CN113521979A - Method for trapping mercury from sulfur-containing mercury-containing flue gas through chlorine selenium mercury deposition - Google Patents
Method for trapping mercury from sulfur-containing mercury-containing flue gas through chlorine selenium mercury deposition Download PDFInfo
- Publication number
- CN113521979A CN113521979A CN202110923109.2A CN202110923109A CN113521979A CN 113521979 A CN113521979 A CN 113521979A CN 202110923109 A CN202110923109 A CN 202110923109A CN 113521979 A CN113521979 A CN 113521979A
- Authority
- CN
- China
- Prior art keywords
- mercury
- flue gas
- sulfur
- selenium
- chlorine
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/46—Removing components of defined structure
- B01D53/64—Heavy metals or compounds thereof, e.g. mercury
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/02—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols by adsorption, e.g. preparative gas chromatography
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/74—General processes for purification of waste gases; Apparatus or devices specially adapted therefor
- B01D53/81—Solid phase processes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D53/00—Separation of gases or vapours; Recovering vapours of volatile solvents from gases; Chemical or biological purification of waste gases, e.g. engine exhaust gases, smoke, fumes, flue gases, aerosols
- B01D53/34—Chemical or biological purification of waste gases
- B01D53/96—Regeneration, reactivation or recycling of reactants
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2251/00—Reactants
- B01D2251/50—Inorganic acids
- B01D2251/502—Hydrochloric acid
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2257/00—Components to be removed
- B01D2257/60—Heavy metals or heavy metal compounds
- B01D2257/602—Mercury or mercury compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2258/00—Sources of waste gases
- B01D2258/02—Other waste gases
- B01D2258/0283—Flue gases
Landscapes
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Environmental & Geological Engineering (AREA)
- Analytical Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Biomedical Technology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Sustainable Development (AREA)
- Treating Waste Gases (AREA)
Abstract
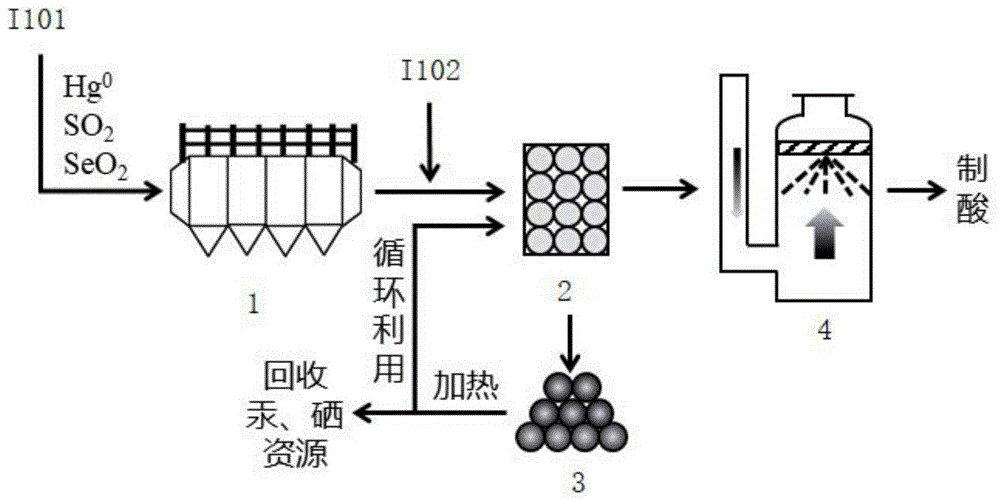
本发明涉及一种通过氯硒汞沉积从含硫含汞烟气中捕集汞的方法,包括以下步骤:(1)有色冶炼原烟气经过除尘及余热回收后,向其中喷入一定量的水雾,使其温度降至露点附近,使烟气中的二氧化硒在二氧化硫与水蒸汽的共同作用下被还原为可与烟气中汞快速反应的活性单质硒,并在烟气中HCl的作用下,生成易于沉积的氯硒汞化合物;(2)然后使烟气与基质材料接触,通过其表面诱导效应,加速氯硒汞的生成及沉积,从而使烟气中的汞被捕集到基质表面。与现有技术相比,本发明方法可在烟气半干状态实现汞的高效捕集与回收,避免其过多进入下游洗涤污酸或制酸系统,从而造成回收率下降及跨介质转移等二次污染问题。
The present invention relates to a method for capturing mercury from sulfur-containing and mercury-containing flue gas by deposition of chloroselenium and mercury, comprising the following steps: (1) after dust removal and waste heat recovery from non-ferrous smelting raw flue gas, a certain amount of flue gas is sprayed into it. The water mist reduces the temperature to around the dew point, so that the selenium dioxide in the flue gas is reduced to active elemental selenium that can react quickly with mercury in the flue gas under the combined action of sulfur dioxide and water vapor, and HCl in the flue gas. Under the action of the chloroselenide mercury compound, which is easy to deposit; (2) the flue gas is then contacted with the matrix material, and through its surface induction effect, the formation and deposition of chloroselenomercuric mercury is accelerated, so that the mercury in the flue gas is trapped to the substrate surface. Compared with the prior art, the method of the present invention can realize the efficient capture and recovery of mercury in the semi-dry state of the flue gas, and avoid excessive entry of the mercury into the downstream scrubbing acid or acid-making system, thereby resulting in a decrease in the recovery rate and cross-media transfer, etc. Secondary pollution problem.
Description
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110923109.2A CN113521979B (en) | 2021-08-12 | 2021-08-12 | Method for trapping mercury from sulfur-containing mercury-containing flue gas through chlorselenomel deposition |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202110923109.2A CN113521979B (en) | 2021-08-12 | 2021-08-12 | Method for trapping mercury from sulfur-containing mercury-containing flue gas through chlorselenomel deposition |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113521979A true CN113521979A (en) | 2021-10-22 |
| CN113521979B CN113521979B (en) | 2024-01-12 |
Family
ID=78090966
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202110923109.2A Active CN113521979B (en) | 2021-08-12 | 2021-08-12 | Method for trapping mercury from sulfur-containing mercury-containing flue gas through chlorselenomel deposition |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113521979B (en) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5607496A (en) * | 1994-06-01 | 1997-03-04 | Brooks Rand, Ltd. | Removal of mercury from a combustion gas stream and apparatus |
| WO2002045825A1 (en) * | 2000-12-08 | 2002-06-13 | Outokumpu Oyj | Method for removing mercury from gas |
| US20110044872A1 (en) * | 2008-01-21 | 2011-02-24 | Mitsubishi Heavy Industries, Ltd. | Flue gas control system of coal combustion boiler and operating method thereof |
| CN102325590A (en) * | 2008-12-19 | 2012-01-18 | 康宁股份有限公司 | Coated flow-through substrates and methods of making and using same |
| CN103623772A (en) * | 2013-12-02 | 2014-03-12 | 上海交通大学 | Absorbent for removing and recovering liquid-phase mercury as well as preparation method and using method thereof |
| CN105238933A (en) * | 2015-11-02 | 2016-01-13 | 郑州轻工业学院 | Method for removing and recycling mercury element from sulfur dioxide containing smoke |
| CN106474907A (en) * | 2015-09-01 | 2017-03-08 | 氧化还原科技集团有限责任公司 | Compositionss and its method for selenium is removed from the industrial gases containing selenium |
| CN108889110A (en) * | 2018-08-08 | 2018-11-27 | 中国恩菲工程技术有限公司 | The method for removing hydrargyrum of flue gas |
-
2021
- 2021-08-12 CN CN202110923109.2A patent/CN113521979B/en active Active
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5607496A (en) * | 1994-06-01 | 1997-03-04 | Brooks Rand, Ltd. | Removal of mercury from a combustion gas stream and apparatus |
| WO2002045825A1 (en) * | 2000-12-08 | 2002-06-13 | Outokumpu Oyj | Method for removing mercury from gas |
| US20110044872A1 (en) * | 2008-01-21 | 2011-02-24 | Mitsubishi Heavy Industries, Ltd. | Flue gas control system of coal combustion boiler and operating method thereof |
| CN102325590A (en) * | 2008-12-19 | 2012-01-18 | 康宁股份有限公司 | Coated flow-through substrates and methods of making and using same |
| CN103623772A (en) * | 2013-12-02 | 2014-03-12 | 上海交通大学 | Absorbent for removing and recovering liquid-phase mercury as well as preparation method and using method thereof |
| CN106474907A (en) * | 2015-09-01 | 2017-03-08 | 氧化还原科技集团有限责任公司 | Compositionss and its method for selenium is removed from the industrial gases containing selenium |
| CN105238933A (en) * | 2015-11-02 | 2016-01-13 | 郑州轻工业学院 | Method for removing and recycling mercury element from sulfur dioxide containing smoke |
| CN108889110A (en) * | 2018-08-08 | 2018-11-27 | 中国恩菲工程技术有限公司 | The method for removing hydrargyrum of flue gas |
Non-Patent Citations (1)
| Title |
|---|
| 《化工百科全书》编辑部: "《金银珠宝贵重金属提取冶炼加工分析技术标准与质量检测鉴定验收规范实验手册 第2卷》", pages: 1131 - 90 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113521979B (en) | 2024-01-12 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN103301749B (en) | The method of flue gas and desulfurizing and denitrifying | |
| CN110280125B (en) | A dry purification method for smelting flue gas containing arsenic and SO3 | |
| CN110124507A (en) | A kind of method and device thereof of multi-pollutant flue gas cleaning treatment | |
| CN102380308A (en) | Sintering flue gas desulfurization and purification method and equipment | |
| CN104841265A (en) | Dry-type removing method for sulfur trioxide and heavy metals in nonferrous smelting acid-making flue gas | |
| CN112708477B (en) | Method for improving combustion heat value of blast furnace gas and simultaneously removing organic sulfur and inorganic sulfur | |
| CN104689679A (en) | Desulfurization and denitrification process for coke oven flue gas | |
| CN103285711B (en) | Method for purifying and recovering mercury in off gas | |
| CN110124490A (en) | A kind of active carbon handles the method and device thereof of multi-pollutant flue gas and waste water reuse | |
| CN107998817B (en) | Single tower flue gas purification device and flue gas purification method | |
| CN115141660B (en) | Dry type fine desulfurization system and method for blast furnace gas | |
| CN106955571A (en) | A kind of industrial smoke ozone step-by-step oxidation absorbs the device and method of simultaneous SO_2 and NO removal | |
| CN110615409A (en) | Sulfur dioxide flue gas acid making process based on smelting system | |
| CN110252068A (en) | The desulfurizing and purifying method of blast furnace gas | |
| CN103877841B (en) | The integrated purifying recovery process of sinter fume pollutant | |
| CN109364659B (en) | A method and device for purifying and recovering thallium in smelting flue gas | |
| CN109569251B (en) | A device and method for producing dilute sulfuric acid by utilizing SO2-containing flue gas | |
| CN102614775A (en) | Method for removing and recovering low concentration sulfur dioxide in industrial exhaust gas | |
| CN115957610A (en) | Waste incineration flue gas treatment system and treatment method | |
| CN112403186B (en) | A method for collaborative treatment of multi-pollutant flue gas and recovery of ammonium ferrous sulfite | |
| CN111375274A (en) | Containing SO2Gas treatment method and apparatus | |
| CN107930388A (en) | A kind of coke oven flue gas sulfur removal technology and coke oven flue gas desulphurization system | |
| CN114100592B (en) | A method for regenerating flue gas mercury removal materials and recovering elemental mercury | |
| CN202224048U (en) | Sintering flue gas treatment device | |
| CN113499753A (en) | Preparation and regeneration method of renewable demercuration adsorbent |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| CB03 | Change of inventor or designer information |
Inventor after: Xu Haomiao Inventor after: Zhang Anbang Inventor after: Yan Naiqiang Inventor after: Li Gui Inventor after: Qu Zan Inventor after: Huang Wenjun Inventor after: Cui Peng Inventor after: Hong Qinyuan Inventor after: Fan Yurui Inventor before: Xu Haomiao Inventor before: Yan Naiqiang Inventor before: Qu Zan Inventor before: Huang Wenjun Inventor before: Hong Qinyuan Inventor before: Fan Yurui |
|
| CB03 | Change of inventor or designer information | ||
| TA01 | Transfer of patent application right |
Effective date of registration: 20231208 Address after: 200240 No. 800, Dongchuan Road, Shanghai, Minhang District Applicant after: SHANGHAI JIAO TONG University Applicant after: HENAN YUGUANG GOLD AND LEAD GROUP CO.,LTD. Address before: 200240 No. 800, Dongchuan Road, Shanghai, Minhang District Applicant before: SHANGHAI JIAO TONG University |
|
| TA01 | Transfer of patent application right | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |