<p>Forest plot for causal effects of chronic hepatitis B on stroke and its subtypes. AS = any stroke, AIS = any ischemic stroke, CES = cardioembolic stroke, LAS = large-artery atherosclerotic stroke, SVS = small-vessel stroke, IVW = inverse variance weighting, SNP = single nucleotide polymorphism, N = number, OR = odds ratio, CI = confidence interval.</p> Full article ">
-
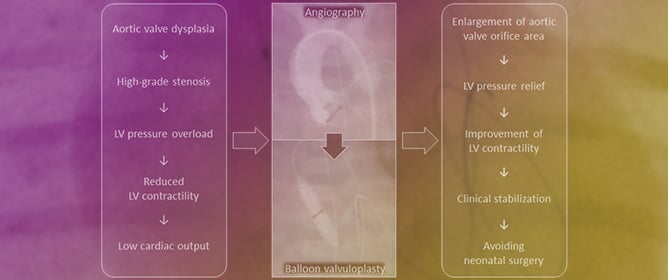 Balloon Valvuloplasty in Congenital Critical Aortic Valve Stenosis in Neonates and Infants: A Rescue Procedure for the Left Ventricle
Balloon Valvuloplasty in Congenital Critical Aortic Valve Stenosis in Neonates and Infants: A Rescue Procedure for the Left Ventricle -
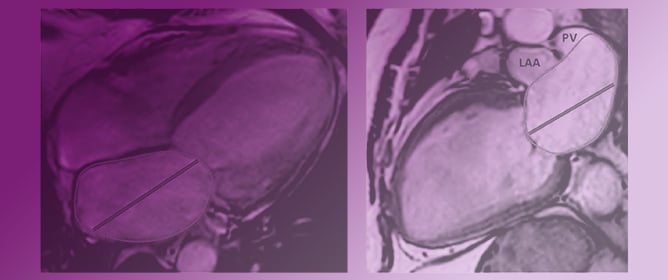 Improved Interpretation of Pulmonary Artery Wedge Pressures through Left Atrial Volumetry—A Cardiac Magnetic Resonance Imaging Study
Improved Interpretation of Pulmonary Artery Wedge Pressures through Left Atrial Volumetry—A Cardiac Magnetic Resonance Imaging Study -
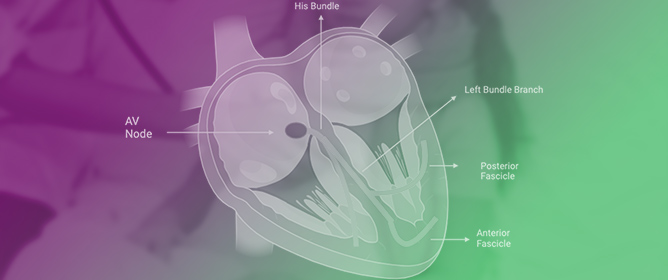 Prevalence and Clinical Significance of Intraventricular Conduction Disturbances in Hospitalized Children
Prevalence and Clinical Significance of Intraventricular Conduction Disturbances in Hospitalized Children -
 Photon-Counting Detector Computed Tomography (PCD-CT): A New Era for Cardiovascular Imaging? Current Status and Future Outlooks
Photon-Counting Detector Computed Tomography (PCD-CT): A New Era for Cardiovascular Imaging? Current Status and Future Outlooks
Journal Description
Journal of Cardiovascular Development and Disease
Journal of Cardiovascular Development and Disease
is an international, scientific, peer-reviewed, open access journal on cardiovascular medicine published monthly online by MDPI.
- Open Access— free for readers, with article processing charges (APC) paid by authors or their institutions.
- High Visibility: indexed within Scopus, SCIE (Web of Science), PubMed, PMC, Embase, CAPlus / SciFinder, and other databases.
- Journal Rank: JCR - Q2 (Cardiac and Cardiovascular Systems) / CiteScore - Q2 (General Pharmacology, Toxicology and Pharmaceutics )
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 22.9 days after submission; acceptance to publication is undertaken in 2.6 days (median values for papers published in this journal in the first half of 2024).
- Recognition of Reviewers: reviewers who provide timely, thorough peer-review reports receive vouchers entitling them to a discount on the APC of their next publication in any MDPI journal, in appreciation of the work done.
Impact Factor:
2.4 (2023);
5-Year Impact Factor:
2.6 (2023)
Latest Articles
Causal Relationship between Chronic Hepatitis B and Stroke in East Asians: A Mendelian Randomization Study
J. Cardiovasc. Dev. Dis. 2024, 11(8), 247; https://doi.org/10.3390/jcdd11080247 (registering DOI) - 10 Aug 2024
Abstract
Both chronic hepatitis B (CHB) and stroke contribute to a high burden of disease in the majority of low- and middle-income countries. Epidemiological studies yield conflicting results on the association between CHB and stroke, and the causal relationship remains inconclusive. This study aimed
[...] Read more.
Both chronic hepatitis B (CHB) and stroke contribute to a high burden of disease in the majority of low- and middle-income countries. Epidemiological studies yield conflicting results on the association between CHB and stroke, and the causal relationship remains inconclusive. This study aimed to assess the causal effects of CHB on stroke and its subtypes in East Asians by Mendelian randomization (MR) analysis. Variants associated with CHB were obtained from a genome-wide association study (GWAS) of Chinese samples as instrumental variables. The summary statistics for stroke in East Asians were derived from the largest published GWAS to date. Two-sample MR analyses were implemented to evaluate the causal effects of CHB on stroke and its subtypes by using the canonical inverse variance weighting method and other supplementary approaches. We observed an association between genetic predisposition to CHB and a decreased risk of large-artery atherosclerotic stroke (odds ratio = 0.872, 95% confidence interval = 0.786–0.967, p = 0.010). The causal effects of CHB on other stroke outcomes were not statistically significant. Evidence for heterogeneity and horizontal pleiotropy were not found in our analyses. This study provides genetic evidence for a negative association between CHB and stroke in East Asians, which helps improve our understanding of the etiology of stroke.
Full article
(This article belongs to the Special Issue Stroke: Risk Factors, Mechanisms, Outcomes and Ethnicity)
►
Show Figures
Figure 1
<p>Forest plot for causal effects of chronic hepatitis B on stroke and its subtypes. AS = any stroke, AIS = any ischemic stroke, CES = cardioembolic stroke, LAS = large-artery atherosclerotic stroke, SVS = small-vessel stroke, IVW = inverse variance weighting, SNP = single nucleotide polymorphism, N = number, OR = odds ratio, CI = confidence interval.</p> Full article ">
<p>Forest plot for causal effects of chronic hepatitis B on stroke and its subtypes. AS = any stroke, AIS = any ischemic stroke, CES = cardioembolic stroke, LAS = large-artery atherosclerotic stroke, SVS = small-vessel stroke, IVW = inverse variance weighting, SNP = single nucleotide polymorphism, N = number, OR = odds ratio, CI = confidence interval.</p> Full article ">
Open AccessArticle
Invasive Mechanical Ventilation Is Associated with Worse Right Ventricular Strain in Acute Respiratory Failure Patients
by
Shuyuan Wang, Zubair Bashir, Edward W. Chen, Vishnu Kadiyala, Charles F. Sherrod, Phinnara Has, Christopher Song, Corey E. Ventetuolo, James Simmons and Philip Haines
J. Cardiovasc. Dev. Dis. 2024, 11(8), 246; https://doi.org/10.3390/jcdd11080246 (registering DOI) - 9 Aug 2024
Abstract
Right ventricular (RV) dysfunction is associated with poor prognosis in acute respiratory failure (ARF). Our study evaluates the efficacy of RV strain in detecting RV dysfunction in ARF patients requiring invasive mechanical ventilation (IMV) compared to tricuspid annular plane systolic excursion (TAPSE). In
[...] Read more.
Right ventricular (RV) dysfunction is associated with poor prognosis in acute respiratory failure (ARF). Our study evaluates the efficacy of RV strain in detecting RV dysfunction in ARF patients requiring invasive mechanical ventilation (IMV) compared to tricuspid annular plane systolic excursion (TAPSE). In this retrospective study involving 376 patients diagnosed with ARF and requiring IMV, we extracted clinical and outcome data from patient records. RV global longitudinal strain (RVGLS), free wall longitudinal strain (FWLS), and TAPSE were measured retrospectively using speckle tracking echocardiography (STE) and traditional echocardiography, respectively. We divided the cohort into three groups: TTE during IMV (TTE-IMV, 223 patients), before IMV (TTE-bIMV, 68 patients), and after IMV (TTE-aIMV, 85 patients). Multivariable regression analysis, adjusted for covariates, revealed significantly higher RVGLS and FWLS in the groups not on IMV at the time of TTE compared to the TTE-IMV group. Specifically, the TTE-bIMV group showed higher RVGLS (β = 7.28, 95% CI 5.07, 9.48) and FWLS (β = 5.83, 95% CI 3.36, 8.31), while the TTE-aIMV group exhibited higher RVGLS (β = 9.39, 95% CI 6.10, 12.69) and FWLS (β = 7.54, 95% CI 4.83, 10.24). TAPSE did not reveal any significant differences across the groups. Our study suggests an association between IMV and lower RVGLS and FWLS in ARF patients, indicating that IMV itself may contribute to RV dysfunction. RVGLS and FWLS appear to be more sensitive than TAPSE in detecting changes in RV function that were previously subclinical in patients on IMV. Prospective studies with TTE before, during, and after IMV are necessary to assess the primary driver of RV dysfunction and to prognosticate STE-detected RV dysfunction in this population.
Full article
(This article belongs to the Special Issue Critical Care in Modern Cardiology)
Open AccessReview
Charting the Unseen: How Non-Invasive Imaging Could Redefine Cardiovascular Prevention
by
Giancarlo Trimarchi, Fausto Pizzino, Umberto Paradossi, Ignazio Alessio Gueli, Matteo Palazzini, Piero Gentile, Francesco Di Spigno, Enrico Ammirati, Andrea Garascia, Andrea Tedeschi and Daniela Aschieri
J. Cardiovasc. Dev. Dis. 2024, 11(8), 245; https://doi.org/10.3390/jcdd11080245 - 9 Aug 2024
Abstract
Cardiovascular diseases (CVDs) remain a major global health challenge, leading to significant morbidity and mortality while straining healthcare systems. Despite progress in medical treatments for CVDs, their increasing prevalence calls for a shift towards more effective prevention strategies. Traditional preventive approaches have centered
[...] Read more.
Cardiovascular diseases (CVDs) remain a major global health challenge, leading to significant morbidity and mortality while straining healthcare systems. Despite progress in medical treatments for CVDs, their increasing prevalence calls for a shift towards more effective prevention strategies. Traditional preventive approaches have centered around lifestyle changes, risk factors management, and medication. However, the integration of imaging methods offers a novel dimension in early disease detection, risk assessment, and ongoing monitoring of at-risk individuals. Imaging techniques such as supra-aortic trunks ultrasound, echocardiography, cardiac magnetic resonance, and coronary computed tomography angiography have broadened our understanding of the anatomical and functional aspects of cardiovascular health. These techniques enable personalized prevention strategies by providing detailed insights into the cardiac and vascular states, significantly enhancing our ability to combat the progression of CVDs. This review focuses on amalgamating current findings, technological innovations, and the impact of integrating advanced imaging modalities into cardiovascular risk prevention, aiming to offer a comprehensive perspective on their potential to transform preventive cardiology.
Full article
(This article belongs to the Special Issue Clinical Burden of Comorbidities on Cardiovascular System and Beyond)
►▼
Show Figures

