CN108042507B - Policosanol microsphere and preparation method and application thereof - Google Patents
Policosanol microsphere and preparation method and application thereof Download PDFInfo
- Publication number
- CN108042507B CN108042507B CN201711421106.9A CN201711421106A CN108042507B CN 108042507 B CN108042507 B CN 108042507B CN 201711421106 A CN201711421106 A CN 201711421106A CN 108042507 B CN108042507 B CN 108042507B
- Authority
- CN
- China
- Prior art keywords
- policosanol
- heating
- microspheres
- drying
- freeze
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- BDERNNFJNOPAEC-UHFFFAOYSA-N propan-1-ol Chemical compound CCCO BDERNNFJNOPAEC-UHFFFAOYSA-N 0.000 title claims abstract description 133
- 229960001109 policosanol Drugs 0.000 title claims abstract description 132
- 239000004005 microsphere Substances 0.000 title claims abstract description 82
- 238000002360 preparation method Methods 0.000 title claims abstract description 19
- 238000004108 freeze drying Methods 0.000 claims abstract description 39
- 239000002245 particle Substances 0.000 claims abstract description 39
- 239000007853 buffer solution Substances 0.000 claims abstract description 22
- 150000003904 phospholipids Chemical class 0.000 claims abstract description 20
- 238000003756 stirring Methods 0.000 claims abstract description 6
- 238000010438 heat treatment Methods 0.000 claims description 74
- 238000001035 drying Methods 0.000 claims description 39
- 238000005192 partition Methods 0.000 claims description 36
- QOEHNLSDMADWEF-UHFFFAOYSA-N I-Dotriacontanol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO QOEHNLSDMADWEF-UHFFFAOYSA-N 0.000 claims description 30
- ULCZGKYHRYJXAU-UHFFFAOYSA-N heptacosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCO ULCZGKYHRYJXAU-UHFFFAOYSA-N 0.000 claims description 30
- IRHTZOCLLONTOC-UHFFFAOYSA-N hexacosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCO IRHTZOCLLONTOC-UHFFFAOYSA-N 0.000 claims description 30
- CNNRPFQICPFDPO-UHFFFAOYSA-N octacosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCO CNNRPFQICPFDPO-UHFFFAOYSA-N 0.000 claims description 30
- TYWMIZZBOVGFOV-UHFFFAOYSA-N tetracosan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCO TYWMIZZBOVGFOV-UHFFFAOYSA-N 0.000 claims description 30
- REZQBEBOWJAQKS-UHFFFAOYSA-N triacontan-1-ol Chemical compound CCCCCCCCCCCCCCCCCCCCCCCCCCCCCCO REZQBEBOWJAQKS-UHFFFAOYSA-N 0.000 claims description 30
- 239000003223 protective agent Substances 0.000 claims description 29
- 238000001816 cooling Methods 0.000 claims description 27
- 238000005086 pumping Methods 0.000 claims description 24
- 229920001612 Hydroxyethyl starch Polymers 0.000 claims description 23
- 229940050526 hydroxyethylstarch Drugs 0.000 claims description 23
- 239000000725 suspension Substances 0.000 claims description 21
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 claims description 18
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 claims description 18
- 229960002666 1-octacosanol Drugs 0.000 claims description 15
- 239000003814 drug Substances 0.000 claims description 15
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 claims description 14
- 239000000463 material Substances 0.000 claims description 14
- 238000005538 encapsulation Methods 0.000 claims description 13
- 238000000034 method Methods 0.000 claims description 13
- 229940079593 drug Drugs 0.000 claims description 12
- 238000007710 freezing Methods 0.000 claims description 12
- 230000008014 freezing Effects 0.000 claims description 12
- 239000008055 phosphate buffer solution Substances 0.000 claims description 12
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 claims description 10
- 150000001720 carbohydrates Chemical class 0.000 claims description 10
- 239000000787 lecithin Substances 0.000 claims description 10
- 235000010445 lecithin Nutrition 0.000 claims description 10
- 229940067606 lecithin Drugs 0.000 claims description 10
- 125000000647 trehalose group Chemical group 0.000 claims description 10
- 239000008187 granular material Substances 0.000 claims description 8
- 238000011068 loading method Methods 0.000 claims description 8
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 claims description 7
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 7
- 229920000168 Microcrystalline cellulose Polymers 0.000 claims description 7
- 229920000881 Modified starch Polymers 0.000 claims description 7
- 239000001863 hydroxypropyl cellulose Substances 0.000 claims description 7
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 claims description 7
- 239000008101 lactose Substances 0.000 claims description 7
- 235000019359 magnesium stearate Nutrition 0.000 claims description 7
- 239000008108 microcrystalline cellulose Substances 0.000 claims description 7
- 235000019813 microcrystalline cellulose Nutrition 0.000 claims description 7
- 229940016286 microcrystalline cellulose Drugs 0.000 claims description 7
- 201000001320 Atherosclerosis Diseases 0.000 claims description 6
- WSMYVTOQOOLQHP-UHFFFAOYSA-N Malondialdehyde Chemical compound O=CCC=O WSMYVTOQOOLQHP-UHFFFAOYSA-N 0.000 claims description 5
- 230000003647 oxidation Effects 0.000 claims description 5
- 238000007254 oxidation reaction Methods 0.000 claims description 5
- TZCPCKNHXULUIY-RGULYWFUSA-N 1,2-distearoyl-sn-glycero-3-phosphoserine Chemical compound CCCCCCCCCCCCCCCCCC(=O)OC[C@H](COP(O)(=O)OC[C@H](N)C(O)=O)OC(=O)CCCCCCCCCCCCCCCCC TZCPCKNHXULUIY-RGULYWFUSA-N 0.000 claims description 4
- ZWZWYGMENQVNFU-UHFFFAOYSA-N Glycerophosphorylserin Natural products OC(=O)C(N)COP(O)(=O)OCC(O)CO ZWZWYGMENQVNFU-UHFFFAOYSA-N 0.000 claims description 4
- 208000035150 Hypercholesterolemia Diseases 0.000 claims description 4
- 208000029078 coronary artery disease Diseases 0.000 claims description 4
- VCMFGDACKBCWDX-UHFFFAOYSA-K dipotassium sodium hydrogen phosphate hydroxide Chemical compound [OH-].[Na+].P(=O)(O)([O-])[O-].[K+].[K+] VCMFGDACKBCWDX-UHFFFAOYSA-K 0.000 claims description 4
- 238000000265 homogenisation Methods 0.000 claims description 4
- AURFVNDXGLQSNN-UHFFFAOYSA-K trisodium 2-hydroxypropane-1,2,3-tricarboxylic acid phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O.OC(=O)CC(O)(C(O)=O)CC(O)=O AURFVNDXGLQSNN-UHFFFAOYSA-K 0.000 claims description 4
- 239000003826 tablet Substances 0.000 claims description 3
- CITHEXJVPOWHKC-UUWRZZSWSA-N 1,2-di-O-myristoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCCCCCCC CITHEXJVPOWHKC-UUWRZZSWSA-N 0.000 claims description 2
- CFWRDBDJAOHXSH-SECBINFHSA-N 2-azaniumylethyl [(2r)-2,3-diacetyloxypropyl] phosphate Chemical compound CC(=O)OC[C@@H](OC(C)=O)COP(O)(=O)OCCN CFWRDBDJAOHXSH-SECBINFHSA-N 0.000 claims description 2
- 208000031226 Hyperlipidaemia Diseases 0.000 claims description 2
- 239000002775 capsule Substances 0.000 claims description 2
- 229960003724 dimyristoylphosphatidylcholine Drugs 0.000 claims description 2
- 239000007924 injection Substances 0.000 claims description 2
- 238000002347 injection Methods 0.000 claims description 2
- 229940118019 malondialdehyde Drugs 0.000 claims description 2
- WTJKGGKOPKCXLL-RRHRGVEJSA-N phosphatidylcholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCCC=CCCCCCCCC WTJKGGKOPKCXLL-RRHRGVEJSA-N 0.000 claims description 2
- 230000008569 process Effects 0.000 claims description 2
- 239000008347 soybean phospholipid Substances 0.000 claims description 2
- 239000007789 gas Substances 0.000 claims 10
- 125000000837 carbohydrate group Chemical group 0.000 claims 3
- HVYWMOMLDIMFJA-DPAQBDIFSA-N cholesterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CCCC(C)C)[C@@]1(C)CC2 HVYWMOMLDIMFJA-DPAQBDIFSA-N 0.000 description 44
- 108010028554 LDL Cholesterol Proteins 0.000 description 23
- 235000012000 cholesterol Nutrition 0.000 description 22
- 239000000243 solution Substances 0.000 description 17
- 108010023302 HDL Cholesterol Proteins 0.000 description 16
- 230000000052 comparative effect Effects 0.000 description 12
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 8
- UFTFJSFQGQCHQW-UHFFFAOYSA-N triformin Chemical compound O=COCC(OC=O)COC=O UFTFJSFQGQCHQW-UHFFFAOYSA-N 0.000 description 8
- 241000700159 Rattus Species 0.000 description 7
- 210000004369 blood Anatomy 0.000 description 7
- 239000008280 blood Substances 0.000 description 7
- 230000000694 effects Effects 0.000 description 7
- 238000002156 mixing Methods 0.000 description 7
- 239000000203 mixture Substances 0.000 description 7
- 108010007622 LDL Lipoproteins Proteins 0.000 description 6
- 102000007330 LDL Lipoproteins Human genes 0.000 description 6
- 230000002829 reductive effect Effects 0.000 description 6
- 238000007873 sieving Methods 0.000 description 6
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 6
- 108010010234 HDL Lipoproteins Proteins 0.000 description 5
- 102000015779 HDL Lipoproteins Human genes 0.000 description 5
- 210000002966 serum Anatomy 0.000 description 5
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 4
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 4
- 230000037396 body weight Effects 0.000 description 4
- 239000005720 sucrose Substances 0.000 description 4
- 239000012085 test solution Substances 0.000 description 4
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 3
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 3
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 3
- 238000008214 LDL Cholesterol Methods 0.000 description 3
- 229930195725 Mannitol Natural products 0.000 description 3
- 229920002472 Starch Polymers 0.000 description 3
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 238000001914 filtration Methods 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 210000002540 macrophage Anatomy 0.000 description 3
- 239000000594 mannitol Substances 0.000 description 3
- 235000010355 mannitol Nutrition 0.000 description 3
- 239000000047 product Substances 0.000 description 3
- 239000000600 sorbitol Substances 0.000 description 3
- 235000010356 sorbitol Nutrition 0.000 description 3
- 239000008107 starch Substances 0.000 description 3
- 235000019698 starch Nutrition 0.000 description 3
- 238000005303 weighing Methods 0.000 description 3
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 2
- 241001465754 Metazoa Species 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 238000001514 detection method Methods 0.000 description 2
- 239000003085 diluting agent Substances 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 235000020828 fasting Nutrition 0.000 description 2
- 210000000497 foam cell Anatomy 0.000 description 2
- 235000013305 food Nutrition 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 210000004185 liver Anatomy 0.000 description 2
- 239000000314 lubricant Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 1
- CABVTRNMFUVUDM-VRHQGPGLSA-N (3S)-3-hydroxy-3-methylglutaryl-CoA Chemical compound O[C@@H]1[C@H](OP(O)(O)=O)[C@@H](COP(O)(=O)OP(O)(=O)OCC(C)(C)[C@@H](O)C(=O)NCCC(=O)NCCSC(=O)C[C@@](O)(CC(O)=O)C)O[C@H]1N1C2=NC=NC(N)=C2N=C1 CABVTRNMFUVUDM-VRHQGPGLSA-N 0.000 description 1
- KJTLQQUUPVSXIM-ZCFIWIBFSA-M (R)-mevalonate Chemical compound OCC[C@](O)(C)CC([O-])=O KJTLQQUUPVSXIM-ZCFIWIBFSA-M 0.000 description 1
- JLPULHDHAOZNQI-ZTIMHPMXSA-N 1-hexadecanoyl-2-(9Z,12Z-octadecadienoyl)-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCCCCCC\C=C/C\C=C/CCCCC JLPULHDHAOZNQI-ZTIMHPMXSA-N 0.000 description 1
- 235000020927 12-h fasting Nutrition 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-M Acetate Chemical compound CC([O-])=O QTBSBXVTEAMEQO-UHFFFAOYSA-M 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 208000037260 Atherosclerotic Plaque Diseases 0.000 description 1
- RGJOEKWQDUBAIZ-IBOSZNHHSA-N CoASH Chemical compound O[C@@H]1[C@H](OP(O)(O)=O)[C@@H](COP(O)(=O)OP(O)(=O)OCC(C)(C)[C@@H](O)C(=O)NCCC(=O)NCCS)O[C@H]1N1C2=NC=NC(N)=C2N=C1 RGJOEKWQDUBAIZ-IBOSZNHHSA-N 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- KJTLQQUUPVSXIM-UHFFFAOYSA-N DL-mevalonic acid Natural products OCCC(O)(C)CC(O)=O KJTLQQUUPVSXIM-UHFFFAOYSA-N 0.000 description 1
- 229920001353 Dextrin Polymers 0.000 description 1
- 239000004375 Dextrin Substances 0.000 description 1
- 102000002322 Egg Proteins Human genes 0.000 description 1
- 108010000912 Egg Proteins Proteins 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 1
- 102000000853 LDL receptors Human genes 0.000 description 1
- 108010001831 LDL receptors Proteins 0.000 description 1
- 102000004895 Lipoproteins Human genes 0.000 description 1
- 108090001030 Lipoproteins Proteins 0.000 description 1
- 241000699670 Mus sp. Species 0.000 description 1
- WHNWPMSKXPGLAX-UHFFFAOYSA-N N-Vinyl-2-pyrrolidone Chemical compound C=CN1CCCC1=O WHNWPMSKXPGLAX-UHFFFAOYSA-N 0.000 description 1
- KNAHARQHSZJURB-UHFFFAOYSA-N Propylthiouracile Chemical compound CCCC1=CC(=O)NC(=S)N1 KNAHARQHSZJURB-UHFFFAOYSA-N 0.000 description 1
- 241000209051 Saccharum Species 0.000 description 1
- TVXBFESIOXBWNM-UHFFFAOYSA-N Xylitol Natural products OCCC(O)C(O)C(O)CCO TVXBFESIOXBWNM-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 1
- 230000000879 anti-atherosclerotic effect Effects 0.000 description 1
- 239000003524 antilipemic agent Substances 0.000 description 1
- 230000009286 beneficial effect Effects 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 239000003613 bile acid Substances 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 239000000378 calcium silicate Substances 0.000 description 1
- 229910052918 calcium silicate Inorganic materials 0.000 description 1
- 235000012241 calcium silicate Nutrition 0.000 description 1
- CJZGTCYPCWQAJB-UHFFFAOYSA-L calcium stearate Chemical compound [Ca+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O CJZGTCYPCWQAJB-UHFFFAOYSA-L 0.000 description 1
- 235000013539 calcium stearate Nutrition 0.000 description 1
- 239000008116 calcium stearate Substances 0.000 description 1
- OYACROKNLOSFPA-UHFFFAOYSA-N calcium;dioxido(oxo)silane Chemical compound [Ca+2].[O-][Si]([O-])=O OYACROKNLOSFPA-UHFFFAOYSA-N 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000001768 carboxy methyl cellulose Substances 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 239000007766 cera flava Substances 0.000 description 1
- 239000003153 chemical reaction reagent Substances 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 229940099352 cholate Drugs 0.000 description 1
- -1 cholesteryl ester Chemical class 0.000 description 1
- BHQCQFFYRZLCQQ-OELDTZBJSA-N cholic acid Chemical compound C([C@H]1C[C@H]2O)[C@H](O)CC[C@]1(C)[C@@H]1[C@@H]2[C@@H]2CC[C@H]([C@@H](CCC(O)=O)C)[C@@]2(C)[C@@H](O)C1 BHQCQFFYRZLCQQ-OELDTZBJSA-N 0.000 description 1
- RGJOEKWQDUBAIZ-UHFFFAOYSA-N coenzime A Natural products OC1C(OP(O)(O)=O)C(COP(O)(=O)OP(O)(=O)OCC(C)(C)C(O)C(=O)NCCC(=O)NCCS)OC1N1C2=NC=NC(N)=C2N=C1 RGJOEKWQDUBAIZ-UHFFFAOYSA-N 0.000 description 1
- 239000005516 coenzyme A Substances 0.000 description 1
- 229940093530 coenzyme a Drugs 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 229960000913 crospovidone Drugs 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- KDTSHFARGAKYJN-UHFFFAOYSA-N dephosphocoenzyme A Natural products OC1C(O)C(COP(O)(=O)OP(O)(=O)OCC(C)(C)C(O)C(=O)NCCC(=O)NCCS)OC1N1C2=NC=NC(N)=C2N=C1 KDTSHFARGAKYJN-UHFFFAOYSA-N 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 235000019425 dextrin Nutrition 0.000 description 1
- 235000005911 diet Nutrition 0.000 description 1
- 230000000378 dietary effect Effects 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- FPAFDBFIGPHWGO-UHFFFAOYSA-N dioxosilane;oxomagnesium;hydrate Chemical compound O.[Mg]=O.[Mg]=O.[Mg]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O.O=[Si]=O FPAFDBFIGPHWGO-UHFFFAOYSA-N 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 235000013345 egg yolk Nutrition 0.000 description 1
- 210000002969 egg yolk Anatomy 0.000 description 1
- 201000005577 familial hyperlipidemia Diseases 0.000 description 1
- 150000002191 fatty alcohols Chemical class 0.000 description 1
- 210000003608 fece Anatomy 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 235000001727 glucose Nutrition 0.000 description 1
- 230000031700 light absorption Effects 0.000 description 1
- 230000003859 lipid peroxidation Effects 0.000 description 1
- 150000002632 lipids Chemical class 0.000 description 1
- 108010022197 lipoprotein cholesterol Proteins 0.000 description 1
- 210000005229 liver cell Anatomy 0.000 description 1
- HCWCAKKEBCNQJP-UHFFFAOYSA-N magnesium orthosilicate Chemical compound [Mg+2].[Mg+2].[O-][Si]([O-])([O-])[O-] HCWCAKKEBCNQJP-UHFFFAOYSA-N 0.000 description 1
- 239000000395 magnesium oxide Substances 0.000 description 1
- CPLXHLVBOLITMK-UHFFFAOYSA-N magnesium oxide Inorganic materials [Mg]=O CPLXHLVBOLITMK-UHFFFAOYSA-N 0.000 description 1
- 235000012245 magnesium oxide Nutrition 0.000 description 1
- 239000000391 magnesium silicate Substances 0.000 description 1
- 229910052919 magnesium silicate Inorganic materials 0.000 description 1
- 235000019792 magnesium silicate Nutrition 0.000 description 1
- AXZKOIWUVFPNLO-UHFFFAOYSA-N magnesium;oxygen(2-) Chemical compound [O-2].[Mg+2] AXZKOIWUVFPNLO-UHFFFAOYSA-N 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- HEBKCHPVOIAQTA-UHFFFAOYSA-N meso ribitol Natural products OCC(O)C(O)C(O)CO HEBKCHPVOIAQTA-UHFFFAOYSA-N 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 235000010981 methylcellulose Nutrition 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 229920000523 polyvinylpolypyrrolidone Polymers 0.000 description 1
- 235000013809 polyvinylpolypyrrolidone Nutrition 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 229920003124 powdered cellulose Polymers 0.000 description 1
- 235000019814 powdered cellulose Nutrition 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 229960002662 propylthiouracil Drugs 0.000 description 1
- 238000012827 research and development Methods 0.000 description 1
- 230000004141 reverse cholesterol transport Effects 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 210000000813 small intestine Anatomy 0.000 description 1
- 239000011734 sodium Substances 0.000 description 1
- 229910052708 sodium Inorganic materials 0.000 description 1
- 235000015424 sodium Nutrition 0.000 description 1
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 1
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 208000010110 spontaneous platelet aggregation Diseases 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 125000000185 sucrose group Chemical group 0.000 description 1
- 239000006228 supernatant Substances 0.000 description 1
- QGVNJRROSLYGKF-UHFFFAOYSA-N thiobarbital Chemical compound CCC1(CC)C(=O)NC(=S)NC1=O QGVNJRROSLYGKF-UHFFFAOYSA-N 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- YNJBWRMUSHSURL-UHFFFAOYSA-N trichloroacetic acid Chemical compound OC(=O)C(Cl)(Cl)Cl YNJBWRMUSHSURL-UHFFFAOYSA-N 0.000 description 1
- 210000003462 vein Anatomy 0.000 description 1
- 238000010792 warming Methods 0.000 description 1
- 239000000811 xylitol Substances 0.000 description 1
- 235000010447 xylitol Nutrition 0.000 description 1
- HEBKCHPVOIAQTA-SCDXWVJYSA-N xylitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)CO HEBKCHPVOIAQTA-SCDXWVJYSA-N 0.000 description 1
- 229960002675 xylitol Drugs 0.000 description 1
- XOOUIPVCVHRTMJ-UHFFFAOYSA-L zinc stearate Chemical compound [Zn+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O XOOUIPVCVHRTMJ-UHFFFAOYSA-L 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/5005—Wall or coating material
- A61K9/5015—Organic compounds, e.g. fats, sugars
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23L—FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES, NOT OTHERWISE PROVIDED FOR; PREPARATION OR TREATMENT THEREOF
- A23L33/00—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof
- A23L33/10—Modifying nutritive qualities of foods; Dietetic products; Preparation or treatment thereof using additives
- A23L33/115—Fatty acids or derivatives thereof; Fats or oils
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23P—SHAPING OR WORKING OF FOODSTUFFS, NOT FULLY COVERED BY A SINGLE OTHER SUBCLASS
- A23P10/00—Shaping or working of foodstuffs characterised by the products
- A23P10/30—Encapsulation of particles, e.g. foodstuff additives
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/045—Hydroxy compounds, e.g. alcohols; Salts thereof, e.g. alcoholates
-
- A—HUMAN NECESSITIES
- A23—FOODS OR FOODSTUFFS; TREATMENT THEREOF, NOT COVERED BY OTHER CLASSES
- A23V—INDEXING SCHEME RELATING TO FOODS, FOODSTUFFS OR NON-ALCOHOLIC BEVERAGES AND LACTIC OR PROPIONIC ACID BACTERIA USED IN FOODSTUFFS OR FOOD PREPARATION
- A23V2002/00—Food compositions, function of food ingredients or processes for food or foodstuffs
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Polymers & Plastics (AREA)
- Food Science & Technology (AREA)
- Epidemiology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Animal Behavior & Ethology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Mycology (AREA)
- Nutrition Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Medicinal Preparation (AREA)
Abstract
The invention relates to policosanol microspheres and a preparation method and application thereof. The policosanol microspheres comprise policosanol and phospholipid, wherein the weight ratio of the policosanol to the phospholipid is (1:3) - (1:9), and the particle size of the policosanol microspheres is 0.01-1 μm; the preparation method comprises dispersing policosanol and phospholipid in buffer solution according to proportion, stirring, homogenizing under high pressure, and freeze drying. The policosanol microsphere disclosed by the invention is stable in quality, high in bioavailability, easy to absorb by a human body, small in dosage and better in safety.
Description
Technical Field
The invention relates to policosanol microspheres and a preparation method thereof, belonging to the field of pharmaceutical preparations.
Background
Policosanol (also called policosanol) is a natural chemical medicine containing 8 kinds of primary fatty alcohols and extracted from Saccharum sinensis Roxb and Cera flava, and has chemical formula of CH3(CH2)nCH2OH, chain length from 24 to 34 carbon atoms. Policosanol has the effects of inhibiting cholesterol synthesis, improving the blood clearance rate of low-density lipoprotein (LDL), effectively reducing the level of low-density lipoprotein cholesterol (LDL-C), improving the level of high-density lipoprotein cholesterol (HDL-C), obviously inhibiting platelet aggregation and the like, and is a novel safe, effective and easily-tolerated lipid-lowering agent.
High density lipoprotein cholesterol (HDL-C), which is cholesterol carried by high density lipoprotein molecules, is an endogenous cholesteryl ester that is transported in the reverse direction, transports it into the liver, and then clears the blood. The high density lipoprotein contains 20-30% of total cholesterol of human. High density lipoprotein cholesterol is colloquially understood as "good" cholesterol, anti-atherosclerotic cholesterol: after macrophages engulf low-density lipoprotein (LDL) or modified LDL, a large amount of cholesterol is accumulated to form foam cells, and the foam cells are deposited on the blood vessel wall to form early atheromatous plaque, so that RCT of the macrophages plays the most important role in the development of atherosclerosis. HDL promotes the efflux of excess cholesterol from macrophages to the liver, where it is secreted in the form of bile acids to the small intestine and excreted in the feces. HDL plays a role in resisting atherosclerosis by participating in the reverse cholesterol transport process. Low density lipoprotein cholesterol (LDL-C): cholesterol is present in the blood in the form of lipoproteins. LDL-cholesterol can be colloquially understood as "bad" cholesterol because elevated LDL-cholesterol (LDL-C) levels increase the risk of coronary heart disease. It has been clearly noted in 7 th edition of internal science that low density lipoprotein cholesterol (LDL-C) is a well-defined independent risk factor for atherosclerosis, and that it plays a role in oxidatively modified low density lipoprotein cholesterol (Ox-LDL-C). Prevention of LDL-C oxidation is a critical factor.
Policosanol inhibits cholesterol biosynthesis during acetate consumption and mevalonate synthesis steps, but it does not directly inhibit the activity of mevalonyl coenzyme A (HMG-CoA). Policosanol can also increase the low density lipoprotein uptake by liver cells by increasing low density lipoprotein receptors, thereby reducing the serum low density lipoprotein-carried low density lipoprotein cholesterol level; policosanol has good effects of reducing cholesterol, resisting platelet and inhibiting lipid peroxidation; therefore, the policosanol has better effect on preventing atherosclerosis and coronary heart disease.
Although policosanol has better activities in reducing blood lipid, cholesterol and low-density lipoprotein cholesterol, increasing high-density lipoprotein cholesterol and the like, the difficult problems of difficult solubility and low bioavailability are solved, and the research and development of the dosage form which has high bioavailability, good curative effect and stable dosage form have great significance.
Disclosure of Invention
The invention provides policosanol microspheres and a preparation method and application thereof, and in order to achieve the purpose, the invention adopts the following technical scheme:
a policosanol microsphere comprises policosanol and phospholipid, wherein the weight ratio of the policosanol to the phospholipid is (1:3) - (1:9), and the particle size of the policosanol microsphere is 0.01-1 μm.
Preferably, the particle size of the policosanol microspheres is 0.01-0.6 μm, and more preferably 0.1-0.2 μm.
Preferably, the weight ratio of the policosanol to the phospholipid is (1:5) - (1: 6).
Further, the encapsulation rate of the policosanol microspheres is more than or equal to 93%, and/or the drug loading rate is more than or equal to 14.0%, and/or the leakage rate is less than or equal to 0.7%, and/or the oxidation degree of phospholipid thereof is calculated by the content of malonaldehyde, and the content of malonaldehyde is less than or equal to 2.5 ng/mL.
Further, the polycosanols comprise tetracosanol, hexacosanol, heptacosanol, octacosanol, triacontanol, dotriacontanol;
preferably, the polycosanol contains 0.5-1.5 percent of tetracosanol (mass fraction, the same below), 5-10 percent of hexacosanol, 1-5 percent of heptacosanol, 60-75 percent of octacosanol, 5-15 percent of triacontanol and 3-8 percent of dotriacontanol; more preferably, the policosanol contains: 0.5-1% of tetracosanol, 6-8% of hexacosanol, 2-4% of heptacosanol, 65-72% of octacosanol, 7-11% of triacontanol and 3-6% of dotriacontanol.
Further, the phospholipid comprises one or more of lecithin, phosphatidylserine, cephalin, soybean phospholipid, dipalmitoyl- α phosphatidylcholine and dimyristoyl phosphatidylcholine, and more preferably lecithin or phosphatidylserine.
The invention also provides a preparation method of the policosanol microspheres, which comprises the following steps:
1) dispersing policosanol and phospholipid in a buffer solution according to the proportion, and fully and uniformly stirring;
2) heating the suspension obtained in the step 1) to 85-99 ℃, homogenizing under high pressure and cooling;
3) adding a freeze-drying protective agent into the material obtained in the step 2), and freeze-drying to obtain the product.
Further, the pH value of the buffer solution in the step 1) is 7.0-8.5, and the buffer solution can be any one of phosphate buffer solution, disodium hydrogen phosphate-sodium citrate buffer solution and dipotassium hydrogen phosphate-sodium hydroxide buffer solution.
Further, the particle size of the policosanol in the step 1) is 1-10 μm, and preferably 3-4 μm.
Further, step 2) is preferably homogenized 5-15 times, each time for generally 1-2 minutes. Preferably, the homogenized material is cooled to 1-10 ℃.
Further, in step 3), preferably, the saccharide is one or more of trehalose, sucrose, mannitol, sorbitol, and the like. Preferably, the amount of the lyoprotectant is 1mg/mL-30 mg/mL.
Further, the freeze-drying method of step 3) comprises: feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30 Pa.
Specifically, the preparation method of the policosanol microspheres comprises the following steps:
1) dispersing policosanol and phospholipid in a buffer solution according to a ratio, and stirring for 20-90 minutes at the temperature of 1-40 ℃; the buffer solution is any one of phosphate buffer solution, disodium hydrogen phosphate-sodium citrate buffer solution and dipotassium hydrogen phosphate-sodium hydroxide buffer solution, and the pH value of the buffer solution is 7.0-8.5; the particle size of the policosanol is 1-10 mu m;
2) heating the suspension obtained in the step 1) to 85-99 ℃, homogenizing for 5-15 times under high pressure for 1-2 minutes each time, wherein the high-pressure homogenizing pressure is 2800-3200 psi; cooling the material to 1-10 ℃ after high-pressure homogenization;
3) adding a freeze-drying protective agent into the material obtained in the step 2), and freeze-drying to obtain the material;
the freeze-drying protective agent is saccharide and hydroxyethyl starch, wherein the weight ratio of the saccharide to the hydroxyethyl starch is (1:1) - (2: 1); the saccharide is one or more of trehalose, sucrose, mannitol and sorbitol; the dosage of the freeze-drying protective agent is 1mg/mL-30 mg/mL;
the freeze-drying method comprises the following steps: feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30 Pa.
The invention also comprises policosanol microspheres prepared by the method.
The invention also comprises the application of the policosanol microspheres in the preparation of medicines, health-care foods, foods and the like; in particular to the application in preparing the medicine for treating hyperlipemia, hypercholesterolemia, atherosclerosis, coronary heart disease and other diseases.
The policosanol microspheres can be prepared into various available formulations in the field, and the specific application formulations comprise tablets, granules, capsules, injections and the like.
The invention also provides a policosanol tablet, which comprises the policosanol microspheres and auxiliary materials and can be prepared by a conventional method in the field.
The auxiliary materials are diluent, disintegrant, glidant and lubricant; the diluent is one or more of starch, dextrin, lactose, glucose, microcrystalline cellulose, sorbitol, mannitol and xylitol, and the starch is potato starch, corn starch and pregelatinized starch; the disintegrating agent is one or more of sodium carboxymethylcellulose, powdered cellulose, crospovidone, methylcellulose, sodium carboxymethyl starch and hydroxypropyl cellulose; the glidant is one of magnesium silicate, magnesium oxide, calcium silicate and talcum powder; the lubricant is one of calcium stearate, magnesium stearate and zinc stearate.
Specifically, the policosanol tablet comprises, by weight, 10-25 parts of policosanol microspheres, 30-50 parts of lactose, 20-40 parts of pregelatinized starch, 5-15 parts of microcrystalline cellulose, 5-15 parts of hydroxypropyl cellulose and 0.5-2 parts of magnesium stearate.
The starting materials used in the present invention are commercially available or may be prepared by methods conventional in the art.
On the basis of the common knowledge in the field, the above preferred conditions can be combined with each other to obtain the preferred embodiments of the invention.
Further, it is to be understood that terms such as "comprising," "including," "containing," and the like, also include "consisting of … …," "consisting of … …," "made of … …," and the like, herein.
The invention has the following beneficial effects: the policosanol microsphere disclosed by the invention is stable in quality, high in bioavailability, easy to absorb by a human body, small in dosage and better in safety.
Detailed Description
The following examples are intended to illustrate the invention but are not intended to limit the scope of the invention. The examples do not show the specific techniques or conditions, according to the technical or conditions described in the literature in the field, or according to the product specifications. The reagents or instruments used are conventional products available from regular distributors, not indicated by the manufacturer.
Policosanol used below was prepared according to the method described in CN 106187677A.
Example 1
A preparation method of policosanol microspheres comprises the following steps:
1Kg of policosanol (containing 0.73 percent of tetracosanol, 7.29 percent of hexacosanol, 2.81 percent of heptacosanol, 69.23 percent of octacosanol, 9.36 percent of triacontanol and 5.13 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 6Kg of lecithin is uniformly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 deg.C, homogenizing for 10 times each time for 1 min under 3000spi pressure, cooling to 5 deg.C, and retaining microspheres with particle size of 0.01-1.0 μm; adding the obtained microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is trehalose and hydroxyethyl starch, and the mass ratio of the trehalose to the hydroxyethyl starch is 1: 1; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Example 2
A preparation method of policosanol microspheres comprises the following steps:
1Kg of policosanol (containing 0.71% of tetracosanol, 7.34% of hexacosanol, 2.59% of heptacosanol, 69.1% of octacosanol, 9.34% of triacontanol and 5.22% of dotriacontanol) is crushed into particles with the average particle size of 4 mu m, 5Kg of phosphatidylserine is uniformly dispersed in 80LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 40 minutes at the temperature of 35 ℃ to obtain suspension; heating the suspension to 94 ℃, homogenizing for 10 times for 1 minute each time under 2800spi pressure, cooling to 5 ℃, and intercepting microspheres with the particle size of 0.01-1.0 μm; adding the obtained microspheres into a solution with the concentration of a freeze-drying protective agent of 25mg/mL, wherein the freeze-drying protective agent is sucrose and hydroxyethyl starch, and the mass ratio of the sucrose to the hydroxyethyl starch is 1.2: 1; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Comparative example 1
1Kg of policosanol (containing 4.0 percent of tetracosanol, 4.0 percent of hexacosanol, 8.0 percent of heptacosanol, 50.0 percent of octacosanol, 20.0 percent of triacontanol and 10.0 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 6Kg of lecithin is evenly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 deg.C, homogenizing for 10 times each time for 1 min under 3000spi pressure, cooling to 5 deg.C, and retaining microspheres with particle size of 0.01-1.0 μm; adding the obtained microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is trehalose and hydroxyethyl starch, and the mass ratio of the trehalose to the hydroxyethyl starch is 1: 1; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Comparative example 2
1Kg of policosanol (containing 0.73 percent of tetracosanol, 7.29 percent of hexacosanol, 2.81 percent of heptacosanol, 69.23 percent of octacosanol, 9.36 percent of triacontanol and 5.13 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 2Kg of lecithin is uniformly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 deg.C, homogenizing for 10 times each time for 1 min under 3000spi pressure, cooling to 5 deg.C, and retaining microspheres with particle size of 0.01-1.0 μm; adding the obtained microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is trehalose and hydroxyethyl starch, and the mass ratio of the trehalose to the hydroxyethyl starch is 1: 1; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Comparative example 3
1Kg of policosanol (containing 0.73 percent of tetracosanol, 7.29 percent of hexacosanol, 2.81 percent of heptacosanol, 69.23 percent of octacosanol, 9.36 percent of triacontanol and 5.13 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 6Kg of lecithin is uniformly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 ℃, homogenizing for 10 times for 1 minute each time under the pressure of 1800spi, cooling to 5 ℃, and intercepting microspheres with the particle size of 0.01-1.0 μm; adding the obtained microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is trehalose and hydroxyethyl starch, and the mass ratio of the trehalose to the hydroxyethyl starch is 1: 1; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Comparative example 4
1Kg of policosanol (containing 0.73 percent of tetracosanol, 7.29 percent of hexacosanol, 2.81 percent of heptacosanol, 69.23 percent of octacosanol, 9.36 percent of triacontanol and 5.13 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 6Kg of lecithin is uniformly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 deg.C, homogenizing for 10 times each time for 1 min under 3000spi pressure, cooling to 5 deg.C, and retaining microspheres with particle size of 0.01-1.0 μm; adding the microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is trehalose; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Comparative example 5
1Kg of policosanol (containing 0.73 percent of tetracosanol, 7.29 percent of hexacosanol, 2.81 percent of heptacosanol, 69.23 percent of octacosanol, 9.36 percent of triacontanol and 5.13 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 6Kg of lecithin is uniformly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 deg.C, homogenizing for 10 times each time for 1 min under 3000spi pressure, cooling to 5 deg.C, and retaining microspheres with particle size of 0.01-1.0 μm; adding the microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is hydroxyethyl starch; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Comparative example 6
1Kg of policosanol (containing 0.73 percent of tetracosanol, 7.29 percent of hexacosanol, 2.81 percent of heptacosanol, 69.23 percent of octacosanol, 9.36 percent of triacontanol and 5.13 percent of dotriacontanol) is crushed into particles with the average particle size of 3 mu m, 6Kg of lecithin is uniformly dispersed in 120LpH7.0 phosphate buffer solution, and 3500r/min is stirred for 60 minutes at the temperature of 30 ℃ to obtain suspension; heating the suspension to 90 deg.C, homogenizing for 10 times each time for 1 min under 3000spi pressure, cooling to 5 deg.C, and retaining microspheres with particle size of 0.01-1.0 μm; adding the obtained microspheres into a solution with the concentration of a freeze-drying protective agent of 20mg/mL, wherein the freeze-drying protective agent is trehalose and hydroxyethyl starch, and the mass ratio of the trehalose to the hydroxyethyl starch is 1: 3; feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition plate to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 pa; the vacuum degree of the front box in the whole drying process is not higher than 30Pa, and the policosanol microspheres are obtained.
Application example 1
Respectively and accurately weighing 51.8g of lactose, 44.4g of pregelatinized starch and 14.8g of microcrystalline cellulose, respectively sieving with a 100-mesh sieve, uniformly mixing with 20.7g of policosanol microspheres prepared in example 1, dissolving 14.8g of hydroxypropyl cellulose in 100g of water to prepare a solution, adding the solution into the mixture to prepare a soft capsule, drying at 50 ℃, sieving dry granules with a 16-mesh sieve, adding 1.5g of magnesium stearate, uniformly mixing, and tabletting, wherein each tablet contains 3mg of policosanol.
Application example 2
43.8g of lactose, 37.5g of pregelatinized starch and 12.5g of microcrystalline cellulose are accurately weighed, respectively sieved by a 100-mesh sieve, and uniformly mixed with 17.5g of policosanol microspheres prepared in example 2, 12.5g of hydroxypropyl cellulose is dissolved in 100g of water to prepare a solution, the solution is added into the mixture to prepare soft granules, the soft granules are dried at 50 ℃, dry granules are sieved by a 16-mesh sieve, 1.3g of magnesium stearate is added, the mixture is uniformly mixed and tabletted, and each tablet contains 3mg of policosanol.
Application example 3
Respectively and accurately weighing 51.2g of lactose, 43.9g of pregelatinized starch and 14.6g of microcrystalline cellulose, respectively sieving with a 100-mesh sieve, and 20.5g of policosanol microspheres of comparative example 1, dissolving 14.6g of hydroxypropyl cellulose in 100g of water to prepare a solution, adding the solution into the mixture to prepare soft granules, drying at 50 ℃, sieving dry granules with a 16-mesh sieve, adding 1.5g of magnesium stearate, uniformly mixing, and tabletting, wherein each tablet contains 3mg of policosanol.
Application example 4
Respectively and accurately weighing 125g of lactose, 107.1g of pregelatinized starch and 35.7g of microcrystalline cellulose, respectively sieving with a 100-mesh sieve, respectively, uniformly mixing with 50g of polycosanol (containing 0.73% of tetracosanol, 7.29% of hexacosanol, 2.81% of heptacosanol, 69.23% of octacosanol, 9.36% of triacontanol and 5.13% of dotriacontanol), dissolving 35.7g of hydroxypropyl cellulose in 250g of water to prepare a solution, adding the solution into the mixture to prepare a soft solution, drying at 50 ℃, sieving with a 16-mesh sieve, adding 3.6g of magnesium stearate, uniformly mixing, and tabletting, wherein each tablet contains 5mg of polycosanol.
Experimental example 1
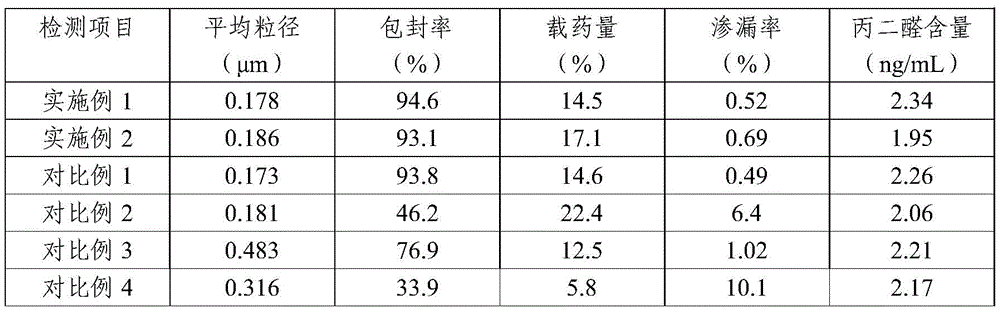
The policosanol microspheres prepared in examples 1-2 and comparative examples 1-6 were tested, and the results are shown in Table 1 below. The detection method comprises the following steps:
1. average particle size detection
0.1g of policosanol microspheres obtained in examples 1-2 and comparative examples 1-6 were weighed, diluted with distilled water to 10mL, and the particle size of the policosanol microspheres was measured by a Zetasizer Nano ZS 90 (Malvern) laser particle sizer, and the average particle size was measured.
2. Encapsulation efficiency and drug loading measurements
Putting 1mg of policosanol microspheres obtained in the examples 1-2 and the comparative examples 1-6 into a 10mL volumetric flask, performing ultrasonic demulsification by using chloroform, performing constant volume, shaking uniformly, passing through a 0.22 mu m filter membrane, injecting 1 mu L of samples, and calculating the total drug amount of the policosanol; and adding acetone into 1mg policosanol microspheres, filtering to obtain filtrate, removing acetone, diluting to 10mL with chloroform, shaking, filtering with 0.22 μm filter membrane, introducing 1 μ L sample, and determining total content of free policosanol.
The encapsulation efficiency and drug loading were calculated according to the following formula:
the encapsulation rate is × 100 percent of the drug content in the policosanol microspheres/the total drug content of encapsulated and non-encapsulated policosanol microspheres
The drug loading rate is × 100 percent of the drug content of the policosanol microspheres/the total weight of the policosanol microspheres
3. Determination of leakage Rate
After the encapsulation efficiency of the policosanol microspheres obtained in examples 1 to 2 and comparative examples 1 to 6 is measured on the same day, the encapsulation efficiency is measured after the microspheres are stored at 4 ℃ for seven days, and the leakage rate is calculated according to the following formula:
the leakage rate (encapsulation rate measured on the day by a certain period)/the encapsulation rate measured on the day of × 100%
4. Degree of oxidation of phospholipid
Preparing TTH test solution: trichloroacetic acid (TCA)30g, Thiobarbital (TBA)0.75g, adding 0.25M hydrochloric acid 200ml, warming to dissolve, cooling, and filtering for use.
And (3) determining the oxidation degree of the policosanol microspheres by adopting a malondialdehyde method. Precisely sucking 1mL of policosanol microspheres, placing in a 10mL centrifuge tube, adding 5mL of TTH test solution, mixing, heating in water bath at 100 ℃ for 35min, taking out, cooling, adding TTH test solution to scale, mixing, centrifuging at 4000r/min for 10min, taking supernatant, and determining absorbance at 534nm with TTH test solution as blank.
The calculation formula of the content of the malonaldehyde is as follows: c ═ A/ba
Wherein A is absorbance; b is the length of the light absorption pool in cm; a is the absorption coefficient in L.g-1.cm-1。
TABLE 1
The results in table 1 show that when the ratio of policosanol to phospholipid is 1:2, the encapsulation efficiency of the obtained policosanol microspheres is obviously reduced, and the leakage rate is also high; when the homogenizing pressure is lower, the obtained policosanol microspheres have larger average particle size, obviously reduced encapsulation efficiency, reduced drug loading and higher leakage rate; when the freeze-drying protective agent is trehalose or hydroxyethyl starch, the encapsulation rate of the obtained policosanol microspheres is obviously reduced, the drug-loading rate is also obviously reduced, and the leakage rate is also higher; when the ratio of the trehalose to the hydroxyethyl starch is 1:3, the encapsulation rate and the drug-loading rate of the policosanol microspheres are reduced, and the leakage rate is not changed remarkably.
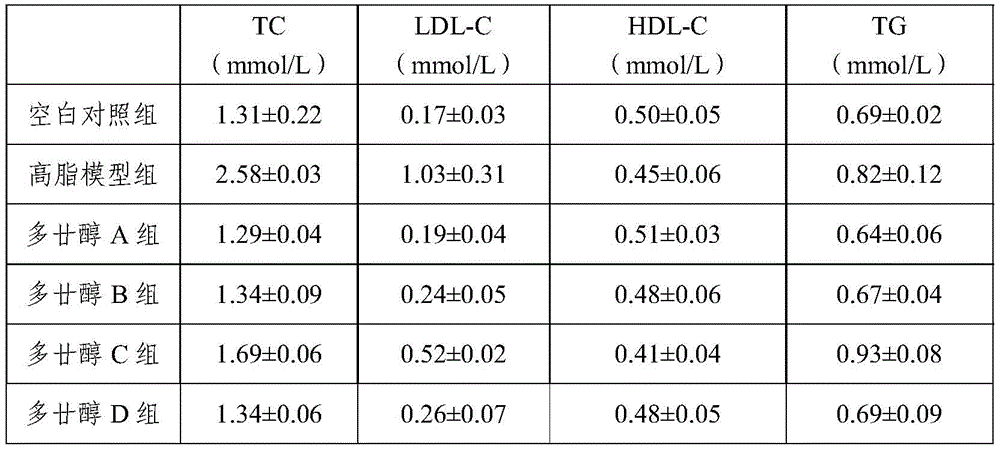
Experimental example 2 animal Experimental study
High-fat feed: 83% of common feed, 10% of lard, 5% of egg yolk, 1.6% of cholesterol, 0.2% of propyl thiouracil and 0.2% of cholate.
Selecting 50 Wistar pure-bred rats with male and female halves, clean grade and weight of 160-180 g. Before the experiment, 60 Wistar pure rats are fed with basic feed under the standard experimental environment, blood is collected from orbital veins after fasting for 12h, serum is prepared, and TC (total cholesterol), LDL-C (low density lipoprotein cholesterol), HDL-C (high density lipoprotein cholesterol) and TG (triglyceride) of the serum of each group of rats are measured. Wistar inbred rats were randomly divided into 2 groups, a blank control group (10), and a high-fat model group (40). The blank control group is fed with basal feed for 4 weeks, the high-fat model group is fed with the high-fat feed for 4 weeks, a rat dietetic hyperlipidemia model is started to be established, and after fasting for 12 hours, orbital venous blood collection is carried out to respectively measure TC (total cholesterol), LDL-C (low density lipoprotein cholesterol), HDL-C (high density lipoprotein cholesterol) and TG (triglyceride) of the serum of two groups of rats.
In the 5 th to 12 th weeks, the high-fat model group (40) was randomly divided into 4 groups, each group was 10, one group was policosanol a group (the policosanol tablet prepared in application example 1 was used, the policosanol content was 3 mg/tablet), one group was policosanol B group (the policosanol tablet prepared in application example 2 was used, the policosanol content was 3 mg/tablet), one group was policosanol C group (the policosanol tablet prepared in application comparative example 1 was used, the policosanol content was 3 mg/tablet), and one group was policosanol D group (the ordinary policosanol tablet obtained in application example 4 was used, the policosanol content was 5 mg/tablet), and the above 4 groups were fed with the basic feed in such a manner that the administration mode was intragastric lavage, and the drug amount was: policosanol A group is 3mg per Kg of body weight policosanol per day, policosanol B group is 3mg per Kg of body weight policosanol per day, policosanol C group is 3mg per Kg of body weight policosanol per day, and policosanol D group is 5mg per Kg of body weight policosanol per day. After 12 weeks, the serum of rats was subjected to orbital venous blood collection for 12 hours after fasting to measure TC (total cholesterol), LDL-C (low density lipoprotein cholesterol), HDL-C (high density lipoprotein cholesterol), and TG (triglyceride). The results are shown in Table 2 below.
From the results in Table 2, it is clear that after 8 weeks of treatment, total cholesterol, triglyceride and LDL cholesterol were decreased and HDL cholesterol was increased in 4 groups of mice, but the effect of policosanol group C was lower than that of the other groups.
Typical cases
Zhangqi, male, age 48, hypercholesterolemia, before taking: total cholesterol 6.36mmol/L, triglyceride 2.67mmol/L, high density lipoprotein cholesterol 0.84mmol/L, low density lipoprotein cholesterol 4.3 mmol/L; after taking policosanol tablets prepared in application example 1 for 2 months, the total cholesterol is 4.08mmol/L, the triglyceride is 1.53mmol/L, the high-density lipoprotein cholesterol is 1.25mmol/L, and the low-density lipoprotein cholesterol is 2.13 mmol/L.
Polygonal disease, female, age 62, hypercholesterolemia, before taking: total cholesterol 7.56mmol/L, triglyceride 1.60mmol/L, high density lipoprotein cholesterol 1.35mmol/L, low density lipoprotein cholesterol 5.48 mmol/L; after taking policosanol tablets prepared in application example 1 for 2 months, the total cholesterol is 5.51mmol/L, the triglyceride is 1.98mmol/L, the high density lipoprotein cholesterol is 1.66mmol/L, and the low density lipoprotein cholesterol is 2.95 mmol/L.
Although the invention has been described in detail hereinabove with respect to a general description and specific embodiments thereof, it will be apparent to those skilled in the art that modifications or improvements may be made thereto based on the invention. Accordingly, such modifications and improvements are intended to be within the scope of the invention as claimed.
Claims (14)
1. A policosanol microsphere comprises policosanol and phospholipid, wherein the weight ratio of the policosanol to the phospholipid is (1:3) - (1:9), and the particle size of the policosanol microsphere is 0.01-1 μm;
the polycosanol contains 0.5-1.5% of tetracosanol, 5-10% of hexacosanol, 1-5% of heptacosanol, 60-75% of octacosanol, 5-15% of triacontanol and 3-8% of dotriacontanol;
the preparation method of the policosanol microspheres comprises the following steps:
1) dispersing policosanol and phospholipid in a buffer solution according to the proportion, and fully and uniformly stirring;
2) heating the suspension obtained in the step 1) to 85-99 ℃, homogenizing under high pressure and cooling; the high pressure homogenization pressure is 2800psi-3200 psi;
3) adding a freeze-drying protective agent into the material obtained in the step 2), and freeze-drying to obtain the material; the freeze-drying protective agent is saccharide and hydroxyethyl starch, wherein the weight ratio of the saccharide to the hydroxyethyl starch is (1:1) - (2: 1); the saccharide is trehalose.
2. The policosanol microsphere of claim 1, wherein the weight ratio of policosanol to phospholipids is (1:5) - (1: 6); and/or the particle size of the policosanol microspheres is 0.01-0.6 mu m.
3. The policosanol microsphere of claim 2, wherein the particle size of the policosanol microsphere is 0.1-0.2 μm.
4. The policosanol microspheres of any one of claims 1-3, wherein the encapsulation efficiency of the policosanol microspheres is not less than 93%, and/or the drug loading is not less than 14.0%, and/or the leakage rate is not more than 0.7%, and/or the oxidation degree of phospholipid thereof is not more than 2.5ng/mL based on the content of malondialdehyde.
5. The policosanol microspheres of any one of claims 1-3, wherein the phospholipids comprise one or more of lecithin, phosphatidylserine, cephalin, soya lecithin, dipalmitoyl- α phosphatidylcholine, dimyristoyl phosphatidylcholine and/or,
the polycosanol comprises tetracosanol, hexacosanol, heptacosanol, octacosanol, triacontanol and dotriacontanol.
6. The policosanol microsphere of claim 5, wherein the policosanol comprises: 0.5-1% of tetracosanol, 6-8% of hexacosanol, 2-4% of heptacosanol, 65-72% of octacosanol, 7-11% of triacontanol and 3-6% of dotriacontanol.
7. The preparation method of the policosanol microspheres according to any one of claims 1 to 6, which is characterized by comprising the following steps:
1) dispersing policosanol and phospholipid in a buffer solution according to the proportion, and fully and uniformly stirring;
2) heating the suspension obtained in the step 1) to 85-99 ℃, homogenizing under high pressure and cooling; the high pressure homogenization pressure is 2800psi to 3200 psi;
3) adding a freeze-drying protective agent into the material obtained in the step 2), and freeze-drying to obtain the material; the freeze-drying protective agent is saccharide and hydroxyethyl starch, wherein the weight ratio of the saccharide to the hydroxyethyl starch is (1:1) - (2: 1).
8. The method according to claim 7, wherein the buffer solution of step 1) has a pH value of 7.0 to 8.5; and/or the presence of a gas in the gas,
the buffer solution in the step 1) is any one of phosphate buffer solution, disodium hydrogen phosphate-sodium citrate buffer solution and dipotassium hydrogen phosphate-sodium hydroxide buffer solution; and/or the presence of a gas in the gas,
the particle size of the policosanol in the step 1) is 1-10 mu m; and/or the presence of a gas in the gas,
step 3), the saccharide is trehalose; and/or the presence of a gas in the gas,
the dosage of the freeze-drying protective agent in the step 3) is 1mg/mL-30 mg/mL; and/or the presence of a gas in the gas,
step 3) the freeze-drying method comprises: feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition board to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 Pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 Pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 Pa; the vacuum degree of the front box in the whole drying process is not higher than 30 Pa.
9. The preparation method according to claim 8, wherein the particle size of the policosanol in the step 1) is 3-4 μm.
10. The method of any one of claims 7 to 9, comprising the steps of:
1) dispersing policosanol and phospholipid in a buffer solution according to a ratio, and stirring for 20-90 minutes at the temperature of 1-40 ℃; the buffer solution is any one of phosphate buffer solution, disodium hydrogen phosphate-sodium citrate buffer solution and dipotassium hydrogen phosphate-sodium hydroxide buffer solution, and the pH value of the buffer solution is 7.0-8.5;
2) heating the suspension obtained in the step 1) to 85-99 ℃, homogenizing for 5-15 times under high pressure for 1-2 minutes each time, wherein the high-pressure homogenizing pressure is 2800-3200 psi; cooling the material to 1-10 ℃ after high-pressure homogenization;
3) adding a freeze-drying protective agent into the material obtained in the step 2), and freeze-drying to obtain the material;
the freeze-drying protective agent is saccharide and hydroxyethyl starch, wherein the weight ratio of the saccharide to the hydroxyethyl starch is (1:1) - (2: 1); the saccharide is trehalose; the dosage of the freeze-drying protective agent is 1mg/mL-30 mg/mL;
the freeze-drying method comprises the following steps: feeding into a drying oven at 5 deg.C, maintaining for 1 hr, cooling to-40 deg.C, pre-freezing for 10 hr, heating to-30 deg.C, maintaining for 8 hr, starting a vacuum pump, and pumping to 5-10 Pa; heating the clapboard to-10 ℃ for 2 hours, keeping for 6 hours, and pumping the vacuum degree of a drying box to 1-5 Pa; heating the partition board to 0 ℃ within 1 hour, maintaining the temperature for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 Pa; heating the partition plate to 10 ℃ within 1 hour, maintaining for 10 hours, and measuring the pressure rise until the pressure rise is less than 1 Pa; heating the partition plate to 25 ℃ within 0.5 hour, maintaining for 8 hours, and measuring the pressure rise until the pressure rise is less than 1 Pa; the vacuum degree of the front box in the whole drying process is not higher than 30 Pa.
11. Policosanol microspheres prepared by the process of any one of claims 8-10.
12. Use of the policosanol microspheres according to any one of claims 1-6 and 11 in the preparation of a medicament for the treatment of hyperlipidemia, hypercholesterolemia, atherosclerosis, and coronary heart disease.
13. The preparation containing the policosanol microspheres of any one of claims 1-6 and 11, wherein the preparation comprises tablets, granules, capsules and injections.
14. The preparation of claim 13, wherein the preparation is a tablet and comprises, by weight, 10-25 parts of the policosanol microspheres, 30-50 parts of lactose, 20-40 parts of pregelatinized starch, 5-15 parts of microcrystalline cellulose, 5-15 parts of hydroxypropyl cellulose and 0.5-2 parts of magnesium stearate.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201711421106.9A CN108042507B (en) | 2017-12-25 | 2017-12-25 | Policosanol microsphere and preparation method and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201711421106.9A CN108042507B (en) | 2017-12-25 | 2017-12-25 | Policosanol microsphere and preparation method and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN108042507A CN108042507A (en) | 2018-05-18 |
| CN108042507B true CN108042507B (en) | 2020-06-30 |
Family
ID=62131707
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201711421106.9A Active CN108042507B (en) | 2017-12-25 | 2017-12-25 | Policosanol microsphere and preparation method and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN108042507B (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN111214440B (en) * | 2018-11-26 | 2023-04-07 | 广州玻思韬控释药业有限公司 | Pharmaceutical composition for injection |
| CN111000811A (en) * | 2019-12-31 | 2020-04-14 | 济南大学 | Orally disintegrating tablet containing policosanol |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2488498A1 (en) * | 2002-06-10 | 2003-12-18 | Elan Pharma International Limited | Nanoparticulate polycosanol formulations and novel polycosanol combinations |
| CN101804029A (en) * | 2010-05-15 | 2010-08-18 | 王丽燕 | Atorvastatin liposome and preparation method thereof, and medicine composition containing atorvastatin |
| CN102292088A (en) * | 2009-02-23 | 2011-12-21 | 奈米瑞斯公司 | Policosanol nanoparticles |
-
2017
- 2017-12-25 CN CN201711421106.9A patent/CN108042507B/en active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CA2488498A1 (en) * | 2002-06-10 | 2003-12-18 | Elan Pharma International Limited | Nanoparticulate polycosanol formulations and novel polycosanol combinations |
| CN102292088A (en) * | 2009-02-23 | 2011-12-21 | 奈米瑞斯公司 | Policosanol nanoparticles |
| CN101804029A (en) * | 2010-05-15 | 2010-08-18 | 王丽燕 | Atorvastatin liposome and preparation method thereof, and medicine composition containing atorvastatin |
Also Published As
| Publication number | Publication date |
|---|---|
| CN108042507A (en) | 2018-05-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US7988994B2 (en) | Capsule formulation of pirfenidone and pharmaceutically acceptable excipients | |
| RU2248218C2 (en) | Solid lipid compositions | |
| TW200838527A (en) | Fumarate salt of (alpha S, beta R)-6-bromo-alpha-[2-(dimethylamino)ethyl]-2-methoxy-alpha-1-naphthalenyl-beta-phenyl-3-quinolineethanol | |
| TW201006466A (en) | Fenofibrate dosage forms | |
| CN108451979B (en) | Lycopene compound preparation with auxiliary treatment effect on prostate cancer and application thereof | |
| CN108042507B (en) | Policosanol microsphere and preparation method and application thereof | |
| CN113398098B (en) | Microcapsule, preparation method thereof and application of microcapsule in preventing and/or treating salivary gland injury caused by radiotherapy | |
| JPH0354926B2 (en) | ||
| JP2003520204A (en) | Cholesterol lowering stanol composition, preparation and use | |
| CN110559352A (en) | hawthorn flavone microcapsule and preparation method and application thereof | |
| CN114404362A (en) | Oral composition of gambogic acid and its application in the preparation of tumor therapeutic drugs | |
| CN110664785A (en) | Hawthorn flavone microcapsule and preparation method and application thereof | |
| EP4346773B1 (en) | A plurality of tasquinimod particles and use thereof | |
| JP3035328B2 (en) | Blood lipid lowering agent | |
| WO2021124316A1 (en) | High cbd cannabis sativa lines and extracts with anti-inflammatory potencies for oral and gut health | |
| JP2021176825A (en) | Solid preparation for oral intake containing curcumin | |
| AU2013201986B2 (en) | Capsule Formulation Of Pirfenidone And Pharmaceutically Acceptable Excipients | |
| JPS6053007B2 (en) | deworming preparation | |
| WO2025252965A1 (en) | Phragmalin-derived compounds for use in the treatment and/or prevention of a metabolic disease and/or disorder | |
| CN110051640A (en) | A kind of simvastatin capsules and preparation method thereof | |
| US3928602A (en) | Substituted phenoxymalonic acids, esters thereof and hypolipaemic agents containing the same for lowering lipid levels | |
| CN118845791A (en) | A crocetin preparation with high bioavailability and its application | |
| WO1997042945A1 (en) | Oral pharmaceutical compositions | |
| HK40107035A (en) | A plurality of tasquinimod particles and use thereof | |
| HK40107035B (en) | A plurality of tasquinimod particles and use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| CB03 | Change of inventor or designer information | ||
| CB03 | Change of inventor or designer information |
Inventor after: Bai Yunsheng Inventor after: Shi Qingdong Inventor after: Wang Jiao Inventor after: Chen Heyan Inventor after: Wang Zhengyuan Inventor before: Shi Qingdong Inventor before: Wang Jiao Inventor before: Chen Heyan Inventor before: Wang Zhengyuan |
|
| GR01 | Patent grant | ||
| GR01 | Patent grant |