CN114957132A - 含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 - Google Patents
含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 Download PDFInfo
- Publication number
- CN114957132A CN114957132A CN202110193453.0A CN202110193453A CN114957132A CN 114957132 A CN114957132 A CN 114957132A CN 202110193453 A CN202110193453 A CN 202110193453A CN 114957132 A CN114957132 A CN 114957132A
- Authority
- CN
- China
- Prior art keywords
- compound
- compound shown
- cancer
- tert
- butyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- -1 aminobenzamide pyridazinone compound Chemical class 0.000 title claims abstract description 21
- 239000008194 pharmaceutical composition Substances 0.000 title claims abstract description 12
- 238000002360 preparation method Methods 0.000 title claims abstract description 10
- 150000001875 compounds Chemical class 0.000 claims abstract description 126
- 102000003964 Histone deacetylase Human genes 0.000 claims abstract description 23
- 108090000353 Histone deacetylase Proteins 0.000 claims abstract description 23
- 150000003839 salts Chemical class 0.000 claims abstract description 16
- 230000000694 effects Effects 0.000 claims description 20
- 206010028980 Neoplasm Diseases 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 14
- 239000003814 drug Substances 0.000 claims description 11
- 208000031261 Acute myeloid leukaemia Diseases 0.000 claims description 10
- WPYMKLBDIGXBTP-UHFFFAOYSA-N Benzoic acid Natural products OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 claims description 8
- 201000003793 Myelodysplastic syndrome Diseases 0.000 claims description 8
- 201000010099 disease Diseases 0.000 claims description 8
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 8
- 230000014509 gene expression Effects 0.000 claims description 8
- 238000011282 treatment Methods 0.000 claims description 8
- 201000011510 cancer Diseases 0.000 claims description 7
- 229910052760 oxygen Inorganic materials 0.000 claims description 7
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 7
- 208000033776 Myeloid Acute Leukemia Diseases 0.000 claims description 6
- 230000002159 abnormal effect Effects 0.000 claims description 6
- 238000006482 condensation reaction Methods 0.000 claims description 6
- 229910052736 halogen Inorganic materials 0.000 claims description 6
- 150000002367 halogens Chemical class 0.000 claims description 6
- 239000005711 Benzoic acid Substances 0.000 claims description 5
- 206010025323 Lymphomas Diseases 0.000 claims description 5
- 235000010233 benzoic acid Nutrition 0.000 claims description 5
- 229910052739 hydrogen Inorganic materials 0.000 claims description 5
- 239000001257 hydrogen Substances 0.000 claims description 5
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 5
- 201000001441 melanoma Diseases 0.000 claims description 5
- 229910052717 sulfur Inorganic materials 0.000 claims description 5
- 206010006187 Breast cancer Diseases 0.000 claims description 4
- 208000026310 Breast neoplasm Diseases 0.000 claims description 4
- 206010009944 Colon cancer Diseases 0.000 claims description 4
- 206010035226 Plasma cell myeloma Diseases 0.000 claims description 4
- 125000003545 alkoxy group Chemical group 0.000 claims description 4
- 125000000217 alkyl group Chemical group 0.000 claims description 4
- 125000003118 aryl group Chemical group 0.000 claims description 4
- 208000029742 colonic neoplasm Diseases 0.000 claims description 4
- 230000032050 esterification Effects 0.000 claims description 4
- 238000005886 esterification reaction Methods 0.000 claims description 4
- 125000005842 heteroatom Chemical group 0.000 claims description 4
- 208000025113 myeloid leukemia Diseases 0.000 claims description 4
- 238000006722 reduction reaction Methods 0.000 claims description 4
- YICPBKWYZXFJNB-UHFFFAOYSA-N 3-chloro-1h-pyridazin-6-one Chemical compound OC1=CC=C(Cl)N=N1 YICPBKWYZXFJNB-UHFFFAOYSA-N 0.000 claims description 3
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 claims description 3
- ZKCFKELMWBDKST-SECBINFHSA-N C[C@H](C(C=C1)=CC=C1C(OC(C)(C)C)=O)O Chemical compound C[C@H](C(C=C1)=CC=C1C(OC(C)(C)C)=O)O ZKCFKELMWBDKST-SECBINFHSA-N 0.000 claims description 3
- 102000004190 Enzymes Human genes 0.000 claims description 3
- 108090000790 Enzymes Proteins 0.000 claims description 3
- 206010061218 Inflammation Diseases 0.000 claims description 3
- 208000008839 Kidney Neoplasms Diseases 0.000 claims description 3
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 3
- 208000034578 Multiple myelomas Diseases 0.000 claims description 3
- 206010038389 Renal cancer Diseases 0.000 claims description 3
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 3
- KHYAFFAGZNCWPT-UHFFFAOYSA-N boron;n,n-diethylaniline Chemical compound [B].CCN(CC)C1=CC=CC=C1 KHYAFFAGZNCWPT-UHFFFAOYSA-N 0.000 claims description 3
- 239000003937 drug carrier Substances 0.000 claims description 3
- 206010017758 gastric cancer Diseases 0.000 claims description 3
- 230000004054 inflammatory process Effects 0.000 claims description 3
- 201000010982 kidney cancer Diseases 0.000 claims description 3
- 208000032839 leukemia Diseases 0.000 claims description 3
- 201000007270 liver cancer Diseases 0.000 claims description 3
- 208000014018 liver neoplasm Diseases 0.000 claims description 3
- 201000005202 lung cancer Diseases 0.000 claims description 3
- 208000020816 lung neoplasm Diseases 0.000 claims description 3
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 3
- 208000025638 primary cutaneous T-cell non-Hodgkin lymphoma Diseases 0.000 claims description 3
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 3
- 201000011549 stomach cancer Diseases 0.000 claims description 3
- 125000001424 substituent group Chemical group 0.000 claims description 3
- QXQXBZLXIAUIBG-UHFFFAOYSA-N tert-butyl 4-acetylbenzoate Chemical compound CC(=O)C1=CC=C(C(=O)OC(C)(C)C)C=C1 QXQXBZLXIAUIBG-UHFFFAOYSA-N 0.000 claims description 3
- LYDRKKWPKKEMNZ-UHFFFAOYSA-N tert-butyl benzoate Chemical compound CC(C)(C)OC(=O)C1=CC=CC=C1 LYDRKKWPKKEMNZ-UHFFFAOYSA-N 0.000 claims description 3
- 208000024893 Acute lymphoblastic leukemia Diseases 0.000 claims description 2
- 208000014697 Acute lymphocytic leukaemia Diseases 0.000 claims description 2
- 206010000871 Acute monocytic leukaemia Diseases 0.000 claims description 2
- 208000032791 BCR-ABL1 positive chronic myelogenous leukemia Diseases 0.000 claims description 2
- 208000010833 Chronic myeloid leukaemia Diseases 0.000 claims description 2
- 201000010374 Down Syndrome Diseases 0.000 claims description 2
- 208000017604 Hodgkin disease Diseases 0.000 claims description 2
- 208000021519 Hodgkin lymphoma Diseases 0.000 claims description 2
- 208000010747 Hodgkins lymphoma Diseases 0.000 claims description 2
- 208000031671 Large B-Cell Diffuse Lymphoma Diseases 0.000 claims description 2
- 208000025205 Mantle-Cell Lymphoma Diseases 0.000 claims description 2
- 208000035490 Megakaryoblastic Acute Leukemia Diseases 0.000 claims description 2
- 208000033761 Myelogenous Chronic BCR-ABL Positive Leukemia Diseases 0.000 claims description 2
- 206010033128 Ovarian cancer Diseases 0.000 claims description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 2
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 2
- 208000006664 Precursor Cell Lymphoblastic Leukemia-Lymphoma Diseases 0.000 claims description 2
- 206010036711 Primary mediastinal large B-cell lymphomas Diseases 0.000 claims description 2
- 208000015634 Rectal Neoplasms Diseases 0.000 claims description 2
- 238000006069 Suzuki reaction reaction Methods 0.000 claims description 2
- 208000029052 T-cell acute lymphoblastic leukemia Diseases 0.000 claims description 2
- 208000000389 T-cell leukemia Diseases 0.000 claims description 2
- 150000008065 acid anhydrides Chemical class 0.000 claims description 2
- 208000013593 acute megakaryoblastic leukemia Diseases 0.000 claims description 2
- 208000020700 acute megakaryocytic leukemia Diseases 0.000 claims description 2
- 239000003054 catalyst Substances 0.000 claims description 2
- 206010012601 diabetes mellitus Diseases 0.000 claims description 2
- 206010012818 diffuse large B-cell lymphoma Diseases 0.000 claims description 2
- 125000000623 heterocyclic group Chemical group 0.000 claims description 2
- 201000004792 malaria Diseases 0.000 claims description 2
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 2
- 230000004770 neurodegeneration Effects 0.000 claims description 2
- 208000015122 neurodegenerative disease Diseases 0.000 claims description 2
- 201000002528 pancreatic cancer Diseases 0.000 claims description 2
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 2
- 238000011321 prophylaxis Methods 0.000 claims description 2
- 125000006239 protecting group Chemical group 0.000 claims description 2
- 125000004076 pyridyl group Chemical group 0.000 claims description 2
- 206010038038 rectal cancer Diseases 0.000 claims description 2
- 201000001275 rectum cancer Diseases 0.000 claims description 2
- 206010044688 Trisomy 21 Diseases 0.000 claims 1
- 230000006197 histone deacetylation Effects 0.000 claims 1
- 230000002401 inhibitory effect Effects 0.000 abstract description 17
- 230000035755 proliferation Effects 0.000 abstract description 11
- 210000004881 tumor cell Anatomy 0.000 abstract description 10
- 230000027455 binding Effects 0.000 abstract description 7
- 238000000338 in vitro Methods 0.000 abstract description 5
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 23
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 22
- 239000000243 solution Substances 0.000 description 22
- 239000002904 solvent Substances 0.000 description 20
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 18
- 210000004027 cell Anatomy 0.000 description 14
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 13
- 238000006243 chemical reaction Methods 0.000 description 13
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 12
- 238000012360 testing method Methods 0.000 description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 12
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 11
- 238000005481 NMR spectroscopy Methods 0.000 description 10
- 229950009221 chidamide Drugs 0.000 description 10
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 10
- INVTYAOGFAGBOE-UHFFFAOYSA-N entinostat Chemical compound NC1=CC=CC=C1NC(=O)C(C=C1)=CC=C1CNC(=O)OCC1=CC=CN=C1 INVTYAOGFAGBOE-UHFFFAOYSA-N 0.000 description 10
- WXHHICFWKXDFOW-BJMVGYQFSA-N n-(2-amino-5-fluorophenyl)-4-[[[(e)-3-pyridin-3-ylprop-2-enoyl]amino]methyl]benzamide Chemical compound NC1=CC=C(F)C=C1NC(=O)C(C=C1)=CC=C1CNC(=O)\C=C\C1=CC=CN=C1 WXHHICFWKXDFOW-BJMVGYQFSA-N 0.000 description 10
- 230000000144 pharmacologic effect Effects 0.000 description 10
- 239000007787 solid Substances 0.000 description 10
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 9
- 239000012071 phase Substances 0.000 description 9
- 238000003756 stirring Methods 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 8
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 8
- 229940079593 drug Drugs 0.000 description 8
- 235000019439 ethyl acetate Nutrition 0.000 description 8
- 239000003276 histone deacetylase inhibitor Substances 0.000 description 8
- 230000005764 inhibitory process Effects 0.000 description 7
- 229940002612 prodrug Drugs 0.000 description 7
- 239000000651 prodrug Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 6
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 239000000460 chlorine Substances 0.000 description 6
- 238000004440 column chromatography Methods 0.000 description 6
- 238000003786 synthesis reaction Methods 0.000 description 6
- 238000012447 xenograft mouse model Methods 0.000 description 6
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical group C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 5
- 102100039996 Histone deacetylase 1 Human genes 0.000 description 5
- 102100039999 Histone deacetylase 2 Human genes 0.000 description 5
- 102100021455 Histone deacetylase 3 Human genes 0.000 description 5
- 101001035024 Homo sapiens Histone deacetylase 1 Proteins 0.000 description 5
- 101001035011 Homo sapiens Histone deacetylase 2 Proteins 0.000 description 5
- 101000899282 Homo sapiens Histone deacetylase 3 Proteins 0.000 description 5
- JICXKULPHWYADC-UHFFFAOYSA-N NC(C=NNC1=O)=C1NC(C1=CC=CC=C1)=O Chemical class NC(C=NNC1=O)=C1NC(C1=CC=CC=C1)=O JICXKULPHWYADC-UHFFFAOYSA-N 0.000 description 5
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 5
- 230000004663 cell proliferation Effects 0.000 description 5
- 239000012043 crude product Substances 0.000 description 5
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- 239000000203 mixture Substances 0.000 description 5
- 239000012074 organic phase Substances 0.000 description 5
- 230000003285 pharmacodynamic effect Effects 0.000 description 5
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 5
- AAILEWXSEQLMNI-UHFFFAOYSA-N 1h-pyridazin-6-one Chemical group OC1=CC=CN=N1 AAILEWXSEQLMNI-UHFFFAOYSA-N 0.000 description 4
- FPQQSJJWHUJYPU-UHFFFAOYSA-N 3-(dimethylamino)propyliminomethylidene-ethylazanium;chloride Chemical compound Cl.CCN=C=NCCCN(C)C FPQQSJJWHUJYPU-UHFFFAOYSA-N 0.000 description 4
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 4
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 4
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 4
- 208000027190 Peripheral T-cell lymphomas Diseases 0.000 description 4
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 4
- 208000031672 T-Cell Peripheral Lymphoma Diseases 0.000 description 4
- 125000004429 atom Chemical group 0.000 description 4
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 4
- 229910052799 carbon Inorganic materials 0.000 description 4
- 230000002255 enzymatic effect Effects 0.000 description 4
- 208000020968 mature T-cell and NK-cell non-Hodgkin lymphoma Diseases 0.000 description 4
- 229910052757 nitrogen Inorganic materials 0.000 description 4
- 239000001301 oxygen Substances 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 3
- PXBFMLJZNCDSMP-UHFFFAOYSA-N 2-Aminobenzamide Chemical class NC(=O)C1=CC=CC=C1N PXBFMLJZNCDSMP-UHFFFAOYSA-N 0.000 description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 3
- NCNRHFGMJRPRSK-MDZDMXLPSA-N belinostat Chemical compound ONC(=O)\C=C\C1=CC=CC(S(=O)(=O)NC=2C=CC=CC=2)=C1 NCNRHFGMJRPRSK-MDZDMXLPSA-N 0.000 description 3
- 125000004432 carbon atom Chemical group C* 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 239000000706 filtrate Substances 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- GBMDVOWEEQVZKZ-UHFFFAOYSA-N methanol;hydrate Chemical compound O.OC GBMDVOWEEQVZKZ-UHFFFAOYSA-N 0.000 description 3
- FPOHNWQLNRZRFC-ZHACJKMWSA-N panobinostat Chemical compound CC=1NC2=CC=CC=C2C=1CCNCC1=CC=C(\C=C\C(=O)NO)C=C1 FPOHNWQLNRZRFC-ZHACJKMWSA-N 0.000 description 3
- 229960005184 panobinostat Drugs 0.000 description 3
- 229910052698 phosphorus Inorganic materials 0.000 description 3
- 239000011574 phosphorus Substances 0.000 description 3
- OHRURASPPZQGQM-GCCNXGTGSA-N romidepsin Chemical compound O1C(=O)[C@H](C(C)C)NC(=O)C(=C/C)/NC(=O)[C@H]2CSSCC\C=C\[C@@H]1CC(=O)N[C@H](C(C)C)C(=O)N2 OHRURASPPZQGQM-GCCNXGTGSA-N 0.000 description 3
- 229960003452 romidepsin Drugs 0.000 description 3
- OHRURASPPZQGQM-UHFFFAOYSA-N romidepsin Natural products O1C(=O)C(C(C)C)NC(=O)C(=CC)NC(=O)C2CSSCCC=CC1CC(=O)NC(C(C)C)C(=O)N2 OHRURASPPZQGQM-UHFFFAOYSA-N 0.000 description 3
- 108010091666 romidepsin Proteins 0.000 description 3
- 239000000741 silica gel Substances 0.000 description 3
- 229910002027 silica gel Inorganic materials 0.000 description 3
- 239000012453 solvate Substances 0.000 description 3
- 238000010998 test method Methods 0.000 description 3
- LMDZBCPBFSXMTL-UHFFFAOYSA-N 1-Ethyl-3-(3-dimethylaminopropyl)carbodiimide Substances CCN=C=NCCCN(C)C LMDZBCPBFSXMTL-UHFFFAOYSA-N 0.000 description 2
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 description 2
- YEDUAINPPJYDJZ-UHFFFAOYSA-N 2-hydroxybenzothiazole Chemical compound C1=CC=C2SC(O)=NC2=C1 YEDUAINPPJYDJZ-UHFFFAOYSA-N 0.000 description 2
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical compound [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 2
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 2
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 2
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 2
- KRKNYBCHXYNGOX-UHFFFAOYSA-K Citrate Chemical compound [O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O KRKNYBCHXYNGOX-UHFFFAOYSA-K 0.000 description 2
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- 102000003893 Histone acetyltransferases Human genes 0.000 description 2
- 108090000246 Histone acetyltransferases Proteins 0.000 description 2
- 102100022537 Histone deacetylase 6 Human genes 0.000 description 2
- 108010033040 Histones Proteins 0.000 description 2
- 101000899330 Homo sapiens Histone deacetylase 6 Proteins 0.000 description 2
- 101001035694 Homo sapiens Polyamine deacetylase HDAC10 Proteins 0.000 description 2
- 241000699666 Mus <mouse, genus> Species 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- WWGBHDIHIVGYLZ-UHFFFAOYSA-N N-[4-[3-[[[7-(hydroxyamino)-7-oxoheptyl]amino]-oxomethyl]-5-isoxazolyl]phenyl]carbamic acid tert-butyl ester Chemical compound C1=CC(NC(=O)OC(C)(C)C)=CC=C1C1=CC(C(=O)NCCCCCCC(=O)NO)=NO1 WWGBHDIHIVGYLZ-UHFFFAOYSA-N 0.000 description 2
- 102100039388 Polyamine deacetylase HDAC10 Human genes 0.000 description 2
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 2
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 2
- 238000011579 SCID mouse model Methods 0.000 description 2
- 108010087230 Sincalide Proteins 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 108010090804 Streptavidin Proteins 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- 208000031673 T-Cell Cutaneous Lymphoma Diseases 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-M Trifluoroacetate Chemical compound [O-]C(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-M 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- 239000007983 Tris buffer Substances 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- WNLRTRBMVRJNCN-UHFFFAOYSA-N adipic acid Chemical compound OC(=O)CCCCC(O)=O WNLRTRBMVRJNCN-UHFFFAOYSA-N 0.000 description 2
- 238000011914 asymmetric synthesis Methods 0.000 description 2
- 229960003094 belinostat Drugs 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- XSIFPSYPOVKYCO-UHFFFAOYSA-N butyl benzoate Chemical compound CCCCOC(=O)C1=CC=CC=C1 XSIFPSYPOVKYCO-UHFFFAOYSA-N 0.000 description 2
- 238000010609 cell counting kit-8 assay Methods 0.000 description 2
- 201000007241 cutaneous T cell lymphoma Diseases 0.000 description 2
- 208000035250 cutaneous malignant susceptibility to 1 melanoma Diseases 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- 229940121372 histone deacetylase inhibitor Drugs 0.000 description 2
- 238000002868 homogeneous time resolved fluorescence Methods 0.000 description 2
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- PNDPGZBMCMUPRI-UHFFFAOYSA-N iodine Chemical compound II PNDPGZBMCMUPRI-UHFFFAOYSA-N 0.000 description 2
- 229910052740 iodine Inorganic materials 0.000 description 2
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 201000005962 mycosis fungoides Diseases 0.000 description 2
- CTSLXHKWHWQRSH-UHFFFAOYSA-N oxalyl chloride Chemical compound ClC(=O)C(Cl)=O CTSLXHKWHWQRSH-UHFFFAOYSA-N 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- TYJJADVDDVDEDZ-UHFFFAOYSA-M potassium hydrogencarbonate Chemical compound [K+].OC([O-])=O TYJJADVDDVDEDZ-UHFFFAOYSA-M 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- IZTQOLKUZKXIRV-YRVFCXMDSA-N sincalide Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)NCC(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](N)CC(O)=O)C1=CC=C(OS(O)(=O)=O)C=C1 IZTQOLKUZKXIRV-YRVFCXMDSA-N 0.000 description 2
- 239000000758 substrate Substances 0.000 description 2
- 238000000967 suction filtration Methods 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 2
- RIOQSEWOXXDEQQ-UHFFFAOYSA-N triphenylphosphine Chemical compound C1=CC=CC=C1P(C=1C=CC=CC=1)C1=CC=CC=C1 RIOQSEWOXXDEQQ-UHFFFAOYSA-N 0.000 description 2
- 229960000237 vorinostat Drugs 0.000 description 2
- WAEXFXRVDQXREF-UHFFFAOYSA-N vorinostat Chemical compound ONC(=O)CCCCCCC(=O)NC1=CC=CC=C1 WAEXFXRVDQXREF-UHFFFAOYSA-N 0.000 description 2
- KZPYGQFFRCFCPP-UHFFFAOYSA-N 1,1'-bis(diphenylphosphino)ferrocene Chemical compound [Fe+2].C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1.C1=CC=C[C-]1P(C=1C=CC=CC=1)C1=CC=CC=C1 KZPYGQFFRCFCPP-UHFFFAOYSA-N 0.000 description 1
- 125000005918 1,2-dimethylbutyl group Chemical group 0.000 description 1
- FLBAYUMRQUHISI-UHFFFAOYSA-N 1,8-naphthyridine Chemical group N1=CC=CC2=CC=CN=C21 FLBAYUMRQUHISI-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- VTNULXUEOJMRKZ-UHFFFAOYSA-N 3-[4-(aminomethyl)-6-(trifluoromethyl)pyridin-2-yl]oxy-N-(2H-tetrazol-5-ylmethyl)benzamide Chemical compound N=1NN=NC=1CNC(C1=CC(=CC=C1)OC1=NC(=CC(=C1)CN)C(F)(F)F)=O VTNULXUEOJMRKZ-UHFFFAOYSA-N 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- QBHDSQZASIBAAI-UHFFFAOYSA-N 4-acetylbenzoic acid Chemical compound CC(=O)C1=CC=C(C(O)=O)C=C1 QBHDSQZASIBAAI-UHFFFAOYSA-N 0.000 description 1
- GAMYYCRTACQSBR-UHFFFAOYSA-N 4-azabenzimidazole Chemical group C1=CC=C2NC=NC2=N1 GAMYYCRTACQSBR-UHFFFAOYSA-N 0.000 description 1
- 229960000549 4-dimethylaminophenol Drugs 0.000 description 1
- KWEWNOOZQVJONF-UHFFFAOYSA-N 4-fluorobenzene-1,2-diamine Chemical compound NC1=CC=C(F)C=C1N KWEWNOOZQVJONF-UHFFFAOYSA-N 0.000 description 1
- FBXGQDUVJBKEAJ-UHFFFAOYSA-N 4h-oxazin-3-one Chemical class O=C1CC=CON1 FBXGQDUVJBKEAJ-UHFFFAOYSA-N 0.000 description 1
- IGDYNWKWXUCIJB-UHFFFAOYSA-N 5-methyl-2-nitroaniline Chemical compound CC1=CC=C([N+]([O-])=O)C(N)=C1 IGDYNWKWXUCIJB-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 125000000041 C6-C10 aryl group Chemical group 0.000 description 1
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- BDAGIHXWWSANSR-UHFFFAOYSA-M Formate Chemical compound [O-]C=O BDAGIHXWWSANSR-UHFFFAOYSA-M 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-N Fumaric acid Chemical compound OC(=O)\C=C\C(O)=O VZCYOOQTPOCHFL-OWOJBTEDSA-N 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 241000238631 Hexapoda Species 0.000 description 1
- 102100039869 Histone H2B type F-S Human genes 0.000 description 1
- 102100038715 Histone deacetylase 8 Human genes 0.000 description 1
- 102000006947 Histones Human genes 0.000 description 1
- 101001035372 Homo sapiens Histone H2B type F-S Proteins 0.000 description 1
- 101001032118 Homo sapiens Histone deacetylase 8 Proteins 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- XEEYBQQBJWHFJM-UHFFFAOYSA-N Iron Chemical compound [Fe] XEEYBQQBJWHFJM-UHFFFAOYSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- OFOBLEOULBTSOW-UHFFFAOYSA-L Malonate Chemical compound [O-]C(=O)CC([O-])=O OFOBLEOULBTSOW-UHFFFAOYSA-L 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical compound CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 229910002651 NO3 Inorganic materials 0.000 description 1
- NHNBFGGVMKEFGY-UHFFFAOYSA-N Nitrate Chemical compound [O-][N+]([O-])=O NHNBFGGVMKEFGY-UHFFFAOYSA-N 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical group C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- XBDQKXXYIPTUBI-UHFFFAOYSA-M Propionate Chemical compound CCC([O-])=O XBDQKXXYIPTUBI-UHFFFAOYSA-M 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical group C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 description 1
- 210000001744 T-lymphocyte Anatomy 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical group C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 1
- 102000044209 Tumor Suppressor Genes Human genes 0.000 description 1
- 108700025716 Tumor Suppressor Genes Proteins 0.000 description 1
- 102000001742 Tumor Suppressor Proteins Human genes 0.000 description 1
- 108010040002 Tumor Suppressor Proteins Proteins 0.000 description 1
- YQYBUJYBXOVWQW-UHFFFAOYSA-N [3-[4-(aminomethyl)-6-(trifluoromethyl)pyridin-2-yl]oxyphenyl]-(3,4-dihydro-1H-isoquinolin-2-yl)methanone Chemical compound NCC1=CC(=NC(=C1)C(F)(F)F)OC=1C=C(C=CC=1)C(=O)N1CC2=CC=CC=C2CC1 YQYBUJYBXOVWQW-UHFFFAOYSA-N 0.000 description 1
