CN1193026C - Z-pyrazoline-5-ones - Google Patents
Z-pyrazoline-5-ones Download PDFInfo
- Publication number
- CN1193026C CN1193026C CNB008136823A CN00813682A CN1193026C CN 1193026 C CN1193026 C CN 1193026C CN B008136823 A CNB008136823 A CN B008136823A CN 00813682 A CN00813682 A CN 00813682A CN 1193026 C CN1193026 C CN 1193026C
- Authority
- CN
- China
- Prior art keywords
- pyrazolin
- methylene radical
- methyl
- pyrroles
- pyrazinyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/415—1,2-Diazoles
- A61K31/4152—1,2-Diazoles having oxo groups directly attached to the heterocyclic ring, e.g. antipyrine, phenylbutazone, sulfinpyrazone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/04—Drugs for disorders of the alimentary tract or the digestive system for ulcers, gastritis or reflux esophagitis, e.g. antacids, inhibitors of acid secretion, mucosal protectants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/14—Prodigestives, e.g. acids, enzymes, appetite stimulants, antidyspeptics, tonics, antiflatulents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P1/00—Drugs for disorders of the alimentary tract or the digestive system
- A61P1/16—Drugs for disorders of the alimentary tract or the digestive system for liver or gallbladder disorders, e.g. hepatoprotective agents, cholagogues, litholytics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P15/00—Drugs for genital or sexual disorders; Contraceptives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/02—Drugs for dermatological disorders for treating wounds, ulcers, burns, scars, keloids, or the like
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P17/00—Drugs for dermatological disorders
- A61P17/06—Antipsoriatics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P27/00—Drugs for disorders of the senses
- A61P27/02—Ophthalmic agents
- A61P27/06—Antiglaucoma agents or miotics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/12—Drugs for disorders of the metabolism for electrolyte homeostasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/08—Antiallergic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/14—Drugs for disorders of the endocrine system of the thyroid hormones, e.g. T3, T4
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P5/00—Drugs for disorders of the endocrine system
- A61P5/14—Drugs for disorders of the endocrine system of the thyroid hormones, e.g. T3, T4
- A61P5/16—Drugs for disorders of the endocrine system of the thyroid hormones, e.g. T3, T4 for decreasing, blocking or antagonising the activity of the thyroid hormones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P7/00—Drugs for disorders of the blood or the extracellular fluid
- A61P7/10—Antioedematous agents; Diuretics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/10—Drugs for disorders of the cardiovascular system for treating ischaemic or atherosclerotic diseases, e.g. antianginal drugs, coronary vasodilators, drugs for myocardial infarction, retinopathy, cerebrovascula insufficiency, renal arteriosclerosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P9/00—Drugs for disorders of the cardiovascular system
- A61P9/14—Vasoprotectives; Antihaemorrhoidals; Drugs for varicose therapy; Capillary stabilisers
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D231/00—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings
- C07D231/02—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings
- C07D231/10—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members
- C07D231/14—Heterocyclic compounds containing 1,2-diazole or hydrogenated 1,2-diazole rings not condensed with other rings having two or three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D231/18—One oxygen or sulfur atom
- C07D231/20—One oxygen atom attached in position 3 or 5
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/06—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings linked by a carbon chain containing only aliphatic carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Endocrinology (AREA)
- Ophthalmology & Optometry (AREA)
- Vascular Medicine (AREA)
- Heart & Thoracic Surgery (AREA)
- Obesity (AREA)
- Immunology (AREA)
- Dermatology (AREA)
- Rheumatology (AREA)
- Oncology (AREA)
- Pulmonology (AREA)
- Cardiology (AREA)
- Physical Education & Sports Medicine (AREA)
- Epidemiology (AREA)
- Communicable Diseases (AREA)
- Emergency Medicine (AREA)
- Nutrition Science (AREA)
- Reproductive Health (AREA)
- Pain & Pain Management (AREA)
- Gastroenterology & Hepatology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Urology & Nephrology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
结构式(I)的化合物及其生理上可接受的盐是丝氨酸/苏氨酸和酪氨酸激酶活性抑制剂。几种其活性被这些化合物抑制的酪氨酸激酶与血管生成过程有关。因此,这些化合物可以改善其中血管生成或内皮细胞增殖为因子的疾病状况。这些化合物可用于治疗癌和增殖疾病。
The compounds of structural formula (I) and their physiologically acceptable salts are inhibitors of serine/threonine and tyrosine kinase activity. Several tyrosine kinases whose activity is inhibited by these compounds are involved in angiogenesis. Therefore, these compounds can improve disease conditions in which angiogenesis or endothelial cell proliferation is a factor. These compounds may be used to treat cancer and proliferative diseases.
Description
技术领域technical field
本申请要求1999年7月30日提交的美国临时申请60/146,563的利益,该临时申请的全部内容结合在本发明中作为参考。This application claims the benefit of US Provisional Application 60/146,563, filed July 30, 1999, which is incorporated herein by reference in its entirety.
本发明涉及一些2-吡唑啉-5-酮,它们是蛋白激酶、特别是酪氨酸激酶和丝氨酸/苏氨酸激酶的抑制剂,其中一些是新化合物,本发明还涉及含有这些吡唑啉酮的药物组合物以及这些吡唑啉酮的制备方法。The present invention relates to certain 2-pyrazolin-5-ones, which are inhibitors of protein kinases, especially tyrosine kinases and serine/threonine kinases, some of which are novel compounds, and to compounds containing these pyrazoles Pharmaceutical compositions of pyrazolones and methods for the preparation of these pyrazolones.
背景技术Background technique
已经识别的蛋白激酶至少有400种。这些酶催化靶蛋白底物的磷酸化作用。磷酸化作用通常是磷酸基由ATP向蛋白底物的转移反应。向其上转移磷酸的靶底物的特定结构是酪氨酸、丝氨酸或苏氨酸残基。由于这些氨基酸残基是磷酰基转移的靶底物,这些蛋白激酶通常被称作酪氨酸激酶或丝氨酸/苏氨酸激酶。At least 400 protein kinases have been identified. These enzymes catalyze the phosphorylation of target protein substrates. Phosphorylation is usually the transfer of a phosphate group from ATP to a protein substrate. The specific structure of the target substrate to which the phosphate is transferred is a tyrosine, serine or threonine residue. Since these amino acid residues are target substrates for phosphoryl transfer, these protein kinases are often referred to as tyrosine kinases or serine/threonine kinases.
在酪氨酸、丝氨酸或苏氨酸残基上进行的磷酸化反应和抗衡的磷酸酶反应涉及无数细胞过程,这些细胞过程以对不同的细胞内信号的应答为基础(通常是由细胞受体介导的),涉及细胞功能的调节以及细胞过程的活化或灭活。蛋白激酶的级联经常参与细胞内信号的传导,并且对于实现这些细胞过程是必须的。由于它们在这些过程中普遍存在,可以发现蛋白激酶作为质膜的整体部分或者作为细胞质酶存在、或者定位在核中,经常作为酶配合物的组分。在许多情况下,这些蛋白激酶是酶和结构蛋白配合物的基本元素,它决定了是否或者何时在细胞中发生细胞过程。Phosphorylation and counterbalancing phosphatase reactions on tyrosine, serine, or threonine residues are involved in a myriad of cellular processes that are based on responses to different intracellular signals (often initiated by cellular receptors mediated), involved in the regulation of cellular functions and the activation or inactivation of cellular processes. Cascades of protein kinases are frequently involved in the transduction of intracellular signals and are essential for the realization of these cellular processes. Because of their ubiquity in these processes, protein kinases can be found as integral parts of the plasma membrane or as cytoplasmic enzymes, or localized in the nucleus, often as components of enzyme complexes. In many cases, these protein kinases are fundamental elements of enzyme and structural protein complexes that determine if or when cellular processes occur in cells.
蛋白酪氨酸激酶。蛋白酪氨酸激酶(PTKs)是催化细胞蛋白中特定的酪氨酸残基磷酸化的酶。这些底物蛋白的后平移修饰,经常是酶本身,起着控制细胞增殖、活化或分化的分子转换器的作用(有关综述,参见Schlessinger和Ulrich,1992,Neuron 9:383-391)。在许多疾病、包括良性和恶性增殖疾病以及由不适当的免疫系统活化导致的疾病(例如自身免疫疾病)、同种移植物排斥和移植物对宿主排斥疾病中都观察到了迷行的或过度的PTKs活动。此外,内皮细胞特异性受体PTKs如KDR和Tie-2介导了血管生成过程,并因此涉及支持癌和其他与不适当的血管形成有关疾病(例如糖尿病性视网膜病、因老化斑状退化导致的脉络膜新生血管、牛皮癣、关节炎、早熟视网膜病、婴儿血管瘤)的进行。protein tyrosine kinase. Protein tyrosine kinases (PTKs) are enzymes that catalyze the phosphorylation of specific tyrosine residues in cellular proteins. Post-translational modifications of these substrate proteins, often the enzymes themselves, function as molecular switches controlling cell proliferation, activation or differentiation (for review, see Schlessinger and Ulrich, 1992, Neuron 9:383-391). Lost or excessive is observed in many diseases, including benign and malignant proliferative diseases and diseases caused by inappropriate activation of the immune system (such as autoimmune diseases), allograft rejection and graft-versus-host rejection diseases PTKs activity. Furthermore, endothelial cell-specific receptor PTKs such as KDR and Tie-2 mediate the angiogenic process and are thus implicated in supporting cancer and other diseases associated with inappropriate vascularization (e.g. diabetic retinopathy, Choroidal neovascularization, psoriasis, arthritis, retinopathy of prematurity, infantile hemangioma).
酪氨酸激酶可以是受体型(具有细胞外、跨膜和细胞内区)或非受体型(完全是细胞内的)。Tyrosine kinases can be receptor-type (having extracellular, transmembrane and intracellular domains) or non-receptor-type (entirely intracellular).
受体酪氨酸激酶(RTKs)。RTKs包括很大一族的跨膜受体,它们具有多种生物活性。目前,至少识别出了19个不同的RTK亚族。受体酪氨酸激酶(RTK)族包括对各种细胞类型生长和分化具有决定性作用的受体(Yarden和Ullrich,Ann.Rev.Biochem.57:433-478,1988;Ullrich和Schlessinger,Cell 61:243-254,1990)。一旦与配体结合,RTKs固有的功能即被活化,导致受体和多细胞底物磷酸化,之后导致多种细胞应答(Ullrich & Schlessinger,1990,Cell 61:203-212)。因此,受体酪氨酸激酶介导的信号传导由与特定生长因子(配体)的细胞外相互作用开始,通常在其后是受体的二聚作用,刺激固有蛋白酪氨酸激酶的活性以及受体磷酸根转移。由此产生了细胞内信号传导分子的结合位点,并导致与一系列胞质信号传导分子形成配合物以利于适当的细胞应答。(例如细胞分裂、分化、新陈代谢作用、在细胞外小环境中的改变),参见Schlessinger和Ullrich,1992,Neuron 9:1-20。Receptor tyrosine kinases (RTKs). RTKs comprise a large family of transmembrane receptors that possess a variety of biological activities. Currently, at least 19 different RTK subfamilies have been identified. The receptor tyrosine kinase (RTK) family includes receptors that are decisive for the growth and differentiation of various cell types (Yarden and Ullrich, Ann. Rev. Biochem. 57:433-478, 1988; Ullrich and Schlessinger, Cell 61 : 243-254, 1990). Upon ligand binding, the intrinsic functions of RTKs are activated, leading to phosphorylation of receptors and multiple cellular substrates, which subsequently lead to a variety of cellular responses (Ullrich & Schlessinger, 1990, Cell 61:203-212). Thus, receptor tyrosine kinase-mediated signaling begins with extracellular interactions with specific growth factors (ligands), often followed by dimerization of the receptor, stimulating the activity of intrinsic protein tyrosine kinases and receptor phosphate transfer. This creates binding sites for intracellular signaling molecules and leads to the formation of complexes with a range of cytoplasmic signaling molecules to facilitate appropriate cellular responses. (eg cell division, differentiation, metabolism, changes in the extracellular niche), see Schlessinger and Ullrich, 1992, Neuron 9:1-20.
具有SH2(src同源性-2)或磷酸酪氨酸结合(PTB)区的蛋白与活化的酪氨酸激酶受体及其具有高度亲合性的底物结合,以使信号传播至细胞。上述两个区均识别磷酸酪氨酸.(Fantl等,1992,Cell 69:413-423;Songyang等,1994,Mol.Cell.Biol.14:2777-2785;Songyang等,1993,Cell 72:767-778;和Koch等,1991,Science 252:668-678;Shoelson,Curr.Opin.Chem.Biol.(1997),1(2),227-234;Cowburn,Curr.Opin.Struct.Biol.(1997),7(6),835-838)。已经识别出几种与受体酪氨酸激酶(RTKs)有关的细胞内底物蛋白。它们可以分为两个主要的组:(1)具有催化区的底物;和(2)缺乏催化区、但用作接合器、并与催化活性分子有关的底物(Songyang等,1993,Cell 72:767-778)。其底物的受体或蛋白与SH2或PTB区相互作用的专一性是由直接围绕磷酸化酪氨酸残基的氨基酸残基决定的。例如,在特定受体上的SH2区与围绕磷酸酪氨酸残基的氨基酸序列之间的结合亲合力的差别与其底物磷酸化作用特性的不同有关(Songyang等,1993,Cell 72:767-778)。这些观察暗示每一受体酪氨酸激酶的功能不仅仅由其表达模式和配体有效性决定,也由被特定的受体活化的下游信号传导路径的排列、那些刺激的定时和持续时间决定。因此,磷酸化作用提供了重要的调控步骤,这决定了由特定的生长因子受体以及分化因子受体补充的信号传导路径的选择性。Proteins with SH2 (src homology-2) or phosphotyrosine-binding (PTB) domains bind activated tyrosine kinase receptors and their substrates with high affinity to propagate the signal to the cell. Both of the above two regions recognize phosphotyrosine. (Fantl et al., 1992, Cell 69:413-423; Songyang et al., 1994, Mol.Cell.Biol.14:2777-2785; Songyang et al., 1993, Cell 72:767 -778; and Koch et al., 1991, Science 252:668-678; Shoelson, Curr.Opin.Chem.Biol. (1997), 1(2), 227-234; Cowburn, Curr.Opin.Struct.Biol.( 1997), 7(6), 835-838). Several intracellular substrate proteins associated with receptor tyrosine kinases (RTKs) have been identified. They can be divided into two main groups: (1) substrates with catalytic domains; and (2) substrates lacking catalytic domains but acting as adapters and associated with catalytically active molecules (Songyang et al., 1993, Cell 72:767-778). The specificity with which the receptor or protein of its substrate interacts with the SH2 or PTB domain is determined by the amino acid residues immediately surrounding the phosphorylated tyrosine residue. For example, differences in binding affinities between the SH2 domain on a particular receptor and the amino acid sequences surrounding the phosphotyrosine residues are related to differences in the phosphorylation properties of their substrates (Songyang et al., 1993, Cell 72:767- 778). These observations imply that the function of each receptor tyrosine kinase is determined not only by its expression pattern and ligand availability, but also by the arrangement of downstream signaling pathways activated by a particular receptor, the timing and duration of those stimuli . Phosphorylation thus provides an important regulatory step that determines the selectivity of signaling pathways complemented by specific growth factor receptors as well as differentiation factor receptors.
已有提示,几种受体酪氨酸激酶如FGFR-1,PDGFR和c-Met,以及与它们结合的生长因子在血管生成中起着作用,尽管某些可能是间接地促进血管生成(Mustonen和Alitalo,J Cell Biol.129:895-898,1995)。一种被称作″胎儿肝激酶1″(FLK-1)的受体酪氨酸激酶是III型RTKs亚类中的一员。或者对于人FLK-1命名为″含激酶插入区的受体″(KDR)(Terman等,Oncogene 6:1677-83,1991)。FLK-1/KDR的另一命名为″血管内皮细胞生长因子受体2″(VEGFR-2),因为它与VEGF的结合具有高度亲合性。鼠的FLK-1/VEGFR-2又被称作NYK(Oelrichs等,Oncogene 8(1):11-15,1993)。已经分离出了DNA编码的小鼠、大鼠和人FLK-1,并且已经报道了核苷酸和编码氨基酸序列(Matthews等,Proc.Natl.Acad.Sci.USA,88:9026-30,1991;Terman等,1991,上文;Terman等,Biochem.Biophys.Res.Comm.187:1579-86,1992;Sarzani等,supra;和Millauer等,Cell 72:835-846,1993)。许多研究如在上文Millauer等的报道中提示了VEGF和FLK-1/KDR/VEGFR-2是配体-受体对,它们在血管内皮细胞的增殖和血管形成与抽芽中起着重要作用,血管内皮细胞增殖和血管形成与抽芽分别被称作血管发生和血管生成。Several receptor tyrosine kinases such as FGFR-1, PDGFR, and c-Met, and the growth factors that bind to them, have been suggested to play a role in angiogenesis, although some may promote angiogenesis indirectly (Mustonen and Alitalo, J Cell Biol. 129:895-898, 1995). A receptor tyrosine kinase known as "fetal liver kinase 1" (FLK-1) is a member of a subclass of type III RTKs. Alternatively for human FLK-1 it is designated "Kinase Inserted Region Receptor" (KDR) (Terman et al., Oncogene 6:1677-83, 1991). Another designation for FLK-1/KDR is "vascular endothelial growth factor receptor 2" (VEGFR-2) because of its high affinity binding to VEGF. Murine FLK-1/VEGFR-2 is also known as NYK (Oelrichs et al., Oncogene 8(1):11-15, 1993). DNA-encoded mouse, rat, and human FLK-1 have been isolated, and nucleotide and encoded amino acid sequences have been reported (Matthews et al., Proc.Natl.Acad.Sci.USA, 88:9026-30, 1991 ; Terman et al., 1991, supra; Terman et al., Biochem. Biophys. Res. Comm. 187:1579-86, 1992; Sarzani et al., supra; and Millauer et al., Cell 72:835-846, 1993). Many studies, such as the above report by Millauer et al., have suggested that VEGF and FLK-1/KDR/VEGFR-2 are ligand-receptor pairs that play an important role in the proliferation of vascular endothelial cells and in angiogenesis and sprouting, Vascular endothelial cell proliferation and vessel formation and sprouting are termed angiogenesis and angiogenesis, respectively.
另一种被命名为″鳍样酪氨酸激酶-1″(Flt1)的III型RTK与FLK-1/KDR有关(DeVries等,Science 255;989-991,1992;Shibuya等,Oncogene 5:519-524,1990)。Flt-1的另一种称谓是″血管内皮细胞生长因子受体1″(VEGFR-1)。迄今为止,已经发现了许多主要在内皮细胞上表达的FLK-1/KDR/VEGFR-2和Flt-1/VEGFR-1亚族。这些亚族成员特定地受许多血管内皮细胞生长因子(VEGF)配体的刺激(Klagsburn和D′Amore,Cytokine & Growth Factor Reviews 7:259-270,1996)。与FLK-1/KDR相比,血管内皮细胞生长因子(VEGF)与Flt-1的结合具有高度亲合性,并且使血管内皮细胞有丝分裂(Terman等,1992,上文;Mustonen等上文;DeVries等,上文)。据信在血管发育中Flt-1对内皮组织是必需的。Flt-1表达与小鼠胚胎中早期血管发展有关,并且与创伤愈合过程中的新血管生成有关(Mustonen和Alitalo,上文)。Flt-1在成熟器官如肾小球中的表达暗示了该受体另外的功能,这些功能与细胞生长无关(Mustonen和Alitalo,上文)。Another type III RTK named "fin-like tyrosine kinase-1" (Flt1) is related to FLK-1/KDR (DeVries et al., Science 255; 989-991, 1992; Shibuya et al., Oncogene 5:519 -524, 1990). Another name for Flt-1 is "vascular endothelial growth factor receptor 1" (VEGFR-1). To date, many FLK-1/KDR/VEGFR-2 and Flt-1/VEGFR-1 subfamilies expressed mainly on endothelial cells have been discovered. Members of these subfamilies are specifically stimulated by a number of vascular endothelial cell growth factor (VEGF) ligands (Klagsburn and D'Amore, Cytokine & Growth Factor Reviews 7:259-270, 1996). Vascular endothelial growth factor (VEGF) binds Flt-1 with high affinity compared to FLK-1/KDR and is mitogenic to vascular endothelial cells (Terman et al., 1992, supra; Mustonen et al., supra; DeVries et al. et al., supra). Flt-1 is believed to be essential for endothelial tissue in vascular development. Flt-1 expression is associated with early vascular development in mouse embryos and with neovascularization during wound healing (Mustonen and Alitalo, supra). Expression of Flt-1 in mature organs such as the glomerulus suggests additional functions for this receptor that are not related to cell growth (Mustonen and Alitalo, supra).
如上所述,最新的证据表明VEGF在刺激正常和病理血管生成中起重要作用(Jakeman等,Endocrinology 133:848-859,1993;Kolch等,Breast Cancer Research and Treatment 36:139-155,1995;Ferrara等,Endocrine Reviews 18(1);4-25,1997;Ferrara等,Regulation ofAngiogenesis(ed.L.D.Goldberg and E.M.Rosen),209-232,1997)。另外,VEGF与控制和增强血管的透过性有关(Connolly,等,J.Biol.Chem.264:20017-20024,1989;Brown等,Regulation of Angiogenesis(ed.L.D.Goldberg and E.M.Rosen),233-269,1997)。As noted above, recent evidence suggests that VEGF plays an important role in stimulating normal and pathological angiogenesis (Jakeman et al., Endocrinology 133:848-859, 1993; Kolch et al., Breast Cancer Research and Treatment 36:139-155, 1995; Ferrara et al. et al., Endocrine Reviews 18(1); 4-25, 1997; Ferrara et al., Regulation of Angiogenesis (ed. L.D. Goldberg and E.M. Rosen), 209-232, 1997). In addition, VEGF is related to the control and enhancement of blood vessel permeability (Connolly, et al., J.Biol.Chem.264:20017-20024, 1989; Brown et al., Regulation of Angiogenesis (ed.L.D.Goldberg and E.M.Rosen), 233- 269, 1997).
已经报道了由另一种mRNA剪接产生的不同形式的VEGF,包括Ferrara等所述的四个种类(J.Cell.Biochem.47:211-218,1991)。Ferrara等(上文)已经识别了与分泌性和主要细胞有关的VEGF,并且已知该蛋白以二硫化物连接的二聚物形式存在。Different forms of VEGF resulting from alternative mRNA splicing have been reported, including the four species described by Ferrara et al. (J. Cell. Biochem. 47:211-218, 1991). Ferrara et al. (supra) have identified VEGF associated with secretory and primary cells, and the protein is known to exist as a disulfide-linked dimer.
近来已经识别几种相关的VEGF同系物。但是,其在正常生理和疾病过程中的作用尚未说明。此外,VEGF中的许多成员经常在许多组织中与VEGF一同表达,并且通常能够与VEGF形成杂二聚体。这种性质或许改变了受体的特异性以及杂二聚体的生物作用,并且进一步使阐明其如下所述的特定功能复杂化(Korpelainen和Alitalo,Curr.Opin.Cell Biol.,159-164,1998,该参考文献引入本文作为参考)。Several related VEGF homologs have recently been identified. However, its role in normal physiological and disease processes has not been elucidated. In addition, many members of VEGF are frequently co-expressed with VEGF in many tissues and are often capable of forming heterodimers with VEGF. This property probably alters the specificity of the receptor and the biological role of the heterodimer, and further complicates the elucidation of its specific function as described below (Korpelainen and Alitalo, Curr. Opin. Cell Biol., 159-164, 1998, which reference is incorporated herein by reference).
胎盘生长因子(P1GF)所具有的氨基酸序列显示出与VEGF序列重要的同源性(Park等,J.Biol.Chem.269:25646-54,1994;Maglione等Oncogene 8:925-31,1993)。与VEGF一样,不同种类的P1GF由另一种mRNA的剪接产生,并且该蛋白质以二聚物的形式存在(Park等,上文)。P1GF-1和P1GF-2与Flt-1高度亲合地结合,并且P1GF-2也热切地与neuropilin-1结合(Migdal等,J.Biol.Chem.273(35):22272-22278),但是不与FLK-1/KDR结合(Park等,上文)。已有报道,当存在低浓度VEGF时,P1GF对血管的透过性和内皮细胞上VEGF的致有丝分裂的作用具有加强作用(据称是因为杂二聚体的形成所致)(Park等,上文)。Placental growth factor (P1GF) has an amino acid sequence showing significant homology to the VEGF sequence (Park et al., J. Biol. Chem. 269: 25646-54, 1994; Maglione et al. Oncogene 8: 925-31, 1993) . Like VEGF, a different species of P1GF results from the splicing of another mRNA, and the protein exists as a dimer (Park et al., supra). P1GF-1 and P1GF-2 bind Flt-1 with high affinity, and P1GF-2 also binds eagerly to neuropilin-1 (Migdal et al., J. Biol. Chem. 273(35):22272-22278), but Does not bind FLK-1/KDR (Park et al., supra). Potential effects of P1GF on vascular permeability and mitogenic effects of VEGF on endothelial cells have been reported in the presence of low concentrations of VEGF (purportedly due to heterodimer formation) (Park et al., supra arts).
VEGF-B的产生有两种同种型(167和185残基),它们还与Flt-1/VEGFR-1结合。它们可以在调控细胞外基质降解、细胞粘附以及通过调节尿激酶型纤溶酶原激活物和纤溶酶原激活物抑制剂1的表达和活性而移行中起作用(Pepper等,Proc.Natl.Acad.Sci.U.S.A.(1998),95(20):11709-11714)。VEGF-B is produced in two isoforms (167 and 185 residues), which also bind Flt-1/VEGFR-1. They may play a role in regulating extracellular matrix degradation, cell adhesion, and migration by modulating the expression and activity of urokinase-type plasminogen activator and plasminogen activator inhibitor 1 (Pepper et al., Proc. Natl Acad. Sci. U.S.A. (1998), 95(20): 11709-11714).
VEGF-C最初是作为VEGFR-3/Flt-4的配体克隆的,它们主要由淋巴内皮细胞表达。在其完全作用形式中,VEGF-C还可以与KDR/VEGFR-2结合,并在刺激体外内皮细胞的增殖和移行以及在体内模型中的血管生成(Lymboussaki等,Am.J.Pathol.(1998),153(2):395-403;Witzenbichler等,Am.J.Pathol.(1998),153(2),381-394)。TheVEGF-C的转基因过分表达仅仅造成淋巴管的增殖和增大,而对血管没有影响。与VEGF不同,VEGF-C的表达不是由氧不足引起的(Ristimaki等,J.Biol.Chem.(1998),273(14),8413-8418)。VEGF-C was originally cloned as a ligand for VEGFR-3/Flt-4, which are mainly expressed by lymphatic endothelial cells. In its fully active form, VEGF-C can also combine with KDR/VEGFR-2, and stimulate proliferation and migration of endothelial cells in vitro and angiogenesis in in vivo models (Lymboussaki et al., Am.J.Pathol.(1998 ), 153(2):395-403; Witzenbichler et al., Am. J. Pathol. (1998), 153(2), 381-394). The transgenic overexpression of TheVEGF-C only caused the proliferation and enlargement of lymphatic vessels, but had no effect on blood vessels. Unlike VEGF, the expression of VEGF-C is not caused by hypoxia (Ristimaki et al., J. Biol. Chem. (1998), 273(14), 8413-8418).
最近发现的VEGF-D在结构上与VEGF-C非常类似。据报道,VEGF-D与至少两种VEGFRs、VEGFR-3/Flt-4和KDR/VEGFR-2结合并使其活化。它最初是作为成纤维细胞的c-fos诱导促细胞分裂剂克隆的,并且最突出的是在肺和皮肤的间质细胞中表达(Achen等,Proc.Natl.Acad.Sci.U.S.A.(1998),95(2),548-553,该文献引入本文作为参考)。Recently discovered VEGF-D is structurally very similar to VEGF-C. VEGF-D has been reported to bind to and activate at least two VEGFRs, VEGFR-3/Flt-4 and KDR/VEGFR-2. It was originally cloned as a c-fos-inducing mitogen for fibroblasts and is most prominently expressed in mesenchymal cells of the lung and skin (Achen et al., Proc. Natl. Acad. Sci. U.S.A. (1998) , 95(2), 548-553, which is incorporated herein by reference).
像VEGF一样,已经声称,当对皮肤组织注射时,VEGF-C和VEGF-D在Miles分析中引起血管透过性增加(PCT/US97/14696;W098/07832,Witzenbichler等,上文)。这些配体在其被表达的组织中在调节血管渗透性过高和内皮应答方面的生理学作用和重要性是不确定的。Like VEGF, VEGF-C and VEGF-D have been claimed to cause increased vascular permeability in the Miles assay when injected into skin tissue (PCT/US97/14696; W098/07832, Witzenbichler et al., supra). The physiological role and importance of these ligands in the regulation of vascular hyperpermeability and endothelial responses in the tissues in which they are expressed is uncertain.
近来报道了病毒编码的、新的血管内皮生长因子VEGF-E(NZ-7VEGF),它优先利用KDR/Flk-1受体并带有有效的有丝分裂活性、而非肝素结合区(Meyer等,EMBO J.(1999),18(2),363-374;Ogawa等,J.Biol.Chem.(1998),273(47),31273-31282.)。VEGF-E序列具有与哺乳动物VEGF约25%的同源性,并且由parapoxvirus Orf病毒(OV)编码。该parapoxvirus影响绵羊和山羊,并且偶而影响人,因而造成伴有血管生成的损害。VEGF-E是既无碱性区、又对肝素没有亲合性的约20kDa的二聚物,但是具有存在于所有哺乳动物VEGF中的特征胱氨酸结基元,并且出人意料地发现其具有与VEGF-A肝素结合VEGF165同种型类似的效能和生物活性,即两种因子均刺激组织因子(TF)的释放,体外培养的血管内皮细胞的增殖、趋化性和抽芽以及体内血管生成。与VEGF165一样,已发现,VEGF-E与VEGF受体-2(KDR)高度亲合地结合,导致受体自磷酸化以及游离细胞内Ca2+浓度二相性升高,同时与VEGF165相反,VEGF-E不与VEGF受体-1(Flt-1)结合。A virally encoded, novel vascular endothelial growth factor, VEGF-E (NZ-7VEGF), has recently been reported that preferentially utilizes the KDR/Flk-1 receptor and possesses potent mitotic activity rather than a heparin-binding domain (Meyer et al., EMBO J. (1999), 18(2), 363-374; Ogawa et al., J. Biol. Chem. (1998), 273(47), 31273-31282.). The VEGF-E sequence has approximately 25% homology to mammalian VEGF and is encoded by the parapoxvirus Orf virus (OV). The parapoxvirus affects sheep and goats, and occasionally humans, causing lesions with angiogenesis. VEGF-E is an approximately 20 kDa dimer that has neither a basic region nor an affinity for heparin, but has a characteristic cystine knot motif present in all mammalian VEGF, and was unexpectedly found to have the same VEGF-A heparin binds VEGF165 isoforms with similar potency and biological activity, i.e. both factors stimulate tissue factor (TF) release, proliferation, chemotaxis and sprouting of vascular endothelial cells cultured in vitro and angiogenesis in vivo. Like VEGF165, it has been found that VEGF-E binds with high affinity to VEGF receptor-2 (KDR), leading to autophosphorylation of the receptor and a diphasic increase in free intracellular Ca2+ concentration, while in contrast to VEGF165, VEGF-E Does not bind to VEGF receptor-1 (Flt-1).
根据现有的其他VEGF和VEGFRs同系物的发现以及配体和受体杂二聚作用的先例,该VEGF同系物的作用可以包括形式VEGF配体杂二聚物,和/或受体的杂二聚作用或与尚未发现的VEGFR结合(Witzenbichler等,上文)。同样,最新的报道提示neuropilin-1(Migdal等,上文)或VEGFR-3/Flt-4(Witzenbichler等,上文)或除KDR/VEGFR-2之外的受体可能与引起血管透过性有关(Stacker,S.A.,Vitali,A.,Domagala,T.,Nice,E.和Wilks,A.F.,″Angiogenesis andCancer″Conference,Amer.Assoc.Cancer Res.,Jan.1998,Orlando,FL;Williams,Diabetelogia 40:S118-120(1997))。迄今为止,尚未公开KDR在VEGF介导的血管渗透性过高中具有重要作用的直接证据。Based on the discovery of other VEGF and VEGFRs homologues and the precedent of ligand and receptor heterodimerization, the effect of this VEGF homologue may include the formation of heterodimers of VEGF ligands, and/or heterodimers of receptors. Polymerization or binding to as yet undiscovered VEGFR (Witzenbichler et al., supra). Likewise, recent reports suggest that neuropilin-1 (Migdal et al., supra) or VEGFR-3/Flt-4 (Witzenbichler et al., supra) or receptors other than KDR/VEGFR-2 may be involved in causing vascular permeability. Regarding (Stacker, S.A., Vitali, A., Domagala, T., Nice, E. and Wilks, A.F., "Angiogenesis and Cancer" Conference, Amer. Assoc. Cancer Res., Jan. 1998, Orlando, FL; Williams, Diabetelogia 40: S118-120 (1997)). To date, direct evidence that KDR has an important role in VEGF-mediated vascular hyperpermeability has not been published.
非受体酪氨酸激酶。非受体酪氨酸激酶代表缺乏细胞外和跨膜序列的细胞酶的集合。目前,已经识别出超过24种单个的非受体酪氨酸激酶,包括11个亚族(Src,Frk,Btk,Csk,Abl,Zap70,Fes/Fps,Fak,Jak,Ack和LIMK)。目前Src亚族非受体酪氨酸激酶由最大量的PTK组成,并且包括Src,Yes,Fyn,Lyn,Lck,Blk,Hck,Fgr和Yrk。Src亚族的酶已经与瘤形成和免疫应答联系在一起。在Bolen,1993,Oncogene 8:2025-2031中提供了有关非受体酪氨酸激酶的更详细讨论,该文献引入本文作为参考。Non-receptor tyrosine kinases. Non-receptor tyrosine kinases represent a collection of cellular enzymes that lack extracellular and transmembrane sequences. Currently, more than 24 individual non-receptor tyrosine kinases have been identified, including 11 subfamilies (Src, Frk, Btk, Csk, Abl, Zap70, Fes/Fps, Fak, Jak, Ack, and LIMK). The Src subfamily of non-receptor tyrosine kinases currently consists of the most abundant PTKs and includes Src, Yes, Fyn, Lyn, Lck, Blk, Hck, Fgr, and Yrk. Enzymes of the Src subfamily have been linked to neoplasia and immune responses. A more detailed discussion of non-receptor tyrosine kinases is provided in Bolen, 1993, Oncogene 8:2025-2031, which is incorporated herein by reference.
已经发现,许多酪氨酸激酶,无论是RTK或是非受体酪氨酸激酶都与很多致病状况包括癌、牛皮癣和其他过度增殖疾病或超免疫应答中涉及的细胞信号传导路径有关。A number of tyrosine kinases, whether RTKs or non-receptor tyrosine kinases, have been found to be involved in cell signaling pathways involved in many pathogenic conditions including cancer, psoriasis and other hyperproliferative diseases or hyperimmune responses.
调节PTKs化合物的开发。考虑到推测的PTKs对细胞增殖、与异常细胞增殖有关疾病和失调的控制、调节和调整的重要性,已经采用各种途径成功地识别了受体和非受体酪氨酸激酶″抑制剂″,所述途径包括使用突变体配体(U.S.A.4,966,849)、可溶性受体和抗体(WO94/10202;Kendall & Thomas,1994,Proc.Natl.Acad.Sci 90:10705-09;Kim等,1993,Nature 362:841-844)、RNA配体(Jellinek,等,Biochemistry 33:10450-56;Takano,等,1993,Mol.Bio.Cell 4:358A;Kinsella,等1992,Exp.Cell Res.199:56-62;Wright,等,1992,J.Cellular Phys.152:448-57)和酪氨酸激酶抑制剂(WO 94/03427;WO92/21660;WO 91/15495;WO 94/14808;U.S.P.5,330,992;Mariani,等,1994,Proc.Am.Assoc.Cancer Res.35:2268)。Development of compounds that regulate PTKs. Given the importance of putative PTKs for the control, regulation and regulation of cell proliferation, diseases and disorders associated with abnormal cell proliferation, various approaches have been employed to successfully identify receptor and non-receptor tyrosine kinase "inhibitors" , the approach involves the use of mutant ligands (U.S.A. 4,966,849), soluble receptors and antibodies (WO94/10202; Kendall & Thomas, 1994, Proc. Natl. Acad. Sci 90: 10705-09; Kim et al., 1993, Nature 362:841-844), RNA ligand (Jellinek, etc., Biochemistry 33:10450-56; Takano, etc., 1993, Mol.Bio.Cell 4:358A; Kinsella, etc. 1992, Exp.Cell Res.199:56 -62; Wright, et al., 1992, J.Cellular Phys.152:448-57) and tyrosine kinase inhibitors (WO 94/03427; WO 92/21660; WO 91/15495; WO 94/14808; U.S.P. 5,330,992; Mariani, et al., 1994, Proc. Am. Assoc. Cancer Res. 35:2268).
最近,已经成功地识别了用作酪氨酸激酶抑制剂的小分子。例如,通常将双单环、双环或杂环芳基化合物(PCT WO 92/20642)和亚乙烯基氮杂吲哚衍生物(PCT WO 94/14808)描述为酪氨酸激酶抑制剂。还描述了苯乙烯基化合物(U.S.P.5,217,999)、苯乙烯基-取代的吡啶基化合物(U.S.P.5,302,606)、某些喹唑啉衍生物(EP申请0 566 266 A1;ExpertOpin.Ther.Pat.(1998),8(4):475-478)、硒基吲哚和硒化物(PCT WO94/03427)、三环多羟基化合物(PCT WO 92/21660)和苄基膦酸化合物(PCT WO 91/15495)用作酪氨酸激酶抑制剂化合物,用于治疗癌。已有描述,苯氨基噌啉(PCT WO97/34876)和喹唑啉衍生物化合物(PCTWO97/22596;PCT WO97/42187)用作血管生成和血管透过性抑制剂。Recently, small molecules have been successfully identified as tyrosine kinase inhibitors. For example, bis monocyclic, bicyclic or heterocyclic aryl compounds (PCT WO 92/20642) and vinylidene azaindole derivatives (PCT WO 94/14808) are generally described as tyrosine kinase inhibitors. Also described styryl compounds (U.S.P. 5,217,999), styryl-substituted pyridyl compounds (U.S.P. 5,302,606), certain quinazoline derivatives (EP application 0 566 266 A1; Expert Opin.Ther.Pat. (1998) , 8(4):475-478), selenoindoles and selenides (PCT WO94/03427), tricyclic polyols (PCT WO 92/21660) and benzylphosphonic acid compounds (PCT WO 91/15495) Used as a tyrosine kinase inhibitor compound for the treatment of cancer. Anilinocinnoline (PCT WO97/34876) and quinazoline derivative compounds (PCT WO97/22596; PCT WO97/42187) have been described as inhibitors of angiogenesis and vascular permeability.
此外,已经成功地识别了用作丝氨酸/苏氨酸激酶抑制剂的小分子。例如,已经描述了二(吲哚基马来酰亚胺)化合物抑制特定PKC丝氨酸/苏氨酸激酶同种型,该同种型的信号传导功能在VEGF-有关的疾病中与改变的血管透过性有关(PCT WO97/40830;PCTWO97/40831)。In addition, small molecules have been successfully identified as inhibitors of serine/threonine kinases. For example, bis(indolylmaleimide) compounds have been described to inhibit specific PKC serine/threonine kinase isoforms whose signaling functions are linked to altered vascular permeability in VEGF-related diseases. Transsexual (PCT WO97/40830; PCTWO97/40831).
Plk-1激酶抑制剂Plk-1 kinase inhibitor
Plk-1是丝氨酸/苏氨酸激酶,它是重要的细胞周期进行的调节剂。它在有丝分裂纺锤体装置的装配和动力功能中起着决定性的作用。Plk-1和相关的激酶还显示出与其他细胞周期调节剂如细胞周期蛋白依赖性激酶的活化和灭活密切相关。高水平Plk-1表达与细胞增殖活性有关。它经常可见于各种起源的恶性肿瘤中。Plk-1 is a serine/threonine kinase that is an important regulator of cell cycle progression. It plays a decisive role in the assembly and power function of the mitotic spindle apparatus. Plk-1 and related kinases have also been shown to be closely associated with the activation and inactivation of other cell cycle regulators such as cyclin-dependent kinases. High levels of Plk-1 expression are associated with cell proliferation activity. It is frequently seen in malignancies of various origins.
预期Plk-1抑制剂通过中断与有丝分裂纺锤体和不适当活化的细胞周期蛋白依赖性激酶的过程而阻断癌细胞增殖。Plk-1 inhibitors are expected to block cancer cell proliferation by interrupting processes associated with the mitotic spindle and inappropriately activated cyclin-dependent kinases.
Cdc2/细胞周期蛋白B激酶抑制剂(Cdc2也称作cdkl) Cdc2/cyclin B kinase inhibitor (Cdc2 also known as cdkl)
Cdc2/细胞周期蛋白B是另一种丝氨酸/苏氨酸激酶,它属于细胞周期蛋白依赖性激酶(cdks)。这些酶涉及细胞周期进行各期之间的决定性的转变。据信不受控制的细胞增殖是癌的标志,它取决于在这些细胞中升高的cdk活性。通过cdc2/细胞周期蛋白B激酶抑制剂抑制癌细胞中升高的cdk活性,可以抑制增殖,并且可以恢复细胞周期进行的正常控制。Cdc2/cyclin B is another serine/threonine kinase that belongs to the cyclin-dependent kinases (cdks). These enzymes are involved in the decisive transitions between the progressive phases of the cell cycle. Uncontrolled cell proliferation is believed to be a hallmark of cancer and depends on elevated cdk activity in these cells. Inhibition of elevated cdk activity in cancer cells by cdc2/cyclin B kinase inhibitors inhibits proliferation and restores normal control of cell cycle progression.
因此需要识别有效的小化合物,这些化合物通过调控受体和非受体酪氨酸和丝氨酸/苏氨酸激酶以调节和调整异常或不适当的细胞增殖、分化或代谢,而专门抑制信号传导和细胞增殖。尤其是,识别方法和专门抑制酪氨酸激酶功能的化合物是有益的,所述酪氨酸激酶对于血管生成过程或血管渗透性过高形成从而导致水肿、腹水、渗漏、渗出物和大分子外渗和基质沉积以及相关疾病是必需的。There is therefore a need to identify potent small compounds that specifically inhibit signaling and Cell Proliferation. In particular, it would be beneficial to identify methods and compounds that specifically inhibit the function of tyrosine kinases that are critical for the angiogenic process or the formation of vascular hyperpermeability leading to edema, ascites, leaks, exudates and large Molecular extravasation and matrix deposition and associated diseases are required.
发明内容Contents of the invention
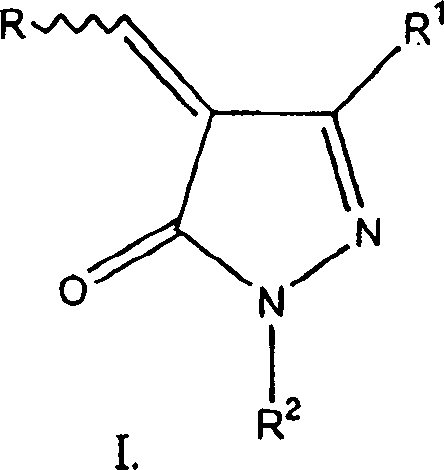
本发明涉及下式化合物及其生理上可接受的盐The present invention relates to compounds of the following formula and physiologically acceptable salts thereof
其中R是取代或未取代的脂族、芳族、杂环或芳烷基,并且R2是氢、低级烷基或芳基。R1是氢或-A-Z,其中A是-(CH2)n-、-(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;并且Z是H、低级烷基、芳烷基、三卤代甲基、R3CO-、三卤代甲基羰基、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3O-或选自下列一组的环系:C3-C6环烷烃、苯、吡咯、异噁唑、异噻唑、吲哚、吡啶、吡嗪、嘧啶、噻吩、咪唑、呋喃、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑、苯并噻唑、四氢呋喃、三嗪及其可应用的N-氧化物,这些环系可以任选地被一个或多个卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳基、-CN、-C(O)NR4R5或-NR4R5取代。每处出现的R3独立地选自取代或未取代的:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基。每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;R4和R5可以任选地与和其相连的氮结合在一起表示吗啉代、吡咯烷子基(pyrrolidino)、哌啶子基、咪唑-1-基、哌嗪子基(piperazino)、硫吗啉代、吖庚因子基(azepino)或全氢化-1,4-二氮杂环庚三烯(diazepin)-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;数字下标n是0-3的整数。wherein R is substituted or unsubstituted aliphatic, aromatic, heterocyclic or aralkyl, and R is hydrogen, lower alkyl or aryl. R 1 is hydrogen or -AZ, where A is -(CH 2 ) n -, -(CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)- or -(CH 2 ) n S(O) 2 -; and Z is H, lower alkyl, aralkyl, trihalomethyl, R 3 CO-, trihalomethyl Carbonyl, R 3 OC(O)-, -NR 4 R 5 , -C(O)NR 4 R 5 , R 3 O- or a ring system selected from the following group: C 3 -C 6 cycloalkane, benzene, Pyrrole, isoxazole, isothiazole, indole, pyridine, pyrazine, pyrimidine, thiophene, imidazole, furan, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole, benzothiazole, tetrahydrofuran, Triazines and applicable N-oxides, these ring systems can optionally be replaced by one or more halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O) -, trihalomethyl, nitro, aryl, -CN, -C(O)NR 4 R 5 or -NR 4 R 5 substituted. Each occurrence of R3 is independently selected from substituted or unsubstituted: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl radical, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl. Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 ) cycloalkyl-alkyl and heterocyclyl-alkyl; R 4 and R 5 can optionally be combined with the nitrogen connected to it to represent morpholino, pyrrolidino ( pyrrolidino), piperidino, imidazol-1-yl, piperazino (piperazino), thiomorpholino, azepino (azepino) or perhydrogenated-1,4-diazepane ( diazepin)-1-yl, wherein each group is optionally substituted by one or more groups selected from the group consisting of lower alkyl, hydroxyl, lower alkoxy lower alkyl, aromatic group, ( C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl; the numerical subscript n is an integer from 0-3.
对于R来说,合适的取代基包括卤素、低级烷基、R3O-、羟基、HOC(O)-、R3OC(O)-、R3OC(O)R6、R3OR6-、三卤代甲基、三卤代甲基羰基、硝基、-C(O)NR4R5、-NR4R5、R3CO-、-(CH2)n-R7、-C(O)(CH2)n-R7、-C(O)-(CH2)n-C(O)-R7、-O(CH2)nR7、-C(O)NR4(CH2)nR7、C(O)O(CH2)nR7、-OC(O)(CH2)nR7、-NR4C(O)(CH2)nR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N[R6-R7]2、-R6C(O)NR4(CH2)nR7、-R6C(O)O(CH2)nR7、-R6OC(O)(CH2)nR7、R6NR4C(O)(CH2)nR7、-R6CH[C(O)OR4][NR5C(O)R4]或取代的芳基或芳烷基,其中取代基选自卤素、三卤代甲基、羟基、-NR4R5、硝基、-CONR4R5、低级烷基、R3O-、C(O)OR4或-OC(O)R3。R6是低级烷基或芳基。R7是烷氧基、卤代烷基、低级烷基哌嗪、羟基、R3O-、R3C(O)-或-NR4R5。Suitable substituents for R include halogen, lower alkyl, R 3 O-, hydroxyl, HOC(O)-, R 3 OC(O)-, R 3 OC(O)R 6 , R 3 OR 6 -, Trihalomethyl, Trihalomethylcarbonyl, Nitro, -C(O)NR 4 R 5 , -NR 4 R 5 , R 3 CO-, -(CH 2 ) n -R 7 , - C(O)(CH 2 ) n -R 7 , -C(O)-(CH 2 ) n -C(O)-R 7 , -O(CH 2 ) n R 7 , -C(O)NR 4 (CH 2 ) n R 7 , C(O)O(CH 2 ) n R 7 , -OC(O)(CH 2 ) n R 7 , -NR 4 C(O)(CH 2 ) n R 7 , - R 6 NR 4 R 5 , -R 6 N(R 4 )-R 6 -R 7 , -R 6 N[R 6 -R 7 ] 2 , -R 6 C(O)NR 4 (CH 2 ) n R 7 , -R 6 C(O)O(CH 2 ) n R 7 , -R 6 OC(O)(CH 2 ) n R 7 , R 6 NR 4 C(O)(CH 2 ) n R 7 , - R 6 CH[C(O)OR 4 ][NR 5 C(O)R 4 ] or substituted aryl or aralkyl, wherein the substituents are selected from halogen, trihalomethyl, hydroxyl, -NR 4 R 5. Nitro, -CONR 4 R 5 , lower alkyl, R 3 O-, C(O)OR 4 or -OC(O)R 3 . R 6 is lower alkyl or aryl. R 7 is alkoxy, haloalkyl, lower alkylpiperazine, hydroxyl, R 3 O-, R 3 C(O)- or -NR 4 R 5 .
对于R3、R4和R5来说,合适的取代基包括一个或多个选自下列的基团:卤素、低级烷基、羟基、低级烷氧基、羧基、低级烷基酯、三卤代甲基优选三氟甲基、硝基、苯基、苯基低级烷基、(C3-C6)环烷基、(C3-C6)环烷基-烷基、CN、氨基、烷氨基、二烷氨基、-C(O)NH2、-C(O)NH(烷基)和-C(O)N(烷基)2。Suitable substituents for R 3 , R 4 and R 5 include one or more groups selected from the group consisting of halogen, lower alkyl, hydroxyl, lower alkoxy, carboxyl, lower alkyl ester, trihalogen Substituted methyl is preferably trifluoromethyl, nitro, phenyl, phenyl-lower alkyl, (C 3 -C 6 )cycloalkyl, (C 3 -C 6 )cycloalkyl-alkyl, CN, amino, Alkylamino, dialkylamino, -C(O)NH2, -C( O )NH(alkyl) and -C(O)N(alkyl) 2 .
脂族基团包括直链或支链C1-C18烃或环C3-C18烃,它们是完全饱和的,或者含有一个或多个不饱和单元。低级烷基是完全饱和的直链或支链C1-C6烃或C3-C6环烃。Aliphatic groups include straight or branched C 1 -C 18 hydrocarbons or cyclic C 3 -C 18 hydrocarbons which are fully saturated or contain one or more units of unsaturation. Lower alkyl is fully saturated straight or branched C 1 -C 6 hydrocarbon or C 3 -C 6 cyclic hydrocarbon.
本文所用的芳族基团一词包括碳环系(例如苄基和亚肉桂基)以及稠合多环芳环系(例如萘基)。此外,芳族基团包括杂芳环系(例如吡啶、噻吩、呋喃、吡咯、咪唑、噁唑、噻唑、吡唑、三唑、嘧啶和吡嗪)以及杂芳基环系,其中碳环芳环、碳环非芳环或杂芳环与一个或多个其他的杂芳环(例如苯并咪唑、苯并噻唑、吲哚、四氢吲哚、氮杂吲哚、吲唑、异噁唑、异噻唑、咪唑和喹啉)稠合。本文所用的芳基一词是指具有5或6个原子的芳族基团。芳烷基是通过具有1-6个碳原子的脂族基团与化合物相连的芳族取代基。本文所用的杂环基一词是指单-、二-或三-环杂环基,它们不是芳族的或者具有一定的不饱和程度,该不饱和不构成芳族基团。The term aromatic as used herein includes carbocyclic systems such as benzyl and cinnamylidene as well as fused polycyclic aromatic ring systems such as naphthyl. In addition, aromatic groups include heteroaryl ring systems (such as pyridine, thiophene, furan, pyrrole, imidazole, oxazole, thiazole, pyrazole, triazole, pyrimidine, and pyrazine) and heteroaryl ring systems in which the carbocyclic aromatic ring, carbocyclic non-aromatic ring or heteroaromatic ring with one or more other heteroaromatic rings (such as benzimidazole, benzothiazole, indole, tetrahydroindole, azaindole, indazole, isoxazole , isothiazole, imidazole and quinoline) fused. The term aryl as used herein refers to an aromatic group having 5 or 6 atoms. Aralkyl is an aromatic substituent attached to a compound through an aliphatic group having 1-6 carbon atoms. The term heterocyclyl as used herein refers to mono-, bi- or tri-cyclic heterocyclyl groups which are not aromatic or which have a degree of unsaturation which does not constitute an aromatic group.
互变异构体、立体异构体、对映体以及这些化合物的混合物包括在本发明中。这些化合物的可药用加成盐也包括在本发明中。Tautomers, stereoisomers, enantiomers and mixtures of these compounds are included in the present invention. Pharmaceutically acceptable addition salts of these compounds are also included in the present invention.
具体实施方式Detailed ways
在一个实施方案中,R2是氢,并且R优选为取代的吲哚或取代或未取代的咪唑、1,2,3-三唑、1,2,4-三唑、苯并咪唑、吡咯、吡唑、4,5,6,7-四氢吲哚、苯并吲哚、氮杂吲哚、吲唑、吡啶、喹啉、嘧啶、苯或哌嗪。In one embodiment R is hydrogen and R is preferably substituted indole or substituted or unsubstituted imidazole, 1,2,3-triazole, 1,2,4-triazole, benzimidazole, pyrrole , pyrazole, 4,5,6,7-tetrahydroindole, benzindole, azaindole, indazole, pyridine, quinoline, pyrimidine, benzene or piperazine.
在一个优选的实施方案中,R2是氢,n是0,并且Z是环丙基、3-吡啶基或吡嗪基。在另一个优选的实施方案中,R2是氢,A是-O-,n是0,并且Z是乙基、丙基或异丙基。在又一个优选的实施方案中,R2是氢,A是CH2,n是2,并且Z是任选地被卤素、三卤代甲基、羟基、-NR4R5、硝基、-C(O)NR4R5、低级烷基、R3O-、-C(O)OR4或-OC(O)R3取代的苯。In a preferred embodiment, R is hydrogen, n is 0, and Z is cyclopropyl, 3-pyridyl or pyrazinyl. In another preferred embodiment, R2 is hydrogen, A is -O-, n is 0, and Z is ethyl, propyl or isopropyl. In yet another preferred embodiment, R 2 is hydrogen, A is CH 2 , n is 2, and Z is optionally halogen, trihalomethyl, hydroxyl, -NR 4 R 5 , nitro, - C(O)NR 4 R 5 , lower alkyl, R 3 O-, -C(O)OR 4 or -OC(O)R 3 substituted benzene.
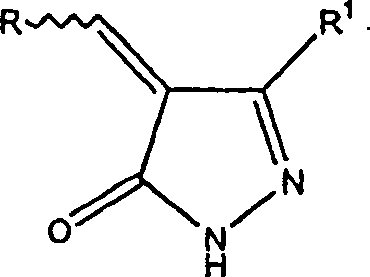
在一个优选的实施方案中,本发明涉及下式表示的化合物的及其生理上可接受的盐:In a preferred embodiment, the present invention relates to compounds represented by the formula: and physiologically acceptable salts thereof:
其中:in:
R选自取代或未取代的:吲哚、咪唑、1,2,3-三唑、1,2,4-三唑、苯并咪唑、4,5,6,7-四氢吲哚、苯并吲哚、氮杂吲哚、吲唑、吡啶、喹啉、嘧啶、苯、吡嗪、吡咯、吡唑、噁唑和噻唑;R is selected from substituted or unsubstituted: indole, imidazole, 1,2,3-triazole, 1,2,4-triazole, benzimidazole, 4,5,6,7-tetrahydroindole, benzene Indole, azaindole, indazole, pyridine, quinoline, pyrimidine, benzene, pyrazine, pyrrole, pyrazole, oxazole and thiazole;
R1是氢或-A-Z;R 1 is hydrogen or -AZ;
A是-(CH2)n-、-(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;A is -(CH 2 ) n -, -(CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)-, or - (CH 2 ) n S(O) 2 -;
Z是-H、低级烷基、芳烷基、三卤代甲基、三卤代甲基羰基、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3CO-、R3O-或选自下列一组的环系:C3-C6环烷烃、异噁唑、异噻唑、咪唑、苯、吡咯、吲哚、吡啶、吡嗪、嘧啶、苯并噻唑、四氢呋喃、噻吩、咪唑、呋喃、三嗪、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑及其N-氧化物,其中所述环系可以任选地被一个或多个选自下列一组的基团取代:卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基、杂环基-烷基、-CN、-C(O)NR4R5或-NR4R5;Z is -H, lower alkyl, aralkyl, trihalomethyl, trihalomethylcarbonyl, R 3 OC(O)-, -NR 4 R 5 , -C(O)NR 4 R 5 , R 3 CO-, R 3 O- or a ring system selected from the following group: C 3 -C 6 cycloalkane, isoxazole, isothiazole, imidazole, benzene, pyrrole, indole, pyridine, pyrazine, pyrimidine, benzothiazole, tetrahydrofuran, thiophene, imidazole, furan, triazine, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole and their N-oxides, wherein the ring system can optionally be Substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O)-, trihalomethyl, nitro , Aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl, heterocyclyl-alkyl, -CN, - C(O)NR 4 R 5 or -NR 4 R 5 ;
每处出现的R3彼此独立地选自取代或未取代的:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R 3 is independently selected from substituted or unsubstituted: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aryl Alkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
任选地,R4和R5与和其相连的氮结合在一起表示吗啉代、吡咯烷子基、哌啶子基、咪唑-1-基、哌嗪子基、硫吗啉代、吖庚因子基或全氢化-1,4-二氮杂环庚三烯-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;并且Optionally, R 4 and R 5 combined with the nitrogen attached to them represent morpholino, pyrrolidino, piperidino, imidazol-1-yl, piperazino, thiomorpholino, acridino Heptinyl or perhydro-1,4-diazepan-1-yl, each of which is optionally substituted by one or more groups selected from the group consisting of lower alkyl, Hydroxy, lower alkoxy lower alkyl, aromatic, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocycle base-alkyl; and
n是0-3的整数;n is an integer of 0-3;
条件是当R是未取代的吲哚-3-基时,R1不是-NH2。在另一个优选的实施方案中,本发明涉及下列结构式表示的化合物及其生理上可接受的盐:With the proviso that when R is unsubstituted indol-3-yl, R1 is other than -NH2 . In another preferred embodiment, the present invention relates to compounds represented by the following structural formula and physiologically acceptable salts thereof:
其中:in:
R选自取代或未取代的:吲哚、咪唑、1,2,3-三唑、1,2,4-三唑、苯并咪唑、4,5,6,7-四氢吲哚、苯并吲哚、氮杂吲哚、吲唑、吡啶、喹啉、嘧啶、苯、吡嗪、吡咯、吡唑、噁唑和噻唑;R is selected from substituted or unsubstituted: indole, imidazole, 1,2,3-triazole, 1,2,4-triazole, benzimidazole, 4,5,6,7-tetrahydroindole, benzene Indole, azaindole, indazole, pyridine, quinoline, pyrimidine, benzene, pyrazine, pyrrole, pyrazole, oxazole and thiazole;
R1是氢或-A-Z;R 1 is hydrogen or -AZ;
A是-(CH2)n-、(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;A is -(CH 2 ) n -, (CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)- or -( CH 2 ) n S(O) 2 -;
Z是-H、低级烷基、芳烷基、三卤代甲基、三卤代甲基羰基、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3CO-、R3O-或选自下列一组的环系:C3-C6环烷烃、异噁唑、异噻唑、咪唑、苯、吡咯、吲哚、吡啶、吡嗪、嘧啶、苯并噻唑、四氢呋喃、噻吩、咪唑、呋喃、三嗪、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑及其N-氧化物,其中所述环系可以任选地被一个或多个选自下列一组的基团取代:卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基、杂环基-烷基、-CN、-C(O)NR4R5或-NR4R5;Z is -H, lower alkyl, aralkyl, trihalomethyl, trihalomethylcarbonyl, R 3 OC(O)-, -NR 4 R 5 , -C(O)NR 4 R 5 , R 3 CO-, R 3 O- or a ring system selected from the following group: C 3 -C 6 cycloalkane, isoxazole, isothiazole, imidazole, benzene, pyrrole, indole, pyridine, pyrazine, pyrimidine, benzothiazole, tetrahydrofuran, thiophene, imidazole, furan, triazine, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole and their N-oxides, wherein the ring system can optionally be Substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O)-, trihalomethyl, nitro , Aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl, heterocyclyl-alkyl, -CN, - C(O)NR 4 R 5 or -NR 4 R 5 ;
每处出现的R3彼此独立地选自取代或未取代的:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R 3 is independently selected from substituted or unsubstituted: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aryl Alkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;任选地,R4和R5与和其相连的氮结合在一起表示吗啉代、吡咯烷子基、哌啶子基、咪唑-1-基、哌嗪子基、硫吗啉代、吖庚因子基或全氢化-1,4-二氮杂环庚三烯-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;并且Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl; Optionally, R 4 and R 5 combined with the nitrogen attached to them represent morpholino, pyrrolidino, Piperidino group, imidazol-1-yl, piperazino group, thiomorpholino, azepin group or perhydrogenated-1,4-diazepan-1-yl, each of which Optionally substituted with one or more groups selected from the group consisting of lower alkyl, hydroxy, lower alkoxy lower alkyl, aromatic, (C 3 -C 6 )cycloalkyl, heterocycle radical, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl; and
n是0-3的整数;n is an integer of 0-3;
条件是:requirement is:
当R是未取代的吲哚-3-基时,R1不是-NH2;和When R is unsubstituted indol-3-yl, R 1 is not -NH 2 ; and
当R是取代或未取代的苯或未取代的咪唑、1,2,3-三唑、1,2,4-三唑、吡啶、嘧啶、吡嗪、吡咯、吡唑、噁唑或噻唑时,R1不是低级烷基、(C3-C6)环烷基、苯或-C(O)NR4R5,其中R4和R5彼此独立地为H、低级烷基或碳环芳基。When R is substituted or unsubstituted benzene or unsubstituted imidazole, 1,2,3-triazole, 1,2,4-triazole, pyridine, pyrimidine, pyrazine, pyrrole, pyrazole, oxazole or thiazole , R 1 is not lower alkyl, (C 3 -C 6 )cycloalkyl, benzene or -C(O)NR 4 R 5 , wherein R 4 and R 5 are independently H, lower alkyl or carbocyclic aromatic base.
在另一个优选的实施方案中,本发明涉及下列结构式表示的化合物及其生理上可接受的盐:In another preferred embodiment, the present invention relates to compounds represented by the following structural formula and physiologically acceptable salts thereof:
其中:in:
R选自取代或未取代的:吲哚、咪唑、1,2,3-三唑、1,2,4-三唑、苯并咪唑、4,5,6,7-四氢吲哚、苯并吲哚、氮杂吲哚、吲唑、吡啶、喹啉、嘧啶、苯、吡嗪、吡咯、吡唑、噁唑和噻唑;R is selected from substituted or unsubstituted: indole, imidazole, 1,2,3-triazole, 1,2,4-triazole, benzimidazole, 4,5,6,7-tetrahydroindole, benzene Indole, azaindole, indazole, pyridine, quinoline, pyrimidine, benzene, pyrazine, pyrrole, pyrazole, oxazole and thiazole;
R1是氢或-A-Z;R 1 is hydrogen or -AZ;
A是-(CH2)n-、(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;A is -(CH 2 ) n -, (CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)- or -( CH 2 ) n S(O) 2 -;
Z是-H、低级烷基、芳烷基、三卤代甲基、三卤代甲基羰基、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3CO-、R3O-或选自下列一组的环系:C3-C6环烷烃、异噁唑、异噻唑、咪唑、苯、吡咯、吲哚、吡啶、吡嗪、嘧啶、苯并噻唑、四氢呋喃、噻吩、咪唑、呋喃、三嗪、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑及其N-氧化物,其中所述环系可以任选地被一个或多个选自下列一组的基团取代:卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基、杂环基-烷基、-CN、-C(O)NR4R5或-NR4R5;Z is -H, lower alkyl, aralkyl, trihalomethyl, trihalomethylcarbonyl, R 3 OC(O)-, -NR 4 R 5 , -C(O)NR 4 R 5 , R 3 CO-, R 3 O- or a ring system selected from the following group: C 3 -C 6 cycloalkane, isoxazole, isothiazole, imidazole, benzene, pyrrole, indole, pyridine, pyrazine, pyrimidine, benzothiazole, tetrahydrofuran, thiophene, imidazole, furan, triazine, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole and their N-oxides, wherein the ring system can optionally be Substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O)-, trihalomethyl, nitro , Aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl, heterocyclyl-alkyl, -CN, - C(O)NR 4 R 5 or -NR 4 R 5 ;
每处出现的R3彼此独立地选自取代或未取代的:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R 3 is independently selected from substituted or unsubstituted: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aryl Alkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
任选地,R4和R5与和其相连的氮结合在一起表示吗啉代、吡咯烷子基、哌啶子基、咪唑-1-基、哌嗪子基、硫吗啉代、吖庚因子基或全氢化-1,4-二氮杂环庚三烯-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;并且Optionally, R 4 and R 5 combined with the nitrogen attached to them represent morpholino, pyrrolidino, piperidino, imidazol-1-yl, piperazino, thiomorpholino, acridino Heptinyl or perhydro-1,4-diazepan-1-yl, each of which is optionally substituted by one or more groups selected from the group consisting of lower alkyl, Hydroxy, lower alkoxy lower alkyl, aromatic, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocycle base-alkyl; and
n是0-3的整数;n is an integer of 0-3;
条件是:requirement is:
a)R不是吡咯-3-基或被烷基或碳环芳基取代的吡咯-3-基;a) R is not pyrrol-3-yl or pyrrol-3-yl substituted by alkyl or carbocyclic aryl;
b)R不是吲哚-3-基或被烷基或碳环芳基取代的吲哚-3-基;和b) R is not indol-3-yl or indol-3-yl substituted by alkyl or carbocyclic aryl; and
c)R不是氮杂吲哚或在2-位被烷基或碳环芳基取代的氮杂吲哚。c) R is not an azaindole or an azaindole substituted at the 2-position with an alkyl or carbocyclic aryl.
在另一个优选的实施方案中,本发明涉及下列结构式表示的化合物及其生理上可接受的盐:In another preferred embodiment, the present invention relates to compounds represented by the following structural formula and physiologically acceptable salts thereof:
其中:in:
R是取代的吡咯或者选自取代或未取代的:吲哚、咪唑、1,2,3-三唑、1,2,4-三唑、苯并咪唑、4,5,6,7-四氢吲哚、苯并吲哚、氮杂吲哚、吲唑、吡啶、喹啉、嘧啶、苯、吡嗪、吡唑、噁唑和噻唑;R is substituted pyrrole or is selected from substituted or unsubstituted: indole, imidazole, 1,2,3-triazole, 1,2,4-triazole, benzimidazole, 4,5,6,7-tetra Indoline, benzoindole, azaindole, indazole, pyridine, quinoline, pyrimidine, benzene, pyrazine, pyrazole, oxazole and thiazole;
R1是氢或-A-Z;R 1 is hydrogen or -AZ;
A是-(CH2)n-、(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;A is -(CH 2 ) n -, (CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)- or -( CH 2 ) n S(O) 2 -;
Z是-H、低级烷基、芳烷基、三卤代甲基、三卤代甲基羰基、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3CO-、R3O-或选自下列一组的环系:C3-C6环烷烃、异噁唑、异噻唑、咪唑、苯、吡咯、吲哚、吡啶、吡嗪、嘧啶、苯并噻唑、四氢呋喃、噻吩、咪唑、呋喃、三嗪、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑及其N-氧化物,其中所述环系可以任选地被一个或多个选自下列一组的基团取代:卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基、杂环基-烷基、-CN、-C(O)NR4R5或-NR4R5;Z is -H, lower alkyl, aralkyl, trihalomethyl, trihalomethylcarbonyl, R 3 OC(O)-, -NR 4 R 5 , -C(O)NR 4 R 5 , R 3 CO-, R 3 O- or a ring system selected from the following group: C 3 -C 6 cycloalkane, isoxazole, isothiazole, imidazole, benzene, pyrrole, indole, pyridine, pyrazine, pyrimidine, benzothiazole, tetrahydrofuran, thiophene, imidazole, furan, triazine, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole and their N-oxides, wherein the ring system can optionally be Substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O)-, trihalomethyl, nitro , Aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl, heterocyclyl-alkyl, -CN, - C(O)NR 4 R 5 or -NR 4 R 5 ;
每处出现的R3彼此独立地选自:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R 3 is independently selected from: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 ) cycloalkyl-alkyl and heterocyclyl-alkyl;
每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
任选地,R4和R5与和其相连的氮结合在一起表示吗啉代、吡咯烷子基、哌啶子基、咪唑-1-基、哌嗪子基、硫吗啉代、吖庚因子基或全氢化-1,4-二氮杂环庚三烯-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;并且Optionally, R 4 and R 5 combined with the nitrogen attached to them represent morpholino, pyrrolidino, piperidino, imidazol-1-yl, piperazino, thiomorpholino, acridino Heptinyl or perhydro-1,4-diazepan-1-yl, each of which is optionally substituted by one or more groups selected from the group consisting of lower alkyl, Hydroxy, lower alkoxy lower alkyl, aromatic, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocycle base-alkyl; and
n是0-3的整数;n is an integer of 0-3;
条件是:requirement is:
a)当R是取代的吡咯时,该吡咯被R3OC(O)R6、R3OR6-、三卤代甲基、三卤代甲基羰基、-(CH2)p-R7、-O(CH2)pR7、-C(O)N(R4)(CH2)pR7、-C(O)O(CH2)pR7、-OC(O)(CH2)pR7、-N(R4)C(O)(CH2)pR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、R6C(O)N(R4)(CH2)pR7、-R6C(O)O(CH2)pR7、-R6OC(O)(CH2)pR7、-R6N(R4)C(O)(CH2)pR7、-R6CH(C(O)OR4)(N(R4)C(O)R4)、取代的芳基或芳烷基取代,其中每处出现的R6独立地为低级烷基或芳基;a) When R is a substituted pyrrole, the pyrrole is replaced by R 3 OC(O)R 6 , R 3 OR 6 -, trihalomethyl, trihalomethylcarbonyl, -(CH 2 ) p -R 7 , -O(CH 2 ) p R 7 , -C(O)N(R 4 )(CH 2 ) p R 7 , -C(O)O(CH 2 ) p R 7 , -OC(O)(CH 2 ) p R 7 , -N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 NR 4 R 5 , -R 6 N(R 4 )-R 6 -R 7 , -R 6 N(R 6 -R 7 ) 2 , R 6 C(O)N(R 4 )(CH 2 ) p R 7 , -R 6 C(O)O(CH 2 ) p R 7 , -R 6 OC( O)(CH 2 ) p R 7 , -R 6 N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 CH(C(O)OR 4 )(N(R 4 )C( O) R 4 ), substituted aryl or aralkyl substituted, wherein each occurrence of R 6 is independently lower alkyl or aryl;
每处出现的R7独立地为烷氧基、卤代烷基、低级烷基哌嗪、羟基、R3O-、R3C(O)-或NR4R3;和Each occurrence of R is independently alkoxy, haloalkyl, lower alkylpiperazine, hydroxyl, R3O- , R3C (O)-, or NR4R3 ; and
p是1-3的整数;p is an integer of 1-3;
b)当R是吲哚-3-基时,该吲哚-3-基被下列基团取代:R3OC(O)R6、R3OR6、三卤代甲基、三卤代甲基羰基、-(CH2)p-R7、-O(CH2)pR7、-C(O)N(R4)(CH2)pR7、-C(O)O(CH2)pR7、-OC(O)(CH2)pR7、-N(R4)C(O)(CH2)pR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、R6C(O)N(R4)(CH2)pR7、-R6C(O)O(CH2)pR7、-R6OC(O)(CH2)pR7、-R6N(R4)C(O)(CH2)pR7、-R6CH(C(O)OR4)(N(R4)C(O)R4)、取代的芳基或芳烷基,和b) When R is indol-3-yl, the indol-3-yl is substituted by the following groups: R 3 OC(O)R 6 , R 3 OR 6 , trihalomethyl, trihaloform Carbonyl, -(CH 2 ) p -R 7 , -O(CH 2 ) p R 7 , -C(O)N(R 4 )(CH 2 ) p R 7 , -C(O)O(CH 2 ) p R 7 , -OC(O)(CH 2 ) p R 7 , -N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 NR 4 R 5 , -R 6 N(R 4 )-R 6 -R 7 , -R 6 N(R 6 -R 7 ) 2 , R 6 C(O)N(R 4 )(CH 2 ) p R 7 , -R 6 C(O)O( CH 2 ) p R 7 , -R 6 OC(O)(CH 2 ) p R 7 , -R 6 N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 CH(C(O )OR 4 )(N(R 4 )C(O)R 4 ), substituted aryl or aralkyl, and
c)当R是吡唑-3-基时,该吡唑-3-基被下列基团取代:R3OC(O)R6、R3OR6、三卤代甲基、三卤代甲基羰基、-(CH2)p-R7、-O(CH2)pR7、-C(O)N(R4)(CH2)pR7、-C(O)O(CH2)pR7、-OC(O)(CH2)pR7、-N(R4)C(O)(CH2)PR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、R6C(O)N(R4)(CH2)pR7、-R6C(O)O(CH2)pR7、-R6OC(O)(CH2)pR7、-R6N(R4)C(O)(CH2)pR7、-R6CH(C(O)OR4)(N(R4)C(O)R4)、取代的芳基或芳烷基。c) When R is pyrazol-3-yl, the pyrazol-3-yl is substituted by the following groups: R 3 OC(O)R 6 , R 3 OR 6 , trihalomethyl, trihaloform Carbonyl, -(CH 2 ) p -R 7 , -O(CH 2 ) p R 7 , -C(O)N(R 4 )(CH 2 ) p R 7 , -C(O)O(CH 2 ) p R 7 , -OC(O)(CH 2 ) p R 7 , -N(R 4 )C(O)(CH 2 ) P R 7 , -R 6 NR 4 R 5 , -R 6 N(R 4 )-R 6 -R 7 , -R 6 N(R 6 -R 7 ) 2 , R 6 C(O)N(R 4 )(CH 2 ) p R 7 , -R 6 C(O)O( CH 2 ) p R 7 , -R 6 OC(O)(CH 2 ) p R 7 , -R 6 N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 CH(C(O )OR 4 )(N(R 4 )C(O)R 4 ), substituted aryl or aralkyl.
在另一个优选的实施方案中,本发明涉及下列结构式表示的化合物及其生理上可接受的盐:In another preferred embodiment, the present invention relates to compounds represented by the following structural formula and physiologically acceptable salts thereof:
其中:in:
R是取代的吡咯或者选自取代或未取代的:吲哚、咪唑、1,2,3-三唑、1,2,4-三唑、苯并咪唑、4,5,6,7-四氢吲哚、苯并吲哚、氮杂吲哚、吲唑、吡啶、喹啉、嘧啶、苯、吡嗪、吡唑、噁唑和噻唑;R is substituted pyrrole or is selected from substituted or unsubstituted: indole, imidazole, 1,2,3-triazole, 1,2,4-triazole, benzimidazole, 4,5,6,7-tetra Indoline, benzoindole, azaindole, indazole, pyridine, quinoline, pyrimidine, benzene, pyrazine, pyrazole, oxazole and thiazole;
R1是氢或-A-Z;R 1 is hydrogen or -AZ;
A是-(CH2)n-、-(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;A is -(CH 2 ) n -, -(CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)-, or - (CH 2 ) n S(O) 2 -;
Z是-H、低级烷基、芳烷基、三卤代甲基、三卤代甲基羰基、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3CO-、R3O-或选自下列一组的环系:C3-C6环烷烃、异噁唑、异噻唑、咪唑、苯、吡咯、吲哚、吡啶、吡嗪、嘧啶、苯并噻唑、四氢呋喃、噻吩、咪唑、呋喃、三嗪、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑及其N-氧化物,其中所述环系可以任选地被一个或多个选自下列一组的基团取代:卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基、杂环基-烷基、-CN、-C(O)NR4R5或-NR4R5;Z is -H, lower alkyl, aralkyl, trihalomethyl, trihalomethylcarbonyl, R 3 OC(O)-, -NR 4 R 5 , -C(O)NR 4 R 5 , R 3 CO-, R 3 O- or a ring system selected from the following group: C 3 -C 6 cycloalkane, isoxazole, isothiazole, imidazole, benzene, pyrrole, indole, pyridine, pyrazine, pyrimidine, benzothiazole, tetrahydrofuran, thiophene, imidazole, furan, triazine, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole and their N-oxides, wherein the ring system can optionally be Substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O)-, trihalomethyl, nitro , Aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl, heterocyclyl-alkyl, -CN, - C(O)NR 4 R 5 or -NR 4 R 5 ;
每处出现的R3彼此独立地选自取代或未取代的:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R 3 is independently selected from substituted or unsubstituted: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aryl Alkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
任选地,R4和R5与和其相连的氮结合在一起表示吗啉代、吡咯烷子基、哌啶子基、咪唑-1-基、哌嗪子基、硫吗啉代、吖庚因子基或全氢化-1,4-二氮杂环庚三烯-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;并且Optionally, R 4 and R 5 combined with the nitrogen attached to them represent morpholino, pyrrolidino, piperidino, imidazol-1-yl, piperazino, thiomorpholino, acridino Heptinyl or perhydro-1,4-diazepan-1-yl, each of which is optionally substituted by one or more groups selected from the group consisting of lower alkyl, Hydroxy, lower alkoxy lower alkyl, aromatic, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocycle base-alkyl; and
n是0-3的整数;n is an integer of 0-3;
条件是:requirement is:
a)当R是未取代的吲哚-3-基时,R1不是-NH2;a) when R is unsubstituted indol-3-yl, R 1 is not -NH 2 ;
b)当R是取代或未取代的苯或未取代的咪唑、1,2,3-三唑、1,2,4-三唑、吡啶、嘧啶、吡嗪、吡咯、吡唑、噁唑或噻唑时;R1不是低级烷基、(C3-C6)环烷基、苯或-C(O)NR4R5,其中R4和R5彼此独立地为H、低级烷基或碳环芳基;b) when R is substituted or unsubstituted benzene or unsubstituted imidazole, 1,2,3-triazole, 1,2,4-triazole, pyridine, pyrimidine, pyrazine, pyrrole, pyrazole, oxazole or When thiazole; R 1 is not lower alkyl, (C 3 -C 6 ) cycloalkyl, benzene or -C (O) NR 4 R 5 , wherein R 4 and R 5 are independently H, lower alkyl or carbon Cycloaryl;
c)R不是吡咯-3-基或被烷基或碳环芳基取代的吡咯-3-基;c) R is not pyrrol-3-yl or pyrrol-3-yl substituted by alkyl or carbocyclic aryl;
d)R不是吲哚-3-基或被烷基或碳环芳基取代的吲哚-3-基;d) R is not indol-3-yl or indol-3-yl substituted by alkyl or carbocyclic aryl;
e)R不是氮杂吲哚或在2-位被烷基或碳环芳基取代的氮杂吲哚;e) R is not an azaindole or an azaindole substituted at the 2-position by an alkyl or carbocyclic aryl;
f)当R是取代的吡咯时,该吡咯被下列基团取代:R3OC(O)R6、R3OR6、三卤代甲基、三卤代甲基羰基、-(CH2)p-R7、-O(CH2)pR7、-C(O)N(R4)(CH2)pR7、-C(O)O(CH2)pR7、-OC(O)(CH2)pR7、-N(R4)C(O)(CH2)pR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、R6C(O)N(R4)(CH2)pR7、-R6C(O)O(CH2)pR7、-R6OC(O)(CH2)pR7、-R6N(R4)C(O)(CH2)pR7、-R6CH(C(O)OR4)(N(R4)C(O)R4)、取代的芳基或芳烷基,其中f) When R is a substituted pyrrole, the pyrrole is substituted by the following groups: R 3 OC(O)R 6 , R 3 OR 6 , trihalomethyl, trihalomethylcarbonyl, -(CH 2 ) p -R 7 , -O(CH 2 ) p R 7 , -C(O)N(R 4 )(CH 2 ) p R 7 , -C(O)O(CH 2 ) p R 7 , -OC( O)(CH 2 ) p R 7 , -N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 NR 4 R 5 , -R 6 N(R 4 )-R 6 -R 7 , -R 6 N(R 6 -R 7 ) 2 , R 6 C(O)N(R 4 )(CH 2 ) p R 7 , -R 6 C(O)O(CH 2 ) p R 7 , - R 6 OC(O)(CH 2 ) p R 7 , -R 6 N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 CH(C(O)OR 4 )(N(R 4 ) C(O)R 4 ), substituted aryl or aralkyl, wherein
每处出现的R6独立地为低级烷基或芳基;Each occurrence of R is independently lower alkyl or aryl;
每处出现的R7独立地为烷氧基、卤代烷基、低级烷基哌嗪、羟基、R3O-、R3C(O)-或NR4R5;和Each occurrence of R7 is independently alkoxy, haloalkyl, lower alkylpiperazine, hydroxyl, R3O- , R3C (O)-, or NR4R5 ; and
p是1-3的整数;p is an integer of 1-3;
g)当R是吲哚-3-基时,该吲哚-3-基被下列基团取代:R3OC(O)R6、R3OR6-、三卤代甲基、三卤代甲基羰基、-(CH2)p-R7、-O(CH2)pR7、-C(O)N(R4)(CH2)pR7、-C(O)O(CH2)pR7、-OC(O)(CH2)pR7、-N(R4)C(O)(CH2)pR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、R6C(O)N(R4)(CH2)pR7、-R6C(O)O(CH2)pR7、-R6OC(O)(CH2)pR7、-R6N(R4)C(O)(CH2)pR7、-R6CH(C(O)OR4)(N(R4)C(O)R4)、取代的芳基或芳烷基,和g) When R is indol-3-yl, the indol-3-yl is substituted by the following groups: R 3 OC(O)R 6 , R 3 OR 6 -, trihalomethyl, trihalo Methylcarbonyl, -(CH 2 ) p -R 7 , -O(CH 2 ) p R 7 , -C(O)N(R 4 )(CH 2 ) p R 7 , -C(O)O(CH 2 ) p R 7 , -OC(O)(CH 2 ) p R 7 , -N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 NR 4 R 5 , -R 6 N( R 4 )-R 6 -R 7 , -R 6 N(R 6 -R 7 ) 2 , R 6 C(O)N(R 4 )(CH 2 ) p R 7 , -R 6 C(O)O (CH 2 ) p R 7 , -R 6 OC(O)(CH 2 ) p R 7 , -R 6 N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 CH(C( O)OR 4 )(N(R 4 )C(O)R 4 ), substituted aryl or aralkyl, and
h)当R是吡唑-3-基时,该吡唑-3-基被下列基团取代:R3OC(O)R6、R3OR6-、三卤代甲基、三卤代甲基羰基、-(CH2)p-R7、-O(CH2)pR7、-C(O)N(R4)(CH2)pR7、-C(O)O(CH2)pR7、-OC(O)(CH2)pR7、-N(R4)C(O)(CH2)pR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、R6C(O)N(R4)(CH2)pR7、-R6C(O)O(CH2)pR7、-R6OC(O)(CH2)pR7、-R6N(R4)C(O)(CH2)pR7、-R6CH(C(O)OR4)(N(R4)C(O)R4)、取代的芳基或芳烷基。h) When R is pyrazol-3-yl, the pyrazol-3-yl is substituted by the following groups: R 3 OC(O)R 6 , R 3 OR 6 -, trihalomethyl, trihalo Methylcarbonyl, -(CH 2 ) p -R 7 , -O(CH 2 ) p R 7 , -C(O)N(R 4 )(CH 2 ) p R 7 , -C(O)O(CH 2 ) p R 7 , -OC(O)(CH 2 ) p R 7 , -N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 NR 4 R 5 , -R 6 N( R 4 )-R 6 -R 7 , -R 6 N(R 6 -R 7 ) 2 , R 6 C(O)N(R 4 )(CH 2 ) p R 7 , -R 6 C(O)O (CH 2 ) p R 7 , -R 6 OC(O)(CH 2 ) p R 7 , -R 6 N(R 4 )C(O)(CH 2 ) p R 7 , -R 6 CH(C( O)OR 4 )(N(R 4 )C(O)R 4 ), substituted aryl or aralkyl.
在更优选的实施方案中,本发明涉及任何上述化合物,其中A是-NH-、-O-、-S-、-S(O)-或-S(O)2-;并且Z是环丙基、3-吡啶基或吡嗪基。In a more preferred embodiment, the invention relates to any of the aforementioned compounds, wherein A is -NH-, -O-, -S-, -S(O)- or -S(O) 2 -; and Z is cyclopropane , 3-pyridyl or pyrazinyl.
在另一更优选的实施方案中,本发明涉及任何上述化合物,其中A是-O-;并且Z是乙基、正丙基或异丙基。In another more preferred embodiment, the invention relates to any of the aforementioned compounds, wherein A is -O-; and Z is ethyl, n-propyl or isopropyl.
在另一更优选的实施方案中,本发明涉及任何上述化合物,其中A是-CH2-;并且Z是苯基,其中所述苯基任选地被一个或多个选自下列的基团取代:卤素、三卤代甲基、羟基、-NR4R5、硝基、-CONR4R5、低级烷基、R3O-、-C(O)OR4和-OC(O)R4。In another more preferred embodiment, the present invention relates to any of the aforementioned compounds, wherein A is -CH2- ; and Z is phenyl, wherein said phenyl is optionally replaced by one or more groups selected from Substitution: Halogen, trihalomethyl, hydroxyl, -NR 4 R 5 , nitro, -CONR 4 R 5 , lower alkyl, R 3 O-, -C(O)OR 4 and -OC(O)R 4 .
在另一更优选的实施方案中,本发明涉及任何上述化合物,其中R彼此独立地被一个或多个选自下列的基团取代:卤素、低级烷基、R3O-、羟基、HOC(O)-、R3OC(O)-、R3OC(O)R6、R3OR6、三卤代甲基、三卤代甲基羰基、硝基、-C(O)NR4R5、-NR4R5、R3CO-、-(CH2)n-R7、-C(O)(CH2)n-R7、-C(O)-(CH2)n-C(O)-R7、-O(CH2)nR7、-C(O)NR4(CH2)nR7、C(O)O(CH2)nR7、-OC(O)(CH2)nR7、-NR4C(O)(CH2)nR7、-R6NR4R5、-R6N(R4)-R6-R7、-R6N(R6-R7)2、-R6C(O)NR4(CH2)nR7、-R6C(O)O(CH2)nR7、-R6OC(O)(CH2)nR7、R6NR4C(O)(CH2)nR7、-R6CH(C(O)OR4)(NR5C(O)R4)和任选取代的芳基和任选取代的芳烷基。任选取代的芳基和任选取代的芳烷基任选地被一个或多个选自下列的取代基取代:卤素、三卤代甲基、羟基、-NR4R5、硝基、-CONR4R5、低级烷基、R3O-、C(O)OR4或-OC(O)R3。R6是低级烷基或芳基。R7是烷氧基、卤代烷基、低级烷基哌嗪、羟基、R3O-、R3C(O)-或-NR4R3。在该实施方案的一个方面中,R是吡咯基、吲哚基、氮杂吲哚基、苯基、吡唑基、咪唑基、噻吩基、4,5,6,7-四氢吲哚基或喹啉基。在该实施方案的另一方面中,R是吡咯-2-基、吡咯-3-基、吲哚-2-基、吲哚-3-基、氮杂吲哚-3-基、吡唑-4-基、咪唑-2-基、咪唑-4-基、噻吩-2-基或喹啉-5-基。In another more preferred embodiment, the present invention relates to any of the aforementioned compounds, wherein R are independently substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, hydroxyl, HOC ( O)-, R 3 OC(O)-, R 3 OC(O)R 6 , R 3 OR 6 , trihalomethyl, trihalomethylcarbonyl, nitro, -C(O)NR 4 R 5 , -NR 4 R 5 , R 3 CO-, -(CH 2 ) n -R 7 , -C(O)(CH 2 ) n -R 7 , -C(O)-(CH 2 ) n -C (O)-R 7 , -O(CH 2 ) n R 7 , -C(O)NR 4 (CH 2 ) n R 7 , C(O)O(CH 2 ) n R 7 , -OC(O) (CH 2 ) n R 7 , -NR 4 C(O)(CH 2 ) n R 7 , -R 6 NR 4 R 5 , -R 6 N(R 4 )-R 6 -R 7 , -R 6 N (R 6 -R 7 ) 2 , -R 6 C(O)NR 4 (CH 2 ) n R 7 , -R 6 C(O)O(CH 2 ) n R 7 , -R 6 OC(O)( CH 2 ) n R 7 , R 6 NR 4 C(O)(CH 2 ) n R 7 , -R 6 CH(C(O)OR 4 )(NR 5 C(O)R 4 ) and optionally substituted Aryl and optionally substituted aralkyl. Optionally substituted aryl and optionally substituted aralkyl are optionally substituted with one or more substituents selected from the group consisting of halogen, trihalomethyl, hydroxyl, -NR 4 R 5 , nitro, - CONR 4 R 5 , lower alkyl, R 3 O-, C(O)OR 4 or -OC(O)R 3 . R 6 is lower alkyl or aryl. R 7 is alkoxy, haloalkyl, lower alkylpiperazine, hydroxyl, R 3 O-, R 3 C(O)- or -NR 4 R 3 . In one aspect of this embodiment, R is pyrrolyl, indolyl, azaindolyl, phenyl, pyrazolyl, imidazolyl, thienyl, 4,5,6,7-tetrahydroindolyl or quinolinyl. In another aspect of this embodiment, R is pyrrol-2-yl, pyrrol-3-yl, indol-2-yl, indol-3-yl, azaindol-3-yl, pyrazole- 4-yl, imidazol-2-yl, imidazol-4-yl, thien-2-yl or quinolin-5-yl.
在又一优选的实施方案中,本发明涉及任何上述化合物,其中R1是三氟甲基、氨基、环丙氨基、甲基、乙基、丙基、异丙基、环丙基、2-甲基环丙基、2,2,3,3-四甲基环丙基、2-苯基环丙基、环丁基、环戊基、环己基、-(CH2)p-O-苯基、-(CH2)p-O-(4-甲氧苯基)、-(CH2)p-O-(4-氯苯基)、-(CH2)p-O-(4-甲基苯基)、-(CH2)p-O-(3-甲基苯基)、-(CH2)p-O-(4-苯基苯基)、-(CH2)p-O-(4-羟苯基)、-(CH2)p-O-(4-硝基苯基)、-(CH2)p-O-(4-氨基苯基)、-(CH2)p-O-(4-氨基甲酰基苯基)、-(CH2)p-O-(4-甲氧羰基苯基)、-NH-苯基、-NH-(4-甲氧苯基)、-NH-(4-氯苯基)、-NH-(4-氟苯基)、-NH-(4-异丙基苯基)、异丙氧基、乙氧基、环戊氧基、-(CH2)p-吲哚基、-(CH2)p-吡啶基、-(CH2)p-苯并噻唑基、-(CH2)p-吡咯基、-(CH2)p-四氢呋喃基、-(CH2)p-吡嗪基、-(CH2)p-呋喃基、-(CH2)p-噻吩基、-(CH2)p-苯基、-(CH2)p-异噁唑基、-(CH2)p-(5-甲基异p噁唑基)、-(CH2)p-嘧啶基、-(CH2)p-哒嗪基、-(CH2)n-C(O)-OMe、-(CH2)n-C(O)-OEt或任选地被一个或多个Cl、F、OMe、甲基或氨基取代的苄基。P是1-3的整数。在该实施方案的一个方面中,R任选地被一个或多个选自下列的基团取代:Br、Cl、F、氨基甲基、N,N-二甲氨基甲基、羧基、羧甲基、羧乙基、羰基甲基、羰基乙基、甲氧羰基、乙氧羰基、苯基、4-吗啉代甲基、-C(O)-O-(CH2)2-N(Me)2、-C(O)-O-(CH2)2-N(Et)2、-C(O)-O-CH2-N(Me)2、-C(O)-O-(CH2)2-N(Me)2、-C(O)-NH-(CH2)2-N(Me)2、-CH2-NH-C(O)-CF3以及选自下列的任选被取代的部分:甲基、乙基、丙基、异丙基、丁基和苯基,其中所述任选被取代的部分任选地被一个或多个Br、Cl、F、羟基、硝基、氨基或低级烷基取代。In yet another preferred embodiment, the present invention relates to any of the aforementioned compounds, wherein R is trifluoromethyl, amino, cyclopropylamino, methyl, ethyl, propyl, isopropyl, cyclopropyl, 2- Methylcyclopropyl, 2,2,3,3-tetramethylcyclopropyl, 2-phenylcyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, -(CH 2 ) p -O-benzene base, -(CH 2 ) p -O-(4-methoxyphenyl), -(CH 2 ) p -O-(4-chlorophenyl), -(CH 2 ) p -O-(4-methyl phenylphenyl), -(CH 2 ) p -O-(3-methylphenyl), -(CH 2 ) p -O-(4-phenylphenyl), -(CH 2 ) p -O- (4-hydroxyphenyl), -(CH 2 ) p -O-(4-nitrophenyl), -(CH 2 ) p -O-(4-aminophenyl), -(CH 2 ) p - O-(4-carbamoylphenyl), -(CH 2 ) p -O-(4-methoxycarbonylphenyl), -NH-phenyl, -NH-(4-methoxyphenyl), - NH-(4-chlorophenyl), -NH-(4-fluorophenyl), -NH-(4-isopropylphenyl), isopropoxy, ethoxy, cyclopentyloxy, -( CH 2 ) p -indolyl, -(CH 2 ) p -pyridyl, -(CH 2 ) p -benzothiazolyl, -(CH 2 ) p -pyrrolyl, -(CH 2 ) p -tetrahydrofuranyl , -(CH 2 ) p -pyrazinyl, -(CH 2 ) p -furyl, -(CH 2 ) p -thienyl, -(CH 2 ) p -phenyl, -(CH 2 ) p -iso Oxazolyl, -(CH 2 ) p -(5-methylisoxazolyl), -(CH 2 ) p -pyrimidinyl, -(CH 2 ) p -pyridazinyl, -(CH 2 ) n -C(O)-OMe, -( CH2 ) n -C(O)-OEt, or benzyl optionally substituted with one or more Cl, F, OMe, methyl or amino. P is an integer of 1-3. In one aspect of this embodiment, R is optionally substituted with one or more groups selected from the group consisting of Br, Cl, F, aminomethyl, N,N-dimethylaminomethyl, carboxy, carboxymethyl Carboxyethyl, carbonylmethyl, carbonylethyl, methoxycarbonyl, ethoxycarbonyl, phenyl, 4-morpholinomethyl, -C(O)-O-(CH 2 ) 2 -N(Me ) 2 , -C(O)-O-(CH 2 ) 2 -N(Et) 2 , -C(O)-O-CH 2 -N(Me) 2 , -C(O)-O-(CH 2 ) 2 -N(Me) 2 , -C(O)-NH-(CH 2 ) 2 -N(Me) 2 , -CH 2 -NH-C(O)-CF 3 and any optional Substituted moieties: methyl, ethyl, propyl, isopropyl, butyl, and phenyl, wherein the optionally substituted moieties are optionally replaced by one or more of Br, Cl, F, hydroxy, nitro Substituted by radical, amino or lower alkyl.
另一方面,本发明提供了药物组合物,其中含有一种或多种如本文所定义的本发明化合物与可药用载体或稀释剂。In another aspect, the invention provides pharmaceutical compositions comprising one or more compounds of the invention as defined herein in association with a pharmaceutically acceptable carrier or diluent.
另一方面,本发明提供了抑制一种或多种蛋白激酶活性的方法,该方法包括施用下式所示的化合物及其生理上可接受的盐和生物活性代谢物:In another aspect, the present invention provides a method for inhibiting the activity of one or more protein kinases, the method comprising administering a compound represented by the following formula and its physiologically acceptable salts and biologically active metabolites:
其中:in:
R选自取代或未取代的:脂族基团、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;R is selected from substituted or unsubstituted: aliphatic, aromatic, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkyl-alkane and heterocyclyl-alkyl;
R1是氢或-A-Z;R 1 is hydrogen or -AZ;
R2是氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基、杂环基-烷基;R 2 is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aralkyl, (C 3 -C 6 )cycloalkane Base-alkyl, heterocyclyl-alkyl;
A是-(CH2)n-、-(CH2)nNH-、-(CH2)nO-、-(CH2)nS-、-(CH2)nS(O)-或-(CH2)nS(O)2-;A is -(CH 2 ) n -, -(CH 2 ) n NH-, -(CH 2 ) n O-, -(CH 2 ) n S-, -(CH 2 ) n S(O)-, or - (CH 2 ) n S(O) 2 -;
Z是-H、低级烷基、芳烷基、三卤代甲基、三卤代甲基羰基、R3CO-、R3OC(O)-、-NR4R5、-C(O)NR4R5、R3O-或选自下列一组的环系:(C3-C6)环烷基、苯、吡咯、异噁唑、异噻唑、吲哚、吡啶、吡嗪、嘧啶、苯并噻唑、四氢呋喃、噻吩、三嗪、咪唑、呋喃、苯并咪唑、哒嗪、喹喔啉、吡唑、噁唑、噻唑及其N-氧化物,其中所述环系可以任选地被一个或多个选自下列一组的基团取代:卤素、低级烷基、R3O-、HO-、HOC(O)-、R3OC(O)-、三卤代甲基、硝基、芳基、-CN、-C(O)NR4R5和-NR4R5;Z is -H, lower alkyl, aralkyl, trihalomethyl, trihalomethylcarbonyl, R 3 CO-, R 3 OC(O)-, -NR 4 R 5 , -C(O) NR 4 R 5 , R 3 O- or a ring system selected from the group consisting of (C 3 -C 6 )cycloalkyl, benzene, pyrrole, isoxazole, isothiazole, indole, pyridine, pyrazine, pyrimidine , benzothiazole, tetrahydrofuran, thiophene, triazine, imidazole, furan, benzimidazole, pyridazine, quinoxaline, pyrazole, oxazole, thiazole and N-oxides thereof, wherein the ring system can be optionally Substituted by one or more groups selected from the group consisting of halogen, lower alkyl, R 3 O-, HO-, HOC(O)-, R 3 OC(O)-, trihalomethyl, nitrate Base, aryl, -CN, -C(O)NR 4 R 5 and -NR 4 R 5 ;
每处出现的R3彼此独立地选自取代或未取代的:低级烷基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;Each occurrence of R 3 is independently selected from substituted or unsubstituted: lower alkyl, lower alkoxy lower alkyl, aromatic group, (C 3 -C 6 )cycloalkyl, heterocyclyl, aryl Alkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl;
每处出现的R4和R5彼此独立地为氢或选自取代或未取代的:低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;任选地,R4和R5与和其相连的氮结合在一起表示吗啉代、吡咯烷子基、哌啶子基、咪唑-1-基、哌嗪子基、硫吗啉代、吖庚因子基或全氢化-1,4-二氮杂环庚三烯-1-基,其中每个基团任选地被一个或多个选自下列一组的基团取代:低级烷基、羟基、低级烷氧基低级烷基、芳族基团、(C3-C6)环烷基、杂环基、芳烷基、(C3-C6)环烷基-烷基和杂环基-烷基;并且Each occurrence of R4 and R5 independently of each other is hydrogen or is selected from substituted or unsubstituted: lower alkyl, aromatic group, (C 3 -C 6 ) cycloalkyl, heterocyclyl, aralkyl , (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl; Optionally, R 4 and R 5 combined with the nitrogen attached to them represent morpholino, pyrrolidino, Piperidino group, imidazol-1-yl, piperazino group, thiomorpholino, azepin group or perhydrogenated-1,4-diazepan-1-yl, each of which Optionally substituted with one or more groups selected from the group consisting of lower alkyl, hydroxy, lower alkoxy lower alkyl, aromatic, (C 3 -C 6 )cycloalkyl, heterocycle radical, aralkyl, (C 3 -C 6 )cycloalkyl-alkyl and heterocyclyl-alkyl; and
n是0-3的整数。n is an integer of 0-3.
上述方法中优选的方法是其中在需要的接受治疗者中抑制蛋白激酶。Preferred of the above methods are those wherein a protein kinase is inhibited in a subject in need thereof.
任何上述方法中优选的方法是其中的化合物为立体异构体的混合物。Preferred methods of any of the above methods are those wherein the compound is a mixture of stereoisomers.
任何上述方法中优选的方法是其中的立体异构体是对映体。A preferred method of any of the above methods is one in which the stereoisomers are enantiomers.
任何上述方法中优选的方法是其中的立体异构体是E和Z异构体。A preferred method of any of the above methods is one wherein the stereoisomers are the E and Z isomers.
任何上述方法中优选的方法是其中的化合物为结构异构体的混合物。Preferred methods of any of the above methods are those wherein the compound is a mixture of structural isomers.
任何上述方法中优选的方法是其中的结构异构体是互变异构体。A preferred method of any of the above methods is one wherein the structural isomers are tautomers.
任何上述方法中优选的方法是其中所述蛋白激酶是受体酪氨酸激酶或非受体酪氨酸激酶。A preferred method of any of the above methods is wherein the protein kinase is a receptor tyrosine kinase or a non-receptor tyrosine kinase.
任何上述方法中优选的方法是其中所述酪氨酸激酶选自KDR、Flt-1、TIE-2、FGFR、PDGFR、IGF-1-R、c-Met、Lck、Src、fyn、Lyn、Blk和yes。A preferred method in any of the above methods is wherein the tyrosine kinase is selected from the group consisting of KDR, Flt-1, TIE-2, FGFR, PDGFR, IGF-1-R, c-Met, Lck, Src, fyn, Lyn, Blk and yes.
另一方面,本发明涉及治疗或根本上抑制需治疗者的过度增殖疾病或炎性疾病的方法,该方法包括对所述接受治疗者施用任何如本文定义的本发明化合物。In another aspect, the invention relates to a method of treating or substantially inhibiting a hyperproliferative or inflammatory disease in a subject in need thereof, the method comprising administering to said subject any compound of the invention as defined herein.
另一方面,本发明涉及治疗或抑制需治疗者血管生成的方法,该方法包括对所述接受治疗者施用任何如本文定义的本发明化合物。In another aspect, the invention relates to a method of treating or inhibiting angiogenesis in a subject in need thereof, the method comprising administering to said subject any compound of the invention as defined herein.
另一方面,本发明涉及诱导需治疗者抗血管生成作用的方法,该方法包括对所述接受治疗者施用任何如本文定义的本发明化合物。In another aspect, the invention relates to a method of inducing an anti-angiogenic effect in a subject in need thereof, the method comprising administering to said subject any compound of the invention as defined herein.
另一方面,本发明涉及治疗或抑制需治疗者疾病或病症进行的方法,该方法包括对所述接受治疗者施用任何如本文定义的本发明化合物,其中所述疾病或病症选自癌、关节炎、动脉粥样硬化、再狭窄、牛皮癣、血管瘤、心肌血管生成、冠状和脑侧突、局部缺血性四肢血管生成、角膜疾病、潮红、新生血管性青光眼、黄斑变性、创伤愈合、消化性溃疡、与螺旋杆菌有关的疾病、病毒引起的血管生成疾病、骨折、糖尿病性视网膜病、节段性回肠炎、炎性肠疾病、猫抓伤引起的发烧、视网膜病或早熟、溃疡、甲状腺增生、灼伤、外伤、急性肺损伤、慢性肺病、中风、息肉、囊肿、滑膜炎、慢性和变应性炎症、卵巢过度刺激综合征、肺和脑水肿、瘢痕瘤、纤维变性、肝硬变、腕管综合征、脓毒症、成人呼吸窘迫综合征、多器官机能障碍综合征、腹水和与肿瘤有关的渗漏积液以及水肿。In another aspect, the present invention relates to a method of treating or inhibiting the progress of a disease or condition in a subject in need thereof, the method comprising administering to said subject any compound of the invention as defined herein, wherein said disease or condition is selected from cancer, joint inflammation, atherosclerosis, restenosis, psoriasis, hemangioma, myocardial angiogenesis, coronary and scoliosis, ischemic extremity angiogenesis, corneal disease, flushing, neovascular glaucoma, macular degeneration, wound healing, digestion Ulcers, diseases associated with Helicobacter, angiogenic diseases caused by viruses, bone fractures, diabetic retinopathy, Crohn's disease, inflammatory bowel disease, fever from cat scratches, retinopathy or precocious puberty, ulcers, thyroid Hyperplasia, burns, trauma, acute lung injury, chronic lung disease, stroke, polyps, cysts, synovitis, chronic and allergic inflammation, ovarian hyperstimulation syndrome, pulmonary and cerebral edema, keloids, fibrosis, cirrhosis , carpal tunnel syndrome, sepsis, adult respiratory distress syndrome, multiple organ dysfunction syndrome, ascites and effusions associated with tumors, and edema.
另一方面,本发明涉及影响需治疗者血管渗透性过高或水肿产生的方法,该方法包括对所述接受治疗者施用任何如本文定义的本发明化合物。该方法中优选的方法是,其中对水肿产生的作用为抗水肿。In another aspect, the invention relates to a method of affecting the development of vascular hyperpermeability or edema in a subject in need thereof, the method comprising administering to said subject any compound of the invention as defined herein. Preferred methods are those wherein the effect on edema is anti-edema.
上述任何方法中的优选方法是,其中蛋白激酶是丝氨酸激酶。A preferred method of any of the above methods is wherein the protein kinase is a serine kinase.
上述任何方法中的优选方法是,其中蛋白激酶是苏氨酸激酶。A preferred method of any of the above methods is wherein the protein kinase is threonine kinase.
本发明特别优选的化合物是:Particularly preferred compounds of the invention are:
3-环丙基-4-{[(3,5-二甲基-4-(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[(3,5-dimethyl-4-(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-2-pyrazole Lin-5-one,
3-环丙基-4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-{[3-(4-甲基-1-哌嗪基甲基)-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-(4-methyl-1-piperazinylmethyl)-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazole Lin-5-one,
3-环丙基-4-[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazolin-5-one,
3-环丙基-4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-(3,5-二溴-4-羟基亚苄基)-2-吡唑啉-5-酮,3-cyclopropyl-4-(3,5-dibromo-4-hydroxybenzylidene)-2-pyrazolin-5-one,
3-环丙基-4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
3-环丙基-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[4-(2-dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[4-(2-dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3-乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-ethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[3-(2-dimethylaminoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[3-(2-dimethylaminoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-环丙基-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-cyclopropyl-2-pyrazolin-5-one,
3-环丙基-4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-{[(3,5-二甲基-4-[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(3-吡啶基)-2-吡唑啉-5-酮,4-{[(3,5-Dimethyl-4-[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(3-pyridyl)- 2-pyrazolin-5-one,
4-[(3,5-二甲基-4-[(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(4-二乙醇氨基甲基-3,5-二甲基-吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethyl-pyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-{[3-(4-甲基-1-哌嗪基甲基)-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(3-吡啶基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinylmethyl)-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(3-pyridyl)-2 -pyrazolin-5-one,
3-(3-吡啶基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(3-pyridyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(3-吡啶基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-(3-吡啶基-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-(3-pyridyl-2-pyrazolin-5-one,
3-(3-吡啶基)-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-pyridyl)-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-(3-吡啶基)-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-(3-pyridyl)-2-pyrazolin-5-one ,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-(3-吡啶基)-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazoline-5 -ketone,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one ,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-isopropoxy-2- pyrazolin-5-one,
4-[(3,5-二甲基-4-[(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one ,
4-[(4-二乙氨基甲基-3,5-二甲基-吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethyl-pyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(哌啶子基甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(piperidinomethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-isopropoxy-4-{[3-(4-methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrrole oxazolin-5-one,
3-异丙氧基-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-isopropoxy-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline-5- ketone,
3-异丙氧基-4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-异丙氧基-2-吡唑啉-5-酮,4-(3,5-Dibromo-4-hydroxybenzylidene)-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-Bromopyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-isopropoxy-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(5-Ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one ,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(5-chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-[[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-ethoxy-2-pyrrole oxazolin-5-one,
4-[(3,5-二甲基-4-[(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(4-二乙氨基甲基-3,5-二甲基-吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethyl-pyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(哌啶子基甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(piperidinomethylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(4-二乙醇氨基甲基-3,5-二甲基-吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethyl-pyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-ethoxy-4-{[3-(4-methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazole Lin-5-one,
3-乙氧基-4-{[[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-ethoxy-4-{[[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline-5- ketone,
3-乙氧基-4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-乙氧基-2-吡唑啉-5-酮,4-(3,5-Dibromo-4-hydroxybenzylidene)-3-ethoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-乙氧基-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-乙氧基-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-ethoxy-2-pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-乙氧基-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-ethoxy-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3-(4-甲基-1-哌嗪基甲基)-4,5-四亚甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinylmethyl)-4,5-tetramethylenepyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
3-吡嗪基-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-pyrazinyl-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazolin-5-one ,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-吡嗪基-2-吡唑啉-5-酮,4-(3,5-Dibromo-4-hydroxybenzylidene)-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-Bromopyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
3-吡嗪基-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-pyrazinyl-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one ,
4-[(吲哚-3-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
3-吡嗪基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-pyrazinyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(Indol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,4-Diethoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(5-chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-(imidazol-2-yl)-2-pyrazoline -5-keto,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazoline -5-keto,
3-(咪唑-2-基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(咪唑-2-基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(imidazol-2-yl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-(咪唑-2-基))-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-(imidazol-2-yl))-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-(imidazol-2-yl)-2-pyrazoline-5- ketone,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-(imidazol-2-yl)-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazoline- 5-keto,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(咪唑-2-基)-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(imidazol-2-yl)-2-pyrazoline-5- ketone,
4-[(4-溴吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(5-甲氧基吲哚-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(5-methoxyindol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
3-(1-甲基环丙基)-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(1-methylcyclopropyl)-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-(1-methylcyclopropyl)-2-pyrrole oxazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrrole oxazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(pyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基))-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl))-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-(1-methylcyclopropyl)-2-pyrazoline- 5-keto,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-{[3-(2-Amino-2-carboxyethyl)indol-2-yl]methylene}-3-(1-methylcyclopropyl)-2-pyrazolin-5-one ,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazole Lin-5-one,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(1-甲基环丙基)-2-吡唑啉-5-酮,4-[(5-chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(1-methylcyclopropyl)-2-pyrazoline- 5-keto,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(2-furyl)-2- pyrazolin-5-one,
4-{[3,5-二甲基-4-(1-吡咯烷基甲基)吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(1-pyrrolidinylmethyl)pyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene}-3-(2-furyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3-二乙氨基乙基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3-Diethylaminoethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-{[3-(4-甲基-1-哌嗪基甲基)-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(2-furyl)-4-{[3-(4-methyl-1-piperazinylmethyl)-4,5-tetramethylenepyrrol-2-yl]methylene}-2 -pyrazolin-5-one,
3-(2-呋喃基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(2-furyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
3-(2-呋喃基)-4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(2-呋喃基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(4-dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazoline- 5-keto,
3-(2-呋喃基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-呋喃基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-furyl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-(3-(2-呋喃基))-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-(3-(2-furyl))-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-(2-furyl)-2-pyrazolin-5-one ,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-(2-furyl)-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazoline-5 -ketone,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one ,
4-[(3,5-二甲基-4-[(吗啉代甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[(morpholinomethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one ,
4-[(3,5-二甲基-4-(4-甲基-1-哌嗪基)甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(4-methyl-1-piperazinyl)methylpyrrol-2-yl]methylene}-3-(2-thienyl)-2- pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-{[3-(4-甲基-1-哌嗪基甲基)-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinylmethyl)-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(2-thienyl)-2 -pyrazolin-5-one,
4-{[3-(1-吡咯烷基甲基)-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-(1-pyrrolidinylmethyl)-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline- 5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(2-噻吩基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(4-氧代-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-oxo-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
3-(2-噻吩基)-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-thienyl)-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(5-甲氧基吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(5-methoxyindol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline- 5-keto,
4-[(吲哚-3-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(pyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,4-Diethoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-(2-thienyl)-2-pyrazolin-5-one ,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazoline-5 -ketone,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one ,
4-[(4-溴吡咯-2-基)亚甲基]-3-环丙氧基-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-cyclopropoxy-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-环丙氧基-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-cyclopropoxy-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-环丙氧基-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-cyclopropoxy-2-pyrazolin-5-one,
3-环丙氧基-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[4-(2-dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[4-(2-dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(3-乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(3-ethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[3-(2-dimethylaminoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[3-(2-dimethylaminoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(3,5-dimethyl-4-ethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[3,5-dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropoxy-4-[3,5-dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-2-pyrazolin-5-one ,
3-环丙氧基-4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-{[3,5-dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-2-pyrazoline-5- ketone,
3-环丙氧基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氧基-4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-环丙氧基-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-cyclopropoxy-2-pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-环丙氧基-2-吡唑啉-5-酮,4-{[3-(2-Amino-2-carboxyethyl)indol-2-yl]methylene}-3-cyclopropoxy-2-pyrazolin-5-one,
3-环丙氧基-4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropoxy-4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-环丙氧基-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-cyclopropoxy-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
3-[2-(5-嘧啶基乙基)]-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(5-pyrimidinylethyl)]-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one ,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one ,
4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[3-(4-Morpholinoethoxycarbonylindol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one ,
4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[3-(4-Morpholinoethylaminocarbonylindol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one ,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazoline-5- ketone,
4-[(1-甲基吲哚-3-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazoline-5- ketone,
4-[(5-甲氧基吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(5-methoxyindol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
3-[2-(5-嘧啶基乙基)]-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(5-pyrimidinylethyl)]-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-[2-(5-pyrimidinylethyl)]- 2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]- 2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
3-[2-(5-嘧啶基乙基)]-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(5-pyrimidinylethyl)]-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-[2-(5-pyrimidinylethyl)]-2-pyridine oxazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-[2-(5-pyrimidinylethyl)]-2-pyrazoline- 5-keto,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2 -pyrazolin-5-one,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-[2-(5-嘧啶基乙基)]-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-[2-(5-pyrimidinylethyl)]-2-pyrrole oxazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
3-(2-苯乙基)-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-phenylethyl)-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(5-甲氧基吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(5-methoxyindol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
3-(2-苯乙基)-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-phenylethyl)-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-(2-苯乙基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-(2-phenylethyl)-2-pyrazoline -5-keto,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazoline -5-keto,
4-[(吲哚-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(3,4-Diethoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-(2-苯乙基)-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-(2-phenylethyl)-2-pyrazoline-5- ketone,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-(2-苯乙基)-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-(2-phenylethyl)-2-pyrazolin-5-one,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazoline- 5-keto,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-(2-苯乙基)-2-吡唑啉-5-酮,4-[(5-Chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-(2-phenylethyl)-2-pyrazoline-5- ketone,
4-[(4-溴吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(4-bromopyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one,
3-[2-(4-羟苯基)乙基]-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-hydroxyphenyl)ethyl]-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one,
4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one ,
4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one ,
4-[(3-乙氧羰基吲哚-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(3-ethoxycarbonylindol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one,
4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazoline-5- ketone,
4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazoline-5- ketone,
3-[2-(4-羟苯基)乙基]-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Hydroxyphenyl)ethyl]-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
3-[2-(4-羟苯基)乙基]-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Hydroxyphenyl)ethyl]-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
3-[2-(4-羟苯基)乙基]-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Hydroxyphenyl)ethyl]-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazoline-5- ketone,
3-[2-(4-羟苯基)乙基]-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Hydroxyphenyl)ethyl]-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazoline-5- ketone,
4-[3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazoline-5 -ketone,
3-[2-(4-羟苯基)乙基]-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Hydroxyphenyl)ethyl]-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-羟苯基)乙基]-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-hydroxyphenyl)ethyl]-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-[2-(4-hydroxyphenyl)ethyl] -2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl] -2-pyrazolin-5-one,
4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-[2-(4-hydroxyphenyl)ethyl]-2- pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-[2-(4-hydroxyphenyl)ethyl]-2-pyrazoline -5-keto,
4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]- 2-pyrazolin-5-one,
4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-3-[2-(4-羟苯基)乙基]-2-吡唑啉-5-酮,4-[(5-chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-3-[2-(4-hydroxyphenyl)ethyl]-2- pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(4-溴吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(4-bromopyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(4-氯吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(4-chloropyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(5-氯吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(5-chloropyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(5-三氟甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(5-trifluoromethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[4-(2-二甲氨基乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Aminophenyl)ethyl]-4-[4-(2-Dimethylaminoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
3-[2-(4-氨基苯基)乙基]-4-[4-(2-二甲氨基乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Aminophenyl)ethyl]-4-[4-(2-Dimethylaminoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
3-[2-(4-氨基苯基)乙基]-4-[(3-乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(3-ethoxycarbonylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[3-(2-二甲氨基乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Aminophenyl)ethyl]-4-[3-(2-Dimethylaminoethoxycarbonylindol-2-yl)methylene]-2-pyrazoline-5- ketone,
3-[2-(4-氨基苯基)乙基]-4-[3-(2-二甲氨基乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-Aminophenyl)ethyl]-4-[3-(2-Dimethylaminoethylaminocarbonylindol-2-yl)methylene]-2-pyrazoline-5- ketone,
3-[2-(4-氨基苯基)乙基]-4-[4-(4-吗啉代乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[4-(4-morpholinoethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
3-[2-(4-氨基苯基)乙基]-4-[4-(4-吗啉代乙氨基羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[4-(4-morpholinoethylaminocarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one ,
3-[2-(4-氨基苯基)乙基]-4-[3-(4-吗啉代乙氧羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[3-(4-morpholinoethoxycarbonylindol-2-yl)methylene]-2-pyrazoline-5- ketone,
3-[2-(4-氨基苯基)乙基]-4-[3-(4-吗啉代乙氨基羰基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[3-(4-morpholinoethylaminocarbonylindol-2-yl)methylene]-2-pyrazoline-5- ketone,
3-[2-(4-氨基苯基)乙基]-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-{[3,5-dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene} -2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-{[3,5-dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl]methylene} -2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(3,4-二乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(3,4-diethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3-(2-乙酰氨基-2-甲氧羰基乙基)吲哚-2-基]亚甲基}-3-[2-(4-氨基苯基)乙基]-2-吡唑啉-5-酮,4-{[3-(2-Acetamido-2-methoxycarbonylethyl)indol-2-yl]methylene}-3-[2-(4-aminophenyl)ethyl]-2- pyrazolin-5-one,
4-{[3-(2-氨基-2-羧乙基)吲哚-2-基]亚甲基}-3-[2-(4-氨基苯基)乙基]-2-吡唑啉-5-酮,4-{[3-(2-amino-2-carboxyethyl)indol-2-yl]methylene}-3-[2-(4-aminophenyl)ethyl]-2-pyrazoline -5-keto,
3-[2-(4-氨基苯基)乙基]-4-[(5-乙氧基-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(5-ethoxy-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]- 2-pyrazolin-5-one,
3-[2-(4-氨基苯基)乙基]-4-[(5-氯-4-甲氧羰基甲基-3-甲氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-[2-(4-aminophenyl)ethyl]-4-[(5-chloro-4-methoxycarbonylmethyl-3-methoxycarbonylpyrrol-2-yl)methylene]-2- pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(4-pyrimidinyl)- 2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(哌啶子基甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(piperidinomethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2 -pyrazolin-5-one,
3-(4-嘧啶基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(4-pyrimidinyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(4-嘧啶基)吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(4-pyrimidinyl)pyrazolin-5-one,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(5-pyrimidinyl)- 2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazoline-5- ketone,
4-[(3,5-二甲基-4-二乙氨基甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-diethylaminomethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(哌啶子基甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(piperidinomethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2 -pyrazolin-5-one,
3-(5-嘧啶基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(5-pyrimidinyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-(3,5-Dibromo-4-hydroxybenzylidene)-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(2-pyrimidinyl)- 2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(哌啶子基甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(piperidinomethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3-diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2 -pyrazolin-5-one,
3-(2-嘧啶基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(2-pyrimidinyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(3-pyridazinyl) -2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one ,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one ,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one ,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3-Diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one ,
4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(3-pyridazinyl)- 2-pyrazolin-5-one,
3-(3-哒嗪基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(3-pyridazinyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline -5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazoline-5- ketone,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(4-pyridazinyl) -2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-(哌啶子基甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(piperidinomethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙醇氨基甲基-3,5-二甲基-吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethyl-pyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one ,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one ,
4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3-Morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one ,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3-Diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one ,
4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[3-(4-Methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-3-(4-pyridazinyl)- 2-pyrazolin-5-one,
3-(4-哒嗪基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(4-pyridazinyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazoline -5-keto,
4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5- ketone,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-(3,5-Dibromo-4-hydroxybenzylidene)-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one ,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one ,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(5-isoxazolyl )-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-[(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazoline -5-keto,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one ,
4-[(4-二乙醇氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one ,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazoline-5- ketone,
3-(5-异噁唑基)-4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(5-isoxazolyl)-4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazoline-5- ketone,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3-Diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazoline-5- ketone,
3-(5-异噁唑基)-4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(5-isoxazolyl)-4-{[3-(4-methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene} -2-pyrazolin-5-one,
3-(5-异噁唑基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(5-isoxazolyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene}-2-pyrazole Lin-5-one,
3-(5-异噁唑基)-4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(5-isoxazolyl)-4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazoline-5 -ketone,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one ,
4-(3,5-二溴-4-羟基亚苄基)-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-(3,5-Dibromo-4-hydroxybenzylidene)-3-(5-isoxazolyl)-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazoline-5- ketone,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrazole Lin-5-one,
4-[(3,5-二甲基-4-{[(4-甲基-1-哌嗪基甲基)吡咯-2-基]亚甲基}-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-{[(4-methyl-1-piperazinylmethyl)pyrrol-2-yl]methylene}-3-(5-methyl-3 -isoxazolyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-[(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-[(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)- 2-pyrazolin-5-one,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrazole Lin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrrole oxazolin-5-one,
4-[(4-二乙醇氨基甲基-3,5-二甲基-吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(4-diethanolaminomethyl-3,5-dimethyl-pyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrrole oxazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrrole oxazolin-5-one,
3-(5-甲基-3-异噁唑基)-4-[(3-吗啉代甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(5-methyl-3-isoxazolyl)-4-[(3-morpholinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrrole oxazolin-5-one,
4-[(3-二乙氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3-Diethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrrole oxazolin-5-one,
3-(5-甲基-3-异噁唑基)-4-{[3-(4-甲基-1-哌嗪基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(5-methyl-3-isoxazolyl)-4-{[3-(4-methyl-1-piperazinyl)methyl-4,5-tetramethylenepyrrol-2-yl ]methylene}-2-pyrazolin-5-one,
3-(5-甲基-3-异噁唑基)-4-{[3-(1-吡咯烷基)甲基-4,5-四亚甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(5-Methyl-3-isoxazolyl)-4-{[3-(1-pyrrolidinyl)methyl-4,5-tetramethylenepyrrol-2-yl]methylene} -2-pyrazolin-5-one,
3-(5-甲基-3-异噁唑基)-4-[(3-哌啶子基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(5-methyl-3-isoxazolyl)-4-[(3-piperidinomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2- pyrazolin-5-one,
4-[(4-二甲氨基-4,5,6,7-四氢吲哚-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Dimethylamino-4,5,6,7-tetrahydroindol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2- pyrazolin-5-one,
4-[(4-二乙氨基乙基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Diethylaminoethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrazole Lin-5-one,
4-(3,5-二溴-4-羟基亚苄基)-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-(3,5-dibromo-4-hydroxybenzylidene)-3-(5-methyl-3-isoxazolyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrazole Lin-5-one,
3-(5-甲基-3-异噁唑基)-4-[(4-氧代-1,5,6,7-四氢吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(5-methyl-3-isoxazolyl)-4-[(4-oxo-1,5,6,7-tetrahydroindol-2-yl)methylene]-2-pyridine oxazolin-5-one,
4-[(4-二甲氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(4-Dimethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrazoline-5 -ketone,
3-环丙基-4-(1-3-[(二甲氨基)甲基]-4,5,6,7-四氢-1H-2-吲哚基亚甲基)-4,5-二氢-1H-5-吡唑酮,3-cyclopropyl-4-(1-3-[(dimethylamino)methyl]-4,5,6,7-tetrahydro-1H-2-indolylmethylene)-4,5- Dihydro-1H-5-pyrazolone,
3-环丙基-4-(1-4-[(1,1-二甲基铵)甲基]-3,5-二甲基-1H-2-吡咯基亚甲基)-4,5-二氢-1H-5-吡唑酮马来酸盐,3-cyclopropyl-4-(1-4-[(1,1-dimethylammonium)methyl]-3,5-dimethyl-1H-2-pyrrolylmethylene)-4,5 - Dihydro-1H-5-pyrazolone maleate,
4-[1-(4-[[2-(二甲氨基)乙基](甲基)氨基]甲基-3,5-二甲基-1H-2-吡咯基)亚甲基]-3-(2-吡嗪基)-4,5-二氢-1H-5-吡唑酮,4-[1-(4-[[2-(Dimethylamino)ethyl](methyl)amino]methyl-3,5-dimethyl-1H-2-pyrrolyl)methylene]-3 -(2-pyrazinyl)-4,5-dihydro-1H-5-pyrazolone,
4-(1-4-[2-(二乙氨基)乙基]-3,5-二甲基-1H-2-吡咯基亚甲基)-3-(2-吡嗪基)-4,5-二氢-1H-5-吡唑酮,4-(1-4-[2-(diethylamino)ethyl]-3,5-dimethyl-1H-2-pyrrolylmethylene)-3-(2-pyrazinyl)-4, 5-dihydro-1H-5-pyrazolone,
4-1-[4-(3-氯丙酰基)-3,5-二甲基-1H-2-吡咯基]亚甲基-3-(2-吡嗪基)-4,5-二氢-1H-5-吡唑酮,4-1-[4-(3-Chloropropionyl)-3,5-dimethyl-1H-2-pyrrolyl]methylene-3-(2-pyrazinyl)-4,5-dihydro -1H-5-pyrazolone,
4-(1-3-[(二甲氨基)甲基]-4,5,6,7-四氢-1H-2-吲哚基亚甲基)-3-(2-吡嗪基)-4,5-二氢-1H-5-吡唑酮,4-(1-3-[(Dimethylamino)methyl]-4,5,6,7-tetrahydro-1H-2-indolylmethylene)-3-(2-pyrazinyl)- 4,5-dihydro-1H-5-pyrazolone,
4-(1-4-[(二甲氨基)甲基]-3,5-二甲基-1H-2-吡咯基亚甲基)-3-(5-甲基-3-异噁唑基)-4,5-二氢-1H-5-吡唑酮,4-(1-4-[(dimethylamino)methyl]-3,5-dimethyl-1H-2-pyrrolylmethylene)-3-(5-methyl-3-isoxazolyl )-4,5-dihydro-1H-5-pyrazolone,
3-环丙基-4-[(4,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-苄基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-benzyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲氧基苯氧基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methoxyphenoxymethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-苯氧基甲基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-phenoxymethyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氯苯氧基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-chlorophenoxymethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲氧基苄基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methoxybenzyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-苄基-4-{[4-(2-羧乙基)-3-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-Benzyl-4-{[4-(2-carboxyethyl)-3-methylpyrrol-2-yl]methylene}-2-pyrazolin-5-one,
3-苄基-4-{[3-(2-羧乙基)-4-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-Benzyl-4-{[3-(2-carboxyethyl)-4-methylpyrrol-2-yl]methylene}-2-pyrazolin-5-one,
3-(4-甲基苯氧基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methylphenoxymethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
2-氨基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,2-amino-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-苯乙基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-phenethyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-苯氨基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-anilino-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲苄基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methylbenzyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲基苯氨基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methylanilino)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲氧基苯氨基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methoxyanilino)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氯苄基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-chlorobenzyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氯苯氨基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Chloroanilino)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3-氯苯氧基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-chlorophenoxymethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(吲哚-3-基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(indol-3-yl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3-甲氧基苄基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-methoxybenzyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3,4-二甲氧基苄基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3,4-dimethoxybenzyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲氧基苯乙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methoxyphenethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-苯基苯氧基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-phenylphenoxymethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-苯基丙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-phenylpropyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3-苯基丙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-phenylpropyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-羟基苯乙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-hydroxyphenethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基吡咯-2-基)亚甲基]-3-苯乙基-2-吡唑啉-5-酮,4-[(3,5-Dimethylpyrrol-2-yl)methylene]-3-phenethyl-2-pyrazolin-5-one,
3-(4-甲基苯乙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methylphenethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-硝基苯乙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-nitrophenethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氟苯氨基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-fluoroanilino)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氯苯乙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-chlorophenethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4,5-二甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4,5-Dimethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-苯乙基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-phenethyl-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吡咯-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-{[1-(4-hydroxybutyl)pyrrol-2-yl]methylene}-3-isopropoxy-2-pyrazolin-5-one,
3-环戊氧基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopentyloxy-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-叔丁基脲基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-tert-butylureido-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(4-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(4-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氨基苯乙基)-4-[(4-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-aminophenethyl)-4-[(4-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(1-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(1-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氨基苯乙基)-4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-aminophenethyl)-4-[(3,5-dimethyl-4-ethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-[(5-甲基吡咯-2-基)亚甲基]2-吡唑啉-5-酮,3-ethoxy-4-[(5-methylpyrrol-2-yl)methylene]2-pyrazolin-5-one,
3-(4-氨基甲酰基苯乙基)-4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-carbamoylphenethyl)-4-[(3,5-dimethyl-4-ethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(4-甲氧羰基苯乙基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(4-methoxycarbonylphenethyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-(4-氨基苯乙基)-4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-aminophenethyl)-4-[(3,5-dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(5-乙基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(5-Ethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(4-羟基苯乙基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(4-hydroxyphenethyl)-2-pyrazolin-5-one,
3-(乙氧羰基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(ethoxycarbonylmethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲氧基苯氨基羰基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Methoxyanilinocarbonylmethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{(3-(2-羧乙基)-4-甲基吡咯-2-基]亚甲基}-3-异丙基-2-吡唑啉-5-酮,4-{(3-(2-carboxyethyl)-4-methylpyrrol-2-yl]methylene}-3-isopropyl-2-pyrazolin-5-one,
3-环丙基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丁基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclobutyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3-吡啶基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-pyridyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[[1-(3,5-二氯苯基)吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[[1-(3,5-dichlorophenyl)pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吡咯-2-基)亚甲基]-3-(2,2,3,3-四甲基环丙基)-2-吡唑啉-5-酮,4-[(pyrrol-2-yl)methylene]-3-(2,2,3,3-tetramethylcyclopropyl)-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环戊基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopentyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-甲基环丙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-methylcyclopropyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-(苯并噻唑-2-基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,4-(benzothiazol-2-yl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4,5-二甲基吡咯-2-基)亚甲基]-4-(3-吡啶基)]-2-吡唑啉-5-酮,4-[(4,5-Dimethylpyrrol-2-yl)methylene]-4-(3-pyridyl)]-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-ethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(吡咯-2-基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(pyrrol-2-yl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(苯并噻唑-2-基)-4-[(4-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(benzothiazol-2-yl)-4-[(4-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
3-环丙基-4-[(5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(5-甲基吡咯-2-基)亚甲基]-3-(-3-吡啶基)-2-吡唑啉-5-酮,4-[(5-methylpyrrol-2-yl)methylene]-3-(-3-pyridyl)-2-pyrazolin-5-one,
3-环丙基-4-[(5-乙基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-ethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(5-苯基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(5-phenylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-丙基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-Propyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-甲基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-methyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-异丙基苯氨基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-isopropylanilino)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3-苯氨基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-phenylamino)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-{[1-(4-羟基丁基)吡咯-2-基]亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-{[1-(4-hydroxybutyl)pyrrol-2-yl]methylene]-2-pyrazolin-5-one,
4-{[4-(2-羧乙基)-3-甲基吡咯-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3-methylpyrrol-2-yl]methylene}-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-羧基-3,5-二甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-carboxy-3,5-dimethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-乙酰基-3,5-二甲基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-acetyl-3,5-dimethylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-isopropoxy-2-pyrazoline-5- ketone,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl]methylene}-3-isopropoxy-2-pyrazoline-5- ketone,
4-[(5-乙氧羰基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(5-ethoxycarbonylpyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(5-羧基吡咯-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(5-carboxypyrrol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-(4-氯苯氨基羰基甲基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Chloroanilinocarbonylmethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(5-苯基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-phenylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(四氢呋喃-3-基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(tetrahydrofuran-3-yl)-2-pyrazolin-5-one,
4-[(4-羧基-3,5-二甲基吡咯-2-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4-[(4-carboxy-3,5-dimethylpyrrol-2-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,4-二甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,4-Dimethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-羧乙基)-3-甲基吡咯-2-基]亚甲基}-3-环丙基-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3-methylpyrrol-2-yl]methylene}-3-cyclopropyl-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(5-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(2-甲氧羰基乙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(2-Methoxycarbonylethyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧羰基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxycarbonyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(反-2-苯基环丙基)-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(trans-2-phenylcyclopropyl)-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环己基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclohexyl-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙氨基-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropylamino-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(5-吗啉代甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-morpholinomethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(5-羧基吡咯-2-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4-[(5-carboxypyrrol-2-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
4-[(4-氯吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-chloropyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-Bromopyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(5-氯吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(5-chloropyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4[(4-氯吡咯-2-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4[(4-chloropyrrol-2-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
4-[(4-溴吡咯-2-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4-[(4-Bromopyrrol-2-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
3-吡嗪基-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-pyrazinyl-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(3-吡啶基)-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-pyridyl)-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-羧基-3,5-二甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-carboxy-3,5-dimethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
3-环丙基-4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3,5-dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-2-pyrazolin-5-one ,
4-{[3,5-二甲基-4-(2-二甲氨基乙氧羰基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethoxycarbonyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮马来酸盐,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one maleate,
4-{[3,5-二甲基-4-(2-吗啉代乙氨基羰基)吡咯-2-基]亚甲基-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-morpholinoethylaminocarbonyl)pyrrol-2-yl]methylene-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基吡咯-2-基)亚甲基]-3-(2-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethylpyrrol-2-yl)methylene]-3-(2-pyridyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-(2-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-(2-pyridyl)-2-pyrazolin-5-one,
4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-3-(2-吡啶基)-2-吡唑啉-5-酮,4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-3-(2-pyridyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3,5-Dimethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
3-环丙基-4-[(4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-三氟乙酰氨基甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-trifluoroacetylaminomethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-aminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(苯并噻唑-2-基)-4-[(3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(benzothiazol-2-yl)-4-[(3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-(苯并噻唑-2-基)-4-[(3,5-二甲基-4-吗啉代甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(benzothiazol-2-yl)-4-[(3,5-dimethyl-4-morpholinomethylpyrrol-2-yl)methylene]-2-pyrazoline-5- ketone,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-乙氧羰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-羧基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-carboxypyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二乙醇氨基甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-diethanolaminomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-三氟乙酰氨基甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-trifluoroacetylaminomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
3-环丙基-4-[(4-乙氧羰基-3-苯基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-ethoxycarbonyl-3-phenylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(1-吡咯烷基甲基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(1-pyrrolidinylmethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-苯基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-phenyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙基吡咯-2-基)亚甲基]-3-苯基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethylpyrrol-2-yl)methylene]-3-phenyl-2-pyrazolin-5-one,
4-[(3,4-二(乙氧羰基)吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,4-bis(ethoxycarbonyl)pyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(2-吡啶基乙基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(2-pyridylethyl)-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(1-甲基-2-苯乙基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(1-methyl-2-phenylethyl)-2-pyrazoline-5 -ketone,
4-[(4-羧基-3,5-二甲基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-carboxy-3,5-dimethylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(5-硝基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(5-nitropyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-[(4-硝基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(4-nitropyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(4-羧基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-carboxypyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-二甲氨基乙氨基羰基)吡咯-2-基)亚甲基}-3-乙氧基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethylaminocarbonyl)pyrrol-2-yl)methylene}-3-ethoxy-2-pyrazolin-5-one ,
4-[(5-氯-3-甲氧羰基-4-甲氧羰基甲基吡咯-2-基)亚甲基-3-乙氧基-2-吡唑啉-5-酮,4-[(5-chloro-3-methoxycarbonyl-4-methoxycarbonylmethylpyrrol-2-yl)methylene-3-ethoxy-2-pyrazolin-5-one,
4-{[3-(2-羧基乙基)-4-甲基吡咯-2-基)亚甲基}-3-乙氧基-2-吡唑啉-5-酮,4-{[3-(2-carboxyethyl)-4-methylpyrrol-2-yl)methylene}-3-ethoxy-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-乙氧基-4-[(4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-三氟甲基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-trifluoromethyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one,
3-(3-氨基苯基)-4-[(3,5-二甲基-4-乙氧羰基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-(3-aminophenyl)-4-[(3,5-dimethyl-4-ethoxycarbonylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-{[2-(4-吗啉代乙基)氨基羰基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-ethoxy-4-{[2-(4-morpholinoethyl)aminocarbonylpyrrol-2-yl]methylene}-2-pyrazolin-5-one,
4-[(4-乙氧羰基-3-苯基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-ethoxycarbonyl-3-phenylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(4-甲基-1-哌嗪基甲基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(4-Methyl-1-piperazinylmethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-哌啶子基甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-piperidinomethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazoline-5- ketone,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(5-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(5-isoxazolyl)-2-pyrazolin-5-one ,
4-[(5-甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(5-methyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3,5-二甲基-4-[N-(2-二甲氨基乙基)-N-甲氨基甲基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[N-(2-dimethylaminoethyl)-N-methylaminomethylpyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
3-环丙基-4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基-4,5-四亚甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethyl-4,5-tetramethylenepyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
3-环丙基-4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮马来酸盐,3-cyclopropyl-4-[(3,5-dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one maleate,
4-{[4-(3-氯丙酰基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(3-chloropropionyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基甲基吡咯-2-基)亚甲基]-3-(5-甲基-3-异噁唑基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminomethylpyrrol-2-yl)methylene]-3-(5-methyl-3-isoxazolyl)-2-pyrazole Lin-5-one,
4-{[3,5-二甲基-4-(4-羟基哌啶子基甲基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(4-hydroxypiperidinomethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-[(4-氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮马来酸盐,4-[(4-Aminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one maleate,
4-{[4-(4-苄基哌啶子基)甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(4-Benzylpiperidino)methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-羟基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-hydroxyethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3,5-二甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
3-环丙基-4-{{3,5-二甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3,5-dimethyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-2-pyrazoline- 5-keto,
3-环丙基-4-{[3,5-二甲基-4-(2-羟基乙基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3,5-dimethyl-4-(2-hydroxyethyl)pyrrol-2-yl]methylene}-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-乙氨基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(3-二乙氨基丙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(3-Diethylaminopropyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(3-羟丙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(3-hydroxypropyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3,5-二甲基-4-二甲氨基乙酰基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮盐酸盐,4-[(3,5-Dimethyl-4-dimethylaminoacetylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one hydrochloride,
4-[(3,5-二甲基-4-二甲氨基乙酰基吡咯-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-dimethylaminoacetylpyrrol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
3-环丙基-4-{[(2-二乙氨基乙基)-3,5-二甲基-4-吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[(2-diethylaminoethyl)-3,5-dimethyl-4-pyrrol-2-yl]methylene}-2-pyrazolin-5-one ,
4-{[3,5-二甲基-4-(2-二甲氨基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-dimethylaminoethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(4-羟基丁基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(4-hydroxybutyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(4-二乙氨基丁基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(4-diethylaminobutyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基-N-氧化物)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl-N-oxide)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[3,5-二甲基-4-(1-吡咯烷基乙酰基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(1-pyrrolidinylacetyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-二乙氨基乙酰基-3,5-二甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-Diethylaminoacetyl-3,5-dimethylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-[3-(1-吡咯烷基丙基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-[3-(1-pyrrolidinylpropyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-[(二甲氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(Dimethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
3-环丙基-4-[(二甲氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(dimethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-乙氧基草酰基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethoxyoxalyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
3-苄基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-benzyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-苄基-4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-2-吡唑啉-5-酮,3-Benzyl-4-{[1-(4-hydroxybutyl)indol-3-yl]methylene}-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲氧基苯氧基甲基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methoxyphenoxymethyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-苯氧基甲基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-phenoxymethyl-2-pyrazolin-5-one,
3-(4-氯苯氧基甲基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-chlorophenoxymethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲氧基苄基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methoxybenzyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲基苯氧基甲基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methylphenoxymethyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-苯乙基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-phenethyl-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲苄基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methylbenzyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-苯氨基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-anilino-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲基苯氨基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methylanilino)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲氧基苯氨基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methoxyanilino)-2-pyrazolin-5-one,
3-(4-氯苄基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-chlorobenzyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-(4-甲氧基苄基)-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-(4-methoxybenzyl)-2-pyrazolin-5-one,
3-(4-氯苯氨基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Chloroanilino)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(3-甲氧基苄基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(3-methoxybenzyl)-2-pyrazolin-5-one,
3-(3,4-二甲氧基苄基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(3,4-dimethoxybenzyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(4-羟基苄基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Hydroxybenzyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(3-氯苯氧基甲基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(3-Chlorophenoxymethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(吲哚-3-基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(indol-3-yl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲氧基苯乙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methoxyphenethyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-苯基苯氧基甲基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-phenylphenoxymethyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(2-苯基丙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(2-phenylpropyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(3-苯基丙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(3-phenylpropyl)-2-pyrazolin-5-one,
3-(4-羟基苯乙基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Hydroxyphenethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲基苯乙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methylphenethyl)-2-pyrazolin-5-one,
3-乙氧基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-isopropoxy-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-苯乙基-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-phenethyl-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-硝基苯乙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-nitrophenethyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-丙氧基-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-propoxy-2-pyrazolin-5-one,
3-(4-氟苯氨基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-fluoroanilino)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(4-氨基苯乙基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-aminophenethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(2-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(2-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(7-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(7-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(7-甲基吲哚-3-基)亚甲基]-3-苯乙基-2-吡唑啉-5-酮,4-[(7-methylindol-3-yl)methylene]-3-phenethyl-2-pyrazolin-5-one,
3-环戊基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopentyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲氧羰基苯乙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methoxycarbonylphenethyl)-2-pyrazolin-5-one,
3-异丙氧基-4-[(1-异丙基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(1-isopropylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-(4-羟基苯乙基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-hydroxyphenethyl)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-异丙基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(4-氯苯氨基羰基甲基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-Chloroanilinocarbonylmethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-乙氧羰基甲基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxycarbonylmethyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-甲氧基苯氨基羰基甲基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-methoxyanilinocarbonylmethyl)-2-pyrazolin-5-one,
3-环丙基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
3-(2-氨基羰基乙基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(2-Aminocarbonylethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-环丁基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclobutyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-(2-二甲氨基羰基乙基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(2-Dimethylaminocarbonylethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[1-(4-hydroxybutyl)indol-3-yl]methylene}-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(4-吡啶基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(4-pyridyl)-2-pyrazolin-5-one,
3-环戊基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopentyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(2,2,3,3-四甲基环丙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(2,2,3,3-tetramethylcyclopropyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(2-甲基环丙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(2-methylcyclopropyl)-2-pyrazolin-5-one,
3-环己基-4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-2-吡唑啉-5-酮,3-cyclohexyl-4-{[1-(4-hydroxybutyl)indol-3-yl]methylene}-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-(4-吡啶基)-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-(4-pyridyl)-2-pyrazolin-5-one,
3-(苯并噻唑-2-基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(Benzothiazol-2-yl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(2-吡啶基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(2-pyridyl)-2-pyrazolin-5-one,
3-二甲氨基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-Dimethylamino-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(吡咯-2-基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(pyrrol-2-yl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(6-甲氧基苯并噻唑-2-基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(6-methoxybenzothiazol-2-yl)-2-pyrazolin-5-one,
3-环丙基-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(7-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(7-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-丙基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-propyl-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-甲基-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-methyl-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-三氟甲基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-trifluoromethyl-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-甲基-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-methyl-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-三氟甲基-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-trifluoromethyl-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-3-(叔丁基)-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)indol-3-yl]methylene}-3-(tert-butyl)-2-pyrazolin-5-one,
3-乙氧羰基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxycarbonyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(2-甲氧羰基乙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(2-methoxycarbonylethyl)-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-(反-2-苯基-1-环丙基)-2-吡唑啉-5-酮,4-[(indol-3-yl)methylene]-3-(trans-2-phenyl-1-cyclopropyl)-2-pyrazolin-5-one,
3-环丁基-4-{[1-(4-羟基丁基)吲哚-3-基]亚甲基}-2-吡唑啉-5-酮,3-cyclobutyl-4-{[1-(4-hydroxybutyl)indol-3-yl]methylene}-2-pyrazolin-5-one,
4-[(6-羧基-1-甲基吲哚-3-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4-[(6-carboxy-1-methylindol-3-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
3-环丙基-4-[(5-甲氧基-1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-methoxy-1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基[-3-吡嗪基-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene[-3-pyrazinyl-2-pyrazolin-5-one,
3-环丙基-4-[(1,7-二甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(1,7-dimethylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-(四氢呋喃-3-基)-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-(tetrahydrofuran-3-yl)-2-pyrazolin-5-one,
3-环丙基氨基-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropylamino-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(6-羧基-1-甲基吲哚-3-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(6-carboxy-1-methylindol-3-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(1,7-二甲基吲哚-3-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(1,7-Dimethylindol-3-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-异丙氧基-4-[(5-甲氧基-1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(5-methoxy-1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-(4-氟苯氨基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-fluoroanilino)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-三氟乙酰氨基-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-trifluoroacetylamino-2-pyrazolin-5-one,
3-(4-氨基苯乙基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-aminophenethyl)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-氨基-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-amino-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-(4-氯苯乙基)-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(4-chlorophenethyl)-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
3-(5-异噁唑基)-4-[(1-甲基吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-(5-isoxazolyl)-4-[(1-methylindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(1-甲基吲哚-3-基)亚甲基]-3-叔丁基-2-吡唑啉-5-酮,4-[(1-methylindol-3-yl)methylene]-3-tert-butyl-2-pyrazolin-5-one,
4-[(6-羧基-1-甲基吲哚-3-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(6-carboxy-1-methylindol-3-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{{1-甲基-6-[2-(4-吗啉代)乙基]氨基羰基吲哚-3-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{1-methyl-6-[2-(4-morpholino)ethyl]aminocarbonylindol-3-yl}methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-[(吲哚-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(Indol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-(4-羟基苯乙基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-hydroxyphenethyl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氨基苯乙基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-aminophenethyl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-甲氧羰基苯乙基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-methoxycarbonylphenethyl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-(4-氨基甲酰基苯乙基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-carbamoylphenethyl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3-二甲氨基甲基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-dimethylaminomethylindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
3-异丙氧基-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(1-甲基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(1-methylindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-(四氢呋喃-3-基)-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-(tetrahydrofuran-3-yl)-2-pyrazolin-5-one,
3-环丙基-4-[(5-甲氧基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-methoxyindol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(5-硝基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(5-nitroindol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-异丙基氨基-2-吡唑啉-5-酮,4-[(Indol-2-yl)methylene]-3-isopropylamino-2-pyrazolin-5-one,
3-(4-羧基苯乙基)-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-(4-carboxyphenethyl)-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-2-基)亚甲基]-3-苯基-2-吡唑啉-5-酮,4-[(indol-2-yl)methylene]-3-phenyl-2-pyrazolin-5-one,
3-异丙氧基-4-[(3-甲基吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(3-methylindol-2-yl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-{[3-(2-甲氧羰基-2-乙酰氨基乙基)吲哚-2-基]亚甲基}-2-吡唑啉-5-酮,3-isopropoxy-4-{[3-(2-methoxycarbonyl-2-acetylaminoethyl)indol-2-yl]methylene}-2-pyrazolin-5-one,
3-乙氧基-4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮,3-ethoxy-4-[(indol-2-yl)methylene]-2-pyrazolin-5-one,
3-乙氧基-4-{[3-(4-吗啉代甲基)吲哚-2-基]亚甲基}-2-吡唑啉-5-酮,3-ethoxy-4-{[3-(4-morpholinomethyl)indol-2-yl]methylene}-2-pyrazolin-5-one,
3-环丙基-4-{[3-(4-吗啉代甲基)吲哚-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-(4-morpholinomethyl)indol-2-yl]methylene}-2-pyrazolin-5-one,
4-[(5-甲氧基吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(5-methoxyindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(5-甲氧基吲哚-2-基)亚甲基]-3-(2-吡啶基)-2-吡唑啉-5-酮,4-[(5-methoxyindol-2-yl)methylene]-3-(2-pyridyl)-2-pyrazolin-5-one,
4-[(4-氯-6,7-二氢吲哚-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(4-Chloro-6,7-dihydroindol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(4-氯-6,7-二氢吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-chloro-6,7-dihydroindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-氧代-1,5,6,7-二氢吲哚-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-oxo-1,5,6,7-dihydroindol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-[(3-二甲氨基甲基吲哚-2-基)亚甲基]-3-乙氧基-2-吡唑啉-5-酮,4-[(3-Dimethylaminomethylindol-2-yl)methylene]-3-ethoxy-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-苄基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-benzyl-2-pyrazolin-5-one,
3-苄基-4-{[1-(4-羟基丁基)-7-氮杂吲哚-3-基]亚甲基}-2-吡唑啉-5-酮,3-Benzyl-4-{[1-(4-hydroxybutyl)-7-azaindol-3-yl]methylene}-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(4-甲氧基苯氧基甲基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(4-methoxyphenoxymethyl)-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-氨基-4-[(7-氮杂吲哚-3-基)亚甲基]-2-吡唑啉-5-酮,3-amino-4-[(7-azaindol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(4-甲氧基苄基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(4-methoxybenzyl)-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-苯氨基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-anilino-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(4-甲苄基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(4-methylbenzyl)-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(4-羟基苯乙基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(4-hydroxyphenethyl)-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)-7-氮杂吲哚-3-基]亚甲基}-3-(4-羟基苯乙基)-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)-7-azaindol-3-yl]methylene}-3-(4-hydroxyphenethyl)-2-pyrazolin-5-one ,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-异丙基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-isopropyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-环丙基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-cyclopropyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-环丁基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-cyclobutyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(3-吡啶基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(3-pyridyl)-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-苯基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-phenyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(4-氟苯氨基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(4-fluoroanilino)-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-丙基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-propyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-甲基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-methyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-三氟甲基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-trifluoromethyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(叔丁基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(tert-butyl)-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)-7-氮杂吲哚-3-基]亚甲基}-3-三氟甲基-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)-7-azaindol-3-yl]methylene}-3-trifluoromethyl-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(4-异丙基苯氨基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(4-isopropylanilino)-2-pyrazolin-5-one,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-(3-甲基苯氨基)-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-(3-methylanilino)-2-pyrazolin-5-one,
4-{[1-(4-羟基丁基)-7-氮杂吲哚-3-基]亚甲基}-3-(3-甲基苯氨基)-2-吡唑啉-5-酮,4-{[1-(4-Hydroxybutyl)-7-azaindol-3-yl]methylene}-3-(3-methylanilino)-2-pyrazolin-5-one ,
4-[(7-氮杂吲哚-3-基)亚甲基]-3-苯乙基-2-吡唑啉-5-酮,4-[(7-azaindol-3-yl)methylene]-3-phenethyl-2-pyrazolin-5-one,
3-苄基-4-[(4-二甲氨基苯基)亚甲基]-2-吡唑啉-5-酮,3-benzyl-4-[(4-dimethylaminophenyl)methylene]-2-pyrazolin-5-one,
4-[(4-二甲氨基苯基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(4-Dimethylaminophenyl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-[(4-二甲氨基苯基)亚甲基]-3-苯乙基-2-吡唑啉-5-酮,4-[(4-Dimethylaminophenyl)methylene]-3-phenethyl-2-pyrazolin-5-one,
3-环丙基-4-[(4-二甲氨基苯基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-dimethylaminophenyl)methylene]-2-pyrazolin-5-one,
3-异丙氧基-4-[(3-甲基吡唑-4-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(3-methylpyrazol-4-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(3-甲基吡唑-4-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-methylpyrazol-4-yl)methylene]-2-pyrazolin-5-one,
4-[(咪唑-2-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(imidazol-2-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-环丙基-4-[(咪唑-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(imidazol-2-yl)methylene]-2-pyrazolin-5-one,
4-[(咪唑-4-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(imidazol-4-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
3-环丙基-4-[(噻吩-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(thiophen-2-yl)methylene]-2-pyrazolin-5-one,
4-[(吲哚-3-基)亚甲基]-3-异丙基-1-甲基-2-吡唑啉-5-酮,4-[(Indol-3-yl)methylene]-3-isopropyl-1-methyl-2-pyrazolin-5-one,
3-异丙氧基-4-[(吡咯-3-基)亚甲基]-2-吡唑啉-5-酮,3-isopropoxy-4-[(pyrrol-3-yl)methylene]-2-pyrazolin-5-one,
4-[(8-羟基喹啉-5-基)亚甲基]-3-异丙氧基-2-吡唑啉-5-酮,4-[(8-hydroxyquinolin-5-yl)methylene]-3-isopropoxy-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazoline-5 -ketone,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyrazoline- 5-keto,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazoline-5 -ketone,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline-5 -ketone,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazoline-5 -ketone,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazoline-5 -ketone,
4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrazoline- 5-keto,
3-环丙基-4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3,5-dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-2-pyrazolin-5-one ,
3-环丙基-4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3,5-dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-2-pyrazolin-5-one,
3-环丙基-4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3,5-dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-2-pyrazoline -5-keto,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-环丙基-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-cyclopropyl-2-pyrazolin-5-one,
3-环丙基-4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(4-diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-2-pyrazoline-5 -ketone,
3-环丙基-4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-2-pyr oxazolin-5-one,
3-环丙基-4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
3-环丙基-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-2-pyrrole oxazolin-5-one,
3-环丙基-4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
3-环丙基-4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-2- pyrazolin-5-one,
3-环丙基-4-{{4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{4-(4-dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-2-pyrazoline-5 -ketone,
3-环丙基-4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮,3-cyclopropyl-4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-2-pyrazolin-5-one,
3-环丙基-4-{[3-环丙基-4-(1-吡咯烷基甲基)-5-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-cyclopropyl-4-(1-pyrrolidinylmethyl)-5-methylpyrrol-2-yl]methylene}-2-pyrazoline-5 -ketone,
3-环丙基-4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-2-pyr oxazolin-5-one,
3-环丙基-4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-2 -pyrazolin-5-one,
3-环丙基-4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-2-pyrazoline-5 -ketone,
3-环丙基-4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl} Methylene}-2-pyrazolin-5-one,
3-环丙基-4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
3-环丙基-4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-2-pyr oxazolin-5-one,
3-环丙基-4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-cyclopropyl-4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-2-pyrazoline- 5-keto,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
3-吡嗪基-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-pyrazinyl-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-2-pyr oxazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
3-吡嗪基-4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-pyrazinyl-4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-2- pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-2-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-2-pyrazinyl-2 -pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -pyrazinyl-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazolin-5-one,
4-[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazoline-5 -ketone,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazoline-5 -ketone,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazoline-5- ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(2呋喃基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(2furyl)-2-pyridine oxazolin-5-one,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazoline-5- ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazole Lin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrazolin-5-one ,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(2-furyl)- 2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrrole oxazolin-5-one,
3-(2-呋喃基)-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(2-furyl)-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}- 2-pyrazolin-5-one,
3-(2-呋喃基)-4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-2-吡唑啉-5-酮,3-(2-furyl)-4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-2-pyridine oxazolin-5-one,
3-(2-呋喃基)-4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(2-furyl)-4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene} -2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2呋喃基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2furyl)-2-pyrazoline -5-keto,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(2-呋喃基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(2-furyl)-2-pyrazoline-5- ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(2-furyl)- 2-pyrazolin-5-one,
4-{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(2-furyl) -2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(2呋喃基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(2furyl)-2-pyrazoline -5-keto,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(2-furyl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(2-furyl)-2-pyrrole oxazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(2-furyl)- 2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(2-呋喃基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(2-furyl)-2-pyr oxazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline-5 -ketone,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazoline-5- ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(2-thienyl)-2- pyrazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazoline-5- ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazole Lin-5-one,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(2-thienyl)- 2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[4-(2-Cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrrole oxazolin-5-one,
4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(2-thienyl)- 2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrrole oxazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(2-thienyl) -2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazole Lin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(2-噻吩基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(2-thienyl)-2-pyrazoline-5- ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(2-thienyl)- 2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(2-thienyl )-2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrazole Lin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(2-thienyl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrrole oxazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(2-thienyl)- 2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(2-噻吩基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(2-thienyl)-2-pyrrole oxazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazoline-5 -ketone,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(4-pyrimidinyl)-2- pyrazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazole Lin-5-one,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(4-pyrimidinyl)- 2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrrole oxazolin-5-one,
3-(4-嘧啶基)-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(4-pyrimidinyl)-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}- 2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(4-pyrimidinyl) -2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazole Lin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(4-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(4-pyrimidinyl)- 2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(4-pyrimidinyl )-2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrazole Lin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(4-pyrimidinyl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(4-pyrimidinyl)- 2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(4-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(4-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazoline-5 -ketone,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(5-pyrimidinyl)-2- pyrazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazole Lin-5-one,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(5-pyrimidinyl)- 2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrrole oxazolin-5-one,
3-(5-嘧啶基)-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(5-pyrimidinyl)-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}- 2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(5-pyrimidinyl) -2-pyrazolin-5-one,
4-{{4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazole Lin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(5-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(5-pyrimidinyl)- 2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(5-pyrimidinyl )-2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrazole Lin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(5-pyrimidinyl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(5-pyrimidinyl)- 2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(5-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(5-pyrimidinyl)-2-pyr oxazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazoline-5 -ketone,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(2-pyrimidinyl)-2- pyrazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazolin-5-one ,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazole Lin-5-one,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(2-pyrimidinyl)- 2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrrole oxazolin-5-one,
3-(2-嘧啶基)-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(2-pyrimidinyl)-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}- 2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(2-pyrimidinyl) -2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazole Lin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(2-pyrimidinyl)-2-pyrazoline-5- ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(2-pyrimidinyl)- 2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(2-pyrimidinyl )-2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazole Lin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(2-pyrimidinyl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrrole oxazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(2-pyrimidinyl)- 2-pyrazolin-5-one,
4-[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(2-嘧啶基)-2-吡唑啉-5-酮,4-[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(2-pyrimidinyl)-2-pyrazole Lin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyrazoline -5-keto,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyrazoline-5 -ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(3-pyridazinyl)-2 -pyrazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyrazoline-5- ketone,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazoline-5 -ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyrrole oxazolin-5-one,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(3-pyridazinyl) -2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2- pyrazolin-5-one,
3-(3-哒嗪基)-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(3-pyridazinyl)-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene} -2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2- pyrazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(3-pyridazinyl )-2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyridine oxazolin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(3-pyridazinyl)-2-pyrazoline-5 -ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(3-pyridazinyl) -2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(3-pyridazine Base) -2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2-pyridine oxazolin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(3-pyridazinyl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2- pyrazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(3-pyridazinyl) -2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(3-哒嗪基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(3-pyridazinyl)-2- pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5 -ketone,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrazoline -5-keto,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrazoline- 5-keto,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrazoline-5 -ketone,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(4-pyridazinyl)-2 -pyrazolin-5-one,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5 -ketone,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrrole oxazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrazoline-5- ketone,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(4-pyridazinyl) -2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2- pyrazolin-5-one,
3-(4-哒嗪基)-4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-2-吡唑啉-5-酮,3-(4-pyridazinyl)-4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene} -2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2- pyrazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(4-pyridazinyl )-2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyrrole oxazolin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(4-pyridazinyl)-2-pyrazoline-5 -ketone,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(4-pyridazinyl) -2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-di(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(4-pyridazine Base) -2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2-pyridine oxazolin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(4-pyridazinyl)-2-pyrazolin-5-one,
4-([3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-([3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2- pyrazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(4-pyridazinyl) -2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(4-哒嗪基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(4-pyridazinyl)-2- pyrazolin-5-one,
4-[(3,5-二甲基-4-(1-吡咯烷基甲基吡咯-2-基)亚甲基]-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-[(3,5-Dimethyl-4-(1-pyrrolidinylmethylpyrrol-2-yl)methylene]-3-(1,2,4-triazin-3-yl)- 2-pyrazolin-5-one,
4-[(4-二乙氨基甲基-3,5-二甲基吡咯-2-基)亚甲基]-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3,5-dimethylpyrrol-2-yl)methylene]-3-(1,2,4-triazin-3-yl)-2-pyrrole oxazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(1,2,4-triazin-3-yl )-2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-乙氨基乙基)-吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-ethylaminoethyl)-pyrrol-2-yl]methylene}-3-(1,2,4-triazin-3-yl) -2-pyrazolin-5-one,
4-{[3,5-二甲基-4-(2-苯氨基乙基)吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[3,5-Dimethyl-4-(2-phenylaminoethyl)pyrrol-2-yl]methylene}-3-(1,2,4-triazin-3-yl)- 2-pyrazolin-5-one,
4-{{3,5-二甲基-4-[2-(4-吡啶基甲氨基)乙基]吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{3,5-Dimethyl-4-[2-(4-pyridylmethylamino)ethyl]pyrrol-2-yl}methylene}-3-(1,2,4-triazine -3-yl)-2-pyrazolin-5-one,
4-{[4-(2-羧乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[4-(2-carboxyethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-(1,2,4-triazin-3-yl)-2 -pyrazolin-5-one,
4-[(4-二乙氨基甲基-3-异丙基-5-甲基吡咯-2-基)亚甲基]-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-[(4-Diethylaminomethyl-3-isopropyl-5-methylpyrrol-2-yl)methylene]-3-(1,2,4-triazin-3-yl)- 2-pyrazolin-5-one,
4-{[4-(1-吡咯烷基甲基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[4-(1-pyrrolidinylmethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(1,2,4-triazine-3 -yl)-2-pyrazolin-5-one,
4-{{3-异丙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{3-isopropyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-(1,2,4- Triazin-3-yl)-2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(1,2,4-triazine- 3-yl)-2-pyrazolin-5-one,
4-{{4-[3-(1-吡咯烷基)丙基]-3-异丙基-5-甲基吡咯-2-基亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{4-[3-(1-pyrrolidinyl)propyl]-3-isopropyl-5-methylpyrrol-2-ylmethylene}-3-(1,2,4-tri Oxin-3-yl)-2-pyrazolin-5-one,
4-{[3-异丙基-5-甲基-4-(4-哌啶子基丁基)吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[3-isopropyl-5-methyl-4-(4-piperidinobutyl)pyrrol-2-yl]methylene}-3-(1,2,4-triazine- 3-yl)-2-pyrazolin-5-one,
4-{{4-[3-(4-吡啶基甲氨基)丙基]-3-异丙基-5-甲基吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{4-[3-(4-pyridylmethylamino)propyl]-3-isopropyl-5-methylpyrrol-2-yl}methylene}-3-(1,2,4 -Triazin-3-yl)-2-pyrazolin-5-one,
4-{[4-(4-二甲氨基丁基)-3-异丙基-5-甲基吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[4-(4-Dimethylaminobutyl)-3-isopropyl-5-methylpyrrol-2-yl]methylene}-3-(1,2,4-triazine-3 -yl)-2-pyrazolin-5-one,
4-[(3-环丙基-4-二乙氨基甲基-5-甲基吡咯-2-基)亚甲基]-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-[(3-cyclopropyl-4-diethylaminomethyl-5-methylpyrrol-2-yl)methylene]-3-(1,2,4-triazin-3-yl)- 2-pyrazolin-5-one,
4-{{3-环丙基-5-甲基-4-[2-(1-吡咯烷基)乙基]吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[2-(1-pyrrolidinyl)ethyl]pyrrol-2-yl}methylene}-3-(1,2,4- Triazin-3-yl)-2-pyrazolin-5-one,
4-{{3-环丙基-4-[2-二(甲氧基乙基)氨基乙基]-5-甲基吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[2-bis(methoxyethyl)aminoethyl]-5-methylpyrrol-2-yl}methylene}-3-(1,2, 4-triazin-3-yl)-2-pyrazolin-5-one,
4-{[3-环丙基-4-(3-二乙氨基丙基)-5-甲基吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-4-(3-diethylaminopropyl)-5-methylpyrrol-2-yl]methylene}-3-(1,2,4-triazine-3 -yl)-2-pyrazolin-5-one,
4-{{3-环丙基-4-[3-(2,5-二甲基-1-吡咯烷基)丙基]-5-甲基吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-4-[3-(2,5-dimethyl-1-pyrrolidinyl)propyl]-5-methylpyrrol-2-yl}methylene}-3 -(1,2,4-triazin-3-yl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(3-苯基甲氨基丙基)吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(3-phenylmethylaminopropyl)pyrrol-2-yl]methylene}-3-(1,2,4-triazine- 3-yl)-2-pyrazolin-5-one,
4-{{3-环丙基-5-甲基-4-[4-(1-吡咯烷基)丁基]吡咯-2-基}亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{{3-cyclopropyl-5-methyl-4-[4-(1-pyrrolidinyl)butyl]pyrrol-2-yl}methylene}-3-(1,2,4- Triazin-3-yl)-2-pyrazolin-5-one,
4-{[3-环丙基-5-甲基-4-(4-环丁基氨基丁基)吡咯-2-基]亚甲基}-3-(1,2,4-三嗪-3-基)-2-吡唑啉-5-酮,4-{[3-cyclopropyl-5-methyl-4-(4-cyclobutylaminobutyl)pyrrol-2-yl]methylene}-3-(1,2,4-triazine- 3-yl)-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{{5-甲基-4-[2-(1-吡咯烷基乙基)]-3-三氟甲基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(1-pyrrolidinylethyl)]-3-trifluoromethylpyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-甲基-4-[2-(4-吡啶基甲氨基乙基)]-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(4-pyridylmethylaminoethyl)]-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-三氟甲基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-trifluoromethylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrrole oxazolin-5-one,
4-{[4-(2-氨基乙基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-甲基-4-[2-(1-吡咯烷基乙基)]-3-叔丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(1-pyrrolidinylethyl)]-3-tert-butylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyr oxazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基)-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene)-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{5-甲基-4-[2-(4-吡啶基甲氨基乙基)]-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{5-methyl-4-[2-(4-pyridylmethylaminoethyl)]-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-叔丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-tert-butylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-氨基乙基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[4-(2-二乙氨基乙基)-3-乙基-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-3-ethyl-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-乙基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-Ethyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[3-乙基-4-(2-乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-Ethyl-4-(2-ethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{3-乙基-5-甲基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-Ethyl-5-methyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-乙基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-ethylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基乙基)-5-甲基-3-乙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-ethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3-环丁基-4-(2-二乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclobutyl-4-(2-diethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{3-环丁基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclobutyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[3-环丁基-4-(2-乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclobutyl-4-(2-ethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-环丁基-5-甲基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclobutyl-5-methyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-环丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-cyclobutylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-氨基乙基)-5-甲基-3-环丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-cyclobutylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[3-环戊基-4-(2-二乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopentyl-4-(2-diethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{3-环戊基-5-甲基-4-[2-(1-吡咯烷基乙基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopentyl-5-methyl-4-[2-(1-pyrrolidinylethyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[3-环戊基-4-(2-乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopentyl-4-(2-ethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-环戊基-5-甲基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopentyl-5-methyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-环戊基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-cyclopentylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-氨基乙基)-5-甲基-3-环戊基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-cyclopentylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[3-环己基-4-(2-二乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclohexyl-4-(2-diethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-环己基-5-甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclohexyl-5-methyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[3-环己基-4-(2-乙氨基乙基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclohexyl-4-(2-ethylaminoethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{3-环己基-5-甲基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclohexyl-5-methyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-环己基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-cyclohexylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基乙基)-5-甲基-3-环己基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-cyclohexylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{5-甲基-3-苯基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-phenyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{5-甲基-3-苯基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-phenyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-苯基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-phenylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基乙基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{5-甲基-3-(4-甲氧苯基)-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(4-methoxyphenyl)-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazine Base-2-pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-ethylaminoethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyr oxazolin-5-one,
4-{{5-甲基-3-(4-甲氧苯基)-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(4-methoxyphenyl)-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyr Azinyl-2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-(4-甲氧苯基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{[4-(2-氨基乙基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{5-甲基-3-(2-吡啶基)-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-pyridyl)-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-吡啶基)-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-pyridyl)-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-(2-吡啶基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-(2-pyridyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-氨基乙基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminoethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-甲基-3-吡嗪基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-pyrazinyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{5-甲基-3-吡嗪基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-pyrazinyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-吡嗪基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-pyrazinylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-环丙基氨基乙基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{5-甲基-3-(2-咪唑基)-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-imidazolyl)-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-咪唑基)-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-imidazolyl)-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-(2-咪唑基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-(2-imidazolyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[5-甲基-3-(2-嘧啶基)-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[5-methyl-3-(2-pyrimidinyl)-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-嘧啶基)-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-pyrimidinyl)-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-(2-嘧啶基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{5-甲基-3-(3-异噁唑基)-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(3-isoxazolyl)-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazine Base-2-pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-ethylaminoethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyr oxazolin-5-one,
4-{{5-甲基-3-(3-异噁唑基)-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(3-isoxazolyl)-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyridine Azinyl-2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-甲基-3-(3-异噁唑基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{5-甲基-3-(2-呋喃基)-4-[2-(1-吡咯烷基乙基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-furyl)-4-[2-(1-pyrrolidinylethyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-呋喃基)-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-furyl)-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{4-[2-(1-咪唑基)乙基]-5-甲基-3-(2-呋喃基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{4-[2-(1-imidazolyl)ethyl]-5-methyl-3-(2-furyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{{5-羟甲基-3-异丙基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-Hydroxymethyl-3-isopropyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-乙氨基乙基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminoethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-羟甲基-3-异丙基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-Hydroxymethyl-3-isopropyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-5-羟甲基-3-异丙基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-5-hydroxymethyl-3-isopropylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-环丙基氨基乙基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-[4-(2-二乙氨基乙基)-3,5-二环丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-[4-(2-Diethylaminoethyl)-3,5-dicyclopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3,5-二环丙基-4-[2-(1-吡咯烷基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-Dicyclopropyl-4-[2-(1-pyrrolidinylethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-乙氨基乙基)-3,5-二环丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-ethylaminoethyl)-3,5-dicyclopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3,5-二环丙基-4-[2-(4-吡啶基甲氨基乙基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-dicyclopropyl-4-[2-(4-pyridylmethylaminoethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{4-[2-(1-咪唑基)乙基]-3,5-二环丙基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)ethyl]-3,5-dicyclopropylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-环丙基氨基乙基)-3,5-二环丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminoethyl)-3,5-dicyclopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{{5-甲基-4-[2-(1-吡咯烷基甲基)]-3-三氟甲基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(1-pyrrolidinylmethyl)]-3-trifluoromethylpyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-甲基-4-[2-(4-吡啶基甲氨基甲基)]-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(4-pyridylmethylaminomethyl)]-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-三氟甲基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-trifluoromethylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrrole oxazolin-5-one,
4-{[4-(2-氨基甲基)-5-甲基-3-三氟甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-trifluoromethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-甲基-4-[2-(1-吡咯烷基甲基)]-3-叔丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(1-pyrrolidinylmethyl)]-3-tert-butylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{5-甲基-4-[2-(4-吡啶基甲氨基甲基)]-3-叔丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-4-[2-(4-pyridylmethylaminomethyl)]-3-tert-butylpyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{4-[2-(1-咪唑基)甲基]-5-甲基-3-叔丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{4-[2-(1-imidazolyl)methyl]-5-methyl-3-tert-butylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基甲基)-5-甲基-3-叔丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-tert-butylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[4-(2-二乙氨基甲基)-3-乙基-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-3-ethyl-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{[4-(2-二甲氨基甲基)-3-乙基-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-3-ethyl-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-乙基-5-甲基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-Ethyl-5-methyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[3-乙基-4-(2-乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-Ethyl-4-(2-ethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{3-乙基-5-甲基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-Ethyl-5-methyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-乙基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-ethylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基甲基)-5-甲基-3-乙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-ethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3-环丁基-4-(2-二乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclobutyl-4-(2-diethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{[3-环丁基-4-(2-二甲氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclobutyl-4-(2-dimethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{3-环丁基-5-甲基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclobutyl-5-methyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[3-环丁基-4-(2-乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclobutyl-4-(2-ethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-环丁基-5-甲基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclobutyl-5-methyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-环丁基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-cyclobutylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-氨基甲基)-5-甲基-3-环丁基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-cyclobutylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[3-环戊基-4-(2-二乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopentyl-4-(2-diethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{[3-环戊基-4-(2-二甲氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopentyl-4-(2-dimethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{3-环戊基-5-甲基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopentyl-5-methyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[3-环戊基-4-(2-乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopentyl-4-(2-ethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-环戊基-5-甲基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclopentyl-5-methyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-环戊基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-cyclopentylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-氨基甲基)-5-甲基-3-环戊基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-cyclopentylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[3-环己基-4-(2-二乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclohexyl-4-(2-diethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{[3-环己基-4-(2-二甲氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclohexyl-4-(2-dimethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{3-环己基-5-甲基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclohexyl-5-methyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[3-环己基-4-(2-乙氨基甲基)-5-甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclohexyl-4-(2-ethylaminomethyl)-5-methylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{3-环己基-5-甲基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3-cyclohexyl-5-methyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-环己基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-cyclohexylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基甲基)-5-甲基-3-环己基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-cyclohexylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{5-甲基-3-苯基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-phenyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{5-甲基-3-苯基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-phenyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-苯基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-phenylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-氨基甲基)-5-甲基-3-苯基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-phenylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{5-甲基-3-(4-甲氧苯基)-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(4-methoxyphenyl)-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazine Base-2-pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-ethylaminomethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyr oxazolin-5-one,
4-{{5-甲基-3-(4-甲氧苯基)-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(4-methoxyphenyl)-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyridine Azinyl-2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-(4-甲氧苯基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{[4-(2-氨基甲基)-5-甲基-3-(4-甲氧苯基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-(4-methoxyphenyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{5-甲基-3-(2-吡啶基)-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-pyridyl)-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-吡啶基)-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-pyridyl)-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-(2-吡啶基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-(2-pyridyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-氨基甲基)-5-甲基-3-(2-吡啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-aminomethyl)-5-methyl-3-(2-pyridyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-甲基-3-吡嗪基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-pyrazinyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5- ketone,
4-{{5-甲基-3-吡嗪基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-pyrazinyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-吡嗪基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-pyrazinylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-环丙基氨基甲基)-5-甲基-3-吡嗪基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminomethyl)-5-methyl-3-pyrazinylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{5-甲基-3-(2-咪唑基)-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-imidazolyl)-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-咪唑基)-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-imidazolyl)-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-(2-咪唑基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-(2-imidazolyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-环丙基氨基甲基)-5-甲基-3-(2-咪唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminomethyl)-5-methyl-3-(2-imidazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{5-甲基-3-(2-嘧啶基)-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{5-methyl-3-(2-pyrimidinyl)-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-嘧啶基)-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-pyrimidinyl)-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基}甲基]-5-甲基-3-(2-嘧啶基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl}methyl]-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-环丙基氨基甲基)-5-甲基-3-(2-嘧啶基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminomethyl)-5-methyl-3-(2-pyrimidinyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-二乙氨基乙基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminoethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-二甲氨基乙基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminoethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{{5-甲基-3-(3-异噁唑基)-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(3-isoxazolyl)-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazine Base-2-pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{{5-甲基-3-(3-异噁唑基)-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(3-isoxazolyl)-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyridine Azinyl-2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-(3-异噁唑基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{[4-(2-环丙基氨基甲基)-5-甲基-3-(3-异噁唑基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminomethyl)-5-methyl-3-(3-isoxazolyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-二乙氨基甲基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[4-(2-二甲氨基甲基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{5-甲基-3-(2-呋喃基)-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-furyl)-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl- 2-pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{{5-甲基-3-(2-呋喃基)-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-methyl-3-(2-furyl)-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl -2-pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-甲基-3-(2-呋喃基)吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-methyl-3-(2-furyl)pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{[4-(2-环丙基氨基甲基)-5-甲基-3-(2-呋喃基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminomethyl)-5-methyl-3-(2-furyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyridine oxazolin-5-one,
4-{[4-(2-二乙氨基甲基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[4-(2-二甲氨基甲基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{{5-羟甲基-3-异丙基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-Hydroxymethyl-3-isopropyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2- pyrazolin-5-one,
4-{[4-(2-乙氨基甲基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Ethylaminomethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone,
4-{{5-羟甲基-3-异丙基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{5-Hydroxymethyl-3-isopropyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2 -pyrazolin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-5-羟甲基-3-异丙基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-5-hydroxymethyl-3-isopropylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrrole oxazolin-5-one,
4-{[4-(2-环丙基氨基甲基)-5-羟甲基-3-异丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-cyclopropylaminomethyl)-5-hydroxymethyl-3-isopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-二乙氨基甲基)-3,5-二环丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Diethylaminomethyl)-3,5-dicyclopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{[4-(2-二甲氨基甲基)-3,5-二环丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-Dimethylaminomethyl)-3,5-dicyclopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one ,
4-{{3,5-二环丙基-4-[2-(1-吡咯烷基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-Dicyclopropyl-4-[2-(1-pyrrolidinylmethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline -5-keto,
4-{[4-(2-乙氨基甲基)-3,5-二环丙基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[4-(2-ethylaminomethyl)-3,5-dicyclopropylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazolin-5-one,
4-{{3,5-二环丙基-4-[2-(4-吡啶基甲氨基甲基)]吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{3,5-Dicyclopropyl-4-[2-(4-pyridylmethylaminomethyl)]pyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{{4-[2-(1-咪唑基)甲基]-3,5-二环丙基吡咯-2-基}亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{{4-[2-(1-imidazolyl)methyl]-3,5-dicyclopropylpyrrol-2-yl}methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-[(4-环丙基氨基甲基-3,5-二环丙基吡咯-2-基)亚甲基]-3-吡嗪基-2-吡唑啉-5-酮,4-[(4-cyclopropylaminomethyl-3,5-dicyclopropylpyrrol-2-yl)methylene]-3-pyrazinyl-2-pyrazolin-5-one,
4-{[3-环丙基氨基甲基-5-甲基-4-(2-二乙氨基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopropylaminomethyl-5-methyl-4-(2-diethylaminoethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazole Lin-5-one,
4-{[3-环丙基氨基甲基-5-甲基-4-(2-羟基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,4-{[3-cyclopropylaminomethyl-5-methyl-4-(2-hydroxyethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto,
4-{[3-二甲氨基甲基-5-甲基-4-(2-二乙氨基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮,和4-{[3-Dimethylaminomethyl-5-methyl-4-(2-diethylaminoethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline -5-keto, and
4-{[3-羟甲基-5-甲基-4-(2-二乙氨基乙基)吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮。4-{[3-Hydroxymethyl-5-methyl-4-(2-diethylaminoethyl)pyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline-5 -ketone.
式I化合物可以以与可药用酸的盐形式存在。本发明包括该盐。该盐的实例包括盐酸盐、氢溴酸盐、硫酸盐、甲磺酸盐、硝酸盐、马来酸盐、乙酸盐、柠檬酸盐、富马酸盐、酒石酸盐[例如(+)-酒石酸盐、(-)-酒石酸盐或其混合物,包括外消旋混合物]、琥珀酸盐、苯甲酸盐和与氨基酸如谷氨酸的盐。这些盐可以按照本领域专业人员已知的方法制备。Compounds of formula I may exist as salts with pharmaceutically acceptable acids. The present invention includes such salts. Examples of such salts include hydrochloride, hydrobromide, sulfate, methanesulfonate, nitrate, maleate, acetate, citrate, fumarate, tartrate [e.g. (+) - tartrates, (-)-tartrates or mixtures thereof, including racemic mixtures], succinates, benzoates and salts with amino acids such as glutamic acid. These salts can be prepared according to methods known to those skilled in the art.
某些具有酸性取代基的式I化合物可以以其与可药用碱的盐形式存在。本发明包括这些盐。该盐的实例包括钠盐、钾盐、赖氨酸盐和精氨酸盐。这些盐可以按照本领域专业人员已知的方法制备。Certain compounds of formula I having acidic substituents may exist as their salts with pharmaceutically acceptable bases. The present invention includes these salts. Examples of such salts include sodium salts, potassium salts, lysine salts and arginine salts. These salts can be prepared according to methods known to those skilled in the art.
某些式I化合物及其盐可以以多于一种晶形存在,并且本发明包括每种晶形及其混合物。Certain compounds of formula I and salts thereof may exist in more than one crystalline form, and the invention includes each crystalline form and mixtures thereof.
某些式I化合物及其盐还可以以溶剂化物、例如水合物的形式存在,并且本发明包括每种溶剂化物及其混合物。Certain compounds of formula I and their salts may also exist in the form of solvates, such as hydrates, and the present invention includes each solvate and mixtures thereof.
某些式I化合物可以含有一个或多个手性中心,并且以不同的旋光活性形式存在。当式I化合物含有一个手性中心时,该化合物以两种对映体的形式的存在,并且本发明包括这两种对映体以及它们的混合物。该两种对映体可以通过本领域专业人员已知的方法拆分,例如通过形成可以分离的非对映异构的盐,如通过结晶法;形成可以分离的非对映异构的衍生物或配合物,例如通过结晶法、气-液色谱或液相色谱;一种对映体与对映体专一的试剂进行选择性反应,例如酶促酯化;或在手性环境中进行气-液或液相色谱,例如在手性载体上,如结合有手性配体的硅胶,或者在手性溶剂存在下。显然,如果所需的对映体通过上述一种分离方法转化为另一种化学实体,则接下来的步骤必须是释放出所需的对映体形式。或者,可以通过使用旋光活性试剂、底物、催化剂或溶剂进行不对称合成,或者通过不对称转化将一种对映体转化为另一种对映体,合成特定的对映体。Certain compounds of formula I may contain one or more chiral centers and exist in different optically active forms. When a compound of formula I contains one chiral center, the compound exists as two enantiomers, and the present invention includes both enantiomers and mixtures thereof. The two enantiomers can be resolved by methods known to those skilled in the art, for example by forming separable diastereomeric salts, such as by crystallization; formation of separable diastereomeric derivatives or complexes, e.g. by crystallization, gas-liquid or liquid chromatography; selective reaction of one enantiomer with an enantiomer-specific reagent, e.g. enzymatic esterification; or gaseous - Liquid or liquid chromatography, for example on a chiral support, such as silica gel bound to a chiral ligand, or in the presence of a chiral solvent. Clearly, if the desired enantiomer is converted to another chemical entity by one of the separation methods described above, the next step must be to liberate the desired enantiomeric form. Alternatively, specific enantiomers may be synthesized by asymmetric synthesis using optically active reagents, substrates, catalysts or solvents, or by converting one enantiomer into the other by asymmetric transformation.
当式I化合物含有多于一个手性中心时,它可以以非对映异构形式存在。该非对映异构对可以按照本领域专业人员已知的方法分离,例如色谱或结晶,并且每一对中的单个对映体可以如上所述进行分离。本发明包括式I化合物的每个非对映异构体及其混合物。When a compound of formula I contains more than one chiral center, it may exist in diastereoisomeric forms. The diastereoisomeric pairs may be separated according to methods known to those skilled in the art, such as chromatography or crystallization, and the individual enantiomers of each pair may be separated as described above. The present invention includes each diastereoisomer of the compound of formula I and mixtures thereof.
某些式I化合物可以以不同的互变异构形式或不同的几何异构形式存在,并且本发明包括式I化合物的每种互变异构体和/或几何异构体及其混合物。Certain compounds of formula I may exist in different tautomeric forms or in different geometric isomeric forms, and the present invention includes each tautomer and/or geometric isomer of the compounds of formula I and mixtures thereof.
某些式I化合物可以以不同的可分离的稳定构象形式存在。扭转不对称是由于不对称单键周围受限制的旋转、例如由于位阻或环张力所致,它使得可以分离不同的构象异构体。本发明包括式I化合物的每种构象异构体及其混合物。Certain compounds of formula I may exist in different separable stable conformational forms. Torsional asymmetry is due to restricted rotation around an asymmetric single bond, for example due to steric hindrance or ring strain, which allows the separation of different conformers. The present invention includes each conformational isomer of the compound of formula I and mixtures thereof.
某些式I化合物可以以两性离子形式存在,并且本发明包括式I化合物的每种两性离子形式及其混合物。Certain compounds of formula I may exist in zwitterionic form, and the present invention includes each zwitterionic form of compounds of formula I and mixtures thereof.
本发明化合物适于用作丝氨酸/苏氨酸和酪氨酸激酶抑制剂。具体地说,本发明化合物适于用作酪氨酸激酶抑制剂,酪氨酸激酶在过度增殖疾病、特别是在血管生成的过程中是重要的。例如,其中一些化合物是受体激酶如KDR、Flt-1、FGFR、PDGFR、c-Met或IGF-1-R的抑制剂。由于这些化合物是抗-血管生成药,它们对于抑制其中血管生成是重要组成的进行性疾病是重要物质。某些本发明的化合物是有效的丝氨酸/苏氨酸激酶如erk、MAP激酶、cdks、Plk-1或Raf-1抑制剂。这些化合物适用于治疗癌和过度增殖疾病。此外,某些化合物是有效的非受体激酶如src、lyn、lck、fyn、blk、hck抑制剂。这些化合物适用于治疗癌、过度增殖疾病和免疫疾病。The compounds of the invention are suitable as inhibitors of serine/threonine and tyrosine kinases. In particular, the compounds of the invention are suitable as inhibitors of tyrosine kinases which are important in hyperproliferative diseases, especially in the process of angiogenesis. For example, some of these compounds are inhibitors of receptor kinases such as KDR, Flt-1, FGFR, PDGFR, c-Met or IGF-1-R. Since these compounds are anti-angiogenic agents, they are important substances for inhibiting progressive diseases in which angiogenesis is an important component. Certain compounds of the invention are potent inhibitors of serine/threonine kinases such as erk, MAP kinase, cdks, Plk-1 or Raf-1. These compounds are useful in the treatment of cancer and hyperproliferative diseases. In addition, certain compounds are potent inhibitors of non-receptor kinases such as src, lyn, lck, fyn, blk, hck. These compounds are useful in the treatment of cancer, hyperproliferative diseases and immune diseases.
本发明提供了抑制酪氨酸激酶和丝氨酸/苏氨酸激酶活性的方法,该方法包括将足以抑制所述激酶活性浓度的式I化合物施用到所述激酶上。The present invention provides methods of inhibiting the activity of tyrosine kinases and serine/threonine kinases comprising administering to said kinases a compound of formula I at a concentration sufficient to inhibit the activity of said kinases.
本发明还包括这些化合物在含有药学有效量的上述化合物和可药用载体或赋形剂的药物组合物中的应用。这些药物组合物可以对个体给药以减缓或停止血管生成帮助的疾病中的血管生成过程,或者治疗水肿、渗漏、渗出液或腹水以及其他与血管渗透性过高有关的疾病。某些药物组合物可以对个体给药,以通过抑制丝氨酸/苏氨酸激酶如cdk、Plk-1、erk等治疗癌和过度增殖疾病。The present invention also includes the application of these compounds in a pharmaceutical composition containing a pharmaceutically effective amount of the above compound and a pharmaceutically acceptable carrier or excipient. These pharmaceutical compositions can be administered to an individual to slow or stop the angiogenic process in angiogenesis-assisted diseases, or to treat edema, leakage, exudate or ascites and other diseases associated with vascular hyperpermeability. Certain pharmaceutical compositions can be administered to individuals to treat cancer and hyperproliferative diseases by inhibiting serine/threonine kinases such as cdk, Plk-1, erk, and the like.
发明详述Detailed description of the invention
本发明化合物具有抗血管生成性质。这些抗血管生成性质是由于至少部分抑制血管生成过程必需的蛋白质酪氨酸激酶产生的。因此,这些化合物可以用作对抗这些疾病的活性药物,所述疾病是例如关节炎、动脉粥样硬化、牛皮癣、血管瘤、心肌血管生成、冠状和脑侧突、局部缺血性四肢血管生成、创伤愈合、消化性溃疡、与螺旋杆菌有关的疾病、病毒引起的血管生成疾病、骨折、Crow-Fukase综合征(POEMS)、先兆子痫、月经过多、猫抓伤引起的发烧、潮红、新血管青光眼和视网膜病如与糖尿病性视网膜病、早熟视网膜病或与年龄相关的黄斑退行性改变有关的疾病。此外,其中一些化合物可用作抗实体瘤、恶性腹水、造血癌(hematopoietic cancers)和过度增殖疾病如 甲状腺增生(特别是格雷夫斯病)和囊肿(如卵巢间质特征的多囊性卵巢综合征(斯-勒综合征))的活性药物,因为这些疾病需要血管细胞的增殖以供其生长和/或转移。The compounds of the invention possess anti-angiogenic properties. These anti-angiogenic properties result from at least partial inhibition of protein tyrosine kinases essential for the angiogenic process. These compounds can therefore be used as active drugs against diseases such as arthritis, atherosclerosis, psoriasis, hemangiomas, myocardial angiogenesis, coronary and lateral processes, ischemic extremity angiogenesis, Wound healing, peptic ulcer, diseases associated with Helicobacter, angiogenic diseases caused by viruses, bone fractures, Crow-Fukase syndrome (POEMS), preeclampsia, menorrhagia, fever from cat scratches, flushing, new Vascular glaucoma and retinopathy such as those associated with diabetic retinopathy, retinopathy of prematurity, or age-related macular degeneration. In addition, some of these compounds are useful against solid tumors, malignant ascites, hematopoietic cancers and hyperproliferative diseases such as thyroid hyperplasia (especially Graves' disease) and cysts (such as polycystic ovary syndrome characterized by ovarian stroma). Syndrome (Stiller Syndrome)) because these diseases require the proliferation of vascular cells for their growth and/or metastasis.
再者,某些化合物可以用作治疗下述疾病的活性药物:灼伤、慢性肺病、中风、息肉、过敏症、慢性和变应性炎症、延迟型过敏、卵巢过度刺激综合征、与脑肿瘤有关的脑水肿、high-altitude、创伤或氧不足引起的脑或肺水肿、眼和黄斑水肿、腹水和其他其中以血管渗透性过高、渗漏、渗出液、蛋白质外渗或水肿为疾病表现的疾病。这些化合物还适用于治疗其中蛋白质外渗导致纤维蛋白和细胞外基质沉积、促进基质增殖的疾病(例如瘢痕瘤,纤维变性,肝硬变和脘管综合征)。VEGF产生增加导致炎性过程加强,例如单核细胞募集与活化。本发明化合物还适用于治疗炎性疾病如炎性肠疾病(IBD)和节段性回肠炎。Furthermore, certain compounds can be used as active drugs in the treatment of burns, chronic lung disease, stroke, polyps, allergies, chronic and allergic inflammation, delayed-type allergy, ovarian hyperstimulation syndrome, associated with brain tumors Cerebral edema, high-altitude, cerebral or pulmonary edema due to trauma or hypoxia, ocular and macular edema, ascites, and other disorders in which vascular hyperpermeability, leakage, exudate, protein extravasation, or edema are manifestations disease. These compounds are also useful in the treatment of diseases in which protein extravasation leads to deposition of fibrin and extracellular matrix, promoting matrix proliferation (eg keloids, fibrosis, cirrhosis and gastric canal syndrome). Increased VEGF production leads to enhanced inflammatory processes, such as monocyte recruitment and activation. The compounds of the invention are also useful in the treatment of inflammatory diseases such as inflammatory bowel disease (IBD) and Crohn's disease.
VEGF是独一无二的,它们是目前仅知的导致血管渗透性过高和水肿形成的血管生成生长因子。实际上,与许多其他生长因子的表达和给药有关的血管渗透性过高和水肿的出现,是由VEGF产生介导的。炎性细胞因子刺激VEGF产生。氧不足导致在多种组织中VEGF的明显上调,因此与梗塞、闭塞、局部缺血、贫血或循环减少有关的情境通常引起VEGF/VPF介导的反应。血管渗透性过高、联合水肿,改变的透内皮交换和大分子外渗(其中经常伴随有血细胞渗出)可以导致基质过度沉积、异常的间质增殖、纤维变性等。因此,VEGF介导的渗透性过高对具有这些病因学特征的病症有重要的贡献。VEGF are unique in that they are the only angiogenic growth factors known to cause vascular hyperpermeability and edema formation. Indeed, the appearance of vascular hyperpermeability and edema associated with the expression and administration of many other growth factors is mediated by VEGF production. Inflammatory cytokines stimulate VEGF production. Hypoxia results in a marked upregulation of VEGF in a variety of tissues, thus situations associated with infarction, occlusion, ischemia, anemia or decreased circulation often elicit VEGF/VPF mediated responses. Vascular hyperpermeability, combined with edema, altered transendothelial exchange, and macromolecular extravasation (often accompanied by extravasation of blood cells) can lead to excessive deposition of matrix, abnormal interstitial proliferation, fibrosis, etc. Thus, VEGF-mediated hyperpermeability has an important contribution to disorders with these etiological features.
应该看到,上面列出的疾病由涉及KDR/VEGFR-2和/或Flt-1/VEGFR-1酪氨酸激酶的蛋白质酪氨酸激酶活性介导至重要的程度。通过抑制这些酪氨酸激酶的活性,上述疾病的进行得以抑制,因为疾病状况的血管生成或血管渗透性过高的成分被严格缩减。某些本发明化合物的作用,通过其对特定酪氨酸激酶的选择性,使得使用低选择性酪氨酸激酶抑制剂出现的副作用减到最小限度。某些本发明化合物还是有效的FGFR、PDGFR、c-Met和IGF-1-R抑制剂。这些受体激酶可以直接或间接加强各种疾病中的血管生成和过度增殖应答,因而其抑制作用可以阻止疾病进行。It should be appreciated that the diseases listed above are mediated to a significant extent by protein tyrosine kinase activity involving KDR/VEGFR-2 and/or Flt-1/VEGFR-1 tyrosine kinases. By inhibiting the activity of these tyrosine kinases, the progression of the aforementioned diseases is inhibited because the angiogenic or vascular hyperpermeable components of the disease state are strictly curtailed. The action of certain compounds of the invention, through their selectivity for specific tyrosine kinases, minimizes the side effects that occur with less selective tyrosine kinase inhibitors. Certain compounds of the invention are also potent inhibitors of FGFR, PDGFR, c-Met and IGF-1-R. These receptor kinases can directly or indirectly potentiate angiogenic and hyperproliferative responses in various diseases so that their inhibition can prevent disease progression.
本发明化合物对蛋白激酶具有抑制活性。即,这些化合物通过蛋白激酶调控信号传导。本发明化合物抑制丝氨酸/苏氨酸和酪氨酸激酶类的蛋白激酶。尤其是,这些化合物选择性地抑制KDR/FLK-1/VEGFR-2酪氨酸激酶活性。某些本发明化合物还抑制其他酪氨酸激酶如Flt-1/VEGFR-1、FGFR、PDGFR、IGF-1R、c-Met、Src-亚族的激酶如Lck、Src、fyn、yes等的活性。另外,某些本发明化合物显著抑制在细胞增殖和细胞周期进行中起重要作用的丝氨酸/苏氨酸激酶如PKC、MAP激酶、erk、CDKs、Plk-1或Raf-1。本发明通式化合物针对特定蛋白激酶的功效和特异性通常可以通过改变取代基(即R1、R2、R3、R4、R5、R6和R7)的性质、数量和排列和构象限制而变化或最佳化。另外,某些化合物的代谢物也具有显著的蛋白质激酶抑制活性。The compounds of the present invention have inhibitory activity against protein kinases. That is, these compounds modulate signaling through protein kinases. The compounds of the present invention inhibit protein kinases of the serine/threonine and tyrosine kinase classes. In particular, these compounds selectively inhibit KDR/FLK-1/VEGFR-2 tyrosine kinase activity. Certain compounds of the present invention also inhibit the activity of other tyrosine kinases such as Flt-1/VEGFR-1, FGFR, PDGFR, IGF-1R, c-Met, Src-subfamily kinases such as Lck, Src, fyn, yes, etc. . In addition, certain compounds of the present invention significantly inhibit serine/threonine kinases such as PKC, MAP kinase, erk, CDKs, Plk-1 or Raf-1 that play an important role in cell proliferation and cell cycle progression. The potency and specificity of the compounds of the general formula of the present invention against specific protein kinases can usually be changed by changing the nature, number and arrangement of the substituents (ie R 1 , R 2 , R 3 , R 4 , R 5 , R 6 and R 7 ) and Change or optimization due to conformational constraints. In addition, metabolites of certain compounds also possess significant protein kinase inhibitory activity.
当对需要的个体施用本发明化合物时,本发明化合物抑制这些个体的血管渗透性过高和水肿形成。据信这些化合物通过抑制与血管渗透性过高和水肿形成过程有关的KDR酪氨酸激酶活性发生作用。KDR酪氨酸激酶又被称作FLK-1酪氨酸激酶、NYK酪氨酸激酶或VEGFR-2酪氨酸激酶。当血管内皮细胞生长因子(VEGF)或另一种活化的配体(例如VEGF-C、VEGF-D、VEGF-E或HIV Tat蛋白)与血管内皮细胞表面上的KDR酪氨酸激酶受体结合时,KDR酪氨酸激酶被活化。在该KDR酪氨酸激酶活化后,血管渗透性过高发生,液体从血液中经血管壁进入间质腔,从而形成水肿区域。血细胞渗出也经常伴有该反应。类似地,过度血管渗透性过高可以打破危象组织和器官(例如肺和肾)中通过内皮的正常分子交换,从而引起大分子外渗和沉积。在对KDR刺激的急性应答(据信这促进了随后的血管生成过程)后,延长的KDR酪氨酸激酶刺激造成血管内皮细胞的增殖和趋化性以及新血管形成。通过抑制KDR酪氨酸激酶活性,或者通过阻断活化配体的产生、通过阻断活化配体与KDR酪氨酸激酶受体结合、通过阻止受体二聚和磷酸根转移、通过抑制KDR酪氨酸激酶活性(抑制酶的磷酸化功能)或者通过某些其他阻断其下游信号传导的机理(D.Mukhopedhyay等,CancerRes.58:1278-1284(1998),该文献结合在本文中作为参考),渗透性过高和有关的外渗、随后的水肿形成和基质沉积以及血管生成反应可以被抑制或最小化。When administered to individuals in need thereof, the compounds of the invention inhibit vascular hyperpermeability and edema formation in these individuals. These compounds are believed to act by inhibiting the activity of the KDR tyrosine kinase involved in the process of vascular hyperpermeability and edema formation. KDR tyrosine kinase is also known as FLK-1 tyrosine kinase, NYK tyrosine kinase or VEGFR-2 tyrosine kinase. When vascular endothelial growth factor (VEGF) or another activating ligand (such as VEGF-C, VEGF-D, VEGF-E, or HIV Tat protein) binds to the KDR tyrosine kinase receptor on the surface of vascular endothelial cells , KDR tyrosine kinase is activated. After activation of this KDR tyrosine kinase, vascular hyperpermeability occurs and fluid passes from the blood through the vessel wall into the interstitial cavity, thereby forming an edematous area. Exudation of blood cells is also often accompanied by this reaction. Similarly, excessive vascular hyperpermeability can disrupt normal molecular exchange across the endothelium in at-risk tissues and organs (eg, lung and kidney), causing extravasation and deposition of macromolecules. Following an acute response to KDR stimulation, which is believed to promote the subsequent angiogenic process, prolonged KDR tyrosine kinase stimulation results in proliferation and chemotaxis of vascular endothelial cells and neovascularization. By inhibiting KDR tyrosine kinase activity, or by blocking the production of activating ligands, by blocking the binding of activating ligands to KDR tyrosine kinase receptors, by preventing receptor dimerization and phosphate transfer, by inhibiting KDR tyrosine kinase amino acid kinase activity (inhibition of the phosphorylation function of the enzyme) or by some other mechanism that blocks its downstream signaling (D. Mukhopedhyay et al., Cancer Res. 58: 1278-1284 (1998), which is incorporated herein by reference ), hyperosmolarity and associated extravasation, subsequent edema formation and matrix deposition, and angiogenic responses can be inhibited or minimized.
一组优选的本发明化合物具有抑制KDR酪氨酸激酶活性、而不显著抑制Flt-1酪氨酸激酶活性(Flt-1酪氨酸激酶也称作VEGFR-1酪氨酸激酶)。KDR酪氨酸激酶和Flt-1酪氨酸激酶分别被与KDR酪氨酸激酶受体和与Flt-1酪氨酸激酶受体结合的VEGF活化。由于Flt-1酪氨酸激酶活性在内皮养护和血管功能中可以介导重要的结果,因此该酶活性的抑制可以导致毒性或不利的作用。至少该抑制对于阻断血管生成应答、诱导血管渗透性过高和水肿形成是不必要的,因此对于个体是不经济和没有价值的。某些优选的本发明化合物是独特的,因为它们抑制通过活化配体活化的VEGF-受体酪氨酸激酶(KDR)活性,而不抑制其他也被某些活化配体活化的受体酪氨酸激酶,例如Flt-1。因此,优选的本发明化合物在其酪氨酸激酶抑制活性上具有选择性。A preferred group of compounds of the invention have the ability to inhibit KDR tyrosine kinase activity without significantly inhibiting Flt-1 tyrosine kinase activity (Flt-1 tyrosine kinase is also known as VEGFR-1 tyrosine kinase). KDR tyrosine kinase and Flt-1 tyrosine kinase are activated by VEGF binding to KDR tyrosine kinase receptor and Flt-1 tyrosine kinase receptor, respectively. Since Flt-1 tyrosine kinase activity can mediate important consequences in endothelial maintenance and vascular function, inhibition of this enzyme activity can lead to toxic or adverse effects. At least this inhibition is unnecessary to block the angiogenic response, induce vascular hyperpermeability and edema formation, and thus be uneconomical and of no value to the individual. Certain preferred compounds of the invention are unique in that they inhibit the activity of VEGF-receptor tyrosine kinase (KDR) activated by activating ligands, while not inhibiting other receptor tyrosine kinases that are also activated by certain activating ligands Acid kinases, such as Flt-1. Accordingly, preferred compounds of the invention are selective in their tyrosine kinase inhibitory activity.
本发明化合物还适用于治疗溃疡-细菌、真菌、莫伦氏溃疡和溃疡性结肠炎。The compounds of the present invention are also useful in the treatment of ulcers - bacterial, fungal, Moran's ulcer and ulcerative colitis.
本发明化合物还适用于治疗其中在病毒感染中出现不希望的血管生成、水肿或间质沉积的疾病,例如单纯性疱疹、带状疱疹、AIDS、parapoxvirus、牛皮癣、卡波济氏肉瘤、原生动物感染和弓形体病、子宫内膜异位、卵巢过度刺激综合征、先兆子痫、月经过多、全身性红斑狼疮、肉样瘤病、滑膜炎、炎性肠疾病、节段性回肠炎、镰刀细胞贫血、莱姆病、类天疱疮、佩吉特氏病、粘滞性过高综合征、Osler-Weber-Rendu病、慢性炎症、慢性闭合性肺病、气喘、类风湿性关节炎和骨关节炎、以及灼伤、外伤、放射或中风后水肿。The compounds of the invention are also suitable for the treatment of diseases in which undesired angiogenesis, edema or interstitial deposition occur in viral infections, such as herpes simplex, herpes zoster, AIDS, parapoxvirus, psoriasis, Kaposi's sarcoma, protozoa Infections and toxoplasmosis, endometriosis, ovarian hyperstimulation syndrome, preeclampsia, menorrhagia, systemic lupus erythematosus, sarcoidosis, synovitis, inflammatory bowel disease, Crohn's disease , sickle cell anemia, Lyme disease, pemphigoid, Paget's disease, hyperviscosity syndrome, Osler-Weber-Rendu disease, chronic inflammation, chronic closed lung disease, asthma, rheumatoid arthritis and osteoarthritis, and edema after burns, trauma, radiation, or stroke.
除了视网膜病和黄斑变性之外,本发明化合物还适用于治疗眼病例如眼和黄斑水肿、眼新血管病、巩膜炎、放射状角膜切开术、眼色素层炎、玻璃体炎、近视、眼凹、慢性视网膜剥离、激光术后并发症、结膜炎、眼底黄色斑点症和伊耳斯氏病。In addition to retinopathy and macular degeneration, the compounds of the present invention are also useful in the treatment of eye diseases such as ocular and macular edema, ocular neovascular disease, scleritis, radial keratotomy, uveitis, vitreitis, myopia, sunken eyes, Chronic retinal detachment, laser postoperative complications, conjunctivitis, xanthomatosis and Earl's disease.
本发明化合物还适用于治疗心血管疾病例如动脉粥样硬化、再狭窄、血管闭合和颈动脉阻塞疾病。The compounds of the invention are also useful in the treatment of cardiovascular diseases such as atherosclerosis, restenosis, vascular occlusion and carotid occlusive disease.
本发明化合物还适用于治疗与癌有关的适应征例如实体瘤、肉瘤(特别是尤因氏肉瘤和骨肉瘤)、成视网膜细胞瘤、横纹肌肉瘤、成神经细胞瘤、造血恶性肿瘤包括白血病和淋巴瘤、肿瘤引起的胸膜或心包积液以及恶性腹水。The compounds of the invention are also useful in the treatment of cancer-related indications such as solid tumors, sarcomas (especially Ewing's sarcoma and osteosarcoma), retinoblastoma, rhabdomyosarcoma, neuroblastoma, hematopoietic malignancies including leukemia and lymphoid Tumors, pleural or pericardial effusions caused by tumors, and malignant ascites.
本发明化合物还适用于治疗Crow-Fukase(POEMS)综合征和糖尿病性疾病如青光眼、糖尿病性视网膜病和微血管病。The compounds of the invention are also useful in the treatment of Crow-Fukase (POEMS) syndrome and diabetic diseases such as glaucoma, diabetic retinopathy and microangiopathy.
应该看到,上面列出的疾病由涉及VEGF受体(例如KDR和Flt-1)的蛋白质酪氨酸激酶活性介导至重要的程度。通过抑制这些受体酪氨酸激酶的活性,上述疾病的进行得以抑制,因为疾病状况的血管生成成分被严格缩减。本发明化合物的作用,通过其对特定酪氨酸激酶的选择性,使得使用低选择性酪氨酸激酶抑制剂出现的副作用减到最小限度。It should be appreciated that the diseases listed above are mediated to a significant extent by the activity of protein tyrosine kinases involving VEGF receptors such as KDR and Flt-1. By inhibiting the activity of these receptor tyrosine kinases, the progression of the aforementioned diseases is inhibited because the angiogenic component of the disease state is strictly curtailed. The action of the compounds of the invention, through their selectivity for specific tyrosine kinases, minimizes the side effects that occur with less selective tyrosine kinase inhibitors.
另一方面,本发明提供了用作药物、特别是用作蛋白激酶活性、例如酪氨酸激酶活性、丝氨酸激酶活性和苏氨酸激酶活性抑制剂的上面开始时定义的式I化合物(包括附带条件)。再一方面,本发明提供了上面开始时定义的式I化合物(包括附带条件)在制备用于抑制蛋白激酶活性的药物中的应用。In another aspect, the present invention provides compounds of the formula I defined at the outset above (including the accompanying condition). In a further aspect, the present invention provides the use of a compound of formula I defined at the outset above (including provisos) for the manufacture of a medicament for inhibiting protein kinase activity.
在本发明中,使用下列定义:In the present invention, the following definitions are used:
″生理上可接受的盐″是指那些保持了游离碱的生物效力和性质,并且通过与无机酸如盐酸、氢溴酸、硫酸、硝酸、磷酸或有机酸如磺酸、羧酸、有机磷酸、甲磺酸、乙磺酸、对甲苯磺酸、水杨酸、乳酸和酒石酸等反应得到的盐。"Physiologically acceptable salts" refer to those that retain the biological potency and properties of the free base, and are obtained by mixing with inorganic acids such as hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid or organic acids such as sulfonic acid, carboxylic acid, organic phosphoric acid. , methanesulfonic acid, ethanesulfonic acid, p-toluenesulfonic acid, salicylic acid, lactic acid and tartaric acid, etc.
″烷基″是指饱和脂族烃,包括具有1-6个碳原子的直链和支链基团或具有3-6个碳原子的环烃。"Alkyl" means a saturated aliphatic hydrocarbon, including straight and branched chain groups having 1-6 carbon atoms or cyclic hydrocarbons having 3-6 carbon atoms.
″烷氧基″是指″O-烷基″,其中″烷基″如上所定义。"Alkoxy" means "O-alkyl", wherein "alkyl" is as defined above.
药物制剂pharmaceutical preparations
本发明化合物可以以其本身或者在药物组合物中对人类患者给药,在药物组合物中该化合物与合适的载体或赋形剂以治疗或改善血管渗透性过高、水肿和有关疾病的剂量混合。还可以将这些化合物的混合物以单纯的混合物或在合适的经配制的药物组合物中对患者给药。治疗有效剂量进一步是指足以预防或减弱不适当的新血管形成、过度增殖疾病进行、水肿、VEGF有关的渗透性过高和/或VEGF有关的低血压的化合物的量。配制和施用本申请化合物的技术可见于″Remington′s Pharmaceutical Sciences,″Mack Publishing Co.,Easton,PA,最近的版本。The compound of the present invention can be administered to human patients by itself or in a pharmaceutical composition in which the compound is combined with a suitable carrier or excipient to treat or improve vascular hyperpermeability, edema and related diseases. mix. Mixtures of these compounds may also be administered to the patient as a pure mixture or in suitable formulated pharmaceutical compositions. A therapeutically effective dose further refers to an amount of compound sufficient to prevent or attenuate inappropriate neovascularization, hyperproliferative disease progression, edema, VEGF-related hyperosmolarity and/or VEGF-related hypotension. Techniques for formulating and administering the compounds of the present application can be found in "Remington's Pharmaceutical Sciences," Mack Publishing Co., Easton, PA, most recent edition.
给药途径Route of administration
合适的给药途径可以例如包括经口、滴眼、直肠、经粘膜、局部或经肠给药;非肠道给药包括肌内、皮下、髓内注射以及鞘内、心室内直接注射、静脉内、腹膜内、鼻内或眼内注射。Suitable routes of administration may, for example, include oral, eye drops, rectal, transmucosal, topical or enteral administration; parenteral administration includes intramuscular, subcutaneous, intramedullary injection and intrathecal, intraventricular direct injection, intravenous Intraperitoneal, intranasal or intraocular injection.
或者,可以局部而非全身施用该化合物,例如将化合物直接注射到水肿部位,通常是以长效制剂或持续释放制剂的形式给药。Alternatively, the compound may be administered locally rather than systemically, for example by injecting the compound directly into the site of edema, usually in a depot or sustained release formulation.
再者,可以将药物以靶向药物释放系统给药,例如用内皮细胞特异性抗体包衣的脂质体。Furthermore, the drug can be administered in a targeted drug delivery system, such as liposomes coated with endothelial cell-specific antibodies.
组合物/制剂Composition/Preparation
本发明药物组合物可以按照本身已知的方式制备,例如通过常规混合、溶解、造粒、制糖衣丸、磨细、乳化、装入胶囊、包载或冻干方法。The pharmaceutical compositions according to the invention can be prepared in a manner known per se, for example by conventional mixing, dissolving, granulating, dragee-making, levigating, emulsifying, encapsulating, entrapping or lyophilizing processes.
因此,用于本发明的药物组合物可以按照常规方式,使用一种或多种生理上可接受的载体进行配制,载体包括赋形剂和助剂以利于将活性化合物加工成可药用的制剂。适当的制剂取决于所选择的给药途径。Therefore, the pharmaceutical compositions used in the present invention can be formulated in a conventional manner using one or more physiologically acceptable carriers, which include excipients and auxiliaries to facilitate processing of the active compounds into pharmaceutically acceptable preparations . Proper formulation is dependent on the route of administration chosen.
对于注射剂,本发明药物可以在含水溶液、优选在生理相容的缓冲剂如Hanks′溶液、林格氏溶液或生理盐水缓冲剂中配制。对于经粘膜给药来说,在制剂中使用对渗透屏障适当的渗透剂。该渗透剂通常是本领域已知的。For injection, the medicaments of the invention can be formulated in aqueous solutions, preferably in physiologically compatible buffers such as Hanks' solution, Ringer's solution or physiological saline buffer. For transmucosal administration, penetrants appropriate to the permeation barrier are used in the formulation. Such penetrants are generally known in the art.
对于口服给药,可以容易地通过将活性化合物与本领域公知的可药用载体混合对该化合物进行配制。对于由接受治疗的患者经口摄食来说,该载体使得本发明化合物可以配制成片剂、丸剂、糖衣丸、胶囊、液体、凝胶、糖浆、浆液和悬浮液等。口服药物制剂可以通过下述方法获得:将活性化合物与固体赋形剂混合,任选地研磨所得混合物,并根据需要加入合适的助剂后将该混合物造粒,得到片剂或糖衣丸芯。合适的赋形剂尤其是填料例如糖包括乳糖、蔗糖、甘露醇或山梨糖醇;纤维素制剂例如玉米淀粉、小麦淀粉、大米淀粉、土豆淀粉、明胶、黄蓍胶、甲基纤维素、羟丙基甲基纤维素、羧甲基纤维素钠和/和聚乙烯吡咯烷酮(PVP)。如果需要,可以加入崩解剂,例如交联聚乙烯吡咯烷酮,琼脂或藻酸或其盐如藻酸钠。For oral administration, the compounds can be formulated readily by combining the active compounds with pharmaceutically acceptable carriers well known in the art. For oral ingestion by a patient to be treated, such carriers enable the compounds of the invention to be formulated as tablets, pills, dragees, capsules, liquids, gels, syrups, slurries, suspensions and the like. Pharmaceutical preparations for oral administration can be obtained by mixing the active compound with a solid excipient, optionally grinding a resulting mixture, and granulating the mixture, after adding suitable auxiliaries, if desired, to obtain tablets or dragee cores. Suitable excipients are especially fillers such as sugars including lactose, sucrose, mannitol or sorbitol; cellulose preparations such as corn starch, wheat starch, rice starch, potato starch, gelatin, tragacanth, methylcellulose, hydroxy Propyl methylcellulose, sodium carboxymethylcellulose, and/and polyvinylpyrrolidone (PVP). If desired, disintegrants such as cross-linked polyvinylpyrrolidone, agar or alginic acid or a salt thereof such as sodium alginate may be added.
糖衣丸芯用合适的包衣层包衣。为此,可以使用浓缩糖溶液,其中可以任选地含有阿拉伯胶、滑石、聚乙烯吡咯烷酮、卡波普(carbopol)胶、聚乙二醇和/或二氧化钛、紫胶漆溶液和适宜的有机溶剂或溶剂混合物。可以向片剂或糖衣丸包衣中加入染料或色素以识别或区分不同组合活性化合物剂量的特征。Dragee cores are coated with suitable coatings. For this purpose, concentrated sugar solutions may be used, which may optionally contain gum arabic, talc, polyvinylpyrrolidone, carbopol gum, polyethylene glycol and/or titanium dioxide, lacquer solutions and suitable organic solvents or solvent mixture. Dyestuffs or pigments may be added to the tablets or dragee coatings for identification or to characterize different doses of combined active compound.
可以经口服给药的药物制剂包括由明胶制备的推-合式(push-fit)胶囊以及由明胶和增塑剂例如甘油或山梨醇制备的软密封胶囊。推-合式胶囊可以含有与填料如乳糖、粘合剂如淀粉和/或润滑剂如滑石或硬脂酸镁以及任选地与稳定剂混合的活性成分。在软胶囊中,活性化合物可以溶解或悬浮在合适的液体例如脂肪油、液体石蜡或液体聚乙二醇中。此外可以加入稳定剂。所有口服制剂均应为适于该给药形式的剂量。Pharmaceutical preparations which can be administered orally include push-fit capsules made of gelatin, as well as soft, sealed capsules made of gelatin and a plasticizer, such as glycerol or sorbitol. The push-fit capsules can contain the active ingredients in admixture with filler such as lactose, binders such as starches, and/or lubricants such as talc or magnesium stearate and, optionally, stabilizers. In soft capsules, the active compounds may be dissolved or suspended in suitable liquids, such as fatty oils, liquid paraffin, or liquid polyethylene glycols. In addition, stabilizers can be added. All oral formulations should be in dosages suitable for the form of administration.
对于颊给药来说,该组合物可以是以常规方式配制的片剂或锭剂。For buccal administration the composition may be in the form of tablets or lozenges formulated in conventional manner.
对于通过吸入给药来说,用于本发明的化合物通常以气溶胶喷雾剂的形式由加压包装或喷雾器给药,其中使用合适的抛射剂例如二氯二氟甲烷、三氯氟甲烷、二氯四氟乙烷、二氧化碳或其他适宜的气体。对于加压气溶胶来说,剂量单位可以通过安装阀以确定释放计量。用于吸入器或吹入器的例如明胶胶囊和药筒可以配制成含有该化合物粉末混合物和适宜粉末基质如乳糖或淀粉的形式。可以将该化合物配制成用于非肠道给药的注射形式,例如浓缩药团(bolus)注射剂或连续输注液。注射制剂可以是单位剂型,例如安瓿或多剂量容器,其中含有防腐剂。该组合物可以是在油或水载体中的悬浮液、溶液或乳液,并且可以含有制剂用试剂例如悬浮剂、稳定剂和/或分散剂。For administration by inhalation, the compounds for use in the present invention are usually administered in the form of an aerosol spray from pressurized packs or nebulizers using suitable propellants such as dichlorodifluoromethane, trichlorofluoromethane, dichlorofluoromethane, Chlorotetrafluoroethane, carbon dioxide or other suitable gas. For pressurized aerosols, the dosage unit may be fitted with a valve to determine the metered release. Capsules and cartridges of eg gelatin for use in an inhaler or insufflator may be formulated containing a powder mix of the compound and a suitable powder base such as lactose or starch. The compounds may be formulated for parenteral administration in the form of injections, such as concentrated bolus injections or continuous infusions. Formulations for injection may be presented in unit dosage form such as ampoules or in multi-dose containers, with a preservative. The compositions may be suspensions, solutions or emulsions in oily or aqueous vehicles, and may contain formulatory agents such as suspending, stabilizing and/or dispersing agents.
非肠道给药的药物制剂包括水溶形式的活性化合物水溶液。此外,活性化合物的悬浮液可以制备成适当的含油注射悬浮液。合适的亲脂性溶剂或载体包括脂肪油如芝麻油或合成脂肪酸酯如油酸乙酯或甘油三酯或脂质体。含水注射悬浮液可以含有增加悬浮液粘度的物质,例如羧甲基纤维素钠、山梨醇或葡聚糖。任选地,该悬浮液还可以含有合适的稳定剂或增加化合物的稳定性以允许制备高度浓缩溶液的试剂。Pharmaceutical formulations for parenteral administration include aqueous solutions of the active compounds in water-soluble form. Additionally, suspensions of the active compounds may be prepared as appropriate oily injection suspensions. Suitable lipophilic solvents or vehicles include fatty oils such as sesame oil, or synthetic fatty acid esters such as ethyl oleate or triglycerides, or liposomes. Aqueous injection suspensions may contain substances which increase the viscosity of the suspension, such as sodium carboxymethyl cellulose, sorbitol, or dextran. Optionally, the suspension may also contain suitable stabilizers or agents which increase the stability of the compounds to allow for the preparation of highly concentrated solutions.
或者,活性成分可以是在使用前用合适的载体例如无菌无热原水配制的粉末形式。Alternatively, the active ingredient may be in powder form for constitution with a suitable vehicle, eg sterile pyrogen-free water, before use.
该化合物还可以配制成直肠给药组合物例如栓剂或保留灌肠剂,例如含有常规栓剂基质如可可脂或其他甘油酯。The compounds may also be formulated in rectal compositions such as suppositories or retention enemas, eg, containing conventional suppository bases such as cocoa butter or other glycerides.
除了上述制剂之外,该化合物还可以配制成长效制剂。该长效制剂可以通过植入法给药(例如皮下或肌内植入或通过肌内注射给药)。因此,例如,该化合物可以与合适的聚合或疏水材料或离子交换树脂一起配制(例如在可接受油中的乳液),或者配制成不易溶的衍生物例如不易溶的盐。In addition to the formulations described above, the compound may also be formulated as a depot formulation. The long-acting formulations may be administered by implantation (eg, subcutaneous or intramuscular implantation or administration by intramuscular injection). Thus, for example, the compounds may be formulated with suitable polymeric or hydrophobic materials or ion exchange resins (eg, as emulsions in acceptable oils), or as poorly soluble derivatives such as poorly soluble salts.
用于本发明疏水化合物的可药用载体的实例是助溶剂系统,包括苄醇、非极性表面活性剂、可与水混溶的有机聚合物和水相。该助溶剂系统可以是VPD助溶剂系统。VPD是3%w/v苄醇、8%w/v非极性表面活性剂吐温80和65%w/v聚乙二醇300在无水乙醇中配制成所需体积的溶液。该VPD助溶剂系统(VPD:5W)由用5%葡萄糖水溶液进行1∶1稀释的VPD组成。该助溶剂系统完全溶解疏水的化合物,并且其本身经系统给药产生的毒性很低。当然,只要不破坏其溶解性和毒性特性,助溶剂系统的比例可以有很大的改变。此外,助溶剂组分的特性可以改变:例如可以使用其他低毒、非极性表面活性剂代替吐温80;聚乙二醇的份数大小可以改变;可以用其他生物相容性聚合物代替聚乙二醇,例如聚乙烯吡咯烷酮;并且可以用其他糖或多糖代替葡萄糖。An example of a pharmaceutically acceptable carrier for a hydrophobic compound of the invention is a co-solvent system comprising benzyl alcohol, a non-polar surfactant, a water-miscible organic polymer, and an aqueous phase. The co-solvent system may be a VPD co-solvent system. VPD is a solution of 3% w/v benzyl alcohol, 8% w/v non-polar surfactant Tween 80 and 65% w/v polyethylene glycol 300 in absolute ethanol to the required volume. The VPD co-solvent system (VPD: 5W) consisted of VPD diluted 1:1 with 5% dextrose in water. The co-solvent system completely dissolves hydrophobic compounds and itself produces low toxicity upon systemic administration. Of course, the proportions of the co-solvent system can be varied considerably so long as the solubility and toxicity properties are not compromised. In addition, the properties of the co-solvent components can be changed: for example, other less toxic, non-polar surfactants can be used instead of Tween 80; the portion size of polyethylene glycol can be changed; other biocompatible polymers can be substituted polyethylene glycols, such as polyvinylpyrrolidone; and other sugars or polysaccharides may be substituted for glucose.
或者,可以使用供疏水药物化合物用的其他给药系统。脂质体和乳液是公知的疏水药物给药赋形剂和载体。也可以使用某些有机溶剂例如二甲基亚砜,尽管其毒性成本通常更大。此外,该化合物可以使用持续释放系统给药,例如含有治疗剂的固体疏水聚合物的半透基质。各种持续释放材料是已经确定的,并且是本领域专业人员公知的。根据其化学性质,持续释放胶囊释放化合物数周至长达100天。根据治疗剂的化学性质和生物稳定性,可以使用稳定蛋白质的其他策略。Alternatively, other delivery systems for hydrophobic pharmaceutical compounds can be used. Liposomes and emulsions are well known hydrophobic drug delivery vehicles and carriers. Certain organic solvents such as dimethylsulfoxide can also be used, although their toxicity cost is usually greater. Additionally, the compounds can be administered using a sustained-release system, such as semipermeable matrices of solid hydrophobic polymers containing the therapeutic agent. Various sustained release materials have been established and are well known to those skilled in the art. Sustained-release capsules release the compound for several weeks to as long as 100 days, depending on their chemical nature. Depending on the chemical nature and biological stability of the therapeutic agent, other strategies for protein stabilization may be used.
药物组合物还可以含有合适的固体或凝胶相载体或赋形剂。所述载体或赋形剂的实例包括、但不限于碳酸钙、磷酸钙、各种糖、淀粉、纤维素衍生物、明胶和聚合物如聚乙二醇。The pharmaceutical composition may also contain suitable solid or gel phase carriers or excipients. Examples of such carriers or excipients include, but are not limited to, calcium carbonate, calcium phosphate, various sugars, starches, cellulose derivatives, gelatin and polymers such as polyethylene glycol.
许多本发明的有机分子化合物可以以其与药物相容性抗衡离子的盐形式使用。可以与许多酸形成药物相容性盐,所述酸包括但不限于盐酸、硫酸、乙酸、乳酸、酒石酸、苹果酸、琥珀酸等。与相应的游离碱相比,盐更易溶于水或其他质子溶剂。Many of the organic molecular compounds of the invention can be used in the form of their salts with pharmaceutically compatible counterions. Pharmaceutically compatible salts can be formed with many acids including, but not limited to, hydrochloric, sulfuric, acetic, lactic, tartaric, malic, succinic, and the like. Salts are more soluble in water or other protic solvents than the corresponding free bases.
有效剂量effective dose
适用于本发明的药物组合物包括其中含有达到其预期目有效量的活性成分的组合物。尤其是,治疗有效量意味着有效预防现有症状发展或减轻接受治疗者现有症状的量。有效量的确定在本领域专业人员的能力范围内。Pharmaceutical compositions suitable for use in the present invention include compositions wherein the active ingredient is contained in an effective amount to achieve its intended purpose. In particular, a therapeutically effective amount means an amount effective to prevent the development of existing symptoms or to alleviate existing symptoms in the subject being treated. Determination of an effective amount is within the capability of those skilled in the art.
对于本发明方法中所用的许多化合物,可以首先由细胞分析估计治疗有效量。例如,可以在细胞和动物模型中制订剂量以达到循环浓度范围,包括在细胞分析中测定的IC50值(即达到指定蛋白激酶活性半数最大抑制的试验化合物浓度)。在某些情况下,在3-5%血清白蛋白存在下测定IC50值是适当的,因为该测定接近血浆蛋白对化合物的结合作用。该信息可用于更精确地确定人的有效剂量。此外,在血浆中可达到安全水平的最优选全身给药化合物有效地抑制蛋白激酶在完整细胞中的信号传导。For many of the compounds used in the methods of the invention, the therapeutically effective amount can be estimated first from cellular assays. For example, a dose can be formulated in cellular and animal models to achieve a range of circulating concentrations that include IC50 values (ie, the concentration of test compound that achieves a half-maximal inhibition of the activity of a indicated protein kinase) determined in cellular assays. In some cases, determination of IC50 values in the presence of 3-5% serum albumin is appropriate because this determination approximates the binding of plasma proteins to the compound. Such information can be used to more accurately determine effective doses in humans. In addition, most preferably systemically administered compounds that achieve safe levels in plasma effectively inhibit protein kinase signaling in intact cells.
治疗有效剂量是指导致患者症状改善的化合物的量。该化合物的毒性和治疗功效可以通过标准药学方法在细胞培养物或试验动物中测定,例如测定最大耐受剂量(MTD)和ED50值(50%最大反应的有效剂量)。毒性与治疗效果之间的剂量比是治疗指数,它可以表示为MTD与ED50之比。优选治疗指数高的化合物。由这些细胞培养物分析和动物研究得到的数据可用于配制人用剂量范围。该化合物的这些剂量优选落入循环浓度范围,包括小的或无毒性的ED50值。根据使用的剂型和给药途径,该剂量可以在该范围内变化。精确的制剂、给药体积和剂量可以由医师根据患者的状况进行选择。(参见例如Fingl等,1975,″The Pharmacological Basis of Therapeutics″,Ch.1 pl)。在危象治疗中,可能需要以急性浓缩药团形式或通过输注途径使用MTD以实现快速反应。A therapeutically effective dose is that amount of a compound that results in amelioration of symptoms in a patient. Toxicity and therapeutic efficacy of the compounds can be determined by standard pharmaceutical methods in cell cultures or experimental animals, eg, determination of the maximum tolerated dose (MTD) and ED50 values (the effective dose at 50% of the maximum response). The dose ratio between toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio between MTD and ED50 . Compounds with high therapeutic indices are preferred. The data obtained from these cell culture assays and animal studies can be used in formulating a range of dosage for human use. Such doses of the compounds lie preferably within a range of circulating concentrations that include the ED50 with little or no toxicity. The dosage may vary within this range depending upon the dosage form employed and the route of administration. The precise formulation, administration volume and dosage can be selected by the physician according to the patient's condition. (See eg Fingl et al., 1975, "The Pharmacological Basis of Therapeutics", Ch. 1 pl). In crisis management, MTDs may be needed in acute bolus form or by infusion to achieve a rapid response.
可以对剂量和时间间隔逐一调节,以使活性物质的血浆水平足以保持激酶调控作用或最小有效浓度(MEC)。每一化合物的MEC不同,但是可以由体外数据估计;例如使用上文所述的分析估计达到50-90%蛋白激酶抑制所需的浓度。达到MEC所需的剂量取决于个体特征和给药途径。然而,可以使用HPLC分析或生物测定来确定血浆浓度。Dosage and time intervals may be adjusted individually so that plasma levels of the active substance are sufficient to maintain the kinase modulating effect or minimum effective concentration (MEC). The MEC will vary for each compound, but can be estimated from in vitro data; for example, the concentration required to achieve 50-90% inhibition of a protein kinase is estimated using the assay described above. Dosages required to achieve the MEC will depend on individual characteristics and route of administration. However, plasma concentrations can be determined using HPLC analysis or bioassays.
给药间隔也可以使用MEC值确定。应该采用保持血浆水平高于MEC 10-90%的时间、优选30-90%、最优选50-90%的时间直至达到所需的症状改善的给药方案施用化合物。如果局部给药或选择性摄取,则药物的局部有效浓度可以不与血浆浓度相关。Dosing intervals can also be determined using MEC values. Compounds should be administered using a dosing regimen that maintains plasma levels above the MEC for 10-90% of the time, preferably 30-90%, most preferably 50-90% of the time until the desired amelioration of symptoms is achieved. If administered locally or selectively uptake, the local effective concentration of the drug may not be related to the plasma concentration.
当然,组合物的给药量取决于接受治疗的对象、患者的体重、病患的严重程度、给药方式和处方医师的判断。The amount of the composition administered will, of course, depend on the subject being treated, the patient's weight, the severity of the affliction, the mode of administration, and the judgment of the prescribing physician.
包装Package
如果需要,该组合物可以以含有一个或多个含活性成分的单位剂型的包装或分配器装置形式提供。该包装可以例如包括金属或塑料箔,例如泡罩包装。该包装或分配器装置可以附带用药说明。也可以制备含有配制在相容性可药用载体中的本发明化合物的组合物,将其置于适当的容器中,并贴上治疗所示疾病的标签。The compositions may, if desired, be presented in a pack or dispenser device containing one or more unit dosage forms containing the active ingredient. The pack may eg comprise metal or plastic foil, eg a blister pack. The pack or dispenser device may be accompanied by instructions for use. Compositions containing a compound of the invention formulated in a compatible pharmaceutically acceptable carrier may also be prepared, placed in an appropriate container, and labeled for treatment of an indicated condition.
在某些制剂中,将本发明化合物以非常小的颗粒形式使用是有利的,例如通过液能磨获得该小颗粒。In certain formulations it may be advantageous to use the compounds of the invention in the form of very small particles, for example obtained by liquid energy milling.
通过下列描述说明本发明化合物在制备药物组合物中的应用。在该描述中,术语″活性化合物″是指任何本发明化合物,但是尤其是指任何在前的一个实施例的最终产物化合物。The use of the compounds of the invention for the preparation of pharmaceutical compositions is illustrated by the following description. In this description, the term "active compound" refers to any compound of the present invention, but especially to the end product compound of any preceding Example.
a) 胶囊 a) Capsules
在胶囊制备中,可以将10重量份的活性化合物与240重量份的乳糖解聚并混合。将该混合物填充到硬凝胶胶囊中,每粒胶囊含有单位剂量或部分单位剂量的活性化合物。In the preparation of capsules, 10 parts by weight of active compound can be depolymerized and mixed with 240 parts by weight of lactose. The mixture is filled into hard gelatin capsules, each capsule containing a unit dose or part of a unit dose of active compound.
b) 片剂 b) tablet
可以由下列成分制备片剂。Tablets can be prepared from the following ingredients.
重量份 parts by weight
活性化合物 10active compound 10
乳糖 190Lactose 190
玉米淀粉 22cornstarch 22
聚乙烯吡咯烷酮 10Polyvinylpyrrolidone 10
硬脂酸镁 3Magnesium stearate 3
将活性化合物、乳糖和一些淀粉解聚、混合,并用聚乙烯吡咯烷酮的乙醇溶液将所得混合物造粒。将干燥的颗粒与硬脂酸镁和剩余的淀粉混合。然后在压片机中将该混合物压制成片,每片含单位剂量或部分单位剂量的活性化合物。The active compound, lactose and some starch are depolymerized, mixed and the resulting mixture is granulated with polyvinylpyrrolidone in ethanol. The dry granules are mixed with the magnesium stearate and the remaining starch. The mixture is then compressed in a tablet machine to yield tablets, each tablet containing a unit dose or part of a unit dose of active compound.
c) 肠溶衣片 c) Enteric-coated tablets
可以按照上述(b)的方法制备片剂。可以使用20%乙酸邻苯二甲酸纤维素和3%邻苯二甲酸二乙酯在乙醇∶二氯甲烷(1∶1)中的溶液,按照常规方法将片剂包肠溶衣。Tablets can be prepared according to the method of (b) above. Tablets may be enteric coated according to conventional methods using a solution of 20% cellulose acetate phthalate and 3% diethyl phthalate in ethanol:dichloromethane (1:1).
d) 栓剂 d) suppositories
在制备栓剂中,可以将100重量份的活性化合物掺入1300重量份的甘油三酯栓剂基质中,并使该混合物形成栓剂,每粒栓剂含有治疗有效量的活性成分。In preparing suppositories, 100 parts by weight of the active compound can be incorporated into 1300 parts by weight of a triglyceride suppository base, and the mixture is formed into suppositories, each suppository containing a therapeutically effective amount of the active ingredient.
如果需要,在本发明的组合物中,活性化合物可以与其他相容的药理活性成分联合。例如,本发明化合物可以与一种或多种附加的药物活性物质组合给药,所述药物活性物质抑制或预防VEGF产生、减弱细胞内对VEGF的应答、阻断细胞内信号传导、抑制血管渗透性过高、减少炎症或抑制或预防水肿形成或新血管形成。本发明化合物可以在附加的药物活性物质之前、之后或同时给药,任何一种给药过程都是适当的。附加的药物活性物质包括、但不限于抗水肿类固醇、NSAIDS、ras抑制剂、抗TNF剂、抗IL-1剂、抗组胺药、PAF-拮抗剂、COX-1抑制剂、COX-2抑制剂、NO合酶抑制剂、PKC抑制剂和PI3激酶抑制剂。本发明化合物与附加的药物活性物质累加或协同发生作用。因此,与单独施用每种物质相比,将抑制血管生成、血管渗透性过高和/或抑制水肿形成的物质组合给药可以大大减轻过度增殖疾病、血管生成、血管渗透性过高或水肿的有害作用。在恶性疾病的治疗中,预期可以与抗增殖或细胞毒素化疗组合。In the compositions of the invention, the active compounds may, if desired, be combined with other compatible pharmacologically active ingredients. For example, compounds of the invention may be administered in combination with one or more additional pharmaceutically active substances that inhibit or prevent VEGF production, attenuate intracellular responses to VEGF, block intracellular signaling, inhibit vascular permeability hyperactivity, reduce inflammation, or inhibit or prevent edema formation or neovascularization. The compound of the invention may be administered before, after or simultaneously with the additional pharmaceutically active substance, whichever course of administration is appropriate. Additional pharmaceutical actives include, but are not limited to, anti-edematous steroids, NSAIDS, ras inhibitors, anti-TNF agents, anti-IL-1 agents, antihistamines, PAF-antagonists, COX-1 inhibitors, COX-2 inhibitors agents, NO synthase inhibitors, PKC inhibitors and PI3 kinase inhibitors. The compounds of the invention act additively or synergistically with additional pharmaceutically active substances. Thus, administration of substances that inhibit angiogenesis, vascular hyperpermeability and/or formation of edema in combination can significantly reduce the effects of hyperproliferative disease, angiogenesis, vascular hyperpermeability or edema compared to administration of each substance alone. Harmful effect. In the treatment of malignant diseases, it is contemplated that it may be combined with antiproliferative or cytotoxic chemotherapy.
本发明还包括式I化合物作为药物的用途。The present invention also includes the use of compounds of formula I as medicaments.
Src和Syk族激酶在调节免疫功能中起着关键作用。Src族目前包括Fyn,Lck,Fgr,Fes,Lyn,Src,Yes,Hck和Blk。目前应该理解,Syk族只包括Zap和Syk。Janus族激酶涉及生长因子和原炎性细胞因子信号通过许多受体的传导。尽管Tec族激酶的成员BTK和ITK在免疫生物学中起着少为人知的作用,但是用抑制剂对其进行调节却证明有治疗益处。激酶RIP,IRAK-1,IRAK-2,NIK,TPL-2/COT,IKK-1和IKK-2涉及关键的原炎性细胞因子TNF和IL-1信号传导路径。由于式I化合物能够抑制这些激酶中的一种或多种,因此它们可以用作用于保持同种移植物和治疗自身免疫疾病的免疫调节剂。通过其调节T细胞活性或加强炎性过程的能力,这些化合物可用于治疗该自身免疫疾病。由于排斥现象,或者是宿主对实体器官的移植物排斥、或者是骨髓移植物对宿主的排斥,移植物目前有效的免疫抑制剂毒性的限制,并且将得益于治疗指数改进的有效药物。基因靶向试验已经证明Src在对骨吸收负有责任的破骨细胞的生物学中起着重要作用。通过其调节Src的能力,式I化合物还可以用于治疗骨质疏松、骨硬化病、佩吉特氏病、肿瘤引起的高钙血和治疗骨转移瘤。Src and Syk family kinases play key roles in regulating immune function. The Src family currently includes Fyn, Lck, Fgr, Fes, Lyn, Src, Yes, Hck, and Blk. It should now be understood that the Syk family includes only Zap and Syk. Janus family kinases are involved in the transmission of growth factor and proinflammatory cytokine signaling through a number of receptors. Although members of the Tec family of kinases, BTK and ITK, play little-understood roles in immunobiology, their modulation with inhibitors has proven therapeutically beneficial. The kinases RIP, IRAK-1, IRAK-2, NIK, TPL-2/COT, IKK-1 and IKK-2 are involved in the key proinflammatory cytokine TNF and IL-1 signaling pathways. Since the compounds of formula I are capable of inhibiting one or more of these kinases, they are useful as immunomodulators for maintenance of allografts and treatment of autoimmune diseases. Through their ability to modulate T cell activity or potentiate inflammatory processes, these compounds are useful in the treatment of this autoimmune disease. Due to the phenomenon of rejection, either host-to-solid-organ graft rejection, or bone-marrow graft-to-host rejection, grafts are currently limited by the toxicity of immunosuppressive agents and would benefit from effective drugs with improved therapeutic indices. Gene targeting experiments have demonstrated that Src plays an important role in the biology of osteoclasts responsible for bone resorption. Through their ability to regulate Src, the compounds of formula I are also useful in the treatment of osteoporosis, osteopetrosis, Paget's disease, tumor-induced hypercalcemia and in the treatment of bone metastases.
已经证明许多蛋白激酶是原癌基因。染色体断裂(在染色体5的ltk激酶断点)、易位例如带有BCR的Abl基因(Philadelphia染色体)、截断例如c-Kit或EGFR或突变(例如Met)导致失控蛋白产生,将其由原癌基因转化为致癌基因产物。在其他肿瘤中,由自分泌或旁分泌配体/生长因子受体相互作用推进瘤形成。许多src-族激酶通常与下游信号传导有关,因而加强了瘤形成,并且其本身通过过分表达或突变可以变成致癌基因。通过抑制这些蛋白的蛋白激酶活性,可以使疾病过程中断。血管再狭窄可能涉及FGF和/或PDGF促进的平滑肌和内皮细胞增殖。FGFR、PDGFR、IGF1-R和c-Met的体内配体刺激是原血管生成的,并且加强了血管生成依赖性疾病。抑制FGFr、PDGFr、c-Met或IGF1-R激酶活性可以是抑制这些现象有效的策略。因此,抑制正常或异常c-kit、c-met、c-fms、src-族成员、EGFr、erbB2、erbB4、BCR-Ab1、PDGFr、FGFr、IGF1-R和其他受体或胞质酪氨酸激酶在治疗良性和新生物增殖疾病中是有价值的。Many protein kinases have been shown to be proto-oncogenes. Chromosomal breaks (ltk kinase breakpoint on chromosome 5), translocations such as the Abl gene with BCR (Philadelphia chromosome), truncations such as c-Kit or EGFR, or mutations (such as Met) leading to unregulated protein production, which is caused by proto-oncogenic Gene conversion into oncogene products. In other tumors, neoplasia is driven by autocrine or paracrine ligand/growth factor receptor interactions. Many src-family kinases are often associated with downstream signaling, thus enhancing neoplasia, and can themselves become oncogenes through overexpression or mutation. By inhibiting the protein kinase activity of these proteins, the disease process can be interrupted. Vascular restenosis may involve smooth muscle and endothelial cell proliferation promoted by FGF and/or PDGF. Ligand stimulation of FGFR, PDGFR, IGF1-R and c-Met in vivo is pro-angiogenic and potentiates angiogenesis-dependent diseases. Inhibition of FGFr, PDGFr, c-Met or IGF1-R kinase activity may be an effective strategy to inhibit these phenomena. Thus, inhibition of normal or abnormal c-kit, c-met, c-fms, src-family members, EGFr, erbB2, erbB4, BCR-Ab1, PDGFr, FGFr, IGF1-R and other receptor or cytoplasmic tyrosine Kinases are valuable in the treatment of benign and neoplastic proliferative diseases.
在许多病理情况下(例如实体原发瘤和转移瘤、卡波济氏肉瘤、类风湿性关节炎、由于不适当的眼新血管生成导致的视觉缺失、牛皮癣和动脉粥样硬化),一旦持续血管形成,疾病的进行是偶然的。通常由疾病组织或相关的炎性细胞产生的多肽生长因子及其相应的内皮细胞特异性受体酪氨酸激酶(例如KDR/VEGFR-2、Flt1/VEGFR-1、Tie-2/Tek和Tie)对于刺激内皮细胞生长、迁移、机化、分化和建立必要的新功能脉管系统是重要的。据信,VEGF″血管渗透性因子″在介导血管渗透性过高、VEGFR激酶的VEGF刺激中的活性在以下病症中起着重要作用:肿瘤腹水形成、脑和肺水肿、胸膜和心包渗漏积液、延迟型过敏反应、外伤后的组织水肿和器官机能障碍、灼伤、局部缺血、糖尿病并发症、子宫内膜异位、成人呼吸窘迫综合征(ARDS)、心肺搭桥后有关的低血压和渗透性过高、以及眼水肿导致的青光眼或由于不适当的新血管生成引起的视觉缺失。除了VEGF之外,最近识别的VEGF-C和VEGF-D以及病毒编码的VEGF-E或HIV-Tat蛋白还可以通过刺激VEGFR激酶引起血管渗透性过高反应。Tie-2也在选择种群的造血干细胞中表达,其中Tie-2在其募集、粘着、调节和分化中起重要作用(Blood、4317-4326(1997));该表达种群Tie-2可以作为循环血管生成内皮祖代。因此,某些能够阻断内皮细胞特异性激酶的激酶活性的式I药物能够抑制涉及这些情境的疾病进行。In many pathological conditions (eg, solid primary tumors and metastases, Kaposi's sarcoma, rheumatoid arthritis, visual loss due to inappropriate ocular neovascularization, psoriasis, and atherosclerosis), once persistent Angiogenesis, the progression of the disease is accidental. Polypeptide growth factors and their corresponding endothelial cell-specific receptor tyrosine kinases (such as KDR/VEGFR-2, Flt1/VEGFR-1, Tie-2/Tek, and Tie ) is important for stimulating endothelial cell growth, migration, organization, differentiation and establishment of the necessary new functional vasculature. The activity of VEGF "vascular permeability factor" in mediating vascular hyperpermeability, VEGF stimulation of VEGFR kinases is believed to play an important role in the following conditions: neoplastic ascites formation, brain and pulmonary edema, pleural and pericardial leaks Effusion, delayed-type hypersensitivity reaction, tissue edema and organ dysfunction after trauma, burns, ischemia, diabetic complications, endometriosis, adult respiratory distress syndrome (ARDS), post-cardiopulmonary bypass-related hypotension and hyperosmolarity, as well as glaucoma due to ocular edema or loss of vision due to inappropriate neovascularization. In addition to VEGF, the recently identified VEGF-C and VEGF-D as well as virally encoded VEGF-E or HIV-Tat proteins can also induce vascular hyperpermeability responses by stimulating VEGFR kinase. Tie-2 is also expressed in selected populations of hematopoietic stem cells, where Tie-2 plays an important role in their recruitment, adhesion, regulation and differentiation (Blood, 4317-4326 (1997)); Angiogenic endothelial progenitors. Thus, certain drugs of formula I capable of blocking the kinase activity of endothelial cell-specific kinases are able to inhibit the progression of diseases involving these contexts.
式I化合物或其盐或含有治疗有效量该化合物或其盐的药物组合物可用于治疗良性和新生物增殖疾病和免疫系统疾病。所述疾病包括自身免疫疾病例如类风湿性关节炎、甲状腺炎、I型糖尿病、多发性脓硬化、肉样瘤病、炎性肠疾病、节段性回肠炎、重症肌无力和全身性红斑狼疮;牛皮癣、器官移植物排斥(例如肾排斥、移植物对宿主疾病排斥)、良性和新生物增殖疾病、人类癌症例如肺、乳腺、胃、膀胱、结肠、胰腺、卵巢、前列腺和直肠癌以及造血恶性肿瘤(白血病和淋巴瘤)、以及涉及不适当血管形成的疾病例如糖尿病性视网膜病、早熟视网膜病、由于与年龄有关的黄斑变性导致的脉络膜新血管形成、和人的婴儿血管瘤。此外,该抑制剂可用于治疗与VEGF介导的水肿有关的疾病、腹水、渗漏和渗出物,包括例如黄斑水肿,脑水肿,急性肺损伤和成人呼吸窘迫综合征(ARDS)。A compound of formula I or a salt thereof or a pharmaceutical composition containing a therapeutically effective amount of the compound or a salt thereof is useful in the treatment of benign and neoplastic proliferative diseases and diseases of the immune system. Such diseases include autoimmune diseases such as rheumatoid arthritis, thyroiditis, type 1 diabetes, multiple sclerosis, sarcoidosis, inflammatory bowel disease, Crohn's disease, myasthenia gravis and systemic lupus erythematosus psoriasis, organ graft rejection (e.g., renal rejection, graft-versus-host disease rejection), benign and neoplastic proliferative diseases, human cancers such as lung, breast, stomach, bladder, colon, pancreas, ovary, prostate, and rectal cancer, and hematopoiesis Malignancies (leukemia and lymphoma), and diseases involving inappropriate vascularization such as diabetic retinopathy, retinopathy of prematurity, choroidal neovascularization due to age-related macular degeneration, and infantile hemangiomas in humans. In addition, the inhibitors are useful in the treatment of diseases associated with VEGF-mediated edema, ascites, leaks and exudates, including, for example, macular edema, cerebral edema, acute lung injury and adult respiratory distress syndrome (ARDS).
本发明化合物还适用于预防上述疾病。The compounds according to the invention are also suitable for the prophylaxis of the diseases mentioned above.
本发明另一方面提供了式I化合物或其盐在制备治疗哺乳动物、特别是人的血管渗透性过高、血管生成依赖性疾病、增殖疾病和/或免疫系统疾病的药物中的应用。Another aspect of the present invention provides the use of a compound of formula I or a salt thereof in the preparation of a medicament for treating mammals, especially human, vascular hyperpermeability, angiogenesis-dependent diseases, proliferative diseases and/or immune system diseases.
本发明还提供了治疗血管渗透性过高、不适当的新血管形成、增殖疾病和/或免疫系统疾病的方法,该方法包括给需要的哺乳动物、特别是人施用治疗有效量的式I化合物。The present invention also provides a method for treating vascular hyperpermeability, inappropriate neovascularization, proliferative diseases and/or immune system diseases, the method comprising administering a therapeutically effective amount of a compound of formula I to a mammal in need, especially a human .
化合物抑制这些蛋白激酶的体外效能可以通过下述方法测定。The in vitro potency of compounds to inhibit these protein kinases can be determined as follows.
通过试验化合物相对于对照的抑制外原底物(例如合成肽)磷酸化的量可以测定化合物的效能(Z.Songyang等,Nature.373:536-539)。The potency of a compound can be determined by the amount of a test compound that inhibits phosphorylation of an exogenous substrate (eg, a synthetic peptide) relative to a control (Z. Songyang et al., Nature. 373:536-539).
使用杆状病毒系统产生KDR酪氨酸激酶:Production of KDR tyrosine kinase using the baculovirus system:
使用由HUVEC细胞分离的cDNAs,通过PCR产生人KDR细胞内区域编码序列(aa789-1354)。还在该蛋白的N-末端引入多-His6序列。将该片段克隆到转染载体pVL1393的Xba 1和Not 1位点。使用BaculoGold转染试剂(PharMingen),通过共转染产生重组杆状病毒(BV)。将重组BV噬斑纯化,并通过Western分析进行检验。为了纯化蛋白,使2×106/ml SF-9细胞在SF-900-II培养基中生长,并以0.5噬斑形成单位/细胞(MOI)感染。在感染后48小时收获细胞。The human KDR intracellular region coding sequence (aa789-1354) was generated by PCR using cDNAs isolated from HUVEC cells. A poly-His6 sequence was also introduced at the N-terminus of the protein. This fragment was cloned into the Xba 1 and Not 1 sites of the transfection vector pVL1393. Recombinant baculovirus (BV) was produced by co-transfection using BaculoGold transfection reagent (PharMingen). Recombinant BV plaques were purified and verified by Western analysis. For protein purification, 2×106/ml SF-9 cells were grown in SF-900-II medium and infected at 0.5 plaque-forming units/cell (MOI). Cells were harvested 48 hours after infection.
纯化KDRPurified KDR
通过向1L细胞培养物的细胞沉淀物中加入50ml Triton X-100溶胞缓冲剂(20mM Tris,pH 8.0,137mM NaCl,10%甘油,1%Triton X-100,1mM PMSF,10μg/ml抑肽酶,1μg/ml亮抑肽酶),使表达(His)6KDR(aa789-1354)的SF-9细胞溶解。溶胞产物以19,000rpm的转速、在Sorval SS-34旋筒中、在4℃离心30分钟。将细胞溶解产物装5ml NiCl2螯合琼脂糖柱,用50mM HEPES,pH7.5,0.3M NaCl平衡。用含有0.25M咪唑的相同缓冲剂洗脱KDR。用SDS-PAGE和测量激酶活性的ELISA分析(下文)对柱馏分进行分析。纯化的KDR在25mM HEPES,pH7.5、25mM NaCl、5mM DTT缓冲剂中交换并在-80℃贮藏。By adding 50 ml Triton X-100 lysis buffer (20 mM Tris, pH 8.0, 137 mM NaCl, 10% glycerol, 1% Triton X-100, 1 mM PMSF, 10 μg/ml aprotinin to the cell pellet of 1 L cell culture enzyme, 1 μg/ml leupeptin), to lyse SF-9 cells expressing (His) 6 KDR (aa789-1354). The lysate was centrifuged at 19,000 rpm in a Sorval SS-34 rotor at 4°C for 30 minutes. The cell lysate was packed on 5ml NiCl 2 chelating agarose column, equilibrated with 50mM HEPES, pH7.5, 0.3M NaCl. KDR was eluted with the same buffer containing 0.25M imidazole. Column fractions were analyzed by SDS-PAGE and ELISA assay (below) measuring kinase activity. Purified KDR was exchanged in 25 mM HEPES, pH 7.5, 25 mM NaCl, 5 mM DTT buffer and stored at -80°C.
人Tie-2激酶的产生与纯化Production and purification of human Tie-2 kinase
使用由人胎盘分离的cDNAs模板,通过PCR产生人Tie-2细胞内区域编码序列(aa775-1124)。在N-末端引入多-His6序列,并将该装配克隆到转染载体pVL1939的Xba 1和Not 1位点。使用BaculoGold转染试剂(PharMingen),通过共转染产生重组BV。将重组BV噬斑纯化,并通过Western分析进行检验。为了纯化蛋白,使2×106/ml SF-9细胞在SF-900-II培养基中生长,并以0.5MOI感染。按照与对KDR所述类似的方法,纯化筛选中使用的His-标记的激酶。The human Tie-2 intracellular region coding sequence (aa775-1124) was generated by PCR using cDNAs template isolated from human placenta. A poly-His6 sequence was introduced at the N-terminus and the assembly was cloned into the Xba 1 and Not 1 sites of the transfection vector pVL1939. Recombinant BV was produced by co-transfection using BaculoGold transfection reagent (PharMingen). Recombinant BV plaques were purified and verified by Western analysis. For protein purification, 2×106/ml SF-9 cells were grown in SF-900-II medium and infected at an MOI of 0.5. The His-tagged kinase used in the screening was purified following a method similar to that described for KDR.
人Flt-1酪氨酸激酶的产生与纯化Production and Purification of Human Flt-1 Tyrosine Kinase
使用杆状病毒表达载体pVL1393(Phar Mingen,Los Angeles,CA)。将编码多-His6的核苷酸序列置于编码整个人Flt-1(氨基酸786-1338)细胞内激酶区的核苷酸区的5′端。使用由HUVEC细胞分离的cDNA文库,通过PCR产生编码激酶区的核苷酸序列。组氨酸残基使得该蛋白按照与KDR和ZAP70类似的方法进行亲合纯化可行。SF-9昆虫细胞以0.5的多重性感染,并在感染后48小时收获。The baculovirus expression vector pVL1393 (Phar Mingen, Los Angeles, CA) was used. The nucleotide sequence encoding poly-His6 was placed 5' to the nucleotide region encoding the intracellular kinase domain of the entire human Flt-1 (amino acids 786-1338). Using a cDNA library isolated from HUVEC cells, the nucleotide sequence encoding the kinase region was generated by PCR. The histidine residues made it possible to affinity purify the protein in a similar way to KDR and ZAP70. SF-9 insect cells were infected at a multiplicity of 0.5 and harvested 48 hours after infection.
EGFR酪氨酸激酶来源EGFR tyrosine kinase source
EGFR购自Sigma(Cat#E-3641;500单位/50μl),并且EGF配体得自Oncogene Research Products/Calbiochem(Cat#PF011-100)。EGFR was purchased from Sigma (Cat#E-3641; 500 units/50μl) and EGF ligand was obtained from Oncogene Research Products/Calbiochem (Cat#PF011-100).
ZAP70的表达Expression of ZAP70
所用杆状病毒表达载体是pVL1393(Pharmingen,Los Angeles,Ca.)。将编码氨基酸M(H)6LVPR9S的核苷酸序列置于编码整个ZAP70(氨基酸1-619)的区的5′端。使用由Jurkat无限增殖化T-细胞分离的cDNA文库,通过PCR产生编码ZAP70编码区的核苷酸序列。组氨酸残基使得该蛋白的进行亲合纯化可行(参见下文)。LVPR9S桥构成了通过凝血酶蛋白水解裂解的识别序列,使得从酶中除去亲合标记可行。SF-9昆虫细胞以0.5的多重性感染,并在感染后48小时收获。The baculovirus expression vector used was pVL1393 (Pharmingen, Los Angeles, Ca.). The nucleotide sequence encoding amino acid M(H) 6LVPR9S was placed 5' to the region encoding the entire ZAP70 (amino acids 1-619). The nucleotide sequence encoding the ZAP70 coding region was generated by PCR using a cDNA library isolated from Jurkat immortalized T-cells. The histidine residues allow affinity purification of the protein (see below). The LVPR 9 S bridge constitutes the recognition sequence for proteolytic cleavage by thrombin, making removal of the affinity tag feasible from the enzyme. SF-9 insect cells were infected at a multiplicity of 0.5 and harvested 48 hours after infection.
ZAP70的提取与纯化Extraction and purification of ZAP70
在由20mM Tris pH8.0、137mM NaCl、10%甘油、1%TritonX-100、1mM PMSF、1μg/ml亮抑肽酶、10μg/ml抑肽酶和1 mM原钒酸钠组成的缓冲剂中,将SF-9细胞溶解。将可溶性溶胞产物装入螯合琼脂糖HiTrap柱(Pharmacia),用50mM HEPES pH7.5、0.3M NaCl平衡。融合蛋白用250mM咪唑洗脱。该酶在含有50mM HEPES pH7.5、50mM NaCl和5mM DTT的缓冲剂中贮藏。In a buffer consisting of 20 mM Tris pH 8.0, 137 mM NaCl, 10% glycerol, 1% TritonX-100, 1 mM PMSF, 1 μg/ml leupeptin, 10 μg/ml aprotinin, and 1 mM sodium orthovanadate , the SF-9 cells were lysed. The soluble lysate was loaded onto a chelating Sepharose HiTrap column (Pharmacia), equilibrated with 50 mM HEPES pH 7.5, 0.3 M NaCl. Fusion proteins were eluted with 250 mM imidazole. The enzyme is stored in a buffer containing 50mM HEPES pH 7.5, 50mM NaCl and 5mM DTT.
Lck来源Source of Lck
Lck或截短形式的Lck可以购买到(例如购自UpstateBiotechnology Inc.(Saranac Lake,N.Y)和Santa Cruz BiotechnologyInc.(Santa Cruz,Ca.))或者采用常规方法由已知的天然或重组来源纯化获得。Lck or truncated forms of Lck are commercially available (e.g. from Upstate Biotechnology Inc. (Saranac Lake, N.Y) and Santa Cruz Biotechnology Inc. (Santa Cruz, Ca.)) or purified from known natural or recombinant sources using conventional methods .
PTKs的酶联免疫吸附测定(ELISA)Enzyme-linked immunosorbent assay (ELISA) for PTKs
采用酶联免疫吸附测定(ELISA)测定和测量酪氨酸激酶活性的存在。该ELISA按照已知的方案进行,这些方案描述在例如Voller等,1980,″酶联免疫吸附测定″:Manual of Clinical Immunology,第2版,edited by Rose and Friedman,pp 359-371 Am.Soc.of Microbiology,Washington,D.C.。The presence of tyrosine kinase activity was determined and measured using an enzyme-linked immunosorbent assay (ELISA). The ELISA is performed according to known protocols described, for example, in Voller et al., 1980, "Enzyme-Linked Immunosorbent Assay": Manual of Clinical Immunology, 2nd Edition, edited by Rose and Friedman, pp 359-371 Am. Soc. of Microbiology, Washington, D.C.
采用该公开的方案测定特定的PTK活性。例如,下面提供了优选的进行ELISA实验的方案。采用这些方案测定化合物对其他受体PTK族成员和非受体酪氨酸激酶的活性是本领域专业人员力所能及的。为了测定抑制剂的选择性,在分析中,将浓度约为表观Km两倍的通用PTK底物(例如聚(Glu4 Tyr)随机共聚物,20,000-50,000MW)与ATP(通常为5μM)一起使用。Specific PTK activities were determined using the published protocol. For example, a preferred protocol for performing an ELISA experiment is provided below. Using these protocols to determine the activity of compounds against other receptor PTK family members and non-receptor tyrosine kinases is within the capabilities of those skilled in the art. To determine inhibitor selectivity, a general-purpose PTK substrate (e.g., poly(Glu 4 Tyr) random copolymer, 20,000-50,000 MW) at a concentration approximately twice the apparent Km is combined with ATP (typically 5 μM) in the assay use together.
使用下列步骤分析本发明化合物对KDR、Flt-1、Flt-4/VEGFR-3、Tie-2、EGFR、FGFR、PDGFR、IGF-1-R、c-Met和ZAP70酪氨酸激酶活性的抑制作用:Inhibition of KDR, Flt-1, Flt-4/VEGFR-3, Tie-2, EGFR, FGFR, PDGFR, IGF-1-R, c-Met, and ZAP70 tyrosine kinase activity by compounds of the invention was assayed using the following procedure effect:
缓冲剂和溶液:Buffers and solutions:
PGT聚(Glu,Tyr)4∶1PGT poly(Glu,Tyr) 4:1
在-20℃贮藏粉末。将粉末溶解在磷酸盐缓冲的盐水(PBS)中,制成50mg/ml的溶液。在-20℃贮藏1ml等分试样。当制备板时,将其在GibcoPBS中稀释至250μg/ml。Store the powder at -20°C. The powder was dissolved in phosphate buffered saline (PBS) to make a 50 mg/ml solution. Store 1 ml aliquots at -20°C. When preparing plates, they were diluted to 250 μg/ml in GibcoPBS.
反应缓冲剂:100mM Hepes,20mM MgCl2,4mM MnCl2,5mM DTT,0.02%BSA,200μM NaVO4,pH7.10。Reaction buffer: 100 mM Hepes, 20 mM MgCl 2 , 4 mM MnCl 2 , 5 mM DTT, 0.02% BSA, 200 μM NaVO 4 , pH 7.10.
ATP:在-20℃贮藏100mM的等分试样。在水中稀释至20μMATP: Store 100 mM aliquots at -20°C. Dilute to 20 μM in water
洗涤缓冲剂:含0.1%Tween 20的PBSWash buffer: PBS with 0.1% Tween 20
抗体稀释的缓冲剂:在PBS中的0.1%牛血清白蛋白(BSA)Buffer for antibody dilution: 0.1% bovine serum albumin (BSA) in PBS
TMB底物:在临用前将TMB底物与过氧化物溶液按9∶1混合或者使用得自Neogen的K-Blue底物。TMB substrate: TMB substrate was mixed 9:1 with peroxide solution immediately before use or K-Blue substrate from Neogen was used.
终止溶液:1M磷酸Stop solution: 1M phosphoric acid
步骤step
1.制板:1. Board making:
将PGT储液(50mg/ml,冷冻的)在PBS中稀释至250μg/ml。向Corning改性的平底高度亲合ELISA板(Corning#25805-96)的每孔中加入125μl。加入125μl PBS至空白孔中。用密封带覆盖,并在37℃培养过夜。用250μl洗涤缓冲剂洗涤1次,并在37℃干燥恒温箱中干燥约2小时。将包封的板在密封袋中于4℃贮藏直至使用。PGT stock solution (50 mg/ml, frozen) was diluted to 250 μg/ml in PBS. 125 [mu]l was added to each well of a Corning modified flat bottom high affinity ELISA plate (Corning #25805-96). Add 125 μl PBS to blank wells. Cover with sealing tape and incubate overnight at 37°C. Wash once with 250 μl of washing buffer, and dry in a 37°C drying incubator for about 2 hours. Store encapsulated plates in sealed bags at 4°C until use.
2.酪氨酸激酶反应:2. Tyrosine kinase reaction:
-制备4×浓度的在20%DMSO水溶液中的抑制剂溶液。- Prepare inhibitor solutions at 4x concentration in 20% DMSO in water.
-制备反应缓冲剂- Preparation of reaction buffer
-制备酶溶液,使得所需单位为50μl,例如对于KDR,制成1ng/μl,在反应中每孔中总计50ng。在冰上贮藏。- Prepare the enzyme solution so that the desired unit is 50 μl, eg for KDR make 1 ng/μl for a total of 50 ng in each well in the reaction. Store on ice.
-由100mM储液制备4×ATP水溶液。在冰上贮藏。- Prepare 4x ATP in water from 100 mM stock solution. Store on ice.
-向每孔中加入50μl酶溶液(根据激酶的特定活性,通常为5-50ng酶/孔)- Add 50 μl of enzyme solution to each well (usually 5-50 ng enzyme/well depending on the specific activity of the kinase)
-加入25μl 4×抑制剂- Add 25 μl 4× inhibitor
-加入25μl 4xATP用于抑制剂分析- Add 25 μl 4xATP for inhibitor analysis
-在室温培养10分钟- Incubate at room temperature for 10 minutes
-向每孔中加入50μl 0.05N HCl以终止反应-Add 50μl 0.05N HCl to each well to stop the reaction
-洗板- washing board
**反应的终浓度:5μM ATP,5%DMSO**Final concentration of the reaction: 5 μM ATP, 5% DMSO
3.抗体结合3. Antibody Binding
-通过两步稀释(100x,然后200x),将1mg/ml PY20-HRP(Pierce)抗体(磷酸酪氨酸抗体)的等分试样在0.1%BSA的PBS溶液中稀释至50ng/ml- Dilute an aliquot of 1 mg/ml PY20-HRP (Pierce) antibody (phosphotyrosine antibody) to 50 ng/ml in 0.1% BSA in PBS by two-step dilution (100x, then 200x)
-向每孔中加入100μl Ab。在室温培养1小时。在4℃培养1小时。- Add 100 μl Ab to each well. Incubate for 1 hour at room temperature. Incubate at 4°C for 1 hour.
-洗涤4x板- Wash 4x plates
4.颜色反应4. Color reaction
-制备TMB底物并向每孔中加入100μl- Prepare TMB substrate and add 100 μl to each well
-在650nm监测OD值,直至达到0.6- Monitor the OD value at 650nm until it reaches 0.6
-用1M磷酸终止反应。在平板读数器上振摇。- Stop the reaction with 1M phosphoric acid. Shake on plate reader.
-立即在450nm读取OD值- Immediate reading of OD value at 450nm
最佳培养时间和酶反应条件随酶制剂略有不同,并且大多靠经验决定。对于Lck,所用反应缓冲剂是100mM MOPSO,pH 6.5,4mMMnCl2,20mM MgCl2,5mM DTT,0.2%BSA,200mM NaVO4,在类似的分析条件下。Optimal incubation time and enzyme reaction conditions vary slightly with enzyme preparations and are mostly determined empirically. For Lck, the reaction buffer used was 100 mM MOPSO, pH 6.5, 4 mM MgCl 2 , 20 mM MgCl 2 , 5 mM DTT, 0.2% BSA, 200 mM NaVO 4 under similar analytical conditions.
式I化合物可用于治疗与被式I化合物抑制的已经确定、包括上面未提及的和尚未确定的蛋白酪氨酸激酶有关的疾病。所有本文列举的化合物在50微摩尔或更低浓度下显著抑制KDR激酶。某些本发明化合物在50微摩尔或更低浓度下还显著抑制其他PTKs如lck。The compounds of formula I are useful in the treatment of diseases associated with the inhibition of established protein tyrosine kinases, including those not mentioned above and which have not been identified, which are inhibited by the compounds of formula I. All compounds listed herein significantly inhibit KDR kinase at concentrations of 50 micromolar or less. Certain compounds of the present invention also significantly inhibit other PTKs such as lck at concentrations of 50 micromolar or less.
Cdc2来源Cdc2 source
人重组酶和分析缓冲剂可以购买到(New England Biolabs,Beverly,MA.USA)或者采用常规方法从已知的天然或重组来源纯化获得。Human recombinant enzymes and assay buffers can be purchased (New England Biolabs, Beverly, MA. USA) or purified from known natural or recombinant sources by conventional methods.
Cdc2测定Cdc2 assay
所用方案是使用购买的试剂并进行较少的改变。简单地说,在补充有新鲜的300μM ATP(31μCi/ml)和终浓度30μg/ml组蛋白IIIss的,由50mM Tris pH7.5、100mM NaCl、1mM EGTA、2mM DTT、0.01%Brij、5%DMSO和10mM MgCl2组成的缓冲剂(商品缓冲剂)中进行该反应。在有或无抑制剂存在下,使含有酶单位的80μL体积在25℃进行20分钟的反应。通过加入120μL 10%乙酸使该反应终止。通过将该混合物点在磷酸纤维素纸上,然后用75mM磷酸洗涤3次,每次洗涤5分钟,将底物与未结合的标记分离。在液体闪烁剂存在下,用β计数器计数。The protocol used was to use purchased reagents with minor changes. Briefly, in 50 mM Tris pH7.5, 100 mM NaCl, 1 mM EGTA, 2 mM DTT, 0.01% Brij, 5% DMSO supplemented with fresh 300 μM ATP (31 μCi/ml) and a final concentration of 30 μg/ml histone IIIss The reaction was carried out in a buffer consisting of 10 mM MgCl 2 (commercial buffer). A volume of 80 μL containing enzyme units was allowed to react for 20 minutes at 25° C. in the presence or absence of inhibitors. The reaction was stopped by adding 120 μL of 10% acetic acid. Substrate was separated from unbound label by spotting the mixture on phosphocellulose paper, followed by three 5 min washes with 75 mM phosphoric acid. Count with a beta counter in the presence of liquid scintillator.
某些本发明化合物在低于50uM的浓度显著抑制cdc2。Certain compounds of the invention significantly inhibited cdc2 at concentrations below 50 uM.
PKC激酶来源PKC kinase source
催化亚单位PKC可以购买到(Calbiochem)。The catalytic subunit PKC is commercially available (Calbiochem).
PKC激酶测定PKC kinase assay
按照公开的方法使用放射活性激酶测定法(Yasuda,I.,Kirshimoto,A.,Tanaka,S.,Tominaga,M.,Sakurai,A.,Nishizuka,Y.Biochemicaland Biophysical Research Communication 3:166,1220-1227(1990))。简单地说,所有反应均在由50mM Tris HCl pH7.5、10mM MgCl2,2mMDTT、1mM EGTA、100μM ATP、8μM肽、5%DMSO和33P ATP(8Ci/mM)组成的激酶缓冲剂中进行。将化合物和酶在反应容器中混合,并通过加入ATP和底物混合物使反应开始。通过加入10μL终止缓冲剂(在75mM磷酸中的5mM ATP)终止反应后,将一部分该混合物点在磷酸纤维素滤纸上。印迹的样品在室温用75mM磷酸洗涤3次,每次洗涤5-15分钟。通过液体闪烁计数法对放射标记结合进行定量。Radioactive kinase assays were used according to published methods (Yasuda, I., Kirshimoto, A., Tanaka, S., Tominaga, M., Sakurai, A., Nishizuka, Y. Biochemical and Biophysical Research Communication 3: 166, 1220- 1227 (1990)). Briefly, all reactions were performed in kinase buffer consisting of 50 mM Tris HCl pH 7.5, 10 mM MgCl 2 , 2 mM DTT, 1 mM EGTA, 100 μM ATP, 8 μM peptide, 5% DMSO and 33 P ATP (8 Ci/mM) . Compounds and enzymes are mixed in a reaction vessel and the reaction is initiated by adding ATP and substrate mixture. After stopping the reaction by adding 10 [mu]L of stop buffer (5 mM ATP in 75 mM phosphoric acid), a portion of this mixture was spotted on phosphocellulose filter paper. Blotted samples were washed 3 times with 75 mM phosphoric acid at room temperature for 5-15 min each. Radiolabel binding was quantified by liquid scintillation counting.
Erk2酶来源Erk2 enzyme source
重组鼠酶和分析缓冲剂可以购买到(New England Biolabs,BeverlyMA.USA)或者可以采用常规方法从已知的天然或重组来源纯化获得。Recombinant mouse enzymes and assay buffers can be purchased (New England Biolabs, Beverly MA. USA) or purified from known natural or recombinant sources by conventional methods.
Erk2酶测定Erk2 enzyme assay
简单地说,在供应商建议的条件下,在补充有新鲜的100μM ATP(31μCi/ml)和30μM髓磷脂碱性蛋白的,由50mM Tris pH 7.5、1mMEGTA、2mM DTT、0.01%Brij、5%DMSO和10mM MgCl2组成的缓冲剂(商品缓冲剂)中进行该反应。反应体积和结合放射活性的测定方法如上对PKC测定所述(参见上文)。Briefly, under the conditions suggested by the supplier, in supplemented with fresh 100 μM ATP (31 μCi/ml) and 30 μM myelin basic protein, prepared by 50 mM Tris pH 7.5, 1 mM GTA, 2 mM DTT, 0.01% Brij, 5% The reaction was carried out in a buffer consisting of DMSO and 10 mM MgCl2 (commercial buffer). Reaction volumes and bound radioactivity were determined as described above for the PKC assay (see above).
T-细胞活化的体外模型In vitro model of T-cell activation
一旦被促细胞分裂剂或抗原活化,将T-细胞引入以分泌IL-2-一种支持其随后增殖期的生长因子。由此,可以测量IL-2的产生或初期T-细胞或适当T-细胞系的细胞增殖作为T-细胞活化替代物。所有这些测定均在文献中有所描述,并且完全引证了其参数(in CurrentProtocols in Immunology,Vol 2,7.10.1-7.11.2)。Once activated by mitogens or antigens, T-cells are introduced to secrete IL-2, a growth factor that supports their subsequent proliferative phase. Thus, IL-2 production or cell proliferation of naive T-cells or appropriate T-cell lines can be measured as a surrogate for T-cell activation. All these assays are described in the literature and their parameters are fully cited (in Current Protocols in Immunology, Vol 2, 7.10.1-7.11.2).
简单地说,通过与同种异型刺激器细胞一同培养,可以使T-细胞活化,这是被称作单向混合淋巴细胞反应的方法。按照制造商的指导,通过Ficoll-Hypaque梯度(Pharmacia),纯化效应器和刺激器外周血管单核细胞。通过用丝裂霉素C(Sigma)或γ照射法处理使刺激器细胞有丝分裂灭活。在有或无试验化合物存在下,将效应器和刺激器细胞按2∶1的比例一同培养。通常在U形底微滴板(Costar Scientific)中将105效应器与5×104刺激器混合并涂板(200μl体积)。该细胞在补充有加热灭活的胎牛血清(Hyclone Laboratories)或得自男性供血者混合人AB血清、5×10-5M 2-巯基乙醇和0.5% DMSO的RPMI 1640中培养。该培养物在收获前一天(通常是第3天)用0.5μCi的3H-胸苷(Amersham)进行脉冲。收获培养物(Betaplate harvester,Wallac),并通过液体闪烁法(Betaplate,Wallac)估价同位素吸收。Briefly, T-cell activation can be achieved by co-cultivation with allogeneic stimulator cells, a method known as the one-way mixed lymphocyte reaction. Effector and stimulator peripheral vascular mononuclear cells were purified by Ficoll-Hypaque gradient (Pharmacia) following the manufacturer's instructions. Stimulator cells were mitotically inactivated by treatment with mitomycin C (Sigma) or gamma irradiation. Effector and stimulator cells were co-cultured in a 2:1 ratio in the presence or absence of test compounds. Typically 105 effectors were mixed with 5 x 104 stimulators and plated (200 μl volume) in U-bottom microtiter plates (Costar Scientific). The cells were cultured in RPMI 1640 supplemented with heat-inactivated fetal calf serum (Hyclone Laboratories) or pooled human AB serum from a male blood donor, 5 x 10 -5 M 2-mercaptoethanol and 0.5% DMSO. The culture was pulsed with 0.5 μCi of3H -thymidine (Amersham) one day before harvest (usually day 3). Cultures were harvested (Betaplate harvester, Wallac) and assessed for isotope uptake by liquid scintillation (Betaplate, Wallac).
可以使用同样的培养系统,通过测量IL-2的产生评价T-细胞活化。在开始培养的18-24小时,除去上清液,并按照制造商的指导,通过ELISA(R和D系统)测量IL-2浓度。T-cell activation can be assessed by measuring IL-2 production using the same culture system. At 18-24 hours from the start of the culture, the supernatant was removed and IL-2 concentrations were measured by ELISA (R and D systems) following the manufacturer's instructions.
T-细胞活化的体内模型In vivo model of T-cell activation
化合物的体内功效可以在直接测量T-细胞活化或者证明其为T-细胞效应器的抑制动物模型中进行检验。通过恒定比例的T-细胞受体与单克隆抗-CD3抗体(Ab)络合可以使T-细胞在体内活化。在该模型中,在放血前2小时,给BALB/c小鼠腹膜内注射10μg抗-CD3 Ab。在注射抗-CD3 Ab之前1小时,预先使动物接受单剂量化合物药物。通过ELISA测定原炎性细胞因子干扰素-γ(IFN-γ)和肿瘤坏死因子-α(TNF-α)的血清水平,这些因子是T-细胞活化指示剂。使用类似的模型,在体内用特定抗原如匙孔虫戚血蓝蛋白(KLH)引发T-细胞,继而用同样的抗原耗尽淋巴结细胞进行体外激发。如上所述,测量细胞因子产量以评估培养细胞的激活状态。简单地说,在第0天,用在完全弗氏佐剂(CFA)中乳化的100μg KLH皮下免疫C57BL/6小鼠。在免疫前一天预先用化合物对动物进行处理,并且在免疫后第1、2和3天用化合物对动物进行处理。在第4天收获耗尽的淋巴结,并在组织培养基(补充有加热灭活胎牛血清(Hyclone Laboratories)、5×10-5M 2-巯基乙醇和0.5%DMSO的RPMI 1640)中以6×106/ml的浓度对其细胞进行24-28小时的培养。然后通过ELISA,对培养物上清液进行自分泌T-细胞生长因子白介素-2(IL-2)和/或IFN-γ水平的的评估。The in vivo efficacy of compounds can be tested in animal models that directly measure T-cell activation or demonstrate inhibition as T-cell effectors. T-cells can be activated in vivo by complexing a constant ratio of T-cell receptors with monoclonal anti-CD3 antibodies (Abs). In this model, BALB/c mice were injected intraperitoneally with 10 μg of anti-CD3 Ab 2 hours before exsanguination. Animals were pre-dosed with a single dose of compound 1 hour prior to anti-CD3 Ab injection. Serum levels of the proinflammatory cytokines interferon-γ (IFN-γ) and tumor necrosis factor-α (TNF-α), which are indicators of T-cell activation, were determined by ELISA. Using a similar model, T-cells are primed in vivo with specific antigens such as keyhole limpet hemocyanin (KLH), followed by depletion of lymph node cells with the same antigen for in vitro challenge. Cytokine production was measured to assess the activation status of cultured cells as described above. Briefly, on day 0, C57BL/6 mice were immunized subcutaneously with 100 μg of KLH emulsified in complete Freund's adjuvant (CFA). Animals were pre-treated with compound the day before immunization and were treated with compound on days 1, 2 and 3 after immunization. Depleted lymph nodes were harvested on day 4 and cultured in tissue culture medium (RPMI 1640 supplemented with heat-inactivated fetal bovine serum (Hyclone Laboratories), 5×10 −5 M 2-mercaptoethanol, and 0.5% DMSO) at 6 The cells were cultured at a concentration of ×10 6 /ml for 24-28 hours. Culture supernatants were then assessed for levels of the autocrine T-cell growth factors interleukin-2 (IL-2) and/or IFN-γ by ELISA.
还对例示化合物进行人体疾病动物模型试验。例如实验性自免疫脑脊髓炎(EAE)和胶原诱导的关节炎(CIA)。模拟人多发性硬化的EAE模型在大鼠和小鼠中均有描述(综述FASEB J.5:2560-2566,1991;murine model:Lab.Invest.4(3):278,1981;rodent model:J.Immunol146(4):1163-8,1991)。简单地说,用髓磷脂碱性蛋白(MBP)或其神经肽衍生物和CFA的乳液免疫小鼠或大鼠。通过加入细菌毒素例如百日咳杆菌可以引起急性疾病。通过T-细胞从MBP/肽免疫的动物上过继转移,引起复发的/缓解的疾病。The exemplified compounds are also tested in animal models of human disease. Examples include experimental autoimmune encephalomyelitis (EAE) and collagen-induced arthritis (CIA). The EAE model simulating human multiple sclerosis has been described in both rats and mice (review FASEB J.5: 2560-2566, 1991; murine model: Lab.Invest.4(3): 278, 1981; rodent model: J. Immunol 146(4):1163-8, 1991). Briefly, mice or rats are immunized with an emulsion of myelin basic protein (MBP) or its neuropeptide derivatives and CFA. Acute disease can be caused by the addition of bacterial toxins such as B. pertussis. Relapsed/remitting disease was induced by adoptive transfer of T-cells from MBP/peptide immunized animals.
通过用II型胶原免疫,可以在DBA/1小鼠上引起CIA(J.Immunol:142(7):2237-2243)。早在抗原激发后10天小鼠即出现关节炎迹象,并且可以在免疫后长达90天进行评分。在EAE和CIA模型中,预防性地或在疾病开始时施用化合物。有效的药物应该减少疾病的严重性和/或发生率。CIA can be induced in DBA/1 mice by immunization with type II collagen (J. Immunol: 142(7):2237-2243). Mice showed signs of arthritis as early as 10 days after antigen challenge and could be scored up to 90 days after immunization. In the EAE and CIA models, compounds are administered prophylactically or at the onset of disease. An effective drug should reduce the severity and/or incidence of disease.
抑制一种或多种血管生成受体PTK和/或蛋白激酶如介导炎性反应的lck的本发明化合物可以在这些动物模型中减轻关节炎的严重程度和发生率。Compounds of the invention that inhibit one or more angiogenic receptors PTK and/or protein kinases such as lck that mediate inflammatory responses can reduce the severity and incidence of arthritis in these animal models.
还可以在小鼠同种移植物模型、或者是皮肤(综述,Ann.Rev.Immunol.,10:333-58,1992;Transplantation:57(12):1701-17D6,1994)或心脏(Am.J.Anat.:113:273,1963)移植模型中对化合物进行试验。简单地说,将全部厚度的皮肤移植物从C57BL/6小鼠移植到BALB/c小鼠上。从第6天开始,每天检查移植物排斥的迹象。在小鼠新生心脏移植模型中,将新生的心脏从C57BL/6小鼠异位移植到成年CBA/J小鼠的耳廓上。在移植后4-7天心脏开始搏动,施用解剖显微镜查看搏动停止以肉眼估计排斥情况。It can also be used in mouse allograft models, or skin (review, Ann. Rev. Immunol., 10: 333-58, 1992; Transplantation: 57 (12): 1701-17D6, 1994) or heart (Am. J. Anat.: 113:273, 1963) the compounds were tested in the transplantation model. Briefly, full thickness skin grafts were transplanted from C57BL/6 mice to BALB/c mice. Beginning on day 6, check daily for signs of graft rejection. In the mouse neonatal heart transplantation model, neonatal hearts were ectopically transplanted from C57BL/6 mice onto the pinnae of adult CBA/J mice. Hearts began to beat 4-7 days after transplantation, and a dissecting microscope was used to view cessation of beating to visually estimate rejection.
细胞受体PTK分析Cell Receptor PTK Analysis
下列细胞分析用于测定不同的本发明化合物对KDR/VEGFR2的活性和作用水平。可以采用本领域公知的技术,按照与其他酪氨酸激酶同样的方案,设计使用特异性配体刺激物的类似受体PTK分析。The following cellular assays were used to determine the activity and level of effect of various compounds of the invention on KDR/VEGFR2. Similar receptor PTK assays using specific ligand stimulators can be designed following the same protocol as for other tyrosine kinases using techniques well known in the art.
通过Western印迹法测量VEGF-诱导的在人脐静脉内皮细胞(HUVEC)中的KDR磷酸化作用:VEGF-induced KDR phosphorylation in human umbilical vein endothelial cells (HUVEC) was measured by Western blotting:
1.HUVEC细胞(来自混合供血者)购自Clonetics(San Diego,CA),并按照制造商的指导进行培养。仅仅使用早期的传代(3-8代)进行该分析。使用完全EBM培养基(Clonetics),将细胞在100mm培养皿(组织培养用的Falcon;Becton Dickinson;Plymouth,England)中培养。1. HUVEC cells (from mixed blood donors) were purchased from Clonetics (San Diego, CA) and cultured according to the manufacturer's instructions. Only the early passages (passages 3-8) were used for this analysis. Cells were cultured in 100 mm dishes (Falcon for Tissue Culture; Becton Dickinson; Plymouth, England) using complete EBM medium (Clonetics).
2.为了评价化合物的抑制活性,将胰蛋白酶作用于细胞,并将细胞以0.5-1.0×105细胞/孔的量接种在6孔簇板(Costar;Cambridge,MA)的每一孔中。2. In order to evaluate the inhibitory activity of the compounds, trypsin was applied to the cells, and the cells were seeded in each well of a 6-well cluster plate (Costar; Cambridge, MA) at an amount of 0.5-1.0×10 5 cells/well.
3.接种3-4天后,该板达到90-100%融合。从所有孔中移出培养基,用5-10ml PBS淋洗细胞,并用无添加物(即无血清)的5ml EBM基础培养基培养18-24小时。3. 3-4 days after inoculation, the plate reaches 90-100% confluency. Remove the medium from all wells, rinse the cells with 5-10ml of PBS, and incubate with 5ml of EBM basal medium without additives (i.e., serum-free) for 18-24 hours.
4.向1ml EBM培养基(25μM、5μM或1μM细胞终浓度)中加入系列稀释的抑制剂,并在37℃培养1小时。然后向所有孔中加入在2mlEBM培养基中、终浓度为50ng/ml的人重组VEGF165(R & DSystems),并在37℃培养10分钟。未处理或者仅用VEGF处理的对照细胞用于评价背景磷酸化作用和由VEGF引起的磷酸化作用。然后用含有1mM原钒酸钠(Sigma)的5-10ml冷的PBS淋洗所有孔,使细胞溶解,并在含有蛋白酶抑制剂(PMSF 1mM、抑肽酶1μg/ml、胃酶抑素1μg/ml,亮抑肽酶1μg/ml、钒酸钠1mM、氟化钠1mM)和1μg/mlDnase的200μl RIPA缓冲剂(50mM Tris-HCl pH7、150mM NaCl、1%NP-40、0.25%脱氧胆酸钠、1mM EDTA)中捣碎(所有化学品均得自Sigma Chemical Company,St Louis,MO)。溶胞产物以14,000rpm的转速旋转以除去核。4. Add serially diluted inhibitors to 1ml EBM medium (25μM, 5μM or 1μM final cell concentration) and incubate at 37°C for 1 hour. Human recombinant VEGF165 (R & D Systems) at a final concentration of 50 ng/ml in 2 ml of EBM medium was then added to all wells and incubated at 37°C for 10 minutes. Control cells, untreated or treated with VEGF only, were used to assess background phosphorylation and phosphorylation induced by VEGF. All wells were then rinsed with 5-10 ml of cold PBS containing 1 mM sodium orthovanadate (Sigma), the cells were lysed, and the wells were incubated with protease inhibitors (PMSF 1 mM, aprotinin 1 μg/ml, pepstatin 1 μg/ml) ml, 200 μl RIPA buffer (50 mM Tris-HCl pH 7, 150 mM NaCl, 1% NP-40, 0.25% deoxycholic acid Sodium, 1 mM EDTA) (all chemicals were obtained from Sigma Chemical Company, St Louis, MO). The lysate was spun at 14,000 rpm to remove nuclei.
然后通过加入冷(-20℃)乙醇(2倍体积)最少1小时或最多过夜,使等量的蛋白沉淀。沉淀物在含有5%巯基乙醇的Laemli样品缓冲剂(BioRad;Hercules,CA)中重新配制,并煮沸5分钟。通过聚丙烯酰胺凝胶电泳(6%,1.5mm Novex,San Deigo,CA)解析蛋白,并使用Novex系统将其转移到硝酸纤维素膜上。用牛血清白蛋白(3%)封闭后,该蛋白在4℃用抗-KDR多克隆抗体(C20,Santa Cruz Biotechnology;SantaCruz,CA)或者用抗磷酸酪氨酸单克隆抗体(4G10,UpstateBiotechnology,Lake Placid,NY)探测过夜。洗涤并用HRP-共轭的F(ab)2山羊抗-兔或山羊-抗-小鼠IgG培养1小时后,使用发射化学发光(ECL)系统(Amersham Life Sciences,Arlington Height,IL)目测检验谱带。某些本发明的实例在小于50μM的浓度显著抑制了细胞VEGF诱导的KDR酪氨酸激酶磷酸化。An equal amount of protein was then precipitated by the addition of cold (-20°C) ethanol (2 volumes) for a minimum of 1 hour or a maximum of overnight. The pellet was reconstituted in Laemli sample buffer (BioRad; Hercules, CA) containing 5% mercaptoethanol and boiled for 5 minutes. Proteins were resolved by polyacrylamide gel electrophoresis (6%, 1.5 mm Novex, San Deigo, CA) and transferred to nitrocellulose membranes using the Novex system. After blocking with bovine serum albumin (3%), the protein was incubated at 4°C with anti-KDR polyclonal antibody (C20, Santa Cruz Biotechnology; Santa Cruz, CA) or with anti-phosphotyrosine monoclonal antibody (4G10, Upstate Biotechnology, Lake Placid, NY) probed overnight. After washing and incubation with HRP-conjugated F(ab) 2 goat anti-rabbit or goat-anti-mouse IgG for 1 h, the spectra were visually inspected using an emission chemiluminescence (ECL) system (Amersham Life Sciences, Arlington Height, IL). bring. Certain embodiments of the invention significantly inhibited cellular VEGF-induced KDR tyrosine kinase phosphorylation at concentrations less than 50 [mu]M.
体内子宫水肿模型In vivo uterine edema model
该分析测量了化合物在小鼠中抑制子宫重量急性增加的能力,小鼠子宫重量急性增加发生在用雌激素刺激后的头几个小时内。这种早期发生的子宫重量增加已知是由于子宫脉管系统透过性增加引起的水肿造成的。Cullinan-Bove和Koss(Endocrinology(1993),133:829-837)证明了雌激素刺激的子宫水肿与子宫中VEGF mRNA表达增加的亲密的暂时关系。这些结果已经通过使用中和VEGF的单克隆抗体得以证实,而中和VEGF的单克隆抗体明显减少了雌激素刺激后子宫重量的急性增加(WO97/42187)。因此,该系统可以用作体内抑制VEGF信号传递以及相关的渗透性过高和水肿的模型。The assay measures the compound's ability to inhibit the acute increase in uterine weight in mice that occurs within the first few hours after stimulation with estrogen. This early onset of uterine weight gain is known to be due to edema caused by increased permeability of the uterine vasculature. Cullinan-Bove and Koss (Endocrinology (1993), 133:829-837) demonstrated an intimate temporal relationship between estrogen-stimulated uterine edema and increased expression of VEGF mRNA in the uterus. These results have been demonstrated using a neutralizing monoclonal antibody against VEGF which significantly reduced the acute increase in uterine weight following estrogen stimulation (WO97/42187). Therefore, this system can be used as a model for the inhibition of VEGF signaling in vivo and the associated hyperosmolarity and edema.
材料:所有激素均以冻干粉末的形式购自Sigma(St.Louis,MO)或Cal Biochem(La Jolla,CA)并按照供应商的说明制备。Materials: All hormones were purchased from Sigma (St. Louis, MO) or Cal Biochem (La Jolla, CA) as lyophilized powders and prepared according to the supplier's instructions.
载体组分(DMSO,Cremaphor EL)购自Sigma(St.Louis,MO)。Vehicle components (DMSO, Cremaphor EL) were purchased from Sigma (St. Louis, MO).
小鼠(Balb/c,8-12周龄)购自Taconic(Germantown,NY),并按照公共的动物保护与使用委员会指南(Animal Care and Use CommitteeGuidlines)将其饲养在无病原体的动物饲养设施中。Mice (Balb/c, 8-12 weeks old) were purchased from Taconic (Germantown, NY) and maintained in a pathogen-free animal husbandry facility according to public Animal Care and Use Committee Guidelines .
方法:method:
第1天:给Balb/c小鼠腹膜内(i.p.)注射12.5单位怀孕母马的血清促性腺激素(PMSG).Day 1: Balb/c mice were injected intraperitoneally (i.p.) with 12.5 units of pregnant mare's serum gonadotropin (PMSG).
第3天:小鼠腹膜内接受15单位的人绒毛膜的促性腺激素(hCG)。Day 3: Mice received 15 units of human chorionic gonadotropin (hCG) intraperitoneally.
第4天:将小鼠随机分成5-10只一组。根据溶解度和载体,通过腹膜内或静脉内或口服途径施用1-100mg/kg的试验化合物。载体对照组只接受载体,并且有两组不做处理。Day 4: Mice were randomly divided into groups of 5-10. Depending on solubility and vehicle, 1-100 mg/kg of test compound is administered by intraperitoneal or intravenous or oral routes. The vehicle control group received vehicle only, and two groups received no treatment.
30分钟后,对实验组、载体组和1个未处理组动物腹膜内注射17-雌二醇(500μg/kg)。2-3小时后,通过吸入CO2处死动物。沿中线切开,分离每只子宫,并在子宫颈下和子宫与输卵管的接合处切除。小心而不破坏子宫完整性地除去脂肪和结缔组织,然后称重(湿重)。通过将子宫在两张滤纸之间用一只装满水的1升玻璃瓶压榨,吸干子宫的液体。吸干后再对子宫称重(吸干后重量)。湿重与吸干后重量的差别为子宫的液体含量。将处理组的平均液体含量与未处理组或载体处理组进行比较。通过学生试验(Student′s test)测定显著性。未刺激的对照组用于监测雌二醇反应。Thirty minutes later, 17-estradiol (500 μg/kg) was intraperitoneally injected into the animals of the experimental group, the vehicle group and one untreated group. After 2-3 hours, sacrifice the animals by CO inhalation. An incision was made along the midline, each uterus was separated, and excised below the cervix and at the junction of the uterus and the fallopian tubes. Fat and connective tissue were removed carefully without disrupting the integrity of the uterus and weighed (wet weight). Uterine fluid was blotted by squeezing the uterus between two sheets of filter paper with a 1-liter glass bottle filled with water. After blotting, the uterus was weighed (weight after blotting). The difference between the wet weight and the blotted weight is the fluid content of the uterus. The mean liquid content of the treated groups was compared to the untreated or vehicle treated groups. Significance was determined by Student's test. An unstimulated control group was used to monitor estradiol responses.
结果表明,当通过各种途径系统给药时,某些本发明化合物抑制水肿形成。The results show that certain compounds of the invention inhibit edema formation when administered systemically by various routes.
作为血管生成受体酪氨酸激酶抑制剂的某些本发明化合物在新血管形成的Matrigel植入物模型中也显示出活性。该Matrigel新血管形成模型涉及在皮下植入的澄清″大理石″细胞外基质中形成新血管,这是由产生肿瘤细胞的原血管生成因子的存在导致的(例如参见:Passaniti,A.,等,Lab.Investig.(1992),67(4),519-528;Anat.Rec.(1997),249(1),63-73;Int.J.Cancer(1995),63(5),694-701;Vasc.Biol.(1995),15(11),1857-6)。该模型优选运行3-4天,并且终点包括与未用抑制剂处理的动物相对的、在切除植入物之后新血管形成的肉眼可见/影像评分、显微镜下的微血管密度测定和血红蛋白定量分析(Drabkinmethod)。该模型还可以施用bFGF或HGF作为刺激物。Certain compounds of the invention that are inhibitors of angiogenic receptor tyrosine kinases also exhibit activity in the Matrigel implant model of neovascularization. The Matrigel model of neovascularization involves the formation of new blood vessels in a subcutaneously implanted clear "marble" extracellular matrix, which results from the presence of pro-angiogenic factors that generate tumor cells (see for example: Passaniti, A., et al. Lab.Investig.(1992), 67(4), 519-528; Anat.Rec.(1997), 249(1), 63-73; Int.J.Cancer(1995), 63(5), 694- 701; Vasc. Biol. (1995), 15(11), 1857-6). The model is preferably run for 3-4 days, and endpoints include macroscopic/visual scoring of neovascularization after excision of the implant, microvessel densitometry and quantitative analysis of hemoglobin relative to animals not treated with inhibitors ( Drabkin method). The model can also administer bFGF or HGF as stimuli.
抑制一种或多种癌基因、原癌基因或增殖依赖性蛋白激酶或血管生成受体PTK的某些本发明化合物还抑制原发小鼠、大鼠或人的异种皮移植肿瘤在小鼠中的生长或者抑制其在小鼠模型中的转移。Certain compounds of the invention that inhibit one or more oncogenes, proto-oncogenes, or proliferation-dependent protein kinases or angiogenic receptor PTKs also inhibit primary mouse, rat, or human xenograft tumors in mice growth or inhibit its metastasis in mouse models.
示例example
通过碱催化羟醛缩合、然后进行消除反应合成本发明化合物的核心结构。反应方案I是该反应的一般表示。按照一般反应方案I合成本发明通式IV化合物的合适的原料II和III是可以购买到的和/或可以按照本领域公知的方法制备和/或按照本文所述方法制备。The core structures of the compounds of the invention were synthesized by base-catalyzed aldol condensation followed by elimination reactions. Reaction Scheme I is a general representation of this reaction. Suitable starting materials II and III for the synthesis of compounds of general formula IV of the present invention according to general reaction scheme I are commercially available and/or can be prepared according to methods well known in the art and/or as described herein.
反应方案I:4-[(取代的)亚甲基]-2-吡唑啉-5-酮的一般合成方法。Reaction Scheme I: General synthesis of 4-[(substituted)methylene]-2-pyrazolin-5-ones.
1. 4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮(V)。1. 4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one (V).
实施例1:3-环丙基-4-[(4,5-二甲基吡咯-2-基)亚甲基]-2-吡唑啉-5-酮。Example 1: 3-cyclopropyl-4-[(4,5-dimethylpyrrol-2-yl)methylene]-2-pyrazolin-5-one.
将3-环丙基-2-吡唑啉-5-酮(790mg,6.3mmol)、4,5-二甲基吡咯-2-甲醛(800mg,6.5mmol)和100mg哌啶在50mL乙醇中的反应混合物在90℃搅拌3.5小时。减压除去溶剂。固体残余物经闪式柱色谱纯化,使用(3∶7)乙酸乙酯∶己烷作流动相,然后通过从甲苯中重结晶进一步纯化。3-cyclopropyl-2-pyrazolin-5-one (790mg, 6.3mmol), 4,5-dimethylpyrrole-2-carbaldehyde (800mg, 6.5mmol) and 100mg piperidine in 50mL ethanol The reaction mixture was stirred at 90°C for 3.5 hours. The solvent was removed under reduced pressure. The solid residue was purified by flash column chromatography using (3:7) ethyl acetate:hexane as mobile phase, followed by further purification by recrystallization from toluene.
实施例40:3-叔丁基脲基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮。Example 40: 3-tert-butylureido-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one.
将3-氨基-4-[(吡咯-2-基)亚甲基]-2-吡唑啉-5-酮(1.7mmol)和异氰酸叔丁酯(1mL)在5mL乙酸乙酯中的反应混合物在80℃搅拌2小时。过滤收集产物并从乙醇中重结晶。A mixture of 3-amino-4-[(pyrrol-2-yl)methylene]-2-pyrazolin-5-one (1.7 mmol) and tert-butyl isocyanate (1 mL) in 5 mL of ethyl acetate The reaction mixture was stirred at 80°C for 2 hours. The product was collected by filtration and recrystallized from ethanol.
实施例88:4-{[4-(2-二甲氨基乙氧羰基)-3,5-二甲基吡咯-2-基]亚甲基}-3-异丙氧基-2-吡唑啉-5-酮。Example 88: 4-{[4-(2-Dimethylaminoethoxycarbonyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-isopropoxy-2-pyrazole Lin-5-one.
将4-[(4-羧基-3,5-二甲基吡咯-2-基)亚甲基]-3异丙氧基-2-吡唑啉-5-酮(200mg,0.68mmol)在二氯甲烷(20ml)和1滴DMF中的混合物用草酰氯(0.2ml)处理。继续搅拌24小时,并加入二甲氨基乙醇(0.6g,6.7mmol)在二氯甲烷(5ml)中的溶液。继续搅拌24小时。蒸发溶剂至干,固体残余物用NaHCO3(5%)处理并用二氯甲烷萃取。除去溶剂后得到的固体从正庚烷中重结晶。4-[(4-carboxy-3,5-dimethylpyrrol-2-yl)methylene]-3 isopropoxy-2-pyrazolin-5-one (200mg, 0.68mmol) in A mixture of methyl chloride (20ml) and 1 drop of DMF was treated with oxalyl chloride (0.2ml). Stirring was continued for 24 hours and a solution of dimethylaminoethanol (0.6 g, 6.7 mmol) in dichloromethane (5 ml) was added. Stirring was continued for 24 hours. The solvent was evaporated to dryness, the solid residue was treated with NaHCO3 (5%) and extracted with dichloromethane. The solid obtained after removal of the solvent was recrystallized from n-heptane.
实施例194:4-{[4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-基]亚甲基}-3-吡嗪基-2-吡唑啉-5-酮N-氧化物Example 194: 4-{[4-(2-Diethylaminoethyl)-3,5-dimethylpyrrol-2-yl]methylene}-3-pyrazinyl-2-pyrazoline- 5-keto N-oxide
将实施例176(0.2g,0.54mmol)和间氯过苯甲酸(0.2g,1.1mmol)在氯仿(50ml)中的混合物在室温搅拌48小时。除去溶剂至干,残余物经闪式色谱纯化(二氯甲烷/乙醇9∶1)。产率30%,熔点192℃(乙醇)。A mixture of EXAMPLE 176 (0.2 g, 0.54 mmol) and m-chloroperbenzoic acid (0.2 g, 1.1 mmol) in chloroform (50 mL) was stirred at room temperature for 48 hours. The solvent was removed to dryness and the residue was purified by flash chromatography (dichloromethane/ethanol 9:1). Yield 30%, melting point 192°C (ethanol).
表1:具有结构式V的其他合成化合物
表1A:具有结构式V的合成化合物的物理数据。
*对于元素分析,所计算的分子量包括所示量的溶剂。 * For elemental analysis, calculated molecular weights include the indicated amounts of solvent.
脚注:在室温下通过在乙醇中向游离碱中加入马来酸的乙醇溶液,形成马来酸盐,然后冷却,并过滤结晶的产品。Footnote: The maleate salt was formed by adding an ethanolic solution of maleic acid to the free base in ethanol at room temperature, then cooled, and the crystallized product was filtered.
II.4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮(VI)。II. 4-[(Indol-3-yl)methylene]-2-pyrazolin-5-one (VI).
实施例210:3-苄基-4-[(吲哚-3-基)亚甲基]-2-吡唑啉-5-酮.Example 210: 3-Benzyl-4-[(indol-3-yl)methylene]-2-pyrazolin-5-one.
将3-苄基-2-吡唑啉-5-酮(905mg,5.2mmol)、3-吲哚甲醛(800mg,5.6mmol)和哌啶(100mg)在乙醇(50mL)中的反应混合物在90℃搅拌3小时。冷却过夜后,过滤收集产物,然后从乙醇中重结晶进行纯化。A reaction mixture of 3-benzyl-2-pyrazolin-5-one (905mg, 5.2mmol), 3-indolecarbaldehyde (800mg, 5.6mmol) and piperidine (100mg) in ethanol (50mL) was heated at 90 °C and stirred for 3 hours. After cooling overnight, the product was collected by filtration and purified by recrystallization from ethanol.
表2:其他具有结构式VI的合成化合物。
表2A:具有结构式VI的合成化合物的物理数据。
*对于元素分析,所计算的分子量包括所示量的溶剂。 * For elemental analysis, calculated molecular weights include the indicated amounts of solvent.
脚注:在室温下通过在乙醇中向游离碱中加入马来酸的乙醇溶液,形成马来酸盐,然后冷却,并过滤结晶的产品。Footnote: The maleate salt was formed by adding an ethanolic solution of maleic acid to the free base in ethanol at room temperature, then cooled, and the crystallized product was filtered.
III.4-[(吲哚-2-基)亚甲基]-2-吡唑啉-5-酮(VII)。III. 4-[(Indol-2-yl)methylene]-2-pyrazolin-5-one (VII).
表3:具有结构式VII的合成化合物。
表3A:具有结构式VII的合成化合物的物理数据。
*对于元素分析,所计算的分子量包括所示量的溶剂。 * For elemental analysis, calculated molecular weights include the indicated amounts of solvent.
IV.4-[(7-氮杂吲哚-3-基)亚甲基]-2-吡唑啉-5-酮(VIII)。IV. 4-[(7-Azaindol-3-yl)methylene]-2-pyrazolin-5-one (VIII).
表4:具有结构式VIII的合成化合物。
表4A:具有结构式VIII的合成化合物的物理数据。
*对于元素分析,所计算的分子量包括所示量的溶剂。 * For elemental analysis, calculated molecular weights include the indicated amounts of solvent.
脚注:在室温下通过在乙醇中向游离碱中加入马来酸的乙醇溶液,形成马来酸盐,然后冷却,并过滤结晶的产品。Footnote: The maleate salt was formed by adding an ethanolic solution of maleic acid to the free base in ethanol at room temperature, then cooled, and the crystallized product was filtered.
V.4-(苯基亚甲基)-2-吡唑啉-5-酮(IX)。V. 4-(Phenylmethylene)-2-pyrazolin-5-one (IX).
表5:具有结构式IX的合成化合物。
表5A:具有结构式IX的合成化合物的物理数据。
VI.4-[(吡唑-4-基)亚甲基]-2-吡唑啉-5-酮(X)。VI. 4-[(Pyrazol-4-yl)methylene]-2-pyrazolin-5-one (X).
表6:具有结构式X的合成化合物。
表6A:具有结构式X的合成化合物的物理数据。
VII.4-[(咪唑-2-基)亚甲基]-2-吡唑啉-5-酮(XI)。VII. 4-[(Imidazol-2-yl)methylene]-2-pyrazolin-5-one (XI).
表7:具有结构式XI的合成化合物。
表7A:具有结构式XI的合成化合物的物理数据。
VIII.4-[(咪唑-4-基)亚甲基]-2-吡唑啉-5-酮(XII)。VIII. 4-[(Imidazol-4-yl)methylene]-2-pyrazolin-5-one (XII).
表8:具有结构式XII的合成化合物。
表8A:具有结构式XII的合成化合物的物理数据。
IX.4-[(噻吩-2-基)亚甲基]-2-吡唑啉-5-酮(XIII)。IX. 4-[(Thien-2-yl)methylene]-2-pyrazolin-5-one (XIII).
表9:具有结构式XIII的合成化合物。
表9A:具有结构式XIII的合成化合物的物理数据。
X.4-[(吲哚-3-基)亚甲基]-1-甲基-2-吡唑啉-5-酮(XIV)。X. 4-[(Indol-3-yl)methylene]-1-methyl-2-pyrazolin-5-one (XIV).
表10:具有结构式XIV的合成化合物。
表10A:具有结构式XIV的合成化合物的物理数据。
XI.4-[(吡咯-3-基)亚甲基]-2-吡唑啉-5-酮(XV)。XI. 4-[(Pyrrol-3-yl)methylene]-2-pyrazolin-5-one (XV).
表11:具有结构式XV的合成化合物。
表11A:具有结构式XV的合成化合物的物理数据。
XII.4-[(喹啉-5-基)亚甲基]-2-吡唑啉-5-酮(XVI)。XII. 4-[(Quinolin-5-yl)methylene]-2-pyrazolin-5-one (XVI).
表12:具有结构式XVI的合成化合物。
表12A:具有结构式XVI的合成化合物的物理数据。
X.原料合成。X. Raw material synthesis.
A.合成3-酮基酯(XIX)。A. Synthesis of 3-ketoesters (XIX).
反应方案II中示出了采用一般方法合成3-酮基酯。The general procedure for the synthesis of 3-ketoesters is shown in Reaction Scheme II.
反应方案II:合成3-酮基酯Reaction Scheme II: Synthesis of 3-ketoesters
实施例371:3-(2,2,3,3-四甲基环丙基)-3-氧代丙酸乙酯。Example 371: Ethyl 3-(2,2,3,3-tetramethylcyclopropyl)-3-oxopropanoate.
在氮气氛下,向冰冷却的2,2-二甲基-1,3-二噁烷-4,7-二酮(4.7g,0.032mol)在无水二氯甲烷(37mL)中的溶液中加入吡啶(5g,0.063mol)和2,2,3,3-四甲基环丙基羰基氯(5g,0.031mol)。在0℃保持1小时,然后使其升至室温过夜。然后将该混合物转入分液漏斗,用3%盐酸洗涤两次,并用水洗涤两次。有机层用硫酸镁干燥,然后过滤并减压浓缩,得到5g深红色油。该油在无水乙醇(60mL)中回流3小时。蒸发溶剂,产物经蒸馏纯化。To an ice-cooled solution of 2,2-dimethyl-1,3-dioxane-4,7-dione (4.7 g, 0.032 mol) in anhydrous dichloromethane (37 mL) under nitrogen atmosphere To this was added pyridine (5 g, 0.063 mol) and 2,2,3,3-tetramethylcyclopropylcarbonyl chloride (5 g, 0.031 mol). It was held at 0°C for 1 hour, then allowed to warm to room temperature overnight. The mixture was then transferred to a separatory funnel and washed twice with 3% hydrochloric acid and twice with water. The organic layer was dried over magnesium sulfate, then filtered and concentrated under reduced pressure to give 5 g of a dark red oil. The oil was refluxed in absolute ethanol (60 mL) for 3 hours. The solvent was evaporated and the product was purified by distillation.
实施例377:3-(6-甲氧基苯并噻唑-2-基)-3-氧代丙酸乙酯。Example 377: Ethyl 3-(6-methoxybenzothiazol-2-yl)-3-oxopropanoate.
向氢化钠(1.5g,50%油分散液,0.032mol)中加入乙酸乙酯(7.2g,0.082mol),保持温度低于25℃。加入2-乙氧羰基-2-(6-甲氧基)苯并噻唑(5.25g,0.022mol)。温热该混合物至轻轻回流,然后在室温搅拌过夜。用冰水(50mL)终止反应,并用浓盐酸调节至pH7.7。产物沉淀,过滤收集,用水洗涤,然后干燥。To sodium hydride (1.5 g, 50% dispersion in oil, 0.032 mol) was added ethyl acetate (7.2 g, 0.082 mol) keeping the temperature below 25°C. 2-Ethoxycarbonyl-2-(6-methoxy)benzothiazole (5.25 g, 0.022 mol) was added. The mixture was warmed to gentle reflux, then stirred at room temperature overnight. The reaction was quenched with ice water (50 mL) and adjusted to pH 7.7 with concentrated hydrochloric acid. The product precipitated and was collected by filtration, washed with water, and dried.
实施例378:4-(4-苯基苯氧基)-3-氧代丁酸乙酯。Example 378: Ethyl 4-(4-phenylphenoxy)-3-oxobutanoate.
在0℃,用2小时,向75mL二甲基甲酰胺(在下文中称作″DMF″)中加入氢化钠(5.0g,0.114mol)和4-苯基苯酚(15.3g,0.09mol)。加入在DMF(75mL)中的4-氯乙酰乙酸乙酯(5.0g,0.03mol)。该混合物在0℃搅拌1小时,并在室温搅拌12小时,然后用50%磷酸中和,并用二氯甲烷萃取。该产物经硅胶色谱纯化,用(8∶2)甲苯∶乙醇作流动相。To 75 mL of dimethylformamide (hereinafter referred to as "DMF") were added sodium hydride (5.0 g, 0.114 mol) and 4-phenylphenol (15.3 g, 0.09 mol) at 0°C over 2 hours. Ethyl 4-chloroacetoacetate (5.0 g, 0.03 mol) in DMF (75 mL) was added. The mixture was stirred at 0°C for 1 hour and at room temperature for 12 hours, then neutralized with 50% phosphoric acid and extracted with dichloromethane. The product was purified by silica gel chromatography using (8:2) toluene:ethanol as mobile phase.
表13:具有结构式XIX的合成化合物。
B.合成环戊氧基硫代羰基乙酸乙酯。B. Synthesis of ethyl cyclopentyloxythiocarbonyl acetate.
反应方案III:合成烷氧基或芳氧基亚胺代羰基乙酸乙酯盐酸盐(R9是取代或未取代的脂族或芳族基团)。Reaction Scheme III: Synthesis of ethyl alkoxy or aryloxy iminocarboacetate hydrochloride (R 9 is a substituted or unsubstituted aliphatic or aromatic group).
实施例381:合成环戊氧基亚胺代羰基乙酸乙酯盐酸盐(XXII)。Example 381: Synthesis of ethyl cyclopentyloxyiminocarboacetate hydrochloride (XXII).
将无水氰基乙酸乙酯(14g,0.12mol)和无水环戊醇(12g,0.15mol的混合物在0℃用氯化氢气饱和3小时,在0℃保持过夜,然后用乙醚稀释。产物沉淀,过滤收集并用乙醚洗涤。A mixture of anhydrous ethyl cyanoacetate (14 g, 0.12 mol) and anhydrous cyclopentanol (12 g, 0.15 mol) was saturated with hydrogen chloride gas at 0°C for 3 hours, kept at 0°C overnight, and then diluted with diethyl ether. The product precipitated , collected by filtration and washed with ether.
产量22.4g,(77%),熔点110-112℃Yield 22.4 g, (77%), melting point 110-112°C
反应方案IV:合成烷氧基硫代或芳氧基硫代羰基乙酸乙酯。Reaction Scheme IV: Synthesis of ethyl alkoxythio or aryloxythiocarbonyl acetates.
实施例382:环戊氧基硫代羰基乙酸乙酯(XXIV)Example 382: Ethyl cyclopentyloxythiocarbonyl acetate (XXIV)
将环戊氧基亚胺代羰基乙酸乙酯盐酸盐(22g 0.09mol)在无水吡啶(150ml)中的混合物用硫化氢处理8小时,在室温保持48小时。该反应混合物用浓盐酸酸化,并用乙醚萃取。醚相用水洗涤,干燥并经蒸馏纯化。A mixture of ethyl cyclopentyloxyiminocarbonyl acetate hydrochloride (22 g 0.09 mol) in anhydrous pyridine (150 ml) was treated with hydrogen sulfide for 8 hours and kept at room temperature for 48 hours. The reaction mixture was acidified with concentrated hydrochloric acid and extracted with ether. The ether phase is washed with water, dried and purified by distillation.
产量15g,(75%),熔点140-142℃(15mm Hg)。Yield 15g, (75%), melting point 140-142°C (15mm Hg).
C.合成2-吡唑啉-5-酮C. Synthesis of 2-pyrazolin-5-one
通过以下四种方法合成2-吡唑啉-5-酮。2-Pyrazolin-5-one was synthesized by the following four methods.
反应方案V:合成2-吡唑啉-5-酮的方法1。Reaction Scheme V: Method 1 for the synthesis of 2-pyrazolin-5-ones.
反应方案VI:合成2-吡唑啉-5-酮的方法2。Reaction Scheme VI: Method 2 for the synthesis of 2-pyrazolin-5-ones.
反应方案VII:合成2-吡唑啉-5-酮的方法3。Reaction Scheme VII: Method 3 for the synthesis of 2-pyrazolin-5-ones.
反应方案VIII:合成2-吡唑啉-5-酮的方法4。Reaction Scheme VIII: Method 4 for the synthesis of 2-pyrazolin-5-ones.
实施例383:3-(2,2,3,3-四甲基环丙基)-2-吡唑啉-5-酮。Example 383: 3-(2,2,3,3-Tetramethylcyclopropyl)-2-pyrazolin-5-one.
向3-(2,2,3,3-四甲基环丙基-3-氧代丙酸乙酯(4.41g,0.020mol)在无水乙醇(60ml)中的溶液中加入水合肼(1.1g,0.024mol)。使反应混合物回流3小时。冷却后,蒸发溶剂,残余物用己烷研制,得到白色固体。Add hydrazine hydrate (1.1 g, 0.024 mol). The reaction mixture was refluxed for 3 hours. After cooling, the solvent was evaporated and the residue was triturated with hexane to give a white solid.
实施例411:3-(4-羟基苄基)-2-吡唑啉-5-酮Example 411: 3-(4-Hydroxybenzyl)-2-pyrazolin-5-one
将搅拌着的3-(4-甲氧基苄基)-2-吡唑啉-5-酮(2.5mmol)在100ml二氯甲烷中的悬浮液在-78℃冷却,用7.5ml三溴化硼(1M二氯甲烷溶液)处理。在-78℃保持1小时后,在室温搅拌反应混合物12小时。用50ml水使该混合物终止反应。分离水相,用5%碳酸氢钠溶液碱化。过滤形成的沉淀,并从甲醇中结晶。A stirred suspension of 3-(4-methoxybenzyl)-2-pyrazolin-5-one (2.5 mmol) in 100 ml of dichloromethane was cooled at -78°C, and 7.5 ml of tribrominated Boron (1M solution in dichloromethane) was treated. After 1 hour at -78°C, the reaction mixture was stirred at room temperature for 12 hours. The mixture was quenched with 50 ml of water. The aqueous phase was separated and basified with 5% sodium bicarbonate solution. The formed precipitate was filtered and crystallized from methanol.
实施例413:3-(4-氨基甲酰基苯乙基)-2-吡唑啉-5-酮Example 413: 3-(4-Carbamoylphenethyl)-2-pyrazolin-5-one
将3-(4-甲氧羰基苯乙基)-2-吡唑啉-5-酮(7.7mmol)在25%氢氧化铵(30ml)中的混合物在室温搅拌12小时。真空浓缩溶剂,过滤形成的固体产物并干燥。A mixture of 3-(4-methoxycarbonylphenethyl)-2-pyrazolin-5-one (7.7 mmol) in 25% ammonium hydroxide (30 ml) was stirred at room temperature for 12 hours. The solvent was concentrated in vacuo and the solid product formed was filtered and dried.
实施例414:3-(2-氨基羰基乙基)-2-吡唑啉-5-酮Example 414: 3-(2-Aminocarbonylethyl)-2-pyrazolin-5-one
将3-(乙氧羰基甲基)-2-吡唑啉-5-酮(1g,5.8mmol)和NaCN(28mg,0.58mmol)在30ml 9M氨的甲醇溶液中的混合物在密封管中于45℃加热3天。冷却后,蒸发溶剂,将残余物悬浮在水中。过滤收集沉淀的固体。A mixture of 3-(ethoxycarbonylmethyl)-2-pyrazolin-5-one (1 g, 5.8 mmol) and NaCN (28 mg, 0.58 mmol) in 30 ml of 9M ammonia in methanol was placed in a sealed tube at 45 °C for 3 days. After cooling, the solvent was evaporated and the residue was suspended in water. The precipitated solid was collected by filtration.
实施例416:3-(4-甲氧基苯氨基)-2-吡唑啉-5-酮Example 416: 3-(4-Methoxyanilino)-2-pyrazolin-5-one
使3-氨基-2-吡唑啉-5-酮(20mmol)、4-甲氧基苯胺(25mol)在50ml乙酸中的混合物回流4小时。蒸发溶剂,将残余物悬浮在水中。过滤固体并从甲醇中结晶。A mixture of 3-amino-2-pyrazolin-5-one (20 mmol), 4-methoxyaniline (25 mol) in 50 ml acetic acid was refluxed for 4 hours. The solvent was evaporated and the residue was suspended in water. The solid was filtered and crystallized from methanol.
表14:具有结构式XXVI的合成化合物。
D.合成醛D. Synthetic aldehydes
实施例426:1-(4-乙酰氧基丁基)-氮杂吲哚-3-甲醛Example 426: 1-(4-Acetoxybutyl)-azaindole-3-carbaldehyde
保持温度在5-10℃,在氮气氛下,向搅拌着的60%氢化钠(油分散液)(1.2g,30mmol)在无水DMF(65ml)中的悬浮液中滴加7-氮杂吲哚-3-甲醛(4.19g,28mmol)在无水N,N-二甲基甲酰胺(50ml)中的溶液。添加完成后,在同样的温度下继续搅拌30分钟,然后滴加乙酸4-溴丁酯(6.16g,31mmol)在无水N,N-二甲基甲酰胺(15ml)中的溶液,使该混合物温热至室温。在室温搅拌反应混合物137小时,然后加入水(100ml),并用二氯甲烷(2×100ml)萃取该混合物。有机相用水(3×100ml)洗涤,用无水硫酸镁干燥,过滤并减压除去溶剂,得到油(6.8g,93%),该油无需进一步纯化即可使用。To a stirred suspension of 60% sodium hydride (oil dispersion) (1.2 g, 30 mmol) in anhydrous DMF (65 ml) was added dropwise 7-azepam under nitrogen atmosphere, keeping the temperature at 5-10 °C. A solution of indole-3-carbaldehyde (4.19 g, 28 mmol) in anhydrous N,N-dimethylformamide (50 ml). After the addition was complete, stirring was continued at the same temperature for 30 minutes, and then a solution of 4-bromobutyl acetate (6.16 g, 31 mmol) in anhydrous N, N-dimethylformamide (15 ml) was added dropwise to make the The mixture was warmed to room temperature. The reaction mixture was stirred at room temperature for 137 hours, then water (100ml) was added and the mixture was extracted with dichloromethane (2 x 100ml). The organic phase was washed with water (3 x 100ml), dried over anhydrous magnesium sulfate, filtered and the solvent removed under reduced pressure to give an oil (6.8g, 93%) which was used without further purification.
实施例427:1-(4-羟基丁基)-7-氮杂吲哚-3-甲醛Example 427: 1-(4-Hydroxybutyl)-7-azaindole-3-carbaldehyde
向1-(4-乙酰氧基丁基)-7-氮杂吲哚-3-甲醛(2.3g,8.8mmol)在甲醇(40ml)中的溶液中加入氢氧化钠(0.2g,17.5mmol)在水(35ml)中的溶液。该混合物在60℃加热0.5小时,然后蒸发溶剂至干。将残余物溶解在乙酸乙酯和水的混合物(50ml∶50ml)中,分离水层。有机相用水洗涤,用硫酸镁干燥,过滤并蒸发至干。残余物无需进一步纯化即可使用。产率78%。熔点90℃。To a solution of 1-(4-acetoxybutyl)-7-azaindole-3-carbaldehyde (2.3g, 8.8mmol) in methanol (40ml) was added sodium hydroxide (0.2g, 17.5mmol) Solution in water (35ml). The mixture was heated at 60°C for 0.5 hours, then the solvent was evaporated to dryness. The residue was dissolved in a mixture of ethyl acetate and water (50ml:50ml), and the aqueous layer was separated. The organic phase was washed with water, dried over magnesium sulfate, filtered and evaporated to dryness. The residue was used without further purification. Yield 78%. The melting point is 90°C.
实施例428:1-(4-羟基丁基)吡咯-2-甲醛Example 428: 1-(4-Hydroxybutyl)pyrrole-2-carbaldehyde
按照实施例51的方法得到产物,不同的是用吡咯-2-甲醛代替7-氮杂吲哚-3-甲醛。该产物经硅胶色谱纯化,用(5∶5)乙酸乙酯∶己烷洗脱。产率67%。The product was obtained as in Example 51, except that pyrrole-2-carbaldehyde was used instead of 7-azaindole-3-carbaldehyde. The product was purified by silica gel chromatography eluting with (5:5) ethyl acetate:hexane. Yield 67%.
实施例429:3-异丙基-5-甲基吡咯-2-甲醛Example 429: 3-Isopropyl-5-methylpyrrole-2-carbaldehyde
在0℃和氮气氛下,向无水DMF(2.1ml,27mmol)在无水二氯乙烷(50ml)中的溶液中滴加POCl3(2.1ml,23mmol)。将该溶液温热至室温1小时。然后,将该悬浮液冷却至0℃,并用20分钟滴加3-异丙基-5-甲基吡咯(2g,16mmol)在二氯乙烷(20ml)中的溶液。在室温搅拌24小时后,加入水,然后加入NaOH至pH:8。萃取有机层,干燥并除去溶剂至干燥。残余物经闪式色谱纯化。产率67%,熔点90-93℃。To a solution of anhydrous DMF (2.1 ml, 27 mmol) in anhydrous dichloroethane (50 ml) was added POCl3 (2.1 ml, 23 mmol) dropwise at 0°C under nitrogen atmosphere. The solution was warmed to room temperature for 1 hour. Then, the suspension was cooled to 0° C., and a solution of 3-isopropyl-5-methylpyrrole (2 g, 16 mmol) in dichloroethane (20 ml) was added dropwise over 20 minutes. After stirring at room temperature for 24 hours, water was added, followed by NaOH to pH:8. The organic layer was extracted, dried and stripped of solvent to dryness. The residue was purified by flash chromatography. Yield 67%, melting point 90-93°C.
实施例430:3,5-二甲基-4-吗啉代甲基吡咯-2-甲醛Example 430: 3,5-Dimethyl-4-morpholinomethylpyrrole-2-carbaldehyde
向吗啉(0.35g,4mmol)、37%的甲醛(0.32g,4mmol)和乙酸(2ml)的溶液中加入3,5-二甲基吡咯-2-甲醛(0.5g,4mmol)。该混合物在室温和氮气氛下搅拌过夜,然后用NaHCO3稀释反应物至pH8。水溶液用乙酸乙酯萃取,有机层用硫酸镁干燥。真空蒸发乙酸乙酯,得到0.4g纯的固体。To a solution of morpholine (0.35g, 4mmol), 37% formaldehyde (0.32g, 4mmol) and acetic acid (2ml) was added 3,5-dimethylpyrrole-2-carbaldehyde (0.5g, 4mmol). The mixture was stirred at room temperature under nitrogen overnight, then the reaction was diluted to pH8 with NaHCO3 . The aqueous solution was extracted with ethyl acetate, and the organic layer was dried over magnesium sulfate. Ethyl acetate was evaporated in vacuo to give 0.4 g of pure solid.
按照与实施例430所述类似的方法合成下列化合物:Following a method similar to that described in Example 430, the following compounds were synthesized:
3,5-二甲基-4-二甲氨基甲基吡咯-2-甲醛,3,5-Dimethyl-4-dimethylaminomethylpyrrole-2-carbaldehyde,
3-(4-吗啉代)甲基-吲哚-2-甲醛,3-(4-morpholino)methyl-indole-2-carbaldehyde,
3,5-二甲基-4-二乙氨基甲基吡咯-2-甲醛,3,5-Dimethyl-4-diethylaminomethylpyrrole-2-carbaldehyde,
3,5-二甲基-4-哌啶子基甲基吡咯-2-甲醛,3,5-Dimethyl-4-piperidinomethylpyrrole-2-carbaldehyde,
3,5-二甲基-4-[(4-甲基-1-哌嗪基)甲基]吡咯-2-甲醛,3,5-Dimethyl-4-[(4-methyl-1-piperazinyl)methyl]pyrrole-2-carbaldehyde,
3,5-二甲基-4-(1-吡咯烷基甲基)吡咯-2-甲醛,3,5-Dimethyl-4-(1-pyrrolidinylmethyl)pyrrole-2-carbaldehyde,
5-甲基-4-二甲氨基甲基吡咯-2-甲醛,5-methyl-4-dimethylaminomethylpyrrole-2-carbaldehyde,
3,5-二甲基-4-[N-甲基-N-(2-二甲氨基乙基)氨基甲基]吡咯-2-甲醛,3,5-Dimethyl-4-[N-methyl-N-(2-dimethylaminoethyl)aminomethyl]pyrrole-2-carbaldehyde,
3,5-二甲基-4-二乙醇氨基甲基吡咯-2-甲醛,3,5-Dimethyl-4-diethanolaminomethylpyrrole-2-carbaldehyde,
4-(4-苄基哌啶子基甲基)-3,5-二甲基吡咯-2-甲醛,4-(4-Benzylpiperidinomethyl)-3,5-dimethylpyrrole-2-carbaldehyde,
4-二甲氨基甲基-3-异丙基-5-甲基吡咯-2-甲醛,4-Dimethylaminomethyl-3-isopropyl-5-methylpyrrole-2-carbaldehyde,
3,5-二甲基-4-(4-羟基哌啶子基甲基)吡咯-2-甲醛,和3,5-Dimethyl-4-(4-hydroxypiperidinomethyl)pyrrole-2-carbaldehyde, and
3-二甲氨基甲基-4,5-四亚甲基吡咯-2-甲醛。3-Dimethylaminomethyl-4,5-tetramethylenepyrrole-2-carbaldehyde.
实施例431:3,5-二甲基-4-三氟乙酰氨基甲基吡咯-2-甲醛Example 431: 3,5-Dimethyl-4-trifluoroacetamidomethylpyrrole-2-carbaldehyde
保持温度低于10℃,将3,5-二甲基吡咯-2-甲醛(2.6g,0.02mol)和N-(羟甲基)三氟乙酰胺(3g,0.02mol)的混合物分批加入硫酸(15ml)中。使反应混合物温热至室温4小时,将反应物倒入冰水(100ml)中,过滤沉淀的固体并用水洗涤。所得白色固体(产率78%)无需进一步纯化即可使用。M.p.200℃。Keeping the temperature below 10 °C, a mixture of 3,5-dimethylpyrrole-2-carbaldehyde (2.6 g, 0.02 mol) and N-(hydroxymethyl)trifluoroacetamide (3 g, 0.02 mol) was added in portions sulfuric acid (15ml). The reaction mixture was allowed to warm to room temperature for 4 hours, the reaction was poured into ice water (100ml), and the precipitated solid was filtered and washed with water. The resulting white solid (78% yield) was used without further purification. M.p.200°C.
实施例432:4-氨基甲基-3,5-二甲基吡咯-2-甲醛Example 432: 4-Aminomethyl-3,5-dimethylpyrrole-2-carbaldehyde
将3,5-二甲基-4-三氟乙酰氨基甲基吡咯-2-甲醛(3.9g,15.7mmol)、NaOH 10%(19ml)和甲醇(100ml)的混合物加热回流1小时。减压浓缩该溶液至一半体积。加入水,过滤沉淀的固体。产率72%,m.p.210-20℃(d)。A mixture of 3,5-dimethyl-4-trifluoroacetamidomethylpyrrole-2-carbaldehyde (3.9 g, 15.7 mmol), NaOH 10% (19 ml) and methanol (100 ml) was heated at reflux for 1 hour. The solution was concentrated under reduced pressure to half volume. Water was added and the precipitated solid was filtered. Yield 72%, m.p. 210-20°C (d).
实施例433:乙酸3-乙酰基-4-氧代戊基酯Example 433: 3-Acetyl-4-oxopentyl acetate
在机械搅拌下,将戊烷-2,4-二酮(60g,0.6mol)、乙酸2-溴乙酯(100g,0.6mol)、无水碳酸钾(83g,0.6mol)、IK(99.6g,0.6mol)和无水丙酮(500ml)的混合物加热回流24小时。减压浓缩该混合物,并将其倒入盐酸(1.7M,400ml)中。通过连续用乙醚、乙酸乙酯和二氯甲烷萃取,分离该产物。合并的有机提取液用盐水洗涤,除去溶剂至干,并蒸馏残余物,得到38g乙酸盐,b.p.75-80℃(0.1mm Hg)。Under mechanical stirring, pentane-2,4-dione (60g, 0.6mol), 2-bromoethyl acetate (100g, 0.6mol), anhydrous potassium carbonate (83g, 0.6mol), IK (99.6g , 0.6mol) and a mixture of anhydrous acetone (500ml) was heated to reflux for 24 hours. The mixture was concentrated under reduced pressure and poured into hydrochloric acid (1.7M, 400ml). The product was isolated by successive extractions with ether, ethyl acetate and dichloromethane. The combined organic extracts were washed with brine, the solvent was removed to dryness, and the residue was distilled to give 38 g of the acetate salt, b.p. 75-80°C (0.1 mm Hg).
按照与实施例433所述类似的方法,制备乙酸4-乙酰基-5-氧代己基酯[产率60%,b.p.110℃(0.2mm Hg)]和乙酸5-乙酰基-6-氧代己基酯[产率77%,b.p.102℃(0.2mm Hg)]。Following a procedure similar to that described in Example 433, 4-acetyl-5-oxohexyl acetate [60% yield, b.p. 110°C (0.2mm Hg)] and 5-acetyl-6-oxoacetate Hexyl ester [77% yield, b.p. 102°C (0.2 mm Hg)].
实施例434:4-(2-乙酰氧基乙基)-3,5-二甲基吡咯-2-甲酸叔丁酯Example 434: tert-Butyl 4-(2-acetoxyethyl)-3,5-dimethylpyrrole-2-carboxylate
在低于20℃温度和搅拌下,通过向乙酰乙酸叔丁酯(26g,0.165mol)在乙酸(48ml)中的溶液中滴加亚硝酸钠(11.3g,0.165mol)在水(17ml)中的溶液对其进行处理,并放置过夜。将乙酸3-乙酰基-4-氧代戊基酯(30g,0.161mol)在乙酸(56ml)中的溶液加热至80℃,此时分批加入锌粉(26.2g,0.40mol)和乙酸钠(26.3g,0.32mol)的混合物,同时滴加上述羟基亚氨基衍生物(97ml,0.165mol)。在添加期间剧烈搅拌反应混合物,使锌总是过量,并且保持温度在90-100℃。添加完成时,将该混合物轻轻煮沸15分钟,并在室温搅拌过夜。将反应混合物倒入冰水中,放置2hr。过滤固体并从乙醇水溶液中重结晶。产率78%,熔点92℃。To a solution of tert-butyl acetoacetate (26g, 0.165mol) in acetic acid (48ml) was added dropwise sodium nitrite (11.3g, 0.165mol) in water (17ml) at a temperature below 20°C under stirring. solution and left overnight. A solution of 3-acetyl-4-oxopentyl acetate (30 g, 0.161 mol) in acetic acid (56 ml) was heated to 80° C., at which point zinc powder (26.2 g, 0.40 mol) and sodium acetate were added in portions (26.3 g, 0.32 mol) of the mixture, while the above hydroxyimino derivative (97 ml, 0.165 mol) was added dropwise. The reaction mixture was stirred vigorously during the addition so that there was always an excess of zinc and the temperature was maintained at 90-100°C. When the addition was complete, the mixture was boiled gently for 15 minutes and stirred at room temperature overnight. The reaction mixture was poured into ice water and left for 2hr. The solid was filtered and recrystallized from aqueous ethanol. Yield 78%, melting point 92°C.
按照与实施例434所述类似的方法,制备4-(3-乙酰氧基丙基)-3,5-二甲基吡咯-2-甲酸叔丁酯[产60%,m.p.73℃(己烷)]和4-(4-乙酰氧基丁基)-3,5-二甲基吡咯-2-甲酸叔丁酯[产率52%,m.p.62℃(己烷)]。Following a procedure similar to that described in Example 434, tert-butyl 4-(3-acetoxypropyl)-3,5-dimethylpyrrole-2-carboxylate was prepared [yield 60%, m.p. 73°C (hexane )] and tert-butyl 4-(4-acetoxybutyl)-3,5-dimethylpyrrole-2-carboxylate [52% yield, m.p. 62°C (hexane)].
实施例435:3,5-二甲基-4-(2-羟基乙基)吡咯-2-甲醛。Example 435: 3,5-Dimethyl-4-(2-hydroxyethyl)pyrrole-2-carbaldehyde.
将4-(2-乙酰氧基乙基)-3,5-二甲基吡咯-2-甲酸叔丁酯(15g,53mmol)溶解在TFA(40ml)中,并在40℃和氮气氛下搅拌该溶液10分钟。然后将反应混合物冷却至0℃,并滴加原甲酸三乙酯(9.6ml,58mmol)。使该混合物温热至20℃,并搅拌1小时,然后倒入冰水中。用二氯甲烷萃取,依次用10%氨水和水洗涤该提取液,并用硫酸镁干燥。4-(2-乙酰氧基乙基)-3,5-二甲基吡咯-2-甲醛经硅胶色谱纯化,用(7∶3)乙酸乙酯∶己烷洗脱。产率45%,m.p.125-27℃(水)。Dissolve tert-butyl 4-(2-acetoxyethyl)-3,5-dimethylpyrrole-2-carboxylate (15 g, 53 mmol) in TFA (40 ml) and stir at 40 °C under nitrogen atmosphere The solution was left for 10 minutes. The reaction mixture was then cooled to 0°C and triethylorthoformate (9.6ml, 58mmol) was added dropwise. The mixture was warmed to 20°C and stirred for 1 hour, then poured into ice water. After extraction with dichloromethane, the extract was washed successively with 10% ammonia and water, and dried over magnesium sulfate. 4-(2-Acetoxyethyl)-3,5-dimethylpyrrole-2-carbaldehyde was purified by silica gel chromatography eluting with (7:3) ethyl acetate:hexane. Yield 45%, m.p. 125-27°C (water).
向上述化合物(5g,23mmol)在乙醇(20ml)中的溶液中加入NaOH10%(10ml)。在室温搅拌该混合物30分钟,浓缩最终的溶液,用水稀释,并用二氯甲烷萃取。干燥有机提取液,并除去溶剂至干。产率98%,m.p.100℃(乙酸乙酯)。To a solution of the above compound (5 g, 23 mmol) in ethanol (20 ml) was added NaOH 10% (10 ml). The mixture was stirred at room temperature for 30 minutes, and the final solution was concentrated, diluted with water, and extracted with dichloromethane. The organic extract was dried and the solvent removed to dryness. Yield 98%, m.p. 100°C (ethyl acetate).
按照与实施例435所述类似的方法,制备3,5-二甲基-4-(3-羟丙基)吡咯-2-甲醛[产率60%,m.p.85℃(乙酸乙酯/己烷)]和3,5-二甲基-4-(4-羟基丁基)吡咯-2-甲醛[产率38%,m.p.99℃(己烷)]。Following a procedure similar to that described in Example 435, 3,5-dimethyl-4-(3-hydroxypropyl)pyrrole-2-carbaldehyde was prepared [60% yield, m.p. 85°C (ethyl acetate/hexane )] and 3,5-dimethyl-4-(4-hydroxybutyl)pyrrole-2-carbaldehyde [38% yield, m.p. 99°C (hexane)].
实施例436:4-(2-二乙氨基乙基)-3,5-二甲基吡咯-2-甲醛。Example 436: 4-(2-Diethylaminoethyl)-3,5-dimethylpyrrole-2-carbaldehyde.
在0-5℃,向搅拌着3,5-二甲基-4-(2-羟基乙基)吡咯-2-甲醛(4.3g,25.7mmol)和三乙胺(5.2g,51mmol)在无水THF(60ml)中的溶液中加入在无水THF(4ml)中的甲磺酰氯(4ml,51mmol),并在室温搅拌该混合物1小时。然后向该混合物中加入水,过滤沉淀的固体。产率89%,m.p.142-44℃。At 0-5°C, 3,5-dimethyl-4-(2-hydroxyethyl)pyrrole-2-carbaldehyde (4.3g, 25.7mmol) and triethylamine (5.2g, 51mmol) were stirred in the absence of To a solution in aqueous THF (60ml) was added methanesulfonyl chloride (4ml, 51mmol) in anhydrous THF (4ml) and the mixture was stirred at room temperature for 1 hour. Water was then added to the mixture, and the precipitated solid was filtered. Yield 89%, m.p. 142-44°C.
将上述甲磺酸酯(23mmol)、K2CO3(23mmol)、二乙胺(90mmol)和2-丙醇(70ml)的混合物加热至100℃,保持0.5小时。除去溶剂至干,残余物用水处理,并用二氯甲烷萃取。干燥有机提取液,并蒸发溶剂至干。产率70%,m.p.70℃(正庚烷)。A mixture of the above mesylate (23mmol), K2CO3 (23mmol), diethylamine (90mmol) and 2-propanol (70ml) was heated to 100°C for 0.5h. The solvent was removed to dryness and the residue was treated with water and extracted with dichloromethane. The organic extract was dried, and the solvent was evaporated to dryness. Yield 70%, mp 70°C (n-heptane).
按照与实施例436所述类似的方法,以油的形式分离并制备3,5-二甲基-4-[2-(1-吡咯烷基乙基)]吡咯-2-甲醛、3,5-二甲基-4-(2-乙氨基乙基)吡咯-2-甲醛、4-(3-二乙氨基丙基)-3,5-二甲基吡咯-2-甲醛、4-(4-二乙氨基丁基)-3,5-二甲基吡咯-2-甲醛、3,5-二甲基-4-[3-(1-吡咯烷基丙基)]吡咯-2-甲醛。3,5-Dimethyl-4-[2-(1-pyrrolidinylethyl)]pyrrole-2-carbaldehyde, 3,5-dimethyl-4-[2-(1-pyrrolidinylethyl)]pyrrole-2-carbaldehyde, 3,5 -Dimethyl-4-(2-ethylaminoethyl)pyrrole-2-carbaldehyde, 4-(3-diethylaminopropyl)-3,5-dimethylpyrrole-2-carbaldehyde, 4-(4 -diethylaminobutyl)-3,5-dimethylpyrrole-2-carbaldehyde, 3,5-dimethyl-4-[3-(1-pyrrolidinylpropyl)]pyrrole-2-carbaldehyde.
实施例437:4-(2-氯乙酰基)-3,5-二甲基吡咯-2-甲醛Example 437: 4-(2-Chloroacetyl)-3,5-dimethylpyrrole-2-carbaldehyde
在室温和氮气氛下,用30分钟,向3,5-二甲基吡咯-2-甲醛(5g,40mmol)在1,2-二氯乙烷(50ml)中的溶液中分批加入无水氯化铝(42g,315mmol)。搅拌15分钟后,用1小时滴加氯乙酰氯(17g,150mmol)。添加完成后,在室温搅拌该混合物16小时。将该混合物倒入碎冰中,分离有机层,用无水硫酸镁干燥,并减压除去溶剂。产率57%,m.p.205-09℃(甲苯)。To a solution of 3,5-dimethylpyrrole-2-carbaldehyde (5 g, 40 mmol) in 1,2-dichloroethane (50 mL) was added portionwise anhydrous Aluminum chloride (42 g, 315 mmol). After stirring for 15 minutes, chloroacetyl chloride (17 g, 150 mmol) was added dropwise over 1 hour. After the addition was complete, the mixture was stirred at room temperature for 16 hours. The mixture was poured into crushed ice, the organic layer was separated, dried over anhydrous magnesium sulfate, and the solvent was removed under reduced pressure. Yield 57%, m.p. 205-09°C (toluene).
实施例438:3,5-二甲基-4-(2-二乙氨基乙酰基)吡咯-2-甲醛Example 438: 3,5-Dimethyl-4-(2-diethylaminoacetyl)pyrrole-2-carbaldehyde
向4-(2-氯乙酰基)-3,5-二甲基吡咯-2-甲醛(1g,5mmol)在THF(20ml)中的溶液中加入二乙胺(5ml)。在室温搅拌该混合物24小时。减压除去溶剂,残余物用水、NaHCO3(5%)处理,并用二氯甲烷萃取。干燥并过滤后,除去溶剂,残余物无需进一步纯化即可使用。To a solution of 4-(2-chloroacetyl)-3,5-dimethylpyrrole-2-carbaldehyde (1 g, 5 mmol) in THF (20 ml) was added diethylamine (5 ml). The mixture was stirred at room temperature for 24 hours. The solvent was removed under reduced pressure and the residue was treated with water, NaHCO3 (5%) and extracted with dichloromethane. After drying and filtration, the solvent was removed and the residue was used without further purification.
按照与实施例438所述类似的方法,合成3,5-二甲基-4-(2-二甲氨基乙酰基)吡咯-2-甲醛和3,5-二甲基-4-[2-(1-吡咯烷基)乙酰基]吡咯-2-甲醛。Following a method similar to that described in Example 438, 3,5-dimethyl-4-(2-dimethylaminoacetyl)pyrrole-2-carbaldehyde and 3,5-dimethyl-4-[2- (1-Pyrrolidinyl)acetyl]pyrrole-2-carbaldehyde.
实施例439:3,5-二甲基-4-乙氧基草酰基吡咯-2-甲醛Example 439: 3,5-Dimethyl-4-ethoxyoxalylpyrrole-2-carbaldehyde
在室温和氮气氛下,用30分钟,向3,5-二甲基吡咯-2-甲醛(7.6g,62mmol)在1,2-二氯乙烷(140ml)中的溶液中分批加入无水氯化铝(37g,277mmol)。搅拌30分钟后,加入硝基甲烷(11.2ml,179mmol)。该混合物在0℃冷却,并用1小时滴加氯乙醛酸乙酯(12.8g,94mmol)。添加完成后,在室温搅拌该混合物4小时。将该混合物倒入碎冰中,分离有机层,干燥并蒸发,得到白色固体。产率45%,m.p.125-127℃(甲苯)。To a solution of 3,5-dimethylpyrrole-2-carbaldehyde (7.6 g, 62 mmol) in 1,2-dichloroethane (140 ml) was added portion-wise at room temperature under a nitrogen atmosphere over 30 minutes without Aluminum chloride hydrate (37 g, 277 mmol). After stirring for 30 minutes, nitromethane (11.2ml, 179mmol) was added. The mixture was cooled at 0°C, and ethyl chloroglyoxylate (12.8 g, 94 mmol) was added dropwise over 1 hour. After the addition was complete, the mixture was stirred at room temperature for 4 hours. The mixture was poured into crushed ice and the organic layer was separated, dried and evaporated to give a white solid. Yield 45%, m.p. 125-127°C (toluene).
前面实施例中所有化合物的NMR谱数据均与相应的化学结构一致。The NMR spectrum data of all the compounds in the previous examples are consistent with the corresponding chemical structures.
尽管参照其优选的实施方案详细地描述了本发明,但是本领域专业人员应该理解,在不背离权利要求书所定义的本发明精神和范围的条件下,可以对其形式和细节进行许多改变。Although the present invention has been described in detail with reference to its preferred embodiments, it will be understood by those skilled in the art that many changes in form and details may be made therein without departing from the spirit and scope of the invention as defined by the claims.
Claims (34)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14656399P | 1999-07-30 | 1999-07-30 | |
| US60/146563 | 1999-07-30 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1377346A CN1377346A (en) | 2002-10-30 |
| CN1193026C true CN1193026C (en) | 2005-03-16 |
Family
ID=22517965
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB008136823A Expired - Fee Related CN1193026C (en) | 1999-07-30 | 2000-07-28 | Z-pyrazoline-5-ones |
Country Status (21)
| Country | Link |
|---|---|
| EP (1) | EP1218373A2 (en) |
| JP (1) | JP2003506368A (en) |
| KR (1) | KR20020091829A (en) |
| CN (1) | CN1193026C (en) |
| AR (1) | AR032279A1 (en) |
| AU (1) | AU6388900A (en) |
| BG (1) | BG106392A (en) |
| BR (1) | BR0012896A (en) |
| CA (1) | CA2380644A1 (en) |
| CZ (1) | CZ2002302A3 (en) |
| HK (1) | HK1049154A1 (en) |
| HU (1) | HUP0400540A3 (en) |
| IL (1) | IL147757A0 (en) |
| MX (1) | MXPA02001088A (en) |
| NO (1) | NO20020487L (en) |
| NZ (1) | NZ516850A (en) |
| PL (1) | PL354144A1 (en) |
| SK (1) | SK1512002A3 (en) |
| TR (1) | TR200200928T2 (en) |
| WO (1) | WO2001009121A2 (en) |
| ZA (1) | ZA200200477B (en) |
Families Citing this family (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6455525B1 (en) | 1999-11-04 | 2002-09-24 | Cephalon, Inc. | Heterocyclic substituted pyrazolones |
| WO2003011287A1 (en) * | 2001-08-03 | 2003-02-13 | Vertex Pharmaceuticals Incorporated | Pyrazolon derivatives as inhibitors of gsk-3 |
| AU2003249865A1 (en) * | 2003-06-24 | 2005-01-21 | Actelion Pharmaceuticals Ltd | Pyrazolidinedione derivatives and their use as platelet aggregation inhibitors |
| WO2005012255A1 (en) * | 2003-08-01 | 2005-02-10 | Mitsubishi Pharma Corporation | Remedy for inflammatory joint diseases |
| EP1681063A4 (en) * | 2003-10-14 | 2009-08-05 | Satoshi Takeo | AGENT FOR ATTENUATING MENTAL DISORDERS |
| PL1696920T3 (en) | 2003-12-19 | 2015-03-31 | Plexxikon Inc | Compounds and methods for development of ret modulators |
| GB0329617D0 (en) * | 2003-12-23 | 2004-01-28 | Astex Technology Ltd | Pharmaceutical compounds |
| US7498342B2 (en) | 2004-06-17 | 2009-03-03 | Plexxikon, Inc. | Compounds modulating c-kit activity |
| WO2006124874A2 (en) * | 2005-05-12 | 2006-11-23 | Kalypsys, Inc. | Inhibitors of b-raf kinase |
| WO2006129587A1 (en) * | 2005-05-30 | 2006-12-07 | Genecare Research Institute Co., Ltd. | Pharmaceutical composition comprising pyrazolone derivative |
| JPWO2006129583A1 (en) * | 2005-05-30 | 2009-01-08 | 株式会社ジーンケア研究所 | Pyrazolone derivatives |
| US7371862B2 (en) | 2005-11-11 | 2008-05-13 | Pfizer Italia S.R.L. | Azaindolylidene derivatives as kinase inhibitors, process for their preparation and pharmaceutical compositions comprising them |
| DE102005057924A1 (en) * | 2005-12-05 | 2007-06-06 | Merck Patent Gmbh | pyridazinone derivatives |
| JP4979710B2 (en) | 2005-12-09 | 2012-07-18 | エフ・ホフマン−ラ・ロシュ・アクチェンゲゼルシャフト | Antiviral nucleoside |
| ES2429290T3 (en) | 2006-10-10 | 2013-11-14 | Gilead Pharmasset Llc | Preparation of ribofuranosylpyrimidine nucleosides |
| WO2008063888A2 (en) | 2006-11-22 | 2008-05-29 | Plexxikon, Inc. | Compounds modulating c-fms and/or c-kit activity and uses therefor |
| US7964580B2 (en) | 2007-03-30 | 2011-06-21 | Pharmasset, Inc. | Nucleoside phosphoramidate prodrugs |
| AU2008276063B2 (en) | 2007-07-17 | 2013-11-28 | Plexxikon Inc. | Compounds and methods for kinase modulation, and indications therefor |
| KR100987557B1 (en) * | 2008-03-18 | 2010-10-12 | 이화여자대학교 산학협력단 | Compositions for treating or preventing vascular restenosis |
| AU2009318098B2 (en) * | 2008-11-20 | 2015-07-16 | Northwestern University | Treatment of amyotrophic lateral sclerosis |
| US9145424B2 (en) | 2008-11-20 | 2015-09-29 | Northwestern University | Treatment of amyotrophic lateral sclerosis |
| US8716263B2 (en) | 2008-12-23 | 2014-05-06 | Gilead Pharmasset Llc | Synthesis of purine nucleosides |
| BRPI1008709B8 (en) | 2009-04-03 | 2021-05-25 | Hoffmann La Roche | solid dispersion, formulation, composition and tablet comprising {3-[5-(4-chloro-phenyl)-1h-pyrrolo[2,3-b]pyridine-3-carbonyl]-2,4-difluoro-phenyl}-amide of propane-1-sulfonic acid |
| TWI576352B (en) | 2009-05-20 | 2017-04-01 | 基利法瑪席特有限責任公司 | Nucleoside phosphoramidates |
| US8618076B2 (en) | 2009-05-20 | 2013-12-31 | Gilead Pharmasset Llc | Nucleoside phosphoramidates |
| US20110112127A1 (en) | 2009-11-06 | 2011-05-12 | Plexxikon, Inc. | Compounds and methods for kinase modulation, and indications therefor |
| MX2012011171A (en) | 2010-03-31 | 2013-02-01 | Gilead Pharmasset Llc | Nucleoside phosphoramidates. |
| JP6069215B2 (en) | 2010-11-30 | 2017-02-01 | ギリアド ファーマセット エルエルシー | Compound |
| MY162950A (en) | 2011-02-07 | 2017-07-31 | Plexxikon Inc | Compounds and methods for kinase modulation, and indications therefor |
| AR085279A1 (en) | 2011-02-21 | 2013-09-18 | Plexxikon Inc | SOLID FORMS OF {3- [5- (4-CHLORINE-PHENYL) -1H-PIRROLO [2,3-B] PIRIDINA-3-CARBONIL] -2,4-DIFLUOR-PHENIL} -AMIDE OF PROPANE ACID-1- SULFONIC |
| US8889159B2 (en) | 2011-11-29 | 2014-11-18 | Gilead Pharmasset Llc | Compositions and methods for treating hepatitis C virus |
| US9150570B2 (en) | 2012-05-31 | 2015-10-06 | Plexxikon Inc. | Synthesis of heterocyclic compounds |
| EP2876109A4 (en) * | 2012-07-23 | 2016-03-16 | Yuhan Corp | FUSHANE-CONTAINING FURANE-CONTAINING COMPOUND OR SALT THEREOF AND PHARMACEUTICAL COMPOSITION COMPRISING THE SAME |
| KR20160115996A (en) | 2014-02-14 | 2016-10-06 | 인셉션 2 인코퍼레이티드 | Pyrazolone compounds and uses thereof |
| CN105037269B (en) * | 2015-07-10 | 2018-01-19 | 中国人民解放军第二军医大学 | Substituted pyrazolecarboxylic ketone secretory protease inhibitors and preparation method thereof |
| ES2966825T3 (en) | 2017-08-15 | 2024-04-24 | Agios Pharmaceuticals Inc | Pyruvate kinase activators for use in the treatment of blood disorders |
| KR102625224B1 (en) * | 2018-10-31 | 2024-01-15 | 주식회사 큐로젠 | Composition for preventing, improving or treating autoimmune diseases comprising pyrazol-one derivatives |
| CN111793032B (en) * | 2019-04-08 | 2021-11-19 | 四川省中医药科学院 | Pyrazolone compounds and preparation method and application thereof |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4369310A (en) * | 1979-11-19 | 1983-01-18 | Ciba-Geigy Ltd. | Bleachable dyes |
| JPS6229570A (en) * | 1985-07-29 | 1987-02-07 | Kanegafuchi Chem Ind Co Ltd | 3,5-diisopropylbenzylidene heterocyclic compound |
| JPH078851B2 (en) * | 1985-07-29 | 1995-02-01 | 鐘淵化学工業株式会社 | 3-phenylthiomethylstyrene derivative |
| JP2678822B2 (en) * | 1990-06-04 | 1997-11-19 | 富士写真フイルム株式会社 | Silver halide photographic material |
-
2000
- 2000-07-28 CN CNB008136823A patent/CN1193026C/en not_active Expired - Fee Related
- 2000-07-28 IL IL14775700A patent/IL147757A0/en unknown
- 2000-07-28 WO PCT/US2000/020628 patent/WO2001009121A2/en not_active Ceased
- 2000-07-28 KR KR1020027001343A patent/KR20020091829A/en not_active Withdrawn
- 2000-07-28 MX MXPA02001088A patent/MXPA02001088A/en unknown
- 2000-07-28 AU AU63889/00A patent/AU6388900A/en not_active Abandoned
- 2000-07-28 CZ CZ2002302A patent/CZ2002302A3/en unknown
- 2000-07-28 CA CA002380644A patent/CA2380644A1/en not_active Abandoned
- 2000-07-28 NZ NZ516850A patent/NZ516850A/en unknown
- 2000-07-28 BR BR0012896-1A patent/BR0012896A/en not_active IP Right Cessation
- 2000-07-28 SK SK151-2002A patent/SK1512002A3/en unknown
- 2000-07-28 HK HK02109301.3A patent/HK1049154A1/en unknown
- 2000-07-28 JP JP2001514324A patent/JP2003506368A/en not_active Withdrawn
- 2000-07-28 PL PL00354144A patent/PL354144A1/en not_active Application Discontinuation
- 2000-07-28 HU HU0400540A patent/HUP0400540A3/en unknown
- 2000-07-28 TR TR2002/00928T patent/TR200200928T2/en unknown
- 2000-07-28 EP EP00950852A patent/EP1218373A2/en not_active Withdrawn
- 2000-07-31 AR ARP000103955A patent/AR032279A1/en unknown
-
2002
- 2002-01-18 ZA ZA200200477A patent/ZA200200477B/en unknown
- 2002-01-30 NO NO20020487A patent/NO20020487L/en not_active Application Discontinuation
- 2002-02-06 BG BG106392A patent/BG106392A/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| PL354144A1 (en) | 2003-12-29 |
| CN1377346A (en) | 2002-10-30 |
| NO20020487D0 (en) | 2002-01-30 |
| AU6388900A (en) | 2001-02-19 |
| CZ2002302A3 (en) | 2002-06-12 |
| WO2001009121A3 (en) | 2002-05-02 |
| KR20020091829A (en) | 2002-12-06 |
| CA2380644A1 (en) | 2001-02-08 |
| NZ516850A (en) | 2004-09-24 |
| BG106392A (en) | 2002-12-29 |
| WO2001009121A2 (en) | 2001-02-08 |
| ZA200200477B (en) | 2003-06-25 |
| BR0012896A (en) | 2002-06-18 |
| JP2003506368A (en) | 2003-02-18 |
| HUP0400540A2 (en) | 2004-06-28 |
| EP1218373A2 (en) | 2002-07-03 |
| TR200200928T2 (en) | 2002-09-23 |
| IL147757A0 (en) | 2002-08-14 |
| NO20020487L (en) | 2002-03-12 |
| MXPA02001088A (en) | 2003-09-22 |
| HK1049154A1 (en) | 2003-05-02 |
| AR032279A1 (en) | 2003-11-05 |
| SK1512002A3 (en) | 2002-11-06 |
| HUP0400540A3 (en) | 2004-09-28 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1193026C (en) | Z-pyrazoline-5-ones | |
| CN1636005A (en) | Benzothiazinone and benzoxazinone compounds | |
| HK1042895A1 (en) | Tricyclic pyrazole derivatives | |
| CN1390220A (en) | Kinase inhibitors as therapeutic agents | |
| CN1137100C (en) | Tricyclic compounds capable of inhibiting tyrosine kinases of the epidermal growth factor receptor family | |
| JP5290972B2 (en) | 2- (Heterocyclylbenzyl) pyridazinone derivatives | |
| CN1319966C (en) | Novel benzoimidazole derivatives useful as antiproliferative agents | |
| CN1196703C (en) | Pyrazolobenzodiazepines as CDK2 inhibitors | |
| JP5210172B2 (en) | Pyridiazinone derivatives for tumor therapy | |
| CN1422262A (en) | 2-benzothiazolyl urea derivatives and their use as protein kinase inhibitors | |
| HK1045687A1 (en) | 4,5,6,7-tetrahydroindazole derivatives as antitumor agents | |
| CN1326457A (en) | 4-aminopyrrolopyrimidines as kinase inhibitors | |
| CN1816529A (en) | Pyrazole-amide compounds useful as kinase inhibitors | |
| CN1520298A (en) | Pyrazolopyrimidines as therapeutic agents | |
| CN1849325A (en) | Thiazolo-, oxazalo and imidazolo-quinazoline compounds capable of inhibiting prot ein kinases | |
| CN1976907A (en) | Heterocyclic amide compound and use thereof as an MMP-13 inhibitor | |
| CN1714092A (en) | Thieno[3,2-b]pyridine-6-carbonitrile and Thieno[2,3-b]pyridine-5-carbonitrile as protein kinase inhibitors | |
| CN1349990A (en) | Heterocyclic compound, its preparation and use | |
| CN1351497A (en) | Aminopyrimidines as sorbitol dehydrogenase inhibitors | |
| CN101039914A (en) | Trifluoromethyl-substituted benzamide compounds as kinase inhibitors | |
| CN101031560A (en) | Cyclic diaryl ureas suitable as tyrosine kinase inhibitors | |
| CN1902186A (en) | Thiazole and pyrazole derivatives as FLT-3 kinase inhibitors | |
| CN1882571A (en) | Pyrrolderivate | |
| CN1368970A (en) | Substituted 1,4-dihydroindeno [1,2-C] pyrazoles as inhibitors of tyrosine kinase | |
| JP5174665B2 (en) | 1-acyl dihydropyrazole derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C06 | Publication | ||
| PB01 | Publication | ||
| ASS | Succession or assignment of patent right |
Owner name: ABBOTT SHARES LIMITED KG CO., LTD. Free format text: FORMER OWNER: BASF COMPANY Effective date: 20021108 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20021108 Applicant after: Abert GmbH & Co. KG Applicant before: BASF AG |
|
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C19 | Lapse of patent right due to non-payment of the annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |