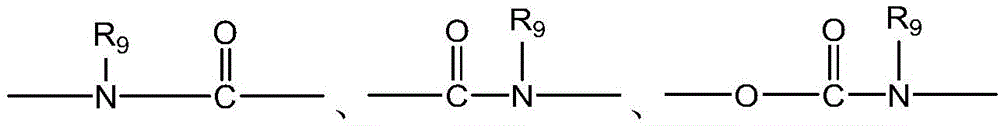
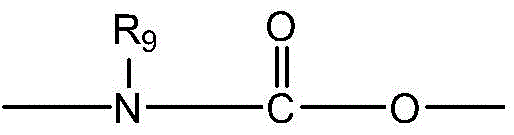
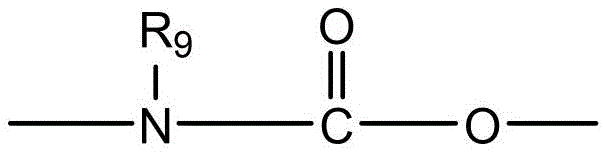
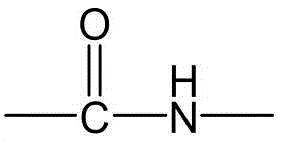
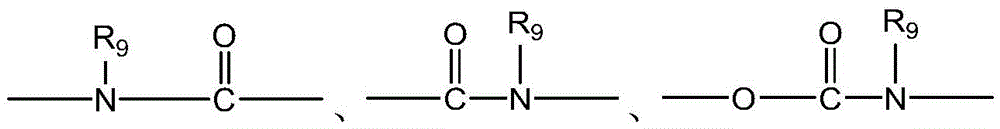
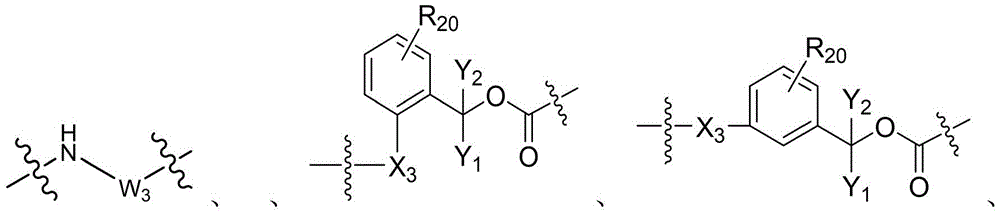
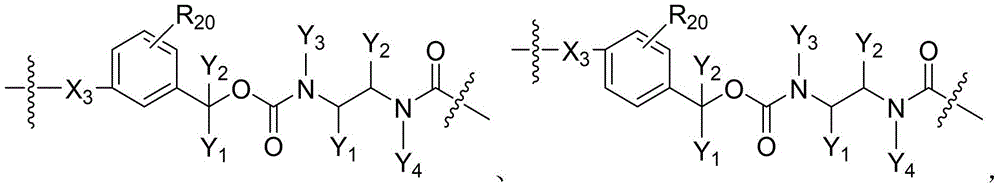
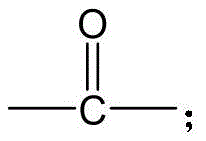
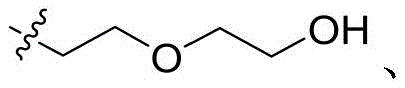
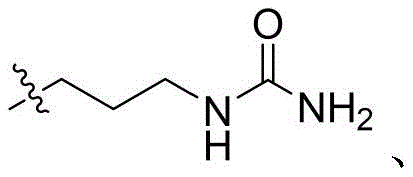
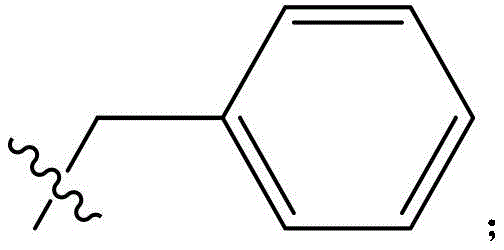
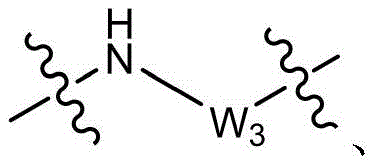
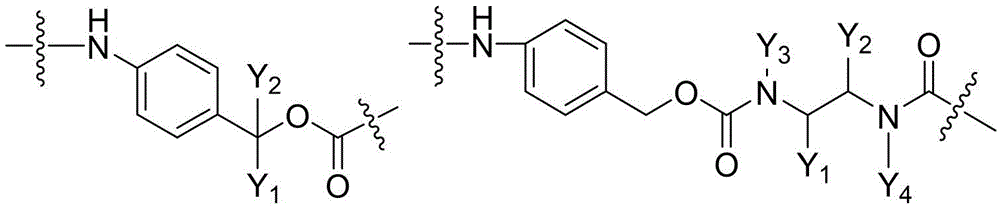
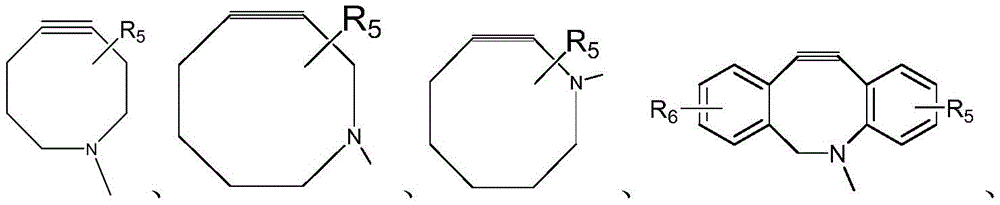
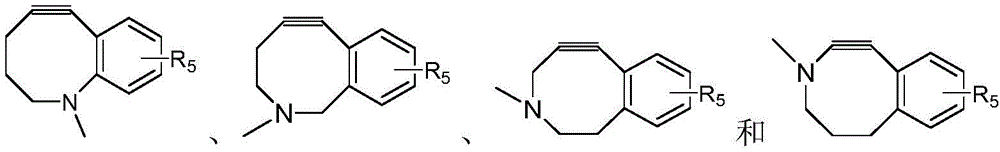
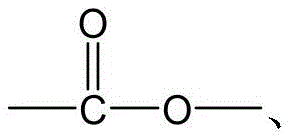
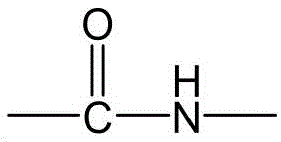
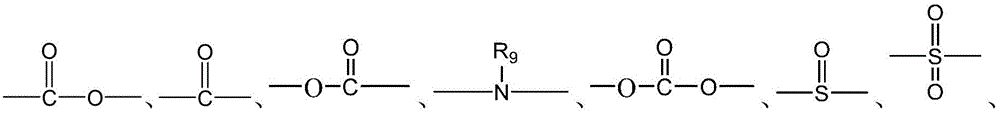
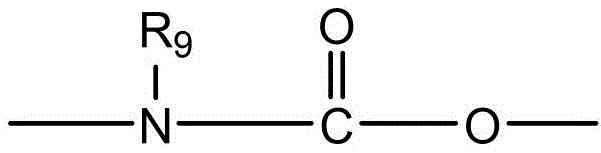
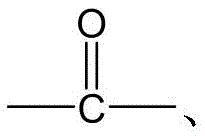
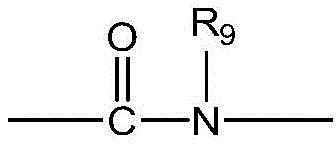
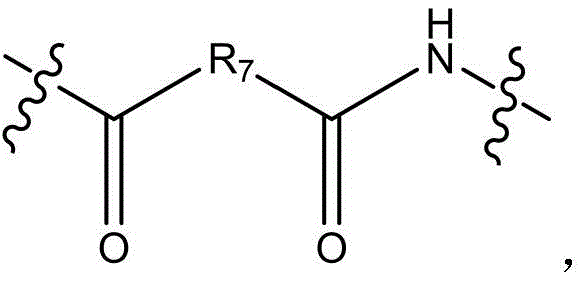
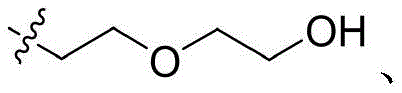
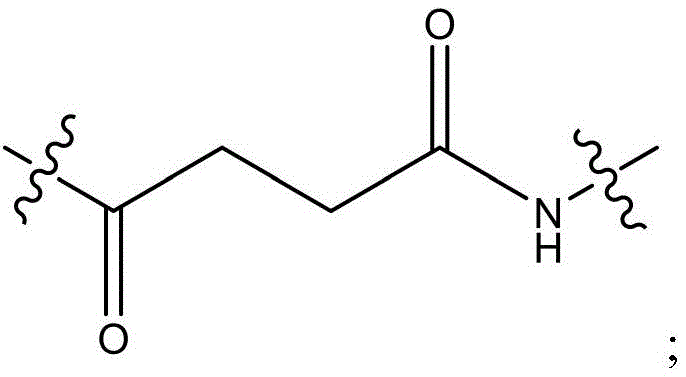
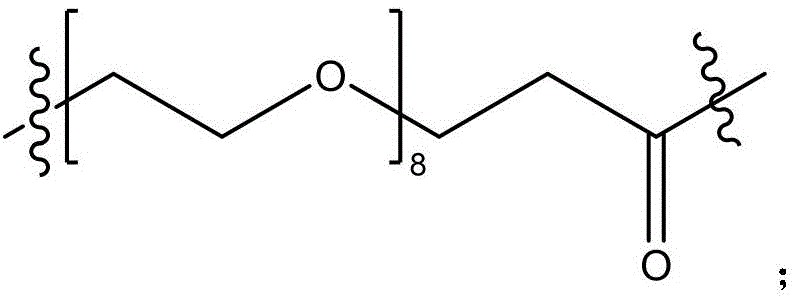
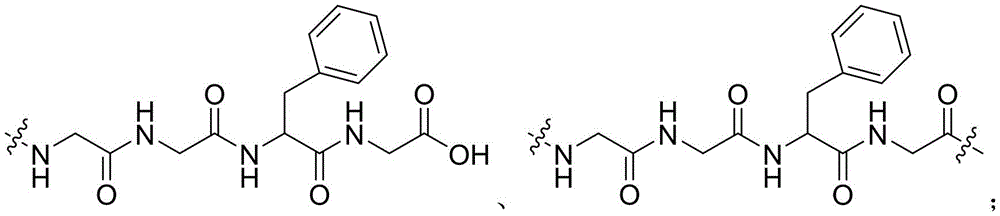
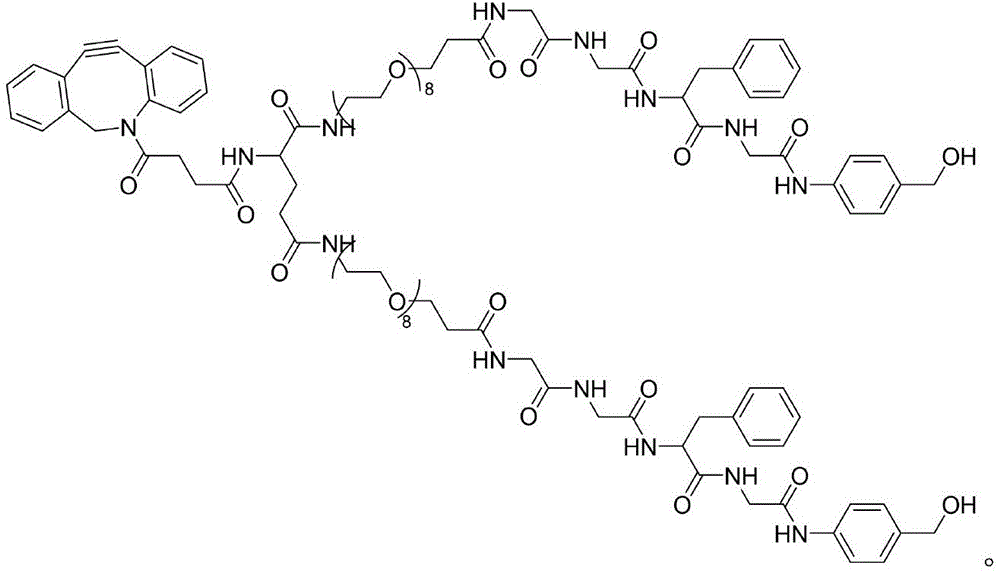
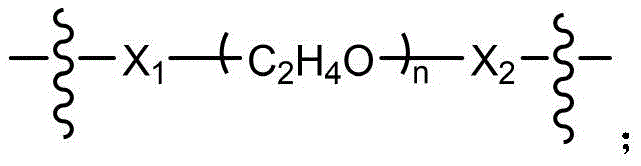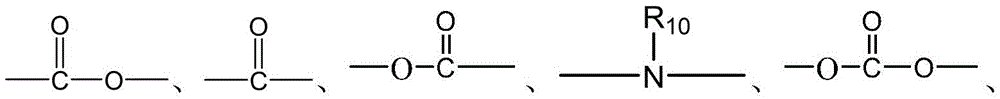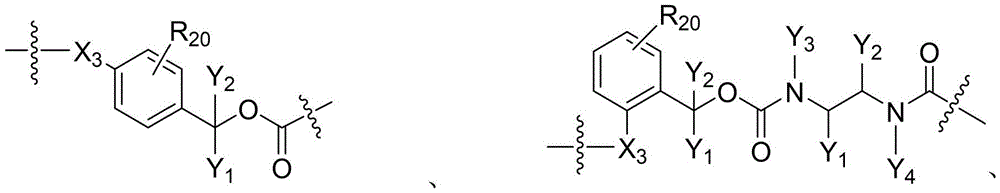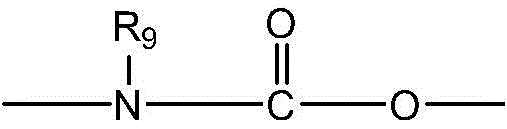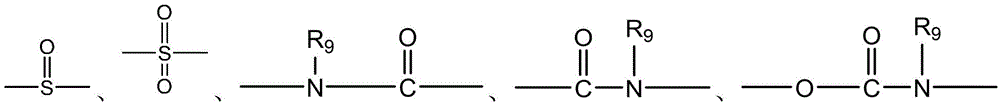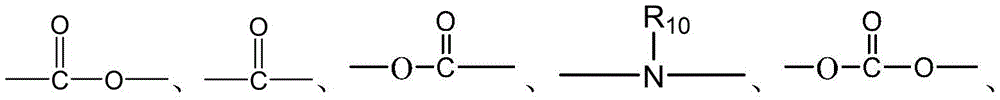CN115505028A - Branched polyethylene glycol linker and application thereof - Google Patents
Branched polyethylene glycol linker and application thereof Download PDFInfo
- Publication number
- CN115505028A CN115505028A CN202211129102.4A CN202211129102A CN115505028A CN 115505028 A CN115505028 A CN 115505028A CN 202211129102 A CN202211129102 A CN 202211129102A CN 115505028 A CN115505028 A CN 115505028A
- Authority
- CN
- China
- Prior art keywords
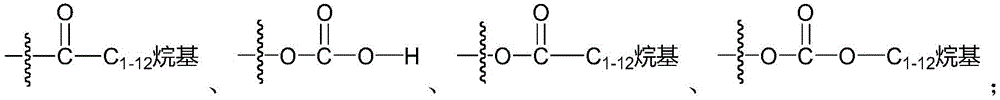
- alkyl
- alkylene
- branched
- straight chain
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
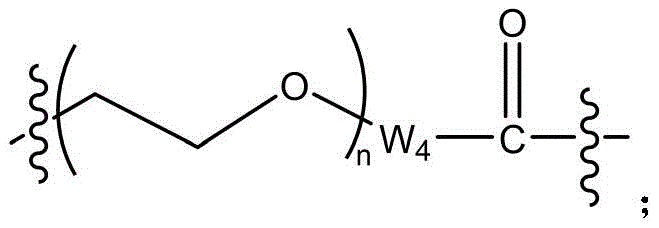
- 229920001223 polyethylene glycol Polymers 0.000 title claims abstract description 21
- 239000002202 Polyethylene glycol Substances 0.000 title claims abstract description 16
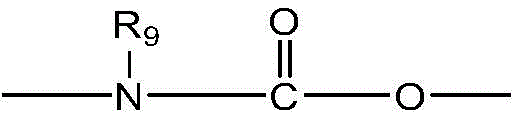
- 239000003814 drug Substances 0.000 claims abstract description 81
- 229940079593 drug Drugs 0.000 claims abstract description 80
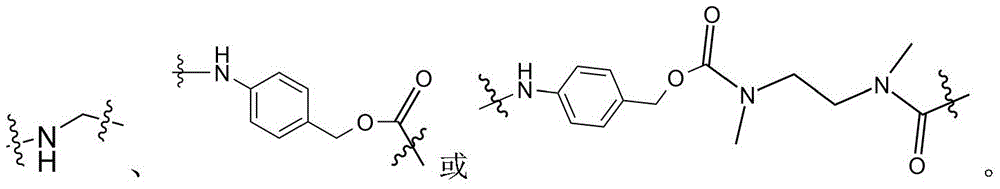
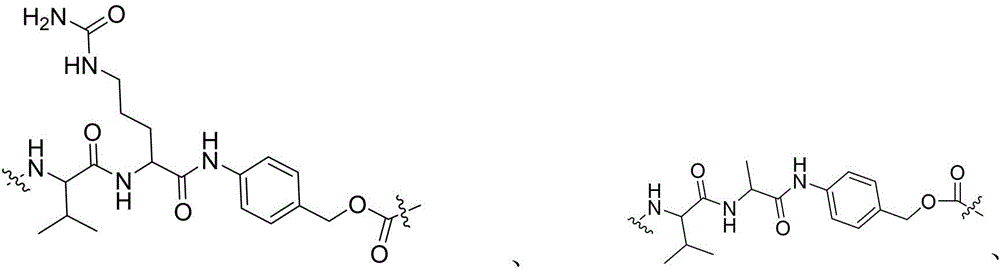
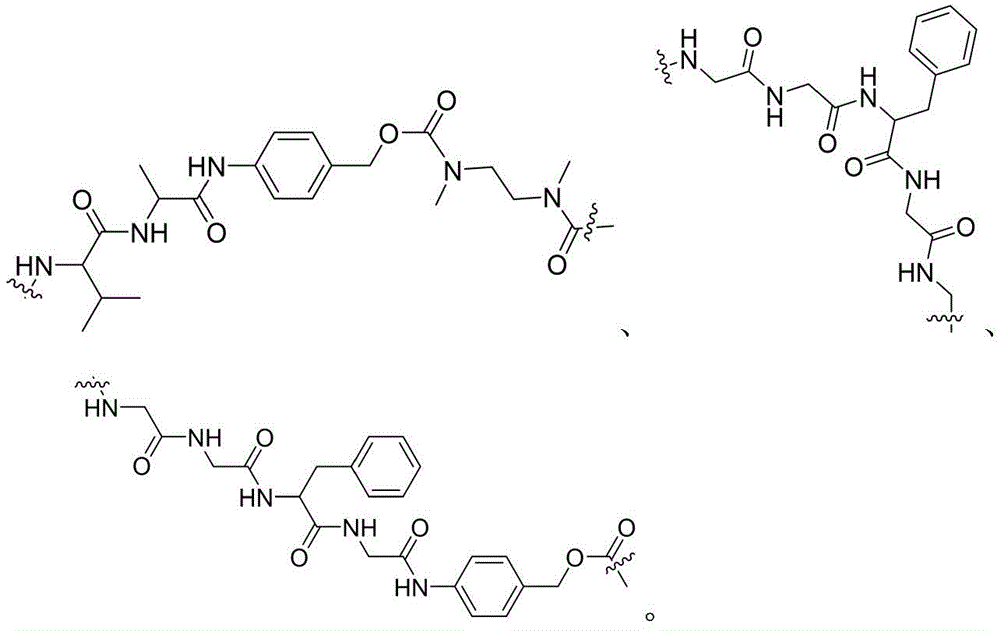
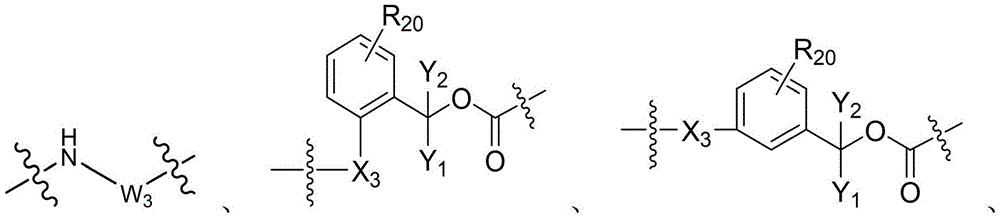
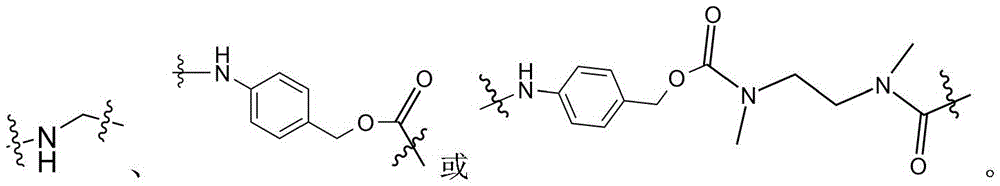
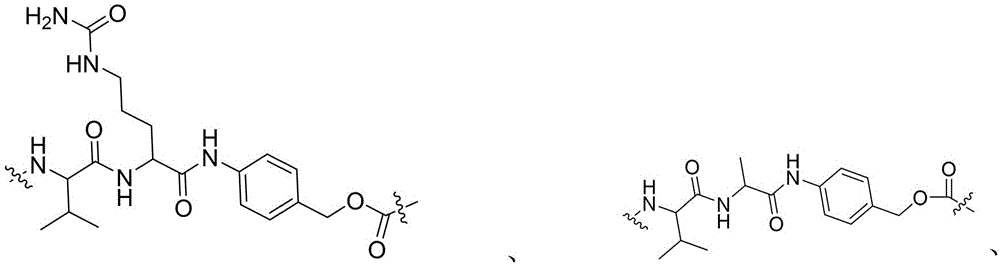
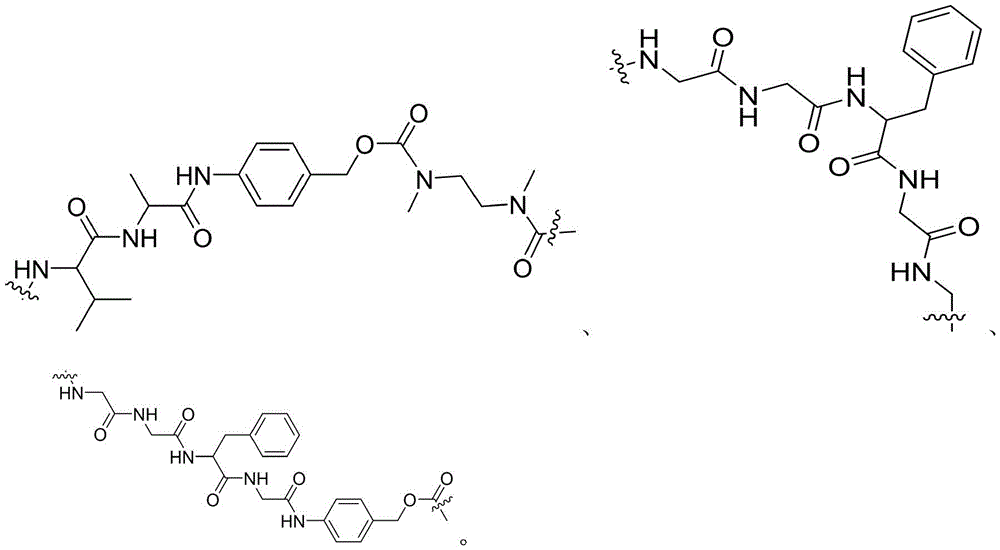
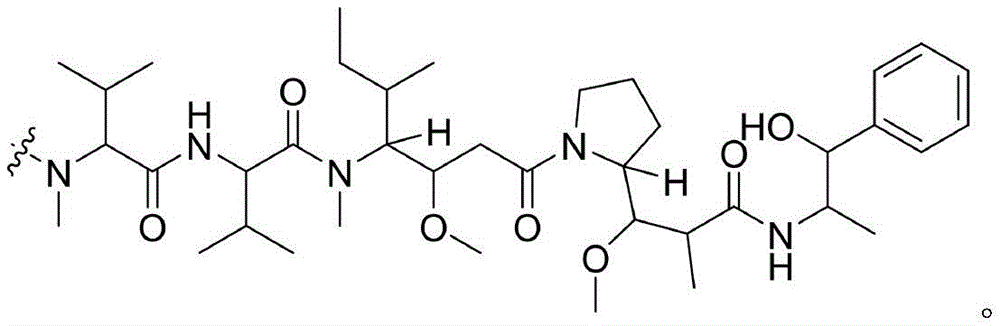
- -1 glycine-glycine-benzene Chemical compound 0.000 claims description 122
- 125000000217 alkyl group Chemical group 0.000 claims description 65
- 125000005647 linker group Chemical group 0.000 claims description 52
- 125000002947 alkylene group Chemical group 0.000 claims description 38
- 229910052739 hydrogen Inorganic materials 0.000 claims description 28
- 229910052799 carbon Inorganic materials 0.000 claims description 25
- 201000010099 disease Diseases 0.000 claims description 25
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 25
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 25
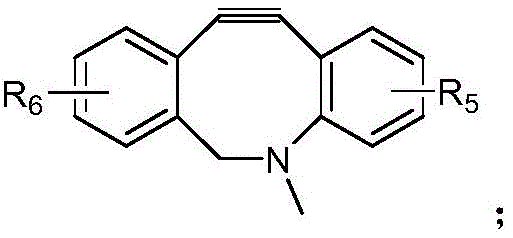
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 claims description 24
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 claims description 18
- DHMQDGOQFOQNFH-UHFFFAOYSA-N Glycine Chemical compound NCC(O)=O DHMQDGOQFOQNFH-UHFFFAOYSA-N 0.000 claims description 16
- 102000004196 processed proteins & peptides Human genes 0.000 claims description 16
- 125000006376 (C3-C10) cycloalkyl group Chemical group 0.000 claims description 15
- 229960004679 doxorubicin Drugs 0.000 claims description 15
- 229920001184 polypeptide Polymers 0.000 claims description 15
- 238000002360 preparation method Methods 0.000 claims description 13
- 125000006652 (C3-C12) cycloalkyl group Chemical group 0.000 claims description 12
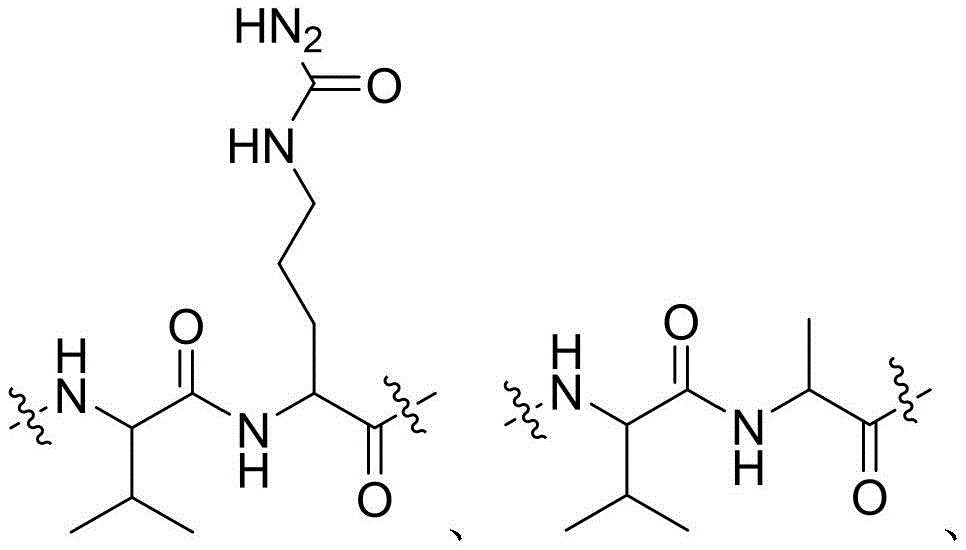
- 229960003767 alanine Drugs 0.000 claims description 12
- 229940024606 amino acid Drugs 0.000 claims description 12
- 229960002173 citrulline Drugs 0.000 claims description 12
- 125000002993 cycloalkylene group Chemical group 0.000 claims description 12
- 235000001014 amino acid Nutrition 0.000 claims description 11
- 150000001413 amino acids Chemical class 0.000 claims description 11
- 125000003118 aryl group Chemical group 0.000 claims description 10
- 229910052736 halogen Inorganic materials 0.000 claims description 10
- 150000002367 halogens Chemical class 0.000 claims description 10
- 108010006654 Bleomycin Proteins 0.000 claims description 9
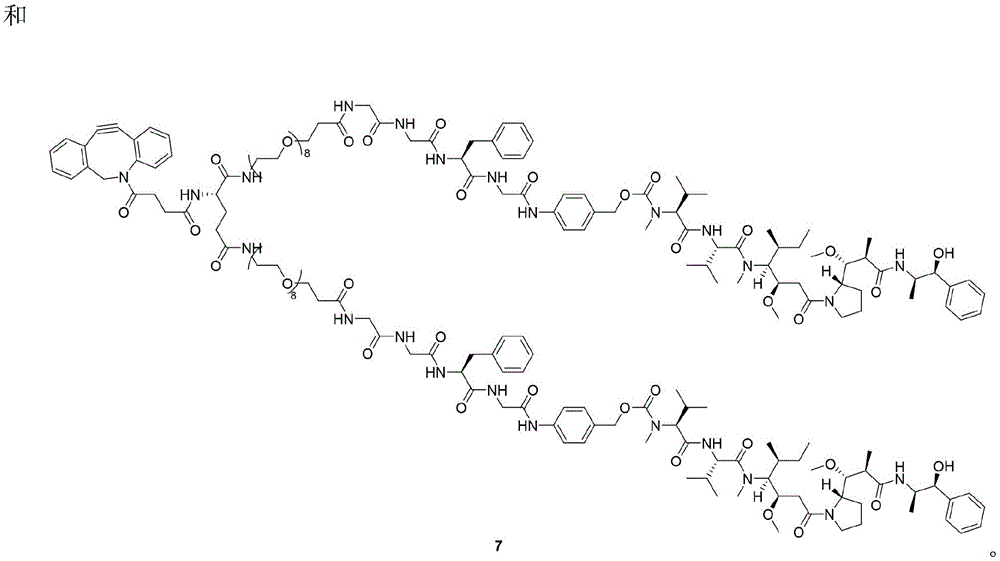
- IEDXPSOJFSVCKU-HOKPPMCLSA-N [4-[[(2S)-5-(carbamoylamino)-2-[[(2S)-2-[6-(2,5-dioxopyrrolidin-1-yl)hexanoylamino]-3-methylbutanoyl]amino]pentanoyl]amino]phenyl]methyl N-[(2S)-1-[[(2S)-1-[[(3R,4S,5S)-1-[(2S)-2-[(1R,2R)-3-[[(1S,2R)-1-hydroxy-1-phenylpropan-2-yl]amino]-1-methoxy-2-methyl-3-oxopropyl]pyrrolidin-1-yl]-3-methoxy-5-methyl-1-oxoheptan-4-yl]-methylamino]-3-methyl-1-oxobutan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]-N-methylcarbamate Chemical compound CC[C@H](C)[C@@H]([C@@H](CC(=O)N1CCC[C@H]1[C@H](OC)[C@@H](C)C(=O)N[C@H](C)[C@@H](O)c1ccccc1)OC)N(C)C(=O)[C@@H](NC(=O)[C@H](C(C)C)N(C)C(=O)OCc1ccc(NC(=O)[C@H](CCCNC(N)=O)NC(=O)[C@@H](NC(=O)CCCCCN2C(=O)CCC2=O)C(C)C)cc1)C(C)C IEDXPSOJFSVCKU-HOKPPMCLSA-N 0.000 claims description 9
- 239000004475 Arginine Substances 0.000 claims description 8
- 239000004471 Glycine Substances 0.000 claims description 8
- XUJNEKJLAYXESH-REOHCLBHSA-N L-Cysteine Chemical compound SC[C@H](N)C(O)=O XUJNEKJLAYXESH-REOHCLBHSA-N 0.000 claims description 8
- QNAYBMKLOCPYGJ-REOHCLBHSA-N L-alanine Chemical compound C[C@H](N)C(O)=O QNAYBMKLOCPYGJ-REOHCLBHSA-N 0.000 claims description 8
- ODKSFYDXXFIFQN-BYPYZUCNSA-P L-argininium(2+) Chemical compound NC(=[NH2+])NCCC[C@H]([NH3+])C(O)=O ODKSFYDXXFIFQN-BYPYZUCNSA-P 0.000 claims description 8
- RHGKLRLOHDJJDR-BYPYZUCNSA-N L-citrulline Chemical compound NC(=O)NCCC[C@H]([NH3+])C([O-])=O RHGKLRLOHDJJDR-BYPYZUCNSA-N 0.000 claims description 8
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 claims description 8
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 claims description 8
- COLNVLDHVKWLRT-QMMMGPOBSA-N L-phenylalanine Chemical compound OC(=O)[C@@H](N)CC1=CC=CC=C1 COLNVLDHVKWLRT-QMMMGPOBSA-N 0.000 claims description 8
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 claims description 8
- KZSNJWFQEVHDMF-BYPYZUCNSA-N L-valine Chemical compound CC(C)[C@H](N)C(O)=O KZSNJWFQEVHDMF-BYPYZUCNSA-N 0.000 claims description 8
- ROHFNLRQFUQHCH-UHFFFAOYSA-N Leucine Natural products CC(C)CC(N)C(O)=O ROHFNLRQFUQHCH-UHFFFAOYSA-N 0.000 claims description 8
- RHGKLRLOHDJJDR-UHFFFAOYSA-N Ndelta-carbamoyl-DL-ornithine Natural products OC(=O)C(N)CCCNC(N)=O RHGKLRLOHDJJDR-UHFFFAOYSA-N 0.000 claims description 8
- KZSNJWFQEVHDMF-UHFFFAOYSA-N Valine Natural products CC(C)C(N)C(O)=O KZSNJWFQEVHDMF-UHFFFAOYSA-N 0.000 claims description 8
- 235000004279 alanine Nutrition 0.000 claims description 8
- 125000000539 amino acid group Chemical group 0.000 claims description 8
- 229960003121 arginine Drugs 0.000 claims description 8
- ODKSFYDXXFIFQN-UHFFFAOYSA-N arginine Natural products OC(=O)C(N)CCCNC(N)=N ODKSFYDXXFIFQN-UHFFFAOYSA-N 0.000 claims description 8
- 235000009697 arginine Nutrition 0.000 claims description 8
- OYVAGSVQBOHSSS-UAPAGMARSA-O bleomycin A2 Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCC[S+](C)C)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1N=CNC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C OYVAGSVQBOHSSS-UAPAGMARSA-O 0.000 claims description 8
- 235000013477 citrulline Nutrition 0.000 claims description 8
- 229960002433 cysteine Drugs 0.000 claims description 8
- 235000018417 cysteine Nutrition 0.000 claims description 8
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 claims description 8
- 229960002449 glycine Drugs 0.000 claims description 8
- 229960000310 isoleucine Drugs 0.000 claims description 8
- AGPKZVBTJJNPAG-UHFFFAOYSA-N isoleucine Natural products CCC(C)C(N)C(O)=O AGPKZVBTJJNPAG-UHFFFAOYSA-N 0.000 claims description 8
- 229960003136 leucine Drugs 0.000 claims description 8
- 229960005190 phenylalanine Drugs 0.000 claims description 8
- COLNVLDHVKWLRT-UHFFFAOYSA-N phenylalanine Natural products OC(=O)C(N)CC1=CC=CC=C1 COLNVLDHVKWLRT-UHFFFAOYSA-N 0.000 claims description 8
- 229960004441 tyrosine Drugs 0.000 claims description 8
- OUYCCCASQSFEME-UHFFFAOYSA-N tyrosine Natural products OC(=O)C(N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-UHFFFAOYSA-N 0.000 claims description 8
- 229960004295 valine Drugs 0.000 claims description 8
- 239000004474 valine Substances 0.000 claims description 8
- 229960001561 bleomycin Drugs 0.000 claims description 7
- AOJJSUZBOXZQNB-VTZDEGQISA-N 4'-epidoxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-VTZDEGQISA-N 0.000 claims description 6
- 108010092160 Dactinomycin Proteins 0.000 claims description 6
- 239000002253 acid Substances 0.000 claims description 6
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 claims description 5
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 5
- WHUUTDBJXJRKMK-UHFFFAOYSA-N Glutamic acid Natural products OC(=O)C(N)CCC(O)=O WHUUTDBJXJRKMK-UHFFFAOYSA-N 0.000 claims description 5
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 claims description 5
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 claims description 5
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 claims description 5
- 239000004472 Lysine Substances 0.000 claims description 5
- 229930126263 Maytansine Natural products 0.000 claims description 5
- 229930012538 Paclitaxel Natural products 0.000 claims description 5
- 125000003545 alkoxy group Chemical group 0.000 claims description 5
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical compound C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 claims description 5
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 claims description 5
- 229960005420 etoposide Drugs 0.000 claims description 5
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 claims description 5
- 229960002989 glutamic acid Drugs 0.000 claims description 5
- 235000013922 glutamic acid Nutrition 0.000 claims description 5
- 239000004220 glutamic acid Substances 0.000 claims description 5
- 229960003646 lysine Drugs 0.000 claims description 5
- WKPWGQKGSOKKOO-RSFHAFMBSA-N maytansine Chemical compound CO[C@@H]([C@@]1(O)C[C@](OC(=O)N1)([C@H]([C@@H]1O[C@@]1(C)[C@@H](OC(=O)[C@H](C)N(C)C(C)=O)CC(=O)N1C)C)[H])\C=C\C=C(C)\CC2=CC(OC)=C(Cl)C1=C2 WKPWGQKGSOKKOO-RSFHAFMBSA-N 0.000 claims description 5
- 229930182817 methionine Natural products 0.000 claims description 5
- 229960004452 methionine Drugs 0.000 claims description 5
- 229960001592 paclitaxel Drugs 0.000 claims description 5
- YUOCYTRGANSSRY-UHFFFAOYSA-N pyrrolo[2,3-i][1,2]benzodiazepine Chemical class C1=CN=NC2=C3C=CN=C3C=CC2=C1 YUOCYTRGANSSRY-UHFFFAOYSA-N 0.000 claims description 5
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 claims description 5
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical compound OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 claims description 4
- DCXYFEDJOCDNAF-UHFFFAOYSA-N Asparagine Natural products OC(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-N 0.000 claims description 4
- UHDGCWIWMRVCDJ-CCXZUQQUSA-N Cytarabine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@@H](O)[C@H](O)[C@@H](CO)O1 UHDGCWIWMRVCDJ-CCXZUQQUSA-N 0.000 claims description 4
- HTIJFSOGRVMCQR-UHFFFAOYSA-N Epirubicin Natural products COc1cccc2C(=O)c3c(O)c4CC(O)(CC(OC5CC(N)C(=O)C(C)O5)c4c(O)c3C(=O)c12)C(=O)CO HTIJFSOGRVMCQR-UHFFFAOYSA-N 0.000 claims description 4
- AHLPHDHHMVZTML-BYPYZUCNSA-N L-Ornithine Chemical compound NCCC[C@H](N)C(O)=O AHLPHDHHMVZTML-BYPYZUCNSA-N 0.000 claims description 4
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 claims description 4
- DCXYFEDJOCDNAF-REOHCLBHSA-N L-asparagine Chemical compound OC(=O)[C@@H](N)CC(N)=O DCXYFEDJOCDNAF-REOHCLBHSA-N 0.000 claims description 4
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 claims description 4
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 claims description 4
- ZDXPYRJPNDTMRX-VKHMYHEASA-N L-glutamine Chemical compound OC(=O)[C@@H](N)CCC(N)=O ZDXPYRJPNDTMRX-VKHMYHEASA-N 0.000 claims description 4
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 claims description 4
- FBOZXECLQNJBKD-ZDUSSCGKSA-N L-methotrexate Chemical compound C=1N=C2N=C(N)N=C(N)C2=NC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 FBOZXECLQNJBKD-ZDUSSCGKSA-N 0.000 claims description 4
- AYFVYJQAPQTCCC-GBXIJSLDSA-N L-threonine Chemical compound C[C@@H](O)[C@H](N)C(O)=O AYFVYJQAPQTCCC-GBXIJSLDSA-N 0.000 claims description 4
- QIVBCDIJIAJPQS-VIFPVBQESA-N L-tryptophane Chemical compound C1=CC=C2C(C[C@H](N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-VIFPVBQESA-N 0.000 claims description 4
- AHLPHDHHMVZTML-UHFFFAOYSA-N Orn-delta-NH2 Natural products NCCCC(N)C(O)=O AHLPHDHHMVZTML-UHFFFAOYSA-N 0.000 claims description 4
- UTJLXEIPEHZYQJ-UHFFFAOYSA-N Ornithine Natural products OC(=O)C(C)CCCN UTJLXEIPEHZYQJ-UHFFFAOYSA-N 0.000 claims description 4
- ONIBWKKTOPOVIA-UHFFFAOYSA-N Proline Natural products OC(=O)C1CCCN1 ONIBWKKTOPOVIA-UHFFFAOYSA-N 0.000 claims description 4
- MTCFGRXMJLQNBG-UHFFFAOYSA-N Serine Natural products OCC(N)C(O)=O MTCFGRXMJLQNBG-UHFFFAOYSA-N 0.000 claims description 4
- AYFVYJQAPQTCCC-UHFFFAOYSA-N Threonine Natural products CC(O)C(N)C(O)=O AYFVYJQAPQTCCC-UHFFFAOYSA-N 0.000 claims description 4
- 239000004473 Threonine Substances 0.000 claims description 4
- QIVBCDIJIAJPQS-UHFFFAOYSA-N Tryptophan Natural products C1=CC=C2C(CC(N)C(O)=O)=CNC2=C1 QIVBCDIJIAJPQS-UHFFFAOYSA-N 0.000 claims description 4
- 229930183665 actinomycin Natural products 0.000 claims description 4
- 125000003710 aryl alkyl group Chemical group 0.000 claims description 4
- 229960001230 asparagine Drugs 0.000 claims description 4
- 235000009582 asparagine Nutrition 0.000 claims description 4
- 229960005261 aspartic acid Drugs 0.000 claims description 4
- 235000003704 aspartic acid Nutrition 0.000 claims description 4
- OQFSQFPPLPISGP-UHFFFAOYSA-N beta-carboxyaspartic acid Natural products OC(=O)C(N)C(C(O)=O)C(O)=O OQFSQFPPLPISGP-UHFFFAOYSA-N 0.000 claims description 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 4
- 229960003067 cystine Drugs 0.000 claims description 4
- VSJKWCGYPAHWDS-UHFFFAOYSA-N dl-camptothecin Natural products C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-UHFFFAOYSA-N 0.000 claims description 4
- AMRJKAQTDDKMCE-UHFFFAOYSA-N dolastatin Chemical compound CC(C)C(N(C)C)C(=O)NC(C(C)C)C(=O)N(C)C(C(C)C)C(OC)CC(=O)N1CCCC1C(OC)C(C)C(=O)NC(C=1SC=CN=1)CC1=CC=CC=C1 AMRJKAQTDDKMCE-UHFFFAOYSA-N 0.000 claims description 4
- 229930188854 dolastatin Natural products 0.000 claims description 4
- 229960001904 epirubicin Drugs 0.000 claims description 4
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 4
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 claims description 4
- 235000004554 glutamine Nutrition 0.000 claims description 4
- 229960002743 glutamine Drugs 0.000 claims description 4
- 229960002885 histidine Drugs 0.000 claims description 4
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 claims description 4
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 4
- 229960000485 methotrexate Drugs 0.000 claims description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 4
- 229960003104 ornithine Drugs 0.000 claims description 4
- 229960002429 proline Drugs 0.000 claims description 4
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 4
- 229960001153 serine Drugs 0.000 claims description 4
- 229960002898 threonine Drugs 0.000 claims description 4
- 229960004799 tryptophan Drugs 0.000 claims description 4
- MFRNYXJJRJQHNW-DEMKXPNLSA-N (2s)-2-[[(2r,3r)-3-methoxy-3-[(2s)-1-[(3r,4s,5s)-3-methoxy-5-methyl-4-[methyl-[(2s)-3-methyl-2-[[(2s)-3-methyl-2-(methylamino)butanoyl]amino]butanoyl]amino]heptanoyl]pyrrolidin-2-yl]-2-methylpropanoyl]amino]-3-phenylpropanoic acid Chemical compound CN[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N(C)[C@@H]([C@@H](C)CC)[C@H](OC)CC(=O)N1CCC[C@H]1[C@H](OC)[C@@H](C)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 MFRNYXJJRJQHNW-DEMKXPNLSA-N 0.000 claims description 3
- NDMPLJNOPCLANR-UHFFFAOYSA-N 3,4-dihydroxy-15-(4-hydroxy-18-methoxycarbonyl-5,18-seco-ibogamin-18-yl)-16-methoxy-1-methyl-6,7-didehydro-aspidospermidine-3-carboxylic acid methyl ester Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 NDMPLJNOPCLANR-UHFFFAOYSA-N 0.000 claims description 3
- 108010027164 Amanitins Proteins 0.000 claims description 3
- 208000023275 Autoimmune disease Diseases 0.000 claims description 3
- PLXLYXLUCNZSAA-QLXKLKPCSA-N CC[C@@]1(O)C(=O)OCC2=C1C=C1N(CC3=C1N=C1C=C(F)C(C)=C4CC[C@H](NC(=O)CO)C3=C14)C2=O Chemical compound CC[C@@]1(O)C(=O)OCC2=C1C=C1N(CC3=C1N=C1C=C(F)C(C)=C4CC[C@H](NC(=O)CO)C3=C14)C2=O PLXLYXLUCNZSAA-QLXKLKPCSA-N 0.000 claims description 3
- KLWPJMFMVPTNCC-UHFFFAOYSA-N Camptothecin Natural products CCC1(O)C(=O)OCC2=C1C=C3C4Nc5ccccc5C=C4CN3C2=O KLWPJMFMVPTNCC-UHFFFAOYSA-N 0.000 claims description 3
- JRZJKWGQFNTSRN-UHFFFAOYSA-N Geldanamycin Natural products C1C(C)CC(OC)C(O)C(C)C=C(C)C(OC(N)=O)C(OC)CCC=C(C)C(=O)NC2=CC(=O)C(OC)=C1C2=O JRZJKWGQFNTSRN-UHFFFAOYSA-N 0.000 claims description 3
- 206010028980 Neoplasm Diseases 0.000 claims description 3
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 claims description 3
- CIORWBWIBBPXCG-JZTFPUPKSA-N amanitin Chemical compound O=C1N[C@@H](CC(N)=O)C(=O)N2CC(O)C[C@H]2C(=O)N[C@@H](C(C)[C@@H](O)CO)C(=O)N[C@@H](C2)C(=O)NCC(=O)N[C@@H](C(C)CC)C(=O)NCC(=O)N[C@H]1CS(=O)C1=C2C2=CC=C(O)C=C2N1 CIORWBWIBBPXCG-JZTFPUPKSA-N 0.000 claims description 3
- 229930195731 calicheamicin Natural products 0.000 claims description 3
- 229940127093 camptothecin Drugs 0.000 claims description 3
- 229960000684 cytarabine Drugs 0.000 claims description 3
- YJGVMLPVUAXIQN-UHFFFAOYSA-N epipodophyllotoxin Natural products COC1=C(OC)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YJGVMLPVUAXIQN-UHFFFAOYSA-N 0.000 claims description 3
- 229930013356 epothilone Natural products 0.000 claims description 3
- 150000003883 epothilone derivatives Chemical class 0.000 claims description 3
- QTQAWLPCGQOSGP-GBTDJJJQSA-N geldanamycin Chemical compound N1C(=O)\C(C)=C/C=C\[C@@H](OC)[C@H](OC(N)=O)\C(C)=C/[C@@H](C)[C@@H](O)[C@H](OC)C[C@@H](C)CC2=C(OC)C(=O)C=C1C2=O QTQAWLPCGQOSGP-GBTDJJJQSA-N 0.000 claims description 3
- UWKQSNNFCGGAFS-XIFFEERXSA-N irinotecan Chemical compound C1=C2C(CC)=C3CN(C(C4=C([C@@](C(=O)OC4)(O)CC)C=4)=O)C=4C3=NC2=CC=C1OC(=O)N(CC1)CCC1N1CCCCC1 UWKQSNNFCGGAFS-XIFFEERXSA-N 0.000 claims description 3
- 229960004768 irinotecan Drugs 0.000 claims description 3
- 230000002147 killing effect Effects 0.000 claims description 3
- 108010093470 monomethyl auristatin E Proteins 0.000 claims description 3
- 229960001237 podophyllotoxin Drugs 0.000 claims description 3
- YJGVMLPVUAXIQN-XVVDYKMHSA-N podophyllotoxin Chemical compound COC1=C(OC)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@H](O)[C@@H]3[C@@H]2C(OC3)=O)=C1 YJGVMLPVUAXIQN-XVVDYKMHSA-N 0.000 claims description 3
- YVCVYCSAAZQOJI-UHFFFAOYSA-N podophyllotoxin Natural products COC1=C(O)C(OC)=CC(C2C3=CC=4OCOC=4C=C3C(O)C3C2C(OC3)=O)=C1 YVCVYCSAAZQOJI-UHFFFAOYSA-N 0.000 claims description 3
- 230000002265 prevention Effects 0.000 claims description 3
- UCFGDBYHRUNTLO-QHCPKHFHSA-N topotecan Chemical compound C1=C(O)C(CN(C)C)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 UCFGDBYHRUNTLO-QHCPKHFHSA-N 0.000 claims description 3
- 229960000303 topotecan Drugs 0.000 claims description 3
- 229930184737 tubulysin Natural products 0.000 claims description 3
- 210000004881 tumor cell Anatomy 0.000 claims description 3
- 229960003048 vinblastine Drugs 0.000 claims description 3
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 claims description 3
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 claims description 3
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 claims description 3
- 229960004528 vincristine Drugs 0.000 claims description 3
- UGGWPQSBPIFKDZ-KOTLKJBCSA-N vindesine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(N)=O)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1N=C1[C]2C=CC=C1 UGGWPQSBPIFKDZ-KOTLKJBCSA-N 0.000 claims description 3
- 229960004355 vindesine Drugs 0.000 claims description 3
- BLUGYPPOFIHFJS-UUFHNPECSA-N (2s)-n-[(2s)-1-[[(3r,4s,5s)-3-methoxy-1-[(2s)-2-[(1r,2r)-1-methoxy-2-methyl-3-oxo-3-[[(1s)-2-phenyl-1-(1,3-thiazol-2-yl)ethyl]amino]propyl]pyrrolidin-1-yl]-5-methyl-1-oxoheptan-4-yl]-methylamino]-3-methyl-1-oxobutan-2-yl]-3-methyl-2-(methylamino)butanamid Chemical compound CN[C@@H](C(C)C)C(=O)N[C@@H](C(C)C)C(=O)N(C)[C@@H]([C@@H](C)CC)[C@H](OC)CC(=O)N1CCC[C@H]1[C@H](OC)[C@@H](C)C(=O)N[C@H](C=1SC=CN=1)CC1=CC=CC=C1 BLUGYPPOFIHFJS-UUFHNPECSA-N 0.000 claims description 2
- DLKUYSQUHXBYPB-NSSHGSRYSA-N (2s,4r)-4-[[2-[(1r,3r)-1-acetyloxy-4-methyl-3-[3-methylbutanoyloxymethyl-[(2s,3s)-3-methyl-2-[[(2r)-1-methylpiperidine-2-carbonyl]amino]pentanoyl]amino]pentyl]-1,3-thiazole-4-carbonyl]amino]-2-methyl-5-(4-methylphenyl)pentanoic acid Chemical compound N([C@@H]([C@@H](C)CC)C(=O)N(COC(=O)CC(C)C)[C@H](C[C@@H](OC(C)=O)C=1SC=C(N=1)C(=O)N[C@H](C[C@H](C)C(O)=O)CC=1C=CC(C)=CC=1)C(C)C)C(=O)[C@H]1CCCCN1C DLKUYSQUHXBYPB-NSSHGSRYSA-N 0.000 claims description 2
- NWXMGUDVXFXRIG-WESIUVDSSA-N (4s,4as,5as,6s,12ar)-4-(dimethylamino)-1,6,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carboxamide Chemical compound C1=CC=C2[C@](O)(C)[C@H]3C[C@H]4[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]4(O)C(=O)C3=C(O)C2=C1O NWXMGUDVXFXRIG-WESIUVDSSA-N 0.000 claims description 2
- INAUWOVKEZHHDM-PEDBPRJASA-N (7s,9s)-6,9,11-trihydroxy-9-(2-hydroxyacetyl)-7-[(2r,4s,5s,6s)-5-hydroxy-6-methyl-4-morpholin-4-yloxan-2-yl]oxy-4-methoxy-8,10-dihydro-7h-tetracene-5,12-dione;hydrochloride Chemical compound Cl.N1([C@H]2C[C@@H](O[C@@H](C)[C@H]2O)O[C@H]2C[C@@](O)(CC=3C(O)=C4C(=O)C=5C=CC=C(C=5C(=O)C4=C(O)C=32)OC)C(=O)CO)CCOCC1 INAUWOVKEZHHDM-PEDBPRJASA-N 0.000 claims description 2
- OBVVKJDJORDBCH-CZDIJEQGSA-N 2-aminoacetic acid (2S)-2-amino-3-phenylpropanoic acid Chemical compound NCC(O)=O.NCC(O)=O.NCC(O)=O.OC(=O)[C@@H](N)CC1=CC=CC=C1 OBVVKJDJORDBCH-CZDIJEQGSA-N 0.000 claims description 2
- TVZGACDUOSZQKY-LBPRGKRZSA-N 4-aminofolic acid Chemical compound C1=NC2=NC(N)=NC(N)=C2N=C1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 TVZGACDUOSZQKY-LBPRGKRZSA-N 0.000 claims description 2
- 208000007934 ACTH-independent macronodular adrenal hyperplasia Diseases 0.000 claims description 2
- WEAHRLBPCANXCN-UHFFFAOYSA-N Daunomycin Natural products CCC1(O)CC(OC2CC(N)C(O)C(C)O2)c3cc4C(=O)c5c(OC)cccc5C(=O)c4c(O)c3C1 WEAHRLBPCANXCN-UHFFFAOYSA-N 0.000 claims description 2
- 206010061218 Inflammation Diseases 0.000 claims description 2
- 229930192392 Mitomycin Natural products 0.000 claims description 2
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 claims description 2
- FONIWJIDLJEJTL-UHFFFAOYSA-N N(8)-acetylspermidine Chemical compound CC(=O)NCCCCNCCCN FONIWJIDLJEJTL-UHFFFAOYSA-N 0.000 claims description 2
- 229940123237 Taxane Drugs 0.000 claims description 2
- PPORNUWQIKUROY-AINMIYBESA-N [(2r,3s,4r,5r,6r)-2-[(2r,3s,4r,5s,6s)-2-[(3s)-3-[[6-amino-2-[(1s)-3-amino-1-[[(2s)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-4-[[(3r,4s,5s)-6-[[(2s)-1-[2-[(4r)-4-[4-[(4-amino-4-iminobutyl)carbamoyl]-1,3-thiazol-2-yl] Chemical compound O([C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@H]1OC(C)[C@H](NC(=O)C=1C(=C(N)N=C(N=1)[C@H](CC(N)=O)NC[C@H](N)C(N)=O)C)C(=O)N[C@H](CCO)[C@@H](O)[C@H](C)C(=O)N[C@H](C(=O)NCCC=1SC[C@@H](N=1)C=1SC=C(N=1)C(=O)NCCCC(N)=N)C(C)(C)O)[C@H]1O[C@H](CO)[C@@H](O)[C@@H](OC(N)=O)[C@@H]1O PPORNUWQIKUROY-AINMIYBESA-N 0.000 claims description 2
- 150000007513 acids Chemical class 0.000 claims description 2
- 229960003896 aminopterin Drugs 0.000 claims description 2
- 108010044540 auristatin Proteins 0.000 claims description 2
- 201000011510 cancer Diseases 0.000 claims description 2
- ZVYVPGLRVWUPMP-FYSMJZIKSA-N exatecan Chemical compound C1C[C@H](N)C2=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC3=CC(F)=C(C)C1=C32 ZVYVPGLRVWUPMP-FYSMJZIKSA-N 0.000 claims description 2
- 229950009429 exatecan Drugs 0.000 claims description 2
- 230000004054 inflammatory process Effects 0.000 claims description 2
- 239000002207 metabolite Substances 0.000 claims description 2
- 229960004857 mitomycin Drugs 0.000 claims description 2
- 108010059074 monomethylauristatin F Proteins 0.000 claims description 2
- LEVWYRKDKASIDU-QWWZWVQMSA-N D-cystine Chemical compound OC(=O)[C@H](N)CSSC[C@@H](N)C(O)=O LEVWYRKDKASIDU-QWWZWVQMSA-N 0.000 claims 2
- WMAXWOOEPJQXEB-UHFFFAOYSA-N 2-phenyl-5-(4-phenylphenyl)-1,3,4-oxadiazole Chemical compound C1=CC=CC=C1C1=NN=C(C=2C=CC(=CC=2)C=2C=CC=CC=2)O1 WMAXWOOEPJQXEB-UHFFFAOYSA-N 0.000 claims 1
- WYTGDNHDOZPMIW-RCBQFDQVSA-N alstonine Natural products C1=CC2=C3C=CC=CC3=NC2=C2N1C[C@H]1[C@H](C)OC=C(C(=O)OC)[C@H]1C2 WYTGDNHDOZPMIW-RCBQFDQVSA-N 0.000 claims 1
- HXCHCVDVKSCDHU-LULTVBGHSA-N calicheamicin Chemical compound C1[C@H](OC)[C@@H](NCC)CO[C@H]1O[C@H]1[C@H](O[C@@H]2C\3=C(NC(=O)OC)C(=O)C[C@](C/3=C/CSSSC)(O)C#C\C=C/C#C2)O[C@H](C)[C@@H](NO[C@@H]2O[C@H](C)[C@@H](SC(=O)C=3C(=C(OC)C(O[C@H]4[C@@H]([C@H](OC)[C@@H](O)[C@H](C)O4)O)=C(I)C=3C)OC)[C@@H](O)C2)[C@@H]1O HXCHCVDVKSCDHU-LULTVBGHSA-N 0.000 claims 1
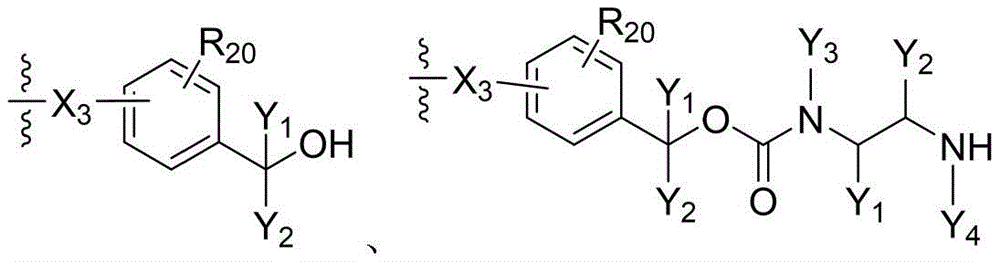
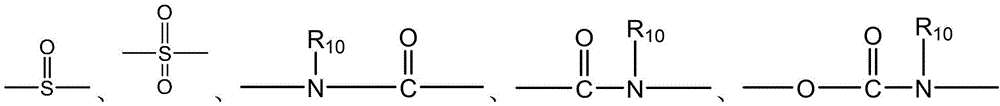
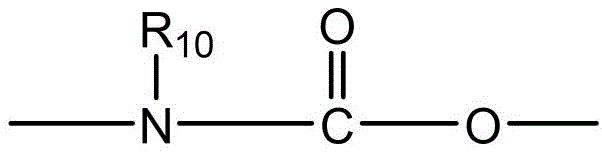
- 238000006243 chemical reaction Methods 0.000 abstract description 100
- 150000001875 compounds Chemical class 0.000 abstract description 20
- 238000010168 coupling process Methods 0.000 abstract description 9
- 238000005859 coupling reaction Methods 0.000 abstract description 8
- 230000008878 coupling Effects 0.000 abstract description 7
- 238000011068 loading method Methods 0.000 abstract description 4
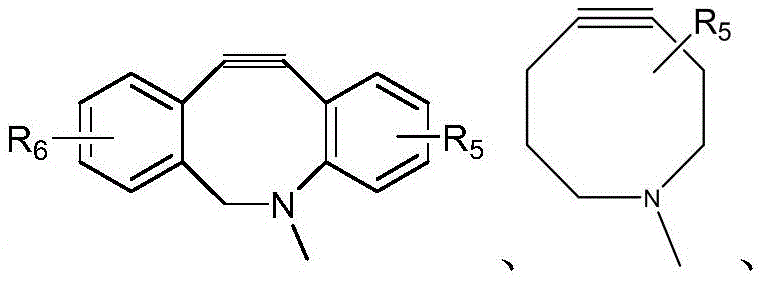
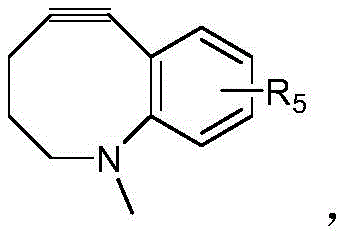
- IVRMZWNICZWHMI-UHFFFAOYSA-N azide group Chemical group [N-]=[N+]=[N-] IVRMZWNICZWHMI-UHFFFAOYSA-N 0.000 abstract description 3
- 231100000433 cytotoxic Toxicity 0.000 abstract description 3
- 230000001472 cytotoxic effect Effects 0.000 abstract description 3
- 229940127121 immunoconjugate Drugs 0.000 abstract description 3
- QRZUPJILJVGUFF-UHFFFAOYSA-N 2,8-dibenzylcyclooctan-1-one Chemical compound C1CCCCC(CC=2C=CC=CC=2)C(=O)C1CC1=CC=CC=C1 QRZUPJILJVGUFF-UHFFFAOYSA-N 0.000 abstract 1
- 238000010276 construction Methods 0.000 abstract 1
- 230000000857 drug effect Effects 0.000 abstract 1
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 94
- 239000000243 solution Substances 0.000 description 66
- 229940049595 antibody-drug conjugate Drugs 0.000 description 44
- 239000000611 antibody drug conjugate Substances 0.000 description 41
- 238000003786 synthesis reaction Methods 0.000 description 41
- 230000015572 biosynthetic process Effects 0.000 description 40
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 39
- 239000012074 organic phase Substances 0.000 description 31
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 30
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 30
- 238000004128 high performance liquid chromatography Methods 0.000 description 29
- 239000012043 crude product Substances 0.000 description 28
- WBHQBSYUUJJSRZ-UHFFFAOYSA-M sodium bisulfate Chemical class [Na+].OS([O-])(=O)=O WBHQBSYUUJJSRZ-UHFFFAOYSA-M 0.000 description 22
- 239000007787 solid Substances 0.000 description 22
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 21
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 21
- 230000002924 anti-infective effect Effects 0.000 description 21
- 239000000562 conjugate Substances 0.000 description 20
- 239000002904 solvent Substances 0.000 description 17
- 239000000539 dimer Substances 0.000 description 16
- 101100434411 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) ADH1 gene Proteins 0.000 description 15
- 101150102866 adc1 gene Proteins 0.000 description 15
- 101150042711 adc2 gene Proteins 0.000 description 15
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 14
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 13
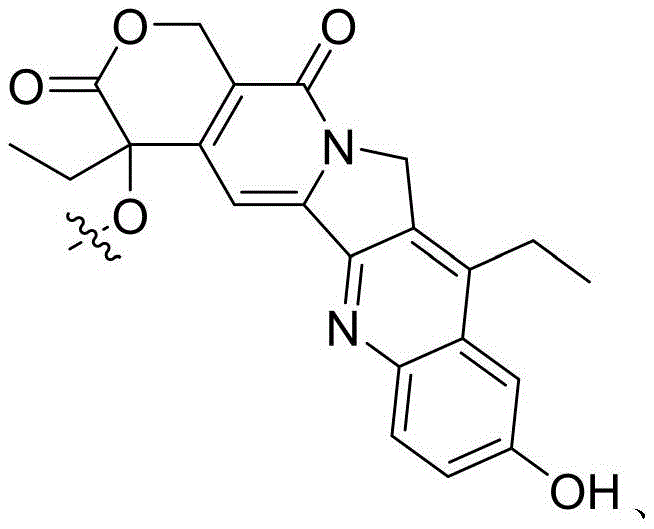
- FJHBVJOVLFPMQE-QFIPXVFZSA-N 7-Ethyl-10-Hydroxy-Camptothecin Chemical compound C1=C(O)C=C2C(CC)=C(CN3C(C4=C([C@@](C(=O)OC4)(O)CC)C=C33)=O)C3=NC2=C1 FJHBVJOVLFPMQE-QFIPXVFZSA-N 0.000 description 12
- 101100162020 Mesorhizobium japonicum (strain LMG 29417 / CECT 9101 / MAFF 303099) adc3 gene Proteins 0.000 description 12
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- 239000007864 aqueous solution Substances 0.000 description 12
- 239000007821 HATU Substances 0.000 description 11
- 210000004027 cell Anatomy 0.000 description 11
- 239000003112 inhibitor Substances 0.000 description 11
- 239000003443 antiviral agent Substances 0.000 description 9
- 239000002994 raw material Substances 0.000 description 9
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 8
- 229920006395 saturated elastomer Polymers 0.000 description 8
- 230000000694 effects Effects 0.000 description 7
- 239000000178 monomer Substances 0.000 description 7
- 230000035772 mutation Effects 0.000 description 7
- 229930186147 Cephalosporin Natural products 0.000 description 6
- 108010004469 allophycocyanin Proteins 0.000 description 6
- 229940124587 cephalosporin Drugs 0.000 description 6
- 150000001780 cephalosporins Chemical class 0.000 description 6
- 239000012141 concentrate Substances 0.000 description 6
- 125000000753 cycloalkyl group Chemical group 0.000 description 6
- 231100000135 cytotoxicity Toxicity 0.000 description 6
- 230000003013 cytotoxicity Effects 0.000 description 6
- 239000000047 product Substances 0.000 description 6
- 238000010898 silica gel chromatography Methods 0.000 description 6
- 239000003053 toxin Substances 0.000 description 6
- 231100000765 toxin Toxicity 0.000 description 6
- 108700012359 toxins Proteins 0.000 description 6
- 239000000579 Gonadotropin-Releasing Hormone Substances 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 5
- 239000003242 anti bacterial agent Substances 0.000 description 5
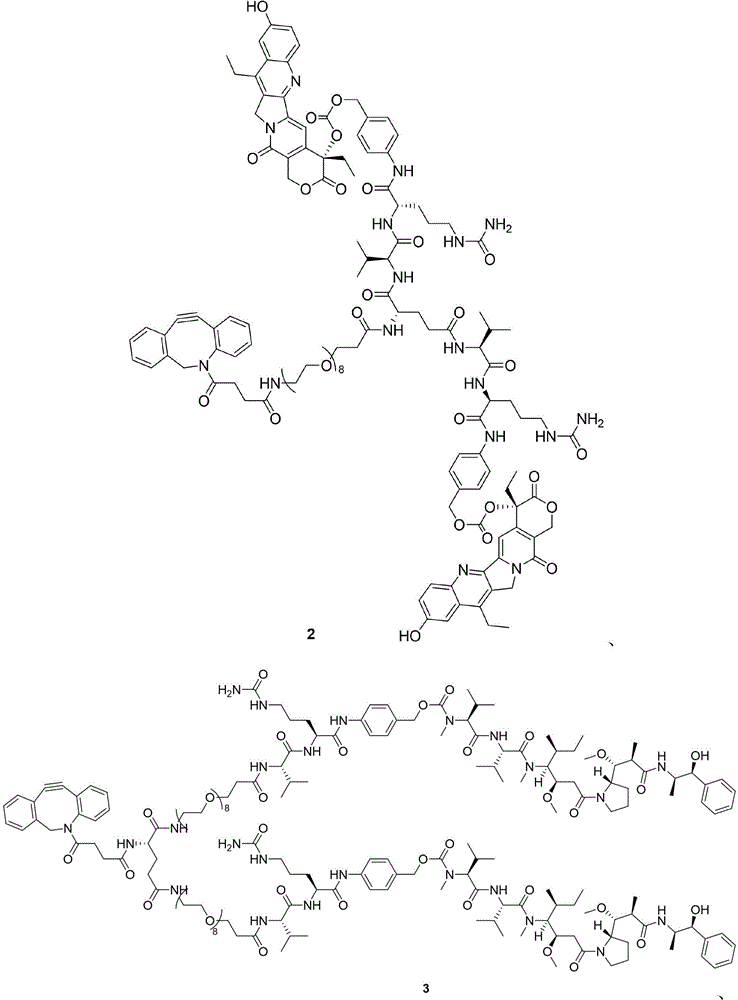
- 229940125904 compound 1 Drugs 0.000 description 5
- XCEBOJWFQSQZKR-UHFFFAOYSA-N dbco-nhs Chemical compound C1C2=CC=CC=C2C#CC2=CC=CC=C2N1C(=O)CCC(=O)ON1C(=O)CCC1=O XCEBOJWFQSQZKR-UHFFFAOYSA-N 0.000 description 5
- 239000005457 ice water Substances 0.000 description 5
- 229910000342 sodium bisulfate Inorganic materials 0.000 description 5
- VAVHMEQFYYBAPR-ITWZMISCSA-N (e,3r,5s)-7-[4-(4-fluorophenyl)-1-phenyl-2-propan-2-ylpyrrol-3-yl]-3,5-dihydroxyhept-6-enoic acid Chemical compound CC(C)C1=C(\C=C\[C@@H](O)C[C@@H](O)CC(O)=O)C(C=2C=CC(F)=CC=2)=CN1C1=CC=CC=C1 VAVHMEQFYYBAPR-ITWZMISCSA-N 0.000 description 4
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 4
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- ULGZDMOVFRHVEP-RWJQBGPGSA-N Erythromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 ULGZDMOVFRHVEP-RWJQBGPGSA-N 0.000 description 4
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 4
- AVKUERGKIZMTKX-NJBDSQKTSA-N ampicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=CC=C1 AVKUERGKIZMTKX-NJBDSQKTSA-N 0.000 description 4
- 229960000723 ampicillin Drugs 0.000 description 4
- 229940088710 antibiotic agent Drugs 0.000 description 4
- 229960001668 cefuroxime Drugs 0.000 description 4
- JFPVXVDWJQMJEE-IZRZKJBUSA-N cefuroxime Chemical compound N([C@@H]1C(N2C(=C(COC(N)=O)CS[C@@H]21)C(O)=O)=O)C(=O)\C(=N/OC)C1=CC=CO1 JFPVXVDWJQMJEE-IZRZKJBUSA-N 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 239000000975 dye Substances 0.000 description 4
- 238000005516 engineering process Methods 0.000 description 4
- 102000052116 epidermal growth factor receptor activity proteins Human genes 0.000 description 4
- 108700015053 epidermal growth factor receptor activity proteins Proteins 0.000 description 4
- 238000004811 liquid chromatography Methods 0.000 description 4
- HPNSFSBZBAHARI-UHFFFAOYSA-N micophenolic acid Natural products OC1=C(CC=C(C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-UHFFFAOYSA-N 0.000 description 4
- WIDKTXGNSOORHA-CJHXQPGBSA-N n,n'-dibenzylethane-1,2-diamine;(2s,5r,6r)-3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid;tetrahydrate Chemical compound O.O.O.O.C=1C=CC=CC=1CNCCNCC1=CC=CC=C1.N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1.N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 WIDKTXGNSOORHA-CJHXQPGBSA-N 0.000 description 4
- YOHYSYJDKVYCJI-UHFFFAOYSA-N n-[3-[[6-[3-(trifluoromethyl)anilino]pyrimidin-4-yl]amino]phenyl]cyclopropanecarboxamide Chemical group FC(F)(F)C1=CC=CC(NC=2N=CN=C(NC=3C=C(NC(=O)C4CC4)C=CC=3)C=2)=C1 YOHYSYJDKVYCJI-UHFFFAOYSA-N 0.000 description 4
- 239000002777 nucleoside Substances 0.000 description 4
- 235000019371 penicillin G benzathine Nutrition 0.000 description 4
- ZJAOAACCNHFJAH-UHFFFAOYSA-N phosphonoformic acid Chemical compound OC(=O)P(O)(O)=O ZJAOAACCNHFJAH-UHFFFAOYSA-N 0.000 description 4
- 235000018102 proteins Nutrition 0.000 description 4
- 102000004169 proteins and genes Human genes 0.000 description 4
- 108090000623 proteins and genes Proteins 0.000 description 4
- 229940044601 receptor agonist Drugs 0.000 description 4
- 239000000018 receptor agonist Substances 0.000 description 4
- 102000005962 receptors Human genes 0.000 description 4
- 108020003175 receptors Proteins 0.000 description 4
- JQXXHWHPUNPDRT-WLSIYKJHSA-N rifampicin Chemical compound O([C@](C1=O)(C)O/C=C/[C@@H]([C@H]([C@@H](OC(C)=O)[C@H](C)[C@H](O)[C@H](C)[C@@H](O)[C@@H](C)\C=C\C=C(C)/C(=O)NC=2C(O)=C3C([O-])=C4C)C)OC)C4=C1C3=C(O)C=2\C=N\N1CC[NH+](C)CC1 JQXXHWHPUNPDRT-WLSIYKJHSA-N 0.000 description 4
- 150000003384 small molecules Chemical class 0.000 description 4
- 239000011780 sodium chloride Substances 0.000 description 4
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 4
- 230000008685 targeting Effects 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- FPZLLRFZJZRHSY-HJYUBDRYSA-N tigecycline Chemical compound C([C@H]1C2)C3=C(N(C)C)C=C(NC(=O)CNC(C)(C)C)C(O)=C3C(=O)C1=C(O)[C@@]1(O)[C@@H]2[C@H](N(C)C)C(O)=C(C(N)=O)C1=O FPZLLRFZJZRHSY-HJYUBDRYSA-N 0.000 description 4
- 229960004089 tigecycline Drugs 0.000 description 4
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 4
- WMBWREPUVVBILR-WIYYLYMNSA-N (-)-Epigallocatechin-3-o-gallate Chemical compound O([C@@H]1CC2=C(O)C=C(C=C2O[C@@H]1C=1C=C(O)C(O)=C(O)C=1)O)C(=O)C1=CC(O)=C(O)C(O)=C1 WMBWREPUVVBILR-WIYYLYMNSA-N 0.000 description 3
- HBENZIXOGRCSQN-VQWWACLZSA-N (1S,2S,6R,14R,15R,16R)-5-(cyclopropylmethyl)-16-[(2S)-2-hydroxy-3,3-dimethylpentan-2-yl]-15-methoxy-13-oxa-5-azahexacyclo[13.2.2.12,8.01,6.02,14.012,20]icosa-8(20),9,11-trien-11-ol Chemical compound N1([C@@H]2CC=3C4=C(C(=CC=3)O)O[C@H]3[C@@]5(OC)CC[C@@]2([C@@]43CC1)C[C@@H]5[C@](C)(O)C(C)(C)CC)CC1CC1 HBENZIXOGRCSQN-VQWWACLZSA-N 0.000 description 3
- FANCTJAFZSYTIS-IQUVVAJASA-N (1r,3s,5z)-5-[(2e)-2-[(1r,3as,7ar)-7a-methyl-1-[(2r)-4-(phenylsulfonimidoyl)butan-2-yl]-2,3,3a,5,6,7-hexahydro-1h-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol Chemical compound C([C@@H](C)[C@@H]1[C@]2(CCCC(/[C@@H]2CC1)=C\C=C\1C([C@@H](O)C[C@H](O)C/1)=C)C)CS(=N)(=O)C1=CC=CC=C1 FANCTJAFZSYTIS-IQUVVAJASA-N 0.000 description 3
- SHAHPWSYJFYMRX-GDLCADMTSA-N (2S)-2-(4-{[(1R,2S)-2-hydroxycyclopentyl]methyl}phenyl)propanoic acid Chemical compound C1=CC([C@@H](C(O)=O)C)=CC=C1C[C@@H]1[C@@H](O)CCC1 SHAHPWSYJFYMRX-GDLCADMTSA-N 0.000 description 3
- PHDIJLFSKNMCMI-ITGJKDDRSA-N (3R,4S,5R,6R)-6-(hydroxymethyl)-4-(8-quinolin-6-yloxyoctoxy)oxane-2,3,5-triol Chemical compound OC[C@@H]1[C@H]([C@@H]([C@H](C(O1)O)O)OCCCCCCCCOC=1C=C2C=CC=NC2=CC=1)O PHDIJLFSKNMCMI-ITGJKDDRSA-N 0.000 description 3
- QKLXBIHSGMPUQS-FGZHOGPDSA-M (3r,5r)-7-[4-(4-fluorophenyl)-2,5-dimethyl-1-phenylpyrrol-3-yl]-3,5-dihydroxyheptanoate Chemical compound CC1=C(CC[C@@H](O)C[C@@H](O)CC([O-])=O)C(C=2C=CC(F)=CC=2)=C(C)N1C1=CC=CC=C1 QKLXBIHSGMPUQS-FGZHOGPDSA-M 0.000 description 3
- NXLNNXIXOYSCMB-UHFFFAOYSA-N (4-nitrophenyl) carbonochloridate Chemical compound [O-][N+](=O)C1=CC=C(OC(Cl)=O)C=C1 NXLNNXIXOYSCMB-UHFFFAOYSA-N 0.000 description 3
- PNHBRYIAJCYNDA-VQCQRNETSA-N (4r)-6-[2-[2-ethyl-4-(4-fluorophenyl)-6-phenylpyridin-3-yl]ethyl]-4-hydroxyoxan-2-one Chemical compound C([C@H](O)C1)C(=O)OC1CCC=1C(CC)=NC(C=2C=CC=CC=2)=CC=1C1=CC=C(F)C=C1 PNHBRYIAJCYNDA-VQCQRNETSA-N 0.000 description 3
- VIMMECPCYZXUCI-MIMFYIINSA-N (4s,6r)-6-[(1e)-4,4-bis(4-fluorophenyl)-3-(1-methyltetrazol-5-yl)buta-1,3-dienyl]-4-hydroxyoxan-2-one Chemical compound CN1N=NN=C1C(\C=C\[C@@H]1OC(=O)C[C@@H](O)C1)=C(C=1C=CC(F)=CC=1)C1=CC=C(F)C=C1 VIMMECPCYZXUCI-MIMFYIINSA-N 0.000 description 3
- QGKMIGUHVLGJBR-UHFFFAOYSA-M (4z)-1-(3-methylbutyl)-4-[[1-(3-methylbutyl)quinolin-1-ium-4-yl]methylidene]quinoline;iodide Chemical class [I-].C12=CC=CC=C2N(CCC(C)C)C=CC1=CC1=CC=[N+](CCC(C)C)C2=CC=CC=C12 QGKMIGUHVLGJBR-UHFFFAOYSA-M 0.000 description 3
- QOLHWXNSCZGWHK-BWBORTOCSA-N (6r,7r)-1-[(4s,5r)-4-acetyloxy-5-methyl-3-methylidene-6-phenylhexyl]-4,7-dihydroxy-6-(11-phenoxyundecylcarbamoyloxy)-2,8-dioxabicyclo[3.2.1]octane-3,4,5-tricarboxylic acid Chemical compound C([C@@H](C)[C@H](OC(C)=O)C(=C)CCC12[C@@H]([C@@H](OC(=O)NCCCCCCCCCCCOC=3C=CC=CC=3)C(O1)(C(O)=O)C(O)(C(O2)C(O)=O)C(O)=O)O)C1=CC=CC=C1 QOLHWXNSCZGWHK-BWBORTOCSA-N 0.000 description 3
- JNPGUXGVLNJQSQ-BGGMYYEUSA-M (e,3r,5s)-7-[4-(4-fluorophenyl)-1,2-di(propan-2-yl)pyrrol-3-yl]-3,5-dihydroxyhept-6-enoate Chemical compound CC(C)N1C(C(C)C)=C(\C=C\[C@@H](O)C[C@@H](O)CC([O-])=O)C(C=2C=CC(F)=CC=2)=C1 JNPGUXGVLNJQSQ-BGGMYYEUSA-M 0.000 description 3
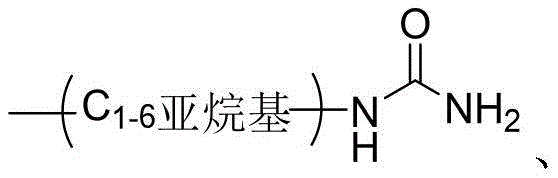
- VSNHCAURESNICA-NJFSPNSNSA-N 1-oxidanylurea Chemical compound N[14C](=O)NO VSNHCAURESNICA-NJFSPNSNSA-N 0.000 description 3
- JGMXNNSYEFOBHQ-OWOJBTEDSA-N 2-[(e)-4-morpholin-4-ylbut-2-enyl]-1,1-dioxothieno[3,2-e]thiazine-6-sulfonamide Chemical compound O=S1(=O)C=2SC(S(=O)(=O)N)=CC=2C=CN1C\C=C\CN1CCOCC1 JGMXNNSYEFOBHQ-OWOJBTEDSA-N 0.000 description 3
- QLVGHFBUSGYCCG-UHFFFAOYSA-N 2-amino-n-(1-cyano-2-phenylethyl)acetamide Chemical compound NCC(=O)NC(C#N)CC1=CC=CC=C1 QLVGHFBUSGYCCG-UHFFFAOYSA-N 0.000 description 3
- YLLRPQWLASQXSI-UHFFFAOYSA-N 3-(4b,8a,9,9a-tetrahydro-4aH-pyrido[2,3-b]indol-4-ylamino)phenol Chemical compound Oc1cccc(NC2=CC=NC3NC4C=CC=CC4C23)c1 YLLRPQWLASQXSI-UHFFFAOYSA-N 0.000 description 3
- XDCOYBQVEVSNNB-UHFFFAOYSA-N 4-[(7-naphthalen-2-yl-1-benzothiophen-2-yl)methylamino]butanoic acid Chemical compound OC(=O)CCCNCc1cc2cccc(-c3ccc4ccccc4c3)c2s1 XDCOYBQVEVSNNB-UHFFFAOYSA-N 0.000 description 3
- HIHOEGPXVVKJPP-JTQLQIEISA-N 5-fluoro-2-[[(1s)-1-(5-fluoropyridin-2-yl)ethyl]amino]-6-[(5-methyl-1h-pyrazol-3-yl)amino]pyridine-3-carbonitrile Chemical compound N([C@@H](C)C=1N=CC(F)=CC=1)C(C(=CC=1F)C#N)=NC=1NC=1C=C(C)NN=1 HIHOEGPXVVKJPP-JTQLQIEISA-N 0.000 description 3
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 3
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 241000219198 Brassica Species 0.000 description 3
- 235000003351 Brassica cretica Nutrition 0.000 description 3
- 235000003343 Brassica rupestris Nutrition 0.000 description 3
- 229940126559 Compound 4e Drugs 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- OVBPIULPVIDEAO-LBPRGKRZSA-N Folic acid Natural products C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-LBPRGKRZSA-N 0.000 description 3
- WMBWREPUVVBILR-UHFFFAOYSA-N GCG Natural products C=1C(O)=C(O)C(O)=CC=1C1OC2=CC(O)=CC(O)=C2CC1OC(=O)C1=CC(O)=C(O)C(O)=C1 WMBWREPUVVBILR-UHFFFAOYSA-N 0.000 description 3
- 108700012941 GNRH1 Proteins 0.000 description 3
- XDXDZDZNSLXDNA-TZNDIEGXSA-N Idarubicin Chemical compound C1[C@H](N)[C@H](O)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2C[C@@](O)(C(C)=O)C1 XDXDZDZNSLXDNA-TZNDIEGXSA-N 0.000 description 3
- 108010000817 Leuprolide Proteins 0.000 description 3
- TZYWCYJVHRLUCT-VABKMULXSA-N N-benzyloxycarbonyl-L-leucyl-L-leucyl-L-leucinal Chemical compound CC(C)C[C@@H](C=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(C)C)NC(=O)OCC1=CC=CC=C1 TZYWCYJVHRLUCT-VABKMULXSA-N 0.000 description 3
- TZCCKCLHNUSAMQ-DUGSHLAESA-N NC(=O)C[C@H](NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](Cc2ccc(F)cc2)NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)Cc5cccs5)C(=O)N Chemical compound NC(=O)C[C@H](NC(=O)[C@H](CCCNC(=N)N)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(=N)N)NC(=O)[C@H](Cc2ccc(F)cc2)NC(=O)[C@H](Cc3c[nH]c4ccccc34)NC(=O)Cc5cccs5)C(=O)N TZCCKCLHNUSAMQ-DUGSHLAESA-N 0.000 description 3
- IWUCXVSUMQZMFG-AFCXAGJDSA-N Ribavirin Chemical compound N1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 IWUCXVSUMQZMFG-AFCXAGJDSA-N 0.000 description 3
- 229940125907 SJ995973 Drugs 0.000 description 3
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 3
- ISAKRJDGNUQOIC-UHFFFAOYSA-N Uracil Chemical compound O=C1C=CNC(=O)N1 ISAKRJDGNUQOIC-UHFFFAOYSA-N 0.000 description 3
- YLEIFZAVNWDOBM-ZTNXSLBXSA-N ac1l9hc7 Chemical compound C([C@H]12)C[C@@H](C([C@@H](O)CC3)(C)C)[C@@]43C[C@@]14CC[C@@]1(C)[C@@]2(C)C[C@@H]2O[C@]3(O)[C@H](O)C(C)(C)O[C@@H]3[C@@H](C)[C@H]12 YLEIFZAVNWDOBM-ZTNXSLBXSA-N 0.000 description 3
- 239000000556 agonist Substances 0.000 description 3
- SRVFFFJZQVENJC-IHRRRGAJSA-N aloxistatin Chemical compound CCOC(=O)[C@H]1O[C@@H]1C(=O)N[C@@H](CC(C)C)C(=O)NCCC(C)C SRVFFFJZQVENJC-IHRRRGAJSA-N 0.000 description 3
- 230000001093 anti-cancer Effects 0.000 description 3
- 239000000427 antigen Substances 0.000 description 3
- 108091007433 antigens Proteins 0.000 description 3
- 102000036639 antigens Human genes 0.000 description 3
- CLKOFPXJLQSYAH-ABRJDSQDSA-N bacitracin A Chemical compound C1SC([C@@H](N)[C@@H](C)CC)=N[C@@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](CCC(O)=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]1C(=O)N[C@H](CCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2N=CNC=2)C(=O)N[C@H](CC(O)=O)C(=O)N[C@@H](CC(N)=O)C(=O)NCCCC1 CLKOFPXJLQSYAH-ABRJDSQDSA-N 0.000 description 3
- QKSKPIVNLNLAAV-UHFFFAOYSA-N bis(2-chloroethyl) sulfide Chemical compound ClCCSCCCl QKSKPIVNLNLAAV-UHFFFAOYSA-N 0.000 description 3
- OSVHLUXLWQLPIY-KBAYOESNSA-N butyl 2-[(6aR,9R,10aR)-1-hydroxy-9-(hydroxymethyl)-6,6-dimethyl-6a,7,8,9,10,10a-hexahydrobenzo[c]chromen-3-yl]-2-methylpropanoate Chemical compound C(CCC)OC(C(C)(C)C1=CC(=C2[C@H]3[C@H](C(OC2=C1)(C)C)CC[C@H](C3)CO)O)=O OSVHLUXLWQLPIY-KBAYOESNSA-N 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 229960004261 cefotaxime Drugs 0.000 description 3
- AZZMGZXNTDTSME-JUZDKLSSSA-M cefotaxime sodium Chemical compound [Na+].N([C@@H]1C(N2C(=C(COC(C)=O)CS[C@@H]21)C([O-])=O)=O)C(=O)\C(=N/OC)C1=CSC(N)=N1 AZZMGZXNTDTSME-JUZDKLSSSA-M 0.000 description 3
- 229960000484 ceftazidime Drugs 0.000 description 3
- ORFOPKXBNMVMKC-DWVKKRMSSA-N ceftazidime Chemical compound S([C@@H]1[C@@H](C(N1C=1C([O-])=O)=O)NC(=O)\C(=N/OC(C)(C)C(O)=O)C=2N=C(N)SC=2)CC=1C[N+]1=CC=CC=C1 ORFOPKXBNMVMKC-DWVKKRMSSA-N 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 238000003776 cleavage reaction Methods 0.000 description 3
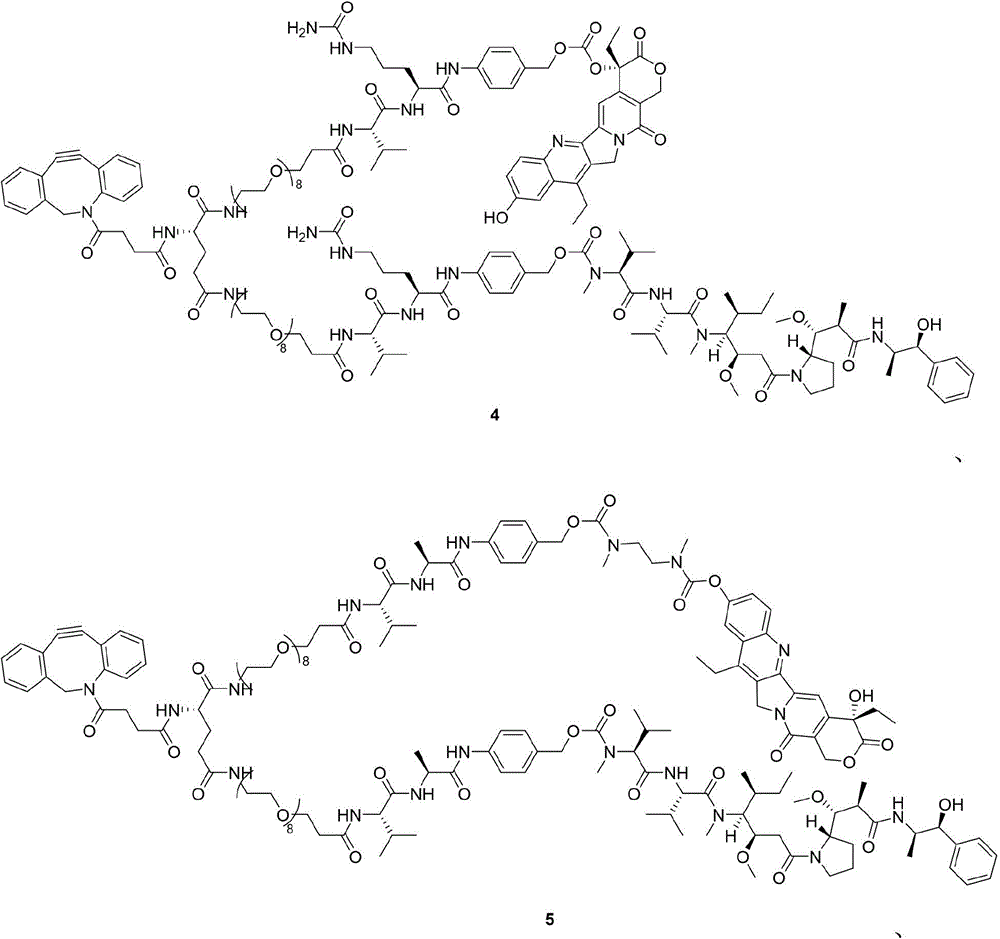
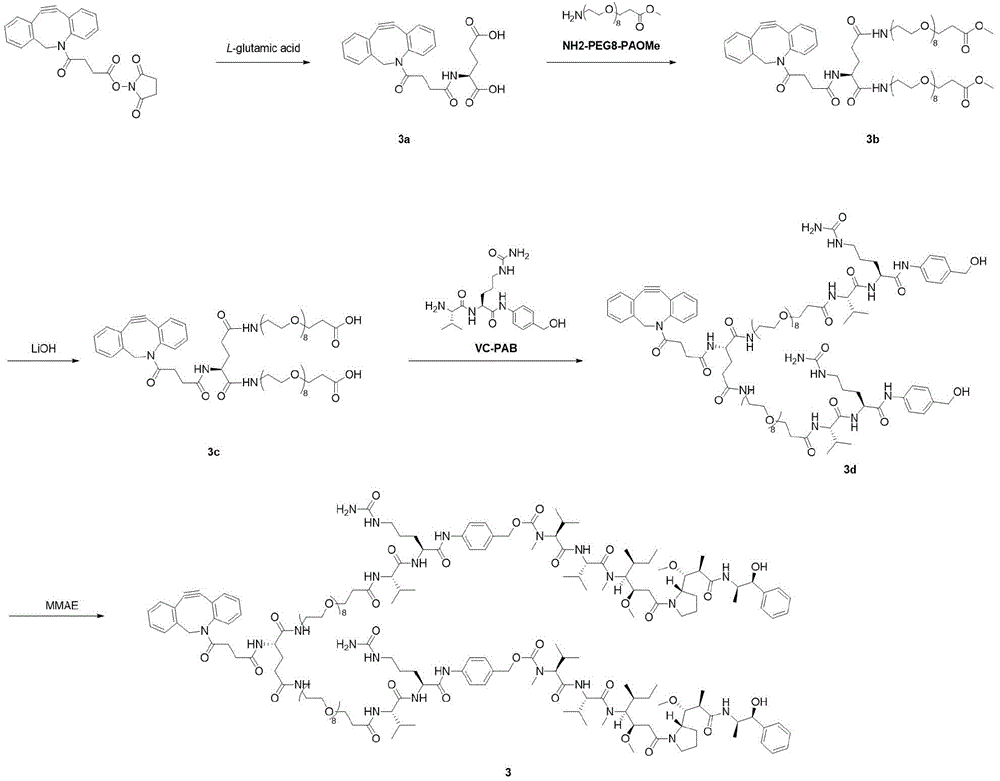
- 229940125796 compound 3d Drugs 0.000 description 3
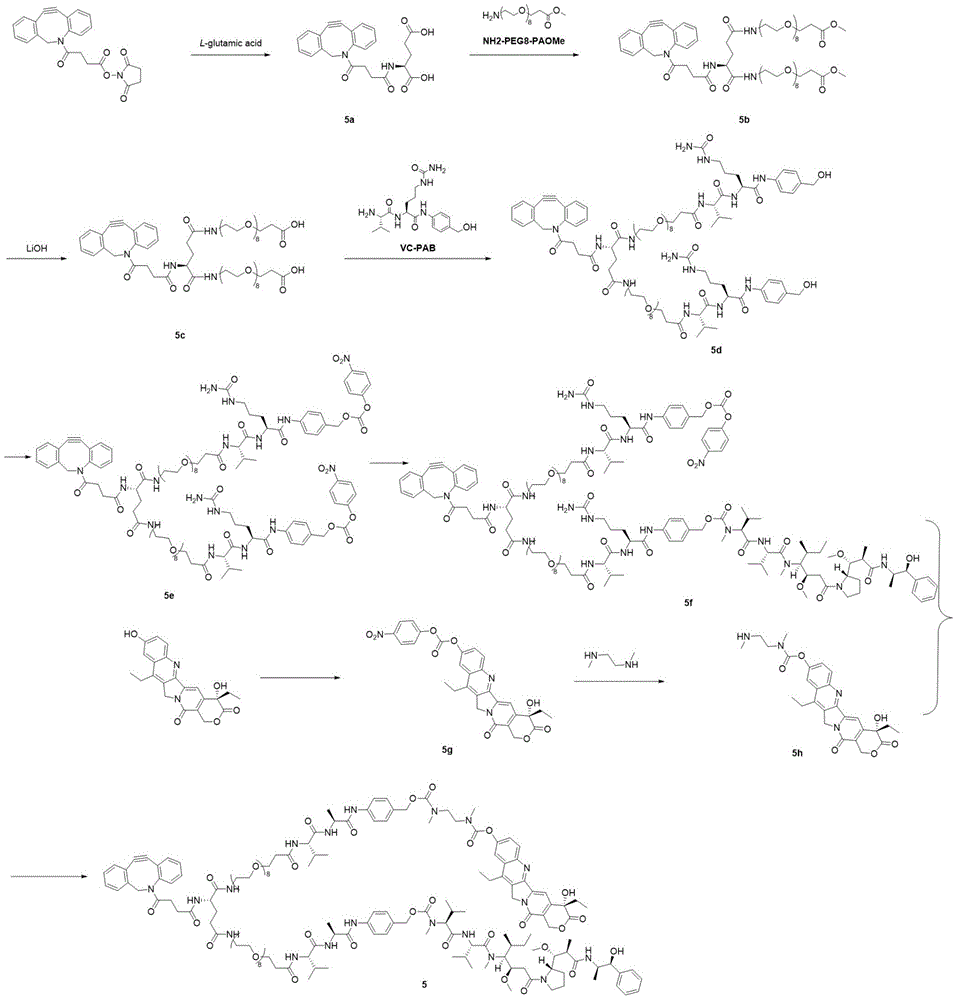
- 229940125872 compound 4d Drugs 0.000 description 3
- 229940126115 compound 4f Drugs 0.000 description 3
- 229940125898 compound 5 Drugs 0.000 description 3
- 238000004132 cross linking Methods 0.000 description 3
- OPTASPLRGRRNAP-UHFFFAOYSA-N cytosine Chemical class NC=1C=CNC(=O)N=1 OPTASPLRGRRNAP-UHFFFAOYSA-N 0.000 description 3
- 229960000975 daunorubicin Drugs 0.000 description 3
- 238000013461 design Methods 0.000 description 3
- YWKVMGDEOUPQGN-UHFFFAOYSA-N ethyl 2-[4-[2-(3-hydroxy-1-azabicyclo[2.2.2]octan-3-yl)ethynyl]phenyl]acetate Chemical compound C1=CC(CC(=O)OCC)=CC=C1C#CC1(O)C(CC2)CCN2C1 YWKVMGDEOUPQGN-UHFFFAOYSA-N 0.000 description 3
- 125000000031 ethylamino group Chemical group [H]C([H])([H])C([H])([H])N([H])[*] 0.000 description 3
- XLXSAKCOAKORKW-AQJXLSMYSA-N gonadorelin Chemical compound C([C@@H](C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 XLXSAKCOAKORKW-AQJXLSMYSA-N 0.000 description 3
- GFIJNRVAKGFPGQ-LIJARHBVSA-N leuprolide Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 GFIJNRVAKGFPGQ-LIJARHBVSA-N 0.000 description 3
- 229960004338 leuprorelin Drugs 0.000 description 3
- JFOZKMSJYSPYLN-QHCPKHFHSA-N lifitegrast Chemical compound CS(=O)(=O)C1=CC=CC(C[C@H](NC(=O)C=2C(=C3CCN(CC3=CC=2Cl)C(=O)C=2C=C3OC=CC3=CC=2)Cl)C(O)=O)=C1 JFOZKMSJYSPYLN-QHCPKHFHSA-N 0.000 description 3
- 238000000034 method Methods 0.000 description 3
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 description 3
- 229960001156 mitoxantrone Drugs 0.000 description 3
- 235000010460 mustard Nutrition 0.000 description 3
- 229960000951 mycophenolic acid Drugs 0.000 description 3
- HPNSFSBZBAHARI-RUDMXATFSA-N mycophenolic acid Chemical compound OC1=C(C\C=C(/C)CCC(O)=O)C(OC)=C(C)C2=C1C(=O)OC2 HPNSFSBZBAHARI-RUDMXATFSA-N 0.000 description 3
- QAPTWHXHEYAIKG-RCOXNQKVSA-N n-[(1r,2s,5r)-5-(tert-butylamino)-2-[(3s)-2-oxo-3-[[6-(trifluoromethyl)quinazolin-4-yl]amino]pyrrolidin-1-yl]cyclohexyl]acetamide Chemical compound CC(=O)N[C@@H]1C[C@H](NC(C)(C)C)CC[C@@H]1N1C(=O)[C@@H](NC=2C3=CC(=CC=C3N=CN=2)C(F)(F)F)CC1 QAPTWHXHEYAIKG-RCOXNQKVSA-N 0.000 description 3
- GVOISEJVFFIGQE-YCZSINBZSA-N n-[(1r,2s,5r)-5-[methyl(propan-2-yl)amino]-2-[(3s)-2-oxo-3-[[6-(trifluoromethyl)quinazolin-4-yl]amino]pyrrolidin-1-yl]cyclohexyl]acetamide Chemical compound CC(=O)N[C@@H]1C[C@H](N(C)C(C)C)CC[C@@H]1N1C(=O)[C@@H](NC=2C3=CC(=CC=C3N=CN=2)C(F)(F)F)CC1 GVOISEJVFFIGQE-YCZSINBZSA-N 0.000 description 3
- JBLLRCOZJMVOAE-HSQYWUDLSA-N n-[(2s)-1-[[(2s)-1-(1,3-benzothiazol-2-yl)-1-oxo-3-[(3s)-2-oxopyrrolidin-3-yl]propan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]-4-methoxy-1h-indole-2-carboxamide Chemical compound C([C@H](NC(=O)[C@H](CC(C)C)NC(=O)C=1NC=2C=CC=C(C=2C=1)OC)C(=O)C=1SC2=CC=CC=C2N=1)[C@@H]1CCNC1=O JBLLRCOZJMVOAE-HSQYWUDLSA-N 0.000 description 3
- XZMHJYWMCRQSSI-UHFFFAOYSA-N n-[5-[2-(3-acetylanilino)-1,3-thiazol-4-yl]-4-methyl-1,3-thiazol-2-yl]benzamide Chemical compound CC(=O)C1=CC=CC(NC=2SC=C(N=2)C2=C(N=C(NC(=O)C=3C=CC=CC=3)S2)C)=C1 XZMHJYWMCRQSSI-UHFFFAOYSA-N 0.000 description 3
- 125000003835 nucleoside group Chemical group 0.000 description 3
- RWWYLEGWBNMMLJ-YSOARWBDSA-N remdesivir Chemical compound NC1=NC=NN2C1=CC=C2[C@]1([C@@H]([C@@H]([C@H](O1)CO[P@](=O)(OC1=CC=CC=C1)N[C@H](C(=O)OCC(CC)CC)C)O)O)C#N RWWYLEGWBNMMLJ-YSOARWBDSA-N 0.000 description 3
- 229960000329 ribavirin Drugs 0.000 description 3
- HZCAHMRRMINHDJ-DBRKOABJSA-N ribavirin Natural products O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1N=CN=C1 HZCAHMRRMINHDJ-DBRKOABJSA-N 0.000 description 3
- 229960000885 rifabutin Drugs 0.000 description 3
- 229960001225 rifampicin Drugs 0.000 description 3
- WDZCUPBHRAEYDL-GZAUEHORSA-N rifapentine Chemical compound O([C@](C1=O)(C)O/C=C/[C@@H]([C@H]([C@@H](OC(C)=O)[C@H](C)[C@H](O)[C@H](C)[C@@H](O)[C@@H](C)\C=C\C=C(C)/C(=O)NC=2C(O)=C3C(O)=C4C)C)OC)C4=C1C3=C(O)C=2\C=N\N(CC1)CCN1C1CCCC1 WDZCUPBHRAEYDL-GZAUEHORSA-N 0.000 description 3
- VHXNKPBCCMUMSW-FQEVSTJZSA-N rubitecan Chemical compound C1=CC([N+]([O-])=O)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VHXNKPBCCMUMSW-FQEVSTJZSA-N 0.000 description 3
- HXCHCVDVKSCDHU-PJKCJEBCSA-N s-[(2r,3s,4s,6s)-6-[[(2r,3s,4s,5r,6r)-5-[(2s,4s,5s)-5-(ethylamino)-4-methoxyoxan-2-yl]oxy-4-hydroxy-6-[[(2s,5z,9r,13e)-9-hydroxy-12-(methoxycarbonylamino)-13-[2-(methyltrisulfanyl)ethylidene]-11-oxo-2-bicyclo[7.3.1]trideca-1(12),5-dien-3,7-diynyl]oxy]-2-m Chemical compound C1[C@H](OC)[C@@H](NCC)CO[C@H]1O[C@H]1[C@H](O[C@@H]2C\3=C(NC(=O)OC)C(=O)C[C@@](C/3=C/CSSSC)(O)C#C\C=C/C#C2)O[C@H](C)[C@@H](NO[C@@H]2O[C@H](C)[C@@H](SC(=O)C=3C(=C(OC)C(O[C@H]4[C@@H]([C@H](OC)[C@@H](O)[C@H](C)O4)O)=C(I)C=3C)OC)[C@@H](O)C2)[C@@H]1O HXCHCVDVKSCDHU-PJKCJEBCSA-N 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 230000007017 scission Effects 0.000 description 3
- IZTQOLKUZKXIRV-YRVFCXMDSA-N sincalide Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)NCC(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](N)CC(O)=O)C1=CC=C(OS(O)(=O)=O)C=C1 IZTQOLKUZKXIRV-YRVFCXMDSA-N 0.000 description 3
- 229960005404 sulfamethoxazole Drugs 0.000 description 3
- JLKIGFTWXXRPMT-UHFFFAOYSA-N sulphamethoxazole Chemical compound O1C(C)=CC(NS(=O)(=O)C=2C=CC(N)=CC=2)=N1 JLKIGFTWXXRPMT-UHFFFAOYSA-N 0.000 description 3
- NHKZSTHOYNWEEZ-AFCXAGJDSA-N taribavirin Chemical compound N1=C(C(=N)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NHKZSTHOYNWEEZ-AFCXAGJDSA-N 0.000 description 3
- 229950006081 taribavirin Drugs 0.000 description 3
- DTQVDTLACAAQTR-UHFFFAOYSA-N trifluoroacetic acid Substances OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 3
- 102000003390 tumor necrosis factor Human genes 0.000 description 3
- DEQANNDTNATYII-OULOTJBUSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-19-[[(2r)-2-amino-3-phenylpropanoyl]amino]-16-benzyl-n-[(2r,3r)-1,3-dihydroxybutan-2-yl]-7-[(1r)-1-hydroxyethyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-1,2-dithia-5,8,11,14,17-pentazacycloicosane-4-carboxa Chemical compound C([C@@H](N)C(=O)N[C@H]1CSSC[C@H](NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](CC=2C3=CC=CC=C3NC=2)NC(=O)[C@H](CC=2C=CC=CC=2)NC1=O)C(=O)N[C@H](CO)[C@H](O)C)C1=CC=CC=C1 DEQANNDTNATYII-OULOTJBUSA-N 0.000 description 2
- WDLWHQDACQUCJR-ZAMMOSSLSA-N (6r,7r)-7-[[(2r)-2-azaniumyl-2-(4-hydroxyphenyl)acetyl]amino]-8-oxo-3-[(e)-prop-1-enyl]-5-thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylate Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@@H]3N(C2=O)C(=C(CS3)/C=C/C)C(O)=O)=CC=C(O)C=C1 WDLWHQDACQUCJR-ZAMMOSSLSA-N 0.000 description 2
- FDKXTQMXEQVLRF-ZHACJKMWSA-N (E)-dacarbazine Chemical compound CN(C)\N=N\c1[nH]cnc1C(N)=O FDKXTQMXEQVLRF-ZHACJKMWSA-N 0.000 description 2
- XDFNWJDGWJVGGN-UHFFFAOYSA-N 2-(2,7-dichloro-3,6-dihydroxy-9h-xanthen-9-yl)benzoic acid Chemical compound OC(=O)C1=CC=CC=C1C1C2=CC(Cl)=C(O)C=C2OC2=CC(O)=C(Cl)C=C21 XDFNWJDGWJVGGN-UHFFFAOYSA-N 0.000 description 2
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 description 2
- MJKVTPMWOKAVMS-UHFFFAOYSA-N 3-hydroxy-1-benzopyran-2-one Chemical compound C1=CC=C2OC(=O)C(O)=CC2=C1 MJKVTPMWOKAVMS-UHFFFAOYSA-N 0.000 description 2
- QXZBMSIDSOZZHK-DOPDSADYSA-N 31362-50-2 Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(N)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H]1NC(=O)CC1)C(C)C)C1=CNC=N1 QXZBMSIDSOZZHK-DOPDSADYSA-N 0.000 description 2
- YXHLJMWYDTXDHS-IRFLANFNSA-N 7-aminoactinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=C(N)C=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 YXHLJMWYDTXDHS-IRFLANFNSA-N 0.000 description 2
- 108700012813 7-aminoactinomycin D Proteins 0.000 description 2
- FUXVKZWTXQUGMW-FQEVSTJZSA-N 9-Aminocamptothecin Chemical compound C1=CC(N)=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 FUXVKZWTXQUGMW-FQEVSTJZSA-N 0.000 description 2
- XKJMBINCVNINCA-UHFFFAOYSA-N Alfalone Chemical compound CON(C)C(=O)NC1=CC=C(Cl)C(Cl)=C1 XKJMBINCVNINCA-UHFFFAOYSA-N 0.000 description 2
- IYMAXBFPHPZYIK-BQBZGAKWSA-N Arg-Gly-Asp Chemical compound NC(N)=NCCC[C@H](N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(O)=O IYMAXBFPHPZYIK-BQBZGAKWSA-N 0.000 description 2
- MLDQJTXFUGDVEO-UHFFFAOYSA-N BAY-43-9006 Chemical compound C1=NC(C(=O)NC)=CC(OC=2C=CC(NC(=O)NC=3C=C(C(Cl)=CC=3)C(F)(F)F)=CC=2)=C1 MLDQJTXFUGDVEO-UHFFFAOYSA-N 0.000 description 2
- 108010001478 Bacitracin Proteins 0.000 description 2
- BPYKTIZUTYGOLE-IFADSCNNSA-N Bilirubin Chemical compound N1C(=O)C(C)=C(C=C)\C1=C\C1=C(C)C(CCC(O)=O)=C(CC2=C(C(C)=C(\C=C/3C(=C(C=C)C(=O)N\3)C)N2)CCC(O)=O)N1 BPYKTIZUTYGOLE-IFADSCNNSA-N 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- GNWUOVJNSFPWDD-XMZRARIVSA-M Cefoxitin sodium Chemical compound [Na+].N([C@]1(OC)C(N2C(=C(COC(N)=O)CS[C@@H]21)C([O-])=O)=O)C(=O)CC1=CC=CS1 GNWUOVJNSFPWDD-XMZRARIVSA-M 0.000 description 2
- 102000020313 Cell-Penetrating Peptides Human genes 0.000 description 2
- 108010051109 Cell-Penetrating Peptides Proteins 0.000 description 2
- 101800001982 Cholecystokinin Proteins 0.000 description 2
- 102100025841 Cholecystokinin Human genes 0.000 description 2
- CMSMOCZEIVJLDB-UHFFFAOYSA-N Cyclophosphamide Chemical compound ClCCN(CCCl)P1(=O)NCCCO1 CMSMOCZEIVJLDB-UHFFFAOYSA-N 0.000 description 2
- PMATZTZNYRCHOR-CGLBZJNRSA-N Cyclosporin A Chemical compound CC[C@@H]1NC(=O)[C@H]([C@H](O)[C@H](C)C\C=C\C)N(C)C(=O)[C@H](C(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](CC(C)C)N(C)C(=O)[C@@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)N(C)C(=O)[C@H](C(C)C)NC(=O)[C@H](CC(C)C)N(C)C(=O)CN(C)C1=O PMATZTZNYRCHOR-CGLBZJNRSA-N 0.000 description 2
- 229930105110 Cyclosporin A Natural products 0.000 description 2
- 108010036949 Cyclosporine Proteins 0.000 description 2
- 239000012129 DRAQ7 reagent Substances 0.000 description 2
- AUGQEEXBDZWUJY-UHFFFAOYSA-N Diacetoxyscirpenol Natural products CC(=O)OCC12CCC(C)=CC1OC1C(O)C(OC(C)=O)C2(C)C11CO1 AUGQEEXBDZWUJY-UHFFFAOYSA-N 0.000 description 2
- AUGQEEXBDZWUJY-ZLJUKNTDSA-N Diacetoxyscirpenol Chemical compound C([C@]12[C@]3(C)[C@H](OC(C)=O)[C@@H](O)[C@H]1O[C@@H]1C=C(C)CC[C@@]13COC(=O)C)O2 AUGQEEXBDZWUJY-ZLJUKNTDSA-N 0.000 description 2
- XQSPYNMVSIKCOC-NTSWFWBYSA-N Emtricitabine Chemical compound C1=C(F)C(N)=NC(=O)N1[C@H]1O[C@@H](CO)SC1 XQSPYNMVSIKCOC-NTSWFWBYSA-N 0.000 description 2
- IECPWNUMDGFDKC-UHFFFAOYSA-N Fusicsaeure Natural products C12C(O)CC3C(=C(CCC=C(C)C)C(O)=O)C(OC(C)=O)CC3(C)C1(C)CCC1C2(C)CCC(O)C1C IECPWNUMDGFDKC-UHFFFAOYSA-N 0.000 description 2
- 102000004862 Gastrin releasing peptide Human genes 0.000 description 2
- 108090001053 Gastrin releasing peptide Proteins 0.000 description 2
- 108010069236 Goserelin Proteins 0.000 description 2
- BLCLNMBMMGCOAS-URPVMXJPSA-N Goserelin Chemical compound C([C@@H](C(=O)N[C@H](COC(C)(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NNC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 BLCLNMBMMGCOAS-URPVMXJPSA-N 0.000 description 2
- XDXDZDZNSLXDNA-UHFFFAOYSA-N Idarubicin Natural products C1C(N)C(O)C(C)OC1OC1C2=C(O)C(C(=O)C3=CC=CC=C3C3=O)=C3C(O)=C2CC(O)(C(C)=O)C1 XDXDZDZNSLXDNA-UHFFFAOYSA-N 0.000 description 2
- LEVWYRKDKASIDU-IMJSIDKUSA-N L-cystine Chemical compound [O-]C(=O)[C@@H]([NH3+])CSSC[C@H]([NH3+])C([O-])=O LEVWYRKDKASIDU-IMJSIDKUSA-N 0.000 description 2
- 239000005511 L01XE05 - Sorafenib Substances 0.000 description 2
- 102000009151 Luteinizing Hormone Human genes 0.000 description 2
- 108010073521 Luteinizing Hormone Proteins 0.000 description 2
- XUMBMVFBXHLACL-UHFFFAOYSA-N Melanin Chemical compound O=C1C(=O)C(C2=CNC3=C(C(C(=O)C4=C32)=O)C)=C2C4=CNC2=C1C XUMBMVFBXHLACL-UHFFFAOYSA-N 0.000 description 2
- OVBPIULPVIDEAO-UHFFFAOYSA-N N-Pteroyl-L-glutaminsaeure Natural products C=1N=C2NC(N)=NC(=O)C2=NC=1CNC1=CC=C(C(=O)NC(CCC(O)=O)C(O)=O)C=C1 OVBPIULPVIDEAO-UHFFFAOYSA-N 0.000 description 2
- 102000012064 NLR Proteins Human genes 0.000 description 2
- 108091005686 NOD-like receptors Proteins 0.000 description 2
- 102000015532 Nicotinamide phosphoribosyltransferase Human genes 0.000 description 2
- 108010064862 Nicotinamide phosphoribosyltransferase Proteins 0.000 description 2
- AKQOBHZKBDHWQI-BZEFIUHZSA-N O[C@H]1C[C@@H](CCC1)N1C(C2(C3=C1N=C(N=C3)NC1=CC=C(C=C1)S(=O)(=O)NC([2H])([2H])[2H])CC2)=O Chemical compound O[C@H]1C[C@@H](CCC1)N1C(C2(C3=C1N=C(N=C3)NC1=CC=C(C=C1)S(=O)(=O)NC([2H])([2H])[2H])CC2)=O AKQOBHZKBDHWQI-BZEFIUHZSA-N 0.000 description 2
- 108010016076 Octreotide Proteins 0.000 description 2
- 229930182555 Penicillin Natural products 0.000 description 2
- 229930195708 Penicillin V Natural products 0.000 description 2
- 108010010522 Phycobilisomes Proteins 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- 102000012338 Poly(ADP-ribose) Polymerases Human genes 0.000 description 2
- 108010061844 Poly(ADP-ribose) Polymerases Proteins 0.000 description 2
- 229920000776 Poly(Adenosine diphosphate-ribose) polymerase Polymers 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 2
- 101000857870 Squalus acanthias Gonadoliberin Proteins 0.000 description 2
- QJJXYPPXXYFBGM-LFZNUXCKSA-N Tacrolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1\C=C(/C)[C@@H]1[C@H](C)[C@@H](O)CC(=O)[C@H](CC=C)/C=C(C)/C[C@H](C)C[C@H](OC)[C@H]([C@H](C[C@H]2C)OC)O[C@@]2(O)C(=O)C(=O)N2CCCC[C@H]2C(=O)O1 QJJXYPPXXYFBGM-LFZNUXCKSA-N 0.000 description 2
- NKANXQFJJICGDU-QPLCGJKRSA-N Tamoxifen Chemical compound C=1C=CC=CC=1C(/CC)=C(C=1C=CC(OCCN(C)C)=CC=1)/C1=CC=CC=C1 NKANXQFJJICGDU-QPLCGJKRSA-N 0.000 description 2
- MUMGGOZAMZWBJJ-DYKIIFRCSA-N Testostosterone Chemical compound O=C1CC[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 MUMGGOZAMZWBJJ-DYKIIFRCSA-N 0.000 description 2
- 239000004098 Tetracycline Substances 0.000 description 2
- 108010003205 Vasoactive Intestinal Peptide Proteins 0.000 description 2
- 102400000015 Vasoactive intestinal peptide Human genes 0.000 description 2
- WREGKURFCTUGRC-POYBYMJQSA-N Zalcitabine Chemical compound O=C1N=C(N)C=CN1[C@@H]1O[C@H](CO)CC1 WREGKURFCTUGRC-POYBYMJQSA-N 0.000 description 2
- DPKHZNPWBDQZCN-UHFFFAOYSA-N acridine orange free base Chemical compound C1=CC(N(C)C)=CC2=NC3=CC(N(C)C)=CC=C3C=C21 DPKHZNPWBDQZCN-UHFFFAOYSA-N 0.000 description 2
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- OIRDTQYFTABQOQ-KQYNXXCUSA-N adenosine Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](CO)[C@@H](O)[C@H]1O OIRDTQYFTABQOQ-KQYNXXCUSA-N 0.000 description 2
- 229940009456 adriamycin Drugs 0.000 description 2
- 229930013930 alkaloid Natural products 0.000 description 2
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 2
- DKNWSYNQZKUICI-UHFFFAOYSA-N amantadine Chemical compound C1C(C2)CC3CC2CC1(N)C3 DKNWSYNQZKUICI-UHFFFAOYSA-N 0.000 description 2
- 229960003805 amantadine Drugs 0.000 description 2
- 229960004821 amikacin Drugs 0.000 description 2
- LKCWBDHBTVXHDL-RMDFUYIESA-N amikacin Chemical compound O([C@@H]1[C@@H](N)C[C@H]([C@@H]([C@H]1O)O[C@@H]1[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O1)O)NC(=O)[C@@H](O)CCN)[C@H]1O[C@H](CN)[C@@H](O)[C@H](O)[C@H]1O LKCWBDHBTVXHDL-RMDFUYIESA-N 0.000 description 2
- 229960003022 amoxicillin Drugs 0.000 description 2
- LSQZJLSUYDQPKJ-NJBDSQKTSA-N amoxicillin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)=CC=C(O)C=C1 LSQZJLSUYDQPKJ-NJBDSQKTSA-N 0.000 description 2
- 239000002246 antineoplastic agent Substances 0.000 description 2
- 108010072041 arginyl-glycyl-aspartic acid Proteins 0.000 description 2
- 229960003005 axitinib Drugs 0.000 description 2
- RITAVMQDGBJQJZ-FMIVXFBMSA-N axitinib Chemical compound CNC(=O)C1=CC=CC=C1SC1=CC=C(C(\C=C\C=2N=CC=CC=2)=NN2)C2=C1 RITAVMQDGBJQJZ-FMIVXFBMSA-N 0.000 description 2
- 229960002170 azathioprine Drugs 0.000 description 2
- LMEKQMALGUDUQG-UHFFFAOYSA-N azathioprine Chemical compound CN1C=NC([N+]([O-])=O)=C1SC1=NC=NC2=C1NC=N2 LMEKQMALGUDUQG-UHFFFAOYSA-N 0.000 description 2
- 229960003071 bacitracin Drugs 0.000 description 2
- 229930184125 bacitracin Natural products 0.000 description 2
- DZBUGLKDJFMEHC-UHFFFAOYSA-N benzoquinolinylidene Natural products C1=CC=CC2=CC3=CC=CC=C3N=C21 DZBUGLKDJFMEHC-UHFFFAOYSA-N 0.000 description 2
- 230000027455 binding Effects 0.000 description 2
- 230000017531 blood circulation Effects 0.000 description 2
- PPKJUHVNTMYXOD-PZGPJMECSA-N c49ws9n75l Chemical compound O=C([C@@H]1N(C2=O)CC[C@H]1S(=O)(=O)CCN(CC)CC)O[C@H](C(C)C)[C@H](C)\C=C\C(=O)NC\C=C\C(\C)=C\[C@@H](O)CC(=O)CC1=NC2=CO1.N([C@@H]1C(=O)N[C@@H](C(N2CCC[C@H]2C(=O)N(C)[C@@H](CC=2C=CC(=CC=2)N(C)C)C(=O)N2C[C@@H](CS[C@H]3C4CCN(CC4)C3)C(=O)C[C@H]2C(=O)N[C@H](C(=O)O[C@@H]1C)C=1C=CC=CC=1)=O)CC)C(=O)C1=NC=CC=C1O PPKJUHVNTMYXOD-PZGPJMECSA-N 0.000 description 2
- 239000011575 calcium Substances 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 238000004364 calculation method Methods 0.000 description 2
- CZPLANDPABRVHX-UHFFFAOYSA-N cascade blue Chemical compound C=1C2=CC=CC=C2C(NCC)=CC=1C(C=1C=CC(=CC=1)N(CC)CC)=C1C=CC(=[N+](CC)CC)C=C1 CZPLANDPABRVHX-UHFFFAOYSA-N 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 229960004069 cefditoren Drugs 0.000 description 2
- KMIPKYQIOVAHOP-YLGJWRNMSA-N cefditoren Chemical compound S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)\C(=N/OC)C=2N=C(N)SC=2)CC=1\C=C/C=1SC=NC=1C KMIPKYQIOVAHOP-YLGJWRNMSA-N 0.000 description 2
- 229960002100 cefepime Drugs 0.000 description 2
- HVFLCNVBZFFHBT-ZKDACBOMSA-N cefepime Chemical compound S([C@@H]1[C@@H](C(N1C=1C([O-])=O)=O)NC(=O)\C(=N/OC)C=2N=C(N)SC=2)CC=1C[N+]1(C)CCCC1 HVFLCNVBZFFHBT-ZKDACBOMSA-N 0.000 description 2
- 229960002129 cefixime Drugs 0.000 description 2
- OKBVVJOGVLARMR-QSWIMTSFSA-N cefixime Chemical compound S1C(N)=NC(C(=N\OCC(O)=O)\C(=O)N[C@@H]2C(N3C(=C(C=C)CS[C@@H]32)C(O)=O)=O)=C1 OKBVVJOGVLARMR-QSWIMTSFSA-N 0.000 description 2
- 229960002682 cefoxitin Drugs 0.000 description 2
- 229960002580 cefprozil Drugs 0.000 description 2
- 229960004755 ceftriaxone Drugs 0.000 description 2
- VAAUVRVFOQPIGI-SPQHTLEESA-N ceftriaxone Chemical compound S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)\C(=N/OC)C=2N=C(N)SC=2)CC=1CSC1=NC(=O)C(=O)NN1C VAAUVRVFOQPIGI-SPQHTLEESA-N 0.000 description 2
- 229940106164 cephalexin Drugs 0.000 description 2
- ZAIPMKNFIOOWCQ-UEKVPHQBSA-N cephalexin Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@@H]3N(C2=O)C(=C(CS3)C)C(O)=O)=CC=CC=C1 ZAIPMKNFIOOWCQ-UEKVPHQBSA-N 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 229940107137 cholecystokinin Drugs 0.000 description 2
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 2
- ZYVSOIYQKUDENJ-WKSBCEQHSA-N chromomycin A3 Chemical compound O([C@@H]1C[C@@H](O[C@H](C)[C@@H]1OC(C)=O)OC=1C=C2C=C3C[C@H]([C@@H](C(=O)C3=C(O)C2=C(O)C=1C)O[C@@H]1O[C@H](C)[C@@H](O)[C@H](O[C@@H]2O[C@H](C)[C@@H](O)[C@H](O[C@@H]3O[C@@H](C)[C@H](OC(C)=O)[C@@](C)(O)C3)C2)C1)[C@H](OC)C(=O)[C@@H](O)[C@@H](C)O)[C@@H]1C[C@@H](O)[C@@H](OC)[C@@H](C)O1 ZYVSOIYQKUDENJ-WKSBCEQHSA-N 0.000 description 2
- MYSWGUAQZAJSOK-UHFFFAOYSA-N ciprofloxacin Chemical compound C12=CC(N3CCNCC3)=C(F)C=C2C(=O)C(C(=O)O)=CN1C1CC1 MYSWGUAQZAJSOK-UHFFFAOYSA-N 0.000 description 2
- 229960002227 clindamycin Drugs 0.000 description 2
- KDLRVYVGXIQJDK-AWPVFWJPSA-N clindamycin Chemical compound CN1C[C@H](CCC)C[C@H]1C(=O)N[C@H]([C@H](C)Cl)[C@@H]1[C@H](O)[C@H](O)[C@@H](O)[C@@H](SC)O1 KDLRVYVGXIQJDK-AWPVFWJPSA-N 0.000 description 2
- 229940125782 compound 2 Drugs 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- 229940127108 compound 5g Drugs 0.000 description 2
- 230000021615 conjugation Effects 0.000 description 2
- PSNOPSMXOBPNNV-VVCTWANISA-N cryptophycin 1 Chemical compound C1=C(Cl)C(OC)=CC=C1C[C@@H]1C(=O)NC[C@@H](C)C(=O)O[C@@H](CC(C)C)C(=O)O[C@H]([C@H](C)[C@@H]2[C@H](O2)C=2C=CC=CC=2)C/C=C/C(=O)N1 PSNOPSMXOBPNNV-VVCTWANISA-N 0.000 description 2
- PSNOPSMXOBPNNV-UHFFFAOYSA-N cryptophycin-327 Natural products C1=C(Cl)C(OC)=CC=C1CC1C(=O)NCC(C)C(=O)OC(CC(C)C)C(=O)OC(C(C)C2C(O2)C=2C=CC=CC=2)CC=CC(=O)N1 PSNOPSMXOBPNNV-UHFFFAOYSA-N 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 229960004397 cyclophosphamide Drugs 0.000 description 2
- 229940127089 cytotoxic agent Drugs 0.000 description 2
- 231100000599 cytotoxic agent Toxicity 0.000 description 2
- 239000002619 cytotoxin Substances 0.000 description 2
- 229960003901 dacarbazine Drugs 0.000 description 2
- 229960000640 dactinomycin Drugs 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 229960003957 dexamethasone Drugs 0.000 description 2
- UREBDLICKHMUKA-CXSFZGCWSA-N dexamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-CXSFZGCWSA-N 0.000 description 2
- 238000009826 distribution Methods 0.000 description 2
- AUZONCFQVSMFAP-UHFFFAOYSA-N disulfiram Chemical compound CCN(CC)C(=S)SSC(=S)N(CC)CC AUZONCFQVSMFAP-UHFFFAOYSA-N 0.000 description 2
- NOPFSRXAKWQILS-UHFFFAOYSA-N docosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCO NOPFSRXAKWQILS-UHFFFAOYSA-N 0.000 description 2
- VLCYCQAOQCDTCN-UHFFFAOYSA-N eflornithine Chemical compound NCCCC(N)(C(F)F)C(O)=O VLCYCQAOQCDTCN-UHFFFAOYSA-N 0.000 description 2
- 229940030275 epigallocatechin gallate Drugs 0.000 description 2
- 229960003276 erythromycin Drugs 0.000 description 2
- 150000002148 esters Chemical class 0.000 description 2
- AEUTYOVWOVBAKS-UWVGGRQHSA-N ethambutol Chemical compound CC[C@@H](CO)NCCN[C@@H](CC)CO AEUTYOVWOVBAKS-UWVGGRQHSA-N 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 229960000390 fludarabine Drugs 0.000 description 2
- GIUYCYHIANZCFB-FJFJXFQQSA-N fludarabine phosphate Chemical compound C1=NC=2C(N)=NC(F)=NC=2N1[C@@H]1O[C@H](COP(O)(O)=O)[C@@H](O)[C@@H]1O GIUYCYHIANZCFB-FJFJXFQQSA-N 0.000 description 2
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 2
- 108010021843 fluorescent protein 583 Proteins 0.000 description 2
- 229960000304 folic acid Drugs 0.000 description 2
- 235000019152 folic acid Nutrition 0.000 description 2
- 239000011724 folic acid Substances 0.000 description 2
- 229960005102 foscarnet Drugs 0.000 description 2
- 229960004675 fusidic acid Drugs 0.000 description 2
- IECPWNUMDGFDKC-MZJAQBGESA-N fusidic acid Chemical compound O[C@@H]([C@@H]12)C[C@H]3\C(=C(/CCC=C(C)C)C(O)=O)[C@@H](OC(C)=O)C[C@]3(C)[C@@]2(C)CC[C@@H]2[C@]1(C)CC[C@@H](O)[C@H]2C IECPWNUMDGFDKC-MZJAQBGESA-N 0.000 description 2
- CHPZKNULDCNCBW-UHFFFAOYSA-N gallium nitrate Chemical compound [Ga+3].[O-][N+]([O-])=O.[O-][N+]([O-])=O.[O-][N+]([O-])=O CHPZKNULDCNCBW-UHFFFAOYSA-N 0.000 description 2
- PUBCCFNQJQKCNC-XKNFJVFFSA-N gastrin-releasingpeptide Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(N)=O)NC(=O)CNC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H](CC(N)=O)NC(=O)CNC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)CNC(=O)[C@H](C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](N)C(C)C)[C@@H](C)O)C(C)C)[C@@H](C)O)C(C)C)C1=CNC=N1 PUBCCFNQJQKCNC-XKNFJVFFSA-N 0.000 description 2
- 229930182470 glycoside Natural products 0.000 description 2
- 150000002338 glycosides Chemical class 0.000 description 2
- 229940035638 gonadotropin-releasing hormone Drugs 0.000 description 2
- 229960002913 goserelin Drugs 0.000 description 2
- OBFGRABVBUQJNE-UHFFFAOYSA-N griffithine Chemical compound C1C2CN(C=O)CC1CN1C(=O)C=CC=C1C(C1)CN(C=O)CC1CN1C(=O)C=CC=C12 OBFGRABVBUQJNE-UHFFFAOYSA-N 0.000 description 2
- JYGXADMDTFJGBT-VWUMJDOOSA-N hydrocortisone Chemical compound O=C1CC[C@]2(C)[C@H]3[C@@H](O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 JYGXADMDTFJGBT-VWUMJDOOSA-N 0.000 description 2
- 229960000908 idarubicin Drugs 0.000 description 2
- 239000002596 immunotoxin Substances 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000003780 insertion Methods 0.000 description 2
- 230000037431 insertion Effects 0.000 description 2
- VBUWHHLIZKOSMS-RIWXPGAOSA-N invicorp Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(N)=O)C(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCCN)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)C(C)C)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=C(O)C=C1 VBUWHHLIZKOSMS-RIWXPGAOSA-N 0.000 description 2
- 229960003350 isoniazid Drugs 0.000 description 2
- QRXWMOHMRWLFEY-UHFFFAOYSA-N isoniazide Chemical compound NNC(=O)C1=CC=NC=C1 QRXWMOHMRWLFEY-UHFFFAOYSA-N 0.000 description 2
- 108010021336 lanreotide Proteins 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 229940040129 luteinizing hormone Drugs 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- HAWPXGHAZFHHAD-UHFFFAOYSA-N mechlorethamine Chemical class ClCCN(C)CCCl HAWPXGHAZFHHAD-UHFFFAOYSA-N 0.000 description 2
- 229960004961 mechlorethamine Drugs 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 2
- 229960000513 necitumumab Drugs 0.000 description 2
- 229960000808 netilmicin Drugs 0.000 description 2
- ZBGPYVZLYBDXKO-HILBYHGXSA-N netilmycin Chemical compound O([C@@H]1[C@@H](N)C[C@H]([C@@H]([C@H]1O)O[C@@H]1[C@]([C@H](NC)[C@@H](O)CO1)(C)O)NCC)[C@H]1OC(CN)=CC[C@H]1N ZBGPYVZLYBDXKO-HILBYHGXSA-N 0.000 description 2
- NQDJXKOVJZTUJA-UHFFFAOYSA-N nevirapine Chemical compound C12=NC=CC=C2C(=O)NC=2C(C)=CC=NC=2N1C1CC1 NQDJXKOVJZTUJA-UHFFFAOYSA-N 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- AICOOMRHRUFYCM-ZRRPKQBOSA-N oxazine, 1 Chemical compound C([C@@H]1[C@H](C(C[C@]2(C)[C@@H]([C@H](C)N(C)C)[C@H](O)C[C@]21C)=O)CC1=CC2)C[C@H]1[C@@]1(C)[C@H]2N=C(C(C)C)OC1 AICOOMRHRUFYCM-ZRRPKQBOSA-N 0.000 description 2
- 229910052760 oxygen Inorganic materials 0.000 description 2
- LSQZJLSUYDQPKJ-UHFFFAOYSA-N p-Hydroxyampicillin Natural products O=C1N2C(C(O)=O)C(C)(C)SC2C1NC(=O)C(N)C1=CC=C(O)C=C1 LSQZJLSUYDQPKJ-UHFFFAOYSA-N 0.000 description 2
- 108010089193 pattern recognition receptors Proteins 0.000 description 2
- 102000007863 pattern recognition receptors Human genes 0.000 description 2
- 229940056367 penicillin v Drugs 0.000 description 2
- 239000008194 pharmaceutical composition Substances 0.000 description 2
- 239000012071 phase Substances 0.000 description 2
- BPLBGHOLXOTWMN-MBNYWOFBSA-N phenoxymethylpenicillin Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)COC1=CC=CC=C1 BPLBGHOLXOTWMN-MBNYWOFBSA-N 0.000 description 2
- IEQIEDJGQAUEQZ-UHFFFAOYSA-N phthalocyanine Chemical compound N1C(N=C2C3=CC=CC=C3C(N=C3C4=CC=CC=C4C(=N4)N3)=N2)=C(C=CC=C2)C2=C1N=C1C2=CC=CC=C2C4=N1 IEQIEDJGQAUEQZ-UHFFFAOYSA-N 0.000 description 2
- 210000002306 phycobilisome Anatomy 0.000 description 2
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 description 2
- 238000000746 purification Methods 0.000 description 2
- RXWNCPJZOCPEPQ-NVWDDTSBSA-N puromycin Chemical compound C1=CC(OC)=CC=C1C[C@H](N)C(=O)N[C@H]1[C@@H](O)[C@H](N2C3=NC=NC(=C3N=C2)N(C)C)O[C@@H]1CO RXWNCPJZOCPEPQ-NVWDDTSBSA-N 0.000 description 2
- 229940052337 quinupristin/dalfopristin Drugs 0.000 description 2
- 108010054624 red fluorescent protein Proteins 0.000 description 2
- ATEBXHFBFRCZMA-VXTBVIBXSA-N rifabutin Chemical compound O([C@](C1=O)(C)O/C=C/[C@@H]([C@H]([C@@H](OC(C)=O)[C@H](C)[C@H](O)[C@H](C)[C@@H](O)[C@@H](C)\C=C\C=C(C)/C(=O)NC(=C2N3)C(=O)C=4C(O)=C5C)C)OC)C5=C1C=4C2=NC13CCN(CC(C)C)CC1 ATEBXHFBFRCZMA-VXTBVIBXSA-N 0.000 description 2
- 229950009213 rubitecan Drugs 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- 239000000741 silica gel Substances 0.000 description 2
- 229910002027 silica gel Inorganic materials 0.000 description 2
- 229960003787 sorafenib Drugs 0.000 description 2
- 229960000268 spectinomycin Drugs 0.000 description 2
- UNFWWIHTNXNPBV-WXKVUWSESA-N spectinomycin Chemical compound O([C@@H]1[C@@H](NC)[C@@H](O)[C@H]([C@@H]([C@H]1O1)O)NC)[C@]2(O)[C@H]1O[C@H](C)CC2=O UNFWWIHTNXNPBV-WXKVUWSESA-N 0.000 description 2
- 150000003431 steroids Chemical class 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 229960005322 streptomycin Drugs 0.000 description 2
- 229940124530 sulfonamide Drugs 0.000 description 2
- SUVMJBTUFCVSAD-UHFFFAOYSA-N sulforaphane Chemical compound CS(=O)CCCCN=C=S SUVMJBTUFCVSAD-UHFFFAOYSA-N 0.000 description 2
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 2
- 229960003250 telithromycin Drugs 0.000 description 2
- LJVAJPDWBABPEJ-PNUFFHFMSA-N telithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)[C@@H](C)C(=O)O[C@@H]([C@]2(OC(=O)N(CCCCN3C=C(N=C3)C=3C=NC=CC=3)[C@@H]2[C@@H](C)C(=O)[C@H](C)C[C@@]1(C)OC)C)CC)[C@@H]1O[C@H](C)C[C@H](N(C)C)[C@H]1O LJVAJPDWBABPEJ-PNUFFHFMSA-N 0.000 description 2
- 125000004213 tert-butoxy group Chemical group [H]C([H])([H])C(O*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 2
- 235000019364 tetracycline Nutrition 0.000 description 2
- 150000003522 tetracyclines Chemical class 0.000 description 2
- MPLHNVLQVRSVEE-UHFFFAOYSA-N texas red Chemical compound [O-]S(=O)(=O)C1=CC(S(Cl)(=O)=O)=CC=C1C(C1=CC=2CCCN3CCCC(C=23)=C1O1)=C2C1=C(CCC1)C3=[N+]1CCCC3=C2 MPLHNVLQVRSVEE-UHFFFAOYSA-N 0.000 description 2
- 229960000707 tobramycin Drugs 0.000 description 2
- NLVFBUXFDBBNBW-PBSUHMDJSA-N tobramycin Chemical compound N[C@@H]1C[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N NLVFBUXFDBBNBW-PBSUHMDJSA-N 0.000 description 2
- 231100000331 toxic Toxicity 0.000 description 2
- 230000002588 toxic effect Effects 0.000 description 2
- 230000001988 toxicity Effects 0.000 description 2
- 231100000419 toxicity Toxicity 0.000 description 2
- 238000000108 ultra-filtration Methods 0.000 description 2
- QYSXJUFSXHHAJI-YRZJJWOYSA-N vitamin D3 Chemical class C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C\C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-YRZJJWOYSA-N 0.000 description 2
- 229960002555 zidovudine Drugs 0.000 description 2
- HBOMLICNUCNMMY-XLPZGREQSA-N zidovudine Chemical compound O=C1NC(=O)C(C)=CN1[C@@H]1O[C@H](CO)[C@@H](N=[N+]=[N-])C1 HBOMLICNUCNMMY-XLPZGREQSA-N 0.000 description 2
- FBTUMDXHSRTGRV-ALTNURHMSA-N zorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(\C)=N\NC(=O)C=1C=CC=CC=1)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 FBTUMDXHSRTGRV-ALTNURHMSA-N 0.000 description 2
- 229960000641 zorubicin Drugs 0.000 description 2
- WHNFPRLDDSXQCL-UAZQEYIDSA-N α-msh Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1NC=NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)NCC(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C(C)C)C(N)=O)NC(=O)[C@H](CO)NC(C)=O)C1=CC=C(O)C=C1 WHNFPRLDDSXQCL-UAZQEYIDSA-N 0.000 description 2
- AYIRNRDRBQJXIF-NXEZZACHSA-N (-)-Florfenicol Chemical compound CS(=O)(=O)C1=CC=C([C@@H](O)[C@@H](CF)NC(=O)C(Cl)Cl)C=C1 AYIRNRDRBQJXIF-NXEZZACHSA-N 0.000 description 1
- LVLLALCJVJNGQQ-SEODYNFXSA-N (1r,3s,5z)-5-[(2e)-2-[(1r,3as,7ar)-1-[(2r,3e,5e)-7-ethyl-7-hydroxynona-3,5-dien-2-yl]-7a-methyl-2,3,3a,5,6,7-hexahydro-1h-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)/C=C/C=C/C(O)(CC)CC)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C LVLLALCJVJNGQQ-SEODYNFXSA-N 0.000 description 1
- KLZOTDOJMRMLDX-YBBVPDDNSA-N (1r,3s,5z)-5-[(2e)-2-[(1s,3as,7as)-1-[(1r)-1-(4-ethyl-4-hydroxyhexoxy)ethyl]-7a-methyl-2,3,3a,5,6,7-hexahydro-1h-inden-4-ylidene]ethylidene]-4-methylidenecyclohexane-1,3-diol Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@@H](C)OCCCC(O)(CC)CC)=C\C=C1\C[C@@H](O)C[C@H](O)C1=C KLZOTDOJMRMLDX-YBBVPDDNSA-N 0.000 description 1
- DENYZIUJOTUUNY-MRXNPFEDSA-N (2R)-14-fluoro-2-methyl-6,9,10,19-tetrazapentacyclo[14.2.1.02,6.08,18.012,17]nonadeca-1(18),8,12(17),13,15-pentaen-11-one Chemical compound FC=1C=C2C=3C=4C(CN5[C@@](C4NC3C1)(CCC5)C)=NNC2=O DENYZIUJOTUUNY-MRXNPFEDSA-N 0.000 description 1
- FWFGIHPGRQZWIW-SQNIBIBYSA-N (2S)-2-[[(2R)-2-[(1S)-1-hydroxy-2-(hydroxyamino)-2-oxoethyl]-4-methyl-1-oxopentyl]amino]-2-phenylacetic acid cyclopentyl ester Chemical compound O=C([C@@H](NC(=O)[C@@H]([C@H](O)C(=O)NO)CC(C)C)C=1C=CC=CC=1)OC1CCCC1 FWFGIHPGRQZWIW-SQNIBIBYSA-N 0.000 description 1
- QDZOEBFLNHCSSF-PFFBOGFISA-N (2S)-2-[[(2R)-2-[[(2S)-1-[(2S)-6-amino-2-[[(2S)-1-[(2R)-2-amino-5-carbamimidamidopentanoyl]pyrrolidine-2-carbonyl]amino]hexanoyl]pyrrolidine-2-carbonyl]amino]-3-(1H-indol-3-yl)propanoyl]amino]-N-[(2R)-1-[[(2S)-1-[[(2R)-1-[[(2S)-1-[[(2S)-1-amino-4-methyl-1-oxopentan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-3-(1H-indol-3-yl)-1-oxopropan-2-yl]pentanediamide Chemical compound C([C@@H](C(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(N)=O)NC(=O)[C@@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCCCN)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](N)CCCNC(N)=N)C1=CC=CC=C1 QDZOEBFLNHCSSF-PFFBOGFISA-N 0.000 description 1
- DQJCDTNMLBYVAY-ZXXIYAEKSA-N (2S,5R,10R,13R)-16-{[(2R,3S,4R,5R)-3-{[(2S,3R,4R,5S,6R)-3-acetamido-4,5-dihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy}-5-(ethylamino)-6-hydroxy-2-(hydroxymethyl)oxan-4-yl]oxy}-5-(4-aminobutyl)-10-carbamoyl-2,13-dimethyl-4,7,12,15-tetraoxo-3,6,11,14-tetraazaheptadecan-1-oic acid Chemical compound NCCCC[C@H](C(=O)N[C@@H](C)C(O)=O)NC(=O)CC[C@H](C(N)=O)NC(=O)[C@@H](C)NC(=O)C(C)O[C@@H]1[C@@H](NCC)C(O)O[C@H](CO)[C@H]1O[C@H]1[C@H](NC(C)=O)[C@@H](O)[C@H](O)[C@@H](CO)O1 DQJCDTNMLBYVAY-ZXXIYAEKSA-N 0.000 description 1
- UDIIBEDMEYAVNG-CORWDWJASA-N (2s)-3-amino-n-[(1r,2s,3s,4r,5s)-5-amino-4-[(3r,4s,5s,6r)-6-(aminomethyl)-3,4,5-trihydroxyoxan-2-yl]oxy-2-[(3r,4r,5r)-3,5-dihydroxy-5-methyl-4-(methylamino)oxan-2-yl]oxy-3-hydroxycyclohexyl]-2-hydroxypropanamide Chemical compound O1C[C@@](O)(C)[C@H](NC)[C@@H](O)C1O[C@@H]1[C@@H](O)[C@H](OC2[C@@H]([C@@H](O)[C@H](O)[C@@H](CN)O2)O)[C@@H](N)C[C@H]1NC(=O)[C@@H](O)CN UDIIBEDMEYAVNG-CORWDWJASA-N 0.000 description 1
- ZRVZOBGMZWVJOS-VMXHOPILSA-N (2s)-6-amino-2-[[(2s)-1-[(2s)-2-[[(2s)-2-[[2-[[(2s)-2-[(2-aminoacetyl)amino]-5-(diaminomethylideneamino)pentanoyl]amino]acetyl]amino]-3-carboxypropanoyl]amino]-3-hydroxypropanoyl]pyrrolidine-2-carbonyl]amino]hexanoic acid Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)[C@@H]1CCCN1C(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CCCN=C(N)N)NC(=O)CN ZRVZOBGMZWVJOS-VMXHOPILSA-N 0.000 description 1
- TVIRNGFXQVMMGB-OFWIHYRESA-N (3s,6r,10r,13e,16s)-16-[(2r,3r,4s)-4-chloro-3-hydroxy-4-phenylbutan-2-yl]-10-[(3-chloro-4-methoxyphenyl)methyl]-6-methyl-3-(2-methylpropyl)-1,4-dioxa-8,11-diazacyclohexadec-13-ene-2,5,9,12-tetrone Chemical compound C1=C(Cl)C(OC)=CC=C1C[C@@H]1C(=O)NC[C@@H](C)C(=O)O[C@@H](CC(C)C)C(=O)O[C@H]([C@H](C)[C@@H](O)[C@@H](Cl)C=2C=CC=CC=2)C/C=C/C(=O)N1 TVIRNGFXQVMMGB-OFWIHYRESA-N 0.000 description 1
- OFPZNTXZCGKCMU-VXBOPZJTSA-N (3z,5e,7r,8s,10s,11z,13s,14r,15s,17s,20r,21s,22s)-22-[(2s,3z)-hexa-3,5-dien-2-yl]-8,10,14,20-tetrahydroxy-7,13,15,17,21-pentamethyl-1-oxacyclodocosa-3,5,11-trien-2-one Chemical compound C=C\C=C/[C@H](C)[C@@H]1OC(=O)\C=C/C=C/[C@@H](C)[C@@H](O)C[C@H](O)\C=C/[C@H](C)[C@H](O)[C@@H](C)C[C@@H](C)CC[C@@H](O)[C@@H]1C OFPZNTXZCGKCMU-VXBOPZJTSA-N 0.000 description 1
- CNPVJJQCETWNEU-CYFREDJKSA-N (4,6-dimethyl-5-pyrimidinyl)-[4-[(3S)-4-[(1R)-2-methoxy-1-[4-(trifluoromethyl)phenyl]ethyl]-3-methyl-1-piperazinyl]-4-methyl-1-piperidinyl]methanone Chemical compound N([C@@H](COC)C=1C=CC(=CC=1)C(F)(F)F)([C@H](C1)C)CCN1C(CC1)(C)CCN1C(=O)C1=C(C)N=CN=C1C CNPVJJQCETWNEU-CYFREDJKSA-N 0.000 description 1
- PUDHBTGHUJUUFI-SCTWWAJVSA-N (4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s,3r)-1-amino-3-hydroxy-1-oxobutan-2-yl]-19-[[(2r)-2-amino-3-naphthalen-2-ylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5,8,11,14,17-p Chemical compound C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 PUDHBTGHUJUUFI-SCTWWAJVSA-N 0.000 description 1
- XIYOPDCBBDCGOE-IWVLMIASSA-N (4s,4ar,5s,5ar,12ar)-4-(dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methylidene-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carboxamide Chemical compound C=C1C2=CC=CC(O)=C2C(O)=C2[C@@H]1[C@H](O)[C@H]1[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]1(O)C2=O XIYOPDCBBDCGOE-IWVLMIASSA-N 0.000 description 1
- SGKRLCUYIXIAHR-AKNGSSGZSA-N (4s,4ar,5s,5ar,6r,12ar)-4-(dimethylamino)-1,5,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1=CC=C2[C@H](C)[C@@H]([C@H](O)[C@@H]3[C@](C(O)=C(C(N)=O)C(=O)[C@H]3N(C)C)(O)C3=O)C3=C(O)C2=C1O SGKRLCUYIXIAHR-AKNGSSGZSA-N 0.000 description 1
- FFTVPQUHLQBXQZ-KVUCHLLUSA-N (4s,4as,5ar,12ar)-4,7-bis(dimethylamino)-1,10,11,12a-tetrahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1C2=C(N(C)C)C=CC(O)=C2C(O)=C2[C@@H]1C[C@H]1[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]1(O)C2=O FFTVPQUHLQBXQZ-KVUCHLLUSA-N 0.000 description 1
- RMVMLZHPWMTQGK-SOUFLCLCSA-N (4s,4as,5as,6s,12ar)-4-(dimethylamino)-1,6,10,11,12a-pentahydroxy-3,12-dioxo-4a,5,5a,6-tetrahydro-4h-tetracene-2-carboxamide Chemical compound C1([C@H]2O)=CC=CC(O)=C1C(O)=C1[C@@H]2C[C@H]2[C@H](N(C)C)C(=O)C(C(N)=O)=C(O)[C@@]2(O)C1=O RMVMLZHPWMTQGK-SOUFLCLCSA-N 0.000 description 1
- XRBSKUSTLXISAB-XVVDYKMHSA-N (5r,6r,7r,8r)-8-hydroxy-7-(hydroxymethyl)-5-(3,4,5-trimethoxyphenyl)-5,6,7,8-tetrahydrobenzo[f][1,3]benzodioxole-6-carboxylic acid Chemical compound COC1=C(OC)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@H](O)[C@@H](CO)[C@@H]2C(O)=O)=C1 XRBSKUSTLXISAB-XVVDYKMHSA-N 0.000 description 1
- FPVKHBSQESCIEP-UHFFFAOYSA-N (8S)-3-(2-deoxy-beta-D-erythro-pentofuranosyl)-3,6,7,8-tetrahydroimidazo[4,5-d][1,3]diazepin-8-ol Natural products C1C(O)C(CO)OC1N1C(NC=NCC2O)=C2N=C1 FPVKHBSQESCIEP-UHFFFAOYSA-N 0.000 description 1
- 125000004400 (C1-C12) alkyl group Chemical group 0.000 description 1
- 125000003161 (C1-C6) alkylene group Chemical group 0.000 description 1
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 description 1
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 description 1
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 description 1
- MINDHVHHQZYEEK-UHFFFAOYSA-N (E)-(2S,3R,4R,5S)-5-[(2S,3S,4S,5S)-2,3-epoxy-5-hydroxy-4-methylhexyl]tetrahydro-3,4-dihydroxy-(beta)-methyl-2H-pyran-2-crotonic acid ester with 9-hydroxynonanoic acid Natural products CC(O)C(C)C1OC1CC1C(O)C(O)C(CC(C)=CC(=O)OCCCCCCCCC(O)=O)OC1 MINDHVHHQZYEEK-UHFFFAOYSA-N 0.000 description 1
- RXZBMPWDPOLZGW-XMRMVWPWSA-N (E)-roxithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=N/OCOCCOC)/[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 RXZBMPWDPOLZGW-XMRMVWPWSA-N 0.000 description 1
- LKJPYSCBVHEWIU-KRWDZBQOSA-N (R)-bicalutamide Chemical compound C([C@@](O)(C)C(=O)NC=1C=C(C(C#N)=CC=1)C(F)(F)F)S(=O)(=O)C1=CC=C(F)C=C1 LKJPYSCBVHEWIU-KRWDZBQOSA-N 0.000 description 1
- QKDHBVNJCZBTMR-LLVKDONJSA-N (R)-temafloxacin Chemical compound C1CN[C@H](C)CN1C(C(=C1)F)=CC2=C1C(=O)C(C(O)=O)=CN2C1=CC=C(F)C=C1F QKDHBVNJCZBTMR-LLVKDONJSA-N 0.000 description 1
- LAMIXXKAWNLXOC-INIZCTEOSA-N (S)-HDAC-42 Chemical compound O=C([C@@H](C(C)C)C=1C=CC=CC=1)NC1=CC=C(C(=O)NO)C=C1 LAMIXXKAWNLXOC-INIZCTEOSA-N 0.000 description 1
- WHTVZRBIWZFKQO-AWEZNQCLSA-N (S)-chloroquine Chemical compound ClC1=CC=C2C(N[C@@H](C)CCCN(CC)CC)=CC=NC2=C1 WHTVZRBIWZFKQO-AWEZNQCLSA-N 0.000 description 1
- XUBOMFCQGDBHNK-JTQLQIEISA-N (S)-gatifloxacin Chemical compound FC1=CC(C(C(C(O)=O)=CN2C3CC3)=O)=C2C(OC)=C1N1CCN[C@@H](C)C1 XUBOMFCQGDBHNK-JTQLQIEISA-N 0.000 description 1
- AGNGYMCLFWQVGX-AGFFZDDWSA-N (e)-1-[(2s)-2-amino-2-carboxyethoxy]-2-diazonioethenolate Chemical compound OC(=O)[C@@H](N)CO\C([O-])=C\[N+]#N AGNGYMCLFWQVGX-AGFFZDDWSA-N 0.000 description 1
- NCCJWSXETVVUHK-ZYSAIPPVSA-N (z)-7-[(2r)-2-amino-2-carboxyethyl]sulfanyl-2-[[(1s)-2,2-dimethylcyclopropanecarbonyl]amino]hept-2-enoic acid;(5r,6s)-3-[2-(aminomethylideneamino)ethylsulfanyl]-6-[(1r)-1-hydroxyethyl]-7-oxo-1-azabicyclo[3.2.0]hept-2-ene-2-carboxylic acid Chemical compound C1C(SCC\N=C/N)=C(C(O)=O)N2C(=O)[C@H]([C@H](O)C)[C@H]21.CC1(C)C[C@@H]1C(=O)N\C(=C/CCCCSC[C@H](N)C(O)=O)C(O)=O NCCJWSXETVVUHK-ZYSAIPPVSA-N 0.000 description 1
- SLLFVLKNXABYGI-UHFFFAOYSA-N 1,2,3-benzoxadiazole Chemical compound C1=CC=C2ON=NC2=C1 SLLFVLKNXABYGI-UHFFFAOYSA-N 0.000 description 1
- UBCHPRBFMUDMNC-UHFFFAOYSA-N 1-(1-adamantyl)ethanamine Chemical compound C1C(C2)CC3CC2CC1(C(N)C)C3 UBCHPRBFMUDMNC-UHFFFAOYSA-N 0.000 description 1
- SWQQELWGJDXCFT-PNHWDRBUSA-N 1-[(2r,3r,4s,5r)-3,4-dihydroxy-5-(hydroxymethyl)oxolan-2-yl]-5-ethynylimidazole-4-carboxamide Chemical compound C#CC1=C(C(=O)N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 SWQQELWGJDXCFT-PNHWDRBUSA-N 0.000 description 1
- WKBPZYKAUNRMKP-UHFFFAOYSA-N 1-[2-(2,4-dichlorophenyl)pentyl]1,2,4-triazole Chemical compound C=1C=C(Cl)C=C(Cl)C=1C(CCC)CN1C=NC=N1 WKBPZYKAUNRMKP-UHFFFAOYSA-N 0.000 description 1
- SPMVMDHWKHCIDT-UHFFFAOYSA-N 1-[2-chloro-4-[(6,7-dimethoxy-4-quinolinyl)oxy]phenyl]-3-(5-methyl-3-isoxazolyl)urea Chemical compound C=12C=C(OC)C(OC)=CC2=NC=CC=1OC(C=C1Cl)=CC=C1NC(=O)NC=1C=C(C)ON=1 SPMVMDHWKHCIDT-UHFFFAOYSA-N 0.000 description 1
- CRSBERNSMYQZNG-UHFFFAOYSA-N 1-dodecene Chemical class CCCCCCCCCCC=C CRSBERNSMYQZNG-UHFFFAOYSA-N 0.000 description 1
- HAWSQZCWOQZXHI-FQEVSTJZSA-N 10-Hydroxycamptothecin Chemical compound C1=C(O)C=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 HAWSQZCWOQZXHI-FQEVSTJZSA-N 0.000 description 1
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 description 1
- CTLOSZHDGZLOQE-UHFFFAOYSA-N 14-methoxy-9-[(4-methylpiperazin-1-yl)methyl]-9,19-diazapentacyclo[10.7.0.02,6.07,11.013,18]nonadeca-1(12),2(6),7(11),13(18),14,16-hexaene-8,10-dione Chemical compound O=C1C2=C3C=4C(OC)=CC=CC=4NC3=C3CCCC3=C2C(=O)N1CN1CCN(C)CC1 CTLOSZHDGZLOQE-UHFFFAOYSA-N 0.000 description 1
- HYZJCKYKOHLVJF-UHFFFAOYSA-N 1H-benzimidazole Chemical class C1=CC=C2NC=NC2=C1 HYZJCKYKOHLVJF-UHFFFAOYSA-N 0.000 description 1
- VGIRNWJSIRVFRT-UHFFFAOYSA-N 2',7'-difluorofluorescein Chemical compound OC(=O)C1=CC=CC=C1C1=C2C=C(F)C(=O)C=C2OC2=CC(O)=C(F)C=C21 VGIRNWJSIRVFRT-UHFFFAOYSA-N 0.000 description 1
- PRDFBSVERLRRMY-UHFFFAOYSA-N 2'-(4-ethoxyphenyl)-5-(4-methylpiperazin-1-yl)-2,5'-bibenzimidazole Chemical compound C1=CC(OCC)=CC=C1C1=NC2=CC=C(C=3NC4=CC(=CC=C4N=3)N3CCN(C)CC3)C=C2N1 PRDFBSVERLRRMY-UHFFFAOYSA-N 0.000 description 1
- BTOTXLJHDSNXMW-POYBYMJQSA-N 2,3-dideoxyuridine Chemical compound O1[C@H](CO)CC[C@@H]1N1C(=O)NC(=O)C=C1 BTOTXLJHDSNXMW-POYBYMJQSA-N 0.000 description 1
- YMHOBZXQZVXHBM-UHFFFAOYSA-N 2,5-dimethoxy-4-bromophenethylamine Chemical compound COC1=CC(CCN)=C(OC)C=C1Br YMHOBZXQZVXHBM-UHFFFAOYSA-N 0.000 description 1
- UEJJHQNACJXSKW-UHFFFAOYSA-N 2-(2,6-dioxopiperidin-3-yl)-1H-isoindole-1,3(2H)-dione Chemical compound O=C1C2=CC=CC=C2C(=O)N1C1CCC(=O)NC1=O UEJJHQNACJXSKW-UHFFFAOYSA-N 0.000 description 1
- KZDCMKVLEYCGQX-UDPGNSCCSA-N 2-(diethylamino)ethyl 4-aminobenzoate;(2s,5r,6r)-3,3-dimethyl-7-oxo-6-[(2-phenylacetyl)amino]-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid;hydrate Chemical compound O.CCN(CC)CCOC(=O)C1=CC=C(N)C=C1.N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 KZDCMKVLEYCGQX-UDPGNSCCSA-N 0.000 description 1
- IZXIZTKNFFYFOF-UHFFFAOYSA-N 2-Oxazolidone Chemical class O=C1NCCO1 IZXIZTKNFFYFOF-UHFFFAOYSA-N 0.000 description 1
- IMSODMZESSGVBE-UHFFFAOYSA-N 2-Oxazoline Chemical compound C1CN=CO1 IMSODMZESSGVBE-UHFFFAOYSA-N 0.000 description 1
- ACTOXUHEUCPTEW-BWHGAVFKSA-N 2-[(4r,5s,6s,7r,9r,10r,11e,13e,16r)-6-[(2s,3r,4r,5s,6r)-5-[(2s,4r,5s,6s)-4,5-dihydroxy-4,6-dimethyloxan-2-yl]oxy-4-(dimethylamino)-3-hydroxy-6-methyloxan-2-yl]oxy-10-[(2s,5s,6r)-5-(dimethylamino)-6-methyloxan-2-yl]oxy-4-hydroxy-5-methoxy-9,16-dimethyl-2-o Chemical compound O([C@H]1/C=C/C=C/C[C@@H](C)OC(=O)C[C@@H](O)[C@@H]([C@H]([C@@H](CC=O)C[C@H]1C)O[C@H]1[C@@H]([C@H]([C@H](O[C@@H]2O[C@@H](C)[C@H](O)[C@](C)(O)C2)[C@@H](C)O1)N(C)C)O)OC)[C@@H]1CC[C@H](N(C)C)[C@@H](C)O1 ACTOXUHEUCPTEW-BWHGAVFKSA-N 0.000 description 1
- IOOMXAQUNPWDLL-UHFFFAOYSA-N 2-[6-(diethylamino)-3-(diethyliminiumyl)-3h-xanthen-9-yl]-5-sulfobenzene-1-sulfonate Chemical compound C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=C(S(O)(=O)=O)C=C1S([O-])(=O)=O IOOMXAQUNPWDLL-UHFFFAOYSA-N 0.000 description 1
- FIDMEHCRMLKKPZ-YSMBQZINSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;[2-methoxy-5-[(z)-2-(3,4,5-trimethoxyphenyl)ethenyl]phenyl] dihydrogen phosphate Chemical compound OCC(N)(CO)CO.C1=C(OP(O)(O)=O)C(OC)=CC=C1\C=C/C1=CC(OC)=C(OC)C(OC)=C1 FIDMEHCRMLKKPZ-YSMBQZINSA-N 0.000 description 1
- RTJUXLYUUDBAJN-KVQBGUIXSA-N 2-amino-9-[(2r,4s,5r)-4-fluoro-5-(hydroxymethyl)oxolan-2-yl]-3h-purin-6-one Chemical compound C1=2NC(N)=NC(=O)C=2N=CN1[C@H]1C[C@H](F)[C@@H](CO)O1 RTJUXLYUUDBAJN-KVQBGUIXSA-N 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000003229 2-methylhexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- CTRPRMNBTVRDFH-UHFFFAOYSA-N 2-n-methyl-1,3,5-triazine-2,4,6-triamine Chemical compound CNC1=NC(N)=NC(N)=N1 CTRPRMNBTVRDFH-UHFFFAOYSA-N 0.000 description 1
- JVDYWBQJOUXQBB-UHFFFAOYSA-N 2-nitro-1,3-benzoxazole Chemical compound C1=CC=C2OC([N+](=O)[O-])=NC2=C1 JVDYWBQJOUXQBB-UHFFFAOYSA-N 0.000 description 1
- BNBQQYFXBLBYJK-UHFFFAOYSA-N 2-pyridin-2-yl-1,3-oxazole Chemical compound C1=COC(C=2N=CC=CC=2)=N1 BNBQQYFXBLBYJK-UHFFFAOYSA-N 0.000 description 1
- RSEBUVRVKCANEP-UHFFFAOYSA-N 2-pyrroline Chemical compound C1CC=CN1 RSEBUVRVKCANEP-UHFFFAOYSA-N 0.000 description 1
- YIMDLWDNDGKDTJ-QLKYHASDSA-N 3'-deamino-3'-(3-cyanomorpholin-4-yl)doxorubicin Chemical compound N1([C@H]2C[C@@H](O[C@@H](C)[C@H]2O)O[C@H]2C[C@@](O)(CC=3C(O)=C4C(=O)C=5C=CC=C(C=5C(=O)C4=C(O)C=32)OC)C(=O)CO)CCOCC1C#N YIMDLWDNDGKDTJ-QLKYHASDSA-N 0.000 description 1
- MAIJHAHHYNROMG-UHFFFAOYSA-N 3,4,12-triazatetracyclo[9.7.0.02,8.013,18]octadeca-1,3,5,7,9,11,13,15,17-nonaene Chemical class C1=CN=NC2=C3C4=CC=CC=C4N=C3C=CC2=C1 MAIJHAHHYNROMG-UHFFFAOYSA-N 0.000 description 1
- PWMYMKOUNYTVQN-UHFFFAOYSA-N 3-(8,8-diethyl-2-aza-8-germaspiro[4.5]decan-2-yl)-n,n-dimethylpropan-1-amine Chemical compound C1C[Ge](CC)(CC)CCC11CN(CCCN(C)C)CC1 PWMYMKOUNYTVQN-UHFFFAOYSA-N 0.000 description 1
- MAUCONCHVWBMHK-UHFFFAOYSA-N 3-[(dimethylamino)methyl]-N-[2-[4-[(hydroxyamino)-oxomethyl]phenoxy]ethyl]-2-benzofurancarboxamide Chemical compound O1C2=CC=CC=C2C(CN(C)C)=C1C(=O)NCCOC1=CC=C(C(=O)NO)C=C1 MAUCONCHVWBMHK-UHFFFAOYSA-N 0.000 description 1
- GSCPDZHWVNUUFI-UHFFFAOYSA-N 3-aminobenzamide Chemical compound NC(=O)C1=CC=CC(N)=C1 GSCPDZHWVNUUFI-UHFFFAOYSA-N 0.000 description 1
- IHXWECHPYNPJRR-UHFFFAOYSA-N 3-hydroxycyclobut-2-en-1-one Chemical class OC1=CC(=O)C1 IHXWECHPYNPJRR-UHFFFAOYSA-N 0.000 description 1
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 1
- WDBQJSCPCGTAFG-QHCPKHFHSA-N 4,4-difluoro-N-[(1S)-3-[4-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)piperidin-1-yl]-1-pyridin-3-ylpropyl]cyclohexane-1-carboxamide Chemical compound FC1(CCC(CC1)C(=O)N[C@@H](CCN1CCC(CC1)N1C(=NN=C1C)C(C)C)C=1C=NC=CC=1)F WDBQJSCPCGTAFG-QHCPKHFHSA-N 0.000 description 1
- BWGRDBSNKQABCB-UHFFFAOYSA-N 4,4-difluoro-N-[3-[3-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)-8-azabicyclo[3.2.1]octan-8-yl]-1-thiophen-2-ylpropyl]cyclohexane-1-carboxamide Chemical compound CC(C)C1=NN=C(C)N1C1CC2CCC(C1)N2CCC(NC(=O)C1CCC(F)(F)CC1)C1=CC=CS1 BWGRDBSNKQABCB-UHFFFAOYSA-N 0.000 description 1
- MSTDXOZUKAQDRL-UHFFFAOYSA-N 4-Chromanone Chemical compound C1=CC=C2C(=O)CCOC2=C1 MSTDXOZUKAQDRL-UHFFFAOYSA-N 0.000 description 1
- SUVMJBTUFCVSAD-JTQLQIEISA-N 4-Methylsulfinylbutyl isothiocyanate Natural products C[S@](=O)CCCCN=C=S SUVMJBTUFCVSAD-JTQLQIEISA-N 0.000 description 1
- ZOPBZHLJXQAQON-VWLOTQADSA-N 4-[[(3s)-3-(dimethylamino)pyrrolidin-1-yl]methyl]-n-[4-methyl-3-[(5-pyrimidin-5-ylpyrimidin-2-yl)amino]phenyl]-3-(trifluoromethyl)benzamide Chemical compound C1[C@@H](N(C)C)CCN1CC1=CC=C(C(=O)NC=2C=C(NC=3N=CC(=CN=3)C=3C=NC=NC=3)C(C)=CC=2)C=C1C(F)(F)F ZOPBZHLJXQAQON-VWLOTQADSA-N 0.000 description 1
- HSBKFSPNDWWPSL-VDTYLAMSSA-N 4-amino-5-fluoro-1-[(2s,5r)-5-(hydroxymethyl)-2,5-dihydrofuran-2-yl]pyrimidin-2-one Chemical compound C1=C(F)C(N)=NC(=O)N1[C@@H]1C=C[C@H](CO)O1 HSBKFSPNDWWPSL-VDTYLAMSSA-N 0.000 description 1
- MDOJTZQKHMAPBK-UHFFFAOYSA-N 4-iodo-3-nitrobenzamide Chemical compound NC(=O)C1=CC=C(I)C([N+]([O-])=O)=C1 MDOJTZQKHMAPBK-UHFFFAOYSA-N 0.000 description 1
- YVMBAUWDIGJRNY-BESUKNQGSA-N 4o8o7q7iu4 Chemical compound C1C(=O)C[C@H](O)\C=C(/C)\C=C\CNC(=O)\C=C\[C@@H](C)[C@@H](C(C)C)OC(=O)C2=CCCN2C(=O)C2=COC1=N2.N([C@@H]1C(=O)N[C@@H](C(N2CCC[C@H]2C(=O)N(C)[C@@H](CC=2C=CC(=CC=2)N(C)C)C(=O)N2CCC(=O)C[C@H]2C(=O)N[C@H](C(=O)O[C@@H]1C)C=1C=CC=CC=1)=O)CC)C(=O)C1=NC=CC=C1O YVMBAUWDIGJRNY-BESUKNQGSA-N 0.000 description 1
- NMUSYJAQQFHJEW-KVTDHHQDSA-N 5-azacytidine Chemical compound O=C1N=C(N)N=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](CO)O1 NMUSYJAQQFHJEW-KVTDHHQDSA-N 0.000 description 1
- WYWHKKSPHMUBEB-UHFFFAOYSA-N 6-Mercaptoguanine Natural products N1C(N)=NC(=S)C2=C1N=CN2 WYWHKKSPHMUBEB-UHFFFAOYSA-N 0.000 description 1
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 description 1
- HGGAKXAHAYOLDJ-FHZUQPTBSA-N 6alpha-[(R)-1-hydroxyethyl]-2-[(R)-tetrahydrofuran-2-yl]pen-2-em-3-carboxylic acid Chemical compound S([C@@H]1[C@H](C(N1C=1C(O)=O)=O)[C@H](O)C)C=1[C@H]1CCCO1 HGGAKXAHAYOLDJ-FHZUQPTBSA-N 0.000 description 1
- MPORYQCGWFQFLA-ONPDANIMSA-N 7-[(7s)-7-amino-5-azaspiro[2.4]heptan-5-yl]-8-chloro-6-fluoro-1-[(1r,2s)-2-fluorocyclopropyl]-4-oxoquinoline-3-carboxylic acid;trihydrate Chemical compound O.O.O.C([C@H]1N)N(C=2C(=C3C(C(C(C(O)=O)=CN3[C@H]3[C@H](C3)F)=O)=CC=2F)Cl)CC11CC1.C([C@H]1N)N(C=2C(=C3C(C(C(C(O)=O)=CN3[C@H]3[C@H](C3)F)=O)=CC=2F)Cl)CC11CC1 MPORYQCGWFQFLA-ONPDANIMSA-N 0.000 description 1
- PLIVFNIUGLLCEK-UHFFFAOYSA-N 7-[4-(3-ethynylanilino)-7-methoxyquinazolin-6-yl]oxy-n-hydroxyheptanamide Chemical compound C=12C=C(OCCCCCCC(=O)NO)C(OC)=CC2=NC=NC=1NC1=CC=CC(C#C)=C1 PLIVFNIUGLLCEK-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- ZGXJTSGNIOSYLO-UHFFFAOYSA-N 88755TAZ87 Chemical compound NCC(=O)CCC(O)=O ZGXJTSGNIOSYLO-UHFFFAOYSA-N 0.000 description 1
- GSDSWSVVBLHKDQ-UHFFFAOYSA-N 9-fluoro-3-methyl-10-(4-methylpiperazin-1-yl)-7-oxo-2,3-dihydro-7H-[1,4]oxazino[2,3,4-ij]quinoline-6-carboxylic acid Chemical compound FC1=CC(C(C(C(O)=O)=C2)=O)=C3N2C(C)COC3=C1N1CCN(C)CC1 GSDSWSVVBLHKDQ-UHFFFAOYSA-N 0.000 description 1
- QMGUSPDJTPDFSF-UHFFFAOYSA-N Aldophosphamide Chemical compound ClCCN(CCCl)P(=O)(N)OCCC=O QMGUSPDJTPDFSF-UHFFFAOYSA-N 0.000 description 1
- 239000012099 Alexa Fluor family Substances 0.000 description 1
- 241001083548 Anemone Species 0.000 description 1
- 108010049777 Ankyrins Proteins 0.000 description 1
- 102000008102 Ankyrins Human genes 0.000 description 1
- WZPBZJONDBGPKJ-UHFFFAOYSA-N Antibiotic SQ 26917 Natural products O=C1N(S(O)(=O)=O)C(C)C1NC(=O)C(=NOC(C)(C)C(O)=O)C1=CSC(N)=N1 WZPBZJONDBGPKJ-UHFFFAOYSA-N 0.000 description 1
- 108010032595 Antibody Binding Sites Proteins 0.000 description 1
- 101000772461 Arabidopsis thaliana Thioredoxin reductase 1, mitochondrial Proteins 0.000 description 1
- 101000772460 Arabidopsis thaliana Thioredoxin reductase 2 Proteins 0.000 description 1
- DDPXDCKYWDGZAL-BQBZGAKWSA-N Asn-Gly-Arg Chemical compound NC(=O)C[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N DDPXDCKYWDGZAL-BQBZGAKWSA-N 0.000 description 1
- AXRYRYVKAWYZBR-UHFFFAOYSA-N Atazanavir Natural products C=1C=C(C=2N=CC=CC=2)C=CC=1CN(NC(=O)C(NC(=O)OC)C(C)(C)C)CC(O)C(NC(=O)C(NC(=O)OC)C(C)(C)C)CC1=CC=CC=C1 AXRYRYVKAWYZBR-UHFFFAOYSA-N 0.000 description 1
- 108010019625 Atazanavir Sulfate Proteins 0.000 description 1
- NOWKCMXCCJGMRR-UHFFFAOYSA-N Aziridine Chemical compound C1CN1 NOWKCMXCCJGMRR-UHFFFAOYSA-N 0.000 description 1
- 108091005950 Azurite Proteins 0.000 description 1
- SPFYMRJSYKOXGV-UHFFFAOYSA-N Baytril Chemical compound C1CN(CC)CCN1C(C(=C1)F)=CC2=C1C(=O)C(C(O)=O)=CN2C1CC1 SPFYMRJSYKOXGV-UHFFFAOYSA-N 0.000 description 1
- KUVIULQEHSCUHY-XYWKZLDCSA-N Beclometasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(Cl)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)COC(=O)CC)(OC(=O)CC)[C@@]1(C)C[C@@H]2O KUVIULQEHSCUHY-XYWKZLDCSA-N 0.000 description 1
- VGGGPCQERPFHOB-MCIONIFRSA-N Bestatin Chemical compound CC(C)C[C@H](C(O)=O)NC(=O)[C@@H](O)[C@H](N)CC1=CC=CC=C1 VGGGPCQERPFHOB-MCIONIFRSA-N 0.000 description 1
- 108010051479 Bombesin Proteins 0.000 description 1
- 102000013585 Bombesin Human genes 0.000 description 1
- VOVIALXJUBGFJZ-KWVAZRHASA-N Budesonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@@H]2[C@@H]1[C@@H]1C[C@H]3OC(CCC)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O VOVIALXJUBGFJZ-KWVAZRHASA-N 0.000 description 1
- MBABCNBNDNGODA-LTGLSHGVSA-N Bullatacin Natural products O=C1C(C[C@H](O)CCCCCCCCCC[C@@H](O)[C@@H]2O[C@@H]([C@@H]3O[C@H]([C@@H](O)CCCCCCCCCC)CC3)CC2)=C[C@H](C)O1 MBABCNBNDNGODA-LTGLSHGVSA-N 0.000 description 1
- KGGVWMAPBXIMEM-ZRTAFWODSA-N Bullatacinone Chemical compound O1[C@@H]([C@@H](O)CCCCCCCCCC)CC[C@@H]1[C@@H]1O[C@@H]([C@H](O)CCCCCCCCCC[C@H]2OC(=O)[C@H](CC(C)=O)C2)CC1 KGGVWMAPBXIMEM-ZRTAFWODSA-N 0.000 description 1
- KGGVWMAPBXIMEM-JQFCFGFHSA-N Bullatacinone Natural products O=C(C[C@H]1C(=O)O[C@H](CCCCCCCCCC[C@H](O)[C@@H]2O[C@@H]([C@@H]3O[C@@H]([C@@H](O)CCCCCCCCCC)CC3)CC2)C1)C KGGVWMAPBXIMEM-JQFCFGFHSA-N 0.000 description 1
- 108010037003 Buserelin Proteins 0.000 description 1
- DGGZCXUXASNDAC-QQNGCVSVSA-N C-1027 chromophore Chemical compound COc1cc2OC(=C)C(=O)Nc2c(c1)C(=O)O[C@H]3COC(=O)C[C@H](N)c4cc(O)c(O[C@@H]5C#C\C=C\3/C#CC6=CC=C[C@]56O[C@@H]7OC(C)(C)[C@H]([C@@H](O)[C@H]7O)N(C)C)c(Cl)c4 DGGZCXUXASNDAC-QQNGCVSVSA-N 0.000 description 1
- 102000003930 C-Type Lectins Human genes 0.000 description 1
- 108090000342 C-Type Lectins Proteins 0.000 description 1
- 239000002126 C01EB10 - Adenosine Substances 0.000 description 1
- QAGYKUNXZHXKMR-UHFFFAOYSA-N CPD000469186 Natural products CC1=C(O)C=CC=C1C(=O)NC(C(O)CN1C(CC2CCCCC2C1)C(=O)NC(C)(C)C)CSC1=CC=CC=C1 QAGYKUNXZHXKMR-UHFFFAOYSA-N 0.000 description 1
- HAWSQZCWOQZXHI-UHFFFAOYSA-N CPT-OH Natural products C1=C(O)C=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)C5(O)CC)C4=NC2=C1 HAWSQZCWOQZXHI-UHFFFAOYSA-N 0.000 description 1
- 108010001789 Calcitonin Receptors Proteins 0.000 description 1
- 102100038520 Calcitonin receptor Human genes 0.000 description 1
- GAGWJHPBXLXJQN-UORFTKCHSA-N Capecitabine Chemical compound C1=C(F)C(NC(=O)OCCCCC)=NC(=O)N1[C@H]1[C@H](O)[C@H](O)[C@@H](C)O1 GAGWJHPBXLXJQN-UORFTKCHSA-N 0.000 description 1
- AOCCBINRVIKJHY-UHFFFAOYSA-N Carmofur Chemical compound CCCCCCNC(=O)N1C=C(F)C(=O)NC1=O AOCCBINRVIKJHY-UHFFFAOYSA-N 0.000 description 1
- DLGOEMSEDOSKAD-UHFFFAOYSA-N Carmustine Chemical compound ClCCNC(=O)N(N=O)CCCl DLGOEMSEDOSKAD-UHFFFAOYSA-N 0.000 description 1
- 102000004225 Cathepsin B Human genes 0.000 description 1
- 108090000712 Cathepsin B Proteins 0.000 description 1
- UQLLWWBDSUHNEB-CZUORRHYSA-N Cefaprin Chemical compound N([C@H]1[C@@H]2N(C1=O)C(=C(CS2)COC(=O)C)C(O)=O)C(=O)CSC1=CC=NC=C1 UQLLWWBDSUHNEB-CZUORRHYSA-N 0.000 description 1
- 108091005944 Cerulean Proteins 0.000 description 1
- JWBOIMRXGHLCPP-UHFFFAOYSA-N Chloditan Chemical compound C=1C=CC=C(Cl)C=1C(C(Cl)Cl)C1=CC=C(Cl)C=C1 JWBOIMRXGHLCPP-UHFFFAOYSA-N 0.000 description 1
- 241000579895 Chlorostilbon Species 0.000 description 1
- 108010089448 Cholecystokinin B Receptor Proteins 0.000 description 1
- 102100028757 Chondroitin sulfate proteoglycan 4 Human genes 0.000 description 1
- RURLVUZRUFHCJO-UHFFFAOYSA-N Chromomycin A3 Natural products COC(C1Cc2cc3cc(OC4CC(OC(=O)C)C(OC5CC(O)C(OC)C(C)O5)C(C)O4)c(C)c(O)c3c(O)c2C(=O)C1OC6CC(OC7CC(C)(O)C(OC(=O)C)C(C)O7)C(O)C(C)O6)C(=O)C(O)C(C)O RURLVUZRUFHCJO-UHFFFAOYSA-N 0.000 description 1
- VWFCHDSQECPREK-LURJTMIESA-N Cidofovir Chemical compound NC=1C=CN(C[C@@H](CO)OCP(O)(O)=O)C(=O)N=1 VWFCHDSQECPREK-LURJTMIESA-N 0.000 description 1
- CYSWUSAYJNCAKA-FYJFLYSWSA-N ClC1=C(C=CC=2N=C(SC=21)OCC)OC1=CC=C(C=N1)/C=C/[C@H](C)NC(C)=O Chemical compound ClC1=C(C=CC=2N=C(SC=21)OCC)OC1=CC=C(C=N1)/C=C/[C@H](C)NC(C)=O CYSWUSAYJNCAKA-FYJFLYSWSA-N 0.000 description 1
- HZZVJAQRINQKSD-UHFFFAOYSA-N Clavulanic acid Natural products OC(=O)C1C(=CCO)OC2CC(=O)N21 HZZVJAQRINQKSD-UHFFFAOYSA-N 0.000 description 1
- 108010078777 Colistin Proteins 0.000 description 1
- 208000035473 Communicable disease Diseases 0.000 description 1
- 229930188224 Cryptophycin Natural products 0.000 description 1
- 108091005943 CyPet Proteins 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- 101710112752 Cytotoxin Proteins 0.000 description 1
- DYDCUQKUCUHJBH-UWTATZPHSA-N D-Cycloserine Chemical compound N[C@@H]1CONC1=O DYDCUQKUCUHJBH-UWTATZPHSA-N 0.000 description 1
- DYDCUQKUCUHJBH-UHFFFAOYSA-N D-Cycloserine Natural products NC1CONC1=O DYDCUQKUCUHJBH-UHFFFAOYSA-N 0.000 description 1
- IGXWBGJHJZYPQS-SSDOTTSWSA-N D-Luciferin Chemical class OC(=O)[C@H]1CSC(C=2SC3=CC=C(O)C=C3N=2)=N1 IGXWBGJHJZYPQS-SSDOTTSWSA-N 0.000 description 1
- 108010013198 Daptomycin Proteins 0.000 description 1
- ZBNZXTGUTAYRHI-UHFFFAOYSA-N Dasatinib Chemical compound C=1C(N2CCN(CCO)CC2)=NC(C)=NC=1NC(S1)=NC=C1C(=O)NC1=C(C)C=CC=C1Cl ZBNZXTGUTAYRHI-UHFFFAOYSA-N 0.000 description 1
- OFPZNTXZCGKCMU-QUQSCIKMSA-N Dictyostatin 1 Natural products CC(C=C/C=C)C1OC(=O)C=C/C=C/C(C)C(O)CC(O)C=C/C(C)C(O)C(C)CC(C)CCC(O)C1C OFPZNTXZCGKCMU-QUQSCIKMSA-N 0.000 description 1
- BXZVVICBKDXVGW-NKWVEPMBSA-N Didanosine Chemical compound O1[C@H](CO)CC[C@@H]1N1C(NC=NC2=O)=C2N=C1 BXZVVICBKDXVGW-NKWVEPMBSA-N 0.000 description 1
- 102100024746 Dihydrofolate reductase Human genes 0.000 description 1
- 108010016626 Dipeptides Proteins 0.000 description 1
- JWCSIUVGFCSJCK-CAVRMKNVSA-N Disodium Moxalactam Chemical compound N([C@]1(OC)C(N2C(=C(CSC=3N(N=NN=3)C)CO[C@@H]21)C(O)=O)=O)C(=O)C(C(O)=O)C1=CC=C(O)C=C1 JWCSIUVGFCSJCK-CAVRMKNVSA-N 0.000 description 1
- SNRUBQQJIBEYMU-UHFFFAOYSA-N Dodecane Natural products CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 1
- 206010059866 Drug resistance Diseases 0.000 description 1
- 108091005941 EBFP Proteins 0.000 description 1
- 108091005947 EBFP2 Proteins 0.000 description 1
- 108091005942 ECFP Proteins 0.000 description 1
- 102000001301 EGF receptor Human genes 0.000 description 1
- 108060006698 EGF receptor Proteins 0.000 description 1
- XPOQHMRABVBWPR-UHFFFAOYSA-N Efavirenz Natural products O1C(=O)NC2=CC=C(Cl)C=C2C1(C(F)(F)F)C#CC1CC1 XPOQHMRABVBWPR-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 108010032976 Enfuvirtide Proteins 0.000 description 1
- 108010008165 Etanercept Proteins 0.000 description 1
- 108010067306 Fibronectins Proteins 0.000 description 1
- 102000016359 Fibronectins Human genes 0.000 description 1
- OZLGRUXZXMRXGP-UHFFFAOYSA-N Fluo-3 Chemical compound CC1=CC=C(N(CC(O)=O)CC(O)=O)C(OCCOC=2C(=CC=C(C=2)C2=C3C=C(Cl)C(=O)C=C3OC3=CC(O)=C(Cl)C=C32)N(CC(O)=O)CC(O)=O)=C1 OZLGRUXZXMRXGP-UHFFFAOYSA-N 0.000 description 1
- OUVXYXNWSVIOSJ-UHFFFAOYSA-N Fluo-4 Chemical compound CC1=CC=C(N(CC(O)=O)CC(O)=O)C(OCCOC=2C(=CC=C(C=2)C2=C3C=C(F)C(=O)C=C3OC3=CC(O)=C(F)C=C32)N(CC(O)=O)CC(O)=O)=C1 OUVXYXNWSVIOSJ-UHFFFAOYSA-N 0.000 description 1
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 description 1
- 102220566467 GDNF family receptor alpha-1_S65A_mutation Human genes 0.000 description 1
- 102220566469 GDNF family receptor alpha-1_S65T_mutation Human genes 0.000 description 1
- 102220566453 GDNF family receptor alpha-1_Y66F_mutation Human genes 0.000 description 1
- 102220566451 GDNF family receptor alpha-1_Y66H_mutation Human genes 0.000 description 1
- 102220566455 GDNF family receptor alpha-1_Y66W_mutation Human genes 0.000 description 1
- RVAQIUULWULRNW-UHFFFAOYSA-N Ganetespib Chemical compound C1=C(O)C(C(C)C)=CC(C=2N(C(O)=NN=2)C=2C=C3C=CN(C)C3=CC=2)=C1O RVAQIUULWULRNW-UHFFFAOYSA-N 0.000 description 1
- 102000052874 Gastrin receptors Human genes 0.000 description 1
- 229930182566 Gentamicin Natural products 0.000 description 1
- CEAZRRDELHUEMR-URQXQFDESA-N Gentamicin Chemical compound O1[C@H](C(C)NC)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](NC)[C@@](C)(O)CO2)O)[C@H](N)C[C@@H]1N CEAZRRDELHUEMR-URQXQFDESA-N 0.000 description 1
- 108010015899 Glycopeptides Proteins 0.000 description 1
- 102000002068 Glycopeptides Human genes 0.000 description 1
- 102400000932 Gonadoliberin-1 Human genes 0.000 description 1
- 101500026183 Homo sapiens Gonadoliberin-1 Proteins 0.000 description 1
- 101000666868 Homo sapiens Vasoactive intestinal polypeptide receptor 2 Proteins 0.000 description 1
- 108090000144 Human Proteins Proteins 0.000 description 1
- 102000003839 Human Proteins Human genes 0.000 description 1
- GRRNUXAQVGOGFE-UHFFFAOYSA-N Hygromycin-B Natural products OC1C(NC)CC(N)C(O)C1OC1C2OC3(C(C(O)C(O)C(C(N)CO)O3)O)OC2C(O)C(CO)O1 GRRNUXAQVGOGFE-UHFFFAOYSA-N 0.000 description 1
- MPBVHIBUJCELCL-UHFFFAOYSA-N Ibandronate Chemical compound CCCCCN(C)CCC(O)(P(O)(O)=O)P(O)(O)=O MPBVHIBUJCELCL-UHFFFAOYSA-N 0.000 description 1
- 102000006992 Interferon-alpha Human genes 0.000 description 1
- 108010047761 Interferon-alpha Proteins 0.000 description 1
- 102000008070 Interferon-gamma Human genes 0.000 description 1
- 108010074328 Interferon-gamma Proteins 0.000 description 1
- 208000005016 Intestinal Neoplasms Diseases 0.000 description 1
- JUZNIMUFDBIJCM-ANEDZVCMSA-N Invanz Chemical compound O=C([C@H]1NC[C@H](C1)SC=1[C@H](C)[C@@H]2[C@H](C(N2C=1C(O)=O)=O)[C@H](O)C)NC1=CC=CC(C(O)=O)=C1 JUZNIMUFDBIJCM-ANEDZVCMSA-N 0.000 description 1
- VQTUBCCKSQIDNK-UHFFFAOYSA-N Isobutene Chemical group CC(C)=C VQTUBCCKSQIDNK-UHFFFAOYSA-N 0.000 description 1
- KJHKTHWMRKYKJE-SUGCFTRWSA-N Kaletra Chemical compound N1([C@@H](C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC=2C=CC=CC=2)NC(=O)COC=2C(=CC=CC=2C)C)CC=2C=CC=CC=2)CCCNC1=O KJHKTHWMRKYKJE-SUGCFTRWSA-N 0.000 description 1
- 239000005517 L01XE01 - Imatinib Substances 0.000 description 1
- 239000005411 L01XE02 - Gefitinib Substances 0.000 description 1
- 239000002147 L01XE04 - Sunitinib Substances 0.000 description 1
- 239000002067 L01XE06 - Dasatinib Substances 0.000 description 1
- 239000002136 L01XE07 - Lapatinib Substances 0.000 description 1
- 239000005536 L01XE08 - Nilotinib Substances 0.000 description 1
- 239000003798 L01XE11 - Pazopanib Substances 0.000 description 1
- 239000002118 L01XE12 - Vandetanib Substances 0.000 description 1
- 239000002144 L01XE18 - Ruxolitinib Substances 0.000 description 1
- 239000002176 L01XE26 - Cabozantinib Substances 0.000 description 1
- FGBAVQUHSKYMTC-UHFFFAOYSA-M LDS 751 dye Chemical compound [O-]Cl(=O)(=O)=O.C1=CC2=CC(N(C)C)=CC=C2[N+](CC)=C1C=CC=CC1=CC=C(N(C)C)C=C1 FGBAVQUHSKYMTC-UHFFFAOYSA-M 0.000 description 1
- 101000591392 Leishmania infantum Probable flavin mononucleotide-dependent alkene reductase Proteins 0.000 description 1
- 229920001491 Lentinan Polymers 0.000 description 1
- GSDSWSVVBLHKDQ-JTQLQIEISA-N Levofloxacin Chemical compound C([C@@H](N1C2=C(C(C(C(O)=O)=C1)=O)C=C1F)C)OC2=C1N1CCN(C)CC1 GSDSWSVVBLHKDQ-JTQLQIEISA-N 0.000 description 1
- OJMMVQQUTAEWLP-UHFFFAOYSA-N Lincomycin Natural products CN1CC(CCC)CC1C(=O)NC(C(C)O)C1C(O)C(O)C(O)C(SC)O1 OJMMVQQUTAEWLP-UHFFFAOYSA-N 0.000 description 1
- 102000019298 Lipocalin Human genes 0.000 description 1
- 108050006654 Lipocalin Proteins 0.000 description 1
- 108010028921 Lipopeptides Proteins 0.000 description 1
- GQYIWUVLTXOXAJ-UHFFFAOYSA-N Lomustine Chemical compound ClCCN(N=O)C(=O)NC1CCCCC1 GQYIWUVLTXOXAJ-UHFFFAOYSA-N 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 101800001751 Melanocyte-stimulating hormone alpha Proteins 0.000 description 1
- 102400000740 Melanocyte-stimulating hormone alpha Human genes 0.000 description 1
- 101710151321 Melanostatin Proteins 0.000 description 1
- 101710200814 Melanotropin alpha Proteins 0.000 description 1
- ZRVUJXDFFKFLMG-UHFFFAOYSA-N Meloxicam Chemical compound OC=1C2=CC=CC=C2S(=O)(=O)N(C)C=1C(=O)NC1=NC=C(C)S1 ZRVUJXDFFKFLMG-UHFFFAOYSA-N 0.000 description 1
- RJQXTJLFIWVMTO-TYNCELHUSA-N Methicillin Chemical compound COC1=CC=CC(OC)=C1C(=O)N[C@@H]1C(=O)N2[C@@H](C(O)=O)C(C)(C)S[C@@H]21 RJQXTJLFIWVMTO-TYNCELHUSA-N 0.000 description 1
- DMUAPQTXSSNEDD-QALJCMCCSA-N Midecamycin Chemical compound C1[C@](O)(C)[C@@H](OC(=O)CC)[C@H](C)O[C@H]1O[C@H]1[C@H](N(C)C)[C@@H](O)[C@H](O[C@@H]2[C@H]([C@H](OC(=O)CC)CC(=O)O[C@H](C)C/C=C/C=C/[C@H](O)[C@H](C)C[C@@H]2CC=O)OC)O[C@@H]1C DMUAPQTXSSNEDD-QALJCMCCSA-N 0.000 description 1
- 229930192051 Mikamycin Natural products 0.000 description 1
- 108010046774 Mikamycin Proteins 0.000 description 1
- PCZOHLXUXFIOCF-UHFFFAOYSA-N Monacolin X Natural products C12C(OC(=O)C(C)CC)CC(C)C=C2C=CC(C)C1CCC1CC(O)CC(=O)O1 PCZOHLXUXFIOCF-UHFFFAOYSA-N 0.000 description 1
- 239000005462 Mubritinib Substances 0.000 description 1
- HRNLUBSXIHFDHP-UHFFFAOYSA-N N-(2-aminophenyl)-4-[[[4-(3-pyridinyl)-2-pyrimidinyl]amino]methyl]benzamide Chemical compound NC1=CC=CC=C1NC(=O)C(C=C1)=CC=C1CNC1=NC=CC(C=2C=NC=CC=2)=N1 HRNLUBSXIHFDHP-UHFFFAOYSA-N 0.000 description 1
- NQTADLQHYWFPDB-UHFFFAOYSA-N N-Hydroxysuccinimide Chemical compound ON1C(=O)CCC1=O NQTADLQHYWFPDB-UHFFFAOYSA-N 0.000 description 1
- LFZAGIJXANFPFN-UHFFFAOYSA-N N-[3-[4-(3-methyl-5-propan-2-yl-1,2,4-triazol-4-yl)piperidin-1-yl]-1-thiophen-2-ylpropyl]acetamide Chemical compound C(C)(C)C1=NN=C(N1C1CCN(CC1)CCC(C=1SC=CC=1)NC(C)=O)C LFZAGIJXANFPFN-UHFFFAOYSA-N 0.000 description 1
- YALNUENQHAQXEA-UHFFFAOYSA-N N-[4-[(hydroxyamino)-oxomethyl]phenyl]carbamic acid [6-(diethylaminomethyl)-2-naphthalenyl]methyl ester Chemical compound C1=CC2=CC(CN(CC)CC)=CC=C2C=C1COC(=O)NC1=CC=C(C(=O)NO)C=C1 YALNUENQHAQXEA-UHFFFAOYSA-N 0.000 description 1
- UBQYURCVBFRUQT-UHFFFAOYSA-N N-benzoyl-Ferrioxamine B Chemical compound CC(=O)N(O)CCCCCNC(=O)CCC(=O)N(O)CCCCCNC(=O)CCC(=O)N(O)CCCCCN UBQYURCVBFRUQT-UHFFFAOYSA-N 0.000 description 1
- ZDZOTLJHXYCWBA-VCVYQWHSSA-N N-debenzoyl-N-(tert-butoxycarbonyl)-10-deacetyltaxol Chemical compound O([C@H]1[C@H]2[C@@](C([C@H](O)C3=C(C)[C@@H](OC(=O)[C@H](O)[C@@H](NC(=O)OC(C)(C)C)C=4C=CC=CC=4)C[C@]1(O)C3(C)C)=O)(C)[C@@H](O)C[C@H]1OC[C@]12OC(=O)C)C(=O)C1=CC=CC=C1 ZDZOTLJHXYCWBA-VCVYQWHSSA-N 0.000 description 1
- 108010047562 NGR peptide Proteins 0.000 description 1
- 102000017921 NTSR1 Human genes 0.000 description 1
- 102000017938 NTSR2 Human genes 0.000 description 1
- 108010021717 Nafarelin Proteins 0.000 description 1
- 229930193140 Neomycin Natural products 0.000 description 1
- 229940123424 Neuraminidase inhibitor Drugs 0.000 description 1
- 108010040718 Neurokinin-1 Receptors Proteins 0.000 description 1
- 102400000064 Neuropeptide Y Human genes 0.000 description 1
- 102000017922 Neurotensin receptor Human genes 0.000 description 1
- 108060003370 Neurotensin receptor Proteins 0.000 description 1
- 101710138657 Neurotoxin Proteins 0.000 description 1
- GRYLNZFGIOXLOG-UHFFFAOYSA-N Nitric acid Chemical compound O[N+]([O-])=O GRYLNZFGIOXLOG-UHFFFAOYSA-N 0.000 description 1
- 239000004104 Oleandomycin Substances 0.000 description 1
- RZPAKFUAFGMUPI-UHFFFAOYSA-N Oleandomycin Natural products O1C(C)C(O)C(OC)CC1OC1C(C)C(=O)OC(C)C(C)C(O)C(C)C(=O)C2(OC2)CC(C)C(OC2C(C(CC(C)O2)N(C)C)O)C1C RZPAKFUAFGMUPI-UHFFFAOYSA-N 0.000 description 1
- 108010038807 Oligopeptides Proteins 0.000 description 1
- 102000015636 Oligopeptides Human genes 0.000 description 1
- QIPQASLPWJVQMH-DTORHVGOSA-N Orbifloxacin Chemical compound C1[C@@H](C)N[C@@H](C)CN1C1=C(F)C(F)=C2C(=O)C(C(O)=O)=CN(C3CC3)C2=C1F QIPQASLPWJVQMH-DTORHVGOSA-N 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 239000004100 Oxytetracycline Substances 0.000 description 1
- VREZDOWOLGNDPW-ALTGWBOUSA-N Pancratistatin Chemical compound C1=C2[C@H]3[C@@H](O)[C@H](O)[C@@H](O)[C@@H](O)[C@@H]3NC(=O)C2=C(O)C2=C1OCO2 VREZDOWOLGNDPW-ALTGWBOUSA-N 0.000 description 1
- VREZDOWOLGNDPW-MYVCAWNPSA-N Pancratistatin Natural products O=C1N[C@H]2[C@H](O)[C@H](O)[C@H](O)[C@H](O)[C@@H]2c2c1c(O)c1OCOc1c2 VREZDOWOLGNDPW-MYVCAWNPSA-N 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- TYMABNNERDVXID-DLYFRVTGSA-N Panipenem Chemical compound C([C@@H]1[C@H](C(N1C=1C(O)=O)=O)[C@H](O)C)C=1S[C@H]1CCN(C(C)=N)C1 TYMABNNERDVXID-DLYFRVTGSA-N 0.000 description 1
- UOZODPSAJZTQNH-UHFFFAOYSA-N Paromomycin II Natural products NC1C(O)C(O)C(CN)OC1OC1C(O)C(OC2C(C(N)CC(N)C2O)OC2C(C(O)C(O)C(CO)O2)N)OC1CO UOZODPSAJZTQNH-UHFFFAOYSA-N 0.000 description 1
- JNTOCHDNEULJHD-UHFFFAOYSA-N Penciclovir Chemical compound N1C(N)=NC(=O)C2=C1N(CCC(CO)CO)C=N2 JNTOCHDNEULJHD-UHFFFAOYSA-N 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 108010057150 Peplomycin Proteins 0.000 description 1
- KMSKQZKKOZQFFG-HSUXVGOQSA-N Pirarubicin Chemical compound O([C@H]1[C@@H](N)C[C@@H](O[C@H]1C)O[C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1CCCCO1 KMSKQZKKOZQFFG-HSUXVGOQSA-N 0.000 description 1
- 239000004698 Polyethylene Substances 0.000 description 1
- 108010093965 Polymyxin B Proteins 0.000 description 1
- 102100027467 Pro-opiomelanocortin Human genes 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- 102000004005 Prostaglandin-endoperoxide synthases Human genes 0.000 description 1
- 108090000459 Prostaglandin-endoperoxide synthases Proteins 0.000 description 1
- 241000937820 Remora Species 0.000 description 1
- AUNGANRZJHBGPY-SCRDCRAPSA-N Riboflavin Chemical compound OC[C@@H](O)[C@@H](O)[C@@H](O)CN1C=2C=C(C)C(C)=CC=2N=C2C1=NC(=O)NC2=O AUNGANRZJHBGPY-SCRDCRAPSA-N 0.000 description 1
- 229940127395 Ribonucleotide Reductase Inhibitors Drugs 0.000 description 1
- URWAJWIAIPFPJE-UHFFFAOYSA-N Rickamicin Natural products O1CC(O)(C)C(NC)C(O)C1OC1C(O)C(OC2C(CC=C(CN)O2)N)C(N)CC1N URWAJWIAIPFPJE-UHFFFAOYSA-N 0.000 description 1
- 229930189077 Rifamycin Natural products 0.000 description 1
- NCDNCNXCDXHOMX-UHFFFAOYSA-N Ritonavir Natural products C=1C=CC=CC=1CC(NC(=O)OCC=1SC=NC=1)C(O)CC(CC=1C=CC=CC=1)NC(=O)C(C(C)C)NC(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-UHFFFAOYSA-N 0.000 description 1
- 108010087230 Sincalide Proteins 0.000 description 1
- 108010003723 Single-Domain Antibodies Proteins 0.000 description 1
- 229930192786 Sisomicin Natural products 0.000 description 1
- 108020004459 Small interfering RNA Proteins 0.000 description 1
- 108010056088 Somatostatin Proteins 0.000 description 1
- 102000005157 Somatostatin Human genes 0.000 description 1
- 102100032889 Sortilin Human genes 0.000 description 1
- 239000004187 Spiramycin Substances 0.000 description 1
- XNKLLVCARDGLGL-JGVFFNPUSA-N Stavudine Chemical compound O=C1NC(=O)C(C)=CN1[C@H]1C=C[C@@H](CO)O1 XNKLLVCARDGLGL-JGVFFNPUSA-N 0.000 description 1
- 108010090804 Streptavidin Proteins 0.000 description 1
- ZSJLQEPLLKMAKR-UHFFFAOYSA-N Streptozotocin Natural products O=NN(C)C(=O)NC1C(O)OC(CO)C(O)C1O ZSJLQEPLLKMAKR-UHFFFAOYSA-N 0.000 description 1
- 102400000096 Substance P Human genes 0.000 description 1
- 101800003906 Substance P Proteins 0.000 description 1
- 102100037346 Substance-P receptor Human genes 0.000 description 1
- NHUHCSRWZMLRLA-UHFFFAOYSA-N Sulfisoxazole Chemical compound CC1=NOC(NS(=O)(=O)C=2C=CC(N)=CC=2)=C1C NHUHCSRWZMLRLA-UHFFFAOYSA-N 0.000 description 1
- 241000779819 Syncarpia glomulifera Species 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- 108010053950 Teicoplanin Proteins 0.000 description 1
- HATRDXDCPOXQJX-UHFFFAOYSA-N Thapsigargin Natural products CCCCCCCC(=O)OC1C(OC(O)C(=C/C)C)C(=C2C3OC(=O)C(C)(O)C3(O)C(CC(C)(OC(=O)C)C12)OC(=O)CCC)C HATRDXDCPOXQJX-UHFFFAOYSA-N 0.000 description 1
- JZRWCGZRTZMZEH-UHFFFAOYSA-N Thiamine Natural products CC1=C(CCO)SC=[N+]1CC1=CN=C(C)N=C1N JZRWCGZRTZMZEH-UHFFFAOYSA-N 0.000 description 1
- FOCVUCIESVLUNU-UHFFFAOYSA-N Thiotepa Chemical compound C1CN1P(N1CC1)(=S)N1CC1 FOCVUCIESVLUNU-UHFFFAOYSA-N 0.000 description 1
- SUJUHGSWHZTSEU-UHFFFAOYSA-N Tipranavir Natural products C1C(O)=C(C(CC)C=2C=C(NS(=O)(=O)C=3N=CC(=CC=3)C(F)(F)F)C=CC=2)C(=O)OC1(CCC)CCC1=CC=CC=C1 SUJUHGSWHZTSEU-UHFFFAOYSA-N 0.000 description 1
- DPXHITFUCHFTKR-UHFFFAOYSA-L To-Pro-1 Chemical compound [I-].[I-].S1C2=CC=CC=C2[N+](C)=C1C=C1C2=CC=CC=C2N(CCC[N+](C)(C)C)C=C1 DPXHITFUCHFTKR-UHFFFAOYSA-L 0.000 description 1
- QHNORJFCVHUPNH-UHFFFAOYSA-L To-Pro-3 Chemical compound [I-].[I-].S1C2=CC=CC=C2[N+](C)=C1C=CC=C1C2=CC=CC=C2N(CCC[N+](C)(C)C)C=C1 QHNORJFCVHUPNH-UHFFFAOYSA-L 0.000 description 1
- MZZINWWGSYUHGU-UHFFFAOYSA-J ToTo-1 Chemical compound [I-].[I-].[I-].[I-].C12=CC=CC=C2C(C=C2N(C3=CC=CC=C3S2)C)=CC=[N+]1CCC[N+](C)(C)CCC[N+](C)(C)CCC[N+](C1=CC=CC=C11)=CC=C1C=C1N(C)C2=CC=CC=C2S1 MZZINWWGSYUHGU-UHFFFAOYSA-J 0.000 description 1
- IVTVGDXNLFLDRM-HNNXBMFYSA-N Tomudex Chemical compound C=1C=C2NC(C)=NC(=O)C2=CC=1CN(C)C1=CC=C(C(=O)N[C@@H](CCC(O)=O)C(O)=O)S1 IVTVGDXNLFLDRM-HNNXBMFYSA-N 0.000 description 1
- 102220615016 Transcription elongation regulator 1_S65C_mutation Human genes 0.000 description 1
- 101800001690 Transmembrane protein gp41 Proteins 0.000 description 1
- SHGAZHPCJJPHSC-NWVFGJFESA-N Tretinoin Chemical compound OC(=O)/C=C(\C)/C=C/C=C(C)C=CC1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-NWVFGJFESA-N 0.000 description 1
- RTKIYFITIVXBLE-UHFFFAOYSA-N Trichostatin A Natural products ONC(=O)C=CC(C)=CC(C)C(=O)C1=CC=C(N(C)C)C=C1 RTKIYFITIVXBLE-UHFFFAOYSA-N 0.000 description 1
- 108010050144 Triptorelin Pamoate Proteins 0.000 description 1
- 102000004243 Tubulin Human genes 0.000 description 1
- 108090000704 Tubulin Proteins 0.000 description 1
- 108091008605 VEGF receptors Proteins 0.000 description 1
- HDOVUKNUBWVHOX-QMMMGPOBSA-N Valacyclovir Chemical compound N1C(N)=NC(=O)C2=C1N(COCCOC(=O)[C@@H](N)C(C)C)C=N2 HDOVUKNUBWVHOX-QMMMGPOBSA-N 0.000 description 1
- 108010059993 Vancomycin Proteins 0.000 description 1
- 102000009484 Vascular Endothelial Growth Factor Receptors Human genes 0.000 description 1
- 102100038388 Vasoactive intestinal polypeptide receptor 1 Human genes 0.000 description 1
- 101710137655 Vasoactive intestinal polypeptide receptor 1 Proteins 0.000 description 1
- 102100038286 Vasoactive intestinal polypeptide receptor 2 Human genes 0.000 description 1
- 241000545067 Venus Species 0.000 description 1
- 229940122803 Vinca alkaloid Drugs 0.000 description 1
- MECHNRXZTMCUDQ-UHFFFAOYSA-N Vitamin D2 Natural products C1CCC2(C)C(C(C)C=CC(C)C(C)C)CCC2C1=CC=C1CC(O)CCC1=C MECHNRXZTMCUDQ-UHFFFAOYSA-N 0.000 description 1
- QYSXJUFSXHHAJI-XFEUOLMDSA-N Vitamin D3 Natural products C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)CCCC(C)C)=C/C=C1\C[C@@H](O)CCC1=C QYSXJUFSXHHAJI-XFEUOLMDSA-N 0.000 description 1
- 108091005971 Wild-type GFP Proteins 0.000 description 1
- GRRMZXFOOGQMFA-UHFFFAOYSA-J YoYo-1 Chemical compound [I-].[I-].[I-].[I-].C12=CC=CC=C2C(C=C2N(C3=CC=CC=C3O2)C)=CC=[N+]1CCC[N+](C)(C)CCC[N+](C)(C)CCC[N+](C1=CC=CC=C11)=CC=C1C=C1N(C)C2=CC=CC=C2O1 GRRMZXFOOGQMFA-UHFFFAOYSA-J 0.000 description 1
- LJFFDOBFKICLHN-IXWHRVGISA-N [(1S,2R,3S,5S,6S,16E,18E,20R,21S)-11-chloro-21-hydroxy-12,20-dimethoxy-2,5,9,16-tetramethyl-8,23-dioxo-4,24-dioxa-9,22-diazatetracyclo[19.3.1.110,14.03,5]hexacosa-10,12,14(26),16,18-pentaen-6-yl] (2S)-2-[methyl(4-sulfanylpentanoyl)amino]propanoate Chemical compound CO[C@@H]([C@@]1(O)C[C@H](OC(=O)N1)[C@@H](C)[C@@H]1O[C@@]1(C)[C@@H](OC(=O)[C@H](C)N(C)C(=O)CCC(C)S)CC(=O)N1C)\C=C\C=C(C)\CC2=CC(OC)=C(Cl)C1=C2 LJFFDOBFKICLHN-IXWHRVGISA-N 0.000 description 1
- NBLHOLNNKJBEDC-XOGQCRKLSA-N [(2r,3s,4s,5r,6r)-2-[(2r,3s,4s,5s,6s)-2-[(1r,2s)-2-[[6-amino-2-[(1s)-3-amino-1-[[(2s)-2,3-diamino-3-oxopropyl]amino]-3-oxopropyl]-5-methylpyrimidine-4-carbonyl]amino]-3-[[(2r,3s,4s)-5-[[(2s,3r)-1-[2-[4-[4-[4-(diaminomethylideneamino)butylcarbamoyl]-1,3-th Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCCCN=C(N)N)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1NC=NC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C NBLHOLNNKJBEDC-XOGQCRKLSA-N 0.000 description 1
- ZWBTYMGEBZUQTK-PVLSIAFMSA-N [(7S,9E,11S,12R,13S,14R,15R,16R,17S,18S,19E,21Z)-2,15,17,32-tetrahydroxy-11-methoxy-3,7,12,14,16,18,22-heptamethyl-1'-(2-methylpropyl)-6,23-dioxospiro[8,33-dioxa-24,27,29-triazapentacyclo[23.6.1.14,7.05,31.026,30]tritriaconta-1(32),2,4,9,19,21,24,26,30-nonaene-28,4'-piperidine]-13-yl] acetate Chemical compound CO[C@H]1\C=C\O[C@@]2(C)Oc3c(C2=O)c2c4NC5(CCN(CC(C)C)CC5)N=c4c(=NC(=O)\C(C)=C/C=C/[C@H](C)[C@H](O)[C@@H](C)[C@@H](O)[C@@H](C)[C@H](OC(C)=O)[C@@H]1C)c(O)c2c(O)c3C ZWBTYMGEBZUQTK-PVLSIAFMSA-N 0.000 description 1
- IFJUINDAXYAPTO-UUBSBJJBSA-N [(8r,9s,13s,14s,17s)-17-[2-[4-[4-[bis(2-chloroethyl)amino]phenyl]butanoyloxy]acetyl]oxy-13-methyl-6,7,8,9,11,12,14,15,16,17-decahydrocyclopenta[a]phenanthren-3-yl] benzoate Chemical compound C([C@@H]1[C@@H](C2=CC=3)CC[C@]4([C@H]1CC[C@@H]4OC(=O)COC(=O)CCCC=1C=CC(=CC=1)N(CCCl)CCCl)C)CC2=CC=3OC(=O)C1=CC=CC=C1 IFJUINDAXYAPTO-UUBSBJJBSA-N 0.000 description 1
- ZHAFUINZIZIXFC-UHFFFAOYSA-N [9-(dimethylamino)-10-methylbenzo[a]phenoxazin-5-ylidene]azanium;chloride Chemical compound [Cl-].O1C2=CC(=[NH2+])C3=CC=CC=C3C2=NC2=C1C=C(N(C)C)C(C)=C2 ZHAFUINZIZIXFC-UHFFFAOYSA-N 0.000 description 1
- DULZJSBFYXKCJG-UHFFFAOYSA-M [OH-].[Si+4].CN(C)CCC[Si](C)(C)[O-].c1ccc2c3nc(nc4[n-]c(nc5nc(nc6[n-]c(n3)c3ccccc63)c3ccccc53)c3ccccc43)c2c1 Chemical compound [OH-].[Si+4].CN(C)CCC[Si](C)(C)[O-].c1ccc2c3nc(nc4[n-]c(nc5nc(nc6[n-]c(n3)c3ccccc63)c3ccccc53)c3ccccc43)c2c1 DULZJSBFYXKCJG-UHFFFAOYSA-M 0.000 description 1
- 229960004748 abacavir Drugs 0.000 description 1
- MCGSCOLBFJQGHM-SCZZXKLOSA-N abacavir Chemical compound C=12N=CN([C@H]3C=C[C@@H](CO)C3)C2=NC(N)=NC=1NC1CC1 MCGSCOLBFJQGHM-SCZZXKLOSA-N 0.000 description 1
- 108010023617 abarelix Proteins 0.000 description 1
- AIWRTTMUVOZGPW-HSPKUQOVSA-N abarelix Chemical compound C([C@@H](C(=O)N[C@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@H](C)C(N)=O)N(C)C(=O)[C@H](CO)NC(=O)[C@@H](CC=1C=NC=CC=1)NC(=O)[C@@H](CC=1C=CC(Cl)=CC=1)NC(=O)[C@@H](CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(O)C=C1 AIWRTTMUVOZGPW-HSPKUQOVSA-N 0.000 description 1
- 229960002184 abarelix Drugs 0.000 description 1
- 229960000446 abciximab Drugs 0.000 description 1
- 229950008805 abexinostat Drugs 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- DEXPIBGCLCPUHE-UISHROKMSA-N acetic acid;(4r,7s,10s,13r,16s,19r)-10-(4-aminobutyl)-n-[(2s,3r)-1-amino-3-hydroxy-1-oxobutan-2-yl]-19-[[(2r)-2-amino-3-naphthalen-2-ylpropanoyl]amino]-16-[(4-hydroxyphenyl)methyl]-13-(1h-indol-3-ylmethyl)-6,9,12,15,18-pentaoxo-7-propan-2-yl-1,2-dithia-5, Chemical compound CC(O)=O.C([C@H]1C(=O)N[C@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(N[C@@H](CSSC[C@@H](C(=O)N1)NC(=O)[C@H](N)CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H]([C@@H](C)O)C(N)=O)=O)C(C)C)C1=CC=C(O)C=C1 DEXPIBGCLCPUHE-UISHROKMSA-N 0.000 description 1
- 108010052004 acetyl-2-naphthylalanyl-3-chlorophenylalanyl-1-oxohexadecyl-seryl-4-aminophenylalanyl(hydroorotyl)-4-aminophenylalanyl(carbamoyl)-leucyl-ILys-prolyl-alaninamide Proteins 0.000 description 1
- BGLGAKMTYHWWKW-UHFFFAOYSA-N acridine yellow Chemical compound [H+].[Cl-].CC1=C(N)C=C2N=C(C=C(C(C)=C3)N)C3=CC2=C1 BGLGAKMTYHWWKW-UHFFFAOYSA-N 0.000 description 1
- 150000001251 acridines Chemical class 0.000 description 1
- 230000003213 activating effect Effects 0.000 description 1
- 230000010933 acylation Effects 0.000 description 1
- 238000005917 acylation reaction Methods 0.000 description 1
- 229960001997 adefovir Drugs 0.000 description 1
- WOZSCQDILHKSGG-UHFFFAOYSA-N adefovir depivoxil Chemical compound N1=CN=C2N(CCOCP(=O)(OCOC(=O)C(C)(C)C)OCOC(=O)C(C)(C)C)C=NC2=C1N WOZSCQDILHKSGG-UHFFFAOYSA-N 0.000 description 1
- 229960005305 adenosine Drugs 0.000 description 1
- 239000000362 adenosine triphosphatase inhibitor Substances 0.000 description 1
- 102000030621 adenylate cyclase Human genes 0.000 description 1
- 108060000200 adenylate cyclase Proteins 0.000 description 1
- 230000001919 adrenal effect Effects 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 229960001686 afatinib Drugs 0.000 description 1
- ULXXDDBFHOBEHA-CWDCEQMOSA-N afatinib Chemical compound N1=CN=C2C=C(O[C@@H]3COCC3)C(NC(=O)/C=C/CN(C)C)=CC2=C1NC1=CC=C(F)C(Cl)=C1 ULXXDDBFHOBEHA-CWDCEQMOSA-N 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 150000003797 alkaloid derivatives Chemical class 0.000 description 1
- 229940045714 alkyl sulfonate alkylating agent Drugs 0.000 description 1
- 150000008052 alkyl sulfonates Chemical class 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- WUOACPNHFRMFPN-UHFFFAOYSA-N alpha-terpineol Chemical compound CC1=CCC(C(C)(C)O)CC1 WUOACPNHFRMFPN-UHFFFAOYSA-N 0.000 description 1
- 229960000473 altretamine Drugs 0.000 description 1
- 229960003099 amcinonide Drugs 0.000 description 1
- ILKJAFIWWBXGDU-MOGDOJJUSA-N amcinonide Chemical compound O([C@@]1([C@H](O2)C[C@@H]3[C@@]1(C[C@H](O)[C@]1(F)[C@@]4(C)C=CC(=O)C=C4CC[C@H]13)C)C(=O)COC(=O)C)C12CCCC1 ILKJAFIWWBXGDU-MOGDOJJUSA-N 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 229960002684 aminocaproic acid Drugs 0.000 description 1
- 229960003437 aminoglutethimide Drugs 0.000 description 1
- ROBVIMPUHSLWNV-UHFFFAOYSA-N aminoglutethimide Chemical compound C=1C=C(N)C=CC=1C1(CC)CCC(=O)NC1=O ROBVIMPUHSLWNV-UHFFFAOYSA-N 0.000 description 1
- 229940126575 aminoglycoside Drugs 0.000 description 1
- 229960002749 aminolevulinic acid Drugs 0.000 description 1
- HTIQEAQVCYTUBX-UHFFFAOYSA-N amlodipine Chemical compound CCOC(=O)C1=C(COCCN)NC(C)=C(C(=O)OC)C1C1=CC=CC=C1Cl HTIQEAQVCYTUBX-UHFFFAOYSA-N 0.000 description 1
- 229960000528 amlodipine Drugs 0.000 description 1
- 229960001830 amprenavir Drugs 0.000 description 1
- YMARZQAQMVYCKC-OEMFJLHTSA-N amprenavir Chemical compound C([C@@H]([C@H](O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1COCC1)C1=CC=CC=C1 YMARZQAQMVYCKC-OEMFJLHTSA-N 0.000 description 1
- XCPGHVQEEXUHNC-UHFFFAOYSA-N amsacrine Chemical compound COC1=CC(NS(C)(=O)=O)=CC=C1NC1=C(C=CC=C2)C2=NC2=CC=CC=C12 XCPGHVQEEXUHNC-UHFFFAOYSA-N 0.000 description 1
- 229960001220 amsacrine Drugs 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 229950000242 ancitabine Drugs 0.000 description 1
- BBDAGFIXKZCXAH-CCXZUQQUSA-N ancitabine Chemical compound N=C1C=CN2[C@@H]3O[C@H](CO)[C@@H](O)[C@@H]3OC2=N1 BBDAGFIXKZCXAH-CCXZUQQUSA-N 0.000 description 1
- 239000003098 androgen Substances 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- 150000001454 anthracenes Chemical class 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- VGQOVCHZGQWAOI-YQRHFANHSA-N anthramycin Chemical compound N1[C@H](O)[C@@H]2CC(\C=C\C(N)=O)=CN2C(=O)C2=CC=C(C)C(O)=C12 VGQOVCHZGQWAOI-YQRHFANHSA-N 0.000 description 1
- 150000004056 anthraquinones Chemical class 0.000 description 1
- 230000002280 anti-androgenic effect Effects 0.000 description 1
- 229940046836 anti-estrogen Drugs 0.000 description 1
- 230000001833 anti-estrogenic effect Effects 0.000 description 1
- 230000003432 anti-folate effect Effects 0.000 description 1
- 230000000692 anti-sense effect Effects 0.000 description 1
- 239000000051 antiandrogen Substances 0.000 description 1
- 229940124350 antibacterial drug Drugs 0.000 description 1
- 229940127074 antifolate Drugs 0.000 description 1
- 229940045687 antimetabolites folic acid analogs Drugs 0.000 description 1
- 239000003080 antimitotic agent Substances 0.000 description 1
- 229940045719 antineoplastic alkylating agent nitrosoureas Drugs 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 229940027998 antiseptic and disinfectant acridine derivative Drugs 0.000 description 1
- 229950006345 antramycin Drugs 0.000 description 1
- 150000008209 arabinosides Chemical class 0.000 description 1
- 229960005397 arbekacin Drugs 0.000 description 1
- MKKYBZZTJQGVCD-XTCKQBCOSA-N arbekacin Chemical compound O([C@@H]1[C@@H](N)C[C@H]([C@@H]([C@H]1O)O[C@@H]1[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O1)O)NC(=O)[C@@H](O)CCN)[C@H]1O[C@H](CN)CC[C@H]1N MKKYBZZTJQGVCD-XTCKQBCOSA-N 0.000 description 1
- 229960003277 atazanavir Drugs 0.000 description 1
- AXRYRYVKAWYZBR-GASGPIRDSA-N atazanavir Chemical compound C([C@H](NC(=O)[C@@H](NC(=O)OC)C(C)(C)C)[C@@H](O)CN(CC=1C=CC(=CC=1)C=1N=CC=CC=1)NC(=O)[C@@H](NC(=O)OC)C(C)(C)C)C1=CC=CC=C1 AXRYRYVKAWYZBR-GASGPIRDSA-N 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- JPIYZTWMUGTEHX-UHFFFAOYSA-N auramine O free base Chemical compound C1=CC(N(C)C)=CC=C1C(=N)C1=CC=C(N(C)C)C=C1 JPIYZTWMUGTEHX-UHFFFAOYSA-N 0.000 description 1
- 229960002756 azacitidine Drugs 0.000 description 1
- VSRXQHXAPYXROS-UHFFFAOYSA-N azanide;cyclobutane-1,1-dicarboxylic acid;platinum(2+) Chemical compound [NH2-].[NH2-].[Pt+2].OC(=O)C1(C(O)=O)CCC1 VSRXQHXAPYXROS-UHFFFAOYSA-N 0.000 description 1
- 229950011321 azaserine Drugs 0.000 description 1
- 150000001541 aziridines Chemical class 0.000 description 1
- 229960004099 azithromycin Drugs 0.000 description 1
- MQTOSJVFKKJCRP-BICOPXKESA-N azithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)N(C)C[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 MQTOSJVFKKJCRP-BICOPXKESA-N 0.000 description 1
- JTWOMNBEOCYFNV-NFFDBFGFSA-N azlocillin Chemical compound N([C@@H](C(=O)N[C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C=1C=CC=CC=1)C(=O)N1CCNC1=O JTWOMNBEOCYFNV-NFFDBFGFSA-N 0.000 description 1
- 229960003623 azlocillin Drugs 0.000 description 1
- WZPBZJONDBGPKJ-VEHQQRBSSA-N aztreonam Chemical compound O=C1N(S([O-])(=O)=O)[C@@H](C)[C@@H]1NC(=O)C(=N/OC(C)(C)C(O)=O)\C1=CSC([NH3+])=N1 WZPBZJONDBGPKJ-VEHQQRBSSA-N 0.000 description 1
- 229960003644 aztreonam Drugs 0.000 description 1
- 108010016899 bacitracin A Proteins 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 229950000210 beclometasone dipropionate Drugs 0.000 description 1
- 229960001192 bekanamycin Drugs 0.000 description 1
- 229960003094 belinostat Drugs 0.000 description 1
- NCNRHFGMJRPRSK-MDZDMXLPSA-N belinostat Chemical compound ONC(=O)\C=C\C1=CC=CC(S(=O)(=O)NC=2C=CC=CC=2)=C1 NCNRHFGMJRPRSK-MDZDMXLPSA-N 0.000 description 1
- LNHWXBUNXOXMRL-VWLOTQADSA-N belotecan Chemical compound C1=CC=C2C(CCNC(C)C)=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 LNHWXBUNXOXMRL-VWLOTQADSA-N 0.000 description 1
- 229950011276 belotecan Drugs 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 150000001557 benzodiazepines Chemical class 0.000 description 1
- 239000003781 beta lactamase inhibitor Substances 0.000 description 1
- IQFYYKKMVGJFEH-UHFFFAOYSA-N beta-L-thymidine Natural products O=C1NC(=O)C(C)=CN1C1OC(CO)C(O)C1 IQFYYKKMVGJFEH-UHFFFAOYSA-N 0.000 description 1
- 229940126813 beta-lactamase inhibitor Drugs 0.000 description 1
- 229960002537 betamethasone Drugs 0.000 description 1
- UREBDLICKHMUKA-DVTGEIKXSA-N betamethasone Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H](C)[C@@](C(=O)CO)(O)[C@@]1(C)C[C@@H]2O UREBDLICKHMUKA-DVTGEIKXSA-N 0.000 description 1
- 229960000397 bevacizumab Drugs 0.000 description 1
- YJEJKUQEXFSVCJ-WRFMNRASSA-N bevirimat Chemical compound C1C[C@H](OC(=O)CC(C)(C)C(O)=O)C(C)(C)[C@@H]2CC[C@@]3(C)[C@]4(C)CC[C@@]5(C(O)=O)CC[C@@H](C(=C)C)[C@@H]5[C@H]4CC[C@@H]3[C@]21C YJEJKUQEXFSVCJ-WRFMNRASSA-N 0.000 description 1
- 229950002892 bevirimat Drugs 0.000 description 1
- 229960003169 biapenem Drugs 0.000 description 1
- MRMBZHPJVKCOMA-YJFSRANCSA-N biapenem Chemical compound C1N2C=NC=[N+]2CC1SC([C@@H]1C)=C(C([O-])=O)N2[C@H]1[C@@H]([C@H](O)C)C2=O MRMBZHPJVKCOMA-YJFSRANCSA-N 0.000 description 1
- 229960000997 bicalutamide Drugs 0.000 description 1
- 230000003115 biocidal effect Effects 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 229950008548 bisantrene Drugs 0.000 description 1
- NBLHOLNNKJBEDC-UHFFFAOYSA-N bleomycin B2 Natural products N=1C(C=2SC=C(N=2)C(=O)NCCCCN=C(N)N)=CSC=1CCNC(=O)C(C(O)C)NC(=O)C(C)C(O)C(C)NC(=O)C(C(OC1C(C(O)C(O)C(CO)O1)OC1C(C(OC(N)=O)C(O)C(CO)O1)O)C=1NC=NC=1)NC(=O)C1=NC(C(CC(N)=O)NCC(N)C(N)=O)=NC(N)=C1C NBLHOLNNKJBEDC-UHFFFAOYSA-N 0.000 description 1
- 230000000903 blocking effect Effects 0.000 description 1
- 229960000517 boceprevir Drugs 0.000 description 1
- LHHCSNFAOIFYRV-DOVBMPENSA-N boceprevir Chemical compound O=C([C@@H]1[C@@H]2[C@@H](C2(C)C)CN1C(=O)[C@@H](NC(=O)NC(C)(C)C)C(C)(C)C)NC(C(=O)C(N)=O)CC1CCC1 LHHCSNFAOIFYRV-DOVBMPENSA-N 0.000 description 1
- 229960001467 bortezomib Drugs 0.000 description 1
- GXJABQQUPOEUTA-RDJZCZTQSA-N bortezomib Chemical compound C([C@@H](C(=O)N[C@@H](CC(C)C)B(O)O)NC(=O)C=1N=CC=NC=1)C1=CC=CC=C1 GXJABQQUPOEUTA-RDJZCZTQSA-N 0.000 description 1
- UBPYILGKFZZVDX-UHFFFAOYSA-N bosutinib Chemical compound C1=C(Cl)C(OC)=CC(NC=2C3=CC(OC)=C(OCCCN4CCN(C)CC4)C=C3N=CC=2C#N)=C1Cl UBPYILGKFZZVDX-UHFFFAOYSA-N 0.000 description 1
- OZVBMTJYIDMWIL-AYFBDAFISA-N bromocriptine Chemical compound C1=CC(C=2[C@H](N(C)C[C@@H](C=2)C(=O)N[C@]2(C(=O)N3[C@H](C(N4CCC[C@H]4[C@]3(O)O2)=O)CC(C)C)C(C)C)C2)=C3C2=C(Br)NC3=C1 OZVBMTJYIDMWIL-AYFBDAFISA-N 0.000 description 1
- 229960002802 bromocriptine Drugs 0.000 description 1
- 229960005520 bryostatin Drugs 0.000 description 1
- MJQUEDHRCUIRLF-TVIXENOKSA-N bryostatin 1 Chemical compound C([C@@H]1CC(/[C@@H]([C@@](C(C)(C)/C=C/2)(O)O1)OC(=O)/C=C/C=C/CCC)=C\C(=O)OC)[C@H]([C@@H](C)O)OC(=O)C[C@H](O)C[C@@H](O1)C[C@H](OC(C)=O)C(C)(C)[C@]1(O)C[C@@H]1C\C(=C\C(=O)OC)C[C@H]\2O1 MJQUEDHRCUIRLF-TVIXENOKSA-N 0.000 description 1
- MUIWQCKLQMOUAT-AKUNNTHJSA-N bryostatin 20 Natural products COC(=O)C=C1C[C@@]2(C)C[C@]3(O)O[C@](C)(C[C@@H](O)CC(=O)O[C@](C)(C[C@@]4(C)O[C@](O)(CC5=CC(=O)O[C@]45C)C(C)(C)C=C[C@@](C)(C1)O2)[C@@H](C)O)C[C@H](OC(=O)C(C)(C)C)C3(C)C MUIWQCKLQMOUAT-AKUNNTHJSA-N 0.000 description 1
- 229960004436 budesonide Drugs 0.000 description 1
- MBABCNBNDNGODA-LUVUIASKSA-N bullatacin Chemical compound O1[C@@H]([C@@H](O)CCCCCCCCCC)CC[C@@H]1[C@@H]1O[C@@H]([C@H](O)CCCCCCCCCC[C@@H](O)CC=2C(O[C@@H](C)C=2)=O)CC1 MBABCNBNDNGODA-LUVUIASKSA-N 0.000 description 1
- CUWODFFVMXJOKD-UVLQAERKSA-N buserelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](COC(C)(C)C)NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)CC1=CC=C(O)C=C1 CUWODFFVMXJOKD-UVLQAERKSA-N 0.000 description 1
- 229960002719 buserelin Drugs 0.000 description 1
- 229960001292 cabozantinib Drugs 0.000 description 1
- ONIQOQHATWINJY-UHFFFAOYSA-N cabozantinib Chemical compound C=12C=C(OC)C(OC)=CC2=NC=CC=1OC(C=C1)=CC=C1NC(=O)C1(C(=O)NC=2C=CC(F)=CC=2)CC1 ONIQOQHATWINJY-UHFFFAOYSA-N 0.000 description 1
- YQXCVAGCMNFUMQ-UHFFFAOYSA-N capravirine Chemical compound C=1C(Cl)=CC(Cl)=CC=1SC1=C(C(C)C)N=C(COC(N)=O)N1CC1=CC=NC=C1 YQXCVAGCMNFUMQ-UHFFFAOYSA-N 0.000 description 1
- 229950008230 capravirine Drugs 0.000 description 1
- JSVCEVCSANKFDY-SFYZADRCSA-N carbacephem Chemical compound C1CC(C)=C(C(O)=O)N2C(=O)[C@@H](NC(=O)C)[C@H]21 JSVCEVCSANKFDY-SFYZADRCSA-N 0.000 description 1
- 125000003917 carbamoyl group Chemical group [H]N([H])C(*)=O 0.000 description 1
- 229940041011 carbapenems Drugs 0.000 description 1
- 229960004562 carboplatin Drugs 0.000 description 1
- 235000012730 carminic acid Nutrition 0.000 description 1
- 229960003261 carmofur Drugs 0.000 description 1
- 229960005243 carmustine Drugs 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 230000003197 catalytic effect Effects 0.000 description 1
- 238000005277 cation exchange chromatography Methods 0.000 description 1
- 229960000603 cefalotin Drugs 0.000 description 1
- 229960003012 cefamandole Drugs 0.000 description 1
- OLVCFLKTBJRLHI-AXAPSJFSSA-N cefamandole Chemical compound CN1N=NN=C1SCC1=C(C(O)=O)N2C(=O)[C@@H](NC(=O)[C@H](O)C=3C=CC=CC=3)[C@H]2SC1 OLVCFLKTBJRLHI-AXAPSJFSSA-N 0.000 description 1
- 229960004350 cefapirin Drugs 0.000 description 1
- 229960001139 cefazolin Drugs 0.000 description 1
- MLYYVTUWGNIJIB-BXKDBHETSA-N cefazolin Chemical compound S1C(C)=NN=C1SCC1=C(C(O)=O)N2C(=O)[C@@H](NC(=O)CN3N=NN=C3)[C@H]2SC1 MLYYVTUWGNIJIB-BXKDBHETSA-N 0.000 description 1
- 229960001817 cefbuperazone Drugs 0.000 description 1
- SMSRCGPDNDCXFR-CYWZMYCQSA-N cefbuperazone Chemical compound O=C1C(=O)N(CC)CCN1C(=O)N[C@H]([C@H](C)O)C(=O)N[C@]1(OC)C(=O)N2C(C(O)=O)=C(CSC=3N(N=NN=3)C)CS[C@@H]21 SMSRCGPDNDCXFR-CYWZMYCQSA-N 0.000 description 1
- 229960003719 cefdinir Drugs 0.000 description 1
- RTXOFQZKPXMALH-GHXIOONMSA-N cefdinir Chemical compound S1C(N)=NC(C(=N\O)\C(=O)N[C@@H]2C(N3C(=C(C=C)CS[C@@H]32)C(O)=O)=O)=C1 RTXOFQZKPXMALH-GHXIOONMSA-N 0.000 description 1
- 229960003791 cefmenoxime Drugs 0.000 description 1
- HJJDBAOLQAWBMH-YCRCPZNHSA-N cefmenoxime Chemical compound S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)\C(=N/OC)C=2N=C(N)SC=2)CC=1CSC1=NN=NN1C HJJDBAOLQAWBMH-YCRCPZNHSA-N 0.000 description 1
- 229960001958 cefodizime Drugs 0.000 description 1
- XDZKBRJLTGRPSS-BGZQYGJUSA-N cefodizime Chemical compound S([C@@H]1[C@@H](C(N1C=1C(O)=O)=O)NC(=O)\C(=N/OC)C=2N=C(N)SC=2)CC=1CSC1=NC(C)=C(CC(O)=O)S1 XDZKBRJLTGRPSS-BGZQYGJUSA-N 0.000 description 1
- 229960004682 cefoperazone Drugs 0.000 description 1
- GCFBRXLSHGKWDP-XCGNWRKASA-N cefoperazone Chemical compound O=C1C(=O)N(CC)CCN1C(=O)N[C@H](C=1C=CC(O)=CC=1)C(=O)N[C@@H]1C(=O)N2C(C(O)=O)=C(CSC=3N(N=NN=3)C)CS[C@@H]21 GCFBRXLSHGKWDP-XCGNWRKASA-N 0.000 description 1
- 229960005495 cefotetan Drugs 0.000 description 1
- SRZNHPXWXCNNDU-RHBCBLIFSA-N cefotetan Chemical compound N([C@]1(OC)C(N2C(=C(CSC=3N(N=NN=3)C)CS[C@@H]21)C(O)=O)=O)C(=O)C1SC(=C(C(N)=O)C(O)=O)S1 SRZNHPXWXCNNDU-RHBCBLIFSA-N 0.000 description 1
- 229960005446 cefpiramide Drugs 0.000 description 1
- PWAUCHMQEXVFJR-PMAPCBKXSA-N cefpiramide Chemical compound C1=NC(C)=CC(O)=C1C(=O)N[C@H](C=1C=CC(O)=CC=1)C(=O)N[C@@H]1C(=O)N2C(C(O)=O)=C(CSC=3N(N=NN=3)C)CS[C@@H]21 PWAUCHMQEXVFJR-PMAPCBKXSA-N 0.000 description 1
- 229960005090 cefpodoxime Drugs 0.000 description 1
- WYUSVOMTXWRGEK-HBWVYFAYSA-N cefpodoxime Chemical compound N([C@H]1[C@@H]2N(C1=O)C(=C(CS2)COC)C(O)=O)C(=O)C(=N/OC)\C1=CSC(N)=N1 WYUSVOMTXWRGEK-HBWVYFAYSA-N 0.000 description 1
- 229960002588 cefradine Drugs 0.000 description 1
- 229960003202 cefsulodin Drugs 0.000 description 1
- SYLKGLMBLAAGSC-QLVMHMETSA-N cefsulodin Chemical compound C1=CC(C(=O)N)=CC=[N+]1CC1=C(C([O-])=O)N2C(=O)[C@@H](NC(=O)[C@@H](C=3C=CC=CC=3)S(O)(=O)=O)[C@H]2SC1 SYLKGLMBLAAGSC-QLVMHMETSA-N 0.000 description 1
- 229960004366 ceftezole Drugs 0.000 description 1
- DZMVCVMFETWNIU-LDYMZIIASA-N ceftezole Chemical compound O=C([C@@H](NC(=O)CN1N=NN=C1)[C@H]1SC2)N1C(C(=O)O)=C2CSC1=NN=CS1 DZMVCVMFETWNIU-LDYMZIIASA-N 0.000 description 1
- 229960004086 ceftibuten Drugs 0.000 description 1
- UNJFKXSSGBWRBZ-BJCIPQKHSA-N ceftibuten Chemical compound S1C(N)=NC(C(=C\CC(O)=O)\C(=O)N[C@@H]2C(N3C(=CCS[C@@H]32)C(O)=O)=O)=C1 UNJFKXSSGBWRBZ-BJCIPQKHSA-N 0.000 description 1
- 229960001991 ceftizoxime Drugs 0.000 description 1
- NNULBSISHYWZJU-LLKWHZGFSA-N ceftizoxime Chemical compound N([C@@H]1C(N2C(=CCS[C@@H]21)C(O)=O)=O)C(=O)\C(=N/OC)C1=CSC(N)=N1 NNULBSISHYWZJU-LLKWHZGFSA-N 0.000 description 1
- 229960000590 celecoxib Drugs 0.000 description 1
- RZEKVGVHFLEQIL-UHFFFAOYSA-N celecoxib Chemical compound C1=CC(C)=CC=C1C1=CC(C(F)(F)F)=NN1C1=CC=C(S(N)(=O)=O)C=C1 RZEKVGVHFLEQIL-UHFFFAOYSA-N 0.000 description 1
- 238000010609 cell counting kit-8 assay Methods 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 230000032823 cell division Effects 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- VUFGUVLLDPOSBC-XRZFDKQNSA-M cephalothin sodium Chemical compound [Na+].N([C@H]1[C@@H]2N(C1=O)C(=C(CS2)COC(=O)C)C([O-])=O)C(=O)CC1=CC=CS1 VUFGUVLLDPOSBC-XRZFDKQNSA-M 0.000 description 1
- UCKZMPLVLCKKMO-LHLIQPBNSA-N cephamycin Chemical compound S1CC(C)=C(C(O)=O)N2C(=O)[C@@H](C)[C@]21OC UCKZMPLVLCKKMO-LHLIQPBNSA-N 0.000 description 1
- 150000001782 cephems Chemical class 0.000 description 1
- RDLPVSKMFDYCOR-UEKVPHQBSA-N cephradine Chemical compound C1([C@@H](N)C(=O)N[C@H]2[C@@H]3N(C2=O)C(=C(CS3)C)C(O)=O)=CCC=CC1 RDLPVSKMFDYCOR-UEKVPHQBSA-N 0.000 description 1
- SBNPWPIBESPSIF-MHWMIDJBSA-N cetrorelix Chemical compound C([C@@H](C(=O)N[C@H](CCCNC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@H](C)C(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](CC=1C=NC=CC=1)NC(=O)[C@@H](CC=1C=CC(Cl)=CC=1)NC(=O)[C@@H](CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(O)C=C1 SBNPWPIBESPSIF-MHWMIDJBSA-N 0.000 description 1
- 108700008462 cetrorelix Proteins 0.000 description 1
- 229960003230 cetrorelix Drugs 0.000 description 1
- 229960005395 cetuximab Drugs 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- DDTDNCYHLGRFBM-YZEKDTGTSA-N chembl2367892 Chemical compound CC(=O)N[C@H]1[C@@H](O)[C@H](O)[C@H](CO)O[C@H]1O[C@@H]([C@H]1C(N[C@@H](C2=CC(O)=CC(O[C@@H]3[C@H]([C@H](O)[C@H](O)[C@@H](CO)O3)O)=C2C=2C(O)=CC=C(C=2)[C@@H](NC(=O)[C@@H]2NC(=O)[C@@H]3C=4C=C(O)C=C(C=4)OC=4C(O)=CC=C(C=4)[C@@H](N)C(=O)N[C@H](CC=4C=C(Cl)C(O5)=CC=4)C(=O)N3)C(=O)N1)C(O)=O)=O)C(C=C1Cl)=CC=C1OC1=C(O[C@H]3[C@H]([C@@H](O)[C@H](O)[C@H](CO)O3)NC(C)=O)C5=CC2=C1 DDTDNCYHLGRFBM-YZEKDTGTSA-N 0.000 description 1
- 239000003638 chemical reducing agent Substances 0.000 description 1
- JCKYGMPEJWAADB-UHFFFAOYSA-N chlorambucil Chemical compound OC(=O)CCCC1=CC=C(N(CCCl)CCCl)C=C1 JCKYGMPEJWAADB-UHFFFAOYSA-N 0.000 description 1
- 229960004630 chlorambucil Drugs 0.000 description 1
- 229960005091 chloramphenicol Drugs 0.000 description 1
- WIIZWVCIJKGZOK-RKDXNWHRSA-N chloramphenicol Chemical compound ClC(Cl)C(=O)N[C@H](CO)[C@H](O)C1=CC=C([N+]([O-])=O)C=C1 WIIZWVCIJKGZOK-RKDXNWHRSA-N 0.000 description 1
- 229960003677 chloroquine Drugs 0.000 description 1
- WHTVZRBIWZFKQO-UHFFFAOYSA-N chloroquine Natural products ClC1=CC=C2C(NC(C)CCCN(CC)CC)=CC=NC2=C1 WHTVZRBIWZFKQO-UHFFFAOYSA-N 0.000 description 1
- CYDMQBQPVICBEU-UHFFFAOYSA-N chlorotetracycline Natural products C1=CC(Cl)=C2C(O)(C)C3CC4C(N(C)C)C(O)=C(C(N)=O)C(=O)C4(O)C(O)=C3C(=O)C2=C1O CYDMQBQPVICBEU-UHFFFAOYSA-N 0.000 description 1
- SOYKEARSMXGVTM-UHFFFAOYSA-N chlorphenamine Chemical compound C=1C=CC=NC=1C(CCN(C)C)C1=CC=C(Cl)C=C1 SOYKEARSMXGVTM-UHFFFAOYSA-N 0.000 description 1
- 229960003291 chlorphenamine Drugs 0.000 description 1
- 229960004475 chlortetracycline Drugs 0.000 description 1
- CYDMQBQPVICBEU-XRNKAMNCSA-N chlortetracycline Chemical compound C1=CC(Cl)=C2[C@](O)(C)[C@H]3C[C@H]4[C@H](N(C)C)C(O)=C(C(N)=O)C(=O)[C@@]4(O)C(O)=C3C(=O)C2=C1O CYDMQBQPVICBEU-XRNKAMNCSA-N 0.000 description 1
- 235000019365 chlortetracycline Nutrition 0.000 description 1
- 235000012000 cholesterol Nutrition 0.000 description 1
- 108010039524 chondroitin sulfate proteoglycan 4 Proteins 0.000 description 1
- 229960001265 ciclosporin Drugs 0.000 description 1
- 229960000724 cidofovir Drugs 0.000 description 1
- 229950009003 cilengitide Drugs 0.000 description 1
- AMLYAMJWYAIXIA-VWNVYAMZSA-N cilengitide Chemical compound N1C(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](C(C)C)N(C)C(=O)[C@H]1CC1=CC=CC=C1 AMLYAMJWYAIXIA-VWNVYAMZSA-N 0.000 description 1
- 229960003405 ciprofloxacin Drugs 0.000 description 1
- 229960004316 cisplatin Drugs 0.000 description 1
- DQLATGHUWYMOKM-UHFFFAOYSA-L cisplatin Chemical compound N[Pt](N)(Cl)Cl DQLATGHUWYMOKM-UHFFFAOYSA-L 0.000 description 1
- 229960002626 clarithromycin Drugs 0.000 description 1
- AGOYDEPGAOXOCK-KCBOHYOISA-N clarithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H](O)[C@@H](C)C(=O)[C@H](C)C[C@](C)([C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)OC)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 AGOYDEPGAOXOCK-KCBOHYOISA-N 0.000 description 1
- HZZVJAQRINQKSD-PBFISZAISA-N clavulanic acid Chemical compound OC(=O)[C@H]1C(=C/CO)/O[C@@H]2CC(=O)N21 HZZVJAQRINQKSD-PBFISZAISA-N 0.000 description 1
- 229960003324 clavulanic acid Drugs 0.000 description 1
- 229960003346 colistin Drugs 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 238000009833 condensation Methods 0.000 description 1
- 230000005494 condensation Effects 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- 150000004775 coumarins Chemical class 0.000 description 1
- 108010006226 cryptophycin Proteins 0.000 description 1
- 108010089438 cryptophycin 1 Proteins 0.000 description 1
- 108010090203 cryptophycin 8 Proteins 0.000 description 1
- 108010082025 cyan fluorescent protein Proteins 0.000 description 1
- 150000001924 cycloalkanes Chemical class 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- ZPWOOKQUDFIEIX-UHFFFAOYSA-N cyclooctyne Chemical compound C1CCCC#CCC1 ZPWOOKQUDFIEIX-UHFFFAOYSA-N 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 229960003077 cycloserine Drugs 0.000 description 1
- 229930182912 cyclosporin Natural products 0.000 description 1
- 125000000151 cysteine group Chemical group N[C@@H](CS)C(=O)* 0.000 description 1
- 229940104302 cytosine Drugs 0.000 description 1
- 239000002254 cytotoxic agent Substances 0.000 description 1
- 231100000263 cytotoxicity test Toxicity 0.000 description 1
- 229960002488 dalbavancin Drugs 0.000 description 1
- 108700009376 dalbavancin Proteins 0.000 description 1
- POZRVZJJTULAOH-LHZXLZLDSA-N danazol Chemical compound C1[C@]2(C)[C@H]3CC[C@](C)([C@](CC4)(O)C#C)[C@@H]4[C@@H]3CCC2=CC2=C1C=NO2 POZRVZJJTULAOH-LHZXLZLDSA-N 0.000 description 1
- 229960000766 danazol Drugs 0.000 description 1
- 125000001295 dansyl group Chemical group [H]C1=C([H])C(N(C([H])([H])[H])C([H])([H])[H])=C2C([H])=C([H])C([H])=C(C2=C1[H])S(*)(=O)=O 0.000 description 1
- DOAKLVKFURWEDJ-QCMAZARJSA-N daptomycin Chemical compound C([C@H]1C(=O)O[C@H](C)[C@@H](C(NCC(=O)N[C@@H](CCCN)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C)C(=O)N[C@@H](CC(O)=O)C(=O)NCC(=O)N[C@H](CO)C(=O)N[C@H](C(=O)N1)[C@H](C)CC(O)=O)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@@H](CC(N)=O)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)CCCCCCCCC)C(=O)C1=CC=CC=C1N DOAKLVKFURWEDJ-QCMAZARJSA-N 0.000 description 1
- 229960005484 daptomycin Drugs 0.000 description 1
- 229960005107 darunavir Drugs 0.000 description 1
- CJBJHOAVZSMMDJ-HEXNFIEUSA-N darunavir Chemical compound C([C@@H]([C@H](O)CN(CC(C)C)S(=O)(=O)C=1C=CC(N)=CC=1)NC(=O)O[C@@H]1[C@@H]2CCO[C@@H]2OC1)C1=CC=CC=C1 CJBJHOAVZSMMDJ-HEXNFIEUSA-N 0.000 description 1
- 229960002448 dasatinib Drugs 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 229960000958 deferoxamine Drugs 0.000 description 1
- 229960002272 degarelix Drugs 0.000 description 1
- MEUCPCLKGZSHTA-XYAYPHGZSA-N degarelix Chemical compound C([C@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCNC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N[C@H](C)C(N)=O)NC(=O)[C@H](CC=1C=CC(NC(=O)[C@H]2NC(=O)NC(=O)C2)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](CC=1C=NC=CC=1)NC(=O)[C@@H](CC=1C=CC(Cl)=CC=1)NC(=O)[C@@H](CC=1C=C2C=CC=CC2=CC=1)NC(C)=O)C1=CC=C(NC(N)=O)C=C1 MEUCPCLKGZSHTA-XYAYPHGZSA-N 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- FMGSKLZLMKYGDP-USOAJAOKSA-N dehydroepiandrosterone Chemical compound C1[C@@H](O)CC[C@]2(C)[C@H]3CC[C@](C)(C(CC4)=O)[C@@H]4[C@@H]3CC=C21 FMGSKLZLMKYGDP-USOAJAOKSA-N 0.000 description 1
- 229950007920 demecycline Drugs 0.000 description 1
- JCSGAUKCDAVARS-UHFFFAOYSA-N demethyltetracycline Natural products CN(C1C(=C(C(C2(C(=C3C(C4=C(C=CC=C4C(C3CC12)O)O)=O)O)O)=O)C(=O)N)O)C JCSGAUKCDAVARS-UHFFFAOYSA-N 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- NIJJYAXOARWZEE-UHFFFAOYSA-N di-n-propyl-acetic acid Natural products CCCC(C(O)=O)CCC NIJJYAXOARWZEE-UHFFFAOYSA-N 0.000 description 1
- 150000004985 diamines Chemical class 0.000 description 1
- 229960003807 dibekacin Drugs 0.000 description 1
- JJCQSGDBDPYCEO-XVZSLQNASA-N dibekacin Chemical compound O1[C@H](CN)CC[C@@H](N)[C@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N JJCQSGDBDPYCEO-XVZSLQNASA-N 0.000 description 1
- 229950001733 difloxacin Drugs 0.000 description 1
- NOCJXYPHIIZEHN-UHFFFAOYSA-N difloxacin Chemical compound C1CN(C)CCN1C(C(=C1)F)=CC2=C1C(=O)C(C(O)=O)=CN2C1=CC=C(F)C=C1 NOCJXYPHIIZEHN-UHFFFAOYSA-N 0.000 description 1
- 108020001096 dihydrofolate reductase Proteins 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 229910001873 dinitrogen Inorganic materials 0.000 description 1
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 description 1
- 238000000375 direct analysis in real time Methods 0.000 description 1
- 229960004100 dirithromycin Drugs 0.000 description 1
- WLOHNSSYAXHWNR-NXPDYKKBSA-N dirithromycin Chemical compound O([C@@H]1[C@@H](C)C(=O)O[C@@H]([C@@]([C@H]2O[C@H](COCCOC)N[C@H]([C@@H]2C)[C@H](C)C[C@@](C)(O)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C)(C)O)CC)[C@H]1C[C@@](C)(OC)[C@@H](O)[C@H](C)O1 WLOHNSSYAXHWNR-NXPDYKKBSA-N 0.000 description 1
- NNEGMXMRXKXKMQ-ZEOBNFASSA-N disnogamycin Chemical compound CO[C@@H]1[C@@](OC)(C)[C@@H](OC)[C@H](C)O[C@H]1O[C@@H]1C2=C(O)C(C(=O)C3=C(O)C=C4[C@@]5(C)O[C@H]([C@H]([C@@H]([C@H]5O)N(C)C)O)OC4=C3C3=O)=C3C=C2C[C@@](C)(O)C1 NNEGMXMRXKXKMQ-ZEOBNFASSA-N 0.000 description 1
- 229960002563 disulfiram Drugs 0.000 description 1
- 239000003534 dna topoisomerase inhibitor Substances 0.000 description 1
- 229960003668 docetaxel Drugs 0.000 description 1
- 229960000735 docosanol Drugs 0.000 description 1
- 125000003438 dodecyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000003291 dopaminomimetic effect Effects 0.000 description 1
- 229960000895 doripenem Drugs 0.000 description 1
- AVAACINZEOAHHE-VFZPANTDSA-N doripenem Chemical compound C=1([C@H](C)[C@@H]2[C@H](C(N2C=1C(O)=O)=O)[C@H](O)C)S[C@@H]1CN[C@H](CNS(N)(=O)=O)C1 AVAACINZEOAHHE-VFZPANTDSA-N 0.000 description 1
- 229960003722 doxycycline Drugs 0.000 description 1
- 238000012362 drug development process Methods 0.000 description 1
- 238000012063 dual-affinity re-targeting Methods 0.000 description 1
- XPOQHMRABVBWPR-ZDUSSCGKSA-N efavirenz Chemical compound C([C@]1(C2=CC(Cl)=CC=C2NC(=O)O1)C(F)(F)F)#CC1CC1 XPOQHMRABVBWPR-ZDUSSCGKSA-N 0.000 description 1
- 229960003804 efavirenz Drugs 0.000 description 1
- 229960002759 eflornithine Drugs 0.000 description 1
- 229960003586 elvitegravir Drugs 0.000 description 1
- JUZYLCPPVHEVSV-LJQANCHMSA-N elvitegravir Chemical compound COC1=CC=2N([C@H](CO)C(C)C)C=C(C(O)=O)C(=O)C=2C=C1CC1=CC=CC(Cl)=C1F JUZYLCPPVHEVSV-LJQANCHMSA-N 0.000 description 1
- 229950006528 elvucitabine Drugs 0.000 description 1
- 239000010976 emerald Substances 0.000 description 1
- 229910052876 emerald Inorganic materials 0.000 description 1
- MLILORUFDVLTSP-UHFFFAOYSA-N emivirine Chemical compound O=C1NC(=O)N(COCC)C(CC=2C=CC=CC=2)=C1C(C)C MLILORUFDVLTSP-UHFFFAOYSA-N 0.000 description 1
- 229950002002 emivirine Drugs 0.000 description 1
- 229960000366 emtricitabine Drugs 0.000 description 1
- 229960002062 enfuvirtide Drugs 0.000 description 1
- PEASPLKKXBYDKL-FXEVSJAOSA-N enfuvirtide Chemical compound C([C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(C)=O)[C@@H](C)O)[C@@H](C)CC)C1=CN=CN1 PEASPLKKXBYDKL-FXEVSJAOSA-N 0.000 description 1
- 108010048367 enhanced green fluorescent protein Proteins 0.000 description 1
- 229960002549 enoxacin Drugs 0.000 description 1
- IDYZIJYBMGIQMJ-UHFFFAOYSA-N enoxacin Chemical compound N1=C2N(CC)C=C(C(O)=O)C(=O)C2=CC(F)=C1N1CCNCC1 IDYZIJYBMGIQMJ-UHFFFAOYSA-N 0.000 description 1
- 229960000740 enrofloxacin Drugs 0.000 description 1
- HKSZLNNOFSGOKW-UHFFFAOYSA-N ent-staurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(C)O1 HKSZLNNOFSGOKW-UHFFFAOYSA-N 0.000 description 1
- 229960000980 entecavir Drugs 0.000 description 1
- YXPVEXCTPGULBZ-WQYNNSOESA-N entecavir hydrate Chemical compound O.C1=NC=2C(=O)NC(N)=NC=2N1[C@H]1C[C@H](O)[C@@H](CO)C1=C YXPVEXCTPGULBZ-WQYNNSOESA-N 0.000 description 1
- INVTYAOGFAGBOE-UHFFFAOYSA-N entinostat Chemical compound NC1=CC=CC=C1NC(=O)C(C=C1)=CC=C1CNC(=O)OCC1=CC=CN=C1 INVTYAOGFAGBOE-UHFFFAOYSA-N 0.000 description 1
- 229950005837 entinostat Drugs 0.000 description 1
- YQGOJNYOYNNSMM-UHFFFAOYSA-N eosin Chemical compound [Na+].OC(=O)C1=CC=CC=C1C1=C2C=C(Br)C(=O)C(Br)=C2OC2=C(Br)C(O)=C(Br)C=C21 YQGOJNYOYNNSMM-UHFFFAOYSA-N 0.000 description 1
- 229960002061 ergocalciferol Drugs 0.000 description 1
- 229960002770 ertapenem Drugs 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 229960001842 estramustine Drugs 0.000 description 1
- FRPJXPJMRWBBIH-RBRWEJTLSA-N estramustine Chemical compound ClCCN(CCCl)C(=O)OC1=CC=C2[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 FRPJXPJMRWBBIH-RBRWEJTLSA-N 0.000 description 1
- 239000000328 estrogen antagonist Substances 0.000 description 1
- 229960000403 etanercept Drugs 0.000 description 1
- 229960000285 ethambutol Drugs 0.000 description 1
- 229960005542 ethidium bromide Drugs 0.000 description 1
- ZMMJGEGLRURXTF-UHFFFAOYSA-N ethidium bromide Chemical compound [Br-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CC)=C1C1=CC=CC=C1 ZMMJGEGLRURXTF-UHFFFAOYSA-N 0.000 description 1
- WHRIKZCFRVTHJH-UHFFFAOYSA-N ethylhydrazine Chemical compound CCNN WHRIKZCFRVTHJH-UHFFFAOYSA-N 0.000 description 1
- LIQODXNTTZAGID-OCBXBXKTSA-N etoposide phosphate Chemical compound COC1=C(OP(O)(O)=O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 LIQODXNTTZAGID-OCBXBXKTSA-N 0.000 description 1
- 229960000752 etoposide phosphate Drugs 0.000 description 1
- 229960002049 etravirine Drugs 0.000 description 1
- PYGWGZALEOIKDF-UHFFFAOYSA-N etravirine Chemical compound CC1=CC(C#N)=CC(C)=C1OC1=NC(NC=2C=CC(=CC=2)C#N)=NC(N)=C1Br PYGWGZALEOIKDF-UHFFFAOYSA-N 0.000 description 1
- 229960004396 famciclovir Drugs 0.000 description 1
- GGXKWVWZWMLJEH-UHFFFAOYSA-N famcyclovir Chemical compound N1=C(N)N=C2N(CCC(COC(=O)C)COC(C)=O)C=NC2=C1 GGXKWVWZWMLJEH-UHFFFAOYSA-N 0.000 description 1
- 229960000379 faropenem Drugs 0.000 description 1
- IKIBJHWXDSKRKV-UHFFFAOYSA-N fijianolide B Natural products CC1CC(=C)CC(O)C2OC2CC(OC(=O)C=C/CC3OC(C)(CC=C3)C1)C(O)C=CC4CC(=CCO4)C IKIBJHWXDSKRKV-UHFFFAOYSA-N 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 229930003935 flavonoid Natural products 0.000 description 1
- 150000002215 flavonoids Chemical class 0.000 description 1
- 235000017173 flavonoids Nutrition 0.000 description 1
- 229960003760 florfenicol Drugs 0.000 description 1
- ODKNJVUHOIMIIZ-RRKCRQDMSA-N floxuridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(F)=C1 ODKNJVUHOIMIIZ-RRKCRQDMSA-N 0.000 description 1
- 229960000961 floxuridine Drugs 0.000 description 1
- 229960000676 flunisolide Drugs 0.000 description 1
- 229960002949 fluorouracil Drugs 0.000 description 1
- 229960002074 flutamide Drugs 0.000 description 1
- MKXKFYHWDHIYRV-UHFFFAOYSA-N flutamide Chemical compound CC(C)C(=O)NC1=CC=C([N+]([O-])=O)C(C(F)(F)F)=C1 MKXKFYHWDHIYRV-UHFFFAOYSA-N 0.000 description 1
- 229960000289 fluticasone propionate Drugs 0.000 description 1
- WMWTYOKRWGGJOA-CENSZEJFSA-N fluticasone propionate Chemical compound C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@]1(F)[C@@H]2[C@@H]2C[C@@H](C)[C@@](C(=O)SCF)(OC(=O)CC)[C@@]2(C)C[C@@H]1O WMWTYOKRWGGJOA-CENSZEJFSA-N 0.000 description 1
- 239000004052 folic acid antagonist Substances 0.000 description 1
- 150000002224 folic acids Chemical class 0.000 description 1
- 229960000308 fosfomycin Drugs 0.000 description 1
- YMDXZJFXQJVXBF-STHAYSLISA-N fosfomycin Chemical compound C[C@@H]1O[C@@H]1P(O)(O)=O YMDXZJFXQJVXBF-STHAYSLISA-N 0.000 description 1
- 229960003704 framycetin Drugs 0.000 description 1
- PGBHMTALBVVCIT-VCIWKGPPSA-N framycetin Chemical compound N[C@@H]1[C@@H](O)[C@H](O)[C@H](CN)O[C@@H]1O[C@H]1[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](N)C[C@@H](N)[C@@H]2O)O[C@@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](CN)O2)N)O[C@@H]1CO PGBHMTALBVVCIT-VCIWKGPPSA-N 0.000 description 1
- 229960001625 furazolidone Drugs 0.000 description 1
- PLHJDBGFXBMTGZ-WEVVVXLNSA-N furazolidone Chemical compound O1C([N+](=O)[O-])=CC=C1\C=N\N1C(=O)OCC1 PLHJDBGFXBMTGZ-WEVVVXLNSA-N 0.000 description 1
- 229940125777 fusion inhibitor Drugs 0.000 description 1
- FBPFZTCFMRRESA-GUCUJZIJSA-N galactitol Chemical compound OC[C@H](O)[C@@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-GUCUJZIJSA-N 0.000 description 1
- 229940044658 gallium nitrate Drugs 0.000 description 1
- 229960003923 gatifloxacin Drugs 0.000 description 1
- 229960002584 gefitinib Drugs 0.000 description 1
- XGALLCVXEZPNRQ-UHFFFAOYSA-N gefitinib Chemical compound C=12C=C(OCCCN3CCOCC3)C(OC)=CC2=NC=NC=1NC1=CC=C(F)C(Cl)=C1 XGALLCVXEZPNRQ-UHFFFAOYSA-N 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 229960005277 gemcitabine Drugs 0.000 description 1
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 1
- 229960003170 gemifloxacin Drugs 0.000 description 1
- ZRCVYEYHRGVLOC-HYARGMPZSA-N gemifloxacin Chemical compound C1C(CN)C(=N/OC)/CN1C(C(=C1)F)=NC2=C1C(=O)C(C(O)=O)=CN2C1CC1 ZRCVYEYHRGVLOC-HYARGMPZSA-N 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 229960002518 gentamicin Drugs 0.000 description 1
- 229950010415 givinostat Drugs 0.000 description 1
- 125000003827 glycol group Chemical group 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- 108010064365 glycyl- arginyl-glycyl-aspartyl-seryl-prolyl-lysine Proteins 0.000 description 1
- 229960001442 gonadorelin Drugs 0.000 description 1
- 239000001056 green pigment Substances 0.000 description 1
- SZWIAFVYPPMZML-YNEHKIRRSA-N heptyl n-[5-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-6-oxo-1,4-dihydro-1,3,5-triazin-2-yl]carbamate Chemical compound C1NC(NC(=O)OCCCCCCC)=NC(=O)N1[C@@H]1O[C@H](CO)[C@@H](O)C1 SZWIAFVYPPMZML-YNEHKIRRSA-N 0.000 description 1
- MCAHMSDENAOJFZ-BVXDHVRPSA-N herbimycin Chemical compound N1C(=O)\C(C)=C\C=C/[C@H](OC)[C@@H](OC(N)=O)\C(C)=C\[C@H](C)[C@@H](OC)[C@@H](OC)C[C@H](C)[C@@H](OC)C2=CC(=O)C=C1C2=O MCAHMSDENAOJFZ-BVXDHVRPSA-N 0.000 description 1
- 229930193320 herbimycin Natural products 0.000 description 1
- 125000001072 heteroaryl group Chemical group 0.000 description 1
- 125000000592 heterocycloalkyl group Chemical group 0.000 description 1
- UUVWYPNAQBNQJQ-UHFFFAOYSA-N hexamethylmelamine Chemical compound CN(C)C1=NC(N(C)C)=NC(N(C)C)=N1 UUVWYPNAQBNQJQ-UHFFFAOYSA-N 0.000 description 1
- 239000003276 histone deacetylase inhibitor Substances 0.000 description 1
- 229940121372 histone deacetylase inhibitor Drugs 0.000 description 1
- 108700020746 histrelin Proteins 0.000 description 1
- HHXHVIJIIXKSOE-QILQGKCVSA-N histrelin Chemical compound CCNC(=O)[C@@H]1CCCN1C(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)CC(N=C1)=CN1CC1=CC=CC=C1 HHXHVIJIIXKSOE-QILQGKCVSA-N 0.000 description 1
- 229960002193 histrelin Drugs 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 238000001794 hormone therapy Methods 0.000 description 1
- 150000007857 hydrazones Chemical class 0.000 description 1
- 229960000890 hydrocortisone Drugs 0.000 description 1
- 229920001477 hydrophilic polymer Polymers 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 125000001165 hydrophobic group Chemical group 0.000 description 1
- 229960004171 hydroxychloroquine Drugs 0.000 description 1
- XXSMGPRMXLTPCZ-UHFFFAOYSA-N hydroxychloroquine Chemical compound ClC1=CC=C2C(NC(C)CCCN(CCO)CC)=CC=NC2=C1 XXSMGPRMXLTPCZ-UHFFFAOYSA-N 0.000 description 1
- GRRNUXAQVGOGFE-NZSRVPFOSA-N hygromycin B Chemical compound O[C@@H]1[C@@H](NC)C[C@@H](N)[C@H](O)[C@H]1O[C@H]1[C@H]2O[C@@]3([C@@H]([C@@H](O)[C@@H](O)[C@@H](C(N)CO)O3)O)O[C@H]2[C@@H](O)[C@@H](CO)O1 GRRNUXAQVGOGFE-NZSRVPFOSA-N 0.000 description 1
- 229940097277 hygromycin b Drugs 0.000 description 1
- 229950010245 ibalizumab Drugs 0.000 description 1
- 229940015872 ibandronate Drugs 0.000 description 1
- HOMGKSMUEGBAAB-UHFFFAOYSA-N ifosfamide Chemical compound ClCCNP1(=O)OCCCN1CCCl HOMGKSMUEGBAAB-UHFFFAOYSA-N 0.000 description 1
- 229960001101 ifosfamide Drugs 0.000 description 1
- 229960002411 imatinib Drugs 0.000 description 1
- KTUFNOKKBVMGRW-UHFFFAOYSA-N imatinib Chemical compound C1CN(C)CCN1CC1=CC=C(C(=O)NC=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)C=C1 KTUFNOKKBVMGRW-UHFFFAOYSA-N 0.000 description 1
- 150000002466 imines Chemical class 0.000 description 1
- 229960002751 imiquimod Drugs 0.000 description 1
- DOUYETYNHWVLEO-UHFFFAOYSA-N imiquimod Chemical compound C1=CC=CC2=C3N(CC(C)C)C=NC3=C(N)N=C21 DOUYETYNHWVLEO-UHFFFAOYSA-N 0.000 description 1
- 229960001936 indinavir Drugs 0.000 description 1
- CBVCZFGXHXORBI-PXQQMZJSSA-N indinavir Chemical compound C([C@H](N(CC1)C[C@@H](O)C[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H]2C3=CC=CC=C3C[C@H]2O)C(=O)NC(C)(C)C)N1CC1=CC=CN=C1 CBVCZFGXHXORBI-PXQQMZJSSA-N 0.000 description 1
- PNDZEEPOYCVIIY-UHFFFAOYSA-N indo-1 Chemical compound CC1=CC=C(N(CC(O)=O)CC(O)=O)C(OCCOC=2C(=CC=C(C=2)C=2N=C3[CH]C(=CC=C3C=2)C(O)=O)N(CC(O)=O)CC(O)=O)=C1 PNDZEEPOYCVIIY-UHFFFAOYSA-N 0.000 description 1
- 208000015181 infectious disease Diseases 0.000 description 1
- 229960000598 infliximab Drugs 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 229950002133 iniparib Drugs 0.000 description 1
- 239000002348 inosinate dehydrogenase inhibitor Substances 0.000 description 1
- 229940124524 integrase inhibitor Drugs 0.000 description 1
- 239000002850 integrase inhibitor Substances 0.000 description 1
- 102000006495 integrins Human genes 0.000 description 1
- 108010044426 integrins Proteins 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 229960003130 interferon gamma Drugs 0.000 description 1
- 201000002313 intestinal cancer Diseases 0.000 description 1
- 230000009545 invasion Effects 0.000 description 1
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 229960004144 josamycin Drugs 0.000 description 1
- XJSFLOJWULLJQS-NGVXBBESSA-N josamycin Chemical compound CO[C@H]1[C@H](OC(C)=O)CC(=O)O[C@H](C)C\C=C\C=C\[C@H](O)[C@H](C)C[C@H](CC=O)[C@@H]1O[C@H]1[C@H](O)[C@@H](N(C)C)[C@H](O[C@@H]2O[C@@H](C)[C@H](OC(=O)CC(C)C)[C@](C)(O)C2)[C@@H](C)O1 XJSFLOJWULLJQS-NGVXBBESSA-N 0.000 description 1
- 229960000318 kanamycin Drugs 0.000 description 1
- 229930027917 kanamycin Natural products 0.000 description 1
- SBUJHOSQTJFQJX-NOAMYHISSA-N kanamycin Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N SBUJHOSQTJFQJX-NOAMYHISSA-N 0.000 description 1
- 229930182823 kanamycin A Natural products 0.000 description 1
- SKKLOUVUUNMCJE-FQSMHNGLSA-N kanamycin B Chemical compound N[C@@H]1[C@@H](O)[C@H](O)[C@@H](CN)O[C@@H]1O[C@H]1[C@H](O)[C@@H](O[C@@H]2[C@@H]([C@@H](N)[C@H](O)[C@@H](CO)O2)O)[C@H](N)C[C@@H]1N SKKLOUVUUNMCJE-FQSMHNGLSA-N 0.000 description 1
- 229940043355 kinase inhibitor Drugs 0.000 description 1
- 150000002596 lactones Chemical class 0.000 description 1
- 229960001627 lamivudine Drugs 0.000 description 1
- JTEGQNOMFQHVDC-NKWVEPMBSA-N lamivudine Chemical compound O=C1N=C(N)C=CN1[C@H]1O[C@@H](CO)SC1 JTEGQNOMFQHVDC-NKWVEPMBSA-N 0.000 description 1
- 229960002437 lanreotide Drugs 0.000 description 1
- 229960004891 lapatinib Drugs 0.000 description 1
- BCFGMOOMADDAQU-UHFFFAOYSA-N lapatinib Chemical compound O1C(CNCCS(=O)(=O)C)=CC=C1C1=CC=C(N=CN=C2NC=3C=C(Cl)C(OCC=4C=C(F)C=CC=4)=CC=3)C2=C1 BCFGMOOMADDAQU-UHFFFAOYSA-N 0.000 description 1
- 229960000433 latamoxef Drugs 0.000 description 1
- MSBQEQDLFWWWMV-XZZGLLCESA-N laulimalide Chemical compound C(/[C@H](O)[C@H]1OC(=O)\C=C/C[C@@H]2C=CC[C@H](O2)C[C@H](CC(=C)C[C@H](O)[C@@H]2O[C@H]2C1)C)=C\[C@@H]1CC(C)=CCO1 MSBQEQDLFWWWMV-XZZGLLCESA-N 0.000 description 1
- 239000002523 lectin Substances 0.000 description 1
- 229960004942 lenalidomide Drugs 0.000 description 1
- GOTYRUGSSMKFNF-UHFFFAOYSA-N lenalidomide Chemical compound C1C=2C(N)=CC=CC=2C(=O)N1C1CCC(=O)NC1=O GOTYRUGSSMKFNF-UHFFFAOYSA-N 0.000 description 1
- 229940115286 lentinan Drugs 0.000 description 1
- WOSKHXYHFSIKNG-UHFFFAOYSA-N lenvatinib Chemical compound C=12C=C(C(N)=O)C(OC)=CC2=NC=CC=1OC(C=C1Cl)=CC=C1NC(=O)NC1CC1 WOSKHXYHFSIKNG-UHFFFAOYSA-N 0.000 description 1
- 229960003784 lenvatinib Drugs 0.000 description 1
- 210000000265 leukocyte Anatomy 0.000 description 1
- 229960003376 levofloxacin Drugs 0.000 description 1
- 229960005535 lidamycin Drugs 0.000 description 1
- 229960005287 lincomycin Drugs 0.000 description 1
- OJMMVQQUTAEWLP-KIDUDLJLSA-N lincomycin Chemical compound CN1C[C@H](CCC)C[C@H]1C(=O)N[C@H]([C@@H](C)O)[C@@H]1[C@H](O)[C@H](O)[C@@H](O)[C@@H](SC)O1 OJMMVQQUTAEWLP-KIDUDLJLSA-N 0.000 description 1
- 229960003907 linezolid Drugs 0.000 description 1
- TYZROVQLWOKYKF-ZDUSSCGKSA-N linezolid Chemical compound O=C1O[C@@H](CNC(=O)C)CN1C(C=C1F)=CC=C1N1CCOCC1 TYZROVQLWOKYKF-ZDUSSCGKSA-N 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 229960002422 lomefloxacin Drugs 0.000 description 1
- ZEKZLJVOYLTDKK-UHFFFAOYSA-N lomefloxacin Chemical compound FC1=C2N(CC)C=C(C(O)=O)C(=O)C2=CC(F)=C1N1CCNC(C)C1 ZEKZLJVOYLTDKK-UHFFFAOYSA-N 0.000 description 1
- 229960002247 lomustine Drugs 0.000 description 1
- 229960004525 lopinavir Drugs 0.000 description 1
- PCZOHLXUXFIOCF-BXMDZJJMSA-N lovastatin Chemical compound C([C@H]1[C@@H](C)C=CC2=C[C@H](C)C[C@@H]([C@H]12)OC(=O)[C@@H](C)CC)C[C@@H]1C[C@@H](O)CC(=O)O1 PCZOHLXUXFIOCF-BXMDZJJMSA-N 0.000 description 1
- 229960004844 lovastatin Drugs 0.000 description 1
- QLJODMDSTUBWDW-UHFFFAOYSA-N lovastatin hydroxy acid Natural products C1=CC(C)C(CCC(O)CC(O)CC(O)=O)C2C(OC(=O)C(C)CC)CC(C)C=C21 QLJODMDSTUBWDW-UHFFFAOYSA-N 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- 108091005958 mTurquoise2 Proteins 0.000 description 1
- 239000003120 macrolide antibiotic agent Substances 0.000 description 1
- 229940041033 macrolides Drugs 0.000 description 1
- 230000014759 maintenance of location Effects 0.000 description 1
- 229940107698 malachite green Drugs 0.000 description 1
- FDZZZRQASAIRJF-UHFFFAOYSA-M malachite green Chemical compound [Cl-].C1=CC(N(C)C)=CC=C1C(C=1C=CC=CC=1)=C1C=CC(=[N+](C)C)C=C1 FDZZZRQASAIRJF-UHFFFAOYSA-M 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- 229960004710 maraviroc Drugs 0.000 description 1
- GSNHKUDZZFZSJB-QYOOZWMWSA-N maraviroc Chemical compound CC(C)C1=NN=C(C)N1[C@@H]1C[C@H](N2CC[C@H](NC(=O)C3CCC(F)(F)CC3)C=3C=CC=CC=3)CC[C@H]2C1 GSNHKUDZZFZSJB-QYOOZWMWSA-N 0.000 description 1
- 229950002736 marizomib Drugs 0.000 description 1
- 238000004949 mass spectrometry Methods 0.000 description 1
- 238000001819 mass spectrum Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000035800 maturation Effects 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 230000010534 mechanism of action Effects 0.000 description 1
- 229960001786 megestrol Drugs 0.000 description 1
- JBVNBBXAMBZTMQ-CEGNMAFCSA-N megestrol Chemical compound C1=CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@@](C(=O)C)(O)[C@@]1(C)CC2 JBVNBBXAMBZTMQ-CEGNMAFCSA-N 0.000 description 1
- 229960001929 meloxicam Drugs 0.000 description 1
- GLVAUDGFNGKCSF-UHFFFAOYSA-N mercaptopurine Chemical compound S=C1NC=NC2=C1NC=N2 GLVAUDGFNGKCSF-UHFFFAOYSA-N 0.000 description 1
- 229960001428 mercaptopurine Drugs 0.000 description 1
- 229960002260 meropenem Drugs 0.000 description 1
- DMJNNHOOLUXYBV-PQTSNVLCSA-N meropenem Chemical compound C=1([C@H](C)[C@@H]2[C@H](C(N2C=1C(O)=O)=O)[C@H](O)C)S[C@@H]1CN[C@H](C(=O)N(C)C)C1 DMJNNHOOLUXYBV-PQTSNVLCSA-N 0.000 description 1
- ANZJBCHSOXCCRQ-FKUXLPTCSA-N mertansine Chemical compound CO[C@@H]([C@@]1(O)C[C@H](OC(=O)N1)[C@@H](C)[C@@H]1O[C@@]1(C)[C@@H](OC(=O)[C@H](C)N(C)C(=O)CCS)CC(=O)N1C)\C=C\C=C(C)\CC2=CC(OC)=C(Cl)C1=C2 ANZJBCHSOXCCRQ-FKUXLPTCSA-N 0.000 description 1
- 229940042016 methacycline Drugs 0.000 description 1
- FNEZBBILNYNQGC-UHFFFAOYSA-N methyl 2-(3,6-diamino-9h-xanthen-9-yl)benzoate Chemical compound COC(=O)C1=CC=CC=C1C1C2=CC=C(N)C=C2OC2=CC(N)=CC=C21 FNEZBBILNYNQGC-UHFFFAOYSA-N 0.000 description 1
- 229960003085 meticillin Drugs 0.000 description 1
- 229960000282 metronidazole Drugs 0.000 description 1
- VAOCPAMSLUNLGC-UHFFFAOYSA-N metronidazole Chemical compound CC1=NC=C([N+]([O-])=O)N1CCO VAOCPAMSLUNLGC-UHFFFAOYSA-N 0.000 description 1
- YPBATNHYBCGSSN-VWPFQQQWSA-N mezlocillin Chemical compound N([C@@H](C(=O)N[C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C=1C=CC=CC=1)C(=O)N1CCN(S(C)(=O)=O)C1=O YPBATNHYBCGSSN-VWPFQQQWSA-N 0.000 description 1
- 229960000198 mezlocillin Drugs 0.000 description 1
- 229960002757 midecamycin Drugs 0.000 description 1
- 229950007764 mikamycin Drugs 0.000 description 1
- 229960003775 miltefosine Drugs 0.000 description 1
- PQLXHQMOHUQAKB-UHFFFAOYSA-N miltefosine Chemical compound CCCCCCCCCCCCCCCCOP([O-])(=O)OCC[N+](C)(C)C PQLXHQMOHUQAKB-UHFFFAOYSA-N 0.000 description 1
- 229960004023 minocycline Drugs 0.000 description 1
- 229960000350 mitotane Drugs 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 229950008814 momelotinib Drugs 0.000 description 1
- ZVHNDZWQTBEVRY-UHFFFAOYSA-N momelotinib Chemical compound C1=CC(C(NCC#N)=O)=CC=C1C1=CC=NC(NC=2C=CC(=CC=2)N2CCOCC2)=N1 ZVHNDZWQTBEVRY-UHFFFAOYSA-N 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 229940125645 monoclonal antibody drug Drugs 0.000 description 1
- 229960003702 moxifloxacin Drugs 0.000 description 1
- FABPRXSRWADJSP-MEDUHNTESA-N moxifloxacin Chemical compound COC1=C(N2C[C@H]3NCCC[C@H]3C2)C(F)=CC(C(C(C(O)=O)=C2)=O)=C1N2C1CC1 FABPRXSRWADJSP-MEDUHNTESA-N 0.000 description 1
- 229950002212 mubritinib Drugs 0.000 description 1
- ZTFBIUXIQYRUNT-MDWZMJQESA-N mubritinib Chemical compound C1=CC(C(F)(F)F)=CC=C1\C=C\C1=NC(COC=2C=CC(CCCCN3N=NC=C3)=CC=2)=CO1 ZTFBIUXIQYRUNT-MDWZMJQESA-N 0.000 description 1
- 229960003128 mupirocin Drugs 0.000 description 1
- 229930187697 mupirocin Natural products 0.000 description 1
- DDHVILIIHBIMQU-YJGQQKNPSA-L mupirocin calcium hydrate Chemical compound O.O.[Ca+2].C[C@H](O)[C@H](C)[C@@H]1O[C@H]1C[C@@H]1[C@@H](O)[C@@H](O)[C@H](C\C(C)=C\C(=O)OCCCCCCCCC([O-])=O)OC1.C[C@H](O)[C@H](C)[C@@H]1O[C@H]1C[C@@H]1[C@@H](O)[C@@H](O)[C@H](C\C(C)=C\C(=O)OCCCCCCCCC([O-])=O)OC1 DDHVILIIHBIMQU-YJGQQKNPSA-L 0.000 description 1
- RTGDFNSFWBGLEC-SYZQJQIISA-N mycophenolate mofetil Chemical compound COC1=C(C)C=2COC(=O)C=2C(O)=C1C\C=C(/C)CCC(=O)OCCN1CCOCC1 RTGDFNSFWBGLEC-SYZQJQIISA-N 0.000 description 1
- 229960004866 mycophenolate mofetil Drugs 0.000 description 1
- KVKFRMCSXWQSNT-UHFFFAOYSA-N n,n'-dimethylethane-1,2-diamine Chemical compound CNCCNC KVKFRMCSXWQSNT-UHFFFAOYSA-N 0.000 description 1
- SHXOKQKTZJXHHR-UHFFFAOYSA-N n,n-diethyl-5-iminobenzo[a]phenoxazin-9-amine;hydrochloride Chemical compound [Cl-].C1=CC=C2C3=NC4=CC=C(N(CC)CC)C=C4OC3=CC(=[NH2+])C2=C1 SHXOKQKTZJXHHR-UHFFFAOYSA-N 0.000 description 1
- JORAUNFTUVJTNG-BSTBCYLQSA-N n-[(2s)-4-amino-1-[[(2s,3r)-1-[[(2s)-4-amino-1-oxo-1-[[(3s,6s,9s,12s,15r,18s,21s)-6,9,18-tris(2-aminoethyl)-3-[(1r)-1-hydroxyethyl]-12,15-bis(2-methylpropyl)-2,5,8,11,14,17,20-heptaoxo-1,4,7,10,13,16,19-heptazacyclotricos-21-yl]amino]butan-2-yl]amino]-3-h Chemical compound CC(C)CCCCC(=O)N[C@@H](CCN)C(=O)N[C@H]([C@@H](C)O)CN[C@@H](CCN)C(=O)N[C@H]1CCNC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCN)NC(=O)[C@H](CCN)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CCN)NC1=O.CCC(C)CCCCC(=O)N[C@@H](CCN)C(=O)N[C@H]([C@@H](C)O)CN[C@@H](CCN)C(=O)N[C@H]1CCNC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCN)NC(=O)[C@H](CCN)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CCN)NC1=O JORAUNFTUVJTNG-BSTBCYLQSA-N 0.000 description 1
- NJSMWLQOCQIOPE-OCHFTUDZSA-N n-[(e)-[10-[(e)-(4,5-dihydro-1h-imidazol-2-ylhydrazinylidene)methyl]anthracen-9-yl]methylideneamino]-4,5-dihydro-1h-imidazol-2-amine Chemical compound N1CCN=C1N\N=C\C(C1=CC=CC=C11)=C(C=CC=C2)C2=C1\C=N\NC1=NCCN1 NJSMWLQOCQIOPE-OCHFTUDZSA-N 0.000 description 1
- ZOGSYTPLAROSTP-UHFFFAOYSA-N n-[bis(ethylamino)phosphoryl]ethanamine Chemical compound CCNP(=O)(NCC)NCC ZOGSYTPLAROSTP-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- RWHUEXWOYVBUCI-ITQXDASVSA-N nafarelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C=C2C=CC=CC2=CC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 RWHUEXWOYVBUCI-ITQXDASVSA-N 0.000 description 1
- 229960002333 nafarelin Drugs 0.000 description 1
- GPXLMGHLHQJAGZ-JTDSTZFVSA-N nafcillin Chemical compound C1=CC=CC2=C(C(=O)N[C@@H]3C(N4[C@H](C(C)(C)S[C@@H]43)C(O)=O)=O)C(OCC)=CC=C21 GPXLMGHLHQJAGZ-JTDSTZFVSA-N 0.000 description 1
- 229960000515 nafcillin Drugs 0.000 description 1
- 150000002790 naphthalenes Chemical class 0.000 description 1
- 229940126662 negative allosteric modulator Drugs 0.000 description 1
- 229960000884 nelfinavir Drugs 0.000 description 1
- QAGYKUNXZHXKMR-HKWSIXNMSA-N nelfinavir Chemical compound CC1=C(O)C=CC=C1C(=O)N[C@H]([C@H](O)CN1[C@@H](C[C@@H]2CCCC[C@@H]2C1)C(=O)NC(C)(C)C)CSC1=CC=CC=C1 QAGYKUNXZHXKMR-HKWSIXNMSA-N 0.000 description 1
- 229960004927 neomycin Drugs 0.000 description 1
- 239000002581 neurotoxin Substances 0.000 description 1
- 231100000618 neurotoxin Toxicity 0.000 description 1
- 229960000689 nevirapine Drugs 0.000 description 1
- VOFUROIFQGPCGE-UHFFFAOYSA-N nile red Chemical compound C1=CC=C2C3=NC4=CC=C(N(CC)CC)C=C4OC3=CC(=O)C2=C1 VOFUROIFQGPCGE-UHFFFAOYSA-N 0.000 description 1
- 229960001346 nilotinib Drugs 0.000 description 1
- HHZIURLSWUIHRB-UHFFFAOYSA-N nilotinib Chemical compound C1=NC(C)=CN1C1=CC(NC(=O)C=2C=C(NC=3N=C(C=CN=3)C=3C=NC=CC=3)C(C)=CC=2)=CC(C(F)(F)F)=C1 HHZIURLSWUIHRB-UHFFFAOYSA-N 0.000 description 1
- 229960001420 nimustine Drugs 0.000 description 1
- VFEDRRNHLBGPNN-UHFFFAOYSA-N nimustine Chemical compound CC1=NC=C(CNC(=O)N(CCCl)N=O)C(N)=N1 VFEDRRNHLBGPNN-UHFFFAOYSA-N 0.000 description 1
- 229910017604 nitric acid Inorganic materials 0.000 description 1
- 229960000564 nitrofurantoin Drugs 0.000 description 1
- NXFQHRVNIOXGAQ-YCRREMRBSA-N nitrofurantoin Chemical compound O1C([N+](=O)[O-])=CC=C1\C=N\N1C(=O)NC(=O)C1 NXFQHRVNIOXGAQ-YCRREMRBSA-N 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- SJYNFBVQFBRSIB-UHFFFAOYSA-N norbornadiene Chemical compound C1=CC2C=CC1C2 SJYNFBVQFBRSIB-UHFFFAOYSA-N 0.000 description 1
- 229960001180 norfloxacin Drugs 0.000 description 1
- OGJPXUAPXNRGGI-UHFFFAOYSA-N norfloxacin Chemical compound C1=C2N(CC)C=C(C(O)=O)C(=O)C2=CC(F)=C1N1CCNCC1 OGJPXUAPXNRGGI-UHFFFAOYSA-N 0.000 description 1
- URPYMXQQVHTUDU-OFGSCBOVSA-N nucleopeptide y Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(N)=O)NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](C)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](N)CC=1C=CC(O)=CC=1)C1=CC=C(O)C=C1 URPYMXQQVHTUDU-OFGSCBOVSA-N 0.000 description 1
- 230000000269 nucleophilic effect Effects 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 229960002700 octreotide Drugs 0.000 description 1
- 229960001699 ofloxacin Drugs 0.000 description 1
- 229960002351 oleandomycin Drugs 0.000 description 1
- RZPAKFUAFGMUPI-KGIGTXTPSA-N oleandomycin Chemical compound O1[C@@H](C)[C@H](O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](C)C(=O)O[C@H](C)[C@H](C)[C@H](O)[C@@H](C)C(=O)[C@]2(OC2)C[C@H](C)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)O)[C@H]1C RZPAKFUAFGMUPI-KGIGTXTPSA-N 0.000 description 1
- 235000019367 oleandomycin Nutrition 0.000 description 1
- 239000010450 olivine Substances 0.000 description 1
- 229910052609 olivine Inorganic materials 0.000 description 1
- 229960004780 orbifloxacin Drugs 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- VHFGEBVPHAGQPI-MYYQHNLBSA-N oritavancin Chemical compound O([C@@H]1C2=CC=C(C(=C2)Cl)OC=2C=C3C=C(C=2O[C@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)O[C@@H]2O[C@@H](C)[C@H](O)[C@@](C)(NCC=4C=CC(=CC=4)C=4C=CC(Cl)=CC=4)C2)OC2=CC=C(C=C2Cl)[C@@H](O)[C@H](C(N[C@@H](CC(N)=O)C(=O)N[C@H]3C(=O)N[C@H]2C(=O)N[C@@H]1C(N[C@H](C1=CC(O)=CC(O)=C1C=1C(O)=CC=C2C=1)C(O)=O)=O)=O)NC(=O)[C@@H](CC(C)C)NC)[C@H]1C[C@](C)(N)[C@@H](O)[C@H](C)O1 VHFGEBVPHAGQPI-MYYQHNLBSA-N 0.000 description 1
- 229960001607 oritavancin Drugs 0.000 description 1
- 108010006945 oritavancin Proteins 0.000 description 1
- VSZGPKBBMSAYNT-RRFJBIMHSA-N oseltamivir Chemical compound CCOC(=O)C1=C[C@@H](OC(CC)CC)[C@H](NC(C)=O)[C@@H](N)C1 VSZGPKBBMSAYNT-RRFJBIMHSA-N 0.000 description 1
- 229960003752 oseltamivir Drugs 0.000 description 1
- UWYHMGVUTGAWSP-JKIFEVAISA-N oxacillin Chemical compound N([C@@H]1C(N2[C@H](C(C)(C)S[C@@H]21)C(O)=O)=O)C(=O)C1=C(C)ON=C1C1=CC=CC=C1 UWYHMGVUTGAWSP-JKIFEVAISA-N 0.000 description 1
- 229960001019 oxacillin Drugs 0.000 description 1
- 150000004866 oxadiazoles Chemical class 0.000 description 1
- 229960001756 oxaliplatin Drugs 0.000 description 1
- DWAFYCQODLXJNR-BNTLRKBRSA-L oxaliplatin Chemical compound O1C(=O)C(=O)O[Pt]11N[C@@H]2CCCC[C@H]2N1 DWAFYCQODLXJNR-BNTLRKBRSA-L 0.000 description 1
- GHTWDWCFRFTBRB-UHFFFAOYSA-M oxazine-170 Chemical compound [O-]Cl(=O)(=O)=O.N1=C2C3=CC=CC=C3C(NCC)=CC2=[O+]C2=C1C=C(C)C(N(C)CC)=C2 GHTWDWCFRFTBRB-UHFFFAOYSA-M 0.000 description 1
- 150000004893 oxazines Chemical class 0.000 description 1
- 239000007800 oxidant agent Substances 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229960000625 oxytetracycline Drugs 0.000 description 1
- IWVCMVBTMGNXQD-PXOLEDIWSA-N oxytetracycline Chemical compound C1=CC=C2[C@](O)(C)[C@H]3[C@H](O)[C@H]4[C@H](N(C)C)C(O)=C(C(N)=O)C(=O)[C@@]4(O)C(O)=C3C(=O)C2=C1O IWVCMVBTMGNXQD-PXOLEDIWSA-N 0.000 description 1
- 235000019366 oxytetracycline Nutrition 0.000 description 1
- VYNDHICBIRRPFP-UHFFFAOYSA-N pacific blue Chemical compound FC1=C(O)C(F)=C2OC(=O)C(C(=O)O)=CC2=C1 VYNDHICBIRRPFP-UHFFFAOYSA-N 0.000 description 1
- VREZDOWOLGNDPW-UHFFFAOYSA-N pancratistatine Natural products C1=C2C3C(O)C(O)C(O)C(O)C3NC(=O)C2=C(O)C2=C1OCO2 VREZDOWOLGNDPW-UHFFFAOYSA-N 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- 229950011346 panipenem Drugs 0.000 description 1
- 229960001972 panitumumab Drugs 0.000 description 1
- 229960005184 panobinostat Drugs 0.000 description 1
- FPOHNWQLNRZRFC-ZHACJKMWSA-N panobinostat Chemical compound CC=1NC2=CC=CC=C2C=1CCNCC1=CC=C(\C=C\C(=O)NO)C=C1 FPOHNWQLNRZRFC-ZHACJKMWSA-N 0.000 description 1
- 229960001914 paromomycin Drugs 0.000 description 1
- UOZODPSAJZTQNH-LSWIJEOBSA-N paromomycin Chemical compound N[C@@H]1[C@@H](O)[C@H](O)[C@H](CN)O[C@@H]1O[C@H]1[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](N)C[C@@H](N)[C@@H]2O)O[C@@H]2[C@@H]([C@@H](O)[C@H](O)[C@@H](CO)O2)N)O[C@@H]1CO UOZODPSAJZTQNH-LSWIJEOBSA-N 0.000 description 1
- 229960000639 pazopanib Drugs 0.000 description 1
- CUIHSIWYWATEQL-UHFFFAOYSA-N pazopanib Chemical compound C1=CC2=C(C)N(C)N=C2C=C1N(C)C(N=1)=CC=NC=1NC1=CC=C(C)C(S(N)(=O)=O)=C1 CUIHSIWYWATEQL-UHFFFAOYSA-N 0.000 description 1
- 229960004236 pefloxacin Drugs 0.000 description 1
- FHFYDNQZQSQIAI-UHFFFAOYSA-N pefloxacin Chemical compound C1=C2N(CC)C=C(C(O)=O)C(=O)C2=CC(F)=C1N1CCN(C)CC1 FHFYDNQZQSQIAI-UHFFFAOYSA-N 0.000 description 1
- 229960001179 penciclovir Drugs 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 150000002960 penicillins Chemical class 0.000 description 1
- JZRYQZJSTWVBBD-UHFFFAOYSA-N pentaporphyrin i Chemical compound N1C(C=C2NC(=CC3=NC(=C4)C=C3)C=C2)=CC=C1C=C1C=CC4=N1 JZRYQZJSTWVBBD-UHFFFAOYSA-N 0.000 description 1
- 229960002340 pentostatin Drugs 0.000 description 1
- FPVKHBSQESCIEP-JQCXWYLXSA-N pentostatin Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(N=CNC[C@H]2O)=C2N=C1 FPVKHBSQESCIEP-JQCXWYLXSA-N 0.000 description 1
- 125000001147 pentyl group Chemical class C(CCCC)* 0.000 description 1
- QIMGFXOHTOXMQP-GFAGFCTOSA-N peplomycin Chemical compound N([C@H](C(=O)N[C@H](C)[C@@H](O)[C@H](C)C(=O)N[C@@H]([C@H](O)C)C(=O)NCCC=1SC=C(N=1)C=1SC=C(N=1)C(=O)NCCCN[C@@H](C)C=1C=CC=CC=1)[C@@H](O[C@H]1[C@H]([C@@H](O)[C@H](O)[C@H](CO)O1)O[C@@H]1[C@H]([C@@H](OC(N)=O)[C@H](O)[C@@H](CO)O1)O)C=1NC=NC=1)C(=O)C1=NC([C@H](CC(N)=O)NC[C@H](N)C(N)=O)=NC(N)=C1C QIMGFXOHTOXMQP-GFAGFCTOSA-N 0.000 description 1
- 229950003180 peplomycin Drugs 0.000 description 1
- 239000000813 peptide hormone Substances 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- XRQDFNLINLXZLB-CKIKVBCHSA-N peramivir Chemical compound CCC(CC)[C@H](NC(C)=O)[C@@H]1[C@H](O)[C@@H](C(O)=O)C[C@H]1NC(N)=N XRQDFNLINLXZLB-CKIKVBCHSA-N 0.000 description 1
- 229960001084 peramivir Drugs 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 1
- 239000003757 phosphotransferase inhibitor Substances 0.000 description 1
- 230000001699 photocatalysis Effects 0.000 description 1
- 238000002428 photodynamic therapy Methods 0.000 description 1
- INAAIJLSXJJHOZ-UHFFFAOYSA-N pibenzimol Chemical compound C1CN(C)CCN1C1=CC=C(N=C(N2)C=3C=C4NC(=NC4=CC=3)C=3C=CC(O)=CC=3)C2=C1 INAAIJLSXJJHOZ-UHFFFAOYSA-N 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000006187 pill Substances 0.000 description 1
- 239000001739 pinus spp. Substances 0.000 description 1
- 229960001221 pirarubicin Drugs 0.000 description 1
- 230000001817 pituitary effect Effects 0.000 description 1
- 229910052697 platinum Inorganic materials 0.000 description 1
- 229960000471 pleconaril Drugs 0.000 description 1
- KQOXLKOJHVFTRN-UHFFFAOYSA-N pleconaril Chemical compound O1N=C(C)C=C1CCCOC1=C(C)C=C(C=2N=C(ON=2)C(F)(F)F)C=C1C KQOXLKOJHVFTRN-UHFFFAOYSA-N 0.000 description 1
- 229920000573 polyethylene Polymers 0.000 description 1
- 229930001119 polyketide Natural products 0.000 description 1
- 238000006116 polymerization reaction Methods 0.000 description 1
- 229920000024 polymyxin B Polymers 0.000 description 1
- XDJYMJULXQKGMM-UHFFFAOYSA-N polymyxin E1 Natural products CCC(C)CCCCC(=O)NC(CCN)C(=O)NC(C(C)O)C(=O)NC(CCN)C(=O)NC1CCNC(=O)C(C(C)O)NC(=O)C(CCN)NC(=O)C(CCN)NC(=O)C(CC(C)C)NC(=O)C(CC(C)C)NC(=O)C(CCN)NC1=O XDJYMJULXQKGMM-UHFFFAOYSA-N 0.000 description 1
- KNIWPHSUTGNZST-UHFFFAOYSA-N polymyxin E2 Natural products CC(C)CCCCC(=O)NC(CCN)C(=O)NC(C(C)O)C(=O)NC(CCN)C(=O)NC1CCNC(=O)C(C(C)O)NC(=O)C(CCN)NC(=O)C(CCN)NC(=O)C(CC(C)C)NC(=O)C(CC(C)C)NC(=O)C(CCN)NC1=O KNIWPHSUTGNZST-UHFFFAOYSA-N 0.000 description 1
- 229960005266 polymyxin b Drugs 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 229960000688 pomalidomide Drugs 0.000 description 1
- UVSMNLNDYGZFPF-UHFFFAOYSA-N pomalidomide Chemical compound O=C1C=2C(N)=CC=CC=2C(=O)N1C1CCC(=O)NC1=O UVSMNLNDYGZFPF-UHFFFAOYSA-N 0.000 description 1
- PHXJVRSECIGDHY-UHFFFAOYSA-N ponatinib Chemical compound C1CN(C)CCN1CC(C(=C1)C(F)(F)F)=CC=C1NC(=O)C1=CC=C(C)C(C#CC=2N3N=CC=CC3=NC=2)=C1 PHXJVRSECIGDHY-UHFFFAOYSA-N 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- JHDKZFFAIZKUCU-ZRDIBKRKSA-N pracinostat Chemical compound ONC(=O)/C=C/C1=CC=C2N(CCN(CC)CC)C(CCCC)=NC2=C1 JHDKZFFAIZKUCU-ZRDIBKRKSA-N 0.000 description 1
- 229960004618 prednisone Drugs 0.000 description 1
- XOFYZVNMUHMLCC-ZPOLXVRWSA-N prednisone Chemical compound O=C1C=C[C@]2(C)[C@H]3C(=O)C[C@](C)([C@@](CC4)(O)C(=O)CO)[C@@H]4[C@@H]3CCC2=C1 XOFYZVNMUHMLCC-ZPOLXVRWSA-N 0.000 description 1
- 230000013823 prenylation Effects 0.000 description 1
- CPTBDICYNRMXFX-UHFFFAOYSA-N procarbazine Chemical compound CNNCC1=CC=C(C(=O)NC(C)C)C=C1 CPTBDICYNRMXFX-UHFFFAOYSA-N 0.000 description 1
- 229960000624 procarbazine Drugs 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- XJMOSONTPMZWPB-UHFFFAOYSA-M propidium iodide Chemical compound [I-].[I-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CCC[N+](C)(CC)CC)=C1C1=CC=CC=C1 XJMOSONTPMZWPB-UHFFFAOYSA-M 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 229940034080 provenge Drugs 0.000 description 1
- 150000003212 purines Chemical class 0.000 description 1
- 229950010131 puromycin Drugs 0.000 description 1
- 229960005206 pyrazinamide Drugs 0.000 description 1
- IPEHBUMCGVEMRF-UHFFFAOYSA-N pyrazinecarboxamide Chemical compound NC(=O)C1=CN=CC=N1 IPEHBUMCGVEMRF-UHFFFAOYSA-N 0.000 description 1
- 150000003220 pyrenes Chemical class 0.000 description 1
- 150000003230 pyrimidines Chemical class 0.000 description 1
- MIXMJCQRHVAJIO-TZHJZOAOSA-N qk4dys664x Chemical compound O.C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O.C1([C@@H](F)C2)=CC(=O)C=C[C@]1(C)[C@@H]1[C@@H]2[C@@H]2C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]2(C)C[C@@H]1O MIXMJCQRHVAJIO-TZHJZOAOSA-N 0.000 description 1
- 238000003908 quality control method Methods 0.000 description 1
- 150000007660 quinolones Chemical class 0.000 description 1
- 150000004053 quinones Chemical class 0.000 description 1
- UOWVMDUEMSNCAV-WYENRQIDSA-N rachelmycin Chemical compound C1([C@]23C[C@@H]2CN1C(=O)C=1NC=2C(OC)=C(O)C4=C(C=2C=1)CCN4C(=O)C1=CC=2C=4CCN(C=4C(O)=C(C=2N1)OC)C(N)=O)=CC(=O)C1=C3C(C)=CN1 UOWVMDUEMSNCAV-WYENRQIDSA-N 0.000 description 1
- 229960004622 raloxifene Drugs 0.000 description 1
- GZUITABIAKMVPG-UHFFFAOYSA-N raloxifene Chemical compound C1=CC(O)=CC=C1C1=C(C(=O)C=2C=CC(OCCN3CCCCC3)=CC=2)C2=CC=C(O)C=C2S1 GZUITABIAKMVPG-UHFFFAOYSA-N 0.000 description 1
- 229960004742 raltegravir Drugs 0.000 description 1
- CZFFBEXEKNGXKS-UHFFFAOYSA-N raltegravir Chemical compound O1C(C)=NN=C1C(=O)NC(C)(C)C1=NC(C(=O)NCC=2C=CC(F)=CC=2)=C(O)C(=O)N1C CZFFBEXEKNGXKS-UHFFFAOYSA-N 0.000 description 1
- 229960004432 raltitrexed Drugs 0.000 description 1
- 229960003876 ranibizumab Drugs 0.000 description 1
- ZAHRKKWIAAJSAO-UHFFFAOYSA-N rapamycin Natural products COCC(O)C(=C/C(C)C(=O)CC(OC(=O)C1CCCCN1C(=O)C(=O)C2(O)OC(CC(OC)C(=CC=CC=CC(C)CC(C)C(=O)C)C)CCC2C)C(C)CC3CCC(O)C(C3)OC)C ZAHRKKWIAAJSAO-UHFFFAOYSA-N 0.000 description 1
- 230000035484 reaction time Effects 0.000 description 1
- 239000002464 receptor antagonist Substances 0.000 description 1
- 229940044551 receptor antagonist Drugs 0.000 description 1
- BXNMTOQRYBFHNZ-UHFFFAOYSA-N resiquimod Chemical compound C1=CC=CC2=C(N(C(COCC)=N3)CC(C)(C)O)C3=C(N)N=C21 BXNMTOQRYBFHNZ-UHFFFAOYSA-N 0.000 description 1
- FECGNJPYVFEKOD-VMPITWQZSA-N resminostat Chemical compound C1=CC(CN(C)C)=CC=C1S(=O)(=O)N1C=C(\C=C\C(=O)NO)C=C1 FECGNJPYVFEKOD-VMPITWQZSA-N 0.000 description 1
- 229950002821 resminostat Drugs 0.000 description 1
- 229930002330 retinoic acid Natural products 0.000 description 1
- 229960003471 retinol Drugs 0.000 description 1
- 235000020944 retinol Nutrition 0.000 description 1
- 239000011607 retinol Substances 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- BTVYFIMKUHNOBZ-QXMMDKDBSA-N rifamycin s Chemical class O=C1C(C(O)=C2C)=C3C(=O)C=C1NC(=O)\C(C)=C/C=C\C(C)C(O)C(C)C(O)C(C)C(OC(C)=O)C(C)C(OC)\C=C/OC1(C)OC2=C3C1=O BTVYFIMKUHNOBZ-QXMMDKDBSA-N 0.000 description 1
- 229940081192 rifamycins Drugs 0.000 description 1
- 229960002599 rifapentine Drugs 0.000 description 1
- 229960002814 rilpivirine Drugs 0.000 description 1
- YIBOMRUWOWDFLG-ONEGZZNKSA-N rilpivirine Chemical compound CC1=CC(\C=C\C#N)=CC(C)=C1NC1=CC=NC(NC=2C=CC(=CC=2)C#N)=N1 YIBOMRUWOWDFLG-ONEGZZNKSA-N 0.000 description 1
- 229960000888 rimantadine Drugs 0.000 description 1
- 229960000311 ritonavir Drugs 0.000 description 1
- NCDNCNXCDXHOMX-XGKFQTDJSA-N ritonavir Chemical compound N([C@@H](C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](CC=1C=CC=CC=1)NC(=O)OCC=1SC=NC=1)CC=1C=CC=CC=1)C(=O)N(C)CC1=CSC(C(C)C)=N1 NCDNCNXCDXHOMX-XGKFQTDJSA-N 0.000 description 1
- MBABCNBNDNGODA-WPZDJQSSSA-N rolliniastatin 1 Natural products O1[C@@H]([C@@H](O)CCCCCCCCCC)CC[C@H]1[C@H]1O[C@@H]([C@H](O)CCCCCCCCCC[C@@H](O)CC=2C(O[C@@H](C)C=2)=O)CC1 MBABCNBNDNGODA-WPZDJQSSSA-N 0.000 description 1
- 229960003452 romidepsin Drugs 0.000 description 1
- OHRURASPPZQGQM-GCCNXGTGSA-N romidepsin Chemical compound O1C(=O)[C@H](C(C)C)NC(=O)C(=C/C)/NC(=O)[C@H]2CSSCC\C=C\[C@@H]1CC(=O)N[C@H](C(C)C)C(=O)N2 OHRURASPPZQGQM-GCCNXGTGSA-N 0.000 description 1
- 108010091666 romidepsin Proteins 0.000 description 1
- OHRURASPPZQGQM-UHFFFAOYSA-N romidepsin Natural products O1C(=O)C(C(C)C)NC(=O)C(=CC)NC(=O)C2CSSCCC=CC1CC(=O)NC(C(C)C)C(=O)N2 OHRURASPPZQGQM-UHFFFAOYSA-N 0.000 description 1
- 229960005224 roxithromycin Drugs 0.000 description 1
- 102200089551 rs5030826 Human genes 0.000 description 1
- 229960000215 ruxolitinib Drugs 0.000 description 1
- HFNKQEVNSGCOJV-OAHLLOKOSA-N ruxolitinib Chemical compound C1([C@@H](CC#N)N2N=CC(=C2)C=2C=3C=CNC=3N=CN=2)CCCC1 HFNKQEVNSGCOJV-OAHLLOKOSA-N 0.000 description 1
- NGWSFRIPKNWYAO-SHTIJGAHSA-N salinosporamide A Chemical compound C([C@@H]1[C@H](O)[C@]23C(=O)O[C@]2([C@H](C(=O)N3)CCCl)C)CCC=C1 NGWSFRIPKNWYAO-SHTIJGAHSA-N 0.000 description 1
- NGWSFRIPKNWYAO-UHFFFAOYSA-N salinosporamide A Natural products N1C(=O)C(CCCl)C2(C)OC(=O)C21C(O)C1CCCC=C1 NGWSFRIPKNWYAO-UHFFFAOYSA-N 0.000 description 1
- 239000012488 sample solution Substances 0.000 description 1
- 229940072272 sandostatin Drugs 0.000 description 1
- 229910052594 sapphire Inorganic materials 0.000 description 1
- 239000010980 sapphire Substances 0.000 description 1
- 229960001852 saquinavir Drugs 0.000 description 1
- QWAXKHKRTORLEM-UGJKXSETSA-N saquinavir Chemical compound C([C@@H]([C@H](O)CN1C[C@H]2CCCC[C@H]2C[C@H]1C(=O)NC(C)(C)C)NC(=O)[C@H](CC(N)=O)NC(=O)C=1N=C2C=CC=CC2=CC=1)C1=CC=CC=C1 QWAXKHKRTORLEM-UGJKXSETSA-N 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- BTIHMVBBUGXLCJ-OAHLLOKOSA-N seliciclib Chemical compound C=12N=CN(C(C)C)C2=NC(N[C@@H](CO)CC)=NC=1NCC1=CC=CC=C1 BTIHMVBBUGXLCJ-OAHLLOKOSA-N 0.000 description 1
- 229950000055 seliciclib Drugs 0.000 description 1
- DYPYMMHZGRPOCK-UHFFFAOYSA-N seminaphtharhodafluor Chemical compound O1C(=O)C2=CC=CC=C2C21C(C=CC=1C3=CC=C(O)C=1)=C3OC1=CC(N)=CC=C21 DYPYMMHZGRPOCK-UHFFFAOYSA-N 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 239000002911 sialidase inhibitor Substances 0.000 description 1
- 229910052710 silicon Inorganic materials 0.000 description 1
- 229960002930 sirolimus Drugs 0.000 description 1
- QFJCIRLUMZQUOT-HPLJOQBZSA-N sirolimus Chemical compound C1C[C@@H](O)[C@H](OC)C[C@@H]1C[C@@H](C)[C@H]1OC(=O)[C@@H]2CCCCN2C(=O)C(=O)[C@](O)(O2)[C@H](C)CC[C@H]2C[C@H](OC)/C(C)=C/C=C/C=C/[C@@H](C)C[C@@H](C)C(=O)[C@H](OC)[C@H](O)/C(C)=C/[C@@H](C)C(=O)C1 QFJCIRLUMZQUOT-HPLJOQBZSA-N 0.000 description 1
- 229960005456 sisomicin Drugs 0.000 description 1
- URWAJWIAIPFPJE-YFMIWBNJSA-N sisomycin Chemical compound O1C[C@@](O)(C)[C@H](NC)[C@@H](O)[C@H]1O[C@@H]1[C@@H](O)[C@H](O[C@@H]2[C@@H](CC=C(CN)O2)N)[C@@H](N)C[C@H]1N URWAJWIAIPFPJE-YFMIWBNJSA-N 0.000 description 1
- 229960003177 sitafloxacin Drugs 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- SNOOUWRIMMFWNE-UHFFFAOYSA-M sodium;6-[(3,4,5-trimethoxybenzoyl)amino]hexanoate Chemical compound [Na+].COC1=CC(C(=O)NCCCCCC([O-])=O)=CC(OC)=C1OC SNOOUWRIMMFWNE-UHFFFAOYSA-M 0.000 description 1
- NHXLMOGPVYXJNR-ATOGVRKGSA-N somatostatin Chemical compound C([C@H]1C(=O)N[C@H](C(N[C@@H](CO)C(=O)N[C@@H](CSSC[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(=O)N1)[C@@H](C)O)NC(=O)CNC(=O)[C@H](C)N)C(O)=O)=O)[C@H](O)C)C1=CC=CC=C1 NHXLMOGPVYXJNR-ATOGVRKGSA-N 0.000 description 1
- 229960000553 somatostatin Drugs 0.000 description 1
- 229940078986 somatuline Drugs 0.000 description 1
- 108010014657 sortilin Proteins 0.000 description 1
- 229960004954 sparfloxacin Drugs 0.000 description 1
- DZZWHBIBMUVIIW-DTORHVGOSA-N sparfloxacin Chemical compound C1[C@@H](C)N[C@@H](C)CN1C1=C(F)C(N)=C2C(=O)C(C(O)=O)=CN(C3CC3)C2=C1F DZZWHBIBMUVIIW-DTORHVGOSA-N 0.000 description 1
- 229960001294 spiramycin Drugs 0.000 description 1
- 235000019372 spiramycin Nutrition 0.000 description 1
- 229930191512 spiramycin Natural products 0.000 description 1
- 229950006315 spirogermanium Drugs 0.000 description 1
- HKSZLNNOFSGOKW-FYTWVXJKSA-N staurosporine Chemical compound C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1[C@H]1C[C@@H](NC)[C@@H](OC)[C@]4(C)O1 HKSZLNNOFSGOKW-FYTWVXJKSA-N 0.000 description 1
- CGPUWJWCVCFERF-UHFFFAOYSA-N staurosporine Natural products C12=C3N4C5=CC=CC=C5C3=C3CNC(=O)C3=C2C2=CC=CC=C2N1C1CC(NC)C(OC)C4(OC)O1 CGPUWJWCVCFERF-UHFFFAOYSA-N 0.000 description 1
- 239000000021 stimulant Substances 0.000 description 1
- 230000000638 stimulation Effects 0.000 description 1
- 229960001052 streptozocin Drugs 0.000 description 1
- ZSJLQEPLLKMAKR-GKHCUFPYSA-N streptozocin Chemical compound O=NN(C)C(=O)N[C@H]1[C@@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O ZSJLQEPLLKMAKR-GKHCUFPYSA-N 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 125000005346 substituted cycloalkyl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 235000000346 sugar Nutrition 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- FKENQMMABCRJMK-RITPCOANSA-N sulbactam Chemical compound O=S1(=O)C(C)(C)[C@H](C(O)=O)N2C(=O)C[C@H]21 FKENQMMABCRJMK-RITPCOANSA-N 0.000 description 1
- 229960005256 sulbactam Drugs 0.000 description 1
- SEEPANYCNGTZFQ-UHFFFAOYSA-N sulfadiazine Chemical compound C1=CC(N)=CC=C1S(=O)(=O)NC1=NC=CC=N1 SEEPANYCNGTZFQ-UHFFFAOYSA-N 0.000 description 1
- 229960004306 sulfadiazine Drugs 0.000 description 1
- 229960000654 sulfafurazole Drugs 0.000 description 1
- GECHUMIMRBOMGK-UHFFFAOYSA-N sulfapyridine Chemical compound C1=CC(N)=CC=C1S(=O)(=O)NC1=CC=CC=N1 GECHUMIMRBOMGK-UHFFFAOYSA-N 0.000 description 1
- 229960002211 sulfapyridine Drugs 0.000 description 1
- 150000003456 sulfonamides Chemical class 0.000 description 1
- 229960005559 sulforaphane Drugs 0.000 description 1
- 235000015487 sulforaphane Nutrition 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 229960001796 sunitinib Drugs 0.000 description 1
- WINHZLLDWRZWRT-ATVHPVEESA-N sunitinib Chemical compound CCN(CC)CCNC(=O)C1=C(C)NC(\C=C/2C3=CC(F)=CC=C3NC\2=O)=C1C WINHZLLDWRZWRT-ATVHPVEESA-N 0.000 description 1
- 239000013589 supplement Substances 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 239000003826 tablet Substances 0.000 description 1
- 229960001967 tacrolimus Drugs 0.000 description 1
- QJJXYPPXXYFBGM-SHYZHZOCSA-N tacrolimus Natural products CO[C@H]1C[C@H](CC[C@@H]1O)C=C(C)[C@H]2OC(=O)[C@H]3CCCCN3C(=O)C(=O)[C@@]4(O)O[C@@H]([C@H](C[C@H]4C)OC)[C@@H](C[C@H](C)CC(=C[C@@H](CC=C)C(=O)C[C@H](O)[C@H]2C)C)OC QJJXYPPXXYFBGM-SHYZHZOCSA-N 0.000 description 1
- 229960001603 tamoxifen Drugs 0.000 description 1
- 238000002626 targeted therapy Methods 0.000 description 1
- 229960003865 tazobactam Drugs 0.000 description 1
- LPQZKKCYTLCDGQ-WEDXCCLWSA-N tazobactam Chemical compound C([C@]1(C)S([C@H]2N(C(C2)=O)[C@H]1C(O)=O)(=O)=O)N1C=CN=N1 LPQZKKCYTLCDGQ-WEDXCCLWSA-N 0.000 description 1
- 229960001608 teicoplanin Drugs 0.000 description 1
- BBAWEDCPNXPBQM-GDEBMMAJSA-N telaprevir Chemical compound N([C@H](C(=O)N[C@H](C(=O)N1C[C@@H]2CCC[C@@H]2[C@H]1C(=O)N[C@@H](CCC)C(=O)C(=O)NC1CC1)C(C)(C)C)C1CCCCC1)C(=O)C1=CN=CC=N1 BBAWEDCPNXPBQM-GDEBMMAJSA-N 0.000 description 1
- ONUMZHGUFYIKPM-MXNFEBESSA-N telavancin Chemical compound O1[C@@H](C)[C@@H](O)[C@](NCCNCCCCCCCCCC)(C)C[C@@H]1O[C@H]1[C@H](OC=2C3=CC=4[C@H](C(N[C@H]5C(=O)N[C@H](C(N[C@@H](C6=CC(O)=C(CNCP(O)(O)=O)C(O)=C6C=6C(O)=CC=C5C=6)C(O)=O)=O)[C@H](O)C5=CC=C(C(=C5)Cl)O3)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](NC(=O)[C@@H](CC(C)C)NC)[C@H](O)C3=CC=C(C(=C3)Cl)OC=2C=4)O[C@H](CO)[C@@H](O)[C@@H]1O ONUMZHGUFYIKPM-MXNFEBESSA-N 0.000 description 1
- 229960005240 telavancin Drugs 0.000 description 1
- 108010089019 telavancin Proteins 0.000 description 1
- 229960005311 telbivudine Drugs 0.000 description 1
- IQFYYKKMVGJFEH-CSMHCCOUSA-N telbivudine Chemical compound O=C1NC(=O)C(C)=CN1[C@H]1O[C@@H](CO)[C@H](O)C1 IQFYYKKMVGJFEH-CSMHCCOUSA-N 0.000 description 1
- 229960004576 temafloxacin Drugs 0.000 description 1
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 1
- 229960001278 teniposide Drugs 0.000 description 1
- 229960004556 tenofovir Drugs 0.000 description 1
- VCMJCVGFSROFHV-WZGZYPNHSA-N tenofovir disoproxil fumarate Chemical compound OC(=O)\C=C\C(O)=O.N1=CN=C2N(C[C@@H](C)OCP(=O)(OCOC(=O)OC(C)C)OCOC(=O)OC(C)C)C=NC2=C1N VCMJCVGFSROFHV-WZGZYPNHSA-N 0.000 description 1
- 150000003505 terpenes Chemical class 0.000 description 1
- 235000007586 terpenes Nutrition 0.000 description 1
- IWVCMVBTMGNXQD-UHFFFAOYSA-N terramycin dehydrate Natural products C1=CC=C2C(O)(C)C3C(O)C4C(N(C)C)C(O)=C(C(N)=O)C(=O)C4(O)C(O)=C3C(=O)C2=C1O IWVCMVBTMGNXQD-UHFFFAOYSA-N 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- BCNZYOJHNLTNEZ-UHFFFAOYSA-N tert-butyldimethylsilyl chloride Chemical compound CC(C)(C)[Si](C)(C)Cl BCNZYOJHNLTNEZ-UHFFFAOYSA-N 0.000 description 1
- 229960003604 testosterone Drugs 0.000 description 1
- 229960002180 tetracycline Drugs 0.000 description 1
- 229930101283 tetracycline Natural products 0.000 description 1
- 229940040944 tetracyclines Drugs 0.000 description 1
- 125000000383 tetramethylene group Chemical group [H]C([H])([*:1])C([H])([H])C([H])([H])C([H])([H])[*:2] 0.000 description 1
- JGVWCANSWKRBCS-UHFFFAOYSA-N tetramethylrhodamine thiocyanate Chemical compound [Cl-].C=12C=CC(N(C)C)=CC2=[O+]C2=CC(N(C)C)=CC=C2C=1C1=CC=C(SC#N)C=C1C(O)=O JGVWCANSWKRBCS-UHFFFAOYSA-N 0.000 description 1
- 229960003433 thalidomide Drugs 0.000 description 1
- IXFPJGBNCFXKPI-FSIHEZPISA-N thapsigargin Chemical compound CCCC(=O)O[C@H]1C[C@](C)(OC(C)=O)[C@H]2[C@H](OC(=O)CCCCCCC)[C@@H](OC(=O)C(\C)=C/C)C(C)=C2[C@@H]2OC(=O)[C@@](C)(O)[C@]21O IXFPJGBNCFXKPI-FSIHEZPISA-N 0.000 description 1
- 231100001274 therapeutic index Toxicity 0.000 description 1
- 229960003495 thiamine Drugs 0.000 description 1
- KYMBYSLLVAOCFI-UHFFFAOYSA-N thiamine Chemical compound CC1=C(CCO)SCN1CC1=CN=C(C)N=C1N KYMBYSLLVAOCFI-UHFFFAOYSA-N 0.000 description 1
- 235000019157 thiamine Nutrition 0.000 description 1
- 239000011721 thiamine Substances 0.000 description 1
- 229960003053 thiamphenicol Drugs 0.000 description 1
- OTVAEFIXJLOWRX-NXEZZACHSA-N thiamphenicol Chemical compound CS(=O)(=O)C1=CC=C([C@@H](O)[C@@H](CO)NC(=O)C(Cl)Cl)C=C1 OTVAEFIXJLOWRX-NXEZZACHSA-N 0.000 description 1
- ACOJCCLIDPZYJC-UHFFFAOYSA-M thiazole orange Chemical compound CC1=CC=C(S([O-])(=O)=O)C=C1.C1=CC=C2C(C=C3N(C4=CC=CC=C4S3)C)=CC=[N+](C)C2=C1 ACOJCCLIDPZYJC-UHFFFAOYSA-M 0.000 description 1
- 229940104230 thymidine Drugs 0.000 description 1
- 229960004659 ticarcillin Drugs 0.000 description 1
- OHKOGUYZJXTSFX-KZFFXBSXSA-N ticarcillin Chemical compound C=1([C@@H](C(O)=O)C(=O)N[C@H]2[C@H]3SC([C@@H](N3C2=O)C(O)=O)(C)C)C=CSC=1 OHKOGUYZJXTSFX-KZFFXBSXSA-N 0.000 description 1
- 229960003087 tioguanine Drugs 0.000 description 1
- MNRILEROXIRVNJ-UHFFFAOYSA-N tioguanine Chemical compound N1C(N)=NC(=S)C2=NC=N[C]21 MNRILEROXIRVNJ-UHFFFAOYSA-N 0.000 description 1
- 229960000838 tipranavir Drugs 0.000 description 1
- SUJUHGSWHZTSEU-FYBSXPHGSA-N tipranavir Chemical compound C([C@@]1(CCC)OC(=O)C([C@H](CC)C=2C=C(NS(=O)(=O)C=3N=CC(=CC=3)C(F)(F)F)C=CC=2)=C(O)C1)CC1=CC=CC=C1 SUJUHGSWHZTSEU-FYBSXPHGSA-N 0.000 description 1
- 229960000940 tivozanib Drugs 0.000 description 1
- 239000011031 topaz Substances 0.000 description 1
- 229910052853 topaz Inorganic materials 0.000 description 1
- 229940044693 topoisomerase inhibitor Drugs 0.000 description 1
- 229950005801 tosedostat Drugs 0.000 description 1
- 229960000575 trastuzumab Drugs 0.000 description 1
- 229960001727 tretinoin Drugs 0.000 description 1
- 229960002117 triamcinolone acetonide Drugs 0.000 description 1
- YNDXUCZADRHECN-JNQJZLCISA-N triamcinolone acetonide Chemical compound C1CC2=CC(=O)C=C[C@]2(C)[C@]2(F)[C@@H]1[C@@H]1C[C@H]3OC(C)(C)O[C@@]3(C(=O)CO)[C@@]1(C)C[C@@H]2O YNDXUCZADRHECN-JNQJZLCISA-N 0.000 description 1
- 150000004654 triazenes Chemical class 0.000 description 1
- RTKIYFITIVXBLE-QEQCGCAPSA-N trichostatin A Chemical compound ONC(=O)/C=C/C(/C)=C/[C@@H](C)C(=O)C1=CC=C(N(C)C)C=C1 RTKIYFITIVXBLE-QEQCGCAPSA-N 0.000 description 1
- GWBUNZLLLLDXMD-UHFFFAOYSA-H tricopper;dicarbonate;dihydroxide Chemical compound [OH-].[OH-].[Cu+2].[Cu+2].[Cu+2].[O-]C([O-])=O.[O-]C([O-])=O GWBUNZLLLLDXMD-UHFFFAOYSA-H 0.000 description 1
- VSQQQLOSPVPRAZ-RRKCRQDMSA-N trifluridine Chemical compound C1[C@H](O)[C@@H](CO)O[C@H]1N1C(=O)NC(=O)C(C(F)(F)F)=C1 VSQQQLOSPVPRAZ-RRKCRQDMSA-N 0.000 description 1
- 229960003962 trifluridine Drugs 0.000 description 1
- IEDVJHCEMCRBQM-UHFFFAOYSA-N trimethoprim Chemical compound COC1=C(OC)C(OC)=CC(CC=2C(=NC(N)=NC=2)N)=C1 IEDVJHCEMCRBQM-UHFFFAOYSA-N 0.000 description 1
- 229960001082 trimethoprim Drugs 0.000 description 1
- VXKHXGOKWPXYNA-PGBVPBMZSA-N triptorelin Chemical compound C([C@@H](C(=O)N[C@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)NCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C2=CC=CC=C2NC=1)NC(=O)[C@H](CC=1N=CNC=1)NC(=O)[C@H]1NC(=O)CC1)C1=CC=C(O)C=C1 VXKHXGOKWPXYNA-PGBVPBMZSA-N 0.000 description 1
- 229960004824 triptorelin Drugs 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 229960005041 troleandomycin Drugs 0.000 description 1
- LQCLVBQBTUVCEQ-QTFUVMRISA-N troleandomycin Chemical compound O1[C@@H](C)[C@H](OC(C)=O)[C@@H](OC)C[C@@H]1O[C@@H]1[C@@H](C)C(=O)O[C@H](C)[C@H](C)[C@H](OC(C)=O)[C@@H](C)C(=O)[C@@]2(OC2)C[C@H](C)[C@H](O[C@H]2[C@@H]([C@H](C[C@@H](C)O2)N(C)C)OC(C)=O)[C@H]1C LQCLVBQBTUVCEQ-QTFUVMRISA-N 0.000 description 1
- 229960000497 trovafloxacin Drugs 0.000 description 1
- WVPSKSLAZQPAKQ-CDMJZVDBSA-N trovafloxacin Chemical compound C([C@H]1[C@@H]([C@H]1C1)N)N1C(C(=CC=1C(=O)C(C(O)=O)=C2)F)=NC=1N2C1=CC=C(F)C=C1F WVPSKSLAZQPAKQ-CDMJZVDBSA-N 0.000 description 1
- 229960001005 tuberculin Drugs 0.000 description 1
- 229940036248 turpentine Drugs 0.000 description 1
- 229950009811 ubenimex Drugs 0.000 description 1
- 238000002604 ultrasonography Methods 0.000 description 1
- 229960004626 umifenovir Drugs 0.000 description 1
- KCFYEAOKVJSACF-UHFFFAOYSA-N umifenovir Chemical compound CN1C2=CC(Br)=C(O)C(CN(C)C)=C2C(C(=O)OCC)=C1CSC1=CC=CC=C1 KCFYEAOKVJSACF-UHFFFAOYSA-N 0.000 description 1
- PSVXZQVXSXSQRO-UHFFFAOYSA-N undecaethylene glycol Chemical compound OCCOCCOCCOCCOCCOCCOCCOCCOCCOCCOCCO PSVXZQVXSXSQRO-UHFFFAOYSA-N 0.000 description 1
- 125000002948 undecyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 229940035893 uracil Drugs 0.000 description 1
- 229940093257 valacyclovir Drugs 0.000 description 1
- MSRILKIQRXUYCT-UHFFFAOYSA-M valproate semisodium Chemical compound [Na+].CCCC(C(O)=O)CCC.CCCC(C([O-])=O)CCC MSRILKIQRXUYCT-UHFFFAOYSA-M 0.000 description 1
- 229960000604 valproic acid Drugs 0.000 description 1
- 229960003165 vancomycin Drugs 0.000 description 1
- MYPYJXKWCTUITO-LYRMYLQWSA-N vancomycin Chemical compound O([C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=C2C=C3C=C1OC1=CC=C(C=C1Cl)[C@@H](O)[C@H](C(N[C@@H](CC(N)=O)C(=O)N[C@H]3C(=O)N[C@H]1C(=O)N[C@H](C(N[C@@H](C3=CC(O)=CC(O)=C3C=3C(O)=CC=C1C=3)C(O)=O)=O)[C@H](O)C1=CC=C(C(=C1)Cl)O2)=O)NC(=O)[C@@H](CC(C)C)NC)[C@H]1C[C@](C)(N)[C@H](O)[C@H](C)O1 MYPYJXKWCTUITO-LYRMYLQWSA-N 0.000 description 1
- MYPYJXKWCTUITO-UHFFFAOYSA-N vancomycin Natural products O1C(C(=C2)Cl)=CC=C2C(O)C(C(NC(C2=CC(O)=CC(O)=C2C=2C(O)=CC=C3C=2)C(O)=O)=O)NC(=O)C3NC(=O)C2NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(CC(C)C)NC)C(O)C(C=C3Cl)=CC=C3OC3=CC2=CC1=C3OC1OC(CO)C(O)C(O)C1OC1CC(C)(N)C(O)C(C)O1 MYPYJXKWCTUITO-UHFFFAOYSA-N 0.000 description 1
- 229960000241 vandetanib Drugs 0.000 description 1
- UHTHHESEBZOYNR-UHFFFAOYSA-N vandetanib Chemical compound COC1=CC(C(/N=CN2)=N/C=3C(=CC(Br)=CC=3)F)=C2C=C1OCC1CCN(C)CC1 UHTHHESEBZOYNR-UHFFFAOYSA-N 0.000 description 1
- GPXBXXGIAQBQNI-UHFFFAOYSA-N vemurafenib Chemical compound CCCS(=O)(=O)NC1=CC=C(F)C(C(=O)C=2C3=CC(=CN=C3NC=2)C=2C=CC(Cl)=CC=2)=C1F GPXBXXGIAQBQNI-UHFFFAOYSA-N 0.000 description 1
- 229960003862 vemurafenib Drugs 0.000 description 1
- 229960001722 verapamil Drugs 0.000 description 1
- 229960003895 verteporfin Drugs 0.000 description 1
- ZQFGRJWRSLZCSQ-ZSFNYQMMSA-N verteporfin Chemical compound C=1C([C@@]2([C@H](C(=O)OC)C(=CC=C22)C(=O)OC)C)=NC2=CC(C(=C2C=C)C)=NC2=CC(C(=C2CCC(O)=O)C)=NC2=CC2=NC=1C(C)=C2CCC(=O)OC ZQFGRJWRSLZCSQ-ZSFNYQMMSA-N 0.000 description 1
- 229950009860 vicriviroc Drugs 0.000 description 1
- GBABOYUKABKIAF-GHYRFKGUSA-N vinorelbine Chemical compound C1N(CC=2C3=CC=CC=C3NC=22)CC(CC)=C[C@H]1C[C@]2(C(=O)OC)C1=CC([C@]23[C@H]([C@]([C@H](OC(C)=O)[C@]4(CC)C=CCN([C@H]34)CC2)(O)C(=O)OC)N2C)=C2C=C1OC GBABOYUKABKIAF-GHYRFKGUSA-N 0.000 description 1
- 229960002066 vinorelbine Drugs 0.000 description 1
- 230000004482 visual phototransduction Effects 0.000 description 1
- 235000001892 vitamin D2 Nutrition 0.000 description 1
- 239000011653 vitamin D2 Substances 0.000 description 1
- MECHNRXZTMCUDQ-RKHKHRCZSA-N vitamin D2 Chemical compound C1(/[C@@H]2CC[C@@H]([C@]2(CCC1)C)[C@H](C)/C=C/[C@H](C)C(C)C)=C\C=C1\C[C@@H](O)CCC1=C MECHNRXZTMCUDQ-RKHKHRCZSA-N 0.000 description 1
- 235000005282 vitamin D3 Nutrition 0.000 description 1
- 239000011647 vitamin D3 Substances 0.000 description 1
- 229940021056 vitamin d3 Drugs 0.000 description 1
- 229960000237 vorinostat Drugs 0.000 description 1
- WAEXFXRVDQXREF-UHFFFAOYSA-N vorinostat Chemical compound ONC(=O)CCCCCCC(=O)NC1=CC=CC=C1 WAEXFXRVDQXREF-UHFFFAOYSA-N 0.000 description 1
- JFCFGYGEYRIEBE-YVLHJLIDSA-N wob38vs2ni Chemical compound CO[C@@H]([C@@]1(O)C[C@H](OC(=O)N1)[C@@H](C)[C@@H]1O[C@@]1(C)[C@@H](OC(=O)[C@H](C)N(C)C(=O)CCC(C)(C)S)CC(=O)N1C)\C=C\C=C(C)\CC2=CC(OC)=C(Cl)C1=C2 JFCFGYGEYRIEBE-YVLHJLIDSA-N 0.000 description 1
- 150000003732 xanthenes Chemical class 0.000 description 1
- 239000008782 xin-kang Substances 0.000 description 1
- 239000001052 yellow pigment Substances 0.000 description 1
- 229940055760 yervoy Drugs 0.000 description 1
- 229960000523 zalcitabine Drugs 0.000 description 1
- ARAIBEBZBOPLMB-UFGQHTETSA-N zanamivir Chemical compound CC(=O)N[C@@H]1[C@@H](N=C(N)N)C=C(C(O)=O)O[C@H]1[C@H](O)[C@H](O)CO ARAIBEBZBOPLMB-UFGQHTETSA-N 0.000 description 1
- 229960001028 zanamivir Drugs 0.000 description 1
- KGPGQDLTDHGEGT-JCIKCJKQSA-N zeven Chemical compound C=1C([C@@H]2C(=O)N[C@H](C(N[C@H](C3=CC(O)=C4)C(=O)NCCCN(C)C)=O)[C@H](O)C5=CC=C(C(=C5)Cl)OC=5C=C6C=C(C=5O[C@H]5[C@@H]([C@@H](O)[C@H](O)[C@H](O5)C(O)=O)NC(=O)CCCCCCCCC(C)C)OC5=CC=C(C=C5)C[C@@H]5C(=O)N[C@H](C(N[C@H]6C(=O)N2)=O)C=2C(Cl)=C(O)C=C(C=2)OC=2C(O)=CC=C(C=2)[C@H](C(N5)=O)NC)=CC=C(O)C=1C3=C4O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O KGPGQDLTDHGEGT-JCIKCJKQSA-N 0.000 description 1
- 229940126085 β‑Lactamase Inhibitor Drugs 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/06—Dipeptides
- C07K5/06008—Dipeptides with the first amino acid being neutral
- C07K5/06017—Dipeptides with the first amino acid being neutral and aliphatic
- C07K5/06034—Dipeptides with the first amino acid being neutral and aliphatic the side chain containing 2 to 4 carbon atoms
- C07K5/06052—Val-amino acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/337—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having four-membered rings, e.g. taxol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/47—Quinolines; Isoquinolines
- A61K31/475—Quinolines; Isoquinolines having an indole ring, e.g. yohimbine, reserpine, strychnine, vinblastine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7042—Compounds having saccharide radicals and heterocyclic rings
- A61K31/7048—Compounds having saccharide radicals and heterocyclic rings having oxygen as a ring hetero atom, e.g. leucoglucosan, hesperidin, erythromycin, nystatin, digitoxin or digoxin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/04—Peptides having up to 20 amino acids in a fully defined sequence; Derivatives thereof
- A61K38/14—Peptides containing saccharide radicals; Derivatives thereof, e.g. bleomycin, phleomycin, muramylpeptides or vancomycin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/56—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule
- A61K47/59—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes
- A61K47/60—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic macromolecular compound, e.g. an oligomeric, polymeric or dendrimeric molecule obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds, e.g. polyureas or polyurethanes the organic macromolecular compound being a polyoxyalkylene oligomer, polymer or dendrimer, e.g. PEG, PPG, PEO or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/65—Peptidic linkers, binders or spacers, e.g. peptidic enzyme-labile linkers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/04—Antibacterial agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/12—Antivirals
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/08—Tripeptides
- C07K5/0802—Tripeptides with the first amino acid being neutral
- C07K5/0804—Tripeptides with the first amino acid being neutral and aliphatic
- C07K5/0806—Tripeptides with the first amino acid being neutral and aliphatic the side chain containing 0 or 1 carbon atoms, i.e. Gly, Ala
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/10—Tetrapeptides
- C07K5/1021—Tetrapeptides with the first amino acid being acidic
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Organic Chemistry (AREA)
- Epidemiology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Genetics & Genomics (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Communicable Diseases (AREA)
- Oncology (AREA)
- Immunology (AREA)
- Gastroenterology & Hepatology (AREA)
- Virology (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
技术领域technical field
本发明属于医药领域,具体涉及一种分支型聚乙二醇连接子及其应用。The invention belongs to the field of medicine, and in particular relates to a branched polyethylene glycol linker and its application.
背景技术Background technique
抗体药物偶联物(Antibodydrugconjugate,ADC)是一类新型的靶向治疗药物,由靶向特异性抗原的单克隆抗体和小分子细胞毒药物通过连接子偶联而成,其联合了抗体及细胞毒药物两者的优势,与传统的小分子抗肿瘤药物相比,具有靶向性强、细胞毒性高、降解半衰期长、毒副作用低等特点。随着和等14个药物由FDA批准上市,近年来ADC药物发展迅速,目前大约有110个ADC药物处于临床试验中。Antibody drug conjugates (Antibodydrugconjugate, ADC) is a new type of targeted therapy drug, which is composed of a monoclonal antibody targeting a specific antigen and a small molecule cytotoxic drug coupled through a linker, which combines antibodies and cells Compared with traditional small molecule antineoplastic drugs, it has the characteristics of strong targeting, high cytotoxicity, long degradation half-life, and low toxic and side effects. along with and 14 other drugs have been approved by the FDA. In recent years, ADC drugs have developed rapidly. Currently, about 110 ADC drugs are in clinical trials.
ADC一般由抗体(antibody)、小分子细胞毒素(cytotoxin)和连接子(linker)三个部分组成。其中连接子的设计对ADC药物具有重要的意义,其本身必须使得药物在血液循环系统中保持稳定,以及到达靶组织之后可以快速有效地释放活性毒素。开发ADC连接子,存在着多种重要的考量因素,包括抗体的偶联位点、每分子抗体平均偶联细胞毒素的数量(drugtoantibodyratio,DAR)、连接子的可裂解性和亲水性等。连接子可分为可裂解连接子和不可裂解连接子两大类,可裂解连接子根据裂解机制的不同分为pH敏感型和酶敏感型。当前,酶敏感型连接子已经成为ADC的主流选择。研究比较成熟的酶敏感型连接子为依赖组织蛋白酶B裂解的二肽连接子,如缬氨酸-瓜氨酸(Valine-Citriline)。目前大多数ADC具有共同的结构特征,如通过马来酰亚胺接头连接。ADC药物已公开的连接子存在设计比较单一,水溶性不好等问题。ADC generally consists of three parts: antibody, small molecule cytotoxin and linker. Among them, the design of the linker is of great significance to the ADC drug, which must keep the drug stable in the blood circulation system and release the active toxin quickly and effectively after reaching the target tissue. In the development of ADC linkers, there are many important considerations, including the conjugation site of the antibody, the average number of cytotoxins per molecule of antibody (drug to antibody ratio, DAR), the cleavability and hydrophilicity of the linker, etc. Linkers can be divided into two categories: cleavable linkers and non-cleavable linkers, and cleavable linkers are divided into pH-sensitive and enzyme-sensitive types according to the different cleavage mechanisms. Currently, enzyme-sensitive linkers have become the mainstream choice for ADCs. The more mature enzyme-sensitive linker is a dipeptide linker dependent on cathepsin B cleavage, such as Valine-Citriline. Most current ADCs share common structural features, such as linkage via maleimide linkers. The published linkers of ADC drugs have problems such as relatively simple design and poor water solubility.
关于毒素与抗体的偶联方式,目前已上市ADC药物多采用赖氨酸或半胱氨酸随机偶联,由于偶联数目和位点的不确定性,导致生成的抗体药物偶联物的不均一性,进而导致产品各组分间药物动力学性质、效价以及毒性的不均一。为了解决ADC均一性问题,近年来定点偶联技术得到了更多的青睐,如基于天然糖基化位点的GlycoConnect技术,抗体的Fc区域N297糖基化位点在酶催化下,对糖链进行切割并连接上叠氮基团,然后叠氮基团与连接子上的环辛炔通过SPAAC(strain-promotedcopper-freeclick)反应,形成均一的DAR值为2的偶联物。Regarding the coupling method of toxins and antibodies, currently marketed ADC drugs are mostly randomly coupled with lysine or cysteine. Due to the uncertainty of the number and sites of coupling, the generated antibody-drug conjugates are inconsistent. Uniformity, which in turn leads to inhomogeneity in pharmacokinetic properties, potency and toxicity among the components of the product. In order to solve the problem of ADC uniformity, site-specific coupling technology has gained more favor in recent years, such as GlycoConnect technology based on natural glycosylation sites. After cleavage and connection of the azide group, the azide group reacts with the cyclooctyne on the linker by SPAAC (strain-promotedcopper-freeclick) to form a uniform conjugate with a DAR value of 2.
专利文件CN108743968A公开了一种半胱氨酸改造的抗体-毒素偶联物,抗体为半胱氨酸定点插入抗体,半胱氨酸插入位点包含kappa/λ轻链恒定区轻链和IgG抗体重链恒定区重链两类。Patent document CN108743968A discloses a cysteine modified antibody-toxin conjugate, the antibody is a cysteine site-directed insertion antibody, and the cysteine insertion site contains kappa/λ light chain constant region light chain and IgG anti- There are two types of heavy chains in the constant region of the heavy chain.
但通过基因工程改造的抗体-毒素偶联物引进了药物的不确定性,需要使用还原剂还原抗体,解除抗体上改造的半胱氨酸残基上的屏蔽,并通过阳离子交换层析或超滤换液等方式去除DTT与屏蔽物,然后使用氧化剂氧化抗体,使抗体的链间二硫键重新连接,过程繁琐复杂限制了该发明的使用。However, the antibody-toxin conjugates introduced by genetic engineering introduce uncertainty in the drug, and it is necessary to use a reducing agent to reduce the antibody, release the shield on the modified cysteine residue on the antibody, and pass through cation exchange chromatography or ultrasonography. Removing DTT and shielding materials by means of filtration and replacement, and then using an oxidizing agent to oxidize the antibody to reconnect the interchain disulfide bonds of the antibody, the cumbersome and complicated process limits the application of this invention.
专利文件CN103083680B公开了一种具有聚乙二醇-氨基酸寡肽-依诺替康结构通式的药物结合物。在所述结合物中,每一个聚乙二醇端基通过氨基酸寡肽可以与多个依诺替康相连,药物的负载率大大提高。通过亲水性聚合物的改性可对依诺替康提供保护,改善药物吸收,延长作用时间,增强疗效,降低给药剂量及避免毒副作用。Patent document CN103083680B discloses a drug conjugate having a general structural formula of polyethylene glycol-amino acid oligopeptide-irnotecan. In the conjugate, each end group of polyethylene glycol can be connected with multiple irinotecans through amino acid oligopeptides, and the drug loading rate is greatly improved. The modification of the hydrophilic polymer can protect inotecan, improve drug absorption, prolong action time, enhance curative effect, reduce dosage and avoid toxic and side effects.
但此抗体药物偶联物的均一性不高,进而导致产品各组分间药物动力学性质、效价以及毒性的不均一、产品批次可重复性较差、治疗指数较低。However, the homogeneity of the antibody-drug conjugate is not high, which leads to inhomogeneity in pharmacokinetic properties, potency and toxicity among the components of the product, poor reproducibility of product batches, and low therapeutic index.
针对上述现有技术中存在的抗体偶联药物均一性较差、连接子设计较单一、水溶性不好等缺陷,迫切需要开发新的连接子技术。引入聚乙二醇基团,增加连接子亲水性,分支型连接子还可以实现增加载药量的目的,同时使用DBCO接头可实现定点偶联,提高抗体药物偶联物的均一性。In view of the defects in the above-mentioned existing technologies such as poor uniformity of antibody-drug conjugates, single linker design, and poor water solubility, it is urgent to develop new linker technologies. The polyethylene glycol group is introduced to increase the hydrophilicity of the linker, and the branched linker can also achieve the purpose of increasing the drug loading. At the same time, the DBCO linker can be used to achieve fixed-point coupling and improve the uniformity of the antibody-drug conjugate.
发明内容Contents of the invention
为了解决现有技术中提到的问题,本发明提供了一种分支型聚乙二醇连接子,其具有式(Ⅰ)所示结构:In order to solve the problems mentioned in the prior art, the present invention provides a branched polyethylene glycol linker, which has the structure shown in formula (I):
其中,所述Q选自: 中的一种;Wherein, the Q is selected from: one of
R5和R6各自独立地具有—X4—Q1的结构,Q1选自-H、-F、-Cl、-Br、-I、-SO2、-NO2、C1-12链烷基、C3-12环烷基、C6-12芳烷基;R 5 and R 6 each independently have the structure of -X 4 -Q 1 , Q 1 is selected from -H, -F, -Cl, -Br, -I, -SO 2 , -NO 2 , C 1-12 chains Alkyl, C 3-12 cycloalkyl, C 6-12 aralkyl;
X4选自由单键、-O-、-S-、C1-12链亚烷基、C3-12环亚烷基、C6-12芳亚烷基、 及中的一种或多种组成的组;X is selected from single bond, -O-, -S-, C 1-12 chain alkylene, C 3-12 cycloalkylene, C 6-12 aralkylene , and A group consisting of one or more of them;
X是连接基团,选自由单键、-O-、-S-、C1-12直链/支链亚烷基、C3-12环亚烷基、C6-12芳亚烷基、 及中的一种或多种组成的组,其中R9选自:-H、C1-10直链/支链烷基;X is a linking group selected from single bond, -O-, -S-, C 1-12 straight chain/branched alkylene, C 3-12 cycloalkylene, C 6-12 aralkylene , and A group consisting of one or more of, wherein R 9 is selected from: -H, C 1-10 straight chain/branched alkyl;
所述W1、W2独立的选自单键、直链或支链的C1-6的烷基;The W 1 and W 2 are independently selected from single bond, linear or branched C 1-6 alkyl groups;
L1a,L1b,L1c独立地选自单键、 L 1a , L 1b , L 1c are independently selected from single bonds,
所述n选自1~24的整数;The n is an integer selected from 1 to 24;
比如,n选自1、2、3、4、5、6、7、8、9、10、11、12、13、14、15、16、17、18、19、20、21、22、23或24。For example, n is selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24.
X1、X2是连接基团,独立地选自由单键、-O-、-S-、C1-12直链/支链亚烷基、C3-12环亚烷基、C6-12芳亚烷基、 及中的一种或多种组成的组;X 1 and X 2 are linking groups, independently selected from single bond, -O-, -S-, C 1-12 linear/branched alkylene, C 3-12 cycloalkylene, C 6- 12 aralkylene, and A group consisting of one or more of them;
L2、L3是连接基团,为其中A为肽类连接子,B为自降解基团或不存在,L 2 and L 3 are linking groups, which are Wherein A is a peptide linker, B is a self-degrading group or does not exist,
当B为自降解基团,B选自: When B is a self-degrading group, B is selected from:
其中,Y1、Y2各自独立地选自:-H、卤素、-OC1-10烷基、C1-10直链/支链烷基、C3-10环烷基、-OH、 Wherein, Y 1 and Y 2 are each independently selected from: -H, halogen, -OC 1-10 alkyl, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl, -OH,
Y3、Y4各自独立地选自:-H、C1-10直链/支链烷基、C3-10环烷基、 Y 3 and Y 4 are each independently selected from: -H, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl,
X3选自由-O-、-S-、C1-12直链/支链烷基、C3-12环烷基、C6-12芳环基、 及或其组合组成的组;X 3 is selected from -O-, -S-, C 1-12 straight chain/branched chain alkyl, C 3-12 cycloalkyl, C 6-12 aromatic ring group, and or a combination thereof;
R20选自:-H、C1-12直链/支链烷基、C1-12烷氧基、-NO2、-CN、-F、-Cl、-Br、-I、-SO2、C3-12环烷基、C6-12芳环基、 R 20 is selected from: -H, C 1-12 straight chain/branched chain alkyl, C 1-12 alkoxy, -NO 2 , -CN, -F, -Cl, -Br, -I, -SO 2 , C 3-12 cycloalkyl, C 6-12 aromatic ring group,
A为肽类连接子,包括一个或多个相同或不同的多肽残基,所述的多肽残基包括两个或两个以上相同或不同的氨基酸残基或氨基酸残基衍生物。A is a peptide linker, including one or more identical or different polypeptide residues, and said polypeptide residues include two or more identical or different amino acid residues or amino acid residue derivatives.
进一步的,所述Q为优选的,所述Q为 Further, the Q is Preferably, the Q is
所述R5、R6独立的选自H、C1-3的烷基,优选的,R5、R6均为H;The R 5 and R 6 are independently selected from H and C 1-3 alkyl groups, preferably, both R 5 and R 6 are H;
所述X选自C1-12直链/支链亚烷基、组成的组,其中R9为H、C1-3直链/支链烷基;Said X is selected from C 1-12 straight chain/branched chain alkylene, The group consisting of, wherein R 9 is H, C 1-3 straight chain/branched alkyl;
优选的,所述X为所述R7为C1-12的亚烷基;更优选的,所述R7为C1-4的亚烷基;Preferably, the X is The R 7 is a C 1-12 alkylene group; more preferably, the R 7 is a C 1-4 alkylene group;
所述L1a,L1b,L1c独立地选自单键、 The L 1a , L 1b , L 1c are independently selected from single bonds,
所述W4选自单键、C1-6直链或支链的亚烷基;The W 4 is selected from a single bond, C 1-6 linear or branched alkylene;
优选的,所述n选自1~12的整数;Preferably, said n is an integer selected from 1 to 12;
所述氨基酸选自:丙氨酸、精氨酸、天冬酰胺、天冬氨酸、半胱氨酸、谷氨酰胺、谷氨酸、甘氨酸、组氨酸、异亮氨酸、亮氨酸、赖氨酸、甲硫氨酸、苯丙氨酸、脯氨酸、丝氨酸、苏氨酸、色氨酸、酪氨酸、缬氨酸、瓜氨酸、鸟氨酸和胱氨酸中的一种或两种以上的组合;The amino acid is selected from: alanine, arginine, asparagine, aspartic acid, cysteine, glutamine, glutamic acid, glycine, histidine, isoleucine, leucine , lysine, methionine, phenylalanine, proline, serine, threonine, tryptophan, tyrosine, valine, citrulline, ornithine and cystine One or a combination of two or more;
优选的,所述的氨基酸选自:缬氨酸、瓜氨酸、甘氨酸、丙氨酸、苯丙氨酸、酪氨酸、异亮氨酸、亮氨酸和精氨酸中的一种或两种以上的组合;Preferably, the amino acid is selected from one or more of valine, citrulline, glycine, alanine, phenylalanine, tyrosine, isoleucine, leucine and arginine A combination of two or more;
更优选的,所述的多肽残基包括缬氨酸-瓜氨酸、缬氨酸-丙氨酸和甘氨酸-甘氨酸-苯丙氨酸-甘氨酸中的一种。More preferably, the polypeptide residues include one of valine-citrulline, valine-alanine and glycine-glycine-phenylalanine-glycine.
进一步的,当B为自降解基团时,所述的A选自 Further, when B is a self-degrading group, said A is selected from
其中,A1、A2、A3……Ay为相同或不同,所述A1、A2、A3……Ay结构独立选自:单键、Wherein, A 1 , A 2 , A 3 ... A y are the same or different, and the structures of A 1 , A 2 , A 3 ... A y are independently selected from: single bond,
所述y选自1~5的整数;The y is an integer selected from 1 to 5;
所述j选自1~10的整数;The j is an integer selected from 1 to 10;
比如,j可以选自1、2、3、4、5、6、7、8、9或10的整数;For example, j can be an integer selected from 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10;
优选的,所述j为2~4的整数;Preferably, the j is an integer of 2-4;
所述Ya选自-H、卤素、-OC1-10烷基、C1-10直链/支链烷基、C3-10环烷基、-OH、-(C1-6亚烷基)-OH、-(C1-6亚烷基)-SH、-(CH2)1-6-环烷基、-(C1-6亚烷基)-芳环基、-(C1-6亚烷基)-杂环烷基、-(C1-6亚烷基)-杂环芳香基、-(C1-6亚烷基)-COOH、-(C1-6亚烷基)-CONH2、-(C1-6亚烷基)-NH2和中的一种;The Y a is selected from -H, halogen, -OC 1-10 alkyl, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl, -OH, -(C 1-6 alkylene base)-OH, -(C 1-6 alkylene)-SH, -(CH 2 ) 1-6 -cycloalkyl, -(C 1-6 alkylene)-aromatic ring group, -(C 1 -6 alkylene)-heterocycloalkyl, -(C 1-6 alkylene)-heterocyclic aryl, -(C 1-6 alkylene)-COOH, -(C 1-6 alkylene )-CONH 2 , -(C 1-6 alkylene)-NH 2 and one of
所述A1、A2、A3……Ay至少有一个结构;Said A 1 , A 2 , A 3 ... A y has at least one structure;
优选地,所述的Ya选自:-H、-C1-6直链/支链烷基、-(C1-6亚烷基)-芳环基;Preferably, said Y a is selected from: -H, -C 1-6 straight chain/branched chain alkyl, -(C 1-6 alkylene)-aromatic ring group;
更优选的,所述Ya选自:-H、甲基、乙基、丙基、异丙基、 More preferably, the Y a is selected from: -H, methyl, ethyl, propyl, isopropyl,
进一步的,所述B不存在或选自: Further, said B does not exist or is selected from:
优选的,所述B选自: Preferably, said B is selected from:
其中,Y1、Y2各自独立地选自:-H、卤素、-OC1-10烷基、C1-10直链/支链烷基、C3-10环烷基、-OH;Wherein, Y 1 and Y 2 are each independently selected from: -H, halogen, -OC 1-10 alkyl, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl, -OH;
Y3、Y4各自独立地选自:-H、C1-10直链/支链烷基、C3-10环烷基、 Y 3 and Y 4 are each independently selected from: -H, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl,
进一步的,所述Q为 Further, the Q is
所述X为 The X is
所述L1a,L1b,L1c独立地选自单键或 Said L 1a , L 1b , L 1c are independently selected from a single bond or
所述A选自 Said A is selected from
所述B为:或不存在。Said B is: or does not exist.
进一步的,所述L2、L3独立的选自:Further, said L 2 and L 3 are independently selected from:
进一步的,所述连接子选自:Further, the linker is selected from:
本发明另一方面还提供一种分支型聚乙二醇连接子-药物缀合物,具有式(II)所示结构:Another aspect of the present invention also provides a branched polyethylene glycol linker-drug conjugate, which has the structure shown in formula (II):
其中,所述Q选自: 中的一种;Wherein, the Q is selected from: one of
R5和R6各自独立地具有—X4—Q1的结构,Q1选自-H、-F、-Cl、-Br、-I、-SO2、-NO2、C1-12链烷基、C3-12环烷基、C6-12芳烷基;R 5 and R 6 each independently have the structure of -X 4 -Q 1 , Q 1 is selected from -H, -F, -Cl, -Br, -I, -SO 2 , -NO 2 , C 1-12 chains Alkyl, C 3-12 cycloalkyl, C 6-12 aralkyl;
X4选自由单键、-O-、-S-、C1-12链亚烷基、C3-12环亚烷基、C6-12芳亚烷基、 及中的一种或多种组成的组;X is selected from single bond, -O-, -S-, C 1-12 chain alkylene, C 3-12 cycloalkylene, C 6-12 aralkylene , and A group consisting of one or more of them;
X是连接基团,选自由单键、-O-、-S-、C1-12直链/支链亚烷基、C3-12环亚烷基、C6-12芳亚烷基、 及中的一种或多种组成的组,其中R9选自:-H、C1-10直链/支链烷基;X is a linking group selected from single bond, -O-, -S-, C 1-12 straight chain/branched alkylene, C 3-12 cycloalkylene, C 6-12 aralkylene , and A group consisting of one or more of, wherein R 9 is selected from: -H, C 1-10 straight chain/branched alkyl;
所述W1、W2独立的选自单键、直链或支链的C1-6的烷基;The W 1 and W 2 are independently selected from single bond, linear or branched C 1-6 alkyl groups;
L1a,L1b,L1c独立地选自单键、 L 1a , L 1b , L 1c are independently selected from single bonds,
所述n选自1~24的整数,可以选自1、2、3、4、5、6、7、8、9、10、11、12、13、14、15、16、17、18、19、20、21、22、23或24。The n is an integer selected from 1 to 24, and may be selected from 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or 24.
X1、X2是连接基团,独立地选自由单键、-O-、-S-、C1-12直链/支链亚烷基、C3-12环亚烷基、C6-12芳亚烷基、 及中的一种或多种组成的组,其中R10选自:-H、C1-10直链/支链烷基;X 1 and X 2 are linking groups, independently selected from single bond, -O-, -S-, C 1-12 linear/branched alkylene, C 3-12 cycloalkylene, C 6- 12 aralkylene, and A group consisting of one or more of, wherein R 10 is selected from: -H, C 1-10 straight chain/branched alkyl;
L2’、L3’是连接基团,为其中A’为肽类连接子,B’为自降解基团,B’选自: 其中,Y1、Y2各自独立地选自:-H、卤素、-OC1-10烷基、C1-10直链/支链烷基、C3-10环烷基、-OH、 L 2 ', L 3 ' are linking groups, which are Wherein A' is a peptide linker, B' is a self-degrading group, and B' is selected from: Wherein, Y 1 and Y 2 are each independently selected from: -H, halogen, -OC 1-10 alkyl, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl, -OH,
Y3、Y4各自独立地选自:-H、C1-10直链/支链烷基、C3-10环烷基、 Y 3 and Y 4 are each independently selected from: -H, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl,
X3选自由-O-、-S-、C1-12直链/支链烷基、C3-12环烷基、C6-12芳环基、 及或其组合组成的组;X 3 is selected from -O-, -S-, C 1-12 straight chain/branched chain alkyl, C 3-12 cycloalkyl, C 6-12 aromatic ring group, and or a combination thereof;
R20选自:-H、C1-12直链/支链烷基、C1-12烷氧基、-NO2、-CN、-F、-Cl、-Br、-I、-SO2、C3-12环烷基、C6-12芳环基、 R 20 is selected from: -H, C 1-12 straight chain/branched chain alkyl, C 1-12 alkoxy, -NO 2 , -CN, -F, -Cl, -Br, -I, -SO 2 , C 3-12 cycloalkyl, C 6-12 aromatic ring group,
W3为C1-6直链/支链亚烷基;W 3 is C 1-6 straight chain/branched chain alkylene;
所述的肽类连接子包括一个或多个相同或不同的多肽残基,所述的多肽残基包括两个或两个以上相同或不同的氨基酸残基或氨基酸残基衍生物;The peptide linker includes one or more identical or different polypeptide residues, and the polypeptide residues include two or more identical or different amino acid residues or amino acid residue derivatives;
优选的,所述氨基酸选自:丙氨酸、精氨酸、天冬酰胺、天冬氨酸、半胱氨酸、谷氨酰胺、谷氨酸、甘氨酸、组氨酸、异亮氨酸、亮氨酸、赖氨酸、甲硫氨酸、苯丙氨酸、脯氨酸、丝氨酸、苏氨酸、色氨酸、酪氨酸、缬氨酸、瓜氨酸、鸟氨酸和胱氨酸中的一种或两种以上的组合,优选地,所述的氨基酸选自:缬氨酸、瓜氨酸、甘氨酸、丙氨酸、苯丙氨酸、酪氨酸、异亮氨酸、亮氨酸和精氨酸中的一种或两种以上的组合;更优选的,所述的多肽残基包括缬氨酸-瓜氨酸、缬氨酸-丙氨酸和甘氨酸-甘氨酸-苯丙氨酸-甘氨酸中的一种;Preferably, the amino acid is selected from: alanine, arginine, asparagine, aspartic acid, cysteine, glutamine, glutamic acid, glycine, histidine, isoleucine, Leucine, Lysine, Methionine, Phenylalanine, Proline, Serine, Threonine, Tryptophan, Tyrosine, Valine, Citrulline, Ornithine, and Cystine One or more combinations of acids, preferably, the amino acid is selected from: valine, citrulline, glycine, alanine, phenylalanine, tyrosine, isoleucine, One or more combinations of leucine and arginine; more preferably, the polypeptide residues include valine-citrulline, valine-alanine and glycine-glycine-benzene One of alanine-glycine;
所述D为药物分子。The D is a drug molecule.
进一步的,所述Q为 优选的,所述Q为 Further, the Q is Preferably, the Q is
所述R5、R6独立的选自H、C1-3的烷基,优选的,R5、R6均为H;The R 5 and R 6 are independently selected from H and C 1-3 alkyl groups, preferably, both R 5 and R 6 are H;
所述X选自C1-12直链/支链亚烷基、组成的组,其中R9为H、C1-3直链/支链烷基;Said X is selected from C 1-12 straight chain/branched chain alkylene, The group consisting of, wherein R 9 is H, C 1-3 straight chain/branched alkyl;
优选的,所述X为所述R7为C1-12的亚烷基;更优选的,所述R7为C1-4的亚烷基;Preferably, the X is The R 7 is a C 1-12 alkylene group; more preferably, the R 7 is a C 1-4 alkylene group;
所述L1a,L1b,L1c独立地选自单键、 The L 1a , L 1b , L 1c are independently selected from single bonds,
所述W4选自单键、C1-6直链或支链的烷基;The W 4 is selected from a single bond, C 1-6 straight chain or branched alkyl;
优选的,所述n选自1~12的整数,比如n可以为1、2、3、4、5、6、7、8、9、10、11或12。Preferably, the n is an integer selected from 1-12, for example, n can be 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11 or 12.
比如,所述的A’选自 For example, said A' is selected from
其中,A1、A2、A3……Ay为相同或不同,所述A1、A2、A3……Ay结构独立选自:单键、Wherein, A 1 , A 2 , A 3 ... A y are the same or different, and the structures of A 1 , A 2 , A 3 ... A y are independently selected from: single bond,
所述y选自1~10的整数;The y is an integer selected from 1 to 10;
比如,所述y可以为1、2、3、4、5、6、7、8、9或10;For example, the y can be 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10;
优选的,所述y为2~4的整数;Preferably, the y is an integer of 2 to 4;
所述j选自1~10的整数;The j is an integer selected from 1 to 10;
比如,所述j可以为1、2、3、4、5、6、7、8、9或10;For example, the j may be 1, 2, 3, 4, 5, 6, 7, 8, 9 or 10;
优选的,所述j为2~4的整数;Preferably, the j is an integer of 2-4;
所述Ya选自-H、卤素、-OC1-10烷基、C1-10直链/支链烷基、C3-10环烷基、-OH、-(C1-6亚烷基)-OH、-(C1-6亚烷基)-SH、-(CH2)1-6-环烷基、-(C1-6亚烷基)-芳环基、-(C1-6亚烷基)-杂环烷基、-(C1-6亚烷基)-杂环芳香基、-(C1-6亚烷基)-COOH、-(C1-6亚烷基)-CONH2、-(C1-6亚烷基)-NH2和中的一种;The Y a is selected from -H, halogen, -OC 1-10 alkyl, C 1-10 straight chain/branched chain alkyl, C 3-10 cycloalkyl, -OH, -(C 1-6 alkylene base)-OH, -(C 1-6 alkylene)-SH, -(CH 2 ) 1-6 -cycloalkyl, -(C 1-6 alkylene)-aromatic ring group, -(C 1 -6 alkylene)-heterocycloalkyl, -(C 1-6 alkylene)-heterocyclic aryl, -(C 1-6 alkylene)-COOH, -(C 1-6 alkylene )-CONH 2 , -(C 1-6 alkylene)-NH 2 and one of
所述A1、A2、A3……Ay至少有一个结构;Said A 1 , A 2 , A 3 ... A y has at least one structure;
优选地,所述的Ya选自:-H、-C1-6直链/支链烷基、-(C1-6亚烷基)-芳环基;Preferably, said Y a is selected from: -H, -C 1-6 straight chain/branched chain alkyl, -(C 1-6 alkylene)-aromatic ring group;
更优选的,所述Ya选自:-H、甲基、乙基、丙基、异丙基、 More preferably, the Y a is selected from: -H, methyl, ethyl, propyl, isopropyl,
所述B’选自: The B' is selected from:
其中,Y1、Y2各自独立地选自:-H、-OC1-10烷基、C1-10直链/支链烷基、-OH;Y3、Y4各自独立地选自:-H、C1-10直链/支链烷基; Wherein, Y 1 , Y 2 are each independently selected from: -H, -OC 1-10 alkyl, C 1-10 straight chain/branched chain alkyl, -OH; Y 3 , Y 4 are each independently selected from: -H, C 1-10 straight chain/branched chain alkyl;
W3选自C1-6直链/支链亚烷基;W 3 is selected from C 1-6 straight chain/branched chain alkylene;
优选的,Y1、Y2、Y3、Y4各自独立地选自H、C1-5直链/支链烷基。Preferably, Y 1 , Y 2 , Y 3 , and Y 4 are each independently selected from H, C 1-5 straight chain/branched chain alkyl.
进一步的,所述Q为 Further, the Q is
所述X为 The X is
所述L1a,L1b,L1c独立地选自单键或 Said L 1a , L 1b , L 1c are independently selected from a single bond or
所述A’选自 The A' is selected from
所述B’选自: The B' is selected from:
进一步的,所述L2’、L3’独立的选自:Further, said L 2 ' and L 3 ' are independently selected from:
进一步的,所述D为化疗剂,选自于:烷基化剂,选自氮芥:氯苯那普,氯普那嗪,环磷酰胺,达喀尔巴嗪,雌二醇氮芥,异环磷酰胺,氮芥,盐酸二甲氧胺,氧化二氮芥,盐酸氨氯地平,麦考酚酸,卫矛醇,呱泊溴烷,新氮芥,苯芥胆甾醇,松龙苯芥,噻替呱,曲磷胺对,尿嘧啶;CC-1065和阿多来新、卡折来新、比折来新或其合成类似物;多卡霉素及其合成类似物,KW-2189或CBI-TMI、;苯并二氮二聚体或吡咯并苯二氮(PBD)二聚体,托美霉素二聚体,吲哚并苯并二氮杂二聚体,咪唑并苯并噻二氮杂二聚体,或恶唑烷并苯并二氮杂的二聚体;亚硝基脲:包括卡莫司汀,洛莫司汀,氯化梭菌素,福莫司汀,尼莫司汀,拉莫司汀;烷基磺酸盐:包括白苏芬、树苏芬、磺胺异丙磺胺和皮苏芬;三氮烯或达喀尔巴嗪;含铂化合物:包括卡铂,顺铂,奥沙利铂;吖丙啶类,苯并二氢吡喃酮,卡洛酮,美妥替派和乌雷多巴;乙烯亚胺和甲基三聚氰胺,包括六甲蜜胺,三亚乙基三胺,三乙基磷酰胺,三亚乙基硫代磷酰胺和三羟甲基甲基胺;Further, the D is a chemotherapeutic agent, selected from: alkylating agents, nitrogen mustards: chlorpheniramine, chlorpronazine, cyclophosphamide, dacarbazine, estramustine, Ifosfamide, nitrogen mustard, dimethoxamine hydrochloride, dinitrogen mustard, amlodipine hydrochloride, mycophenolic acid, dulcitol, pipbromide, new nitrogen mustard, benzene mustard cholesterol, turpentine mustard, thiotegua, trifosamide, uracil; CC-1065 and adolaisine, cardzelesine, bizelesine or their synthetic analogues; duocamycin and its synthetic analogues, KW- 2189 or CBI-TMI, benzodiazepine dimer or pyrrolobenzodiazepine (PBD) dimer, tomemycin dimer, indolobenzodiazepine dimer, imidazobenzene Dimers of thiadiazepines, or dimers of oxazolidinebenzodiazepines; nitrosoureas: including carmustine, lomustine, clomustine, formustine , nimustine, lamustine; alkyl sulfonates: including basiphen, shusuphen, sulfaprosulfone, and pisuphen; triazenes or dacarbazine; platinum-containing compounds: including carboplatin , cisplatin, oxaliplatin; aziridines, chromanone, caloxone, methutepa, and uredopa; ethyleneimine and methylmelamine, including hexamethylmelamine, Sanya Ethyltriamine, triethylphosphoramide, triethylenethiophosphoramide and trimethylolmethylamine;
所述D为植物生物碱:选自长春花生物碱,包括长春新碱,长春碱,长春地辛,长春瑞滨,去甲长春碱;紫杉醇类:包括紫杉醇,多西紫杉醇及其类似物;美登素类包括DM1、DM2、DM3、DM4、DM5、DM6、DM7、美登素、安沙霉素及其类似物);cryptophycin(包括cryptophycin1和cryptophycin8);埃博霉素,软珊瑚醇,迪莫利德,草苔虫内酯,海兔毒素,澳瑞斯汀,tubulysins,cephalostatin;pancratistatin;erbulins;sarcodictyin;海绵抑制素;The D is a plant alkaloid: selected from vinca alkaloids, including vincristine, vinblastine, vindesine, vinorelbine, norvinblastine; paclitaxel: including paclitaxel, docetaxel and analogs thereof; Maytansinoids include DM1, DM2, DM3, DM4, DM5, DM6, DM7, maytansine, ansamycin and their analogs); cryptophycin (including cryptophycin1 and cryptophycin8); Molide, bryostatin, dolastatin, auristin, tubulysins, cephalostatin; pancratistatin; erbulins; sarcodictyn; spongostatin;
所述D为DNA拓扑异构酶抑制剂:选自依托泊苷替尼,包括9-氨基喜树碱、喜树碱、克立那托、多拉霉素、依托泊苷、磷酸依托泊苷、伊立替康、依喜替康、Dxd、SN38、米托蒽醌、诺消灵、视黄酸(视黄醇)、替尼泊苷、拓扑替康、9-硝基喜树碱或RFS2000、丝裂霉素及其类似物;The D is a DNA topoisomerase inhibitor: selected from etoposide tinib, including 9-aminocamptothecin, camptothecin, clinator, doramycin, etoposide, etoposide phosphate, Irinotecan, exinotecan, Dxd, SN38, mitoxantrone, nodiclofen, retinoic acid (retinol), teniposide, topotecan, 9-nitrocamptothecin or RFS2000, Mitomycin and its analogues;
所述D为选自抗叶酸剂(DHFR抑制剂,包括甲氨蝶呤、曲麦克特、二甲叶酸、蝶罗呤、氨喋呤(4-氨基苯甲酸)或其他叶酸类似物);IMP脱氢酶抑制剂(包括麦考酚酸、噻唑呋林、利巴韦林、EICAR);核糖核苷酸还原酶抑制剂(包括羟基脲、去铁胺);嘧啶类似物:尿嘧啶类似物(包括安西他滨、阿扎胞苷、6-氮尿嘧啶、卡培他滨(希罗达)、卡莫氟、阿糖胞苷、双脱氧尿苷、脱氧氟尿苷、依诺他滨、5-氟尿嘧啶、氟尿苷、ratitrexed(Tomudex))和胞嘧啶类似物(包括阿糖胞苷,胞嘧啶阿拉伯糖苷,氟达拉滨);嘌呤类似物(包括硫唑嘌呤,氟达拉滨,巯嘌呤,硫胺素,硫鸟嘌呤);叶酸补充剂,弗洛林酸;和烟酰胺磷酸核糖基转移酶(NAMPT)的抑制剂;Said D is selected from antifolates (DHFR inhibitors, including methotrexate, trimact, dimethyl folic acid, pteroxin, aminopterin (4-aminobenzoic acid) or other folic acid analogues); IMP Dehydrogenase inhibitors (including mycophenolic acid, thiazofurin, ribavirin, EICAR); ribonucleotide reductase inhibitors (including hydroxyurea, deferoxamine); pyrimidine analogs: uracil analogs (including ancitabine, azacitidine, 6-azuracil, capecitabine (Xeloda), carmofur, cytarabine, dideoxyuridine, deoxyfluridine, enoxitabine , 5-fluorouracil, floxuridine, ratitrexed (Tomudex)) and cytosine analogs (including cytarabine, cytosine arabinoside, fludarabine); purine analogs (including azathioprine, fludarabine , mercaptopurine, thiamine, thioguanine); folic acid supplements, florinic acid; and inhibitors of nicotinamide phosphoribosyltransferase (NAMPT);
所述D为激素疗法剂:选自受体拮抗剂:抗雌激素(包括甲地孕酮,雷洛昔芬,他莫昔芬),LHRH兴奋剂(包括戈斯他林,醋酸亮丙瑞林);抗雄激素药(包括比卡鲁胺,氟他胺,卡鲁司酮,丙酸倍他雄酮,表雄甾醇,戈舍瑞林,亮丙瑞林,美替利定,尼鲁米特,睾内酯,曲洛司坦及其他雄激素抑制剂);维甲类化合物:维生素D3类似物(包括CB1093,EB1089,KH1060,胆钙化醇,麦角钙化甾醇);光动力疗法剂(包括维替泊芬,酞菁,光敏剂Pc4,去甲氧基-竹红菌素A);细胞因子(干扰素-α,干扰素-γ,肿瘤坏死因子(TNF),含TNF的人蛋白);The D is a hormone therapy agent: selected from receptor antagonists: antiestrogens (including megestrol, raloxifene, tamoxifen), LHRH stimulants (including gostaline, leuprolide acetate Lin); anti-androgen drugs (including bicalutamide, flutamide, caluspristone, betandrosterone propionate, epiandrosterol, goserelin, leuprolide, metilidine, nitric acid Luthimide, testolide, trilosteine and other androgen inhibitors); retinoids: vitamin D3 analogs (including CB1093, EB1089, KH1060, cholecalciferol, ergocalciferol); photodynamic therapy agents (including verteporfin, phthalocyanine, photosensitizer Pc4, demethoxy-hypocretin A); cytokines (interferon-α, interferon-γ, tumor necrosis factor (TNF), TNF-containing human protein);
所述D为激酶抑制剂,选自BIBW2992(抗-EGFR/Erb2),伊马替尼,吉非替尼,呱加他尼,索拉非尼,达沙替尼,舒尼替尼,厄洛替尼,尼洛替尼,拉帕替尼,阿西替尼,帕唑帕尼,凡德他尼,E7080(抗VEGFR2),mubritinib,普纳替尼(AP24534),bafetinib(INNO-406),bosutinib(SKI-606),卡博替尼,维莫德吉,iniparib,鲁索利替尼,CYT387,阿西替尼,tivozanib,索拉非尼,贝伐单抗,西妥昔单抗,曲妥珠单抗,雷珠单抗,帕尼单抗,伊斯平斯;The D is a kinase inhibitor selected from BIBW2992 (anti-EGFR/Erb2), imatinib, gefitinib, guagatanib, sorafenib, dasatinib, sunitinib, erb2 Lotinib, nilotinib, lapatinib, axitinib, pazopanib, vandetanib, E7080 (anti-VEGFR2), mubritinib, ponatinib (AP24534), bafetinib (INNO-406 ), bosutinib (SKI-606), cabozantinib, vimodegib, iniparib, ruxolitinib, CYT387, axitinib, tivozanib, sorafenib, bevacizumab, cetuximab Anti-, trastuzumab, ranibizumab, panitumumab, Espins;
所述D为聚(ADP-核糖)聚合酶(PARP)抑制剂,选自奥拉帕里、尼拉帕里、依尼帕里、塔拉佐帕里、维利帕里、维利帕里、CEP9722(Cephalon)、E7016(Eisai)、BGB-290(Beigene)、3-氨基苯甲酰酰胺;Described D is poly (ADP-ribose) polymerase (PARP) inhibitor, is selected from Olapari, Nirapari, Inipari, Tarazopari, Velipari, Velipari , CEP9722 (Cephalon), E7016 (Eisai), BGB-290 (Beigene), 3-aminobenzamide;
所述D为抗生素:选自烯二炔类抗生素(选自加利车霉素、加利车霉素γ1、δ1、α1和β1,达因霉素,包括达因霉素A和脱氧米霉素,埃斯培拉霉素,卡塔尔霉素,C-1027,maduropeptin,或新卡嗪奥斯汀和相关色蛋白烯二炔抗生素),aclacinomysins,放线菌素,安曲霉素,重氮丝氨酸,博来霉素,卡诺霉素,卡拉霉素,洋红霉素,嗜癌素,色霉素,达金霉素,柔红霉素,去柔红霉素,6-重氮-5-氧代-L-去甲亮氨酸,阿霉素,吗啉-阿霉素,氰基吗啉-阿霉素,2-吡咯啉阿霉素和脱氧柔红霉素,表柔比星,阿柔比星,伊达比星,马可霉素,nitomycin,霉酚酸,诺加霉素,橄榄霉素,Peplomycin,potfiromycin,嘌呤霉素,奎拉霉素,罗道霉素,链黑霉素,链脲霉素,杀结核菌素,乌苯美司,净司他丁,佐柔比星;Said D is an antibiotic: selected from enediyne antibiotics (selected from calicheamicin, calicheamicin γ1, δ1, α1 and β1, dynemycin, including dynemycin A and deoxyoryzae esperamycin, catamycin, C-1027, maduropeptin, or neocarbazine Austin and related chromopin enediyne antibiotics), aclacinomysins, actinomycin, antramycin, azaserine, Bleomycin, kanomycin, karamycin, carmine, carcinophilin, chromomycin, daunorubicin, daunorubicin, dedaunorubicin, 6-diazo-5-oxo Di-L-norleucine, doxorubicin, morpholine-doxorubicin, cyanomorpholine-doxorubicin, 2-pyrroline doxorubicin and deoxydaunorubicin, epirubicin, a Ruubicin, idarubicin, marcomycin, nitomycin, mycophenolic acid, nogamycin, olivine, peplomycin, potfiromycin, puromycin, quilamycin, rodaumycin, streptomycin streptozotocin, tuberculin, ubenimex, netastatin, zorubicin;
所述D为聚酮化合物(番荔素),bullatacin和bullatacinone;吉西他滨,环氧酶素和卡菲偌米布,硼替佐米,沙利度胺,来那度胺,pomalidomide,tosedostat,zybrestat,PLX4032,STA-9090,Stimuvax,allovectin-7,Xegeva,Provenge,Yervoy,异戊二烯化抑制剂和洛伐他汀,多巴胺能神经毒素(选自星形孢菌素),放线菌素(包含放线菌素D,更生霉素),鹅膏毒素,博莱霉素(包括博来霉素A2,博莱霉素B2,培洛霉素),蒽环类抗生素,包括柔红霉素,阿霉素(亚德里亚霉素),伊达比星,表柔比星,佐柔比星,米托蒽醌,MDR抑制剂(或维拉帕米),Ca2+ATP酶抑制剂(或毒胡萝卜素),组蛋白去乙酰酶抑制剂(伏立诺他,罗米地辛,帕比司他,丙戊酸,Mocetinostat(MGCD0103),Belinostat,PCI-24781,恩替诺特,SB939,Resminostat,Givinostat,AR-42,CUDC-101,萝卜硫素,曲古抑菌素A);塞来昔布,格列酮类,表没食子儿茶素没食子酸酯,双硫仑,SalinosporamideA;抗肾上腺药物,选自:氨鲁米特,米托坦,曲洛司坦,醋葡醛内酯,醛磷酰胺,氨基乙酰丙酸,安吖啶,阿拉伯糖苷,bestrabucil,比生群,edatraxate,defofamine,美可辛,地吖醌,依氟鸟氨酸(DFMO),elfomithine,依利醋铵,依托格鲁,硝酸镓,胞嘧啶,羟基脲,伊班膦酸盐,香菇多糖,氯尼达明,米托胍腙,米托蒽醌,莫呱达醇,二胺硝吖啶,喷司他丁,蛋氨氮芥,吡柔比星,鬼臼酸,2-乙肼,甲基苄肼;呱嗪二酮丙烷;根霉素;西佐;螺环锗;细格孢氮杂酸;三亚胺醌;三氯三乙胺;单端孢霉烯(包括T-2毒素,疣孢菌素A,杆孢菌素A和anguidine),聚氨酯,siRNA,反义药物;The D is a polyketide compound (Annonaline), bullatacin and bullatacinone; gemcitabine, cyclooxygenase and carfilomib, bortezomib, thalidomide, lenalidomide, pomalidomide, tosedostat, zybrestat, PLX4032, STA-9090, Stimuvax, allowctin-7, Xegeva, Provenge, Yervoy, prenylation inhibitors and lovastatin, dopaminergic neurotoxin (selected from staurosporine), actinomycin (contains Actinomycin D, dactinomycin), amanitin, bleomycin (including bleomycin A2, bleomycin B2, pelomycin), anthracyclines, including daunorubicin, Adriamycin (adriamycin), idarubicin, epirubicin, zorubicin, mitoxantrone, MDR inhibitors (or verapamil), Ca2+ATPase inhibitors (or thapsigargin), histone deacetylase inhibitors (vorinostat, romidepsin, panobinostat, valproic acid, Mocetinostat (MGCD0103), Belinostat, PCI-24781, entinostat, SB939, Resminostat, Givinostat, AR-42, CUDC-101, sulforaphane, trichostatin A); celecoxib, glitazones, epigallocatechin gallate, disulfiram, Salinosporamide A); anti Adrenal drug selected from the group consisting of: aminoglutethimide, mitotane, trolosteine, aceglucuronide, aldophosphamide, aminolevulinic acid, amsacrine, arabinoside, bestrabucil, bisantrene, edatraxate, defofamine, methacin, decquinone, eflornithine (DFMO), elfomithine, etricetium, etoglu, gallium nitrate, cytosine, hydroxyurea, ibandronate, lentinan, clonida Ming, Mitoguanidine Hydrazone, Mitoxantrone, Mopraditol, Diamine Niacridine, Pentostatin, Methionine, Pirarubicin, Podophyllic Acid, 2-Ethylhydrazine, Procarbazine ; piperazine diketopane; rhizoctidine; sizole; spirogermanium; A, bacitracin A and anguidine), polyurethane, siRNA, antisense drugs;
所述D为自身免疫疾病药物,选自环孢菌素,环孢菌素A,氨基己酸,硫唑嘌呤,溴隐亭,苯丁酸氮芥,氯喹,环磷酰胺,皮质类固醇(包括安西奈德,倍他米松,布地奈德,氢化可的松,氟尼缩松,丙酸氟替卡松,氟可龙达那唑,地塞米松,曲安奈德,二丙酸倍氯米松),DHEA,依那西普,羟基氯喹,英夫利昔单抗,美洛昔康,甲氨蝶呤,麦考酚酸酯,泼尼松,西罗莫司,他克莫司;The D is an autoimmune disease drug selected from cyclosporin, cyclosporine A, aminocaproic acid, azathioprine, bromocriptine, chlorambucil, chloroquine, cyclophosphamide, corticosteroids (including Amcinonide, betamethasone, budesonide, hydrocortisone, flunisolide, fluticasone propionate, flurcoron (danazol), dexamethasone, triamcinolone acetonide, beclomethasone dipropionate), DHEA , etanercept, hydroxychloroquine, infliximab, meloxicam, methotrexate, mycophenolate mofetil, prednisone, sirolimus, tacrolimus;
所述D为抗感染性疾病药物,包括氨基糖苷类:选自阿米卡星,阿司米星,庆大霉素(奈替米星,西索米星,异帕米星),潮霉素B,卡那霉素(阿米卡星,阿贝卡星,氨基脱氧卡那霉素,地贝卡星,妥布霉素),新霉素(framycetin,巴龙霉素,核糖霉素),奈替米星,壮观霉素,链霉素,妥布霉素,甲基姿苏霉素;The D is an anti-infective disease drug, including aminoglycosides: selected from amikacin, asemicin, gentamicin (netilmicin, sisomicin, isopamicin), hygromycin B, kanamycin (amikacin, arbekacin, aminodeoxykanamycin, dibekacin, tobramycin), neomycin (framycetin, paromomycin, ribomycin ), netilmicin, spectinomycin, streptomycin, tobramycin, methylzithramycin;
所述D为抗感染性疾病药物,包括酰胺醇类:选自迭氮氯霉素,氯霉素,氟苯尼考,甲砜霉素;The D is an anti-infective disease drug, including amide alcohols: selected from azidochloramphenicol, chloramphenicol, florfenicol, thiamphenicol;
所述D为抗感染性疾病药物,包括安沙霉素:选自格尔德霉素或除莠霉素;The D is an anti-infective disease drug, including ansamycin: selected from geldanamycin or herbimycin;
所述D为抗感染性疾病药物,包括碳青霉烯类:选自比阿培南,多利培南,厄他培南,亚胺培南/西司他丁,美罗培南,帕尼培南;The D is an anti-infective disease drug, including carbapenems: selected from biapenem, doripenem, ertapenem, imipenem/cilastatin, meropenem, panipenem ;
所述D为抗感染性疾病药物,包括头孢烯:选自碳头孢烯(洛拉卡比),头孢乙腈,氯氨苄青霉素,头孢拉定,头孢羟氨,头孢洛宁,头孢噻啶,头孢噻吩或头孢金素,头孢氨苄,头孢来星,头孢孟多,头孢匹林,羟胺唑头孢菌素,氟唑头孢菌素,孢西酮,唑啉头孢菌素,头孢拉宗,头孢卡品,头孢达肟,头孢吡,头孢克肟,头孢西丁,头孢罗齐,头孢甲氧环烯胺,头孢替唑,头孢呋辛,头孢克肟,头孢地尼,头孢托仑,头孢吡,头孢他美,头孢甲肟,头孢地嗪,头孢尼西,头孢呱酮,头孢雷特,头孢噻肟,噻乙胺唑头孢菌素,头孢唑兰,头孢氨苄,头孢咪唑,头孢匹胺,头孢匹罗,头孢泊肟,头孢罗齐,头孢喹诺,头孢磺啶,头孢他啶,头孢特仑,头孢布腾,头孢噻林,头孢唑肟,头孢吡普,头孢曲松,头孢呋辛,头孢唑南,头霉素(头孢西丁,头孢替坦,头孢氰唑),氧(碳)头孢烯(氟氧头孢,拉氧头孢);The D is an anti-infective disease drug, including cephems: selected from carbacephem (loracarb), cefuroxime, ampicillin, cephradine, cefadroxidine, cefuroxime, cefotaxime, cephalothin or Cephalosporin, cephalexin, cephalosporin, cefamandole, cefapirin, cephalosporin oxamazole, cephalosporin fluxazone, fenidone, cephalosporin oxazoline, cefbuperazone, cefcapine, cephalosporin Daxime, cefepime, cefixime, cefoxitin, cefprozil, cefmethoxam, ceftezole, cefuroxime, cefixime, cefdinir, cefditoren, cefepime, ceftazidime The United States, cefmenoxime, cefodizime, cefanixime, cefoperazone, cefreide, cefotaxime, cefazolin, cefazolan, cephalexin, cefamizole, cefpiramide, cefpirin Luo, cefpodoxime, cefprozil, cefquinol, cefsulodin, ceftazidime, cefditoren, ceftibuten, cefotaxime, ceftizoxime, ceftazidime, ceftriaxone, cefuroxime, ceftizole South, cephamycin (cefoxitin, cefotetan, ceftriaxone), oxygen (carbon) cephem (fluoxyceph, latamoxef);
所述D为抗感染性疾病药物,包括糖肽:选自博来霉素,万古霉素(奥利万星,特拉万星),替考拉宁(达巴万星),雷莫拉宁;The D is an anti-infective disease drug, including glycopeptide: selected from bleomycin, vancomycin (oritavancin, telavancin), teicoplanin (dalbavancin), remora rather;
所述D为抗感染性疾病药物,包括甘氨酰环素:如替加环素;The D is an anti-infective disease drug, including glycylcycline: such as tigecycline;
所述D为抗感染性疾病药物,包括β-内酰胺酶抑制剂:选自青霉烷(舒巴坦,他唑巴坦),氧青霉烷(克拉维酸);The D is an anti-infective disease drug, including a β-lactamase inhibitor: selected from penicillane (sulbactam, tazobactam), oxypenicillane (clavulanic acid);
所述D为抗感染性疾病药物,包括林可酰胺:选自克林霉素或林可霉素;The D is an anti-infective disease drug, including lincosamide: selected from clindamycin or lincomycin;
所述D为抗感染性疾病药物,包括脂肽:选自达托霉素,A54145,钙依赖性抗生素(CDA);The D is an anti-infective disease drug, including lipopeptide: selected from daptomycin, A54145, calcium-dependent antibiotics (CDA);
所述D为抗感染性疾病药物,包括大环内酯类:选自阿奇霉素,克霉素,克拉霉素,地红霉素,红霉素,氟雷霉素,交沙霉素,酮内酯(泰利霉素,塞红霉素),麦迪霉素,米卡霉素,竹桃霉素,利福霉素(异烟肼、利福平,利福布丁,利福喷汀),罗匹霉素,罗红霉素,大观霉素,螺旋霉素,他克莫司(FK506),醋竹桃霉素,泰利霉素;The D is an anti-infective disease drug, including macrolides: selected from azithromycin, clindamycin, clarithromycin, dirithromycin, erythromycin, fluremycin, josamycin, ketone Esters (telithromycin, erythromycin), midecamycin, mikamycin, oleandomycin, rifamycins (isoniazid, rifampicin, rifabutin, rifapentine), Piramycin, Roxithromycin, Spectinomycin, Spiramycin, Tacrolimus (FK506), Troleandomycin, Telithromycin;
所述D为抗感染性疾病药物,包括单环胺:选自氨曲南或替吉莫南;The D is an anti-infective disease drug, including monocyclic amine: selected from aztreonam or tigemonan;
所述D为抗感染性疾病药物,包括恶唑烷酮类:如利奈唑胺;The D is an anti-infective disease drug, including oxazolidinones: such as linezolid;
所述D为抗感染性疾病药物,包括青霉素类:选自阿莫西林,氨苄青霉素(匹氨西林,海洛西林,巴氨西林,氨苄青霉素,阿霉素),阿替代西林,阿洛西林,苄青霉素,苄星青霉素苄青霉素,苄星青霉素苯氧甲基青霉素,克洛西林,普鲁卡因青霉素(美替西林),美洛西林,甲氧西林,萘夫西林,苯唑西林,醋甲西林,青霉素,非奈西林,苯氧基甲基青霉素,呱拉西林,氨苄西林,磺苯西林,替莫西林,替卡西林;The D is an anti-infective disease drug, including penicillins: selected from amoxicillin, ampicillin (piamicillin, heroicillin, bamicillin, ampicillin, doxorubicin), amoxicillin, azlocillin , benzathine penicillin, benzathine penicillin benzathine penicillin, benzathine penicillin phenoxymethyl penicillin, clotillin, procaine penicillin (methenicillin), mezlocillin, methicillin, nafcillin, oxacillin, Acemethicillin, penicillin, phenecillin, phenoxymethylpenicillin, gualacillin, ampicillin, sulfbenzillin, temoxicillin, ticarcillin;
所述D为抗感染性疾病药物,包括多肽:选自杆菌肽,粘菌素,多粘菌素B;The D is an anti-infectious disease drug, including a polypeptide: selected from bacitracin, colistin, and polymyxin B;
所述D为抗感染性疾病药物,包括喹诺酮类:选自阿拉曲沙星,巴洛沙星,环丙沙星,克林沙,达氟沙星,二氟沙星,依诺沙星,恩诺沙星,加雷沙星,加替沙星,吉米沙星,格帕沙星,卡诺曲伐沙星,左氧氟沙星,洛美沙星,麻保沙星,莫西沙星,那氟沙星,诺氟沙星,奥比沙星,氧氟沙星,培氟沙星,曲伐沙星,格帕沙星,西他沙星,司帕沙星,替马沙星,托沙星,曲伐沙星;The D is an anti-infective disease drug, including quinolones: selected from alatrafloxacin, baloxacin, ciprofloxacin, Kelinsha, dafloxacin, difloxacin, enoxacin, Enrofloxacin, Garafloxacin, Gatifloxacin, Gemifloxacin, Gepafloxacin, Canotrovafloxacin, Levofloxacin, Lomefloxacin, Mabofloxacin, Moxifloxacin, Nafloxacin , Norfloxacin, Orbifloxacin, Ofloxacin, Pefloxacin, Trovafloxacin, Gepafloxacin, Sitafloxacin, Sparfloxacin, Temafloxacin, Tofloxacin, Qu Valfloxacin;
所述D为抗感染性疾病药物,包括链阳性菌素:选自普那霉素,奎奴普丁/达福普汀;The D is an anti-infective disease drug, including streptavidin: selected from prunamycin, quinupristin/dalfopristin;
所述D为抗感染性疾病药物,包括磺胺类:选自氨芐磺胺,偶氮磺胺,磺胺嘧啶,磺胺甲异唑、磺胺酰亚胺、磺胺吡啶、磺胺异恶唑、甲氧苄啶、磺胺甲恶唑(复方磺胺甲恶唑);The D is an anti-infective disease drug, including sulfonamides: selected from ambenzylsulfonamide, azosulfonamide, sulfadiazine, sulfamethoxazole, sulfimide, sulfapyridine, sulfisoxazole, trimethoprim, sulfamethoxazole Methoxazole (compound sulfamethoxazole);
所述D为抗感染性疾病药物,包括类固醇抗菌药物:如夫西地酸;The D is an anti-infective disease drug, including a steroid antibacterial drug: such as fusidic acid;
所述D为抗感染性疾病药物,包括四环素类:选自强力霉素,金霉素,氯米西环素,地美环素,雷莫昔林,美西环素,美他环素,米诺环素,土霉素,潘美环素,吡咯烷甲基四环素,四环素,甘氨酰环素(如替加环素);The D is an anti-infective disease drug, including tetracyclines: selected from doxycycline, aureomycin, clomixicycline, demecycline, ramoxiline, mexicycline, metacycline, Minocycline, oxytetracycline, pamecycline, pyrrolidinylmethyltetracycline, tetracycline, glycylcycline (such as tigecycline);
所述D为抗感染性疾病药物,包括其他类型的抗生素:番荔枝素,胂凡纳明,细菌萜醇抑制剂(杆菌肽),DANAL/AR抑制剂(环丝氨酸),dictyostatin,圆皮海绵内酯,软珊瑚醇,埃博霉素,乙胺丁醇,依托泊苷,法罗培南,夫西地酸,呋喃唑酮,异烟肼,laulimalide,甲硝唑,莫匹罗星,NAM合成抑制剂(例如磷霉素),呋喃妥因,紫杉醇,普兰西霉素,吡嗪酰胺,奎奴普丁/达福普汀,利福平,他唑巴坦替硝唑,乌菊花素;Said D is an anti-infective disease drug, including other types of antibiotics: anemone, arsenadine, bacterial terpene alcohol inhibitor (bacitracin), DANAL/AR inhibitor (cycloserine), dictyostatin, and disc sponge Lactone, soft coralline, epothilone, ethambutol, etoposide, faropenem, fusidic acid, furazolidone, isoniazid, laulimalide, metronidazole, mupirocin, NAM synthesis inhibitor (e.g. fosfomycin), nitrofurantoin, paclitaxel, prancimycin, pyrazinamide, quinupristin/dalfopristin, rifampicin, tazobactantinidazole, sclerosin;
所述D为抗病毒药物,包括侵入/融合抑制剂:选自阿帕韦洛,马拉韦罗,vicriviroc,gp41(恩夫韦肽),PRO140或CD4(艾巴利珠单抗);The D is an antiviral drug, including an invasion/fusion inhibitor: selected from apavero, maraviroc, vicriviroc, gp41 (enfuvirtide), PRO140 or CD4 (ibalizumab);
所述D为抗病毒药物,包括整合酶抑制剂:选自雷特格韦,elvite-gravir或globoidnanA;The D is an antiviral drug, including an integrase inhibitor: selected from Raltegravir, elvite-gravir or globoidnanA;
所述D为所述D为抗病毒药物,包括成熟抑制剂:选自bevirimat或vivecon;The D is that the D is an antiviral drug, including a maturation inhibitor: selected from bevirimat or vivecon;
所述D为抗病毒药物,包括神经氨酸酶抑制剂:选自奥司他韦,扎那米韦或帕拉米韦;The D is an antiviral drug, including a neuraminidase inhibitor: selected from oseltamivir, zanamivir or peramivir;
所述D为抗病毒药物,包括核苷和核苷酸:选自阿巴卡韦,阿昔单韦,阿德福韦,阿莫西韦,阿昔单抗,溴夫定,西多福韦,克拉夫定,地塞米松,去羟肌苷(ddI),elvucitabine,恩曲他滨(FTC),恩替卡韦,泛昔洛韦,氟拉西林(5-FU),3’-氟取代的2’,3’-脱氧核苷类似物,包括3’-氟-2’,3’-双脱氧胸苷(FLT)和3’-氟-2’,3’-双脱氧鸟苷(FLG),福米韦生,9-鸟嘌呤,碘苷,拉米夫定(3TC),1-核苷(包括β-1-胸苷和β-1-2'-脱氧胞苷),喷昔洛韦,racivir,利巴韦林,迪替丁,司他夫定(d4T),塔利巴韦林(viramidine),替比夫定,替诺福韦,三氟尿苷伐昔洛韦,缬更昔洛韦,扎西他滨(ddC)或齐多夫定(AZT);The D is an antiviral drug, including nucleosides and nucleotides: selected from abacavir, aciximavir, adefovir, amoxivir, abciximab, brovudine, cidofo vir, clavudine, dexamethasone, didanosine (ddI), elvucitabine, emtricitabine (FTC), entecavir, famciclovir, fluracillin (5-FU), 3'-fluoro substituted 2', 3'-Deoxynucleoside analogs, including 3'-fluoro-2',3'-dideoxythymidine (FLT) and 3'-fluoro-2',3'-dideoxyguanosine (FLG), Fumi Weisheng, 9-guanine, iodine, lamivudine (3TC), 1-nucleosides (including β-1-thymidine and β-1-2'-deoxycytidine), penciclovir, racivir , ribavirin, detidine, stavudine (d4T), talibavirin (viramidine), telbivudine, tenofovir, trifluridine valacyclovir, valganciclo Wei, zalcitabine (ddC) or zidovudine (AZT);
所述D为抗病毒药物,包括非核苷类:选自金刚烷胺,阿替吡啶,卡普韦林,二芳基嘧啶(依曲韦林,rilpivirine),地拉夫定,二十二烷醇,乙米韦林,依法韦仑,膦甲酸(磷酰基甲酸),咪喹莫特,聚乙二醇干扰素,洛韦胺,洛德腺苷,甲吲噻腙,奈韦拉平,NOV-205,长效干扰素α,鬼臼毒素,利福平,金刚乙胺,瑞喹莫德(R-848)或醋胺金刚烷;The D is an antiviral drug, including non-nucleosides: selected from amantadine, atipyridine, capravirine, diarylpyrimidine (etravirine, rilpivirine), delavudine, docosanol , emivirine, efavirenz, foscarnet (phosphoroformic acid), imiquimod, peginterferon, loviramide, lord adenosine, inthizone, nevirapine, NOV-205, peginterferon alfa, podophyllotoxin, rifampicin, rimantadine, resiquimod (R-848) or amantadine;
所述D为抗病毒药物,包括蛋白酶抑制剂:选自安普那韦,阿扎那韦,boceprevir,darunavir,福萨那韦,印地那韦,洛匹那韦,奈非那韦,普来可那立,利托那韦,沙奎那韦,telaprevir(VX-950)或替拉那韦;The D is an antiviral drug, including protease inhibitors: selected from amprenavir, atazanavir, boceprevir, darunavir, fosanavir, indinavir, lopinavir, nelfinavir, Leconali, ritonavir, saquinavir, telaprevir (VX-950) or tipranavir;
所述D为抗病毒药物,包括其它类型的抗病毒药物:如抗体酶,阿比朵尔,calanolidea,ceragenin,氰维林-n,二芳基嘧啶,表没食子儿茶素没食子酸酯(EGCG),膦甲酸,格里菲辛,taribavirin(viramidine),羟基脲,KP-1461,米替福新,普来可那立,混成抑制剂,利巴韦林或seliciclib;The D is an antiviral drug, including other types of antiviral drugs: such as abzyme, arbidol, calanolidea, ceragenin, cyanverine-n, diarylpyrimidine, epigallocatechin gallate (EGCG ), foscarnet, griffithine, taribavirin (viramidine), hydroxyurea, KP-1461, miltefosine, pleconaril, mixed inhibitors, ribavirin or seliciclib;
所述D为放射性同位素,可以选自(放射性核素)3H、11C、14C、18F、32P、35S、64Cu、68Ga、86Y、99Tc、111In、123I、124I、125I、131I、133Xe、177Lu、211At、或213Bi。The D is a radioisotope, which can be selected from (radionuclide) 3H, 11C, 14C, 18F, 32P, 35S, 64Cu, 68Ga, 86Y, 99Tc, 111In, 123I, 124I, 125I, 131I, 133Xe, 177Lu, 211At , or 213Bi.
所述D为发色团分子,能够吸收紫外线、荧光、红外光、近红外光、可见光;黄色素、红细胞、虹彩色素、白细胞、黑色素和蓝绿色素的一类或一个亚类、荧光分子吸收光后再发光的荧光化学物质。视觉光转导分子、荧光团分子、发光分子、萤光素化合物。非蛋白质有机荧光团,选自:氧杂蒽衍生物(包括荧光素、罗丹明、俄勒冈绿、伊红和德克萨斯红);花青衍生物(包括花青、吲哚羰花青、氧杂花青、硫代花青和部花青);方酸衍生物和环取代的方酸、包括Seta、SeTau和Square染料;萘衍生物(包括丹酰和氟硅酸钠衍生物);香豆素衍生物;恶二唑衍生物(包括吡啶基恶唑、硝基苯并恶唑和苯并恶二唑);蒽衍生物(包括蒽醌类、包括DRAQ5、DRAQ7和CyTRAK橙);芘衍生物(包括级联蓝等);恶嗪衍生物(包括尼罗红、尼罗蓝、甲酚紫、恶嗪170等);吖啶衍生物(包括黄醇黄素、吖啶橙、吖啶黄等);芳基甲胺衍生物(包括金胺、结晶紫、孔雀石绿)和四吡咯衍生物(卟吩、酞菁、胆红素)。The D is a chromophore molecule capable of absorbing ultraviolet rays, fluorescence, infrared light, near-infrared light, and visible light; yellow pigments, red blood cells, iridescent pigments, white blood cells, melanin, and a class or a subclass of blue-green pigments, fluorescent molecules absorb Fluorescent chemicals that glow after light. Visual phototransduction molecules, fluorophore molecules, luminescent molecules, luciferin compounds. Non-proteinaceous organic fluorophores selected from the group consisting of: xanthene derivatives (including fluorescein, rhodamine, Oregon green, eosin and Texas red); cyanine derivatives (including cyanine, indocarbocyanine, oxacyanines, thiocyanines, and merocyanines); squaraine derivatives and ring-substituted squaraines, including Seta, SeTau, and Square dyes; naphthalene derivatives (including dansyl and sodium fluorosilicate derivatives); Coumarin derivatives; oxadiazole derivatives (including pyridyloxazole, nitrobenzoxazole, and benzoxadiazole); anthracene derivatives (including anthraquinones, including DRAQ5, DRAQ7, and CyTRAK orange); Pyrene derivatives (including cascade blue, etc.); oxazine derivatives (including Nile red, Nile blue, cresyl violet, oxazine 170, etc.); acridine derivatives (including flavin, acridine orange, acridine yellow, etc.); arylmethylamine derivatives (including auramine, crystal violet, malachite green) and tetrapyrrole derivatives (porphine, phthalocyanine, bilirubin).
所述D为生色分子,选自以下荧光化合物的任何类似物和衍生物:CF染料、DRAQ和CyTRAK探针、BODIPY、AlexaFluor、DyLightFluor、Atto和Tracy、FluoProbes、AbberiorDyes、DY和MegaStokesDyes、SulfoCy染料、HiLyteFluor、Seta、SeTau和SquareDyes、Quasar和CalFluordyes、SureLightDyes(APC、RPEPerCP、Phycobilisomes)、APC、APCXL、RPE、BPE、别藻蓝蛋白(APC)、氨基胭脂蛋白、APC-Cy7偶联物、BODIPY-FL、CascadeBlue、Cy2、Cy3、Cy3.5、Cy3B、Cy5、Cy5.5、Cy7、F荧光素、FluorX、羟基香豆素、丽丝胺罗丹明B、萤光黄、Me-甲氧基香豆素、NBD、PacificBlue、PacificOrange、PE-Cy5偶联物、PE-Cy7偶联物、PerCP、R-藻红蛋白(PE)、Red613、Seta-555-叠氮化物、Seta-555-DBCO、Seta-555-NHS、Seta-580-NHS、Seta-680-NHS、Seta-780-NHS、Seta-APC-780、Seta-PerCP-680、Seta-R-PE-670、SeTau-380-NHS、SeTau-405-马来酰亚胺、SeTau-405-NHS、SeTau-425-NHS、SeTau-647-NHS、TexasRed、TRITC、TruRed、X-Rhodamine、7-AAD(7-氨基放线菌素D、CG-选择性的)、吖啶橙、色霉素A3、CyTRAK橙(redexcitationdark)、DAPI、DRAQ5、DRAQ7、溴化乙锭、Hoechst33258、Hoechst33342、LDS751、光辉霉素、碘化丙啶(PI)、SYTOX蓝、SYTOX绿、SYTOX橙、噻唑橙、TO-PRO:菁染料单体、TOTO-1、TO-PRO-1、TOTO-3、TO-PRO-3、YOSeta-1、YOYO-1;荧光化合物:包括DCFH(2'、7'-二氯二氢荧光素、氧化形式)、DHR(二氢罗丹明123、氧化形式、光催化氧化)、Fluo-3(AM酯,pH>6)、Fluo-4(AM酯,pH7.2)、Indo-1(AM酯,低/高钙(Ca2+))、SNARF(pH6/9)、别藻蓝蛋白(APC)、AmCyan1(四聚体、Clontech)、AsRed2(四聚体、Clontech)、AzamiGreen(单体)、Azurite、B-藻红蛋白(BPE)、Cerulean、CyPet、DsRed单体(Clontech)、DsRed2("RFP")、EBFP、EBFP2、ECFP、EGFP(弱二聚体)、Emerald(弱二聚体)、EYFP(弱二聚体)、GFP(S65A突变)、GFP(S65C突变)、GFP(S65L突变)、GFP(S65T突变)、GFP(Y66F突变)、GFP(Y66H突变)、GFP(Y66W突变)、GFPuv、HcRed1、J-Red、Katusha、KusabiraOrange(单体、MBL)、mCFP、mCherry、mCitrine、MidoriishiCyan(二聚体、MBL)、mKate(TagFP635、单体)、mKeima-Red(单体)、mKO、mOrange、mPlum、mRaspberry、mRFP1(单体)、mStrawberry、mTFP1、mTurquoise2、P3(藻胆体复合物)、多甲藻黄素-叶绿素-蛋白质复合物(PerCP)、R-phycoerythrin(RPE)、T-Sapphire、TagCFP(二聚体)、TagGFP(二聚体)、TagRFP(二聚体)、TagYFP(二聚体)、tdTomato(tandem二聚体)、Topaz、TurboFP602(二聚体)、TurboFP635(二聚体)、TurboGFP(二聚体)、TurboRFP(二聚体)、TurboYFP(二聚体)、Venus、WildTypeGFP、YPet、ZsGreen1(四聚体)、ZsYellow1(四聚体)及其衍生物。Said D is a chromogenic molecule selected from any analog and derivative of the following fluorescent compounds: CF dyes, DRAQ and CyTRAK probes, BODIPY, AlexaFluor, DyLightFluor, Atto and Tracy, FluoProbes, AbberiorDyes, DY and MegaStokesDyes, SulfoCy dyes , HiLyteFluor, Seta, SeTau and SquareDyes, Quasar and CalFluordyes, SureLightDyes (APC, RPEPerCP, Phycobilisomes), APC, APCXL, RPE, BPE, Allophycocyanin (APC), Aminonopain, APC-Cy7 Conjugate, BODIPY -FL, CascadeBlue, Cy2, Cy3, Cy3.5, Cy3B, Cy5, Cy5.5, Cy7, F Fluorescein, FluorX, Hydroxycoumarin, Lissamine Rhodamine B, Fluorescent Yellow, Me-Methoxy Coumarin, NBD, PacificBlue, PacificOrange, PE-Cy5 Conjugate, PE-Cy7 Conjugate, PerCP, R-Phycoerythrin (PE), Red613, Seta-555-Azide, Seta-555-DBCO , Seta-555-NHS, Seta-580-NHS, Seta-680-NHS, Seta-780-NHS, Seta-APC-780, Seta-PerCP-680, Seta-R-PE-670, Seta-380-NHS , SeTau-405-maleimide, SeTau-405-NHS, SeTau-425-NHS, SeTau-647-NHS, TexasRed, TRITC, TruRed, X-Rhodamine, 7-AAD (7-aminoactinomycin D, CG-selective), acridine orange, chromomycin A3, CyTRAK orange (reexcitationdark), DAPI, DRAQ5, DRAQ7, ethidium bromide, Hoechst33258, Hoechst33342, LDS751, mithromycin, propidium iodide ( PI), SYTOX blue, SYTOX green, SYTOX orange, thiazole orange, TO-PRO: cyanine dye monomer, TOTO-1, TO-PRO-1, TOTO-3, TO-PRO-3, YOSeta-1, YOYO- 1. Fluorescent compounds: including DCFH (2', 7'-dichlorodihydrofluorescein, oxidized form), DHR (dihydrorhodamine 123, oxidized form, photocatalytic oxidation), Fluo-3 (AM ester, pH> 6), Fluo-4 (AM ester, pH7.2), Indo-1 ( AM Ester, Low/High Calcium (Ca2+), SNARF (pH6/9), Allophycocyanin (APC), AmCyan1 (Tetramer, Clontech), AsRed2 (Tetramer, Clontech), AzamiGreen (Monomer) , Azurite, B-phycoerythrin (BPE), Cerulean, CyPet, DsRed monomer (Clontech), DsRed2 ("RFP"), EBFP, EBFP2, ECFP, EGFP (weak dimer), Emerald (weak dimer ), EYFP (weak dimer), GFP (S65A mutation), GFP (S65C mutation), GFP (S65L mutation), GFP (S65T mutation), GFP (Y66F mutation), GFP (Y66H mutation), GFP (Y66W mutation ), GFPuv, HcRed1, J-Red, Katusha, KusabiraOrange (monomer, MBL), mCFP, mCherry, mCitrine, MidoriishiCyan (dimer, MBL), mKate (TagFP635, monomer), mKeima-Red (monomer) , mKO, mOrange, mPlum, mRaspberry, mRFP1 (monomer), mStrawberry, mTFP1, mTurquoise2, P3 (phycobilisome complex), peridinoxanthin-chlorophyll-protein complex (PerCP), R-phycoerythrin (RPE ), T-Sapphire, TagCFP (dimer), TagGFP (dimer), TagRFP (dimer), TagYFP (dimer), tdTomato (tandem dimer), Topaz, TurboFP602 (dimer) , TurboFP635 (dimer), TurboGFP (dimer), TurboRFP (dimer), TurboYFP (dimer), Venus, WildTypeGFP, YPet, ZsGreen1 (tetramer), ZsYellow1 (tetramer) and derivative.
所述D为细胞结合配体或受体激动剂,可以选自:叶酸衍生物、谷氨酸尿素衍生物、生长抑素及其类似物(选自奥曲肽(Sandostatin)和兰瑞肽(Somatuline))、芳香磺酰胺、Aromaticsulfonamides、垂体腺苷酸环化酶激活肽(PACAP)(PAC1)、血管活性肠肽(VIP/PACAP)(VPAC1,VPAC2)、α-黑素细胞刺激激素(α-MSH)、胆囊收缩素(CCK)/胃泌素受体激动剂、蛙皮素(选自Pyr-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met-NH2)/胃泌素释放肽(GRP)、神经降压素受体及其受体亚型(NTR1,NTR2,NTR3)、物质P(NK1受体)配体、神经肽Y(Y1–Y6)、归巢肽包括RGD(Arg-Gly-Asp),NGR(Asn-Gly-Arg),二聚和多聚环状RGD肽(选自cRGDfV),TAASGVRSMH和LTLRWVGLMS(硫酸软骨素蛋白多糖NG2受体)和F3肽、细胞穿透肽(CPPs)、肽激素,选自促黄体激素释放激素(LHRH)激动剂和拮抗剂,和促性腺激素释放激素(GnRH)激动剂,通过靶向卵泡刺激素(FSH)和黄体生成素(LH)起作用,以及睾酮产物,选自布舍瑞林(Pyr-His-Trp-Ser-Tyr-D-Ser(OtBu)-Leu-Arg-Pro-NHEt)、戈那瑞林(Pyr-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-gly-NH2)、戈舍瑞林(Pyr-His-Trp-Ser-Tyr-D-Ser(OtBu)-Leu-Arg-Pro-Azgly-NH2)、组氨瑞林(Pyr-His-Trp-Ser-Tyr-D-His(N-苯基)-Leu-Arg-Pro-NHEt)、醋酸亮丙瑞林(Pyr-His-Trp-Ser-Tyr-D-Leu-Leu-Arg-Pro-NHEt)、那法瑞林(Pyr-His-Trp-Ser-Tyr-2Nal-Leu-Arg-Pro-Gly-NH2)、曲普瑞林(Pyr-His-Trp-Ser-Tyr-D-Trp-Leu-Arg-Pro-Gly-NH2)、德舍瑞林、阿巴瑞克(Ac-D-2Nal-D-4-氯-D-3-(3-吡啶基)Ala-Ser-(N-Me)Tyr-D-Asn-Leu-异丙基-Lys-Pro-D-Ala-NH2)、西曲瑞克(Ac-D-2Nal-D-4-chloro-Phe-D-3-(3-pyridyl)Ala-Ser-Tyr-D-Cit-Leu-Arg-Pro-D-Ala-NH2)、地加瑞克(Ac-D-2Nal-D-4-chloroPhe-D-3-(3-吡啶基)Ala-Ser-4-氨基Phe(L-hydroorotyl)-D-4-氨基Phe(氨基甲酰基)-Leu-异丙基Lys-Pro-D-Ala-NH2)和加尼瑞克(Ac-D-2Nal-D-4-chloroPhe-D-3-(3-吡啶基)Ala-Ser-Tyr-D-(N9,N10-二乙基)-homoArg-Leu-(N9,N10-二乙基)-homoArg-Pro-D-Ala-NH2),模式识别受体(PRRs),选自Toll受体(TLRs)、C型凝集素和Nodlike受体(NLRs)、降钙素受体激动剂、整合素受体及其受体亚型(选自αVβ1、αVβ3、αVβ5、αVβ6、α6β4、α7β1、αLβ2、αIIbβ3)激动剂(选自GRGDSPK,环(RGDfV)(L1)及其衍生物[环(-N(Me)R-GDfV)、环(R-Sar-DfV)、环(RG-N(Me)D-fV)、环(RGD-N(Me)f-V)、环(RGDf-N(Me)V-)(Cilengitide)]、纳米抗体(VHH的衍生物(骆驼Ig))、域抗体(dAb、VH或VL域的衍生物)、双特异性T细胞Engager(BiTE、双特异性双抗体)、双亲和重新定位(DART,双特异性双抗体)、四价串联抗体(TandAb,二聚双特异双抗体)、Anticalin(Lipocalins的衍生物)、Adnectins(10thFN3(Fibronectin))、设计的锚蛋白重复蛋白(DARPins)、Avimers、EGF受体和VEGF受体激动剂。The D is a cell-binding ligand or receptor agonist, which can be selected from: folic acid derivatives, glutamic acid urea derivatives, somatostatin and analogs thereof (selected from octreotide (Sandostatin) and lanreotide (Somatuline) ), aromatic sulfonamides, Aromaticsulfonamides, pituitary adenylate cyclase activating peptide (PACAP) (PAC1), vasoactive intestinal peptide (VIP/PACAP) (VPAC1, VPAC2), α-melanocyte stimulating hormone (α-MSH ), cholecystokinin (CCK)/gastrin receptor agonist, bombesin (selected from Pyr-Gln-Arg-Leu-Gly-Asn-Gln-Trp-Ala-Val-Gly-His-Leu-Met -NH2)/gastrin-releasing peptide (GRP), neurotensin receptor and its receptor subtypes (NTR1, NTR2, NTR3), substance P (NK1 receptor) ligand, neuropeptide Y (Y1–Y6 ), homing peptides including RGD (Arg-Gly-Asp), NGR (Asn-Gly-Arg), dimeric and polycyclic RGD peptides (selected from cRGDfV), TAASGVRSMH and LTLRWVGLMS (chondroitin sulfate proteoglycan NG2 receptor body) and F3 peptides, cell-penetrating peptides (CPPs), peptide hormones selected from luteinizing hormone-releasing hormone (LHRH) agonists and antagonists, and gonadotropin-releasing hormone (GnRH) agonists, by targeting follicle stimulation Hormone (FSH) and Luteinizing Hormone (LH) act, and testosterone product, selected from Buserelin (Pyr-His-Trp-Ser-Tyr-D-Ser(OtBu)-Leu-Arg-Pro-NHEt) , Gonadorelin (Pyr-His-Trp-Ser-Tyr-Gly-Leu-Arg-Pro-gly-NH2), Goserelin (Pyr-His-Trp-Ser-Tyr-D-Ser(OtBu) -Leu-Arg-Pro-Azgly-NH2), Histrelin (Pyr-His-Trp-Ser-Tyr-D-His(N-Phenyl)-Leu-Arg-Pro-NHEt), Leuprolide Acetate Lin (Pyr-His-Trp-Ser-Tyr-D-Leu-Leu-Arg-Pro-NHEt), Nafarelin (Pyr-His-Trp-Ser-Tyr-2Nal-Leu-Arg-Pro-Gly- NH2), triptorelin (Pyr-His-Trp-Ser-Tyr-D-Trp-Leu-Arg-Pro-Gly-NH2), deserelin, abarelix (Ac-D-2Nal-D -4-Chloro-D-3-(3-pyridyl)Ala-Ser-(N-Me)Tyr-D-Asn-Leu-Isopropyl-Lys-Pro-D-Ala-NH2) , Cetrorelix (Ac-D-2Nal-D-4-chloro-Phe-D-3-(3-pyridyl)Ala-Ser-Tyr-D-Cit-Leu-Arg-Pro-D-Ala-NH2 ), Degarelix (Ac-D-2Nal-D-4-chloroPhe-D-3-(3-pyridyl)Ala-Ser-4-aminoPhe(L-hydroorotyl)-D-4-aminoPhe( Carbamoyl)-Leu-IsopropylLys-Pro-D-Ala-NH2) and Ganyrelix (Ac-D-2Nal-D-4-chloroPhe-D-3-(3-pyridyl)Ala- Ser-Tyr-D-(N9,N10-diethyl)-homoArg-Leu-(N9,N10-diethyl)-homoArg-Pro-D-Ala-NH2), pattern recognition receptors (PRRs), selected From Toll receptors (TLRs), C-type lectins and Nodlike receptors (NLRs), calcitonin receptor agonists, integrin receptors and their receptor subtypes (selected from αVβ1, αVβ3, αVβ5, αVβ6, α6β4 , α7β1, αLβ2, αIIbβ3) agonist (selected from GRGDSPK, cyclic (RGDfV) (L1) and its derivatives [cyclic (-N(Me)R-GDfV), cyclic (R-Sar-DfV), cyclic (RG -N(Me)D-fV), Cyclo(RGD-N(Me)f-V), Cyclo(RGDf-N(Me)V-)(Cilengitide)], Nanobodies (Derivatives of VHH (Camel Ig)), Domain antibody (dAb, derivative of VH or VL domain), bispecific T cell Engager (BiTE, bispecific diabody), double affinity retargeting (DART, bispecific diabody), tetravalent tandem antibody (TandAb , dimeric bispecific diabodies), Anticalins (derivatives of Lipocalins), Adnectins (10thFN3 (Fibronectin)), Designer Ankyrin Repeat Proteins (DARPins), Avimers, EGF receptor and VEGF receptor agonists.
所述D为上述药物药学上可接受的盐,酸或其衍生物,水合物或水合盐、或晶体结构或任何上述药物的旋光异构体,外消旋物,非对映异构体或对映异构体。Said D is a pharmaceutically acceptable salt of the above drug, acid or its derivative, hydrate or hydrated salt, or crystal structure or any optical isomer, racemate, diastereoisomer or Enantiomers.
优选的,所述的D选自:氨基酸、蛋白质、酶、核苷、糖类、有机酸、甙类、黄酮类、醌类、萜类、苯丙素酚类、甾体及其甙类和生物碱中的一种。Preferably, said D is selected from the group consisting of: amino acids, proteins, enzymes, nucleosides, sugars, organic acids, glycosides, flavonoids, quinones, terpenes, phenylpropanol phenols, steroids and their glycosides and One of the alkaloids.
更优选的,所述D为用于杀灭肿瘤细胞的药物;More preferably, the D is a drug for killing tumor cells;
和/或,所述的D优选自:鹅膏菌素、奥瑞他汀、加利车霉素、喜树碱、喜树碱衍生物和代谢物(如:SN-38及其衍生物、10-羟基喜树碱、9-氨基喜树碱、9-硝基喜树碱),依喜替康、Dxd、隐藻霉素、道诺霉素、多拉司他丁、多柔比星、多卡霉素、埃坡霉素、埃斯帕霉素、格尔德霉素、美登素及其衍生物、甲氨蝶呤、单甲基奥瑞他汀E(“MMAE”),单甲基auristatinF(“MMAF”)、吡咯并苯二氮卓、根瘤菌素、SG2285、微管溶素、长春地辛、类毒素、伊立替康、拓扑替康、贝洛替康、依沙替康、卢托替康、二氟替康、吉尼替康、卡尼替康、阿霉素、表柔比星、吗啉代阿霉素、氰基吗啉代-阿霉素、2-吡咯啉基阿霉素、、MMAD、PBD及其衍生物、TPL、紫杉烷、袢霉素、博霉素B及其衍生物、美登素及其衍生物、多拉司他汀、丝裂霉素、氨基蝶呤、放线菌素、博来霉素、N8-乙酰基亚精胺、1-(2-氯乙基)-1,2-二甲磺酰肼、他利霉素、阿糖胞苷、依托泊苷、紫杉醇、拉霉素、鬼臼毒素、蛇形菌素(anguidine)、长春新碱、长春碱、吗啉-阿霉素、正-(5,5-二乙酰氧基-戊基)阿霉素及其衍生物中的一种或两种以上的组合。And/or, said D is preferably selected from: amanitin, auristatin, calicheamicin, camptothecin, camptothecin derivatives and metabolites (such as: SN-38 and derivatives thereof, 10 -Hydroxycamptothecin, 9-aminocamptothecin, 9-nitrocamptothecin), exitecan, Dxd, cryptomycin, daunomycin, dolastatin, doxorubicin, Duocamycin, Epothilone, Espamycin, Geldanamycin, Maytansine and its derivatives, Methotrexate, Monomethyl Auristatin E (“MMAE”), Monomethyl Auristatin F ("MMAF"), pyrrolobenzodiazepine, rhizobin, SG2285, tubulysin, vindesine, toxoid, irinotecan, topotecan, belotecan, exatecan , rutotecan, diflutecan, genitecan, canitecan, doxorubicin, epirubicin, morpholino doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrole Phenyl doxorubicin, MMAD, PBD and its derivatives, TPL, taxanes, cyclomycin, bleomycin B and its derivatives, maytansine and its derivatives, dolastatin, mitomyces Aminopterin, actinomycin, bleomycin, N8-acetylspermidine, 1-(2-chloroethyl)-1,2-dimethylsulfonyl hydrazide, talithromycin, a Cytoside, etoposide, paclitaxel, laramycin, podophyllotoxin, anguidine, vincristine, vinblastine, morpholine-doxorubicin, n-(5,5-diacetoxy One or a combination of two or more of doxorubicin and its derivatives.
在本发明的一些实施例中,所述D选自 In some embodiments of the present invention, the D is selected from
进一步的,所述的缀合物选自:Further, the conjugate is selected from:
本发明的缀合物属于分支型单分散聚乙二醇连接子-药物缀合物。The conjugates of the present invention belong to branched monodisperse polyethylene glycol linker-drug conjugates.
本发明另一方面还提供前述的缀合物在制备预防和/或治疗疾病药物中的应用,其特征在于,所述的疾病为癌症、炎症、自身免疫疾病。Another aspect of the present invention also provides the application of the aforementioned conjugates in the preparation of drugs for the prevention and/or treatment of diseases, characterized in that the diseases are cancer, inflammation and autoimmune diseases.
本发明另一方面还提供一种药物组合物包含治疗有效量的前述缀合物及药学上可接受的盐,载体,稀释剂或赋形剂或其组合。Another aspect of the present invention also provides a pharmaceutical composition comprising a therapeutically effective amount of the aforementioned conjugate and a pharmaceutically acceptable salt, a carrier, a diluent or an excipient or a combination thereof.
所述药物组合物为片剂、栓剂、丸剂、软和硬明胶胶囊剂、散剂、溶液剂、混悬剂或气雾剂的剂型。The pharmaceutical composition is in the form of tablets, suppositories, pills, soft and hard gelatin capsules, powders, solutions, suspensions or aerosols.
本发明另一方面还提供一种前述缀合物在制备预防和/或治疗疾病药物中的应用,所述药物具有抗癌活性,具体地,所述抗癌活性包括对肠癌、卵巢癌和肺癌具有抑制性的活性。Another aspect of the present invention also provides an application of the aforementioned conjugate in the preparation of a drug for the prevention and/or treatment of diseases, the drug has anticancer activity, specifically, the anticancer activity includes anticancer activity against intestinal cancer, ovarian cancer and Lung cancer has inhibitory activity.
本发明中出现的术语“烷基”可以是直链或支链的烷基,典型的烷基含有1至12(例如1、2、3、4、5、6、7、8、9、10、11、12)个碳原子,即C1-12烷基,例如C1-8烷基,C1-6烷基,C1-3烷基,烷基的实例如甲基、乙基、丙基、异丙基、正丁基、仲丁基、异丁基、叔丁基、2-乙基丁基、正戊基、异戊基、1-甲基戊基、1,3-二甲基丁基、正己基、1-甲基己基、正庚基、异庚基、1,1,3,3-四甲基丁基、1-甲基庚基、3-甲基庚基、正辛基、2-乙基己基、1,1,3-三甲基己基、1,1,3,3-四甲基戊基、壬基、癸基、十一烷基、1-甲基十一烷基、十二烷基、1,1,3,3,5,5-六甲基己基。The term "alkyl" appearing in the present invention can be a linear or branched alkyl group, and a typical alkyl group contains 1 to 12 (such as 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 , 11, 12) carbon atoms, namely C1-12 alkyl, such as C1-8 alkyl, C1-6 alkyl, C1-3 alkyl, examples of alkyl such as methyl, ethyl, propyl, iso Propyl, n-butyl, sec-butyl, isobutyl, tert-butyl, 2-ethylbutyl, n-pentyl, isopentyl, 1-methylpentyl, 1,3-dimethylbutyl , n-hexyl, 1-methylhexyl, n-heptyl, isoheptyl, 1,1,3,3-tetramethylbutyl, 1-methylheptyl, 3-methylheptyl, n-octyl, 2-ethylhexyl, 1,1,3-trimethylhexyl, 1,1,3,3-tetramethylpentyl, nonyl, decyl, undecyl, 1-methylundecyl , dodecyl, 1,1,3,3,5,5-hexamethylhexyl.
本发明中出现的术语“亚烷基”可以是直链或支链的烷基,典型的亚烷基含有1至12(例如1、2、3、4、5、6、7、8、9、10、11、12)个碳原子,即C1-12亚烷基,例如C1-18亚烷基,C1-12亚烷基,C1-8亚烷基,C1-6亚烷基,C1-3亚烷基,亚烷基的实例如亚乙基、亚丙基、亚异丙基、亚丁基、亚仲丁基、亚异丁基、2-乙基亚丁基、亚戊基、亚异戊基、1-甲基亚戊基、1,3-二甲基亚丁基、亚己基、1-甲基亚己基、亚庚基、1,1,3,3-四甲基亚丁基、1-甲基亚庚基、3-甲基亚庚基、亚辛基、2-乙基亚己基、1,1,3-三甲基亚己基、1,1,3,3-四甲基亚戊基、亚壬基、亚癸基、亚十一烷基和亚十二烷基的支化和直链的异构体。The term "alkylene" appearing in the present invention can be a linear or branched alkyl group, and a typical alkylene group contains 1 to 12 (such as 1, 2, 3, 4, 5, 6, 7, 8, 9 , 10, 11, 12) carbon atoms, that is, C1-12 alkylene, such as C1-18 alkylene, C1-12 alkylene, C1-8 alkylene, C1-6 alkylene, C1- 3 Alkylene, examples of alkylene are ethylene, propylene, isopropylene, butylene, sec-butylene, isobutylene, 2-ethylbutylene, pentylene, isobutylene Pentyl, 1-methylpentylene, 1,3-dimethylbutylene, hexylene, 1-methylhexylene, heptylene, 1,1,3,3-tetramethylbutylene, 1 -Methylheptylene, 3-methylheptylene, octylene, 2-ethylhexylene, 1,1,3-trimethylhexylene, 1,1,3,3-tetramethylhexylene Branched and straight-chain isomers of pentyl, nonylene, decylene, undecylene and dodecylene.
本发明中出现的术语“环烷基”包括取代环烷基和不饱和环烷基,典型的环烷基含有1至10(例如1、2、3、4、5、6、7、8、9、10)个碳原子,即C1-10环烷基,例如C3-6环烷基,环烷基的实例如环丙基、环丁基、甲基环丁基、环戊基、甲基环戊基、二甲基环戊基、三甲基环戊基、乙基环戊基、丙基环戊基、异丙基环戊基、环己基、甲基环己基、二甲基环己基、乙基环己基、环庚基、甲基环庚基、环辛基。The term "cycloalkyl" appearing in the present invention includes substituted cycloalkyl and unsaturated cycloalkyl, typical cycloalkyl contains 1 to 10 (such as 1, 2, 3, 4, 5, 6, 7, 8, 9,10) carbon atoms, that is, C 1-10 cycloalkyl, such as C3-6 cycloalkyl, examples of cycloalkyl such as cyclopropyl, cyclobutyl, methylcyclobutyl, cyclopentyl, methyl Cyclopentyl, dimethylcyclopentyl, trimethylcyclopentyl, ethylcyclopentyl, propylcyclopentyl, isopropylcyclopentyl, cyclohexyl, methylcyclohexyl, dimethylcyclopentyl Hexyl, ethylcyclohexyl, cycloheptyl, methylcycloheptyl, cyclooctyl.
本发明中,杂环烷基的实例是含有N、O、S、Si等元素的环烷基。In the present invention, examples of the heterocycloalkyl group are cycloalkyl groups containing elements such as N, O, S, and Si.
本发明中的术语芳基包括全碳芳基和杂芳基,其可以被一个或多个C1-C12烷基、卤素、C3-C8环烷基、C3-C8环烷氧基、C1-C18烷氧基、芳基取代。The term aryl in the present invention includes full carbon aryl and heteroaryl, which can be replaced by one or more C 1 -C 12 alkyl, halogen, C 3 -C 8 cycloalkyl, C 3 -C 8 cycloalkane Oxygen, C 1 -C 18 alkoxy, aryl substitution.
本发明中,术语“DMAP”是一种超强亲核的酰化作用催化剂,中文名是4-二甲氨基吡啶,它广泛应用于化学合成的新型高效催化剂,有较高的催化能力,对提高收率有极其明显的效果。Among the present invention, term "DMAP" is a kind of super-strong nucleophilic acylation catalyst, and Chinese name is 4-dimethylaminopyridine, and it is widely used in the novel high-efficiency catalyst of chemical synthesis, has higher catalytic ability, to Increasing the yield has an extremely obvious effect.
本发明中,术语“TFA”表示三氟乙酸。In the present invention, the term "TFA" means trifluoroacetic acid.
本发明中,术语“ADC”为Antibody-DrugConjugate,抗体药物偶联物。抗体药物偶联物ADC是具有靶向作用的单克隆抗体与具有特定药理学特性(如细胞毒作用)的化合物的结合,两部分通过连接子偶联为一个整体。In the present invention, the term "ADC" refers to Antibody-Drug Conjugate, antibody-drug conjugate. Antibody-drug conjugate ADC is a combination of a monoclonal antibody with a targeting effect and a compound with specific pharmacological properties (such as cytotoxicity), and the two parts are coupled into a whole through a linker.
本发明中,术语“DAR”(Drug-to-AntibodyRatio,药物抗体比值)是抗体药物偶联物的一个关键属性,是ADC药物研发过程重要的质控环节。In the present invention, the term "DAR" (Drug-to-Antibody Ratio, drug-to-antibody ratio) is a key attribute of antibody-drug conjugates and an important quality control link in the ADC drug development process.
本发明中,术语“mAb”为抗体。In the present invention, the term "mAb" is an antibody.
本发明中,术语“DCM”为二氯甲烷纯溶液。In the present invention, the term "DCM" refers to a pure solution of dichloromethane.
本发明中,术语“ACN”为乙腈的纯溶液。In the present invention, the term "ACN" refers to a pure solution of acetonitrile.
本发明中,术语“MPA”为流动相A;In the present invention, the term "MPA" is mobile phase A;
本发明中,术语“MPB”为流动相B。In the present invention, the term "MPB" refers to mobile phase B.
本发明中,术语“SPR”(SPR,SurfacePlasmonResonance),为光学表面等离子共振,可以通过监测生物反应过程中SPR角的动态变化,得到生物分子之间结合和相互作用的特异性信号。In the present invention, the term "SPR" (SPR, Surface Plasmon Resonance) refers to optical surface plasmon resonance, which can obtain specific signals of binding and interaction between biomolecules by monitoring the dynamic change of the SPR angle during the biological reaction process.
本发明中,术语“h”代表小时。In the present invention, the term "h" stands for hours.
本发明中,术语“室温”表示25±5℃。In the present invention, the term "room temperature" means 25±5°C.
本发明中,术语“DIEA”是指N,N-二异丙基乙胺,广泛用作溶剂,用于氨基酸多肽合成等。In the present invention, the term "DIEA" refers to N,N-diisopropylethylamine, which is widely used as a solvent for the synthesis of amino acid polypeptides and the like.
本发明中,术语“DMF”是指二甲基甲酰胺(DMF)是一种透明液体,常用作溶剂,能和水及大部分有机溶剂互溶。In the present invention, the term "DMF" means that dimethylformamide (DMF) is a transparent liquid, commonly used as a solvent, and can be miscible with water and most organic solvents.
本发明中,术语“HATU”是一种常用的多肽缩合试剂,应用在羧基与氨基合成肽键的反应之中。其系统命名为2-(7-氮杂苯并三氮唑)-N,N,N',N'-四甲基脲六氟磷酸酯,英文名2-(7-Azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluroniumhexafluorophosphate。In the present invention, the term "HATU" is a commonly used polypeptide condensation reagent, which is used in the reaction of carboxyl and amino groups to synthesize peptide bonds. Its system name is 2-(7-azabenzotriazole)-N,N,N',N'-tetramethyluronium hexafluorophosphate, English name 2-(7-Azabenzotriazol-1-yl) -N,N,N',N'-tetramethyluronium hexafluorophosphate.
本发明中,术语“MMAE”是指一甲基澳瑞他汀E简称MMAE,是抗有丝分裂剂,能通过阻断微管蛋白的聚合抑制细胞分裂,在抗体偶联药物(ADC)中颇受关注。In the present invention, the term "MMAE" refers to monomethyl auristatin E, referred to as MMAE, which is an anti-mitotic agent that can inhibit cell division by blocking the polymerization of tubulin, and has attracted much attention in antibody-drug conjugates (ADC) .
本发明中,术语“KD”是指亲和力常数KD(单位为升/质量摩尔浓度)表示,其计算公式为Among the present invention, term " KD " refers to the expression of affinity constant KD (unit is liter/mass molar concentration), and its calculation formula is
KD=[Ab·H]/([Ab][H])KD=[Ab·H]/([Ab][H])
其中[]表示摩尔浓度,Ab表示抗体,H表示半抗原,Ab—H表示抗体与半抗原的结合物Wherein [ ] represents the molar concentration, Ab represents the antibody, H represents the hapten, and Ab—H represents the conjugate of the antibody and the hapten
亲和力常数KD越高,则抗体结合半抗原的能力越强。抗体亲和力的强弱取决于抗体对位(Paratope)与所用抗原表位(epitope)之间的配合程度,包括接触面积的大小、吻合的密切程度、以及带点基团与疏水基团的分布等。The higher the affinity constant KD, the stronger the ability of the antibody to bind the hapten. The strength of antibody affinity depends on the degree of coordination between the paratope of the antibody and the epitope used, including the size of the contact area, the closeness of the matching, and the distribution of the dotted group and the hydrophobic group, etc. .
本发明的有益效果:Beneficial effects of the present invention:
(1)本发明提供的分支型聚乙二醇连接子-药物缀合物,可同时偶联两个细胞毒药物,构建双有效载荷抗体偶联药物。连接两种相同的毒素时,可提高载药量,实现构建高DAR值抗体偶联药物的目的;连接相同作用机制的不同毒素时,可实现协同作用,提高疗效;连接两种不同作用机制的毒素时,可降低耐药性的发生率。(1) The branched polyethylene glycol linker-drug conjugate provided by the present invention can simultaneously couple two cytotoxic drugs to construct a double payload antibody-drug conjugate. When connecting two identical toxins, the drug loading capacity can be increased to realize the purpose of constructing antibody-drug conjugates with high DAR value; when connecting different toxins with the same mechanism of action, synergistic effect can be achieved and the curative effect can be improved; connecting two different mechanisms of action Toxins can reduce the incidence of drug resistance.
(2)本发明提供的分支型聚乙二醇连接子-药物缀合物,其特定的结构赋予了其高亲水性,提高了其在血液循环中的保留时间;同时,平衡了细胞毒有效载荷的疏水性,缓解了带有疏水性毒素的ADC易聚集的问题。(2) The branched polyethylene glycol linker-drug conjugate provided by the present invention, its specific structure endows it with high hydrophilicity, improves its retention time in blood circulation; at the same time, balances cytotoxicity The hydrophobicity of the payload alleviates the problem of easy aggregation of ADCs with hydrophobic toxins.
(3)本发明提供的分支型聚乙二醇连接子-药物缀合物,与传统抗体药物偶联物相比,制备简单,其DAR值分布较窄,产品均一性高。(3) Compared with traditional antibody-drug conjugates, the branched polyethylene glycol linker-drug conjugate provided by the present invention is simpler to prepare, has a narrower distribution of DAR values, and has higher product uniformity.
(4)本发明提供的分支型聚乙二醇连接子-药物缀合物,裸抗和低交联度的ADC占比几乎为零(质谱检测不出DAR为0和1的组份)。(4) For the branched polyethylene glycol linker-drug conjugate provided by the present invention, the ratio of naked antibody and ADC with low cross-linking degree is almost zero (mass spectrometry cannot detect components with DAR of 0 and 1).
(5)本发明提供的分支型聚乙二醇连接子-药物缀合物,偶联后乙二醇所赋予的亲水性可以用来调节生物分子特性,在治疗肿瘤方面具有一定安全性和有效性。(5) The branched polyethylene glycol linker-drug conjugate provided by the present invention, after coupling, the hydrophilicity endowed by ethylene glycol can be used to adjust the characteristics of biomolecules, and has certain safety and safety in treating tumors. effectiveness.
(6)本发明分支型聚乙二醇连接子-药物缀合物的连接子中引入了PEG链,增加了细胞毒性药物的溶解性,使ADC偶联反应更充分,偶联效率更高。(6) The PEG chain is introduced into the linker of the branched polyethylene glycol linker-drug conjugate of the present invention, which increases the solubility of cytotoxic drugs, makes the ADC coupling reaction more complete and the coupling efficiency is higher.
(7)本发明提供的偶联方法,适用于大部分抗体,因此具有广泛的应用前景,具有较快的交联速度,交联反应时间通常在2-4小时以内便可反应完毕。(7) The coupling method provided by the present invention is applicable to most antibodies, so it has a wide application prospect and has a relatively fast cross-linking speed, and the cross-linking reaction time can usually be completed within 2-4 hours.
附图说明Description of drawings
图1所示为化合物1的质谱图。Figure 1 shows the mass spectrum of compound 1.
具体实施方式detailed description
为了能够更清楚地理解本发明的技术内容,特举以下实施例详细说明,对本发明的技术方案进行清楚、完整地描述,其目的仅在于更好理解本发明的内容而非限制本发明的保护范围领域普通技术人员在没有作出创造性劳动前提下所获得的所有其他实施例,都属于本发明保护的范围。In order to understand the technical content of the present invention more clearly, the following examples are given in detail to describe the technical solution of the present invention clearly and completely, the purpose of which is only to better understand the content of the present invention rather than limit the protection of the present invention Scope All other embodiments obtained by persons of ordinary skill in the field without creative efforts fall within the protection scope of the present invention.
本发明实施例中所用试剂均为市面上可以购买到的常规试剂。The reagents used in the examples of the present invention are commercially available conventional reagents.
实施例1化合物1的合成The synthesis of embodiment 1 compound 1
1.化合物1a的合成1. Synthesis of Compound 1a
将原料DBCO-NHS(8.0g,19mmol)加入到80mLDMF中,在依次加入L-glutanicacid(3.89g,23mmol)和TEA(4.0g,39mmol),反应室温搅拌15h,HPLC显示反应完全。反应液用200mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色固体6.5g(化合物1a),产率75.5%。The raw material DBCO-NHS (8.0g, 19mmol) was added to 80mL DMF, followed by the addition of L-glutanicacid (3.89g, 23mmol) and TEA (4.0g, 39mmol), and the reaction was stirred at room temperature for 15h. HPLC showed that the reaction was complete. The reaction solution was washed three times with 200 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 6.5 g of light yellow solid (Compound 1a), with a yield of 75.5%.
MSm/z(ESI):435.2[M+1]MSm/z(ESI):435.2[M+1]
2.化合物1b的合成2. Synthesis of compound 1b
将化合物1a(6.2g,14.3mmol),加入到60mLDCM中,再依次加入NH2-PEG8-PAOMe(14.3g,31.3mmol),DIEA(5.5g,43mmol),HATU(16.3g,43mmol)。将反应体系置于室温下搅拌6h,HPLC显示反应完全。反应液用100mL饱和氯化铵洗涤三次,有机相用饱和氯化钠溶液洗涤一次,再用无水硫酸钠干燥,旋干溶剂得到粗品15g。粗品用硅胶柱层析纯化(DCM/MeOH50/1-20/1)得到淡黄色油状物10.5g(化合物1b),产率58%。Compound 1a (6.2g, 14.3mmol) was added to 60mL DCM, and then NH2-PEG8-PAOMe (14.3g, 31.3mmol), DIEA (5.5g, 43mmol), HATU (16.3g, 43mmol) were added sequentially. The reaction system was stirred at room temperature for 6 h, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated ammonium chloride, the organic phase was washed once with saturated sodium chloride solution, then dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 15 g of crude product. The crude product was purified by silica gel column chromatography (DCM/MeOH 50/1-20/1) to obtain 10.5 g of light yellow oil (compound 1b), with a yield of 58%.
MSm/z(ESI):1309.7[M+1]MSm/z(ESI):1309.7[M+1]
3.化合物1c的合成3. Synthesis of Compound 1c
将化合物1b(10g,7mmol)溶于50mLTHF中,再加入预先溶解好的50mLLiOH(1.7g,74mmol)水溶液。反应体系置于室温下搅拌1h,HPLC显示反应完全。反应液用100mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色油状物8.0g(化合物1c),产率81%。Compound 1b (10g, 7mmol) was dissolved in 50mL THF, and then 50mL LiOH (1.7g, 74mmol) aqueous solution dissolved in advance was added. The reaction system was stirred at room temperature for 1 h, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 8.0 g of light yellow oil (compound 1c), with a yield of 81%.
MSm/z(ESI):1281.6[M+1]MSm/z(ESI):1281.6[M+1]
4.化合物1d的合成4. Synthesis of Compound 1d
将化合物1c(8.0g,6mmol)加入到80mLDMF中,再依次加入VC-PAB(4.97g,13mmol),DIEA(2.4g,18mmol),HATU(7.1g,18mmol)。将反应体系置于室温下搅拌2h,HPLC显示反应完全。反应液直接制备纯化。最后得到黄色固体8.5g(化合物1d),产率68%。Compound 1c (8.0g, 6mmol) was added into 80mL DMF, followed by VC-PAB (4.97g, 13mmol), DIEA (2.4g, 18mmol), HATU (7.1g, 18mmol). The reaction system was stirred at room temperature for 2 h, and HPLC showed that the reaction was complete. The reaction solution was directly prepared and purified. Finally, 8.5 g of yellow solid (compound 1d) was obtained with a yield of 68%.
MSm/z(ESI):2004.1[M+1]MSm/z(ESI):2004.1[M+1]
5.化合物1e的合成5. Synthesis of compound 1e
将原料SN38(30g,76mmol)加入到300mLDCM中,再依次加入DIEA(14.8g,0.1mol)和TBSCl(13.8g,91mmol),反应室温搅拌5h,TLC显示反应完全。反应液用400mL饱和氯化钠溶液洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到粗品,再用硅胶柱层析纯化(正己烷/EA20/1-8/1)得到白色固体42g(化合物1e),产率82.3%。The raw material SN38 (30g, 76mmol) was added to 300mL DCM, and then DIEA (14.8g, 0.1mol) and TBSCl (13.8g, 91mmol) were added in sequence, and the reaction was stirred at room temperature for 5h. TLC showed that the reaction was complete. The reaction solution was washed three times with 400 mL of saturated sodium chloride solution, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain a crude product, which was then purified by silica gel column chromatography (n-hexane/EA20/1-8/1) to obtain 42 g of a white solid (Compound 1e), the yield was 82.3%.
MSm/z(ESI):507.2[M+1]MSm/z(ESI):507.2[M+1]
6.化合物1f的合成6. Synthesis of Compound 1f
将化合物1e(10g,14.8mmol)溶于70mLDCM中,冰水浴降温至0-5℃,依次加入对硝基氯甲酸苯酯(3.3g,16mmol)和DMAP(2.89g,24mmol),自然至室温搅拌2H,TLC显示反应完全后。分别用饱和硫酸氢钠溶液50mL和NaCl50mL洗涤一次,有机相用无水硫酸钠干燥,浓缩得到粗品15g。硅胶柱纯化(DCM/EA20/1-1/1)得到黄色固体8g(化合物1f),产率61.5%。Compound 1e (10g, 14.8mmol) was dissolved in 70mL DCM, cooled to 0-5°C in an ice-water bath, and p-nitrophenylchloroformate (3.3g, 16mmol) and DMAP (2.89g, 24mmol) were added successively, and allowed to cool to room temperature After stirring for 2H, TLC showed that the reaction was complete. Wash once with 50 mL of saturated sodium bisulfate solution and 50 mL of NaCl, dry the organic phase with anhydrous sodium sulfate, and concentrate to obtain 15 g of crude product. Silica gel column purification (DCM/EA20/1-1/1) gave 8 g of yellow solid (compound 1f), yield 61.5%.
MSm/z(ESI):672.2[M+1]MSm/z(ESI):672.2[M+1]
7.化合物1g的合成7. Synthesis of Compound 1g
将化合物1f(5.6g,8mmol)溶于50mLDCM和10mLDMSO中,再加入化合物1d(8.0g,4mmol)和DMAP(1.07g,8.7mmol),室温下搅拌1h,HPLC显示反应完全。向反应体系加入100mLDCM和50mL饱和硫酸氢钠水溶液,待其分层后,有机相用50mL饱和氯化钠洗涤一次,无水硫酸钠干燥,浓缩后得到13g粗品(化合物1g)。Compound 1f (5.6g, 8mmol) was dissolved in 50mL DCM and 10mL DMSO, then compound 1d (8.0g, 4mmol) and DMAP (1.07g, 8.7mmol) were added, stirred at room temperature for 1h, HPLC showed that the reaction was complete. 100mL DCM and 50mL saturated aqueous sodium bisulfate solution were added to the reaction system. After the layers were separated, the organic phase was washed once with 50mL saturated sodium chloride, dried over anhydrous sodium sulfate, and concentrated to obtain 13g of crude product (compound 1g).
MSm/z(ESI):1534.7[M/2+1]MSm/z(ESI):1534.7[M/2+1]
7.化合物1的合成7. Synthesis of compound 1
将上步粗品(化合物1g)溶于THF(80mL)中,再加入1mol/LHCl水溶液(20mL),室温搅拌3h,HPLC显示原料反应完全。反应体系用NaHCO3饱和水溶液100mL调pH=7~8,然后用二氯甲烷(100mL)萃取两次,有机相用无水硫酸钠干燥,浓缩得到粗品。粗品用高压制备系统纯化,最后得到黄色的固体5.1g(化合物1),产率46%,为化合物1.The crude product from the previous step (compound 1g) was dissolved in THF (80 mL), and then 1 mol/L HCl aqueous solution (20 mL) was added, and stirred at room temperature for 3 h. HPLC showed that the reaction of the raw material was complete. The reaction system was adjusted to pH=7-8 with 100 mL of NaHCO3 saturated aqueous solution, then extracted twice with dichloromethane (100 mL), the organic phase was dried over anhydrous sodium sulfate, and concentrated to obtain a crude product. The crude product was purified with a high-pressure preparation system to finally obtain 5.1 g of a yellow solid (compound 1), with a yield of 46%, which was compound 1.
MSm/z(ESI):1421.6[M/2+1]MSm/z(ESI):1421.6[M/2+1]
实施例2化合物2的合成The synthesis of embodiment 2 compound 2
1.化合物2a的合成1. Synthesis of Compound 2a
将原料DBCO-NHS(4.0g,10mmol)加入到40mLDMF中,依次加入NH2-PEG8-PA(4.9g,11mmol)和TEA(2.0g,20mmol),反应室温搅拌15h,HPLC显示反应完全。反应液用200mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色固体5.1g(化合物2a),产率71%。The raw material DBCO-NHS (4.0g, 10mmol) was added to 40mL DMF, NH 2 -PEG 8 -PA (4.9g, 11mmol) and TEA (2.0g, 20mmol) were added in sequence, and the reaction was stirred at room temperature for 15h. HPLC showed that the reaction was complete. The reaction solution was washed three times with 200 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 5.1 g of a light yellow solid (compound 2a), with a yield of 71%.
MSm/z(ESI):729.4[M+1]MSm/z(ESI):729.4[M+1]
2.化合物2b的合成2. Synthesis of compound 2b
将化合物2a(5.0g,7.0mmol)和N-羟基琥珀酰亚胺(1.0g,8.40mmol)溶于60mLDCM中,加入1-(3-二甲基氨基丙基)-3-乙基碳二亚胺(1.6g,8.40mmol),反应室温搅拌4h,得到化合物2b的反应液,直接用于下一步反应。Compound 2a (5.0g, 7.0mmol) and N-hydroxysuccinimide (1.0g, 8.40mmol) were dissolved in 60mL DCM, and 1-(3-dimethylaminopropyl)-3-ethylcarbodi Imine (1.6g, 8.40mmol) was stirred at room temperature for 4h to obtain the reaction solution of compound 2b, which was directly used in the next reaction.
MSm/z(ESI):826.4[M+1]MSm/z(ESI):826.4[M+1]
3.化合物2c的合成3. Synthesis of compound 2c
将L-glutanicacid(1.2g,8mmol)加入上述得到的化合物2b的反应液中,加入TEA(0.8g,8mmol),室温搅拌15h,HPLC显示反应完全。反应液用100mL饱和硫酸氢钠溶液和DCM60mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色油状物4.2g(化合物2c),产率70.2%。Add L-glutanic acid (1.2g, 8mmol) to the reaction solution of compound 2b obtained above, add TEA (0.8g, 8mmol), stir at room temperature for 15h, HPLC shows that the reaction is complete. The reaction solution was washed three times with 100 mL of saturated sodium bisulfate solution and 60 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 4.2 g of light yellow oil (compound 2c), with a yield of 70.2%.
MSm/z(ESI):858.4[M+1]MSm/z(ESI):858.4[M+1]
4.化合物2d的合成4. Synthesis of compound 2d
将化合物2c(4.0g,4.6mmol)加入到40mLDMF中,再依次加入VC-PAB(3.8g,10mmol),DIEA(1.9g,14mmol),HATU(5.5g,14mmol)。将反应体系置于室温下搅拌2h,HPLC显示反应完全。反应液直接制备纯化。最后得到黄色固体3.9g(化合物2d),产率54%。Compound 2c (4.0g, 4.6mmol) was added to 40mL DMF, followed by VC-PAB (3.8g, 10mmol), DIEA (1.9g, 14mmol), HATU (5.5g, 14mmol). The reaction system was stirred at room temperature for 2 h, and HPLC showed that the reaction was complete. The reaction solution was directly prepared and purified. Finally, 3.9 g of yellow solid (compound 2d) was obtained with a yield of 54%.
MSm/z(ESI):1580.8[M+1]MSm/z(ESI):1580.8[M+1]
5.化合物2e的合成5. Synthesis of Compound 2e
将化合物1f(2.8g,4mmol)溶于30mLDCM和5mLDMSO中,再加入化合物2d(3.2g,2mmol)和DMAP(0.5g,4.4mmol),室温下搅拌2h,HPLC显示反应完全。将反应体系加入50mLDCM和50mL饱和硫酸氢钠水溶液,有机相用50mL饱和氯化钠洗涤一次,无水硫酸钠干燥,浓缩后得到2.6g粗品。Compound 1f (2.8g, 4mmol) was dissolved in 30mL DCM and 5mL DMSO, then compound 2d (3.2g, 2mmol) and DMAP (0.5g, 4.4mmol) were added, stirred at room temperature for 2h, HPLC showed that the reaction was complete. Add 50 mL of DCM and 50 mL of saturated aqueous sodium bisulfate to the reaction system, wash the organic phase once with 50 mL of saturated sodium chloride, dry over anhydrous sodium sulfate, and concentrate to obtain 2.6 g of crude product.
MSm/z(ESI):1323.1[M/2+1]MSm/z(ESI):1323.1[M/2+1]
6.化合物2的合成6. Synthesis of compound 2
将上步粗品溶于THF(20mL)中,再加入1mol/LHCl水溶液(5mL),室温搅拌3h,HPLC显示原料反应完全。反应体系用NaHCO3饱和水溶液调pH=7~8,然后用二氯甲烷(100mL)萃取两次,有机相用无水硫酸钠干燥,浓缩得到粗品。粗品用高压制备系统纯化,最后得到黄色固体1.2g(化合物2),产率48%。The crude product from the previous step was dissolved in THF (20 mL), and then 1 mol/L HCl aqueous solution (5 mL) was added, and stirred at room temperature for 3 h. HPLC showed that the reaction of the raw material was complete. The reaction system was adjusted to pH=7-8 with saturated NaHCO 3 aqueous solution, then extracted twice with dichloromethane (100 mL), the organic phase was dried over anhydrous sodium sulfate, and concentrated to obtain a crude product. The crude product was purified by a high-pressure preparation system to finally obtain 1.2 g of a yellow solid (compound 2), with a yield of 48%.
MSm/z(ESI):1209.0[M/2+1]MSm/z(ESI):1209.0[M/2+1]
实施例3化合物3的合成The synthesis of embodiment 3 compound 3
1.化合物3a的合成1. Synthesis of compound 3a
将原料DBCO-NHS(8.0g,19mmol)加入到80mLDMF中,在依次加入L-glutanicacid(3.89g,23mmol)和TEA(4.0g,39mmol),反应室温搅拌15h,HPLC显示反应完全。反应液用200mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色固体6.5g(化合物3a),产率75.5%。The raw material DBCO-NHS (8.0g, 19mmol) was added to 80mL DMF, followed by the addition of L-glutanicacid (3.89g, 23mmol) and TEA (4.0g, 39mmol), and the reaction was stirred at room temperature for 15h. HPLC showed that the reaction was complete. The reaction solution was washed three times with 200 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 6.5 g of a light yellow solid (compound 3a), with a yield of 75.5%.
MSm/z(ESI):435.2[M+1]MSm/z(ESI):435.2[M+1]
2.化合物3b的合成2. Synthesis of compound 3b
将化合物3a(6.2g,14.3mmol),加入到60mLDCM中,再依次加入NH2-PEG8-PAOMe(14.3g,31.3mmol),DIEA(5.5g,43mmol),HATU(16.3g,43mmol)。将反应体系置于室温下搅拌6H,HPLC显示反应完全。反应液用100mL饱和氯化铵洗涤三次,有机相用饱和氯化钠溶液洗涤一次,再用无水硫酸钠干燥,旋干溶剂得到粗品15g。粗品用硅胶柱层析纯化(DCM/MeOH50/1-20/1)得到淡黄色油状物10.5g(化合物3b),产率58%。Compound 3a (6.2g, 14.3mmol) was added to 60mL DCM, and then NH 2 -PEG 8 -PAOMe (14.3g, 31.3mmol), DIEA (5.5g, 43mmol), HATU (16.3g, 43mmol) were added in sequence. The reaction system was stirred at room temperature for 6H, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated ammonium chloride, the organic phase was washed once with saturated sodium chloride solution, then dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 15 g of crude product. The crude product was purified by silica gel column chromatography (DCM/MeOH 50/1-20/1) to obtain 10.5 g of light yellow oil (compound 3b), with a yield of 58%.
MSm/z(ESI):1309.7[M+1]MSm/z(ESI):1309.7[M+1]
3.化合物3c的合成3. Synthesis of compound 3c
将化合物3b(10g,7mmol)溶于50mLTHF中,再加入预先溶解好的50mLLiOH(1.7g,74mmol)水溶液。反应体系置于室温下搅拌1h,HPLC显示反应完全。反应液用100mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色油状物8.0g(化合物3c),产率81%。Compound 3b (10g, 7mmol) was dissolved in 50mL THF, and then 50mL LiOH (1.7g, 74mmol) aqueous solution dissolved in advance was added. The reaction system was stirred at room temperature for 1 h, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 8.0 g of light yellow oil (compound 3c), with a yield of 81%.
MSm/z(ESI):1281.6[M+1]MSm/z(ESI):1281.6[M+1]
4.化合物3d的合成4. Synthesis of Compound 3d
将化合物3c(8.0g,6mmol)加入到80mLDMF中,再依次加入VC-PAB(4.97g,13mmol),DIEA(2.4g,18mmol),HATU(7.1g,18mmol)。将反应体系置于室温下搅拌2h,HPLC显示反应完全。反应液直接制备纯化。最后得到黄色固体8.5g(化合物3d),产率68%。Compound 3c (8.0g, 6mmol) was added into 80mL DMF, and then VC-PAB (4.97g, 13mmol), DIEA (2.4g, 18mmol), HATU (7.1g, 18mmol) were added sequentially. The reaction system was stirred at room temperature for 2 h, and HPLC showed that the reaction was complete. The reaction solution was directly prepared and purified. Finally, 8.5 g of yellow solid (compound 3d) was obtained with a yield of 68%.
MSm/z(ESI):2004.1[M+1]MSm/z(ESI):2004.1[M+1]
6.化合物3的合成6. Synthesis of compound 3
将化合物3d(8g,4mmol)溶于50mLDCM中,冰水浴降温至0-5℃,依次加入对硝基氯甲酸苯酯(2.1g,10mmol)和DMAP(1.9g,16mmol),室温搅拌2h,TLC显示反应完全后,加入MMAE(6.5g,9mmol)和DMAP(1.1g,9mmol)。分别用饱和硫酸氢钠溶液50mL和NaCl50mL洗涤一次,有机相用无水硫酸钠干燥,浓缩有机相得到粗品。经硅胶柱层析高压制备分离得到黄色固体4.7g(化合物3),产率34%。Compound 3d (8g, 4mmol) was dissolved in 50mL DCM, the temperature was cooled to 0-5°C in an ice-water bath, p-nitrophenylchloroformate (2.1g, 10mmol) and DMAP (1.9g, 16mmol) were added sequentially, and stirred at room temperature for 2h, After TLC showed the reaction was complete, MMAE (6.5 g, 9 mmol) and DMAP (1.1 g, 9 mmol) were added. Wash once with 50 mL of saturated sodium bisulfate solution and 50 mL of NaCl, dry the organic phase with anhydrous sodium sulfate, and concentrate the organic phase to obtain a crude product. 4.7 g of yellow solid (compound 3) was obtained by silica gel column chromatography and high-pressure preparation, and the yield was 34%.
MSm/z(ESI):1746[M/2+1]MSm/z(ESI):1746[M/2+1]
实施例4化合物4的合成The synthesis of embodiment 4 compound 4
1.化合物4a的合成1. Synthesis of compound 4a
将原料DBCO-NHS(8.0g,19mmol)加入到80mLDMF中,在依次加入L-glutanicacid(3.89g,23mmol)和TEA(4.0g,39mmol),反应室温搅拌15h,HPLC显示反应完全。反应液用200mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色固体6.5g(化合物4a),产率75.5%。The raw material DBCO-NHS (8.0g, 19mmol) was added to 80mL DMF, followed by the addition of L-glutanicacid (3.89g, 23mmol) and TEA (4.0g, 39mmol), and the reaction was stirred at room temperature for 15h. HPLC showed that the reaction was complete. The reaction solution was washed three times with 200 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 6.5 g of a light yellow solid (compound 4a), with a yield of 75.5%.
MSm/z(ESI):435.2[M+1]MSm/z(ESI):435.2[M+1]
2.化合物4b的合成2. Synthesis of Compound 4b
将化合物4a(6.2g,14.3mmol),加入到60mLDCM中,再依次加入NH2-PEG8-PAOMe(14.3g,31.3mmol),DIEA(5.5g,43mmol),HATU(16.3g,43mmol)。将反应体系置于室温下搅拌6h,HPLC显示反应完全。反应液用100mL饱和氯化铵洗涤三次,有机相用饱和氯化钠溶液洗涤一次,再用无水硫酸钠干燥,旋干溶剂得到粗品15g。粗品用硅胶柱层析纯化(DCM/MeOH50/1-20/1)得到淡黄色油状物10.5g(化合物4b),产率58%。Compound 4a (6.2g, 14.3mmol) was added to 60mL DCM, and NH 2 -PEG 8 -PAOMe (14.3g, 31.3mmol), DIEA (5.5g, 43mmol), HATU (16.3g, 43mmol) were added in sequence. The reaction system was stirred at room temperature for 6 h, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated ammonium chloride, the organic phase was washed once with saturated sodium chloride solution, then dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 15 g of crude product. The crude product was purified by silica gel column chromatography (DCM/MeOH 50/1-20/1) to obtain 10.5 g of light yellow oil (compound 4b), with a yield of 58%.
MSm/z(ESI):1309.7[M+1]MSm/z(ESI):1309.7[M+1]
3.化合物4c的合成3. Synthesis of Compound 4c
将化合物4b(10g,7mmol)溶于50mLTHF中,再加入预先溶解好的50mLLiOH(1.7g,74mmol)水溶液。反应体系置于室温下搅拌1h,HPLC显示反应完全。反应液用100mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色油状物8.0g(化合物4c),产率81%。Compound 4b (10g, 7mmol) was dissolved in 50mL THF, and then 50mL LiOH (1.7g, 74mmol) aqueous solution dissolved in advance was added. The reaction system was stirred at room temperature for 1 h, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 8.0 g of light yellow oil (compound 4c), with a yield of 81%.
MSm/z(ESI):1281.6[M+1]MSm/z(ESI):1281.6[M+1]
4.化合物4d的合成4. Synthesis of compound 4d
将化合物4c(8.0g,6mmol)加入到80mLDMF中,再依次加入VC-PAB(4.97g,13mmol),DIEA(2.4g,18mmol),HATU(7.1g,18mmol)。将反应体系置于室温下搅拌2h,HPLC显示反应完全,得到反应液。将前述反应液直接制备纯化。最后得到黄色固体8.5g(化合物4d),产率68%。Compound 4c (8.0g, 6mmol) was added into 80mL DMF, and then VC-PAB (4.97g, 13mmol), DIEA (2.4g, 18mmol), HATU (7.1g, 18mmol) were added sequentially. The reaction system was stirred at room temperature for 2 h, HPLC showed that the reaction was complete, and a reaction solution was obtained. The aforementioned reaction solution was directly prepared and purified. Finally, 8.5 g of yellow solid (compound 4d) was obtained with a yield of 68%.
MSm/z(ESI):2004.1[M+1]MSm/z(ESI):2004.1[M+1]
5.化合物4e的合成5. Synthesis of Compound 4e
将化合物4d(8g,4mmol)溶于50mLDCM中,冰水浴降温至0-5℃,依次加入对硝基氯甲酸苯酯(2.1g,10mmol)和DMAP(1.9g,16mmol),室温搅拌2h,TLC显示反应完全后,分别用饱和硫酸氢钠溶液50mL和NaCl50mL洗涤一次,使反应体系分层,有机相用无水硫酸钠干燥,浓缩得到粗品6.0g(化合物4e),产率65%。Compound 4d (8g, 4mmol) was dissolved in 50mL DCM, the temperature was cooled to 0-5°C in an ice-water bath, p-nitrochloroformate (2.1g, 10mmol) and DMAP (1.9g, 16mmol) were added successively, and stirred at room temperature for 2h, After TLC showed that the reaction was complete, the reaction system was washed once with 50 mL of saturated sodium bisulfate solution and 50 mL of NaCl, and the reaction system was separated. The organic phase was dried with anhydrous sodium sulfate and concentrated to obtain 6.0 g of crude product (compound 4e), with a yield of 65%.
MSm/z(ESI):1167.5[M/2+1]MSm/z(ESI):1167.5[M/2+1]
6.化合物4f的合成6. Synthesis of compound 4f
将化合物4e(4.6g,2mmol)溶于20mLDCM和5mLDMSO中,再加入化合物SN38-OTBS(0.5g,1mmol)和DMAP(0.2g,1.5mmol),室温下搅拌2h,HPLC显示反应完全。将反应体系加入50mLDCM和50mL饱和硫酸氢钠水溶液,使反应体系分层,有机相用50mL饱和氯化钠溶液洗涤一次,无水硫酸钠干燥,浓缩后得到粗品,经高压制备分离得到黄色固体1.6g(化合物4f),产率61%。Compound 4e (4.6g, 2mmol) was dissolved in 20mL DCM and 5mLDMSO, then compound SN38-OTBS (0.5g, 1mmol) and DMAP (0.2g, 1.5mmol) were added, stirred at room temperature for 2h, HPLC showed that the reaction was complete. Add 50mL DCM and 50mL saturated sodium bisulfate aqueous solution to the reaction system to separate the reaction system, wash the organic phase once with 50mL saturated sodium chloride solution, dry over anhydrous sodium sulfate, and concentrate to obtain the crude product, which is separated by high-pressure preparation to obtain a yellow solid 1.6 g (compound 4f), yield 61%.
MSm/z(ESI):1351.2[M/2+1]MSm/z(ESI):1351.2[M/2+1]
7.化合物4g的合成7. Synthesis of compound 4g
将化合物4f(1.3g,0.5mmol)溶于10mLDCM中,再加入化合物MMAE(0.4g,0.6mmol)和DMAP(0.1g,1.0mmol),室温下搅拌2H,HPLC显示反应完全。将反应体系加入20mLDCM和20mL饱和硫酸氢钠水溶液,使反应体系分层,有机相用20mL饱和氯化钠溶液洗涤一次,无水硫酸钠干燥,浓缩后得到粗品,经高压制备分离得到黄色固体1.6g(化合物4g),产率70%。Compound 4f (1.3g, 0.5mmol) was dissolved in 10mL DCM, compound MMAE (0.4g, 0.6mmol) and DMAP (0.1g, 1.0mmol) were added, stirred at room temperature for 2H, HPLC showed that the reaction was complete. Add 20mL DCM and 20mL saturated sodium bisulfate aqueous solution to the reaction system to separate the reaction system, wash the organic phase once with 20mL saturated sodium chloride solution, dry over anhydrous sodium sulfate, and concentrate to obtain the crude product, which is separated by high-pressure preparation to obtain a yellow solid 1.6 g (compound 4g), yield 70%.
MSm/z(ESI):1640.4[M/2+1]MSm/z(ESI):1640.4[M/2+1]
8.化合物4的合成8. Synthesis of Compound 4
将化合物4g(1.5g,0.46mmol)溶于THF(10mL)中,再加入1mol/LHCl水溶液(3mL),室温搅拌3h,HPLC显示原料反应完全。反应体系用NaHCO3饱和水溶液调pH=7~8,然后用二氯甲烷(100mL)萃取两次,有机相用无水硫酸钠干燥,浓缩得到粗品。粗品用高压制备纯化,最后得到黄色固体700mg(化合物4),产率48%。Compound 4g (1.5g, 0.46mmol) was dissolved in THF (10mL), then 1mol/L HCl aqueous solution (3mL) was added, stirred at room temperature for 3h, HPLC showed that the reaction of the raw materials was complete. The reaction system was adjusted to pH=7-8 with saturated NaHCO 3 aqueous solution, then extracted twice with dichloromethane (100 mL), the organic phase was dried over anhydrous sodium sulfate, and concentrated to obtain a crude product. The crude product was purified by high-pressure preparation, and finally 700 mg of yellow solid (compound 4) was obtained with a yield of 48%.
MSm/z(ESI):1583.4[M/2+1]MSm/z(ESI):1583.4[M/2+1]
实施例5化合物5的合成The synthesis of embodiment 5 compound 5
1.化合物5a的合成1. Synthesis of Compound 5a
将原料DBCO-NHS(8.0g,19mmol)加入到80mLDMF中,在依次加入L-glutanicacid(3.89g,23mmol)和TEA(4.0g,39mmol),反应室温搅拌15h,HPLC显示反应完全。反应液用200mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色固体6.5g(化合物5a),产率75.5%。The raw material DBCO-NHS (8.0g, 19mmol) was added to 80mL DMF, followed by the addition of L-glutanicacid (3.89g, 23mmol) and TEA (4.0g, 39mmol), and the reaction was stirred at room temperature for 15h. HPLC showed that the reaction was complete. The reaction solution was washed three times with 200 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 6.5 g of a light yellow solid (compound 5a), with a yield of 75.5%.
MSm/z(ESI):435.2[M+1]MSm/z(ESI):435.2[M+1]
2.化合物5b的合成2. Synthesis of Compound 5b
将化合物5a(6.2g,14.3mmol),加入到60mLDCM中,再依次加入NH2-PEG8-PAOMe(14.3g,31.3mmol),DIEA(5.5g,43mmol),HATU(16.3g,43mmol)。将反应体系置于室温下搅拌6H,HPLC显示反应完全。反应液用100mL饱和氯化铵洗涤三次,有机相用饱和氯化钠溶液洗涤一次,再用无水硫酸钠干燥,旋干溶剂得到粗品15g。粗品用硅胶柱层析纯化(DCM/MeOH50/1-20/1)得到淡黄色油状物10.5g(化合物5b),产率58%。Compound 5a (6.2g, 14.3mmol) was added to 60mL DCM, and then NH2-PEG8-PAOMe (14.3g, 31.3mmol), DIEA (5.5g, 43mmol), HATU (16.3g, 43mmol) were added sequentially. The reaction system was stirred at room temperature for 6H, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated ammonium chloride, the organic phase was washed once with saturated sodium chloride solution, then dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 15 g of crude product. The crude product was purified by silica gel column chromatography (DCM/MeOH 50/1-20/1) to obtain 10.5 g of light yellow oil (compound 5b), with a yield of 58%.
MSm/z(ESI):1309.7[M+1]MSm/z(ESI):1309.7[M+1]
3.化合物5c的合成3. Synthesis of compound 5c
将化合物5b(10g,7mmol)溶于50mLTHF中,再加入预先溶解好的50mLLiOH(1.7g,74mmol)水溶液。反应体系置于室温下搅拌1h,HPLC显示反应完全。反应液用100mL饱和硫酸氢钠溶液和DCM100mL洗涤三次,有机相用无水硫酸钠干燥,旋干溶剂得到淡黄色油状物8.0g(化合物5c),产率81%。Compound 5b (10g, 7mmol) was dissolved in 50mL THF, and then 50mL LiOH (1.7g, 74mmol) aqueous solution dissolved in advance was added. The reaction system was stirred at room temperature for 1 h, and HPLC showed that the reaction was complete. The reaction solution was washed three times with 100 mL of saturated sodium bisulfate solution and 100 mL of DCM, the organic phase was dried with anhydrous sodium sulfate, and the solvent was spin-dried to obtain 8.0 g of light yellow oil (compound 5c), with a yield of 81%.
MSm/z(ESI):1281.6[M+1]MSm/z(ESI):1281.6[M+1]
4.化合物5d的合成4. Synthesis of compound 5d
将化合物5c(8.0g,6mmol)加入到80mLDMF中,再依次加入VC-PAB(4.97g,13mmol),DIEA(2.4g,18mmol),HATU(7.1g,18mmol)。将反应体系置于室温下搅拌2h,HPLC显示反应完全。反应液直接经高压制备系统纯化。最后得到黄色固体8.5g(.化合物5d),产率68%。Compound 5c (8.0g, 6mmol) was added into 80mL DMF, and then VC-PAB (4.97g, 13mmol), DIEA (2.4g, 18mmol), HATU (7.1g, 18mmol) were added sequentially. The reaction system was stirred at room temperature for 2 h, and HPLC showed that the reaction was complete. The reaction solution is directly purified by a high-pressure preparation system. Finally, 8.5 g of yellow solid (.compound 5d) was obtained with a yield of 68%.
MSm/z(ESI):2004.1[M+1]MSm/z(ESI):2004.1[M+1]
5.化合物5e的合成5. Synthesis of compound 5e
将化合物5d(8g,4mmol)溶于50mLDCM中,冰水浴降温至0-5℃,依次加入对硝基氯甲酸苯酯(2.1g,10mmol)和DMAP(1.9g,16mmol),室温搅拌2h,TLC显示反应完全后,分别用饱和硫酸氢钠溶液50mL和饱氯化钠溶液50mL洗涤一次,有机相用无水硫酸钠干燥,浓缩得到粗品6.0g(化合物5e),产率65%。Compound 5d (8g, 4mmol) was dissolved in 50mL DCM, the temperature was cooled to 0-5°C in an ice-water bath, p-nitrophenylchloroformate (2.1g, 10mmol) and DMAP (1.9g, 16mmol) were added sequentially, and stirred at room temperature for 2h, After TLC showed that the reaction was complete, it was washed once with 50 mL of saturated sodium bisulfate solution and 50 mL of saturated sodium chloride solution, and the organic phase was dried over anhydrous sodium sulfate and concentrated to obtain 6.0 g of crude product (compound 5e), with a yield of 65%.
MSm/z(ESI):1167.5[M/2+1]MSm/z(ESI):1167.5[M/2+1]
6.化合物5f的合成6. Synthesis of compound 5f
将化合物5e(4.6g,2mmol)溶于20mLDCM和5mLDMSO中,再加入化合物MMAE(0.7g,1mmol)和DMAP(0.2g,1.5mmol),室温下搅拌2h,HPLC显示反应完全。将反应体系加入50mLDCM和50mL饱和硫酸氢钠水溶液,有机相用50mL饱和氯化钠溶液洗涤一次,无水硫酸钠干燥,浓缩后得到粗品,经高压制备分离得到黄色固体1.9g(化合物5f),产率65%。Compound 5e (4.6g, 2mmol) was dissolved in 20mL DCM and 5mL DMSO, then compound MMAE (0.7g, 1mmol) and DMAP (0.2g, 1.5mmol) were added, stirred at room temperature for 2h, HPLC showed that the reaction was complete. The reaction system was added 50mL DCM and 50mL saturated aqueous sodium bisulfate solution, the organic phase was washed once with 50mL saturated sodium chloride solution, dried over anhydrous sodium sulfate, and concentrated to obtain a crude product, which was separated by high-pressure preparation to obtain 1.9 g of a yellow solid (compound 5f). Yield 65%.
MSm/z(ESI):1456.8[M/2+1]MSm/z(ESI):1456.8[M/2+1]
7.化合物5g的合成7. Synthesis of Compound 5g
将SN38(5.5g,14mmol)(购自湖北信康,≥95%)溶于50mLDCM中,冰水浴降温至0-5℃,依次加入对硝基氯甲酸苯酯(3.3g,16mmol)和DMAP(2.89g,24mmol),自然至室温搅拌2h,TLC显示反应完全后。分别用饱和硫酸氢钠溶液50mL和NaCl50mL洗涤一次,有机相用无水硫酸钠干燥,浓缩得到粗品15g。硅胶柱纯化(DCM/EA20/1-1/1)得到黄色固体4.6g(化合物5g),产率60%。SN38 (5.5g, 14mmol) (purchased from Hubei Xinkang, ≥95%) was dissolved in 50mL DCM, cooled to 0-5°C in an ice-water bath, and p-nitrochloroformate phenyl (3.3g, 16mmol) and DMAP ( 2.89g, 24mmol), naturally to room temperature and stirred for 2h, TLC showed that the reaction was complete. Wash once with 50 mL of saturated sodium bisulfate solution and 50 mL of NaCl, dry the organic phase with anhydrous sodium sulfate, and concentrate to obtain 15 g of crude product. Silica gel column purification (DCM/EA20/1-1/1) gave 4.6 g of yellow solid (compound 5 g), yield 60%.
MSm/z(ESI):558.1[M+1]MSm/z(ESI):558.1[M+1]
8.化合物5h的合成8. Synthesis of compound 5h
将化合物5g(4.5g,8mmol)溶于30mLDCM和5mLDMSO中,再加入N,N'-二甲基乙二胺(0.8g,9.0mmol)和DMAP(1.07g,8.7mmol),室温下搅拌1h,HPLC显示反应完全。将反应体系加入100mLDCM和50mL饱和硫酸氢钠水溶液,有机相用50mL饱和氯化钠溶液洗涤一次,无水硫酸钠干燥,浓缩,柱层析分离得到黄色固体2.3g(化合物5h),产率58%。Compound 5g (4.5g, 8mmol) was dissolved in 30mLDCM and 5mLDMSO, then N,N'-dimethylethylenediamine (0.8g, 9.0mmol) and DMAP (1.07g, 8.7mmol) were added, and stirred at room temperature for 1h , HPLC showed that the reaction was complete. The reaction system was added 100mL DCM and 50mL saturated aqueous sodium bisulfate solution, the organic phase was washed once with 50mL saturated sodium chloride solution, dried over anhydrous sodium sulfate, concentrated, and separated by column chromatography to obtain 2.3g of a yellow solid (compound 5h), yield 58 %.
MSm/z(ESI):507.2[M+1]MSm/z(ESI):507.2[M+1]
9.化合物5的合成9. Synthesis of Compound 5
将化合物5f(1.4g,0.5mmol)溶于10mLDCM和2mLDMSO中,再加入化合物5h(0.3g,0.6mmol)和DMAP(0.1g,1.0mmol),室温下搅拌2h,HPLC显示反应完全。将反应体系加入20mLDCM和20mL饱和硫酸氢钠水溶液,有机相用20mL饱和氯化钠溶液洗涤一次,无水硫酸钠干燥,浓缩后得到粗品,经高压制备分离得到黄色固体700mg(化合物5),产率47%。Compound 5f (1.4g, 0.5mmol) was dissolved in 10mL DCM and 2mL DMSO, then compound 5h (0.3g, 0.6mmol) and DMAP (0.1g, 1.0mmol) were added, stirred at room temperature for 2h, HPLC showed that the reaction was complete. The reaction system was added 20mL DCM and 20mL saturated aqueous sodium bisulfate solution, the organic phase was washed once with 20mL saturated sodium chloride solution, dried over anhydrous sodium sulfate, and concentrated to obtain a crude product, which was separated by high-pressure preparation to obtain 700 mg of a yellow solid (compound 5). rate 47%.
MSm/z(ESI):1554.3[M/2+1]MSm/z(ESI):1554.3[M/2+1]
实施例6 ADC的制备The preparation of embodiment 6 ADC
6.1 mAb与PEG12反应过程6.1 Reaction process of mAb and PEG12
按照mAb与N3-PEG12-SPA的摩尔比为1:20的投料比进行反应。先量取208μL的mAb溶液(浓度:9.6mg/mL)置于1.5mL的离心管中,则量取的mAb为2mg。使用5mMpH3.0的PBS溶液配制10mg/mL的N3-PEG12-SPA溶液,量取20μL加入到离心管中,充分混合,震荡反应2h。反应完毕后用超滤的方式去除未反应的N3-PEG12-SPA。The reaction was carried out according to the molar ratio of mAb and N 3 -PEG12-SPA being 1:20. First measure 208 μL of mAb solution (concentration: 9.6 mg/mL) and place it in a 1.5 mL centrifuge tube, then the measured mAb is 2 mg. A 10 mg/mL N 3 -PEG12-SPA solution was prepared using 5 mM PBS solution at pH 3.0, and 20 μL was added to a centrifuge tube, mixed thoroughly, and oscillated for 2 hours. After the reaction, the unreacted N 3 -PEG12-SPA was removed by ultrafiltration.
mAb为EGFR单抗-耐昔妥珠单抗(Necitumumab),购自礼来(EliLilly)公司mAb is EGFR monoclonal antibody-Necitumumab (Necitumumab), purchased from Eli Lilly (EliLilly)
N3-PEG12-SPA为自制,纯度95%,MSm/z(ESI):741.4[M+1]。N 3 -PEG12-SPA is self-made, with a purity of 95%, MSm/z (ESI): 741.4[M+1].
6.2 mAb-PEG12与linker-drug反应过程6.2 Reaction process of mAb-PEG12 and linker-drug
取制备好的mAb-PEG12样品,测定蛋白浓度为2.05mg/mL,量取490μL样品溶液置于1.5mL的离心管中。称量10mg linker-drug,溶解在DMSO中,制备成1mg/mL的溶液。按照mAb与linker-drug投料比为1:4计算,量取41μL linker-drug溶液加入到离心管中,再向离心管中加入510μL的PBS和59μL的DMSO,保持蛋白浓度为1mg/mL,水与DMSO的比例为10:1,充分混合,震荡反应2h。反应完毕后用超滤的方式去除未反应的小分子,得到ADC分子,如下表所示。Take the prepared mAb-PEG12 sample, determine the protein concentration to be 2.05 mg/mL, measure 490 μL of the sample solution and place it in a 1.5 mL centrifuge tube. Weigh 10 mg linker-drug, dissolve in DMSO, and prepare a 1 mg/mL solution. According to the calculation that the ratio of mAb to linker-drug is 1:4, measure 41 μL of linker-drug solution and add it to the centrifuge tube, then add 510 μL of PBS and 59 μL of DMSO to the centrifuge tube to keep the protein concentration at 1 mg/mL, water The ratio with DMSO is 10:1, mix well, and shake for 2 hours. After the reaction was completed, unreacted small molecules were removed by ultrafiltration to obtain ADC molecules, as shown in the table below.
表1 Linker-drug的种类以及对应的ADC分子的名称Table 1 Types of Linker-drugs and names of corresponding ADC molecules
其中,化合物1为实施例1制备,化合物4为实施例4制备,DBCO-VC-PAB-SN38,为自制,纯度90%,MSm/z(ESI):1087.4[M+1]Among them, compound 1 was prepared in Example 1, compound 4 was prepared in Example 4, DBCO-VC-PAB-SN38, self-made, purity 90%, MSm/z (ESI): 1087.4 [M+1]
DBCO-VC-PAB-SN38结构式如下图所示:The structural formula of DBCO-VC-PAB-SN38 is shown in the figure below:
实施例7 DAR值的测量The measurement of embodiment 7 DAR value
用液相色谱方法检测实施例6中的ADC的DAR值。The DAR value of the ADC in Example 6 was detected by liquid chromatography.
液相色谱设置如下:The LC settings were as follows:
表2液相色谱条件Table 2 Liquid Chromatography Conditions
溶解实施例6制备的ADC1、ADC2、ADC3,按照表2中的液相色谱条件对ADC1、ADC2、ADC3进行检测,该液相色谱条件下SN38与ADC1、ADC2、ADC3分离度大于1.5。制备不同浓度标准曲线,建立SN38峰面积与浓度之间的线性标准曲线,分别对ADC1、ADC2和ADC3上的SN38进行定量。根据ADC的抗体浓度和SN38的浓度计算ADC上SN38的偶联个数。测定实施例6制备的ADC1的DAR值为8,ADC2的DAR值为8,ADC3的DAR值为4,ADC1和ADC2的DAR值高于ADC3,实验结果表明,相较于传统该发明获得的ADC1和ADC2的DAR值更高。Dissolve the ADC1, ADC2, and ADC3 prepared in Example 6, and detect ADC1, ADC2, and ADC3 according to the liquid chromatography conditions in Table 2. Under the liquid chromatography conditions, the separation between SN38 and ADC1, ADC2, and ADC3 is greater than 1.5. Prepare standard curves of different concentrations, establish a linear standard curve between the peak area of SN38 and the concentration, and quantify SN38 on ADC1, ADC2 and ADC3, respectively. According to the antibody concentration of ADC and the concentration of SN38, the conjugation number of SN38 on ADC was calculated. The DAR value of ADC1 prepared in Example 6 is 8, the DAR value of ADC2 is 8, the DAR value of ADC3 is 4, and the DAR value of ADC1 and ADC2 is higher than that of ADC3. The experimental results show that compared with the traditional ADC1 obtained by this invention and ADC2 have higher DAR values.
实施例8 ADC与EGFR的亲和力测定Example 8 Affinity Determination of ADC and EGFR
ADC药物的亲和力用SPR的方法进行检测,试验仪器为GE公司的产品biacore。The affinity of the ADC drug is detected by the method of SPR, and the test instrument is biacore, a product of GE Company.
简单操作步骤为:将EGFR抗原偶联到CM芯片上。用不同浓度的ADC药物或单抗药物测试ADC药物的抗体或游离的抗体与抗原的亲和力。The simple operation steps are: coupling the EGFR antigen to the CM chip. Use different concentrations of ADC drugs or monoclonal antibody drugs to test the affinity of ADC drug antibodies or free antibodies to antigens.
结果显示实施例6制备的ADC1、ADC2和ADC3,其抗体的亲和力下降不是很明显,ADC1和ADC2的亲和力高于ADC3。The results showed that ADC1, ADC2 and ADC3 prepared in Example 6, the affinity decrease of the antibodies was not obvious, and the affinity of ADC1 and ADC2 was higher than that of ADC3.
表3实验结果:Table 3 Experimental results:
实施例9体外细胞毒性实验Example 9 In Vitro Cytotoxicity Experiment
用BXPC-3(人胰腺癌细胞)细胞进行体外细胞毒性测试。试验操作如下:In vitro cytotoxicity tests were performed with BXPC-3 (human pancreatic cancer cell) cells. The test operation is as follows:
1、96孔板每孔加入细胞100μL(留2个空白组不加细胞,加入同体积的培养基)。细胞置于37℃的5%CO2细胞培养箱中培养24h。细胞毒性实验每孔加入100μL约含10000个细胞。1. Add 100 μL of cells to each well of the 96-well plate (keep 2 blank groups without adding cells, and add the same volume of medium). The cells were cultured in a 5% CO 2 incubator at 37°C for 24 h. For cytotoxicity experiments, add 100 μL to each well to contain about 10,000 cells.
2、每孔加入10μL不同浓度ADC或SN38的药物。2. Add 10 μL of ADC or SN38 drugs with different concentrations to each well.
3、将96孔板在37℃,含5%CO2空气及100%湿度的细胞培养箱中孵育24小时。3. Incubate the 96-well plate at 37°C for 24 hours in a cell culture incubator containing 5% CO 2 air and 100% humidity.
4、每孔加入10μL的CCK-8溶液。37℃,5%CO2培养箱中孵育3h。4. Add 10 μL of CCK-8 solution to each well. Incubate for 3 h at 37°C in a 5% CO 2 incubator.
5、酶标仪测定450nm处的吸光度。5. Measure the absorbance at 450nm with a microplate reader.
6、结果分析:6. Result analysis:
A.细胞存活率:将各测试孔的OD值减去本底OD值(空白组),各重复孔的OD值取平均数±SD。A. Cell survival rate: subtract the background OD value (blank group) from the OD value of each test well, and take the mean ± SD of the OD value of each repeated well.
细胞存活率%=(加药细胞OD/对照细胞OD)×100%。Cell viability%=(OD of drug-added cells/OD of control cells)×100%.
B.求出T/C=50%时的药物浓度(IC50)及T/C=10%时的药物浓度(IC90)。B. Determine the drug concentration (IC50) when T/C=50% and the drug concentration (IC90) when T/C=10%.
结果显示实施例6制备的ADC1的IC50值小于ADC2的IC50值,均小于SN38的IC50值。The results showed that the IC50 value of ADC1 prepared in Example 6 was less than that of ADC2, and both were less than the IC50 value of SN38.
表4细胞毒性结果:Table 4 Cytotoxicity Results:
结果表明,该发明获得的ADC1和ADC2不仅能够有效地发挥EGFR抗体部分的生物学功能,还兼有SN38对肿瘤细胞的高效杀伤活性。相较于传统的直线型linker制备得到的ADC3,本发明得到的具有分支型linker的ADC1和ADC2具有更优的活性。The results show that the ADC1 and ADC2 obtained by the invention can not only effectively play the biological function of the EGFR antibody part, but also have the high-efficiency killing activity of SN38 on tumor cells. Compared with ADC3 prepared by traditional linear linker, ADC1 and ADC2 with branched linker obtained in the present invention have better activity.
最后应说明的是:以上各实施例仅用以说明本发明的技术方案,而非对其限制;尽管参照前述各实施例对本发明进行了详细的说明,但本领域的普通技术人员应当理解:其依然可以对前述各实施例所记载的技术方案进行修改,或者对其中部分或者全部技术特征进行等同替换;而这些修改或者替换,并不使相应技术方案的本质脱离本发明各实施例技术方案的范围。It should be noted that at last: above each embodiment is only in order to illustrate technical scheme of the present invention, and is not intended to limit; Although the present invention has been described in detail with reference to foregoing each embodiment, those of ordinary skill in the art should understand that: It is still possible to modify the technical solutions described in the foregoing embodiments, or perform equivalent replacements for some or all of the technical features; and these modifications or replacements do not make the essence of the corresponding technical solutions deviate from the technical solutions of the various embodiments of the present invention. range.
Claims (14)
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202211129102.4A CN115505028A (en) | 2022-09-16 | 2022-09-16 | Branched polyethylene glycol linker and application thereof |
| PCT/CN2023/117323 WO2024055886A1 (en) | 2022-09-16 | 2023-09-06 | Branched polyethylene glycol linker and use thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202211129102.4A CN115505028A (en) | 2022-09-16 | 2022-09-16 | Branched polyethylene glycol linker and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN115505028A true CN115505028A (en) | 2022-12-23 |
Family
ID=84503535
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202211129102.4A Pending CN115505028A (en) | 2022-09-16 | 2022-09-16 | Branched polyethylene glycol linker and application thereof |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN115505028A (en) |
| WO (1) | WO2024055886A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024055886A1 (en) * | 2022-09-16 | 2024-03-21 | 辽宁键凯科技有限公司 | Branched polyethylene glycol linker and use thereof |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014177042A1 (en) * | 2013-04-28 | 2014-11-06 | Qin Gang | Novel linker and preparation method thereof |
| WO2015155753A2 (en) * | 2015-08-10 | 2015-10-15 | Suzhou M-Conj Biotech Co., Ltd | Novel linkers and their uses in specific conjugation of drugs to a biological molecule |
| CN107469089A (en) * | 2016-06-07 | 2017-12-15 | 北京键凯科技股份有限公司 | A kind of PEG connexons and aglucon drug conjugates |
| US20200215206A1 (en) * | 2016-02-04 | 2020-07-09 | Suzhou M-Conj Biotech Co., Ltd. | Specific conjugation linkers, specific immunoconjugates thereof, methods of making and uses such conjugates thereof |
| CN113382985A (en) * | 2018-11-20 | 2021-09-10 | 里珍纳龙药品有限公司 | Bis-octahydrophenanthrenecarboxamide derivatives and protein conjugates thereof as LXR agonists |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103083680B (en) * | 2011-11-07 | 2014-12-24 | 北京键凯科技有限公司 | Polyethylene glycol (PEG)-amino acid oligopeptide-irinotecan combo and its medicinal composition |
| EP3524631B1 (en) * | 2016-10-07 | 2025-12-03 | Institute Of Science Tokyo | Branched type hetero monodispersed polyethylene glycol, production method thereof, and conjugate thereof |
| BR112022020124A2 (en) * | 2020-04-15 | 2022-11-29 | Shenzhen Enduring Biotech Ltd | ANTIBODY-DRUG CONJUGATE |
| CN115505028A (en) * | 2022-09-16 | 2022-12-23 | 辽宁键凯科技有限公司 | Branched polyethylene glycol linker and application thereof |
-
2022
- 2022-09-16 CN CN202211129102.4A patent/CN115505028A/en active Pending
-
2023
- 2023-09-06 WO PCT/CN2023/117323 patent/WO2024055886A1/en not_active Ceased
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014177042A1 (en) * | 2013-04-28 | 2014-11-06 | Qin Gang | Novel linker and preparation method thereof |
| WO2015155753A2 (en) * | 2015-08-10 | 2015-10-15 | Suzhou M-Conj Biotech Co., Ltd | Novel linkers and their uses in specific conjugation of drugs to a biological molecule |
| US20200215206A1 (en) * | 2016-02-04 | 2020-07-09 | Suzhou M-Conj Biotech Co., Ltd. | Specific conjugation linkers, specific immunoconjugates thereof, methods of making and uses such conjugates thereof |
| CN107469089A (en) * | 2016-06-07 | 2017-12-15 | 北京键凯科技股份有限公司 | A kind of PEG connexons and aglucon drug conjugates |
| CN113382985A (en) * | 2018-11-20 | 2021-09-10 | 里珍纳龙药品有限公司 | Bis-octahydrophenanthrenecarboxamide derivatives and protein conjugates thereof as LXR agonists |
Non-Patent Citations (1)
| Title |
|---|
| AIKO YAMAGUCHI ET AL.: "Chemical generation of small molecule-based bispecific antibody-drug conjugates for broadening the target scope", BIOORGANIC & MEDICINAL CHEMISTRY, vol. 32, 9 January 2021 (2021-01-09), pages 1 - 9, XP086472983, DOI: 10.1016/j.bmc.2021.116013 * |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024055886A1 (en) * | 2022-09-16 | 2024-03-21 | 辽宁键凯科技有限公司 | Branched polyethylene glycol linker and use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2024055886A1 (en) | 2024-03-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN118434765A (en) | BCMA monoclonal antibodies and antibody-drug conjugates | |
| CN120248020B (en) | Bioactive substance conjugate and its preparation method and use | |
| CN110099682B (en) | Coupled connector, cell binding molecule-drug conjugate containing same, and preparation and application thereof | |
| JPWO2023078021A5 (en) | ||
| CN113195487A (en) | 2,3-Diaminosuccinyl conjugate linker | |
| AU2018368520B2 (en) | Ligand-drug-conjugates as substrates for selective cleavage by the exopeptidase activity of Cathepsin B | |
| CN115335370A (en) | Fibroblast activation protein ligands for targeted delivery applications | |
| CN119630672B (en) | Camptothecin derivatives and their conjugates, their preparation methods and pharmaceutical uses | |
| IL304960B1 (en) | Bivalent fibroblast activation protein ligands for targeted delivery applications | |
| WO2025011419A1 (en) | Linker-drug molecule and antibody drug conjugate, preparation method therefor and use thereof | |
| WO2024055886A1 (en) | Branched polyethylene glycol linker and use thereof | |
| CN120835792A (en) | Antibody-drug conjugates for targeted therapy of prostate cancer and other tumors | |
| CN118510515A (en) | Pyridazindione-based double-heterocycle covalent linker, method and application thereof | |
| US20250249113A1 (en) | Ligand-drug-conjugates with improved pharmacokinetic and drug release properties | |
| WO2024133845A1 (en) | Conjugates comprising a phosphorus(v) moiety and a drug | |
| KR20250108120A (en) | Anti-BCAM antibodies and conjugates thereof | |
| WO2026006506A1 (en) | Two-step drug delivery system | |
| KR20250174687A (en) | Antibody-drug conjugates for delivering cytotoxic agents | |
| HK40120540A (en) | Linker-drug molecule and antibody drug conjugate, preparation method therefor and use thereof | |
| EA045708B1 (en) | NEW PEPTIDE LINKERS AND CONJUGATES BASED ON CRYPTOPHICIN, THEIR PREPARATION AND THERAPEUTIC APPLICATION | |
| NZ744940B2 (en) | Conjugation linkers, antibody-drug conjugates thereof, and methods of synthesis and use of such conjugates |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |