CN103221406A - Novel organic electroluminescent compounds and organic electroluminescent device using the same - Google Patents
Novel organic electroluminescent compounds and organic electroluminescent device using the same Download PDFInfo
- Publication number
- CN103221406A CN103221406A CN2011800555279A CN201180055527A CN103221406A CN 103221406 A CN103221406 A CN 103221406A CN 2011800555279 A CN2011800555279 A CN 2011800555279A CN 201180055527 A CN201180055527 A CN 201180055527A CN 103221406 A CN103221406 A CN 103221406A
- Authority
- CN
- China
- Prior art keywords
- substituted
- unsubstituted
- aryl
- alkyl
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- 0 C*=Cc1c(C=*)ccc(*2)c1-c1c2cccc1 Chemical compound C*=Cc1c(C=*)ccc(*2)c1-c1c2cccc1 0.000 description 11
- RRYLBSMPBZACTD-UHFFFAOYSA-N Bc(cc1)ccc1-[n]1c(cc(CC)cc2)c2c2c1ccc1c2cccc1 Chemical compound Bc(cc1)ccc1-[n]1c(cc(CC)cc2)c2c2c1ccc1c2cccc1 RRYLBSMPBZACTD-UHFFFAOYSA-N 0.000 description 1
- SOBALCNFJKGZSH-UHFFFAOYSA-N Bc(cc12)ccc1[nH]c1c2c2ccccc2cc1 Chemical compound Bc(cc12)ccc1[nH]c1c2c2ccccc2cc1 SOBALCNFJKGZSH-UHFFFAOYSA-N 0.000 description 1
- LTBWKAYPXIIVPC-UHFFFAOYSA-N Brc1ccc2[nH]c(cccc3)c3c2c1 Chemical compound Brc1ccc2[nH]c(cccc3)c3c2c1 LTBWKAYPXIIVPC-UHFFFAOYSA-N 0.000 description 1
- FUVCEJQZYTYZGD-UHFFFAOYSA-N C(C(C1C2C=CC=CC12)C=C1)C=C1[n](c(cc1)c2cc1-c(cc1c3c4cccc3)ccc1[n]4-c1nc3ccccc3c(-c3ccccc3)n1)c1c2c(cccc2)c2cc1 Chemical compound C(C(C1C2C=CC=CC12)C=C1)C=C1[n](c(cc1)c2cc1-c(cc1c3c4cccc3)ccc1[n]4-c1nc3ccccc3c(-c3ccccc3)n1)c1c2c(cccc2)c2cc1 FUVCEJQZYTYZGD-UHFFFAOYSA-N 0.000 description 1
- QFKOPCKVIDVXME-UHFFFAOYSA-N C(C1)C([n]2c(cccc3)c3c3ccccc23)=Cc(cc2)c1c(c1cc(-c(cc3c4ccccc44)ccc3[n]4-c3nc4ccccc4c(-c4ccccc4)n3)ccc11)c2[n]1-c1ccccc1 Chemical compound C(C1)C([n]2c(cccc3)c3c3ccccc23)=Cc(cc2)c1c(c1cc(-c(cc3c4ccccc44)ccc3[n]4-c3nc4ccccc4c(-c4ccccc4)n3)ccc11)c2[n]1-c1ccccc1 QFKOPCKVIDVXME-UHFFFAOYSA-N 0.000 description 1
- BYFRDIJRTNYYGG-UHFFFAOYSA-N C(C12)=CC=CC1=NC(c(cc1)ccc1-[n](c1c3cccc1)c1c3c(cccc3)c3cc1)=NC2c1ccccc1 Chemical compound C(C12)=CC=CC1=NC(c(cc1)ccc1-[n](c1c3cccc1)c1c3c(cccc3)c3cc1)=NC2c1ccccc1 BYFRDIJRTNYYGG-UHFFFAOYSA-N 0.000 description 1
- YEDUGIOMBBZSIG-UHFFFAOYSA-N C(C12)=Cc3ccccc3C1c(cc(cc1)-c(cc3c4ccccc44)ccc3[n]4-c3nc4ccccc4c(-c4ccccc4)n3)c1N2c1cc(-c2ccccc2)cc(-c2ccccc2)c1 Chemical compound C(C12)=Cc3ccccc3C1c(cc(cc1)-c(cc3c4ccccc44)ccc3[n]4-c3nc4ccccc4c(-c4ccccc4)n3)c1N2c1cc(-c2ccccc2)cc(-c2ccccc2)c1 YEDUGIOMBBZSIG-UHFFFAOYSA-N 0.000 description 1
- SXBCONQKVGMTPU-XDOYNYLZSA-N C/N=C(/c(cc1)ccc1-c1ccccc1)\c1ccccc1N Chemical compound C/N=C(/c(cc1)ccc1-c1ccccc1)\c1ccccc1N SXBCONQKVGMTPU-XDOYNYLZSA-N 0.000 description 1
- MNXTXWYYCYQTQT-UHFFFAOYSA-N C1C2c(ccc3ccccc33)c3-c3cc(-c4ccc5[nH]c(cccc6)c6c5c4)ccc3C12 Chemical compound C1C2c(ccc3ccccc33)c3-c3cc(-c4ccc5[nH]c(cccc6)c6c5c4)ccc3C12 MNXTXWYYCYQTQT-UHFFFAOYSA-N 0.000 description 1
- DNKDSLCFVMYSCL-UHFFFAOYSA-N C1C=C2C(c3cc(-c4ccccc4)cc(-c4ccccc4)c3)=NC([n](c(cccc3)c3c3c4)c3ccc4-c(cc3)cc(c4c5ccc6c4cccc6)c3[n]5-c3ccccc3)=NC2=CC1 Chemical compound C1C=C2C(c3cc(-c4ccccc4)cc(-c4ccccc4)c3)=NC([n](c(cccc3)c3c3c4)c3ccc4-c(cc3)cc(c4c5ccc6c4cccc6)c3[n]5-c3ccccc3)=NC2=CC1 DNKDSLCFVMYSCL-UHFFFAOYSA-N 0.000 description 1
- BQJSTWLNZLTBNZ-UHFFFAOYSA-N CC(C(CC(C)C=C1)=C1[n](c(cc1)c2cc1-c(cc1c3c4cccc3)ccc1[n]4-c(cc1)ccc1-c1nc(cccc3)c3c(-c3ccccc3)n1)c1c2c2ccccc2cc1)S Chemical compound CC(C(CC(C)C=C1)=C1[n](c(cc1)c2cc1-c(cc1c3c4cccc3)ccc1[n]4-c(cc1)ccc1-c1nc(cccc3)c3c(-c3ccccc3)n1)c1c2c2ccccc2cc1)S BQJSTWLNZLTBNZ-UHFFFAOYSA-N 0.000 description 1
- HUTPNNRZQPWEFU-UHFFFAOYSA-N CC(C)(C)c(cc1)ccc1-[n](c(cc1)c2cc1-c(cc1c3c4cccc3)ccc1[n]4-c1nc3ccccc3c(-c3ccccc3)n1)c1c2c(cccc2)c2cc1 Chemical compound CC(C)(C)c(cc1)ccc1-[n](c(cc1)c2cc1-c(cc1c3c4cccc3)ccc1[n]4-c1nc3ccccc3c(-c3ccccc3)n1)c1c2c(cccc2)c2cc1 HUTPNNRZQPWEFU-UHFFFAOYSA-N 0.000 description 1
- NOFFFWCFDHVZQW-NMTYMURDSA-N CC(C1[n]2c3ccc(C=CCC4C)c4c3c3c2ccc(-c(cc2)cc4c2[s]c2c4C=CC(C)C2)c3)N1/C(/C(CC1)=CC=C1c1ccccc1)=C(/C=CC=C1)\C1=N Chemical compound CC(C1[n]2c3ccc(C=CCC4C)c4c3c3c2ccc(-c(cc2)cc4c2[s]c2c4C=CC(C)C2)c3)N1/C(/C(CC1)=CC=C1c1ccccc1)=C(/C=CC=C1)\C1=N NOFFFWCFDHVZQW-NMTYMURDSA-N 0.000 description 1
- LZMOMXSYCODWJL-UHFFFAOYSA-N CC(C1c(cc2)ccc2-c2n[nH]c3ccccc23)C=Cc2c1[o]c1c2c(-c(cc2)cc3c2c(cccc2-c(cc4c5c6ccc7c5cccc7)ccc4[n]6-c4nc5ccccc5c(-c5ccc(c6ccccc6[n]6-c7ccccc7)c6c5)n4)c2[o]3)ccc1 Chemical compound CC(C1c(cc2)ccc2-c2n[nH]c3ccccc23)C=Cc2c1[o]c1c2c(-c(cc2)cc3c2c(cccc2-c(cc4c5c6ccc7c5cccc7)ccc4[n]6-c4nc5ccccc5c(-c5ccc(c6ccccc6[n]6-c7ccccc7)c6c5)n4)c2[o]3)ccc1 LZMOMXSYCODWJL-UHFFFAOYSA-N 0.000 description 1
- KYHJAKXYDDRYMI-UHFFFAOYSA-N CC(C1c2c3ccc(-c4ccc5[nH]c(cccc6)c6c5c4)c2)(C=Cc2c1cccc2)N3c(cc1)ccc1-c1cc2ccccc2cc1 Chemical compound CC(C1c2c3ccc(-c4ccc5[nH]c(cccc6)c6c5c4)c2)(C=Cc2c1cccc2)N3c(cc1)ccc1-c1cc2ccccc2cc1 KYHJAKXYDDRYMI-UHFFFAOYSA-N 0.000 description 1
- OOMOBTKTKNLVPG-UHFFFAOYSA-N CC(CC(C)=C1)C=C1[n](c(cc1)c2cc1-c(cc1c3ccccc33)ccc1[n]3C(C=C1)=CCC1(C)c1nc(cccc3)c3c(-c3ccccc3)n1)c1c2c(cccc2)c2cc1 Chemical compound CC(CC(C)=C1)C=C1[n](c(cc1)c2cc1-c(cc1c3ccccc33)ccc1[n]3C(C=C1)=CCC1(C)c1nc(cccc3)c3c(-c3ccccc3)n1)c1c2c(cccc2)c2cc1 OOMOBTKTKNLVPG-UHFFFAOYSA-N 0.000 description 1
- QZZVXMMRVYEVFQ-UHFFFAOYSA-N CC1(C)c(c(-c(cc2c3c4ccc5c3cccc5)ccc2[n]4-c2nc3ccccc3c(-c3ccccc3)n2)ccc2)c2-c2ccccc12 Chemical compound CC1(C)c(c(-c(cc2c3c4ccc5c3cccc5)ccc2[n]4-c2nc3ccccc3c(-c3ccccc3)n2)ccc2)c2-c2ccccc12 QZZVXMMRVYEVFQ-UHFFFAOYSA-N 0.000 description 1
- GCHXGHHMBCRNHE-UHFFFAOYSA-N CC1(C)c(cc(cc2)-[n](c3c4cc(C(C)(C5)C=Cc6c5c(cccc5)c5[n]6-c5nc(-c(cc6)ccc6-c6ccccc6)c(cccc6)c6n5)cc3)c3c4c(cccc4)c4cc3)c2-c2c1cccc2 Chemical compound CC1(C)c(cc(cc2)-[n](c3c4cc(C(C)(C5)C=Cc6c5c(cccc5)c5[n]6-c5nc(-c(cc6)ccc6-c6ccccc6)c(cccc6)c6n5)cc3)c3c4c(cccc4)c4cc3)c2-c2c1cccc2 GCHXGHHMBCRNHE-UHFFFAOYSA-N 0.000 description 1
- KVEZYAURQFZCGB-UHFFFAOYSA-N CC1(C=CC([n](c(cccc2)c2c2c3)c2ccc3-c(cc23)ccc2[s]c2c3c3ccccc3cc2)=CC1)c1nc(cccc2)c2c(-c2ccccc2)n1 Chemical compound CC1(C=CC([n](c(cccc2)c2c2c3)c2ccc3-c(cc23)ccc2[s]c2c3c3ccccc3cc2)=CC1)c1nc(cccc2)c2c(-c2ccccc2)n1 KVEZYAURQFZCGB-UHFFFAOYSA-N 0.000 description 1
- PYCXKVWUVQNNTB-UHFFFAOYSA-N CC1=Nc2ccccc2C(c2ccccc2)N1C Chemical compound CC1=Nc2ccccc2C(c2ccccc2)N1C PYCXKVWUVQNNTB-UHFFFAOYSA-N 0.000 description 1
- HZSZILMUNUIJMZ-UHFFFAOYSA-N CC1C=CC([n](c(cc2)c3cc2-c2ccc4[nH]c5ccccc5c4c2)c2c3c(cccc3)c3cc2)=CC1 Chemical compound CC1C=CC([n](c(cc2)c3cc2-c2ccc4[nH]c5ccccc5c4c2)c2c3c(cccc3)c3cc2)=CC1 HZSZILMUNUIJMZ-UHFFFAOYSA-N 0.000 description 1
- MWTMNICJMIGHAF-UHFFFAOYSA-N CN1C(c2ccccc2)=C(C=CC=C2)C2=CC1c(cc1)ccc1-[n]1c2ccc(cccc3)c3c2c2c1ccc(-c(cc1)cc(c3c4cccc3)c1[n]4-c1ccccc1)c2 Chemical compound CN1C(c2ccccc2)=C(C=CC=C2)C2=CC1c(cc1)ccc1-[n]1c2ccc(cccc3)c3c2c2c1ccc(-c(cc1)cc(c3c4cccc3)c1[n]4-c1ccccc1)c2 MWTMNICJMIGHAF-UHFFFAOYSA-N 0.000 description 1
- OUBJSTLNCBPWRG-UHFFFAOYSA-N Cc1cc(-c2nc(-c(cc3)ccc3-[n](c3ccccc3c3c4)c3ccc4-c(cc34)ccc3[s]c3c4c4ccccc4cc3)nc3c2cccc3)cc(C)c1 Chemical compound Cc1cc(-c2nc(-c(cc3)ccc3-[n](c3ccccc3c3c4)c3ccc4-c(cc34)ccc3[s]c3c4c4ccccc4cc3)nc3c2cccc3)cc(C)c1 OUBJSTLNCBPWRG-UHFFFAOYSA-N 0.000 description 1
- OCNMJXBWHHIIGW-UHFFFAOYSA-N Cc1ccc2c(c(cccc3)c3cc3)c3[nH]c2c1 Chemical compound Cc1ccc2c(c(cccc3)c3cc3)c3[nH]c2c1 OCNMJXBWHHIIGW-UHFFFAOYSA-N 0.000 description 1
- SFKMVPQJJGJCMI-UHFFFAOYSA-N Clc1nc2ccccc2c(-c2ccccc2)n1 Chemical compound Clc1nc2ccccc2c(-c2ccccc2)n1 SFKMVPQJJGJCMI-UHFFFAOYSA-N 0.000 description 1
- XZEKOCKQKUIHEJ-UHFFFAOYSA-N Fc(cc1)ccc1-[n](c(c1c2)ccc2-c(cc2c3c4cccc3)ccc2[n]4-c(cc2)ccc2-c2nc(cccc3)c3c(-c3ccccc3)n2)c2c1c(cccc1)c1cc2 Chemical compound Fc(cc1)ccc1-[n](c(c1c2)ccc2-c(cc2c3c4cccc3)ccc2[n]4-c(cc2)ccc2-c2nc(cccc3)c3c(-c3ccccc3)n2)c2c1c(cccc1)c1cc2 XZEKOCKQKUIHEJ-UHFFFAOYSA-N 0.000 description 1
- MPABXPJSFAGQMI-UHFFFAOYSA-N Fc(cc1)ccc1-c1nc(-c(cc2)ccc2-[n](c2ccccc2c2c3)c2ccc3-c(cc23)ccc2[o]c2c3c3ccccc3cc2)nc2c1cccc2 Chemical compound Fc(cc1)ccc1-c1nc(-c(cc2)ccc2-[n](c2ccccc2c2c3)c2ccc3-c(cc23)ccc2[o]c2c3c3ccccc3cc2)nc2c1cccc2 MPABXPJSFAGQMI-UHFFFAOYSA-N 0.000 description 1
- UCAUXKMNUFNZDA-UHFFFAOYSA-N I[IH][n]1c(C=CC2C=CC=CC22)c2c2c1C=CC2 Chemical compound I[IH][n]1c(C=CC2C=CC=CC22)c2c2c1C=CC2 UCAUXKMNUFNZDA-UHFFFAOYSA-N 0.000 description 1
- SVEXKQQHSUBFPU-UHFFFAOYSA-N I[IH][n]1c(C=CCC2)c2c2c1C=CCC2 Chemical compound I[IH][n]1c(C=CCC2)c2c2c1C=CCC2 SVEXKQQHSUBFPU-UHFFFAOYSA-N 0.000 description 1
- UGFOTZLGPPWNPY-UHFFFAOYSA-N c(cc1)cc2c1[nH]c1c2c(cccc2)c2cc1 Chemical compound c(cc1)cc2c1[nH]c1c2c(cccc2)c2cc1 UGFOTZLGPPWNPY-UHFFFAOYSA-N 0.000 description 1
- ZPFWAPBOFOIKNC-UHFFFAOYSA-N c(cc1)ccc1-c(cc1)ccc1-c1nc(-[n](c(cccc2)c2c2c3)c2ccc3-c(cc2)cc(c3c4ccc5c3cccc5)c2[n]4-c(cc2)ccc2-c2ccccc2)nc2ccccc12 Chemical compound c(cc1)ccc1-c(cc1)ccc1-c1nc(-[n](c(cccc2)c2c2c3)c2ccc3-c(cc2)cc(c3c4ccc5c3cccc5)c2[n]4-c(cc2)ccc2-c2ccccc2)nc2ccccc12 ZPFWAPBOFOIKNC-UHFFFAOYSA-N 0.000 description 1
- ZHMCYXWGPNMDLM-UHFFFAOYSA-N c(cc1)ccc1-c1cccc(-c2cccc(-c3nc(-[n](c(cccc4)c4c4c5)c4ccc5-c(cc4)cc(c5c6ccc7c5cccc7)c4[n]6-c4ccccc4)nc4c3cccc4)c2)c1 Chemical compound c(cc1)ccc1-c1cccc(-c2cccc(-c3nc(-[n](c(cccc4)c4c4c5)c4ccc5-c(cc4)cc(c5c6ccc7c5cccc7)c4[n]6-c4ccccc4)nc4c3cccc4)c2)c1 ZHMCYXWGPNMDLM-UHFFFAOYSA-N 0.000 description 1
- QNEQEFJWFISXJB-UHFFFAOYSA-N c(cc1)ccc1-c1nc(-[n](c2ccccc2c2c3)c2ccc3-c(cc2)cc(c3c4ccc5c3cccc5)c2[n]4-c2ccccc2)nc2c1cccc2 Chemical compound c(cc1)ccc1-c1nc(-[n](c2ccccc2c2c3)c2ccc3-c(cc2)cc(c3c4ccc5c3cccc5)c2[n]4-c2ccccc2)nc2c1cccc2 QNEQEFJWFISXJB-UHFFFAOYSA-N 0.000 description 1
- GIJLUPOUEGVXSF-UHFFFAOYSA-N c(cc1)ccc1-c1nc(-c2cccc(-[n](c(cccc3)c3c3c4)c3ccc4-c(cc3)cc(c4c5ccc6ccccc46)c3[n]5-c3ccccc3)c2)nc2c1cccc2 Chemical compound c(cc1)ccc1-c1nc(-c2cccc(-[n](c(cccc3)c3c3c4)c3ccc4-c(cc3)cc(c4c5ccc6ccccc46)c3[n]5-c3ccccc3)c2)nc2c1cccc2 GIJLUPOUEGVXSF-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings
- C07D403/04—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D403/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00
- C07D403/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, not provided for by group C07D401/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/20—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the material in which the electroluminescent material is embedded
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/342—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising iridium
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1088—Heterocyclic compounds characterised by ligands containing oxygen as the only heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1092—Heterocyclic compounds characterised by ligands containing sulfur as the only heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Crystallography & Structural Chemistry (AREA)
- Inorganic Chemistry (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Plural Heterocyclic Compounds (AREA)
- Laminated Bodies (AREA)
Abstract
Description
技术领域technical field
本发明涉及新颖的有机电致发光化合物以及包括该化合物的有机电致发光器件。The present invention relates to novel organic electroluminescent compounds and organic electroluminescent devices comprising the same.
技术背景technical background
在显示器件中,自发射显示器件的电致发光(EL)器件是优选的,因为它们提供宽视角、优异的对比度和快速的响应速率。伊斯曼柯达公司(EastmanKodak)在1987年首先开发了一种有机EL器件,该器件使用低分子量芳香族二胺和铝配合物作为形成电致发光层的物质[Appl.Phys.Lett.51,913,1987]。Among display devices, electroluminescent (EL) devices of self-emissive display devices are preferred because they provide wide viewing angles, excellent contrast, and fast response rates. Eastman Kodak first developed an organic EL device in 1987, which used low-molecular-weight aromatic diamines and aluminum complexes as materials for forming the electroluminescent layer [Appl.Phys.Lett.51,913, 1987].
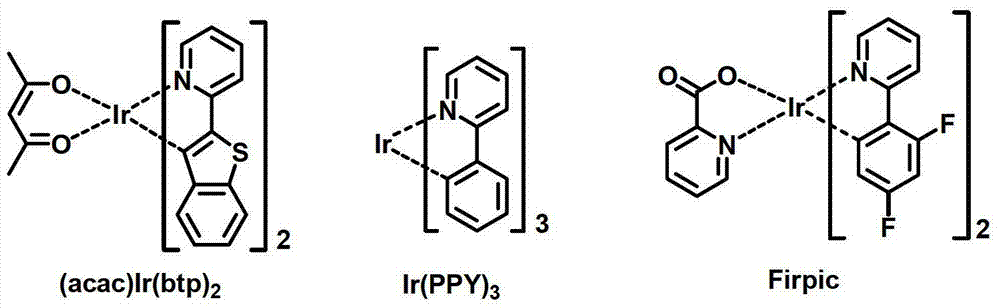
决定有机发光二极管(OLED)的发光效率的最重要因素是电致发光材料。尽管到目前为止荧光材料已经广泛用作电致发光材料,但是从电致发光机理来看,开发磷光材料是理论上将发光效率提高最高到4倍的最好途径之一。到目前为止,铱(III)络合物是众所周知的一种磷光材料,包括(acac)Ir(btp)2、Ir(ppy)3和Firpic,分别作为红色、绿色和蓝色磷光材料。具体来说,许多磷光材料目前已经在日本、欧洲和美国进行研究。The most important factor determining the luminous efficiency of an organic light-emitting diode (OLED) is the electroluminescent material. Although fluorescent materials have been widely used as electroluminescent materials so far, from the perspective of electroluminescence mechanism, developing phosphorescent materials is one of the best ways to theoretically increase the luminous efficiency up to 4 times. So far, iridium(III) complexes are well known as a phosphorescent material, including (acac)Ir(btp) 2 , Ir(ppy) 3 and Firpic as red, green and blue phosphorescent materials, respectively. Specifically, many phosphorescent materials are currently being researched in Japan, Europe, and the United States.
目前,已知CBP是最广泛用作磷光材料的基质材料。已经报道了使用包含BCP、BAlq等的空穴阻挡层的高效OLED。先锋公司(日本)等已经报道了使用BAlq衍生物作为基质的高性能OLED。Currently, CBP is known to be the most widely used host material for phosphorescent materials. High-efficiency OLEDs using hole-blocking layers comprising BCP, BAlq, etc. have been reported. Pioneer Corporation (Japan) and others have reported high-performance OLEDs using BAlq derivatives as substrates.
尽管这些材料提供了良好的电致发光特性,但是由于它们具有低的玻璃化转变温度和差的热稳定性,在真空中的高温沉积加工过程中可能会发生降解,因而是不利的。因为OLED的功率效率是由(π/电压)×电流效率确定的,所以功率效率与电压成反比。需要高的功率效率来降低OLED的功率消耗。实际上,使用磷光材料的OLED提供比使用荧光材料的OLED好得多的电流效率(cd/A)。但是,当现有材料例如BAlq、CBP等用作磷光材料的基质时,相比使用荧光材料的OLED在功率效率(lm/W)方面没有明显优势,这是因为驱动电压较高。另外,使用此类材料的OLED器件的寿命也不令人满意。Although these materials provide good electroluminescent properties, they are disadvantageous due to their low glass transition temperature and poor thermal stability, which may degrade during high temperature deposition processes in vacuum. Since the power efficiency of an OLED is determined by (π/voltage)×current efficiency, the power efficiency is inversely proportional to the voltage. High power efficiency is required to reduce the power consumption of OLEDs. In fact, OLEDs using phosphorescent materials provide much better current efficiency (cd/A) than OLEDs using fluorescent materials. However, when existing materials such as BAlq, CBP, etc. are used as hosts of phosphorescent materials, there is no obvious advantage in power efficiency (lm/W) compared to OLEDs using fluorescent materials because of higher driving voltage. In addition, the lifetime of OLED devices using such materials is not satisfactory.
另一方面,国际专利申请第WO2006/049013号揭示了一种用于有机电致发光材料的化合物,其主链具有稠合二环基团。然而,该文献并没有具体揭示具有以下两种特征的化合物:用与芳环稠合的环烷基或杂环烷基取代的咔唑主链和含氮稠合二环基团。On the other hand, International Patent Application No. WO2006/049013 discloses a compound for an organic electroluminescent material, the main chain of which has a fused bicyclic group. However, this document does not specifically disclose a compound having two features: a carbazole main chain substituted with a cycloalkyl or heterocycloalkyl group fused to an aromatic ring and a nitrogen-containing fused bicyclic group.
发明内容Contents of the invention
技术问题technical problem
因此,本发明注意到相关技术存在的问题,本发明的一个目的在于提供具有主链的有机电致发光化合物,从而相对于常规材料能提供更好的发光效率和具有合适的色坐标的器件寿命。Therefore, the present invention has noticed the problems in the related art, and an object of the present invention is to provide an organic electroluminescent compound with a main chain, thereby providing better luminous efficiency and device life with suitable color coordinates compared to conventional materials .
本发明的另一个目的在于提供具有高效率和长工作寿命的有机电致发光器件,该发光器件使用所述有机电致发光化合物作为电致发光材料。Another object of the present invention is to provide an organic electroluminescent device having high efficiency and long operating life, which uses the organic electroluminescent compound as an electroluminescent material.
技术方案Technical solutions
提供了一种用于如下化学式1表示的有机电致发光化合物的化合物,以及使用该化合物的有机电致发光器件。由于本发明的有机电致发光化合物具有优越的发光效率和优异的寿命性质,它可用于制造OLED器件,该器件具有极优越的工作寿命并且由于改进的功率效率而消耗较少的功率。Provided are a compound for an organic electroluminescent compound represented by Chemical Formula 1 below, and an organic electroluminescent device using the compound. Since the organic electroluminescent compound of the present invention has superior luminous efficiency and excellent lifetime properties, it can be used to manufacture OLED devices which have an extremely superior operating lifetime and consume less power due to improved power efficiency.
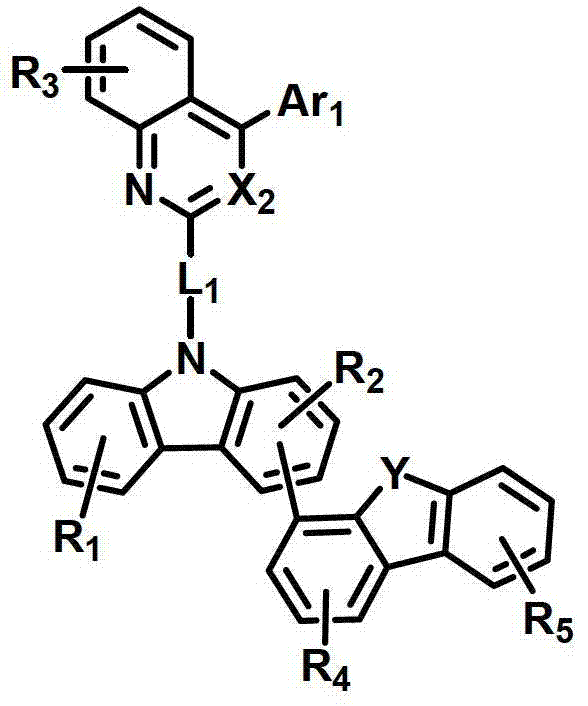
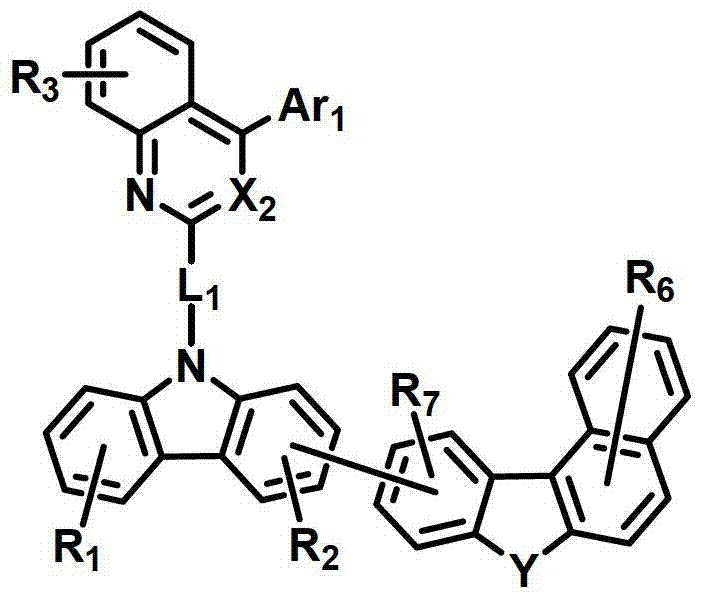
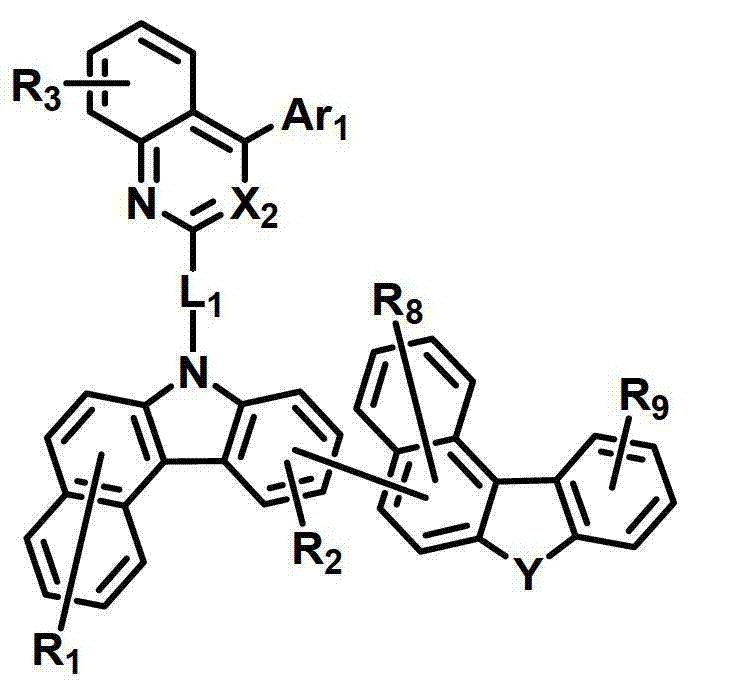
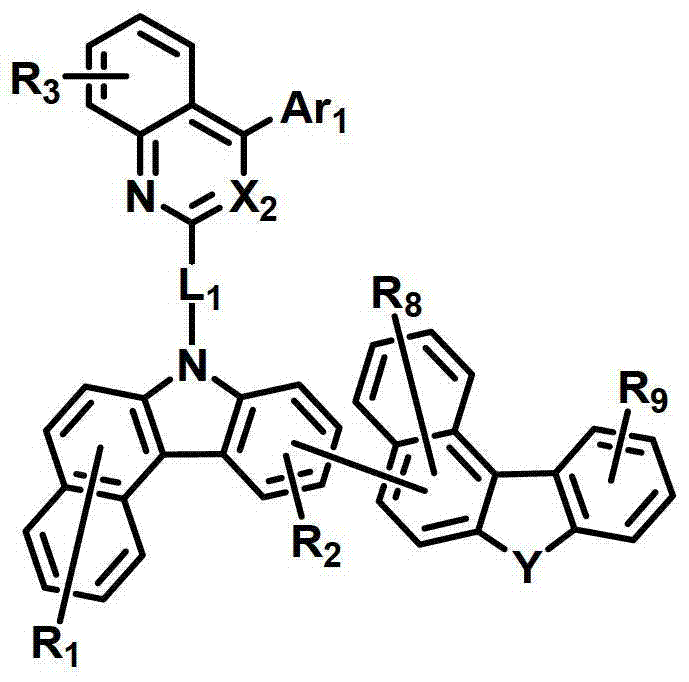
化学式1chemical formula 1
其中:in:
环A表示单环或多环芳环;Ring A represents a monocyclic or polycyclic aromatic ring;
X1和X2独立地表示N或CR’;X 1 and X 2 independently represent N or CR';
L1表示单键、取代或未取代的(C6-C30)亚芳基、取代或未取代的(C2-C30)杂亚芳基或取代或未取代的(C3-C30)环亚烷基; L represents a single bond, a substituted or unsubstituted (C6-C30) arylene group, a substituted or unsubstituted (C2-C30) heteroarylene group or a substituted or unsubstituted (C3-C30) cycloalkylene group;
Ar1表示氢、氘、卤素、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基、或者取代或未取代的(C2-C30)杂芳基;Ar 1 represents hydrogen, deuterium, halogen, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl, or substituted or unsubstituted (C2-C30) heteroaryl;
Z独立地选自以下结构:Z is independently selected from the following structures:
并且当所述环A是单环芳环时,Z仅选自以下结构:And when the ring A is a monocyclic aromatic ring, Z is only selected from the following structures:
Y表示-O-、-S-、-C(R11R12)-、-Si(R13R14)-或-N(R15)-;Y represents -O-, -S-, -C(R 11 R 12 )-, -Si(R 13 R 14 )- or -N(R 15 )-;
R1至R9独立地表示氢、氘、卤素、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基、取代或未取代的(C2-C30)杂芳基、取代或未取代的(C3-C30)环烷基、取代或未取代的5-至7-元杂环烷基、取代或未取代的(C6-C30)芳基(C1-C30)烷基、与一个或多个(C3-C30)环烷基稠合的取代或未取代的(C6-C30)芳基、与取代或未取代的一个或多个芳环稠合的5-至7-元杂环烷基、与取代或未取代的一个或多个芳环稠合的(C3-C30)环烷基、-NR16R17、-SiR18R19R20、-SR21、-OR22、氰基、硝基或羟基;或者R1至R9可以通过具有或不具有稠环的取代或未取代的(C3-C30)亚烯基或取代或未取代的(C3-C30)亚烷基与相邻取代基相连形成脂环和单环或多环芳环,并且该脂环和单环或多环芳环的碳原子可以被一个或多个选自N、O和S的杂原子取代;R 1 to R 9 independently represent hydrogen, deuterium, halogen, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl, substituted or unsubstituted (C2-C30) Heteroaryl, substituted or unsubstituted (C3-C30) cycloalkyl, substituted or unsubstituted 5- to 7-membered heterocycloalkyl, substituted or unsubstituted (C6-C30) aryl (C1-C30 ) alkyl, substituted or unsubstituted (C6-C30) aryl fused to one or more (C3-C30) cycloalkyl, 5- fused to one or more substituted or unsubstituted aromatic rings To 7-membered heterocycloalkyl, (C3-C30)cycloalkyl fused with one or more substituted or unsubstituted aromatic rings, -NR 16 R 17 , -SiR 18 R 19 R 20 , -SR 21 , -OR 22 , cyano, nitro or hydroxyl; or R1 to R9 can be substituted or unsubstituted (C3-C30) alkenylene or substituted or unsubstituted (C3-C30) with or without fused rings The alkylene group is connected with adjacent substituents to form an alicyclic ring and a monocyclic or polycyclic aromatic ring, and the carbon atoms of the alicyclic ring and a monocyclic or polycyclic aromatic ring can be replaced by one or more selected from N, O and S Heteroatom substitution;
R’以及R11至R22独立地表示氢、氘、卤素、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基、取代或未取代的(C2-C30)杂芳基、取代或未取代的5-至7-元杂环烷基或取代或未取代的(C3-C30)环烷基;或者它们可通过具有或不具有稠环的取代或未取代的(C3-C30)亚烯基或取代或未取代的(C3-C30)亚烷基与相邻取代基相连形成脂环和单环或多环芳环,并且该脂环和单环或多环芳环的碳原子可以被一个或多个选自N、O和S的杂原子取代;R' and R 11 to R 22 independently represent hydrogen, deuterium, halogen, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl, substituted or unsubstituted (C2 -C30)heteroaryl, substituted or unsubstituted 5- to 7-membered heterocycloalkyl or substituted or unsubstituted (C3-C30)cycloalkyl; or they may be substituted or unsubstituted with or without fused rings Unsubstituted (C3-C30) alkenylene or substituted or unsubstituted (C3-C30) alkylene is connected with adjacent substituents to form alicyclic and monocyclic or polycyclic aromatic rings, and the alicyclic and monocyclic Or the carbon atoms of the polycyclic aromatic ring can be replaced by one or more heteroatoms selected from N, O and S;
a、c、e和i独立地表示1至4的整数,并且当a、c、e和I是大于或等于2的整数时,各R1、R3、R5和R9可以相同或彼此不同;a, c, e and i independently represent an integer of 1 to 4, and when a, c, e and i are integers greater than or equal to 2, each of R 1 , R 3 , R 5 and R 9 may be the same or each other different;
b、d和g独立地表示1至3的整数,并且当b、d和g是大于或等于2的整数时,各R2、R4和R7可以相同或彼此不同;b, d and g independently represent an integer of 1 to 3, and when b, d and g are integers greater than or equal to 2, each of R 2 , R 4 and R 7 may be the same or different from each other;
f表示1至6的整数,并且当f是大于或等于2的整数时,各R6可以相同或彼此不同;f represents an integer from 1 to 6, and when f is an integer greater than or equal to 2, each R 6 can be the same or different from each other;
h表示1至5的整数,并且当h是大于或等于2的整数时,各R8可以相同或彼此不同;以及h represents an integer from 1 to 5, and when h is an integer greater than or equal to 2, each R may be the same or different from each other; and
所述杂芳环、杂亚芳基、杂环烷基和杂芳基包含一个或多个选自B、N、O、S、P(=O)、Si和P的杂原子。The heteroaryl ring, heteroarylene, heterocycloalkyl, and heteroaryl include one or more heteroatoms selected from B, N, O, S, P(=O), Si, and P.
本文所用术语“烷基”、“烷氧基”和其它含有“烷基”部分的取代基包括直链和支链部分,并且“环烷基”包括多环烃环(例如取代或未取代的金刚烷基或者取代或未取代的(C7-C30)二环烷基)以及单环烃环。本文所用术语“芳基”指的是通过从芳香烃去除一个氢原子得到的有机基团,其可包括4-至7-元、特别是5-或6-元的单环或稠环,甚至还包括通过单键连接的多个芳基的结构。其具体例子包括但不限于:苯基、萘基、联苯基、三联苯基、蒽基、茚基、芴基、菲基、苯并[9,10]菲基(triphenylenyl)、芘基、苝基(perylenyl)、基(chrysenyl)、并四苯基(naphthacenyl)、荧蒽基(fluoranthenyl)等。所述萘基包括1-萘基和2-萘基,所述蒽基包括1-蒽基、2-蒽基和9-蒽基,所述菲基包括1-菲基、2-菲基、3-菲基、4-菲基和9-菲基,所述并四苯基(naphthacenyl)包括1-并四苯基、2-并四苯基和9-并四苯基。所述芘基包括1-芘基、2-芘基和4-芘基,所述联苯基包括2-联苯基、3-联苯基和4-联苯基,所述三联苯基包括对-三联苯基-4-基、对-三联苯基-3-基、对-三联苯基-2-基、间-三联苯基-4-基、间-三联苯基-3-基和间-三联苯基-2-基,所述芴基包括1-芴基、2-芴基、3-芴基、4-芴基和9-芴基。As used herein, the terms "alkyl", "alkoxy" and other substituents containing "alkyl" moieties include straight and branched chain moieties, and "cycloalkyl" includes polycyclic hydrocarbon rings (such as substituted or unsubstituted adamantyl or substituted or unsubstituted (C7-C30) bicycloalkyl) and monocyclic hydrocarbon rings. The term "aryl" as used herein refers to an organic group obtained by removing one hydrogen atom from an aromatic hydrocarbon, which may include 4- to 7-membered, especially 5- or 6-membered monocyclic or condensed rings, even Structures in which a plurality of aryl groups are linked by single bonds are also included. Specific examples thereof include, but are not limited to: phenyl, naphthyl, biphenyl, terphenyl, anthracenyl, indenyl, fluorenyl, phenanthrenyl, benzo[9,10]phenanthrenyl (triphenylenyl), pyrenyl, perylenyl, Chrysenyl, naphthacenyl, fluoroanthenyl, etc. The naphthyl includes 1-naphthyl and 2-naphthyl, the anthracenyl includes 1-anthracenyl, 2-anthracenyl and 9-anthracenyl, and the phenanthrenyl includes 1-phenanthrenyl, 2-phenanthrenyl, 3-phenanthrenyl, 4-phenanthrenyl and 9-phenanthrenyl, the naphthacenyl includes 1-naphthacenyl, 2-naphthacenyl and 9-naphthacenyl. The pyrenyl includes 1-pyrenyl, 2-pyrenyl and 4-pyrenyl, the biphenyl includes 2-biphenyl, 3-biphenyl and 4-biphenyl, and the terphenyl includes P-terphenyl-4-yl, p-terphenyl-3-yl, p-terphenyl-2-yl, m-terphenyl-4-yl, m-terphenyl-3-yl and m-terphenyl-2-yl, the fluorenyl includes 1-fluorenyl, 2-fluorenyl, 3-fluorenyl, 4-fluorenyl and 9-fluorenyl.
本文所用术语“杂芳基”表示包含1-4个选自B、N、O、S、P(=O)、Si和P的杂原子作为芳环骨架原子,并且其他芳环骨架原子为碳的芳基基团。它可以是与一个或多个苯环缩合得到的5元或6元单环杂芳基或多环杂芳基,且可以是部分饱和的。在本发明中,“杂芳基”包含通过单键连接的一个或多个杂芳基的结构。所述杂芳基包括二价芳基,其中环中的杂原子可被氧化或季铵化,形成例如N-氧化物或季铵盐。它们具体的例子包括但并不限于,单环杂芳基例如呋喃基、噻吩基(thiophenyl)、吡咯基、咪唑基、吡唑基、噻唑基、噻二唑基、异噻唑基、异噁唑基、噁唑基、噁二唑基、三嗪基、四嗪基、三唑基、呋咱基(furazanyl)、吡啶基、吡嗪基、嘧啶基、哒嗪基等;多环杂芳基例如苯并呋喃基、苯并噻吩基、二苯并呋喃基、二苯并噻吩基、异苯并呋喃基、苯并咪唑基、苯并噻唑基、苯并异噻唑基、苯并异噁唑基、苯并噁唑基、异吲哚基、吲哚基、吲唑基、苯并噻二唑基、喹啉基、异喹啉基、噌啉基(cinnolinyl)、喹唑啉基、喹喔啉基(quinoxalinyl)、咔唑基、菲啶基(phenanthridinyl)、苯并间二氧杂环戊烯基(benzodioxolyl)、吖啶基、菲咯啉基、吩嗪基、吩噻嗪基、吩噁嗪基等;及其N-氧化物(例如吡啶基N-氧化物、喹啉基N-氧化物);或其季铵盐等。The term "heteroaryl" as used herein means comprising 1-4 heteroatoms selected from B, N, O, S, P(=O), Si and P as aromatic ring skeletal atoms, and the other aromatic ring skeletal atoms are carbon aryl group. It may be a 5-membered or 6-membered monocyclic heteroaryl or polycyclic heteroaryl obtained by condensation with one or more benzene rings, and may be partially saturated. In the present invention, "heteroaryl" includes a structure in which one or more heteroaryl groups are linked by a single bond. The heteroaryl groups include divalent aryl groups in which ring heteroatoms can be oxidized or quaternized to form, for example, N-oxides or quaternary ammonium salts. Specific examples thereof include, but are not limited to, monocyclic heteroaryl groups such as furyl, thiophenyl, pyrrolyl, imidazolyl, pyrazolyl, thiazolyl, thiadiazolyl, isothiazolyl, isoxazole oxazolyl, oxadiazolyl, triazinyl, tetrazinyl, triazolyl, furazanyl (furazanyl), pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, etc.; polycyclic heteroaryl For example, benzofuryl, benzothienyl, dibenzofuryl, dibenzothienyl, isobenzofuryl, benzimidazolyl, benzothiazolyl, benzisothiazolyl, benzisoxazole Base, benzoxazolyl, isoindolyl, indolyl, indazolyl, benzothiadiazolyl, quinolinyl, isoquinolyl, cinnolinyl (cinnolinyl), quinazolinyl, quinolinyl quinoxalinyl, carbazolyl, phenanthridinyl, benzodioxolyl, acridinyl, phenanthrolinyl, phenazinyl, phenothiazinyl, Phenoxazinyl, etc.; N-oxides thereof (such as pyridyl N-oxide, quinolinyl N-oxide); or quaternary ammonium salts thereof, etc.
吡咯基包括:1-吡咯基、2-吡咯基和3-吡咯基;吡啶基包括2-吡啶基、3-吡啶基和4-吡啶基;吲哚基包括1-吲哚基、2-吲哚基、3-吲哚基、4-吲哚基、5-吲哚基、6-吲哚基和7-吲哚基;异吲哚基包括1-异吲哚基、2-异吲哚基、3-异吲哚基、4-异吲哚基、5-异吲哚基、6-异吲哚基和7-异吲哚基;呋喃基包括2-呋喃基和3-呋喃基;苯并呋喃基包括2-苯并呋喃基、3-苯并呋喃基、4-苯并呋喃基、5-苯并呋喃基、6-苯并呋喃基和7-苯并呋喃基;异苯并呋喃基包括1-异苯并呋喃基、3-异苯并呋喃基、4-异苯并呋喃基、5-异苯并呋喃基、6-异苯并呋喃基和7-异苯并呋喃基;喹啉基包括3-喹啉基、4-喹啉基、5-喹啉基、6-喹啉基、7-喹啉基和8-喹啉基;异喹啉基包括1-异喹啉基、3-异喹啉基、4-异喹啉基、5-异喹啉基、6-异喹啉基、7-异喹啉基和8-异喹啉基;喹喔啉基包括2-喹喔啉基、5-喹喔啉基和6-喹喔啉基;咔唑基包括1-咔唑基、2-咔唑基、3-咔唑基、4-咔唑基和9-咔唑基;菲啶基包括1-菲啶基、2-菲啶基、3-菲啶基、4-菲啶基、6-菲啶基、7-菲啶基、8-菲啶基、9-菲啶基和10-菲啶基;吖啶基包括1-吖啶基、2-吖啶基、3-吖啶基、4-吖啶基和9-吖啶基;菲咯啉基包括1,7-菲咯啉-2-基、1,7-菲咯啉-3-基、1,7-菲咯啉-4-基、1,7-菲咯啉-5-基、1,7-菲咯啉-6-基、1,7-菲咯啉-8-基、1,7-菲咯啉-9-基、1,7-菲咯啉-10-基、1,8-菲咯啉-2-基、1,8-菲咯啉-3-基、1,8-菲咯啉-4-基、1,8-菲咯啉-5-基、1,8-菲咯啉-6-基、1,8-菲咯啉-7-基、1,8-菲咯啉-9-基、1,8-菲咯啉-10-基、1,9-菲咯啉-2-基、1,9-菲咯啉-3-基、1,9-菲咯啉-4-基、1,9-菲咯啉-5-基、1,9-菲咯啉-6-基、1,9-菲咯啉-7-基、1,9-菲咯啉-8-基、1,9-菲咯啉-10-基、1,10-菲咯啉-2-基、1,10-菲咯啉-3-基、1,10-菲咯啉-4-基、1,10-菲咯啉-5-基、2,9-菲咯啉-1-基、2,9-菲咯啉-3-基、2,9-菲咯啉-4-基、2,9-菲咯啉-5-基、2,9-菲咯啉-6-基、2,9-菲咯啉-7-基、2,9-菲咯啉-8-基、2,9-菲咯啉-10-基、2,8-菲咯啉-1-基、2,8-菲咯啉-3-基、2,8-菲咯啉-4-基、2,8-菲咯啉-5-基、2,8-菲咯啉-6-基、2,8-菲咯啉-7-基、2,8-菲咯啉-9-基、2,8-菲咯啉-10-基、2,7-菲咯啉-1-基、2,7-菲咯啉-3-基、2,7-菲咯啉-4-基、2,7-菲咯啉-5-基、2,7-菲咯啉-6-基、2,7-菲咯啉-8-基、2,7-菲咯啉-9-基和2,7-菲咯啉-10-基;吩嗪基包括1-吩嗪基和2-吩嗪基;吩噻嗪基包括1-吩噻嗪基、2-吩噻嗪基、3-吩噻嗪基、4-吩噻嗪基和10-吩噻嗪基;吩噁嗪基包括1-吩噁嗪基、2-吩噁嗪基、3-吩噁嗪基、4-吩噁嗪基和10-吩噁嗪基;噁唑基包括2-噁唑基、4-噁唑基和5-噁唑基;噁二唑包括2-噁二唑基和5-噁二唑基;呋咱基包括3-呋咱基;二苯并呋喃基包括1-二苯并呋喃基、2-二苯并呋喃基、3-二苯并呋喃基和4-二苯并呋喃基;二苯并噻吩基包括1-二苯并噻吩基、2-二苯并噻吩基、3-二苯并噻吩基和4-二苯并噻吩基。Pyrrolyl includes: 1-pyrrolyl, 2-pyrrolyl and 3-pyrrolyl; pyridyl includes 2-pyridyl, 3-pyridyl and 4-pyridyl; indolyl includes 1-indolyl, 2-indolyl Indolyl, 3-indolyl, 4-indolyl, 5-indolyl, 6-indolyl, and 7-indolyl; isoindolyl includes 1-isoindolyl, 2-isoindolyl Base, 3-isoindolyl, 4-isoindolyl, 5-isoindolyl, 6-isoindolyl and 7-isoindolyl; furyl includes 2-furyl and 3-furyl; Benzofuryl includes 2-benzofuryl, 3-benzofuryl, 4-benzofuryl, 5-benzofuryl, 6-benzofuryl and 7-benzofuryl; isobenzo Furanyl includes 1-isobenzofuryl, 3-isobenzofuryl, 4-isobenzofuryl, 5-isobenzofuryl, 6-isobenzofuryl and 7-isobenzofuryl ; Quinolinyl includes 3-quinolyl, 4-quinolyl, 5-quinolyl, 6-quinolyl, 7-quinolyl and 8-quinolyl; isoquinolyl includes 1-isoquinolyl Linyl, 3-isoquinolinyl, 4-isoquinolinyl, 5-isoquinolinyl, 6-isoquinolinyl, 7-isoquinolinyl and 8-isoquinolinyl; quinoxalinyl includes 2-quinoxalinyl, 5-quinoxalinyl and 6-quinoxalinyl; carbazolyl includes 1-carbazolyl, 2-carbazolyl, 3-carbazolyl, 4-carbazolyl and 9 -carbazolyl; phenanthridinyl includes 1-phenanthridinyl, 2-phenanthridinyl, 3-phenanthridinyl, 4-phenanthridinyl, 6-phenanthridinyl, 7-phenanthridinyl, 8-phenanthridinyl , 9-phenanthridinyl and 10-phenanthridinyl; acridinyl includes 1-acridinyl, 2-acridinyl, 3-acridinyl, 4-acridinyl and 9-acridinyl; phenanthroline The base includes 1,7-phenanthrolin-2-yl, 1,7-phenanthrolin-3-yl, 1,7-phenanthrolin-4-yl, 1,7-phenanthrolin-5-yl, 1,7-phenanthrolin-6-yl, 1,7-phenanthrolin-8-yl, 1,7-phenanthrolin-9-yl, 1,7-phenanthrolin-10-yl, 1, 8-phenanthrolin-2-yl, 1,8-phenanthrolin-3-yl, 1,8-phenanthrolin-4-yl, 1,8-phenanthrolin-5-yl, 1,8- Phenanthroline-6-yl, 1,8-phenanthroline-7-yl, 1,8-phenanthroline-9-yl, 1,8-phenanthroline-10-yl, 1,9-phenanthrole Phenanthroline-2-yl, 1,9-phenanthroline-3-yl, 1,9-phenanthroline-4-yl, 1,9-phenanthroline-5-yl, 1,9-phenanthroline- 6-yl, 1,9-phenanthroline-7-yl, 1,9-phenanthroline-8-yl, 1,9-phenanthroline-10-yl, 1,10-phenanthroline-2- Base, 1,10-phenanthrolin-3-yl, 1,10-phenanthrolin-4-yl, 1,10-phenanthrolin-5-yl, 2,9-phenanthrolin-1-yl, 2,9-phenanthrolin-3-yl, 2,9-phenanthrolin-4-yl, 2,9-phenanthrolin-5-yl, 2,9-phenanthrolin-6-yl, 2, 9-phenanthrolin-7-yl, 2,9-phenanthrolin-8-yl, 2,9-phenanthrolin-10-yl, 2,8-phenanthrolin-1-yl, 2,8- Philo Phenanthroline-3-yl, 2,8-phenanthroline-4-yl, 2,8-phenanthroline-5-yl, 2,8-phenanthroline-6-yl, 2,8-phenanthroline- 7-yl, 2,8-phenanthroline-9-yl, 2,8-phenanthroline-10-yl, 2,7-phenanthroline-1-yl, 2,7-phenanthroline-3- Base, 2,7-phenanthrolin-4-yl, 2,7-phenanthrolin-5-yl, 2,7-phenanthrolin-6-yl, 2,7-phenanthrolin-8-yl, 2,7-phenanthrolin-9-yl and 2,7-phenanthrolin-10-yl; phenazinyl includes 1-phenazinyl and 2-phenazinyl; phenothiazinyl includes 1-phenothiazine Base, 2-phenothiazinyl, 3-phenothiazinyl, 4-phenothiazinyl and 10-phenothiazinyl; phenoxazinyl includes 1-phenoxazinyl, 2-phenoxazinyl, 3 - phenoxazinyl, 4-phenoxazinyl and 10-phenoxazinyl; oxazolyl includes 2-oxazolyl, 4-oxazolyl and 5-oxazolyl; oxadiazoles include 2-oxadio Azolyl and 5-oxadiazolyl; Furazanyl includes 3-furazanyl; Dibenzofuryl includes 1-dibenzofuryl, 2-dibenzofuryl, 3-dibenzofuryl and 4-Dibenzofuryl; dibenzothienyl includes 1-dibenzothienyl, 2-dibenzothienyl, 3-dibenzothienyl and 4-dibenzothienyl.
本文所用术语“(C1-C30)烷基”包括(C1-C20)烷基或(C1-C10)烷基,术语“(C6-C30)芳基”包括(C6-C20)芳基。术语“(C2-C30)杂芳基”包括(C2-C20)杂芳基,术语“(C3-C30)环烷基”包括(C3-C20)环烷基或(C3-C7)环烷基。术语“(C2-C30)烯基或炔基”包括(C2-C20)烯基或炔基,或者(C2-C10)烯基或炔基。The term "(C1-C30)alkyl" as used herein includes (C1-C20)alkyl or (C1-C10)alkyl, and the term "(C6-C30)aryl" includes (C6-C20)aryl. The term "(C2-C30) heteroaryl" includes (C2-C20) heteroaryl, and the term "(C3-C30) cycloalkyl" includes (C3-C20) cycloalkyl or (C3-C7) cycloalkyl . The term "(C2-C30)alkenyl or alkynyl" includes (C2-C20)alkenyl or alkynyl, or (C2-C10)alkenyl or alkynyl.
在本文所用表述“取代或未取代的”(或者“具有或不具有取代基”)中,术语“取代的(具有取代基)”表示未取代的取代基进一步被取代基取代。L1、Ar1、R1至R9、R以及R11至R22的各取代基还可被选自下组的一个或多个取代基取代:氘、卤素、卤素取代或未取代的(C1-C30)烷基、(C6-C30)芳基、(C6-C30)芳基取代或未取代的(C2-C30)杂芳基、5-至7-元杂环烷基、与一个或多个芳环稠合的5-至7-元杂环烷基、(C3-C30)环烷基、与一个或多个芳环稠合的(C6-C30)环烷基、RaRbRcSi-、(C2-C30)烯基、(C2-C30)炔基、氰基、咔唑基、-NRdRe、-BRfRg、-PRhRi、-P(=O)RjRk、(C6-C30)芳基(C1-C30)烷基、(C1-C30)烷基(C6-C30)芳基、RlT-、RmC(=O)-、RmC(=O)O-、羧基、硝基和羟基,其中Ra至Rl独立地表示(C1-C30)烷基、(C6-C30)芳基或(C2-C30)杂芳基;T是S或O;Rm表示(C1-C30)烷基、(C1-C30)烷氧基、(C6-C30)芳基或(C6-C30)芳氧基。In the expression "substituted or unsubstituted" (or "with or without a substituent") used herein, the term "substituted (with a substituent)" means that the unsubstituted substituent is further substituted with a substituent. Each substituent of L 1 , Ar 1 , R 1 to R 9 , R, and R 11 to R 22 may also be substituted by one or more substituents selected from the group consisting of deuterium, halogen, halogen substituted or unsubstituted ( C1-C30) alkyl, (C6-C30) aryl, (C6-C30) aryl substituted or unsubstituted (C2-C30) heteroaryl, 5- to 7-membered heterocycloalkyl, and one or 5- to 7-membered heterocycloalkyl, (C3-C30)cycloalkyl, (C6-C30)cycloalkyl fused to one or more aromatic rings, R a R b R c Si-, (C2-C30) alkenyl, (C2-C30) alkynyl, cyano, carbazolyl, -NR d R e , -BR f R g , -PR h R i , -P(= O)R j R k , (C6-C30)aryl(C1-C30)alkyl, (C1-C30)alkyl(C6-C30)aryl, R l T-, R m C(=O)- , R m C (= O) O-, carboxyl, nitro and hydroxyl, wherein R a to R l independently represent (C1-C30) alkyl, (C6-C30) aryl or (C2-C30) heteroaryl base; T is S or O; R m represents (C1-C30) alkyl, (C1-C30) alkoxy, (C6-C30) aryl or (C6-C30) aryloxy.
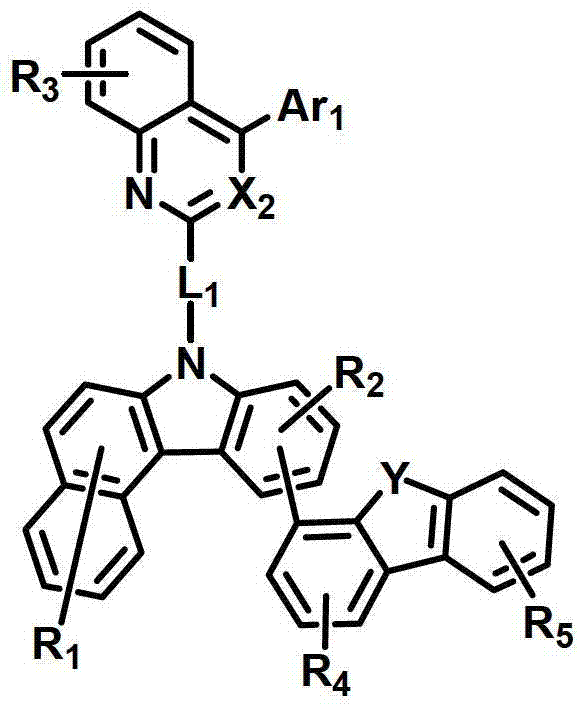
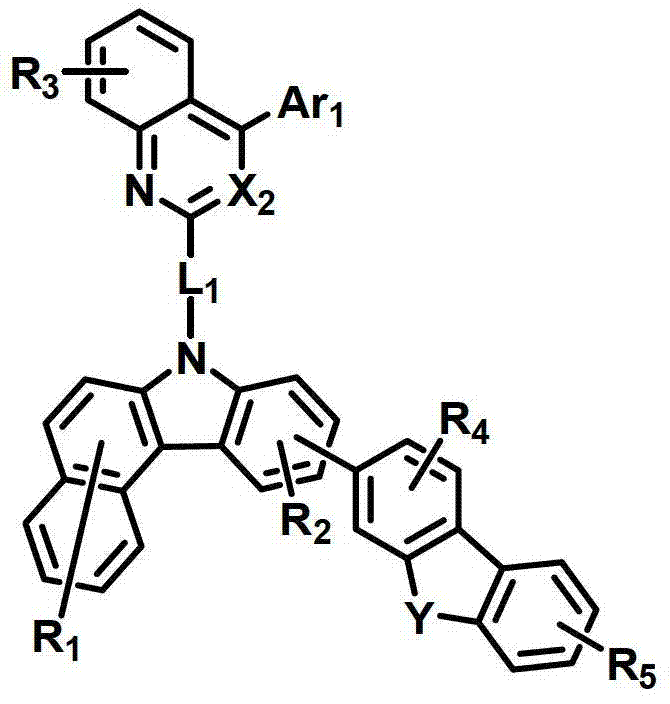
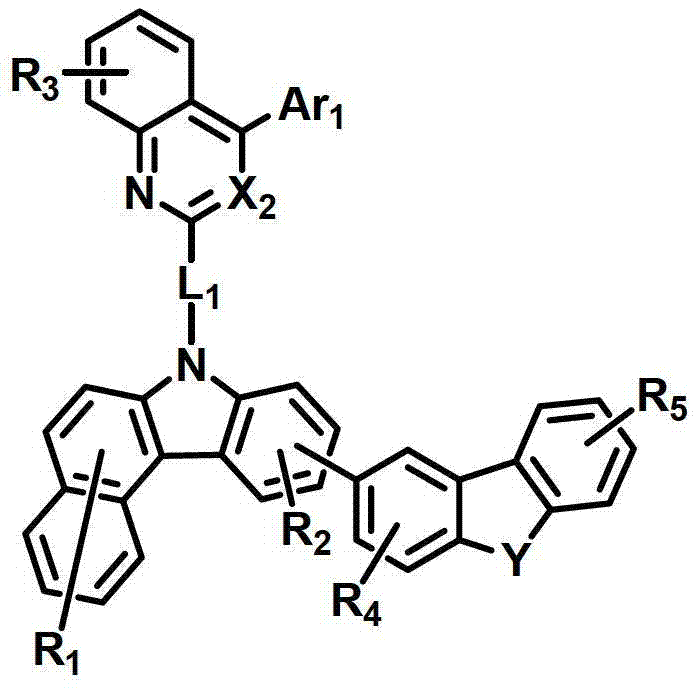
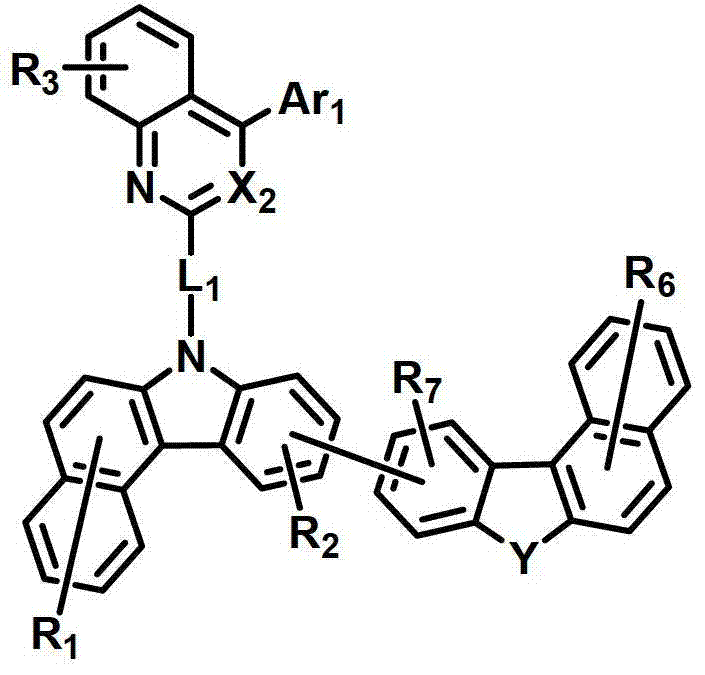
所述有机电致发光化合物可由以下化学式2-9来表示。The organic electroluminescent compound may be represented by the following Chemical Formulas 2-9.
化学式2chemical formula 2
化学式3chemical formula 3
化学式4chemical formula 4
化学式5chemical formula 5
化学式6chemical formula 6
化学式7chemical formula 7
化学式8chemical formula 8
化学式9chemical formula 9
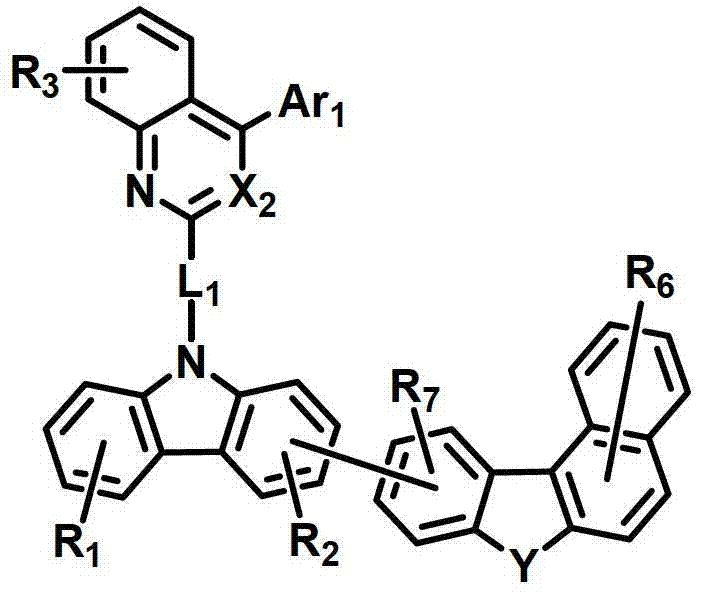
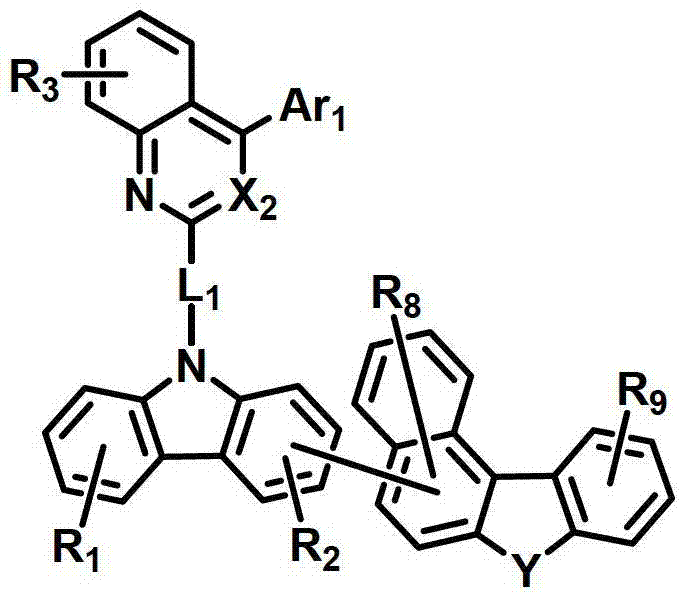
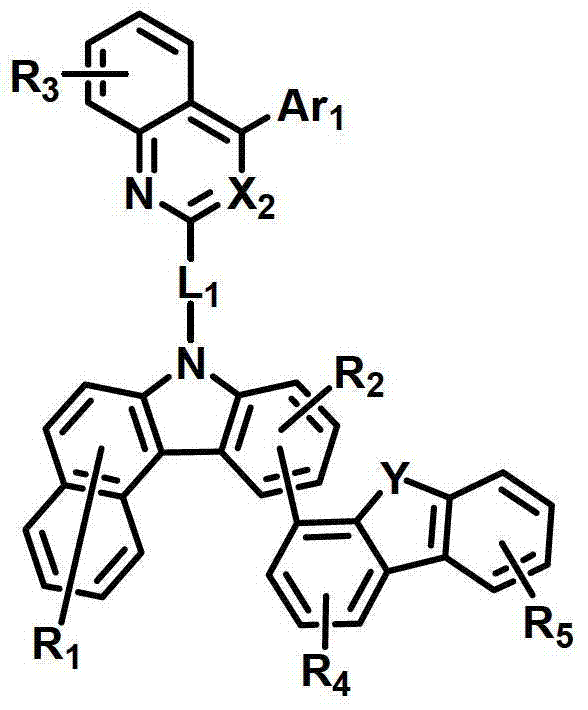
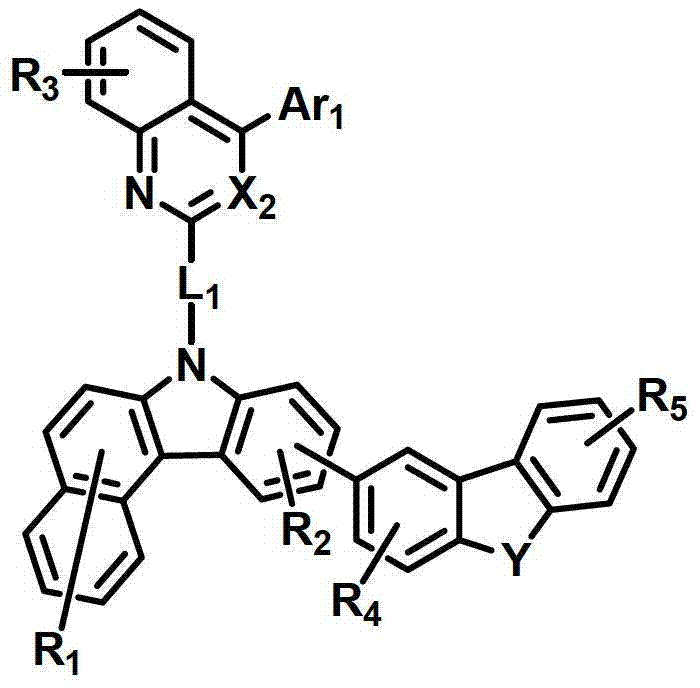
式中,X2是N或CH;Y是-O-、-S-、-C(R11R12)-或-N(R15)-;L1表示单键、取代或未取代的(C6-C30)亚芳基、或取代或未取代的(C2-C30)杂亚芳基;Ar1表示氢、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基、或取代或未取代的(C2-C30)杂芳基;R1至R9独立地表示氢、氘、取代或未取代的(C6-C30)芳基、取代或未取代的(C2-C30)杂芳基、NR16R17或SiR18R19R20;R11、R12、R15和R16至R20独立地表示氢、氘、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基或取代或未取代的(C2-C30)杂芳基;或者R16和R17可通过具有或不具有稠环的取代或未取代的(C3-C30)亚烯基或取代或未取代的(C3-C30)亚烷基相连接形成脂环或单环或多环芳环,并且该脂环和单环或多环芳环的碳原子可以被一个或多个选自N、O和S的杂原子取代。In the formula, X 2 is N or CH; Y is -O-, -S-, -C(R 11 R 12 )- or -N(R 15 )-; L 1 represents a single bond, substituted or unsubstituted ( C6-C30) arylene, or substituted or unsubstituted (C2-C30) heteroarylene; Ar 1 represents hydrogen, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6- C30) aryl, or substituted or unsubstituted (C2-C30) heteroaryl; R 1 to R 9 independently represent hydrogen, deuterium, substituted or unsubstituted (C6-C30) aryl, substituted or unsubstituted (C2-C30) Heteroaryl, NR 16 R 17 or SiR 18 R 19 R 20 ; R 11 , R 12 , R 15 and R 16 to R 20 independently represent hydrogen, deuterium, substituted or unsubstituted (C1- C30) alkyl, substituted or unsubstituted (C6-C30) aryl or substituted or unsubstituted (C2-C30) heteroaryl; or R 16 and R 17 can be substituted or unsubstituted with or without fused rings A substituted (C3-C30) alkenylene group or a substituted or unsubstituted (C3-C30) alkylene group is connected to form an alicyclic ring or a monocyclic or polycyclic aromatic ring, and the alicyclic ring and the monocyclic or polycyclic aromatic ring The carbon atoms in may be substituted by one or more heteroatoms selected from N, O and S.
具体而言,X2表示N或CH;Y表示-O-、-S-、-C(R11R12)-或-N(R15)-;Specifically, X 2 represents N or CH; Y represents -O-, -S-, -C(R 11 R 12 )- or -N(R 15 )-;
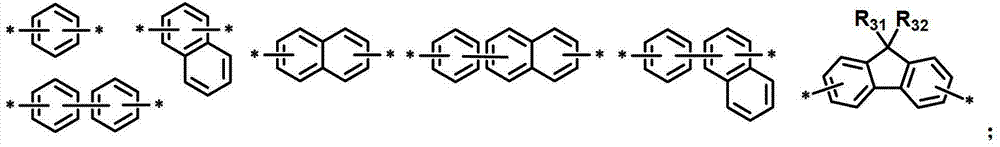
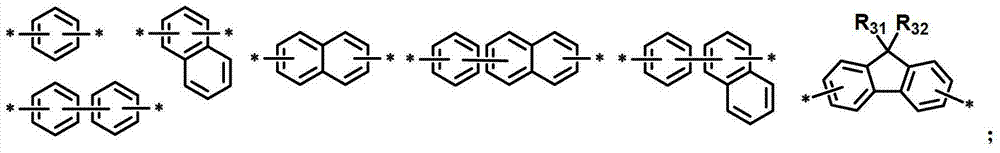
L1表示单键或者选自以下结构亚芳基: L represents a single bond or is selected from the following structures of arylene:
R31和R32独立地表示甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基、正己基、正庚基、正辛基、2-乙基己基、正壬基、癸基、十二烷基、十六烷基、三氟甲基、全氟乙基、三氟乙基、全氟丙基、全氟丁基、苯基、萘基、吡啶基或喹啉基;R 31 and R 32 independently represent methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isopentyl, n-hexyl, n-heptyl, n- Octyl, 2-ethylhexyl, n-nonyl, decyl, dodecyl, hexadecyl, trifluoromethyl, perfluoroethyl, trifluoroethyl, perfluoropropyl, perfluorobutyl , phenyl, naphthyl, pyridyl or quinolinyl;
Ar1表示氢、甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基、正己基、正庚基、正辛基、2-乙基己基、正壬基、癸基、十二烷基、十六烷基、三氟甲基、全氟乙基、三氟乙基、全氟丙基、全氟丁基、苯基、联苯基、三联苯基、萘基、9,9-二苯基芴基、9,9-二甲基芴基、荧蒽基、吡啶基、二苯并呋喃基、二苯并噻吩基或N-苯基咔唑基,并且Ar1的苯基、联苯基、三联苯基、萘基和咔唑基还可被一个或多个选自下组的取代基取代:氘、氟、甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基、正己基、正庚基、正辛基、2-乙基己基、正壬基、癸基、十二烷基、十六烷基、三氟甲基、全氟乙基、三氟乙基、全氟丙基、全氟丁基、三苯基甲硅烷基、三甲基甲硅烷基、二甲基苯基甲硅烷基、二苯基甲基甲硅烷基、苯基、萘基、9,9-二苯基芴基、9,9-二甲基芴基、苯基吡啶基、咔唑基、荧蒽基、二苯并呋喃基和二苯并噻吩基;以及Ar 1 represents hydrogen, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isopentyl, n-hexyl, n-heptyl, n-octyl, 2-Ethylhexyl, n-nonyl, decyl, dodecyl, hexadecyl, trifluoromethyl, perfluoroethyl, trifluoroethyl, perfluoropropyl, perfluorobutyl, phenyl , biphenyl, terphenyl, naphthyl, 9,9-diphenylfluorenyl, 9,9-dimethylfluorenyl, fluoranthene, pyridyl, dibenzofuranyl, dibenzothienyl Or N-phenylcarbazolyl, and the phenyl, biphenyl, terphenyl, naphthyl and carbazolyl of Ar 1 can also be substituted by one or more substituents selected from the group: deuterium, fluorine, Methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isopentyl, n-hexyl, n-heptyl, n-octyl, 2-ethylhexyl , n-nonyl, decyl, dodecyl, hexadecyl, trifluoromethyl, perfluoroethyl, trifluoroethyl, perfluoropropyl, perfluorobutyl, triphenylsilyl, Trimethylsilyl, Dimethylphenylsilyl, Diphenylmethylsilyl, Phenyl, Naphthyl, 9,9-Diphenylfluorenyl, 9,9-Dimethylfluorenyl , phenylpyridyl, carbazolyl, fluoranthenyl, dibenzofuranyl and dibenzothienyl; and
R1至R9独立地表示氢、氘、苯基、吡啶基、二苯并呋喃基、二苯并噻吩基、氨基或咔唑基;R11、R12和R15独立地表示氢、氘、甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基、正己基、正庚基、正辛基、2-乙基己基、正壬基、癸基、十二烷基、十六烷基、三氟甲基、全氟乙基、三氟乙基、全氟丙基、全氟丁基、苯基、联苯基、9,9-二苯基芴基、9,9-二甲基芴基、萘基、吡啶基、N-苯基咔唑基或喹啉基,并且R11、R12和R15的苯基还可被一个或多个选自下组的取代基取代:氘、卤素、甲基、乙基、正丙基、异丙基、正丁基、异丁基、叔丁基、正戊基、异戊基、正己基、正庚基、正辛基、2-乙基己基、正壬基、癸基、十二烷基、十六烷基、三氟甲基、全氟乙基、三氟乙基、全氟丙基、全氟丁基、苯基和萘基,并且R11和R12可相互连接形成环。R 1 to R 9 independently represent hydrogen, deuterium, phenyl, pyridyl, dibenzofuryl, dibenzothienyl, amino or carbazolyl; R 11 , R 12 and R 15 independently represent hydrogen, deuterium , methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl, isopentyl, n-hexyl, n-heptyl, n-octyl, 2-ethyl Hexyl, n-nonyl, decyl, dodecyl, hexadecyl, trifluoromethyl, perfluoroethyl, trifluoroethyl, perfluoropropyl, perfluorobutyl, phenyl, biphenyl , 9,9-diphenylfluorenyl, 9,9-dimethylfluorenyl, naphthyl, pyridyl, N-phenylcarbazolyl or quinolinyl, and the benzene of R 11 , R 12 and R 15 The group can also be substituted by one or more substituents selected from the group consisting of deuterium, halogen, methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl, tert-butyl, n-pentyl , isopentyl, n-hexyl, n-heptyl, n-octyl, 2-ethylhexyl, n-nonyl, decyl, dodecyl, hexadecyl, trifluoromethyl, perfluoroethyl, three Fluoroethyl, perfluoropropyl, perfluorobutyl, phenyl and naphthyl, and R 11 and R 12 may be connected to each other to form a ring.
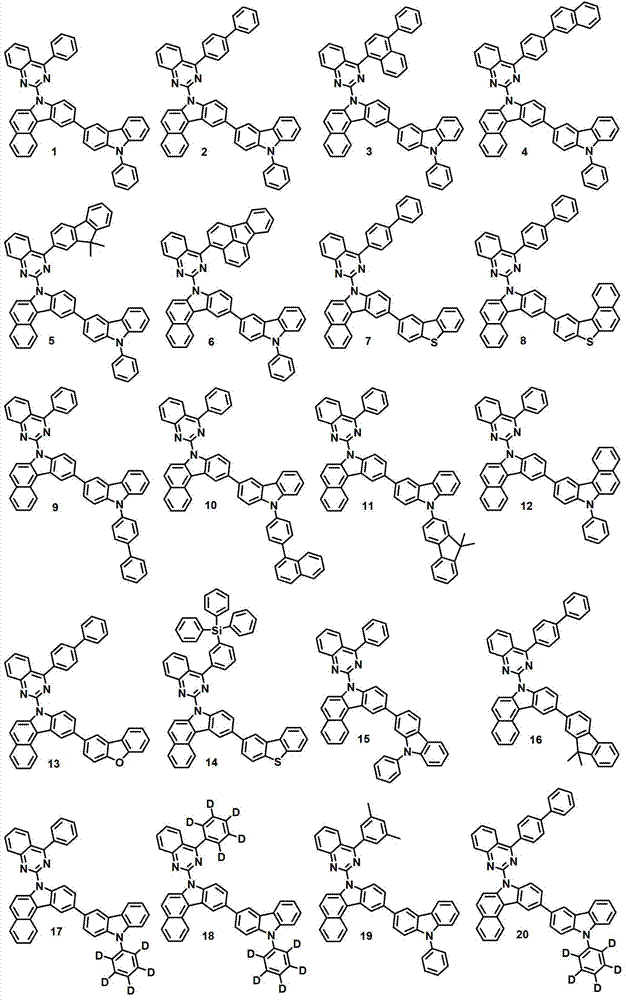
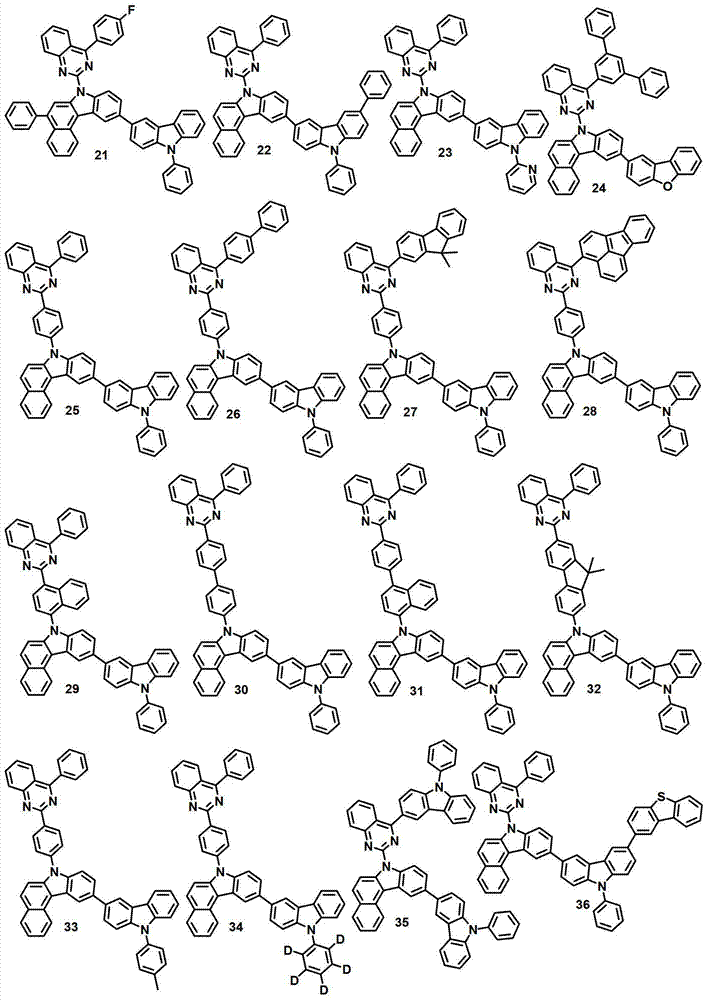
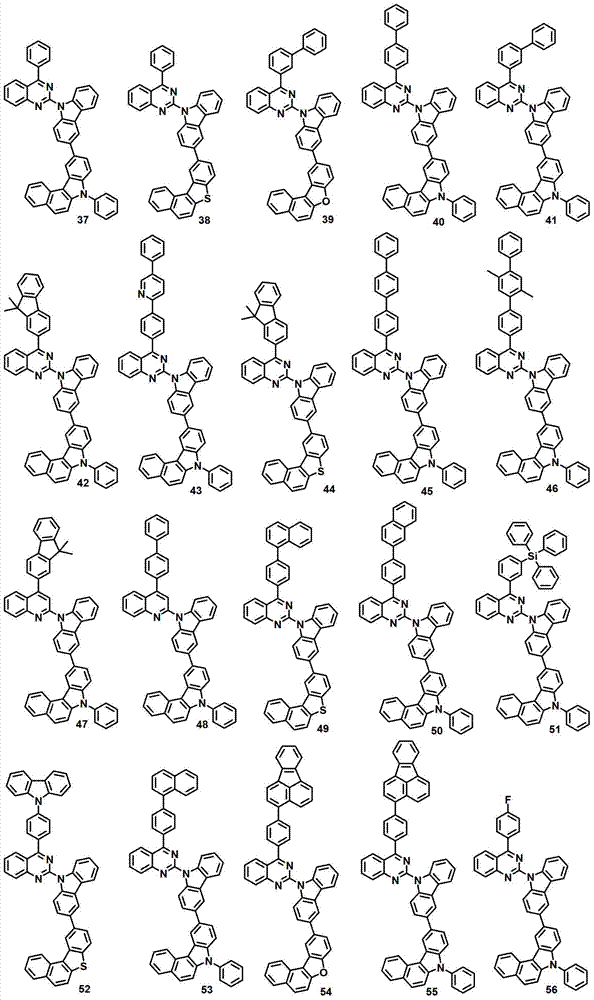
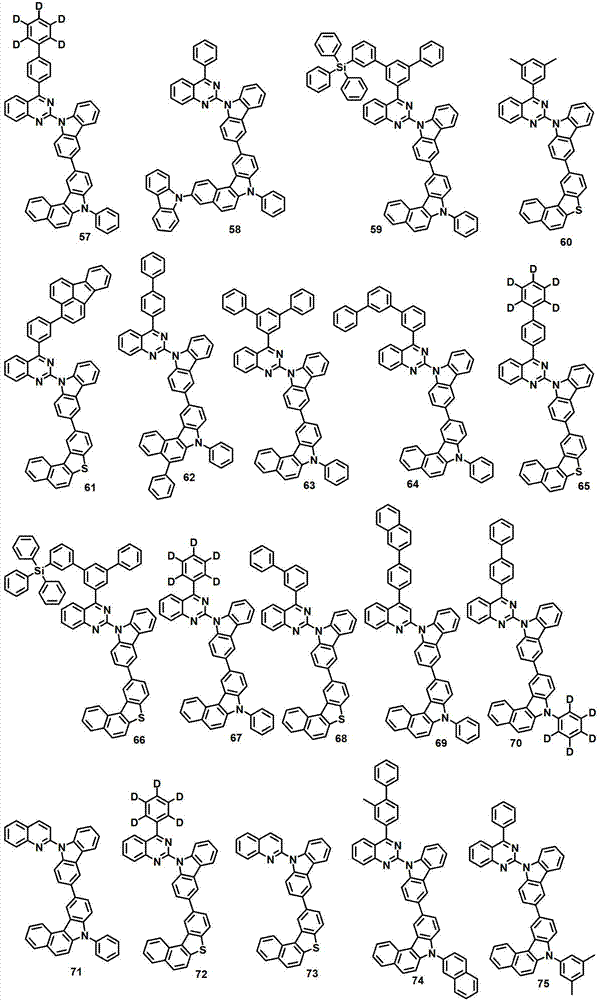
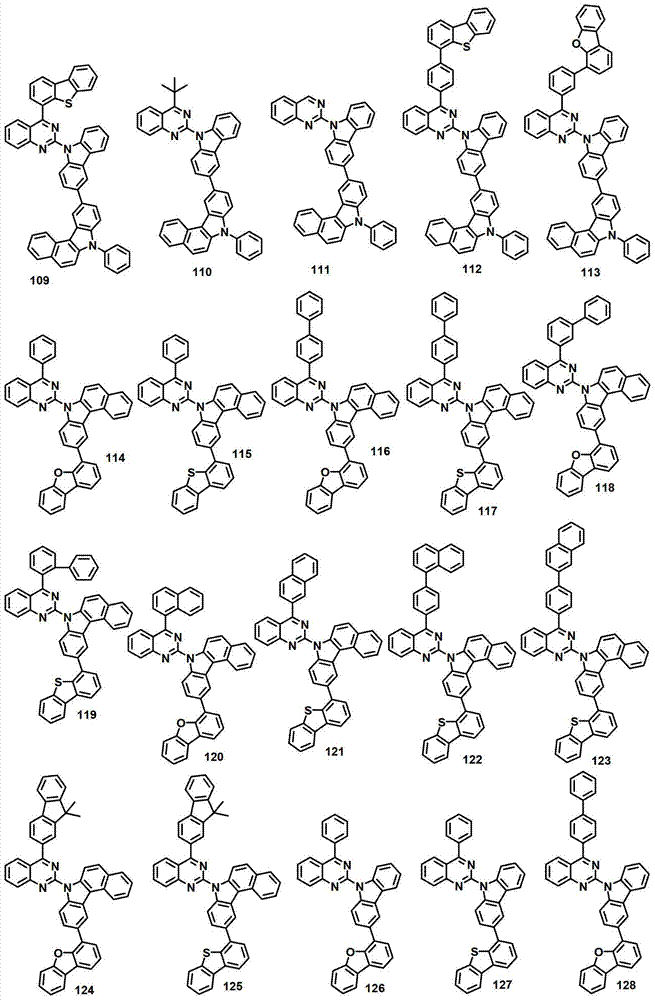
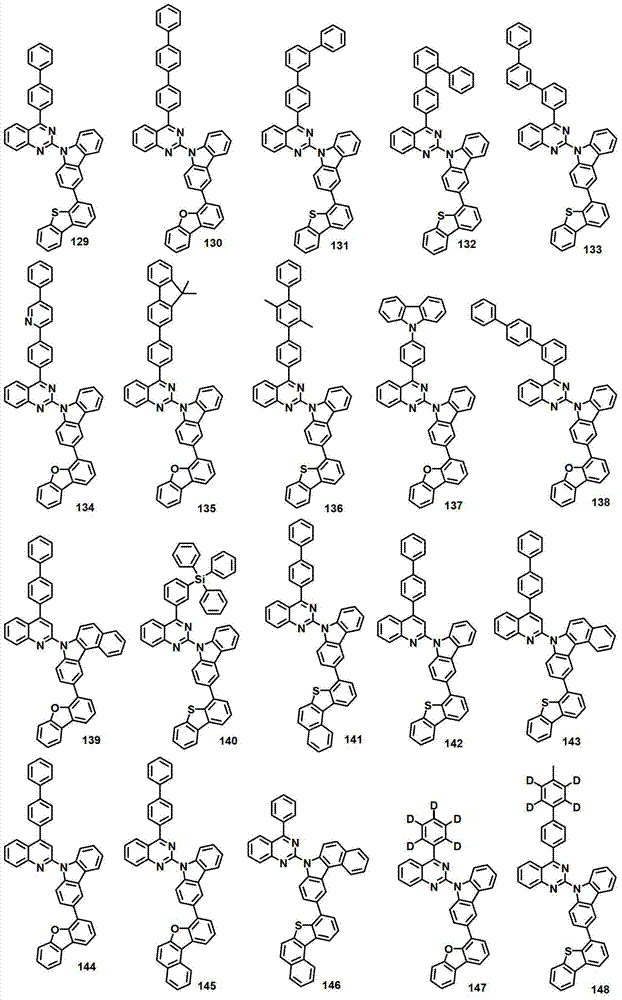
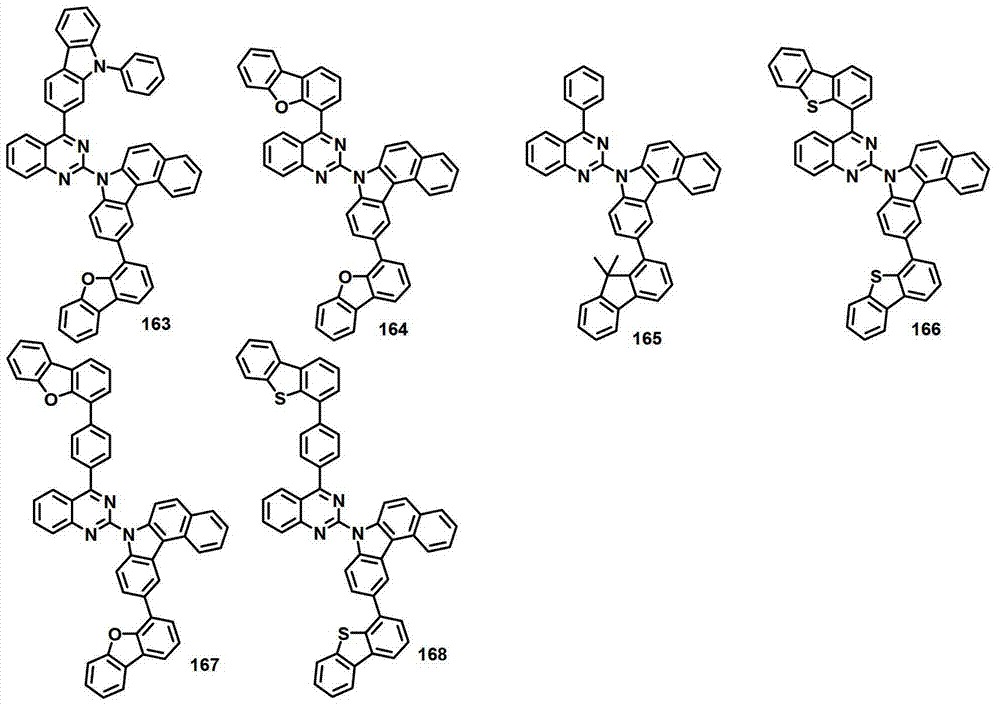
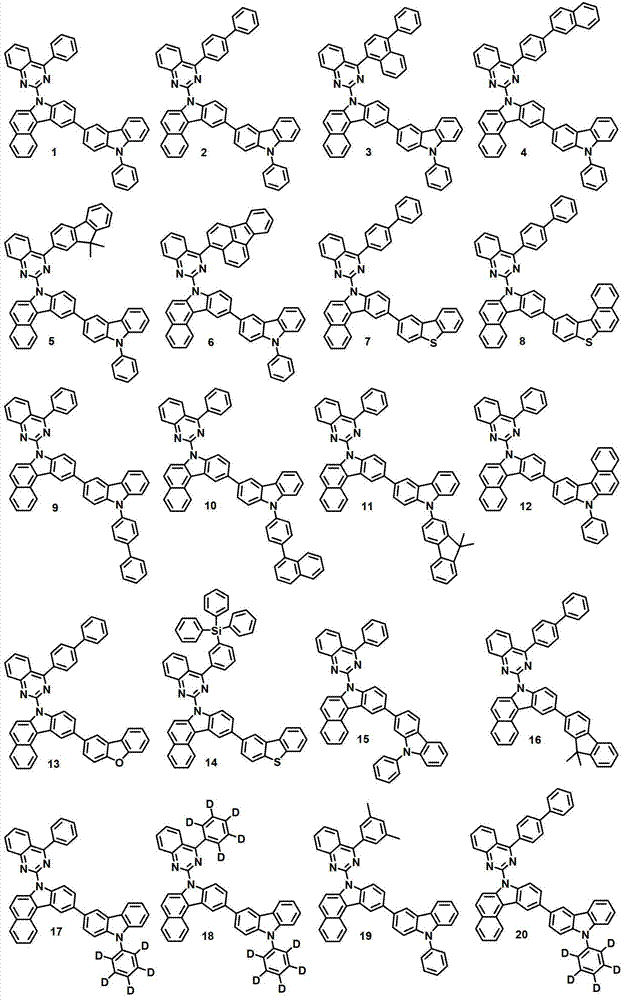
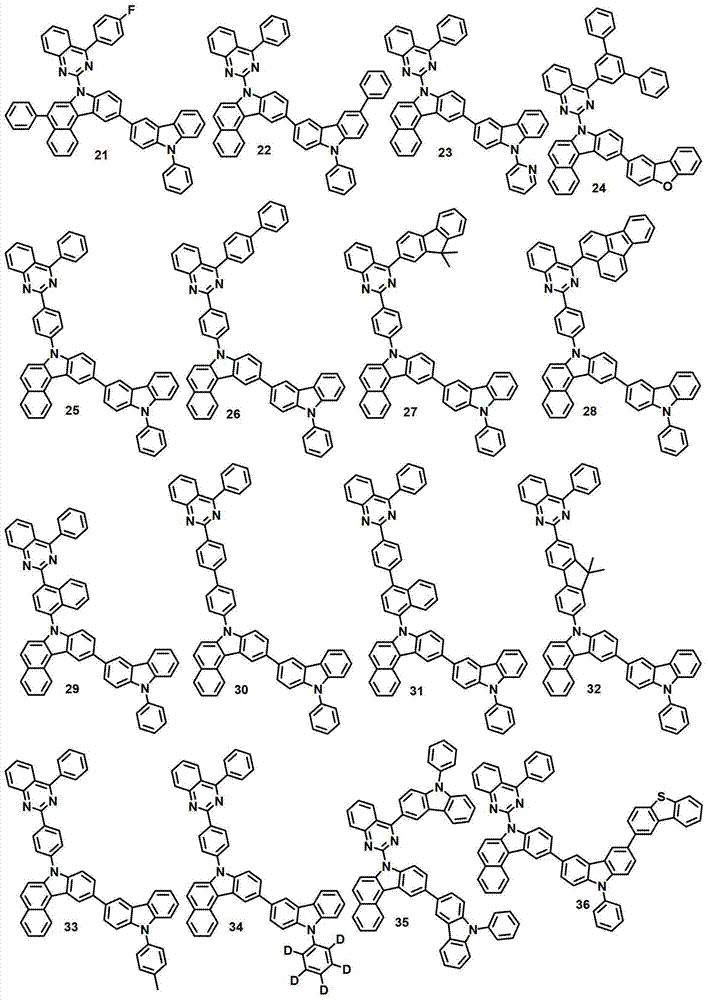
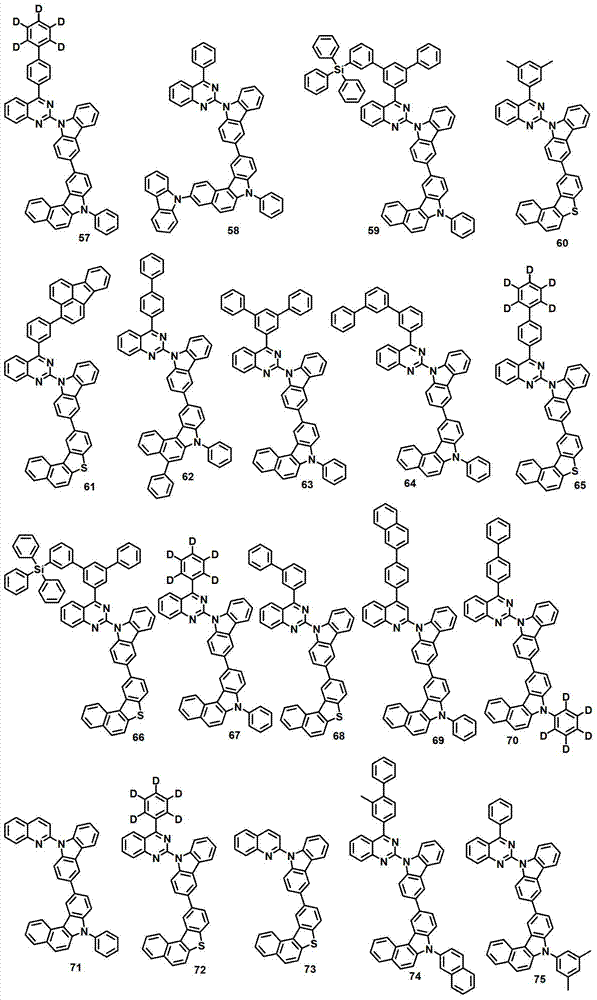
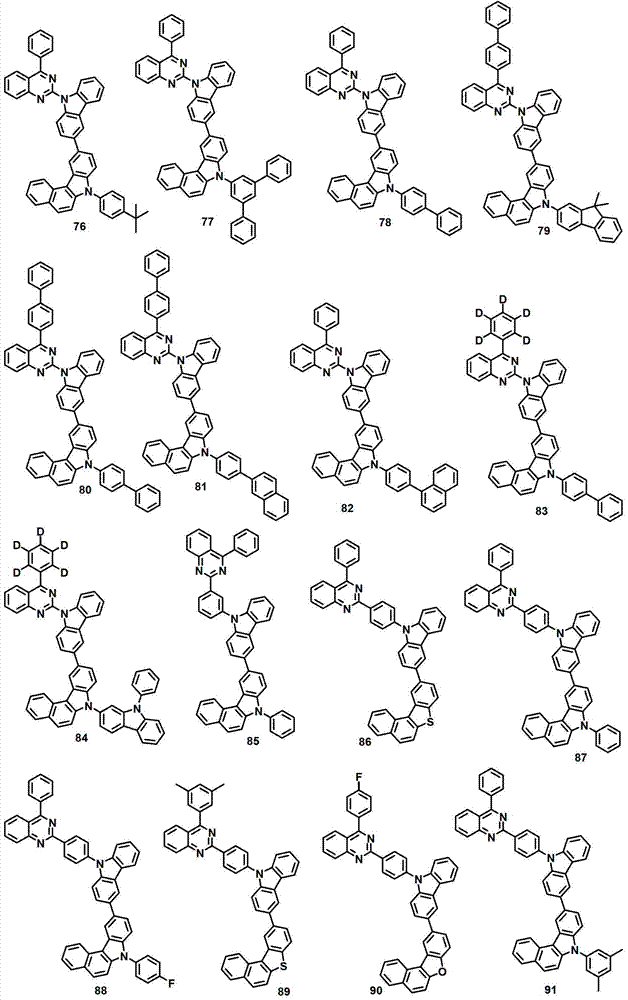
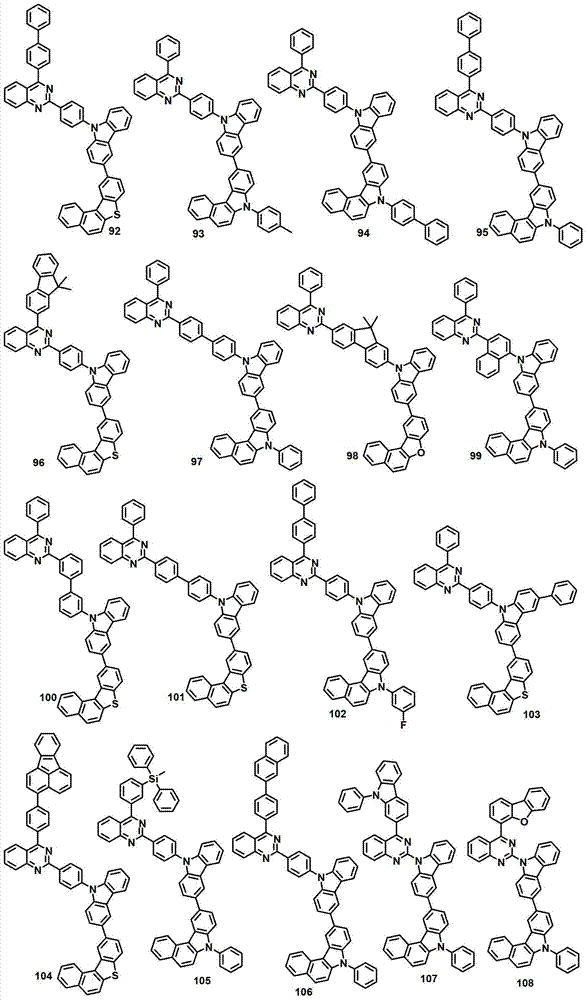
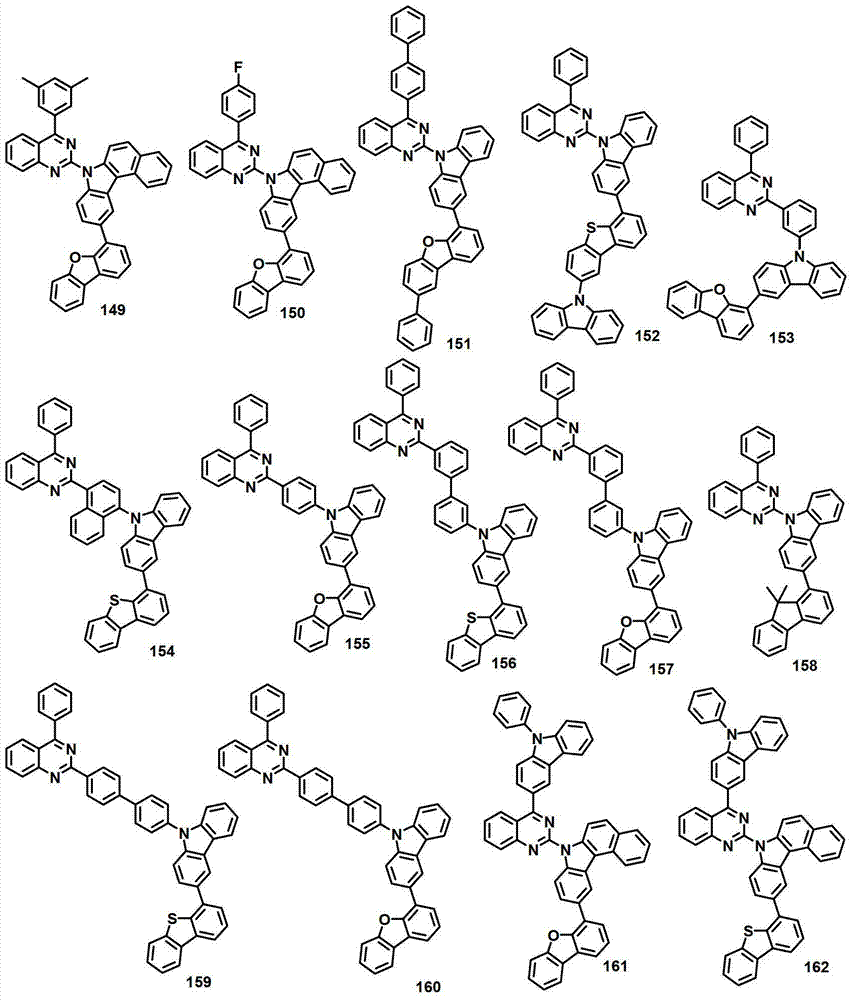
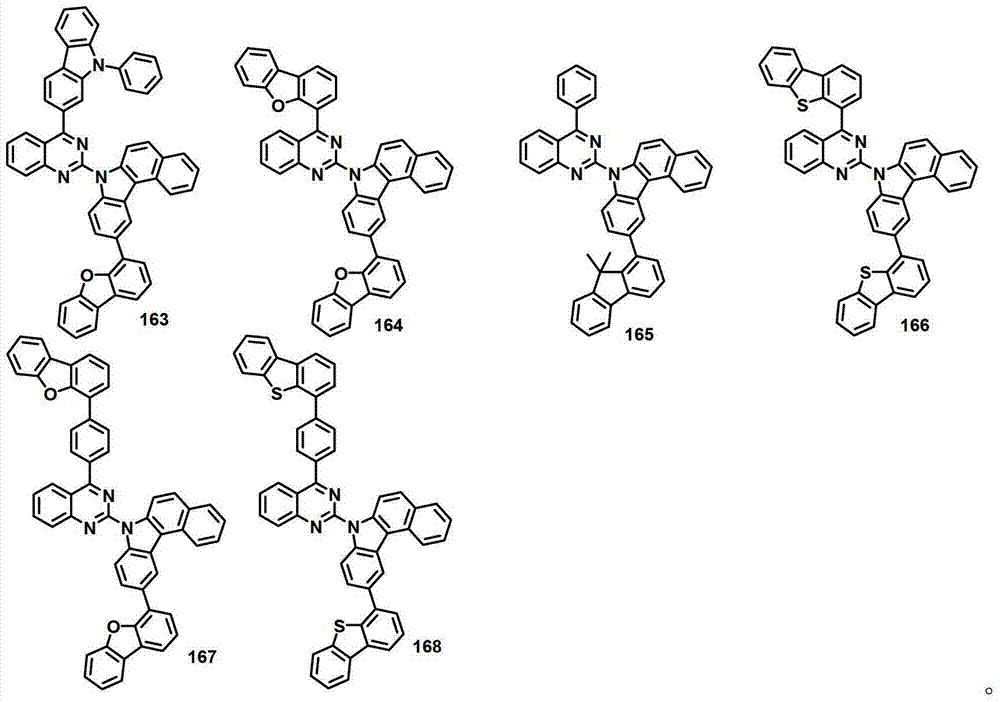
本发明的有机电致发光化合物可列举为下述化合物,但它们并不对本发明构成限制:The organic electroluminescent compound of the present invention can be listed as following compound, but they do not limit the present invention:
可以如(例如)以下方案1所示来制备本发明的有机电致发光化合物,但不限于此。The organic electroluminescent compound of the present invention can be prepared, for example, as shown in Scheme 1 below, but is not limited thereto.
[方案1][plan 1]
在方案1中,环A、X1、X2、L1、Ar1、R1、R2、R3、a、b、c和Z与化学式1所定义的相同;Hal表示卤素。In Scheme 1, rings A, X 1 , X 2 , L 1 , Ar 1 , R 1 , R 2 , R 3 , a, b, c, and Z are the same as defined in Chemical Formula 1; Hal represents halogen.
提供了一种有机电致发光器件,它包含第一电极;第二电极;和插入所述第一电极和第二电极之间的一层或多层有机层,其中所述有机层包含由化学式1表示的一种或多种有机电致发光化合物。所述有机层包括电致发光层,并且化学式1的有机电致发光化合物可用作所述电致发光层中的基质。An organic electroluminescent device is provided, comprising a first electrode; a second electrode; and one or more organic layers interposed between the first electrode and the second electrode, wherein the organic layer comprises the formula One or more organic electroluminescent compounds represented by 1. The organic layer includes an electroluminescent layer, and the organic electroluminescent compound of Chemical Formula 1 may be used as a host in the electroluminescent layer.
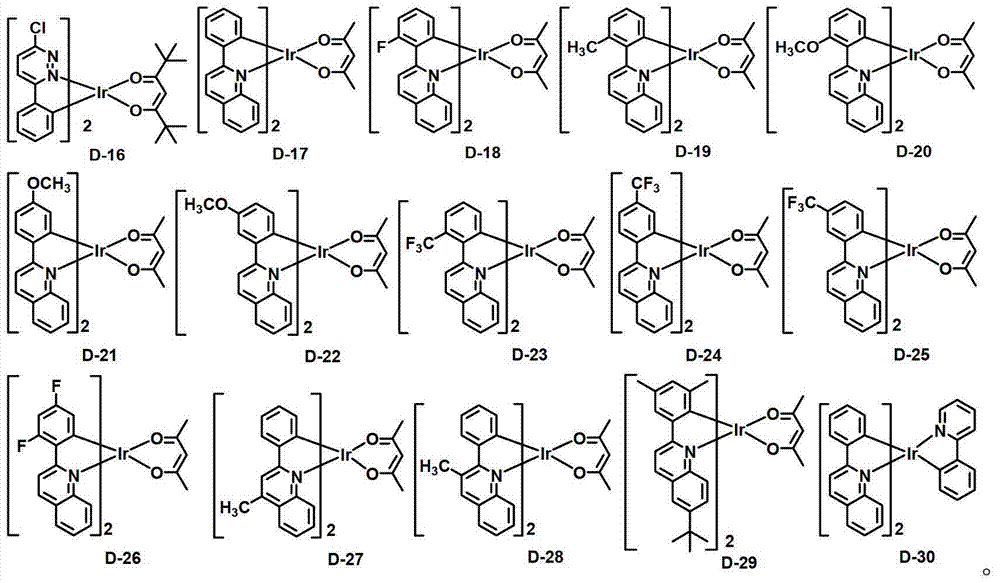
在电致发光层中,当化学式1的有机电致发光化合物用作基质时,包含一种或多种磷光掺杂剂。用于本发明的有机电致发光器件的磷光掺杂剂并没有具体限制,但可选自如下化学式10所表示的化合物:In the electroluminescent layer, when the organic electroluminescent compound of Chemical Formula 1 is used as a host, one or more phosphorescent dopants are contained. The phosphorescent dopant used in the organic electroluminescent device of the present invention is not specifically limited, but can be selected from compounds represented by the following chemical formula 10:
化学式10chemical formula 10
M1L101L102L103 M 1 L 101 L 102 L 103
其中:in:
M1是选自元素周期表第7族、第8族、第9族、第10族、第11族、第13族、第14族、第15族和第16族的金属,配体L101、L102、和L103独立地选自以下结构:M 1 is a metal selected from Group 7, Group 8, Group 9, Group 10, Group 11, Group 13, Group 14, Group 15 and Group 16 of the Periodic Table of the Elements, ligand L 101 , L 102 , and L 103 are independently selected from the following structures:
R201至R203独立地表示氢、氘、卤素取代或未取代的(C1-C30)烷基、(C1-C30)烷基取代或未取代的(C6-C30)芳基或卤素;R 201 to R 203 independently represent hydrogen, deuterium, halogen substituted or unsubstituted (C1-C30) alkyl, (C1-C30) alkyl substituted or unsubstituted (C6-C30) aryl or halogen;
R204至R219独立地表示氢、氘、取代或未取代的(C1-C30)烷基、取代或未取代的(C1-C30)烷氧基、取代或未取代的(C3-C30)环烷基、取代或未取代的(C2-C30)烯基、取代或未取代的(C6-C30)芳基、取代或未取代的单-(C1-C30)烷基氨基或者取代或未取代的二-(C1-C30)烷基氨基、取代或未取代的单-(C6-C30)芳基氨基或者取代或未取代的二-(C6-C30)芳基氨基、SF5、取代或未取代的三(C1-C30)烷基甲硅烷基、取代或未取代的二(C1-C30)烷基(C6-C30)芳基甲硅烷基、取代或未取代的三(C6-C30)芳基甲硅烷基、氰基或者卤素;R 204 to R 219 independently represent hydrogen, deuterium, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C1-C30) alkoxy, substituted or unsubstituted (C3-C30) ring Alkyl, substituted or unsubstituted (C2-C30)alkenyl, substituted or unsubstituted (C6-C30)aryl, substituted or unsubstituted mono-(C1-C30)alkylamino or substituted or unsubstituted Di-(C1-C30)alkylamino, substituted or unsubstituted mono-(C6-C30)arylamino or substituted or unsubstituted di-(C6-C30)arylamino, SF 5 , substituted or unsubstituted Tri(C1-C30)alkylsilyl, substituted or unsubstituted di(C1-C30)alkyl(C6-C30)arylsilyl, substituted or unsubstituted tri(C6-C30)aryl Silyl, cyano or halogen;
R220至R223独立地表示氢、氘、卤素取代或未取代的(C1-C30)烷基或者(C1-C30)烷基取代或未取代的(C6-C30)芳基;R 220 to R 223 independently represent hydrogen, deuterium, halogen substituted or unsubstituted (C1-C30) alkyl or (C1-C30) alkyl substituted or unsubstituted (C6-C30) aryl;
R224和R225独立地表示氢、氘、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基或卤素,或者R224和R225可通过具有或不具有稠环的(C3-C12)亚烯基或(C3-C12)亚烷基连接形成脂环和单环或多环芳环;R 224 and R 225 independently represent hydrogen, deuterium, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl or halogen, or R 224 and R 225 can be represented by having or A (C3-C12) alkenylene group or (C3-C12) alkylene group without a fused ring is connected to form an alicyclic ring and a monocyclic or polycyclic aromatic ring;
R226表示取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基、取代或未取代的(C2-C30)杂芳基或卤素;R 226 represents substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl, substituted or unsubstituted (C2-C30) heteroaryl or halogen;
R227至R229独立地表示氢、氘、取代或未取代的(C1-C30)烷基、取代或未取代的(C6-C30)芳基或卤素;以及R 227 to R 229 independently represent hydrogen, deuterium, substituted or unsubstituted (C1-C30) alkyl, substituted or unsubstituted (C6-C30) aryl or halogen; and
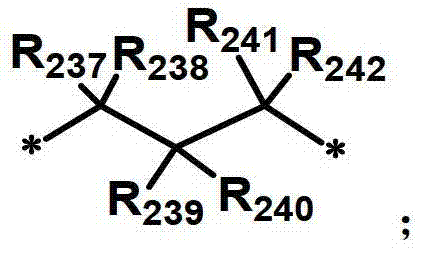
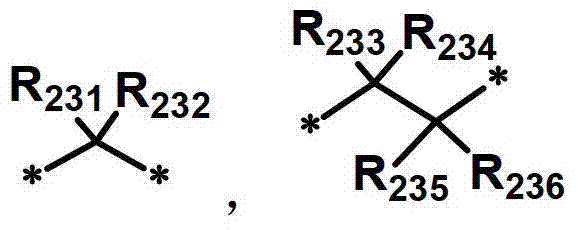
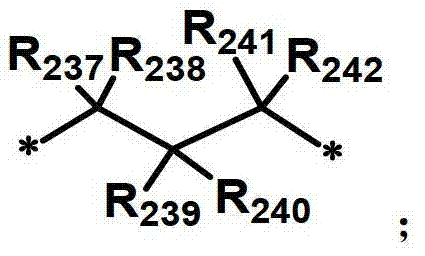
R231至R242独立地表示氢、氘、卤素取代或未取代的(C1-C30)烷基、(C1-C30)烷氧基、卤素、取代或未取代的(C6-C30)芳基、氰基、取代或未取代的(C3-C30)环烷基,或者它们可通过亚烷基或者亚烯基与相邻取代基连接形成螺环或稠环,或者可通过亚烷基或者亚烯基与R207或R208连接形成饱和或不饱和的稠环。R 231 to R 242 independently represent hydrogen, deuterium, halogen substituted or unsubstituted (C1-C30) alkyl, (C1-C30) alkoxy, halogen, substituted or unsubstituted (C6-C30) aryl, Cyano, substituted or unsubstituted (C3-C30) cycloalkyl groups, or they can be connected to adjacent substituents to form spiro or fused rings through alkylene or alkenylene, or they can be connected through alkylene or alkenene The group is connected with R207 or R208 to form a saturated or unsaturated condensed ring.
化学式10的掺杂剂化合物可列举为以下化合物,但不限于此:The dopant compound of Chemical Formula 10 can be listed as the following compounds, but not limited thereto:
在本发明的有机电子器件中,除了化学式1所表示的有机电致发光化合物以外,所述有机层还可同时包括一种或多种选自芳胺化合物和苯乙烯基芳胺化合物的化合物。所述芳胺化合物或苯乙烯基芳胺化合物的例子参见韩国专利申请第10-2008-0123276、10-2008-0107606或10-2008-0118428号,但不限于此。In the organic electronic device of the present invention, in addition to the organic electroluminescent compound represented by Chemical Formula 1, the organic layer may also include one or more compounds selected from arylamine compounds and styrylarylamine compounds. For examples of the arylamine compound or styrylarylamine compound, see Korean Patent Application No. 10-2008-0123276, 10-2008-0107606, or 10-2008-0118428, but is not limited thereto.
此外,在本发明的有机电致发光器件中,除了化学式1所表示的有机电致发光化合物以外,所述有机层还可包括一种或多种选自元素周期表第1族、第2族的有机金属、第四周期和第五周期过渡金属、镧系金属和d-过渡元素的金属或络合物。所述有机层可包含电致发光层和电荷产生层。In addition, in the organic electroluminescent device of the present invention, in addition to the organic electroluminescent compound represented by Chemical Formula 1, the organic layer may also include one or more elements selected from Group 1 and Group 2 of the Periodic Table of Elements. metals or complexes of organometallics, fourth and fifth period transition metals, lanthanide metals and d-transition elements. The organic layer may include an electroluminescent layer and a charge generation layer.
另外,除了所述化学式1表示的有机电致发光化合物之外,所述有机层还可同时包括一种或多种发射蓝光、绿光或红光的有机电致发光层,以实现发射白光的有机电致发光器件。发蓝光、绿光或红光的化合物的例子可以是韩国专利申请第10-2008-0123276、10-2008-0107606或10-2008-0118428号所述的化合物,但不限于此。In addition, in addition to the organic electroluminescent compound represented by the chemical formula 1, the organic layer may also include one or more organic electroluminescent layers that emit blue light, green light or red light, so as to achieve white light emission. Organic electroluminescent devices. Examples of compounds emitting blue, green, or red light may be compounds described in Korean Patent Application No. 10-2008-0123276, 10-2008-0107606, or 10-2008-0118428, but are not limited thereto.
在本发明的有机电致发光器件中,选自硫属化物层、金属卤化物层和金属氧化物层的层(以下称为“表面层”)可放置在电极对中的一个或两个电极的内表面上。更具体地说,硅或铝的金属硫属化物(包括氧化物)层可置于电致发光介质层的阳极表面上,金属卤化物层或金属氧化物层可置于所述电致发光介质层的阴极表面上。从而得到工作稳定性。例如,硫属化物可以是SiOx(1≤x≤2)、AlOx(1≤x≤1.5)、SiON、SiAlON等。例如,金属卤化物可以是LiF、MgF2、CaF2、稀土金属氟化物等。例如,金属氧化物可以是Cs2O、Li2O、MgO、SrO、BaO、CaO等。In the organic electroluminescence device of the present invention, a layer selected from a chalcogenide layer, a metal halide layer, and a metal oxide layer (hereinafter referred to as "surface layer") may be placed on one or both electrodes of the electrode pair on the inner surface. More specifically, a metal chalcogenide (including oxide) layer of silicon or aluminum can be placed on the anode surface of the electroluminescent medium layer, and a metal halide layer or a metal oxide layer can be placed on the electroluminescent medium layer on the cathode surface. resulting in job stability. For example, the chalcogenide may be SiOx (1≤x≤2), AlOx (1≤x≤1.5), SiON, SiAlON, or the like. For example, the metal halides may be LiF, MgF 2 , CaF 2 , rare earth metal fluorides, and the like. For example, the metal oxide may be Cs2O , Li2O , MgO, SrO, BaO, CaO, or the like.
在本发明的有机电致发光器件中,也优选在所制备的电极对的至少一个表面上设置电子传输化合物和还原性掺杂剂的混合区,或者空穴传输化合物和氧化性掺杂剂的混合区。在该情况下,由于电子传输化合物被还原成阴离子,从而促进电子从混合区注入并传输到电致发光介质。另外,由于空穴传输化合物被氧化形成阳离子,从而促进空穴从混合区注入并传输到电致发光介质。优选的氧化性掺杂剂包括各种路易斯酸和受体化合物。优选的还原性掺杂剂包括碱金属、碱金属化合物、碱土金属、稀土金属及其混合物。此外,具有两层或更多层电致发光层的发白光的电致发光器件可通过使用还原性掺杂剂层作为电荷产生层来制备。In the organic electroluminescent device of the present invention, it is also preferred that a mixed region of an electron-transport compound and a reducing dopant, or a mixture of a hole-transport compound and an oxidizing dopant is provided on at least one surface of the prepared electrode pair. mixed zone. In this case, electron injection and transport from the mixed region to the electroluminescent medium is facilitated since the electron transport compound is reduced to an anion. In addition, since the hole transport compound is oxidized to form a cation, injection and transport of holes from the mixed region to the electroluminescent medium is facilitated. Preferred oxidative dopants include various Lewis acids and acceptor compounds. Preferred reductive dopants include alkali metals, alkali metal compounds, alkaline earth metals, rare earth metals, and mixtures thereof. In addition, white-light emitting electroluminescent devices having two or more electroluminescent layers can be produced by using a reducing dopant layer as a charge generation layer.
本发明的有益的效果Beneficial effect of the present invention
根据本发明,有机电致发光化合物可具有高发光效率并且可具有优异的材料寿命,可用于制备具有极优越工作寿命的OLED器件。According to the present invention, the organic electroluminescent compound can have high luminous efficiency and excellent material lifetime, and can be used to prepare OLED devices with extremely superior working lifetime.
本发明的实施方式Embodiments of the present invention
下文以本发明的代表性化合物为例,对本发明的有机电致发光化合物、其制备方法以及该器件的电致发光性质进行进一步描述。但是这些实施例仅用于说明实施方式目的,并不旨在限制本发明的范围。The organic electroluminescent compound of the present invention, its preparation method and the electroluminescent properties of the device are further described below by taking representative compounds of the present invention as examples. However, these examples are for illustrative purposes only and are not intended to limit the scope of the present invention.
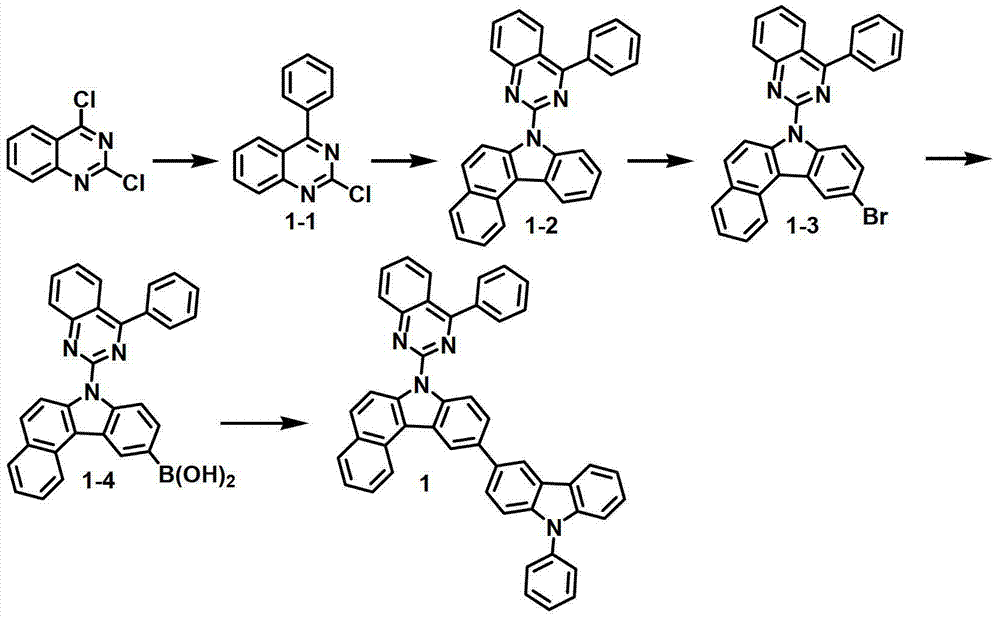
[制备例1]制备化合物1[Preparation Example 1] Preparation of Compound 1
化合物1-1的制备Preparation of compound 1-1
将2,4-二氯喹唑啉(8.1克,40.6毫摩尔)、苯基硼酸(5.0克,40.6毫摩尔)、甲苯200毫升、50毫升乙醇和50毫升水进行混合,再加入Pd(PPh3)4(1.9克,1.64毫摩尔)和K2CO3(12.9克,122毫摩尔)。将该混合物在120℃下搅拌5小时,并冷却至室温,然后用200毫升氯化铵水溶液终止该反应。用500毫升乙酸乙酯(EA)对该混合物进行萃取,再用50毫升蒸馏水对其进行洗涤。用无水MgSO4对获得的有机层进行干燥,在减压条件下去除有机溶剂。随后,进行硅胶过滤并重结晶,得到化合物1-1(6.6克,68%)。2,4-dichloroquinazoline (8.1 g, 40.6 mmol), phenylboronic acid (5.0 g, 40.6 mmol), 200 ml of toluene, 50 ml of ethanol and 50 ml of water were mixed, and then Pd (PPh 3 ) 4 (1.9 g, 1.64 mmol) and K 2 CO 3 (12.9 g, 122 mmol). The mixture was stirred at 120°C for 5 hours and cooled to room temperature, then quenched with 200 ml of aqueous ammonium chloride. The mixture was extracted with 500 mL of ethyl acetate (EA), and washed with 50 mL of distilled water. The obtained organic layer was dried with anhydrous MgSO 4 , and the organic solvent was removed under reduced pressure. Subsequently, silica gel filtration and recrystallization were performed to obtain Compound 1-1 (6.6 g, 68%).
化合物1-2的制备Preparation of compound 1-2
在回流下对7H-苯并[c]咔唑(8.9克,41.10毫摩尔)、化合物1-1(11.9克,49.32毫摩尔)、Pd(OAc)2(0.46克、NaOt-bu7.9克,82.20毫摩尔)、100毫升甲苯、P(t-bu)3(2毫升,4.11毫摩尔,50%的甲苯溶液)进行搅拌。10小时后,将该混合物冷却至室温,且向其中加入蒸馏水。用EA对所得到的混合物进行萃取,用无水MgSO4干燥,再在减压条件下进行干燥。随后,进行柱分离,得到化合物1-2(14.5克,84%)。Under reflux, 7H-benzo[c]carbazole (8.9 g, 41.10 mmol), compound 1-1 (11.9 g, 49.32 mmol), Pd(OAc) 2 (0.46 g, NaOt-bu7.9 g , 82.20 mmol), 100 ml of toluene, P(t-bu) 3 (2 ml, 4.11 mmol, 50% solution in toluene) were stirred. After 10 hours, the mixture was cooled to room temperature, and distilled water was added thereto. The resulting mixture was extracted with EA, dried over anhydrous MgSO4 , and dried under reduced pressure. Subsequently, column separation was performed to obtain compound 1-2 (14.5 g, 84%).
化合物1-3的制备Preparation of Compound 1-3
将化合物1-2(14.3克,33.98毫摩尔)置入单颈烧瓶中,形成真空气氛,再用氩气对该烧瓶进行填充。加入THF(500毫升),在0℃下将该混合物搅拌10分钟。加入NBS(7.35克,40.78毫摩尔),在室温下将该混合物搅拌一天。终止反应,用蒸馏水和EA对所得混合物进行萃取。用无水MgSO4对有机层进行干燥,使用旋转蒸发仪除去溶剂,再使用己烷和EA作为显影剂进行柱色谱,得到化合物1-3(14.6克,85%)。Put compound 1-2 (14.3 g, 33.98 mmol) into a single-necked flask to form a vacuum atmosphere, and then fill the flask with argon. THF (500 mL) was added, and the mixture was stirred at 0°C for 10 minutes. NBS (7.35 g, 40.78 mmol) was added and the mixture was stirred at room temperature for one day. The reaction was terminated, and the resulting mixture was extracted with distilled water and EA. The organic layer was dried with anhydrous MgSO 4 , the solvent was removed using a rotary evaporator, and column chromatography was performed using hexane and EA as developers to obtain compound 1-3 (14.6 g, 85%).
化合物1-4的制备Preparation of Compound 1-4
将化合物1-3(13.2克,26.30毫摩尔)置入单颈烧瓶中,形成真空气氛,再用氩气对该烧瓶进行填充。加入500毫升THF,在-78℃下将该混合物搅拌10分钟。逐滴加入正-BuLi(2.5M的己烷溶液)(15.8毫升,39.45毫摩尔),在-78℃下将该混合物搅拌1小时30分钟。在-78℃下加入硼酸三甲酯(4.85毫升,39.45毫摩尔)。在-78℃下搅拌该混合物30分钟,然后在室温下搅拌4小时。终止反应,用蒸馏水和EA对所得混合物进行萃取。用无水MgSO4对有机层进行干燥,使用旋转蒸发仪除去溶剂,再使用己烷和EA作为显影剂进行柱色谱,得到化合物1-4(6.9克,18.05毫摩尔,65%)。Put compound 1-3 (13.2 g, 26.30 mmol) into a single-necked flask to form a vacuum atmosphere, and then fill the flask with argon. 500 mL of THF was added, and the mixture was stirred at -78°C for 10 minutes. n-BuLi (2.5M in hexane) (15.8 mL, 39.45 mmol) was added dropwise, and the mixture was stirred at -78°C for 1 hour 30 minutes. Trimethyl borate (4.85 mL, 39.45 mmol) was added at -78°C. The mixture was stirred at -78°C for 30 minutes, then at room temperature for 4 hours. The reaction was terminated, and the resulting mixture was extracted with distilled water and EA. The organic layer was dried with anhydrous MgSO 4 , the solvent was removed using a rotary evaporator, and column chromatography was performed using hexane and EA as developers to obtain compound 1-4 (6.9 g, 18.05 mmol, 65%).
化合物1的制备Preparation of compound 1
在回流下将化合物1-4(8.1克,17.4毫摩尔)、3-溴-9-苯基-9H-咔唑(6.7克,20.88毫摩尔)、Pd(PPh3)4(0.8克,0.7毫摩尔)、20毫升2M K2CO3水溶液、100毫升甲苯和50毫升乙醇搅拌12小时。用蒸馏水对该混合物进行洗涤,并用EA进行萃取。用无水MgSO4干燥,在减压下进行蒸馏,并进行柱分离,得到化合物1(6.8克,10.3毫摩尔,58%)。Compound 1-4 (8.1 g, 17.4 mmol), 3-bromo-9-phenyl-9H-carbazole (6.7 g, 20.88 mmol), Pd(PPh 3 ) 4 (0.8 g, 0.7 mmol), 20 mL of 2M aqueous K2CO3 , 100 mL of toluene and 50 mL of ethanol were stirred for 12 hours. The mixture was washed with distilled water and extracted with EA. Drying with anhydrous MgSO 4 , distillation under reduced pressure, and column separation gave compound 1 (6.8 g, 10.3 mmol, 58%).
MS/FAB:663(实测),662.78(计算)MS/FAB: 663 (measured), 662.78 (calculated)
[制备例2]制备化合物2[Preparation Example 2] Preparation of Compound 2
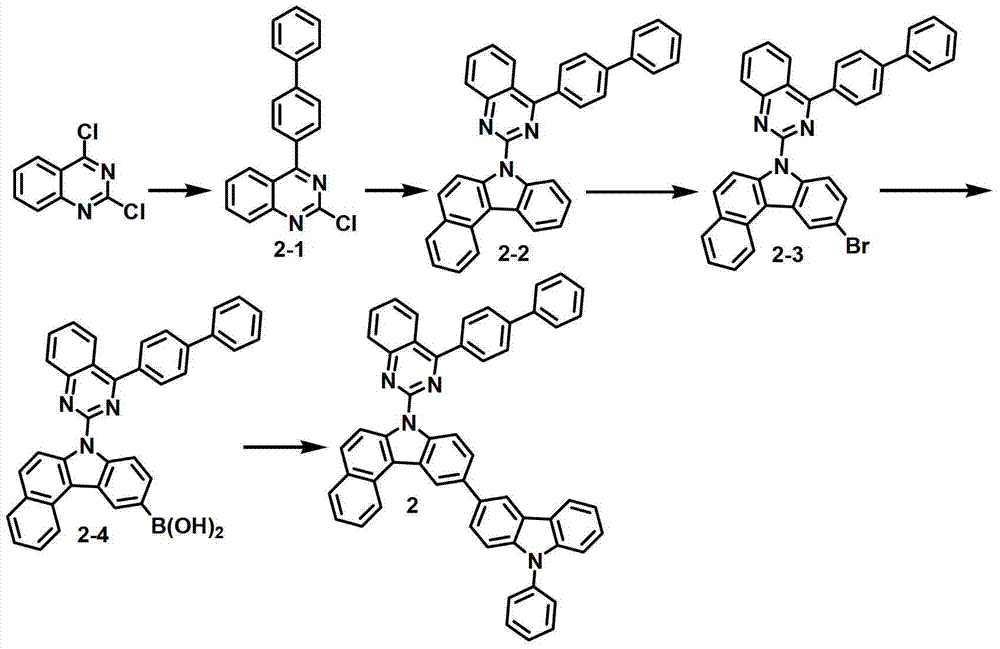
化合物2-1的制备Preparation of compound 2-1
将2,4-二氯喹唑啉(8.1克,40.6毫摩尔)、联苯基苯基硼酸(8.0克,40.6毫摩尔)、200毫升甲苯、50毫升乙醇和50毫升水进行混合,再加入Pd(PPh3)4(1.9克,1.64毫摩尔)和K2CO3(12.9克,122毫摩尔)。将该混合物在120℃下搅拌5小时,并冷却至室温,用200毫升氯化铵水溶液终止反应。用500毫升EA对该混合物进行萃取,用50毫升蒸馏水对其进行洗涤。用无水MgSO4对获得的有机层进行干燥,在减压条件下去除有机溶剂。随后,进行硅胶过滤并重结晶,得到化合物2-1(8.2克,25.9毫摩尔,64%)。Mix 2,4-dichloroquinazoline (8.1 g, 40.6 mmol), biphenylphenylboronic acid (8.0 g, 40.6 mmol), 200 mL toluene, 50 mL ethanol and 50 mL water, and add Pd ( PPh3 ) 4 (1.9 g, 1.64 mmol) and K2CO3 (12.9 g, 122 mmol) . The mixture was stirred at 120°C for 5 hours, cooled to room temperature, and quenched with 200 ml of aqueous ammonium chloride. The mixture was extracted with 500 mL of EA and washed with 50 mL of distilled water. The obtained organic layer was dried with anhydrous MgSO 4 , and the organic solvent was removed under reduced pressure. Subsequently, silica gel filtration and recrystallization were performed to obtain compound 2-1 (8.2 g, 25.9 mmol, 64%).
化合物2-2的制备Preparation of compound 2-2
通过与制备化合物1-2相同的方法制备化合物2-2(9.5克,19.1毫摩尔,74%)。Compound 2-2 (9.5 g, 19.1 mmol, 74%) was prepared by the same method as that of compound 1-2.
化合物2-3的制备Preparation of compound 2-3
通过与制备化合物1-3相同的方法制备化合物2-3(9.0克,15.6毫摩尔,82%)。Compound 2-3 (9.0 g, 15.6 mmol, 82%) was prepared by the same method as that of compound 1-3.
化合物2-4的制备Preparation of Compound 2-4
通过与制备化合物1-4相同的方法制备化合物2-4(4.0克,7.4毫摩尔,47%)。Compound 2-4 (4.0 g, 7.4 mmol, 47%) was prepared by the same method as that of compound 1-4.
化合物2的制备Preparation of compound 2
通过与制备化合物1相同的方法由化合物2-4(4.0克,7.4毫摩尔)和3-溴-9-苯基-9H-咔唑(6.7克,20.88毫摩尔)来制备化合物2(2.8克,4.6毫摩尔,51%)。Compound 2 (2.8 g , 4.6 mmol, 51%).
MS/FAB:739(实测),738.87(计算)MS/FAB: 739 (measured), 738.87 (calculated)
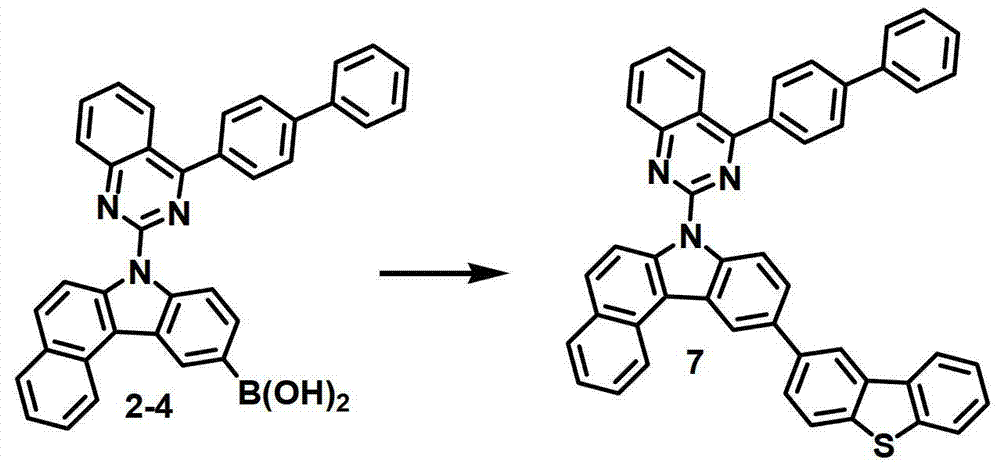
[制备例3]制备化合物7[Preparation Example 3] Preparation of Compound 7
化合物7的制备Preparation of Compound 7
通过与制备化合物1相同的方法由化合物2-4(4.0克,7.4毫摩尔)和2-溴代二苯并[b,d]噻吩(5.5克,20.88毫摩尔)来制备化合物7(2.8克,4.6毫摩尔,51%)。Compound 7 (2.8 g , 4.6 mmol, 51%).
MS/FAB:604(实测),603.73(计算)MS/FAB: 604 (measured), 603.73 (calculated)
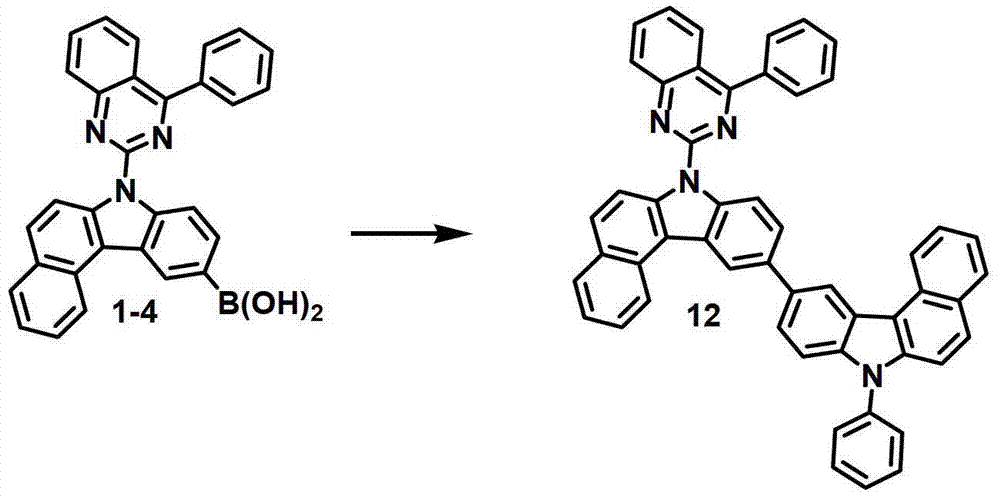
[制备例4]制备化合物12[Preparation Example 4] Preparation of Compound 12
化合物12的制备Preparation of compound 12
通过与制备化合物1相同的方法由化合物1-4(6.9克,18.05毫摩尔)和10-溴-7-苯基-7H-苯并[c]咔唑(7.8克,20.88毫摩尔)来制备化合物12(6.8克,9.5毫摩尔,53%)。Prepared from compound 1-4 (6.9 g, 18.05 mmol) and 10-bromo-7-phenyl-7H-benzo[c]carbazole (7.8 g, 20.88 mmol) by the same method as compound 1 Compound 12 (6.8 g, 9.5 mmol, 53%).
MS/FAB:713(实测),712.84(计算)。MS/FAB: 713 (found), 712.84 (calculated).
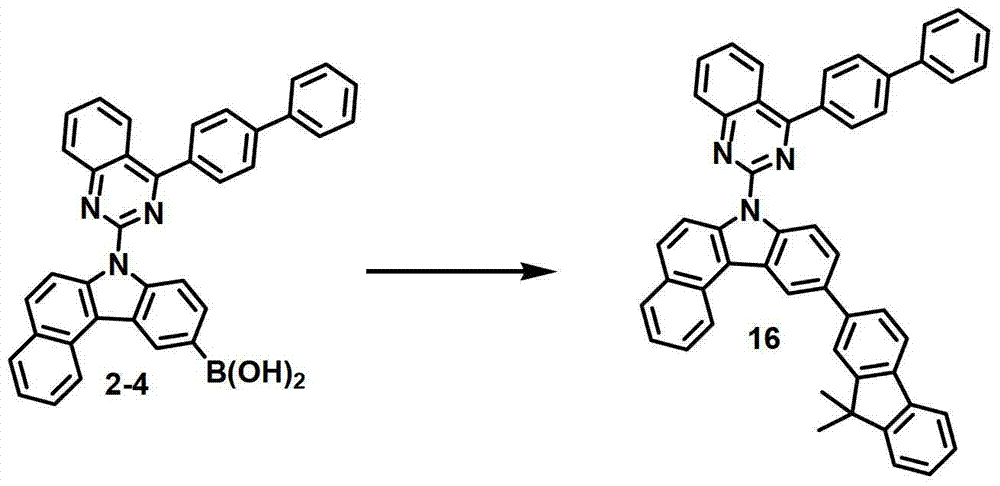
[制备例5]制备化合物16[Preparation Example 5] Preparation of Compound 16
化合物16的制备Preparation of compound 16
通过与制备化合物1相同的方法由化合物2-4(9.8克,18.05毫摩尔)和2-溴-9,9-二甲基-9H-芴(5.7克,20.88毫摩尔)来制备化合物16(7.6克,11.0毫摩尔,61%)。Compound 16 ( 7.6 g, 11.0 mmol, 61%).
MS/FAB:690(实测),689.84(计算)MS/FAB: 690 (measured), 689.84 (calculated)
[制备例6]制备化合物25[Preparation Example 6] Preparation of Compound 25
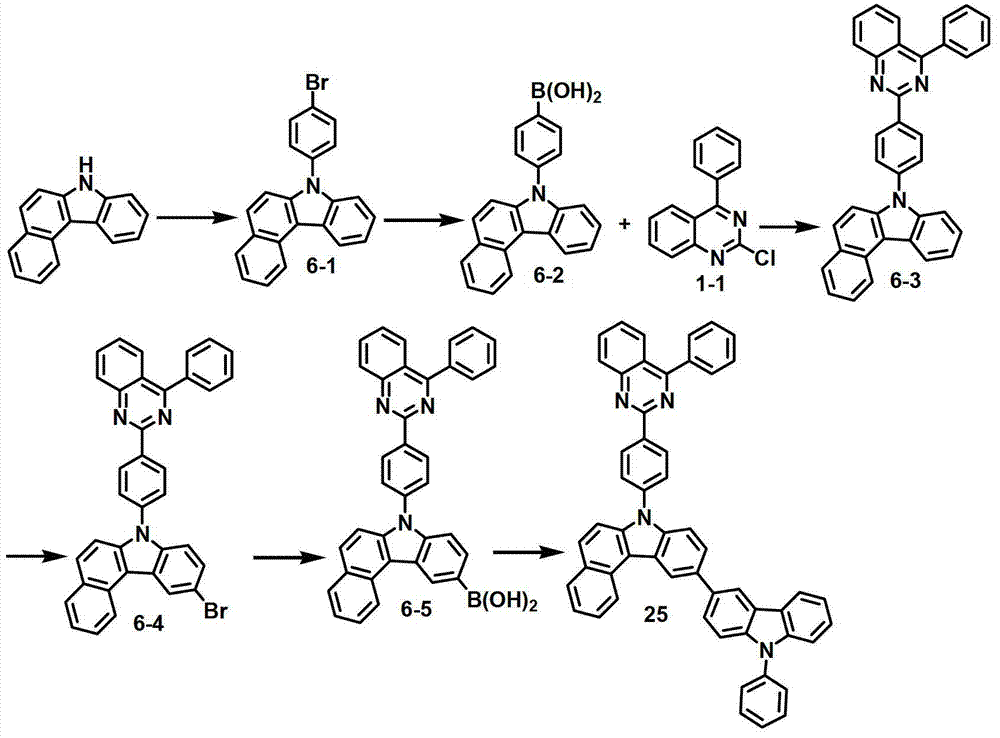
化合物6-1的制备Preparation of compound 6-1
将7H-苯并[c]咔唑(20克,92毫摩尔)和1-溴-4-碘苯(43.5克,184毫摩尔)溶解于500毫升甲苯中,并添加CuI(8.8克,46毫摩尔)、二氨基乙烷(6.2毫升,92毫摩尔)和K3PO4(8.7克,276毫摩尔),再将该混合物回流30小时。将该混合物冷却至室温,并用50毫升2.0M HCl水溶液终止该反应。用1升EA对所得混合物进行萃取,再用50毫升蒸馏水对其进行洗涤。用无水MgSO4对获得的有机层进行干燥,在减压条件下去除有机溶剂。再进行硅胶柱色谱,得到化合物6-1(19克,56%)。7H-Benzo[c]carbazole (20 g, 92 mmol) and 1-bromo-4-iodobenzene (43.5 g, 184 mmol) were dissolved in 500 mL of toluene, and CuI (8.8 g, 46 mmol), diaminoethane (6.2 mL, 92 mmol) and K 3 PO 4 (8.7 g, 276 mmol), and the mixture was refluxed for 30 hours. The mixture was cooled to room temperature and quenched with 50 mL of 2.0M aqueous HCl. The resulting mixture was extracted with 1 L of EA and washed with 50 mL of distilled water. The obtained organic layer was dried with anhydrous MgSO 4 , and the organic solvent was removed under reduced pressure. Silica gel column chromatography was then carried out to obtain compound 6-1 (19 g, 56%).
化合物6-2的制备Preparation of compound 6-2
将化合物6-1(19克,51毫摩尔)溶解于250毫升THF中,再冷却至-78℃,并在-78℃下加入正-BuLi(2.5M的己烷溶液)(24.5毫升)。将该混合物在-78℃下搅拌1小时,加入B(OMe)3(8.5毫升),然后将该混合物搅拌2小时,用100毫升氯化铵水溶液终止该反应。用500毫升EA对所得混合物进行萃取,再用100毫升蒸馏水对其进行洗涤。用无水MgSO4对获得的有机层进行干燥,在减压条件下去除有机溶剂。然后进行重结晶,得到化合物6-2(14克,81%)。Compound 6-1 (19 g, 51 mmol) was dissolved in 250 mL of THF, cooled to -78°C, and n-BuLi (2.5M in hexane) (24.5 mL) was added at -78°C. The mixture was stirred at -78°C for 1 hour, B(OMe) 3 (8.5 mL) was added, then the mixture was stirred for 2 hours, and the reaction was quenched with 100 mL of aqueous ammonium chloride. The resulting mixture was extracted with 500 ml of EA and washed with 100 ml of distilled water. The obtained organic layer was dried with anhydrous MgSO 4 , and the organic solvent was removed under reduced pressure. Recrystallization was then performed to obtain compound 6-2 (14 g, 81%).
化合物6-3的制备Preparation of compound 6-3
将化合物1-1(3.3克,13.7毫摩尔)和化合物6-2(5.5克,16.4毫摩尔)与100毫升甲苯、20毫升乙醇和20毫升水进行混合,加入Pd(PPh3)4(1.6克,1.4毫摩尔)和K2CO3(5.7克,41.1毫摩尔)。将该混合物在120℃下搅拌5小时,并冷却至室温,用20毫升氯化铵水溶液终止反应。用250毫升EA对该混合物进行萃取,用30毫升蒸馏水对其进行洗涤。用无水MgSO4对获得的有机层进行干燥,在减压条件下去除有机溶剂。然后进行硅胶过滤和重结晶,得到化合物6-3(4.9克,9.8毫摩尔,72%)。Compound 1-1 (3.3 g, 13.7 mmol) and compound 6-2 (5.5 g, 16.4 mmol) were mixed with 100 ml of toluene, 20 ml of ethanol and 20 ml of water, and Pd(PPh 3 ) 4 (1.6 g, 1.4 mmol) and K 2 CO 3 (5.7 g, 41.1 mmol). The mixture was stirred at 120°C for 5 hours, cooled to room temperature, and quenched with 20 ml of aqueous ammonium chloride. The mixture was extracted with 250 mL of EA and washed with 30 mL of distilled water. The obtained organic layer was dried with anhydrous MgSO 4 , and the organic solvent was removed under reduced pressure. Silica gel filtration and recrystallization were then performed to obtain compound 6-3 (4.9 g, 9.8 mmol, 72%).
化合物6-4的制备Preparation of Compound 6-4
通过与制备化合物1-3相同的方法制备化合物6-4(4.3克,7.5毫摩尔,76%)。Compound 6-4 (4.3 g, 7.5 mmol, 76%) was prepared by the same method as that of compound 1-3.
化合物6-5的制备Preparation of compound 6-5
通过与制备化合物1-4相同的方法制备化合物6-5(1.7克,3.1毫摩尔,43%)。Compound 6-5 (1.7 g, 3.1 mmol, 43%) was prepared by the same method as that of compound 1-4.
化合物25的制备Preparation of compound 25
通过与制备化合物1相同的方法由化合物6-5(9.8克,18.05毫摩尔)和3-溴-9-苯基-9H-咔唑(6.7克,20.88毫摩尔)制备化合物25(7.6克,10.3毫摩尔,57%)。Compound 25 (7.6 g, 10.3 mmol, 57%).
MS/FAB:739(实测),738.87(计算)MS/FAB: 739 (measured), 738.87 (calculated)
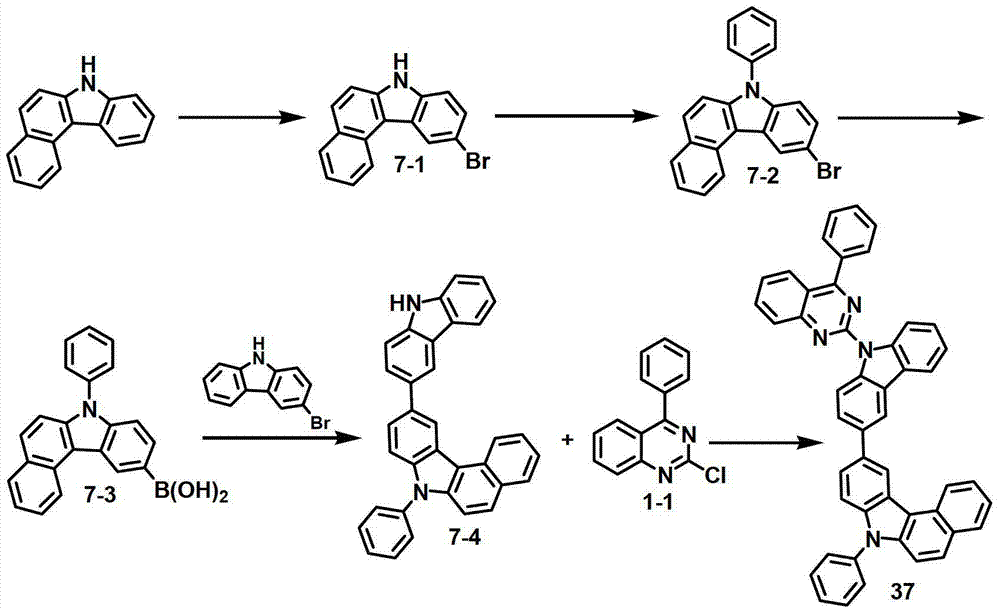
[制备例7]制备化合物37[Preparation Example 7] Preparation of Compound 37
化合物7-1的制备Preparation of compound 7-1
将7H-苯并[c]咔唑(50克,0.23摩尔)溶解于1.4升DMF中,加入NBS(41克,0.23摩尔),然后在室温条件下将该混合物搅拌24小时。反应终止后,用EA对所得混合物进行萃取,将有机层在减压条件下进行蒸馏。然后进行硅胶柱分离,得到化合物7-1(53.2克,78%)。7H-Benzo[c]carbazole (50 g, 0.23 mol) was dissolved in 1.4 L of DMF, NBS (41 g, 0.23 mol) was added, and the mixture was stirred at room temperature for 24 hours. After the reaction was terminated, the resulting mixture was extracted with EA, and the organic layer was distilled under reduced pressure. Then silica gel column separation was carried out to obtain compound 7-1 (53.2 g, 78%).
化合物7-2的制备Preparation of compound 7-2
将化合物7-1(30克,0.10摩尔)、碘苯(22.6毫升,0.20毫摩尔)、CuI(9.6克,0.05摩尔)、Cs2CO3(99克,0.030摩尔)和EDA(13.7毫升,0.20摩尔)加入至500毫升甲苯中,并在回流下搅拌24小时。用EA对该混合物进行萃取,在减压条件下进行蒸馏,并用MC/Hex进行柱分离,得到化合物7-2(20克,53%)。Compound 7-1 (30 g, 0.10 mol), iodobenzene (22.6 ml, 0.20 mmol), CuI (9.6 g, 0.05 mol), Cs 2 CO 3 (99 g, 0.030 mol) and EDA (13.7 ml, 0.20 mol) was added to 500 ml of toluene, and stirred under reflux for 24 hours. The mixture was extracted with EA, distilled under reduced pressure, and column separated with MC/Hex to obtain compound 7-2 (20 g, 53%).
化合物7-3的制备Preparation of compound 7-3
将化合物7-2(18克,48.4毫摩尔)溶解在250毫升THF中,在-78℃下加入2.5M正-BuLi(在己烷中)(23.2毫升,58.0毫摩尔),然后将该混合物搅拌1小时。缓慢添加B(Oi-Pr)3(16.7毫升,72.5毫摩尔),将该溶液搅拌2小时。添加2M HCl,并用蒸馏水和EA对该混合物进行猝灭和提取。再使用MC和Hex进行重结晶,得到化合物7-3(13.6克,83.4%)。Compound 7-2 (18 g, 48.4 mmol) was dissolved in 250 mL of THF, 2.5M n-BuLi (in hexane) (23.2 mL, 58.0 mmol) was added at -78°C, and the mixture Stir for 1 hour. B(Oi-Pr) 3 (16.7 mL, 72.5 mmol) was added slowly and the solution was stirred for 2 hours. 2M HCl was added and the mixture was quenched and extracted with distilled water and EA. Then recrystallization was carried out using MC and Hex to obtain compound 7-3 (13.6 g, 83.4%).
化合物7-4的制备Preparation of compound 7-4
将化合物7-3(13.6克,40.3毫摩尔)、溴代咔唑(9.9克,40.3毫摩尔)、Pd(PPh3)4(2.3克,2.0毫摩尔)、K2CO3(13.4克,96.7毫摩尔)、200毫升甲苯和48毫升蒸馏水进行混合,再在90℃下搅拌2小时。在减压条件下对有机层进行蒸馏,并用MeOH进行研磨。将获得的固体溶解于MC中,二氧化硅过滤,并用MC和己烷进行研磨,得到化合物7-4(15克,81%)。Compound 7-3 (13.6 g, 40.3 mmol), bromocarbazole (9.9 g, 40.3 mmol), Pd(PPh 3 ) 4 (2.3 g, 2.0 mmol), K 2 CO 3 (13.4 g, 96.7 mmol), 200 ml of toluene and 48 ml of distilled water were mixed, and stirred at 90° C. for 2 hours. The organic layer was distilled under reduced pressure and triturated with MeOH. The obtained solid was dissolved in MC, filtered on silica, and triturated with MC and hexane to obtain compound 7-4 (15 g, 81%).
化合物37的制备Preparation of compound 37
将化合物1-1(4克,16.6毫摩尔)和化合物7-4(7.6克,16.6毫摩尔)悬浮于80毫升DMF中,在室温条件下添加60%NaH(1.1克,28.2毫摩尔),将该混合物搅拌12小时。添加1升蒸馏水,并在减压条件下对该混合物进行过滤。用MeOH/EA对所得固体进行研磨,溶解于MC中,进行二氧化硅过滤,再用MC/正己烷进行研磨,得到化合物37(2.4克,21.8%)。Compound 1-1 (4 g, 16.6 mmol) and compound 7-4 (7.6 g, 16.6 mmol) were suspended in 80 ml of DMF, and 60% NaH (1.1 g, 28.2 mmol) was added at room temperature, The mixture was stirred for 12 hours. 1 liter of distilled water was added, and the mixture was filtered under reduced pressure. The resulting solid was triturated with MeOH/EA, dissolved in MC, filtered on silica, and triturated with MC/n-hexane to give compound 37 (2.4 g, 21.8%).
MS/FAB的实测值662.78,理论值662.25The measured value of MS/FAB is 662.78, and the theoretical value is 662.25
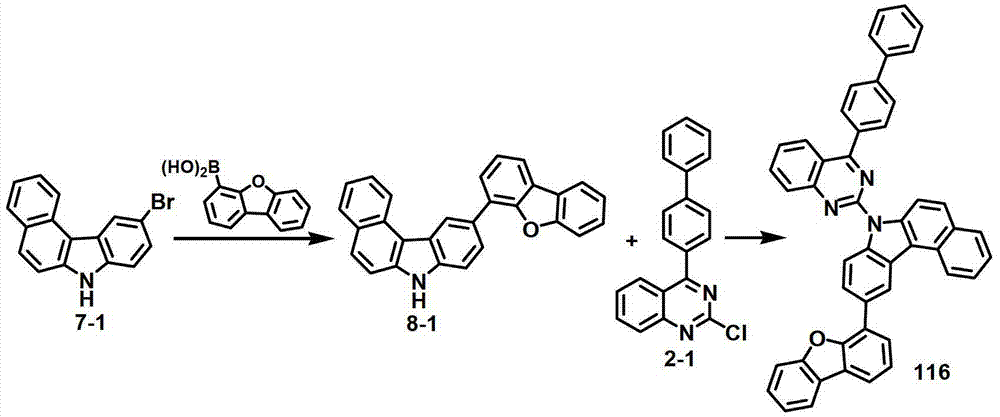
[制备例8]制备化合物116[Preparation Example 8] Preparation of Compound 116
化合物8-1的制备Preparation of Compound 8-1
将化合物7-1(10克,33.8毫摩尔)、二苯并[b,d]呋喃-4-基硼酸(8.6克,40.56毫摩尔)、Pd(PPh3)4(2克,1.7毫摩尔)、K2CO3(34克,321毫摩尔)、60毫升甲苯、12毫升EtOH和12毫升纯化水进行混合,再在120℃下搅拌15小时。反应终止后,将所得混合物静置,除去水层,然后将有机层浓缩。然后进行硅胶柱纯化,得到化合物8-1(10.2克,78%)。Compound 7-1 (10 g, 33.8 mmol), dibenzo[b,d]furan-4-ylboronic acid (8.6 g, 40.56 mmol), Pd(PPh 3 ) 4 (2 g, 1.7 mmol ), K 2 CO 3 (34 g, 321 mmol), 60 ml of toluene, 12 ml of EtOH and 12 ml of purified water were mixed and stirred at 120° C. for 15 hours. After the reaction was terminated, the resulting mixture was left standing, the aqueous layer was removed, and then the organic layer was concentrated. Silica gel column purification was then performed to obtain Compound 8-1 (10.2 g, 78%).
化合物116的制备Preparation of compound 116
将化合物8-1(3克,7.8毫摩尔)和化合物2-1(2.1克,7.8毫摩尔)悬浮于30毫升DMF中,在室温条件下添加60%NaH(376毫克,9.4毫摩尔),将该混合物搅拌12小时。添加500毫升纯化水,并在减压条件下对该混合物进行过滤。依次用MeOH/EA、DMF和EA/THF对获得的固体进行研磨。将获得的产物溶解于MC中,二氧化硅过滤,并用MeOH/EA进行研磨,得到化合物116(2.4克,46%)。Compound 8-1 (3 g, 7.8 mmol) and compound 2-1 (2.1 g, 7.8 mmol) were suspended in 30 ml of DMF, and 60% NaH (376 mg, 9.4 mmol) was added at room temperature, The mixture was stirred for 12 hours. 500 ml of purified water was added, and the mixture was filtered under reduced pressure. The obtained solid was triturated sequentially with MeOH/EA, DMF and EA/THF. The obtained product was dissolved in MC, filtered on silica and triturated with MeOH/EA to give compound 116 (2.4 g, 46%).
MS/FAB的实测值663.76,理论值663.23The measured value of MS/FAB is 663.76, and the theoretical value is 663.23
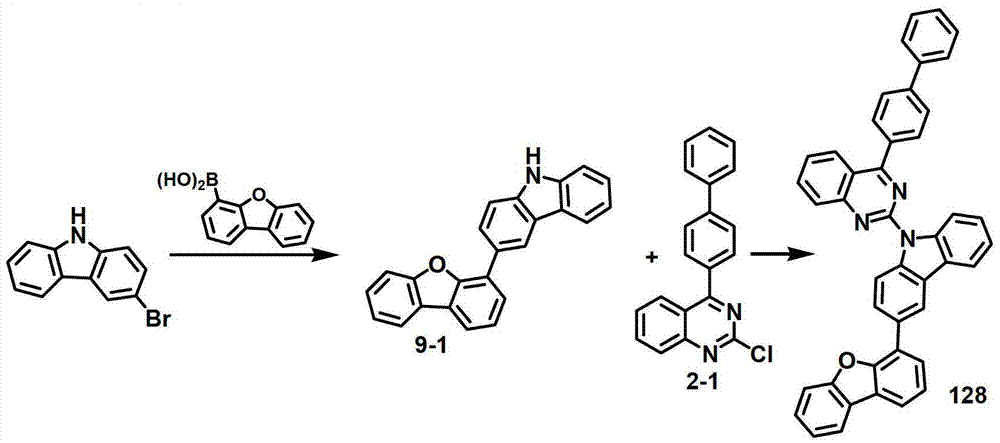
[制备例9]制备化合物128[Preparation Example 9] Preparation of Compound 128
化合物9-1的制备Preparation of compound 9-1
将3-溴-9H-咔唑(5克,20.32毫摩尔)、二苯并[b,d]呋喃-4-基硼酸(4.7克,22.35毫摩尔)、K2CO3(7克,50.79毫摩尔)、Pd(PPh3)4(1.17克,1.01毫摩尔)、100毫升甲苯、25毫升EtOH和25毫升纯化水进行混合,再在100℃下搅拌3小时。反应终止后,将该混合物冷却至室温,将其静置,除去水层。将有机层浓缩,再使用硅胶柱进行纯化,得到化合物9-1(4.3克,64%)。3-Bromo-9H-carbazole (5 g, 20.32 mmol), dibenzo[b,d]furan-4-ylboronic acid (4.7 g, 22.35 mmol), K 2 CO 3 (7 g, 50.79 mmol), Pd(PPh 3 ) 4 (1.17 g, 1.01 mmol), 100 mL of toluene, 25 mL of EtOH and 25 mL of purified water were mixed and stirred at 100°C for 3 hours. After the reaction was terminated, the mixture was cooled to room temperature, allowed to stand, and the water layer was removed. The organic layer was concentrated and purified using a silica gel column to obtain compound 9-1 (4.3 g, 64%).
化合物128的制备Preparation of Compound 128
将化合物2-1(3克,8.99毫摩尔)和化合物9-1(3.14克,9.89毫摩尔)悬浮于50毫升无水DMF中,在室温下添加60%NaH(0.54克,13.5毫摩尔)。室温下搅拌该混合物5小时。反应终止后,逐滴加入3毫升MeOH,由于过量的MeOH而获得固体,进行硅胶柱纯化、用EA进行悬浮,过滤,再用THF进行结晶,得到化合物128(1克,18%)。Compound 2-1 (3 g, 8.99 mmol) and compound 9-1 (3.14 g, 9.89 mmol) were suspended in 50 mL of anhydrous DMF, and 60% NaH (0.54 g, 13.5 mmol) was added at room temperature . The mixture was stirred at room temperature for 5 hours. After the reaction was terminated, 3 ml of MeOH was added dropwise, and a solid was obtained due to excess MeOH, which was purified on a silica gel column, suspended with EA, filtered, and crystallized with THF to obtain compound 128 (1 g, 18%).
MS/FAB的实测值613.70,理论值613.22The measured value of MS/FAB is 613.70, and the theoretical value is 613.22
[实施例1]使用本发明的有机电致发光化合物制备OLED器件[Example 1] Preparation of OLED devices using the organic electroluminescent compound of the present invention
使用本发明的有机电子材料的化合物制造OLED器件。首先,将由玻璃制成的用于OLED的透明电极ITO薄膜(15Ω/□)(购自三星康宁公司(Samsung-Corning))依次用三氯乙烯、丙酮、乙醇和蒸馏水进行超声清洗,在使用之前保存于异丙醇中。然后,将ITO基片装在真空沉积设备的基片夹(folder)中,将N1,N1’-([1,1’-联苯基]-4,4’-二基)二(N1-(萘-1-基)-N4,N4-二苯基苯-1,4-二胺置于真空沉积设备的小室(cell)中,然后排气至室内真空度最高达到10-6托。接着,向所述小室施加电流使其蒸发,从而在ITO基片上沉积厚度为60纳米的空穴注入层。随后,将N,N’-二(4-联苯基)-N,N’-二(4-联苯基)-4,4’-二氨基联苯基置于真空沉积设备的另一个小室中,通过向该小室施加电流以使NPB蒸发,从而在所述空穴注入层上沉积厚度为20纳米的空穴传输层。在形成空穴注入层和空穴传输层后,在其上面形成电致发光层,具体如下。在真空气相沉积设备内,将作为基质的化合物1置于一个小室中,将作为掺杂剂的化合物D-11置于另一个小室中。以不同的速率使这两种材料蒸发,使得发生4重量%的掺杂,从而厚度为30纳米的电致发光层气相沉积在空穴传输层上。随后,将2-(4-(9,10-二(萘-2-基)蒽-2-基)苯基)-1-苯基-1H-苯并[d]咪唑置于一个小室中,将喹啉合锂另一个小室内,然后将这两种材料以相同的速率进行蒸发,使得发生50重量%掺杂,从而在所述电致发光层上气相沉积厚度为30纳米的电子传输层。接着,气相沉积厚度为2纳米的Liq(喹啉合锂)作为电子注入层,然后使用另一真空气相沉积设备来气相沉积厚度为150纳米的Al阴极,从而制得OLED。OLED devices are fabricated using the inventive compounds of organic electronic materials. First, the transparent electrode ITO film (15Ω/□) for OLED made of glass (purchased from Samsung-Corning) was ultrasonically cleaned with trichlorethylene, acetone, ethanol and distilled water in sequence, before use Store in isopropanol. Then, the ITO substrate was placed in the substrate folder of the vacuum deposition equipment, and N 1 , N 1' -([1,1'-biphenyl]-4,4'-diyl)di( N 1 -(naphthalene-1-yl)-N 4 , N 4 -diphenylbenzene-1,4-diamine is placed in the small chamber (cell) of the vacuum deposition equipment, and then exhausted to a maximum vacuum of 10 -6 torr.Then, apply electric current to described small chamber and make it evaporate, thereby on the ITO substrate, deposit the hole injection layer that thickness is 60 nanometers.Subsequently, N, N '-bis (4-biphenyl)-N , N'-bis(4-biphenyl)-4,4'-diaminobiphenyl is placed in another small chamber of the vacuum deposition equipment, and the NPB is evaporated by applying an electric current to the small chamber, so that in the air Deposition thickness is the hole transport layer of 20 nanometers on the hole injection layer.After forming hole injection layer and hole transport layer, form electroluminescent layer on it, specifically as follows.In vacuum vapor deposition equipment, will serve as substrate The compound 1 of is placed in a small chamber, and the compound D-11 as dopant is placed in another small chamber.These two kinds of materials are evaporated with different rates, so that 4% by weight of doping occurs, thus the thickness is 30 A nanometer electroluminescent layer is vapor deposited on the hole transport layer. Subsequently, 2-(4-(9,10-bis(naphthalene-2-yl)anthracen-2-yl)phenyl)-1-phenyl -1H-benzo[d]imidazole is placed in one chamber, quinolate is lithium-treated in another chamber, and then the two materials are evaporated at the same rate so that 50% by weight doping occurs, thereby in the On the electroluminescence layer, vapor deposition thickness is an electron transport layer of 30 nanometers. Then, vapor deposition thickness is 2 nanometers of Liq (quinoline alloy lithium) as electron injection layer, and then use another vacuum vapor deposition equipment to vapor deposition thickness of 150nm Al cathode to make an OLED.
在使用前,用于OLED器件的各化合物通过在10-6托下真空升华得以纯化。Each compound used in OLED devices was purified by vacuum sublimation at 10 −6 Torr before use.
结果为,证实在4.4V的电压下电流流动为8.8毫安/厘米2,并且发射900cd/m2的红光。As a result, it was confirmed that the current flow was 8.8 mA/cm 2 at a voltage of 4.4 V, and red light of 900 cd/m 2 was emitted.
[实施例2]使用本发明的有机电致发光化合物制备OLED器件[Example 2] Preparation of OLED devices using the organic electroluminescent compound of the present invention
使用与实施例1相同的方法来制造OLED器件,不同之处在于,在电致发光层中使用化合物2作为基质材料,并使用D-7作为掺杂剂。An OLED device was fabricated using the same method as in Example 1, except that Compound 2 was used as a host material and D-7 was used as a dopant in the electroluminescent layer.
结果为,证实在4.9V的电压下电流流动为22.9毫安/厘米2,并且发射2710cd/m2的红光。As a result, it was confirmed that a current of 22.9 mA/cm 2 flowed at a voltage of 4.9 V, and red light of 2710 cd/m 2 was emitted.
[实施例3]使用本发明的有机电致发光化合物制备OLED器件[Example 3] Preparation of OLED devices using the organic electroluminescent compound of the present invention
使用与实施例1相同的方法来制造OLED器件,不同之处在于,在电致发光层中使用化合物25作为基质材料。An OLED device was fabricated using the same method as in Example 1, except that Compound 25 was used as a host material in the electroluminescent layer.
结果为,证实在4.8V的电压下电流流动为16.9毫安/厘米2,并且发射1780cd/m2的红光。As a result, it was confirmed that a current of 16.9 mA/cm 2 flowed at a voltage of 4.8 V, and red light of 1780 cd/m 2 was emitted.
[实施例4]使用本发明的有机电致发光化合物制备OLED器件[Example 4] Preparation of OLED devices using the organic electroluminescent compound of the present invention
使用与实施例1相同的方法来制造OLED器件,不同之处在于,在电致发光层中使用化合物37作为基质材料,并使用D-7作为掺杂剂。An OLED device was fabricated using the same method as in Example 1, except that Compound 37 was used as a host material and D-7 was used as a dopant in the electroluminescent layer.
结果为,证实在5.5V的电压下电流流动为41.2毫安/厘米2,并且发射4800cd/m2的红光。As a result, it was confirmed that a current of 41.2 mA/cm 2 flowed at a voltage of 5.5 V, and red light of 4800 cd/m 2 was emitted.
[实施例5]使用本发明的有机电致发光化合物制备OLED器件[Example 5] Preparation of OLED devices using the organic electroluminescent compound of the present invention
使用与实施例1相同的方法来制造OLED器件,不同之处在于,在电致发光层中使用化合物128作为基质材料,并使用D-7作为掺杂剂。An OLED device was fabricated using the same method as in Example 1, except that Compound 128 was used as a host material and D-7 was used as a dopant in the electroluminescent layer.
结果为,证实在4.2V的电压下电流流动为8.7mA/cm2,并且发射900cd/m2的红光。As a result, it was confirmed that the current flow was 8.7 mA/cm 2 at a voltage of 4.2 V, and red light of 900 cd/m 2 was emitted.
[实施例6]使用本发明的有机电致发光化合物制备OLED器件[Example 6] Preparation of OLED devices using the organic electroluminescent compound of the present invention
使用与实施例1相同的方法来制造OLED器件,不同之处在于,在电致发光层中使用化合物139作为基质材料,并使用D-7作为掺杂剂。An OLED device was fabricated using the same method as in Example 1, except that Compound 139 was used as a host material and D-7 was used as a dopant in the electroluminescent layer.
结果为,证实在4.1V的电压下电流流动为3.2mA/cm2,并且发射400cd/m2的红光。As a result, it was confirmed that the current flow was 3.2 mA/cm 2 at a voltage of 4.1 V, and red light of 400 cd/m 2 was emitted.
[比较例1]使用现有技术的电致发光材料的OLED器件的电致发光性质[Comparative Example 1] Electroluminescent properties of OLED devices using prior art electroluminescent materials
通过与实施例1相同的方法来制造OLED器件,不同之处在于,使用4,4’-N,N’-二咔唑-联苯基作为基质以及化合物D-11作为掺杂剂通过气相沉积获得电致发光层,使用二(2-甲基-8-羟基喹啉合(quinolinato))4-苯基苯酚铝(III)在所述电致发光层和电子传输层之间气相沉积获得厚度为10纳米的空穴阻挡层。OLED devices were fabricated by the same method as in Example 1, except that 4,4'-N,N'-dicarbazole-biphenyl was used as a host and compound D-11 was used as a dopant by vapor deposition The electroluminescent layer was obtained, and a thickness of 10 nm hole blocking layer.
结果为,证实在8.2V的电压下电流流动为20.0mA/cm2,并且发射1000cd/m2的红光。As a result, it was confirmed that a current flowed at 20.0 mA/cm 2 at a voltage of 8.2 V, and red light of 1000 cd/m 2 was emitted.
证实相比于常规材料,本发明的有机电致发光化合物展现出优异的电致发光性质。使用本发明的有机电致发光化合物作为基质材料的器件能展现出优异的电致发光性质并能够降低工作电压,从而增加功率效率,并进而改进功率消耗。It was confirmed that the organic electroluminescent compound of the present invention exhibits excellent electroluminescent properties compared to conventional materials. A device using the organic electroluminescent compound of the present invention as a host material can exhibit excellent electroluminescent properties and can reduce operating voltage, thereby increasing power efficiency and further improving power consumption.
虽然出于阐述的目的描述了本发明的优选实施方式,但是本领域技术人员应理解,各种改进、增加和替代方式也是可能的,不背离所附权利要求书所限定的本发明的范围和精神。Although the preferred embodiment of the present invention has been described for illustrative purposes, those skilled in the art will appreciate that various improvements, additions and substitutions are possible without departing from the scope and scope of the present invention as defined by the appended claims. Spirit.
工业应用性Industrial Applicability
根据本发明,有机电致发光化合物可具有高发光效率并且可具有优异的材料寿命,可用于制备具有极优越工作寿命的OLED器件。According to the present invention, the organic electroluminescent compound can have high luminous efficiency and excellent material lifetime, and can be used to prepare OLED devices with extremely superior working lifetime.
Claims (10)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810756909.8A CN109020960B (en) | 2010-09-17 | 2011-09-16 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Applications Claiming Priority (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR10-2010-0091545 | 2010-09-17 | ||
| KR20100091545 | 2010-09-17 | ||
| KR10-2011-0092422 | 2011-09-14 | ||
| KR1020110092422A KR101477614B1 (en) | 2010-09-17 | 2011-09-14 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| PCT/KR2011/006810 WO2012036482A1 (en) | 2010-09-17 | 2011-09-16 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201810756909.8A Division CN109020960B (en) | 2010-09-17 | 2011-09-16 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN103221406A true CN103221406A (en) | 2013-07-24 |
Family
ID=46134276
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2011800555279A Pending CN103221406A (en) | 2010-09-17 | 2011-09-16 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| CN201810756909.8A Active CN109020960B (en) | 2010-09-17 | 2011-09-16 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201810756909.8A Active CN109020960B (en) | 2010-09-17 | 2011-09-16 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2616462A4 (en) |
| JP (3) | JP2013539750A (en) |
| KR (1) | KR101477614B1 (en) |
| CN (2) | CN103221406A (en) |
| TW (1) | TWI560258B (en) |
| WO (1) | WO2012036482A1 (en) |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103502243A (en) * | 2011-03-08 | 2014-01-08 | 罗门哈斯电子材料韩国有限公司 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| CN103534251A (en) * | 2011-03-25 | 2014-01-22 | 罗门哈斯电子材料韩国有限公司 | Novel compounds for organic electronic material and organic electroluminescent device using the same |
| CN103732590A (en) * | 2011-05-30 | 2014-04-16 | 罗门哈斯电子材料韩国有限公司 | New compounds for organic electronic materials and organic electroluminescent devices using the compounds |
| CN104066728A (en) * | 2011-11-18 | 2014-09-24 | 罗门哈斯电子材料韩国有限公司 | Novel organic electroluminescence compounds and organic electroluminescence device comprising the same |
| CN104342124A (en) * | 2014-10-23 | 2015-02-11 | 上海道亦化工科技有限公司 | Benzimidazole based phosphorescent luminescent main body material |
| WO2015089853A1 (en) * | 2013-12-16 | 2015-06-25 | 深圳市华星光电技术有限公司 | Blue fluorescent organic material and organic light emitting diode panel |
| CN105408311A (en) * | 2013-10-03 | 2016-03-16 | 出光兴产株式会社 | Compound, organic electroluminescent element, and electronic device |
| CN105440027A (en) * | 2014-09-18 | 2016-03-30 | 株式会社Lg化学 | Heterocyclic compound and organic light-emitting device comprising same |
| CN105601621A (en) * | 2014-11-18 | 2016-05-25 | 株式会社Lg化学 | Nitrogen-containing polycyclic compound and organic electroluminescence device using same |
| CN105794010A (en) * | 2013-12-18 | 2016-07-20 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compound and multi-component host material and organic electroluminescent device comprising same |
| CN105837392A (en) * | 2016-04-15 | 2016-08-10 | 中节能万润股份有限公司 | Organic electroluminescence material and electroluminescence device including same |
| CN105980520A (en) * | 2014-08-20 | 2016-09-28 | 株式会社Lg化学 | organic light emitting device |
| CN106414407A (en) * | 2014-05-22 | 2017-02-15 | 三星Sdi株式会社 | Organic compound, composition, organic electroluminescent device, and display device |
| CN106543152A (en) * | 2016-10-25 | 2017-03-29 | 上海道亦化工科技有限公司 | Phosphorescence host compound and its organic luminescent device based on naphtho- benzofuran |
| CN106715393A (en) * | 2014-10-14 | 2017-05-24 | 株式会社Lg化学 | Nitrogen-containing polycyclic compound and organic light-emitting device using same |
| CN106749341A (en) * | 2017-01-18 | 2017-05-31 | 江西冠能光电材料有限公司 | Balancing charge injects organic semiconductor and its Organic Light Emitting Diode application |
| WO2018108108A1 (en) * | 2016-12-13 | 2018-06-21 | 广州华睿光电材料有限公司 | Conjugated polymer and use thereof in organic electronic device |
| CN108623480A (en) * | 2017-03-17 | 2018-10-09 | 吉林奥来德光电材料股份有限公司 | New organic luminescent compounds and use its organic electroluminescence device |
| CN109721588A (en) * | 2017-10-27 | 2019-05-07 | 北京鼎材科技有限公司 | Organic electroluminescence device compound and the organic electroluminescence device for using it |
| CN109824671A (en) * | 2017-11-23 | 2019-05-31 | 北京鼎材科技有限公司 | A kind of quinazolinotriazole derivative and its application in the field of organic electroluminescence |
| CN109824672A (en) * | 2017-11-23 | 2019-05-31 | 北京鼎材科技有限公司 | A kind of quinazo triazole derivatives and its application in field of organic electroluminescence |
| CN110143952A (en) * | 2019-06-17 | 2019-08-20 | 上海天马有机发光显示技术有限公司 | A kind of compound, display panel and display device |
| CN113227084A (en) * | 2018-12-26 | 2021-08-06 | Lt素材株式会社 | Compound, composition, organic photoelectric device and display device |
| CN113260615A (en) * | 2018-12-27 | 2021-08-13 | Lt素材株式会社 | Polycyclic compound and organic light-emitting element including same |
| CN114031609A (en) * | 2021-12-14 | 2022-02-11 | 北京燕化集联光电技术有限公司 | Compound containing carbazole and quinazoline structure and application thereof |
Families Citing this family (55)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5623996B2 (en) | 2010-09-21 | 2014-11-12 | 株式会社半導体エネルギー研究所 | Carbazole derivatives |
| JP5856266B2 (en) * | 2010-09-21 | 2016-02-09 | 株式会社半導体エネルギー研究所 | Organic semiconductor device |
| KR20120038060A (en) * | 2010-10-13 | 2012-04-23 | 롬엔드하스전자재료코리아유한회사 | Novel compounds for organic electronic material and organic electroluminescent device using the same |
| KR101720444B1 (en) | 2011-02-07 | 2017-03-27 | 이데미쓰 고산 가부시키가이샤 | Biscarbazole derivative and organic electroluminescent element using same |
| TWI591154B (en) | 2011-02-07 | 2017-07-11 | Idemitsu Kosan Co | Biscarbazole derivatives and organic electroluminescent devices using the same |
| US9287512B2 (en) | 2011-03-08 | 2016-03-15 | Rohm And Haas Electronic Materials Korea Ltd. | Organic electroluminescent compounds, layers and organic electroluminescent device using the same |
| CN103429570A (en) * | 2011-03-24 | 2013-12-04 | 出光兴产株式会社 | Biscarbazole derivative and organic electroluminescent device using same |
| KR20120116269A (en) * | 2011-04-12 | 2012-10-22 | 롬엔드하스전자재료코리아유한회사 | Novel compounds for organic electronic material and organic electroluminescence device using the same |
| WO2012150826A1 (en) * | 2011-05-03 | 2012-11-08 | Rohm And Haas Electronic Materials Korea Ltd. | Novel organic electroluminescent compounds and an organic electroluminescent device using the same |
| KR101396171B1 (en) * | 2011-05-03 | 2014-05-27 | 롬엔드하스전자재료코리아유한회사 | Novel organic electroluminescent compounds and an organic electroluminescent device using the same |
| WO2013012298A1 (en) * | 2011-07-21 | 2013-01-24 | Rohm And Haas Electronic Materials Korea Ltd. | 9h-carbazole compounds and electroluminescent devices involving them |
| KR20130025087A (en) * | 2011-09-01 | 2013-03-11 | 롬엔드하스전자재료코리아유한회사 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| KR102004387B1 (en) * | 2012-04-18 | 2019-07-26 | 에스에프씨 주식회사 | Heterocyclic com pounds and organic light-emitting diode including the same |
| KR102017507B1 (en) * | 2012-04-24 | 2019-10-21 | 에스에프씨 주식회사 | Heterocyclic com pounds and organic light-emitting diode including the same |
| KR20130121479A (en) * | 2012-04-27 | 2013-11-06 | 롬엔드하스전자재료코리아유한회사 | Novel organic electroluminescent compounds and organic electroluminescent device comprising the same |
| JP2014072417A (en) | 2012-09-28 | 2014-04-21 | Idemitsu Kosan Co Ltd | Material for organic electroluminescent element and organic electroluminescent element |
| CN104797571A (en) * | 2012-12-04 | 2015-07-22 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compounds and organic electroluminescent device comprising the same |
| KR101995191B1 (en) * | 2012-12-10 | 2019-07-02 | 코니카 미놀타 가부시키가이샤 | Material for organic electroluminescent element, organic electroluminescent element, illumination device, and display device |
| KR102061571B1 (en) * | 2012-12-24 | 2020-01-02 | 덕산네오룩스 주식회사 | Compound for organic electronic element, organic electronic element using the same, and a electronic device thereof |
| US10157687B2 (en) | 2012-12-28 | 2018-12-18 | Terrapower, Llc | Iron-based composition for fuel element |
| KR102134843B1 (en) | 2013-01-04 | 2020-07-17 | 삼성디스플레이 주식회사 | Carbazole-based compound and organic light emitting diode comprising the same |
| TW201427967A (en) * | 2013-01-08 | 2014-07-16 | Ind Tech Res Inst | Bipolar compound having quinoline and carbazole and organic light emitting diode device using the same |
| KR102081617B1 (en) * | 2013-02-15 | 2020-02-28 | 에스에프씨주식회사 | Deuterated organic compounds for organic light-emitting diode and organic light-emitting diode including the same |
| KR102104633B1 (en) * | 2013-02-15 | 2020-04-27 | 에스에프씨주식회사 | Novel organic compounds for organic light-emitting diode and organic light-emitting diode including the same |
| KR102149450B1 (en) * | 2013-02-15 | 2020-09-01 | 에스에프씨주식회사 | Deuterated organic compounds for organic light-emitting diode and organic light-emitting diode including the same |
| JP6430709B2 (en) * | 2013-03-15 | 2018-11-28 | ダウ グローバル テクノロジーズ エルエルシー | Quinazoline-derived compounds for electronic films and devices |
| KR101658111B1 (en) * | 2013-05-13 | 2016-09-20 | 제일모직 주식회사 | COMPOUND FOR OPTOELECTRIC DEVICE, ORGANIC LiGHT EMITTING DIODE INCLUDING THE SAME AND DISPLAY INCLUDING THE ORGANIC LiGHT EMITTING DIODE |
| US10205099B2 (en) * | 2013-05-31 | 2019-02-12 | Idemitsu Kosan Co., Ltd. | Condensed fluoranthene compound, organic electroluminescence element material including same, organic electroluminescence element using same, and electronic device |
| WO2014196580A1 (en) | 2013-06-04 | 2014-12-11 | 出光興産株式会社 | Nitrogen-containing heterocyclic derivative, organic electroluminescence element material using same, and organic electroluminescence element and electronic device using same |
| KR102194819B1 (en) | 2013-08-27 | 2020-12-24 | 삼성디스플레이 주식회사 | Organic light emitting diode comprising the same |
| CN105473577B (en) * | 2013-08-29 | 2019-05-14 | 株式会社半导体能源研究所 | Heterocyclic compound, light-emitting element, light-emitting device, electronic equipment, and lighting device |
| WO2015093878A1 (en) * | 2013-12-18 | 2015-06-25 | Rohm And Haas Electronic Materials Korea Ltd. | Organic electroluminescent compound, and multi-component host material and organic electroluminescent device comprising the same |
| CN106255687B (en) | 2014-04-30 | 2020-06-05 | 默克专利有限公司 | Material for electronic devices |
| KR102372950B1 (en) * | 2014-05-02 | 2022-03-14 | 롬엔드하스전자재료코리아유한회사 | Organic Electroluminescent Compounds and Organic Electroluminescent Device Comprising the Same |
| TWI555752B (en) * | 2014-06-27 | 2016-11-01 | 喜星素材股份有限公司 | Heterocyclic compound and organic light-emitting device using same |
| KR102273047B1 (en) * | 2014-06-30 | 2021-07-06 | 삼성디스플레이 주식회사 | Organic light-emitting device |
| KR101539729B1 (en) * | 2014-07-23 | 2015-07-28 | 성균관대학교산학협력단 | Organic electroluminescent compound, producing method of the same, and organic electroluminescent device including the same |
| US10355222B2 (en) | 2015-02-06 | 2019-07-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| JP2018525331A (en) | 2015-06-10 | 2018-09-06 | メルク パテント ゲーエムベーハー | Materials for organic electroluminescent devices |
| US10367151B2 (en) * | 2015-06-23 | 2019-07-30 | Samsung Electronics Co., Ltd. | Condensed cyclic compound and organic light-emitting device including the same |
| KR102510392B1 (en) | 2015-09-10 | 2023-03-16 | 삼성디스플레이 주식회사 | Carbazole-based compound and organic light-emitting device including the same |
| KR102552274B1 (en) | 2015-10-08 | 2023-07-07 | 삼성디스플레이 주식회사 | Condensed-cyclic compound and organic light emitting device comprising the same |
| KR102292768B1 (en) * | 2015-10-13 | 2021-08-25 | 롬엔드하스전자재료코리아유한회사 | Organic Electroluminescent Compounds and Organic Electroluminescent Devices Comprising the Same |
| KR102456075B1 (en) | 2015-10-15 | 2022-10-19 | 삼성디스플레이 주식회사 | Compound and Organic light emitting device comprising same |
| KR101984082B1 (en) * | 2016-05-27 | 2019-05-31 | 주식회사 엘지화학 | Organic light emitting device |
| KR102520279B1 (en) * | 2016-10-14 | 2023-04-12 | 롬엔드하스전자재료코리아유한회사 | Organic Electroluminescence Device |
| KR102111149B1 (en) * | 2016-11-11 | 2020-05-14 | 주식회사 엘지화학 | Organic light emitting device |
| WO2018182294A1 (en) | 2017-03-27 | 2018-10-04 | 주식회사 엘지화학 | Benzocarbazole-based compound and organic light-emitting device comprising same |
| US11192884B2 (en) | 2017-03-27 | 2021-12-07 | Lg Chem, Ltd. | Heterocyclic compound and organic light-emitting device comprising same |
| KR102118142B1 (en) | 2017-09-27 | 2020-06-02 | 삼성에스디아이 주식회사 | Compound for organic optoelectronic device, and organic optoelectronic device and display device |
| CN110330506A (en) * | 2019-04-29 | 2019-10-15 | 宁波卢米蓝新材料有限公司 | A kind of fused-polycyclic compounds and its preparation method and application |
| KR20220029590A (en) * | 2019-07-08 | 2022-03-08 | 소니그룹주식회사 | Compounds, polymers and organic materials, and optical devices, optical components and image display devices using the same |
| CN112279847B (en) * | 2019-07-25 | 2021-12-03 | 南京高光半导体材料有限公司 | Green phosphorescent host material and OLED (organic light emitting diode) light emitting device containing same |
| KR20230049947A (en) * | 2021-10-07 | 2023-04-14 | 엘지디스플레이 주식회사 | Organic light emitting diode and organic light emitting device having the same |
| WO2025192265A1 (en) * | 2024-03-13 | 2025-09-18 | キヤノン株式会社 | Organic compound and organic light-emitting element |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1808433A1 (en) * | 2004-11-04 | 2007-07-18 | Idemitsu Kosan Company Limited | Compound containing fused ring and organic electroluminescent element employing the same |
| US20080122344A1 (en) * | 2006-11-24 | 2008-05-29 | Samsung Electronics Co., Ltd. | Organic light emitting compound and organic light emitting device comprising the same, and method of manufacturing the organic light emitting device |
| EP2138551A2 (en) * | 2008-06-25 | 2009-12-30 | Gracel Display Inc. | Fluorene-derivatives and organic electroluminescent device using the same |
| EP2149555A1 (en) * | 2008-07-30 | 2010-02-03 | Samsung Mobile Display Co., Ltd. | Amine-based compound, organic light emitting device comprising the amine-based compound, and flat panel display device including the organic light emitting device |
| WO2010021524A2 (en) * | 2008-08-22 | 2010-02-25 | 주식회사 엘지화학 | Material for organic electronic device, and organic electronic device using the same |
| US20100051914A1 (en) * | 2008-09-03 | 2010-03-04 | Samsung Mobile Display Co., Ltd. | Silicon-containing compound and organic electroluminescent device employing the same |
| WO2011019156A1 (en) * | 2009-08-10 | 2011-02-17 | Rohm And Haas Electronic Materials Korea Ltd. | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Family Cites Families (12)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP4116225B2 (en) * | 1999-10-27 | 2008-07-09 | 富士フイルム株式会社 | Novel condensed heterocyclic compound, light emitting device material, and light emitting device using the same |
| KR100957288B1 (en) * | 2002-03-15 | 2010-05-12 | 이데미쓰 고산 가부시키가이샤 | Material for organic electroluminescent device and organic electroluminescent device using same |
| JP4580342B2 (en) * | 2003-01-24 | 2010-11-10 | 出光興産株式会社 | Organic electroluminescence device |
| US20040247933A1 (en) * | 2003-06-03 | 2004-12-09 | Canon Kabushiki Kaisha | Bipolar asymmetric carbazole-based host materials for electrophosphorescent guest-host OLED systems |
| JP4585786B2 (en) * | 2004-04-01 | 2010-11-24 | キヤノン株式会社 | Light emitting element and display device |
| US7329722B2 (en) * | 2004-08-30 | 2008-02-12 | Samsung Electronics Co., Ltd | Polymeric charge transport materials having carbazolyl repeating units |
| JP5055818B2 (en) * | 2006-04-19 | 2012-10-24 | コニカミノルタホールディングス株式会社 | ORGANIC ELECTROLUMINESCENT ELEMENT MATERIAL, ORGANIC ELECTROLUMINESCENT ELEMENT, DISPLAY DEVICE AND LIGHTING DEVICE |
| JP5018138B2 (en) * | 2007-03-02 | 2012-09-05 | Jnc株式会社 | Luminescent material and organic electroluminescent device using the same |
| US9145363B2 (en) * | 2009-02-18 | 2015-09-29 | Idemitsu Kosan Co., Ltd. | Aromatic amine derivative and organic electroluminescent element |
| WO2011014039A1 (en) * | 2009-07-31 | 2011-02-03 | Rohm And Haas Electronic Materials Korea Ltd. | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| KR101219492B1 (en) * | 2009-12-11 | 2013-01-28 | 삼성디스플레이 주식회사 | Compound Containing 5-Membered Heterocycle And Organic Electronic Element Using The Same, Terminal Thereof |
| KR20120057561A (en) * | 2010-04-20 | 2012-06-05 | 이데미쓰 고산 가부시키가이샤 | Biscarbazole derivative, material for organic electroluminescence device and organic electroluminescence device using the same |
-
2011
- 2011-09-14 KR KR1020110092422A patent/KR101477614B1/en active Active
- 2011-09-16 CN CN2011800555279A patent/CN103221406A/en active Pending
- 2011-09-16 WO PCT/KR2011/006810 patent/WO2012036482A1/en not_active Ceased
- 2011-09-16 TW TW100133700A patent/TWI560258B/en active
- 2011-09-16 CN CN201810756909.8A patent/CN109020960B/en active Active
- 2011-09-16 EP EP11825442.4A patent/EP2616462A4/en not_active Withdrawn
- 2011-09-16 JP JP2013529060A patent/JP2013539750A/en active Pending
-
2016
- 2016-09-05 JP JP2016172842A patent/JP2017031167A/en active Pending
-
2018
- 2018-10-10 JP JP2018192058A patent/JP6672416B2/en active Active
Patent Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1808433A1 (en) * | 2004-11-04 | 2007-07-18 | Idemitsu Kosan Company Limited | Compound containing fused ring and organic electroluminescent element employing the same |
| US20080122344A1 (en) * | 2006-11-24 | 2008-05-29 | Samsung Electronics Co., Ltd. | Organic light emitting compound and organic light emitting device comprising the same, and method of manufacturing the organic light emitting device |
| EP2138551A2 (en) * | 2008-06-25 | 2009-12-30 | Gracel Display Inc. | Fluorene-derivatives and organic electroluminescent device using the same |
| EP2149555A1 (en) * | 2008-07-30 | 2010-02-03 | Samsung Mobile Display Co., Ltd. | Amine-based compound, organic light emitting device comprising the amine-based compound, and flat panel display device including the organic light emitting device |
| WO2010021524A2 (en) * | 2008-08-22 | 2010-02-25 | 주식회사 엘지화학 | Material for organic electronic device, and organic electronic device using the same |
| US20100051914A1 (en) * | 2008-09-03 | 2010-03-04 | Samsung Mobile Display Co., Ltd. | Silicon-containing compound and organic electroluminescent device employing the same |
| WO2011019156A1 (en) * | 2009-08-10 | 2011-02-17 | Rohm And Haas Electronic Materials Korea Ltd. | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
Cited By (41)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103502243A (en) * | 2011-03-08 | 2014-01-08 | 罗门哈斯电子材料韩国有限公司 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| CN104447709A (en) * | 2011-03-08 | 2015-03-25 | 罗门哈斯电子材料韩国有限公司 | Novel compounds for organic electronic material and organic electroluminescence device using the same |
| CN103502243B (en) * | 2011-03-08 | 2017-02-15 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compounds and organic electroluminescent device using the same |
| CN103534251A (en) * | 2011-03-25 | 2014-01-22 | 罗门哈斯电子材料韩国有限公司 | Novel compounds for organic electronic material and organic electroluminescent device using the same |
| CN103732590A (en) * | 2011-05-30 | 2014-04-16 | 罗门哈斯电子材料韩国有限公司 | New compounds for organic electronic materials and organic electroluminescent devices using the compounds |
| CN104066728A (en) * | 2011-11-18 | 2014-09-24 | 罗门哈斯电子材料韩国有限公司 | Novel organic electroluminescence compounds and organic electroluminescence device comprising the same |
| CN105408311B (en) * | 2013-10-03 | 2019-03-01 | 出光兴产株式会社 | Compound, organic electroluminescent element, and electronic device |
| CN105408311A (en) * | 2013-10-03 | 2016-03-16 | 出光兴产株式会社 | Compound, organic electroluminescent element, and electronic device |
| GB2534106B (en) * | 2013-12-16 | 2021-03-24 | Shenzhen China Star Optoelect | Blue fluorescent organic material and organic light emitting diode panel thereof |
| GB2534106A (en) * | 2013-12-16 | 2016-07-13 | Shenzhen China Star Optoelect | Blue fluorescent organic material and light emitting diode panel |
| EA029982B1 (en) * | 2013-12-16 | 2018-06-29 | Шэньчжэнь Чайна Стар Оптоэлектроникс Текнолоджи Ко., Лтд. | Blue fluorescent organic material and organic light emitting diode panel thereof |
| WO2015089853A1 (en) * | 2013-12-16 | 2015-06-25 | 深圳市华星光电技术有限公司 | Blue fluorescent organic material and organic light emitting diode panel |
| CN105794010A (en) * | 2013-12-18 | 2016-07-20 | 罗门哈斯电子材料韩国有限公司 | Organic electroluminescent compound and multi-component host material and organic electroluminescent device comprising same |
| US10446764B2 (en) | 2014-05-22 | 2019-10-15 | Samsung Sdi Co., Ltd. | Organic compound, composition, organic optoelectric device, and display device |
| CN106414407B (en) * | 2014-05-22 | 2019-07-12 | 三星Sdi株式会社 | Organic compound, composition, organic photoelectric device and display device |
| CN106414407A (en) * | 2014-05-22 | 2017-02-15 | 三星Sdi株式会社 | Organic compound, composition, organic electroluminescent device, and display device |
| CN105980520A (en) * | 2014-08-20 | 2016-09-28 | 株式会社Lg化学 | organic light emitting device |
| US9947878B2 (en) | 2014-08-20 | 2018-04-17 | Lg Chem, Ltd. | Organic light-emitting device |
| CN105440027A (en) * | 2014-09-18 | 2016-03-30 | 株式会社Lg化学 | Heterocyclic compound and organic light-emitting device comprising same |
| CN105440027B (en) * | 2014-09-18 | 2018-10-12 | 株式会社Lg化学 | Heterocyclic compound and organic light-emitting device comprising same |
| CN106715393A (en) * | 2014-10-14 | 2017-05-24 | 株式会社Lg化学 | Nitrogen-containing polycyclic compound and organic light-emitting device using same |
| CN106715393B (en) * | 2014-10-14 | 2019-12-17 | 株式会社Lg化学 | Nitrogen-containing polycyclic compound and organic light-emitting device using same |
| CN104342124A (en) * | 2014-10-23 | 2015-02-11 | 上海道亦化工科技有限公司 | Benzimidazole based phosphorescent luminescent main body material |
| CN105601621A (en) * | 2014-11-18 | 2016-05-25 | 株式会社Lg化学 | Nitrogen-containing polycyclic compound and organic electroluminescence device using same |
| CN105601621B (en) * | 2014-11-18 | 2019-05-28 | 株式会社Lg化学 | Nitrogen-containing polycyclic compound and organic electroluminescent device using the same |
| CN105837392B (en) * | 2016-04-15 | 2018-06-19 | 中节能万润股份有限公司 | A kind of electroluminescent organic material and the electroluminescent device comprising the material |
| CN105837392A (en) * | 2016-04-15 | 2016-08-10 | 中节能万润股份有限公司 | Organic electroluminescence material and electroluminescence device including same |
| CN106543152A (en) * | 2016-10-25 | 2017-03-29 | 上海道亦化工科技有限公司 | Phosphorescence host compound and its organic luminescent device based on naphtho- benzofuran |
| CN109790118A (en) * | 2016-12-13 | 2019-05-21 | 广州华睿光电材料有限公司 | Conjugated polymer and its application in organic electronic device |
| US11161933B2 (en) | 2016-12-13 | 2021-11-02 | Guangzhou Chinaray Optoelectronic Materials Ltd. | Conjugated polymer and use thereof in organic electronic device |
| WO2018108108A1 (en) * | 2016-12-13 | 2018-06-21 | 广州华睿光电材料有限公司 | Conjugated polymer and use thereof in organic electronic device |
| CN106749341A (en) * | 2017-01-18 | 2017-05-31 | 江西冠能光电材料有限公司 | Balancing charge injects organic semiconductor and its Organic Light Emitting Diode application |
| CN108623480A (en) * | 2017-03-17 | 2018-10-09 | 吉林奥来德光电材料股份有限公司 | New organic luminescent compounds and use its organic electroluminescence device |
| CN109721588A (en) * | 2017-10-27 | 2019-05-07 | 北京鼎材科技有限公司 | Organic electroluminescence device compound and the organic electroluminescence device for using it |
| CN109824671A (en) * | 2017-11-23 | 2019-05-31 | 北京鼎材科技有限公司 | A kind of quinazolinotriazole derivative and its application in the field of organic electroluminescence |
| CN109824672A (en) * | 2017-11-23 | 2019-05-31 | 北京鼎材科技有限公司 | A kind of quinazo triazole derivatives and its application in field of organic electroluminescence |
| CN113227084A (en) * | 2018-12-26 | 2021-08-06 | Lt素材株式会社 | Compound, composition, organic photoelectric device and display device |
| CN113260615A (en) * | 2018-12-27 | 2021-08-13 | Lt素材株式会社 | Polycyclic compound and organic light-emitting element including same |
| CN113260615B (en) * | 2018-12-27 | 2025-03-28 | Lt素材株式会社 | Polycyclic compound and organic light-emitting element comprising the same |
| CN110143952A (en) * | 2019-06-17 | 2019-08-20 | 上海天马有机发光显示技术有限公司 | A kind of compound, display panel and display device |
| CN114031609A (en) * | 2021-12-14 | 2022-02-11 | 北京燕化集联光电技术有限公司 | Compound containing carbazole and quinazoline structure and application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| EP2616462A1 (en) | 2013-07-24 |
| TW201224110A (en) | 2012-06-16 |
| KR20120030009A (en) | 2012-03-27 |
| CN109020960A (en) | 2018-12-18 |
| TWI560258B (en) | 2016-12-01 |
| JP2013539750A (en) | 2013-10-28 |
| KR101477614B1 (en) | 2014-12-31 |
| JP2017031167A (en) | 2017-02-09 |
| JP6672416B2 (en) | 2020-03-25 |
| WO2012036482A1 (en) | 2012-03-22 |
| EP2616462A4 (en) | 2014-02-19 |
| JP2019050382A (en) | 2019-03-28 |
| CN109020960B (en) | 2022-05-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109020960B (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| JP6543211B2 (en) | Electroluminescent device using electroluminescent compound as luminescent material | |
| CN103228661A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN103249722B (en) | New compounds for organic electronic materials and organic electroluminescent devices using the compounds | |
| CN102918134B (en) | Organic electroluminescent compound and organic electroluminescent device using the same | |
| CN102449109B (en) | Novel organic electroluminescent compound and organic electroluminescent device using the same | |
| JP5771644B2 (en) | Material for organic electroluminescence device and organic electroluminescence device using the same | |
| CN102933531B (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN103384712B (en) | The organic electroluminescence device of new organic electroluminescent compounds and this compound of use | |
| CN103189469A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN103298800A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN102449106A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20110097784A (en) | Aromatic Amine Derivatives and Organic Electroluminescent Devices | |
| CN104364344A (en) | Biscarbazole Derivative Host Materials and Red Emitters for OLED Emitting Region | |
| CN105359292A (en) | Combination of dopant compound and host compound and organic electroluminescent device comprising the combination | |
| WO2011162162A1 (en) | Light-emitting device material and light-emitting device | |
| CN102933530A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN103857680A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| TW202026399A (en) | A plurality of host materials and organic electroluminescent device comprising the same | |
| KR20120021215A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| KR20190101281A (en) | Organic electroluminescent compound and organic electroluminescent device comprising the same | |
| CN102574871A (en) | Novel organic electroluminescent compounds and organic electroluminescent device using the same | |
| CN113321588B (en) | Nitrogen-containing compound, electronic component, and electronic device | |
| CN117466852A (en) | Organic compounds, organic electroluminescent devices and electronic devices |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| RJ01 | Rejection of invention patent application after publication | ||
| RJ01 | Rejection of invention patent application after publication |
Application publication date: 20130724 |