Journal Description
Journal of Clinical & Translational Ophthalmology
Journal of Clinical & Translational Ophthalmology
is an international, peer-reviewed, open access journal on ophthalmology published quarterly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: first decisions in 16 days; acceptance to publication in 5.8 days (median values for MDPI journals in the first half of 2024).
- Recognition of Reviewers: APC discount vouchers, optional signed peer review, and reviewer names published annually in the journal.
- Journal of Clinical & Translational Ophthalmology is a companion journal of JCM.
subject
Imprint Information
Open Access
ISSN: 2813-1053
Latest Articles
Rapamycin’s Impact on Age-Related Macular Degeneration—A Systematic Review and Hormesis Perspective
J. Clin. Transl. Ophthalmol. 2024, 2(3), 99-112; https://doi.org/10.3390/jcto2030009 - 17 Sep 2024
Abstract
►
Show Figures
Background: Pre-clinical studies related to the use of rapamycin (Sirolimus®), a mammalian target of rapamycin (mTOR) inhibitors, for age-related macular degeneration (AMD) have shown improved therapeutic outcomes. However, knowledge of its dose–effect relationship in humans with AMD has been limited and
[...] Read more.
Background: Pre-clinical studies related to the use of rapamycin (Sirolimus®), a mammalian target of rapamycin (mTOR) inhibitors, for age-related macular degeneration (AMD) have shown improved therapeutic outcomes. However, knowledge of its dose–effect relationship in humans with AMD has been limited and requires further investigation. Objective: The aim of this study is to assess the safety and efficacy of Sirolimus® for treatment of AMD in humans and determine the dose range for its application in the eye. Methods: A systematic literature review was conducted following the PRISMA guidelines. The MEDLINE, Embase, CINAHL, Scopus and Cochrane Central Registry of Controlled Trials databases were searched for original clinical studies examining the effects of Sirolimus® on outcomes linked to AMD in humans. This review has been registered in the PROSPERO database. Results: Only four studies were found to satisfy the inclusion and exclusion criteria and were analyzed in this systematic review in a narrative way. The dose range of rapamycin in the limited number of studies appears to be toxic to the retina. Conclusion: Future studies should focus on establishing the optimal low-dose range of Sirolimus® that effectively induces autophagy without causing retinal toxicity, as current data indicate a potential therapeutic window that remains underexplored. Specifically, longitudinal, controlled studies with larger, heterogeneous patient populations are necessary to determine the precise dosing that balances efficacy and safety in treating AMD.
Full article
Open AccessArticle
Possible Correlation between Mucin Gene Expression and Symptoms of Dry Eye Syndrome Secondary to Sjogren’s Disease
by
Natalie Brossard-Barbosa, Matias Agoglia, Maria Elena Vergara, Monique Costa, Ernesto Cairoli and Teresa Freire
J. Clin. Transl. Ophthalmol. 2024, 2(3), 87-98; https://doi.org/10.3390/jcto2030008 - 28 Aug 2024
Abstract
►▼
Show Figures
(1) Background: It is estimated that 10% of dry eye disease (DED) occurs in patients with Sjogren’s syndrome (SS-DED) and represents a challenge when it comes to treatment. Both innate and adaptive immunity participate in the pathogenesis of SS-DED. Previous studies suggest that
[...] Read more.
(1) Background: It is estimated that 10% of dry eye disease (DED) occurs in patients with Sjogren’s syndrome (SS-DED) and represents a challenge when it comes to treatment. Both innate and adaptive immunity participate in the pathogenesis of SS-DED. Previous studies suggest that Th1 and Th17 cell immune responses are the main actors associated with the pathogenesis of this disease. Ocular surface mucins play a fundamental role in ocular surface homeostasis. In particular, the main transmembrane mucins, MUC1, MUC4 and MUC16, are dysregulated in DED and could be involved in the activation of pro-inflammatory cytokines at the ocular interface. Thus, the objective of this work was to analyze mucin and cytokine expression in ocular surface (OS) damage and correlate it with clinical symptoms.; (2) Methods: 18 patients with SS-DED and 15 healthy controls were included in the study. Samples of conjunctival cells were obtained through cytology impression. RNA was extracted from the collected samples and used to determine the expression of MUC1, 4 and 16 by qRT-PCR. Pro-inflammatory cytokines associated with DED pathogenesis (IL17 and IL-22) were also evaluated. The results were contrasted with the clinical findings on examination of the patients. (3) Results: We observed a significant increase in the expression of MUC1 and MUC4 in patients with SS-DED. MUC4 significantly correlated with both lower production and stability of the tear film, as well as greater superficial keratopathy. On the other hand, MUC1 and MUC16 were positively correlated with the presence of more severe DED symptoms. However, we could not reproduce an increase in IL-17 and IL-22 in DED patients as previously reported; (4) Conclusions: This work constitutes an approach to understanding how the gene expression of transmembrane mucins associates with SS-DED symptoms and clinical signs.
Full article

Figure 1
Figure 1
<p>mRNA levels of MUC1, MUC4 and MUC16 in patients with SS-DED (<b>A</b>), PSS-DED (<b>B</b>) and SSS-DED (<b>C</b>). mRNA was extracted from conjunctival cells obtained by conjunctival impression and amplified by RT-qPCR. Asterisks correspond to * <span class="html-italic">p</span> < 0.05 and ** <span class="html-italic">p</span> < 0.01; ns = no significant difference between the groups. The y-axis indicates the mucin fold expression in relation to <span class="html-italic">gapdh</span>. Squares correspond to healthy controls and circles to patients with DED.</p> Full article ">Figure 2
<p>Correlation of mucin mRNA levels. mRNA was extracted from conjunctival cells obtained by conjunctival impression and amplified by RT-qPCR (* = <span class="html-italic">p</span> < 0.05). The axes indicate the mucin fold expression in relation to <span class="html-italic">gapdh</span>.</p> Full article ">Figure 3
<p>Correlation of MUC1, MUC4 and MUC16 mRNA levels with TBUT score (<b>A</b>), Schirmer I (<b>B</b>), Oxford Keratopathy score (<b>C</b>) and OSDI score (<b>D</b>). mRNA was extracted from conjunctival cells obtained by conjunctival impression, extracted and amplified by RT-qPCR (* = <span class="html-italic">p</span> < 0.05). The y-axis indicates the mucin fold expression in relation to <span class="html-italic">gapdh</span>.</p> Full article ">Figure 4
<p>IL-17 and IL-22 mRNA levels in conjunctival cells in patients with SS-DED and in healthy controls (<b>A</b>) and patients with SSS and PSS (<b>B</b>). Messenger RNA was extracted from conjunctival cells obtained using the conjunctival impression technique and amplified by RT-qPCR. The y-axis indicates the cytokine fold expression in relation to <span class="html-italic">gapdh</span>. ns = no significant difference between the groups.</p> Full article ">Figure 5
<p>Correlation between IL-17 and IL-22 mRNA levels in conjunctival cells in patients with SS-DED. Messenger RNA was extracted from conjunctival cells obtained using the conjunctival impression technique described in the Materials and Methods section.</p> Full article ">
<p>mRNA levels of MUC1, MUC4 and MUC16 in patients with SS-DED (<b>A</b>), PSS-DED (<b>B</b>) and SSS-DED (<b>C</b>). mRNA was extracted from conjunctival cells obtained by conjunctival impression and amplified by RT-qPCR. Asterisks correspond to * <span class="html-italic">p</span> < 0.05 and ** <span class="html-italic">p</span> < 0.01; ns = no significant difference between the groups. The y-axis indicates the mucin fold expression in relation to <span class="html-italic">gapdh</span>. Squares correspond to healthy controls and circles to patients with DED.</p> Full article ">Figure 2
<p>Correlation of mucin mRNA levels. mRNA was extracted from conjunctival cells obtained by conjunctival impression and amplified by RT-qPCR (* = <span class="html-italic">p</span> < 0.05). The axes indicate the mucin fold expression in relation to <span class="html-italic">gapdh</span>.</p> Full article ">Figure 3
<p>Correlation of MUC1, MUC4 and MUC16 mRNA levels with TBUT score (<b>A</b>), Schirmer I (<b>B</b>), Oxford Keratopathy score (<b>C</b>) and OSDI score (<b>D</b>). mRNA was extracted from conjunctival cells obtained by conjunctival impression, extracted and amplified by RT-qPCR (* = <span class="html-italic">p</span> < 0.05). The y-axis indicates the mucin fold expression in relation to <span class="html-italic">gapdh</span>.</p> Full article ">Figure 4
<p>IL-17 and IL-22 mRNA levels in conjunctival cells in patients with SS-DED and in healthy controls (<b>A</b>) and patients with SSS and PSS (<b>B</b>). Messenger RNA was extracted from conjunctival cells obtained using the conjunctival impression technique and amplified by RT-qPCR. The y-axis indicates the cytokine fold expression in relation to <span class="html-italic">gapdh</span>. ns = no significant difference between the groups.</p> Full article ">Figure 5
<p>Correlation between IL-17 and IL-22 mRNA levels in conjunctival cells in patients with SS-DED. Messenger RNA was extracted from conjunctival cells obtained using the conjunctival impression technique described in the Materials and Methods section.</p> Full article ">
Open AccessOpinion
The Ocular Surface–Gut Axis in Spaceflight: Implications of Intestinal Changes in Microgravity on Tear Film Physiology
by
Ritu Sampige, Joshua Ong, Ethan Waisberg, John Berdahl and Andrew G. Lee
J. Clin. Transl. Ophthalmol. 2024, 2(3), 79-86; https://doi.org/10.3390/jcto2030007 - 31 Jul 2024
Cited by 1
Abstract
►▼
Show Figures
With increasing space exploration, there is a rising need to evaluate the impact of spaceflight on astronauts’ health, including the effects of space-associated hazards such as microgravity. Astronauts’ reports of experienced symptoms upon spaceflight include a notable prevalence of dry eye disease (DED).
[...] Read more.
With increasing space exploration, there is a rising need to evaluate the impact of spaceflight on astronauts’ health, including the effects of space-associated hazards such as microgravity. Astronauts’ reports of experienced symptoms upon spaceflight include a notable prevalence of dry eye disease (DED). Hence, there is a pressing need to understand the pathogenesis and mechanism behind space-associated DED onset, which will subsequently guide the development of necessary therapies to reduce dry eye symptoms among astronauts. One critical effect of spaceflight includes alterations to the gut microbiome. On Earth, the prior literature has established the presence of an ocular surface–gut axis and the potential role of gut dysbiosis in DED onset. Meanwhile, the literature about astronauts’ health underscores the presence of space-associated gut microbiome composition alterations and the presence of DED separately. Therefore, in this opinion article, we review and present the current literature regarding the ocular surface–gut axis on Earth and regarding potential translations to spaceflight. We present the view that, based on the existing literature, the ocular surface–gut axis may be a critical mechanism for the pathogenesis of DED in space, and this axis needs to be further explored in the context of identifying ways to reduce astronauts’ experiences of DED during spaceflight.
Full article

Figure 1
Figure 1
<p>Schematic depiction of the ocular surface–gut axis in spaceflight. Parts of <a href="#jcto-02-00007-f001" class="html-fig">Figure 1</a> were created using unmodified art from Servier Medical Art under a Creative Commons Attribution 4.0 License (<a href="https://creativecommons.org/licenses/by/4.0/" target="_blank">https://creativecommons.org/licenses/by/4.0/</a>) and Clkr [<a href="#B6-jcto-02-00007" class="html-bibr">6</a>] (accessed on 3 March 2024).</p> Full article ">
<p>Schematic depiction of the ocular surface–gut axis in spaceflight. Parts of <a href="#jcto-02-00007-f001" class="html-fig">Figure 1</a> were created using unmodified art from Servier Medical Art under a Creative Commons Attribution 4.0 License (<a href="https://creativecommons.org/licenses/by/4.0/" target="_blank">https://creativecommons.org/licenses/by/4.0/</a>) and Clkr [<a href="#B6-jcto-02-00007" class="html-bibr">6</a>] (accessed on 3 March 2024).</p> Full article ">
Open AccessReview
Hereditary Optic Neuropathies: An Updated Review
by
Samuel K. Lee, Caroline Mura, Nicolas J. Abreu, Janet C. Rucker, Steven L. Galetta, Laura J. Balcer and Scott N. Grossman
J. Clin. Transl. Ophthalmol. 2024, 2(3), 64-78; https://doi.org/10.3390/jcto2030006 - 26 Jun 2024
Abstract
►▼
Show Figures
Hereditary optic neuropathies (HONs) are a class of genetic disorders that may lead to vision loss due to either acute or progressive injury to the optic nerve. Although HONs may commonly manifest as isolated optic atrophy, these disorders can also have a variety
[...] Read more.
Hereditary optic neuropathies (HONs) are a class of genetic disorders that may lead to vision loss due to either acute or progressive injury to the optic nerve. Although HONs may commonly manifest as isolated optic atrophy, these disorders can also have a variety of characteristic clinical features and time courses that may narrow the differential diagnosis. While the two most prevalent HONs are Leber Hereditary Optic Neuropathy (LHON) and Dominant Optic Atrophy (DOA), the phenotypic spectrum of these conditions, as well as genetic landscape of less common optic neuropathies, have been better characterized through advances in molecular diagnostic testing. Treatment targeting various pathogenic mechanisms has been investigated, although studies of clinical applicability remain nascent. Present management largely remains supportive. In this review, we discuss the clinical features, molecular diagnosis, current treatment, and future directions for HONs.
Full article

Figure 1
Open AccessReview
Artificial Intelligence in Glaucoma: A New Landscape of Diagnosis and Management
by
Patrick Xiang Ji, Vethushan Ramalingam, Michael Balas, Lauren Pickel and David J. Mathew
J. Clin. Transl. Ophthalmol. 2024, 2(2), 47-63; https://doi.org/10.3390/jcto2020005 - 25 May 2024
Cited by 1
Abstract
►▼
Show Figures
Glaucoma refers to a spectrum of progressive optic neuropathies and remains the leading cause of irreversible blindness worldwide. Its insidious onset poses serious challenges to conventional diagnostic methods and clinicians striving to detect early-stage disease for timely and effective intervention. Artificial intelligence (AI)
[...] Read more.
Glaucoma refers to a spectrum of progressive optic neuropathies and remains the leading cause of irreversible blindness worldwide. Its insidious onset poses serious challenges to conventional diagnostic methods and clinicians striving to detect early-stage disease for timely and effective intervention. Artificial intelligence (AI) has demonstrated its ability to process and analyze large datasets which can help identify subtle changes in early glaucomatous clinical presentation. This study reviews the current state of AI utilization in glaucoma and elucidates the strengths and limitations of existing approaches. We dissect the role of AI in various domains: enhancing early detection and diagnosis, monitoring disease progression, and refining treatment strategies to optimize patient outcomes. Furthermore, we address the ethical, legal, and social implications, alongside the inherent limitations of AI in the clinical setting. Despite these challenges, AI holds transformative potential for glaucoma management. Future directions emphasize the need for interdisciplinary collaboration, advanced and explainable algorithm development, and equitable healthcare access to fully realize the promise of AI in combating this vision-threatening condition.
Full article

Figure 1
Open AccessArticle
Mutational Analysis of the SLC4A11 Gene in a (Filipino) Family with Congenital Hereditary Endothelial Dystrophy
by
Vicente Lorenzo O. Cabahug, John Paul S. Llido, Loraine Kay D. Cabral, Tricia L. Maynes, Cathlyn Leigh Pinuela, Tommee Lynne Tayengco-Tiu, Ruben Lim Bon Siong and Ma. Luisa D. Enriquez
J. Clin. Transl. Ophthalmol. 2024, 2(2), 34-46; https://doi.org/10.3390/jcto2020004 - 1 Apr 2024
Abstract
►▼
Show Figures
Aim: The study aims to identify if mutations in the SLC4A11 gene are present in Filipino families affected with congenital hereditary endothelial dystrophy (CHED). Methods: This is a family cohort study that investigated the genetic profile of a selected family in northern Luzon,
[...] Read more.
Aim: The study aims to identify if mutations in the SLC4A11 gene are present in Filipino families affected with congenital hereditary endothelial dystrophy (CHED). Methods: This is a family cohort study that investigated the genetic profile of a selected family in northern Luzon, Philippines, whose members were diagnosed with congenital hereditary endothelial dystrophy (CHED). A patient who was diagnosed with CHED prior to this study served as the proband for this family. A detailed family history was obtained and a complete ophthalmologic examination was performed on each of the family members. A total of six affected members and three unaffected members were included in this study. DNA was isolated from peripheral blood samples of the family members, polymerase chain reaction (PCR) was used to amplify the gene’s entire coding region (19 exons and 2 putative promoter regions), and finally, the amplified regions were analyzed using DNA sequencing. Results: Consanguinity was not present in the family. Corneal haze was reported to have been present since birth or shortly thereafter in all the affected patients. Slit-lamp examination showed edematous corneas. Molecular studies of the SLC4A11 gene revealed four novel homozygous point mutations variably presenting in the six affected members, as well as the three unaffected members. One unaffected family member (I-1) had a novel sense mutation absent in the other family members. All affected siblings showed little phenotypic variability. Conclusions: This is the first report that gives us a genetic profile of a northern Luzon family with members affected by CHED. This study supports earlier findings that mutations in the SLC4A11 gene are not consistently the same among different ethnic groups worldwide, probably due to the disease’s genetic heterogeneity. Our study documented five novel mutations, adding to the growing list of mutations probably responsible for acquiring the CHED phenotype. It is possible that there are more novel mutations waiting to be discovered in this hereditary disease. Screening for these specific mutations in other families may prove useful for genetic counseling, prenatal diagnosis, and the future development of gene therapy.
Full article
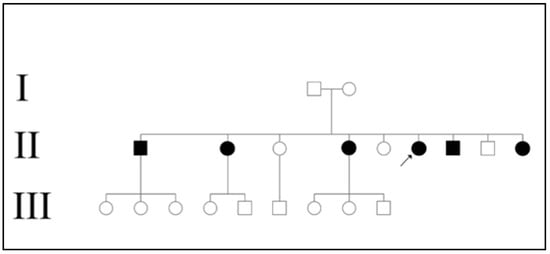
Figure 1
Figure 1
<p>Pedigree showing the three generations (Labelled as I, II, and III) of the family being investigated. Male members are shown as squares while female members are shown as circles. Affected individuals are shown in black solid shapes. Proband is marked with an arrowhead.</p> Full article ">Figure 2
<p>Representative photographs of the eyes of patient II-2 (<b>above</b>) and patient II-9 (<b>below</b>) showing (described eye).</p> Full article ">Figure 3
<p>Representative electropherograms of amplified DNA sequences analyzed in comparison with representative <span class="html-italic">SLC4A11</span> reference sequence (Green: Adenine, Red: Thymine, Blue: Cytosine, Black: Guanine). Five point mutations were observed, mostly located outside the exon regions, except for 10294G>C and 10307G>A, which were both located in Exon 6. Electropherograms were generated using the consensus of triplicate sequences, viewed and analyzed using BioEdit, Seqotron, and 4 Peaks.</p> Full article ">Figure 4
<p>Amino acid sequence alignment of reference and from patient samples. Two point mutations, 10294 G>C and 10307 G>A, at Exon 6 had different effects on translated amino acid sequence. The 10294G>C is a missense mutation, resulting in a change in translated amino acid at location 193 (Red Arrow) to change from arginine (R) to proline (P), while the point mutation at 10307G>A did not result in an amino acid change. Amino acid sequence was generated via ExPASy using the reconstructed DNA sequences as input. Seqotron was used for multiple sequence alignment (Note: Amino acids are highlighted in different color schemes).</p> Full article ">Figure 5
<p>Results of prediction/confidence on the effect of the change in the translated amino acid 193 from arginine to proline due missense mutation 10294G>C. The missense mutation was predicted to be “BENIGN” on both HumDiv and HumVar. According to the PolyPhen2 website (<a href="http://genetics.bwh.harvard.edu/pph2/dokuwiki/overview" target="_blank">http://genetics.bwh.harvard.edu/pph2/dokuwiki/overview</a>, accessed on 15 July 2020) “HumDiv was compiled from all damaging alleles with known effects on the molecular function causing human Mendelian diseases, present in the UniProtKB database, together with differences between human proteins and their closely related mammalian homologs, assumed to be non-damaging; HumVar consisted of all human disease-causing mutations from UniProtKB, together with common human nsSNPs (MAF > 1%) without annotated involvement in disease, which were treated as non-damaging”.</p> Full article ">
<p>Pedigree showing the three generations (Labelled as I, II, and III) of the family being investigated. Male members are shown as squares while female members are shown as circles. Affected individuals are shown in black solid shapes. Proband is marked with an arrowhead.</p> Full article ">Figure 2
<p>Representative photographs of the eyes of patient II-2 (<b>above</b>) and patient II-9 (<b>below</b>) showing (described eye).</p> Full article ">Figure 3
<p>Representative electropherograms of amplified DNA sequences analyzed in comparison with representative <span class="html-italic">SLC4A11</span> reference sequence (Green: Adenine, Red: Thymine, Blue: Cytosine, Black: Guanine). Five point mutations were observed, mostly located outside the exon regions, except for 10294G>C and 10307G>A, which were both located in Exon 6. Electropherograms were generated using the consensus of triplicate sequences, viewed and analyzed using BioEdit, Seqotron, and 4 Peaks.</p> Full article ">Figure 4
<p>Amino acid sequence alignment of reference and from patient samples. Two point mutations, 10294 G>C and 10307 G>A, at Exon 6 had different effects on translated amino acid sequence. The 10294G>C is a missense mutation, resulting in a change in translated amino acid at location 193 (Red Arrow) to change from arginine (R) to proline (P), while the point mutation at 10307G>A did not result in an amino acid change. Amino acid sequence was generated via ExPASy using the reconstructed DNA sequences as input. Seqotron was used for multiple sequence alignment (Note: Amino acids are highlighted in different color schemes).</p> Full article ">Figure 5
<p>Results of prediction/confidence on the effect of the change in the translated amino acid 193 from arginine to proline due missense mutation 10294G>C. The missense mutation was predicted to be “BENIGN” on both HumDiv and HumVar. According to the PolyPhen2 website (<a href="http://genetics.bwh.harvard.edu/pph2/dokuwiki/overview" target="_blank">http://genetics.bwh.harvard.edu/pph2/dokuwiki/overview</a>, accessed on 15 July 2020) “HumDiv was compiled from all damaging alleles with known effects on the molecular function causing human Mendelian diseases, present in the UniProtKB database, together with differences between human proteins and their closely related mammalian homologs, assumed to be non-damaging; HumVar consisted of all human disease-causing mutations from UniProtKB, together with common human nsSNPs (MAF > 1%) without annotated involvement in disease, which were treated as non-damaging”.</p> Full article ">
Open AccessCase Report
A Case Report of Secondary Glaucoma in a Patient with HTLV-1: Discussion about the Management of Secondary Glaucoma Due to Recurrent Granulomatous Uveitis
by
Kenta Ashikaga, Teruhiko Hamanaka, Mayumi Hosogai, Takao Tanaka, Shintaro Nakao and Toshinari Funaki
J. Clin. Transl. Ophthalmol. 2024, 2(2), 26-33; https://doi.org/10.3390/jcto2020003 - 29 Mar 2024
Abstract
►▼
Show Figures
Purpose: to investigate secondary glaucoma resulting from uveitis in a patient infected with Human T-cell Leukemia Virus Type 1 (HTLV-1) pathologically and discuss the management of glaucoma with recurrent uveitis. Clinical course: An octogenarian woman diagnosed as a carrier of HTLV-1 experienced recurrent
[...] Read more.
Purpose: to investigate secondary glaucoma resulting from uveitis in a patient infected with Human T-cell Leukemia Virus Type 1 (HTLV-1) pathologically and discuss the management of glaucoma with recurrent uveitis. Clinical course: An octogenarian woman diagnosed as a carrier of HTLV-1 experienced recurrent uveitis and a sudden rise in intraocular pressure (IOP) in both eyes. Due to the uncontrolled IOP and severely damaged visual field in her left eye, a combined procedure of trabeculectomy and DGIS (glaucoma drainage implant surgery, Baerveldt 350) was performed. The presence of HTLV-1 provirus was detected in the aqueous humor. Her trabeculectomy sample was processed for light microscopic observation. Following an irregular follow-up, she presented with a sudden decrease in vision and pain in her fellow eye, four years after the glaucoma surgeries. Her right eye exhibited a significant accumulation of mutton-fat-like keratic precipitates. Results: Clinical manifestations revealed the presence of granulomatous uveitis. The combined glaucoma surgery, along with continuous topical corticosteroid medication post-surgery in her left eye, effectively suppressed the high IOP spikes and the recurrence of uveitis for 4 years. The pathological examination of the outflow pathways showed a range of damages in Schlemm’s canal (SC), including SC endothelial loss, narrowing, and occlusion, as well as loss of trabecular meshwork (TM) cells and fused TM beams. Conclusion: Combined GDIS and trabeculectomy represents a promising approach for managing such refractory cases of secondary glaucoma. Continuous topical corticosteroid medication is strongly recommended to prevent irreversible changes in SC and TM associated with granulomatous uveitis.
Full article
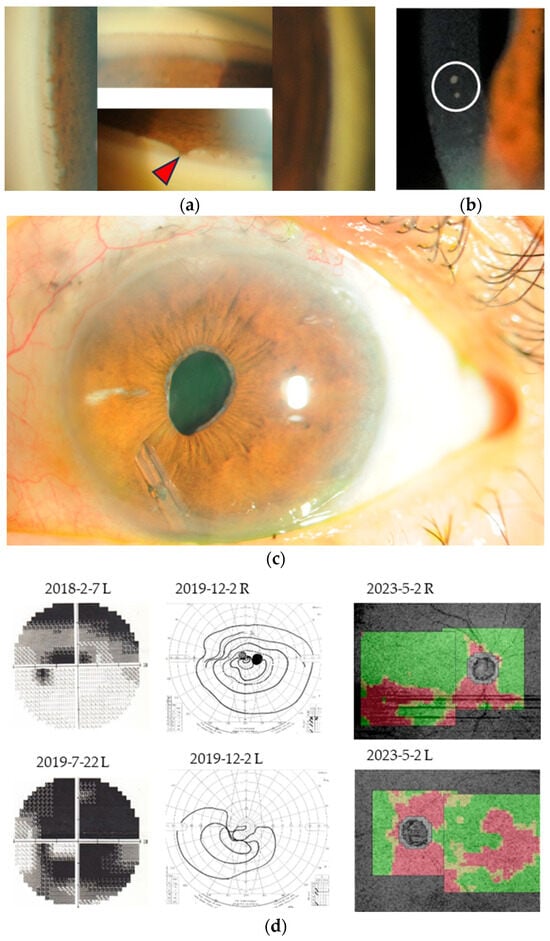
Figure 1
Figure 1
<p>(<b>a</b>): Gonioscopic observations reveal the presence of tent-like peripheral anterior synechia (red arrowhead) at the 12 o’clock position in the right eye but normal angle in the other parts. (<b>b</b>): As illustrated in the image of the slit lamp photograph, a significant reduction in the abundance of keratic precipitates (KPs) was observed, with only two KPs remaining (circled), within a two-week period following the initiation of steroid medication. (<b>c</b>) Slit lamp image capturing the left eye featuring the glaucoma drainage implant (Baerveldt 350). The tube is inserted from the 6 o’clock position in order to locate as much as far from corneal endothelium. Although, tube was in contact with the iris, no inflammation in the anterior chamber was observed for more than 4 years. (<b>d</b>) The outcomes of visual field (VF) examinations and the optic disc’s presentation in optic coherence tomography (OCT) were investigated. Over the duration of the study, a progressive deterioration in VF of the left eye was observed. Concurrently, a substantial decline in OCT was evident in both eyes, aligning with characteristic alterations associated with glaucoma.</p> Full article ">Figure 2
<p>(<b>a</b>–<b>d</b>) Ligh microscopic photographs capturing the details of the Schlemm’s canal (SC) and trabecular meshwork (TM) under light microscopy, bar: 50 µm: (<b>a</b>) Immunohistochemical staining of thrombomodulin in block No. 2; (<b>b</b>) immunohistochemical staining of CD34 in block No. 1; (<b>c</b>) immunohistochemical staining of podoplanin in block No. 3; (<b>d</b>) immunohistochemical staining of CD68 in block No. 2. The Schlemm’s canal (SC) exhibited complete openness and appeared histologically normal (<b>a</b>). However, the SC endothelium locally showed signs of disappearance (indicated by black arrowheads in (<b>a</b>); additionally, certain regions within the trabecular meshwork (TM) displayed localized absence of TM cells (stars in (<b>a</b>,<b>b</b>)) in block No. 2. Furthermore, the SC experienced narrowing (indicated by open arrowheads in (<b>b</b>) or complete collapse (solid arrowheads in (<b>b</b>)). The SC underwent complete collapse (<b>c</b>), while the corneoscleral and juxta-canalicular meshwork fused together, displaying sparse podoplanin staining (indicated by stars in (<b>c</b>)). In this region, a reduced number of TM cells were observed, and there were no spaces available for the aqueous humor. CD68-positive cells (<b>d</b>) were observed in the trabecular meshwork (TM), Schlemm’s canal (SC), ciliary muscle (CM), ciliary processes (CP), and iris. These CD68-positive cells appeared to be infiltrated around the SC and collector channels (CC).</p> Full article ">Figure 2 Cont.
<p>(<b>a</b>–<b>d</b>) Ligh microscopic photographs capturing the details of the Schlemm’s canal (SC) and trabecular meshwork (TM) under light microscopy, bar: 50 µm: (<b>a</b>) Immunohistochemical staining of thrombomodulin in block No. 2; (<b>b</b>) immunohistochemical staining of CD34 in block No. 1; (<b>c</b>) immunohistochemical staining of podoplanin in block No. 3; (<b>d</b>) immunohistochemical staining of CD68 in block No. 2. The Schlemm’s canal (SC) exhibited complete openness and appeared histologically normal (<b>a</b>). However, the SC endothelium locally showed signs of disappearance (indicated by black arrowheads in (<b>a</b>); additionally, certain regions within the trabecular meshwork (TM) displayed localized absence of TM cells (stars in (<b>a</b>,<b>b</b>)) in block No. 2. Furthermore, the SC experienced narrowing (indicated by open arrowheads in (<b>b</b>) or complete collapse (solid arrowheads in (<b>b</b>)). The SC underwent complete collapse (<b>c</b>), while the corneoscleral and juxta-canalicular meshwork fused together, displaying sparse podoplanin staining (indicated by stars in (<b>c</b>)). In this region, a reduced number of TM cells were observed, and there were no spaces available for the aqueous humor. CD68-positive cells (<b>d</b>) were observed in the trabecular meshwork (TM), Schlemm’s canal (SC), ciliary muscle (CM), ciliary processes (CP), and iris. These CD68-positive cells appeared to be infiltrated around the SC and collector channels (CC).</p> Full article ">Figure 3
<p>(<b>a</b>,<b>b</b>) A light microscopic image of the Schlemm’s canal (SC), trabecular meshwork (TM), iris, and ciliary muscle (CM) is presented in the immunohistochemical staining of CD4 (<b>a</b>) and CD8 (<b>b</b>) in block No. 3 and No. 2, respectively (bar: 50 µm). No CD4-positive cells (<b>a</b>) or CD8-positive cells (<b>b</b>) were detected in the anterior segment of the eye. Light microscopic images of a formalin-fixed tonsil specimen from a patient with HTLV-1 are presented in the immunohistochemical staining of CD4 (inset (<b>a</b>)) and CD8 (inset (<b>b</b>)) (bar: 50 µm). Abundant CD4-positive cells and CD8-positive cells were observed.</p> Full article ">
<p>(<b>a</b>): Gonioscopic observations reveal the presence of tent-like peripheral anterior synechia (red arrowhead) at the 12 o’clock position in the right eye but normal angle in the other parts. (<b>b</b>): As illustrated in the image of the slit lamp photograph, a significant reduction in the abundance of keratic precipitates (KPs) was observed, with only two KPs remaining (circled), within a two-week period following the initiation of steroid medication. (<b>c</b>) Slit lamp image capturing the left eye featuring the glaucoma drainage implant (Baerveldt 350). The tube is inserted from the 6 o’clock position in order to locate as much as far from corneal endothelium. Although, tube was in contact with the iris, no inflammation in the anterior chamber was observed for more than 4 years. (<b>d</b>) The outcomes of visual field (VF) examinations and the optic disc’s presentation in optic coherence tomography (OCT) were investigated. Over the duration of the study, a progressive deterioration in VF of the left eye was observed. Concurrently, a substantial decline in OCT was evident in both eyes, aligning with characteristic alterations associated with glaucoma.</p> Full article ">Figure 2
<p>(<b>a</b>–<b>d</b>) Ligh microscopic photographs capturing the details of the Schlemm’s canal (SC) and trabecular meshwork (TM) under light microscopy, bar: 50 µm: (<b>a</b>) Immunohistochemical staining of thrombomodulin in block No. 2; (<b>b</b>) immunohistochemical staining of CD34 in block No. 1; (<b>c</b>) immunohistochemical staining of podoplanin in block No. 3; (<b>d</b>) immunohistochemical staining of CD68 in block No. 2. The Schlemm’s canal (SC) exhibited complete openness and appeared histologically normal (<b>a</b>). However, the SC endothelium locally showed signs of disappearance (indicated by black arrowheads in (<b>a</b>); additionally, certain regions within the trabecular meshwork (TM) displayed localized absence of TM cells (stars in (<b>a</b>,<b>b</b>)) in block No. 2. Furthermore, the SC experienced narrowing (indicated by open arrowheads in (<b>b</b>) or complete collapse (solid arrowheads in (<b>b</b>)). The SC underwent complete collapse (<b>c</b>), while the corneoscleral and juxta-canalicular meshwork fused together, displaying sparse podoplanin staining (indicated by stars in (<b>c</b>)). In this region, a reduced number of TM cells were observed, and there were no spaces available for the aqueous humor. CD68-positive cells (<b>d</b>) were observed in the trabecular meshwork (TM), Schlemm’s canal (SC), ciliary muscle (CM), ciliary processes (CP), and iris. These CD68-positive cells appeared to be infiltrated around the SC and collector channels (CC).</p> Full article ">Figure 2 Cont.
<p>(<b>a</b>–<b>d</b>) Ligh microscopic photographs capturing the details of the Schlemm’s canal (SC) and trabecular meshwork (TM) under light microscopy, bar: 50 µm: (<b>a</b>) Immunohistochemical staining of thrombomodulin in block No. 2; (<b>b</b>) immunohistochemical staining of CD34 in block No. 1; (<b>c</b>) immunohistochemical staining of podoplanin in block No. 3; (<b>d</b>) immunohistochemical staining of CD68 in block No. 2. The Schlemm’s canal (SC) exhibited complete openness and appeared histologically normal (<b>a</b>). However, the SC endothelium locally showed signs of disappearance (indicated by black arrowheads in (<b>a</b>); additionally, certain regions within the trabecular meshwork (TM) displayed localized absence of TM cells (stars in (<b>a</b>,<b>b</b>)) in block No. 2. Furthermore, the SC experienced narrowing (indicated by open arrowheads in (<b>b</b>) or complete collapse (solid arrowheads in (<b>b</b>)). The SC underwent complete collapse (<b>c</b>), while the corneoscleral and juxta-canalicular meshwork fused together, displaying sparse podoplanin staining (indicated by stars in (<b>c</b>)). In this region, a reduced number of TM cells were observed, and there were no spaces available for the aqueous humor. CD68-positive cells (<b>d</b>) were observed in the trabecular meshwork (TM), Schlemm’s canal (SC), ciliary muscle (CM), ciliary processes (CP), and iris. These CD68-positive cells appeared to be infiltrated around the SC and collector channels (CC).</p> Full article ">Figure 3
<p>(<b>a</b>,<b>b</b>) A light microscopic image of the Schlemm’s canal (SC), trabecular meshwork (TM), iris, and ciliary muscle (CM) is presented in the immunohistochemical staining of CD4 (<b>a</b>) and CD8 (<b>b</b>) in block No. 3 and No. 2, respectively (bar: 50 µm). No CD4-positive cells (<b>a</b>) or CD8-positive cells (<b>b</b>) were detected in the anterior segment of the eye. Light microscopic images of a formalin-fixed tonsil specimen from a patient with HTLV-1 are presented in the immunohistochemical staining of CD4 (inset (<b>a</b>)) and CD8 (inset (<b>b</b>)) (bar: 50 µm). Abundant CD4-positive cells and CD8-positive cells were observed.</p> Full article ">
Open AccessArticle
Relationship of Visual Function with Incident Fall and Mortality among Patients with Anterior Ischemic Optic Neuropathy
by
Braden V. Saba, Joshua A. Grosser, Dominic Prado, Brian A. Robusto, Javier Nahmias, Gillian Treadwell and Atalie C. Thompson
J. Clin. Transl. Ophthalmol. 2024, 2(1), 15-25; https://doi.org/10.3390/jcto2010002 - 7 Feb 2024
Abstract
►▼
Show Figures
The purpose of this study was to determine if worse visual acuity is associated with an increased risk of incident fall or mortality, independent of other relevant cofounders. This was a single-center retrospective cohort study of 428 new cases of anterior ischemic optic
[...] Read more.