- YKKPYMXANSSQCA-UHFFFAOYSA-N [3-[4-(aminomethyl)-6-(trifluoromethyl)pyridin-2-yl]oxyphenyl]-(3-pyrazol-1-ylazetidin-1-yl)methanone Chemical compound N1(N=CC=C1)C1CN(C1)C(=O)C1=CC(=CC=C1)OC1=NC(=CC(=C1)CN)C(F)(F)F YKKPYMXANSSQCA-UHFFFAOYSA-N 0.000 description 1
- 230000001594 aberrant effect Effects 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 239000001361 adipic acid Substances 0.000 description 1
- 235000011037 adipic acid Nutrition 0.000 description 1
- 239000002671 adjuvant Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 229920000615 alginic acid Polymers 0.000 description 1
- 235000010443 alginic acid Nutrition 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 235000019270 ammonium chloride Nutrition 0.000 description 1
- 230000003698 anagen phase Effects 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 239000002246 antineoplastic agent Substances 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 125000005334 azaindolyl group Chemical group N1N=C(C2=CC=CC=C12)* 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 229910052791 calcium Inorganic materials 0.000 description 1
- 229910000019 calcium carbonate Inorganic materials 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 125000002837 carbocyclic group Chemical group 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 230000022131 cell cycle Effects 0.000 description 1
- 230000010261 cell growth Effects 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 239000007810 chemical reaction solvent Substances 0.000 description 1
- 230000010428 chromatin condensation Effects 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 239000012059 conventional drug carrier Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- 230000006196 deacetylation Effects 0.000 description 1
- 238000003381 deacetylation reaction Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 150000002012 dioxanes Chemical class 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 238000009509 drug development Methods 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 239000003623 enhancer Substances 0.000 description 1
- 230000001973 epigenetic effect Effects 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-M ethanesulfonate Chemical compound CCS([O-])(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-M 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 238000001914 filtration Methods 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- QEWYKACRFQMRMB-UHFFFAOYSA-N fluoroacetic acid Chemical compound OC(=O)CF QEWYKACRFQMRMB-UHFFFAOYSA-N 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 230000006870 function Effects 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 238000002523 gelfiltration Methods 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 125000005843 halogen group Chemical group 0.000 description 1
- 208000019691 hematopoietic and lymphoid cell neoplasm Diseases 0.000 description 1
- 231100000086 high toxicity Toxicity 0.000 description 1
- 230000006195 histone acetylation Effects 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 239000012535 impurity Substances 0.000 description 1
- 238000000099 in vitro assay Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 238000009776 industrial production Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000000155 isotopic effect Effects 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 229940049920 malate Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 1
- 230000007721 medicinal effect Effects 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 125000002950 monocyclic group Chemical group 0.000 description 1
- 201000000050 myeloid neoplasm Diseases 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 239000012044 organic layer Substances 0.000 description 1
- 239000003960 organic solvent Substances 0.000 description 1
- 230000002018 overexpression Effects 0.000 description 1
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- 229940075930 picrate Drugs 0.000 description 1
- OXNIZHLAWKMVMX-UHFFFAOYSA-M picrate anion Chemical compound [O-]C1=C([N+]([O-])=O)C=C([N+]([O-])=O)C=C1[N+]([O-])=O OXNIZHLAWKMVMX-UHFFFAOYSA-M 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- VVWRJUBEIPHGQF-MDZDMXLPSA-N propan-2-yl (ne)-n-propan-2-yloxycarbonyliminocarbamate Chemical compound CC(C)OC(=O)\N=N\C(=O)OC(C)C VVWRJUBEIPHGQF-MDZDMXLPSA-N 0.000 description 1
- VVWRJUBEIPHGQF-UHFFFAOYSA-N propan-2-yl n-propan-2-yloxycarbonyliminocarbamate Chemical compound CC(C)OC(=O)N=NC(=O)OC(C)C VVWRJUBEIPHGQF-UHFFFAOYSA-N 0.000 description 1
- 238000001742 protein purification Methods 0.000 description 1
- 108090000623 proteins and genes Proteins 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- PBMFSQRYOILNGV-UHFFFAOYSA-N pyridazine Chemical group C1=CC=NN=C1 PBMFSQRYOILNGV-UHFFFAOYSA-N 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 150000003856 quaternary ammonium compounds Chemical class 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 238000010992 reflux Methods 0.000 description 1
- 238000007634 remodeling Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 239000012047 saturated solution Substances 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 125000003548 sec-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000000344 soap Substances 0.000 description 1
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 1
- 235000017557 sodium bicarbonate Nutrition 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- 238000001179 sorption measurement Methods 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000005556 structure-activity relationship Methods 0.000 description 1
- KDYFGRWQOYBRFD-UHFFFAOYSA-L succinate(2-) Chemical compound [O-]C(=O)CCC([O-])=O KDYFGRWQOYBRFD-UHFFFAOYSA-L 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 235000012222 talc Nutrition 0.000 description 1
- 229940095064 tartrate Drugs 0.000 description 1
- IBKSOWUYKOORFF-UHFFFAOYSA-N tert-butyl n-(2-amino-4-fluorophenyl)carbamate Chemical compound CC(C)(C)OC(=O)NC1=CC=C(F)C=C1N IBKSOWUYKOORFF-UHFFFAOYSA-N 0.000 description 1
- RUTRVXDBOSVFFT-UHFFFAOYSA-N tert-butyl n-(2-amino-4-methoxyphenyl)carbamate Chemical compound COC1=CC=C(NC(=O)OC(C)(C)C)C(N)=C1 RUTRVXDBOSVFFT-UHFFFAOYSA-N 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N toluene-4-sulfonic acid Chemical compound CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000009466 transformation Effects 0.000 description 1
- LENZDBCJOHFCAS-UHFFFAOYSA-N tris Chemical compound OCC(N)(CO)CO LENZDBCJOHFCAS-UHFFFAOYSA-N 0.000 description 1
- 241000701447 unidentified baculovirus Species 0.000 description 1
- 239000003981 vehicle Substances 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D237/00—Heterocyclic compounds containing 1,2-diazine or hydrogenated 1,2-diazine rings
- C07D237/02—Heterocyclic compounds containing 1,2-diazine or hydrogenated 1,2-diazine rings not condensed with other rings
- C07D237/06—Heterocyclic compounds containing 1,2-diazine or hydrogenated 1,2-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D237/10—Heterocyclic compounds containing 1,2-diazine or hydrogenated 1,2-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D237/14—Oxygen atoms
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/02—Antiprotozoals, e.g. for leishmaniasis, trichomoniasis, toxoplasmosis
- A61P33/06—Antimalarials
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/10—Preparation of carboxylic acid esters by reacting carboxylic acids or symmetrical anhydrides with ester groups or with a carbon-halogen bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Public Health (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Diabetes (AREA)
- Hematology (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Tropical Medicine & Parasitology (AREA)
- Oncology (AREA)
- Neurosurgery (AREA)
- Pain & Pain Management (AREA)
- Rheumatology (AREA)
- Neurology (AREA)
- Emergency Medicine (AREA)
- Endocrinology (AREA)
- Obesity (AREA)
- Biomedical Technology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
本发明涉及含S构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用。具体地,本发明涉及一种以下通式I所示的化合物或其药学上可接受的盐,其制备方法、药物组合物及应用。本申请的S构型化合物对I类组蛋白去乙酰化酶(HDAC1)具有很强的结合活性,对多种肿瘤细胞体外增殖显示出抑制活性。
Description
技术领域
本发明涉及氨基苯甲酰胺基哒嗪酮类化合物,更具体地,本发明涉及一种含S构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物以及在制备用于预防或治疗与I类组蛋白去乙酰化酶(class I HDAC)活性异常表达相关的疾病的药物中的用途。
背景技术
细胞内的组蛋白或非组蛋白乙酰化水平是受组蛋白乙酰转移酶(HAT)和组蛋白去乙酰化酶(HDAC)控制,进而影响细胞周期、分化、血管生成及凋亡等。HDAC的过度表达,促使组蛋白或非组蛋白发生去乙酰化导致染色质凝聚,从而抑制抑癌基因的转录。研究表明,抑制HDAC的活性可以实现对表观遗传学功能的重塑,已被证明是一种有效的治疗恶性肿瘤的方法(Nat.Rev.Drug.Discov.2014,13(9),673-691)。
迄今为止,FDA批准了四种HDAC抑制剂用于治疗皮肤T细胞淋巴瘤(CTCL)、外周T细胞淋巴瘤(PTCL)和多发性骨髓瘤(MM),包括2006年批准的第一个HDAC抑制剂伏立诺他(Vorinostat,SAHA)、2009年批准的罗米地辛(Romidepsin,FK228)、2014年批准的贝林司他(Belinostat,PXD-101)和2015年批准的帕比司他(Panobinostat,LBH589)。另一类氨基苯甲酰胺类化合物属于I类(class I)HDAC的抑制剂,其中最具代表性的化合物包括西达本胺(Chidamide,CS055),由NMPA批准用于治疗PTCL,与依西美坦联用,用于治疗HR+乳腺癌,但单用有效性不足;以及处于临床三期的恩替诺特(Entinostat,MS-275),但该化合物毒性较大,安全窗小,限制了进一步的临床应用(Adv.Cancer.Res.2018,138,183-211)。此外,还有大量的HDAC抑制剂处于药物研发的不同阶段。
CN109280032A公开一种哒嗪酮母核结构的化合物,可以作为HDAC抑制剂,用于与HDAC活性异常表达的相关哺乳动物疾病,如肿瘤、炎症等,其中代表性化合物HYH-072与HYH-073显示出良好的酶和肿瘤细胞增殖抑制活性,但是该类代表性化合物为消旋体。
发明内容
发明人进一步的研究表明,不同构型的氨基苯甲酰胺基哒嗪酮类化合物对酶和细胞增殖抑制活性具有较大差异,含S构型的氨基苯甲酰胺基哒嗪酮类化合物具有更高的活性,由此完成了本发明。
本发明的第一个目的是提供含S构型的氨基苯甲酰胺基哒嗪酮类化合物,或其药学上可接受的盐。
本发明的第二个目的是提供所述化合物的制备方法。
本发明的第三个目的是提供一种药物组合物,其包含治疗有效量的选自上述化合物及其药学上可接受的盐中的一种或多种。
本发明的第四个目的是提供上述化合物或其药学上可接受的盐在制备或上述药物组合物用于预防或治疗与I类组蛋白去乙酰化酶(class I HDAC)活性异常表达相关的疾病的药物中的用途。
一方面,本发明提供了一种通式I所示的化合物或其药学上可接受的盐,
其中:
环A选自C6-C10芳基、含1-3个选自N、O和S中的杂原子的5-10元杂芳基;
R1代表一个或多个取代基,所述取代基各自独立地选自H、卤素、C1-C6直链或支链烷基、C1-C6直链或支链烷氧基、(Ra)(Rb)N(CH2)-;其中,Ra和Rb各自独立地C1-C3烷基或者与相连的N一起形成3-7元的杂环基;
R2、R3和R4彼此相同或不同,并各自独立地选自氢、卤素、C1-C3烷基、C1-C3烷氧基。
在一些实施方式中,环A选自苯基、吡啶基或吡唑基。
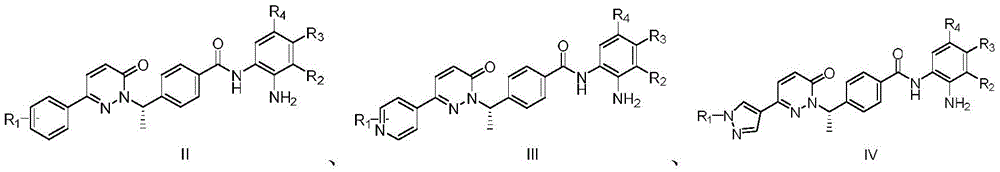
在一些实施方式中,所述化合物选自:
其中:
R1、R2、R3和R4的定义分别与上述相同。
在一些实施方式中,所述化合物选自以下化合物:
本发明的化合物还可以以溶剂化物(包括水合物)、多晶型物、同位素标记物(例如氘代物)、互变异构体或前药的形式存在,这些存在形式也都包括在本发明的保护范围内。
第二方面,本发明提供了上文所述化合物的制备方法,所述方法为如下路线之一,
路线一:
包括步骤:
(1)将H3所示的(R)-4-(1-羟乙基)苯甲酸叔丁酯与6-氯哒嗪酮经Mitsunobu反应得到构型翻转的H4所示的(S)-4-(1-(3-氯-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸叔丁酯;
(2)H4所示的(S)-4-(1-(3-氯-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸叔丁酯与H5所示的化合物发生Suzuki偶联反应得到H6所示的化合物;
(3)H6所示的化合物脱去叔丁基保护得到H7所示的化合物;
(4)H7所示的化合物与H8所示的化合物经缩合反应得到式I所示的化合物;
路线二:
包括步骤:
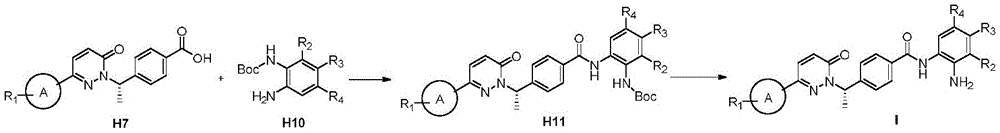
(1)H7所示的化合物与H10所示的化合物经缩合反应得到H11所示化合物;
(2)将H11所示的化合物脱去Boc保护基得到式I所示的化合物;
包括步骤:
(1)H7所示的化合物与H13所示的化合物经缩合反应得到H14所示的化合物;
(2)H14所示的化合物经还原反应得到式I所示的化合物;
其中,环A、R1、R2、R3和R4的定义分别如前文所述。
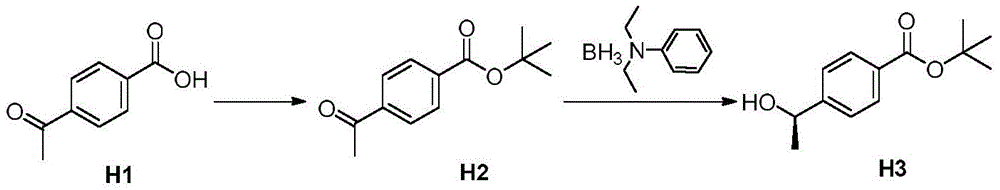
在所述方法中,手性中间体H3可以为市售的,也可以是自合成的。
在一个实施方式中,H3可以如下合成:
(1)H1所示的对乙酰基苯甲酸与Boc酸酐发生酯化反应得到H2所示的对乙酰基苯甲酸叔丁酯;
(2)将H2所示的对乙酰基苯甲酸叔丁酯在手性催化剂(S)-2-甲基-CBS-恶唑硼烷((S)-2-Me-CBS)的作用下与N,N-二乙苯胺硼烷复合物发生还原反应得到H3所示的(R)-4-(1-羟乙基)苯甲酸叔丁酯。
上述手性中间体H3的合成操作简单、手性ee值高(97.2%,见图5)、适合工业化生产。
第三方面,本发明提供了一种下式H7所示的中间体:
其中,环A和R1的定义分别如前文所述。
第四方面,本发明提供了一种药物组合物,其包含选自上文所述化合物和其药学上可接受的盐中的至少一种。所述药物组合物还可以包含药学上可接受的载体。
第五方面,本发明提供了上文所述化合物或其药学上可接受的盐或上文所述药物组合物在制备用于预防或治疗与I类组蛋白去乙酰化酶(class I HDAC)活性异常表达相关的疾病的药物中的用途。
在一些实施方式中,所述与I类组蛋白去乙酰化酶(class I HDAC)活性异常表达相关的疾病包括癌症、炎症、神经变性疾病、疟疾或糖尿病。
在一些实施方式中,所述癌症选自骨髓增生异常综合征、白血病(包括单核细胞白血病、急性髓系白血病、唐氏综合征急性巨核细胞白血病、T细胞急性淋巴细胞白血病、急性淋巴细胞白血病、多发性骨髓性白血病、慢性粒细胞白血病、人T淋巴细胞白血病、急性髓细胞样白血病)、淋巴瘤(包括弥漫性大B细胞淋巴瘤、套细胞淋巴瘤、原发性皮肤T细胞非霍奇金淋巴瘤、原发性纵隔大B细胞淋巴瘤、Hodgkin淋巴瘤)、多发性骨髓瘤、肺癌、肾癌、胃癌、乳腺癌、黑色素瘤、结肠癌、肝癌、卵巢癌、胰腺癌、直肠癌。
应理解,在本发明范围内,本发明的上述各技术特征和在下文(如实施例)中具体描述的各技术特征之间都可以互相组合,从而构成新的或优选的技术方案。限于篇幅,在此不再一一累述。
有益效果
本发明人研究此类含S构型氨基苯甲酰胺基哒嗪酮类I型HDAC抑制剂的构效关系,发现将哒嗪酮2位N上连接苄基为S构型时,其对HDAC1、HDAC2和HDAC3各个亚型的抑制活性以及对人骨髓增生异常综合征细胞SKM-1、人结肠癌细胞HCT-116的增殖抑制活性明显优于R构型,高于Chidamide和MS-275,说明S构型化合物在分子及细胞活性中发挥主要作用。
更重要的是,本发明所提供的化合物在同等剂量下,对人骨髓增生异常综合征细胞SKM-1(见图1)、急性髓性白血病细胞OCI-AML-3(见图2)小鼠异种移植瘤模型的抑制效果远优于上市药物Chidamide,在药效相当情况下,安全性优于MS-275。连续口服给药化合物I-1 60mg/kg 18天,对人骨髓增生异常综合征细胞SKM-1NOD/SCID小鼠移植瘤有治疗作用,能有效抑制小鼠移植瘤的增殖,相对肿瘤增殖率T/C为11.78%,明显优于口服给药Chidamide60 mg/kg qd实验组药效(T/C为73.45%),优于口服给药MS-275 20mg/kg qd实验组药效(T/C为18.23%)。连续口服给药化合物I-1 60mg/kg 14天,对人急性髓性白血病细胞OCI-AML-3NOD/SCID小鼠移植瘤有治疗作用,能有效抑制小鼠移植瘤的增殖,相对肿瘤增殖率T/C为7.75%,明显优于口服给药Chidamide80mg/kg qd实验组药效(T/C为69.91%),优于口服给药MS-275 20mg/kg qd实验组药效(T/C为14.16%)。且实验过程中小鼠体重基本无变化,表明化合物I-1具有较好的安全性。
综上所述,本发明所提供的化合物活性有了较大的提高,口服吸收效果良好,同时药效显著,因而具有更好的开发前景。
另外,本发明的化合物对多种实体瘤(如肺癌、肾癌、胃癌、乳腺癌、黑色素瘤、结肠癌、肝癌、淋巴瘤等)也具备十分优异的抑制活性。
附图说明
图1示出本申请的化合物I-1在人骨髓增生异常综合征SKM-1小鼠移植瘤模型上的药效结果。
图2示出本申请的化合物I-1在人急性髓性白血病OCI-AML-3小鼠移植瘤模型上的药效结果。
图3示出本申请的化合物I-1、I-2在人恶性黑色素瘤A375小鼠移植瘤模型上的药效结果。
图4示出本申请的化合物I-1不对称合成前(B)和不对称合成后(A)的ee值。
图5示出本申请实施例1中的中间体化合物H不对称合成前(B)和不对称合成后(A)的ee值。
具体实施方式
术语
在本发明中,除非另有具体说明外,对于取代基的含义做出如下定义:
所述C6-C10芳基指的是环上具有6至10个碳原子的芳香性碳环基,其具体的实例包括苯基、萘基等。
所述卤素原子指的是F、Cl、Br、I。
所述C1-C6直链或支链烷基指的是具有1至6个碳原子的直链或支链烷基,其具体实例包括甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、仲丁基、正戊基,1-甲基丁基、2-甲基丁基、3-甲基丁基、异戊基、1-乙基丙基、新戊基、正己基、1-甲基戊基、2-甲基戊基、3-甲基戊基、异己基、1,1-二甲基丁基、2,2-二甲基丁基、3,3-二甲基丁基、1,2-二甲基丁基、1,3-二甲基丁基、2,3-二甲基丁基、2-乙基丁基等。
所述C1-C6直链或支链烷氧基指的是具有1至6个碳原子的直链或支链烷氧基,其具体实例包括甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、异丁氧基、叔丁氧基、仲丁氧基、正戊氧基、异戊氧基、新戊氧基、正己氧基、异己氧基、3-甲基戊氧基等。
所述含1-3个选自N、O和S中的杂原子的5-10元杂芳基指的是在环上具有5至10个原子且包含1-3个选自N、O、S中的杂原子的芳香性环,其可以为单环或双环,例如吡啶环、吡咯环、嘧啶环、吡嗪环、哒嗪环、噻吩环、呋喃环、吡唑环、咪唑环、噁唑环、噻唑环、吲哚环、氮杂吲哚环、萘啶环、苯并咪唑环、吡啶并咪唑环、嘧啶并咪唑环或喹啉环等。
所述药学上可接受的盐,例如可以是无机酸盐,例如盐酸盐、氢溴酸盐、氢碘酸盐、硫酸盐、硝酸盐、磷酸盐、碳酸盐等;有机酸盐,例如甲酸盐、乙酸盐、丙酸盐、草酸盐、丙二酸盐、琥珀酸盐、富马酸盐、马来酸盐、己二酸、乳酸盐、苹果酸盐、柠檬酸盐、枸橼酸盐、酒石酸盐、碳酸盐、苦味酸盐、甲磺酸盐、乙磺酸盐、对甲苯磺酸盐、谷氨酸盐、双羟萘酸盐等。
本发明提供的通式I所示的含S构型氨基苯甲酰胺基哒嗪酮类化合物的药学上可接受的盐可以通过将通式I所示的含S构型氨基苯甲酰胺基哒嗪酮类化合物溶于用相应的酸饱和的醇溶液或二氧六环溶液中进行反应而制备,例如:将本发明提供的含S构型的氨基苯甲酰胺基哒嗪酮类化合物溶于HCl饱和的二氧六环溶液,室温搅拌30分钟,将溶剂蒸干,即制得相应的盐酸盐。
本发明的化合物还可以存在前药,因此,这些前药也包括在本发明化合物的保护范围内。所述前药是指药物经过化学结构修饰后得到的在体外无活性或活性较小、在体内经酶或非酶的转化释放出活性药物而发挥药效的化合物。本发明中的前药形式不做特别限定,只要其在体内经酶或化学作用释放具有活性的原药,从而发挥预期的药理作用即可,可以是载体前体药物或生物前体。
本发明的化合物可以以非溶剂化形式和含有药学上可接受的溶剂(如水、乙醇等)的溶剂化形式(也称为溶剂合物)存在,本发明的化合物包括溶剂化和非溶剂化形式。所述溶剂合物为通式I的化合物与药学上可接受的溶剂形成的配合物,可选地,所述药学上可接受的溶剂包括水、乙醇、乙酸、N,N-二甲基甲酰胺或二甲基亚砜等。
本发明的化合物还可以以不同互变异构形式存在,所有这些形式均包括在本发明范围内。术语“互变异构体”或“互变异构形式”是指经由低能垒相互转化的不同能量的结构异构体。
本发明也涵盖经同位素标记的本发明化合物,除了一个或多个原子是被原子质量或质量数不同于自然中常见的原子质量或质量数之一原子所置换的事实之外,其是与此述者相同。可纳入本发明的化合物中的同位素实例,包括氢、碳、氮、氧、磷、硫、氟、碘及氯的同位素,其分别例如:2氢、3氢、11碳、13碳、14碳、13氮、15氮、15氧、17氧、18氧、31磷、32磷、35硫、18氟、123碘、125碘及36氯。本发明的同位素标记的化合物一般可以遵循类似于在方案和/或下文实施例中所公开的方法,通过用同位素标记的试剂替代非同位素标记的试剂来制备。
所述药学上可接受的载体是指药学领域常规的药物载体,例如:稀释剂,如水等;填充剂,如淀粉、蔗糖等;粘合剂,如纤维素衍生物、藻酸盐、明胶、聚乙烯吡咯烷酮;湿润剂,如甘油;崩解剂,如琼脂、碳酸钙和碳酸氢钠;吸收促进剂,如季铵化合物;表面活性剂,如十六烷醇;吸附载体,如高岭土和皂粘土;润滑剂,如滑石粉、硬脂酸钙和硬脂酸镁、和聚乙二醇等。另外,还可以在上述药物组合物中加入其它辅剂,如香味剂和甜味剂等。
下面结合具体实施例,进一步阐述本发明。应理解,这些实施例仅用于说明本发明而不用于限制本发明的范围。
下面的实施例用于具体地说明本发明提供的含S构型的氨基苯甲酰胺基哒嗪酮类化合物的制备过程,以及其作为I类HDAC抑制剂的生物学活性,但本发明并不局限于这些实施例。
在以下实施例中,核磁共振氢谱用BrukerAMX-400型、BrukerAMX-500型或AMX-600型核磁共振仪记录,化学位移δ的单位为ppm。如无特别说明,所有反应溶剂均按照常规方法进行纯化。柱层析用硅胶(200-300目)为青岛海洋化工分厂生产。如无特别标注,所有溶剂均为分析纯试剂,所用试剂均购自国药集团化学试剂有限公司。采用紫外荧光法显色。减压蒸除有机溶剂在旋转蒸发仪中进行。
实施例1:化合物I-1的合成
将对乙酰基苯甲酸(1,100.0g,609mmol)置于2L三颈瓶中,加入300ml四氢呋喃,冰浴冷却后加入4-二甲氨基吡啶(DMAP,14.88g,121mmol),保持内温为10℃,滴加溶于700ml四氢呋喃(THF)的(Boc)2O(279.1g,1278.9mmol)溶液,滴毕后室温反应24小时。TLC监测反应结束后,减压除去溶剂,加入750ml乙酸乙酯,依次用0.1N盐酸(2×750ml),饱和碳酸氢钠溶液(2×750ml),饱和食盐水(2×500ml)洗涤,无水硫酸钠干燥后减压除去溶剂,加入正庚烷750ml,用硅胶过滤得浅黄色固体130g(化合物2),收率97%。1H NMR(400MHz,CDCl3)δ8.06(d,J=8.7Hz,2H),7.98(d,J=8.7Hz,2H),2.64(s,3H),1.61(s,9H);ESI-MS:m/z=221[M+H]+。
氮气保护下向2L三颈瓶中加入(S)-2-甲基-CBS-恶唑硼烷((S)-2-Me-CBS,8.2g,29mmol)、N,N-二乙苯胺硼烷复合物(96.2g,590mmol)和300ml无水甲基叔丁基醚,冰浴冷却至10℃,滴加溶于300ml无水甲基叔丁基醚的4-乙酰基苯甲酸叔丁酯(2,130g,590mmol)溶液,温度控制在15~25℃。滴加完毕,升至室温,继续搅拌5小时直至原料反应完全。冰浴冷却至10℃,缓慢滴加60ml甲醇,继续搅拌30分钟。依次用0.5N盐酸(2×600ml),饱和食盐水(2×500ml)洗涤,无水硫酸钠干燥后减压除去溶剂,得浅黄色油状液体112g(化合物3),收率85%,手性ee值97.2%。1H NMR(400MHz,CDCl3)δ7.97(d,J=8.3Hz,2H),7.42(d,J=8.3Hz,2H),5.02–4.91(m,1H),1.88(d,J=3.4Hz,1H),1.59(s,9H),1.49(d,J=6.5Hz,3H);ESI-MS:m/z=223[M+H]+。
反应物(R)-4-(1-羟乙基)苯甲酸叔丁酯(3,111.5g,501mmol)、6-氯哒嗪酮(62.3g,477mmol)和三苯基膦(PPh3,150.2g,572mmol)置于3L三颈瓶中,N2保护下加入600mL无水THF溶解,随后,冷却至0℃,缓慢滴加偶氮二甲酸二异丙酯(DIAD,115.8g,572mmol),室温反应过夜。将反应液中固体过滤,滤液加入200ml乙酸乙酯和1000ml正庚烷,室温搅拌1小时。抽滤,滤液重新加入200ml乙酸乙酯和1000ml正庚烷打浆,抽滤,滤饼用乙酸乙酯:正庚烷=5:1(600mL)洗涤,减压除去溶剂,得黄色油状液体178.9g(化合物4),未经纯化直接用于下一步反应。ESI-MS:m/z=335[M+H]+。
将上述得到的(S)-4-(1-(3-氯-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸叔丁酯粗品(4,177g)、(4-((二甲氨基)甲基)苯基)硼酸盐酸盐(5,88.9g,412mmol)、Pd(dppf)Cl2(12.0g,17mmol)和碳酸钾(104g,755mmol)加入到1,4-二氧六环(1000ml)和水(100ml)中,N2置换后加热至85℃反应6h。将反应液冷却至室温,过滤到固体杂质,滤液减压浓缩至干,加入乙酸乙酯(EtOAc,1200ml)和水(800ml),分出有机层。加入醋酸(59ml),室温搅拌30分钟后加水(800ml),水相冰浴冷却至4℃,滴加2N氢氧化钠溶液,调节pH至10~11,再用乙酸乙酯(1200ml)萃取,硅胶过滤后减压除去溶剂,得粗品100g(化合物6),两步收率48%。1HNMR(400MHz,CDCl3)δ7.94(d,J=8.4Hz,2H),7.72(d,J=8.3Hz,2H),7.65(d,J=9.7Hz,1H),7.52(d,J=8.4Hz,2H),7.40(d,J=8.2Hz,2H),6.98(d,J=9.7Hz,1H),6.44(q,J=14.1,6.9Hz,1H),3.46(s,2H),2.26(s,6H),1.86(d,J=7.0Hz,3H),1.57(s,9H);ESI-MS:m/z=434[M+H]+.
(S)-4-(1-(3-(4-(((二甲基氨基)甲基)苯基)-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸叔丁酯(6,100g,230mmol)溶于175ml CH2Cl2,冰浴冷却至0℃,加入三氟乙酸(230ml),室温反应3小时。反应液减压蒸除溶剂,加入甲基叔丁基醚500ml打浆,将上清液倾出,如此反复三次,最后减压除去溶剂得白色固体(化合物7)80g,收率71%。1H NMR(400MHz,CDCl3)δ12.74(s,1H),8.03(d,J=8.4Hz,2H),7.83(d,J=8.3Hz,2H),7.65(d,J=9.7Hz,1H),7.54(d,J=8.3Hz,4H),7.07(d,J=9.7Hz,1H),6.47(q,J=7.0Hz,1H),4.26(d,J=8.5Hz,2H),2.83(s,6H),1.87(d,J=7.1Hz,3H);ESI-MS:m/z=378[M+H]+.
(S)-4-(1-(3-(4-(((二甲基氨基)甲基)苯基)-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸三氟乙酸盐(7,0.79g,1.60mmol)置于25ml三颈瓶中,加入2ml二甲基甲酰胺(DMF)溶解,随即加入1-羟基苯并三氮唑(HOBT,0.26g,1.93mmol)、N-(3-二甲氨基丙基)-N’-乙基碳二亚胺盐酸盐(EDCI,0.46g,2.41mmol),室温搅拌30分钟。N2保护下滴加溶于1.2ml DMF的4-氟-1,2-苯二胺(0.24g,1.93mmol)溶液和二异丙基乙基胺(0.79ml,4.82mmol)。室温搅拌过夜,向反应液中加入10%碳酸钾溶液(6.6ml)和二氯甲烷(15.0ml),分离出有机相。加入3N盐酸(6.4ml),室温搅拌2小时,分离出水相,水相冰浴冷却至4℃,滴加2N氢氧化钠溶液,调节pH至10~11,再用二氯甲烷(25ml)萃取,减压除去溶剂,粗品用反相柱层析分离(C18柱,甲醇-水体系),得白色固体(化合物I-1)0.32g,手性ee值97%,收率41%。1H NMR(400MHz,CDCl3)δ7.97(s,1H),7.85(d,J=8.1Hz,2H),7.73(d,J=8.2Hz,2H),7.65(d,J=9.7Hz,1H),7.56(d,J=8.0Hz,2H),7.41(d,J=8.2Hz,2H),7.13(dd,J=8.2,6.1Hz,1H),6.94(d,J=9.7Hz,1H),6.49(dt,J=12.2,5.5Hz,2H),6.42(dd,J=14.1,7.1Hz,1H),4.00(s,2H),3.46(s,2H),2.26(s,6H),1.87(d,J=7.0Hz,3H);ESI-MS:m/z=486[M+H]+.
实施例2-10:化合物I-2~I-10的合成
如下,以与实施例1类似的方法制备化合物I-2~I-10:
实施例11:化合物I-11的合成
将(S)-4-(1-(3-(4-(((二甲基氨基)甲基)苯基)-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸三氟乙酸盐(7,0.79g,1.60mmol)置于25ml三颈瓶中,加入2ml DMF溶解,随即加入1-羟基苯并三氮唑(HOBT,0.26g,1.93mmol)、N-(3-二甲氨基丙基)-N’-乙基碳二亚胺盐酸盐(EDCI,0.46g,2.41mmol),室温搅拌30分钟。N2保护下滴加溶于1.2ml DMF的(2-氨基-4-甲氧基苯基)氨基甲酸叔丁酯(0.46g,1.93mmol)溶液和二异丙基乙基胺(0.79ml,4.82mmol)。室温搅拌过夜,向反应液中加入10%碳酸钾溶液(6.6ml)和二氯甲烷(15.0ml),分离出有机相。加入3N盐酸(6.4ml),室温搅拌2小时,分离出水相,水相冰浴冷却至4℃,滴加2N氢氧化钠溶液,调节pH至10~11,再用二氯甲烷(25ml)萃取,减压除去溶剂,粗品用柱层析分离,得白色固体(中间体10)0.68g,收率73%。1H NMR(400MHz,CDCl3)δ8.05–7.96(m,1H),7.91(d,J=8.3Hz,2H),7.72(d,J=8.3Hz,2H),7.64(d,J=9.7Hz,1H),7.55(d,J=8.3Hz,2H),7.45(s,1H),7.42(d,J=8.3Hz,2H),7.08(d,J=8.8Hz,1H),7.04–7.01(m,1H),6.98(d,J=9.7Hz,1H),6.86(s,1H),6.44(q,J=7.0Hz,1H),3.75(s,3H),3.55(s,2H),2.30(s,6H),1.86(d,J=7.0Hz,3H),1.48(s,9H);ESI-MS:m/z=598[M+H]+.
中间体10(0.68g,1.16mmol)溶于2ml CH2Cl2,冰浴冷却至0℃,加入HCl饱和的二氧六环溶液(10ml),室温反应3小时。反应液减压蒸除溶剂,加入20ml饱和碳酸氢钠溶液,乙酸乙酯萃取(25ml×2),合并有机相,再用饱和食盐水溶液(15ml×1)洗涤,无水硫酸钠干燥后减压除去溶剂,反相柱层析分离(C18柱,甲醇-水体系)得白色固体(化合物I-11)0.43g,收率75%。1H NMR(400MHz,CDCl3)δ8.35(s,1H),7.86(d,J=8.2Hz,2H),7.73(d,J=8.2Hz,2H),7.65(d,J=9.7Hz,1H),7.58(d,J=8.2Hz,2H),7.41(d,J=8.2Hz,2H),7.31(d,J=2.9Hz,1H),6.97(d,J=9.7Hz,1H),6.80(d,J=8.6Hz,1H),6.63(dd,J=8.6,2.9Hz,1H),6.43(q,J=7.0Hz,1H),3.75(s,3H),3.47(s,2H),3.38(s,2H),2.26(s,6H),1.88(d,J=7.1Hz,3H).ESI-MS:m/z=498[M+H]+.
实施例12:化合物I-12的合成
除了使用(2-氨基-4-氟苯基)氨基甲酸叔丁酯代替(2-氨基-4-甲氧基苯基)氨基甲酸叔丁酯之外,以与实施例11相同的方法制备化合物I-12(白色固体,收率72%)。
1H NMR(400MHz,CDCl3)δ8.24(s,1H),7.85(d,J=8.3Hz,2H),7.74(d,J=8.3Hz,2H),7.66(d,J=9.7Hz,1H),7.58(d,J=8.3Hz,2H),7.48–7.37(m,3H),6.98(d,J=9.7Hz,1H),6.85–6.72(m,2H),6.43(q,J=7.1Hz,1H),3.49(s,2H),2.28(s,6H),1.89–1.86(m,5H);ESI-MS:m/z=486[M+H]+.
实施例13:化合物I-13的合成
将(S)-4-(1-(3-(4-(((二甲基氨基)甲基)苯基)-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸三氟乙酸盐(7,0.25g,0.51mmol)置于25ml三颈瓶中,N2保护下加入3ml无水DCM,冰浴冷却至0℃,加入一滴无水DMF并逐渐滴加草酰氯(52μL,0.61mmol),室温搅拌30分钟后减压蒸除溶剂。加入8ml无水DCM、5-甲基-2硝基苯胺(0.093g,0.61mmol)和二异丙基乙基胺(252μL,1.53mmol)。室温搅拌过夜,向反应液中加入饱和碳酸氢钠溶液(15ml)和二氯甲烷(15ml),分离出有机相,再用饱和食盐水(15ml)洗涤。无水硫酸钠干燥后减压除去溶剂,粗品用柱层析分离,得浅黄色固体(中间体12)0.21g,收率80%。1H NMR(400MHz,CDCl3)δ11.39(s,1H),8.80(s,1H),8.15(d,J=8.6Hz,1H),7.94(d,J=8.3Hz,2H),7.74(d,J=8.2Hz,2H),7.67(d,J=9.7Hz,1H),7.63(d,J=8.3Hz,2H),7.41(d,J=8.2Hz,2H),7.05–6.96(m,2H),6.45(q,J=7.0Hz,1H),3.49(s,2H),2.46(s,3H),2.26(s,6H),1.89(d,J=7.1Hz,3H);ESI-MS:m/z=512[M+H]+.
中间体12(0.20g,0.4mmol)溶于2ml乙醇和0.4ml水中,加入铁粉(0.11g,2.0mmol)和氯化铵(0.21g,4.0mmol),N2保护下加热回流5小时。降至室温,硅藻土过滤,滤饼用乙醇洗涤两次,减压蒸除溶剂,加入饱和碳酸氢钠溶液(20ml)和二氯甲烷(30ml),分离有机相,再用饱和食盐水(20ml)洗涤。无水硫酸钠干燥后减压除去溶剂,反相柱层析分离(C18柱,甲醇-水体系)得白色固体(化合物I-13)0.12g,收率62%。1H NMR(400MHz,CDCl3)δ8.47(s,1H),7.82(d,J=8.0Hz,2H),7.72(d,J=8.2Hz,2H),7.61(d,J=9.7Hz,1H),7.49(d,J=7.6Hz,2H),7.40(d,J=8.1Hz,2H),7.08(s,1H),6.89(dd,J=9.6,2.3Hz,1H),6.81(d,J=7.9Hz,1H),6.66(d,J=7.9Hz,1H),6.38(q,J=6.6Hz,1H),3.67(s,2H),3.45(s,2H),2.24(s,6H),2.17(s,3H),1.84(d,J=6.9Hz,3H);ESI-MS:m/z=482[M+H]+.
药理试验实施例
HYH-073(I-1消旋)、HYH-072(I-2消旋)、I-3(消旋)、I-4(消旋)和I-10(消旋)按照CN109280032A中公开的方法制备。
I-1(R构型)、I-2(R构型)、I-10(R构型)通过商业公司将上面制备的I-1(消旋)、I-2(消旋)和I-10(消旋)拆分得到。
MS-275和Chidamide购自MCE MedChemExpress;
药理试验实施例1:
组蛋白去乙酰化酶体外测活方法
利用昆虫杆状病毒表达系统表达带有His标签和GST标签的人源重组HDAC家族蛋白,通过Ni亲和柱蛋白纯化获得具有生物活性的HDAC家族重组蛋白。以H3(1-21)K9Ac-生物素(biotin)为底物,采用HTRF检测法,在白色384孔平底微孔板(proxiPlate-384 Plus,PerkinElmer)中加入HDAC蛋白、梯度稀释的化合物以及底物,反应1小时后,加入Ea标记的H3K9抗体和XL665标记的链霉亲和物的混合液,室温下平衡0.5小时,利用HTRF的原理,用酶标仪检测615nm和665nm处的时间分辨荧光,算出比值,用GraphPad进行分析计算相应的酶活抑制率。简单来说,20μl的测活体系中,包括HDAC底物(0.2μM,4μl),人源重组蛋白HDAC(2-5ng/μl,4μl)以及化合物(2μl),Eu标记的H3K9抗体和XL665标记的链霉亲和物的混合液(10μl),所有成分均是用Tris缓冲液稀释(50mM Tris-HCl,pH 8.0,137mM NaCl,2.7mM KCland 1mM MgCl2)。
药理学数据:以下表1中示出本发明的化合物的药理学试验结果,测试中采用的对照为MS-275和西达本胺(Chidamide)。
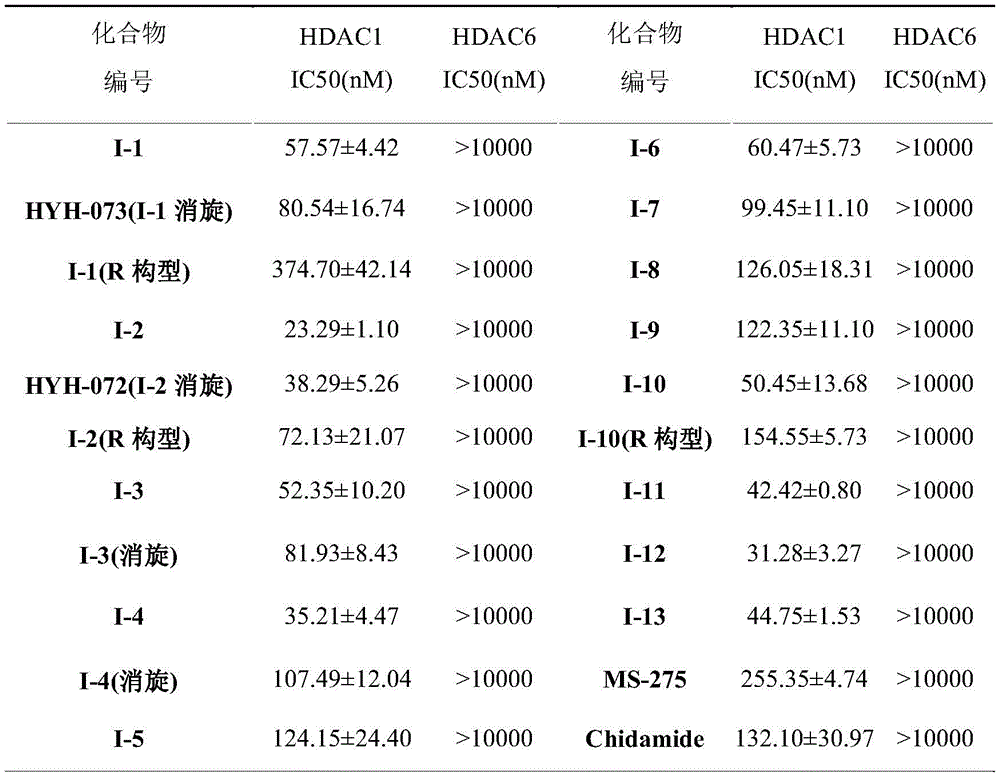
表1化合物对HDAC1、HDAC6抑制活性
从上表可以看到,该类含S构型化合物分子水平测试实验结果表明本发明化合物对HDAC1具有很强的结合活性,多数化合物的分子水平抑制活性优于阳性对照MS-275,优于上市药物西达本胺(Chidamide)或与其相当,并且对HDAC6亚型具有较高的选择性。此外,S构型化合物对HDAC1的结合活性优于R构型化合物,说明S构型化合物对HDAC1的结合活性发挥着主要作用。
药理实验实施例2:化合物对肿瘤细胞体外增殖抑制活性测试
所用的肿瘤细胞购自商业化的细胞库(HCT116购自ATCC,SKM 1购自JCRB)。细胞的生长抑制检测采用CCK-8方法。具体步骤如下:处于对数生长期的细胞按合适密度接种至96孔培养板,每孔90μL,培养过夜后,加入不同浓度的药物作用72h,每个浓度设三复孔,并设相应浓度的溶媒对照及无细胞调零孔。作用结束后,每孔加入10μL CCK-8,培养箱中孵育2-4h后,SpectraMax 190酶标仪测定450nm波长下的光密度(OD值)。按照以下公式计算药物对肿瘤细胞增殖抑制程度:抑制率(%)=(OD对照孔-OD给药孔)/OD对照孔×100%,并据此按四参数法(4-parameters)拟合IC50值。实验重复两次,计算平均值和SD。
实验结果见下表2(N.T.代表未测试)。
参考HCT-116细胞增殖抑制实验方法测试化合物对人骨髓增生异常综合征细胞SKM-1增殖抑制活性,实验结果见表2和图1。图1示出了化合物I-1在不同给药浓度下在人骨髓增生异常综合征SKM-1小鼠移植瘤模型上的药效结果。
表2化合物对HCT-116和SKM-1增殖抑制活性
NT=未检测(not test)
从表2的实验结果可知,该类化合物对不同类型肿瘤细胞株均表现出较强的体外增殖抑制活性,细胞活性优于MS-275和Chidamide。此外,对比I-1、I-1(R构型)、I-2、I-2(R构型)和I-10、I-10(R构型),S构型化合物的细胞活性明显优于R构型化合物,表明S构型对细胞活性发挥主要作用,该类化合物可以作为一类新型抗肿瘤药物进行开发。
药理实验实施例3:本申请的部分化合物对HDAC多个亚型选择性测试
生物学活性的测试方法与药理试验实施例1中记载的方法相同,结果如下表3中所示。
表3部分化合物对HDAC2、HDAC3、HDAC8和HDAC10抑制活性
从上表3可以看到,该类含S构型的化合物分子水平测试实验结果表明本发明化合物对HDAC2和HDAC3具有强效的结合活性,优于阳性对照MS-275;对HDAC10亚型的抑制活性较弱,选择性大于100倍,表明该类化合物是一类高选择性的class I HDAC抑制剂。此外,S构型化合物对HDAC2和HDAC3的结合活性优于R构型化合物,说明S构型化合物对HDAC2和HDAC3的结合活性发挥着主要作用。
药理实验实施例4:本申请的化合物I-1对多种肿瘤细胞体外增殖抑制活性测试
细胞活性的测试方法与药理试验实施例2中记载的方法相同,结果如下表4和图2-3所示。图2示出化合物I-1在人急性髓性白血病OCI-AML-3小鼠移植瘤模型上的药效结果。图3示出化合物I-1在人恶性黑色素瘤A375小鼠移植瘤模型上的药效结果。
表4化合物I-1对多种肿瘤细胞株增殖抑制活性
结果表明,化合物I-1对多种血液瘤细胞株和多种实体瘤细胞株均有强效的抑制活性,属于广谱类肿瘤抑制剂。
在本发明提及的所有文献都在本申请中引用作为参考,就如同每一篇文献被单独引用作为参考那样。此外应理解,在阅读了本发明的上述讲授内容之后,本领域技术人员可以对本发明作各种改动或修改,这些等价形式同样落于本申请所附权利要求书所限定的范围。
Claims (10)
5.一种制备权利要求1-4中任一项所述的化合物的方法,所述方法为如下路线之一,
路线一:
包括步骤:
(1)将H3所示的(R)-4-(1-羟乙基)苯甲酸叔丁酯与6-氯哒嗪酮经Mitsunobu反应得到构型翻转的H4所示的(S)-4-(1-(3-氯-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸叔丁酯;
(2)H4所示的(S)-4-(1-(3-氯-6-氧并哒嗪-1(6H)-基)乙基)苯甲酸叔丁酯与H5所示的化合物发生Suzuki偶联反应得到H6所示的化合物;
(3)H6所示的化合物脱去叔丁基保护得到H7所示的化合物;
(4)H7所示的化合物与H8所示的化合物经缩合反应得到式I所示的化合物;
路线二:
包括步骤:
(1)H7所示的化合物与H10所示的化合物经缩合反应得到H11所示化合物;
(2)将H11所示的化合物脱去Boc保护基得到式I所示的化合物;
路线三:
包括步骤:
(1)H7所示的化合物与H13所示的化合物经缩合反应得到H14所示的化合物;
(2)H14所示的化合物经还原反应得到式I所示的化合物;
其中,环A、R1、R2、R3和R4的定义分别如对应权利要求所述。
7.一种药物组合物,其包含选自权利要求1至4中任一项所述的化合物和其药学上可接受的盐中的至少一种,任选地还包含药学上可接受的载体。
8.根据权利要求1至4中任一项所述的化合物或其药学上可接受的盐,或权利要求7所述的药物组合物在制备用于预防或治疗与I类组蛋白去乙酰化酶活性异常表达相关的疾病的药物中的用途。
9.根据权利要求8所述的用途,其中,所述与I类组蛋白去乙酰化酶活性异常表达相关的疾病包括癌症、炎症、神经变性疾病、疟疾及糖尿病。
10.根据权利要求9所述的用途,其中,所述癌症选自骨髓增生异常综合征、白血病、淋巴瘤、多发性骨髓瘤、肺癌、肾癌、胃癌、乳腺癌、黑色素瘤、结肠癌、肝癌、卵巢癌、胰腺癌、直肠癌,
特别地,所述白血病包括单核细胞白血病、急性髓系白血病、唐氏综合征急性巨核细胞白血病、T细胞急性淋巴细胞白血病、急性淋巴细胞白血病、多发性骨髓性白血病、慢性粒细胞白血病、人T淋巴细胞白血病、急性髓细胞样白血病;
所述淋巴瘤包括弥漫性大B细胞淋巴瘤、套细胞淋巴瘤、原发性皮肤T细胞非霍奇金淋巴瘤、原发性纵隔大B细胞淋巴瘤、Hodgkin淋巴瘤。
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110193453.0A CN114957132A (zh) | 2021-02-20 | 2021-02-20 | 含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 |
| US18/278,063 US20240150300A1 (en) | 2021-02-20 | 2022-02-18 | S-configuration-containing amino benzamide pyridazinone compound, preparation method therefor, and pharmaceutical composition and application thereof |
| EP22755576.0A EP4296266A1 (en) | 2021-02-20 | 2022-02-18 | S-configuration-containing amino benzamide pyridazinone compound, preparation method therefor, and pharmaceutical composition and application thereof |
| JP2023550056A JP2024504862A (ja) | 2021-02-20 | 2022-02-18 | S配置含有アミノベンズアミドピリダジノン系化合物、その製造方法、医薬組成物および応用 |
| PCT/CN2022/076727 WO2022174803A1 (zh) | 2021-02-20 | 2022-02-18 | 含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110193453.0A CN114957132A (zh) | 2021-02-20 | 2021-02-20 | 含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114957132A true CN114957132A (zh) | 2022-08-30 |
Family
ID=82932095
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110193453.0A Pending CN114957132A (zh) | 2021-02-20 | 2021-02-20 | 含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20240150300A1 (zh) |
| EP (1) | EP4296266A1 (zh) |
| JP (1) | JP2024504862A (zh) |
| CN (1) | CN114957132A (zh) |
| WO (1) | WO2022174803A1 (zh) |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004058234A2 (en) * | 2002-12-27 | 2004-07-15 | Schering Aktiengesellschaft | Pharmaceutical combinations of phthalazine vegf inhibitors and benzamide hdac inhibitors |
| WO2008136378A1 (ja) * | 2007-04-27 | 2008-11-13 | Toyama Chemical Co., Ltd. | 新規なスルホンアミド誘導体またはその塩 |
| CN103732578A (zh) * | 2011-07-22 | 2014-04-16 | 辉瑞大药厂 | 喹啉基胰高血糖素受体调节剂 |
| CN104968658A (zh) * | 2013-02-07 | 2015-10-07 | 默克专利股份公司 | 哒嗪酮-酰胺衍生物 |
| CN107235960A (zh) * | 2016-03-29 | 2017-10-10 | 浙江海正药业股份有限公司 | 酰胺类衍生物、其制备方法及其在医药上的用途 |
| CN107417687A (zh) * | 2016-05-24 | 2017-12-01 | 中国科学院上海药物研究所 | 五元杂环并[3,4‑d]哒嗪酮类化合物、其制备方法、药物组合物及其应用 |
| CN107879975A (zh) * | 2016-09-30 | 2018-04-06 | 中国科学院上海药物研究所 | 组蛋白去乙酰化酶抑制剂及其应用 |
| CN108026057A (zh) * | 2015-05-22 | 2018-05-11 | 株式会社钟根堂 | 用作选择性组蛋白脱乙酰酶抑制剂的杂环烷基衍生化合物及含其的药物组合物 |
| CN108752322A (zh) * | 2018-09-12 | 2018-11-06 | 广州新民培林医药科技有限公司 | 一种新型Tepotinib衍生物和制备方法及其在抗肿瘤药物中的应用 |
| CN109280032A (zh) * | 2017-07-19 | 2019-01-29 | 中国科学院上海药物研究所 | 一种哒嗪酮母核结构的组蛋白去乙酰化酶抑制剂及其制备方法和用途 |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BR112012015633A2 (pt) * | 2009-12-25 | 2018-05-29 | Daiichi Sankyo Company, Limited | "composto com anel de sete membros e seu uso farmacêutico" |
| CA2809662C (en) * | 2010-09-01 | 2019-04-16 | Gilead Connecticut, Inc. | Pyridazinones, method of making, and method of use thereof |
| WO2012149540A1 (en) * | 2011-04-28 | 2012-11-01 | The Broad Institute Inc | Inhibitors of histone deacetylase |
-
2021
- 2021-02-20 CN CN202110193453.0A patent/CN114957132A/zh active Pending
-
2022
- 2022-02-18 WO PCT/CN2022/076727 patent/WO2022174803A1/zh not_active Ceased
- 2022-02-18 JP JP2023550056A patent/JP2024504862A/ja active Pending
- 2022-02-18 US US18/278,063 patent/US20240150300A1/en active Pending
- 2022-02-18 EP EP22755576.0A patent/EP4296266A1/en not_active Withdrawn
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2004058234A2 (en) * | 2002-12-27 | 2004-07-15 | Schering Aktiengesellschaft | Pharmaceutical combinations of phthalazine vegf inhibitors and benzamide hdac inhibitors |
| WO2008136378A1 (ja) * | 2007-04-27 | 2008-11-13 | Toyama Chemical Co., Ltd. | 新規なスルホンアミド誘導体またはその塩 |
| CN103732578A (zh) * | 2011-07-22 | 2014-04-16 | 辉瑞大药厂 | 喹啉基胰高血糖素受体调节剂 |
| CN104968658A (zh) * | 2013-02-07 | 2015-10-07 | 默克专利股份公司 | 哒嗪酮-酰胺衍生物 |
| CN108026057A (zh) * | 2015-05-22 | 2018-05-11 | 株式会社钟根堂 | 用作选择性组蛋白脱乙酰酶抑制剂的杂环烷基衍生化合物及含其的药物组合物 |
| CN107235960A (zh) * | 2016-03-29 | 2017-10-10 | 浙江海正药业股份有限公司 | 酰胺类衍生物、其制备方法及其在医药上的用途 |
| CN107417687A (zh) * | 2016-05-24 | 2017-12-01 | 中国科学院上海药物研究所 | 五元杂环并[3,4‑d]哒嗪酮类化合物、其制备方法、药物组合物及其应用 |
| CN107879975A (zh) * | 2016-09-30 | 2018-04-06 | 中国科学院上海药物研究所 | 组蛋白去乙酰化酶抑制剂及其应用 |
| CN109280032A (zh) * | 2017-07-19 | 2019-01-29 | 中国科学院上海药物研究所 | 一种哒嗪酮母核结构的组蛋白去乙酰化酶抑制剂及其制备方法和用途 |
| CN108752322A (zh) * | 2018-09-12 | 2018-11-06 | 广州新民培林医药科技有限公司 | 一种新型Tepotinib衍生物和制备方法及其在抗肿瘤药物中的应用 |
Non-Patent Citations (2)
| Title |
|---|
| DAVID K. HERRON等: "1,2-Dibenzamidobenzene Inhibitors of Human Factor Xa", J. MED. CHEM. * |
| JIABIN YANG等: "Synthesis of 3,4-diaminobenzoyl derivatives as factor Xa inhibitors", EUROPEAN JOURNAL OF MEDICINAL CHEMISTRY * |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4296266A4 (en) | 2023-12-27 |
| US20240150300A1 (en) | 2024-05-09 |
| WO2022174803A1 (zh) | 2022-08-25 |
| EP4296266A1 (en) | 2023-12-27 |
| JP2024504862A (ja) | 2024-02-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101414931B1 (ko) | 아실티오우레아 화합물 또는 그 염 및 그 용도 | |
| CN110770242B (zh) | 作为vanin抑制剂的杂芳族化合物 | |
| EP1627869A1 (en) | Amide derivative | |
| CN113527195B (zh) | 一类5-芳基烟酰胺类lsd1/hdac双靶点抑制剂、其制备方法及应用 | |
| CN109843890B (zh) | 三氮唑并嘧啶、三氮唑并吡啶化合物及其组合物用于治疗prc2介导的疾病 | |
| JP2023036991A (ja) | Ehmt2阻害剤としてのアミン置換複素環化合物、その塩、及びそれらの合成方法 | |
| JP2024528251A (ja) | Hdacとnad合成を標的とする多標的阻害剤及びその用途 | |
| AU2019218187A1 (en) | Dioxinoquinoline compounds, preparation method and uses thereof | |
| WO2001004125A1 (en) | Staurosporin derivatives | |
| EP3760633B1 (en) | Oxazino-quinazoline and oxazino-quinazoline type compound, preparation method therefor, and uses thereof | |
| CN117069696B (zh) | 一种双靶点小分子抑制剂及其制备方法和应用 | |
| CN103833756A (zh) | 一类哒嗪酮类化合物及其制备方法和用途 | |
| CN102026969A (zh) | 新型的n-(2-氨基-苯基)-丙烯酰胺类 | |
| CN113166148B (zh) | 作为cdk-hdac双通路抑制剂的杂环化合物 | |
| CN112341390B (zh) | 用于制备靶向组蛋白甲基转移酶ezh2共价抑制剂的化合物及其制备方法和用途 | |
| WO2022174803A1 (zh) | 含s构型的氨基苯甲酰胺基哒嗪酮类化合物、其制备方法、药物组合物及应用 | |
| CN115368306B (zh) | 含四氢异喹啉类结构的hdac抑制剂、组合物及其用途 | |
| EP3750893A1 (en) | Dioxazoline compound, preparation method therefor, and uses thereof | |
| CN109111439A (zh) | 一种酰胺类化合物及包含该化合物的组合物及其用途 | |
| JP2007332061A (ja) | 新規ピラゾロ[1,5−a]ピリミジン誘導体及びその用途 | |
| CN106496132B (zh) | N-(4-取代苯基)-2-取代乙酰胺类化合物及其作为sirt2蛋白抑制剂的用途 | |
| WO2013159698A1 (zh) | 稠环喹唑啉羟肟酸类化合物及其作为抗肿瘤药物的应用 | |
| CN109705057B (zh) | 组蛋白去乙酰化酶抑制剂及其制备方法与用途 | |
| WO2024239186A1 (zh) | 吡唑甲酰基哌嗪酮类化合物、药物组合物及其应用 | |
| WO2025082225A1 (en) | Benzo [c] [1, 2] oxaborol-1 (3h) -ol derivatives as shp2 inhibitors, compositions and methods thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |