CN114591334A - Dihydropyrazolopyrimidinone derivatives - Google Patents
Dihydropyrazolopyrimidinone derivatives Download PDFInfo
- Publication number
- CN114591334A CN114591334A CN202111465345.0A CN202111465345A CN114591334A CN 114591334 A CN114591334 A CN 114591334A CN 202111465345 A CN202111465345 A CN 202111465345A CN 114591334 A CN114591334 A CN 114591334A
- Authority
- CN
- China
- Prior art keywords
- membered
- cancer
- alkyl
- amino
- hydroxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
- 150000001875 compounds Chemical class 0.000 claims abstract description 108
- 150000003839 salts Chemical class 0.000 claims abstract description 44
- 239000003814 drug Substances 0.000 claims abstract description 22
- 101150040313 Wee1 gene Proteins 0.000 claims abstract description 20
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 17
- -1 hydroxy, amino Chemical group 0.000 claims description 414
- 125000005843 halogen group Chemical group 0.000 claims description 75
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 72
- 150000003254 radicals Chemical class 0.000 claims description 60
- 229910052736 halogen Inorganic materials 0.000 claims description 58
- 150000002367 halogens Chemical class 0.000 claims description 58
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 48
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 41
- 125000001424 substituent group Chemical group 0.000 claims description 36
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 32
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 32
- 125000004432 carbon atom Chemical group C* 0.000 claims description 27
- 206010028980 Neoplasm Diseases 0.000 claims description 26
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 25
- 201000011510 cancer Diseases 0.000 claims description 23
- 229910052760 oxygen Inorganic materials 0.000 claims description 21
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical group [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 20
- 239000001301 oxygen Substances 0.000 claims description 20
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 claims description 19
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 18
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 17
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 17
- 229910052739 hydrogen Inorganic materials 0.000 claims description 16
- 239000001257 hydrogen Substances 0.000 claims description 16
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 claims description 15
- 125000003545 alkoxy group Chemical group 0.000 claims description 15
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 15
- 201000010099 disease Diseases 0.000 claims description 14
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 14
- 125000003003 spiro group Chemical group 0.000 claims description 14
- 125000004076 pyridyl group Chemical group 0.000 claims description 13
- 230000001404 mediated effect Effects 0.000 claims description 12
- 125000002098 pyridazinyl group Chemical group 0.000 claims description 12
- 125000006620 amino-(C1-C6) alkyl group Chemical group 0.000 claims description 11
- 210000004027 cell Anatomy 0.000 claims description 11
- 125000003373 pyrazinyl group Chemical group 0.000 claims description 11
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 claims description 11
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 11
- 125000001153 fluoro group Chemical group F* 0.000 claims description 10
- 239000003112 inhibitor Substances 0.000 claims description 10
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 10
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 9
- 238000011282 treatment Methods 0.000 claims description 9
- 125000001072 heteroaryl group Chemical group 0.000 claims description 7
- 239000013543 active substance Substances 0.000 claims description 6
- 125000004448 alkyl carbonyl group Chemical group 0.000 claims description 6
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 claims description 6
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 6
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 claims description 5
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 5
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 claims description 5
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 5
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 5
- 125000006714 (C3-C10) heterocyclyl group Chemical group 0.000 claims description 4
- 125000000041 C6-C10 aryl group Chemical group 0.000 claims description 4
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 4
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 4
- 230000002265 prevention Effects 0.000 claims description 4
- 102000004169 proteins and genes Human genes 0.000 claims description 4
- 108090000623 proteins and genes Proteins 0.000 claims description 4
- 206010005949 Bone cancer Diseases 0.000 claims description 3
- 208000018084 Bone neoplasm Diseases 0.000 claims description 3
- 208000003174 Brain Neoplasms Diseases 0.000 claims description 3
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 3
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 3
- 206010009944 Colon cancer Diseases 0.000 claims description 3
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 3
- 206010025323 Lymphomas Diseases 0.000 claims description 3
- 201000004404 Neurofibroma Diseases 0.000 claims description 3
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 3
- 208000000453 Skin Neoplasms Diseases 0.000 claims description 3
- 206010041067 Small cell lung cancer Diseases 0.000 claims description 3
- 208000036765 Squamous cell carcinoma of the esophagus Diseases 0.000 claims description 3
- 208000024313 Testicular Neoplasms Diseases 0.000 claims description 3
- 206010057644 Testis cancer Diseases 0.000 claims description 3
- 208000024770 Thyroid neoplasm Diseases 0.000 claims description 3
- 208000029742 colonic neoplasm Diseases 0.000 claims description 3
- 208000035475 disorder Diseases 0.000 claims description 3
- 239000002552 dosage form Substances 0.000 claims description 3
- 208000007276 esophageal squamous cell carcinoma Diseases 0.000 claims description 3
- 210000005002 female reproductive tract Anatomy 0.000 claims description 3
- 125000003253 isopropoxy group Chemical group [H]C([H])([H])C([H])(O*)C([H])([H])[H] 0.000 claims description 3
- 201000005202 lung cancer Diseases 0.000 claims description 3
- 208000020816 lung neoplasm Diseases 0.000 claims description 3
- 230000000394 mitotic effect Effects 0.000 claims description 3
- 239000000825 pharmaceutical preparation Substances 0.000 claims description 3
- 201000000849 skin cancer Diseases 0.000 claims description 3
- 208000000587 small cell lung carcinoma Diseases 0.000 claims description 3
- 201000003120 testicular cancer Diseases 0.000 claims description 3
- 201000002510 thyroid cancer Diseases 0.000 claims description 3
- 208000036764 Adenocarcinoma of the esophagus Diseases 0.000 claims description 2
- 108020004491 Antisense DNA Proteins 0.000 claims description 2
- 108020005544 Antisense RNA Proteins 0.000 claims description 2
- 206010005003 Bladder cancer Diseases 0.000 claims description 2
- 206010006187 Breast cancer Diseases 0.000 claims description 2
- 208000026310 Breast neoplasm Diseases 0.000 claims description 2
- 229940123587 Cell cycle inhibitor Drugs 0.000 claims description 2
- 206010008342 Cervix carcinoma Diseases 0.000 claims description 2
- 206010014733 Endometrial cancer Diseases 0.000 claims description 2
- 206010014759 Endometrial neoplasm Diseases 0.000 claims description 2
- 206010051066 Gastrointestinal stromal tumour Diseases 0.000 claims description 2
- 208000032612 Glial tumor Diseases 0.000 claims description 2
- 206010018338 Glioma Diseases 0.000 claims description 2
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 claims description 2
- 208000008839 Kidney Neoplasms Diseases 0.000 claims description 2
- 208000034578 Multiple myelomas Diseases 0.000 claims description 2
- 206010030137 Oesophageal adenocarcinoma Diseases 0.000 claims description 2
- 206010061534 Oesophageal squamous cell carcinoma Diseases 0.000 claims description 2
- 206010033128 Ovarian cancer Diseases 0.000 claims description 2
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 2
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 claims description 2
- 206010060862 Prostate cancer Diseases 0.000 claims description 2
- 229940079156 Proteasome inhibitor Drugs 0.000 claims description 2
- 208000015634 Rectal Neoplasms Diseases 0.000 claims description 2
- 206010038389 Renal cancer Diseases 0.000 claims description 2
- 206010039491 Sarcoma Diseases 0.000 claims description 2
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 claims description 2
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 claims description 2
- 239000002168 alkylating agent Substances 0.000 claims description 2
- 229940100198 alkylating agent Drugs 0.000 claims description 2
- 239000004037 angiogenesis inhibitor Substances 0.000 claims description 2
- 229940121369 angiogenesis inhibitor Drugs 0.000 claims description 2
- 239000003242 anti bacterial agent Substances 0.000 claims description 2
- 230000000340 anti-metabolite Effects 0.000 claims description 2
- 230000000259 anti-tumor effect Effects 0.000 claims description 2
- 229940088710 antibiotic agent Drugs 0.000 claims description 2
- 229940100197 antimetabolite Drugs 0.000 claims description 2
- 239000002256 antimetabolite Substances 0.000 claims description 2
- 239000003816 antisense DNA Substances 0.000 claims description 2
- 208000035269 cancer or benign tumor Diseases 0.000 claims description 2
- 201000010881 cervical cancer Diseases 0.000 claims description 2
- 239000003184 complementary RNA Substances 0.000 claims description 2
- 239000003534 dna topoisomerase inhibitor Substances 0.000 claims description 2
- 239000002532 enzyme inhibitor Substances 0.000 claims description 2
- 208000028653 esophageal adenocarcinoma Diseases 0.000 claims description 2
- 206010017758 gastric cancer Diseases 0.000 claims description 2
- 201000011243 gastrointestinal stromal tumor Diseases 0.000 claims description 2
- 239000003102 growth factor Substances 0.000 claims description 2
- 239000003966 growth inhibitor Substances 0.000 claims description 2
- 201000010536 head and neck cancer Diseases 0.000 claims description 2
- 208000014829 head and neck neoplasm Diseases 0.000 claims description 2
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 claims description 2
- 239000000367 immunologic factor Substances 0.000 claims description 2
- 201000010982 kidney cancer Diseases 0.000 claims description 2
- 208000032839 leukemia Diseases 0.000 claims description 2
- 201000007270 liver cancer Diseases 0.000 claims description 2
- 208000014018 liver neoplasm Diseases 0.000 claims description 2
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 2
- 201000006512 mast cell neoplasm Diseases 0.000 claims description 2
- 201000001441 melanoma Diseases 0.000 claims description 2
- 208000002154 non-small cell lung carcinoma Diseases 0.000 claims description 2
- 201000002528 pancreatic cancer Diseases 0.000 claims description 2
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 2
- 201000002628 peritoneum cancer Diseases 0.000 claims description 2
- 229940096701 plain lipid modifying drug hmg coa reductase inhibitors Drugs 0.000 claims description 2
- 125000001844 prenyl group Chemical group [H]C([*])([H])C([H])=C(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 2
- 239000003207 proteasome inhibitor Substances 0.000 claims description 2
- 206010038038 rectal cancer Diseases 0.000 claims description 2
- 201000001275 rectum cancer Diseases 0.000 claims description 2
- 102000027483 retinoid hormone receptors Human genes 0.000 claims description 2
- 108091008679 retinoid hormone receptors Proteins 0.000 claims description 2
- 230000011664 signaling Effects 0.000 claims description 2
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 2
- 201000011549 stomach cancer Diseases 0.000 claims description 2
- 230000008685 targeting Effects 0.000 claims description 2
- 229940044693 topoisomerase inhibitor Drugs 0.000 claims description 2
- 239000003558 transferase inhibitor Substances 0.000 claims description 2
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 claims description 2
- 201000005112 urinary bladder cancer Diseases 0.000 claims description 2
- 201000000498 stomach carcinoma Diseases 0.000 claims 2
- 229940088597 hormone Drugs 0.000 claims 1
- 238000004519 manufacturing process Methods 0.000 claims 1
- 239000000546 pharmaceutical excipient Substances 0.000 claims 1
- 201000001514 prostate carcinoma Diseases 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 66
- 229940079593 drug Drugs 0.000 abstract description 10
- 238000000034 method Methods 0.000 abstract description 7
- 229940043355 kinase inhibitor Drugs 0.000 abstract description 3
- 239000003757 phosphotransferase inhibitor Substances 0.000 abstract description 3
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 84
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 59
- 125000000623 heterocyclic group Chemical group 0.000 description 41
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 39
- 238000006243 chemical reaction Methods 0.000 description 32
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 30
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 28
- 125000004122 cyclic group Chemical group 0.000 description 27
- 239000000243 solution Substances 0.000 description 27
- 230000000694 effects Effects 0.000 description 21
- 125000004433 nitrogen atom Chemical group N* 0.000 description 21
- 239000000203 mixture Substances 0.000 description 20
- 125000006413 ring segment Chemical group 0.000 description 20
- 229920006395 saturated elastomer Polymers 0.000 description 20
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 20
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-diisopropylethylamine Substances CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 18
- 239000012043 crude product Substances 0.000 description 18
- 229910052757 nitrogen Inorganic materials 0.000 description 18
- 125000002911 monocyclic heterocycle group Chemical group 0.000 description 17
- 125000004434 sulfur atom Chemical group 0.000 description 16
- 238000012360 testing method Methods 0.000 description 16
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 15
- 239000007924 injection Substances 0.000 description 15
- 238000002347 injection Methods 0.000 description 15
- 239000000047 product Substances 0.000 description 15
- 229910052717 sulfur Inorganic materials 0.000 description 15
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 14
- 229910052799 carbon Inorganic materials 0.000 description 14
- 150000001721 carbon Chemical group 0.000 description 13
- 125000005842 heteroatom Chemical group 0.000 description 12
- 239000012074 organic phase Substances 0.000 description 12
- 125000003118 aryl group Chemical group 0.000 description 11
- 238000011534 incubation Methods 0.000 description 11
- 210000001853 liver microsome Anatomy 0.000 description 11
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 10
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 10
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 10
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 10
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 10
- 238000004440 column chromatography Methods 0.000 description 10
- 238000001959 radiotherapy Methods 0.000 description 10
- 150000002431 hydrogen Chemical class 0.000 description 9
- 230000005764 inhibitory process Effects 0.000 description 9
- 239000002609 medium Substances 0.000 description 9
- 125000004430 oxygen atom Chemical group O* 0.000 description 9
- 239000000843 powder Substances 0.000 description 9
- 238000005160 1H NMR spectroscopy Methods 0.000 description 8
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 8
- PAYRUJLWNCNPSJ-UHFFFAOYSA-N Aniline Chemical compound NC1=CC=CC=C1 PAYRUJLWNCNPSJ-UHFFFAOYSA-N 0.000 description 8
- 238000002474 experimental method Methods 0.000 description 8
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 7
- 229960005277 gemcitabine Drugs 0.000 description 7
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 7
- 239000013641 positive control Substances 0.000 description 7
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 238000002512 chemotherapy Methods 0.000 description 6
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 6
- 125000002883 imidazolyl group Chemical group 0.000 description 6
- 230000002401 inhibitory effect Effects 0.000 description 6
- 239000012071 phase Substances 0.000 description 6
- 210000004881 tumor cell Anatomy 0.000 description 6
- BKWJAKQVGHWELA-UHFFFAOYSA-N 1-[6-(2-hydroxypropan-2-yl)-2-pyridinyl]-6-[4-(4-methyl-1-piperazinyl)anilino]-2-prop-2-enyl-3-pyrazolo[3,4-d]pyrimidinone Chemical compound C1CN(C)CCN1C(C=C1)=CC=C1NC1=NC=C2C(=O)N(CC=C)N(C=3N=C(C=CC=3)C(C)(C)O)C2=N1 BKWJAKQVGHWELA-UHFFFAOYSA-N 0.000 description 5
- WFQDTOYDVUWQMS-UHFFFAOYSA-N 1-fluoro-4-nitrobenzene Chemical compound [O-][N+](=O)C1=CC=C(F)C=C1 WFQDTOYDVUWQMS-UHFFFAOYSA-N 0.000 description 5
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 5
- 230000005971 DNA damage repair Effects 0.000 description 5
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 description 5
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 5
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 5
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 5
- 229950009557 adavosertib Drugs 0.000 description 5
- 239000000706 filtrate Substances 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 210000001589 microsome Anatomy 0.000 description 5
- 239000011259 mixed solution Substances 0.000 description 5
- 239000003208 petroleum Substances 0.000 description 5
- 238000000746 purification Methods 0.000 description 5
- 230000002441 reversible effect Effects 0.000 description 5
- 239000000741 silica gel Substances 0.000 description 5
- 229910002027 silica gel Inorganic materials 0.000 description 5
- 238000012546 transfer Methods 0.000 description 5
- GAWIXWVDTYZWAW-UHFFFAOYSA-N C[CH]O Chemical group C[CH]O GAWIXWVDTYZWAW-UHFFFAOYSA-N 0.000 description 4
- 230000005778 DNA damage Effects 0.000 description 4
- 231100000277 DNA damage Toxicity 0.000 description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 4
- 125000000217 alkyl group Chemical group 0.000 description 4
- 239000002246 antineoplastic agent Substances 0.000 description 4
- 125000005605 benzo group Chemical group 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 239000002775 capsule Substances 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 238000010790 dilution Methods 0.000 description 4
- 239000012895 dilution Substances 0.000 description 4
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 4
- 239000008187 granular material Substances 0.000 description 4
- CATSNJVOTSVZJV-UHFFFAOYSA-N heptan-2-one Chemical compound CCCCCC(C)=O CATSNJVOTSVZJV-UHFFFAOYSA-N 0.000 description 4
- 230000003287 optical effect Effects 0.000 description 4
- 239000002953 phosphate buffered saline Substances 0.000 description 4
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 4
- 125000000168 pyrrolyl group Chemical group 0.000 description 4
- 230000008439 repair process Effects 0.000 description 4
- 241000894007 species Species 0.000 description 4
- 239000000829 suppository Substances 0.000 description 4
- 239000003826 tablet Substances 0.000 description 4
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 4
- 239000012224 working solution Substances 0.000 description 4
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 description 3
- 241000282472 Canis lupus familiaris Species 0.000 description 3
- 102000002427 Cyclin B Human genes 0.000 description 3
- 108010068150 Cyclin B Proteins 0.000 description 3
- 102100032857 Cyclin-dependent kinase 1 Human genes 0.000 description 3
- 101710106279 Cyclin-dependent kinase 1 Proteins 0.000 description 3
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 3
- 241000282414 Homo sapiens Species 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 150000007513 acids Chemical class 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 3
- 230000022131 cell cycle Effects 0.000 description 3
- 229910052801 chlorine Inorganic materials 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 239000003085 diluting agent Substances 0.000 description 3
- 238000001035 drying Methods 0.000 description 3
- 229910052731 fluorine Inorganic materials 0.000 description 3
- 238000009472 formulation Methods 0.000 description 3
- 125000000524 functional group Chemical group 0.000 description 3
- 230000002503 metabolic effect Effects 0.000 description 3
- 238000002156 mixing Methods 0.000 description 3
- 125000002950 monocyclic group Chemical group 0.000 description 3
- 125000003226 pyrazolyl group Chemical group 0.000 description 3
- 238000010791 quenching Methods 0.000 description 3
- 239000007921 spray Substances 0.000 description 3
- BZMMRNKDONDVIB-UHFFFAOYSA-N (1-ethoxycyclopropyl)oxy-trimethylsilane Chemical compound CCOC1(O[Si](C)(C)C)CC1 BZMMRNKDONDVIB-UHFFFAOYSA-N 0.000 description 2
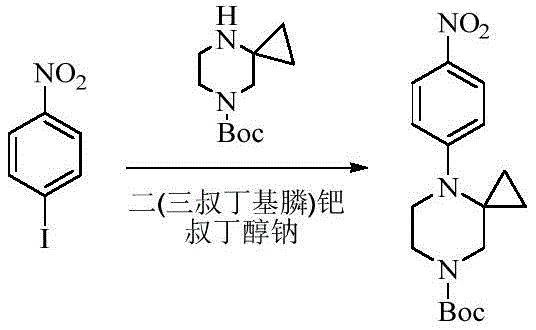
- SCCCFNJTCDSLCY-UHFFFAOYSA-N 1-iodo-4-nitrobenzene Chemical compound [O-][N+](=O)C1=CC=C(I)C=C1 SCCCFNJTCDSLCY-UHFFFAOYSA-N 0.000 description 2
- OXSDDDKLMCHNHF-UHFFFAOYSA-N 2-(6-bromopyridin-2-yl)propan-2-ol Chemical compound CC(C)(O)C1=CC=CC(Br)=N1 OXSDDDKLMCHNHF-UHFFFAOYSA-N 0.000 description 2
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 2
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- IANWOCUEQOUQPB-UHFFFAOYSA-N 4-(6-methyl-2,6-diazaspiro[3.3]heptan-2-yl)aniline Chemical compound CN1CC2(C1)CN(C2)C3=CC=C(C=C3)N IANWOCUEQOUQPB-UHFFFAOYSA-N 0.000 description 2
- RMMHBBLZYIKZCV-UHFFFAOYSA-N 6-methyl-2-(4-nitrophenyl)-2,6-diazaspiro[3.3]heptane Chemical compound CN1CC2(C1)CN(C2)C3=CC=C(C=C3)[N+](=O)[O-] RMMHBBLZYIKZCV-UHFFFAOYSA-N 0.000 description 2
- JKBQCALHIQOSTC-UHFFFAOYSA-N 6-methylsulfanyl-2-prop-2-enyl-1h-pyrazolo[3,4-d]pyrimidin-3-one Chemical compound CSC1=NC=C2C(=O)N(CC=C)NC2=N1 JKBQCALHIQOSTC-UHFFFAOYSA-N 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 2
- 241000282693 Cercopithecidae Species 0.000 description 2
- 229910021595 Copper(I) iodide Inorganic materials 0.000 description 2
- 108020004414 DNA Proteins 0.000 description 2
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 2
- 230000004668 G2/M phase Effects 0.000 description 2
- 101000777293 Homo sapiens Serine/threonine-protein kinase Chk1 Proteins 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 2
- ACFIXJIJDZMPPO-NNYOXOHSSA-N NADPH Chemical compound C1=CCC(C(=O)N)=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OC[C@@H]2[C@H]([C@@H](OP(O)(O)=O)[C@@H](O2)N2C3=NC=NC(N)=C3N=C2)O)O1 ACFIXJIJDZMPPO-NNYOXOHSSA-N 0.000 description 2
- 108091000080 Phosphotransferase Proteins 0.000 description 2
- 102000001253 Protein Kinase Human genes 0.000 description 2
- 102100031081 Serine/threonine-protein kinase Chk1 Human genes 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical class [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 102000004142 Trypsin Human genes 0.000 description 2
- 108090000631 Trypsin Proteins 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 2
- 125000004414 alkyl thio group Chemical group 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 239000012298 atmosphere Substances 0.000 description 2
- 125000002785 azepinyl group Chemical group 0.000 description 2
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 2
- ACFIXJIJDZMPPO-UHFFFAOYSA-N beta-NADPH Natural products C1=CCC(C(=O)N)=CN1C1C(O)C(O)C(COP(O)(=O)OP(O)(=O)OCC2C(C(OP(O)(O)=O)C(O2)N2C3=NC=NC(N)=C3N=C2)O)O1 ACFIXJIJDZMPPO-UHFFFAOYSA-N 0.000 description 2
- 229910052794 bromium Inorganic materials 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 230000004663 cell proliferation Effects 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 239000000460 chlorine Substances 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- LSXDOTMGLUJQCM-UHFFFAOYSA-M copper(i) iodide Chemical compound I[Cu] LSXDOTMGLUJQCM-UHFFFAOYSA-M 0.000 description 2
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 2
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 230000007547 defect Effects 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 239000012091 fetal bovine serum Substances 0.000 description 2
- 239000012065 filter cake Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000011737 fluorine Substances 0.000 description 2
- 239000008098 formaldehyde solution Substances 0.000 description 2
- 238000001640 fractional crystallisation Methods 0.000 description 2
- 239000012458 free base Substances 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 125000002632 imidazolidinyl group Chemical group 0.000 description 2
- 239000000463 material Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 125000002757 morpholinyl group Chemical group 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 230000036961 partial effect Effects 0.000 description 2
- 230000037361 pathway Effects 0.000 description 2
- 102000020233 phosphotransferase Human genes 0.000 description 2
- 239000006187 pill Substances 0.000 description 2
- 125000004193 piperazinyl group Chemical group 0.000 description 2
- 125000004928 piperidonyl group Chemical group 0.000 description 2
- 125000005936 piperidyl group Chemical group 0.000 description 2
- 229910000027 potassium carbonate Inorganic materials 0.000 description 2
- 108060006633 protein kinase Proteins 0.000 description 2
- 230000002685 pulmonary effect Effects 0.000 description 2
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 2
- 125000004943 pyrimidin-6-yl group Chemical group N1=CN=CC=C1* 0.000 description 2
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 2
- 230000008929 regeneration Effects 0.000 description 2
- 238000011069 regeneration method Methods 0.000 description 2
- 238000005070 sampling Methods 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- BEOOHQFXGBMRKU-UHFFFAOYSA-N sodium cyanoborohydride Chemical compound [Na+].[B-]C#N BEOOHQFXGBMRKU-UHFFFAOYSA-N 0.000 description 2
- MFRIHAYPQRLWNB-UHFFFAOYSA-N sodium tert-butoxide Chemical compound [Na+].CC(C)(C)[O-] MFRIHAYPQRLWNB-UHFFFAOYSA-N 0.000 description 2
- 239000012321 sodium triacetoxyborohydride Substances 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 239000000725 suspension Substances 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- FMTWZULNMBYWPQ-UHFFFAOYSA-N tert-butyl 4-oxo-3,6-diazabicyclo[3.1.1]heptane-6-carboxylate Chemical compound C1C2N(C(=O)OC(C)(C)C)C1CNC2=O FMTWZULNMBYWPQ-UHFFFAOYSA-N 0.000 description 2
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 125000001984 thiazolidinyl group Chemical group 0.000 description 2
- 238000004809 thin layer chromatography Methods 0.000 description 2
- 125000004205 trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 2
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 2
- 239000012588 trypsin Substances 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- 125000006273 (C1-C3) alkyl group Chemical group 0.000 description 1
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 1
- 125000006569 (C5-C6) heterocyclic group Chemical group 0.000 description 1
- 125000004502 1,2,3-oxadiazolyl group Chemical group 0.000 description 1
- 125000004529 1,2,3-triazinyl group Chemical group N1=NN=C(C=C1)* 0.000 description 1
- 125000001399 1,2,3-triazolyl group Chemical group N1N=NC(=C1)* 0.000 description 1
- 125000004504 1,2,4-oxadiazolyl group Chemical group 0.000 description 1
- 125000001376 1,2,4-triazolyl group Chemical group N1N=C(N=C1)* 0.000 description 1
- 125000004506 1,2,5-oxadiazolyl group Chemical group 0.000 description 1
- 125000005918 1,2-dimethylbutyl group Chemical group 0.000 description 1
- 125000001781 1,3,4-oxadiazolyl group Chemical group 0.000 description 1
- 125000003363 1,3,5-triazinyl group Chemical group N1=C(N=CN=C1)* 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- WORJRXHJTUTINR-UHFFFAOYSA-N 1,4-dioxane;hydron;chloride Chemical compound Cl.C1COCCO1 WORJRXHJTUTINR-UHFFFAOYSA-N 0.000 description 1
- 125000005940 1,4-dioxanyl group Chemical group 0.000 description 1
- 125000000196 1,4-pentadienyl group Chemical group [H]C([*])=C([H])C([H])([H])C([H])=C([H])[H] 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000006039 1-hexenyl group Chemical group 0.000 description 1
- 125000006023 1-pentenyl group Chemical group 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- SGTNSNPWRIOYBX-UHFFFAOYSA-N 2-(3,4-dimethoxyphenyl)-5-{[2-(3,4-dimethoxyphenyl)ethyl](methyl)amino}-2-(propan-2-yl)pentanenitrile Chemical compound C1=C(OC)C(OC)=CC=C1CCN(C)CCCC(C#N)(C(C)C)C1=CC=C(OC)C(OC)=C1 SGTNSNPWRIOYBX-UHFFFAOYSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- GXIMUJOQZCMEFY-UHFFFAOYSA-N 2-methyl-2,5-diazabicyclo[2.2.2]octane Chemical compound C1CC2N(C)CC1NC2 GXIMUJOQZCMEFY-UHFFFAOYSA-N 0.000 description 1
- YFAQWQPCBYBDEK-UHFFFAOYSA-N 2-methyl-2,6-diazaspiro[3.3]heptane;dihydrochloride Chemical compound Cl.Cl.C1N(C)CC11CNC1 YFAQWQPCBYBDEK-UHFFFAOYSA-N 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- 125000006024 2-pentenyl group Chemical group 0.000 description 1
- 125000000850 2H-chromenyl group Chemical group O1C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000000844 2H-pyran-2-onyl group Chemical group O1C(C(=CC=C1)*)=O 0.000 description 1
- 125000001698 2H-pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- QTFBPMJATBTHSY-UHFFFAOYSA-N 2h-furo[3,2-b]pyrrole Chemical compound C1=NC2=CCOC2=C1 QTFBPMJATBTHSY-UHFFFAOYSA-N 0.000 description 1
- 125000006041 3-hexenyl group Chemical group 0.000 description 1
- 125000003542 3-methylbutan-2-yl group Chemical group [H]C([H])([H])C([H])(*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- QCQCHGYLTSGIGX-GHXANHINSA-N 4-[[(3ar,5ar,5br,7ar,9s,11ar,11br,13as)-5a,5b,8,8,11a-pentamethyl-3a-[(5-methylpyridine-3-carbonyl)amino]-2-oxo-1-propan-2-yl-4,5,6,7,7a,9,10,11,11b,12,13,13a-dodecahydro-3h-cyclopenta[a]chrysen-9-yl]oxy]-2,2-dimethyl-4-oxobutanoic acid Chemical compound N([C@@]12CC[C@@]3(C)[C@]4(C)CC[C@H]5C(C)(C)[C@@H](OC(=O)CC(C)(C)C(O)=O)CC[C@]5(C)[C@H]4CC[C@@H]3C1=C(C(C2)=O)C(C)C)C(=O)C1=CN=CC(C)=C1 QCQCHGYLTSGIGX-GHXANHINSA-N 0.000 description 1
- 125000001819 4H-chromenyl group Chemical group O1C(=CCC2=CC=CC=C12)* 0.000 description 1
- XAUDJQYHKZQPEU-KVQBGUIXSA-N 5-aza-2'-deoxycytidine Chemical compound O=C1N=C(N)N=CN1[C@@H]1O[C@H](CO)[C@@H](O)C1 XAUDJQYHKZQPEU-KVQBGUIXSA-N 0.000 description 1
- RDHOEMPRFDWIHL-UHFFFAOYSA-N 5h-thieno[3,2-c]pyrazole Chemical compound N1=NC2=CCSC2=C1 RDHOEMPRFDWIHL-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 238000003727 ADP Glo Kinase Assay Methods 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- NLXLAEXVIDQMFP-UHFFFAOYSA-N Ammonia chloride Chemical class [NH4+].[Cl-] NLXLAEXVIDQMFP-UHFFFAOYSA-N 0.000 description 1
- ASVYXWZOBCRSFB-UHFFFAOYSA-N C(C)S(=O)(O)=S.OCCN1CCNCC1 Chemical compound C(C)S(=O)(O)=S.OCCN1CCNCC1 ASVYXWZOBCRSFB-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 208000009458 Carcinoma in Situ Diseases 0.000 description 1
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 108010024986 Cyclin-Dependent Kinase 2 Proteins 0.000 description 1
- 102100036239 Cyclin-dependent kinase 2 Human genes 0.000 description 1
- 230000033616 DNA repair Effects 0.000 description 1
- 230000004543 DNA replication Effects 0.000 description 1
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 1
- 102100030011 Endoribonuclease Human genes 0.000 description 1
- 101710199605 Endoribonuclease Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- YCKRFDGAMUMZLT-UHFFFAOYSA-N Fluorine atom Chemical compound [F] YCKRFDGAMUMZLT-UHFFFAOYSA-N 0.000 description 1
- 230000008051 G1/S transition checkpoint Effects 0.000 description 1
- 230000020172 G2/M transition checkpoint Effects 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000624643 Homo sapiens M-phase inducer phosphatase 3 Proteins 0.000 description 1
- 101000580039 Homo sapiens Ras-specific guanine nucleotide-releasing factor 1 Proteins 0.000 description 1
- 101000621390 Homo sapiens Wee1-like protein kinase Proteins 0.000 description 1
- 102100023330 M-phase inducer phosphatase 3 Human genes 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 102000005877 Peptide Initiation Factors Human genes 0.000 description 1
- 108010044843 Peptide Initiation Factors Proteins 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 230000018199 S phase Effects 0.000 description 1
- 241000235347 Schizosaccharomyces pombe Species 0.000 description 1
- 101710113029 Serine/threonine-protein kinase Proteins 0.000 description 1
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 1
- UIIMBOGNXHQVGW-DEQYMQKBSA-M Sodium bicarbonate-14C Chemical class [Na+].O[14C]([O-])=O UIIMBOGNXHQVGW-DEQYMQKBSA-M 0.000 description 1
- JLRGJRBPOGGCBT-UHFFFAOYSA-N Tolbutamide Chemical compound CCCCNC(=O)NS(=O)(=O)C1=CC=C(C)C=C1 JLRGJRBPOGGCBT-UHFFFAOYSA-N 0.000 description 1
- OKJPEAGHQZHRQV-UHFFFAOYSA-N Triiodomethane Natural products IC(I)I OKJPEAGHQZHRQV-UHFFFAOYSA-N 0.000 description 1
- 102100023037 Wee1-like protein kinase Human genes 0.000 description 1
- DGEZNRSVGBDHLK-UHFFFAOYSA-N [1,10]phenanthroline Chemical compound C1=CN=C2C3=NC=CC=C3C=CC2=C1 DGEZNRSVGBDHLK-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 239000003513 alkali Substances 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 150000001449 anionic compounds Chemical class 0.000 description 1
- 229940041181 antineoplastic drug Drugs 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 239000007864 aqueous solution Substances 0.000 description 1
- 125000002393 azetidinyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 125000000499 benzofuranyl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000004619 benzopyranyl group Chemical group O1C(C=CC2=C1C=CC=C2)* 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 1
- 125000004935 benzoxazolinyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 229910002091 carbon monoxide Inorganic materials 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 150000001767 cationic compounds Chemical class 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000036755 cellular response Effects 0.000 description 1
- 239000012295 chemical reaction liquid Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- OTAFHZMPRISVEM-UHFFFAOYSA-N chromone Chemical compound C1=CC=C2C(=O)C=COC2=C1 OTAFHZMPRISVEM-UHFFFAOYSA-N 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
- 230000001276 controlling effect Effects 0.000 description 1
- 239000012050 conventional carrier Substances 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical compound C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 125000001047 cyclobutenyl group Chemical group C1(=CCC1)* 0.000 description 1
- 125000002197 cyclohepta-2,4,6-trienyl group Chemical group 0.000 description 1
- 125000001162 cycloheptenyl group Chemical group C1(=CCCCCC1)* 0.000 description 1
- MGNZXYYWBUKAII-UHFFFAOYSA-N cyclohexa-1,3-diene Chemical compound C1CC=CC=C1 MGNZXYYWBUKAII-UHFFFAOYSA-N 0.000 description 1
- UVJHQYIOXKWHFD-UHFFFAOYSA-N cyclohexa-1,4-diene Chemical compound C1C=CCC=C1 UVJHQYIOXKWHFD-UHFFFAOYSA-N 0.000 description 1
- 125000001944 cycloocta-2,4,6-trienyl group Chemical group [H]\C1=C(/[H])\C(\[H])=C([H])/C([H])(*)C([H])([H])\C([H])=C/1\[H] 0.000 description 1
- 125000000522 cyclooctenyl group Chemical group C1(=CCCCCCC1)* 0.000 description 1
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 description 1
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 1
- 238000007405 data analysis Methods 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 125000004856 decahydroquinolinyl group Chemical group N1(CCCC2CCCCC12)* 0.000 description 1
- 229960003603 decitabine Drugs 0.000 description 1
- 230000001934 delay Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 125000005054 dihydropyrrolyl group Chemical group [H]C1=C([H])C([H])([H])C([H])([H])N1* 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 230000005782 double-strand break Effects 0.000 description 1
- 230000000857 drug effect Effects 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 150000002085 enols Chemical group 0.000 description 1
- 230000002255 enzymatic effect Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- SNNHLSHDDGJVDM-UHFFFAOYSA-N ethyl 4-chloro-2-methylsulfanylpyrimidine-5-carboxylate Chemical compound CCOC(=O)C1=CN=C(SC)N=C1Cl SNNHLSHDDGJVDM-UHFFFAOYSA-N 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 239000013022 formulation composition Substances 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 230000004927 fusion Effects 0.000 description 1
- OKKDEIYWILRZIA-OSZBKLCCSA-N gemcitabine hydrochloride Chemical compound [H+].[Cl-].O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 OKKDEIYWILRZIA-OSZBKLCCSA-N 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 230000002440 hepatic effect Effects 0.000 description 1
- 125000000592 heterocycloalkyl group Chemical group 0.000 description 1
- 229940125697 hormonal agent Drugs 0.000 description 1
- TUJKJAMUKRIRHC-UHFFFAOYSA-N hydroxyl Chemical compound [OH] TUJKJAMUKRIRHC-UHFFFAOYSA-N 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 201000004933 in situ carcinoma Diseases 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 238000009776 industrial production Methods 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- INQOMBQAUSQDDS-UHFFFAOYSA-N iodomethane Chemical compound IC INQOMBQAUSQDDS-UHFFFAOYSA-N 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 230000002045 lasting effect Effects 0.000 description 1
- 231100000518 lethal Toxicity 0.000 description 1
- 230000001665 lethal effect Effects 0.000 description 1
- 238000012417 linear regression Methods 0.000 description 1
- 239000000865 liniment Substances 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000001294 liquid chromatography-tandem mass spectrometry Methods 0.000 description 1
- 239000007791 liquid phase Substances 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- NXPHGHWWQRMDIA-UHFFFAOYSA-M magnesium;carbanide;bromide Chemical compound [CH3-].[Mg+2].[Br-] NXPHGHWWQRMDIA-UHFFFAOYSA-M 0.000 description 1
- 230000036210 malignancy Effects 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 208000011645 metastatic carcinoma Diseases 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- OKKJLVBELUTLKV-VMNATFBRSA-N methanol-d1 Chemical compound [2H]OC OKKJLVBELUTLKV-VMNATFBRSA-N 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- SGNCOKUHMXLGAH-UHFFFAOYSA-N methyl 6-bromopyridine-2-carboxylate Chemical compound COC(=O)C1=CC=CC(Br)=N1 SGNCOKUHMXLGAH-UHFFFAOYSA-N 0.000 description 1
- 230000003228 microsomal effect Effects 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 230000035772 mutation Effects 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001624 naphthyl group Chemical group 0.000 description 1
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 1
- 230000035407 negative regulation of cell proliferation Effects 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000012148 non-surgical treatment Methods 0.000 description 1
- 238000011275 oncology therapy Methods 0.000 description 1
- 229940100688 oral solution Drugs 0.000 description 1
- 229940100692 oral suspension Drugs 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000002891 organic anions Chemical class 0.000 description 1
- 150000002892 organic cations Chemical class 0.000 description 1
- 229940127084 other anti-cancer agent Drugs 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000000160 oxazolidinyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 125000005740 oxycarbonyl group Chemical group [*:1]OC([*:2])=O 0.000 description 1
- MXQOYLRVSVOCQT-UHFFFAOYSA-N palladium;tritert-butylphosphane Chemical compound [Pd].CC(C)(C)P(C(C)(C)C)C(C)(C)C.CC(C)(C)P(C(C)(C)C)C(C)(C)C MXQOYLRVSVOCQT-UHFFFAOYSA-N 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 239000008024 pharmaceutical diluent Substances 0.000 description 1
- 125000004934 phenanthridinyl group Chemical group C1(=CC=CC2=NC=C3C=CC=CC3=C12)* 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 230000000865 phosphorylative effect Effects 0.000 description 1
- 125000004592 phthalazinyl group Chemical group C1(=NN=CC2=CC=CC=C12)* 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 238000007747 plating Methods 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 238000011533 pre-incubation Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 description 1
- 125000000561 purinyl group Chemical group N1=C(N=C2N=CNC2=C1)* 0.000 description 1
- RQGPLDBZHMVWCH-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole Chemical compound C1=NC2=CC=NC2=C1 RQGPLDBZHMVWCH-UHFFFAOYSA-N 0.000 description 1
- GZTPJDLYPMPRDF-UHFFFAOYSA-N pyrrolo[3,2-c]pyrazole Chemical compound N1=NC2=CC=NC2=C1 GZTPJDLYPMPRDF-UHFFFAOYSA-N 0.000 description 1
- 230000000171 quenching effect Effects 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 238000011127 radiochemotherapy Methods 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000002271 resection Methods 0.000 description 1
- 230000000979 retarding effect Effects 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000012279 sodium borohydride Substances 0.000 description 1
- 229910000033 sodium borohydride Inorganic materials 0.000 description 1
- 239000012312 sodium hydride Substances 0.000 description 1
- 229910000104 sodium hydride Inorganic materials 0.000 description 1
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 description 1
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 1
- 239000002904 solvent Substances 0.000 description 1
- 238000009987 spinning Methods 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 238000000967 suction filtration Methods 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- UXAWXZDXVOYLII-UHFFFAOYSA-N tert-butyl 2,5-diazabicyclo[2.2.1]heptane-2-carboxylate Chemical compound C1C2N(C(=O)OC(C)(C)C)CC1NC2 UXAWXZDXVOYLII-UHFFFAOYSA-N 0.000 description 1
- HNINFCBLGHCFOJ-UHFFFAOYSA-N tert-butyl 3,8-diazabicyclo[3.2.1]octane-8-carboxylate Chemical compound C1NCC2CCC1N2C(=O)OC(C)(C)C HNINFCBLGHCFOJ-UHFFFAOYSA-N 0.000 description 1
- XNLYPHAMXHERHS-UHFFFAOYSA-N tert-butyl 4,7-diazaspiro[2.5]octane-4-carboxylate Chemical compound CC(C)(C)OC(=O)N1CCNCC11CC1 XNLYPHAMXHERHS-UHFFFAOYSA-N 0.000 description 1
- XXKCYKIJWLFVDG-UHFFFAOYSA-N tert-butyl 4,7-diazaspiro[2.5]octane-7-carboxylate Chemical compound C1N(C(=O)OC(C)(C)C)CCNC11CC1 XXKCYKIJWLFVDG-UHFFFAOYSA-N 0.000 description 1
- OGPRTHQPTQFNAX-UHFFFAOYSA-N tert-butyl 7-(4-nitrophenyl)-4,7-diazaspiro[2.5]octane-4-carboxylate Chemical compound [N+](=O)([O-])C1=CC=C(C=C1)N1CCN(C2(CC2)C1)C(=O)OC(C)(C)C OGPRTHQPTQFNAX-UHFFFAOYSA-N 0.000 description 1
- ZTWGIZOCAXOUOE-UHFFFAOYSA-N tert-butyl n-amino-n-prop-2-enylcarbamate Chemical compound CC(C)(C)OC(=O)N(N)CC=C ZTWGIZOCAXOUOE-UHFFFAOYSA-N 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 239000012085 test solution Substances 0.000 description 1
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 description 1
- RWRDLPDLKQPQOW-UHFFFAOYSA-N tetrahydropyrrole Substances C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 1
- 125000005958 tetrahydrothienyl group Chemical group 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 229960005371 tolbutamide Drugs 0.000 description 1
- 229960001722 verapamil Drugs 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 238000003260 vortexing Methods 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 210000005253 yeast cell Anatomy 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D519/00—Heterocyclic compounds containing more than one system of two or more relevant hetero rings condensed among themselves or condensed with a common carbocyclic ring system not provided for in groups C07D453/00 or C07D455/00
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The invention belongs to the technical field of medicines, and particularly relates to a dihydropyrazolopyrimidinone derivative compound serving as a Wee1 kinase inhibitor, a pharmaceutically acceptable salt or a stereoisomer thereof, a pharmaceutical composition and a preparation containing the compound, the pharmaceutically acceptable salt or the stereoisomer thereof, a method for preparing the compound, the pharmaceutically acceptable salt or the stereoisomer thereof, and application of the compound, the pharmaceutically acceptable salt or the stereoisomer thereof.
Description
Technical Field
The invention belongs to the technical field of medicines, and particularly relates to a dihydropyrazolopyrimidinone derivative compound, pharmaceutically acceptable salt and a stereoisomer thereof, a pharmaceutical composition and a preparation containing the compound, the pharmaceutically acceptable salt and the stereoisomer thereof, a method for preparing the compound, the pharmaceutically acceptable salt and the stereoisomer thereof, and application of the compound, the pharmaceutically acceptable salt and the stereoisomer thereof.
Background
Cancer is a malignant disease which is difficult to treat all over the world, and has high treatment difficulty and high mortality rate. The recent global tumor statistics in 2018 show that, to date, 1819 ten thousand new cases of cancer and 960 ten thousand cases of cancer death are estimated globally. In 2019, month 1, the national cancer center released the latest national cancer statistics for the first phase. About 392.9 million people develop national malignancies each year, with an average of over 1 million diagnosed as cancer per day and 7.5 diagnosed as cancer per minute. Cancer has become a major disease affecting the health of residents in China, and the prevention and treatment of cancer also faces a severe form.
Currently, radiotherapy and chemotherapy are the most effective means of treating cancer in addition to surgical resection, while radiotherapy is the most effective non-surgical treatment for malignant tumors. Both radiation and a considerable number of anticancer drugs can cause DNA damage. After DNA damage, a series of cellular responses such as damaged DNA repair can be initiated to improve the survival of tumor cells, which is also one of the mechanisms of tumor cells against chemoradiotherapy. If the damaged DNA is not repaired in time and intact, the tumor cells die due to apoptosis or/and mitotic disorders. Therefore, by inhibiting the repair of such DNA damage, the sensitivity of cancer cells to radiotherapy and chemotherapy can be improved, and the proliferation of cells can be inhibited.
Wee1 protein kinase is a member of the serine/threonine protein kinase family, and was first isolated by Nurse et al in fission yeast cells (S.pombe). In humans, Wee1 contains 647 amino acids with a molecular weight of 96 kDa. In the DNA single-strand damage repair channel, Wee1 is positioned at the downstream of an ATR signal channel, after the ATR signal channel is activated, CHK1 is phosphorylated, activated CHK1 activates Wee1, CDC25 is inhibited (the phosphorylation of CDK1/Cyclin B complex is relieved, the activity of regulating and controlling the cell cycle is recovered), CDK1/Cyclin B is further phosphorylated, the CDK1/Cyclin B complex enters an inactive state, the cell cycle is blocked in a G2/M phase, and the time is gained for DNA damage repair; in addition, Weel can also regulate the repair of DNA double strand breaks that occur during DNA replication by phosphorylating CDK2, retarding the S phase of the cell cycle.
In the entire DDR (DNA damage repair) pathway, Wee1 plays a role primarily at the G2/M checkpoint. For p53 mutant tumor cells, the G2/M phase checkpoint is more dependent on the G1/S checkpoint defect itself to repair DNA damage, and the Wee1 inhibitor is more sensitive to p53 mutant tumor cells in terms of mechanism. Furthermore, Wee1 inhibitors can also be combined with other targets on the DDR pathway, such as ATM defects/mutations in tumor cells, combination with ATR inhibitors, etc., to more completely block DNA damage repair, achieving a "synthetic lethal" effect.
In conclusion, the Wee1 kinase inhibitor can not only synergistically enhance the effects of radiotherapy and chemotherapy, effectively inhibit tumor growth, but also reduce damage to normal cells and reduce side effects. At present, the research on the target medicine is still in the clinical experimental stage, and no medicine is on the market. Therefore, the development of a high-efficiency Wee1 kinase inhibitor has important clinical significance, and has wide market prospect in single use or combined use with other medicines.
Disclosure of Invention
The technical problem to be solved by the invention is to provide a dihydropyrazolopyrimidinone derivative compound which has a novel structure and has an inhibitory activity on Wee 1. Furthermore, the compounds can be used for inhibiting the activity of Wee1 kinase, thereby enhancing the immunity of organisms to tumors. Furthermore, the compounds can be used for treating one or more diseases mediated by Wee1, especially cancers. The compounds have good inhibition effect on various cancer cells, and have higher exposure and better in-vivo drug effect in organisms.
The technical scheme of the invention is as follows:
in one aspect, the present invention provides a compound represented by the following general formula (I), a pharmaceutically acceptable salt thereof, or a stereoisomer thereof,
wherein,
X1、X2、X3are each independently selected from-C (R)2) -or-N-;
X4、X5each independently selected from-CH-or-N-;
each L is independently selected from-C (R)3)(R4)-、-O-、-N(R5) -or-S-;
ring A is selected from a 3-10 membered cycloalkyl, 3-10 membered heterocyclyl, 6-10 membered aryl or 5-10 membered heteroaryl optionally substituted with 1-4 of Q1;
ring B is selected from 3-10 membered cycloalkyl, 3-10 membered heterocyclyl, 6-10 membered aryl or 5-10 membered heteroaryl optionally substituted with 1-4Q 2;
ring C is selected from 5-15 membered bridged ring group, 5-10 membered spiro ring group, 5-15 membered bridged ring group or 5-10 membered spiroheterocyclic group optionally substituted with 1-4 of Q3, wherein ring carbon atoms in said 5-15 membered bridged ring group, 5-10 membered spiro ring group, 5-15 membered bridged ring group or 5-10 membered spiroheterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q1 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
each Q2, each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, halo C1-6Alkoxy, halo C1-6Alkylthio, hydroxy C1-6Alkoxy, hydroxy C1-6Alkylthio, amino C1-6Alkoxy radicalRadical, amino C1-6Alkylthio, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
R1selected from C optionally substituted by substituents1-6Alkyl radical, C2-6Alkenyl or C2-6Alkynyl, the substituents are respectively and independently selected from halogen, hydroxyl, amino, carboxyl, cyano, C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkoxy or C1-6An alkylcarbonyl group;
R2、R3、R4、R5each independently selected from hydrogen, halogen, hydroxyl, amino, carboxyl and C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkoxy or C1-6An alkylcarbonyl group;
m and n are respectively and independently selected from 0, 1,2 or 3.
In certain embodiments, the compound, a pharmaceutically acceptable salt thereof, or a stereoisomer thereof, wherein,
ring a is selected from 3-8 membered monocyclic cycloalkyl, 3-8 membered monocyclic heterocyclyl, phenyl or 5-8 membered monocyclic heteroaryl, optionally substituted with 1-4Q 1;
each Q1 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
ring B is selected from phenyl or 5-8 membered mono heteroaryl optionally substituted with 1-4Q 2;
each Q2 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, halo C1-6Alkoxy, halo C1-6Alkylthio, hydroxy C1-6Alkoxy, hydroxy C1-6Alkylthio, amino C1-6Alkoxy, amino C1-6An alkylthio group;
ring C is selected from 5-15 membered bridged heterocyclic group or 5-10 membered spiroheterocyclic group optionally substituted with 1-4 of Q3, wherein ring-forming carbon atoms in said 5-15 membered bridged heterocyclic group or 5-10 membered spiroheterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, halo C1-6Alkoxy, halo C1-6Alkylthio, hydroxy C1-6Alkoxy, hydroxy C1-6Alkylthio, amino C1-6Alkoxy or amino C1-6Alkylthio, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
R1selected from C optionally substituted by substituents1-6Alkyl radical, C2-6Alkenyl or C2-6Alkynyl, the substituents are respectively and independently selected from halogen, hydroxyl, amino, carboxyl, cyano, C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkoxy or C1-6An alkylcarbonyl group;
R2、R3、R4、R5each independently selected from hydrogen, halogen, hydroxyl, amino, carboxyl and C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkoxy or C1-6An alkylcarbonyl group;
m and n are respectively and independently selected from 0, 1 or 2.
In certain embodiments, ring a is selected from phenyl or 5-6 membered nitrogen containing monoheteroaryl optionally substituted with 1-3Q 1;
each Q1 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
in certain embodiments, ring a is selected from phenyl or 5-6 membered nitrogen containing monoheteroaryl substituted with 1Q 1;
preferably, ring a is selected from phenyl, pyrrolyl, imidazolyl, pyrazolyl, pyridyl, pyrimidinyl, pyrazinyl or pyridazinyl substituted by 1Q 1; q1 is selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl.
In certain embodiments, ring a is selected from phenyl, pyrrolyl, imidazolyl, pyrazolyl, pyridyl, pyrimidinyl, pyrazinyl, or pyridazinyl substituted with 1Q 1; q1 is selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-6 membered cycloalkyl, - (CH)2)m-3-6 membered heterocycloalkyl, - (CH)2)m-5-6 membered heteroaryl or- (CH)2)m-6-8 membered aryl.
In certain embodiments, ring a is selected from pyridinyl, pyrimidinyl, pyrazinyl, or pyridazinyl substituted with 1Q 1; q1 is selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, wherein the substituents are respectively and independently selected from halogen, hydroxyl, amino, nitro, cyano or carboxyl.
In certain embodiments, ring B is selected from phenyl or 5-6 membered monoheteroaryl optionally substituted with 1 or 2Q 2; each Q2 is independently selected from halogen, hydroxy, amino, nitro, cyano, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or amino C1-6An alkoxy group.
In certain embodiments, ring B is selected from phenyl, pyridyl, pyrazinyl, or pyridazinyl optionally substituted with 1 or 2Q 2; each Q2 is independently selected from halogen, hydroxy, amino, nitro, cyano, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxyC1-6Alkoxy or amino C1-6An alkoxy group.
In certain embodiments, ring B is selected from phenyl, pyridyl, pyrazinyl, or pyridazinyl optionally substituted with 1 or 2Q 2; each Q2 is independently selected from fluoro, chloro, bromo, iodo, cyano, methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, methylamino, ethylamino, propylamino, isopropylamino, dimethylamino, diethylamino, monofluoromethyl, difluoromethyl, trifluoromethyl, methoxy, ethoxy, propoxy, isopropoxy, monofluoromethoxy, difluoromethoxy, or trifluoromethoxy.
In certain embodiments, ring a is selected from phenyl or 5-6 membered nitrogen containing monoheteroaryl optionally substituted with 1-3Q 1;
each Q1 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
ring B is selected from phenyl or 5-6 membered monoheteroaryl optionally substituted with 1 or 2Q 2; each Q2 is independently selected from halogen, hydroxy, amino, nitro, cyano, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or amino C1-6An alkoxy group.
In certain embodiments, ring C is selected from a 5-15 membered nitrogen-containing bridged heterocyclic group or a 5-10 membered nitrogen-containing spiro heterocyclic group, optionally substituted with 1-4Q 3, wherein the ring carbon atoms in the 5-15 membered nitrogen-containing bridged heterocyclic group or the 5-10 membered nitrogen-containing spiro heterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, halo C1-6Alkoxy, halo C1-6Alkylthio, hydroxy C1-6Alkoxy, hydroxy C1-6Alkylthio, amino C1-6Alkoxy or amino C1-6An alkylthio group.
In certain embodiments, ring C is selected from 7-9 membered nitrogen-containing bridged heterocyclic group or 7-9 membered nitrogen-containing spiro heterocyclic group, optionally substituted with 1-2Q 3, wherein the ring carbon atoms in said 7-9 membered nitrogen-containing bridged heterocyclic group or 7-9 membered nitrogen-containing spiro heterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy or hydroxy C1-6An alkoxy group.
In certain embodiments, ring C is selected from 7-8 membered nitrogen-containing bridged heterocyclic group or 7-8 membered nitrogen-containing spiro heterocyclic group, optionally substituted with 1-2Q 3, wherein the ring carbon atoms in said 7-8 membered nitrogen-containing bridged heterocyclic group or 7-8 membered nitrogen-containing spiro heterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from a 7-8 membered 1-2 nitrogen containing bridged heterocyclic group optionally substituted with 1-2Q 3 or a 7-8 membered 1-2 nitrogen containing spiroheterocyclic group, wherein the ring-forming carbon atoms in said bridged heterocyclic group or spiroheterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxyAmino, nitro, cyano, carboxyl, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from a 7-8 membered 2 nitrogen-containing bridged heterocyclyl group optionally substituted with 1-2Q 3, or a 7-8 membered 2 nitrogen-containing spiroheterocyclyl group, wherein the ring-forming carbon atoms in said bridged heterocyclyl or spiroheterocyclyl group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3:
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from 7-9 membered nitrogen-containing bridged heterocyclic groups optionally substituted with 1-2 of Q3, wherein the ring-forming carbon atoms of said 7-9 membered nitrogen-containing bridged heterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl. In certain embodiments, ring C is selected from 7-8 membered 1-containing rings optionally substituted with 1-2Q 3-a bridged heterocyclic group of 2 nitrogens, wherein the ring-forming carbon atoms of said bridged heterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 2-azabicyclo [2.2.1] heptanyl, 7-azabicyclo [2.2.1] heptanyl, 3-azabicyclo [3.2.1] octanyl, 8-azabicyclo [3.2.1] octanyl, 2-azabicyclo [2.2.2] octanyl, 2, 5-diazabicyclo [2.2.1] heptanyl, 3, 6-diazabicyclo [3.1.1] heptanyl, 3, 8-diazabicyclo [3.2.1] octanyl, 2-oxa-5-azabicyclo [2.2.1] heptanyl, 8-oxa-3-azabicyclo [3.2.1] octanyl, 3, 8-diazabicyclo [3.2.1] octan-6-enyl or 3, 9-diazabicyclo [3.3.1] nonanyl, wherein the oxycarbonyl group forms a ring carbon atom;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3:
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-to 4-memberedA cycloalkyl group.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 2-azabicyclo [2.2.1] heptanyl, 7-azabicyclo [2.2.1] heptanyl, 3-azabicyclo [3.2.1] octanyl, 8-azabicyclo [3.2.1] octanyl, 2-azabicyclo [2.2.2] octanyl, 2, 5-diazabicyclo [2.2.1] heptanyl, 3, 6-diazabicyclo [3.1.1] heptanyl or 3, 8-diazabicyclo [3.2.1] octanyl, wherein the groups are optionally substituted at a ring carbon atom by oxygen to form a carbonyl group;
each Q3 is independently selected from fluoro, chloro, hydroxy, amino, nitro, cyano, carboxy, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, hydroxymethyl, hydroxyethyl, hydroxyisopropyl, trifluoromethyl, trifluoromethoxy, or cyclopropyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 2, 5-diazabicyclo [2.2.2] octane-yl, 2, 5-diazabicyclo [2.2.1] heptylalkyl, 3, 6-diazabicyclo [3.1.1] heptylalkyl or 3, 8-diazabicyclo [3.2.1] octane-yl, wherein the groups are optionally substituted with oxygen to form a carbonyl group at a ring carbon atom;
each Q3 is independently selected from fluoro, chloro, hydroxy, amino, nitro, cyano, carboxy, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, hydroxymethyl, hydroxyethyl, hydroxyisopropyl, trifluoromethyl, trifluoromethoxy, or cyclopropyl.
In certain embodiments, ring C is selected from 7-9 membered nitrogen containing spiroheterocyclic group optionally substituted with 1-2Q 3, wherein ring-forming carbon atoms in said 7-9 membered nitrogen containing spiroheterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the group consisting of a 7-8 membered 1-2 nitrogen containing spiroheterocyclic group optionally substituted with 1-2Q 3 wherein the ring-forming carbon atoms in said spiroheterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 5-azaspiro [2.4] heptyl, 2-azaspiro [3.3] heptyl, 2-azaspiro [3.5] nonanyl, 2, 6-diazaspiro [3.3] heptanyl, 2, 7-diazaspiro [3.5] nonanyl, 2, 5-diazaspiro [3.4] octanyl, 2-oxa-6-azaspiro [3.3] heptanyl, 6-oxa-2-azaspiro [3.4] octanyl, 6-azaspiro [3.4] octanyl, 2-azaspiro [4.4] nonanyl, 2-oxa-7-azaspiro [4.4] nonanyl, 6-azaspiro [3.4] oct-7-enyl, 2-oxa-6-azaspiro [3.4] oct-7-enyl or 2-azaspiro [4.4] non-7-enyl, wherein said group-forming carbon atoms are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3:
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, haloGeneration C1-6Alkoxy, hydroxy C1-6Alkoxy or 3-4 membered cycloalkyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 5-azaspiro [2.4] heptylalkyl, 2-azaspiro [3.3] heptylalkyl, 2-azaspiro [3.5] nonanyl, 2, 6-diazaspiro [3.3] heptanyl, 2, 7-diazaspiro [3.5] nonanyl, 2, 5-diazaspiro [3.4] octanyl, 2-oxa-6-azaspiro [3.3] heptanyl, 6-oxa-2-azaspiro [3.4] octanyl, 6-azaspiro [3.4] octanyl, 2-azaspiro [4.4] nonanyl, wherein the groups may optionally be substituted at a ring carbon atom with oxygen to form a carbonyl group;
each Q3 is independently selected from fluoro, chloro, hydroxy, amino, nitro, cyano, carboxy, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, hydroxymethyl, hydroxyethyl, hydroxyisopropyl, trifluoromethyl, trifluoromethoxy, or cyclopropyl.
In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 2, 6-diazaspiro [3.3] heptanyl, 2, 7-diazaspiro [3.5] nonanyl, 2, 5-diazaspiro [3.4] octanyl, wherein the ring carbon atoms are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from fluoro, chloro, hydroxy, amino, nitro, cyano, carboxy, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, hydroxymethyl, hydroxyethyl, hydroxyisopropyl, trifluoromethyl, trifluoromethoxy, or cyclopropyl. In certain embodiments, ring C is selected from the following optionally substituted with 1-2Q 3: 2-azabicyclo [2.2.1] heptanyl, 7-azabicyclo [2.2.1] heptanyl, 3-azabicyclo [3.2.1] octanyl, 8-azabicyclo [3.2.1] octanyl, 2-azabicyclo [2.2.2] octanyl, 2, 5-diazabicyclo [2.2.1] heptanyl, 3, 6-diazabicyclo [3.1.1] heptanyl, 3, 8-diazabicyclo [3.2.1] octanyl, 2-oxa-5-azabicyclo [2.2.1] heptanyl, 8-oxa-3-azabicyclo [3.2.1] octanyl, 3, 8-diazabicyclo [3.2.1] octan-6-enyl, 3, 9-diazabicyclo [3.3.1] nonanyl, 5-azabicyclo [4.1 ] heptanyl, 2-azaspiro [3.3] heptylalkyl, 2-azaspiro [3.5] nonanyl, 2, 6-diazaspiro [3.3] heptanyl, 2, 7-diazaspiro [3.5] nonanyl, 2, 5-diazaspiro [3.4] octanyl, 2-oxa-6-azaspiro [3.3] heptanyl, 6-oxa-2-azaspiro [3.4] octanyl, 6-azaspiro [3.4] octanyl, 2-azaspiro [4.4] nonanyl, 2-oxa-7-azaspiro [4.4] nonanyl, 6-azaspiro [3.4] oct-7-enyl, 2-oxa-6-azaspiro [3.4] oct-7-enyl or 2-azaspiro [4.4] non-7-enyl, wherein the ring-forming carbon atoms of the group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy or hydroxy C1-6An alkoxy group.
In certain embodiments, R1Selected from C optionally substituted by substituents2-4Alkenyl, the substituents are respectively and independently selected from halogen, hydroxyl, amino, carboxyl, cyano and C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkyl or halo C1-6An alkoxy group.
In certain embodiments, R1Selected from-CH optionally substituted by a substituent2-CH=CH2The substituents are respectively and independently selected from halogen, hydroxyl, amino, carboxyl, cyano and C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkyl or halo C1-6An alkoxy group.
In certain embodiments, R1Is selected from-CH2-CH=CH2。
In certain embodiments, the compounds of the present invention, pharmaceutically acceptable salts thereof, or stereoisomers thereof, wherein X is1、X2、X3Are each independently selected from-C (R)2) -or-N-;
X4、X5each independently selected from-CH-or-N-;
each L is independently selected from-C (R)3)(R4)-、-O-、-N(R5) -or-S-;
R2、R3、R4、R5each independently selected from hydrogen and C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl or halo C1-6An alkoxy group;
R1selected from C optionally substituted by substituents2-6Alkenyl, each of said substituents being independently selected from halogen, hydroxy, amino, carboxy, cyano, C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl or halo C1-6An alkoxy group.
In certain embodiments, R2、R3、R4、R5Each independently selected from hydrogen, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, monofluoromethyl, difluoromethyl, trifluoromethyl or trifluoromethoxy.
In certain embodiments, X1、X2、X3Are each independently selected from-C (R)2) -or-N-;
X4、X5each independently selected from-CH-or-N-;
each L is independently selected from-C (R)3)(R4)-、-O-、-N(R5) -or-S-;
R2、R3、R4、R5each independently selected from hydrogen, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, monofluoromethyl, difluoromethyl, trifluoromethyl or trifluoromethoxy;
R1selected from C optionally substituted by substituents2-6Alkenyl, the substituents are respectively and independently selected from halogen, hydroxyl, amino, carboxyl, cyano and C1-6Alkyl radical, C1-6Alkoxy radicalBase, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl or halo C1-6An alkoxy group.
In certain embodiments, R1Selected from C optionally substituted by substituents2-4Alkenyl, preferably propen-3-yl, optionally substituted with substituents each independently selected from halogen, hydroxy, amino, carboxy, cyano, C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkyl or halo C1-6An alkoxy group.
In certain embodiments, X1、X2Each independently selected from-N-.
In certain embodiments, X3is-CH-.
In certain embodiments, X4、X5Are each independently-N-.
In certain embodiments, the compounds of the present invention, pharmaceutically acceptable salts thereof, or stereoisomers thereof, have the structure shown in formula (II) below:
wherein, ring A, ring B, ring C, Q1, Q2, Q3, L, R3、R4、R5、R1M, n are as defined above.
In certain embodiments, the compounds of the present invention, pharmaceutically acceptable salts thereof, or stereoisomers thereof, have the structure shown in formula (III) below:
wherein, ring A, ring B, ring C, Q1, Q2, Q3, L, R3、R4、R5M, n are as defined above.
In certain embodiments, the compounds of the present invention, pharmaceutically acceptable salts thereof, or stereoisomers thereof, have the structure shown in formula (III) below:
wherein ring a is selected from pyridyl, pyrimidinyl, pyrazinyl, or pyridazinyl substituted with 1Q 1; q1 is selected from fluoro, chloro, bromo, hydroxy, amino, nitro, cyano or the following optionally substituted with 1-3 substituents: methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, methoxy, ethoxy, propoxy and isopropoxy, wherein the substituents are respectively and independently selected from fluorine, chlorine, bromine, hydroxyl, amino, nitro, cyano, carboxyl, - (CH)2)m-cyclopropane, - (CH)2)m-cyclobutyl, - (CH)2)m-cyclopentyl or- (CH)2)m-a cyclohexane group;
ring B is phenyl or pyridyl;
each L is independently selected from-C (R)3)(R4) -or-N (R)5)-;R3、R4、R5Each independently selected from hydrogen, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, monofluoromethyl, difluoromethyl, trifluoromethyl, trifluoroethyl, monofluoromethoxy, difluoromethoxy or trifluoromethoxy;
m is selected from 0, 1 or 2; n is 1.
In certain embodiments, the compounds of the present invention, pharmaceutically acceptable salts thereof, or stereoisomers thereof, wherein,
ring a is selected from pyridyl, pyrimidinyl, pyrazinyl, or pyridazinyl substituted with 1Q 1; q1 is selected from fluoro, chloro, bromo, hydroxy, amino, nitro, cyano or the following optionally substituted with 1-3 substituents: methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, methoxy, ethoxy, propoxy and isopropoxy, wherein the substituents are respectively and independently selected from fluorine, chlorine, bromine and hydroxylRadical, amino, nitro, cyano, carboxyl, - (CH)2)m-cyclopropylalkyl, - (CH)2)m-cyclobutyl, - (CH)2)m-cyclopentyl or- (CH)2)m-a cyclohexane group;
ring B is selected from phenyl, pyridyl, pyrimidinyl, pyrazinyl or pyridazinyl.
In certain embodiments, ring B is phenyl or pyridyl.
In certain embodiments, each L is independently selected from-C (R)3)(R4) -or-N (R)5)-;R3、R4、R5Each independently selected from hydrogen, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, monofluoromethyl, difluoromethyl, trifluoromethyl, trifluoroethyl, monofluoromethoxy, difluoromethoxy or trifluoromethoxy.
In certain embodiments, each L is independently selected from-N (R)5)-;R5Each independently selected from hydrogen, methyl, ethyl, propyl, isopropyl, methoxy, trifluoromethyl or trifluoromethoxy; preferably, L is NH.
In certain embodiments, m is selected from 0, 1 or 2.
In certain embodiments, n is selected from 0, 1 or 2; preferably, n is 1.
The technical solutions of the present invention can be combined with each other to form a new technical solution, and the formed new technical solution is also included in the scope of the present invention.
In certain embodiments, the compound of formula (I), formula (II), or formula (III), a pharmaceutically acceptable salt thereof, or a stereoisomer thereof, is selected from the group consisting of:
the invention also provides a pharmaceutical composition, which contains the compound shown in the general formula (I), the general formula (II) or the general formula (III), the pharmaceutically acceptable salt or the stereoisomer thereof, and one or more pharmaceutical carriers and/or diluents; the pharmaceutical composition can be prepared into any clinically or pharmaceutically acceptable dosage form, such as tablets, capsules, pills, granules, solutions, suspensions, syrups, injections (including injection, sterile powder for injection and concentrated solution for injection), suppositories, inhalants or sprays and the like.
In certain embodiments of the invention, the above-described pharmaceutical formulations may be administered to a patient or subject in need of such treatment by oral, parenteral, rectal, or pulmonary administration, and the like. For oral administration, the pharmaceutical composition can be prepared into oral preparations, for example, conventional oral solid preparations such as tablets, capsules, pills, granules and the like; it can also be made into oral liquid, such as oral solution, oral suspension, syrup, etc. When the composition is formulated into oral preparations, appropriate filler, binder, disintegrating agent, lubricant, etc. can be added. For parenteral administration, the pharmaceutical preparations can also be prepared into injections, including injections, sterile powders for injection, and concentrated solutions for injection. The injection can be prepared by conventional method in the existing pharmaceutical field, and can be prepared without adding additives or adding suitable additives according to the properties of the medicine. For rectal administration, the pharmaceutical composition may be formulated as a suppository or the like. For pulmonary administration, the pharmaceutical composition may be formulated as an inhalant or a spray.
The pharmaceutically acceptable carrier and/or diluent useful in the pharmaceutical composition or pharmaceutical formulation of the present invention may be any conventional carrier and/or diluent in the art of pharmaceutical formulation, and the selection of a particular carrier and/or diluent will depend on the mode of administration or the type and state of the disease used to treat a particular patient. The preparation of suitable pharmaceutical compositions for a particular mode of administration is well within the knowledge of those skilled in the pharmaceutical art.
In another aspect, the present invention also relates to the use of a compound of the aforementioned formula (I), formula (II) or formula (III), a pharmaceutically acceptable salt thereof, or a stereoisomer thereof, for the preparation of a medicament for the prevention and/or treatment of diseases mediated by Wee1 and related diseases, which can be used in combination with one or more other medicaments for the prevention or treatment of diseases mediated by Wee1 and related conditions. The disease and related conditions are selected from cancer or benign tumors, including carcinoma in situ and metastatic carcinoma. Further, the cancer includes, but is not limited to, lung cancer, squamous cell carcinoma, bladder cancer, stomach cancer, ovarian cancer, peritoneal cancer, pancreatic cancer, breast cancer, head and neck cancer, cervical cancer, endometrial cancer, rectal cancer, liver cancer, kidney cancer, esophageal adenocarcinoma, esophageal squamous cell carcinoma, prostate cancer, thyroid cancer, cancer of the female genital tract, lymphoma, neurofibroma, bone cancer, skin cancer, brain cancer, colon cancer, testicular cancer, small cell lung cancer, non-small cell lung cancer, gastrointestinal stromal tumor, mast cell tumor, multiple myeloma, melanoma, leukemia, glioma, or sarcoma, and the like.
Further, the invention also relates to the application of a pharmaceutical preparation containing the compound shown in the general formula (I), the general formula (II) or the general formula (III), the pharmaceutically acceptable salt thereof or the stereoisomer thereof in preparing a medicament which can be combined with one or more medicaments for treating and/or preventing diseases mediated by Wee1 and related symptoms.
In another aspect, the invention is directed to medicaments containing a compound of formula (I), formula (II), or formula (III), pharmaceutically acceptable salts, or stereoisomers thereof, as described above, alone or in combination with one or more second therapeutically active agents that are useful in combination with the Wee1 inhibitor compounds of the present application for the treatment and/or prevention of diseases and related conditions mediated by Wee 1. Thus, in certain embodiments, the pharmaceutical composition further comprises one or more second therapeutically active agents. In certain embodiments, the second therapeutically active agent is selected from the group consisting of anti-cancer agents, including mitotic inhibitors, alkylating agents, anti-metabolites, antisense DNA or RNA, anti-tumor antibiotics, growth factor inhibitors, signaling inhibitors, cell cycle inhibitors, enzyme inhibitors, retinoid receptor modulators, proteasome inhibitors, topoisomerase inhibitors, biological response modifiers, hormonal agents, angiogenesis inhibitors, cell growth inhibitors, targeting antibodies, HMG-CoA reductase inhibitors, and prenyl protein transferase inhibitors.
In certain embodiments, the ingredients to be combined (e.g., the compound of the invention, a pharmaceutically acceptable salt thereof, a stereoisomer thereof, and the second therapeutically active agent) may be administered simultaneously or separately, sequentially and separately. For example, the second therapeutically active agent may be administered before, simultaneously with, or after the administration of the compound of the present invention, a pharmaceutically acceptable salt thereof, or a stereoisomer thereof. Furthermore, the components to be combined may also be administered in combination in the same formulation or in separate and distinct formulations.
In another aspect, the present invention also provides a method for treating diseases mediated by Wee1 and related disorders, comprising administering to a patient in need thereof an effective amount of a compound of formula (I) as described above, a pharmaceutically acceptable salt thereof, or a stereoisomer thereof, a formulation or pharmaceutical composition as described above; the diseases and related conditions mediated by Wee1 are as defined above.
By "effective amount" is meant a dosage of a drug that reduces, delays, inhibits or cures a condition in a subject. The size of the administered dose is determined by the administration mode of the drug, the pharmacokinetics of the medicament, the severity of the disease, the individual physical signs (sex, weight, height, age) of the subject, and the like.
The medicine containing the compound shown in the general formula (I), the general formula (II) or the general formula (III), the pharmaceutically acceptable salt or the stereoisomer thereof can be combined with radiotherapy to improve the treatment effect of the radiotherapy, and can be used as a radiotherapy sensitizer.
In another aspect, the compounds of the invention may also be used as sensitizers for other anticancer agents in the field of cancer therapy. The anticancer agent is as described above. The sensitizer means a drug which enhances the therapeutic effect of radiotherapy and/or chemotherapy by being used in combination with radiotherapy and/or chemotherapy using an anticancer agent, additively or synergistically, in the field of cancer treatment.
[ detailed description of the invention ]
In the present invention, unless otherwise defined, scientific and technical terms used herein have meanings commonly understood by those skilled in the art, however, in order to better understand the present invention, definitions of some terms are provided below. To the extent that the definitions and explanations of terms provided herein do not conform to the meanings commonly understood by those skilled in the art, the definitions and explanations of terms provided herein shall control.
The "halogen" as referred to herein means a fluorine atom, a chlorine atom, a bromine atom or an iodine atom.
"C" according to the invention1-6Alkyl "denotes straight or branched alkyl having 1 to 6 carbon atoms, including for example" C1-4Alkyl group "," C1-3Alkyl group "," C1-2Alkyl group "," C2-6Alkyl group "," C2-5Alkyl group "," C2-4Alkyl group "," C2-3Alkyl group "," C3-6Alkyl group "," C3-5Alkyl group "," C3-4Alkyl "and the like, specific examples include, but are not limited to: methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, 2-methylbutyl, neopentyl, 1-ethylpropyl, n-hexyl, isohexyl, 3-methylpentyl, 2-methylpentyl, 1-methylpentyl, 3-dimethylbutyl, 2-dimethylbutyl, 1-dimethylbutyl, 1, 2-dimethylbutyl, 1, 3-dimethylbutyl, 2-ethylbutyl, 1, 2-dimethylpropyl, and the like. "C" according to the invention1-4Alkyl "means C1-6Specific examples of the alkyl group having 1 to 4 carbon atoms.
"C" according to the invention1-6Alkoxy "means" C1-6alkyl-O- ", said" C1-6Alkyl "is as defined above. "C" according to the invention1-4Alkoxy "means" C1-4alkyl-O- ", said" C1-4Alkyl "is as defined above.
"C" according to the invention1-6Alkyl sulfideThe radical "means" C1-6alkyl-S- ", said" C1-6Alkyl "is as defined above. "C" according to the invention1-4Alkylthio "means" C1-4alkyl-S- ", said" C1-4Alkyl "is as defined above.
The "hydroxyl group C" of the present invention1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkyl "means C1-6One or more hydrogens of the alkyl group are each replaced by one or more hydroxyl, amino or halogen. C1-6Alkyl is as previously defined
The "hydroxyl group C" of the present invention1-6Alkoxy, amino C1-6Alkoxy, halo C1-6Alkoxy "means" C1-6One or more hydrogens of "alkoxy" are replaced with one or more hydroxy, amino, or halogen.
The "hydroxy group C" of the present invention1-6Alkylthio, amino C1-6Alkylthio, halo C1-6Alkylthio "means" C1-6Alkylthio "is one in which one or more hydrogens are replaced with one or more hydroxy, amino, or halogen.
"C" according to the invention1-6Alkylamino radical, di (C)1-6Alkyl) amino "means independently C1-6alkyl-NH-),
"C" according to the invention2-6Alkenyl "means a straight, branched or cyclic alkenyl group having 2 to 6 carbon atoms containing at least one double bond, and includes, for example," C2-4Alkenyl groups "and the like. Examples thereof include, but are not limited to: vinyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 1, 3-butadienyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 1, 3-pentadienyl, 1, 4-pentadienyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 1, 4-hexadienyl, cyclopentenyl, 1, 3-cyclopentadienyl, cyclohexenyl, 1, 4-cyclohexadienyl and the like.
"C" according to the invention2-6Alkynyl means containing at least one triple bond and having the number of carbon atomsStraight-chain or branched alkynyl radicals having 2 to 6 inclusive, e.g. "C2-4Alkynyl "and the like. Examples include, but are not limited to: ethynyl, propynyl, 2-butynyl, 2-pentynyl, 3-pentynyl, 4-methyl-2-pentynyl, 2-hexynyl, 3-hexynyl, 5-methyl-2-hexynyl and the like.
The "3-to 10-membered cycloalkyl" as referred to herein is meant to include "3-to 8-membered monocycloalkyl" and "8-to 10-membered fused ring alkyl".
The "3-to 8-membered monocyclic alkyl" as used herein means a saturated or partially saturated monocyclic cyclic alkyl group having 3 to 8 carbon atoms and having no aromaticity, and includes "3-to 8-membered saturated monocyclic alkyl" and "3-to 8-membered partially saturated monocyclic alkyl"; preferred are "3-to 4-membered monocycloalkyl", "3-to 5-membered monocycloalkyl", "3-to 6-membered monocycloalkyl", "3-to 7-membered monocycloalkyl", "4-to 5-membered monocycloalkyl", "4-to 6-membered monocycloalkyl", "4-to 7-membered monocycloalkyl", "5-to 6-membered monocycloalkyl", "5-to 7-membered monocycloalkyl", "6-to 8-membered monocycloalkyl", "7-to 8-membered monocycloalkyl", "3-to 6-membered saturated monocycloalkyl", "5-to 8-membered saturated monocycloalkyl", "5-to 7-membered saturated monocycloalkyl", "5-to 6-membered saturated monocycloalkyl", and the like. Specific examples of said "3-to 8-membered saturated monocycloalkyl" include, but are not limited to: a cyclopropane group (cyclopropyl), a cyclobutane group (cyclobutyl), a cyclopentyl group (cyclopentyl), a cyclohexane group (cyclohexyl), a cycloheptyl group (cycloheptyl), a cyclooctyl group (cyclooctyl), etc.; specific examples of the "3-to 8-membered partially saturated monocycloalkyl" include, but are not limited to, cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclopentadienyl, cyclohexenyl, cyclohex-1, 3-diene, cyclohex-1, 4-diene, cycloheptenyl, cyclohepta-1, 3-dienyl, cyclohepta-1, 4-dienyl, cyclohepta-1, 3, 5-trienyl, cyclooctenyl, cycloocta-1, 3-dienyl, cycloocta-1, 4-dienyl, cycloocta-1, 5-dienyl, cycloocta-1, 3, 5-trienyl, cyclooctatetraenyl and the like.
The 8-10-membered fused ring group is a saturated or partially saturated non-aromatic cyclic group containing 8-10 ring atoms, which is formed by two or more cyclic structures sharing two adjacent carbon atoms, wherein one ring in the fused ring can be an aromatic ring, but the fused ring does not have aromatic property as a whole; including "8-9-membered fused ring group", "9-10-membered fused ring group", etc., the fusion mode may be: 5-6 membered cycloalkyl and 5-6 membered cycloalkyl, benzo 5-6 membered saturated cycloalkyl and the like. Examples include, but are not limited to: bicyclo [3.1.0] hexanyl, bicyclo [4.1.0] heptanyl, bicyclo [2.2.0] hexanyl, bicyclo [3.2.0] heptanyl, bicyclo [4.2.0] octanyl, octahydropentanyl, octahydro-1H-indenyl, decahydronaphthyl, tetradecahydrophenanthryl, bicyclo [3.1.0] hex-2-enyl, bicyclo [4.1.0] hept-3-enyl, bicyclo [3.2.0] hept-3-enyl, bicyclo [4.2.0] oct-3-enyl, 1,2,3,3 a-tetrahydropentanyl, 2,3,3a,4,7,7 a-hexahydro-1H-indenyl, 1,2,3,4,4a,5,6,8 a-octahydronaphthyl, 1,2,4a,5,6,8 a-hexahydronaphthyl, 1,2,3,4,5, 8 a-hexahydronaphthyl, 10-decahydrophenanthryl, benzocyclopentyl, benzocyclohexyl, benzocyclohexenyl, benzocyclopentenyl, and the like.
The "3-to 10-membered heterocyclic group" described in the present invention includes "3-to 8-membered heteromonocyclic group" and "8-to 10-membered fused heterocyclic group".
The "3-to 8-membered heteromonocyclic group" according to the present invention means a saturated or partially saturated and non-aromatic monocyclic cyclic group containing at least one hetero atom (e.g., 1,2,3,4 or 5) which is a nitrogen atom, an oxygen atom and/or a sulfur atom and has 3 to 8 ring atoms, and optionally, a ring atom (e.g., a carbon atom, a nitrogen atom or a sulfur atom) in the cyclic structure may be oxo. The "3-to 8-membered heteromonocyclic group" described in the present invention includes "3-to 8-membered saturated heteromonocyclic group" and "3-to 8-membered partially saturated heteromonocyclic group". Preferably, the "3-8 membered heteromonocyclic group" described herein contains 1-3 heteroatoms; preferably, the "3-to 8-membered heteromonocyclic group" of the present invention contains 1 to 2 hetero atoms selected from nitrogen atom and/or oxygen atom; preferably, the "3-to 8-membered heteromonocyclic group" described herein contains 1 nitrogen atom. The "3-to 8-membered heteromonocyclic group" is preferably "3-to 7-membered heteromonocyclic group", "3-to 6-membered heteromonocyclic group", "4-to 7-membered heteromonocyclic group", "4-to 6-membered heteromonocyclic group", "6-to 8-membered heteromonocyclic group", "5-to 7-membered heteromonocyclic group", "5-to 6-membered heteromonocyclic group", "3-to 6-membered saturated nitrogen-containing heteromonocyclic group", "5-to 6-membered saturated nitrogen-containing heteromonocyclic group", and the like. For example, containing only 1 or 2 nitrogen atoms, or, alternatively, containing one nitrogen atom and 1 or 2 other heteroatoms (e.g., oxygen and/or sulfur atoms). Specific examples of "3-8 membered heteromonocyclic group" include, but are not limited to: aziridinyl, 2H-aziridinyl, diazacyclopropenyl, 3H-diazacyclopropenyl, azetidinyl, 1, 4-dioxanyl, 1, 3-dioxolanyl, 1, 4-dioxadienyl, tetrahydrofuryl, dihydropyrrolyl, pyrrolidinyl, imidazolidinyl, 4, 5-dihydroimidazolyl, pyrazolidinyl, 4, 5-dihydropyrazolyl, 2, 5-dihydrothienyl, tetrahydrothienyl, 4, 5-dihydrothiazolyl, thiazolidinyl, piperidinyl, tetrahydropyridinyl, piperidonyl, piperazinyl, morpholinyl, 4, 5-dihydrooxazolyl, 4, 5-dihydroisoxazolyl, 2, 3-dihydroisoxazolyl, 3H-diazacyclopropenyl, imidazolidinyl, 4, 5-dihydroimidazolyl, pyrazolidinyl, 4, 5-dihydrothiazolyl, thiazolidinyl, piperidyl, piperidonyl, tetrahydropyridinyl, piperidyl, piperazinyl, morpholinyl, 4, 5-dihydroisoxazolyl, 2, 3-dihydroisoxazolyl, and the like, Oxazolidinyl, 2H-1, 2-oxazinyl, 4H-1, 2-oxazinyl, 6H-1, 2-oxazinyl, 4H-1, 3-oxazinyl, 6H-1, 3-oxazinyl, 4H-1, 4-oxazinyl, 4H-1, 3-thiazinyl, 6H-1, 3-thiazinyl, 2H-pyranyl, 2H-pyran-2-onyl, 3, 4-dihydro-2H-pyranyl and the like.
The "8-to 10-membered fused heterocyclic group" as used herein refers to a saturated or partially saturated, nonaromatic cyclic group containing 8 to 10 ring atoms, wherein at least one ring atom of the fused ring may be an aromatic ring, but the fused ring as a whole does not have aromaticity, which is formed by two or more cyclic structures sharing two adjacent atoms with each other, and at least one ring atom of the fused ring is a heteroatom, which is a nitrogen atom, an oxygen atom and/or a sulfur atom, and optionally, a ring atom (e.g., a carbon atom, a nitrogen atom or a sulfur atom) of the cyclic structure may be oxo, and includes, but is not limited to, "8-to 9-membered fused heterocyclic group", "9-to 10-membered fused heterocyclic group" and the like, and may be fused in such a manner that the fused ring is a 5-to 6-membered heterocyclic group, a 5-to 6-membered cycloalkyl group, Benzo 5-6 membered heterocyclyl, benzo 5-6 membered saturated heterocyclyl, 5-6 membered heteroarylo 5-6 membered saturated heterocyclyl; 5-6 membered heteroaryl is as previously defined; specific examples of the "8-to 10-membered fused heterocyclic group" include, but are not limited to: pyrrolidinyl cyclopropyl, cyclopent aziridinyl, pyrrolidinyl cyclobutyl, pyrrolidinyl, pyrrolidinyl piperidinyl, pyrrolidinyl piperazinyl, pyrrolidinyl morpholinyl, piperidinyl morpholinyl, benzopyrrolidinyl, benzocyclopentyl, benzocyclohexyl, benzotetrahydrofuranyl, benzopyrrolidinyl, benzimidazolyl, benzoxazolinyl, benzothiazolinyl, benzisoxazolidinyl, benzisothiazolidinyl, benzopyriperidinyl, benzomorpholinyl, benzopyrazinyl, benzopyranyl, pyridocyclopentyl, pyridocyclohexyl, pyridotetrahydrofuranyl, pyridopyrrolidinyl, pyridoimidazolidinyl, pyridooxazolidinyl, pyridothiazolidinyl, pyridoisoxazolidinyl, pyridoisothiazolidinyl, pyridopiperidinyl, pyridomorpholinyl, and the like, Pyridopiperazinyl, pyridotetrahydropyranyl, pyrimidocyclopentyl, pyrimidocyclohexyl, pyrimidotetrahydrofuranyl, pyrimidopyrrolidinyl, pyrimidoimidazoimidazolidinyl, pyrimidooxazolidoalkyl, pyrimidoiizolidinyl, pyrimidoiisoxazolidinyl, pyrimidoiisothiazolidinyl, pyrimidoipiperidinyl, pyrimidoimorpholinyl, pyrimidoipiperazinyl, pyrimidoitetrahydropyranyl; tetrahydroimidazo [4,5-c ] pyridyl, 3, 4-dihydroquinazolinyl, 1, 2-dihydroquinoxalinyl, benzo [ d ] [1,3] dioxolyl, 2H-chromenyl, 2H-chromen-2-one, 4H-chromenyl, 4H-chromen-4-one, 4H-1, 3-benzoxazinyl, 4, 6-dihydro-1H-furo [3,4-d ] imidazolyl, 3a,4,6,6 a-tetrahydro-1H-furo [3,4-d ] imidazolyl, 4, 6-dihydro-1H-thieno [3,4-d ] imidazolyl, 4, 6-dihydro-1H-pyrrolo [3,4-d ] imidazolyl, octahydro-benzo [ d ] imidazolyl, decahydroquinolinyl, hexahydrothienoimidazolyl, hexahydrofuroimidazolyl, 4,5,6, 7-tetrahydro-1H-benzo [ d ] imidazolyl, octahydro-cyclopenta [ c ] pyrrolyl, 4H-1, 3-benzoxazinyl and the like.
The term "benzocyclopentyl", the structure of which refers to(also referred to as 2, 3-dihydro-1H-indenyl); the term "benzo-pyrrolidine" structurally includesEtc.; the term "pyridotetrahydrofuranyl" includes Specific examples of the other "other condensed heterocyclic group as defined above" have a cyclic structure similar thereto.
The "6-to 10-membered aryl" as referred to in the present invention includes "6-to 8-membered monocyclic aryl" and "8-to 10-membered fused ring aryl".
The "6-to 8-membered monocyclic aryl" as referred to herein means a monocyclic aryl group containing 6 to 8 ring carbon atoms, examples of which include, but are not limited to: phenyl, cyclooctatetraenyl and the like; phenyl is preferred.
The "8-to 10-membered fused ring aryl" as referred to herein means an unsaturated aromatic cyclic group having 8 to 10 ring carbon atoms, formed by two or more cyclic structures sharing two adjacent atoms with each other, and is preferably a "9-to 10-membered fused ring aryl", and specific examples thereof are naphthyl and the like.
The "5-to 10-membered heteroaryl" as referred to herein includes "5-to 8-membered monoheteroaryl" and "8-to 10-membered fused heteroaryl".
The "5-to 8-membered monoheteroaryl group" according to the present invention means a monocyclic cyclic group having aromaticity, which contains 5 to 8 ring atoms, at least one of which is a heteroatom such as nitrogen atom, oxygen atom or sulfur atom. Optionally, a ring atom (e.g., a carbon atom, a nitrogen atom, or a sulfur atom) in the cyclic structure may be oxo. The "5-to 8-membered monoheteroaryl group" includes, for example, "5-to 7-membered monoheteroaryl group", "5-to 6-membered nitrogen-containing monoheteroaryl group", "6-membered nitrogen-containing monoheteroaryl group", and the like, in which the hetero atom contains at least one nitrogen atom, for example, contains only 1 or 2 nitrogen atoms, or contains one nitrogen atom and the other 1 or 2 hetero atoms (for example, oxygen atom and/or sulfur atom), or contains 2 nitrogen atoms and the other 1 or 2 hetero atoms (for example, oxygen atom and/or sulfur atom). Specific examples of "5-to 8-membered monocyclic heteroaryl" include, but are not limited to, furyl, thienyl, pyrrolyl, thiazolyl, isothiazolyl, thiadiazolyl, oxazolyl, isoxazolyl, oxadiazolyl, imidazolyl, pyrazolyl, 1,2, 3-triazolyl, 1,2, 4-triazolyl, 1,2, 3-oxadiazolyl, 1,2, 4-oxadiazolyl, 1,2, 5-oxadiazolyl, 1,3, 4-oxadiazolyl, pyridyl, 2-pyridonyl, 4-pyridonyl, pyrimidinyl, pyridazinyl, pyrazinyl, 1,2, 3-triazinyl, 1,3, 5-triazinyl, 1,2,4, 5-tetrazinyl, azepinyl, 1, 3-diazacycloheptenyl, azepinyl, and the like. The "5-6 membered monoheteroaryl" refers to a specific example containing 5 to 6 ring atoms in the 5-8 membered heteroaryl.
The "8-to 10-membered fused heteroaryl group" as used herein refers to an unsaturated aromatic cyclic structure having 8 to 10 ring atoms (at least one of which is a heteroatom such as nitrogen atom, oxygen atom or sulfur atom) formed by two or more cyclic structures sharing two adjacent atoms with each other. Optionally, a ring atom (e.g., a carbon atom, a nitrogen atom, or a sulfur atom) in the cyclic structure may be oxo. Including "9-10 membered fused heteroaryl", "8-9 membered fused heteroaryl", etc., which can be fused in a benzo-5-6 membered heteroaryl, 5-6 membered heteroaryl and 5-6 membered heteroaryl, etc.; specific examples include, but are not limited to: pyrrolopyrrole, pyrrolofuran, pyrazolopyrrole, pyrazolothiophene, furothiophene, pyrazoloxazole, benzofuranyl, benzisofuranyl, benzothiophenyl, indolyl, isoindolyl, benzoxazolyl, benzimidazolyl, indazolyl, benzotriazolyl, quinolinyl, 2-quinolinonyl, 4-quinolinonyl, 1-isoquinolinyl, acridinyl, phenanthridinyl, pyridazinyl, phthalazinyl, quinazolinyl, quinoxalinyl, purinyl, naphthyridinyl, and the like.
The 5-10 membered spiro ring group is two or moreCyclic structures containing from 5 to 10 ring carbon atoms formed by the upper cyclic structures sharing a carbon atom with each other. Optionally, the carbon atoms in the cyclic structure may be oxidized. The "5-to 10-membered spiro ring group" includes, for example, "4-to 10-membered spiro ring group", "6-to 10-membered spiro ring group", "7-to 10-membered spiro ring group", "6-to 9-membered spiro ring group", "7-to 9-membered spiro ring group", "9-to 10-membered spiro ring group" and the like. Specific examples include, but are not limited to:
and the like. The "7-9 membered spirocyclic group" means a specific example containing 7 to 9 ring atoms in the 5-10 membered spirocyclic group.
The "5-15 membered bridged cyclic group" as used herein means a cyclic structure containing 5 to 15 ring carbon atoms, which is formed by two or more cyclic structures sharing two non-adjacent carbon atoms with each other. Optionally, the carbon atom in the cyclic structure may be oxo. "5-15-membered bridge ring group" includes, for example, "5-11-membered bridge ring group", "6-11-membered bridge ring group", "5-10-membered bridge ring group", "7-10-membered bridge ring group", "6-9-membered bridge ring group", "7-9-membered bridge ring group", "9-10-membered bridge ring group" and the like. Specific examples include, but are not limited to:
and the like. The "7-to 9-membered bridged ring group" means a specific example in which the 5-to 15-membered bridged ring group contains 7 to 9 ring atoms.
The 5-to 10-membered spiro complex of the inventionThe "cyclic group" means a cyclic structure containing 5 to 10 ring atoms (at least one of which is a hetero atom such as a nitrogen atom, an oxygen atom or a sulfur atom) formed by two or more cyclic structures sharing one ring atom with each other, and includes "5 to 10-membered saturated spiroheterocyclic group" and "5 to 10-membered partially saturated spiroheterocyclic group". Optionally, a ring atom (e.g., a carbon atom, a nitrogen atom, or a sulfur atom) in the cyclic structure may be oxidized. The "5-to 10-membered spiroheterocyclic group" includes, for example, "6-to 11-membered spiroheterocyclic group", "6-to 9-membered spiroheterocyclic group", "7-to 9-membered spiroheterocyclic group", "9-to 10-membered spiroheterocyclic group", "5-to 10-membered nitrogen-containing spiroheterocyclic group", "7-to 9-membered nitrogen-containing spiroheterocyclic group", "8-to 9-membered nitrogen-containing spiroheterocyclic group", "7-to 9-membered nitrogen-containing saturated spiroheterocyclic group", "8-to 9-membered nitrogen-containing saturated spiroheterocyclic group" and the like. Specific examples include, but are not limited to: and the like.
The "5-to 15-membered bridged heterocyclic group" as used herein means a cyclic structure containing 5 to 15 ring atoms (at least one of which is a hetero atom such as a nitrogen atom, an oxygen atom or a sulfur atom) formed by two or more cyclic structures sharing two non-adjacent ring atoms with each other, and includes "5-to 15-membered saturated bridged heterocyclic group" and "5-to 15-membered partially saturated bridged heterocyclic group". Optionally, a ring atom (e.g., a carbon atom, a nitrogen atom, or a sulfur atom) in the cyclic structure may be oxidized. The "5-to 15-membered bridged heterocyclic group" includes, for example, "5-to 10-membered bridged heterocyclic group", "6-to 11-membered bridged heterocyclic group", "6-to 9-membered bridged heterocyclic group", "6-to 10-membered bridged heterocyclic group", "7-to 9-membered nitrogen-containing bridged heterocyclic group", "7-to 8-membered nitrogen-containing bridged heterocyclic group", "5-to 9-membered nitrogen-containing bridged heterocyclic group", "5-to 15-membered nitrogen-containing bridged heterocyclic group", "5-to 10-membered bridged heterocyclic group", "7-to 9-membered nitrogen-containing bridged heterocyclic groupNitrogen-saturated bridged heterocyclic group ", and the like. Specific examples include, but are not limited to: and so on.
The expression "carbon atom, nitrogen atom or sulfur atom is oxo" as used herein means that C-O, N-O, S-O or SO is formed2The structure of (1).
The term "optionally substituted" as used herein means both the case where one or more hydrogen atoms on a substituent may be "substituted" or "unsubstituted" by one or more substituents.
"pharmaceutically acceptable salt" as used herein refers to an acidic functional group (e.g., -COOH, -OH, -SO) present in a compound3H, etc.) with a suitable inorganic or organic cation (base), including salts with alkali or alkaline earth metals, ammonium salts, and salts with nitrogen-containing organic bases; and salts of basic functional groups present in the compounds (e.g., -NH2, etc.) with suitable inorganic or organic anions (acids), including salts with inorganic or organic acids (e.g., carboxylic acids, etc.).
"isomers" as used herein refers to compounds of the present invention when they contain one or more asymmetric centers and thus may be present as racemates and racemic mixtures, single enantiomers, diastereomeric mixtures and individual diastereomers. The compounds of the present invention may have asymmetric centers that each independently produce two optical isomers. The scope of the present invention includes all possible optical isomers and mixtures thereof. The compounds of the present invention, if they contain an olefinic double bond, include cis-isomers and trans-isomers, unless otherwise specified. The compounds of the invention may exist in tautomeric (one of the functional group isomers) forms having different points of attachment of hydrogen through one or more double bond shifts, e.g., a ketone and its enol form are keto-enol tautomers. The compounds of the present invention contain a spiro ring structure, and substituents on the ring may be present on both sides of the ring to form the opposite cis (cis) and trans (trans) isomers, depending on the steric structure of the ring. Each tautomer and mixtures thereof are included within the scope of the present invention. All enantiomers, diastereomers, racemates, meso, cis-trans isomers, tautomers, geometric isomers, epimers, mixtures thereof and the like of the compounds are included within the scope of the present invention.
The compounds of the invention may be prepared by enantiospecific synthesis or by resolution from a mixture of enantiomers in such a way as to give the individual enantiomers. Conventional resolution techniques include the formation of salts of the free base of each of the enantiomers of an enantiomeric pair using optically active acids (followed by fractional crystallization and regeneration of the free base), the formation of salts of the acid form of each of the enantiomers of an enantiomeric pair using optically active amines (followed by fractional crystallization and regeneration of the free acid), the formation of esters or amides of each of the enantiomers of an enantiomeric pair using optically pure acids, amines or alcohols (followed by chromatographic separation and removal of the chiral auxiliary), or the resolution of mixtures of the enantiomers of the starting materials or final products using various well-known chromatographic methods.
When the stereochemistry of the disclosed compounds is named or depicted by structure, the named or depicted stereoisomer is at least 60%, 70%, 80%, 90%, 99% or 99.9% pure by weight relative to the other stereoisomers. When a single isomer is named or depicted by structure, the depicted or named enantiomer is at least 60%, 70%, 80%, 90%, 99%, or 99.9% pure by weight. The optical purity wt% is the ratio of the weight of an enantiomer to the weight of the enantiomer plus the weight of its optical isomer.
The "dosage form" of the present invention refers to a form prepared from the drug suitable for clinical use, including, but not limited to, powders, tablets, granules, capsules, solutions, emulsions, suspensions, injections (including injections, sterile powders for injections and concentrated solutions for injections), sprays, aerosols, powders, lotions, liniments, ointments, plasters, pastes, patches, gargles or suppositories, more preferably powders, tablets, granules, capsules, solutions, injections, ointments, gargles or suppositories.
Advantageous effects of the invention
1. The compound, the pharmaceutically acceptable salt thereof or the stereoisomer thereof has excellent Wee1 activity inhibition effect, has good pharmacokinetic property in organisms, has lasting effect and high exposure and bioavailability, and can treat and/or prevent diseases mediated by Wee 1.
2. The compound, the pharmaceutically acceptable salt thereof or the stereoisomer thereof has a better therapeutic effect on Wee 1-mediated cancer.
3. The compound, the pharmaceutically acceptable salt thereof or the stereoisomer thereof can synergistically enhance the effects of radiotherapy and chemotherapy, effectively inhibit the growth of tumors, and can reduce the damage to normal cells and reduce side effects.
4. The compound of the invention has simple preparation process, high medicine purity, stable quality and easy large-scale industrial production.
Detailed description of the preferred embodiments
The technical solutions of the present invention will be described below in conjunction with the specific embodiments, and the above-mentioned contents of the present invention will be further described in detail, but it should not be understood that the scope of the above-mentioned subject matter of the present invention is limited to the following examples. All the technologies realized based on the above contents of the present invention belong to the scope of the present invention.
Example 1: preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- ((4- (8-methyl-3, 8-diazabicyclo [3.2.1] oct-3-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (compound 1)
Preparation of ethyl 4- (2-allyl-2- (tert-butoxycarbonyl) hydrazino) -2- (methylthio) pyrimidine-5-carboxylate
Tert-butyl 1-allylhydrazine-1-carboxylate (0.37g,21.5mmol) and ethyl 4-chloro-2- (methylthio) pyrimidine-5-carboxylate (0.5g,21.5mmol) were dissolved in tetrahydrofuran (100.0mL), N-diisopropylethylamine (0.69g,53.8mmol) was added, reacted at 70 ℃ for 16h, the solvent was dried, and column chromatography (ethyl acetate/petroleum ether ═ 25%) gave the product (0.62g, 78.5%).
Preparation of ethyl 4- (2-allylhydrazino) -3- (methylthio) -1,2, 4-triazine-6-carboxylate
Ethyl 4- (2-allyl-2- (tert-butoxycarbonyl) hydrazino) -2- (methylthio) pyrimidine-5-carboxylate (0.62g,16.8mmol) was dissolved in trifluoroacetic acid (50.0mL), reacted at 70 ℃ for 1 hour, and spin-dried to give the crude product which was used directly in the next step.
Preparation of 2-allyl-6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
4- (2-allylhydrazino) -3- (methylthio) -1,2, 4-triazine-6-carboxylic acid ethyl ester (crude in the above step) was dissolved in 70.0mL of ethanol, an aqueous solution of sodium hydroxide (25.6mL,179mmol) was added, the reaction was carried out at 20 ℃ for 30 minutes, 3N diluted hydrochloric acid was added to adjust the pH to 4, dichloromethane and 5% methanol (50.0mL) were extracted, and the organic phase was dried by spinning to give 4.0g of crude product.
Preparation of 2- (6-bromopyridin-2-yl) propan-2-ol
Methyl 6-bromopicolinate (5.0g, 23.3mmol) was dissolved in 150mL tetrahydrofuran, and methyl magnesium bromide solution (16.0mL,100.0mmol) was added at 0 deg.C, reacted at 25 deg.C for 2h, quenched with saturated ammonium chloride solution, extracted with ethyl acetate, and concentrated by column chromatography to give 4.5g, 90% yield.
Preparation of 2-allyl-1- (6- (2- (hydroxyprop-2-yl) pyridin-2-yl) -6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
2-allyl-6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (crude from the above step) was dissolved in 1, 4-dioxane (150mL), cuprous iodide (3.4g,18.0mmol), potassium carbonate (3.5g,25.2mmol), N' -dimethylethylenediamine (3.2g, 36.0mmol), 2- (6-bromopyridin-2-yl) propan-2-ol (5.1g,23.4mmol) were added, reacted at 95 ℃ for 16H, and column chromatography was concentrated (ethyl acetate/petroleum ether ═ 50%) to give 4.0g of product, three-step total yield: 66.61 percent.
Preparation of tert-butyl (1R,5S) -3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane-8-carboxylate
Tert-butyl 3, 8-diazabicyclo [3.2.1] octane-8-carboxylate (0.5g,2.4mmol) was dissolved in N, N-dimethylacetamide (10.0mL), 1-fluoro-4-nitrobenzene (0.33g,2.4mmol), N-diisopropylethylamine (0.9g,7.2mmol) was added, reacted at 90 ℃ for 16h, and column chromatography was concentrated (ethyl acetate/petroleum ether ═ 25%) to give the product (0.33g, 41.3%).
Preparation of (1R,5S) -3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane
Tert-butyl (1R,5S) -3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane-8-carboxylate (0.15g,0.45mmol), dissolved in 10.0mL of dichloromethane, added with 3.0mL of trifluoroacetic acid, reacted at room temperature for 1h, and spin-dried to give 190.0mg of crude product.
Preparation of (1R,5S) -8-methyl-3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane
(1R,5S) -3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane (crude product from the previous step) was dissolved in 12.0mL of N, N-dimethylformamide, sodium triacetoxyborohydride (0.3g,1.6mmol) and a formaldehyde solution (0.2g, 5.4mmol) were added, the reaction was carried out at 20 ℃ for 3 hours, 10.0mL of water was added to quench the reaction, and dichloromethane was extracted and concentrated to obtain 0.27 g.
Preparation of 9.4- ((1R,5S) -8-methyl-3, 8-diazabicyclo [3.2.1] octan-3-yl) aniline
(1R,5S) -8-methyl-3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane (0.25g, 1.0mmol), dissolved in 6.0mL of methanol, 0.2g of palladium on carbon (10%), reacted for 1h, and concentrated to give 0.23g of crude product.
Preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- ((4- (8-methyl-3, 8-diazabicyclo [3.2.1] oct-3-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
2-allyl-1- (6- (2- (hydroxypropan-2-yl) pyridin-2-yl) -6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (0.29g, 0.81mmol), dissolved in 3.0mL of toluene, 85% m-chloroperoxybenzoic acid (m-CPBA,0.21g,1.2mmol) was added and reacted at room temperature for 30 minutes, N-diisopropylethylamine (0.54g,4.2mmol), 4- ((1R,5S) -8-methyl-3, 8-diazabicyclo [3.2.1] octan-3-yl) aniline (0.23g,1.1mmol) was added and reacted at room temperature for 16 hours, saturated sodium bicarbonate was reacted, extracted with ethyl acetate, dried, concentration and reverse phase column chromatography (water/methanol 60%) gave 23.0mg, 5.5% yield.
Molecular formula C29H34N8O2Molecular weight 526.0LC-MS (M/e):527.0(M + H)+)
1H-NMR(400MHz,CDCl3)δ:8.81(s,1H),7.84(t,J=7.8Hz,1H),7.75(d,J=8.0Hz,1H),7.51-7.39(m,2H),7.32(d,J=7.6Hz,1H),6.78(d,J=8.2Hz,2H),5.74-5.65(m,1H),5.03(d,J=10.1Hz,1H),4.93(d,J=17.2Hz,1H),4.73(d,J=6.0Hz,2H),3.96(s,1H),3.34(d,J=10.5Hz,2H),3.28(s,2H),3.02(d,J=10.5Hz,2H),2.36(s,3H),2.04-2.03(m,2H),1.79-1.77(m,2H),1.58(s,6H).
Example 2: preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- ((4- (5-methyl-2, 5-diazabicyclo [2.2.2] oct-2-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (compound 2)
Preparation of 2-methyl-5- (4-nitrophenyl) -2, 5-diazabicyclo [2.2.2] octane
2-methyl-2, 5-diazabicyclo [2.2.2] octane (100mg,0.5mmol) was dissolved in 10ml of acetonitrile, and 1-fluoro-4-nitrobenzene (141mg,1.0mmol) and N, N-diisopropylethylamine (323mg,2.5mmol) were added and reacted at 60 ℃ for 16 hours. Concentration and silica gel column purification (methanol: dichloromethane ═ 1:7) gave 74mg of the objective compound in a yield of 60.0%.
Preparation of 4- (5-methyl-2, 5-diazabicyclo [2.2.2] oct-2-yl) aniline
2-methyl-5- (4-nitrophenyl) -2, 5-diazabicyclo [2.2.2] octane (70mg,0.28mmol) was dissolved in methanol (5mL), and palladium on carbon (40mg) was added to the solution to conduct a reaction at 16 ℃ for 1 hour under a hydrogen atmosphere. Concentration gave the crude product (72mg) which was used directly in the next reaction.
Preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- ((4- (5-methyl-2, 5-diazabicyclo [2.2.2] oct-2-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
2-allyl-1- (6- (2- (hydroxypropan-2-yl) pyridin-2-yl) -6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (77mg,0.22mmol) was dissolved in toluene (10mL), 62.5% m-CPBA (77mg,0.28mmol) was added and reacted at 16 ℃ for 1 hour, N-diisopropylethylamine (148mg,1.14mmol) and 4- (5-methyl-2, 5-diazabicyclo [2.2.2] oct-2-yl) aniline (70mg,0.32mmol) were added and reacted at 16 ℃ for 5 hours, followed by concentration and purification with a C18 column (water: methanol ═ 3:1) to give 29mg of the title compound in 25.0% yield.
Molecular formula C29H34N8O2Molecular weight 526.6LC-MS (M/e):527.2(M + H)+)
1H-NMR(400MHz,DMSO-d6)δ:10.12(s,1H),8.79(s,1H),8.03(s,1H),7.76(d,J=7.6Hz,1H),7.60(d,J=8.0Hz,1H),7.59-7.52(m,2H),6.64(d,J=8.4Hz,2H),5.76-5.63(m,1H),5.32(s,1H),5.00(d,J=10.4Hz,1H),4.85-4.81(m,1H),4.69-4.67(m,2H),3.86(s,1H),3.63(d,J=9.6Hz,1H),3.23(d,J=9.2Hz,1H),2.90(d,J=8.8Hz,1H),2.33(s,3H),2.03-1.96(m,2H),1.83-1.56(m,3H),1.44(s,6H).
Example 3: preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (((4- (6-methyl-2, 6-diazaspiro [3.3] hept-2-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (compound 3)
Preparation of 2-methyl-6- (4-nitrophenyl) -2, 6-diazaspiro [3.3] heptane
2-methyl-2, 6-diazaspiro [3.3] heptane dihydrochloride (100mg,0.54mmol), p-fluoronitrobenzene (83mg,0.59mmol) and N, N-diisopropylethylamine (349mg,2.7mmol) were dissolved in acetonitrile (20mL) and reacted at 60 ℃ for 6h, the reaction was completed, a saturated sodium bicarbonate (3mL) solution was added, concentrated, and the crude product was purified by silica gel column chromatography (dichloromethane: methanol ═ 10:1) to give the product (100mg, yield 79.1%).
Preparation of 4- (6-methyl-2, 6-diazaspiro [3.3] hept-2-yl) aniline
2-methyl-6- (4-nitrophenyl) -2, 6-diazaspiro [3.3] heptane (30mg,0.13mmol) was dissolved in methanol (10mL), and palladium on carbon (15mg) was added to react at 25 ℃ for 0.5 hour under a hydrogen atmosphere. Filtration and concentration gave the crude product (28mg) which was used directly in the next reaction.
Preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (((4- (6-methyl-2, 6-diazaspiro [3.3] hept-2-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
2-allyl-1- (6- (2- (hydroxypropan-2-yl) pyridin-2-yl) -6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (32mg, 0.09mmol) was dissolved in toluene (7mL), m-chloroperoxybenzoic acid (20mg, 0.12mmol) was added and the reaction was completed at 25 ℃ for 0.5 hour, a solution of N, N-diisopropylethylamine (69mg, 0.53mmol) and 4- (6-methyl-2, 6-diazaspiro [3.3] hept-2-yl) aniline (28mg) in toluene (7mL) was added and the mixture was reacted at 25 ℃ for 18 hours, followed by concentration, purification with a C18 column (water: methanol ═ 1:4) to obtain the crude target compound (20 mg), purification by prep-TLC (dichloromethane: methanol: 12:1) gave 18mg of crude target compound, which was purified by preparative liquid phase purification under high pressure (water: methanol: 1:5) to give 2.3mg of product in 5.0% yield.
Molecular formula C28H32N8O2Molecular weight 512.3LC-MS (M/e):513.2(M + H)+)
1H-NMR(400MHz,CDCl3)δ:8.83(s,1H),7.86(t,J=8.0Hz,1H),7.76(d,J=8.0Hz,1H),7.54-7.26(m,4H),6.46(d,J=8.0Hz,2H),5.76-5.67(m,1H),5.07-4.94(m,2H),4.75(d,J=6.0Hz,2H),3.96(s,4H),3.42(s,4H),2.35(s,3H),1.60(s,6H).
Example 4: preparation of 6- (4- ((2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -3-oxo-2, 3-dihydro-1H-pyrazolo [3,4-d ] pyrimidin-6-yl) amino) phenyl) -3-methyl-3, 6-diazabicyclo [3.1.1] hept-2-one (compound 4)
Preparation of tert-butyl 3-methyl-2-oxo-3, 6-diazabicyclo [3.1.1] heptane-6-carboxylate
Tert-butyl 2-oxo-3, 6-diazabicyclo [3.1.1] heptane-6-carboxylate (106mg,0.5mmol) was dissolved in tetrahydrofuran (10mL), 60% sodium hydride (40mg,1mmol) was added, reaction was carried out at 25 ℃ for 2h, iodomethane (142mg,1mmol) was added, reaction was carried out at 25 ℃ for 2h, quenching was carried out with saturated saline (20mL), extraction was carried out with ethyl acetate (30mL), the organic phase was dried over anhydrous sodium sulfate and spin-dried to give the crude product, which was used directly in the next step.
Preparation of 2.3-methyl-3, 6-diazabicyclo [3.1.1] hept-2-one hydrochloride
The crude product of 3-methyl-2-oxo-3, 6-diazabicyclo [3.1.1] heptane-6-carboxylic acid tert-butyl ester obtained in the previous step was dissolved in 4M dioxane hydrochloride (2mL), reacted at 20 ℃ for 16 hours, and spin-dried to obtain a crude product, which was used directly in the next step.
Preparation of 3-methyl-6- (4-nitrophenyl) -3, 6-diazabicyclo [3.1.1] heptan-2-one
The crude 3-methyl-3, 6-diazabicyclo [3.1.1] hept-2-one hydrochloride salt from the above step, 1-fluoro-4-nitrobenzene (141mg,1mmol) and diisopropylethylamine (194mg,1.5mmol) were dissolved in DMA (7mL), reacted at 90 ℃ for 16 hours, quenched with saturated brine (15mL), extracted with ethyl acetate (30mL), and organic phase spin-dried medium pressure reverse phase preparation (0-40% methanol/water) gave the product (40mg, 32.4%).
Preparation of 6- (4-aminophenyl) -3-methyl-3, 6-diazabicyclo [3.1.1] heptan-2-one
3-methyl-6- (4-nitrophenyl) -3, 6-diazabicyclo [3.1.1] heptan-2-one (50mg,0.2mmol) was reacted with 10% wet palladium on carbon (5mg) in methanol (7mL) under hydrogen (15Psi) at 15 ℃ for 1 hour, and the filtrate was filtered with suction and concentrated to give the product (40mg, 91.0%).
Preparation of 2-allyl-1- (6- (2- (hydroxyprop-2-yl) pyridin-2-yl) -6- (methylsulfinyl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (methylthio) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (100mg,0.28mmol) was reacted with 65% m-chloroperoxybenzoic acid (89mg,0.34mmol) in toluene (2mL) at 15 ℃ for 3 hours, concentrated, and purified by column chromatography (30% tetrahydrofuran/ethyl acetate) to give the product (80mg, 76.6%).
Preparation of 6- (4- ((2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -3-oxo-2, 3-dihydro-1H-pyrazolo [3,4-d ] pyrimidin-6-yl) amino) phenyl) -3-methyl-3, 6-diazabicyclo [3.1.1] hept-2-one
2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (methylsulfinyl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (103mg,0.27mmol), 6- (4-aminophenyl) -3-methyl-3, 6-diazabicyclo [3.1.1] heptan-2-one (40mg,0.18mmol), and diisopropylethylamine (48mg,0.36mmol) were dissolved in THF (12mL) and reacted at 15 ℃ for 16 hours, spun dry, and purified by medium pressure reverse phase preparation (0-40% acetonitrile/water) to give the product (10mg, 10.3%).
The molecular formula is as follows: C28H30N8O3 molecular weight: 526.6LC-MS (M/e):527.2(M + H +)
1HNMR(400MHz,CDCl3):δ:10.11(s,1H),8.82(s,1H),8.10-7.95(m,1H),7.80-7.55(m,2H),7.55-7.45(m,2H),6.60-6.50(m,2H),5.70-5.60(m,1H),5.33(s,1H),5.05-4.95(m,1H),4.85-4.75(m,1H),4.75-4.65(m,2H),4.60-4.50(m,1H),4.10-4.05(m,1H),3.65-3.55(m,1H),3.30-3.20(m,1H),2.80-2.70(m,1H),2.57(m,3H),1.95-1.85(m,1H),1.45(s,6H).
Example 5: preparation of 3- (4- ((2-allyl-1- (6- (2-hydroxypropyl-2-yl) pyridin-2-yl) -3-oxo-2, 3-dihydro-1H-pyrazolo [3,4-d ] pyrimidin-6-yl) amino) phenyl) -6-methyl-3, 6-diazabicyclo [3.1.1] heptan-2-one (Compound 5)
Preparation of tert-butyl 3- (4-nitrophenyl) -2-oxo-3, 6-diazabicyclo [3.1.1] heptane-6-carboxylate
2-oxo 3, 6-diazabicyclo [3.1.1] heptane-6-carboxylic acid tert-butyl ester (149mg,0.70mmol), p-nitroiodobenzene (262mg,1.05mmol), cuprous iodide (27mg,0.14mmol), potassium phosphate (149mg,3.5mmol) and 1, 10-phenanthroline (25mg,0.14mmol) are dissolved in DMF (15mL), reacted at 110 ℃ for 5 hours, quenched with saturated saline (20mL), extracted with ethyl acetate (30mL), dried with anhydrous sodium sulfate of organic phase, spin-dried, and purified by column chromatography (ethyl acetate/petroleum ether ═ 30%) to obtain the product (140mg, 59.8%).
Preparation of 3- (4-Nitrophenyl) -3, 6-diazabicyclo [3.1.1] heptan-2-one trifluoroacetate salt
Tert-butyl 3- (4-nitrophenyl) -2-oxo-3, 6-diazabicyclo [3.1.1] heptane-6-carboxylate (140mg,0.42mmol) was dissolved in dichloromethane (10mL) and trifluoroacetic acid (5mL), reacted at 20 ℃ for 1 hour, and spin-dried to give the crude product which was used directly in the next step.
Preparation of 3.6-methyl-3- (4-nitrophenyl) -3, 6-diazabicyclo [3.1.1] heptan-2-one
The crude 3- (4-nitrophenyl) -3, 6-diazabicyclo [3.1.1] heptan-2-one trifluoroacetate salt from the previous step, 37% aqueous formaldehyde (341mg,4.2mmol) and sodium borohydride acetate (267mg,1.26mmol) were dissolved in DMF (6mL) and reacted at 20 ℃ for 2 hours, quenched with saturated sodium bicarbonate (20mL), extracted with ethyl acetate (30mL), dried over anhydrous sodium sulfate for the organic phase, spun dry and purified by column chromatography (methanol/dichloromethane ═ 4%) to give the product (85mg, 81.9%).
Preparation of 3- (4-aminophenyl) -6-methyl-3, 6-diazabicyclo [3.1.1] heptan-2-one
6-methyl-3- (4-nitrophenyl) -3, 6-diazabicyclo [3.1.1] heptan-2-one (85mg,0.34mmol) was reacted with 10% wet palladium on carbon (8.5mg) in methanol (12mL) at 15 ℃ under hydrogen (15Psi) for 1 hour, filtered with suction, and the filtrate was concentrated to give the product (70mg, 93.7%).
Preparation of 3- (4- ((2-allyl-1- (6- (2-hydroxypropyl-2-yl) pyridin-2-yl) -3-oxo-2, 3-dihydro-1H-pyrazolo [3,4-d ] pyrimidin-6-yl) amino) phenyl) -6-methyl-3, 6-diazabicyclo [3.1.1] heptan-2-one
2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (methylsulfinyl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (103mg,0.27mmol) and 3- (4-aminophenyl) -6-methyl-3, 6-diazabicyclo [3.1.1] heptan-2-one (48mg,0.22mmol) were dissolved in toluene (8mL) and reacted at 40 ℃ for 16 hours, spun dry, medium pressure reverse phase preparation (0-40% acetonitrile/water) to give the product (20mg, 17.2%).
The molecular formula is as follows: c28H30N8O3Molecular weight: 526.6LC-MS (M/e):527.2(M + H)+)
1HNMR(400MHz,CDCl3):δ:10.38(s,1H),8.91(s,1H),8.15-8.05(m,1H),7.80-7.70(m,3H),7.65-7.55(m,1H),7.50-7.40(m,2H),5.70-5.60(m,1H),5.36(s,1H),5.05-4.95(m,1H),4.85-4.75(m,1H),4.75-4.65(m,2H),3.90-3.70(m,3H),3.55-3.45(m,1H),2.60-2.50(m,1H),2.25(m,3H),1.85-1.80(m,1H),1.45(s,6H).
Example 6: preparation of 2-allyl-6- ((4- (8-cyclopropyl-3, 8-diazabicyclo [3.2.1] octan-3-yl) phenyl) amino) -1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (compound 6)
Preparation of 1.8-cyclopropyl-3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane
Dissolving 3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane trifluoroacetate (2.2g, crude product) in methanol (30mL), adding sodium cyanoborohydride (3.8g,60.5mmol), acetic acid (5.7mL,99.6mmol) and (1-ethoxycyclopropoxy) trimethylsilane (3.8g,21.8mmol) in sequence at 0 ℃, reacting for 4h at 60 ℃, detecting the completion of the reaction by LC-MS, diluting the reaction liquid with water, extracting a dichloromethane/methanol (10:1) mixed solvent, drying an organic phase, spin-drying, and separating the residue by column chromatography (dichloromethane: methanol ═ 10:1) to obtain the target compound (1.5 g).
Preparation of 4- (8-cyclopropyl-3, 8-diazabicyclo [3.2.1] octyl-3-yl) aniline
Dissolving 8-cyclopropyl-3- (4-nitrophenyl) -3, 8-diazabicyclo [3.2.1] octane (0.6g, 2.2mmol) in methanol (20mL), adding palladium/carbon (1.0g), replacing with hydrogen for three times, reacting at 25 ℃ for 1.5h, detecting by LC-MS (liquid chromatography-mass spectrometry) to finish the reaction, filtering the reaction solution, and spin-drying the filtrate to obtain a crude target compound (480mg), wherein the crude target compound is directly used in the next step.
Preparation of 2-allyl-6- ((4- (8-cyclopropyl-3, 8-diazabicyclo [3.2.1] oct-3-yl) phenyl) amino) -1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
Dissolving 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (methylsulfinyl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (580mg, crude) in toluene (20mL), adding 4- (8-cyclopropyl-3, 8-diazabicyclo [3.2.1] octyl-3-yl) aniline (400mg, crude) and DIEA (628mg, 4.86mmol), reacting at 25 ℃ for 14H, detecting by LC-MS, draining the reaction solution, separating by thin-layer chromatography (dichloromethane: methanol ═ 12:1, Rf ∼ 0.5) to obtain crude (500mg), separating by reverse phase column chromatography (C18 column, water: methanol ═ 90:10-40:60) to obtain target compound (98mg, yield 11%).
Molecular formula C31H36N8O2Molecular weight 552.7LC-MS (M/z):553.1(M + H)+)
1H-NMR(400MHz,CD3OD)δ:8.82(s,1H),7.88-7.82(m,3H),7.42-7.25(m,3H),6.80-6.73(m,2H),5.75-5.65(m,1H),5.04(d,1H,J=10.4),4.95(d,1H,J=17.2),4.76-4.74(m,2H),4.00(s,1H),3.50-3.46(m,3H),3.38-3.33(m,2H),3.31-2.90(m,2H),2.15-2.00(m,2H),1.93-1.80(m,2H),1.70-1.30(m,6H),0.55-0.45(m,4H).
Example 7: preparation of 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- ((4- (7-methyl-4, 7-diazaspiro [2.5] octyl-4-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (compound 8)
Preparation of tert-butyl 4- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane-7-carboxylate
1-iodo-4-nitrobenzene (2.1g,8.3mmol) was dissolved in toluene (40mL), 4, 7-diazaspiro [2.5] octane-7-carboxylic acid tert-butyl ester (1.5g, 7.1mmol), bis (tri-tert-butylphosphine) palladium (562mg, 1.1mmol) and sodium tert-butoxide (1.3g, 13.8mmol) were added, reacted at 110 ℃ for 4h, LCMS detected that the reaction was complete, the reaction solution was poured into water, EA extracted, dried over anhydrous sodium sulfate, the organic phase was spin-dried, and chromatographed on silica gel column (EA: PE ═ 1: 3) to give the title compound (1.8g, yield 76.0%).
Preparation of 4- (4-Nitrophenyl) -4, 7-diazaspiro [2.5] octane trifluoroacetate
Tert-butyl 4- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane-7-carboxylate (900mg,2.7mmol) was dissolved in dichloromethane (8mL), trifluoroacetic acid (2mL) was added dropwise at 25 ℃ and reacted at 25 ℃ for 1h, the reaction was completed by LCMS detection, and the reaction solution was spin-dried to give a crude product (1.4 g).
Preparation of 3.7-methyl-4- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane
4- (4-Nitrophenyl) -4, 7-diazaspiro [2.5] octane trifluoroacetate (1.4g, crude product) was dissolved in N, N-dimethylformamide (20mL), an aqueous formaldehyde solution (37%) (2.2g, 27mmol) and sodium triacetoxyborohydride (1.7g,8.1mmol) were added, the mixture was left to react at 25 ℃ for 1 hour, water was added to the system to quench the reaction, the organic phase was extracted with dichloromethane, the organic phase was concentrated, and the product was purified with silica gel (methanol/dichloromethane ═ 1:10) to obtain the objective compound (650mg, yield in two steps 97.4%).
Preparation of 4- (7-methyl-4, 7-diazaspiro [2.5] octan-4-yl) aniline
7-methyl-4- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane (650mg, 2.63mmol) was dissolved in methanol (20mL), palladium on carbon (300mg) was added, the mixture was reacted at 25 ℃ for 1h, suction filtered, and the filtrate was concentrated to give a crude product (600mg) which was used directly in the next reaction.
Preparation of 2-allyl-1- (6- (2-hydroxyprop-2-yl) pyridin-2-yl) -6- ((4- (7-methyl-4, 7-diazaspiro [2.5] octyl-4-yl) phenyl) amino) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
2-allyl-1- (6-2-hydroxypropan-2-yl) pyridin-2-yl) -6- (methylsulfinyl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (440mg,1.18mmol) was dissolved in toluene (20mL), 4- (7-methyl-4, 7-diazaspiro [2.5] octan-4-yl) aniline (395mg, crude) and DIEA (458mg, 3.54mmol) were added, the reaction was allowed to react for 16H at 25 ℃, LCMS was checked for completion, the reaction was spun dry, silica gel column chromatography (MeOH: DCM ═ 1:10) to obtain a crude product, which was then slurried with ethyl acetate to obtain the desired compound (15mg, yield 2.6%).
Molecular formula C29H34N8O2Molecular weight 526.6LC-MS (M/e):527.1(M + H)+)
1H-NMR(400MHz,MeOD)δ:8.81(s,1H),7.95-8.05(m,1H),7.70-7.80(m,1H),7.62-7.68(m,1H),7.55-7.61(m,2H),7.06-7.09(m,2H),5.65-5.75(m,1H),5.05(d,J=10.4Hz,1H),4.90-4.96(m,1H),4.80-4.90(m,2H),4.60(s,1H),3.80-4.00(m,2H),2.95-3.10(m,3H),2.81(s,3H),1.59(s,6H),1.10-1.20(m,2H),0.90-1.10(m,2H).
Example 8: preparation of 2-allyl-6- ((4- (4-cyclopropyl-4, 7-diazaspiro [2.5] octan-7-yl) phenyl) amino) -1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (compound 9)
Preparation of tert-butyl 1.7- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane-4-carboxylate
To dimethyl sulfoxide (50.0mL) of 1-fluoro-4-nitrobenzene (1.0g,7.1mmol) were added tert-butyl 4, 7-diazaspiro [2.5] octane-4-carboxylate (1.7g, 8.0mmol) and potassium carbonate (2.0g, 14.2mmol) and reacted at 70 ℃ for 5 hours, the reaction mixture was poured into water, the solid was filtered by suction, the filter cake was washed with water, and the filter cake was spin-dried to give the desired compound (2.4g, yield: 99.7%).
Preparation of 7- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane
After trifluoroacetic acid (6.0mL) was added to 7- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane-4-carboxylic acid tert-butyl ester (1.8g,5.4mmol) in dichloromethane (12.0mL), the mixture was allowed to react at 25 ℃ for 1 hour, the reaction was concentrated and dried, diluted with water, adjusted to alkaline with saturated sodium bicarbonate solution, extracted with dichloromethane, the organic phase was dried over anhydrous sodium sulfate, and concentrated and dried directly for the next step.
Preparation of 4-cyclopropyl-7- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane
After adding acetic acid (1.3g,22.3mmol), sodium cyanoborohydride (841.5mg,13.4mmol) and (1-ethoxycyclopropoxy) trimethylsilane (836.6g,4.8mmol) to methanol (15.0mL) of 7- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane (crude) (500.0mg, 2.1mmol), the mixture was allowed to react at 65 ℃ for 5 hours, the system was quenched with water, extracted with dichloromethane, the organic phase was concentrated, and purified with a silica gel column (ethyl acetate/petroleum ether ═ 1:2) to obtain the objective compound (560.0mg, yield 95.6%).
Preparation of 4- (4-cyclopropyl-4, 7-diazaspiro [2.5] octyl-7-yl) aniline
To 4-cyclopropyl-7- (4-nitrophenyl) -4, 7-diazaspiro [2.5] octane (560.0mg, 2.0mmol) in methanol (6.0mL) was added palladium on charcoal (200mg), and the mixture was reacted at 25 ℃ for 1h, followed by suction filtration, and the filtrate was concentrated to give a crude product (500.0mg) which was used directly in the next reaction.
Preparation of 2-allyl-6- ((4- (4-cyclopropyl-4, 7-diazaspiro [2.5] octan-7-yl) phenyl) amino) -1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one
In 2-allyl-1- (6- (2-hydroxypropan-2-yl) pyridin-2-yl) -6- (methylsulfinyl) -1, 2-dihydro-3H-pyrazolo [3,4-d ] pyrimidin-3-one (crude, 500.0mg,1.3mmol) in toluene (20.0mL), n, N-diisopropylethylamine (840.5mg,6.5mmol) and 4- (4-cyclopropyl-4, 7-diazaspiro [2.5] octan-7-yl) aniline (486.8mg,2.0mmol) were added, and the mixture was reacted at 25 ℃ for 16 hours, quenched with saturated sodium bicarbonate, extracted with dichloromethane, the organic phase was concentrated, and purified with silica gel (methanol/dichloromethane ═ 1:20) to give the title compound (8.5 mg).
Molecular formula C31H36N8O2Molecular weight 552.7LC-MS (M/e):523.0(M + H)+)
1H-NMR(400MHz,CDCl3)δ:10.20-10.05(m,1H),8.81(s,1H),7.95-8.15(m,1H),7.85-7.70(m,1H),7.59-7.61(m,2H),6.90-6.88(m,2H),5.70-5.50(m,2H),5.31(s,1H),5.05-4.95(m,1H),4.80-4.90(m,1H),4.72-4.62(m,2H),3.20-3.10(m,2H),3.05-2.95(m,4H),2.00(s,1H),1.48(s,6H),0.80-0.70(m,2H),0.55-0.40(m,4H),0.32-0.20(m,2H).
Experimental protocol
An exemplary experimental scheme of a portion of the compounds of the invention is provided below to show the advantageous activity and advantageous technical effects of the compounds of the invention. It should be understood, however, that the following experimental protocols are only illustrative of the present disclosure and are not intended to limit the scope of the present disclosure.
Experimental example 1 inhibition of cell proliferation Activity by Compounds of the present invention
Test article: the structural formula and the preparation method of the partial compound are shown in the preparation examples of the invention.
Positive control drug: AZD1775, prepared according to the method disclosed in prior art WO2007126128, and having the structure shown below:
experimental methods
1. Test materials and reagents
| Name (R) | Source | Goods number |
| DMEM high-sugar medium | Gibco | 11995-065 |
| MEM medium | Gibco | 10370-021 |
| FBS (fetal bovine serum) | Gibco | 10099-141C |
| Horse serum | Solarbio | S9050 |
| Pyruvic acid sodium salt | Gibco | 11360-070 |
| Glutamine | Gibco | 35050-061 |
| Penicillin streptomycin double antibody | Gibco | 1507-063 |
| 0.25% Trypsin | Gibco | 25200-072 |
| 10 XDPBS (Du's phosphate buffer) | Gibco | 14200-075 |
| 96-hole bottom transparent white board | Corning | 3610 |
| Gemcitabine (Guitar decitabine) | MCE | HY-B0003 |
| CTG | Promega | G7571 |
2. Cell culture media mix and plating number
3. Procedure of experiment
3.1 preparation of Compounds
Test compounds and reference positive control in 10mM DMSO were diluted in DMSO 3-fold gradient at 8 concentrations. Test compounds and reference positive control 10mM DMSO solutions were prepared in DMSO at 1000-fold concentrations for combination, i.e., 300. mu.M, 100. mu.M, 30. mu.M.
Gemcitabine (Gemcitabine)10mM stock solution was prepared and diluted 1000-fold to the highest final concentration, i.e., 100. mu.M, and then diluted 5 concentrations with a 3-fold gradient of DMSO.
3.2 test procedure
Culturing cells according to the recommended conditions of the source prescription, removing the culture medium, washing with PBS once, digesting with 0.25% trypsin, collecting cells after the complete culture medium is terminated, resuspending to a proper concentration, inoculating to a 96-well plate with 90 μ L/well, 37 deg.C, 5% CO2The incubator was incubated for 24 hours.
The compound is used alone: the final concentration of 1000 times compound solution, medium dilution 100 times, each well of 10 u L,37 degrees C, 5% CO2And (5) detecting after the incubator is cultured for 72 hours.
Combination of compound and gemcitabine: gemcitabine solution 1000 times final concentration, medium diluted 100 times, each well was added with 10. mu.L of 5% CO at 37 ℃2The incubator continues to incubate for 24 hours. The final concentration of the compound solution was 1000 times, the medium was diluted 20 times, and 5. mu.L of the diluted medium was added to each well at 37 ℃ and 5% CO2The incubator is used for detection after 24 hours of continuous culture.
The culture plate is placed at room temperature in advance, 60 mu L of CTG detection solution is added into each hole, the plate is protected from light, the plate is shaken on an orbital shaker for 2 minutes to crack cells, and after the reaction lasts for 20 minutes, the cold light value is read by a multifunctional microplate reader.
4. Data processing
Data were analyzed using GraphPad Prism 8.0 software, fitted to the data using non-linear sigmoidal regression to derive a dose-effect curve, and IC was calculated therefrom50The value is obtained.
Results of the experiment
TABLE 1 in vitro cell inhibitory Activity of Compounds of the invention alone
TABLE 2 in vitro cell inhibitory Activity of the Compounds of the invention in combination with Gemcitabine
Conclusion of the experiment
The compound disclosed by the invention has a good inhibition effect on the cell proliferation activity of A431, SK-MES-1 when being used alone, and has a remarkable synergistic effect on the cell proliferation activity inhibition of MIAPaCa2 when being used together with gemcitabine.
Experimental example 2 Metabolic stability of liver microsomes in various species of the Compound of the present invention
And (3) testing the sample: the chemical name and the preparation method of the compound 3 are shown in the preparation examples of the compound.
Positive control drug: AZD1775, prepared according to methods disclosed in the prior art.
Experimental materials:
cyno monkey mixed liver microsomes were purchased from the reid liver disease research center (shanghai ltd) under the batch number: SHZS, liver microsome protein concentration of 20mg mL-1.
CD-1 mouse, SD rat, Beagle dog, and human mixed liver microsomes were purchased from corning, under the respective lot numbers 9025002 (mouse), 0112002(SD rat), 9259006(Beagle dog), and 38295 (human), and the concentrations of liver microsomes were 20 mg/mL-1.
The experimental initiation factor beta-NADPH is purchased from Solarbio company; phosphate Buffered Saline (PBS) pH 7.4 was self-prepared by the laboratory.
Preparing a test solution:
a proper amount of test powder is precisely weighed, a proper amount of dimethyl sulfoxide (DMSO) is added to dissolve the test powder to 1mM, and the test powder is diluted by 20 times to 50 mu M of working solution by using methanol.
The experimental method comprises the following steps:
TABLE 3 liver microsome metabolic stability experiment incubation system composition
The experimental operation steps are as follows:
(1) according to the ratio of "Experimental incubation System constitution" in Table 3 above, 5.85mL of 100mM PBS and 20mM MgCl were used for each compound2Solution 0.585mL and H2O3.57 mL, and a mixed solution 1 (not containing microsomes, a sample and. beta. -NADPH) for incubation was prepared. The positive pair drug verapamil of the experimental incubation system was also performed to demonstrate normal liver microsomal enzyme activity.
(2) Liver microsomes (20mg protein/mL) were removed from the-80 ℃ freezer and placed on a 37 ℃ water bath constant temperature shaker for pre-incubation for 3 min.
(3) For each compound, 1.9mL of mixed solution 1 of incubation system was taken for each species, and 56. mu.L of microsomes of different species was added to prepare mixed solution 2 of incubation system (containing no test substance and. beta. -NADPH).
(4) Sample set (microsome and β -NADPH containing): and adding 14 mu L of the test sample working solution with the concentration of 50 mu M into 616 mu L of the mixed solution 2 of the incubation system, and adding 70 mu L of 10mM beta-NADPH working solution. Mixing, and repeating the steps. The sampling time points are 0min, 5min, 10min, 20min, 30min and 60 min. This sample set was used to evaluate the metabolic stability of compounds mediated via β -NADPH.
(5) Control group (microsome-containing, no β -NADPH, water instead of β -NADPH): 264 mu L of the mixed solution 2 of the incubation system is taken, 6 mu L of the working solution of the test article with the concentration of 50 mu M is added, and 30 mu L of water is added. Mixing, and repeating the steps. Sampling time points were 0min and 60 min. This negative control group was used to evaluate whether compounds present non- β -NADPH mediated metabolism in the liver microsome incubation system.
(6) At each predetermined time point, 50. mu.L of the incubation sample was sampled from the incubation sample tube, added to a stop sample tube (containing 300. mu.L of cold stop reagent, containing 50ng/mL acetonitrile of the internal standard tolbutamide), vortexed, and the reaction was stopped.
(7) After vortexing for 10min, centrifuge for 5min (12000 rpm).
(8) Taking 100 mu L of supernatant, adding 100 mu L of water, mixing uniformly by vortex, and carrying out LC-MS/MS sample injection analysis.
And (3) data analysis:
the percent residual was converted by the ratio of the peak area of the test article to the internal standard in the following equation.
The experimental results are as follows:
TABLE 4 hepatic microsome stability results for the compounds of the invention
The experimental conclusion is that:
the compound has good stability in human, monkey, dog, rat and mouse liver microsomes, and the stability in the mouse liver microsomes is equivalent to that of a control; in the other species of liver microsomes, the stability of the compounds of the invention was significantly better than that of the control AZD 1775.
Experimental example 3 inhibition of in vitro kinase Activity by Compounds of the present invention
Test article: the structural formula and the preparation method of the partial compound are shown in the preparation examples of the invention. The abbreviations used in the following experiments have the following meanings:
DMSO, DMSO: dimethyl sulfoxide; HEPES (high efficiency particulate air): hydroxyethyl piperazine ethanethiosulfonic acid.
The experimental method comprises the following steps: compounds were evaluated on WEE1 Kinase in vitro using the ADP-Glo Kinase Assay.
The experimental steps are as follows:
1. compound dilution:
test compounds (10mM stock) were diluted 100-fold with 100% DMSO in 384 dilution plates (labcyte, PP-0200) at 1: 3, carrying out equal ratio dilution on the concentration of the compound to be tested: 100. mu.M, 33.33. mu.M, 11.11. mu.M, 3.7. mu.M, 1.23. mu.M, 0.41. mu.M, 0.137. mu.M, 0.046. mu.M, 0.015. mu.M, 0.005. mu.M, 0. mu.M.
2. Transfer 0.1. mu.L of test compound to 384 reaction plates (PE,6007290) with Echo, 1000rpm/min, centrifuge for 1 min.
3. Transfer 5. mu.L of kinase to 384 reaction plates, 1000rpm/min, centrifuge for 1min, and incubate for 15min at 25 ℃.
4. Transfer 5. mu.L of substrate mixture to 384 reaction plates, 1000rpm/min, centrifuge for 1min, and incubate at 25 ℃ for 60 min. In the reaction, the final concentration of the test compound was 1000nM, 333.33nM, 111.11nM, 37.04nM, 12.35nM, 4.12nM, 1.37nM, 0.46nM, 0.15nM, 0.05nM, 0 nM. The final concentration of DMSO was 1%.
5. Transfer 5. mu.L ADP-Glo to 384 reaction plates at 1000rpm/min, centrifuge for 1min, and incubate at 25 ℃ for 60 min.
6. Transfer 10. mu.L of Detection solution to 384 reaction plates at 1000rpm/min, centrifuge for 1min, and incubate for 60min at 25 ℃.
7. The RLU (relative luminescence unit) signal is read using an Envision multifunction board reader.
8. Data computation
1) The mean data and Standard Deviation (SD) of DMSO and the positive control compound AZD-1775 (100% inhibition at 1000 nM) were calculated for each sieve plate as high and low controls.
2) Percent inhibition (% inh) 100 × (max-compound signal)/(max-min)
"minimum" is the negative control well reading without compound added; "Max" is the reading of the positive control well plus positive control compound AZD-1775 (100% inhibition at 1000 nM).
3) Fitting Compound IC Using non-Linear regression equation50。
The experimental results are as follows:
TABLE 5 in vitro enzymatic inhibitory Activity of the Compounds of the invention
And (4) experimental conclusion:
the compound of the invention has good inhibition activity on WEE 1.
Claims (10)
1. A compound shown as a formula (I), pharmaceutically acceptable salt thereof or stereoisomer thereof,
wherein,
X1、X2、X3are each independently selected from-C (R)2) -or-N-;
X4、X5each independently selected from-CH-or-N-;
each L is independently selected from-C (R)3)(R4)-、-O-、-N(R5) -or-S-;
ring A is selected from a 3-10 membered cycloalkyl, 3-10 membered heterocyclyl, 6-10 membered aryl or 5-10 membered heteroaryl optionally substituted with 1-4 of Q1;
ring B is selected from 3-10 membered cycloalkyl, 3-10 membered heterocyclyl, 6-10 membered aryl or 5-10 membered heteroaryl optionally substituted with 1-4Q 2;
ring C is selected from 5-15 membered bridged ring group, 5-10 membered spiro ring group, 5-15 membered bridged ring group or 5-10 membered spiroheterocyclic group optionally substituted with 1-4 of Q3, wherein ring carbon atoms in said 5-15 membered bridged ring group, 5-10 membered spiro ring group, 5-15 membered bridged ring group or 5-10 membered spiroheterocyclic group are optionally substituted with oxygen to form a carbonyl group;
each Q1 is uniqueSelected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
each Q2, each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, halo C1-6Alkoxy, halo C1-6Alkylthio, hydroxy C1-6Alkoxy, hydroxy C1-6Alkylthio, amino C1-6Alkoxy, amino C1-6Alkylthio, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
R1selected from C optionally substituted by substituents1-6Alkyl radical, C2-6Alkenyl or C2-6Alkynyl, the substituent groups are respectively and independently selected from halogen, hydroxyl, amino, carboxyl, cyano, C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkoxy or C1-6An alkylcarbonyl group;
R2、R3、R4、R5each independently selected from hydrogen, halogen, hydroxyl, amino, carboxyl and C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl, halo C1-6Alkoxy or C1-6An alkylcarbonyl group;
m and n are respectively and independently selected from 0, 1,2 or 3.
2. The compound of claim 1, a pharmaceutically acceptable salt thereof, or a stereoisomer thereof,
ring a is selected from phenyl or a 5-6 membered nitrogen containing monoheteroaryl optionally substituted with 1-3Q 1;
each Q1 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy or the following optionally substituted with 1-3 substituents: c1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylthio, each of which substituents is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-3-10 membered cycloalkyl, - (CH)2)m-3-10 membered heterocycloalkyl, - (CH)2)m-5-10 membered heteroaryl or- (CH)2)m-6-10 membered aryl;
ring B is selected from phenyl or 5-6 membered mono heteroaryl optionally substituted with 1 or 2Q 2; each Q2 is independently selected from halogen, hydroxy, amino, nitro, cyano, C1-6Alkyl radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy, hydroxy C1-6Alkoxy or amino C1-6An alkoxy group.
3. The compound of any one of claims 1-2, a pharmaceutically acceptable salt thereof, or a stereoisomer thereof, wherein:
ring C is selected from the following optionally substituted with 1-2Q 3: 2-azabicyclo [2.2.1] heptanyl, 7-azabicyclo [2.2.1] heptanyl, 3-azabicyclo [3.2.1] octanyl, 8-azabicyclo [3.2.1] octanyl, 2-azabicyclo [2.2.2] octanyl, 2, 5-diazabicyclo [2.2.1] heptanyl, 3, 6-diazabicyclo [3.1.1] heptanyl, 3, 8-diazabicyclo [3.2.1] octanyl, 2-oxa-5-azabicyclo [2.2.1] heptanyl, 8-oxa-3-azabicyclo [3.2.1] octanyl, 3, 8-diazabicyclo [3.2.1] octan-6-enyl, 3, 9-diazabicyclo [3.3.1] nonanyl, 5-azabicyclo [4.1 ] heptanyl, 2-azaspiro [3.3] heptylalkyl, 2-azaspiro [3.5] nonanyl, 2, 6-diazaspiro [3.3] heptanyl, 2, 7-diazaspiro [3.5] nonanyl, 2, 5-diazaspiro [3.4] octanyl, 2-oxa-6-azaspiro [3.3] heptanyl, 6-oxa-2-azaspiro [3.4] octanyl, 6-azaspiro [3.4] octanyl, 2-azaspiro [4.4] nonanyl, 2-oxa-7-azaspiro [4.4] nonanyl, 6-azaspiro [3.4] oct-7-enyl, 2-oxa-6-azaspiro [3.4] oct-7-enyl or 2-azaspiro [4.4] non-7-enyl, wherein the ring-forming carbon atoms of the group are optionally substituted with oxygen to form a carbonyl group;
each Q3 is independently selected from halogen, hydroxy, amino, nitro, cyano, carboxy, C1-6Alkyl, halo C1-6Alkyl, hydroxy C1-6Alkyl radical, C1-6Alkoxy, halo C1-6Alkoxy or hydroxy C1-6An alkoxy group.
4. The compound, pharmaceutically acceptable salt thereof, or stereoisomer thereof according to any one of claims 1-3,
X1、X2、X3are each independently selected from-C (R)2) -or-N-;
X4、X5each independently selected from-CH-or-N-;
each L is independently selected from-C (R)3)(R4)-、-O-、-N(R5) -or-S-;
R2、R3、R4、R5each independently selected from hydrogen, methyl, ethyl, propyl, isopropyl, methoxy, ethoxy, propoxy, isopropoxy, monofluoromethyl, difluoromethyl, trifluoromethyl or trifluoromethoxy;
R1selected from C optionally substituted by substituents2-6Alkenyl, the substituents are each independentlyIs selected from halogen, hydroxy, amino, carboxyl, cyano, C1-6Alkyl radical, C1-6Alkoxy radical, C1-6Alkylamino radical, di (C)1-6Alkyl) amino, halo C1-6Alkyl, hydroxy C1-6Alkyl, amino C1-6Alkyl or halo C1-6An alkoxy group.
6. The compound, pharmaceutically acceptable salt thereof, or stereoisomer thereof according to any one of claims 1-5,
l is NH;
ring a is selected from pyridyl, pyrimidinyl, pyrazinyl, or pyridazinyl substituted with 1Q 1; q1 is selected from fluoro, chloro, bromo, hydroxy, amino, nitro, cyano or the following optionally substituted with 1-3 substituents: methyl, ethyl, propyl, isopropyl, butyl, isobutyl, sec-butyl, tert-butyl, methoxy, ethoxy, propoxy and isopropoxy, wherein the substituents are independently selected from fluoro, chloro, bromo, hydroxy, amino, nitro, cyano, carboxy, - (CH)2)m-cyclopropane, - (CH)2)m-cyclobutyl, - (CH)2)m-cyclopentyl or- (CH)2)m-a cyclohexane group;
ring B is selected from phenyl, pyridyl, pyrimidinyl, pyrazinyl, or pyridazinyl;
m is selected from 0, 1 or 2;
n is 1.
8. a pharmaceutical formulation comprising a compound according to any one of claims 1 to 7, a pharmaceutically acceptable salt thereof or a stereoisomer thereof, in a pharmaceutically acceptable dosage form, comprising one or more pharmaceutically acceptable excipients.
9. A pharmaceutical composition comprising a compound, pharmaceutically acceptable salt thereof, or stereoisomer thereof according to any one of claims 1-7, comprising one or more second therapeutically active agent selected from mitotic inhibitors, alkylating agents, antimetabolites, antisense DNA or RNA, antitumor antibiotics, growth factor inhibitors, signaling inhibitors, cell cycle inhibitors, enzyme inhibitors, retinoid receptor modulators, proteasome inhibitors, topoisomerase inhibitors, biological response modifiers, hormonal drugs, angiogenesis inhibitors, cell growth inhibitors, targeting antibodies, HMG-CoA reductase inhibitors, and prenyl protein transferase inhibitors.
10. Use of a compound of any one of claims 1-7, a pharmaceutically acceptable salt or stereoisomer thereof, a pharmaceutical preparation of claim 8, or a pharmaceutical composition of claim 9 for the manufacture of a medicament for the treatment and/or prevention of a disease and related disorder mediated by Wee1 selected from cancer or benign tumor selected from lung cancer, squamous cell carcinoma, bladder cancer, gastric cancer, ovarian cancer, peritoneal cancer, pancreatic cancer, breast cancer, head and neck cancer, cervical cancer, endometrial cancer, rectal cancer, liver cancer, kidney cancer, esophageal adenocarcinoma, esophageal squamous cell carcinoma, prostate cancer, thyroid cancer, female genital tract cancer, lymphoma, neurofibroma, bone cancer, skin cancer, brain cancer, colon cancer, testicular cancer, small cell lung cancer, non-small cell lung cancer, cancer of the stomach, cancer, squamous cell carcinoma of the esophagus, cancer of the prostate, thyroid cancer, cancer of the female genital tract, lymphoma, neurofibroma, bone cancer, skin cancer, brain cancer, colon cancer, testicular cancer, small cell lung cancer, non-cell lung cancer, cancer of the stomach cancer, cancer of the subject for example, cancer of the subject for example, cancer, Gastrointestinal stromal tumors, mast cell tumors, multiple myeloma, melanoma, leukemia, glioma, or sarcoma.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2020114072937 | 2020-12-04 | ||
| CN202011407293 | 2020-12-04 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN114591334A true CN114591334A (en) | 2022-06-07 |
| CN114591334B CN114591334B (en) | 2023-10-20 |
Family
ID=81813674
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111465345.0A Active CN114591334B (en) | 2020-12-04 | 2021-12-03 | Dihydropyrazolopyrimidinone derivatives |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114591334B (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023179196A1 (en) * | 2022-03-25 | 2023-09-28 | 药雅科技(上海)有限公司 | Preparation and application of wee1 kinase inhibitor |
| CN117327077A (en) * | 2023-10-07 | 2024-01-02 | 中国药科大学 | Anilinopyrimidino heterocyclic compound, and preparation method, pharmaceutical composition and application thereof |
Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009054332A1 (en) * | 2007-10-23 | 2009-04-30 | Banyu Pharmaceutical Co., Ltd. | Pyridone-substituted-dihydropyrazolopyrimidinone derivative |
| CN109422754A (en) * | 2017-08-24 | 2019-03-05 | 上海迪诺医药科技有限公司 | Pyrazolo [3,4-d] pyrimidine -3- ketone derivatives, its pharmaceutical composition and application |
| WO2019165204A1 (en) * | 2018-02-23 | 2019-08-29 | Newave Pharmaceutical Inc. | 1,2-dihydro-3h-pyrazolo[3,4-d]pyrimidine-3-one compounds as inhibitors of wee-1 kinase |
| CN110198943A (en) * | 2017-01-23 | 2019-09-03 | 石家庄智康弘仁新药开发有限公司 | 1,2-Dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one derivatives as Wee1 inhibitors |
| CN110872296A (en) * | 2018-08-31 | 2020-03-10 | 上海弘翊生物科技有限公司 | isoindoline-1H-pyrazolo [3,4-d ] pyrimidone compound, and preparation method and application thereof |
| CN111315747A (en) * | 2018-01-05 | 2020-06-19 | 四川科伦博泰生物医药股份有限公司 | Dihydropyrazolopyrimidine compounds and preparation method and use thereof |
| CN112142747A (en) * | 2019-06-28 | 2020-12-29 | 上海医药集团股份有限公司 | A kind of pyrazolone pyrimidine compound, its preparation method and application |
| CN112142748A (en) * | 2019-06-28 | 2020-12-29 | 上海医药集团股份有限公司 | Pyrazolone pyrimidine compound, preparation method and application thereof |
| CN113387962A (en) * | 2020-03-12 | 2021-09-14 | 上海迪诺医药科技有限公司 | Pyrazolo [3,4-d ] pyrimidine-3-one derivative, pharmaceutical composition and application thereof |
| CN115698006A (en) * | 2020-06-17 | 2023-02-03 | 微境生物医药科技(上海)有限公司 | Pyrazolo[3,4-d]pyrimidin-3-one derivatives as Wee-1 inhibitors |
-
2021
- 2021-12-03 CN CN202111465345.0A patent/CN114591334B/en active Active
Patent Citations (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009054332A1 (en) * | 2007-10-23 | 2009-04-30 | Banyu Pharmaceutical Co., Ltd. | Pyridone-substituted-dihydropyrazolopyrimidinone derivative |
| US20100221211A1 (en) * | 2007-10-23 | 2010-09-02 | Hidetomo Furuyama | Pyridone-substituted-dihydropyrazolopyrimidinone derivative |
| CN110198943A (en) * | 2017-01-23 | 2019-09-03 | 石家庄智康弘仁新药开发有限公司 | 1,2-Dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one derivatives as Wee1 inhibitors |
| CN109422754A (en) * | 2017-08-24 | 2019-03-05 | 上海迪诺医药科技有限公司 | Pyrazolo [3,4-d] pyrimidine -3- ketone derivatives, its pharmaceutical composition and application |
| CN111315747A (en) * | 2018-01-05 | 2020-06-19 | 四川科伦博泰生物医药股份有限公司 | Dihydropyrazolopyrimidine compounds and preparation method and use thereof |
| WO2019165204A1 (en) * | 2018-02-23 | 2019-08-29 | Newave Pharmaceutical Inc. | 1,2-dihydro-3h-pyrazolo[3,4-d]pyrimidine-3-one compounds as inhibitors of wee-1 kinase |
| CN110872296A (en) * | 2018-08-31 | 2020-03-10 | 上海弘翊生物科技有限公司 | isoindoline-1H-pyrazolo [3,4-d ] pyrimidone compound, and preparation method and application thereof |
| CN112142747A (en) * | 2019-06-28 | 2020-12-29 | 上海医药集团股份有限公司 | A kind of pyrazolone pyrimidine compound, its preparation method and application |
| CN112142748A (en) * | 2019-06-28 | 2020-12-29 | 上海医药集团股份有限公司 | Pyrazolone pyrimidine compound, preparation method and application thereof |
| CN113387962A (en) * | 2020-03-12 | 2021-09-14 | 上海迪诺医药科技有限公司 | Pyrazolo [3,4-d ] pyrimidine-3-one derivative, pharmaceutical composition and application thereof |
| CN115698006A (en) * | 2020-06-17 | 2023-02-03 | 微境生物医药科技(上海)有限公司 | Pyrazolo[3,4-d]pyrimidin-3-one derivatives as Wee-1 inhibitors |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2023179196A1 (en) * | 2022-03-25 | 2023-09-28 | 药雅科技(上海)有限公司 | Preparation and application of wee1 kinase inhibitor |
| CN117327077A (en) * | 2023-10-07 | 2024-01-02 | 中国药科大学 | Anilinopyrimidino heterocyclic compound, and preparation method, pharmaceutical composition and application thereof |
| WO2025073221A1 (en) * | 2023-10-07 | 2025-04-10 | 中国药科大学 | Anilinopyrimido heterocyclic compound, and preparation method therefor, pharmaceutical composition thereof and use thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| CN114591334B (en) | 2023-10-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6726677B2 (en) | Substituted 2-H-pyrazole derivatives as anticancer agents | |
| JP5752232B2 (en) | Substituted pyrrolotriazine compounds as protein kinase inhibitors | |
| TWI751163B (en) | Fgfr4 inhibitor, its preparation method and use thereof | |
| CA3092770A1 (en) | Substituted imidazolidin-2-one derivatives as prmt5 inhibitors | |
| AU2012310168B2 (en) | 6 - substituted 3 - (quinolin- 6 - ylthio) - [1,2,4] triazolo [4, 3 -a] pyradines as tyrosine kinase | |
| CN112939967A (en) | Pyrazolo [1,5-a ] pyridine compound and preparation method and application thereof | |
| CN111171049B (en) | Tyrosine kinase inhibitors and uses thereof | |
| JP6667014B2 (en) | Pyrimidine derivatives, their preparation and use in medicine | |
| CN118684684B (en) | Compounds as WRN helicase inhibitors | |
| WO2018167276A1 (en) | Tricyclic compounds for use in treatment of proliferative disorders | |
| CN116323562B (en) | A class of compounds having kinase inhibitory activity | |
| CN118369322A (en) | Substituted monocyclic or bicyclic heterocyclic compounds, their preparation and medical use | |
| CN121013849A (en) | Heterocyclic compounds as WRN inhibitors | |
| KR20250048262A (en) | Aza-quinazoline compounds and methods of use | |
| CN114591334B (en) | Dihydropyrazolopyrimidinone derivatives | |
| KR20210108433A (en) | Thienopyridinone compound | |
| CN115197221A (en) | Dihydropyrazolopyrimidinone macrocyclic derivatives and application thereof | |
| TW202310845A (en) | Heteroaryl derivative compounds, and uses thereof | |
| TW202214634A (en) | Heterocyclic compound and derivative thereof | |
| JP6977038B2 (en) | GSK-3 inhibitor | |
| TWI749518B (en) | Piperazine amide derivative, its preparation method and its use in medicine | |
| CA3216045A1 (en) | Compounds as pd1/pd-l1 inhibitors and methods thereof | |
| CN115707708A (en) | Wee1 kinase inhibitors | |
| US20240360145A1 (en) | Pyrimidine-2,4-diamine derivatives as well as preparation method therefor and use thereof | |
| TW202519531A (en) | Crystal form of propylene ketone-containing bioinhibitor, preparation method therefor and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |