CN102204900A - Application of sodium oxamate in preparing drugs for radiotherapy sensitization - Google Patents
Application of sodium oxamate in preparing drugs for radiotherapy sensitization Download PDFInfo
- Publication number
- CN102204900A CN102204900A CN 201110093858 CN201110093858A CN102204900A CN 102204900 A CN102204900 A CN 102204900A CN 201110093858 CN201110093858 CN 201110093858 CN 201110093858 A CN201110093858 A CN 201110093858A CN 102204900 A CN102204900 A CN 102204900A
- Authority
- CN
- China
- Prior art keywords
- sodium oxamate
- radiotherapy
- cells
- application
- oxamate
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
- RQVZIJIQDCGIKI-UHFFFAOYSA-M sodium;oxamate Chemical compound [Na+].NC(=O)C([O-])=O RQVZIJIQDCGIKI-UHFFFAOYSA-M 0.000 title claims abstract description 34
- 238000001959 radiotherapy Methods 0.000 title claims abstract description 18
- 239000003814 drug Substances 0.000 title claims abstract description 17
- 229940079593 drug Drugs 0.000 title abstract description 12
- 206010070834 Sensitisation Diseases 0.000 title abstract description 4
- 230000008313 sensitization Effects 0.000 title abstract description 4
- 238000002360 preparation method Methods 0.000 claims abstract description 5
- 208000002454 Nasopharyngeal Carcinoma Diseases 0.000 claims description 6
- 206010061306 Nasopharyngeal cancer Diseases 0.000 claims description 6
- 201000011216 nasopharynx carcinoma Diseases 0.000 claims description 6
- 210000004881 tumor cell Anatomy 0.000 abstract description 5
- 230000000694 effects Effects 0.000 abstract description 4
- 230000035945 sensitivity Effects 0.000 abstract description 4
- 230000001235 sensitizing effect Effects 0.000 abstract description 4
- 231100000331 toxic Toxicity 0.000 abstract description 2
- 230000002588 toxic effect Effects 0.000 abstract description 2
- 210000004027 cell Anatomy 0.000 description 35
- 230000005855 radiation Effects 0.000 description 8
- 230000004083 survival effect Effects 0.000 description 8
- 206010028980 Neoplasm Diseases 0.000 description 5
- 239000002609 medium Substances 0.000 description 5
- 201000011510 cancer Diseases 0.000 description 4
- 239000003153 chemical reaction reagent Substances 0.000 description 4
- 238000002474 experimental method Methods 0.000 description 4
- 230000012010 growth Effects 0.000 description 4
- IZTQOLKUZKXIRV-YRVFCXMDSA-N sincalide Chemical compound C([C@@H](C(=O)N[C@@H](CCSC)C(=O)NCC(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC=1C=CC=CC=1)C(N)=O)NC(=O)[C@@H](N)CC(O)=O)C1=CC=C(OS(O)(=O)=O)C=C1 IZTQOLKUZKXIRV-YRVFCXMDSA-N 0.000 description 4
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 4
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 3
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 3
- 108010087230 Sincalide Proteins 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 230000003698 anagen phase Effects 0.000 description 3
- 244000309466 calf Species 0.000 description 3
- 238000010609 cell counting kit-8 assay Methods 0.000 description 3
- 238000004113 cell culture Methods 0.000 description 3
- 230000005251 gamma ray Effects 0.000 description 3
- 230000000637 radiosensitizating effect Effects 0.000 description 3
- 210000002966 serum Anatomy 0.000 description 3
- 239000000243 solution Substances 0.000 description 3
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- 231100000111 LD50 Toxicity 0.000 description 2
- 229930182555 Penicillin Natural products 0.000 description 2
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 239000006143 cell culture medium Substances 0.000 description 2
- 230000010261 cell growth Effects 0.000 description 2
- 238000004737 colorimetric analysis Methods 0.000 description 2
- 238000012258 culturing Methods 0.000 description 2
- 201000010099 disease Diseases 0.000 description 2
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 2
- 238000000034 method Methods 0.000 description 2
- 229940049954 penicillin Drugs 0.000 description 2
- 239000000843 powder Substances 0.000 description 2
- 239000002534 radiation-sensitizing agent Substances 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 229960005322 streptomycin Drugs 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 230000036962 time dependent Effects 0.000 description 2
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 101800005151 Cholecystokinin-8 Proteins 0.000 description 1
- 102400000888 Cholecystokinin-8 Human genes 0.000 description 1
- 208000000461 Esophageal Neoplasms Diseases 0.000 description 1
- 206010030155 Oesophageal carcinoma Diseases 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- 102000004142 Trypsin Human genes 0.000 description 1
- 108090000631 Trypsin Proteins 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 238000006243 chemical reaction Methods 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 201000004101 esophageal cancer Diseases 0.000 description 1
- 239000012737 fresh medium Substances 0.000 description 1
- 230000004153 glucose metabolism Effects 0.000 description 1
- ZDXPYRJPNDTMRX-UHFFFAOYSA-N glutamine Natural products OC(=O)C(N)CCC(N)=O ZDXPYRJPNDTMRX-UHFFFAOYSA-N 0.000 description 1
- 201000010536 head and neck cancer Diseases 0.000 description 1
- 208000014829 head and neck neoplasm Diseases 0.000 description 1
- 230000005865 ionizing radiation Effects 0.000 description 1
- 231100000636 lethal dose Toxicity 0.000 description 1
- 239000000463 material Substances 0.000 description 1
- 238000005259 measurement Methods 0.000 description 1
- 239000010413 mother solution Substances 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- -1 organic synthesis Substances 0.000 description 1
- 235000006408 oxalic acid Nutrition 0.000 description 1
- SOWBFZRMHSNYGE-UHFFFAOYSA-N oxamic acid Chemical compound NC(=O)C(O)=O SOWBFZRMHSNYGE-UHFFFAOYSA-N 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 239000012588 trypsin Substances 0.000 description 1
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 1
Images
Landscapes
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
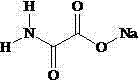
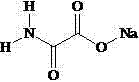
本发明属于放疗增敏药物领域,涉及草氨酸钠的新用途,即在制备放疗增敏药物中的应用,所述草氨酸钠的结构式如下所示:
。本发明所述草氨酸钠可以用于制备放射治疗增敏药物,并且可以在无毒副作用的低浓度下,提高肿瘤细胞的放疗敏感性。The invention belongs to the field of radiotherapy sensitizing drugs, and relates to the new application of sodium oxamate, that is, the application in the preparation of radiotherapy sensitizing drugs. The structural formula of the sodium oxamate is as follows:
. The sodium oxamate described in the present invention can be used to prepare radiotherapy sensitization drugs, and can improve the radiotherapy sensitivity of tumor cells at a low concentration without toxic and side effects.Description
技术领域technical field
本发明属于放疗增敏药物领域,涉及草氨酸钠的新用途,即在制备放疗增敏药物中的应用。The invention belongs to the field of radiotherapy sensitizing drugs, and relates to a new application of sodium oxamate, that is, an application in preparing radiotherapy sensitizing drugs.
背景技术Background technique
恶性肿瘤是一种常见病、多发病,目前已成为严重威胁人类生命健康的主要疾病之一。放射治疗作为肿瘤治疗的三大主要手段之一,已广泛和常规地应用在肿瘤治疗中。据国内各大肿瘤防治中心统计,经诊治的肿瘤患者约有65%-75%需用放疗,尤其是头颈部肿瘤、前列腺癌、食管癌等。但即便是放射治疗效果较好的鼻咽癌患者,其放射治疗后仍会发生较高的局部复发和耐辐射等问题。而仅仅依靠单纯提高辐射剂量又同时会带来更严重的副作用。因此,降低肿瘤细胞的辐射抗性、提高肿瘤细胞辐射敏感性,成为近年来肿瘤治疗研究的热点和难点。Malignant tumor is a common and frequently-occurring disease, and has become one of the major diseases that seriously threaten human life and health. As one of the three main means of tumor treatment, radiotherapy has been widely and routinely used in tumor treatment. According to the statistics of major cancer prevention and treatment centers in China, about 65%-75% of cancer patients who have been diagnosed and treated need radiotherapy, especially head and neck cancer, prostate cancer, and esophageal cancer. However, even for patients with nasopharyngeal carcinoma who have a good radiotherapy effect, there will still be high local recurrence and radiation resistance after radiotherapy. But relying on simply increasing the radiation dose will bring more serious side effects at the same time. Therefore, reducing the radiation resistance of tumor cells and increasing the radiation sensitivity of tumor cells has become a hot and difficult point in cancer treatment research in recent years.
草氨酸钠(sodium oxamate),纯化学制药,由草氨酸或草酸酰胺乙酯与氢氧化钠反应制得,广泛用于在生化试剂、有机合成、制药等方面。国外有研究发现草氨酸钠可通过影响乳腺癌MDA-MB-231细胞内的糖代谢活动,而影响该细胞的生长。Sodium oxamate (sodium oxamate), a pure chemical pharmaceutical, is prepared by the reaction of oxalic acid or amidoethyl oxamate and sodium hydroxide, and is widely used in biochemical reagents, organic synthesis, and pharmaceuticals. Foreign studies have found that sodium oxamate can affect the growth of breast cancer MDA-MB-231 cells by affecting the glucose metabolism in the cells.
现有技术中未见关于草氨酸钠放疗增敏方面的报道。There is no report on radiosensitization of sodium oxamate in the prior art.
发明内容Contents of the invention
本发明的发明目的是提供草氨酸钠的新用途,即在制备放疗增敏药物中的应用。The purpose of the present invention is to provide a new application of sodium oxamate, that is, the application in the preparation of radiosensitizing drugs.
为达到上述发明目的,本发明采用的技术方案是:草氨酸钠在制备放疗增敏药物中的应用,所述草氨酸钠的结构式如下所示: 。In order to achieve the above-mentioned purpose of the invention, the technical solution adopted in the present invention is: the application of sodium oxamate in the preparation of radiosensitizing drugs, and the structural formula of said sodium oxamate is as follows: .
发明人将不同浓度的草氨酸钠直接加入指数生长的鼻咽癌CNE-1细胞培养基中,细胞继续培养至12、24或48小时不同时间后收样,采用CCK8比色方法检测细胞的生长,结果显示:草氨酸钠可明显抑制CNE-1细胞的生长,并呈现显著的剂量和时间依赖关系,24小时和48小时草氨酸钠处理的细胞半数致死浓度(IC50)分别为2.3 μmol/L和1.6 μmol/L。The inventors directly added different concentrations of sodium oxamate to the exponentially growing nasopharyngeal carcinoma CNE-1 cell culture medium, and the cells continued to be cultured for 12, 24 or 48 hours at different times before collecting samples, and the CCK8 colorimetric method was used to detect the cell growth. growth, the results showed that sodium oxamate could significantly inhibit the growth of CNE-1 cells, and showed a significant dose- and time-dependent relationship. The median lethal concentrations (IC 50 ) of cells treated with sodium oxamate in 24 hours and 48 hours were respectively 2.3 μmol/L and 1.6 μmol/L.
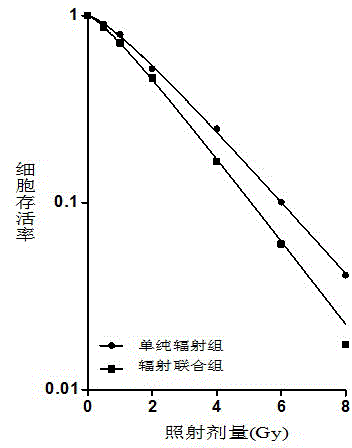
同时,发明人采用低浓度0.75μmol/L草氨酸钠(采用草氨酸钠这个浓度处理24小时后>90%以上的细胞成活)预处理细胞24小时后接受不同剂量(0-8 Gy)的60Co-γ线照射,照射后再继续培养24小时,然后更换新鲜培养基,继续培养2-3周,以完成细胞克隆形成实验,采用单击多靶模型拟合两组细胞存活曲线,结果发现:与单γ线照射组相比,0.75 μmol/L草氨酸钠预处理后的辐照联合组的生存曲线明显下移,且随着照射剂量的升高,其下移越明显,各辐射敏感性检测指标显示,在暴露于电离辐射前72小时内,给与1 μmol/L以下剂量的草氨酸钠,可以肿瘤细胞的放疗敏感性,从而有效地降低细胞存活率。At the same time, the inventor pretreated the cells with a low concentration of 0.75 μmol/L sodium oxamate (more than 90% of the cells survived after 24 hours of treatment with this concentration of sodium oxamate) and received different doses (0-8 Gy) for 24 hours. The 60 Co-γ ray was irradiated, and the culture was continued for 24 hours after irradiation, and then the fresh medium was replaced, and the culture was continued for 2-3 weeks to complete the cell clone formation experiment. The single-click multi-target model was used to fit the two groups of cell survival curves. The results showed that: compared with the single γ-ray irradiation group, the survival curve of the combined irradiation group after 0.75 μmol/L sodium oxamate pretreatment significantly shifted down, and the downward shift was more obvious with the increase of the irradiation dose. The radiosensitivity detection indicators showed that giving sodium oxamate at a dose below 1 μmol/L within 72 hours before exposure to ionizing radiation could reduce the radiosensitivity of tumor cells and effectively reduce the cell survival rate.
优选的技术方案中,所述放疗增敏药物为适用鼻咽癌的放疗增敏药物。In a preferred technical solution, the radiosensitizing drug is a radiosensitizing drug suitable for nasopharyngeal carcinoma.
由于上述技术方案运用,本发明与现有技术相比具有下列优点:Due to the use of the above-mentioned technical solutions, the present invention has the following advantages compared with the prior art:
1.本发明所述草氨酸钠可以用于制备放射治疗增敏药物,并且可以在无毒副作用的低浓度下,提高肿瘤细胞的放疗敏感性。1. The sodium oxamate described in the present invention can be used to prepare radiotherapy sensitization drugs, and can improve the radiotherapy sensitivity of tumor cells at a low concentration without toxic and side effects.
附图说明Description of drawings
图1是实施例一中小于5 μmol/L草氨酸钠 处理24小时对CNE-1细胞生长和增殖的影响;Fig. 1 is the influence that is less than 5 μ mol/L sodium oxamate treatment 24 hours on CNE-1 cell growth and proliferation in embodiment one;
图2是是实施例二中草氨酸钠预处理CNE-1细胞的放射治疗敏感性改变。Fig. 2 is the radiotherapy sensitivity change of CNE-1 cells pretreated with sodium oxamate in Example 2.
具体实施方式Detailed ways
下面结合附图及实施例对本发明作进一步描述:The present invention will be further described below in conjunction with accompanying drawing and embodiment:
材料:细胞株和细胞培养:人类鼻咽癌CNE-1细胞株为本研究室传代并保存的细胞系。细胞培养于含10%热灭活小牛血清的DMEM培养液(内含100 U/mL青霉素和100 μg/mL链霉素),并在5%CO2、饱和湿度的37oC培养箱中培养。药物与主要试剂:药物草氨酸钠,溶于无水乙醇中,配成母液为5mmol/L于4度冰箱中保存。1640培养基干粉,小牛血清,胰酶粉末购自Gibco公司;CCK-8试剂购于日本同仁化学研究所。Materials: Cell lines and cell culture: Human nasopharyngeal carcinoma CNE-1 cell line is a cell line passed down and preserved in our laboratory. Cells were cultured in DMEM medium containing 10% heat-inactivated calf serum (containing 100 U/mL penicillin and 100 μg/mL streptomycin), and placed in a 37 o C incubator with 5% CO 2 and saturated humidity. nourish. Drugs and main reagents: drug sodium oxamate, dissolved in absolute ethanol, made into a mother solution of 5mmol/L, and stored in a 4-degree refrigerator. 1640 medium dry powder, calf serum, and trypsin powder were purchased from Gibco; CCK-8 reagent was purchased from Dojin Chemical Research Institute, Japan.
实施例一:Embodiment one:
(1) 细胞培养:将细胞培养于含10%小牛血清, 谷氨酸胺及100万U/L青霉素和链霉素的DMEM培养基。细胞置于5%CO2,37℃培养箱内培养,每2-3天传代1次,取对数生长期细胞用于实验。(1) Cell culture: Cells were cultured in DMEM medium containing 10% calf serum, glutamine and 1 million U/L penicillin and streptomycin. The cells were cultured in a 5% CO 2 , 37°C incubator, passaged once every 2-3 days, and the cells in the logarithmic growth phase were used for experiments.
(2) CCK-8比色法:在药物处理前24小时将对数生长期细胞(5×104/mL)接种至96孔板中,每种浓度设6个平行孔。加入不同浓度的草氨酸钠继续培养12、24或48小时。细胞培养液中加入10 μL CCK-8试剂继续培养1-4小时。采用酶标仪测定在450 nm处的吸光度。最后采用细胞存活率=加药组值/对照组值×100%的公式计算细胞存活率。(2) CCK-8 colorimetric method: cells in logarithmic growth phase (5×10 4 /mL) were inoculated into 96-well plates 24 hours before drug treatment, and 6 parallel wells were set for each concentration. Different concentrations of sodium oxamate were added to continue culturing for 12, 24 or 48 hours. Add 10 μL CCK-8 reagent to the cell culture medium and continue culturing for 1-4 hours. The absorbance at 450 nm was measured using a microplate reader. Finally, the cell survival rate was calculated by the formula of cell survival rate=dosage group value/control group value×100%.
所得结果如图1所示,结果表明草氨酸钠可明显抑制CNE-1细胞的生长,并呈现显著的剂量和时间依赖关系。24小时和48小时草氨酸钠处理的细胞半数致死浓度(IC50)分别为2.3 μmol/L和1.6 μmol/L。The results obtained are shown in Figure 1, and the results show that sodium oxamate can significantly inhibit the growth of CNE-1 cells, and present a significant dose- and time-dependent relationship. The median lethal concentrations (IC 50 ) of cells treated with sodium oxamate at 24 hours and 48 hours were 2.3 μmol/L and 1.6 μmol/L, respectively.
实施例二Embodiment two
(1) 细胞培养同实施例一;(1) cell culture is the same as embodiment one;
(2) 细胞克隆形成方法:取对数生长期细胞,并按不同照射剂量要求接种不同数量的细胞到60 mm培养皿中(0, 2, 4, 6, 8 Gy剂量接种细胞数量分别为400, 2000, 6000, 1000和15000)。24小时后,草氨酸钠与辐射联合组加入0.75 μmol/L草氨酸钠处理24小时,对照组采用等量无水乙醇作为阴性对照。两组细胞同时接受不同剂量(0-8Gy)的γ-射线照射,照后继续培养24小时,两组均换成普通培养基,并继续直至培养皿中出现明显肉眼可见的细胞克隆时终止培养。弃培养基,PBS洗两次,并采用无水甲醇固定30分钟,Gimsa染液染色30分钟,流水冲洗后晾干。在显微镜下计数超过50个细胞的克隆集落,各计量点至少设3个平行样,取平均值,计算出细胞存活率。并利用Sigma Plot 9.0软件,运用单击多靶模型拟合曲线(参见图2),求出平均致死剂量(D0)、SER(辐射增敏比)、准阈剂量(Dq)、K及n值等,参见表1。(2) Cell clone formation method: Take cells in the logarithmic growth phase, and inoculate different numbers of cells into 60 mm culture dishes according to different irradiation dose requirements (400 cells for 0, 2, 4, 6, 8 Gy doses, respectively , 2000, 6000, 1000 and 15000). After 24 hours, the combined sodium oxamate and radiation group was treated with 0.75 μmol/L sodium oxamate for 24 hours, and the control group was treated with an equal amount of absolute ethanol as a negative control. The two groups of cells were irradiated with different doses (0-8Gy) of γ-rays at the same time, and continued to culture for 24 hours after irradiation. Both groups were replaced with ordinary medium, and continued to stop the culture until obvious cell clones visible to the naked eye appeared in the culture dish. . Discard the medium, wash twice with PBS, fix with anhydrous methanol for 30 minutes, stain with Gimsa stain for 30 minutes, rinse with running water and dry in the air. Count the clonal colonies of more than 50 cells under a microscope, set at least 3 parallel samples at each measurement point, take the average value, and calculate the cell survival rate. And using Sigma Plot 9.0 software, using the single-click multi-target model to fit the curve (see Figure 2), the average lethal dose (D 0 ), SER (Radiation Sensitization Ratio), quasi-threshold dose (D q ), K and For n values, etc., see Table 1.
步骤(2)中的照射条件为:采用60Co γ-射线照射,照射野为10 cm × 10 cm,源皮距为100 cm,采用单次照射,机头0度从培养皿上方照射,剂量率为0.58 Gy/min。The irradiation conditions in step (2) were as follows: 60 Co γ-ray irradiation was used, the irradiation field was 10 cm × 10 cm, the source-to-skin distance was 100 cm, a single irradiation was adopted, the machine head was irradiated from above the petri dish at 0 degrees, and the dose The rate was 0.58 Gy/min.
表1. 草氨酸钠作用于鼻咽癌CNE-1细胞的辐射克隆实验结果Table 1. Results of radiation cloning experiment of sodium oxamate on nasopharyngeal carcinoma CNE-1 cells
图2表明:采用单击多靶模型拟合单照射细胞组与草氨酸钠+照射细胞组的细胞存活曲线表明,与单线照射组相比,0.75μmol/L草氨酸钠预处理后的辐照联合组的生存曲线明显下移,且随着照射剂量的升高,其下移越明显。Figure 2 shows: the cell survival curves of the single-irradiation cell group and the sodium oxamate+irradiation cell group were fitted by the single-click multi-target model. The survival curve of combined irradiation group moved down significantly, and with the increase of irradiation dose, the downward shift was more obvious.
统计学处理:上述实施例的所有实验均重复3次以上,结果采用± S表示。采用SAS统计软件对相关数据进行t检验,以P<0.05为有显著性差异。Statistical processing: all experiments of above-mentioned embodiment are all repeated more than 3 times, the result adopts ±S means. SAS statistical software was used to conduct t -test on relevant data, and P <0.05 was considered significant difference.
Claims (2)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201110093858 CN102204900A (en) | 2011-04-14 | 2011-04-14 | Application of sodium oxamate in preparing drugs for radiotherapy sensitization |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 201110093858 CN102204900A (en) | 2011-04-14 | 2011-04-14 | Application of sodium oxamate in preparing drugs for radiotherapy sensitization |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN102204900A true CN102204900A (en) | 2011-10-05 |
Family
ID=44694145
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 201110093858 Pending CN102204900A (en) | 2011-04-14 | 2011-04-14 | Application of sodium oxamate in preparing drugs for radiotherapy sensitization |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102204900A (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114617900A (en) * | 2022-02-14 | 2022-06-14 | 苏州大学 | Application of dGMP in preparation of medicine for improving sensitivity of tumor to radiotherapy |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006024492A2 (en) * | 2004-08-30 | 2006-03-09 | Interstitial Therapeutics | Medical implant provided with inhibitors of atp synthesis |
-
2011
- 2011-04-14 CN CN 201110093858 patent/CN102204900A/en active Pending
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006024492A2 (en) * | 2004-08-30 | 2006-03-09 | Interstitial Therapeutics | Medical implant provided with inhibitors of atp synthesis |
Non-Patent Citations (1)
| Title |
|---|
| 《Breast Cancer Research》 20081015 Thornburg et al. Targeting aspartate aminotransferase in breast cancer 第10卷, 第5期 * |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN114617900A (en) * | 2022-02-14 | 2022-06-14 | 苏州大学 | Application of dGMP in preparation of medicine for improving sensitivity of tumor to radiotherapy |
| CN114617900B (en) * | 2022-02-14 | 2023-03-10 | 苏州大学 | Application of dGMP in preparation of medicine for improving sensitivity of tumor to radiotherapy |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| Zhou et al. | Paeonol enhances the sensitivity of human ovarian cancer cells to radiotherapy-induced apoptosis due to downregulation of the phosphatidylinositol-3-kinase/Akt/phosphatase and tensin homolog pathway and inhibition of vascular endothelial growth factor | |
| CN107823211A (en) | Application of the gucosamine in preparing ionising radiation and causing induced lung injury protective agents | |
| Pang et al. | The combination of the HDAC1 inhibitor SAHA and doxorubicin has synergic efficacy in triple negative breast cancer in vivo | |
| CN115531546B (en) | Combined antimetabolite for treating high-grade glioma and preparation method thereof | |
| Gong et al. | Radiation‑induced pulmonary injury accelerated pulmonary metastasis in a mouse model of breast cancer | |
| CN102362871A (en) | Preparation method of toad venom injection | |
| CN102579425B (en) | Caulis Spatholobi extract, application thereof and new application of isoliquiritigenin | |
| CN102204900A (en) | Application of sodium oxamate in preparing drugs for radiotherapy sensitization | |
| CN113230251A (en) | Albendazole and multiple myeloma clinical medicine composition and application | |
| Wang et al. | Huaier enhances the tumor-killing effect and reverses gemcitabine-induced stemness by suppressing FoxM1 | |
| CN102688228A (en) | Pharmaceutical composition containing apigenin, apigenin derivatives, oridonin and oridonin derivatives and application thereof | |
| CN109843289B (en) | A kind of diaryl chalcogenide compound against Candida albicans and its preparation and application | |
| CN117323336A (en) | Application of PRMT7 inhibitor SGC3027 in lung cancer treatment | |
| CN117338777A (en) | Targeting IKK epsilon kinase inhibitor and application thereof in antitumor drugs | |
| CN107519188B (en) | Gucosamine is preparing the application in radiotherapy hypersitization medicine | |
| CN116650474A (en) | New medical application of xanthine | |
| CN113846161A (en) | Pharmaceutical composition containing circ-ITCH and application thereof | |
| CN115245517A (en) | Application of dihydrotanshinone I in preparation of breast cancer and lung metastasis inhibitor thereof | |
| CN113181166A (en) | Application of curzerene alcohol in preparation of anti-lung cancer drugs and anti-lung cancer drugs | |
| CN105456247A (en) | Application of nafamostat mesylate in aspect of treating colorectal cancer | |
| Pang et al. | Huashi jiedu decoction blocks cell cycle and inhibits EMT of gastric cells via LncRNA-p21 | |
| CN112535732A (en) | Application of cyclin and inhibitor thereof in preparation of medicine for enhancing pancreatic cancer radiotherapy effect | |
| CN118593495B (en) | Application of quinoline alkaloid derivative in preparation of medicines for treating cancers | |
| CN115444927B (en) | Application of Amodex in the preparation of drugs for the treatment of triple-negative breast cancer | |
| Liu et al. | Comparative transcriptome analysis providing inhibitory mechanism of lung cancer A549 cells by radioactive 125I seed |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Application publication date: 20111005 |