CN114369083A - Heterocyclic compound, pharmaceutical composition containing same, preparation method and application thereof - Google Patents
Heterocyclic compound, pharmaceutical composition containing same, preparation method and application thereof Download PDFInfo
- Publication number
- CN114369083A CN114369083A CN202011104889.XA CN202011104889A CN114369083A CN 114369083 A CN114369083 A CN 114369083A CN 202011104889 A CN202011104889 A CN 202011104889A CN 114369083 A CN114369083 A CN 114369083A
- Authority
- CN
- China
- Prior art keywords
- compound
- cycloalkyl
- alkyl
- optionally substituted
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 239000008194 pharmaceutical composition Substances 0.000 title claims description 40
- 238000002360 preparation method Methods 0.000 title abstract description 9
- 150000002391 heterocyclic compounds Chemical class 0.000 title abstract description 8
- 150000001875 compounds Chemical class 0.000 claims abstract description 217
- MWUXSHHQAYIFBG-UHFFFAOYSA-N Nitric oxide Chemical compound O=[N] MWUXSHHQAYIFBG-UHFFFAOYSA-N 0.000 claims abstract description 112
- 150000003839 salts Chemical class 0.000 claims abstract description 45
- 239000000651 prodrug Substances 0.000 claims abstract description 42
- 229940002612 prodrug Drugs 0.000 claims abstract description 42
- 150000002148 esters Chemical class 0.000 claims abstract description 41
- 230000000155 isotopic effect Effects 0.000 claims abstract description 40
- 239000012453 solvate Substances 0.000 claims abstract description 40
- 239000002207 metabolite Substances 0.000 claims abstract description 39
- ZRKFYGHZFMAOKI-QMGMOQQFSA-N tgfbeta Chemical compound C([C@H](NC(=O)[C@H](C(C)C)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@H](CC(C)C)NC(=O)CNC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@@H](NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CCSC)C(C)C)[C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C=CC=CC=1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O)C1=CC=C(O)C=C1 ZRKFYGHZFMAOKI-QMGMOQQFSA-N 0.000 claims abstract description 18
- 125000000217 alkyl group Chemical class 0.000 claims description 90
- -1 isotopic label Substances 0.000 claims description 77
- 229910052739 hydrogen Inorganic materials 0.000 claims description 70
- 239000001257 hydrogen Substances 0.000 claims description 69
- 125000000623 heterocyclic group Chemical group 0.000 claims description 54
- 150000002431 hydrogen Chemical class 0.000 claims description 50
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 44
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 43
- 229910052736 halogen Inorganic materials 0.000 claims description 43
- 150000002367 halogens Chemical class 0.000 claims description 43
- 125000006552 (C3-C8) cycloalkyl group Chemical group 0.000 claims description 40
- 238000006467 substitution reaction Methods 0.000 claims description 34
- 125000001424 substituent group Chemical group 0.000 claims description 28
- 125000005913 (C3-C6) cycloalkyl group Chemical group 0.000 claims description 26
- 201000010099 disease Diseases 0.000 claims description 26
- 239000003814 drug Substances 0.000 claims description 25
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 23
- 125000000171 (C1-C6) haloalkyl group Chemical group 0.000 claims description 22
- 125000004076 pyridyl group Chemical group 0.000 claims description 18
- 125000002393 azetidinyl group Chemical group 0.000 claims description 17
- 208000035475 disorder Diseases 0.000 claims description 17
- 229910052757 nitrogen Inorganic materials 0.000 claims description 17
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 16
- 125000001313 C5-C10 heteroaryl group Chemical group 0.000 claims description 16
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 16
- 125000004433 nitrogen atom Chemical group N* 0.000 claims description 16
- 125000003386 piperidinyl group Chemical group 0.000 claims description 16
- 206010028980 Neoplasm Diseases 0.000 claims description 14
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 claims description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 claims description 13
- 229940124597 therapeutic agent Drugs 0.000 claims description 13
- 125000004890 (C1-C6) alkylamino group Chemical group 0.000 claims description 12
- 125000004193 piperazinyl group Chemical group 0.000 claims description 11
- 230000001404 mediated effect Effects 0.000 claims description 10
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 9
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 claims description 9
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 claims description 9
- 201000007270 liver cancer Diseases 0.000 claims description 9
- 208000014018 liver neoplasm Diseases 0.000 claims description 9
- 230000002265 prevention Effects 0.000 claims description 9
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 claims description 9
- 125000003226 pyrazolyl group Chemical group 0.000 claims description 9
- 125000006570 (C5-C6) heteroaryl group Chemical group 0.000 claims description 8
- 201000011510 cancer Diseases 0.000 claims description 8
- 238000004519 manufacturing process Methods 0.000 claims description 7
- 150000003254 radicals Chemical class 0.000 claims description 7
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 6
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 5
- 125000006310 cycloalkyl amino group Chemical group 0.000 claims description 5
- 125000001559 cyclopropyl group Chemical group [H]C1([H])C([H])([H])C1([H])* 0.000 claims description 5
- 229910052805 deuterium Inorganic materials 0.000 claims description 5
- 239000003937 drug carrier Substances 0.000 claims description 5
- ZSXGLVDWWRXATF-UHFFFAOYSA-N N,N-dimethylformamide dimethyl acetal Chemical compound COC(OC)N(C)C ZSXGLVDWWRXATF-UHFFFAOYSA-N 0.000 claims description 4
- 238000006482 condensation reaction Methods 0.000 claims description 4
- 125000001072 heteroaryl group Chemical group 0.000 claims description 4
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 claims description 4
- 238000004806 packaging method and process Methods 0.000 claims description 4
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 claims description 4
- 238000007363 ring formation reaction Methods 0.000 claims description 4
- 125000005843 halogen group Chemical group 0.000 claims description 3
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 claims description 2
- 125000001153 fluoro group Chemical group F* 0.000 claims description 2
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 claims description 2
- 238000007347 radical substitution reaction Methods 0.000 claims 4
- 229910003827 NRaRb Inorganic materials 0.000 claims 3
- 125000004356 hydroxy functional group Chemical group O* 0.000 claims 3
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims 1
- 125000006569 (C5-C6) heterocyclic group Chemical group 0.000 claims 1
- 230000005764 inhibitory process Effects 0.000 abstract description 22
- 239000000203 mixture Substances 0.000 abstract description 19
- 239000003112 inhibitor Substances 0.000 abstract description 11
- 230000000694 effects Effects 0.000 abstract description 7
- 230000000259 anti-tumor effect Effects 0.000 abstract description 2
- 239000003550 marker Substances 0.000 abstract 1
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 39
- 238000006243 chemical reaction Methods 0.000 description 36
- 238000000034 method Methods 0.000 description 36
- 238000012360 testing method Methods 0.000 description 34
- 125000003545 alkoxy group Chemical group 0.000 description 29
- WEVYAHXRMPXWCK-UHFFFAOYSA-N Acetonitrile Chemical compound CC#N WEVYAHXRMPXWCK-UHFFFAOYSA-N 0.000 description 27
- 239000012071 phase Substances 0.000 description 26
- 239000000243 solution Substances 0.000 description 24
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 21
- 102000004887 Transforming Growth Factor beta Human genes 0.000 description 20
- 108090001012 Transforming Growth Factor beta Proteins 0.000 description 20
- 238000003786 synthesis reaction Methods 0.000 description 17
- 230000015572 biosynthetic process Effects 0.000 description 16
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 15
- 238000005481 NMR spectroscopy Methods 0.000 description 14
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 13
- 125000004432 carbon atom Chemical group C* 0.000 description 13
- 230000019491 signal transduction Effects 0.000 description 13
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 12
- 125000003118 aryl group Chemical group 0.000 description 12
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 12
- 230000002401 inhibitory effect Effects 0.000 description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 11
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 10
- 210000004027 cell Anatomy 0.000 description 10
- WMFOQBRAJBCJND-UHFFFAOYSA-M Lithium hydroxide Chemical compound [Li+].[OH-] WMFOQBRAJBCJND-UHFFFAOYSA-M 0.000 description 8
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 8
- 239000003153 chemical reaction reagent Substances 0.000 description 8
- 239000012043 crude product Substances 0.000 description 8
- 238000002474 experimental method Methods 0.000 description 8
- 230000000069 prophylactic effect Effects 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 7
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 7
- 125000004429 atom Chemical group 0.000 description 7
- 125000000000 cycloalkoxy group Chemical group 0.000 description 7
- 150000007529 inorganic bases Chemical class 0.000 description 7
- 150000007530 organic bases Chemical class 0.000 description 7
- 239000012074 organic phase Substances 0.000 description 7
- 229910052717 sulfur Inorganic materials 0.000 description 7
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 6
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical class CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 6
- 125000002619 bicyclic group Chemical group 0.000 description 6
- 125000004122 cyclic group Chemical group 0.000 description 6
- 125000005366 cycloalkylthio group Chemical group 0.000 description 6
- 125000001995 cyclobutyl group Chemical group [H]C1([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 6
- 238000001514 detection method Methods 0.000 description 6
- 229940079593 drug Drugs 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- BDAGIHXWWSANSR-UHFFFAOYSA-N methanoic acid Natural products OC=O BDAGIHXWWSANSR-UHFFFAOYSA-N 0.000 description 6
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 6
- 239000002904 solvent Substances 0.000 description 6
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 5
- 125000000041 C6-C10 aryl group Chemical group 0.000 description 5
- 108010074922 Cytochrome P-450 CYP1A2 Proteins 0.000 description 5
- 108010001237 Cytochrome P-450 CYP2D6 Proteins 0.000 description 5
- 108010081668 Cytochrome P-450 CYP3A Proteins 0.000 description 5
- 102100026533 Cytochrome P450 1A2 Human genes 0.000 description 5
- 102100021704 Cytochrome P450 2D6 Human genes 0.000 description 5
- 102100039205 Cytochrome P450 3A4 Human genes 0.000 description 5
- 239000000460 chlorine Substances 0.000 description 5
- 125000005842 heteroatom Chemical group 0.000 description 5
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 5
- 125000002950 monocyclic group Chemical group 0.000 description 5
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 5
- 239000013642 negative control Substances 0.000 description 5
- 230000007170 pathology Effects 0.000 description 5
- 125000006239 protecting group Chemical group 0.000 description 5
- 229920006395 saturated elastomer Polymers 0.000 description 5
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 5
- 229910000104 sodium hydride Inorganic materials 0.000 description 5
- HPALAKNZSZLMCH-UHFFFAOYSA-M sodium;chloride;hydrate Chemical class O.[Na+].[Cl-] HPALAKNZSZLMCH-UHFFFAOYSA-M 0.000 description 5
- 208000024891 symptom Diseases 0.000 description 5
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- 125000006700 (C1-C6) alkylthio group Chemical group 0.000 description 4
- 101150051438 CYP gene Proteins 0.000 description 4
- 102000004190 Enzymes Human genes 0.000 description 4
- 108090000790 Enzymes Proteins 0.000 description 4
- DFPAKSUCGFBDDF-UHFFFAOYSA-N Nicotinamide Chemical compound NC(=O)C1=CC=CN=C1 DFPAKSUCGFBDDF-UHFFFAOYSA-N 0.000 description 4
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical compound [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 4
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 4
- 150000001408 amides Chemical class 0.000 description 4
- 125000003277 amino group Chemical group 0.000 description 4
- 230000037396 body weight Effects 0.000 description 4
- 239000000872 buffer Substances 0.000 description 4
- 229940125904 compound 1 Drugs 0.000 description 4
- 238000009472 formulation Methods 0.000 description 4
- 238000011534 incubation Methods 0.000 description 4
- 238000006317 isomerization reaction Methods 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 238000002953 preparative HPLC Methods 0.000 description 4
- 235000017557 sodium bicarbonate Nutrition 0.000 description 4
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 4
- 230000001225 therapeutic effect Effects 0.000 description 4
- OSWFIVFLDKOXQC-UHFFFAOYSA-N 4-(3-methoxyphenyl)aniline Chemical compound COC1=CC=CC(C=2C=CC(N)=CC=2)=C1 OSWFIVFLDKOXQC-UHFFFAOYSA-N 0.000 description 3
- 241000124008 Mammalia Species 0.000 description 3
- LRHPLDYGYMQRHN-UHFFFAOYSA-N N-Butanol Chemical compound CCCCO LRHPLDYGYMQRHN-UHFFFAOYSA-N 0.000 description 3
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 3
- ACFIXJIJDZMPPO-NNYOXOHSSA-N NADPH Chemical compound C1=CCC(C(=O)N)=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](COP(O)(=O)OP(O)(=O)OC[C@@H]2[C@H]([C@@H](OP(O)(O)=O)[C@@H](O2)N2C3=NC=NC(N)=C3N=C2)O)O1 ACFIXJIJDZMPPO-NNYOXOHSSA-N 0.000 description 3
- SAHIZENKTPRYSN-UHFFFAOYSA-N [2-[3-(phenoxymethyl)phenoxy]-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound O(C1=CC=CC=C1)CC=1C=C(OC2=NC(=CC(=C2)CN)C(F)(F)F)C=CC=1 SAHIZENKTPRYSN-UHFFFAOYSA-N 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 238000007792 addition Methods 0.000 description 3
- 125000004414 alkyl thio group Chemical group 0.000 description 3
- 239000007900 aqueous suspension Substances 0.000 description 3
- HONIICLYMWZJFZ-UHFFFAOYSA-N azetidine Chemical compound C1CNC1 HONIICLYMWZJFZ-UHFFFAOYSA-N 0.000 description 3
- 125000005605 benzo group Chemical group 0.000 description 3
- FJDQFPXHSGXQBY-UHFFFAOYSA-L caesium carbonate Chemical compound [Cs+].[Cs+].[O-]C([O-])=O FJDQFPXHSGXQBY-UHFFFAOYSA-L 0.000 description 3
- 239000002775 capsule Substances 0.000 description 3
- 229910052799 carbon Inorganic materials 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 238000002038 chemiluminescence detection Methods 0.000 description 3
- 235000019253 formic acid Nutrition 0.000 description 3
- 125000001188 haloalkyl group Chemical group 0.000 description 3
- 125000000592 heterocycloalkyl group Chemical group 0.000 description 3
- 229910052740 iodine Inorganic materials 0.000 description 3
- 229930027945 nicotinamide-adenine dinucleotide Natural products 0.000 description 3
- 210000000056 organ Anatomy 0.000 description 3
- 239000003960 organic solvent Substances 0.000 description 3
- 239000003208 petroleum Substances 0.000 description 3
- 125000003367 polycyclic group Chemical group 0.000 description 3
- 229910000027 potassium carbonate Inorganic materials 0.000 description 3
- LPNYRYFBWFDTMA-UHFFFAOYSA-N potassium tert-butoxide Chemical compound [K+].CC(C)(C)[O-] LPNYRYFBWFDTMA-UHFFFAOYSA-N 0.000 description 3
- 238000001953 recrystallisation Methods 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 230000008929 regeneration Effects 0.000 description 3
- 238000011069 regeneration method Methods 0.000 description 3
- 238000012216 screening Methods 0.000 description 3
- 238000000926 separation method Methods 0.000 description 3
- 238000010898 silica gel chromatography Methods 0.000 description 3
- 239000003826 tablet Substances 0.000 description 3
- LWIHDJKSTIGBAC-UHFFFAOYSA-K tripotassium phosphate Chemical compound [K+].[K+].[K+].[O-]P([O-])([O-])=O LWIHDJKSTIGBAC-UHFFFAOYSA-K 0.000 description 3
- SZUVGFMDDVSKSI-WIFOCOSTSA-N (1s,2s,3s,5r)-1-(carboxymethyl)-3,5-bis[(4-phenoxyphenyl)methyl-propylcarbamoyl]cyclopentane-1,2-dicarboxylic acid Chemical compound O=C([C@@H]1[C@@H]([C@](CC(O)=O)([C@H](C(=O)N(CCC)CC=2C=CC(OC=3C=CC=CC=3)=CC=2)C1)C(O)=O)C(O)=O)N(CCC)CC(C=C1)=CC=C1OC1=CC=CC=C1 SZUVGFMDDVSKSI-WIFOCOSTSA-N 0.000 description 2
- WSLDOOZREJYCGB-UHFFFAOYSA-N 1,2-Dichloroethane Chemical compound ClCCCl WSLDOOZREJYCGB-UHFFFAOYSA-N 0.000 description 2
- 238000005160 1H NMR spectroscopy Methods 0.000 description 2
- ZKHQWZAMYRWXGA-KQYNXXCUSA-J ATP(4-) Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]1O ZKHQWZAMYRWXGA-KQYNXXCUSA-J 0.000 description 2
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 2
- ZKHQWZAMYRWXGA-UHFFFAOYSA-N Adenosine triphosphate Natural products C1=NC=2C(N)=NC=NC=2N1C1OC(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)C(O)C1O ZKHQWZAMYRWXGA-UHFFFAOYSA-N 0.000 description 2
- ATRRKUHOCOJYRX-UHFFFAOYSA-N Ammonium bicarbonate Chemical compound [NH4+].OC([O-])=O ATRRKUHOCOJYRX-UHFFFAOYSA-N 0.000 description 2
- 229910000013 Ammonium bicarbonate Inorganic materials 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- OKKJLVBELUTLKV-MZCSYVLQSA-N Deuterated methanol Chemical compound [2H]OC([2H])([2H])[2H] OKKJLVBELUTLKV-MZCSYVLQSA-N 0.000 description 2
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- MHAJPDPJQMAIIY-UHFFFAOYSA-N Hydrogen peroxide Chemical compound OO MHAJPDPJQMAIIY-UHFFFAOYSA-N 0.000 description 2
- AVXURJPOCDRRFD-UHFFFAOYSA-N Hydroxylamine Chemical class ON AVXURJPOCDRRFD-UHFFFAOYSA-N 0.000 description 2
- 238000004566 IR spectroscopy Methods 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 2
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N Phenol Chemical compound OC1=CC=CC=C1 ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 2
- 108091000080 Phosphotransferase Proteins 0.000 description 2
- JUJWROOIHBZHMG-UHFFFAOYSA-N Pyridine Chemical compound C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 2
- KEAYESYHFKHZAL-UHFFFAOYSA-N Sodium Chemical compound [Na] KEAYESYHFKHZAL-UHFFFAOYSA-N 0.000 description 2
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 description 2
- 239000012317 TBTU Substances 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- ZEEBGORNQSEQBE-UHFFFAOYSA-N [2-(3-phenylphenoxy)-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound C1(=CC(=CC=C1)OC1=NC(=CC(=C1)CN)C(F)(F)F)C1=CC=CC=C1 ZEEBGORNQSEQBE-UHFFFAOYSA-N 0.000 description 2
- CLZISMQKJZCZDN-UHFFFAOYSA-N [benzotriazol-1-yloxy(dimethylamino)methylidene]-dimethylazanium Chemical compound C1=CC=C2N(OC(N(C)C)=[N+](C)C)N=NC2=C1 CLZISMQKJZCZDN-UHFFFAOYSA-N 0.000 description 2
- SPEUIVXLLWOEMJ-UHFFFAOYSA-N acetaldehyde dimethyl acetal Natural products COC(C)OC SPEUIVXLLWOEMJ-UHFFFAOYSA-N 0.000 description 2
- 235000012538 ammonium bicarbonate Nutrition 0.000 description 2
- 239000001099 ammonium carbonate Substances 0.000 description 2
- 230000006907 apoptotic process Effects 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 239000013060 biological fluid Substances 0.000 description 2
- 229910000024 caesium carbonate Inorganic materials 0.000 description 2
- 210000000170 cell membrane Anatomy 0.000 description 2
- 229910052801 chlorine Inorganic materials 0.000 description 2
- 229940126543 compound 14 Drugs 0.000 description 2
- 229940125898 compound 5 Drugs 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 229940099112 cornstarch Drugs 0.000 description 2
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 239000003480 eluent Substances 0.000 description 2
- 230000002255 enzymatic effect Effects 0.000 description 2
- 210000002919 epithelial cell Anatomy 0.000 description 2
- 229910052731 fluorine Inorganic materials 0.000 description 2
- 150000008282 halocarbons Chemical class 0.000 description 2
- 230000002140 halogenating effect Effects 0.000 description 2
- 238000005658 halogenation reaction Methods 0.000 description 2
- 150000002430 hydrocarbons Chemical group 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000007918 intramuscular administration Methods 0.000 description 2
- 150000002500 ions Chemical class 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 230000000670 limiting effect Effects 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 238000004949 mass spectrometry Methods 0.000 description 2
- 230000004060 metabolic process Effects 0.000 description 2
- CKJNUZNMWOVDFN-UHFFFAOYSA-N methanone Chemical compound O=[CH-] CKJNUZNMWOVDFN-UHFFFAOYSA-N 0.000 description 2
- 125000001624 naphthyl group Chemical group 0.000 description 2
- 235000005152 nicotinamide Nutrition 0.000 description 2
- 239000011570 nicotinamide Substances 0.000 description 2
- 229960003966 nicotinamide Drugs 0.000 description 2
- 150000002825 nitriles Chemical class 0.000 description 2
- 239000002674 ointment Substances 0.000 description 2
- 230000003287 optical effect Effects 0.000 description 2
- 125000004430 oxygen atom Chemical group O* 0.000 description 2
- 230000000144 pharmacologic effect Effects 0.000 description 2
- RLOWWWKZYUNIDI-UHFFFAOYSA-N phosphinic chloride Chemical compound ClP=O RLOWWWKZYUNIDI-UHFFFAOYSA-N 0.000 description 2
- 102000020233 phosphotransferase Human genes 0.000 description 2
- 235000011181 potassium carbonates Nutrition 0.000 description 2
- 238000000634 powder X-ray diffraction Methods 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 125000006413 ring segment Chemical group 0.000 description 2
- 238000004467 single crystal X-ray diffraction Methods 0.000 description 2
- 229910000029 sodium carbonate Inorganic materials 0.000 description 2
- 235000017550 sodium carbonate Nutrition 0.000 description 2
- APSBXTVYXVQYAB-UHFFFAOYSA-M sodium docusate Chemical compound [Na+].CCCCC(CC)COC(=O)CC(S([O-])(=O)=O)C(=O)OCC(CC)CCCC APSBXTVYXVQYAB-UHFFFAOYSA-M 0.000 description 2
- QDRKDTQENPPHOJ-UHFFFAOYSA-N sodium ethoxide Chemical compound [Na+].CC[O-] QDRKDTQENPPHOJ-UHFFFAOYSA-N 0.000 description 2
- 239000012312 sodium hydride Substances 0.000 description 2
- NESLWCLHZZISNB-UHFFFAOYSA-M sodium phenolate Chemical compound [Na+].[O-]C1=CC=CC=C1 NESLWCLHZZISNB-UHFFFAOYSA-M 0.000 description 2
- GEHJYWRUCIMESM-UHFFFAOYSA-L sodium sulfite Chemical compound [Na+].[Na+].[O-]S([O-])=O GEHJYWRUCIMESM-UHFFFAOYSA-L 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000007858 starting material Substances 0.000 description 2
- 238000007920 subcutaneous administration Methods 0.000 description 2
- 125000004434 sulfur atom Chemical group 0.000 description 2
- 238000001308 synthesis method Methods 0.000 description 2
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 2
- 238000002411 thermogravimetry Methods 0.000 description 2
- FYSNRJHAOHDILO-UHFFFAOYSA-N thionyl chloride Chemical compound ClS(Cl)=O FYSNRJHAOHDILO-UHFFFAOYSA-N 0.000 description 2
- 238000012546 transfer Methods 0.000 description 2
- SNICXCGAKADSCV-JTQLQIEISA-N (-)-Nicotine Chemical compound CN1CCC[C@H]1C1=CC=CN=C1 SNICXCGAKADSCV-JTQLQIEISA-N 0.000 description 1
- HXKKHQJGJAFBHI-GSVOUGTGSA-N (2R)-1-aminopropan-2-ol Chemical compound C[C@@H](O)CN HXKKHQJGJAFBHI-GSVOUGTGSA-N 0.000 description 1
- HXKKHQJGJAFBHI-VKHMYHEASA-N (2s)-1-aminopropan-2-ol Chemical compound C[C@H](O)CN HXKKHQJGJAFBHI-VKHMYHEASA-N 0.000 description 1
- 125000004455 (C1-C3) alkylthio group Chemical group 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- YBYIRNPNPLQARY-UHFFFAOYSA-N 1H-indene Natural products C1=CC=C2CC=CC2=C1 YBYIRNPNPLQARY-UHFFFAOYSA-N 0.000 description 1
- WCXFPLXZZSWROM-UHFFFAOYSA-N 1h-pyrazolo[4,3-c]pyridine Chemical compound C1=NC=C2C=NNC2=C1 WCXFPLXZZSWROM-UHFFFAOYSA-N 0.000 description 1
- KJJPLEZQSCZCKE-UHFFFAOYSA-N 2-aminopropane-1,3-diol Chemical compound OCC(N)CO KJJPLEZQSCZCKE-UHFFFAOYSA-N 0.000 description 1
- JLUZCHOYSPEHES-UHFFFAOYSA-N 3-aminocyclobutan-1-ol Chemical compound NC1CC(O)C1 JLUZCHOYSPEHES-UHFFFAOYSA-N 0.000 description 1
- GZPHSAQLYPIAIN-UHFFFAOYSA-N 3-pyridinecarbonitrile Chemical compound N#CC1=CC=CN=C1 GZPHSAQLYPIAIN-UHFFFAOYSA-N 0.000 description 1
- YIQXUVPRCWNHPP-UHFFFAOYSA-N 6-methylpyridine-2-carboxamide Chemical compound CC1=CC=CC(C(N)=O)=N1 YIQXUVPRCWNHPP-UHFFFAOYSA-N 0.000 description 1
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical compound [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 1
- 108010059616 Activins Proteins 0.000 description 1
- 102000007350 Bone Morphogenetic Proteins Human genes 0.000 description 1
- 108010007726 Bone Morphogenetic Proteins Proteins 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical compound [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- OJRUSAPKCPIVBY-KQYNXXCUSA-N C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N Chemical compound C1=NC2=C(N=C(N=C2N1[C@H]3[C@@H]([C@@H]([C@H](O3)COP(=O)(CP(=O)(O)O)O)O)O)I)N OJRUSAPKCPIVBY-KQYNXXCUSA-N 0.000 description 1
- JGLMVXWAHNTPRF-CMDGGOBGSA-N CCN1N=C(C)C=C1C(=O)NC1=NC2=CC(=CC(OC)=C2N1C\C=C\CN1C(NC(=O)C2=CC(C)=NN2CC)=NC2=CC(=CC(OCCCN3CCOCC3)=C12)C(N)=O)C(N)=O Chemical compound CCN1N=C(C)C=C1C(=O)NC1=NC2=CC(=CC(OC)=C2N1C\C=C\CN1C(NC(=O)C2=CC(C)=NN2CC)=NC2=CC(=CC(OCCCN3CCOCC3)=C12)C(N)=O)C(N)=O JGLMVXWAHNTPRF-CMDGGOBGSA-N 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 206010008342 Cervix carcinoma Diseases 0.000 description 1
- ZAMOUSCENKQFHK-UHFFFAOYSA-N Chlorine atom Chemical compound [Cl] ZAMOUSCENKQFHK-UHFFFAOYSA-N 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- 241000938605 Crocodylia Species 0.000 description 1
- 102000002004 Cytochrome P-450 Enzyme System Human genes 0.000 description 1
- 108010015742 Cytochrome P-450 Enzyme System Proteins 0.000 description 1
- 102000004127 Cytokines Human genes 0.000 description 1
- 108090000695 Cytokines Proteins 0.000 description 1
- GSNUFIFRDBKVIE-UHFFFAOYSA-N DMF Natural products CC1=CC=C(C)O1 GSNUFIFRDBKVIE-UHFFFAOYSA-N 0.000 description 1
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical class [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 1
- 206010013710 Drug interaction Diseases 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 101001059454 Homo sapiens Serine/threonine-protein kinase MARK2 Proteins 0.000 description 1
- 206010020751 Hypersensitivity Diseases 0.000 description 1
- 206010062016 Immunosuppression Diseases 0.000 description 1
- 102100026818 Inhibin beta E chain Human genes 0.000 description 1
- 102000002746 Inhibins Human genes 0.000 description 1
- 108010004250 Inhibins Proteins 0.000 description 1
- 108060001084 Luciferase Proteins 0.000 description 1
- 239000005089 Luciferase Substances 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- 102100025725 Mothers against decapentaplegic homolog 4 Human genes 0.000 description 1
- 101710143112 Mothers against decapentaplegic homolog 4 Proteins 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- 206010061902 Pancreatic neoplasm Diseases 0.000 description 1
- 241001494479 Pecora Species 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 238000001069 Raman spectroscopy Methods 0.000 description 1
- 229940124639 Selective inhibitor Drugs 0.000 description 1
- 102100028904 Serine/threonine-protein kinase MARK2 Human genes 0.000 description 1
- 102000057208 Smad2 Human genes 0.000 description 1
- 108700032504 Smad2 Proteins 0.000 description 1
- 102000049939 Smad3 Human genes 0.000 description 1
- 108700031297 Smad3 Proteins 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 208000005718 Stomach Neoplasms Diseases 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 102000046299 Transforming Growth Factor beta1 Human genes 0.000 description 1
- 102000011117 Transforming Growth Factor beta2 Human genes 0.000 description 1
- 101800002279 Transforming growth factor beta-1 Proteins 0.000 description 1
- 101800000304 Transforming growth factor beta-2 Proteins 0.000 description 1
- 102000056172 Transforming growth factor beta-3 Human genes 0.000 description 1
- 108090000097 Transforming growth factor beta-3 Proteins 0.000 description 1
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical class [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 1
- 208000006105 Uterine Cervical Neoplasms Diseases 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- ABRVLXLNVJHDRQ-UHFFFAOYSA-N [2-pyridin-3-yl-6-(trifluoromethyl)pyridin-4-yl]methanamine Chemical compound FC(C1=CC(=CC(=N1)C=1C=NC=CC=1)CN)(F)F ABRVLXLNVJHDRQ-UHFFFAOYSA-N 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 239000002253 acid Substances 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 230000004913 activation Effects 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000008186 active pharmaceutical agent Substances 0.000 description 1
- 239000000488 activin Substances 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000001931 aliphatic group Chemical group 0.000 description 1
- 125000003282 alkyl amino group Chemical group 0.000 description 1
- 150000001350 alkyl halides Chemical group 0.000 description 1
- 230000033115 angiogenesis Effects 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- 239000008346 aqueous phase Substances 0.000 description 1
- 125000000477 aza group Chemical group 0.000 description 1
- GMWFCJXSQQHBPI-UHFFFAOYSA-N azetidin-3-ol Chemical compound OC1CNC1 GMWFCJXSQQHBPI-UHFFFAOYSA-N 0.000 description 1
- 125000003828 azulenyl group Chemical group 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 229940112869 bone morphogenetic protein Drugs 0.000 description 1
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Substances BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- 230000024245 cell differentiation Effects 0.000 description 1
- 230000012292 cell migration Effects 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 230000036755 cellular response Effects 0.000 description 1
- 201000010881 cervical cancer Diseases 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000004587 chromatography analysis Methods 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 229940125758 compound 15 Drugs 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 235000009508 confectionery Nutrition 0.000 description 1
- 210000002808 connective tissue Anatomy 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 125000006547 cyclononyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000000640 cyclooctyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000004855 decalinyl group Chemical group C1(CCCC2CCCCC12)* 0.000 description 1
- 230000003111 delayed effect Effects 0.000 description 1
- 238000004807 desolvation Methods 0.000 description 1
- 238000000113 differential scanning calorimetry Methods 0.000 description 1
- 238000009792 diffusion process Methods 0.000 description 1
- 150000004683 dihydrates Chemical class 0.000 description 1
- 230000006806 disease prevention Effects 0.000 description 1
- 125000005883 dithianyl group Chemical group 0.000 description 1
- 230000008482 dysregulation Effects 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 230000001804 emulsifying effect Effects 0.000 description 1
- 238000003821 enantio-separation Methods 0.000 description 1
- XHTRNTBSWKGZOE-UHFFFAOYSA-N ethyl 2-formyl-3-methylbutanoate Chemical compound CCOC(=O)C(C=O)C(C)C XHTRNTBSWKGZOE-UHFFFAOYSA-N 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000003176 fibrotic effect Effects 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000000796 flavoring agent Substances 0.000 description 1
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000013355 food flavoring agent Nutrition 0.000 description 1
- 235000003599 food sweetener Nutrition 0.000 description 1
- 125000002541 furyl group Chemical group 0.000 description 1
- 206010017758 gastric cancer Diseases 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 125000003707 hexyloxy group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])O* 0.000 description 1
- 150000004677 hydrates Chemical class 0.000 description 1
- 125000002768 hydroxyalkyl group Chemical group 0.000 description 1
- 206010020718 hyperplasia Diseases 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- 230000001506 immunosuppresive effect Effects 0.000 description 1
- 125000003454 indenyl group Chemical group C1(C=CC2=CC=CC=C12)* 0.000 description 1
- 239000000893 inhibin Substances 0.000 description 1
- ZPNFWUPYTFPOJU-LPYSRVMUSA-N iniprol Chemical compound C([C@H]1C(=O)NCC(=O)NCC(=O)N[C@H]2CSSC[C@H]3C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(N[C@H](C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC=4C=CC=CC=4)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC=4C=CC=CC=4)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC2=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H]2N(CCC2)C(=O)[C@@H](N)CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N2[C@@H](CCC2)C(=O)N2[C@@H](CCC2)C(=O)N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N2[C@@H](CCC2)C(=O)N3)C(=O)NCC(=O)NCC(=O)N[C@@H](C)C(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@H](C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H](C(=O)N1)C(C)C)[C@@H](C)O)[C@@H](C)CC)=O)[C@@H](C)CC)C1=CC=C(O)C=C1 ZPNFWUPYTFPOJU-LPYSRVMUSA-N 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 238000007917 intracranial administration Methods 0.000 description 1
- 238000007912 intraperitoneal administration Methods 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 239000011630 iodine Substances 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 125000001786 isothiazolyl group Chemical group 0.000 description 1
- 125000000842 isoxazolyl group Chemical group 0.000 description 1
- 231100001231 less toxic Toxicity 0.000 description 1
- 210000004185 liver Anatomy 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000007937 lozenge Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 210000004072 lung Anatomy 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- 210000004324 lymphatic system Anatomy 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 208000015486 malignant pancreatic neoplasm Diseases 0.000 description 1
- 238000000048 melt cooling Methods 0.000 description 1
- 238000013508 migration Methods 0.000 description 1
- 150000007522 mineralic acids Chemical class 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 150000004682 monohydrates Chemical class 0.000 description 1
- 125000002757 morpholinyl group Chemical group 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 1
- IJGRMHOSHXDMSA-UHFFFAOYSA-N nitrogen Substances N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 1
- 239000000346 nonvolatile oil Substances 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 230000002611 ovarian Effects 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 201000002528 pancreatic cancer Diseases 0.000 description 1
- 208000008443 pancreatic carcinoma Diseases 0.000 description 1
- 230000036961 partial effect Effects 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 230000008506 pathogenesis Effects 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 125000005561 phenanthryl group Chemical group 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 235000015320 potassium carbonate Nutrition 0.000 description 1
- 229910000160 potassium phosphate Inorganic materials 0.000 description 1
- 235000011009 potassium phosphates Nutrition 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000002062 proliferating effect Effects 0.000 description 1
- 210000002307 prostate Anatomy 0.000 description 1
- 102000004169 proteins and genes Human genes 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000001725 pyrenyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 125000000168 pyrrolyl group Chemical group 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 238000013341 scale-up Methods 0.000 description 1
- 238000004626 scanning electron microscopy Methods 0.000 description 1
- 208000012201 sexual and gender identity disease Diseases 0.000 description 1
- 208000015891 sexual disease Diseases 0.000 description 1
- 238000010583 slow cooling Methods 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- KKCBUQHMOMHUOY-UHFFFAOYSA-N sodium oxide Chemical compound [O-2].[Na+].[Na+] KKCBUQHMOMHUOY-UHFFFAOYSA-N 0.000 description 1
- 229910001948 sodium oxide Inorganic materials 0.000 description 1
- 235000010265 sodium sulphite Nutrition 0.000 description 1
- SUBJHSREKVAVAR-UHFFFAOYSA-N sodium;methanol;methanolate Chemical compound [Na+].OC.[O-]C SUBJHSREKVAVAR-UHFFFAOYSA-N 0.000 description 1
- 125000003003 spiro group Chemical group 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 201000011549 stomach cancer Diseases 0.000 description 1
- 238000000859 sublimation Methods 0.000 description 1
- 230000008022 sublimation Effects 0.000 description 1
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 description 1
- 150000003460 sulfonic acids Chemical class 0.000 description 1
- 239000000829 suppository Substances 0.000 description 1
- 239000000375 suspending agent Substances 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000013268 sustained release Methods 0.000 description 1
- 239000012730 sustained-release form Substances 0.000 description 1
- 239000003765 sweetening agent Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 238000003419 tautomerization reaction Methods 0.000 description 1
- 150000003512 tertiary amines Chemical class 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- 125000001412 tetrahydropyranyl group Chemical group 0.000 description 1
- NQRYJNQNLNOLGT-UHFFFAOYSA-N tetrahydropyridine hydrochloride Natural products C1CCNCC1 NQRYJNQNLNOLGT-UHFFFAOYSA-N 0.000 description 1
- CZDYPVPMEAXLPK-UHFFFAOYSA-N tetramethylsilane Chemical group C[Si](C)(C)C CZDYPVPMEAXLPK-UHFFFAOYSA-N 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 125000001113 thiadiazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- XSKZXGDFSCCXQX-UHFFFAOYSA-N thiencarbazone-methyl Chemical compound COC(=O)C1=CSC(C)=C1S(=O)(=O)NC(=O)N1C(=O)N(C)C(OC)=N1 XSKZXGDFSCCXQX-UHFFFAOYSA-N 0.000 description 1
- 125000001544 thienyl group Chemical group 0.000 description 1
- 125000004568 thiomorpholinyl group Chemical group 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 238000013518 transcription Methods 0.000 description 1
- 230000035897 transcription Effects 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 125000004306 triazinyl group Chemical group 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-M triflate Chemical compound [O-]S(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-M 0.000 description 1
- 150000004684 trihydrates Chemical class 0.000 description 1
- 229910000404 tripotassium phosphate Inorganic materials 0.000 description 1
- 235000019798 tripotassium phosphate Nutrition 0.000 description 1
- 125000005455 trithianyl group Chemical group 0.000 description 1
- 210000004881 tumor cell Anatomy 0.000 description 1
- 239000012808 vapor phase Substances 0.000 description 1
- 239000003643 water by type Substances 0.000 description 1
- 230000003442 weekly effect Effects 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
技术领域technical field
本发明属于药物化学领域,涉及一种具有TGFβR1抑制活性的杂环类化合物、包含其的药物组合物,其制备方法及其用途。The invention belongs to the field of medicinal chemistry, and relates to a heterocyclic compound with TGFβR1 inhibitory activity, a pharmaceutical composition comprising the same, a preparation method and use thereof.
背景技术Background technique
转化生长因子-β(transforming growth factor-β,TGF-β)是一种多功能细胞因子,其调节各种细胞应答,例如细胞增殖、分化、迁移和凋亡。TGF-β超家族包括TGF-β1、TGF-β2、TGF-β3、活化素、抑制素、骨形态发生蛋白等。TGF-β通过两种高保守性单跨膜丝氨酸/苏氨酸激酶TGFβR1和TGFβR2进行信号转导(ACS Med.Chem.Lett.,2018,9,1117)。Transforming growth factor-β (TGF-β) is a multifunctional cytokine that regulates various cellular responses such as cell proliferation, differentiation, migration and apoptosis. The TGF-β superfamily includes TGF-β1, TGF-β2, TGF-β3, activin, inhibin, bone morphogenetic protein, and the like. TGF-β signals through two highly conserved single-transmembrane serine/threonine kinases, TGFβR1 and TGFβR2 (ACS Med. Chem. Lett., 2018, 9, 1117).
Smads是细胞内重要的TGF-β信号转导和调节分子,可以将TGF-β信号直接由细胞膜转导至细胞核内,TGF-β/Smads信号通路在肿瘤的发生和发展中起到重要的作用。在TGF-β/Smads信号转导中,活化的TGF-β首先与细胞膜表面的TGFβR2结合,形成异源二聚体复合物,并进一步被TGFβR1识别和结合。活化的TGFβR1进一步磷酸化Smad2/Smad3蛋白,后者再进一步与Smad4结合,以形成异源三聚体复合物,这一复合物进入细胞核内与辅助活化/抑制因子协同作用来调节靶基因的转录(Nature,2003,425,577)。只要TGF-β/Smads信号通路的任一环节发生改变,都会导致信号转导通路的异常(PNAS,2019,116,9166)。Smads are important TGF-β signal transduction and regulation molecules in cells, which can directly transduce TGF-β signals from the cell membrane to the nucleus. The TGF-β/Smads signaling pathway plays an important role in the occurrence and development of tumors . In TGF-β/Smads signal transduction, activated TGF-β first binds to TGFβR2 on the cell membrane surface to form a heterodimeric complex, which is further recognized and bound by TGFβR1. Activated TGFβR1 further phosphorylates Smad2/Smad3 protein, which further binds to Smad4 to form a heterotrimeric complex, which enters the nucleus and cooperates with co-activators/repressors to regulate the transcription of target genes (Nature, 2003, 425, 577). As long as any part of the TGF-β/Smads signaling pathway is altered, it will lead to abnormalities in the signal transduction pathway (PNAS, 2019, 116, 9166).
TGF-β信号通路在包括癌症在内的许多疾病中失调,胃癌、结肠直肠癌、前列腺癌、卵巢癌、胰腺癌、肝癌、肺癌、宫颈癌和头颈部癌细胞系和肿瘤组织中的TGFβR1蛋白水平显著升高。TGF-β信号通路的活化在肿瘤基质中引发明显的病理效应,包括免疫抑制、血管再生和结缔组织增生。此外,TGF-β信号通路能够增强肿瘤细胞的侵袭力,促进上皮细胞向间质转化以及提高对肿瘤上皮细胞治疗的耐受度(Nat.Neurosci.,2014,17,943)。The TGF-β signaling pathway is dysregulated in many diseases including cancer, TGFβR1 in gastric, colorectal, prostate, ovarian, pancreatic, liver, lung, cervical and head and neck cancer cell lines and tumor tissues Protein levels were significantly elevated. Activation of the TGF-β signaling pathway triggers pronounced pathological effects in the tumor stroma, including immunosuppression, angiogenesis, and connective tissue hyperplasia. In addition, the TGF-β signaling pathway can enhance the invasiveness of tumor cells, promote the transition of epithelial cells to mesenchyme, and improve the tolerance of tumor epithelial cells (Nat. Neurosci., 2014, 17, 943).
目前,针对TGF-β信号通路中关键靶点TGFβR1的抑制剂的开发在医药工业界已逐渐得到重视,已公开的专利申请包括WO 02/094833 A1、WO 2009/150547 A1、WO 2017/035118 A1、WO 2018/019106 A1等。但本领域仍然亟需新的TGFβR1抑制剂,特别是具有高活性、高选择性的TGFβR1抑制剂。At present, the development of inhibitors targeting TGFβR1, a key target in the TGF-β signaling pathway, has gradually attracted attention in the pharmaceutical industry, and published patent applications include WO 02/094833 A1, WO 2009/150547 A1, and WO 2017/035118 A1 , WO 2018/019106 A1, etc. However, there is still an urgent need in the art for new TGFβR1 inhibitors, especially TGFβR1 inhibitors with high activity and selectivity.
发明内容SUMMARY OF THE INVENTION
本发明通过大量的研究,惊喜地发现了一种杂环类化合物及其相应的制备方法。该类化合物可以显著抑制TGFβR1的活性,并且在TGFβR1和TGFβR2之间具有良好的选择性,其作为TGFβR1抑制剂可用于治疗至少部分由TGF-β信号通路介导的增殖性病症及细胞凋亡失调性病症,尤其是至少部分由TGFβR1介导的疾病,如癌症,例如肝癌。The present invention surprisingly finds a heterocyclic compound and its corresponding preparation method through a large number of researches. The compounds can significantly inhibit the activity of TGFβR1 and have good selectivity between TGFβR1 and TGFβR2, and can be used as TGFβR1 inhibitors for the treatment of proliferative disorders and dysregulation of apoptosis mediated at least in part by TGF-β signaling pathway Sexual disorders, especially diseases mediated at least in part by TGF[beta]Rl, such as cancer, eg liver cancer.
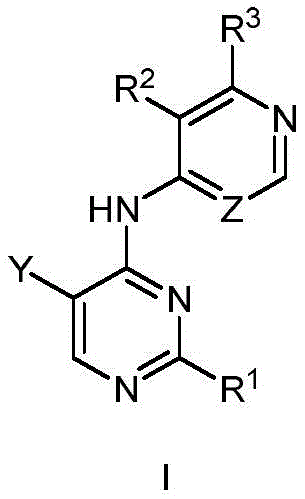
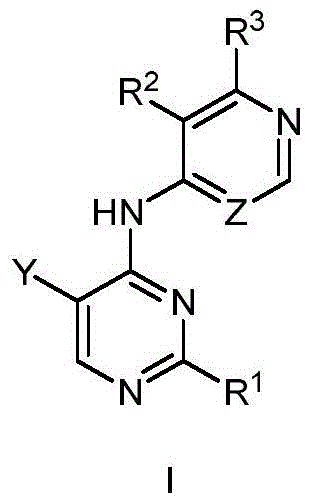
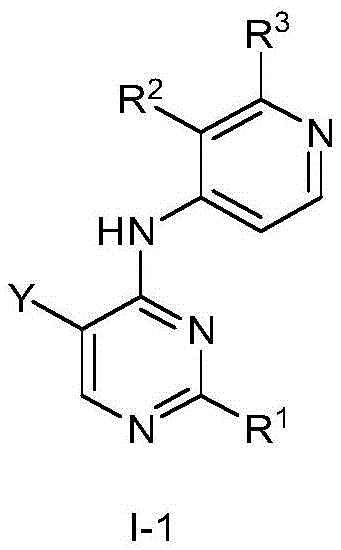
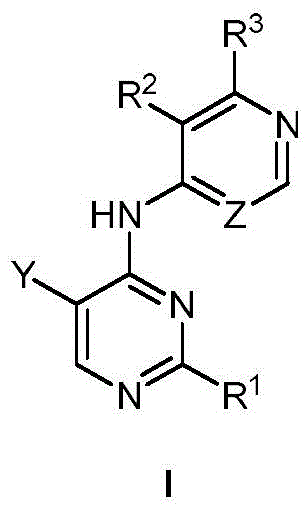
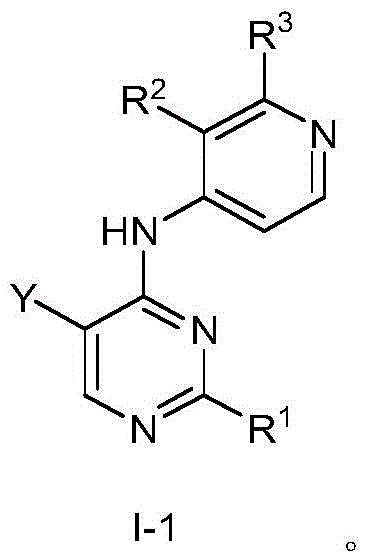
第一方面,本发明提供了一种具有式I结构的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药:In the first aspect, the present invention provides a compound having the structure of formula I or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitrogen oxide thereof , isotopic labels, metabolites or prodrugs:
其中,in,
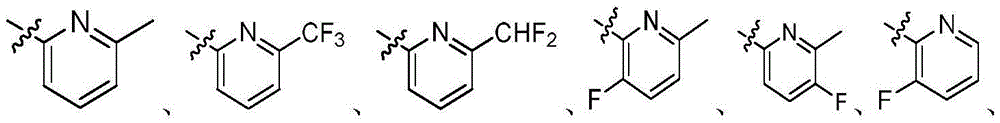
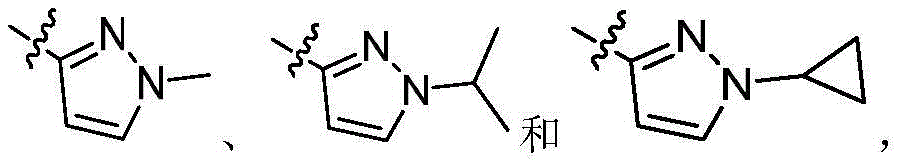
R1为5-10元杂芳基,所述5-10元杂芳基任选地被一或多个R4取代;R 1 is a 5-10-membered heteroaryl group optionally substituted with one or more R 4 ;
R2选自氢、卤素、C1-6烷基、C1-6卤代烷基、C3-8环烷基、氰基、-C(=O)NRaRb和-C(=O)ORa;R 2 is selected from hydrogen, halogen, C 1-6 alkyl, C 1-6 haloalkyl, C 3-8 cycloalkyl, cyano, -C(=O)NR a R b and -C(=O) OR a ;
Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-8环烷基和4-8元杂环基,所述C1-6烷基、C3-8环烷基或4-8元杂环基任选地被一或多个R5取代;或者Ra、Rb和与它们连接的氮原子一起形成4-8元杂环,所述4-8元杂环任选地被一或多个R5取代;R a and R b are each independently selected at each occurrence from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl and 4-8 membered heterocyclyl, the C 1-6 alkyl, C 3-8 cycloalkyl or 4-8 membered heterocyclyl optionally substituted with one or more R 5 ; or R a , R b and the nitrogen atom to which they are attached together form a 4-8 membered heterocycle, the The 4-8 membered heterocycle is optionally substituted with one or more R5;
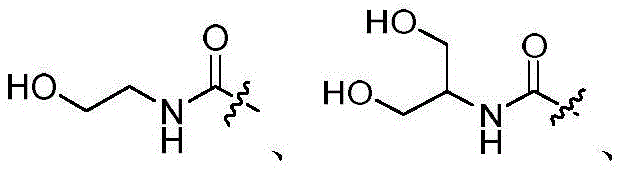
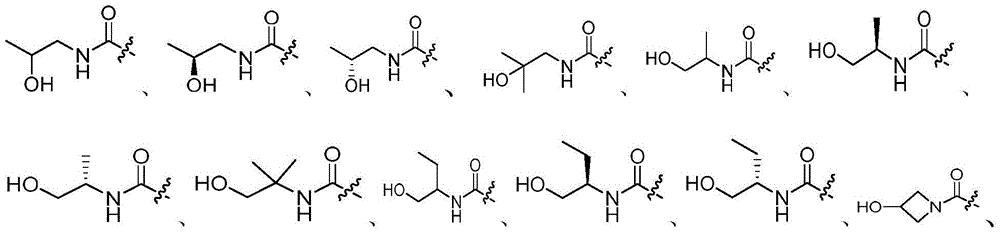
R3选自氢和-NRcRd;R 3 is selected from hydrogen and -NR c R d ;
Rc和Rd在每次出现时各自独立地选自氢、C1-6烷基、C3-8环烷基、4-8元杂环基、-C(=O)Ra、C6-10芳基和5-10元杂芳基,所述C1-6烷基、C3-8环烷基、4-8元杂环基、C6-10芳基或5-10元杂芳基任选地被一或多个R6取代;R c and R d at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl, 4-8 membered heterocyclyl, -C(=O)R a , C 6-10 -membered aryl and 5-10-membered heteroaryl, the C 1-6 alkyl, C 3-8 cycloalkyl, 4-8-membered heterocyclyl, C 6-10 -membered aryl or 5-10-membered Heteroaryl is optionally substituted with one or more R ;
R4在每次出现时各自独立地选自氘、卤素、C1-6烷基、C1-6烷氧基和C3-8环烷基,所述C1-6烷基、C1-6烷氧基或C3-8环烷基任选地被一或多个卤素取代;R 4 at each occurrence is independently selected from deuterium, halogen, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, the C 1-6 alkyl, C 1-6 alkyl -6 alkoxy or C 3-8 cycloalkyl optionally substituted with one or more halogens;
R5在每次出现时各自独立地选自卤素、羟基、C1-6烷基、C1-6烷氧基、氨基、C1-6烷基氨基、C3-8环烷基和4-8元杂环基,所述C1-6烷基或C3-8环烷基任选地被一或多个羟基取代;R 5 at each occurrence is independently selected from halogen, hydroxy, C 1-6 alkyl, C 1-6 alkoxy, amino, C 1-6 alkylamino, C 3-8 cycloalkyl and 4 -8-membered heterocyclyl, the C 1-6 alkyl or C 3-8 cycloalkyl is optionally substituted with one or more hydroxyl groups;
R6在每次出现时各自独立地选自卤素、羟基、C1-6烷基、C1-6烷氧基、氨基、C1-6烷基氨基、C3-8环烷基和4-8元杂环基,所述C1-6烷基、C1-6烷氧基、C1-6烷基氨基、C3-8环烷基或4-8元杂环基任选地被一或多个选自羟基和C1-6烷基的基团取代;R 6 at each occurrence is independently selected from halogen, hydroxy, C 1-6 alkyl, C 1-6 alkoxy, amino, C 1-6 alkylamino, C 3-8 cycloalkyl and 4 -8-membered heterocyclyl, the C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, C 3-8 cycloalkyl or 4-8 membered heterocyclyl are optionally substituted with one or more groups selected from hydroxy and C 1-6 alkyl;
Y选自氢、C1-6烷基、C3-8环烷基、C1-6卤代烷基、C1-6烷氧基、C3-8环烷氧基、C1-6烷硫基、C3-8环烷硫基、C1-6烷基氨基、C3-8环烷基氨基和4-8元杂环基;Y is selected from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl, C 1-6 haloalkyl, C 1-6 alkoxy, C 3-8 cycloalkoxy, C 1-6 alkylthio base, C 3-8 cycloalkylthio, C 1-6 alkylamino, C 3-8 cycloalkylamino and 4-8 membered heterocyclyl;
Z选自-CR7-和-N-;且Z is selected from -CR7- and -N-; and
R7选自氢、卤素、C1-6烷基、C3-8环烷基、C1-6卤代烷基和氰基。R 7 is selected from hydrogen, halogen, C 1-6 alkyl, C 3-8 cycloalkyl, C 1-6 haloalkyl and cyano.
第二方面,本发明提供了一种药物组合物,其包含至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药,以及一种或多种药学上可接受的载体。In a second aspect, the present invention provides a pharmaceutical composition comprising at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, A solvate, nitrogen oxide, isotopic label, metabolite or prodrug, and one or more pharmaceutically acceptable carriers.
第三方面,本发明提供了一种药盒,其包含:A third aspect, the present invention provides a kind of medicine box, it comprises:
a)作为第一治疗剂的至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药,或者作为第一药物组合物的本发明的药物组合物;a) at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitroxide, an isotopic label, metabolite or prodrug, or a pharmaceutical composition of the invention as the first pharmaceutical composition;
b)任选存在的作为第二治疗剂的至少一种其他治疗剂,或者作为第二药物组合物的包含其他治疗剂的药物组合物;和b) optionally present at least one other therapeutic agent as a second therapeutic agent, or a pharmaceutical composition comprising the other therapeutic agent as a second pharmaceutical composition; and
c)任选存在的包装和/或说明书。c) Optional packaging and/or instructions.
第四方面,本发明提供了本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物,其用作TGFβR1抑制剂,用于预防和/或治疗至少部分由TGFβR1介导的疾病或病症(尤其是癌症,例如肝癌)。In a fourth aspect, the present invention provides compounds of the present invention or pharmaceutically acceptable salts, esters, stereoisomers, tautomers, polymorphs, solvates, nitrogen oxides, isotopic labels thereof , metabolites or prodrugs or pharmaceutical compositions of the invention for use as TGFβR1 inhibitors for the prevention and/or treatment of diseases or disorders (especially cancers, eg liver cancer) mediated at least in part by TGFβR1.
第五方面,本发明提供了本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物用作TGFβR1抑制剂的用途。In a fifth aspect, the present invention provides compounds of the present invention or pharmaceutically acceptable salts, esters, stereoisomers, tautomers, polymorphs, solvates, nitrogen oxides, isotopic labels thereof Use of a , metabolite or prodrug or a pharmaceutical composition of the invention as an inhibitor of TGFβR1.
第六方面,本发明提供了本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物在制备用于预防和/或治疗至少部分由TGFβR1介导的疾病或病症(尤其是癌症,例如肝癌)的药物中的用途。In a sixth aspect, the present invention provides compounds of the present invention or pharmaceutically acceptable salts, esters, stereoisomers, tautomers, polymorphs, solvates, nitrogen oxides, isotopic labels thereof , metabolite or prodrug or use of a pharmaceutical composition of the invention in the manufacture of a medicament for the prevention and/or treatment of a disease or disorder mediated at least in part by TGFβR1, especially cancer, eg liver cancer.
第七方面,本发明提供了一种用于预防和/或治疗至少部分由TGFβR1介导的疾病或病症(尤其是癌症,例如肝癌)的方法,其包括向有此需要的个体给药预防和/或治疗有效量的本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物。In a seventh aspect, the present invention provides a method for the prevention and/or treatment of a disease or disorder mediated at least in part by TGFβR1, in particular cancer, such as liver cancer, comprising administering to an individual in need thereof the prophylactic and A therapeutically effective amount of a compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitrogen oxide, isotopic label, metabolite drug or prodrug or pharmaceutical composition of the present invention.
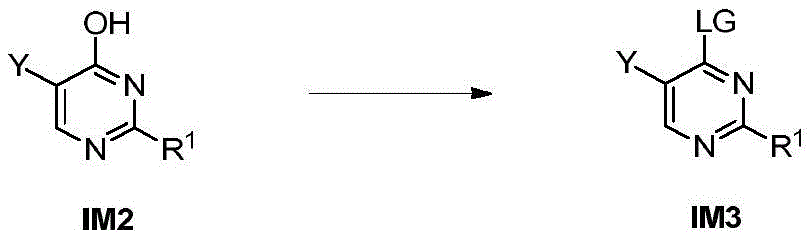
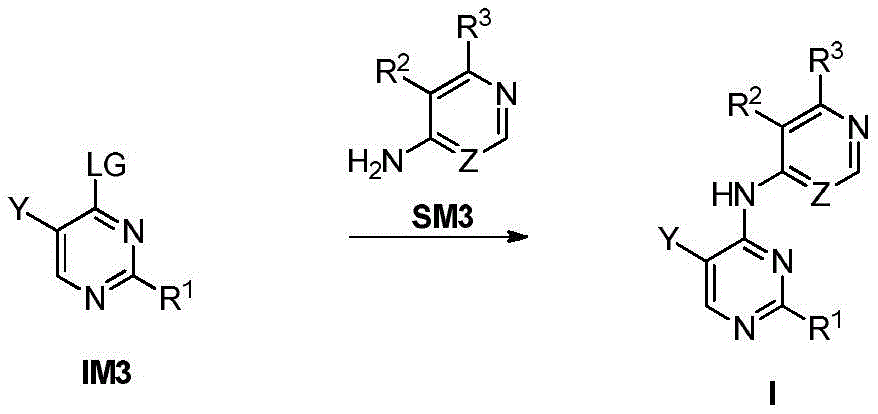
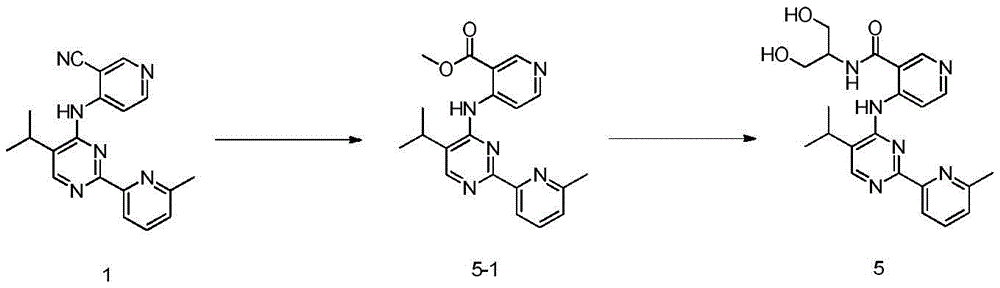
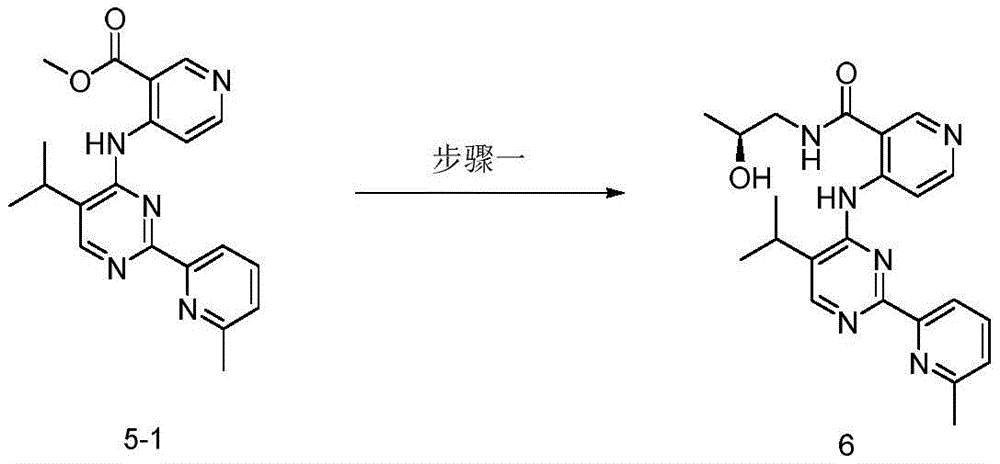
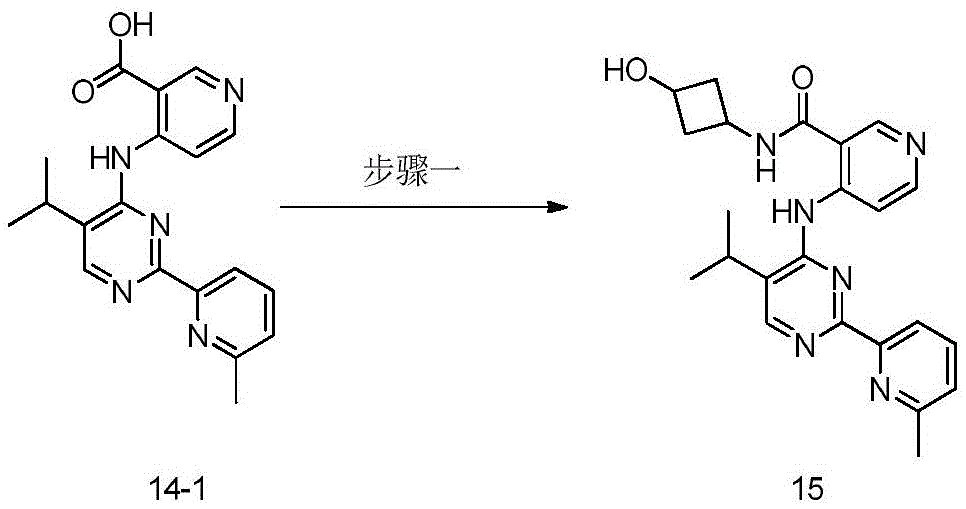
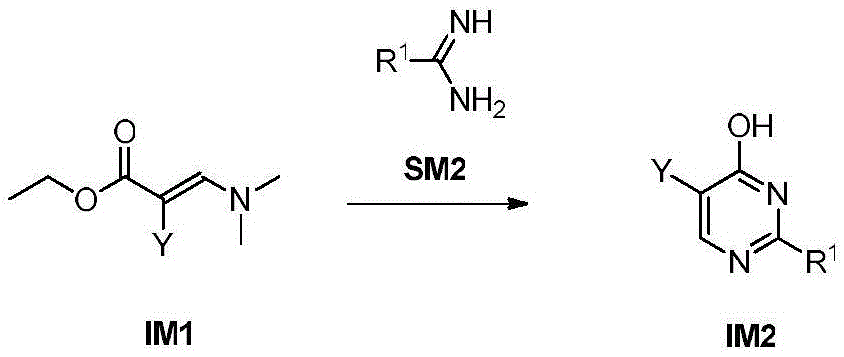
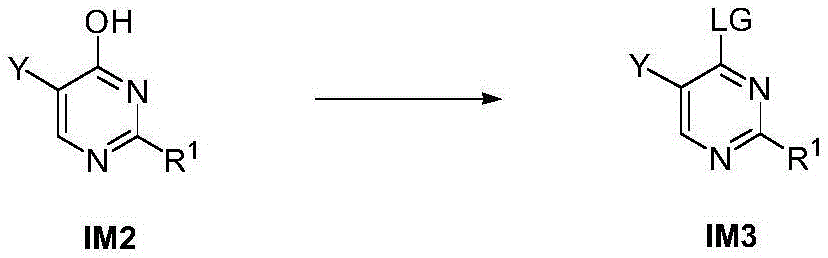
第八方面,本发明提供了制备本发明的化合物的方法,其包括:In an eighth aspect, the present invention provides a method for preparing the compound of the present invention, comprising:
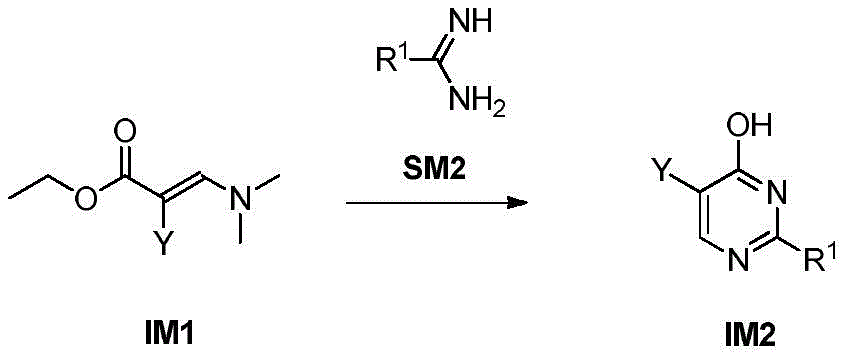
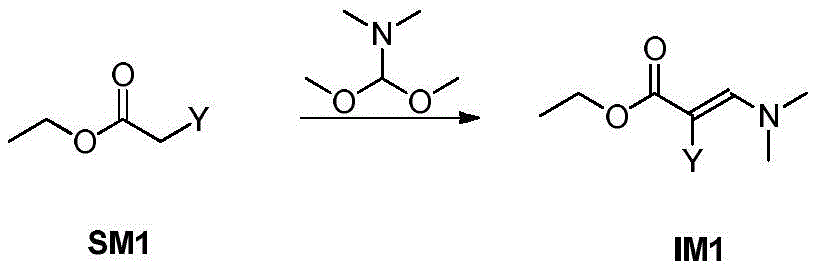
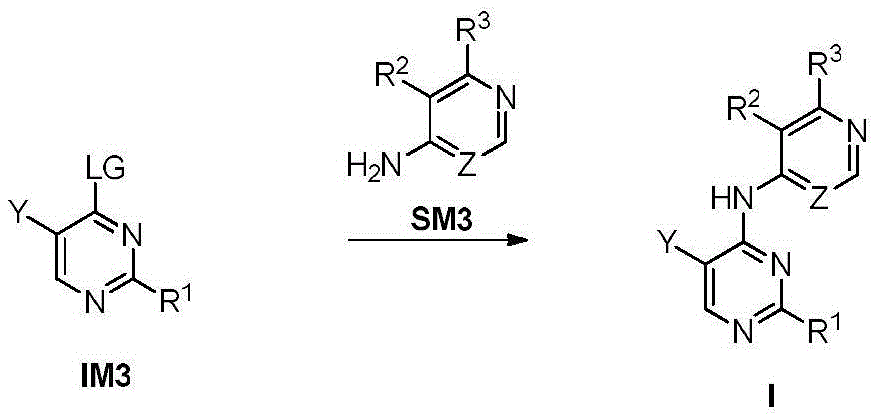
步骤1-1:化合物SM1和N,N-二甲基甲酰胺二甲基缩醛发生缩合反应得到化合物IM1Step 1-1: Compound SM1 and N,N-dimethylformamide dimethyl acetal undergo condensation reaction to obtain compound IM1
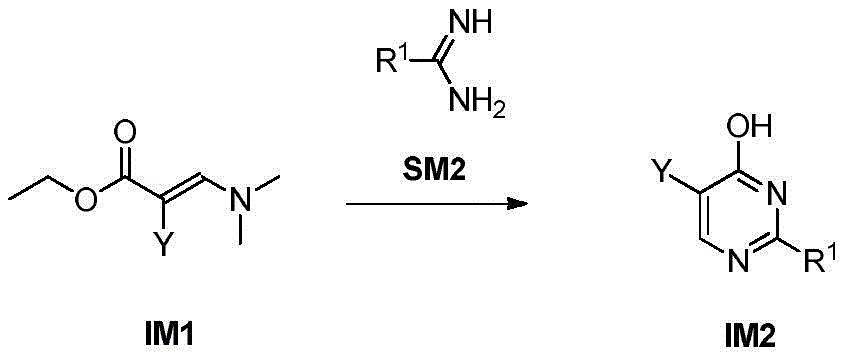
步骤1-2:化合物IM1和化合物SM2发生环化反应得到化合物IM2Step 1-2: Compound IM1 and compound SM2 undergo cyclization reaction to obtain compound IM2
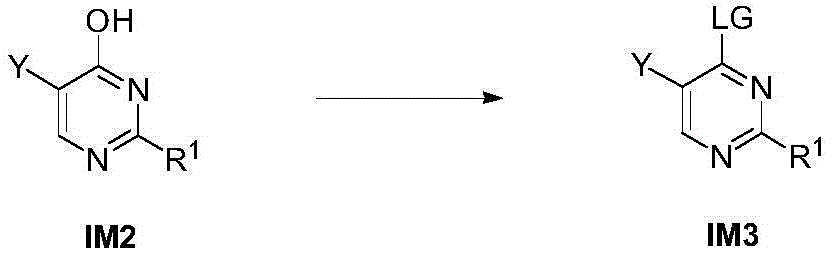
步骤1-3:化合物IM2发生取代反应得到化合物IM3Step 1-3: Compound IM2 undergoes substitution reaction to obtain compound IM3
步骤1-4:化合物IM3和SM3发生取代反应得到式I化合物Step 1-4: Substitution reaction of compound IM3 and SM3 to obtain compound of formula I
其中,LG为离去基团,例如苯氧基和卤素;并且,where LG is a leaving group such as phenoxy and halogen; and,
Y、Z、R1、R2和R3如上文所定义。Y, Z, R 1 , R 2 and R 3 are as defined above.
本发明的化合物可以作为高效且高选择性的TGFβR1抑制剂,具有抗肿瘤活性,且毒副作用和药物相互作用较少,合成方法温和,操作简单易行,适合工业放大量生产。The compound of the present invention can be used as an efficient and highly selective TGFβR1 inhibitor, has antitumor activity, less toxic side effects and drug interactions, mild synthesis method, simple and easy operation, and is suitable for industrial scale-up production.
具体实施方式Detailed ways
在进一步描述本发明之前,应当理解,本发明不限于文中所述的特定实施方案;还应该理解,文中所使用的术语仅用于描述而非限制特定实施方案。Before the present invention is further described, it is to be understood that this invention is not limited to the specific embodiments described herein; it is also to be understood that the terminology used herein is for the purpose of describing and not limiting the specific embodiments.
定义definition
除非在下文中另有定义,本文中所用的所有技术术语和科学术语的含义意图与本领域技术人员通常所理解的相同。提及本文中使用的技术意图指在本领域中通常所理解的技术,包括那些对本领域技术人员显而易见的技术的变化或等效技术的替换。虽然相信以下术语对于本领域技术人员很好理解,但仍然阐述以下定义以更好地解释本发明。Unless otherwise defined below, all technical and scientific terms used herein are intended to have the same meaning as commonly understood by one of ordinary skill in the art. References to techniques used herein are intended to refer to techniques commonly understood in the art, including those variations or substitutions of equivalent techniques that would be apparent to those skilled in the art. While the following terms are believed to be well understood by those skilled in the art, the following definitions are set forth to better explain the present invention.
如本文中所使用,术语“包括”、“包含”、“具有”、“含有”或“涉及”及其在本文中的其它变体形式为包含性的(inclusive)或开放式的,且不排除其它未列举的元素或方法步骤。As used herein, the terms "comprising", "comprising", "having", "containing" or "involving" and other variations thereof herein are inclusive or open-ended, and do not Other unlisted elements or method steps are excluded.
如本文中所使用,术语“烷基”定义为直链或支链的饱和脂肪族烃基。例如,如本文中所使用,术语“C1-6烷基”指具有1至6个碳原子的直链或支链的基团(例如甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、正戊基或正己基),其任选地被一或多个(诸如1至3个)适合的取代基如卤素取代。As used herein, the term "alkyl" is defined as a straight or branched chain saturated aliphatic hydrocarbon group. For example, as used herein, the term "C 1-6 alkyl" refers to a straight or branched chain group having 1 to 6 carbon atoms (eg, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl or n-hexyl), which are optionally substituted with one or more (such as 1 to 3) suitable substituents such as halogen.
如本文中所使用,术语“环烷基”指饱和或部分不饱和的非芳族单环或多环(诸如双环)烃环(例如单环,诸如环丙基、环丁基、环戊基、环己基、环庚基、环辛基、环壬基,或双环,包括螺环、稠合或桥连系统,诸如双环[1.1.1]戊基、双环[2.2.1]庚基、双环[3.2.1]辛基或双环[5.2.0]壬基、十氢化萘基等),其任选地被一个或多个(诸如1至3个)适合的取代基取代。所述环烷基具有3至15个,例如3至10个碳原子、3至8个碳原子或3至6个碳原子。例如,如本文中所使用,术语“C3-8环烷基”指具有3至8个成环碳原子的饱和或不饱和的非芳族单环或多环(诸如双环)烃环(例如环丙基、环丁基、环戊基、环己基、环庚基),其任选地被一或多个(诸如1至3个)适合的取代基取代,例如甲基取代的环丙基。As used herein, the term "cycloalkyl" refers to a saturated or partially unsaturated non-aromatic monocyclic or polycyclic (such as bicyclic) hydrocarbon ring (eg, monocyclic, such as cyclopropyl, cyclobutyl, cyclopentyl) , cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, or bicyclic rings, including spirocyclic, fused or bridged systems such as bicyclo[1.1.1]pentyl, bicyclo[2.2.1]heptyl, bicyclo [3.2.1]octyl or bicyclo[5.2.0]nonyl, decalinyl, etc.), optionally substituted with one or more (such as 1 to 3) suitable substituents. The cycloalkyl group has 3 to 15, eg, 3 to 10 carbon atoms, 3 to 8 carbon atoms, or 3 to 6 carbon atoms. For example, as used herein, the term "C 3-8 cycloalkyl" refers to a saturated or unsaturated non-aromatic monocyclic or polycyclic (such as bicyclic) hydrocarbon ring having 3 to 8 ring carbon atoms (eg cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl) optionally substituted with one or more (such as 1 to 3) suitable substituents, for example methyl substituted cyclopropyl .
如本文中所使用,术语“烷氧基”是指通过氧原子连接至母体分子部分的如上文所定义的“烷基”,例如C1-6烷氧基、C1-3烷氧基。C1-6烷氧基的代表性实例包括但不限于甲氧基、乙氧基、正丙氧基、异丙氧基、正丁氧基、异丁氧基、叔丁氧基、戊氧基、己氧基等,所述烷氧基可以任选地被一或多个(诸如1至3个)相同或不同的取代基取代。As used herein, the term "alkoxy" refers to an "alkyl" as defined above attached to the parent molecular moiety through an oxygen atom, eg, C 1-6 alkoxy, C 1-3 alkoxy. Representative examples of C 1-6 alkoxy include, but are not limited to, methoxy, ethoxy, n-propoxy, isopropoxy, n-butoxy, isobutoxy, tert-butoxy, pentoxy alkoxy groups, hexyloxy groups, etc., the alkoxy groups may be optionally substituted with one or more (such as 1 to 3) substituents which may be the same or different.
如本文中所使用,术语“环烷氧基”是指通过氧原子连接至母体分子部分的如上文所定义的“环烷基”,例如C3-8环烷氧基、C3-6环烷氧基。C3-8环烷氧基的代表性实例包括但不限于环丙氧基、环丁氧基、环戊氧基、环己氧基等,所述的环烷氧基可以任选地被一或多个(诸如1至3个)相同或不同的取代基取代。As used herein, the term "cycloalkoxy" refers to a "cycloalkyl" as defined above attached to the parent molecular moiety through an oxygen atom, eg, C3-8 cycloalkoxy, C3-6 ring alkoxy. Representative examples of C 3-8 cycloalkoxy include, but are not limited to, cyclopropoxy, cyclobutoxy, cyclopentyloxy, cyclohexyloxy, etc., said cycloalkoxy may be optionally or multiple (such as 1 to 3) of the same or different substituents are substituted.
如本文中所使用,术语“烷硫基”是指通过硫原子连接至母体分子部分的如上文所定义的“烷基”,例如C1-6烷硫基、C1-3烷硫基。C1-6烷硫基的代表性实例包括但不限于甲硫基、乙硫基、正丙硫基、异丙硫基、正丁硫基、异丁硫基、叔丁硫基、戊硫基、己硫基等,所述烷硫基可以任选地被一或多个(诸如1至3个)相同或不同的取代基取代。As used herein, the term "alkylthio" refers to an "alkyl group" as defined above attached to the parent molecular moiety through a sulfur atom, eg, C1-6 alkylthio, C1-3 alkylthio. Representative examples of C 1-6 alkylthio include, but are not limited to, methylthio, ethylthio, n-propylthio, isopropylthio, n-butylthio, isobutylthio, tert-butylthio, pentylthio The alkylthio group may be optionally substituted with one or more, such as 1 to 3, the same or different substituents.
如本文中所使用,术语“环烷硫基”是指通过硫原子连接至母体分子部分的如上文所定义的“环烷基”,例如C3-8环烷硫基、C3-6环烷硫基。C3-8环烷硫基的代表性实例包括但不限于环丙硫基、环丁硫基、环戊硫基、环己硫基等,所述的环烷硫基可以任选地被一或多个(诸如1至3个)相同或不同的取代基取代。As used herein, the term "cycloalkylthio" refers to a "cycloalkyl" as defined above attached to the parent molecular moiety through a sulfur atom, eg, C3-8 cycloalkylthio, C3-6 ring Alkylthio. Representative examples of C 3-8 cycloalkylthio include, but are not limited to, cyclopropylthio, cyclobutylthio, cyclopentylthio, cyclohexylthio, etc., and the cycloalkylthio can be optionally or multiple (such as 1 to 3) of the same or different substituents are substituted.
如本文中所使用,术语“卤代”或“卤素”是指氟(F)、氯(Cl)、溴(Br)和碘(I)。As used herein, the term "halo" or "halogen" refers to fluorine (F), chlorine (Cl), bromine (Br) and iodine (I).
如本文中所使用,术语“卤代烷基”是指被一或多个(诸如1至3个)相同或不同的卤素原子取代的烷基。例如,本文中所使用的术语“C1-6卤代烷基”是指具有1至6个碳原子的卤代烷基,卤代烷基包括(但不限于)-CH2F、-CHF2、-CF3、-CH2CF3、-CF2CF3、-CH2CH2CF3、-CH2Cl等。本文中的卤代烷基任选地被一或多个(诸如1至3个)本文所描述的取代基取代。As used herein, the term "haloalkyl" refers to an alkyl group substituted with one or more, such as 1 to 3, the same or different halogen atoms. For example, the term "C 1-6 haloalkyl" as used herein refers to a haloalkyl having 1 to 6 carbon atoms, including but not limited to -CH 2 F, -CHF 2 , -CF 3 , -CH 2 CF 3 , -CF 2 CF 3 , -CH 2 CH 2 CF 3 , -CH 2 Cl and the like. The haloalkyl groups herein are optionally substituted with one or more (such as 1 to 3) substituents described herein.
如本文中所使用,术语“杂环基”指单环或多环基团,其在环中例如具有2、3、4、5、6、7、8、9个碳原子和一个或多个(例如1个、2个、3个或4个)选自C(=O)、O、S、S(=O)、S(=O)2、N和NR(R表示氢原子或取代基,例如但不限于烷基或环烷基)的基团。杂环基可以是饱和的或不饱和的。饱和杂环基可称为杂环烷基,例如3-8元杂环烷基、5-6元杂环烷基等。除非本说明书中另外特别指明,否则杂环基可以为单环、双环、三环或更多环的环体系,其可包括稠合环体系、并环体系、桥环体系或螺环体系。特别地,3-8元杂环基为在环中具有3-8个碳原子及杂原子的基团,例如,其具有4至8、4至7、4至6、5至8、5至7或者5至6个碳原子及杂原子(分别称作4至8元、4至7元、4至6元、5至8元、5至7元以及5至6元杂环基),例如但不限于环氧乙烷基、氮丙啶基、氮杂环丁基、氮杂环庚基、氧杂环丁基、四氢呋喃基、吡咯烷基、吡咯烷酮基、咪唑烷基、吡唑烷基、四氢吡喃基、氮杂基、哌啶基、吗啉基、二噻烷基(dithianyl)、硫吗啉基、哌嗪基、三噻烷基(trithianyl)等;以及它们的并环衍生物或苯并衍生物或杂芳基并衍生物或螺环衍生物等。As used herein, the term "heterocyclyl" refers to a monocyclic or polycyclic group having, for example, 2, 3, 4, 5, 6, 7, 8, 9 carbon atoms and one or more carbon atoms in the ring (eg 1, 2, 3 or 4) selected from C(=O), O, S, S(=O), S(=O) 2 , N and NR (R represents a hydrogen atom or a substituent , such as, but not limited to, alkyl or cycloalkyl). Heterocyclyl groups can be saturated or unsaturated. Saturated heterocyclyl groups may be referred to as heterocycloalkyl groups, such as 3-8 membered heterocycloalkyl, 5-6 membered heterocycloalkyl, and the like. Unless specifically stated otherwise in this specification, a heterocyclyl group may be a monocyclic, bicyclic, tricyclic or more ring ring system, which may include a fused ring system, a paracyclic ring system, a bridged ring system or a spiro ring system. In particular, a 3-8 membered heterocyclyl group is a group having 3-8 carbon atoms and heteroatoms in the ring, for example, it has 4 to 8, 4 to 7, 4 to 6, 5 to 8, 5 to 7 or 5 to 6 carbon atoms and heteroatoms (referred to as 4 to 8 membered, 4 to 7 membered, 4 to 6 membered, 5 to 8 membered, 5 to 7 membered and 5 to 6 membered heterocyclyl, respectively), for example Without limitation , tetrahydropyranyl, aza group, piperidinyl, morpholinyl, dithianyl, thiomorpholinyl, piperazinyl, trithianyl, etc.; and their cyclic derivatives or benzo derivatives or hetero derivatives Aryl derivatives or spiro derivatives, etc.
如本文中所使用,术语“芳基”指具有共轭π电子系统的全碳单环或稠合环多环芳族基团。常见的芳基包括(但不限于)苯基、萘基、蒽基、菲基、苊基、薁基、芴基、茚基、芘基等。例如,术语“C6-10芳基”指含有6至10个碳原子的芳族基团,诸如苯基或萘基。芳基任选地被一或多个(诸如1至3个)适合的取代基(例如卤素、-OH、-CN、-NO2、C1-6烷基等)取代。As used herein, the term "aryl" refers to an all carbon monocyclic or fused ring polycyclic aromatic group having a conjugated pi electron system. Common aryl groups include, but are not limited to, phenyl, naphthyl, anthracenyl, phenanthryl, acenaphthyl, azulenyl, fluorenyl, indenyl, pyrenyl, and the like. For example, the term " C6-10 aryl" refers to an aromatic group containing 6 to 10 carbon atoms, such as phenyl or naphthyl. Aryl groups are optionally substituted with one or more (such as 1 to 3) suitable substituents (eg, halogen, -OH, -CN, -NO2 , C1-6 alkyl, etc.).
如本文中所使用,术语“杂芳基”指含有至少一个选自N、O和S的杂原子的单环、双环或三环芳族环系,其具有例如5、6、8、9、10、11、12、13或14个环原子,特别是具有1或2或3或4或5或6或9或10个碳原子,并且,另外在每一种情况下可为苯并稠合的。例如,如本文中所使用,术语“5-10元杂芳基”意指具有5-10个环原子的单环、双环或三环芳族环系统,并且其中包含至少一个可以相同或不同的杂原子(所述杂原子是例如N、O或S)。5-10元杂芳基的实例包括但不限于噻吩基、呋喃基、吡咯基、噁唑基、噻唑基、咪唑基、吡唑基、异噁唑基、异噻唑基、噁二唑基、三唑基、噻二唑基等,以及它们的苯并衍生物;或吡啶基、哒嗪基、嘧啶基、吡嗪基、三嗪基等,以及它们的苯并衍生物。杂芳基任选地被1或多个(诸如1至3个)适合的取代基(例如卤素、C1-6烷基等)取代。As used herein, the term "heteroaryl" refers to a monocyclic, bicyclic or tricyclic aromatic ring system containing at least one heteroatom selected from N, O and S, having, for example, 5, 6, 8, 9, 10, 11, 12, 13 or 14 ring atoms, in particular 1 or 2 or 3 or 4 or 5 or 6 or 9 or 10 carbon atoms, and, in addition, in each case may be benzo-fused of. For example, as used herein, the term "5-10 membered heteroaryl" means a monocyclic, bicyclic or tricyclic aromatic ring system having 5-10 ring atoms and comprising at least one which may be the same or different Heteroatoms (the heteroatoms are eg N, O or S). Examples of 5-10 membered heteroaryl groups include, but are not limited to, thienyl, furyl, pyrrolyl, oxazolyl, thiazolyl, imidazolyl, pyrazolyl, isoxazolyl, isothiazolyl, oxadiazolyl, Triazolyl, thiadiazolyl, etc., and their benzo derivatives; or pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl, etc., and their benzo derivatives. Heteroaryl groups are optionally substituted with 1 or more (such as 1 to 3) suitable substituents (eg, halogen, C1-6 alkyl, etc.).
如本文中所使用,术语“烷基氨基”是指氨基上的一个氢被烷基取代的基团,例如,如本文所使用,术语“C1-6烷基氨基”是指氨基上的一个氢被C1-6烷基取代的基团,可表示为C1-6烷基-NH-。As used herein, the term "alkylamino" refers to a group in which one hydrogen on an amino group is replaced by an alkyl group, eg, as used herein, the term " C1-6 alkylamino" refers to a group on an amino group A group in which hydrogen is replaced by a C 1-6 alkyl group can be represented as C 1-6 alkyl-NH-.
如本文中所使用,术语“环烷基氨基”是指氨基上的一个氢被环烷基取代的基团,例如,如本文所使用,术语“C3-8环烷基氨基”是指氨基上的一个氢被C3-8环烷基取代的基团,可表示为C3-8环烷基-NH-。As used herein, the term "cycloalkylamino" refers to a group in which one hydrogen on an amino group is replaced by a cycloalkyl group, eg, as used herein, the term " C3-8cycloalkylamino " refers to an amino group A group in which one hydrogen is replaced by a C 3-8 cycloalkyl group can be represented as C 3-8 cycloalkyl-NH-.
如本文中所使用,术语“羟基烷基”是指烷基上的一个氢被羟基取代的基团,例如,如本文中所使用,术语“羟基C1-6烷基”是指C1-6烷基上的一个氢被羟基取代的基团,可表示为-C1-6烷基-OH。As used herein, the term "hydroxyalkyl" refers to a group in which one hydrogen on an alkyl group is replaced by a hydroxy group, eg, as used herein, the term "hydroxyC 1-6 alkyl" refers to C 1- A group in which one hydrogen on the 6 alkyl group is replaced by a hydroxyl group can be represented as -C 1-6 alkyl-OH.
如本文中所使用,术语“羟基”是指-OH。As used herein, the term "hydroxy" refers to -OH.
如本文中所使用,术语“氰基”是指-CN。As used herein, the term "cyano" refers to -CN.
如本文中所使用,术语“氨基”是指-NH2。As used herein, the term "amino" refers to -NH2 .
术语“取代”是指所指定的基团上的一或多个(例如1、2、3或4个)原子(例如氢原子)或原子团(例如三氟甲磺酸酯基)被其他原子或原子团代替,条件是所指定的基团在当前情况下满足价键要求并且在取代之后形成稳定的化合物。取代基和/或变量的组合仅当该组合能够形成稳定的化合物时才是允许的。如果取代基被描述为“任选地被…取代”,则该取代基可以是未被取代的,或是被取代的。如果第一取代基被描述为任选地被第二取代基列表中的一或多个取代,则第一取代基中的一或多个氢原子可以单独地(individually)或各自独立地(independently)被第二取代基列表中的一或多个代替,或者未被代替。The term "substituted" means that one or more (eg, 1, 2, 3, or 4) atoms (eg, hydrogen atoms) or radicals (eg, triflate) of the specified group are replaced by other atoms or Atomic group substitution, provided that the specified group satisfies the valence requirements in the present case and forms a stable compound after substitution. Combinations of substituents and/or variables are permissible only if such combinations form stable compounds. If a substituent is described as "optionally substituted", the substituent can be unsubstituted or substituted. If the first substituent is described as being optionally substituted with one or more of the list of second substituents, one or more hydrogen atoms in the first substituent may be individually or independently ) is replaced by one or more of the second list of substituents, or is not replaced.
如本文中所使用,术语“一或多个”意指在合理条件下的1个或超过1个,例如2个、3个、4个、5个、6个、7个、8个、9个或10个。As used herein, the term "one or more" means 1 or more than 1 under reasonable conditions, such as 2, 3, 4, 5, 6, 7, 8, 9 or 10.
除非指明,否则如本文中所使用,取代基的连接点可来自取代基的任意适宜位置。Unless indicated, as used herein, the point of attachment of a substituent can be from any suitable position on the substituent.
本发明所用术语“药学上可接受的”是指物质或组合物必须与构成制剂的其他组分和/或用其治疗的哺乳动物在化学和/或毒理学上相容。The term "pharmaceutically acceptable" as used herein means that a substance or composition must be chemically and/or toxicologically compatible with the other components that make up the formulation and/or the mammal to be treated with it.
术语“药学上可接受的盐”通常包括(但不限于)本发明的化合物与药学上可接受的无机/有机酸或无机/有机碱反应而形成的盐,此类盐又被称为酸加成盐或碱加成盐。The term "pharmaceutically acceptable salts" generally includes, but is not limited to, salts formed by the reaction of the compounds of the present invention with pharmaceutically acceptable inorganic/organic acids or inorganic/organic bases, such salts are also known as acid additions. Salt or base addition salt.
术语“药学上可接受的酯”通常包括(但不限于)本发明的化合物与药学上可接受的羧酸或磺酸形成的酯,此类酯又被称为羧酸酯或磺酸酯。The term "pharmaceutically acceptable ester" generally includes, but is not limited to, esters of compounds of the present invention with pharmaceutically acceptable carboxylic or sulfonic acids, such esters also known as carboxylate or sulfonate.
术语“异构体”是指因具有相同的原子数和原子类型而具有相同的分子量,但原子的空间排列或构型不同的化合物。The term "isomers" refers to compounds that have the same molecular weight due to the same number and type of atoms, but differ in the spatial arrangement or configuration of the atoms.
术语“立体异构体”(或称“旋光异构体”)是指由于具有至少一个手性因素(包括手性中心、手性轴、手性面等)而导致具有垂直的不对称平面,从而能够使平面偏振光旋转的稳定异构体。由于本发明的化合物存在可能导致立体异构的不对称中心以及其他化学结构,因此本发明也包括这些立体异构体及其混合物。由于本发明的化合物(或其药学上可接受的盐)包括不对称碳原子,因而能够以单一立体异构体形式、外消旋物、对映异构体和非对映异构体的混合物形式存在。通常,这些化合物能够以外消旋物的形式制备。然而,如果需要的话,可以将这类化合物制备或分离后得到纯的立体异构体,即单一对映异构体或非对映异构体,或者单一立体异构体富集化(纯度≥98%、≥95%、≥93%、≥90%、≥88%、≥85%或≥80%)的混合物。如下文中所述,化合物的单一立体异构体是由含有所需手性中心的旋光起始原料合成制备得到的,或者是通过制备得到对映异构体产物的混合物之后再分离或拆分制备得到的,例如转化为非对映异构体的混合物之后再进行分离或重结晶、色谱处理、使用手性拆分试剂,或者在手性色谱柱上将对映异构体进行直接分离。具有特定立体化学的起始化合物既可以商购得到,也可以按照下文中描述的方法制备再通过本领域熟知的方法拆分得到。术语“对映异构体”是指彼此具有不能重叠的镜像的一对立体异构体。术语“非对映异构体”或“非对映体”是指彼此不构成镜像的旋光异构体。术语“外消旋混合物”或“外消旋物”是指含有等份的单一对映异构体的混合物(即两种R和S对映体的等摩尔量混合物)。术语“非外消旋混合物”是指含有不等份的单一对映异构体的混合物。除非另外指出,本发明的化合物的所有立体异构体形式都在本发明的范围之内。The term "stereoisomer" (or "optical isomer") refers to having at least one chiral factor (including chiral center, chiral axis, chiral plane, etc.) resulting in a vertical asymmetric plane, Thus, a stable isomer capable of rotating plane-polarized light. Since the compounds of the present invention have asymmetric centers and other chemical structures that may lead to stereoisomerism, the present invention also includes these stereoisomers and mixtures thereof. Since the compounds of the present invention (or pharmaceutically acceptable salts thereof) include asymmetric carbon atoms, they can be in the form of single stereoisomers, racemates, mixtures of enantiomers and diastereomers form exists. Generally, these compounds can be prepared in the form of racemates. However, if desired, such compounds may be prepared or isolated as pure stereoisomers, i.e. single enantiomers or diastereomers, or enriched for single stereoisomers (purity ≥ 98%, ≥95%, ≥93%, ≥90%, ≥88%, ≥85% or ≥80%). Single stereoisomers of compounds are prepared synthetically from optically active starting materials containing the desired chiral centers, as described hereinafter, or by preparation of mixtures of enantiomeric products followed by separation or resolution The resulting mixture is, for example, converted to a mixture of diastereomers followed by separation or recrystallization, chromatography, use of chiral resolving agents, or direct separation of the enantiomers on a chiral chromatography column. Starting compounds with specific stereochemistry are either commercially available or can be prepared according to the methods described below and resolved by methods well known in the art. The term "enantiomers" refers to a pair of stereoisomers that have non-superimposable mirror images of each other. The term "diastereomer" or "diastereomer" refers to optical isomers that do not form mirror images of each other. The term "racemic mixture" or "racemate" refers to a mixture containing equal parts of a single enantiomer (ie, an equimolar mixture of the two R and S enantiomers). The term "non-racemic mixture" refers to a mixture containing unequal parts of a single enantiomer. Unless otherwise indicated, all stereoisomeric forms of the compounds of the present invention are within the scope of the present invention.
术语“互变异构体”(或称“互变异构形式”)是指具有不同能量的,可通过低能垒互相转化的结构异构体。若互变异构是可能的(如在溶液中),则可以达到互变异构体的化学平衡。例如,质子互变异构体(或称质子转移互变异构体)包括(但不限于)通过质子迁移来进行的互相转化,如酮-烯醇异构化、亚胺-烯胺异构化、酰胺-亚胺醇异构化等。除非另外指出,本发明的化合物的所有互变异构体形式都在本发明的范围之内。The term "tautomer" (or "tautomeric form") refers to structural isomers of different energies that can be interconverted through a low energy barrier. A chemical equilibrium of tautomers can be achieved if tautomerism is possible (eg, in solution). For example, proton tautomers (or proton transfer tautomers) include, but are not limited to, interconversion by proton transfer, such as keto-enol isomerization, imine-enamine isomerization isomerization, amide-imino alcohol isomerization, etc. Unless otherwise indicated, all tautomeric forms of the compounds of the present invention are within the scope of the present invention.
术语“多晶型物”(或称“多晶型形式”)是指化合物或复合物的固体晶体形式。本领域技术人员可通过许多已知的方法获得分子的多晶型物。这些方法包括(但不限于)熔融重结晶、熔融冷却、溶剂重结晶、去溶剂化、快速蒸发、快速冷却、慢速冷却、汽相扩散和升华。另外,可用熟知的技术检测、分类和鉴定多晶型物,这些技术包括(但不限于)差示扫描量热法(DSC)、热重分析法(TGA)、X射线粉末衍射法(XRPD)、单晶X射线衍射法(SCXRD)、固态核磁共振(NMR)、红外光谱法(IR)、拉曼光谱法和扫描电镜术(SEM)等。The term "polymorph" (or "polymorphic form") refers to a solid crystalline form of a compound or complex. Polymorphs of the molecule can be obtained by one of skill in the art by a number of known methods. These methods include, but are not limited to, melt recrystallization, melt cooling, solvent recrystallization, desolvation, rapid evaporation, rapid cooling, slow cooling, vapor phase diffusion, and sublimation. Additionally, polymorphs may be detected, classified and identified using well known techniques including, but not limited to, Differential Scanning Calorimetry (DSC), Thermogravimetric Analysis (TGA), X-ray Powder Diffraction (XRPD) , Single Crystal X-ray Diffraction (SCXRD), Solid State Nuclear Magnetic Resonance (NMR), Infrared Spectroscopy (IR), Raman Spectroscopy and Scanning Electron Microscopy (SEM), etc.
术语“溶剂合物”是指由本发明的化合物(或其药学上可接受的盐)与至少一种溶剂分子通过非共价分子间作用力结合而形成的物质。常见的溶剂合物包括(但不限于)水合物(包括半水合物、一水合物、二水合物、三水合物等)、乙醇合物、丙酮合物等。The term "solvate" refers to a substance formed by the association of a compound of the present invention (or a pharmaceutically acceptable salt thereof) with at least one solvent molecule through non-covalent intermolecular forces. Common solvates include, but are not limited to, hydrates (including hemihydrates, monohydrates, dihydrates, trihydrates, and the like), ethanolates, acetonates, and the like.
术语“氮氧化物”是指叔胺类或含氮(芳)杂环类化合物结构中的氮原子经氧化而形成的化合物。例如,式I化合物母核中的1位氮原子可以形成相应的氮氧化物。The term "nitrogen oxide" refers to compounds formed by oxidation of nitrogen atoms in the structure of tertiary amines or nitrogen-containing (aromatic) heterocyclic compounds. For example, a nitrogen atom in position 1 in the parent nucleus of a compound of formula I can form the corresponding nitrogen oxide.
术语“同位素标记物”是指将本发明的化合物中的特定原子替换为其同位素原子而形成的衍生化合物。除非另外指出,本发明的化合物包括H、C、N、O、F、P、S、Cl的各种同位素,如2H(D)、3H(T)、13C、14C、15N、17O、18O、18F、31P、32P、35S、36S和37Cl。The term "isotopic label" refers to a derivative compound formed by substituting a particular atom in a compound of the present invention for its isotopic atom. Unless otherwise indicated, compounds of the present invention include various isotopes of H, C, N, O, F, P, S, Cl, such as 2 H(D), 3 H(T), 13 C, 14 C, 15 N , 17 O, 18 O, 18 F, 31 P, 32 P, 35 S, 36 S and 37 Cl.
术语“代谢物”是指本发明的化合物经代谢后形成的衍生化合物。关于代谢的进一步信息可参见Goodman and Gilman's:The Pharmacological Basis of Therapeutics(9thed.)[M],McGraw-Hill International Editions,1996。The term "metabolite" refers to a derivative compound formed upon metabolism of a compound of the present invention. Further information on metabolism can be found in Goodman and Gilman's: The Pharmacological Basis of Therapeutics (9th ed .) [M], McGraw-Hill International Editions, 1996.
术语“前药”是指在施用于个体后能够直接或间接地提供本发明的化合物的衍生化合物。特别优选的衍生化合物或前药是在施用于个体时可以提高本发明的化合物的生物利用度的化合物(例如,更易吸收入血),或者促进母体化合物向作用位点(例如,淋巴系统)递送的化合物。除非另外指出,本发明的化合物的所有前药形式都在本发明的范围之内,且各种前药形式是本领域熟知的。The term "prodrug" refers to a derivative compound capable of providing, directly or indirectly, a compound of the present invention upon administration to an individual. Particularly preferred derivative compounds or prodrugs are those that, when administered to an individual, increase the bioavailability of the compounds of the invention (eg, more readily absorbed into the blood), or facilitate delivery of the parent compound to the site of action (eg, the lymphatic system) compound of. Unless otherwise indicated, all prodrug forms of the compounds of the present invention are within the scope of this invention, and various prodrug forms are well known in the art.
术语“各自独立地”是指结构中存在的取值范围相同或相近的至少两个基团(或环系)可以在特定情形下具有相同或不同的含义。例如,取代基X和取代基Y各自独立地为氢、卤素、羟基、氰基、烷基或芳基,则当取代基X为氢时,取代基Y既可以为氢,也可以为卤素、羟基、氰基、烷基或芳基;同理;当取代基Y为氢时,取代基X既可以为氢,也可以为卤素、羟基、氰基、烷基或芳基。The term "each independently" means that at least two groups (or ring systems) present in the structure with the same or similar value ranges may have the same or different meanings under certain circumstances. For example, Substituent X and Substituent Y are each independently hydrogen, halogen, hydroxyl, cyano, alkyl or aryl, then when Substituent X is hydrogen, Substituent Y can be either hydrogen or halogen, Hydroxyl, cyano, alkyl or aryl; the same is true; when the substituent Y is hydrogen, the substituent X can be either hydrogen or halogen, hydroxyl, cyano, alkyl or aryl.
本发明还涵盖含有保护基的本发明的化合物。在制备本发明的化合物的任何过程中,保护在任何有关分子上的敏感基团或反应基团可能是必需的和/或期望的,由此形成本发明的化合物的化学保护的形式。这可以通过常规的保护基实现,例如,在ProtectiveGroups in Organic Chemistry,ed.J.F.W.McOmie,Plenum Press,1973;和T.W.Greene&P.G.M.Wuts,Protective Groups in Organic Synthesis,John Wiley&Sons,1991中所述的那些保护基,这些参考文献通过援引加入本文。使用本领域已知的方法,可以在适当的后续阶段移除保护基。The present invention also encompasses compounds of the present invention that contain protecting groups. In any process for preparing the compounds of the present invention, it may be necessary and/or desirable to protect sensitive or reactive groups on any relevant molecule, thereby forming chemically protected forms of the compounds of the present invention. This can be accomplished by conventional protecting groups, such as those described in Protective Groups in Organic Chemistry, ed. J.F.W. McOmie, Plenum Press, 1973; and T.W. Greene & P.G.M. Wuts, Protective Groups in Organic Synthesis, John Wiley & Sons, 1991 basis, these references are incorporated herein by reference. Protecting groups can be removed at an appropriate subsequent stage using methods known in the art.
化合物compound
本发明提供了的一个目的在于提供一种式I所示的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药,An object provided by the present invention is to provide a compound represented by formula I or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitrogen oxide compounds, isotopic labels, metabolites or prodrugs,
其中,in,
R1为5-10元杂芳基,所述5-10元杂芳基任选地被一或多个R4取代;R 1 is a 5-10-membered heteroaryl group optionally substituted with one or more R 4 ;
R2选自氢、卤素、C1-6烷基、C1-6卤代烷基、C3-8环烷基、氰基、-C(=O)NRaRb和-C(=O)ORa;R 2 is selected from hydrogen, halogen, C 1-6 alkyl, C 1-6 haloalkyl, C 3-8 cycloalkyl, cyano, -C(=O)NR a R b and -C(=O) OR a ;
Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-8环烷基和4-8元杂环基,所述C1-6烷基、C3-8环烷基或4-8元杂环基任选地被一或多个R5取代;或者Ra、Rb和与它们连接的氮原子一起形成4-8元杂环,所述4-8元杂环任选地被一或多个R5取代;R a and R b are each independently selected at each occurrence from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl and 4-8 membered heterocyclyl, the C 1-6 alkyl, C 3-8 cycloalkyl or 4-8 membered heterocyclyl optionally substituted with one or more R 5 ; or R a , R b and the nitrogen atom to which they are attached together form a 4-8 membered heterocycle, the The 4-8 membered heterocycle is optionally substituted with one or more R5;
R3选自氢和-NRcRd;R 3 is selected from hydrogen and -NR c R d ;
Rc和Rd在每次出现时各自独立地选自氢、C1-6烷基、C3-8环烷基、4-8元杂环基、-C(=O)Ra、C6-10芳基和5-10元杂芳基,所述C1-6烷基、C3-8环烷基、4-8元杂环基、C6-10芳基或5-10元杂芳基任选地被一或多个R6取代;R c and R d at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl, 4-8 membered heterocyclyl, -C(=O)R a , C 6-10 -membered aryl and 5-10-membered heteroaryl, the C 1-6 alkyl, C 3-8 cycloalkyl, 4-8-membered heterocyclyl, C 6-10 -membered aryl or 5-10-membered Heteroaryl is optionally substituted with one or more R ;
R4在每次出现时各自独立地选自氘、卤素、C1-6烷基、C1-6烷氧基和C3-8环烷基,所述C1-6烷基、C1-6烷氧基或C3-8环烷基任选地被一或多个卤素取代;R 4 at each occurrence is independently selected from deuterium, halogen, C 1-6 alkyl, C 1-6 alkoxy and C 3-8 cycloalkyl, the C 1-6 alkyl, C 1-6 alkyl -6 alkoxy or C 3-8 cycloalkyl optionally substituted with one or more halogens;
R5在每次出现时各自独立地选自卤素、羟基、C1-6烷基、C1-6烷氧基、氨基、C1-6烷基氨基、C3-8环烷基和4-8元杂环基,所述C1-6烷基或C3-8环烷基任选地被一或多个羟基取代;R 5 at each occurrence is independently selected from halogen, hydroxy, C 1-6 alkyl, C 1-6 alkoxy, amino, C 1-6 alkylamino, C 3-8 cycloalkyl and 4 -8-membered heterocyclyl, the C 1-6 alkyl or C 3-8 cycloalkyl is optionally substituted with one or more hydroxyl groups;
R6在每次出现时各自独立地选自卤素、羟基、C1-6烷基、C1-6烷氧基、氨基、C1-6烷基氨基、C3-8环烷基和4-8元杂环基,所述C1-6烷基、C1-6烷氧基、C1-6烷基氨基、C3-8环烷基或4-8元杂环基任选地被一或多个选自羟基和C1-6烷基的基团取代;R 6 at each occurrence is independently selected from halogen, hydroxy, C 1-6 alkyl, C 1-6 alkoxy, amino, C 1-6 alkylamino, C 3-8 cycloalkyl and 4 -8-membered heterocyclyl, the C 1-6 alkyl, C 1-6 alkoxy, C 1-6 alkylamino, C 3-8 cycloalkyl or 4-8 membered heterocyclyl are optionally substituted with one or more groups selected from hydroxy and C 1-6 alkyl;
Y选自氢、C1-6烷基、C3-8环烷基、C1-6卤代烷基、C1-6烷氧基、C3-8环烷氧基、C1-6烷硫基、C3-8环烷硫基、C1-6烷基氨基、C3-8环烷基氨基和4-8元杂环基;Y is selected from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl, C 1-6 haloalkyl, C 1-6 alkoxy, C 3-8 cycloalkoxy, C 1-6 alkylthio base, C 3-8 cycloalkylthio, C 1-6 alkylamino, C 3-8 cycloalkylamino and 4-8 membered heterocyclyl;
Z选自-CR7-和-N-;且Z is selected from -CR7- and -N-; and
R7选自氢、卤素、C1-6烷基、C3-8环烷基、C1-6卤代烷基和氰基。R 7 is selected from hydrogen, halogen, C 1-6 alkyl, C 3-8 cycloalkyl, C 1-6 haloalkyl and cyano.
根据本发明的一些实施方案,本发明的化合物为式I-1的化合物:According to some embodiments of the present invention, the compound of the present invention is a compound of formula I-1:
其中,in,
R1、R2、R3和Y如上文所定义。R 1 , R 2 , R 3 and Y are as defined above.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,R4在每次出现时各自独立地选自氘、卤素、C1-6烷基、C1-6卤代烷基和C3-8环烷基。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, each occurrence of R 4 is independently selected from deuterium, halogen, C 1-6 alkyl, C 1-6 Haloalkyl and C 3-8 cycloalkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R4在每次出现时各自独立地选自卤素、C1-6烷基、C1-6卤代烷基和C3-6环烷基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, each occurrence of R 4 is independently selected from halogen, C 1-6 alkyl, C 1-6 haloalkane group and C 3-6 cycloalkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R4在每次出现时各自独立地选自氟、甲基、三氟甲基、二氟甲基、一氟甲基、异丙基和环丙基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 4 at each occurrence is independently selected from fluoro, methyl, trifluoromethyl, difluoromethyl , monofluoromethyl, isopropyl and cyclopropyl.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,R1为5-6元杂芳基,所述5-6元杂芳基任选地被一或多个R4取代。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is a 5-6 membered heteroaryl group optionally formed by one or more R 4 is substituted.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R1为5-6元杂芳基,所述5-6元杂芳基任选地被一或多个独立地选自以下的基团取代:卤素、C1-6烷基、C1-6卤代烷基和C3-6环烷基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is a 5-6 membered heteroaryl, optionally substituted by a or A plurality of groups independently selected from halogen, C1-6 alkyl, C1-6 haloalkyl, and C3-6 cycloalkyl are substituted.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R1为5-6元含氮杂芳基,所述5-6元含氮杂芳基任选地被一或多个R4取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is a 5-6 membered nitrogen-containing heteroaryl, and the 5-6 membered nitrogen-containing heteroaryl is optionally is substituted with one or more R 4 .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R1为5-6元含氮杂芳基,所述5-6元含氮杂芳基任选地被一或多个独立地选自以下的基团取代:卤素、C1-6烷基、C1-6卤代烷基和C3-6环烷基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is a 5-6 membered nitrogen-containing heteroaryl, and the 5-6 membered nitrogen-containing heteroaryl is optionally is substituted with one or more groups independently selected from halogen, C1-6 alkyl, C1-6 haloalkyl, and C3-6 cycloalkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R1选自吡啶基和吡唑基,所述吡啶基或吡唑基任选地被一或多个R4取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is selected from pyridyl and pyrazolyl, said pyridyl or pyrazolyl optionally being replaced by one or more R 4 is substituted.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R1选自吡啶基和吡唑基,所述吡啶基或吡唑基任选地被一或多个独立地选自以下的基团取代:卤素、C1-6烷基、C1-6卤代烷基和C3-6环烷基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is selected from pyridyl and pyrazolyl, said pyridyl or pyrazolyl optionally being replaced by one or more substituted with groups independently selected from the group consisting of halogen, C1-6 alkyl, C1-6 haloalkyl, and C3-6 cycloalkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R1选自 其中波浪线表示该基团与分子其余部分的连接点。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 1 is selected from where the wavy line Indicates the point of attachment of this group to the rest of the molecule.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,R5在每次出现时各自独立地选自卤素、羟基、C1-6烷基、羟基C1-6烷基、C1-6烷氧基、氨基、C1-6烷基氨基、C3-6环烷基、羟基C3-6环烷基和4-8元杂环基。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, each occurrence of R 5 is independently selected from halogen, hydroxy, C 1-6 alkyl, hydroxy C 1- 6 alkyl, C 1-6 alkoxy, amino, C 1-6 alkylamino, C 3-6 cycloalkyl, hydroxy C 3-6 cycloalkyl and 4-8 membered heterocyclic group.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R5在每次出现时各自独立地选自羟基和C1-6烷基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, each occurrence of R 5 is independently selected from hydroxy and C 1-6 alkyl at each occurrence.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基和4-6元杂环基,所述C1-6烷基、C3-6环烷基或4-6元杂环基任选地被一或多个R5取代;或者Ra、Rb和与它们连接的氮原子一起形成4-6元杂环,所述4-6元杂环任选地被一或多个R5取代。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R a and R b at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3- 6 -cycloalkyl and 4-6 membered heterocyclyl, the C1-6 alkyl, C3-6 cycloalkyl or 4-6 membered heterocyclyl optionally substituted with one or more R5 ; or Ra , Rb and the nitrogen atom to which they are attached together form a 4-6 membered heterocycle optionally substituted with one or more R5 .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基和4-6元杂环基,所述C1-6烷基、C3-6环烷基或4-6元杂环基任选地被一或多个独立地选自羟基和C1-6烷基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成4-6元杂环,所述4-6元杂环任选地被一或多个独立地选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of Formula I and Formula I-1 of the present invention, R a and R b at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3 -6 cycloalkyl and 4-6 membered heterocyclyl, the C 1-6 alkyl, C 3-6 cycloalkyl or 4-6 membered heterocyclyl optionally being independently selected from one or more hydroxy and C 1-6 alkyl groups; or R a , R b and the nitrogen atom to which they are attached together form a 4-6 membered heterocycle optionally substituted by one or more substituted with groups independently selected from hydroxy and C 1-6 alkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基、氮杂环丁基和哌啶基,所述C1-6烷基、C3-6环烷基、氮杂环丁基或哌啶基任选地被一或多个R5取代;或者Ra、Rb和与它们连接的氮原子一起形成4-6元杂环,所述4-6元杂环任选地被一或多个R5取代。In some embodiments of the present invention, in the compounds of Formula I and Formula I-1 of the present invention, R a and R b at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3 -6 cycloalkyl, azetidinyl and piperidinyl, the C 1-6 alkyl, C 3-6 cycloalkyl, azetidinyl or piperidinyl optionally being replaced by one or more R5 is substituted ; or Ra , Rb and the nitrogen atom to which they are attached together form a 4-6 membered heterocycle optionally substituted with one or more R5 .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基、氮杂环丁基和哌啶基,所述C1-6烷基、C3-6环烷基、氮杂环丁基或哌啶基任选地被一或多个独立地选自羟基和C1-6烷基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成4-6元杂环,所述4-6元杂环任选地被一或多个独立地选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of Formula I and Formula I-1 of the present invention, R a and R b at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3 -6 cycloalkyl, azetidinyl and piperidinyl, the C 1-6 alkyl, C 3-6 cycloalkyl, azetidinyl or piperidinyl optionally being replaced by one or more substituted with groups independently selected from hydroxy and C 1-6 alkyl; or R a , R b and the nitrogen atom to which they are attached together form a 4-6 membered heterocycle optionally Substituted with one or more groups independently selected from hydroxy and C 1-6 alkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基、氮杂环丁基和哌啶基,所述C1-6烷基、C3-6环烷基、氮杂环丁基或哌啶基任选地被一或多个独立地选自羟基和C1-6烷基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成氮杂环丁基,所述氮杂环丁基任选地被一或多个独立地选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of Formula I and Formula I-1 of the present invention, R a and R b at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3 -6 cycloalkyl, azetidinyl and piperidinyl, the C 1-6 alkyl, C 3-6 cycloalkyl, azetidinyl or piperidinyl optionally being replaced by one or more substituted with groups independently selected from hydroxy and C 1-6 alkyl; or R a , R b and the nitrogen atom to which they are attached together form an azetidine optionally substituted by a substituted with more than one group independently selected from hydroxy and C 1-6 alkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Ra和Rb在每次出现时各自独立地选自氢、甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、环丁基、氮杂环丁基和哌啶基,所述甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、环丁基、氮杂环丁基和哌啶基任选地被一个或多个独立选自羟基、甲基和乙基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成氮杂环丁基,所述氮杂环丁基任选地被一或多个独立地选自羟基和甲基的基团取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R a and R b at each occurrence are each independently selected from hydrogen, methyl, ethyl, n-propyl , isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, cyclobutyl, azetidinyl and piperidinyl, the methyl, ethyl, n-propyl, isopropyl , n-butyl, isobutyl, sec-butyl, tert-butyl, cyclobutyl, azetidinyl, and piperidinyl optionally surrounded by one or more groups independently selected from hydroxy, methyl, and ethyl or R a , R b and the nitrogen atom to which they are attached together form an azetidine group optionally with one or more groups independently selected from hydroxy and methyl replace.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,R2选自氢、卤素、氰基和-C(=O)NRaRb。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 2 is selected from hydrogen, halogen, cyano and -C(=O)NR a R b .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R2选自氢、氰基和-C(=O)NRaRb。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 2 is selected from hydrogen, cyano and -C(=O)NR a R b .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R2选自氢、氰基和-C(=O)NRaRb,其中Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基、氮杂环丁基和哌啶基,所述C1-6烷基、C3-6环烷基、氮杂环丁基或哌啶基任选地被一或多个独立地选自羟基和C1-6烷基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成4-6元杂环,所述4-6元杂环任选地被一或多个独立地选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 2 is selected from hydrogen, cyano and -C(=O)NR a R b , wherein R a and R b Each occurrence is independently selected from hydrogen, C 1-6 alkyl, C 3-6 cycloalkyl, azetidinyl, and piperidinyl, the C 1-6 alkyl, C 3-6 cycloalkyl, azetidinyl or piperidinyl optionally substituted with one or more groups independently selected from hydroxy and C1-6 alkyl; or R a , R b and the nitrogen to which they are attached The atoms together form a 4-6 membered heterocycle optionally substituted with one or more groups independently selected from hydroxy and C1-6 alkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R2选自氢、氰基和-C(=O)NRaRb,其中Ra和Rb在每次出现时各自独立地选自氢、甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、环丁基、氮杂环丁基和哌啶基,所述甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、环丁基、氮杂环丁基和哌啶基任选地被一个或多个独立选自羟基、甲基和乙基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成氮杂环丁基,所述氮杂环丁基任选地被一或多个独立地选自羟基和甲基的基团取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 2 is selected from hydrogen, cyano and -C(=O)NR a R b , wherein R a and R b Each occurrence is independently selected from hydrogen, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, cyclobutyl, azetidine and piperidinyl, the methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, cyclobutyl, azetidinyl and piperidine radicals are optionally substituted with one or more groups independently selected from hydroxy, methyl and ethyl; or R a , R b and the nitrogen atom to which they are attached together form an azetidine, the azacyclo The butyl group is optionally substituted with one or more groups independently selected from hydroxy and methyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R2选自氢、氰基、-C(=O)NH2、-C(=O)N(H)CH3、 其中波浪线表示该基团与分子其余部分的连接点。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 2 is selected from hydrogen, cyano, -C(=O)NH 2 , -C(=O)N( H)CH 3 , where the wavy line Indicates the point of attachment of this group to the rest of the molecule.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,R6在每次出现时各自独立地选自C1-6烷基、C1-6烷氧基和4-8元杂环基,所述C1-6烷基、C1-6烷氧基或4-8元杂环基任选地被一或多个选自羟基和C1-6烷基的基团取代。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 6 at each occurrence is each independently selected from C 1-6 alkyl, C 1-6 alkoxy and 4-8-membered heterocyclyl, the C 1-6 alkyl, C 1-6 alkoxy or 4-8-membered heterocyclyl is optionally by one or more selected from hydroxyl and C 1-6 alkyl group substitution.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R6在每次出现时各自独立地选自C1-6烷基、C1-6烷氧基和5-6元杂环基,所述C1-6烷基、C1-6烷氧基或5-6元杂环基任选地被一或多个选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 6 at each occurrence is independently selected from C 1-6 alkyl, C 1-6 alkoxy and 5-6 membered heterocyclyl, the C 1-6 alkyl, C 1-6 alkoxy or 5-6 membered heterocyclyl is optionally replaced by one or more selected from hydroxyl and C 1-6 alkane radical group substitution.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,R6在每次出现时各自独立地选自C1-6烷基、C1-6烷氧基和哌嗪基,所述C1-6烷基、C1-6烷氧基或哌嗪基任选地被选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R 6 at each occurrence is independently selected from C 1-6 alkyl, C 1-6 alkoxy and piperazinyl, the C 1-6 alkyl, C 1-6 alkoxy or piperazinyl is optionally substituted with a group selected from hydroxy and C 1-6 alkyl.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,Rc和Rd在每次出现时各自独立地选自氢、C6-10芳基和5-10元杂芳基,所述C6-10芳基或5-10元杂芳基任选地被一或多个R6取代。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R c and R d at each occurrence are each independently selected from hydrogen, C 6-10 aryl and 5-10 Membered heteroaryl, the C 6-10 aryl or 5-10 membered heteroaryl is optionally substituted with one or more R 6 .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Rc和Rd在每次出现时各自独立地选自氢、苯基和5-6元杂芳基,所述苯基或5-6元杂芳基任选地被一或多个R6取代。In some embodiments of the invention, in the compounds of formula I and formula I-1 of the invention, R c and R d at each occurrence are each independently selected from hydrogen, phenyl and 5-6 membered heteroaryl group, the phenyl or 5-6 membered heteroaryl is optionally substituted with one or more R 6 .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Rc和Rd在每次出现时各自独立地选自氢、苯基和吡啶基,所述苯基或吡啶基任选地被一或多个R6取代。In some embodiments of the present invention, in the compounds of Formula I and Formula I-1 of the present invention, Rc and Rd are each independently selected at each occurrence from hydrogen, phenyl, and pyridyl, the benzene or pyridyl is optionally substituted with one or more R 6 .
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Rc和Rd在每次出现时各自独立地选自氢、苯基和吡啶基,所述苯基或吡啶基任选地被一或多个各自独立地选自以下的基团取代:C1-6烷基、C1-6烷氧基和哌嗪基,所述C1-6烷基或哌嗪基任选地被选自羟基和C1-6烷基的基团取代。In some embodiments of the present invention, in the compounds of Formula I and Formula I-1 of the present invention, Rc and Rd are each independently selected at each occurrence from hydrogen, phenyl, and pyridyl, the benzene or pyridyl optionally substituted with one or more groups each independently selected from the group consisting of C 1-6 alkyl, C 1-6 alkoxy and piperazinyl, the C 1-6 alkyl Or piperazinyl is optionally substituted with a group selected from hydroxy and C 1-6 alkyl.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,R3选自氢、 其中波浪线表示该基团与分子其余部分的连接点。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, R is selected from hydrogen, where the wavy line Indicates the point of attachment of this group to the rest of the molecule.
根据本发明的一些实施方案,在本发明的式I和式I-1的化合物中,Y选自氢、C1-6烷基、C3-8环烷基、C1-6卤代烷基、C1-6烷氧基、C3-8环烷氧基、C1-6烷基氨基和C3-8环烷基氨基。According to some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, Y is selected from hydrogen, C 1-6 alkyl, C 3-8 cycloalkyl, C 1-6 haloalkyl, C 1-6 alkoxy, C 3-8 cycloalkoxy, C 1-6 alkylamino and C 3-8 cycloalkylamino.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Y为C1-6烷基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, Y is C1-6 alkyl.
在本发明的一些实施方案中,在本发明的式I和式I-1的化合物中,Y为甲基、乙基和异丙基。In some embodiments of the present invention, in the compounds of formula I and formula I-1 of the present invention, Y is methyl, ethyl and isopropyl.
根据本发明的一些实施方案,在本发明的式I的化合物中,R7选自氢、卤素、C1-6卤代烷基和氰基。According to some embodiments of the present invention, in the compounds of formula I of the present invention, R 7 is selected from hydrogen, halogen, C 1-6 haloalkyl and cyano.
在本发明的一些实施方案中,在本发明的式I的化合物中,R7为氢。In some embodiments of the present invention, in the compounds of formula I of the present invention, R7 is hydrogen.
根据本发明的一些实施方案,在本发明的式I的化合物中,Z为-CR7-。According to some embodiments of the present invention, in the compounds of formula I of the present invention, Z is -CR7- .
本发明涵盖对上述优选基团进行任意组合所得的式I和I-1的化合物。在本发明的一些实施方案,本发明的式I和I-1的化合物中,The present invention encompasses compounds of formulae I and I-1 resulting from any combination of the above preferred groups. In some embodiments of the invention, in the compounds of formula I and I-1 of the invention,
R1选自吡啶基和吡唑基,所述吡啶基或吡唑基任选地被一或多个独立地选自以下的基团取代:卤素、C1-6烷基、C1-6卤代烷基和C3-6环烷基;R 1 is selected from pyridyl and pyrazolyl optionally substituted with one or more groups independently selected from halogen, C 1-6 alkyl, C 1-6 haloalkyl and C 3-6 cycloalkyl;
R2选自氢、氰基和-C(=O)NRaRb,其中Ra和Rb在每次出现时各自独立地选自氢、C1-6烷基、C3-6环烷基和4-6元杂环基,所述C1-6烷基、C3-6环烷基或4-6元杂环基任选地被一或多个独立地选自羟基和C1-6烷基的基团取代;或者Ra、Rb和与它们连接的氮原子一起形成4-6元杂环,所述4-6元杂环任选地被一或多个独立地选自羟基和C1-6烷基的基团取代;R 2 is selected from hydrogen, cyano and -C(=O)NR a R b , wherein R a and R b at each occurrence are each independently selected from hydrogen, C 1-6 alkyl, C 3-6 ring Alkyl and 4-6 membered heterocyclyl, the C 1-6 alkyl, C 3-6 cycloalkyl or 4-6 membered heterocyclyl optionally being one or more independently selected from hydroxy and C 1-6 alkyl group substitution; alternatively R a , R b and the nitrogen atom to which they are attached together form a 4-6 membered heterocycle optionally substituted by one or more independently substituted with a group selected from hydroxy and C 1-6 alkyl;
R3选自氢和-NRcRd;R 3 is selected from hydrogen and -NR c R d ;
Rc和Rd在每次出现时各自独立地选自氢、苯基和吡啶基,所述苯基或吡啶基任选地被一或多个各自独立地选自以下的基团取代:C1-6烷基、C1-6烷氧基和哌嗪基,所述C1-6烷基或哌嗪基任选地被选自羟基和C1-6烷基的基团取代;且R c and R d are each independently selected at each occurrence from hydrogen, phenyl and pyridyl optionally substituted with one or more groups each independently selected from: C 1-6 alkyl, C 1-6 alkoxy and piperazinyl, the C 1-6 alkyl or piperazinyl optionally substituted with a group selected from hydroxy and C 1-6 alkyl; and
Y为C1-6烷基。Y is C 1-6 alkyl.
根据本发明的一些实施方案,本发明的化合物选自:According to some embodiments of the present invention, the compounds of the present invention are selected from:
制备方法Preparation
本发明的另一目的在于提供制备本发明的化合物的方法,其包括:Another object of the present invention is to provide a method for preparing the compound of the present invention, comprising:
步骤1-1:化合物SM1和N,N-二甲基甲酰胺二甲基缩醛发生缩合反应得到化合物IM1Step 1-1: Compound SM1 and N,N-dimethylformamide dimethyl acetal undergo condensation reaction to obtain compound IM1
步骤1-2:化合物IM1和化合物SM2发生环化反应得到化合物IM2Step 1-2: Compound IM1 and compound SM2 undergo cyclization reaction to obtain compound IM2
步骤1-3:化合物IM2发生取代反应得到化合物IM3Step 1-3: Compound IM2 undergoes substitution reaction to obtain compound IM3
步骤1-4:化合物IM3和SM3发生取代反应得到式I化合物Step 1-4: Substitution reaction of compound IM3 and SM3 to obtain compound of formula I
其中,LG为离去基团,例如苯氧基和卤素;并且,where LG is a leaving group such as phenoxy and halogen; and,
Y、Z、R1、R2和R3如上文所定义。Y, Z, R 1 , R 2 and R 3 are as defined above.
步骤1-1:化合物SM1和N,N-二甲基甲酰胺二甲基缩醛发生缩合反应得到化合物IM1Step 1-1: Compound SM1 and N,N-dimethylformamide dimethyl acetal undergo condensation reaction to obtain compound IM1
在本发明的一些实施方案中,该反应在适合的温度下进行,所述温度为0-200℃,优选0-100℃。In some embodiments of the invention, the reaction is carried out at a suitable temperature, said temperature being 0-200°C, preferably 0-100°C.
步骤1-2:化合物IM1和化合物SM2发生环化反应得到化合物IM2:Step 1-2: Compound IM1 and compound SM2 undergo cyclization reaction to obtain compound IM2:
在本发明的一些实施方案中,该反应在适合的有机溶剂中进行,所述有机溶剂可选自醇类(例如MeOH、EtOH、n-BuOH等)、THF、AcOH、Diox及其任意组合,优选甲醇或乙醇。In some embodiments of the present invention, the reaction is carried out in a suitable organic solvent, which may be selected from alcohols (eg, MeOH, EtOH, n-BuOH, etc.), THF, AcOH, Diox, and any combination thereof, Methanol or ethanol is preferred.
在本发明的一些实施方案中,该反应在适合的碱的存在下进行,所述碱包括有机碱或无机碱,所述有机碱可选自DIPEA、TEA、t-BuOK、甲醇钠、乙醇钠和Py,所述无机碱可选自K3PO4、NaH、K2CO3、Na2CO3、Cs2CO3和NaOH,优选甲醇钠或乙醇钠。In some embodiments of the present invention, the reaction is carried out in the presence of a suitable base, including an organic base or an inorganic base, the organic base may be selected from DIPEA, TEA, t-BuOK, sodium methoxide, sodium ethoxide and Py, the inorganic base can be selected from K 3 PO 4 , NaH, K 2 CO 3 , Na 2 CO 3 , Cs 2 CO 3 and NaOH, preferably sodium methoxide or sodium ethoxide.
在本发明的一些实施方案中,该反应在适合的温度下进行,所述温度为0-200℃,优选100-200℃。In some embodiments of the present invention, the reaction is carried out at a suitable temperature, said temperature being 0-200°C, preferably 100-200°C.
步骤1-3:化合物IM2发生取代反应得到化合物IM3Step 1-3: Compound IM2 undergoes substitution reaction to obtain compound IM3
在本发明的一些实施方案中,该反应在适合的有机溶剂中进行,所述有机溶剂可选自卤代烃类(例如DCM、TCM、1,2-DCE等)、腈类(例如AN等)、NMP、DMF、DMA、THF、Diox、DMSO、芳烃类(例如TL、XY)及其任意组合,优选DMF、THF或DCM。In some embodiments of the invention, the reaction is carried out in a suitable organic solvent, which may be selected from halogenated hydrocarbons (eg, DCM, TCM, 1,2-DCE, etc.), nitriles (eg, AN, etc.) ), NMP, DMF, DMA, THF, Diox, DMSO, aromatics (eg TL, XY) and any combination thereof, preferably DMF, THF or DCM.
当LG为卤素时:When LG is halogen:
在本发明的一些实施方案中,该反应在适合的卤代试剂的存在下进行,所述卤代试剂可选自SOCl2、POBr3和POCl3,优选SOCl2。In some embodiments of the invention, the reaction is carried out in the presence of a suitable halogenating reagent, which may be selected from SOCl 2 , POBr 3 and POCl 3 , preferably SOCl 2 .
在本发明的一些实施方案中,该反应在适合的温度下进行,所述温度为0-200℃,优选50-150℃。In some embodiments of the present invention, the reaction is carried out at a suitable temperature, said temperature being 0-200°C, preferably 50-150°C.
当LG为苯氧基时:When LG is phenoxy:
在本发明的一些实施方案中,该反应可通过先后进行卤代反应和进一步取代反应来发生。所述卤代反应在适合的卤代试剂的存在下进行,所述卤代试剂可选自SOCl2、POBr3和POCl3,优选SOCl2;所述进一步取代反应在合适的取代试剂的存在下进行,所述取代试剂可选自苯酚和苯酚钠。In some embodiments of the present invention, this reaction may occur by performing a halogenation reaction followed by a further substitution reaction. The halogenation reaction is carried out in the presence of a suitable halogenating reagent, which can be selected from SOCl 2 , POBr 3 and POCl 3 , preferably SOCl 2 ; the further substitution reaction is carried out in the presence of a suitable substitution reagent carried out, the substitution reagent may be selected from phenol and sodium phenate.
在本发明的一些实施方案中,所述进一步取代反应在适合的碱的存在下进行,所述碱包括有机碱或无机碱,所述有机碱可选自DIPEA、TEA、t-BuOK和Py,所述无机碱可选自K3PO4、NaH、K2CO3、Na2CO3、Cs2CO3和NaOH,优选NaH。In some embodiments of the present invention, the further substitution reaction is carried out in the presence of a suitable base, including an organic base or an inorganic base, the organic base may be selected from DIPEA, TEA, t-BuOK and Py, The inorganic base may be selected from K3PO4 , NaH , K2CO3 , Na2CO3 , Cs2CO3 and NaOH , preferably NaH .
在本发明的一些实施方案中,所述进一步取代反应在适合的温度下进行,所述温度为0-200℃,优选0-80℃。In some embodiments of the present invention, the further substitution reaction is carried out at a suitable temperature, the temperature being 0-200°C, preferably 0-80°C.
步骤1-4:化合物IM3和SM3发生取代反应得到式I化合物Step 1-4: Substitution reaction of compound IM3 and SM3 to obtain compound of formula I
在本发明的一些实施方案中,该反应在适合的有机溶剂中进行,所述有机溶剂可选自卤代烃类(例如二氯甲烷、氯仿、1,2-二氯乙烷等)、腈类(例如乙腈等)、N-甲基吡咯烷酮、DMF、DMA、四氢呋喃、二氧六环、DMSO及其任意组合,优选DMSO或DMF。In some embodiments of the present invention, the reaction is carried out in a suitable organic solvent, which may be selected from halogenated hydrocarbons (eg, dichloromethane, chloroform, 1,2-dichloroethane, etc.), nitrile (eg acetonitrile, etc.), N-methylpyrrolidone, DMF, DMA, tetrahydrofuran, dioxane, DMSO and any combination thereof, preferably DMSO or DMF.
在本发明的一些实施方案中,该反应在适合的碱的存在下进行,所述碱包括有机碱或无机碱,所述有机碱可选自DIPEA、三乙胺、叔丁醇钾和吡啶,所述无机碱可选自磷酸钾、氢化钠、碳酸钾、碳酸钠、碳酸铯、氢氧化锂和氢氧化钠,优选碳酸钾、氢氧化锂或氢化钠。In some embodiments of the invention, the reaction is carried out in the presence of a suitable base, including an organic base or an inorganic base, the organic base may be selected from DIPEA, triethylamine, potassium tert-butoxide, and pyridine, The inorganic base may be selected from potassium phosphate, sodium hydride, potassium carbonate, sodium carbonate, cesium carbonate, lithium hydroxide and sodium hydroxide, preferably potassium carbonate, lithium hydroxide or sodium hydride.
在本发明的一些实施方案中,该反应在适合的温度下进行,所述反应温度为0-200℃,优选50-150℃。In some embodiments of the present invention, the reaction is carried out at a suitable temperature, the reaction temperature being 0-200°C, preferably 50-150°C.
药物组合物和药盒Pharmaceutical compositions and kits
本发明的另一目的在于提供一种药物组合物,其包含至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药,以及一种或多种药学上可接受的载体。Another object of the present invention is to provide a pharmaceutical composition comprising at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, A solvate, nitrogen oxide, isotopic label, metabolite or prodrug, and one or more pharmaceutically acceptable carriers.
本发明的另一目的在于提供一种药盒,其包含:Another object of the present invention is to provide a kind of medicine kit, it comprises:
a)作为第一治疗剂的至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药,或者作为第一药物组合物的本发明的药物组合物;a) at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitroxide, an isotopic label, metabolite or prodrug, or a pharmaceutical composition of the invention as the first pharmaceutical composition;
b)任选存在的作为第二治疗剂的至少一种其他治疗剂,或者作为第二药物组合物的包含其他治疗剂的药物组合物;和b) optionally present at least one other therapeutic agent as a second therapeutic agent, or a pharmaceutical composition comprising the other therapeutic agent as a second pharmaceutical composition; and
c)任选存在的包装和/或说明书。c) Optional packaging and/or instructions.
术语“药学上可接受的载体”是指与治疗剂一同给药的辅料,并且其在合理的医学判断的范围内适于接触人类和/或其它动物的组织而没有过度的毒性、刺激性、过敏反应或与合理的益处/风险比相应的其它问题或并发症。The term "pharmaceutically acceptable carrier" refers to an excipient with which a therapeutic agent is administered and which, within the scope of sound medical judgment, is suitable for contact with human and/or other animal tissue without undue toxicity, irritation, Allergic reactions or other problems or complications with a reasonable benefit/risk ratio.
本发明的药物组合物可以系统地作用和/或局部地作用。为此目的,它们可以通过适合的途径给药,例如通过胃肠外、局部、静脉内、口服、皮下、动脉内、真皮内、经皮、直肠、颅内、腹膜内、鼻内、肌内途径或作为吸入剂给药。The pharmaceutical compositions of the present invention may act systemically and/or locally. For this purpose they can be administered by suitable routes, for example by parenteral, topical, intravenous, oral, subcutaneous, intraarterial, intradermal, transdermal, rectal, intracranial, intraperitoneal, intranasal, intramuscular route or as an inhalant.
对于这些给药途径,可以适合的剂型给药本发明的药物组合物。在本发明中可使用的剂型包括(但不限于):片剂、胶囊剂、锭剂、硬糖剂、散剂、喷雾剂、乳膏剂、软膏剂、栓剂、凝胶剂、糊剂、洗剂、软膏剂、水性混悬剂、可注射溶液剂、酏剂、糖浆剂等。For these routes of administration, the pharmaceutical compositions of the present invention can be administered in suitable dosage forms. Dosage forms that can be used in the present invention include, but are not limited to: tablets, capsules, lozenges, hard candies, powders, sprays, creams, ointments, suppositories, gels, pastes, lotions , ointments, aqueous suspensions, injectable solutions, elixirs, syrups, etc.
当口服给药时,可将本发明的药物组合物制成任意口服可接受的制剂形式,包括(但不限于)片剂、胶囊剂、水溶液剂、水混悬剂等。其中,片剂中使用的载体一般包括乳糖和玉米淀粉,另外也可加入润滑剂,例如硬脂酸镁。胶囊剂中使用的载体一般包括乳糖和干燥玉米淀粉。水混悬剂则通常通过将API与适宜的乳化剂和助悬剂混合而制成。任选地,以上口服制剂中还可加入一些甜味剂、芳香剂、着色剂等。When administered orally, the pharmaceutical compositions of the present invention can be formulated into any orally acceptable formulation, including but not limited to tablets, capsules, aqueous solutions, aqueous suspensions, and the like. Among them, the carriers used in tablets generally include lactose and corn starch, and lubricants such as magnesium stearate may also be added. Carriers used in capsules typically include lactose and dried cornstarch. Aqueous suspensions are usually prepared by mixing the API with suitable emulsifying and suspending agents. Optionally, some sweetening agents, flavoring agents, coloring agents, etc. may also be added to the above oral formulations.
本发明的药物组合物还可以无菌注射剂的形式给药,包括无菌注射水或油混悬剂,或者无菌注射水或油溶液剂。其中,可使用的载体包括(但不限于):水、林格氏溶液和等渗氯化钠溶液。另外,灭菌的非挥发油也可用作溶剂或悬浮介质,如单甘油酯或二甘油酯。The pharmaceutical compositions of the present invention can also be administered in the form of sterile injectable preparations, including sterile injectable aqueous or oily suspensions, or sterile injectable aqueous or oily solutions. Among them, the carriers that can be used include (but are not limited to): water, Ringer's solution and isotonic sodium chloride solution. In addition, sterile fixed oils can also be employed as a solvent or suspending medium, such as mono- or diglycerides.
本发明的药物组合物可以包含0.01mg至1000mg的至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药。The pharmaceutical compositions of the present invention may contain from 0.01 mg to 1000 mg of at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate thereof , nitrogen oxides, isotopic labels, metabolites or prodrugs.
在一些实施方案中,本发明提供了制备本发明的药物组合物或药物制剂的方法,所述方法包括将至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药与一种或多种药学上可接受的载体组合。In some embodiments, the present invention provides a method of preparing a pharmaceutical composition or pharmaceutical formulation of the present invention, the method comprising combining at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer thereof , tautomer, polymorph, solvate, nitrogen oxide, isotopic label, metabolite or prodrug in combination with one or more pharmaceutically acceptable carriers.
本发明的药盒可以包含0.01mg至1000mg的至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药。The kits of the present invention may contain from 0.01 mg to 1000 mg of at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, Nitrogen oxides, isotopic labels, metabolites or prodrugs.
在一些实施方案中,本发明提供了制备本发明的药盒的方法,所述方法包括将至少一种本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物与任选存在的至少一种其他治疗剂或者包含其他治疗剂的药物组合物、包装和/或说明书组合。In some embodiments, the present invention provides a method of making a kit of the present invention, the method comprising combining at least one compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer thereof Conforms, polymorphs, solvates, nitrogen oxides, isotopic labels, metabolites or prodrugs or pharmaceutical compositions of the invention with optionally at least one other therapeutic agent or a drug comprising other therapeutic agent Composition, Packaging and/or Instructions Combination.
治疗方法和用途Treatment methods and uses
本发明的化合物能够对TGFβR1表现出较强的抑制作用,IC50值能够达到100nM以下,个别甚至达到10nM以下,同时对TGFβR2表现出较弱的抑制作用,可以用作TGFβR1抑制剂(尤其是TGFβR1选择性抑制剂)。The compounds of the present invention can show a strong inhibitory effect on TGFβR1, the IC 50 value can reach below 100nM, some even below 10nM, and at the same time show a weak inhibitory effect on TGFβR2, and can be used as TGFβR1 inhibitors (especially TGFβR1 selective inhibitor).
本发明的另一目的在于提供本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物,其用作TGFβR1抑制剂,用于预防和/或治疗至少部分由TGFβR1介导的疾病或病症(尤其是癌症,例如肝癌)。Another object of the present invention is to provide the compounds of the present invention or their pharmaceutically acceptable salts, esters, stereoisomers, tautomers, polymorphs, solvates, nitrogen oxides, isotopic labels , metabolites or prodrugs or pharmaceutical compositions of the invention for use as TGFβR1 inhibitors for the prevention and/or treatment of diseases or disorders (especially cancers, eg liver cancer) mediated at least in part by TGFβR1.
本发明的另一目的在于提供本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物用作TGFβR1抑制剂的用途。Another object of the present invention is to provide the compounds of the present invention or their pharmaceutically acceptable salts, esters, stereoisomers, tautomers, polymorphs, solvates, nitrogen oxides, isotopic labels Use of a , metabolite or prodrug or a pharmaceutical composition of the invention as an inhibitor of TGFβR1.
本发明的另一目的在于提供本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物在制备用于预防和/或治疗至少部分由TGFβR1介导的疾病或病症(尤其是癌症,例如肝癌)的药物中的用途。Another object of the present invention is to provide the compounds of the present invention or their pharmaceutically acceptable salts, esters, stereoisomers, tautomers, polymorphs, solvates, nitrogen oxides, isotopic labels , metabolite or prodrug or use of a pharmaceutical composition of the invention in the manufacture of a medicament for the prevention and/or treatment of a disease or disorder mediated at least in part by TGFβR1, especially cancer, eg liver cancer.
本发明的另一目的在于提供一种用于预防和/或治疗至少部分由TGFβR1介导的疾病或病症(尤其是癌症,例如肝癌)的方法,其包括向有此需要的个体给药预防和/或治疗有效量的本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物。Another object of the present invention is to provide a method for the prevention and/or treatment of a disease or disorder mediated at least in part by TGFβR1, in particular cancer, such as liver cancer, comprising administering to an individual in need thereof prophylactic and A therapeutically effective amount of a compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitrogen oxide, isotopic label, metabolite drug or prodrug or pharmaceutical composition of the present invention.
术语“至少部分由TGFβR1介导的疾病或病症”是指发病机理中至少包含一部分与TGFβR1有关的因素的疾病,这些疾病包括(但不限于)癌症或纤维化疾病。The term "disease or disorder mediated at least in part by TGF[beta]Rl" refers to diseases in which at least a portion of the pathogenesis of TGF[beta]Rl is involved, including but not limited to cancer or fibrotic diseases.
术语“有效量”是指能够诱发细胞、组织、器官或生物体(例如个体)产生生物或医学反应,并且足以实现所需预防和/或治疗效果的剂量。The term "effective amount" refers to an amount sufficient to induce a biological or medical response in a cell, tissue, organ or organism (eg, an individual) and which is sufficient to achieve the desired prophylactic and/or therapeutic effect.
可调整给药方案以提供最佳所需响应。例如,可单次给药,可随时间分剂量给药,或可根据实际情况按比例减少或增加剂量后给药。可以理解的是,对于任何特定个体,具体的给药方案应根据需要以及给药组合物或监督组合物的给药人员的专业判断而调整。Dosage regimens can be adjusted to provide the optimal desired response. For example, it may be administered in a single dose, may be administered in divided doses over time, or may be administered in proportionately reduced or increased doses as appropriate. It will be appreciated that, for any particular individual, the specific dosing regimen should be adjusted as needed and according to the professional judgment of the person administering the composition or supervising the administration of the composition.
本发明的化合物的给药量将取决于个体情况、疾病或病症的严重性、给药的速率、化合物的处置及处方医师的判断。一般而言,有效量为约0.001-10000mg/kg受试者体重/天。在合适的情况下,有效量为约0.01-1000mg/kg受试者体重/天。可以每天、每两天或每三天给药约0.01-1000mg/kg受试者体重,通常约0.1-500mg/kg受试者体重。示例性的给药方案为每天一次或多次,或者每周一次或多次,或者每月一次或多次。多次给药时,单次剂量之间的间隔通常可以是每天、每周、每月或每年。或者,可以缓释制剂的形式给药,在这种情况下需要较低的给药频率。给药剂量和频率可根据药物在受试者体内的半衰期而不同,也可以根据是预防性应用还是治疗性应用而不同。在预防性应用中,以相对低频率的间隔长期施用相对低的剂量;在治疗性应用中,有时需要以较短的间隔施用相对高的剂量,直至疾病的进展被延缓或停止,优选直至个体表现出疾病症状的部分或完全改善,此后可以采用预防性应用。The amount of a compound of the present invention administered will depend on the individual circumstances, the severity of the disease or disorder, the rate of administration, the disposition of the compound, and the judgment of the prescribing physician. In general, an effective amount is about 0.001-10000 mg/kg subject body weight/day. Where appropriate, an effective amount is about 0.01-1000 mg/kg subject body weight/day. About 0.01-1000 mg/kg of the subject's body weight can be administered daily, every two days, or every three days, usually about 0.1-500 mg/kg of the subject's body weight. Exemplary dosing regimens are one or more times per day, or one or more times per week, or one or more times per month. In the case of multiple administrations, the interval between single doses can generally be daily, weekly, monthly or yearly. Alternatively, it may be administered in a sustained release formulation, in which case less frequent dosing is required. The dose and frequency of administration can vary depending on the half-life of the drug in the subject, as well as whether it is for prophylactic or therapeutic use. In prophylactic applications, relatively low doses are administered chronically at relatively infrequent intervals; in therapeutic applications, it is sometimes necessary to administer relatively high doses at shorter intervals until progression of the disease is delayed or stopped, preferably until the individual Shows partial or complete improvement in disease symptoms, after which prophylactic applications can be employed.
术语“治疗”是指减轻或消除所针对的疾病或病症。如果受试者接受了治疗量的本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物,该受试者的至少一种指标和症状表现出可观察到的和/或可检测出的缓解和/或改善,则表明该受试者已被成功地“治疗”。可以理解的是,治疗不仅包括完全地治疗,还包括未达到完全地治疗,但实现了一些生物学或医学相关的结果。具体而言,“治疗”表示本发明的化合物或其药学上可接受的盐、酯、立体异构体、互变异构体、多晶型物、溶剂合物、氮氧化物、同位素标记物、代谢物或前药或者本发明的药物组合物可以实现下列效果中的至少一种,例如:(1)在可能有疾病倾向,但尚未经历或显示疾病病理学或症状学的动物中防止疾病发生;(2)在正在经历或显示疾病病理学或症状学的动物中抑制疾病(即阻止病理学和/或症状学的进一步发展);(3)在正在经历或显示疾病病理学或症状学的动物中改善疾病(即逆转病理学和/或症状学)。The term "treating" refers to alleviating or eliminating the targeted disease or condition. If the subject receives a therapeutic amount of a compound of the present invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitrogen oxide, isotopic label A substance, metabolite or prodrug or a pharmaceutical composition of the present invention, the subject exhibits observable and/or detectable alleviation and/or improvement in at least one indicator and symptom, indicating that the subject have been successfully "treated". It is understood that treatment includes not only complete treatment, but also incomplete treatment, but the achievement of some biologically or medically relevant result. In particular, "treatment" means a compound of the invention or a pharmaceutically acceptable salt, ester, stereoisomer, tautomer, polymorph, solvate, nitrogen oxide, isotopic label thereof , metabolites or prodrugs, or pharmaceutical compositions of the present invention may achieve at least one of the following effects, for example: (1) prevention of disease in animals that may be predisposed to disease but have not experienced or displayed disease pathology or symptomatology Occurs; (2) inhibits disease (i.e. prevents further progression of the pathology and/or symptomology) in an animal that is experiencing or exhibits disease pathology or symptomology; (3) occurs in animals experiencing or showing disease pathology or symptomology improved disease (ie, reversal of pathology and/or symptomatology) in animals.
术语“预防”是指预先施用药物以避免或预防疾病或病症的一种或多种症状的出现。医学领域的普通技术人员认识到术语“预防”不是绝对术语。在医学领域中,应理解为预防性施用药物以基本上减少病症的可能性或严重性或病症的症状,这是本公开内容中意图的意义。如其中所使用的,关于病症或疾病的术语“预防”是指在疾病或病症完全表现出来之前避免疾病或病症的原因、作用、症状或进展。The term "prevention" refers to the pre-administration of a drug to avoid or prevent the appearance of one or more symptoms of a disease or disorder. One of ordinary skill in the medical arts recognizes that the term "prevention" is not an absolute term. In the medical arts, prophylactic administration of a drug to substantially reduce the likelihood or severity of a disorder or symptoms of a disorder is understood in the intended sense of this disclosure. As used herein, the term "preventing" with respect to a condition or disorder refers to avoiding the cause, effect, symptom, or progression of the disease or disorder until the disease or disorder is fully manifested.
术语“施用”或“给药”是指将药物活性成分(比如本发明的化合物)或包含药物活性成分的药物组合物(例如本发明的药物组合物)应用于个体或其细胞、组织、器官、生物流体等部位,以便使药物活性成分或药物组合物与个体或其细胞、组织、器官、生物流体等部位接触的过程。常见的施用方式包括(但不限于)口服施用、皮下施用、肌内施用、腹膜下施用、眼部施用、鼻部施用、舌下施用、直肠施用、阴道施用等。The term "administration" or "administration" refers to the application of a pharmaceutically active ingredient (such as a compound of the present invention) or a pharmaceutical composition comprising a pharmaceutically active ingredient (eg, a pharmaceutical composition of the present invention) to an individual or its cells, tissues, organs , biological fluids, etc., in order to make a pharmaceutical active ingredient or pharmaceutical composition come into contact with an individual or its cells, tissues, organs, biological fluids, etc. Common modes of administration include, but are not limited to, oral, subcutaneous, intramuscular, subperitoneal, ocular, nasal, sublingual, rectal, vaginal, and the like.
术语“有此需要”是指医生或其他护理人员对个体需要或者将要从预防和/或治疗过程中获益的判断,该判断的得出基于医生或其他护理人员在其专长领域中的各种因素。The term "needs" refers to a physician or other caregiver's judgment that an individual needs or will benefit from a prophylactic and/or therapeutic procedure based on the physician's or other caregiver's various areas of expertise. factor.
术语“个体”(或称受试者)包括人类或非人动物。本发明的个体包括患有疾病和/或病症的个体(患者)和正常的个体。本发明中的非人动物包括所有脊椎动物,例如非哺乳动物,例如鸟类、两栖类、爬行类等;和哺乳动物,例如非人灵长类、家畜和/或驯化动物(例如绵羊、犬、猫、奶牛、猪等)。The term "individual" (or subject) includes humans or non-human animals. Individuals of the present invention include individuals (patients) with a disease and/or disorder and normal individuals. Non-human animals in the present invention include all vertebrates, such as non-mammals, such as birds, amphibians, reptiles, etc.; and mammals, such as non-human primates, livestock and/or domesticated animals (such as sheep, dogs, etc.) , cats, cows, pigs, etc.).
实施例Example
为了使本发明的目的和技术方案更加清楚,以下结合实施例对本发明的实施方案进行详细描述。但是本领域技术人员将会理解,下列实施例仅用于说明本发明,而不应视为限定本发明的范围。In order to make the objectives and technical solutions of the present invention clearer, the embodiments of the present invention are described in detail below with reference to the examples. However, those skilled in the art will understand that the following examples are only used to illustrate the present invention and should not be construed as limiting the scope of the present invention.
实施例中所使用的试剂或仪器均为可以通过市购获得的常规产品。未注明具体条件者,均按照常规条件或制造商建议的条件进行。本发明中所使用的术语“室温”是指20℃±5℃。在用于修饰某一数值或数值范围时,本发明中所使用的术语“约”是指包括该数值或数值范围以及该数值或数值范围的本领域技术人员可接受的误差范围,例如该误差范围为±10%、±5%、±4%、±3%、±2%、±1%、±0.5%等。The reagents or instruments used in the examples are conventional products that can be obtained commercially. If the specific conditions are not specified, it is carried out in accordance with the conventional conditions or the conditions suggested by the manufacturer. The term "room temperature" used in the present invention refers to 20°C ± 5°C. When used to modify a value or range of values, the term "about" as used herein is meant to include the value or range of values and the range of errors acceptable to those skilled in the art for the value or range of values, such as the error The range is ±10%, ±5%, ±4%, ±3%, ±2%, ±1%, ±0.5%, etc.
在常规的合成法以及实施例和中间体合成例中,各缩写的含义如下表所示。In the conventional synthesis methods, examples and intermediate synthesis examples, the meanings of each abbreviation are shown in the following table.
以下实施例中记载的化合物的结构通过核磁共振(1H-NMR)和/或质谱(MS)来确定。The structures of the compounds described in the following examples were determined by nuclear magnetic resonance ( 1 H-NMR) and/or mass spectrometry (MS).
核磁共振(1H-NMR)的测定仪器使用Bruker 400MHz核磁共振仪,测定溶剂为氘代甲醇(CD3OD)、氘代氯仿(CDCl3)、六氘代二甲基亚砜(DMSO-d6),内标物质为四甲基硅烷(TMS)。The measuring instrument of nuclear magnetic resonance ( 1 H-NMR) was a Bruker 400 MHz nuclear magnetic resonance apparatus, and the measuring solvents were deuterated methanol (CD 3 OD), deuterated chloroform (CDCl 3 ), hexadeuterated dimethyl sulfoxide (DMSO-d) 6 ), the internal standard substance is tetramethylsilane (TMS).
以下实施例中的核磁共振(NMR)数据中的缩写代表的含义如下:The meanings of the abbreviations in the nuclear magnetic resonance (NMR) data in the following examples are as follows:
s:单峰(singlet)、d:二重峰(doublet)、t:三重峰(triplet)、q:四重峰(quartet)、dd:双二重峰(double doublet)、qd:四二重峰(quartet doublet)、ddd:双双二重峰(double double doublet)、ddt:双双三重峰(double double triplet)、dddd:双双双二重峰(double double double doublet)、m:多重峰(multiplet)、br:宽峰(broad)、J:偶合常数、Hz:赫兹、δ:化学位移。s: singlet (singlet), d: doublet (doublet), t: triplet (triplet), q: quartet (quartet), dd: double doublet (double doublet), qd: quadruplet quartet doublet, ddd: double double doublet, ddt: double double triplet, dddd: double double double doublet, m: multiplet , br: broad, J: coupling constant, Hz: Hertz, δ: chemical shift.
全部化学位移(δ)值以百万分之一(ppm)的单位给出。All chemical shift (δ) values are given in parts per million (ppm).
质谱(MS)的测定仪器使用Agilent 6120B质谱仪,离子源为电喷雾离子源(ESI)。An Agilent 6120B mass spectrometer was used as the measuring instrument for mass spectrometry (MS), and the ion source was an electrospray ion source (ESI).
本发明的实施例采用如下所示方法进行制备型高效液相色谱(Prep-HPLC)纯化。Examples of the present invention were purified by preparative high performance liquid chromatography (Prep-HPLC) using the method shown below.
方法A:Method A:
色谱柱:YMC(5μm*19mm*150mm)Chromatographic column: YMC (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%三氟乙酸)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% trifluoroacetic acid)
方法B:Method B:
色谱柱:Sunfire C18 OBD(5μm*19mm*150mm)Column: Sunfire C 18 OBD (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%甲酸)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% formic acid)
方法C:Method C:
色谱柱:Sunfire C18 OBD(5μm*19mm*150mm)Chromatographic column: Sunfire C18 OBD (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%碳酸氢铵)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% ammonium bicarbonate)
方法D:Method D:
色谱柱:Sunfire C18 OBD(5μm*19mm*150mm)Column: Sunfire C 18 OBD (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%甲酸)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% formic acid)
方法E:Method E:
色谱柱:Waters XBridge Prep C18 OBD 5μm 19x 150mmColumn: Waters XBridge Prep C18 OBD 5μm 19x 150mm
流动相A:乙腈;流动相B:水(含有0.05%碳酸氢铵)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% ammonium bicarbonate)
方法F:Method F:
色谱柱:Sunfire C18 OBD(5μm*19mm*150mm)Column: Sunfire C 18 OBD (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%甲酸)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% formic acid)
方法G:Method G:
色谱柱:Sunfire C18 OBD(5μm*19mm*150mm)Column: Sunfire C 18 OBD (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%碳酸氢钠)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% sodium bicarbonate)
方法H:Method H:
色谱柱:C18 OBD(5μm*19mm*150mm)Chromatographic column: C 18 OBD (5μm*19mm*150mm)
流动相A:乙腈;流动相B:水(含有0.05%碳酸氢钠)Mobile phase A: acetonitrile; mobile phase B: water (containing 0.05% sodium bicarbonate)
化合物的制备Preparation of compounds
实施例一:4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟腈(化合物1)的合成Example 1: Synthesis of 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinonitrile (Compound 1)
步骤一:5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-醇(化合物1-2)的合成Step 1: Synthesis of 5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-ol (Compound 1-2)
25℃下,将2-甲酰3-甲基丁酸乙酯(化合物1-1,6g,37.93mmol)和6-甲基吡啶-2-甲酰胺(1.85g,13.69mmol)溶于甲醇(5mL)中,加入甲醇钠甲醇溶液(5mol/L,5mL),在120℃下反应16小时。将反应液用二氯甲烷(10mL*3)萃取,用饱和食盐水(10mL)洗涤,合并有机相,经无水硫酸钠干燥后浓缩得粗品,将粗品经硅胶柱层析分离纯化(洗脱剂:石油醚/乙酸乙酯=2/1(v/v))),得到标题化合物(810mg)。At 25°C, ethyl 2-formyl 3-methylbutanoate (compound 1-1, 6 g, 37.93 mmol) and 6-methylpyridine-2-carboxamide (1.85 g, 13.69 mmol) were dissolved in methanol ( 5 mL), sodium methoxide methanol solution (5 mol/L, 5 mL) was added, and the reaction was carried out at 120° C. for 16 hours. The reaction solution was extracted with dichloromethane (10 mL*3), washed with saturated brine (10 mL), the organic phases were combined, dried over anhydrous sodium sulfate, and concentrated to obtain a crude product, which was separated and purified by silica gel column chromatography (eluting solvent: petroleum ether/ethyl acetate = 2/1 (v/v))) to obtain the title compound (810 mg).
ESI-MS(m/z):230.1[M+H]+。ESI-MS (m/z): 230.1 [M+H] + .
步骤二:4-氯-5-异丙基-2-(6-甲基吡啶-2-基)嘧啶(化合物1-3)的合成Step 2: Synthesis of 4-chloro-5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidine (Compound 1-3)
25℃下,将5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-醇(化合物1-2,800mg,3.49mmol)溶于二氯亚砜(10mL)中,滴入两滴N,N-二甲基甲酰胺催化反应,在90℃下反应0.5小时。将反应液浓缩,用碳酸氢钠水溶液中和后,用乙酸乙酯(10mL)萃取3次,合并有机相,用饱和食盐水(10mL)洗涤,经无水硫酸钠干燥后浓缩得粗品。将粗品经硅胶柱层析分离纯化(洗脱剂:石油醚/乙酸乙酯=2/1(v/v))),得到标题化合物(850mg)。5-Isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-ol (compound 1-2, 800 mg, 3.49 mmol) was dissolved in thionyl chloride (10 mL) at 25°C , drop two drops of N,N-dimethylformamide to catalyze the reaction, and react at 90°C for 0.5 hours. The reaction solution was concentrated, neutralized with aqueous sodium bicarbonate solution, and extracted three times with ethyl acetate (10 mL). The organic phases were combined, washed with saturated brine (10 mL), dried over anhydrous sodium sulfate, and concentrated to obtain a crude product. The crude product was separated and purified by silica gel column chromatography (eluent: petroleum ether/ethyl acetate = 2/1 (v/v)) to obtain the title compound (850 mg).
ESI-MS(m/z):248.1[M+H]+。ESI-MS (m/z): 248.1 [M+H] + .
步骤三:5-异丙基-2-(6-甲基吡啶-2-基)-4-苯氧基嘧啶(化合物1-4)的合成Step 3: Synthesis of 5-isopropyl-2-(6-methylpyridin-2-yl)-4-phenoxypyrimidine (Compound 1-4)
25℃下,将4-氯-5-异丙基-2-(6-甲基吡啶-2-基)嘧啶(化合物1-4,850mg,3.43mmol)和苯酚钠(796.69mg,6.86mmol)溶于THF(5mL)中,在120℃下反应2小时。将反应液浓缩,用乙酸乙酯(10mL)萃取3次,合并有机相,用饱和食盐水(10mL)洗涤,经无水硫酸钠干燥后浓缩得粗品。将粗品经硅胶柱层析分离纯化(洗脱剂:石油醚/乙酸乙酯=1/1(v/v))),得到标题化合物(880mg).4-Chloro-5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidine (compound 1-4, 850 mg, 3.43 mmol) and sodium phenolate (796.69 mg, 6.86 mmol) were combined at 25°C It was dissolved in THF (5 mL) and reacted at 120°C for 2 hours. The reaction solution was concentrated, extracted three times with ethyl acetate (10 mL), the organic phases were combined, washed with saturated brine (10 mL), dried over anhydrous sodium sulfate, and concentrated to obtain a crude product. The crude product was separated and purified by silica gel column chromatography (eluent: petroleum ether/ethyl acetate = 1/1 (v/v)) to obtain the title compound (880 mg).
ESI-MS(m/z):306.1[M+H]+。ESI-MS (m/z): 306.1 [M+H] + .
步骤四:4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟腈(化合物1)的合成Step 4: Synthesis of 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinonitrile (Compound 1)
25℃下,将5-异丙基-2-(6-甲基吡啶-2-基)-4-苯氧基嘧啶(化合物1-4,30mg,98.24umol)和1H-吡唑并[4,3-c]吡啶(23.41mg,196.48umol)溶于DMSO(3mL)中,加入LiOH(8.06mg,196.48umol),在80℃下反应2小时。将反应液用乙酸乙酯(10mL)萃取3次,合并有机相,用饱和食盐水(10mL)洗涤,经无水硫酸钠干燥后浓缩得粗品。将粗品经高效液相色谱制备纯化(方法A),得标题化合物(22mg)。At 25 °C, 5-isopropyl-2-(6-methylpyridin-2-yl)-4-phenoxypyrimidine (compound 1-4, 30 mg, 98.24 umol) and 1H-pyrazolo[4 ,3-c]pyridine (23.41 mg, 196.48 umol) was dissolved in DMSO (3 mL), LiOH (8.06 mg, 196.48 umol) was added, and the reaction was carried out at 80° C. for 2 hours. The reaction solution was extracted three times with ethyl acetate (10 mL), and the organic phases were combined, washed with saturated brine (10 mL), dried over anhydrous sodium sulfate, and concentrated to obtain a crude product. The crude product was preparatively purified by high performance liquid chromatography (Method A) to give the title compound (22 mg).
ESI-MS(m/z):331.1[M+H]+。ESI-MS (m/z): 331.1 [M+H] + .
1H NMR(400MHz,DMSO-d6)δ9.01(s,1H),8.69(s,1H),8.34(s,1H),8.08–8.04(m,2H),7.84(d,J=6.1Hz,1H),7.66–7.62(m,1H),3.30(p,J=6.8Hz,1H),2.68(s,3H),1.35(d,J=6.8Hz,6H). 1 H NMR (400MHz, DMSO-d 6 )δ9.01(s,1H), 8.69(s,1H), 8.34(s,1H), 8.08-8.04(m,2H), 7.84(d, J=6.1 Hz, 1H), 7.66–7.62 (m, 1H), 3.30 (p, J=6.8Hz, 1H), 2.68 (s, 3H), 1.35 (d, J=6.8Hz, 6H).
实施例二:4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酰胺(化合物2)的合成Example 2: Synthesis of 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinamide (compound 2)
将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟腈(20mg,60.54umol)溶于四氢呋喃(3mL)中,加入氢氧化钠(4.84mg,121.07umol),再缓慢加入双氧水(2mL),在25℃下反应1小时。将反应液用乙酸乙酯(10mL*3)萃取,用饱和亚硫酸钠水溶液(10mL)洗涤,合并有机相。用无水硫酸钠干燥,然后减压蒸干得粗品,经高效液相色谱制备纯化(方法B),得到标题化合物(4mg)。4-((5-Isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinonitrile (20 mg, 60.54 umol) was dissolved in tetrahydrofuran (3 mL), hydrogen was added Sodium oxide (4.84 mg, 121.07 umol) was slowly added with hydrogen peroxide (2 mL), and the reaction was carried out at 25° C. for 1 hour. The reaction solution was extracted with ethyl acetate (10 mL*3), washed with saturated aqueous sodium sulfite solution (10 mL), and the organic phases were combined. It was dried over anhydrous sodium sulfate, and then evaporated to dryness under reduced pressure to obtain the crude product, which was preparatively purified by high performance liquid chromatography (Method B) to obtain the title compound (4 mg).
其结构表征如下:Its structure is characterized as follows:
1H NMR(400MHz,DMSO-d6)δ12.39(s,1H),9.44(d,J=5.9Hz,1H),8.97(s,1H),8.66–8.52(m,3H),8.17(d,J=7.7Hz,1H),8.00(s,1H),7.87(t,J=7.7Hz,1H),7.40(d,J=7.6Hz,1H),3.13(p,J=6.8Hz,1H),2.62(s,3H),1.36(d,J=6.8Hz,6H). 1 H NMR (400MHz, DMSO-d 6 )δ12.39(s,1H),9.44(d,J=5.9Hz,1H),8.97(s,1H),8.66-8.52(m,3H),8.17( d, J=7.7Hz, 1H), 8.00(s, 1H), 7.87(t, J=7.7Hz, 1H), 7.40(d, J=7.6Hz, 1H), 3.13(p, J=6.8Hz, 1H), 2.62(s, 3H), 1.36(d, J=6.8Hz, 6H).
ESI-MS(m/z):349.1[M+H]+。ESI-MS (m/z): 349.1 [M+H] + .
实施例三:N-(1,3-二羟基丙-2-基)-4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酰胺(化合物5)的合成Example three: N-(1,3-dihydroxypropan-2-yl)-4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino ) Synthesis of Nicotinamide (Compound 5)
步骤一:4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸甲酯(化合物5-1)的合成将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟腈(化合物1,1g,1.82mmol)溶解于MeOH(10mL)中,缓慢加入H2SO4(1.82mmol,5mL),加毕,升温至100℃,反应15小时。将反应液冷却至室温,用饱和碳酸氢钠水溶液调节至PH=8,然后用EA(50mL*3)萃取,合并有机相,用饱和食盐水(50mL)洗涤,经无水硫酸钠干燥,过滤,将滤液减压蒸干得到标题化合物(589mg)。Step 1: Synthesis of methyl 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinate (compound 5-1) (5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinonitrile (compound 1, 1 g, 1.82 mmol) was dissolved in MeOH (10 mL) and H was added slowly 2 SO 4 (1.82 mmol, 5 mL), the addition was completed, the temperature was raised to 100° C., and the reaction was carried out for 15 hours. The reaction solution was cooled to room temperature, adjusted to pH=8 with saturated aqueous sodium bicarbonate solution, then extracted with EA (50 mL*3), the organic phases were combined, washed with saturated brine (50 mL), dried over anhydrous sodium sulfate, and filtered. , the filtrate was evaporated to dryness under reduced pressure to obtain the title compound (589 mg).
ESI-MS(m/z):364.2[M+H]+。ESI-MS (m/z): 364.2 [M+H] + .
步骤二:N-(1,3-二羟基丙-2-基)-4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酰胺(化合物5)的合成Step 2: N-(1,3-Dihydroxypropan-2-yl)-4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino) Synthesis of Nicotinamide (Compound 5)
将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸甲酯(化合物5-1,22mg,0.06mmol)和2-氨基丙烷-1,3-二醇(55mg,0.6mmol)用DMSO(0.6mL)溶解,在100℃下反应3小时。将反应液冷却至室温,直接经高效液相色谱制备纯化(方法C),将制备液冻干后得到标题化合物(16mg)。其结构表征如下:Methyl 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinate (compound 5-1, 22 mg, 0.06 mmol) and 2- Aminopropane-1,3-diol (55 mg, 0.6 mmol) was dissolved in DMSO (0.6 mL) and reacted at 100°C for 3 hours. The reaction solution was cooled to room temperature, and directly purified by high performance liquid chromatography (Method C). The title compound (16 mg) was obtained after the preparation solution was lyophilized. Its structure is characterized as follows:
1H NMR(400MHz,DMSO-d6)δ11.8(s,1H),9.36(d,J=6.0Hz,1H),8.96(s,1H),8.70–8.57(m,3H),8.16(d,J=8.0Hz,1H),7.87(t,J=8.0Hz,1H),7.39(d,J=7.6Hz,1H),4.74(t,J=6.0Hz,2H),4.15–4.00(m,1H),3.67–3.45(m,4H),3.17–3.04(m,1H),2.62(s,3H),1.37(d,J=6.8Hz,6H). 1 H NMR (400MHz, DMSO-d 6 )δ11.8(s, 1H), 9.36(d, J=6.0Hz, 1H), 8.96(s, 1H), 8.70-8.57(m, 3H), 8.16( d, J=8.0Hz, 1H), 7.87(t, J=8.0Hz, 1H), 7.39(d, J=7.6Hz, 1H), 4.74(t, J=6.0Hz, 2H), 4.15–4.00( m, 1H), 3.67–3.45 (m, 4H), 3.17–3.04 (m, 1H), 2.62 (s, 3H), 1.37 (d, J=6.8Hz, 6H).
ESI-MS(m/z):423.2[M+H]+。ESI-MS (m/z): 423.2 [M+H] + .
实施例四:(S)-N-(2-羟丙基)-4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酰胺(化合物6)的合成Example 4: (S)-N-(2-hydroxypropyl)-4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinum Synthesis of Amide (Compound 6)
25℃下,将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸甲酯(化合物5-1,20mg,55.03umol)和(S)-1-氨基丙烷-2-醇(41.34mg,550.34μmol)溶于二甲基亚砜(1mL)中,升温至100℃,反应3小时。将反应液直接经高效液相色谱制备纯化(方法D),得标题化合物(8.5mg)。At 25°C, methyl 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinate (compound 5-1, 20mg, 55.03umol ) and (S)-1-aminopropan-2-ol (41.34 mg, 550.34 μmol) were dissolved in dimethyl sulfoxide (1 mL), and the temperature was raised to 100° C. and reacted for 3 hours. The reaction solution was directly purified by high performance liquid chromatography (Method D) to give the title compound (8.5 mg).
其结构表征如下:Its structure is characterized as follows:
1H NMR(400MHz,DMSO-d6)δ11.86(s,1H),9.38(d,J=5.9Hz,1H),9.00(t,J=5.8Hz,1H),8.93(s,1H),8.63–8.56(m,2H),8.20–8.13(m,1H),7.87(t,J=7.7Hz,1H),7.39(d,J=7.6Hz,1H),4.84(d,J=4.8Hz,1H),3.85(dd,J=6.4,5.0Hz,1H),3.27(t,J=5.9Hz,2H),3.17–3.05(m,1H),2.62(s,3H),1.36(dd,J=6.8,1.5Hz,6H),1.10(d,J=6.2Hz,3H). 1 H NMR (400MHz, DMSO-d 6 ) δ 11.86 (s, 1H), 9.38 (d, J=5.9Hz, 1H), 9.00 (t, J=5.8Hz, 1H), 8.93 (s, 1H) , 8.63–8.56 (m, 2H), 8.20–8.13 (m, 1H), 7.87 (t, J=7.7Hz, 1H), 7.39 (d, J=7.6Hz, 1H), 4.84 (d, J=4.8 Hz, 1H), 3.85(dd, J=6.4, 5.0Hz, 1H), 3.27(t, J=5.9Hz, 2H), 3.17–3.05(m, 1H), 2.62(s, 3H), 1.36(dd , J=6.8, 1.5Hz, 6H), 1.10(d, J=6.2Hz, 3H).
ESI-MS(m/z):407.1[M+H]+。ESI-MS (m/z): 407.1 [M+H] + .
实施例五:(R)-N-(2-羟基丙基)-4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酰胺(化合物7)的合成Embodiment 5: (R)-N-(2-hydroxypropyl)-4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinamide Synthesis of Amide (Compound 7)
将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸甲酯(化合物5-1,20mg,0.055mmol)和(R)-1-氨基丙烷-2-醇(41mg,0.55mmol)溶于DMSO(0.5mL)中,升温至100℃,反应2h。将反应液冷至室温后,经制备高效液相色谱(方法E)分离纯化,制备液经冷冻干燥,得标题化合物(12mg)。Methyl 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinate (compound 5-1, 20 mg, 0.055 mmol) and (R )-1-aminopropan-2-ol (41 mg, 0.55 mmol) was dissolved in DMSO (0.5 mL), the temperature was raised to 100 °C, and the reaction was carried out for 2 h. After cooling the reaction solution to room temperature, it was separated and purified by preparative high performance liquid chromatography (Method E), and the preparative solution was lyophilized to obtain the title compound (12 mg).
其结构表征如下:Its structure is characterized as follows:
1H NMR(400MHz,DMSO-d6)δ11.85(s,1H),9.37(d,J=5.9Hz,1H),9.00(t,J=5.7Hz,1H),8.93(s,1H),8.63-8.57(m,2H),8.16(d,J=7.7Hz,1H),7.86(t,J=7.7Hz,1H),7.39(d,J=7.6Hz,1H),4.84(d,J=4.8Hz,1H),3.85(dt,J=11.1,6.1Hz,1H),3.26(t,J=6.0Hz,2H),3.14-3.07(m,1H),2.62(s,3H),1.42-1.30(m,6H),1.10(d,J=6.2Hz,3H). 1 H NMR (400MHz, DMSO-d 6 )δ 11.85(s, 1H), 9.37(d, J=5.9Hz, 1H), 9.00(t, J=5.7Hz, 1H), 8.93(s, 1H) ,8.63-8.57(m,2H),8.16(d,J=7.7Hz,1H),7.86(t,J=7.7Hz,1H),7.39(d,J=7.6Hz,1H),4.84(d, J=4.8Hz, 1H), 3.85(dt, J=11.1, 6.1Hz, 1H), 3.26(t, J=6.0Hz, 2H), 3.14-3.07(m, 1H), 2.62(s, 3H), 1.42-1.30(m, 6H), 1.10(d, J=6.2Hz, 3H).
ESI-MS(m/z):407.2[M+H]+。ESI-MS (m/z): 407.2 [M+H]+.
实施例六:(3-羟基氮杂环丁-1-基)(4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)吡啶-3-基)甲酮(化合物14)的合成Embodiment 6: (3-hydroxyazetidin-1-yl)(4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)pyridine Synthesis of -3-yl)methanone (Compound 14)
步骤一:4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸(化合物14-1)的合成Step 1: Synthesis of 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinic acid (Compound 14-1)
25℃下,将硫酸(1mL)溶于水(1mL)中,再将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟腈(化合物1,100mg,302.68umol)加入到反应液中,在98℃下反应16h。将反应液冷却,在冰浴下,用1N NaOH水溶液调节PH至6~7,用二氯甲烷(10ml)萃取,将水相再用盐酸调PH至3~4,用正丁醇萃取,将有机相合并浓缩,经高效液相色谱制备纯化(方法F),得标题化合物(60mg)。At 25°C, sulfuric acid (1 mL) was dissolved in water (1 mL), followed by 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino) Nicotinonitrile (compound 1, 100 mg, 302.68 umol) was added to the reaction solution, and the reaction was carried out at 98° C. for 16 h. The reaction solution was cooled, adjusted to pH 6-7 with 1N NaOH aqueous solution under ice bath, extracted with dichloromethane (10ml), the aqueous phase was adjusted to pH 3-4 with hydrochloric acid, extracted with n-butanol, and the The organic phases were combined, concentrated, and purified by preparative high performance liquid chromatography (Method F) to give the title compound (60 mg).
ESI-MS(m/z):350.1[M+H]+。ESI-MS (m/z): 350.1 [M+H] + .
步骤二:(3-羟基氮杂环丁-1-基)(4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)吡啶-3-基)甲酮(化合物14)的合成Step 2: (3-Hydroxyazetidin-1-yl)(4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)pyridine- Synthesis of 3-yl)methanone (Compound 14)
25℃下,将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸(化合物14-1,20mg,57.24μmol)和氮杂环丁烷-3-醇(4.60mg,62.97μmol)溶于DMF(2mL)中,再依次加入TBTU(17.58mg,68.69μmol)和DIPEA(22.19mg,171.73μmol),在25℃下反应1小时,反应液直接经高效液相色谱制备纯化(方法G),得标题化合物(1.51mg)。At 25°C, 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinic acid (compound 14-1, 20 mg, 57.24 μmol) and Azetidine-3-ol (4.60mg, 62.97μmol) was dissolved in DMF (2mL), then TBTU (17.58mg, 68.69μmol) and DIPEA (22.19mg, 171.73μmol) were added in sequence, and the reaction was carried out at 25°C After 1 hour, the reaction solution was directly purified by high performance liquid chromatography (Method G) to give the title compound (1.51 mg).
其结构表征如下:Its structure is characterized as follows:
1H NMR(400MHz,DMSO-d6)δ11.23(s,1H),9.28(d,J=5.9Hz,1H),8.64(s,1H),8.61(s,1H),8.58(d,J=5.9Hz,1H),8.16(d,J=7.7Hz,1H),7.87(t,J=7.7Hz,1H),7.39(d,J=7.6Hz,1H),5.82(d,J=6.1Hz,1H),4.60(d,J=8.6Hz,1H),4.50(q,J=5.9,5.3Hz,1H),4.36(d,J=10.0Hz,1H),4.23(s,1H),3.87(d,J=10.8Hz,1H),3.07(p,J=6.8Hz,1H),2.61(s,3H),1.37(d,J=6.8Hz,6H). 1 H NMR (400MHz, DMSO-d 6 )δ11.23(s, 1H), 9.28(d, J=5.9Hz, 1H), 8.64(s, 1H), 8.61(s, 1H), 8.58(d, J=5.9Hz, 1H), 8.16 (d, J=7.7Hz, 1H), 7.87 (t, J=7.7Hz, 1H), 7.39 (d, J=7.6Hz, 1H), 5.82 (d, J= 6.1Hz,1H),4.60(d,J=8.6Hz,1H),4.50(q,J=5.9,5.3Hz,1H),4.36(d,J=10.0Hz,1H),4.23(s,1H) ,3.87(d,J=10.8Hz,1H),3.07(p,J=6.8Hz,1H),2.61(s,3H),1.37(d,J=6.8Hz,6H).
ESI-MS(m/z):405.1[M+H]+。ESI-MS (m/z): 405.1 [M+H] + .
实施例七:N-(3-羟基环丁基)-4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酰胺(化合物15)的合成Embodiment 7: N-(3-hydroxycyclobutyl)-4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinamide (compound 15) Synthesis
25℃下,将4-((5-异丙基-2-(6-甲基吡啶-2-基)嘧啶-4-基)氨基)烟酸(化合物14-1,20mg,57.24μmol)和3-氨基环丁烷-1-醇(5.49mg,62.97μmol)溶于DMF(2mL)中,再依次加入TBTU(14.65mg,57.24μmol)和DIPEA(22.19mg,171.73μmol),在25℃下反应1小时,反应液直接经高效液相色谱制备纯化(方法H),得标题化合物(1.35mg)。At 25°C, 4-((5-isopropyl-2-(6-methylpyridin-2-yl)pyrimidin-4-yl)amino)nicotinic acid (compound 14-1, 20 mg, 57.24 μmol) and 3-Aminocyclobutan-1-ol (5.49 mg, 62.97 μmol) was dissolved in DMF (2 mL), followed by adding TBTU (14.65 mg, 57.24 μmol) and DIPEA (22.19 mg, 171.73 μmol), at 25°C After reacting for 1 hour, the reaction solution was directly purified by high performance liquid chromatography (Method H) to obtain the title compound (1.35 mg).
其结构表征如下:Its structure is characterized as follows:
1H NMR(400MHz,DMSO-d6)δ11.90–11.80(m,1H),9.37(d,J=5.9Hz,1H),9.15(t,J=7.6Hz,1H),8.93(s,1H),8.59(d,J=8.4Hz,2H),8.16(d,J=7.8Hz,1H),7.87(t,J=7.7Hz,1H),7.40(d,J=7.6Hz,1H),5.20–5.10(m,1H),4.53–3.68(m,2H),3.10(p,J=6.7Hz,1H),2.65–2.55(m,5H),2.32–2.16(m,1H),1.99–1.95(m,2H),1.40–1.30(m,6H). 1 H NMR (400MHz, DMSO-d 6 ) δ 11.90-11.80 (m, 1H), 9.37 (d, J=5.9Hz, 1H), 9.15 (t, J=7.6Hz, 1H), 8.93 (s, 1H), 8.59(d, J=8.4Hz, 2H), 8.16(d, J=7.8Hz, 1H), 7.87(t, J=7.7Hz, 1H), 7.40(d, J=7.6Hz, 1H) , 5.20–5.10 (m, 1H), 4.53–3.68 (m, 2H), 3.10 (p, J=6.7Hz, 1H), 2.65–2.55 (m, 5H), 2.32–2.16 (m, 1H), 1.99 –1.95(m,2H),1.40–1.30(m,6H).
ESI-MS(m/z):419.1[M+H]+。ESI-MS (m/z): 419.1 [M+H] + .
药理活性测试Pharmacological activity test
试验例一:体外酶学活性抑制试验(TGFβR1)。Test Example 1: In vitro enzymatic activity inhibition test (TGFβR1).
实验方法:根据ADP-GloTM激酶检测试剂盒(Promega)的说明测定本发明的化合物对TGFβR1酶活性的抑制作用,步骤如下:Experimental method: The inhibitory effect of the compounds of the present invention on the enzymatic activity of TGFβR1 was determined according to the instructions of ADP-Glo ™ Kinase Detection Kit (Promega). The steps are as follows:
将TGFβR1酶分别与不同浓度的测试化合物(1000nM、100nM、10nM)在30℃下预孵育30min后,加入TGFβR1肽和三磷酸腺苷(ATP)启动反应。在30℃下孵育3h后加入ADP-GloTM试剂,室温下孵育90min后加入激酶检测试剂。室温下孵育30min后进行检测化学发光信号值。以溶剂组(DMSO)为阴性对照,缓冲液组(不含TGFβR1酶)为空白对照,按照下式计算不同浓度化合物的百分比抑制率:After pre-incubating TGFβR1 enzyme with different concentrations of test compounds (1000 nM, 100 nM, 10 nM) at 30°C for 30 min, TGFβR1 peptide and adenosine triphosphate (ATP) were added to initiate the reaction. ADP-Glo TM reagent was added after incubation at 30°C for 3 h, and kinase detection reagent was added after incubation at room temperature for 90 min. Chemiluminescence signal value was detected after 30min incubation at room temperature. Taking the solvent group (DMSO) as the negative control and the buffer group (without TGFβR1 enzyme) as the blank control, the percentage inhibition rate of different concentrations of compounds was calculated according to the following formula:
百分比抑制率=(1-(不同浓度化合物的化学发光信号值-空白对照的化学发光信号值)/(阴性对照的化学发光信号值-空白对照的化学发光信号值))×100%;Percent inhibition rate=(1-(chemiluminescence signal value of compounds with different concentrations-chemiluminescence signal value of blank control)/(chemiluminescence signal value of negative control-chemiluminescence signal value of blank control))×100%;
当百分比抑制率介于30-80%时,按照下述公式计算化合物的半数抑制浓度(IC50)或范围:When the percent inhibition was between 30-80%, the median inhibitory concentration ( IC50 ) or range of the compound was calculated according to the following formula:
IC50=X×(1-百分比抑制率(%))/百分比抑制率(%),其中:X为化合物的测试浓度。 IC50 =XX(1-percent inhibition (%))/percent inhibition (%), where: X is the test concentration of the compound.
实验结果如下表1所示:The experimental results are shown in Table 1 below:
表1.本发明的化合物对TGFβR1的抑制活性Table 1. Inhibitory activity of compounds of the invention on TGFβR1
由表1可以看出,本发明的化合物对TGFβR1具有明显的抑制作用。It can be seen from Table 1 that the compounds of the present invention have obvious inhibitory effect on TGFβR1.
试验例二:体外细胞活性抑制试验。Test Example 2: In vitro cell activity inhibition test.
实验方法:根据Bright-GloTM荧光素酶检测试剂盒(Promega)的说明测定本发明的化合物对HEK293-SBE细胞TGFβ/Smad信号通路的抑制作用,步骤如下:Experimental method: The inhibitory effect of the compounds of the present invention on the TGFβ/Smad signaling pathway in HEK293-SBE cells was determined according to the instructions of Bright-Glo TM luciferase detection kit (Promega). The steps are as follows:
将HEK293-SBE细胞(Bps bioscience)加入到96孔板(含10%FBS的MEM培养基)中,3万/孔,37℃,5%CO2培养过夜。将培养基换成0.5%FBS培养基,并加入0.5%FBS培养基稀释的测试化合物,测试化合物最高终浓度为10μM,化合物4倍稀释,共8个浓度梯度。培养4-5小时后,加入10μl TGFβ。TGFβ的终浓度为0.5ng/ml。加入10μl培养基以代替TGFβ作为阴性对照组。空白对照中不加测试化合物,加入TGFβ。每孔加入Bright Glo试剂,在酶标仪上读取化学发光信号值。HEK293-SBE cells (Bps bioscience) were added to a 96-well plate (MEM medium containing 10% FBS), 30,000/well, and cultured overnight at 37° C., 5% CO 2 . The medium was changed to 0.5% FBS medium, and the test compound diluted in 0.5% FBS medium was added. The highest final concentration of the test compound was 10 μM, and the compound was diluted 4 times, with a total of 8 concentration gradients. After 4-5 hours of incubation, 10 [mu]l of TGF[beta] was added. The final concentration of TGFβ was 0.5 ng/ml. 10 μl of medium was added in place of TGFβ as a negative control. In the blank control, no test compound was added, and TGFβ was added. Bright Glo reagent was added to each well, and the chemiluminescence signal value was read on a microplate reader.
按照下述公式计算不同浓度化合物的百分比抑制率:The percent inhibition of different concentrations of compounds was calculated according to the following formula:
百分比抑制率=(1-(测试化合物的化学发光信号值-空白对照的化学发光信号值)/(阴性对照的化学发光信号值-空白对照的化学发光信号值))×100%;Percent inhibition rate=(1-(chemiluminescence signal value of test compound-chemiluminescence signal value of blank control)/(chemiluminescence signal value of negative control-chemiluminescence signal value of blank control))×100%;
将不同浓度化合物的百分比抑制率相对于化合物浓度作图,按照四参数模型拟合曲线,通过下式计算IC50值:The percent inhibition rates of different concentrations of compounds were plotted against compound concentrations, the curves were fitted according to a four-parameter model, and the IC50 values were calculated by the following formula:
y=Min+(Max-Min)/(1+(x/IC50)^(-Hillslope)),其中:y为百分比抑制率;Max和Min分别为拟合曲线的最大值与最小值;x为化合物的对数浓度;且Hillslope为曲线斜率。y=Min+(Max-Min)/(1+(x/IC 50 )^(-Hillslope)), where: y is the percentage inhibition rate; Max and Min are the maximum and minimum values of the fitted curve, respectively; x is The logarithmic concentration of the compound; and Hillslope is the slope of the curve.
实验结果如下表2所示:The experimental results are shown in Table 2 below:
表2.本发明的化合物对HEK293-SBE细胞TGFβ/Smad信号通路的抑制作用Table 2. Inhibitory effect of compounds of the present invention on TGFβ/Smad signaling pathway in HEK293-SBE cells
由表2可以看出,本发明的化合物对HEK293-SBE细胞TGFβ/Smad信号通路具有显著的抑制作用。It can be seen from Table 2 that the compounds of the present invention have a significant inhibitory effect on the TGFβ/Smad signaling pathway in HEK293-SBE cells.
试验例三:CYP酶(细胞色素P450)抑制试验。Test Example 3: CYP enzyme (cytochrome P450) inhibition test.
1、试验系统:1. Test system:
P450-GloTM CYP1A2筛选系统(Promega);P450-Glo ™ CYP1A2 Screening System (Promega);
P450-GloTM CYP2D6筛选系统(Promega);P450-Glo ™ CYP2D6 Screening System (Promega);
P450-GloTM CYP3A4筛选系统(Promega)。P450-Glo ™ CYP3A4 Screening System (Promega).
2、测试仪器:2. Test equipment:
BMG PHERAstar FS Luminescent。BMG PHERAstar FS Luminescent.
3、试验方法:3. Test method:
分别按照试剂盒说明书进行试验,步骤如下:Test according to the kit instructions, the steps are as follows:
3.1.对CYP1A2的抑制:3.1. Inhibition of CYP1A2:
测试组:将不同浓度的待测化合物加入到微孔板中,向每孔中加入Luciferin-ME(100μM)、K3PO4(100mM)和CYP1A2(0.01pmol/μL),在室温下预孵育10min,随后加入NADPH再生系统,在室温下反应30min,最后加入等体积的检测缓冲液,在室温下孵育20min,然后进行化学发光检测。Test group: different concentrations of test compounds were added to the microplate, Luciferin-ME (100 μM), K 3 PO 4 (100 mM) and CYP1A2 (0.01 pmol/μL) were added to each well, and pre-incubated at room temperature 10min, then add NADPH regeneration system, react at room temperature for 30min, finally add an equal volume of detection buffer, incubate at room temperature for 20min, and then perform chemiluminescence detection.
阴性对照组:实验方法同测试组,但不加待测化合物。Negative control group: the experimental method is the same as that of the test group, but the compound to be tested is not added.
空白对照组:实验方法同测试组,但不加待测化合物,并且用CYP1A2 Membrance(0.01pmol/μL)代替CYP1A2。Blank control group: the experimental method is the same as that of the test group, but no test compound is added, and CYP1A2 Membrance (0.01pmol/μL) is used instead of CYP1A2.
3.2.对CYP2D6的抑制:3.2. Inhibition of CYP2D6:
测试组:将不同浓度的待测化合物加入到微孔板中,向每孔中加入Luciferin-MEEGE(3μM)、K3PO4(100mM)和CYP2D6(5nM),在室温下预孵育10min,随后加入NADPH再生系统,在37℃下反应30min,最后加入等体积的检测缓冲液,在室温下孵育20min,然后进行化学发光检测。Test group: different concentrations of test compounds were added to the microplate, Luciferin-MEEGE (3 μM), K 3 PO 4 (100 mM) and CYP2D6 (5 nM) were added to each well, pre-incubated at room temperature for 10 min, and then The NADPH regeneration system was added, and the reaction was carried out at 37 °C for 30 min. Finally, an equal volume of detection buffer was added and incubated at room temperature for 20 min, followed by chemiluminescence detection.
阴性对照组:实验方法同测试组,但不加待测化合物。Negative control group: the experimental method is the same as that of the test group, but the compound to be tested is not added.
空白对照组:实验方法同测试组,但不加待测化合物,并且用CYP2D6 Membrance(5nM)代替CYP2D6。Blank control group: the experimental method is the same as that of the test group, but no test compound is added, and CYP2D6 Membrance (5nM) is used instead of CYP2D6.
3.3.对CYP3A4的抑制:3.3. Inhibition of CYP3A4:
测试组:将不同浓度的待测化合物加入到微孔板中,向每孔中加入Luciferin-IPA(3μM)、K3PO4(100mM)和CYP3A4(2nM),在室温下预孵育10min,随后加入NADPH再生系统,在室温下反应30min,最后加入等体积的检测缓冲液,在室温下孵育20min,然后进行化学发光检测。Test group: different concentrations of test compounds were added to the microplate, Luciferin-IPA (3 μM), K 3 PO 4 (100 mM) and CYP3A4 (2 nM) were added to each well, pre-incubated at room temperature for 10 min, and then Add the NADPH regeneration system, react at room temperature for 30 minutes, and finally add an equal volume of detection buffer, incubate at room temperature for 20 minutes, and then perform chemiluminescence detection.
阴性对照组:实验方法同测试组,但不加待测化合物。Negative control group: the experimental method is the same as that of the test group, but the compound to be tested is not added.
空白对照组:实验方法同测试组,但不加待测化合物,并且用CYP3A4 Membrance(2nM)代替CYP3A4。Blank control group: the experimental method is the same as that of the test group, but no test compound is added, and CYP3A4 Membrance (2nM) is used instead of CYP3A4.
4、数据处理:4. Data processing:
百分比抑制率(%)=(1-(待测化合物的化学发光信号值-空白对照的化学发光信号值)/(阴性对照的化学发光信号值-空白对照的化学发光信号值))×100%;Percent inhibition rate (%)=(1-(chemiluminescence signal value of test compound-chemiluminescence signal value of blank control)/(chemiluminescence signal value of negative control-chemiluminescence signal value of blank control))×100% ;
根据不同浓度化合物对CYP酶的抑制率,估算化合物的半数抑制浓度(IC50)或范围:According to the inhibition rate of CYP enzymes by different concentrations of compounds, the median inhibitory concentration (IC 50 ) or range of the compounds was estimated:
IC50=X×(1-百分比抑制率(%))/百分比抑制率(%),其中:X为化合物的测试浓度。 IC50 =XX(1-percent inhibition (%))/percent inhibition (%), where: X is the test concentration of the compound.
5、实验结果:5. Experimental results:
按照上述方法测定本发明化合物对三种CYPs的抑制,结果如下表3中所示。Inhibition of the three CYPs by the compounds of the present invention was determined as described above and the results are shown in Table 3 below.
表3.CYPs抑制试验结果Table 3. Results of CYPs inhibition test
Claims (18)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011104889.XA CN114369083A (en) | 2020-10-15 | 2020-10-15 | Heterocyclic compound, pharmaceutical composition containing same, preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202011104889.XA CN114369083A (en) | 2020-10-15 | 2020-10-15 | Heterocyclic compound, pharmaceutical composition containing same, preparation method and application thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN114369083A true CN114369083A (en) | 2022-04-19 |
Family
ID=81138227
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202011104889.XA Pending CN114369083A (en) | 2020-10-15 | 2020-10-15 | Heterocyclic compound, pharmaceutical composition containing same, preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN114369083A (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1694708A (en) * | 2002-09-10 | 2005-11-09 | 西奥斯股份有限公司 | Inhibitors of tgfbeta |
| WO2006105222A2 (en) * | 2005-03-25 | 2006-10-05 | Scios Inc. | Carboxamide inhibitors of tgfb |
| CN106132950A (en) * | 2014-01-01 | 2016-11-16 | 麦迪韦逊技术股份有限公司 | Compounds and Methods of Use |
| WO2017157991A1 (en) * | 2016-03-18 | 2017-09-21 | Bayer Pharma Aktiengesellschaft | 1-alkyl-pyrazoles and -indazoles as bub1 inhibitors for the treatment of hyperproliferative diseases |
-
2020
- 2020-10-15 CN CN202011104889.XA patent/CN114369083A/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN1694708A (en) * | 2002-09-10 | 2005-11-09 | 西奥斯股份有限公司 | Inhibitors of tgfbeta |
| WO2006105222A2 (en) * | 2005-03-25 | 2006-10-05 | Scios Inc. | Carboxamide inhibitors of tgfb |
| CN106132950A (en) * | 2014-01-01 | 2016-11-16 | 麦迪韦逊技术股份有限公司 | Compounds and Methods of Use |
| WO2017157991A1 (en) * | 2016-03-18 | 2017-09-21 | Bayer Pharma Aktiengesellschaft | 1-alkyl-pyrazoles and -indazoles as bub1 inhibitors for the treatment of hyperproliferative diseases |
Non-Patent Citations (2)
| Title |
|---|
| PIERRE RABOISSON: "Evaluation of the anti-hepatitis C virus effect of novel potent,selective, and orally bioavailable JNK and VEGFR kinase inhibitors", BIOORGANIC & MEDICINAL CHEMISTRY LETTERS, vol. 17, no. 7, 1 April 2007 (2007-04-01), pages 1843 * |
| STN结构检索: "202011104889X", 8 July 2011, pages: 1 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN114096544B (en) | KRAS G12C inhibitors and uses thereof | |
| CN106220635B (en) | Fused tetracyclic or pentacyclic dihydrodiaza*carbazolones as PARP inhibitors | |
| TWI827017B (en) | Pyridine derivatives and their applications in medicine | |
| CN104470902B (en) | N-(3-heteroarylaryl)-4-arylaryl carboxamides and analogues as Hedgehog pathway inhibitors and their use | |
| CN116332948A (en) | A nitrogen-containing tetracyclic compound and its preparation method and medicinal use | |
| CN110770242B (en) | Heteroaromatic compounds as VANIN inhibitors | |
| BR112013025732B1 (en) | Imizazo-oxazine compounds, their uses and pharmaceutical and drug composition comprising the same | |
| CN116969904B (en) | Compounds used as autophagy regulators, their preparation methods and uses | |
| JP2022528780A (en) | Heterocyclic compounds as kinase inhibitors for therapeutic use | |
| FR2941948A1 (en) | AZAINDOLE DERIVATIVES AS INHIBITOR OF ABL KINASE PROTEINS AND SRC | |
| WO2018214866A1 (en) | Azaaryl derivative, preparation method therefor, and application thereof for use in pharmacy | |
| WO2018068759A1 (en) | Five-membered heteroaryl ring bridged ring derivative, preparation method therefor and medical use thereof | |
| WO2024026423A1 (en) | Substituted quinoline derivatives as pi3k inhibitors | |
| CN114920690A (en) | Amide derivative and application thereof in medicine | |
| CA3069602A1 (en) | Formylpyridine derivative having fgfr4 inhibitory activity, preparation method therefor and use thereof | |
| CN116655602A (en) | PI3K alpha allosteric inhibitors | |
| CN112341477A (en) | Imidazo pyrimidine compound containing polycyclic group, preparation method and application thereof | |
| CN116969976A (en) | Deubiquitinating enzyme inhibitors and their applications | |
| CN114380818A (en) | Pyrazolo pyridine compound, and pharmaceutical composition, preparation method and application thereof | |
| CN114369083A (en) | Heterocyclic compound, pharmaceutical composition containing same, preparation method and application thereof | |
| CN109384785B (en) | Pyrrolopyridinone derivatives, preparation method and medical application thereof | |
| WO2022063050A1 (en) | Pyrazole compound and preparation method therefor and use thereof | |
| CN116891465A (en) | A class of N-substituted quinolinone compounds, their preparation methods and uses | |
| CN114149410B (en) | Pyridocyclic compounds and preparation methods and uses thereof | |
| CN114907350A (en) | Nitrogen-containing condensed ring compounds, preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination |