JP2009294202A - Differentiation method for involvement of tight junction in material transport - Google Patents
Differentiation method for involvement of tight junction in material transport Download PDFInfo
- Publication number
- JP2009294202A JP2009294202A JP2009092355A JP2009092355A JP2009294202A JP 2009294202 A JP2009294202 A JP 2009294202A JP 2009092355 A JP2009092355 A JP 2009092355A JP 2009092355 A JP2009092355 A JP 2009092355A JP 2009294202 A JP2009294202 A JP 2009294202A
- Authority
- JP
- Japan
- Prior art keywords
- skin
- involvement
- distribution
- tight junction
- transport
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 210000001578 tight junction Anatomy 0.000 title claims abstract description 51
- 102000000591 Tight Junction Proteins Human genes 0.000 title claims abstract description 34
- 108010002321 Tight Junction Proteins Proteins 0.000 title claims abstract description 34
- 238000000034 method Methods 0.000 title claims abstract description 23
- 230000004069 differentiation Effects 0.000 title claims abstract description 4
- 239000000463 material Substances 0.000 title description 2
- 210000003491 skin Anatomy 0.000 claims description 78
- 238000011282 treatment Methods 0.000 claims description 22
- 230000006378 damage Effects 0.000 claims description 18
- 208000027418 Wounds and injury Diseases 0.000 claims description 17
- 208000014674 injury Diseases 0.000 claims description 17
- YDNKGFDKKRUKPY-JHOUSYSJSA-N C16 ceramide Natural products CCCCCCCCCCCCCCCC(=O)N[C@@H](CO)[C@H](O)C=CCCCCCCCCCCCCC YDNKGFDKKRUKPY-JHOUSYSJSA-N 0.000 claims description 15
- CRJGESKKUOMBCT-VQTJNVASSA-N N-acetylsphinganine Chemical compound CCCCCCCCCCCCCCC[C@@H](O)[C@H](CO)NC(C)=O CRJGESKKUOMBCT-VQTJNVASSA-N 0.000 claims description 15
- 229940106189 ceramide Drugs 0.000 claims description 15
- ZVEQCJWYRWKARO-UHFFFAOYSA-N ceramide Natural products CCCCCCCCCCCCCCC(O)C(=O)NC(CO)C(O)C=CCCC=C(C)CCCCCCCCC ZVEQCJWYRWKARO-UHFFFAOYSA-N 0.000 claims description 15
- VVGIYYKRAMHVLU-UHFFFAOYSA-N newbouldiamide Natural products CCCCCCCCCCCCCCCCCCCC(O)C(O)C(O)C(CO)NC(=O)CCCCCCCCCCCCCCCCC VVGIYYKRAMHVLU-UHFFFAOYSA-N 0.000 claims description 15
- 210000000434 stratum corneum Anatomy 0.000 claims description 9
- 238000002372 labelling Methods 0.000 claims description 4
- 229920006395 saturated elastomer Polymers 0.000 claims description 4
- 235000014113 dietary fatty acids Nutrition 0.000 claims description 3
- 238000012850 discrimination method Methods 0.000 claims description 3
- 229930195729 fatty acid Natural products 0.000 claims description 3
- 239000000194 fatty acid Substances 0.000 claims description 3
- 150000004665 fatty acids Chemical class 0.000 claims description 3
- 238000010306 acid treatment Methods 0.000 claims description 2
- 230000037204 skin physiology Effects 0.000 abstract description 11
- 230000008591 skin barrier function Effects 0.000 abstract description 4
- 239000000470 constituent Substances 0.000 abstract 2
- 230000035479 physiological effects, processes and functions Effects 0.000 abstract 1
- 239000002609 medium Substances 0.000 description 36
- FIWQZURFGYXCEO-UHFFFAOYSA-M sodium;decanoate Chemical compound [Na+].CCCCCCCCCC([O-])=O FIWQZURFGYXCEO-UHFFFAOYSA-M 0.000 description 25
- 238000012423 maintenance Methods 0.000 description 23
- 230000032258 transport Effects 0.000 description 17
- 239000000126 substance Substances 0.000 description 15
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 8
- 238000002073 fluorescence micrograph Methods 0.000 description 8
- 241000206755 Palmaria Species 0.000 description 6
- 210000004027 cell Anatomy 0.000 description 6
- 239000012228 culture supernatant Substances 0.000 description 6
- 239000008187 granular material Substances 0.000 description 6
- 239000007850 fluorescent dye Substances 0.000 description 5
- 230000006870 function Effects 0.000 description 5
- 239000001963 growth medium Substances 0.000 description 5
- 239000001569 carbon dioxide Substances 0.000 description 4
- 229910002092 carbon dioxide Inorganic materials 0.000 description 4
- 239000002537 cosmetic Substances 0.000 description 4
- 238000012258 culturing Methods 0.000 description 4
- GHVNFZFCNZKVNT-UHFFFAOYSA-N decanoic acid Chemical compound CCCCCCCCCC(O)=O GHVNFZFCNZKVNT-UHFFFAOYSA-N 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- LNLLNTMHVMIMOG-YUMQZZPRSA-N Gamma-glutamyl-Lysine Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CC[C@H](N)C(O)=O LNLLNTMHVMIMOG-YUMQZZPRSA-N 0.000 description 3
- 230000004888 barrier function Effects 0.000 description 3
- 210000004207 dermis Anatomy 0.000 description 3
- 210000002615 epidermis Anatomy 0.000 description 3
- 230000005284 excitation Effects 0.000 description 3
- 230000002285 radioactive effect Effects 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 239000005632 Capric acid (CAS 334-48-5) Substances 0.000 description 2
- 208000018380 Chemical injury Diseases 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 102220645376 Sodium- and chloride-dependent betaine transporter_C10R_mutation Human genes 0.000 description 2
- 229940098773 bovine serum albumin Drugs 0.000 description 2
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 238000001215 fluorescent labelling Methods 0.000 description 2
- 239000012634 fragment Substances 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- WWZKQHOCKIZLMA-UHFFFAOYSA-N octanoic acid Chemical compound CCCCCCCC(O)=O WWZKQHOCKIZLMA-UHFFFAOYSA-N 0.000 description 2
- 239000002953 phosphate buffered saline Substances 0.000 description 2
- 238000002360 preparation method Methods 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- 230000028327 secretion Effects 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- WWUZIQQURGPMPG-UHFFFAOYSA-N (-)-D-erythro-Sphingosine Natural products CCCCCCCCCCCCCC=CC(O)C(N)CO WWUZIQQURGPMPG-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 239000005635 Caprylic acid (CAS 124-07-2) Substances 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 241000700199 Cavia porcellus Species 0.000 description 1
- 102000002029 Claudin Human genes 0.000 description 1
- 108050009302 Claudin Proteins 0.000 description 1
- 102000008186 Collagen Human genes 0.000 description 1
- 108010035532 Collagen Proteins 0.000 description 1
- 102000029816 Collagenase Human genes 0.000 description 1
- 108060005980 Collagenase Proteins 0.000 description 1
- 102000016942 Elastin Human genes 0.000 description 1
- 108010014258 Elastin Proteins 0.000 description 1
- 231100000948 EpiDerm Skin Irritation Test Toxicity 0.000 description 1
- 102000009123 Fibrin Human genes 0.000 description 1
- 108010073385 Fibrin Proteins 0.000 description 1
- BWGVNKXGVNDBDI-UHFFFAOYSA-N Fibrin monomer Chemical compound CNC(=O)CNC(=O)CN BWGVNKXGVNDBDI-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 108010063907 Glutathione Reductase Proteins 0.000 description 1
- 102100036442 Glutathione reductase, mitochondrial Human genes 0.000 description 1
- 102000005720 Glutathione transferase Human genes 0.000 description 1
- 108010070675 Glutathione transferase Proteins 0.000 description 1
- JZNWSCPGTDBMEW-UHFFFAOYSA-N Glycerophosphorylethanolamin Natural products NCCOP(O)(=O)OCC(O)CO JZNWSCPGTDBMEW-UHFFFAOYSA-N 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- 108060001084 Luciferase Proteins 0.000 description 1
- 239000005089 Luciferase Substances 0.000 description 1
- 102400000740 Melanocyte-stimulating hormone alpha Human genes 0.000 description 1
- 101710200814 Melanotropin alpha Proteins 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- -1 OCT compound Chemical class 0.000 description 1
- 102000003940 Occludin Human genes 0.000 description 1
- 108090000304 Occludin Proteins 0.000 description 1
- 241000283973 Oryctolagus cuniculus Species 0.000 description 1
- 102000016387 Pancreatic elastase Human genes 0.000 description 1
- 108010067372 Pancreatic elastase Proteins 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 102000003992 Peroxidases Human genes 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 241000282898 Sus scrofa Species 0.000 description 1
- ATBOMIWRCZXYSZ-XZBBILGWSA-N [1-[2,3-dihydroxypropoxy(hydroxy)phosphoryl]oxy-3-hexadecanoyloxypropan-2-yl] (9e,12e)-octadeca-9,12-dienoate Chemical compound CCCCCCCCCCCCCCCC(=O)OCC(COP(O)(=O)OCC(O)CO)OC(=O)CCCCCCC\C=C\C\C=C\CCCCC ATBOMIWRCZXYSZ-XZBBILGWSA-N 0.000 description 1
- 239000004480 active ingredient Substances 0.000 description 1
- AWUCVROLDVIAJX-UHFFFAOYSA-N alpha-glycerophosphate Natural products OCC(O)COP(O)(O)=O AWUCVROLDVIAJX-UHFFFAOYSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 230000008901 benefit Effects 0.000 description 1
- 229910052799 carbon Inorganic materials 0.000 description 1
- 150000001732 carboxylic acid derivatives Chemical class 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 229920001436 collagen Polymers 0.000 description 1
- 229960002424 collagenase Drugs 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 229920002549 elastin Polymers 0.000 description 1
- 210000001339 epidermal cell Anatomy 0.000 description 1
- 238000011156 evaluation Methods 0.000 description 1
- 229950003499 fibrin Drugs 0.000 description 1
- 210000002950 fibroblast Anatomy 0.000 description 1
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 150000002339 glycosphingolipids Chemical class 0.000 description 1
- 125000002887 hydroxy group Chemical group [H]O* 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 210000004692 intercellular junction Anatomy 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 210000002510 keratinocyte Anatomy 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 239000003068 molecular probe Substances 0.000 description 1
- 230000003472 neutralizing effect Effects 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 229960002446 octanoic acid Drugs 0.000 description 1
- 238000010647 peptide synthesis reaction Methods 0.000 description 1
- 108040007629 peroxidase activity proteins Proteins 0.000 description 1
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 description 1
- 150000008104 phosphatidylethanolamines Chemical class 0.000 description 1
- 150000003905 phosphatidylinositols Chemical class 0.000 description 1
- 150000003904 phospholipids Chemical class 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 150000003839 salts Chemical class 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 210000004927 skin cell Anatomy 0.000 description 1
- 230000004215 skin function Effects 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- WWUZIQQURGPMPG-KRWOKUGFSA-N sphingosine Chemical compound CCCCCCCCCCCCC\C=C\[C@@H](O)[C@@H](N)CO WWUZIQQURGPMPG-KRWOKUGFSA-N 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 229910052713 technetium Inorganic materials 0.000 description 1
- GKLVYJBZJHMRIY-UHFFFAOYSA-N technetium atom Chemical compound [Tc] GKLVYJBZJHMRIY-UHFFFAOYSA-N 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
- WHNFPRLDDSXQCL-UAZQEYIDSA-N α-msh Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(N)=O)NC(=O)[C@H](CO)NC(C)=O)C1=CC=C(O)C=C1 WHNFPRLDDSXQCL-UAZQEYIDSA-N 0.000 description 1
Images
Landscapes
- Cosmetics (AREA)
- Medicines Containing Plant Substances (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Investigating Or Analysing Biological Materials (AREA)
Abstract
Description
本発明は、化粧料などの皮膚外用剤の生理学的作用の探求に有用な、皮膚内における物質輸送、物質分布の鑑別法に関し、更に詳細には、タイトジャンクションがどの程度に特定の物質の皮膚内における物質輸送、物質分布に関与するかを鑑別する技術に関する。 The present invention relates to a method for distinguishing substance transport and substance distribution in the skin, which is useful for exploring the physiological effects of external preparations for skin such as cosmetics, and more particularly, to what extent a tight junction is a specific substance skin. The present invention relates to a technique for discriminating whether a substance is involved in substance transport or substance distribution.
皮膚におけるバリア機能は、生体にとっての異物の生体内への侵入を防ぐ意味で重要な機能の一つとなっている。かかるバリア機能の不全は肌荒れ、炎症などの原因ともなり、その保全策は化粧料分野においては一大課題となっている。この様な皮膚バリア機能の因子としては、角層細胞の形状や接合状態に起因する角層のバリア機能、表皮・真皮の含水量などの皮膚保湿性などが挙げられ、単純なものではなく、多くの因子が絡み合っていることが既に知られている。これらの因子で近年特に注目されているのは、表皮顆粒層に存在するタイトジャンクション蛋白であり、かかるタイトジャンクション蛋白を介した細胞接合の不全に皮膚バリア機能の低下が起因すると言う説である。(例えば、特許文献1、特許文献2を参照)このタイトジャンクションの機能に注目した化粧料素材のスクリーニング法も前記の特許文献に開示されている。この方法では、タイトジャンクション機能の不全による皮膚バリア機能の低下を改善する成分を、数値として評価でき、タイトジャンクションが皮膚内の物質の移動に対して、何らかの機能を担っていることは推定させているが、皮膚機能としての物質の輸送、分布に対しての働きは明らかにしていない。この様なタイトジャンクションの物質の輸送、分布に対して担っている働きを明らかにすることは、皮膚生理において、タイトジャンクションの果たすべき役割を明確にすることにつながり、非常に有益なことと言える。
The barrier function in the skin is one of the important functions in terms of preventing foreign bodies from entering the living body. Such insufficiency of the barrier function causes rough skin, inflammation, and the like, and its conservation measures are a major issue in the cosmetics field. Such skin barrier function factors include the stratum corneum barrier function due to the shape and bonding state of the stratum corneum cells, skin moisture retention such as the water content of the epidermis and dermis, and is not simple, It is already known that many factors are intertwined. In recent years, these factors have attracted particular attention as a tight junction protein present in the epidermal granule layer, and the theory is that a decrease in skin barrier function is caused by the failure of cell junction via the tight junction protein. (See, for example,
タイトジャンクションの障害因子として、紫外線(例えば、特許文献3を参照)、カプリン酸などが存することは既に知られている(例えば、非特許文献1を参照)が、これらを用いて、培養皮膚三次元モデルのタイトジャンクションを傷害し、それが皮膚内の物質の輸送、分布にどの様な影響を与えるかについては何らの検討も存しない。 It is already known that ultraviolet rays (for example, see Patent Document 3), capric acid, and the like exist as obstacle factors for tight junctions (for example, see Non-Patent Document 1). There is no examination of how the original tight junction is damaged and how it affects the transport and distribution of substances in the skin.
本発明は、この様な状況下為されたものであり、皮膚生理学的に有用な、皮膚内の物質の輸送と分布に対してのタイトジャンクションの関与の程度を明らかにせしめる技術を提供することを課題とする。 The present invention has been made under such circumstances, and provides a technique for clarifying the degree of involvement of tight junctions in the transport and distribution of substances in the skin, which is useful in skin physiology. Is an issue.
この様な状況に鑑みて、本発明者らは、皮膚生理学的に有用な、皮膚内の物質の輸送と分布に対してのタイトジャンクションの関与の程度を明らかにせしめる技術を求めて、鋭意研究努力を重ねた結果、皮膚乃至は培養皮膚三次元モデルのタイトジャンクションを傷害処理して、タイトジャンクション傷害皮膚モデルを作製し、標識してなる皮膚関連成分を含む培地中で培養し、前記標識の分布状況を指標とすることにより、この様な課題が解決できることを見出し、発明を完成させるに至った。即ち、本発明は、以下に示す通りである。
(1)皮膚乃至は培養皮膚三次元モデルのタイトジャンクションを傷害処理して、タイトジャンクション傷害皮膚モデルを作成し、標識してなる皮膚関連成分を含む培地中で培養し、前記標識の分布状況を指標とすることを特徴とする、皮膚関連成分の分布又は輸送へのタイトジャンクションの関与の程度の鑑別法。
(2)対照として、タイトジャンクションの傷害処理を施さない皮膚乃至は培養皮膚三次元モデルを用いることを特徴とする、(1)に記載の皮膚関連成分の分布又は輸送へのタイトジャンクションの関与の程度の鑑別法。
(3)前記鑑別法が、角層側の培地中に放出された、標識してなる皮膚関連成分を計測することを特徴とする、請求項1又は2に記載の皮膚関連成分の輸送へのタイトジャンクションの関与の程度の鑑別法。
(4)前記皮膚乃至は皮膚三次元モデルが、培養ヒト皮膚三次元モデルであることを特徴とする、(1)〜(3)の何れか1項に記載の皮膚関連成分の分布又は輸送へのタイトジャンクションの関与の程度の鑑別法。
(5)前記タイトジャンクションの傷害処理が、中鎖飽和直鎖脂肪酸処理であることを特徴とする、(1)〜(4)何れか1項に記載の皮膚関連成分の分布又は輸送へのタイトジャンクションの関与の程度の鑑別法。
(6)標識してなる皮膚関連成分は、蛍光標識されたセラミドであることを特徴とする、(1)〜(5)何れか1項に記載の皮膚関連成分の分布又は輸送へのタイトジャンクションの関与の程度の鑑別法。
In view of such a situation, the present inventors have sought for a technique for clarifying the degree of involvement of tight junctions in the transport and distribution of substances in the skin, which are useful for skin physiology. As a result of repeated efforts, the tight junction of the skin or the cultured skin three-dimensional model is injured to prepare a tight junction injured skin model, which is cultured in a medium containing a labeled skin-related component. The inventors have found that such a problem can be solved by using the distribution status as an index, and have completed the invention. That is, the present invention is as follows.
(1) Tight junction of skin or cultured skin three-dimensional model is injured, tight junction injury skin model is prepared, cultured in a medium containing skin-related components formed by labeling, and the distribution status of the label is determined. A method of distinguishing the degree of involvement of tight junctions in the distribution or transport of skin-related components, characterized by being an index.
(2) As a control, a skin or a cultured skin three-dimensional model not subjected to tight junction injury treatment is used, and the involvement of tight junctions in the distribution or transport of skin-related components according to (1) How to differentiate degree.
(3) The said discrimination method measures the skin-related component formed by labeling, which is released into the medium on the stratum corneum side, and the transport of the skin-related component according to
(4) To the distribution or transport of skin-related components according to any one of (1) to (3), wherein the skin or skin three-dimensional model is a cultured human skin three-dimensional model A method to differentiate the degree of involvement of tight junctions.
(5) The tight treatment to distribution or transport of skin-related components according to any one of (1) to (4), wherein the injury treatment of the tight junction is a medium-chain saturated linear fatty acid treatment A method of distinguishing the degree of junction involvement.
(6) The skin-related component formed by labeling is a ceramide that is fluorescently labeled, and the tight junction to the distribution or transport of the skin-related component according to any one of (1) to (5) The method of differentiation of the degree of involvement.
本発明によれば、皮膚生理学的に有用な、皮膚内の物質の輸送と分布に対してのタイトジャンクションの関与の程度を明らかにせしめる技術を提供することができる。 ADVANTAGE OF THE INVENTION According to this invention, the technique which makes clear the degree of participation of the tight junction with respect to the transport and distribution of the substance in skin useful for skin physiology can be provided.
本発明の皮膚関連成分の分布又は輸送へのタイトジャンクションの関与の程度の鑑別法は、皮膚乃至は培養皮膚三次元モデルのタイトジャンクションを傷害処理して、タイトジャンクション傷害皮膚モデルを作成し、標識してなる皮膚関連成分を含む培地中で培養し、前記標識の分布状況を指標とすることを特徴とする。前記の皮膚としては、所望により体毛を除去し、生体より採取した皮膚断片であって、角層などを除いた、表皮顆粒層、表皮基底層及び真皮部分からなるものが好ましく、マウス、ラット、モルモット、ブタ、ウサギの皮膚断片が好ましい。又、前記培養皮膚三次元モデルとしては、ヒト乃至はヒトを除く動物の皮膚より採取した、正常な(癌化していない)ケラチノサイト、フィブロブラストなどの皮膚細胞を培養し、三次元構造を構築し、皮膚の皮膚の構造に疑似させたものが好ましく、この様な形態の市販品を購入して使用することも出来る。好ましい市販品としては、例えば、倉敷紡績株式会社から販売されている「EFT-400」(正常培養ヒト三次元皮膚モデル)などが好適に例示できる。特に表皮成分のみで構成される「EPI-200」や、USA MaTek社から販売されている「EpiDerm」などがさらに好適に例示できる。かかる皮膚乃至は培養皮膚三次元モデルは、顆粒層側から傷害手段を講じてタイトジャンクション部分を傷害する。傷害手段としては、例えば、紫外線や化学物質などが好ましく例示でき、紫外線であれば、波長280〜320nmの紫外線を7.5〜200mJ/cm2、さらに好ましくは50〜160mJ/cm2の単位あたりのエネルギー量で照射すれば良く、化学的な処置であれば、カプリン酸、カプリル酸などの中鎖長(炭素数8〜12)の脂肪族飽和直鎖脂肪酸またはその一価金属塩の0.1〜10mM、さらに好ましくは0.5〜2mMの溶液を真皮側乃至は表皮基底層側から、5〜24時間、さらに好ましくは10〜15時間作用させれば良い。また、オクルディンやクローディンの細胞外ドメインを認識する中和抗体を使用することもできる。これらの傷害処置の内、特に好ましいものは化学的傷害処置であり、なかでもカプリン酸ナトリウムによる処理が特に好ましい。これはタイトジャンクションに対して均質な損傷がなしうるからである。この様な均質な傷害は、後記の傷害タイトジャンクションの回復促進剤のスクリーニングにおいては、そのメカニズムを的確に鑑別する上で非常に重要な因子となる。処置後傷害手段は直ちに皮膚乃至は培養皮膚三次元モデルより離脱させる。離脱は、紫外線照射であれば照射を終了することによりできるし、化学的傷害手段であれば、培地乃至はPBS(リン酸緩衝生理食塩水)などで洗浄することによりなしうる。斯くして得られたタイトジャンクション傷害皮膚モデルは、標識してなる皮膚関連成分を含む培地中で培養し、しかる後に、タイトジャンクション傷害皮膚モデルに取り込まれなかった標識された皮膚関連成分を洗浄などによって除去した後に、暫く培養を続け、培養上清中に放出される、標識してなる皮膚関連成分を測定したり、組織片を切り出し、標本に加工し、標識を認識しうる観察手段によって標識の分布状況を観察される。 The method for distinguishing the degree of involvement of tight junctions in the distribution or transport of skin-related components according to the present invention is to treat a tight junction of a skin or a cultured skin three-dimensional model, create a tight junction injury skin model, It is cultured in a medium containing a skin-related component, and the distribution state of the label is used as an index. The skin is preferably a skin fragment collected from a living body by removing body hair as desired, and excluding the stratum corneum, and composed of an epidermal granule layer, an epidermal basal layer, and a dermis portion, such as a mouse, rat, Guinea pig, pig and rabbit skin fragments are preferred. In addition, as the cultured skin three-dimensional model, normal (non-cancerous) keratinocytes, fibroblasts and other skin cells collected from human or non-human animal skin are cultured to construct a three-dimensional structure. Those which simulate the skin structure of the skin are preferable, and commercially available products of such a form can also be purchased and used. Preferable examples of commercially available products include “EFT-400” (normally cultured human three-dimensional skin model) sold by Kurashiki Boseki Co., Ltd. In particular, “EPI-200” composed only of an epidermis component, “EpiDerm” sold by USA MaTek, and the like can be more suitably exemplified. Such skin or cultured skin three-dimensional models injure tight junctions by taking injury means from the granule layer side. As the injury means, for example, ultraviolet rays and chemical substances can be preferably exemplified. In the case of ultraviolet rays, ultraviolet rays having a wavelength of 280 to 320 nm are per unit of 7.5 to 200 mJ / cm 2 , more preferably 50 to 160 mJ / cm 2 . In the case of chemical treatment, the saturated saturated linear fatty acid having a medium chain length (carbon number of 8 to 12) such as capric acid or caprylic acid or a monovalent metal salt thereof may be used. A solution of 1 to 10 mM, more preferably 0.5 to 2 mM, may be allowed to act for 5 to 24 hours, more preferably 10 to 15 hours from the dermis side or epidermal basal layer side. A neutralizing antibody that recognizes the extracellular domain of occludin or claudin can also be used. Among these treatments for injury, particularly preferred is a treatment for chemical injury, and treatment with sodium caprate is particularly preferred. This is because homogeneous damage can occur to tight junctions. Such homogeneous injury is a very important factor in accurately identifying the mechanism in screening for a recovery accelerator for injury tight junction described later. The post-treatment injury means is immediately removed from the skin or cultured skin three-dimensional model. Detachment can be achieved by terminating the irradiation if ultraviolet irradiation is performed, and can be performed by washing with a medium or PBS (phosphate buffered saline) or the like if chemical injury means. The tight junction injury skin model thus obtained is cultured in a medium containing a labeled skin-related component, and then the labeled skin-related component that has not been incorporated into the tight junction injury skin model is washed. After removing by the above, continue culturing for a while, measure the labeled skin-related components released into the culture supernatant, cut out the tissue piece, process it into a specimen, and label it with observation means that can recognize the label The distribution situation is observed.
ここで、前記標識してなる皮膚関連成分を構成する皮膚関連成分としては、皮膚生理学上、皮膚の生理に関与する蓋然性の高い成分であり、例えば、タイプ1〜7のセラミド、スフィンゴシン、スフィンゴミエリン、スフィンゴ糖脂質、スフィンゴ燐脂質、ホスファチジルコリン、ホスファチジルイノシトール、ホスファチジルグリセロール、ホスファチジルエタノールアミンなどの燐脂質類、コラーゲン、エラスチンなどの繊維素、αーMSH、コラゲナーゼ、エラスターゼ、グルタチオントランスフェラーゼ、グルタチオンレダクターゼ、プロテアーゼなどの酵素類が好適に例示できる。標識としては、例えば、フルオレセイン等の蛍光発色基をエステル結合やアミド結合で導入したり、アミノ基にヨウ素、テクネチウムなどの放射性同位体を導入したり、水酸基を放射性のフッ素で置換したりした放射性同位体に誘導する方法、ルシェフェラーゼやペルオキシダーゼなどの酵素とコンジュゲートを形成せしめ、基質を反応させて発色や発光をさせる方法などが好ましく例示できる。これらの標識の内、好ましいものは、検知感度及び安全性がともに高い蛍光標識である。この様な蛍光標識は、例えば、対象となる成分と蛍光標識基を有するカルボン酸とをDCC等のペプチド合成試薬の存在下反応させることにより得ることができる。又、この様な標識皮膚関連成分には市販されているものも存し、この様な市販品を購入し利用することもできる。この様な市販品としては例えば、セラミドの蛍光標識体である「BODIPY(登録商標)FL-C5-Ceramide-BSA」(Molecular Probe社製)が特に好ましく例示できる。 Here, the skin-related component constituting the labeled skin-related component is a component highly likely to be involved in skin physiology in terms of skin physiology. For example, type 1-7 ceramide, sphingosine, sphingomyelin , Glycosphingolipid, sphingophospholipid, phosphatidylcholine, phosphatidylinositol, phosphatidylglycerol, phosphatidylethanolamine and other phospholipids, collagen, elastin and other fibrin, α-MSH, collagenase, elastase, glutathione transferase, glutathione reductase, protease, etc. These enzymes can be preferably exemplified. Examples of the label include a radioactive chromophore such as fluorescein introduced by an ester bond or an amide bond, a radioactive isotope such as iodine or technetium introduced into an amino group, or a hydroxyl group substituted with radioactive fluorine. Preferred examples include an isotope induction method, a method in which a conjugate is formed with an enzyme such as luciferase or peroxidase, and a substrate is reacted to develop color or emit light. Among these labels, preferred are fluorescent labels having high detection sensitivity and safety. Such a fluorescent label can be obtained, for example, by reacting a target component with a carboxylic acid having a fluorescent labeling group in the presence of a peptide synthesis reagent such as DCC. In addition, some of such labeled skin-related components are commercially available, and such commercially available products can be purchased and used. As such a commercially available product, for example, “BODIPY (registered trademark) FL-C5-Ceramide-BSA” (manufactured by Molecular Probe), which is a fluorescent label of ceramide, can be particularly preferably exemplified.
この様な標識された皮膚生理関連成分は、例えば市販の培地、「EPI-100」(倉敷紡績株式会社)などの液体培地中に1〜10μM、好ましくは2.5〜7.5μM溶解させ、かかる培地で前記タイトジャンクション傷害皮膚モデルを0.5〜10時間、好ましくは1〜5時間培養し、皮膚モデル中に取り込ませる。この時、培養時間のドーズを振り、輸送・分布速度の指標としても良い。斯くして、標識してなる皮膚生理関連成分をチャージしたタイトジャンクション傷害皮膚モデルは、そのまま培養を継続して培養上清中に放出される標識してなる皮膚生理関連成分を測定することができるし、所望により組織固定をして、切片などの観察標本に切り出す。観察標本は、標識検知に適した観察手段で観察される。蛍光標識であれば、顕微鏡標本に切り出し、蛍光顕微鏡下観察される。 Such a labeled skin physiology-related component is dissolved in a liquid medium such as a commercially available medium, “EPI-100” (Kurashiki Boseki Co., Ltd.), for example, 1 to 10 μM, preferably 2.5 to 7.5 μM, The tight junction injury skin model is cultured in such a medium for 0.5 to 10 hours, preferably 1 to 5 hours, and incorporated into the skin model. At this time, the dose of the culture time may be varied to serve as an index for transportation / distribution speed. Thus, the tight junction injury skin model charged with the labeled skin physiology-related component can measure the labeled skin physiology-related component released into the culture supernatant by continuing the culture as it is. Then, if desired, the tissue is fixed and cut into an observation specimen such as a section. The observation specimen is observed by an observation means suitable for detecting a sign. If it is a fluorescent label, it is cut out into a microscope specimen and observed under a fluorescent microscope.
観察に際しては、タイトジャンクションを傷害せずに、同様の処理で作成した、タイトジャンクション非傷害皮膚モデルを対照において観察することにより、タイトジャンクションの皮膚生理関連成分の輸送と分布に対する寄与が明確になり好ましい。 When observing a tight junction non-injured skin model created in the same manner without damaging the tight junction in the control, the contribution of the tight junction to the transport and distribution of skin physiology-related components becomes clear. preferable.
以下に、実施例を挙げて、本発明について更に詳細に説明を加える。 Hereinafter, the present invention will be described in more detail with reference to examples.
<工程1>「EPI-200」を培養プレートに移し、表皮基底層側に200μlの5μM 「BODIPY FL-C5-Ceramide-BSA」を含む維持培地(EPI-100 クラボウ製)を加え、37℃、5%二酸化炭素で4時間培養した後、4℃冷蔵庫で1時間静置した。
<工程2>500μlの新鮮な維持培地に置換し、37℃、5% CO2で20時間培養した。
<工程3>300μlの5%脱脂牛血清アルブミンを含む維持培地に置換し、4℃で30分間静置した。
<工程4>工程3を2回繰り返した。
<工程5>新鮮な維持培地及び1mMカプリン酸ナトリウムを含む維持培地にそれぞれ置換し、37℃、5%二酸化炭素で培養した。
<工程6>培養12時間後、1mM C10を含まない新鮮な維持培地に置換した(C10R)。経時的に培養上清の蛍光強度(励起波長485nm、蛍光波長535nm)を測定した。
<
<Step 2> The medium was replaced with 500 μl of a fresh maintenance medium, and cultured at 37 ° C. and 5% CO 2 for 20 hours.
<Step 3> The medium was replaced with a maintenance medium containing 300 μl of 5% defatted bovine serum albumin and allowed to stand at 4 ° C. for 30 minutes.
<Step 4> Step 3 was repeated twice.
<
<Step 6> After 12 hours of culture, the culture medium was replaced with a fresh maintenance medium not containing 1 mM C10 (C10R). The fluorescence intensity (excitation wavelength: 485 nm, fluorescence wavelength: 535 nm) of the culture supernatant was measured over time.
図1は培養上清の蛍光強度を測定することにより放出されたセラミドの相対量を示したグラフである。カプリン酸ナトリウムで処理していない場合(図1中Control)は時間経過とともに培養上清中に放出されるセラミドが増加していくが、1mMカプリン酸ナトリウム処理により(図1中C10)、セラミドの増加が妨げられた。培養5時間後にカプリン酸ナトリウムを含まない新鮮な維持培地に交換することにより(図1中C10R)、セラミドの放出量が再び増加した。 FIG. 1 is a graph showing the relative amount of ceramide released by measuring the fluorescence intensity of the culture supernatant. When not treated with sodium caprate (Control in FIG. 1), the amount of ceramide released into the culture supernatant increases with time. By treatment with 1 mM sodium caprate (C10 in FIG. 1), The increase was hindered. By changing to a fresh maintenance medium not containing sodium caprate after 5 hours of culture (C10R in FIG. 1), the amount of ceramide released increased again.
<工程1>「EPI-200」を培養プレートに移し、表皮基底層側に200μlの5μM 「BODIPY FL-C5-Ceramide-BSA」を含む維持培地(EPI-100、クラボウ製)を加え、37℃、5%二酸化炭素で4時間培養した後、4℃冷蔵庫で1時間静置した。
<工程2>500μlの新鮮な維持培地に置換し、37℃、5% CO2で20時間培養した。
<工程3>300μlの5%脱脂牛血清アルブミンを含む維持培地に置換し、4℃で30分間静置した。
<工程4>工程3を2回繰り返した。
<工程5>新鮮な維持培地及び1mMカプリン酸ナトリウムを含む維持培地にそれぞれ置換し、37℃、5%二酸化炭素で培養した。
<工程6>培養12時間後、1mMカプリン酸ナトリウムを含まない新鮮な維持培地に置換した。
<工程7>一定時間培養後「EPI-200」を支持体から切り離し、「OCTコンパウンド」(サクラファインテック社製)に包埋して液体窒素で凍結した。
<工程8>凍結切片を作製し、蛍光顕微鏡下、励起波長470nm/蛍光波長525nmで観察した。
<
<Step 2> The medium was replaced with 500 μl of a fresh maintenance medium, and cultured at 37 ° C. and 5% CO 2 for 20 hours.
<Step 3> The medium was replaced with a maintenance medium containing 300 μl of 5% defatted bovine serum albumin and allowed to stand at 4 ° C. for 30 minutes.
<Step 4> Step 3 was repeated twice.
<
<Step 6> After 12 hours of culture, the culture medium was replaced with a fresh maintenance medium not containing 1 mM sodium caprate.
<Step 7> After culturing for a certain period of time, “EPI-200” was detached from the support, embedded in “OCT compound” (manufactured by Sakura Finetech), and frozen with liquid nitrogen.
<Step 8> Frozen sections were prepared and observed under a fluorescence microscope at an excitation wavelength of 470 nm / fluorescence wavelength of 525 nm.
図2はカプリン酸ナトリウム未処理、図3は1mMカプリン酸ナトリウム処理12時間後、図4は1mMカプリン酸ナトリウム処理12時間後、カプリン酸ナトリウムを含まない新鮮な維持培地に置換したときの「EPI-200」の凍結切片の蛍光画像である。角層(図中点線で囲まれた部分)直下の表皮顆粒層(矢印)の蛍光(セラミド)がカプリン酸ナトリウム未処理では細胞間の上部分に多く認められるのに対して(図2)、カプリン酸ナトリウム処理により消失していることが判る(図3)。この後カプリン酸ナトリウムを含まない新鮮な維持培地に置換することにより、再び表皮基底層の細胞間の上部分(矢印)に蛍光(セラミド)が認められる(図4)。 FIG. 2 shows an untreated sodium caprate, FIG. 3 shows 12 hours after treatment with 1 mM sodium caprate, and FIG. 4 shows “EPI when replaced with a fresh maintenance medium not containing sodium caprate after 12 hours of 1 mM sodium caprate treatment. -200 "is a fluorescence image of a frozen section. Whereas the fluorescence (ceramide) of the epidermal granule layer (arrow) just below the stratum corneum (part surrounded by a dotted line in the figure) is not observed with sodium caprate, it is often observed in the upper part between cells (FIG. 2). It turns out that it has disappeared by the treatment with sodium caprate (FIG. 3). Thereafter, by replacing with a fresh maintenance medium not containing sodium caprate, fluorescence (ceramide) is observed again in the upper part (arrow) between cells of the epidermal basal layer (FIG. 4).
実施例2と同様に<工程6>まで行うが、<工程6>の後、別個の「EPI-200」を、500μg/mlε,γ−グルタミルリジン、0.1%パルマリア抽出物それぞれ単独で含有する維持培地に置換し6時間培養を続けた。その後培地を回収し、細胞外に分泌された「BODIPY FL-C5-Ceramide-BSA」の蛍光強度(励起波長485nm、蛍光波長535nm)を、ARVO SX Multilabel Counter(PerkinElmer社製)を用いて測定した。さらに、その後<工程8>まで進めた。 <Step 6> is performed in the same manner as in Example 2, but after <Step 6>, separate "EPI-200" is contained alone at 500 µg / mlε, γ-glutamyllysine and 0.1% palmaria extract, respectively. The culture medium was replaced with the maintenance medium to be cultured for 6 hours. Thereafter, the medium was collected, and the fluorescence intensity (excitation wavelength: 485 nm, fluorescence wavelength: 535 nm) of “BODIPY FL-C5-Ceramide-BSA” secreted outside the cells was measured using ARVO SX Multilabel Counter (manufactured by PerkinElmer). . Furthermore, it advanced to <process 8> after that.
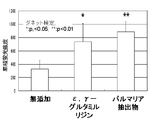
結果を図5に示すが、ε,γ−グルタミルリジン、パルマリア抽出物は、いずれも無添加コントロールよりも有意に蛍光標識セラミド「BODIPY FL-C5-Ceramide-BSA」の細胞外への分泌量を促進する。また図6は1mMカプリン酸ナトリウム処理12時間後、維持培地に置換して12時間後、図7は1mMカプリン酸ナトリウム処理12時間後、500μg/mlε,γ−グルタミルリジンを含有する維持培地に置換して12時間後、図8は1mMカプリン酸ナトリウム処理12時間後、0.1%パルマリア抽出物を含有する維持培地に置換して12時間後の「EPI-200」の凍結切片の蛍光画像である。角層直下の表皮顆粒層の蛍光(セラミド)が図6では細胞間の上部分には多く認められないのに対して、図7、8ではそれぞれ表皮顆粒層の細胞間の上部分(矢印)に多く認められる。つまり、ε,γ−グルタミルリジン及びパルマリア抽出物はそれぞれ単独で、セラミドを角層方向に分泌する表皮細胞の働きを早める効果を有することが判る。即ち本鑑別法は、セラミドの組織内輸送へのタイトジャンクションの関与の程度の評価に適していることが判る。 The results are shown in FIG. 5, and ε, γ-glutamyllysine and palmaria extract all significantly increased the amount of secretion of fluorescently labeled ceramide “BODIPY FL-C5-Ceramide-BSA” to the outside of the control without addition. Facilitate. 6 shows 12 hours after treatment with 1 mM sodium caprate and 12 hours after replacement with maintenance medium. FIG. 7 shows 12 hours after treatment with 1 mM sodium caprate and replacement with maintenance medium containing 500 μg / ml ε, γ-glutamyllysine. 12 hours later, FIG. 8 is a fluorescence image of a frozen section of “EPI-200” after 12 hours after treatment with 1 mM sodium caprate and after replacement with a maintenance medium containing 0.1% palmaria extract. is there. In FIG. 6, the fluorescence (ceramide) in the epidermal granule layer just below the stratum corneum is not observed in the upper part between cells, whereas in FIGS. 7 and 8, the upper part (arrow) between the cells in the epidermal granule layer, respectively. Many are recognized. That is, it turns out that (epsilon), (gamma) -glutamyl lysine and a palmaria extract each have the effect which accelerates | stimulates the function of the epidermal cell which secretes ceramide toward a stratum corneum. That is, it can be seen that this discrimination method is suitable for evaluating the degree of involvement of tight junctions in the intracerebral transport of ceramide.
本発明は、化粧料などの皮膚外用剤の有効成分の評価に応用できる。 The present invention can be applied to the evaluation of active ingredients of external preparations for skin such as cosmetics.
Claims (6)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2009092355A JP2009294202A (en) | 2008-05-09 | 2009-04-06 | Differentiation method for involvement of tight junction in material transport |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2008123071 | 2008-05-09 | ||
| JP2009092355A JP2009294202A (en) | 2008-05-09 | 2009-04-06 | Differentiation method for involvement of tight junction in material transport |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013028561A Division JP2013100355A (en) | 2008-05-09 | 2013-02-18 | Promoter of recovery of injured tight junction |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2009294202A true JP2009294202A (en) | 2009-12-17 |
| JP2009294202A5 JP2009294202A5 (en) | 2012-05-31 |
Family
ID=41542493
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2009092355A Pending JP2009294202A (en) | 2008-05-09 | 2009-04-06 | Differentiation method for involvement of tight junction in material transport |
| JP2013028561A Pending JP2013100355A (en) | 2008-05-09 | 2013-02-18 | Promoter of recovery of injured tight junction |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2013028561A Pending JP2013100355A (en) | 2008-05-09 | 2013-02-18 | Promoter of recovery of injured tight junction |
Country Status (1)
| Country | Link |
|---|---|
| JP (2) | JP2009294202A (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2009294200A (en) * | 2008-05-09 | 2009-12-17 | Pola Chem Ind Inc | Differentiation method for tight junction function restoring agent |
Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05301814A (en) * | 1992-02-25 | 1993-11-16 | Pola Chem Ind Inc | Cosmetic |
| JP2005035926A (en) * | 2003-07-14 | 2005-02-10 | Japan Science & Technology Agency | Drug for promoting ceramide transport, base sequence for producing the drug, method for measuring activity for promoting ceramide release, and method for measuring activity for promoting transmembrane movement of ceramide |
| JP2006250786A (en) * | 2005-03-11 | 2006-09-21 | Pola Chem Ind Inc | Method for identifying action to skin |
| JP2007174931A (en) * | 2005-12-27 | 2007-07-12 | Pola Chem Ind Inc | Epidermal keratinocyte layer membrane and utilization of the epidermal keratinocyte layer membrane |
| WO2008006235A2 (en) * | 2006-07-10 | 2008-01-17 | Esbatech Ag | Scfv antibodies which pass epithelial and/or endothelial layers |
| JP2008026092A (en) * | 2006-07-20 | 2008-02-07 | Pola Chem Ind Inc | Discrimination method of serviceability of material for improving skin barrier function |
Family Cites Families (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP3807782B2 (en) * | 1995-06-22 | 2006-08-09 | ライオン株式会社 | Hyaluronidase inhibitor |
| JP4050799B2 (en) * | 1995-07-19 | 2008-02-20 | 雪印乳業株式会社 | Tight junction formation accelerator |
| JPH10236918A (en) * | 1997-02-21 | 1998-09-08 | Lion Corp | Skin patch |
| JP2005137307A (en) * | 2003-11-07 | 2005-06-02 | Japan Science & Technology Agency | Method and apparatus for evaluating damage of cells and tissues |
| US20080194611A1 (en) * | 2005-06-03 | 2008-08-14 | Alverdy John C | Modulation of Cell Barrier Dysfunction |
| TW200831125A (en) * | 2006-12-08 | 2008-08-01 | Pola Chem Ind Inc | A discrimination method of skin barrier function, the screening method of skin barrier function reinforced material by using the discrimination method, the skin barrier function reinforced materials, and the cosmetic material containing the skin barrier |
-
2009
- 2009-04-06 JP JP2009092355A patent/JP2009294202A/en active Pending
-
2013
- 2013-02-18 JP JP2013028561A patent/JP2013100355A/en active Pending
Patent Citations (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH05301814A (en) * | 1992-02-25 | 1993-11-16 | Pola Chem Ind Inc | Cosmetic |
| JP2005035926A (en) * | 2003-07-14 | 2005-02-10 | Japan Science & Technology Agency | Drug for promoting ceramide transport, base sequence for producing the drug, method for measuring activity for promoting ceramide release, and method for measuring activity for promoting transmembrane movement of ceramide |
| JP2006250786A (en) * | 2005-03-11 | 2006-09-21 | Pola Chem Ind Inc | Method for identifying action to skin |
| JP2007174931A (en) * | 2005-12-27 | 2007-07-12 | Pola Chem Ind Inc | Epidermal keratinocyte layer membrane and utilization of the epidermal keratinocyte layer membrane |
| WO2008006235A2 (en) * | 2006-07-10 | 2008-01-17 | Esbatech Ag | Scfv antibodies which pass epithelial and/or endothelial layers |
| JP2008026092A (en) * | 2006-07-20 | 2008-02-07 | Pola Chem Ind Inc | Discrimination method of serviceability of material for improving skin barrier function |
Non-Patent Citations (3)
| Title |
|---|
| JPN6013012572; 杉山義宣: '皮膚の機能制御とスキンケア' 化学と生物 Vol.46,No.2, 20080201, P.135-141 * |
| JPN6013012573; 倉沢真澄ほか: '表皮接着装置タイトジャンクションと表皮内カルシウムイオン濃度勾配の関係の研究' NIRS-M (Natl. Inst. Radiol. Sci.) , 20080313, P.22 * |
| JPN6013012574; 杉林康ほか: '中鎖脂肪酸適用に伴うタイトジャンクションタンパク質の分布変化' 日本薬学会年会要旨集 Vol.128th,No.4, 20080305, P.87 * |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2013100355A (en) | 2013-05-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Noskovicova et al. | Suppression of the fibrotic encapsulation of silicone implants by inhibiting the mechanical activation of pro-fibrotic TGF-β | |
| de Miranda et al. | Effects of hydrolyzed collagen supplementation on skin aging: a systematic review and meta‐analysis | |
| Musumeci et al. | Biosynthesis of collagen I, II, RUNX2 and lubricin at different time points of chondrogenic differentiation in a 3D in vitro model of human mesenchymal stem cells derived from adipose tissue | |
| Vičanová et al. | Normalization of epidermal calcium distribution profile in reconstructed human epidermis is related to improvement of terminal differentiation and stratum corneum barrier formation | |
| Ohto-Fujita et al. | Solubilized eggshell membrane supplies a type III collagen-rich elastic dermal papilla | |
| US20230364000A1 (en) | Compositions comprising combinations of collagen or elastin polypeptides with active ingredients and methods of using same | |
| Nebelung et al. | Simultaneous anabolic and catabolic responses of human chondrocytes seeded in collagen hydrogels to long-term continuous dynamic compression | |
| Glicksman et al. | Differentiation of motor nerve terminals formed in the absence of muscle fibres | |
| Ni et al. | Preparation of injectable porcine skin-derived collagen and its application in delaying skin aging by promoting the adhesion and chemotaxis of skin fibroblasts | |
| Oikarinen et al. | Pachydermoperiostosis: analysis of the connective tissue abnormality in one family | |
| Roelandse et al. | Focal motility determines the geometry of dendritic spines☆ | |
| Sharma et al. | Biomimetic molecules lower catabolic expression and prevent chondroitin sulfate degradation in an osteoarthritic ex vivo model | |
| Samra et al. | Melatonin exerts prominent, differential epidermal and dermal anti-aging properties in aged human eyelid skin ex vivo | |
| Choi et al. | Tonsil-derived mesenchymal stem cells incorporated in reactive oxygen species-releasing hydrogel promote bone formation by increasing the translocation of cell surface GRP78 | |
| Konstantinou et al. | Mechanisms of senescence and anti-senescence strategies in the skin | |
| Iwai et al. | Protein carbonyls damage the water-holding capacity of the stratum corneum | |
| US6079415A (en) | Process for evaluating the damage induced in skin by UV-A radiation | |
| Khandelwal et al. | Age-dependent increase in green autofluorescence of blood erythrocytes | |
| Satake et al. | Estimation of extracellular matrix production using a cultured-chondrocyte-based gate ion-sensitive field-effect transistor | |
| JP2009294202A (en) | Differentiation method for involvement of tight junction in material transport | |
| Hajem et al. | The regulatory role of the tetrapeptide A c SDKP in skin and hair physiology and the prevention of ageing effects in these tissues–a potential cosmetic role | |
| Potokar et al. | Trafficking of astrocytic vesicles in hippocampal slices | |
| Sun et al. | Topical retinol restores type I collagen production in photoaged forearm skin within four weeks | |
| WO2016194760A1 (en) | Cosmetic method and dermatological topical agent for use therein, migration imparting agent, and method for screening components for use in cosmetic method for improving skin state | |
| Smith et al. | Cartilage tissue homeostasis |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20120405 |
|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20120405 |
|
| RD03 | Notification of appointment of power of attorney |
Free format text: JAPANESE INTERMEDIATE CODE: A7423 Effective date: 20120405 |
|
| A977 | Report on retrieval |
Free format text: JAPANESE INTERMEDIATE CODE: A971007 Effective date: 20121210 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20121218 |
|
| A521 | Written amendment |
Free format text: JAPANESE INTERMEDIATE CODE: A523 Effective date: 20130218 |
|
| A131 | Notification of reasons for refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A131 Effective date: 20130319 |
|
| A02 | Decision of refusal |
Free format text: JAPANESE INTERMEDIATE CODE: A02 Effective date: 20130827 |