが770MPa以上、0.2%耐力が750MPa以上、かつ破断伸びが8%以上であることを基準として、これらを満たすものを合格とした。 In the tensile test, the tensile strength, 0.2% proof stress, and elongation at break were measured. In the tensile test, those satisfying these criteria based on a tensile strength of 770 MPa or more, a 0.2% proof stress of 750 MPa or more, and a breaking elongation of 8% or more were judged as acceptable.
次いで、上記の引張試験で合格したA、B、Cについて、耐応力腐食割れ性(耐SCC性)を評価した。
Cリング試験片を製作し、JISH8711に準拠し、応力腐食割れ試験(SCC試験)を実施した。Cリング試験片は外径19mm、厚さ19mmとした。応力は耐力値の75%に相当する応力を負荷した。腐食液は3.5%NaClであり、この水溶液に、10分間浸漬、50分間乾燥を1サイクルとして、30サイクル実施し、割れの有無を判断した。
Next, stress corrosion cracking resistance (SCC resistance) was evaluated for A, B, and C that passed the above tensile test.
A C-ring test piece was manufactured, and a stress corrosion cracking test (SCC test) was performed in accordance with JIS H8711. The C-ring test piece had an outer diameter of 19 mm and a thickness of 19 mm. As the stress, a stress corresponding to 75% of the proof stress was applied. The etchant was 3.5% NaCl, and 30 cycles were performed, with one cycle consisting of immersion in this aqueous solution for 10 minutes and drying for 50 minutes, and the presence or absence of cracks was determined.
上記の引張特性と耐SCC性の目標値を満足したものを、総合評価で合格(○)とした。 Those satisfying the target values of the above tensile properties and SCC resistance were evaluated as acceptable (O) in the overall evaluation.
下記の[表3]に、実施例AからJについての、特性評価の結果を示す。 [Table 3] below shows the results of the characteristic evaluations for Examples A to J.
上記の結果によれば、A及びBでは、0.2%耐力、伸び、耐SCC性の条件を満たし総合評価が合格となった。この結果から、本発明で規定するアルミニウム合金加工材では、高い強度が得られることがわかる。 According to the above results, A and B satisfied the conditions of 0.2% proof stress, elongation, and SCC resistance, and passed the comprehensive evaluation. From this result, it is understood that the aluminum alloy processed material specified in the present invention can obtain high strength.
Cは、耐SCC性が満足なものではなかった。これは、Crの添加量が本発明で規定する下限値未満であるためだと考えられる。
Dは、0.2%耐力の結果が合格基準を満たしていない。これは、Cuの添加量が本発明で規定する上限値より多く、Mg/Cu比が、本発明で規定する範囲外であるため、析出強化が不十分であったためだと考えられる。
Eは、0.2%耐力の結果が合格基準を満たしていない。これは、Cuの添加量が本発明で規定する上限値より多く、Mg/Cu比が、本発明で規定する範囲外であるため、析出強化が不十分であったためだと考えられる。
C did not have satisfactory SCC resistance. This is presumably because the amount of Cr added is less than the lower limit specified in the present invention.
As for D, the result of 0.2% proof stress does not satisfy the acceptance criteria. This is considered to be because the amount of Cu added was larger than the upper limit specified in the present invention, and the Mg / Cu ratio was out of the range specified in the present invention, so that precipitation strengthening was insufficient.
E shows that the result of 0.2% proof stress does not satisfy the acceptance criteria. This is considered to be because the amount of Cu added was larger than the upper limit specified in the present invention, and the Mg / Cu ratio was out of the range specified in the present invention, so that precipitation strengthening was insufficient.
Fは、0.2%耐力の結果が合格基準を満たしていない。これは、Zn添加量が規定する値より少なく、かつ、Cuの添加量が本発明で規定する上限値より多く、Mg/Cu比が、本発明で規定する範囲外であるため、析出強化が不十分であったためだと考えられる。
Gは、伸びの結果が合格基準を満たしていない。これは、Znの添加量が本発明で規定する上限値より多いため、晶出部が増加したためであると考えられる。
As for F, the result of 0.2% proof stress did not satisfy the acceptance criteria. This is because the amount of Zn added is smaller than the value specified, the amount of Cu added is larger than the upper limit specified in the present invention, and the Mg / Cu ratio is out of the range specified in the present invention. Probably because it was insufficient.
G shows that the elongation results do not meet the acceptance criteria. This is considered to be because the amount of Zn added was larger than the upper limit specified in the present invention, and the crystallized portion increased.
Hは、0.2%耐力と伸びの結果が合格基準を満たしていない。これは、Mgの添加量が本発明で規定する上限値より多く、Mg/Cu比が、本発明で規定する範囲外であり、Mg+Cuが本発明で規定する上限値より多いため、晶出部が増加したためであると考えられる。
Iは、0.2%耐力の結果が合格基準を満たしていない。これは、Mgの添加量が本発明で規定する下限値未満であり、かつMg/Cu比が、本発明で規定する範囲外であり、析出強化が不十分であったためだと考えられる。
Jは、0.2%耐力の結果が合格基準を満たしていない。ZrとCrの添加量が本発明で規定する下限値未満であるため、押出加工組織が形成されず、所望の強度が得られなかったと考えられる。
As for H, the results of 0.2% proof stress and elongation do not satisfy the acceptance criteria. This is because the addition amount of Mg is larger than the upper limit specified in the present invention, the Mg / Cu ratio is outside the range specified in the present invention, and Mg + Cu is larger than the upper limit specified in the present invention. It is thought that this was due to an increase in
As for I, the result of 0.2% proof stress does not satisfy the acceptance criteria. This is considered to be because the amount of Mg added was less than the lower limit specified in the present invention, the Mg / Cu ratio was outside the range specified in the present invention, and the precipitation strengthening was insufficient.
As for J, the result of 0.2% proof stress did not satisfy the acceptance criteria. Since the addition amounts of Zr and Cr are less than the lower limit specified in the present invention, it is considered that an extruded structure was not formed and a desired strength was not obtained.
上記の結果から、本発明で規定する組成を満たすアルミニウム合金加工材は、本発明で規定する組成を満たさないアルミニウム合金加工材に比べて、高い強度が得られることがわかる。 From the above results, it is understood that the aluminum alloy processed material satisfying the composition specified in the present invention can obtain higher strength than the aluminum alloy processed material not satisfying the composition specified in the present invention.
Claims (3)
残部がAlと不可避的不純物からなり、
前記Mg,Cuの含有量は、Mg/Cu≧2.5の関係を満たし、Zr、Crの含有量の合計が、0.12質量%以上0.42質量%以下であり、塑性加工組織を有し、結晶粒組織が繊維状組織を含み、再結晶組織の再結晶の厚さが1.0mm以下であることを特徴とするアルミニウム合金加工材の製造方法であって、均質化処理、塑性加工、溶体化処理、焼き入れ処理及び時効処理を順次施すことを特徴とする、製造方法。 Zn: 9% by mass to 11% by mass, Mg: 2.0% by mass to less than 2.8% by mass, Cu: more than 0.8% by mass to 1.2% by mass, Zr: 0.08% by mass Not less than 0.25% by mass and Cr; not less than 0.02% by mass and not more than 0.30% by mass;
The balance consists of Al and inevitable impurities,
The content of Mg and Cu satisfies the relationship of Mg / Cu ≧ 2.5, and the total content of Zr and Cr is 0.12% by mass or more and 0.42% by mass or less. A method for producing a processed aluminum alloy material , comprising: a crystal grain structure including a fibrous structure; and a recrystallized structure having a recrystallized thickness of 1.0 mm or less. A manufacturing method comprising sequentially performing processing, solution treatment, quenching, and aging .
塑性加工として、押出比が5から100、鋳塊温度が300℃から450℃の熱間押出し加工、
440℃から470℃に保持する溶体化処理、
450℃から100℃の範囲を1000℃/分以上の冷却速度で冷却する焼き入れ処理、
100℃から180℃で10時間から30時間の時効処理を順次施すことを特徴とする請求項1又は2に記載の製造方法。 Homogenization treatment at a heating rate of 50 ° C./h or less,
As the plastic working, an extrusion ratio of 5 to 100 and an ingot temperature of 300 ° C. to 450 ° C. are hot extruded.
Solution treatment maintained at 440 ° C. to 470 ° C.
A quenching process of cooling a range of 450 ° C. to 100 ° C. at a cooling rate of 1000 ° C./min or more;
The method according to claim 1, wherein the aging treatment is sequentially performed at 100 ° C. to 180 ° C. for 10 hours to 30 hours.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015031364A JP6638192B2 (en) | 2015-02-20 | 2015-02-20 | Aluminum alloy processing material and method of manufacturing the same |
| PCT/JP2016/053994 WO2016132994A1 (en) | 2015-02-20 | 2016-02-10 | Aluminum alloy worked material, and manufacturing method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2015031364A JP6638192B2 (en) | 2015-02-20 | 2015-02-20 | Aluminum alloy processing material and method of manufacturing the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2016153516A JP2016153516A (en) | 2016-08-25 |
| JP6638192B2 true JP6638192B2 (en) | 2020-01-29 |
Family
ID=56689378
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2015031364A Active JP6638192B2 (en) | 2015-02-20 | 2015-02-20 | Aluminum alloy processing material and method of manufacturing the same |
Country Status (2)
| Country | Link |
|---|---|
| JP (1) | JP6638192B2 (en) |
| WO (1) | WO2016132994A1 (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106399777B (en) * | 2016-11-11 | 2018-03-09 | 佛山科学技术学院 | A kind of high intensity high-hardenability ultra-high-strength aluminum alloy and preparation method thereof |
| BR112021024430A2 (en) * | 2019-06-03 | 2022-01-18 | Novelis Inc | Ultra-high strength aluminum alloy products and methods for manufacturing them |
| TWI741962B (en) * | 2021-04-16 | 2021-10-01 | 圓融金屬粉末股份有限公司 | Aluminum-nickel-copper alloy and manufacturing method thereof |
| CN115354201A (en) * | 2022-08-29 | 2022-11-18 | 广东中色研达新材料科技股份有限公司 | High-strength 7-series aluminum alloy for 3C electronic products and processing technology thereof |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH08295977A (en) * | 1995-04-21 | 1996-11-12 | Sumitomo Light Metal Ind Ltd | High strength aluminum alloy extruded material with excellent fatigue strength and motorcycle front fork outer tube material made of the extruded material |
| FR2838135B1 (en) * | 2002-04-05 | 2005-01-28 | Pechiney Rhenalu | CORROSIVE ALLOY PRODUCTS A1-Zn-Mg-Cu WITH VERY HIGH MECHANICAL CHARACTERISTICS, AND AIRCRAFT STRUCTURE ELEMENTS |
| JP5343333B2 (en) * | 2007-07-06 | 2013-11-13 | 日本軽金属株式会社 | Method for producing high-strength aluminum alloy material with excellent resistance to stress corrosion cracking |
| CN102732761B (en) * | 2012-06-18 | 2014-01-08 | 中国航空工业集团公司北京航空材料研究院 | A kind of 7000 series aluminum alloy material and preparation method thereof |
| JP6249435B2 (en) * | 2012-08-10 | 2017-12-20 | 日本軽金属株式会社 | Aluminum-zinc alloy extruded material and method for producing the same |
-
2015
- 2015-02-20 JP JP2015031364A patent/JP6638192B2/en active Active
-
2016
- 2016-02-10 WO PCT/JP2016/053994 patent/WO2016132994A1/en not_active Ceased
Also Published As
| Publication number | Publication date |
|---|---|
| WO2016132994A1 (en) | 2016-08-25 |
| JP2016153516A (en) | 2016-08-25 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6955483B2 (en) | High-strength aluminum alloy extruded material with excellent corrosion resistance and good hardenability and its manufacturing method | |
| JP5345056B2 (en) | Heat-treatable high-strength aluminum alloy | |
| JP5561846B2 (en) | High strength aluminum alloy material and manufacturing method thereof | |
| JP6022882B2 (en) | High strength aluminum alloy extruded material and manufacturing method thereof | |
| JP6420553B2 (en) | Aluminum alloy, aluminum alloy wire, aluminum alloy wire manufacturing method, aluminum alloy member manufacturing method, and aluminum alloy member | |
| JP6344816B2 (en) | High-strength aluminum alloy extruded thin section and method for producing the same | |
| JP5834077B2 (en) | Aluminum alloy and method for producing extruded profile using the same | |
| CN115427598B (en) | Magnesium alloy, magnesium alloy sheet, magnesium alloy rod, method for producing magnesium alloy, and magnesium alloy member | |
| JP2015519475A5 (en) | Improved free-cutting wrought aluminum alloy product and manufacturing method thereof | |
| SI24911A (en) | High-strength aluminum alloy Al-Mg-Si and procedure for its manufacture | |
| JP2009013479A (en) | High strength aluminum alloy material excellent in stress corrosion cracking resistance and method for producing the same | |
| JPH10219381A (en) | High strength aluminum alloy excellent in intergranular corrosion resistance and method for producing the same | |
| JP7459496B2 (en) | Manufacturing method for aluminum alloy forgings | |
| JP6612029B2 (en) | High strength aluminum alloy extruded material with excellent impact resistance and method for producing the same | |
| JP2004084058A (en) | Method for producing aluminum alloy forging for transport structural material and aluminum alloy forging | |
| JP5204793B2 (en) | High strength aluminum alloy extruded material with excellent stress corrosion cracking resistance | |
| JP2020066752A (en) | Al-Mg-Si-BASED ALUMINUM ALLOY EXTRUSION MATERIAL AND METHOD FOR MANUFACTURING THE SAME | |
| JP6638192B2 (en) | Aluminum alloy processing material and method of manufacturing the same | |
| JP7093611B2 (en) | Aluminum alloy for extruded material and method for manufacturing extruded material and extruded material using it | |
| JP2009203516A (en) | Aluminum alloy | |
| KR101680041B1 (en) | Wrought magnesium alloy having high ductility and high toughness and method for preparing the same | |
| JPH10219413A (en) | Manufacturing method of high strength aluminum alloy with excellent intergranular corrosion resistance | |
| JP2022161591A (en) | Aluminum alloy-made forged member and method for manufacturing the same | |
| JP2022506542A (en) | 2XXX Aluminum Lithium Alloy | |
| JP6638193B2 (en) | Aluminum alloy processing material and method of manufacturing the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20170703 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20180717 |
|
| A601 | Written request for extension of time |
Free format text: JAPANESE INTERMEDIATE CODE: A601 Effective date: 20180918 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20181108 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20190416 |
|
| A521 | Request for written amendment filed |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20190611 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20191126 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20191209 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 6638192 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |
|
| R250 | Receipt of annual fees |
Free format text: JAPANESE INTERMEDIATE CODE: R250 |