Figure 1
Figure 1
<p>How imaging redefines cardiovascular prevention.</p> Full article ">Figure 2
<p>Supra-aortic trunks ultrasound: (<b>a</b>) short axis view of internal carotid artery stenosis with calcific plaque; (<b>b</b>) pulsed wave Doppler spectrum of an internal carotid artery with not hemodynamically relevant stenosis; (<b>c</b>) long axis view of a calcific plaque of internal carotid artery; (<b>d</b>) normal pulsed wave Doppler spectrum of a vertebral artery.</p> Full article ">Figure 3
<p>Carotid-femoral pulse wave velocity measurement: (<b>a</b>) normal value of pulse wave velocity in a young man without cardiovascular risk factors; (<b>b</b>) increased pulse wave velocity in a 78-year-old male smoker with hypertension and type 2 diabetes.</p> Full article ">Figure 4
<p>A 65-year-old male smoker with hypertension and dyslipidemia underwent coronary artery calcium (CAC) score calculation for exertional angina. CAC score value is increased in the absence of critic lesions. CAC—coronary artery calcium; CCTA—coronary computed tomography angiography; Cx—circumflex artery; LAD—left anterior descending artery; LM—left main.</p> Full article ">Figure 5
<p>Coronary computed tomography angiography: A 75-year-old female patient with hypertension, diabetes, and dyslipidemia underwent CCTA as a screening procedure before TAVI intervention. The CCTA shows widespread atherosclerotic disease in all three vessels with extensive non-critical calcified lesions. Abbreviations: CCTA—coronary computed tomography angiography; Cx—circumflex artery; LAD—left anterior descending artery; RCA—right coronary artery; TAVI—transcatheter aortic valve implantation.</p> Full article ">Figure 6
<p>Photon-counting detector CT angiography (PCD-CTA) of the heart showing coronary stent implantation in the proximal left anterior descending artery (LAD) (yellow arrow in <b>a</b>–<b>c</b>). Three-dimensional cinematic rendering of the heart, showing the stent in the LAD (<b>a</b>), three-dimensional rendering of the ascending aorta and left coronary artery and the stent in proximal LAD (<b>b</b>), curved multiplanar reformation showing the absence of in-stent stenosis (<b>c</b>).</p> Full article ">Figure 7
<p>Quantification of epicardial fat by cardiac magnetic resonance imaging in a 55-year-old obese, smoking, dyslipidemic male patient with poor echocardiographic acoustic window and symptomatic for exertional angina.</p> Full article ">
<p>How imaging redefines cardiovascular prevention.</p> Full article ">Figure 2
<p>Supra-aortic trunks ultrasound: (<b>a</b>) short axis view of internal carotid artery stenosis with calcific plaque; (<b>b</b>) pulsed wave Doppler spectrum of an internal carotid artery with not hemodynamically relevant stenosis; (<b>c</b>) long axis view of a calcific plaque of internal carotid artery; (<b>d</b>) normal pulsed wave Doppler spectrum of a vertebral artery.</p> Full article ">Figure 3
<p>Carotid-femoral pulse wave velocity measurement: (<b>a</b>) normal value of pulse wave velocity in a young man without cardiovascular risk factors; (<b>b</b>) increased pulse wave velocity in a 78-year-old male smoker with hypertension and type 2 diabetes.</p> Full article ">Figure 4
<p>A 65-year-old male smoker with hypertension and dyslipidemia underwent coronary artery calcium (CAC) score calculation for exertional angina. CAC score value is increased in the absence of critic lesions. CAC—coronary artery calcium; CCTA—coronary computed tomography angiography; Cx—circumflex artery; LAD—left anterior descending artery; LM—left main.</p> Full article ">Figure 5
<p>Coronary computed tomography angiography: A 75-year-old female patient with hypertension, diabetes, and dyslipidemia underwent CCTA as a screening procedure before TAVI intervention. The CCTA shows widespread atherosclerotic disease in all three vessels with extensive non-critical calcified lesions. Abbreviations: CCTA—coronary computed tomography angiography; Cx—circumflex artery; LAD—left anterior descending artery; RCA—right coronary artery; TAVI—transcatheter aortic valve implantation.</p> Full article ">Figure 6
<p>Photon-counting detector CT angiography (PCD-CTA) of the heart showing coronary stent implantation in the proximal left anterior descending artery (LAD) (yellow arrow in <b>a</b>–<b>c</b>). Three-dimensional cinematic rendering of the heart, showing the stent in the LAD (<b>a</b>), three-dimensional rendering of the ascending aorta and left coronary artery and the stent in proximal LAD (<b>b</b>), curved multiplanar reformation showing the absence of in-stent stenosis (<b>c</b>).</p> Full article ">Figure 7
<p>Quantification of epicardial fat by cardiac magnetic resonance imaging in a 55-year-old obese, smoking, dyslipidemic male patient with poor echocardiographic acoustic window and symptomatic for exertional angina.</p> Full article ">
Open AccessArticle
Comparison of Two Generations of Self-Expandable Transcatheter Heart Valves in Nine Surgical Valves: An In Vitro Study
by
Najla Sadat, Michael Scharfschwerdt, Stephan Ensminger and Buntaro Fujita
J. Cardiovasc. Dev. Dis. 2024, 11(8), 244; https://doi.org/10.3390/jcdd11080244 - 8 Aug 2024
Abstract
►▼
Show Figures
(1) Background: This study aimed to analyse the hydrodynamic performance of two generations of self-expanding transcatheter heart valves (THV) as a valve-in-valve (ViV) in different surgical aortic valve (SAV) models under standardised conditions. The nitinol-based Evolut R valve is frequently used in ViV
[...] Read more.
(1) Background: This study aimed to analyse the hydrodynamic performance of two generations of self-expanding transcatheter heart valves (THV) as a valve-in-valve (ViV) in different surgical aortic valve (SAV) models under standardised conditions. The nitinol-based Evolut R valve is frequently used in ViV procedures. It is unclear whether its successor, the Evolut PRO, is superior in ViV procedures, particularly considering the previously implanted SAV model. (2) Methods: EvolutTM R 26 mm and EvolutTM PRO 26 mm prostheses were implanted in nine 21 mm labelled size SAV models (Hancock® II, Mosaic® UltraTM, EpicTM Supra, TrifectaTM GT, Perimount®, Perimount® Magna Ease, AvalusTM, IntuityTM, Freestyle®) to analyse their hydrodynamic performance under defined circulatory conditions in a pulse duplicator. (3) Results: Both THVs presented with the lowest effective orifice area (EOA) and highest mean pressure gradient (MPG) inside Hancock® II, whereas THVs in Intuity showed the highest EOA and lowest MPG. Evolut R and Evolut PRO showed significant hydrodynamic differences depending on the SAV. Both THVs performed similarly in porcine valves. Although the Evolut R performed better than Evolut PRO in stented bovine SAVs, the Evolut PRO was superior inside the Intuity. Further, the SAV model design markedly influenced the TAV’s geometric orifice area and pin-wheeling index. (4) Conclusions: These findings show that the Evolut R and Evolut PRO perform differently depending on the previously implanted SAV model. THV selection for treatment of a specific SAV model should consider these results.
Full article

Figure 1
Figure 1
<p>Images of Evolut R and Evolut PRO as valve-in-valve inside nine surgical aortic bioprostheses models. Stented porcine surgical aortic surgical bioprostheses (Hancock II, Mosaic Ultra and Epic Supra), stented bovine bioprostheses (Trifecta, Perimount, Magna Ease, Avalus and Intuity) and stentless porcine bioprosthesis (Freestyle).</p> Full article ">Figure 2
<p>Fluoroscopic images of Evolut R and Evolut PRO as valve-in-valve inside nine surgical aortic bioprostheses models. Stented porcine surgical aortic surgical bioprostheses (Hancock II, Mosaic Ultra and Epic Supra), stented bovine bioprostheses (Trifecta, Perimount, Magna Ease, Avalus and Intuity) and stentless porcine bioprostheses (Freestyle).</p> Full article ">Figure 3
<p>A custom-made pulse duplicator system according to ISO 5840 regulations for heart valve testing [<a href="#B10-jcdd-11-00244" class="html-bibr">10</a>,<a href="#B11-jcdd-11-00244" class="html-bibr">11</a>].</p> Full article ">Figure 4
<p>The PWI is calculated from the straight distance (L<sub>ideal</sub>) between the valve frame and the coaptation centre (marked in blue) and the traced length (L<sub>actual</sub>) of the leaflet’s free edge between these two points (marked in red).</p> Full article ">Figure 5
<p>Hydrodynamic data I: (<b>A</b>) effective orifice area (EOA) and (<b>B</b>) mean pressure gradient (MPG) of Evolut R vs. Evolut PRO as valve-in-valve. Data are presented as boxplots (median and 25th–75th interquartile; dots indicate outliers) of 180 measurements for each THV model (5 per circulatory condition 50 mL, 70 mL, 90 mL, and 105 mL per 9 surgical valve models).</p> Full article ">Figure 6
<p>Hydrodynamic data II: (<b>A</b>) effective orifice area (EOA) and (<b>B</b>) mean pressure gradient (MPG) of Evolut R vs. Evolut PRO inside specific surgical valve models. Data are presented as boxplots (median and 25th–75th interquartile; dots indicate outliers) of 20 measurements per valve combination (5 per circulatory condition 50 mL, 70 mL, 90 mL, and 105 mL).</p> Full article ">Figure 7
<p>Images of Evolut R and PRO as valve-in-valve inside surgical aortic valve models at maximal opening during perfusion (pulse duplicator testing). The black area with the THVs represents the maximal geometric orifice area. Measurement was performed at a frequency of 64/min and a stroke volume of 70 mL.</p> Full article ">Figure 8
<p>Images of Evolut R and PRO as valve-in-valve inside surgical aortic valve models at closed state during perfusion (pulse duplicator testing). The white lines represent the leaflet folding of the THV as valve-in-valve. Measurement was performed at a frequency of 64/min and a stroke volume of 70 mL.</p> Full article ">
<p>Images of Evolut R and Evolut PRO as valve-in-valve inside nine surgical aortic bioprostheses models. Stented porcine surgical aortic surgical bioprostheses (Hancock II, Mosaic Ultra and Epic Supra), stented bovine bioprostheses (Trifecta, Perimount, Magna Ease, Avalus and Intuity) and stentless porcine bioprosthesis (Freestyle).</p> Full article ">Figure 2
<p>Fluoroscopic images of Evolut R and Evolut PRO as valve-in-valve inside nine surgical aortic bioprostheses models. Stented porcine surgical aortic surgical bioprostheses (Hancock II, Mosaic Ultra and Epic Supra), stented bovine bioprostheses (Trifecta, Perimount, Magna Ease, Avalus and Intuity) and stentless porcine bioprostheses (Freestyle).</p> Full article ">Figure 3
<p>A custom-made pulse duplicator system according to ISO 5840 regulations for heart valve testing [<a href="#B10-jcdd-11-00244" class="html-bibr">10</a>,<a href="#B11-jcdd-11-00244" class="html-bibr">11</a>].</p> Full article ">Figure 4
<p>The PWI is calculated from the straight distance (L<sub>ideal</sub>) between the valve frame and the coaptation centre (marked in blue) and the traced length (L<sub>actual</sub>) of the leaflet’s free edge between these two points (marked in red).</p> Full article ">Figure 5
<p>Hydrodynamic data I: (<b>A</b>) effective orifice area (EOA) and (<b>B</b>) mean pressure gradient (MPG) of Evolut R vs. Evolut PRO as valve-in-valve. Data are presented as boxplots (median and 25th–75th interquartile; dots indicate outliers) of 180 measurements for each THV model (5 per circulatory condition 50 mL, 70 mL, 90 mL, and 105 mL per 9 surgical valve models).</p> Full article ">Figure 6
<p>Hydrodynamic data II: (<b>A</b>) effective orifice area (EOA) and (<b>B</b>) mean pressure gradient (MPG) of Evolut R vs. Evolut PRO inside specific surgical valve models. Data are presented as boxplots (median and 25th–75th interquartile; dots indicate outliers) of 20 measurements per valve combination (5 per circulatory condition 50 mL, 70 mL, 90 mL, and 105 mL).</p> Full article ">Figure 7
<p>Images of Evolut R and PRO as valve-in-valve inside surgical aortic valve models at maximal opening during perfusion (pulse duplicator testing). The black area with the THVs represents the maximal geometric orifice area. Measurement was performed at a frequency of 64/min and a stroke volume of 70 mL.</p> Full article ">Figure 8
<p>Images of Evolut R and PRO as valve-in-valve inside surgical aortic valve models at closed state during perfusion (pulse duplicator testing). The white lines represent the leaflet folding of the THV as valve-in-valve. Measurement was performed at a frequency of 64/min and a stroke volume of 70 mL.</p> Full article ">
Open AccessReview
Antiarrhythmic Drug Use in Pregnancy: Considerations and Safety Profiles
by
Marco Valerio Mariani, Nicola Pierucci, Vincenzo Mirco La Fazia, Pietro Cipollone, Marco Micillo, Andrea D’Amato, Francesca Fanisio, Giuseppe Ammirati, Nethuja Salagundla, Carlo Lavalle and Marco Alfonso Perrone
J. Cardiovasc. Dev. Dis. 2024, 11(8), 243; https://doi.org/10.3390/jcdd11080243 - 7 Aug 2024
Abstract
Pregnancy entails notable physiological alterations and hormonal fluctuations that affect the well-being of both the fetus and the mother. Cardiovascular events and arrhythmias are a major concern during pregnancy, especially in women with comorbidities or a history of arrhythmias. This paper provides an
[...] Read more.
Pregnancy entails notable physiological alterations and hormonal fluctuations that affect the well-being of both the fetus and the mother. Cardiovascular events and arrhythmias are a major concern during pregnancy, especially in women with comorbidities or a history of arrhythmias. This paper provides an overview of the prevalence, therapies, and prognoses of different types of arrhythmias during pregnancy. The administration of antiarrhythmic drugs (AADs) during pregnancy demands careful consideration because of their possible effect on the mother and fetus. AADs can cross the placenta or be present in breast milk, potentially leading to adverse effects such as teratogenicity, growth restriction, or premature birth. The safety profiles of different classes of AADs are discussed. Individualized treatment approaches and close monitoring of pregnant women prescribed AADs are essential to ensure optimal maternal and fetal outcomes.
Full article
(This article belongs to the Section Cardiovascular Clinical Research)
Open AccessArticle
The Great Saphenous Vein Proximal Part: Branches, Anatomical Variations, and Their Implications for Clinical Practice and Venous Reflux Surgery
by
Krisztina Munteanu, Ovidiu Ghirlea, Daniel Breban-Schwarzkopf, Alexandra-Ioana Dănilă, Roxana-Georgeta Iacob, Ioan Adrian Petrache, Gabriel Veniamin Cozma, Anca Bordianu and Sorin Lucian Bolintineanu
J. Cardiovasc. Dev. Dis. 2024, 11(8), 242; https://doi.org/10.3390/jcdd11080242 - 7 Aug 2024
Abstract
The anatomical variations in the lower limb veins play a critical role in venous reflux surgeries. This study presents an analysis of the great saphenous vein (GSV) proximal part’s anatomical peculiarities, with 257 patients included, who were operated for venous reflux. This study
[...] Read more.
The anatomical variations in the lower limb veins play a critical role in venous reflux surgeries. This study presents an analysis of the great saphenous vein (GSV) proximal part’s anatomical peculiarities, with 257 patients included, who were operated for venous reflux. This study highlighted a progressive increase in the GSV diameter in conjunction with the complexity of the anatomical variations, ranging from no tributaries to more than five tributaries, an anterior accessory GSV, or venous aneurysms. Statistical analysis evidenced this expansion to be significantly correlated with the variations. Additionally, the progression of the chronic venous disease (CVD) stages was notably more prevalent in the complex anatomical variations, suggesting a nuanced interplay between the GSV anatomy and CVD severity. Conclusively, our research articulates the paramount importance of recognizing GSV anatomical variations in optimizing surgical outcomes for CVD patients. These insights not only pave the way for enhanced diagnostic accuracy but also support the strategic framework within which surgical and interventional treatments are devised, advocating for personalized approaches to venous reflux surgery.
Full article
Open AccessReview
Current and Clinically Relevant Echocardiographic Parameters to Analyze Left Atrial Function
by
Mario Mangia, Emilio D’Andrea, Antonella Cecchetto, Riccardo Beccari, Donato Mele and Stefano Nistri
J. Cardiovasc. Dev. Dis. 2024, 11(8), 241; https://doi.org/10.3390/jcdd11080241 - 5 Aug 2024
Abstract
The evaluation of the left atrial (LA) size using the LA volume index (LAVI) is clinically relevant due to its prognostic significance in various conditions. Nonetheless, adding a LA function assessment to the LAVI provides further clinical and prognostic information in different cardiovascular
[...] Read more.
The evaluation of the left atrial (LA) size using the LA volume index (LAVI) is clinically relevant due to its prognostic significance in various conditions. Nonetheless, adding a LA function assessment to the LAVI provides further clinical and prognostic information in different cardiovascular (CV) diseases. The assessment of LA function by echocardiography primarily includes volumetric measurements (LA ejection fraction [LAEF]), tissue Doppler imaging (TDI) (mitral annular late diastolic velocity [a’]), and speckle-tracking methods, such as LA longitudinal reservoir strain (LA strain). This review analyzes and discusses the current medical evidence and potential clinical usefulness of these different methods to analyze LA function.
Full article
(This article belongs to the Special Issue Advances in the Diagnosis of Cardiovascular Diseases)
►▼
Show Figures

Figure 1
Figure 1
<p>Left atrial pressure-volume loop, with a qualitative representation of a left atrial (LA) pressure-volume loop and corresponding echocardiographic evaluations of LA volumes. Maximal LA volume (usually reported in clinical echocardiography as LAVI) occurs at the end of the reservoir phase, while the conduit phase can be captured by LA volume pre-A. Minimum LA volume corresponds to the end of the LA contractile phase. Superimposed arrows indicate the time-course.</p> Full article ">Figure 2
<p>Measurement of LA volume and function in a healthy individual. LA maximal and minimal volumes by four-chamber (upper left) and two-chamber (lower left) views in a 30-year-old healthy man. LAVI is 31 mL/m<sup>2</sup> and LAEF 59%. In the right panel, the LA function by biplane STE is depicted: the average LA strain is 43%.</p> Full article ">Figure 3
<p>Measurement of LA volume and function in a patient with heart failure. LA maximal and minimal volumes by four-chamber (upper left) and two-chamber (lower left) views in a 56-year-old male with heart failure and mildly reduced ejection fraction. LAVI is 43 mL/m<sup>2</sup> and LAEF 35%. In the right panel, the LA function by biplane STE is depicted: the average peak LA strain is 19%.</p> Full article ">Figure 4
<p>Age and diastolic velocities by tissue Doppler imaging (TDI). Septal TDI traces measured in three healthy male individuals (aged 17, 33, and 62 years) are reported to show the inverse behaviors of early (e’) and late (a’) diastolic TDI velocities across different ages. At 17 years, TDI-e’ and a’ were16 and 6 cm/s, respectively (left panel); at 33 years (mid panel), TDI-e’ and a’ were 11 and 9 cm/s, respectively; at 62 years (right panel), TDI-e’ and a’ were 8 and 11 cm/s, respectively.</p> Full article ">Figure 5
<p>Measurements of left atrial volume index (LAVI) over TDI-a’. LAVI over TDI-a’, renamed LA volumetric/mechanical coupling index (LACI) by Benfari et al. [<a href="#B136-jcdd-11-00241" class="html-bibr">136</a>], has been shown to mirror the severity of diastolic dysfunction and to correlate with outcomes in different settings. The left panel shows a 58-year-old woman with uncomplicated hypertension, with mildly enlarged LAVI (40 mL/m<sup>2</sup>) and TDI-a’ = 9 cm/s, resulting in LACI = 4.4. The right panel reports a 78-year-old woman with heart failure with preserved ejection fraction due to transthyretin amyloidosis, with severely enlarged LAVI (60 mL/m<sup>2</sup>) and a TDI-a’ = 4 cm/s, reduced in comparison with what is expected for age, resulting in a LACI = 15.</p> Full article ">
<p>Left atrial pressure-volume loop, with a qualitative representation of a left atrial (LA) pressure-volume loop and corresponding echocardiographic evaluations of LA volumes. Maximal LA volume (usually reported in clinical echocardiography as LAVI) occurs at the end of the reservoir phase, while the conduit phase can be captured by LA volume pre-A. Minimum LA volume corresponds to the end of the LA contractile phase. Superimposed arrows indicate the time-course.</p> Full article ">Figure 2
<p>Measurement of LA volume and function in a healthy individual. LA maximal and minimal volumes by four-chamber (upper left) and two-chamber (lower left) views in a 30-year-old healthy man. LAVI is 31 mL/m<sup>2</sup> and LAEF 59%. In the right panel, the LA function by biplane STE is depicted: the average LA strain is 43%.</p> Full article ">Figure 3
<p>Measurement of LA volume and function in a patient with heart failure. LA maximal and minimal volumes by four-chamber (upper left) and two-chamber (lower left) views in a 56-year-old male with heart failure and mildly reduced ejection fraction. LAVI is 43 mL/m<sup>2</sup> and LAEF 35%. In the right panel, the LA function by biplane STE is depicted: the average peak LA strain is 19%.</p> Full article ">Figure 4
<p>Age and diastolic velocities by tissue Doppler imaging (TDI). Septal TDI traces measured in three healthy male individuals (aged 17, 33, and 62 years) are reported to show the inverse behaviors of early (e’) and late (a’) diastolic TDI velocities across different ages. At 17 years, TDI-e’ and a’ were16 and 6 cm/s, respectively (left panel); at 33 years (mid panel), TDI-e’ and a’ were 11 and 9 cm/s, respectively; at 62 years (right panel), TDI-e’ and a’ were 8 and 11 cm/s, respectively.</p> Full article ">Figure 5
<p>Measurements of left atrial volume index (LAVI) over TDI-a’. LAVI over TDI-a’, renamed LA volumetric/mechanical coupling index (LACI) by Benfari et al. [<a href="#B136-jcdd-11-00241" class="html-bibr">136</a>], has been shown to mirror the severity of diastolic dysfunction and to correlate with outcomes in different settings. The left panel shows a 58-year-old woman with uncomplicated hypertension, with mildly enlarged LAVI (40 mL/m<sup>2</sup>) and TDI-a’ = 9 cm/s, resulting in LACI = 4.4. The right panel reports a 78-year-old woman with heart failure with preserved ejection fraction due to transthyretin amyloidosis, with severely enlarged LAVI (60 mL/m<sup>2</sup>) and a TDI-a’ = 4 cm/s, reduced in comparison with what is expected for age, resulting in a LACI = 15.</p> Full article ">
Open AccessArticle
Cardiovascular Risk Factors Predicting Cardiovascular and Cancer Deaths in a Middle-Aged Population Followed-Up for 61 Years until Extinction
by
Alessandro Menotti, Paolo Emilio Puddu and Paolo Piras
J. Cardiovasc. Dev. Dis. 2024, 11(8), 240; https://doi.org/10.3390/jcdd11080240 - 5 Aug 2024
Abstract
Background and Aim. To study the relationships of cardiovascular risk factors with cancer and cardiovascular mortality in a cohort of middle-aged men followed-up for 61 years. Materials and Methods. A rural cohort of 1611 cancer- and cardiovascular disease-free men aged 40–59 years was
[...] Read more.
Background and Aim. To study the relationships of cardiovascular risk factors with cancer and cardiovascular mortality in a cohort of middle-aged men followed-up for 61 years. Materials and Methods. A rural cohort of 1611 cancer- and cardiovascular disease-free men aged 40–59 years was examined in 1960 within the Italian Section of the Seven Countries Study, and 28 risk factors measured at baseline were used to predict cancer (n = 459) and cardiovascular deaths (n = 678) that occurred during 61 years of follow-up until the extinction of the cohort with Cox proportional hazard models. Results. A model with 28 risk factors and cancer deaths as the end-point produced eight statistically significant coefficients for age, smoking habits, mother early death, corneal arcus, xanthelasma and diabetes directly related to events, and arm circumference and healthy diet inversely related. In the corresponding models for major cardiovascular diseases and their subgroups, only the coefficients of age and smoking habits were significant among those found for cancer deaths, to which healthy diet can be added if considering coronary heart disease alone. Following a competing risks analysis by the Fine–Gray method, risk factors significantly common to both conditions were only age, smoking, and xanthelasma. Conclusions. A sizeable number of traditional cardiovascular risk factors were not predictors of cancer death in a middle-aged male cohort followed-up until extinction.
Full article
(This article belongs to the Section Epidemiology, Lifestyle, and Cardiovascular Health)
►▼
Show Figures

Figure 1
Open AccessCase Report
Noninvasive Mapping System for the Stereotactic Radioablation Treatment of Ventricular Tachycardia: A Case Description
by
Imma Romanazzi, Antonio Di Monaco, Ilaria Bonaparte, Noemi Valenti, Alessia Surgo, Fiorella Di Guglielmo, Alba Fiorentino and Massimo Grimaldi
J. Cardiovasc. Dev. Dis. 2024, 11(8), 239; https://doi.org/10.3390/jcdd11080239 - 5 Aug 2024
Abstract
Objectives: Sustained monomorphic ventricular tachycardia (SMVT) is a life-threatening condition that is often observed in patients with structural heart disease. Catheter ablation (CA) ablation is an effective and well-established treatment for the scar-related ventricular tachycardias (VTs). Sometimes, due to patient fragility or contraindications
[...] Read more.
Objectives: Sustained monomorphic ventricular tachycardia (SMVT) is a life-threatening condition that is often observed in patients with structural heart disease. Catheter ablation (CA) ablation is an effective and well-established treatment for the scar-related ventricular tachycardias (VTs). Sometimes, due to patient fragility or contraindications to CA, a noninvasive procedure is required. In these cases, VT ablation with stereotactic arrhythmia radioablation (STAR) for SMVTs supported by the CardioInsight mapping system seems to be a promising and effective noninvasive approach. Methods and results: We report a case of a 55-year-old male smoker and heavy alcohol consumer who developed ischemic heart disease and frequent refractory SMVT relative to antiarrhythmic drugs. Catheter ablation was not practicable due to the presence of an apical thrombosis in the left ventricle. The CardioInsightTM system (Cardioinsight Technologies Inc., Cleveland, OH, USA) was useful for noninvasively mapping the VTs, identifying two target areas on the septum and anterior wall of the left ventricle. A personalized STAR treatment plan was carefully designed, and it was delivered in a few minutes. During follow-up, a significant reduction in the arrhythmia burden was documented. Conclusions: Stereotactic arrhythmia radioablation supported by the CardioInsight system could be an alternative treatment for VTs when catheter ablation is not possible. Larger studies are needed to investigate this technique.
Full article
(This article belongs to the Special Issue Ventricular Arrhythmias: Epidemiology, Diagnosis and Treatment)
►▼
Show Figures

Figure 1
Figure 1
<p>Electrocardiogram showing the sinus rhythm and signs of myocardial necrosis in anterior–lateral leads.</p> Full article ">Figure 2
<p>Summary of the patient’s medical history.</p> Full article ">Figure 3
<p>Echocardiographic image (parasternal long axis) showing a severely dilated left ventricle with apical thrombosis (red arrows).</p> Full article ">Figure 4
<p>Ventricular tachycardias induced by programmed ventricular stimulation using ICD. The red arrows correspond to the stimulated beats.</p> Full article ">Figure 5
<p>Noninvasive mapping of ventricular tachycardia (VT) using the CardioInsight system. Two VTs were induced and mapped. On the left, the system identified the anterior wall as the site of earliest activation during the VT1 (red area). On the right, the system identified the middle septum as the site of earliest activation during VT2 (red area). The right anterior oblique (RAO) projection is shown for all images.</p> Full article ">Figure 6
<p>Treatment plan for STAR. The area outlined by the red line corresponds to the Clinical target volume (CTV). The area outlined by the blue line corresponds to the Planning target volume (PTV). The area outlined by the pink area corresponds to the heart volume.</p> Full article ">Figure 7
<p>Dose distribution and treatment plan shown in sagittal CT images (<b>left</b>) and coronal plan (<b>right</b>). The areas outlined by the yellow, violet and red lines correspond to the organs at risk (OARs). The area outlined by the pink area corresponds to the heart volume. Colored areas correspond to isodose level of RTplan. Notice that the ICD lead was excluded from treatment (red arrow).</p> Full article ">
<p>Electrocardiogram showing the sinus rhythm and signs of myocardial necrosis in anterior–lateral leads.</p> Full article ">Figure 2
<p>Summary of the patient’s medical history.</p> Full article ">Figure 3
<p>Echocardiographic image (parasternal long axis) showing a severely dilated left ventricle with apical thrombosis (red arrows).</p> Full article ">Figure 4
<p>Ventricular tachycardias induced by programmed ventricular stimulation using ICD. The red arrows correspond to the stimulated beats.</p> Full article ">Figure 5
<p>Noninvasive mapping of ventricular tachycardia (VT) using the CardioInsight system. Two VTs were induced and mapped. On the left, the system identified the anterior wall as the site of earliest activation during the VT1 (red area). On the right, the system identified the middle septum as the site of earliest activation during VT2 (red area). The right anterior oblique (RAO) projection is shown for all images.</p> Full article ">Figure 6
<p>Treatment plan for STAR. The area outlined by the red line corresponds to the Clinical target volume (CTV). The area outlined by the blue line corresponds to the Planning target volume (PTV). The area outlined by the pink area corresponds to the heart volume.</p> Full article ">Figure 7
<p>Dose distribution and treatment plan shown in sagittal CT images (<b>left</b>) and coronal plan (<b>right</b>). The areas outlined by the yellow, violet and red lines correspond to the organs at risk (OARs). The area outlined by the pink area corresponds to the heart volume. Colored areas correspond to isodose level of RTplan. Notice that the ICD lead was excluded from treatment (red arrow).</p> Full article ">
Open AccessReview
Optimization of Hypothermic Protocols for Neurocognitive Preservation in Aortic Arch Surgery: A Literature Review
by
Jordan Llerena-Velastegui, Sebastian Velastegui-Zurita, Kristina Zumbana-Podaneva, Melany Mejia-Mora, Ana Clara Fonseca Souza de Jesus and Pedro Moraes Coelho
J. Cardiovasc. Dev. Dis. 2024, 11(8), 238; https://doi.org/10.3390/jcdd11080238 - 1 Aug 2024
Abstract
Shifts from deep to moderate hypothermic circulatory arrest (HCA) in aortic arch surgery necessitate an examination of their differential impacts on neurocognitive functions, especially structured verbal memory, given its significance for patient recovery and quality of life. This study evaluates and synthesizes evidence
[...] Read more.
Shifts from deep to moderate hypothermic circulatory arrest (HCA) in aortic arch surgery necessitate an examination of their differential impacts on neurocognitive functions, especially structured verbal memory, given its significance for patient recovery and quality of life. This study evaluates and synthesizes evidence on the effects of deep (≤20.0 °C), low-moderate (20.1–24.0 °C), and high-moderate (24.1–28.0 °C) hypothermic temperatures on structured verbal memory preservation and overall cognitive health in patients undergoing aortic arch surgery. We evaluated the latest literature from major medical databases such as PubMed and Scopus, focusing on research from 2020 to 2024, to gather comprehensive insights into the current landscape of temperature management during HCA. This comparative analysis highlights the viability of moderate hypothermia (20.1–28.0 °C), supported by recent trials and observational studies, as a method to achieve comparable neuroprotection with fewer complications than traditional deep hypothermia. Notably, low-moderate and high-moderate temperatures have been shown to support substantial survival rates, with impacts on structured verbal memory preservation that necessitate careful selection based on individual surgical risks and patient profiles. The findings advocate for a nuanced approach to selecting hypothermic protocols in aortic arch surgeries, emphasizing the importance of tailoring temperature management to optimize neurocognitive outcomes and patient recovery. This study fills a critical gap in the literature by providing evidence-based recommendations for temperature ranges during HCA, calling for ongoing updates to clinical guidelines and further research to refine these recommendations. The implications of temperature on survival rates, complications, and success rates underpin the necessity for evolving cardiopulmonary bypass techniques and cerebral perfusion strategies to enhance patient outcomes in complex cardiovascular procedures.
Full article
Open AccessArticle
Long-Term Prognostic Impact of Three-Dimensional Speckle-Tracking Echocardiography-Derived Left Ventricular Global Longitudinal Strain in Healthy Adults—Insights from the MAGYAR-Healthy Study
by
Attila Nemes, Árpád Kormányos, Dorottya Lilla Olajos, Alexandru Achim, Zoltán Ruzsa, Nóra Ambrus and Csaba Lengyel
J. Cardiovasc. Dev. Dis. 2024, 11(8), 237; https://doi.org/10.3390/jcdd11080237 - 31 Jul 2024
Abstract
Introduction. Three-dimensional (3D) speckle-tracking echocardiography (STE) combines the advantages of STE and volumetric 3D echocardiography, which shows the left ventricle (LV) in 3D during the cardiac cycle and is also suitable for accurate strain measurements in addition to volumetric assessments using the same
[...] Read more.
Introduction. Three-dimensional (3D) speckle-tracking echocardiography (STE) combines the advantages of STE and volumetric 3D echocardiography, which shows the left ventricle (LV) in 3D during the cardiac cycle and is also suitable for accurate strain measurements in addition to volumetric assessments using the same virtual 3D LV cast. The present study aimed to confirm the prognostic impact of 3DSTE-derived LV global longitudinal strain (GLS) in healthy adults during a 12-year follow-up period. Patients and methods. The current study comprised 124 healthy individuals with a mean age of 31.0 ± 11.7 years (64 males) at the time of complete two-dimensional Doppler echocardiography (2DE) and 3DSTE. Results. During a mean follow-up of 8.01 ± 4.12 years, 10 healthy individuals suffered cardiovascular events, including 2 cardiac deaths. Using ROC analysis, 3DSTE-derived LV-GLS ≥ 14.77% was found to be a significant predictor for cardiovascular event-free survival (sensitivity 70%, specificity 71%, area under the curve 76%, p = 0.007). Using 2DE, higher LV end-diastolic and end-systolic volumes, a larger LV end-systolic diameter and a lower LV ejection fraction could be detected in subjects with LV-GLS < 14.77% as compared to cases with LV-GLS ≥ 14.77%. Subjects with events had thicker interventricular septa, a larger LV mass and lower 3DSTE-derived LV-GLS and a higher ratio of cases had LV-GLS < 14.77%. From subjects with LV-GLS < 14.77%, seven individuals (18%) had events. Multivariate regression analysis identified age and LV-GLS as independent predictors of event-free survival. Conclusions. 3DSTE-derived LV-GLS is a strong independent predictor of cardiovascular survival in healthy adults.
Full article
(This article belongs to the Section Imaging)
►▼
Show Figures

Figure 1
Figure 1
<p>Analysis of the left ventricle (LV) from a three-dimensional (3D) speckle-tracking echocardiographic dataset: (A) apical four-chamber view, (B) apical two-chamber view and (C3) apical, (C5) midventricular and (C7) basal LV short-axis views. A virtual 3D cast of the LV (red D), LV volumetric data during the cardiac cycle (red E), time—LV global longitudinal strain (GLS, white line) and time—LV volume change (dashed white line) curves during the cardiac cycle (red F) are presented in a healthy subject. Abbreviations. LV = left ventricle, LA = left atrium, RV = right ventricle, RA = right atrium.</p> Full article ">Figure 2
<p>Receiver operating characteristic analysis illustrating the diagnostic accuracy of three-dimensional speckle-tracking echocardiography-derived left ventricular global longitudinal strain (GLS) in predicting cardiovascular morbidity and mortality.</p> Full article ">Figure 3
<p>Kaplan–Meier survival curves illustrating the predictive role of three-dimensional speckle-tracking echocardiography-derived left ventricular global longitudinal strain (GLS).</p> Full article ">
<p>Analysis of the left ventricle (LV) from a three-dimensional (3D) speckle-tracking echocardiographic dataset: (A) apical four-chamber view, (B) apical two-chamber view and (C3) apical, (C5) midventricular and (C7) basal LV short-axis views. A virtual 3D cast of the LV (red D), LV volumetric data during the cardiac cycle (red E), time—LV global longitudinal strain (GLS, white line) and time—LV volume change (dashed white line) curves during the cardiac cycle (red F) are presented in a healthy subject. Abbreviations. LV = left ventricle, LA = left atrium, RV = right ventricle, RA = right atrium.</p> Full article ">Figure 2
<p>Receiver operating characteristic analysis illustrating the diagnostic accuracy of three-dimensional speckle-tracking echocardiography-derived left ventricular global longitudinal strain (GLS) in predicting cardiovascular morbidity and mortality.</p> Full article ">Figure 3
<p>Kaplan–Meier survival curves illustrating the predictive role of three-dimensional speckle-tracking echocardiography-derived left ventricular global longitudinal strain (GLS).</p> Full article ">
Open AccessArticle
Compromised Cerebral Arterial Perfusion, Altered Brain Tissue Integrity, and Cognitive Impairment in Adolescents with Complex Congenital Heart Disease
by
Nancy A. Pike, Bhaswati Roy, Cristina Cabrera-Mino, Nancy J. Halnon, Alan B. Lewis, Xingfeng Shao, Danny J. J. Wang and Rajesh Kumar
J. Cardiovasc. Dev. Dis. 2024, 11(8), 236; https://doi.org/10.3390/jcdd11080236 - 29 Jul 2024
Abstract
(1) Introduction: Adolescents with complex congenital heart disease (CCHD) show brain tissue injuries in regions associated with cognitive deficits. Alteration in cerebral arterial perfusion (CAP), as measured by arterial transit time (ATT), may lead to perfusion deficits and potential injury. Our study aims
[...] Read more.
(1) Introduction: Adolescents with complex congenital heart disease (CCHD) show brain tissue injuries in regions associated with cognitive deficits. Alteration in cerebral arterial perfusion (CAP), as measured by arterial transit time (ATT), may lead to perfusion deficits and potential injury. Our study aims to compare ATT values between CCHD patients and controls and assess the associations between ATT values, MD values, and cognitive scores in adolescents with CCHD. (2) Methods: 37 CCHD subjects, 14–18 years of age, who had undergone surgical palliation and 30 healthy controls completed cognitive testing and brain MRI assessments using a 3.0-Tesla scanner. ATT values and regional brain mean diffusivity [MD] were assessed for the whole brain using diffusion tensor imaging. (3) Results: The mean MoCA values [23.1 ± 4.1 vs. 28.1 ± 2.3; p < 0.001] and General Memory Index, with a subscore of WRAML2 [86.8 ± 15.4 vs. 110.3 ± 14.5; p < 0.001], showed significant cognitive deficits in CCHD patients compared to controls. The mean global ATT was significantly higher in CCHD patients versus controls (mean ± SD, s, 1.26 ± 0.11 vs. 1.19 ± 0.11, p = 0.03), respectively. The partial correlations between ATT values, MD values, and cognitive scores (p < 0.005) showed significant associations in areas including the hippocampus, prefrontal cortices, cerebellum, caudate, anterior and mid cingulate, insula, thalamus, and lingual gyrus. (4) Conclusions: Adolescents with CCHD had prolonged ATTs and showed correlation with clinical measurements of cognitive impairment and MRI measurements of brain tissue integrity. This suggests that altered CAP may play a role in brain tissue injury and cognitive impairment after surgical palliation.
Full article
(This article belongs to the Section Pediatric Cardiology and Congenital Heart Disease)
►▼
Show Figures

Figure 1
Figure 1
<p>Arterial transit time (ATT) values and cognitive scores. Negative correlations emerged between ATT, MoCA, subscores, WRAML2, and the subscales of CCHD subjects. Negative correlations appeared between ATT and cognition scores at the bilateral cerebellum (a, b), prefrontal cortices (c), bilateral occipital cortices (d) in the MoCA subscale. These were also found for the right cerebellum (e), bilateral occipital (f, g), bilateral mid-brain (h, i), bilateral cerebellar tonsil (j, k), right cerebellum (l), and pons (m) when examining WRAML2 subscores. All images are given according to neurological convention (L = left; R = right). The color bar indicates t-statistic values.</p> Full article ">Figure 2
<p>Mean diffusivity (MD) values and cognitive scores. Cognition showed negative associations with MD values in CCHD subjects in several brain sites. Negative correlations appeared between MD and MoCA and subscores at the bilateral prefrontal cortices (a, d, i, l, s), bilateral insula (b, c, j, k), anterior (m, p, r), mid (n) cingulate, and the caudate (o). These were also found for WRAML2 subscales, the hippocampus (e, f, q), prefrontal cortices (g), and the caudate (h, l). All images are presented according to neurological convention (L = left; R = right). The color bar indicates t-statistic values.</p> Full article ">
<p>Arterial transit time (ATT) values and cognitive scores. Negative correlations emerged between ATT, MoCA, subscores, WRAML2, and the subscales of CCHD subjects. Negative correlations appeared between ATT and cognition scores at the bilateral cerebellum (a, b), prefrontal cortices (c), bilateral occipital cortices (d) in the MoCA subscale. These were also found for the right cerebellum (e), bilateral occipital (f, g), bilateral mid-brain (h, i), bilateral cerebellar tonsil (j, k), right cerebellum (l), and pons (m) when examining WRAML2 subscores. All images are given according to neurological convention (L = left; R = right). The color bar indicates t-statistic values.</p> Full article ">Figure 2
<p>Mean diffusivity (MD) values and cognitive scores. Cognition showed negative associations with MD values in CCHD subjects in several brain sites. Negative correlations appeared between MD and MoCA and subscores at the bilateral prefrontal cortices (a, d, i, l, s), bilateral insula (b, c, j, k), anterior (m, p, r), mid (n) cingulate, and the caudate (o). These were also found for WRAML2 subscales, the hippocampus (e, f, q), prefrontal cortices (g), and the caudate (h, l). All images are presented according to neurological convention (L = left; R = right). The color bar indicates t-statistic values.</p> Full article ">
Open AccessReview
Revolutionizing Donor Heart Procurement: Innovations and Future Directions for Enhanced Transplantation Outcomes
by
Marc Leon
J. Cardiovasc. Dev. Dis. 2024, 11(8), 235; https://doi.org/10.3390/jcdd11080235 - 27 Jul 2024
Abstract
►▼
Show Figures
Heart failure persists as a critical public health challenge, with heart transplantation esteemed as the optimal treatment for patients with end-stage heart failure. However, the limited availability of donor hearts presents a major obstacle to meeting patient needs. In recent years, the most
[...] Read more.
Heart failure persists as a critical public health challenge, with heart transplantation esteemed as the optimal treatment for patients with end-stage heart failure. However, the limited availability of donor hearts presents a major obstacle to meeting patient needs. In recent years, the most groundbreaking progress in heart transplantation has been in donor heart procurement, significantly expanding the donor pool and enhancing clinical outcomes. This review comprehensively examines these advancements, including the resurgence of heart donation after circulatory death and innovative recovery and evaluation technologies such as normothermic machine perfusion and thoraco-abdominal normothermic regional perfusion. Additionally, novel preservation methods, including controlled hypothermic preservation and hypothermic oxygenated perfusion, are evaluated. The review also explores the use of extended-criteria donors, post-cardiopulmonary resuscitation donors, and high-risk donors, all contributing to increased donor availability without compromising outcomes. Future directions, such as xenotransplantation, biomarkers, and artificial intelligence in donor heart evaluation and procurement, are discussed. These innovations promise to address current limitations and optimize donor heart utilization, ultimately enhancing transplantation success. By identifying recent advancements and proposing future research directions, this review aims to provide insights into advancing heart transplantation and improving patient outcomes.
Full article

Figure 1
Figure 1
<p>TransMedics OCS Heart System. (<b>A</b>). TransMedics OCS device, front view with the cover removed. (<b>B</b>). Schematic diagram of the OCS working principle. The OCS circulates warmed, oxygenated perfusate through the heart preservation module circuit. Blood, supplemented with TransMedics solutions, is pumped from the reservoir through an oxygenator and warmer. This warm, oxygenated blood is directed to the aorta to perfuse the coronary arteries. Deoxygenated blood returns from the coronary circulation to the right atrium, passes through the tricuspid valve to the right ventricle, and is ejected through the pulmonary artery back to the reservoir for recirculation. CF: coronary flow; PA: pulmonary artery; SvO<sub>2</sub>: mixed venous oxygen saturation. The images are sourced from publicly available materials and are used with permission from TransMedics, Inc.</p> Full article ">Figure 2
<p>Paragonix SherpaPak Cardiac Transport System. (<b>A</b>). Exploded view of the SCTS, demonstrating its components and assembly process. (<b>B</b>). Cross-sectional view of the assembled SCTS, showing a preserved heart within the system. The SCTS employs controlled hypothermic preservation to maintain donor heart viability. The system ensures stable temperatures between 4 °C and 8 °C, minimizing myocardial damage during transport. The images are sourced from publicly available materials and are used with permission from Paragonix Technologies, Inc.</p> Full article ">Figure 3
<p>XVIVO Heart Perfusion System. (<b>A</b>). Side view with the cover. (<b>B</b>). Top view without the cover. The XVIVO Heart Perfusion System is designed for hypothermic oxygenated perfusion, providing continuous oxygen and nutrient supply to the donor heart. The images are used with permission from XVIVO Inc.</p> Full article ">
<p>TransMedics OCS Heart System. (<b>A</b>). TransMedics OCS device, front view with the cover removed. (<b>B</b>). Schematic diagram of the OCS working principle. The OCS circulates warmed, oxygenated perfusate through the heart preservation module circuit. Blood, supplemented with TransMedics solutions, is pumped from the reservoir through an oxygenator and warmer. This warm, oxygenated blood is directed to the aorta to perfuse the coronary arteries. Deoxygenated blood returns from the coronary circulation to the right atrium, passes through the tricuspid valve to the right ventricle, and is ejected through the pulmonary artery back to the reservoir for recirculation. CF: coronary flow; PA: pulmonary artery; SvO<sub>2</sub>: mixed venous oxygen saturation. The images are sourced from publicly available materials and are used with permission from TransMedics, Inc.</p> Full article ">Figure 2
<p>Paragonix SherpaPak Cardiac Transport System. (<b>A</b>). Exploded view of the SCTS, demonstrating its components and assembly process. (<b>B</b>). Cross-sectional view of the assembled SCTS, showing a preserved heart within the system. The SCTS employs controlled hypothermic preservation to maintain donor heart viability. The system ensures stable temperatures between 4 °C and 8 °C, minimizing myocardial damage during transport. The images are sourced from publicly available materials and are used with permission from Paragonix Technologies, Inc.</p> Full article ">Figure 3
<p>XVIVO Heart Perfusion System. (<b>A</b>). Side view with the cover. (<b>B</b>). Top view without the cover. The XVIVO Heart Perfusion System is designed for hypothermic oxygenated perfusion, providing continuous oxygen and nutrient supply to the donor heart. The images are used with permission from XVIVO Inc.</p> Full article ">
Open AccessReview
A Comprehensive Review of Percutaneous and Surgical Left Atrial Appendage Occlusion
by
Michał Święczkowski, Emil Julian Dąbrowski, Paweł Muszyński, Piotr Pogorzelski, Piotr Jemielita, Joanna Maria Dudzik, Tomasz Januszko, Małgorzata Duzinkiewicz, Maciej Południewski, Łukasz Kuźma, Marcin Kożuch, Paweł Kralisz and Sławomir Dobrzycki
J. Cardiovasc. Dev. Dis. 2024, 11(8), 234; https://doi.org/10.3390/jcdd11080234 - 27 Jul 2024
Abstract
Atrial fibrillation (AF) is the most common arrhythmia worldwide, and is associated with a significant risk of thromboembolic events. Left atrial appendage occlusion (LAAO) has emerged as a promising alternative for patients with contraindications or intolerance to anticoagulant therapy. This review summarises the
[...] Read more.
Atrial fibrillation (AF) is the most common arrhythmia worldwide, and is associated with a significant risk of thromboembolic events. Left atrial appendage occlusion (LAAO) has emerged as a promising alternative for patients with contraindications or intolerance to anticoagulant therapy. This review summarises the current evidence, indications, and technical advancements in surgical and percutaneous LAAO. Preprocedural planning relies on various imaging techniques, each with unique advantages and limitations. The existing randomised clinical trials and meta-analyses demonstrate favourable results for both percutaneous and surgical LAAO. Postprocedural management emphasises personalised anticoagulation strategies and comprehensive imaging surveillance to ensure device stability and detect complications. Future focus should be put on antithrombotic regimens, investigating predictors of device-related complications, and simplifying procedural aspects to enhance patient outcomes. In summary, LAAO is presented as a valuable therapeutic option for preventing AF-related thromboembolic events, with ongoing research aimed at refining techniques and improving patient care.
Full article
(This article belongs to the Special Issue Feature Review Papers in Stroke and Cerebrovascular Disease)
►▼
Show Figures

Figure 1
Figure 1
<p>Imaging methods. (<b>A</b>) TOE—2D image. LAA with a thrombus (blue arrow). (<b>B</b>) TOE—3D image. LAA with a thrombus (blue arrow). (<b>C</b>) Intraoperative DSA. LAA occluder (red arrow) and TOE probe (blue arrow). (<b>D</b>) Follow-up CCT. LAA occluder (red arrows). (<b>E</b>) Follow-up TOE—2D image. LAA occluder (red arrow). (<b>F</b>) Follow-up TOE—3D image. LAA occluder (red arrow). Abbreviations: CCT—computer tomography; DSA—digital subtraction angiography; LAA—left atrial appendage; TOE—transoesophageal echocardiography.</p> Full article ">
<p>Imaging methods. (<b>A</b>) TOE—2D image. LAA with a thrombus (blue arrow). (<b>B</b>) TOE—3D image. LAA with a thrombus (blue arrow). (<b>C</b>) Intraoperative DSA. LAA occluder (red arrow) and TOE probe (blue arrow). (<b>D</b>) Follow-up CCT. LAA occluder (red arrows). (<b>E</b>) Follow-up TOE—2D image. LAA occluder (red arrow). (<b>F</b>) Follow-up TOE—3D image. LAA occluder (red arrow). Abbreviations: CCT—computer tomography; DSA—digital subtraction angiography; LAA—left atrial appendage; TOE—transoesophageal echocardiography.</p> Full article ">
Open AccessReview
Cardiovascular Sequelae of Bronchopulmonary Dysplasia in Preterm Neonates Born before 32 Weeks of Gestational Age: Impact of Associated Pulmonary and Systemic Hypertension
by
Pramod Pharande, Arvind Sehgal and Samuel Menahem
J. Cardiovasc. Dev. Dis. 2024, 11(8), 233; https://doi.org/10.3390/jcdd11080233 - 26 Jul 2024
Abstract
Bronchopulmonary dysplasia (BPD) remains the most common respiratory disorder of prematurity for infants born before 32 weeks of gestational age (GA). Early and prolonged exposure to chronic hypoxia and inflammation induces pulmonary hypertension (PH) with the characteristic features of a reduced number and
[...] Read more.
Bronchopulmonary dysplasia (BPD) remains the most common respiratory disorder of prematurity for infants born before 32 weeks of gestational age (GA). Early and prolonged exposure to chronic hypoxia and inflammation induces pulmonary hypertension (PH) with the characteristic features of a reduced number and increased muscularisation of the pulmonary arteries resulting in an increase in the pulmonary vascular resistance (PVR) and a fall in their compliance. BPD and BPD-associated pulmonary hypertension (BPD-PH) together with systemic hypertension (sHTN) are chronic cardiopulmonary disorders which result in an increased mortality and long-term problems for these infants. Previous studies have predominantly focused on the pulmonary circulation (right ventricle and its function) and developing management strategies accordingly for BPD-PH. However, recent work has drawn attention to the importance of the left-sided cardiac function and its impact on BPD in a subset of infants arising from a unique pathophysiology termed postcapillary PH. BPD infants may have a mechanistic link arising from chronic inflammation, cytokines, oxidative stress, catecholamines, and renin–angiotensin system activation along with systemic arterial stiffness, all of which contribute to the development of BPD-sHTN. The focus for the treatment of BPD-PH has been improvement of the right heart function through pulmonary vasodilators. BPD-sHTN and a subset of postcapillary PH may benefit from afterload reducing agents such as angiotensin converting enzyme inhibitors. Preterm infants with BPD-PH are at risk of later cardiac and respiratory morbidities as young adults. This paper reviews the current knowledge of the pathophysiology, diagnosis, and treatment of BPD-PH and BPD-sHTN. Current knowledge gaps and emerging new therapies will also be discussed.
Full article
(This article belongs to the Special Issue Echocardiography in Pediatric Heart Disease)
►▼
Show Figures

Figure 1
Figure 1
<p>Continuous Doppler trace showing tricuspid regurgitant jet velocity (TRJV) Systolic pulmonary artery pressure estimated using the Bernoulli equation = 84 mmHg [PA pressure = 4 × (TRJV)<sup>2</sup> + right atrial pressure (5 mmHg)].</p> Full article ">Figure 2
<p>Apical 4-chamber view showing the dilated right atrium (RA) and right ventricle (RV).</p> Full article ">Figure 3
<p>Short axis view of left ventricle showing flattening of the interventricular septum and high systolic eccentricity index (D1/D2) Eccentricity index in systole = 2 [D1(3.9 cm)/D2(1.9 cm) = 2, normal 1.2].</p> Full article ">Figure 4
<p>Pulmonary artery pulse Doppler trace showing a shortened time to peak velocity (TPV)/right ventricular ejection time ratio (RVET) TPV/RVET = 0.1 [TPV(0.04 s)/RVET(0.42 s) = 0.1, normal ≥ 0.31].</p> Full article ">
<p>Continuous Doppler trace showing tricuspid regurgitant jet velocity (TRJV) Systolic pulmonary artery pressure estimated using the Bernoulli equation = 84 mmHg [PA pressure = 4 × (TRJV)<sup>2</sup> + right atrial pressure (5 mmHg)].</p> Full article ">Figure 2
<p>Apical 4-chamber view showing the dilated right atrium (RA) and right ventricle (RV).</p> Full article ">Figure 3
<p>Short axis view of left ventricle showing flattening of the interventricular septum and high systolic eccentricity index (D1/D2) Eccentricity index in systole = 2 [D1(3.9 cm)/D2(1.9 cm) = 2, normal 1.2].</p> Full article ">Figure 4
<p>Pulmonary artery pulse Doppler trace showing a shortened time to peak velocity (TPV)/right ventricular ejection time ratio (RVET) TPV/RVET = 0.1 [TPV(0.04 s)/RVET(0.42 s) = 0.1, normal ≥ 0.31].</p> Full article ">
Open AccessArticle
Complex Pulmonary Artery Rehabilitation in Children with Alagille Syndrome: An Early Single-Center Experience of a Successful Collaborative Work
by
Farida Karim, Gurumurthy Hiremath, Juan Carlos Samayoa and Sameh M. Said
J. Cardiovasc. Dev. Dis. 2024, 11(8), 232; https://doi.org/10.3390/jcdd11080232 - 25 Jul 2024
Abstract
Objective: In this paper, we share our single-center experience of successful multidisciplinary management of patients with Alagille syndrome. In addition, we aim to highlight the need for an Alagille program for effectively managing these patients, in general, and particularly peripheral pulmonary artery stenosis
[...] Read more.
Objective: In this paper, we share our single-center experience of successful multidisciplinary management of patients with Alagille syndrome. In addition, we aim to highlight the need for an Alagille program for effectively managing these patients, in general, and particularly peripheral pulmonary artery stenosis associated with this syndrome. Study Design: This is a retrospective review of six children with Alagille syndrome and advanced liver involvement who underwent pulmonary artery reconstruction between 2021 and 2022. Cardiac diagnosis, co-existing liver disease burdens, management approach, and short-term outcomes were analyzed. Results: All the patients underwent one-stage extensive bilateral branch pulmonary rehabilitation. Concomitant procedures included repair of tetralogy of Fallot in one patient and repair of supravalvar pulmonary artery stenosis in two. One patient had balloon pulmonary branch angioplasty before surgery. In all patients, there was a decrease in right ventricular systolic pressure post-operatively. Three patients underwent liver transplantation for pre-existing liver dysfunction. At a median 3-year follow-up, all the patients were alive with their right ventricular systolic pressure less than half of their systemic systolic pressure. One patient underwent balloon angioplasty due to new and recurrent left pulmonary artery stenosis 13 months after surgery. Conclusion: Pulmonary arteries can be successfully rehabilitated surgically in the presence of complex branch disease. Patients with advanced liver disease can undergo successful complex pulmonary artery reconstruction, which can facilitate their future liver transplantation course. A multidisciplinary team approach is a key for successful management of Alagille patients.
Full article
(This article belongs to the Section Cardiac Surgery)
►▼
Show Figures

Figure 1
Figure 1
<p>(<b>A</b>,<b>B</b>) Intra-operative photos showing (<b>A</b>) the thoroughly dissected lobar and segmental branches of the right and (<b>B</b>) left pulmonary arteries. This is an important step that has to be performed prior to heparinization and initiation of cardiopulmonary bypass to avoid the potential for pulmonary hemorrhage from the parenchymal dissection.</p> Full article ">Figure 2
<p>(<b>A</b>–<b>D</b>) Intra-operative photos showing (<b>A</b>) the main pulmonary artery (PA) and both branches being transected which is an important step to facilitate the dissection and reconstruction. (<b>B</b>) Decellularized pulmonary homograft (H) patch augmentation of the right pulmonary artery is being performed, and the completed reconstruction is shown in (<b>C</b>). The procedure is then repeated on the left side, as shown in (<b>D</b>). LPA: left pulmonary artery; RPA: right pulmonary artery; PA: main pulmonary artery; RV: right ventricle; RA: right atrium, H: homograft.</p> Full article ">Figure 3
<p>(<b>A</b>–<b>D</b>) Intra-operative photos showing the final appearance of the augmented right (<b>A</b>) and left (<b>B</b>) branch pulmonary arteries. Pressure tracings showing (<b>C</b>) pre-cardiopulmonary bypass direct measurement of the right ventricular pressure (yellow tracing) which is suprasystemic, and (<b>D</b>) immediate post-operative right ventricular pressure which is less than 50% of the systemic pressure. All measurements were through a direct right ventricular pressure line that was placed intra-operatively. Ao: ascending aorta; PA: main pulmonary artery; RV: right ventricle; SVC: superior vena cava; RUL: right upper lobar branch; RLL: right lower lobar branch.</p> Full article ">Figure 4
<p>(<b>A</b>–<b>D</b>) Pre-operative pulmonary angiography: anteroposterior (<b>A</b>) and lateral (<b>B</b>) views showing severe central branch pulmonary arteries stenoses and hypoplasia. The left and right main branches measured about 3 mm and 4.5 mm, respectively which were quite small for the patient. Multiple segmental stenoses of the lobar branches can be seen as well (black arrows). Post-operative computerized tomographic 3D reconstruction images (<b>C</b>,<b>D</b>) on the same patient performed 17 months after the pulmonary arterial rehabilitation showing widely patent and much larger central branch pulmonary arteries with minimal residual segmental pulmonary artery branch stenosis. RPA: Right pulmonary artery, LPA: Left pulmonary artery.</p> Full article ">
<p>(<b>A</b>,<b>B</b>) Intra-operative photos showing (<b>A</b>) the thoroughly dissected lobar and segmental branches of the right and (<b>B</b>) left pulmonary arteries. This is an important step that has to be performed prior to heparinization and initiation of cardiopulmonary bypass to avoid the potential for pulmonary hemorrhage from the parenchymal dissection.</p> Full article ">Figure 2
<p>(<b>A</b>–<b>D</b>) Intra-operative photos showing (<b>A</b>) the main pulmonary artery (PA) and both branches being transected which is an important step to facilitate the dissection and reconstruction. (<b>B</b>) Decellularized pulmonary homograft (H) patch augmentation of the right pulmonary artery is being performed, and the completed reconstruction is shown in (<b>C</b>). The procedure is then repeated on the left side, as shown in (<b>D</b>). LPA: left pulmonary artery; RPA: right pulmonary artery; PA: main pulmonary artery; RV: right ventricle; RA: right atrium, H: homograft.</p> Full article ">Figure 3
<p>(<b>A</b>–<b>D</b>) Intra-operative photos showing the final appearance of the augmented right (<b>A</b>) and left (<b>B</b>) branch pulmonary arteries. Pressure tracings showing (<b>C</b>) pre-cardiopulmonary bypass direct measurement of the right ventricular pressure (yellow tracing) which is suprasystemic, and (<b>D</b>) immediate post-operative right ventricular pressure which is less than 50% of the systemic pressure. All measurements were through a direct right ventricular pressure line that was placed intra-operatively. Ao: ascending aorta; PA: main pulmonary artery; RV: right ventricle; SVC: superior vena cava; RUL: right upper lobar branch; RLL: right lower lobar branch.</p> Full article ">Figure 4
<p>(<b>A</b>–<b>D</b>) Pre-operative pulmonary angiography: anteroposterior (<b>A</b>) and lateral (<b>B</b>) views showing severe central branch pulmonary arteries stenoses and hypoplasia. The left and right main branches measured about 3 mm and 4.5 mm, respectively which were quite small for the patient. Multiple segmental stenoses of the lobar branches can be seen as well (black arrows). Post-operative computerized tomographic 3D reconstruction images (<b>C</b>,<b>D</b>) on the same patient performed 17 months after the pulmonary arterial rehabilitation showing widely patent and much larger central branch pulmonary arteries with minimal residual segmental pulmonary artery branch stenosis. RPA: Right pulmonary artery, LPA: Left pulmonary artery.</p> Full article ">
Open AccessReview
A Review Paper on Optical Coherence Tomography Evaluation of Coronary Calcification Pattern: Is It Relevant Today?
by
Horea-Laurentiu Onea, Maria Olinic, Florin-Leontin Lazar, Calin Homorodean, Mihai Claudiu Ober, Mihail Spinu, Alexandru Achim, Dan Alexandru Tataru and Dan Mircea Olinic
J. Cardiovasc. Dev. Dis. 2024, 11(8), 231; https://doi.org/10.3390/jcdd11080231 - 24 Jul 2024
Abstract
The process of coronary calcification represents one of the numerous pathophysiological mechanisms involved in the atherosclerosis continuum. Optical coherence tomography (OCT) represents an ideal imaging modality to assess plaque components, especially calcium. Different calcification patterns have been contemporarily described in both early stages
[...] Read more.
The process of coronary calcification represents one of the numerous pathophysiological mechanisms involved in the atherosclerosis continuum. Optical coherence tomography (OCT) represents an ideal imaging modality to assess plaque components, especially calcium. Different calcification patterns have been contemporarily described in both early stages and advanced atherosclerosis. Microcalcifications and spotty calcifications correlate positively with macrophage burden and inflammatory markers and are more frequently found in the superficial layers of ruptured plaques in acute coronary syndrome patients. More compact, extensive calcification may reflect a later stage of the disease and was traditionally associated with plaque stability. Nevertheless, a small number of culprit coronary lesions demonstrates the presence of dense calcified plaques. The purpose of the current paper is to review the most recent OCT data on coronary calcification and the interrelation between calcification pattern and plaque vulnerability. How different calcified plaques influence treatment strategies and associated prognostic implications is of great interest.
Full article
(This article belongs to the Special Issue Feature Review Papers in Imaging)
►▼
Show Figures

Figure 1
Figure 1
<p>Representative images of each OCT-identified calcification pattern. (<b>A</b>) Microcalcification with calcium arc < 22.5° and (<b>a</b>) calcium length < 1 mm. (<b>B</b>) Spotty calcification with calcium arc < 90° and (<b>b</b>) calcium length < 4 mm. (<b>C</b>) Superficial calcified plates with evidence of a small white thrombus (white arrow). (<b>D</b>) Eruptive calcified nodule, white arrow indicating expulsion of small nodules. (<b>E</b>) Calcified protrusion. OCT: optical coherence tomography.</p> Full article ">Figure 2
<p>Example of intrinsic calcification angle measurement in comparison to calcium arc (<b>a</b>) in case of a spotty calcification (calcium arc < 90°); (<b>b</b>) in case of macrocalcification or irregular calcification, the smallest ICA for every OCT section was considered. ICA: intrinsic calcification angle; OCT: optical coherence tomography.</p> Full article ">Figure 3
<p>OCT-derived calcium parameters predictive of stent underexpansion. OCT: optical coherence tomography.</p> Full article ">
<p>Representative images of each OCT-identified calcification pattern. (<b>A</b>) Microcalcification with calcium arc < 22.5° and (<b>a</b>) calcium length < 1 mm. (<b>B</b>) Spotty calcification with calcium arc < 90° and (<b>b</b>) calcium length < 4 mm. (<b>C</b>) Superficial calcified plates with evidence of a small white thrombus (white arrow). (<b>D</b>) Eruptive calcified nodule, white arrow indicating expulsion of small nodules. (<b>E</b>) Calcified protrusion. OCT: optical coherence tomography.</p> Full article ">Figure 2
<p>Example of intrinsic calcification angle measurement in comparison to calcium arc (<b>a</b>) in case of a spotty calcification (calcium arc < 90°); (<b>b</b>) in case of macrocalcification or irregular calcification, the smallest ICA for every OCT section was considered. ICA: intrinsic calcification angle; OCT: optical coherence tomography.</p> Full article ">Figure 3
<p>OCT-derived calcium parameters predictive of stent underexpansion. OCT: optical coherence tomography.</p> Full article ">
Open AccessArticle
Ablation Parameters Predicting Pulmonary Vein Reconnection after Very High-Power Short-Duration Pulmonary Vein Isolation
by
Márton Boga, Gábor Orbán, Zoltán Salló, Klaudia Vivien Nagy, István Osztheimer, Arnold Béla Ferencz, Ferenc Komlósi, Patrik Tóth, Edit Tanai, Péter Perge, Béla Merkely, László Gellér and Nándor Szegedi
J. Cardiovasc. Dev. Dis. 2024, 11(8), 230; https://doi.org/10.3390/jcdd11080230 - 24 Jul 2024
Abstract
Background: Recurrences due to discontinuity in ablation lines are substantial after pulmonary vein isolation (PVI) with radiofrequency ablation for atrial fibrillation. Data are scarce regarding the durability predictors for very high-power short-duration (vHPSD, 90 W/4 s) ablation. Methods: A total of 20 patients
[...] Read more.
Background: Recurrences due to discontinuity in ablation lines are substantial after pulmonary vein isolation (PVI) with radiofrequency ablation for atrial fibrillation. Data are scarce regarding the durability predictors for very high-power short-duration (vHPSD, 90 W/4 s) ablation. Methods: A total of 20 patients were enrolled, who underwent 90 W PVI and a mandatory remapping procedure at 3 months. First-pass isolation (FPI) gaps, and acute pulmonary vein reconnection (PVR) sites were identified at the index procedure; and chronic PVR sites were identified at the repeated procedure. We analyzed parameters of ablation points (n = 1357), and evaluated their roles in predicting a composite endpoint of FPI gaps, acute and chronic PVR. Results: In total, 45 initial ablation points corresponding to gaps in the ablation lines were analyzed. Parameters associated with gaps were interlesion distance (ILD), baseline generator impedance, mean current, total charge, and loss of catheter–tissue contact. The optimal ILD cut-off for predicting gaps was 3.5 mm anteriorly, and 4 mm posteriorly. Conclusions: Biophysical characteristics dependent on generator impedance could affect the efficacy of vHPSD PVI. The use of smaller ILDs is required for effective and durable PVI with vHPSD compared to the consensus targets with lower power ablation, and lower ILDs for anterior applications seem necessary compared to posterior points.
Full article
(This article belongs to the Special Issue Biophysical- and Engineering-Oriented Approaches to Energy-Based Cardiac Ablation Techniques)
►▼
Show Figures

Figure 1
Figure 1
<p>Graph showing the changes of ablation parameters during the 4-s RF applications with the QMODE+ setting (90 W/4 s, temperature-controlled mode). Yellow: power (W); red: temperature (°C); blue: contact-force (g), dashed lines: target contact-force range (5–40 g); green: impedance (Ω). (<b>A</b>) The target temperature of 55 °C is not reached, stable 90 W of power is delivered for 4 s. (<b>B</b>) The target temperature is reached, power is downregulated to prevent overheating. RF = radiofrequency.</p> Full article ">Figure 2
<p>(<b>A</b>) Electroanatomical map of 90 W pulmonary vein isolation (posteroanterior view). (<b>B</b>) High-density voltage map of a patient with four isolated pulmonary veins at the repeat procedure (posteroanterior view) [<a href="#B9-jcdd-11-00230" class="html-bibr">9</a>]. Red area on right panel: bipolar voltage < 0.5 mV; purple area: bipolar voltage > 0.5 mV.</p> Full article ">Figure 3
<p>Location of gaps on a 16-segment pulmonary vein ostium model. Empty stars indicate gaps in the first-pass circle, dashed stars indicate acute PVR, and bold stars indicate chronic PVR. PVR = pulmonary vein reconnection. Vertical dashed lines indicate the border between anterior/posterior segments.</p> Full article ">Figure 4
<p>Association between variables and gap probability. (<b>A</b>) Bar chart showing the percentage of ablation points at gap sites in the case of anterior location, loss-of-contact, and ILD > 3.5 mm. (<b>B</b>–<b>D</b>) Logistic regression curves of total energy, total charge, and mean current delivered by ablation point. ILD = interlesion distance, LOC = loss-of-contact.</p> Full article ">Figure 5
<p>ROC curves of ILDs for predicting gaps and reconnections in the case all applications (<b>A</b>), anterior (<b>B</b>), and posterior (<b>C</b>) ablation points. AUC = area under curve, CI = confidence interval, ILD = inter-lesion distance, ROC = receiver operating characteristic.</p> Full article ">
<p>Graph showing the changes of ablation parameters during the 4-s RF applications with the QMODE+ setting (90 W/4 s, temperature-controlled mode). Yellow: power (W); red: temperature (°C); blue: contact-force (g), dashed lines: target contact-force range (5–40 g); green: impedance (Ω). (<b>A</b>) The target temperature of 55 °C is not reached, stable 90 W of power is delivered for 4 s. (<b>B</b>) The target temperature is reached, power is downregulated to prevent overheating. RF = radiofrequency.</p> Full article ">Figure 2
<p>(<b>A</b>) Electroanatomical map of 90 W pulmonary vein isolation (posteroanterior view). (<b>B</b>) High-density voltage map of a patient with four isolated pulmonary veins at the repeat procedure (posteroanterior view) [<a href="#B9-jcdd-11-00230" class="html-bibr">9</a>]. Red area on right panel: bipolar voltage < 0.5 mV; purple area: bipolar voltage > 0.5 mV.</p> Full article ">Figure 3
<p>Location of gaps on a 16-segment pulmonary vein ostium model. Empty stars indicate gaps in the first-pass circle, dashed stars indicate acute PVR, and bold stars indicate chronic PVR. PVR = pulmonary vein reconnection. Vertical dashed lines indicate the border between anterior/posterior segments.</p> Full article ">Figure 4
<p>Association between variables and gap probability. (<b>A</b>) Bar chart showing the percentage of ablation points at gap sites in the case of anterior location, loss-of-contact, and ILD > 3.5 mm. (<b>B</b>–<b>D</b>) Logistic regression curves of total energy, total charge, and mean current delivered by ablation point. ILD = interlesion distance, LOC = loss-of-contact.</p> Full article ">Figure 5
<p>ROC curves of ILDs for predicting gaps and reconnections in the case all applications (<b>A</b>), anterior (<b>B</b>), and posterior (<b>C</b>) ablation points. AUC = area under curve, CI = confidence interval, ILD = inter-lesion distance, ROC = receiver operating characteristic.</p> Full article ">
Open AccessArticle
Hemodynamic Evaluation of Coronary Artery Lesions after Kawasaki Disease: Comparison of Fractional Flow Reserve during Cardiac Catheterization with Myocardial Flow Reserve during 13N-Ammonia PET
by
Makoto Watanabe, Ryuji Fukazawa, Tomonari Kiriyama, Shogo Imai, Ryosuke Matsui, Kanae Shimada, Yoshiaki Hashimoto, Koji Hashimoto, Masanori Abe, Mitsuhiro Kamisago and Yasuhiko Itoh
J. Cardiovasc. Dev. Dis. 2024, 11(8), 229; https://doi.org/10.3390/jcdd11080229 - 23 Jul 2024
Abstract
Coronary artery lesions (CALs) after Kawasaki disease present complex coronary hemodynamics. We investigated the relationship between coronary fractional flow reserve (FFR), myocardial flow reserve (MFR), and myocardial blood flow volume fraction (MBF) and their clinical usefulness in CALs after Kawasaki disease. Nineteen patients
[...] Read more.
Coronary artery lesions (CALs) after Kawasaki disease present complex coronary hemodynamics. We investigated the relationship between coronary fractional flow reserve (FFR), myocardial flow reserve (MFR), and myocardial blood flow volume fraction (MBF) and their clinical usefulness in CALs after Kawasaki disease. Nineteen patients (18 men, 1 woman) who underwent cardiac catheterization and 13N-ammonia positron emission tomography, with 24 coronary artery branches, were included. Five branches had inconsistent FFR and MFR values, two had normal FFR but abnormal MFR, and three had abnormal FFR and normal MFR. The abnormal MFR group had significantly higher MBF at rest than the normal group (0.86 ± 0.13 vs. 1.08 ± 0.09, p = 0.001). The abnormal FFR group had significantly lower MBF at adenosine loading than the normal group (2.23 ± 0.23 vs. 1.88 ± 0.29, p = 0.021). The three branches with abnormal FFR only had stenotic lesions, but the MFR may have been normal because blood was supplied by collateral vessels. Combining FFR, MFR, and MBF will enable a more accurate assessment of peripheral coronary circulation and stenotic lesions in CALs and help determine treatment strategy and timing of intervention.
Full article
(This article belongs to the Special Issue Clinical Burden of Comorbidities on Cardiovascular System and Beyond)
►▼
Show Figures

Figure 1
Figure 1
<p>Adenosine-loaded 13N-ammonia PET examination protocol. The same protocol was used for both the resting and loading examinations except for the adenosine administration, and the interval between the resting and loading examinations was 4–5 half-lives. CT: computed tomography, PET: positron emission tomography.</p> Full article ">Figure 2
<p>(<b>A</b>) Comparison of resting MBF in the normal and abnormal FFR groups. There was no significant difference in resting MBF between the normal and abnormal FFR groups. (<b>B</b>) Comparison of stress MBF in the normal and abnormal FFR groups. The abnormal FFR group had significantly lower stress MBF than the normal FFR group. FFR: coronary fractional flow reserve, MBF: myocardial blood flow volume fraction.</p> Full article ">Figure 3
<p>(<b>A</b>) Comparison of resting MBF in the normal and abnormal MFR groups. The rest MBF was significantly higher in the abnormal MFR group than in the normal MFR group. (<b>B</b>) Comparison of stress MBF in the normal and abnormal MFR groups. There was no significant difference in stress MBF between the normal and abnormal MFR groups. MBF: myocardial blood flow volume fraction, MFR: myocardial flow reserve.</p> Full article ">Figure 4
<p>(<b>A</b>) Coronary angiography image of Case T. (<b>B</b>) 13N-ammonia PET image of Case T. Owing to occlusion of the LAD and RCA, only LCX is responsible for coronary circulation. MFR may be low due to high blood flow at rest. LAD: left anterior descending artery, LCX: left circumflex artery, MFR: myocardial blood flow reserve, RCA: right coronary artery, PET: positron emission tomography.</p> Full article ">Figure 5
<p>(<b>A</b>) Coronary angiography image of Case U. (<b>B</b>) 13N-ammonia PET image of Case U. The FFR was normal because of the giant aneurysm, and the MFR was low because the peripheral coronary arteries were already dilated at rest. FFR: coronary fractional flow reserve, MFR: myocardial flow reserve, PET: positron emission tomography.</p> Full article ">Figure 6
<p>(<b>A</b>) Coronary angiography image of Case V. (<b>B</b>) 13N-ammonia PET image of Case V. The FFR was low due to the presence of a stenotic lesion, but it was thought the peripheral circulation was not impaired due to the collateral blood vessels. FFR: coronary fractional flow reserve; PET: positron emission tomography.</p> Full article ">
<p>Adenosine-loaded 13N-ammonia PET examination protocol. The same protocol was used for both the resting and loading examinations except for the adenosine administration, and the interval between the resting and loading examinations was 4–5 half-lives. CT: computed tomography, PET: positron emission tomography.</p> Full article ">Figure 2
<p>(<b>A</b>) Comparison of resting MBF in the normal and abnormal FFR groups. There was no significant difference in resting MBF between the normal and abnormal FFR groups. (<b>B</b>) Comparison of stress MBF in the normal and abnormal FFR groups. The abnormal FFR group had significantly lower stress MBF than the normal FFR group. FFR: coronary fractional flow reserve, MBF: myocardial blood flow volume fraction.</p> Full article ">Figure 3
<p>(<b>A</b>) Comparison of resting MBF in the normal and abnormal MFR groups. The rest MBF was significantly higher in the abnormal MFR group than in the normal MFR group. (<b>B</b>) Comparison of stress MBF in the normal and abnormal MFR groups. There was no significant difference in stress MBF between the normal and abnormal MFR groups. MBF: myocardial blood flow volume fraction, MFR: myocardial flow reserve.</p> Full article ">Figure 4
<p>(<b>A</b>) Coronary angiography image of Case T. (<b>B</b>) 13N-ammonia PET image of Case T. Owing to occlusion of the LAD and RCA, only LCX is responsible for coronary circulation. MFR may be low due to high blood flow at rest. LAD: left anterior descending artery, LCX: left circumflex artery, MFR: myocardial blood flow reserve, RCA: right coronary artery, PET: positron emission tomography.</p> Full article ">Figure 5
<p>(<b>A</b>) Coronary angiography image of Case U. (<b>B</b>) 13N-ammonia PET image of Case U. The FFR was normal because of the giant aneurysm, and the MFR was low because the peripheral coronary arteries were already dilated at rest. FFR: coronary fractional flow reserve, MFR: myocardial flow reserve, PET: positron emission tomography.</p> Full article ">Figure 6
<p>(<b>A</b>) Coronary angiography image of Case V. (<b>B</b>) 13N-ammonia PET image of Case V. The FFR was low due to the presence of a stenotic lesion, but it was thought the peripheral circulation was not impaired due to the collateral blood vessels. FFR: coronary fractional flow reserve; PET: positron emission tomography.</p> Full article ">
Open AccessArticle
Clinical Outcome Using Different Catheter Interventional Treatment Modalities in High-Risk Pulmonary Artery Embolism
by
Luise Antonia Mentzel, Parham Shahidi, Stephan Blazek, Dmitry Sulimov, Holger Thiele and Karl Fengler
J. Cardiovasc. Dev. Dis. 2024, 11(7), 228; https://doi.org/10.3390/jcdd11070228 - 22 Jul 2024
Abstract
Background: For patients with high-risk pulmonary artery embolism (PE), catheter-directed therapies pose a viable alternative treatment option to systemic thrombolysis or anticoagulation. Right now, there are multiple devices available which have been proven to be safe and effective in lower-risk settings. There is,
[...] Read more.
Background: For patients with high-risk pulmonary artery embolism (PE), catheter-directed therapies pose a viable alternative treatment option to systemic thrombolysis or anticoagulation. Right now, there are multiple devices available which have been proven to be safe and effective in lower-risk settings. There is, however, little data comparing their efficacies in high-risk PE. Methods: We performed a retrospective, single-center study on patients with high-risk PE undergoing catheter interventional treatment. Patients receiving large-bore catheter thrombectomy were compared to patients receiving alternative treatment forms. Results: Of the 20 patients included, 9 received large-bore thrombectomy, and 11 received alternative interventional treatments. While the baseline characteristics were comparable between the two groups, periprocedural and in-hospital mortality tended to be significantly lower with large-bore thrombectomy when compared to other treatment forms (0 vs. 55% and 33 vs. 82%, p = 0.07 and 0.01, respectively). Conclusions: In this small, retrospective study, large-bore thrombectomy was associated with lower mortality as compared to alternative treatment forms. Future prospective research is needed to corroborate these findings.
Full article
(This article belongs to the Section Cardiovascular Clinical Research)
►▼
Show Figures

Figure 1
Figure 1
<p>Flowchart depicting patient selection for this study.</p> Full article ">Figure 2
<p>All values are depicted as mean ± standard deviation. (<b>A</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.03; FT: <span class="html-italic">p</span> = 0.05; and nonFT: <span class="html-italic">p</span> = 0.12; (<b>B</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.001; FT: <span class="html-italic">p</span> = 0.01; and nonFT: <span class="html-italic">p</span> = 0.06; (<b>C</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.16; FT <span class="html-italic">p</span> = 0.34; and nonFT: <span class="html-italic">p</span> = 0.17; and (<b>D</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.004; FT <span class="html-italic">p</span> = 0.02; and nonFT: <span class="html-italic">p</span> = 0.03.</p> Full article ">Figure 3
<p><span class="html-italic">p</span>-values for between-group comparison.</p> Full article ">
<p>Flowchart depicting patient selection for this study.</p> Full article ">Figure 2
<p>All values are depicted as mean ± standard deviation. (<b>A</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.03; FT: <span class="html-italic">p</span> = 0.05; and nonFT: <span class="html-italic">p</span> = 0.12; (<b>B</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.001; FT: <span class="html-italic">p</span> = 0.01; and nonFT: <span class="html-italic">p</span> = 0.06; (<b>C</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.16; FT <span class="html-italic">p</span> = 0.34; and nonFT: <span class="html-italic">p</span> = 0.17; and (<b>D</b>) <span class="html-italic">p</span>-values (<span class="html-italic">t</span>-test): total: <span class="html-italic">p</span> = 0.004; FT <span class="html-italic">p</span> = 0.02; and nonFT: <span class="html-italic">p</span> = 0.03.</p> Full article ">Figure 3
<p><span class="html-italic">p</span>-values for between-group comparison.</p> Full article ">

Journal Menu
► ▼ Journal Menu-
- JCDD Home
- Aims & Scope
- Editorial Board
- Reviewer Board
- Topical Advisory Panel
- Instructions for Authors
- Special Issues
- Topics
- Sections
- Article Processing Charge
- Indexing & Archiving
- Editor’s Choice Articles
- Most Cited & Viewed
- Journal Statistics
- Journal History
- Journal Awards
- Conferences
- Editorial Office
Journal Browser
► ▼ Journal BrowserHighly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
JCDD, JCM, Medicina, Membranes, ARM
Extracorporeal Membrane Oxygenation (ECMO)
Topic Editors: Patrick Honore, Isabelle MichauxDeadline: 30 November 2024
Topic in
Antioxidants, Biomolecules, JCDD, Metabolites, Neurology International, Pharmaceutics
Tissue-Specific, Disease-Signatured Macrophages in Control of Redox and Antioxidation in Metabolic Diseases
Topic Editors: Xiangwei Xiao, Yingmei Feng, Zhiyong LeiDeadline: 5 July 2025
Topic in
Antioxidants, Biomedicines, Disabilities, JCDD
Cardiovascular Disease in Special Populations: From Basic Science to Clinical Practice
Topic Editors: Song-Young Park, Gwenael LayecDeadline: 31 December 2025
Topic in
CIMB, IJMS, JCDD, Organoids, Biomedicines
Molecular and Cellular Mechanisms of Heart Disease
Topic Editors: Pasi Tavi, Ebru Arioglu-InanDeadline: 31 December 2026

Conferences
Special Issues
Special Issue in
JCDD
Hypertrophic Cardiomyopathy: Pathogenesis, Diagnosis and Management
Guest Editors: Dan G. Halpern, Óscar Lorenzo GonzálezDeadline: 15 August 2024
Special Issue in
JCDD
Cardiovascular Disease: Risk Factors and Prevention
Guest Editors: Roman Leischik, Markus StraußDeadline: 15 August 2024
Special Issue in
JCDD
Pulmonary Hypertension: Current Status of Diagnosis and Treatment
Guest Editors: Roxana Sulica, Himanshu DeshwalDeadline: 20 August 2024
Special Issue in
JCDD
Stroke: Risk Factors, Mechanisms, Outcomes and Ethnicity
Guest Editor: Narayanaswamy VenketasubramanianDeadline: 30 August 2024