The purpose of this study was to determine if worse visual acuity is associated with an increased risk of incident fall or mortality, independent of other relevant cofounders. This was a single-center retrospective cohort study of 428 new cases of anterior ischemic optic neuropathy (AION). Separate Cox proportional hazards models were constructed to evaluate the relationship between either logMAR visual acuity (VA) or legal blindness (20/200 or worse VA) and survival time to (1) encounter for fall, (2) mortality, and (3) a composite adverse outcome. Multivariable models were adjusted for a priori confounders. In adjusted models, a 1-unit increase in logMAR VA was associated with a significantly greater risk of an incident fall (adjusted HR 1.36, 95% CI (1.06–1.73), p = 0.014) and of mortality (adjusted HR 1.44, 95% CI (1.15–1.82), p = 0.002). Meeting criteria for legal blindness was also significantly associated with a higher risk of incident fall (HR 1.80, 95% CI (1.05–3.07), p = 0.032) and mortality (adjusted HR 2.16, 95% CI (1.29–3.63), p = 0.004). Among patients with AION, worse visual acuity or legal blindness conferred a significantly increased risk of falls and mortality, independent of coexistent comorbidities. Future studies should consider fall reduction interventions for patients with poor vision from severe eye disease such as AION.
Full article
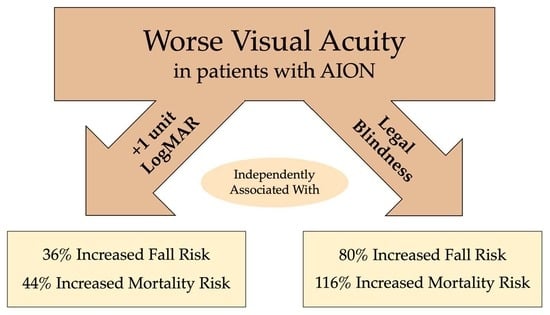
Graphical abstract
Graphical abstract
Full article ">Figure 1
<p>The unadjusted and adjusted association of visual acuity function with risk of fall, mortality, and a composite outcome among patients with acute anterior ischemic optic neuropathy. <b>Legend:</b> The point is the Hazard ratio and the bar represents the 95% confidence interval. LogMAR VA = logarithm of the minimum angle of resolution visual acuity. Legal Blindness = visual acuity 20/200 or worse.</p> Full article ">
Full article ">Figure 1
<p>The unadjusted and adjusted association of visual acuity function with risk of fall, mortality, and a composite outcome among patients with acute anterior ischemic optic neuropathy. <b>Legend:</b> The point is the Hazard ratio and the bar represents the 95% confidence interval. LogMAR VA = logarithm of the minimum angle of resolution visual acuity. Legal Blindness = visual acuity 20/200 or worse.</p> Full article ">
Open AccessArticle
Preparation and In Vitro Testing of Brinzolamide-Loaded Poly Lactic-Co-Glycolic Acid (PLGA) Nanoparticles for Sustained Drug Delivery
by
Ann-Marie Ako-Adounvo and Pradeep K. Karla
J. Clin. Transl. Ophthalmol. 2024, 2(1), 1-14; https://doi.org/10.3390/jcto2010001 - 9 Jan 2024
Cited by 1
Abstract
►▼
Show Figures
Glaucoma therapy aims at lowering intra-ocular pressure (IOP). Brinzolamide, a carbonic anhydrase inhibitor, is utilized as a second-line medication for treating ocular hypertension and primary open-angle glaucoma (POAG). The drug lowers the IOP making it a therapeutic agent against glaucoma, and due to
[...] Read more.
Glaucoma therapy aims at lowering intra-ocular pressure (IOP). Brinzolamide, a carbonic anhydrase inhibitor, is utilized as a second-line medication for treating ocular hypertension and primary open-angle glaucoma (POAG). The drug lowers the IOP making it a therapeutic agent against glaucoma, and due to its poor water solubility, is commercially available as Azopt®, a 1% ophthalmic suspension. Adverse effects such as blurred vision, ocular irritation, discomfort, and bitter taste are associated with the use of the marketed brinzolamide formulation. This study aims to test the feasibility of formulating and in vitro testing of brinzolamide-PLGA nanoparticles for improved toxicity profile. The nanoparticles were prepared by the oil-in-water (O/W) emulsion-solvent evaporation method. Particle size and zeta potential were determined by dynamic light scattering (DLS). The morphology of the nanoparticles was determined by scanning electron microscopy (SEM). Encapsulation of the drug was verified by high-performance liquid chromatography (HPLC) and the compatibility of the polymer and drug was verified by Fourier transform infrared (FTIR) spectroscopy. The in vitro drug release profile was assessed employing the dialysis method. Intracellular localization of the nanoparticles was assessed by confocal microscopy utilizing Rhodamine 123-loaded nanoparticles. Cytotoxicity of the formulation was assessed on Statens Seruminstitut Rabbit Cornea (SIRC) and transfected Human Corneal Epithelial (SV40 HCEC) cell lines. The particle size of the nanoparticle formulations ranged from 202.3 ± 2.9 nm to 483.1 ± 27.9 nm for blank nanoparticles, and 129.6 ± 1.5 nm to 350.9 ± 8.5 nm for drug-loaded nanoparticles. The polydispersity of the formulations ranged from 0.071 ± 0.032 to 0.247 ± 0.043 for blank nanoparticles, and 0.089 ± 0.028 to 0.158 ± 0.004 for drug-loaded nanoparticles. Drug loading and encapsulation efficiencies ranged from 7.42–15.84% and 38.93–74.18%, respectively. The in vitro drug release profile for the optimized formulation was biphasic, with a ~54% burst release for the initial 3 h, followed by a cumulative 85% and 99% released at 24 and 65 h, respectively. Uptake study showed nanoparticles(NPs) localization in the cytoplasm and around the nuclei of the cells. Brinzolamide-PLGA nanoparticles were successfully developed, characterized, and tested in vitro. Preliminary data show intracellular localization of the nanoparticles in the cytoplasm of SIRC and SV40 HCEC cells. The formulations appeared to be relatively non-cytotoxic to the cells. The research data from the study provided preliminary data for successful development and promising in vitro absorption efficacy for brinzolamide-loaded PLGA nanoparticle formulation.
Full article
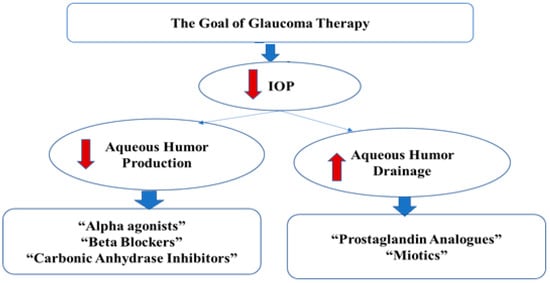
Figure 1
Figure 1
<p>Glaucoma disease pathology and treatment.</p> Full article ">Figure 2
<p>Chemical structure of brinzolamide.</p> Full article ">Figure 3
<p>Size distribution of: (<b>A</b>) Blank PLGA NPs (Formulation F10B), (<b>B</b>) brinzolamide-loaded PLGA NPs (Formulation BNZ001).</p> Full article ">Figure 4
<p>Typical SEM images of freeze-dried brinzolamide-loaded PLGA nanoparticles (BNZ004) at various magnifications.</p> Full article ">Figure 5
<p>FTIR spectra of pure drug, blank PLGA nanoparticles, and drug-loaded PLGA nanoparticles (BNZ004).</p> Full article ">Figure 6
<p>(<b>a</b>) HPLC chromatogram of brinzolamide. (<b>b</b>) Calibration curve of brinzolamide.</p> Full article ">Figure 7
<p>Drug release profile of optimized formulation BNZ004 (N = 3, mean ± Stdev).</p> Full article ">Figure 8
<p>Cytotoxicity study of blank PLGA NPs, positive control (Lysis Solution), Brinzolamide-PLGA NPs (BNZ004) and pure drug suspension. Statistical significance was measured by two-factor ANOVA.</p> Full article ">Figure 9
<p>Confocal images showing intracellular localization of nanoparticles.</p> Full article ">
<p>Glaucoma disease pathology and treatment.</p> Full article ">Figure 2
<p>Chemical structure of brinzolamide.</p> Full article ">Figure 3
<p>Size distribution of: (<b>A</b>) Blank PLGA NPs (Formulation F10B), (<b>B</b>) brinzolamide-loaded PLGA NPs (Formulation BNZ001).</p> Full article ">Figure 4
<p>Typical SEM images of freeze-dried brinzolamide-loaded PLGA nanoparticles (BNZ004) at various magnifications.</p> Full article ">Figure 5
<p>FTIR spectra of pure drug, blank PLGA nanoparticles, and drug-loaded PLGA nanoparticles (BNZ004).</p> Full article ">Figure 6
<p>(<b>a</b>) HPLC chromatogram of brinzolamide. (<b>b</b>) Calibration curve of brinzolamide.</p> Full article ">Figure 7
<p>Drug release profile of optimized formulation BNZ004 (N = 3, mean ± Stdev).</p> Full article ">Figure 8
<p>Cytotoxicity study of blank PLGA NPs, positive control (Lysis Solution), Brinzolamide-PLGA NPs (BNZ004) and pure drug suspension. Statistical significance was measured by two-factor ANOVA.</p> Full article ">Figure 9
<p>Confocal images showing intracellular localization of nanoparticles.</p> Full article ">
Open AccessArticle
Pulsed Ultrasound Modulates the Cytotoxic Effect of Cisplatin and Doxorubicin on Cultured Human Retinal Pigment Epithelium Cells (ARPE-19)
by
Seyed Omid Mohammadi, Megan C. LaRocca, Christopher D. Yang, Jordan Jessen, M. Cristina Kenney and Ken Y. Lin
J. Clin. Transl. Ophthalmol. 2023, 1(4), 107-116; https://doi.org/10.3390/jcto1040013 - 27 Nov 2023
Abstract
►▼
Show Figures
Objective: Pulsed ultrasound has been proposed as a tool to enhance ocular drug delivery, but its effects on drug potency are not well understood. Doxorubicin-HCl and cisplatin are two drugs commonly used to treat ocular melanoma. We report the effects of pulsed ultrasound
[...] Read more.
Objective: Pulsed ultrasound has been proposed as a tool to enhance ocular drug delivery, but its effects on drug potency are not well understood. Doxorubicin-HCl and cisplatin are two drugs commonly used to treat ocular melanoma. We report the effects of pulsed ultrasound on the cytotoxicity of doxorubicin-HCl and cisplatin in vitro. Methods: Cultured human retinal pigment epithelium cells (ARPE-19) were treated with doxorubicin-HCl or cisplatin in the presence or absence of ultrasound. MTT and Trypan blue assays were performed at 24 and 48 h post treatment to assess cell metabolism and death. Results: Cells treated with ultrasound plus doxorubicin-HCl demonstrated a significant decrease in metabolism compared with cells treated with doxorubicin-HCl alone. In contrast, cells treated with ultrasound plus cisplatin exhibited a significant increase in metabolism compared with cells treated with cisplatin alone for 48 h. Cells treated with cisplatin and pretreated with ultrasound (US–Cis) exhibited a significant decrease in metabolism. Cell death was similar in doxorubicin- and cisplatin-treated cells with and without ultrasound. Conclusion: Pulsed ultrasound enhances the cytotoxicity of doxorubicin-HCl at 24 and 48 h post treatment but abrogates cisplatin toxicity 48 h post treatment. This suggests ultrasound modulates cell–drug interactions in a drug-specific manner. These findings may influence the future development of ultrasound-assisted ocular drug delivery systems.
Full article
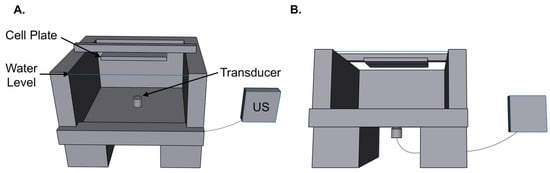
Figure 1
Figure 1
<p>(<b>A</b>) Three-dimensional CAD render of the experimental ultrasound (US) set-up. Inverted cell plates were placed 1 inch from the transducer tip at its focal point. The blue line denotes a water level allowing full submersion of the cell plate. The apparatus allowed only lateral movement (in the x- and y-direction) of the cell plate and maintained a fixed 1-inch distance between the cell plate and transducer. (<b>B</b>) Anterior view of our CAD render of the experimental US set-up.</p> Full article ">Figure 2
<p>The effects of ultrasound treatment alone on ARPE-19 cell metabolism at a pulse frequency of 1 MHz, a power of 4.93 W/cm<sup>2</sup>, and a PRF of 30 Hz (<b>A</b>) and 100 Hz (<b>B</b>). After 48 h, there was no significant decrease in cell metabolism at a PRF of 30 Hz (<span class="html-italic">p</span> = 0.1398). There was a significant decrease in cell metabolism at a PRF of 100 Hz (24 h, <span class="html-italic">p</span> = 0.0002; 48 h, <span class="html-italic">p</span> = 0.0181). These results confirm the effectiveness of our ultrasound setup. <span class="html-italic">p</span>-values < 0.05, <0.001, are denoted as *, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">Figure 3
<p>Doxorubicin-HCl toxicity in ARPE-19 cells. At 24 h post incubation (<b>A</b>), results were variable, including a significant increase in cell metabolism at 100 μg/mL and decrease in metabolism at 10 μg/mL (100 μg/mL, <span class="html-italic">p</span> = 0.0002; 10 μg/mL, <span class="html-italic">p</span> < 0.0001). At 48 h post incubation (<b>B</b>), cells displayed a significant dose-dependent decrease in cell metabolism (0.01 μg/mL, <span class="html-italic">p</span> = 0.0231; 0.1 μg/mL, <span class="html-italic">p</span> < 0.0001; 1 μg/mL, <span class="html-italic">p</span> < 0.0001; 10 μg/mL, <span class="html-italic">p</span> < 0.0001; 100 μg/mL, <span class="html-italic">p</span> < 0.0001). <span class="html-italic">p</span>-values < 0.05, <0.001, are denoted as *, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">Figure 4
<p>Ultrasound application with varying concentrations of doxorubicin-HCl at 24 h (<b>A</b>) and 48 h (<b>B</b>). The graphs demonstrate the effects of doxorubicin-HCl-treated cells with and without ultrasound exposure. When exposed to ultrasound, there was a significantly greater dose-dependent decrease in cell metabolism at 0.1 μg/mL, greater at both 24 and 48 h (<span class="html-italic">p</span> < 0.05). There was no effect visualized on 0.01 μg/mL of doxorubicin-HCl (24 h, <span class="html-italic">p</span> = 0.1488, 48 h, <span class="html-italic">p</span> = 0.607). <span class="html-italic">p</span>-values < 0.05, <0.01, <0.001 are denoted as *, **, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">Figure 5
<p>Trypan blue analysis of doxorubicin- and cisplatin-treated cells with and without ultrasound (US) exposure at 24 h (<b>A</b>) and 48 h (<b>B</b>). Amount of 100 μg/mL and 1 μg/mL of doxorubicin-HCl and 40 μM and 20 μM of cisplatin were tested with and without pulsed ultrasound. At both 24 and 48 h, there was no significant differences seen between cells treated with both doxorubicin-HCl and ultrasound and the doxorubicin-treated control (1 μg/mL: 24 h, <span class="html-italic">p</span> = 0.995; 48 h, <span class="html-italic">p</span> = 0.937; 100 μg/mL: 24 h, <span class="html-italic">p</span> = 0.606; 48 h, <span class="html-italic">p</span> = 0.794). There was no significant relationship seen between cisplatin-treated and ultrasound-treated cells and the cisplatin-treated controls (20 μM: 24 h, <span class="html-italic">p</span> = 0.870; 48 h, <span class="html-italic">p</span> = 0.151; 40 μM: 24 h, <span class="html-italic">p</span> = 0.715; 48 h, <span class="html-italic">p</span> = 0.0863).</p> Full article ">Figure 6
<p>Effects of ultrasound plus cisplatin (<b>A</b>,<b>B</b>) on ARPE-19 cells at 24 h (<b>A</b>), 48 h (<b>B</b>). Panel (<b>C</b>) shows cells treated with US–Cis (cisplatin exposed to US prior to cell plating) after 24 h. There were no significant effects seen at 24 h post incubation (<b>A</b>). After 48 h, ultrasound-treated plus cisplatin-treated cells had increased cell metabolism compared with the metabolism of the cisplatin-treated controls (<span class="html-italic">p</span> < 0.0001) (<b>B</b>). The 20 μM concentrations of US–Cis had a decrease in cell metabolism (<span class="html-italic">p</span> = 0.0093) (<b>C</b>). No significant difference in metabolism was found between 40 μM US–Cis and cisplatin-treated cells (<span class="html-italic">p</span> = 0.4004, <b>C</b>). <span class="html-italic">p</span>-values < 0.01, <0.001, are denoted as **, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">
<p>(<b>A</b>) Three-dimensional CAD render of the experimental ultrasound (US) set-up. Inverted cell plates were placed 1 inch from the transducer tip at its focal point. The blue line denotes a water level allowing full submersion of the cell plate. The apparatus allowed only lateral movement (in the x- and y-direction) of the cell plate and maintained a fixed 1-inch distance between the cell plate and transducer. (<b>B</b>) Anterior view of our CAD render of the experimental US set-up.</p> Full article ">Figure 2
<p>The effects of ultrasound treatment alone on ARPE-19 cell metabolism at a pulse frequency of 1 MHz, a power of 4.93 W/cm<sup>2</sup>, and a PRF of 30 Hz (<b>A</b>) and 100 Hz (<b>B</b>). After 48 h, there was no significant decrease in cell metabolism at a PRF of 30 Hz (<span class="html-italic">p</span> = 0.1398). There was a significant decrease in cell metabolism at a PRF of 100 Hz (24 h, <span class="html-italic">p</span> = 0.0002; 48 h, <span class="html-italic">p</span> = 0.0181). These results confirm the effectiveness of our ultrasound setup. <span class="html-italic">p</span>-values < 0.05, <0.001, are denoted as *, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">Figure 3
<p>Doxorubicin-HCl toxicity in ARPE-19 cells. At 24 h post incubation (<b>A</b>), results were variable, including a significant increase in cell metabolism at 100 μg/mL and decrease in metabolism at 10 μg/mL (100 μg/mL, <span class="html-italic">p</span> = 0.0002; 10 μg/mL, <span class="html-italic">p</span> < 0.0001). At 48 h post incubation (<b>B</b>), cells displayed a significant dose-dependent decrease in cell metabolism (0.01 μg/mL, <span class="html-italic">p</span> = 0.0231; 0.1 μg/mL, <span class="html-italic">p</span> < 0.0001; 1 μg/mL, <span class="html-italic">p</span> < 0.0001; 10 μg/mL, <span class="html-italic">p</span> < 0.0001; 100 μg/mL, <span class="html-italic">p</span> < 0.0001). <span class="html-italic">p</span>-values < 0.05, <0.001, are denoted as *, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">Figure 4
<p>Ultrasound application with varying concentrations of doxorubicin-HCl at 24 h (<b>A</b>) and 48 h (<b>B</b>). The graphs demonstrate the effects of doxorubicin-HCl-treated cells with and without ultrasound exposure. When exposed to ultrasound, there was a significantly greater dose-dependent decrease in cell metabolism at 0.1 μg/mL, greater at both 24 and 48 h (<span class="html-italic">p</span> < 0.05). There was no effect visualized on 0.01 μg/mL of doxorubicin-HCl (24 h, <span class="html-italic">p</span> = 0.1488, 48 h, <span class="html-italic">p</span> = 0.607). <span class="html-italic">p</span>-values < 0.05, <0.01, <0.001 are denoted as *, **, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">Figure 5
<p>Trypan blue analysis of doxorubicin- and cisplatin-treated cells with and without ultrasound (US) exposure at 24 h (<b>A</b>) and 48 h (<b>B</b>). Amount of 100 μg/mL and 1 μg/mL of doxorubicin-HCl and 40 μM and 20 μM of cisplatin were tested with and without pulsed ultrasound. At both 24 and 48 h, there was no significant differences seen between cells treated with both doxorubicin-HCl and ultrasound and the doxorubicin-treated control (1 μg/mL: 24 h, <span class="html-italic">p</span> = 0.995; 48 h, <span class="html-italic">p</span> = 0.937; 100 μg/mL: 24 h, <span class="html-italic">p</span> = 0.606; 48 h, <span class="html-italic">p</span> = 0.794). There was no significant relationship seen between cisplatin-treated and ultrasound-treated cells and the cisplatin-treated controls (20 μM: 24 h, <span class="html-italic">p</span> = 0.870; 48 h, <span class="html-italic">p</span> = 0.151; 40 μM: 24 h, <span class="html-italic">p</span> = 0.715; 48 h, <span class="html-italic">p</span> = 0.0863).</p> Full article ">Figure 6
<p>Effects of ultrasound plus cisplatin (<b>A</b>,<b>B</b>) on ARPE-19 cells at 24 h (<b>A</b>), 48 h (<b>B</b>). Panel (<b>C</b>) shows cells treated with US–Cis (cisplatin exposed to US prior to cell plating) after 24 h. There were no significant effects seen at 24 h post incubation (<b>A</b>). After 48 h, ultrasound-treated plus cisplatin-treated cells had increased cell metabolism compared with the metabolism of the cisplatin-treated controls (<span class="html-italic">p</span> < 0.0001) (<b>B</b>). The 20 μM concentrations of US–Cis had a decrease in cell metabolism (<span class="html-italic">p</span> = 0.0093) (<b>C</b>). No significant difference in metabolism was found between 40 μM US–Cis and cisplatin-treated cells (<span class="html-italic">p</span> = 0.4004, <b>C</b>). <span class="html-italic">p</span>-values < 0.01, <0.001, are denoted as **, and ***, respectively, and all denote statistical significance; nonsignificance is denoted as “ns”.</p> Full article ">
Open AccessArticle
A Case Series of Transscleral Cyclophotocoagulation Nd:YAG Laser in Refractory Glaucoma
by
Gregorio Lo Giudice, Giuseppe Miceli, Giuseppe Tumminello, Alessandro Trapani and Antonino Pioppo
J. Clin. Transl. Ophthalmol. 2023, 1(4), 97-106; https://doi.org/10.3390/jcto1040012 - 26 Oct 2023
Abstract
►▼
Show Figures
Purpose: To describe the efficacy and safety of surgical treatment in refractory glaucoma using transscleral cyclophotocoagulation with a diode laser (TSCPC). Cyclo-G6 laser with a G-probe delivery system (Iridex, Silicon Valley, CA, USA). Materials and Methods: 134 patients (134 eyes) with refractory glaucoma
[...] Read more.
Purpose: To describe the efficacy and safety of surgical treatment in refractory glaucoma using transscleral cyclophotocoagulation with a diode laser (TSCPC). Cyclo-G6 laser with a G-probe delivery system (Iridex, Silicon Valley, CA, USA). Materials and Methods: 134 patients (134 eyes) with refractory glaucoma were included in this study. Patients received transscleral cyclophotocoagulation (TSCPC) using the IRIDEX Cyclo-G6 diode laser equipped with a G-probe delivery system. The eyes were treated with 1250–1500 mW for 3500–4000 ms depending on the iris colour. Intraocular pressure (IOP) and the count of antiglaucoma medications (AGM) were documented at the initial assessment and again at 3, 6, and 12 months following the treatment. Results: A reduction in IOP was observed in 97.73% of eyes at 12 months. At 12 months, 55.97% of the 134 eyes decreased IOP under 21 mmHg. At the 3-month mark, 96.27% of eyes attained an IOP reduction in 30% or more from their initial baseline levels, 85.61% at 6 months, and 82.17% at 12 months. A reduction in AGM was recorded for 86.07% of patients at 6 months and 87.31% at 12 months. There were no instances of severe complications reported, and minor complications were observed in 3% (4 out of 134) of the treated eyes. Conclusions: IRIDEX Cyclo-G6 laser is a surgical method effective in reducing IOP in patients with refractory glaucoma
Full article
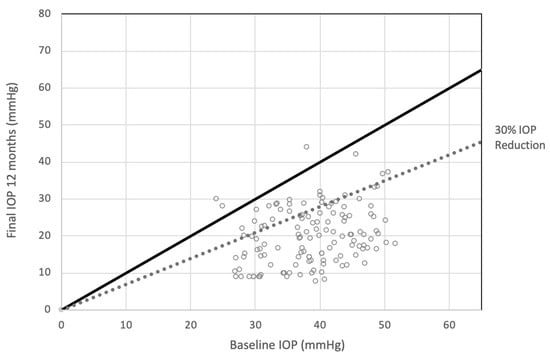
Figure 1
Figure 1
<p>Scattergram showing intraocular pressure (IOP) at baseline and at the end of follow-up at 12 months. At 12 months after treatment, the 82.17% of patients achieved post-treatment IOP reduction of 30% or more.</p> Full article ">Figure 2
<p>Kaplan–Meier survival curves were generated to assess surgical success in all eyes, based on the following criteria: achieving an IOP of <21 mmHg or a 30% reduction from baseline at the last visit (12 months), without the need for additional glaucoma medication and in the absence of other indicators of glaucoma progression.</p> Full article ">Figure 3
<p>Cumulative survival for different types of glaucoma.</p> Full article ">Figure 4
<p>Cumulative survival for IOP baseline. No statistically significant differences for baseline IOP.</p> Full article ">Figure 5
<p>Cumulative survival for different types of surgery. No statistically significant differences for different types of surgery.</p> Full article ">
<p>Scattergram showing intraocular pressure (IOP) at baseline and at the end of follow-up at 12 months. At 12 months after treatment, the 82.17% of patients achieved post-treatment IOP reduction of 30% or more.</p> Full article ">Figure 2
<p>Kaplan–Meier survival curves were generated to assess surgical success in all eyes, based on the following criteria: achieving an IOP of <21 mmHg or a 30% reduction from baseline at the last visit (12 months), without the need for additional glaucoma medication and in the absence of other indicators of glaucoma progression.</p> Full article ">Figure 3
<p>Cumulative survival for different types of glaucoma.</p> Full article ">Figure 4
<p>Cumulative survival for IOP baseline. No statistically significant differences for baseline IOP.</p> Full article ">Figure 5
<p>Cumulative survival for different types of surgery. No statistically significant differences for different types of surgery.</p> Full article ">
Open AccessArticle
The Effect of Antiopioid State Laws on Prescribing Patterns in an Oculoplastic Practice
by
Elahhe Afkhamnejad, Cooper Stevenson and Praveena Gupta
J. Clin. Transl. Ophthalmol. 2023, 1(3), 91-96; https://doi.org/10.3390/jcto1030011 - 18 Aug 2023
Abstract
►▼
Show Figures
This study aimed to find out whether three Texas state policies restricting opioid prescriptions have influenced the prescribing patterns of the oculoplastic department at the University of Texas Medical Branch (UTMB). This is a retrospective chart review of 520 patients at UTMB between
[...] Read more.
This study aimed to find out whether three Texas state policies restricting opioid prescriptions have influenced the prescribing patterns of the oculoplastic department at the University of Texas Medical Branch (UTMB). This is a retrospective chart review of 520 patients at UTMB between 2019 and 2021, reporting the amount of morphine milliequivalents (MMEs) prescribed to patients undergoing one of eight oculoplastic procedures before and after passage of each state policy. Of 520 patients, 218 received opioids. There were no changes in MMEs/month after the first policy (95% CI: −0.1531–0.1256, p = 0.85), second policy (95% CI: −0.000997–0.00093, p = 0.97), or third policy (95% CI: −0.001431–0.00164, p = 0.096). The passage and implementation of three Texas state antiopioid policies were not associated with significant changes in opioid prescribing patterns following oculoplastic and orbital surgeries at UTMB. Average MMEs/month were driven by orbital-floor fractures, which composed most procedures overall. Limitations included the small population and that only a single provider was reviewed. Future studies would be beneficial in further evaluating the role of state legislation on opioid prescribing patterns and usage among patients.
Full article
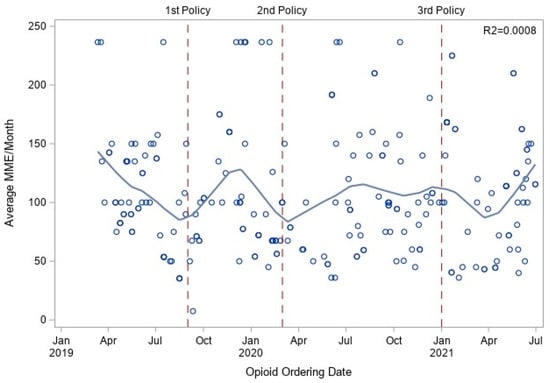
Figure 1
Open AccessArticle
Synergistic Effect of Substance P with Insulin and Insulin-Like Growth Factor-I on Epithelial Migration of the Transformed Human Corneal Epithelial Cells (SV-40)
by
Phat Tran, Jonathan Kopel and Ted W. Reid
J. Clin. Transl. Ophthalmol. 2023, 1(3), 79-90; https://doi.org/10.3390/jcto1030010 - 17 Jul 2023
Abstract
►▼
Show Figures
On the cornea, daily epithelial resurfacing is a critical process to prevent the loss of normal function, ocular morbidity, corneal structure, and vision loss. There are several components to wound healing, including cellular attachment, migration, and growth. To develop a treatment for corneal
[...] Read more.
On the cornea, daily epithelial resurfacing is a critical process to prevent the loss of normal function, ocular morbidity, corneal structure, and vision loss. There are several components to wound healing, including cellular attachment, migration, and growth. To develop a treatment for corneal epithelial healing, we studied the effect of substance P (SP) on corneal epithelial cell migration using a cell culture system of either transformed human corneal epithelial cells (SV-40), or rabbit corneal epithelial cells (SIRC). We investigated the effect of SP with insulin and insulin-like growth factor-I (IGF-1). We found that SP is synergistic with insulin and IGF-1 on the stimulation of transformed human corneal epithelial migration in a cell culture system, as well as in rabbit SIRC cells. The addition of either SP, insulin, or IGF-1 alone did not greatly affect epithelial migration, while the combination of SP and insulin or SP and IGF-1 markedly stimulated epithelial migration in a dose-dependent manner. The synergistic effects of SP with insulin and SP with IGF-1 were inhibited by the addition of the SP-specific inhibitor (CP96345). However, the effect of insulin and IGF-1 alone were not inhibited by the NK-1-specific inhibitor (CP96345). Our results are consistent with the need for the involvement of the neuropeptide SP in corneal epithelial wound healing of diabetic corneas where nerve-ending dropout occurs. Additionally, almost identical results were obtained with human and rabbit corneal epithelial cells. These results suggest that the maintenance of the normal integrity of the corneal epithelium might be regulated by both humoral and neural factors.
Full article
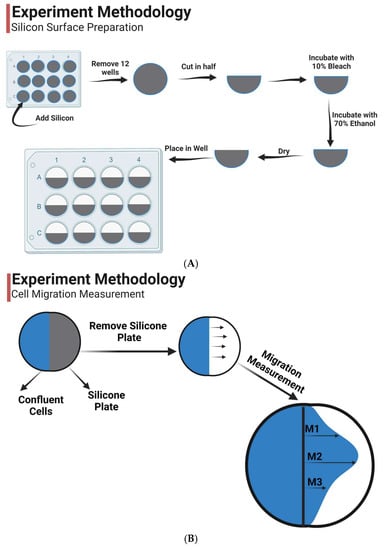
Figure 1
Figure 1
<p>(<b>A</b>) Experiment methodology for silicon surface preparation. (<b>B</b>) Cell migration protocol summary (M1, M2, and M3 are the three measurements used for the analysis).</p> Full article ">Figure 2
<p>(<b>A</b>) The effect of SP on cellular migration of SV-40 Cells. SV-40 cells were treated with various concentrations of SP in DMEM-F12-free serum media. Error bars represent the SEM from three determinations. (<b>B</b>) Cell morphology of SV-40 cells [<a href="#B24-jcto-01-00010" class="html-bibr">24</a>]. The measurements were taken after 7 days.</p> Full article ">Figure 3
<p>Cellular migration of SV-40—transformed human corneal epithelial cells increases with time after treatment with SP at 1 nM and various concentrations of IGF-1. Error bars represent the SEM from three determinations.</p> Full article ">Figure 4
<p>Cellular migration of SV-40—transformed human corneal epithelial cells increases with time after treatment with SP at 1 nM and various concentrations of insulin. Error bars represent the SEM from three determinations.</p> Full article ">Figure 5
<p>The SP-stimulated migration in the presence of IGF-1 is inhibited by the NK-1-specific inhibitor CP96345. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 6
<p>The SP-stimulated migration in the presence of insulin is inhibited by the NK-1-specific inhibitor CP96345. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 7
<p>SP is synergistic with IGF-1 on cellular migration of SV-40 at 1 nM. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 8
<p>Effect of SP (1 nM) with insulin, on cellular migration of SV-40. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance * <span class="html-italic">p</span> < 0.05 and ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 9
<p>(<b>A</b>) The effect of IGF-1 on human SV-40-transformed cell migration with and without the SP receptor-specific inhibitor CP96345. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. (<b>B</b>) Effect of insulin on cellular migration of human SV-40-transformed cells after 7 days, with and without the SP receptor-specific inhibitor CP96345. Error bars represent the SEM from three determinations.</p> Full article ">
<p>(<b>A</b>) Experiment methodology for silicon surface preparation. (<b>B</b>) Cell migration protocol summary (M1, M2, and M3 are the three measurements used for the analysis).</p> Full article ">Figure 2
<p>(<b>A</b>) The effect of SP on cellular migration of SV-40 Cells. SV-40 cells were treated with various concentrations of SP in DMEM-F12-free serum media. Error bars represent the SEM from three determinations. (<b>B</b>) Cell morphology of SV-40 cells [<a href="#B24-jcto-01-00010" class="html-bibr">24</a>]. The measurements were taken after 7 days.</p> Full article ">Figure 3
<p>Cellular migration of SV-40—transformed human corneal epithelial cells increases with time after treatment with SP at 1 nM and various concentrations of IGF-1. Error bars represent the SEM from three determinations.</p> Full article ">Figure 4
<p>Cellular migration of SV-40—transformed human corneal epithelial cells increases with time after treatment with SP at 1 nM and various concentrations of insulin. Error bars represent the SEM from three determinations.</p> Full article ">Figure 5
<p>The SP-stimulated migration in the presence of IGF-1 is inhibited by the NK-1-specific inhibitor CP96345. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 6
<p>The SP-stimulated migration in the presence of insulin is inhibited by the NK-1-specific inhibitor CP96345. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 7
<p>SP is synergistic with IGF-1 on cellular migration of SV-40 at 1 nM. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 8
<p>Effect of SP (1 nM) with insulin, on cellular migration of SV-40. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. Significance (*) was defined as a <span class="html-italic">p</span> < 0.05 using Student’s <span class="html-italic">t</span>-test. The different number of stars represent significance * <span class="html-italic">p</span> < 0.05 and ** <span class="html-italic">p</span> < 0.01.</p> Full article ">Figure 9
<p>(<b>A</b>) The effect of IGF-1 on human SV-40-transformed cell migration with and without the SP receptor-specific inhibitor CP96345. The measurements were taken after 7 days. Error bars represent the SEM from three determinations. (<b>B</b>) Effect of insulin on cellular migration of human SV-40-transformed cells after 7 days, with and without the SP receptor-specific inhibitor CP96345. Error bars represent the SEM from three determinations.</p> Full article ">
Open AccessCase Report
Cataract Surgery following Corneal Allogenic Intrastromal Ring Segments and Implantable Collamer Lens
by
Arsalan Akbar Ali, Bobby Saenz and Taj Nasser
J. Clin. Transl. Ophthalmol. 2023, 1(3), 72-78; https://doi.org/10.3390/jcto1030009 - 26 Jun 2023
Abstract
►▼
Show Figures
The management of cataracts in keratoconus patients poses a challenge due to the irregular corneal shape and variability in corneal topography, which may lead to errors in determining corneal power. In this report, we present a case of a 48-year-old male with a
[...] Read more.
The management of cataracts in keratoconus patients poses a challenge due to the irregular corneal shape and variability in corneal topography, which may lead to errors in determining corneal power. In this report, we present a case of a 48-year-old male with a history of keratoconus and prior Visian Implantable Collamer lenses and Corneal Allogenic Intrastromal Ring Segments procedures, who presented with a nuclear cataract in his right eye. To address this patient’s complex case, he underwent ICL explantation, cataract extraction, and intraocular lens (IOL) implantation, utilizing the Johnson & Johnson Sensar AR40 monofocal 3-piece lens with a power of −9.5. The Barrett True K formula predicted a spherical equivalent of −1.76, and at the post-operative one-month follow-up, the uncorrected distance visual acuity (UDVA) was 20/60, with pinhole improvement to 20/50. The manifest refraction was −2.50–3.25 × 145, and the best corrected visual acuity was 20/25. This case report highlights the unique challenges encountered in managing keratoconus patients with a history of prior ICL and CAIRS procedures, followed by cataract extraction. Our findings underscore the importance of a comprehensive approach in the management of progressive keratoconus and cataracts to ensure optimal outcomes.
Full article
Figure 1
Figure 1
<p>Sheimpflug tomography demonstrates severe keratoconus as the image displays significant steepening of the cornea before and after the CAIRS procedure. We can appreciate the 10 D flattening of the central cornea that occurred after the surgery.</p> Full article ">Figure 2
<p>Anterior segment optical coherence tomography measuring a normal vault, which is the distance between posterior surface of the ICL and anterior lens capsule.</p> Full article ">
<p>Sheimpflug tomography demonstrates severe keratoconus as the image displays significant steepening of the cornea before and after the CAIRS procedure. We can appreciate the 10 D flattening of the central cornea that occurred after the surgery.</p> Full article ">Figure 2
<p>Anterior segment optical coherence tomography measuring a normal vault, which is the distance between posterior surface of the ICL and anterior lens capsule.</p> Full article ">
Open AccessPerspective
Recontextualizing Neuromyelitis Optica as a Systemic Condition: A Perspective
by
Parker Webber, Brianna C. Landis and Amanda E. Brooks
J. Clin. Transl. Ophthalmol. 2023, 1(2), 61-71; https://doi.org/10.3390/jcto1020008 - 24 May 2023
Abstract
►▼
Show Figures
Neuromyelitis optica spectrum disorder (NMOSD), a demyelinating CNS disorder in which inflammatory cells infiltrate the spinal cord and optic nerve, has been identified as an AQP4-IgG-positive disease. Some of its most common clinical characteristics are optic neuritis, acute myelitis, area postrema syndrome, and
[...] Read more.
Neuromyelitis optica spectrum disorder (NMOSD), a demyelinating CNS disorder in which inflammatory cells infiltrate the spinal cord and optic nerve, has been identified as an AQP4-IgG-positive disease. Some of its most common clinical characteristics are optic neuritis, acute myelitis, area postrema syndrome, and brainstem syndrome. However, the relationship between aquaporin-4 (AQP4) and NMOSD appears to be involved in pathologies outside of the CNS due to the fact that autoimmune, muscular, and paraneoplastic syndromes are more common in patients with NMOSD. This perspective presents an analysis of the current literature on neuromyelitis optica in an effort to further understand and compile pathologies that arise outside of the CNS secondary to NMOSD. Recontextualizing neuromyelitis optica as a systemic condition will facilitate greater diagnostic ability and improved treatment approaches.
Full article
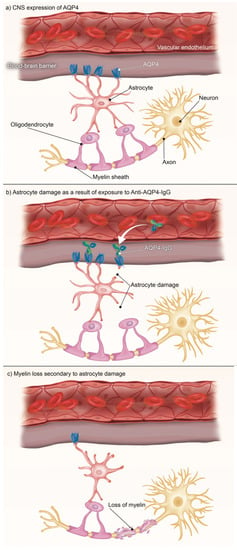
Figure 1
Figure 1
<p>Anti-AQP4-antibody disease in the CNS occurs when a disruption to the blood–brain barrier allows circulating AQP4-IgG to access the CNS and bind with the AQP4 channels found on astrocytes. These astrocytes can no longer support the oligodendrocytes that myelinate the neurons in the CNS, and this loss of myelination leads to the various symptoms of NMOSD.</p> Full article ">Figure 2
<p>NMOSD is an anti-AQP4 antibody disease. AQP4 is found throughout the body [<xref ref-type="bibr" rid="B60-jcto-01-00008">60</xref>], particularly in the regions in the above figure. As the traditional definition of NMOSD as a CNS-exclusive disorder is challenged, further connections are being discovered that demonstrate the possibility of pathologies arising in any body region where AQP4 is expressed.</p> Full article ">
<p>Anti-AQP4-antibody disease in the CNS occurs when a disruption to the blood–brain barrier allows circulating AQP4-IgG to access the CNS and bind with the AQP4 channels found on astrocytes. These astrocytes can no longer support the oligodendrocytes that myelinate the neurons in the CNS, and this loss of myelination leads to the various symptoms of NMOSD.</p> Full article ">Figure 2
<p>NMOSD is an anti-AQP4 antibody disease. AQP4 is found throughout the body [<xref ref-type="bibr" rid="B60-jcto-01-00008">60</xref>], particularly in the regions in the above figure. As the traditional definition of NMOSD as a CNS-exclusive disorder is challenged, further connections are being discovered that demonstrate the possibility of pathologies arising in any body region where AQP4 is expressed.</p> Full article ">
Open AccessFeature PaperArticle
In Vitro Anti-Inflammatory Potential of Pomegranate Extract (Pomanox®) in a Reconstituted Human Corneal Epithelium Model
by
Marcos Peñalver-Mellado, Fredy Silva-Fuentes, Agustín Villar, Anna Mula and Andrea Zangara
J. Clin. Transl. Ophthalmol. 2023, 1(2), 52-60; https://doi.org/10.3390/jcto1020007 - 19 Apr 2023
Abstract
►▼
Show Figures
An in vitro study was conducted using a model of reconstituted human corneal epithelial (SkinEthic™ HCE/Human Corneal Epithelium) to test the modulation of cytokines secretion activity of Pomanox® (PMX), a standardized commercial extract of pomegranate fruit characterized by high punicalagin α +
[...] Read more.
An in vitro study was conducted using a model of reconstituted human corneal epithelial (SkinEthic™ HCE/Human Corneal Epithelium) to test the modulation of cytokines secretion activity of Pomanox® (PMX), a standardized commercial extract of pomegranate fruit characterized by high punicalagin α + β content. Cell viability and inhibition of the release of interleukin-8 (IL-8) was evaluated in four conditions: negative control, positive inflammatory control with lipopolysaccharide (LPS) from Escherichia coli, positive anti-inflammatory control (LPS plus dexamethasone), and LPS plus PMX after 24 h of culture. The mean (±standard error of mean (SEM)) IL-8 level was 48.7 ± 5.1 pg/mL in the PMX condition vs. 172.7 ± 19.3 and 26.6 ± 1.2 in the LPS from E. coli and negative control, respectively (p < 0.05) and 93.8 ± 8.7 pg/mL in LPS plus dexamethasone (p = 0.165). The percentages of inhibition of IL-8 release were 45.7% for LPS plus dexamethasone and 63.8% for LPS plus PMX. The percentage of cell viability (86%) was also higher for the LPS plus PMX condition. The present findings add evidence to the anti-inflammatory effect of a PMX in an in vitro model of reconstituted corneal epithelial cells.
Full article
Figure 1
Figure 1
<p>Percentage of cell viability in the different study groups: phosphate-buffered saline (PBS), negative control (NC); lipopolysaccharide from <italic>E. coli</italic> (LPS), positive inflammatory control; LPS plus PMX, test sample; and LPS plus dexamethasone (DXM), positive anti-inflammatory control. See number of runs in <xref ref-type="table" rid="jcto-01-00007-t001">Table 1</xref>.</p> Full article ">Figure 2
<p>Release of IL-8 in the different study groups. * <italic>p</italic> < 0.05 of LPS and LPS + PMX vs. negative control; #: <italic>p</italic> < 0.05 vs. LPS. (NC) (LPS: lipopolysaccharide, PMX: Pomanox<sup>®</sup>; DXM: dexamethasone). Data expressed as mean ± SEM. See number of runs in <xref ref-type="table" rid="jcto-01-00007-t001">Table 1</xref>.</p> Full article ">
<p>Percentage of cell viability in the different study groups: phosphate-buffered saline (PBS), negative control (NC); lipopolysaccharide from <italic>E. coli</italic> (LPS), positive inflammatory control; LPS plus PMX, test sample; and LPS plus dexamethasone (DXM), positive anti-inflammatory control. See number of runs in <xref ref-type="table" rid="jcto-01-00007-t001">Table 1</xref>.</p> Full article ">Figure 2
<p>Release of IL-8 in the different study groups. * <italic>p</italic> < 0.05 of LPS and LPS + PMX vs. negative control; #: <italic>p</italic> < 0.05 vs. LPS. (NC) (LPS: lipopolysaccharide, PMX: Pomanox<sup>®</sup>; DXM: dexamethasone). Data expressed as mean ± SEM. See number of runs in <xref ref-type="table" rid="jcto-01-00007-t001">Table 1</xref>.</p> Full article ">
Open AccessReview
The Activity of Substance P (SP) on the Corneal Epithelium
by
Jonathan Kopel, Caezaan Keshvani, Kelly Mitchell and Ted Reid
J. Clin. Transl. Ophthalmol. 2023, 1(2), 35-51; https://doi.org/10.3390/jcto1020006 - 27 Mar 2023
Cited by 1
Abstract
►▼
Show Figures
In 1931, Von Euler and Gaddum isolated substance P (SP), an undecapeptide from the tachykinin family, from equine brain and intestine tissue extracts. Numerous types of cells, including neurons, astrocytes, microglia, epithelial, and endothelial cells, as well as immune cells including T-cells, dendritic
[...] Read more.
In 1931, Von Euler and Gaddum isolated substance P (SP), an undecapeptide from the tachykinin family, from equine brain and intestine tissue extracts. Numerous types of cells, including neurons, astrocytes, microglia, epithelial, and endothelial cells, as well as immune cells including T-cells, dendritic cells, and eosinophils, are responsible for its production. The corneal epithelium, immune cells, keratocytes, and neurons all express the two isoforms of NK1R, which has the highest affinity for SP. The most recent research supports SP’s contribution to corneal healing by encouraging epithelial cell migration and proliferation. Additionally, when applied to the eyes, SP has proinflammatory effects that result in miosis, intraocular inflammation, and conjunctival hyperemia. In this review article, we examine the role of substance P within the eye. We focus on the role of SP with regards to maintenance and healing of the corneal epithelium.
Full article
Figure 1
Figure 1
<p>Corneal healing using SP in dogs. (<bold>A</bold>) Cornea of capsaicin rabbit after healing—not treated with Substance P (note epithelium is not well attached). (<bold>B</bold>) Cornea of capsaicin rabbit after healing—treated with Substance P (note epithelium is well attached). (<bold>C</bold>) Dog eye with corneal ulcer in the lower left corner (haze in the background is a cataract). (<bold>D</bold>) Dog cornea after one week treatment with substance P, which shows resolution of the corneal ulcer.</p> Full article ">Figure 2
<p>The effect of Substance P and IGF-1 on corneal ulcer healing in a human patient. (<bold>A</bold>) Two-year-old patient with skin flap over ulcer in right eye. (<bold>B</bold>) Same patient with large corneal ulcer in left eye. (<bold>C</bold>) Left eye after two weeks treatment with SP with no ulcer remaining. (<bold>D</bold>) Right eye, six months after cornea transplant and treatment with SP.</p> Full article ">Figure 3
<p>Substance P release from trigeminal nerve: Trigeminal nerve (V1) innervates the corneal epithelium, and it releases SP from its axonal terminals. Keratinocytes have been shown to express Neurokinin 1 (Nk1) receptors that promote inflammation and are important for wound regeneration. Mast cells have been shown to express MgrpX2 receptor that stimulates mast cells to release cytokines and promote inflammation.</p> Full article ">
<p>Corneal healing using SP in dogs. (<bold>A</bold>) Cornea of capsaicin rabbit after healing—not treated with Substance P (note epithelium is not well attached). (<bold>B</bold>) Cornea of capsaicin rabbit after healing—treated with Substance P (note epithelium is well attached). (<bold>C</bold>) Dog eye with corneal ulcer in the lower left corner (haze in the background is a cataract). (<bold>D</bold>) Dog cornea after one week treatment with substance P, which shows resolution of the corneal ulcer.</p> Full article ">Figure 2
<p>The effect of Substance P and IGF-1 on corneal ulcer healing in a human patient. (<bold>A</bold>) Two-year-old patient with skin flap over ulcer in right eye. (<bold>B</bold>) Same patient with large corneal ulcer in left eye. (<bold>C</bold>) Left eye after two weeks treatment with SP with no ulcer remaining. (<bold>D</bold>) Right eye, six months after cornea transplant and treatment with SP.</p> Full article ">Figure 3
<p>Substance P release from trigeminal nerve: Trigeminal nerve (V1) innervates the corneal epithelium, and it releases SP from its axonal terminals. Keratinocytes have been shown to express Neurokinin 1 (Nk1) receptors that promote inflammation and are important for wound regeneration. Mast cells have been shown to express MgrpX2 receptor that stimulates mast cells to release cytokines and promote inflammation.</p> Full article ">
Open AccessCase Report
Visual Rehabilitation in Post Mild Traumatic Brain Injury. Case-Based Review
by
Carmen López-de-la-Fuente, Hermes Barriga-Longás and Elvira Orduna-Hospital
J. Clin. Transl. Ophthalmol. 2023, 1(1), 25-34; https://doi.org/10.3390/jcto1010005 - 24 Feb 2023
Abstract
►▼
Show Figures
Background: Traumatic brain injury (TBI) can cause visual dysfunction affecting binocularity, spatial orientation, posture, and balance. Currently, there are several options for treating manifested visual disturbances; vision therapy is one of the possible treatment options. Methods: A 14-year-old female fainted and sustained trauma
[...] Read more.
Background: Traumatic brain injury (TBI) can cause visual dysfunction affecting binocularity, spatial orientation, posture, and balance. Currently, there are several options for treating manifested visual disturbances; vision therapy is one of the possible treatment options. Methods: A 14-year-old female fainted and sustained trauma to the upper-temporal part of her right eyebrow. The patient presented with eye pain, a decreased visual field, and blurred vision and she exhibited great difficulties when reading. Both neurological tests and exploration of the anterior and posterior ocular segments showed results within normal limits. The patient was diagnosed with fusional vergence dysfunction, associated with accommodative infacility and oculomotor dysfunction. To eliminate her symptoms, a visual rehabilitation program was implemented; it consisted of accommodative, anti-suppressive, vergential, motility, hand–eye coordination, and peripheral vision exercises. Results: The symptoms manifested by the patient gradually dissipated throughout the course of therapy. However, not all optometric parameters reached normal values after visual therapy, which could indicate a traumatic injury that limited the achievement of normal optometric ranges. Conclusions: After a mild traumatic brain injury (mTBI), it is necessary to carry out a complete examination of the patient’s visual function. This visual examination must include an analysis of vergences, ocular motility, and the accommodative system to obtain an accurate diagnosis. The outcomes of the present clinical case imply that visual therapy should be considered as a treatment in cases of mTBIs.
Full article
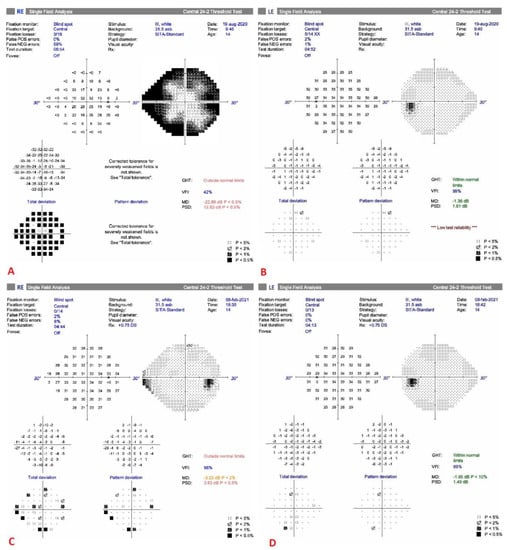
Figure 1
Figure 1
<p>(<bold>A</bold>) Humphrey visual field (HVF, threshold 24-2) of the right eye (RE) after a mild traumatic brain injury (mTBI) and before starting visual therapy showing generalized loss of sensitivity. (<bold>B</bold>) HVF 24-2 of the left eye (LE) after an mTBI and before starting visual therapy. (<bold>C</bold>) HVF 24-2 of the RE after visual therapy showing generalized improvement in sensitivity and (<bold>D</bold>) HVF 24-2 of the LE after visual therapy. Abbreviations: RE: right eye; LE: left eye; POS: positive; NEG: negative; GHT: glaucoma hemifield test; VFI: visual field index; MD: mean deviation; PSD: pattern standard deviation.</p> Full article ">
<p>(<bold>A</bold>) Humphrey visual field (HVF, threshold 24-2) of the right eye (RE) after a mild traumatic brain injury (mTBI) and before starting visual therapy showing generalized loss of sensitivity. (<bold>B</bold>) HVF 24-2 of the left eye (LE) after an mTBI and before starting visual therapy. (<bold>C</bold>) HVF 24-2 of the RE after visual therapy showing generalized improvement in sensitivity and (<bold>D</bold>) HVF 24-2 of the LE after visual therapy. Abbreviations: RE: right eye; LE: left eye; POS: positive; NEG: negative; GHT: glaucoma hemifield test; VFI: visual field index; MD: mean deviation; PSD: pattern standard deviation.</p> Full article ">
Open AccessFeature PaperArticle
Filtering Bleb Characteristics in Combined Cataract Surgery with Ex-PRESS Implant vs. Non-Penetrating Deep Sclerectomy. A Prospective, Randomized, Multi-Center Study
by
Alfonso Anton, Marcos Muñoz, Marta Castany, Alfonso Gil, Alberto Martinez, Francisco Muñoz-Negrete, Jose Urcelay and Javier Moreno-Montañes
J. Clin. Transl. Ophthalmol. 2023, 1(1), 15-24; https://doi.org/10.3390/jcto1010004 - 20 Jan 2023
Cited by 1
Abstract
►▼
Show Figures
(1) Background: After filtering surgery, bleb morphology is an indicator of the factors that may determine the final intraocular pressure (IOP). The present study aimed to evaluate and compare filtering bleb characteristics after combined cataract and glaucoma surgery. (2) Methods: We conducted a
[...] Read more.
(1) Background: After filtering surgery, bleb morphology is an indicator of the factors that may determine the final intraocular pressure (IOP). The present study aimed to evaluate and compare filtering bleb characteristics after combined cataract and glaucoma surgery. (2) Methods: We conducted a prospective multi-center randomized trial. Eyes with glaucoma and cataract were randomly assigned to either phacoemulsification and filtration surgery with an EX-PRESS implant (Alcon) or non-penetrating deep sclerectomy (NPDS) with an ESNOPER implant (AJL). The bleb characteristics were assessed with the Moorfields bleb grading system at months 1 and 12 of follow-up, and the relationship with IOP was analyzed. (3) Results: There were significant changes in bleb appearance between the assessments at month 1 and month 12. The changes in bleb appearance were more evident in the EX-PRESS group. The central area in the EX-PRESS group decreased from 2.9 at month 1 to 2.4 at month 12 (p = 0.014). Bleb height in the EX-PRESS group decreased from 2.3 at month 1 to 1.8 at month 12 (p = 0.034). The vascularity of the central area in the NPDS group decreased from 1.8 at month 1 to 1.3 at month 12 (p = 0.02). The maximal bleb area was inversely related (r = −0.39; p = 0.03) to the IOP in the NPDS group at month 1. Vascularity in the central area was directly related (r = 0.39; p = 0.01) to a higher IOP in the EX-PRESS group at month 1. Vascularity in the central area (r = 0.56; p < 0.001) and maximal area (r = 0.37; p = 0.012) at month 1 was directly related to a higher IOP in the EX-PRESS group at month 12. (4) Conclusions: More intense vascularity at month 1 was related to a higher final IOP in the EX-PRESS group. Larger blebs were associated with a lower IOP in the NPDS group.
Full article
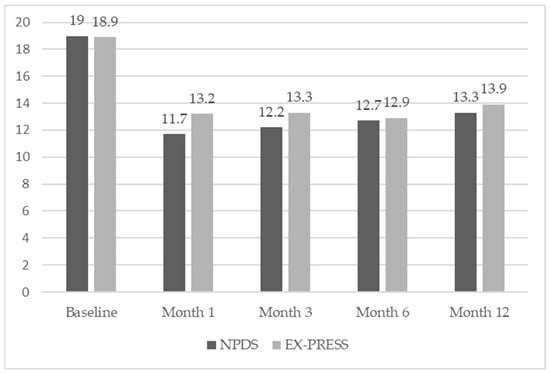
Figure 1
Figure 1
<p>Mean IOP at baseline and follow-up visits in the NPDS vs. EX-PRESS groups in mmHg. The study was completed by 47 persons in the NPDS group and 48 persons in the EX-PRESS group. The mean IOP was similar in the two groups at all time points.</p> Full article ">Figure 2
<p>(<bold>a</bold>) Good prognosis bleb. Moorfields bleb grading system (MBGS): 1a,b (3), 2 (2), 3a,b,c (2). (<bold>b</bold>) Poor prognosis bleb. MBGS: 1a,b (3), 2 (3), 3a (4), 3b (4), 3c (2). White arrow, central zone; yellow arrow, maximal area zone; black arrow, non-bleb zone.</p> Full article ">
<p>Mean IOP at baseline and follow-up visits in the NPDS vs. EX-PRESS groups in mmHg. The study was completed by 47 persons in the NPDS group and 48 persons in the EX-PRESS group. The mean IOP was similar in the two groups at all time points.</p> Full article ">Figure 2
<p>(<bold>a</bold>) Good prognosis bleb. Moorfields bleb grading system (MBGS): 1a,b (3), 2 (2), 3a,b,c (2). (<bold>b</bold>) Poor prognosis bleb. MBGS: 1a,b (3), 2 (3), 3a (4), 3b (4), 3c (2). White arrow, central zone; yellow arrow, maximal area zone; black arrow, non-bleb zone.</p> Full article ">
Open AccessCase Report
Recurrent Uveitis-Glaucoma-Hyphema Syndrome Due to Positional Pupillary Capture after Sutureless Scleral-Fixated Secondary Intraocular Lens Placement
by
Sagar Patel and Hossein Ameri
J. Clin. Transl. Ophthalmol. 2023, 1(1), 11-14; https://doi.org/10.3390/jcto1010003 - 31 Dec 2022
Abstract
►▼
Show Figures
Here, we present a case of floppy iris leading to positional pupillary capture by a sutureless, scleral-fixated intraocular lens (IOL) causing recurrent uveitis-glaucoma-hyphema (UGH) syndrome. The patient developed recurrent episodes of UGH syndrome after dislocated IOL removal and the placement of sutureless, scleral-fixated
[...] Read more.
Here, we present a case of floppy iris leading to positional pupillary capture by a sutureless, scleral-fixated intraocular lens (IOL) causing recurrent uveitis-glaucoma-hyphema (UGH) syndrome. The patient developed recurrent episodes of UGH syndrome after dislocated IOL removal and the placement of sutureless, scleral-fixated IOL. Gravitationally dependent pupillary capture was noted with the superior iris moving in front of and behind the IOL, depending on head positioning. Ultrasonography showed a floppy iris that moved with shifting gaze. The lack of the capsular bag may have contributed to extreme iris movements. This finding may be secondary to a combination of a lack of zonular support and capsular bag support as well as the lack of vitreous support following vitrectomy. When possible, secondary IOL placement behind a peripherally preserved capsular bag may reduce the risk of UGH.
Full article
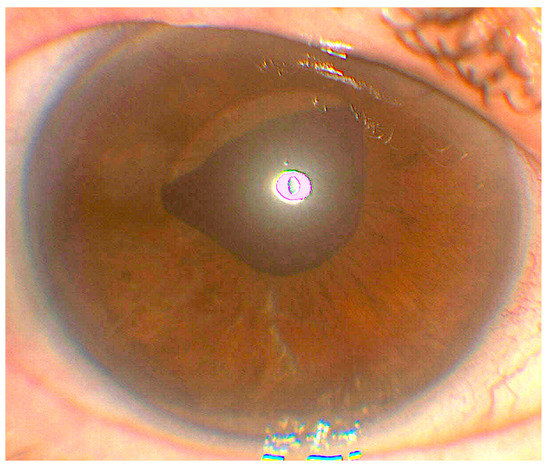
Figure 1
Figure 1
<p>Slit lamp photo demonstrating pupillary capture by the IOL superotemporaly.</p> Full article ">Figure 2
<p>Anterior segment ultrasound biomicroscopy showing the iris and IOL in primary position (<bold>left</bold>) and marked iridodonesis, posterior-iris-bowing, and IOL-chafing when the eye moved down (<bold>right</bold>).</p> Full article ">Figure 3
<p>Model demonstrating aqueous flow with eye movement leading to anterior movement of the superior iris with up gaze (<bold>top left</bold>), posterior movement of the superior iris with down gaze (<bold>top right</bold>), and blockage of aqueous flow when the capsular bag is in place (shown in black) behind the IOL (<bold>bottom left</bold>) or in front of the IOL (<bold>bottom right</bold>). Note the haptics of the IOL are not shown.</p> Full article ">
<p>Slit lamp photo demonstrating pupillary capture by the IOL superotemporaly.</p> Full article ">Figure 2
<p>Anterior segment ultrasound biomicroscopy showing the iris and IOL in primary position (<bold>left</bold>) and marked iridodonesis, posterior-iris-bowing, and IOL-chafing when the eye moved down (<bold>right</bold>).</p> Full article ">Figure 3
<p>Model demonstrating aqueous flow with eye movement leading to anterior movement of the superior iris with up gaze (<bold>top left</bold>), posterior movement of the superior iris with down gaze (<bold>top right</bold>), and blockage of aqueous flow when the capsular bag is in place (shown in black) behind the IOL (<bold>bottom left</bold>) or in front of the IOL (<bold>bottom right</bold>). Note the haptics of the IOL are not shown.</p> Full article ">
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Diagnostics, JCTO, JCM, JPM, Tomography
Optical Coherence Tomography (OCT) and OCT Angiography – Recent Advances
Topic Editors: Sumit Randhir Singh, Jay ChhablaniDeadline: 26 May 2025

Conferences
Special Issues
Special Issue in
JCTO
Advancements in Treating Corneal Injuries and Diseases
Guest Editor: Ratnakar TripathiDeadline: 15 May 2025
Special Issue in
JCTO
Retinal Diseases: Recent Advances in Diagnosis and Treatment
Guest Editors: Dario Giorgio, Dario Pasquale MuccioloDeadline: 15 June 2025
Special Issue in
JCTO
Multimodal Ophthalmic Imaging and Therapeutic Advances in Retinal Diseases: Real-World Insights and Clinical Outcomes
Guest Editors: Stefano Mercuri, Fabrizio GiansantiDeadline: 15 June 2025
Special Issue in
JCTO
Augmented and Artificial Intelligence in Ophthalmology
Guest Editors: Edsel B. Ing, Tina Felfeli, David Jose Mathew, Michael BalasDeadline: 30 June 2025




