CN115707708A - Wee1 kinase inhibitors - Google Patents
Wee1 kinase inhibitors Download PDFInfo
- Publication number
- CN115707708A CN115707708A CN202110952817.9A CN202110952817A CN115707708A CN 115707708 A CN115707708 A CN 115707708A CN 202110952817 A CN202110952817 A CN 202110952817A CN 115707708 A CN115707708 A CN 115707708A
- Authority
- CN
- China
- Prior art keywords
- alkyl
- amino
- alkoxy
- halogenated
- ring
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 101150040313 Wee1 gene Proteins 0.000 title claims description 16
- 229940043355 kinase inhibitor Drugs 0.000 title abstract description 5
- 239000003757 phosphotransferase inhibitor Substances 0.000 title abstract description 5
- 150000001875 compounds Chemical class 0.000 claims abstract description 73
- 150000003839 salts Chemical class 0.000 claims abstract description 49
- 238000002360 preparation method Methods 0.000 claims abstract description 21
- 239000003814 drug Substances 0.000 claims abstract description 16
- 239000008194 pharmaceutical composition Substances 0.000 claims abstract description 9
- 239000000825 pharmaceutical preparation Substances 0.000 claims abstract description 9
- 125000000217 alkyl group Chemical group 0.000 claims description 237
- 125000003545 alkoxy group Chemical group 0.000 claims description 182
- -1 amino, carboxyl Chemical group 0.000 claims description 159
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 124
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 80
- 125000006620 amino-(C1-C6) alkyl group Chemical group 0.000 claims description 49
- 229910052736 halogen Inorganic materials 0.000 claims description 49
- 150000002367 halogens Chemical class 0.000 claims description 49
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 claims description 43
- 125000005842 heteroatom Chemical group 0.000 claims description 39
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 35
- 206010028980 Neoplasm Diseases 0.000 claims description 26
- 125000003373 pyrazinyl group Chemical group 0.000 claims description 26
- 125000002098 pyridazinyl group Chemical group 0.000 claims description 26
- 125000004076 pyridyl group Chemical group 0.000 claims description 26
- 125000000714 pyrimidinyl group Chemical group 0.000 claims description 26
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 claims description 25
- 201000011510 cancer Diseases 0.000 claims description 25
- 125000004306 triazinyl group Chemical group 0.000 claims description 25
- 125000006619 (C1-C6) dialkylamino group Chemical group 0.000 claims description 24
- 229910020008 S(O) Inorganic materials 0.000 claims description 24
- 125000000475 sulfinyl group Chemical group [*:2]S([*:1])=O 0.000 claims description 24
- 229910052757 nitrogen Inorganic materials 0.000 claims description 20
- 229910052760 oxygen Inorganic materials 0.000 claims description 20
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 19
- 125000003118 aryl group Chemical group 0.000 claims description 18
- 229910052717 sulfur Inorganic materials 0.000 claims description 18
- 201000010099 disease Diseases 0.000 claims description 16
- 125000000623 heterocyclic group Chemical group 0.000 claims description 16
- 230000001093 anti-cancer Effects 0.000 claims description 15
- 125000002883 imidazolyl group Chemical group 0.000 claims description 15
- 125000000168 pyrrolyl group Chemical group 0.000 claims description 15
- 150000001335 aliphatic alkanes Chemical class 0.000 claims description 14
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 14
- 125000002971 oxazolyl group Chemical group 0.000 claims description 14
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 14
- 125000000335 thiazolyl group Chemical group 0.000 claims description 14
- 125000004916 (C1-C6) alkylcarbonyl group Chemical group 0.000 claims description 13
- QWENRTYMTSOGBR-UHFFFAOYSA-N 1H-1,2,3-Triazole Chemical group C=1C=NNN=1 QWENRTYMTSOGBR-UHFFFAOYSA-N 0.000 claims description 13
- NSPMIYGKQJPBQR-UHFFFAOYSA-N 4H-1,2,4-triazole Chemical group C=1N=CNN=1 NSPMIYGKQJPBQR-UHFFFAOYSA-N 0.000 claims description 13
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 12
- 229940079593 drug Drugs 0.000 claims description 12
- 125000001072 heteroaryl group Chemical group 0.000 claims description 12
- 125000004435 hydrogen atom Chemical class [H]* 0.000 claims description 12
- 125000004454 (C1-C6) alkoxycarbonyl group Chemical group 0.000 claims description 11
- 125000002541 furyl group Chemical group 0.000 claims description 11
- 229910052739 hydrogen Inorganic materials 0.000 claims description 11
- 239000001257 hydrogen Substances 0.000 claims description 11
- 239000003112 inhibitor Substances 0.000 claims description 10
- 125000002911 monocyclic heterocycle group Chemical group 0.000 claims description 10
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 claims description 10
- 125000001544 thienyl group Chemical group 0.000 claims description 10
- 239000013543 active substance Substances 0.000 claims description 9
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 claims description 9
- 125000004309 pyranyl group Chemical group O1C(C=CC=C1)* 0.000 claims description 9
- 229910052799 carbon Inorganic materials 0.000 claims description 8
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 claims description 8
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 claims description 7
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 7
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 7
- 230000001404 mediated effect Effects 0.000 claims description 7
- 125000002950 monocyclic group Chemical group 0.000 claims description 7
- 239000001301 oxygen Substances 0.000 claims description 7
- 125000003342 alkenyl group Chemical group 0.000 claims description 6
- 125000004429 atom Chemical group 0.000 claims description 6
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 6
- BASFCYQUMIYNBI-UHFFFAOYSA-N platinum Chemical compound [Pt] BASFCYQUMIYNBI-UHFFFAOYSA-N 0.000 claims description 6
- 125000000304 alkynyl group Chemical group 0.000 claims description 5
- 125000001624 naphthyl group Chemical group 0.000 claims description 5
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 claims description 5
- 125000002568 propynyl group Chemical group [*]C#CC([H])([H])[H] 0.000 claims description 5
- 125000000882 C2-C6 alkenyl group Chemical group 0.000 claims description 4
- 125000003601 C2-C6 alkynyl group Chemical group 0.000 claims description 4
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Chemical group C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 claims description 4
- 230000000259 anti-tumor effect Effects 0.000 claims description 4
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 claims description 4
- 125000001424 substituent group Chemical group 0.000 claims description 4
- 125000003718 tetrahydrofuranyl group Chemical group 0.000 claims description 4
- 125000001984 thiazolidinyl group Chemical group 0.000 claims description 4
- 238000011282 treatment Methods 0.000 claims description 4
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 claims description 4
- 229920002554 vinyl polymer Chemical group 0.000 claims description 4
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 claims description 3
- 125000000041 C6-C10 aryl group Chemical group 0.000 claims description 3
- 229940123587 Cell cycle inhibitor Drugs 0.000 claims description 3
- 206010009944 Colon cancer Diseases 0.000 claims description 3
- 102000005454 Dimethylallyltranstransferase Human genes 0.000 claims description 3
- 108010006731 Dimethylallyltranstransferase Proteins 0.000 claims description 3
- 229940121710 HMGCoA reductase inhibitor Drugs 0.000 claims description 3
- 102000014150 Interferons Human genes 0.000 claims description 3
- 108010050904 Interferons Proteins 0.000 claims description 3
- 206010033128 Ovarian cancer Diseases 0.000 claims description 3
- 206010061535 Ovarian neoplasm Diseases 0.000 claims description 3
- 206010061902 Pancreatic neoplasm Diseases 0.000 claims description 3
- 229940079156 Proteasome inhibitor Drugs 0.000 claims description 3
- 239000002168 alkylating agent Substances 0.000 claims description 3
- 229940100198 alkylating agent Drugs 0.000 claims description 3
- 239000004037 angiogenesis inhibitor Substances 0.000 claims description 3
- 229940121369 angiogenesis inhibitor Drugs 0.000 claims description 3
- 239000005557 antagonist Substances 0.000 claims description 3
- 239000003242 anti bacterial agent Substances 0.000 claims description 3
- 229940088710 antibiotic agent Drugs 0.000 claims description 3
- 239000003101 antineoplastic hormone agonist and antagonist Substances 0.000 claims description 3
- 239000003886 aromatase inhibitor Substances 0.000 claims description 3
- VSJKWCGYPAHWDS-FQEVSTJZSA-N camptothecin Chemical class C1=CC=C2C=C(CN3C4=CC5=C(C3=O)COC(=O)[C@]5(O)CC)C4=NC2=C1 VSJKWCGYPAHWDS-FQEVSTJZSA-N 0.000 claims description 3
- 239000000824 cytostatic agent Substances 0.000 claims description 3
- 208000035475 disorder Diseases 0.000 claims description 3
- 239000003534 dna topoisomerase inhibitor Substances 0.000 claims description 3
- 239000002552 dosage form Substances 0.000 claims description 3
- 239000003102 growth factor Substances 0.000 claims description 3
- 239000002471 hydroxymethylglutaryl coenzyme A reductase inhibitor Substances 0.000 claims description 3
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 claims description 3
- 230000002503 metabolic effect Effects 0.000 claims description 3
- 201000002528 pancreatic cancer Diseases 0.000 claims description 3
- 208000008443 pancreatic carcinoma Diseases 0.000 claims description 3
- 229940096701 plain lipid modifying drug hmg coa reductase inhibitors Drugs 0.000 claims description 3
- 229910052697 platinum Inorganic materials 0.000 claims description 3
- 239000003207 proteasome inhibitor Substances 0.000 claims description 3
- 102000027483 retinoid hormone receptors Human genes 0.000 claims description 3
- 108091008679 retinoid hormone receptors Proteins 0.000 claims description 3
- 206010041823 squamous cell carcinoma Diseases 0.000 claims description 3
- 230000008685 targeting Effects 0.000 claims description 3
- 125000003554 tetrahydropyrrolyl group Chemical group 0.000 claims description 3
- 125000003507 tetrahydrothiofenyl group Chemical group 0.000 claims description 3
- 229940044693 topoisomerase inhibitor Drugs 0.000 claims description 3
- 239000003558 transferase inhibitor Substances 0.000 claims description 3
- 229940121358 tyrosine kinase inhibitor Drugs 0.000 claims description 3
- 239000005483 tyrosine kinase inhibitor Substances 0.000 claims description 3
- 125000006714 (C3-C10) heterocyclyl group Chemical group 0.000 claims description 2
- 208000036764 Adenocarcinoma of the esophagus Diseases 0.000 claims description 2
- 206010005003 Bladder cancer Diseases 0.000 claims description 2
- 206010005949 Bone cancer Diseases 0.000 claims description 2
- 208000018084 Bone neoplasm Diseases 0.000 claims description 2
- 208000003174 Brain Neoplasms Diseases 0.000 claims description 2
- 206010006187 Breast cancer Diseases 0.000 claims description 2
- 208000026310 Breast neoplasm Diseases 0.000 claims description 2
- 206010008342 Cervix carcinoma Diseases 0.000 claims description 2
- 206010014733 Endometrial cancer Diseases 0.000 claims description 2
- 206010014759 Endometrial neoplasm Diseases 0.000 claims description 2
- 206010051066 Gastrointestinal stromal tumour Diseases 0.000 claims description 2
- 208000032612 Glial tumor Diseases 0.000 claims description 2
- 206010018338 Glioma Diseases 0.000 claims description 2
- 208000008839 Kidney Neoplasms Diseases 0.000 claims description 2
- 206010058467 Lung neoplasm malignant Diseases 0.000 claims description 2
- 206010025323 Lymphomas Diseases 0.000 claims description 2
- 208000034578 Multiple myelomas Diseases 0.000 claims description 2
- 201000004404 Neurofibroma Diseases 0.000 claims description 2
- 206010030137 Oesophageal adenocarcinoma Diseases 0.000 claims description 2
- 206010061534 Oesophageal squamous cell carcinoma Diseases 0.000 claims description 2
- ZCQWOFVYLHDMMC-UHFFFAOYSA-N Oxazole Chemical compound C1=COC=N1 ZCQWOFVYLHDMMC-UHFFFAOYSA-N 0.000 claims description 2
- 206010035226 Plasma cell myeloma Diseases 0.000 claims description 2
- 206010060862 Prostate cancer Diseases 0.000 claims description 2
- 208000000236 Prostatic Neoplasms Diseases 0.000 claims description 2
- 208000015634 Rectal Neoplasms Diseases 0.000 claims description 2
- 206010038389 Renal cancer Diseases 0.000 claims description 2
- 206010039491 Sarcoma Diseases 0.000 claims description 2
- 208000000453 Skin Neoplasms Diseases 0.000 claims description 2
- 206010041067 Small cell lung cancer Diseases 0.000 claims description 2
- 208000036765 Squamous cell carcinoma of the esophagus Diseases 0.000 claims description 2
- 208000005718 Stomach Neoplasms Diseases 0.000 claims description 2
- 208000024313 Testicular Neoplasms Diseases 0.000 claims description 2
- 206010057644 Testis cancer Diseases 0.000 claims description 2
- 208000024770 Thyroid neoplasm Diseases 0.000 claims description 2
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 claims description 2
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 claims description 2
- 239000002246 antineoplastic agent Substances 0.000 claims description 2
- 229940046844 aromatase inhibitors Drugs 0.000 claims description 2
- 230000008512 biological response Effects 0.000 claims description 2
- 201000010881 cervical cancer Diseases 0.000 claims description 2
- 208000029742 colonic neoplasm Diseases 0.000 claims description 2
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 2
- 208000028653 esophageal adenocarcinoma Diseases 0.000 claims description 2
- 208000007276 esophageal squamous cell carcinoma Diseases 0.000 claims description 2
- 210000005002 female reproductive tract Anatomy 0.000 claims description 2
- 206010017758 gastric cancer Diseases 0.000 claims description 2
- 201000011243 gastrointestinal stromal tumor Diseases 0.000 claims description 2
- 201000010536 head and neck cancer Diseases 0.000 claims description 2
- 208000014829 head and neck neoplasm Diseases 0.000 claims description 2
- 229940047124 interferons Drugs 0.000 claims description 2
- 201000010982 kidney cancer Diseases 0.000 claims description 2
- 208000032839 leukemia Diseases 0.000 claims description 2
- 201000007270 liver cancer Diseases 0.000 claims description 2
- 208000014018 liver neoplasm Diseases 0.000 claims description 2
- 201000005202 lung cancer Diseases 0.000 claims description 2
- 208000020816 lung neoplasm Diseases 0.000 claims description 2
- 201000006512 mast cell neoplasm Diseases 0.000 claims description 2
- 201000001441 melanoma Diseases 0.000 claims description 2
- 230000011278 mitosis Effects 0.000 claims description 2
- 208000002154 non-small cell lung carcinoma Diseases 0.000 claims description 2
- 201000002628 peritoneum cancer Diseases 0.000 claims description 2
- 230000002265 prevention Effects 0.000 claims description 2
- 206010038038 rectal cancer Diseases 0.000 claims description 2
- 201000001275 rectum cancer Diseases 0.000 claims description 2
- 201000000849 skin cancer Diseases 0.000 claims description 2
- 208000000587 small cell lung carcinoma Diseases 0.000 claims description 2
- 201000011549 stomach cancer Diseases 0.000 claims description 2
- 201000003120 testicular cancer Diseases 0.000 claims description 2
- 201000002510 thyroid cancer Diseases 0.000 claims description 2
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 claims description 2
- 201000005112 urinary bladder cancer Diseases 0.000 claims description 2
- 239000000546 pharmaceutical excipient Substances 0.000 claims 1
- 230000011664 signaling Effects 0.000 claims 1
- 238000000034 method Methods 0.000 abstract description 12
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 21
- 229920006395 saturated elastomer Polymers 0.000 description 20
- 125000004433 nitrogen atom Chemical group N* 0.000 description 16
- 239000000243 solution Substances 0.000 description 16
- 210000004027 cell Anatomy 0.000 description 13
- 125000004432 carbon atom Chemical group C* 0.000 description 12
- 230000000694 effects Effects 0.000 description 12
- 125000006413 ring segment Chemical group 0.000 description 12
- 125000004434 sulfur atom Chemical group 0.000 description 12
- 238000001959 radiotherapy Methods 0.000 description 11
- 239000000203 mixture Substances 0.000 description 10
- 125000005843 halogen group Chemical group 0.000 description 9
- 238000006243 chemical reaction Methods 0.000 description 8
- 125000004430 oxygen atom Chemical group O* 0.000 description 8
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 7
- 239000002585 base Substances 0.000 description 7
- 238000002512 chemotherapy Methods 0.000 description 7
- 239000012091 fetal bovine serum Substances 0.000 description 7
- SDUQYLNIPVEERB-QPPQHZFASA-N gemcitabine Chemical compound O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 SDUQYLNIPVEERB-QPPQHZFASA-N 0.000 description 7
- 229960005277 gemcitabine Drugs 0.000 description 7
- 230000002401 inhibitory effect Effects 0.000 description 7
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 7
- SCVJRXQHFJXZFZ-KVQBGUIXSA-N 2-amino-9-[(2r,4s,5r)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-3h-purine-6-thione Chemical compound C1=2NC(N)=NC(=S)C=2N=CN1[C@H]1C[C@H](O)[C@@H](CO)O1 SCVJRXQHFJXZFZ-KVQBGUIXSA-N 0.000 description 6
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 6
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 description 5
- 125000006569 (C5-C6) heterocyclic group Chemical group 0.000 description 5
- 239000002253 acid Substances 0.000 description 5
- 239000012043 crude product Substances 0.000 description 5
- 125000004122 cyclic group Chemical group 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 229910052698 phosphorus Inorganic materials 0.000 description 5
- 238000012360 testing method Methods 0.000 description 5
- 210000004881 tumor cell Anatomy 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 230000005778 DNA damage Effects 0.000 description 4
- 231100000277 DNA damage Toxicity 0.000 description 4
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 4
- 125000005605 benzo group Chemical group 0.000 description 4
- 230000015572 biosynthetic process Effects 0.000 description 4
- 229910002091 carbon monoxide Inorganic materials 0.000 description 4
- 239000003085 diluting agent Substances 0.000 description 4
- 239000001963 growth medium Substances 0.000 description 4
- 238000001727 in vivo Methods 0.000 description 4
- 230000003287 optical effect Effects 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 230000008439 repair process Effects 0.000 description 4
- 230000019491 signal transduction Effects 0.000 description 4
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- 208000009458 Carcinoma in Situ Diseases 0.000 description 3
- 102000002427 Cyclin B Human genes 0.000 description 3
- 108010068150 Cyclin B Proteins 0.000 description 3
- 102100032857 Cyclin-dependent kinase 1 Human genes 0.000 description 3
- 101710106279 Cyclin-dependent kinase 1 Proteins 0.000 description 3
- 230000005971 DNA damage repair Effects 0.000 description 3
- 230000004668 G2/M phase Effects 0.000 description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 3
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 3
- 230000022131 cell cycle Effects 0.000 description 3
- 230000004663 cell proliferation Effects 0.000 description 3
- 238000012054 celltiter-glo Methods 0.000 description 3
- 230000006378 damage Effects 0.000 description 3
- 125000000524 functional group Chemical group 0.000 description 3
- 201000004933 in situ carcinoma Diseases 0.000 description 3
- 238000000338 in vitro Methods 0.000 description 3
- 239000002609 medium Substances 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 230000001225 therapeutic effect Effects 0.000 description 3
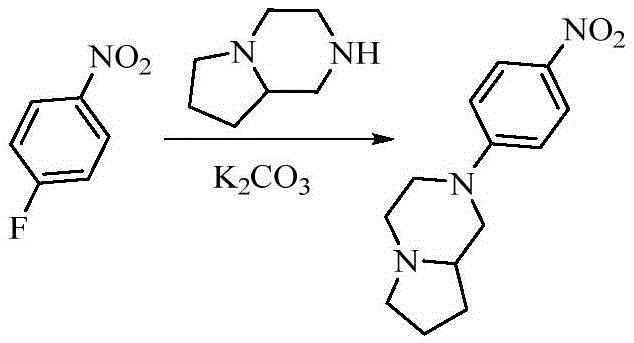
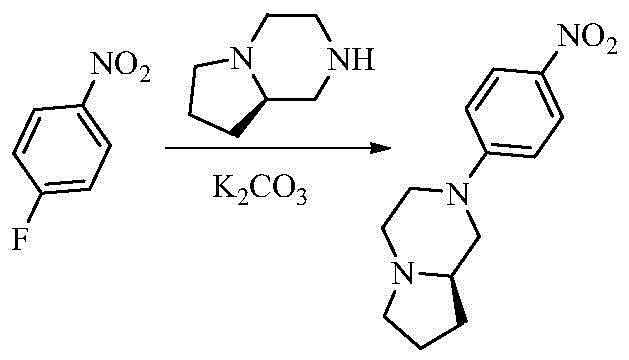
- FTTATHOUSOIFOQ-SSDOTTSWSA-N (8ar)-1,2,3,4,6,7,8,8a-octahydropyrrolo[1,2-a]pyrazine Chemical compound C1NCCN2CCC[C@@H]21 FTTATHOUSOIFOQ-SSDOTTSWSA-N 0.000 description 2
- AZJZYOFVHUGUDB-CYBMUJFWSA-N (8ar)-2-(4-nitrophenyl)-3,4,6,7,8,8a-hexahydro-1h-pyrrolo[1,2-a]pyrazine Chemical compound C1=CC([N+](=O)[O-])=CC=C1N1C[C@H]2CCCN2CC1 AZJZYOFVHUGUDB-CYBMUJFWSA-N 0.000 description 2
- 125000005940 1,4-dioxanyl group Chemical group 0.000 description 2
- WFQDTOYDVUWQMS-UHFFFAOYSA-N 1-fluoro-4-nitrobenzene Chemical compound [O-][N+](=O)C1=CC=C(F)C=C1 WFQDTOYDVUWQMS-UHFFFAOYSA-N 0.000 description 2
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
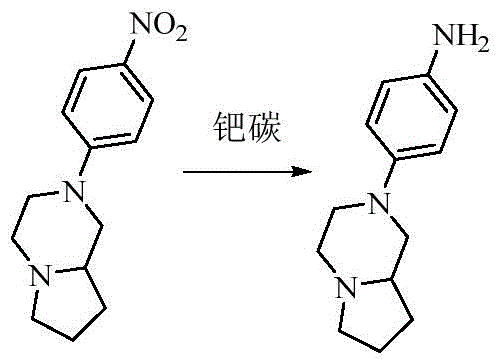
- AZJZYOFVHUGUDB-UHFFFAOYSA-N 2-(4-nitrophenyl)-3,4,6,7,8,8a-hexahydro-1h-pyrrolo[1,2-a]pyrazine Chemical compound C1=CC([N+](=O)[O-])=CC=C1N1CC2CCCN2CC1 AZJZYOFVHUGUDB-UHFFFAOYSA-N 0.000 description 2
- RWTJVGLEXNIAME-UHFFFAOYSA-N 4-(3,4,6,7,8,8a-hexahydro-1h-pyrrolo[1,2-a]pyrazin-2-yl)aniline Chemical compound C1=CC(N)=CC=C1N1CC2CCCN2CC1 RWTJVGLEXNIAME-UHFFFAOYSA-N 0.000 description 2
- RWTJVGLEXNIAME-CYBMUJFWSA-N 4-[(8ar)-3,4,6,7,8,8a-hexahydro-1h-pyrrolo[1,2-a]pyrazin-2-yl]aniline Chemical compound C1=CC(N)=CC=C1N1C[C@H]2CCCN2CC1 RWTJVGLEXNIAME-CYBMUJFWSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- 102100025064 Cellular tumor antigen p53 Human genes 0.000 description 2
- 101000777293 Homo sapiens Serine/threonine-protein kinase Chk1 Proteins 0.000 description 2
- KDLHZDBZIXYQEI-UHFFFAOYSA-N Palladium Chemical compound [Pd] KDLHZDBZIXYQEI-UHFFFAOYSA-N 0.000 description 2
- 102000001253 Protein Kinase Human genes 0.000 description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 2
- 102100031081 Serine/threonine-protein kinase Chk1 Human genes 0.000 description 2
- 102000004142 Trypsin Human genes 0.000 description 2
- 108090000631 Trypsin Proteins 0.000 description 2
- ZEEBGORNQSEQBE-UHFFFAOYSA-N [2-(3-phenylphenoxy)-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound C1(=CC(=CC=C1)OC1=NC(=CC(=C1)CN)C(F)(F)F)C1=CC=CC=C1 ZEEBGORNQSEQBE-UHFFFAOYSA-N 0.000 description 2
- ABRVLXLNVJHDRQ-UHFFFAOYSA-N [2-pyridin-3-yl-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound FC(C1=CC(=CC(=N1)C=1C=NC=CC=1)CN)(F)F ABRVLXLNVJHDRQ-UHFFFAOYSA-N 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 150000001336 alkenes Chemical class 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 230000009286 beneficial effect Effects 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 125000000596 cyclohexenyl group Chemical group C1(=CCCCC1)* 0.000 description 2
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 2
- 125000002433 cyclopentenyl group Chemical group C1(=CCCC1)* 0.000 description 2
- 230000001085 cytostatic effect Effects 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 238000001640 fractional crystallisation Methods 0.000 description 2
- 239000012458 free base Substances 0.000 description 2
- 239000008187 granular material Substances 0.000 description 2
- 208000037819 metastatic cancer Diseases 0.000 description 2
- 208000011575 metastatic malignant neoplasm Diseases 0.000 description 2
- OKKJLVBELUTLKV-VMNATFBRSA-N methanol-d1 Chemical compound [2H]OC OKKJLVBELUTLKV-VMNATFBRSA-N 0.000 description 2
- 230000000394 mitotic effect Effects 0.000 description 2
- 230000035772 mutation Effects 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 239000012074 organic phase Substances 0.000 description 2
- 125000000160 oxazolidinyl group Chemical group 0.000 description 2
- 230000037361 pathway Effects 0.000 description 2
- 239000002953 phosphate buffered saline Substances 0.000 description 2
- 108060006633 protein kinase Proteins 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N pyridine Substances C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 2
- 230000008929 regeneration Effects 0.000 description 2
- 238000011069 regeneration method Methods 0.000 description 2
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 2
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 description 2
- 239000000829 suppository Substances 0.000 description 2
- 239000003826 tablet Substances 0.000 description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 2
- 239000012588 trypsin Substances 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- VEEGZPWAAPPXRB-BJMVGYQFSA-N (3e)-3-(1h-imidazol-5-ylmethylidene)-1h-indol-2-one Chemical compound O=C1NC2=CC=CC=C2\C1=C/C1=CN=CN1 VEEGZPWAAPPXRB-BJMVGYQFSA-N 0.000 description 1
- FTTATHOUSOIFOQ-ZETCQYMHSA-N (8as)-1,2,3,4,6,7,8,8a-octahydropyrrolo[1,2-a]pyrazine Chemical compound C1NCCN2CCC[C@H]21 FTTATHOUSOIFOQ-ZETCQYMHSA-N 0.000 description 1
- FTTATHOUSOIFOQ-UHFFFAOYSA-N 1,2,3,4,6,7,8,8a-octahydropyrrolo[1,2-a]pyrazine Chemical compound C1NCCN2CCCC21 FTTATHOUSOIFOQ-UHFFFAOYSA-N 0.000 description 1
- 125000004502 1,2,3-oxadiazolyl group Chemical group 0.000 description 1
- 125000004529 1,2,3-triazinyl group Chemical group N1=NN=C(C=C1)* 0.000 description 1
- 125000001399 1,2,3-triazolyl group Chemical group N1N=NC(=C1)* 0.000 description 1
- BBVIDBNAYOIXOE-UHFFFAOYSA-N 1,2,4-oxadiazole Chemical compound C=1N=CON=1 BBVIDBNAYOIXOE-UHFFFAOYSA-N 0.000 description 1
- 125000001376 1,2,4-triazolyl group Chemical group N1N=C(N=C1)* 0.000 description 1
- 125000004506 1,2,5-oxadiazolyl group Chemical group 0.000 description 1
- 125000005918 1,2-dimethylbutyl group Chemical group 0.000 description 1
- 125000001781 1,3,4-oxadiazolyl group Chemical group 0.000 description 1
- 125000003363 1,3,5-triazinyl group Chemical group N1=C(N=CN=C1)* 0.000 description 1
- IGERFAHWSHDDHX-UHFFFAOYSA-N 1,3-dioxanyl Chemical group [CH]1OCCCO1 IGERFAHWSHDDHX-UHFFFAOYSA-N 0.000 description 1
- JPRPJUMQRZTTED-UHFFFAOYSA-N 1,3-dioxolanyl Chemical group [CH]1OCCO1 JPRPJUMQRZTTED-UHFFFAOYSA-N 0.000 description 1
- 125000000196 1,4-pentadienyl group Chemical group [H]C([*])=C([H])C([H])([H])C([H])=C([H])[H] 0.000 description 1
- BKWJAKQVGHWELA-UHFFFAOYSA-N 1-[6-(2-hydroxypropan-2-yl)-2-pyridinyl]-6-[4-(4-methyl-1-piperazinyl)anilino]-2-prop-2-enyl-3-pyrazolo[3,4-d]pyrimidinone Chemical compound C1CN(C)CCN1C(C=C1)=CC=C1NC1=NC=C2C(=O)N(CC=C)N(C=3N=C(C=CC=3)C(C)(C)O)C2=N1 BKWJAKQVGHWELA-UHFFFAOYSA-N 0.000 description 1
- TVOBVNNUECNRHY-UHFFFAOYSA-N 1-[6-(2-hydroxypropan-2-yl)pyridin-2-yl]-6-(4-morpholin-4-ylanilino)-2-prop-2-enylpyrazolo[3,4-d]pyrimidin-3-one Chemical compound CC(C)(O)c1cccc(n1)-n1n(CC=C)c(=O)c2cnc(Nc3ccc(cc3)N3CCOCC3)nc12 TVOBVNNUECNRHY-UHFFFAOYSA-N 0.000 description 1
- 125000004973 1-butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000006039 1-hexenyl group Chemical group 0.000 description 1
- 125000006023 1-pentenyl group Chemical group 0.000 description 1
- 125000006017 1-propenyl group Chemical group 0.000 description 1
- WOHLSTOWRAOMSG-UHFFFAOYSA-N 2,3-dihydro-1,3-benzothiazole Chemical compound C1=CC=C2SCNC2=C1 WOHLSTOWRAOMSG-UHFFFAOYSA-N 0.000 description 1
- 125000004974 2-butenyl group Chemical group C(C=CC)* 0.000 description 1
- 125000000069 2-butynyl group Chemical group [H]C([H])([H])C#CC([H])([H])* 0.000 description 1
- 125000006176 2-ethylbutyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(C([H])([H])*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006040 2-hexenyl group Chemical group 0.000 description 1
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 1
- 125000005916 2-methylpentyl group Chemical group 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000000850 2H-chromenyl group Chemical group O1C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000000844 2H-pyran-2-onyl group Chemical group O1C(C(=CC=C1)*)=O 0.000 description 1
- 125000001698 2H-pyranyl group Chemical group O1C(C=CC=C1)* 0.000 description 1
- QTFBPMJATBTHSY-UHFFFAOYSA-N 2h-furo[3,2-b]pyrrole Chemical compound C1=NC2=CCOC2=C1 QTFBPMJATBTHSY-UHFFFAOYSA-N 0.000 description 1
- NHQDETIJWKXCTC-UHFFFAOYSA-N 3-chloroperbenzoic acid Chemical compound OOC(=O)C1=CC=CC(Cl)=C1 NHQDETIJWKXCTC-UHFFFAOYSA-N 0.000 description 1
- 125000006041 3-hexenyl group Chemical group 0.000 description 1
- 125000003542 3-methylbutan-2-yl group Chemical group [H]C([H])([H])C([H])(*)C([H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 125000005917 3-methylpentyl group Chemical group 0.000 description 1
- HETSDWRDICBRSQ-UHFFFAOYSA-N 3h-quinolin-4-one Chemical compound C1=CC=C2C(=O)CC=NC2=C1 HETSDWRDICBRSQ-UHFFFAOYSA-N 0.000 description 1
- 125000001819 4H-chromenyl group Chemical group O1C(=CCC2=CC=CC=C12)* 0.000 description 1
- RDHOEMPRFDWIHL-UHFFFAOYSA-N 5h-thieno[3,2-c]pyrazole Chemical compound N1=NC2=CCSC2=C1 RDHOEMPRFDWIHL-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 229940122815 Aromatase inhibitor Drugs 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical group [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- 108010024986 Cyclin-Dependent Kinase 2 Proteins 0.000 description 1
- 102100036239 Cyclin-dependent kinase 2 Human genes 0.000 description 1
- 230000033616 DNA repair Effects 0.000 description 1
- 230000004543 DNA replication Effects 0.000 description 1
- 239000006145 Eagle's minimal essential medium Substances 0.000 description 1
- 102100030011 Endoribonuclease Human genes 0.000 description 1
- 101710199605 Endoribonuclease Proteins 0.000 description 1
- 230000008051 G1/S transition checkpoint Effects 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101000624643 Homo sapiens M-phase inducer phosphatase 3 Proteins 0.000 description 1
- 101000580039 Homo sapiens Ras-specific guanine nucleotide-releasing factor 1 Proteins 0.000 description 1
- 102100023330 M-phase inducer phosphatase 3 Human genes 0.000 description 1
- 206010027476 Metastases Diseases 0.000 description 1
- NIPNSKYNPDTRPC-UHFFFAOYSA-N N-[2-oxo-2-(2,4,6,7-tetrahydrotriazolo[4,5-c]pyridin-5-yl)ethyl]-2-[[3-(trifluoromethoxy)phenyl]methylamino]pyrimidine-5-carboxamide Chemical compound O=C(CNC(=O)C=1C=NC(=NC=1)NCC1=CC(=CC=C1)OC(F)(F)F)N1CC2=C(CC1)NN=N2 NIPNSKYNPDTRPC-UHFFFAOYSA-N 0.000 description 1
- 230000018199 S phase Effects 0.000 description 1
- 241000235347 Schizosaccharomyces pombe Species 0.000 description 1
- 101710113029 Serine/threonine-protein kinase Proteins 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- 125000000641 acridinyl group Chemical group C1(=CC=CC2=NC3=CC=CC=C3C=C12)* 0.000 description 1
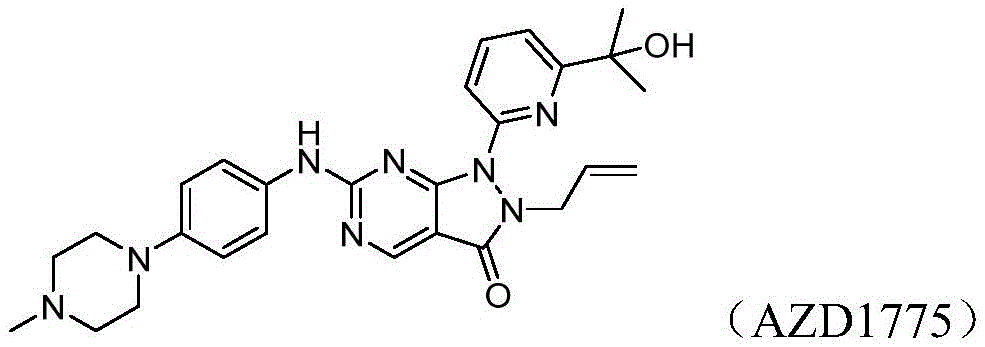
- 229950009557 adavosertib Drugs 0.000 description 1
- 239000000443 aerosol Substances 0.000 description 1
- 125000003158 alcohol group Chemical group 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 229910052784 alkaline earth metal Inorganic materials 0.000 description 1
- 150000001342 alkaline earth metals Chemical class 0.000 description 1
- 125000004414 alkyl thio group Chemical group 0.000 description 1
- 150000001408 amides Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 125000003277 amino group Chemical group 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 230000003698 anagen phase Effects 0.000 description 1
- 150000001449 anionic compounds Chemical class 0.000 description 1
- 230000006907 apoptotic process Effects 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- 125000003725 azepanyl group Chemical group 0.000 description 1
- 125000004069 aziridinyl group Chemical group 0.000 description 1
- 125000003785 benzimidazolyl group Chemical group N1=C(NC2=C1C=CC=C2)* 0.000 description 1
- 125000004618 benzofuryl group Chemical group O1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000004196 benzothienyl group Chemical group S1C(=CC2=C1C=CC=C2)* 0.000 description 1
- 125000003354 benzotriazolyl group Chemical group N1N=NC2=C1C=CC=C2* 0.000 description 1
- 125000004541 benzoxazolyl group Chemical group O1C(=NC2=C1C=CC=C2)* 0.000 description 1
- 239000003560 cancer drug Substances 0.000 description 1
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 150000001767 cationic compounds Chemical class 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 230000036755 cellular response Effects 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 125000001309 chloro group Chemical group Cl* 0.000 description 1
- 238000013375 chromatographic separation Methods 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- OTAFHZMPRISVEM-UHFFFAOYSA-N chromone Chemical compound C1=CC=C2C(=O)C=COC2=C1 OTAFHZMPRISVEM-UHFFFAOYSA-N 0.000 description 1
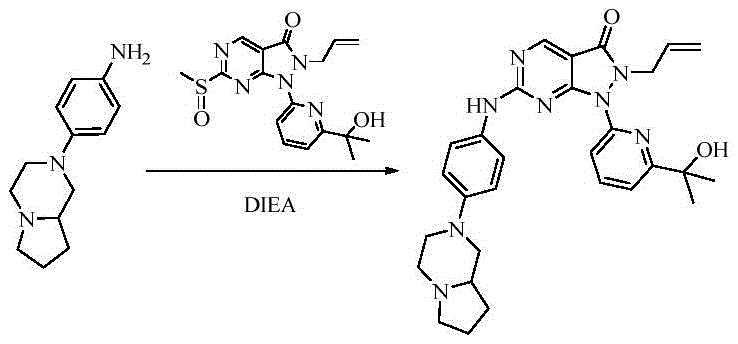
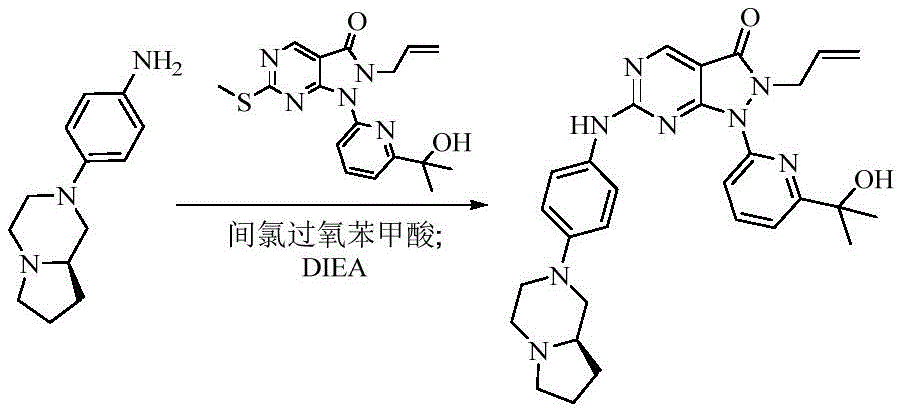
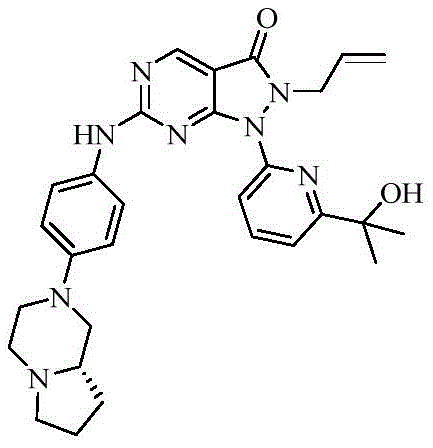
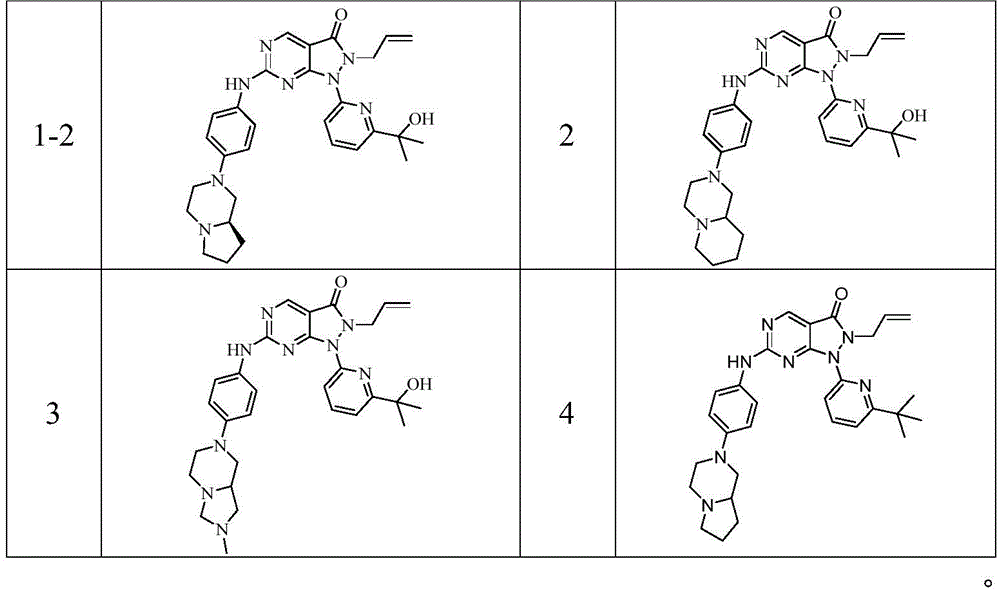
- 229940125904 compound 1 Drugs 0.000 description 1
- 239000012050 conventional carrier Substances 0.000 description 1
- ZYGHJZDHTFUPRJ-UHFFFAOYSA-N coumarin Chemical compound C1=CC=C2OC(=O)C=CC2=C1 ZYGHJZDHTFUPRJ-UHFFFAOYSA-N 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 125000001047 cyclobutenyl group Chemical group C1(=CCC1)* 0.000 description 1
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- CHVJITGCYZJHLR-UHFFFAOYSA-N cyclohepta-1,3,5-triene Chemical compound C1C=CC=CC=C1 CHVJITGCYZJHLR-UHFFFAOYSA-N 0.000 description 1
- 125000001162 cycloheptenyl group Chemical group C1(=CCCCCC1)* 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- UVJHQYIOXKWHFD-UHFFFAOYSA-N cyclohexa-1,4-diene Chemical compound C1C=CCC=C1 UVJHQYIOXKWHFD-UHFFFAOYSA-N 0.000 description 1
- 125000000058 cyclopentadienyl group Chemical group C1(=CC=CC1)* 0.000 description 1
- 125000000298 cyclopropenyl group Chemical group [H]C1=C([H])C1([H])* 0.000 description 1
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 231100000517 death Toxicity 0.000 description 1
- 125000004856 decahydroquinolinyl group Chemical group N1(CCCC2CCCCC12)* 0.000 description 1
- 125000004855 decalinyl group Chemical group C1(CCCC2CCCCC12)* 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- DIXBSCZRIZDQGC-UHFFFAOYSA-N diaziridine Chemical group C1NN1 DIXBSCZRIZDQGC-UHFFFAOYSA-N 0.000 description 1
- GLUUGHFHXGJENI-UHFFFAOYSA-N diethylenediamine Natural products C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 125000005054 dihydropyrrolyl group Chemical group [H]C1=C([H])C([H])([H])C([H])([H])N1* 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 238000006073 displacement reaction Methods 0.000 description 1
- 231100000673 dose–response relationship Toxicity 0.000 description 1
- 230000005782 double-strand break Effects 0.000 description 1
- 238000001647 drug administration Methods 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- ZSWFCLXCOIISFI-UHFFFAOYSA-N endo-cyclopentadiene Natural products C1C=CC=C1 ZSWFCLXCOIISFI-UHFFFAOYSA-N 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 description 1
- 230000003203 everyday effect Effects 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000012467 final product Substances 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- OKKDEIYWILRZIA-OSZBKLCCSA-N gemcitabine hydrochloride Chemical compound [H+].[Cl-].O=C1N=C(N)C=CN1[C@H]1C(F)(F)[C@H](O)[C@@H](CO)O1 OKKDEIYWILRZIA-OSZBKLCCSA-N 0.000 description 1
- 239000006481 glucose medium Substances 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 230000012010 growth Effects 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002632 imidazolidinyl group Chemical group 0.000 description 1
- 230000036039 immunity Effects 0.000 description 1
- 239000000367 immunologic factor Substances 0.000 description 1
- 125000003453 indazolyl group Chemical group N1N=C(C2=C1C=CC=C2)* 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 238000009776 industrial production Methods 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 229910052740 iodine Inorganic materials 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000004491 isohexyl group Chemical group C(CCC(C)C)* 0.000 description 1
- 125000000904 isoindolyl group Chemical group C=1(NC=C2C=CC=CC12)* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- VDBNYAPERZTOOF-UHFFFAOYSA-N isoquinolin-1(2H)-one Chemical compound C1=CC=C2C(=O)NC=CC2=C1 VDBNYAPERZTOOF-UHFFFAOYSA-N 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 150000002576 ketones Chemical class 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229940040145 liniment Drugs 0.000 description 1
- 239000000865 liniment Substances 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 238000004020 luminiscence type Methods 0.000 description 1
- 201000005243 lung squamous cell carcinoma Diseases 0.000 description 1
- 230000003211 malignant effect Effects 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000009401 metastasis Effects 0.000 description 1
- UKVIEHSSVKSQBA-UHFFFAOYSA-N methane;palladium Chemical compound C.[Pd] UKVIEHSSVKSQBA-UHFFFAOYSA-N 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 239000007758 minimum essential medium Substances 0.000 description 1
- 239000003607 modifier Substances 0.000 description 1
- SYSQUGFVNFXIIT-UHFFFAOYSA-N n-[4-(1,3-benzoxazol-2-yl)phenyl]-4-nitrobenzenesulfonamide Chemical class C1=CC([N+](=O)[O-])=CC=C1S(=O)(=O)NC1=CC=C(C=2OC3=CC=CC=C3N=2)C=C1 SYSQUGFVNFXIIT-UHFFFAOYSA-N 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004593 naphthyridinyl group Chemical group N1=C(C=CC2=CC=CN=C12)* 0.000 description 1
- 125000001971 neopentyl group Chemical group [H]C([*])([H])C(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000012148 non-surgical treatment Methods 0.000 description 1
- 238000011275 oncology therapy Methods 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 150000002891 organic anions Chemical class 0.000 description 1
- 150000002892 organic cations Chemical class 0.000 description 1
- 229940127084 other anti-cancer agent Drugs 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 125000003538 pentan-3-yl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000004934 phenanthridinyl group Chemical group C1(=CC=CC2=NC=C3C=CC=CC3=C12)* 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 230000000865 phosphorylative effect Effects 0.000 description 1
- 125000004592 phthalazinyl group Chemical group C1(=NN=CC2=CC=CC=C12)* 0.000 description 1
- 125000003386 piperidinyl group Chemical group 0.000 description 1
- 125000004928 piperidonyl group Chemical group 0.000 description 1
- 239000011505 plaster Substances 0.000 description 1
- 229940098458 powder spray Drugs 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000035755 proliferation Effects 0.000 description 1
- 125000000561 purinyl group Chemical group N1=C(N=C2N=CNC2=C1)* 0.000 description 1
- 125000003072 pyrazolidinyl group Chemical group 0.000 description 1
- 125000000719 pyrrolidinyl group Chemical group 0.000 description 1
- RQGPLDBZHMVWCH-UHFFFAOYSA-N pyrrolo[3,2-b]pyrrole Chemical compound C1=NC2=CC=NC2=C1 RQGPLDBZHMVWCH-UHFFFAOYSA-N 0.000 description 1
- GZTPJDLYPMPRDF-UHFFFAOYSA-N pyrrolo[3,2-c]pyrazole Chemical compound N1=NC2=CC=NC2=C1 GZTPJDLYPMPRDF-UHFFFAOYSA-N 0.000 description 1
- 125000002294 quinazolinyl group Chemical group N1=C(N=CC2=CC=CC=C12)* 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 125000001567 quinoxalinyl group Chemical group N1=C(C=NC2=CC=CC=C12)* 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 229940075993 receptor modulator Drugs 0.000 description 1
- 230000001105 regulatory effect Effects 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000002271 resection Methods 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 230000035945 sensitivity Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 230000001568 sexual effect Effects 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 229940054269 sodium pyruvate Drugs 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 239000000126 substance Substances 0.000 description 1
- 230000004083 survival effect Effects 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 125000004853 tetrahydropyridinyl group Chemical group N1(CCCC=C1)* 0.000 description 1
- 125000005942 tetrahydropyridyl group Chemical group 0.000 description 1
- 125000005958 tetrahydrothienyl group Chemical group 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000876 trifluoromethoxy group Chemical group FC(F)(F)O* 0.000 description 1
- 230000001960 triggered effect Effects 0.000 description 1
- 210000005253 yeast cell Anatomy 0.000 description 1
Landscapes
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明属于医药技术领域,具体涉及一类式(I)所示的二氢吡唑并嘧啶酮类Wee1激酶抑制剂、其药学上可接受的盐或其立体异构体,含有所述化合物、其药学上可接受的盐或其立体异构体的药物组合物及制剂,制备所述化合物、其药学上可接受的盐或其立体异构体的方法,以及所述化合物、其药学上可接受的盐或其立体异构体的用途, The present invention belongs to the technical field of medicine, and specifically relates to a class of dihydropyrazolopyrimidinone Weel kinase inhibitors represented by formula (I), pharmaceutically acceptable salts thereof or stereoisomers thereof, comprising the compound, Pharmaceutical compositions and preparations of pharmaceutically acceptable salts or stereoisomers thereof, methods for preparing said compounds, pharmaceutically acceptable salts or stereoisomers thereof, and said compounds, their pharmaceutically acceptable Use of accepted salts or stereoisomers thereof,
Description
技术领域technical field
本发明属于医药技术领域,具体涉及一类二氢吡唑并嘧啶酮类Wee1激酶抑制剂、其药学上可接受的盐、及其立体异构体,含有所述化合物、其药学上可接受的盐及其立体异构体的药物组合物及制剂,制备所述化合物、其药学上可接受的盐及其立体异构体的方法,以及所述化合物、其药学上可接受的盐及其立体异构体的用途。The invention belongs to the technical field of medicine, and in particular relates to a class of dihydropyrazolopyrimidinone Weel kinase inhibitors, pharmaceutically acceptable salts thereof, and stereoisomers thereof, containing the compound, its pharmaceutically acceptable Pharmaceutical compositions and preparations of salts and stereoisomers thereof, processes for preparing said compounds, pharmaceutically acceptable salts and stereoisomers thereof, and said compounds, pharmaceutically acceptable salts and stereoisomers thereof Use of isomers.
背景技术Background technique
癌症是全世界难治疗的恶性疾病,治疗难度大,死亡率高。2018年最新的全球肿瘤统计结果显示,目前为止,全球估计有1819万癌症新增病例以及960万癌症死亡病例。2019年1月,国家癌症中心发布了最新一期的全国癌症统计数据。每年全国恶性肿瘤发病约392.9万人,平均每天超过1万人被确诊为癌症,每分钟有7.5人被确诊为癌症。癌症已成为影响我国居民健康的主要疾病,癌症防治也面临着严峻的形式。Cancer is a malignant disease that is difficult to treat all over the world. It is difficult to treat and has a high mortality rate. According to the latest global cancer statistics in 2018, so far, there are an estimated 18.19 million new cancer cases and 9.6 million cancer deaths worldwide. In January 2019, the National Cancer Center released the latest national cancer statistics. About 3.929 million people are diagnosed with malignant tumors nationwide every year, and an average of more than 10,000 people are diagnosed with cancer every day, and 7.5 people are diagnosed with cancer every minute. Cancer has become a major disease affecting the health of Chinese residents, and cancer prevention and treatment is also facing a severe situation.
目前,放疗以及化疗是除手术切除外治疗癌症的最有效手段,同时放疗是对恶性肿瘤最有效的非手术治疗方法。放射线和相当多的抗癌药物均可导致DNA损伤。DNA损伤后,会引发受损DNA修复等一系列细胞反应,以提高肿瘤细胞的存活,这也是肿瘤细胞对放化疗抵抗的机制之一。损伤的DNA如果不及时和完整修复,肿瘤细胞则会因细胞凋亡或/和有丝分裂障碍导致细胞死亡。因此,只要抑制这些DNA损伤的修复,就可以提高癌细胞对放化疗的敏感性,抑制细胞增殖。At present, radiotherapy and chemotherapy are the most effective means of treating cancer besides surgical resection, and radiotherapy is the most effective non-surgical treatment for malignant tumors. Radiation and quite a few cancer drugs can cause DNA damage. After DNA damage, a series of cellular responses such as damaged DNA repair will be triggered to improve the survival of tumor cells, which is also one of the mechanisms of tumor cell resistance to radiotherapy and chemotherapy. If the damaged DNA is not repaired in a timely and complete manner, tumor cells will die due to apoptosis or/and mitotic disorders. Therefore, as long as the repair of these DNA damages is inhibited, the sensitivity of cancer cells to radiotherapy and chemotherapy can be improved and cell proliferation can be inhibited.
Wee1蛋白激酶是丝氨酸/苏氨酸蛋白激酶家族的一员,最早由Nurse等人在裂殖酵母细胞(S.pombe)中分离出来。在人体中,Wee1含有647个氨基酸,具有96kDa的分子量。DNA单链损伤修复的通路中,Wee1处于ATR信号通路的下游,在ATR信号通路激活之后,随之磷酸化CHK1,活化的CHK1会激活Wee1,同时抑制CDC25(解除CDK1/Cyclin B复合物的磷酸化,恢复其调控细胞周期的活性),进而磷酸化CDK1/Cyclin B,使CDK1/Cyclin B复合物进入非活跃状态,使细胞周期在G2/M期阻滞,为DNA损伤修复赢得时间;除此之外,Weel还可以通过磷酸化CDK2,阻滞细胞周期S期,调控DNA复制过程中出现的DNA双链断裂的修复。Wee1 protein kinase is a member of the serine/threonine protein kinase family, which was first isolated from fission yeast cells (S. pombe) by Nurse et al. In humans, Wee1 contains 647 amino acids and has a molecular weight of 96 kDa. In the pathway of DNA single-strand damage repair, Wee1 is in the downstream of the ATR signaling pathway. After the ATR signaling pathway is activated, it phosphorylates CHK1, and the activated CHK1 activates Wee1, and at the same time inhibits CDC25 (releasing the phosphorylation of the CDK1/Cyclin B complex. to restore its activity of regulating the cell cycle), and then phosphorylate CDK1/Cyclin B, so that the CDK1/Cyclin B complex enters an inactive state, arresting the cell cycle in the G2/M phase, and gaining time for DNA damage repair; In addition, Weel can also arrest the S phase of the cell cycle by phosphorylating CDK2, and regulate the repair of DNA double-strand breaks that occur during DNA replication.
在整个DDR(DNA damage repair)通路中,Wee1主要在G2/M期检查点发挥作用。对于p53突变的肿瘤细胞,由于本身存在G1/S检查点缺陷,因此更加依赖于G2/M期检查点来修复DNA损伤,从机制而言,Wee1抑制剂对p53突变的肿瘤细胞更加敏感。In the entire DDR (DNA damage repair) pathway, Wee1 mainly plays a role in the G2/M phase checkpoint. For tumor cells with p53 mutations, due to the G1/S checkpoint defect, they are more dependent on the G2/M phase checkpoint to repair DNA damage. Mechanistically, Wee1 inhibitors are more sensitive to tumor cells with p53 mutations.
综上所述,Wee1激酶抑制剂不仅可协同增强放疗和化疗效果,有效抑制肿瘤生长,还可降低对正常细胞的损伤和减少副作用。目前,该靶点药物的研究仍处于临床实验阶段,尚无药物上市。因此,开发一款高效的Wee1激酶抑制剂具有重要的临床意义,无论是单用还是联合其他药物应用均有广阔的市场前景。In summary, Wee1 kinase inhibitors can not only synergistically enhance the effect of radiotherapy and chemotherapy, effectively inhibit tumor growth, but also reduce damage to normal cells and reduce side effects. At present, the research on this target drug is still in the clinical trial stage, and no drug has been marketed yet. Therefore, the development of a highly effective Wee1 kinase inhibitor has important clinical significance, whether it is used alone or in combination with other drugs, it has broad market prospects.
发明内容Contents of the invention
本发明要解决的技术问题是提供一种结构新颖的、对Wee1有抑制活性的二氢吡唑并嘧啶酮类化合物。进一步的,该类化合物可用于抑制Wee1激酶活性,从而增强机体对肿瘤的免疫作用。更进一步的,该类化合物还可用于治疗由Wee1所介导的一种或多种疾病,尤其是癌症。该类化合物对多种癌细胞具有良好的抑制作用,在生物体内具有较高的暴露量和较好的体内药效。The technical problem to be solved by the present invention is to provide a dihydropyrazolopyrimidinone compound with novel structure and inhibitory activity on Wee1. Furthermore, this kind of compound can be used to inhibit the activity of Wee1 kinase, thereby enhancing the immunity of the body against tumors. Furthermore, the compound can also be used to treat one or more diseases mediated by Wee1, especially cancer. This type of compound has good inhibitory effect on various cancer cells, has higher exposure in vivo and better drug efficacy in vivo.
本发明的技术方案如下:Technical scheme of the present invention is as follows:
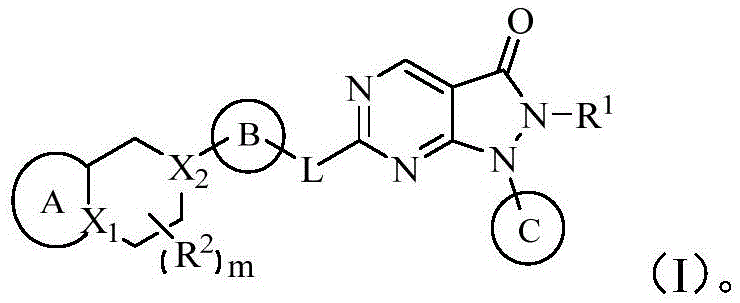
在一方面,本发明提供了如下通式(I)所示的化合物、其药学上可接受的盐或其立体异构体,In one aspect, the present invention provides a compound represented by the following general formula (I), a pharmaceutically acceptable salt thereof or a stereoisomer thereof,
其中,X1、X2分别独立地选自C、C(R3)或N;Wherein, X 1 and X 2 are independently selected from C, C(R 3 ) or N;
L选自-C(R4)(R5)-、-O-、-N(R6)-或-S-;L is selected from -C(R 4 )(R 5 )-, -O-, -N(R 6 )- or -S-;
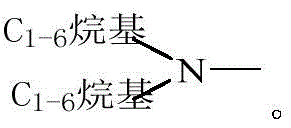
R1选自任选被取代基取代的C1-6烷基、C2-6烯基或C2-6炔基,所述取代基分别独立地选自卤素、羟基、氨基、羧基、氰基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷氧基或C1-6烷基羰基; R is selected from C 1-6 alkyl, C 2-6 alkenyl or C 2-6 alkynyl optionally substituted by substituents independently selected from halogen, hydroxyl, amino, carboxyl, cyano Base, C 1-6 alkoxy, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halogenated C 1-6 alkoxy or C 1-6 alkylcarbonyl;
每一R2分别独立地选自卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷基羰基;Each R is independently selected from halogen, hydroxyl, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di(C 1-6 alkyl)amino , halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1-6 alkylcarbonyl;
环A选自任选被1-3个Q1取代的5-7元单环环烷基、5-7元单环杂环基、6-8元单环芳基或5-8元单环杂芳基;Ring A is selected from 5-7 membered monocyclic cycloalkyl, 5-7 membered monocyclic heterocyclic group, 6-8 membered monocyclic aryl or 5-8 membered monocyclic heterocyclic group optionally substituted by 1-3 Q1 Aryl;
环B选自任选被1-3个Q2取代的5-7元单环环烷基、5-7元单环杂环基、6-8元单环芳基或5-8元单环杂芳基;Ring B is selected from 5-7 membered monocyclic cycloalkyl, 5-7 membered monocyclic heterocyclyl, 6-8 membered monocyclic aryl or 5-8 membered monocyclic heterocyclic optionally substituted by 1-3 Q2 Aryl;
环C选自任选被1-3个Q3取代的3-10元环烷基、3-10元杂环基、6-10元芳基或5-10元杂芳基;Ring C is selected from 3-10 membered cycloalkyl, 3-10 membered heterocyclyl, 6-10 membered aryl or 5-10 membered heteroaryl optionally substituted by 1-3 Q3;
每一Q1、每一Q2、每一Q3分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基;Each Q1, each Q2, and each Q3 are independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl , amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkane Oxygen, C 1-6 alkylthio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio, hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, amino C 1-6 alkylthio;
每一R3、R4、R5分别独立的选自氢、卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基、C1-6烷基羰基或C1-6烷氧基C1-6烷基;Each R 3 , R 4 , and R 5 are independently selected from hydrogen, halogen, hydroxyl, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di( C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy, C 1-6 alkyl Carbonyl or C 1-6 alkoxy C 1-6 alkyl;
R6分别独立的选自氢、C1-6烷基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基或C1-6烷氧基C1-6烷基;R 6 are independently selected from hydrogen, C 1-6 alkyl, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl or C 1-6 alkoxy C 1 -6 alkyl;
m选自0、1、2或3。m is selected from 0, 1, 2 or 3.
在上述某些实施方案中,X1、X2分别独立地选自C、CH或N;In some of the above embodiments, X 1 and X 2 are independently selected from C, CH or N;
L选自-CH2-、-O-、-NH或-S-;L is selected from -CH 2 -, -O-, -NH or -S-;
R1选自C1-6烷基、C2-6烯基或C2-6炔基;R 1 is selected from C 1-6 alkyl, C 2-6 alkenyl or C 2-6 alkynyl;
每一R2分别独立地选自卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷基羰基;Each R is independently selected from halogen, hydroxyl, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di(C 1-6 alkyl)amino , halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1-6 alkylcarbonyl;
环A选自任选被1-2个Q1取代的5-6元单环环烷基、5-6元单环杂环基、苯基或5-6元单环杂芳基;Ring A is selected from 5-6 membered monocyclic cycloalkyl, 5-6 membered monocyclic heterocyclyl, phenyl or 5-6 membered monocyclic heteroaryl optionally substituted by 1-2 Q1;
环B选自任选被1-2个Q2取代的苯基或5-6元单环杂芳基;Ring B is selected from phenyl or 5-6 membered monocyclic heteroaryl optionally substituted by 1-2 Q2;
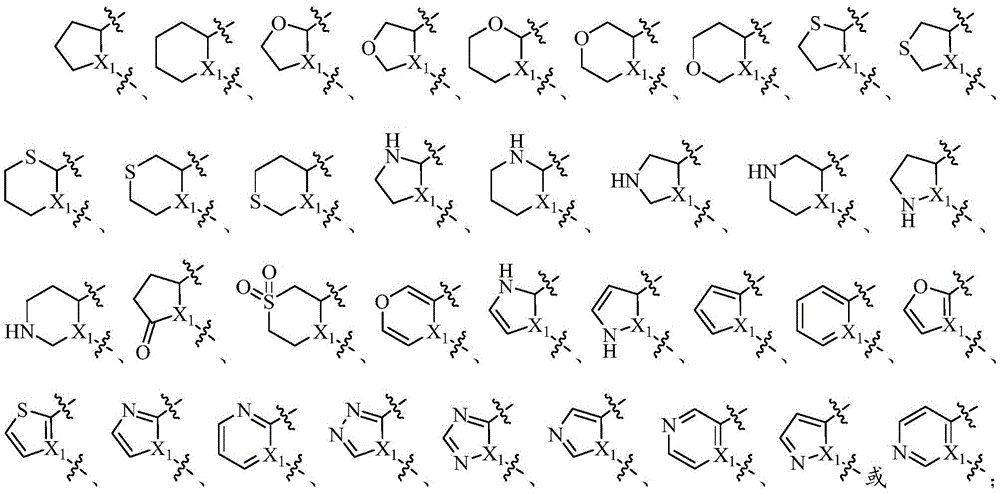
环C选自任选被1-2个Q3取代的5-7元单环环烷基、8-10元稠环环烷基、5-7元单环杂环基、8-10元稠环杂环基、苯基、萘基、5-7元单环杂芳基或8-10元稠杂芳基;Ring C is selected from 5-7 membered monocyclic cycloalkyl, 8-10 membered fused ring cycloalkyl, 5-7 membered monocyclic heterocyclyl, 8-10 membered fused ring optionally substituted by 1-2 Q3 Heterocyclyl, phenyl, naphthyl, 5-7 membered monocyclic heteroaryl or 8-10 membered condensed heteroaryl;
每一Q1、每一Q2、每一Q3分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基;Each Q1, each Q2, and each Q3 are independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl , amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkane Oxygen, C 1-6 alkylthio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio, hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, amino C 1-6 alkylthio;
m选自0、1或2。m is selected from 0, 1 or 2.
在某些实施方案中,X1、X2分别独立地选自C、CH或N;In some embodiments, X 1 and X 2 are independently selected from C, CH or N;
L选自-CH2-、-O-、-NH或-S-;L is selected from -CH 2 -, -O-, -NH or -S-;
R1选自C1-4烷基、C2-4烯基或C2-4炔基;R 1 is selected from C 1-4 alkyl, C 2-4 alkenyl or C 2-4 alkynyl;
每一R2分别独立地选自卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷基羰基;Each R is independently selected from halogen, hydroxyl, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di(C 1-6 alkyl)amino , halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1-6 alkylcarbonyl;
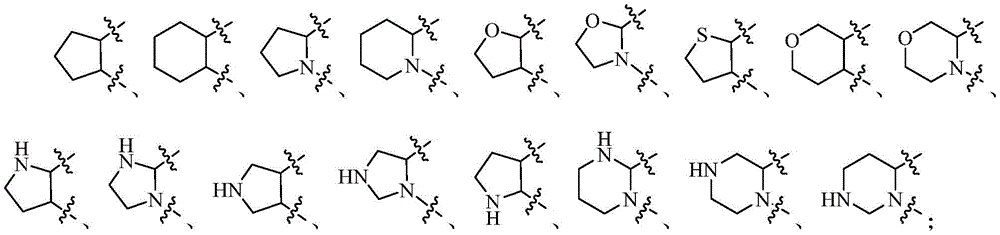
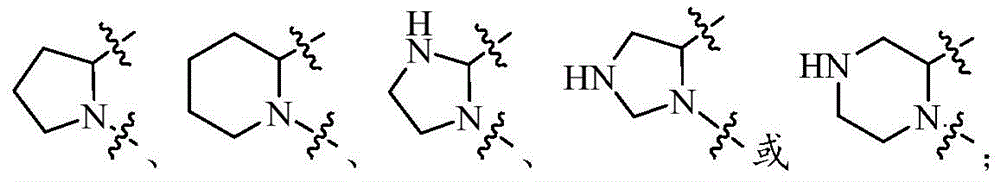
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环B选自任选被1-2个Q2取代的苯基或含1-3个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;Ring B is selected from phenyl optionally substituted by 1-2 Q2 or 5-6 membered monocyclic heteroaryl containing 1-3 heteroatoms or groups independently selected from N, NH, O, S, S(O) or S(O) 2 ;
环C选自任选被1-2个Q3取代的5-6元单环环烷基、8-10元稠环环烷基、5-6元单环杂环基、含1-3个杂原子或基团的8-10元稠环杂环基、苯基、萘基、5-6元单环杂芳基或含1-3个杂原子或基团的8-10元稠杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;Ring C is selected from 5-6 membered monocyclic cycloalkyl, 8-10 membered condensed ring cycloalkyl, 5-6 membered monocyclic heterocyclic group, containing 1-3 heterocyclic groups optionally substituted by 1-2 Q3 Atom or group 8-10 membered fused ring heterocyclic group, phenyl, naphthyl, 5-6 membered monocyclic heteroaryl group or 8-10 membered fused heteroaryl group containing 1-3 heteroatoms or groups , the heteroatoms or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ;
每一Q1、每一Q2、每一Q3分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基;Each Q1, each Q2, and each Q3 are independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl , amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkane Oxygen, C 1-6 alkylthio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio, hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, amino C 1-6 alkylthio;
m选自0、1或2。m is selected from 0, 1 or 2.
在某些实施方案中,X1、X2分别独立地选自CH或N;In certain embodiments, X 1 and X 2 are independently selected from CH or N;
L选自-CH2-、-O-、-NH或-S-;L is selected from -CH 2 -, -O-, -NH or -S-;
R1选自甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基或丙炔基; R is selected from methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl or propynyl;
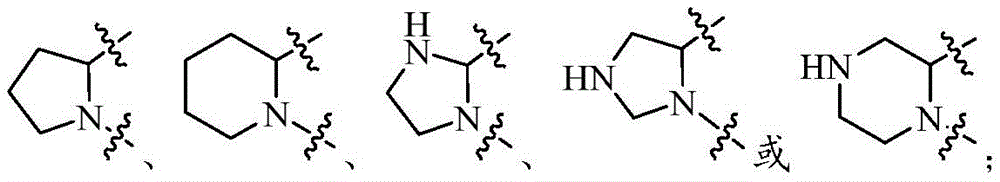
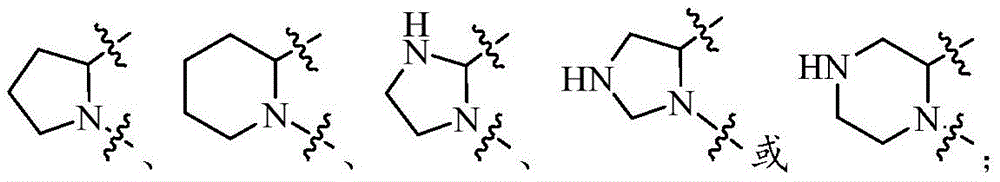
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环B选自任选被1-2个Q2取代的苯基、呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring B is selected from phenyl, furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1,2,4-Triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
环C选自任选被1-2个Q3取代的环戊基、环己基、苯基、含1-3个杂原子或基团的5-6元单环杂环基或含1-3个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;Ring C is selected from cyclopentyl, cyclohexyl, phenyl, 5-6 membered monocyclic heterocyclic groups containing 1-3 heteroatoms or groups optionally substituted by 1-2 Q3 or 1-3 5-6 membered monocyclic heteroaryls of heteroatoms or groups, said heteroatoms or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ;
每一Q1、每一Q2、每一Q3分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基;Each Q1, each Q2, and each Q3 are independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl , amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkane Oxygen, C 1-6 alkylthio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio, hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, amino C 1-6 alkylthio;
m选自0或1。m is selected from 0 or 1.
在某些实施方案中,X1、X2分别为N;L为NH;R1为丙烯基;In certain embodiments, X 1 and X 2 are each N; L is NH; R 1 is propenyl;
每一R2分别独立地选自卤素、羟基、氨基、C1-6烷基、C1-6烷氧基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基或卤代C1-6烷氧基;Each R is independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1 -6 alkyl or halogenated C 1-6 alkoxy;
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环B选自苯基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位;Ring B is selected from phenyl, pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl, preferably, L and X are in the para position of ring B;
环C选自任选被1-2个Q3取代的呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1,2 ,4-triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q1,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1 and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy , hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m选自0、1或2。m is selected from 0, 1 or 2.
在某些实施方案中,X1、X2分别为N;In certain embodiments, X 1 and X 2 are each N;
L为NH;L is NH;
R1为丙烯基;R 1 is propenyl;
R2选自卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷基羰基;R 2 is selected from halogen, hydroxyl, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1 -6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1-6 alkylcarbonyl;
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环B选自苯基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位;Ring B is selected from phenyl, pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl, preferably, L and X are in the para position of ring B;
环C选自任选被1-2个Q3取代的环戊基、环己基、苯基、四氢呋喃基、四氢噻吩基、四氢吡咯基、四氢咪唑基、四氢吡唑基、噁唑烷基、噻唑烷基、呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from cyclopentyl, cyclohexyl, phenyl, tetrahydrofuryl, tetrahydrothiophenyl, tetrahydropyrrolyl, tetrahydroimidazolyl, tetrahydropyrazolyl, oxazole optionally substituted by 1-2 Q3 Alkyl, thiazolidinyl, furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1,2,4-triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q1,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1 and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy , hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m选自0或1。m is selected from 0 or 1.
在某些实施方案中,X1、X2分别为N;In certain embodiments, X 1 and X 2 are each N;
L为NH;L is NH;
R1为丙烯基;R 1 is propenyl;
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环B选自苯基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位;Ring B is selected from phenyl, pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl, preferably, L and X are in the para position of ring B;
环C选自任选被1-2个Q3取代的苯基、吡咯基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from phenyl, pyrrolyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1,2,4- Triazole, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q1,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1 and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy , hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m选自0。m is selected from 0.
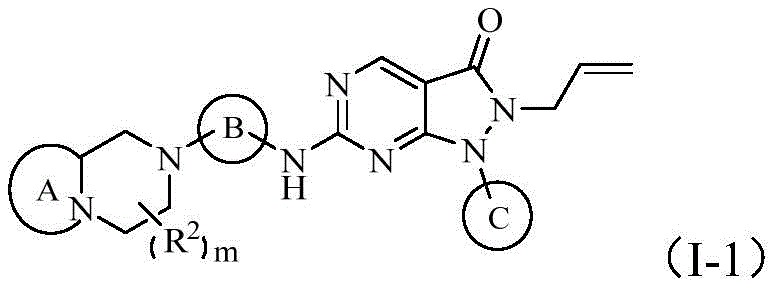
在某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(I-1)所示的结构:In some embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (I-1):
每一R2分别独立地选自卤素、羟基、氨基、C1-6烷基、C1-6烷氧基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基或卤代C1-6烷氧基;Each R is independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1 -6 alkyl or halogenated C 1-6 alkoxy;
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环B选自苯基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位;Ring B is selected from phenyl, pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl, preferably, L and X are in the para position of ring B;
环C选自任选被1-2个Q3取代的吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl optionally substituted by 1-2 Q3;
每一Q1,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1 and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy , halogenated C 1-6 alkoxy , hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m选自0、1或2。m is selected from 0, 1 or 2.
在上述某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(II)所示的结构:In some of the above embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (II):
其中,环A、环C、Q1、Q2、Q3、X1、X2、L、R1、R2、R3、R4、R5、R6、m的定义如上述任一方案所述;n选自0、1或2。Among them, ring A, ring C, Q1, Q2, Q3, X 1 , X 2 , L, R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , and m are defined as described in any of the above schemes ; n is selected from 0, 1 or 2.
在上述某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(III)所示的结构:In some of the above embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (III):
其中,环A、环C、Q1、Q2、Q3、X1、X2、L、R2、R3、R4、R5、R6、m的定义如上述任一方案所述;n选自0、1或2。Among them, ring A, ring C, Q1, Q2, Q3, X 1 , X 2 , L, R 2 , R 3 , R 4 , R 5 , R 6 , and m are defined as described in any of the above schemes; Since 0, 1 or 2.
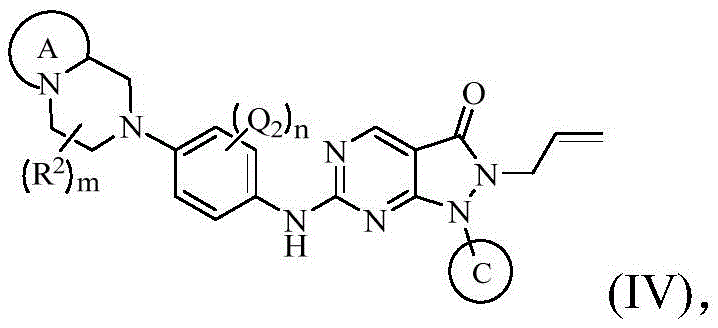
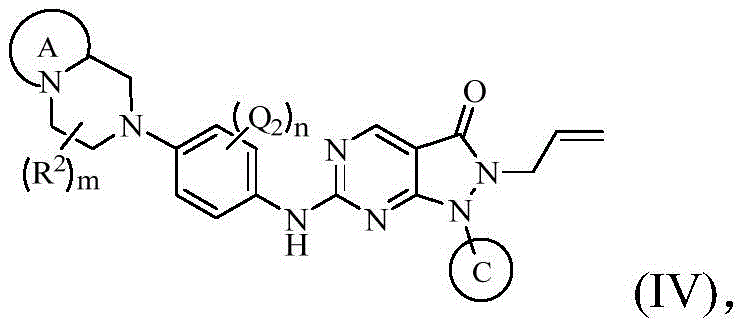
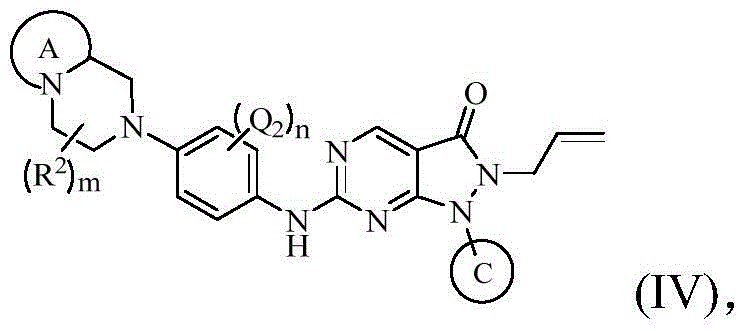
在上述某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(IV)所示的结构:In some of the above embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (IV):
其中,环A、环C、Q1、Q2、Q3、R2、m的定义如上述任一方案所述;n选自0、1或2。Wherein, ring A, ring C, Q1, Q2, Q3, R 2 and m are defined as described in any of the schemes above; n is selected from 0, 1 or 2.
在某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(IV)所示的结构:In certain embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (IV):
其中,in,
每一R2分别独立地选自卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷基羰基;Each R is independently selected from halogen, hydroxyl, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di(C 1-6 alkyl)amino , halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1-6 alkylcarbonyl;
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环C选自任选被1-2个Q3取代的吡咯基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from pyrrolyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1,2,4-triazole optionally substituted by 1-2 Q3 , pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q1,每一Q2,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1, each Q2, and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy , halogenated C 1- 6 alkoxy, hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m选自0或1;m is selected from 0 or 1;
n选自0或1。n is selected from 0 or 1.
在某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(IV)所示的结构:In certain embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (IV):
其中,in,
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环C选自任选被1-2个Q3取代的吡咯基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from pyrrolyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1,2,4-triazole optionally substituted by 1-2 Q3 , pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q1,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1 and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy , halogenated C 1-6 alkoxy , hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m为0;m is 0;
n为0。n is 0.
在某些实施方案中,本发明所述的化合物、其药学上可接受的盐或其立体异构体,具有如下通式(IV)所示的结构:In certain embodiments, the compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has the structure shown in the following general formula (IV):
其中,in,
环A选自任选被1-2个Q1取代的以下基团:Ring A is selected from the following groups optionally substituted by 1-2 Q1:
环C选自任选被1-2个Q3取代的吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;Ring C is selected from pyridyl, pyrimidyl, pyridazinyl, pyrazinyl or triazinyl optionally substituted by 1-2 Q3;
每一Q1,每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基;Each Q1 and each Q3 are independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy , halogenated C 1-6 alkoxy , hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy;
m为0;m is 0;
n为0。n is 0.
在上述某些实施方案中,R1选自C1-4烷基、C2-4烯基或C2-4炔基。在上述某些实施方案中,R1选自甲基、乙基、丙基、异丙基、乙烯基、丙烯基、乙炔基或丙炔基。在上述某些实施方案中,R1选自丙基、丙烯基或丙炔基。在上述某些实施方案中,R1选自丙烯基。In certain embodiments above, R 1 is selected from C 1-4 alkyl, C 2-4 alkenyl or C 2-4 alkynyl. In certain embodiments of the above, R is selected from methyl, ethyl, propyl, isopropyl, vinyl, propenyl, ethynyl or propynyl. In certain embodiments of the above, R is selected from propyl, propenyl or propynyl. In certain embodiments above, R 1 is selected from propenyl.
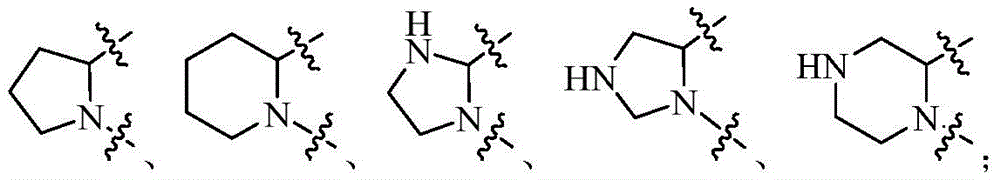
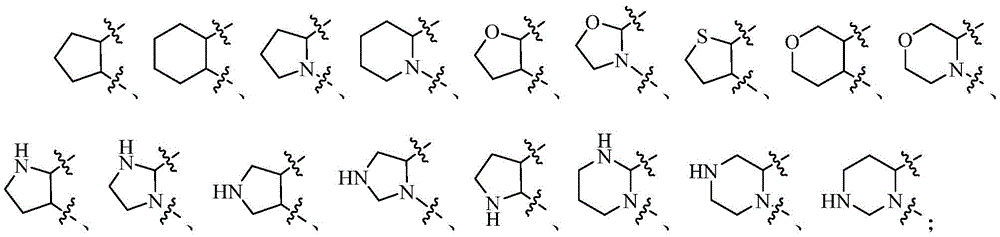
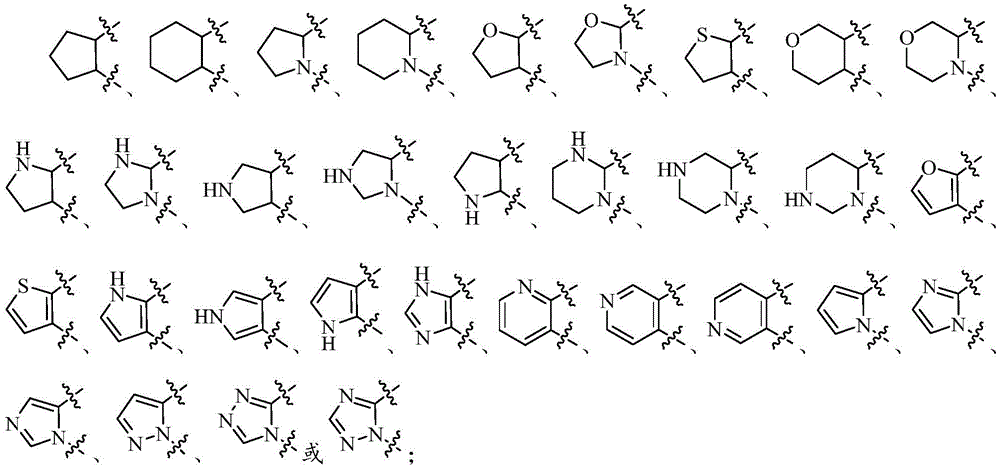
在上述某些实施方案中,环A选自任选被1-2个Q1取代的以下基团:In some of the above embodiments, ring A is selected from the following groups optionally substituted with 1-2 Q1:
X1选自C、-CH-或-N-; X1 is selected from C, -CH- or -N-;
每一Q1分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。Each Q1 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkane Thio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio , hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, Amino C 1-6 alkylthio.
在上述某些实施方案中,环A选自任选被1-2个Q1取代的以下基团:In some of the above embodiments, ring A is selected from the following groups optionally substituted with 1-2 Q1:
每一Q1分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。Each Q1 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkane Thio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio , hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, Amino C 1-6 alkylthio.
在上述某些实施方案中,环A选自任选被1-2个Q1取代的以下基团:In some of the above embodiments, ring A is selected from the following groups optionally substituted with 1-2 Q1:
每一Q1分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基。Each Q1 is independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxyl C 1-6 alkyl, amino C 1-6 alkyl , C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, hydroxyl C 1 -6 alkoxy or amino C 1-6 alkoxy.
在上述某些实施方案中,环A选自任选被1-2个Q1取代的以下基团:In some of the above embodiments, ring A is selected from the following groups optionally substituted with 1-2 Q1:
在上述某些实施方案中,环A选自任选被1-2个Q1取代的以下基团:In some of the above embodiments, ring A is selected from the following groups optionally substituted with 1-2 Q1:
在上述某些实施方案中,环A选自任选被1-2个Q1取代的以下基团: In some of the above embodiments, ring A is selected from the following groups optionally substituted with 1-2 Q1:
在上述某些实施方案中,每一Q1分别独立的选自卤素、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基或卤代C1-6烷氧基。In some of the above embodiments, each Q1 is independently selected from halogen, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 Alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy or halogenated C 1-6 alkoxy.
在上述某些实施方案中,每一Q1分别独立的选自C1-6烷基、卤代C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基或卤代C1-6烷氧基。In some of the above embodiments, each Q1 is independently selected from C 1-6 alkyl, halogenated C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 Alkoxy or halogenated C 1-6 alkoxy.
在上述某些实施方案中,每一Q1分别独立的选自甲基、乙基、丙基、异丙基、一氟甲基、二氟甲基、三氟甲基、1,1,1-三氟乙基、甲氧基甲基、甲氧基乙基,乙氧基甲基、甲氧基、乙氧基、丙氧基、异丙氧基、一氟甲氧基、二氟甲基或三氟甲氧基。In some of the above embodiments, each Q1 is independently selected from methyl, ethyl, propyl, isopropyl, monofluoromethyl, difluoromethyl, trifluoromethyl, 1,1,1- Trifluoroethyl, methoxymethyl, methoxyethyl, ethoxymethyl, methoxy, ethoxy, propoxy, isopropoxy, fluoromethoxy, difluoromethyl or trifluoromethoxy.
在上述某些实施方案中,环B选自任选被1-2个Q2取代的苯基或含1-3个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;In some of the above embodiments, ring B is selected from phenyl optionally substituted by 1-2 Q2 or 5-6 membered monocyclic heteroaryl containing 1-3 heteroatoms or groups, said heteroatoms Or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ;
每一Q2分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。Each Q2 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkane Thio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio , hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, Amino C 1-6 alkylthio.
在上述某些实施方案中,环B选自任选被1-2个Q2取代的苯基或含1-2个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;每一Q2分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。In some of the above embodiments, ring B is selected from phenyl optionally substituted by 1-2 Q2 or 5-6 membered monocyclic heteroaryl containing 1-2 heteroatoms or groups, said heteroatoms or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ; each Q2 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, di(C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy , C 1-6 alkylthio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio, Hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, amino C 1-6 alkylthio.
在上述某些实施方案中,环B选自苯基或含1-2个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2,优选地,L和X2处于环B的对位。In some of the above embodiments, ring B is selected from phenyl or 5-6 membered monocyclic heteroaryl containing 1-2 heteroatoms or groups independently selected from N, NH, O, S, S(O) or S(O) 2 , preferably, L and X 2 are in the para position of ring B.
在上述某些实施方案中,环B选自任选被1-2个Q2取代的苯基、呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;In certain embodiments above, ring B is selected from phenyl, furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1, 2,3-triazole, 1,2,4-triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q2分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。Each Q2 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkane Thio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio , hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, Amino C 1-6 alkylthio.
在上述某些实施方案中,环B选自苯基、呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位。In some of the above embodiments, ring B is selected from phenyl, furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1, 2,4-Triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl, preferably, L and X are in the para position of ring B.
在上述某些实施方案中,环B选自任选被1-2个Q2取代的苯基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位;In some of the above embodiments, ring B is selected from phenyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl optionally substituted by 1-2 Q2, preferably, L and X2 in the paraposition of ring B;
每一Q2分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基。Each Q2 is independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxyl C 1-6 alkyl, amino C 1-6 alkyl , C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, hydroxyl C 1 -6 alkoxy or amino C 1-6 alkoxy.
在上述某些实施方案中,环B选自苯基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基,优选地,L和X2处于环B的对位。In certain embodiments above, ring B is selected from phenyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl, preferably, L and X are at the para position of ring B.
在上述某些实施方案中,环B选自苯基。In certain embodiments of the above, ring B is selected from phenyl.
在上述某些实施方案中,环C选自任选被1-2个Q3取代的5-6元单环环烷基、8-10元稠环环烷基、5-6元单环杂环基、含1-3个杂原子或基团的8-10元稠环杂环基、苯基、萘基、5-6元单环杂芳基或含1-3个杂原子或基团的8-10元稠杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;In some of the above embodiments, Ring C is selected from 5-6 membered monocyclic cycloalkyl, 8-10 membered fused ring cycloalkyl, 5-6 membered monocyclic heterocycle optionally substituted by 1-2 Q3 group, 8-10 membered condensed ring heterocyclic group containing 1-3 heteroatoms or groups, phenyl, naphthyl, 5-6 membered monocyclic heteroaryl group or 1-3 heteroatoms or groups 8-10 membered condensed heteroaryl groups, the heteroatoms or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ;
优选地,环C选自任选被1-2个Q3取代的5-6元单环环烷基、5-6元单环杂环基、苯基或5-6元单环杂芳基;Preferably, ring C is selected from 5-6 membered monocyclic cycloalkyl, 5-6 membered monocyclic heterocyclyl, phenyl or 5-6 membered monocyclic heteroaryl optionally substituted by 1-2 Q3;
优选地,环C选自任选被1-2个Q3取代的环戊基、环己基、苯基、含1-3个杂原子或基团的5-6元单环杂环基或含1-3个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;Preferably, ring C is selected from cyclopentyl, cyclohexyl, phenyl, 5-6 membered monocyclic heterocyclyl containing 1-3 heteroatoms or groups optionally substituted by 1-2 Q3 or 1 -3 heteroatoms or groups of 5-6 membered monocyclic heteroaryls, said heteroatoms or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ;
优选地,环C选自任选被1-2个Q3取代的环戊基、环己基、苯基、含1-2个杂原子或基团的5-6元单环杂环基或含1-2个杂原子或基团的5-6元单环杂芳基,所述杂原子或基团分别独立地选自N、NH、O、S、S(O)或S(O)2;Preferably, ring C is selected from cyclopentyl, cyclohexyl, phenyl, 5-6 membered monocyclic heterocyclyl containing 1-2 heteroatoms or groups optionally substituted by 1-2 Q3 or containing 1 -2 heteroatoms or groups of 5-6 membered monocyclic heteroaryls, said heteroatoms or groups are independently selected from N, NH, O, S, S(O) or S(O) 2 ;
每一Q3分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。Each Q3 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkane Thio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio , hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, Amino C 1-6 alkylthio.
在上述某些实施方案中,环C选自任选被1-2个Q3取代的环戊基、环己基、苯基、四氢呋喃基、四氢噻吩基、四氢吡咯基、四氢咪唑基、四氢吡唑基、噁唑烷基、噻唑烷基、呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;In some of the above embodiments, ring C is selected from cyclopentyl, cyclohexyl, phenyl, tetrahydrofuranyl, tetrahydrothiophenyl, tetrahydropyrrolyl, tetrahydroimidazolyl, optionally substituted by 1-2 Q3, Tetrahydropyrazolyl, oxazolidinyl, thiazolidinyl, furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3-triazole, 1, 2,4-triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q3分别独立地选自卤素、羟基、氨基、硝基、氰基、羧基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、C1-6烷硫基、卤代C1-6烷氧基、卤代C1-6烷硫基、羟基C1-6烷氧基、羟基C1-6烷硫基、氨基C1-6烷氧基、氨基C1-6烷硫基。Each Q3 is independently selected from halogen, hydroxyl, amino, nitro, cyano, carboxyl, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halo C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkane Thio, halogenated C 1-6 alkoxy, halogenated C 1-6 alkylthio , hydroxy C 1-6 alkoxy, hydroxy C 1-6 alkylthio, amino C 1-6 alkoxy, Amino C 1-6 alkylthio.
在上述某些实施方案中,环C选自任选被1-2个Q3取代的呋喃基、吡咯基、噻吩基、咪唑基、吡唑基、噁唑基、噻唑基、1,2,3-三氮唑、1,2,4-三氮唑、吡喃基、噻喃基、吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;In some of the above embodiments, ring C is selected from furyl, pyrrolyl, thienyl, imidazolyl, pyrazolyl, oxazolyl, thiazolyl, 1,2,3 - triazole, 1,2,4-triazole, pyranyl, thiopyranyl, pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl;
每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基。Each Q3 is independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxyl C 1-6 alkyl, amino C 1-6 alkyl , C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, hydroxyl C 1 -6 alkoxy or amino C 1-6 alkoxy.
在上述某些实施方案中,环C选自任选被1-2个Q3取代的吡啶基、嘧啶基、哒嗪基、吡嗪基或三嗪基;In some of the above embodiments, Ring C is selected from pyridyl, pyrimidinyl, pyridazinyl, pyrazinyl or triazinyl optionally substituted by 1-2 Q3;
每一Q3分别独立的选自卤素、羟基、氨基、C1-6烷基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基。Each Q3 is independently selected from halogen, hydroxyl, amino, C 1-6 alkyl, C 1-6 alkylamino, two (C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxyl C 1-6 alkyl, amino C 1-6 alkyl , C 1-6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, hydroxyl C 1 -6 alkoxy or amino C 1-6 alkoxy.
在上述某些实施方案中,每一Q3分别独立的选自C1-6烷基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、C1-6烷氧基C1-6烷基、C1-6烷氧基、卤代C1-6烷氧基、羟基C1-6烷氧基或氨基C1-6烷氧基。In some of the above embodiments, each Q3 is independently selected from C 1-6 alkyl, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, C 1 -6 alkoxy C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkoxy, hydroxy C 1-6 alkoxy or amino C 1-6 alkoxy.
在上述某些实施方案中,每一Q3分别独立的选自C1-6烷基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基或C1-6烷氧基C1-6烷基。In some of the above embodiments, each Q3 is independently selected from C 1-6 alkyl, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl or C 1 -6 alkoxy C 1-6 alkyl.
在上述某些实施方案中,X1、X2分别独立地选自C、CH或N。在上述某些实施方案中,X1、X2分别独立地选自CH或N。在上述某些实施方案中,X1、X2均为N。In certain embodiments above, X 1 and X 2 are independently selected from C, CH or N. In certain embodiments above, X 1 and X 2 are each independently selected from CH or N. In certain embodiments above, both X 1 and X 2 are N.
在上述某些实施方案中,L选自-CH(R5)-、-O-、-N(R6)-或-S-;R5选自氢、卤素、羟基、氨基、C1-6烷基、C1-6烷氧基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷氧基C1-6烷基;R6选自氢、C1-6烷基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基或C1-6烷氧基C1-6烷基。In some of the above embodiments, L is selected from -CH(R 5 )-, -O-, -N(R 6 )- or -S-; R 5 is selected from hydrogen, halogen, hydroxyl, amino, C 1- 6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkyl , hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1- 6 alkoxy C 1-6 alkyl; R 6 is selected from hydrogen, C 1-6 alkyl, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl or C 1-6 alkoxy C 1-6 alkyl.
在上述某些实施方案中,L选自-CH(R5)-、-O-、-N(R6)-或-S-;R5选自氢、卤素、羟基、氨基、甲基、乙基、异丙基、甲氧基或三氟甲基;R6选自氢、甲基、乙基或异丙基。In some of the above embodiments, L is selected from -CH(R 5 )-, -O-, -N(R 6 )- or -S-; R 5 is selected from hydrogen, halogen, hydroxyl, amino, methyl, Ethyl, isopropyl, methoxy or trifluoromethyl; R 6 is selected from hydrogen, methyl, ethyl or isopropyl.
在上述某些实施方案中,L选自-CH2-、-O-、-NH-或-S-。在上述某些实施方案中,L选自-O-、-NH-或-S-。在上述某些实施方案中,L为-NH-。In certain embodiments of the above, L is selected from -CH2- , -O-, -NH- or -S-. In certain embodiments of the above, L is selected from -O-, -NH- or -S-. In certain embodiments of the above, L is -NH-.
在上述某些实施方案中,每一R2分别独立地选自卤素、羟基、氨基、羧基、C1-6烷基、C1-6烷氧基、C1-6烷基氨基、二(C1-6烷基)氨基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷氧基或C1-6烷基羰基。In certain embodiments above, each R is independently selected from halogen, hydroxy, amino, carboxyl, C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, di( C 1-6 alkyl) amino, halogenated C 1-6 alkyl, hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkoxy or C 1-6 alkyl carbonyl.
在上述某些实施方案中,每一R2分别独立地选自卤素、羟基、氨基、C1-6烷基、C1-6烷氧基、卤代C1-6烷基、羟基C1-6烷基、氨基C1-6烷基或卤代C1-6烷氧基。In certain embodiments above, each R is independently selected from halogen, hydroxy, amino, C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkyl, hydroxy C 1 -6 alkyl, amino C 1-6 alkyl or halogenated C 1-6 alkoxy.
在上述某些实施方案中,每一R2分别独立地选自卤素C1-6烷基、C1-6烷氧基、卤代C1-6烷基或卤代C1-6烷氧基。In some of the above embodiments, each R is independently selected from halogenated C 1-6 alkyl, C 1-6 alkoxy, halogenated C 1-6 alkyl or halogenated C 1-6 alkoxy base.
在上述某些实施方案中,m选自0、1或2。在上述某些实施方案中,m选自0或1。在上述某些实施方案中,m选自0。In certain embodiments above, m is selected from 0, 1 or 2. In certain embodiments above, m is selected from 0 or 1. In certain embodiments above, m is selected from zero.
在上述某些实施方案中,n选自0或1。在上述某些实施方案中,n选自0。In certain embodiments above, n is selected from 0 or 1. In certain embodiments above, n is selected from zero.
本发明中各技术方案之间可以相互组合形成新的技术方案,所形成的新的技术方案同样包括在本发明的范围之内。Various technical solutions in the present invention can be combined with each other to form new technical solutions, and the formed new technical solutions are also included in the scope of the present invention.
在某些实施方案,前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所示的化合物、其药学上可接受的盐或其立体异构体,选自如下化合物:In certain embodiments, the compound represented by the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), and a pharmaceutically acceptable salt thereof or its stereoisomers, selected from the following compounds:
本发明还提供了一种药物制剂,其含有前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述的化合物、其药学上可接受的盐或其立体异构体,及一种或多种药用载体和/或稀释剂;所述药物制剂可以制成临床上或药学上可接受的任一剂型。The present invention also provides a pharmaceutical preparation, which contains the compound described in the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), its A pharmaceutically acceptable salt or its stereoisomer, and one or more pharmaceutically acceptable carriers and/or diluents; the pharmaceutical preparation can be made into any clinically or pharmaceutically acceptable dosage form.
本发明还提供了一种药物组合物,其包含前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述的化合物、其药学上可接受的盐或其立体异构体,还包含一种或多种第二治疗活性剂,所述的第二治疗活性剂选自有丝分裂抑制剂、抗癌性烷化剂、抗癌性代谢拮抗剂、抗癌性铂配位化合物、抗癌性喜树碱衍生物、抗肿瘤抗生素、生长因子抑制剂、信号传导抑制剂、细胞周期抑制剂、抗癌性酪氨酸激酶抑制剂、干扰素、类维生素A受体调控剂、蛋白酶体抑制剂、拓扑异构酶抑制剂、生物应答调节剂、激素类抗癌剂、血管再生抑制剂、芳香化酶抑制剂、细胞生长抑制剂、靶向抗体、HMG-CoA还原酶抑制剂和蛋白质异戊二烯基转移酶抑制剂。The present invention also provides a pharmaceutical composition, which comprises the compound described in the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), A pharmaceutically acceptable salt thereof or a stereoisomer thereof, further comprising one or more second therapeutically active agents selected from the group consisting of mitotic inhibitors, anticancer alkylating agents, anticancer Sexual metabolic antagonists, anticancer platinum coordination compounds, anticancer camptothecin derivatives, antitumor antibiotics, growth factor inhibitors, signal transduction inhibitors, cell cycle inhibitors, anticancer tyrosine kinase inhibitors , interferon, retinoid receptor modulator, proteasome inhibitor, topoisomerase inhibitor, biological response modifier, hormonal anticancer agent, angiogenesis inhibitor, aromatase inhibitor, cytostatic agent , targeting antibodies, HMG-CoA reductase inhibitors and protein prenyl transferase inhibitors.
本发明的药物组合物或药物制剂中可用的药用载体和/或稀释剂可以是药物制剂领域中任何常规的载体和/或稀释剂,特定载体和/或稀释剂的选择将取决于用于治疗特定患者的给药方式或疾病类型和状态。The pharmaceutically acceptable carrier and/or diluent available in the pharmaceutical composition or pharmaceutical preparation of the present invention can be any conventional carrier and/or diluent in the pharmaceutical preparation field, and the selection of specific carrier and/or diluent will depend on the The mode of administration or disease type and state to treat a particular patient.
在另一方面,本发明还涉及前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述化合物、其药学上可接受的盐或其立体异构体在制备预防和/或治疗由Wee1所介导的疾病及相关疾病的药物中的用途,所述疾病及相关病症选自癌症,所述癌症包括原位癌和转移的癌症。In another aspect, the present invention also relates to the compound described in the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), and its pharmaceutically acceptable Use of salts or stereoisomers thereof in the preparation of medicines for preventing and/or treating diseases mediated by Weel and related diseases, the diseases and related diseases being selected from cancer, the cancer including carcinoma in situ and metastasis of cancer.
进一步的,本发明还涉及含有前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述化合物、其药学上可接受的盐或其立体异构体的药物制剂在制备药物中的用途,所述疾病及相关病症选自癌症,所述癌症包括原位癌和转移的癌症。Further, the present invention also relates to a compound containing the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), and its pharmaceutically acceptable Use of a pharmaceutical formulation of a salt or a stereoisomer thereof for the manufacture of a medicament for the disease and related conditions selected from cancer, including carcinoma in situ and metastatic cancer.
进一步的,本发明还涉及含有前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述化合物、其药学上可接受的盐或其立体异构体以及一种或多种第二治疗活性剂的药物组合物在制备用于治疗和/或预防由Wee1所介导的疾病和相关病症的药物中的用途,所述疾病及相关病症选自癌症,所述癌症包括原位癌和转移的癌症。Further, the present invention also relates to a compound containing the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), and its pharmaceutically acceptable Use of a pharmaceutical composition of a salt or a stereoisomer thereof and one or more second therapeutically active agents for the preparation of a medicament for the treatment and/or prevention of diseases and related conditions mediated by Weel, said diseases and related disorders are selected from cancers including carcinoma in situ and metastatic cancers.
上述方案中,所述癌症包括但不限于肺癌、鳞状上皮细胞癌、膀胱癌、胃癌、卵巢癌、腹膜癌、胰腺癌、乳腺癌、头颈癌、子宫颈癌、子宫内膜癌、直肠癌、肝癌、肾癌、食管腺癌、食管鳞状细胞癌、前列腺癌、甲状腺癌、雌性生殖道癌、淋巴瘤、神经纤维瘤、骨癌、皮肤癌、脑癌、结肠癌、睾丸癌、小细胞肺癌、非小细胞肺癌、胃肠道间质瘤、肥大细胞肿瘤、多发性骨髓瘤、黑色素瘤、白血病、胶质瘤或肉瘤。In the above scheme, the cancers include but not limited to lung cancer, squamous cell carcinoma, bladder cancer, gastric cancer, ovarian cancer, peritoneal cancer, pancreatic cancer, breast cancer, head and neck cancer, cervical cancer, endometrial cancer, rectal cancer , liver cancer, kidney cancer, esophageal adenocarcinoma, esophageal squamous cell carcinoma, prostate cancer, thyroid cancer, female reproductive tract cancer, lymphoma, neurofibroma, bone cancer, skin cancer, brain cancer, colon cancer, testicular cancer, small Cell lung cancer, non-small cell lung cancer, gastrointestinal stromal tumor, mast cell tumor, multiple myeloma, melanoma, leukemia, glioma, or sarcoma.
上述方案中,所述第二治疗活性剂选自有丝分裂抑制剂、抗癌性烷化剂、抗癌性代谢拮抗剂、抗癌性铂配位化合物、抗癌性喜树碱衍生物、抗肿瘤抗生素、生长因子抑制剂、信号传导抑制剂、细胞周期抑制剂、抗癌性酪氨酸激酶抑制剂、干扰素、类维生素A受体调控剂、蛋白酶体抑制剂、拓扑异构酶抑制剂、生物应答调节剂、激素类抗癌剂、血管再生抑制剂、芳香化酶抑制剂、细胞生长抑制剂、靶向抗体、HMG-CoA还原酶抑制剂和蛋白质异戊二烯基转移酶抑制剂。In the above scheme, the second therapeutically active agent is selected from the group consisting of mitosis inhibitors, anticancer alkylating agents, anticancer metabolic antagonists, anticancer platinum coordination compounds, anticancer camptothecin derivatives, antitumor Antibiotics, growth factor inhibitors, signal transduction inhibitors, cell cycle inhibitors, anticancer tyrosine kinase inhibitors, interferons, retinoid receptor modulators, proteasome inhibitors, topoisomerase inhibitors, Biological response modifiers, hormonal anticancer agents, angiogenesis inhibitors, aromatase inhibitors, cytostatic agents, targeting antibodies, HMG-CoA reductase inhibitors and protein prenyl transferase inhibitors.
在某些实施方案中,待组合的各成分(例如,本发明的化合物、其药学上可接受的盐、其立体异构体与第二治疗活性剂)可同时给药或依次顺序地分开用药。例如,可以在施用本发明化合物、其药学上可接受的盐或其立体异构体之前、同时或之后,施用第二治疗活性剂。此外,待组合的各成分还可以以同一制剂形式或以分开的不同制剂的形式联合给药。In certain embodiments, the components to be combined (e.g., a compound of the invention, a pharmaceutically acceptable salt thereof, a stereoisomer thereof, and a second therapeutically active agent) may be administered simultaneously or sequentially and separately . For example, the second therapeutically active agent may be administered before, simultaneously with, or after administration of a compound of the invention, a pharmaceutically acceptable salt thereof, or a stereoisomer thereof. Furthermore, the individual components to be combined can also be administered jointly in the form of the same formulation or in the form of separate different formulations.
在另一方面,本发明还提供了一种治疗由Wee1介导的疾病及相关病症的方法,该方法包括向有需要的患者施用有效量的前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述的化合物、其药学上可接受的盐或其立体异构体,前述制剂或药物组合物;所述由Wee1介导的疾病及相关病症如上定义。In another aspect, the present invention also provides a method for treating diseases mediated by Weel and related diseases, the method comprising administering an effective amount of the aforementioned general formula (I), general formula (I-1) to patients in need ), the compound described in general formula (II), general formula (III) or general formula (IV), its pharmaceutically acceptable salt or its stereoisomer, the aforementioned preparation or pharmaceutical composition; Leading diseases and related conditions are as defined above.
所述的“有效量”是指能够减轻、延缓、抑制或治愈受试者病症的药物剂量。给药剂量的大小与药物给药方式、药剂的药代动力学、疾病的严重程度、受试者的个性体征(性别、体重、身高、年龄)等来确定。The "effective amount" refers to the drug dose that can reduce, delay, inhibit or cure the subject's symptoms. The size of the dosage is determined by the way of drug administration, the pharmacokinetics of the drug, the severity of the disease, and the individual signs (gender, weight, height, age) of the subject.
本发明涉及的含有前述通式(I)、通式(I-1)、通式(II)、通式(III)或通式(IV)所述化合物、其药学上可接受的盐或其立体异构体的药物还可通过与放疗组合,提高放疗的治疗效果,即可作为放疗敏化剂使用。The present invention relates to the compound containing the aforementioned general formula (I), general formula (I-1), general formula (II), general formula (III) or general formula (IV), its pharmaceutically acceptable salt or its Stereoisomer drugs can also be combined with radiotherapy to improve the therapeutic effect of radiotherapy, and can be used as radiotherapy sensitizers.
在另一方面,本发明化合物在癌症治疗领域还可作为其他抗癌剂(第二治疗活性剂)的敏化剂使用。所述第二治疗活性剂如前所述。所述敏化剂表示,在癌症治疗领域,通过与放疗和或使用抗癌剂的化学疗法组合使用,相加或协同地提高这些放疗和/或化学疗法的治疗效果的药物。In another aspect, the compounds of the invention are also useful in the field of cancer therapy as sensitizers to other anticancer agents (second therapeutically active agents). The second therapeutically active agent is as previously described. The sensitizer means, in the field of cancer treatment, a drug that additively or synergistically increases the therapeutic effect of radiation therapy and/or chemotherapy by using it in combination with radiation therapy and/or chemotherapy using an anticancer agent.
【发明详述】【Detailed description of the invention】
在本发明中,除非另有说明,否则本文中使用的科学和技术名词具有本领域技术人员通常理解的含义,然而为了更好的理解本发明,下面提供了部分术语的定义。当本发明所提供的术语的定义和解释与本领域技术人员所通常理解的含义不符的时候,以本发明所提供的术语的定义和解释为准。In the present invention, unless otherwise specified, the scientific and technical terms used herein have the meanings generally understood by those skilled in the art. However, in order to better understand the present invention, definitions of some terms are provided below. When the definitions and explanations of the terms provided in the present invention are inconsistent with the meanings commonly understood by those skilled in the art, the definitions and explanations of the terms provided in the present invention shall prevail.
本发明所述的“卤素”是指氟原子、氯原子、溴原子或碘原子。The "halogen" in the present invention refers to fluorine atom, chlorine atom, bromine atom or iodine atom.
本发明所述的“C1-6烷基”表示直链或支链的含有1-6个碳原子的烷基,包括例如“C1-4烷基”、“C1-3烷基”、“C1-2烷基”、“C2-6烷基”、“C2-5烷基”、“C2-4烷基”、“C2-3烷基”、“C3-6烷基”、“C3-5烷基”、“C3-4烷基”等,具体实例包括但不限于:甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、正戊基、异戊基、2-甲基丁基、新戊基、1-乙基丙基、正己基、异己基、3-甲基戊基、2-甲基戊基、1-甲基戊基、3,3-二甲基丁基、2,2-二甲基丁基、1,1-二甲基丁基、1,2-二甲基丁基、1,3-二甲基丁基、2,3-二甲基丁基、2-乙基丁基、1,2-二甲基丙基等。本发明所述的“C1-4烷基”指C1-6烷基中的含有1-4个碳原子的具体实例。The "C 1-6 alkyl" in the present invention means a linear or branched alkyl group containing 1-6 carbon atoms, including, for example, "C 1-4 alkyl", "C 1-3 alkyl" , "C 1-2 alkyl", "C 2-6 alkyl", "C 2-5 alkyl", "C 2-4 alkyl", "C 2-3 alkyl", "C 3- 6 alkyl", "C 3-5 alkyl", "C 3-4 alkyl", etc. Specific examples include but are not limited to: methyl, ethyl, n-propyl, isopropyl, n-butyl, iso Butyl, sec-butyl, tert-butyl, n-pentyl, isopentyl, 2-methylbutyl, neopentyl, 1-ethylpropyl, n-hexyl, isohexyl, 3-methylpentyl, 2-methylpentyl, 1-methylpentyl, 3,3-dimethylbutyl, 2,2-dimethylbutyl, 1,1-dimethylbutyl, 1,2-dimethyl butyl, 1,3-dimethylbutyl, 2,3-dimethylbutyl, 2-ethylbutyl, 1,2-dimethylpropyl, etc. The "C 1-4 alkyl" in the present invention refers to specific examples of C 1-6 alkyl containing 1-4 carbon atoms.
本发明所述的“C1-6烷氧基”是指“C1-6烷基-O-”,所述的“C1-6烷基”如前文所定义。本发明所述的“C1-4烷氧基”是指“C1-4烷基-O-”,所述的“C1-4烷基”如前文所定义。The "C 1-6 alkoxy" in the present invention refers to "C 1-6 alkyl-O-", and the "C 1-6 alkyl" is as defined above. The "C 1-4 alkoxy" in the present invention refers to "C 1-4 alkyl-O-", and the "C 1-4 alkyl" is as defined above.
本发明所述的“C1-6烷硫基”是指“C1-6烷基-S-”,所述的“C1-6烷基”如前文所定义。本发明所述的“C1-4烷硫基”是指“C1-4烷基-S-”,所述的“C1-4烷基”如前文所定义。The "C 1-6 alkylthio" in the present invention refers to "C 1-6 alkyl-S-", and the "C 1-6 alkyl" is as defined above. The "C 1-4 alkylthio" in the present invention refers to "C 1-4 alkyl-S-", and the "C 1-4 alkyl" is as defined above.
本发明所述的“羟基C1-6烷基、氨基C1-6烷基、卤代C1-6烷基”是指C1-6烷基中的一个或多个氢分别被一个或多个羟基、氨基或卤素所取代。C1-6烷基如前文所定义"Hydroxy C 1-6 alkyl, amino C 1-6 alkyl, halogenated C 1-6 alkyl" in the present invention means that one or more hydrogens in C 1-6 alkyl are respectively replaced by one or Substituted by multiple hydroxyl groups, amino groups or halogens. C 1-6 alkyl is as defined above
本发明所述“羟基C1-6烷氧基、氨基C1-6烷氧基、卤代C1-6烷氧基”是指“C1-6烷氧基”中的一个或多个氢被一个或多个羟基、氨基或卤素所取代。"Hydroxy C 1-6 alkoxy, amino C 1-6 alkoxy, halogenated C 1-6 alkoxy" in the present invention refers to one or more of "C 1-6 alkoxy" Hydrogens are replaced by one or more hydroxy, amino or halo.
本发明所述“羟基C1-6烷硫基、氨基C1-6烷硫基、卤代C1-6烷硫基”是指“C1-6烷硫基”中的一个或多个氢被一个或多个羟基、氨基或卤素所取代。"Hydroxy C 1-6 alkylthio, amino C 1-6 alkylthio, halogenated C 1-6 alkylthio" in the present invention refers to one or more of "C 1-6 alkylthio" Hydrogens are replaced by one or more hydroxy, amino or halo.
本发明所述的“C1-6烷基氨基、二(C1-6烷基)氨基”分别是指C1-6烷基-NH-、 "C 1-6 alkylamino, di(C 1-6 alkyl) amino" in the present invention refers to C 1-6 alkyl-NH-,
本发明所述的“C2-6烯基”是指含有至少一个双键且碳原子数为2-6的直链、支链或环状的烯基,包括例如“C2-4烯基”等。其实例包括但不限于:乙烯基、1-丙烯基、2-丙烯基、1-丁烯基、2-丁烯基、1,3-丁二烯基、1-戊烯基、2-戊烯基、3-戊烯基、1,3-戊二烯基、1,4-戊二烯基、1-己烯基、2-己烯基、3-己烯基、1,4-己二烯基、环戊烯基、1,3-环戊二烯基、环己烯基、1,4-环己二烯基等。The "C 2-6 alkenyl" in the present invention refers to a straight-chain, branched or cyclic alkenyl group containing at least one double bond and having 2-6 carbon atoms, including, for example, "C 2-4 alkenyl "wait. Examples include, but are not limited to: vinyl, 1-propenyl, 2-propenyl, 1-butenyl, 2-butenyl, 1,3-butadienyl, 1-pentenyl, 2-pentenyl Alkenyl, 3-pentenyl, 1,3-pentadienyl, 1,4-pentadienyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 1,4-hex Dienyl, cyclopentenyl, 1,3-cyclopentadienyl, cyclohexenyl, 1,4-cyclohexadienyl and the like.
本发明所述的“C2-6炔基”是指含有至少一个三键且碳原子数为2-6的直链或支链的炔基,包括例如“C2-4炔基”等。其实例包括但不限于:乙炔基、丙炔基、2-丁炔基、2-戊炔基、3-戊炔基、4-甲基-2-戊炔基、2-己炔基、3-己炔基、5-甲基-2-己炔基等。The "C 2-6 alkynyl" in the present invention refers to a straight-chain or branched alkynyl containing at least one triple bond and having 2-6 carbon atoms, including, for example, "C 2-4 alkynyl". Examples include, but are not limited to: ethynyl, propynyl, 2-butynyl, 2-pentynyl, 3-pentynyl, 4-methyl-2-pentynyl, 2-hexynyl, 3 -hexynyl, 5-methyl-2-hexynyl and the like.
本发明所述的“3-10元环烷基”是指包括“3-8元单环烷基”和“8-10元稠环烷基”。The "3-10 membered cycloalkyl group" in the present invention refers to include "3-8 membered monocycloalkyl group" and "8-10 membered condensed cycloalkyl group".
本发明所述的“3-8元单环烷基”是指含有3-8个碳原子的饱和或部分饱和的且不具有芳香性的单环环状烷基,包括“3-8元饱和单环烷基”和“3-8元部分饱和单环烷基”;优选“3-4元单环烷基”、“3-5元单环烷基”、“3-6元单环烷基”、“3-7元单环烷基”、“4-6元单环烷基”、“4-7元单环烷基”、“4-8元单环烷基”、“5-6元单环烷基”、“5-7元单环烷基”、“5-8元单环烷基”、“6-7元单环烷基”、“6-8元单环烷基”、“7-8元单环烷基”、“3-6元饱和单环烷基”、“5-8元饱和单环烷基”、“5-7元饱和单环烷基”、“5-6元饱和单环烷基”等。所述的“3-8元饱和单环烷基”的具体实例包括但不限于:环丙基、环丁基、环戊基、环己基、环庚基、环辛基;所述的“3-8元部分饱和单环烷基”的具体实例包括但不限于环丙烯基、环丁烯基、环戊烯基、环戊二烯基、环己烯基、环己-1,3-二烯、环己-1,4-二烯、环庚烯基、环庚-1,3-二烯基、环庚-1,4-二烯基、环庚-1,3,5-三烯基,环辛烯基、环辛-1,3-二烯基、环辛-1,4-二烯基、环辛-1,5-二烯基、环辛-1,3,5-三烯基、环辛四烯基。The "3-8 membered monocycloalkyl" in the present invention refers to a saturated or partially saturated monocyclic cyclic alkyl group containing 3-8 carbon atoms and does not have aromaticity, including "3-8 membered saturated Monocycloalkyl" and "3-8 membered partially saturated monocycloalkyl"; preferably "3-4 membered monocycloalkyl", "3-5 membered monocycloalkyl", "3-6 membered monocycloalkane ", "3-7 membered monocycloalkyl", "4-6 membered monocycloalkyl", "4-7 membered monocycloalkyl", "4-8 membered monocycloalkyl", "5- 6-membered monocycloalkyl", "5-7-membered monocycloalkyl", "5-8-membered monocycloalkyl", "6-7-membered monocycloalkyl", "6-8-membered monocycloalkyl ", "7-8 membered monocycloalkyl", "3-6 membered saturated monocycloalkyl", "5-8 membered saturated monocycloalkyl", "5-7 membered saturated monocycloalkyl", " 5-6 membered saturated monocycloalkyl", etc. Specific examples of the "3-8 membered saturated monocycloalkyl" include, but are not limited to: cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl; the "3 - Specific examples of 8-membered partially saturated monocycloalkyl" include, but are not limited to, cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclopentadienyl, cyclohexenyl, cyclohexa-1,3-di Alkene, cyclohexa-1,4-diene, cycloheptenyl, cyclohepta-1,3-dienyl, cyclohepta-1,4-dienyl, cyclohepta-1,3,5-triene Base, cyclooctyl, cyclooct-1,3-dienyl, cyclooct-1,4-dienyl, cyclooct-1,5-dienyl, cyclooct-1,3,5-tri alkenyl, cyclooctatetraenyl.
本发明所述的“8-10元稠环基”是指由两个或两个以上环状结构彼此共用两个相邻的碳原子所形成的含有8-10个环原子的、饱和或部分饱和的、非芳香性环状基团,所述的稠环中的一个环可以为芳香性环,但稠环整体不具备芳香性;包括“8-9元稠环基”、“9-10元稠环基”等,其稠和方式可以为:5-6元环烷基并5-6元环烷基、苯并5-6元环烷基、苯并5-6元饱和环烷基等。其实例包括但不限于:二环[3.1.0]己烷基、二环[4.1.0]庚烷基、二环[2.2.0]己烷基、二环[3.2.0]庚烷基、二环[4.2.0]辛烷基、八氢并环戊二烯基、八氢-1H-茚基、十氢化萘基、十四氢菲基、双环[3.1.0]己-2-烯基、双环[4.1.0]庚-3-烯基、双环[3.2.0]庚-3-烯基、双环[4.2.0]辛-3-烯基、1,2,3,3a-四氢并环戊二烯基、2,3,3a,4,7,7a-六氢-1H-茚基、1,2,3,4,4a,5,6,8a-八氢化萘基、1,2,4a,5,6,8a-六氢化萘基、1,2,3,4,5,6,7,8,9,10-十氢菲基、苯并环戊基、苯并环己基、苯并环己烯基、苯并环戊烯基等。The "8-10 membered condensed ring group" in the present invention refers to a saturated or partial ring structure containing 8-10 ring atoms formed by sharing two adjacent carbon atoms with each other. Saturated, non-aromatic cyclic group, one ring in the fused ring may be an aromatic ring, but the fused ring as a whole is not aromatic; including "8-9 membered fused ring group", "9-10 Member fused ring group", etc., the condensing method can be: 5-6 membered cycloalkyl and 5-6 membered cycloalkyl, benzo 5-6 membered cycloalkyl, benzo 5-6 membered saturated cycloalkyl wait. Examples include, but are not limited to: bicyclo[3.1.0]heptyl, bicyclo[4.1.0]heptanyl, bicyclo[2.2.0]hexyl, bicyclo[3.2.0]heptanyl , bicyclo[4.2.0]octyl, octahydropentalenyl, octahydro-1H-indenyl, decalinyl, tetrahydrophenanthryl, bicyclo[3.1.0]hex-2- Alkenyl, bicyclo[4.1.0]hept-3-enyl, bicyclo[3.2.0]hept-3-enyl, bicyclo[4.2.0]oct-3-enyl, 1,2,3,3a- Tetrahydropentalenyl, 2,3,3a,4,7,7a-hexahydro-1H-indenyl, 1,2,3,4,4a,5,6,8a-octahydronaphthyl, 1,2,4a,5,6,8a-hexahydronaphthyl, 1,2,3,4,5,6,7,8,9,10-decahydrophenanthrenyl, benzocyclopentyl, benzo Cyclohexyl, benzocyclohexenyl, benzocyclopentenyl, etc.
本发明所述的“3-10元杂环基”包括“3-8元单杂环基”和“8-10元稠杂环基”。The "3-10 membered heterocyclic group" in the present invention includes "3-8 membered monoheterocyclic group" and "8-10 membered condensed heterocyclic group".
本发明所述的“3-8元单杂环基”是指至少含有一个杂原子或基团(例如,含有1个、2个、3个、4个或5个)的且环原子数为3-8个的饱和或部分饱和的且不具有芳香性的单环环状基团,所述杂原子为氮原子、氧原子和/或硫原子,且环状结构中的环原子(例如碳原子、氮原子或硫原子)可以被氧代。本发明所述的“3-8元单杂环基”包括“3-8元饱和单杂环基”和“3-8元部分饱和单杂环基”。优选地,本发明所述的“3-8元单杂环基”含有1-3个杂原子或基团;优选地,本发明所述的“3-8元单杂环基”含有1-2个杂原子或基团,且所述的杂原子选自氮原子和/或氧原子;优选地,本发明所述的“3-8元单杂环基”含有1-2个氮原子。所述“3-8元单杂环基”优选“3-7元单杂环基”、“3-6元单杂环基”、“4-7元单杂环基”、“4-6元单杂环基”、“6-8元单杂环基”、“5-7元单杂环基”、“5-6元单杂环基”、“3-6元饱和单杂环基”、“5-6元饱和单杂环基”、“3-6元含氮单杂环基”、“3-6元饱和含氮单杂环基”、“5-6元含氮单杂环基”、“5-6元饱和含氮单杂环基”等。例如,仅包含1个或2个氮原子,或者,包含一个氮原子和其他的1个或2个杂原子(例如氧原子和/或硫原子)。“3-8元单杂环基”的具体实例包括但不仅限于:氮杂环丙烷基、2H-氮杂环丙烷基、二氮杂环丙烷基、3H-二氮杂环丙烯基、氮杂环丁烷基、1,4-二氧杂环己烷基、1,3-二氧杂环己烷基、1,3-二氧杂环戊烷基、1,4-二氧杂环己二烯基、四氢呋喃基、二氢吡咯基、吡咯烷基、咪唑烷基、4,5-二氢咪唑基、吡唑烷基、4,5-二氢吡唑基、2,5-二氢噻吩基、四氢噻吩基、4,5-二氢噻唑基、噻唑烷基、哌啶基、四氢吡啶基、哌啶酮基、四氢吡啶酮基、二氢哌啶酮基、哌嗪基、吗啉基、4,5-二氢噁唑基、4,5-二氢异噁唑基、2,3-二氢异噁唑基、噁唑烷基、2H-1,2-噁嗪基、4H-1,2-噁嗪基、6H-1,2-噁嗪基、4H-1,3-噁嗪基、6H-1,3-噁嗪基、4H-1,4-噁嗪基、4H-1,3-噻嗪基、6H-1,3-噻嗪基、2H-吡喃基、2H-吡喃-2-酮基、3,4-二氢-2H-吡喃基等。The "3-8 membered monoheterocyclic group" in the present invention refers to a heterocyclic group containing at least one heteroatom or group (for example, containing 1, 2, 3, 4 or 5) and the number of ring atoms is 3-8 saturated or partially saturated and non-aromatic monocyclic ring groups, the heteroatoms are nitrogen atoms, oxygen atoms and/or sulfur atoms, and the ring atoms in the ring structure (such as carbon atom, nitrogen atom or sulfur atom) may be oxo-substituted. The "3-8 membered monoheterocyclic group" in the present invention includes "3-8 membered saturated monoheterocyclic group" and "3-8 membered partially saturated monoheterocyclic group". Preferably, the "3-8 membered monoheterocyclic group" described in the present invention contains 1-3 heteroatoms or groups; preferably, the "3-8 membered monoheterocyclic group" described in the present invention contains 1- 2 heteroatoms or groups, and the heteroatoms are selected from nitrogen atoms and/or oxygen atoms; preferably, the "3-8 membered monoheterocyclic group" in the present invention contains 1-2 nitrogen atoms. The "3-8 membered monoheterocyclic group" is preferably "3-7 membered monoheterocyclic group", "3-6 membered monoheterocyclic group", "4-7 membered monoheterocyclic group", "4-6 Monocyclic heterocyclic group", "6-8 monoheterocyclic group", "5-7 monoheterocyclic group", "5-6 monoheterocyclic group", "3-6 saturated monoheterocyclic group ", "5-6 membered saturated monoheterocyclic group", "3-6 membered nitrogen-containing monoheterocyclic group", "3-6 membered saturated nitrogen-containing monoheterocyclic group", "5-6 membered nitrogen-containing monoheterocyclic group" Cyclic group", "5-6 membered saturated nitrogen-containing monoheterocyclic group" and so on. For example, containing only 1 or 2 nitrogen atoms, or containing 1 nitrogen atom and other 1 or 2 heteroatoms (such as oxygen atoms and/or sulfur atoms). Specific examples of "3-8 membered monoheterocyclyl" include, but are not limited to: aziridine group, 2H-aziridine group, diaziridine group, 3H-diaziridine group, aza Cyclobutanyl, 1,4-dioxanyl, 1,3-dioxanyl, 1,3-dioxolanyl, 1,4-dioxanyl Dienyl, tetrahydrofuranyl, dihydropyrrolyl, pyrrolidinyl, imidazolidinyl, 4,5-dihydroimidazolyl, pyrazolidinyl, 4,5-dihydropyrazolyl, 2,5-dihydro Thienyl, tetrahydrothienyl, 4,5-dihydrothiazolyl, thiazolidinyl, piperidinyl, tetrahydropyridyl, piperidonyl, tetrahydropyridinyl, dihydropiperidinyl, piperazine morpholinyl, 4,5-dihydrooxazolyl, 4,5-dihydroisoxazolyl, 2,3-dihydroisoxazolyl, oxazolidinyl, 2H-1,2-oxa Azinyl, 4H-1,2-oxazinyl, 6H-1,2-oxazinyl, 4H-1,3-oxazinyl, 6H-1,3-oxazinyl, 4H-1,4-oxazinyl Azinyl, 4H-1,3-thiazinyl, 6H-1,3-thiazinyl, 2H-pyranyl, 2H-pyran-2-onyl, 3,4-dihydro-2H-pyranyl Base etc.
本发明所述“8-10元稠杂环基”是指由两个或两个以上环状结构彼此共用两个相邻的原子所形成的含有8-10个环原子的、且至少一个环原子为杂原子或基团的、饱和或部分饱和的、非芳香性环状基团,所述的稠环中其中一个环可以为芳香性环,但稠环整体不具备芳香性,所述杂原子为氮原子、氧原子和/或硫原子,环状结构中的环原子(例如碳原子、氮原子或硫原子)可以被氧代,包括但不限于“8-9元稠杂环基”、“9-10元稠杂环基”等,其稠和方式包括但不限于:5-6元杂环基并5-6元杂环基、5-6元杂环基并5-6元环烷基、苯并5-6元杂环基、苯并5-6元饱和杂环基、5-6元杂芳基并5-6元杂环基、5-6元杂芳基并5-6元饱和杂环基;5-6元杂芳基如前文所定义;所述“8-10元稠杂环基”具体实例包括但不仅限于:吡咯烷基并环丙基、环戊基并氮杂环丙基、吡咯烷基并环丁基、吡咯烷基并吡咯烷基、吡咯烷基并哌啶基、吡咯烷基并哌嗪基、吡咯烷基并吗啉基、哌啶基并吗啉基、苯并吡咯烷基、苯并环戊基、苯并环己基、苯并四氢呋喃基、苯并吡咯烷基、苯并咪唑烷基、苯并噁唑烷基、苯并噻唑烷基、苯并异噁唑烷基、苯并异噻唑烷基、苯并哌啶基、苯并吗啉基、苯并哌嗪基、苯并四氢吡喃基、吡啶并环戊基、吡啶并环己基、吡啶并四氢呋喃基、吡啶并吡咯烷基、吡啶并咪唑烷基、吡啶并噁唑烷基、吡啶并噻唑烷基、吡啶并异噁唑烷基、吡啶并异噻唑烷基、吡啶并哌啶基、吡啶并吗啉基、吡啶并哌嗪基、吡啶并四氢吡喃基、嘧啶并环戊基、嘧啶并环己基、嘧啶并四氢呋喃基、嘧啶并吡咯烷基、嘧啶并咪唑烷基、嘧啶并噁唑烷基、嘧啶并噻唑烷基、嘧啶并异噁唑烷基、嘧啶并异噻唑烷基、嘧啶并哌啶基、嘧啶并吗啉基、嘧啶并哌嗪基、嘧啶并四氢吡喃基;四氢咪唑并[4,5-c]吡啶基、3,4-二氢喹唑啉基、1,2-二氢喹喔啉基、苯并[d][1,3]二氧杂环戊烯基、2H-色原烯基、2H-色原烯-2-酮基、4H-色烯基、4H-色烯-4-酮基、4H-1,3-苯并噁嗪基、4,6-二氢-1H-呋喃并[3,4-d]咪唑基、3a,4,6,6a-四氢-1H-呋喃并[3,4-d]咪唑基、4,6-二氢-1H-噻吩并[3,4-d]咪唑基、4,6-二氢-1H-吡咯并[3,4-d]咪唑基、八氢-苯并[d]咪唑基、十氢喹啉基、六氢噻吩并咪唑基、六氢呋喃并咪唑基、4,5,6,7-四氢-1H-苯并[d]咪唑基、八氢环戊烯并[c]吡咯基、4H-1,3-苯并噁嗪基等。The "8-10 membered condensed heterocyclic group" in the present invention refers to a ring structure containing 8-10 ring atoms and at least one ring structure formed by sharing two adjacent atoms with each other. Atoms are heteroatoms or groups, saturated or partially saturated, non-aromatic cyclic groups, one of the rings in the condensed ring may be an aromatic ring, but the fused ring as a whole does not have aromaticity, and the hetero Atoms are nitrogen atoms, oxygen atoms and/or sulfur atoms, and ring atoms (such as carbon atoms, nitrogen atoms or sulfur atoms) in the ring structure can be oxo, including but not limited to "8-9 membered fused heterocyclic group" , "9-10 membered fused heterocyclic group", etc., the fused methods include but not limited to: 5-6 membered heterocyclic group and 5-6 membered heterocyclic group, 5-6 membered heterocyclic group and 5-6 membered heterocyclic group Cycloalkyl, benzo 5-6 membered heterocyclic group, benzo 5-6 membered saturated heterocyclic group, 5-6 membered heteroaryl and 5-6 membered heterocyclic group, 5-6 membered heteroaryl and 5 -6-membered saturated heterocyclic group; 5-6-membered heteroaryl group as defined above; specific examples of the "8-10-membered condensed heterocyclic group" include, but are not limited to: pyrrolidinocyclopropyl, cyclopentyl Aziridyl, pyrrolidinocyclobutyl, pyrrolidinopyrrolidinyl, pyrrolidinopiperidinyl, pyrrolidinopiperazinyl, pyrrolidinomorpholinyl, piperidinyl Morpholinyl, benzopyrrolidinyl, benzocyclopentyl, benzocyclohexyl, benzotetrahydrofuranyl, benzopyrrolidinyl, benzimidazolidinyl, benzoxazolidinyl, benzothiazolidine base, benzisoxazolidinyl, benzisothiazolidinyl, benzopiperidinyl, benzomorpholinyl, benzopiperazinyl, benzotetrahydropyranyl, pyridopentyl, pyridine Cyclohexyl, pyridotetrahydrofuranyl, pyridopyrrolidinyl, pyridimidazolidinyl, pyridoxazolidinyl, pyridothiazolidinyl, pyridisoxazolidinyl, pyridylisothiazolidinyl, pyridine Piperidinyl, pyridomorpholinyl, pyridopiperazinyl, pyridotetrahydropyranyl, pyrimidocyclopentyl, pyrimidocyclohexyl, pyrimidotetrahydrofuranyl, pyrimidopyrrolidinyl, pyrimidimidazole Alkyl, pyrimidooxazolidinyl, pyrimidothiazolidinyl, pyrimidoisoxazolidinyl, pyrimidoisothiazolidinyl, pyrimidopiperidinyl, pyrimidomorpholinyl, pyrimidopiperazinyl, pyrimidine Tetrahydropyranyl; tetrahydroimidazo[4,5-c]pyridyl, 3,4-dihydroquinazolinyl, 1,2-dihydroquinoxalinyl, benzo[d][1 ,3] Dioxolyl, 2H-chromenyl, 2H-chromen-2-one, 4H-chromenyl, 4H-chromen-4-one, 4H-1,3 -Benzoxazinyl, 4,6-dihydro-1H-furo[3,4-d]imidazolyl, 3a,4,6,6a-tetrahydro-1H-furo[3,4-d] Imidazolyl, 4,6-dihydro-1H-thieno[3,4-d]imidazolyl, 4,6-dihydro-1H-pyrrolo[3,4-d]imidazolyl, octahydro-benzo [d] imidazolyl, decahydroquinolinyl, hexahydrothienoimidazolyl, hexahydrofuranoimidazolyl, 4,5,6,7-tetrahydro-1H-benzo[d]imidazolyl, octahydrocyclo Penteno[c]pyrrolyl, 4H-1,3-benzoxazinyl, etc.
术语“苯并环戊基”,其结构是指(亦可称之为2,3-二氢-1H-茚基);术语“苯并吡咯烷”其结构包括等;术语“吡啶并四氢呋喃基”其结构包括 前文所定义的其他“定义的其它稠杂环基”的具体实例具有与之类似的环状结构。The term "benzocyclopentyl", its structure refers to (also known as 2,3-dihydro-1H-indenyl); the structure of the term "benzopyrrolidine" includes etc.; the term "pyridotetrahydrofuranyl" has a structure including Specific examples of other "other defined condensed heterocyclic groups" defined above have ring structures similar thereto.
本发明所述的“6-10元芳基”包括“6-8元单环芳基”和“8-10元稠环芳基”。The "6-10 membered aryl group" in the present invention includes "6-8 membered monocyclic aryl group" and "8-10 membered fused ring aryl group".
本发明所述的“6-8元单环芳基”是指含有6-8个环碳原子的单环芳基,其实例包括但不限于:苯基、环辛四烯基等;优选苯基。The "6-8 membered monocyclic aryl group" in the present invention refers to a monocyclic aryl group containing 6-8 ring carbon atoms, examples of which include but are not limited to: phenyl, cyclooctatetraenyl, etc.; preferably benzene base.
本发明所述的“8-10元稠环芳基”是指由两个或两个以上环状结构彼此共用两个相邻的原子所形成的、含有8-10个环碳原子的、不饱和的、具有芳香性的环状基团,优选“9-10元稠环芳基”,具体实例如萘基等。The "8-10 membered fused ring aryl group" in the present invention refers to a group formed by two or more ring structures sharing two adjacent atoms, containing 8-10 ring carbon atoms, not Saturated, aromatic cyclic group, preferably "9-10 membered fused ring aryl", specific examples include naphthyl and the like.
本发明所述的“5-10元杂芳基”包括“5-8元单杂芳基”和“8-10元稠杂芳基”。The "5-10 membered heteroaryl" in the present invention includes "5-8 membered monoheteroaryl" and "8-10 membered condensed heteroaryl".
本发明所述的“5-8元单杂芳基”是指含有5-8个环原子(其中至少一个环原子为杂原子,例如氮原子、氧原子或硫原子)的具有芳香性的单环环状基团。环状结构中的环原子(例如碳原子、氮原子或硫原子)可以被氧代。“5-8元单杂芳基”包括例如“5-7元单杂芳基”、“5-6元单杂芳基”、“5-6元含氮单杂芳基”、“6元含氮单杂芳基”等,所述的“含氮杂芳基”中的杂原子至少含有一个氮原子,例如,仅包含1个或2个氮原子,或者,包含一个氮原子和其他的1个或2个杂原子(例如氧原子和/或硫原子),或者,包含2个氮原子和其他的1个或2个杂原子(例如氧原子和/或硫原子)。“5-8元单环杂芳基”的具体实例包括但不仅限于呋喃基、噻吩基、吡咯基、噻唑基、异噻唑基、噻二唑基、噁唑基、异噁唑基、噁二唑基、咪唑基、吡唑基、1,2,3-三唑基、1,2,4-三唑基、1,2,3-噁二唑基、1,2,4-噁二唑基、1,2,5-噁二唑基、1,3,4-噁二唑基、吡啶基、2-吡啶酮基、4-吡啶酮基、嘧啶基、哒嗪基、吡嗪基、1,2,3-三嗪基、1,3,5-三嗪基、1,2,4,5-四嗪基、氮杂环庚三烯基、1,3-二氮杂环庚三烯基、氮杂环辛四烯基等。所述“5-6元单杂芳基”是指5-8元杂芳基中含有5-6个环原子的具体实例。The "5-8 membered monoheteroaryl" in the present invention refers to an aromatic monoheteroaryl group containing 5-8 ring atoms (wherein at least one ring atom is a heteroatom, such as a nitrogen atom, an oxygen atom or a sulfur atom). ring ring group. A ring atom (such as a carbon atom, a nitrogen atom or a sulfur atom) in a ring structure may be oxo substituted. "5-8 membered monoheteroaryl" includes, for example, "5-7 membered monoheteroaryl", "5-6 membered monoheteroaryl", "5-6 membered nitrogen-containing monoheteroaryl", "6 membered Nitrogen-containing monoheteroaryl", etc., the heteroatom in the "nitrogen-containing heteroaryl" contains at least one nitrogen atom, for example, contains only 1 or 2 nitrogen atoms, or contains one nitrogen atom and other 1 or 2 heteroatoms (such as oxygen atom and/or sulfur atom), or, contain 2 nitrogen atoms and other 1 or 2 heteroatoms (such as oxygen atom and/or sulfur atom). Specific examples of "5-8 membered monocyclic heteroaryl" include, but are not limited to, furyl, thienyl, pyrrolyl, thiazolyl, isothiazolyl, thiadiazolyl, oxazolyl, isoxazolyl, oxadi Azolyl, imidazolyl, pyrazolyl, 1,2,3-triazolyl, 1,2,4-triazolyl, 1,2,3-oxadiazolyl, 1,2,4-oxadiazole Base, 1,2,5-oxadiazolyl, 1,3,4-oxadiazolyl, pyridyl, 2-pyridonyl, 4-pyridonyl, pyrimidinyl, pyridazinyl, pyrazinyl, 1,2,3-triazinyl, 1,3,5-triazinyl, 1,2,4,5-tetrazinyl, azepanyl, 1,3-diazepanyl alkenyl, azacyclooctatetraenyl, etc. The "5-6 membered monoheteroaryl" refers to a specific example of 5-6 ring atoms in the 5-8 membered heteroaryl.
本发明所述的“8-10元稠杂芳基”是指由两个或两个以上环状结构彼此共用两个相邻的原子所形成的、含有8-10个环原子(其中至少一个环原子为杂原子,例如氮原子、氧原子或硫原子)的、不饱和的具有芳香性的环状结构。任选地,环状结构中的环原子(例如碳原子、氮原子或硫原子)可以被氧代。包括“9-10元稠杂芳基”,“8-9元稠杂芳基”等,其稠和方式可以为苯并5-6元杂芳基、5-6元杂芳基并5-6元杂芳基等;具体实例包括但不限于:吡咯并吡咯、吡咯并呋喃、吡唑并吡咯、吡唑并噻吩、呋喃并噻吩、吡唑并噁唑、苯并呋喃基、苯并异呋喃基、苯并噻吩基、吲哚基、异吲哚基、苯并噁唑基、苯并咪唑基、吲唑基、苯并三唑基、喹啉基、2-喹啉酮基、4-喹啉酮基、1-异喹啉酮基、异喹啉基、吖啶基、菲啶基、苯并哒嗪基、酞嗪基、喹唑啉基、喹喔啉基、嘌呤基、萘啶基等。The "8-10 membered condensed heteroaryl" in the present invention refers to a group formed by two or more ring structures sharing two adjacent atoms with each other, containing 8-10 ring atoms (at least one of which is An unsaturated aromatic ring structure in which the ring atoms are heteroatoms, such as nitrogen atoms, oxygen atoms or sulfur atoms). Optionally, ring atoms (eg carbon, nitrogen or sulfur atoms) in the ring structure can be oxo substituted. Including "9-10 membered fused heteroaryl", "8-9 membered fused heteroaryl", etc., the condensed way can be benzo 5-6 membered heteroaryl, 5-6 membered heteroaryl and 5- 6-membered heteroaryl, etc.; specific examples include, but are not limited to: pyrrolopyrrole, pyrrolofuran, pyrazolopyrrole, pyrazolothiophene, furothiophene, pyrazoloxazole, benzofuryl, benziso Furyl, benzothienyl, indolyl, isoindolyl, benzoxazolyl, benzimidazolyl, indazolyl, benzotriazolyl, quinolinyl, 2-quinolinonyl, 4 -Quinolinone, 1-isoquinolinone, isoquinolinyl, acridinyl, phenanthridinyl, benzopyridazinyl, phthalazinyl, quinazolinyl, quinoxalinyl, purinyl, naphthyridinyl etc.
本发明所述的“碳原子、氮原子或硫原子被氧代”是指形成C=O、N=O、S=O或SO2的结构。"Carbon atom, nitrogen atom or sulfur atom is oxo-substituted" in the present invention refers to forming a structure of C=O, N=O, S=O or SO 2 .
本发明所述“任选被取代”是指被取代基上的一个或多个氢原子可以被一个或多个取代基“取代”或“不取代”的两种情形。The term "optionally substituted" in the present invention refers to two situations in which one or more hydrogen atoms on the substituted group can be "substituted" or "not substituted" by one or more substituents.
本发明所述的“药学上可接受的盐”指化合物中存在的酸性官能团(例如-COOH、-OH、-SO3H等)与适当的无机或者有机阳离子(碱)形成的盐,包括与碱金属或碱土金属形成的盐、铵盐,以及与含氮有机碱形成的盐;以及化合物中存在的碱性官能团(例如-NH2等)与适当的无机或者有机阴离子(酸)形成的盐,包括与无机酸或有机酸(例如羧酸等)形成的盐。The "pharmaceutically acceptable salt" in the present invention refers to the salt formed by the acidic functional group (such as -COOH, -OH, -SO 3 H, etc.) present in the compound and an appropriate inorganic or organic cation (base), including Salts formed by alkali metals or alkaline earth metals, ammonium salts, and salts formed with nitrogen-containing organic bases; and salts formed by basic functional groups (such as -NH2, etc.) present in compounds and suitable inorganic or organic anions (acids), Salts with inorganic acids or organic acids (eg, carboxylic acids, etc.) are included.
本发明所述的“异构体”是指当本发明化合物含有一个或多个不对称中心,因而可作为外消旋体和外消旋混合物、单一对映异构体、非对映异构体混合物和单一非对映异构体。本发明化合物可以有不对称中心,这类不对称中心各自独立地产生两个光学异构体。本发明的范围包括所有可能的光学异构体和它们的混合物。本发明所述的化合物若含有烯烃双键,除非特别说明,包括顺式异构体和反式异构体。本发明所述的化合物可以以互变异构体(官能团异构体的一种)形式存在,其通过一个或多个双键位移而具有不同的氢的连接点,例如,酮和它的烯醇形式是酮-烯醇互变异构体。本发明化合物含有螺环结构,受环的立体空间结构的影响,环上的取代基可存在于环两侧从而形成相对的顺式(cis)和反式(trans)异构体。各互变异构体及其混合物都包括在本发明的范围中。所有化合物的对映异构体、非对映异构体、外消旋体、内消旋体、顺反异构体、互变异构体、几何异构体、差向异构体及其混合物等,均包括在本发明范围中。The "isomer" in the present invention means that when the compound of the present invention contains one or more asymmetric centers, it can be used as racemate and racemic mixture, single enantiomer, diastereoisomer mixtures and individual diastereomers. The compounds of the present invention may have asymmetric centers, each of which independently produces two optical isomers. The scope of the present invention includes all possible optical isomers and mixtures thereof. If the compounds of the present invention contain olefinic double bonds, unless otherwise specified, they include cis-isomers and trans-isomers. The compounds described in the present invention may exist in the form of tautomers (one of functional group isomers), which have different points of attachment of hydrogen by displacement of one or more double bonds, for example, ketones and its alkenes The alcohol form is a keto-enol tautomer. The compound of the present invention contains a spiro ring structure. Affected by the three-dimensional structure of the ring, substituents on the ring may exist on both sides of the ring to form relative cis (cis) and trans (trans) isomers. Each tautomer and mixtures thereof are included within the scope of the present invention. Enantiomers, diastereoisomers, racemates, mesoisomers, cis-trans isomers, tautomers, geometric isomers, epimers and their Mixtures and the like are included in the scope of the present invention.
本发明化合物可通过对映体特异性合成或从对映异构体混合物拆分以得到个别对映异构体的形式制备。常规拆分技术包括使用光学活性酸形成对映异构体对的每一异构体的游离碱的盐(接着分步结晶和游离碱再生)、使用光学活性胺形成对映异构体对的每一对映异构体的酸形式的盐(接着分步结晶和游离酸再生)、使用光学纯酸、胺或醇形成对映异构体对的每一对映异构体中的每一种的酯或酰胺(接着为色谱分离和手性助剂去除)或使用各种众所周知的色谱方法拆分起始物质或最终产物的对映异构体的混合物。The compounds of the present invention may be prepared in the form of individual enantiomers by enantiospecific synthesis or resolution from enantiomeric mixtures. Conventional resolution techniques include formation of the free base salt of each isomer of the enantiomeric pair using an optically active acid (followed by fractional crystallization and regeneration of the free base), formation of the salt of the enantiomeric pair using an optically active amine, Salts of the acid forms of each enantiomer (followed by fractional crystallization and regeneration of the free acid), formation of each of each enantiomer of the enantiomeric pair using optically pure acids, amines or alcohols The esters or amides of the species (followed by chromatographic separation and removal of the chiral auxiliary) or the resolution of the starting material or the mixture of enantiomers of the final product using various well-known chromatographic methods.
当公开的化合物的立体化学通过结构命名或描绘时,命名或描绘的立体异构体相对于其他立体异构体为至少60%重量、70%重量、80%重量、90%重量、99%重量或99.9%重量纯。当单一异构体通过结构命名或描绘时,所描绘或命名的对映异构体为至少60%重量、70%重量、80%重量、90%重量、99%重量或99.9%重量纯。光学纯度重量%为对映异构体的重量与对映异构体重量加上其光学异构体的重量比率。When the stereochemistry of a disclosed compound is named or depicted by a structure, the named or depicted stereoisomer is at least 60%, 70%, 80%, 90%, 99% by weight relative to the other stereoisomers Or 99.9% pure by weight. When a single isomer is named or depicted by structure, the depicted or named enantiomer is at least 60%, 70%, 80%, 90%, 99% or 99.9% pure by weight. Optical purity wt% is the ratio of the weight of an enantiomer to the weight of the enantiomer plus the weight of its optical isomer.
本发明所述的“剂型”指将药物制成的适用于临床使用的形式,包括但不限于散剂、片剂、颗粒剂、胶囊剂、溶液剂、乳剂、混悬剂、注射剂(包括注射液、注射用无菌粉末与注射用浓溶液)、喷雾剂、气雾剂、粉雾剂、洗剂、搽剂、软膏剂、硬膏剂、糊剂、贴剂、含漱剂或栓剂,更优选散剂、片剂、颗粒剂、胶囊剂、溶液剂、注射剂、软膏剂、含漱剂或栓剂。The "dosage form" mentioned in the present invention refers to the form suitable for clinical use made of medicines, including but not limited to powders, tablets, granules, capsules, solutions, emulsions, suspensions, injections (including injections) , sterile powder for injection and concentrated solution for injection), spray, aerosol, powder spray, lotion, liniment, ointment, plaster, paste, patch, gargle or suppository, more preferably Powder, tablet, granule, capsule, solution, injection, ointment, gargle, or suppository.
发明的有益效果Beneficial Effects of the Invention
1、本发明化合物、其药学上可接受的盐或其立体异构体具有优异的Wee1活性抑制作用和抗肿瘤细胞增殖活性,其在生物体内具有良好的药代动力学性质,作用持久,暴露量和生物利用度高,能够治疗和/或预防由Wee1介导的疾病。1. The compound of the present invention, its pharmaceutically acceptable salt or its stereoisomer has excellent Wee1 activity inhibitory effect and anti-tumor cell proliferation activity, which has good pharmacokinetic properties in vivo, long-lasting effect, exposure The amount and bioavailability are high, and it can treat and/or prevent diseases mediated by Wee1.
2、本发明化合物、其药学上可接受的盐或其立体异构体在生物体内外对Wee1介导的癌症具有较好的抑制和治疗作用。2. The compounds of the present invention, their pharmaceutically acceptable salts or their stereoisomers have good inhibitory and therapeutic effects on Wee1-mediated cancer in vivo and in vitro.
3、本发明化合物、其药学上可接受的盐或其立体异构体可协同增强放疗和化疗效果,有效抑制肿瘤生长,还可降低对正常细胞的损伤并减少副作用。3. The compounds of the present invention, their pharmaceutically acceptable salts or their stereoisomers can synergistically enhance the effects of radiotherapy and chemotherapy, effectively inhibit tumor growth, and reduce damage to normal cells and side effects.
4、本发明化合物制备工艺简单,药品纯度高,质量稳定,易于进行大规模工业生产。4. The preparation process of the compound of the present invention is simple, the drug has high purity and stable quality, and is easy to carry out large-scale industrial production.
具体实施方案specific implementation plan
下面将结合具体实施方式对本发明技术方案进行描述,对本发明的上述内容作进一步的详细说明,但不应将此理解为本发明上述主题的范围仅限于以下实施例。凡基于本发明上述内容所实现的技术均属于本发明的范围。The technical solution of the present invention will be described below in conjunction with specific embodiments, and the above content of the present invention will be further described in detail, but it should not be understood that the scope of the above subject of the present invention is limited to the following examples. All technologies realized based on the above contents of the present invention belong to the scope of the present invention.
实施例1:2-烯丙基-6-((4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯基)氨基)-1-(6-(2-羟基丙-2-基)吡啶-2基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮的制备(化合物1)的制备Example 1: 2-allyl-6-((4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)phenyl)amino)-1-(6-( Preparation of 2-hydroxypropan-2-yl)pyridin-2-yl)-1,2-dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one (compound 1)
1、2-(4-硝基苯基)八氢吡咯并[1,2-a]吡嗪的制备1. Preparation of 2-(4-nitrophenyl) octahydropyrrolo[1,2-a]pyrazine
将1-氟-4-硝基苯(338mg,2.4mmol)溶于DMSO(10mL)中,加入K2CO3(662mg,4.8mmol),八氢吡咯并[1,2-a]吡嗪(300mg,2.4mmol),70℃下反应16h。反应完成,反应液加入EA与水,分液萃取三次,有机相旋干,经正向制备分离(PE:EA=1:5)得产物(480mg,收率81.6%)。Dissolve 1-fluoro-4-nitrobenzene (338 mg, 2.4 mmol) in DMSO (10 mL), add K 2 CO 3 (662 mg, 4.8 mmol), octahydropyrrolo[1,2-a]pyrazine ( 300mg, 2.4mmol), react at 70°C for 16h. After the reaction was completed, EA and water were added to the reaction solution, separated and extracted three times, the organic phase was spin-dried, and separated by forward preparation (PE:EA=1:5) to obtain the product (480 mg, yield 81.6%).
2、4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯胺的制备2, Preparation of 4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)aniline
将2-(4-硝基苯基)八氢吡咯并[1,2-a]吡嗪(470mg,1.9mmol)溶于甲醇(10mL)中,加入钯碳(40mg),后将混合物置于下反应25℃反应1h,抽滤,滤液浓缩得粗品(400mg)。Dissolve 2-(4-nitrophenyl)octahydropyrrolo[1,2-a]pyrazine (470mg, 1.9mmol) in methanol (10mL), add palladium on carbon (40mg), and place the mixture on The reaction was carried out at 25°C for 1 h, filtered with suction, and the filtrate was concentrated to obtain the crude product (400 mg).
3、2-烯丙基-6-((4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯基)氨基)-1-(6-(2-羟基丙-2-基)吡啶-2基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮的制备3. 2-allyl-6-((4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)phenyl)amino)-1-(6-(2- Preparation of Hydroxypropan-2-yl)pyridin-2yl)-1,2-dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one
将2-烯丙基-1-(6-(2-羟基丙-2-基)吡啶-2-基)-6-(甲基亚磺酰基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮(76mg)溶于甲苯(3mL)中,加入4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯胺(130mg粗品)和DIEA(129mg,1mmol),25℃反应10h。反应完毕,反应液旋干,经反相色谱纯化(水:乙腈=1:3)得产品(25mg)。2-allyl-1-(6-(2-hydroxyprop-2-yl)pyridin-2-yl)-6-(methylsulfinyl)-1,2-dihydro-3H-pyrazole Con[3,4-d]pyrimidin-3-one (76 mg) was dissolved in toluene (3 mL), and 4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)aniline was added (130mg crude product) and DIEA (129mg, 1mmol), react at 25°C for 10h. After the reaction was completed, the reaction solution was spin-dried and purified by reverse phase chromatography (water:acetonitrile=1:3) to obtain the product (25 mg).
分子式:C29H34N8O2分子量:526.6LC-MS(M/e):527.2(M+H+)Molecular formula: C 29 H 34 N 8 O 2 Molecular weight: 526.6LC-MS (M/e): 527.2 (M+H + )
1H-NMR(400MHz,MeOD)δ:10.1(s,1H),8.95(s,1H),8.10(t,1H),7.75(m,1H),7.65(m,1H),7.50–7.60(m,2H),6.90-7.00(m,2H),5.60–5.70(m,1H),5.30(s,1H),5.00(d,1H),4.83(d,1H),4.67(d,2H),3.75(m,1H),3.55(m,1H),3.05(m,2H),2.65(t,1H),2.35(t,1H),2.25(t,1H),2.05(m,2H),1.85(m,1H),1.70(m,2H),1.46(s,6H),1.40(m,1H). 1 H-NMR (400MHz, MeOD) δ: 10.1(s,1H), 8.95(s,1H), 8.10(t,1H), 7.75(m,1H), 7.65(m,1H), 7.50–7.60( m,2H),6.90-7.00(m,2H),5.60–5.70(m,1H),5.30(s,1H),5.00(d,1H),4.83(d,1H),4.67(d,2H) ,3.75(m,1H),3.55(m,1H),3.05(m,2H),2.65(t,1H),2.35(t,1H),2.25(t,1H),2.05(m,2H), 1.85(m,1H),1.70(m,2H),1.46(s,6H),1.40(m,1H).
实施例2:(R)-2-烯丙基-6-((4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯基)氨基)-1-(6-(2-羟基丙-2-基)吡啶-2基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮(化合物1-2)的制备Example 2: (R)-2-allyl-6-((4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)phenyl)amino)-1- (6-(2-hydroxyprop-2-yl)pyridin-2-yl)-1,2-dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one (compound 1-2) preparation
1、(R)-2-(4-硝基苯基)八氢吡咯并[1,2-a]吡嗪的制备1. Preparation of (R)-2-(4-nitrophenyl)octahydropyrrolo[1,2-a]pyrazine
将1-氟-4-硝基苯(350mg,2.5mmol)溶于DMSO(6mL)中,加入K2CO3(685mg,5.0mmol),(R)-八氢吡咯并[1,2-a]吡嗪(313mg,2.5mmol),70℃下反应7小时。反应完成,用水(20mL)和乙酸乙酯(20mL x2)萃取,有机相用水(20mL x3)洗涤,粗品用硅胶柱层析纯化(PE:EA=1:4)得产物(400mg,收率65.2%)。Dissolve 1-fluoro-4-nitrobenzene (350mg, 2.5mmol) in DMSO (6mL), add K 2 CO 3 (685mg, 5.0mmol), (R)-octahydropyrrolo[1,2-a ] Pyrazine (313mg, 2.5mmol), react at 70°C for 7 hours. The reaction was completed, extracted with water (20mL) and ethyl acetate (20mL x2), the organic phase was washed with water (20mL x3), and the crude product was purified by silica gel column chromatography (PE:EA=1:4) to obtain the product (400mg, yield 65.2 %).
2、(R)-4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯胺的制备2. Preparation of (R)-4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)aniline
将(R)-2-(4-硝基苯基)八氢吡咯并[1,2-a]吡嗪(400mg,1.6mmol)溶于甲醇(40mL)中,加入钯碳(120mg),用氢气置换气体三次,在氢气氛围下30℃反应1h,过滤除去固体,浓缩,得粗品(350mg)。Dissolve (R)-2-(4-nitrophenyl)octahydropyrrolo[1,2-a]pyrazine (400mg, 1.6mmol) in methanol (40mL), add palladium carbon (120mg), and use The gas was replaced by hydrogen three times, reacted at 30° C. for 1 h under a hydrogen atmosphere, filtered to remove the solid, and concentrated to obtain a crude product (350 mg).
3、(R)-2-烯丙基-6-((4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯基)氨基)-1-(6-(2-羟基丙-2-基)吡啶-2基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮的制备3. (R)-2-allyl-6-((4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)phenyl)amino)-1-(6 Preparation of -(2-hydroxypropan-2-yl)pyridin-2yl)-1,2-dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one
将2-烯丙基-1-(6-(2-羟基丙-2-基)吡啶-2-基)-6-(甲硫基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮(100mg,0.28mmol)溶于甲苯(15mL),加入间氯过氧苯甲酸(63mg,0.31mmol),25℃反应0.5小时。反应完成。加入N,N-二异丙基乙胺(180mg,1.39mmol)和(R)-4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯胺(67mg,0.31mmol),25℃反应16h。用乙酸乙酯(20mL)和饱和碳酸氢钠溶液(15mL)萃取粗品经反相色谱纯化(水:乙腈=1:2)得产物(42mg,收率28.5%)。分子式:C29H34N8O2分子量:526.6LC-MS(M/e):527.2(M+H+)2-allyl-1-(6-(2-hydroxyprop-2-yl)pyridin-2-yl)-6-(methylthio)-1,2-dihydro-3H-pyrazolo[ 3,4-d]Pyrimidin-3-one (100mg, 0.28mmol) was dissolved in toluene (15mL), m-chloroperoxybenzoic acid (63mg, 0.31mmol) was added, and reacted at 25°C for 0.5 hours. The reaction is complete. Add N,N-diisopropylethylamine (180 mg, 1.39 mmol) and (R)-4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)aniline (67 mg, 0.31mmol), reacted at 25°C for 16h. The crude product was extracted with ethyl acetate (20 mL) and saturated sodium bicarbonate solution (15 mL) and purified by reverse phase chromatography (water: acetonitrile = 1:2) to obtain the product (42 mg, yield 28.5%). Molecular formula: C 29 H 34 N 8 O 2 Molecular weight: 526.6LC-MS (M/e): 527.2 (M+H + )
1H-NMR(400MHz,MeOD)δ:10.20-10.05(brs,1H),8.82(s,1H),8.12-8.03(m,1H),7.81-7.75(m,1H),7.65-7.46(m,3H),6.93(d,J=8.8Hz,2H),5.71-5.60(m,1H),5.32(s,1H),5.00(d,J=9.2Hz,1H),4.90-4.80(m,1H),4.68-4.67(m,2H),3.80-3.70(m,1H),3.65-3.55(m,1H),3.06-3.01(m,2H),2.75-2.65(m,1H),2.40-2.32(m,1H),2.27-2.20(m,1H),2.10-1.99(m,2H),1.90-1.80(m,1H),1.78-1.65(m,2H),1.46(s,6H),1.44-1.35(m,1H)。 1 H-NMR (400MHz,MeOD)δ:10.20-10.05(brs,1H),8.82(s,1H),8.12-8.03(m,1H),7.81-7.75(m,1H),7.65-7.46(m ,3H),6.93(d,J=8.8Hz,2H),5.71-5.60(m,1H),5.32(s,1H),5.00(d,J=9.2Hz,1H),4.90-4.80(m, 1H),4.68-4.67(m,2H),3.80-3.70(m,1H),3.65-3.55(m,1H),3.06-3.01(m,2H),2.75-2.65(m,1H),2.40- 2.32(m,1H),2.27-2.20(m,1H),2.10-1.99(m,2H),1.90-1.80(m,1H),1.78-1.65(m,2H),1.46(s,6H), 1.44-1.35 (m, 1H).
实施例3:(S)-2-烯丙基-6-((4-(六氢吡咯并[1,2-a]吡嗪-2(1H)-基)苯基)氨基)-1-(6-(2-羟基丙-2-基)吡啶-2基)-1,2-二氢-3H-吡唑并[3,4-d]嘧啶-3-酮(化合物1-1)的制备Example 3: (S)-2-allyl-6-((4-(hexahydropyrrolo[1,2-a]pyrazin-2(1H)-yl)phenyl)amino)-1- (6-(2-hydroxyprop-2-yl)pyridin-2-yl)-1,2-dihydro-3H-pyrazolo[3,4-d]pyrimidin-3-one (compound 1-1) preparation
参照化合物1-2的制备方法制备化合物1-1,将原料(R)-八氢吡咯并[1,2-a]吡嗪替换为(S)-八氢吡咯并[1,2-a]吡嗪。Compound 1-1 was prepared according to the preparation method of compound 1-2, and the raw material (R)-octahydropyrrolo[1,2-a]pyrazine was replaced by (S)-octahydropyrrolo[1,2-a] pyrazine.
采用与实施例1-3相同或相似的方法制备得到化合物2-4。Compound 2-4 was prepared by the same or similar method as in Example 1-3.
实验方案Experimental program
以下提供本发明部分化合物的示例性实验方案,以显示本发明化合物有利活性和有益技术效果。但是应当理解,下述实验方案仅仅是对本发明内容的示例,而不是对本发明范围的限制。Exemplary experimental schemes of some compounds of the present invention are provided below to show the advantageous activities and beneficial technical effects of the compounds of the present invention. However, it should be understood that the following experimental scheme is only an example of the content of the present invention, rather than limiting the scope of the present invention.
实验例1本发明化合物对细胞增殖活性的抑制实验Experimental example 1 The compound of the present invention is to the inhibitory experiment of cell proliferation activity
试验方法:CellTiter-GloTest method: CellTiter-Glo
测试物:本发明部分化合物,其结构式和制备方法见本发明制备例。Test substance: some compounds of the present invention, see the preparation examples of the present invention for their structural formulas and preparation methods.
AZD1775,按照现有技术WO2007126128公开的方法制备,其结构如下所示:AZD1775, prepared according to the method disclosed in the prior art WO2007126128, its structure is as follows:
实验方法experimental method
1.实验材料和试剂1. Experimental Materials and Reagents
2.细胞培养基配比及铺板数量2. The ratio of cell culture medium and the number of plates
3.实验过程3. Experimental process
3.1化合物准备3.1 Compound preparation
测试化合物的10mM DMSO溶液,用DMSO 3倍梯度稀释,8个浓度。测试化合物10mMDMSO溶液,用DMSO配制为联用浓度的1000倍溶液,即300μM,100μM,30μM。The 10 mM DMSO solution of the test compound was diluted with 3-fold gradient in DMSO, with 8 concentrations. The 10 mM DMSO solution of the test compound was prepared with DMSO as a 1000-fold solution of the combined concentration, that is, 300 μM, 100 μM, and 30 μM.
配制吉西他滨(Gemcitabine)10mM的储备液,稀释至最高终浓度1000倍,即100μM,再用DMSO 3倍梯度稀释5个浓度。A 10 mM stock solution of Gemcitabine was prepared, diluted to 1000 times the highest final concentration, that is, 100 μM, and then diluted to 5 concentrations with 3 times of DMSO.
3.2试验过程3.2 Test process
细胞按照来源方建议条件培养,至状态良好的对数生长期,去除培养基,PBS洗一遍,0.25%胰蛋白酶消化,完全培养基终止后收集细胞,重悬至合适的浓度,接种到96孔板,每孔90μL,37℃,5%CO2培养箱培养24小时。The cells were cultured according to the conditions recommended by the source, until they reached the logarithmic growth phase in good condition, the medium was removed, washed once with PBS, digested with 0.25% trypsin, and the cells were collected after the completion of the complete medium, resuspended to an appropriate concentration, and inoculated into 96 wells plate, 90 μL per well, and incubate for 24 hours at 37°C in a 5% CO 2 incubator.
化合物单用:终浓度1000倍的化合物溶液,培养基稀释100倍,每孔加入10μL,37℃,5%CO2培养箱培养72h后检测。Compound single use: compound solution with a final concentration of 1000 times, diluted 100 times in the culture medium, added 10 μL to each well, incubated at 37°C, 5% CO 2 incubator for 72 hours, and then detected.
化合物和吉西他滨联用:吉西他滨终浓度1000倍的溶液,培养基稀释100倍,每孔加入10μL,37℃,5%CO2培养箱继续培养24小时。终浓度1000倍的化合物溶液,培养基稀释20倍,每孔加入5μL,37℃,5%CO2培养箱继续培养24小时后检测。Combination of compound and gemcitabine: 1000 times the final concentration of gemcitabine, dilute the culture medium 100 times, add 10 μL to each well, and continue to incubate for 24 hours at 37°C in a 5% CO 2 incubator. For the compound solution with a final concentration of 1000 times, the culture medium was diluted 20 times, 5 μL was added to each well, and the culture was continued for 24 hours at 37° C. in a 5% CO 2 incubator for detection.
培养板提前放置室温,每孔加入60μL的CTG检测液,避光,在定轨摇床上震动2分钟使细胞裂解,反应共20分钟后,用多功能酶标仪读取冷光值。Place the culture plate at room temperature in advance, add 60 μL of CTG detection solution to each well, avoid light, and shake on an orbital shaker for 2 minutes to lyse the cells. After a total of 20 minutes of reaction, read the luminescence value with a multi-functional microplate reader.
4.数据处理4. Data processing
利用非线性S曲线回归来拟合数据得出剂量-效应曲线,并由此计算IC50值。The data were fitted using non-linear S-curve regression to generate dose-response curves from which IC50 values were calculated.
实验结果Experimental results
表1本发明化合物单用体外细胞抑制活性Table 1 Compounds of the present invention have single-use in vitro cytostatic activity
表2本发明化合物与吉西他滨联用体外细胞抑制活性Table 2 Compounds of the present invention combined with gemcitabine in vitro cytostatic activity
注*:联用时显示的数据为吉西他滨的IC50值。Note*: The data displayed in combination is the IC 50 value of gemcitabine.
实验结论Experimental results
本发明化合物单用对测试细胞株具有良好的抑制作用,与吉西他滨联用,对MIAPaCa2细胞增殖活性抑制有显著的协同作用。The compound of the present invention has a good inhibitory effect on test cell lines when used alone, and has a significant synergistic effect on inhibiting the proliferation activity of MIAPaCa2 cells when used in combination with gemcitabine.
Claims (11)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110952817.9A CN115707708A (en) | 2021-08-19 | 2021-08-19 | Wee1 kinase inhibitors |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110952817.9A CN115707708A (en) | 2021-08-19 | 2021-08-19 | Wee1 kinase inhibitors |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN115707708A true CN115707708A (en) | 2023-02-21 |
Family
ID=85212545
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110952817.9A Pending CN115707708A (en) | 2021-08-19 | 2021-08-19 | Wee1 kinase inhibitors |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN115707708A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116462687A (en) * | 2022-01-18 | 2023-07-21 | 江苏天士力帝益药业有限公司 | WEE1 inhibitor and preparation and application thereof |
-
2021
- 2021-08-19 CN CN202110952817.9A patent/CN115707708A/en active Pending
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116462687A (en) * | 2022-01-18 | 2023-07-21 | 江苏天士力帝益药业有限公司 | WEE1 inhibitor and preparation and application thereof |
| WO2023138362A1 (en) * | 2022-01-18 | 2023-07-27 | 江苏天士力帝益药业有限公司 | Wee1 inhibitor, preparation therefor, and use thereof |
| CN116462687B (en) * | 2022-01-18 | 2025-01-07 | 江苏天士力帝益药业有限公司 | WEE1 inhibitors and their preparation and use |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112047948B (en) | Kras mutant inhibitors | |
| JP5752232B2 (en) | Substituted pyrrolotriazine compounds as protein kinase inhibitors | |
| CN106029659B (en) | Glutaminase inhibitors | |
| US9765076B2 (en) | Aurora kinase inhibitors for inhibiting mitotic progression | |
| US10300058B2 (en) | Tyrosine kinase inhibitor and uses thereof | |
| EP3260454B1 (en) | Condensed pyrimidine compound or salt thereof | |
| CN114846005A (en) | SHP2 inhibitor and application thereof | |
| US20210238184A1 (en) | Therapeutic compounds | |
| ES2668479T3 (en) | Substituted pyrimidine compounds and their use as SYK inhibitors | |
| EA007063B1 (en) | AMINOBENZAMIDE DERIVATIVES AS GLYCOGEN SYNTHASE KINASE 3β INHIBITORS | |
| EP4232020A1 (en) | Treatment of hematological malignancies with inhibitors of menin | |
| CN107635986A (en) | Substituted quinoxaline derivatives | |
| JP2016531858A (en) | Novel 3- (1H-pyrazol-4-yl) -1H-pyrrolo [2,3-c] pyridine derivatives as NIK inhibitors | |
| US20200017485A1 (en) | Compounds and methods for regulating insulin secretion | |
| US20200071326A1 (en) | Tam kinase inhibitors | |
| US9724331B2 (en) | Use of maleimide derivatives for preventing and treating leukemia | |
| EP1914234A1 (en) | Pyrido[2,3-d]pyrimidines and their use as kinase inhibitors | |
| JP2022506802A (en) | Macrocyclic tyrosine kinase inhibitors and their uses | |
| CN111171049A (en) | Tyrosine kinase inhibitors and uses thereof | |
| WO2021043208A1 (en) | 3, 5-disubstituted pyrazole compounds as kinase inhibitors and uses thereof | |
| CN105884695B (en) | Heterocyclic derivatives species tyrosine kinase inhibitor | |
| US20210163478A1 (en) | Fused ring derivative used as fgfr4 inhibitor | |
| JP6426745B2 (en) | Conformationally immobilized PI3K and mTOR inhibitors | |
| CN115197221B (en) | Dihydropyrazolopyrimidinone macrocyclic derivatives and uses thereof | |
| CN104804016B (en) | Four and ring class anaplastic lymphoma kinase inhibitor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20230221 |
|
| WD01 | Invention patent application deemed withdrawn after publication |