CN111763302B - TPU (thermoplastic polyurethane) formula for thin film and preparation method thereof - Google Patents
TPU (thermoplastic polyurethane) formula for thin film and preparation method thereof Download PDFInfo
- Publication number
- CN111763302B CN111763302B CN202010650573.4A CN202010650573A CN111763302B CN 111763302 B CN111763302 B CN 111763302B CN 202010650573 A CN202010650573 A CN 202010650573A CN 111763302 B CN111763302 B CN 111763302B
- Authority
- CN
- China
- Prior art keywords
- parts
- antioxidant
- corn starch
- butanediol
- mixing
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 229920002803 thermoplastic polyurethane Polymers 0.000 title claims abstract description 42
- 238000002360 preparation method Methods 0.000 title claims abstract description 23
- 239000004433 Thermoplastic polyurethane Substances 0.000 title abstract description 34
- 239000010409 thin film Substances 0.000 title description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 claims abstract description 46
- 239000000463 material Substances 0.000 claims abstract description 37
- 229920000881 Modified starch Polymers 0.000 claims abstract description 31
- 239000002994 raw material Substances 0.000 claims abstract description 30
- 239000003963 antioxidant agent Substances 0.000 claims abstract description 23
- 230000003078 antioxidant effect Effects 0.000 claims abstract description 23
- 239000004611 light stabiliser Substances 0.000 claims abstract description 23
- 238000002156 mixing Methods 0.000 claims abstract description 23
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims abstract description 19
- UPMLOUAZCHDJJD-UHFFFAOYSA-N 4,4'-Diphenylmethane Diisocyanate Chemical compound C1=CC(N=C=O)=CC=C1CC1=CC=C(N=C=O)C=C1 UPMLOUAZCHDJJD-UHFFFAOYSA-N 0.000 claims abstract description 18
- 239000003054 catalyst Substances 0.000 claims abstract description 17
- 238000006243 chemical reaction Methods 0.000 claims abstract description 15
- 229920001730 Moisture cure polyurethane Polymers 0.000 claims abstract description 14
- 238000001035 drying Methods 0.000 claims abstract description 14
- 238000001125 extrusion Methods 0.000 claims abstract description 7
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 15
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 15
- 229920003225 polyurethane elastomer Polymers 0.000 claims description 12
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 11
- 239000007864 aqueous solution Substances 0.000 claims description 10
- 229920002261 Corn starch Polymers 0.000 claims description 6
- 239000008120 corn starch Substances 0.000 claims description 6
- 238000005303 weighing Methods 0.000 claims description 6
- UWSMKYBKUPAEJQ-UHFFFAOYSA-N 5-Chloro-2-(3,5-di-tert-butyl-2-hydroxyphenyl)-2H-benzotriazole Chemical group CC(C)(C)C1=CC(C(C)(C)C)=CC(N2N=C3C=C(Cl)C=CC3=N2)=C1O UWSMKYBKUPAEJQ-UHFFFAOYSA-N 0.000 claims description 5
- JKIJEFPNVSHHEI-UHFFFAOYSA-N Phenol, 2,4-bis(1,1-dimethylethyl)-, phosphite (3:1) Chemical compound CC(C)(C)C1=CC(C(C)(C)C)=CC=C1OP(OC=1C(=CC(=CC=1)C(C)(C)C)C(C)(C)C)OC1=CC=C(C(C)(C)C)C=C1C(C)(C)C JKIJEFPNVSHHEI-UHFFFAOYSA-N 0.000 claims description 5
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical compound CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 claims description 5
- 229920002472 Starch Polymers 0.000 claims description 5
- BGYHLZZASRKEJE-UHFFFAOYSA-N [3-[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoyloxy]-2,2-bis[3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoyloxymethyl]propyl] 3-(3,5-ditert-butyl-4-hydroxyphenyl)propanoate Chemical compound CC(C)(C)C1=C(O)C(C(C)(C)C)=CC(CCC(=O)OCC(COC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)(COC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)COC(=O)CCC=2C=C(C(O)=C(C=2)C(C)(C)C)C(C)(C)C)=C1 BGYHLZZASRKEJE-UHFFFAOYSA-N 0.000 claims description 5
- UKLDJPRMSDWDSL-UHFFFAOYSA-L [dibutyl(dodecanoyloxy)stannyl] dodecanoate Chemical group CCCCCCCCCCCC(=O)O[Sn](CCCC)(CCCC)OC(=O)CCCCCCCCCCC UKLDJPRMSDWDSL-UHFFFAOYSA-L 0.000 claims description 5
- 238000001816 cooling Methods 0.000 claims description 5
- 239000012975 dibutyltin dilaurate Substances 0.000 claims description 5
- 239000012153 distilled water Substances 0.000 claims description 5
- 235000013336 milk Nutrition 0.000 claims description 5
- 239000008267 milk Substances 0.000 claims description 5
- 210000004080 milk Anatomy 0.000 claims description 5
- 230000007935 neutral effect Effects 0.000 claims description 5
- 238000007873 sieving Methods 0.000 claims description 5
- UGTZMIPZNRIWHX-UHFFFAOYSA-K sodium trimetaphosphate Chemical compound [Na+].[Na+].[Na+].[O-]P1(=O)OP([O-])(=O)OP([O-])(=O)O1 UGTZMIPZNRIWHX-UHFFFAOYSA-K 0.000 claims description 5
- 235000019698 starch Nutrition 0.000 claims description 5
- 239000008107 starch Substances 0.000 claims description 5
- 238000003756 stirring Methods 0.000 claims description 5
- KSBAEPSJVUENNK-UHFFFAOYSA-L tin(ii) 2-ethylhexanoate Chemical compound [Sn+2].CCCCC(CC)C([O-])=O.CCCCC(CC)C([O-])=O KSBAEPSJVUENNK-UHFFFAOYSA-L 0.000 claims description 5
- 238000005406 washing Methods 0.000 claims description 5
- 238000000034 method Methods 0.000 claims description 4
- QQGISFDJEJMKIL-JAIQZWGSSA-N (5z)-5-[[3-(hydroxymethyl)thiophen-2-yl]methylidene]-10-methoxy-2,2,4-trimethyl-1h-chromeno[3,4-f]quinolin-9-ol Chemical group C1=CC=2NC(C)(C)C=C(C)C=2C2=C1C=1C(OC)=C(O)C=CC=1O\C2=C/C=1SC=CC=1CO QQGISFDJEJMKIL-JAIQZWGSSA-N 0.000 claims description 3
- 150000001875 compounds Chemical class 0.000 abstract description 2
- 230000007547 defect Effects 0.000 abstract description 2
- 239000010408 film Substances 0.000 description 19
- 230000000052 comparative effect Effects 0.000 description 6
- 229920001971 elastomer Polymers 0.000 description 3
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 1
- 238000007792 addition Methods 0.000 description 1
- 239000002390 adhesive tape Substances 0.000 description 1
- 229920001400 block copolymer Polymers 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 150000002009 diols Chemical class 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000000806 elastomer Substances 0.000 description 1
- 239000004744 fabric Substances 0.000 description 1
- 238000011056 performance test Methods 0.000 description 1
- 230000035699 permeability Effects 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920002635 polyurethane Polymers 0.000 description 1
- 239000004814 polyurethane Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/70—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the isocyanates or isothiocyanates used
- C08G18/72—Polyisocyanates or polyisothiocyanates
- C08G18/74—Polyisocyanates or polyisothiocyanates cyclic
- C08G18/76—Polyisocyanates or polyisothiocyanates cyclic aromatic
- C08G18/7657—Polyisocyanates or polyisothiocyanates cyclic aromatic containing two or more aromatic rings
- C08G18/7664—Polyisocyanates or polyisothiocyanates cyclic aromatic containing two or more aromatic rings containing alkylene polyphenyl groups
- C08G18/7671—Polyisocyanates or polyisothiocyanates cyclic aromatic containing two or more aromatic rings containing alkylene polyphenyl groups containing only one alkylene bisphenyl group
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/08—Processes
- C08G18/10—Prepolymer processes involving reaction of isocyanates or isothiocyanates with compounds having active hydrogen in a first reaction step
- C08G18/12—Prepolymer processes involving reaction of isocyanates or isothiocyanates with compounds having active hydrogen in a first reaction step using two or more compounds having active hydrogen in the first polymerisation step
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/30—Low-molecular-weight compounds
- C08G18/32—Polyhydroxy compounds; Polyamines; Hydroxyamines
- C08G18/3203—Polyhydroxy compounds
- C08G18/3206—Polyhydroxy compounds aliphatic
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/40—High-molecular-weight compounds
- C08G18/64—Macromolecular compounds not provided for by groups C08G18/42 - C08G18/63
- C08G18/6484—Polysaccharides and derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G18/00—Polymeric products of isocyanates or isothiocyanates
- C08G18/06—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen
- C08G18/28—Polymeric products of isocyanates or isothiocyanates with compounds having active hydrogen characterised by the compounds used containing active hydrogen
- C08G18/65—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen
- C08G18/6505—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen the low-molecular compounds being compounds of group C08G18/32 or polyamines of C08G18/38
- C08G18/6511—Low-molecular-weight compounds having active hydrogen with high-molecular-weight compounds having active hydrogen the low-molecular compounds being compounds of group C08G18/32 or polyamines of C08G18/38 compounds of group C08G18/3203
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J5/00—Manufacture of articles or shaped materials containing macromolecular substances
- C08J5/18—Manufacture of films or sheets
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/04—Oxygen-containing compounds
- C08K5/13—Phenols; Phenolates
- C08K5/134—Phenols containing ester groups
- C08K5/1345—Carboxylic esters of phenolcarboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/16—Nitrogen-containing compounds
- C08K5/34—Heterocyclic compounds having nitrogen in the ring
- C08K5/3467—Heterocyclic compounds having nitrogen in the ring having more than two nitrogen atoms in the ring
- C08K5/3472—Five-membered rings
- C08K5/3475—Five-membered rings condensed with carbocyclic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K5/00—Use of organic ingredients
- C08K5/49—Phosphorus-containing compounds
- C08K5/51—Phosphorus bound to oxygen
- C08K5/52—Phosphorus bound to oxygen only
- C08K5/524—Esters of phosphorous acids, e.g. of H3PO3
- C08K5/526—Esters of phosphorous acids, e.g. of H3PO3 with hydroxyaryl compounds
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08J—WORKING-UP; GENERAL PROCESSES OF COMPOUNDING; AFTER-TREATMENT NOT COVERED BY SUBCLASSES C08B, C08C, C08F, C08G or C08H
- C08J2375/00—Characterised by the use of polyureas or polyurethanes; Derivatives of such polymers
- C08J2375/04—Polyurethanes
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L2203/00—Applications
- C08L2203/16—Applications used for films
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Manufacturing & Machinery (AREA)
- Materials Engineering (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Polyurethanes Or Polyureas (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
Abstract
The invention relates to the technical field of high molecular compounds, and particularly discloses a TPU (thermoplastic polyurethane) formula for a film, which comprises the following raw material components in parts by weight: 25-35 parts of bis (4-isocyanatophenyl) methane, 4-6 parts of 1, 4-butanediol, 50-60 parts of dihydric alcohol, 8-12 parts of modified corn starch, 2-3 parts of antioxidant and 1-2 parts of light stabilizer; the preparation method comprises the following steps: drying the raw materials in a drying oven at 80 deg.C for 8 hr; mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer; and (2) mixing the polyurethane prepolymer with 1, 4-butanediol and modified corn starch in a formula amount, standing for reaction, then mixing with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film. The invention overcomes the defects of the prior art, and the prepared TPU material has high elasticity, better waterproof and moisture-permeable performances when being applied to a film, and has the advantages of simple preparation method, easily obtained raw materials, low price and easy realization.
Description
Technical Field
The invention relates to the technical field of high molecular compounds, and particularly belongs to a TPU (thermoplastic polyurethane) formula for a film and a preparation method thereof.
Background
Thermoplastic polyurethane elastomers (TPU) are a class of heat-moldable, solvent-soluble polyurethane materials with unique soft and hard block copolymer structures that are widely used in many applications due to their rubber elasticity and plastic processability, for example: external parts of an automobile body, a cable sheath, an adhesive tape, a ski boot, a gear, a rubber tube, a garment material, an inflatable product and the like.
In general, different fields of application have a certain higher demand for TPU materials, such as: the TPU fabric is required to have better waterproof and moisture-permeable performances for clothing and shoe materials, mechanical equipment requires the TPU product to have higher mechanical strength, and inflatable products (such as inflatable cushions, safety chairs, inflatable beds and the like) require the TPU material to have better elasticity and wear resistance.
Therefore, how to prepare TPU materials with high elasticity and better waterproof and moisture-permeable performances is a problem to be solved urgently in the industry.
Disclosure of Invention
The invention aims to provide a TPU formula for a film and a preparation method thereof, overcomes the defects of the prior art, and the prepared TPU material has high elasticity and better waterproof and moisture-permeable performances when being applied to the film, and has the advantages of simple preparation method, easily obtained raw materials, low price and easy realization.
In order to solve the problems, the technical scheme adopted by the invention is as follows: the material comprises the following raw materials in parts by weight: 25-35 parts of bis (4-isocyanatophenyl) methane, 4-6 parts of 1, 4-butanediol, 50-60 parts of dihydric alcohol, 8-12 parts of modified corn starch, 2-3 parts of antioxidant and 1-2 parts of light stabilizer.
Further, the material comprises the following raw material components in parts by weight: 25 parts of bis (4-isocyanatophenyl) methane, 4 parts of 1, 4-butanediol, 60 parts of dihydric alcohol, 8 parts of modified corn starch, 2 parts of antioxidant and 1 part of light stabilizer.
Further, the material comprises the following raw material components in parts by weight: 30 parts of bis (4-isocyanatophenyl) methane, 5 parts of 1, 4-butanediol, 55 parts of dihydric alcohol, 10 parts of modified corn starch, 2.5 parts of antioxidant and 1.5 parts of light stabilizer.
Further, the material comprises the following raw material components in parts by weight: 35 parts of bis (4-isocyanatophenyl) methane, 6 parts of 1, 4-butanediol, 20 parts of dihydric alcohol, 12 parts of modified corn starch, 3 parts of antioxidant and 2 parts of light stabilizer.
Further, the diol consists of 20 wt% of ethylene glycol and 80 wt% of 1, 4-butanediol.
Further, the specific preparation method of the modified corn starch comprises the following steps: adding distilled water with the same amount into corn starch to prepare starch milk, adding a 3% sodium hydroxide aqueous solution to adjust the pH value under the conditions of constant-temperature water bath and stirring, slowly dropwise adding a 12% sodium trimetaphosphate aqueous solution within 2h, slowly dropwise adding propylene oxide after full reaction, naturally cooling after reacting for 24h, adjusting the pH value to be neutral, washing, drying, crushing and sieving to obtain the modified corn starch.
Further, the antioxidant is composed of an antioxidant 1010 and an antioxidant 168 according to a weight ratio of 1: 2.
Further, the light stabilizer is 2- (2' -hydroxy-3 ',5' -di-tert-butylphenyl) -5-chlorobenzotriazole.
A preparation method of a TPU formula for a film based on the formula specifically comprises the following steps:
(1) weighing the raw materials according to the formula, and putting the raw materials into a drying oven to be dried for 8 hours at the temperature of 80 ℃;
(2) mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer;
(3) mixing the polyurethane prepolymer obtained in the step (2) with 1, 4-butanediol and modified corn starch according to the formula amount, and standing for reaction to generate a polyurethane elastomer;
(4) and (4) mixing the polyurethane elastomer obtained in the step (3) with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film.
Further, in the step (2), the catalyst is dibutyltin dilaurate and/or stannous octoate; the catalyst accounts for 0.5% of the total mass.
Compared with the prior art, the invention has the following implementation effects:
1. the TPU material prepared by the formula for the film and the preparation method thereof provided by the invention is applied to the film, has high elasticity and better waterproof moisture permeability, and is simple in preparation method, easy in obtaining of raw materials, low in price and easy to realize.
2. According to the invention, the modified corn starch is added for blending, so that the strength of the TPU material is enhanced, and the water absorption and degradability of the material are improved to a certain extent.
3. According to the invention, the raw materials are dried and then blended, so that the waterproof and moisture-permeable performances of the material are improved.
Detailed Description
The present invention will be further described with reference to the following examples, but the present invention is not limited to these examples, and any modification is within the scope of the present invention without departing from the spirit of the present invention.
Example 1
The TPU formula for the film comprises the following raw materials in parts by weight: 25 parts of bis (4-isocyanatophenyl) methane, 4 parts of 1, 4-butanediol, 60 parts of dihydric alcohol, 8 parts of modified corn starch, 2 parts of antioxidant and 1 part of light stabilizer.
Wherein the dihydric alcohol consists of 20 wt% of ethylene glycol and 80 wt% of 1, 4-butanediol; the specific preparation method of the modified corn starch comprises the following steps: taking 400g of corn starch, adding 400ml of distilled water to prepare starch milk, adding a 3% sodium hydroxide aqueous solution to adjust the pH under the conditions of constant-temperature water bath and stirring, slowly dropwise adding 50ml of 12% sodium trimetaphosphate aqueous solution within 2h, slowly dropwise adding 36ml of propylene oxide after full reaction, naturally cooling after 24h of reaction, adjusting the pH to be neutral, washing, drying, crushing and sieving to obtain modified corn starch; the antioxidant consists of an antioxidant 1010 and an antioxidant 168 according to the weight ratio of 1: 2; the light stabilizer is 2- (2' -hydroxy-3 ',5' -di-tert-butylphenyl) -5-chlorobenzotriazole.
The preparation method of the TPU formula for the film specifically comprises the following steps:
(1) weighing the raw materials according to the formula, and putting the raw materials into a drying oven to be dried for 8 hours at the temperature of 80 ℃;
(2) mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer; wherein the catalyst is dibutyltin dilaurate and/or stannous octoate; the catalyst accounts for 0.5 percent of the total mass;
(3) mixing the polyurethane prepolymer obtained in the step (2) with 1, 4-butanediol and modified corn starch according to the formula amount, and standing for reaction to generate a polyurethane elastomer;
(4) and (4) mixing the polyurethane elastomer obtained in the step (3) with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film.
Example 2
The TPU formula for the film comprises the following raw materials in parts by weight: 30 parts of bis (4-isocyanatophenyl) methane, 5 parts of 1, 4-butanediol, 55 parts of dihydric alcohol, 10 parts of modified corn starch, 2.5 parts of antioxidant and 1.5 parts of light stabilizer.
Wherein the dihydric alcohol consists of 20 wt% of ethylene glycol and 80 wt% of 1, 4-butanediol; the specific preparation method of the modified corn starch comprises the following steps: taking 400g of corn starch, adding 400ml of distilled water to prepare starch milk, adding a 3% sodium hydroxide aqueous solution to adjust the pH under the conditions of constant-temperature water bath and stirring, slowly dropwise adding 50ml of 12% sodium trimetaphosphate aqueous solution within 2h, slowly dropwise adding 36ml of propylene oxide after full reaction, naturally cooling after 24h of reaction, adjusting the pH to be neutral, washing, drying, crushing and sieving to obtain modified corn starch; the antioxidant consists of an antioxidant 1010 and an antioxidant 168 according to the weight ratio of 1: 2; the light stabilizer is 2- (2' -hydroxy-3 ',5' -di-tert-butylphenyl) -5-chlorobenzotriazole.
The preparation method of the TPU formula for the film specifically comprises the following steps:
(1) weighing the raw materials according to the formula, and putting the raw materials into a drying oven to be dried for 8 hours at the temperature of 80 ℃;
(2) mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer; wherein the catalyst is dibutyltin dilaurate and/or stannous octoate; the catalyst accounts for 0.5 percent of the total mass;
(3) mixing the polyurethane prepolymer obtained in the step (2) with 1, 4-butanediol and modified corn starch according to the formula amount, and standing for reaction to generate a polyurethane elastomer;
(4) and (4) mixing the polyurethane elastomer obtained in the step (3) with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film.
Example 3
The TPU formula for the film comprises the following raw materials in parts by weight: 35 parts of bis (4-isocyanatophenyl) methane, 6 parts of 1, 4-butanediol, 20 parts of dihydric alcohol, 12 parts of modified corn starch, 3 parts of antioxidant and 2 parts of light stabilizer.
Wherein the dihydric alcohol consists of 20 wt% of ethylene glycol and 80 wt% of 1, 4-butanediol; the specific preparation method of the modified corn starch comprises the following steps: taking 400g of corn starch, adding 400ml of distilled water to prepare starch milk, adding a 3% sodium hydroxide aqueous solution to adjust the pH under the conditions of constant-temperature water bath and stirring, slowly dropwise adding 50ml of 12% sodium trimetaphosphate aqueous solution within 2h, slowly dropwise adding 36ml of propylene oxide after full reaction, naturally cooling after 24h of reaction, adjusting the pH to be neutral, washing, drying, crushing and sieving to obtain modified corn starch; the antioxidant consists of an antioxidant 1010 and an antioxidant 168 according to the weight ratio of 1: 2; the light stabilizer is 2- (2' -hydroxy-3 ',5' -di-tert-butylphenyl) -5-chlorobenzotriazole.
The preparation method of the TPU formula for the film specifically comprises the following steps:
(1) weighing the raw materials according to the formula, and putting the raw materials into a drying oven to be dried for 8 hours at the temperature of 80 ℃;
(2) mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer; wherein the catalyst is dibutyltin dilaurate and/or stannous octoate; the catalyst accounts for 0.5 percent of the total mass;
(3) mixing the polyurethane prepolymer obtained in the step (2) with 1, 4-butanediol and modified corn starch according to the formula amount, and standing for reaction to generate a polyurethane elastomer;
(4) and (4) mixing the polyurethane elastomer obtained in the step (3) with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film.
Comparative example 1
The materials and preparation process were essentially the same as those of example 3, except that no modified corn starch was added.
Comparative example 2
The materials and preparation process were essentially the same as in example 3, except that the corn starch was not modified.
Comparative example 3
The materials and preparation process were substantially the same as those of example 3, except that the raw materials were not dried in step (1).
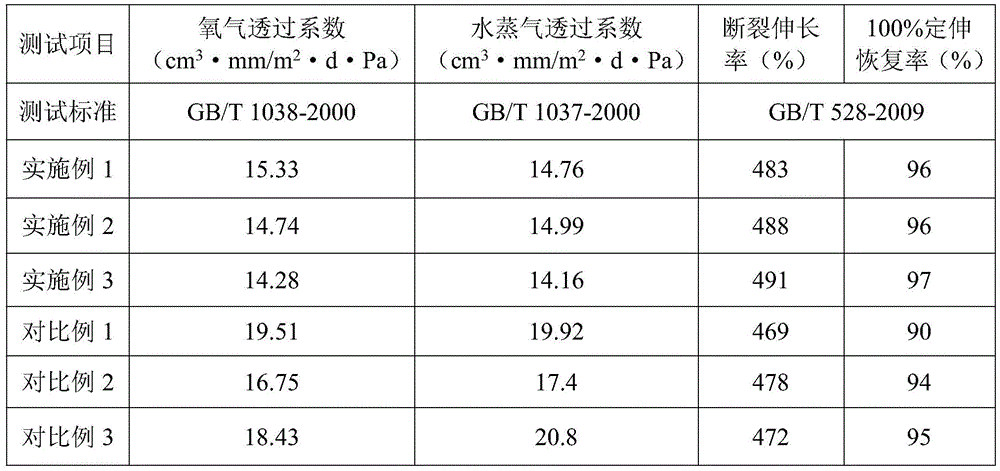
Physical detection
Equal amounts of the TPU materials prepared by the methods of examples 1-3 and comparative examples 1-3 are respectively taken to prepare films, and the prepared films are subjected to the following performance tests, and the test results are shown in Table 1:
TABLE 1 statistical table of mechanical property test results
As can be seen from Table 1, the TPU material bonding film prepared by the formula and the preparation method provided by the invention has high elasticity and better waterproof and moisture-permeable performances; as can be seen from the comparison of the results of the example 3 and the comparative examples 1-2, the strength and the waterproof and moisture-permeable performance of the material are better improved by adding the modified corn starch; as can be seen from the comparison of the results of example 3 and comparative example 3, the waterproof and moisture-permeable performance of the material can be effectively improved by drying the raw materials before blending.
The foregoing is merely exemplary and illustrative of the present inventive concept and various modifications, additions and substitutions of similar embodiments may be made to the specific embodiments described by those skilled in the art without departing from the inventive concept or exceeding the scope of the claims as defined in the accompanying claims.
Claims (5)
1. A TPU material for films, characterized by: the material comprises the following raw materials in parts by weight:
25 parts of bis (4-isocyanatophenyl) methane, 4 parts of 1, 4-butanediol, 60 parts of dihydric alcohol, 8 parts of modified corn starch, 2 parts of antioxidant and 1 part of light stabilizer;
or comprises the following raw material components in parts by weight:
30 parts of bis (4-isocyanatophenyl) methane, 5 parts of 1, 4-butanediol, 55 parts of dihydric alcohol, 10 parts of modified corn starch, 2.5 parts of antioxidant and 1.5 parts of light stabilizer;
or comprises the following raw material components in parts by weight:
35 parts of bis (4-isocyanatophenyl) methane, 6 parts of 1, 4-butanediol, 20 parts of dihydric alcohol, 12 parts of modified corn starch, 3 parts of antioxidant and 2 parts of light stabilizer;
the dihydric alcohol consists of 20 wt% of ethylene glycol and 80 wt% of 1, 4-butanediol;
the specific preparation method of the modified corn starch comprises the following steps: adding equal amount of distilled water into corn starch to prepare starch milk, adding 3% sodium hydroxide aqueous solution to adjust pH under the conditions of constant-temperature water bath and stirring, slowly dropwise adding 12% sodium trimetaphosphate aqueous solution within 2h, slowly dropwise adding propylene oxide after full reaction, naturally cooling after reacting for 24h, adjusting pH to be neutral, washing, drying, crushing and sieving to obtain modified corn starch;
the preparation method of the TPU material for the film specifically comprises the following steps:
(1) weighing the raw materials according to the formula, and putting the raw materials into a drying oven to be dried for 8 hours at the temperature of 80 ℃;
(2) mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer;
(3) mixing the polyurethane prepolymer obtained in the step (2) with 1, 4-butanediol and modified corn starch according to the formula amount, and standing for reaction to generate a polyurethane elastomer;
(4) and (4) mixing the polyurethane elastomer obtained in the step (3) with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film.
2. The TPU material for films as set forth in claim 1 wherein: the antioxidant consists of an antioxidant 1010 and an antioxidant 168 according to the weight ratio of 1: 2.
3. The TPU material for films as set forth in claim 1 wherein: the light stabilizer is 2- (2' -hydroxy-3 ',5' -di-tert-butylphenyl) -5-chlorobenzotriazole.
4. A method of preparing a TPU material for films as set forth in any of claims 1-3, characterized in that: the method specifically comprises the following steps:
(1) weighing the raw materials according to the formula, and putting the raw materials into a drying oven to be dried for 8 hours at the temperature of 80 ℃;
(2) mixing bis (4-isocyanatophenyl) methane and dihydric alcohol, and reacting in the presence of a catalyst to generate a polyurethane prepolymer;
(3) mixing the polyurethane prepolymer obtained in the step (2) with 1, 4-butanediol and modified corn starch according to the formula amount, and standing for reaction to generate a polyurethane elastomer;
(4) and (4) mixing the polyurethane elastomer obtained in the step (3) with an antioxidant and a light stabilizer, and performing melt extrusion through an extruder to obtain the TPU material for the film.
5. A method of preparing a TPU material for films as set forth in any of claims 1-3, characterized in that: in the step (2), the catalyst is dibutyltin dilaurate and/or stannous octoate; the catalyst accounts for 0.5% of the total mass.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010650573.4A CN111763302B (en) | 2020-07-08 | 2020-07-08 | TPU (thermoplastic polyurethane) formula for thin film and preparation method thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010650573.4A CN111763302B (en) | 2020-07-08 | 2020-07-08 | TPU (thermoplastic polyurethane) formula for thin film and preparation method thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN111763302A CN111763302A (en) | 2020-10-13 |
| CN111763302B true CN111763302B (en) | 2022-01-07 |
Family
ID=72726476
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010650573.4A Expired - Fee Related CN111763302B (en) | 2020-07-08 | 2020-07-08 | TPU (thermoplastic polyurethane) formula for thin film and preparation method thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN111763302B (en) |
Family Cites Families (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| LU76474A1 (en) * | 1976-12-24 | 1978-07-10 | ||
| GB2070041B (en) * | 1980-02-20 | 1983-06-22 | Dunlop Ltd | Polyether-urethane foams |
| JP2663390B2 (en) * | 1991-02-04 | 1997-10-15 | 工業技術院長 | Method for producing biodegradable polyurethane |
| US5138006A (en) * | 1991-02-11 | 1992-08-11 | Eastman Kodak Company | Radiation polymerizable starch ester-urethanes |
| CN1039648C (en) * | 1991-08-01 | 1998-09-02 | 诺瓦蒙特股份公司 | Polymer composition obtainable from a melt comprising a modified starch and a thermoplastic polymer |
| DE19520093A1 (en) * | 1995-06-01 | 1996-12-05 | Bayer Ag | Polymer blends containing starch and polyurethanes |
| CN1548460A (en) * | 2003-05-22 | 2004-11-24 | 徐林坤 | Dry hydroxyalkyl starch producing process |
| CN101418119B (en) * | 2008-12-02 | 2011-11-16 | 江苏大学 | Method for preparing oxidative crosslinked starch modified waterborne polyurethane hybridization material |
| US8889945B2 (en) * | 2010-12-08 | 2014-11-18 | Kimberly-Clark Worldwide, Inc. | Elastic film containing a renewable starch polymer |
| FR2992652B1 (en) * | 2012-06-27 | 2014-06-20 | Arkema France | USE OF A THERMOPLASTIC STARCH ALLOY AND TPE FOR THE MANUFACTURE OF A ULTRA-FINE IMPER-BREATHABLE ADHESIVE FILM. |
| CN102977330A (en) * | 2012-11-14 | 2013-03-20 | 华北电力大学 | Preparation method for degradable polyurethane materials |
| CN104119493B (en) * | 2014-08-05 | 2016-06-29 | 苏州巨联科技有限公司 | A kind of soft dry method water-proof air-moisture-permeable polyurethane resin and preparation method thereof |
| CN105061720B (en) * | 2015-08-06 | 2018-01-23 | 吉林师范大学 | Polyurethane material and preparation method thereof |
| CN105949424A (en) * | 2016-06-08 | 2016-09-21 | 苏州市雄林新材料科技有限公司 | TPU film for food packing and preparation method thereof |
| CN111117207A (en) * | 2019-12-26 | 2020-05-08 | 南通纳科达聚氨酯科技有限公司 | Environment-friendly photochromic TPU (thermoplastic polyurethane) automobile paint protective film |
-
2020
- 2020-07-08 CN CN202010650573.4A patent/CN111763302B/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN111763302A (en) | 2020-10-13 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN107501505B (en) | Surface layer polyurethane resin for waterproof moisture-permeable synthetic leather and preparation method thereof | |
| JP7093492B2 (en) | Eco-friendly odorless foam sole material and sole manufacturing method | |
| KR20190104057A (en) | Thermoplastic Silicone-Polyurethane Elastomer and Method for Making the Same | |
| CN104140520B (en) | High Wet-type polyurethane resin of peeling off of a kind of ultra-soft matter high resiliency hydrolysis and preparation method thereof | |
| EP3318663A1 (en) | Single fibre including thermally responsive liquid-crystal elastomer, filament yarn, and fibre product | |
| EP2484709A1 (en) | Heat resistant high moisture vapor transmission thermoplastic polyurethane | |
| CN105505280A (en) | TPU (thermoplastic polyurethane) hot melt adhesive and preparation method thereof | |
| EP1353970A1 (en) | Thermoplastic polyurethane containing structural units of ethylene oxide polyol or ethylene oxide capped propylene oxide polyol | |
| KR100878883B1 (en) | Moisture-Proof Waterproof Thermoplastic Polyurethane for Extrusion | |
| CN109485881A (en) | A kind of high-strength electronic protection takes TPU film material and preparation method thereof | |
| CN113527624B (en) | Transparent polyurethane elastomer and preparation method thereof | |
| JP5823504B2 (en) | Thermoplastic polyurethane comprising as a plasticizer glycerol esterified with at least one aliphatic carboxylic acid | |
| CN109970939A (en) | Biomass thermoplastic polyurethane | |
| CN111763302B (en) | TPU (thermoplastic polyurethane) formula for thin film and preparation method thereof | |
| CN108264626A (en) | A kind of high resilience spandex slice | |
| CN114149559B (en) | A high-strength, high-elongation thermoplastic polyurethane material and its preparation method and use | |
| CN118620374B (en) | A high-transmittance packaging material for optical communication equipment and a preparation method thereof | |
| CN103992412B (en) | Modified Huaihe River sweet potato starch prepolymer and the degradable polyurethane expanded material prepared with this prepolymer | |
| US4000117A (en) | Novel compositions | |
| CN113121985A (en) | Ultraviolet-resistant TPU film and preparation method thereof | |
| CN116376266B (en) | A thermal insulation foam shoe material containing silicon dioxide aerogel | |
| JP3968736B2 (en) | Polyurethane yarn and process for producing the same | |
| CN117777925A (en) | A reactive polyurethane soft foam hot melt adhesive | |
| CN114957605A (en) | TPU (thermoplastic polyurethane) adhesive film material for traceless underwear and preparation method thereof | |
| CN109705310B (en) | Polyurethane resin for waterproof breathable leather and preparation method thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20220107 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |