Chlorofluorocarbon
Appearance

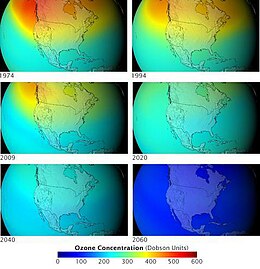
A chlorofluorocarbon (CFC) is a gas used for various purposes including solvents, refrigerants, and aerosol sprays. They are organic chemicals and contain carbon, hydrogen, chlorine, and fluorine. They were much used in the middle of the 20th century, replacing chemicals that were toxic or flammable or had other problems.
In 1978, Sweden became the first country that banned CFC products. Later, the US and Canada did the same. Now, CFC products are not allowed in most countries, because they cause ozone depletion. CFCs are greenhouse gases. An alternative to chlorofluorocarbons is hydrofluorocarbons (HFCs). These do not destroy the ozone layer or increase global warming.[1][2]
Related pages
[change | change source]- Thomas Midgley (American inventor)
- Freon
References
[change | change source]- ↑ John M. Broder (November 9, 2010). "A novel tactic in climate fight". The New York Times. p. A9. Retrieved 2013-02-05.
- ↑ M. Rossberg et al. Chlorinated hydrocarbons in Ullmann's Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a06_233.pub2