CN106336413B - Compounds as JAK inhibitors and their uses - Google Patents
Compounds as JAK inhibitors and their uses Download PDFInfo
- Publication number
- CN106336413B CN106336413B CN201610536665.3A CN201610536665A CN106336413B CN 106336413 B CN106336413 B CN 106336413B CN 201610536665 A CN201610536665 A CN 201610536665A CN 106336413 B CN106336413 B CN 106336413B
- Authority
- CN
- China
- Prior art keywords
- formula
- compound
- mmol
- methyl
- pyrrolo
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/4353—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems
- A61K31/437—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom ortho- or peri-condensed with heterocyclic ring systems the heterocyclic ring system containing a five-membered ring having nitrogen as a ring hetero atom, e.g. indolizine, beta-carboline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/4985—Pyrazines or piperazines ortho- or peri-condensed with heterocyclic ring systems
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/519—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim ortho- or peri-condensed with heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/54—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one sulfur as the ring hetero atoms, e.g. sulthiame
- A61K31/541—Non-condensed thiazines containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K45/00—Medicinal preparations containing active ingredients not provided for in groups A61K31/00 - A61K41/00
- A61K45/06—Mixtures of active ingredients without chemical characterisation, e.g. antiphlogistics and cardiaca
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains three hetero rings
- C07D471/14—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains three hetero rings
- C07D487/14—Ortho-condensed systems
Landscapes
- Chemical & Material Sciences (AREA)
- Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
发明领域Field of Invention
本发明属于医药技术领域,具体涉及一类具有JAK抑制活性的化合物,包含本发明化合物的药物组合物,以及本发明的化合物或药学上包含本发明化合物的药物组合物在药物中的应用。The present invention belongs to the technical field of medicine, and specifically relates to a class of compounds with JAK inhibitory activity, a pharmaceutical composition comprising the compound of the present invention, and the application of the compound of the present invention or the pharmaceutical composition pharmaceutically comprising the compound of the present invention in medicine.
发明背景Background of the Invention
Janus激酶(JAK)属于酪氨酸激酶家族,由JAK1,JAK2,JAK3和TYK2组成。JAK在细胞因子信号传导中起着重要的作用。JAK家族激酶的下游底物包括转录信号传感与活化(STAT)蛋白。JAK/STAT信号发送已经在很多异常免疫应答的介导中有牵连,例如变态反应、哮喘、自身免疫疾病,例如移植排斥、类风湿性关节炎、肌萎缩性侧索硬化和多发性硬化,以及实体与血液恶性肿瘤,例如白血病和淋巴瘤。JAK1,JAK2和TYK2可以抑制多种基因表达,然而JAK3仅在粒细胞中发挥作用。细胞因子受体的典型功能是作为异二聚体形式存在,因此通常不是一种JAK激酶与细胞因子受体作用。Janus kinases (JAKs) belong to the tyrosine kinase family consisting of JAK1, JAK2, JAK3 and TYK2. JAKs play an important role in cytokine signaling. Downstream substrates of JAK family kinases include transcriptional signal sensing and activation (STAT) proteins. JAK/STAT signaling has been implicated in the mediation of many abnormal immune responses, such as allergy, asthma, autoimmune diseases such as transplant rejection, rheumatoid arthritis, amyotrophic lateral sclerosis and multiple sclerosis, and Solid and hematological malignancies such as leukemia and lymphoma. JAK1, JAK2 and TYK2 can inhibit the expression of various genes, whereas JAK3 only functions in granulocytes. Cytokine receptors typically function as heterodimers and are therefore not usually a JAK kinase interacting with cytokine receptors.
遗传生物学研究表明,JAK1通过与IFNalpha,IFNgamma,IL-2,IL-6等细胞因子受体作用而发挥作用,JAK1敲除小鼠由于LIF受体信号缺失而死亡。观察JAK1敲除小鼠的特征组织,发现JAK1在IFN,IL-10,IL-2/IL-4和IL-6等细胞通路中起重要作用。Genetic biology studies have shown that JAK1 acts by interacting with cytokine receptors such as IFNalpha, IFNgamma, IL-2, and IL-6, and JAK1 knockout mice die due to the lack of LIF receptor signaling. Observing the characteristic tissues of JAK1 knockout mice, it was found that JAK1 plays an important role in cellular pathways such as IFN, IL-10, IL-2/IL-4 and IL-6.
JAK2也已经在骨髓增殖性疾患中有牵连,这包括真性红细胞增多、特发性血小板增多、慢性自发性骨髓纤维化、伴有骨髓纤维化的骨髓性组织变形、慢性骨髓性白血病、慢性骨髓单核细胞性白细胞、慢性嗜酸细胞性白血病、嗜酸细胞过多综合征和系统性肥大细胞病。JAK2 has also been implicated in myeloproliferative disorders, including polycythemia vera, idiopathic thrombocythemia, chronic idiopathic myelofibrosis, myeloid tissue deformity with myelofibrosis, chronic myeloid leukemia, chronic myelogenous monocytogenes Nucleocytic leukocytes, chronic eosinophilic leukemia, hypereosinophilic syndrome, and systemic mastocytosis.
JAK3特异性的作用于γ细胞因子受体链,它在IL-2,IL-4,IL-7,IL-9,IL-15,IL-21等细胞因子受体中存在。JAK3在淋巴细胞生长,增生,变异过程中起到重要作用,发生异常可以导致严重的免疫缺失。JAK3在很多异常免疫应答的介导中有牵连,如变态反应,哮喘,自身免疫疾病如抑制移植排斥,类风湿性关节炎,肌肉缩性侧索硬化和多发性硬化以及实体和血液恶性肿瘤如白血病,淋巴瘤。JAK3抑制剂作为用于以下的免疫抑制剂是有用的治疗剂:器官移植、异种移植、狼疮、多发性硬化、类风湿性关节炎、银屑癣、I型糖尿病和来自糖尿病的并发症、癌症、哮喘、特应性皮炎、自身免疫甲状腺障碍、溃疡性结肠炎、克罗恩病、早老性痴呆、白血病和免疫抑制适宜的其他症状。还报道了JAK3的非造血性表达,尽管此功能上的意义还不清楚(J.Immunol.,2002,168:2475-2482)。JAK3 specifically acts on the γ-cytokine receptor chain, which exists in cytokine receptors such as IL-2, IL-4, IL-7, IL-9, IL-15, and IL-21. JAK3 plays an important role in the growth, proliferation and mutation of lymphocytes, and its abnormality can lead to severe immune deficiency. JAK3 has been implicated in the mediation of many abnormal immune responses, such as allergy, asthma, autoimmune diseases such as suppression of transplant rejection, rheumatoid arthritis, musculoskeletal lateral sclerosis and multiple sclerosis, as well as solid and hematological malignancies such as Leukemia, lymphoma. JAK3 inhibitors are useful therapeutics as immunosuppressants for organ transplantation, xenotransplantation, lupus, multiple sclerosis, rheumatoid arthritis, psoriasis, type I diabetes and complications from diabetes, cancer , asthma, atopic dermatitis, autoimmune thyroid disorders, ulcerative colitis, Crohn's disease, Alzheimer's disease, leukemia and other conditions where immunosuppression is appropriate. Non-hematopoietic expression of JAK3 has also been reported, although the functional significance of this is unclear (J. Immunol., 2002, 168:2475-2482).
TYK2作用于I型干扰素,IL-6,IL-10,IL-12,IL-23等细胞因子受体复合物。与之相一致的是,衍生于TYK2缺失的人的初级细胞,在I型干扰素,IL-6,IL-10,IL-12,IL-23的信号传导中存在障碍。TYK2 acts on type I interferon, IL-6, IL-10, IL-12, IL-23 and other cytokine receptor complexes. Consistently, primary cells derived from TYK2-deficient humans are impaired in the signaling of type I interferons, IL-6, IL-10, IL-12, and IL-23.
布鲁顿酪氨酸激酶(Bruton’s tyrosine kinase,BTK),一种非受体酪氨酸激酶Tec家族的成员,是在除了T淋巴细胞和自然杀伤细胞之外的所有造血细胞类型中表达的关键信号酶。BTK对变应性紊乱和/或自身免疫性疾病和/或炎性疾病的作用已经在BTK-缺失小鼠模型中得到证实。例如,在全身红斑狼疮(SLE)的标准鼠科动物前临床(preclinical)的模型中,已显示BTK缺失会明显改变疾病进程。Bruton's tyrosine kinase (BTK), a member of the Tec family of non-receptor tyrosine kinases, is critical for expression in all hematopoietic cell types except T lymphocytes and natural killer cells signaling enzymes. The effects of BTK on allergic disorders and/or autoimmune and/or inflammatory diseases have been demonstrated in BTK-null mouse models. For example, in a standard murine preclinical model of systemic lupus erythematosus (SLE), BTK deletion has been shown to significantly alter disease progression.
表皮生长因子受体(epidermal growth factor receptor,EGFR)是一种受体型酪氨酸激酶,广泛分布于人体各组织细胞膜上的多功能糖蛋白,是鸟类成红细胞白血病病毒(avian erythroblastic leukemia viral,v-erb-b)致癌基因同源体。EGFR等在上皮来源的肿瘤,如头颈部鳞状细胞癌、乳腺癌、直肠癌、卵巢癌、前列腺癌、非小细胞肺癌等多种肿瘤中都过表达,它们的表达与癌细胞增殖、转移等现象相关。Epidermal growth factor receptor (EGFR) is a receptor-type tyrosine kinase, a multifunctional glycoprotein widely distributed on the cell membrane of various tissues of the human body. , v-erb-b) oncogene homologues. EGFR and other tumors are overexpressed in epithelial-derived tumors, such as head and neck squamous cell carcinoma, breast cancer, rectal cancer, ovarian cancer, prostate cancer, non-small cell lung cancer and other tumors. related to transfer.
T790M突变是EGFR20外显因子中的一个点突变,是目前较为认可的耐药机制之一。T790M位于EGFR和ATP结合口袋的入口,其边链的大小直接影响EGFR和ATP的结合能力。T790M突变在空间上阻碍EGFR抑制剂与ATP结合位点的作用,增加EGFR对ATP的亲和力,从而使细胞对EGFR抑制剂产 生耐药。最初,T790M只在治疗失败的NSCLC患者标本中被发现,但随后在未经任何治疗的标本中也被发现,故目前认为,该突变也存在于未经TKI治疗的肿瘤组织中,但仅见于少数克隆细胞,由于这些克隆细胞对TKI的抵抗性而在治疗后被选择出来。T790M mutation is a point mutation in EGFR20 exon factor, which is one of the more recognized drug resistance mechanisms. T790M is located at the entrance of the EGFR and ATP binding pockets, and the size of its side chain directly affects the binding capacity of EGFR and ATP. The T790M mutation sterically hinders the action of EGFR inhibitors with the ATP binding site, increasing the affinity of EGFR for ATP, thereby making cells resistant to EGFR inhibitors. Initially, T790M was only found in specimens from patients with NSCLC who failed treatment, but was subsequently found in specimens without any treatment, so it is currently believed that this mutation is also present in TKI-untreated tumor tissues, but only in A small number of clones were selected after treatment due to their resistance to TKIs.
因此,提供治疗疾病(例如自身免疫性疾病、炎性疾病以及癌症)的抑制蛋白激酶的化合物存在需要。本发明的化合物能够有效抑制蛋白激酶的活性,如JAK1,JAK2,JAK3,BTK,EGFR或EGFR T790M。这类化合物将在治疗自体免疫疾病和/或炎性疾病和/或癌症中发挥潜在的作用。Accordingly, there exists a need to provide compounds that inhibit protein kinases for the treatment of diseases such as autoimmune diseases, inflammatory diseases, and cancer. The compounds of the present invention can effectively inhibit the activity of protein kinases, such as JAK1, JAK2, JAK3, BTK, EGFR or EGFR T790M. Such compounds would have potential roles in the treatment of autoimmune and/or inflammatory diseases and/or cancer.
发明内容SUMMARY OF THE INVENTION
本发明的化合物对蛋白激酶活性有抑制作用。更让人满意的是,本发明的化合物有多重的抑制功能,可以抑制JAK1,JAK2,JAK3,BTK,EGFR或EGFR T790M。特别地,本发明涉及的化合物及药学上可接受的药物组合物,都可以有效地作为JAK1,JAK2,JAK3,BTK,EGFR或EGFR T790M抑制剂。The compounds of the present invention have inhibitory effects on protein kinase activity. More satisfyingly, the compounds of the present invention have multiple inhibitory functions, and can inhibit JAK1, JAK2, JAK3, BTK, EGFR or EGFR T790M. In particular, the compounds and pharmaceutically acceptable pharmaceutical compositions of the present invention can effectively act as JAK1, JAK2, JAK3, BTK, EGFR or EGFR T790M inhibitors.
一方面,本发明涉及一种化合物,其为如式(I)所示的化合物或式(I)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In one aspect, the present invention relates to a compound, which is a compound represented by formula (I) or a stereoisomer, geometric isomer, tautomer, racemate of the compound represented by formula (I), Nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs:
其中,A环进一步独立任选地被1,2,3或4个R1取代;wherein the A ring is further independently optionally substituted with 1, 2, 3 or 4 R 1 ;
A环、E环、各R1、R2、R3、R5和m具有如本发明所述的含义。Ring A, ring E, each of R 1 , R 2 , R 3 , R 5 and m have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(Ia)所示的化合物或式(Ia)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In some embodiments, the present invention relates to a compound, which is a compound represented by formula (Ia) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (Ia) Forms, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs:
其中,A环进一步独立任选地被1,2,3或4个R1取代;wherein the A ring is further independently optionally substituted with 1, 2, 3 or 4 R 1 ;
A环、E环、各R1、R2、R3、各R4、R5和m具有如本发明所述的含义。Ring A, ring E, each of R 1 , R 2 , R 3 , each of R 4 , R 5 and m have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(Ib)所示的化合物或式(Ib)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In some embodiments, the present invention relates to a compound, which is a compound represented by formula (Ib) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (Ib) Forms, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs:
其中,A环进一步独立任选地被1,2,3或4个R1取代;wherein the A ring is further independently optionally substituted with 1, 2, 3 or 4 R 1 ;
A环、R5和各R1具有如本发明所述的含义。Ring A, R 5 and each R 1 have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(Ic)所示的化合物或式(Ic)所示化合物的立体异 构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In some embodiments, the present invention relates to a compound, which is a compound represented by formula (Ic) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (Ic) Forms, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs:
其中,A环进一步独立任选地被1,2,3或4个R1取代;wherein the A ring is further independently optionally substituted with 1, 2, 3 or 4 R 1 ;
A环、R5和各R1具有如本发明所述的含义。Ring A, R 5 and each R 1 have the meanings as described in the present invention.
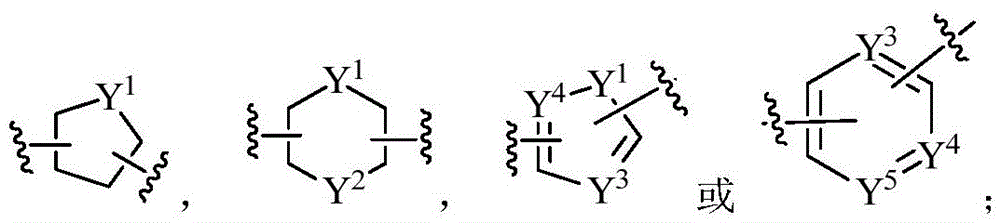
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,A环为C3-9环烷基,C5-9环烯基,C1-9杂环基,C6-10芳基,C1-9杂芳基,C6-12稠合双环基,C5-12稠合杂双环基,C6-12螺双环基或C5-12螺杂双环基;A环进一步独立任选地被1,2,3或4个R1取代;各R1具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) The above acceptable salts or prodrugs: wherein, ring A is C 3-9 cycloalkyl, C 5-9 cycloalkenyl, C 1-9 heterocyclyl, C 6-10 aryl, C 1-9 heterocyclyl Aryl, C 6-12 fused bicyclic group, C 5-12 fused heterobicyclic group, C 6-12 spirobicyclic group or C 5-12 spiro heterobicyclic group; A ring is further independently optionally separated by 1,2 , 3 or 4 R 1 substitutions; each R 1 has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,A环为 A环进一步独立任选地被1,2,3或4个R1取代;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein the A ring is Ring A is further independently optionally substituted with 1 , 2, 3 or 4 R1;
其中,X1和X2各自独立地为-C(R14R14a)-,-N(R15)-,-O-,-S(=O)p-或-C(=O)-;wherein, X 1 and X 2 are each independently -C(R 14 R 14a )-, -N(R 15 )-, -O-, -S(=O) p - or -C(=O)-;
X3,X4,X5和X6各自独立地为CR14或N;X 3 , X 4 , X 5 and X 6 are each independently CR 14 or N;
各R14和R14a独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基;each R 14 and R 14a is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl;
各R15独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基;each R 15 is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl;
各R1和p具有如本发明所述的含义。Each of R 1 and p has the meaning as described in the present invention.
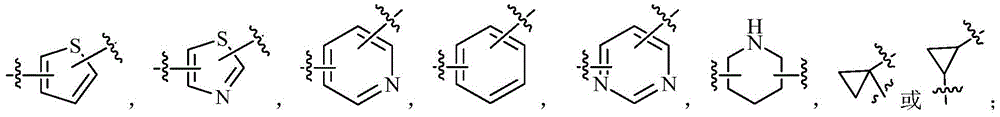
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,A环为 A环进一步独立任选地被1,2,3或4个R1取代;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein the A ring is Ring A is further independently optionally substituted with 1 , 2, 3 or 4 R1;
各R1具有如本发明所述的含义。Each R 1 has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,A环为 In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein the A ring is
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia))所示的化合物或式(I)(或式(Ia))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,E环为如下所示的子结构式: “*”表示与R5相连的一端;其中,当E环为时,A环为环丙基;In some embodiments, the present invention relates to a compound that is a compound of formula (I) (or formula (Ia)) or a stereoisomer of a compound of formula (I) (or formula (Ia)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein the E ring is the substructure shown below : "*" represents the end connected to R 5 ; wherein, when the E ring is When, A ring is cyclopropyl;
其中,R5、各R4和m具有如本发明所述的含义。Wherein, R 5 , each R 4 and m have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R1独立地为H,氘,F,Cl,Br,I,CN,烷基,烯基,炔基,卤代烷基,R-(CR6R7)n-O-,R-(CR6R7)n1-,R8O-(CR6R7)n-,R8-C(=O)-N(R9)-,CN-(CR6R7)n-R0-(CR6R7)n-N(R9)-,R9a-N(R9)-C(=O)-,R或R-N(R9)-;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R 1 is independently H, deuterium, F, Cl, Br, I, CN, alkyl, alkenyl, alkynyl, haloalkyl, R-(CR 6 R 7 ) n -O-, R-(CR 6 R 7 ) n1 -, R 8 O-(CR 6 R 7 ) n -, R 8 -C(=O)-N(R 9 )-, CN-(CR 6 R 7 ) n -R 0 -(CR 6 R 7 ) n -N(R 9 )-, R 9a -N(R 9 )-C(=O)-, R or RN(R 9 )-;
其中,各R、各R0、各R6、各R7、各R8、各R9、各R9a、各n和各n1具有如本发明所述的含义。Here, each R, each R 0 , each R 6 , each R 7 , each R 8 , each R 9 , each R 9a , each n and each n1 have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R1独立地为H,氘,F,Cl,Br,I,CN,C1-4烷基,C2-6烯基,C2-6炔基,C1-4卤代烷基,CN-(CR6R7)n-R0-(CR6R7)n-N(R9)-,R-(CR6R7)n-O-,R-(CR6R7)n1-,R8O-(CR6R7)n-,R8-C(=O)-N(R9)-,CN-(CR6R7)n-S(=O)p-R0-(CR6R7)n-N(R9)-,R9a-N(R9)-C(=O)-,R或R-N(R9)-;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R1 is independently H, deuterium, F , Cl, Br, I, CN, C1-4 alkyl, C2-6 alkenyl, C2-6 alkynyl , C 1-4 haloalkyl, CN-(CR 6 R 7 ) n -R 0 -(CR 6 R 7 ) n -N(R 9 )-, R-(CR 6 R 7 ) n -O-, R -(CR 6 R 7 ) n1 -, R 8 O-(CR 6 R 7 ) n -, R 8 -C(=O)-N(R 9 )-, CN-(CR 6 R 7 ) n -S (=O) p -R 0 -(CR 6 R 7 ) n -N(R 9 )-, R 9a -N(R 9 )-C(=O)-, R or RN(R 9 )-;
其中,各R、各R0、各R6、各R7、各R8、各R9、各R9a、各n和各n1具有如本发明所述的含义。Here, each R, each R 0 , each R 6 , each R 7 , each R 8 , each R 9 , each R 9a , each n and each n1 have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R1独立地为H,氘,F,Cl,Br,I,CN,甲基,乙基,正丙基,异丙基,三氟甲基,CN-(CR6R7)n-R0-(CR6R7)n-N(R9)-,R-(CR6R7)n-O-,R-(CR6R7)n1-,R8O-(CR6R7)n-,R8-C(=O)-N(R9)-,CN-(CR6R7)n-S(=O)p-R0-(CR6R7)n-N(R9)-,R9a-N(R9)-C(=O)-,R或R-N(R9)-;其中,各R0、各R、各R6、各R7、各R8、各R9、各R9a、各n和各n1具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R is independently H, deuterium, F , Cl, Br, I, CN, methyl, ethyl, n-propyl, isopropyl, trifluoromethyl, CN -(CR 6 R 7 ) n -R 0 -(CR 6 R 7 ) n -N(R 9 )-, R-(CR 6 R 7 ) n -O-, R-(CR 6 R 7 ) n1 - , R 8 O-(CR 6 R 7 ) n -, R 8 -C(=O)-N(R 9 )-, CN-(CR 6 R 7 ) n -S(=O) p -R 0 - (CR 6 R 7 ) n -N(R 9 )-, R 9a -N(R 9 )-C(=O)-, R or RN(R 9 )-; wherein each R 0 , each R, each R 6 , each R 7 , each R 8 , each R 9 , each R 9a , each n and each n1 have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R0独立地为C3-9亚环烷基,C5-9亚环烯基,C1-9亚杂环基,C6-10亚芳基,C1-9亚杂芳基,C6-12亚稠合双环基,C5-12亚稠合杂双环基,C6-12亚螺双环基或C5-12亚螺杂双环基;各R0独立任选地被1,2,3或4个R6a取代;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above : wherein each R is independently C 3-9 cycloalkylene, C 5-9 cycloalkenylene, C 1-9 heterocyclylene, C 6-10 arylene , C 1-9 heteroarylene, C 6-12 condensed bicyclic group, C 5-12 condensed heterobicyclic group, C 6-12 spiro bicyclic group or C 5-12 spiro heterobicyclic group; Each R 0 is independently optionally substituted with 1, 2, 3 or 4 R 6a ;
各R独立地为C3-9环烷基,C5-9环烯基,C1-9杂环基,C6-10芳基,C1-9杂芳基,C6-12稠合双环基,C5-12稠合杂双环基,C6-12螺双环基或C5-12螺杂双环基;各R独立任选地被1,2,3或4个R6a取代;其中,各R6a具有如本发明所述的含义。Each R is independently C 3-9 cycloalkyl, C 5-9 cycloalkenyl, C 1-9 heterocyclyl, C 6-10 aryl, C 1-9 heteroaryl, C 6-12 fused Bicyclyl, C5-12 fused heterobicyclyl, C6-12 spirobicyclyl or C5-12 spiroheterobicyclyl; each R is independently optionally substituted with 1, 2, 3 or 4 R6a ; wherein , each R 6a has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R0独立地为 各R0独立任选地被1,2,3或4个R6a取代;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above : wherein each R is independently Each R 0 is independently optionally substituted with 1, 2, 3 or 4 R 6a ;
各R独立地为各R独立任选地被1,2,3或4个R6a取代;Each R is independently Each R is independently optionally substituted with 1, 2, 3 or 4 R 6a ;
其中,Y1和Y2各自独立地为-C(R16R16a)-,-N(R17)-,-O-,-S(=O)p-或-C(=O)-;wherein, Y 1 and Y 2 are each independently -C(R 16 R 16a )-, -N(R 17 )-, -O-, -S(=O) p - or -C(=O)-;
Y3,Y4和Y5各自独立地为CR16或N;Y 3 , Y 4 and Y 5 are each independently CR 16 or N;
各R16和R16a独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基;Each R 16 and R 16a is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl;
各R17独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基;each R 17 is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl;
其中,各R6a具有如本发明所述的含义。Here, each R 6a has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R0独立地为 各R0独立任选地被1,2,3或4个R6a取代;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above : wherein each R is independently Each R 0 is independently optionally substituted with 1, 2, 3 or 4 R 6a ;
各R独立地为各R独立任选地被1,2,3或4个R6a取代;其中,各R6a具有如本发明所述的含义。Each R is independently Each R is independently optionally substituted with 1, 2, 3 or 4 R 6a ; wherein each R 6a has the meaning as described herein.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R6a独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基,F,Cl,Br,I,CN,R6b-(CR6R7)n-S(=O)p-,羟基,硝基,氨基,羧基或C1-4烷氧基;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R is independently hydrogen, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, F, Cl, Br, I, CN , R 6b -(CR 6 R 7 ) n -S(=O) p -, hydroxyl, nitro, amino, carboxyl or C 1-4 alkoxy;
其中,各R6、各R7、R6b、n和p具有如本发明所述的含义。Wherein, each R 6 , each R 7 , R 6b , n and p has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R6b独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基,F,Cl,Br,I,CN,羟基,硝基,C1-4卤代烷基,氨基,羧基或C1-4烷氧基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R is independently hydrogen, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, F, Cl, Br, I, CN , hydroxy, nitro, C 1-4 haloalkyl, amino, carboxyl or C 1-4 alkoxy.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R6独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基,F,Cl,Br,I,CN,羟基,硝基,C1-4卤代烷基,氨基,羧基或C1-4烷氧基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl , F, Cl, Br, I, CN , hydroxy, nitro, C 1-4 haloalkyl, amino, carboxyl or C 1-4 alkoxy.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R7独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基,F,Cl,Br,I,CN,羟基,硝基,C1-4卤代烷基,氨基,羧基或C1-4烷氧基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R is independently hydrogen, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, F, Cl, Br, I, CN , hydroxy, nitro, C 1-4 haloalkyl, amino, carboxyl or C 1-4 alkoxy.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R8独立地为氢,氘,C1-4烷基,C1-4卤代烷基或HO-(CR6R7)n-;其中,各R6、各R7和各n具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 8 is independently hydrogen, deuterium, C 1-4 alkyl, C 1-4 haloalkyl or HO-(CR 6 R 7 ) n -; wherein each R 6 , each R 7 and each n have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R9独立地为氢,氘,C1-4烷基,C1-4卤代烷基或HO-(CR6R7)n-;其中,各R6、各R7和各n具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 9 is independently hydrogen, deuterium, C 1-4 alkyl, C 1-4 haloalkyl or HO-(CR 6 R 7 ) n -; wherein each R 6 , each R 7 and each n have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R9a独立地为氢,氘,C1-4烷基,C1-4卤代烷基或HO-(CR6R7)n-;其中,各R6、各R7和各n具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 9a is independently hydrogen, deuterium, C 1-4 alkyl, C 1-4 haloalkyl or HO-(CR 6 R 7 ) n -; wherein each R 6 , each R 7 and each n have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia))所示的化合物或式(I)(或式(Ia))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中R2为H,氘,F,Cl,Br,I,CN,C1-4烷基,C2-6烯基,C2-6炔基,C1-4卤代烷基,R8O-(CR6R7)n-,R8-C(=O)-N(R9)-,R9a-N(R9)-C(=O)-或R9a-N(R9)-;其中,各R6、各R7、各R8、各R9、各R9a和各n具有如本发明所述的含义。In some embodiments, the present invention relates to a compound that is a compound of formula (I) (or formula (Ia)) or a stereoisomer of a compound of formula (I) (or formula (Ia)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs : where R is H, deuterium, F, Cl , Br, I, CN, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-4 haloalkyl, R 8 O-(CR 6 R 7 ) n -, R 8 -C(=O)-N(R 9 )-, R 9a -N(R 9 )-C(=O)- or R 9a -N(R 9 )-; wherein each R 6 , each R 7 , Each R 8 , each R 9 , each R 9a and each n has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia))所示的化合物或式(I)(或式(Ia))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中R3为H,氘,F,Cl,Br,I,CN,C1-4烷基,C2-6烯基,C2-6炔基,C1-4卤代烷基,R8O-(CR6R7)n-,R8-C(=O)-N(R9)-,R9a-N(R9)-C(=O)-或R9a-N(R9)-;其中,各R6、各R7、各R8、各R9、各R9a和各n具有如本发明所述的含义。In some embodiments, the present invention relates to a compound that is a compound of formula (I) (or formula (Ia)) or a stereoisomer of a compound of formula (I) (or formula (Ia)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein R is H, deuterium, F, Cl , Br, I, CN, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-4 haloalkyl, R 8 O-(CR 6 R 7 ) n -, R 8 -C(=O)-N(R 9 )-, R 9a -N(R 9 )-C(=O)- or R 9a -N(R 9 )-; wherein each R 6 , each R 7 , Each R 8 , each R 9 , each R 9a and each n has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia))所示的化合物或式(I)(或式(Ia))所 示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中R4为H,氘,F,Cl,Br,I,CN,C1-4烷基,C2-6烯基,C2-6炔基,C1-4卤代烷基,R8O-(CR6R7)n-,R8-C(=O)-N(R9)-,R9a-N(R9)-C(=O)-或R9a-N(R9)-;其中,各R6、各R7、各R8、各R9、各R9a和各n具有如本发明所述的含义。In some embodiments, the present invention relates to a compound that is a compound of formula (I) (or formula (Ia)) or a stereoisomer of a compound of formula (I) (or formula (Ia)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: where R is H, deuterium , F, Cl , Br, I, CN, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-4 haloalkyl, R 8 O-(CR 6 R 7 ) n -, R 8 -C(=O)-N(R 9 )-, R 9a -N(R 9 )-C(=O)- or R 9a -N(R 9 )-; wherein each R 6 , each R 7 , Each R 8 , each R 9 , each R 9a and each n has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中R5为CN-(CR10R11)n1-N(R12)-,CN-(CR10R11)n1-C(=O)-N(R12)-,CN-(CR10R11)n-C3-9环烷基-N(R12)-,R13-O-(CR10R11)n1-N(R12)-,CN-(CR10R11)n1-C1-9杂环基-C(=O)-N(R12)-,CN-(CR10R11)n-C(=O)-C1-9杂环基-(CR10R11)n-N(R12)-,R13-(CR10R11)n1-S(=O)p-(CR10R11)n-N(R12)-,R13-(CR10R11)n-S(=O)p-(R12a)N-C3-9环烷基-N(R12)-,R13-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-,CN-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-,CN-(CR10R11)n-C1-9杂环基-(CR10R11)n-N(R12)-,CN-(CR10R11)n1-S(=O)p-(R12a)N-,CN-(CR10R11)n-C(=O)-C1-9杂环基-(CR10R11)n-,CN-(CR10R11)n-C3-9环烷基-(CR10R11)n1-,HO-(CR10R11)n-C3-9环烷基-(CR10R11)n1-,CN-(CR10R11)n-C1-9杂环基-(CR10R11)n1-,CN-(CR10R11)n1-N(R12)-C(=O)-,CN-(CR10R11)n1-,C6-10芳基-(CR10R11)n1-,(R12a)N(R12)-C(=O)-(CR10R11)n1-,CN-(CR10R11)n-C1-9杂环基-(CR10R11)n-N(R12)-S(=O)p-,CN-(CR10R11)n-C1-9杂环基-(CR10R11)n-S(=O)p-,CN-(CR10R11)n-S(=O)p-,R13-(CR10R11)n-(R12a)N-S(=O)p-,R13-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-S(=O)p-或CN-(CR10R11)n1-(R12a)N-S(=O)p-;其中所述的芳基,杂环基和环烷基独立任选地被1,2,3或4个R6c取代;其中,各R6c、各R10、各R11、各R12、各R12a、各R13、各n、各n1和各p具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) The above acceptable salts or prodrugs: wherein R 5 is CN-(CR 10 R 11 ) n1 -N(R 12 )-, CN-(CR 10 R 11 ) n1 -C(=O)-N(R 12 )-, CN-(CR 10 R 11 ) n -C 3-9 cycloalkyl-N(R 12 )-, R 13 -O-(CR 10 R 11 ) n1 -N(R 12 )-, CN- (CR 10 R 11 ) n1 -C 1-9 heterocyclyl-C(=O)-N(R 12 )-, CN-(CR 10 R 11 ) n -C(=O)-C 1-9 heterocycle Cyclic group -(CR 10 R 11 ) n -N(R 12 )-, R 13 -(CR 10 R 11 ) n1 -S(=O) p -(CR 10 R 11 ) n -N(R 12 )- , R 13 -(CR 10 R 11 ) n -S(=O) p -(R 12a )NC 3-9 cycloalkyl-N(R 12 )-, R 13 -(CR 10 R 11 ) n -N (R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )N-, CN-(CR 10 R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )N-, CN-(CR 10 R 11 ) n -C 1-9 heterocyclyl-(CR 10 R 11 ) n -N(R 12 )-, CN-(CR 10 R 11 ) n1 -S(=O) p -(R 12a )N-, CN-(CR 10 R 11 ) n -C(=O)-C 1-9 heterocyclyl-(CR 10 R 11 ) n -, CN-(CR 10 R 11 ) n -C 3-9 cycloalkyl-(CR 10 R 11 ) n1 -, HO-(CR 10 R 11 ) n -C 3-9 cycloalkyl-(CR 10 R 11 ) n1 -, CN-(CR 10 R 11 ) n -C 1-9 heterocyclyl-(CR 10 R 11 ) n1 -, CN-(CR 10 R 11 ) n1 -N(R 12 )- C(=O)-, CN-(CR 10 R 11 ) n1 -, C 6-1 0 aryl-(CR 10 R 11 ) n1 -, (R 12a )N(R 12 )-C(=O)-(CR 10 R 11 ) n1 -, CN-(CR 10 R 11 ) n -C 1 -9 heterocyclyl-(CR 10 R 11 ) n -N(R 12 )-S(=O) p -, CN-(CR 10 R 11 ) n -C 1-9 heterocyclyl-(CR 10 R 11 ) n -S(=O) p -, CN-(CR 10 R 11 ) n -S(=O) p -, R 13 -(CR 10 R 11 ) n -(R 12a )NS(=O) p -, R 13 -(CR 10 R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )NS(=O) p - or CN-(CR 10 R 11 ) n1 -(R 12a )NS(=O) p -; wherein said aryl, heterocyclyl and cycloalkyl are independently optionally substituted with 1, 2, 3 or 4 R 6c ; wherein , each R 6c , each R 10 , each R 11 , each R 12 , each R 12a , each R 13 , each n, each n1 and each p have the meanings described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中R5为CN-(CR10R11)n1-N(R12)-,CN-(CR10R11)n1-C(=O)-N(R12)-,CN-(CR10R11)n-C3-6环烷基-N(R12)-,R13-O-(CR10R11)n1-N(R12)-,CN-(CR10R11)n1-C3-6杂环基-C(=O)-N(R12)-,CN-(CR10R11)n-C(=O)-C3-6杂环基-(CR10R11)n-N(R12)-,R13-(CR10R11)n-S(=O)p-(R12a)N-C3-6环烷基-N(R12)-,R13-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-,CN-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-,R13-(CR10R11)n1-S(=O)p-(CR10R11)n-N(R12)-,CN-(CR10R11)n-C3-6杂环基-(CR10R11)n-N(R12)-,CN-(CR10R11)n1-S(=O)p-(R12a)N-,CN-(CR10R11)n-C(=O)-C3-6杂环基-(CR10R11)n-,CN-(CR10R11)n-C3-6环烷基-(CR10R11)n1-,HO-(CR10R11)n-C3-6环烷基-(CR10R11)n1-,CN-(CR10R11)n-C3-6杂环基-(CR10R11)n1-,CN-(CR10R11)n1-,苯基-(CR10R11)n1-,(R12a)N(R12)-C(=O)-(CR10R11)n1-,CN-(CR10R11)n1-N(R12)-C(=O)-,CN-(CR10R11)n-C3-6杂环基-(CR10R11)n-S(=O)p-,CN-(CR10R11)n-S(=O)p-,CN-(CR10R11)n-C3-6杂环基-(CR10R11)n-N(R12)-S(=O)p-,R13-(CR10R11)n-(R12a)N-S(=O)p-,R13-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-S(=O)p-或CN-(CR10R11)n1-(R12a)N-S(=O)p-;其中所述的苯基,杂环基和环烷基独立任选地被1,2,3或4个R6c取代;其中,各R6c、各R10、各R11、各R12、各R12a、各R13、各n、各n1和各p具有如本发明所述的含义。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) The above acceptable salts or prodrugs: wherein R 5 is CN-(CR 10 R 11 ) n1 -N(R 12 )-, CN-(CR 10 R 11 ) n1 -C(=O)-N(R 12 )-, CN-(CR 10 R 11 ) n -C 3-6 cycloalkyl-N(R 12 )-, R 13 -O-(CR 10 R 11 ) n1 -N(R 12 )-, CN- (CR 10 R 11 ) n1 -C 3-6 heterocyclyl-C(=O)-N(R 12 )-, CN-(CR 10 R 11 ) n -C(=O)-C 3-6 heterocycle Cyclo-(CR 10 R 11 ) n -N(R 12 )-, R 13 -(CR 10 R 11 ) n -S(=O) p -(R 12a )NC 3-6 cycloalkyl-N( R 12 )-, R 13 -(CR 10 R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )N-, CN-(CR 10 R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )N-, R 13 -(CR 10 R 11 ) n1 -S(=O) p -(CR 10 R 11 ) n -N(R 12 )-, CN-(CR 10 R 11 ) n -C 3-6 heterocyclyl-(CR 10 R 11 ) n -N(R 12 )-, CN-(CR 10 R 11 ) n1 -S(=O) p -(R 12a )N-, CN-(CR 10 R 11 ) n -C(=O)-C 3-6 heterocyclyl-(CR 10 R 11 ) n -, CN-(CR 10 R 11 ) n -C 3-6 cycloalkyl-(CR 10 R 11 ) n1 -, HO-(CR 10 R 11 ) n -C 3-6 cycloalkyl-(CR 10 R 11 ) n1 -, CN-(CR 10 R 11 ) n -C 3-6 heterocyclyl-(CR 10 R 11 ) n1 -, CN-(CR 10 R 11 ) n1 -, phenyl-(CR 10 R 11 ) n1 -, (R 12a )N(R 12 )-C(= O)-(CR 10 R 11 ) n1 -, CN-(CR 10 R 11 ) n1 -N(R 12 )-C(=O)-, CN-(CR 10 R 11 ) n -C 3-6 hetero Cyclyl-(CR 10 R 11 ) n -S(=O) p -, CN-(CR 10 R 11 ) n -S(=O) p -, CN-(CR 10 R 11 ) n -C 3- 6heterocyclyl- (CR 10 R 11 ) n -N(R 12 )-S(=O) p -, R 13 -(CR 10 R 11 ) n -(R 12a )NS(=O) p -, R 13 -(CR 10 R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )NS(=O) p - or CN-(CR 10 R 11 ) n1 -(R 12a )NS(=O) p -; wherein said phenyl, heterocyclyl and cycloalkyl are independently optionally substituted with 1, 2, 3 or 4 R 6c ; wherein each R 6c , each R 10 , each R 11 , each R 12 , each R 12a , each R 13 , each n, each n1 and each p have the meanings as defined in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中R5为 In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein R 5 is
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R10独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 10 is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R10独立地为氢,氘,甲基,乙基,正丙基,异丙基,2,2,2-三氟乙基或三氟甲基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 10 is independently hydrogen, deuterium, methyl, ethyl, n-propyl, isopropyl, 2,2,2-trifluoroethyl or trifluoromethyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R11独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 11 is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R11独立地为氢,氘,甲基,乙基,正丙基,异丙基,2,2,2-三氟乙基或三氟甲基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 11 is independently hydrogen, deuterium, methyl, ethyl, n-propyl, isopropyl, 2,2,2-trifluoroethyl or trifluoromethyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R13独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 13 is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R13独立地为氢,氘,甲基,乙基,正丙基,异丙基,2,2,2-三氟乙基或三氟甲基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 13 is independently hydrogen, deuterium, methyl, ethyl, n-propyl, isopropyl, 2,2,2-trifluoroethyl or trifluoromethyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R12独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 12 is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R12独立地为氢,氘,甲基,正丙基,乙基或异丙基;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R is independently hydrogen, deuterium, methyl, n-propyl, ethyl or isopropyl;
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R12a独立地为氢,氘,C1-4烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R 12a is independently hydrogen, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R12a独立地为氢,氘,甲基,正丙基,乙基或异丙基;In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each R is independently hydrogen, deuterium , methyl, n-propyl, ethyl or isopropyl;
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R6c独立地为氢,氘,C1-4烷基,F,Cl,Br,I,CN,羟基,硝基,氨基,羧基或C1-4烷氧基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R6c is independently hydrogen, deuterium, C1-4 alkyl, F, Cl, Br, I, CN, hydroxy, nitro, amino, carboxyl or C1-4 alkoxy.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各R6c独立地为氢,氘,甲氧基,F,Cl,Br,I,CN,羟基,硝基,氨基,甲基,正丙基,乙基或异丙基。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) Acceptable salts or prodrugs of the above: wherein each R is independently hydrogen, deuterium , methoxy, F, Cl, Br, I, CN, hydroxy, nitro, amino, methyl, n-propyl, ethyl or isopropyl.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各n独立地为0,1,2或3。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each n is independently 0, 1, 2 or 3.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式 (I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各n1独立地为1,2,3或4。In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each n1 is independently 1, 2, 3 or 4.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia))所示的化合物或式(I)(或式(Ia))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中m为0,1,2,3或4。In some embodiments, the present invention relates to a compound that is a compound of formula (I) (or formula (Ia)) or a stereoisomer of a compound of formula (I) (or formula (Ia)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: where m is 0, 1, 2, 3 or 4.
一些实施例中,本发明涉及一种化合物,其为如式(I)(或式(Ia)、式(Ib)或式(Ic))所示的化合物或式(I)(或式(Ia)、式(Ib)或式(Ic))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中各p独立地为0,1或2In some embodiments, the present invention relates to a compound, which is a compound of formula (I) (or formula (Ia), formula (Ib), or formula (Ic)) or formula (I) (or formula (Ia) ), stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceuticals of compounds represented by formula (Ib) or formula (Ic)) An acceptable salt or prodrug of the above: wherein each p is independently 0, 1 or 2
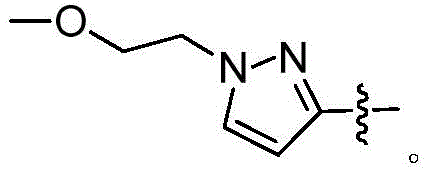
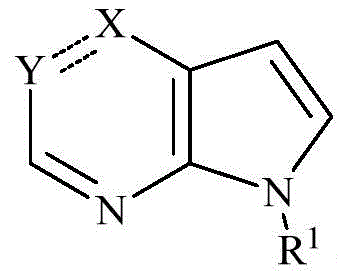
一方面,本发明涉及一种化合物,其为如式(II)所示的化合物或式(II)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In one aspect, the present invention relates to a compound, which is a compound represented by formula (II) or a stereoisomer, geometric isomer, tautomer, racemate of the compound represented by formula (II), Nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs:
其中,W环任选地进一步被1、2、3或4个独立地选自氟,氯,溴,C1-4卤代烷基,硝基,氰基,HO-(CR3R4)m-,H-(CR3R4)m-O-,R-N(R5)-C(=O)-,R-C(=O)-N(R5)-,R-N(R5)-,R-C(=O)-,R-C(=O)O-,R-OC(=O)-,R-S(=O)2-,R-S(=O)-,R-S-,R-N(R5)-S(=O)2-,R-S(=O)2-N(R5)-,R-N(R5)-C(=O)-N(R6)-或R的取代基所取代;wherein the W ring is optionally further selected by 1, 2, 3 or 4 independently selected from fluoro, chloro, bromo, C 1-4 haloalkyl, nitro, cyano, HO-(CR 3 R 4 ) m - , H-(CR 3 R 4 ) m -O-, RN(R 5 )-C(=O)-, RC(=O)-N(R 5 )-, RN(R 5 )-, RC(= O)-, RC(=O)O-, R-OC(=O)-, RS(=O) 2 -, RS(=O)-, RS-, RN(R 5 )-S(=O) 2- , RS(=O) 2 -N(R 5 )-, RN(R 5 )-C(=O)-N(R 6 )- or R’s substituent;
R、R3、R4、R5、R6、m、W环、R0、R00、L、Z1、Z2和n具有如本发明所述的含义。R, R 3 , R 4 , R 5 , R 6 , m, W ring, R 0 , R 00 , L, Z 1 , Z 2 and n have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(IIa)所示的化合物或式(IIa)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In some embodiments, the present invention relates to a compound, which is a compound represented by formula (IIa) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (IIa) Forms, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs:
其中,任选地进一步被1、2、3或4个独立地选自氟,氯,溴,C1-4卤代烷基,硝基,氰基,HO-(CR3R4)m-,H-(CR3R4)m-O-,R-N(R5)-C(=O)-,R-C(=O)-N(R5)-,R-N(R5)-,R-C(=O)-,R-C(=O)O-,R-OC(=O)-,R-S(=O)2-,R-S(=O)-,R-S-,R-N(R5)-S(=O)2-,R-S(=O)2-N(R5)-,R-N(R5)-C(=O)-N(R6)-或R的取代基所取代;表示单键或双键;in, optionally further selected by 1, 2, 3 or 4 independently selected from fluoro, chloro, bromo, C 1-4 haloalkyl, nitro, cyano, HO-(CR 3 R 4 ) m -, H-( CR 3 R 4 ) m -O-, RN(R 5 )-C(=O)-, RC(=O)-N(R 5 )-, RN(R 5 )-, RC(=O)-, RC(=O)O-, R-OC(=O)-, RS(=O) 2- , RS(=O) - , RS-, RN(R5)-S(=O) 2- , RS (=O) 2 -N(R 5 )-, RN(R 5 )-C(=O)-N(R 6 )- or the substituent of R; Indicates a single bond or a double bond;
R、R3、R4、R5、R6、m、X、Y、R1、R0、R00、L、Z1、Z2和n具有如本发明所述的含义。R, R 3 , R 4 , R 5 , R 6 , m, X, Y, R 1 , R 0 , R 00 , L, Z 1 , Z 2 and n have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,Z1和Z2各自独立地为N或CH。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein Z 1 and Z 2 are each independently N or CH.
一些实施例中,本发明涉及一种化合物,其为如式(II)所示的化合物或式(II)所示化合物的立体异 构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,W环为如下所示的环:W环任选地进一步被1、2、3或4个独立地选自氟,氯,溴,C1-4卤代烷基,硝基,氰基,HO-(CR3R4)m-,H-(CR3R4)m-O-,R-N(R5)-C(=O)-,R-C(=O)-N(R5)-,R-N(R5)-,R-C(=O)-,R-C(=O)O-,R-OC(=O)-,R-S(=O)2-,R-S(=O)-,R-S-,R-N(R5)-S(=O)2-,R-S(=O)2-N(R5)-,R-N(R5)-C(=O)-N(R6)-或R的取代基所取代;表示单键或双键;In some embodiments, the present invention relates to a compound, which is a compound represented by formula (II) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (II) Forms, nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs: wherein the W ring is a ring as shown below: The W ring is optionally further 1, 2, 3 or 4 independently selected from fluoro, chloro, bromo, C 1-4 haloalkyl, nitro, cyano, HO-(CR 3 R 4 ) m -, H -(CR 3 R 4 ) m -O-, RN(R 5 )-C(=O)-, RC(=O)-N(R 5 )-, RN(R 5 )-, RC(=O) -, RC(=O)O-, R-OC(=O)-, RS(=O) 2- , RS(=O) - , RS-, RN(R5)-S(=O) 2- , RS(=O) 2 -N(R 5 )-, RN(R 5 )-C(=O)-N(R 6 )- or R's substituent; Indicates a single bond or a double bond;
其中R、R3、R4、R5、R6、m、X、Y、R1、R0、R00、Z1、Z2和n具有如本发明所述的含义。wherein R, R 3 , R 4 , R 5 , R 6 , m, X, Y, R 1 , R 0 , R 00 , Z 1 , Z 2 and n have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,X为N,NRa或CRb;Y为CRc;In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: where X is N, NR a or CR b ; Y is CR c ;
Ra、Rb和Rc各自独立地为氢或C1-4烷基;Ra和Rc与他们所连接的环原子一起形成五元或六元的杂芳基;或Rb和Rc与他们所连接的环原子一起形成五元或六元的杂芳基;所述杂芳基可独立任选地被R2取代;R a , R b and R c are each independently hydrogen or C 1-4 alkyl; R a and R c together with the ring atoms to which they are attached form a five- or six-membered heteroaryl group; or R b and R c together with the ring atoms to which they are attached form a five- or six-membered heteroaryl; the heteroaryls may independently be optionally substituted with R ;
其中R2具有如本发明所述的含义。wherein R 2 has the meaning as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(II)所示的化合物或式(II)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,W环为如下所示的子结构式: In some embodiments, the present invention relates to a compound, which is a compound represented by formula (II) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (II) Forms, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein the W ring is the substructure shown below:
W环任选地进一步被1、2、3或4个独立地选自氟,氯,溴,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,硝基,氰基,HO-(CR3R4)m-,H-(CR3R4)m-O-,R-N(R5)-C(=O)-,R-C(=O)-N(R5)-,R-N(R5)-,R-C(=O)-,R-C(=O)O-,R-OC(=O)-,R-S(=O)2-,R-S(=O)-,R-S-,R-N(R5)-S(=O)2-,R-S(=O)2-N(R5)-,R-N(R5)-C(=O)-N(R6)-或R的取代基所取代;The W ring is optionally further 1, 2, 3 or 4 independently selected from fluoro, chloro, bromo, trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, Nitro, cyano, HO-(CR 3 R 4 ) m -, H-(CR 3 R 4 ) m -O-, RN(R 5 )-C(=O)-, RC(=O)-N (R 5 )-, RN(R 5 )-, RC(=O)-, RC(=O)O-, R-OC(=O)-, RS(=O) 2 -, RS(=O) -, RS-, RN(R 5 )-S(=O) 2 -, RS(=O) 2 -N(R 5 )-, RN(R 5 )-C(=O)-N(R 6 ) - or R substituents;
其中R、R3、R4、R5、R6、m、R1和R2具有如本发明所述的含义。wherein R, R 3 , R 4 , R 5 , R 6 , m, R 1 and R 2 have the meanings as described in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R1为H,氘,C1-4烷基,C2-6烯基,C2-6炔基或羟基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein R 1 is H, deuterium, C 1 -4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or hydroxy.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R1为H,氘,甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein R 1 is H, deuterium, methyl , ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa)) 所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R2为H,氘,C1-4烷基,C2-6烯基,C2-6炔基或羟基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein R 2 is H, deuterium, C 1 -4 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or hydroxy.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R2为H,氘,甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs : wherein R is H, deuterium, methyl , ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R0为氟,氯,溴,C1-4卤代烷基,硝基,氰基,HO-(CR3R4)m-,H-(CR3R4)m-O-,R-N(R5)-C(=O)-,R-C(=O)-N(R5)-,R-N(R5)-,R-C(=O)-,R-C(=O)O-,R-OC(=O)-,R-S(=O)2-,R-S(=O)-,R-S-,R-N(R5)-S(=O)2-,R-S(=O)2-N(R5)-,R-N(R5)-C(=O)-N(R6)-或R;In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs: wherein, R 0 is fluorine, chlorine, bromine, C 1-4 haloalkyl, nitro, cyano, HO-(CR 3 R 4 ) m -, H-(CR 3 R 4 ) m -O-, RN(R 5 )-C(=O)-, RC(=O)-N(R 5 )-, RN(R 5 )-, RC(=O)-, RC(=O)O-, R-OC(=O)-, RS(=O) 2 -, RS(=O)-, RS-, RN(R 5 )-S(=O) 2 -, RS(=O) 2 -N(R 5 )-, RN(R 5 )-C(=O )-N(R 6 )- or R;
其中R、R3、R4、R5、R6和m具有如本发明所述的含义。wherein R, R 3 , R 4 , R 5 , R 6 and m have the meanings as defined in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R0为氟,氯,溴,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,硝基,氰基,HO-(CR3R4)m-,H-(CR3R4)m-O-,R-N(R5)-C(=O)-,R-C(=O)-N(R5)-,R-N(R5)-,R-C(=O)-,R-C(=O)O-,R-OC(=O)-,R-S(=O)2-,R-S(=O)-,R-S-,R-N(R5)-S(=O)2-,R-S(=O)2-N(R5)-,R-N(R5)-C(=O)-N(R6)-或R;In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs: wherein, R 0 is fluorine, chlorine, bromine, Trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, nitro, cyano, HO-(CR 3 R 4 ) m -, H-(CR 3 R 4 ) m -O-, RN(R 5 )-C(=O)-, RC(=O)-N(R 5 )-, RN(R 5 )-, RC(=O)-, RC(=O) O-, R-OC(=O)-, RS(=O) 2 -, RS(=O)-, RS-, RN(R 5 )-S(=O) 2 -, RS(=O) 2 -N(R 5 )-, RN(R 5 )-C(=O)-N(R 6 )- or R;
其中R、R3、R4、R5、R6和m具有如本发明所述的含义。wherein R, R 3 , R 4 , R 5 , R 6 and m have the meanings as defined in the present invention.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R00为氟,氯,溴,硝基,氰基,HO-,H-S-,H2N-C(=O)-,H-C(=O)-NH-,NH2-,H-C(=O)-,H-C(=O)O-,H-OC(=O)-,H-S(=O)4-,H-S(=O)-,H2N-S(=O)2-,H-S(=O)2-NH-,H2N-C(=O)-NH-,CH3O-,苯基,C1-4烷基,C2-6烯基,C1-4卤代烷基或C2-6炔基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs: wherein, R 00 is fluorine, chlorine, bromine, Nitro, cyano, HO-, HS-, H 2 NC(=O)-, HC(=O)-NH-, NH 2 -, HC(=O)-, HC(=O)O-, H -OC(=O)-, HS(=O) 4 -, HS(=O)-, H 2 NS(=O) 2 -, HS(=O) 2 -NH-, H 2 NC(=O) -NH-, CH 3 O-, phenyl, C 1-4 alkyl, C 2-6 alkenyl, C 1-4 haloalkyl or C 2-6 alkynyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,L为-NH-S(=O)2-或-NH-。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs: wherein L is -NH-S(=O ) 2 - or -NH-.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R独立地为H,氘,C1-4烷基,C2-6烯基,C1-4卤代烷基或C2-6炔基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium, C 1-4 alkyl, C 2-6 alkenyl, C 1-4 haloalkyl or C 2-6 alkynyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R独立地为H,氘,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium, Trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, methyl, ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R3独立地为H,氘,C1-4烷基,C2-6烯基,C1-4卤代烷基或C2-6炔基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 1-4 haloalkyl or C 2-6 alkynyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R3独立地为H,氘,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基, 甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, methyl, ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R4独立地为H,氘,C1-4烷基,C2-6烯基,C1-4卤代烷基或C2-6炔基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 1-4 haloalkyl or C 2-6 alkynyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R4独立地为H,氘,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, methyl, ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R5独立地为H,氘,C1-4烷基,C2-6烯基,C1-4卤代烷基或C2-6炔基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 1-4 haloalkyl or C 2-6 alkynyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R5独立地为H,氘,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, methyl, ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R6独立地为H,氘,C1-4烷基,C2-6烯基,C1-4卤代烷基或C2-6炔基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , C 1-4 alkyl, C 2-6 alkenyl, C 1-4 haloalkyl or C 2-6 alkynyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各R6独立地为H,氘,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,甲基,乙基,异丙基或正丙基。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each R is independently H, deuterium , trifluoromethyl, 2,2-difluoroethyl, 3,3,3-trifluoropropyl, methyl, ethyl, isopropyl or n-propyl.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,各n独立地为1,2,3或4。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: wherein each n is independently 1, 2, 3 or 4.
一些实施例中,本发明涉及一种化合物,其为如式(II)(或式(IIa))所示的化合物或式(II)(或式(IIa))所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,m为0,1,2,3或4。In some embodiments, the present invention relates to a compound that is a compound of formula (II) (or formula (IIa)) or a stereoisomer of a compound of formula (II) (or formula (IIa)) , geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, and pharmaceutically acceptable salts or prodrugs: where m is 0, 1, 2, 3 or 4.
一方面,本发明涉及一种化合物,其为如式(III)所示的化合物或式(III)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:In one aspect, the present invention relates to a compound, which is a compound represented by formula (III) or a stereoisomer, geometric isomer, tautomer, racemate of a compound represented by formula (III), Nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs:
V环为其中“*”表示与-NH-相连的一端;The V ring is Wherein "*" represents the end connected to -NH-;
q为1,2,3或4;q is 1, 2, 3 or 4;
p为1,2,3或4;p is 1, 2, 3 or 4;
R8为HO-(CR10R11)p-,H-(CR10R11)p-O-,R9-N(R12)-,R9-C(=O)O-或R9;R 8 is HO-(CR 10 R 11 ) p -, H-(CR 10 R 11 ) p -O-, R 9 -N(R 12 )-, R 9 -C(=O)O- or R 9 ;
R12为H,氘,C1-6烷基,C2-6烯基,C2-6炔基或C1-4卤代烷基;R 12 is H, deuterium, C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl or C 1-4 haloalkyl;
各R9、R11和R10独立地为H,氘,C1-6烷基,C2-6烯基,C2-6炔基,C1-4卤代烷基或C1-4烷氧基。Each of R 9 , R 11 and R 10 is independently H, deuterium, C 1-6 alkyl, C 2-6 alkenyl, C 2-6 alkynyl, C 1-4 haloalkyl or C 1-4 alkoxy base.
一些实施例中,本发明涉及一种化合物,其为如式(III)所示的化合物或式(III)所示化合物的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物以及药学上可接受的盐或前药:其中,R12为H,氘,三氟甲基,2,2-二氟乙基,3,3,3-三氟丙基,甲基,乙基,异丙基或正丙基;In some embodiments, the present invention relates to a compound, which is a compound represented by formula (III) or a stereoisomer, geometric isomer, tautomer, racemic of a compound represented by formula (III) Forms, nitrogen oxides, hydrates, solvates, metabolites and pharmaceutically acceptable salts or prodrugs: wherein, R 12 is H, deuterium, trifluoromethyl, 2,2-difluoroethyl, 3, 3,3-trifluoropropyl, methyl, ethyl, isopropyl or n-propyl;
各R9、R11和R10独立地为H,氘,甲基,乙基,正丙基,异丙基,叔丁基,2-甲基丙基,三氟甲基,甲氧基或乙氧基。Each of R 9 , R 11 and R 10 is independently H, deuterium, methyl, ethyl, n-propyl, isopropyl, tert-butyl, 2-methylpropyl, trifluoromethyl, methoxy or Ethoxy.
一种化合物,其为具有下列之一结构的化合物或具有下列之一结构化合物的立体异构体,几何异构体,互变异构体,氮氧化物,水合物,溶剂化物,代谢产物,药学上可接受的盐或前药:A compound that is a compound having one of the following structures or a stereoisomer, geometric isomer, tautomer, nitrogen oxide, hydrate, solvate, metabolite of a compound having one of the following structures, Pharmaceutically acceptable salts or prodrugs:
本发明一方面涉及药物组合物,包含本发明的化合物,或它们的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物,代谢前体,药学上可接受的盐或它们的前药,或任选的药学上可接受的载体,赋形剂,稀释剂,辅剂,媒介物,或它们的组合。One aspect of the present invention pertains to pharmaceutical compositions comprising a compound of the present invention, or their stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites , metabolic precursors, pharmaceutically acceptable salts or prodrugs thereof, or optional pharmaceutically acceptable carriers, excipients, diluents, adjuvants, vehicles, or combinations thereof.
其中一些实施例是,本发明所述的药物组合物进一步地包含附加治疗剂,其选自化疗剂或抗增殖剂,抗炎药,免疫调节剂或免疫抑制剂,神经营养因子,用于治疗心血管疾病的活性剂,用于治疗糖尿病的活性剂和用于治疗自体免疫疾病的活性剂。In some embodiments, the pharmaceutical composition of the present invention further comprises an additional therapeutic agent selected from chemotherapeutic agents or anti-proliferative agents, anti-inflammatory agents, immunomodulatory agents or immunosuppressive agents, neurotrophic factors for treatment Active agents for cardiovascular disease, active agents for the treatment of diabetes and active agents for the treatment of autoimmune diseases.
本发明另一方面涉及使用一种本发明的化合物或包含本发明的化合物的药物组合物来制备用于预防、处理或治疗患者自体免疫疾病或增殖性疾病,并减轻其严重程度的药品的用途。Another aspect of the present invention pertains to the use of a compound of the present invention or a pharmaceutical composition comprising a compound of the present invention for the manufacture of a medicament for the prevention, management or treatment of, and alleviating the severity of, autoimmune or proliferative disease in a patient .
其中一些实施例是,本发明所述的自体免疫疾病是狼疮,多发性硬化,肌肉缩性侧索硬化,类风湿性关节炎,银屑病,I型糖尿病,因器官移植导致的并发症,异物移植,糖尿病,癌症,哮喘,特应性皮炎,自身免疫性甲状腺病,溃疡性结肠炎,克罗恩病,阿尔茨海默病,白血病或淋巴瘤。In some embodiments, the autoimmune disease of the present invention is lupus, multiple sclerosis, musculoskeletal lateral sclerosis, rheumatoid arthritis, psoriasis, type I diabetes, complications due to organ transplantation, Xenotransplantation, diabetes, cancer, asthma, atopic dermatitis, autoimmune thyroid disease, ulcerative colitis, Crohn's disease, Alzheimer's disease, leukemia or lymphoma.
其中一些实施例是,本发明所述的增殖性疾病是转移癌,结肠癌,胃腺癌,膀胱癌,乳腺癌,肾癌,肝癌,肺癌,甲状腺癌,头颈癌,前列腺癌,胰腺癌,CNS(中枢神经系统)的癌症,恶性胶质瘤,骨髓增生病,动脉粥样硬化或肺纤维化。In some embodiments, the proliferative disease of the present invention is metastatic cancer, colon cancer, gastric adenocarcinoma, bladder cancer, breast cancer, kidney cancer, liver cancer, lung cancer, thyroid cancer, head and neck cancer, prostate cancer, pancreatic cancer, CNS cancer (central nervous system) cancer, malignant glioma, myeloproliferative disease, atherosclerosis or pulmonary fibrosis.
另一方面,本发明涉及本发明化合物或包含本发明化合物的药物组合物来制备用于抑制或调节蛋白激酶活性的药物的用途。In another aspect, the present invention relates to the use of a compound of the present invention or a pharmaceutical composition comprising the compound of the present invention for the manufacture of a medicament for inhibiting or modulating the activity of protein kinases.
其中一些实施例是,蛋白激酶为JAK1,JAK2,JAK3,BTK,EGFR或EGFRT790M。In some embodiments, the protein kinase is JAK1, JAK2, JAK3, BTK, EGFR or EGFRT790M.
一方面,本发明涉及制备式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所包含的化合物的中间体。In one aspect, the invention relates to intermediates for the preparation of compounds encompassed by formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III).
本发明另一方面涉及式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所包含的化合物的制备、分离和纯化的方法。Another aspect of the present invention relates to the preparation, isolation and purification of compounds encompassed by formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III) Methods.
本发明还包含本发明的化合物及其药学上可接受的盐的应用,用于生产医药产品治疗自体免疫疾病或增殖性疾病,包括那些本发明所描述的。The present invention also encompasses the use of the compounds of the present invention, and pharmaceutically acceptable salts thereof, for the manufacture of medicinal products for the treatment of autoimmune or proliferative diseases, including those described herein.
本发明包含药物组合物,该药物组合物包括式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所代表的化合物与至少一个药学上可接受的载体,辅剂或稀释剂的结合所需的有效治疗用量。The present invention includes a pharmaceutical composition comprising a compound represented by formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III) The therapeutically effective amount required in combination with at least one pharmaceutically acceptable carrier, adjuvant or diluent.
本发明同样包含治疗患者自体免疫疾病或增殖性疾病,或对此病症敏感的方法,该方法包含使用式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所代表化合物的治疗有效量对患者进行治疗。The present invention also includes a method of treating, or susceptibility to, an autoimmune or proliferative disease in a patient comprising the use of formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II) , a therapeutically effective amount of a compound represented by formula (IIa) or formula (III) to treat a patient.
除非其他方面表明,本发明的化合物所有的立体异构体,几何异构体,互变异构体,消旋体,氮氧化物,水合物,溶剂化物,代谢产物,药学上可接受的盐和前药都属于本发明的范围。Unless otherwise indicated, all stereoisomers, geometric isomers, tautomers, racemates, nitrogen oxides, hydrates, solvates, metabolites, pharmaceutically acceptable salts of the compounds of the present invention and prodrugs are within the scope of the present invention.
本发明描述的化合物可以具有一个或多个立构中心,并且每一个中心可以存在R或S构型。本发明提供的化合物包括所有非对映异构体、对映异构体和差向异构体的形式,及其合适的混合物。如果需要,立体异构体可以通过本领域已知的方法获得,例如通过手性色谱柱分离立体异构体。The compounds described herein may have one or more stereocenters, and each center may exist in the R or S configuration. The compounds provided herein include all diastereomeric, enantiomeric and epimeric forms, as well as suitable mixtures thereof. If desired, stereoisomers can be obtained by methods known in the art, such as separation of stereoisomers by chiral chromatography columns.
本发明的化合物的盐,还包括用于制备或纯化式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所示化合物的中间体或式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所示化合物分离的对映异构体的盐,但不一定是药学上可接受的盐。The salts of the compounds of the present invention also include those of formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III) used in the preparation or purification Intermediates of compounds or separated enantiomers of compounds of formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III) salts, but not necessarily pharmaceutically acceptable salts.
另外,本发明公开的化合物、包括它们的盐,也可以以它们的水合物形式或包含其溶剂(例如乙醇、DMSO,等等)的形式得到,用于它们的结晶。本发明公开的化合物可以与药学上可接受的溶剂(包括水)固有地或通过设计形成溶剂化物;因此,本发明旨在包括溶剂化的和未溶剂化的形式。In addition, the compounds disclosed herein, including their salts, can also be obtained in their hydrate form or in a form containing a solvent (eg, ethanol, DMSO, etc.) for their crystallization. The compounds disclosed herein may form solvates, inherently or by design, with pharmaceutically acceptable solvents, including water; thus, the present invention is intended to include both solvated and unsolvated forms.
本发明给出的任何结构式也意欲表示这些化合物未被同位素富集的形式以及同位素富集的形式。同位素富集的化合物具有本发明给出的通式描绘的结构,除了一个或多个原子被具有所选择原子量或质量数的 原子替换。可引入本发明化合物中的示例性同位素包括氢、碳、氮、氧、磷、硫、氟和氯的同位素,如2H, 3H,11C,13C,14C,15N,17O,18O,18F,31P,32P,35S,36Cl和125I。Any structural formula given herein is also intended to represent both isotopically unenriched as well as isotopically enriched forms of these compounds. Isotopically enriched compounds have the structures depicted by the general formulae given herein, except that one or more atoms are replaced by atoms having a selected atomic weight or mass number. Exemplary isotopes that can be incorporated into the compounds of the present invention include isotopes of hydrogen , carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, and chlorine, such as 2H, 3H , 11C , 13C , 14C , 15N , 17O , 18 O, 18 F, 31 P, 32 P, 35 S, 36 Cl and 125 I.
另一方面,本发明所述化合物包括同位素富集的本发明所定义的化合物,例如,其中存在放射性同位素,如3H,14C和18F的那些化合物,或者其中存在非放射性同位素,如2H和13C。该类同位素富集的化合物可用于代谢研究(使用14C)、反应动力学研究(使用例如2H或3H)、检测或成像技术,如正电子发射断层扫描术(PET)或包括药物或底物组织分布测定的单光子发射计算机断层成像术(SPECT),或可用于患者的放疗中。18F富集的化合物对PET或SPECT研究而言是特别理想的。同位素富集的式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所示化合物可以通过本领域技术人员熟悉的常规技术或本发明中的实施例和制备过程所描述使用合适的同位素标记试剂替代原来使用过的未标记试剂来制备。In another aspect, the compounds of the present invention include isotopically enriched compounds as defined by the present invention, for example, those in which radioactive isotopes, such as 3 H, 14 C and 18 F are present, or in which non-radioactive isotopes, such as 2 H and 13 C. Such isotopically enriched compounds can be used in metabolic studies (using 14 C), reaction kinetic studies (using eg 2 H or 3 H), detection or imaging techniques such as positron emission tomography (PET) or including drugs or Single photon emission computed tomography (SPECT) for substrate tissue distribution measurements, or may be used in radiation therapy of patients. 18 F-enriched compounds are particularly ideal for PET or SPECT studies. Isotopically enriched compounds of formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III) can be obtained by conventional methods familiar to those skilled in the art. The techniques or the examples and preparations described in this invention are prepared using a suitable isotopically-labeled reagent in place of the unlabeled reagent originally used.
此外,较重同位素特别是氘(即,2H或D)的取代可提供某些治疗优点,这些优点是由代谢稳定性更高带来的。例如,体内半衰期增加或剂量需求降低或治疗指数得到改善带来的。应当理解,本发明中的氘被看作式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所示化合物的取代基。可以用同位素富集因子来定义该类较重同位素特别是氘的浓度。本发明所使用的术语“同位素富集因子”是指所指定同位素的同位素丰度和天然丰度之间的比例。如果本发明化合物的取代基被指定为氘,该化合物对各指定的氘原子而言具有至少3500(各指定氘原子处52.5%的氘掺入)、至少4000(60%的氘掺入)、至少4500(67.5%的氘掺入),至少5000(75%的氘掺入),至少5500(82.5%的氘掺入)、至少6000(90%的氘掺入)、至少6333.3(95%的氘掺入)、至少6466.7(97%的氘掺入)、至少6600(99%的氘掺入)或至少6633.3(99.5%的氘掺入)的同位素富集因子。本发明可药用的溶剂化物包括其中结晶溶剂可以是同位素取代的例如D2O、丙酮-d6、DMSO-d6的那些溶剂化物。In addition, substitution of heavier isotopes, particularly deuterium (ie, 2 H or D), may provide certain therapeutic advantages that result from greater metabolic stability. For example, increased in vivo half-life or reduced dosage requirements or improved therapeutic index. It should be understood that deuterium in the present invention is regarded as the substitution of compounds of formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula (III) base. The concentration of such heavier isotopes, especially deuterium, can be defined by an isotopic enrichment factor. The term "isotopic enrichment factor" as used herein refers to the ratio between the isotopic abundance and the natural abundance of a given isotope. If a substituent of a compound of the present invention is designated as deuterium, the compound has for each designated deuterium atom at least 3500 (52.5% deuterium incorporation at each designated deuterium atom), at least 4000 (60% deuterium incorporation), At least 4500 (67.5% deuterium incorporated), at least 5000 (75% deuterium incorporated), at least 5500 (82.5% deuterium incorporated), at least 6000 (90% deuterium incorporated), at least 6333.3 (95% deuterium incorporated) deuterium incorporation), an isotopic enrichment factor of at least 6466.7 (97% deuterium incorporation), at least 6600 (99% deuterium incorporation), or at least 6633.3 (99.5% deuterium incorporation). Pharmaceutically acceptable solvates of the present invention include those in which the crystallization solvent may be isotopically substituted, eg, D2O , acetone - d6, DMSO -d6.
前面所述内容只概述了本发明的某些方面,但并不限于这些方面及其他的方面的内容将在下面作更加具体完整的描述。The foregoing has outlined only certain aspects of the present invention, but is not limited to these and other aspects which will be described in more detail below.
本发明详细说明书Detailed Description of the Invention
定义和一般术语Definitions and General Terms
现在详细描述本发明的某些实施方案,其实例由随附的结构式和化学式说明。本发明意图涵盖所有的替代、修改和等同技术方案,它们均包括在如权利要求定义的本发明范围内。本领域技术人员应认识到,许多与本发明所述类似或等同的方法和材料能够用于实践本发明。本发明绝不限于本发明所述的方法和材料。在所结合的文献、专利和类似材料的一篇或多篇与本申请不同或相矛盾的情况下(包括但不限于所定义的术语、术语应用、所描述的技术,等等),以本申请为准。Certain embodiments of the invention will now be described in detail, examples of which are illustrated by the accompanying structural and chemical formulae. The present invention is intended to cover all alternatives, modifications and equivalents, which are included within the scope of the present invention as defined by the claims. One skilled in the art will recognize that many methods and materials similar or equivalent to those described herein could be used in the practice of the present invention. The present invention is in no way limited to the methods and materials described herein. In the event that one or more of the incorporated literature, patents, and similar materials differs from or contradicts this application (including, but not limited to, terms defined, uses of terms, techniques described, etc.), this Application shall prevail.
应进一步认识到,本发明的某些特征,为清楚可见,在多个独立的实施方案中进行了描述,但也可以在单个实施例中以组合形式提供。反之,本发明的各种特征,为简洁起见,在单个实施方案中进行了描述,但也可以单独或以任意适合的子组合提供。It should further be appreciated that certain features of the invention, which are, for clarity, described in the context of multiple separate embodiments, can also be provided in combination in a single embodiment. Conversely, various features of the invention, which are, for brevity, described in the context of a single embodiment, can also be provided separately or in any suitable subcombination.
除非另外说明,本发明所使用的所有科技术语具有与本发明所属领域技术人员的通常理解相同的含义。本发明涉及的所有专利和公开出版物通过引用方式整体并入本发明。Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. All patents and publications referred to herein are incorporated by reference in their entirety.
除非另外说明,应当应用本发明所使用的下列定义。出于本发明的目的,化学元素与元素周期表CAS版,和《化学和物理手册》,第75版,1994一致。此外,有机化学一般原理可参考"Organic Chemistry",Thomas Sorrell,University Science Books,Sausalito:1999,和"March's Advanced Organic Chemistry"by Michael B.Smith and JerryMarch,John Wiley&Sons,New York:2007中的描述,其全部内容通过引用并入本发明。Unless otherwise stated, the following definitions used herein shall apply. For the purposes of the present invention, chemical elements are in accordance with the Periodic Table of the Elements, CAS Edition, and Handbook of Chemistry and Physics, 75th Edition, 1994. In addition, general principles of organic chemistry may be referred to as described in "Organic Chemistry", Thomas Sorrell, University Science Books, Sausalito: 1999, and "March's Advanced Organic Chemistry" by Michael B. Smith and Jerry March, John Wiley & Sons, New York: 2007, Its entire contents are incorporated herein by reference.
除非另有说明或者上下文中有明显的冲突,本文所使用的冠词“一”、“一个(种)”和“所述”旨在包括“至少一个”或“一个或多个”。因此,本文所使用的这些冠词是指一个或多于一个(即至少一个)宾语的冠词。例如,“一组分”指一个或多个组分,即可能有多于一个的组分被考虑在所述实施方案的实 施方式中采用或使用。As used herein, the articles "a", "an" and "the" are intended to include "at least one" or "one or more" unless stated otherwise or otherwise clearly contradicted by context. Thus, as used herein, these articles refer to one or more than one (ie, at least one) object of the article. For example, "a component" refers to one or more components, i.e. there may be more than one component contemplated for adoption or use in the implementation of the described embodiments.
术语“包含”为开放式表达,即包括本发明所指明的内容,但并不排除其他方面的内容。The term "comprising" is an open-ended expression, that is, it includes the contents specified in the present invention, but does not exclude other aspects.
“立体异构体”是指具有相同化学构造,但原子或基团在空间上排列方式不同的化合物。立体异构体包括对映异构体、非对映异构体、构象异构体(旋转异构体)、几何异构体(顺/反)异构体、阻转异构体,等等。"Stereoisomers" refer to compounds that have the same chemical structure, but differ in the arrangement of atoms or groups in space. Stereoisomers include enantiomers, diastereomers, conformational isomers (rotamers), geometric isomers (cis/trans), atropisomers, etc. .
“手性”是具有与其镜像不能重叠性质的分子;而“非手性”是指与其镜像可以重叠的分子。"Chiral" is a molecule that has the property of being non-superimposable with its mirror image; while "achiral" refers to a molecule that is superimposable with its mirror image.
“对映异构体”是指一个化合物的两个不能重叠但互成镜像关系的异构体。"Enantiomer" refers to two nonsuperimposable, but mirror-image isomers of a compound.
“非对映异构体”是指有两个或多个手性中心并且其分子不互为镜像的立体异构体。非对映异构体具有不同的物理性质,如熔点、沸点、光谱性质和反应性。非对映异构体混合物可通过高分辨分析操作如电泳和色谱,例如HPLC来分离。"Diastereomer" refers to a stereoisomer having two or more centers of chirality and whose molecules are not mirror images of each other. Diastereomers have different physical properties such as melting point, boiling point, spectral properties and reactivity. Diastereomeric mixtures can be separated by high resolution analytical procedures such as electrophoresis and chromatography, eg HPLC.
本发明所使用的立体化学定义和规则一般遵循S.P.Parker,Ed.,McGraw-HillDictionary of Chemical Terms(1984)McGraw-Hill Book Company,New York;andEliel,E.and Wilen,S.,“Stereochemistry of Organic Compounds”,John Wiley&Sons,Inc.,NewYork,1994。Stereochemical definitions and rules used in the present invention generally follow S.P. Parker, Ed., McGraw-Hill Dictionary of Chemical Terms (1984) McGraw-Hill Book Company, New York; and Eliel, E. and Wilen, S., "Stereochemistry of Organic Compounds", John Wiley & Sons, Inc., New York, 1994.
许多有机化合物以光学活性形式存在,即它们具有使平面偏振光的平面发生旋转的能力。在描述光学活性化合物时,使用前缀D和L或R和S来表示分子关于其一个或多个手性中心的绝对构型。前缀d和l或(+)和(-)是用于指定化合物所致平面偏振光旋转的符号,其中(-)或l表示化合物是左旋的。前缀为(+)或d的化合物是右旋的。一种具体的立体异构体是对映异构体,这种异构体的混合物称作对映异构体混合物。对映异构体的50:50混合物称为外消旋混合物或外消旋体,当在化学反应或过程中没有立体选择性或立体特异性时,可出现这种情况。Many organic compounds exist in optically active forms, that is, they have the ability to rotate the plane of plane-polarized light. In describing optically active compounds, the prefixes D and L or R and S are used to denote the absolute configuration of the molecule about one or more of its chiral centers. The prefixes d and 1 or (+) and (-) are symbols used to designate the rotation of plane polarized light by the compound, where (-) or 1 indicates that the compound is levorotatory. Compounds prefixed with (+) or d are dextrorotatory. A specific stereoisomer is an enantiomer, and a mixture of such isomers is called an enantiomeric mixture. A 50:50 mixture of enantiomers is called a racemic mixture or racemate, which can occur when there is no stereoselectivity or stereospecificity in a chemical reaction or process.
本发明公开化合物的任何不对称原子(例如,碳等)都可以以外消旋或对映体富集的形式存在,例如(R)-、(S)-或(R,S)-构型形式存在。在某些实施方案中,各不对称原子在(R)-或(S)-构型方面具有至少50%对映体过量,至少60%对映体过量,至少70%对映体过量,至少80%对映体过量,至少90%对映体过量,至少95%对映体过量,或至少99%对映体过量。Any asymmetric atom (eg, carbon, etc.) of the compounds disclosed herein may exist in racemic or enantiomerically enriched forms, such as (R)-, (S)- or (R,S)-configurations exist. In certain embodiments, each asymmetric atom has at least 50% enantiomeric excess, at least 60% enantiomeric excess, at least 70% enantiomeric excess, at least 70% enantiomeric excess in the (R)- or (S)-configuration 80% enantiomeric excess, at least 90% enantiomeric excess, at least 95% enantiomeric excess, or at least 99% enantiomeric excess.
依据起始物料和方法的选择,本发明化合物可以以可能的异构体中的一个或它们的混合物,例如外消旋体和非对应异构体混合物(这取决于不对称碳原子的数量)的形式存在。光学活性的(R)-或(S)-异构体可使用手性合成子或手性试剂制备,或使用常规技术拆分。如果化合物含有一个双键,取代基可能为E或Z构型;如果化合物中含有二取代的环烷基,环烷基的取代基可能有顺式或反式构型;如本发明实施例37存在吡咯烷基的顺势和反式构型。Depending on the choice of starting materials and process, the compounds of the present invention may be present as one of the possible isomers or as mixtures thereof, such as racemates and mixtures of diastereomers (depending on the number of asymmetric carbon atoms) form exists. Optically active (R)- or (S)-isomers can be prepared using chiral synthons or chiral reagents, or resolved using conventional techniques. If the compound contains a double bond, the substituent may have E or Z configuration; if the compound contains a disubstituted cycloalkyl, the substituent of the cycloalkyl may have cis or trans configuration; as in Example 37 of the present invention There are cis and trans configurations of pyrrolidinyl.
术语“互变异构体”或“互变异构形式”是指具有不同能量的可通过低能垒(lowenergy barrier)互相转化的结构异构体。若互变异构是可能的(如在溶液中),则可以达到互变异构体的化学平衡。例如,质子互变异构体(proton tautomer)(也称为质子转移互变异构体(prototropic tautomer))包括通过质子迁移来进行的互相转化,如酮-烯醇异构化和亚胺-烯胺异构化。价键互变异构体(valence tautomer)包括通过一些成键电子的重组来进行的互相转化。酮-烯醇互变异构的具体实例是戊烷-2,4-二酮和4-羟基戊-3-烯-2-酮互变异构体的互变。互变异构的另一个实例是酚-酮互变异构。酚-酮互变异构的一个具体实例是吡啶-4-醇和吡啶-4(1H)-酮互变异构体的互变。除非另外指出,本发明化合物的所有互变异构体形式都在本发明的范围之内。The term "tautomer" or "tautomeric form" refers to structural isomers of different energies that are interconvertible through a low energy barrier. A chemical equilibrium of tautomers can be achieved if tautomerism is possible (eg, in solution). For example, proton tautomers (also known as prototropic tautomers) include interconversions by migration of protons, such as keto-enol isomerization and imine- Enamine isomerization. Valence tautomers include interconversions by recombination of some of the bonding electrons. A specific example of keto-enol tautomerism is the interconversion of pentane-2,4-dione and 4-hydroxypent-3-en-2-one tautomers. Another example of tautomerism is phenol-ketone tautomerism. A specific example of phenol-ketone tautomerism is the interconversion of pyridin-4-ol and pyridin-4(lH)-one tautomers. Unless otherwise indicated, all tautomeric forms of the compounds of the present invention are within the scope of the present invention.
像本发明所描述的,本发明的化合物可以任选地被一个或多个取代基所取代,如上面的通式化合物,或者像实施例里面特殊的例子,子类,和本发明所包含的一类化合物。应了解“任选取代的”这个术语与“取代或非取代的”这个术语可以交换使用。一般而言,术语“取代的”表示所给结构中的一个或多个氢原子被具体取代基所取代。除非其他方面表明,一个任选的取代基团可以在基团各个可取代的位置进行取 代。当所给出的结构式中不只一个位置能被选自具体基团的一个或多个取代基所取代,那么取代基可以相同或不同地在各个位置取代。其中所述的取代基可以是,但并不限于,氘,氟,氯,溴,碘,氰基,羟基,硝基,氨基,羧基,烷基,烷氧基,芳氧基,杂芳基氧基,杂环基氧基,芳基烷氧基,杂芳基烷氧基,烷基酰基,杂烷基,环烷基,环烯基,环烷基烷基,杂环基,杂环基烷基,杂环基酰基,芳基,芳基烷基,芳氨基,杂芳基,杂芳基烷基,杂芳基氨基,酰胺基,磺酰基,氨基磺酰基等等。As described herein, the compounds of the present invention may be optionally substituted with one or more substituents, such as the compounds of the general formula above, or as in the Examples, subclasses, and subclasses encompassed by the present invention. a class of compounds. It is to be understood that the term "optionally substituted" is used interchangeably with the term "substituted or unsubstituted". In general, the term "substituted" means that one or more hydrogen atoms in a given structure have been replaced with a specified substituent. Unless otherwise indicated, an optional substituent group may be substituted at each substitutable position of the group. When more than one position in a given formula can be substituted with one or more substituents selected from a particular group, the substituents can be substituted identically or differently at each position. The substituents mentioned therein can be, but are not limited to, deuterium, fluorine, chlorine, bromine, iodine, cyano, hydroxyl, nitro, amino, carboxyl, alkyl, alkoxy, aryloxy, heteroaryl Oxy, heterocyclyloxy, arylalkoxy, heteroarylalkoxy, alkylacyl, heteroalkyl, cycloalkyl, cycloalkenyl, cycloalkylalkyl, heterocyclyl, heterocycle ylalkyl, heterocyclylacyl, aryl, arylalkyl, arylamino, heteroaryl, heteroarylalkyl, heteroarylamino, amido, sulfonyl, aminosulfonyl and the like.
另外,需要说明的是,除非以其他方式明确指出,在本发明中所采用的描述方式“各…独立地为”与“…各自独立地为”和“…独立地为”可以互换,均应做广义理解,其既可以是指在不同基团中,相同符号之间所表达的具体选项之间互相不影响,也可以表示在相同的基团中,相同符号之间所表达的具体选项之间互相不影响。以R10为例,结构式“R13-(CR10R11)n1-S(=O)p-(CR10R11)n-N(R12)-”和结构式“R13-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-”两者之间R10的具体选项互相之间不受影响,同时,在同一化学式“R13-(CR10R11)n-N(R12)-C(=O)-(CR10R11)n-(R12a)N-”内,两个R10的具体选项互相之间不受影响。In addition, it should be noted that, unless clearly stated otherwise, the description modes "each independently" and "...independently" and "...independently" used in the present invention can be interchanged, and both are interchangeable. It should be understood in a broad sense. It can either mean that in different groups, the specific options expressed between the same symbols do not affect each other, or it can mean that in the same group, the specific options expressed between the same symbols do not affect each other. Taking R 10 as an example, the structural formula "R 13 -(CR 10 R 11 ) n1 -S(=O) p -(CR 10 R 11 ) n -N(R 12 )-" and the structural formula "R 13 -(CR 10 )-" R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n -(R 12a )N-" The specific options of R 10 are not affected by each other, and at the same time, In the same chemical formula "R 13 -(CR 10 R 11 ) n -N(R 12 )-C(=O)-(CR 10 R 11 ) n - (R 12a )N-, the specific Options do not affect each other.
在本说明书的各部分,本发明公开化合物的取代基按照基团种类或范围公开。特别指出,本发明包括这些基团种类和范围的各个成员的每一个独立的次级组合。例如,术语“C1-C6烷基”或“C1-6烷基”特别指独立公开的甲基、乙基、C3烷基、C4烷基、C5烷基和C6烷基。In various parts of this specification, the substituents of the compounds disclosed in the present invention are disclosed in terms of group type or scope. Specifically, the present invention includes each and every independent subcombination of each member of these group species and ranges. For example, the term " C1 - C6 alkyl" or " C1-6 alkyl" specifically refers to the independently disclosed methyl, ethyl, C3 alkyl, C4 alkyl, C5 alkyl and C6 alkanes base.
在本发明的各部分,描述了连接取代基。当该结构清楚地需要连接基团时,针对该基团所列举的马库什变量应理解为连接基团。例如,如果该结构需要连接基团并且针对该变量的马库什基团定义列举了“烷基”或“芳基”,则应该理解,该“烷基”或“芳基”分别代表连接的亚烷基基团或亚芳基基团。例如,“CN-(CR10R11)n1-C1-9杂环基-C(=O)-N(R12)-”,分别通过C1-9杂环基与结构式的其余部分相连,当列举杂环基时,应理解为“杂环基”代表连接的亚杂环基基团。又例如“CN-(CR6R7)n-R0-(CR6R7)n-N(R9)-”中,R0为环烷基等,其中的环烷基等可以理解为亚环烷基基团等。In various parts of the present invention, linking substituents are described. When the structure clearly requires a linking group, the Markush variables listed for that group should be understood to be the linking group. For example, if the structure requires a linking group and the Markush group definition for that variable recites "alkyl" or "aryl", it should be understood that the "alkyl" or "aryl" respectively represents the linking An alkylene group or an arylene group. For example, "CN-(CR 10 R 11 ) n1 -C 1-9 heterocyclyl-C(=O)-N(R 12 )-", connected to the rest of the formula through C 1-9 heterocyclyl, respectively , when listing a heterocyclyl group, it should be understood that "heterocyclyl" represents the attached heterocyclylene group. For another example, in "CN-(CR 6 R 7 ) n -R 0 -(CR 6 R 7 ) n -N(R 9 )-", R 0 is a cycloalkyl group, etc., and the cycloalkyl group etc. can be understood as Cycloalkylene groups, etc.
本发明使用的术语“烷基”或“烷基基团”,表示含有1至10个碳原子,饱和的直链或支链一价烃基基团,其中,所述烷基基团可以任选地被一个或多个本发明描述的取代基所取代。除非另外详细说明,烷基基团含有1-6个碳原子;在又一实施方案中,烷基基团含有1-4个碳原子;还在一实施方案中,烷基基团含有1-3个碳原子。烷基基团的实例包含,但并不限于,甲基,乙基,正丙基,异丙基,正丁基,异丁基,仲丁基,叔丁基,正戊基,2-戊基,3-戊基,2-甲基-2-丁基,3-甲基-2-丁基,3-甲基-1-丁基,正己基,2-己基,等等。The term "alkyl" or "alkyl group" as used in the present invention refers to a saturated linear or branched monovalent hydrocarbon group containing 1 to 10 carbon atoms, wherein the alkyl group may be optionally is substituted with one or more substituents described herein. Unless otherwise specified, alkyl groups contain 1-6 carbon atoms; in yet another embodiment, alkyl groups contain 1-4 carbon atoms; in yet another embodiment, alkyl groups contain 1- 3 carbon atoms. Examples of alkyl groups include, but are not limited to, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, sec-butyl, tert-butyl, n-pentyl, 2-pentyl , 3-pentyl, 2-methyl-2-butyl, 3-methyl-2-butyl, 3-methyl-1-butyl, n-hexyl, 2-hexyl, etc.
术语“烯基”基团的实例包括,但并不限于,乙烯基,烯丙基,丙烯基等等。Examples of the term "alkenyl" group include, but are not limited to, vinyl, allyl, propenyl, and the like.
术语“炔基”基团的实例包括,但并不限于,乙炔基(-C≡CH),炔丙基(-CH2C≡CH)、1-丙炔基(-C≡C-CH3)等等。Examples of the term "alkynyl" group include, but are not limited to, ethynyl (-C≡CH), propargyl (-CH2C≡CH), 1 -propynyl (-C≡C- CH3) )and many more.
术语“烷氧基”表示烷基基团通过氧原子与分子其余部分相连,其中烷基基团具有如本发明所述的含义。除非另外详细说明,所述烷氧基基团含有1-6个碳原子;在另一实施方案中,烷氧基基团含有1-4个碳原子;在又一实施方案中,烷氧基基团含有1-3个碳原子。所述烷氧基基团可以任选地被一个或多个本发明描述的取代基所取代。烷氧基基团的实例包括,但并不限于,甲氧基,乙氧基,1-丙氧基,2-丙氧基,1-丁氧基,等等。The term "alkoxy" means that an alkyl group is attached to the remainder of the molecule through an oxygen atom, wherein the alkyl group has the meaning as described herein. Unless otherwise specified, the alkoxy group contains 1-6 carbon atoms; in another embodiment, the alkoxy group contains 1-4 carbon atoms; in yet another embodiment, the alkoxy group The group contains 1-3 carbon atoms. The alkoxy groups may be optionally substituted with one or more substituents described herein. Examples of alkoxy groups include, but are not limited to, methoxy, ethoxy, 1-propoxy, 2-propoxy, 1-butoxy, and the like.
术语“卤代烷基”,表示烷基基团被一个或多个卤素原子所取代,这样的实例包含,但并不限于,-CH2F,-CHF2,三氟甲基,-CH2Cl,-CHCl2,-CCl3,-CH2CF3,-CH2CH2CF3等。The term "haloalkyl" means an alkyl group substituted with one or more halogen atoms, examples of which include, but are not limited to, -CH2F , -CHF2 , trifluoromethyl, -CH2Cl , -CHCl 2 , -CCl 3 , -CH 2 CF 3 , -CH 2 CH 2 CF 3 , etc.
术语“碳环基”或“碳环”表示含有3-12个碳原子的,单价或多价的饱和或部分不饱和单环、双环或者三环体系,其中单环、双环或三环中不包含芳香环。碳双环基包括螺碳双环基和稠合碳双环基,合适的碳环基基团包括,但并不限于,环烷基、环烯基和环炔基。碳环基基团的实例进一步包括,环丙基,环丁 基,环戊基,1-环戊基-1-烯基,1-环戊基-2-烯基,1-环戊基-3-烯基,环己基,1-环己基-1-烯基,1-环己基-2-烯基,1-环己基-3-烯基,环己二烯基,环庚基,环辛基,环壬基,等等。The term "carbocyclyl" or "carbocycle" refers to a monovalent or polyvalent saturated or partially unsaturated monocyclic, bicyclic or tricyclic ring system containing 3 to 12 carbon atoms in which no Contains aromatic rings. Carbobicyclyl groups include spirocarbobicyclyl groups and fused carbobicyclyl groups, and suitable carbocyclyl groups include, but are not limited to, cycloalkyl, cycloalkenyl, and cycloalkynyl. Examples of carbocyclyl groups further include, cyclopropyl, cyclobutyl, cyclopentyl, 1-cyclopentyl-1-enyl, 1-cyclopentyl-2-enyl, 1-cyclopentyl- 3-alkenyl, cyclohexyl, 1-cyclohexyl-1-enyl, 1-cyclohexyl-2-enyl, 1-cyclohexyl-3-enyl, cyclohexadienyl, cycloheptyl, cyclooctyl base, cyclononyl, etc.
术语“环烷基”表示含有3-12个碳原子的,单价或多价的饱和单环,双环或三环体系,其中单环、双环或三环中不包含芳香环。在一实施方案中,环烷基包含3-9个碳原子;在又一实施方案中,环烷基包含3-6个碳原子。所述环烷基基团可以独立地未被取代或被一个或多个本发明所描述的取代基所取代。这样的实例包括,但并不限于,环丙基,环丁基,环戊基,环己基,环庚基,环辛基,环壬基,环癸基,环十一烷基,环十二烷基,等等。The term "cycloalkyl" refers to a monovalent or polyvalent saturated monocyclic, bicyclic or tricyclic ring system containing 3 to 12 carbon atoms, wherein the monocyclic, bicyclic or tricyclic rings do not contain aromatic rings. In one embodiment, the cycloalkyl group contains 3-9 carbon atoms; in yet another embodiment, the cycloalkyl group contains 3-6 carbon atoms. The cycloalkyl groups may independently be unsubstituted or substituted with one or more substituents described herein. Such examples include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, cycloundecyl, cyclododecyl Alkyl, etc.
术语“环烯基”的实例包括,但并不限于,环丁烯基,环戊烯基,环戊二烯基,环己烯基,环己二烯基,环庚烯基,环辛烯基,环壬烯基和环癸烯基,等等。Examples of the term "cycloalkenyl" include, but are not limited to, cyclobutenyl, cyclopentenyl, cyclopentadienyl, cyclohexenyl, cyclohexadienyl, cycloheptenyl, cyclooctene , cyclononenyl and cyclodecenyl, among others.
术语“杂环基”和“杂环”在此处可交换使用,都是指包含3-12个环原子的饱和或部分不饱和的单环、双环或三环,其中单环、双环或三环中不包含芳香环,且至少一个环原子选自氮、硫和氧原子。除非另外说明,杂环基可以是碳基或氮基,且-CH2-基团可以任选地被-C(=O)-替代。环的硫原子可以任选地被氧化成S-氧化物。环的氮原子可以任选地被氧化成N-氧化合物。杂环基中-CH2-基团被-C(=O)-取代的实例包括,但不限于,2-氧代吡咯烷基,氧代-1,3-噻唑烷基,2-哌啶酮基,3,5-二氧代哌啶基。杂环基中硫原子被氧化的实例包括,但不限于,环丁砜基,1,1-二氧代硫代吗啉基。The terms "heterocyclyl" and "heterocycle" are used interchangeably herein, and both refer to a saturated or partially unsaturated monocyclic, bicyclic or tricyclic ring containing from 3 to 12 ring atoms, wherein a monocyclic, bicyclic or tricyclic ring The ring contains no aromatic rings and at least one ring atom is selected from nitrogen, sulfur and oxygen atoms. Unless otherwise specified, heterocyclyl can be carbon or nitrogen, and -CH2- groups can be optionally replaced by -C(=O)-. Ring sulfur atoms can optionally be oxidized to S-oxides. The nitrogen atoms of the rings can be optionally oxidized to N-oxygen compounds. Examples of heterocyclyl groups where the -CH2- group is substituted with -C(=O)- include, but are not limited to, 2-oxopyrrolidinyl, oxo-1,3-thiazolidinyl, 2-piperidine Keto, 3,5-dioxopiperidinyl. Examples of oxidized sulfur atoms in a heterocyclyl group include, but are not limited to, sulfolanyl, 1,1-dioxothiomorpholinyl.
在一实施方案中,杂环基为4-7个原子组成的杂环基,是指包含4-7个环原子的饱和或部分不饱和的单环,其中至少一个环原子选自氮、硫和氧原子。实例包括,但不限于:氮杂环丁基,氧杂环丁基,硫杂环丁基,吡咯烷基,2-吡咯啉基,3-吡咯啉基,吡唑烷基,咪唑啉基,咪唑烷基,四氢呋喃基,二氢呋喃基,四氢噻吩基,二氢噻吩基,1,3-二氧环戊基,二硫环戊基,四氢吡喃基,二氢吡喃基,2H-吡喃基,4H-吡喃基,四氢噻喃基,哌啶基,四氢吡啶基,吗啉基,硫代吗啉基,哌嗪基,二噁烷基,二噻烷基,噻噁烷基,高哌嗪基,高哌啶基,氧杂环庚烷基,硫杂环庚烷基。杂环基中-CH2-基团被-C(=O)-取代的实例包括,但不限于,2-氧代吡咯烷基,氧代-1,3-噻唑烷基,2-哌啶酮基,3,5-二氧代哌啶基。杂环基中硫原子被氧化的实例包括,但不限于,环丁砜基,1,1-二氧代硫代吗啉基。所述的4-7个原子组成的杂环基基团可以任选地被一个或多个本发明所描述的取代基所取代。在另一实施方案中,杂环基为4个原子组成的杂环基,实例包括,但不限于:氮杂环丁基,氧杂环丁基,硫杂环丁基。在另一实施方案中,杂环基为5个原子组成的杂环基,实例包括,但不限于:吡咯烷基,2-吡咯啉基,3-吡咯啉基,吡唑烷基,咪唑啉基,咪唑烷基,四氢呋喃基,二氢呋喃基,四氢噻吩基,二氢噻吩基,1,3-二氧环戊基,二硫环戊基,2-氧代吡咯烷基,氧代-1,3-噻唑烷基,环丁砜基。在另一实施方案中,杂环基为6个原子组成的杂环基,实例包括,但不限于:四氢吡喃基,二氢吡喃基,2H-吡喃基,4H-吡喃基,四氢噻喃基,哌啶基,吗啉基,硫代吗啉基,哌嗪基,二噁烷基,二噻烷基,噻噁烷基,2-哌啶酮基,3,5-二氧代哌啶基,1,1-二氧代硫代吗啉基。所述的6个原子组成的杂环基基团可以任选地被一个或多个本发明所描述的取代基所取代。In one embodiment, the heterocyclyl group is a heterocyclyl group consisting of 4-7 atoms, which refers to a saturated or partially unsaturated monocyclic ring containing 4-7 ring atoms, wherein at least one ring atom is selected from nitrogen, sulfur and oxygen atoms. Examples include, but are not limited to: azetidinyl, oxetanyl, thietanyl, pyrrolidinyl, 2-pyrrolinyl, 3-pyrrolinyl, pyrazolidinyl, imidazolinyl, Imidazolidinyl, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothienyl, dihydrothienyl, 1,3-dioxopentyl, dithiocyclopentyl, tetrahydropyranyl, dihydropyranyl, 2H-pyranyl, 4H-pyranyl, tetrahydrothiopyranyl, piperidinyl, tetrahydropyridyl, morpholinyl, thiomorpholinyl, piperazinyl, dioxanyl, dithianyl , thioxanyl, homopiperazinyl, homopiperidinyl, oxepanyl, thiepanyl. Examples of heterocyclyl groups where the -CH2- group is substituted with -C(=O)- include, but are not limited to, 2-oxopyrrolidinyl, oxo-1,3-thiazolidinyl, 2-piperidine Keto, 3,5-dioxopiperidinyl. Examples of oxidized sulfur atoms in heterocyclyl groups include, but are not limited to, sulfolanyl, 1,1-dioxothiomorpholinyl. Said heterocyclyl group consisting of 4-7 atoms can be optionally substituted by one or more substituents described in the present invention. In another embodiment, the heterocyclyl group is a 4-atom heterocyclyl group, examples include, but are not limited to: azetidine, oxetanyl, thietanyl. In another embodiment, the heterocyclyl group is a 5-atom heterocyclyl group, examples include, but are not limited to: pyrrolidinyl, 2-pyrrolinyl, 3-pyrrolidinyl, pyrazolidinyl, imidazoline base, imidazolidinyl, tetrahydrofuranyl, dihydrofuranyl, tetrahydrothienyl, dihydrothienyl, 1,3-dioxocyclopentyl, dithiocyclopentyl, 2-oxopyrrolidinyl, oxo -1,3-thiazolidinyl, sulfolane. In another embodiment, the heterocyclyl group is a 6-atom heterocyclyl group, examples include, but are not limited to: tetrahydropyranyl, dihydropyranyl, 2H-pyranyl, 4H-pyranyl , tetrahydrothiopyranyl, piperidinyl, morpholinyl, thiomorpholinyl, piperazinyl, dioxanyl, dithianyl, thioxanyl, 2-piperidinyl, 3,5 -Dioxopiperidyl, 1,1-dioxothiomorpholinyl. The 6-atom heterocyclyl group may be optionally substituted with one or more substituents described herein.
术语“稠合双环”和“稠合双环基”在此处可交换使用,是指单价或多价的饱和或部分不饱和的桥环体系,所述桥环体系是指双环体系。术语“桥环”是指任意两个环共用两个直接相连或不直接相连的原子。实例包括,但并不限于,六氢呋喃并[3,2-b]呋喃,2,3,3a,4,7,7a-六氢-1H-茚,7-氮杂双环[2.2.1]庚烷,稠合双环[3.3.0]辛烷,稠合双环[3.1.0]己烷,1,2,3,4,4a,5,8,8a-八氢萘。The terms "fused bicyclic" and "fused bicyclyl" are used interchangeably herein to refer to a monovalent or polyvalent saturated or partially unsaturated bridged ring system, which refers to a bicyclic ring system. The term "bridged ring" refers to any two rings that share two atoms that may or may not be directly connected. Examples include, but are not limited to, hexahydrofuro[3,2-b]furan, 2,3,3a,4,7,7a-hexahydro-1H-indene, 7-azabicyclo[2.2.1] Heptane, fused bicyclo[3.3.0]octane, fused bicyclo[3.1.0]hexane, 1,2,3,4,4a,5,8,8a-octahydronaphthalene.
术语“稠合杂双环”表示饱和或部分不饱和的稠环体系,且至少一个环体系包含一个或多个杂原子,其中每一个环体系包含3-7元环,即包含1-6个碳原子和选自N,O,P,S的1-3个杂原子,在此S或P任选地被一个或多个氧原子所取代得到像SO,SO2,PO,PO2的基团,这样的实例包括,但并不限于六氢呋喃并[3,2-b]呋喃,7-氮杂双环[2.2.1]庚烷,3-氮杂双环[3.3.0]辛烷,3,5,8-三氧杂双环[5,1,0]辛烷,1-氮杂-4,6-二氧双环[3.3.0]辛烷等,并且所述稠合杂双环基可以是取代或非取代的。The term "fused heterobicycle" means a saturated or partially unsaturated fused ring system, and at least one ring system contains one or more heteroatoms, wherein each ring system contains a 3-7 membered ring, ie, contains 1-6 carbons atom and 1-3 heteroatoms selected from N, O, P, S, where S or P is optionally substituted with one or more oxygen atoms to give groups like SO, SO 2 , PO, PO 2 , such examples include, but are not limited to, hexahydrofuro[3,2-b]furan, 7-azabicyclo[2.2.1]heptane, 3-azabicyclo[3.3.0]octane, 3 , 5,8-trioxabicyclo[5,1,0]octane, 1-aza-4,6-dioxabicyclo[3.3.0]octane, etc., and the fused heterobicyclo group may be substituted or unsubstituted.
术语“螺双环基”或“螺双环”在此处可交换使用,是指单价或多价的饱和或部分不饱和环体系,其中一个环起源于另一个环上特定的环碳原子。螺双环基中的每个环都可以是碳环基或杂环基,并且每个环任选地被一个或多个本发明所描述的取代基所取代。The terms "spirobicyclyl" or "spirobicycle" are used interchangeably herein and refer to a monovalent or polyvalent saturated or partially unsaturated ring system wherein one ring originates from a specified ring carbon atom on the other ring. Each ring in a spirobicyclyl can be carbocyclyl or heterocyclyl, and each ring is optionally substituted with one or more substituents described herein.
术语“螺杂双环基”表示一个环起源于另一个环上特殊的环状碳,且至少一个环体系包含一个或多个杂原子,其中每一个环体系包含3-7元环,即包含1-6个碳原子和选自N,O,P,S的1-3个杂原子,在此S或P任选地被一个或多个氧原子所取代得到例如SO,SO2,PO,PO2的基团,这样的实例包括,但并不限于4-氮杂螺[2.4]庚烷-5-基,4-氧杂螺[2.4]庚烷-5-基,5-氮杂螺[2.4]庚烷-5-基,7-羟基-5-氮杂螺[2.4]庚烷-5-基等。The term "spiroheterobicyclyl" means that one ring originates from a particular cyclic carbon on the other ring, and at least one ring system contains one or more heteroatoms, wherein each ring system contains a 3-7 membered ring, i.e. contains 1 - 6 carbon atoms and 1-3 heteroatoms selected from N, O, P, S, where S or P are optionally substituted by one or more oxygen atoms to give eg SO, SO 2 , PO, PO 2 groups, such examples include, but are not limited to, 4-azaspiro[2.4]heptan-5-yl, 4-oxaspiro[2.4]heptan-5-yl, 5-azaspiro[ 2.4]Heptan-5-yl, 7-hydroxy-5-azaspiro[2.4]heptan-5-yl and the like.
术语“杂原子”是指O、S、N、P和Si,包括N、S和P任何氧化态的形式;伯、仲、叔胺和季铵盐的形式;或者杂环中氮原子上的氢被取代的形式,例如,N(像3,4-二氢-2H-吡咯基中的N),NH(像吡咯烷基中的NH)或NR(像N-取代的吡咯烷基中的NR)。The term "heteroatom" refers to O, S, N, P, and Si, including N, S, and P in any oxidation state; in the form of primary, secondary, tertiary amines, and quaternary ammonium salts; or on a nitrogen atom in a heterocyclic ring. Hydrogen substituted form, for example, N (like N in 3,4-dihydro-2H-pyrrolidinyl), NH (like NH in pyrrolidinyl) or NR (like in N-substituted pyrrolidinyl) NR).
术语“卤素”是指氟(F)、氯(Cl)、溴(Br)或碘(I)。The term "halogen" refers to fluorine (F), chlorine (Cl), bromine (Br) or iodine (I).
术语“芳基”表示含有6-14个环原子,或6-12个环原子,或6-10个环原子的单环、双环和三环的碳环体系,其中,至少一个环体系是芳香族的,其中每一个环体系包含3-7个原子组成的环,且有一个或多个附着点与分子的其余部分相连。术语“芳基”可以和术语“芳香环”交换使用。芳基基团的实例可以包括苯基,茚基,萘基和蒽基。The term "aryl" refers to monocyclic, bicyclic and tricyclic carbocyclic ring systems containing 6-14 ring atoms, or 6-12 ring atoms, or 6-10 ring atoms, wherein at least one ring system is aromatic A family of rings in which each ring system contains a ring of 3-7 atoms with one or more points of attachment to the rest of the molecule. The term "aryl" is used interchangeably with the term "aromatic ring". Examples of aryl groups may include phenyl, indenyl, naphthyl, and anthracenyl.
术语“杂芳基”表示含有5-12个环原子,或5-10个环原子,或5-6个环原子的单环、双环和三环体系,其中至少一个环体系是芳香族的,且至少一个环体系包含一个或多个杂原子,其中每一个环体系包含5-7个原子组成的环,且有一个或多个附着点与分子其余部分相连。术语“杂芳基”可以与术语“杂芳环”或“杂芳族化合物”交换使用。所述杂芳基基团任选地被一个或多个本发明所描述的取代基所取代。在一实施方案中,5-10个原子组成的杂芳基包含1,2,3或4个独立选自O,S和N的杂原子。杂芳基基团的实例包括,但并不限于,2-呋喃基,3-呋喃基,N-咪唑基,2-咪唑基,4-咪唑基,5-咪唑基,3-异噁唑基,4-异噁唑基,5-异噁唑基,2-噁唑基,4-噁唑基,5-噁唑基,N-吡咯基,2-吡咯基,3-吡咯基,2-吡啶基,3-吡啶基,4-吡啶基,2-嘧啶基,4-嘧啶基,5-嘧啶基,哒嗪基(如3-哒嗪基),2-噻唑基,4-噻唑基,5-噻唑基,四唑基(如5-四唑基),三唑基(如2-三唑基和5-三唑基),2-噻吩基,3-噻吩基,吡唑基(如2-吡唑基),异噻唑基,1,2,3-噁二唑基,1,2,5-噁二唑基,1,2,4-噁二唑基,1,2,3-三唑基,1,2,3-硫代二唑基,1,3,4-硫代二唑基,1,2,5-硫代二唑基,吡嗪基,1,3,5-三嗪基,嘧啶酮基,吡啶酮基;也包括以下的双环,但绝不限于这些双环:苯并咪唑基,苯并呋喃基,苯并四氢呋喃基,苯并噻吩基,吲哚基(如2-吲哚基),嘌呤基,喹啉基(如2-喹啉基,3-喹啉基,4-喹啉基),四氢喹啉基(如1,2,3,4-四氢喹啉基),异喹啉基(如1-异喹啉基、3-异喹啉基或4-异喹啉基),四氢异喹啉基(如1,2,3,4-四氢异喹啉基),咪唑并[1,2-a]吡啶基,吡唑并[1,5-a]吡啶基,吡唑并[1,5-a]嘧啶基,咪唑并[1,2-b]哒嗪基,[1,2,4]三唑并[4,3-b]哒嗪基,[1,2,4]三唑并[1,5-a]嘧啶基,[1,2,4]三唑并[1,5-a]吡啶基,等等。The term "heteroaryl" refers to monocyclic, bicyclic and tricyclic ring systems containing 5-12 ring atoms, or 5-10 ring atoms, or 5-6 ring atoms, wherein at least one ring system is aromatic, And at least one ring system contains one or more heteroatoms, wherein each ring system contains a ring of 5-7 atoms and has one or more points of attachment to the rest of the molecule. The term "heteroaryl" may be used interchangeably with the terms "heteroaromatic ring" or "heteroaromatic". The heteroaryl group is optionally substituted with one or more substituents described herein. In one embodiment, a heteroaryl group of 5-10 atoms contains 1, 2, 3 or 4 heteroatoms independently selected from O, S and N. Examples of heteroaryl groups include, but are not limited to, 2-furyl, 3-furyl, N-imidazolyl, 2-imidazolyl, 4-imidazolyl, 5-imidazolyl, 3-isoxazolyl , 4-isoxazolyl, 5-isoxazolyl, 2-oxazolyl, 4-oxazolyl, 5-oxazolyl, N-pyrrolyl, 2-pyrrolyl, 3-pyrrolyl, 2- Pyridyl, 3-pyridyl, 4-pyridyl, 2-pyrimidinyl, 4-pyrimidinyl, 5-pyrimidinyl, pyridazinyl (such as 3-pyridazinyl), 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, tetrazolyl (such as 5-tetrazolyl), triazolyl (such as 2-triazolyl and 5-triazolyl), 2-thienyl, 3-thienyl, pyrazolyl (such as 2-pyrazolyl), isothiazolyl, 1,2,3-oxadiazolyl, 1,2,5-oxadiazolyl, 1,2,4-oxadiazolyl, 1,2,3- Triazolyl, 1,2,3-thiodiazolyl, 1,3,4-thiodiazolyl, 1,2,5-thiodiazolyl, pyrazinyl, 1,3,5- Triazinyl, pyrimidinonyl, pyridone; also including, but in no way limited to, the following bicyclic rings: benzimidazolyl, benzofuranyl, benzotetrahydrofuranyl, benzothienyl, indolyl (eg 2-indolyl), purinyl, quinolinyl (such as 2-quinolinyl, 3-quinolinyl, 4-quinolinyl), tetrahydroquinolinyl (such as 1,2,3,4-tetrahydroquinolinyl) Hydroquinolyl), isoquinolyl (such as 1-isoquinolyl, 3-isoquinolyl or 4-isoquinolyl), tetrahydroisoquinolyl (such as 1,2,3,4- tetrahydroisoquinolinyl), imidazo[1,2-a]pyridyl, pyrazolo[1,5-a]pyridyl, pyrazolo[1,5-a]pyrimidinyl, imidazo[1 ,2-b]pyridazinyl, [1,2,4]triazolo[4,3-b]pyridazinyl, [1,2,4]triazolo[1,5-a]pyrimidinyl, [1,2,4]Triazolo[1,5-a]pyridyl, and the like.
在一实施方案中,杂芳基基团为以下子结构式:In one embodiment, the heteroaryl group is of the following substructure:
等等。 and many more.
术语“羧基”,无论是单独使用还是和其他术语连用,如“羧烷基”,表示-CO2H。The term "carboxy", whether used alone or in combination with other terms, such as "carboxyalkyl", means -CO2H .
如本发明所描述,取代基R由一个键连接到中心的环上形成的环体系代表取代基R可以在环上任何可取代或任何合理的位置进行取代。例如,式a代表D环和B环上任何可能被取代的位置均可被R取代,如式b,式c,式d,式e,式f,式g和式h所示。As described herein, a ring system in which a substituent R is attached to a central ring by a bond represents that the substituent R can be substituted at any substitutable or any reasonable position on the ring. For example, formula a represents ring D and any possible substituted position on ring B can be substituted by R, as shown in formula b, formula c, formula d, formula e, formula f, formula g and formula h.
如本发明所描述,取代基(R)n由一个键连接到中心的环上形成的环体系代表n个取代基R可以在环上任何可取代的位置进行取代。例如,式i代表D环和B环上任何可能被取代的位置均可被n个R取代。As described in the present invention, a ring system formed by a substituent (R) n attached to the central ring by a bond represents that the n substituents R may be substituted at any substitutable position on the ring. For example, Formula i represents that any possible substituted position on the D and B rings can be substituted with n Rs.
像本发明所描述的,环C上有两个连接点可与分子其余部分相连,例如,如式j所示,表示既可以是E”端也可以是E’端与分子的其余部分相连,即两端的连接方式可以互换。As described in the present invention, there are two points of attachment on the ring C that can be connected to the rest of the molecule, for example, as shown in formula j, indicating that either the E" end or the E' end is connected to the rest of the molecule, That is, the connection methods at both ends can be interchanged.
如本发明所描述,附着点可以在环上任何可连接的位置与分子其余部分连接。例如,式k代表环上任何可能被连接的位置均可作为连接的点。An attachment point can be attached to the rest of the molecule at any linkable position on the loop as described herein. For example, formula k represents that any position on the ring that may be attached can serve as a point of attachment.
如本发明所描述,附着点可以在环上任何可连接的位置与分子其余部分连接,同时连接的两端可以互换。例如,式m代表环上任何可能被连接的位置均可作为连接的点,同时连接点的两端可以互换。As described herein, the point of attachment can be attached to the rest of the molecule at any attachable position on the loop, while the two ends of the attachment can be interchanged. For example, the formula m represents that any position on the ring that may be connected can be used as a connection point, and the two ends of the connection point can be interchanged.
术语“保护基团”或“PG”是指一个取代基与其他官能团起反应的时候,通常用来阻断或保护特殊的 功能性。例如,“氨基的保护基团”是指一个取代基与氨基基团相连来阻断或保护化合物中氨基的功能性,合适的氨基保护基团包括乙酰基,三氟乙酰基,叔丁氧羰基(BOC,Boc),苄氧羰基(CBZ,Cbz)和9-芴甲氧羰基(Fmoc)。相似地,“羟基保护基团”是指羟基的取代基用来阻断或保护羟基的功能性,合适的保护基团包括乙酰基和甲硅烷基。“羧基保护基团”是指羧基的取代基用来阻断或保护羧基的功能性,一般的羧基保护基包括-CH2CH2SO2Ph,氰基乙基,2-(三甲基硅烷基)乙基,2-(三甲基硅烷基)乙氧基甲基,2-(对甲苯磺酰基)乙基,2-(对硝基苯磺酰基)乙基,2-(二苯基膦基)乙基,硝基乙基,等等。对于保护基团一般的描述可参考文献:T W.Greene,Protective Groups in Organic Synthesis,John Wiley&Sons,New York,1991;and P.J.Kocienski,Protecting Groups,Thieme,Stuttgart,2005。The term "protecting group" or "PG" refers to a substituent group that is commonly used to block or protect specific functionality when it reacts with other functional groups. For example, "amino protecting group" refers to a substituent attached to the amino group to block or protect the functionality of the amino group in the compound. Suitable amino protecting groups include acetyl, trifluoroacetyl, t-butoxycarbonyl (BOC, Boc), benzyloxycarbonyl (CBZ, Cbz) and 9-fluorenemethoxycarbonyl (Fmoc). Similarly, a "hydroxyl protecting group" refers to a substituent of a hydroxy group used to block or protect the functionality of the hydroxy group, suitable protecting groups include acetyl and silyl. "Carboxyl protecting group" means that the substituent of the carboxyl group is used to block or protect the functionality of the carboxyl group. General carboxyl protecting groups include -CH 2 CH 2 SO 2 Ph, cyanoethyl, 2-(trimethylsilane) yl)ethyl, 2-(trimethylsilyl)ethoxymethyl, 2-(p-toluenesulfonyl)ethyl, 2-(p-nitrobenzenesulfonyl)ethyl, 2-(diphenyl) phosphino)ethyl, nitroethyl, and the like. For a general description of protecting groups, reference can be made to the literature: TW. Greene, Protective Groups in Organic Synthesis, John Wiley & Sons, New York, 1991; and PJ Kocienski, Protecting Groups, Thieme, Stuttgart, 2005.
本发明所使用的术语“前药”,代表一个化合物在体内转化为式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所示的化合物。这样的转化受前体药物在血液中水解或在血液或组织中经酶转化为母体结构的影响。本发明前体药物类化合物可以是酯,在现有的发明中酯可以作为前体药物的有苯酯类,脂肪族(C1-24)酯类,酰氧基甲基酯类,碳酸酯,氨基甲酸酯类和氨基酸酯类。例如本发明里的一个化合物包含羟基,即可以将其酰化得到前体药物形式的化合物。其他的前体药物形式包括磷酸酯,如这些磷酸酯类化合物是经母体上的羟基磷酸化得到的。关于前体药物完整的讨论可以参考以下文献:T.Higuchiand V.Stella,Pro-drugs as Novel Delivery Systems,Vol.14of the A.C.S.SymposiumSeries,Edward B.Roche,ed.,Bioreversible Carriers in Drug Design,AmericanPharmaceutical Association and Pergamon Press,1987,J.Rautio et al.,Prodrugs:Design and Clinical Applications,Nature Review Drug Discovery,2008,7,255-270,and S.J.Hecker et al.,Prodrugs of Phosphates and Phosphonates,Journal ofMedicinal Chemistry,2008,51,2328-2345。The term "prodrug", as used in the present invention, represents the in vivo conversion of a compound of formula (I), formula (Ia), formula (Ib), formula (Ic), formula (II), formula (IIa) or formula ( The compound shown in III). Such conversion is effected by hydrolysis of the prodrug in blood or enzymatic conversion to the parent structure in blood or tissue. The prodrug compounds of the present invention can be esters. In the existing invention, esters that can be used as prodrugs include phenyl esters, aliphatic (C 1-24 ) esters, acyloxymethyl esters, carbonate esters , carbamates and amino acid esters. For example, a compound of the present invention contains a hydroxyl group, which can be acylated to give the compound in prodrug form. Other prodrug forms include phosphates, such as these phosphates are phosphorylated by the hydroxyl group on the parent. A complete discussion of prodrugs can be found in the following references: T. Higuchi and V. Stella, Pro-drugs as Novel Delivery Systems, Vol. 14 of the ACSSymposium Series, Edward B. Roche, ed., Bioreversible Carriers in Drug Design, American Pharmaceutical Association and Pergamon Press, 1987, J. Rautio et al., Prodrugs: Design and Clinical Applications, Nature Review Drug Discovery, 2008, 7, 255-270, and SJ Hecker et al., Prodrugs of Phosphates and Phosphonates, Journal of Medicinal Chemistry, 2008, 51, 2328-2345.
“代谢产物”是指具体的化合物或其盐在体内通过代谢作用所得到的产物。一个化合物的代谢产物可以通过所属领域公知的技术来进行鉴定,其活性可以通过如本发明所描述的那样采用试验的方法进行表征。这样的产物可以是通过给药化合物经过氧化,还原,水解,酰氨化,脱酰氨作用,酯化,脱脂作用,酶裂解等等方法得到。相应地,本发明包括化合物的代谢产物,包括将本发明的化合物与哺乳动物充分接触一段时间所产生的代谢产物。"Metabolite" refers to a product obtained by metabolism of a specific compound or salt thereof in vivo. Metabolites of a compound can be identified by techniques well known in the art, and their activity can be characterized using assays as described herein. Such products may be obtained by subjecting the administered compound to oxidation, reduction, hydrolysis, amidation, deamidation, esterification, delipidation, enzymatic cleavage, and the like. Accordingly, the present invention includes metabolites of compounds, including metabolites produced by contacting a compound of the present invention with a mammal for a sufficient period of time.
本发明所使用的“药学上可接受的盐”是指本发明的化合物的有机盐和无机盐。药学上可接受的盐在所属领域是为我们所熟知的,如文献:S.M.Berge et al.,describepharmaceutically acceptable salts in detail in J.Pharmaceutical Sciences,1977,66:1-19.所记载的。药学上可接受的无毒的酸形成的盐包括,但并不限于,与氨基基团反应形成的无机酸盐有盐酸盐,氢溴酸盐,磷酸盐,硫酸盐,高氯酸盐,和有机酸盐如乙酸盐,草酸盐,马来酸盐,酒石酸盐,柠檬酸盐,琥珀酸盐,丙二酸盐,或通过书籍文献上所记载的其他方法如离子交换法来得到这些盐。其他药学上可接受的盐包括己二酸盐,藻酸盐,抗坏血酸盐,天冬氨酸盐,苯磺酸盐,苯甲酸盐,重硫酸盐,硼酸盐,丁酸盐,樟脑酸盐,樟脑磺酸盐,环戊基丙酸盐,二葡萄糖酸盐,十二烷基硫酸盐,乙磺酸盐,甲酸盐,反丁烯二酸盐,葡庚糖酸盐,甘油磷酸盐,葡萄糖酸盐,半硫酸盐,庚酸盐,己酸盐,氢碘酸盐,2-羟基-乙磺酸盐,乳糖醛酸盐,乳酸盐,月桂酸盐,月桂基硫酸盐,苹果酸盐,丙二酸盐,甲磺酸盐,2-萘磺酸盐,烟酸盐,硝酸盐,油酸盐,棕榈酸盐,扑酸盐,果胶酸盐,过硫酸盐,3-苯基丙酸盐,苦味酸盐,特戊酸盐,丙酸盐,硬脂酸盐,硫氰酸盐,对甲苯磺酸盐,十一酸盐,戊酸盐,等等。通过适当的碱得到的盐包括碱金属,碱土金属,铵和N+(C1-4烷基)4的盐。本发明也拟构思了任何所包含N的基团的化合物所形成的季铵盐。水溶性或油溶性或分散产物可以通过季铵化作用得到。碱金属或碱土金属盐包括钠,锂,钾,钙,镁,等等。药学上可接受的盐进一步包括适当的、无毒的铵,季铵盐和抗平衡离子形成的胺阳离子,如卤化物,氢氧化物,羧化物,硫酸化物,磷酸化物,硝酸化物,C1-8磺酸化物和芳香磺酸化物。As used herein, "pharmaceutically acceptable salts" refer to organic and inorganic salts of the compounds of the present invention. Pharmaceutically acceptable salts are well known in the art, as described in the literature: SM Berge et al., describe pharmaceutically acceptable salts in detail in J. Pharmaceutical Sciences, 1977, 66: 1-19. Pharmaceutically acceptable non-toxic acid salts include, but are not limited to, inorganic acid salts formed by reaction with amino groups including hydrochloride, hydrobromide, phosphate, sulfate, perchlorate, And organic acid salts such as acetate, oxalate, maleate, tartrate, citrate, succinate, malonate, or obtained by other methods such as ion exchange method described in books and literature these salts. Other pharmaceutically acceptable salts include adipate, alginate, ascorbate, aspartate, besylate, benzoate, bisulfate, borate, butyrate, camphoric acid Salt, camphorsulfonate, cyclopentylpropionate, digluconate, lauryl sulfate, ethanesulfonate, formate, fumarate, glucoheptonate, glycerophosphate Salt, Gluconate, Hemisulfate, Heptanoate, Caproate, Hydroiodide, 2-Hydroxy-ethanesulfonate, Lacturonate, Lactate, Laurate, Lauryl Sulfate, Malonate, Malonate, Mesylate, 2-Naphthalenesulfonate, Nicotinate, Nitrate, Oleate, Palmitate, Pamoate, Pectate, Persulfate, 3 - Phenylpropionate, picrate, pivalate, propionate, stearate, thiocyanate, p-toluenesulfonate, undecanoate, valerate, etc. Salts obtained with appropriate bases include alkali metal, alkaline earth metal, ammonium and N + (C 1-4 alkyl) 4 salts. The present invention also contemplates quaternary ammonium salts formed from any compound containing an N-group. Water- or oil-soluble or dispersible products can be obtained by quaternization. Alkali metal or alkaline earth metal salts include sodium, lithium, potassium, calcium, magnesium, and the like. Pharmaceutically acceptable salts further include appropriate, non-toxic ammonium, quaternary ammonium salts and amine cations that resist the formation of counterions, such as halides, hydroxides, carboxylates, sulfates, phosphates, nitrates, C1 -8 Sulfonates and aromatic sulfonates.
本发明的“溶剂化物”是指一个或多个溶剂分子与本发明的化合物所形成的缔合物。形成溶剂化物的 溶剂包括,但并不限于,水,异丙醇,乙醇,甲醇,二甲亚砜,乙酸乙酯,乙酸和氨基乙醇。术语“水合物”是指溶剂分子是水所形成的缔合物。A "solvate" of the present invention refers to an association of one or more solvent molecules with a compound of the present invention. Solvate-forming solvents include, but are not limited to, water, isopropanol, ethanol, methanol, dimethyl sulfoxide, ethyl acetate, acetic acid, and aminoethanol. The term "hydrate" refers to an association in which the solvent molecule is water.
本发明所使用的“炎性疾病”是指由于过度或失控的炎性响应所导致的过度的炎性症状、宿主组织损害或组织功能丧失的任何疾病,紊乱或症状。“炎性疾病”还指受白细胞流入和/或嗜中性粒细胞趋化性介导的病理学状态。As used herein, "inflammatory disease" refers to any disease, disorder or symptom of excessive inflammatory symptoms, host tissue damage, or loss of tissue function due to an excessive or uncontrolled inflammatory response. "Inflammatory disease" also refers to pathological conditions mediated by leukocyte influx and/or neutrophil chemotaxis.
本发明所使用的“炎症”是指由组织受损或破坏引起的局部保护性响应,它用于破坏、稀释或隔开(隔绝)有害的物质和受损的组织。炎症与白细胞流入和/或嗜中性粒细胞趋化性有显著的联系。炎症可以产生于病原性生物体和病毒的感染以及非传染性方式,如心肌梗塞或中风后的创伤或再灌注,对外来抗原的免疫应答和自身免疫应答。因此,可以用本发明公开化合物治疗的炎性疾病包括:与特异性防御系统反应以及非特异性防御系统反应相关的疾病。"Inflammation," as used herein, refers to a localized protective response caused by tissue damage or destruction, which serves to destroy, dilute, or isolate (sequester) harmful substances and damaged tissue. Inflammation is significantly associated with leukocyte influx and/or neutrophil chemotaxis. Inflammation can arise from infection by pathogenic organisms and viruses as well as from non-infectious means such as trauma or reperfusion following myocardial infarction or stroke, immune responses to foreign antigens and autoimmune responses. Accordingly, inflammatory diseases that can be treated with the disclosed compounds include diseases associated with specific defense system responses as well as non-specific defense system responses.
本发明所使用的“自体免疫疾病”或“自身免疫性疾病”是指与体液或细胞介导的对身体自身组分应答相关的组织损伤的任意疾病的集合。自体免疫疾病的实例包括狼疮,多发性硬化,肌肉缩性侧索硬化,类风湿性关节炎,银屑病,I型糖尿病,因器官移植导致的并发症,异物移植,糖尿病,癌症,哮喘,特应性皮炎,自身免疫性甲状腺病,溃疡性结肠炎,克罗恩病,阿尔茨海默病,白血病和淋巴瘤。As used herein, "autoimmune disease" or "autoimmune disease" refers to any collection of diseases associated with tissue damage associated with a humoral or cell-mediated response to the body's own components. Examples of autoimmune diseases include lupus, multiple sclerosis, lateral sclerosis, rheumatoid arthritis, psoriasis, type I diabetes, complications from organ transplantation, xenotransplantation, diabetes, cancer, asthma, Atopic dermatitis, autoimmune thyroid disease, ulcerative colitis, Crohn's disease, Alzheimer's disease, leukemia and lymphoma.
如本发明所使用的“关节炎疾病”是指以可归因于各种病因学的关节炎性损伤为特征的任意疾病。如本发明所使用的“皮炎”是指以可归因于各种病因学的皮肤炎症为特征的皮肤疾病的大家族中的任意一种。如本发明所使用的“移植排斥”是指以移植或周围组织的功能丧失、疼痛、肿胀、白细胞增多和血小板减少为特征的对抗移植组织,如器官或细胞(如骨髓)的任意免疫反应。本发明的治疗方法包括用于治疗与炎性细胞活化相关的疾病的方法。"Arthritis disease" as used herein refers to any disease characterized by arthritic lesions attributable to various etiologies. "Dermatitis" as used herein refers to any of a large family of skin diseases characterized by skin inflammation attributable to various etiologies. "Transplant rejection" as used herein refers to any immune response against transplanted tissue, such as organs or cells (eg, bone marrow), characterized by loss of function of the transplant or surrounding tissue, pain, swelling, leukocytosis, and thrombocytopenia. The therapeutic methods of the present invention include methods for treating diseases associated with inflammatory cell activation.
术语“癌症”和“癌的”是指或描述患者中通常以失控的细胞生长为特征的生理学病症。“肿瘤”包含一种或多种癌细胞。癌症的实例包括但不限于癌(carcinoma)、淋巴瘤、胚细胞瘤、肉瘤和白血病,或恶性淋巴增殖性疾病(lymphoid malignancies)。此类癌症的更具体的实例包括鳞状细胞癌(如上皮鳞状细胞癌)、肺癌(包括小细胞肺癌、非小细胞肺癌(NSCLC)、肺腺癌和肺鳞状癌)、腹膜癌、肝细胞癌(hepatocellular cancer)、胃癌(gastricor stomach cancer)(包括胃肠癌)、胰腺癌、恶性胶质瘤、宫颈癌、卵巢癌、肝癌(livercancer)、膀胱癌、肝细胞瘤(hepatoma)、乳腺癌、结肠癌、直肠癌、结直肠癌、子宫内膜癌或子宫癌、唾液腺癌、肾癌或肾脏癌(kidney or renal cancer)、前列腺癌、外阴癌、甲状腺癌、肝脏癌(hepatic carcinoma)、肛门癌、阴茎癌以及头颈癌。The terms "cancer" and "cancerous" refer to or describe the physiological condition in a patient that is often characterized by uncontrolled cell growth. A "tumor" includes one or more cancer cells. Examples of cancer include, but are not limited to, carcinoma, lymphoma, blastoma, sarcoma, and leukemia, or lymphoid malignancies. More specific examples of such cancers include squamous cell carcinoma (eg, epithelial squamous cell carcinoma), lung cancer (including small cell lung cancer, non-small cell lung cancer (NSCLC), lung adenocarcinoma, and lung squamous cell carcinoma), peritoneal carcinoma, hepatocellular cancer, gastric cancer (including gastrointestinal cancer), pancreatic cancer, glioblastoma, cervical cancer, ovarian cancer, liver cancer, bladder cancer, hepatoma, Breast, colon, rectal, colorectal, endometrial or uterine cancer, salivary gland cancer, kidney or renal cancer, prostate cancer, vulvar cancer, thyroid cancer, hepatic carcinoma ), anal cancer, penile cancer, and head and neck cancer.
另外,本发明公开的化合物、包括它们的盐,也可以以它们的水合物形式或包含其溶剂(例如乙醇、DMSO,等等)的形式得到,用于它们的结晶。本发明公开化合物可以与药学上可接受的溶剂(包括水)固有地或通过设计形成溶剂化物;因此,本发明旨在包括溶剂化的和未溶剂化的形式。In addition, the compounds disclosed herein, including their salts, can also be obtained in their hydrate form or in a form containing a solvent (eg, ethanol, DMSO, etc.) for their crystallization. Compounds of the present disclosure may form solvates, inherently or by design, with pharmaceutically acceptable solvents, including water; thus, the present invention is intended to include both solvated and unsolvated forms.
本发明给出的任何结构式也意欲表示这些化合物未被同位素富集的形式以及同位素富集的形式。同位素富集的化合物具有本发明给出的通式描绘的结构,除了一个或多个原子被具有所选择原子量或质量数的原子替换。可引入本发明化合物中的示例性同位素包括氢、碳、氮、氧、磷、硫、氟和氯的同位素,如2H, 3H,11C,13C,14C,15N,17O,18O,18F,31P,32P,35S,36Cl和125I。Any structural formula given herein is also intended to represent both isotopically unenriched as well as isotopically enriched forms of these compounds. Isotopically enriched compounds have the structures depicted by the general formulae given herein, except that one or more atoms are replaced by atoms having a selected atomic weight or mass number. Exemplary isotopes that can be incorporated into the compounds of the present invention include isotopes of hydrogen , carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, and chlorine, such as 2H, 3H , 11C , 13C , 14C , 15N , 17O , 18 O, 18 F, 31 P, 32 P, 35 S, 36 Cl and 125 I.
本发明化合物的药物组合物,制剂和给药PHARMACEUTICAL COMPOSITIONS, FORMULATIONS AND ADMINISTRATION OF THE COMPOUNDS OF THE INVENTION
本发明提供一种药物组合物,其包含本发明公开化合物和药学上可接受的赋形剂、载体、佐剂、溶媒或它们的组合。本发明公开的药物组合物中化合物的量是指能有效检测到抑制生物样本或患者体内蛋白激酶的量。The present invention provides a pharmaceutical composition comprising a compound disclosed herein and a pharmaceutically acceptable excipient, carrier, adjuvant, vehicle or a combination thereof. The amount of the compound in the pharmaceutical composition disclosed in the present invention refers to the amount that can effectively detect the inhibition of protein kinases in biological samples or patients.
也应认识到,本发明的某些化合物可以以游离形式存在用于治疗,或者如果适当可以以其药学上可接受的衍生物的形式存在。药学上可接受衍生物的一些非限制性的实施方案包括药学上可接受的前药,盐, 酯,这些酯的盐,或者对有需要的患者给药时能直接或间接提供本发明所述化合物或其代谢产物或残留物的任何另外的加合物或衍生物。It will also be recognized that certain compounds of the present invention may exist in free form for use in therapy or, if appropriate, in the form of their pharmaceutically acceptable derivatives. Some non-limiting embodiments of pharmaceutically acceptable derivatives include pharmaceutically acceptable prodrugs, salts, esters, salts of these esters, or can provide, directly or indirectly, the invention described herein when administered to a patient in need thereof. Any additional adducts or derivatives of the compound or its metabolites or residues.
本发明公开的药物药物组合物可制备并包装为散装(bulk)形式,其中可提取安全有效量的本发明所述化合物,然后以粉末或糖浆形式给予患者。或者,本发明公开的药物组合物可制备并包装为单位剂型,其中每个物理上离散的单位含有安全有效量的本发明所述的化合物。当以单位剂型制备时,本发明公开的药物组合物通常可含,例如,0.5mg至1g、或1mg至700mg、或5mg至100mg的本发明公开的化合物。The pharmaceutical compositions disclosed herein can be prepared and packaged in bulk form, wherein a safe and effective amount of the compounds of the present invention can be extracted and then administered to patients in powder or syrup form. Alternatively, the pharmaceutical compositions disclosed herein can be prepared and packaged in unit dosage form, wherein each physically discrete unit contains a safe and effective amount of a compound described herein. When prepared in unit dosage form, the presently disclosed pharmaceutical compositions may generally contain, for example, 0.5 mg to 1 g, or 1 mg to 700 mg, or 5 mg to 100 mg of the presently disclosed compound.
本发明所用“药学上可接受的赋形剂”意指与给药剂型或药物组合物一致性相关的药学上可接受的材料,混合物或溶媒。每种赋形剂在混合时必须与药物组合物的其它成分相容,以避免对患者给药时会大大降低本发明公开化合物的功效的相互作用和会导致不是药学上可接受的药物组合物的相互作用。此外,每种赋形剂必须是药学上可接受的,例如,具有足够高的纯度。"Pharmaceutically acceptable excipient" as used in the present invention means a pharmaceutically acceptable material, admixture or vehicle that is relevant to the consistency of the administered dosage form or pharmaceutical composition. Each excipient must, when mixed, be compatible with the other ingredients of the pharmaceutical composition to avoid interactions that would greatly reduce the efficacy of the disclosed compounds when administered to a patient and would result in a pharmaceutical composition that is not pharmaceutically acceptable Interaction. In addition, each excipient must be pharmaceutically acceptable, eg, of sufficiently high purity.
合适的药学上可接受的赋形剂包括以下类型的赋形剂:稀释剂、填充剂、粘合剂、崩解剂、润滑剂、助流剂、造粒剂、包衣剂、润湿剂、溶剂、共溶剂、助悬剂、乳化剂、甜味剂、矫味剂、掩味剂、着色剂、防结块剂、保湿剂、螯合剂、塑化剂、增粘剂、抗氧化剂、防腐剂、稳定剂、表面活性剂和缓冲剂。技术人员可认识到,某些药学上可接受的赋形剂可提供不止一种功能,并提供可供选择的功能,这取决于制剂中存在多少该赋形剂和制剂中存在那些其他赋形剂。Suitable pharmaceutically acceptable excipients include the following types of excipients: diluents, fillers, binders, disintegrants, lubricants, glidants, granulating agents, coating agents, wetting agents , solvent, co-solvent, suspending agent, emulsifier, sweetener, flavoring agent, taste masking agent, coloring agent, anti-caking agent, humectant, chelating agent, plasticizer, tackifier, antioxidant, Preservatives, stabilizers, surfactants and buffers. The skilled artisan will recognize that certain pharmaceutically acceptable excipients may serve more than one function, and provide alternative functions, depending on how much of that excipient is present in the formulation and those other excipients present in the formulation. agent.
示例性的药学可接受的载体或其组分是糖类,例如乳糖,葡萄糖和蔗糖;淀粉,例如玉米淀粉和马铃薯淀粉;纤维素及其衍生物,如羧甲基纤维素钠、乙基纤维素和甲基纤维素;西黄蓍胶粉;麦芽;明胶;滑石;固体润滑剂,例如硬脂酸和硬脂酸镁;硫酸钙;合成油;植物油,例如花生油,棉籽油,芝麻油,橄榄油和玉米油;多元醇,如丙二醇、甘油、山梨醇、甘露醇和聚乙二醇;藻酸;磷酸盐缓冲溶液;乳化剂,例如吐温类;润湿剂,例如十二烷基硫酸钠;着色剂;调味剂;压片剂;稳定剂;抗氧化剂;防腐剂;无热原水;等渗盐水;和磷酸盐缓冲溶液。Exemplary pharmaceutically acceptable carriers or components thereof are carbohydrates such as lactose, glucose and sucrose; starches such as corn starch and potato starch; cellulose and derivatives thereof such as sodium carboxymethyl cellulose, ethyl cellulose vegan and methylcellulose; tragacanth powder; malt; gelatin; talc; solid lubricants such as stearic acid and magnesium stearate; calcium sulfate; synthetic oils; vegetable oils such as peanut oil, cottonseed oil, sesame oil, olive oil Oils and corn oils; polyols such as propylene glycol, glycerol, sorbitol, mannitol and polyethylene glycol; alginic acid; phosphate buffered solutions; emulsifiers such as Tween; wetting agents such as sodium lauryl sulfate ; coloring agent; flavoring agent; compressed tablet; stabilizer; antioxidant; preservative; pyrogen-free water; isotonic saline; and phosphate buffered solution.
本发明所公开的化合物也可以与作为靶向药物载体的可溶性聚合物结合。这样的聚合物包括聚乙烯吡咯烷酮、吡喃共聚物、聚羟丙基甲基丙烯酰胺-苯酚、聚羟乙基天冬酰胺苯酚或棕榈酰残基取代的聚氧乙烯聚赖氨酸。此外,本发明所公开的化合物可以与在实现药物的控制释放中使用的一类生物可降解的聚合物结合,例如,聚乳酸、聚ε-己内酯、聚羟基丁酸、聚原酸酯、聚缩醛、聚二氢吡喃、聚氰基丙烯酸酯和水凝胶的交联或两亲嵌段共聚物。The disclosed compounds can also be combined with soluble polymers as targetable drug carriers. Such polymers include polyvinylpyrrolidone, pyran copolymers, polyhydroxypropylmethacrylamide-phenol, polyhydroxyethylaspartamidephenol, or polyoxyethylene polylysine substituted with palmitoyl residues. In addition, the compounds disclosed herein can be combined with a class of biodegradable polymers used in achieving controlled release of drugs, eg, polylactic acid, poly-epsilon-caprolactone, polyhydroxybutyric acid, polyorthoesters , crosslinked or amphiphilic block copolymers of polyacetal, polydihydropyran, polycyanoacrylate and hydrogel.
本发明提供的药物组合物可以与不会损害预期的治疗作用的其它活性成分共同配制,或者与补充预期的作用的物质共同配制。The pharmaceutical compositions provided by the present invention can be co-formulated with other active ingredients that do not impair the intended therapeutic effect, or with substances that supplement the intended effect.
合适的含水运载体包括,但不限于:水、盐水、生理盐水或磷酸盐缓冲盐水(PBS)、氯化钠注射液、Ringers注射液、等渗葡萄糖注射液、无菌水注射液、葡萄糖和乳酸化的Ringers注射液。非水运载体包括,但不限于,植物来源的非挥发油、蓖麻油、玉米油、棉籽油、橄榄油、花生油、薄荷油、红花油、芝麻油、豆油、氢化植物油、氢化豆油和椰子油的中链甘油三酯、及棕榈种子油。水混溶性运载体包括,但不限于,乙醇、1,3-丁二醇、液体聚乙二醇(例如聚乙二醇300和聚乙二醇400)、丙二醇、甘油、N-甲基-2-吡咯烷酮、N,N-二甲基乙酰胺和二甲亚砜。Suitable aqueous vehicles include, but are not limited to: water, saline, normal saline or phosphate buffered saline (PBS), Sodium Chloride Injection, Ringers Injection, Isotonic Dextrose Injection, Sterile Water Injection, Dextrose and Lactated Ringers Injection. Non-aqueous carriers include, but are not limited to, medium of vegetable derived fixed oils, castor oil, corn oil, cottonseed oil, olive oil, peanut oil, peppermint oil, safflower oil, sesame oil, soybean oil, hydrogenated vegetable oil, hydrogenated soybean oil and coconut oil. Chain triglycerides, and palm seed oil. Water-miscible carriers include, but are not limited to, ethanol, 1,3-butanediol, liquid polyethylene glycols (eg, polyethylene glycol 300 and polyethylene glycol 400), propylene glycol, glycerol, N-methyl- 2-pyrrolidone, N,N-dimethylacetamide and dimethylsulfoxide.
合适的抗微生物剂或防腐剂包括,但不限于,苯酚、甲酚、汞剂、苯甲醇、氯代丁醇、对羟基苯甲酸甲酯和对羟基苯甲酸丙酯、硫柳汞、苯扎氯铵(例如苄索氯铵)、尼泊金甲酯和尼泊金丙酯及山梨酸。合适的等渗剂包括,但不限于,氯化钠、甘油和葡萄糖。合适的缓冲剂包括,但不限于,磷酸盐和柠檬酸盐。合适的抗氧化剂为如本发明描述的那些,包括亚硫酸氢盐和偏亚硫酸氢钠。合适的局部麻醉剂包括,但不限于盐酸普鲁卡因。合适的助悬剂和分散剂为如本发明描述的那些,包括羧甲基纤维素钠、羟丙基甲基纤 维素和聚乙烯吡咯烷酮。合适的乳化剂包括本发明描述的那些,包括聚氧乙烯脱水山梨醇单月桂酸酯。聚氧乙烯脱水山梨醇单油酸酯80和油酸三乙醇胺酯。合适的多价螯合剂或螯合剂包括,但不限于EDTA。合适的pH调节剂包括,但不限于氢氧化钠、盐酸、柠檬酸和乳酸。合适的络合剂包括,但不限于环糊精,包括α-环糊精、β-环糊精、羟丙基-β-环糊精、磺基丁基醚-β-环糊精和磺基丁基醚7-β-环糊精(CyDex,Lenexa,KS)。Suitable antimicrobial agents or preservatives include, but are not limited to, phenol, cresol, amalgam, benzyl alcohol, chlorobutanol, methyl and propylparaben, thimerosal, benzalkonium chloride (eg benzethonium chloride), methyl and propyl parabens, and sorbic acid. Suitable isotonicity agents include, but are not limited to, sodium chloride, glycerol, and dextrose. Suitable buffers include, but are not limited to, phosphates and citrates. Suitable antioxidants are those as described herein, including bisulfite and sodium metabisulfite. Suitable local anesthetics include, but are not limited to, procaine hydrochloride. Suitable suspending and dispersing agents are those as described herein including sodium carboxymethyl cellulose, hydroxypropyl methyl cellulose and polyvinylpyrrolidone. Suitable emulsifiers include those described herein, including polyoxyethylene sorbitan monolaurate. Polyoxyethylene sorbitan monooleate 80 and triethanolamine oleate. Suitable sequestrants or chelating agents include, but are not limited to, EDTA. Suitable pH adjusting agents include, but are not limited to, sodium hydroxide, hydrochloric acid, citric acid, and lactic acid. Suitable complexing agents include, but are not limited to, cyclodextrins, including alpha-cyclodextrin, beta-cyclodextrin, hydroxypropyl-beta-cyclodextrin, sulfobutyl ether-beta-cyclodextrin and sulfonic acid butyl ether 7-β-cyclodextrin ( CyDex, Lenexa, KS).
本发明提供的药物组合物可以配制成单剂量或多剂量给药。所述单剂量制剂被包装在安瓿剂、小瓶或注射器中。所述多剂量肠胃外制剂必须包含抑菌或抑真菌浓度的抗微生物剂。所有的肠胃外制剂都必须是无菌的,如本领域已知和实践的。The pharmaceutical compositions provided by the present invention can be formulated for single-dose or multiple-dose administration. The single-dose formulation is packaged in ampoules, vials or syringes. The multiple-dose parenteral formulation must contain a bacteriostatic or fungistatic concentration of the antimicrobial agent. All parenteral formulations must be sterile, as known and practiced in the art.
用途use
本发明提供使用本发明所公开的化合物和药物组合物治疗、预防或改善由JAK激酶包括JAK1、JAK2、JAK3或TYK2激酶行为介导或以其他方式影响的疾病或紊乱或者由JAK激酶包括JAK1、JAK2、JAK3或TYK2激酶行为介导或以其他方式影响的疾病或紊乱的一种或多种症状的方法。The present invention provides the use of the compounds and pharmaceutical compositions disclosed herein for the treatment, prevention or amelioration of diseases or disorders mediated or otherwise affected by the behavior of JAK kinases including JAK1, JAK2, JAK3 or TYK2 kinases or by JAK kinases including JAK1, A method for one or more symptoms of a disease or disorder that is mediated or otherwise affected by the behavior of JAK2, JAK3 or TYK2 kinases.
JAK激酶可以是JAK1、JAK2、JAK3或TYK2激酶的野生型和/或突变。The JAK kinase can be wild-type and/or mutated of a JAK1, JAK2, JAK3 or TYK2 kinase.
在一实施方案中,本发明提供一类本发明所公开的化合物或包含本发明所公开化合物的药物组合物,用于治疗、预防或改善由不适当的JAK1激酶行为介导或以其他方式影响的疾病或紊乱或者由不适当的JAK1激酶行为介导或以其他方式影响的疾病或紊乱的一种或多种症状。In one embodiment, the present invention provides a class of compounds disclosed herein, or pharmaceutical compositions comprising compounds disclosed herein, for use in treating, preventing, or ameliorating effects mediated or otherwise affected by inappropriate JAK1 kinase behavior disease or disorder, or one or more symptoms of a disease or disorder mediated or otherwise affected by inappropriate JAK1 kinase behavior.
在另一实施方案中,所述疾病、紊乱或者疾病或紊乱的一种或多种症状与不适当的JAK2激酶行为相关。In another embodiment, the disease, disorder, or one or more symptoms of a disease or disorder is associated with inappropriate JAK2 kinase behavior.
还在一实施方案中,所述疾病、紊乱或者疾病或紊乱的一种或多种症状与不适当的JAK3激酶行为相关。In yet another embodiment, the disease, disorder, or one or more symptoms of a disease or disorder is associated with inappropriate JAK3 kinase behavior.
“不适当的JAK激酶行为”是指发生在特定患者身上偏离正常JAK激酶行为的JAK激酶行为。不适当的JAK激酶行为可以表现为例如活性的不正常增长、或JAK激酶行为时间点和控制上的偏差的形式。这种不适当的激酶行为源于,例如,蛋白激酶的过度表达或突变而导致的不适当或不受控的行为。因此,本发明提供治疗这些疾病和紊乱的方法。"Inappropriate JAK kinase behavior" refers to JAK kinase behavior that deviates from normal JAK kinase behavior that occurs in a particular patient. Inappropriate JAK kinase behavior can take the form of, for example, an abnormal increase in activity, or deviations in timing and control of JAK kinase behavior. Such inappropriate kinase behavior results from, for example, inappropriate or uncontrolled behavior caused by overexpression or mutation of protein kinases. Accordingly, the present invention provides methods of treating these diseases and disorders.
同上面的描述相一致,这样的疾病或紊乱包括但不限于:骨髓增殖性疾病,例如真性红细胞增多症(PCV)、特发性血小板增多症、特发性骨髓纤维化(IMF);白血病,例如髓系白血病包括慢性髓系白血病(CML)、耐伊马替尼的CML形式、急性髓系白血病(AML)和AML的亚型、急性成巨核细胞白血病(AMKL);淋巴增殖性疾病,例如骨髓瘤;癌症包括头颈部癌、前列腺癌乳癌、卵巢癌、黑素瘤、肺癌、脑肿瘤、胰腺癌和肾癌;和与免疫功能紊乱、免疫缺陷、免疫调节有关的炎症性疾病或紊乱、自身免疫性疾病、组织移植排斥、移植物抗宿主病、伤口愈合、肾病、多发性硬化、甲状腺炎、I型糖尿病、结节病、银屑病、变应性鼻炎、炎症性肠病包括克罗恩病和溃疡性结肠炎(UC)、系统性红斑狼疮(SLE)、关节炎、骨关节炎、类风湿性关节炎、骨质疏松症、哮喘和慢性阻塞性肺病(COPD)和干眼综合征(或干燥性角膜结膜炎(KCS))。Consistent with the description above, such diseases or disorders include, but are not limited to: myeloproliferative diseases such as polycythemia vera (PCV), essential thrombocythemia, idiopathic myelofibrosis (IMF); leukemia, For example, myeloid leukemias include chronic myeloid leukemia (CML), imatinib-resistant forms of CML, acute myeloid leukemia (AML) and subtypes of AML, acute megakaryoblastic leukemia (AMKL); lymphoproliferative disorders such as Myeloma; cancers including head and neck, prostate, breast, ovarian, melanoma, lung, brain, pancreatic, and kidney cancers; and inflammatory diseases or disorders associated with immune dysfunction, immune deficiency, immune regulation , autoimmune disease, tissue transplant rejection, graft-versus-host disease, wound healing, kidney disease, multiple sclerosis, thyroiditis, type I diabetes, sarcoidosis, psoriasis, allergic rhinitis, inflammatory bowel disease including Crohn's disease and ulcerative colitis (UC), systemic lupus erythematosus (SLE), arthritis, osteoarthritis, rheumatoid arthritis, osteoporosis, asthma and chronic obstructive pulmonary disease (COPD) and dry Ocular syndrome (or keratoconjunctivitis sicca (KCS)).
一方面,本发明提供一类本发明所公开的化合物或包含本发明所公开化合物的药物组合物,用于预防和/或治疗哺乳动物(包括人类)的增殖性疾病、自体免疫疾病、过敏性疾病、炎性疾病、或移植排斥。In one aspect, the present invention provides a class of compounds disclosed in the present invention or pharmaceutical compositions comprising the compounds disclosed in the present invention for preventing and/or treating proliferative diseases, autoimmune diseases, allergic diseases in mammals (including humans) Disease, inflammatory disease, or transplant rejection.
在另一方面,本发明提供一种治疗罹患或有风险罹患本文所公开疾病的哺乳动物的方法,所述方法包括给予有效治疗病症量或有效预防病症量的一种或多种本文公开的药物组合物或化合物。在另一方面,本文提供一种治疗罹患或有风险罹患增殖性疾病、自体免疫疾病、过敏性疾病、炎性疾病或移植排斥的哺乳动物的方法。In another aspect, the present invention provides a method of treating a mammal suffering from or at risk of suffering from a disorder disclosed herein, the method comprising administering an amount effective to treat a disorder or an amount effective to prevent a disorder of one or more of the agents disclosed herein composition or compound. In another aspect, provided herein is a method of treating a mammal suffering from or at risk of suffering from a proliferative disease, autoimmune disease, allergic disease, inflammatory disease, or transplant rejection.
在治疗方面的一种方法中,本发明提供治疗和/或预防易患或患有增殖性疾病的哺乳动物的方法,所述 方法包括给予有效治疗量或有效预防量的一种或多种本文公开的药物组合物或化合物。在特定实例中,增殖性疾病选自癌症(例如,实体瘤例如子宫平滑肌肉瘤或前列腺癌)、真性红细胞增多症、原发性血小板增多症、骨髓纤维化、白血病(例如,AML、CML、ALL或CLL)和多发性骨髓瘤。In one method of therapy, the present invention provides a method of treating and/or preventing a mammal susceptible to or suffering from a proliferative disease, the method comprising administering a therapeutically effective or prophylactically effective amount of one or more of the herein Disclosed pharmaceutical compositions or compounds. In specific examples, the proliferative disease is selected from cancer (eg, solid tumors such as uterine leiomyosarcoma or prostate cancer), polycythemia vera, essential thrombocythemia, myelofibrosis, leukemia (eg, AML, CML, ALL) or CLL) and multiple myeloma.
另一方面,本文提供一类本文公开的化合物,用于治疗和/或预防增殖性疾病。在特定的实施方案中,增殖性疾病选自癌症(例如,实体瘤例如子宫平滑肌肉瘤或前列腺癌)、真性红细胞增多症、原发性血小板增多症、骨髓纤维化、白血病(例如,AML、CML、ALL或CLL)和多发性骨髓瘤。In another aspect, provided herein is a class of compounds disclosed herein for use in the treatment and/or prevention of proliferative diseases. In specific embodiments, the proliferative disease is selected from cancer (eg, solid tumors such as uterine leiomyosarcoma or prostate cancer), polycythemia vera, essential thrombocythemia, myelofibrosis, leukemia (eg, AML, CML) , ALL or CLL) and multiple myeloma.
另一方面,本文提供一类本文公开的化合物,或包含本文公开化合物的药物组合物,用于制备治疗或预防增殖性疾病的药品。在特定实例中,增殖性疾病选自癌症(例如,实体瘤例如子宫平滑肌肉瘤或前列腺癌)、真性红细胞增多症、原发性血小板增多症、骨髓纤维化、白血病(例如,AML、CML、ALL或CLL)和多发性骨髓瘤。In another aspect, provided herein is a class of compounds disclosed herein, or pharmaceutical compositions comprising the compounds disclosed herein, for use in the manufacture of a medicament for the treatment or prevention of proliferative diseases. In specific examples, the proliferative disease is selected from cancer (eg, solid tumors such as uterine leiomyosarcoma or prostate cancer), polycythemia vera, essential thrombocythemia, myelofibrosis, leukemia (eg, AML, CML, ALL) or CLL) and multiple myeloma.
在另一方面,本文提供治疗和/或预防易患或患有自体免疫疾病的哺乳动物的方法,所述方法包括给予有效治疗量或有效预防量的一种或多种本文公开的药物组合物或化合物。在特定实例中,自体免疫疾病选自COPD、哮喘、系统性红斑狼疮、皮肤型红斑狼疮、狼疮性肾炎、皮肌炎、干燥综合征、银屑病、I型糖尿病和炎性肠病。In another aspect, provided herein is a method of treating and/or preventing a mammal susceptible to or suffering from an autoimmune disease, the method comprising administering a therapeutically effective or prophylactically effective amount of one or more of the pharmaceutical compositions disclosed herein or compound. In specific examples, the autoimmune disease is selected from the group consisting of COPD, asthma, systemic lupus erythematosus, cutaneous lupus erythematosus, lupus nephritis, dermatomyositis, Sjögren's syndrome, psoriasis, type I diabetes, and inflammatory bowel disease.
另一方面,本文提供一类本文公开的化合物,用于治疗和/或预防自体免疫疾病。在特定的实施方案中,自体免疫疾病选自COPD、哮喘、系统性红斑狼疮、皮肤型红斑狼疮、狼疮性肾炎、皮肌炎、干燥综合征、银屑病、I型糖尿病和炎性肠病。In another aspect, provided herein is a class of compounds disclosed herein for use in the treatment and/or prevention of autoimmune diseases. In specific embodiments, the autoimmune disease is selected from the group consisting of COPD, asthma, systemic lupus erythematosus, cutaneous lupus erythematosus, lupus nephritis, dermatomyositis, Sjögren's syndrome, psoriasis, type I diabetes and inflammatory bowel disease .
另一方面,本文提供一类本文公开的化合物,或包含本文公开化合物的药物组合物,用于制备治疗或预防自体免疫疾病的药品。在特定的实施方案中,自体免疫疾病选自COPD、哮喘、系统性红斑狼疮、皮肤型红斑狼疮、狼疮性肾炎、皮肌炎、干燥综合征、银屑病、I型糖尿病和炎性肠病。In another aspect, provided herein is a class of compounds disclosed herein, or pharmaceutical compositions comprising the compounds disclosed herein, for use in the manufacture of a medicament for the treatment or prevention of autoimmune diseases. In specific embodiments, the autoimmune disease is selected from the group consisting of COPD, asthma, systemic lupus erythematosus, cutaneous lupus erythematosus, lupus nephritis, dermatomyositis, Sjögren's syndrome, psoriasis, type I diabetes and inflammatory bowel disease .
在另一方面,本文提供治疗和/或预防易患或患有过敏性疾病的哺乳动物的方法,所述方法包括给予有效治疗量或有效预防量的一种或多种本文公开的药物组合物或化合物。在特定的实施方案中,过敏性疾病选自呼吸道过敏性疾病、鼻窦炎、湿疹和麻疹、食物过敏和昆虫毒液过敏。In another aspect, provided herein are methods of treating and/or preventing mammals susceptible to or suffering from allergic disease, the methods comprising administering a therapeutically effective or prophylactically effective amount of one or more of the pharmaceutical compositions disclosed herein or compound. In particular embodiments, the allergic disease is selected from the group consisting of respiratory allergic disease, sinusitis, eczema and measles, food allergy and insect venom allergy.
另一方面,本文提供一类本文公开的化合物,用于治疗和/或预防过敏性疾病。在特定的实施方案中,过敏性疾病选自呼吸道过敏性疾病、鼻窦炎、湿疹和麻疹、食物过敏和昆虫毒液过敏。In another aspect, provided herein is a class of compounds disclosed herein for use in the treatment and/or prevention of allergic diseases. In particular embodiments, the allergic disease is selected from the group consisting of respiratory allergic disease, sinusitis, eczema and measles, food allergy and insect venom allergy.
另一方面,本文提供一类本文公开的化合物,或包含本文公开化合物的药物组合物,用于制备治疗或预防过敏性疾病的药品。在特定的实施方案中,过敏性疾病选自呼吸道过敏性疾病、鼻窦炎、湿疹和麻疹,食物过敏和昆虫毒液过敏。In another aspect, provided herein is a class of compounds disclosed herein, or pharmaceutical compositions comprising the compounds disclosed herein, for use in the manufacture of a medicament for the treatment or prevention of allergic diseases. In particular embodiments, the allergic disease is selected from the group consisting of respiratory allergic disease, sinusitis, eczema and measles, food allergy and insect venom allergy.
另一方面,本文提供一类本文公开的化合物,用于治疗和/或预防炎性疾病。在特定的实施方案中,炎性疾病选自炎性肠病、克罗恩病、类风湿性关节炎、幼年型关节炎和银屑病性关节炎。In another aspect, provided herein is a class of compounds disclosed herein for use in the treatment and/or prevention of inflammatory diseases. In specific embodiments, the inflammatory disease is selected from the group consisting of inflammatory bowel disease, Crohn's disease, rheumatoid arthritis, juvenile arthritis, and psoriatic arthritis.
另一方面,本文提供一类本文公开的化合物,或包含本文公开化合物的药物组合物,用于制备治疗或预防炎性疾病的药品。在特定的实施方案中,炎性疾病选自炎性肠病、克罗恩病、类风湿性关节炎、幼年型关节炎和银屑病性关节炎。In another aspect, provided herein is a class of compounds disclosed herein, or pharmaceutical compositions comprising the compounds disclosed herein, for use in the manufacture of a medicament for the treatment or prevention of inflammatory diseases. In specific embodiments, the inflammatory disease is selected from the group consisting of inflammatory bowel disease, Crohn's disease, rheumatoid arthritis, juvenile arthritis, and psoriatic arthritis.
另一方面,本文提供一类本文公开的化合物,用于治疗和/或预防移植排斥。在特定的实施方案中,移植排斥是器官移植排斥、组织移植排斥和细胞移植排斥。In another aspect, provided herein is a class of compounds disclosed herein for use in the treatment and/or prevention of transplant rejection. In specific embodiments, transplant rejection is organ transplant rejection, tissue transplant rejection, and cell transplant rejection.
另一方面,本文提供一类本文公开的化合物,或包含本文公开化合物的药物组合物,用于制备治疗或预防移植排斥的药品。在特定实例中,移植排斥是器官移植排斥、组织移植排斥和细胞移植排斥。In another aspect, provided herein is a class of compounds disclosed herein, or pharmaceutical compositions comprising the compounds disclosed herein, for use in the manufacture of a medicament for the treatment or prevention of transplant rejection. In specific examples, transplant rejection is organ transplant rejection, tissue transplant rejection, and cell transplant rejection.
在另一方面,本文提供一类用作药物尤其用作治疗和/或预防前面所述疾病药物的本文公开的化合物。也提供使用本文公开化合物制造治疗和/或预防前面所述疾病的药物。In another aspect, provided herein is a class of compounds disclosed herein for use as a medicament, particularly as a medicament for the treatment and/or prevention of the aforementioned diseases. Also provided is the use of the compounds disclosed herein for the manufacture of medicaments for the treatment and/or prevention of the aforementioned diseases.
本方法的一个特别方案包括给予患有炎症的受试对象有效量的本发明公开化合物一段时间,所述时间 足以降低受试对象的炎症水平,并且优选终止所述炎症的进程。该方法的特别实施方案包括给予患有或易患骨类风湿性关节炎的受试患者有效量的本发明公开化合物一段时间,所述时间足以分别降低或预防所述患者的关节炎症,并且优选终止所述炎症的进程。A particular aspect of the method comprises administering to a subject suffering from inflammation an effective amount of a compound of the present disclosure for a period of time sufficient to reduce the level of inflammation in the subject, and preferably to terminate the progression of said inflammation. Particular embodiments of this method comprise administering to a subject suffering from or susceptible to rheumatoid arthritis of the bone an effective amount of a compound of the present disclosure for a period of time sufficient to reduce or prevent, respectively, joint inflammation in said patient, and preferably The process of the inflammation is terminated.
本方法的另一个特别方案包括给予患有增殖性疾病的受试对象有效量的本发明公开化合物一段时间,所述时间足以降低受试对象的增殖疾病水平,并且优选终止所述增殖性疾病的进程。该方法的特别实施方案包括给予患有癌症的受试患者有效量的本文公开化合物一段时间,所述时间足以分别降低或预防所述患者的癌症病征,并且优选终止所述癌症的进程。Another particular embodiment of this method comprises administering to a subject suffering from a proliferative disease an effective amount of a compound of the present disclosure for a period of time sufficient to reduce the level of the proliferative disease in the subject, and preferably to terminate the proliferation of the proliferative disease. process. Particular embodiments of this method comprise administering to a subject suffering from cancer an effective amount of a compound disclosed herein for a period of time sufficient to reduce or prevent, respectively, the symptoms of cancer in said patient, and preferably to terminate the progression of said cancer.
本发明所公开的化合物和药物组合物是激酶抑制剂,包含Btk抑制剂。这些抑制剂可以用于治疗哺乳动物中的一种或多种响应激酶抑制的疾病,包括响应Btk抑制和/或B-细胞增殖抑制的疾病。不希望束缚于任何特定的理论,相信本发明化合物与Btk的相互作用导致Btk活性的抑制,并因此得到这些化合物的药学应用。因此,本发明包括用于治疗具有响应Btk活性的抑制和/或抑制B-细胞增殖的疾病的哺乳动物,例如人的方法,该方法包括:向具有这样的疾病的哺乳动物给药有效量的至少一种在本文中提供的化学实体。可以在实验上例如通过测定化合物的血液浓度,或理论上通过计算生物利用度,确定有效浓度。除了Btk之外,还可能受到影响的其它激酶包括但不限于,其它酪氨酸激酶和丝氨酸/苏氨酸激酶。The compounds and pharmaceutical compositions disclosed herein are kinase inhibitors, including Btk inhibitors. These inhibitors can be used to treat one or more diseases in mammals responsive to kinase inhibition, including diseases responsive to Btk inhibition and/or B-cell proliferation inhibition. Without wishing to be bound by any particular theory, it is believed that the interaction of the compounds of the present invention with Btk results in the inhibition of Btk activity and, therefore, the pharmaceutical use of these compounds. Accordingly, the present invention includes a method for treating a mammal, eg, a human, having a disease responsive to inhibition of Btk activity and/or inhibition of B-cell proliferation, the method comprising: administering to the mammal having such a disease an effective amount of At least one chemical entity provided herein. Effective concentrations can be determined experimentally, eg, by measuring blood concentrations of the compound, or theoretically by calculating bioavailability. In addition to Btk, other kinases that may be affected include, but are not limited to, other tyrosine kinases and serine/threonine kinases.
在Btk与疾病有关的范围内,疾病和疾病症状的减轻、预防法和预防性治疗都在本发明的范围内。To the extent Btk is associated with disease, alleviation, prophylaxis and prophylactic treatment of disease and disease symptoms are within the scope of the present invention.
在一些实施方案中,应答Btk活性和/或B细胞和/或髓样细胞活性抑制的病症是癌症、骨病、变态反应性疾病和/或自身免疫和/或炎性疾病和/或急性炎症反应。In some embodiments, the disorder responsive to inhibition of Btk activity and/or B cell and/or myeloid cell activity is cancer, bone disease, allergic disease and/or autoimmune and/or inflammatory disease and/or acute inflammation reaction.
本发明包括治疗患有癌症、骨病、变态反应性疾病和/或自身免疫和/或炎性疾病和/或急性炎症反应的患者的方法,其通过给药有效量的本发明化合物及其药学可接受的盐、溶剂合物和混合物。The present invention includes methods of treating patients suffering from cancer, bone disease, allergic disease and/or autoimmune and/or inflammatory disease and/or acute inflammatory response by administering an effective amount of a compound of the present invention and the pharmacy thereof Acceptable salts, solvates and mixtures.
在一些实施方案中,使用本发明化合物可以影响的病症和疾病包括但不限于:In some embodiments, conditions and diseases that can be affected by the use of the compounds of the present invention include, but are not limited to:
变态反应性疾病,包括但不限于湿疹、变应性鼻炎或鼻炎、花粉症、支气管哮喘、荨麻疹(寻麻疹)和食物过敏和其它特应性病症;Allergic diseases, including but not limited to eczema, allergic rhinitis or rhinitis, hay fever, bronchial asthma, urticaria (hives) and food allergies and other atopic conditions;
自身免疫和/或炎性疾病,包括但不限于银屑病、克罗恩病、肠易激综合症、干燥综合征、组织移植排斥反应、以及移植器官的超急性排斥反应、哮喘、系统性红斑狼疮(和相关的肾小球肾炎)、皮肌炎、多发性硬化、硬皮病、血管炎(ANCA相关和其它血管炎)、自身免疫溶血性疾病及血小板减少性状态、肺出血肾炎综合征(和相关的肾小球肾炎和肺出血)、动脉粥样硬化、类风湿关节炎、骨关节炎、慢性特发性血小板减少性紫癜(ITP)、艾迪生病、帕金森病、阿尔茨海默病、糖尿病(1型)、感染性休克、重症肌无力、溃疡性结肠炎、再生障碍性贫血、Coeliac病、韦格纳肉芽肿、和其中细胞和抗体由个体的自身组织引起并直接对抗个体的自身组织的其它疾病;Autoimmune and/or inflammatory diseases, including but not limited to psoriasis, Crohn's disease, irritable bowel syndrome, Sjögren's syndrome, tissue transplant rejection, and hyperacute rejection of transplanted organs, asthma, systemic Lupus erythematosus (and related glomerulonephritis), dermatomyositis, multiple sclerosis, scleroderma, vasculitis (ANCA-related and other vasculitis), autoimmune hemolytic diseases and thrombocytopenic states, pulmonary hemorrhagic nephritis complex symptoms (and associated glomerulonephritis and pulmonary hemorrhage), atherosclerosis, rheumatoid arthritis, osteoarthritis, chronic idiopathic thrombocytopenic purpura (ITP), Addison's disease, Parkinson's disease, Alzheimer's disease Haimer's disease, diabetes (type 1), septic shock, myasthenia gravis, ulcerative colitis, aplastic anemia, Coeliac's disease, Wegener's granulomatosis, and where cells and antibodies arise from the individual's own tissues and directly against other diseases of the individual's own tissues;
急性炎症反应,包括但不限于皮肤晒伤、盆腔炎症性疾病、炎性肠病、尿道炎、葡萄膜炎、鼻窦炎、肺炎、脑炎、脑膜炎、心肌炎、肾炎、骨髓炎、肌炎、肝炎、胃炎、肠炎、皮炎、龈炎、阑尾炎、胰腺炎和胆囊炎;Acute inflammatory reactions, including but not limited to skin sunburn, pelvic inflammatory disease, inflammatory bowel disease, urethritis, uveitis, sinusitis, pneumonia, encephalitis, meningitis, myocarditis, nephritis, osteomyelitis, myositis, Hepatitis, gastritis, enteritis, dermatitis, gingivitis, appendicitis, pancreatitis and cholecystitis;
癌症,包括但不限于恶性血液病如B细胞淋巴瘤、和急性成淋巴细胞性白血病、急性髓性白血病、慢性髓性白血病、慢性和急性淋巴细胞白血病、多毛细胞白血病、霍奇金病、非霍奇金淋巴瘤、多发性骨髓瘤、和其他以血液或淋巴系统的癌症为特征的疾病;和Cancer, including but not limited to hematological malignancies such as B-cell lymphoma, and acute lymphoblastic leukemia, acute myeloid leukemia, chronic myeloid leukemia, chronic and acute lymphocytic leukemia, hairy cell leukemia, Hodgkin's disease, non- Hodgkin lymphoma, multiple myeloma, and other diseases characterized by cancers of the blood or lymphatic system; and
骨病,包括但不限于骨质疏松。Bone disease, including but not limited to osteoporosis.
Btk是已知的淋巴瘤B细胞凋亡的抑制剂。缺陷凋亡有助于人白血病和淋巴瘤的发病和抗药性。因此,还提供促进或诱导表达Btk的细胞凋亡的方法,其包括使所述细胞与本发明化合物及其药学可接受的盐、溶剂合物和混合物接触。Btk is a known inhibitor of lymphoma B cell apoptosis. Defective apoptosis contributes to the pathogenesis and drug resistance of human leukemias and lymphomas. Accordingly, also provided are methods of promoting or inducing apoptosis in a Btk-expressing cell comprising contacting the cell with a compound of the present invention and pharmaceutically acceptable salts, solvates and mixtures thereof.
本发明公开化合物可以与一种或多种其它治疗剂同时,或在其之前或之后给药。本发明化合物可以与 其他治疗剂通过相同或不同给药途径分别给药,或与之以同以药物组合物形式给药。The disclosed compounds may be administered concurrently with, prior to or subsequent to one or more other therapeutic agents. The compounds of the present invention may be administered separately from the other therapeutic agents by the same or different route of administration, or in the same pharmaceutical composition.
对于约50-70kg的个体,本发明公开药物组合物和联合物可以是含有约1-1000mg、或约1-500mg、或约1-250mg、或约1-150mg、或约0.5-100mg、或约1-50mg活性成分的单位剂量形式。化合物、药物组合物或其联合物的治疗有效量是取决于个体的物种、体重、年龄及个体情况、被治疗的疾病(disorder)或疾病(disease)或其严重程度。具备常用技能的医师、临床医师或兽医可以容易决定预防、治疗或抑制疾病(disorder)或疾病(disease)发展过程中所需各活性成分的有效量。For an individual of about 50-70 kg, the presently disclosed pharmaceutical compositions and combinations may contain about 1-1000 mg, or about 1-500 mg, or about 1-250 mg, or about 1-150 mg, or about 0.5-100 mg, or A unit dosage form of about 1-50 mg of active ingredient. A therapeutically effective amount of a compound, pharmaceutical composition or combination thereof will depend on the species, body weight, age and individual condition of the individual, the disorder or disease being treated or its severity. A physician, clinician or veterinarian of ordinary skill can readily determine the effective amount of each active ingredient required to prevent, treat or inhibit the progression of a disorder or disease.
以上所引用的剂量特性已在采用有利的哺乳动物(例如小鼠、大鼠、狗、猴)或其离体器官、组织及标本的体外及体内试验中证实。本发明公开化合物以溶液,例如水溶液形式在体外使用,也可以例如悬浮液或水溶液形式在体内的肠内,胃肠外,尤其是静脉内使用。The dosage characteristics cited above have been demonstrated in in vitro and in vivo tests using advantageous mammals (eg, mice, rats, dogs, monkeys) or isolated organs, tissues and specimens thereof. The disclosed compounds are used in vitro as solutions, eg, aqueous solutions, and enterally, parenterally, especially intravenously, in vivo, eg, as suspensions or aqueous solutions.
在一实施方案中,本发明公开化合物的治疗有效剂量为每天约0.1mg至约2,000mg。其药物组合物应当提供约0.1mg至约2,000mg剂量的该化合物。在一特定实施方案中,制备的药物剂量单位形式能提供约1mg至约2,000mg,约10mg至约1,000mg,约20mg至约500mg,或约25mg至约250mg的主要活性成分或每剂量单位形式中各主要成分的组合。在一特定实施方案中,制备的药物剂量单位形式能提供约10mg,20mg,25mg,50mg,100mg,250mg,500mg,1000mg或2000mg主要活性成分。In one embodiment, the therapeutically effective dose of the compounds disclosed herein is from about 0.1 mg to about 2,000 mg per day. Pharmaceutical compositions thereof should provide a dose of about 0.1 mg to about 2,000 mg of the compound. In a specific embodiment, pharmaceutical dosage unit forms are prepared to provide from about 1 mg to about 2,000 mg, from about 10 mg to about 1,000 mg, from about 20 mg to about 500 mg, or from about 25 mg to about 250 mg of the primary active ingredient or per dosage unit form combination of the main components. In a particular embodiment, a pharmaceutical dosage unit form is prepared to provide about 10 mg, 20 mg, 25 mg, 50 mg, 100 mg, 250 mg, 500 mg, 1000 mg or 2000 mg of the primary active ingredient.
此外,本发明公开的化合物可以以前药形式给药。在本发明中,本发明公开化合物的“前药”是对患者给药时,最终能在体内释放出本发明公开化合物的功能性衍生物。以前药形式给予本发明公开的化合物时,本领域技术人员可实施下列方式中的一种及以上:(a)变更化合物的体内起效时间;(b)变更化合物的体内作用持续时间;(c)变更化合物的体内输送或分布;(d)变更化合物的体内溶解度;及(e)克服化合物所面临的副作用或其他难点。用于制备前药的典型的功能性衍生物,包含在体内以化学方式或酶的方式裂解的化合物的变体。包含制备磷酸盐、酰胺、酯、硫代酯、碳酸盐及氨基甲酸盐的这些变体对本领域技术人员来讲是众所周知的。In addition, the compounds disclosed herein can be administered in prodrug form. In the present invention, "prodrugs" of the compounds disclosed in the present invention are functional derivatives that can eventually release the compounds disclosed in the present invention in vivo when administered to a patient. When administering the compounds disclosed herein in prodrug form, those skilled in the art can implement one or more of the following methods: (a) altering the in vivo onset time of the compound; (b) altering the in vivo duration of action of the compound; (c) ) altering the in vivo delivery or distribution of the compound; (d) altering the in vivo solubility of the compound; and (e) overcoming side effects or other difficulties faced by the compound. Typical functional derivatives used to prepare prodrugs include variants of compounds that are chemically or enzymatically cleaved in vivo. These variants, including the preparation of phosphates, amides, esters, thioesters, carbonates and carbamates, are well known to those skilled in the art.
一般合成过程General synthetic procedure
一般地,本发明的化合物可以通过本发明所描述的方法制备得到,除非有进一步的说明,其中取代基的定义如式(I)、式(Ia)、式(Ib)、式(Ic)、式(II)、式(IIa)或式(III)所示。下面的反应方案和实施例1-100用于进一步举例说明本发明的内容。Generally, the compounds of the present invention can be prepared by the methods described in the present invention, unless otherwise specified, wherein the substituents are as defined in formula (I), formula (Ia), formula (Ib), formula (Ic), Formula (II), formula (IIa) or formula (III). The following reaction schemes and Examples 1-100 serve to further illustrate the present invention.
所属领域的技术人员将认识到:本发明所描述的化学反应可以用来合适地制备许多本发明的其他化合物,且用于制备本发明的化合物的其它方法都被认为是在本发明的范围之内。例如,根据本发明那些非例证的化合物的合成可以成功地被所属领域的技术人员通过修饰方法完成,如适当的保护干扰基团,通过利用其他已知的试剂除了本发明所描述的,或将反应条件做一些常规的修改。另外,本发明所公开的反应或已知的反应条件也公认地适用于本发明其他化合物的制备。Those skilled in the art will recognize that the chemical reactions described herein can be used to suitably prepare many other compounds of the present invention, and that other methods for preparing the compounds of the present invention are considered to be within the scope of the present invention Inside. For example, the synthesis of those non-exemplified compounds according to the present invention can be successfully accomplished by those skilled in the art by modifying methods, such as appropriate protection of interfering groups, by using other known reagents in addition to those described herein, or by combining The reaction conditions were modified routinely. In addition, the reactions disclosed herein or known reaction conditions are also acknowledged to be applicable to the preparation of other compounds of the present invention.
下面所描述的实施例,除非其他方面表明所有的温度定为摄氏度。试剂购买于商品供应商如Aldrich Chemical Company,Arco Chemical Company and Alfa ChemicalCompany,使用时都没有经过进一步纯化,除非其他方面表明。一般的试剂从汕头西陇化工厂,广东光华化学试剂厂,广州化学试剂厂,天津好寓宇化学品有限公司,青岛腾龙化学试剂有限公司和青岛海洋化工厂购买得到。In the examples described below, all temperatures are in degrees Celsius unless otherwise indicated. Reagents were purchased from commercial suppliers such as Aldrich Chemical Company, Arco Chemical Company and Alfa Chemical Company and were used without further purification unless otherwise indicated. Common reagents were purchased from Shantou Xilong Chemical Reagent Factory, Guangdong Guanghua Chemical Reagent Factory, Guangzhou Chemical Reagent Factory, Tianjin Haoyuyu Chemical Co., Ltd., Qingdao Tenglong Chemical Reagent Co., Ltd. and Qingdao Ocean Chemical Factory.
无水四氢呋喃,二氧六环,甲苯,乙醚是经过金属钠回流干燥得到。无水二氯甲烷和氯仿是经过氢化钙回流干燥得到。乙酸乙酯,石油醚,正己烷,N,N-二甲基乙酰胺和N,N-二甲基甲酰胺是经无水硫酸钠事先干燥使用。Anhydrous tetrahydrofuran, dioxane, toluene and diethyl ether were obtained by refluxing and drying with metallic sodium. Anhydrous dichloromethane and chloroform were obtained by refluxing with calcium hydride. Ethyl acetate, petroleum ether, n-hexane, N,N-dimethylacetamide and N,N-dimethylformamide were dried over anhydrous sodium sulfate before use.
以下反应一般是在氮气或氩气正压下或在无水溶剂上套一干燥管(除非其他方面表明),反应瓶都塞上合适的橡皮塞,底物通过注射器打入。玻璃器皿都是干燥过的。The following reactions are generally carried out under positive nitrogen or argon pressure or a drying tube is set over anhydrous solvent (unless otherwise indicated), the reaction flasks are plugged with suitable rubber stoppers, and the substrate is injected through a syringe. Glassware is dry.
色谱柱是使用硅胶柱。硅胶(300-400目)购于青岛海洋化工厂。核磁共振氢谱的测试条件是:室温 条件下,布鲁克(Bruker)400MHz或600MHz的核磁仪,以CDC13,d6-DMSO,CD3OD或d6-丙酮为溶剂(报导以ppm为单位),用TMS(0ppm)或氯仿(7.26ppm)作为参照标准。当出现多重峰的时候,将使用下面的缩写:s(singlet,单峰),d(doublet,双峰),t(triplet,三重峰),q(quartet,四重峰),m(multiplet,多重峰),br(broadened,宽峰),dd(doublet of doublets,双二重峰),ddd(doublet of doublet of doublets,双二倍二重峰),dt(doublet of triplets,双三重峰)。偶合常数,用赫兹(Hz)表示。The chromatographic column is a silica gel column. Silica gel (300-400 mesh) was purchased from Qingdao Ocean Chemical Factory. The test conditions for NMR spectroscopy are: Bruker 400MHz or 600MHz NMR instrument at room temperature, with CDC1 3 , d 6 -DMSO, CD 3 OD or d 6 -acetone as solvent (reported in ppm) , with TMS (0ppm) or chloroform (7.26ppm) as the reference standard. When multiplets are present, the following abbreviations are used: s (singlet, singlet), d (doublet, doublet), t (triplet, triplet), q (quartet, quartet), m (multiplet, multiplet), br (broadened, broad peak), dd (doublet of doublets, double doublet), ddd (doublet of doublet of doublets, double doublet doublet), dt (doublet of triplets, double triplet) . Coupling constant, expressed in Hertz (Hz).
低分辨率质谱(MS)数据测定的条件是:Agilent 6120Quadrupole HPLC-MS(柱子型号:Zorbax SB-C18,2.1x30mm,3.5μm,6min,流速为0.6mL/min,流动相:5%-95%(含0.1%甲酸的CH3CN)在(含0.1%甲酸的H2O)中的比例)),在210/254nm用UV检测,用电喷雾电离模式(ESI)。The conditions for low-resolution mass spectrometry (MS) data determination are: Agilent 6120Quadrupole HPLC-MS (column model: Zorbax SB-C18, 2.1x30mm, 3.5μm, 6min, flow rate 0.6mL/min, mobile phase: 5%-95% (Ratio of CH3CN with 0.1% formic acid in ( H2O with 0.1% formic acid))), detected by UV at 210/254 nm, in electrospray ionization mode (ESI).
化合物纯度的表征方式为:Agilent 1260制备型高效液相色谱(Pre-HPLC)或Calesep Pump 250制备型高效液相色谱(Pre-HPLC)(柱子型号:NOVASEP,50/80mm,DAC),在210nm/254nm用UV检测。The purity of the compound was characterized by: Agilent 1260 preparative high performance liquid chromatography (Pre-HPLC) or Calesep Pump 250 preparative high performance liquid chromatography (Pre-HPLC) (column type: NOVASEP, 50/80mm, DAC), at 210nm /254nm with UV detection.
下面简写词的使用贯穿本发明:The following abbreviations are used throughout this disclosure:
H2O水;MeOH,CH3OH甲醇;CD3OD氘代甲醇;SEMCl 2-(三甲硅烷基)乙氧甲基氯;H 2 O water; MeOH, CH 3 OH methanol; CD 3 OD deuterated methanol; SEMCl 2-(trisilyl)ethoxymethyl chloride;
CHCl3氯仿,三氯甲烷;CDCl3氘代氯仿;DMSO二甲基亚砜;DMSO-d6氘代二甲基亚砜;CHCl 3 chloroform, chloroform; CDCl 3 deuterated chloroform; DMSO dimethyl sulfoxide; DMSO-d 6 deuterated dimethyl sulfoxide;
DMF N,N-二甲基甲酰胺;HOBT 1-羟基苯并三唑;Na2SO4硫酸钠;Pd/C钯碳;DMF N,N-dimethylformamide; HOBT 1-hydroxybenzotriazole; Na 2 SO 4 sodium sulfate; Pd/C palladium carbon;
HATU 2-(7-偶氮苯并三氮唑)-N,N,N',N'-四甲基脲六氟磷酸酯;TsCl对甲苯磺酰氯;HATU 2-(7-azobenzotriazole)-N,N,N',N'-tetramethylurea hexafluorophosphate; TsCl p-toluenesulfonyl chloride;
Pd(dppf)Cl2[1,1'-双(二苯基膦基)二茂铁]二氯化钯;Xantphos 4,5-双二苯基膦-9,9-二甲基氧杂蒽;Pd(dppf)Cl 2 [1,1'-bis(diphenylphosphino)ferrocene]palladium dichloride; Xantphos 4,5-bisdiphenylphosphino-9,9-dimethylxanthene ;
X-Phos 2-二环己基磷-2,4,6-三异丙基联苯;Pd2(dba)3三(二亚苄基丙酮)二钯;X-Phos 2-dicyclohexylphosphorus-2,4,6-triisopropylbiphenyl; Pd 2 (dba) 3 tris(dibenzylideneacetone)dipalladium;
t-BuDavePhos 2-(二-叔丁基膦)-2'-(N,N-二甲基氨基)联苯;DBU 1,8-二氮杂双环[5.4.0]十一碳-7-烯;t-BuDavePhos 2-(di-tert-butylphosphine)-2'-(N,N-dimethylamino)biphenyl; DBU 1,8-diazabicyclo[5.4.0]undec-7- alkene;
g克;mg毫克;mol摩尔;mmol毫摩尔;h小时;min分钟;L升;mL,ml毫升;r.t,RT室温;g grams; mg milligrams; mol moles; mmol millimoles; h hours; min minutes; L liters; mL, ml ml; r.t, RT room temperature;
Rt保留时间;HEPES羟乙基哌嗪乙硫磺酸;Brij-35十二烷基聚乙二醇醚;DTT二硫苏糖醇;Rt retention time; HEPES hydroxyethylpiperazine ethanethiosulfonic acid; Brij-35 dodecyl polyethylene glycol ether; DTT dithiothreitol;
EDTA乙二胺四乙酸;EGFR表皮生长因子受体;BTK布鲁顿酪氨酸激酶;EDTA ethylenediaminetetraacetic acid; EGFR epidermal growth factor receptor; BTK Bruton's tyrosine kinase;
EGFR T790M表皮生长因子受体T790M突变体;Peptide FAM-P22荧光素标记肽22;EGFR T790M epidermal growth factor receptor T790M mutant; Peptide FAM-P22 fluorescein-labeled peptide 22;
ATP三磷酸腺苷;96-well plate 96孔板;384-well plate 384孔板;Staurosporine星孢菌素;ATP adenosine triphosphate; 96-well plate 96-well plate; 384-well plate 384-well plate; Staurosporine staurosporine;
Coating Reagent#3 #3被膜剂。Coating Reagent #3 #3 Coating Reagent.
中间体合成方案1Intermediate Synthesis Scheme 1
中间体化合物(6)可以通过中间体合成方案1制备得到,其中R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;R3具有如本发明式(I)中所述的含义。化合物(1)通过过渡金属催化与三甲基硅乙炔在极性溶剂(如:二甲亚砜,N,N-二甲基酰胺,1,4-二氧六环等)中,反应得到化合物(3);化合物 (3)在碱(如:叔丁醇钠,叔丁醇钾,乙醇钠等),或氟化物(如:四丁基氟化铵)的作用下得到化合物(4);化合物(4)在卤代试剂(如:NIS,NBS等)作用下得到化合物(5);化合物(5)与R20Cl(如Ph3CCl,SEMCl等)反应生成化合物(6)。The intermediate compound (6) can be prepared by the intermediate synthesis scheme 1, wherein R 19 and R 19a are each independently halogen; R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; R 3 has the formula of the present invention The meaning described in (I). Compound (1) is catalyzed by transition metals to react with trimethylsilylacetylene in polar solvents (such as dimethyl sulfoxide, N,N-dimethylamide, 1,4-dioxane, etc.) to obtain compounds (3) ; Compound (3) obtains compound (4) under the effect of alkali (such as: sodium tert-butoxide, potassium tert-butoxide, sodium ethoxide etc.), or fluoride (such as: tetrabutylammonium fluoride) ; Compound (4) can obtain compound (5) under the action of halogenated reagent (such as NIS, NBS, etc.); compound (5) is reacted with R 20 Cl (such as Ph 3 CCl, SEMCl, etc.) to form compound (6) .
中间体合成方案2Intermediate Synthesis Scheme 2
中间体化合物(10)可以通过中间体合成方案2制备得到,其中R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;R3和R2具有如本发明式(I)中所述的含义。化合物(1)通过过渡金属催化与 (如:二甲亚砜,N,N-二甲基酰胺,1,4-二氧六环等)中,反应得到化合物(7);化合物(7)在碱(如:叔丁醇钠,叔丁醇钾,乙醇钠等),或氟化物(如:四丁基氟化铵)的作用下得到化合物(8);化合物(8)在卤代试剂(如:NIS,NBS等)作用下得到化合物(9);化合物(9)与R20Cl(如Ph3CCl,SEMCl等)反应生成化合物(10)。The intermediate compound (10) can be prepared by the intermediate synthesis scheme 2, wherein R 19 and R 19a are each independently halogen; R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; R 3 and R 2 have such as The meanings described in formula (I) of the present invention. Compound (1) is catalyzed by transition metals with (such as: dimethyl sulfoxide, N,N-dimethylamide, 1,4-dioxane, etc.), the reaction obtains compound (7) ; compound (7) in a base (such as: sodium tert-butoxide, Under the action of potassium tert-butoxide, sodium ethoxide, etc.), or fluoride (such as: tetrabutylammonium fluoride), compound (8) is obtained; compound (8) is obtained under the action of halogenated reagents (such as: NIS, NBS, etc.) Compound (9) is obtained; compound (9) is reacted with R 20 Cl (such as Ph 3 CCl, SEMCl, etc.) to generate compound (10) .
中间体合成方案3Intermediate Synthesis Scheme 3
中间体化合物(12)可以通过中间体合成方案3制备得到,其中R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(10)和化合物(11)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(12)。Intermediate compound (12) can be prepared by intermediate synthesis scheme 3, wherein R 19 and R 19a are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; E ring, m, R 4 , R 5 , R 3 and R 2 have the meanings as described in formula (I) of the present invention. Compound (10) and compound (11) undergo Suzuki cross-coupling reaction under the catalysis of transition metal to obtain compound (12) .
中间体合成方案4Intermediate Synthesis Scheme 4
中间体化合物(12)可以通过中间体合成方案4制备得到,其中R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(10)和化合物(16)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(12)。Intermediate compound (12) can be prepared by intermediate synthesis scheme 4, wherein R 19 and R 19a are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; E ring, m, R 4 , R 5 , R 3 and R 2 have the meanings as described in formula (I) of the present invention. Compound (10) and compound (16) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (12) .
中间体合成方案5Intermediate Synthesis Scheme 5
中间体化合物(18)可以通过中间体合成方案5制备得到,其中R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;A环、R3和R2具有如本发明式(I)中所述的含义。化合物(10)和化合物(17)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(18)。Intermediate compound (18) can be prepared by intermediate synthesis scheme 5, wherein R 19 and R 19a are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; A ring, R 3 and R 2 has the meaning as described in formula (I) of the present invention. Compound (10) and compound (17) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (18) .
中间体合成方案6Intermediate Synthesis Scheme 6
中间体化合物(18)可以通过中间体合成方案6制备得到,其中R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;A环、R3和R2具有如本发明式(I)中所述的含义。化合物(10)和化合物(13)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(18)。Intermediate compound (18) can be prepared by intermediate synthesis scheme 6, wherein R 19 and R 19a are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; A ring, R 3 and R 2 has the meaning as described in formula (I) of the present invention. Compound (10) and compound (13) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (18) .
中间体合成方案7Intermediate Synthesis Scheme 7
中间体化合物(14)可以通过中间体合成方案7制备得到,其中R19为卤素;R20为Ph3C-,SEM-等保护基团;A环、E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(12)和化合物(13)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(14)。The intermediate compound (14) can be prepared through the intermediate synthesis scheme 7, wherein R 19 is halogen; R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; A ring, E ring, m, R 4 , R 5 , R 3 and R 2 have the meanings as described in formula (I) of the present invention. Compound (12) and compound (13) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (14) .
中间体合成方案8Intermediate Synthesis Scheme 8
中间体化合物(14)可以通过中间体合成方案8制备得到,其中R19为卤素;R20为Ph3C-,SEM-等保护基团;A环、E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(12)和化合物(17)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(14)。The intermediate compound (14) can be prepared by the intermediate synthesis scheme 8, wherein R 19 is halogen; R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; A ring, E ring, m, R 4 , R 5 , R 3 and R 2 have the meanings as described in formula (I) of the present invention. Compound (12) and compound (17) undergo Suzuki cross-coupling reaction under the catalysis of transition metal to obtain compound (14) .
中间体合成方案9Intermediate Synthesis Scheme 9
中间体化合物(14)可以通过中间体合成方案9制备得到,其中R19a为卤素;R20为Ph3C-,SEM-等保护基团;A环、E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(18)和化合物(16)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(14)。The intermediate compound (14) can be prepared through the intermediate synthesis scheme 9, wherein R 19a is halogen; R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; A ring, E ring, m, R 4 , R 5 , R 3 and R 2 have the meanings as described in formula (I) of the present invention. Compound (18) and compound (16) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (14) .
中间体合成方案10Intermediate Synthesis Scheme 10
中间体化合物(14)可以通过中间体合成方案10制备得到,其中R19a为卤素;R20为Ph3C-,SEM-等保护基团;A环、E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(18)和化合物(11)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(14)。The intermediate compound (14) can be prepared through the intermediate synthesis scheme 10, wherein R 19a is halogen; R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; A ring, E ring, m, R 4 , R 5 , R 3 and R 2 have the meanings as described in formula (I) of the present invention. Compound (18) and compound (11) undergo Suzuki cross-coupling reaction under the catalysis of transition metal to obtain compound (14) .
中间体合成方案11Intermediate Synthesis Scheme 11
中间体化合物(12a)可以通过中间体合成方案11制备得到,其中R18、R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;Z1、Z2、R0、R00、n和L具有如本发明式(II)中所述的含义。化合物(10a)和化合物(16a)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(12a)。Intermediate compound (12a) can be prepared by intermediate synthesis scheme 11, wherein R 18 , R 19 and R 19a are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; Z 1 , Z 2 , R 0 , R 00 , n and L have the meanings as described in formula (II) of the present invention. Compound (10a) and compound (16a) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (12a) .
中间体合成方案12Intermediate Synthesis Scheme 12
中间体化合物(12a)可以通过中间体合成方案12制备得到,其中R18、R19和R19a各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;Z1、Z2、R0、R00、n和L具有如本发明式(II)中所述的含义。化合物(10a)和化合物(11a)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(12a)。Intermediate compound (12a) can be prepared by intermediate synthesis scheme 12, wherein R 18 , R 19 and R 19a are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; Z 1 , Z 2 , R 0 , R 00 , n and L have the meanings as described in formula (II) of the present invention. Compound (10a) and compound (11a) undergo Suzuki cross-coupling reaction under the catalysis of transition metal to obtain compound (12a) .
合成方案1Synthesis Scheme 1
化合物(15)可以通过合成方案1制备得到,R20为Ph3C-,SEM-等保护基团;A环、E环、m、R4、R5、R3和R2具有如本发明式(I)中所述的含义。化合物(14)在脱保护反应得到化合物(15)。Compound (15) can be prepared by synthetic scheme 1, R 20 is a protecting group such as Ph 3 C-, SEM-, etc.; A ring, E ring, m, R 4 , R 5 , R 3 and R 2 have the same properties as the present invention. The meanings described in formula (I). Compound (14) is deprotected to obtain compound (15) .
合成方案2Synthesis Scheme 2
化合物(15a)可以通过合成方案2制备得到,其中R18和R19各自独立地为卤素;R20为Ph3C-,SEM-等保护基团;Z1、Z2、R1、R0、R00、n和L具有如本发明式(II)中所述的含义。化合物(12a)发生脱保护反应得到化合物(15a)。Compound (15a) can be prepared by synthetic scheme 2, wherein R 18 and R 19 are each independently halogen; R 20 is Ph 3 C-, SEM- and other protecting groups; Z 1 , Z 2 , R 1 , R 0 , R 00 , n and L have the meanings as described in formula (II) of the present invention. Compound (12a) undergoes deprotection reaction to obtain compound (15a) .
合成方案3Synthesis Scheme 3
化合物(2a)可以通过合成方案3制备得到,其中R19a为卤素;Z1、Z2、W环、R0、R00、n和L具有如本发明式(II)中所述的含义。化合物(1a)和化合物(16a)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(2a)。Compound (2a) can be prepared by Synthesis Scheme 3, wherein R 19a is halogen; Z 1 , Z 2 , W ring, R 0 , R 00 , n and L have the meanings as described in formula (II) of the present invention. Compound (1a) and compound (16a) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (2a) .
合成方案4Synthesis Scheme 4
化合物(2a)可以通过合成方案4制备得到,其中R19a为卤素;Z1、Z2、W环、R0、R00、n和L具有如本发明式(II)中所述的含义。化合物(1a)和化合物(11a)在过渡金属催化下发生Suzuki交叉偶联反应得到化合物(2a)。Compound (2a) can be prepared by Synthesis Scheme 4, wherein R 19a is halogen; Z 1 , Z 2 , W ring, R 0 , R 00 , n and L have the meanings as described in formula (II) of the present invention. Compound (1a) and compound (11a) undergo Suzuki cross-coupling reaction under transition metal catalysis to obtain compound (2a) .
中间体实施例1Intermediate Example 1
2-溴-3-甲基-7-碘-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪2-Bromo-3-methyl-7-iodo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine
步骤1:化合物3,5-二溴-6-氯吡嗪-2-胺的合成Step 1: Synthesis of compound 3,5-dibromo-6-chloropyrazin-2-amine
室温下,将溴素(5.89g,36.54mmol,1.90mL)的氯仿(15mL)溶液在5分钟内缓慢滴加到6-甲基吡嗪-2-胺(2.00g,18.33mmol)的氯仿(170mL)溶液中。滴加完毕后,在室温下搅拌反应4.5h。加水(100mL)淬灭反应,有机层用水(100mL)洗涤,无水硫酸镁干燥,有机层减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到4.20g米黄色固体,产率:86.0%。A solution of bromine (5.89 g, 36.54 mmol, 1.90 mL) in chloroform (15 mL) was slowly added dropwise to 6-methylpyrazin-2-amine (2.00 g, 18.33 mmol) in chloroform ( 170 mL) solution. After the dropwise addition, the reaction was stirred at room temperature for 4.5 h. Water (100 mL) was added to quench the reaction, the organic layer was washed with water (100 mL), dried over anhydrous magnesium sulfate, the organic layer was concentrated under reduced pressure, and subjected to column separation (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 4.20 g beige solid, yield: 86.0%.
1H NMR(400MHz,DMSO-d6):δ(ppm)6.83(s,2H),2.34(s,3H); 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 6.83 (s, 2H), 2.34 (s, 3H);
MS(ESI,pos.ion)m/z:266.0[M+1]+。MS(ESI, pos.ion) m/z: 266.0[M+1] + .
步骤2:化合物5-溴-6-甲基-3-((三甲基硅烷基)乙炔基)吡嗪-2-胺的合成Step 2: Synthesis of compound 5-bromo-6-methyl-3-((trimethylsilyl)ethynyl)pyrazin-2-amine
氮气保护下,在15℃时,将三甲基硅乙炔(1.50g,16.00mmol)缓慢加入到3,5-二溴-6-甲基吡嗪-2-胺(4.00g,15.00mmol)、碘化亚铜(600mg,3.15mmol)、二(三苯基膦)二氯化钯(Pd(PPh3)2Cl2)(600mg,0.85mmol)和三乙胺(2.40g,24.00mmol,3.30mL)的四氢呋喃(42mL)混合物中,滴加完毕后,自然升至室温后,继续反应16h。硅藻土过滤,用乙酸乙酯洗涤,减压浓缩,浓缩残渣加入100mL水,用二氯甲烷萃取(100mL x 3),有机层无水硫酸钠干燥,减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到3.50g米黄色固体,产率:82.0%。Under nitrogen protection, at 15°C, trimethylsilylacetylene (1.50 g, 16.00 mmol) was slowly added to 3,5-dibromo-6-methylpyrazin-2-amine (4.00 g, 15.00 mmol), Cuprous iodide (600 mg, 3.15 mmol), bis(triphenylphosphine)palladium dichloride (Pd(PPh 3 ) 2 Cl 2 ) (600 mg, 0.85 mmol) and triethylamine (2.40 g, 24.00 mmol, 3.30 mL) in tetrahydrofuran (42 mL), after the dropwise addition was completed, the temperature was naturally raised to room temperature, and the reaction was continued for 16 h. Celite was filtered, washed with ethyl acetate, concentrated under reduced pressure, the concentrated residue was added with 100 mL of water, extracted with dichloromethane (100 mL x 3), the organic layer was dried over anhydrous sodium sulfate, concentrated under reduced pressure, and subjected to column separation (petroleum ether). /ethyl acetate (v/v)=2/1) to give 3.50 g of a beige solid, yield: 82.0%.
1H NMR(400MHz,DMSO-d6):δ(ppm)6.69(s,2H),2.39(s,3H),0.26(s,9H); 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 6.69 (s, 2H), 2.39 (s, 3H), 0.26 (s, 9H);
MS(ESI,pos.ion)m/z:284.1[M+1]+。MS(ESI, pos.ion) m/z: 284.1[M+1] + .
步骤3:化合物2-溴-3-甲基-5H-吡咯并[2,3-b]吡嗪的合成Step 3: Synthesis of compound 2-bromo-3-methyl-5H-pyrrolo[2,3-b]pyrazine
N2保护下,-30℃下将5-溴-6-甲基-3-((三甲基硅烷基)乙炔基)吡嗪-2-胺(3.50g,12.00mmol)的四氢呋喃溶液(11mL)缓慢滴加到叔丁醇钾(1.80g,16.00mmol)的四氢呋喃(18mL)溶液中。滴加完毕以后,在-30℃下反应1h后,自然升至室温搅拌反应过夜。反应液硅藻土过滤,乙酸乙酯洗涤,滤液减压浓缩,浓缩残渣加入水(150mL),用乙酸乙酯萃取(150mL x 3),有机层无水硫酸钠干燥,减压浓缩得到2.1g橘黄色固体,粗品不进一步纯化直接用于下一步反应,产率:60.0%。Under N2 protection, a solution of 5-bromo-6-methyl-3-((trimethylsilyl)ethynyl)pyrazin-2-amine (3.50 g, 12.00 mmol) in tetrahydrofuran (11 mL) was added at -30 °C. ) was slowly added dropwise to a solution of potassium tert-butoxide (1.80 g, 16.00 mmol) in tetrahydrofuran (18 mL). After the dropwise addition, the reaction was carried out at -30° C. for 1 h, and then the reaction was naturally raised to room temperature and stirred overnight. The reaction solution was filtered through celite, washed with ethyl acetate, the filtrate was concentrated under reduced pressure, the concentrated residue was added with water (150 mL), extracted with ethyl acetate (150 mL x 3), the organic layer was dried over anhydrous sodium sulfate, and concentrated under reduced pressure to obtain 2.1 g Orange solid, the crude product was used in the next reaction without further purification, yield: 60.0%.
MS(ESI,pos.ion)m/z:212.1[M+1]+。MS(ESI, pos.ion) m/z: 212.1[M+1] + .
步骤4:化合物2-溴-3-甲基-7-碘-5H-吡咯并[2,3-b]吡嗪的合成Step 4: Synthesis of compound 2-bromo-3-methyl-7-iodo-5H-pyrrolo[2,3-b]pyrazine
0℃下,将N-碘代丁二酰亚胺(2.60g,11.00mmol)分批加入到2-溴-3-甲基-5H-吡咯并[2,3-b]吡嗪(2.05g,7.25mmol)的丙酮(30mL)溶液中,室温搅拌反应3h。减压除去溶剂,往残渣中加入水(30mL)和二氯甲烷(30mL),固体过滤,用少许水和二氯甲烷洗涤固体,棕色固体再60℃下真空干燥得到2.50g棕色固体,粗品不进一步纯化直接用于下一步反应。At 0°C, N-iodosuccinimide (2.60 g, 11.00 mmol) was added portionwise to 2-bromo-3-methyl-5H-pyrrolo[2,3-b]pyrazine (2.05 g , 7.25mmol) in acetone (30mL) solution, the reaction was stirred at room temperature for 3h. The solvent was removed under reduced pressure, water (30 mL) and dichloromethane (30 mL) were added to the residue, the solid was filtered, the solid was washed with a little water and dichloromethane, and the brown solid was vacuum-dried at 60°C to obtain 2.50 g of a brown solid. Further purification was used directly in the next reaction.
MS(ESI,pos.ion)m/z:337.7[M+1]+。MS(ESI, pos.ion) m/z: 337.7[M+1] + .
步骤5:化合物2-溴-3-甲基-7-碘-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪的合成Step 5: Compound 2-Bromo-3-methyl-7-iodo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyridine Synthesis of oxazine
0℃下,将氢化钠(450mg,11.25mmol)分批加入到2-溴-3-甲基-7-碘-5H-吡咯并[2,3-b]吡嗪(2.50g,7.4mmol)的N,N-二甲基甲酰胺(30mL)混合液中。室温下搅拌2h后。在0℃下缓慢滴加三甲基硅基乙氧基甲基氯(1.90g,11.00mmol,2.00mL),滴加完毕后,室温下搅拌反应过夜。加水(100mL)淬灭反应,二氯甲烷(100mL x 3)萃取,有机层用饱和食盐水(100mL)洗涤,无水硫酸钠干燥,浓缩除去溶剂,残留物进行柱层析分离(石油醚/乙酸乙酯(v/v)=10/1)得到1.20g淡黄色油状物,两步总产率:35.0%。At 0°C, sodium hydride (450 mg, 11.25 mmol) was added portionwise to 2-bromo-3-methyl-7-iodo-5H-pyrrolo[2,3-b]pyrazine (2.50 g, 7.4 mmol) of N,N-dimethylformamide (30 mL). After stirring for 2 h at room temperature. Trimethylsilylethoxymethyl chloride (1.90 g, 11.00 mmol, 2.00 mL) was slowly added dropwise at 0° C. After the dropwise addition, the reaction was stirred at room temperature overnight. The reaction was quenched by adding water (100 mL), extracted with dichloromethane (100 mL x 3), the organic layer was washed with saturated brine (100 mL), dried over anhydrous sodium sulfate, concentrated to remove the solvent, and the residue was separated by column chromatography (petroleum ether/ Ethyl acetate (v/v) = 10/1) gave 1.20 g of a pale yellow oil, two-step overall yield: 35.0%.
MS(ESI,pos.ion)m/z:467.9[M+1]+。MS(ESI, pos.ion) m/z: 467.9[M+1] + .
采用相应的原料,按照中间体实施例1的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthesis method of Intermediate Example 1:
中间体实施例6Intermediate Example 6
2-氯-7-碘-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪2-Chloro-7-iodo-5-trityl-5H-pyrrolo[2,3-b]pyrazine
步骤1:化合物2-氯-5H-吡咯并[2,3-b]吡嗪的合成Step 1: Synthesis of compound 2-chloro-5H-pyrrolo[2,3-b]pyrazine
向2-溴-5H-吡咯并[2,3-b]吡嗪(3.1g,16mmol)的1,4-二氧六环(20mL)溶液中加入三氯氧磷(2.27mL,24.1mmol)和三乙胺(220μL,1.58mmol),110℃回流反应6小时,慢慢加入到水中(100mL)淬灭,氢氧化钾溶液调至pH>7,乙酸乙酯萃取(50mLx3),无水硫酸钠干燥,浓缩后,打浆(石油醚/乙酸乙酯(v/v)=2/1),过滤,干燥得到1.3g灰色固体,产率:54%。To a solution of 2-bromo-5H-pyrrolo[2,3-b]pyrazine (3.1 g, 16 mmol) in 1,4-dioxane (20 mL) was added phosphorus oxychloride (2.27 mL, 24.1 mmol) and triethylamine (220 μL, 1.58 mmol), refluxed at 110 °C for 6 hours, slowly added to water (100 mL) to quench, potassium hydroxide solution was adjusted to pH>7, extracted with ethyl acetate (50 mL×3), anhydrous sulfuric acid It was dried over sodium, concentrated, slurried (petroleum ether/ethyl acetate (v/v)=2/1), filtered, and dried to obtain 1.3 g of a gray solid, yield: 54%.
MS(ESI,pos.ion)m/z:154.1[M+1]+。MS(ESI, pos.ion) m/z: 154.1[M+1] + .
步骤2:化合物2-氯-7-碘-5H-吡咯并[2,3-b]吡嗪的合成Step 2: Synthesis of compound 2-chloro-7-iodo-5H-pyrrolo[2,3-b]pyrazine
向2-氯-5H-吡咯并[2,3-b]吡嗪(1.3g,8.5mmol)的丙酮(15mL)溶液中加入N-碘代琥珀酰亚胺(2.9g,13mmol),室温搅拌12小时,加水(40mL)稀释,二氯甲烷萃取(55mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到2.2mg灰白色固体,产率:93%。To a solution of 2-chloro-5H-pyrrolo[2,3-b]pyrazine (1.3 g, 8.5 mmol) in acetone (15 mL) was added N-iodosuccinimide (2.9 g, 13 mmol) and stirred at room temperature 12 hours, diluted with water (40 mL), extracted with dichloromethane (55 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 2.2 mg of off-white solid, Yield: 93%.
MS(ESI,pos.ion)m/z:280.0[M+1]+。MS(ESI, pos.ion) m/z: 280.0[M+1] + .
步骤3:化合物2-氯-7-碘-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪的合成Step 3: Synthesis of compound 2-chloro-7-iodo-5-trityl-5H-pyrrolo[2,3-b]pyrazine
向2-氯-7-碘-5H-吡咯并[2,3-b]吡嗪(300mg,1.07mmol)的N,N-二甲基甲酰胺(8mL)溶液中加入碳酸钠(230mg,2.17mmol)和三苯基氯甲烷(330mg,1.18mmol),45℃加热反应5小时,加水(20mL)稀释,二氯甲烷萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=8/1)得到320mg白色固体,产率:57.13%。To a solution of 2-chloro-7-iodo-5H-pyrrolo[2,3-b]pyrazine (300 mg, 1.07 mmol) in N,N-dimethylformamide (8 mL) was added sodium carbonate (230 mg, 2.17 mmol) and triphenylchloromethane (330 mg, 1.18 mmol), heated at 45°C for 5 hours, diluted with water (20 mL), extracted with dichloromethane (25 mL×3), dried over anhydrous sodium sulfate, and concentrated by column chromatography (petroleum ether/ Ethyl acetate (v/v)=8/1) gave 320 mg of white solid, yield: 57.13%.
MS(ESI,pos.ion)m/z:522.10[M+1]+;MS(ESI, pos.ion) m/z: 522.10[M+1] + ;
1H NMR(400MHz,CDCl3)δ(ppm)7.92(s,1H),7.65(s,1H),7.31(m,9H),7.17(m,6H)。 1 H NMR (400 MHz, CDCl 3 ) δ (ppm) 7.92 (s, 1H), 7.65 (s, 1H), 7.31 (m, 9H), 7.17 (m, 6H).
中间体实施例7Intermediate Example 7
2,7-二溴-6-甲基-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪2,7-Dibromo-6-methyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine
步骤1:化合物5-溴-3-(丙炔-1-基)吡嗪-2-胺的合成Step 1: Synthesis of compound 5-bromo-3-(propyn-1-yl)pyrazin-2-amine
向3,5-二溴吡嗪-2-胺(200mg,0.79mmol)的四氢呋喃(8mL)溶液中,依次加入碘化亚铜(15mg,0.07mmol),二(三苯基膦)二氯化钯(56mg,0.08mmol)和三乙胺(340μL,2.44mmol),氮气保护下,冰浴下慢慢滴加加入3%丙炔正庚烷溶液(2.3mL,1.2mmol),自然升至室温后搅拌3小时,硅藻提过滤,浓缩,柱层析(石油醚/乙酸乙酯(v/v)=8/1)得到150mg淡黄色固体,产率:89.45%。To a solution of 3,5-dibromopyrazin-2-amine (200 mg, 0.79 mmol) in tetrahydrofuran (8 mL) were added cuprous iodide (15 mg, 0.07 mmol), followed by bis(triphenylphosphine)dichloride Palladium (56 mg, 0.08 mmol) and triethylamine (340 μL, 2.44 mmol), under nitrogen protection, slowly dropwise add 3% propyne n-heptane solution (2.3 mL, 1.2 mmol) under ice bath, and naturally warm to room temperature After stirring for 3 hours, diatom extraction and filtration, concentration, column chromatography (petroleum ether/ethyl acetate (v/v)=8/1) to obtain 150 mg of pale yellow solid, yield: 89.45%.
MS(ESI,pos.ion)m/z:212.1,214.1[M+1]+。MS(ESI, pos.ion) m/z: 212.1, 214.1[M+1] + .
步骤2:化合物2-溴-6-甲基-5H-吡唑并[2,3-b]吡嗪的合成Step 2: Synthesis of compound 2-bromo-6-methyl-5H-pyrazolo[2,3-b]pyrazine
室温下,氮气保护下,向叔丁醇钾(118mg,1.05mmol)的四氢呋喃(8mL)溶液中,加入5-溴-3-(丙炔-1-基)吡嗪-2-胺(150mg,0.70mmol)的四氢呋喃溶液(5mL),回流反应1小时,硅藻土过滤,除去溶剂,残留物柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到124mg淡黄色固体产物,产率:82.67%。To a solution of potassium tert-butoxide (118 mg, 1.05 mmol) in tetrahydrofuran (8 mL) was added 5-bromo-3-(propyn-1-yl)pyrazin-2-amine (150 mg, 0.70 mmol) in tetrahydrofuran (5 mL), refluxed for 1 hour, filtered through celite, removed the solvent, and the residue was subjected to column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 124 mg of pale yellow solid Product, yield: 82.67%.
MS(ESI,pos.ion)m/z:212.1,214.1[M+1]+;MS(ESI, pos.ion) m/z: 212.1, 214.1[M+1] + ;
1H NMR(400MHz,DMSO-d6)δ(ppm)12.19(s,1H),8.21(s,1H),6.35(s,1H),2.49(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 ) δ (ppm) 12.19 (s, 1H), 8.21 (s, 1H), 6.35 (s, 1H), 2.49 (s, 3H).
步骤3:化合物2,7-二溴-6-甲基-5H-吡咯并[2,3-b]吡嗪的合成Step 3: Synthesis of compound 2,7-dibromo-6-methyl-5H-pyrrolo[2,3-b]pyrazine
室温下,向2-溴-6-甲基-5H-吡唑并[2,3-b]吡嗪(500mg,2.35mmol)的N,N-二甲基甲酰胺(12mL)溶液中,加入N-溴代琥珀酰亚胺(630mg,3.54mmol),室温搅拌5小时,加水(20mL)稀释,二氯甲烷萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到450mg淡黄色固体,产率:65.95%。To a solution of 2-bromo-6-methyl-5H-pyrazolo[2,3-b]pyrazine (500 mg, 2.35 mmol) in N,N-dimethylformamide (12 mL) at room temperature was added N-bromosuccinimide (630 mg, 3.54 mmol), stirred at room temperature for 5 hours, diluted with water (20 mL), extracted with dichloromethane (25 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate) Ester (v/v)=2/1) to give 450 mg of pale yellow solid, yield: 65.95%.
MS(ESI,pos.ion)m/z:292.0[M+1]+。MS(ESI, pos.ion) m/z: 292.0[M+1] + .
步骤4:化合物2,7-二溴-6-甲基-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪的合成Step 4: Compound 2,7-Dibromo-6-methyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine Synthesis
0℃下,向2,7-二溴-6-甲基-5H-吡咯并[2,3-b]吡嗪(450mg,1.54mmol)的四氢呋喃(12mL)溶液中加入氢化钠(92mg,2.30mmol),继续在此温度下反应1小时后,慢慢滴加入SEMCl(410μL,2.32mmol),继续室温搅拌,加水(20mL)稀释,二氯甲烷萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到360mg棕色油状物,产率:55.26%。To a solution of 2,7-dibromo-6-methyl-5H-pyrrolo[2,3-b]pyrazine (450 mg, 1.54 mmol) in tetrahydrofuran (12 mL) was added sodium hydride (92 mg, 2.30 mL) at 0°C mmol), continue to react at this temperature for 1 hour, slowly add SEMCl (410 μL, 2.32 mmol) dropwise, continue to stir at room temperature, add water (20 mL) to dilute, extract with dichloromethane (25 mL×3), dry over anhydrous sodium sulfate, and concentrate Column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) gave 360 mg of brown oil, yield: 55.26%.
MS(ESI,pos.ion)m/z:422.1[M+1]+。MS(ESI, pos.ion) m/z: 422.1[M+1] + .
中间体实施例8Intermediate Example 8
7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪7-Bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2, 3-b]pyrazine
向2,7-二溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(1.0g,2.5mmol)的乙腈(6mL)溶液中依次加入1-甲基吡唑-4-硼酸酯(620mg,2.98mmol),碳酸钠(650mg,6.13mmol),Pd(PPh3)2Cl2(180mg,0.26mmol)和水(6mL),80℃加热搅拌3小时,加水(40mL)稀释,二氯甲烷萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到1.0g黄色固体,产率:99%。To 2,7-dibromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (1.0 g, 2.5 mmol) To the acetonitrile (6 mL) solution was added 1-methylpyrazole-4-boronate (620 mg, 2.98 mmol), sodium carbonate (650 mg, 6.13 mmol), Pd(PPh 3 ) 2 Cl 2 (180 mg, 0.26 mmol) in turn and water (6 mL), heated and stirred at 80°C for 3 hours, diluted with water (40 mL), extracted with dichloromethane (25 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/v) = 2/1) 1.0 g of a yellow solid was obtained, yield: 99%.
MS(ESI,pos.ion)m/z:408.10,410.10[M+1]+。MS(ESI, pos.ion) m/z: 408.10, 410.10 [M+1] + .
采用相应的原料,按照中间体实施例8的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 8:
中间体实施例12Intermediate Example 12
N-(2-(甲磺酰基)乙基)-4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)胺N-(2-(Methylsulfonyl)ethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)amine
步骤1:化合物4-碘-N-(2-(甲磺酰基)乙基)胺的合成Step 1: Synthesis of compound 4-iodo-N-(2-(methylsulfonyl)ethyl)amine
氮气保护下,将DMSO(15mL)加入到对二碘苯(408mg,1.24mmol)、2-(甲磺酰基)乙胺盐酸盐(220mg,1.38mmol)、碘化亚铜(250mg,1.31mmol)、碳酸铯(1.20g,3.70mmol)、二甲基甘氨酸(208mg,1.21mmol)的混合物中,80℃反应22h,反应液冷却至室温,用乙酸乙酯萃取(50mLx3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=1/1)得到78mg白色固体,产率:19.4%。Under nitrogen protection, DMSO (15 mL) was added to p-diiodobenzene (408 mg, 1.24 mmol), 2-(methylsulfonyl)ethylamine hydrochloride (220 mg, 1.38 mmol), cuprous iodide (250 mg, 1.31 mmol) ), cesium carbonate (1.20g, 3.70mmol) and dimethylglycine (208mg, 1.21mmol), react at 80°C for 22h, the reaction solution was cooled to room temperature, extracted with ethyl acetate (50mL×3), and the organic phase was spin-dried , and separated by column chromatography (petroleum ether/ethyl acetate (v/v)=1/1) to obtain 78 mg of white solid, yield: 19.4%.
MS(ESI,pos.ion)m/z:326.1[M+1]+。MS(ESI, pos.ion) m/z: 326.1[M+1] + .
步骤2:化合物N-(2-(甲磺酰基)乙基)-4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)胺的合成Step 2: Compound N-(2-(methylsulfonyl)ethyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)amine Synthesis
氮气保护下,将DMSO(12mL)加入到4-碘-N-(2-(甲磺酰基)乙基)胺(78mg,0.24mmol)、联硼酸频哪醇酯(75mg,0.30mmol)、醋酸钾(75mg,0.76mmol)、Pd(dppf)Cl2(9mg,0.01mmol)混合物中,105℃反应17h,反应液冷却至室温,用乙酸乙酯萃取(50mLx3),浓缩有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=1/2)得到58mg无色油状物,产率:74.4%。Under nitrogen, DMSO (12 mL) was added to 4-iodo-N-(2-(methylsulfonyl)ethyl)amine (78 mg, 0.24 mmol), pinacol biboronate (75 mg, 0.30 mmol), acetic acid Potassium (75mg, 0.76mmol), Pd(dppf)Cl 2 (9mg, 0.01mmol) mixture, react at 105°C for 17h, the reaction solution was cooled to room temperature, extracted with ethyl acetate (50mL×3), the organic phase was concentrated, column chromatography Separation (petroleum ether/ethyl acetate (v/v)=1/2) gave 58 mg of colorless oil, yield: 74.4%.
MS(ESI,pos.ion)m/z:326.3[M+1]+。MS(ESI, pos.ion) m/z: 326.3[M+1] + .
采用相应的原料,按照中间体实施例12的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 12:
中间体实施例32Intermediate Example 32
4,4,5,5-四甲基-2-(1,4-二氧螺[4.5]葵烷-7-烯-8-基)-1,3,2-二氧硼戊烷4,4,5,5-Tetramethyl-2-(1,4-dioxospiro[4.5]decane-7-en-8-yl)-1,3,2-dioxaboropentane
步骤1:化合物1,4-二氧螺[4.5]葵烷-7-烯-8-三氟甲磺酸酯的合成Step 1: Synthesis of Compound 1,4-dioxospiro[4.5]decane-7-ene-8-trifluoromethanesulfonate
-78℃下,向1,4-二氧螺[4.5]葵烷-8-酮(2.0g,13mmol)的四氢呋喃(15mL)溶液中加入六甲基二硅基 胺基钾(1M,26mL,26mmol)的四氢呋喃溶液(26mL),继续在该温度下搅拌,加入N,N-二(三氟甲磺酰基)苯胺(5.46g,15.3mmol)的四氢呋喃溶液(15mL),自然升至室温搅拌过夜,加水(30mL)淬灭,二氯甲烷萃取(30mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=5/1)得到4.5g淡黄色油状物,产率:80.23%。To a solution of 1,4-dioxospiro[4.5]decane-8-one (2.0 g, 13 mmol) in tetrahydrofuran (15 mL) at -78 °C was added potassium hexamethyldisilazide (1 M, 26 mL, 26 mmol) in tetrahydrofuran (26 mL), continue to stir at this temperature, add N,N-bis(trifluoromethanesulfonyl)aniline (5.46 g, 15.3 mmol) in tetrahydrofuran (15 mL), naturally warm to room temperature and stir overnight , quenched with water (30 mL), extracted with dichloromethane (30 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=5/1) to obtain 4.5 g of pale yellow oil , Yield: 80.23%.
MS(ESI,pos.ion)m/z:289.0[M+1]+。MS(ESI, pos.ion) m/z: 289.0[M+1] + .
步骤2:化合物4,4,5,5-四甲基-2-(1,4-二氧螺[4.5]葵烷-7-烯-8-基)-1,3,2-二氧硼戊烷的合成Step 2: Compound 4,4,5,5-Tetramethyl-2-(1,4-dioxospiro[4.5]decane-7-en-8-yl)-1,3,2-dioxoboron Synthesis of Pentane
1,4-二氧螺[4.5]葵烷-7-烯-8-三氟甲磺酸酯(500mg,1.73mmol)的1,4-二氧六环(8mL)溶液中依次加入醋酸钾(260mg,2.64mmol),联硼酸频哪醇脂(530mg,2.08mmol),Pd(dppf)Cl2(130mg,0.17mmol),氮气保护,110℃回流反应10小时,过滤,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到540mg黄色油状物,产率:99%。Potassium acetate ( 260mg ; Ether/ethyl acetate (v/v)=2/1) to give 540 mg of yellow oil, yield: 99%.
MS(ESI,pos.ion)m/z:267.30[M+1]+。MS(ESI, pos.ion) m/z: 267.30[M+1] + .
采用相应的原料,按照中间体实施例32的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 32:
中间体实施例34Intermediate Example 34
(R)-2-甲基-2-((3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基)-N-(2,2,2-三氟乙基)丁酰胺(R)-2-Methyl-2-((3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl)amino)- N-(2,2,2-Trifluoroethyl)butanamide
步骤1:化合物(R)-2-((3-溴苯基)氨基)-2-甲基丁酸的合成Step 1: Synthesis of compound (R)-2-((3-bromophenyl)amino)-2-methylbutanoic acid
向间溴碘苯(500mg,1.76mmol)的DMF(5mL)溶液中依次加入碘化亚铜(340mg,1.78mmol),碳酸铯(870mg,2.67mmol),(R)-2-氨基-2-甲基丁酸(550mg,4.55mmol),80℃加热反应8小时,直接浓缩拌样,柱层析(石油醚/乙酸乙酯(v/v)=1/1)得到130mg棕色固体,产率:27.02%。To a solution of m-bromoiodobenzene (500 mg, 1.76 mmol) in DMF (5 mL) were added cuprous iodide (340 mg, 1.78 mmol), cesium carbonate (870 mg, 2.67 mmol), (R)-2-amino-2- Methylbutyric acid (550mg, 4.55mmol), heated at 80°C for 8 hours, directly concentrated and mixed with sample, column chromatography (petroleum ether/ethyl acetate (v/v)=1/1) gave 130mg of brown solid, yield : 27.02%.
MS(ESI,pos.ion)m/z:271.9,273.9[M+1]+。MS(ESI, pos.ion) m/z: 271.9, 273.9[M+1] + .
步骤2:化合物(R)-2-((3-溴苯基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺的合成Step 2: Synthesis of compound (R)-2-((3-bromophenyl)amino)-2-methyl-N-(2,2,2-trifluoroethyl)butanamide
向(R)-2-((3-溴苯基)氨基)-2-甲基丁酸(70mg,0.25mmol)的DMF(5mL)溶液中依次加入N-甲基吗啉(80mg,0.79mmol),2,2,2-三氟乙胺盐酸盐(60mg,0.44mmol)和HATU(146mg,0.38mmol),室温搅拌2小时,加水(30mL)稀释,二氯甲烷萃取(30mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=1/1)得到70mg黄色固体,产率:77.05%。To a solution of (R)-2-((3-bromophenyl)amino)-2-methylbutanoic acid (70 mg, 0.25 mmol) in DMF (5 mL) was sequentially added N-methylmorpholine (80 mg, 0.79 mmol) ), 2,2,2-trifluoroethylamine hydrochloride (60mg, 0.44mmol) and HATU (146mg, 0.38mmol), stirred at room temperature for 2 hours, diluted with water (30mL), extracted with dichloromethane (30mLx3), no It was dried over sodium sulfate and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=1/1) to obtain 70 mg of a yellow solid, yield: 77.05%.
MS(ESI,pos.ion)m/z:353.2,355.1[M+1]+。MS(ESI, pos.ion) m/z: 353.2, 355.1[M+1] + .
步骤3:化合物(R)-2-甲基-2-((3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基)-N-(2,2,2-三氟乙基)丁酰胺的合成Step 3: Compound (R)-2-methyl-2-((3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl) )amino)-N-(2,2,2-trifluoroethyl)butanamide
向(R)-2-((3-溴苯基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺(350mg,0.99mmol)的二甲基亚砜(8mL)溶液中依次加入醋酸钾(250mg,2.54mmol),联硼酸频哪醇酯(380mg,1.49mmol)和Pd(dppf)Cl2(70mg, 0.09mmol),氮气氛围下,110℃加热反应8小时,加水(30mL)稀释,二氯甲烷萃取(30mL x 3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=8/1)得到350mg淡黄色油状物,产率:60.52%。Dimethylidene to (R)-2-((3-bromophenyl)amino)-2-methyl-N-(2,2,2-trifluoroethyl)butanamide (350 mg, 0.99 mmol) To the sulfone (8 mL) solution was sequentially added potassium acetate (250 mg, 2.54 mmol), biboronic acid pinacol ester (380 mg, 1.49 mmol) and Pd(dppf)Cl 2 (70 mg, 0.09 mmol), heated at 110 ° C under nitrogen atmosphere The reaction was carried out for 8 hours, diluted with water (30 mL), extracted with dichloromethane (30 mL x 3), dried over anhydrous sodium sulfate, and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=8/1) to obtain 350 mg of light Yellow oil, yield: 60.52%.
MS(ESI,pos.ion)m/z:401.0[M+1]+。MS(ESI, pos.ion) m/z: 401.0[M+1] + .
采用相应的原料,按照中间体实施例34的合成方法制备如下所示的中间体:The intermediate shown below was prepared according to the synthetic method of Intermediate Example 34 using the corresponding raw materials:
中间体实施例37Intermediate Example 37
1-(2-氰乙基)-N-(3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)吡咯烷-3-甲酰胺1-(2-cyanoethyl)-N-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl)pyrrolidine- 3-Carboxamide
步骤1:化合物1-甲酸叔丁酯-吡咯烷-3-羧酸的合成Step 1: Synthesis of compound 1-tert-butylcarboxylate-pyrrolidine-3-carboxylic acid
将氢氧化钠(0.80g,20.00mmol)的水(15mL)溶液加入到1-甲酸叔丁酯-吡咯烷-3-甲酸甲酯(2.30g,10.00mmol)的甲醇(30mL)溶液,反应液在室温下反应3小时,减压浓缩除去甲醇,用2M的冰醋酸将pH调至pH=4左右,二氯甲烷(50mL x 3)萃取,无水硫酸钠干燥,减压浓缩得到油状物1.80g,产率:83.00%。A solution of sodium hydroxide (0.80 g, 20.00 mmol) in water (15 mL) was added to a solution of methyl 1-carboxylate-pyrrolidine-3-carboxylate (2.30 g, 10.00 mmol) in methanol (30 mL), the reaction solution The reaction was carried out at room temperature for 3 hours, concentrated under reduced pressure to remove methanol, adjusted to pH=4 with 2M glacial acetic acid, extracted with dichloromethane (50 mL x 3), dried over anhydrous sodium sulfate, and concentrated under reduced pressure to obtain an oily product 1.80 g, Yield: 83.00%.
MS(ESI,pos.ion)m/z:160.1[M-55]+。MS (ESI, pos.ion) m/z: 160.1 [M-55] + .
步骤2:化合物3-((3-溴苯基)氨基甲酰基)吡咯烷-1-甲酸叔丁酯的合成Step 2: Synthesis of compound 3-((3-bromophenyl)carbamoyl)pyrrolidine-1-carboxylic acid tert-butyl ester
将3-溴苯胺(1.40g,8.40mmol,0.91mL)加入到1-甲酸叔丁酯-吡咯烷-3-羧酸(1.80g,8.40mmol),HATU(6.40g,17.00mmol)和N,N-二异丙基乙胺(3.30g,25.00mmol,4.40mL)的二氯甲烷(20mL)溶液中,室温下反应4h。用水(50mL)淬灭反应,二氯甲烷(50mL x3)萃取,有机层用饱和食盐水(50mL)洗涤,无水硫酸钠干燥,减压浓缩,浓缩液进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到2.40g棕黄色油状物,产率:78.0%。3-Bromoaniline (1.40 g, 8.40 mmol, 0.91 mL) was added to tert-butyl 1-carboxylate-pyrrolidine-3-carboxylic acid (1.80 g, 8.40 mmol), HATU (6.40 g, 17.00 mmol) and N, In a solution of N-diisopropylethylamine (3.30 g, 25.00 mmol, 4.40 mL) in dichloromethane (20 mL), the reaction was carried out at room temperature for 4 h. The reaction was quenched with water (50 mL), extracted with dichloromethane (50 mL×3), the organic layer was washed with saturated brine (50 mL), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the concentrate was subjected to column separation (petroleum ether/ethyl acetate) (v/v)=2/1) to give 2.40 g of a brownish yellow oil, yield: 78.0%.
MS(ESI,pos.ion)m/z:312.9[M-55]+。MS(ESI, pos.ion) m/z: 312.9[M-55] + .
步骤3:化合物3-((3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基甲酰基)吡咯烷-1-甲酸叔丁酯的合成Step 3: Compound 3-((3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl)carbamoyl)pyrrole Synthesis of tert-butyl alkane-1-carboxylate
在氮气保护下,向3-((3-溴苯基)氨基甲酰基)吡咯烷-1-甲酸叔丁酯(2.40g,6.50mmol),联硼酸频哪醇酯(9.80mg,2.50mmol),醋酸钾(1.30mg,13.00mmol),Pd(dppf)Cl2(240mg,0.32mmol)的混合物中注入二甲亚砜(20mL),反应液在100℃反应12h。冷却至室温,用水(150mL)和乙酸乙酯(150mL)将反应液分层,水相用乙酸乙酯萃取(150mLx3),有机层用饱和盐水洗涤(100mL x 2),无水硫酸钠干燥,减压 浓缩,浓缩液进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到1.20g淡黄色油状物,产率:44.0%。Under nitrogen protection, tert-butyl 3-((3-bromophenyl)carbamoyl)pyrrolidine-1-carboxylate (2.40 g, 6.50 mmol), pinacol biboronate (9.80 mg, 2.50 mmol) was added , dimethyl sulfoxide (20 mL) was injected into the mixture of potassium acetate (1.30 mg, 13.00 mmol), and Pd(dppf)Cl 2 (240 mg, 0.32 mmol), and the reaction solution was reacted at 100° C. for 12 h. Cooled to room temperature, the reaction solution was separated with water (150 mL) and ethyl acetate (150 mL), the aqueous phase was extracted with ethyl acetate (150 mL×3), the organic layer was washed with saturated brine (100 mL×2), dried over anhydrous sodium sulfate, It was concentrated under reduced pressure, and the concentrated solution was subjected to column separation (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 1.20 g of pale yellow oil, yield: 44.0%.
MS(ESI,pos.ion)m/z:361.3[M+1]+。MS(ESI, pos.ion) m/z: 361.3[M+1] + .
步骤4:化合物N-(3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)吡咯烷-3-甲酰胺盐酸盐的合成Step 4: Compound N-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl)pyrrolidine-3-carboxamide hydrochloride salt synthesis
室温下将氯化氢的乙酸乙酯溶液(2M,14.00mmol,7.00mL)加入到3-((3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基甲酰基)吡咯烷-1-甲酸叔丁酯(1.20g,2.90mmol)的乙酸乙酯(15mL)溶液中,反应2h。减压浓缩得到1.20g油状物,产率:120.0%。A solution of hydrogen chloride in ethyl acetate (2M, 14.00 mmol, 7.00 mL) was added to 3-((3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxo) at room temperature Borpentan-2-yl)phenyl)carbamoyl)pyrrolidine-1-carboxylic acid tert-butyl ester (1.20g, 2.90mmol) in ethyl acetate (15mL) solution, react for 2h. Concentrate under reduced pressure to obtain 1.20g Oil, yield: 120.0%.
MS(ESI,pos.ion)m/z:317.1[M+1]+。MS(ESI, pos.ion) m/z: 317.1[M+1] + .
步骤5:化合物1-(2-氰乙基)-N-(3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)吡咯烷-3-甲酰胺的合成Step 5: Compound 1-(2-cyanoethyl)-N-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl ) Synthesis of pyrrolidine-3-carboxamide
室温下,将3-溴丙腈(1.00g,7.00mmol)加入到N-(3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)吡咯烷-3-甲酰胺盐酸盐(1.20g,3.4mmol)和碳酸铯(5.50g,17.00mmol)的乙腈(25mL)溶剂中,反应在90℃下回流反应4h。硅藻土过滤,用乙酸乙酯洗涤滤饼,减压浓缩,浓缩液进行柱分离(二氯甲烷/甲醇(v/v)=15/1)得到550mg淡黄色油状物,产率:44.0%。3-Bromopropionitrile (1.00 g, 7.00 mmol) was added to N-(3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentane-2-) at room temperature yl)phenyl)pyrrolidine-3-carboxamide hydrochloride (1.20 g, 3.4 mmol) and cesium carbonate (5.50 g, 17.00 mmol) in acetonitrile (25 mL) solvent, the reaction was refluxed at 90 °C for 4 h. The filter cake was filtered through celite, washed with ethyl acetate, concentrated under reduced pressure, and the concentrated solution was subjected to column separation (dichloromethane/methanol (v/v)=15/1) to obtain 550 mg of pale yellow oil, yield: 44.0% .
MS(ESI,pos.ion)m/z:370.0[M+1]+。MS(ESI, pos.ion) m/z: 370.0[M+1] + .
中间体实施例38Intermediate Example 38
(R)-2-甲基-2-((3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基)-N-(2,2,2-三氟乙基)丁酰胺(R)-2-Methyl-2-((3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl)amino)- N-(2,2,2-Trifluoroethyl)butanamide
步骤1:化合物(R)-2-((叔丁氧羰基)氨基)-2-甲基丁酸的合成Step 1: Synthesis of compound (R)-2-((tert-butoxycarbonyl)amino)-2-methylbutanoic acid
氮气保护下,将(R)-2-氨基-2-甲基丁酸(2.05g,17.50mmol)、三乙胺(7.01mL,50.20mmol)、二羧酸二叔丁酯(5.70g,26.00mmol)加到一起,再加入四氢呋喃(20mL),升温至70℃反应3h,反应液冷却至室温,乙酸乙酯(50mL×3)萃取,用无水Na2SO4干燥,除去溶剂,进行柱分离(二氯甲烷/甲醇(v/v)=10/1)得到3.60g无色油状物,产率:97.0%。Under nitrogen protection, (R)-2-amino-2-methylbutanoic acid (2.05 g, 17.50 mmol), triethylamine (7.01 mL, 50.20 mmol), di-tert-butyl dicarboxylate (5.70 g, 26.00 mmol) were added together, then tetrahydrofuran (20 mL) was added, the temperature was raised to 70 °C and reacted for 3 h, the reaction solution was cooled to room temperature, extracted with ethyl acetate (50 mL×3), dried with anhydrous Na 2 SO 4 , removed the solvent, and carried out a column Separation (dichloromethane/methanol (v/v)=10/1) gave 3.60 g of colorless oil, yield: 97.0%.
MS(ESI,pos.ion)m/z:118.2[M-99]+。MS(ESI, pos.ion) m/z: 118.2[M-99] + .
步骤2:化合物(R)-(2-甲基-1-氧-1-((2,2,2-三氟乙基)氨基)丁基-2-基)氨基甲酸酯的合成Step 2: Synthesis of compound (R)-(2-methyl-1-oxo-1-((2,2,2-trifluoroethyl)amino)butyl-2-yl)carbamate
氮气保护下,将2,2,2-三氟乙基胺盐酸盐(2.85g,21.01mmol)、(R)-2-((叔丁氧羰基)氨基)-2-甲基丁酸(3.71g,17.01mmol)、HATU(8.81g,23.02mmol)、N-甲基吗啉(5.21g,51.01mmol)加到一起,再加入DMF(30mL),室温搅拌5h,乙酸乙酯(100mL×3)萃取,用无水Na2SO4干燥,除去溶剂,进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到3.57g白色固体,产率:70.1%。Under nitrogen protection, 2,2,2-trifluoroethylamine hydrochloride (2.85 g, 21.01 mmol), (R)-2-((tert-butoxycarbonyl)amino)-2-methylbutanoic acid ( 3.71g, 17.01mmol), HATU (8.81g, 23.02mmol), N-methylmorpholine (5.21g, 51.01mmol) were added together, then DMF (30mL) was added, stirred at room temperature for 5h, ethyl acetate (100mL× 3) Extraction, drying with anhydrous Na 2 SO 4 , removing the solvent, and performing column separation (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 3.57 g of white solid, yield: 70.1%.
MS(ESI,pos.ion)m/z:199.1[M-99]+。MS(ESI, pos.ion) m/z: 199.1[M-99] + .
步骤3:化合物(R)-2-((3-溴苯基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺的合成Step 3: Synthesis of compound (R)-2-((3-bromophenyl)amino)-2-methyl-N-(2,2,2-trifluoroethyl)butanamide
室温下,(R)-(2-甲基-1-氧-1-((2,2,2-三氟乙基)氨基)丁基-2-基)氨基甲酸酯(3.57g,12.01mmol)溶解于氯化氢的乙酸乙酯溶液(15mL)中,室温搅拌5h,减压浓缩反应液。氮气保护下,加入碘化亚铜(1.60g,8.40mmol)、碳酸铯(11.80g,36.20mmol)、L-脯氨酸(970mg,8.43mmol)、间溴碘苯(3.73g,13.20mmol)、DMF(40mL),升温至70℃搅拌17h,乙酸乙酯(100mL×3)萃取,用无水Na2SO4干燥,除去溶剂,进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到3.61g暗黄色油状物,产率:84.9%。At room temperature, (R)-(2-methyl-1-oxo-1-((2,2,2-trifluoroethyl)amino)butyl-2-yl)carbamate (3.57g, 12.01 g mmol) was dissolved in an ethyl acetate solution of hydrogen chloride (15 mL), stirred at room temperature for 5 h, and the reaction solution was concentrated under reduced pressure. Under nitrogen protection, cuprous iodide (1.60g, 8.40mmol), cesium carbonate (11.80g, 36.20mmol), L-proline (970mg, 8.43mmol), m-bromoiodobenzene (3.73g, 13.20mmol) were added , DMF (40 mL), heated to 70 °C, stirred for 17 h, extracted with ethyl acetate (100 mL×3), dried with anhydrous Na 2 SO 4 , removed the solvent, and carried out column separation (petroleum ether/ethyl acetate (v/v) =2/1) 3.61 g of dark yellow oil were obtained, yield: 84.9%.
MS(ESI,pos.ion)m/z:355.2[M+1]+。MS(ESI, pos.ion) m/z: 355.2[M+1] + .
步骤4:化合物(R)-2-甲基-2-((3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基)-N-(2,2,2-三氟乙基) 丁酰胺的合成Step 4: Compound (R)-2-methyl-2-((3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl) )amino)-N-(2,2,2-trifluoroethyl)butyramide
氮气保护下,将(R)-2-((3-溴苯基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺(3.59g,10.21mmol)、联硼酸频哪醇酯(3.08g,12.11mmol)、醋酸钾(3.21g,33.02mmol)和Pd(dppf)Cl2(450mg,0.60mmol)加到一起,再加入DMSO(45.00mL),115℃反应17h,反应液冷却至室温,用乙酸乙酯萃取(100mL×3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=2/1)得到2.86g黑色油状物,产率:70.2%。Under nitrogen protection, (R)-2-((3-bromophenyl)amino)-2-methyl-N-(2,2,2-trifluoroethyl)butanamide (3.59 g, 10.21 mmol) , pinacol biboronate (3.08 g, 12.11 mmol), potassium acetate (3.21 g, 33.02 mmol) and Pd(dppf)Cl 2 (450 mg, 0.60 mmol) were added together, followed by DMSO (45.00 mL), 115 The reaction was carried out at ℃ for 17 h, the reaction solution was cooled to room temperature, extracted with ethyl acetate (100 mL×3), the organic phase was spin-dried, and separated by column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 2.86 g Black oil, yield: 70.2%.
MS(ESI,pos.ion)m/z:401.4[M+1]+。MS(ESI, pos.ion) m/z: 401.4[M+1] + .
采用相应的原料,按照中间体实施例38的合成方法制备如下所示的中间体:The intermediate shown below was prepared according to the synthetic method of Intermediate Example 38 using the corresponding raw materials:
中间体实施例40Intermediate Example 40
1-(2-氟苯基)-4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑1-(2-Fluorophenyl)-4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H-pyrazole
氮气保护下,将DMF(12mL)加入到邻氟溴苄(706mg,3.71mmol)、4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑(480mg,2.47mmol)、碳酸铯(2.10g,6.19mmol)和碘化钾(150mg,1.24mmol)混合物中,室温反应12h,用乙酸乙酯萃取(50mLx3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=10/1)得到400mg无色油状物,产率:53.5%。Under nitrogen, DMF (12 mL) was added to o-fluorobenzyl bromide (706 mg, 3.71 mmol), 4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentane-2 -yl)-1H-pyrazole (480mg, 2.47mmol), cesium carbonate (2.10g, 6.19mmol) and potassium iodide (150mg, 1.24mmol) mixture, react at room temperature for 12h, extract with ethyl acetate (50mLx3), spin dry The organic phase was separated by column chromatography (petroleum ether/ethyl acetate (v/v)=10/1) to obtain 400 mg of colorless oil, yield: 53.5%.
MS(ESI,pos.ion)m/z:303.3[M+1]+。MS(ESI, pos.ion) m/z: 303.3[M+1] + .
采用相应的原料,按照中间体实施例40的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 40:
中间体实施例46Intermediate Example 46
2-(1-(乙腈磺酰基)-3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑-1-基)氮杂环丁烷-3-基)乙腈2-(1-(acetonitrilesulfonyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H-pyrazole -1-yl)azetidin-3-yl)acetonitrile
步骤1:化合物3-(氰基乙基)-3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑-1-基)氮杂环丁烷-1-甲酸叔丁酯的合成Step 1: Compound 3-(cyanoethyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H- Synthesis of Pyrazol-1-yl)azetidine-1-carboxylate tert-butyl ester
向1H-吡唑硼酸脂(200mg,1.03mmol),3-(氰基亚甲基)氮杂环丁烷-1-甲酸叔丁酯(220mg,1.13mmol)的乙腈(8mL)溶液中加入DBU(80μL,0.53mmol),50℃加热反应4小时,直接浓缩,柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到340mg淡黄色油状物,产率:84.93%。To a solution of 1H-pyrazoleborate (200 mg, 1.03 mmol), tert-butyl 3-(cyanomethylene)azetidine-1-carboxylate (220 mg, 1.13 mmol) in acetonitrile (8 mL) was added DBU (80 μL, 0.53 mmol), heated at 50° C. for 4 hours, concentrated directly, column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) gave 340 mg of pale yellow oil, yield: 84.93%.
MS(ESI,pos.ion)m/z:333.3[M-55]+。MS (ESI, pos.ion) m/z: 333.3[M-55] + .
步骤2:化合物2-(1-(乙腈磺酰基)-3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑-1-基)氮杂环丁烷-3-基)乙腈的合成Step 2: Compound 2-(1-(acetonitrilesulfonyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)- Synthesis of 1H-pyrazol-1-yl)azetidine-3-yl)acetonitrile
向3-(氰基乙基)-3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑-1-基)氮杂环丁烷-1-甲酸叔丁酯(330mg,0.85mmol)的二氯甲烷(8mL)溶液中加入三氟乙酸(0.7mL,9mmol),室温搅拌2小时,浓缩溶剂,加入二氯甲烷(8mL)溶解,冰浴下加入Et3N(400μL,2.8mmol),乙基磺酰氯(0.15mL,1.6mmol),室温搅拌2小时,加水(30mL)淬灭,二氯甲烷萃取(30mL x 3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=1/1)得到120mg淡黄色油状物,产率:37.13%。To 3-(cyanoethyl)-3-(4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H-pyrazole- To a solution of tert-butyl 1-yl)azetidine-1-carboxylate (330 mg, 0.85 mmol) in dichloromethane (8 mL) was added trifluoroacetic acid (0.7 mL, 9 mmol), stirred at room temperature for 2 hours, concentrated the solvent, Add dichloromethane (8 mL) to dissolve, add Et 3 N (400 μL, 2.8 mmol), ethylsulfonyl chloride (0.15 mL, 1.6 mmol) under ice bath, stir at room temperature for 2 hours, add water (30 mL) to quench, dichloromethane Extraction (30 mL x 3), drying over anhydrous sodium sulfate, and concentration column chromatography (petroleum ether/ethyl acetate (v/v)=1/1) gave 120 mg of pale yellow oil, yield: 37.13%.
MS(ESI,pos.ion)m/z:381.2[M+1]+。MS(ESI, pos.ion) m/z: 381.2[M+1] + .
中间体实施例47Intermediate Example 47
1-甲酸叔丁酯-4-((4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡咯-1-基))哌啶tert-Butyl 1-carboxylate-4-((4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H-pyrrol-1-yl )) Piperidine
步骤1:化合物1-甲酸叔丁酯-4-甲烷磺酰氧基哌啶的合成Step 1: Synthesis of compound 1-tert-butyl formate-4-methanesulfonyloxypiperidine
冰浴下,将甲基磺酰氯(2.44g,21.3mmol,1.65mL)缓慢滴加到N-甲酸叔丁酯-4-羟基哌啶(3.10g,15.00mmol)和三乙胺(3.00g,30mmol,4.00mL)的二氯甲烷溶液中(40mL),滴加完毕后,室温下反应1.5h。向反应液加入用的盐酸(1M,40mL)淬灭反应,有机层用饱和食盐水(30mL)洗涤,无水硫酸钠干燥,减压浓缩,得到4.30g米白色固体,产率:100.0%。Under ice bath, methanesulfonyl chloride (2.44g, 21.3mmol, 1.65mL) was slowly added dropwise to N-tert-butyl formate-4-hydroxypiperidine (3.10g, 15.00mmol) and triethylamine (3.00g, 30mmol, 4.00mL) in dichloromethane solution (40mL), after the dropwise addition, the reaction was carried out at room temperature for 1.5h. To the reaction solution was added hydrochloric acid (1 M, 40 mL) to quench the reaction, the organic layer was washed with saturated brine (30 mL), dried over anhydrous sodium sulfate, and concentrated under reduced pressure to obtain 4.30 g of an off-white solid, yield: 100.0%.
MS(ESI,pos.ion)m/z:224.2[M-55]+;MS(ESI, pos.ion) m/z: 224.2[M-55] + ;
1H NMR(400MHz,CDCl3):δ(ppm)4.90(m,1H),3.72(m,2H),3.31(m,2H),3.05(s,3H),1.97(m,2H),1.84(m,2H),1.47(s,9H)。 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 4.90 (m, 1H), 3.72 (m, 2H), 3.31 (m, 2H), 3.05 (s, 3H), 1.97 (m, 2H), 1.84 (m, 2H), 1.47 (s, 9H).
步骤2:化合物1-甲酸叔丁酯-4-((4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡咯-1-基))哌啶的合成Step 2: Compound 1-tert-butylcarboxylate-4-((4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H-pyrrole Synthesis of -1-yl))piperidine
将1-甲酸叔丁酯-4-甲烷磺酰氧基哌啶(2.00g,7.16mmol)加入到4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑(1.50g,7.70mmol)和碳酸铯(3.50g,11.00mmol)的N,N-二甲基甲酰胺(15mL)的溶液中,反应液100℃反应24h,冷却至室温,用水(100mL)和乙酸乙酯(100mL)萃取,水相用乙酸乙酯(100mL x 3)萃取,合并有机相后用饱和食盐水(100mL x 3)洗涤,无水硫酸钠干燥,减压浓缩,浓缩液拌样过柱(石油醚/乙酸乙酯(v/v)=2/1)得到950mg无色固体,产率:35.2%。tert-Butyl 1-carboxylate-4-methanesulfonyloxypiperidine (2.00 g, 7.16 mmol) was added to 4-(4,4,5,5-tetramethyl-1,3,2-dioxoboron) Pentan-2-yl)-1H-pyrazole (1.50g, 7.70mmol) and cesium carbonate (3.50g, 11.00mmol) in N,N-dimethylformamide (15mL) solution, reaction solution 100 ℃ The reaction was carried out for 24 h, cooled to room temperature, extracted with water (100 mL) and ethyl acetate (100 mL), the aqueous phase was extracted with ethyl acetate (100 mL x 3), the organic phases were combined and washed with saturated brine (100 mL x 3), anhydrous It was dried over sodium sulfate, concentrated under reduced pressure, and the concentrated solution was mixed and passed through a column (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 950 mg of a colorless solid, yield: 35.2%.
MS(ESI,pos.ion)m/z:378.4[M+1]+;MS(ESI, pos.ion) m/z: 378.4[M+1] + ;
1H NMR(400MHz,CDCl3):δ(ppm)7.81(s,1H),7.74(s,1H),4.28(m,3H),2.90(m,2H),2.14(m,2H),1.92(m,2H),1.49(s,9H),1.33(s,12H)。 1 H NMR (400 MHz, CDCl 3 ): δ (ppm) 7.81 (s, 1H), 7.74 (s, 1H), 4.28 (m, 3H), 2.90 (m, 2H), 2.14 (m, 2H), 1.92 (m, 2H), 1.49 (s, 9H), 1.33 (s, 12H).
采用相应的原料,按照中间体实施例47的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 47:
中间体实施例49Intermediate Example 49
6-氟-1-甲基-3-(三正丁基锡基)-1H-吲唑6-Fluoro-1-methyl-3-(tri-n-butyltinyl)-1H-indazole
步骤1:化合物6-氟-3-碘-1H-吲唑的合成Step 1: Synthesis of compound 6-fluoro-3-iodo-1H-indazole
室温下,向6-氟-1H-吲唑(97mg,0.71mmol)的DMF(5mL)溶液中,依次加入氢氧化钾(130mg,2.32mmol)和碘单质(280mg,1.10mmol),室温搅拌11h,饱和硫代硫酸钠溶液溶液(10mL)淬灭,乙酸乙酯萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=6/1)得到150mg棕色固体,产率:80.34%。To a solution of 6-fluoro-1H-indazole (97 mg, 0.71 mmol) in DMF (5 mL) at room temperature, potassium hydroxide (130 mg, 2.32 mmol) and iodine (280 mg, 1.10 mmol) were sequentially added, and the mixture was stirred at room temperature for 11 h. , quenched with saturated sodium thiosulfate solution (10 mL), extracted with ethyl acetate (15 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=6/1) to obtain 150 mg of brown solid, yield: 80.34%.
MS(ESI,pos.ion)m/z:263.0[M+1]+。MS(ESI, pos.ion) m/z: 263.0[M+1] + .
步骤2:化合物6-氟-3-碘-1-甲基-1H-吲唑的合成Step 2: Synthesis of compound 6-fluoro-3-iodo-1-methyl-1H-indazole
室温下,向6-氟-3-碘-1H-吲唑(97mg,0.37mmol)的DMSO(5mL)溶液中,依次加入碳酸铯(60mg,0.18mmol)和碘甲烷(70μL,0.76mmol),室温搅拌1.5小时,加水(10mL)淬灭,乙酸乙酯萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=6/1)得到100mg黄色固体,产率:99%。To a solution of 6-fluoro-3-iodo-1H-indazole (97 mg, 0.37 mmol) in DMSO (5 mL) at room temperature, cesium carbonate (60 mg, 0.18 mmol) and methyl iodide (70 μL, 0.76 mmol) were sequentially added, Stir at room temperature for 1.5 hours, add water (10 mL) to quench, extract with ethyl acetate (15 mL×3), dry over anhydrous sodium sulfate, and concentrate by column chromatography (petroleum ether/ethyl acetate (v/v)=6/1) to obtain 100 mg of yellow Solid, yield: 99%.
MS(ESI,pos.ion)m/z:277.0[M+1]+。MS(ESI, pos.ion) m/z: 277.0[M+1] + .
步骤3:化合物6-氟-1-甲基-3-(三正丁基锡基)-1H-吲唑的合成Step 3: Synthesis of compound 6-fluoro-1-methyl-3-(tri-n-butyltinyl)-1H-indazole
-16℃下,向6-氟-3-碘-1-甲基-1H-吲唑(4.1g,15mmol)的四氢呋喃(30mL)溶液中,依次加入异丙基氯化镁(2M,9mL,18mmol),在此温度下搅拌20分钟后加入三正丁基氯化锡(4.8mL,18mmol)后,自然升至室温后继续搅拌5小时,加入饱和氯化铵溶液(30mL)淬灭,乙酸乙酯萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=8/1)得到6.1g淡黄色油状物,产率:94%。To a solution of 6-fluoro-3-iodo-1-methyl-1H-indazole (4.1 g, 15 mmol) in tetrahydrofuran (30 mL) at -16 °C, was sequentially added isopropylmagnesium chloride (2M, 9 mL, 18 mmol) , after stirring at this temperature for 20 minutes, after adding tri-n-butyltin chloride (4.8mL, 18mmol), naturally rising to room temperature and continuing to stir for 5 hours, adding saturated ammonium chloride solution (30mL) to quench, ethyl acetate Extraction (15 mL×3), drying over anhydrous sodium sulfate, and concentration column chromatography (petroleum ether/ethyl acetate (v/v)=8/1) gave 6.1 g of pale yellow oil, yield: 94%.
MS(ESI,pos.ion)m/z:440.2[M+1]+。MS(ESI, pos.ion) m/z: 440.2[M+1] + .
中间体实施例50Intermediate Example 50
N-(3-(2-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)-2-氰基乙酰胺N-(3-(2-Bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)benzene yl)-2-cyanoacetamide
向2-溴-7-碘-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(30mg,0.07mmol)、联硼酸频哪醇酯(25mg,0.08mmol)、碳酸钠(20mg,0.19mmol)和二氯二(三苯基膦)钯(6mg,0.01mmol)的混合物中依次加入乙腈(4mL)和水(1mL),氮气氛围下,65℃加热反应4.5小时。加入饱和氯化铵溶液(10mL)淬 灭,乙酸乙酯萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到29mg黄色固体,产率:90%。To 2-bromo-7-iodo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (30 mg, 0.07 mmol), Acetonitrile (4 mL) and water ( 1 mL), heated at 65°C for 4.5 hours under nitrogen atmosphere. Saturated ammonium chloride solution (10 mL) was added to quench, extracted with ethyl acetate (15 mL×3), dried over anhydrous sodium sulfate, and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 29 mg of yellow Solid, yield: 90%.
MS(ESI,pos.ion)m/z:487.4[M+1]+。MS(ESI, pos.ion) m/z: 487.4[M+1] + .
采用相应的原料,按照中间体实施例50的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 50:
中间体实施例65Intermediate Example 65
(R)-2-((2-(2-氯-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺(R)-2-((2-(2-Chloro-5-trityl-5H-pyrrolo[2,3-b]pyrazin-7-yl)pyrimidin-4-yl)amino)-2 -Methyl-N-(2,2,2-trifluoroethyl)butanamide
步骤1:化合物(R)-2-((2-氯嘧啶-4-基)氨基)-2-甲基丁酸的合成Step 1: Synthesis of compound (R)-2-((2-chloropyrimidin-4-yl)amino)-2-methylbutanoic acid
向2,4-二氯嘧啶(600mg,4.02mmol)的异丙醇(8mL)溶液中依次加入R-异缬氨酸(550mg,4.55mmol),碳酸钾(850mg,6.15mmol),90℃回流反应5.5小时,直接加入硅胶拌样,柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到600mg白色固体,产率:93%。To the isopropanol (8 mL) solution of 2,4-dichloropyrimidine (600 mg, 4.02 mmol) was added R-isovaline (550 mg, 4.55 mmol), potassium carbonate (850 mg, 6.15 mmol) successively, and refluxed at 90°C After the reaction was carried out for 5.5 hours, silica gel was directly added to mix the sample, and column chromatography (petroleum ether/ethyl acetate (v/v)=0/1) was used to obtain 600 mg of white solid, yield: 93%.
MS(ESI,pos.ion)m/z:230.0[M+1]+。MS(ESI, pos.ion) m/z: 230.0[M+1] + .
步骤2:化合物(R)-2-((2-氯嘧啶-4-基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺的合成Step 2: Synthesis of compound (R)-2-((2-chloropyrimidin-4-yl)amino)-2-methyl-N-(2,2,2-trifluoroethyl)butanamide
0℃,向(R)-2-((2-氯嘧啶-4-基)氨基)-2-甲基丁酸(50mg,0.21mmol),2,2,2-三氟乙胺盐酸盐(44mg,0.32mmol)和二异丙基乙基胺(120μL,0.68mmol)的N,N-二甲基甲酰胺(4mL)溶液中加入50%丙基磷酸酐乙酸乙酯溶液(510mg,0.80mmol),室温搅拌12小时,加水(15mL)稀释,二氯甲烷萃取(20mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到30mg淡黄色固体,产率:44.35%。0 °C, to (R)-2-((2-chloropyrimidin-4-yl)amino)-2-methylbutanoic acid (50 mg, 0.21 mmol), 2,2,2-trifluoroethylamine hydrochloride (44 mg, 0.32 mmol) and diisopropylethylamine (120 μL, 0.68 mmol) in N,N-dimethylformamide (4 mL) was added a 50% solution of propylphosphoric anhydride in ethyl acetate (510 mg, 0.80 mmol), stirred at room temperature for 12 hours, diluted with water (15 mL), extracted with dichloromethane (20 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 30 mg of pale yellow solid, yield: 44.35%.
MS(ESI,pos.ion)m/z:311.1[M+1]+。MS(ESI, pos.ion) m/z: 311.1[M+1] + .
步骤3:化合物2-氯-7-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-5-三苯基甲基-5H-吡咯并[2,3-b]吡嗪的合成Step 3: Compound 2-chloro-7-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-5-triphenylmethyl-5H- Synthesis of pyrrolo[2,3-b]pyrazine
-20℃,向2-氯-7-碘-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪(380mg,0.73mmol),异丙醇频哪醇硼酸酯(200μL,0.98mmol)的四氢呋喃(8mL)溶液中慢慢加入异丙基氯化镁的四氢呋喃溶液(2.0M,850μL,1.1mmol),在此温度下继续反应1.5小时,加入乙酸乙酯(20mL)稀释,加入饱和氯化铵溶液(20mL)淬灭,乙酸乙酯萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=10/1)得到320mg淡黄色固体,产率:84.21%。-20 °C, to 2-chloro-7-iodo-5-trityl-5H-pyrrolo[2,3-b]pyrazine (380 mg, 0.73 mmol), isopropanol pinacol borate ( 200 μL, 0.98 mmol) in tetrahydrofuran (8 mL) was slowly added isopropylmagnesium chloride in tetrahydrofuran (2.0 M, 850 μL, 1.1 mmol), the reaction was continued at this temperature for 1.5 hours, and ethyl acetate (20 mL) was added to dilute, Saturated ammonium chloride solution (20 mL) was added to quench, extracted with ethyl acetate (25 mL×3), dried over anhydrous sodium sulfate, and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=10/1) to obtain 320 mg of light Yellow solid, yield: 84.21%.
MS(ESI,pos.ion)m/z:521.8[M+1]+。MS(ESI, pos.ion) m/z: 521.8[M+1] + .
步骤4:化合物(R)-2-((2-(2-氯-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-2-甲基-N-(2,2,2- 三氟乙基)丁酰胺的合成Step 4: Compound (R)-2-((2-(2-Chloro-5-trityl-5H-pyrrolo[2,3-b]pyrazin-7-yl)pyrimidin-4-yl) Synthesis of Amino)-2-methyl-N-(2,2,2-trifluoroethyl)butanamide
向2-氯-7-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-5-三苯基甲基-5H-吡咯并[2,3-b]吡嗪(30mg,0.057mmol)的1,4-二氧六环(4mL)溶液中依次加入(R)-2-((2-氯嘧啶-4-基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺(17mg,0.05mmol),碳酸钾(12mg,0.08mmol),Pd(dppf)Cl2(5mg,0.01mmol)和水(1mL),氮气氛围下,110℃回流反应4.5小时,加入饱和氯化铵溶液(20mL)淬灭,二氯甲烷萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到220mg淡黄色油状物,产率:46.72%。To 2-chloro-7-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-5-triphenylmethyl-5H-pyrrolo[ To a solution of 2,3-b]pyrazine (30 mg, 0.057 mmol) in 1,4-dioxane (4 mL) was added (R)-2-((2-chloropyrimidin-4-yl)amino)- 2-methyl-N-(2,2,2-trifluoroethyl)butanamide (17 mg, 0.05 mmol), potassium carbonate (12 mg, 0.08 mmol), Pd(dppf)Cl 2 (5 mg, 0.01 mmol) and Water (1 mL), under nitrogen atmosphere, refluxed at 110°C for 4.5 hours, quenched by adding saturated ammonium chloride solution (20 mL), extracted with dichloromethane (25 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ Ethyl acetate (v/v)=2/1) gave 220 mg of pale yellow oil, yield: 46.72%.
MS(ESI,pos.ion)m/z:669.7[M+1]+。MS(ESI, pos.ion) m/z: 669.7[M+1] + .
采用相应的原料,按照中间体实施例65的合成方法制备如下所示的中间体:Using the corresponding raw materials, the following intermediates were prepared according to the synthetic method of Intermediate Example 65:
中间体实施例67Intermediate Example 67
N-异丙基-5-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡唑并[2,3-b]吡啶-3-甲酰胺N-Isopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1-((2-(trimethylsilyl )ethoxy)methyl)-1H-pyrazolo[2,3-b]pyridine-3-carboxamide
步骤1:化合物5-溴-1H-吡咯并[2,3-b]吡啶-3-羧酸的合成Step 1: Synthesis of compound 5-bromo-1H-pyrrolo[2,3-b]pyridine-3-carboxylic acid
将5-溴-7-氮杂吲哚(10.00g,50.75mmol)溶于二氯甲烷(200mL)中,加入三氯化铝(34.00g,254.99mmol),室温搅拌10分钟,加入三氯乙酰氯(8.6mL,77mmol),继续室温搅拌过夜,将反应液倒入冰水中,充分搅拌,过滤得到白色固体,滤液用乙酸乙酯萃取(200mL x3),浓缩合并过滤出来的白色固体,加入四氢呋喃(250mL)和水(125mL)溶解,加入三乙胺(50mL),室温搅拌24h,将溶剂浓缩蒸干,用1.0M稀盐酸调pH至5-6,过滤,50℃真空干燥,得到产物为8.20g灰白色固体,产率:67.0%。Dissolve 5-bromo-7-azaindole (10.00 g, 50.75 mmol) in dichloromethane (200 mL), add aluminum trichloride (34.00 g, 254.99 mmol), stir at room temperature for 10 minutes, add trichloroethyl Acyl chloride (8.6 mL, 77 mmol), continued stirring at room temperature overnight, poured the reaction solution into ice water, stirred well, filtered to obtain a white solid, the filtrate was extracted with ethyl acetate (200 mL x 3), concentrated and combined the filtered white solid, added tetrahydrofuran (250 mL) and water (125 mL) were dissolved, triethylamine (50 mL) was added, stirred at room temperature for 24 h, the solvent was concentrated and evaporated to dryness, the pH was adjusted to 5-6 with 1.0 M dilute hydrochloric acid, filtered, and dried under vacuum at 50 °C to obtain the product as 8.20 g off-white solid, yield: 67.0%.
MS(ESI,pos.ion)m/z:241.2[M+1]+。MS(ESI, pos.ion) m/z: 241.2[M+1] + .
步骤2:化合物5-溴-N-异丙基-1H-吡咯并[2,3-b]吡啶-3-甲酰胺的合成Step 2: Synthesis of compound 5-bromo-N-isopropyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide
向5-溴-1H-吡咯并[2,3-b]吡啶-3-羧酸(600mg,2.49mmol)的N,N-二甲基甲酰胺(20mL)溶液中依次加入1-(3-二甲氨基丙基)-3-乙基碳二亚胺盐酸盐(1.44g,7.43mmol),1-羟基苯并三氮唑(1.01g,7.46mmol)和2-异丙胺(1.27mL,14.90mmol),室温搅拌过夜。加入饱和食盐水(20mL),二氯甲烷(15mLx 3)萃取,用无水硫酸钠干燥,除去溶剂,浓缩液进行柱色谱分离(淋洗剂:石油醚/乙酸乙酯(v/v)=5/1)得到产物为360mg棕色固体,产率:51.3%。To a solution of 5-bromo-1H-pyrrolo[2,3-b]pyridine-3-carboxylic acid (600 mg, 2.49 mmol) in N,N-dimethylformamide (20 mL) was sequentially added 1-(3- Dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (1.44 g, 7.43 mmol), 1-hydroxybenzotriazole (1.01 g, 7.46 mmol) and 2-isopropylamine (1.27 mL, 14.90 mmol) and stirred at room temperature overnight. Saturated brine (20 mL) was added, extracted with dichloromethane (15 mL×3), dried over anhydrous sodium sulfate, the solvent was removed, and the concentrated solution was subjected to column chromatography (eluent: petroleum ether/ethyl acetate (v/v)= 5/1) The product was obtained as 360 mg of brown solid, yield: 51.3%.
MS(ESI,pos.ion)m/z:282.0[M+1]+。MS(ESI, pos.ion) m/z: 282.0[M+1] + .
步骤3:化合物5-溴-N-异丙基-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡啶-3-甲酰胺的合成Step 3: Compound 5-Bromo-N-isopropyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-3- Synthesis of formamide
将5-溴-N-异丙基-1H-吡咯并[2,3-b]吡啶-3-甲酰胺(360mg,1.28mmol)溶于N,N-二甲基甲酰胺(20mL)中,冰浴下,加入60%氢化钠(102.1mg,2.55mmol),室温搅拌0.5h后加入2-(三甲基硅基)乙氧基甲基 氯(338.7μL,1.82mmol),继续室温搅拌过夜,加入饱和食盐水(20mL),二氯甲烷(15mL x 3)萃取,用无水硫酸钠干燥,除去溶剂,浓缩液进行柱色谱分离(淋洗剂:石油醚/乙酸乙酯(v/v)=6/1)得到产物为500mg棕色固体,产率:99.0%。5-Bromo-N-isopropyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide (360 mg, 1.28 mmol) was dissolved in N,N-dimethylformamide (20 mL), Under ice bath, 60% sodium hydride (102.1 mg, 2.55 mmol) was added, stirred at room temperature for 0.5 h, then 2-(trimethylsilyl)ethoxymethyl chloride (338.7 μL, 1.82 mmol) was added, and stirring was continued at room temperature overnight , added saturated brine (20 mL), extracted with dichloromethane (15 mL x 3), dried with anhydrous sodium sulfate, removed the solvent, and the concentrated solution was subjected to column chromatography (eluent: petroleum ether/ethyl acetate (v/v )=6/1) The product was obtained as 500 mg of brown solid, yield: 99.0%.
MS(ESI,pos.ion)m/z:412.1[M+1]+。MS(ESI, pos.ion) m/z: 412.1[M+1] + .
步骤4:化合物N-异丙基-5-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡唑并[2,3-b]吡啶-3-甲酰胺的合成Step 4: Compound N-isopropyl-5-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1-((2-(tri Synthesis of methylsilyl)ethoxy)methyl)-1H-pyrazolo[2,3-b]pyridine-3-carboxamide
将5-溴-N-异丙基-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡啶-3-甲酰胺(600mg,1.45mmol)溶于1,4-二氧六环(10mL)中,依次加入联硼酸频哪醇酯(443.7mg,1.74mmol),碳酸钾(357.3mg,3.64mmol)和Pd(dppf)Cl2(106.4mg,0.14mmol),氮气保护,115℃回流反应4小时,冷却至室温,硅藻土过滤,浓缩滤液,浓缩液进行柱色谱分离(淋洗剂:石油醚/乙酸乙酯(v/v)=2/1)得到产物为610mg黄色固体,产率:91.25%。5-Bromo-N-isopropyl-1-((2-(trimethylsilyl)ethoxy)methyl)-1H-pyrrolo[2,3-b]pyridine-3-carboxamide ( 600mg, 1.45mmol) was dissolved in 1,4-dioxane (10mL), followed by adding pinacol biboronate (443.7mg, 1.74mmol), potassium carbonate (357.3mg, 3.64mmol) and Pd (dppf) Cl 2 (106.4 mg, 0.14 mmol), under nitrogen protection, refluxed at 115°C for 4 hours, cooled to room temperature, filtered through celite, concentrated filtrate, and the concentrated solution was subjected to column chromatography (eluent: petroleum ether/ethyl acetate ( v/v)=2/1) The product was obtained as 610 mg of yellow solid, yield: 91.25%.
MS(ESI,pos.ion)m/z:460.30[M+1]+。MS(ESI, pos.ion) m/z: 460.30[M+1] + .
实施例1Example 1
3-((3-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈3-((3-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile
步骤1:化合物3-((3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 1: Compound 3-((3-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- Synthesis of 5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile
向7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(30mg,0.07mmol)的1,4-二氧六环(4mL)溶液中依次加入3-((3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基)丙腈(24mg,0.08mmol),碳酸钾(16mg,0.12mmol),Pd(dppf)Cl2(10mg,0.01mmol)和水(1mL),氮气氛围下,110℃加热反应7小时,加水(10mL)稀释,二氯甲烷萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到26mg棕色油状物,产率:74.74%。To 7-bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2 ,3-b]pyrazine (30mg, 0.07mmol) in 1,4-dioxane (4mL) solution was added 3-((3-(4,4,5,5-tetramethyl-1, 3,2-Dioxaboropentan-2-yl)phenyl)amino)propionitrile (24 mg, 0.08 mmol), potassium carbonate (16 mg, 0.12 mmol), Pd(dppf)Cl 2 (10 mg, 0.01 mmol) and Water (1 mL), heated at 110°C for 7 hours under nitrogen atmosphere, diluted with water (10 mL), extracted with dichloromethane (15 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/ v)=2/1) 26 mg of brown oil, yield: 74.74%.
MS(ESI,pos.ion)m/z:474.3[M+1]+。MS(ESI, pos.ion) m/z: 474.3[M+1] + .
步骤2:化合物3-((3-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 2: Compound 3-((3-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl) Synthesis of Amino)Propionitrile
向3-((3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(260mg,0.55mmol)的二氯甲烷(6mL)溶液中,加入三氟乙酸(2.5mL),室温搅拌2h,浓缩溶剂,加入四氢呋喃(6mL)溶解,滴加氢氧化钠溶液(2N)至pH>7,室温搅拌2h,加水(20mL)稀释,二氯甲烷萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到50mg黄色固体,产率:31.83%。To 3-((3-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrole To [2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile (260 mg, 0.55 mmol) in dichloromethane (6 mL) was added trifluoroacetic acid (2.5 mL), and stirred at room temperature for 2 h , concentrated the solvent, added tetrahydrofuran (6 mL) to dissolve, added dropwise sodium hydroxide solution (2N) to pH>7, stirred at room temperature for 2 h, added water (20 mL) to dilute, extracted with dichloromethane (15 mL×3), dried over anhydrous sodium sulfate, concentrated Column chromatography (petroleum ether/ethyl acetate (v/v)=0/1) gave 50 mg of a yellow solid, yield: 31.83%.
MS(ESI,pos.ion)m/z:344.2[M+1]+;MS(ESI, pos.ion) m/z: 344.2[M+1] + ;
1H NMR(600MHz,DMSO-d6)δ(ppm)12.10(s,1H),8.62(m,1H),8.37(s,1H),8.26(s,1H),8.15(s,1H),7.67(m,1H),7.46(d,J=7.7Hz,1H),7.15(m,1H),6.52(dd,J=8.0,1.7Hz,1H),5.95(t,J=6.2Hz,1H),3.94(s,3H),3.46(dd,J=12.9,6.5Hz,2H),2.80(t,J=6.6Hz,2H); 1 H NMR (600MHz, DMSO-d 6 )δ(ppm) 12.10(s,1H), 8.62(m,1H), 8.37(s,1H), 8.26(s,1H), 8.15(s,1H), 7.67(m, 1H), 7.46(d, J=7.7Hz, 1H), 7.15(m, 1H), 6.52(dd, J=8.0, 1.7Hz, 1H), 5.95(t, J=6.2Hz, 1H) ), 3.94(s, 3H), 3.46(dd, J=12.9, 6.5Hz, 2H), 2.80(t, J=6.6Hz, 2H);
13C NMR(150MHz,DMSO-d6)δ(ppm)148.3,141.8,140.9,137.3,136.3,135.0,134.4,129.6,129.3,128.3,121.8,120.2,114.8,113.8,110.7,110.2,40.5,39.2,17.9。 13 C NMR (150MHz, DMSO-d 6 )δ(ppm) 148.3, 141.8, 140.9, 137.3, 136.3, 135.0, 134.4, 129.6, 129.3, 128.3, 121.8, 120.2, 114.8, 113.8, 110.7, 110.2, 40.5, 39 , 17.9.
采用相应的原料,按照实施例1的合成方法制备如下所示的化合物:Using the corresponding raw materials, the following compounds were prepared according to the synthetic method of Example 1:
实施例2Example 2
3-((3-(2-环丙基-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙酰胺3-((3-(2-Cyclopropyl-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propanamide
步骤1:化合物3-((3-(2-环丙基-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 1: Compound 3-((3-(2-Cyclopropyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyridine Synthesis of oxazin-7-yl)phenyl)amino)propionitrile
氮气保护下,将甲苯(10mL)和水(1.2mL)加入到7-溴-2-环丙基-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(200mg,0.42mmol)、环丙基硼酸(72mg,0.85mmol)、七水磷酸钾(440mg,1.36mmol)、醋酸钯(6mg,0.02mmol)、三环己基磷(15mg,0.04mmol)混合物中,110℃回流反应19h,反应液冷却至室温,硅藻土过滤,滤液减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=4/1)得到160mg浅红色固体,产率:87.2%。Under nitrogen, toluene (10 mL) and water (1.2 mL) were added to 7-bromo-2-cyclopropyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H- Pyrrolo[2,3-b]pyrazine (200mg, 0.42mmol), cyclopropylboronic acid (72mg, 0.85mmol), potassium phosphate heptahydrate (440mg, 1.36mmol), palladium acetate (6mg, 0.02mmol), tris Cyclohexylphosphorus (15mg, 0.04mmol) mixture was refluxed at 110°C for 19h, the reaction solution was cooled to room temperature, filtered through celite, the filtrate was concentrated under reduced pressure, and subjected to column separation (petroleum ether/ethyl acetate (v/v)= 4/1) 160 mg of light red solid were obtained, yield: 87.2%.
MS(ESI,pos.ion)m/z:434.3[M+1]+。MS(ESI, pos.ion) m/z: 434.3[M + 1] + .
步骤2:化合物3-((3-(2-环丙基-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙酰胺的合成Step 2: Synthesis of compound 3-((3-(2-cyclopropyl-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionamide
室温下,3-((3-(2-环丙基-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(158mg,0.36mmol)溶解于二氯甲烷(6mL)中,加入三氟乙酸(6mL),室温下搅拌反应12h。减压浓缩反应液,加入二氯甲烷(6mL)、乙二胺(1mL),室温下搅拌2h,反应液加入硅胶,直接旋干拌样,柱层析分离(二氯甲烷/甲醇(v/v)=50/1)得到86mg浅黄色固体,产率:77.8%。3-((3-(2-Cyclopropyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine at room temperature -7-yl)phenyl)amino)propionitrile (158 mg, 0.36 mmol) was dissolved in dichloromethane (6 mL), trifluoroacetic acid (6 mL) was added, and the reaction was stirred at room temperature for 12 h. The reaction solution was concentrated under reduced pressure, dichloromethane (6 mL) and ethylenediamine (1 mL) were added, stirred at room temperature for 2 h, silica gel was added to the reaction solution, the sample was directly spin-dried, and the sample was separated by column chromatography (dichloromethane/methanol (v/ v)=50/1) 86 mg of pale yellow solid were obtained, yield: 77.8%.
MS(ESI,pos.ion)m/z:304.1[M+1]+;MS(ESI, pos.ion) m/z: 304.1[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)11.99(s,1H),8.26(s,1H),8.21(s,1H),7.54(s,1H),7.36(d,J=7.6Hz,1H),7.13(t,J=7.8Hz,1H),6.50(dd,J1=8.0Hz,J2=1.9Hz,1H),5.89(t,J=6.2Hz,1H),3.43(q,J=6.6Hz,2H),2.79(t,J=6.6Hz,2H),2.32–2.28(m,1H),1.05–1.02(m,4H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 11.99(s, 1H), 8.26(s, 1H), 8.21(s, 1H), 7.54(s, 1H), 7.36(d, J= 7.6Hz,1H),7.13(t,J=7.8Hz,1H),6.50(dd,J1 = 8.0Hz,J2=1.9Hz,1H ) ,5.89(t,J=6.2Hz,1H),3.43 (q, J=6.6Hz, 2H), 2.79 (t, J=6.6Hz, 2H), 2.32–2.28 (m, 1H), 1.05–1.02 (m, 4H);
13C NMR(150MHz,DMSO-d6):δ(ppm)151.1,148.2,141.3,136.3,136.0,135.2,129.5,127.9,120.1,114.7,113.5,111.3,109.5,39.6,18.0,14.8,10.3。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 151.1, 148.2, 141.3, 136.3, 136.0, 135.2, 129.5, 127.9, 120.1, 114.7, 113.5, 111.3, 109.5, 39.6, 18.0, 14.8, 10.3.
采用相应的原料,按照实施例2的合成方法制备如下所示的化合物:Using the corresponding raw materials, the following compounds were prepared according to the synthetic method of Example 2:
实施例3Example 3
3-((3-(2-(6-氟-1-甲基-1H-吲唑-3-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈3-((3-(2-(6-Fluoro-1-methyl-1H-indazol-3-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl) amino)propionitrile
步骤1:化合物3-((3-(2-(6-氟-1-甲基-1H-吲唑-3-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b] 吡嗪-7-基)苯基)氨基)丙腈的合成Step 1: Compound 3-((3-(2-(6-Fluoro-1-methyl-1H-indazol-3-yl)-5-((2-(trimethylsilyl)ethoxy) Synthesis of methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile
氮气保护下,将3-((3-(2-溴-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(200mg,0.42mmol)、6-氟-1-甲基-3-(三丁基锡烷)-1H-吲唑(380mg,0.85mmol)、碘化亚铜(16mg,0.08mmol)、四三苯基膦钯(26mg,0.02mmol)加到一起,再加入DMF(15mL),升温至90℃反应4.5h,反应液冷却至室温,乙酸乙酯(50mL×3)萃取,用无水Na2SO4干燥,除去溶剂,进行柱分离(石油醚/乙酸乙酯(v/v)=4/1)得到200mg浅黄色油状物,产率:84.7%。Under nitrogen protection, 3-((3-(2-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine -7-yl)phenyl)amino)propionitrile (200 mg, 0.42 mmol), 6-fluoro-1-methyl-3-(tributylstannane)-1H-indazole (380 mg, 0.85 mmol), iodide Copper (16 mg, 0.08 mmol) and palladium tetrakistriphenylphosphine (26 mg, 0.02 mmol) were added together, then DMF (15 mL) was added, the temperature was raised to 90° C. for 4.5 h, the reaction solution was cooled to room temperature, and ethyl acetate (50 mL) was added. ×3) extraction, drying with anhydrous Na 2 SO 4 , removing the solvent, and performing column separation (petroleum ether/ethyl acetate (v/v)=4/1) to obtain 200 mg of pale yellow oil, yield: 84.7%.
MS(ESI,pos.ion)m/z:542.3[M+1]+。MS(ESI, pos.ion) m/z: 542.3[M+1] + .
步骤2:化合物3-((3-(2-(6-氟-1-甲基-1H-吲唑-3-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 2: Compound 3-((3-(2-(6-Fluoro-1-methyl-1H-indazol-3-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl Synthesis of )phenyl)amino)propionitrile
3-((3-(2-(6-氟-1-甲基-1H-吲唑-3-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(240mg,0.44mmol)按照实施例2步骤2的合成方法制备得到180mg浅黄色固体,产率:96.2%。3-((3-(2-(6-Fluoro-1-methyl-1H-indazol-3-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- 5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile (240 mg, 0.44 mmol) was prepared according to the synthetic method of Example 2, step 2 to obtain 180 mg of a pale yellow solid, yield: 96.2%.
MS(ESI,pos.ion)m/z:412.2[M+1]+;MS(ESI, pos.ion) m/z: 412.2[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.34(s,1H),9.05(s,1H),8.71(dd,J1=8.7Hz,J2=5.5Hz,1H),8.33(s,1H),7.63(dd,J1=9.6Hz,J2=1.5Hz,1H),7.53(d,J=7.5Hz,1H),7.49(s,1H),7.30–7.19(m,2H),6.60(d,J=7.9Hz,1H),5.92(t,J=6.0Hz,1H),4.14(s,3H),3.46(dd,J1=12.6Hz,J2=6.3Hz,1H),2.79(t,J=6.5Hz,2H); 1 H NMR (600 MHz, DMSO-d 6 ): δ (ppm) 12.34 (s, 1H), 9.05 (s, 1H), 8.71 (dd, J 1 =8.7 Hz, J 2 =5.5 Hz, 1H), 8.33 (s, 1H), 7.63 (dd, J 1 =9.6 Hz, J 2 =1.5 Hz, 1 H), 7.53 (d, J = 7.5 Hz, 1 H), 7.49 (s, 1 H), 7.30–7.19 (m, 2H), 6.60(d, J=7.9Hz, 1H), 5.92(t, J=6.0Hz, 1H), 4.14(s, 3H), 3.46(dd, J1 = 12.6Hz , J2=6.3Hz, 1H), 2.79(t, J=6.5Hz, 2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)162.9,161.3,148.6,142.4,142.1,142.1,141.8,141.4,135.9,135.2,134.8,129.8,128.8,125.1,125.0,120.2,119.0,115.4,114.8,111.8,111.6,111.4,110.1,96.4,96.2,39.5,36.3,18.0。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 162.9, 161.3, 148.6, 142.4, 142.1, 142.1, 141.8, 141.4, 135.9, 135.2, 134.8, 129.8, 128.8, 125.1, 125.0, 120.2, 119.0 115.4, 114.8, 111.8, 111.6, 111.4, 110.1, 96.4, 96.2, 39.5, 36.3, 18.0.
采用相应的原料,按照实施例3的合成方法制备如下所示的化合物:Using the corresponding raw materials, the following compounds were prepared according to the synthetic method of Example 3:
实施例16Example 16
1-(氰甲基)-3-(4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)脲1-(Cyanomethyl)-3-(4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl) phenyl)urea
步骤1:化合物4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯胺的合成Step 1: Compound 4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)aniline
4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯胺(120mg,0.55mmol)、7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(114mg,0.28mmol)、碳酸钾(115mg,0.82mmol)和Pd(dppf)Cl2(13mg,0.02mmol)按照实施例1步骤1的合成方法制备得到104mg黄色固体,产率:88.6%。4-(4,4,5,5-Tetramethyl-1,3,2-dioxaboropentan-2-yl)aniline (120 mg, 0.55 mmol), 7-bromo-2-(1-methyl) -1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (114 mg, 0.28 mmol ), potassium carbonate (115 mg, 0.82 mmol) and Pd(dppf)Cl 2 (13 mg, 0.02 mmol) were prepared according to the synthetic method of Example 1, step 1 to obtain 104 mg of a yellow solid, yield: 88.6%.
MS(ESI,pos.ion)m/z:421.4[M+1]+。MS(ESI, pos.ion) m/z: 421.4[M+1] + .
步骤2:化合物4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯胺的合成Step 2: Synthesis of compound 4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)aniline
4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯胺(300mg,0.71mmol)、三氟乙酸(6mL)和乙二胺(1mL)按照实施例2步骤2的合成方法制备得到146mg黄色固体,产率:71.2%。4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3 -b]pyrazin-7-yl)aniline (300mg, 0.71mmol), trifluoroacetic acid (6mL) and ethylenediamine (1mL) were prepared according to the synthetic method of Example 2, step 2 to obtain 146mg of yellow solid, yield: 71.2 %.
MS(ESI,pos.ion)m/z:291.4[M+1]+。MS(ESI, pos.ion) m/z: 291.4[M+1] + .
步骤3:化合物1-(氰甲基)-3-(4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)脲的合成Step 3: Compound 1-(cyanomethyl)-3-(4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine- Synthesis of 7-yl)phenyl)urea
将甲苯(5mL)加入到4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯胺(36mg,0.12mmol)、三光气(33mg,0.11mmol)中,再加入两滴三乙胺,110℃回流反应5h,反应液冷却至室温,加入氨基乙腈盐酸盐(28mg,0.30mmol)、三乙胺(100mg,0.10mmol),室温搅拌12h,反应加入硅胶,直接旋干拌样,进行柱分离(二氯甲烷/甲醇(v/v)=15/1)得到35mg白色固体,产率:75.8%。Toluene (5 mL) was added to 4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)aniline (36 mg, 0.12mmol) and triphosgene (33mg, 0.11mmol), then two drops of triethylamine were added, and the reaction was refluxed at 110°C for 5h. The reaction solution was cooled to room temperature, and aminoacetonitrile hydrochloride (28mg, 0.30mmol), triethylamine were added. (100mg, 0.10mmol), stirred at room temperature for 12h, added silica gel to the reaction, directly spin-dried and mixed the sample, and carried out column separation (dichloromethane/methanol (v/v)=15/1) to obtain 35mg of white solid, yield: 75.8% .
MS(ESI,neg.ion)m/z:371.2[M-1]-;MS(ESI,neg.ion)m/z:371.2[M-1] - ;
1H NMR(600MHz,DMSO-d6):δ(ppm)9.32(s,1H),9.14(s,1H),8.78(s,1H),8.67(s,1H),8.54(s,1H),8.28(d,J=8.6Hz,2H),8.24(s,1H),7.57(d,J=8.6Hz,2H),6.83(t,J=5.7Hz,1H),4.17(d,J=5.6Hz,2H),3.96(s,3H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 9.32(s,1H), 9.14(s,1H), 8.78(s,1H), 8.67(s,1H), 8.54(s,1H) ,8.28(d,J=8.6Hz,2H),8.24(s,1H),7.57(d,J=8.6Hz,2H),6.83(t,J=5.7Hz,1H),4.17(d,J= 5.6Hz, 2H), 3.96(s, 3H);
13C NMR(150MHz,DMSO-d6):δ(ppm)155.2,150.2,144.5,139.7,139.6,138.6,137.7,134.1,130.4,127.6,125.7,125.5,120.6,119.0,118.7,118.0,117.0,39.3,29.6。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 155.2, 150.2, 144.5, 139.7, 139.6, 138.6, 137.7, 134.1, 130.4, 127.6, 125.7, 125.5, 120.6, 119.0, 118.7, 118.0, 117.0 39.3, 29.6.
采用相应的原料,按照实施例16的合成方法制备如下所示的化合物:Using the corresponding raw materials, the following compounds were prepared according to the synthetic method of Example 16:
实施例20Example 20
3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexyl)amino)propionitrile
步骤1:化合物2-(1-甲基-1H-吡唑-4-基)-7-(1,4-二氧螺[4.5]葵烷-7-烯-8-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪的合成Step 1: Compound 2-(1-Methyl-1H-pyrazol-4-yl)-7-(1,4-dioxospiro[4.5]decane-7-en-8-yl)-5-( Synthesis of (2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine
7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(30mg,0.07mmol),4,4,5,5-四甲基-2-(1,4-二氧螺[4.5]葵烷-7-烯-8-基)-1,3,2-二氧硼戊烷(40mg,0.15mmol),碳酸钾(15mg,0.10mmol),Pd(dppf)Cl2(6mg,0.01mmol)和水(1mL)根据实施例1步骤1的合成方法制备得到21mg淡黄色油状物,产率:61.14%。7-Bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2, 3-b]pyrazine (30 mg, 0.07 mmol), 4,4,5,5-tetramethyl-2-(1,4-dioxospiro[4.5]decane-7-en-8-yl)- 1,3,2-Dioxaboropentane (40 mg, 0.15 mmol), potassium carbonate (15 mg, 0.10 mmol), Pd(dppf)Cl2 ( 6 mg, 0.01 mmol) and water (1 mL) according to Example 1, Step 1 The synthesis method of 21 mg of pale yellow oil was obtained, yield: 61.14%.
MS(ESI,pos.ion)m/z:468.3[M+1]+。MS(ESI, pos.ion) m/z: 468.3[M+1] + .
步骤2:化合物2-(1-甲基-1H-吡唑-4-基)-7-(1,4-二氧螺[4.5]葵烷-8-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪的合成Step 2: Compound 2-(1-methyl-1H-pyrazol-4-yl)-7-(1,4-dioxospiro[4.5]decan-8-yl)-5-((2-( Synthesis of Trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine
向2-(1-甲基-1H-吡唑-4-基)-7-(1,4-二氧螺[4.5]葵烷-7-烯-8-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(320mg,0.68mmol)和甲醇(8mL)溶液中加入Pd/C(10%,156mg,0.14mmol),在氢气氛围下室温搅拌3小时,过滤,浓缩,得到330mg黄色油状物粗产品,直接进行下一步反应,产率:99%。To 2-(1-methyl-1H-pyrazol-4-yl)-7-(1,4-dioxospiro[4.5]decane-7-en-8-yl)-5-((2- To a solution of (trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (320 mg, 0.68 mmol) and methanol (8 mL) was added Pd/C (10%, 156 mg) , 0.14 mmol), stirred at room temperature for 3 hours under a hydrogen atmosphere, filtered and concentrated to obtain 330 mg of crude yellow oily product, which was directly carried out to the next step, yield: 99%.
MS(ESI,pos.ion)m/z:470.4[M+1]+。MS(ESI, pos.ion) m/z: 470.4[M+1] + .
步骤3:化合物4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己酮的合成Step 3: Compound 4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)cyclohexanone
向2-(1-甲基-1H-吡唑-4-基)-7-(1,4-二氧螺[4.5]葵烷-8-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(330mg,0.70mmol)的丙酮(10mL)溶液中,加入对甲苯磺酸(363mg,2.10mmol),室温搅拌8小时,直接浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到180mg黄色固体,产率:60.18%。To 2-(1-methyl-1H-pyrazol-4-yl)-7-(1,4-dioxospiro[4.5]decan-8-yl)-5-((2-(trimethyl To a solution of silyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (330 mg, 0.70 mmol) in acetone (10 mL) was added p-toluenesulfonic acid (363 mg, 2.10 mmol), The mixture was stirred at room temperature for 8 hours, and was directly concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 180 mg of a yellow solid, yield: 60.18%.
MS(ESI,pos.ion)m/z:426.4[M+1]+。MS(ESI, pos.ion) m/z: 426.4[M+1] + .
步骤4:化合物3-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈的合成Step 4: Compound 3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- Synthesis of 5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexyl)amino)propionitrile
室温下,向4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己酮(15mg,0.03mmol),3-氨基丙腈(30μL,0.41mmol)的四氢呋喃(4mL)溶液中加入三乙酰氧基硼氢化钠(12mg,0.05mmol),室温搅拌6小时,加水(20mL)淬灭,二氯甲烷萃取(20mLx3),无水硫酸钠干燥,浓缩柱层析(二氯甲烷/甲醇(v/v)=10/1)得到12mg黄色油状物,产率:70.98%。To 4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo at room temperature To a solution of [2,3-b]pyrazin-7-yl)cyclohexanone (15 mg, 0.03 mmol), 3-aminopropionitrile (30 μL, 0.41 mmol) in tetrahydrofuran (4 mL) was added sodium triacetoxyborohydride (12 mg, 0.05 mmol), stirred at room temperature for 6 hours, quenched by adding water (20 mL), extracted with dichloromethane (20 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (dichloromethane/methanol (v/v)=10 /1) 12 mg of yellow oil was obtained, yield: 70.98%.
MS(ESI,pos.ion)m/z:480.0[M+1]+。MS(ESI, pos.ion) m/z: 480.0[M+1] + .
步骤5:化合物3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈的合成Step 5: Compound 3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexyl) Synthesis of Amino)Propionitrile
3-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈(130mg,0.27mmol)、三氟乙酸(2mL)和乙二胺(1mL)根据实施例2步骤2的合成方法制备得到16mg淡黄色固体,产率:16.90%。3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo [2,3-b]pyrazin-7-yl)cyclohexyl)amino)propionitrile (130 mg, 0.27 mmol), trifluoroacetic acid (2 mL) and ethylenediamine (1 mL) according to the synthesis method of Example 2, Step 2 16 mg of pale yellow solid were prepared, yield: 16.90%.
MS(ESI,pos.ion)m/z:350.30[M+1]+;MS(ESI, pos.ion) m/z: 350.30[M+1] + ;
1H NMR(600MHz,DMSO-d6)δ(ppm)11.72(s,1H),9.27(s,1H),8.55(s,1H),8.29(s,1H),8.03(s,1H),7.58(d,J=103.8Hz,1H),3.91(s,3H),2.94(m,5H),2.19(d,J=7.4Hz,1H),1.82(m,6H),1.26(d,J=28.4Hz,2H)。 1 H NMR (600MHz, DMSO-d 6 )δ(ppm) 11.72(s,1H), 9.27(s,1H), 8.55(s,1H), 8.29(s,1H), 8.03(s,1H), 7.58(d, J=103.8Hz, 1H), 3.91(s, 3H), 2.94(m, 5H), 2.19(d, J=7.4Hz, 1H), 1.82(m, 6H), 1.26(d, J =28.4Hz, 2H).
实施例21Example 21
3-(甲基(4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈3-(methyl(4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino) Propionitrile
步骤1:化合物3-(甲基(4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 1: Compound 3-(methyl(4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl) )-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile synthesis
0℃下,将氢化钠(60%,55mg,1.38mmol)分批加入到3-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(320mg,0.54mmol)的N,N-二甲基甲酰胺 (15mL)混合液中。室温下搅拌1.5h后,再在0℃下缓慢滴加碘甲烷(200mg,2.00mmol),滴加完毕后,室温下搅拌反应15h。加水(50mL)淬灭反应,乙酸乙酯(50mL x 3)萃取,有机层用饱和食盐水(50mL)洗涤,无水硫酸钠干燥,浓缩除去溶剂,残留物进行柱层析分离(石油醚/乙酸乙酯(v/v)=1/1)得到180mg淡黄色固体,产率:54.6%。At 0°C, sodium hydride (60%, 55 mg, 1.38 mmol) was added portionwise to 3-((4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2 -(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile (320 mg, 0.54 mmol) in N, N-dimethylformamide (15 mL) mixture. After stirring at room temperature for 1.5 h, methyl iodide (200 mg, 2.00 mmol) was slowly added dropwise at 0 °C. After the dropwise addition, the reaction was stirred at room temperature for 15 h. The reaction was quenched by adding water (50 mL), extracted with ethyl acetate (50 mL x 3), the organic layer was washed with saturated brine (50 mL), dried over anhydrous sodium sulfate, concentrated to remove the solvent, and the residue was separated by column chromatography (petroleum ether/ Ethyl acetate (v/v)=1/1) gave 180 mg of pale yellow solid, yield: 54.6%.
MS(ESI,pos.ion)m/z:487.9[M+1]+。MS(ESI, pos.ion) m/z: 487.9[M+1] + .
步骤2:化合物3-(甲基(4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 2: Compound 3-(methyl(4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)benzene Synthesis of base)amino)propionitrile
3-(甲基(4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(180mg,0.37mmol)、三氟乙酸(2mL)和乙二胺(1mL)按照实施例2步骤2的合成方法制备得到40mg橘黄色固体,产率:30.3%。3-(methyl(4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H- Pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile (180 mg, 0.37 mmol), trifluoroacetic acid (2 mL) and ethylenediamine (1 mL) were prepared as in Example 2, Step 2 The synthetic method prepared 40 mg of orange-yellow solid, yield: 30.3%.
MS(ESI,pos.ion)m/z:376.1[M+1]+。MS(ESI, pos.ion) m/z: 376.1[M+1] + .
1H NMR(400MHz,DMSO-d6):δ(ppm)11.97(d,J=2.3Hz,1H),8.61(s,1H),8.36(s,1H),8.16(m,2H),8.13(s,1H),8.11(s,1H),6.87(d,J=8.9Hz,2H),3.94(s,3H),3.71(t,J=6.7Hz,2H),2.99(s,3H),2.75(t,J=6.7Hz,2H); 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 11.97 (d, J=2.3 Hz, 1H), 8.61 (s, 1H), 8.36 (s, 1H), 8.16 (m, 2H), 8.13 (s,1H),8.11(s,1H),6.87(d,J=8.9Hz,2H),3.94(s,3H),3.71(t,J=6.7Hz,2H),2.99(s,3H) ,2.75(t,J=6.7Hz,2H);
13C NMR(100MHz,DMSO-d6):δ(ppm)146.80,141.5,140.9,137.1,136.2,134.2,129.3,127.2,126.4,123.1,121.9,120.1,113.7,113.1,48.4,38.4,32.0,15.2。 13 C NMR (100MHz, DMSO-d 6 ): δ(ppm) 146.80, 141.5, 140.9, 137.1, 136.2, 134.2, 129.3, 127.2, 126.4, 123.1, 121.9, 120.1, 113.7, 113.1, 48.4, 38.4, 32.0, 15.2.
实施例22Example 22
4-(4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)哌啶-1-基)丁腈4-(4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)piperidin-1-yl)butane Nitrile
步骤1:化合物4-(2-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-5,6-二氢吡啶-1(2H)-甲酸叔丁酯的合成Step 1: Compound 4-(2-Bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl) Synthesis of -5,6-dihydropyridine-1(2H)-carboxylate tert-butyl ester
2-溴-7-碘-5-((2-(三甲硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(600mg,1.32mmol)、4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-5,6-二氢吡啶-1(2H)-甲酸叔丁酯(425mg,1.37mmol)、碳酸钠(356mg,3.35mmol)、Pd(PPh3)2Cl2(100mg,0.14mmol)和水(6mL)根据实施例1步骤1的合成方法制备得到540mg淡黄色固体,产率:80.23%。2-Bromo-7-iodo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (600 mg, 1.32 mmol), 4-( 4,4,5,5-Tetramethyl-1,3,2-dioxaboropentan-2-yl)-5,6-dihydropyridine-1(2H)-carboxylic acid tert-butyl ester (425 mg, 1.37 mmol), sodium carbonate (356 mg, 3.35 mmol), Pd(PPh 3 ) 2 Cl 2 (100 mg, 0.14 mmol) and water (6 mL) were prepared according to the synthetic method of Example 1, step 1 to obtain 540 mg of pale yellow solid, yield: 80.23%.
MS(ESI,pos.ion)m/z:509.2,511.2[M+1]+。MS(ESI, pos.ion) m/z: 509.2,511.2[M+1] + .
步骤2:化合物4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-5,6-二氢吡啶-1(2H)-甲酸叔丁酯的合成Step 2: Compound 4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)-5,6-dihydropyridine-1(2H)-carboxylic acid tert-butyl ester
4-(2-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-5,6-二氢吡啶-1(2H)-甲酸叔丁酯(325mg,0.63mmo),碳酸钾(140mg,1.01mmol),1-甲基吡唑硼酸酯(172mg,0.82mmol)和Pd(dppf)Cl2(50mg,0.06mmol)根据实施例2步骤1的合成方法制备得到270mg黄色固体,产率:82.88%。4-(2-Bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)-5,6 - tert-butyl dihydropyridine-1(2H)-carboxylate (325mg, 0.63mmol), potassium carbonate (140mg, 1.01mmol), 1-methylpyrazole boronate (172mg, 0.82mmol) and Pd(dppf) Cl 2 (50 mg, 0.06 mmol) was prepared according to the synthetic method of Example 2, step 1 to obtain 270 mg of a yellow solid, yield: 82.88%.
MS(ESI,pos.ion)m/z:511.4[M+1]+。MS(ESI, pos.ion) m/z: 511.4[M+1] + .
步骤3:化合物4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)哌啶-1-甲酸叔丁酯的合成Step 3: Compound 4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)piperidine-1-carboxylate tert-butyl ester
向4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-5,6-二氢吡 啶-1(2H)-甲酸叔丁酯(270mg,0.52mmol)的甲醇(8mL)溶液中加入Pd/C(10%,30mg,0.028mmol),在氢气氛围下,室温搅拌过夜,过滤,浓缩后,柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到250mg黄色油状物,产率:92.23%。To 4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2, 3-b]pyrazin-7-yl)-5,6-dihydropyridine-1(2H)-carboxylic acid tert-butyl ester (270 mg, 0.52 mmol) in methanol (8 mL) was added Pd/C (10%, 30 mg, 0.028 mmol), stirred overnight at room temperature under a hydrogen atmosphere, filtered, concentrated, and subjected to column chromatography (petroleum ether/ethyl acetate (v/v)=0/1) to obtain 250 mg of yellow oil, yield: 92.23 %.
MS(ESI,pos.ion)m/z:513.4[M+1]+。MS(ESI, pos.ion) m/z: 513.4[M+1] + .
步骤4:化合物2-(1-甲基-1H-吡唑-4-基)-7-(哌啶-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪的合成Step 4: Compound 2-(1-Methyl-1H-pyrazol-4-yl)-7-(piperidin-4-yl)-5-((2-(trimethylsilyl)ethoxy) Synthesis of methyl)-5H-pyrrolo[2,3-b]pyrazine
冰浴下,向4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)哌啶-1-甲酸叔丁酯(250mg,0.48mmol)的甲醇(8mL)溶液中,加入乙酰氯(70μL,0.97mmol),室温搅拌过夜,反应完毕,直接浓缩,柱层析(二氯甲烷/甲醇(v/v)=8/1)得到260mg黄色固体,产率:99%。Under ice bath, add 4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrole Acetyl chloride (70 μL, 0.97 mmol) was added to a solution of tert-butyl iso[2,3-b]pyrazin-7-yl)piperidine-1-carboxylate (250 mg, 0.48 mmol) in methanol (8 mL), and stirred at room temperature After overnight, the reaction was completed, directly concentrated, and column chromatography (dichloromethane/methanol (v/v)=8/1) obtained 260 mg of yellow solid, yield: 99%.
MS(ESI,pos.ion)m/z:413.4[M+1]+。MS(ESI, pos.ion) m/z: 413.4[M+1] + .
步骤5:化合物4-(4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)哌啶-1-基)丁腈的合成Step 5: Compound 4-(4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrole Synthesis of [2,3-b]pyrazin-7-yl)piperidin-1-yl)butyronitrile
向2-(1-甲基-1H-吡唑-4-基)-7-(哌啶-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(130mg,0.31mmol)的乙腈(8mL)溶液中依次加入碳酸钾(110mg,0.79mmol),碘化钾(10mg,0.06mmol)和4-氯丁腈(0.05mL,0.5mmol),50℃加热反应24小时,加水(20mL)稀释,二氯甲烷萃取(20mL x 3),无水硫酸钠干燥,浓缩柱层析(二氯甲烷/甲醇(v/v)=8/1)得到120mg黄色油状物,产率:79.40%。To 2-(1-methyl-1H-pyrazol-4-yl)-7-(piperidin-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl) To a solution of -5H-pyrrolo[2,3-b]pyrazine (130 mg, 0.31 mmol) in acetonitrile (8 mL) were sequentially added potassium carbonate (110 mg, 0.79 mmol), potassium iodide (10 mg, 0.06 mmol) and 4-chlorobutane Nitrile (0.05mL, 0.5mmol), heated at 50°C for 24 hours, diluted with water (20mL), extracted with dichloromethane (20mL x 3), dried over anhydrous sodium sulfate, concentrated by column chromatography (dichloromethane/methanol (v /v)=8/1) to give 120 mg of yellow oil, yield: 79.40%.
MS(ESI,pos.ion)m/z:480.0[M+1]+。MS(ESI, pos.ion) m/z: 480.0[M+1] + .
步骤6:化合物4-(4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)哌啶-1-基)丁腈的合成Step 6: Compound 4-(4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)piperidine-1 -Base) Synthesis of Butyronitrile
4-(4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)哌啶-1-基)丁腈(120mg,0.25mmol)和三氟乙酸(2mL)根据实施例2步骤2的合成方法制备得到36mg淡黄色固体,产率:41.19%。4-(4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2, 3-b]pyrazin-7-yl)piperidin-1-yl)butyronitrile (120 mg, 0.25 mmol) and trifluoroacetic acid (2 mL) were prepared according to the synthetic method of Example 2, step 2 to obtain 36 mg of a pale yellow solid, yielding Rate: 41.19%.
MS(ESI,pos.ion)m/z:351.00[M+1]+;MS(ESI, pos.ion) m/z: 351.00[M+1] + ;
1H NMR(400MHz,DMSO-d6)δ(ppm)11.69(s,1H),8.55(s,1H),8.32(s,1H),8.04(s,1H),7.58(s,1H),3.91(s,3H),3.01(dd,J=126.5,49.0Hz,5H),2.57(s,2H),1.99(m,8H)。 1 H NMR (400MHz, DMSO-d 6 )δ(ppm) 11.69(s,1H), 8.55(s,1H), 8.32(s,1H), 8.04(s,1H), 7.58(s,1H), 3.91(s, 3H), 3.01(dd, J=126.5, 49.0Hz, 5H), 2.57(s, 2H), 1.99(m, 8H).
实施例23Example 23
3-((3-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈3-((3-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexyl)amino)propionitrile
步骤1:化合物3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己烯酮的合成Step 1: Compound 3-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)cyclohexenone
氮气保护下,将1,4-二氧六环(16mL)/水(4mL)加入到7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(500mg,1.22mmol)、3-(4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)环己烯酮(650mg,2.34mmol)、碳酸钾(340mg,0.2.46mmol)和Pd(dppf)Cl2(45mg,0.06mmol)混合物中,110℃回流反应8h,反应液冷却至室温,硅藻土过滤,滤液减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=0/1)得到450mg黄色固体,产率:69.4%。Under nitrogen, 1,4-dioxane (16 mL)/water (4 mL) was added to 7-bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2 -(Trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (500 mg, 1.22 mmol), 3-(4-(4,4,5,5- Tetramethyl-1,3,2-dioxaboropentan-2-yl)cyclohexenone (650 mg, 2.34 mmol), potassium carbonate (340 mg, 0.2.46 mmol) and Pd(dppf)Cl 2 (45 mg, 0.06mmol) mixture, refluxed at 110°C for 8h, the reaction solution was cooled to room temperature, filtered through celite, the filtrate was concentrated under reduced pressure, and subjected to column separation (petroleum ether/ethyl acetate (v/v)=0/1) to obtain 450mg Yellow solid, yield: 69.4%.
MS(ESI,pos.ion)m/z:424.0[M+1]+。MS(ESI, pos.ion) m/z: 424.0[M+1] + .
步骤2:化合物3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7- 基)环己醇的合成Step 2: Compound 3-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)cyclohexanol
将Pd/C(10%,200mg)加入到3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己烯酮的甲醇溶液中,室温下反应24h,反应液硅藻土过滤,用二氯甲烷洗涤滤饼,滤液减压浓缩,进行柱分离(二氯甲烷/甲醇(v/v)=15/1)得到350mg黄色油状物,产率:77.0%。Pd/C (10%, 200 mg) was added to 3-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy) Methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexenone in methanol solution, react at room temperature for 24h, the reaction solution is filtered through celite, and the filter cake is washed with dichloromethane , the filtrate was concentrated under reduced pressure and subjected to column separation (dichloromethane/methanol (v/v)=15/1) to obtain 350 mg of yellow oil, yield: 77.0%.
MS(ESI,pos.ion)m/z:428.0[M+1]+。MS(ESI, pos.ion) m/z: 428.0[M+1] + .
步骤3:化合物3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己酮的合成Step 3: Compound 3-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)cyclohexanone
将Dess-Martin试剂(360mg,0.85mmol)加入到3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己醇(300mg,0.70mmol)的二氯甲烷(15mL)溶液中,室温下反应3天,用饱和的硫代硫酸钠溶液(30mL)淬灭反应,水相用二氯甲烷(20mL x 3)萃取,有机层无水硫酸钠干燥,进行柱分离(石油醚/乙酸乙酯(v/v)=1/3)得到100mg黄色油状物,产率:33.5%。Dess-Martin reagent (360 mg, 0.85 mmol) was added to 3-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy ) methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexanol (300 mg, 0.70 mmol) in dichloromethane (15 mL) solution, reacted at room temperature for 3 days, saturated with of sodium thiosulfate solution (30 mL) to quench the reaction, the aqueous phase was extracted with dichloromethane (20 mL x 3), the organic layer was dried over anhydrous sodium sulfate, and subjected to column separation (petroleum ether/ethyl acetate (v/v) = 1/3) 100 mg of yellow oil was obtained, yield: 33.5%.
MS(ESI,pos.ion)m/z:426.0[M+1]+。MS(ESI, pos.ion) m/z: 426.0[M+1] + .
步骤4:化合物3-((3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈的合成Step 4: Compound 3-((3-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- Synthesis of 5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexyl)amino)propionitrile
将三乙酰氧基硼氢化钠(70mg,0.33mmol)加入到3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己酮(80mg,0.19mmol)和3-氨基丙腈(20mg,0.28mmol)的四氢呋喃溶液(5mL)中,室温下反应2h。加水(30mL)淬灭反应,用二氯甲烷(30mL x 3)萃取,有机层无水硫酸钠干燥,进行柱分离(二氯甲烷/甲醇(v/v)=30/1)得到50mg黄色油状物,产率:55.4%.Sodium triacetoxyborohydride (70 mg, 0.33 mmol) was added to 3-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl) Ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexanone (80 mg, 0.19 mmol) and 3-aminopropionitrile (20 mg, 0.28 mmol) in tetrahydrofuran (5mL), the reaction was carried out at room temperature for 2h. The reaction was quenched by adding water (30 mL), extracted with dichloromethane (30 mL x 3), the organic layer was dried over anhydrous sodium sulfate, and subjected to column separation (dichloromethane/methanol (v/v)=30/1) to obtain 50 mg of yellow oil product, yield: 55.4%.
MS(ESI,pos.ion)m/z:480.4[M+1]+。MS(ESI, pos.ion) m/z: 480.4[M+1] + .
步骤5:化合物3-((3-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己烷)氨基)丙腈的合成Step 5: Compound 3-((3-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)cyclohexane ) Amino) Propionitrile Synthesis
3-((3-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)环己基)氨基)丙腈(110mg,0.20mmol)、三氟乙酸(2mL)和乙二胺(1mL)按照实施例2步骤2的合成方法制备得到25mg黄色固体,产率:76.3%。3-((3-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo [2,3-b]pyrazin-7-yl)cyclohexyl)amino)propionitrile (110 mg, 0.20 mmol), trifluoroacetic acid (2 mL) and ethylenediamine (1 mL) were synthesized according to Example 2, Step 2 25 mg of yellow solid were prepared, yield: 76.3%.
MS(ESI,pos.ion)m/z:350.0[M+1]+。MS(ESI, pos.ion) m/z: 350.0[M+1] + .
1H NMR(600MHz,DMSO-d6):δ(ppm)11.58(d,J=1.8Hz,1H),8.54(s,1H),8.26(s,1H),8.03(s,1H),7.51(d,J=2.3Hz,1H),3.91(s,3H),2.94(s,1H),2.86(m,2H),2.65(td,J=6.7,1.7Hz,2H),2.04(dd,J=31.6,10.8Hz,2H),1.80(m,2H),1.64(m,2H),1.51(m,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 11.58(d, J=1.8Hz, 1H), 8.54(s, 1H), 8.26(s, 1H), 8.03(s, 1H), 7.51 (d, J=2.3Hz, 1H), 3.91(s, 3H), 2.94(s, 1H), 2.86(m, 2H), 2.65(td, J=6.7, 1.7Hz, 2H), 2.04(dd, J=31.6, 10.8Hz, 2H), 1.80(m, 2H), 1.64(m, 2H), 1.51(m, 2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)140.7,140.6,137.3,136.9,134.0,129.0,126.7,121.9,120.7,119.6,51.8,43.0,39.1,36.9,32.6,30.9,28.1,20.7,18.7。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 140.7, 140.6, 137.3, 136.9, 134.0, 129.0, 126.7, 121.9, 120.7, 119.6, 51.8, 43.0, 39.1, 36.9, 32.6, 30.9, 28.1, 20.7, 18.7.
实施例24Example 24
1-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)磺酰基)氮杂环丁烷-3-甲腈1-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)sulfonyl)nitrogen tetracyclobutane-3-carbonitrile
步骤1:化合物1-((4-溴苯基)磺酰基)氮杂环丁烷-3-甲腈的合成Step 1: Synthesis of compound 1-((4-bromophenyl)sulfonyl)azetidine-3-carbonitrile
氮气保护下,将二氯甲烷(12mL)加入到4-溴苯磺酰氯(252mg,0.99mmol)、3-乙腈环丁胺盐酸盐(90mg,0.76mmol)、三乙胺(230mg,2.28mmol)中,室温反应12h,反应液用乙酸乙酯萃取(50mLx3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=3/1)得到179mg白色固体,产率:78.3%。Under nitrogen protection, dichloromethane (12 mL) was added to 4-bromobenzenesulfonyl chloride (252 mg, 0.99 mmol), 3-acetonitrile cyclobutylamine hydrochloride (90 mg, 0.76 mmol), triethylamine (230 mg, 2.28 mmol) ), reacted at room temperature for 12 h, the reaction solution was extracted with ethyl acetate (50 mL×3), the organic phase was spin-dried, and separated by column chromatography (petroleum ether/ethyl acetate (v/v)=3/1) to obtain 179 mg of white solid, the product Rate: 78.3%.
1H NMR(400MHz,DMSO-d6):δ(ppm)7.94(d,J=8.5Hz,2H),7.78(d,J=8.5Hz,2H),4.00(t,J=8.7Hz,2H),3.87(dd,J1=8.5Hz,J2=5.9Hz,2H),3.69–3.58(m,1H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 7.94 (d, J=8.5 Hz, 2H), 7.78 (d, J=8.5 Hz, 2H), 4.00 (t, J=8.7 Hz, 2H) ), 3.87 (dd, J 1 =8.5 Hz, J 2 =5.9 Hz, 2H), 3.69-3.58 (m, 1H).
步骤2:化合物1-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)磺酰基)氮杂环丁烷-3-甲腈的合成Step 2: Compound 1-((4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- Synthesis of 5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)sulfonyl)azetidine-3-carbonitrile
氮气保护下,将1,4-二氧六环(12mL)加入到1-((4-溴苯基)磺酰基)氮杂环丁烷-3-甲腈(148mg,0.49mmol)、联硼酸频哪醇酯(138mg,0.54mmol)、醋酸钾(150mg,1.53mmol)、Pd(dppf)Cl2(23mg,0.03mmol)混合物中,110℃回流反应11h,反应液冷却至室温,氮气保护下,加入水(2.5mL)、碳酸钾(130mg,0.93mmol)、7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(120mg,0.29mmol)、Pd(dppf)Cl2(23mg,0.03mmol),110℃回流反应10h,反应液冷却至室温,硅藻土过滤,滤液减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=1/2)得到150mg黄色固体,产率:55.5%。Under nitrogen, 1,4-dioxane (12 mL) was added to 1-((4-bromophenyl)sulfonyl)azetidine-3-carbonitrile (148 mg, 0.49 mmol), biboronic acid In a mixture of pinacol ester (138 mg, 0.54 mmol), potassium acetate (150 mg, 1.53 mmol) and Pd(dppf)Cl 2 (23 mg, 0.03 mmol), the reaction was refluxed at 110° C. for 11 h, and the reaction solution was cooled to room temperature under nitrogen protection. , added water (2.5 mL), potassium carbonate (130 mg, 0.93 mmol), 7-bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilane) Base)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (120mg, 0.29mmol), Pd(dppf)Cl 2 (23mg, 0.03mmol), 110 ℃ reflux reaction for 10h, the reaction The liquid was cooled to room temperature, filtered through celite, the filtrate was concentrated under reduced pressure, and subjected to column separation (petroleum ether/ethyl acetate (v/v)=1/2) to obtain 150 mg of yellow solid, yield: 55.5%.
MS(ESI,pos.ion)m/z:549.8[M+1]+。MS(ESI, pos.ion) m/z: 549.8[M+1] + .
步骤3:化合物1-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)磺酰基)氮杂环丁烷-3-甲腈的合成Step 3: Compound 1-((4-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl) Synthesis of sulfonyl)azetidine-3-carbonitrile
1-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)磺酰基)氮杂环丁烷-3-甲腈(150mg,0.27mmol)、三氟乙酸(6mL)和乙二胺(1mL)根据实施例2步骤2的合成方法制备得到91mg浅黄色固体,产率:79.5%。1-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo [2,3-b]pyrazin-7-yl)phenyl)sulfonyl)azetidine-3-carbonitrile (150 mg, 0.27 mmol), trifluoroacetic acid (6 mL) and ethylenediamine (1 mL) According to the synthesis method of Example 2, step 2, 91 mg of light yellow solid was obtained, yield: 79.5%.
MS(ESI,pos.ion)m/z:419.9[M+1]+;MS(ESI, pos.ion) m/z: 419.9[M+1] + ;
1H NMR(600MHz,DMSO-d6+D2O):δ(ppm)8.72(d,J=8.4Hz,2H),8.72(s,1H),8.65(s,1H),8.49(s,1H),8.21(s,1H),7.90(d,J=8.4Hz,2H),4.02(t,J=8.7Hz,2H),3.95(s,3H),3.88(dd,J1=8.3Hz,J2=6.2Hz,2H),3.65(m,1H); 1 H NMR (600MHz, DMSO-d 6 +D 2 O): δ(ppm) 8.72(d, J=8.4Hz, 2H), 8.72(s, 1H), 8.65(s, 1H), 8.49(s, 1H), 8.21(s, 1H), 7.90(d, J=8.4Hz, 2H), 4.02(t, J=8.7Hz, 2H), 3.95(s, 3H), 3.88(dd, J 1 =8.3Hz , J 2 =6.2Hz, 2H), 3.65(m, 1H);
13C NMR(150MHz,DMSO-d6):δ(ppm)142.5,141.2,140.2,137.4,136.3,135.1,130.6,129.8,129.3,129.2,126.3,121.5,120.3,111.3,53.8,40.5,39.3,17.2。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 142.5, 141.2, 140.2, 137.4, 136.3, 135.1, 130.6, 129.8, 129.3, 129.2, 126.3, 121.5, 120.3, 111.3, 53.8, 40.5, 39.3, 17.2.
实施例25Example 25
3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)磺酰基)丙腈3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)sulfonyl)propane Nitrile
步骤1:化合物3-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)硫代)丙腈的合成Step 1: Compound 3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- Synthesis of 5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)thio)propionitrile
碳酸钾(76mg,0.55mmol)、7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(90mg,0.22mmol)、3-((4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)硫代)丙腈(95mg,0.33mmol)和Pd(dppf)Cl2(7mg,0.01mmol)根据实施例1步骤1的合成方法制备得到45mg黄色油状物,产率:41.6%。Potassium carbonate (76 mg, 0.55 mmol), 7-bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl )-5H-pyrrolo[2,3-b]pyrazine (90 mg, 0.22 mmol), 3-((4-(4,4,5,5-tetramethyl-1,3,2-dioxoboron) Pentan-2-yl)phenyl)thio)propionitrile (95 mg, 0.33 mmol) and Pd(dppf)Cl 2 (7 mg, 0.01 mmol) were prepared according to the synthetic method of Example 1, step 1 to give 45 mg of yellow oil, Yield: 41.6%.
MS(ESI,pos.ion)m/z:491.3[M+1]+。MS(ESI, pos.ion) m/z: 491.3[M+1] + .
步骤2:化合物3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)硫代)丙腈的合成Step 2: Compound 3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl) Synthesis of Thio)propionitrile
3-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)硫代)丙腈(45mg,0.09mmol)、三氟乙酸(6mL)和乙二胺(1mL)根据实施例2步骤2的合成方法制备得到27mg浅白色固体,产率:81.7%。3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)phenyl)thio)propionitrile (45 mg, 0.09 mmol), trifluoroacetic acid (6 mL) and ethylenediamine (1 mL) according to Example 2, Step 2 The method prepared 27 mg of light white solid, yield: 81.7%.
MS(ESI,pos.ion)m/z:361.2[M+1]+。MS(ESI, pos.ion) m/z: 361.2[M+1] + .
步骤3:化合物3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)磺酰基)丙腈的合成Step 3: Compound 3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl) Synthesis of Sulfonyl)Propionitrile
室温下,3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)硫代)丙腈(36mg,0.10mmol)溶解于二氯甲烷(10mL)中,加入间氯过氧苯甲酸(60mg,0.30mmol),室温反应12h,反应液加入硅胶,室温旋干拌样,柱层析分离(二氯甲烷/甲醇(v/v)=20/1)得到43mg浅黄色固体,然后用制备薄层纯化得到6mg土灰色固体,产率:15.3%。3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)sulfur at room temperature Substance) propionitrile (36mg, 0.10mmol) was dissolved in dichloromethane (10mL), m-chloroperoxybenzoic acid (60mg, 0.30mmol) was added, and the reaction was carried out at room temperature for 12h. Chromatography (dichloromethane/methanol (v/v) = 20/1) gave 43 mg of a pale yellow solid, which was then purified by preparative thin layer to give 6 mg of an earthy grey solid, yield: 15.3%.
MS(ESI,pos.ion)m/z:393.2[M+1]+;MS(ESI, pos.ion) m/z: 393.2[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)8.71(s,1H),8.66(d,J=8.4Hz,2H),8.64(s,1H),8.48(s,1H),8.19(s,1H),7.97(d,J=8.4Hz,2H),3.95(s,3H),3.74(t,J=6.8Hz,2H),2.89(t,J=6.8Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 8.71(s, 1H), 8.66(d, J=8.4Hz, 2H), 8.64(s, 1H), 8.48(s, 1H), 8.19 (s, 1H), 7.97 (d, J=8.4Hz, 2H), 3.95 (s, 3H), 3.74 (t, J=6.8Hz, 2H), 2.89 (t, J=6.8Hz, 2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)142.5,140.5,137.4,136.3,135.1,134.4,130.7,130.1,129.8,129.0,126.2,121.5,118.3,111.3,50.3,39.3,12.1。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 142.5, 140.5, 137.4, 136.3, 135.1, 134.4, 130.7, 130.1, 129.8, 129.0, 126.2, 121.5, 118.3, 111.3, 50.3, 39.3, 12.1.
采用相应的原料,按照实施例24的合成方法制备如下所示的化合物:Using the corresponding raw materials, the following compounds were prepared according to the synthetic method of Example 24:
实施例38Example 38
2-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)乙腈2-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)acetonitrile
步骤1:化合物4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯胺的合成Step 1: Compound 4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo Synthesis of [2,3-b]pyrazin-7-yl)aniline
7-溴-2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(200mg,0.49mmol)、4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯胺(150mg,0.68mmol)、碳酸钾(120mg,0.87mmol)和1,1'-双二苯基膦二茂铁二氯化钯(20mg,0.03mmol)根据实施例1步骤1的合成方法制备得到180mg黄色油状物,产率:72.3%。7-Bromo-2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2, 3-b]pyrazine (200mg, 0.49mmol), 4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)aniline (150mg, 0.68mmol) ), potassium carbonate (120mg, 0.87mmol) and 1,1'-bisdiphenylphosphinoferrocene palladium dichloride (20mg, 0.03mmol) were prepared according to the synthetic method of Example 1, step 1 to obtain 180mg of yellow oil, Yield: 72.3%.
MS(ESI,pos.ion)m/z:421.0[M+1]+。MS(ESI, pos.ion) m/z: 421.0[M+1] + .
步骤2:化合物2-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)乙腈的合成Step 2: Compound 2-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)- Synthesis of 5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)acetonitrile
将对2-溴乙腈(70mg,0.60mmol,0.04mL)加入到4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯胺(180mg,0.42mmol)和碳酸钾(90mg,0.65mmol)、碘化钾(110mg,0.66mmol)的乙腈(15mL)的溶液中,反应液在90℃回流反应过夜,冷却至室温,硅藻土过滤,减压浓缩,浓缩液进行柱分离(石油醚/乙酸乙酯(v/v)=0/1)得到140mg土黄色固体,产率:71.2%。p-2-Bromoacetonitrile (70 mg, 0.60 mmol, 0.04 mL) was added to 4-(2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl) )ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)aniline (180 mg, 0.42 mmol) and potassium carbonate (90 mg, 0.65 mmol), potassium iodide (110 mg, 0.66 mmol) ) in acetonitrile (15 mL), the reaction solution was refluxed at 90°C overnight, cooled to room temperature, filtered through celite, concentrated under reduced pressure, and the concentrated solution was subjected to column separation (petroleum ether/ethyl acetate (v/v)= 0/1) gave 140 mg of a khaki solid, yield: 71.2%.
MS(ESI,pos.ion)m/z:460.4[M+1]+。MS(ESI, pos.ion) m/z: 460.4[M+1] + .
步骤3:化合物2-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)乙腈的合成Step 3: Compound 2-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl) Synthesis of amino)acetonitrile
2-((4-(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)乙腈(130mg,0.28mmol)、三氟乙酸(2mL)和乙二胺(1mL)根据实施例2步骤2的合成方法制备得到50mg黄色固体,产率:53.7%。2-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo [2,3-b]pyrazin-7-yl)phenyl)amino)acetonitrile (130 mg, 0.28 mmol), trifluoroacetic acid (2 mL) and ethylenediamine (1 mL) were prepared according to the synthetic method of Example 2, Step 2 50 mg of yellow solid were obtained, yield: 53.7%.
MS(ESI,pos.ion)m/z:329.9[M+1]+;MS(ESI, pos.ion) m/z: 329.9[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)11.99(s,1H),8.62(s,1H),8.38(s,1H),8.17(d,J=2.6Hz,1H),8.13(s,1H),8.12(s,2H),6.83(d,J=8.6Hz,2H),6.24(t,J=6.9Hz,1H),4.30(d,J=6.8Hz,2H),3.94(s,3H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 11.99(s, 1H), 8.62(s, 1H), 8.38(s, 1H), 8.17(d, J=2.6Hz, 1H), 8.13 (s,1H),8.12(s,2H),6.83(d,J=8.6Hz,2H),6.24(t,J=6.9Hz,1H),4.30(d,J=6.8Hz,2H),3.94 (s, 3H);
13C NMR(150MHz,DMSO-d6):δ(ppm)145.1,141.5,140.8,137.1,136.1,134.3,129.4,127.1,126.6,124.4,121.8,119.2,113.7,113.6,40.4,39.2。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 145.1, 141.5, 140.8, 137.1, 136.1, 134.3, 129.4, 127.1, 126.6, 124.4, 121.8, 119.2, 113.7, 113.6, 40.4, 39.2.
实施例40Example 40
2-甲基-2-((2-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-N-(2,2,2-三氟乙基)丁酰胺2-Methyl-2-((2-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)pyrimidine- 4-yl)amino)-N-(2,2,2-trifluoroethyl)butanamide
步骤1:化合物(R)-2-甲基-2-((2-(2-(1-甲基-1H-吡唑-4-基)-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-N-(2,2,2-三氟乙基)丁酰胺的合成Step 1: Compound (R)-2-methyl-2-((2-(2-(1-methyl-1H-pyrazol-4-yl)-5-trityl-5H-pyrrolo[ Synthesis of 2,3-b]pyrazin-7-yl)pyrimidin-4-yl)amino)-N-(2,2,2-trifluoroethyl)butanamide
(R)-2-((2-(2-氯-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-2-甲基-N-(2,2,2-三氟乙基)丁酰胺(240mg,0.36mmol),碳酸钾(75mg,0.54mmol),1-甲基-4-吡唑硼酸酯(115mg,0.55mmol)和Pd(dppf)Cl2(30mg,0.04mmol)根据实施例2步骤1的合成方法制备得到120mg灰白色固体,产率:46.81%。(R)-2-((2-(2-Chloro-5-trityl-5H-pyrrolo[2,3-b]pyrazin-7-yl)pyrimidin-4-yl)amino)-2 - methyl-N-(2,2,2-trifluoroethyl)butanamide (240mg, 0.36mmol), potassium carbonate (75mg, 0.54mmol), 1-methyl-4-pyrazole boronate (115mg , 0.55 mmol) and Pd(dppf)Cl 2 (30 mg, 0.04 mmol) were prepared according to the synthetic method of Example 2, step 1 to obtain 120 mg of off-white solid, yield: 46.81%.
MS(ESI,pos.ion)m/z:716.45[M+1]+。MS(ESI, pos.ion) m/z: 716.45[M+1] + .
步骤2:化合物2-甲基-2-((2-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-N-(2,2,2-三氟乙基)丁酰胺的合成Step 2: Compound 2-methyl-2-((2-(2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine-7- Synthesis of pyrimidin-4-yl)amino)-N-(2,2,2-trifluoroethyl)butanamide
(R)-2-甲基-2-((2-(2-(1-甲基-1H-吡唑-4-基)-5-三苯甲基-5H-吡咯并[2,3-b]吡嗪-7-基)嘧啶-4-基)氨基)-N-(2,2,2-三氟乙基)丁酰胺(110mg,0.15mmol)、三氟乙酸(330μL,4.443mmol)和三乙基硅烷(670μL,4.19mmol)根据实施例2步骤2的合成方法制备得到20mg黄色固体,产率:27.49%。(R)-2-Methyl-2-((2-(2-(1-methyl-1H-pyrazol-4-yl)-5-trityl-5H-pyrrolo[2,3- b] Pyrazin-7-yl)pyrimidin-4-yl)amino)-N-(2,2,2-trifluoroethyl)butanamide (110 mg, 0.15 mmol), trifluoroacetic acid (330 μL, 4.443 mmol) and triethylsilane (670 μL, 4.19 mmol) according to the synthesis method of Example 2, step 2, to obtain 20 mg of a yellow solid, yield: 27.49%.
MS(ESI,pos.ion)m/z:474.30[M+1]+;MS(ESI, pos.ion) m/z: 474.30[M+1] + ;
1H NMR(600MHz,CD3OD)δ(ppm)8.70(s,1H),8.49(s,1H),8.45(s,1H),8.31(s,1H),8.26(m,1H),6.62(s,1H),4.02(s,3H),3.85(m,2H),2.24(dd,J=21.7,6.4Hz,1H),2.03(dd,J=19.9,10.2Hz,1H),1.66(s,3H),0.98(t,J=9.4Hz,3H); 1 H NMR (600MHz, CD 3 OD) δ(ppm) 8.70(s, 1H), 8.49(s, 1H), 8.45(s, 1H), 8.31(s, 1H), 8.26(m, 1H), 6.62 (s,1H),4.02(s,3H),3.85(m,2H),2.24(dd,J=21.7,6.4Hz,1H),2.03(dd,J=19.9,10.2Hz,1H),1.66( s, 3H), 0.98(t, J=9.4Hz, 3H);
13C NMR(150MHz,CD3OD)δ(ppm)175.8,161.2,143.2,140.8,137.4,135.8,135.3,129.9,129.4,128.4,125.5,125.3,123.5,121.2,103.8,60.8,48.1,40.1,37.7,20.5,6.7。 13 C NMR (150MHz, CD 3 OD) δ (ppm) 175.8, 161.2, 143.2, 140.8, 137.4, 135.8, 135.3, 129.9, 129.4, 128.4, 125.5, 125.3, 123.5, 121.2, 103.8, 60.8, 48.1, 40.1 37.7, 20.5, 6.7.
实施例62Example 62
(R)-N-(2-腈乙基)-3-(2-(1-(2-羟丙基)-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯磺酰胺(R)-N-(2-cyanoethyl)-3-(2-(1-(2-hydroxypropyl)-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b ]pyrazin-7-yl)benzenesulfonamide
步骤1:化合物(R)-N-(2-氰乙基)-3-(2-(1-(2-羟丙基)-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯磺酰胺的合成Step 1: Compound (R)-N-(2-cyanoethyl)-3-(2-(1-(2-hydroxypropyl)-1H-pyrazol-4-yl)-5-((2- Synthesis of (trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)benzenesulfonamide
室温下,将碳酸铯(840mg,2.58mmol)、4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑(250mg,1.29mmol)和(R)-环氧丙烷(285mg,4.91mmol)加到一起,再加入乙腈(15ml),封管升温至130℃反应22h,反应液用乙酸乙酯萃取(50mL×3),旋干有机相。氮气保护下,加入3-(2-溴-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-N-(2-氰乙基)苯磺酰胺(150mg,0.28mmol)、碳酸钾(100mg,0.72mmol)、[1,1'-双(二苯基膦基)二茂铁]二氯化钯(12mg,0.02mmol),再加入二氧六环(15mL)、水(3ml),110℃反应24h,反应液冷却至室温,硅藻土过滤,滤液减压浓缩,进行柱分离(乙酸乙酯)得到150mg黄色油状物,产率:92.2%。Cesium carbonate (840 mg, 2.58 mmol), 4-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)-1H-pyrazole ( 250mg, 1.29mmol) and (R)-propylene oxide (285mg, 4.91mmol) were added together, then acetonitrile (15ml) was added, the temperature was heated to 130°C for 22h in a sealed tube, and the reaction solution was extracted with ethyl acetate (50mL×3 ), spin dry the organic phase. Under nitrogen protection, 3-(2-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl was added )-N-(2-cyanoethyl)benzenesulfonamide (150mg, 0.28mmol), potassium carbonate (100mg, 0.72mmol), [1,1'-bis(diphenylphosphino)ferrocene]dichloro Palladium (12 mg, 0.02 mmol) was added, dioxane (15 mL) and water (3 ml) were added, and the reaction was carried out at 110° C. for 24 h. The reaction solution was cooled to room temperature, filtered through celite, and the filtrate was concentrated under reduced pressure, and subjected to column separation (acetic acid). ethyl ester) to give 150 mg of yellow oil, yield: 92.2%.
MS(ESI,pos.ion)m/z:582.3[M+1]+。MS(ESI, pos.ion) m/z: 582.3[M+1] + .
步骤2:化合物(R)-N-(2-腈乙基)-3-(2-(1-(2-羟丙基)-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯磺酰胺的合成Step 2: Compound (R)-N-(2-cyanoethyl)-3-(2-(1-(2-hydroxypropyl)-1H-pyrazol-4-yl)-5H-pyrrolo[2 Synthesis of ,3-b]pyrazin-7-yl)benzenesulfonamide
(R)-N-(2-氰乙基)-3-(2-(1-(2-羟丙基)-1H-吡唑-4-基)-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯磺酰胺(160mg,0.28mmol)、三氟乙酸(6mL)和乙二胺(1mL)根据实施例2步骤2的合成方法制备得到56mg白色固体,产率:45.1%。(R)-N-(2-cyanoethyl)-3-(2-(1-(2-hydroxypropyl)-1H-pyrazol-4-yl)-5-((2-(trimethyl) Silyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)benzenesulfonamide (160 mg, 0.28 mmol), trifluoroacetic acid (6 mL) and ethylenediamine ( 1 mL) was prepared according to the synthetic method of step 2 of Example 2 to obtain 56 mg of white solid, yield: 45.1%.
MS(ESI,pos.ion)m/z:452.4[M+1]+;MS(ESI, pos.ion) m/z: 452.4[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)8.98(s,1H),8.71(s,1H),8.52(s,1H),8.49(d,J=7.3Hz,1H),8.39(s,1H),8.19(s,1H),7.72–7.63(m,2H),5.00(s,2H),4.16–3.99(m,3H),3.09(t,J=6.4Hz,2H),2.68(t,J=6.4Hz,2H),1.10(d,J=5.8Hz,3H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 8.98(s, 1H), 8.71(s, 1H), 8.52(s, 1H), 8.49(d, J=7.3Hz, 1H), 8.39 (s, 1H), 8.19 (s, 1H), 7.72–7.63 (m, 2H), 5.00 (s, 2H), 4.16–3.99 (m, 3H), 3.09 (t, J=6.4Hz, 2H), 2.68 (t, J=6.4Hz, 2H), 1.10 (d, J=5.8Hz, 3H);
13C NMR(150MHz,DMSO-d6):δ(ppm)142.3,141.0,140.9,137.4,136.0,135.5,135.0,130.1,129.4,129.4,129.3,123.9,123.8,121.2,119.4,111.5,65.9,59.4,39.2,21.4,19.1。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 142.3, 141.0, 140.9, 137.4, 136.0, 135.5, 135.0, 130.1, 129.4, 129.4, 129.3, 123.9, 123.8, 121.2, 119.4, 111.5, 65.9, 59.4, 39.2, 21.4, 19.1.
运用相应的原料,按照实施例62的合成方法制备如下所示的化合物:Using the corresponding raw materials, the following compounds were prepared according to the synthetic method of Example 62:
实施例72Example 72
3-(4-(4-(2-环丙基-5H-吡咯并[2,3-b]吡嗪-7-基)-1H-吡唑-1-基)哌啶-1-基)-3-氧丙腈3-(4-(4-(2-Cyclopropyl-5H-pyrrolo[2,3-b]pyrazin-7-yl)-1H-pyrazol-1-yl)piperidin-1-yl) -3-Oxypropionitrile
步骤1:化合物1-甲酸叔丁酯-4-((4-(2-环丙基-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-1H-吡唑-1-基))哌啶的合成Step 1: Compound 1-tert-Butylcarboxylate-4-((4-(2-cyclopropyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[ Synthesis of 2,3-b]pyrazin-7-yl)-1H-pyrazol-1-yl))piperidine
1,4-二氧六环(16mL)/水(4mL)、1-甲酸叔丁酯-4-((4-(2-溴-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-1H-吡唑-1-基))哌啶(430mg,0.74mmol)、环丙基硼酸(95mg,1.11mmol)、碳酸钾(160mg,1.16mmol)和1,1'-双二苯基膦二茂铁二氯化钯(30mg,0.04mmol)的混合物根据实施例2步骤1的合成方法制备得到280mg黄色油状物,产率:69.8%。1,4-dioxane (16 mL)/water (4 mL), tert-butyl 1-carboxylate-4-((4-(2-bromo-5-((2-(trimethylsilyl)ethoxy) yl)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)-1H-pyrazol-1-yl))piperidine (430mg, 0.74mmol), cyclopropylboronic acid (95mg , 1.11 mmol), potassium carbonate (160 mg, 1.16 mmol) and 1,1'-bis-diphenylphosphinoferrocene palladium dichloride (30 mg, 0.04 mmol) were prepared according to the synthetic method of Example 2, step 1 280 mg of yellow oil, yield: 69.8%.
MS(ESI,pos.ion)m/z:539.4[M+1]+。MS(ESI, pos.ion) m/z: 539.4[M+1] + .
步骤2:化合物2-环丙基-7-(1-(哌啶-4-基)-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪的合成Step 2: Synthesis of compound 2-cyclopropyl-7-(1-(piperidin-4-yl)-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine
1-甲酸叔丁酯-4-((4-(2-环丙基-5-((2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)-1H-吡唑-1-基))哌啶(260mg,0.48mmol)、三氟乙酸(2mL)和乙二胺(1mL)根据实施例2步骤2的合成方法制备得到110mg淡黄色固体,产率:73.9%。tert-Butyl 1-carboxylate-4-((4-(2-cyclopropyl-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3- b] Pyrazin-7-yl)-1H-pyrazol-1-yl))piperidine (260 mg, 0.48 mmol), trifluoroacetic acid (2 mL) and ethylenediamine (1 mL) Synthesis according to Example 2, Step 2 The method prepared 110 mg of pale yellow solid, yield: 73.9%.
MS(ESI,pos.ion)m/z:309.3[M+1]+。MS(ESI, pos.ion) m/z: 309.3[M+1] + .
步骤3:化合物3-(4-(4-(2-环丙基-5H-吡咯并[2,3-b]吡嗪-7-基)-1H-吡唑-1-基)哌啶-1-基)-3-氧丙腈的合成Step 3: Compound 3-(4-(4-(2-Cyclopropyl-5H-pyrrolo[2,3-b]pyrazin-7-yl)-1H-pyrazol-1-yl)piperidine- Synthesis of 1-yl)-3-oxopropionitrile
将2-环丙基-7-(1-(哌啶-4-基)-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪(100mg,0.32mmol)加入到氰基乙酸(65mg,0.76mmol),1-羟基苯并三唑(HOBT)(110mg,0.81mmol),1-乙基-(3-二甲基氨基丙基)碳酰二亚胺盐酸盐(150mg,0.78mmol)和三乙胺(0.08g,0.1mL,0.80mmol)的N,N-二甲基甲酰胺(5mL)混合物中,室温下反应6h,用水(50mL)淬灭反应,乙酸乙酯萃取(50mL x 4),有机层用饱和食盐水(50mL x 2)洗涤,无水硫酸钠干燥,减压浓缩,浓缩液进行柱分离(二氯甲烷/甲醇(v/v)=12/1)得到80mg淡黄色固体,产率:65.7%。2-Cyclopropyl-7-(1-(piperidin-4-yl)-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine (100 mg, 0.32 mmol) Added to cyanoacetic acid (65mg, 0.76mmol), 1-hydroxybenzotriazole (HOBT) (110mg, 0.81mmol), 1-ethyl-(3-dimethylaminopropyl)carbodiimide salt A mixture of acid salt (150 mg, 0.78 mmol) and triethylamine (0.08 g, 0.1 mL, 0.80 mmol) in N,N-dimethylformamide (5 mL), reacted at room temperature for 6 h, and quenched with water (50 mL) , extracted with ethyl acetate (50 mL x 4), the organic layer was washed with saturated brine (50 mL x 2), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and the concentrate was subjected to column separation (dichloromethane/methanol (v/v) =12/1) 80 mg of pale yellow solid were obtained, yield: 65.7%.
MS(ESI,pos.ion)m/z:375.9[M+1]+;MS(ESI, pos.ion) m/z: 375.9[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)11.81(s,1H),8.23(d,J=6.8Hz,2H),8.01(s,1H),8.00(d,J=2.6Hz,1H),4.52(m,1H),4.43(d,J=13.2Hz,1H),4.12(q,J=18.9Hz,2H),3.79(d,J=13.7Hz,1H),3.25(m,1H),2.86(t,J=11.8Hz,1H),2.28(m,1H),2.09(m,2H),1.99(m,1H),1.82(m,1H),1.03(m,4H); 1 H NMR (600MHz, DMSO-d 6 ): δ (ppm) 11.81 (s, 1H), 8.23 (d, J=6.8Hz, 2H), 8.01 (s, 1H), 8.00 (d, J=2.6Hz ,1H),4.52(m,1H),4.43(d,J=13.2Hz,1H),4.12(q,J=18.9Hz,2H),3.79(d,J=13.7Hz,1H),3.25(m ,1H),2.86(t,J=11.8Hz,1H),2.28(m,1H),2.09(m,2H),1.99(m,1H),1.82(m,1H),1.03(m,4H) ;
13C NMR(150MHz,DMSO-d6):δ(ppm)161.8,150.8,140.8,136.4,136.1,135.8,125.9,124.7,116.6,114.2,106.5,57.9,44.8,41.1,32.7,32.1,25.3,14.8,10.2。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 161.8, 150.8, 140.8, 136.4, 136.1, 135.8, 125.9, 124.7, 116.6, 114.2, 106.5, 57.9, 44.8, 41.1, 32.7, 32.1, 25.3, 14.8, 10.2.
运用相应的原料按照实施例72的合成方法制备得到,制备如下化合物:The corresponding raw materials are used to prepare according to the synthetic method of Example 72, and the following compounds are prepared:
实施例90Example 90
5-((3-丙酰胺基苯基)胺基)吡唑并[1,5-a]嘧啶-3-甲酸甲酯Methyl 5-((3-Propionamidophenyl)amino)pyrazolo[1,5-a]pyrimidine-3-carboxylate
步骤1:化合物3-碘代吡唑[1,5-a]嘧啶-5-羟基的合成Step 1: Synthesis of compound 3-iodopyrazolo[1,5-a]pyrimidine-5-hydroxy
将吡唑[1,5-a]嘧啶-5-羟基(1g,7.40mmol)和N-碘代丁二酰亚胺(1.83g,8.14mmol)在室温下溶入N,N-二甲基甲酰胺(20mL),反应会慢慢放热,几分钟内有大量固体生成,反应在室温下搅拌1小时。冰浴下冷却,过滤,收集白色固体,用二氯甲烷(20mL)洗滤饼,干燥得到1.7g白色固体,产率:88%。Pyrazo[1,5-a]pyrimidine-5-hydroxy (1 g, 7.40 mmol) and N-iodosuccinimide (1.83 g, 8.14 mmol) were dissolved in N,N-dimethyl dimethyl at room temperature Formamide (20 mL), the reaction was slowly exothermic, a large amount of solid formed within a few minutes, and the reaction was stirred at room temperature for 1 hour. Cool under an ice bath, filter, collect the white solid, wash the filter cake with dichloromethane (20 mL), and dry to obtain 1.7 g of white solid, yield: 88%.
1H NMR(600MHz,DMSO-d6):δ(ppm)12.24(s,1H),8.48(s,1H),7.84(s,1H),6.00(s,1H)。 1 H NMR (600 MHz, DMSO-d 6 ): δ (ppm) 12.24 (s, 1H), 8.48 (s, 1H), 7.84 (s, 1H), 6.00 (s, 1H).
步骤2:化合物5-羟基吡唑[1,5-a]嘧啶-3-甲酸甲酯的合成Step 2: Synthesis of compound 5-hydroxypyrazo[1,5-a]pyrimidine-3-carboxylic acid methyl ester
N2保护下将甲醇(10mL)注入3-碘代吡唑并[1,5-a]嘧啶-5-羟基(0.2g,0.76mmol)、醋酸钯(35mg,0.15mmol)和三乙胺(0.32mL,2.3mmol)的混合物中,通入一氧化碳,反应在55℃下反应5h。硅藻土过滤,减压浓缩,残渣水洗(35mL),用二氯甲烷萃取(35mLx3),无水硫酸钠干燥,减压浓缩,进行柱分离(二氯甲烷/甲醇(v/v)=30/1)得到45mg白色固体,产率:30.4%。Methanol (10 mL) was injected under N2 into 3-iodopyrazolo[1,5-a]pyrimidine-5-hydroxy (0.2 g, 0.76 mmol), palladium acetate (35 mg, 0.15 mmol) and triethylamine ( 0.32 mL, 2.3 mmol), carbon monoxide was introduced into the mixture, and the reaction was carried out at 55° C. for 5 h. Celite was filtered, concentrated under reduced pressure, the residue was washed with water (35 mL), extracted with dichloromethane (35 mL×3), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and subjected to column separation (dichloromethane/methanol (v/v)=30 /1) 45 mg of white solid were obtained, yield: 30.4%.
1H NMR(400MHz,DMSO-d6):δ(ppm)11.87(s,1H),8.57(d,J=7.9Hz,1H),8.15(s,1H),6.16(s,1H),3.77(s,3H)。 1 H NMR (400MHz, DMSO-d 6 ): δ(ppm) 11.87(s, 1H), 8.57(d, J=7.9Hz, 1H), 8.15(s, 1H), 6.16(s, 1H), 3.77 (s, 3H).
步骤3:化合物5-氯吡唑并[1,5-a]嘧啶-3-甲酸甲酯的合成Step 3: Synthesis of compound 5-chloropyrazolo[1,5-a]pyrimidine-3-carboxylic acid methyl ester
5-羟基吡唑[1,5-a]嘧啶-3-甲酸甲酯(0.028g,0.14mmol)、三氯氧磷(0.8mL,8mmol)和N,N-二异丙基乙胺(0.01mL,0.06mmol)在甲苯(2mL)溶液中回流反应2h。冷却至室温,减压除去三氯氧磷,残渣用温水(35mL)淬灭,二氯甲烷(35mL x 3)萃取,无水硫酸钠干燥,减压浓缩,进行柱分离(二氯甲烷/甲醇(v/v)=80/1)得到19mg白色固体,产率:61.9%。Methyl 5-hydroxypyrazo[1,5-a]pyrimidine-3-carboxylate (0.028 g, 0.14 mmol), phosphorus oxychloride (0.8 mL, 8 mmol) and N,N-diisopropylethylamine (0.01 mL, 0.06 mmol) was refluxed in toluene (2 mL) solution for 2 h. Cooled to room temperature, phosphorus oxychloride was removed under reduced pressure, the residue was quenched with warm water (35 mL), extracted with dichloromethane (35 mL x 3), dried over anhydrous sodium sulfate, concentrated under reduced pressure, and subjected to column separation (dichloromethane/methanol). (v/v)=80/1) 19 mg of white solid were obtained, yield: 61.9%.
1H NMR(400MHz,DMSO-d6):δ(ppm)9.33(d,J=7.2Hz,1H),8.68(s,1H),7.41(d,J=7.2Hz,1H),3.83(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 9.33 (d, J=7.2 Hz, 1H), 8.68 (s, 1H), 7.41 (d, J=7.2 Hz, 1H), 3.83 (s , 3H).
步骤4:化合物5-((3-丙酰胺基苯基)胺基)吡唑[1,5-a]嘧啶-3-甲酸甲酯的合成Step 4: Synthesis of compound 5-((3-propionamidophenyl)amino)pyrazo[1,5-a]pyrimidine-3-carboxylic acid methyl ester
N2保护下,将1,4-二氧六环(8mL)加入到5-氯吡唑[1,5-a]嘧啶-3-甲酸甲酯(0.08g,0.37mmol)、Xantphos(0.011g,0.02mmol)、醋酸钯(4mg,0.02mmol)、碳酸铯(0.14g,0.42mmol)和N-(3-胺基苯)丙酰胺(0.08g,0.48mmol)的混合物中,110℃回流反应6h,过滤,滤液用水(30mL)洗,水相用二氯甲烷反萃(30mL x 3),合并有机层,无水硫酸钠干燥,减压浓缩,进行柱分离(二氯甲烷/甲醇(v/v)=30/1)得到棕黄色固体,再利用高效色谱法分离得到20mg米黄色固体,产率:15.59%。Under N2 protection, 1,4-dioxane (8 mL) was added to methyl 5-chloropyrazolo[1,5-a]pyrimidine-3-carboxylate (0.08 g, 0.37 mmol), Xantphos (0.011 g , 0.02mmol), palladium acetate (4mg, 0.02mmol), cesium carbonate (0.14g, 0.42mmol) and N-(3-aminobenzene) propionamide (0.08g, 0.48mmol) in a mixture, 110 ° C reflux reaction 6h, filtered, the filtrate was washed with water (30 mL), the aqueous phase was back-extracted with dichloromethane (30 mL x 3), the organic layers were combined, dried over anhydrous sodium sulfate, concentrated under reduced pressure, and subjected to column separation (dichloromethane/methanol (v /v)=30/1) to obtain a brownish-yellow solid, which was then separated by high performance chromatography to obtain 20 mg of a beige solid, yield: 15.59%.
MS(ESI,pos.ion)m/z:340.1[M+1]+;MS(ESI, pos.ion) m/z: 340.1[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)10.08(s,1H),9.87(s,1H),8.75(d,J=7.6Hz,1H),8.36(d,J=6.7Hz,1H),8.29(s,1H),7.94(s,1H),7.30(t,J=8.1Hz,1H),7.12(d,J=7.8Hz,1H),6.70(d,J=7.6Hz,1H),3.81(s,3H),2.34(q,J=7.5Hz,2H),1.11(t,J=7.5Hz,3H); 1 H NMR (600 MHz, DMSO-d 6 ): δ (ppm) 10.08 (s, 1H), 9.87 (s, 1H), 8.75 (d, J=7.6 Hz, 1H), 8.36 (d, J=6.7 Hz) ,1H),8.29(s,1H),7.94(s,1H),7.30(t,J=8.1Hz,1H),7.12(d,J=7.8Hz,1H),6.70(d,J=7.6Hz , 1H), 3.81(s, 3H), 2.34(q, J=7.5Hz, 2H), 1.11(t, J=7.5Hz, 3H);
13C NMR(150MHz,DMSO-d6):δ(ppm)172.0,162.6,154.7,147.2,146.0,140.0,139.4,136.4,129.0,114.2,113.9,110.3,102.2,98.5,50.7,29.5,9.7。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 172.0, 162.6, 154.7, 147.2, 146.0, 140.0, 139.4, 136.4, 129.0, 114.2, 113.9, 110.3, 102.2, 98.5, 50.7, 29.5, 9.7.
实施例91Example 91
N-(2-氰乙基)-3-(1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺N-(2-cyanoethyl)-3-(1,6-dihydropyrrole[2,3-b][1,2,3]triazolo[4,5-d]pyridin-8-yl) Benzenesulfonamide
步骤1:化合物4-氯-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶的合成Step 1: Synthesis of compound 4-chloro-1-p-toluenesulfonyl-1H-pyrro[2,3-b]pyridine
室温下,将4-氯-1H-吡咯[2,3-b]吡啶(1.50g,9.80mmol)、三乙胺(1.81g,18.01mmol)、DMAP(205mg,1.59mmol)加到一起,再依次加入二氯甲烷(20mL)、对甲苯磺酰氯(2.25g,11.80mmol),室温反应36h,反应液加水(50mL)淬灭,用二氯甲烷萃取(50mL×3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=10/1)得到1.46g白色固体,产率49.1%。4-Chloro-1H-pyrrole[2,3-b]pyridine (1.50 g, 9.80 mmol), triethylamine (1.81 g, 18.01 mmol), DMAP (205 mg, 1.59 mmol) were added together at room temperature, and then Dichloromethane (20 mL) and p-toluenesulfonyl chloride (2.25 g, 11.80 mmol) were successively added, and the reaction was carried out at room temperature for 36 h. The reaction solution was quenched by adding water (50 mL), extracted with dichloromethane (50 mL×3), and the organic phase was spin-dried. Column chromatography separation (petroleum ether/ethyl acetate (v/v)=10/1) gave 1.46 g of white solid in a yield of 49.1%.
MS(ESI,pos.ion)m/z:306.9[M+1]+。MS(ESI, pos.ion) m/z: 306.9[M+1] + .
步骤2:化合物4-氯-5-硝基-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶的合成Step 2: Synthesis of compound 4-chloro-5-nitro-1-p-toluenesulfonyl-1H-pyrro[2,3-b]pyridine
-5℃下,将4-氯-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶(1.48g,4.82mmol)、四丁基硝酸铵(1.84g,5.86mmol)、二氯甲烷(30mL)加到一起,然后缓慢加入三氟乙酸酐(0.92mL,6.50mmol),加完后升至室温反应6h,-5℃下补加四丁基硝酸铵(0.46g,1.45mmol)、三氟乙酸酐(0.28mL,2.01mmol),加完后升至室温反应18h,反应液加水(70mL)淬灭,用二氯甲烷萃取(100mL×3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=10/1)得到1.21g白色固体,产率:71.6%。At -5 °C, 4-chloro-1-p-toluenesulfonyl-1H-pyrrole[2,3-b]pyridine (1.48g, 4.82mmol), tetrabutylammonium nitrate (1.84g, 5.86mmol), di Chloromethane (30 mL) was added together, then trifluoroacetic anhydride (0.92 mL, 6.50 mmol) was slowly added, and after the addition, the reaction was raised to room temperature for 6 h, and tetrabutylammonium nitrate (0.46 g, 1.45 mmol) was added at -5°C. ), trifluoroacetic anhydride (0.28mL, 2.01mmol), after adding, the reaction was raised to room temperature for 18h, the reaction solution was quenched by adding water (70mL), extracted with dichloromethane (100mL×3), spin-dried the organic phase, the column layer Separation (petroleum ether/ethyl acetate (v/v)=10/1) gave 1.21 g of white solid, yield: 71.6%.
MS(ESI,pos.ion)m/z:352.1[M+1]+。MS(ESI, pos.ion) m/z: 352.1[M+1] + .
步骤3:化合物5-硝基-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶-4-胺的合成Step 3: Synthesis of compound 5-nitro-1-p-toluenesulfonyl-1H-pyrro[2,3-b]pyridin-4-amine
室温下,将4-氯-5-硝基-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶(300mg,0.85mmol)、浓氨水(2mL)加到一起,再依次加入二氧六环(20mL),80℃反应6h,反应液冷却至室温,加水(50mL)稀释,用二氯甲烷萃取(50mL×3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=4/1)得到130mg黄色固体,产率:45.9%。At room temperature, 4-chloro-5-nitro-1-p-toluenesulfonyl-1H-pyrrole[2,3-b]pyridine (300 mg, 0.85 mmol) and concentrated ammonia (2 mL) were added together, followed by Dioxane (20 mL) was reacted at 80 °C for 6 h, the reaction solution was cooled to room temperature, diluted with water (50 mL), extracted with dichloromethane (50 mL×3), the organic phase was spin-dried, separated by column chromatography (petroleum ether/acetic acid) Ethyl ester (v/v)=4/1) gave 130 mg of yellow solid, yield: 45.9%.
MS(ESI,pos.ion)m/z:333.2[M+1]+。MS(ESI, pos.ion) m/z: 333.2[M+1] + .
步骤4:化合物1-对甲苯磺酰基-1H-吡咯[2,3-b]吡嗪-4,5-二胺的合成Step 4: Synthesis of Compound 1-p-Tosyl-1H-pyrrole[2,3-b]pyrazine-4,5-diamine
室温下,将5-硝基-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶-4-胺(130mg,0.39mmol)、氯化铵(124mg,2.34mmol)、铁粉(90mg,1.61mmol)加到一起,再加入乙醇(10mL)、水(3mL),90℃反应4h,反应液冷却至室温,硅藻土过滤后加水(50mL)稀释,用乙酸乙酯萃取(50mL×3),旋干有机相,柱层析分离(二氯甲烷/甲醇(v/v)=20/1)得到80mg棕色油状物,产率:67.6%。At room temperature, 5-nitro-1-p-toluenesulfonyl-1H-pyrrole[2,3-b]pyridin-4-amine (130mg, 0.39mmol), ammonium chloride (124mg, 2.34mmol), iron powder (90 mg, 1.61 mmol) were added together, then ethanol (10 mL) and water (3 mL) were added, and the reaction was carried out at 90° C. for 4 h. The reaction solution was cooled to room temperature, filtered through celite, diluted with water (50 mL), and extracted with ethyl acetate ( 50 mL×3), spin dry the organic phase, and separate by column chromatography (dichloromethane/methanol (v/v)=20/1) to obtain 80 mg of brown oil, yield: 67.6%.
MS(ESI,pos.ion)m/z:303.2[M+1]+。MS(ESI, pos.ion) m/z: 303.2[M+1] + .
步骤5:化合物6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶的合成Step 5: Synthesis of compound 6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine
将1-对甲苯磺酰基-1H-吡咯[2,3-b]吡嗪-4,5-二胺(70mg,0.23mmol)、亚硝酸钠(19mg,0.28mmol)加到一起,再加入乙酸(8mL),室温反应3h,加水(50mL)稀释,用乙酸乙酯萃取(50mL×3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=1/2)得到40mg浅黄色固体,产率:55.2%。1-p-Tosyl-1H-pyrrole[2,3-b]pyrazine-4,5-diamine (70 mg, 0.23 mmol), sodium nitrite (19 mg, 0.28 mmol) were added together followed by acetic acid (8mL), react at room temperature for 3h, add water (50mL) to dilute, extract with ethyl acetate (50mL×3), spin dry the organic phase, separate by column chromatography (petroleum ether/ethyl acetate (v/v)=1/2 ) to obtain 40 mg of a pale yellow solid, yield: 55.2%.
MS(ESI,pos.ion)m/z:314.3[M+1]+;MS(ESI, pos.ion) m/z: 314.3[M+1] + ;
1H NMR(400MHz,DMSO-d6):δ(ppm)9.19(s,1H),8.03(t,J=5.5Hz,3H),7.43(d,J=8.3Hz,2H),7.07(d,J=3.9Hz,1H),2.34(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 9.19 (s, 1H), 8.03 (t, J=5.5 Hz, 3H), 7.43 (d, J=8.3 Hz, 2H), 7.07 (d , J=3.9Hz, 1H), 2.34 (s, 3H).
步骤6:化合物8-溴-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶的合成Step 6: Synthesis of compound 8-bromo-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine
室温下,将6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶(150mg,0.48mmol)、NBS(128mg,0.72mmol)加到一起,再加入DMF(10mL),室温反应10h,加水(50mL)稀释,用乙酸乙酯萃取(50mL×3),旋干有机相,柱层析分离(石油醚/乙酸乙酯(v/v)=1/2)得到160mg浅黄色固体,送HPLC制备得到79mg浅黄色固体,产率:42.1%。At room temperature, 6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine (150 mg, 0.48 mmol), NBS (128 mg, 0.72 mmol) was added together, then DMF (10 mL) was added, the reaction was carried out at room temperature for 10 h, diluted with water (50 mL), extracted with ethyl acetate (50 mL×3), the organic phase was spin-dried, separated by column chromatography (petroleum ether/ethyl acetate (v/v)=1/2) to obtain 160 mg of pale yellow solid, which was sent to HPLC to prepare 79 mg of pale yellow solid, yield: 42.1%.
MS(ESI,pos.ion)m/z:392.0[M+1]+;MS(ESI, pos.ion) m/z: 392.0[M+1] + ;
1H NMR(400MHz,DMSO-d6+D2O):δ(ppm)9.23(s,1H),8.26(s,1H),8.04(d,J=8.4Hz,2H),7.42(d,J=8.2Hz,2H),2.31(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 +D 2 O): δ (ppm) 9.23 (s, 1H), 8.26 (s, 1H), 8.04 (d, J=8.4 Hz, 2H), 7.42 (d, J=8.2Hz, 2H), 2.31 (s, 3H).
步骤7:化合物N-(2-氰乙基)-3-(6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺的合成Step 7: Compound N-(2-cyanoethyl)-3-(6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4 Synthesis of ,5-d]pyridin-8-yl)benzenesulfonamide
氮气保护下,将1,4-二氧六环(12mL)/水(3mL)加入到N-(2-氰乙基)-3-(4,4,5,5-四甲基-1,3,2-二氧杂硼烷-2-基)苯磺酰胺(132mg,0.39mmol)、8-溴-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶(79mg,0.20mmol)、碳酸钾(73mg,0.52mmol)、Pd(dppf)Cl2(12mg,0.02mmol)混合物中,110℃回流反应7h,反应液冷却至室温,硅藻土过滤,滤液减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=1/3)得到95mg棕色油状物,产率:90.5%。Under nitrogen, 1,4-dioxane (12 mL)/water (3 mL) was added to N-(2-cyanoethyl)-3-(4,4,5,5-tetramethyl-1, 3,2-dioxaboran-2-yl)benzenesulfonamide (132 mg, 0.39 mmol), 8-bromo-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][ 1,2,3]Triazolo[4,5-d]pyridine (79mg, 0.20mmol), potassium carbonate (73mg, 0.52mmol), Pd(dppf)Cl 2 (12mg, 0.02mmol) mixture, 110°C The reaction was refluxed for 7 h, the reaction solution was cooled to room temperature, filtered through celite, the filtrate was concentrated under reduced pressure, and subjected to column separation (petroleum ether/ethyl acetate (v/v)=1/3) to obtain 95 mg of brown oil, yield: 90.5 %.
MS(ESI,pos.ion)m/z:522.2[M+1]+。MS(ESI, pos.ion) m/z: 522.2[M+1] + .
步骤8:化合物N-(2-氰乙基)-3-(1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺的合成Step 8: Compound N-(2-cyanoethyl)-3-(1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine- Synthesis of 8-yl)benzenesulfonamide
室温下,将N-(2-氰乙基)-3-(6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺(106mg,0.20mmol)、叔丁醇钾(100mg,0.84mmol)加到一起,再加入无水四氢呋喃(5mL),室温反应3h,反应液加入硅胶室温旋干拌样,柱层析分离(二氯甲烷/甲醇(v/v)=10/1)得到100mg红色固体,然后用HPLC制备得到50mg白色固体,产率:67.0%。At room temperature, N-(2-cyanoethyl)-3-(6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4 ,5-d]pyridin-8-yl)benzenesulfonamide (106mg, 0.20mmol), potassium tert-butoxide (100mg, 0.84mmol) were added together, then anhydrous tetrahydrofuran (5mL) was added, and the reaction was carried out at room temperature for 3h. The reaction solution Silica gel was added to dry at room temperature, and the sample was mixed, and separated by column chromatography (dichloromethane/methanol (v/v)=10/1) to obtain 100 mg of red solid, which was then prepared by HPLC to obtain 50 mg of white solid, yield: 67.0%.
MS(ESI,pos.ion)m/z:368.0[M+1]+;MS(ESI, pos.ion) m/z: 368.0[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)11.77(s,1H),9.16(d,J=7.8Hz,1H),8.94(s,1H),8.75(s,1H),7.80(s,1H),7.65(t,J=7.8Hz,1H),7.57(d,J=7.9Hz,1H),5.33(t,J=4.6Hz,1H),3.21(t,J=6.3Hz,2H),2.80(t,J=6.3Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 11.77(s, 1H), 9.16(d, J=7.8Hz, 1H), 8.94(s, 1H), 8.75(s, 1H), 7.80 (s, 1H), 7.65 (t, J=7.8Hz, 1H), 7.57 (d, J=7.9Hz, 1H), 5.33 (t, J=4.6Hz, 1H), 3.21 (t, J=6.3Hz) ,2H),2.80(t,J=6.3Hz,2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)143.5,143.3,141.1,138.1,136.2,130.7,130.1,129.7,123.9,122.5,119.7,119.4,114.8,105.1,35.6,19.2。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 143.5, 143.3, 141.1, 138.1, 136.2, 130.7, 130.1, 129.7, 123.9, 122.5, 119.7, 119.4, 114.8, 105.1, 35.6, 19.2.
实施例92Example 92
N-(2-氰乙基)-3-(1-甲基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺N-(2-cyanoethyl)-3-(1-methyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine -8-yl)benzenesulfonamide
步骤1:化合物N-甲基-5-硝基-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶-4-胺的合成Step 1: Synthesis of compound N-methyl-5-nitro-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridin-4-amine
将4-氯-5-硝基-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶(300mg,0.85mmol)、甲胺乙醇溶液(6.38M,15mL,95.7mmol)按照实施例15步骤3的合成方法制备得到184mg黄色固体,产率:62.3%。4-Chloro-5-nitro-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridine (300 mg, 0.85 mmol), methylamine ethanol solution (6.38 M, 15 mL, 95.7 mmol) were combined according to The synthetic method of step 3 of Example 15 prepared 184 mg of yellow solid, yield: 62.3%.
MS(ESI,pos.ion)m/z:333.2[M+1]+。MS(ESI, pos.ion) m/z: 333.2[M+1] + .
步骤2:化合物N-甲基-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶-4,5-二胺的合成Step 2: Synthesis of compound N-methyl-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridine-4,5-diamine
将N-甲基-5-硝基-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶-4-胺(40mg,0.12mmol)、氯化铵(37mg,0.70mmol)、铁粉(26mg,0.47mmol)乙醇(10mL)、水(3mL)按照实施例15步骤4的合成方法制备得到 20mg红色固体,产率:54.7%。N-methyl-5-nitro-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridin-4-amine (40 mg, 0.12 mmol), ammonium chloride (37 mg, 0.70 mmol) , iron powder (26 mg, 0.47 mmol), ethanol (10 mL) and water (3 mL) were prepared according to the synthesis method of step 4 of Example 15 to obtain 20 mg of red solid, yield: 54.7%.
MS(ESI,pos.ion)m/z:317.2[M+1]+。MS(ESI, pos.ion) m/z: 317.2[M+1] + .
步骤3:化合物1-甲基-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶的合成Step 3: Synthesis of compound 1-methyl-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine
将N-甲基-1-对甲苯磺酰基-1H-吡咯[2,3-b]吡啶-4,5-二胺(215mg,0.68mmol)、亚硝酸钠(56mg,0.81mmol)和乙酸(8mL)按照实施例15步骤5的合成方法制备得到150mg浅黄色固体,产率:67.2%。N-methyl-1-p-toluenesulfonyl-1H-pyrro[2,3-b]pyridine-4,5-diamine (215 mg, 0.68 mmol), sodium nitrite (56 mg, 0.81 mmol) and acetic acid ( 8 mL) was prepared according to the synthesis method of step 5 of Example 15 to obtain 150 mg of light yellow solid, yield: 67.2%.
MS(ESI,pos.ion)m/z:328.2[M+1]+。MS(ESI, pos.ion) m/z: 328.2[M+1] + .
步骤4:化合物1-甲基-8-溴-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶的合成Step 4: Compound 1-methyl-8-bromo-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d ] Pyridine Synthesis
将1-甲基-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶(156mg,0.48mmol)、NBS(128mg,0.72mmol)、DMF(10mL)按照实施例15步骤6的合成方法制备得到160mg浅黄色固体,送HPLC制备得到53mg浅黄色固体,产率:27.3%。1-Methyl-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d]pyridine (156 mg, 0.48 mmol ), NBS (128 mg, 0.72 mmol), DMF (10 mL) were prepared according to the synthetic method of step 6 of Example 15 to obtain 160 mg of light yellow solid, and sent to HPLC to prepare 53 mg of light yellow solid, yield: 27.3%.
MS(ESI,pos.ion)m/z:406.1[M+1]+;MS(ESI, pos.ion) m/z: 406.1[M+1] + ;
1H NMR(400MHz,DMSO-d6):δ(ppm)9.26(s,1H),8.34(s,1H),8.07(d,J=8.4Hz,2H),7.44(d,J=8.2Hz,2H),4.69(s,3H),2.34(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 9.26 (s, 1H), 8.34 (s, 1H), 8.07 (d, J=8.4 Hz, 2H), 7.44 (d, J=8.2 Hz , 2H), 4.69(s, 3H), 2.34(s, 3H).
步骤5:化合物N-(2-氰乙基)-3-(1-甲基-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺的合成Step 5: Compound N-(2-cyanoethyl)-3-(1-methyl-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3] Synthesis of Triazolo[4,5-d]pyridin-8-yl)benzenesulfonamide
1,4-二氧六环(12mL)/水(3mL)、N-(2-氰乙基)-3-(4,4,5,5-四甲基-1,3,2-二氧杂硼烷-2-基)苯磺酰胺(82mg,0.24mmol)、1-甲基-8-溴-6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶(53mg,0.14mmol)、碳酸钾(50mg,0.36mmol)、Pd(dppf)Cl2(7mg,0.01mmol)的混合物按照实施例15步骤7的合成方法制备得到60mg浅黄色固体,产率:85.8%。1,4-dioxane (12mL)/water (3mL), N-(2-cyanoethyl)-3-(4,4,5,5-tetramethyl-1,3,2-dioxo Boran-2-yl)benzenesulfonamide (82 mg, 0.24 mmol), 1-methyl-8-bromo-6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1 ,2,3] A mixture of triazo[4,5-d]pyridine (53 mg, 0.14 mmol), potassium carbonate (50 mg, 0.36 mmol), Pd(dppf)Cl 2 (7 mg, 0.01 mmol) according to Example 15 The synthesis method of step 7 prepared 60 mg of pale yellow solid, yield: 85.8%.
MS(ESI,pos.ion)m/z:536.2[M+1]+。MS(ESI, pos.ion) m/z: 536.2[M+1] + .
步骤6:化合物N-(2-氰乙基)-3-(1-甲基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺的合成Step 6: Compound N-(2-cyanoethyl)-3-(1-methyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5 Synthesis of -d]pyridin-8-yl)benzenesulfonamide
N-(2-氰乙基)-3-(6-对甲苯磺酰基-1,6-二氢吡咯[2,3-b][1,2,3]三氮唑[4,5-d]吡啶-8-基)苯磺酰胺(76mg,0.14mmol)、叔丁醇钾(50mg,0.42mmol)按照实施例15步骤8的合成方法制备得到60mg浅黄色油状物,然后用二氯甲烷/甲醇的混合液打浆得到30mg土灰色固体,产率:55.4%。N-(2-cyanoethyl)-3-(6-p-toluenesulfonyl-1,6-dihydropyrrole[2,3-b][1,2,3]triazole[4,5-d ] Pyridin-8-yl)benzenesulfonamide (76mg, 0.14mmol), potassium tert-butoxide (50mg, 0.42mmol) were prepared according to the synthetic method of Example 15 step 8 to obtain 60mg of pale yellow oil, and then dichloromethane/ The methanol mixture was slurried to obtain 30 mg of earthy gray solids, yield: 55.4%.
MS(ESI,pos.ion)m/z:382.2[M+1]+;MS(ESI, pos.ion) m/z: 382.2[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.67(s,1H),9.08(s,1H),8.15(s,1H),7.98(s,1H),7.92(d,J=7.5Hz,1H),7.87(d,J=7.8Hz,1H),7.73(t,J=7.7Hz,1H),7.69(s,1H),3.82(s,3H),3.05(t,J=6.3Hz,2H),2.65(t,J=6.3Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 12.67(s, 1H), 9.08(s, 1H), 8.15(s, 1H), 7.98(s, 1H), 7.92(d, J= 7.5Hz, 1H), 7.87(d, J=7.8Hz, 1H), 7.73(t, J=7.7Hz, 1H), 7.69(s, 1H), 3.82(s, 3H), 3.05(t, J= 6.3Hz, 2H), 2.65(t, J=6.3Hz, 2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)145.5,140.5,140.2,137.6,137.5,134.8,132.9,130.0,128.3,125.9,124.1,119.4,114.9,101.5,39.0,38.2,19.2。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 145.5, 140.5, 140.2, 137.6, 137.5, 134.8, 132.9, 130.0, 128.3, 125.9, 124.1, 119.4, 114.9, 101.5, 39.0, 38.2, 19.2.
实施例93Example 93
N-(2-氰基乙基)-3-(3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-1-基)苯磺酰胺N-(2-cyanoethyl)-3-(3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazin-1-yl)benzenesulfonamide
步骤1:化合物(7-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-2-基)氨基甲酸叔丁酯的合成Step 1: Compound (7-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-2-yl)carbamic acid Synthesis of tert-butyl ester
向2,7-二溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪(100mg,0.24mmol)的1,4-二氧六环(4mL)溶液中依次加入xantphos(30mg,0.05mmol),碳酸钾(50mg,0.36mmol),醋酸钯(6mg,0.03mmol)和氨基甲酸叔丁酯(45mg,0.38mmol),氮气氛围下,115℃回流反应6小时,硅藻土过滤,浓缩,柱层析(石油醚/乙酸乙酯(v/v)=8/1)得到100mg棕色油状物,产率:91.83%。To 2,7-dibromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine (100 mg, 0.24 mmol) in 1 , 4-dioxane (4mL) solution was successively added xantphos (30mg, 0.05mmol), potassium carbonate (50mg, 0.36mmol), palladium acetate (6mg, 0.03mmol) and tert-butyl carbamate (45mg, 0.38mmol) ), under nitrogen atmosphere, refluxed at 115°C for 6 hours, filtered through celite, concentrated, column chromatography (petroleum ether/ethyl acetate (v/v)=8/1) to obtain 100 mg of brown oil, yield: 91.83 %.
MS(ESI,pos.ion)m/z:443.2,445.3[M+1]+。MS(ESI, pos.ion) m/z: 443.2, 445.3[M+1] + .
步骤2:化合物7-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-2-氨基的合成Step 2: Synthesis of compound 7-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine-2-amino
向(7-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-2-基)氨基甲酸叔丁酯(100mg,0.22mmol)的二氯甲烷(2mL)溶液中,加入氯化氢异丙醇溶液(7N,4mL),室温搅拌6小时,除去溶剂,加入饱和碳酸氢钠调至pH>7,二氯甲烷萃取(25mL x 3),干燥浓缩,残留物柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到58mg淡黄色固体产物,产率:74.91%。tert-butyl to (7-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-2-yl)carbamate (100mg, 0.22mmol) in dichloromethane (2mL), add hydrogen chloride isopropanol solution (7N, 4mL), stir at room temperature for 6 hours, remove the solvent, add saturated sodium bicarbonate to adjust pH>7, dichloromethane Extraction (25 mL x 3), drying and concentration, the residue was subjected to column chromatography (petroleum ether/ethyl acetate (v/v)=0/1) to obtain 58 mg of pale yellow solid product, yield: 74.91%.
MS(ESI,pos.ion)m/z:343.2,345.1[M+1]+。MS(ESI, pos.ion) m/z: 343.2,345.1[M+1] + .
步骤3:化合物1-溴-3-((2-(三甲基硅基)乙氧基)甲基)-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪的合成Step 3: Compound 1-bromo-3-((2-(trimethylsilyl)ethoxy)methyl)-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyridine Synthesis of oxazine
向7-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-2-氨基(58mg,0.17mmol)的乙醇(6mL)溶液中依次加入40%氯乙醛水溶液(70μL,0.42mmol),85℃回流反应6小时,直接加硅胶拌样,柱层析得到60mg淡黄色固体,产率:96.69%。To 7-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazine-2-amino (58 mg, 0.17 mmol) 40% aqueous chloroacetaldehyde solution (70 μL, 0.42 mmol) was sequentially added to the ethanol (6 mL) solution, and the reaction was refluxed at 85° C. for 6 hours. Silica gel was directly added to stir the sample, and 60 mg of pale yellow solid was obtained by column chromatography, yield: 96.69%.
MS(ESI,pos.ion)m/z:368.6[M+1]+。MS(ESI, pos.ion) m/z: 368.6[M+1] + .
步骤4:化合物N-(2-氰基乙基)-3-(3-((2-(三甲基硅基)乙氧基)甲基)-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-1-基)苯磺酰胺的合成Step 4: Compound N-(2-cyanoethyl)-3-(3-((2-(trimethylsilyl)ethoxy)methyl)-3H-imidazo[1,2-a] Synthesis of pyrrolo[2,3-e]pyrazin-1-yl)benzenesulfonamide
1-溴-3-((2-(三甲基硅基)乙氧基)甲基)-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪(20mg,0.05mmol)的1,4-二氧六环(5mL)溶液、碳酸钾(12mg,0.08mmol),N-(2-氰基乙基)-3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯磺酰胺(30mg,0.09mmol)、Pd(dppf)Cl2(6mg,0.008mmol)和水(1mL)按照实施例15步骤7的合成方法制备得到7mg黄色固体,产率:25.88%。1-Bromo-3-((2-(trimethylsilyl)ethoxy)methyl)-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine (20 mg, 0.05 mmol) in 1,4-dioxane (5 mL), potassium carbonate (12 mg, 0.08 mmol), N-(2-cyanoethyl)-3-(4,4,5,5-tetramethyl) yl-1,3,2-dioxaboropentan-2-yl)benzenesulfonamide (30 mg, 0.09 mmol), Pd(dppf)Cl 2 (6 mg, 0.008 mmol) and water (1 mL) following the procedure of Example 15 The synthetic method of 7 prepared 7 mg of yellow solid, yield: 25.88%.
MS(ESI,pos.ion)m/z:497.4[M+1]+。MS(ESI, pos.ion) m/z: 497.4[M+1] + .
步骤5:化合物N-(2-氰基乙基)-3-(3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-1-基)苯磺酰胺的合成Step 5: Synthesis of Compound N-(2-cyanoethyl)-3-(3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazin-1-yl)benzenesulfonamide
向N-(2-氰基乙基)-3-(3-((2-(三甲基硅基)乙氧基)甲基)-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-1-基)苯磺酰胺(100mg,0.20mmol)的二氯甲烷(6mL)溶液中加入三氟乙酸(2mL,26.93mmol),室温搅拌3小时,浓缩溶剂,加入四氢呋喃(6mL)溶解,加入乙二胺调至pH>7,继续室温搅拌,加水(20mL)稀释,二氯甲烷萃取(20mL x 3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到37mg黄色固体,产率:50.16%。to N-(2-cyanoethyl)-3-(3-((2-(trimethylsilyl)ethoxy)methyl)-3H-imidazo[1,2-a]pyrrolo[ Trifluoroacetic acid (2 mL, 26.93 mmol) was added to a solution of 2,3-e]pyrazin-1-yl)benzenesulfonamide (100 mg, 0.20 mmol) in dichloromethane (6 mL), stirred at room temperature for 3 hours, and the solvent was concentrated, Add tetrahydrofuran (6mL) to dissolve, add ethylenediamine to adjust to pH>7, continue to stir at room temperature, add water (20mL) to dilute, extract with dichloromethane (20mL x 3), dry over anhydrous sodium sulfate, and concentrate by column chromatography (petroleum ether). /ethyl acetate (v/v)=0/1) to give 37 mg of yellow solid, yield: 50.16%.
MS(ESI,pos.ion)m/z:367.25[M+1]+;MS(ESI, pos.ion) m/z: 367.25[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.62(s,1H),8.74(s,1H),7.99(s,1H),7.89(d,J=7.6Hz,1H),7.85(d,J=7.9Hz,1H),7.78(d,J=7.8Hz,1H),7.77(s,1H),7.74(s,1H),7.69(s,1H),3.09(t,J=6.3Hz,2H),2.66(t,J=6.3Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 12.62(s, 1H), 8.74(s, 1H), 7.99(s, 1H), 7.89(d, J=7.6Hz, 1H), 7.85 (d, J=7.9Hz, 1H), 7.78(d, J=7.8Hz, 1H), 7.77(s, 1H), 7.74(s, 1H), 7.69(s, 1H), 3.09(t, J= 6.3Hz, 2H), 2.66(t, J=6.3Hz, 2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)141.1,140.0,135.3,134.7,134.6,134.4,133.4,130.4,127.0,125.3,122.0,119.3,112.9,112.2,110.3,39.1,19.1。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 141.1, 140.0, 135.3, 134.7, 134.6, 134.4, 133.4, 130.4, 127.0, 125.3, 122.0, 119.3, 112.9, 112.2, 110.3, 39.1, 19.1.
实施例94Example 94
N-(2-氰乙基)-3-(3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-8-基)苯磺酰胺N-(2-cyanoethyl)-3-(3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazin-8-yl)benzenesulfonamide
步骤1:化合物2-溴-5-对甲苯磺酰基-5H-吡咯并[2,3-b]吡嗪的合成Step 1: Synthesis of compound 2-bromo-5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazine
-15℃,向2-溴-5H-吡咯并[2,3-b]吡嗪(10g,50.5mmol)的四氢呋喃(150mL)溶液中加入氢化钠(60%,3.1g,78mmol)后,室温搅拌1小时,冰浴下加入对甲苯磺酰氯(12.6g,65.4mmol),继续室温搅拌,加水(100mL)稀释,二氯甲烷萃取(100mL x 3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=8/1)得到14.1g淡黄色絮状固体,产率:79.3%。-15°C, sodium hydride (60%, 3.1 g, 78 mmol) was added to a solution of 2-bromo-5H-pyrrolo[2,3-b]pyrazine (10 g, 50.5 mmol) in tetrahydrofuran (150 mL). Stir for 1 hour, add p-toluenesulfonyl chloride (12.6 g, 65.4 mmol) under ice bath, continue to stir at room temperature, add water (100 mL) to dilute, extract with dichloromethane (100 mL x 3), dry over anhydrous sodium sulfate, and concentrate for column chromatography (Petroleum ether/ethyl acetate (v/v)=8/1) 14.1 g of pale yellow flocculent solid were obtained, yield: 79.3%.
MS(ESI,pos.ion)m/z:352.1,354.1[M+1]+。MS(ESI, pos.ion) m/z: 352.1, 354.1[M+1] + .
步骤2:化合物(5-对甲苯磺酰基-5H-吡咯并[2,3-b]吡嗪-2-基)氨基甲酸叔丁酯的合成Step 2: Synthesis of compound (5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazin-2-yl)carbamate tert-butyl ester
2-溴-5-对甲苯磺酰基-5H-吡咯并[2,3-b]吡嗪(100mg,0.28mmol)的1,4-二氧六环(6mL)溶液、xantphos(34mg,0.06mmol)、碳酸钾(60mg,0.43mmol)、醋酸钯(7mg,0.03mmol)和氨基甲酸叔丁酯(50mg,0.43mmol)根据实施例17步骤1的合成方法制备得到87mg红棕色固体产物,产率:78.89%。2-Bromo-5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazine (100 mg, 0.28 mmol) in 1,4-dioxane (6 mL), xantphos (34 mg, 0.06 mmol) ), potassium carbonate (60mg, 0.43mmol), palladium acetate (7mg, 0.03mmol) and tert-butyl carbamate (50mg, 0.43mmol) were prepared according to the synthetic method of Example 17 step 1 to obtain 87mg reddish-brown solid product, the yield : 78.89%.
MS(ESI,pos.ion)m/z:389.3[M+1]+;MS(ESI, pos.ion) m/z: 389.3[M+1] + ;
1H NMR(400MHz,DMSO-d6):δ(ppm)10.11(s,1H),8.77(s,1H),8.18(d,J=4.0Hz,1H),7.97(d,J=8.2Hz,2H),7.43(d,J=8.2Hz,2H),6.85(d,J=4.0Hz,1H),2.34(s,3H),1.37(s,10H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 10.11 (s, 1H), 8.77 (s, 1H), 8.18 (d, J=4.0 Hz, 1H), 7.97 (d, J=8.2 Hz , 2H), 7.43 (d, J=8.2Hz, 2H), 6.85 (d, J=4.0Hz, 1H), 2.34 (s, 3H), 1.37 (s, 10H).
步骤3:化合物2-氨基-5-对甲苯磺酰基-5H-吡咯并[2,3-b]吡嗪的合成Step 3: Synthesis of compound 2-amino-5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazine
(5-对甲苯磺酰基-5H-吡咯并[2,3-b]吡嗪-2-基)氨基甲酸叔丁酯(900mg,2.32mmol)的二氯甲烷(3mL)溶液和氯化氢-异丙醇溶液(6N,7mL,42mmol)按照实施例17步骤2的合成方法制备得到440mg淡黄色干燥固体,产率:65.86%A solution of tert-butyl (5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazin-2-yl)carbamate (900 mg, 2.32 mmol) in dichloromethane (3 mL) and hydrogen chloride-isopropyl Alcohol solution (6N, 7mL, 42mmol) was prepared according to the synthetic method of step 2 of Example 17 to obtain 440mg of light yellow dry solid, yield: 65.86%
MS(ESI,pos.ion)m/z:289.0[M+1]+。MS(ESI, pos.ion) m/z: 289.0[M+1] + .
步骤4:化合物3-对甲苯磺酰基-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪的合成Step 4: Synthesis of Compound 3-p-Tosyl-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine
2-氨基-5-对甲苯磺酰基-5H-吡咯并[2,3-b]吡嗪(170mg,0.5897mmol)的乙醇(6mL)溶液和40%2-氯乙醛(150μL,0.9mmol)按照实施例17步骤3的合成方法制备得到170mg黄绿色,产率:92.29%。2-Amino-5-p-toluenesulfonyl-5H-pyrrolo[2,3-b]pyrazine (170 mg, 0.5897 mmol) in ethanol (6 mL) and 40% 2-chloroacetaldehyde (150 μL, 0.9 mmol) According to the synthetic method of step 3 of Example 17, 170 mg of yellow-green color was obtained, yield: 92.29%.
MS(ESI,pos.ion)m/z:313.2[M+1]+;MS(ESI, pos.ion) m/z: 313.2[M+1] + ;
1H NMR(400MHz,DMSO-d6):δ(ppm)8.90(s,1H),8.58(s,1H),8.04(dd,J=13.5,6.1Hz,4H),7.45(d,J=8.2Hz,2H),7.35(d,J=3.8Hz,1H),2.35(s,3H)。1H NMR (400MHz, DMSO-d 6 ): δ (ppm) 8.90 (s, 1H), 8.58 (s, 1H), 8.04 (dd, J=13.5, 6.1 Hz, 4H), 7.45 (d, J=8.2 Hz, 2H), 7.35 (d, J=3.8 Hz, 1H), 2.35 (s, 3H).
步骤5:化合物8-碘-3-对甲苯磺酰基-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪的合成Step 5: Synthesis of compound 8-iodo-3-p-toluenesulfonyl-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine
向3-对甲苯磺酰基-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪(420mg,1.34mmol)的DMF(8mL)溶液中加入N-碘代琥珀酰亚胺(450mg,2.0mmol),室温搅拌3小时,加入硫代硫酸钠溶液(20mL)淬灭,二氯甲烷萃取(25mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=1/1)得到599mg淡黄色固体,产率:100%。To a solution of 3-p-toluenesulfonyl-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine (420 mg, 1.34 mmol) in DMF (8 mL) was added N-iodosuccinimide (450 mg, 2.0 mmol), stirred at room temperature for 3 hours, quenched by adding sodium thiosulfate solution (20 mL), extracted with dichloromethane (25 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate ( v/v)=1/1) 599 mg of pale yellow solid were obtained, yield: 100%.
MS(ESI,pos.ion)m/z:439.10[M+1]+;MS(ESI, pos.ion) m/z: 439.10[M+1] + ;
1H NMR(400MHz,DMSO-d6):δ(ppm)8.78(d,J=6.7Hz,1H),8.00(m,3H),7.92(s,1H),7.72(d,J=4.1Hz,1H),7.45(d,J=8.3Hz,2H),2.35(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 8.78 (d, J=6.7 Hz, 1H), 8.00 (m, 3H), 7.92 (s, 1H), 7.72 (d, J=4.1 Hz) , 1H), 7.45 (d, J=8.3 Hz, 2H), 2.35 (s, 3H).
步骤6:化合物N-(2-氰基乙基)-3-(3-对甲苯磺酰胺-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-8-基)苯磺酰胺的合成Step 6: Compound N-(2-cyanoethyl)-3-(3-p-toluenesulfonamide-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine-8- Synthesis of phenyl)benzenesulfonamide
8-碘-3-对甲苯磺酰基-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪(300mg,0.68mmol)的1,4-二氧六环(8mL)、 N-(2-氰基乙基)-3-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯磺酰胺(210mg,0.62mmol)、碳酸钾(150mg,1.08mmol)、Pd(dppf)Cl2(50mg,0.07mmol)和水(2mL)根据实施例15步骤7的合成方法得到200mg淡黄色固体,产率:56.11%。8-Iodo-3-p-toluenesulfonyl-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine (300 mg, 0.68 mmol) in 1,4-dioxane (8 mL) ), N-(2-cyanoethyl)-3-(4,4,5,5-tetramethyl-1,3,2-dioxaboropentan-2-yl)benzenesulfonamide (210mg, 0.62 mmol), potassium carbonate (150 mg, 1.08 mmol), Pd(dppf)Cl 2 (50 mg, 0.07 mmol) and water (2 mL) 200 mg of pale yellow solid were obtained according to the synthetic method of Example 15, step 7, yield: 56.11% .
MS(ESI,pos.ion)m/z:521.20[M+1]+。MS(ESI, pos.ion) m/z: 521.20[M+1] + .
步骤7:化合物N-(2-氰乙基)-3-(3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-8-基)苯磺酰胺的合成Step 7: Synthesis of Compound N-(2-cyanoethyl)-3-(3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazin-8-yl)benzenesulfonamide
室温下,向N-(2-氰基乙基)-3-(3-对甲苯磺酰胺-3H-咪唑并[1,2-a]吡咯并[2,3-e]吡嗪-8-基)苯磺酰胺(180mg,0.35mmol)的四氢呋喃(8mL)溶液中加入叔丁醇钾(124mg,1.10mmol),室温搅拌2小时,直接加入硅胶拌样,柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到98mg淡黄色固体,产率:77.36%,纯度:96.08%(HPLC)。At room temperature, to N-(2-cyanoethyl)-3-(3-p-toluenesulfonamide-3H-imidazo[1,2-a]pyrrolo[2,3-e]pyrazine-8- To the solution of tetrahydrofuran (8 mL) of benzenesulfonamide (180 mg, 0.35 mmol), potassium tert-butoxide (124 mg, 1.10 mmol) was added, and the mixture was stirred at room temperature for 2 hours. Ester (v/v)=0/1) gave 98 mg of pale yellow solid, yield: 77.36%, purity: 96.08% (HPLC).
MS(ESI,pos.ion)m/z:367.25[M+1]+;MS(ESI, pos.ion) m/z: 367.25[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.38(s,1H),8.72(s,1H),8.22(t,J=5.8Hz,1H),8.09(d,J=15.1Hz,1H),7.96(m,2H),7.92(s,1H),7.84(t,J=7.8Hz,1H),7.37(t,J=3.1Hz,1H),6.06(dd,J=3.0,2.0Hz,1H),3.09(q,J=6.2Hz,2H),2.66(t,J=6.4Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ (ppm) 12.38 (s, 1H), 8.72 (s, 1H), 8.22 (t, J=5.8Hz, 1H), 8.09 (d, J=15.1Hz ,1H),7.96(m,2H),7.92(s,1H),7.84(t,J=7.8Hz,1H),7.37(t,J=3.1Hz,1H),6.06(dd,J=3.0, 2.0Hz, 1H), 3.09 (q, J=6.2Hz, 2H), 2.66 (t, J=6.4Hz, 2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)141.1,140.3,134.8,134.8,134.6,133.7,130.5,130.4,127.3,126.9,125.2,122.2,119.3,114.8,95.2,39.1,19.1。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 141.1, 140.3, 134.8, 134.8, 134.6, 133.7, 130.5, 130.4, 127.3, 126.9, 125.2, 122.2, 119.3, 114.8, 95.2, 39.1, 19.1.
实施例95Example 95
N-环丁基-2-(3-丙酰胺基苯基)-5H-吡咯[2,3-b]吡嗪-7-甲酰胺N-Cyclobutyl-2-(3-propionamidophenyl)-5H-pyrro[2,3-b]pyrazine-7-carboxamide
步骤1:化合物N-环丁基-2-(3-丙酰胺基苯基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯[2,3-b]吡嗪-7-甲酰胺的合成Step 1: Compound N-Cyclobutyl-2-(3-propionamidophenyl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrole[2,3 -b]Synthesis of pyrazine-7-carboxamide
1,4二氧六环(8mL)/水(2mL),2-溴-N-环丁基-5-(2-(三甲基硅烷基)乙氧基)甲基)-5H-吡咯[2,3-b]吡嗪-7-甲酰胺(0.22g,0.51mmol),N-(3-苯硼酸频哪醇酯苯基)丙酰胺(0.21g,0.76mmol),碳酸钾(0.2g,1.44mmol)和Pd(dppf)Cl2(18mg,0.02mmol)根据实施例1步骤1的合成方法制备得到220mg米白色固体,产率:86.17%。1,4 dioxane (8 mL)/water (2 mL), 2-bromo-N-cyclobutyl-5-(2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrole[ 2,3-b]pyrazine-7-carboxamide (0.22g, 0.51mmol), N-(3-phenylboronic acid pinacol ester phenyl)propionamide (0.21g, 0.76mmol), potassium carbonate (0.2g , 1.44 mmol) and Pd(dppf)Cl 2 (18 mg, 0.02 mmol) were prepared according to the synthesis method of Example 1, step 1 to obtain 220 mg of an off-white solid, yield: 86.17%.
MS(ESI,Pos.ion)m/z:493.3[M+1]+。MS(ESI, Pos.ion) m/z: 493.3[M+1] + .
步骤2:化合物N-环丁基-2-(3-丙酰胺基苯基)-5H-吡咯[2,3-b]吡嗪-7-甲酰胺的合成Step 2: Synthesis of compound N-cyclobutyl-2-(3-propionamidophenyl)-5H-pyrro[2,3-b]pyrazine-7-carboxamide
室温下,将N-环丁基-2-(3-丙酰胺基苯基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯[2,3-b]吡嗪-7-甲酰胺的(0.22g,0.44mmol)溶解于二氯甲烷(20mL)中,加入三氟乙酸(8mL),室温下搅拌6h后。减压浓缩,残渣溶入四氢呋喃(15mL),用饱和碳酸氢钠溶液调至碱性,室温下反应过夜。加水稀释(30mL),二氯甲烷萃取(30mL x 3),有机层用无水硫酸钠干燥,减压浓缩液(二氯甲烷/甲醇(v/v)=30/1),进行柱分离得到黄色固体150mg,产率:92.63%。At room temperature, N-cyclobutyl-2-(3-propionamidophenyl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrole[2,3 -b] Pyrazine-7-carboxamide (0.22 g, 0.44 mmol) was dissolved in dichloromethane (20 mL), trifluoroacetic acid (8 mL) was added, and the mixture was stirred at room temperature for 6 h. Concentrated under reduced pressure, the residue was dissolved in tetrahydrofuran (15 mL), made basic with saturated sodium bicarbonate solution, and reacted overnight at room temperature. Diluted with water (30 mL), extracted with dichloromethane (30 mL x 3), the organic layer was dried over anhydrous sodium sulfate, concentrated under reduced pressure (dichloromethane/methanol (v/v)=30/1), and subjected to column separation to obtain Yellow solid 150 mg, yield: 92.63%.
MS(ESI,pos.ion)m/z:364.2[M+1]+;MS(ESI, pos.ion) m/z: 364.2[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.79(s,1H),10.05(s,1H),8.96(s,1H),8.84(s,1H),8.59(d,J=8.2Hz,1H),8.41(s,1H),7.85(d,J=7.5Hz,1H),7.52–7.43(m,2H),4.56(dd,J=16.5,8.2Hz,1H),2.38(q,J=7.5Hz,2H),2.33(dd,J=16.5,8.0Hz,2H),2.19(dd,J=15.0,5.8Hz,2H),1.83–1.66(m,2H),1.12(t,J=7.5Hz,3H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 12.79(s, 1H), 10.05(s, 1H), 8.96(s, 1H), 8.84(s, 1H), 8.59(d, J= 8.2Hz, 1H), 8.41(s, 1H), 7.85(d, J=7.5Hz, 1H), 7.52–7.43(m, 2H), 4.56(dd, J=16.5, 8.2Hz, 1H), 2.38( q, J=7.5Hz, 2H), 2.33 (dd, J=16.5, 8.0Hz, 2H), 2.19 (dd, J=15.0, 5.8Hz, 2H), 1.83–1.66 (m, 2H), 1.12 (t , J=7.5Hz, 3H);
13C NMR(150MHz,DMSO-d6):δ(ppm)172.6,161.6,146.1,141.6,140.7,137.7,136.1,135.7,135.1,129.8,121.4,120.1,117.7,109.2,43.9,31.5,30.0,15.2,10.0。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 172.6, 161.6, 146.1, 141.6, 140.7, 137.7, 136.1, 135.7, 135.1, 129.8, 121.4, 120.1, 117.7, 109.2, 43.9, 31.5, 30.0, 15.2, 10.0.
实施例96Example 96
5-(4-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1H-吡咯并[2,3-b]吡啶-3-甲酰胺5-(4-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide
步骤1:化合物5-(4-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡嗪-3-甲酰胺的合成Step 1: Compound 5-(4-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1-((2-(trimethylsilyl)ethoxy Synthesis of yl)methyl)-1H-pyrrolo[2,3-b]pyrazine-3-carboxamide
将4-(4-溴苄基)二氧硫代吗啉(16mg,0.05mmol)、N-异丙基-5-硼酸酯-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡啶-3-甲酰胺(20mg,0.04mmol),碳酸钾(15mg,0.11mmol)和Pd(dppf)Cl2(6.4mg,0.01mmol)混合,加入1,4-二氧六环(8mL),氮气氛围下,115℃加热4小时,过滤,硅藻土过滤,滤液浓缩拌样柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到12.7mg黄色油状物,产率:52.4%。4-(4-Bromobenzyl)dioxothiomorpholine (16 mg, 0.05 mmol), N-isopropyl-5-boronate-1-((2-(trimethylsilyl)ethoxy yl)methyl)-1H-pyrrolo[2,3-b]pyridine-3-carboxamide (20 mg, 0.04 mmol), potassium carbonate (15 mg, 0.11 mmol) and Pd(dppf)Cl 2 (6.4 mg, 0.01 mmol) and mixed, 1,4-dioxane (8 mL) was added, heated at 115 °C for 4 hours under nitrogen atmosphere, filtered, filtered through celite, and the filtrate was concentrated by stirring column chromatography (petroleum ether/ethyl acetate (v /v)=2/1) to give 12.7 mg of yellow oil, yield: 52.4%.
MS(ESI,pos.ion)m/z:557.1[M+1]+。MS(ESI, pos.ion) m/z: 557.1[M+1] + .
步骤2:化合物5-(4-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1H-吡咯并[2,3-b]吡啶-3-甲酰胺的合成Step 2: Compound 5-(4-((1,1-Dioxothiomorpholine)methyl)phenyl)-N-isopropyl-1H-pyrrolo[2,3-b]pyridine-3- Synthesis of formamide
将5-(4-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡啶-3-甲酰胺(140mg,0.25mmol)溶于二氯甲烷(6mL)中,加入三氟乙酸(2mL),室温搅拌2h,浓缩溶剂,加入四氢呋喃(6mL)溶解,滴加氢氧化锂溶液(2N)至pH>7,室温搅拌2h,加水(20mL)稀释,二氯甲烷萃取(15mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=1/1)得到63mg白色固体,产率:58.75%。5-(4-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1-((2-(trimethylsilyl)ethoxy)methyl base)-1H-pyrrolo[2,3-b]pyridine-3-carboxamide (140 mg, 0.25 mmol) was dissolved in dichloromethane (6 mL), trifluoroacetic acid (2 mL) was added, stirred at room temperature for 2 h, and the solvent was concentrated , add tetrahydrofuran (6mL) to dissolve, dropwise add lithium hydroxide solution (2N) to pH>7, stir at room temperature for 2h, add water (20mL) to dilute, extract with dichloromethane (15mLx3), dry over anhydrous sodium sulfate, and concentrate for column chromatography (Petroleum ether/ethyl acetate (v/v)=1/1) 63 mg of white solid were obtained, yield: 58.75%.
MS(ESI,pos.ion)m/z:427.0[M+1]+;MS(ESI, pos.ion) m/z: 427.0[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.13(s,1H),8.65(t,J=9.2Hz,1H),8.56(d,J=1.7Hz,1H),8.21(d,J=2.3Hz,1H),7.83(d,J=7.7Hz,1H),7.68(d,J=8.0Hz,2H),7.47(d,J=7.8Hz,2H),4.12(dt,J=13.5,6.7Hz,1H),3.75(d,J=25.8Hz,2H),3.13(s,4H),2.92(s,4H),1.19(d,J=6.5Hz,6H); 1 H NMR (600 MHz, DMSO-d 6 ): δ (ppm) 12.13 (s, 1H), 8.65 (t, J=9.2 Hz, 1H), 8.56 (d, J=1.7 Hz, 1H), 8.21 (d ,J=2.3Hz,1H),7.83(d,J=7.7Hz,1H),7.68(d,J=8.0Hz,2H),7.47(d,J=7.8Hz,2H),4.12(dt,J =13.5,6.7Hz,1H),3.75(d,J=25.8Hz,2H),3.13(s,4H),2.92(s,4H),1.19(d,J=6.5Hz,6H);
13C NMR(150MHz,DMSO-d6):δ(ppm)163.6,148.4,142.7,138.2,137.1,130.0,129.7,129.1,127.6,127.3,119.1,110.4,59.7,50.8,50.6,40.6,23.1。 13 C NMR (150 MHz, DMSO-d 6 ): δ (ppm) 163.6, 148.4, 142.7, 138.2, 137.1, 130.0, 129.7, 129.1, 127.6, 127.3, 119.1, 110.4, 59.7, 50.8, 50.6, 40.6, 23.1.
实施例97Example 97
5-(3-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1H-吡咯并[2,3-b]吡啶-3-甲酰胺5-(3-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1H-pyrrolo[2,3-b]pyridine-3-carboxamide
步骤1:化合物5-(3-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡嗪-3-甲酰胺的合成Step 1: Compound 5-(3-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1-((2-(trimethylsilyl)ethoxy Synthesis of yl)methyl)-1H-pyrrolo[2,3-b]pyrazine-3-carboxamide
4-(3-溴苄基)二氧硫代吗啉(175mg,0.57mmol)、N-异丙基-5-硼酸酯-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡啶-3-甲酰胺(220mg,0.47mmol),碳酸钾(165mg,1.19mmol)、Pd(dppf)Cl2(73mg,0.09mmol)和1,4-二氧六环(10mL)根据实施例20步骤1的合成方法制备得到190mg黄色固体, 产率:71.3%。4-(3-Bromobenzyl)dioxothiomorpholine (175 mg, 0.57 mmol), N-isopropyl-5-boronate-1-((2-(trimethylsilyl)ethoxy) )methyl)-1H-pyrrolo[2,3-b]pyridine-3-carboxamide (220 mg, 0.47 mmol), potassium carbonate (165 mg, 1.19 mmol), Pd(dppf)Cl 2 (73 mg, 0.09 mmol) and 1,4-dioxane (10 mL) were prepared according to the synthetic method of step 1 of Example 20 to obtain 190 mg of yellow solid, yield: 71.3%.
MS(ESI,pos.ion)m/z:557.1[M+1]+。MS(ESI, pos.ion) m/z: 557.1[M+1] + .
步骤2:化合物5-(3-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1H-吡咯并[2,3-b]吡啶-3-甲酰胺的合成Step 2: Compound 5-(3-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1H-pyrrolo[2,3-b]pyridine-3- Synthesis of formamide
5-(3-((1,1-二氧硫代吗啉)甲基)苯基)-N-异丙基-1-((2-(三甲基硅基)乙氧基)甲基)-1H-吡咯并[2,3-b]吡嗪-3-甲酰胺(190mg,0.34mmol)、三氟乙酸(2mL)和氢氧化锂溶液(2N)根据实施例20步骤2的合成方法制备得到71mg白色固体,产率:48.78%。5-(3-((1,1-Dioxythiomorpholine)methyl)phenyl)-N-isopropyl-1-((2-(trimethylsilyl)ethoxy)methyl )-1H-pyrrolo[2,3-b]pyrazine-3-carboxamide (190 mg, 0.34 mmol), trifluoroacetic acid (2 mL) and lithium hydroxide solution (2N) according to the synthesis method of Example 20, Step 2 71 mg of white solid were prepared, yield: 48.78%.
MS(ESI,pos.ion)m/z:427.20[M+1]+;MS(ESI, pos.ion) m/z: 427.20[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.16(s,1H),8.70(s,1H),8.59(s,1H),8.25(s,1H),7.86(d,J=7.5Hz,1H),7.68(s,1H),7.62(d,J=7.4Hz,1H),7.46(t,J=7.5Hz,1H),7.34(d,J=7.3Hz,1H),4.14(dq,J=13.1,6.4Hz,1H),3.76(s,2H),3.16(d,J=24.8Hz,4H),2.93(s,4H),1.19(d,J=6.4Hz,6H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 12.16(s, 1H), 8.70(s, 1H), 8.59(s, 1H), 8.25(s, 1H), 7.86(d, J= 7.5Hz, 1H), 7.68(s, 1H), 7.62(d, J=7.4Hz, 1H), 7.46(t, J=7.5Hz, 1H), 7.34(d, J=7.3Hz, 1H), 4.14 (dq,J=13.1,6.4Hz,1H),3.76(s,2H),3.16(d,J=24.8Hz,4H),2.93(s,4H),1.19(d,J=6.4Hz,6H) ;
13C NMR(150MHz,DMSO-d6):δ(ppm)163.6,148.4,142.9,139.3,139.0,129.8,129.5,129.1,128.1,127.8,127.6,126.3,119.1,110.3,60.0,50.7,50.6,40.6,23.1。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 163.6, 148.4, 142.9, 139.3, 139.0, 129.8, 129.5, 129.1, 128.1, 127.8, 127.6, 126.3, 119.1, 110.3, 60.0, 50.7, 50.6, 40.6, 23.1.
实施例98Example 98
N-(3-(3-(1H-吲哚-5-基)-1H-吡咯并[2,3-b]吡啶-5-基)苯基)-3-甲氧基丙酰胺N-(3-(3-(1H-Indol-5-yl)-1H-pyrrolo[2,3-b]pyridin-5-yl)phenyl)-3-methoxypropionamide
步骤1:化合物5-溴-3-(1H-吲哚-5-基)-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶的合成Step 1: Synthesis of compound 5-bromo-3-(1H-indol-5-yl)-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridine
将5-溴-3-碘-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶(20mg,0.04mmol)溶于乙腈(4mL)中,依次加入5-硼酸频哪醇酯-1H-吲哚(11.2mg,0.04mmol)、1N碳酸钠水溶液(0.11mL,0.11mmol)、Pd(dppf)Cl2(6.5mg,0.01mmol),换N2,室温搅拌2h,加入饱和食盐水(10mL),二氯甲烷(10mL x 3)萃取,用无水硫酸钠干燥,除去溶剂,浓缩液进行柱分离(石油醚/乙酸乙酯(v/v)=6/1),得到7mg棕色固体,产率:35.8%。5-Bromo-3-iodo-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridine (20 mg, 0.04 mmol) was dissolved in acetonitrile (4 mL), followed by the addition of 5-boronic acid pinacol Ester-1H-indole (11.2 mg, 0.04 mmol), 1N aqueous sodium carbonate solution (0.11 mL, 0.11 mmol), Pd(dppf)Cl 2 (6.5 mg, 0.01 mmol), changed to N 2 , stirred at room temperature for 2 h, added saturated Brine (10 mL), extracted with dichloromethane (10 mL x 3), dried with anhydrous sodium sulfate, the solvent was removed, and the concentrated solution was subjected to column separation (petroleum ether/ethyl acetate (v/v)=6/1) to obtain 7 mg of brown solid, yield: 35.8%.
MS(ESI,pos.ion)m/z:466.0[M+1]。MS (ESI, pos.ion) m/z: 466.0 [M+1].
步骤2:化合物N-(3-(3-(1H-吲哚-5-基)-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡嗪-5-基)苯基)丙烯酰胺的合成Step 2: Compound N-(3-(3-(1H-Indol-5-yl)-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyrazin-5-yl)phenyl ) Synthesis of acrylamide
将5-溴-3-(1H-吲哚-5-基)-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡啶(10mg,0.02mmol),N-(3-苯硼酸频哪醇酯苯基)丙烯酰胺(8.8mg,0.032mmol),Pd(dppf)Cl2(3.1mg,0.004mmol)和1mol/L的碳酸钠溶液(0.052mL,0.052mmol)的化合物置于单口瓶中,加入二氧六环(4mL),115℃室温搅拌5小时,加入饱和食盐水(10mL),二氯甲烷(10mL x 3)萃取,用无水硫酸钠干燥,除去溶剂,浓缩液进行柱分离(石油醚/乙酸乙酯(v/v)=2/1)得到3mg棕色固体,产率:26.27%。5-Bromo-3-(1H-indol-5-yl)-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyridine (10 mg, 0.02 mmol), N-(3-benzene The compound of pinacol borate phenyl)acrylamide (8.8mg, 0.032mmol), Pd(dppf)Cl 2 (3.1mg, 0.004mmol) and 1mol/L sodium carbonate solution (0.052mL, 0.052mmol) was placed in In a single-necked flask, add dioxane (4 mL), stir at room temperature at 115°C for 5 hours, add saturated brine (10 mL), extract with dichloromethane (10 mL x 3), dry with anhydrous sodium sulfate, remove the solvent, and concentrate the solution Column separation (petroleum ether/ethyl acetate (v/v)=2/1) was performed to obtain 3 mg of a brown solid, yield: 26.27%.
MS(ESI,pos.ion)m/z:533.2[M+1]+。MS(ESI, pos.ion) m/z: 533.2[M+1] + .
步骤3:化合物N-(3-(3-(1H-吲哚-5-基)-1H-吡咯并[2,3-b]吡啶-5-基)苯基)-3-甲氧基丙酰胺的合成Step 3: Compound N-(3-(3-(1H-Indol-5-yl)-1H-pyrrolo[2,3-b]pyridin-5-yl)phenyl)-3-methoxyprop amide synthesis
将N-(3-(3-(1H-吲哚-5-基)-1-对甲苯磺酰基-1H-吡咯并[2,3-b]吡嗪-5-基)苯基)丙烯酰胺(7mg,0.01mmol)溶于甲醇(3mL)中,加入5N氢氧化钠溶液(13.1μL,0.06mmol)溶液,50℃加热反应3小时,加水稀释(10mL),二氯甲烷萃取(15mL x 3),用无水硫酸钠干燥,除去溶剂,浓缩液进行柱分离,(石油醚/乙酸乙酯(v/v)=2/1)得到2mg淡黄色固体,产率:37.07%。N-(3-(3-(1H-Indol-5-yl)-1-p-toluenesulfonyl-1H-pyrrolo[2,3-b]pyrazin-5-yl)phenyl)acrylamide (7 mg, 0.01 mmol) was dissolved in methanol (3 mL), 5N sodium hydroxide solution (13.1 μL, 0.06 mmol) was added, the reaction was heated at 50°C for 3 hours, diluted with water (10 mL), and extracted with dichloromethane (15 mL x 3 ), dried over anhydrous sodium sulfate, the solvent was removed, and the concentrated solution was subjected to column separation (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 2 mg of pale yellow solid, yield: 37.07%.
MS(ESI,pos.ion)m/z:411.15[M+1]+;MS(ESI, pos.ion) m/z: 411.15[M+1] + ;
1H NMR(600MHz,CD3OD):δ(ppm)8.49(s,1H),8.48(s,1H),7.91(s,1H),7.87(s,1H),7.65–7.58(m,2H),7.47(m,4H),7.28(d,J=3.0Hz,1H),6.53(d,J=2.9Hz,1H),3.81–3.70(m,3H),3.39(s,3H),2.66(t,J=6.1Hz,2H);1H NMR (600MHz, CD3OD ): δ(ppm) 8.49(s,1H), 8.48(s,1H), 7.91(s,1H), 7.87(s,1H), 7.65–7.58(m,2H) ,7.47(m,4H),7.28(d,J=3.0Hz,1H),6.53(d,J=2.9Hz,1H),3.81–3.70(m,3H),3.39(s,3H),2.66( t, J=6.1Hz, 2H);
13C NMR(150MHz,CD3OD):δ(ppm)171.0,148.0,140.9,140.0,139.1,135.3,129.1,129.1,128.6,126.4,125.5,124.7,122.7,122.6,121.0,119.2,118.4,118.4,118.2,117.9,111.2,101.1,68.2,57.5,37.0。 13 C NMR (150MHz, CD 3 OD): δ(ppm) 171.0, 148.0, 140.9, 140.0, 139.1, 135.3, 129.1, 129.1, 128.6, 126.4, 125.5, 124.7, 122.7, 122.6, 121.0, 119.2, 118.4, 1 , 118.2, 117.9, 111.2, 101.1, 68.2, 57.5, 37.0.
实施例99Example 99
3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-羰基)苯基)氨基)丙腈3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine-7-carbonyl)phenyl)amino)propionitrile
步骤1:化合物(2-溴-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲醇的合成Step 1: Synthesis of compound (2-bromo-5H-pyrrolo[2,3-b]pyrazin-7-yl)(4-nitrophenyl)methanol
室温下,向2-溴-5H-吡咯并[2,3-b]吡嗪(3g,15.15mmol)和对硝基苯甲醛(5.4g,36mmol)的混合物中加入甲醇(25mL),室温搅拌10分钟后加入加入氢氧化钾(5.4g,96mmol),继续室温搅拌48小时,停止反应,加水(50mL)稀释,二氯甲烷萃取(50mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到1.35g淡黄色固体,产率:25.3%。At room temperature, methanol (25 mL) was added to a mixture of 2-bromo-5H-pyrrolo[2,3-b]pyrazine (3 g, 15.15 mmol) and p-nitrobenzaldehyde (5.4 g, 36 mmol), and the mixture was stirred at room temperature After 10 minutes, potassium hydroxide (5.4 g, 96 mmol) was added, and stirring was continued for 48 hours at room temperature. The reaction was stopped, diluted with water (50 mL), extracted with dichloromethane (50 mL×3), dried over anhydrous sodium sulfate, and concentrated by column chromatography (petroleum Ether/ethyl acetate (v/v)=2/1) gave 1.35 g of pale yellow solid, yield: 25.3%.
MS(ESI,pos.ion)m/z:348.8,350.8[M+1]+。MS(ESI, pos.ion) m/z: 348.8, 350.8[M+1] + .
步骤2:化合物(2-溴-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲酮的合成Step 2: Synthesis of compound (2-bromo-5H-pyrrolo[2,3-b]pyrazin-7-yl)(4-nitrophenyl)methanone
向(2-溴-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲醇(210mg,0.60mmol)的四氢呋喃(7mL)溶液中,加入DESS-MARTIN氧化剂(530mg,1.23mmol),室温搅拌4小时,加水(15mL)稀释,二氯甲烷萃取(20mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到170mg淡黄色固体,产率:81.42%。To a solution of (2-bromo-5H-pyrrolo[2,3-b]pyrazin-7-yl)(4-nitrophenyl)methanol (210 mg, 0.60 mmol) in tetrahydrofuran (7 mL) was added DESS- MARTIN oxidant (530 mg, 1.23 mmol), stirred at room temperature for 4 hours, diluted with water (15 mL), extracted with dichloromethane (20 mL×3), dried over anhydrous sodium sulfate, concentrated by column chromatography (petroleum ether/ethyl acetate (v/v) =2/1) 170 mg of pale yellow solid were obtained, yield: 81.42%.
MS(ESI,pos.ion)m/z:347.05,349.10[M+1]+。MS (ESI, pos.ion) m/z: 347.05, 349.10 [M+1] + .
步骤3:化合物(2-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲酮的合成Step 3: Compound (2-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)(4 Synthesis of -nitrophenyl)methanone
向(2-溴-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲酮(170mg,0.49mmol)的DMF(8mL)溶液中加入氢化钠(60%,40mg,1.0mmol),室温搅拌30分钟,加入SEMCl(120μL,0.67mmol),继续室温搅拌4小时,加水(30mL)淬灭,二氯甲烷萃取(30mLx3),无水硫酸钠干燥,浓缩后,柱层析(石油醚/乙酸乙酯(v/v)=3/1),过滤,干燥得到180mg黄色固体,产率:76.98%。To a solution of (2-bromo-5H-pyrrolo[2,3-b]pyrazin-7-yl)(4-nitrophenyl)methanone (170 mg, 0.49 mmol) in DMF (8 mL) was added sodium hydride (60%, 40 mg, 1.0 mmol), stirred at room temperature for 30 minutes, added SEMCl (120 μL, 0.67 mmol), continued to stir at room temperature for 4 hours, quenched by adding water (30 mL), extracted with dichloromethane (30 mL×3), and dried over anhydrous sodium sulfate , after concentration, column chromatography (petroleum ether/ethyl acetate (v/v)=3/1), filtration and drying to obtain 180 mg of yellow solid, yield: 76.98%.
MS(ESI,pos.ion)m/z:477.75.479.75[M+1]+。MS(ESI, pos.ion) m/z: 477.75.479.75[M+1] + .
步骤4:化合物(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲酮的合成Step 4: Compound (2-(1-Methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3 Synthesis of -b]pyrazin-7-yl)(4-nitrophenyl)methanone
向(2-溴-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲酮(180mg,0.37mmol)的1,4-二氧六环(6mL)溶液中,依次加入碳酸钾(80mg,0.57mmol),1-甲基吡唑硼酸脂(106mg,0.50mmol)和Pd(dppf)Cl2(30mg,0.04mmol),水(1.5mL),氮气保护,115℃回流加热反应5.5小时,加水(40mL)稀释,二氯甲烷萃取(55mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到120mg黄色固体,产率:66.50%。To (2-bromo-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)(4-nitro Phenyl)methanone (180mg, 0.37mmol) in 1,4-dioxane (6mL) solution, potassium carbonate (80mg, 0.57mmol), 1-methylpyrazole borate (106mg, 0.50mmol) were added successively ) and Pd(dppf)Cl 2 (30mg, 0.04mmol), water (1.5mL), nitrogen protection, heated at 115°C under reflux for 5.5 hours, diluted with water (40mL), extracted with dichloromethane (55mLx3), anhydrous sodium sulfate It was dried and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=2/1) to obtain 120 mg of a yellow solid, yield: 66.50%.
MS(ESI,pos.ion)m/z:479.30[M+1]+。MS(ESI, pos.ion) m/z: 479.30[M+1] + .
步骤5:化合物(4-氨基苯基)(2-(1-甲基-1H-吡咯并-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)甲酮的合成Step 5: Compound (4-aminophenyl)(2-(1-methyl-1H-pyrrolo-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl) Synthesis of -5H-pyrrolo[2,3-b]pyrazin-7-yl)methanone
向(2-(1-甲基-1H-吡唑-4-基)-5-((2-(三甲硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)(4-硝基苯基)甲酮(120mg,0.25mmol)的甲醇(6mL)溶液中加入钯碳(10%,10mg,0.01mmol),氢气置换,室温搅拌1.5小时,过滤,浓缩柱层析(石油醚/乙酸乙酯(v/v)=0/1)得到44mg淡黄色油状物,产率:39.12%。To (2-(1-methyl-1H-pyrazol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo[2,3-b] Palladium on carbon (10%, 10 mg, 0.01 mmol) was added to a solution of pyrazin-7-yl)(4-nitrophenyl)methanone (120 mg, 0.25 mmol) in methanol (6 mL), replaced with hydrogen, and stirred at room temperature for 1.5 hours , filtered, and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v)=0/1) to obtain 44 mg of pale yellow oil, yield: 39.12%.
MS(ESI,pos.ion)m/z:448.90[M+1]+。MS(ESI, pos.ion) m/z: 448.90[M+1] + .
步骤6:化合物3-((4-(2-(1-甲基-1H-吡咯-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-羰基)苯基)氨基)丙腈的合成Step 6: Compound 3-((4-(2-(1-methyl-1H-pyrrol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H - Synthesis of pyrrolo[2,3-b]pyrazine-7-carbonyl)phenyl)amino)propionitrile
向(4-氨基苯基)(2-(1-甲基-1H-吡咯并-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-基)甲酮(100mg,0.22mmol)的丙烯腈(10mL)溶液中,加入三氯化铝(45mg,0.33mmol),85℃回流反应24小时,加入少量甲醇(5mL)淬灭反应,过滤,滤液加水(40mL)稀释,二氯甲烷萃取(55mLx3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=2/1)得到70mg黄色油状物,产率:70.64%。To (4-aminophenyl)(2-(1-methyl-1H-pyrrolo-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H- To a solution of pyrrolo[2,3-b]pyrazin-7-yl)methanone (100 mg, 0.22 mmol) in acrylonitrile (10 mL), was added aluminum trichloride (45 mg, 0.33 mmol), and the reaction was carried out at 85 °C for 24 After 1 hour, a small amount of methanol (5 mL) was added to quench the reaction, filtered, the filtrate was diluted with water (40 mL), extracted with dichloromethane (55 mL×3), dried over anhydrous sodium sulfate, and concentrated by column chromatography (petroleum ether/ethyl acetate (v/v) )=2/1) to give 70 mg of yellow oil, yield: 70.64%.
MS(ESI,pos.ion)m/z:502.3[M+1]+。MS(ESI, pos.ion) m/z: 502.3[M+1] + .
步骤7:化合物3-((4-(2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-羰基)苯基)氨基)丙腈的合成Step 7: Compound 3-((4-(2-(1-Methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine-7-carbonyl)phenyl) Synthesis of Amino)Propionitrile
向3-((4-(2-(1-甲基-1H-吡咯-4-基)-5-((2-(三甲基硅基)乙氧基)甲基)-5H-吡咯并[2,3-b]吡嗪-7-羰基)苯基)氨基)丙腈(79mg,0.15mmol)的二氯甲烷(6mL)溶液中,加入三氟乙酸(2mL)室温搅拌过夜,浓缩溶剂,加入四氢呋喃(6mL)溶解,加乙二胺中和至pH>7,室温搅拌3小时,加水(20mL)稀释,二氯甲烷萃取(20mL x 3),无水硫酸钠干燥,浓缩柱层析(石油醚/乙酸乙酯(v/v)=0/1)后通过制备板分离得到7mg黄色固体,产率:11.97%。To 3-((4-(2-(1-methyl-1H-pyrrol-4-yl)-5-((2-(trimethylsilyl)ethoxy)methyl)-5H-pyrrolo [2,3-b]pyrazine-7-carbonyl)phenyl)amino)propionitrile (79 mg, 0.15 mmol) in dichloromethane (6 mL) was added trifluoroacetic acid (2 mL), stirred at room temperature overnight, and the solvent was concentrated , add tetrahydrofuran (6mL) to dissolve, add ethylenediamine to neutralize to pH>7, stir at room temperature for 3 hours, add water (20mL) to dilute, extract with dichloromethane (20mL x 3), dry over anhydrous sodium sulfate, and concentrate for column chromatography (Petroleum ether/ethyl acetate (v/v) = 0/1), 7 mg of yellow solid was obtained by preparative plate isolation, yield: 11.97%.
MS(ESI,pos.ion)m/z:372.25[M+1]+;MS(ESI, pos.ion) m/z: 372.25[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)12.75(s,1H),8.69(s,1H),8.31(s,1H),8.26(s,1H),7.99(s,1H),7.88(d,J=8.7Hz,2H),6.88(t,J=6.0Hz,1H),6.74(d,J=8.7Hz,2H),3.90(s,3H),3.48(dd,J=12.6,6.3Hz,2H),2.79(t,J=6.5Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 12.75(s, 1H), 8.69(s, 1H), 8.31(s, 1H), 8.26(s, 1H), 7.99(s, 1H) ,7.88(d,J=8.7Hz,2H),6.88(t,J=6.0Hz,1H),6.74(d,J=8.7Hz,2H),3.90(s,3H),3.48(dd,J= 12.6,6.3Hz,2H),2.79(t,J=6.5Hz,2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)207.0,152.1,143.1,140.4,137.3,136.8,136.0,135.3,132.7,129.5,127.1,121.5,120.0,115.2,111.2,45.8,40.5,17.8。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 207.0, 152.1, 143.1, 140.4, 137.3, 136.8, 136.0, 135.3, 132.7, 129.5, 127.1, 121.5, 120.0, 115.2, 111.2, 45.8, 40.5, 17.8.
实施例100Example 100
3-((4-(5-甲基-2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈3-((4-(5-Methyl-2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl )amino)propionitrile
步骤1:化合物2-溴-7-碘-5-甲基-5H-吡咯并[2,3-b]吡嗪的合成Step 1: Synthesis of compound 2-bromo-7-iodo-5-methyl-5H-pyrrolo[2,3-b]pyrazine
0℃下,将氢化钠(550mg,13.75mmol)分批加入到2-溴-7-碘-5H-吡咯并[2,3-b]吡嗪(3.00g,8.37mmol)的N,N-二甲基甲酰胺(20mL)混合液中。室温下搅拌1.5h后,再在0℃下缓慢滴加碘甲烷(2.2g,16.00mmol),滴加完毕后,室温下搅拌反应5h。加水(50mL)淬灭反应,乙酸乙酯(50mL x 3)萃取,有机层用饱和食盐水(50mL)洗涤,无水硫酸钠干燥,浓缩除去溶剂,残留物进行柱层析分离(石油醚/乙酸乙酯(v/v)=3/1)得到2.00g淡黄色固体,产率:64.2%。At 0°C, sodium hydride (550 mg, 13.75 mmol) was added portionwise to a solution of 2-bromo-7-iodo-5H-pyrrolo[2,3-b]pyrazine (3.00 g, 8.37 mmol) in N,N- dimethylformamide (20 mL) mixture. After stirring at room temperature for 1.5 h, methyl iodide (2.2 g, 16.00 mmol) was slowly added dropwise at 0°C. After the dropwise addition, the reaction was stirred at room temperature for 5 h. The reaction was quenched by adding water (50 mL), extracted with ethyl acetate (50 mL x 3), the organic layer was washed with saturated brine (50 mL), dried over anhydrous sodium sulfate, concentrated to remove the solvent, and the residue was separated by column chromatography (petroleum ether/ Ethyl acetate (v/v)=3/1) gave 2.00 g of a pale yellow solid, yield: 64.2%.
MS(ESI,pos.ion)m/z:337.7[M+1]+;MS(ESI, pos.ion) m/z: 337.7[M+1] + ;
1H NMR(400MHz,DMSO-d6):δ(ppm)8.52–8.30(m,1H),8.18(d,J=1.8Hz,1H),3.85(s,3H)。 1 H NMR (400 MHz, DMSO-d 6 ): δ (ppm) 8.52-8.30 (m, 1H), 8.18 (d, J=1.8 Hz, 1H), 3.85 (s, 3H).
步骤2:化合物3-((4-(2-溴-5-甲基-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 2: Synthesis of compound 3-((4-(2-bromo-5-methyl-5H-pyrrolo[2,3-b]pyrazin-7-yl)phenyl)amino)propionitrile
乙腈(12mL)/水(4mL)、2-溴-7-碘-5-甲基-5H-吡咯并[2,3-b]吡嗪(350mg,1.04mmol)、3-((4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)苯基)氨基)丙腈(350mg,1.03mmol)、碳酸钠(330mg,3.11mmol)和二(三苯基膦)二氯化钯(Pd(PPh3)2Cl2)(40mg,0.05mmol)的混合物按照实施例1步骤1的合成方法制备得到80mg黄色固体,产率:21.7%。Acetonitrile (12 mL)/water (4 mL), 2-bromo-7-iodo-5-methyl-5H-pyrrolo[2,3-b]pyrazine (350 mg, 1.04 mmol), 3-((4-( 4,4,5,5-Tetramethyl-1,3,2-dioxaboropentan-2-yl)phenyl)amino)propionitrile (350 mg, 1.03 mmol), sodium carbonate (330 mg, 3.11 mmol) A mixture of bis(triphenylphosphine) palladium dichloride (Pd(PPh 3 ) 2 Cl 2 ) (40 mg, 0.05 mmol) was prepared according to the synthetic method of Example 1, step 1 to obtain 80 mg of a yellow solid, yield: 21.7% .
MS(ESI,pos.ion)m/z:356.2[M+1]+。MS(ESI, pos.ion) m/z: 356.2[M+1] + .
步骤3:化合物3-((4-(5-甲基-2-(1-甲基-1H-吡唑-4-基)-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈的合成Step 3: Compound 3-((4-(5-Methyl-2-(1-methyl-1H-pyrazol-4-yl)-5H-pyrrolo[2,3-b]pyrazine-7- Synthesis of base)phenyl)amino)propionitrile
氮气保护下,将1,4-二氧六环(4mL)/水(1mL)加入到3-((4-(2-溴-5-甲基-5H-吡咯并[2,3-b]吡嗪-7-基)苯基)氨基)丙腈(75mg,0.18mmol)、1-甲基-4-(4,4,5,5-四甲基-1,3,2-二氧硼戊烷-2-基)-1H-吡唑(65mg,0.31mmol)、碳酸钾(45mg,0.33mmol)和1,1'-双二苯基膦二茂铁二氯化钯(Pd(dppf)Cl2)(8mg,0.06mmol)混合物中,110℃回流反应过夜,反应液冷却至室温,硅藻土过滤,滤液减压浓缩,进行柱分离(石油醚/乙酸乙酯(v/v)=0/1)得到52mg黄色固体,产率:81.3%。Under nitrogen, 1,4-dioxane (4 mL)/water (1 mL) was added to 3-((4-(2-bromo-5-methyl-5H-pyrrolo[2,3-b]) Pyrazin-7-yl)phenyl)amino)propionitrile (75 mg, 0.18 mmol), 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxoboron) Pentan-2-yl)-1H-pyrazole (65 mg, 0.31 mmol), potassium carbonate (45 mg, 0.33 mmol) and 1,1'-bisdiphenylphosphinoferrocene palladium dichloride (Pd(dppf) Cl 2 ) (8 mg, 0.06 mmol), refluxed at 110° C. overnight for reaction, the reaction solution was cooled to room temperature, filtered through celite, the filtrate was concentrated under reduced pressure, and subjected to column separation (petroleum ether/ethyl acetate (v/v)= 0/1) gave 52 mg of a yellow solid, yield: 81.3%.
MS(ESI,pos.ion)m/z:358.0[M+1]+;MS(ESI, pos.ion) m/z: 358.0[M+1] + ;
1H NMR(600MHz,DMSO-d6):δ(ppm)8.64(s,1H),8.38(s,1H),8.14(s,1H),8.11(s,1H),8.00(d,J=8.6Hz,2H),6.73(d,J=8.7Hz,2H),3.94(s,3H),3.85(s,3H),3.82(s,1H),3.40(t,J=6.4Hz,2H),2.76(t,J=6.5Hz,2H); 1 H NMR (600MHz, DMSO-d 6 ): δ(ppm) 8.64(s, 1H), 8.38(s, 1H), 8.14(s, 1H), 8.11(s, 1H), 8.00(d, J= 8.6Hz, 2H), 6.73(d, J=8.7Hz, 2H), 3.94(s, 3H), 3.85(s, 3H), 3.82(s, 1H), 3.40(t, J=6.4Hz, 2H) ,2.76(t,J=6.5Hz,2H);
13C NMR(150MHz,DMSO-d6):δ(ppm)146.4,141.5,140.0,137.1,136.3,136.2,134.0,129.8,129.4,127.1,122.5,121.7,120.2,113.1,39.5,39.2,31.4,17.9。 13 C NMR (150MHz, DMSO-d 6 ): δ(ppm) 146.4, 141.5, 140.0, 137.1, 136.3, 136.2, 134.0, 129.8, 129.4, 127.1, 122.5, 121.7, 120.2, 113.1, 39.5, 39.2, 31.4, 17.9.
生物学活性biological activity
生物实施例1 JAK1/2/3体外活性测试方法Biological Example 1 JAK1/2/3 in vitro activity test method
本发明采用以下方法对所示的化合物进行生物试验:The present invention adopts the following methods to carry out biological tests on the compounds shown:
1.采用Caliper Mobility Shift Assay检测化合物对JAK1/2/3酶抑制作用。1. Caliper Mobility Shift Assay was used to detect the inhibitory effect of compounds on JAK1/2/3 enzymes.
2.配制1倍激酶反应液:JAK2/3:50mM HEPES,pH 7.5;0.0015%Brij-35;10mMMgCl2;2mM DTT。JAK1:25mM HEPES,pH 7.5;0.001%Brij-35;0.01%Triton;0.5mM EGTA;10mM MgCl2。2. Prepare 1x kinase reaction solution: JAK2/3: 50 mM HEPES, pH 7.5; 0.0015% Brij-35; 10 mM MgCl 2 ; 2 mM DTT. JAK1: 25 mM HEPES, pH 7.5; 0.001% Brij-35; 0.01% Triton; 0.5 mM EGTA; 10 mM MgCl2 .
3.配制反应终止液:100mM HEPES,pH 7.5;0.0015%Brij-35;0.2%CoatingReagent#3(Caliper,货号760050);50mM EDTA。3. Prepare reaction stop solution: 100 mM HEPES, pH 7.5; 0.0015% Brij-35; 0.2% Coating Reagent #3 (Caliper, Cat. No. 760050); 50 mM EDTA.
4.酶配制(JAK1/2/3):用1倍激酶反应液配制酶溶液,酶配制终浓度为JAK1(30nM),JAK2(2nM),JAK 3(4nM)。4. Enzyme preparation (JAK1/2/3): prepare enzyme solution with 1-fold kinase reaction solution, and the final concentration of enzyme preparation is JAK1 (30nM), JAK2 (2nM), JAK3 (4nM).
5.底物配制:用1倍激酶反应液配制底物溶液,底物配制终浓度见表1。5. Substrate preparation: prepare the substrate solution with 1-fold kinase reaction solution. The final concentration of the substrate preparation is shown in Table 1.
表1底物配制终浓度Table 1 Substrate preparation final concentration
根据实验方法优化结果,实验采用384孔板(Corning,Cat.No.3573,Lot.No.12608008)进行检测,将JAK1/2/3酶浓度配制为JAK1(75nM),JAK2(5nM),JAK 3(10nM),反应终浓度为JAK1(30nM),JAK2(2nM),JAK 3(4nM);底物Peptide FAM-P22浓度配制为7.5μM,反应终浓度为3μM;ATP配制浓度为JAK1(225μM),JAK2(50μM),JAK3(15.5μM),反应终浓度为JAK1(90μM),JAK2(20 μM),JAK3(6.2μM);Peptide D(序列5-FAM-C6-KKHTDDGYMPMSPGVA-NH2)浓度配制为7.5μM,反应终浓度为3μM;酶及底物均使用1倍激酶反应液配制。反应体系如表2所示。According to the optimization results of the experimental method, the experiment used 384-well plate (Corning, Cat.No.3573, Lot.No.12608008) for detection, and the concentration of JAK1/2/3 enzymes was prepared as JAK1 (75nM), JAK2 (5nM), JAK 3 (10nM), the final reaction concentration was JAK1 (30nM), JAK2 (2nM), JAK 3 (4nM); the substrate Peptide FAM-P22 was prepared at a concentration of 7.5μM, and the final reaction concentration was 3μM; ATP was prepared at a concentration of JAK1 (225μM) ), JAK2 (50μM), JAK3 (15.5μM), the final reaction concentration is JAK1 (90μM), JAK2 (20 μM), JAK3 (6.2μM); Peptide D (sequence 5-FAM-C6-KKHTDDGYMPMSPGVA-NH2) concentration preparation 7.5 μM, the final reaction concentration is 3 μM; both enzyme and substrate are prepared with 1-fold kinase reaction solution. The reaction system is shown in Table 2.
表2化合物对JAK1/2/3酶IC50检测体系Table 2 IC 50 detection system for JAK1/2/3 enzymes
采用384孔板进行检测,实验设置受试样品孔、阳性对照孔、阴性对照孔,每个样品利用双复孔检测8个浓度下化合物对JAK1/2/3酶的抑制作用,利用酶及底物反应孔作为阳性对照,无酶孔(激酶反应液)作为阴性对照。各孔按表2顺序加入相应样品、缓冲液及酶后,25℃(RT)恒温箱孵育10min,然后每孔加入已配置好的Peptide solution,并于28℃恒温孵育60min,加入反应终止液后,利用Caliper EZ Reader在FP485nM激发/525nM发射波长处进行检测,读取数据为转化率。利用Graph Pad Prism 5软件对化合物不同浓度下对JAK1/2/3酶抑制作用进行作图,计算IC50,实验结果见表3。A 384-well plate was used for detection. The test sample wells, positive control wells and negative control wells were set up in the experiment. Each sample was tested by double wells to detect the inhibitory effect of compounds on JAK1/2/3 enzymes at 8 concentrations. Substrate reaction wells were used as positive controls, and no enzyme wells (kinase reaction solution) were used as negative controls. After adding the corresponding samples, buffers and enzymes to each well in the order of Table 2, incubate at 25°C (RT) for 10 minutes, then add the prepared Peptide solution to each well, and incubate at 28°C for 60 minutes. After adding the reaction stop solution , detected at FP485nM excitation/525nM emission wavelength using Caliper EZ Reader, and read the data as conversion rate. The Graph Pad Prism 5 software was used to plot the inhibitory effects of compounds on JAK1/2/3 enzymes at different concentrations, and IC 50 was calculated. The experimental results are shown in Table 3.
表3化合物的酶(JAK1/2/3)抑制数据Table 3 Enzyme (JAK1/2/3) Inhibition Data for Compounds
表3数据显示,本发明部分化合物对JAK1、JAK2和JAK3均有一定抑制作用,特别是对JAK3有较强的抑制作用,能有效的用于多种适应症的治疗。The data in Table 3 shows that some of the compounds of the present invention have certain inhibitory effects on JAK1, JAK2 and JAK3, and especially have a strong inhibitory effect on JAK3, which can be effectively used for the treatment of various indications.
生物实施例2 测试化合物大鼠体内PK研究Biological Example 2 In vivo PK study of test compounds in rats
SD大鼠经口灌胃给予5mg/kg或经尾静脉注射1mg/kg的测试化合物。给药后按时间点(0.083小时,0.25小时,0.5小时,1小时,2小时,4小时,6小时,8小时和24小时)眼眶静脉采血,收集于加K2EDTA的抗凝管内。血浆样品经液液萃取后,在三重四极杆串联质谱仪上,以多重反应离子监测(MRM)方式进行定量分析。采用WinNonlin 6.1软件用非房室模型法计算药动学参数。SD rats were orally administered 5 mg/kg or 1 mg/kg of the test compound via tail vein injection. Orbital vein blood was collected at time points (0.083 hours, 0.25 hours, 0.5 hours, 1 hour, 2 hours, 4 hours, 6 hours, 8 hours and 24 hours) after administration, and collected in anticoagulant tubes with K 2 EDTA. After liquid-liquid extraction, the plasma samples were quantitatively analyzed on a triple quadrupole tandem mass spectrometer with multiple reactive ion monitoring (MRM). Pharmacokinetic parameters were calculated by non-compartmental model method using WinNonlin 6.1 software.
结论:本发明化合物清除率中等,在SD大鼠口服给药5mg/kg后,表现出较高的血药暴露量,同时半衰期合理,具备比较好的成药性。Conclusion: The compound of the present invention has a moderate clearance rate. After oral administration of 5 mg/kg in SD rats, it shows a higher blood drug exposure, and at the same time, the half-life is reasonable, and it has a relatively good druggability.
Claims (3)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201510402418 | 2015-07-09 | ||
| CN201510402418X | 2015-07-09 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN106336413A CN106336413A (en) | 2017-01-18 |
| CN106336413B true CN106336413B (en) | 2021-04-20 |
Family
ID=57825336
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201610536665.3A Active CN106336413B (en) | 2015-07-09 | 2016-07-08 | Compounds as JAK inhibitors and their uses |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN106336413B (en) |
Families Citing this family (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| NZ702485A (en) | 2010-06-03 | 2016-04-29 | Pharmacyclics Llc | The use of inhibitors of bruton’s tyrosine kinase (btk) |
| MX2015001081A (en) | 2012-07-24 | 2015-10-14 | Pharmacyclics Inc | Mutations associated with resistance to inhibitors of bruton's tyrosine kinase (btk). |
| CN109422753B (en) * | 2017-09-03 | 2021-12-31 | 上海美志医药科技有限公司 | Compounds capable of inhibiting and degrading tyrosine protein kinase JAK1 or JAK2 activity |
| CN110105363A (en) * | 2019-06-11 | 2019-08-09 | 南京新酶合医药科技有限公司 | The preparation method of (5- tosyl -5H- pyrrolo- [2,3-b] pyrazine -2- base) methyl carbamate |
| TWI848141B (en) * | 2019-07-04 | 2024-07-11 | 英屬開曼群島商百濟神州有限公司 | PYRROLO[2,3-b]PYRAZINES AS HPK1 INHIBITOR AND THE USE THEREOF |
| TW202116771A (en) | 2019-07-17 | 2021-05-01 | 英屬開曼群島商百濟神州有限公司 | Tricyclic compounds as hpk1 inhibitor and the use thereof |
| CN110862380A (en) * | 2019-10-24 | 2020-03-06 | 嘉兴特科罗生物科技有限公司 | Small molecule compound |
| CN116836171B (en) * | 2023-06-30 | 2024-06-04 | 凌科药业(杭州)有限公司 | A sulfonamide compound and its application |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101098872A (en) * | 2004-11-22 | 2008-01-02 | 沃泰克斯药物股份有限公司 | Pyrrolopyrazines and pyrazolopyrazines useful as protein kinase inhibitors |
| CN102906095A (en) * | 2010-05-20 | 2013-01-30 | 弗·哈夫曼-拉罗切有限公司 | Pyrrolopyrazine derivatives as SYK and JAK inhibitors |
| WO2014172513A2 (en) * | 2013-04-18 | 2014-10-23 | Arrien Pharmaceuticals Llc | 3,5-(un)substituted-1h-pyrrolo[2,3-b]pyridine, 1h-pyrazolo[3,4-b]pyridine and 5h-pyrrolo [2-,3-b]pyrazine dual itk and jak3 kinase inhibitors |
| CN105777756A (en) * | 2014-07-02 | 2016-07-20 | 广东东阳光药业有限公司 | Heteroaryl compound and application thereof in medicines |
-
2016
- 2016-07-08 CN CN201610536665.3A patent/CN106336413B/en active Active
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101098872A (en) * | 2004-11-22 | 2008-01-02 | 沃泰克斯药物股份有限公司 | Pyrrolopyrazines and pyrazolopyrazines useful as protein kinase inhibitors |
| CN102906095A (en) * | 2010-05-20 | 2013-01-30 | 弗·哈夫曼-拉罗切有限公司 | Pyrrolopyrazine derivatives as SYK and JAK inhibitors |
| WO2014172513A2 (en) * | 2013-04-18 | 2014-10-23 | Arrien Pharmaceuticals Llc | 3,5-(un)substituted-1h-pyrrolo[2,3-b]pyridine, 1h-pyrazolo[3,4-b]pyridine and 5h-pyrrolo [2-,3-b]pyrazine dual itk and jak3 kinase inhibitors |
| CN105777756A (en) * | 2014-07-02 | 2016-07-20 | 广东东阳光药业有限公司 | Heteroaryl compound and application thereof in medicines |
Also Published As
| Publication number | Publication date |
|---|---|
| CN106336413A (en) | 2017-01-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN106336413B (en) | Compounds as JAK inhibitors and their uses | |
| TWI891666B (en) | Bicyclic heterocycles as fgfr inhibitors | |
| CN105732636B (en) | Heteroaromatic compounds and their application in medicine | |
| CN103038233B (en) | Pyridone and azapyridone compounds and methods of use | |
| CN106478651B (en) | Substituted heteroaryl compound and combinations thereof and purposes | |
| ES2699948T3 (en) | Substituted heteroaryl compounds and methods of use | |
| CN108570048B (en) | Substituted heteroaryl compounds, compositions and uses thereof | |
| CN105777756A (en) | Heteroaryl compound and application thereof in medicines | |
| CN109776522B (en) | Substituted heteroaryl compounds and compositions and uses thereof | |
| CN110036004A (en) | Inhibitors of cyclin-dependent kinase 7 (CDK7) | |
| CN114302886B (en) | Triazolopyridazine derivative, preparation method, pharmaceutical composition and application thereof | |
| CN105566321B (en) | Heteroaromatic compounds and their application in medicine | |
| WO2023109883A1 (en) | Aromatic heterocycle-substituted compounds, and preparation method therefor and use thereof | |
| ES2902365T3 (en) | Substituted furanopyrimidine compounds as PDE1 inhibitors | |
| CN105732637B (en) | Heteroaromatic compounds and their application in medicine | |
| CN106478607B (en) | Substituted heteroaryl compounds and compositions and uses thereof | |
| JP7254076B2 (en) | Substituted heteroaryl compounds and methods of use | |
| CN103261167A (en) | Substituted 6,6-used nitrogenous heterocyclic compounds and uses thereof | |
| WO2020103897A1 (en) | Heterocyclic fused pyrimidine derivative, pharmaceutical composition thereof, and application thereof | |
| CN106432246B (en) | Heteroaromatic compounds and their application in medicine | |
| WO2022033544A1 (en) | Substituted heteroaryl compound, and composition and application thereof | |
| WO2018169700A1 (en) | Substituted heteroaryl compounds and methods of use | |
| JP2018527362A (en) | Substituted heteroaryl compounds and methods of use | |
| CN104926795B (en) | Substituted heteroaryl compound and combinations thereof and purposes | |
| CN111278840A (en) | Pyrrolo [2, 3-B ] pyridine compounds and their use for treating cancer |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CP03 | Change of name, title or address |
Address after: 523808 No.1, Gongye North Road, Songshanhu Park, Dongguan City, Guangdong Province Patentee after: Guangdong Dongyangguang Pharmaceutical Co.,Ltd. Address before: 523808 No. 1 Industrial North Road, Songshan Industrial Park, Songshan, Guangdong, Dongguan, Hubei Patentee before: SUNSHINE LAKE PHARMA Co.,Ltd. |
|
| CP03 | Change of name, title or address |