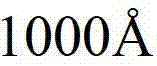CN103296138A - Low-cost solar cells and methods for their production - Google Patents
Low-cost solar cells and methods for their production Download PDFInfo
- Publication number
- CN103296138A CN103296138A CN2013100950349A CN201310095034A CN103296138A CN 103296138 A CN103296138 A CN 103296138A CN 2013100950349 A CN2013100950349 A CN 2013100950349A CN 201310095034 A CN201310095034 A CN 201310095034A CN 103296138 A CN103296138 A CN 103296138A
- Authority
- CN
- China
- Prior art keywords
- layer
- forming
- wafer
- silicon
- doped
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 238000000034 method Methods 0.000 title claims abstract description 70
- 238000004519 manufacturing process Methods 0.000 title claims abstract description 37
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 claims abstract description 104
- 229910052710 silicon Inorganic materials 0.000 claims abstract description 102
- 239000010703 silicon Substances 0.000 claims abstract description 102
- 238000000151 deposition Methods 0.000 claims abstract description 17
- 229910021420 polycrystalline silicon Inorganic materials 0.000 claims abstract description 16
- 235000012431 wafers Nutrition 0.000 claims description 96
- 229910052751 metal Inorganic materials 0.000 claims description 21
- 239000002184 metal Substances 0.000 claims description 21
- 238000000623 plasma-assisted chemical vapour deposition Methods 0.000 claims description 21
- 239000007789 gas Substances 0.000 claims description 19
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 claims description 15
- 229910052709 silver Inorganic materials 0.000 claims description 15
- 239000004332 silver Substances 0.000 claims description 15
- BLRPTPMANUNPDV-UHFFFAOYSA-N Silane Chemical compound [SiH4] BLRPTPMANUNPDV-UHFFFAOYSA-N 0.000 claims description 13
- 229920005591 polysilicon Polymers 0.000 claims description 13
- 229910000077 silane Inorganic materials 0.000 claims description 11
- 229910052782 aluminium Inorganic materials 0.000 claims description 10
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 claims description 9
- 229910052739 hydrogen Inorganic materials 0.000 claims description 8
- 238000005530 etching Methods 0.000 claims description 7
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 6
- 239000001257 hydrogen Substances 0.000 claims description 6
- 238000012545 processing Methods 0.000 claims description 6
- XYFCBTPGUUZFHI-UHFFFAOYSA-N Phosphine Chemical compound P XYFCBTPGUUZFHI-UHFFFAOYSA-N 0.000 claims description 5
- 238000007650 screen-printing Methods 0.000 claims description 5
- 238000004544 sputter deposition Methods 0.000 claims description 4
- 229910000073 phosphorus hydride Inorganic materials 0.000 claims description 3
- 150000002431 hydrogen Chemical class 0.000 claims description 2
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 claims description 2
- 239000004020 conductor Substances 0.000 claims 4
- 239000000383 hazardous chemical Substances 0.000 abstract description 10
- 238000002309 gasification Methods 0.000 abstract description 8
- 230000036541 health Effects 0.000 abstract description 4
- 239000010410 layer Substances 0.000 description 112
- 239000000758 substrate Substances 0.000 description 54
- 239000010409 thin film Substances 0.000 description 40
- 230000008569 process Effects 0.000 description 37
- 238000006243 chemical reaction Methods 0.000 description 26
- 239000010408 film Substances 0.000 description 26
- 229910021417 amorphous silicon Inorganic materials 0.000 description 21
- 239000000463 material Substances 0.000 description 21
- 239000012535 impurity Substances 0.000 description 18
- 238000009792 diffusion process Methods 0.000 description 15
- 239000004065 semiconductor Substances 0.000 description 12
- 150000003376 silicon Chemical class 0.000 description 11
- 239000011521 glass Substances 0.000 description 10
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 9
- 239000006096 absorbing agent Substances 0.000 description 9
- 238000002161 passivation Methods 0.000 description 9
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 7
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N silicon dioxide Inorganic materials O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 7
- 239000002019 doping agent Substances 0.000 description 6
- 125000004429 atom Chemical group 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 229910052799 carbon Inorganic materials 0.000 description 5
- 229910002804 graphite Inorganic materials 0.000 description 5
- 239000010439 graphite Substances 0.000 description 5
- 229910052796 boron Inorganic materials 0.000 description 4
- 239000013078 crystal Substances 0.000 description 4
- 230000008021 deposition Effects 0.000 description 4
- 230000006870 function Effects 0.000 description 4
- 239000000155 melt Substances 0.000 description 4
- 238000002844 melting Methods 0.000 description 4
- 230000008018 melting Effects 0.000 description 4
- 230000002829 reductive effect Effects 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 101100520660 Drosophila melanogaster Poc1 gene Proteins 0.000 description 3
- 101100520662 Saccharomyces cerevisiae (strain ATCC 204508 / S288c) PBA1 gene Proteins 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 239000000969 carrier Substances 0.000 description 3
- 150000001875 compounds Chemical class 0.000 description 3
- 238000001816 cooling Methods 0.000 description 3
- 230000007613 environmental effect Effects 0.000 description 3
- 231100001261 hazardous Toxicity 0.000 description 3
- BHEPBYXIRTUNPN-UHFFFAOYSA-N hydridophosphorus(.) (triplet) Chemical compound [PH] BHEPBYXIRTUNPN-UHFFFAOYSA-N 0.000 description 3
- 229910021421 monocrystalline silicon Inorganic materials 0.000 description 3
- 239000008188 pellet Substances 0.000 description 3
- RLOWWWKZYUNIDI-UHFFFAOYSA-N phosphinic chloride Chemical compound ClP=O RLOWWWKZYUNIDI-UHFFFAOYSA-N 0.000 description 3
- 238000007639 printing Methods 0.000 description 3
- 239000010453 quartz Substances 0.000 description 3
- 230000009467 reduction Effects 0.000 description 3
- 229910052814 silicon oxide Inorganic materials 0.000 description 3
- XKRFYHLGVUSROY-UHFFFAOYSA-N Argon Chemical compound [Ar] XKRFYHLGVUSROY-UHFFFAOYSA-N 0.000 description 2
- ZOXJGFHDIHLPTG-UHFFFAOYSA-N Boron Chemical compound [B] ZOXJGFHDIHLPTG-UHFFFAOYSA-N 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 230000003667 anti-reflective effect Effects 0.000 description 2
- 238000013459 approach Methods 0.000 description 2
- 238000005266 casting Methods 0.000 description 2
- 229910052804 chromium Inorganic materials 0.000 description 2
- 238000010276 construction Methods 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 229910021419 crystalline silicon Inorganic materials 0.000 description 2
- 238000013461 design Methods 0.000 description 2
- 238000010586 diagram Methods 0.000 description 2
- 238000005516 engineering process Methods 0.000 description 2
- 229910052742 iron Inorganic materials 0.000 description 2
- 230000031700 light absorption Effects 0.000 description 2
- 229910052748 manganese Inorganic materials 0.000 description 2
- 239000006060 molten glass Substances 0.000 description 2
- 229910052759 nickel Inorganic materials 0.000 description 2
- XHXFXVLFKHQFAL-UHFFFAOYSA-N phosphoryl trichloride Chemical compound ClP(Cl)(Cl)=O XHXFXVLFKHQFAL-UHFFFAOYSA-N 0.000 description 2
- 238000005498 polishing Methods 0.000 description 2
- 238000010248 power generation Methods 0.000 description 2
- 239000011241 protective layer Substances 0.000 description 2
- 229920005989 resin Polymers 0.000 description 2
- 239000011347 resin Substances 0.000 description 2
- 239000000523 sample Substances 0.000 description 2
- LIVNPJMFVYWSIS-UHFFFAOYSA-N silicon monoxide Chemical compound [Si-]#[O+] LIVNPJMFVYWSIS-UHFFFAOYSA-N 0.000 description 2
- 239000011863 silicon-based powder Substances 0.000 description 2
- 238000010583 slow cooling Methods 0.000 description 2
- 229910021422 solar-grade silicon Inorganic materials 0.000 description 2
- 238000000859 sublimation Methods 0.000 description 2
- 230000008022 sublimation Effects 0.000 description 2
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- 229910004613 CdTe Inorganic materials 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 1
- ZOKXTWBITQBERF-UHFFFAOYSA-N Molybdenum Chemical compound [Mo] ZOKXTWBITQBERF-UHFFFAOYSA-N 0.000 description 1
- 229910019213 POCl3 Inorganic materials 0.000 description 1
- 229910020286 SiOxNy Inorganic materials 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 239000012670 alkaline solution Substances 0.000 description 1
- AZDRQVAHHNSJOQ-UHFFFAOYSA-N alumane Chemical compound [AlH3] AZDRQVAHHNSJOQ-UHFFFAOYSA-N 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 239000006117 anti-reflective coating Substances 0.000 description 1
- 229910052786 argon Inorganic materials 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- DQXBYHZEEUGOBF-UHFFFAOYSA-N but-3-enoic acid;ethene Chemical compound C=C.OC(=O)CC=C DQXBYHZEEUGOBF-UHFFFAOYSA-N 0.000 description 1
- 239000006227 byproduct Substances 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000002800 charge carrier Substances 0.000 description 1
- 238000003486 chemical etching Methods 0.000 description 1
- 238000004140 cleaning Methods 0.000 description 1
- 239000003245 coal Substances 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 239000000356 contaminant Substances 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 238000000354 decomposition reaction Methods 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000003795 desorption Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 229910003460 diamond Inorganic materials 0.000 description 1
- 239000010432 diamond Substances 0.000 description 1
- MROCJMGDEKINLD-UHFFFAOYSA-N dichlorosilane Chemical compound Cl[SiH2]Cl MROCJMGDEKINLD-UHFFFAOYSA-N 0.000 description 1
- 238000005553 drilling Methods 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 230000005611 electricity Effects 0.000 description 1
- 239000005038 ethylene vinyl acetate Substances 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 239000002803 fossil fuel Substances 0.000 description 1
- 238000005247 gettering Methods 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 231100000206 health hazard Toxicity 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 238000011065 in-situ storage Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000009434 installation Methods 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 150000004767 nitrides Chemical class 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 238000010422 painting Methods 0.000 description 1
- 239000003208 petroleum Substances 0.000 description 1
- 238000000206 photolithography Methods 0.000 description 1
- 238000000053 physical method Methods 0.000 description 1
- 238000005268 plasma chemical vapour deposition Methods 0.000 description 1
- 229920001200 poly(ethylene-vinyl acetate) Polymers 0.000 description 1
- 229920000642 polymer Polymers 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000002994 raw material Substances 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000002441 reversible effect Effects 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- 238000007788 roughening Methods 0.000 description 1
- 239000004576 sand Substances 0.000 description 1
- 229910052990 silicon hydride Inorganic materials 0.000 description 1
- 239000002210 silicon-based material Substances 0.000 description 1
- 238000007711 solidification Methods 0.000 description 1
- 230000008023 solidification Effects 0.000 description 1
- 239000000243 solution Substances 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 229910052723 transition metal Inorganic materials 0.000 description 1
- 150000003624 transition metals Chemical class 0.000 description 1
- ZDHXKXAHOVTTAH-UHFFFAOYSA-N trichlorosilane Chemical compound Cl[SiH](Cl)Cl ZDHXKXAHOVTTAH-UHFFFAOYSA-N 0.000 description 1
- 239000005052 trichlorosilane Substances 0.000 description 1
- 238000009827 uniform distribution Methods 0.000 description 1
- 238000007740 vapor deposition Methods 0.000 description 1
- 238000003631 wet chemical etching Methods 0.000 description 1
Images
Classifications
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10F—INORGANIC SEMICONDUCTOR DEVICES SENSITIVE TO INFRARED RADIATION, LIGHT, ELECTROMAGNETIC RADIATION OF SHORTER WAVELENGTH OR CORPUSCULAR RADIATION
- H10F71/00—Manufacture or treatment of devices covered by this subclass
- H10F71/121—The active layers comprising only Group IV materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10F—INORGANIC SEMICONDUCTOR DEVICES SENSITIVE TO INFRARED RADIATION, LIGHT, ELECTROMAGNETIC RADIATION OF SHORTER WAVELENGTH OR CORPUSCULAR RADIATION
- H10F10/00—Individual photovoltaic cells, e.g. solar cells
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10F—INORGANIC SEMICONDUCTOR DEVICES SENSITIVE TO INFRARED RADIATION, LIGHT, ELECTROMAGNETIC RADIATION OF SHORTER WAVELENGTH OR CORPUSCULAR RADIATION
- H10F10/00—Individual photovoltaic cells, e.g. solar cells
- H10F10/10—Individual photovoltaic cells, e.g. solar cells having potential barriers
- H10F10/16—Photovoltaic cells having only PN heterojunction potential barriers
- H10F10/164—Photovoltaic cells having only PN heterojunction potential barriers comprising heterojunctions with Group IV materials, e.g. ITO/Si or GaAs/SiGe photovoltaic cells
- H10F10/165—Photovoltaic cells having only PN heterojunction potential barriers comprising heterojunctions with Group IV materials, e.g. ITO/Si or GaAs/SiGe photovoltaic cells the heterojunctions being Group IV-IV heterojunctions, e.g. Si/Ge, SiGe/Si or Si/SiC photovoltaic cells
- H10F10/166—Photovoltaic cells having only PN heterojunction potential barriers comprising heterojunctions with Group IV materials, e.g. ITO/Si or GaAs/SiGe photovoltaic cells the heterojunctions being Group IV-IV heterojunctions, e.g. Si/Ge, SiGe/Si or Si/SiC photovoltaic cells the Group IV-IV heterojunctions being heterojunctions of crystalline and amorphous materials, e.g. silicon heterojunction [SHJ] photovoltaic cells
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10F—INORGANIC SEMICONDUCTOR DEVICES SENSITIVE TO INFRARED RADIATION, LIGHT, ELECTROMAGNETIC RADIATION OF SHORTER WAVELENGTH OR CORPUSCULAR RADIATION
- H10F77/00—Constructional details of devices covered by this subclass
- H10F77/10—Semiconductor bodies
- H10F77/12—Active materials
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/547—Monocrystalline silicon PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/548—Amorphous silicon PV cells
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P70/00—Climate change mitigation technologies in the production process for final industrial or consumer products
- Y02P70/50—Manufacturing or production processes characterised by the final manufactured product
Landscapes
- Photovoltaic Devices (AREA)
Abstract
本发明涉及低成本的太阳能电池及其生产方法,其无需进行冶金级硅的气化来制造太阳能电池,因此避免了在太阳能级或硅级硅的制造中涉及的成本和健康以及环境危害。该生产太阳能电池的方法包括:获得基本上由被掺杂为p-型或n-型的冶金级硅组成的多晶硅晶片;织构该晶片的正面;直接在该晶片的正面上并且与该晶片的正面接触地沉积本征层;在所述本征层上并且与该本征层接触地沉积与该晶片相反极性的掺杂层;在所述掺杂层上形成顶导电接触;以及在所述晶片的底面上形成底导电接触。
The present invention relates to low cost solar cells and methods of production thereof which do not require the gasification of metallurgical grade silicon to manufacture solar cells, thus avoiding the costs and health and environmental hazards involved in the manufacture of solar grade or silicon grade silicon. The method of producing solar cells comprises: obtaining a polycrystalline silicon wafer consisting essentially of metallurgical grade silicon doped p-type or n-type; texturing the front side of the wafer; directly on the front side of the wafer and with the wafer depositing an intrinsic layer in contact with the front side of the intrinsic layer; depositing a doped layer of opposite polarity to the wafer on and in contact with the intrinsic layer; forming a top conductive contact on the doped layer; and Bottom conductive contacts are formed on the bottom surface of the wafer.
Description
本申请是原申请的申请日为2008年11月7日,申请号为200880115434.9,发明名称为《低成本的太阳能电池及其生产方法》的中国专利申请的分案申请。相关申请This application is a divisional application of the Chinese patent application with the filing date of the original application being November 7, 2008, the application number being 200880115434.9, and the invention title being "low-cost solar cell and its production method". related application
本申请要求2007年11月9日提交的美国临时申请序列号60/986,996的优先权,其公开的内容全部结合到本文。This application claims priority to US Provisional Application Serial No. 60/986,996, filed November 9, 2007, the disclosure of which is incorporated herein in its entirety.
技术领域technical field
本发明涉及太阳能光伏电池,更具体地涉及用于供这样的电池使用的低成本基本材料的制造方法以及用于制造低成本电池的方法以及所获得的电池器件结构体。The present invention relates to solar photovoltaic cells, and more particularly to methods of manufacturing low cost base materials for use in such cells and methods for manufacturing low cost cells and resulting cell device structures.
背景技术Background technique
常规的由化石燃料产生能源扮演着自上次冰川期以来对地球安宁的最大威胁。在所有的替代能源中,除节能之外,与其它途径如乙醇、水电和风能相比,可以论证太阳能光伏电池是最干净的、普遍存在的并且可能是最可靠的选择。原理是将光转换为小的直流电压的简单固态p-n结。可以将电池层叠以对车用电池充电或通过DC/AC转换输入电网。在可用于该目的各种半导体材料中,硅占据光伏太阳能电池产量的99%。与其它基于化合物半导体的太阳能电池相比,尽管其具有较高的转换效率,特别是在小面积电池中,但硅在地壳中丰富得多,并在全世界各种气候下在经风吹雨打的屋顶上提供高达30年的得到证明的可靠性。此外,利用硅的大规模商业制造技术已经使用了数十年并且发展良好和便于理解。因此,硅很可能仍然是占优势的用于太阳能电池的基本材料。Conventional energy generation from fossil fuels poses the greatest threat to the peace of the planet since the last ice age. Of all the alternative energy sources, solar photovoltaic cells are arguably the cleanest, ubiquitous and probably the most reliable option compared to other sources such as ethanol, hydropower and wind energy, in addition to energy savings. The principle is a simple solid-state p-n junction that converts light into a small DC voltage. Batteries can be stacked to charge a car battery or feed into the grid through DC/AC conversion. Among the various semiconductor materials that can be used for this purpose, silicon accounts for 99% of the production of photovoltaic solar cells. Compared to other compound semiconductor-based solar cells, despite its high conversion efficiency, especially in small-area cells, silicon is much more abundant in the Earth's crust and is weathered in all climates around the world. Hit roofs offer up to 30 years of proven reliability. Furthermore, large-scale commercial fabrication techniques utilizing silicon have been in use for decades and are well developed and well understood. Therefore, silicon is likely to remain the predominant basic material for solar cells.
然而,尽管经三十年的发展,硅基太阳能电池还没有发挥出其用于大规模发电的潜能。对其认可的主要障碍是与制造太阳能电池有关的成本,尤其是用于制造太阳能电池的原料、基本材料(基板)的成本。与半导体微芯片的情况下仅约10%相比,材料占太阳能电池制造总成本的超过一半。讽刺地是,因为巨大的需求和高生产成本,用于太阳能电池的硅材料的价格实际上和油价同步增加。例如,在过去几年中,用于生产太阳能硅晶片的多晶硅材料每kg的成本显著增加,对于薄膜太阳电池,用于沉积该膜的硅烷气体的成本以及在沉积后清洗反应器的NF3气体的成本同样地增加。相反,半导体芯片价格(即,每单元存储或逻辑功能)在过去的三十年按照摩尔定律指数地降低。学习曲线的这种不同可能涉及技术上以及材料相对于成本的比重(相比于日益提高的每单位面积器件密度的工艺和设计)的主要差别。However, despite three decades of development, silicon-based solar cells have yet to realize their potential for large-scale power generation. The main obstacle to its acceptance is the cost associated with manufacturing solar cells, especially the cost of the raw material, the base material (substrate) used to manufacture solar cells. The material accounts for more than half of the total cost of solar cell manufacturing, compared to only about 10% in the case of semiconductor microchips. Ironically, the price of silicon, the material used in solar cells, has actually risen in tandem with the price of oil because of huge demand and high production costs. For example, the cost per kg of polysilicon material used to produce solar silicon wafers has increased significantly over the past few years, and for thin-film solar cells, the cost of silane gas to deposit the film and NF gas to clean the reactor after deposition The cost also increases. In contrast, semiconductor chip prices (ie, per unit of memory or logic function) have decreased exponentially over the past three decades in accordance with Moore's Law. This difference in learning curves may involve major differences in technology and the weighting of materials versus cost (compared to process and design for increasing device density per unit area).
根据本领域的目前状态,基于多晶硅的太阳能电池生产按三个主要阶段进行。第一,为较适中的25MW容量工厂生产用于基板的大量硅晶片――典型地每月百万个晶片。第二,通过形成p-n结并金属化将这些晶片加工成太阳能电池。第三,然后将这些晶片“封装”成供用户设施中的安装使用的模块。According to the current state of the art, the production of solar cells based on polycrystalline silicon proceeds in three main stages. First, a relatively modest 25MW capacity plant produces large quantities of silicon wafers for substrates - typically millions of wafers per month. Second, these wafers are processed into solar cells by forming p-n junctions and metallizing them. Third, these chips are then "packaged" into modules for installation in the user's facility.
通过热分解含Si-H-Cl如二氯硅烷和三氯硅烷的危险气体生产超高纯的多晶硅,通常指九个九,即99.9999999%纯度,制造用于太阳能电池的基本硅晶片。这些气体是极易燃且有毒的。然而,由于在硅的气化中危害环境和健康,世界上仅有很少的工厂在运转,从而导致半导体和太阳能电池行业的瓶颈。新计划的硅气化工厂面临来自当地社区基于环境和安全关切的阻力。这些工厂还需要大量的资本投入和较长的建设周期。因此,在裸硅晶片的需求与供给之间始终存在不平衡。Ultra-high-purity polysilicon, usually referred to as nine nines, or 99.9999999% pure, is produced by thermally decomposing hazardous gases containing Si-H-Cl such as dichlorosilane and trichlorosilane, making the basic silicon wafers used in solar cells. These gases are extremely flammable and toxic. However, due to environmental and health hazards in the gasification of silicon, only a few factories are operating in the world, causing bottlenecks in the semiconductor and solar cell industries. Newly planned silicon gasification plants face resistance from local communities based on environmental and safety concerns. These plants also require significant capital investment and long construction periods. Therefore, there is always an imbalance between the demand and supply of bare silicon wafers.
通常按适合半导体和太阳能电池应用的颗粒状来提供纯硅(称为多晶硅,在硅烷基化合物的气化和分解之后)。然后使颗粒熔融并利用晶种提拉单晶棒或多晶带。或者,将多晶硅铸成柱状。将提拉的柱体锯切、成型并抛光成5~6英寸的圆形晶片,其后可以将其切割成正方形晶片。然后在碱性化学品例如KOH中进行湿化学蚀刻以织构。利用POC13熔炉扩散形成p-n结。然后利用PECVD SiON进行抗反射膜钝化。将丝网印刷的银糊涂覆到n-型面以及将铝糊涂覆到p-型面。然后烧结该糊以形成电接触。最后,测试电池并根据它们的特性如它们的I-V曲线来分类。Pure silicon (called polysilicon, after gasification and decomposition of silane-based compounds) is usually supplied in granular form suitable for semiconductor and solar cell applications. The pellets are then melted and the single crystal rods or polycrystalline ribbons are pulled using the seed crystals. Alternatively, the polysilicon is cast into pillars. The pulled cylinder is sawn, shaped and polished into 5-6 inch circular wafers which can then be cut into square wafers. Wet chemical etching is then performed in alkaline chemicals such as KOH to texture. Use POC1 3 furnace diffusion to form pn junction. The antireflective film passivation is then performed using PECVD SiON. Screen printed silver paste was applied to the n-type side and aluminum paste was applied to the p-type side. The paste is then sintered to form electrical contacts. Finally, the cells are tested and sorted according to their characteristics such as their IV curves.
上述工艺在本行业中是众所周知的并且已经实施多年。然而,虽然在半导体中大部分成本(即,价值)在于将抛光的硅晶片转换为功能性集成电路的过程,但在太阳能电池制造中,将抛光后的晶片转变为功能性太阳能电池的过程比生产抛光晶片自身的过程成本低。也就是说,在商业意义上,将硅晶片转变成太阳能电池的过程在整个太阳电池板制造链中不是高附加价值的步骤。因此,与电池制造技术相反――在制造初始晶片的成本方面的任何改进或降低将能够使最终的太阳电池板的价格大幅度降低。The above process is well known in the industry and has been practiced for many years. However, while in semiconductors most of the cost (i.e., value) lies in the process of converting polished silicon wafers into functional integrated circuits, in solar cell manufacturing the process of converting polished wafers into functional solar cells is less expensive than The process of producing the polished wafer itself is low cost. That said, the process of turning silicon wafers into solar cells is not a high value-added step in the overall solar panel manufacturing chain in a commercial sense. So, in contrast to cell manufacturing technology - any improvement or reduction in the cost of making the initial wafer will enable a substantial reduction in the price of the final solar panel.
为了克服用于太阳能电池的硅原料的问题,已经沿着两个主要的途径积极地致力于降低太阳能电池每瓦消耗的硅量。这些是:In order to overcome the problem of silicon feedstock for solar cells, there have been active efforts to reduce the amount of silicon consumed per watt by solar cells along two main avenues. these are:
1.将晶片厚度从标准的500μm降低到~300μm。该途径受到晶片强度的限制,在高速通过加工设备期间晶片趋向破裂。1. Reduce wafer thickness from standard 500 μm to ~300 μm. This approach is limited by the strength of the wafer, which tends to crack during passage through the processing equipment at high speeds.
2.采用各种太阳能电池材料例如硅、CdTe、CuInGaSe的薄膜,典型地在玻璃上以及在其它较便宜的基板上。为了使光照射在太阳能电池上,电极之一由导电的透明氧化物(CTO)例如InSnOx或者ZnO2构成。2. Using thin films of various solar cell materials such as silicon, CdTe, CuInGaSe, typically on glass and on other less expensive substrates. To allow light to impinge on the solar cell, one of the electrodes consists of a conductive transparent oxide (CTO) such as InSnO x or ZnO 2 .
在各种薄膜太阳电池材料中,硅同样是最经济合算的材料。在该太阳能结构体中,将晶片厚度从300~500μm降低至约1~10μm。在该1~10μm中,大部分沉积的膜的厚度典型地由未掺杂的Si-H聚合物本征非晶层组成,缩写成ia-Si:H层。夹在掺杂的n-型a-Si:H和p-型a-Si:H膜之间的该i aSi:H层提供吸收入射阳光所需的容积,从而在其中产生电子空穴对。然后这些载流子扩散到太阳能电池的n-和p-电极来产生用于发电的光伏电压和电流。然而,因为太阳光谱的红外线波长具有通过硅的长透射深度,所以损失大量太阳辐射,从而降低了光伏转换的效率。也就是说,损失了转换的量子效率,特别对于在红外线范围内的较长波长。薄膜结构的另一个固有局限性在于少数载流子的扩散长度被膜的厚度限制到远小于10μm。这是预测成品的太阳能电池效率的品质因素。对于纯的晶体硅基太阳能电池,扩散长度典型地为约80μm。Among various thin-film solar cell materials, silicon is also the most cost-effective material. In this solar structure, the wafer thickness is reduced from 300-500 μm to about 1-10 μm. Within this 1-10 μm, most of the deposited film thickness typically consists of an intrinsic amorphous layer of undoped Si-H polymer, abbreviated as ia-Si:H layer. This i aSi:H layer, sandwiched between doped n-type a-Si:H and p-type a-Si:H films, provides the volume needed to absorb incident sunlight, generating electron-hole pairs therein. These carriers then diffuse to the n- and p-electrodes of the solar cell to generate photovoltaic voltage and current for power generation. However, because the infrared wavelengths of the solar spectrum have a long transmission depth through silicon, a large amount of solar radiation is lost, reducing the efficiency of photovoltaic conversion. That is, the quantum efficiency of the conversion is lost, especially for longer wavelengths in the infrared range. Another inherent limitation of thin-film structures is that the minority carrier diffusion length is limited by the thickness of the film to much less than 10 μm. This is the figure of merit that predicts the efficiency of a finished solar cell. For pure crystalline silicon-based solar cells, the diffusion length is typically around 80 μm.
薄膜太阳电池结构存在其它根本性的局限性,与基于硅晶片的太阳能电池超过总的太阳电池板市场的80%相比,迄今这些局限性将薄膜太阳电池产量限制在约5%。这些局限性中部分如下:There are other fundamental limitations to thin-film solar cell construction that have so far limited thin-film solar cell production to about 5%, compared with silicon wafer-based solar cells, which account for more than 80% of the total solar panel market. Some of these limitations are as follows:
1.由于与多晶硅的价格相同的原因,即用于沉积a-Si:H膜的硅烷气体的产量不足,该极易燃的气体的成本快速上升。除硅烷之外,用于生产太阳能薄膜的等离子增强CVD反应器需要大量专门的NF3气体来执行PECVD反应器的原位等离子净化以确保生产设备的正常运转时间长。1. Due to the same reason as the price of polysilicon, that is, insufficient production of silane gas for depositing a-Si:H films, the cost of this highly flammable gas is rapidly rising. In addition to silane, plasma-enhanced CVD reactors for the production of solar thin films require large quantities of specialized NF3 gases to perform in-situ plasma cleaning of PECVD reactors to ensure high production equipment uptime.
2.薄膜硅太阳能电池的光伏转换效率低,有时小于基于硅晶片的太阳能电池的一半。2. The photovoltaic conversion efficiency of thin-film silicon solar cells is low, sometimes less than half that of silicon wafer-based solar cells.
3.建立薄膜太阳电池工厂所需的通常在约$50M的固定设备是用于能量输出相当的基于硅晶片的太阳能电池工厂的将近10倍。基本费用主要由用于沉积a-Si:H和SiN钝化膜的基于真空的等离子CVD反应器以及用于沉积CTO膜的基于真空的PVD反应器所推动。3. The capital equipment required to set up a thin film solar cell factory, typically at about $50M, is nearly 10 times that for a comparable energy output silicon wafer based solar cell factory. Base costs are mainly driven by vacuum-based plasma CVD reactors for deposition of a-Si:H and SiN passivation films, and vacuum-based PVD reactors for deposition of CTO films.
如同能够由上述所理解的那样,太阳能电池行业已经被分为两个阵营:设法利用高纯硅晶片来获得高电池效率的基于硅晶片的太阳能电池阵营,以及为了降低成本避开使用硅晶片的薄膜阵营。因此,基于硅晶片的阵营受到纯硅晶片可获得性的限制,而薄膜阵营受到主要由于玻璃基板中光的吸收不足所导致的转换效率的限制,以及受到生产较厚的本征氢化硅吸收层所需的SiH4气体的成本的限制。As can be understood from the above, the solar cell industry has been divided into two camps: the camp of silicon wafer-based solar cells that seek to utilize high-purity silicon wafers to obtain high cell efficiencies, and those that eschew the use of silicon wafers in order to reduce costs. film camp. Thus, the silicon wafer-based camp is limited by the availability of pure silicon wafers, while the thin-film camp is limited by conversion efficiencies mainly due to insufficient absorption of light in glass substrates, and by the production of thicker intrinsic hydrogenated silicon absorber layers The cost of the required SiH 4 gas is constrained.
发明内容Contents of the invention
为了提供本发明的一些方面的基本理解及特征,包括如下发明概述。该概述不是本发明的广泛综述,因而不是意图特别区分本发明的关键或重要的要素或者描绘本发明的范围。其唯一的目的是以简化的形式给出本发明的一些原理,作为下面给出的更详细描述的前奏。The following Summary of the Invention is included to provide a basic understanding and characterization of some aspects of the invention. This summary is not an extensive overview of the invention and as such it is not intended to particularly identify key or critical elements of the invention or to delineate the scope of the invention. Its sole purpose is to present some principles of the invention in a simplified form as a prelude to the more detailed description that is presented below.
本发明的各种实施方式提供无需进行硅的气化来制造硅基板的方法。因此,避免了在制造九个九等级的硅中涉及的成本和健康以及环境危害。该基板可以用于制造效率与薄膜太阳电池相当甚至超过的太阳能电池。Various embodiments of the present invention provide methods for fabricating silicon substrates without the need for gasification of silicon. Thus, the costs and health and environmental hazards involved in manufacturing nine-nine-grade silicon are avoided. The substrate can be used to fabricate solar cells with efficiencies that match or exceed those of thin-film solar cells.
本发明的特征解决了太阳能电池行业面临的一个以上如下重要问题:The features of the present invention solve one or more important problems faced by the solar cell industry as follows:
a)用于晶片和薄膜的“具有太阳能能力的(solar capable)”硅材料的可获得性和成本a) Availability and cost of "solar capable" silicon materials for wafers and thin films
b)用于太阳能电池工厂的投资费用b) Investment costs for solar cell factories
c)未来太阳能电池的每瓦所需的成本。c) Cost per watt required for future solar cells.
d)用于大体积的生产工艺的规模性d) Scalability for large volume production processes
e)环境适应性和25年的可靠性e) Environmental adaptability and 25-year reliability
本发明的特征使得能够获得对上述问题的有生产价值的解决方案,尤其通过制造投资在体硅晶片的转换效率和薄膜电池结构的益处的太阳能电池结构。根据本发明的方面,通过利用由成本极低的冶金级硅制成的硅晶片作为基板并在该基板上制造薄膜太阳电池,制造太阳能电池。根据本发明的特征,通过沉积比常规的薄膜太阳电池薄得多的(例如,10%)薄膜来制造电池。除降低基板和膜材料的成本外,建议的结构允许超过常规薄膜太阳电池的提高的转换效率。也就是说,通过利用冶金级硅晶片,基板的制造变得危害较少并且更环保,同时还降低基板的成本。此外,利用冶金级硅晶片作为基板,与在玻璃上形成的薄膜结构相比提高了转换效率。The features of the present invention make it possible to obtain a production-worthy solution to the above-mentioned problems, in particular by manufacturing solar cell structures investing in the conversion efficiency of bulk silicon wafers and the benefits of thin-film cell structures. According to aspects of the invention, solar cells are fabricated by utilizing silicon wafers made of very low cost metallurgical grade silicon as substrates and fabricating thin film solar cells on the substrates. According to a feature of the invention, the cell is fabricated by depositing a much thinner (eg 10%) film than conventional thin film solar cells. In addition to reducing the cost of substrate and membrane materials, the proposed structure allows for improved conversion efficiencies over conventional thin film solar cells. That is, by utilizing metallurgical grade silicon wafers, the manufacture of the substrate becomes less hazardous and more environmentally friendly, while also reducing the cost of the substrate. In addition, utilizing metallurgical-grade silicon wafers as substrates improves conversion efficiency compared to thin-film structures formed on glass.
附图说明Description of drawings
根据参照下列附图的详细描述,本发明的其它方面和特征将会变得清楚。应当理解的是,详细描述和附图提供由所附权利要求限定的本发明的各种实施方式的各种非限制性的实施例。Other aspects and features of the present invention will become apparent from the detailed description with reference to the following drawings. It should be understood that the detailed description and drawings provide various non-limiting examples of various embodiments of the invention as defined by the appended claims.
结合到该说明书中并构成其一部分的附图举例说明了本发明的实施方式,并和描述一起起到解释和说明本发明的原则的作用。附图意图以图示的方式说明示例性的实施方式的特征。附图不是意图描绘实际实施方式的每个特征或所描绘的元件的相对尺寸,并且未按比例描绘。The accompanying drawings, which are incorporated in and constitute a part of this specification, illustrate embodiments of the invention and, together with the description, serve to explain and illustrate the principles of the invention. The drawings are intended to illustrate features of the exemplary embodiments by way of illustration. The drawings are not intended to depict every feature of actual implementations or relative dimensions of the depicted elements, and are not drawn to scale.
图1是说明根据本发明一个实施方式的工艺的流程图。Figure 1 is a flow diagram illustrating a process according to one embodiment of the present invention.
图2说明根据本发明一个实施方式的工艺。Figure 2 illustrates a process according to one embodiment of the invention.
图3说明根据本发明另一个实施方式的工艺。Figure 3 illustrates a process according to another embodiment of the invention.
图4说明根据本发明一个实施方式的工艺。Figure 4 illustrates a process according to one embodiment of the invention.
图5说明本发明的另一个实施方式。Figure 5 illustrates another embodiment of the invention.
图6说明本发明的另一个实施方式。Figure 6 illustrates another embodiment of the present invention.
图7说明本发明的另一个实施方式。Figure 7 illustrates another embodiment of the present invention.
图8说明本发明的另一个实施方式,其类似于图7的实施方式,除了将掺杂剂颠倒。Figure 8 illustrates another embodiment of the invention which is similar to the embodiment of Figure 7 except that the dopants are reversed.
图9A说明用于制造通常本文中称为SmartSiTM的准备好的太阳能电池基板(a solar-cell ready substrate)的工艺的实施例。Figure 9A illustrates an embodiment of a process for fabricating a solar-cell ready substrate, generally referred to herein as SmartSi ™ .
图9B说明可用于将SmartSi晶片转换为SmartSi PV太阳能电池的工艺的实施例。Figure 9B illustrates an embodiment of a process that may be used to convert SmartSi wafers to SmartSi PV solar cells.
图10说明根据本发明的实施方式制造的完整的太阳能电池。Figure 10 illustrates a complete solar cell fabricated in accordance with an embodiment of the invention.
图11A和11B说明多结SmartSi太阳能电池的实施方式。11A and 11B illustrate an embodiment of a multi-junction SmartSi solar cell.
图12A和12B说明具有扩散结的多结SmartSi太阳能电池的实施方式。12A and 12B illustrate an embodiment of a multi-junction SmartSi solar cell with diffused junctions.
图13A和13B说明两面夹在i-Si/掺杂的Si:H薄膜之间的冶金硅基板的对称配置的实施方式。13A and 13B illustrate an embodiment of a symmetrical configuration of a metallurgical silicon substrate sandwiched on both sides between i-Si/doped Si:H thin films.
具体实施方式Detailed ways
本发明的实施方式提供用于低成本制造太阳能电池的方法,同时减少常规的太阳能电池制造中所涉及的健康和环境危害。如在美国能源部的太阳能网站上指出的那样:“要用作太阳能电池中的半导体材料,硅必须被提纯到99.9999%的纯度。”(可在http://wwwl.eere.energy.gov/solar/silicon.html上获得)。这通常被称为6N或者太阳能级硅、SoG Si。与传统智慧相反,本发明提供利用纯度3N~5N的冶金级硅MG Si生产基板和太阳能电池的方法。各种实施方式公开了结合硅基太阳能电池和薄膜基太阳能电池的益处以提供转换效率约14%的电池。Embodiments of the present invention provide methods for low-cost fabrication of solar cells while reducing health and environmental hazards involved in conventional solar cell fabrication. As noted on the U.S. Department of Energy's Solar Energy website: "To be used as a semiconductor material in solar cells, silicon must be purified to a purity of 99.9999 percent." (Available at http://wwwl.eere.energy.gov/ available at solar/silicon.html ). This is commonly referred to as 6N or solar grade silicon, SoG Si. Contrary to conventional wisdom, the present invention provides a method for producing substrates and solar cells utilizing metallurgical grade silicon MG Si of purity 3N-5N. Various embodiments disclose combining the benefits of silicon-based solar cells and thin-film based solar cells to provide cells with conversion efficiencies of about 14%.
图1是说明根据本发明一个实施方式的工艺的流程图。在图1中,该工艺开始于步骤100,通过用石墨还原石英来生产冶金硅颗粒原料。所得的纯度水平可以是99.9%或99.999%的纯度,即约三个九到约五个九的纯度。值得注意的是,石英容易低成本地获得。此外,该工艺省略了气化步骤,从而避免了气化和硅烷生产的危险过程。Figure 1 is a flow diagram illustrating a process according to one embodiment of the present invention. In FIG. 1 , the process begins at step 100 with the production of metallurgical silicon particulate feedstock by reducing quartz with graphite. The resulting purity level may be 99.9% or 99.999% pure, ie, about three nines to about five nines. Notably, quartz is readily and inexpensively available. In addition, the process omits the gasification step, thereby avoiding the hazardous process of gasification and silane production.
在下一步200中,将粉末熔融到5英寸、6英寸、8英寸等正方形或圆形的模具中,并使液体缓慢凝固成具有大硅晶粒的圆柱体。任选地,将凝固的圆柱体重熔,然后分部分凝固,以便将杂质移到圆柱体的一侧。在该过程中沿水平方向控制冷却速率和温度梯度,以便使杂质移动到圆柱体的表面并沿垂直方向使杂质集中在硅晶界。在步骤200中,任选地用少量硼掺杂该熔融硅以生产含1E17~1E18cm-3硼的p-Si。或者可以利用常规的Czochralsky工艺从熔体提拉p-型单晶。在步骤300中,将凝固圆柱体的表面加工到抛光状态。在步骤400中,用例如金刚石或钢丝锯将该圆柱体切割成约20密耳,即0.020英寸厚的Si晶片,然后在一面上抛光表面,同时将另一面化学蚀刻成光洁表面。可以收集废料在熔融中重新使用。一旦完成太阳能电池的制造(完成下述全部过程),如果希望,可以进一步使晶片变薄,使得p-层为约0.007英寸。In a
图2说明根据本发明一个实施方式的工艺。该工艺开始于由“脏的”硅,即例如通过图1的实施方式的工艺获得的3~5个九的冶金硅制成的晶片DSi200。采用常规的炉在约900℃下将晶片200暴露于POC13,以便在晶片210的顶部上提供浓度为1.0e16原子/cm3的n-层。然后用等离子室在该n-型层210上提供SiN层220。然后利用在高于900℃的温度下的POC13处理将杂质从DSi层200提取到熔融玻璃层230中。这提高了p-型层200的纯度,尤其是此前形成的结的周围。然而采用例如化学机械抛光(CMP)后的蚀刻处理剥离层230。这除去了含有提取自层200的杂质的层230。最后在n-掺杂层220上设置银接触240,并在层200上提供铝电极250。然后在700℃左右使整个结构体退火,使银电极对n-型层210以及使铝电极250对p-型层200完成低电阻的欧姆接触。Figure 2 illustrates a process according to one embodiment of the invention. The process starts with a
图3说明根据本发明另一个实施方式的工艺。该工艺开始于步骤100,从含冶金硅的坩埚中升华2μm厚的非晶硅层。这将在约10E-6托的Ar背景真空度下在低于硅熔点的1000~1200℃下进行。该步骤确定较纯的有源结层,因为在1200℃下任何碳和金属杂质不升华。此外,在氩气背景环境中少量的残余氧通过在坩埚表面上形成少量一氧化硅帮助促进升华。在步骤200,在O2加N2或Ar的环境下将晶片暴露于含P气体如POC13或PBr3。该步骤通过掺杂n-型面并使B得以从“脏的”基板向外扩散到干净的升华后的硅层中来而形成p-n结。Figure 3 illustrates a process according to another embodiment of the invention. The process begins at step 100 with the sublimation of a 2 μm thick layer of amorphous silicon from a crucible containing metallurgical silicon. This will be done at 1000-1200°C below the melting point of silicon with an Ar background vacuum of about 10E-6 Torr. This step identifies a purer active junction layer since any carbon and metal impurities do not sublime at 1200°C. In addition, the small amount of residual oxygen in the argon background environment helps to promote sublimation by forming a small amount of silicon monoxide on the crucible surface. In
然而在步骤300中该晶片的背面进行蚀刻或CMP(化学机械抛光)以除去任何掺杂磷的玻璃。其次,在步骤400中,等离子用于在该晶片的正面(n-型)沉积SiN抗反射膜。在步骤500中形成接触,例如,可用激光来为接触钻孔。在步骤600,制造导电电极,例如,可以采用丝网或其它方法在正面和背面沉积金属糊以限定电极。然后在600℃~700℃下烧结晶片以形成接触。在通过丝网将银沉积在SiN抗反射膜上而没有穿过该层的任何激光钻孔的接触孔的情况下,利用较高的温度来使银得以穿过整个SiN层。However in
图4说明根据本发明一个实施方式的工艺。该工艺从脏的p-型硅晶片400开始。然后利用蒸发过程在晶片400上产生蒸发的SiOx层410。在SiOx层410上提供n-型层420(这可以通过沉积掺杂磷的Si层或通过其它可接受的方法部分扩散到该层410中来进行)。然后用层430密封层410,利用吸杂(gettering)将杂质向晶片400的底部440牵引以提供纯度提高的层400。然后可以在沉积导线之前除去该底部440。Figure 4 illustrates a process according to one embodiment of the invention. The process starts with a dirty p-
根据又一个实施方式,首先蚀刻脏的硅晶片以在其顶面上提供织构。然后在POC13炉中处理该晶片以形成该晶片的p-n结。用等离子沉积的SiN层覆盖该晶片的顶面。然后将该晶片再次暴露于POC13以将全部金属杂质收到(getter)背面,并使该结硬化避免漏出。然后通过例如背面蚀刻除去晶片背面上的玻璃。然后利用例如激光打孔或接触蚀刻形成接触孔。然后利用常规的技术形成金属接触。不然的话,直接在等离子沉积的氮化物层上形成丝网的银糊,然后在~700℃下退火,使银扩散到靠近晶片顶部的掺杂磷的层,而不使用任何接触孔。According to yet another embodiment, the dirty silicon wafer is first etched to provide texture on its top surface. The wafer was then processed in a POCl 3 furnace to form the pn junction of the wafer. The top surface of the wafer was covered with a plasma deposited SiN layer. The wafer is then exposed again to POC1 3 to getter all metal impurities on the backside and harden the junction from leakage. The glass on the backside of the wafer is then removed by, for example, backside etching. Contact holes are then formed using, for example, laser drilling or contact etching. Metal contacts are then formed using conventional techniques. Otherwise, a screened silver paste is formed directly on the plasma deposited nitride layer and then annealed at ~700°C to diffuse the silver into the phosphorous doped layer near the top of the wafer without using any contact holes.
值得注意的是,一旦形成冶金Si p-n结,由于在结界面中的金属杂质,极易漏出。POCl3的一个效果是形成n-层,将杂质牵引到形成结的表面附近。因此,为了从正面的结中将金属杂质移到晶片的背面,在该实施方式中执行第二POC13步骤,同时用SiN保护有源的正面。可以使金属集中在晶片背面上的低温熔融的玻璃中,然后通过化学蚀刻或CMP除去。It is worth noting that once a metallurgical Si pn junction is formed, it is very easy to leak out due to metal impurities in the junction interface. One effect of POCl3 is to form an n-layer that pulls impurities near the surface where the junction is formed. Therefore, in order to move metal impurities from the junction on the front side to the back side of the wafer, a second POC1 3 step is performed in this embodiment while protecting the active front side with SiN. The metal can be concentrated in the low temperature molten glass on the backside of the wafer and then removed by chemical etching or CMP.
代替硅基板,人们可以使用由涂覆有升华Si的不锈钢或玻璃制成的基板,并通过从离心浇铸的B、P玻璃中扩散在该基板中形成p-n结。这不同于用于薄膜晶体管平板应用而沉积的非晶PECVD硅,因为升华的膜不具有任何被捕获的氢。因此,在随后的高温扩散步骤时,它们不分解。PECVD膜随时间失效,可能因为与H解吸有关的组成变化。Instead of a silicon substrate, one can use a substrate made of stainless steel or glass coated with sublimated Si and form a p-n junction in this substrate by diffusion from centrifugally cast B, P glass. This differs from amorphous PECVD silicon deposited for thin film transistor flat panel applications, as the sublimated film does not have any trapped hydrogen. Therefore, they do not decompose during the subsequent high temperature diffusion step. PECVD films fail over time, probably because of compositional changes related to H desorption.
图5说明本发明的另一个实施方式。用于图5的实施方式的原料是通过浇铸,随后缓慢冷却制造的纯度为约四个9到五个9即99.99%到99.999%的低成本多晶冶金硅晶片。冶金硅简便地由石英(SiO2)与石墨(C)基于熔炉的化学反应来制造,二者存在于世界各地的矿山中。这两种材料基本上是砂和煤的较纯形式。石墨能够用其它包含纯C的石油副产物或有机植物物质来代替。使冶金硅粉熔融,根据需要向熔体中添加测试量的B以产生约5E17原子*cm-3的p-型掺杂剂浓度。使熔体缓慢冷却以生产含硅的多晶晶粒的圆柱形铸锭,含约10ppm的杂质例如Cr、Fe、Ni、Mn和C。调节冷却过程以便使杂质分布在所谓沉淀物的原子簇中。它们趋向电活性小于均匀分布原子,后者典型地占据硅晶体晶格上取代的电活性位点。取代的杂质起到用于电子空穴复合的陷阱或中心的作用,该复合被认为通过降低光吸收层中载流子的扩散长度而降低了太阳能电池的光伏转换效率。通过众所周知的测试与光的波长成函数关系的光转换为电荷载流子的量子效率的物理法能够估算扩散长度。将铸造的材料,即铸锭加工成较小的圆柱体、锯成晶片、蚀刻以除去表面损伤,然后根据标准的行业操作在一面或两面上抛光。这将会得到的冶金级晶片用作生产太阳能电池的基板。不象常规的多晶硅太阳能电池所用的硅基板,该实施方式不需要使用通常由气相SiHxCly化合物还原制造的七个9或更高纯度的多晶硅。Figure 5 illustrates another embodiment of the invention. The feedstock for the embodiment of FIG. 5 is low cost polycrystalline metallurgical silicon wafers of about four nines to five nines or 99.99% to 99.999% purity manufactured by casting followed by slow cooling. Metallurgical silicon is simply produced by the furnace-based chemical reaction of quartz (SiO 2 ) and graphite (C), both found in mines around the world. These two materials are basically the purer forms of sand and coal. Graphite can be replaced with other petroleum by-products or organic plant matter containing pure C. Metallurgical silicon powder was melted and measured amounts of B were added to the melt as needed to produce a p-type dopant concentration of about 5E17 atoms*cm −3 . The melt was cooled slowly to produce a cylindrical ingot of polycrystalline grains containing silicon with about 10 ppm of impurities such as Cr, Fe, Ni, Mn and C. The cooling process is adjusted so that the impurities are distributed in atomic clusters called precipitates. They tend to be less electroactive than the uniform distribution of atoms that typically occupy substituted electroactive sites on the silicon crystal lattice. The substituted impurities act as traps or centers for electron-hole recombination, which is believed to reduce the photovoltaic conversion efficiency of solar cells by reducing the diffusion length of carriers in the light-absorbing layer. The diffusion length can be estimated by the well-known physical method of measuring the quantum efficiency of the conversion of light to charge carriers as a function of the wavelength of the light. The cast material, the ingot, is machined into smaller cylinders, sawn into wafers, etched to remove surface damage, and then polished on one or both sides according to standard industry practice. The resulting metallurgical-grade wafers are used as substrates for the production of solar cells. Unlike the silicon substrates used for conventional polysilicon solar cells, this embodiment does not require the use of seven nines or higher purity polysilicon typically produced by reduction of gas phase SiHxCly compounds.
基板500经过预沉积清洗,典型地涉及用100:1HF除去任何天然的氧化物,用NH4OH/H2O2除去有机污染物,然后用HCl除去任何金属杂质。然后在没有掺杂气存在的情况下利用在SiH4和H2中产生的等离子在标准的PECVD设备中涂覆极薄()的本征、未掺杂的非晶Si:H层505,典型地低于。接下来,通过沉积n-掺杂的a-Si:H层510形成结的有源部件,这可以方便地在相同装置中进行,但利用含SiH4和H2以及PH3的等离子。这随后是透明的导电氧化物520例如ZnO2或InSnO以及如果需要由SiOxNy构成的抗反射膜515的连续层。这些形成顶电极,通过该电极能够将日光传输到硅吸收层本体。为了额外的电荷收集效率,可以在该透明的导电氧化物层520上形成典型地由银糊构成的一系列电极。对于电池结构背面的低电阻接触,通过PVD工艺沉积或丝网印刷含Al的糊在晶片的底面涂覆Al层525,然后烧结以形成低电阻接触。
于是得到的太阳能电池包含至少下列新特征。通过在由成本比利用太阳能或半导体级多晶硅制造的常规硅晶片少约十倍的p-型多晶冶金级硅晶片制成的吸收晶片上沉积非晶Si:H薄膜的n-层来形成p-n结。由铸造含B掺杂剂的冶金硅粉制造的250~500μm厚的冶金p-型多晶硅光吸收层代替采用贵得多的太阳能级多晶硅。在p-型冶金基板和n-Si:H膜之间插入任选的本征(无掺杂的)Si:H膜中间层以使由于多晶性质和材料中的杂质典型地具有断裂(悬挂)键的冶金硅的表面钝化,从而改进光伏转换效率。为了节约成本可以省略ARC层515,代之以通过在KOH中蚀刻冶金级硅的表面使其粗糙以在大体上(100)取向的晶粒中暴露(111)面。该粗糙化使光反射最小化,以致ARC层可以是不必要的。The solar cell thus obtained contains at least the following new features. The p-n is formed by depositing an n-layer of amorphous Si:H thin film on an absorber wafer made of p-type polycrystalline metallurgical grade silicon wafers that cost about ten times less than conventional silicon wafers made with solar or semiconductor grade polysilicon Knot. The 250-500 μm thick metallurgical p-type polysilicon light absorbing layer produced by casting metallurgical silicon powder containing B dopant replaces the much more expensive solar-grade polysilicon. An optional intrinsic (undoped) Si:H film interlayer is inserted between the p-type metallurgical substrate and the n-Si:H film to typically have fracture (hanging) due to the polycrystalline nature and impurities in the material. ) bond to the surface passivation of metallurgical silicon, thereby improving photovoltaic conversion efficiency. The
图6说明本发明的另一个实施方式。图6的实施方式类似于图5,除了将掺杂颠倒。也就是说,将吸收层600制造为n-型冶金硅。沉积的非晶层610具有反极性,即用于结的p-型。Figure 6 illustrates another embodiment of the present invention. The embodiment of Fig. 6 is similar to Fig. 5 except that the doping is reversed. That is,
图7说明本发明的另一个实施方式。图7的实施方式类似于图5。然而,在图7的实施方式中,在制造背面接触的铝层725之前制造任选的由a-i Si:H膜730随后a-n Si:H膜735构成的结构,以便利用异质结提高高于吸收基板的转换效率,该异质结具有含沉积的极薄本征Si-H层的本征钝化层结构,继之以反极性的电学上有源的Si-H薄层。在这方面,对于图5~8所述的实施方式,用带括号的字母表示对于各说明的层建议的制造顺序。图8说明本发明的另一个实施方式,其类似于图7的实施方式,除了将掺杂剂颠倒。也就是说,基板被指定为n-型冶金级硅,结层810是p-型,以及层835是n-型。Figure 7 illustrates another embodiment of the present invention. The embodiment of FIG. 7 is similar to FIG. 5 . However, in the embodiment of FIG. 7, the optional structure consisting of a-i Si:
如同能够被理解的那样,图5~8的实施方式通过在冶金级硅基板上构建薄膜结来提供太阳能电池。由于冶金硅的性能,与具有极薄吸收层的常规薄膜电池相比,这具有光吸收较好的优点。因此,提高了转换效率。另一方面,使用冶金硅片提供低于常规的太阳能或半导体级硅晶片的成本。此外,如本文所述通过使用冶金级硅晶片,降低了健康和环境危害。As can be appreciated, the embodiments of Figures 5-8 provide solar cells by building thin film junctions on metallurgical grade silicon substrates. Due to the properties of metallurgical silicon, this has the advantage of better light absorption compared to conventional thin-film cells with extremely thin absorber layers. Therefore, conversion efficiency is improved. On the other hand, the use of metallurgical silicon wafers offers lower cost than conventional solar or semiconductor grade silicon wafers. Furthermore, by using metallurgical grade silicon wafers as described herein, health and environmental hazards are reduced.
涉及图5~8的实施方式的意图是区分与PV工艺联系的三个功能,该工艺首先在硅中吸收光以产生电子空穴对,然后通过利用p-n结的带隙产生少数载流子(电子)流,把光转换为电流。通常,在具有扩散的p-n结结构的多晶或单晶硅中,两个过程同时发生。当从常规的多晶硅到Czochralsky单晶硅到区域熔融的单晶硅时,少数载流子扩散长度可以从50μm到100μm到300μm变化。相应的PV转换效率为约18%、22%和25%。在另一端,沉积非晶单结的薄膜太阳电池依靠典型地约1μm厚的中间aSi:H层为吸收层。扩散长度被薄膜层的厚度限制到约1μm。相应的PV转换效率降低到约6%。在本发明的该实施方式中,少数载流子扩散长度不受薄膜的限制,而是由冶金硅基板的特性来决定。The intent of the embodiments referring to Figures 5-8 is to distinguish three functions associated with the PV process, which first absorbs light in silicon to generate electron-hole pairs, and then generates minority carriers by exploiting the bandgap of the p-n junction ( Electron) flow, which converts light into electric current. Usually, in polycrystalline or monocrystalline silicon with a diffused p-n junction structure, both processes occur simultaneously. The minority carrier diffusion length can vary from 50 μm to 100 μm to 300 μm when going from conventional polysilicon to Czochralsky single crystal silicon to zone fused single crystal silicon. The corresponding PV conversion efficiencies are about 18%, 22% and 25%. At the other end, deposited amorphous single-junction thin-film solar cells rely on an intermediate aSi:H layer typically about 1 μm thick as the absorber layer. The diffusion length is limited to about 1 μm by the thickness of the thin film layer. The corresponding PV conversion efficiency is reduced to about 6%. In this embodiment of the invention, the minority carrier diffusion length is not limited by the thin film, but is determined by the properties of the metallurgical silicon substrate.
实施例1Example 1
通过在约1.5m×1.5m的石墨坩埚中感应熔融两个九的硅颗粒,然后经24小时缓慢冷却成圆柱形来生产三个九的冶金级硅。除去富碳的表面壳层,并将圆柱体粉碎为晶粒或颗粒。得到的材料含B和P,但通常是具有在0.1~1ohm·cm范围内的电阻率的p-型。然后将得到的材料铸成约0.5m×1m的冶金级硅铸锭,伴随着控制冷却和掺杂剂调节。从铸锭中加工出6英寸的芯,平整圆柱体表面,然后从圆柱体上锯断500μm厚的晶片,制造冶金级硅晶片。机械抛光一个面,轻微蚀刻两个面以露出在晶片背面上的多边形大晶粒结构。这产生四个九和五个九纯度的约500片冶金级硅晶片。利用4点探针测量将晶片分为两组,主要的组具有0.3~0.5ohm·cm的电阻率,其余的在~1ohm·cm。4N和5N材料的SIMS组成分布类似,具有IE14原子cm-3的过渡金属杂质浓度。金属杂质典型地是与冶金硅有关的那些,即Fe、Cr、Mn、Co、Ni、Cu。另外,存在IE15原子cm-3的碳浓度。Three nines of metallurgical grade silicon was produced by induction melting two nines of silicon pellets in an approximately 1.5 m x 1.5 m graphite crucible, followed by slow cooling into a cylindrical shape over 24 hours. The carbon-rich surface shell is removed and the cylinders are crushed into grains or granules. The resulting material contains B and P, but is usually p-type with a resistivity in the range of 0.1-1 ohm·cm. The resulting material was then cast into approximately 0.5m x 1m ingots of metallurgical grade silicon with controlled cooling and dopant adjustment. A 6-inch core is machined from the ingot, the surface of the cylinder is flattened, and a 500 μm thick wafer is sawn from the cylinder to produce metallurgical-grade silicon wafers. One side is mechanically polished and both sides are lightly etched to reveal the polygonal large grain structure on the backside of the wafer. This yielded approximately 500 wafers of metallurgical-grade silicon of four-nine and five-nine purity. The wafers were divided into two groups using 4-point probe measurements, the main group having a resistivity of 0.3-0.5 ohm·cm and the remainder at ~1 ohm·cm. The SIMS composition profiles of the 4N and 5N materials are similar, with transition metal impurity concentrations of IE14 atoms cm −3 . Metallic impurities are typically those associated with metallurgical silicon, ie Fe, Cr, Mn, Co, Ni, Cu. In addition, there is a carbon concentration of IE15 atom cm −3 .
用晶片的样品制造太阳能电池。在含适当的掺杂气PH3和B2H6的SiH4、H2中利用rf等离子,用PECVD(等离子体增强化学气相沉积)设备沉积i型a-Si:H薄膜、p型a-Si:H和n型a-Si:H膜。采用PVD(等离子气相沉积)溅射设备沉积约的InxSnyOz,用作用于顶电极和底电极的透明导电氧化物。采用蚀刻设备蚀刻约10μm深的硅台地(silicon mesas)以产生与晶片其余部分隔离的二极管。利用该工艺,从0.1Ω·cm p-型(100)冶金级硅晶片开始,产生具有含扩散的p+背接触的本征钝化层结构的单异质结,并测量该结的二极管I-V和穿过该光谱范围的量子效率。利用1/QE对照λ波长的曲线,斜率按μm提供扩散长度L。长度L和IDsat是众所周知的PV转换效率的预测因子。该结构提供400mA的IDsat和80μm的长度L,对应于约20%的PV转换效率。在0.4Ω·cm p-型冶金级硅晶片上形成的结构也工作得很好,具有7μm的少数载流子(电子)扩散长度Le,对应于12~13%的PV转换效率,假定结构具有良好控制的串联电阻。在1.0Ω·cm p-型冶金级硅晶片上形成的结构也工作得很好,具有8μm的少数载流子(电子)扩散长度Le,对应于14%的PV转换效率,假定结构具有良好控制的串联电阻。A sample of the wafer is used to fabricate solar cells. Using rf plasma in SiH 4 and H 2 containing appropriate dopant gas PH 3 and B 2 H 6 , use PECVD (plasma enhanced chemical vapor deposition) equipment to deposit i-type a-Si:H film, p-type a- Si:H and n-type a-Si:H films. Using PVD (Plasma Vapor Deposition) sputtering equipment to deposit about In x Sny O z , used as the transparent conductive oxide for the top and bottom electrodes. Silicon mesas are etched approximately 10 μm deep using an etch tool to create diodes isolated from the rest of the wafer. Using this process, starting from a 0.1 Ω cm p-type (100) metallurgical grade silicon wafer, a single heterojunction with an intrinsic passivation layer structure with a diffused p+ back contact was produced and the diode IV and Quantum efficiency across this spectral range. Using a plot of 1/QE versus lambda wavelength, the slope provides the diffusion length L in μm. The lengths L and I Dsat are well known predictors of PV conversion efficiency. This structure provides an I Dsat of 400 mA and a length L of 80 μm, corresponding to a PV conversion efficiency of about 20%. Structures formed on 0.4Ω·cm p-type metallurgical grade silicon wafers also work well, with a minority carrier (electron) diffusion length L e of 7 μm, corresponding to a PV conversion efficiency of 12-13%, assuming the structure with well-controlled series resistance. The structure formed on a 1.0 Ω cm p-type metallurgical grade silicon wafer also works well, with a minority carrier (electron) diffusion length L e of 8 μm, corresponding to a PV conversion efficiency of 14%, assuming the structure has a good controlled series resistance.
实施例2Example 2
通过在正面即“器件”侧沉积纳米级Si:H膜堆栈并在背面“接触”侧沉积相反掺杂的a-Si:H膜,在低成本的冶金级基板上形成含本征钝化层器件结构的单异质结。冶金级基板不必如对结晶Si基板所做的那样将基板特别地从500μm变薄到250μm,免除了损耗。较厚的晶片在自动生产线中提供更稳固的操作。该材料还避免了基于多晶硅的气化、凝固、熔融和提拉工艺的成本、周期和复杂性,因为通过恰好在被纳米级本征a-Si:H膜钝化的冶金级基板面之外的薄Si:H膜来产生该有源器件。Formation of intrinsic passivation layers on low-cost metallurgical-grade substrates by depositing nanoscale Si:H film stacks on the front, “device” side and oppositely doped a-Si:H films on the back, “contact” side Single heterojunction for device structures. Metallurgical grade substrates do not have to specifically thin the substrate from 500 μm to 250 μm as is done for crystalline Si substrates, eliminating losses. Thicker wafers provide more robust handling in automated production lines. The material also avoids the cost, cycle, and complexity of polysilicon-based gasification, solidification, melting, and pulling processes, as it passes just outside the metallurgical-grade substrate face passivated by nanoscale intrinsic a-Si:H films thin Si:H films to create the active device.
冶金级基板可以按标准的尺寸例如6英寸、8英寸、12英寸来形成,可以在标准的半导体PECVD加工设备中进行加工。相反,在大面积(通常,4×6ft或6×7ft)的玻璃上产生常规的基于薄膜的太阳能电池,这需要专门的内部容积大的腔室,导致难以抽到低压并导致用于形成薄膜层的活性气体浪费。因此,这些PECVD反应器购买昂贵并且由于消耗品(即,废弃的活性气体)成本高运转昂贵。这些专门的腔室的高内部容积还造成折旧困难及成本。反之,在标准尺寸的晶片上形成薄膜能够在内部容积小的标准反应器中进行,以致运转和折旧的问题最小。由于在冶金硅基板中较长的少数载流子扩散长度的数量级,在冶金硅基板上得到的薄膜器件结构具有大于常规的薄膜太阳电池约两倍的PV效率。Metallurgical grade substrates can be formed in standard sizes such as 6 inches, 8 inches, and 12 inches, and can be processed in standard semiconductor PECVD processing equipment. In contrast, conventional thin-film based solar cells are produced on large areas (typically, 4×6ft or 6×7ft) of glass, which requires specialized chambers with large internal volumes, making it difficult to pump low pressures and resulting in Layers of reactive gas are wasted. Consequently, these PECVD reactors are expensive to purchase and expensive to operate due to the high cost of consumables (ie, spent reactive gases). The high internal volume of these specialized chambers also creates depreciation difficulties and costs. Conversely, thin film formation on standard sized wafers can be performed in standard reactors with small internal volumes so that handling and depreciation problems are minimized. Due to the order of magnitude longer minority carrier diffusion lengths in metallurgical silicon substrates, the resulting thin film device structures on metallurgical silicon substrates have PV efficiencies approximately twice greater than conventional thin film solar cells.
实施例3Example 3
图9A说明通常本文中称为SmartSiTM的用于制造准备好的太阳能电池基板的工艺的一个实施例。在步骤900中,在含石墨电极的电解槽中将冶金级石英熔融并还原,然后让其冷却并凝固以提供约两个九的冶金硅铸锭。将铸锭破碎成颗粒,在化学品中处理以浸出表面杂质,然后铸成铸锭。然后剥离铸锭的外壳并破碎成三个到四个九的冶金硅块。得到的块按照它们的电阻率分类。Figure 9A illustrates one embodiment of a process for fabricating a prepared solar cell substrate, generally referred to herein as SmartSi ™ . In step 900, metallurgical grade quartz is melted and reduced in an electrolytic cell containing graphite electrodes, then allowed to cool and solidify to provide a metallurgical silicon ingot of about two nines. The ingot is broken into pellets, treated in chemicals to leach surface impurities, and cast into ingots. The ingot is then peeled off and broken into metallurgical silicon blocks of three to four nines. The resulting blocks are sorted according to their resistivity.
在步骤915中对分类的MG硅块进行铸造。使熔体凝固成铸锭,在步骤920中加工,切成晶片,并将晶片抛光。在步骤925中,用PECVD室来形成本征非晶硅薄层i-a-Si:H以钝化MG-Si基板的表面。在步骤930中,用PECVD室在钝化层上形成n-型层n-a-Si:H。此时,已经产生了“SmartSi”晶片935,使得能够在世界上无论何处用很少的投资、较少量的简单机械和极少的技术知识实际上形成PV太阳能电池工业。也就是说,如同能够被理解的那样,为了将SmartSi晶片转换为太阳能电池所需要的一切是制造正面和背面的接触,也许还有抗反射层和保护层。这采用现有的丝网印刷或印刷技术能够容易地进行。此外,如标注所示,可以执行PECVD的另一个步骤930'以在基板的背面形成p-型后面(later)935',以便改进对随后的导电层的接触。In step 915 the sorted MG silicon ingots are cast. The melt is solidified into an ingot, processed in step 920, sliced into wafers, and the wafers are polished. In
图9B说明可用于将SmartSi晶片转换为SmartSi PV太阳能电池的工艺的一个实施例。如上所述,所需要的是在SmartSi基板的背面和正面中形成接触。至于正面,一种常规方法是形成导电的金属网格。通常的方法是设计具有散布到电池表面每个部分的许多薄的导电触头的网格。网格的触头必须足够宽以良好导电(具有低电阻),但要足够窄以免阻断大量入射光。这种网格保持低的电阻损耗,同时仅遮蔽电池表面的约3%到5%。顶面网格可以由例如铝、银或钼金属构成,通过在电池上经掩模沉积金属蒸气、通过网印刷法在电池上涂抹它们,或者用提供最高的性能但成本最高的光刻法。Figure 9B illustrates one embodiment of a process that can be used to convert SmartSi wafers to SmartSi PV solar cells. As mentioned above, all that is required is to form contacts in the back and front sides of the SmartSi substrate. As for the front side, a conventional method is to form a conductive metal grid. The usual approach is to design a grid with many thin conductive contacts scattered to every part of the battery's surface. The mesh's contacts must be wide enough to conduct electricity well (with low resistance), but narrow enough not to block much of the incoming light. This mesh maintains low resistive losses while shading only about 3 to 5 percent of the cell's surface. The top grid can be made of eg aluminum, silver or molybdenum metal by depositing metal vapors through a mask on the cell, painting them on the cell by screen printing, or by photolithography which offers the highest performance but the highest cost.
可替代金属网格接触的是透明的导电氧化物(TCO)层,例如氧化锡(SnO2)或通常被称为ITO的氧化铟锡。TCO的优点在于它们对入射光几乎是不可见的,并且它们形成从半导体材料到外部电路的良好桥接。图9B中所示的实施方式利用TCO作为对电池正面的接触。在步骤940中,利用CVD工艺形成TCO层。在步骤945中采用丝网、印刷等在正面上利用例如微量描绘的金属糊将正面接触金属化。在步骤950中,采用丝网、印刷等利用在背面上描绘的例如金属糊(例如,银糊)或者通过在基板的背面上溅射铝或其它金属使背面接触金属化以形成集电极。当用糊将正面接触金属化时,如步骤955所示,为了形成良好的欧姆接触烧结该晶片是合乎需要的。在步骤960中,将晶片切成所希望的形状,例如,如果用圆形晶片进行加工,在该步骤可以将它们切成正方形。然后根据转换效率将晶片分类,以便产生SmartSi PV电池970。An alternative to the metal grid contact is a transparent conductive oxide (TCO) layer such as tin oxide (SnO 2 ) or indium tin oxide, commonly known as ITO. The advantage of TCOs is that they are nearly invisible to incident light, and they form a good bridge from the semiconductor material to the external circuitry. The embodiment shown in Figure 9B utilizes a TCO as the contact to the front side of the cell. In step 940, a TCO layer is formed using a CVD process. The front side contacts are metallized in step 945 with, for example, trace traced metal paste on the front side using screen, printing or the like. In
在上述所有实施方式中,在形成任何层之前,可以通过例如在碱性溶液如氢氧化钾溶液使MG Si基板蚀刻而使得其一个或两个面被织构化。然后可以冲洗该基板,并通过例如加热该基板使其干燥。此外,可以用氢气的等离子体放电来减少基板表面上的碳量。可以在PECVD室中采用与氢气(H2)混合的硅烷气(SiH4)来形成本征非晶硅薄膜层。可以在PECVD室中采用硅烷、氢气和磷化氢气体(PH3)来形成n-型非晶硅薄层。可以在PECVD室中采用硅烷、氢气和硼乙烷气体(B2H6)来形成p-型非晶硅薄层。In all of the embodiments described above, one or both faces of the MG Si substrate may be textured by, for example, etching in an alkaline solution such as potassium hydroxide solution, before any layers are formed. The substrate can then be rinsed and dried, for example by heating the substrate. In addition, a plasma discharge of hydrogen can be used to reduce the amount of carbon on the substrate surface. The intrinsic amorphous silicon thin film layer can be formed using silane gas (SiH 4 ) mixed with hydrogen gas (H 2 ) in a PECVD chamber. A thin n-type amorphous silicon layer can be formed using silane, hydrogen and phosphine gas (PH 3 ) in a PECVD chamber. A thin layer of p-type amorphous silicon can be formed using silane, hydrogen, and boroethane gas (B 2 H 6 ) in a PECVD chamber.
图10说明根据本发明的实施方式制造的完整的太阳能电池。在冶金级硅基板1000上形成太阳能电池,该基板在本实施例中是掺杂的p-型。然后在顶面形成本征非晶硅层1005,随后形成n-型非晶硅层1010。在n-型层上形成TCO层1020,并且在TCO上形成接触例如银接触1025以形成良好的欧姆接触。可以用例如铝形成背接触。此时电池完成并可供使用;然而,为了使它免受元素影响,进行下列进一步的加工。正面受到任选的树脂膜层1015的保护,例如乙烯-乙酸乙烯共聚物,随后是玻璃1045。背面也可以用树脂膜1035、随后玻璃或其它保护层1040来保护。Figure 10 illustrates a complete solar cell fabricated in accordance with an embodiment of the invention. The solar cell is formed on a metallurgical
如图9A和9B所示,可以用上面讨论的实施方式来制造SmartSi晶片,可以将晶片进一步加工以制造SmartSi太阳能电池。根据本发明的另一个方面,为了提高光伏转换效率,可以进一步加工SmartSi太阳能电池来制造具有多个带隙的多结SmartSi太阳能电池。在图11A中说明多结SmartSi太阳能电池的一个实施方式。在图11A中,冶金级硅基板1100是掺杂的p-型。该p-型基板的顶面被本征非晶硅薄层1105钝化,该非晶硅具有分散其中并占据硅悬挂键的氢原子。这有时被称为氢化硅。如上述SmartSi太阳能电池实施方式中所示,在本征层1105上形成n-型非晶氢化硅的薄层1110,从而形成第一p-i-n结。本征和n-型层1105和1110比典型的常规薄膜太阳电池的薄膜层相对薄得多,在本实施方式中,第一薄膜结构不必起光吸收体的作用,而是在冶金硅基板中吸收光。As shown in Figures 9A and 9B, the embodiments discussed above can be used to fabricate SmartSi wafers, which can be further processed to fabricate SmartSi solar cells. According to another aspect of the present invention, in order to improve the photovoltaic conversion efficiency, the SmartSi solar cell can be further processed to manufacture a multi-junction SmartSi solar cell with multiple bandgaps. One embodiment of a multi-junction SmartSi solar cell is illustrated in FIG. 11A . In FIG. 11A , the metallurgical grade silicon substrate 1100 is doped p-type. The top surface of the p-type substrate is passivated by a
为了提高SmartSi太阳能电池的转换效率,现在在SmartSi太阳能电池的上面形成常规的薄膜太阳电池p-i-n结构。首先,在SmartSi太阳能电池上形成薄膜p-型非晶氢化硅层1120。然后在p-型层1120上形成薄膜本征非晶氢化硅层1125,并在该本征层1125上形成薄膜n-型非晶氢化硅层1130。本征层1125起另一光吸收体的作用并产生电子空穴对,从而将光转换为电能。为了收集电能,在n-型层1130上形成顶透明电极ITO1135,然后在ITO1135上形成金属接触1140。这里金属接触1140由银制成,例如采用银糊,然后烧结该结构以形成良好的欧姆接触。另外,在基板1100的底部形成金属电极1145。这里接触1145用铝制成。图11B说明类似的多结结构,除了将层的极性颠倒。In order to improve the conversion efficiency of SmartSi solar cells, a conventional thin-film solar cell p-i-n structure is now formed on top of SmartSi solar cells. First, a thin p-type amorphous hydrogenated
图12A和12B说明具有扩散结的多结SmartSi太阳能电池的实施方式。图12A和12B的实施方式基本上相同,除了将层的极性颠倒。因此,仅对它们中的一种即图12A的实施方式进行描述。在图12A中,根据如上所述的实施方式制造冶金硅基板1200,并且其是掺杂的n-型。然后,使基板的顶层扩散以形成p-型扩散层1260。这在冶金硅基板中形成p-n结并提供太阳能电池的转换区域,类似于标准的硅基太阳能电池。然后在扩散的p-型层上形成本征非晶氢化硅的薄钝化层1205。在该本征层1205上形成n-型非晶氢化硅层1215,以致层1215、1205和1260形成具有不同于基板1200内p-n结的带隙的p-i-n结,因此以不同的频率吸收光。然后通过形成p-型非晶氢化硅层1220、本征非晶氢化硅层1225和n-型非晶氢化硅层1230在层1215上形成常规的薄膜p-i-n结。在该结构中,当本征层1225起光吸收体的作用时,它具有远高于本征层1205的厚度。此外,该薄膜p-i-n结构具有与其下的结构不同的带隙,因此以不同的频率吸收光。因此,通过仔细地选择层的厚度,人们能够“调整”该结构以在宽的频率范围吸收光。12A and 12B illustrate an embodiment of a multi-junction SmartSi solar cell with diffused junctions. The embodiments of Figures 12A and 12B are essentially the same except that the polarity of the layers is reversed. Therefore, only one of them, the embodiment of Fig. 12A, will be described. In Fig. 12A, a
图13A和13B说明两面夹在本征Si/掺杂的Si:H薄膜之间的冶金硅基板的对称配置的实施方式。图13A和13B是彼此的对映体,除了将层的极性颠倒。因此仅解释图13A。在图13A中,p-型冶金硅基板700具有作为钝化层而不是吸收体的上本征层705和下本征层730。然后在本征层705上形成n-型非晶氢化硅的薄层710,并在本征层730上形成另一个n-型层735。然后如相对于其它实施方式所述形成接触720和725。13A and 13B illustrate an embodiment of a symmetrical configuration of a metallurgical silicon substrate sandwiched on both sides between intrinsic Si/doped Si:H thin films. Figures 13A and 13B are antipodes of each other, except that the polarity of the layers is reversed. Therefore only FIG. 13A is explained. In FIG. 13A, a p-type
应理解本文所述的工艺和技术不是固有地与任何装置相关,并且可以通过任何适合的组件的组合来实施。此外,根据本文所述的教导可以采用各种类型的通用设备。还可以证明构建执行本文所述的方法步骤的专用装置是有利的。已经相对于具体的实施例描述了本发明,从所有的方面意图是说明性的而非限制性的。本领域技术人员将理解硬件、软件和固件的许多不同组合将适于实施本发明。It should be understood that the processes and techniques described herein are not inherently related to any device, and may be implemented by any suitable combination of components. In addition, various types of general-purpose devices may be employed in accordance with the teachings described herein. It may also prove advantageous to construct dedicated apparatus for carrying out the method steps described herein. The present invention has been described with respect to specific embodiments, which are intended in all respects to be illustrative and not restrictive. Those skilled in the art will appreciate that many different combinations of hardware, software, and firmware will be suitable for implementing the invention.
已经相对于特别的实施例描述了本发明,从所有的方面意图是说明性的而非限制性的。本领域技术人员将理解硬件、软件和固件的许多不同组合将适于实施本发明。此外,对于本领域技术人员,考虑了本文公开的本发明的说明和实施,本发明的其它实施将是显而易见的。意图将说明书和实施例仅当作是示例性的,通过下列权利要求表明本发明的真实范围和实质。The invention has been described with respect to particular embodiments, which are intended in all respects to be illustrative and not restrictive. Those skilled in the art will appreciate that many different combinations of hardware, software, and firmware will be suitable for implementing the invention. Furthermore, other implementations of the invention will be apparent to those skilled in the art from consideration of the specification and practice of the invention disclosed herein. It is intended that the specification and examples be considered exemplary only, with a true scope and spirit of the invention indicated by the following claims.
Claims (20)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US98699607P | 2007-11-09 | 2007-11-09 | |
| US60/986,996 | 2007-11-09 |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008801154349A Division CN101960618B (en) | 2007-11-09 | 2008-11-07 | Low-cost solar cells and methods for their production |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN103296138A true CN103296138A (en) | 2013-09-11 |
Family
ID=40326264
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2013100950349A Pending CN103296138A (en) | 2007-11-09 | 2008-11-07 | Low-cost solar cells and methods for their production |
| CN2008801154349A Expired - Fee Related CN101960618B (en) | 2007-11-09 | 2008-11-07 | Low-cost solar cells and methods for their production |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008801154349A Expired - Fee Related CN101960618B (en) | 2007-11-09 | 2008-11-07 | Low-cost solar cells and methods for their production |
Country Status (7)
| Country | Link |
|---|---|
| US (4) | US7956283B2 (en) |
| EP (2) | EP2618387A1 (en) |
| KR (1) | KR101293162B1 (en) |
| CN (2) | CN103296138A (en) |
| ES (1) | ES2422256T3 (en) |
| PT (1) | PT2208238E (en) |
| WO (1) | WO2009062117A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104505429A (en) * | 2014-11-21 | 2015-04-08 | 广东爱康太阳能科技有限公司 | Photolithographic process applied to crystalline silicon solar cells |
Families Citing this family (56)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103296138A (en) * | 2007-11-09 | 2013-09-11 | 森普雷姆有限公司 | Low-cost solar cells and methods for their production |
| US8377734B2 (en) * | 2008-12-02 | 2013-02-19 | Mitsubishi Electric Corporation | Method for manufacturing solar battery cell |
| US20100206367A1 (en) * | 2009-02-18 | 2010-08-19 | Korea Institute Of Industrial Technology | Method for fabricating silicon nano wire, solar cell including silicon nano wire and method for fabricating solar cell |
| US20100279492A1 (en) * | 2009-05-02 | 2010-11-04 | Atomic Energy Council-Institute Of Nuclear Energy Research | Method of Fabricating Upgraded Metallurgical Grade Silicon by External Gettering Procedure |
| US20110070744A1 (en) * | 2009-09-18 | 2011-03-24 | Zhi-Wen Sun | Silicon Texturing Formulations for Solar Applications |
| US9012766B2 (en) | 2009-11-12 | 2015-04-21 | Silevo, Inc. | Aluminum grid as backside conductor on epitaxial silicon thin film solar cells |
| KR101228140B1 (en) * | 2009-11-27 | 2013-01-31 | 엘지전자 주식회사 | Solar Cell |
| US20110126877A1 (en) * | 2009-11-27 | 2011-06-02 | Jinah Kim | Solar cell |
| US8324015B2 (en) * | 2009-12-01 | 2012-12-04 | Sunpower Corporation | Solar cell contact formation using laser ablation |
| US20110186119A1 (en) * | 2009-12-24 | 2011-08-04 | Atwater Harry A | Light-trapping plasmonic back reflector design for solar cells |
| WO2011093967A2 (en) * | 2009-12-24 | 2011-08-04 | California Institute Of Technology | Light-trapping plasmonic back reflector design for solar cells |
| US9096437B2 (en) | 2010-03-08 | 2015-08-04 | William Marsh Rice University | Growth of graphene films from non-gaseous carbon sources |
| US9908282B2 (en) | 2010-05-25 | 2018-03-06 | Mossey Creek Technologies, Inc. | Method for producing a semiconductor using a vacuum furnace |
| WO2011150057A2 (en) * | 2010-05-25 | 2011-12-01 | Mossey Creek Solar, LLC | Method of producing a solar cell |
| US9214576B2 (en) | 2010-06-09 | 2015-12-15 | Solarcity Corporation | Transparent conducting oxide for photovoltaic devices |
| CN102386285B (en) * | 2010-08-24 | 2014-12-10 | 森普雷姆有限公司 | Low-cost solar cells and methods for fabricating low cost substrates for solar cells |
| US8207013B2 (en) * | 2010-09-17 | 2012-06-26 | Atomic Energy Council Institute Of Nuclear Energy Research | Method of fabricating silicon nanowire solar cell device having upgraded metallurgical grade silicon substrate |
| KR101134730B1 (en) * | 2011-01-25 | 2012-04-19 | 엘지이노텍 주식회사 | Solar cell apparatus and method of fabricating the same |
| US20120247543A1 (en) * | 2011-03-31 | 2012-10-04 | Integrated Photovoltaic, Inc. | Photovoltaic Structure |
| WO2013013138A1 (en) | 2011-07-20 | 2013-01-24 | Mossey Creek Solar, LLC | Substrate for use in preparing solar cells |
| JP5927027B2 (en) | 2011-10-05 | 2016-05-25 | 株式会社半導体エネルギー研究所 | Photoelectric conversion device |
| DE102011115028A1 (en) * | 2011-10-07 | 2013-04-11 | Ewe-Forschungszentrum Für Energietechnologie E. V. | Photovoltaic multiple solar cell |
| US20130125968A1 (en) * | 2011-11-18 | 2013-05-23 | Sunpreme, Ltd. | Low-cost solar cell metallization over tco and methods of their fabrication |
| US9543493B2 (en) | 2011-11-22 | 2017-01-10 | Mossey Creek Technologies, Inc. | Packaging for thermoelectric subcomponents |
| CN102605418A (en) * | 2012-01-16 | 2012-07-25 | 上澎太阳能科技(嘉兴)有限公司 | Solar cell substrate, manufacturing method of solar cell and crucible used for same |
| US20140020743A1 (en) * | 2012-07-23 | 2014-01-23 | E I Du Pont De Nemours And Company | Solar cell and manufacturing method thereof |
| CN102856189B (en) * | 2012-09-20 | 2015-09-09 | 苏州易益新能源科技有限公司 | A kind of crystal silicon chip surface-treated method |
| US20140099780A1 (en) | 2012-10-05 | 2014-04-10 | International Business Machines Corporation | Laser Doping of Crystalline Semiconductors Using a Dopant-Containing Amorphous Silicon Stack For Dopant Source and Passivation |
| US20140110264A1 (en) * | 2012-10-24 | 2014-04-24 | Atomic Energy Council-Institute of Nuclear Research | Light induced nickel plating method for p-type silicon and n/p solar cell material |
| US9219174B2 (en) | 2013-01-11 | 2015-12-22 | Solarcity Corporation | Module fabrication of solar cells with low resistivity electrodes |
| US10074755B2 (en) | 2013-01-11 | 2018-09-11 | Tesla, Inc. | High efficiency solar panel |
| US9412884B2 (en) | 2013-01-11 | 2016-08-09 | Solarcity Corporation | Module fabrication of solar cells with low resistivity electrodes |
| US20140305478A1 (en) | 2013-04-15 | 2014-10-16 | Mossey Creek Solar, LLC | Method for Producting a Thermoelectric Material |
| CN104253178A (en) * | 2013-06-26 | 2014-12-31 | 联景光电股份有限公司 | Solar cell and manufacturing method thereof |
| JP6350979B2 (en) * | 2013-09-04 | 2018-07-04 | パナソニックIpマネジメント株式会社 | Solar cell |
| US10403901B2 (en) * | 2014-02-11 | 2019-09-03 | Crown Battery Manufacturing Company | Silicon current collector for lead acid battery |
| US9783901B2 (en) | 2014-03-11 | 2017-10-10 | Macdermid Acumen, Inc. | Electroplating of metals on conductive oxide substrates |
| US20150280018A1 (en) * | 2014-03-26 | 2015-10-01 | Seung Bum Rim | Passivation of light-receiving surfaces of solar cells |
| US10309012B2 (en) | 2014-07-03 | 2019-06-04 | Tesla, Inc. | Wafer carrier for reducing contamination from carbon particles and outgassing |
| US9559236B2 (en) * | 2014-09-24 | 2017-01-31 | Sunpower Corporation | Solar cell fabricated by simplified deposition process |
| US9530921B2 (en) | 2014-10-02 | 2016-12-27 | International Business Machines Corporation | Multi-junction solar cell |
| CN104362220A (en) * | 2014-11-13 | 2015-02-18 | 京东方科技集团股份有限公司 | Method for manufacturing thin film solar cell and thin film solar cell |
| US9899546B2 (en) | 2014-12-05 | 2018-02-20 | Tesla, Inc. | Photovoltaic cells with electrodes adapted to house conductive paste |
| EP3038164B1 (en) * | 2014-12-22 | 2018-12-12 | Total S.A. | Opto-electronic device with textured surface and method of manufacturing thereof |
| US9842956B2 (en) * | 2015-12-21 | 2017-12-12 | Tesla, Inc. | System and method for mass-production of high-efficiency photovoltaic structures |
| TWI619262B (en) * | 2016-01-04 | 2018-03-21 | 有成精密股份有限公司 | High power solar module |
| CN105742379B (en) * | 2016-04-18 | 2017-06-30 | 浙江贝盛新能源开发有限公司 | A kind of crystal silicon solar energy battery and preparation method thereof |
| CN105870252B (en) * | 2016-04-18 | 2017-12-22 | 浙江贝盛新能源开发有限公司 | A kind of crystal silicon solar energy battery of high-absorbility and preparation method thereof |
| US10115838B2 (en) | 2016-04-19 | 2018-10-30 | Tesla, Inc. | Photovoltaic structures with interlocking busbars |
| US10672919B2 (en) | 2017-09-19 | 2020-06-02 | Tesla, Inc. | Moisture-resistant solar cells for solar roof tiles |
| US11190128B2 (en) | 2018-02-27 | 2021-11-30 | Tesla, Inc. | Parallel-connected solar roof tile modules |
| CN110634962A (en) * | 2018-06-01 | 2019-12-31 | 君泰创新(北京)科技有限公司 | A kind of solar cell and preparation method thereof |
| EP3660927B1 (en) * | 2018-11-30 | 2021-11-10 | CSEM Centre Suisse D'electronique Et De Microtechnique SA | Photovoltaic module |
| CN111883621A (en) * | 2020-07-07 | 2020-11-03 | 江苏爱康能源研究院有限公司 | A kind of TCO coating process method of high-efficiency crystalline silicon heterojunction solar cell |
| JP2023137909A (en) * | 2022-03-18 | 2023-09-29 | 日亜化学工業株式会社 | Sensor element and manufacturing method thereof |
| KR102840417B1 (en) * | 2023-06-02 | 2025-07-29 | 인천대학교 산학협력단 | Color-tunable transparent photovoltaics for onsite power production under sunlight and indoor light |
Family Cites Families (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3969163A (en) | 1974-09-19 | 1976-07-13 | Texas Instruments Incorporated | Vapor deposition method of forming low cost semiconductor solar cells including reconstitution of the reacted gases |
| US3961997A (en) | 1975-05-12 | 1976-06-08 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Fabrication of polycrystalline solar cells on low-cost substrates |
| US4084024A (en) | 1975-11-10 | 1978-04-11 | J. C. Schumacher Co. | Process for the production of silicon of high purity |
| US4124410A (en) * | 1977-11-21 | 1978-11-07 | Union Carbide Corporation | Silicon solar cells with low-cost substrates |
| US4270018A (en) | 1979-12-26 | 1981-05-26 | Gibbons James F | Amorphous solar cells |
| US4490573A (en) * | 1979-12-26 | 1984-12-25 | Sera Solar Corporation | Solar cells |
| US4253882A (en) * | 1980-02-15 | 1981-03-03 | University Of Delaware | Multiple gap photovoltaic device |
| EP0309000B1 (en) | 1981-07-17 | 1992-10-14 | Kanegafuchi Kagaku Kogyo Kabushiki Kaisha | Amorphous semiconductor and amorphous silicon photovoltaic device |
| US4496788A (en) * | 1982-12-29 | 1985-01-29 | Osaka Transformer Co., Ltd. | Photovoltaic device |
| US4528082A (en) | 1983-09-26 | 1985-07-09 | Exxon Research And Engineering Co. | Method for sputtering a PIN amorphous silicon semi-conductor device having partially crystallized P and N-layers |
| US4536607A (en) * | 1984-03-01 | 1985-08-20 | Wiesmann Harold J | Photovoltaic tandem cell |
| US4668840A (en) | 1984-06-29 | 1987-05-26 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| US4689438A (en) * | 1984-10-17 | 1987-08-25 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| US4633034A (en) * | 1985-02-08 | 1986-12-30 | Energy Conversion Devices, Inc. | Photovoltaic device and method |
| US4755475A (en) | 1986-02-18 | 1988-07-05 | Sanyo Electric Co., Ltd. | Method of manufacturing photovoltaic device |
| US4818337A (en) | 1986-04-11 | 1989-04-04 | University Of Delaware | Thin active-layer solar cell with multiple internal reflections |
| US4981525A (en) | 1988-02-19 | 1991-01-01 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| JP2846651B2 (en) | 1989-03-31 | 1999-01-13 | 三洋電機株式会社 | Photovoltaic device |
| JP2740284B2 (en) | 1989-08-09 | 1998-04-15 | 三洋電機株式会社 | Photovoltaic element |
| US5155051A (en) | 1990-06-22 | 1992-10-13 | Sanyo Electric Co., Ltd. | Method of manufacturing photovoltaic device |
| US5213628A (en) | 1990-09-20 | 1993-05-25 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| US5455430A (en) | 1991-08-01 | 1995-10-03 | Sanyo Electric Co., Ltd. | Photovoltaic device having a semiconductor grade silicon layer formed on a metallurgical grade substrate |
| US5705828A (en) | 1991-08-10 | 1998-01-06 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| JP3048732B2 (en) | 1991-11-25 | 2000-06-05 | 三洋電機株式会社 | Photovoltaic device |
| JP3203078B2 (en) | 1992-12-09 | 2001-08-27 | 三洋電機株式会社 | Photovoltaic element |
| US5639314A (en) | 1993-06-29 | 1997-06-17 | Sanyo Electric Co., Ltd. | Photovoltaic device including plural interconnected photoelectric cells, and method of making the same |
| US5538902A (en) | 1993-06-29 | 1996-07-23 | Sanyo Electric Co., Ltd. | Method of fabricating a photovoltaic device having a three-dimensional shape |
| US5549763A (en) | 1993-07-26 | 1996-08-27 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| US5401331A (en) | 1993-09-07 | 1995-03-28 | Midwest Research Institute | Substrate for thin silicon solar cells |
| US5584941A (en) | 1994-03-22 | 1996-12-17 | Canon Kabushiki Kaisha | Solar cell and production process therefor |
| JPH0864851A (en) | 1994-06-14 | 1996-03-08 | Sanyo Electric Co Ltd | Photovoltaic device and manufacturing method thereof |
| JP3246189B2 (en) * | 1994-06-28 | 2002-01-15 | 株式会社日立製作所 | Semiconductor display device |
| US5614785A (en) | 1995-09-28 | 1997-03-25 | Texas Instruments Incorporated | Anode plate for flat panel display having silicon getter |
| JP3349308B2 (en) | 1995-10-26 | 2002-11-25 | 三洋電機株式会社 | Photovoltaic element |
| JP3357808B2 (en) | 1996-01-29 | 2002-12-16 | 三洋電機株式会社 | Solar cell device |
| US5907766A (en) | 1996-10-21 | 1999-05-25 | Electric Power Research Institute, Inc. | Method of making a solar cell having improved anti-reflection passivation layer |
| ATE261612T1 (en) * | 1996-12-18 | 2004-03-15 | Canon Kk | METHOD FOR PRODUCING A SEMICONDUCTOR ARTICLE USING A SUBSTRATE HAVING A POROUS SEMICONDUCTOR LAYER |
| DE69811511T2 (en) | 1997-03-21 | 2004-02-19 | Sanyo Electric Co., Ltd., Moriguchi | MANUFACTURING METHOD FOR A PHOTOVOLTAIC COMPONENT |
| US6060400A (en) | 1998-03-26 | 2000-05-09 | The Research Foundation Of State University Of New York | Highly selective chemical dry etching of silicon nitride over silicon and silicon dioxide |
| EP0949688A1 (en) * | 1998-03-31 | 1999-10-13 | Phototronics Solartechnik GmbH | Thin film solar cell, method of manufacturing the same, and apparatus for carrying out the method of manufacturing |
| JP3545203B2 (en) | 1998-05-22 | 2004-07-21 | 三洋電機株式会社 | Inverter operation method and power supply system |
| EP1071136B1 (en) | 1999-07-23 | 2006-12-13 | Sanyo Electric Co., Ltd. | Solar electric power apparatus, solar module, and installation method of solar modules |
| US6402974B1 (en) | 1999-07-27 | 2002-06-11 | Applied Materials, Inc. | Method for etching polysilicon to have a smooth surface |
| JP2001077382A (en) | 1999-09-08 | 2001-03-23 | Sanyo Electric Co Ltd | Photovoltaic device |
| JP2001148500A (en) | 1999-11-22 | 2001-05-29 | Sanyo Electric Co Ltd | Solar cell module |
| US6340788B1 (en) * | 1999-12-02 | 2002-01-22 | Hughes Electronics Corporation | Multijunction photovoltaic cells and panels using a silicon or silicon-germanium active substrate cell for space and terrestrial applications |
| US6602767B2 (en) * | 2000-01-27 | 2003-08-05 | Canon Kabushiki Kaisha | Method for transferring porous layer, method for making semiconductor devices, and method for making solar battery |
| JP2001291881A (en) | 2000-01-31 | 2001-10-19 | Sanyo Electric Co Ltd | Solar cell module |
| JP4171162B2 (en) | 2000-05-30 | 2008-10-22 | 三洋電機株式会社 | Photovoltaic element and manufacturing method thereof |
| US6521883B2 (en) | 2000-07-18 | 2003-02-18 | Sanyo Electric Co., Ltd. | Photovoltaic device |
| US20030150377A1 (en) | 2000-08-31 | 2003-08-14 | Nobuhiro Arimoto | Silicon monoxide vapor deposition material, process for producing the same, raw material for producing the same, and production apparatus |
| JP3490964B2 (en) | 2000-09-05 | 2004-01-26 | 三洋電機株式会社 | Photovoltaic device |
| JP2002305315A (en) * | 2001-01-31 | 2002-10-18 | Canon Inc | Method of forming semiconductor device and semiconductor device |
| US20020175143A1 (en) | 2001-05-22 | 2002-11-28 | Seh America, Inc. | Processes for polishing wafers |
| US6749685B2 (en) | 2001-08-16 | 2004-06-15 | Cree, Inc. | Silicon carbide sublimation systems and associated methods |
| JP3902534B2 (en) | 2001-11-29 | 2007-04-11 | 三洋電機株式会社 | Photovoltaic device and manufacturing method thereof |
| JP4070483B2 (en) | 2002-03-05 | 2008-04-02 | 三洋電機株式会社 | Photovoltaic device and manufacturing method thereof |
| JP3679771B2 (en) | 2002-03-19 | 2005-08-03 | 三洋電機株式会社 | Photovoltaic device and method for manufacturing photovoltaic device |
| JP2004128060A (en) * | 2002-09-30 | 2004-04-22 | Canon Inc | Silicon film growth method, solar cell manufacturing method, semiconductor substrate, and solar cell |
| JP4171428B2 (en) | 2003-03-20 | 2008-10-22 | 三洋電機株式会社 | Photovoltaic device |
| JP2005142268A (en) * | 2003-11-05 | 2005-06-02 | Canon Inc | Photovoltaic element and manufacturing method thereof |
| NO333319B1 (en) * | 2003-12-29 | 2013-05-06 | Elkem As | Silicon material for the production of solar cells |
| ES2385720T3 (en) * | 2005-02-25 | 2012-07-30 | Sanyo Electric Co., Ltd. | Photovoltaic cell |
| EP1882275A1 (en) | 2005-05-17 | 2008-01-30 | Interuniversitair Microelektronica Centrum Vzw | Method for the production of photovoltaic cells |
| US7875486B2 (en) * | 2007-07-10 | 2011-01-25 | Applied Materials, Inc. | Solar cells and methods and apparatuses for forming the same including I-layer and N-layer chamber cleaning |
| CN103296138A (en) * | 2007-11-09 | 2013-09-11 | 森普雷姆有限公司 | Low-cost solar cells and methods for their production |
| US7951640B2 (en) * | 2008-11-07 | 2011-05-31 | Sunpreme, Ltd. | Low-cost multi-junction solar cells and methods for their production |
| TWI379430B (en) * | 2009-04-16 | 2012-12-11 | Atomic Energy Council | A method of fabricating a thin interface for internal light reflection and impurities isolation |
-
2008
- 2008-11-07 CN CN2013100950349A patent/CN103296138A/en active Pending
- 2008-11-07 PT PT88465091T patent/PT2208238E/en unknown
- 2008-11-07 WO PCT/US2008/082908 patent/WO2009062117A1/en not_active Ceased
- 2008-11-07 US US12/267,528 patent/US7956283B2/en not_active Expired - Fee Related
- 2008-11-07 CN CN2008801154349A patent/CN101960618B/en not_active Expired - Fee Related
- 2008-11-07 KR KR1020107012683A patent/KR101293162B1/en not_active Expired - Fee Related
- 2008-11-07 US US12/267,530 patent/US7960644B2/en not_active Expired - Fee Related
- 2008-11-07 ES ES08846509T patent/ES2422256T3/en active Active
- 2008-11-07 EP EP13164313.2A patent/EP2618387A1/en not_active Withdrawn
- 2008-11-07 EP EP08846509.1A patent/EP2208238B1/en not_active Not-in-force
-
2011
- 2011-05-03 US US13/099,874 patent/US20110207259A1/en not_active Abandoned
- 2011-05-16 US US13/107,962 patent/US20110223708A1/en not_active Abandoned
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104505429A (en) * | 2014-11-21 | 2015-04-08 | 广东爱康太阳能科技有限公司 | Photolithographic process applied to crystalline silicon solar cells |
Also Published As
| Publication number | Publication date |
|---|---|
| US20110223708A1 (en) | 2011-09-15 |
| US20090120493A1 (en) | 2009-05-14 |
| KR101293162B1 (en) | 2013-08-12 |
| US20090120492A1 (en) | 2009-05-14 |
| CN101960618B (en) | 2013-05-01 |
| EP2208238B1 (en) | 2013-04-24 |
| US20110207259A1 (en) | 2011-08-25 |
| ES2422256T3 (en) | 2013-09-10 |
| US7960644B2 (en) | 2011-06-14 |
| CN101960618A (en) | 2011-01-26 |
| EP2618387A1 (en) | 2013-07-24 |
| PT2208238E (en) | 2013-07-10 |
| EP2208238A1 (en) | 2010-07-21 |
| US7956283B2 (en) | 2011-06-07 |
| KR20100095442A (en) | 2010-08-30 |
| WO2009062117A1 (en) | 2009-05-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101960618B (en) | Low-cost solar cells and methods for their production | |
| US8084683B2 (en) | Low-cost multi-junction solar cells and methods for their production | |
| Tao | Inorganic photovoltaic solar cells: silicon and beyond | |
| Kilner et al. | Functional materials for sustainable energy applications | |
| KR101000064B1 (en) | Heterojunction solar cell and its manufacturing method | |
| CN102064216A (en) | Novel crystalline silicon solar cell and manufacturing method thereof | |
| CN101232030A (en) | Method and apparatus for forming a semiconductor structure with at least one via | |
| US8796066B2 (en) | Low-cost solar cells and methods for fabricating low cost substrates for solar cells | |
| Benda | Crystalline Silicon Solar Cell and Module Technology | |
| CN102751371A (en) | Solar thin film battery and manufacturing method thereof | |
| De Nicolas | a-Si: H/c-Si heterojunction solar cells: back side assessment and improvement | |
| Petti et al. | 10-thin films in photovoltaics | |
| JP5975841B2 (en) | Manufacturing method of photovoltaic device and photovoltaic device | |
| Angadi et al. | A review on different types of materials employed in solar photovoltaic panel | |
| CN102386285B (en) | Low-cost solar cells and methods for fabricating low cost substrates for solar cells | |
| KR101228140B1 (en) | Solar Cell | |
| Panek et al. | Crystalline silicon solar cells with high resistivity emitter | |
| Saga | Crystalline and polycrystalline silicon PV technology | |
| Panda et al. | Fabrication of Crystalline-Silicon Based Solar Cell | |
| Moulin et al. | Amorphous and Nanocrystalline Silicon Solar Cells | |
| Prashanth | INVESTIGATING THE CRYSTALLINE TECHNOLOGY FOR SOLAR PANELS | |
| Solanki et al. | c-Si Solar Cells: Physics and Technology | |
| Jiang et al. | Materials Chemistry and Physics for Low‐Cost Silicon Photovoltaics | |
| Buck et al. | Crystalline Si thin film n-type solar cells: a screen printed rear junction approach | |
| WO2011102352A1 (en) | Solar cell, and process for production of solar cell |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20130911 |