CN100384913C - Amino-functional polysiloxane wax - Google Patents
Amino-functional polysiloxane wax Download PDFInfo
- Publication number
- CN100384913C CN100384913C CNB2004800291745A CN200480029174A CN100384913C CN 100384913 C CN100384913 C CN 100384913C CN B2004800291745 A CNB2004800291745 A CN B2004800291745A CN 200480029174 A CN200480029174 A CN 200480029174A CN 100384913 C CN100384913 C CN 100384913C
- Authority
- CN
- China
- Prior art keywords
- general formula
- sub
- polysiloxane
- wax
- waxes
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/04—Polysiloxanes
- C08L83/08—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/38—Polysiloxanes modified by chemical after-treatment
- C08G77/382—Polysiloxanes modified by chemical after-treatment containing atoms other than carbon, hydrogen, oxygen or silicon
- C08G77/388—Polysiloxanes modified by chemical after-treatment containing atoms other than carbon, hydrogen, oxygen or silicon containing nitrogen
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L83/00—Compositions of macromolecular compounds obtained by reactions forming in the main chain of the macromolecule a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon only; Compositions of derivatives of such polymers
- C08L83/04—Polysiloxanes
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M15/00—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment
- D06M15/19—Treating fibres, threads, yarns, fabrics, or fibrous goods made from such materials, with macromolecular compounds; Such treatment combined with mechanical treatment with synthetic macromolecular compounds
- D06M15/37—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds
- D06M15/643—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain
- D06M15/6436—Macromolecular compounds obtained otherwise than by reactions only involving carbon-to-carbon unsaturated bonds containing silicon in the main chain containing amino groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/045—Polysiloxanes containing less than 25 silicon atoms
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/14—Polysiloxanes containing silicon bound to oxygen-containing groups
- C08G77/18—Polysiloxanes containing silicon bound to oxygen-containing groups to alkoxy or aryloxy groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/24—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen halogen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/26—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen nitrogen-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/04—Polysiloxanes
- C08G77/22—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen
- C08G77/28—Polysiloxanes containing silicon bound to organic groups containing atoms other than carbon, hydrogen and oxygen sulfur-containing groups
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G77/00—Macromolecular compounds obtained by reactions forming a linkage containing silicon with or without sulfur, nitrogen, oxygen or carbon in the main chain of the macromolecule
- C08G77/42—Block-or graft-polymers containing polysiloxane sequences
- C08G77/452—Block-or graft-polymers containing polysiloxane sequences containing nitrogen-containing sequences
- C08G77/455—Block-or graft-polymers containing polysiloxane sequences containing nitrogen-containing sequences containing polyamide, polyesteramide or polyimide sequences
-
- D—TEXTILES; PAPER
- D06—TREATMENT OF TEXTILES OR THE LIKE; LAUNDERING; FLEXIBLE MATERIALS NOT OTHERWISE PROVIDED FOR
- D06M—TREATMENT, NOT PROVIDED FOR ELSEWHERE IN CLASS D06, OF FIBRES, THREADS, YARNS, FABRICS, FEATHERS OR FIBROUS GOODS MADE FROM SUCH MATERIALS
- D06M2200/00—Functionality of the treatment composition and/or properties imparted to the textile material
- D06M2200/50—Modified hand or grip properties; Softening compositions
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Textile Engineering (AREA)
- Treatments For Attaching Organic Compounds To Fibrous Goods (AREA)
- Silicon Polymers (AREA)
- Compositions Of Macromolecular Compounds (AREA)
Abstract
Description
本发明涉及具有多个叔或者季氨基基团的聚硅氧烷蜡,其制备方法以及其作为软化剂在纺织工业中的应用。The present invention relates to a polysiloxane wax with multiple tertiary or quaternary amino groups, its preparation method and its use as a softener in the textile industry.
有大量的关于氨基官能硅氧烷化合物的文献。这些化合物例如被用作纺织工业中的增塑剂、用作表面处理剂、用作增稠剂或者被用于化妆品工业。尽管如此,纺织工业中仍然对能够得到具有优越的手感的产品并且不具有通用的不稳定乳液的缺点的软化性物质存在需求。There is an extensive literature on aminofunctional silicone compounds. These compounds are used, for example, as plasticizers in the textile industry, as surface treatment agents, as thickeners or in the cosmetics industry. Nevertheless, there is still a need in the textile industry for emollient substances which allow to obtain products with a superior hand without the disadvantages of conventional unstable emulsions.
现在已经发现,某些具有多个叔或者季氮原子的聚硅氧烷蜡可以被加工成稳定的含水分散体,并且当被用作纺织工业中的软化剂时具有令人惊奇地优良的性能,并且得到具有令人愉快的、柔软手感的产品。It has now been found that certain polysiloxane waxes with multiple tertiary or quaternary nitrogen atoms can be processed into stable aqueous dispersions and have surprisingly good properties when used as softeners in the textile industry , and a product with a pleasant, soft hand is obtained.
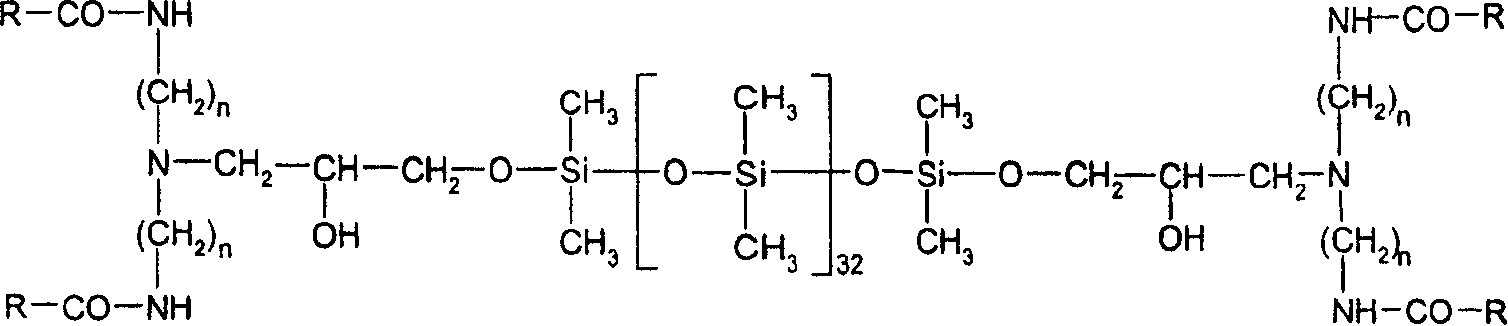
因此,本发明提供了具有通式(I)到(IV)的氨基官能的聚硅氧烷蜡Accordingly, the present invention provides amino-functional polysiloxane waxes having the general formulas (I) to (IV)
其中in
R是直链或者支链的C11-C22-烷基,R is straight-chain or branched C 11 -C 22 -alkyl,
R1是C1-C7-烷基或者苄基,优选甲基或者苄基,R1 is C 1 -C 7 -alkyl or benzyl, preferably methyl or benzyl,
R2是-OH,-CH3,-OCH3,-OC2H5,R2 is -OH , -CH3 , -OCH3 , -OC2H5 ,
A-是CH3OSO3 -,氯根,溴根,碘根,或者甲苯磺酸根,优选CH3OSO3 -或者氯根,A - is CH 3 OSO 3 - , chloride, bromide, iodide, or tosylate, preferably CH 3 OSO 3 - or chloride,
n是2或者3,n is 2 or 3,
p是10-200,优选20-50,p is 10-200, preferably 20-50,
q+z是10-400,优选15-200,并且q+z is 10-400, preferably 15-200, and
q/z是5-50,优选10-30。q/z is 5-50, preferably 10-30.
这些蜡,无论是季铵化的形式(II)和(IV),或者非季铵化的形式(I)和(III),当用作软化剂时均显示出良好的性能。其重要的优点在于,它们可以分散体的形式使用,该分散体与乳液相比具有较高的剪切力稳定性。These waxes, whether in the quaternized forms (II) and (IV) or in the non-quaternized forms (I) and (III), show good performance when used as softeners. An important advantage of this is that they can be used in the form of dispersions which, compared to emulsions, have a higher shear stability.
本发明进一步提供了上述聚硅氧烷蜡的制备方法。初始步骤是通过脂肪酸例如硬脂酸或者月桂酸与二亚乙基三胺或者二亚丙基二胺的缩合制备脂肪酸二酰胺。The present invention further provides a preparation method of the above polysiloxane wax. The initial step is the preparation of fatty acid diamides by condensation of fatty acids such as stearic acid or lauric acid with diethylenetriamine or dipropylenediamine.
使得到的脂肪酸二酰胺与具有通式(V)的聚硅氧烷油反应Reaction of the resulting fatty acid diamide with a polysiloxane oil of general formula (V)
其中p具有与通式(I)或者(II)中的含义相同的含义,以制备通式(I)或者(II)的蜡,wherein p has the same meaning as in general formula (I) or (II), to prepare the wax of general formula (I) or (II),
或者随后用通常的季铵化剂季铵化,所述季铵化剂例如是硫酸甲酯、硫酸乙酯、卤代甲烷、氯化苄或者甲苯磺酸酯,以制备通式(II)的蜡。起始材料和其制备方法是本领域技术人员已知的。Or subsequently quaternized with common quaternizing agents such as methyl sulfate, ethyl sulfate, methyl halide, benzyl chloride or tosylate, to prepare the compound of general formula (II) wax. Starting materials and methods for their preparation are known to those skilled in the art.
为了制备通式(III)或者(IV)的蜡,将得到的脂肪酸二酰胺与通式(VI)的聚硅氧烷油反应To prepare waxes of general formula (III) or (IV), the fatty acid diamides obtained are reacted with polysiloxane oils of general formula (VI)
其中R2、(q+z)和q/z具有与通式(III)或者(IV)中的含义相同的含义,wherein R2, (q+z) and q/z have the same meaning as in general formula (III) or (IV),
并且,为了制备通式(IV)的蜡,将得到的脂肪酸二酰胺随后用通常的、上述的季铵化剂季铵化。Furthermore, in order to prepare the waxes of the general formula (IV), the fatty acid diamides obtained are subsequently quaternized with the customary, abovementioned quaternizing agents.
本发明进一步提供了上述的聚硅氧烷蜡作为软化剂在纺织工业中的用途。获得的聚硅氧烷蜡可以借助于分散剂转化成含水分散体,该含水分散体与乳液相比具有较高的剪切力稳定性,并且对于软化纺织品是非常有用的,能够赋予材料以令人愉快的柔软手感。The present invention further provides the use of the above polysiloxane wax as a softener in the textile industry. The polysiloxane waxes obtained can be converted with the aid of dispersants into aqueous dispersions which have a higher shear stability compared to emulsions and are very useful for softening textiles, giving the material a Pleasant soft touch.
实施例Example
1.脂肪酸二酰胺的制备1. Preparation of Fatty Acid Diamides
(脂肪酸:RCOOH)(fatty acid: RCOOH)
1.1R-CONH(CH2)2NH(CH2)2NHCO-R的制备1.1 Preparation of R-CONH(CH 2 ) 2 NH(CH 2 ) 2 NHCO-R
在氮气下将2摩尔脂肪酸加热到105℃;然后滴加1摩尔二亚乙基三胺,同时将温度提高到130℃。然后,使用蒸馏桥,在缓慢的氮气流下,在130℃下缩合24小时。随后排出反应混合物。反应混合物的酸值(中和1克产品需要的KOH的毫克数)低于7。得到的缩合产物的量需要少于1.1摩尔的高氯酸用以滴定自由氨基。2 moles of fatty acid were heated to 105°C under nitrogen; then 1 mole of diethylenetriamine was added dropwise while raising the temperature to 130°C. Then, condensation was carried out at 130° C. for 24 hours under a slow nitrogen flow using a distillation bridge. The reaction mixture was subsequently discharged. The acid number (mg of KOH required to neutralize 1 gram of product) of the reaction mixture was below 7. The amount of condensation product obtained required less than 1.1 moles of perchloric acid for titration of free amino groups.
1.2R-CONH(CH2)3NH(CH2)3NHCO-R的制备1.2 Preparation of R-CONH(CH 2 ) 3 NH(CH 2 ) 3 NHCO-R
使用二亚丙基二胺代替二亚乙基二胺重复1.1。Repeat 1.1 using dipropylenediamine instead of diethylenediamine.
2.聚硅氧烷蜡的制备2. Preparation of Polysiloxane Wax
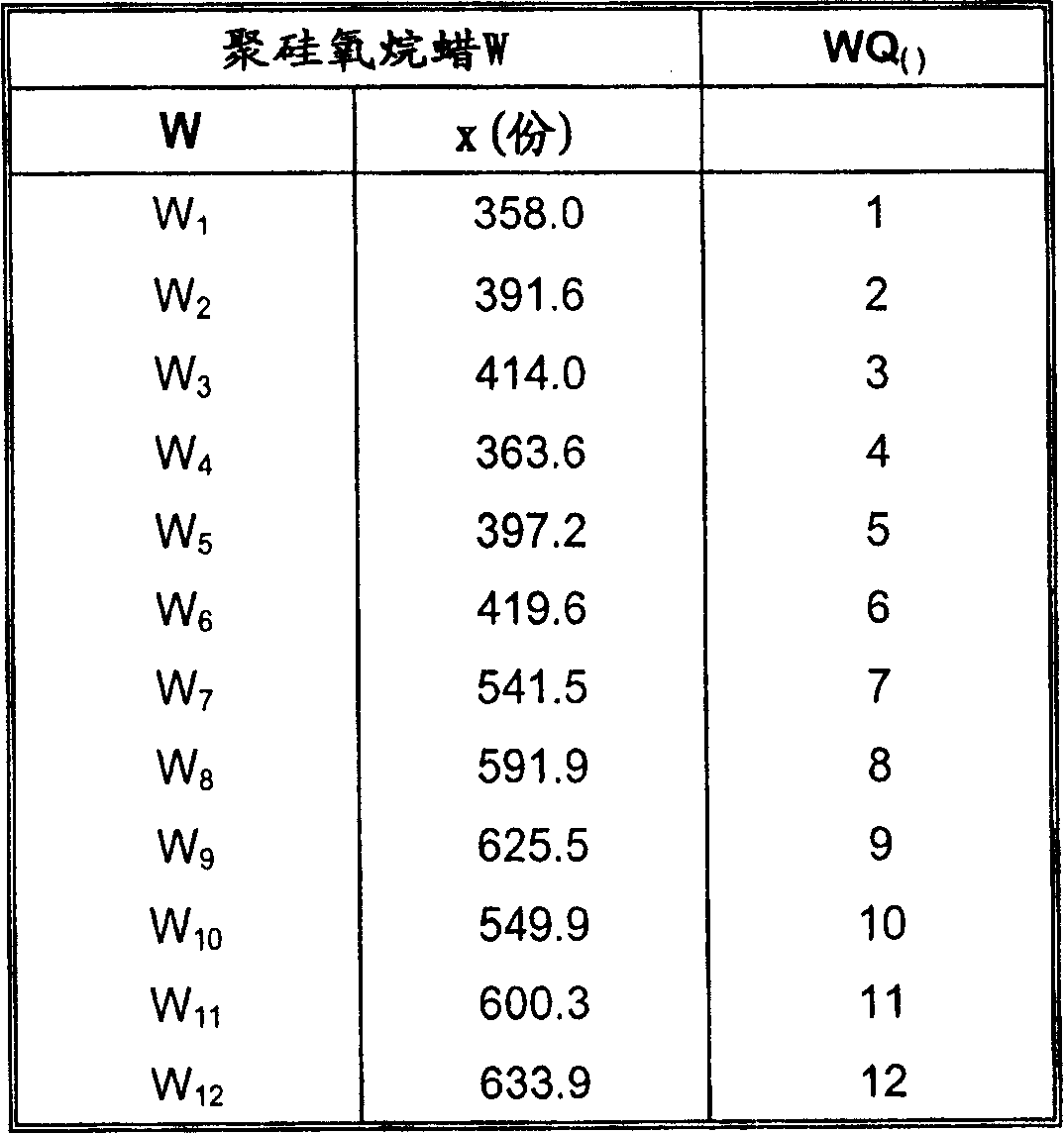
2.1聚硅氧烷蜡W1-W62.1 Silicone wax W1-W6
使661.5份的以下通式的硅油Make 661.5 parts of silicone oil of the following general formula
在150℃下,在氮气下,与x份的二酰胺D1到D6反应14小时。随后进行检验,看是否缩水甘油基不再存在。获得了以下蜡:Reaction with x parts of diamides D 1 to D 6 at 150° C. under nitrogen for 14 hours. A test is then carried out to see if glycidyl groups are no longer present. The following waxes were obtained:
所述蜡具有以下结构:The wax has the following structure:
n=2或者3,R=C11-C22烷基n=2 or 3, R=C 11 -C 22 alkyl
2.2聚硅氧烷蜡W7-W12 2.2 Silicone wax W 7 -W 12
使661.5份的以下通式的硅油Make 661.5 parts of silicone oil of the following general formula
在150℃下,在氮气下,与x份的二酰胺D1到D6反应14小时。随后进行检验,看是否缩水甘油基已经定量地反应。获得了以下蜡:Reaction with x parts of diamides D 1 to D 6 at 150° C. under nitrogen for 14 hours. A test was then carried out to see if the glycidyl groups had reacted quantitatively. The following waxes were obtained:
所述蜡具有以下结构:The wax has the following structure:
n=2或者3,R=C11-C22烷基n=2 or 3, R=C 11 -C 22 alkyl
3.季铵化的聚硅氧烷蜡WQ1-WQ12的制备3. Preparation of quaternized polysiloxane waxes WQ 1 -WQ 12
将x份的聚硅氧烷蜡W熔融,并且允许与25.2份的硫酸二甲酯在大约70-75℃下反应2小时。x parts of silicone wax W were melted and allowed to react with 25.2 parts of dimethyl sulfate at about 70-75°C for 2 hours.
获得具有以下结构的蜡:A wax with the following structure is obtained:
和and
n=2或者3,R=C11-C21烷基。n=2 or 3, R=C 11 -C 21 alkyl.
4.最终产品的制备4. Preparation of the final product
4.1基于聚硅氧烷蜡W1-W12 4.1 Silicone-based waxes W 1 -W 12
将125.0份聚硅氧烷蜡(W)熔融。然后加入以下化合物(分散剂1)Melt 125.0 parts of silicone wax (W). Then add the following compound (Dispersant 1)
在丙二醇中的70%溶液25.0份以及硬脂基聚-10-乙二醇醚(分散剂2)37.5份。一旦得到均匀的熔体,在搅拌下增量地加入800.0份的水。最后加入12.5份的冰醋酸。这样得到1000.0份的12.5%聚硅氧烷蜡分散体(W-E)。70% solution in propylene glycol 25.0 parts and stearyl poly-10-glycol ether (dispersant 2) 37.5 parts. Once a homogeneous melt is obtained, 800.0 parts of water are added in increments with stirring. Finally 12.5 parts of glacial acetic acid are added. This gives 1000.0 parts of a 12.5% silicone wax dispersion (W-E).
4.2基于季铵聚硅氧烷蜡WQ1-WQ12 4.2 Based on quaternary ammonium polysiloxane waxes WQ 1 -WQ 12
重复4.1,除了使用聚硅氧烷蜡(WQ)代替聚硅氧烷蜡(W),以及最后加入12.5份的水,代替12.5份的冰醋酸。这样得到1000.0份的12.5%聚硅氧烷蜡分散体(WQ-E)。4.1 was repeated except that silicone wax (WQ) was used instead of silicone wax (W), and finally 12.5 parts of water were added instead of 12.5 parts of glacial acetic acid. This gives 1000.0 parts of a 12.5% silicone wax dispersion (WQ-E).
产品W1-E到W12-E以及WQ1-E到WQ12-E对于剪切力是很稳定的;它们可以在浸轧工艺中和在排空(exhaust)工艺中被施加到纺织材料(棉花和合成纤维)两者中。最终的纺织材料具有优良的、柔软的、令人愉快的平滑手感。Products W 1 -E to W 12 -E and WQ 1 -E to WQ 12 -E are very stable against shear forces; they can be applied to textile materials in the padding process and in the exhaust process (cotton and synthetic fibers) of both. The final textile material has an excellent, soft, pleasantly smooth hand.
应用实施例Application example
进行了以下精整操作:The following finishing operations were performed:
排空工艺Empty process
在大约40℃和6∶1到20∶1的液比下,在实验室喷射器中,将精整底物加入含水溶液中,基于所述底物该溶液包含0.5%到4.0%的在实施例中描述的最终产品。在pH5.0到6.0(40℃)和对底物进行连续搅动下在20分钟之后,将底物从所述溶液中除去,在140℃下无张力地离心和干燥70-90秒。At about 40°C and a liquid ratio of 6:1 to 20:1, in a laboratory injector, the finishing substrate is added to an aqueous solution containing 0.5% to 4.0% of The final product described in the example. After 20 minutes at pH 5.0 to 6.0 (40°C) with continuous agitation of the substrate, the substrate was removed from the solution, centrifuged and dried without tension at 140°C for 70-90 seconds.
浸轧工艺padding process
在室温下,使用包含15到60g/l的最终产品W-E或者WQ-E的含水溶液,将精整底物浸轧,达到100%干重增量。随后将浸轧的材料在140℃下干燥70-90秒。The finishing substrate is padded to 100% dry weight gain at room temperature using an aqueous solution comprising 15 to 60 g/l of the final product W-E or WQ-E. The padded material was then dried at 140°C for 70-90 seconds.
底物:Substrate:
测试:test:
使用织物手感测定仪(例如211-5 Twing Albert)成对地测试柔软性。在评价之前首先对精整样品进行调节(24小时,20℃,65%相对湿度)。Softness is tested in pairs using a Handle Tester (eg 211-5 Twing Albert). Finished samples were first conditioned (24 hours, 20°C, 65% relative humidity) prior to evaluation.
所有的精整均显示出与未精整的织物相比柔软性获得明显改善。All finishes showed a clear improvement in softness compared to unfinished fabrics.
Claims (7)
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP03022447.1 | 2003-10-07 | ||
| EP03022447 | 2003-10-07 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1863844A CN1863844A (en) | 2006-11-15 |
| CN100384913C true CN100384913C (en) | 2008-04-30 |
Family
ID=34429232
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CNB2004800291745A Expired - Fee Related CN100384913C (en) | 2003-10-07 | 2004-10-04 | Amino-functional polysiloxane wax |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP1678239A1 (en) |
| CN (1) | CN100384913C (en) |
| BR (1) | BRPI0415170A (en) |
| WO (1) | WO2005035631A1 (en) |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE102008001867A1 (en) * | 2008-05-19 | 2009-11-26 | Wacker Chemie Ag | Process for the preparation of quaternary ammonium organopolysiloxanes |
| JP5940556B2 (en) | 2010-12-28 | 2016-06-29 | ユニリーバー・ナームローゼ・ベンノートシヤープ | Method for producing emulsion |
| DE102012213248A1 (en) * | 2012-07-27 | 2014-01-30 | Henkel Ag & Co. Kgaa | Nourishing hair dye with silicone (s) and polymer (s) |
| US9845410B2 (en) * | 2014-06-12 | 2017-12-19 | The Chemours Company Fc, Llc | Wax and urethane based extender blends for surface effect compositions |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10500431A (en) * | 1995-04-18 | 1998-01-13 | ローヌ−プーラン シミ | Use of silicone waxes containing ester functional groups for thickening oily media |
| CN1176093A (en) * | 1996-06-07 | 1998-03-18 | 莱雅公司 | Transfer-free make-up or care composition containing alkylpolysiloxane |
| US6165444A (en) * | 1995-09-29 | 2000-12-26 | L'oreal | Composition for treating keratinous material, including at least one silicone-grafted polymer and at least one fatty-chain amide, and uses thereof |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB8919669D0 (en) * | 1989-08-31 | 1989-10-11 | Unilever Plc | Fabric-softening compositions |
-
2004
- 2004-10-04 CN CNB2004800291745A patent/CN100384913C/en not_active Expired - Fee Related
- 2004-10-04 BR BRPI0415170-4A patent/BRPI0415170A/en not_active IP Right Cessation
- 2004-10-04 WO PCT/IB2004/003227 patent/WO2005035631A1/en not_active Ceased
- 2004-10-04 EP EP04769548A patent/EP1678239A1/en not_active Withdrawn
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPH10500431A (en) * | 1995-04-18 | 1998-01-13 | ローヌ−プーラン シミ | Use of silicone waxes containing ester functional groups for thickening oily media |
| US6165444A (en) * | 1995-09-29 | 2000-12-26 | L'oreal | Composition for treating keratinous material, including at least one silicone-grafted polymer and at least one fatty-chain amide, and uses thereof |
| CN1176093A (en) * | 1996-06-07 | 1998-03-18 | 莱雅公司 | Transfer-free make-up or care composition containing alkylpolysiloxane |
Also Published As
| Publication number | Publication date |
|---|---|
| EP1678239A1 (en) | 2006-07-12 |
| BRPI0415170A (en) | 2006-11-28 |
| HK1095846A1 (en) | 2007-05-18 |
| CN1863844A (en) | 2006-11-15 |
| WO2005035631A1 (en) | 2005-04-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP2752449B2 (en) | Aqueous compositions and methods for treating textiles | |
| EP0342834B1 (en) | Treatment of fibrous materials | |
| JP5553505B2 (en) | Polyamino and / or polyammonium / polysiloxane copolymer compound in which polyalkylene oxide units are arranged in a comb shape | |
| US7834122B2 (en) | Polysiloxanes with quaternary ammonium groups, preparation thereof and use thereof as textile softeners | |
| US4624794A (en) | Compositions and process for treating textiles | |
| CN112074559B (en) | Silicones for the treatment of textiles and in cleaning and care formulations | |
| DE102010062156A1 (en) | Polysiloxanes with nitrogen-containing groups | |
| WO2004090007A2 (en) | Reactive amino- and/or ammonium-polysiloxane compounds | |
| US5115049A (en) | Fatty carboxylic silicone amine salts | |
| EP0200325B1 (en) | Method of improving the draining of water from textiles during a laundering operation | |
| JPH04289276A (en) | Method for softening fiber substance and impart- ing hydrophilic nature thereto using polyorgano- silane containing composition | |
| CN100384913C (en) | Amino-functional polysiloxane wax | |
| JPH05239071A (en) | Organosilicon compound | |
| US20040219371A1 (en) | Polysiloxane and textile adjunct comprising a polysiloxane | |
| US7511165B2 (en) | Amino-functional silicone waxes | |
| CN105658749B (en) | Polysiloxanes with quaternized heterocyclic group | |
| JPH07116675B2 (en) | Method for producing softener composition | |
| HK1095846B (en) | Amino-functional silicone waxes | |
| US20040102594A1 (en) | Polysiloxanes with quarternary groups featuring nitrogen atoms | |
| DE10004321A1 (en) | Organosilicon compounds with side chains containing quaternary ammonium groups derived from amine compounds with at least two tert. nitrogen atoms, used as softeners for textile treatment | |
| JP3313073B2 (en) | Liquid fabric softener | |
| CN106700083A (en) | Polyquaternary-ammonium-based glucose polydimethyl siloxane polyether, a preparation method thereof and hand-feeling finishing agent applying polyquaternary-ammonium-based glucose polydimethyl siloxane polyether | |
| JP2009513742A (en) | Polyorganosiloxane | |
| JP2003003369A (en) | Softening base agent and softening and finishing agent | |
| JP2015522670A (en) | Composition and method for treating textile products |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1095846 Country of ref document: HK |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: GR Ref document number: 1095846 Country of ref document: HK |
|
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20080430 Termination date: 20091104 |