CN109843289B - A kind of diaryl chalcogenide compound against Candida albicans and its preparation and application - Google Patents
A kind of diaryl chalcogenide compound against Candida albicans and its preparation and application Download PDFInfo
- Publication number
- CN109843289B CN109843289B CN201780025371.7A CN201780025371A CN109843289B CN 109843289 B CN109843289 B CN 109843289B CN 201780025371 A CN201780025371 A CN 201780025371A CN 109843289 B CN109843289 B CN 109843289B
- Authority
- CN
- China
- Prior art keywords
- candida albicans
- diaryl
- compound
- chalcogenide
- preparation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/095—Sulfur, selenium, or tellurium compounds, e.g. thiols
- A61K31/10—Sulfides; Sulfoxides; Sulfones
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
- A61K31/403—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil condensed with carbocyclic rings, e.g. carbazole
- A61K31/404—Indoles, e.g. pindolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P31/00—Antiinfectives, i.e. antibiotics, antiseptics, chemotherapeutics
- A61P31/10—Antimycotics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/02—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom condensed with one carbocyclic ring
- C07D209/04—Indoles; Hydrogenated indoles
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Epidemiology (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Oncology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Communicable Diseases (AREA)
- Inorganic Chemistry (AREA)
- Birds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
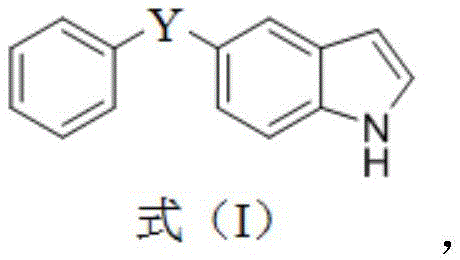
本发明公开了一种抗白色念珠菌的二芳基硫族化合物及其制备和应用。所述二芳基硫族化合物的结构如式(I)所示:
其中,Y为S或Se。该二芳基硫族化合物对白色念珠菌的粘附性、菌丝形态转换和致病性具有很好的抑制作用。而且,该化合物本身毒性较小,不影响人类细胞的生长,不易产生耐药性,在新型抗真菌药物的开发,尤其是抗白色念珠菌感染药物的开发方面具有很好的应用前景。The invention discloses a diaryl chalcogenide compound against Candida albicans and its preparation and application. The structure of the diaryl chalcogenide is shown in formula (I):
Wherein, Y is S or Se. The diaryl chalcogenide has a good inhibitory effect on the adhesion, hyphal morphological transformation and pathogenicity of Candida albicans. Moreover, the compound itself is less toxic, does not affect the growth of human cells, and is not easy to develop drug resistance, and has a good application prospect in the development of new antifungal drugs, especially in the development of anti-Candida albicans infection drugs.Description
Technical Field
The invention belongs to the technical field of biological medicines. More particularly, relates to a diaryl sulfur group compound for resisting candida albicans, and a preparation method and an application thereof.
Background
Candida albicans (Candida albicans) is a widely spread fungal disease in humans, is an important conditionally pathogenic fungus, usually causes acute, subacute or chronic infections, and is one of the most important pathogens of hospital-acquired infections at present. Candida albicans does not normally cause disease on mucosal surfaces of healthy persons, such as the oral cavity and intestinal tract, but causes serious systemic infection in patients with compromised or suppressed immune systems, such as chemotherapy patients, organ transplant patients or AIDS patients, with mortality rates as high as 40%.
At present, clinically, antifungal medicines are limited in species, wherein azole medicines (fluconazole) are widely applied, the fluconazole plays a role in inhibiting bacteria by inhibiting fungal replication, but the phenomenon of drug resistance is more serious along with abuse of antibiotics.
Disclosure of Invention
The invention aims to solve the technical problems of overcoming the defects and shortcomings of the existing anti-candida albicans drugs, designing the anti-candida albicans compound by using a novel antibacterial strategy, starting from the unique yeast-hypha diphasic property of candida albicans, designing a novel compound which is efficient, low in toxicity and not easy to generate drug resistance, and having important scientific significance and application prospect.
The invention aims to provide a diaryl sulfur group compound resisting candida albicans.
Another object of the invention is to provide a process for the preparation of said diarylchalcogenides.
It is a further object of the invention to provide the use of said diarylchalcogenides.
The above purpose of the invention is realized by the following technical scheme:
an anti-candida albicans diaryl sulfur group compound, which has a structure shown as a formula (I):
wherein Y is S or Se.
The preparation method of the diaryl sulfur group compound comprises the following steps: mixing a hydrogenated nitrogen-containing heterocyclic compound and a diaryl sulfur/selenium ether compound, adding dimethyl sulfoxide as a solvent and iodine as an accelerator, cooling to room temperature after stirring reaction is finished, diluting a reaction solution with ethyl acetate, washing and drying, removing the solvent by reduced pressure evaporation to obtain a crude product, and finally purifying by column chromatography to obtain the diaryl sulfur group compound.
Wherein, preferably, the molar ratio of the hydrogenated nitrogen-containing heterocyclic compound to the diarylsulfur/selenium ether compound to the iodine simple substance is 0.5-1.5: 0.5-1.5: 1 to 2.
More preferably, the molar ratio of the hydrogenated nitrogen-containing heterocyclic compound, the diarylsulfur/selenide compound, and the elemental iodine is 1: 1: 1.5.
preferably, the hydrogenated nitrogen-containing heterocyclic compound is indoline.
Preferably, the diaryl sulfide/selenide compound is diphenyl sulfide or diphenyl selenide.
Preferably, the dosage ratio of the dimethyl sulfoxide solvent to the iodine simple substance is 1-2 mL: 1 to 2 mmol.
More preferably, the dosage ratio of the dimethyl sulfoxide solvent to the iodine simple substance is 2 mL: 1.5 mmol.
Preferably, the stirring reaction condition is that the stirring reaction is carried out for 4-20 hours at the temperature of 100 ℃.
Preferably, the washing is several times with water.
Preferably, wash 3 times with water.
Yeast-hyphal diphasic is a characteristic feature of Candida albicans. The free yeast state is not toxic to the host during infection, and mainly performs the function of adhering to receptor tissues, then the yeast state is converted into hyphal state to promote invasion, and then the hyphal state is taken as a pathogen to enter the infected tissues to further exert toxic effect. The yeast-hyphal morphological transformation is an important process for Candida albicans to exert virulence. Therefore, the invention designs the compound for resisting the candida albicans by utilizing a novel bacteriostasis strategy, starts from the unique yeast-hypha diphasic property of the candida albicans, and designs a novel compound which has high efficiency, low toxicity and difficult generation of drug resistance in a targeted way. Then, Candida albicans (Candida albicans) is taken as a test object, the influence of the diaryl chalcogenide compound designed and synthesized by the invention on the adhesiveness, the hypha formation rate, the cytotoxicity and the survival rate of mice is examined, and the aim is to further influence the toxicity of Candida albicans in mammals by detecting the interference of the compound on the toxicity formation factor of Candida albicans. The results show that the compound has good inhibition effect on the adhesion, hyphal form transformation and pathogenicity of the candida albicans. Moreover, the compound has low toxicity and does not influence the growth of candida albicans and human cells.
Therefore, the application of the diaryl chalcogenide compound in preparing the medicines for resisting candida albicans infection and the medicines for preventing and/or treating infectious diseases caused by candida albicans are both within the protection scope of the invention.
Specifically, the anti-candida albicans refers to inhibiting the adhesion, hyphal morphology transformation, pathogenicity and/or virulence effect of candida albicans on cells.
An anti-candida albicans drug or a drug for preventing and treating infectious diseases caused by candida albicans, which contains the diaryl chalcogenide compound, also belongs to the protection scope of the invention.
The invention has the following beneficial effects:
the invention provides a novel diaryl sulfur group compound which has good inhibition effect on the adhesion, hypha form conversion and pathogenicity of candida albicans. Moreover, the compound has low toxicity, does not influence the growth of human cells, and has good application prospect in the development of novel antifungal medicines, especially in the development of medicines for resisting Candida albicans infection.
The study of the invention also finds that the diaryl sulfur group compound only has certain inhibition effect on the growth of the candida albicans, which indicates that the effect of the compound on the candida albicans strain is not mainly based on killing bacteria, but is achieved by inhibiting the adhesion, hypha form conversion and pathogenicity of the bacteria, so that the drug resistance is not easy to generate.
Drawings
FIG. 1 is a diagram of the synthesis of a diaryl chalcogenide.
FIG. 2 is a nuclear magnetic hydrogen spectrum of Compound No. 3.
Fig. 3 is a carbon spectrum of compound No. 3.
Fig. 4 is a nuclear magnetic hydrogen spectrum of compound No. 8.
Fig. 5 is a carbon spectrum of compound No. 8.
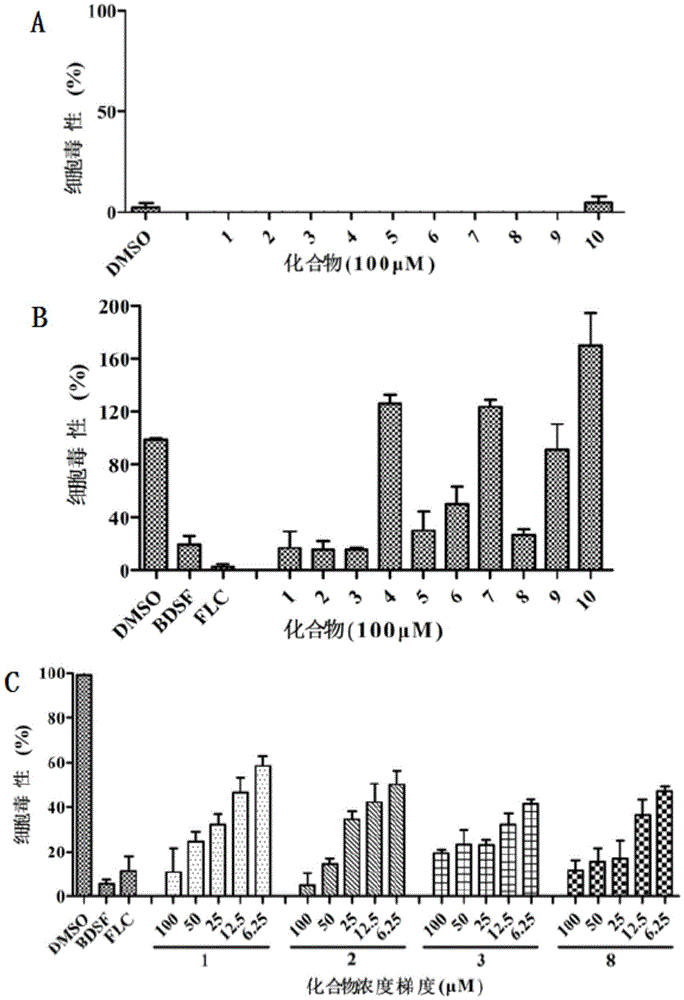
FIG. 6 is a graph of the effect of diarylchalcogenides on adhesion to Candida albicans; wherein, the graph (A) is a graph showing the effect of 13 compounds at a final concentration of 100. mu.M on the adhesion of Candida albicans cells to polystyrene; FIG. (B) is a graph showing the results of the inhibition rates of 4 compounds Nos. 1, 2, 3 and 8 at different concentrations of 6.25. mu.M to 100. mu.M; DMSO as control; data are shown as the average of 8 biological replicates, with error bars reflecting standard deviations.
FIG. 7 is a graph showing the effect of diarylchalcogenides on Candida albicans hyphae formation; wherein, the graph (A) is a graph showing the results of measurement of the inhibitory rate of 13 synthetic diarylchalcogenides at a final concentration of 100. mu.M on the formation of Candida albicans hyphae; FIG. (B) is a graph showing the inhibitory effect of Compound No. 1, 2, 3 or 8 at various concentrations ranging from 6.25. mu.M to 100. mu.M; FIG. (C) is a photograph showing the microscopic observation of the inhibition of the generation of hyphae by DMSO, fluconazole, compound No. 1, 2, 3, 8 at a final concentration of 100. mu.M; this data shows the average of 3 biological experiments, with error bars reflecting the standard deviation.
FIG. 8 is a graph showing the effect of diarylchalcogenides on the growth rate of Candida albicans; DMSO as control; data shown are the average of 3 biological replicates, and error bars reflect standard deviations.
FIG. 9 is the effect of diarylchalcogenides on the pathogenicity of A549 cells by Candida albicans; (A) cytotoxicity of 13 diarylchalcogenides against a549 cells at a final concentration of 100 μ M; (B) the effect of 13 diarylchalcogenides at a final concentration of 100 μ M on cells infected with Candida albicans; (C) effect of compounds No. 1, 2, 3, 8 on Candida albicans infection of cells at various concentrations ranging from 6.25. mu.M to 100. mu.M. The release amount of LDH is detected to detect the toxicity of cells, and when the cytotoxicity of Candida albicans is detected, the release amount of LDH added into a DMSO group is taken as 100%, so that the release ratio of LDH added into other diaryl chalcogenide groups is regulated. Data shown are the average of 4 biological replicates, and error bars reflect standard deviations.
Detailed Description
The invention is further described with reference to the drawings and the following detailed description, which are not intended to limit the invention in any way. Reagents, methods and apparatus used in the present invention are conventional in the art unless otherwise indicated.
Unless otherwise indicated, reagents and materials used in the following examples are commercially available.
EXAMPLE 1 Synthesis of diarylchalcogenides
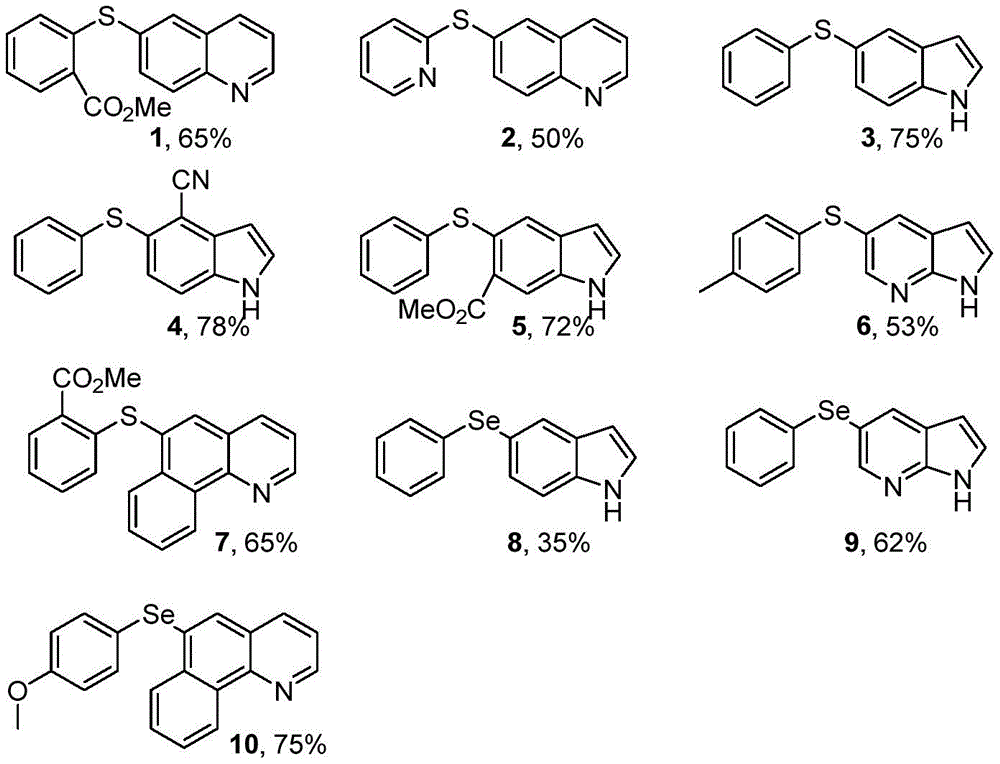
1. The synthesis scheme of the diaryl chalcogenide is shown in figure 1, and the specific synthesis method comprises the following steps:
(1) adding 1mmol of hydrogenated nitrogen-containing heterocyclic compound (compound A-1-A-6) and 1mmol of diaryl sulfur/selenium ether compound (compound B-1-B-6) into a reactor, adding 2mL of dimethyl sulfoxide serving as a solvent and 1.5mmol of iodine elementary substance serving as an accelerator, and stirring for reacting for 4-20 hours at 100 ℃;
wherein, the compounds A-1 to A-6 are respectively: tetrahydroquinoline, indoline, 4-cyanoindoline, methyl 6-indolinoate, 7-azaindoline, 7, 8-benzotetrahydroquinoline (structure formula shown in figure 1);
the compounds B-1 to B-6 are respectively: methyl 2, 2 '-diphenyl sulfide, 2' -dipyridyl sulfide, diphenyl sulfide, 4 '-dimethyl diphenyl sulfide, diphenyl selenide, 4' -dimethoxy diphenyl selenide (structural formula shown in figure 1).
(2) And cooling to room temperature after the reaction is finished, diluting the reaction solution with ethyl acetate, washing for three times, drying, evaporating under reduced pressure to remove the solvent to obtain a crude product, and finally purifying by column chromatography to obtain the diaryl sulfur group compound No. 1-10.
2. Wherein, the nuclear magnetic hydrogen spectrum and the carbon spectrum of the compound 3 and the compound 8 are respectively shown in figures 2-4.
EXAMPLE 2 examination of the Effect of diaryl chalcogenides
1. The test method comprises the following steps:
(1) activation of candida albicans strains: candida albicans standard strain SC5314 was activated in LB medium (tryptone 10g/L, yeast extract 5g/L, NaCl 10g/L, agar 15g/L), and cultured overnight in an incubator at 30 ℃.
(2) Effect of compounds on adhesion of candida albicans strain SC 5314:
SC5314 strain on LB solid plate was picked, inoculated into YNB medium (6.7g/L, 0.2% glucose was added before use), cultured overnight at 30 ℃ under shaking at 200rpm, and the OD of the bacterial solution was measured600. Then diluted the bacterial liquid to OD with GMM600(0.5), 1mL of the suspension was put into a 1.5mL EP tube, and the final concentration of 100. mu.M compound was added in order, followed by shakingMix well and add 200 μ L each to a 96 well plate, set to 4 replicates per treatment, and set to treatment with DMSO alone. Incubating 96-well plate at 37 deg.C, discarding bacteria solution after 4 hr, adding 50 μ L of 0.5% crystal violet, and allowing to act at room temperature for 45 min. Discarding crystal violet and using ice ddH2O washing 10 times, adding 200 μ L75% ethanol, standing at room temperature for 30 min, and measuring OD590Data were processed using GraphPad Prism 6 software.
(3) Effect of compounds on hyphae of candida albicans strain SC 5314:
selecting SC5314 strain on LB solid plate, inoculating into GMM culture solution, shaking at 30 deg.C and 200rpm for overnight culture, and determining OD of bacterial solution600Diluting the bacterial liquid to OD with GMM6000.1. 500 μ L of the bacterial solution was taken into a 1.5mL EP tube, and a compound with a final concentration of 100 μ M was added, and DMSO and BDSF (b.cenocepacia differential signal factor, which has a good inhibitory effect on the formation of SC5314 hyphae) were set as positive and negative controls, respectively. Shaking and mixing, placing in a water bath kettle at 37 ℃ for incubation, centrifuging at 5000rpm for 10min after 6h, discarding the supernatant, adding 40 mu L of GMM culture solution to resuspend the thalli, observing the formation of hyphae under a Zeiss Axioplan 2 microscope, and taking pictures with different visual fields.
(4) Determination of the effect of the compounds on the growth of candida albicans strain SC 5314:
selecting single colony of strain SC5314, inoculating to GMM culture solution, performing shake culture at 30 deg.C and 200rpm overnight, and determining OD of bacterial solution600Diluting the bacterial liquid to OD with GMM6000.05. And (3) putting 1mL of the bacterial liquid into a 1.5mL EP tube, sequentially adding a compound with the final concentration of 100 mu M, shaking and uniformly mixing, adding 300 mu L of the compound into a 100-pore plate, setting 4 times of each treatment, and setting the treatment of only adding DMSO and adding fluconazole at the same time. Placing in a growth curve tester, measuring OD every 2h at 30 deg.C and 200rpm600Values, observed after 2d experimental results, GraphPad Prism 6 processed data.
(5) Effect of compounds on the cytotoxicity of candida albicans strain SC 5314:
(a) recovery and culture of A549 cells: the freeze-thawed A549 cells were transferred to DMEM medium (Gioco Corp.) containing 10% FBS, 37 ℃,5%CO2cultured overnight under the conditions.
(b) Preparation of a549 cells: a549 cells in DMEM (high glucose Medium) containing 10% fetal bovine serum at 1.5X 104Cell concentration per well was cultured overnight in 96-well plates. When the cells were 80% full of the bottom of the 96-well plate, the culture medium was discarded, and the cells were washed 3 times with 1 × PBS.
(c) Preparation of candida albicans: selecting fresh SC5314, inoculating into GMM culture solution, and shake culturing at 30 deg.C and 200rpm overnight; adjustment to OD with DMEM cell maintenance solution containing 1% FBS6001.0, 10-fold dilution with DMEM (1% FBS) (10 ≈ 10)8cfu/mL) for use.
(d) And (3) determining the cytotoxicity: adding compound with final concentration of 100 μ M into cell maintenance solution containing bacteria, adding 100 μ L into prepared A549 cells, standing at 37 deg.C and 5% CO2Culturing for 8h in a cell culture box, repeating every treatment for 4 times, simultaneously setting DMSO only, BDSF and fluconazole as controls, and simultaneously determining the toxicity of the compound on the cells, namely adding the compound with the final concentration of 100 mu M into a cell maintenance solution without bacteria, and treating as above. Reference is made to Promega corporation CytoToxCellular LDH activity was determined by the NonRadioactive cytoxicity Assay protocol followed by GraphPad Prism 6 treatment of the data.
(6) Effect of compounds on infection of candida albicans strain SC5314 in mice:
(a) breeding the mice: male BALB/c mice of 6-8 weeks old, purchased from Experimental animals center, Guangdong province, were housed in Experimental animals center, university of south China agricultural. 8/group were randomly assigned, weight recorded and labeled for each mouse.
(b) Preparation of candida albicans: selecting fresh SC5314, inoculating to GMM culture solution, shake culturing at 30 deg.C and 200rpm overnight, centrifuging at 5000rpm for 5min to collect thallus, washing thallus with 1 × PBS for 3 times, and culturing at 5 × 108The concentration of cfu/mL was dispersed in PBS.
(c) Mouse tail vein injection: adding compound No. 3 and compound No. 8 with final concentration of 100 μ M into PBS containing bacteria, and mixing well for use. The test was conducted according to the care and use regulations of experimental animals under the health guidelines of the national institutes of health (NIH 8023 publication, revised 1978). Mice were weighed, injected tail vein with 100 μ L/10g, and injected with 1 XPBS, 100 μ M fluconazole as a control. Mice were observed continuously for survival, recorded, and data were counted after 20d and processed with GraphPad Prism 6.
2. Results of the experiment
(1) Compounds inhibit adhesion of Candida albicans strain SC5314
As shown in a diagram in fig. 6, the results of the reference on the adhesion of candida albicans in the DMSO group showed that the decrease in cell adhesion after treatment with compound nos. 3 and 8 was around 35% among 10 compounds, which significantly inhibited the adhesion of candida albicans SC 5314.
In combination with the biological activity of the compounds in terms of hyphal formation rate and cytotoxicity, we selected compounds No. 1, No. 2, No. 3 and No. 8 for concentration gradient testing. The results show (panel B in fig. 6) that compounds No. 3 and No. 8 exhibited concentration-gradient-dependent inhibition of adhesion, with the decrease in compound concentration, the inhibition of adhesion to candida albicans decreased.
(2) Compounds inhibit the formation of hyphae of Candida albicans strain SC5314
By comparing the hypha formation rates of DMSO and BDSF, as shown in A of FIG. 7, 6 of 10 compounds have significant inhibitory effect on Candida albicans SC5314, and the hypha formation rate is reduced to below 35%. The results of hyphal formation observed under a microscope are shown in panel C of FIG. 7.
Similarly, in combination with other activities, we tested the rate of hyphal formation of C.albicans strain SC5314 for compounds No. 1, 2, 3 and 8 at a concentration gradient of 6.25. mu.M to 100. mu.M. The results (B in FIG. 7) show that the four compounds all exhibit concentration-dependent inhibitory activity against Candida albicans hyphae, and that the hypha formation rate of Compound No. 8 was reduced to about 40% at 6.25. mu.M.
(3) The compound has certain effect on the growth of Candida albicans strain SC5314
The results are shown in A in figure 8, and 100 μ M of compounds No. 1, 2, 3, and 8 have certain inhibitory effect on the growth of Candida albicans strain SC5314 with DMSO and fluconazole as controls. At the same time, we tested the effect of different concentration gradients of compound nos. 3 and 8 on candida albicans. The results show (panels B and C in fig. 8) that compound No. 3 had a small effect, compound No. 8 had a relatively large effect, and had some inhibitory effect on the growth of SC 5314.
(4) The compound can inhibit the virulence of cells caused by Candida albicans strain SC5314
The results of the cytotoxicity experiments (see a in fig. 9) showed that 10 compounds were not toxic to cells in the absence of bacteria in the DMSO control.
Under the condition of adding candida albicans SC5314, the cytotoxicity of DMSO and BDSF treated cells is taken as a control, and B and C in figure 9 show that the compounds No. 1, 2, 3 and 8 have protective effect in inhibiting the infection of the strain SC5314 to cells, and can reduce the toxicity of the candida albicans to below 30%.
(5) The compound has protective effect on mice infected by Candida albicans strain SC5314
By combining the performance results of all aspects of diaryl chalcogenide, No. 1, No. 2, No. 3 and No. 8 which are better in all aspects are selected for carrying out mouse experiments, and the experiments are continuously observed for 20 d.
The results show that compound nos. 1 and 2 have no protective effect in vivo experiments, whereas the survival rate of mice treated with compound nos. 3 and 8 is 100%, as shown in table 1.
TABLE 1 Effect of Compounds on Candida albicans pathogenicity in mouse infection model
| Compound (I) | Survival rate (%) |
| |
0 |
| |
100 |
| No. 3 | 100 |
| |
100 |
From the results of the experiments, it can be seen that compounds 3 and 8 are novel candidates for anti-candida albicans drugs. In general, the present invention synthesizes a series of diaryl chalcogenides and screens them for their ability to combat fungal pathogens. Some of them showed excellent properties of inhibiting cell adhesion, hyphal formation and pathogenicity of candida albicans; although they are not toxic to Candida albicans cells themselves, they are also not toxic to human cells. The experimental results show that some of the compounds can be developed into novel medicaments for resisting the candida albicans infection.
The above embodiments are preferred embodiments of the present invention, but the present invention is not limited to the above embodiments, and any other changes, modifications, substitutions, combinations, and simplifications which do not depart from the spirit and principle of the present invention should be construed as equivalents thereof, and all such changes, modifications, substitutions, combinations, and simplifications are intended to be included in the scope of the present invention.
Claims (8)
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2017/105400 WO2019071395A1 (en) | 2017-10-09 | 2017-10-09 | Anti-candida albicans diaryl chalcogenide and preparation and use thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN109843289A CN109843289A (en) | 2019-06-04 |
| CN109843289B true CN109843289B (en) | 2021-10-29 |
Family
ID=66100204
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201780025371.7A Active CN109843289B (en) | 2017-10-09 | 2017-10-09 | A kind of diaryl chalcogenide compound against Candida albicans and its preparation and application |
Country Status (2)
| Country | Link |
|---|---|
| CN (1) | CN109843289B (en) |
| WO (1) | WO2019071395A1 (en) |
Families Citing this family (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN110003080B (en) * | 2019-04-30 | 2022-05-06 | 成都大学 | A kind of selenium-containing compound and its preparation method and use |
| CN112494483B (en) * | 2020-11-13 | 2022-09-02 | 兰州大学 | Application of dithiolethiones compound in antibacterial aspect |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101068777A (en) * | 2004-10-08 | 2007-11-07 | 万有制药株式会社 | The preparation method of sulfide compound |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1515716A2 (en) * | 2002-04-03 | 2005-03-23 | Astrazeneca AB | Indole derivatives having anti-angiogenetic activity |
-
2017
- 2017-10-09 CN CN201780025371.7A patent/CN109843289B/en active Active
- 2017-10-09 WO PCT/CN2017/105400 patent/WO2019071395A1/en not_active Ceased
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101068777A (en) * | 2004-10-08 | 2007-11-07 | 万有制药株式会社 | The preparation method of sulfide compound |
Non-Patent Citations (6)
| Title |
|---|
| A general palladium-catalyzed coupling of aryl bromides/triflates and thiols;Takahiro Itoh等;《ORGANIC LETTERS》;20041028;第6卷(第24期);4587-4590 * |
| Cobalt-catalyzed aryl-sulfur bond formation;Ying-Chieh Wong等;《Org. Lett.》;20061103;第8卷(第24期);5613-5616 * |
| Preparation and antifungal action of a series of 6-quinolyl sulfides and sulfones;Gialdi, F.等;《Farmaco, Edizione Scientifica》;19591231;第14卷;288-303 * |
| Selective inhibitors of Candida albicans dihydrofolate reductase: activity and selectivity of 5-(arylthio)-2,4-diaminoquinazolines;Joseph H. Chan等;《Journal of Medicinal Chemistry》;19951231;第38卷(第18期);3608-3616 * |
| Site-Specific Oxidative C-H Chalcogenation of (Hetero)Aryl-Fused Cyclic Amines Enabled by Nanocobalt Oxides;Zhenda Tan等;《Org. Lett.》;20181004;第20卷(第20期);6554-6558 * |
| Takahiro Itoh等.A general palladium-catalyzed coupling of aryl bromides/triflates and thiols.《ORGANIC LETTERS》.2004,第6卷(第24期),4587-4590. * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN109843289A (en) | 2019-06-04 |
| WO2019071395A1 (en) | 2019-04-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2021109549A1 (en) | Joint application of quercetin and antibacterial medicament | |
| CN109843289B (en) | A kind of diaryl chalcogenide compound against Candida albicans and its preparation and application | |
| CN118436666A (en) | Huang Tongxi complexes and their use | |
| CN113648303A (en) | Application of cafestol or derivative thereof in preparation of anti-candida albicans medicine or anti-candida albicans daily product | |
| CN104606175A (en) | Application of plumbagin for inhibiting growth of staphylococcus aureus biofilm | |
| CN108084089B (en) | A kind of 2-alkylamine quinoline compound for preventing and treating Candida albicans and its preparation method and application | |
| CN109384823B (en) | Two tetromycin glucosides and their application in anti-renal cancer drugs | |
| CN109985027B (en) | Application of myricetin in preparation of gram-negative bacterium virulence resisting medicine | |
| WO2019019049A1 (en) | Anti-candida albicans piperazine derivative, preparation method therefor and application thereof | |
| CN108785291A (en) | Application of the hypocrellin in the product for preparing anti-candida albicans | |
| CN113730384A (en) | Application of 4-methoxy phenanthrene-2, 5-diol in preparation of anti-candida albicans medicine or anti-candida albicans daily product | |
| CN111909021A (en) | Sorbicillinoids compound and preparation method and application thereof | |
| CN108707090B (en) | A kind of chlorine-containing aromatic compound and its preparation method and use | |
| CN111499649A (en) | Benzodifuranone compound with anti-tumor activity, preparation method and application thereof | |
| CN101803531A (en) | China cryptoporus sinensis and solid cultural method and application thereof | |
| CN108498784A (en) | Application of the Radix pseudostellariae cyclic peptides B in preparing anti-candida albicans drug | |
| CN103585277A (en) | Application of clove leaf extractives in inhibition of streptococcus suis and intervention and elimination of streptococcus suis biofilm | |
| CN109803656B (en) | A kind of compound against Candida albicans and its preparation method and application | |
| CN115006383B (en) | Use of a diphenyl ether compound in the preparation of anti-coronavirus drugs | |
| Raghavendra et al. | Analgesic, Antibacterial and Antiviral Activities of 2-(5-Alkyl-1, 3, 4-oxadiazol-2-yl)-3H-benzo [f] chromen-3-ones | |
| CN115634227B (en) | Use of N-hydroxypyridone compounds in the preparation of anti-coronavirus drugs | |
| CN113786399A (en) | Application of glabridin in preparation of salmonella III type secretion system inhibitor | |
| CN110786341B (en) | Preparation method and application of lotus plumule extracting solution | |
| CN101525578A (en) | Preparation of peltate yam endophytic fungi spiro-dinaphthyl compound and application as germicide thereof | |
| CN112603916A (en) | Application of lauric acid in preparation of salmonella III type secretion system inhibitor |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |