WO2025145020A2 - Methods, systems, compounds, and kits for determining an analyte of interest in a test sample - Google Patents
Methods, systems, compounds, and kits for determining an analyte of interest in a test sample Download PDFInfo
- Publication number
- WO2025145020A2 WO2025145020A2 PCT/US2024/062078 US2024062078W WO2025145020A2 WO 2025145020 A2 WO2025145020 A2 WO 2025145020A2 US 2024062078 W US2024062078 W US 2024062078W WO 2025145020 A2 WO2025145020 A2 WO 2025145020A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- formula
- analyte
- acridinium
- so3h
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/543—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals
- G01N33/54313—Immunoassay; Biospecific binding assay; Materials therefor with an insoluble carrier for immobilising immunochemicals the carrier being characterised by its particulate form
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D219/00—Heterocyclic compounds containing acridine or hydrogenated acridine ring systems
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/566—Immunoassay; Biospecific binding assay; Materials therefor using specific carrier or receptor proteins as ligand binding reagents where possible specific carrier or receptor proteins are classified with their target compounds
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/569—Immunoassay; Biospecific binding assay; Materials therefor for microorganisms, e.g. protozoa, bacteria, viruses
- G01N33/56983—Viruses
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/53—Immunoassay; Biospecific binding assay; Materials therefor
- G01N33/576—Immunoassay; Biospecific binding assay; Materials therefor for hepatitis
- G01N33/5761—Hepatitis B
- G01N33/5762—Hepatitis B core antigen
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/005—Assays involving biological materials from specific organisms or of a specific nature from viruses
- G01N2333/01—DNA viruses
- G01N2333/02—Hepadnaviridae, e.g. hepatitis B virus
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/005—Assays involving biological materials from specific organisms or of a specific nature from viruses
- G01N2333/08—RNA viruses
- G01N2333/165—Coronaviridae, e.g. avian infectious bronchitis virus
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2333/00—Assays involving biological materials from specific organisms or of a specific nature
- G01N2333/435—Assays involving biological materials from specific organisms or of a specific nature from animals; from humans
- G01N2333/575—Hormones
- G01N2333/59—Follicle-stimulating hormone [FSH]; Chorionic gonadotropins, e.g. HCG; Luteinising hormone [LH]; Thyroid-stimulating hormone [TSH]
Definitions
- acridinium-containing and acridone-containing compounds for use in assays to detect the presence of or determine the amount or level of an analyte of interest in a biological sample.
- the acridinium-containing and acridone-containing compounds described herein can be used in the methods, systems, and kits for determining the concentration or level of an analyte of interest using a competitive and sandwich immunoassay.
- Immunoassays typically involve combining a sample suspected of containing an antigen or antibody with a reagent comprising a first antibody or first antigen which can be attached to a solid support, e.g., a microparticle, to form a reaction mixture.
- a reagent comprising a first antibody or first antigen which can be attached to a solid support, e.g., a microparticle, to form a reaction mixture.
- the antigen or antibody if present in the sample, specifically binds to the first antibody or first antigen.
- a conjugate which comprises a second antibody or second antigen having a detectable label attached thereto, is introduced to the reaction mixture and specifically binds to the antigen or antibody, which is specifically bound to the first antibody or first antigen, which, as stated previously, can be attached to a solid support.
- Such an assay is referred to as a sandwich immunoassay or an immunometric assay.
- the signal attributable to the detectable label is measured after unbound conjugate is removed from the reaction mixture, typically by performing a wash step.
- the signal that is derived from the total volume of the reaction mixture is measured and compared to a calibration curve to establish the concentration of antigen or antibody present in the sample.
- Immunoassays generally require that the sample containing the analyte of interest be diluted (e.g., pre-dilution) prior to performing the assay. Samples are diluted to lower the concentration of the analyte in the sample to ensure that the amount of analyte is below acceptable limits and to help minimize interferences from other compounds in the sample that could artificially affect the analysis. When samples are not diluted sufficiently enough, the analyte concentration can be underestimated for high analyte positive samples. When samples are over-diluted, low analyte positive samples can be missed as a negative sample.
- diluted e.g., pre-dilution
- An immunoassay that includes a washing step to remove unbound sample analyte before introducing the conjugate antibody is generally referred to as a “two-step assay.”
- An immunoassay that introduces the conjugate antibody and the analyte to antibody-coated microparticles together without intermediate washing steps is generally referred to as a “one- step” assay.
- Both one-step and two-step formats can exhibit limited assay dynamic range, thus some samples require re-testing with dilution.
- a “hook effect” or “prozone phenomenon” is a phenomenon of falsely low values observed in an immunoassay when an overwhelming amount of antigen or antibody is present in a “one-step assay” format. Hook effect is caused by insufficient capture reagent and detection reagent in an immunoassay, which limits the assay dynamic range.
- Acridinium molecules with chemiluminescent properties are useful in the field of in vitro diagnostics (IVDs), where the amount of light produced by acridinium in an assay is proportional to an analyte of interest.
- IVDs in vitro diagnostics
- 9-Carbonylacridiniums are a common type of acridinium used in commercialized chemiluminescence based IVDs, where a phenyl ester or acylsulfonamide moiety at the acridinium 9-position facilitates an efficient reaction pathway for the generation of blue light (ca. 445 nm) upon treatment with an alkaline peroxide solution.
- Substituted acridiniums have previously been prepared through multiple steps beginning with a substituted isatin. This procedure is limited by availability of the isatin and the stability of the substituent to the subsequent reaction conditions. Given the importance of having modified acridiniums for the field of IVDs, improved methods for synthesizing substituted acridiniums are needed. A more preferable method would involve direct modification of an existing commercially available acridinium molecule. Because acridinium is electron deficient and chemically unstable, there are few reactions that can be employed for direct modification.
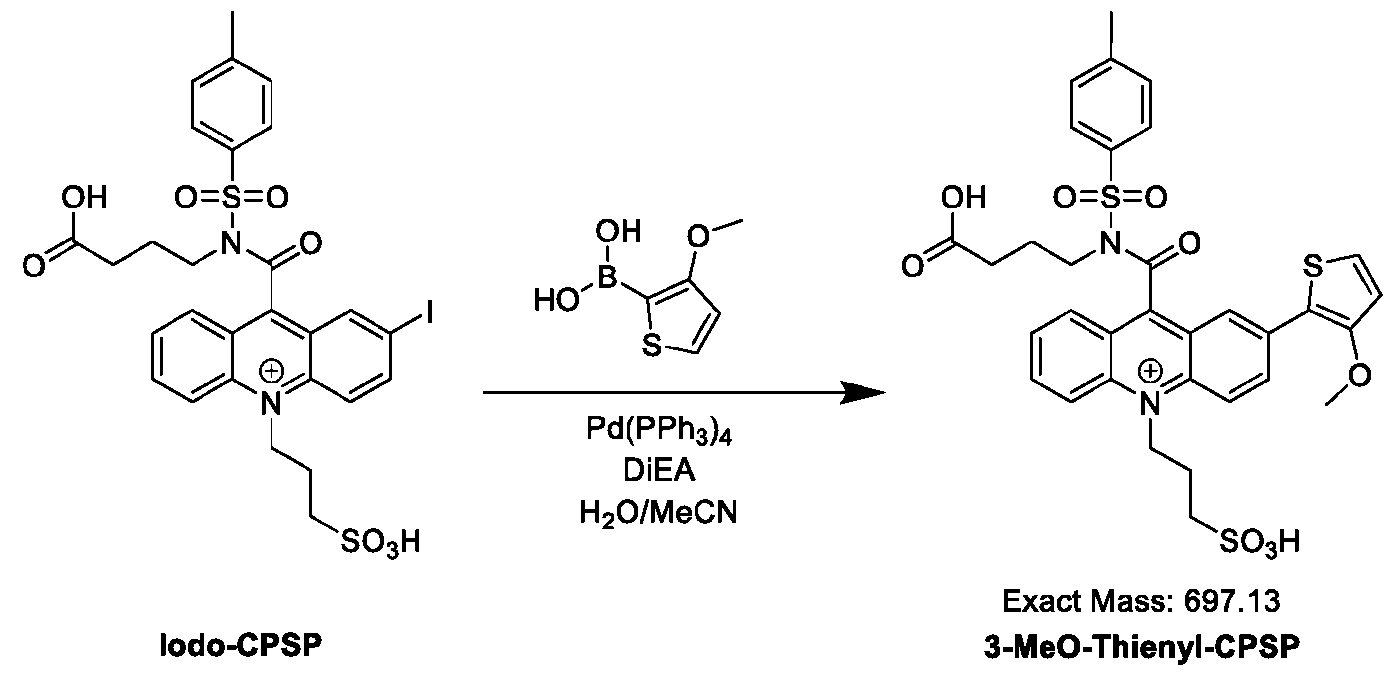
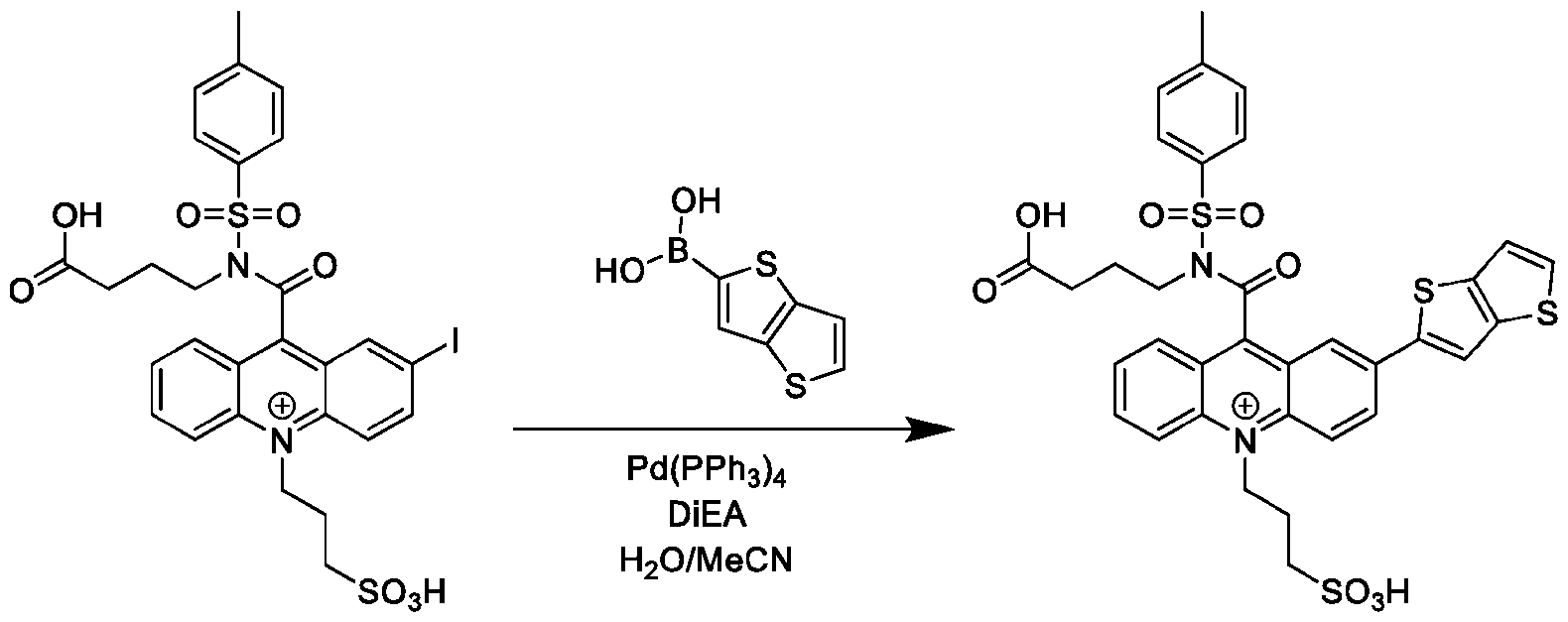
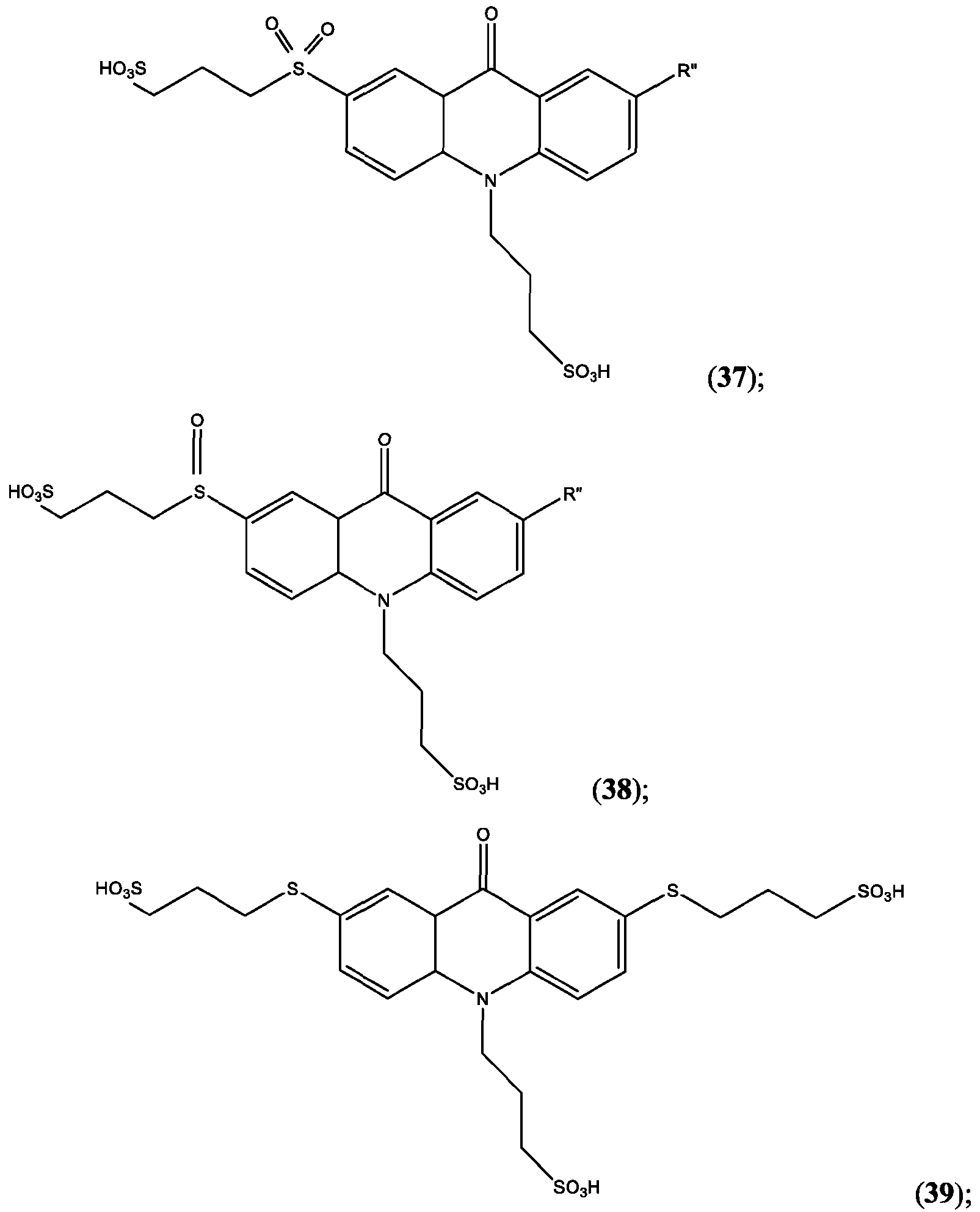
- FIG. 36B is the chemiluminescence spectrum of Thienothiophenyl-CPSP.
- FIG. 37A is the absorbance spectrum of Bis(5-Me-Thienyl)-CPSP.
- FIG. 37B is the chemiluminescence spectrum of Bis(5-Me-Thienyl)-CPSP.
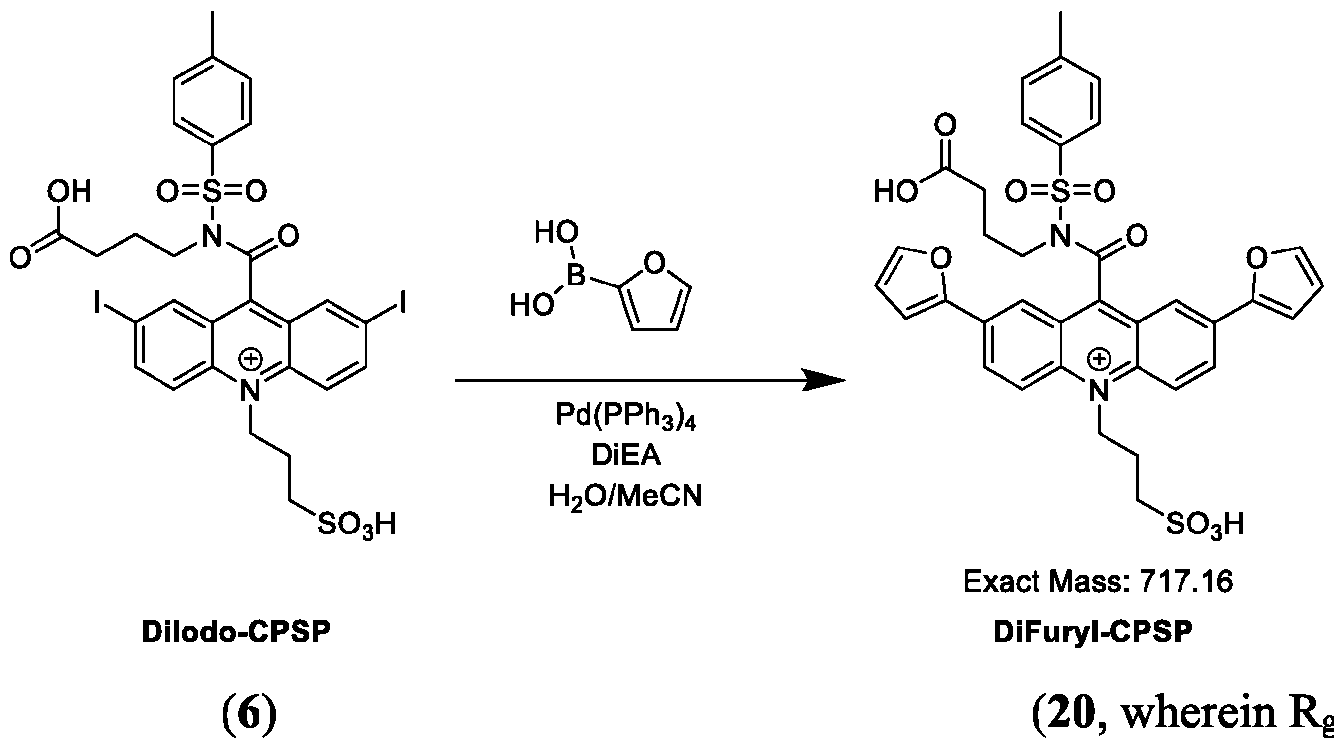
- FIG. 38A is the absorbance spectrum of DiFuranyl-CPSP.
- FIG. 38B is the chemiluminescence spectrum of DiFuranyl-CPSP.
- FIG. 39A is the absorbance spectrum of TAMRA-Ph-CPSP.
- FIG. 39B is the chemiluminescence spectrum of TAMRA-Ph-CPSP.
- CL chemiluminescence
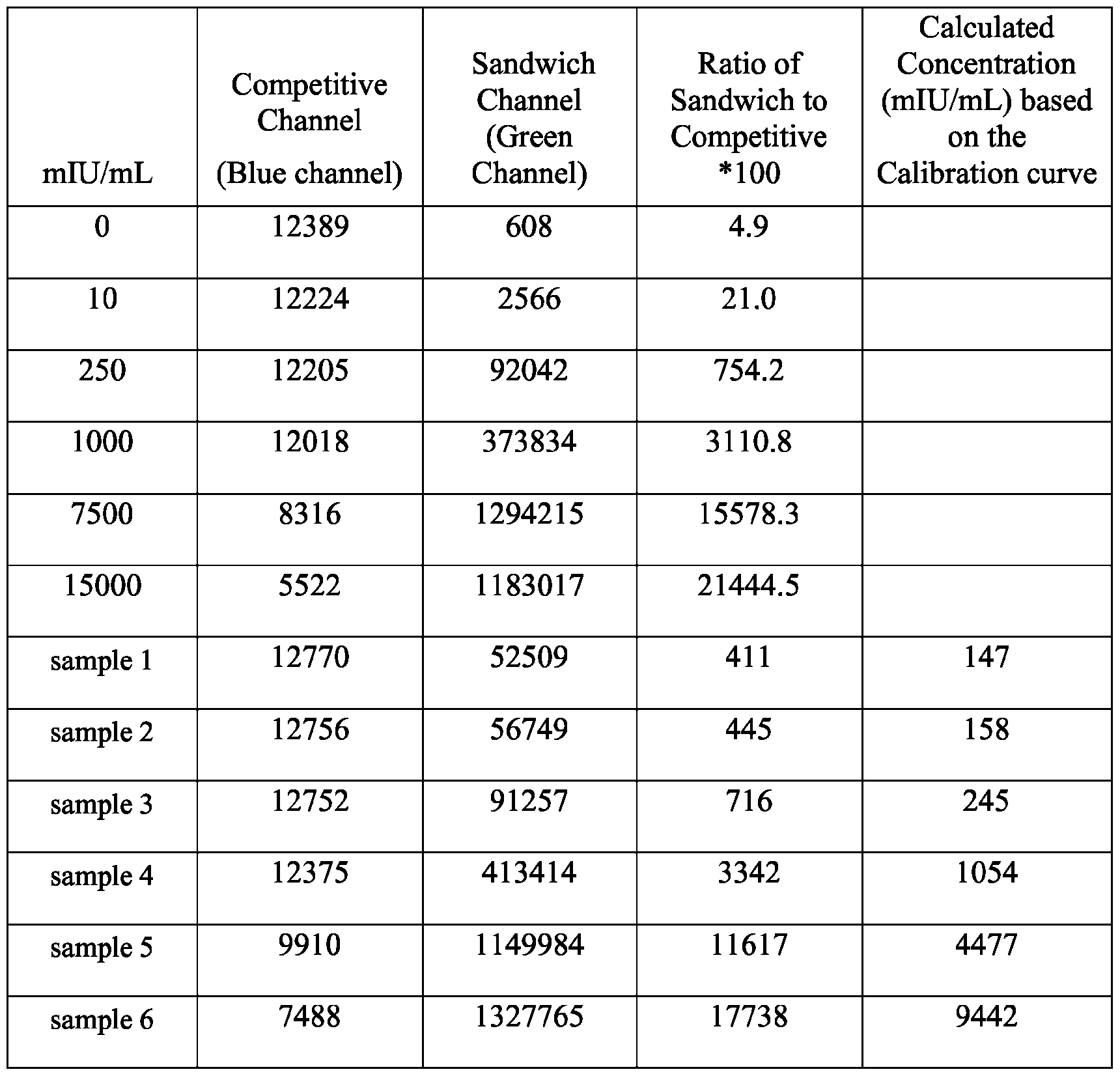
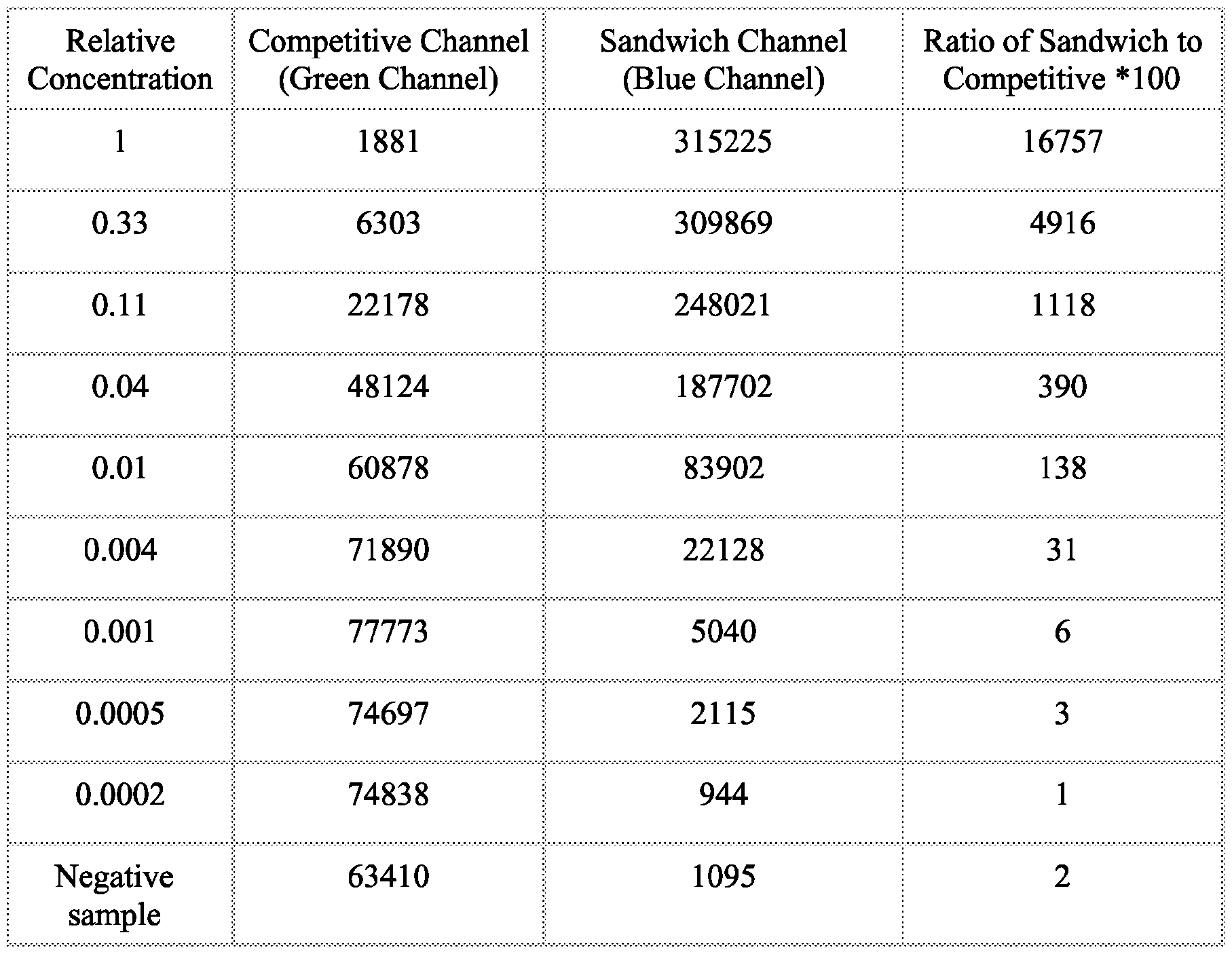
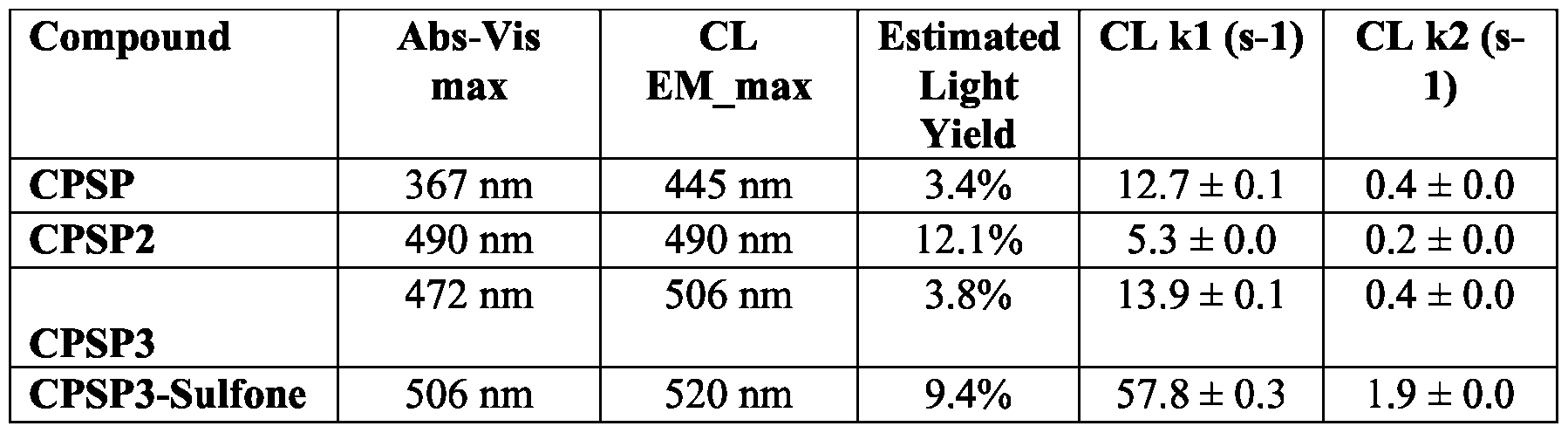
- FIG. 41 illustrates that with the chosen optical filters (channel 1 is blue and channel 2 is green in the figure), signals from CPSP or CPSP3 can be resolved at high precision.
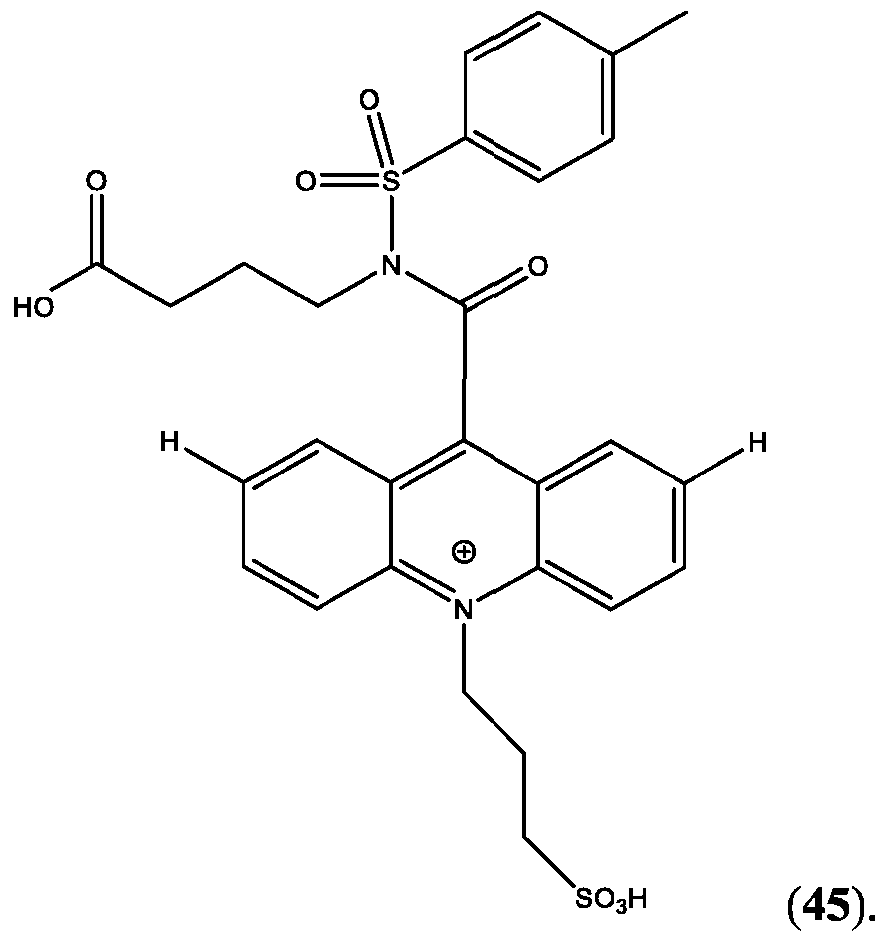
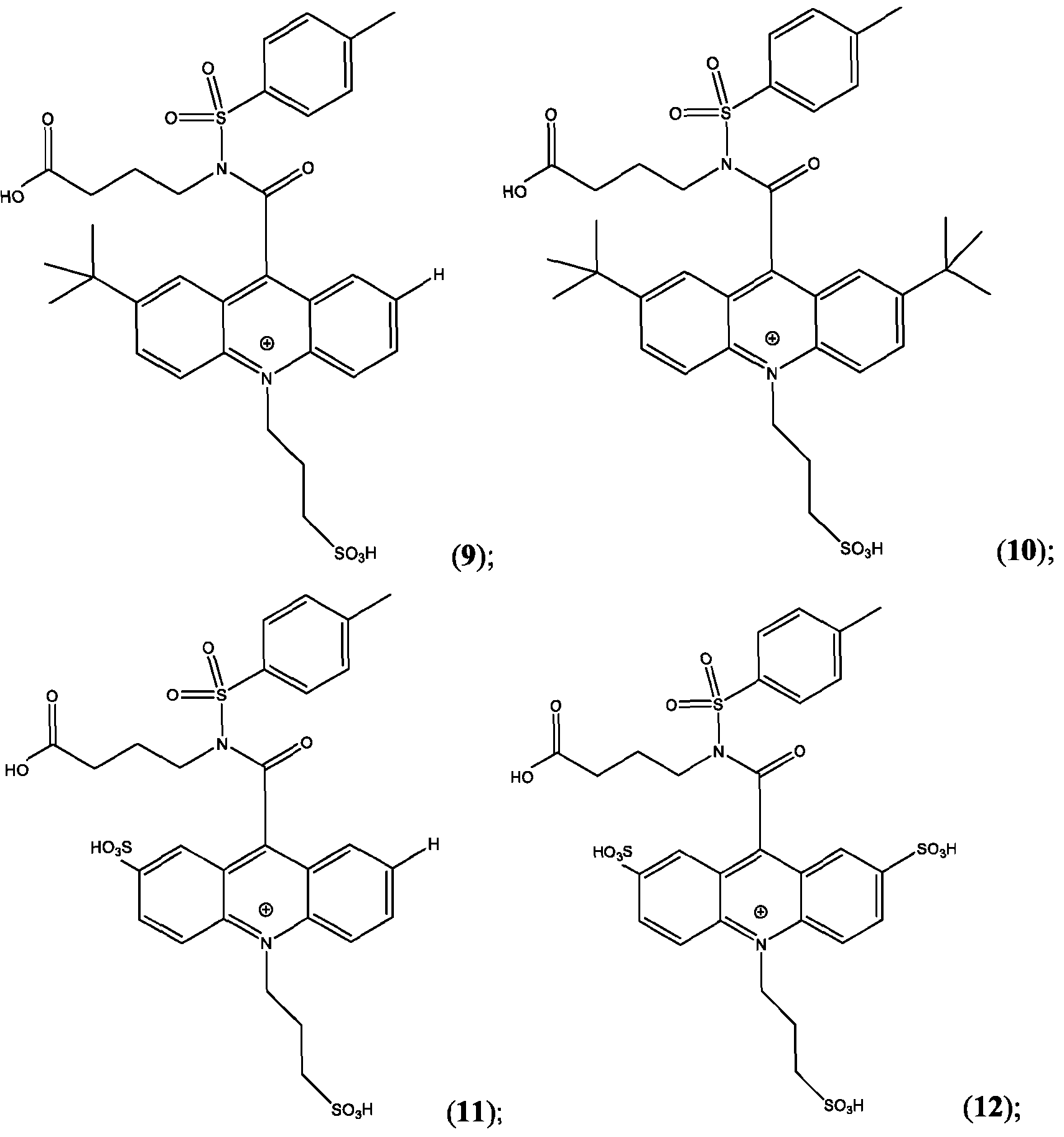
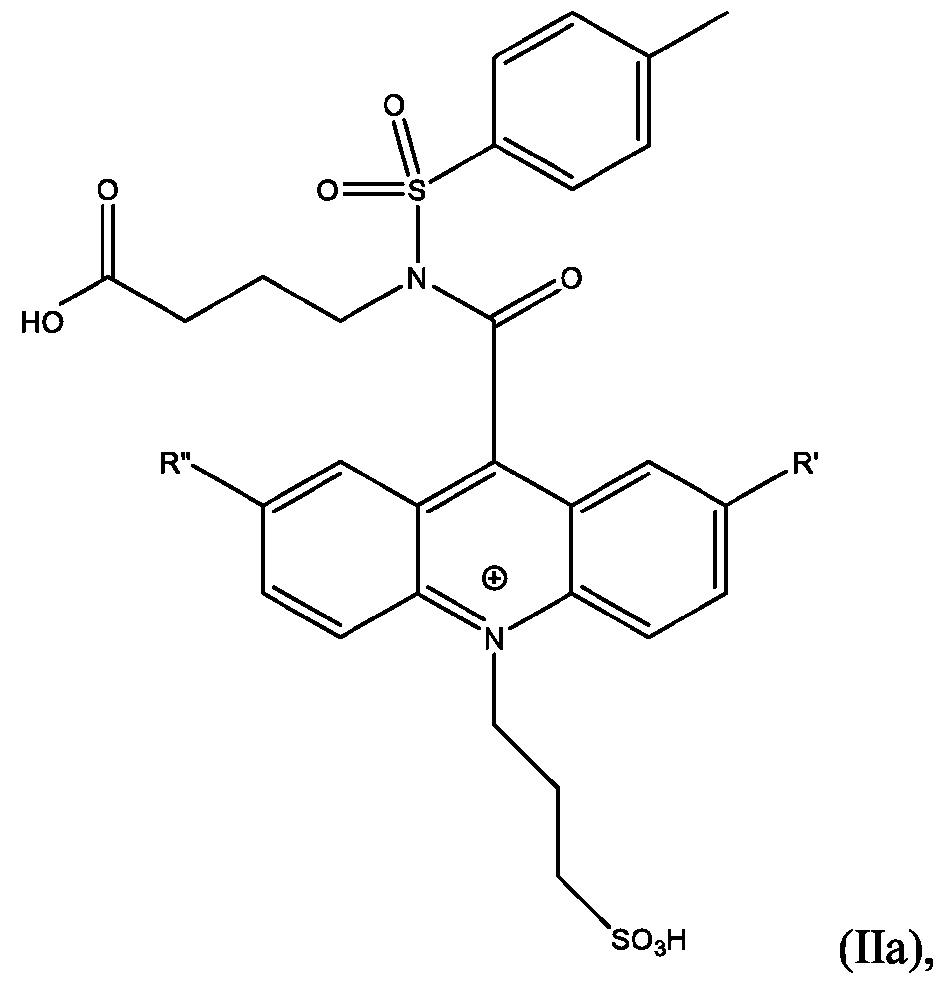
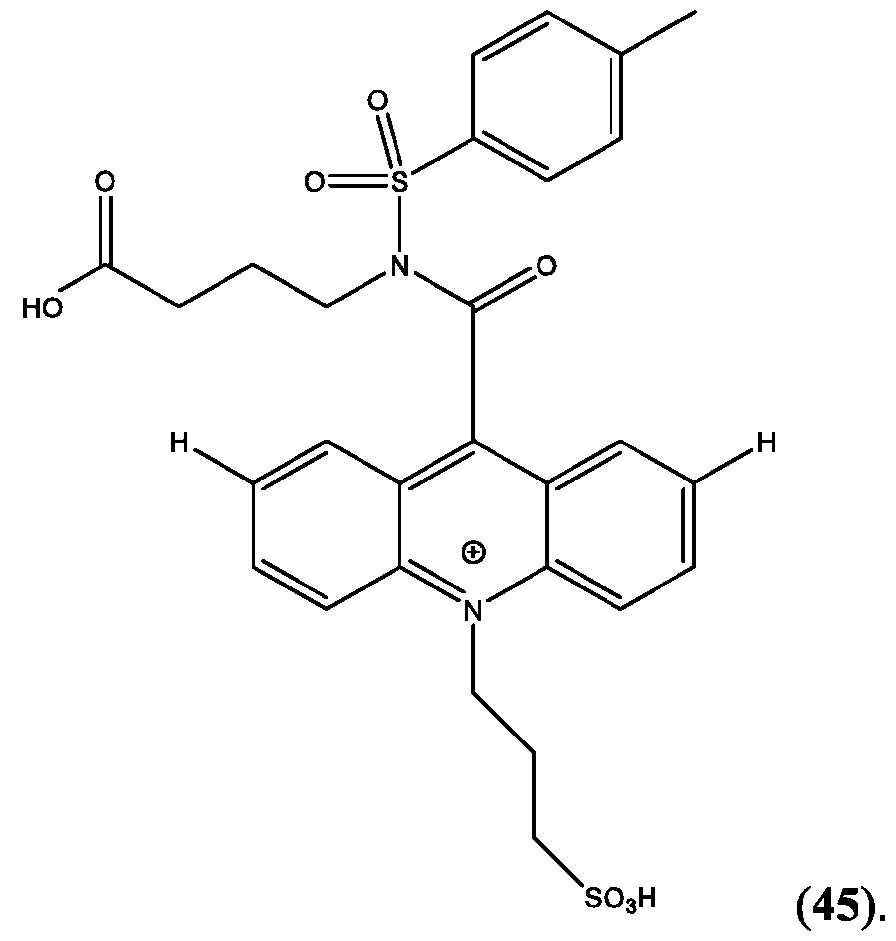
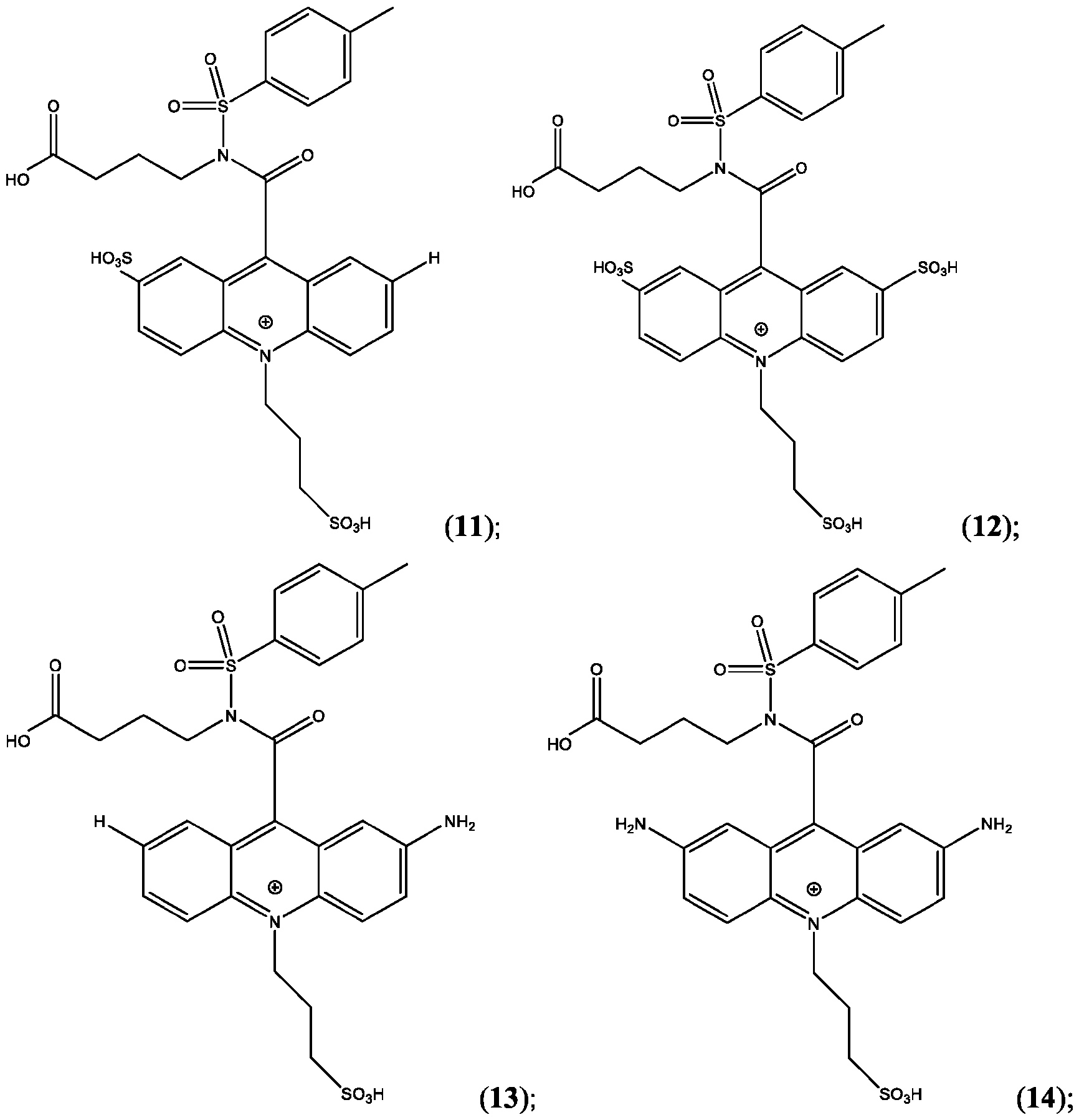
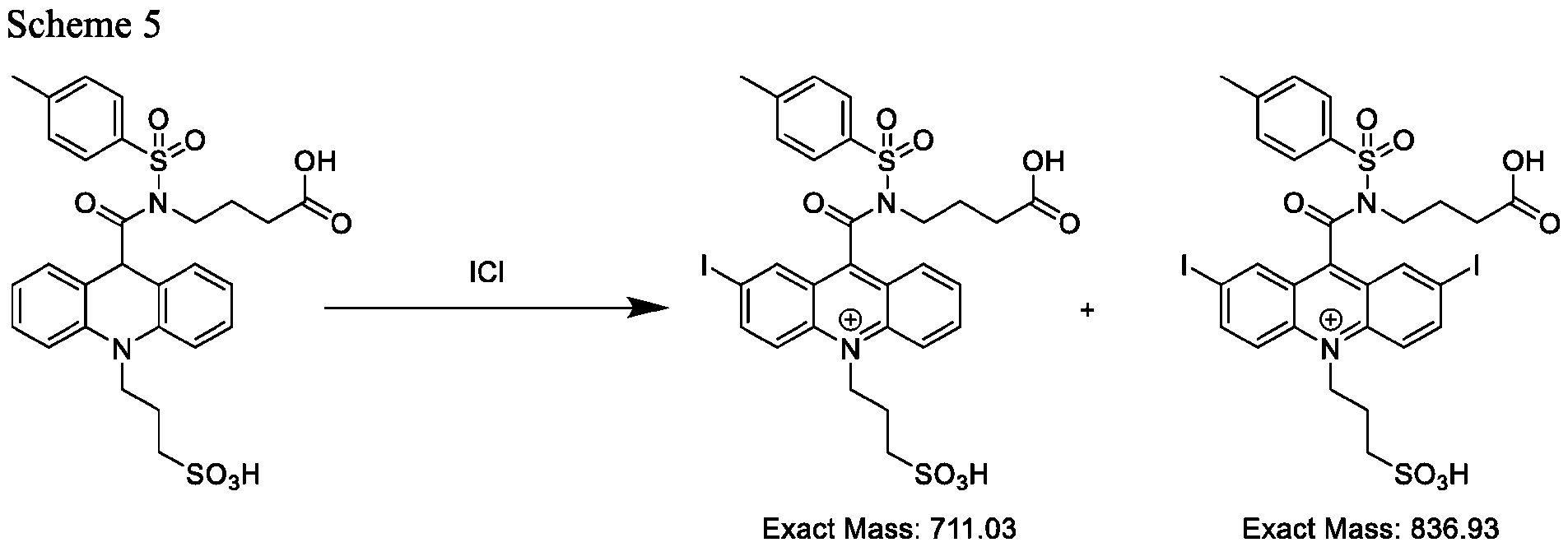
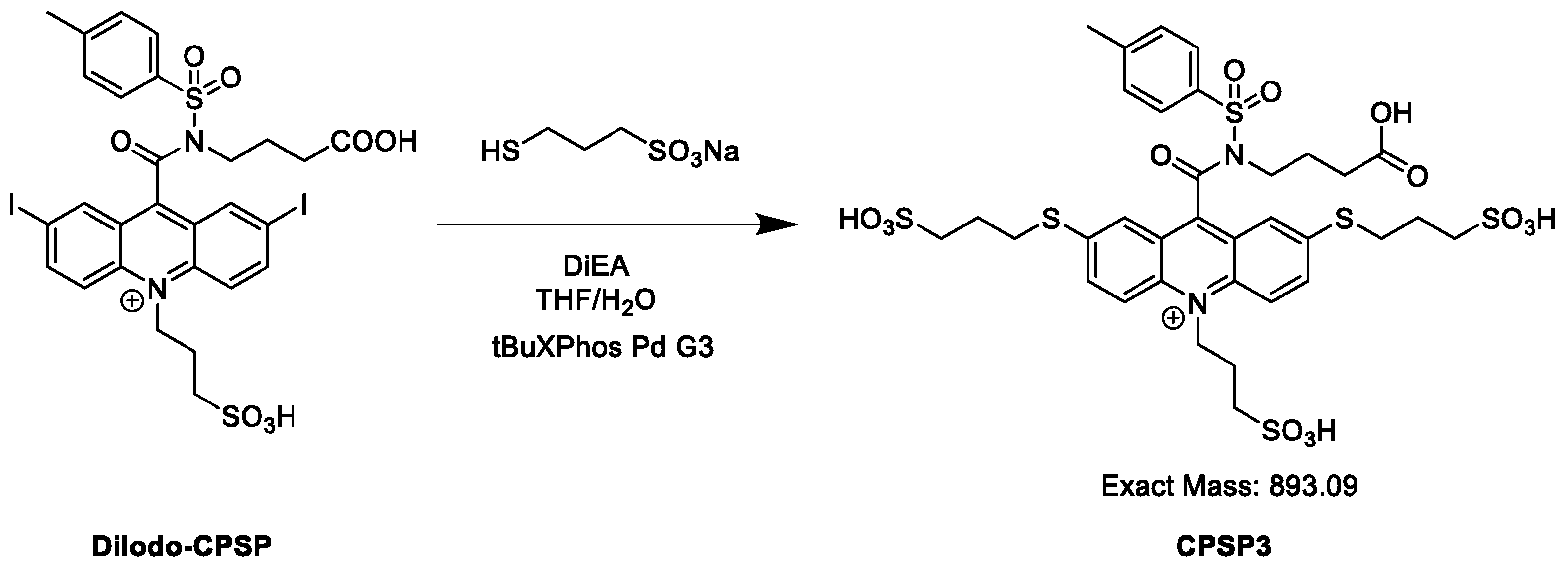
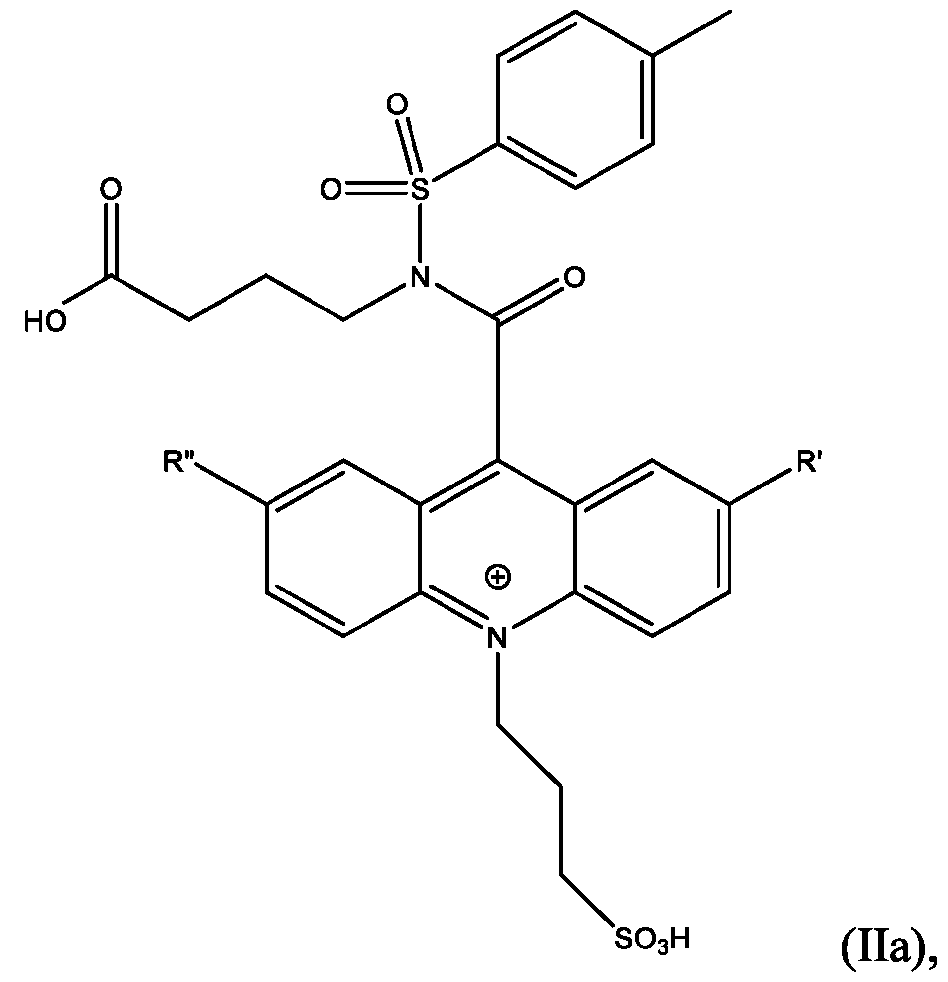
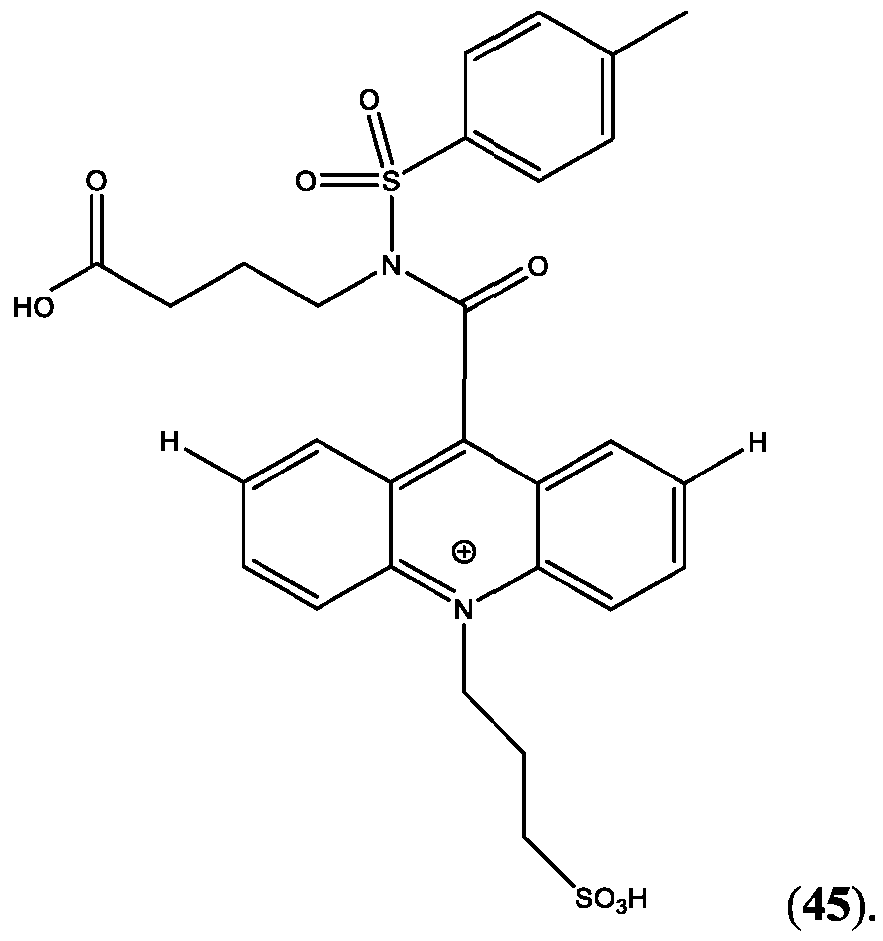
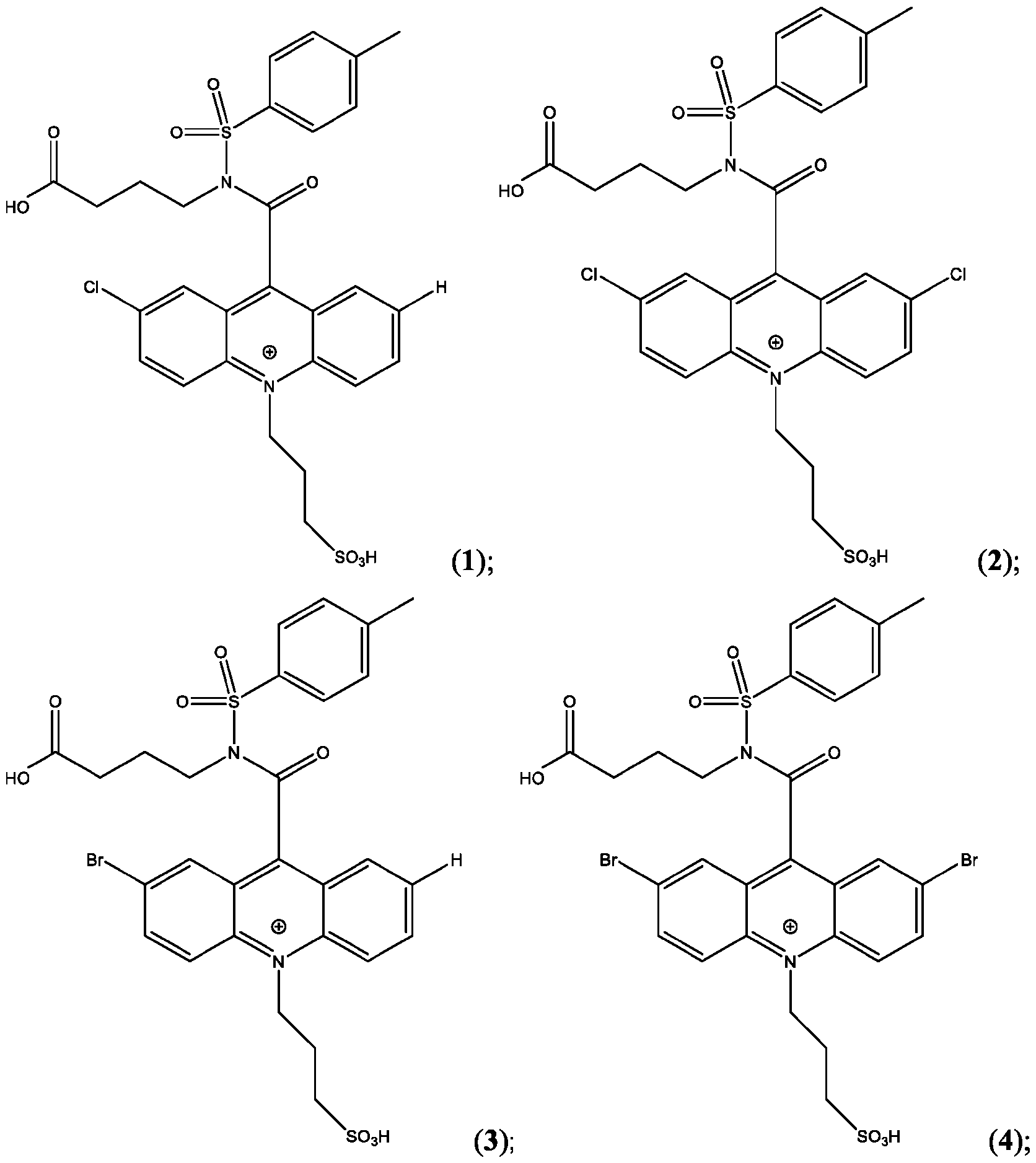
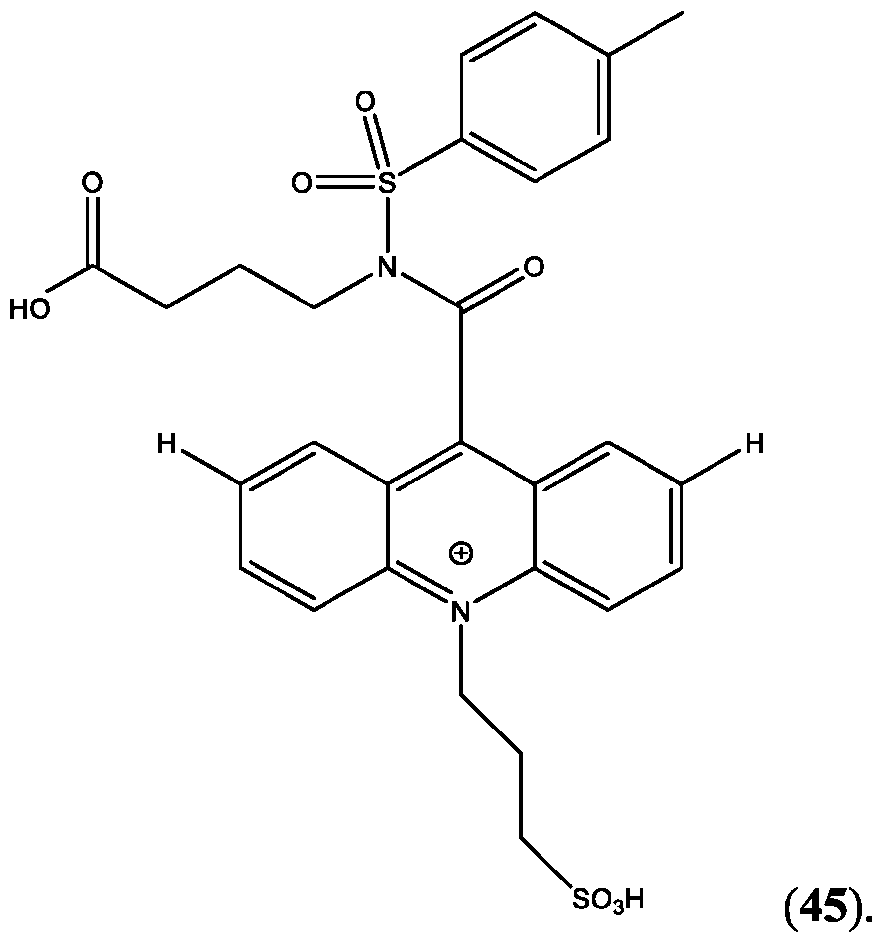
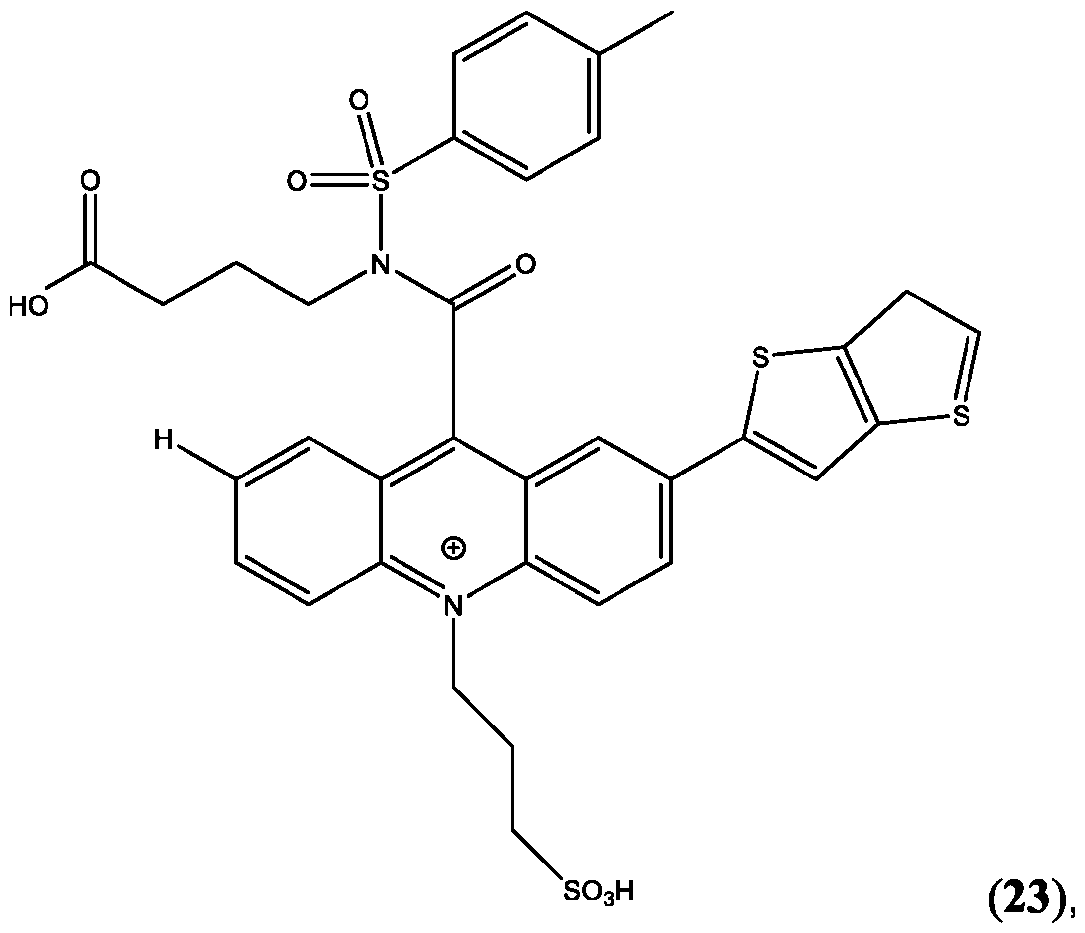
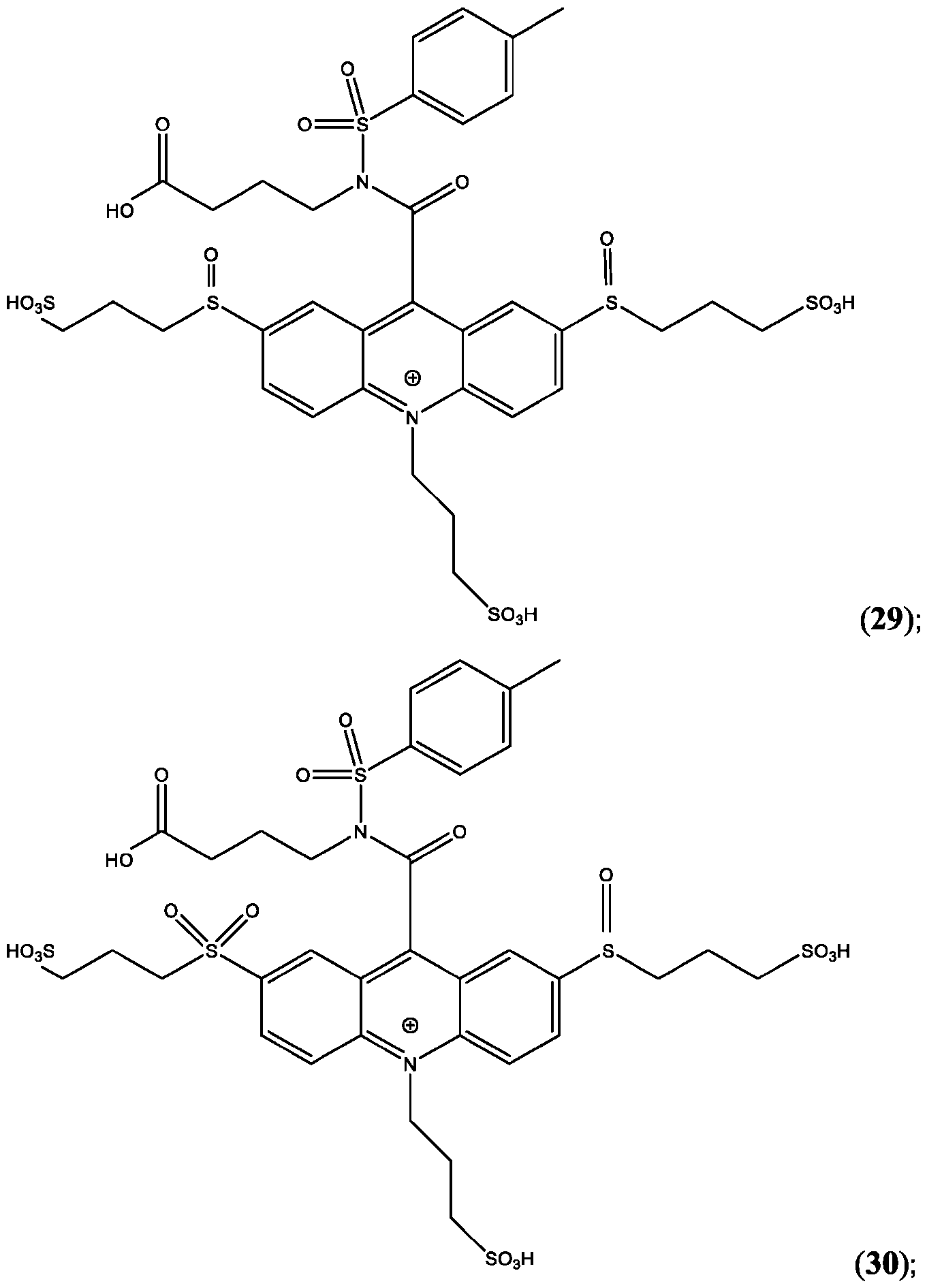
- CPSP 4- ( 10-(3 -sulfopropyl)-N-tosyl- 10X 4 -acridine-9-carboxamido)butanoic acid.
- FIG. 43 shows the immunoglobulin-G (IgG) and immunoglobulin-M (IgM) antibody levels from a single patient post vaccination using simultaneous dual channel measurements.
- FIG. 44A is a schematic of the extension of the dynamic range using a two-step assay using two different acridinium-containing compounds described herein in Example 45.
- FIG. 45B is an assay curve plotted based on signal measured in green (sandwich) channel according to Example 45.
- FIG. 48 illustrates the resolved signal of a human IgG and IgM mixture using dichloro-CPSP and ditbutyl-CPSP and a single channel detection system according to Example 47.
- the method further comprises using a first detection component to detect the first detectable signal generated by the first detectable label and a second detection component to detect the second detectable signal generated by the second detectable label.
- the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
- the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”), that competes with the analyte of interest wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
- the first detection reagent or the second detection reagent is an antigen or an antibody.
- the analyte of interest and the competing analyte are each captured on a solid support.
- the sandwich assay is performed in one step or two steps.
- the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained from the subject.
- the analyte of interest is bHCG, SARS- CoV-2, hepatitis B core antigen (HBcAg), phosphorylated hepatitis B core antigen, or combinations thereof.
- a method for determining the amount of an analyte of interest in a biological sample comprises the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a first reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c.
- the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein: (i) the sandwich immunoassay and competitive immunoassay are performed simultaneously, or sequentially, in any order: (ii) the second detectable label is identical to the first detectable label; (iii) the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and (iv) the first detectable signal and the second detectable signal are detected by an instrument comprising a single detection component; d.
- a system for determining the amount of an analyte of interest in a biological sample can comprise: a. a calibration curve for the analyte of interest; b. a sandwich immunoassay wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c.
- a competitive immunoassay wherein the competitive immunoassay comprises a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. a device comprising: i. a reaction vessel for performing the sandwich immunoassay and the competition assay; ii. a first detection component to determine the amount of the first detectable signal generated by the first detectable label; and iii.
- the first detectable signal comprises a first color and the second detectable signal comprises a second color.
- the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
- analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
- the first detection reagent, the second detection reagent or the first detection agent and the second detection reagent is an antigen or an antibody.
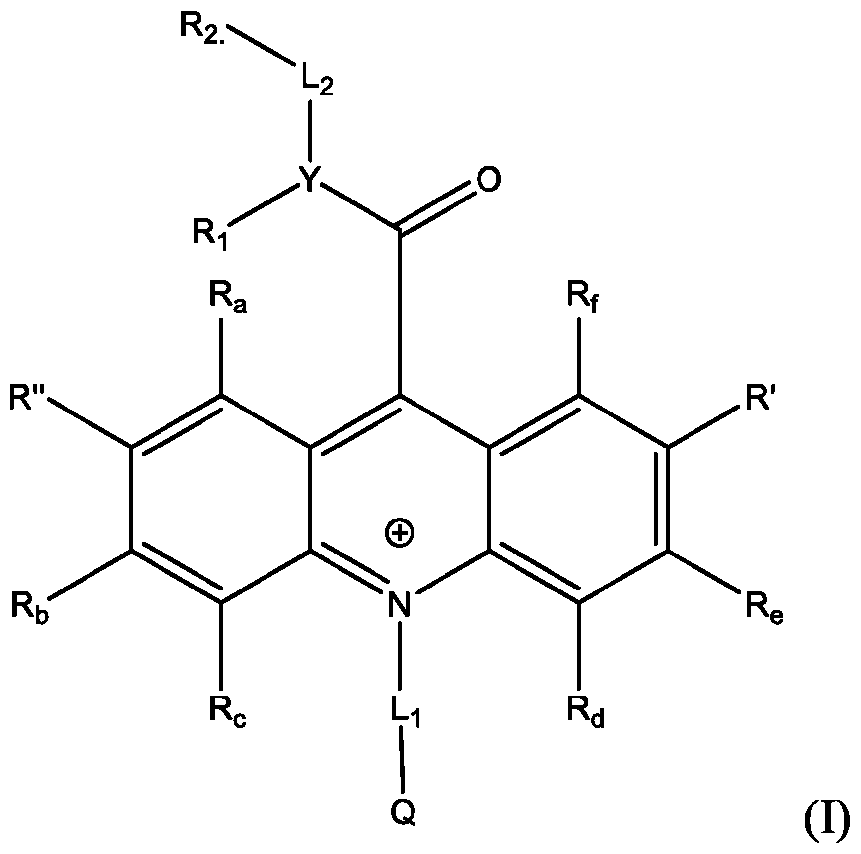
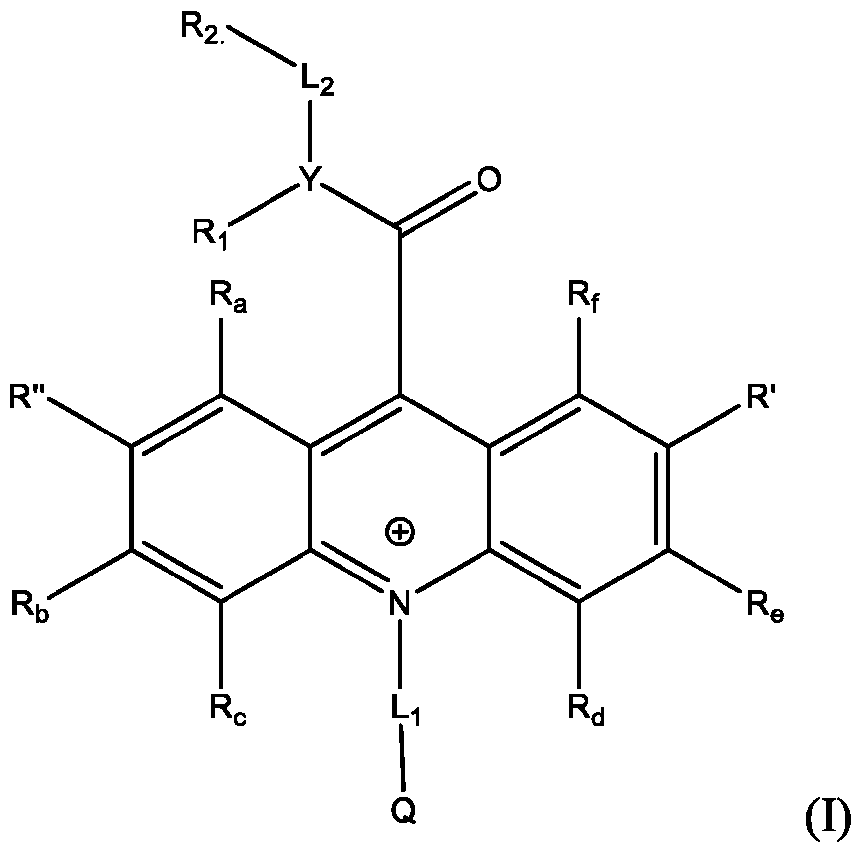
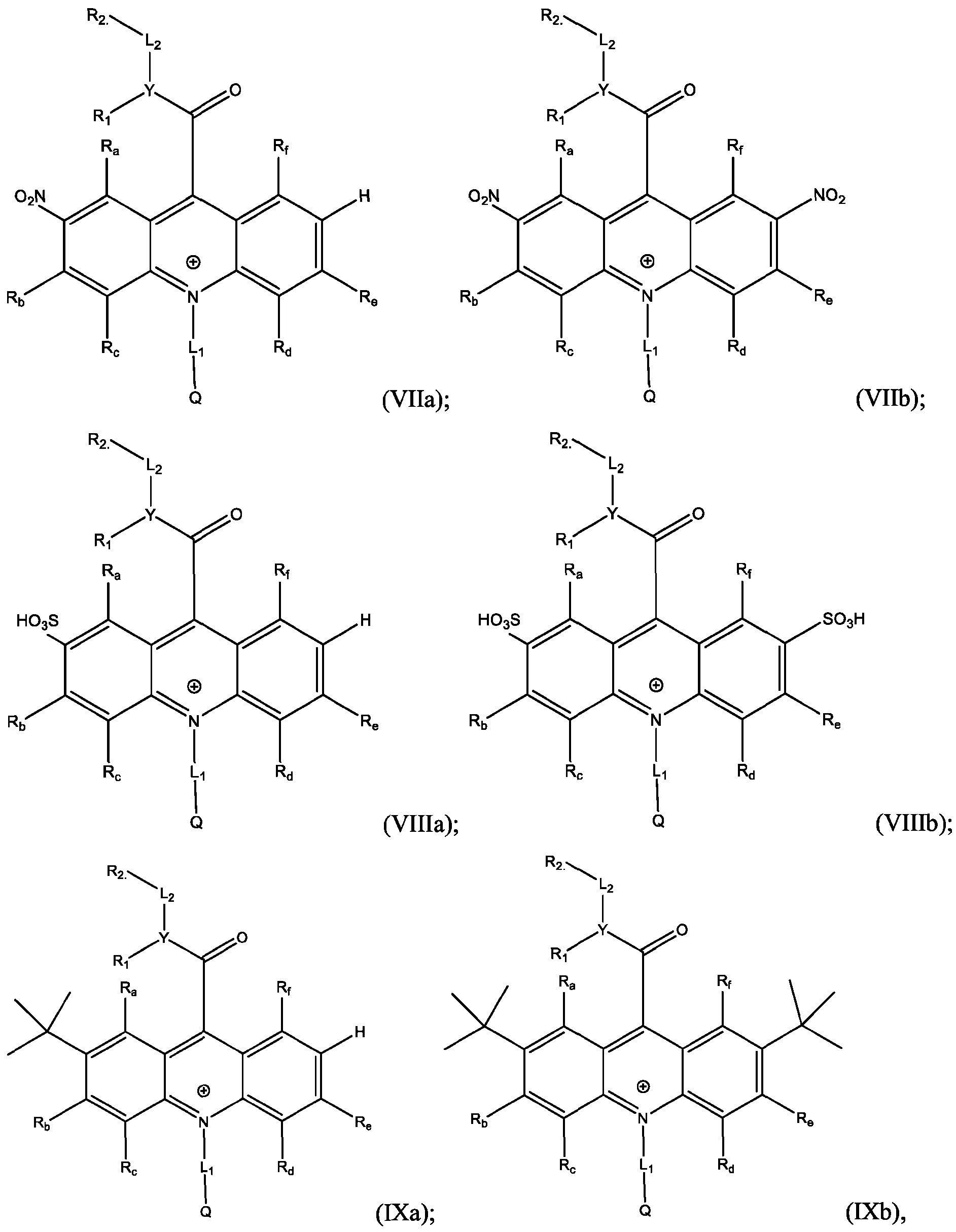
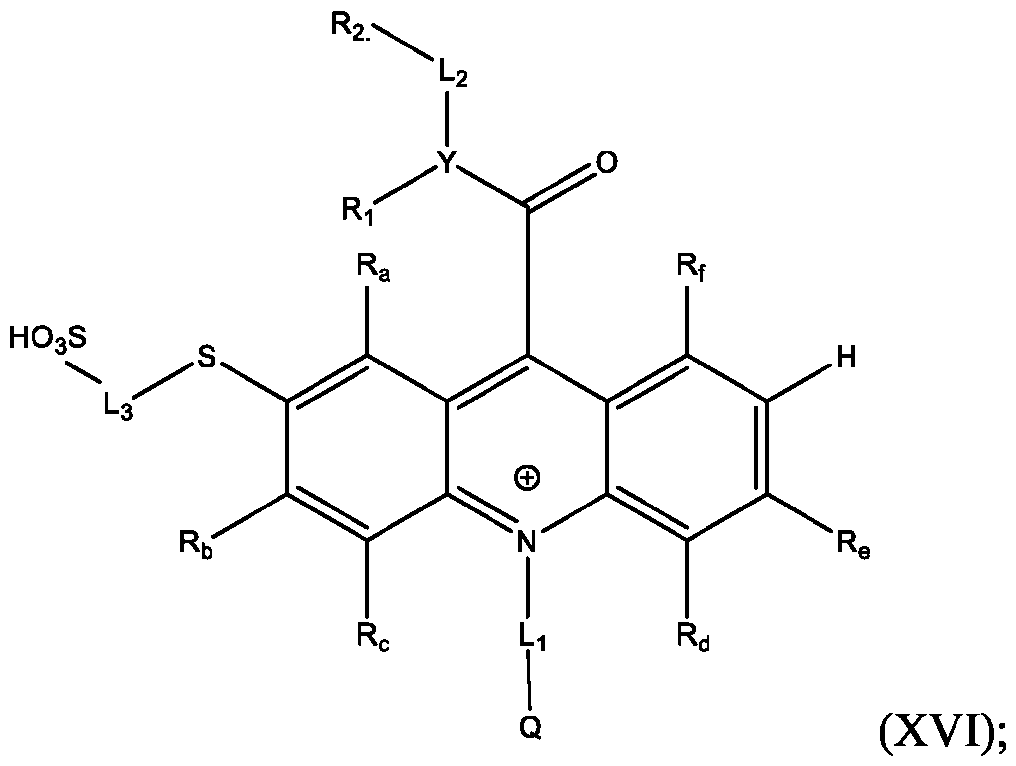
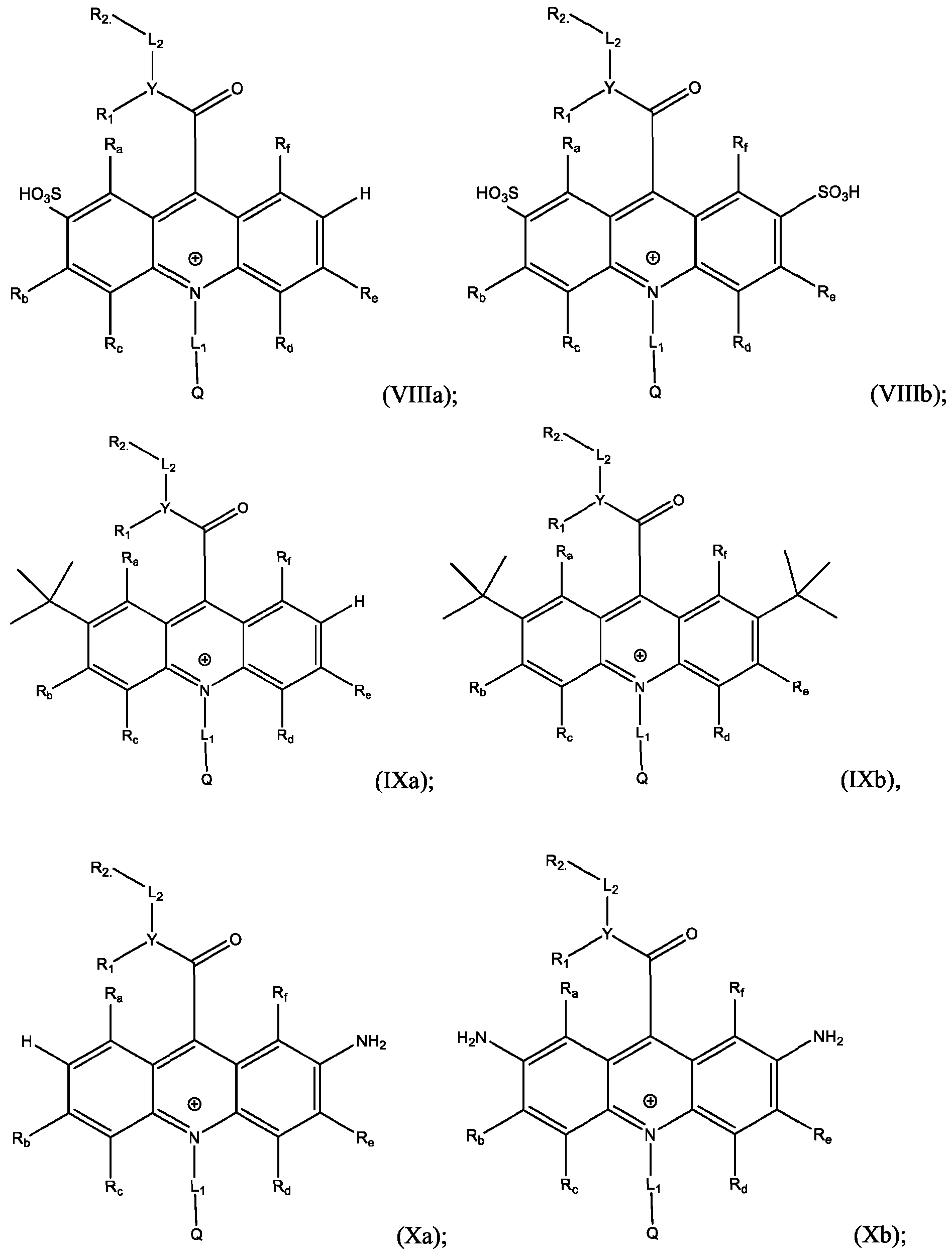
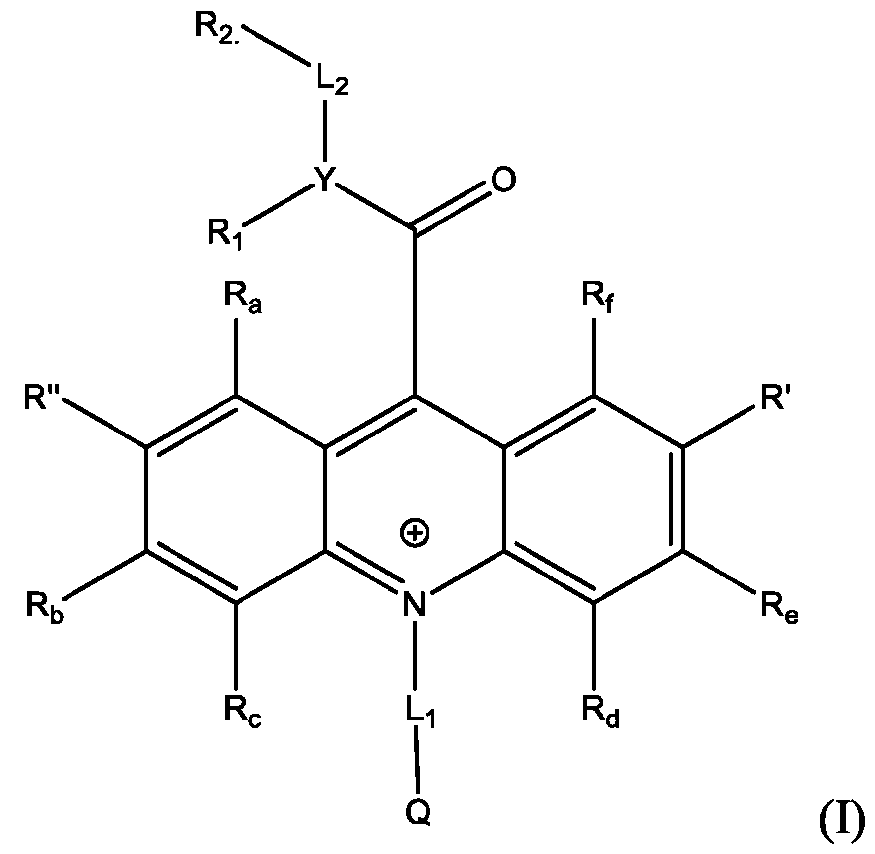
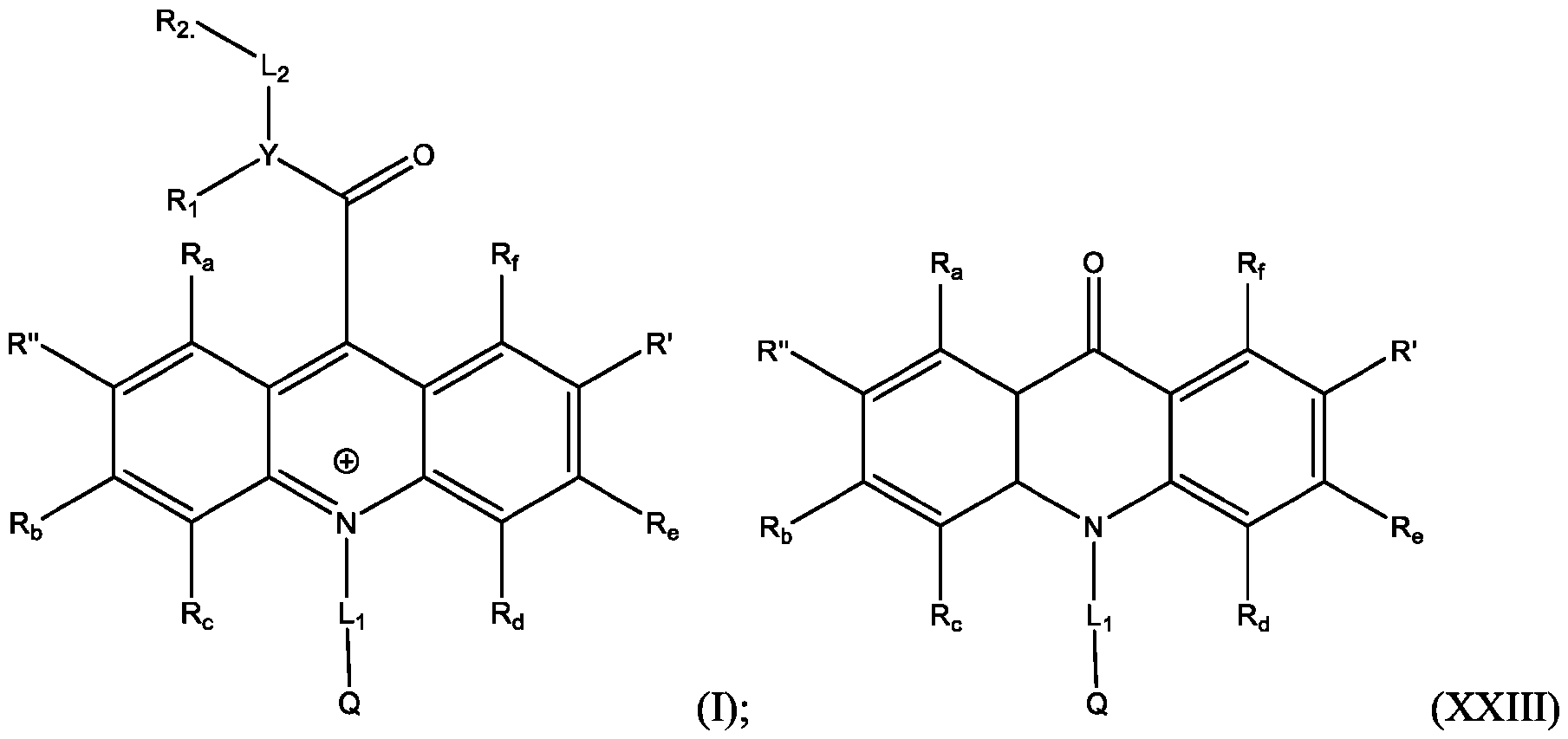
- Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
- R 2 is selected from -SO3Z and -COOZ
- Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
- R a , R b , R c , R d , R e , and R f are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
- Y is nitrogen
- A is aryl that is unsubstituted or substituted with 1, 2, 3, 4, or 5 substituents selected from C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl.
- R 2 is -COOZ.
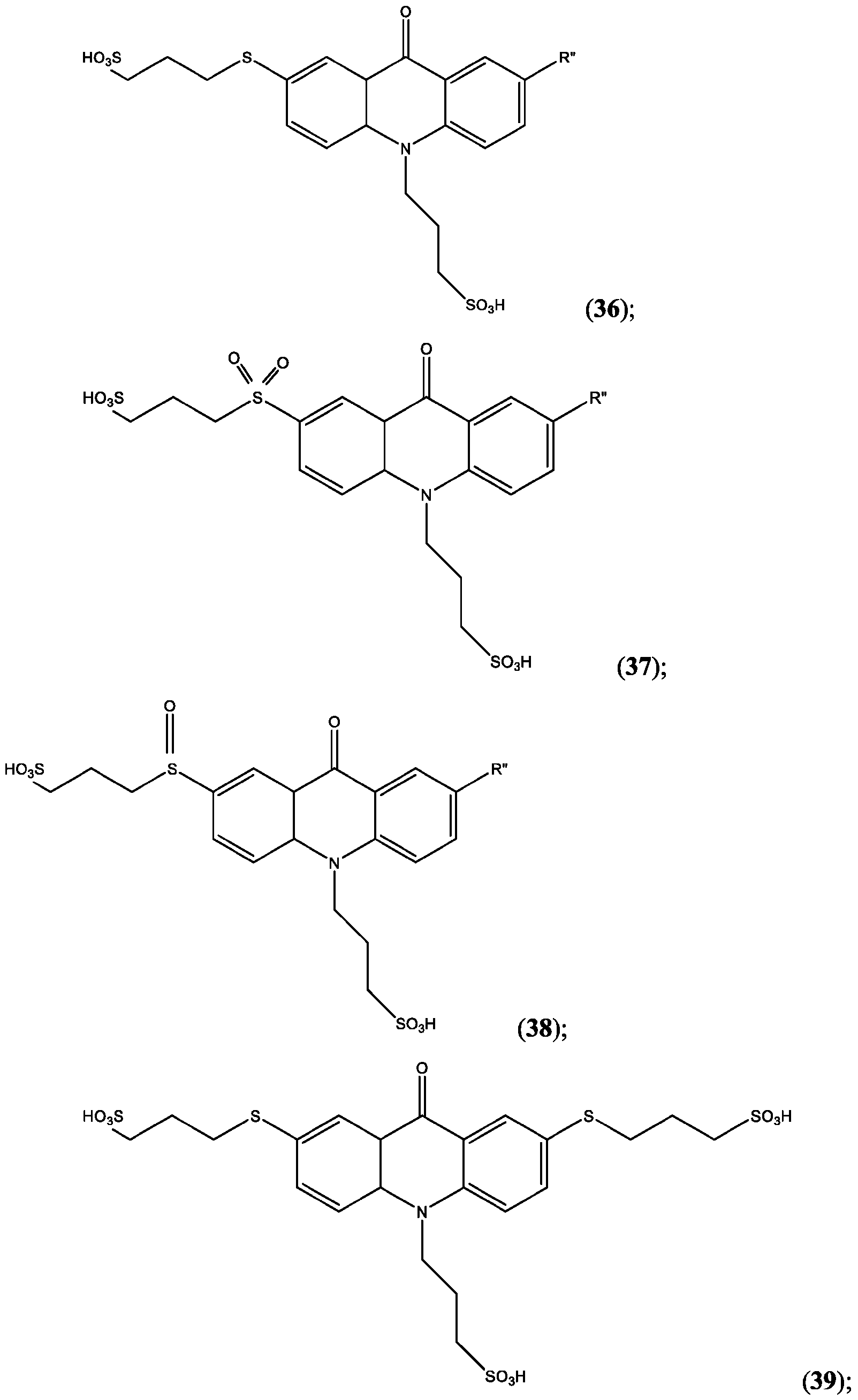
- R’, R”, or both R’ and R” are hydrogen.
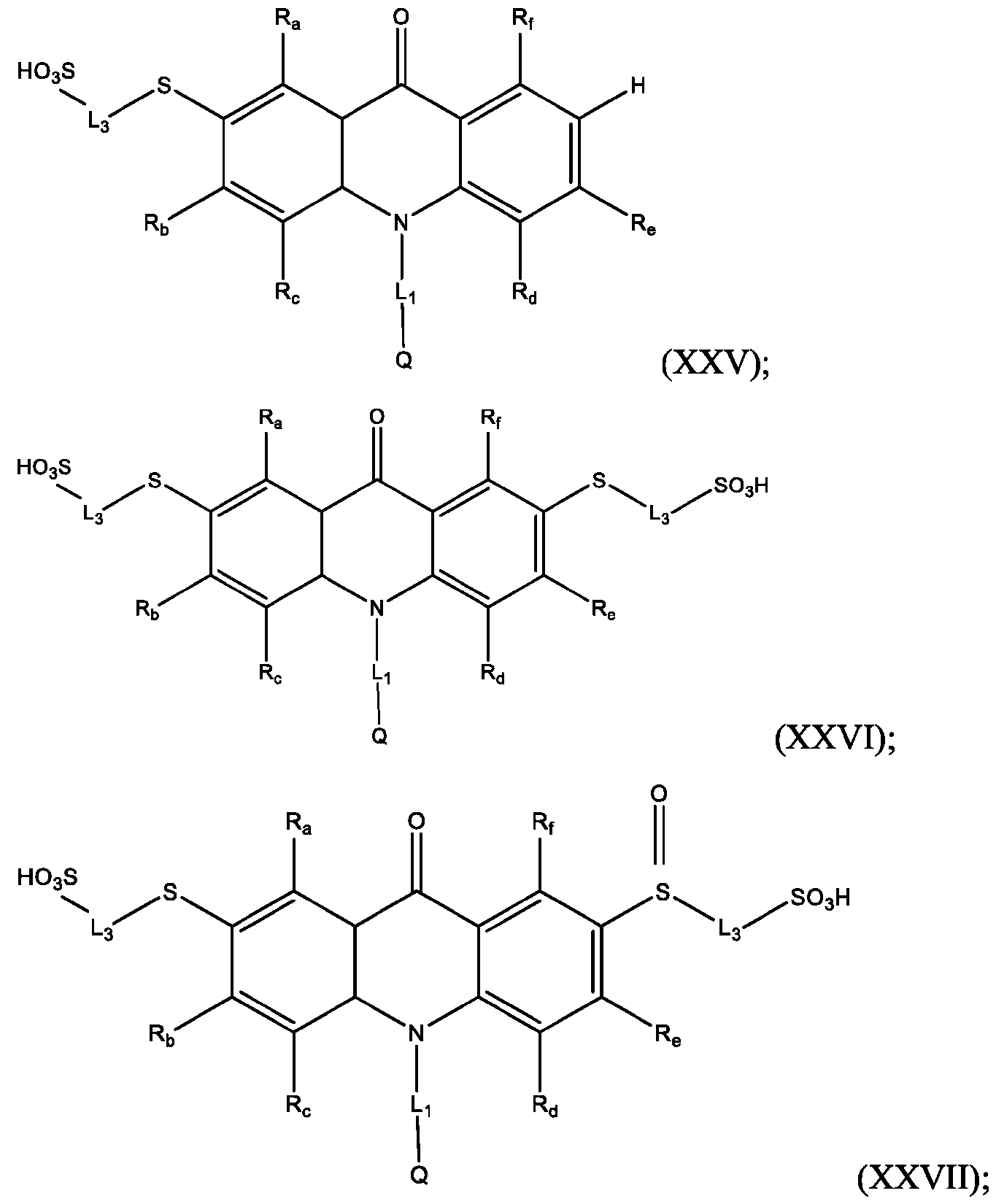
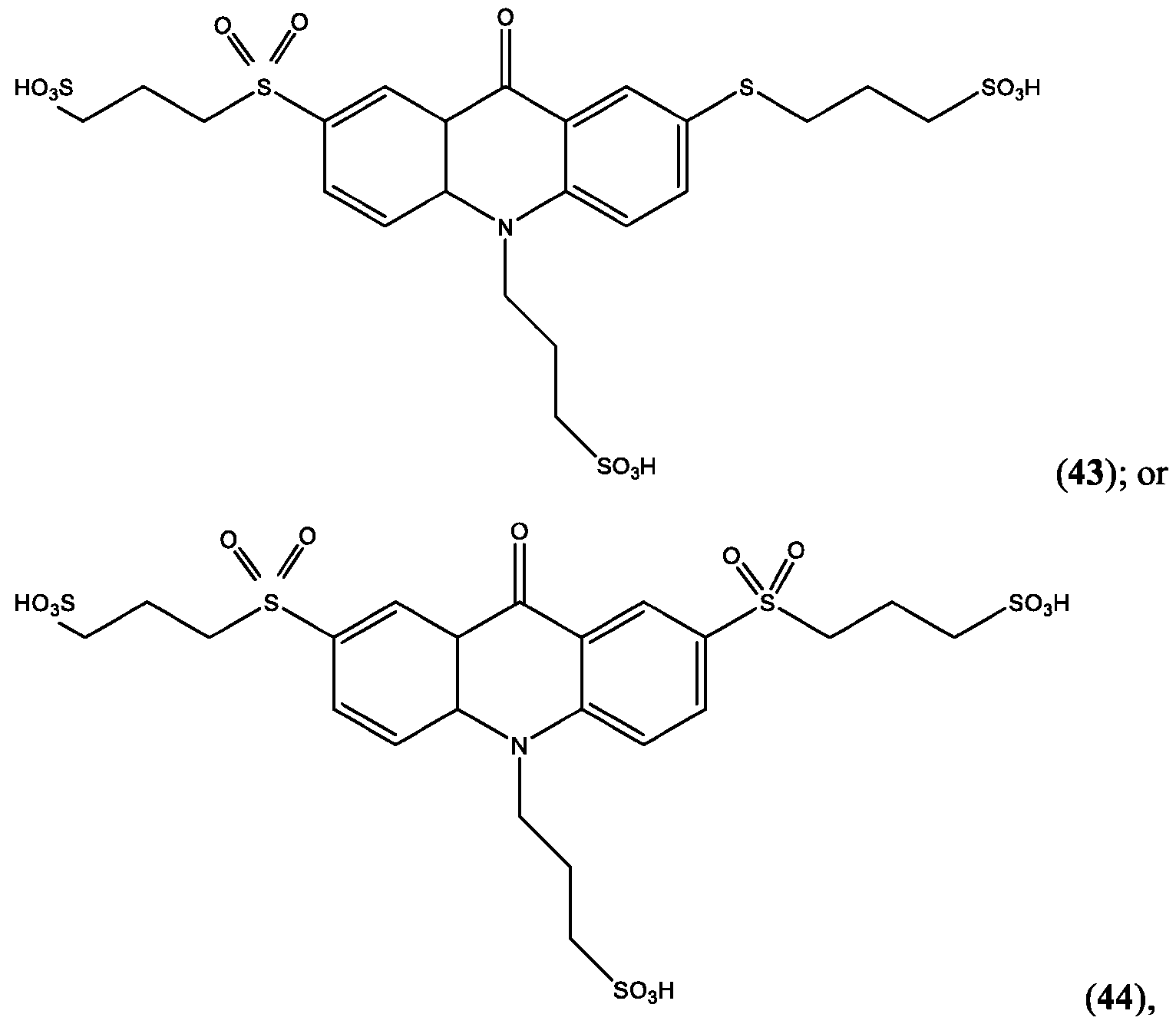
- R’ is -SO2-L3-SO3H and R” is -S-L3-SO3H.
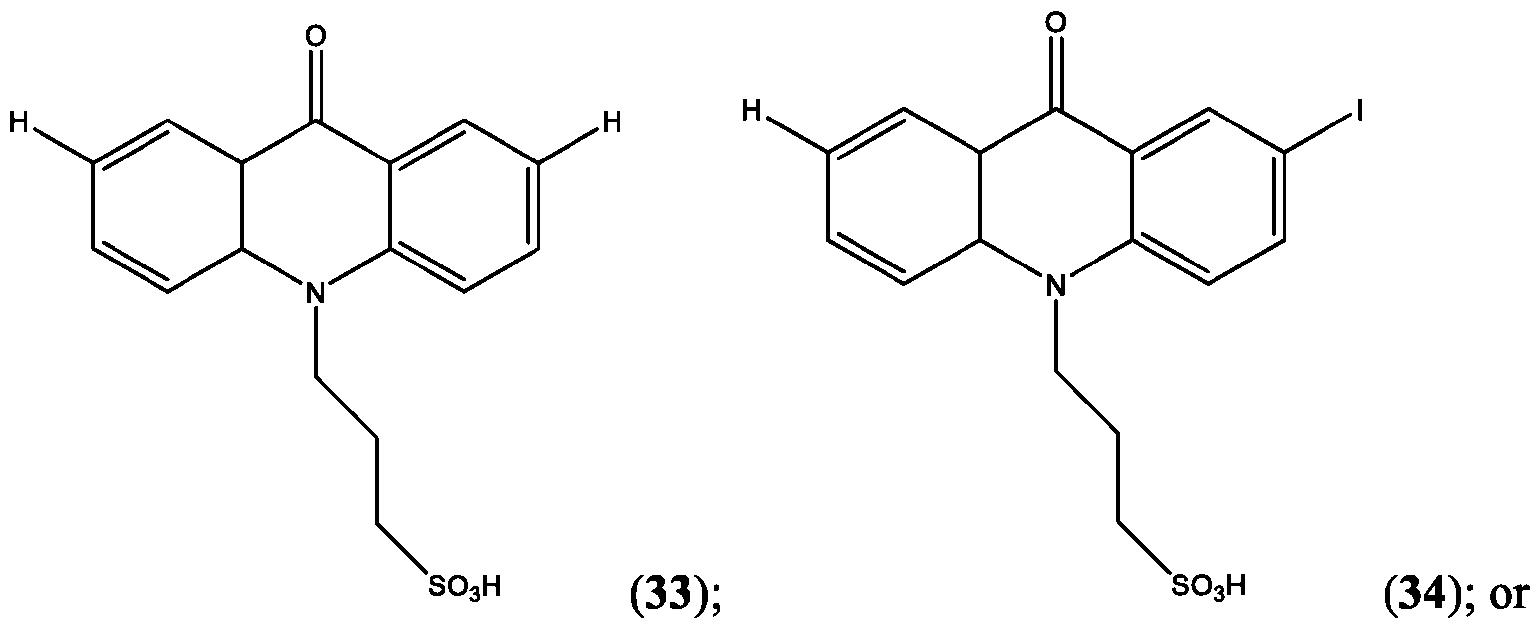
- R’ is -S-L3-SO3H and R” is one of H, Cl, Br, I, -NO2, t-butyl, or -SO3H.
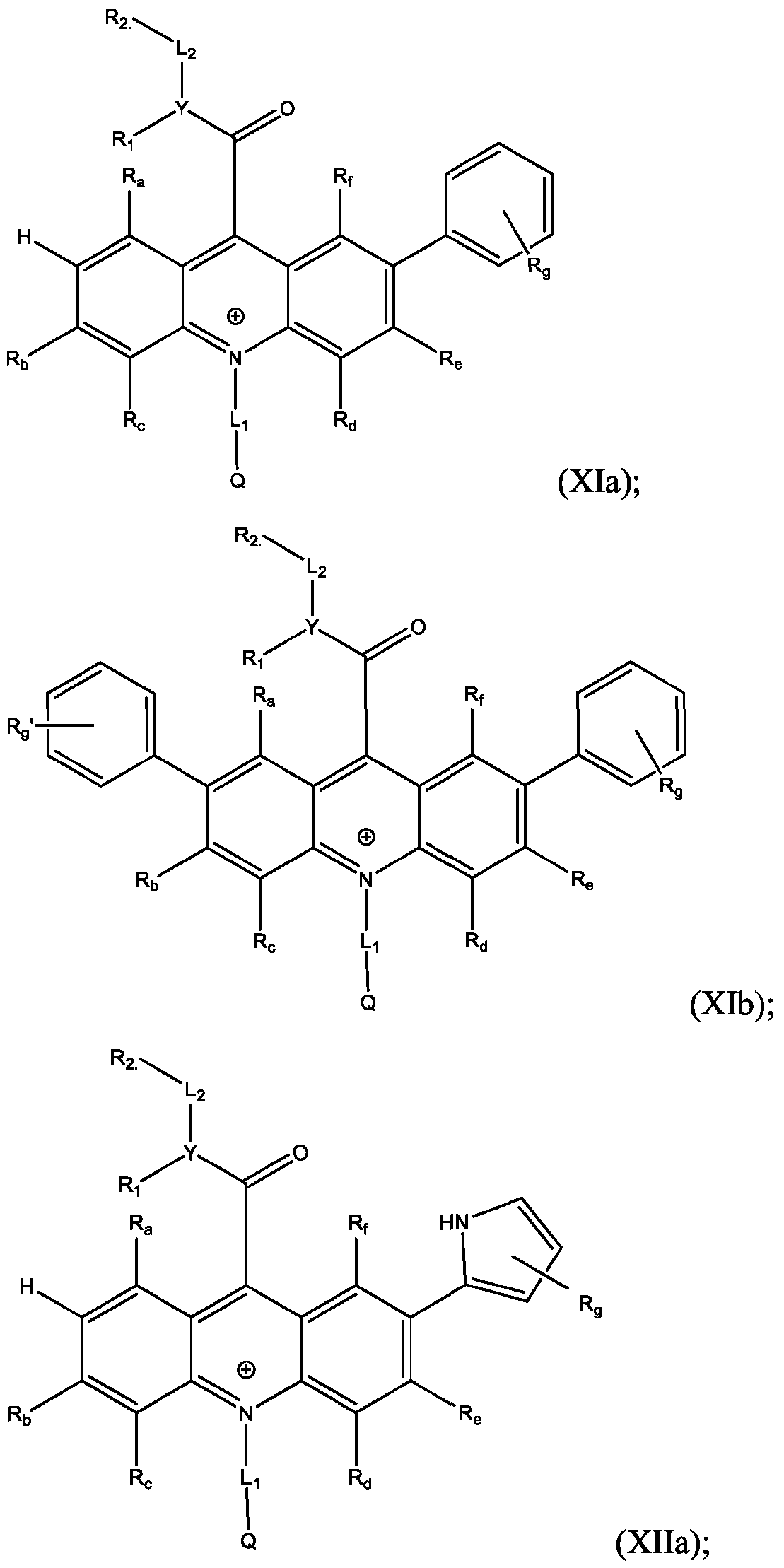
- R’, R”, or both R’ and R” are a phenyl group substituted with at least one of hydroxy, cyano, alkylamino, dialkylamino, or aminoalkyl.
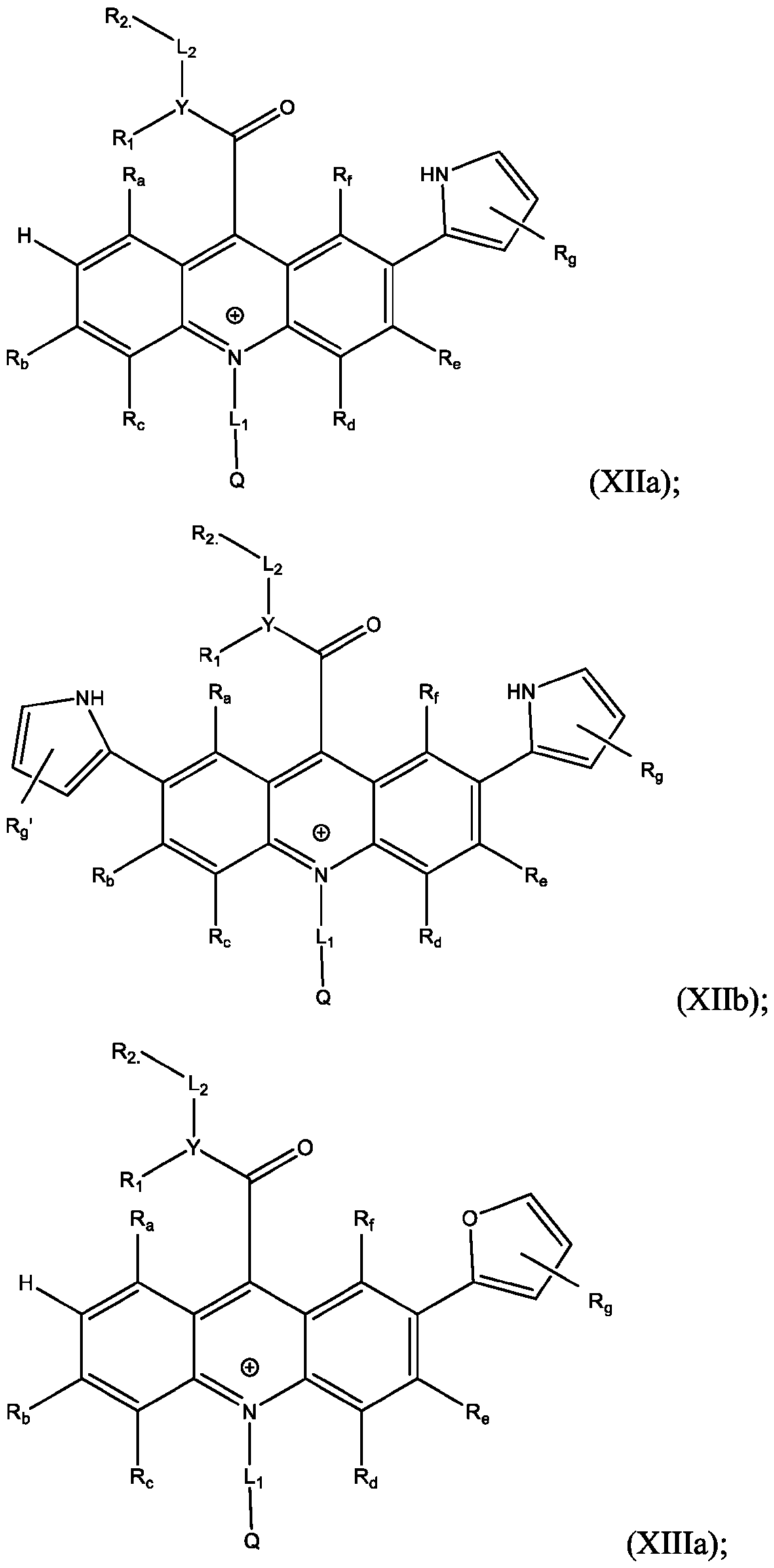
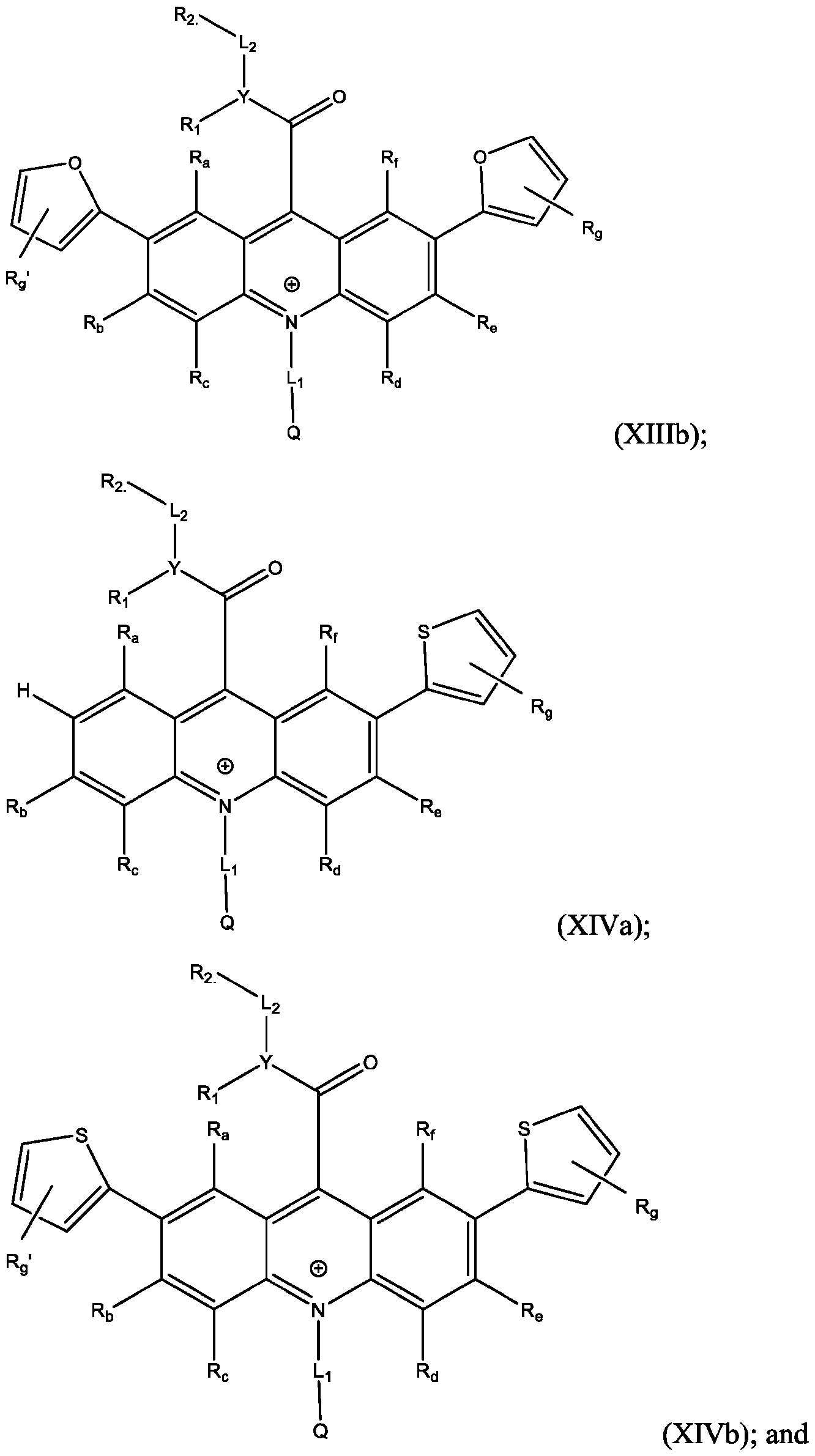
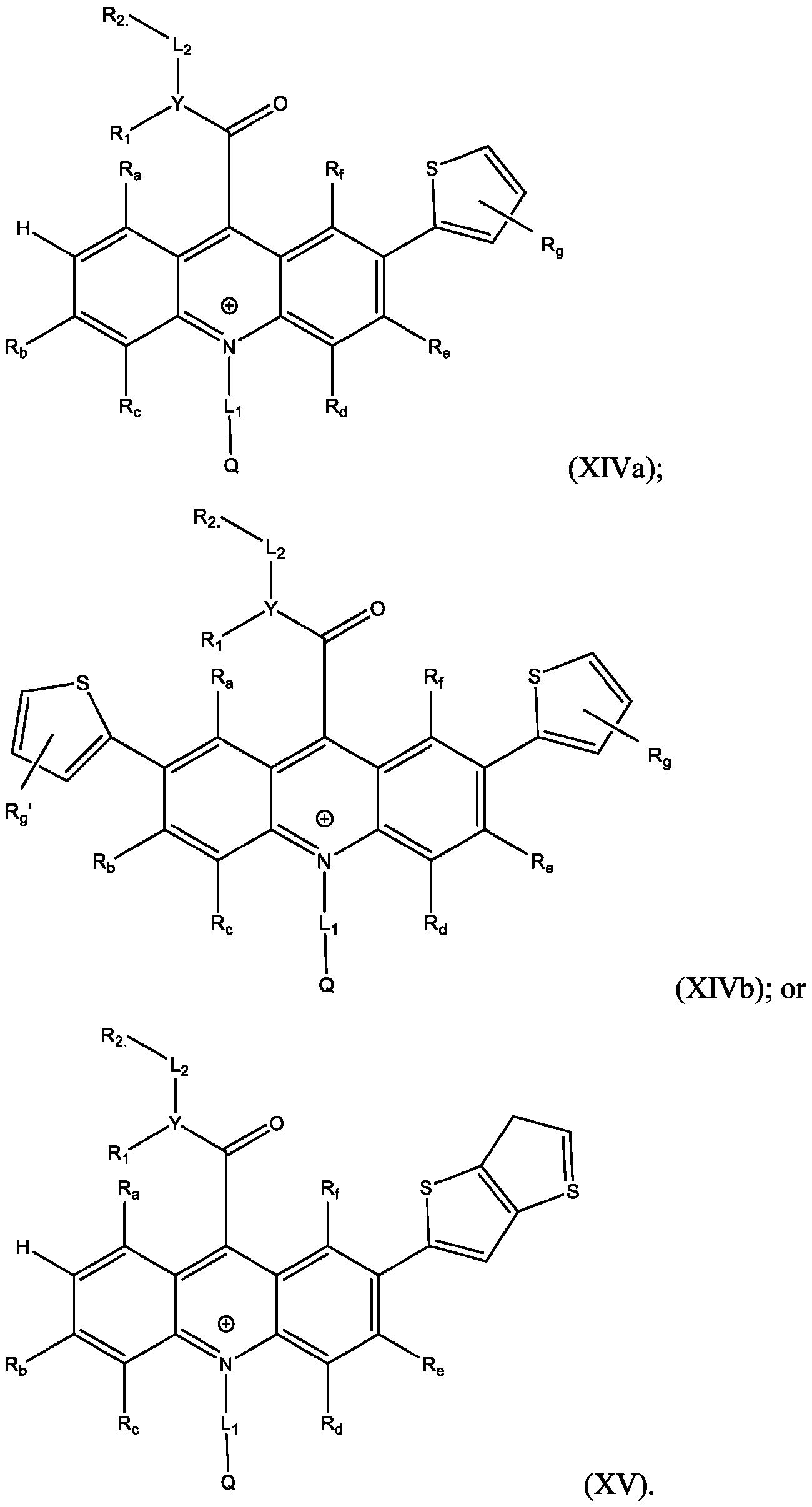
- R’, R”, or both R’ and R are a substituted or unsubstituted heteroaryl group.
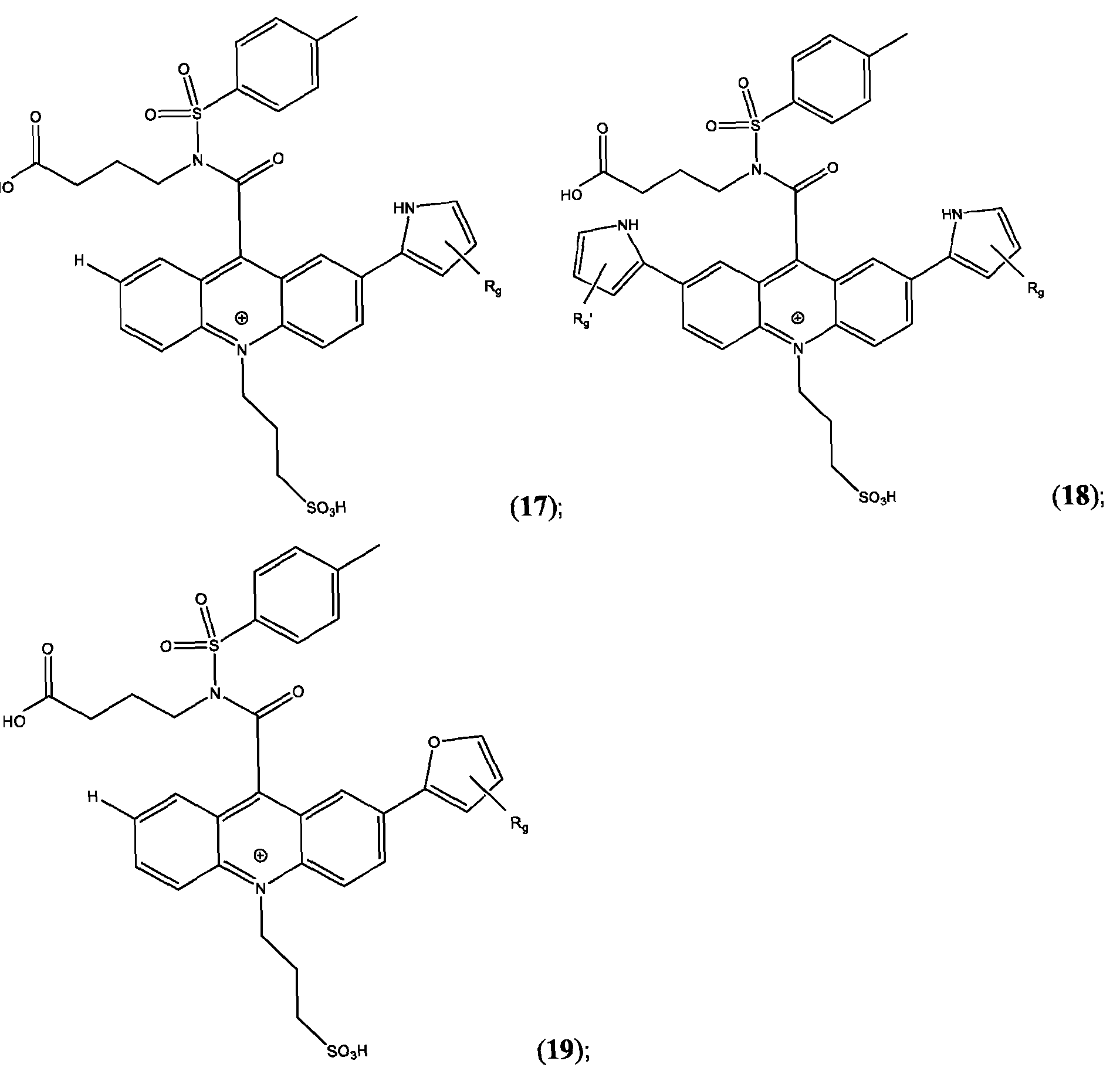
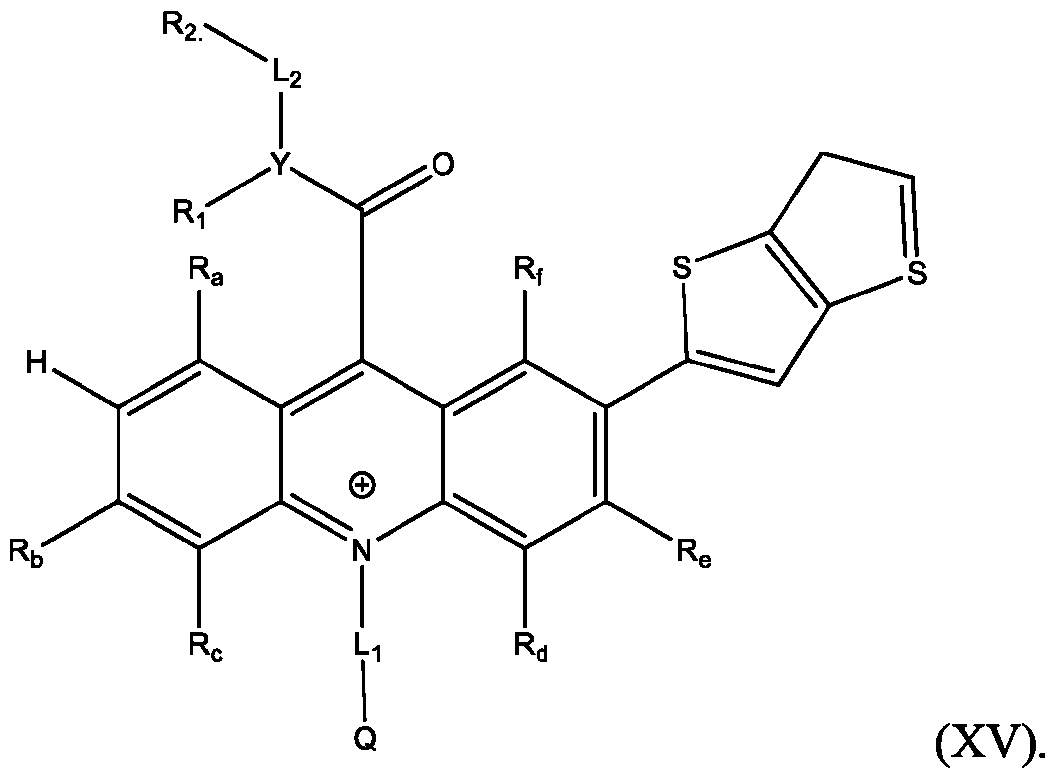
- R’, R”, or both R’ and R are a substituted or unsubstituted heteroaryl group selected from pyrrolyl, furanyl, thienyl, or thienothiophenyl.
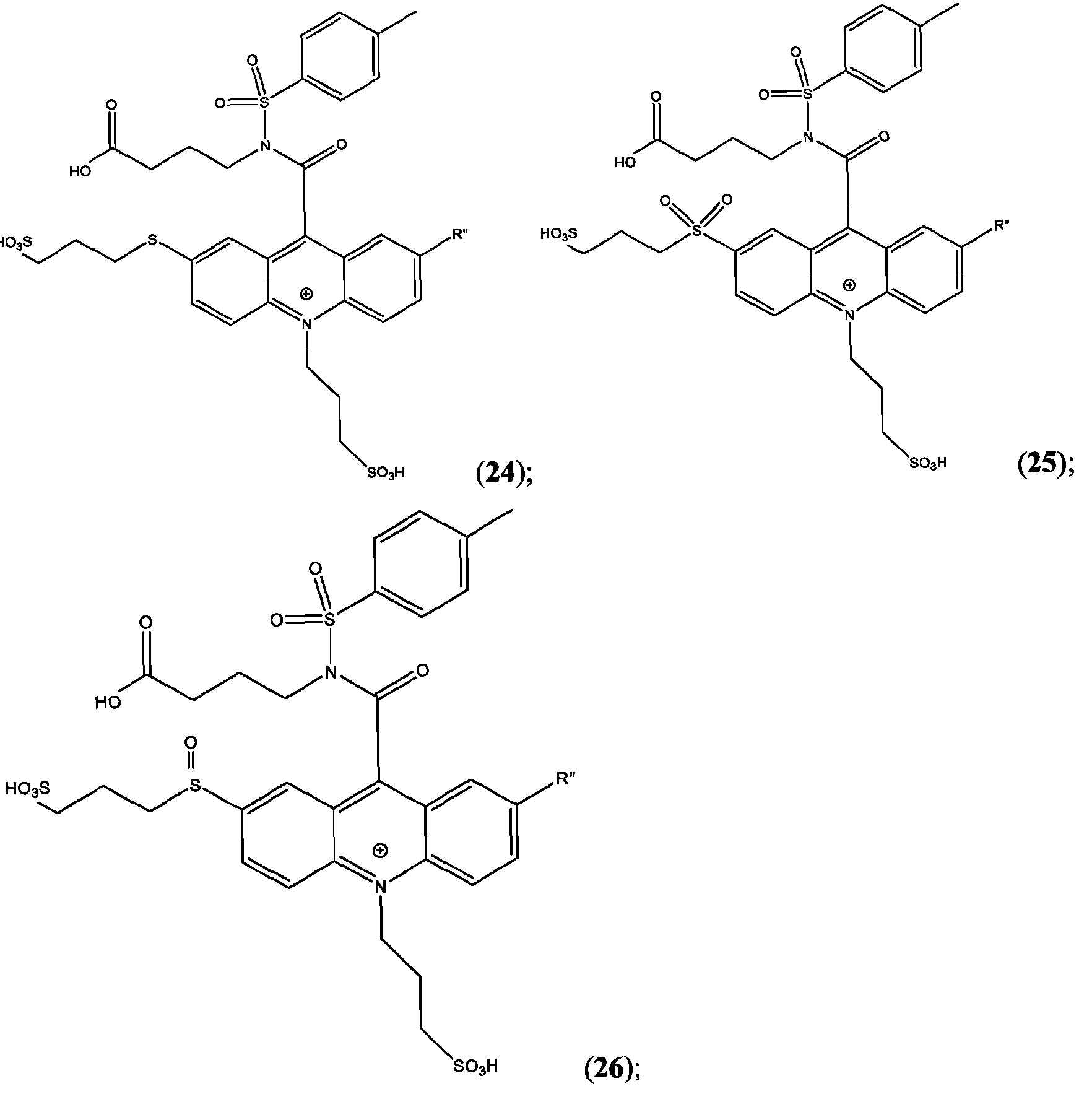
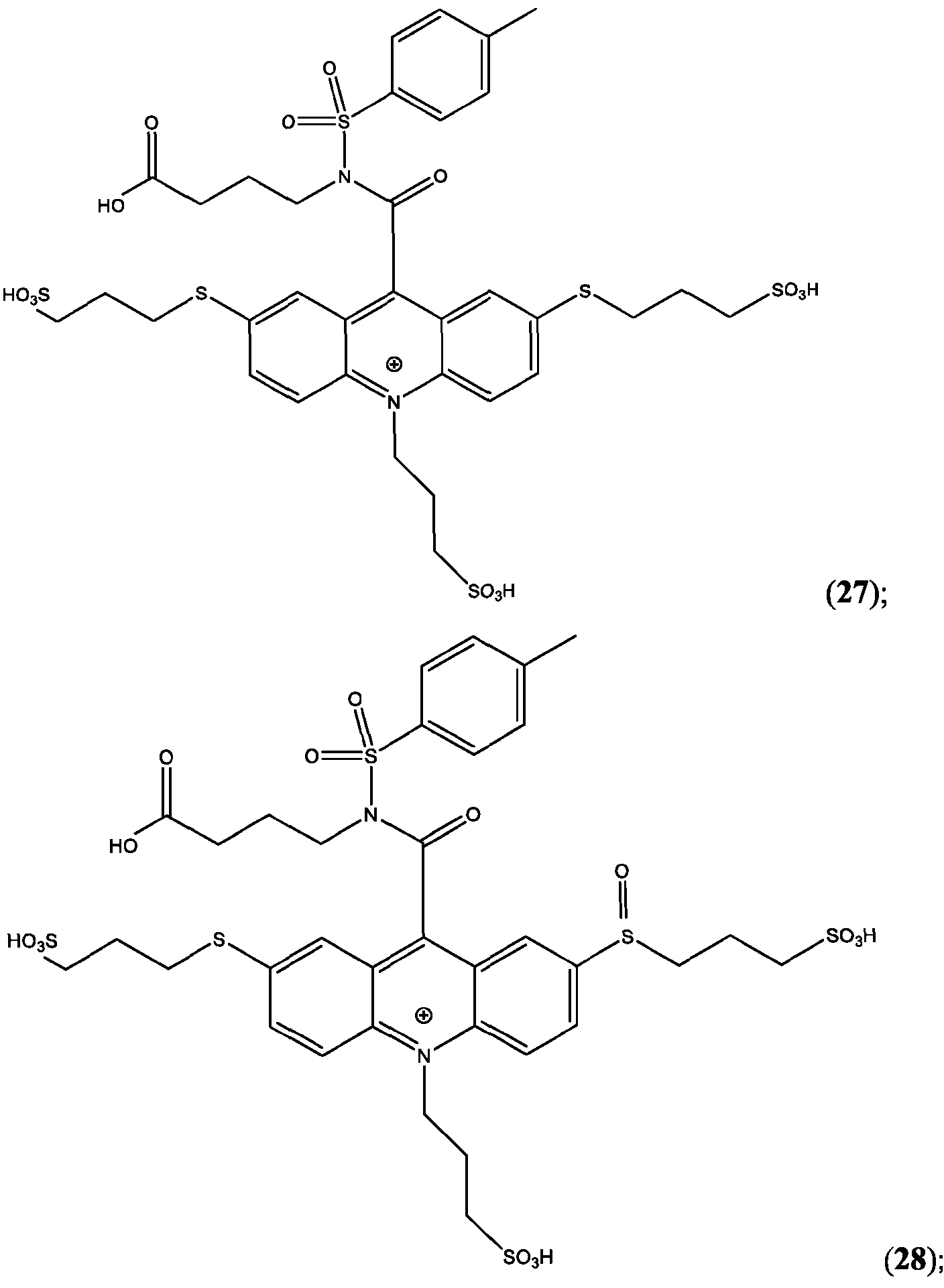
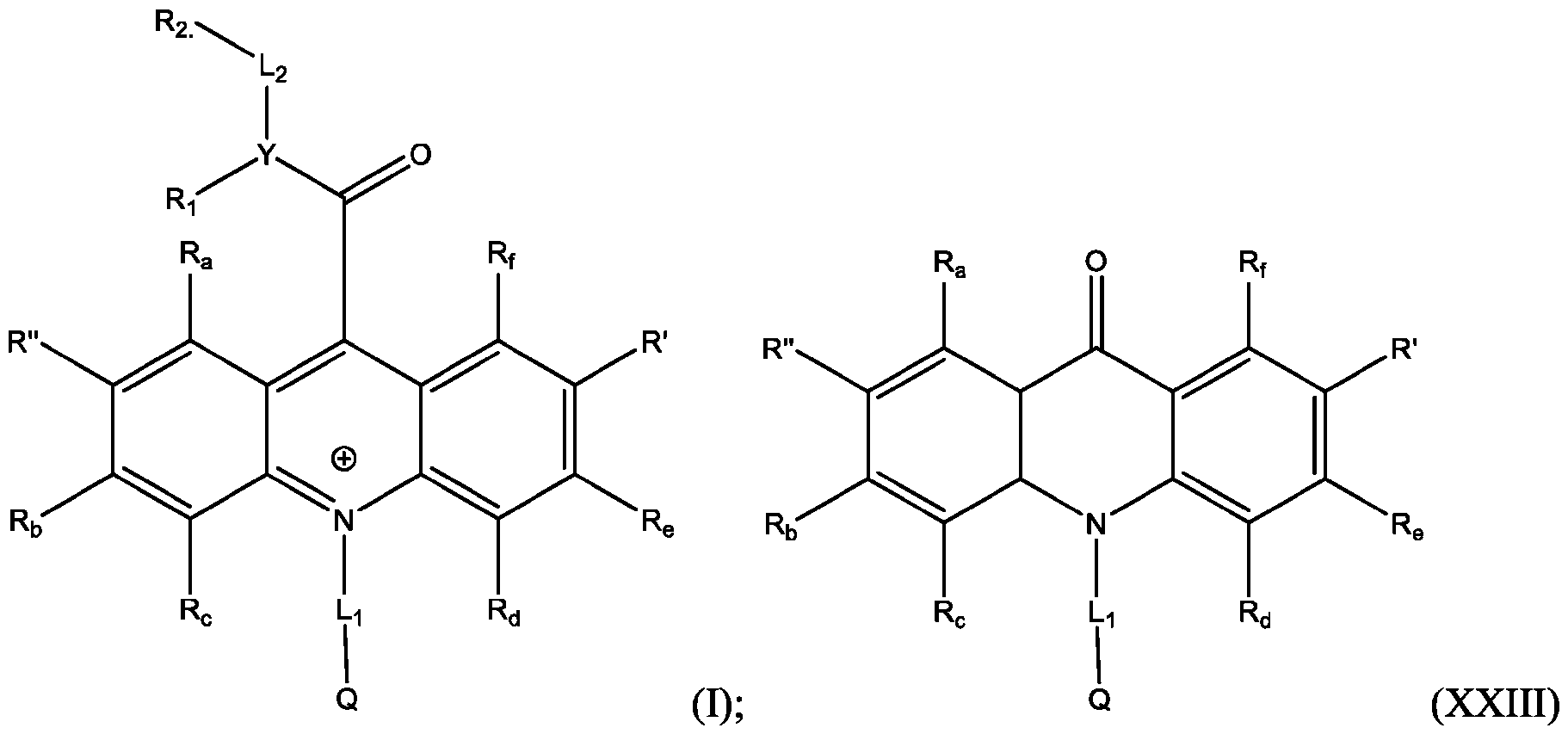
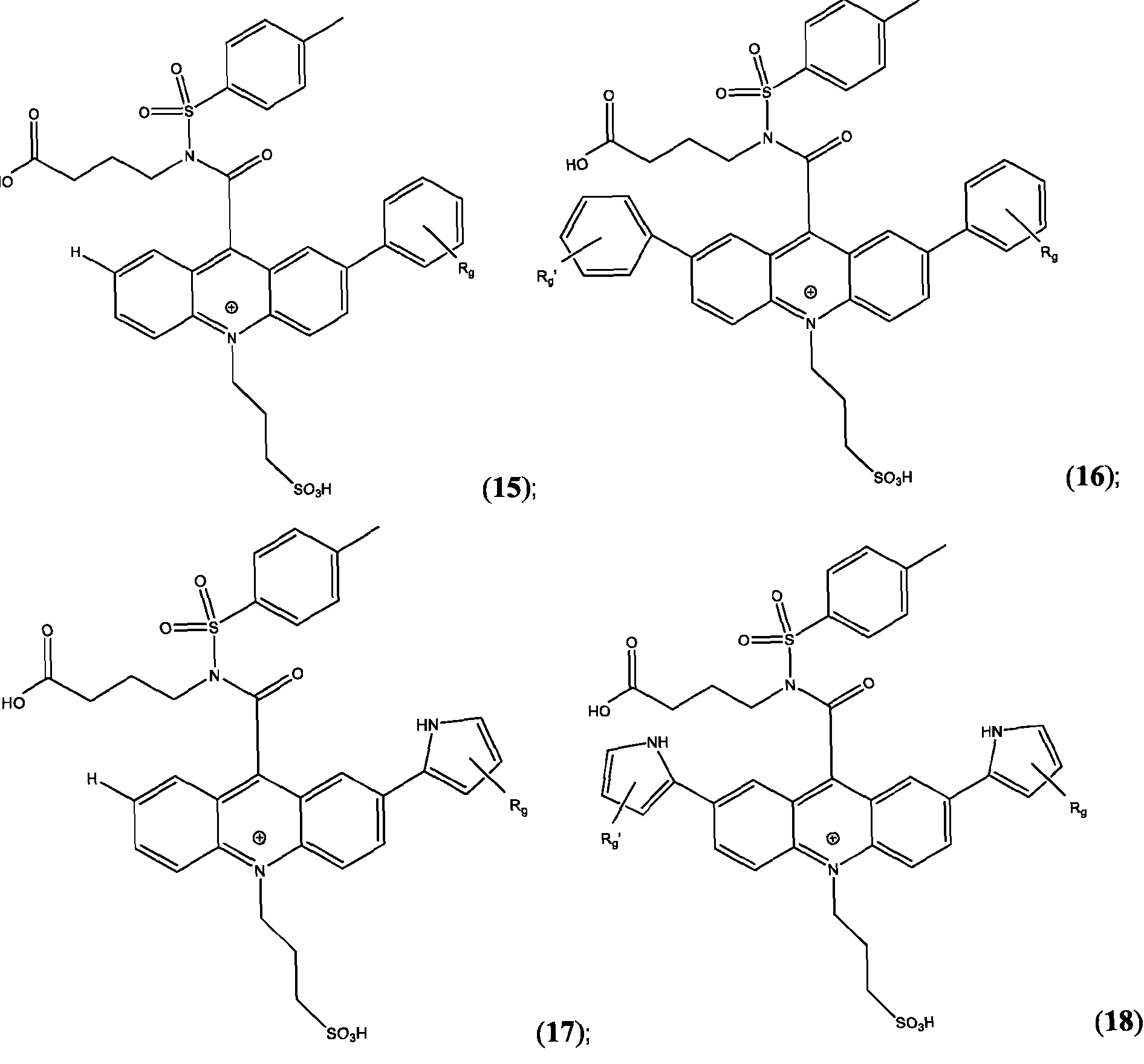
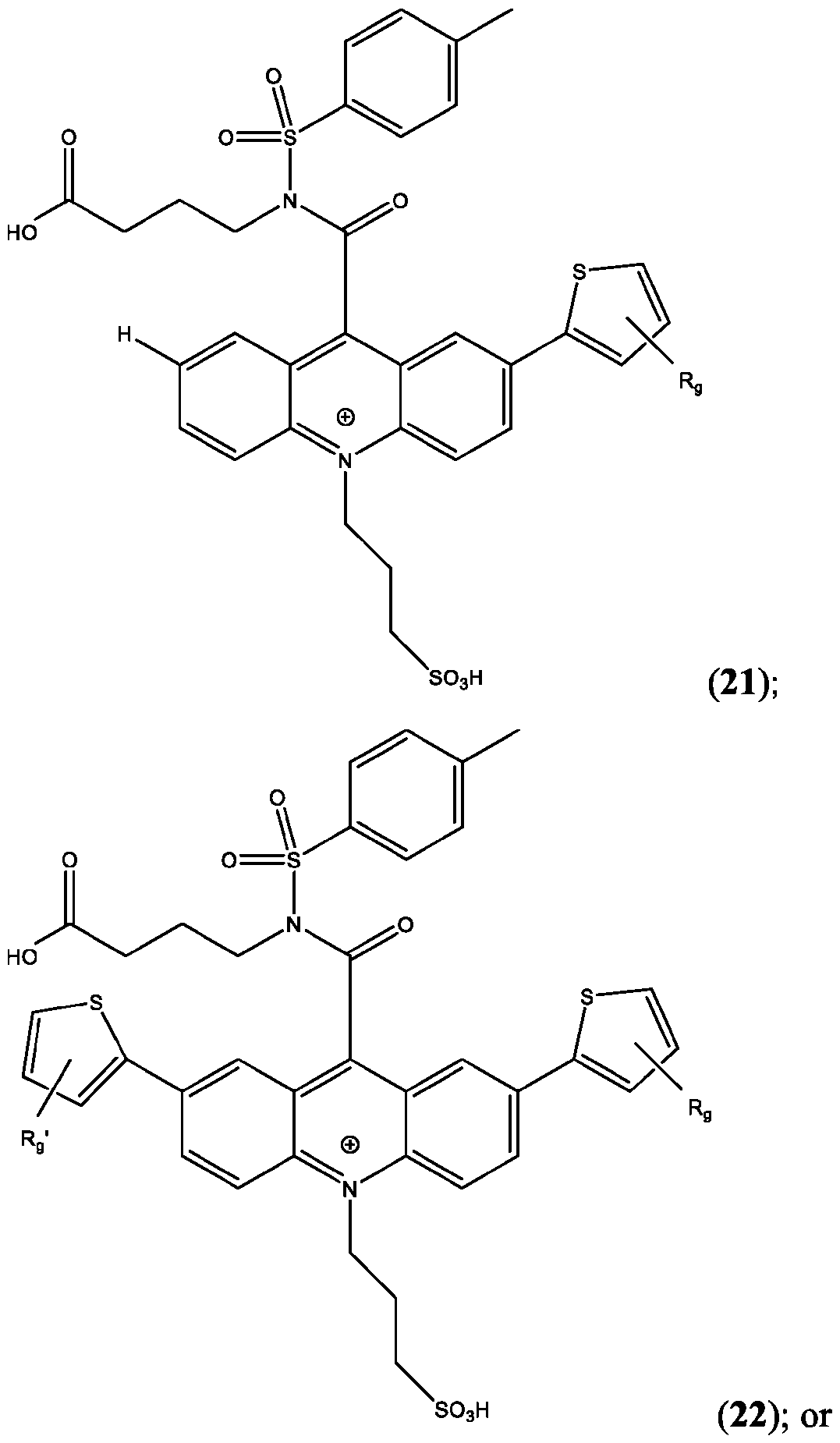
- the compound has the Formula (Ila): wherein R’ and R” are defined above for Formula (I).
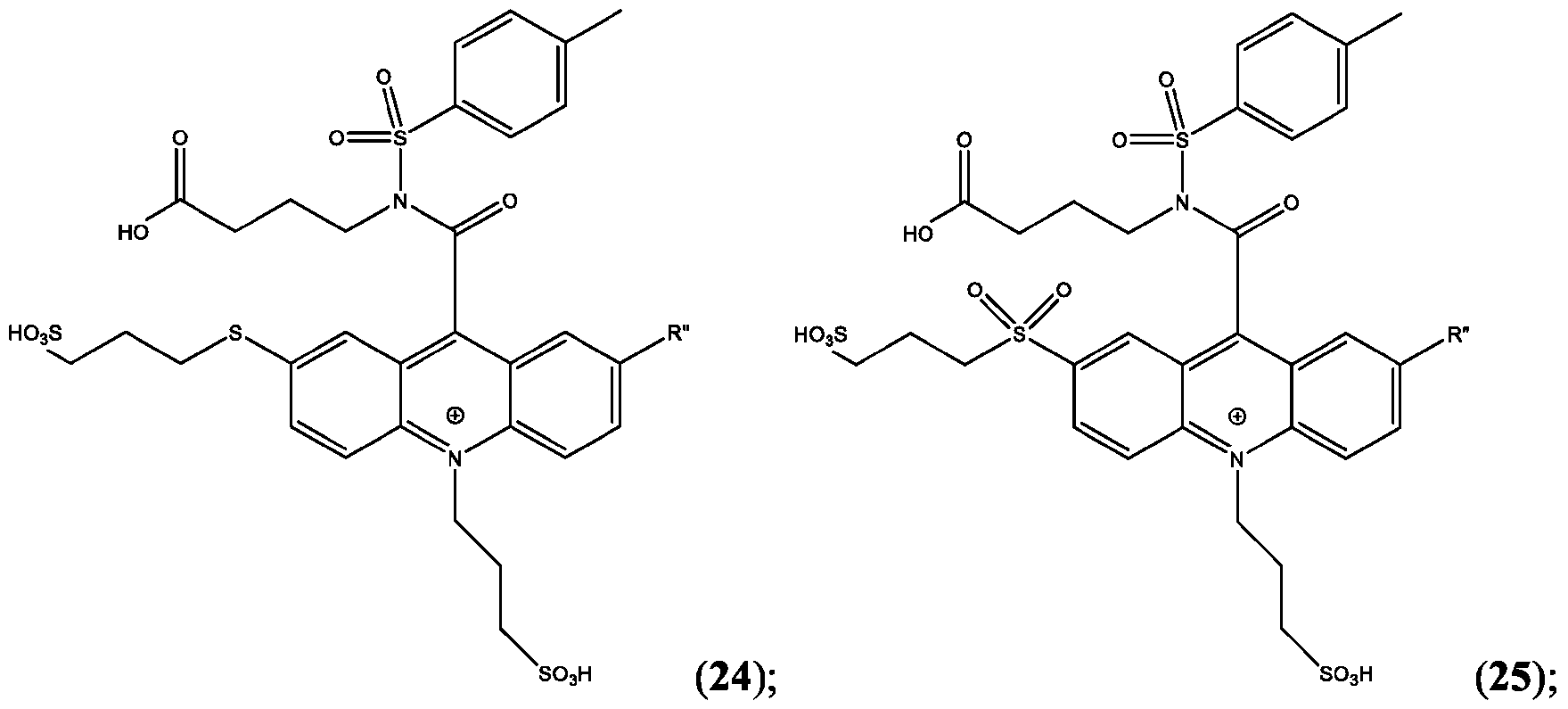
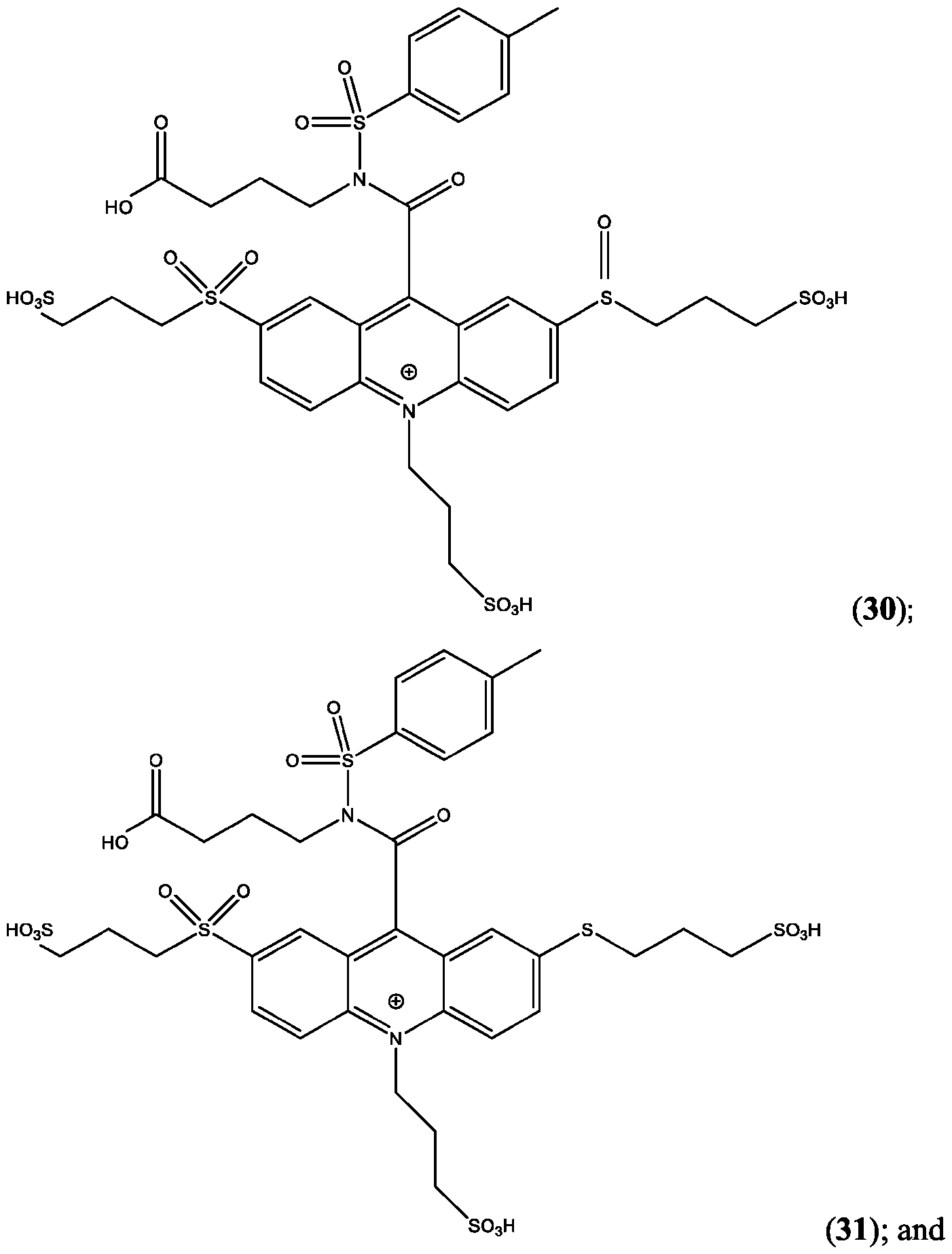
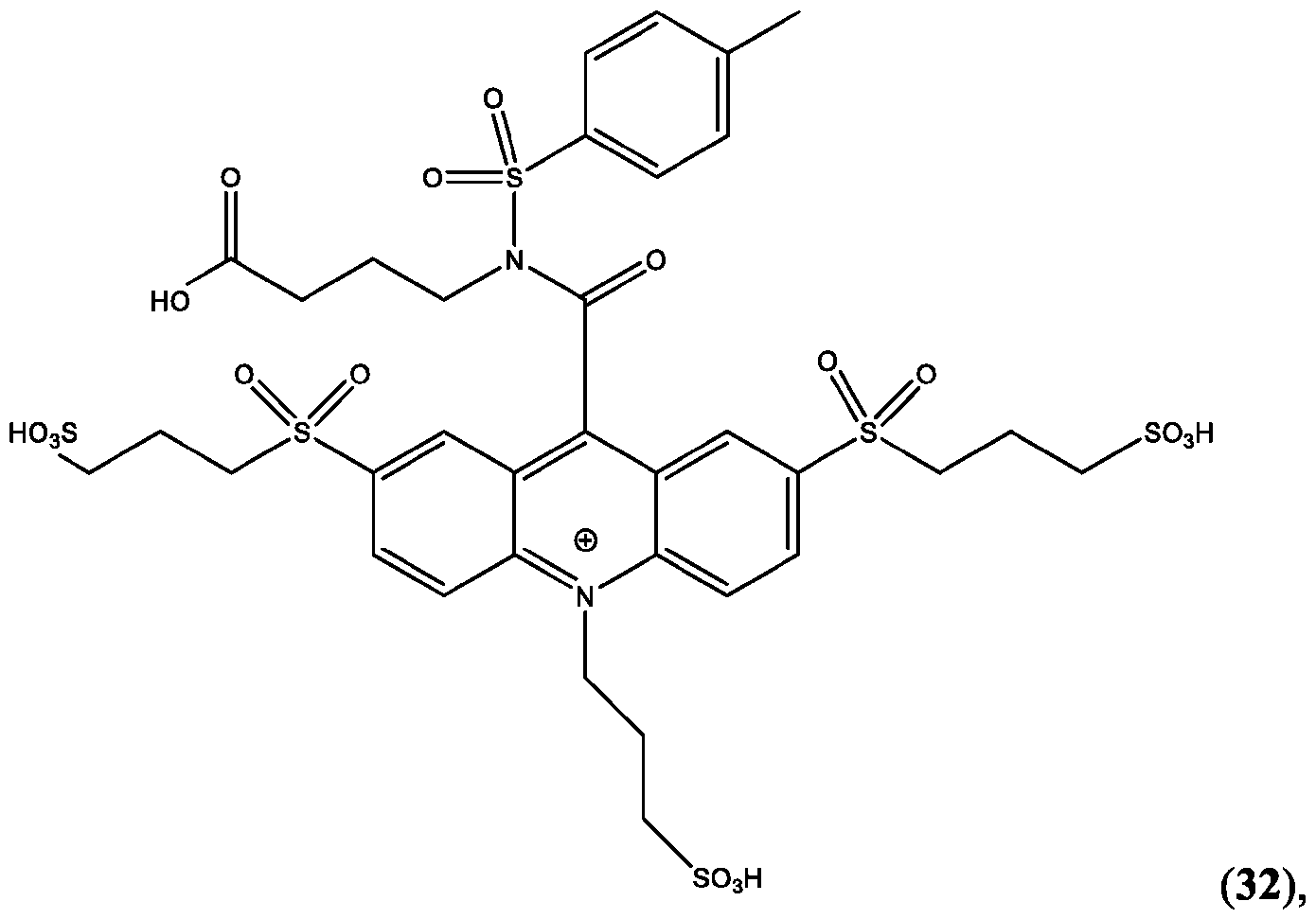
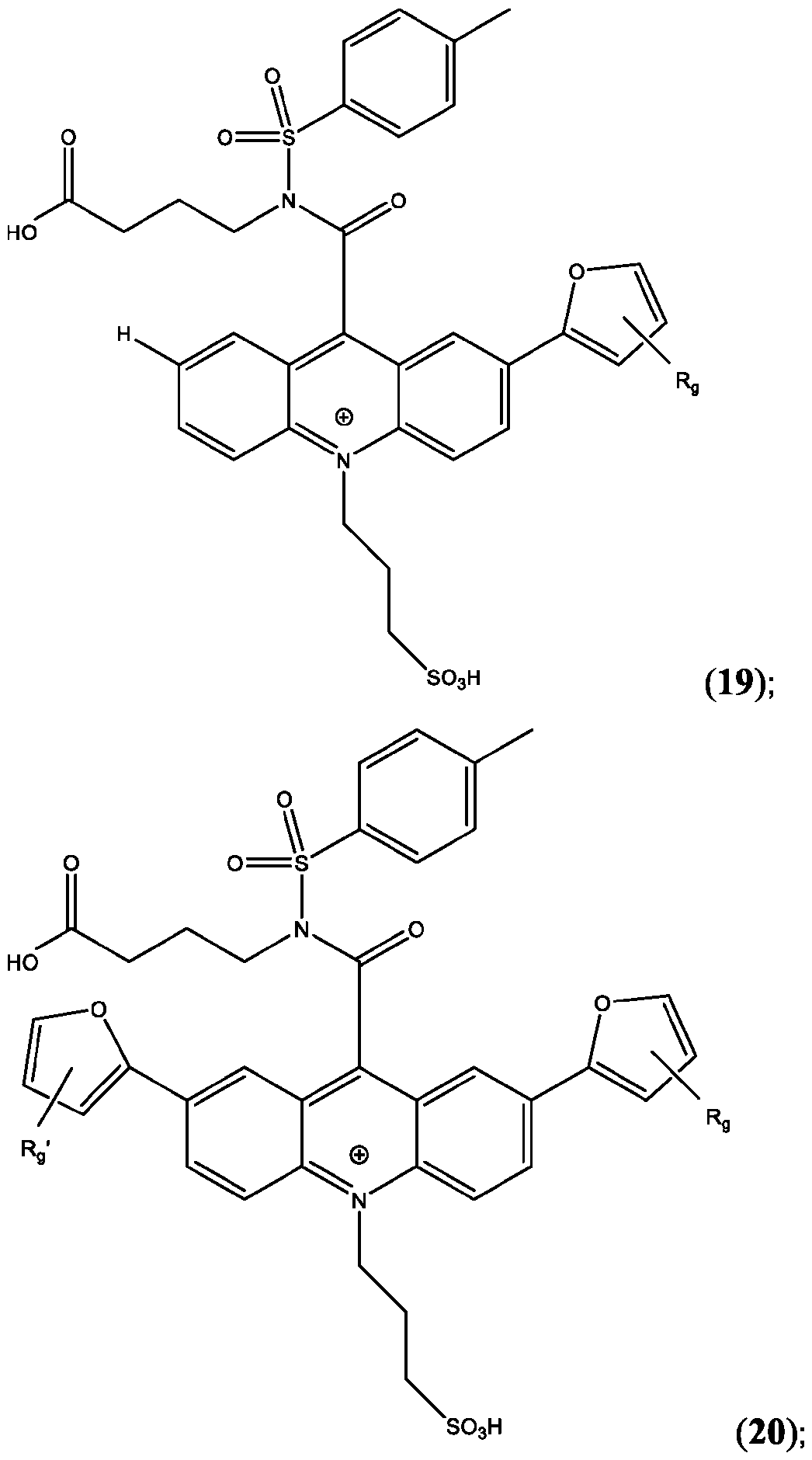
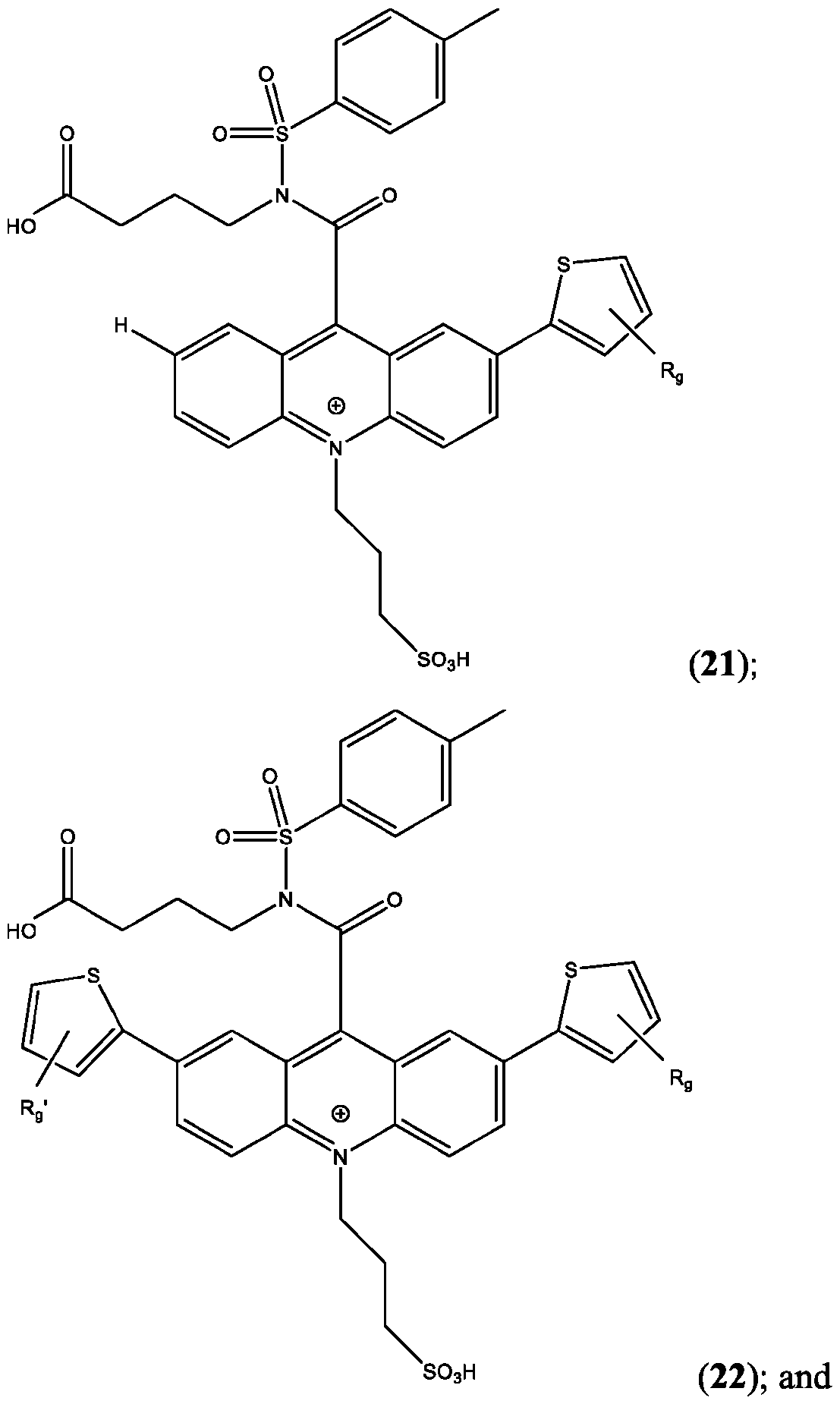
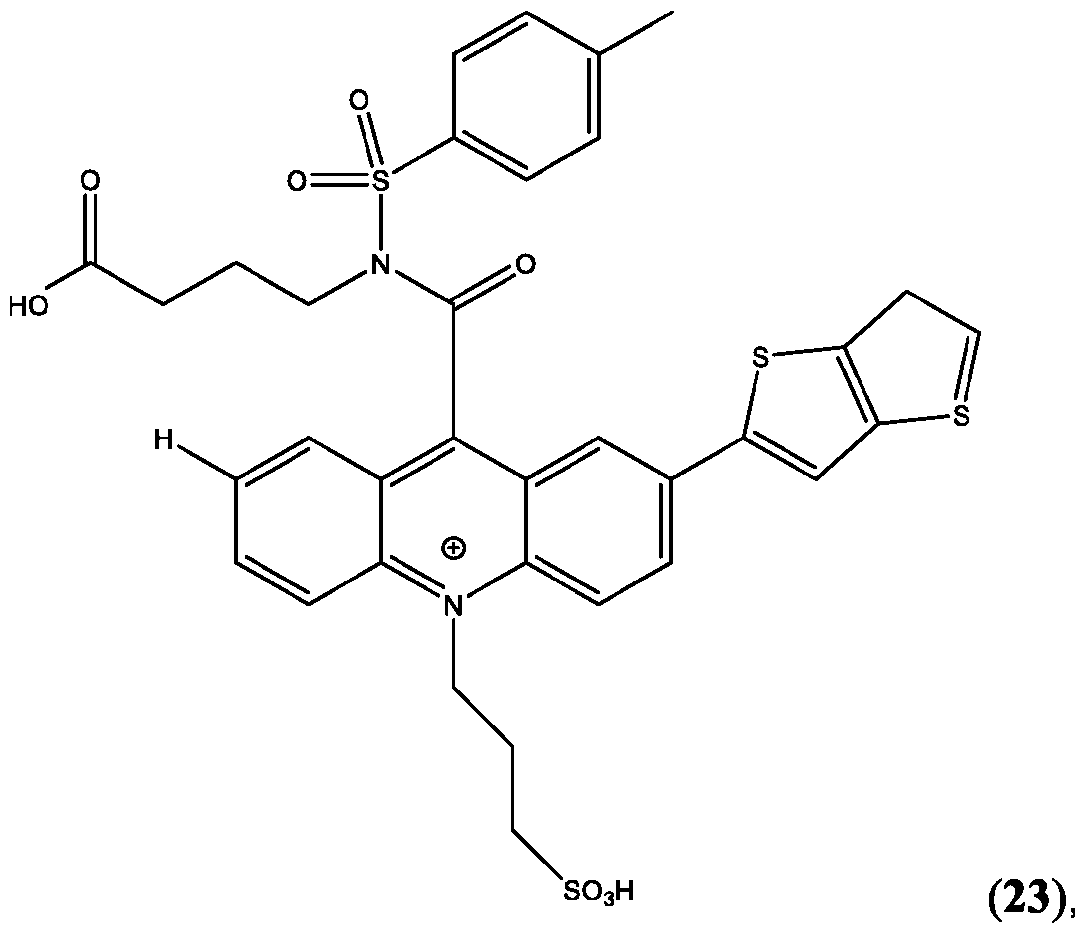
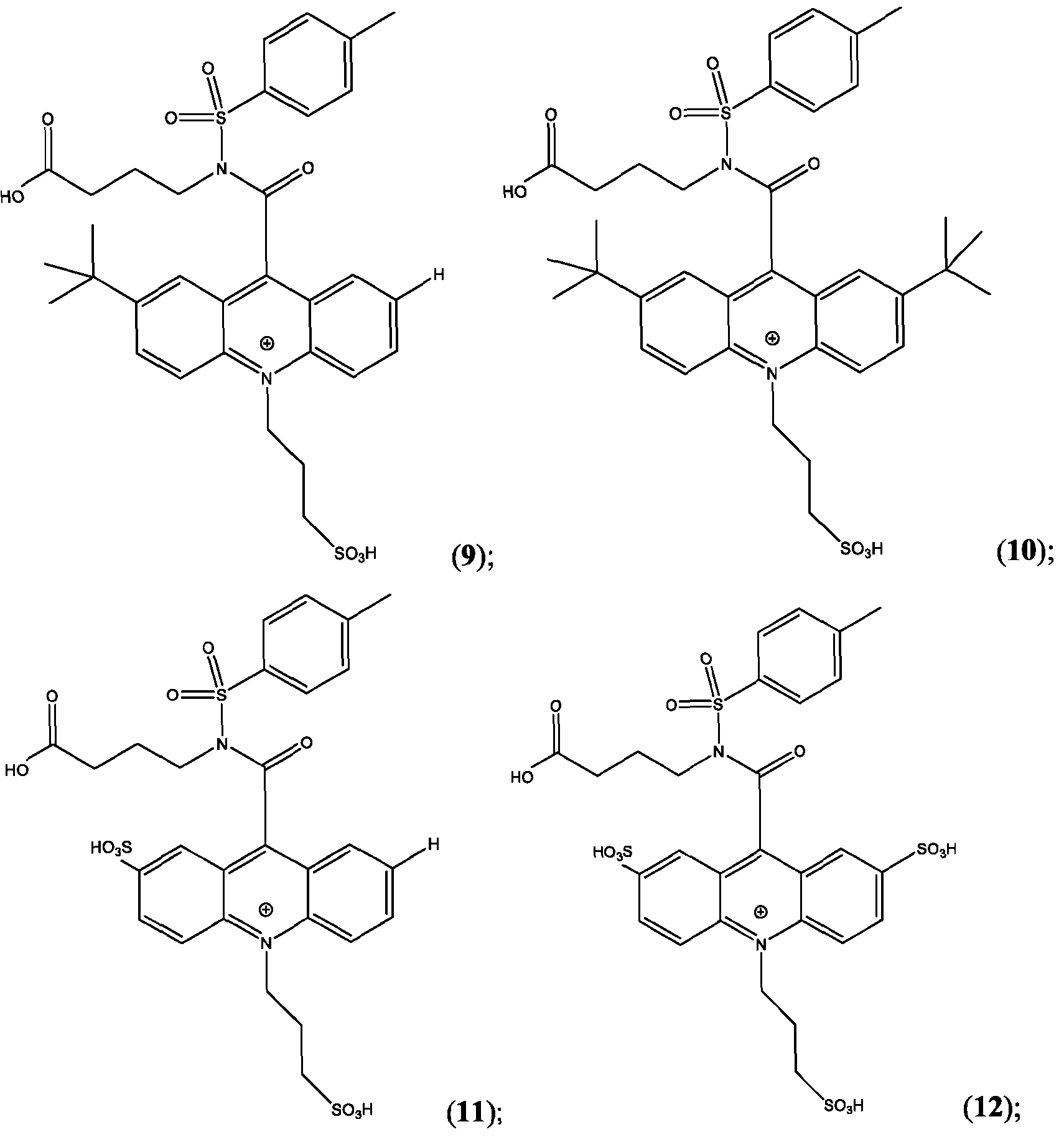
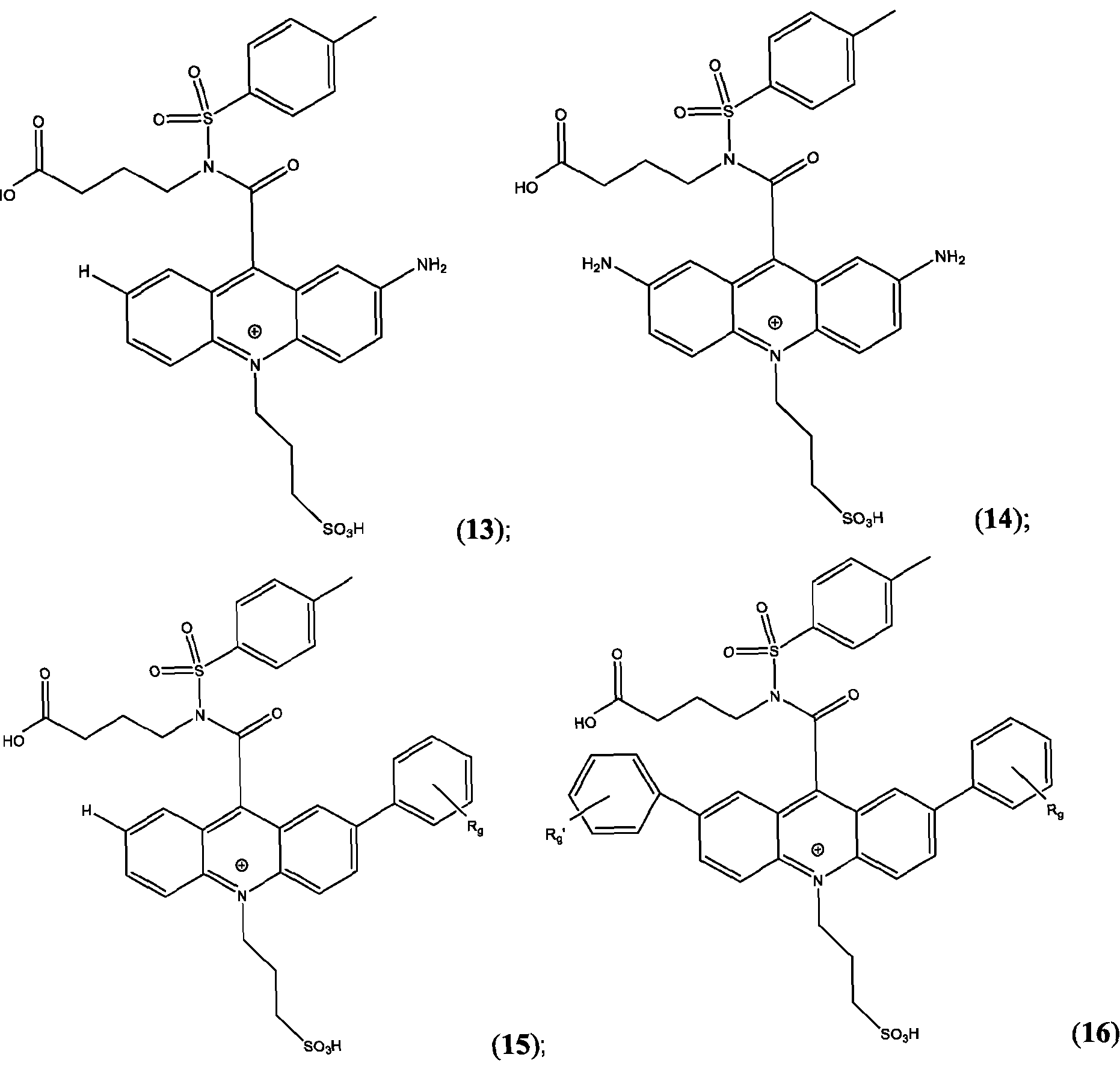
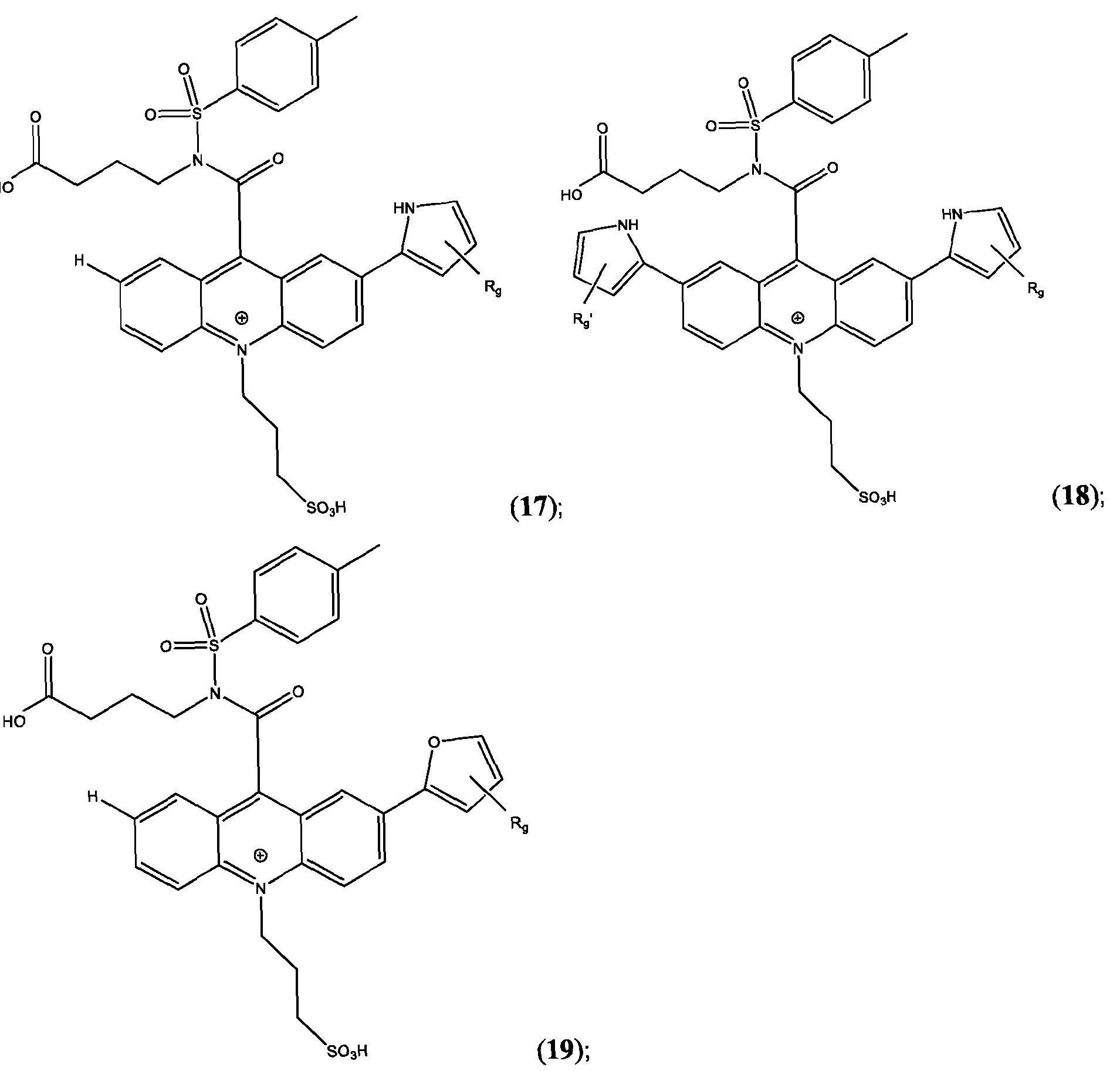
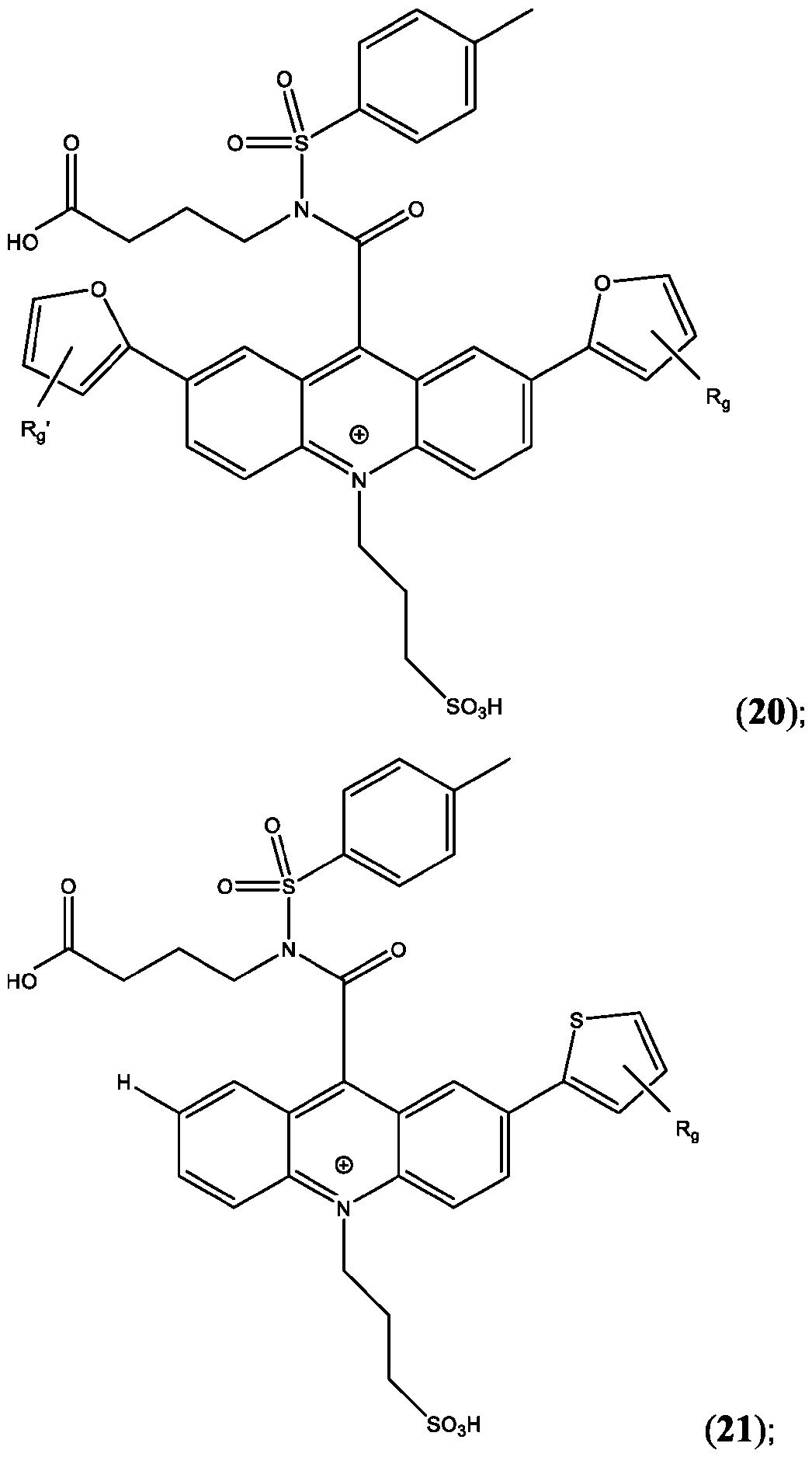
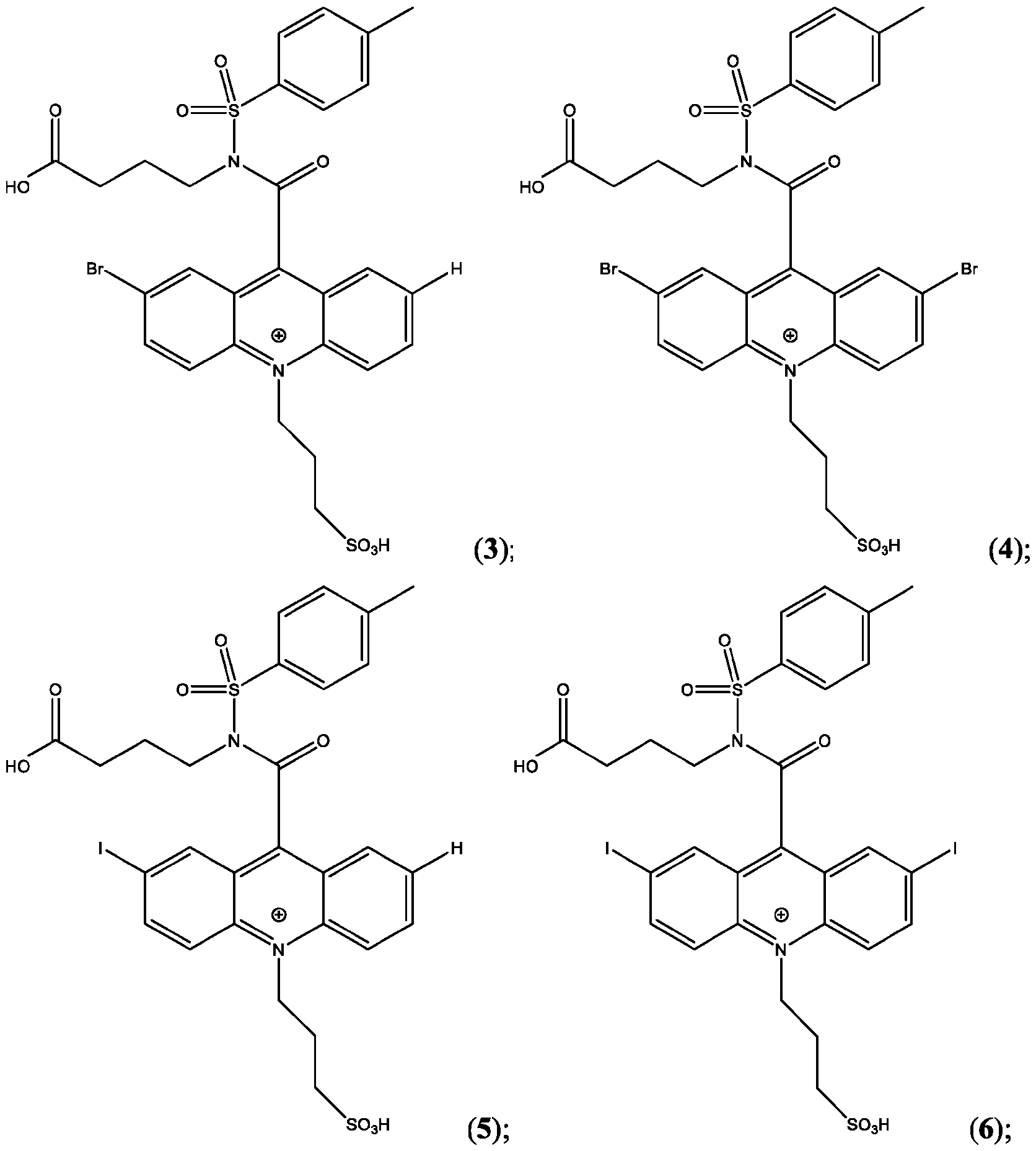
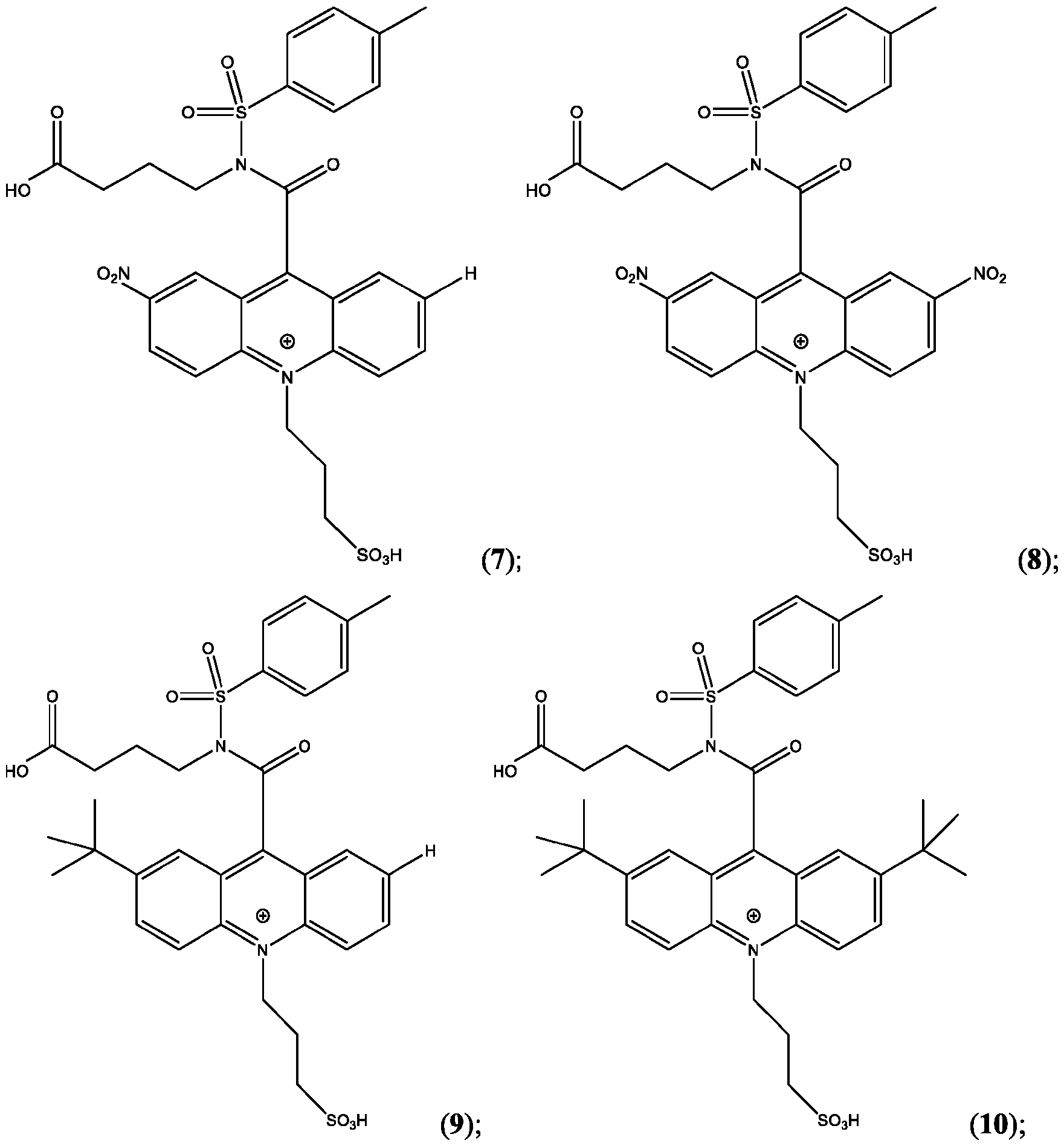
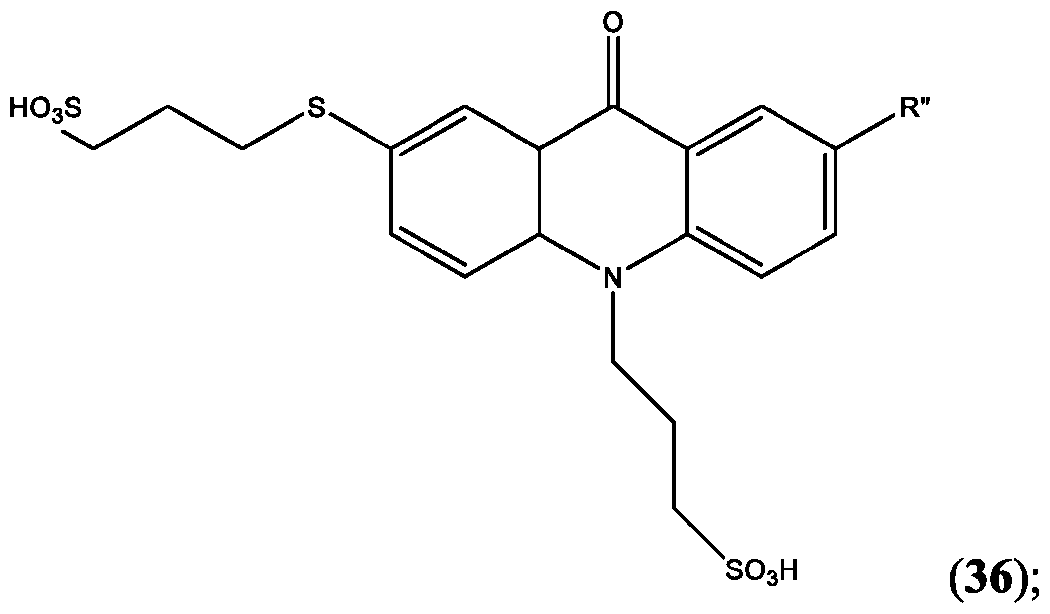
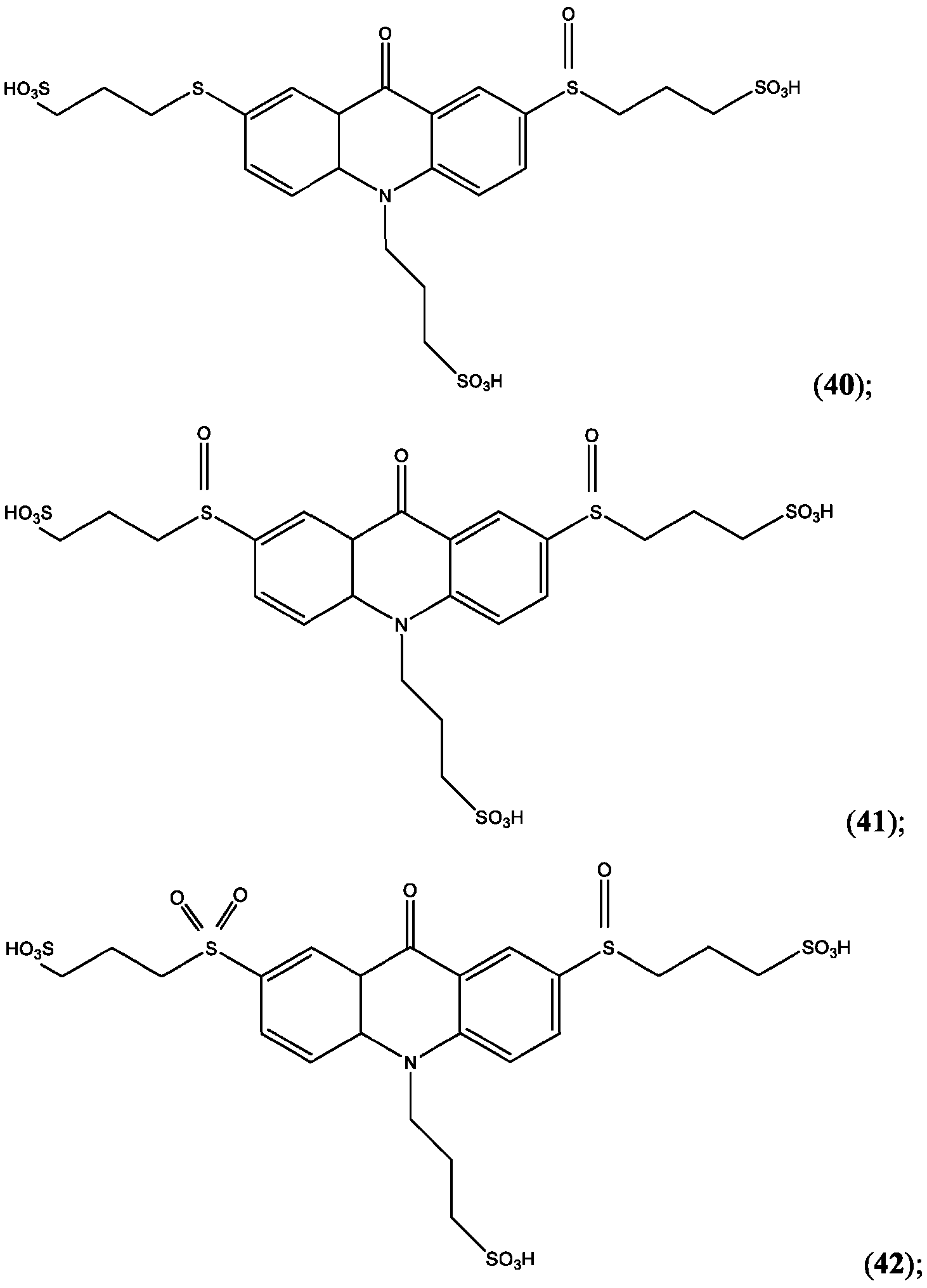
- the compound is selected from:
- the compound is selected from:
- R is as is defined above for Formula (I).
- the method can comprise: reducing the acridinium ring to an acridan intermediate using a first solution comprising at least one reducing agent and at least one acid; and reacting the acridan intermediate with an electrophile to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring.
- the at least one reducing agent comprises zinc or a hydride.
- the zinc comprises elemental zinc, a zinc alloy, or a combination of both.
- the acridinium ring is reduced at temperature in a range from about 50°C to about 90°C, preferably about 60°C to about 80°C.
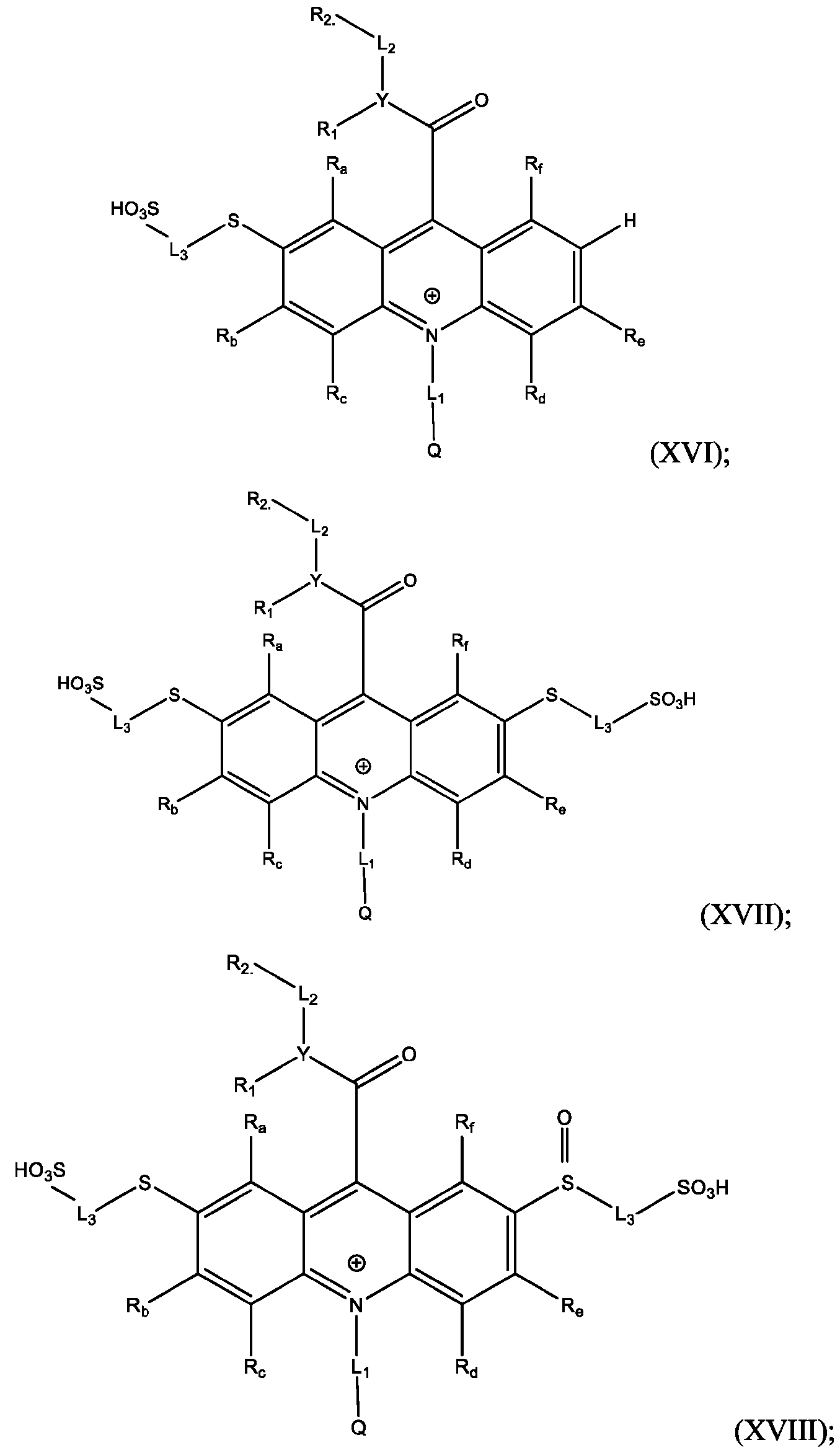
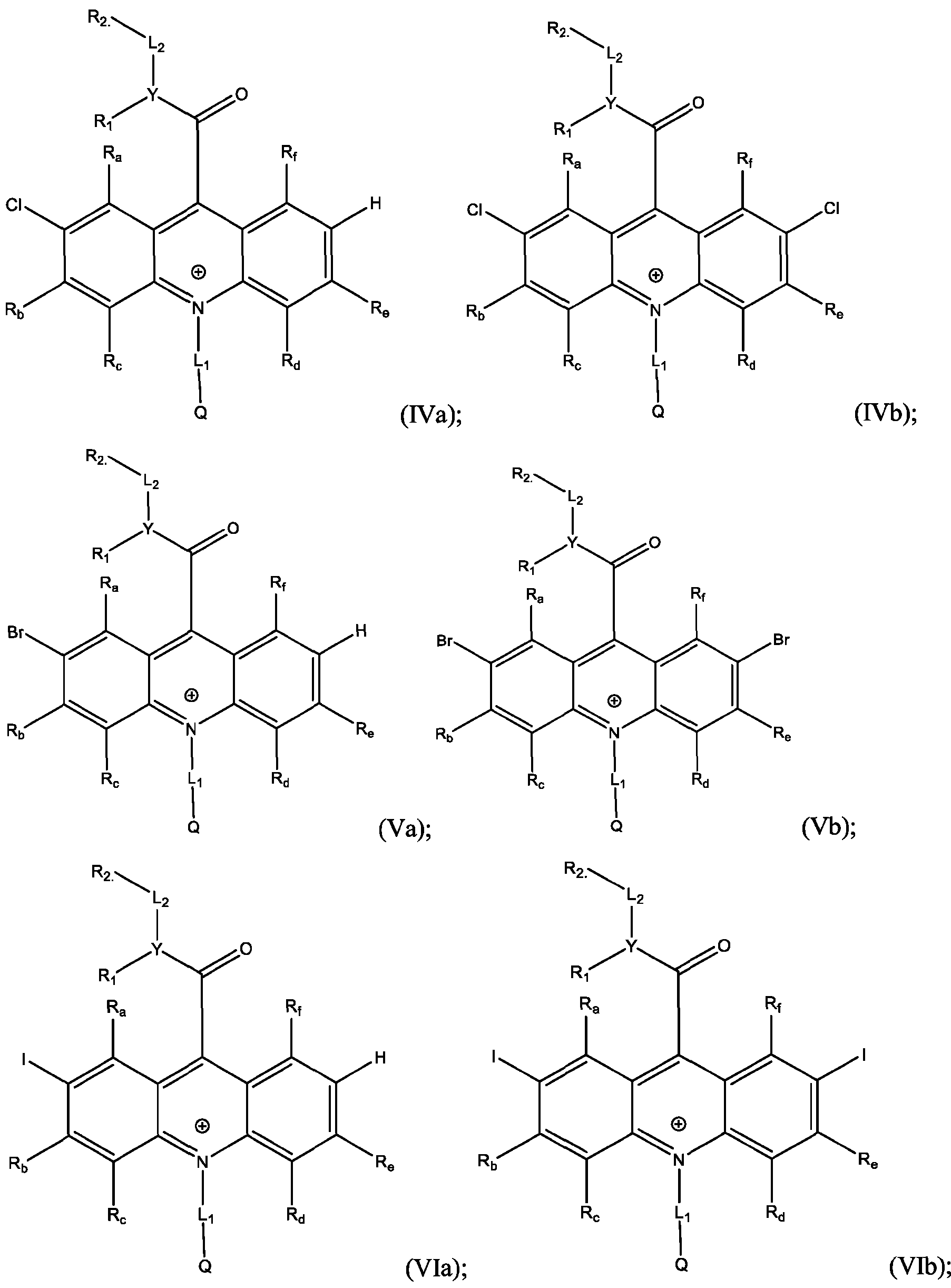
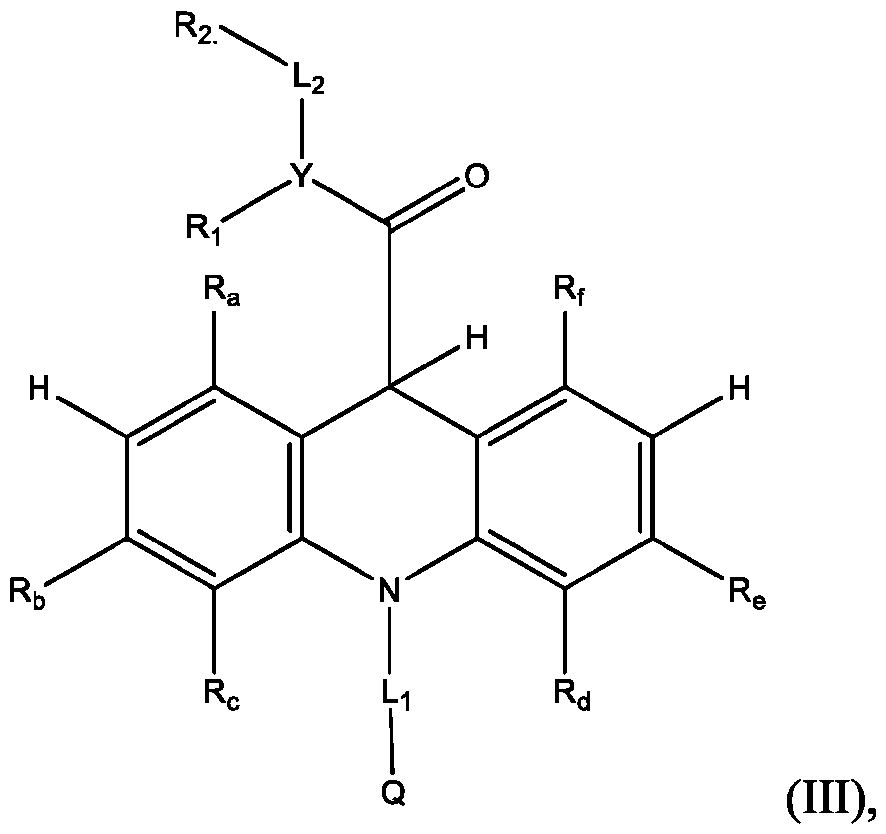
- the acridinium-containing compound is represented by the compound of Formula (I) as described above.
- the acridan intermediate is represented by Formula (III): f are defined above for Formula (I).
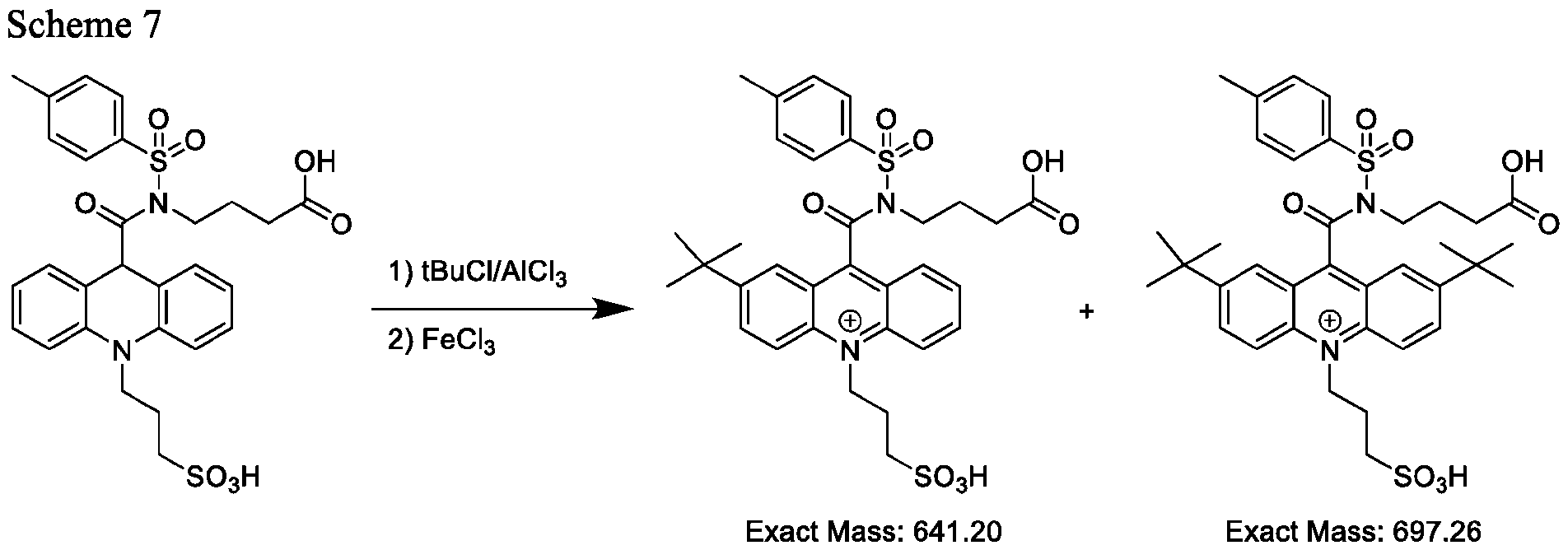
- the electrophile comprises at least one of Cl, Br, I, NO2, SO3H, or t-butyl.
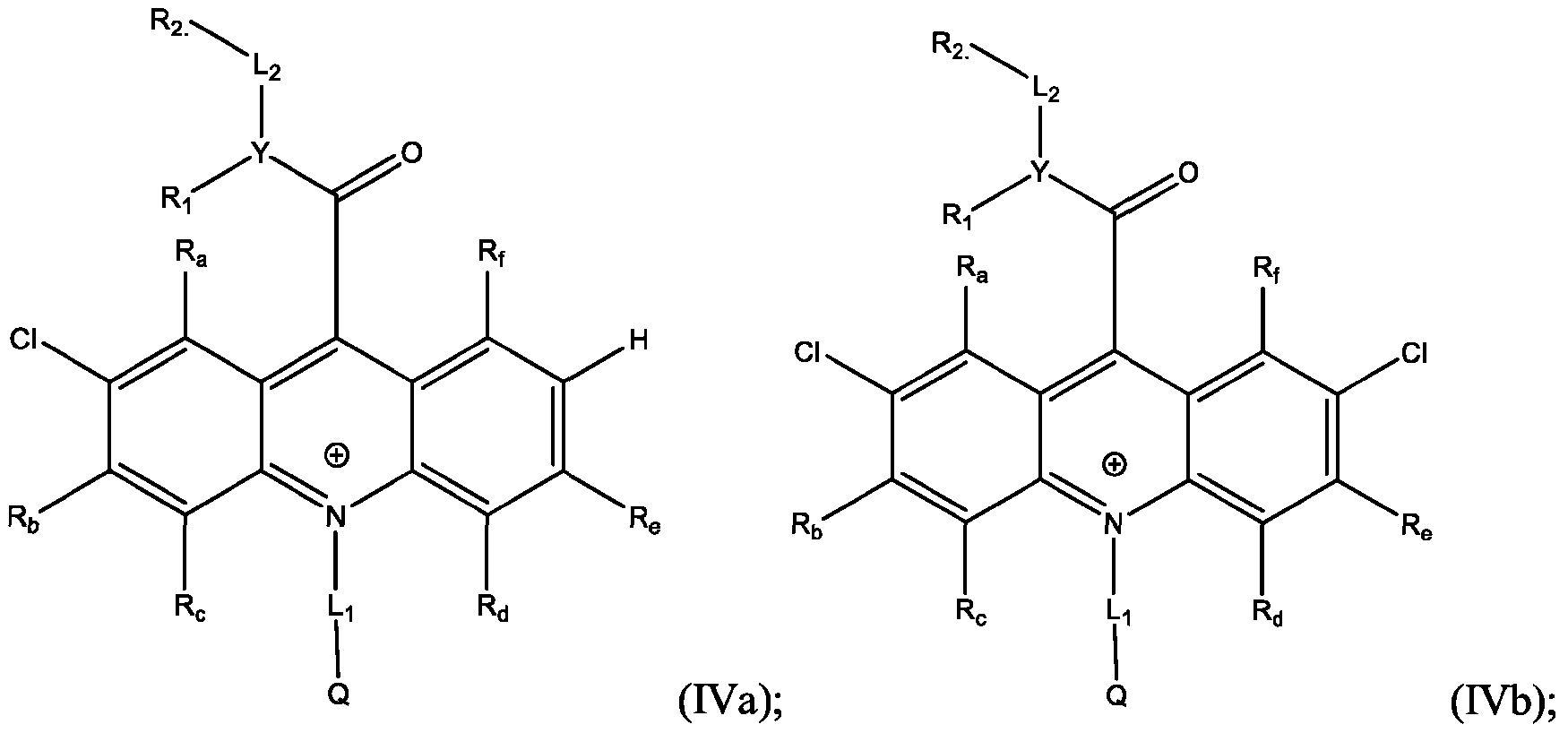
- the electrophile is Cl and the products are Formula (IVa), (IVb), or both (IVa) and (IVb).
- the method further comprises mixing the acridan intermediate of Formula (III) with a second solution in a container, and stirring for about 12 hours to about 24 hours, preferably about 16 hours to about 20 hours; adding an aqueous solution of at least one oxidant to the container, in portions, over about 30 minutes to about 90 minutes, preferably about 45 minutes to about 75 minutes; and isolating and optionally purifying the product comprising the compound of Formula (IVa), (IVb), or both (TVa) and (IVb).
- the second solution comprises a chloride salt, N-chlorosuccinimide, and p-toluenesulfonic acid in a water/acetonitrile mixture.
- the oxidant comprises FeCh, 2,3- Dichloro-5,6-dicyano-l,4-benzoquinone (DDQ), chloranil, or ceric ammonium nitrate (CAN).
- DDQ 2,3- Dichloro-5,6-dicyano-l,4-benzoquinone
- CAN ceric ammonium nitrate
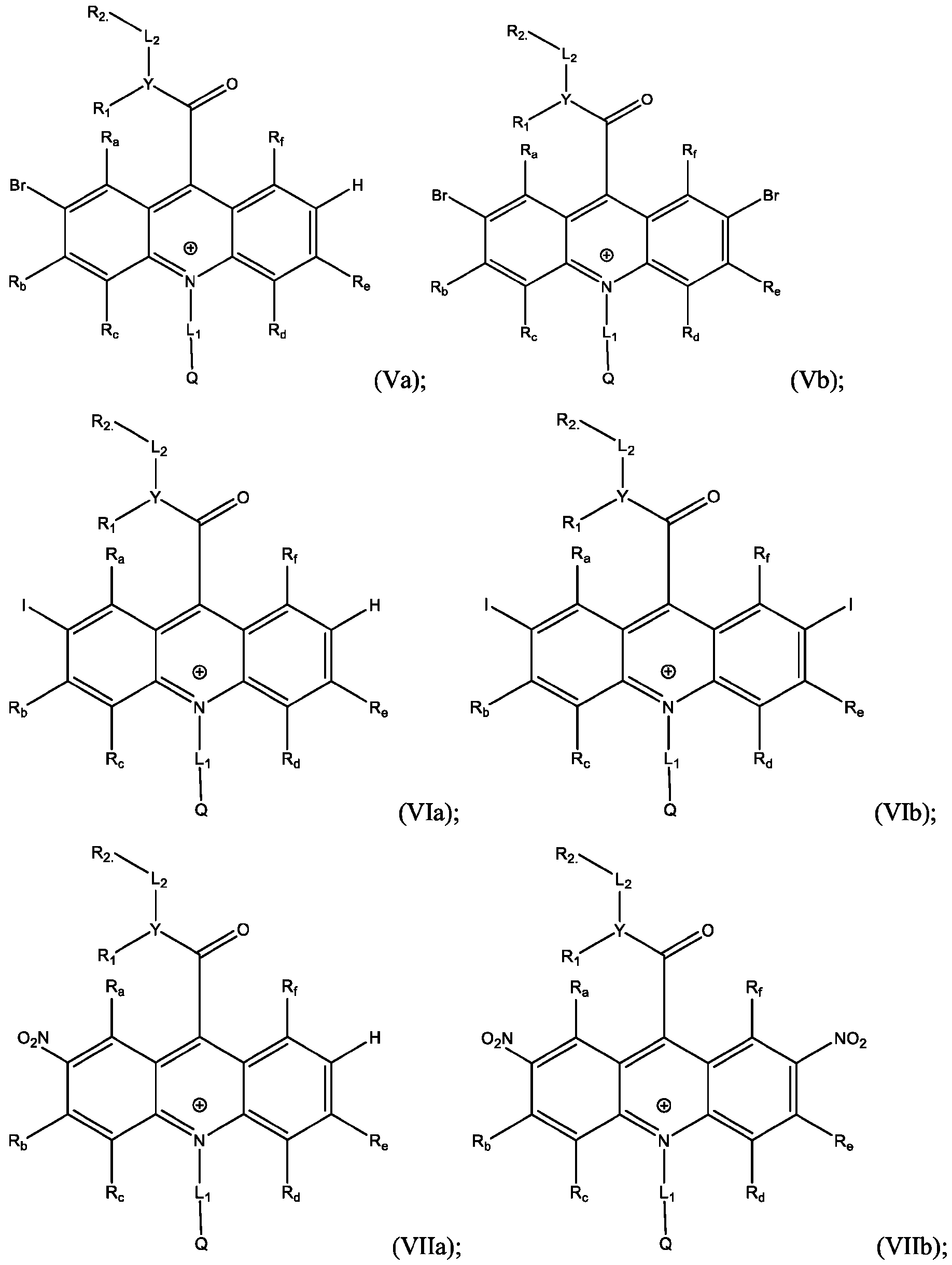
- the electrophile is Br and the products are Formula (Va), (Vb), or both (Va) and (Vb).
- a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with Br at the 2- or the 2,7- position, or both, of the acridinium ring includes a bromination reaction.
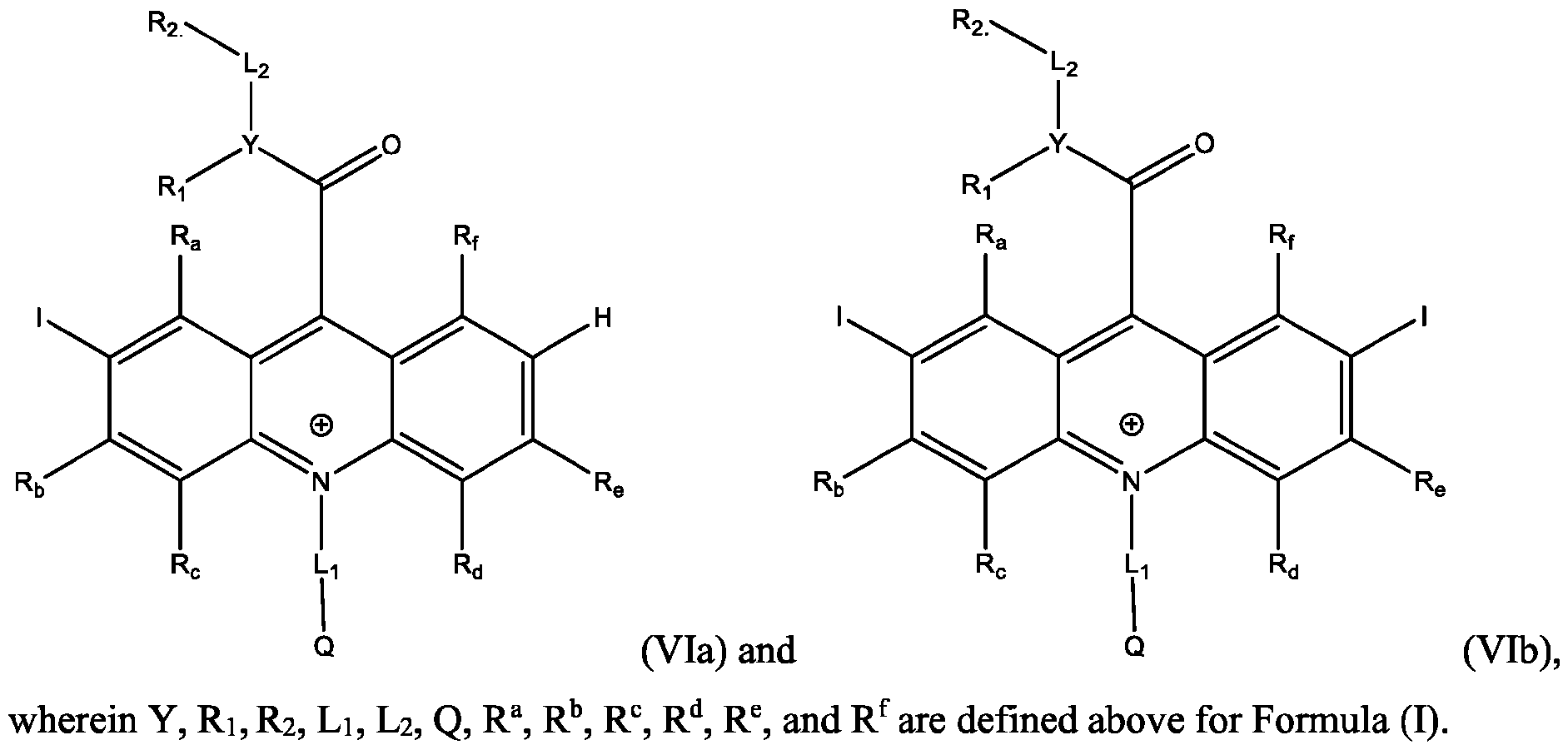
- the electrophile is I and the products are Formula (Via), (VIb), or both (Via) and (VIb).
- a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with I at the 2- or the 2,7- position, or both, of the acridinium ring includes an iodination reaction.
- the method further comprises dissolving the acridan intermediate in a third solution in a container; adding iodine monochloride to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb).
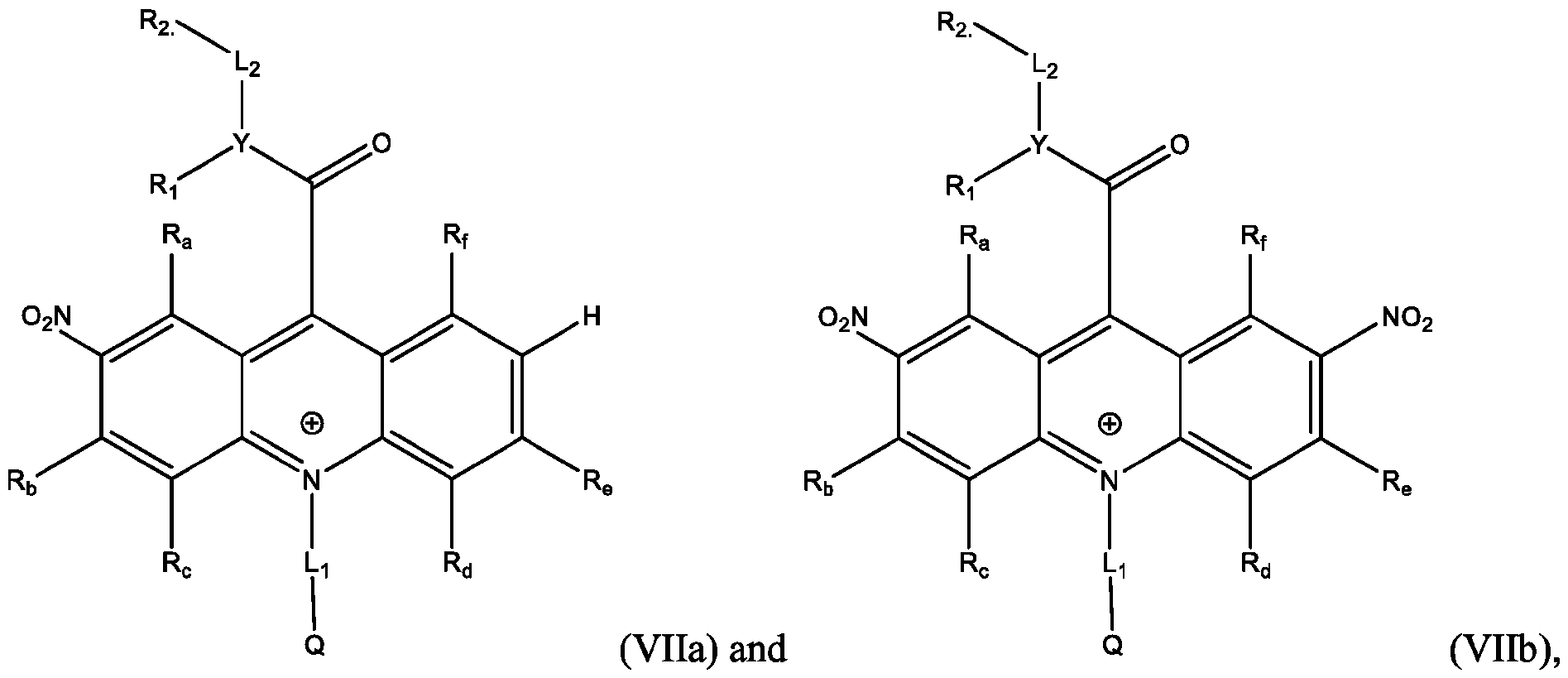
- the electrophile is NO2 and the products are Formula (Vila), (Vllb), or both (Vila) and (Vllb).
- the electrophile is SO3H and the products are Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
- a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with SO3H at the 2- or the 2,7- position, or both, of the acridinium ring includes a sulfonation reaction.
- the method further comprises: dissolving the acridan intermediate in a fourth solution in a container; adding chlorosulfonic acid to the container and stirring for about 5 minutes to about 30 minutes; adding water to the container and stirring for about 10 minutes to about 30 minutes; and isolating and optionally purifying the product comprising the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
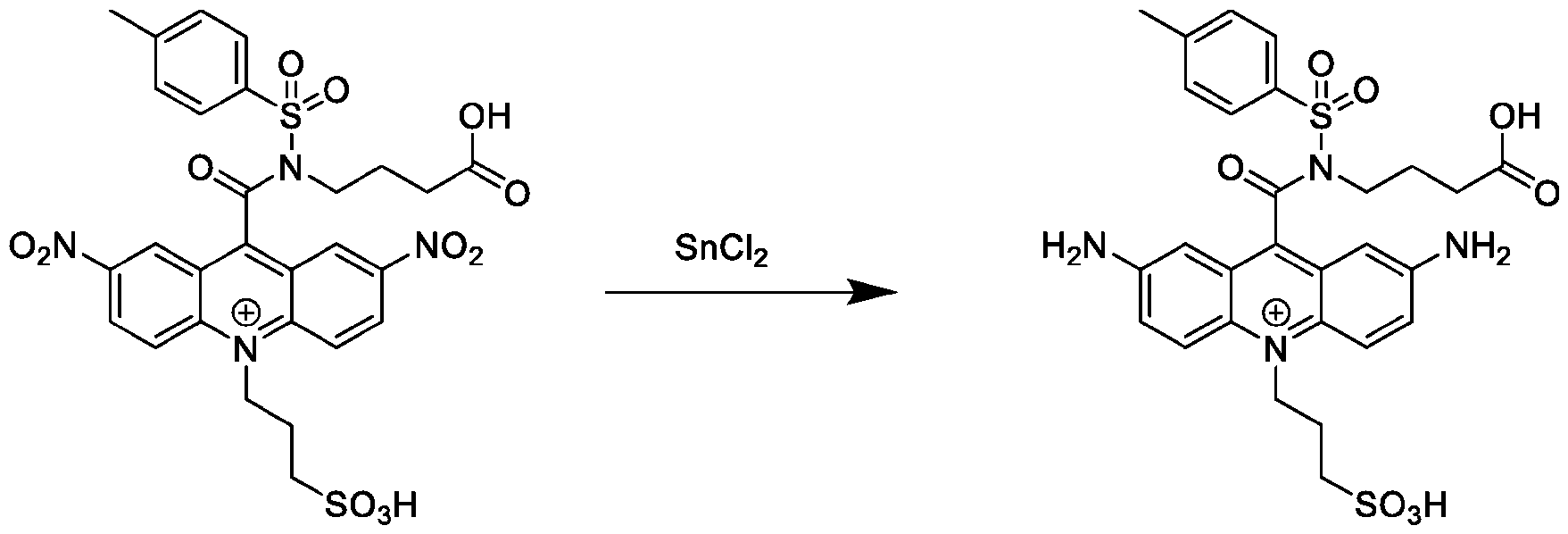
- the reducing agent comprises SnCh-
- Also provided herein are methods of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring comprising: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a boronic acid comprising A’ or a boronate ester comprising A’, wherein A’ is a substituted or unsubstituted aryl group or a substituted or unsubstituted heteroaryl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
- the acridinium compound is functionalized by:
- the oxidizing agent comprises mCPBA.
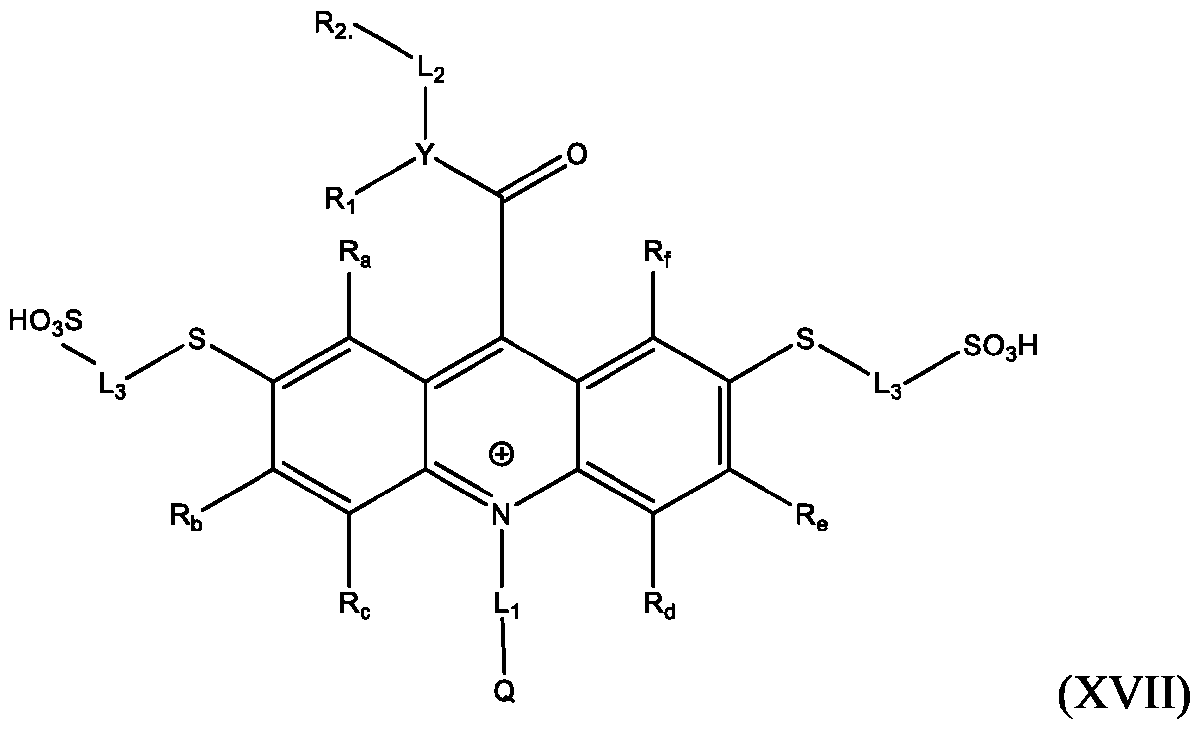
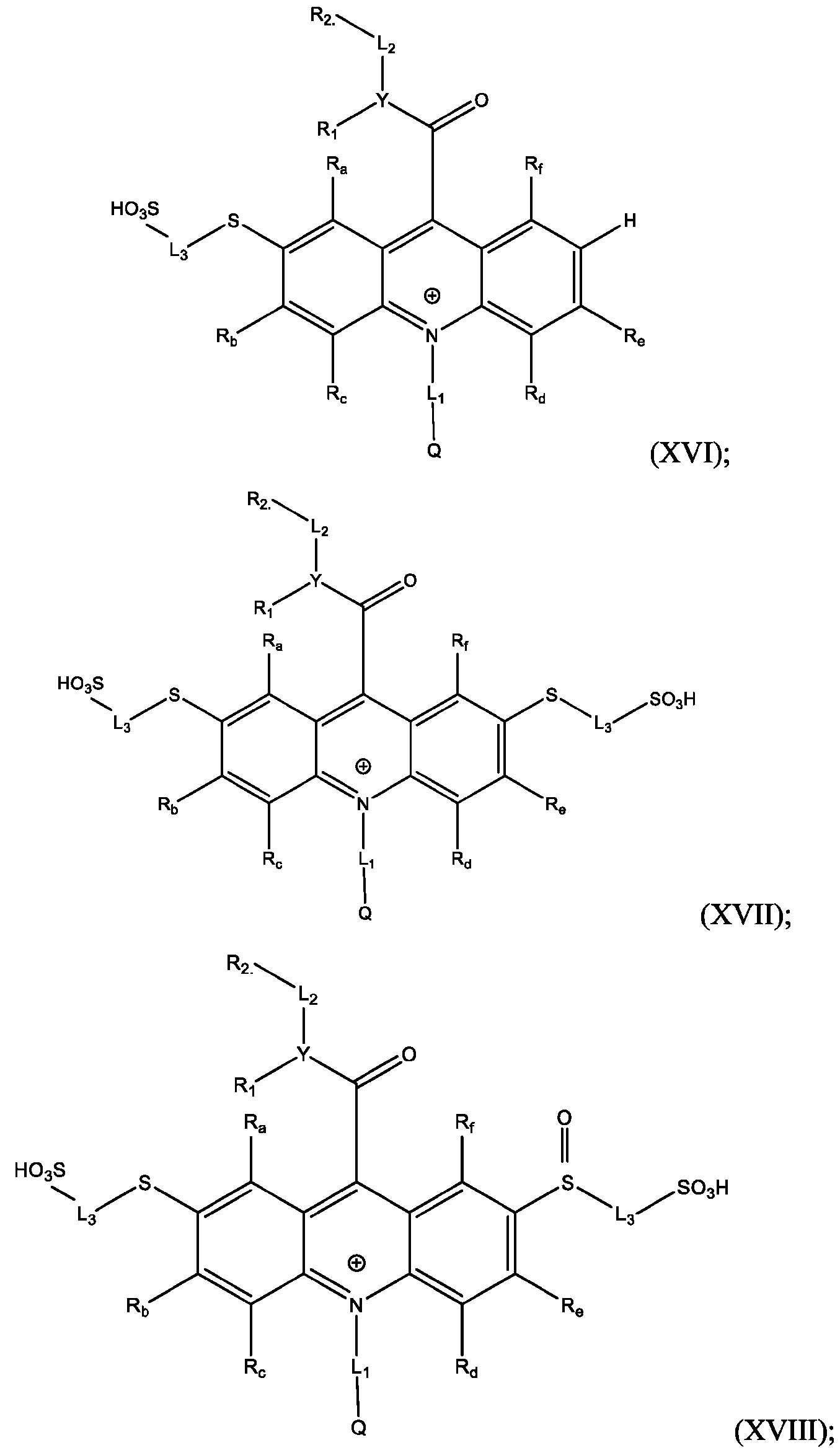
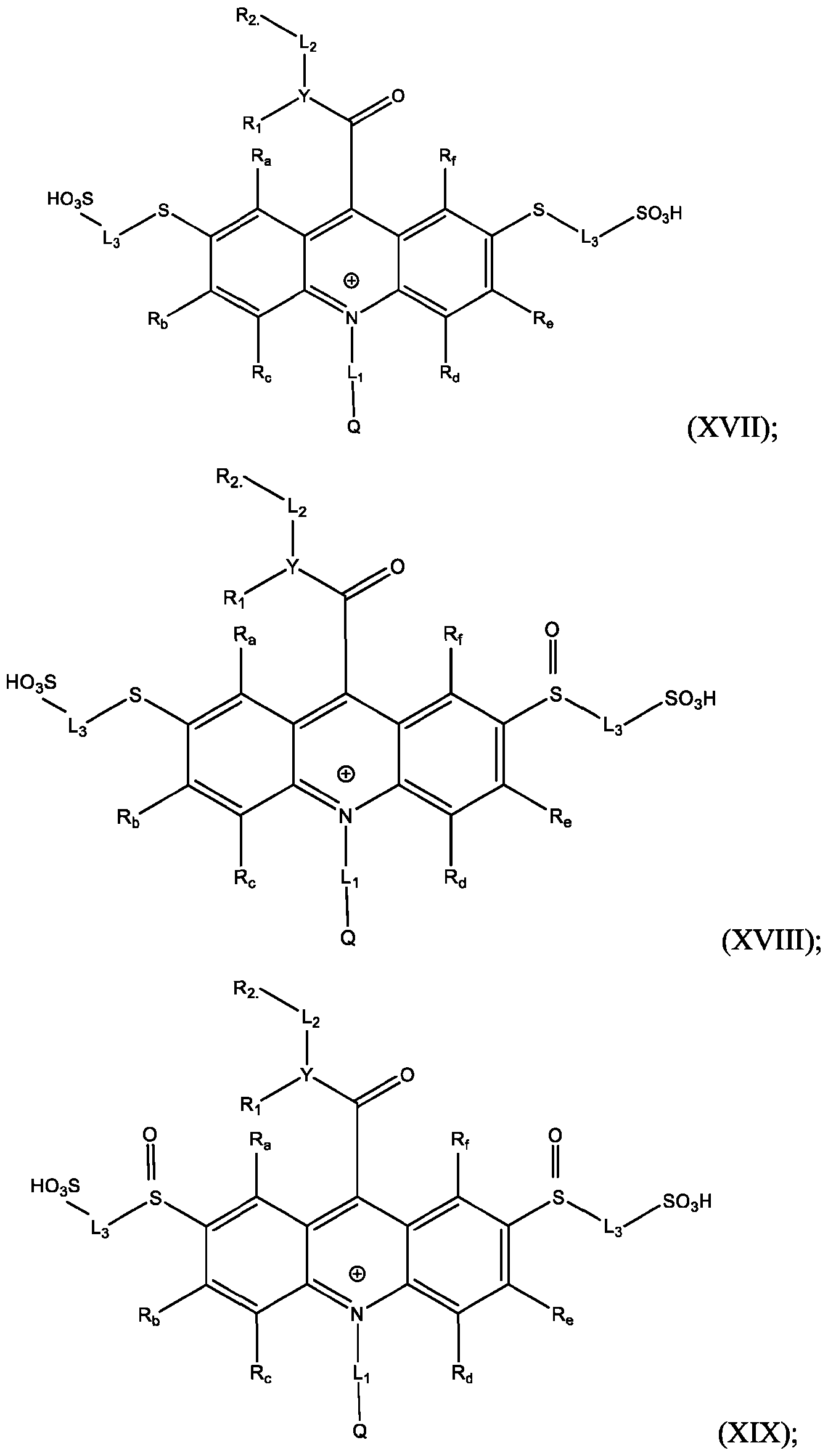
- the compound of Formula (XVII) is CPSP3.
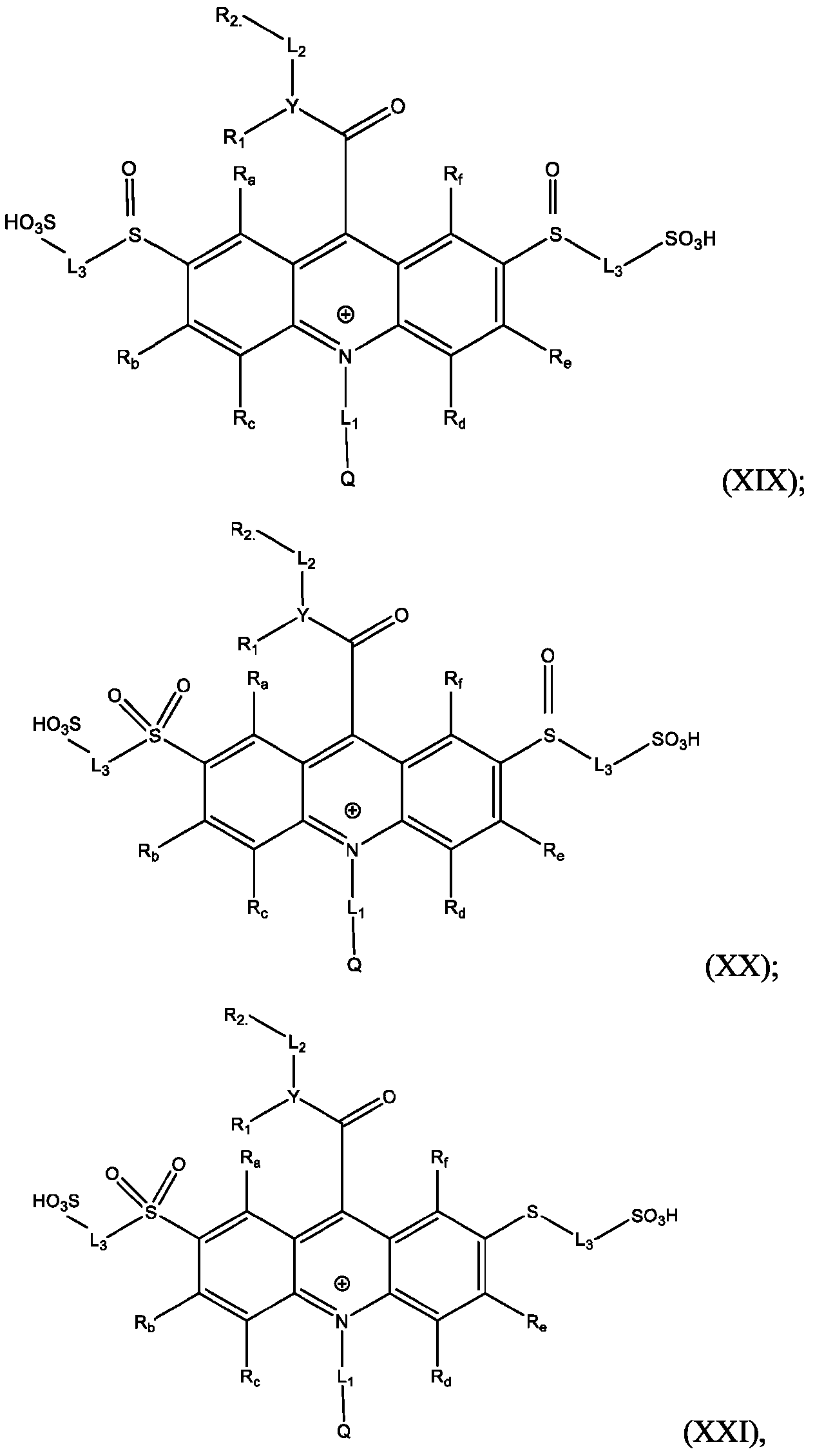
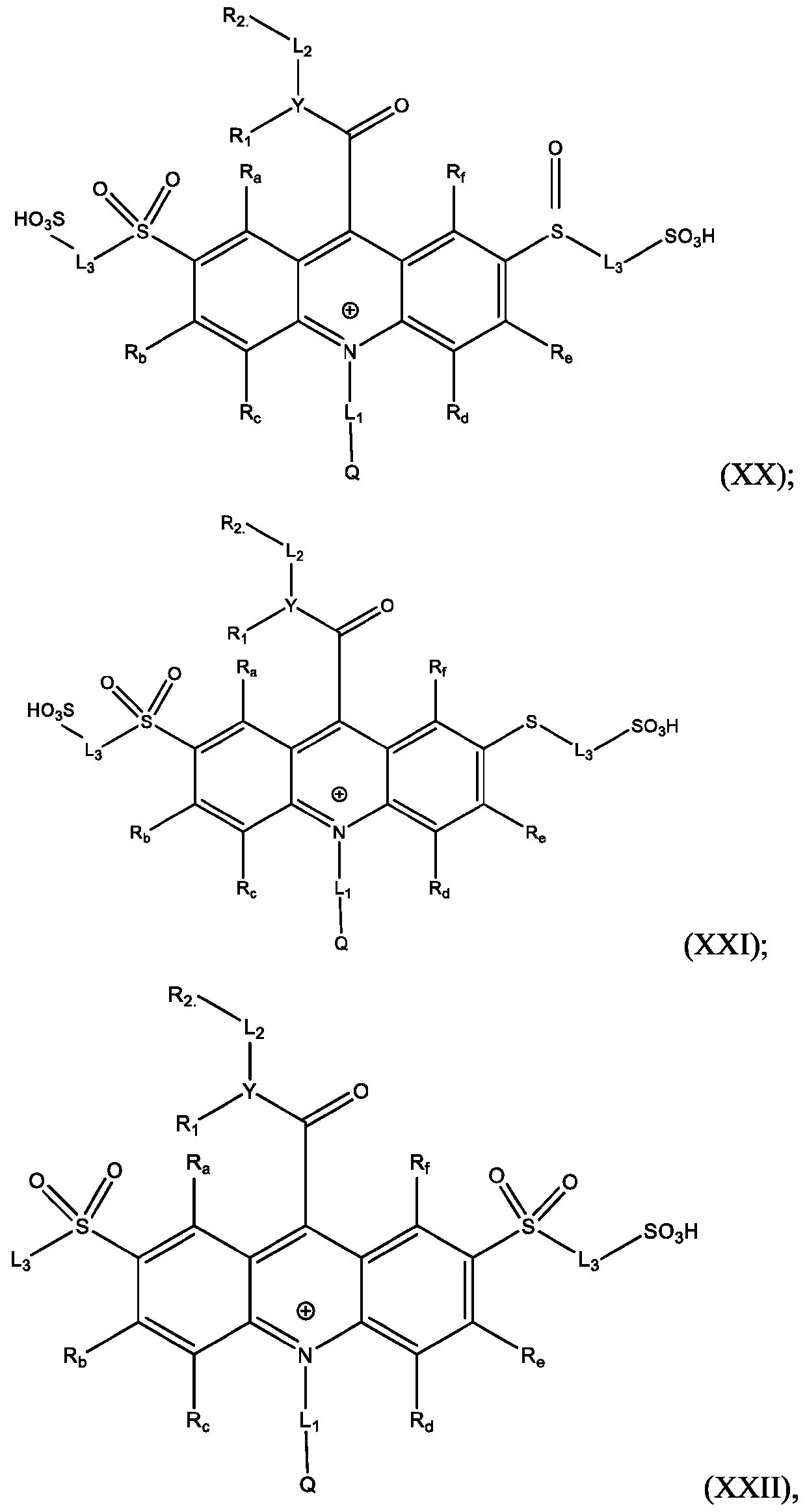
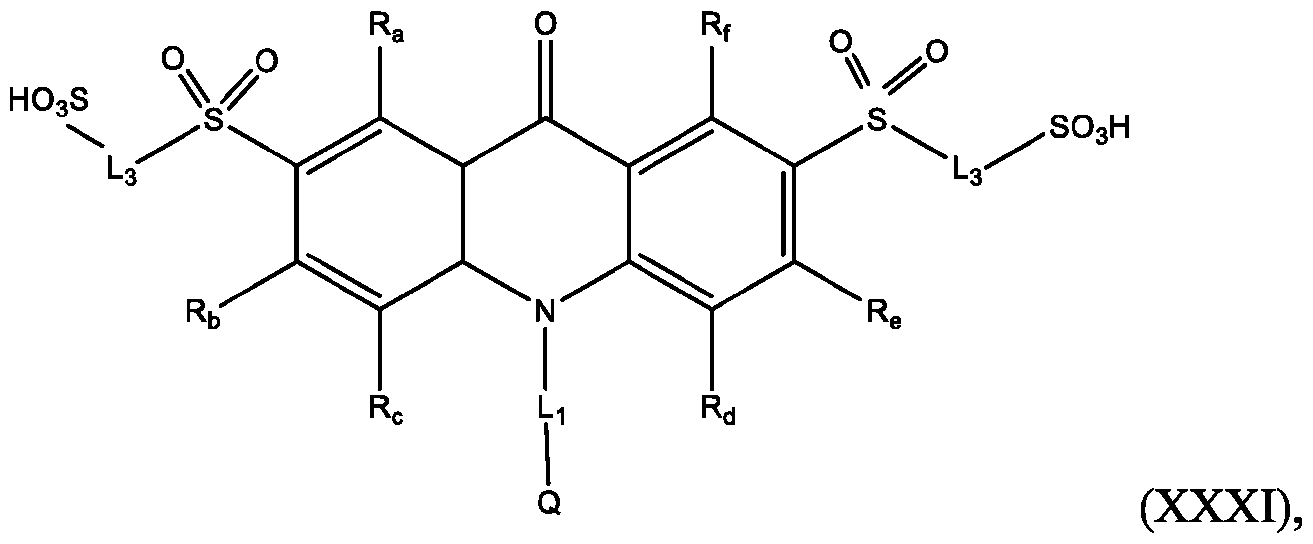
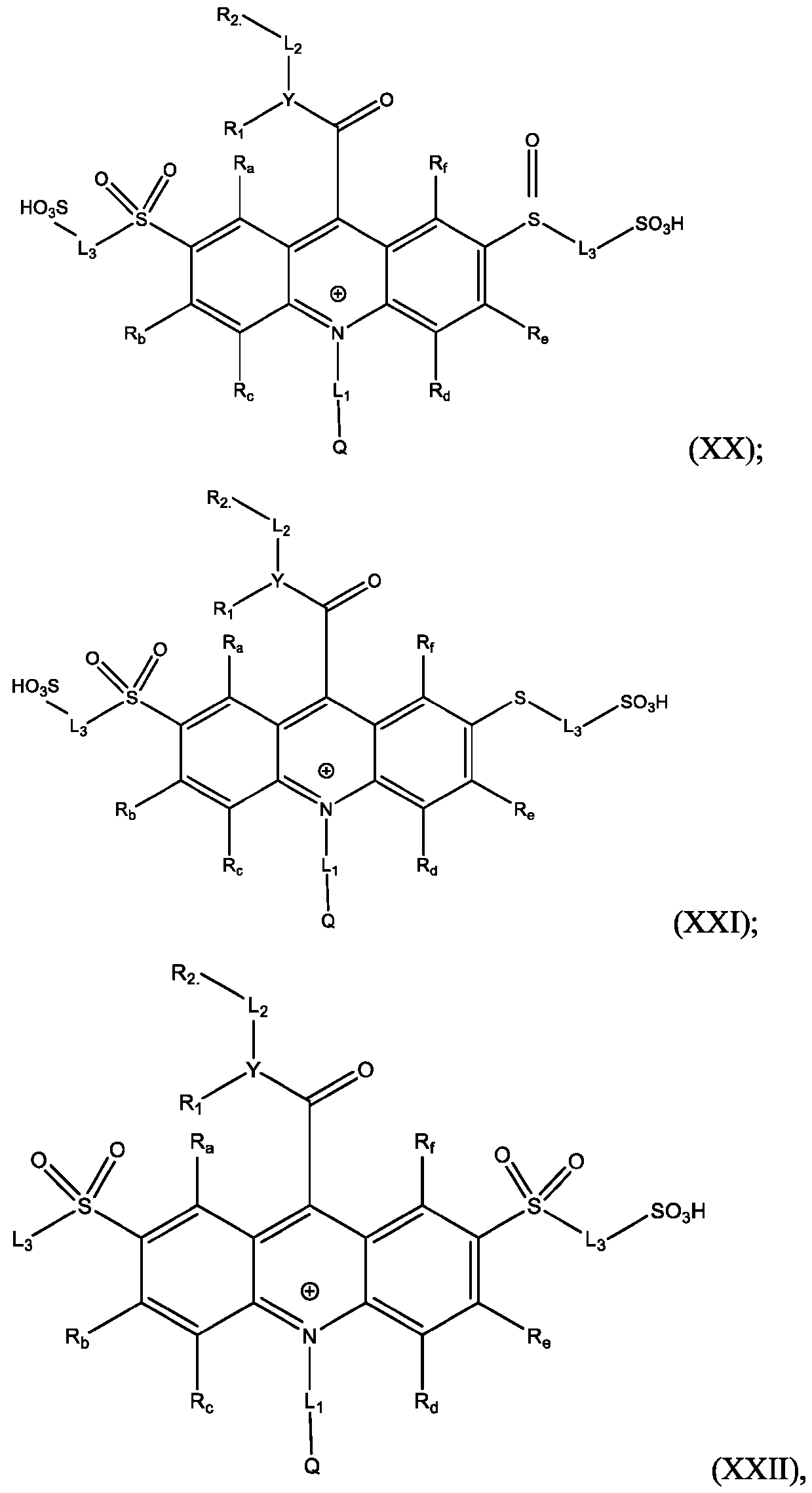
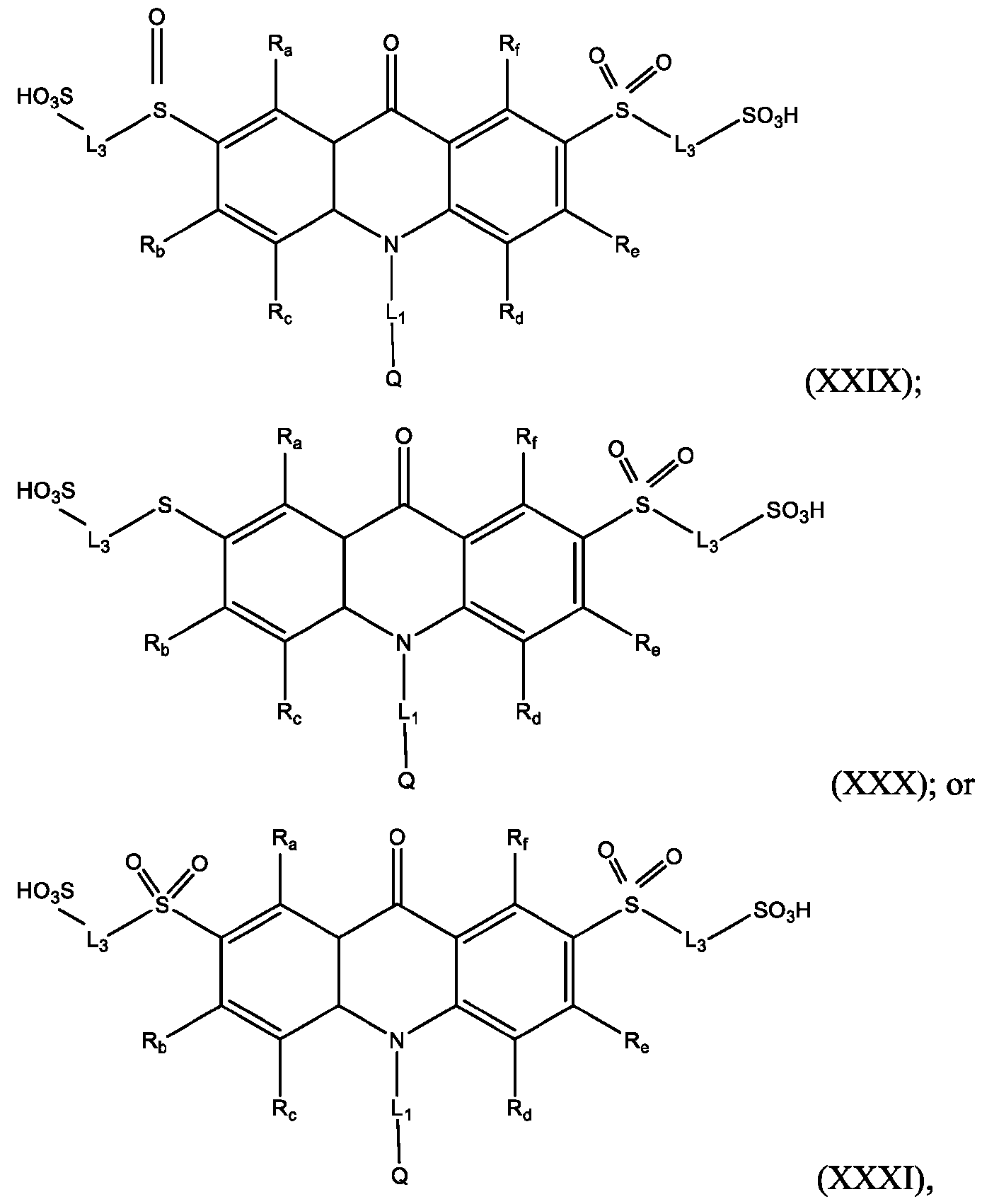
- Also provided herein are methods of producing a BisSulfone compound of Formula (XXII) comprises: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; quenching an oxidation reaction with dimethyl sulfide and adding an amount of water; and isolating and optionally purifying the BisSulfone compound of Formula (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- the at least one organic solvent comprises tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
- the oxidizing agent comprises mCPBA.
- the compound of Formula (XVII) is CPSP3.
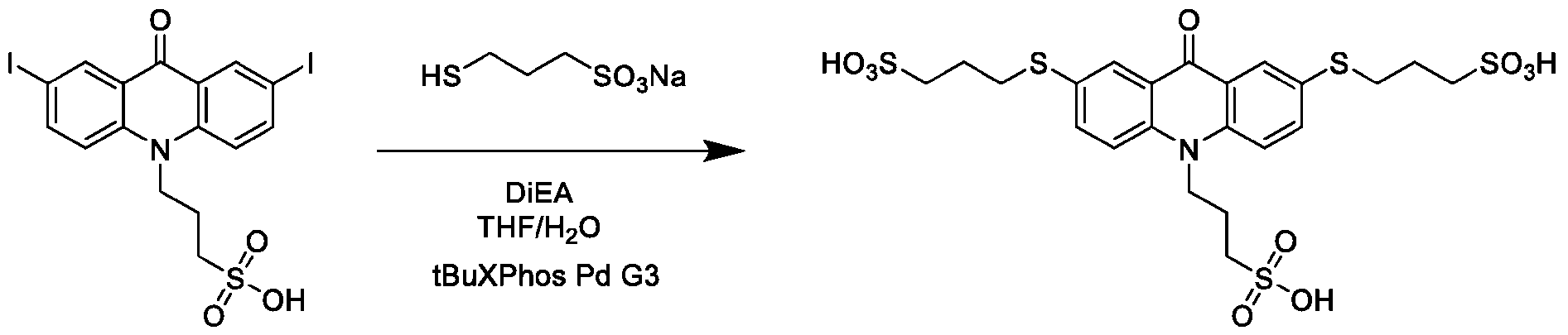
- the method further comprises: reacting the product comprising the acridinium functionalized at the 2 -position with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (XVI).
- the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
- reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
- reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
- DIEA N,N-diisopropylethylamine
- THF tetrahydrofuran
- the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 ,l'-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (rBuXPhos Pd G3).
- the method further comprises isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
- the method further comprises reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium disulfide compound of Formula (XVII).
- reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
- reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
- DIEA N,N-diisopropylethylamine
- THF tetrahydrofuran
- the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
- the method further comprises: isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
- the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
- MSA mercaptopropanesulfonic acid
- the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
- reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
- DIEA N,N-diisopropylethylamine
- THF tetrahydrofuran
- the Pd-containing catalyst comprises 2-Di-terributylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
- R’ is -SO2-L3-SO3H and R” is -S-L3-SO3H.
- R’, R”, or both R’ and R’ are a halogen, preferably I.
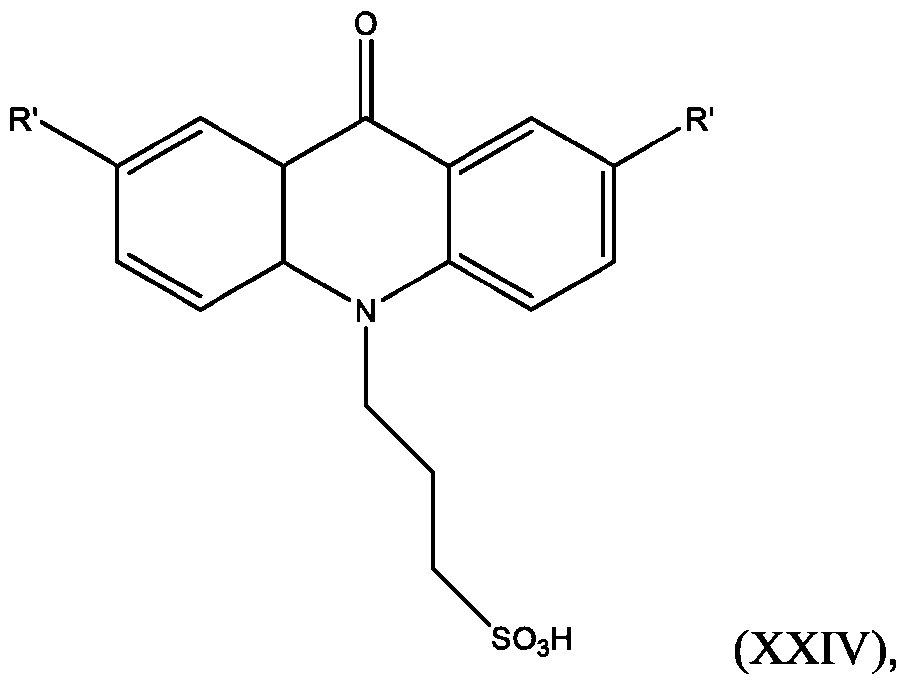
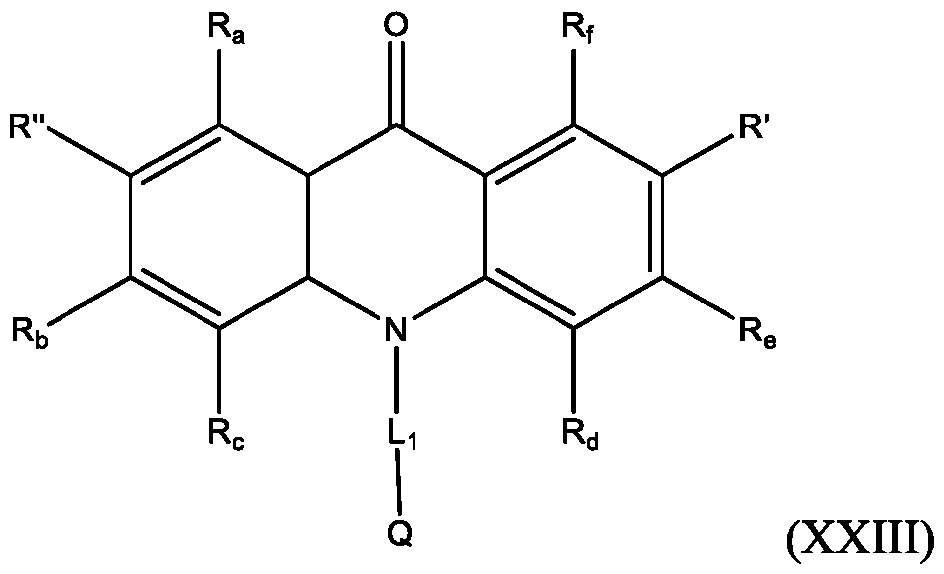
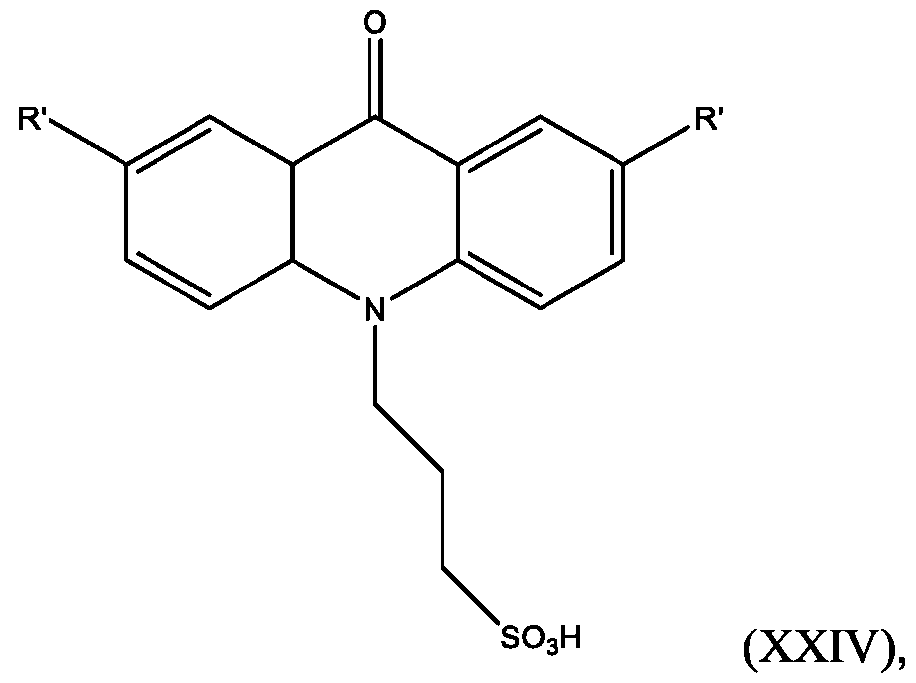
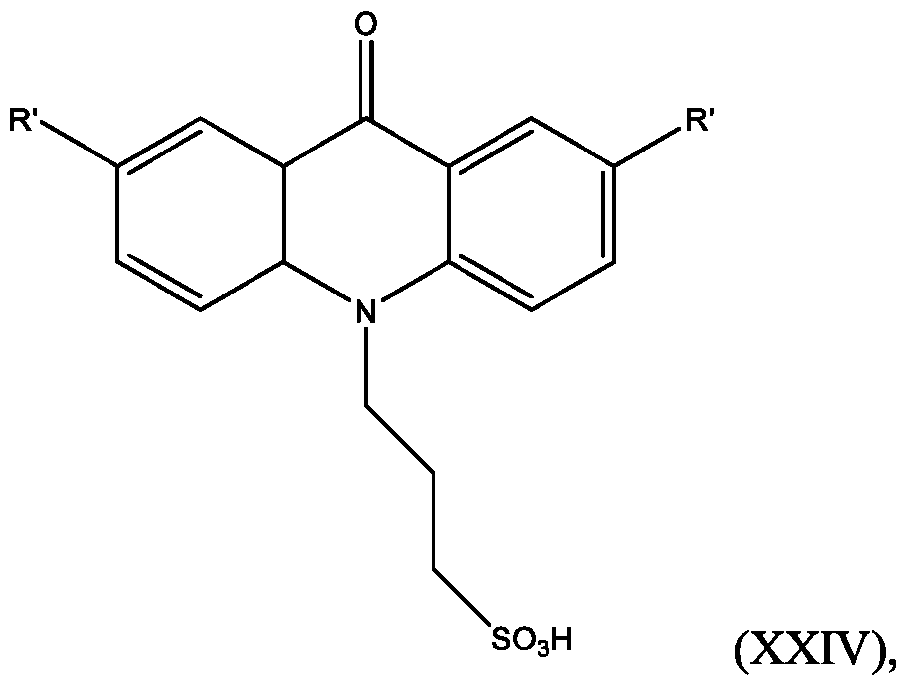
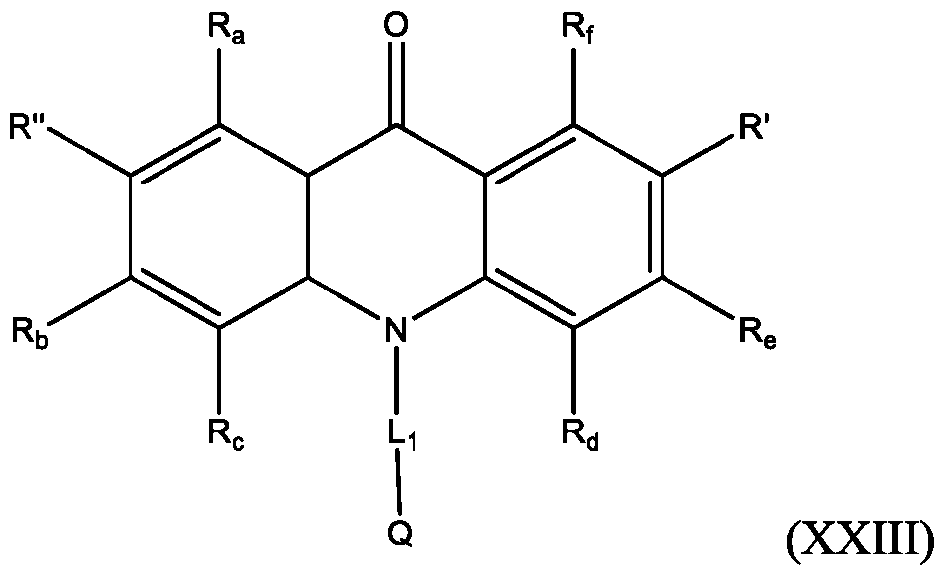
- the compound has the Formula (XXIV): wherein R’ and R” are defined above for Formula (XXIII).
- the acridone compound is any one of the following compounds:
- the compound is selected from:
- R is defined above for Formula (XXIII). In some embodiments, R” is H.
- reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
- DIEA N,N-diisopropylethylamine
- THF tetrahydrofuran
- the oxidizing agent comprises mCPBA.
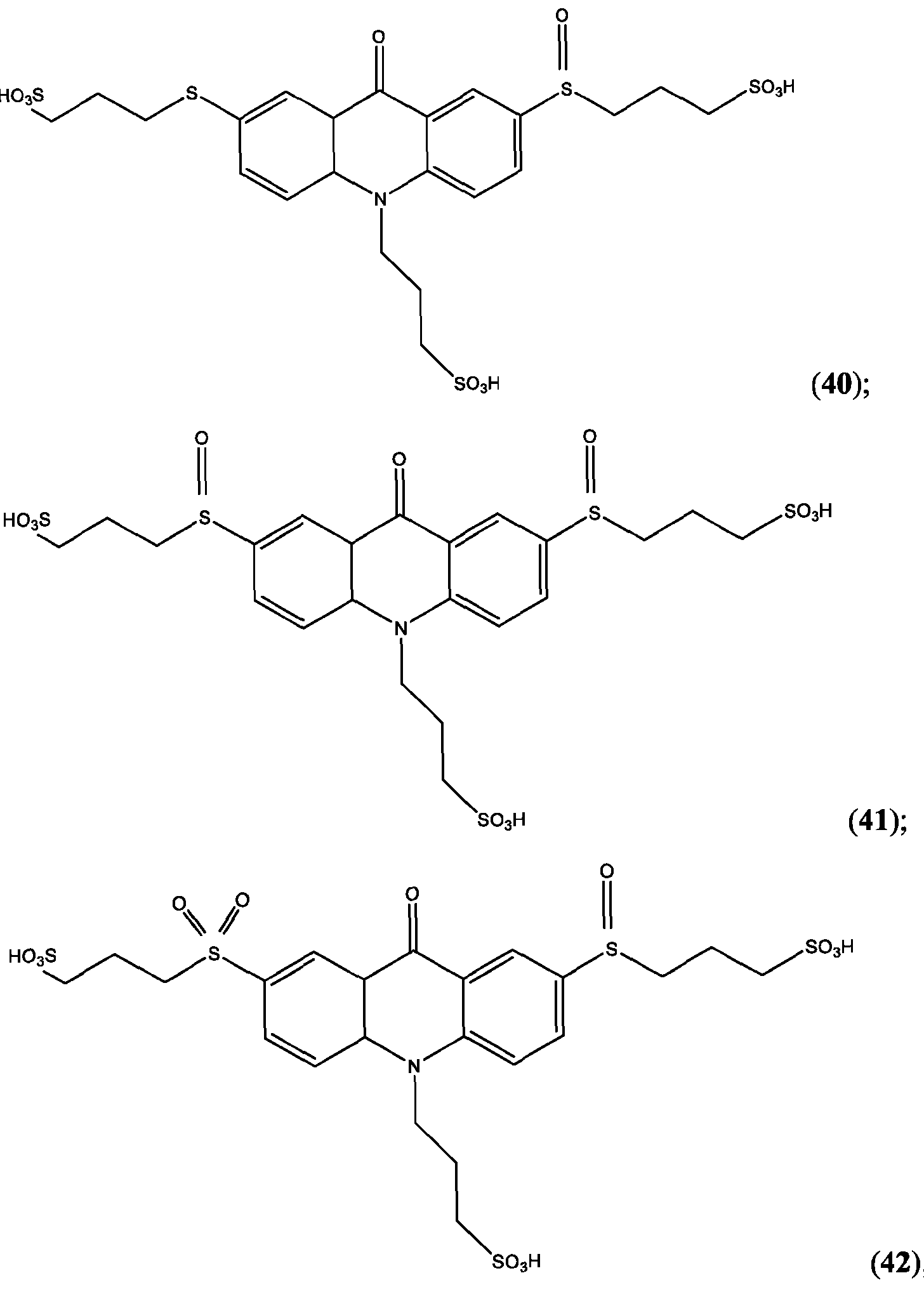
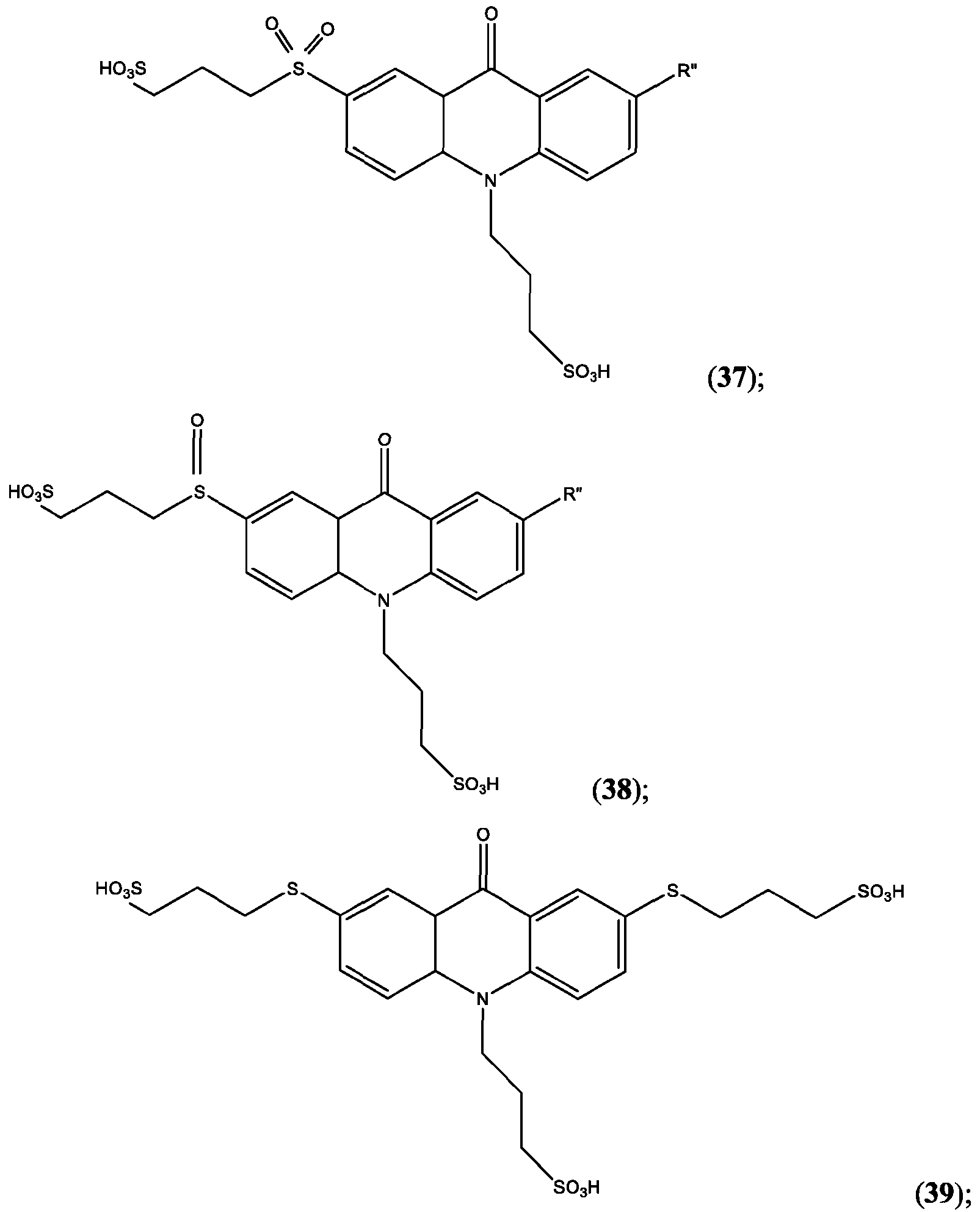
- the compound of Formula (XXVI) is compound (39).
- Also provided herein are methods of producing a sulfoxide of Formula (XXVII) comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; dropwise adding an eighth solution comprising an oxidizing agent to the contents of the container; stirring the contents of the container; quenching the reaction with dimethyl sulfide; and isolating the sulfoxide of Formula (XXVII) and optionally purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, or dimethylformamide (DMF).
- THF tetrahydrofuran
- acetonitrile acetonitrile
- dioxane dimethylsulfoxide
- DMF dimethylformamide
- the eighth solution further comprises at least one organic solvent.
- the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
- the method comprises the steps of: a) performing an assay to detect the presence or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject diagnosed with chronic HBV or receiving a treatment for chronic HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds
- Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R 1 is -SO2-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyh heteroarylalkyh cycloalkylalkyl, and heterocycly lalkyl ; when Y is oxygen or sulfur, R 1 is absent;
- Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
- R 2 is selected from -SO3Z and -COOZ
- Z is selected from hydrogen, alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
- R a , R b , R c , R d , R e , and R f are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
- the method comprises the steps of: a) performing an assay to detect the presence of or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject suspected of having HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg- second detection antibody complex is formed; and b) detecting HBV infection based on the
- Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R 1 is -SO2-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, or heterocycly lalkyl ; when Y is oxygen or sulfur, R 1 is absent;
- Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
- R 2 is selected from -SO3Z and -COOZ;
- Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
- R a , R b , R c , R d , R e , and R f are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
- analyte refers to a substance, material or chemical constituent the presence, absence and/or amount of which is being analyzed in a sample (e.g., a capillary blood sample, a venous blood sample, a plasma sample, a serum sample) obtained from a subject.
- a sample e.g., a capillary blood sample, a venous blood sample, a plasma sample, a serum sample
- the analyte is a biomolecule.
- biomolecules include macromolecules such as, proteins, lipids, and carbohydrates.
- the analyte may be hormones, antibodies, growth factors, cytokines, enzymes, receptors (e.g., neural, hormonal, nutrient, and cell surface receptors) or their ligands, cancer markers (e.g., PSA, TNF-alpha), markers of myocardial infarction (e.g., troponin, creatine kinase, and the like), toxins, drugs (e.g., drugs of addiction), metabolic agents (e.g., including vitamins), and the like.
- cancer markers e.g., PSA, TNF-alpha
- markers of myocardial infarction e.g., troponin, creatine kinase, and the like
- toxins e.g., drugs of addiction
- metabolic agents e.g., including vitamins
- protein analytes include peptides, polypeptides, protein fragments, protein complexes, fusion proteins, recombinant proteins, phosphoproteins, glycoproteins, lipo
- the analyte may be a post-translationally modified protein (e.g., phosphorylated, methylated, glycosylated protein) and the first or the second binding member may be an antibody specific to a post-translational modification.
- a modified protein may be bound to a first binding member immobilized on a solid support where the first binding member binds to the modified protein but not the unmodified protein.
- the first binding member may bind to both the unmodified and the modified protein, and the second binding member may be specific to the post-translationally modified protein.
- monocyclic heterocycles include, but are not limited to, azetidinyl, azepanyl, aziridinyl, diazepanyl, 1,3-dioxanyl, 1,3-dioxolanyl, 1,3-dithiolanyl, 1,3- dithianyl, imidazolinyl, imidazolidinyl, isothiazolinyl, isothiazolidinyl, isoxazolinyl, isoxazolidinyl, morpholinyl, oxadiazolinyl, oxadiazolidinyl, oxazolinyl, oxazolidinyl, oxetanyl, piperazinyl, piperidinyl, pyranyl, pyrazolinyl, pyrazolidinyl, pyrrolinyl, pyrrolidinyl, tetrahydrofuranyl, tetrahydropyr
- the bicyclic heterocycle is a monocyclic heterocycle fused to a phenyl group, or a monocyclic heterocycle fused to a monocyclic cycloalkyl, or a monocyclic heterocycle fused to a monocyclic cycloalkenyl, or a monocyclic heterocycle fused to a monocyclic heterocycle, or a spiro heterocycle group, or a bridged monocyclic heterocycle ring system in which two non-adjacent atoms of the ring are linked by an alkylene bridge of 1, 2, 3, or 4 carbon atoms, or an alkenylene bridge of two, three, or four carbon atoms.
- bicyclic heterocycles include, but are not limited to, benzopyranyl, benzothiopyranyl, chromanyl, 2,3- dihydrobenzofuranyl, 2,3 -dihydrobenzothienyl, 2,3-dihydroisoquinoline, 2-azaspiro[3.3]heptan- 2-yl, azabicyclo[2.2.1]heptyl (including 2-azabicyclo[2.2.1]hept-2-yl), 2,3 -dihydro- lH-indolyl, isoindolinyl, octahydrocyclopenta[c]pyrrolyl, octahydropyrrolopyridinyl, and tetrahydroisoquinolinyl.
- Tricyclic heterocycles are exemplified by a bicyclic heterocycle fused to a phenyl group, or a bicyclic heterocycle fused to a monocyclic cycloalkyl, or a bicyclic heterocycle fused to a monocyclic cycloalkenyl, or a bicyclic heterocycle fused to a monocyclic heterocycle, or a bicyclic heterocycle in which two non-adjacent atoms of the bicyclic ring are linked by an alkylene bridge of 1, 2, 3, or 4 carbon atoms, or an alkenylene bridge of two, three, or four carbon atoms.
- tricyclic heterocycles include, but are not limited to, octahydro-2, 5-epoxypentalene, hexahydro-2H-2,5-methanocyclopenta[b]furan, hexahydro-lH- l,4-methanocyclopenta[c]fiiran, aza-adamantane (l-azatricyclo[3.3.1.13,7]decane), and oxaadamantane (2-oxatricyclo[3.3.1.13,7]decane).
- the monocyclic, bicyclic, and tricyclic heterocycles are connected to the parent molecular moiety through any carbon atom or any nitrogen atom contained within the rings.
- heterocycloalkylene means a divalent heterocycloalkyl group.
- heterocyclylalkyl refers to a heterocyclyl group, as defined herein, appended to the parent molecular moiety through an alkylene group, as defined herein.
- Representative examples of heterocyclylalkyl include, but are not limited to, piperidin-4- ylmethyl, piperazin- 1-ylmethyl, 3 -methyl- 1-pyrrolidin-l-ylbutyl, ( lR)-3 -methyl- 1-pyrrolidin-l- ylbutyl, (1 S)-3 -methyl- 1-pyrrolidin-l-ylbutyl, and 3 -morpholinopropyl.
- hydroxy means an — OH group.
- hydroxyalkyl refers to an alkyl group, as defined herein, substituted with at least one hydroxy group.
- Representative examples of hydroxyalkyl include, but are not limited to, hydroxymethyl, 2-hydroxyethyl, 3 -hydroxypropyl, 2,3-dihydroxypropyl, 2,3-dihydroxypentyl, 4-hydroxybutyl, 2-ethyl-4-hydroxyheptyl, 3,4-dihydroxybutyl, and 5- hydroxypentyl.
- the percentage may be calculated by optimally aligning the two sequences, comparing the two sequences over the specified region, determining the number of positions at which the identical residue occurs in both sequences to yield the number of matched positions, dividing the number of matched positions by the total number of positions in the specified region, and multiplying the result by 100 to yield the percentage of sequence identity.
- the residues of single sequence are included in the denominator but not the numerator of the calculation.
- label refers to any atom or molecule that can be used to provide a detectable and/or quantifiable signal.
- the label can be attached, directly or indirectly, to an antigen or antibody.
- Suitable labels that can be attached to an antigen or antibody include, but are not limited to, radioisotopes, fluorophores, chromophores, mass labels, electron dense particles, magnetic particles, spin labels, molecules that emit chemiluminescence, electrochemically active molecules, enzymes, cofactors, and enzyme substrates.
- Non-point-of-care device refers to a device that is not a point-of-care device or a single use device.
- a “point-of-care” device refers to a device used to provide medical diagnostic testing at or near the point-of-care (namely, outside of a laboratory), at the time and place of patient care (such as in a hospital, physician’s office, urgent or other medical care facility, a patient’s home, a nursing home and/or a long-term care and/or hospice facility).
- a point-of-care instrument does not perform an assay on more than one clinical sample simultaneously.
- point-of-care devices examples include those produced by Abbott Laboratories (Abbott Park, IL) (e.g., i-STAT and i-STAT Alinity, Universal Biosensors (Rowville, Australia) (see U.S. Patent Publication No. 2006/0134713), Axis-Shield PoC AS (Oslo, Norway) and Clinical Lab Products (Los Angeles, USA).
- the point-of-care device is a single-use device.
- the term “single-use device” or “single-use instrument” refers to a clinical diagnostic instrument that processes and performs a clinical diagnostic assay on a unit use basis (such as a single-use cartridge) for a single patient sample.
- a non-point-of-care device refers to any device that does not meet any of the above limitations of a point-of-care or a single use device.
- the non-point-of-care device may be a relatively large instrument, such as a tabletop instrument. Accordingly, in some embodiments the non-point-of-care device is not a handheld instrument.
- the non-point-of-care device is capable of performing an assay on more than one clinical sample simultaneously. Suitable non-point-of- care devices include, for example, the ARCHITECT® or Alinity platforms produced by Abbott Laboratories.
- nucleic acid refers to a nucleotide polymer, and unless otherwise limited, includes known analogs of natural nucleotides that can function in a similar manner (e.g., hybridize) to naturally occurring nucleotides.
- nucleic acid includes any form of DNA or RNA, including, for example, genomic DNA; complementary DNA (cDNA), which is a DNA representation of mRNA, usually obtained by reverse transcription of messenger RNA (mRNA) or by amplification; DNA molecules produced synthetically or by amplification; and mRNA.
- genomic DNA genomic DNA
- cDNA complementary DNA
- mRNA messenger RNA
- mRNA messenger RNA
- mRNA messenger RNA
- mRNA messenger RNA
- mRNA messenger RNA
- mRNA messenger RNA
- mRNA messenger RNA
- nucleic acid encompasses double- or triple-stranded nucleic acids, as well as single-stranded molecules.
- nucleic acid strands need not be coextensive (i.e., a double-stranded nucleic acid need not be double-stranded along the entire length of both strands).
- nucleic acid also encompasses any chemical modification thereof, such as by methylation and/or by capping.
- Nucleic acid modifications can include addition of chemical groups that incorporate additional charge, polarizability, hydrogen bonding, electrostatic interaction, and functionality to the individual nucleic acid bases or to the nucleic acid as a whole. Such modifications may include base modifications such as 2'-position sugar modifications, 5-position pyrimidine modifications, 8-position purine modifications, modifications at cytosine exocyclic amines, substitutions of 5 -bromo-uracil, backbone modifications, unusual base pairing combinations such as the isobases isocytidine and isoguanidine, and the like.
- “Positive predictive value” or “PPV” as used interchangeably herein refers to the probability that a subject has a positive outcome given that they have a positive test result.
- “Quality control reagents” in the context of immunoassays (e.g., competitive immunoassay and sandwich immunoassay) and kits described herein include, but are not limited to, calibrators, controls, and sensitivity panels.
- a “calibrator” or “standard” typically is used (e.g., one or more, such as a plurality) in order to establish calibration (standard) curves for interpolation of the concentration of an analyte, such as an antibody or an analyte.
- a single calibrator which is near a reference level or control level (e.g., “low”, “medium”, or “high” levels), can be used.
- Multiple calibrators i.e., more than one calibrator or a varying amount of calibrator(s) can be used in conjunction to comprise a “sensitivity panel.”
- reaction vessel refers to a holder or receiver, such as a container, receptacle, tube, and/or cartridge, in or upon which one or more assays is performed.
- a “reagent” refers broadly to any agent used in a reaction, other than the analyte of interest.
- Illustrative reagents for immunoassay include, for example, antigens or antibodies specific for an analyte of interest, detection (e.g., labeled) antigens or antibodies, controls, diluents, buffers, and the like.
- single molecule detection refers to the detection and/or measurement of a single molecule of an analyte in a test sample at very low levels of concentration (such as pg/mL or femtogram/mL levels).
- concentration such as pg/mL or femtogram/mL levels.
- single molecule analyzers or devices include nanopore and nanowell devices. Examples of nanopore devices are described in International Patent Publication No. WO 2016/161402, which is hereby incorporated by reference in its entirety. Examples of nanowell device are described in International Patent Publication No. WO 2016/161400, which is hereby incorporated by reference in its entirety.
- substituted refers to a group substituted on an atom of the indicated group.
- test sample generally refers to a material (e.g., a biological material) being tested for and/or suspected of containing an analyte of interest, such as a marker described herein.
- the test sample may be derived from any biological source, such as, a physiological fluid, including, but not limited to, whole blood, serum, plasma, interstitial fluid, saliva, ocular lens fluid, cerebral spinal fluid, sweat, urine, milk, ascites fluid, mucous, nasal fluid, sputum, synovial fluid, peritoneal fluid, vaginal fluid, menses, amniotic fluid, semen and so forth.
- the sample is a whole blood sample.
- the sample is a plasma sample. In yet other embodiments, the sample is a serum sample.
- the test sample may be used directly as obtained from the biological source or following a pretreatment to modify the character of the sample. For example, such pretreatment may include preparing plasma from blood, diluting viscous fluids and so forth. Methods of pretreatment may also involve filtration, precipitation, dilution, distillation, mixing, concentration, inactivation of interfering components, the addition of reagents, lysing, etc. Moreover, it may also be beneficial to modify a solid test sample to form a liquid medium or to release the analyte.
- Solid phase refers to any material that can be used to attach and/or attract and immobilize (1) one or more capture reagents or capture specific binding partners, or (2) one or more detection reagents or detection specific binding partners.
- the solid phase can be chosen for its intrinsic ability to attract and immobilize a capture reagent.
- the solid phase can have affixed thereto a linking agent that has the ability to attract and immobilize the (1) capture reagent or capture specific binding partner, or (2) detection reagent or detection specific binding partner.
- the linking agent can include a charged substance that is oppositely charged with respect to the capture reagent (e.g., capture specific binding partner) or detection reagent (e.g., detection specific binding partner) itself or to a charged substance conjugated to the (1) capture reagent or capture specific binding partner or (2) detection reagent or detection specific binding partner.
- the linking agent can be any binding partner (preferably specific) that is immobilized on (attached to) the solid phase and that has the ability to immobilize the (1) capture reagent or capture specific binding partner, or (2) detection reagent or detection specific binding partner through a binding reaction.
- the linking agent enables the indirect binding of the capture reagent to a solid phase material before the performance of the assay or during the performance of the assay.
- the solid phase can be plastic, derivatized plastic, magnetic, or non-magnetic metal, glass or silicon, including, for example, a test tube, microtiter well, sheet, bead, microparticle, chip, and other configurations known to those of ordinary skill in the art.
- “Specific binding” or “specifically binding” as used herein may refer to the interaction of an antibody or antibody fragment thereof, a protein, or a peptide (e.g., an antigen) with a second chemical species, wherein the interaction is dependent upon the presence of a particular structure (e.g., an antigenic determinant or epitope) on the chemical species; for example, an antibody recognizes and binds to a specific protein structure rather than to proteins generally. If an antibody is specific for epitope “A”, the presence of a molecule containing epitope A (or free, unlabeled A), in a reaction containing labeled “A” and the antibody, will reduce the amount of labeled A bound to the antibody.
- a particular structure e.g., an antigenic determinant or epitope
- Specific binding partner is a member of a specific binding pair.
- a specific binding pair comprises two different molecules, which specifically bind to each other through chemical or physical means. Therefore, in addition to antigen and antibody specific binding pairs of common immunoassays, other specific binding pairs can include biotin and avidin (or streptavidin), carbohydrates and lectins, complementary nucleotide sequences, effector and receptor molecules, cofactors and enzymes, enzymes and enzyme inhibitors, and the like.
- specific binding pairs can include members that are analogs of the original specific binding members, for example, an analyte-analog.
- Immunoreactive specific binding members include antigens, antigen fragments, and antibodies, including monoclonal and polyclonal antibodies as well as complexes and fragments thereof, whether isolated or recombinantly produced.
- substantially identical may mean that a first and second sequence are at least from about 50% to about 99% identical over a region of from about 8 to about 100 or more residues (including any range within from about 8 to about 100 residues).
- the subject may be a human or a non-human.
- the subject or patient may be undergoing forms of treatment.
- “Mammal” as used herein refers to any member of the class Mammalia, including, without limitation, humans and nonhuman primates such as chimpanzees and other apes and monkey species; farm animals such as cattle, sheep, pigs, goats, llamas, camels, and horses; domestic mammals such as dogs and cats; laboratory animals including rodents such as mice, rats, rabbits, guinea pigs, and the like. The term does not denote a particular age or sex. Thus, adult and newborn subjects, as well as fetuses, whether male or female, are intended to be included within the scope of this term.
- Treatment are each used interchangeably herein to describe reversing, alleviating, or inhibiting the progress of a disease and/or injury, or one or more symptoms of such disease, to which such term applies.
- a treatment may be either performed in an acute or chronic way.
- the term also refers to preventing a disease or injury, and includes preventing the onset of a disease or injury, or preventing the symptoms associated with a disease or injury.
- Preventing also refers to preventing the recurrence of a disease or injury or of one or more symptoms associated with such disease or injury.
- Treatment and “therapeutically,” refer to the act of treating, as “treating” is defined above.
- the prevention or treatment of a disease can be done prior to affliction or injury, such as, for example, to reduce the severity of a disease or injury or symptoms associated with a disease or an injury.
- Such prevention or reduction can include (a) administration of one or more pharmaceutical composition and/or one or more nutritional compositions to a subject; (b) the use of one or more of physical therapy, occupational therapy, and/or counseling; or (c) any combinations of (a) and (b).
- Variant is used herein to describe a peptide or polypeptide that differs in amino acid sequence by the insertion, deletion, or conservative substitution of amino acids, but retain at least one biological activity.
- SNP refers to a variant that is a single nucleotide polymorphism.
- biological activity include the ability to be bound by a specific antibody or to promote an immune response.
- Variant is also used herein to describe a protein with an amino acid sequence that is substantially identical to a referenced protein with an amino acid sequence that retains at least one biological activity.
- a conservative substitution of an amino acid i.e., replacing an amino acid with a different amino acid of similar properties (e.g., hydrophilicity, degree, and distribution of charged regions) is recognized in the art as typically involving a minor change. These minor changes can be identified, in part, by considering the hydropathic index of amino acids, as understood in the art. Kyte et al., J. Mol. Biol. 157:105-132 (1982). The hydropathic index of an amino acid is based on a consideration of its hydrophobicity and charge. It is known in the art that amino acids of similar hydropathic indexes can be substituted and still retain protein function. In one aspect, amino acids having hydropathic indexes of ⁇ 2 are substituted.
- hydrophilicity of amino acids can also be used to reveal substitutions that would result in proteins retaining biological junction.
- a consideration of the hydrophilicity of amino acids in the context of a peptide permits calculation of the greatest local average hydrophilicity of that peptide, a useful measure that has been reported to correlate well with antigenicity and immunogenicity.
- U.S. Patent No. 4,554,101 incorporated fully herein by reference.
- Substitution of amino acids having similar hydrophilicity values can result in peptides retaining biological activity, for example immunogenicity, as is understood in the art. Substitutions may be performed with amino acids having hydrophilicity values within ⁇ 2 of each other.
- hydrophobicity index and the hydrophilicity value of amino acids are influenced by the particular side chain of that amino acid. Consistent with that observation, amino acid substitutions that are compatible with biological function are understood to depend on the relative similarity of the amino acids, including the side chains of those amino acids, as revealed by the hydrophobicity, hydrophilicity, charge, size, and other properties.
- Vector is used herein to describe a nucleic acid molecule that can transport another nucleic acid to which it has been linked.
- plasmid refers to a circular double-stranded DNA loop into which additional DNA segments may be ligated.
- viral vector Another type of vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome.
- Certain vectors can replicate autonomously in a host cell into which they are introduced (e.g., bacterial vectors having a bacterial origin of replication and episomal mammalian vectors).
- a biological sample suspected of containing an analyte of interest is combined with one or more reagents comprising a capture reagent and a detection reagent to form a reaction mixture.
- the biological sample and capture reagent are subject to a washing step to remove any unbound analyte prior to the addition of the detection reagent to the reaction mixture.
- the biological sample containing the analyte of interest, the capture reagent, and the detection reagent are all combined and subjected to a washing step to remove any unbound analyte.
- the detectable label used in the detection reagent in the sandwich immunoassay is a different than the detectable label used in the detection reagent used in the competitive immunoassay.
- the competitive immunoassay and the sandwich immunoassay are performed in the same reaction vessel. In other aspects, the competitive immunoassay and the sandwich immunoassay are performed in different reaction vessels. Additionally, in still yet other aspects, when generating the calibration curve, the capture reagent used in the competitive immunoassay and the sandwich immunoassay are identical. In still yet even further aspects, when generating the calibration curve, the detectable label used in detection reagent in the competitive immunoassay and the detectable label used in the detection reagent in the sandwich immunoassay are not identical.
- a single (e.g., the same) biological sample obtained from the subject can be used to perform each of the competitive immunoassay and the sandwich immunoassay.
- multiple biological samples can be obtained from the subject and used to perform each of the competitive immunoassay and the sandwich immunoassay.
- the biological samples used in the methods of the present disclosure do not require dilution prior to performing the competitive immunoassay, prior to performing the sandwich immunoassay, or prior to perform both the competitive immunoassay and prior to performing the sandwich immunoassay.
- the biological sample requires no dilution prior to performing any of the assays described herein. No pre-dilution of the biological sample avoids the problems known in the art with under and over diluting biological samples prior to performing a competitive immunoassay or sandwich immunoassay.
- the competitive immunoassay and the sandwich immunoassay are performed in a single (e.g., the same) reaction vessel using two at least two different detectable labels (e.g., the competitive immunoassay and the sandwich immunoassay each use different detectable labels).
- the competitive immunoassay and the sandwich immunoassay are performed in two different (e.g., separate) reaction vessels using at least two different detectable labels (e.g., the competitive immunoassay and the sandwich immunoassay each use different detectable labels).
- the competitive immunoassay signal detectable signal is detected by a first detection component in an instrument and the sandwich immunoassay detectable signal is detected by a second detection component in an instrument.
- the first detection component can be a first photomultiplier tube with a first specific filter (e.g., a blue filter) in an instrument
- the second detection component can be a second photomultiplier tube with a second specific filter (e.g., a green filter) in an instrument.
- the competitive immunoassay and the sandwich immunoassay are performed in two different (e.g., separate) reaction vessels (a first reaction vessel and a second reaction vessel) but employ the same detectable label (e.g., the competitive immunoassay and the sandwich immunoassay each use the same detectable label).
- the competitive immunoassay and the sandwich immunoassay are performed in two different (e.g., separate) reaction vessels, the immunoassays can be performed simultaneously, or sequentially, in any order.
- the competitive immunoassay signal detectable signal and the sandwich immunoassay detectable signal can be detected by a single detection component in an instrument.
- the detection component can be a single photomultiplier tube with a specific filter (e.g., a blue filter) in an instrument that can be used to detect the chemiluminescent signal from each of the competitive immunoassay and sandwich immunoassay in their respective reaction vessels.
- a method of detecting an analyte of interest in a biological sample comprising the steps of: a) contacting a biological sample with at least one specific binding member that binds to the analyte of interest to form at least one complex, wherein the specific binding member comprises the compound of Formula (I), as described herein; and b) detecting the presence or absence of a signal from the specific binding member, wherein detection of the signal indicates that the analyte is present in the sample and the absence of the signal indicates that the analyte is not present in the sample.
- the compound of Formula (I) does not have a fluorophore conjugated to it.
- the competitive immunoassay detectable signal and the sandwich immunoassay detectable signal are communicated on or from at least one instrument.
- Suitable instruments are described herein, including non-point-of-care devices that may contain a user interface that communicate by displaying the determination.
- Suitable instruments are described herein, including non-point-of-care devices (e.g. the ARCHITECT® and Alinity platforms marketed by Abbott Laboratories) that may contain a user interface that communicate by displaying the determination.
- suitable instruments include point-of-care devices (such as the iSTAT® Abbott Point of Care devices).
- the instrument contains software to execute one or more tasks. In some embodiments, the instrument contains software to automatically determine the next appropriate step in a method as described herein. For example, the instrument may contain software that determines the amount or level of an analyte of interest in a biological sample. The software may display this determination, such as on a graphical user interface.
- the instrument stores software that instructs a processor to execute a given task.
- the software stores machine readable instructions that instruct a processor to execute a given task.
- the machine readable instructions may be one or more executable programs or portion(s) of an executable program for execution by a computer.
- the programs may be embodied in software stored on a non-transitory computer readable storage medium such as a CD-ROM, a floppy disk, a hard drive, a DVD, a Blu-ray disk, or a memory associated with the processors.
- the entire programs and/or parts thereof could alternatively be executed by a device other than the processors and/or embodied in firmware or dedicated hardware.
- processes may be implemented by one or more hardware circuits (e.g., discrete and/or integrated analog and/or digital circuitry, an FPGA, an ASIC, a comparator, an operational-amplifier (op-amp), a logic circuit, etc.) structured to perform the corresponding operation without executing software or firmware.
- hardware circuits e.g., discrete and/or integrated analog and/or digital circuitry, an FPGA, an ASIC, a comparator, an operational-amplifier (op-amp), a logic circuit, etc.
- the machine readable instructions may be stored in one or more of a compressed format, an encrypted format, a fragmented format, a compiled format, an executable format, a packaged format, etc.
- Machine readable instructions as described herein may be stored as data (e.g., portions of instructions, code, representations of code, etc.) that may be utilized to create, manufacture, and/or produce machine executable instructions.
- the machine readable instructions may be fragmented and stored on one or more storage devices and/or computing devices (e.g., servers).
- the machine readable instructions may require one or more of installation, modification, adaptation, updating, combining, supplementing, configuring, decryption, decompression, unpacking, distribution, reassignment, compilation, etc.
- the machine readable instructions may be stored in multiple parts, which are individually compressed, encrypted, and stored on separate computing devices, wherein the parts when decrypted, decompressed, and combined form a set of executable instructions that implement a program such as that described herein.
- the machine readable instructions may be stored in a state in which they may be read by a computer, but require addition of a library (e.g., a dynamic link library (DLL)), a software development kit (SDK), an application programming interface (API), etc. in order to execute the instructions on a particular computing device or other device.
- a library e.g., a dynamic link library (DLL)
- SDK software development kit
- API application programming interface
- the machine readable instructions may need to be configured (e.g., settings stored, data input, network addresses recorded, etc.) before the machine readable instructions and/or the corresponding program(s) can be executed in whole or in part.
- the disclosed machine readable instructions and/or corresponding program(s) are intended to encompass such machine readable instructions and/or program(s) regardless of the particular format or state of the machine readable instructions and/or program(s) when stored or otherwise at rest or in transit.
- the machine readable instructions described herein can be represented by any past, present, or future instruction language, scripting language, programming language, etc.
- the machine readable instructions may be represented using any of the following languages: C, C++, Java, C#, Perl, Python, JavaScript, HyperText Markup Language (HTML), Structured Query Language (SQL), Swift, etc.
- the machine readable instructions may be stored on a non-transitory computer and/or machine readable medium such as a hard disk drive, a flash memory, a read-only memory, a compact disk, a digital versatile disk, a cache, a random-access memory and/or any other storage device or storage disk in which information is stored for any duration (e.g., for extended time periods, permanently, for brief instances, for temporarily buffering, and/or for caching of the information).
- a non-transitory computer readable medium is expressly defined to include any type of computer readable storage device and/or storage disk and to exclude propagating signals and to exclude transmission media.
- the disclosed methods and systems for determining the concentration or level of analyte of interest present in a sample may be as described herein.
- the methods may also be adapted in view of other methods for analyzing analytes such as single molecule detection described below.
- the methods, systems, and kits as described herein may also involve single molecule counting.
- the device can be a microfluidics device, digital microfluidics device, a surface acoustic wave based microfluidic device (SAW), an integrated digital microfluidics device and analyte detection device, an integrated SAW and analyte detection device, or robotics based assay processing unit.
- SAW surface acoustic wave based microfluidic device
- SMCTM single molecule counting
- Nanopore devices are described in International Patent Publication No. WO 2016/161402, which is hereby incorporated by reference in its entirety.
- nanowell device are described in International Patent Publication No. WO 2016/161400, which is hereby incorporated by reference in its entirety.
- methods for analyte analysis can involve mass spectrometry using DIAMS, DDA-MS or SRM/MRM-MS or PRM-MS.
- methods for analyte analysis can involve assessing a sample for the presence of an analyte.
- assessing a sample for the presence of an analyte can be used for determining presence of and/or concentration of an analyte or a fragment in a sample.
- a method can also be used for determining presence of and/or concentration of a plurality of different analytes or analyte fragments present in a sample. Quantification can be performed using internal control proteins or peptide fragments.
- the methods, systems, and kits optionally are adapted for use on an automated or semi-automated system.
- Some of the differences between an automated or semi-automated system as compared to a non-automated system include the substrate to which the capture antibody is attached (which can impact sandwich formation and analyte reactivity), and the length and timing of the capture, detection and/or any optional wash steps.
- a non-automated format may include a relatively longer incubation time with sample and capture reagent (e.g., about 2 hours) an automated or semi-automated format (e.g., ARCHITECT® or Alinity) may have a relatively shorter incubation time (e.g., approximately 18 minutes for ARCHITECT®).
- an automated or semi-automated format e.g., ARCHITECT® or Alinity
- may have a relatively shorter incubation time e.g., approximately 4 minutes for the ARCHITECT®).
- the power of a diagnostic test to correctly predict status is commonly measured as the sensitivity of the assay, the specificity of the assay or the area under a receiver operated characteristic (“ROC”) curve.
- Sensitivity is the percentage of true positives that are predicted by a test to be positive, while specificity is the percentage of true negatives that are predicted by a test to be negative.
- a ROC curve provides the sensitivity of a test as a function of 1 -specificity. The greater the area under the ROC curve, the more powerful the predictive value of the test.
- Other useful measures of the utility of a test are positive predictive value and negative predictive value. Positive predictive value is the percentage of people who test positive that are actually positive. Negative predictive value is the percentage of people who test negative that are actually negative.
- the values measured for an analyte of interest can be mathematically combined with other values measured for other analytes and the combined value is correlated to the underlying diagnostic question.
- Biomarker values may be combined by any appropriate state of the art mathematical method.
- the method used in a correlating a biomarker combination is selected from DA (e.g., Linear-, Quadratic-, Regularized Discriminant Analysis), DFA, Kernel Methods (e.g., SVM), MDS, Nonparametric Methods (e.g., k-Nearest-Neighbor Classifiers), PLS (Partial Least Squares), Tree-Based Methods (e.g., Logic Regression, CART, Random Forest Methods, Boosting Methods), or Generalized Linear Models (e.g., Logistic Regression), and Principal Components Analysis. Details relating to these statistical methods are found in the following references: Ruczinski et al., 12
- Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R 1 is -SO2-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, and heterocyciylalkyl; when Y is oxygen or sulfur, R 1 is absent;
- Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
- Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyciylalkyl, aryloxy, or heteroalkyl;
- Y is selected from nitrogen, oxygen, and sulfur. In some embodiments, Y is nitrogen. In some embodiments, when Y is nitrogen, R 1 is -SO 2 -A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, and heterocyclylalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Y is nitrogen and R 1 is -SO 2 -A, and A is a substituted or unsubstituted aryl group. In some embodiments, Y is nitrogen and R 1 is -SO 2 -A, and A is a aryl group with a methyl substitute. In some embodiments, when Y is oxygen or sulfur, R 1 is absent;
- Q is -SO3Z, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- Q is -SO3H.
- Q is -SO3Z wherein Z is a Ci -Ci6 group.
- Li and L 2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- Li and L2 are each independently selected from C1-C10 alkylene.
- Li is — CH2CH2CH2 — .
- L2 is — CH2CH2CH2— .
- R 2 is selected from -SO3Z and -COOZ, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R 2 is -SO3H.
- R 2 is -SO3Z, wherein Z is a Ci -Ci6 group.
- R 2 is -COOZ, wherein Z is H.
- R 2 is -COOZ, wherein Z is is a Ci -Ci6 group.
- R 2 is - COOZ, wherein Z is N-hydroxysuccinimide (NHS).
- R a , R b , R c , R d , R e , and R f are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R a is hydrogen.
- R b is hydrogen.
- R c is hydrogen.
- R d is hydrogen.
- R e is hydrogen.
- R f is hydrogen.
- R’ and/or R are H. In some embodiments, R’ and/or R” are Cl. In some embodiments, R’ and/or R” are Br. In some embodiments, R’ and/or R” are I. In some embodiments, R’ and/or R” are NO 2 . In some embodiments, R’ and/or R” are NH 2 . In some embodiments, R’ and/or R” are t-butyl. In some embodiments, R’ and/or R” are SO3H. In some embodiments, R’ and/or R” are substituted aryl groups. In some embodiments, R’ and/or R” are unsubstituted aryl groups.
- R’ and/or R are substituted heteroaryl groups. In some embodiments, R’ and/or R” are unsubstituted heteroaryl groups. In some embodiments, R’ and/or R” are substituted phenyl groups. In some embodiments, R’ and/or R” are unsubstituted phenyl groups. In some embodiments, R’ and/or R” are substituted pyrrolyl groups. In some embodiments, R’ and/or R” are unsubstituted pyrrolyl groups. In some embodiments, R’ and/or R” are substituted furanyl groups. In some embodiments, R’ and/or R” are unsubstituted furanyl groups.
- R’ and/or R are substituted thienyl groups. In some embodiments, R’ and/or R” are unsubstituted thienyl groups. In some embodiments, R’ and/or R” are substituted thienothiophenyl groups. In some embodiments, R’ and/or R” are unsubstituted thienothiophenyl groups.
- the zinc comprises elemental zinc, a zinc alloy, or a combination of both.
- the first solution further comprises a water-miscible alcohol, preferably ethanol.
- the acridinium ring is reduced at temperature in a range from about 50°C to about 90°C, preferably about 60°C to about 80°C.
- the acridan intermediate is isolated prior to reacting with the electrophile.
- the compound of Formula (Va) is separated from the compound of Formula (Vb).
- the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
- a method of producing the compound of Formula (Via), (VIb), or both (Via) and (VIb) comprises: dissolving the acridan intermediate of Formula (III) in a third solution in a container; adding iodine monochloride to the container and stirring for about 30 min to about 1 h; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb).
- the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
- the compound of Formula (Via) is separated from the compound of Formula (VIb).
- the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
- the electrophile is NO2
- the reaction of the acridan intermediate with NO2 to form a product comprising the acridinium functionalized at the 2,7- position of the acridinium ring includes a nitration reaction
- the products are represented by Formulas (Vila) and (Vllb): wherein Y, Ri, R2, Li, L2, Q, R a , R b , R c , R d , R e , and R f are defined above for Formula (I).
- a method of producing the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb) comprises: dissolving the acridan intermediate in a fourth solution in a container; adding chlorosulfonic acid to the container and stirring for about 5 min to about 30 min; adding water to the container and stirring for about 10 min to about 30 min; and isolating and optionally purifying the product comprising the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
- the fourth solution comprises acetonitrile.
- the electrophile is t-butyl
- the reaction of the acridan intermediate with t-butyl to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring includes a Friedel-Craft alkylation reaction, and the products are represented by Formulas (IXa) and (IXb):
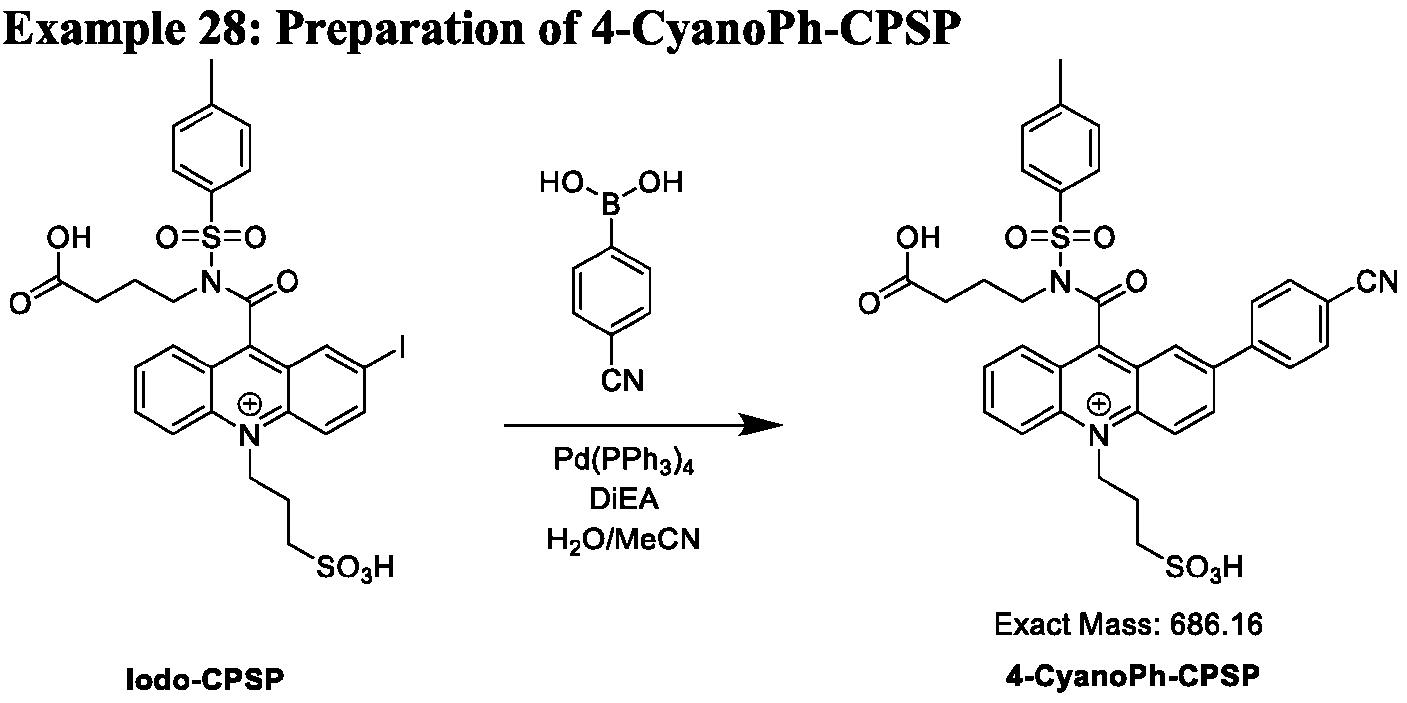
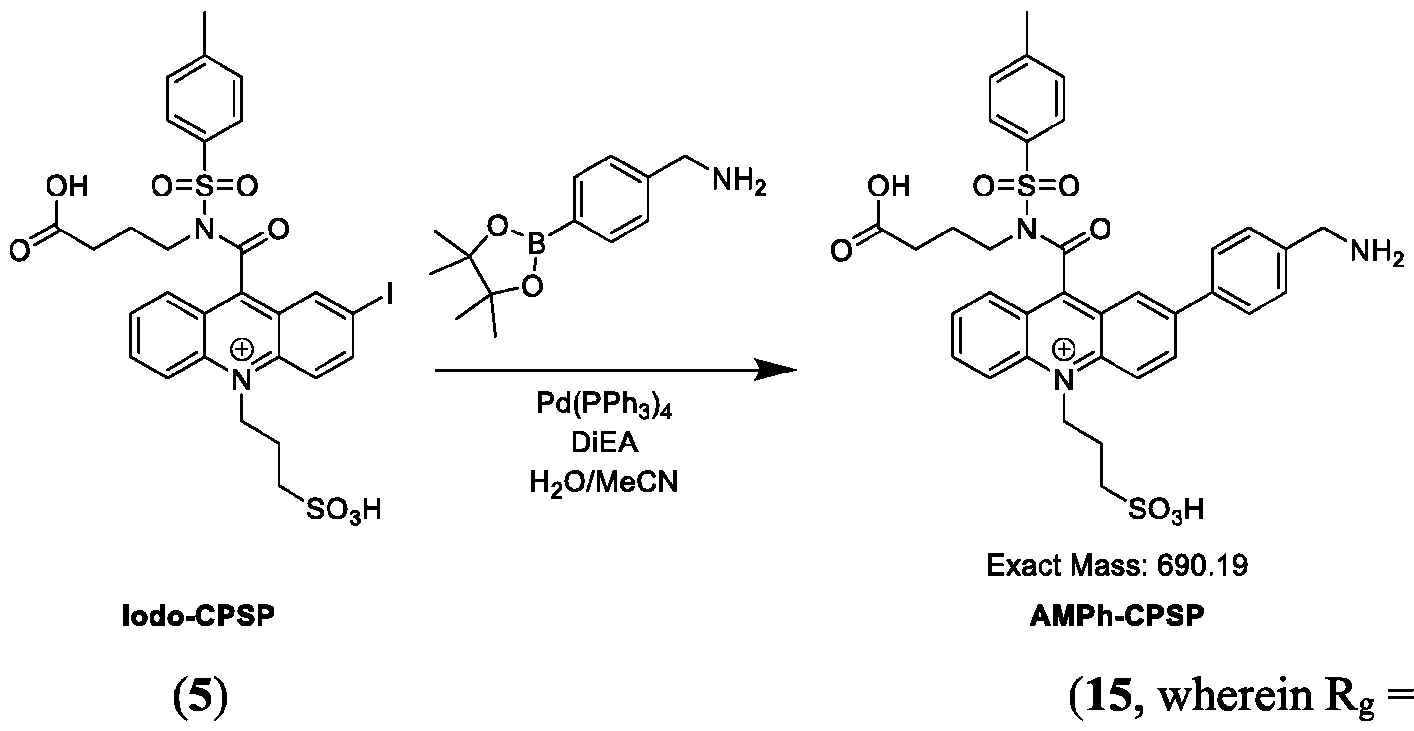
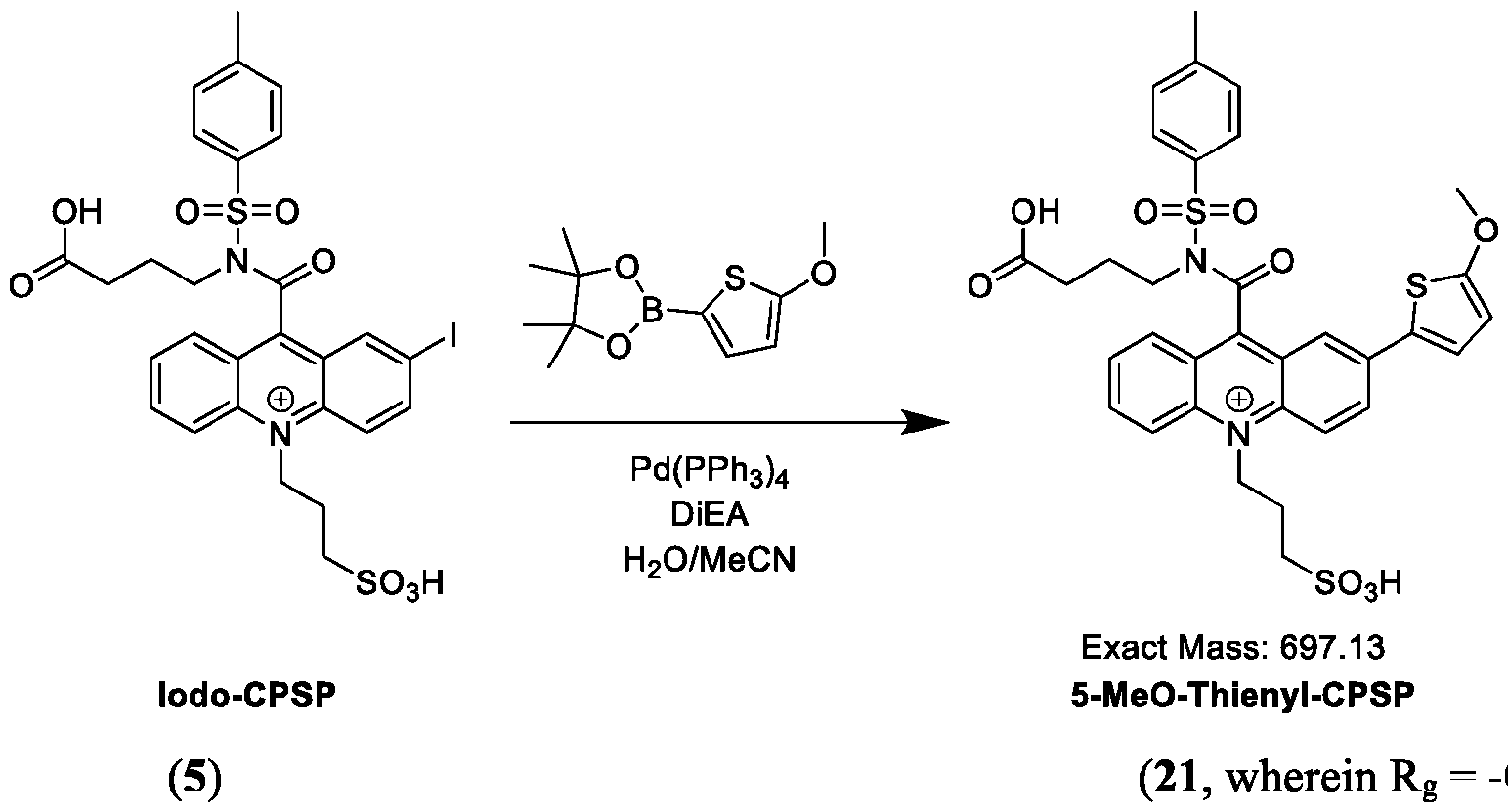
- a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring comprises: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a boronic acid comprising A’ or a boronate ester comprising A’, wherein A’ is a substituted or unsubstituted aryl group or a substituted or unsubstituted heteroaryl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
- a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring with a substituted or unsubstituted pyrrolyl group comprises: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a tert-butyloxycarbonyl (Boc)-protected boronic acid comprising A’ or a Boc-protected boronate ester comprising A’, wherein A’ is a substituted or unsubstituted pyrrolyl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating the product comprising the acridinium-containing compound comprising the Boc-protect
- isolated, functionalized acridinium-containing compounds e.g., any of those of Formulas (IV)-(IX)
- the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA).
- the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water.
- DIEA N,N-diisopropylethylamine
- THF tetrahydrofuran
- DMF dimethylformamide
- the Pd-containing catalyst includes, but is not limited to, 2-Di-tert-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'- amino- 1,1 '-biphenyl)] palladium (II) methanesulfonate (tBuXPhos Pd G3).
- the method further comprises isolating and optionally purifying the acridinium monosulfide product.
- the monosulfide moiety of Formula (XVI) is oxidized to a sulfone using, e.g., meta-Chloroperoxybenzoic acid (mCPBA).
- the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
- the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA).
- the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water.
- the Pd-containing catalyst includes, but is not limited to, 2-Di- terf-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1 , 1 '-biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3).
- the method further comprises isolating and optionally purifying the acridinium monosulfide product.
- a method of producing an acridinium disulfide of Formula (XVII) comprises: reacting a product of any one of Formulas (IVb)-(IXb) with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium disulfide product of Formula (XVII): wherein Y, Ri, R2, Li, L2, L3, Q, R a , R b , R c , R d , R e , and R f are defined above for Formula (I).
- Q is -SO3Z or H
- Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
- R’ and/or R are H. In some embodiments, R’ and/or R” are Cl. In some embodiments, R’ and/or R” are Br. In some embodiments, R’ and/or R” are I. In some embodiments, R’ and/or R” are -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R’ is -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R’ is -S-L3-SO3H, wherein L3 is a C1-C10 alkylene.
- R’ is -S-L3-SO3H, wherein L3 is — CH2CH2CH2 — .
- R” is - S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R" is -S-L3-SO3H, wherein L3 is a C1-C10 alkylene.
- R” is - S-L3-SO3H, wherein L3 is — CH2CH2CH2 — .
- R’ and/or R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R’ is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R’ is -SO2-L3-SO3H, wherein L3 is a C1-C10 alkylene.
- R’ is -SO2-L3-SO3H, wherein L3 is — CH2CH2CH2 — .
- R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- R" is -SO2-L3-SO3H, wherein L3 is a C1-C10 alkylene.
- R” is -SO2-L3-SO3H, wherein L3 is — CH2CH2CH2 — .
- the acridone compound is a compound of Formula (XXIV): wherein R’ and R” are defined above for Formula (XXIII).
- the acridone compound is any one of the following compounds:
- the acridone compound is a compound of any one of Formulas:
- the compound of Formula (XXIII) is functionalized at the 2- or 2,7 positions and is selected from:
- R is defined above for Formula (XXIII). In some embodiments, R” is H.
- groups and substituents thereof may be selected in accordance with permitted valence of the atoms and the substituents, such that the selections and substitutions result in a stable compound, e.g., which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc.
- Stereoisomers include enantiomers and diastereomers and mixtures of enantiomers or diastereomers. Individual stereoisomers of the compounds may be prepared synthetically from commercially available starting materials, which contain asymmetric or chiral centers or by preparation of racemic mixtures followed by methods of resolution well-known to those of ordinary skill in the art.
- the present disclosure also includes isotopically-labeled compounds, which are identical to those recited in Formula (XXIII), but for the fact that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature.
- isotopes suitable for inclusion in the compounds of the invention are hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, and chlorine, such as, but not limited to, 2 H, 3 H, l3 C, U C, t5 N, 1S O, ;7 0, 3 i P, 32 P, 33 S, 1S F, and 36 C1, respectively.
- the compound may incorporate positronemitting isotopes for medical imaging and positron-emitting tomography (PET) studies for determining the distribution of receptors.
- positron-emitting isotopes that can be incorporated in compounds of Formula (I) are 13 N, ’ : 5 O, and ' 8 F.
- Isotopically-labeled compounds of Formula (XXIII) can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying examples using appropriate isotopically-labeled reagent in place of non-isotopicaily-labeied reagent.
- a compound disclosed herein may be in the form of a salt.
- the salts may be prepared during the final isolation and purification of the compounds or separately, for example byreacting a basic group of the compound (e.g., an amino group) with a suitable acid or by reacting an acidic group of the compound (e.g., a carboxylic acid group) with a suitable base.
- Acid salts may be prepared during the final isolation and purification of the compounds or separately by reacting a suitable group of the compound, such as an amino group, with a suitable acid.
- a compound may be dissolved in a suitable solvent, such as but not limited to methanol and water, and treated with at least one equivalent of an acid, such hydrochloric acid.
- the resulting salt may precipitate cut and be isolated by filtration and dried under reduced pressure. Alternatively, the solvent and excess acid may be removed under reduced pressure to provide a salt.
- Representative salts include acetate, adipate, alginate, citrate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, camphorate, eamphorsulfonate, digluconate, glycerophosphate, hemisuliate, heptanoate, hexanoate, formate, isethionate, fumarate, lactate, maleate, methanesulfonate, naphthylenesulfonate, nicotinate, oxalate, pamoate, pectinate, persulfate, 3 -phenylpropionate, picrate, oxalate, maleate, pivalate, propionate, succinate, tartrate, trichloroacetate, trifluoroacetate
- the amino groups of the compounds may also be quatemized with alkyd chlorides, bromides and iodides such as methyl, ethyl, propyl, isopropyl, butyl, lauryl, myristyl, stearyl and the like.
- Basic addition salts may be prepared during the final isolation and purification of the disclosed compounds by reaction of a carboxyl group with a suitable base such as the hydroxide, carbonate, or bicarbonate of a metal cation such as lithium, sodium, potassium, calcium, magnesium, or aluminum, or an organic primary, secondary, or tertiary amine.
- Quaternary amine salts can be prepared, such as those derived from methylamine, dimethylamine, trimethylamine, triethylamine, diethylamine, ethylamine, tributylamine, pyridine, N,N-dimethylaniline, N-methylpiperidine, N-methylmorpholine, dicyclohexylamine, procaine, dibenzylamine, N,N-dibenzylphenethylamine, 1-ephenamine and N,N'- dibenzylethylenediamine, ethylenediamine, ethanolamine, diethanolamine, piperidine, piperazine, and the like.
- the present disclosure relates to methods of synthesizing the compounds of Formula (XXIII).
- the compounds of Formula (XXIII) are readily prepared by oxidizing an acridinium-containing compound of Formula (I) in an alkaline solution.
- an acridinium-containing compound of any of Formulas (XVI)-(XXII) or compounds (24)-(32) can be oxidized to produce an acridone of Formulas (XXV)-(XXXII) or compounds (36)-(44), respectively.
- the oxidizing agent comprises H2O2.
- a method of functionalizing an acridone-containing compound at a 2,7-position of an acridone ring with iodine comprises: dissolving an acridone-containing compound in a third solution; adding iodine monochloride to the third solution and stirring for about 6 h to about 18 h under pressure to produce the acridone-containing compound functionalized at the 2,7-positions in a mixture; dissolving the mixture in DMSO; and isolating and optionally purifying the diiodo-acridone compound.
- the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
- a method of producing an acridone disulfide of Formula (XXVI) comprises: reacting a dihalo-acridone, e.g., a diiodo-acridone of compound (35), with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridone disulfide product of Formula (XXVI): wherein Li, L3, Q, R a , R b , R c , R d , R e , and R f are defined above for Formula (XXIII).
- a compound of Formula (XXVI) can be oxidized to produce a sulfoxide of Formula (XXVII), a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- a method of producing a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI) comprises: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating the bis sulfoxide of Formula (XXVIII), sulfoxide sulfone of Formula (XXIX), and/or bis sulfone of Formula (XXXI) and optionally separating and purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, or heterocycloalky
- the at least one organic solvent includes, but is not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
- the oxidizing agent comprises mCPBA.
- the compound of Formula (XXVI) is compound (39).
- the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
- a method of producing a sulfoxide of Formula (XXVII) comprises: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; dropwise adding an eighth solution comprising an oxidizing agent to the contents of the container; stirring the contents of the container; quenching the reaction with dimethyl sulfide; and isolating the sulfoxide of Formula (XXVII) and optionally purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
- the at least one organic solvent includes, but is not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
- the oxidizing agent comprises mCPBA.
- the eighth solution further comprises at least one organic solvent, e.g., DMF.
- the compound of Formula (XXVI) is compound (39).
- the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
- the dimethyl sulfide is added dropwise. In some embodiments, immediately after the addition of dimethyl sulfide, a bolus of water is added.
- compounds of Formula (I) or Formula (XXIII) can be used in assays, such as in, for example, duplex assays, including those described herein in Section 2.
- the compounds of Formula (I) or Formula (XXIII) can be used in methods for detecting the presence of or determining the concentration or amount one or more analytes of interest in a biological sample obtained from a subject. Detecting the presence of or determining the concentration or amount of an analyte of interest in a biological sample can be used for detecting, identifying and/or monitoring a disease, condition, or illness or monitoring the response of a particular disease, condition, or illness to treatment with a therapeutic (e.g., small or large molecule).
- a therapeutic e.g., small or large molecule
- the compounds of Formula (I) or Formula (XXIII) can be used to assess the stage and/or monitor the stage or phase of chronic Hepatitis B (HBV) infection or monitoring response to a treatment for chronic HBV.
- a method can involve the steps of: a) performing an assay to detect the presence or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject diagnosed with chronic HBV or receiving a treatment for chronic HBV, where the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody
- the compounds of Formula (I) or Formula (XXIII) can be used to detect the presence of or the level of HBV in a sample obtained from a subject.
- a method can involve the steps of: a) performing an assay to detect the presence of or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject suspect of having HBV, where the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg- first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitop
- the first detectable label and second detectable label can each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII): wherein:
- Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R 1 is -SOa-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyL heteroarylalkyl, cycloalkylalkyl, and heterocycly lalkyi ; when Y is oxygen or sulfur, R 1 is absent; Q is -SO3Z or H;
- Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
- R 2 is selected from -SO3Z and -COOZ
- Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
- the fluid sample may be diluted prior to use in an assay.
- the source containing an analyte of interest is a human body fluid (e.g., blood, serum)
- the fluid may be diluted with an appropriate solvent (e.g., a buffer such as PBS buffer).
- an appropriate solvent e.g., a buffer such as PBS buffer.
- a fluid sample may be diluted about 1-fold, about 2-fold, about 3 -fold, about 4-fold, about 5 -fold, about 6-fold, about 10-fold, about 100-fold, or greater, prior to use.
- the fluid sample is not diluted prior to use in an assay.
- the sample may undergo pre-analytical processing.
- Pre-analytical processing may offer additional functionality such as nonspecific protein removal and/or effective yet cheaply implementable mixing functionality.
- General methods of pre-analytical processing may include the use of electrokinetic trapping, AC electrokinetics, surface acoustic waves, isotachophoresis, dielectrophoresis, electrophoresis, or other pre-concentration techniques known in the art.
- the fluid sample may be concentrated prior to use in an assay.
- the fluid may be concentrated by precipitation, evaporation, filtration, centrifugation, or a combination thereof.
- a fluid sample may be concentrated about 1-fold, about 2-fold, about 3-fold, about 4-fold, about 5-fold, about 6-fold, about 10-fold, about 100-fold, or greater, prior to use.
- control may be analyzed concurrently with the sample from the subject as described above.
- the results obtained from the subject sample can be compared to the results obtained from the control sample.
- Standard curves may be provided, with which assay results for the sample may be compared.
- Such standard curves present levels of marker as a function of assay units, i.e., fluorescent signal intensity, if a fluorescent label is used.
- standard curves can be provided for reference levels of an analyte of interest in normal healthy tissue, as well as for “at- risk” levels of the analyte of interest in tissue taken from donors, who may have one or more of the characteristics set forth above.
- a method for determining the presence, amount, or concentration of an analyte of interest in a test sample comprises assaying the test sample for an analyte of interest by an immunoassay, for example, employing at least one capture antibody that binds to an epitope on an analyte of interest and at least one detection antibody that binds to an epitope on an analyte of interest which is different from the epitope for the capture antibody and optionally includes a detectable label, and comprising comparing a signal generated by the detectable label as a direct or indirect indication of the presence, amount or concentration of an analyte of interest in the test sample to a signal generated as a direct or indirect indication of the presence, amount or concentration of an analyte of interest in a calibrator.
- the calibrator is optionally, and in some embodiments, is part of a series of calibrators in which each of the calibrators differs from the other calibrators in the series
- kits and systems for detecting the presence, level, or status of an analyte of interest in a sample.
- the kits or systems find use in multiplex and/or automated analysis methods.
- Exemplary reagents include, but are not limited to, antigens, antibodies, colorimetric reagents, enzymes, buffers, etc.
- the kit can also contain at least one calibrator or control. Any calibrator or control can be included in the kit.
- kits comprising one or more antibodies or other detection reagents.
- the assays, kits and kit components of the disclosure are optimized for use on commercial platforms (e.g., immunoassays on the PRISM®, AxSYM®, ARCHITECT®, Alinity, and EIA (Bead) platforms of Abbott Laboratories, Abbott Park, IL, as well as other commercial and/or in vitro diagnostic assays).
- kits include quality control reagents (for example, sensitivity panels, calibrators, and positive controls).
- quality control reagents for example, sensitivity panels, calibrators, and positive controls.
- Preparation of quality control reagents is well known in the art, and is described, e.g., on a variety of immunodiagnostic or nucleic acid product insert sheets.
- Example 1 A one step dual channel combo assay run on Dual-Channel Detection System to Extend Assay Dynamic Range
- Microparticles from bHCG ARCHITECT® assay kit (Abbott Laboratories, Abbott Park, IL).
- Competing analyte recombinant bHCG labeled with CPSP (blue color).
- Analyte anti-RBD antibody from CO VID- 19 patient sample or vaccinated patient sample.
- Dual Channel 25 pL of 0.1% RBD coated microparticles + 50pL calibrators or patient sample + 50pL (5 nM) Rabbit anti-RBD polyclonal antibody were incubated for 18 minutes and washed.
- Table C lists the signal response measured in the Competitive and Sandwich (dual) Channels, and the calculated signal ratio for a serial diluted patient sample.
- methods of functionalizing chemiluminescent acridiniums at the 2 and 2,7-position of the acridinium ring begin with a H + /2e reduction of an acridinium ring to an acridan intermediate using a metal, e.g., zinc, and weakly acidic solution (e.g., as shown in Scheme 1).
- Acridans are electron rich and capable of selectively reacting with electrophiles (X + ) at the 2 and 7 positions of the acridinium ring.
- the electrophiles can include, but are not limited to, Cl, Br, I, NO2, SO3H, and t-butyl. Electrophiles are generated from classical electrophilic aromatic substitution reaction conditions.
- reaction mixture was injected on a 100g Cl 8 Aq flash column and eluted using 100% water to 60% acetonitrile (MeCN or ACN) gradient. Fractions containing the CPSP-acridan product were collected and lyophilized to dryness. Yield - 450 mg white powder.
- the aqueous filtrate was discarded, and the resin washed with 25 mL MeCN.
- the reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min.
- a manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% trifluoroacetic acid (TFA) Yield of 2- Monochloro-CPSP was 3.5 mg. Yield of 2,7-Dichloro-CPSP was 8.9 mg.
- the solid resin was transferred to a Buchner funnel and washed with 25 mL of water. The aqueous filtrate was discarded, and the resin washed with 25 mL MeCN.
- the reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min.
- a manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H 2 O/H 2 O-0.5% TFA Yield of Di-t-butyl-CPSP was 6.8 mg. Yield of Mono-t-butyl- CPSP was 3.5 mg.
- Example 10 Summary of the Spectroscopic Properties of Functionalized Acridinium- Containing Compounds
- Abs absorbance
- CL chemiluminescence
- FIG. 11. illustrates the chemiluminescence reaction rates of DiChloro-CPSP and Di-t- butyl-CPSP after adding alkaline peroxide.
- Example 12 Preparation of CPSP2
- a step gradient method (Flow rate 40 mL/Minute; mobile phase of ACN/H2O/IO % H2O- 0.5% TFA) was applied using acetonitrile (Solvent A), water (Solvent B), and (Solvent C) 0.5 % TFA in water (held at a constant 10 % throughout the gradient run). Recorder chart speed 5 mm/min. Fractions containing the product were combined and evaporated to dryness. Yield of CPSP3-BisSulfone, 1.7 mg. The absorbance spectrum and chemiluminescence spectrum of CPSP3-BisSulfone is shown in FIGs. 17A and 17B, respectively. FIRMS (M-H) observed m/z 955.0524, calculated neutral mass 956.0600, found 956.0597 (mass error -0.4 mDa).
- Acridone is the product from the acridinium chemiluminescence reaction. As such, the fluorescence spectrum of acridone should match the spectrum observed from the chemiluminescence reaction.
- the SP3 -Acridone produced from alkaline peroxide-triggered CPSP3 was isolated. The fluorescence spectrum of SP3 -Acridone was found to match the chemiluminescence spectrum of CPSP3.
- An alternative method of producing SP3-Acridone involves synthesis of Dilodo-Acridone.
- a step gradient method was applied using acetonitrile (Solvent A), water (Solvent B), and 0.5% TFA in water (Solvent C). Recorder chart speed 5 mm/min.
- FIGs. 19A and 19B The absorbance spectrum and chemiluminescence spectrum of SP3-Acridone BisSulfoxide is shown in FIGs. 19A and 19B, respectively.
- the absorbance spectrum and fluorescence spectrum of SP3-Acridone Sulfoxide Sulfone is shown in FIGs. 20A and 20B, respectively.
- the absorbance spectrum and fluorescence spectrum of SP3-Acridone BisSulfone is shown in FIGs. 21 A and 2 IB, respectively.
- Ph-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of phenylboronic acid pinacol ester. Yield was 4 mg of Ph-CPSP.
- the absorbance spectrum and chemiluminescence spectrum of Ph-CPSP is shown in FIGs. 24A and
- HPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 4-hydroxyphenylboronic acid pinacol ester. Yield was 2.5 mg of HPh- CPSP.
- the absorbance spectrum and chemiluminescence spectrum of HPh-CPSP is shown in FIGs. 25A and 25B, respectively.
- 4-cyanoPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg Cyanophenylboronic acid. Yield was 3.2 mg of 4-CyanoPh-CPSP.
- the absorbance spectrum and chemiluminescence spectrum of 4-CyanoPh-CPSP is shown in FIGs. 27A and 27B, respectively.
- AMPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 4-(Aminomethyl)phenylboronic acid pinacol ester hydrochloride. Yield was 4.5 mg of AMPh-CPSP.
- Furanyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 2-furanylboronic acid. Yield was 3.0 mg of Furanyl-CPSP.
- the absorbance spectrum and chemiluminescence spectrum of Furanyl-CPSP is shown in FIGs. 29A and 29B, respectively.
- Thienyl-CPSP is shown in FIGs. 30A and 30B, respectively.
- 5-Me-thienyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 5-Methylthiophene-2-boronic acidpinacol ester. Yield was 2.8 mg of 5-Me-Thienyl-CPSP.
- the absorbance spectrum and chemiluminescence spectrum of 5- Me-Thienyl-CPSP is shown in FIGs. 31 A and 3 IB, respectively.
- 5-MeO-Thienyl-CPSP is shown in FIGs. 32A and 32B, respectively.
- MeO-Thienyl-CPSP The absorbance spectrum and chemiluminescence spectrum of 3-MeO-
- Thienyl-CPSP is shown in FIGs. 34A and 34B, respectively.
- Thienothiophenyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of Thienothiophene-2-boronic acid. Yield was 4.5 mg of Thienothiophenyl-CPSP.
- the absorbance spectrum and chemiluminescence spectrum of Thienothiophenyl-CPSP is shown in FIGs. 36A and 36B, respectively.
- Difuranyl-CPSP was prepared from 5 mg of Dilodo-CPSP and 5 mg of 2-Furanylboronic acid. Yield was 3.9 mg of DiFuranyl-CPSP.
- the absorbance spectrum and chemiluminescence spectrum of DiFuranyl-CPSP is shown in FIGs. 38A and 38B, respectively.
- Example 42 Summary of Chemiluminescence of Acridinium Compounds Having Sulfide Substitutions at 2- and 2,7 Positions
- acridinium-containing compounds with at least one sulfide, or with one sulfide and one sulfone, substituted at the 2 or 2,7-position were synthesized.
- the resulting sulfide-containing acridinium compounds are highly water-soluble species.
- further spectral shifts were observed when the oxidation state of the sulfur atom is increased.
- Example 43 Resolving CPSP and CPSP3 signals in the CPSP and CPSP3 mixture with high precision
- Analytes from a patient sample have a wide range of concentration. Often, the assays could not cover the required dynamic range, thus sample dilution is required. More than 10% of current immunoassays have a sample dilution protocol implemented in the assay file. This is not cost or time effective. With the implement of dual color acridinium-containing compounds and a dual channel instrument, the assay dynamic range can be easily extended such that sample dilution is not needed. The following example shows the bHCG assay range was expanded from 0-15,000 mIU/mL to 0-450,000 mIU/mL with improved sensitivity.
- the sample was triggered and read with a dual channel system.
- the assay curves were ploted based on signal measured in blue channel and green channel (see, FIGs. 45A-45C), the green channel also serves as the flag channel to indicate whether the measured signal corresponds to the rising section or the descending section of the Alternative Cal Curve.
- the assay dynamic range can cover all bHCG levels from a patient sample, thus eliminating the need for retest or auto-dilution.
- FIGS. 46A-46C when comparing to standard ARCHITECT® bHCG assay (e.g., Alternative Cal Curve), the method using the acridinium-containing compounds described herein has improved sensitivity and wider dynamic range.
- P-HBcAg/HBcAg detection Recombinant phosphorylated HBcAg (P-rHBcAg) and non-phosphorylated HBcAg (rHBcAg) were fully automated analyzed on the Alinity i dual channel instrument. P-HBcAg/HBcAg was captured and detected by monoclonal antibodies directed to phosphorylated or non-phosphorylated HBcAg. Detection was achieved by antibodies labeled with two different luminescent molecules, which emit photons of light at different wavelengths (blue and green). Using a dual detection system and specific filters enables the simultaneously detection of P-HBcAg and HBcAg.
- Table F shows the P-HBcAg/HBcAg duplex assay with simultaneous detection of P- HBcAg/HBcAg, which were determined in triplicate using recombinant P-HBcAg/HBcAg.
- RLUs for CPSP were corrected by subtraction of residual RLUs obtained from CPSP3 that passed the filter and vice versa.
- Abl binds to P-rHBcAg and Ab2 binds to rHBcAg.
- Example 47 Resolving IgG and IgM mixture with Dichloro-CPSP and Ditbutyl-CPSP and a Single Channel Detection System
- FIG. 48 shows the resolved signal of IgG and IgM from its mixture.
- the advantage of using the time domain for signal separation is simplicity of hardware requirement. It can be directly measured on a single channel system instead of dual channel system.
- Example 48 Duplex SARS-Cov2 IgG and IgM assay performed with Dichloro-CPSP and Ditbutyl-CPSP and a single channel detection system [585] A SARS-Cov2 IgG and IgM assay was performed on COVTD-vaccinated patient sample using Abbott on-market ARCHITECT® kit. The conjugates were replaced with dichloro- CPSP labeled anti-human IgG antibody and /or ditbutyl-CPSP labeled anti-human IgM antibody.
- FIG. 49A-49C show the separate measurement of IgG or IgM antibody levels from a single patient pre- and post-vaccination (Day 0, 15, 29, 57, 82) using either dichloro-CPSP labeled anti-human IgG antibody or ditbutyl-CPSP labeled anti-human IgM antibody.
- IgM levels initially spike 1 -2 weeks after exposure and then drop. IgG levels rise more slowly and then remain in high abundance over the course of months.
- FIG 49C shows simultaneous measurement of IgG and IgM antibody levels from a single patient pre and post vaccination (Day 0, 15, 29, 57, 82) using dichloro-CPSP labeled anti-human IgG antibody and Di-t-butyl-CPSP labeled anti-human IgM antibody.
- the dose response profile of the individual IgG and IgM assay agrees well with the duplex IgG and IgM assay.
- Example 49 A twostep dual channel combo assay run on Dual-Channel Detection System to Extend Assay Dynamic Range
- the assay kit comprises a microparticle bottle (off shelf from Abbott bHCG ARCHITECT® assay kit), a Conjugate bottle (1 nM Fab-CPSP3 Conjugate diluted in phosphate buffer), A Tracer bottle (6.25 IU/L bHCG-CPSP tracer in MES buffer).
- FIGs. 50C-50E shows the correlation of pregnant patients’ bHCG value measured on the two systems. For any sample above 15000 mIU/mL, they were first flagged on the Abbott commercial instrument, then the instrument performed a 15X sample dilution before re-running the test, whereas the dual channel system does not require the extra step of dilution and re-testing.
- a method for determining the amount of an analyte of interest in a biological sample comprising the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c.
- a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
- the calibration curve is obtained by the steps of: a. performing, in a reaction vessel, a sandwich immunoassay on the analyte of interest having a known concentration, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; b.
- a competitive immunoassay on analyte of interest having a known concentration
- the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical
- c. calculating the signal ratio of first detectable signal and the second detectable signal and d. varying concentrations of the analyte of interest and repeating steps a-c to generate a calibration curve using the signal ratios generated in step c.
- Clause 3 The method of clause 1 or clause 2, wherein the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
- Clause 4 The method of any of clauses 1 to 3, wherein the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a chromophore, a chemiluminescent label, or any combinations thereof.
- Clause 5 The method of any of clauses 1 to 4, wherein the first detectable signal comprises a first color and the second detectable signal comprises a second color.
- Clause 6 The method of any of clauses 1 to 4, wherein the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
- Clause 7 The method of any of clauses 1 to 6, wherein the method further comprises using a first detection component to detect the first detectable signal generated by the first detectable label and a second detection component to detect the second detectable signal generated by the second detectable label.
- Clause 8 The method of clause 7, wherein the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
- Clause 9 The method of any of clauses 1 to 8, wherein the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”), that competes with the analyte of interest wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
- analyte labeled with a detectable label (“competing analyte”)
- Clause 10 The method of any of clauses 1 to 8, wherein the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
- Clause 11 The method of any of clauses 1 to 10, wherein the first detection reagent or the second detection reagent is an antigen or an antibody.
- Clause 13 The method of any of clauses 1 to 12, wherein the capture reagent comprises an antibody or an antigen.
- Clause 14 The method of any of clauses 1 to 13, wherein the sandwich assay is performed in one step or two steps.
- a system for determining the amount of an analyte of interest in a biological sample comprising: a. a calibration curve for the analyte of interest; b. a sandwich immunoassay wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. a competitive immunoassay wherein the competitive immunoassay comprises a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d.
- a device comprising: i. a reaction vessel for performing the sandwich immunoassay and the competition assay; ii. a first detection component to determine the amount of the first detectable signal generated by the first detectable label; and iii. a second detection component to determine the amount of the second detectable signal generated by the second label, wherein: (i) the amount of the first detectable signal determined by the first detection component and the amount of the second detectable signal determined by the second detection component is used to calculate the signal ratio of first detectable signal and second detectable signal; and (ii) the signal ratio is used to determine the amount of the analyte of interest in the biological sample.
- Clause 17 The system of clause 16, wherein the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
- Clause 18 The system of clause 16 or clause 17, wherein the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a biotin/avidin label, a chromophore, a chemiluminescent label, or any combinations thereof.
- Clause 19 The system of any of clauses 16 to 18, wherein the first detectable signal comprises a first color and the second detectable signal comprises a second color.
- Clause 20 The system of any of clauses 16 to 18, wherein the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
- Clause 21 The system of any of clauses 16 to 20, wherein the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
- Clause 22 The system of any of clauses 16 to 21, wherein the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
- the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
- R 2 is selected from -SO3Z and -COOZ
- Clause 29 The compound of clause 28, wherein A is aryl that is unsubstituted or substituted with 1, 2, 3, 4, or 5 substituents selected from C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, and selenyl.
- Clause 30 The compound of any of clauses 27-29, wherein R 2 is -COOZ.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Immunology (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Molecular Biology (AREA)
- Hematology (AREA)
- Biomedical Technology (AREA)
- Urology & Nephrology (AREA)
- Physics & Mathematics (AREA)
- Biochemistry (AREA)
- Microbiology (AREA)
- Pathology (AREA)
- Biotechnology (AREA)
- Food Science & Technology (AREA)
- Medicinal Chemistry (AREA)
- General Physics & Mathematics (AREA)
- Analytical Chemistry (AREA)
- Cell Biology (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Virology (AREA)
- Communicable Diseases (AREA)
- Tropical Medicine & Parasitology (AREA)
- Plural Heterocyclic Compounds (AREA)
- Investigating Or Analysing Materials By The Use Of Chemical Reactions (AREA)
Abstract
Provided herein are methods, systems, and kits for determining the concentration or level of an analyte of interest using a competitive and sandwich immunoassay. Also provided herein are acridinium-containing and acridone-containing compounds for use in assays to detect the presence of or determine the concentration or level an analyte of interest in a sample, e.g., a biological sample.
Description
METHODS, SYSTEMS, COMPOUNDS, AND KITS FOR DETERMINING AN ANALYTE OF INTEREST IN A TEST SAMPLE
RELATED APPLICATION INFORMATION
[1] This application claims priority to U.S. Application No. 63/615,305 filed on December 28, 2023, the contents of which are herein incorporated by reference.
FIELD
[2j Provided herein are methods, systems, and kits for determining the concentration or level of an analyte of interest using a competitive and sandwich immunoassay. Also provided herein are acridinium-containing and acridone-containing compounds for use in assays to detect the presence of or determine the amount or level of an analyte of interest in a biological sample. In some aspects, the acridinium-containing and acridone-containing compounds described herein can be used in the methods, systems, and kits for determining the concentration or level of an analyte of interest using a competitive and sandwich immunoassay.
BACKGROUND
[3] For the past several decades, immunoassays have been performed using fluorescence, chemiluminescence, or other means of generating a signal in response to an analyte of interest. Immunoassays typically involve combining a sample suspected of containing an antigen or antibody with a reagent comprising a first antibody or first antigen which can be attached to a solid support, e.g., a microparticle, to form a reaction mixture. The antigen or antibody, if present in the sample, specifically binds to the first antibody or first antigen. A conjugate, which comprises a second antibody or second antigen having a detectable label attached thereto, is introduced to the reaction mixture and specifically binds to the antigen or antibody, which is specifically bound to the first antibody or first antigen, which, as stated previously, can be attached to a solid support. Such an assay is referred to as a sandwich immunoassay or an immunometric assay. The signal attributable to the detectable label is measured after unbound conjugate is removed from the reaction mixture, typically by performing a wash step. The signal that is derived from the total volume of the reaction mixture is measured and compared to a calibration curve to establish the concentration of antigen or antibody present in the sample.
[4] Immunoassays generally require that the sample containing the analyte of interest be diluted (e.g., pre-dilution) prior to performing the assay. Samples are diluted to lower the concentration of the analyte in the sample to ensure that the amount of analyte is below acceptable limits and to help minimize interferences from other compounds in the sample that could artificially affect the analysis. When samples are not diluted sufficiently enough, the analyte concentration can be underestimated for high analyte positive samples. When samples are over-diluted, low analyte positive samples can be missed as a negative sample.
[5] An immunoassay that includes a washing step to remove unbound sample analyte before introducing the conjugate antibody is generally referred to as a “two-step assay.” An immunoassay that introduces the conjugate antibody and the analyte to antibody-coated microparticles together without intermediate washing steps is generally referred to as a “one- step” assay. Both one-step and two-step formats can exhibit limited assay dynamic range, thus some samples require re-testing with dilution. A “hook effect” or “prozone phenomenon” is a phenomenon of falsely low values observed in an immunoassay when an overwhelming amount of antigen or antibody is present in a “one-step assay” format. Hook effect is caused by insufficient capture reagent and detection reagent in an immunoassay, which limits the assay dynamic range.
[6] Acridinium molecules with chemiluminescent properties are useful in the field of in vitro diagnostics (IVDs), where the amount of light produced by acridinium in an assay is proportional to an analyte of interest. 9-Carbonylacridiniums are a common type of acridinium used in commercialized chemiluminescence based IVDs, where a phenyl ester or acylsulfonamide moiety at the acridinium 9-position facilitates an efficient reaction pathway for the generation of blue light (ca. 445 nm) upon treatment with an alkaline peroxide solution. The molecular structure of acridinium has been modified extensively in the prior art to affect solubility, charge, reaction kinetics, and quantum efficiency of the chemiluminescence (CL). By comparison, there are few reports on structural modifications which change the color of light emitted from the triggered acridinium.
[7] Previously, multicolor acridinium was demonstrated by attaching fluorophores to acridinium to red-shift the CL color, where triggering of acridinium resulted in an energy transfer and subsequent emission from the corresponding fluorophore. Although this technique was used to generate acridinium multicolor CL emission (green, yellow, red, or near-IR light),
the energy transfer inevitably has some energy loss (and lower CL intensity) proportional to the quantum yield of the fluorophore. Also, the energy transfer efficiency is not absolute with a small percentage of blue light being emitted, which is increased if the fluorophore is chemically unstable to the harsh CL pre-trigger/trigger conditions. Bioconjugation of the acridinium- fluorophore constructs was also problematic as the addition of the fluorophore caused an increase in hydrophobicity resulting in unstable, and difficult to purify, protein conjugates.
[8] Substituted acridiniums have previously been prepared through multiple steps beginning with a substituted isatin. This procedure is limited by availability of the isatin and the stability of the substituent to the subsequent reaction conditions. Given the importance of having modified acridiniums for the field of IVDs, improved methods for synthesizing substituted acridiniums are needed. A more preferable method would involve direct modification of an existing commercially available acridinium molecule. Because acridinium is electron deficient and chemically unstable, there are few reactions that can be employed for direct modification.
[9] There remains a need for methods (e.g., immunoassays), systems, and kits for detecting analytes of interest in a sample which exhibit increased sensitivity and wide dynamic range, while exhibiting reducing hook effect. Further, there remains a need for modified acridiniums that have unique chemical and chemiluminescent properties, as well methods of producing same.
BRIEF DESCRIPTION OF THE DRAWINGS
[10] FIG. 1 shows a drawing of the beta-human chorionic gonadotropin (bHCG) assay performed in a competitive and sandwich dual channel assay format to Example 1. CPSP: 4- ( 10-(3 -sulfopropyl)-N-tosyl- 10X.4-acridine-9-carboxamido)butanoic acid.
[11] FIG. 2A-2C show the signal generated from the blue and green channels as described in Example 1 that reflect a bHCG assay in competitive format (FIG. 2A) and in an one-step sandwich format (FIG. 2B). FIG. 2C shows a final calibration curve based on the signal ratio between the competitive format (FIG. 2A) and the one-step sandwich format (FIG. 2B).
[12] FIG. 2D shows that with decreased sample and microparticle volumes, the assay dynamic range can be extended using the proposed dual channel assay format to 450,000 mIU/mL, which is 30 times that of the current on-market assay (15,000m lU/mL).
[13] FIG. 3 shows competitive immunoassay and two-step sandwich immunoassay being performed in a single reaction vessel according to Example -2. CPSP: 4-(10-(3-sulfopropyl)-N- tosyl- 10X4-acridine-9-carboxamido)butanoic acid.
[14] FIG. 4A-4C show the signal generated from the blue and green channels as described in Example 2 that reflect a SARS-CoV-2 assay in competitive format (FIG. 4A) and in a two- step sandwich format (FIG. 4B). FIG. 4C shows the ratio of signal detected in the sandwich immunoassay format and the signal detected in the competitive immunoassay format.
[15] FIG. 5A is the absorbance spectrum of Dichloro-CPSP.
[16] FIG. 5B is the chemiluminescence spectrum of Dichloro-CPSP.
[17] FIG. 6A is the absorbance spectrum of Dibromo-CPSP.
[18] FIG. 6B is the chemiluminescence spectrum of Dibromo-CPSP.
[19] FIG. 7A is the absorbance spectrum of Diiodo-CPSP.
[20] FIG. 7B is the chemiluminescence spectrum of Diiodo-CPSP.
[21] FIG. 8A is the absorbance spectrum of Disulfanato-CPSP.
[22] FIG. 8B is the chemiluminescence spectrum of Disulfanato-CPSP.
[23] FIG. 9A is the absorbance spectrum of Dinitro-CPSP.
[24] FIG. 9B is the chemiluminescence spectrum of Dinitro-CPSP.
[25] FIG. 10A is the absorbance spectrum of Di-t-butyl-CPSP.
[26] FIG. 10B is the chemiluminescence spectrum of Di-t-butyl-CPSP.
[27] FIG. 11 illustrates the chemiluminescence reaction rates of DiChloro-CPSP and Di-t- butyl-CPSP after adding alkaline peroxide. CPSP: 4-(10-(3-sulfopropyl)-N-tosyl-10X4-acridine- 9-carboxamido)butanoic acid.
[28] FIG. 12A is the absorbance spectrum of CPSP.
[29] FIG. 12B is the chemiluminescence spectrum of CPSP.
[30] FIG. 13A is the absorbance spectrum of CPSP2. CPSP2: 4-(10-(3-sulfopropyl)-2-((3- sulfopropyl)thio)-N-tosyl- 10X4-acridine-9-carboxamido)butanoic acid.
[31] FIG. 13B is the chemiluminescence spectrum of CPSP2.
[32] FIG. 14A is the absorbance spectrum of CPSP3. CPSP3: 4-(10-(3-sulfopropyl)-2,7- bis((3-sulfopropyl)thio)-N-tosyl-10X4-acridine-9-carboxamido)butanoic acid.
[33] FIG. 14B is the chemiluminescence spectrum of CPSP3.
[34] FIG. 15A is the absorbance spectrum of CPSP3 -sulfone.
[35] FIG. 15B is the chemiluminescence spectrum of CPSP3 -sulfone.
[36] FIG. 16A is the absorbance spectrum of CPSP3 -sulfoxide.
[37] FIG. 16B is the chemiluminescence spectrum of CPSP3 -sulfoxide.
[38] FIG. 17A is the absorbance spectrum of CPSP3-BisSulfone.
[39] FIG. 17B is the chemiluminescence spectrum of CPSP3-BisSulfone.
[40] FIG. 18A is the absorbance spectrum of SP3-Acridone. SP3-Acridone: 3,3'-((9-oxo-
10-(3 -sulfopropyl)-9, 10-dihydroacridine-2,7 -diyl)bis(sulfanediyl))bis(propane- 1 -sulfonic acid).
[41] FIG. 18B is the fluorescence spectrum of SP3-Acridone.
[42] FIG. 19A is the absorbance spectrum of SP3-Acridone BisSulfoxide.
[43] FIG. 19B is the fluorescence spectrum of SP3-Acridone BisSulfoxide.
[44] FIG. 20A is the absorbance spectrum of SP3-Acridone Sulfoxide Sulfone.
[45] FIG. 20B is the fluorescence spectrum of SP3-Acridone Sulfoxide Sulfone.
[46] FIG. 21 A is the absorbance spectrum of SP3-Acridone BisSulfone.
[47] FIG. 2 IB is the fluorescence spectrum of SP3-Acridone BisSulfone.
[48] FIG. 22 A is the absorbance spectrum of SP3-Acridone Sulfoxide.
[49] FIG. 22B is the fluorescence spectrum of SP3-Acridone Sulfoxide.
[50] FIG. 23 A is the absorbance spectrum of DiAmino-CPSP.
[51] FIG. 23B is the chemiluminescence spectrum of DiAmino-CPSP.
[52] FIG. 24A is the absorbance spectrum of Ph-CPSP.
[53] FIG. 24B is the chemiluminescence spectrum of Ph-CPSP.
[54] FIG. 25 A is the absorbance spectrum of HPh-CPSP.
[55] FIG. 25B is the chemiluminescence spectrum of HPh-CPSP.
[56] FIG. 26A is the absorbance spectrum of DMAPh-CPSP.
[57] FIG. 26B is the chemiluminescence spectrum of DMAPh-CPSP.
[58] FIG. 27A is the absorbance spectrum of 4-CyanoPh-CPSP.
[59] FIG. 27B is the chemiluminescence spectrum of 4-CyanoPh-CPSP.
[60] FIG. 28 A is the absorbance spectrum of Pyrrolyl-CPSP.
[61] FIG. 28B is the chemiluminescence spectrum of Pyrrolyl-CPSP.
[62] FIG. 29 A is the absorbance spectrum of Furanyl-CPSP.
[63] FIG. 29B is the chemiluminescence spectrum of Furanyl-CPSP.
[64] FIG. 30A is the absorbance spectrum of 5-Cyano-Thienyl-CPSP.
[65] FIG. 30B is the chemiluminescence spectrum of 5-Cyano-Thienyl-CPSP.
[66] FIG. 31 A is the absorbance spectrum of 5-Me-Thienyl-CPSP.
[67] FIG. 3 IB is the chemiluminescence spectrum of 5-Me-Thienyl-CPSP.
[68] FIG. 32A is the absorbance spectrum of 5-MeO-Thienyl-CPSP.
[69] FIG. 32B is the chemiluminescence spectrum of 5-MeO-Thienyl-CPSP.
[70] FIG. 33A is the absorbance spectrum of 4-MeO-Thienyl-CPSP.
[71] FIG. 33B is the chemiluminescence spectrum of 4-MeO-Thienyl-CPSP.
[72] FIG. 34A is the absorbance spectrum of 3-MeO-Thienyl-CPSP.
[73] FIG. 34B is the chemiluminescence spectrum of 3-MeO-Thienyl-CPSP.
[74] FIG. 35A is the absorbance spectrum of 5-MeS-Thienyl-CPSP.
[75] FIG. 35B is the chemiluminescence spectrum of 5-MeS-Thienyl-CPSP.
[76] FIG. 36A is the absorbance spectrum of Thienothiophenyl-CPSP.
[77] FIG. 36B is the chemiluminescence spectrum of Thienothiophenyl-CPSP.
[78] FIG. 37A is the absorbance spectrum of Bis(5-Me-Thienyl)-CPSP.
[79] FIG. 37B is the chemiluminescence spectrum of Bis(5-Me-Thienyl)-CPSP.
[80] FIG. 38A is the absorbance spectrum of DiFuranyl-CPSP.
[81] FIG. 38B is the chemiluminescence spectrum of DiFuranyl-CPSP.
[82] FIG. 39A is the absorbance spectrum of TAMRA-Ph-CPSP.
[83] FIG. 39B is the chemiluminescence spectrum of TAMRA-Ph-CPSP.
[84] FIG. 40 illustrates the treatment of the mono- or di-substituted acridinium-sulfides with standard alkaline peroxide conditions resulted in a bright blue-green or green chemiluminescence (CL) color, with a CL Xmax = 490 nm and 505 nm, respectively.
[85] FIG. 41 illustrates that with the chosen optical filters (channel 1 is blue and channel 2 is green in the figure), signals from CPSP or CPSP3 can be resolved at high precision. CPSP: 4- ( 10-(3 -sulfopropyl)-N-tosyl- 10X4-acridine-9-carboxamido)butanoic acid.
[86] FIG. 42 illustrates the minimal crosstalk between CPSP and CPSP3 when the ratio between the two compounds is within a 100-fold difference. CPSP: 4-(10-(3-sulfopropyl)-N- tosyl- 10X4-acridine-9-carboxamido)butanoic acid.
[87] FIG. 43 shows the immunoglobulin-G (IgG) and immunoglobulin-M (IgM) antibody levels from a single patient post vaccination using simultaneous dual channel measurements.
[88] FIG. 44A is a schematic of the extension of the dynamic range using a two-step assay using two different acridinium-containing compounds described herein in Example 45.
[89] FIG. 44B is a schematic of the dual channel system of Example 45. The blue channel shown in this figure assesses signal coming from the tracer and the green channel assesses the signal coming from CPSP3 (as part of the antibody conjugate which contains CPSP3).
[90] FIG. 45A is an assay curve plotted based on signal measured in blue (hook) channel according to Example 45.
[91] FIG. 45B is an assay curve plotted based on signal measured in green (sandwich) channel according to Example 45.
[92] FIG. 45C is the combination of FIGs. 45 A and 45B illustrating that the assay dynamic range can cover all bHCG levels from a patient sample.
[93] FIG. 46A-46C illustrates the comparison between the standard ARCHITECT® bHCG assay (e.g., Alternative Cal Curve) and the assay disclosed herein using the acridinium- containing compounds, showing improved sensitivity and wider dynamic range.
[94] FIG. 47 illustrates the oxidation of SP3-Acridone, as described in Example 22.
[95] FIG. 48 illustrates the resolved signal of a human IgG and IgM mixture using dichloro-CPSP and ditbutyl-CPSP and a single channel detection system according to Example 47.
[96] FIG. 49A shows the result of a single channel SARS-Cov2 IgM assay performed with ditbutyl-CPSP according to Example 48.
[97] FIG. 49B shows the result of a single channel SARS-Cov2 IgG assay performed with dichloro-CPSP according to Example 48.
[98] FIG. 49C shows the result of a duplex SARS-Cov2 IgG and IgM assay performed with dichloro-CPSP and ditbutyl-CPSP, respectively, according to Example 48.
[99] FIG. 50A illustrates the low-end calibration curve for the bHCG assay in Example 49.
[100] FIG. 50B illustrates the high-end calibration curve for the bHCG assay in Example 49.
[101] FIG. 50C illustrates the platform comparison of patient samples for the bHCG assay (low end) in Example 49.
[102] FIG. 50D illustrates the platform comparison of patient samples for the bHCG assay (mid-range) in Example 49.
[103] FIG. 50E illustrates the platform comparison of patient samples for the bHCG assay (high range) in Example 49.
SUMMARY
[104] In some aspects, provided herein are methods for determining the amount of an analyte of interest in a biological sample. The method comprises the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. performing, in the same reaction vessel, a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
[105] In still fiirther aspects of the above method, the calibration curve is obtained by the steps of: a. performing, in a reaction vessel, a sandwich immunoassay on the analyte of interest having a known concentration, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; b. performing, in the same reaction vessel, a competitive immunoassay on analyte of interest having a known concentration, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second
detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and c. calculating the signal ratio of first detectable signal and the second detectable signal; and d. varying concentrations of the analyte of interest and repeating steps a-c to generate a calibration curve using the signal ratios generated in step c.
[106] In still further aspects of the above method, the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
[107] In yet still further aspects of the above method, the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a chromophore, a chemiluminescent label, or any combinations thereof.
[108] In yet still further aspects of the above method, the first detectable signal comprises a first color and the second detectable signal comprises a second color.
[109] In yet still further aspects of the above method, the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
[110] In yet still further aspects of the above method, the method further comprises using a first detection component to detect the first detectable signal generated by the first detectable label and a second detection component to detect the second detectable signal generated by the second detectable label.
[111] In yet still further aspects of the above method, the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
[112] In yet still further aspects of the above method, the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”), that competes with the analyte of interest wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
[113] In yet still further aspects of the above method, the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
[114] In yet still further aspects of the above method, the first detection reagent or the second detection reagent is an antigen or an antibody.
[115] In yet still further aspects of the above method, the analyte of interest and the competing analyte are each captured on a solid support.
[116] In yet still further aspects of the above method, the capture reagent comprises an antibody or an antigen.
[117] In yet still further aspects of the above method, the sandwich assay is performed in one step or two steps.
[118] In yet still further aspects of the above method, the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained from the subject.
[119] In still further aspects of the above method, the analyte of interest is bHCG, SARS- CoV-2, hepatitis B core antigen (HBcAg), phosphorylated hepatitis B core antigen, or combinations thereof.
[120] In yet another aspect, also provided herein is a method for determining the amount of an analyte of interest in a biological sample. The method comprises the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a first reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. performing, in the second reaction vessel, a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein: (i) the sandwich immunoassay and competitive immunoassay are performed simultaneously, or sequentially, in any order: (ii) the second detectable label is identical to the first detectable label; (iii) the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and (iv) the first detectable signal and the second detectable signal are detected by an instrument comprising a single detection component; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
[121] In yet another aspect, also provided herein is a system for determining the amount of an analyte of interest in a biological sample. The system can comprise: a. a calibration curve for the analyte of interest; b. a sandwich immunoassay wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. a competitive immunoassay wherein the competitive immunoassay comprises a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. a device comprising: i. a reaction vessel for performing the sandwich immunoassay and the competition assay; ii. a first detection component to determine the amount of the first detectable signal generated by the first detectable label; and iii. a second detection component to determine the amount of the second detectable signal generated by the second label, wherein: (i) the amount of the first detectable signal determined by the first detection component and the amount of the second detectable signal determined by the second detection component is used to calculate the signal ratio of first detectable signal and second detectable signal; and (ii) the signal ratio is used to determine the amount of the analyte of interest in the biological sample.
[122] In the above-described system, the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
[123] In the above-described system, the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a biotin/avidin label, a chromophore, a chemiluminescent label, or any combinations thereof.
[124] In the above-described system, the first detectable signal comprises a first color and the second detectable signal comprises a second color.
[125] In the above-described system, the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
[126] In the above-described system, the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
[127] In the above-described system, the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
[128] In the above-described system, the first detection reagent, the second detection reagent or the first detection agent and the second detection reagent is an antigen or an antibody.
[129] In the above-described system, the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
[130] In the above-described system, the capture reagent comprises an antibody or an antigen.
[131] In the above-described system, the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained ftom the subject.
Y is selected from nitrogen, oxygen, and sulfur;
when Y is nitrogen, R1 is -SCh-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryi, eycloalkyl, heterocyclyl, arylalkyl. heteroarylalkyl. cycloalkylalkyl, or heter ocycly lalkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)-
N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents.
[133] In some aspects in the above compound, Y is nitrogen.
[134] In some aspects in the above compound, A is aryl that is unsubstituted or substituted with 1, 2, 3, 4, or 5 substituents selected from C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl.
[135] In some aspects in the above compound, R2 is -COOZ.
[136] In some aspects in the above compound, Z is selected from hydrogen, C1-C4 alkyl, or N-hydroxysuccinimide.
[137] In some aspects in the above compound, Li and L2 are each independently C1-C4 alkylene.
[138] In some aspects in the above compound, Ra, Rb, Rc, Rd, Re, and Rf are each hydrogen.
[139] In some aspects in the above compound, R’, R”, or both R’ and R” are hydrogen.
[140] In some aspects in the above compound, R’, R”, or both R’ and R” are -S-L3-SO3H.
[141] In some aspects in the above compound, R’, R”, or both R’ and R” are -S(=O)-L3-
SO3H.
[142] In some aspects in the above compound, R’, R”, or both R’ and R” are -SO2-L3-SO3H.
[143] In some aspects in the above compound, R’ is -SO2-L3-SO3H and R” is -S-L3-SO3H.
[144] In some aspects in the above compound, R’ is -S-L3-SO3H and R” is one of H, Cl, Br, I, -NO2, t-butyl, or -SO3H.
[145] In some aspects in the above compound, R’ is -S-L3-SO3H and R” is H.
[146] In some aspects in the above compound, R’ is -SO2-L3-SO3H and R” is one of Cl, Br,
I, -NO2, t-butyl, or -SO3H.
[147] In some aspects in the above compound, R’ is -(S=O)-L3-SO3H and R” is -S-L3-SO3H.
[148] In some aspects in the above compound, R’ is -(S=O)-L3-SO3H and R” is one of Cl,
Br, I, -NO2, t-butyl, or -SO3H.
[149] In some aspects in the above compound, R’, R”, or both R’ and R” are a substituted or unsubstituted phenyl group.
[150] In some aspects in the above compound, R’, R”, or both R’ and R” are a phenyl group substituted with at least one of hydroxy, cyano, alkylamino, dialkylamino, or aminoalkyl.
[151] In some aspects in the above compound, R’, R”, or both R’ and R” are a substituted or unsubstituted heteroaryl group.
[152] In some aspects in the above compound, R’, R”, or both R’ and R” are a substituted or unsubstituted heteroaryl group selected from pyrrolyl, furanyl, thienyl, or thienothiophenyl.
[153] In some aspects in the above compound, the compound has the Formula (Ila):
wherein R’ and R” are defined above for Formula (I).
[154] In some aspects in the above compound, the compound is:
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), and Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
[159] Also provided herein are methods of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring. The method can comprise: reducing the acridinium ring to an acridan intermediate using a first solution comprising at least one reducing agent and at least one acid; and reacting the acridan intermediate with an electrophile to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring.
[160] In some aspects of the above-described method, the at least one acid comprises at least one of ammonium chloride, formic acid, acetic acid, benzoic acid, hydrofluoric acid, phosphoric acid, sulfurous acid, hypochlorous acid, citric acid, carbonic acid, nitrous acid, oxalic acid, boric acid, ammonium sulfate, and ammonium oxalate.
[161] In still further aspects of above-described method, the at least one reducing agent comprises zinc or a hydride.
[162] In still further aspects of above-described method, the zinc comprises elemental zinc, a zinc alloy, or a combination of both.
[163] In still further aspects of above-described method, the hydride comprises sodium borohydride, lithium aluminum hydride, sodium cyanoborohydride, or sodium triacetoxyborohydride.
[164] In still further aspects of above-described method, the first solution further comprises a water-miscible alcohol, preferably ethanol.
[165] In still further aspects of above-described method, the acridinium ring is reduced at temperature in a range from about 50°C to about 90°C, preferably about 60°C to about 80°C.
[166] In still further aspects of above-described method, the acridinium-containing compound is represented by the compound of Formula (I) as described above.
[167] In still further aspects of above-described method, R’ = R” = H.
[168] In still further aspects of above-described method, the acridan intermediate is represented by Formula (III):
f are defined above for Formula (I).
[169] In still further aspects of above-described method, the electrophile comprises at least one of Cl, Br, I, NO2, SO3H, or t-butyl.
[170] In still further aspects of above-described method, the electrophile is Cl and the products are Formula (IVa), (IVb), or both (IVa) and (IVb).
[171] In still further aspects of above-described method, a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with Cl at the 2- or the 2,7- position, or both, of the acridinium ring includes a chlorination reaction.
[172] In still further aspects of above-described method, the method further comprises mixing the acridan intermediate of Formula (III) with a second solution in a container, and stirring for about 12 hours to about 24 hours, preferably about 16 hours to about 20 hours; adding an aqueous solution of at least one oxidant to the container, in portions, over about 30 minutes to about 90 minutes, preferably about 45 minutes to about 75 minutes; and isolating and optionally
purifying the product comprising the compound of Formula (IVa), (IVb), or both (TVa) and (IVb).
[173] In still fiirther aspects of above-described method, the second solution comprises a chloride salt, N-chlorosuccinimide, and p-toluenesulfonic acid in a water/acetonitrile mixture.
[174] In still further aspects of above-described method, the oxidant comprises FeCh, 2,3- Dichloro-5,6-dicyano-l,4-benzoquinone (DDQ), chloranil, or ceric ammonium nitrate (CAN).
[175] In still further aspects of above-described method, the electrophile is Br and the products are Formula (Va), (Vb), or both (Va) and (Vb).
[176] In still further aspects of above-described method, a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with Br at the 2- or the 2,7- position, or both, of the acridinium ring includes a bromination reaction.
[177] In still further aspects of above-described method, the method further comprises dissolving the acridan intermediate in a third solution in a container; adding bromine to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Va), (Vb), or both (Va) and (Vb).
[178] In still further aspects of above-described method, the electrophile is I and the products are Formula (Via), (VIb), or both (Via) and (VIb).
[179] In still further aspects of above-described method, a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with I at the 2- or the 2,7- position, or both, of the acridinium ring includes an iodination reaction.
[180] In still further aspects of above-described method, the method further comprises dissolving the acridan intermediate in a third solution in a container; adding iodine monochloride to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb).
[181] In still further aspects of above-described method, the electrophile is NO2 and the products are Formula (Vila), (Vllb), or both (Vila) and (Vllb).
[182] In still further aspects of above-described method, a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with NO2 at the 2- or the 2,7- position, or both, of the acridinium ring includes a nitration reaction.
[183] In still further aspects of above-described method, the method further comprises dissolving the acridan intermediate in a third solution in a container; adding nitric acid to the container; and isolating and optionally purifying the product comprising the compound of Formula (Vila), (Vllb), or both (Vila) and (Vllb).
[184] In still further aspects of above-described method, the electrophile is SO3H and the products are Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
[185] In still further aspects of above-described method, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with SO3H at the 2- or the 2,7- position, or both, of the acridinium ring includes a sulfonation reaction.
[186] In still further aspects of above-described method, the method further comprises: dissolving the acridan intermediate in a fourth solution in a container; adding chlorosulfonic acid to the container and stirring for about 5 minutes to about 30 minutes; adding water to the container and stirring for about 10 minutes to about 30 minutes; and isolating and optionally purifying the product comprising the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
[187] In still further aspects of above-described method, the electrophile is t-butyl and the products are Formula (IXa), (IXb), or both (IXa) and (IXb).
[188] In still further aspects of above-described method, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with t- butyl at the 2- or the 2,7- position, or both, of the acridinium ring includes a Friedel-Craft alkylation reaction.
[189] In still further aspects of above-described method, wherein the method further comprises: dissolving the acridan intermediate in a fifth solution in a container; adding a Lewis acid to the container and heating at about 40°C to about 60°C for about 18-24 hours; reducing solvent in vacuo followed by the addition of a water/acetonitrile solution to the container; adding an aqueous solution of an oxidant to the container dropwise over time; and isolating and optionally purifying the product comprising the compound of Formula (IXa), (IXb), or both (IXa) and (IXb).
[190] In still further aspects of the above-described method, the nitro group(s) of the compound of Formula (Vila) or (Vllb) are reduced to an amine group using a reducing agent to produce the compound of Formula (Xa) or (Xb), respectively.
[1.91] In still further aspects of the above-described method, the reducing agent comprises SnCh-
[192] Also provided herein are methods of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring comprising: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a boronic acid comprising A’ or a boronate ester comprising A’, wherein A’ is a substituted or unsubstituted aryl group or a substituted or unsubstituted heteroaryl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
[193] In still further aspects of the above-described method, the acridinium compound is functionalized by:
(i) at least one phenyl group to produce a compound of Formula (Xia) or (Xlb);
(ii) at least one furanyl group to produce a compound of Formula (Xllla) or (Xlllb);
(iii) at least one thienyl group to produce a compound of Formula (XlVa) or (XlVb); or
(iv) at least one thienothiophenyl group to produce a compound of Formula (XV), wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
[194] In still further aspects of the above-described method, the acridinium compound is functionalized by at least one pyrrolyl group to produce a compound of Formula (Xlla) or
(Xllb), wherein the boronic acid comprising A’ or the boronate ester comprising A’ is tertbutyloxycarbonyl (Boc)-protected, and wherein the method comprises after the stirring step: isolating the product comprising the acridinium-containing compound comprising a Boc-protected pyrrolyl group; dissolving the acridinium-containing compound comprising the Boc-protected pyrrolyl group in dichloromethane and trifluoroacetic acid for about 2-6 hours; removing the solvent in vacuo; and optionally purifying the product comprising the compounds of Formula Xlla or Xllb, wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =0, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
[195] In still further aspects of the above-described method, the reactant compound comprises iodo-CPSP or diiodo-CPSP.
[196] Also provided herein are methods of producing a sulfoxide of Formula (XVIII) or (XIX), said method comprising: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating and optionally purifying the sulfoxide compound of Formula (XVIII) or (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[197] In still further aspects of the above-described method, the at least one organic solvent comprises tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, or dimethylformamide (DMF).
[198] In still further aspects of the above-described method, the oxidizing agent comprises mCPBA.
[199] In still further aspects of the above-described method, the compound of Formula (XVII) is CPSP3.
[200] Also provided herein are methods of producing a BisSulfone compound of Formula (XXII) comprises: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; quenching an oxidation reaction with dimethyl sulfide and adding an amount of water; and isolating and optionally purifying the BisSulfone compound of Formula (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[201] In still further aspects of the above-described method, the at least one organic solvent comprises tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
[202] In still further aspects of the above-described method, the oxidizing agent comprises mCPBA.
[203] In still further aspects of the above-described method, the compound of Formula (XVII) is CPSP3.
[204] In still further aspects of above-described method, wherein the method further comprises: reacting the product comprising the acridinium functionalized at the 2 -position with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (XVI).
[205] In still further aspects of above-described method, the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
[206] In still further aspects of above-described method, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
[207] In still further aspects of above-described method, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
[208] In still further aspects of above-described method, wherein the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 ,l'-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (rBuXPhos Pd G3).
[209] In still further aspects of above-described method, the method further comprises isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
[210] In still further aspects of above-described method, the method further comprises reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium disulfide compound of Formula (XVII).
[211] In still further aspects of above-described method, the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
[212] In still further aspects of above-described method, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
[213] In still further aspects of above-described method, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
[214] In still further aspects of above-described method, wherein the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
[215] In still further aspects of above-described method, the method further comprises: isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
[216] In still further aspects of above-described method, the method further comprises: reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (I), wherein R’ is -S-L3-SO3H and R” is selected from Cl, Br, I, -NO2, t-butyl, or -SO3H; oxidizing the sulfide moiety to a sulfone; reacting the R” group selected from Cl, Br, I, -NO2, t-butyl, or -SO3H with the reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium sulfone compound of Formula (XXI).
[217] In still further aspects of above-described method, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
[218] In still further aspects of above-described method, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
[219] In still further aspects of above-described method, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
[220] In still further aspects of above-described method, wherein the Pd-containing catalyst comprises 2-Di-terributylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
[221] In still further aspects of above-described method, wherein the sulfide moiety is oxidized using meta-Chloroperoxybenzoic acid (mCPBA).
[222] In still further aspects of above-described method, wherein the method further comprises isolating and optionally purifying the acridinium sulfone compound of Formula (XXI).
Q is -SO3Z or H;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Li is selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl, and wherein at least one of R’ or R” has to be -S(=O)-L3-X, -S-L3-X, or -SO2-L3-X; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents.
[224] In some aspects in the above compound, Q is -SO3Z, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents, preferably -SO3H.
[225] In some aspects in the above compound, Li is a C1-C10 alkylene, preferably — CH2CH2CH2— .
[226] In some aspects in the above compound, Ra, Rb, Rc, Rd, Re, and Rf are each hydrogen.
[227] In some aspects in the above compound, R’, R”, or both R’ and R” are -S-L3-SO3H.
[228] In some aspects in the above compound, R’, R”, or both R’ and R” are -S(=O)-L3- SO3H.
[229] In some aspects in the above compound, R’, R”, or both R’ and R” are -SO2-L3-SO3H.
[230] In some aspects in the above compound, R’ is -SO2-L3-SO3H and R” is -S-L3-SO3H.
[231] In some aspects in the above compound, R’ is -SO2-L3-SO3H and R” is -S(=O)-L3-
SO3H.
[232] In some aspects in the above compound, R’, R”, or both R’ and R’ are a halogen, preferably I.
[233] In some aspects, the compound has the Formula (XXIV):
wherein R’ and R” are defined above for Formula (XXIII).
[237] Also provided herein are methods of functionalizing an acridone-containing compound at a 2,7-position of an acridone ring with iodine, comprising: dissolving an acridone-containing compound in a third solution; adding iodine monochloride to the third solution and stirring for about 6 h to about 18 h under pressure to produce the acridone-containing compound functionalized at the 2,7-positions in a mixture; dissolving the mixture in DMSO; and isolating and optionally purifying the diiodo-acridone compound.
[238] In further aspects of above-described method, the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H.
[239] In still further aspects of above-described method, the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H and Li is — CH2CH2CH2 — .
[240] In still further aspects of above-described method, the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H, Li is — CH2CH2CH2 — , and Q is -SChH.Also provided herein are methods of producing an acridone disulfide of Formula (XXVI) comprising: reacting a dihalo-acridone with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridone disulfide product of Formula (XXVI), wherein Li, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (XXIII).
[241] In further aspects of above-described method, the dihalo-acridone is compound (35).
[242] In still further aspects of above-described method, the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
[243] In still further aspects of above-described method, the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
[244] In still further aspects of above-described method, the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
[245] In still further aspects of above-described method, the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3).
[246] In still further aspects of above-described method, the method further comprises isolating and optionally purifying the acridone disulfide compound of Formula (XXVI).
[247] Also provided herein are methods of producing a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI), comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating the bis sulfoxide of Formula (XXVIII), sulfoxide sulfone of Formula (XXIX), and/or bis sulfone of Formula (XXXI) and optionally separating and purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[248] In further aspects of above-described method, the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, or dimethylformamide (DMF).
[249] In still further aspects of above-described method, the oxidizing agent comprises mCPBA.
[250] In still further aspects of above-described method, the compound of Formula (XXVI) is compound (39).
[251] In still further aspects of above-described method, after addition of the oxidizing agent, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
[252] Also provided herein are methods of producing a sulfoxide of Formula (XXVII) comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; dropwise adding an eighth solution comprising an oxidizing agent to the contents of the container; stirring the contents of the container; quenching the reaction with dimethyl sulfide; and isolating the sulfoxide of Formula (XXVII) and optionally purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[253] In further aspects of above-described method, the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, or dimethylformamide (DMF).
[254] In still further aspects of above-described method, the oxidizing agent comprises mCPBA.
[255] In still further aspects of above-described method, wherein the compound of Formula (XXVI) is compound (39).
[256] In still further aspects of above-described method, wherein the eighth solution further comprises at least one organic solvent.
[257] In still further aspects of above-described method, wherein after addition of the eighth solution, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
[258] In still further aspects of above-described method, wherein the dimethyl sulfide is added dropwise.
[259] In still further aspects of above-described method, wherein immediately after the addition of dimethyl sulfide, water is added.
[260] Also provided herein are methods of assessing and monitoring stage or phase of chronic Hepatitis B (HBV) infection or monitoring response to a treatment for chronic HB V in a subject. The method comprises the steps of: a) performing an assay to detect the presence or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject diagnosed with chronic HBV or receiving a treatment for chronic HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg-second detection antibody complex is formed; and b) assessing and monitoring stage or phase of chronic HBV infection or monitoring in response to the treatment for chronic HBV based on the presence or level of HBcAg and P-HBcAg in the at least one sample, wherein the first detectable label and second detectable label each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyh heteroarylalkyh cycloalkylalkyl, and heterocycly lalkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, -S-L3- X, -S(=O)-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or
unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2J -O-N(R*)2J -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCh, CF2C1, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=0)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
[261] Further provided herein are methods of detecting the presence of or level of Hepatitis
B (HBV) infection. The method comprises the steps of: a) performing an assay to detect the presence of or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject suspected of having HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that
a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg- second detection antibody complex is formed; and b) detecting HBV infection based on the presence or level of HBcAg and P-HBcAg in the at least one sample, wherein the first detectable label and second detectable label each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
wherein:
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, or heterocycly lalkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=0)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
[262] Section headings as used in this section and the entire disclosure herein are merely for organizational purposes and are not intended to be limiting.
DETAILED DESCRIPTION
1. Definitions
[263] Unless otherwise defined, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art. In case of conflict, the present document, including definitions, will control. Various embodiments of the methods and materials are described below, although methods and materials similar or equivalent to those described herein can be used in practice or testing of the present disclosure. All publications, patent applications, patents and other references mentioned herein are incorporated by reference in their entirety. The materials, methods, and examples disclosed herein are illustrative only and not intended to be limiting.
[264] For the recitation of numeric ranges herein, each intervening number there between with the same degree of precision is explicitly contemplated. For example, for the range of 6-9, the numbers 7 and 8 are contemplated in addition to 6 and 9, and for the range 6.0-7.0, the number 6.0, 6.1, 6.2, 6.3, 6.4, 6.5, 6.6, 6.7, 6.8, 6.9, and 7.0 are explicitly contemplated.
[265] The terms “comprise(s),” “include(s),” “having,” “has,” “can,” “contain(s),” and variants thereof, as used herein, are intended to be open-ended transitional phrases, terms, or words that do not preclude the possibility of additional acts or structures. The singular forms “a,” “an” and “the” include plural references unless the context clearly dictates otherwise. The present disclosure also contemplates other embodiments “comprising,” “consisting of’ and “consisting essentially of,” the embodiments or elements presented herein, whether explicitly set forth or not.
[266] "Analyte", "target analyte", "analyte of interest" as used interchangeably herein, refers to a substance, material or chemical constituent the presence, absence and/or amount of which is being analyzed in a sample (e.g., a capillary blood sample, a venous blood sample, a plasma sample, a serum sample) obtained from a subject. In some aspects, the analyte is a biomolecule. Non-limiting examples of biomolecules include macromolecules such as, proteins, lipids, and carbohydrates. In certain instances, the analyte may be hormones, antibodies, growth factors, cytokines, enzymes, receptors (e.g., neural, hormonal, nutrient, and cell surface receptors) or
their ligands, cancer markers (e.g., PSA, TNF-alpha), markers of myocardial infarction (e.g., troponin, creatine kinase, and the like), toxins, drugs (e.g., drugs of addiction), metabolic agents (e.g., including vitamins), and the like. Non-limiting examples of protein analytes include peptides, polypeptides, protein fragments, protein complexes, fusion proteins, recombinant proteins, phosphoproteins, glycoproteins, lipoproteins, or the like.
[267] In some aspects, the analyte may be a post-translationally modified protein (e.g., phosphorylated, methylated, glycosylated protein) and the first or the second binding member may be an antibody specific to a post-translational modification. A modified protein may be bound to a first binding member immobilized on a solid support where the first binding member binds to the modified protein but not the unmodified protein. In other embodiments, the first binding member may bind to both the unmodified and the modified protein, and the second binding member may be specific to the post-translationally modified protein.
[268] In yet other aspects, the analyte may be a cell, such as, a circulating tumor cell, pathogenic bacteria, viruses (including retroviruses, herpesviruses, adenoviruses, lentiviruses, Filoviruses (ebola), hepatitis viruses (e.g., A, B, C, D, and E); HPV, spores, etc. For example, in some aspects, the analyte can be phosphorylated Hepatitis core antigen, such as Hepatitis B core antigen (P-HBcAg). In other aspects, the analyte can be a non-phosphorylated Hepatitis core antigen, such as Hepatitis B core antigen (HBcAg).
[269] A non-limiting list of analytes that may be analyzed by the methods described herein include: Ap42 amyloid beta-protein, fetuin-A, tau, secretogranin II, prion protein, Alpha- synuclein, tau protein, neurofilament light chain, parkin, PTEN induced putative kinase 1, DJ-1, leucine-rich repeat kinase 2, mutated ATP13A2, Apo H, alanine aminotranseferase (ALT), aspartate aminotransferase (AST), ceruloplasmin, Peroxisome proliferator-activated receptor gamma coactivator- 1 alpha (PGC-la), transthyretin, Vitamin D-binding Protein, proapoptotic kinase R (PKR) and its phosphorylated PKR (pPKR), CXCL13, IL-12p40, CXCL13, IL-8, Dkk- 3 (semen), pl4 endocan fragment, Serum, ACE2, autoantibody to CD25, hTERT, CAI25 (MUC 16), VEGF, sIL-2, Osteopontin, Human epididymis protein 4 (HE4), Alpha-Fetoprotein, Albumin, albuminuria, microalbuminuria, neutrophil gelatinase-associated lipocalin (NGAL), hepatitis B surface antigen, homocysteine, interleukin 18 (IL- 18), Kidney Injury Molecule- 1 (KIM-1), Liver Fatty Acid Binding Protein (L-FABP), LMP1, BARF1, IL-8, carcinoembryonic antigen (CEA), BRAF, CCNI, EGRF, FGF19, FRS2, GREB1, and LZTS1, alpha-amylase,
carcinoembryonic antigen, CA 125, IL8, thioredoxin, beta-2 microglobulin levels— monitor activity of the virus, tumor necrosis factor-alpha receptors— monitor activity of the virus, CAI 5- 3, follicle-stimulating hormone (FSH), leutinizing hormone (LH), T-cell lymphoma invasion and metastasis 1 (TIAM1), N-cadherin, EC39, amphiregulin, dUTPase, secretory gelsolin (pGSN), PSA (prostate specific antigen), thymosin 1315, insulin, plasma C-peptide, glycosylated hemoglobin (HBAlc), C-Reactive Protein (CRP), Interleukin-6 (IL-6), ARHGDIB (Rho GDP- dissociation inhibitor 2), CFL1 (Cofilin-1), PFN1 (profilin-1), GSTP1 (Glutathione S-transferase P), S100A11 (Protein S100-A11), PRDX6 (Peroxiredoxin-6), HSPE1 (10 kDa heat shock protein, mitochondrial), LYZ (Lysozyme C precursor), GPI (Glucose-6-phosphate isomerase), HIST2H2AA (Histone H2A type 2- A), GAPDH (Glyceraldehyde-3 -phosphate dehydrogenase), HSPG2 (Basement membrane-specific heparan sulfate proteoglycan core protein precursor), LGALS3BP (Galectin-3 -binding protein precursor), CTSD (Cathepsin D precursor), APOE (Apolipoprotein E precursor), IQGAP1 (Ras GTPase-activating-like protein IQGAP1), CP (Ceruloplasmin precursor), and IGLC2 (IGLC1 protein), PCDGF/GP88, EGFR, HER2, MUC4, IGF-IR, p27(kipl), Akt, HER3, HER4, PTEN, PIK3CA, SHIP, Grb2, Gab2, PDK-1 (3- phosphoinositide dependent protein kinase- 1), TSC1, TSC2, mTOR, MIG-6 (ERBB receptor feedback inhibitor 1), S6K, src, KRAS, MEK mitogen-activated protein kinase 1, cMYC, TOPO II topoisomerase (DNA) II alpha 170 kDa, FRAP1, NRG1, ESRI, ESR2, PGR, CDKN1B, MAP2K1, NEDD4-1, FOXO3A, PPP1R1B, PXN, ELA2, CTNNB1, AR, EPHB2, KLF6, ANXA7, NKX3-1, PITX2, MKI67, PHLPP, adiponectin (ADIPOQ), fibrinogen alpha chain (FGA), leptin (LEP), advanced glycosylation end product-specific receptor (AGER aka RAGE), alpha-2-HS-glycoprotein (AHSG), angiogenin (ANG), CD 14 molecule (CD 14), ferritin (FTH1), insulin-like growth factor binding protein 1 (IGFBP1), interleukin 2 receptor, alpha (IL2RA), vascular cell adhesion molecule 1 (VCAM1) and Von Willebrand factor (VWF), myeloperoxidase (MPO), IL la, TNFa, perinuclear anti -neutrophil cytoplasmic antibody (p- ANCA), lactoferrin, calprotectin, Wilm's Tumor- 1 protein, Aquaporin-1, MLL3, AMBP, VDAC1, E. coli enterotoxins (heat-labile exotoxin, heat-stable enterotoxin), influenza HA antigen, tetanus toxin, diphtheria toxin, botulinum toxins, troponin (such as troponin I, troponin T and/or troponin C), Shiga toxin, Shiga-like toxin I, Shiga-like toxin II, Clostridium difficile toxins A andB, thyroid stimulating hormone (TSH), P1GF1, sFlt-1, GFAP, UCH-L1, NF-1, SARS-CoV-2 IgG antibody, SARS-CoV-2 IgM antibody, SARS-CoV-2 IgA antibody, etc. In
another aspect the analyte is thyroid stimulating hormone (TSH), estradiol, hepatitis B surface antigen, free prostate specific antigen (PSA), C-reactive protein (CRP), alanine aminotransferase (ALT), aspartate aminotransferase (AST), SARS-CoV-2 IgG antibody, homocysteine, troponin, or any combinations thereof.
[270] Exemplary aptamers (e.g., nucleic acid or protein or peptide aptamers) that may be analyzed by the methods described herein include: drugs of abuse (e.g. cocaine), protein biomarkers (including, but not limited to, Nucleolin, nuclear factor-kB essential modulator (NEMO), CD-30, protein tyrosine kinase 7 (PTK7), vascular endothelial growth factor (VEGF), MUC1 glycoform, immunoglobulin p Heavy Chains (IGHM), Immunoglobulin E, av03 integrin, a-thrombin, HIV gpl20, NF-KB, E2F transcription factor, HER3, Plasminogen activator inhibitor, Tenascin C, CXCL12/SDF-1, prostate specific membrane antigen (PSMA), gastric cancer cells, HGC-27); cells (including, but not limited to, non-small cell lung cancer (NSCLC), colorectal cancer cells, (DLD-1), H23 lung adenocarcinoma cells, Ramos cells, T-cell acute lymphoblastic leukemia (T-ALL) cells, CCRF-CEM, acute myeloid leukemia (AML) cells (HL60), small-cell lung cancer (SCLC) cells, NCIH69, human glioblastoma cells, U118-MG, PC-3 cells, HER-2-overexpressing human breast cancer cells, SK-BR-3, pancreatic cancer cell line (Mia-PaCa-2)); infectious agents (including, but not limited to, Mycobacterium tuberculosis, Staphylococcus aureus, Shigella dysenteriae, Escherichia coli O157:H7, Campylobacter jejuni, Listeria monocytogenes, Pseudomonas aeruginosa, Salmonella 08, Salmonella enteritidis), HBV core capsid protein, CDK2, E2F transcription factor, Thymidylate synthase, Ras, EB1, and Receptor for Advanced Glycated End products (RAGE).
[271] The term “alkenyl,” as used herein, refers to a straight or branched hydrocarbon chain containing from 2 to 16 carbon atoms and containing at least one carbon-carbon double bond. Representative examples of alkenyl include, but are not limited to, ethenyl, 2-propenyl, 2- methyl-2-propenyl, 3-butenyl, 4-pentenyl, 5-hexenyl, 2-heptenyl, 2-methyl-l -heptenyl, and 3- decenyl.
[272] As used herein, the term “alkenylene” refers a divalent straight chain or branched alkenyl linking group. Examples of “alkenylene” include ethen- 1,1 -diyl, ethen-l,2-diyl, propen- 1,3-diyl, 2-buten-l,4-diyl, 3-penten-l,5-diyl, 3-hexen-l,6-diyl, 3-hexen-l,5-diyl, and the like.
[273] The term “alkoxy,” as used herein, refers to an alkyl group, as defined herein, appended to the parent molecular moiety through an oxygen atom. Representative examples of
alkoxy include, but are not limited to, methoxy, ethoxy, propoxy, 2 -propoxy, butoxy and tertbutoxy.
[274] The term “alkyl,” as used herein, means a straight or branched saturated hydrocarbon chain containing from 1 to 16 carbon atoms (Ci -Ci6 alkyl), for example 1 to 14 carbon atoms (Ci -CM alkyl), 1 to 12 carbon atoms (Ci -C12 alkyl), 1 to 10 carbon atoms (Ci -C10 alkyl), 1 to 8 carbon atoms (Ci -Cs alkyl), 1 to 6 carbon atoms (Ci -Ce alkyl), or 1 to 4 carbon atoms (Ci -C4 alkyl). Representative examples of alkyl include, but are not limited to, methyl, ethyl, n-propyl, iso-propyl, n-butyl, sec-butyl, iso-butyl, tert-butyl (or t-butyl), n-pentyl, isopentyl, neopentyl, n- hexyl, 3 -methylhexyl, 2,2-dimethylpentyl, 2, 3 -dimethylpentyl, n-heptyl, n-octyl, n-nonyl, n- decyl, n-undecyl, and n-dodecyl.
[275] The term “alkylene,” as used herein, refers to a divalent group derived from a straight or branched chain hydrocarbon of 1 to 10 carbon atoms (C1-C10 alkylene), for example, of 1 to 6 carbon atoms (Ci-Ce alkylene). Representative examples of alkylene include, but are not limited to, — CH2— , — CH2CH2— , — CH(CH)— , — CH2CH2CH2— , — CH2CH(CH)— , — CH2CH2CH2CH2— , — CH2CH(CH3)CH2— , — CH2CH2CH(CH)— , — CH2CH2CH2CH2CH2— , — CH2CH(CH3)CH2CH2— , — CH(CH3)CH2CH2CH2— , — CH2CH2CH2CH2CH2CH2— , — CH2CH2CH(CH3)CH2CH2— , — CH2CH(CH3)CH2CH2CH2— , and — CH(CH3)CH2CH2CH2CH2— .
[276] The term “alkynyl,” as used herein, refers to a straight or branched hydrocarbon chain containing from 2 to 16 carbon atoms and containing at least one carbon-carbon triple bond. Representative examples of alkynyl include, but are not limited to, ethynyl, propynyl, and butynyl.
[277] As used herein, the term “alkynylene” refers a divalent straight chain or branched alkynyl linking group. Examples of “alkynylene groups” include propyn- 1,3 -diyl, 2-butyn-l,4- diyl, 3-pentyn-l,5-diyl, 3-hexyn-l,6-diyl, 3-hexyn-l,5-diyl, and the like.
[278] “Antibody” and “antibodies” as used herein refers to monoclonal antibodies, multispecific antibodies, human antibodies, humanized antibodies (fully or partially humanized), animal antibodies such as, but not limited to, a bird (for example, a duck or a goose), a shark, a whale, and a mammal, including a non-primate (for example, a cow, a pig, a camel, a llama, a horse, a goat, a rabbit, a sheep, a hamster, a guinea pig, a cat, a dog, a rat, a mouse, etc.) or a non-human primate (for example, a monkey, a chimpanzee, etc.), recombinant antibodies,
chimeric antibodies, single-chain Fvs (“scFv”), single chain antibodies, single domain antibodies, Fab fragments, F(ab') fragments, F(ab')2 fragments, disulfide-linked Fvs (“sdFv”), and anti-idiotypic (“anti-Id”) antibodies, dual-domain antibodies, dual variable domain (DVD) or triple variable domain (TVD) antibodies (dual-variable domain immunoglobulins and methods for making them are described in Wu, C., et al., Nature Biotechnology, 25(11):1290-1297 (2007) and PCT International Application WO 2001/058956, the contents of each of which are herein incorporated by reference), and functionally active epitope-binding fragments of any of the above. Antibodies include immunoglobulin molecules and immunologically active fragments of immunoglobulin molecules, namely, molecules that contain an analyte-binding site. Immunoglobulin molecules can be of any type (for example, IgG, IgE, IgM, IgD, IgA, and IgY), class (for example, IgGl, IgG2, IgG3, IgG4, IgAl, and IgA2), or subclass. For simplicity sake, an antibody against an analyte is frequently referred to herein as being either an “anti-analyte antibody” or merely an “analyte antibody” (e.g., an anti-UCH-Ll antibody or a UCH-L1 antibody).
[279] An antibody as used herein also refers non-Ig derived alternatives (so-called antibody ‘mimetics’) such as, e.g., aptamers (described previously above), as well as, DARPins, Affimers, Avimers, Knottins, Monobodies, and Affinity Clamps. As used herein, the term “aptamer” refers to a nucleic acid that has a specific binding affinity for a target analyte or molecule. It is recognized that affinity interactions are a matter of degree; however, in this context, the “specific binding affinity” of an aptamer for its analyte or target means that the aptamer binds to its analyte or target generally with a much higher degree of affinity than it binds to other components in a sample. An “aptamer” is a set of copies of one type or species of nucleic acid molecule that comprises a particular nucleotide sequence. An aptamer can include any suitable number of nucleotides, including any number of chemically modified nucleotides. “Aptamers” refers to more than one such set of molecules. Different aptamers can have either the same or different numbers of nucleotides. Aptamers can be DNA or RNA or chemically modified nucleic acids and can be single-stranded, double-stranded, or contain double-stranded regions, and can include higher ordered structures. An aptamer can also be a photoaptamer, where a photoreactive or chemically reactive functional group is included in the aptamer to allow it to be covalently linked to its corresponding analyte or target. In some aspects, an aptamer may include a detectable label. If an aptamer includes a detectable label, all copies of the aptamer need not have
the same detectable label. Moreover, if different aptamers each include a detectable label, these different aptamers can have either the same detectable label or a different detectable label.
[280] “Antibody fragment” as used herein refers to a portion of an intact antibody comprising the antigen-binding site or variable region. The portion does not include the constant heavy chain domains (i.e., CH2, CH3, or CH4, depending on the antibody isotype) of the Fc region of the intact antibody. Examples of antibody fragments include, but are not limited to, Fab fragments, Fab’ fragments, Fab’-SH fragments, F(ab’)2 fragments, Fd fragments, Fv fragments, diabodies, single-chain Fv (scFv) molecules, single-chain polypeptides containing only one light chain variable domain, single-chain polypeptides containing the three CDRs of the light-chain variable domain, single-chain polypeptides containing only one heavy chain variable region, and single-chain polypeptides containing the three CDRs of the heavy chain variable region.
[281] The “area under curve” or “AUC” refers to area under a ROC curve. AUC under a ROC curve is a measure of accuracy. An AUC of 1 represents a perfect test, whereas an AUC of 0.5 represents an insignificant test. A preferred AUC may be at least approximately 0.700, at least approximately 0.750, at least approximately 0.800, at least approximately 0.850, at least approximately 0.900, at least approximately 0.910, at least approximately 0.920, at least approximately 0.930, at least approximately 0.940, at least approximately 0.950, at least approximately 0.960, at least approximately 0.970, at least approximately 0.980, at least approximately 0.990, or at least approximately 0.995.
[282] The term “aryl,” as used herein, refers to a phenyl group, or a bicyclic or tricyclic aromatic fused ring system. Bicyclic fused ring systems are exemplified by a phenyl group appended to the parent molecular moiety and fused to a phenyl group. Tricyclic fused ring systems are exemplified by a phenyl group appended to the parent molecular moiety and fused to two other phenyl groups. Representative examples of bicyclic aryls include, but are not limited to, naphthyl. Representative examples of tricyclic aryls include, but are not limited to, anthracenyl and phenanthreneyl.
[283] The term “arylalkyl,” as used herein, refers to an aryl group, as defined herein, appended to the parent molecular moiety through an alkyl group, as defined herein. Representative examples of arylalkyl include, but are not limited to, phenylmethyl (i.e., benzyl) and phenylethyl.
[284] The term “arylene” refers to an aryl group having a valence of two.
[285] The term “aryloxy,” as used herein, means an aryl group, as defined herein, appended to the parent molecular moiety through an oxygen atom.
[286] “Bead” and “particle” are used herein interchangeably and refer to a substantially spherical solid support. One example of a bead or particle is a microparticle. Microparticles that can be used herein can be any type known in the art. For example, the bead or particle can be a magnetic bead or magnetic particle. Magnetic beads/particles may be ferromagnetic, ferrimagnetic, paramagnetic, superparamagnetic or ferrofluidic. Exemplary ferromagnetic materials include Fe, Co, Ni, Gd, Dy, CrO?, MnAs, MnBi, EuO, and NiO/Fe. Examples of ferrimagnetic materials include NiFe2O4, CoFe2O4, Fe3O4 (or FeO Fe2O3). Beads can have a solid core portion that is magnetic and is surrounded by one or more non-magnetic layers. Alternately, the magnetic portion can be a layer around a non-magnetic core. The microparticles can be of any size that would work in the methods described herein, e.g., from about 0.75 to about 5 nm, or from about 1 to about 5 nm, or from about 1 to about 3 nm.
[287] “Binding protein” is used herein to refer to a monomeric or multimeric protein that binds to and forms a complex with a binding partner, such as, for example, a polypeptide, an antigen, a chemical compound or other molecule, or a substrate of any kind. A binding protein specifically binds a binding partner. Binding proteins include antibodies, as well as antigenbinding fragments thereof and other various forms and derivatives thereof as are known in the art and described herein below, and other molecules comprising one or more antigen-binding domains that bind to an antigen molecule or a particular site (epitope) on the antigen molecule. Accordingly, a binding protein includes, but is not limited to, an antibody a tetrameric immunoglobulin, an IgG molecule, an IgGl molecule, a monoclonal antibody, a chimeric antibody, a CDR-grafted antibody, a humanized antibody, an affinity matured antibody, and fragments of any such antibodies that retain the ability to bind to an antigen.
[288] “Bispecific antibody” is used herein to refer to a full-length antibody that is generated by quadroma technology (also referred to as hybrid-hybridoma technology; see Milstein et al., Nature, 305(5934): 537-540 (1983)), by chemical conjugation of two different monoclonal antibodies (see, Staerz et al., Nature, 314(6012): 628-631 (1985)), or by knob-into-hole or similar approaches, which introduce mutations in the Fc region (see Holliger et al., Proc. Natl. Acad. Sci. USA, 90(14): 6444-6448 (1993)), resulting in multiple different immunoglobulin
species of which only one is the functional bispecific antibody. A bispecific antibody binds one antigen (or epitope) on one of its two binding arms (one pair of HC/LC), and binds a different antigen (or epitope) on its second arm (a different pair of HC/LC). By this definition, a bispecific antibody has two distinct antigen-binding arms (in both specificity and CDR sequences), and is monovalent for each antigen to which it binds to.
[289] As used herein, the term “calibration curve” refers to a curve that is used to determine the concentration of an analyte of interest in a biological sample, as well as to calculate the limit of detection and limit of quantitation of an assay (such as, for example, an immunoassay, clinical chemistry assay, etc.). A calibration curve is created from the working range of the technique (e.g., instrument) being used to a set of standard samples (e.g., at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least night, or at least ten samples) at a range of concentrations. Calibration curves for use in the present disclosure can be generated using routine techniques known in the art.
[290] As used herein, the term “capture reagent”, refers to a specific binding partner comprising (i) an antibody or antibody fragment thereof that binds to an antigen (e.g., polypeptide) of interest (e.g., analyte of interest) in a biological sample; or (ii) an antigen (e.g., polypeptide or fragment thereof) that binds to an antibody or antibody fragment thereof of interest (e.g., analyte of interest) in a biological sample. In some aspects, the capture reagent may be bound on to a solid support, such as a bead or particle. In other aspects, the capture reagent is not bound to a solid support.
[291] “Communicated” or “communicating” as used herein refers to the conveying, transmitting and/or reporting of an item of information. In some aspects, the information that is communicated is an item of information obtained by performing an assay (e.g., a competitive immunoassay and/or a sandwich immunoassay), such as, the amount or level of an analyte of interest in a sample (e.g., a result). The amount or level of the analyte of interest in the sample may be determining from a competitive immunoassay detectable signal and/or a sandwich immunoassay detectable signal. The information obtained by performing an assay can be communicated by a computer, in a document and/or spreadsheet, on a mobile device (e.g., a smart phone), on a website, in an e-mail, or any combination thereof. In some other aspects, information is communicated on or from an instrument or device. In other aspects, the information is communicated by being displayed, such as on an instrument or device.
[ 292 ] As used herein, the term “complementary” refers to the capacity for precise pairing between two nucleotides, i.e., if a nucleotide at a given position of a nucleic acid is capable of hydrogen bonding with a nucleotide of another nucleic acid, then the two nucleic acids are considered to be complementary to one another at that position. Complementarity between two single-stranded nucleic acid molecules may be “partial,” in which only some of the nucleotides bind, or it may be complete when total complementarity exists between the single-stranded molecules. The degree of complementarity between nucleic acid strands has significant effects on the efficiency and strength of hybridization between nucleic acid strands.
[293] “CDR” is used herein to refer to the “complementarity determining region” within an antibody variable sequence. There are three CDRs in each of the variable regions of the heavy chain and the light chain. Proceeding from the N-terminus of a heavy or light chain, these regions are denoted “CDR1”, “CDR2”, and “CDR3”, for each of the variable regions. The term “CDR set” as used herein refers to a group of three CDRs that occur in a single variable region that binds the antigen. An antigen-binding site, therefore, may include six CDRs, comprising the CDR set from each of a heavy and a light chain variable region. A polypeptide comprising a single CDR, (e.g., a CDR1, CDR2, or CDR3) may be referred to as a “molecular recognition unit.” Crystallographic analyses of antigen-antibody complexes have demonstrated that the amino acid residues of CDRs form extensive contact with bound antigen, wherein the most extensive antigen contact is with the heavy chain CDR3. Thus, the molecular recognition units may be primarily responsible for the specificity of an antigen-binding site. In general, the CDR residues are directly and most substantially involved in influencing antigen binding.
[294] The exact boundaries of these CDRs have been defined differently according to different systems. The system described by Kabat (Kabat et al., Sequences of Proteins of Immunological Interest (National Institutes of Health, Bethesda, Md. (1987) and (1991)) not only provides an unambiguous residue numbering system applicable to any variable region of an antibody, but also provides precise residue boundaries defining the three CDRs. These CDRs may be referred to as “Kabat CDRs”. Chothia and coworkers (Chothia and Lesk, J. Mol. Biol., 196: 901-917 (1987); and Chothia et al., Nature, 342: 877-883 (1989)) found that certain subportions within Kabat CDRs adopt nearly identical peptide backbone conformations, despite having great diversity at the level of amino acid sequence. These sub-portions were designated as “LI”, “L2”, and “L3”, or “Hl”, “H2”, and “H3”, where the “L” and the “H” designate the
light chain and the heavy chain regions, respectively. These regions may be referred to as “Chothia CDRs”, which have boundaries that overlap with Kabat CDRs. Other boundaries defining CDRs overlapping with the Kabat CDRs have been described by Padlan, FASEB J., 9: 133-139 (1995), and MacCallum, J. Mol. Biol., 262(5): 732-745 (1996). Still other CDR boundary definitions may not strictly follow one of the herein systems, but will nonetheless overlap with the Kabat CDRs, although they may be shortened or lengthened in light of prediction or experimental findings that particular residues or groups of residues or even entire CDRs do not significantly impact antigen binding. The methods used herein may utilize CDRs defined according to any of these systems, although certain embodiments use Kabat- or Chothia- defined CDRs.
[295] “Component,” “components,” or “at least one component,” refer generally to a capture reagent, a detection reagent or conjugate a calibrator, a control, a sensitivity panel, a container, a buffer, a diluent, a salt, an enzyme, a co-factor for an enzyme, a detection reagent, a pretreatment reagent/ solution, a substrate (e.g., as a solution), a stop solution, and the like that can be included in a kit for assay of a test sample, such as a patient urine, whole blood, serum or plasma sample, in accordance with the methods described herein and other methods known in the art. Some components can be in solution or lyophilized for reconstitution for use in an immunoassay.
[296] “Controls” as used herein generally refers to a reagent whose purpose is to evaluate the performance of a measurement system in order to assure that it continues to produce results within permissible boundaries (e.g., boundaries ranging from measures appropriate for a research use assay on one end to analytic boundaries established by quality specifications for a commercial assay on the other end). To accomplish this, a control should be indicative of patient results and optionally should somehow assess the impact of error on the measurement (e.g., error due to reagent stability, calibrator variability, instrument variability, and the like).
[297] As used herein, “CPSP” corresponds to carboxypropylsulfopropyl-acridinium and has the IUPAC name 4-(10-(3-sulfopropyl)-N-tosyl-10X4-acridine-9-carboxamido)butanoic acid. “CPSP2” has the IUPAC name 4-(10-(3-sulfopropyl)-2-((3-sulfopropyl)thio)-N-tosyl-10X4- acridine-9-carboxamido)butanoic acid. “CPSP3” has the IUPAC name 4-(10-(3-sulfopropyl)- 2,7-bis((3-sulfopropyl)thio)-N-tosyl-10X4-acridine-9-carboxamido)butanoic acid. “SP3” has the IUPAC name 3,3'-((9-oxo- 10-(3-sulfopropyl)-9,l 0-dihydroacridine-2,7- diyl)bis(sulfanediyl))bis(propane-l -sulfonic acid). For ease of reference, it is understood that
“CPSP” and “CPSP-acridinium” can be used interchangeably, that “CPSP2” and “CPSP2- acridinium” can be used interchangeably, and that “CPSP3” and “CPSP3-acridinium” can be used interchangeably.
[298] The term “cycloalkyl,” as used herein, refers to a saturated carbocyclic ring system containing three to ten carbon atoms and zero heteroatoms. The cycloalkyl may be monocyclic, bicyclic, bridged, fused, or spirocyclic. Representative examples of cycloalkyl include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl, cyclononyl, cyclodecyl, adamantyl, bicyclo [l.l.l]pentanyl, bicyclo[2.2.1]heptanyl, bicyclo[3.2. l]octanyl, and bicyclo[5.2.0]nonanyl.
[299] The term “cycloalkenyl,” as used herein, means a non-aromatic monocyclic or multicyclic carbocyclic ring system containing at least one carbon-carbon double bond and preferably having from 5-10 carbon atoms per ring. Exemplary monocyclic cycloalkenyl rings include, but are not limited to, cyclopentenyl, cyclohexenyl, cycloheptenyl, and bicyclo[2.2. l]heptenyl.
[300] As used herein, the term “cycloalkylalkyl” refers to a cycloalkyl group, as defined herein, appended to the parent molecular moiety through an alkylene group, as defined herein. Representative examples of cycloalkylalkyl include, but are not limited to, cyclohexylmethyl.
[301] The term “cycloalkylene” means a divalent cycloalkyl group, such as 1,2- cyclohexylene, 1,3 -cyclohexylene, or 1,4-cyclohexylene.
[302] “Derivative” of an antibody as used herein may refer to an antibody having one or more modifications to its amino acid sequence when compared to a genuine or parent antibody and exhibits a modified domain structure. The derivative may still be able to adopt the typical domain configuration found in native antibodies, as well as an amino acid sequence, which is able to bind to targets (antigens) with specificity. Typical examples of antibody derivatives are antibodies coupled to other polypeptides, rearranged antibody domains, or fragments of antibodies. The derivative may also comprise at least one further compound, e.g., a protein domain, said protein domain being linked by covalent or non-covalent bonds. The linkage can be based on genetic fusion according to the methods known in the art. The additional domain present in the fusion protein comprising the antibody may be linked by a flexible linker, advantageously a peptide linker, wherein said peptide linker comprises plural, hydrophilic, peptide-bonded amino acids of a length sufficient to span the distance between the C-terminal
end of the further protein domain and the N-terminal end of the antibody or vice versa. The antibody may be linked to an effector molecule having a conformation suitable for biological activity or selective binding to a solid support, a biologically active substance (e.g., a cytokine or growth hormone), a chemical agent, a peptide, a protein, or a drug, for example.
[303] As used herein, the term “detection reagent”, or “conjugate” as used interchangeably herein, refers to a specific binding partner that comprises (i) an antibody or antibody fragment thereof that is conjugated to one or more detectable labels; or (ii) an antigen (e.g., polypeptide or fragment thereof) that is conjugated to one or more detectable labels.
[304] “Dynamic range” as used herein refers to range over which an assay readout is proportional to the amount of target molecule or analyte of interest in the sample being analyzed.
[305] “Epitope,” or “epitopes,” or “epitopes of interest” refer to a site(s) on any molecule that is recognized and can bind to a complementary site(s) on its specific binding partner. The molecule and specific binding partner are part of a specific binding pair. For example, an epitope can be on a polypeptide, a protein, a hapten, a carbohydrate antigen (such as, but not limited to, glycolipids, glycoproteins or lipopolysaccharides), or a polysaccharide. Its specific binding partner can be, but is not limited to, an antibody.
[306] The term “halogen” or “halo,” as used herein, means F, Cl, Br, or I.
[307] The term “haloalkyl,” as used herein, means an alkyl group, as defined herein, in which one or more hydrogen atoms are replaced by a halogen. For example, one, two, three, four, five, six, seven or eight hydrogen atoms can be replaced by a halogen, or all hydrogen atoms can be replaced by a halogen. Representative examples of haloalkyl include, but are not limited to, fluoromethyl, difluoromethyl, trifluoromethyl, chloromethyl, dichloromethyl, trichloromethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, perfluoroethyl, 2-fluoro-2- methylpropyl, and 3,3,3-trifluoropropyl.
[308] The term “haloalkoxy,” as used herein, means at least one haloalkyl group, as defined herein, is appended to the parent molecular moiety through an oxygen atom. Representative examples of haloalkoxy include, but are not limited to, trifluoromethoxy.
[309] The term “heteroalkyl,” as used herein, refers to an alkyl group, as defined herein, in which at least one carbon atom has been replaced with a heteroatom such as N, O, P, or S. Representative examples of heteroalkyls include, but are not limited to, alkyl ethers, secondary and tertiary alkyl amines, amides, and alkyl sulfides.
[310] The term “heteroalkylene,” as used herein, refers to an alkylene group, as defined herein, in which at least one carbon atom has been replaced with a heteroatom such as N, O, P, or S. Representative examples of heteroalkylene groups include polyethylene oxide and polypropylene oxide chains, polyethyleneimine groups, and the like.
[311] The term “heteroaryl,” as used herein, refers to an aromatic monocyclic ring or an aromatic bicyclic ring system or an aromatic tricyclic ring system. The aromatic monocyclic rings are five or six membered rings containing at least one heteroatom independently selected from the group consisting of N, O, and S (e.g., 1, 2, 3, or 4 heteroatoms independently selected from O, S, and N). The five-membered aromatic monocyclic rings have two double bonds and the six membered six membered aromatic monocyclic rings have three double bonds. The bicyclic heteroaryl groups are exemplified by a monocyclic heteroaryl ring appended fused to a monocyclic aryl group, as defined herein, or a monocyclic heteroaryl group, as defined herein. The tricyclic heteroaryl groups are exemplified by a monocyclic heteroaryl ring fused to two rings independently selected from a monocyclic aryl group, as defined herein or a monocyclic heteroaryl group as defined herein. Representative examples of monocyclic heteroaryl include, but are not limited to, pyridinyl (including pyridin-2-yl, pyridin-3-yl, pyridin-4-yl), pyrimidinyl, pyrazinyl, pyridazinyl, pyrrolyl, benzopyrazolyl, 1,2,3-triazolyl, 1,3,4-thiadiazolyl, 1,2,4- thiadiazolyl, 1,3,4-oxadiazolyl, 1,2,4-oxadiazolyl, imidazolyl, thiazolyl, isothiazolyl, thienyl, furanyl, oxazolyl, isoxazolyl, 1,2,4-triazinyl, and 1,3,5-triazinyl. Representative examples of bicyclic heteroaryl include, but are not limited to, benzimidazolyl, benzodioxolyl, benzofuranyl, benzooxadiazolyl, benzopyrazolyl, benzothiazolyl, benzothienyl, benzotriazolyl, benzoxadiazolyl, benzoxazolyl, chromenyl, imidazopyridine, imidazothiazolyl, indazolyl, indolyl, isobenzofuranyl, isoindolyl, isoquinolinyl, naphthyridinyl, purinyl, pyridoimidazolyl, quinazolinyl, quinolinyl, quinoxalinyl, thiazolopyridinyl, thiazolopyrimidinyl, thienopyrrolyl, thienothiophenyl, and thienothienyl. Representative examples of tricyclic heteroaryl include, but are not limited to, dibenzofuranyl and dibenzothienyl. The monocyclic, bicyclic, and tricyclic heteroaryls are connected to the parent molecular moiety through any carbon atom or any nitrogen atom contained within the rings.
[312] The term “heteroarylalkyl,” as used herein, refers to a heteroaryl group, as defined herein, appended to the parent molecular moiety through an alkylene group, as defined herein. Representative examples of heteroarylalkyl include, but are not limited to, fur-3 -ylmethyl, 1H-
imidazol-2-ylmethyl, lH-imidazol-4-ylmethyl, l-(pyridin-4-yl)ethyl, pyridin-3-ylmethyl, 6- chloropyridin-3-ylmethyl, pyridin-4-ylmethyl, (6-(trifluoromethyl)pyridin-3-yl)methyl, (6- (cyano)pyridin-3-yl)methyl, (2-(cyano)pyridin-4-yl)methyl, (5-(cyano)pyridin-2-yl)methyl, (2- (chloro)pyridin-4-yl)methyl, pyrimidin-5-ylmethyl, 2-(pyrimidin-2-yl)propyl, thien-2-ylmethyl, and thien-3-ylmethyl.
[313] The term “heteroarylene” refers to an heteroaryl group having a valence of two.
[314] The term “heterocycle” or “heterocyclic” as used herein, means a monocyclic heterocycle, a bicyclic heterocycle, or a tricyclic heterocycle. The monocyclic heterocycle is a three-, four-, five-, six-, seven-, or eight-membered ring containing at least one heteroatom independently selected from the group consisting of O, N, and S. The three- or four-membered ring contains zero or one double bond, and one heteroatom selected from the group consisting of O, N, and S. The five-membered ring contains zero or one double bond and one, two or three heteroatoms selected from the group consisting of O, N and S. The six-membered ring contains zero, one or two double bonds and one, two, or three heteroatoms selected from the group consisting of O, N, and S. The seven- and eight-membered rings contains zero, one, two, or three double bonds and one, two, or three heteroatoms selected from the group consisting of O, N, and S. Representative examples of monocyclic heterocycles include, but are not limited to, azetidinyl, azepanyl, aziridinyl, diazepanyl, 1,3-dioxanyl, 1,3-dioxolanyl, 1,3-dithiolanyl, 1,3- dithianyl, imidazolinyl, imidazolidinyl, isothiazolinyl, isothiazolidinyl, isoxazolinyl, isoxazolidinyl, morpholinyl, oxadiazolinyl, oxadiazolidinyl, oxazolinyl, oxazolidinyl, oxetanyl, piperazinyl, piperidinyl, pyranyl, pyrazolinyl, pyrazolidinyl, pyrrolinyl, pyrrolidinyl, tetrahydrofuranyl, tetrahydropyranyl, tetrahydropyridinyl, tetrahydrothienyl, thiadiazolinyl, thiadiazolidinyl, 1,2-thiazinanyl, 1,3-thiazinanyl, thiazolinyl, thiazolidinyl, thiomorpholinyl, 1,1- dioxidothiomorpholinyl (thiomorpholine sulfone), thiopyranyl, and trithianyl. The bicyclic heterocycle is a monocyclic heterocycle fused to a phenyl group, or a monocyclic heterocycle fused to a monocyclic cycloalkyl, or a monocyclic heterocycle fused to a monocyclic cycloalkenyl, or a monocyclic heterocycle fused to a monocyclic heterocycle, or a spiro heterocycle group, or a bridged monocyclic heterocycle ring system in which two non-adjacent atoms of the ring are linked by an alkylene bridge of 1, 2, 3, or 4 carbon atoms, or an alkenylene bridge of two, three, or four carbon atoms. Representative examples of bicyclic heterocycles include, but are not limited to, benzopyranyl, benzothiopyranyl, chromanyl, 2,3-
dihydrobenzofuranyl, 2,3 -dihydrobenzothienyl, 2,3-dihydroisoquinoline, 2-azaspiro[3.3]heptan- 2-yl, azabicyclo[2.2.1]heptyl (including 2-azabicyclo[2.2.1]hept-2-yl), 2,3 -dihydro- lH-indolyl, isoindolinyl, octahydrocyclopenta[c]pyrrolyl, octahydropyrrolopyridinyl, and tetrahydroisoquinolinyl. Tricyclic heterocycles are exemplified by a bicyclic heterocycle fused to a phenyl group, or a bicyclic heterocycle fused to a monocyclic cycloalkyl, or a bicyclic heterocycle fused to a monocyclic cycloalkenyl, or a bicyclic heterocycle fused to a monocyclic heterocycle, or a bicyclic heterocycle in which two non-adjacent atoms of the bicyclic ring are linked by an alkylene bridge of 1, 2, 3, or 4 carbon atoms, or an alkenylene bridge of two, three, or four carbon atoms. Examples of tricyclic heterocycles include, but are not limited to, octahydro-2, 5-epoxypentalene, hexahydro-2H-2,5-methanocyclopenta[b]furan, hexahydro-lH- l,4-methanocyclopenta[c]fiiran, aza-adamantane (l-azatricyclo[3.3.1.13,7]decane), and oxaadamantane (2-oxatricyclo[3.3.1.13,7]decane). The monocyclic, bicyclic, and tricyclic heterocycles are connected to the parent molecular moiety through any carbon atom or any nitrogen atom contained within the rings.
[315] The term “heterocycloalkylene” means a divalent heterocycloalkyl group.
[316] As used herein, the term “heterocyclylalkyl” refers to a heterocyclyl group, as defined herein, appended to the parent molecular moiety through an alkylene group, as defined herein. Representative examples of heterocyclylalkyl include, but are not limited to, piperidin-4- ylmethyl, piperazin- 1-ylmethyl, 3 -methyl- 1-pyrrolidin-l-ylbutyl, ( lR)-3 -methyl- 1-pyrrolidin-l- ylbutyl, (1 S)-3 -methyl- 1-pyrrolidin-l-ylbutyl, and 3 -morpholinopropyl.
[317] The term “hydroxy,” as used herein, means an — OH group.
[318] The term “hydroxyalkyl,” as used herein, refers to an alkyl group, as defined herein, substituted with at least one hydroxy group. Representative examples of hydroxyalkyl include, but are not limited to, hydroxymethyl, 2-hydroxyethyl, 3 -hydroxypropyl, 2,3-dihydroxypropyl, 2,3-dihydroxypentyl, 4-hydroxybutyl, 2-ethyl-4-hydroxyheptyl, 3,4-dihydroxybutyl, and 5- hydroxypentyl.
[319] In some instances, the number of carbon atoms in a group (e.g., alkyl, alkoxy, or cycloalkyl) is indicated by the prefix “Cx-Cy-”, wherein x is the minimum and y is the maximum number of carbon atoms in the group. Thus, for example, “Ci-Cj-alkyl” refers to an alkyl group containing from 1 to 3 carbon atoms.
[320] “Identical” or “identity” as used herein in the context of two or more polypeptide or polynucleotide sequences, may mean that the sequences have a specified percentage of residues that are the same over a specified region. The percentage may be calculated by optimally aligning the two sequences, comparing the two sequences over the specified region, determining the number of positions at which the identical residue occurs in both sequences to yield the number of matched positions, dividing the number of matched positions by the total number of positions in the specified region, and multiplying the result by 100 to yield the percentage of sequence identity. In cases where the two sequences are of different lengths or the alignment produces one or more staggered ends and the specified region of comparison includes only a single sequence, the residues of single sequence are included in the denominator but not the numerator of the calculation.
[321] The term “label,” as used herein, refers to any atom or molecule that can be used to provide a detectable and/or quantifiable signal. In some cases, the label can be attached, directly or indirectly, to an antigen or antibody. Suitable labels that can be attached to an antigen or antibody include, but are not limited to, radioisotopes, fluorophores, chromophores, mass labels, electron dense particles, magnetic particles, spin labels, molecules that emit chemiluminescence, electrochemically active molecules, enzymes, cofactors, and enzyme substrates.
[322] “Negative predictive value” or “NPV” as used interchangeably herein refers to the probability that a subject has a negative outcome given that they have a negative test result.
[323] “Non-point-of-care device” refers to a device that is not a point-of-care device or a single use device. A “point-of-care” device refers to a device used to provide medical diagnostic testing at or near the point-of-care (namely, outside of a laboratory), at the time and place of patient care (such as in a hospital, physician’s office, urgent or other medical care facility, a patient’s home, a nursing home and/or a long-term care and/or hospice facility). A point-of-care instrument does not perform an assay on more than one clinical sample simultaneously.
Examples of point-of-care devices include those produced by Abbott Laboratories (Abbott Park, IL) (e.g., i-STAT and i-STAT Alinity, Universal Biosensors (Rowville, Australia) (see U.S. Patent Publication No. 2006/0134713), Axis-Shield PoC AS (Oslo, Norway) and Clinical Lab Products (Los Angeles, USA). In some embodiments, the point-of-care device is a single-use device. The term “single-use device” or “single-use instrument” refers to a clinical diagnostic instrument that processes and performs a clinical diagnostic assay on a unit use basis (such as a
single-use cartridge) for a single patient sample. A non-point-of-care device refers to any device that does not meet any of the above limitations of a point-of-care or a single use device. In some embodiments, the non-point-of-care device may be a relatively large instrument, such as a tabletop instrument. Accordingly, in some embodiments the non-point-of-care device is not a handheld instrument. In some embodiments, the non-point-of-care device is capable of performing an assay on more than one clinical sample simultaneously. Suitable non-point-of- care devices include, for example, the ARCHITECT® or Alinity platforms produced by Abbott Laboratories.
[324] The term “nucleic acid” refers to a nucleotide polymer, and unless otherwise limited, includes known analogs of natural nucleotides that can function in a similar manner (e.g., hybridize) to naturally occurring nucleotides.
[325] The term nucleic acid includes any form of DNA or RNA, including, for example, genomic DNA; complementary DNA (cDNA), which is a DNA representation of mRNA, usually obtained by reverse transcription of messenger RNA (mRNA) or by amplification; DNA molecules produced synthetically or by amplification; and mRNA.
[326] The term nucleic acid encompasses double- or triple-stranded nucleic acids, as well as single-stranded molecules. In double- or triple-stranded nucleic acids, the nucleic acid strands need not be coextensive (i.e., a double-stranded nucleic acid need not be double-stranded along the entire length of both strands).
[327] The term nucleic acid also encompasses any chemical modification thereof, such as by methylation and/or by capping. Nucleic acid modifications can include addition of chemical groups that incorporate additional charge, polarizability, hydrogen bonding, electrostatic interaction, and functionality to the individual nucleic acid bases or to the nucleic acid as a whole. Such modifications may include base modifications such as 2'-position sugar modifications, 5-position pyrimidine modifications, 8-position purine modifications, modifications at cytosine exocyclic amines, substitutions of 5 -bromo-uracil, backbone modifications, unusual base pairing combinations such as the isobases isocytidine and isoguanidine, and the like.
[328] “Positive predictive value” or “PPV” as used interchangeably herein refers to the probability that a subject has a positive outcome given that they have a positive test result.
[329] “Quality control reagents” in the context of immunoassays (e.g., competitive immunoassay and sandwich immunoassay) and kits described herein, include, but are not limited to, calibrators, controls, and sensitivity panels. A “calibrator” or “standard” typically is used (e.g., one or more, such as a plurality) in order to establish calibration (standard) curves for interpolation of the concentration of an analyte, such as an antibody or an analyte. Alternatively, a single calibrator, which is near a reference level or control level (e.g., “low”, “medium”, or “high” levels), can be used. Multiple calibrators (i.e., more than one calibrator or a varying amount of calibrator(s)) can be used in conjunction to comprise a “sensitivity panel.”
[330] The terms “R” and “S” used herein are configurations as defined in IUPAC 1974 Recommendations for Section E, Fundamental Stereochemistry, in Pure Appl. C'hem., 1976, 45: 13-30.
[331] As used herein, a “reaction vessel” refers to a holder or receiver, such as a container, receptacle, tube, and/or cartridge, in or upon which one or more assays is performed.
[332] A “reagent” refers broadly to any agent used in a reaction, other than the analyte of interest. Illustrative reagents for immunoassay include, for example, antigens or antibodies specific for an analyte of interest, detection (e.g., labeled) antigens or antibodies, controls, diluents, buffers, and the like.
[333] A “receiver operating characteristic” curve or “ROC” curve refers to a graphical plot that illustrates the performance of a binary classifier system as its discrimination threshold is varied. For example, a ROC curve can be a plot of the true positive rate against the false positive rate for the different possible cutoff points of a diagnostic test. It is created by plotting the fraction of true positives out of the positives (TPR = true positive rate) vs. the fraction of false positives out of the negatives (FPR = false positive rate), at various threshold settings. TPR is also known as sensitivity, and FPR is one minus the specificity or true negative rate. The ROC curve demonstrates the tradeoff between sensitivity and specificity (any increase in sensitivity will be accompanied by a decrease in specificity); the closer the curve follows the left-hand border and then the top border of the ROC space, the more accurate the test; the closer the curve comes to the 45 -degree diagonal of the ROC space, the less accurate the test; the slope of the tangent line at a cutoff point gives the likelihood ratio (LR) for that value of the test; and the area under the curve is a measure of test accuracy.
[334] “Sensitivity” of an assay as used herein refers to the proportion of subjects for whom the outcome is positive that are correctly identified as positive (e.g., correctly identifying those subjects who do have a disease or medical condition for which they are being tested).
[335] As used herein the term “single molecule detection” refers to the detection and/or measurement of a single molecule of an analyte in a test sample at very low levels of concentration (such as pg/mL or femtogram/mL levels). A number of different single molecule analyzers or devices are known in the art and include nanopore and nanowell devices. Examples of nanopore devices are described in International Patent Publication No. WO 2016/161402, which is hereby incorporated by reference in its entirety. Examples of nanowell device are described in International Patent Publication No. WO 2016/161400, which is hereby incorporated by reference in its entirety.
[336] The term “substituent” refers to a group substituted on an atom of the indicated group.
[337] When a group or moiety can be substituted, the term “substituted” indicates that one or more (e.g., 1, 2, 3, 4, 5, or 6; in some embodiments 1, 2, or 3; and in other embodiments 1 or 2) hydrogens on the group indicated in the expression using “substituted” can be replaced with a selection of recited indicated groups or with a suitable group known to those of skill in the art (e.g., one or more of the groups recited below). Substituent groups include, but are not limited to, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, and acyl.
[338] As used herein, the term “test sample” or “sample” generally refers to a material (e.g., a biological material) being tested for and/or suspected of containing an analyte of interest, such as a marker described herein. The test sample may be derived from any biological source, such as, a physiological fluid, including, but not limited to, whole blood, serum, plasma, interstitial fluid, saliva, ocular lens fluid, cerebral spinal fluid, sweat, urine, milk, ascites fluid, mucous, nasal fluid, sputum, synovial fluid, peritoneal fluid, vaginal fluid, menses, amniotic fluid, semen and so forth. In some embodiments, the sample is a whole blood sample. In some embodiments, the sample is a plasma sample. In yet other embodiments, the sample is a serum sample. The
test sample may be used directly as obtained from the biological source or following a pretreatment to modify the character of the sample. For example, such pretreatment may include preparing plasma from blood, diluting viscous fluids and so forth. Methods of pretreatment may also involve filtration, precipitation, dilution, distillation, mixing, concentration, inactivation of interfering components, the addition of reagents, lysing, etc. Moreover, it may also be beneficial to modify a solid test sample to form a liquid medium or to release the analyte.
[339] “Solid phase” or “solid support” as used interchangeably herein, refers to any material that can be used to attach and/or attract and immobilize (1) one or more capture reagents or capture specific binding partners, or (2) one or more detection reagents or detection specific binding partners. The solid phase can be chosen for its intrinsic ability to attract and immobilize a capture reagent. Alternatively, the solid phase can have affixed thereto a linking agent that has the ability to attract and immobilize the (1) capture reagent or capture specific binding partner, or (2) detection reagent or detection specific binding partner. For example, the linking agent can include a charged substance that is oppositely charged with respect to the capture reagent (e.g., capture specific binding partner) or detection reagent (e.g., detection specific binding partner) itself or to a charged substance conjugated to the (1) capture reagent or capture specific binding partner or (2) detection reagent or detection specific binding partner. In general, the linking agent can be any binding partner (preferably specific) that is immobilized on (attached to) the solid phase and that has the ability to immobilize the (1) capture reagent or capture specific binding partner, or (2) detection reagent or detection specific binding partner through a binding reaction. The linking agent enables the indirect binding of the capture reagent to a solid phase material before the performance of the assay or during the performance of the assay. For examples, the solid phase can be plastic, derivatized plastic, magnetic, or non-magnetic metal, glass or silicon, including, for example, a test tube, microtiter well, sheet, bead, microparticle, chip, and other configurations known to those of ordinary skill in the art.
[340] “Specific binding” or “specifically binding” as used herein may refer to the interaction of an antibody or antibody fragment thereof, a protein, or a peptide (e.g., an antigen) with a second chemical species, wherein the interaction is dependent upon the presence of a particular structure (e.g., an antigenic determinant or epitope) on the chemical species; for example, an antibody recognizes and binds to a specific protein structure rather than to proteins generally. If an antibody is specific for epitope “A”, the presence of a molecule containing epitope A (or free,
unlabeled A), in a reaction containing labeled “A” and the antibody, will reduce the amount of labeled A bound to the antibody.
[341] “Specific binding partner” is a member of a specific binding pair. A specific binding pair comprises two different molecules, which specifically bind to each other through chemical or physical means. Therefore, in addition to antigen and antibody specific binding pairs of common immunoassays, other specific binding pairs can include biotin and avidin (or streptavidin), carbohydrates and lectins, complementary nucleotide sequences, effector and receptor molecules, cofactors and enzymes, enzymes and enzyme inhibitors, and the like.
Furthermore, specific binding pairs can include members that are analogs of the original specific binding members, for example, an analyte-analog. Immunoreactive specific binding members include antigens, antigen fragments, and antibodies, including monoclonal and polyclonal antibodies as well as complexes and fragments thereof, whether isolated or recombinantly produced.
[342] “Specificity” of an assay as used herein refers to the proportion of subjects for whom the outcome is negative that are correctly identified as negative (e.g., correctly identifying those subjects who do not have a disease or medical condition for which they are being tested).
[343] “Substantially identical,” as used herein may mean that a first and second sequence are at least from about 50% to about 99% identical over a region of from about 8 to about 100 or more residues (including any range within from about 8 to about 100 residues).
[344] “Subject” and “patient” as used herein interchangeably refers to any vertebrate, including, but not limited to, a mammal and a human. In some embodiments, the subject may be a human or a non-human. The subject or patient may be undergoing forms of treatment.
“Mammal” as used herein refers to any member of the class Mammalia, including, without limitation, humans and nonhuman primates such as chimpanzees and other apes and monkey species; farm animals such as cattle, sheep, pigs, goats, llamas, camels, and horses; domestic mammals such as dogs and cats; laboratory animals including rodents such as mice, rats, rabbits, guinea pigs, and the like. The term does not denote a particular age or sex. Thus, adult and newborn subjects, as well as fetuses, whether male or female, are intended to be included within the scope of this term.
[345] “Treat,” “treating” or “treatment” are each used interchangeably herein to describe reversing, alleviating, or inhibiting the progress of a disease and/or injury, or one or more
symptoms of such disease, to which such term applies. In some aspects, a treatment may be either performed in an acute or chronic way. Depending on the condition of the subject, the term also refers to preventing a disease or injury, and includes preventing the onset of a disease or injury, or preventing the symptoms associated with a disease or injury. "Preventing" also refers to preventing the recurrence of a disease or injury or of one or more symptoms associated with such disease or injury. "Treatment" and "therapeutically," refer to the act of treating, as "treating" is defined above. In some aspects, the prevention or treatment of a disease can be done prior to affliction or injury, such as, for example, to reduce the severity of a disease or injury or symptoms associated with a disease or an injury. Such prevention or reduction can include (a) administration of one or more pharmaceutical composition and/or one or more nutritional compositions to a subject; (b) the use of one or more of physical therapy, occupational therapy, and/or counseling; or (c) any combinations of (a) and (b).
[346] “Variant” is used herein to describe a peptide or polypeptide that differs in amino acid sequence by the insertion, deletion, or conservative substitution of amino acids, but retain at least one biological activity. “SNP” refers to a variant that is a single nucleotide polymorphism. Representative examples of “biological activity” include the ability to be bound by a specific antibody or to promote an immune response. Variant is also used herein to describe a protein with an amino acid sequence that is substantially identical to a referenced protein with an amino acid sequence that retains at least one biological activity. A conservative substitution of an amino acid, i.e., replacing an amino acid with a different amino acid of similar properties (e.g., hydrophilicity, degree, and distribution of charged regions) is recognized in the art as typically involving a minor change. These minor changes can be identified, in part, by considering the hydropathic index of amino acids, as understood in the art. Kyte et al., J. Mol. Biol. 157:105-132 (1982). The hydropathic index of an amino acid is based on a consideration of its hydrophobicity and charge. It is known in the art that amino acids of similar hydropathic indexes can be substituted and still retain protein function. In one aspect, amino acids having hydropathic indexes of ±2 are substituted. The hydrophilicity of amino acids can also be used to reveal substitutions that would result in proteins retaining biological junction. A consideration of the hydrophilicity of amino acids in the context of a peptide permits calculation of the greatest local average hydrophilicity of that peptide, a useful measure that has been reported to correlate well with antigenicity and immunogenicity. U.S. Patent No. 4,554,101, incorporated fully herein
by reference. Substitution of amino acids having similar hydrophilicity values can result in peptides retaining biological activity, for example immunogenicity, as is understood in the art. Substitutions may be performed with amino acids having hydrophilicity values within ±2 of each other. Both the hydrophobicity index and the hydrophilicity value of amino acids are influenced by the particular side chain of that amino acid. Consistent with that observation, amino acid substitutions that are compatible with biological function are understood to depend on the relative similarity of the amino acids, including the side chains of those amino acids, as revealed by the hydrophobicity, hydrophilicity, charge, size, and other properties.
[347] “Vector” is used herein to describe a nucleic acid molecule that can transport another nucleic acid to which it has been linked. One type of vector is a “plasmid”, which refers to a circular double-stranded DNA loop into which additional DNA segments may be ligated. Another type of vector is a viral vector, wherein additional DNA segments may be ligated into the viral genome. Certain vectors can replicate autonomously in a host cell into which they are introduced (e.g., bacterial vectors having a bacterial origin of replication and episomal mammalian vectors). Other vectors (e.g., non-episomal mammalian vectors) can be integrated into the genome of a host cell upon introduction into the host cell, and thereby are replicated along with the host genome. Moreover, certain vectors are capable of directing the expression of genes to which they are operatively linked. Such vectors are referred to herein as “recombinant expression vectors” (or simply, “expression vectors”). In general, expression vectors of utility in recombinant DNA techniques are often in the form of plasmids. “Plasmid” and “vector” may be used interchangeably as the plasmid is the most commonly used form of vector. However, other forms of expression vectors, such as viral vectors (e.g., replication defective retroviruses, adenoviruses and adeno-associated viruses), which serve equivalent functions, can be used. In this regard, RNA versions of vectors (including RNA viral vectors) may also find use in the context of the present disclosure.
[348] Unless otherwise defined herein, scientific and technical terms used in connection with the present disclosure shall have the meanings that are commonly understood by those of ordinary skill in the art. For example, any nomenclatures used in connection with, and techniques of, cell and tissue culture, molecular biology, immunology, microbiology, genetics and protein and nucleic acid chemistry and hybridization described herein are those that are well known and commonly used in the art. The meaning and scope of the terms should be
clear; in the event, however of any latent ambiguity, definitions provided herein take precedent over any dictionary or extrinsic definition. Further, unless otherwise required by context, singular terms shall include pluralities and plural terms shall include the singular.
2. Methods, Systems, and Kits for Determining the Concentration or Level of an Analyte of Interest using a Competitive and Sandwich Immunoassay Assay Format
[349] In some aspects, the present disclosure relates to methods, systems, and kits for determining the concentration or level of an analyte of interest in a sample, e.g., a biological sample) using a combination of a competitive immunoassay and a sandwich immunoassay. The use of a combination of a competitive immunoassay and a sandwich immunoassay in the methods, systems, and kits described herein provides several advantages over using a competitive immunoassay or a sandwich immunoassay alone or individually to determine the concentration or level of an analyte of interest in a biological sample. Specifically, the use of a competitive immunoassay and a sandwich immunoassay to determine the concentration or level of an analyte of interest in a biological sample extends the dynamic range of the assays, and eliminates the need for pre-sample dilution or sample re-testing.
[350] As mentioned above, the methods, systems, and kits employ both a competitive immunoassay and a sandwich immunoassay. In a competitive immunoassay, a biological sample suspected of containing an analyte of interest is combined with one or more reagents comprising an analyte of interest labeled with a detectable label (a “competing analyte”), a capture reagent, and a detection reagent to form a reaction mixture. The competing analyte competes with the analyte of interest contained in the biological sample for binding to the capture reagent. The biological sample, competing analyte, capture reagent and detection reagent can be added in any order to form the reaction mixture. In some aspects, the competing analyte is the same analyte as the analyte of interest or an analog thereof. In yet other aspects, the capture reagent is immobilized on a solid support. In still other aspects, the capture reagent used in the competitive immunoassay is the same or identical to the capture reagent used in the sandwich immunoassay.
[351] The signal that is derived from the total volume of the reaction mixture of the competitive immunoassay (“competitive immunoassay detectable signal”) can be quantified using routine techniques known in the art. For example, if an enzymatic label is used, the labeled complex that is formed (capture reagent-analyte of interest-detection reagent) is reacted
with a substrate for the label that gives a quantifiable reaction such as the development of color, chemiluminescence, fluorescence, or any other forms of light, or current. If the label is a radioactive label, the label is quantified using a scintillation counter. If the label is a fluorescent label, the label is quantified by stimulating the label with a light of one color (which is known as the “excitation wavelength”) and detecting another color (which is known as the “emission wavelength”) that is emitted by the label in response to the stimulation. If the label is a chemiluminescent label, the label is quantified detecting the light emitted either visually or by using luminometers, x-ray film, high speed photographic film, a charge-coupled device (CCD) camera, etc.
[352] Any solid support known in the art can be used in the present disclosure, including but not limited to, solid supports made out of polymeric materials in the forms of wells of a reaction tray, test tubes, particles, or beads (for example, polystyrene beads, magnetic beads), nitrocellulose strips, membranes, microparticles (for example, latex particles, sheep and DURACYTES® (Abbott Laboratories, Abbott Park, IL; DURACYTES® are red blood cells that have been “fixed” by pyruvic aldehyde and formaldehyde)).
[353] The solid phase also can comprise any suitable porous material with sufficient porosity to allow access by a detection reagent and a suitable surface affinity to bind the analyte of interest. Microporous structures are generally used, but materials with gel structure in the hydrated state may be used as well. Such useful solid supports include, but are not limited to, nitrocellulose and nylon. Such porous solid supports are in the form of sheets of thickness from about 0.01 to 0.5 mm, including about 0.1 mm. The pore size may vary within wide limits, and can be from about 0.025 to about 15 microns, especially from about 0.15 to about 15 microns. The surface of such supports may be activated by chemical processes which cause covalent linkage of the capture reagent to the support. The irreversible binding of the capture reagent is obtained, however, in general, by adsorption on the porous material by poorly understood hydrophobic forces.
[354] The capture reagent can be bound to the solid support or solid phase by adsorption, by covalent bonding using a chemical coupling agent or by other means known in the art, provided that such binding does not interfere with the ability of the capture reagent to bind to the analyte of interest. Alternatively, the capture reagent can be bound with microparticles that have previously coated with streptavidin or biotin (for example, using Power-BindTM-SA-MP
streptavidin coated microparticles, available from Seradyn, Indianapolis, Indiana, with a capture reagent that has been biotinylated using means known in the art). Alternatively, the capture reagent can be bound using microparticles that have been previously coated with anti-species specific monoclonal antibodies. Moreover, if necessary, the solid support can be derivatized to allow reactivity with various functional groups on the capture reagent. Such derivatization requires the use of certain coupling agents such as, but not limited to, maleic anhydride, N- hydroxysuccinimide and 1 -ethyl-3-(3-dimethylaminopropyl)carbodiimide.
[355] The analyte of interest or analog thereof used as the competing analyte and the antigen or antibody or antibody fragment thereof used in detection reagent each comprise at least one detectable label. Any detectable label known in the art can be used. For example, the detectable label can be a radioactive label, such as, 3H, 1251, 35S, 14C, 32P, 33P, an enzymatic label, such as horseradish peroxidase, alkaline phosphatase, glucose 6-phosphate dehydrogenase, etc., a chemiluminescent label, such as, acridinium (e.g., acridium esters, acridinium SPSP (N10-(3- sulfopropyl)-N-(3-sulfopropyl, etc.), luminol, isoluminol, thioesters, sulfonamides, phenanthridinium esters, etc. a fluorescence label, such as, fluorescein (5 -fluorescein, 6- carboxyfluorescein, 3’6-carboxyfluorescein, 5(6)-carboxyfluorescein, 6-hexachloro-fluorescein, 6-tetrachlorofluorescein, fluorescein isothiocyanate, etc.), rhodamine, phycobiliproteins, R- phycoerythrin, quantum dots (zinc sulfide-capped cadmium selenide), a thermometric label or an immuno-polymerase chain reaction label. An introduction to labels, labeling procedures and detection of labels is found in Polak and Van Noorden, Introduction to Immunocytochemistry, 2nd ed., Springer Verlag, N.Y. (1997) and in Haugland, Handbook of Fluorescent Probes and Research Chemicals (1996), which is a combined handbook and catalogue published by Molecular Probes, Inc., Eugene, Oregon. In some embodiments, the detectable label comprises N10-(3-sulfopropyl)-N-(3-carboxypropyl)-acridinium-9-carboxamide (CPSP-acridinium ester aka CPSP). In some embodiments, the detectable label comprises CPSP2-acridium ester (aka CPSP2), which is a derivative of CPSP, as described herein. In some embodiments, the detectable label comprises CPSP3 -acridinium ester (aka CPSP3), which is a derivative of CPSP, as described herein.
[356] In addition to the detectable labels described above, the compounds described in Sections 3 and 4 herein, can also be used as detectable labels.
[357] The detectable label can be bound to the analyte of interest used in the competing analyte or to the antigen or antibody or antibody fragment thereof in the detection reagent either directly or through a coupling agent. An example of a coupling agent that can be used is ED AC (l-ethyl-3 -(3 -dimethylaminopropyl) carbodiimide, hydrochloride) that is commercially available from Sigma- Aldrich, St. Louis, MO. Other coupling agents that can be used are known in the art. Methods for binding a detectable label to an antibody are known in the art. Additionally, many detectable labels can be purchased or synthesized that already contain end groups that facilitate the coupling of the detectable label to the antibody, such as, N10-(3-sulfopropyl)-N-(3- carboxypropyl)-acridinium-9-carboxamide, otherwise known as CPSP-Acridinium Ester or N10- (3-sulfopropyl)-N-(3-sulfopropyl)-acridinium-9-carboxamide, otherwise known as SPSP- Acridinium Ester.
[358] In some further aspects, the detectable label used in the detection reagent in the competitive immunoassay is different than the detectable label used in the detection reagent used in the sandwich immunoassay.
[359] In a sandwich immunoassay, a biological sample suspected of containing an analyte of interest is combined with one or more reagents comprising a capture reagent and a detection reagent to form a reaction mixture. In some aspects, in a “two step” sandwich immunoassay, the biological sample and capture reagent are subject to a washing step to remove any unbound analyte prior to the addition of the detection reagent to the reaction mixture. In a “one step” sandwich immunoassay, the biological sample containing the analyte of interest, the capture reagent, and the detection reagent are all combined and subjected to a washing step to remove any unbound analyte. The biological sample, capture reagent, and detection reagent can be added in any order to form the reaction mixture for “one step” sandwich immunoassay. In some aspects, the capture reagent is immobilized on a solid support. In other aspects, the detection reagent is immobilized on a solid support. In still other aspects, the capture reagent used in the sandwich immunoassay is the same or identical to the capture reagent used in the competitive immunoassay.
[360] The detectable signal that is derived from the total volume of the reaction mixture of the sandwich immunoassay (“sandwich immunoassay detectable signal”) is measured using routine techniques known in the art. For example, if an enzymatic label is used, the labeled complex (capture reagent-analyte of interest-detection reagent) is reacted with a substrate for the
label that gives a quantifiable reaction such as the development of color, chemiluminescence, fluorescence or any other forms of light, or current. If the label is a radioactive label, the label is quantified using a scintillation counter. If the label is a fluorescent label, the label is quantified by stimulating the label with a light of one color (which is known as the “excitation wavelength”) and detecting another color (which is known as the “emission wavelength”) that is emitted by the label in response to the stimulation. If the label is a chemiluminescent label, the label is quantified detecting the light emitted either visually or by using luminometers, x-ray film, high speed photographic film, a CCD camera, etc.
[361] Any solid support known in the art can be used in the present disclosure, including but not limited to, solid supports made out of polymeric materials in the forms of wells of a reaction tray, test tubes, particles, or beads (for example, polystyrene beads, magnetic beads), nitrocellulose strips, membranes, microparticles (for example, latex particles, sheep and DURACYTES® (Abbott Laboratories, Abbott Park, IL; DURACYTES® are red blood cells that have been “fixed” by pyruvic aldehyde and formaldehyde)).
[362] The solid phase also can comprise any suitable porous material with sufficient porosity to allow access by a capture or detection reagent and a suitable surface affinity to bind the analyte of interest. Microporous structures are generally used, but materials with gel structure in the hydrated state may be used as well. Such useful solid supports include, but are not limited to, nitrocellulose and nylon. Such porous solid supports are in the form of sheets of thickness from about 0.01 to 0.5 mm, including about 0.1 mm. The pore size may vary within wide limits, and can be from about 0.025 to about 15 microns, especially from about 0.15 to about 15 microns. The surface of such supports may be activated by chemical processes which cause covalent linkage of the capture reagent or detection reagent to the support. The irreversible binding of the capture reagent or detection reagent is obtained, however, in general, by adsorption on the porous material by poorly understood hydrophobic forces.
[363] The capture reagent or detection reagent can be bound to the solid support or solid phase by adsorption, by covalent bonding using a chemical coupling agent or by other means known in the art, provided that such binding does not interfere with the ability of the capture reagent or detection reagent to bind to the analyte of interest. Alternatively, the capture reagent or detection reagent can be bound with microparticles that have previously coated with streptavidin or biotin (for example, using Power-BindTM-SA-MP streptavidin coated
microparticles, available from Seradyn, Indianapolis, Indiana, with capture reagents or detection reagents that have been biotinylated using means known in the art). Alternatively, the capture reagent or detection reagents can be bound using microparticles that have been previously coated with anti-species specific monoclonal antibodies. Moreover, if necessary, the solid support can be derivatized to allow reactivity with various functional groups on the capture reagent or detection reagent. Such derivatization requires the use of certain coupling agents such as, but not limited to, maleic anhydride, N-hydroxy succinimide and l-ethyl-3-(3- dimethylaminopropyl)carbodiimide.
[364] The antigen or antibody or antibody fragment thereof used in detection reagent comprises at least one detectable label. Any detectable label known in the art can be used. For example, the detectable label can be a radioactive label, such as, 3H, 1251, 35S, 14C, 32P, 33P, an enzymatic label, such as horseradish peroxidase, alkaline phosphatase, glucose 6-phosphate dehydrogenase, etc., a chemiluminescent label, such as, acridinium (e.g., acridium esters, acridinium SPSP (N10-(3-sulfopropyl)-N-(3-sulfopropyl, etc.), luminol, isoluminol, thioesters, sulfonamides, phenanthridinium esters, etc. a fluorescence label, such as, fluorescein (5- fluorescein, 6-carboxyfhiorescein, 3’6-carboxyfluorescein, 5(6)-carboxyfluorescein, 6- hexachloro-fluorescein, 6-tetrachlorofluorescein, fluorescein isothiocyanate, etc.), rhodamine, phycobiliproteins, R-phycoerythrin, quantum dots (zinc sulfide-capped cadmium selenide), a thermometric label or an immuno-polymerase chain reaction label. An introduction to labels, labeling procedures and detection of labels is found in Polak and Van Noorden, Introduction to Immunocytochemistry, 2nd ed., Springer Verlag, N.Y. (1997) and in Haugland, Handbook of Fluorescent Probes and Research Chemicals (1996), which is a combined handbook and catalogue published by Molecular Probes, Inc., Eugene, Oregon. In some embodiments, the detectable label comprises CPSP. In some embodiments, the detectable label comprises CPSP2, as described herein. In some embodiments, the detectable label comprises CPSP3, as described herein.
[365] In addition to the detectable labels described above, the compounds described in Section 3 herein, can also be used as detectable labels.
[366] The detectable label can be bound to the antigen or antibody or antibody fragment thereof in the detection reagent either directly or through a coupling agent. An example of a coupling agent that can be used is ED AC (l-ethyl-3 -(3 -dimethylaminopropyl) carbodiimide,
hydrochloride) that is commercially available from Sigma-Aldrich, St. Louis, MO. Other coupling agents that can be used are known in the art. Methods for binding a detectable label to an antibody are known in the art. Additionally, many detectable labels can be purchased or synthesized that already contain end groups that facilitate the coupling of the detectable label to the antibody, such as, N10-(3-sulfopropyl)-N-(3-carboxypropyl)-acridinium-9-carboxamide, otherwise known as CPSP-Acridinium Ester or N10-(3-sulfopropyl)-N-(3-sulfopropyl)- acridinium-9-carboxamide, otherwise known as SPSP-Acridinium Ester.
[367] In some further aspects, the detectable label used in the detection reagent in the sandwich immunoassay is a different than the detectable label used in the detection reagent used in the competitive immunoassay.
[368] In some further aspects, the first detectable signal and second detectable signal used in the sandwich immunoassay have distinguishable reaction kinetics.
[369] The methods of the present disclosure involve obtaining a calibration curve for the analyte of interest. The calibration curve can be generated using routine techniques known in the art. Typically, calibration curves are generated using varying concentrations or serial dilutions of an analyte of interest at a known concentration. Other than using serial dilutions of the marker, the standard curve can be generated gravimetrically, by mass spectroscopy and by other techniques known in the art.
[370] In some aspects, the calibration curve is generated by: (a) performing a sandwich immunoassay on the analyte of interest as described above; (b) performing a competitive immunoassay on analyte of interest as described above; (c) calculating the signal ratio of first detectable signal and the second detectable signal; and (d) the varying concentrations of the analyte of interest and repeating steps a-c as many times as necessary (e.g., 1 time, 2 times, 3 times, 4 times, 5 times, 6 times, 7 times, 8 times, 9 times, or 10 times) to generate a calibration curve using the signal ratios generated in step c. In some aspects, when generating the calibration curve, the competitive immunoassay and the sandwich immunoassay are performed in the same reaction vessel. In other aspects, the competitive immunoassay and the sandwich immunoassay are performed in different reaction vessels. Additionally, in still yet other aspects, when generating the calibration curve, the capture reagent used in the competitive immunoassay and the sandwich immunoassay are identical. In still yet even further aspects, when generating the calibration curve, the detectable label used in detection reagent in the competitive
immunoassay and the detectable label used in the detection reagent in the sandwich immunoassay are not identical.
[371] The method of the present disclosure fiirther involves performing a competitive immunoassay and a sandwich immunoassay on a biological sample containing an analyte of interest. The order in which the competitive immunoassay and the sandwich immunoassay are performed is not critical and can be performed sequentially or simultaneously. For example, the competitive immunoassay can be performed first on the biological sample followed by the sandwich immunoassay. Alternatively, the sandwich immunoassay can be performed first on the biological sample followed by the competitive immunoassay. Still fiirther alternatively, the competitive immunoassay and the sandwich immunoassay can be performed simultaneously.
[372] In still other aspects, a single (e.g., the same) biological sample obtained from the subject can be used to perform each of the competitive immunoassay and the sandwich immunoassay. Alternatively, multiple biological samples can be obtained from the subject and used to perform each of the competitive immunoassay and the sandwich immunoassay. Additionally, the biological samples used in the methods of the present disclosure do not require dilution prior to performing the competitive immunoassay, prior to performing the sandwich immunoassay, or prior to perform both the competitive immunoassay and prior to performing the sandwich immunoassay. In fact, one of the advantages of the methods of the disclosure is that the biological sample requires no dilution prior to performing any of the assays described herein. No pre-dilution of the biological sample avoids the problems known in the art with under and over diluting biological samples prior to performing a competitive immunoassay or sandwich immunoassay.
[373] In some aspects, the competitive immunoassay and the sandwich immunoassay are performed in a single (e.g., the same) reaction vessel using two at least two different detectable labels (e.g., the competitive immunoassay and the sandwich immunoassay each use different detectable labels). In other aspects, the competitive immunoassay and the sandwich immunoassay are performed in two different (e.g., separate) reaction vessels using at least two different detectable labels (e.g., the competitive immunoassay and the sandwich immunoassay each use different detectable labels). When the competitive immunoassay and the sandwich immunoassay are performed in a single (e.g., the same) reaction vessel, the competitive immunoassay signal detectable signal is detected by a first detection component in an instrument
and the sandwich immunoassay detectable signal is detected by a second detection component in an instrument. For example, if the label is a chemiluminescent label, the first detection component can be a first photomultiplier tube with a first specific filter (e.g., a blue filter) in an instrument, and the second detection component can be a second photomultiplier tube with a second specific filter (e.g., a green filter) in an instrument.
[374] In some aspects, the competitive immunoassay and the sandwich immunoassay are performed in two different (e.g., separate) reaction vessels (a first reaction vessel and a second reaction vessel) but employ the same detectable label (e.g., the competitive immunoassay and the sandwich immunoassay each use the same detectable label). When the competitive immunoassay and the sandwich immunoassay are performed in two different (e.g., separate) reaction vessels, the immunoassays can be performed simultaneously, or sequentially, in any order. When the competitive immunoassay and the sandwich immunoassay are performed in different (e.g., separate) reaction vessels, the competitive immunoassay signal detectable signal and the sandwich immunoassay detectable signal can be detected by a single detection component in an instrument. For example, if the label used in the competitive immunoassay and sandwich immunoassay is a chemiluminescent label, the detection component can be a single photomultiplier tube with a specific filter (e.g., a blue filter) in an instrument that can be used to detect the chemiluminescent signal from each of the competitive immunoassay and sandwich immunoassay in their respective reaction vessels.
[375] In some aspects, a method of detecting an analyte of interest in a biological sample is disclosed, the method comprising the steps of: a) contacting a biological sample with at least one specific binding member that binds to the analyte of interest to form at least one complex, wherein the specific binding member comprises the compound of Formula (I), as described herein; and b) detecting the presence or absence of a signal from the specific binding member, wherein detection of the signal indicates that the analyte is present in the sample and the absence of the signal indicates that the analyte is not present in the sample. In some embodiments, the compound of Formula (I) does not have a fluorophore conjugated to it. In some embodiments, the first specific binding member and/or second specific binding member are immobilized on a solid support. In some embodiments, the method is performed using a clinical chemistry assay, an immunoassay, or single molecule detection assay. In some embodiments, the method further
comprises adding hydrogen peroxide to the biological sample prior to the detecting step. In some embodiments, the compound of Formula (I) has an emission wavelength in a range from about 445 nm to about 520 nm. In some embodiments, the emission is detected using a photomultiplier tube (PMT).
[376] In some other aspects, a method of detecting two or more analytes of interest in a biological sample, the method comprising the steps of: a) contacting the biological sample either simultaneously or sequentially with (i) at least one first specific binding member that binds to a first analyte of interest to form at least one first complex; and (ii) at least one second specific binding member that binds to a second analyte of interest to form at least one second complex, wherein each of the first and second specific binding members comprise the compound of Formula (I), and wherein the compound associated with each of the first and second specific binding members is different; and b) detecting the presence or absence of a signal from each of the first and second specific binding members, wherein (i) detection of a signal from the first specific binding member indicates that the first analyte is present in the sample and the absence of a signal from the first specific binding member indicates that the first analyte is not present in the sample; and (ii) detection of a signal from the second specific binding member indicates that the second analyte is present in the sample and the absence of a signal from the second specific binding member indicates that the second analyte is not present in the sample. In some embodiments, the compound of Formula (I) does not have a fluorophore conjugated to it. In some embodiments, the first specific binding member and/or second specific binding member are immobilized on a solid support. In some embodiments, the method is performed using a clinical chemistry assay, an immunoassay, or single molecule detection assay. In some embodiments, the method further comprises adding hydrogen peroxide to the biological sample prior to the detecting step. In some embodiments, the compound of Formula (I) has an emission wavelength in a range from about 445 nm to about 520 nm. In some embodiments, the emission is detected using a PMT.
[377] In some aspects, the competitive immunoassay detectable signal and the sandwich immunoassay detectable signal are communicated on or from at least one instrument. Suitable instruments are described herein, including non-point-of-care devices that may contain a user interface that communicate by displaying the determination. Suitable instruments are described herein, including non-point-of-care devices (e.g. the ARCHITECT® and Alinity platforms
marketed by Abbott Laboratories) that may contain a user interface that communicate by displaying the determination. In other aspects, suitable instruments include point-of-care devices (such as the iSTAT® Abbott Point of Care devices).
[378] In some embodiments, the instrument contains software to execute one or more tasks. In some embodiments, the instrument contains software to automatically determine the next appropriate step in a method as described herein. For example, the instrument may contain software that determines the amount or level of an analyte of interest in a biological sample. The software may display this determination, such as on a graphical user interface.
[379] In some embodiments, the instrument stores software that instructs a processor to execute a given task. In some embodiments, the software stores machine readable instructions that instruct a processor to execute a given task. The machine readable instructions may be one or more executable programs or portion(s) of an executable program for execution by a computer. The programs may be embodied in software stored on a non-transitory computer readable storage medium such as a CD-ROM, a floppy disk, a hard drive, a DVD, a Blu-ray disk, or a memory associated with the processors. Alternatively, the entire programs and/or parts thereof could alternatively be executed by a device other than the processors and/or embodied in firmware or dedicated hardware. Additionally or alternatively, processes may be implemented by one or more hardware circuits (e.g., discrete and/or integrated analog and/or digital circuitry, an FPGA, an ASIC, a comparator, an operational-amplifier (op-amp), a logic circuit, etc.) structured to perform the corresponding operation without executing software or firmware.
[389] The machine readable instructions may be stored in one or more of a compressed format, an encrypted format, a fragmented format, a compiled format, an executable format, a packaged format, etc. Machine readable instructions as described herein may be stored as data (e.g., portions of instructions, code, representations of code, etc.) that may be utilized to create, manufacture, and/or produce machine executable instructions. For example, the machine readable instructions may be fragmented and stored on one or more storage devices and/or computing devices (e.g., servers). The machine readable instructions may require one or more of installation, modification, adaptation, updating, combining, supplementing, configuring, decryption, decompression, unpacking, distribution, reassignment, compilation, etc. in order to make them directly readable, interpretable, and/or executable by a computing device and/or other machine. For example, the machine readable instructions may be stored in multiple parts, which
are individually compressed, encrypted, and stored on separate computing devices, wherein the parts when decrypted, decompressed, and combined form a set of executable instructions that implement a program such as that described herein.
[381] In another example, the machine readable instructions may be stored in a state in which they may be read by a computer, but require addition of a library (e.g., a dynamic link library (DLL)), a software development kit (SDK), an application programming interface (API), etc. in order to execute the instructions on a particular computing device or other device. In another example, the machine readable instructions may need to be configured (e.g., settings stored, data input, network addresses recorded, etc.) before the machine readable instructions and/or the corresponding program(s) can be executed in whole or in part. Thus, the disclosed machine readable instructions and/or corresponding program(s) are intended to encompass such machine readable instructions and/or program(s) regardless of the particular format or state of the machine readable instructions and/or program(s) when stored or otherwise at rest or in transit.
[382] The machine readable instructions described herein can be represented by any past, present, or future instruction language, scripting language, programming language, etc. For example, the machine readable instructions may be represented using any of the following languages: C, C++, Java, C#, Perl, Python, JavaScript, HyperText Markup Language (HTML), Structured Query Language (SQL), Swift, etc.
[383] The machine readable instructions may be stored on a non-transitory computer and/or machine readable medium such as a hard disk drive, a flash memory, a read-only memory, a compact disk, a digital versatile disk, a cache, a random-access memory and/or any other storage device or storage disk in which information is stored for any duration (e.g., for extended time periods, permanently, for brief instances, for temporarily buffering, and/or for caching of the information). As used herein, the term non-transitory computer readable medium is expressly defined to include any type of computer readable storage device and/or storage disk and to exclude propagating signals and to exclude transmission media.
[384] Once each of the competitive immunoassay detectable signal and sandwich immunoassay detectable signal are obtained from the instrument, a signal ratio of (i) the competitive immunoassay detectable signal to sandwich immunoassay detectable signal; or (2) the sandwich immunoassay detectable signal to competitive immunoassay detectable signal, is
calculated. Once the signal ratio is calculated, the final concentration or level of the analyte of interest in the biological sample can be determined using the calibration curve.
[385] The disclosed methods and systems for determining the concentration or level of analyte of interest present in a sample may be as described herein. The methods may also be adapted in view of other methods for analyzing analytes such as single molecule detection described below. For example, the methods, systems, and kits as described herein may also involve single molecule counting.
[386] Any device known in the art that allows for the detection of a single molecule of one or more analytes of interest can be used in the systems described herein. For example, the device can be a microfluidics device, digital microfluidics device, a surface acoustic wave based microfluidic device (SAW), an integrated digital microfluidics device and analyte detection device, an integrated SAW and analyte detection device, or robotics based assay processing unit. Examples of other devices that can be used include the Quanterix SIMOA™ (Lexington, MA), Singulex’s single molecule counting (SMC™) technology (Alameda, CA, see for example, U.S. patent No. 9,239,284, the contents of which are herein incorporated by reference), etc.
[387] Other methods of detection include the use of or can be adapted for use on a nanopore device or nanowell device. Examples of nanopore devices are described in International Patent Publication No. WO 2016/161402, which is hereby incorporated by reference in its entirety. Examples of nanowell device are described in International Patent Publication No. WO 2016/161400, which is hereby incorporated by reference in its entirety.
[388] The methods and kits as described herein can involve mass spectrometry using DIAMS, DDA-MS or SRM/MRM-MS or PRM-MS. In certain embodiments, methods for analyte analysis can involve assessing a sample for the presence of an analyte. In certain embodiments, assessing a sample for the presence of an analyte can be used for determining presence of and/or concentration of an analyte or a fragment in a sample. In certain embodiments, a method can also be used for determining presence of and/or concentration of a plurality of different analytes or analyte fragments present in a sample. Quantification can be performed using internal control proteins or peptide fragments.
[389] The methods, systems, and kits as described herein encompass other reagents and methods for carrying out the competitive immunoassay and the sandwich immunoassay. For instance, encompassed are various buffers such as are known in the art and/or which can be
readily prepared or optimized to be employed, e.g., for washing, as a conjugate diluent, and/or as a calibrator diluent. An exemplary conjugate diluent is an ARCHITECT® diluent (Abbott Laboratories, Abbott Park, IL) containing 2-(7V-morpholino)ethanesulfonic acid (MES), another salt, protein blockers, an antimicrobial and detergent. An exemplary calibrator diluent is ARCHITECT® calibrator diluent (Abbott Laboratories, Abbott Park, IL), which comprises a buffer containing MES, another salt, a protein blocker and an antimicrobial.
[390] Furthermore, as previously mentioned, the methods, systems, and kits optionally are adapted for use on an automated or semi-automated system. Some of the differences between an automated or semi-automated system as compared to a non-automated system include the substrate to which the capture antibody is attached (which can impact sandwich formation and analyte reactivity), and the length and timing of the capture, detection and/or any optional wash steps. Whereas a non-automated format may include a relatively longer incubation time with sample and capture reagent (e.g., about 2 hours) an automated or semi-automated format (e.g., ARCHITECT® or Alinity) may have a relatively shorter incubation time (e.g., approximately 18 minutes for ARCHITECT®). Similarly, whereas a non-automated format may incubate a detection antibody for a relatively longer incubation time (e.g., about 2 hours), an automated or semi-automated format (e.g., ARCHITECT® or Alinity) may have a relatively shorter incubation time (e.g., approximately 4 minutes for the ARCHITECT®).
[391] The power of a diagnostic test to correctly predict status is commonly measured as the sensitivity of the assay, the specificity of the assay or the area under a receiver operated characteristic (“ROC”) curve. Sensitivity is the percentage of true positives that are predicted by a test to be positive, while specificity is the percentage of true negatives that are predicted by a test to be negative. A ROC curve provides the sensitivity of a test as a function of 1 -specificity. The greater the area under the ROC curve, the more powerful the predictive value of the test. Other useful measures of the utility of a test are positive predictive value and negative predictive value. Positive predictive value is the percentage of people who test positive that are actually positive. Negative predictive value is the percentage of people who test negative that are actually negative.
[392] Furthermore, in certain embodiments, the values measured for an analyte of interest can be mathematically combined with other values measured for other analytes and the combined value is correlated to the underlying diagnostic question. Biomarker values may be
combined by any appropriate state of the art mathematical method. Well-known mathematical methods for correlating a marker combination to a disease status employ methods like discriminant analysis (DA) (e.g., linear-, quadratic-, regularized-DA), Discriminant Functional Analysis (DFA), Kernel Methods (e.g., SVM), Multidimensional Scaling (MDS), Nonparametric Methods (e.g., k-Nearest-Neighbor Classifiers), PLS (Partial Least Squares), Tree-Based Methods (e.g., Logic Regression, CART, Random Forest Methods, Boosting/Bagging Methods), Generalized Linear Models (e.g., Logistic Regression), Principal Components based Methods (e.g., SIMCA), Generalized Additive Models, Fuzzy Logic based Methods, Neural Networks and Genetic Algorithms based Methods. The skilled artisan will have no problem in selecting an appropriate method to evaluate a biomarker combination of the present disclosure. In one embodiment of the present disclosure, the method used in a correlating a biomarker combination (e.g. to diagnose HBV status) is selected from DA (e.g., Linear-, Quadratic-, Regularized Discriminant Analysis), DFA, Kernel Methods (e.g., SVM), MDS, Nonparametric Methods (e.g., k-Nearest-Neighbor Classifiers), PLS (Partial Least Squares), Tree-Based Methods (e.g., Logic Regression, CART, Random Forest Methods, Boosting Methods), or Generalized Linear Models (e.g., Logistic Regression), and Principal Components Analysis. Details relating to these statistical methods are found in the following references: Ruczinski et al., 12
J. OF COMPUTATIONAL AND GRAPHICAL STATISTICS 475-511 (2003); Friedman, J. H., 84 J. OF THE AMERICAN STATISTICAL ASSOCIATION 165-75 (1989); Hastie, Trevor, Tibshirani, Robert, Friedman, Jerome, The Elements of Statistical Learning, Springer Series in Statistics (2001); Breiman, L., Friedman, J. H., Olshen, R. A., Stone, C. J. Classification and regression trees, California: Wadsworth (1984); Breiman, L., 45 MACHINE LEARNING 5-32 (2001); Pepe, M. S., The Statistical Evaluation of Medical Tests for Classification and Prediction, Oxford Statistical Science Series, 28 (2003); and Duda, R. O., Hart, P. E., Stork, D. G., Pattern Classification, Wiley Interscience, 2nd Edition (2001).
3. Acridinium-Containing Compounds and Methods of Synthesizing Same
[393] In some aspects, the present disclosure relates to a compound of Formula (I) comprising:
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, and heterocyciylalkyl; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyciylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, and wherein X is selected from -F, -Cl, -Br, -I, -OH, -
OR*, -NH2, -NHR*, -N(R*)2, -N(R*)3J -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, - N(R*)-N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, - SR*, -CN, -NC, -CHF2, -CCI3, CF2C1, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O- NO, -O-ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O- (C=O)-H, -O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)- NHNH2, -O-C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, - N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)- C(=O)-NHR*, -N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)- C(=S)-N(R*)2, -S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*- S(=O)2-R*, -S(=O)2-N(R*)2, -P(R*)2, -O-P(-O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or - P(=O)(OH)-N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl, wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents.
[394] In some embodiments, Y is selected from nitrogen, oxygen, and sulfur. In some embodiments, Y is nitrogen. In some embodiments, when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, and heterocyclylalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Y is nitrogen and R1 is -SO2-A, and A is a substituted or unsubstituted aryl group. In some embodiments, Y is nitrogen and R1 is -SO2-A, and A is a aryl group with a methyl substitute. In some embodiments, when Y is oxygen or sulfur, R1 is absent;
[395] In some embodiments, Q is -SO3Z, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Q is -SO3H. In some embodiments, Q is -SO3Z wherein Z is a Ci -Ci6 group.
[396] In some embodiments, Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene,
heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Li and L2 are each independently selected from C1-C10 alkylene. In some embodiments, Li is — CH2CH2CH2 — . In some embodiments, L2 is — CH2CH2CH2— .
[397] In some embodiments, R2 is selected from -SO3Z and -COOZ, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R2 is -SO3H. In some embodiments, R2 is -SO3Z, wherein Z is a Ci -Ci6 group. In some embodiments, R2 is -COOZ, wherein Z is H. In some embodiments, R2 is -COOZ, wherein Z is is a Ci -Ci6 group. In some embodiments, R2 is - COOZ, wherein Z is N-hydroxysuccinimide (NHS).
[398] In some embodiments, Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Ra is hydrogen. In some embodiments, Rb is hydrogen. In some embodiments, Rc is hydrogen. In some embodiments, Rd is hydrogen. In some embodiments, Re is hydrogen. In some embodiments, Rf is hydrogen.
[399] In some embodiments, R’ and R” are each independently selected from H, Cl, Br, I, - NO2, -NH2, t-butyl, -SO3H, -S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, -NH2, -NHR*, - N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)-N(R*)2, -C=N- R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, -CN, -NC, - CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O-ON2, -N3, - N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(-O)-S-R*, -O-(C-O)-H, -O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O-C(C=O)- NHNH2, -C(-S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, - C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2J -
S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2J -O-P(=O)(OR*)2J -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl. In some embodiments, R’ and/or R” are H. In some embodiments, R’ and/or R” are Cl. In some embodiments, R’ and/or R” are Br. In some embodiments, R’ and/or R” are I. In some embodiments, R’ and/or R” are NO2. In some embodiments, R’ and/or R” are NH2. In some embodiments, R’ and/or R” are t-butyl. In some embodiments, R’ and/or R” are SO3H. In some embodiments, R’ and/or R” are substituted aryl groups. In some embodiments, R’ and/or R” are unsubstituted aryl groups. In some embodiments, R’ and/or R” are substituted heteroaryl groups. In some embodiments, R’ and/or R” are unsubstituted heteroaryl groups. In some embodiments, R’ and/or R” are substituted phenyl groups. In some embodiments, R’ and/or R” are unsubstituted phenyl groups. In some embodiments, R’ and/or R” are substituted pyrrolyl groups. In some embodiments, R’ and/or R” are unsubstituted pyrrolyl groups. In some embodiments, R’ and/or R” are substituted furanyl groups. In some embodiments, R’ and/or R” are unsubstituted furanyl groups. In some embodiments, R’ and/or R” are substituted thienyl groups. In some embodiments, R’ and/or R” are unsubstituted thienyl groups. In some embodiments, R’ and/or R” are substituted thienothiophenyl groups. In some embodiments, R’ and/or R” are unsubstituted thienothiophenyl groups. In some embodiments, R’ and/or R” are -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -S-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R’ is - S-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R” is -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R" is -S-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R” is -
S-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R’ and/or R” are -(S=O)- L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -(S=O)-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -(S=O)-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R’ is -(S=O)-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R” is (S=O)-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R" is -(S=O)-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R” is -(S=O)-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R’ and/or R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -SO2-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R’ is -SO2-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R" is -SO2-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R” is -SO2-L3-SO3H, wherein L3 is — CH2CH2CH2 — .
[400] In some embodiments, the compound of Formula (I) does not have a fluorophore conjugated to it.
[401] In some embodiments, the compound is a compound of Formula (Ila):
[403] In some embodiments, the compound of Formula (I) is functionalized at the 2- or 2,7 positions and is selected from Formulas:
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), and Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
[404] In some embodiments, the compound of Formula (I) is functionalized at the 2- or 2,7 positions and is selected from:
wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
[405] In some embodiments, the compound comprises a sulfide at the 2- or 2,7 position of the acridinium ring and is selected from Formulas:
[406] In some embodiments, the compound comprises a sulfide at the 2- or 2,7 position of the acridinium ring and is selected from:
[407] For compounds described herein, groups and substituents thereof may be selected in accordance with permitted valence of the atoms and the substituents, such that the selections and substitutions result in a stable compound, e.g., which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc.
[408] The compounds described herein may exist as stereoisomers wherein asymmetric or chiral centers are present. The stereoisomers are “R” or “S” depending on the configuration of substituents around the chiral carbon atom. The disclosure contemplates various stereoisomers and mixtures thereof, and these are specifically included within the scope of this disclosure.
Stereoisomers indude enantiomers and diastereomers and mixtures of enantiomers or diastereomers. Individual stereoisomers of the compounds may be prepared synthetically from commercially available starting materials, which contain asymmetric or chiral centers or by preparation of racemic mixtures followed by methods of resolution well-known to those of ordinary skill in the art, These methods of resolution are exemplified by (1) attachment of a mixture of enantiomers to a chiral auxiliary, separation of the resulting mixture of diastereomers by recrystallization or chromatography, and optional liberation of the optically pure product from the auxiliary as described in Fumiss, Hannaford, Smith, and Tatchell, “Vogel's Textbook of Practical Organic Chemistry’", 5th edition (1989), Longman Scientific & Technical, Essex CM20 2JE, England, or (2) direct separation of the mixture of optical enantiomers on chiral chromatographic columns, or (3) fractional recrystallization methods.
[409] It should be understood that the compounds may possess tautomeric forms as well as geometric isomers, and that these also constitute an aspect of the invention.
[410] The present disclosure also includes isotopically-Iabeled compounds, which are identical to those recited in Formula (I), but for the fact that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature. Examples of isotopes suitable for inclusion in the compounds of the invention are hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, and chlorine, such as, but not limited to, 2H, 3H, 1
respectively. Substitution with heavier isotopes such as deuterium, i.e., 2H, can afford certain advantages resulting from greater metabolic stability, for example increased in vivo half-life, and may therefore be preferred in some circumstances. The compound may incorporate positronemitting isotopes for medical imaging and positron-emitting tomography (PET) studies for determining the distribution of receptors. Suitable positron-emitting isotopes that can be incorporated in compounds of Formula (I) are :'!C. 13N, 15O, and :*F. Isotopically-Iabeled compounds of Formula (I) can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying examples using appropriate isotopically-Iabeled reagent in place of non-isotopically-labeled reagent.
[411] A compound disclosed herein may be in the form of a salt. The salts may be prepared during the final isolation and purification of the compounds or separately, for example by reacting a basic group of the compound (e.g., an amino group) with a suitable acid or by reacting
an acidic group of the compound (e.g., a carboxylic acid group) with a suitable base. Acid salts may be prepared during the final isolation and purification of the compounds or separately by reacti ng a suitable group of the compound, such as an amino group, with a suitable acid. For example, a compound may be dissolved in a suitable solvent, such as but not limited to methanol and water, and treated with at least one equivalent of an acid, such hydrochloric acid, The resulting salt may precipitate out and be isolated by filtration and dried under reduced pressure. Alternatively, the solvent and excess acid may be removed under reduced pressure to provide a salt. Representative salts include acetate, adipate, alginate, citrate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, camphorate, camphorsulfonate, digluconate, glycerophosphate, hemisulfate, heptauoate, hexanoate, formate, isethionate, fumarate, lactate, maleate, methanesulfonate, naphthylenesulfonate, nicotinate, oxalate, pamoate, pectinate, persulfate, 3 -phenylpropionate, picrate, oxalate, maleate, pivalate, propionate, succinate, tartrate, trichloroacetate, trifiuoroacetate, glutamate, para-toluenesulfonate, undecanoate, hydrochloric, hydrobromic, sulfuric, phosphoric and the like. The amino groups of the compounds may also be quatemized with alkyl chlorides, bromides and iodides such as methyl, ethyl, propyl, isopropyl, butyl, lauryl, myristyl, stearyl and the like, Basic addition salts may be prepared during the final isolation and purification of the disclosed compounds by reaction of a carboxyl group with a suitable base such as the hydroxide, carbonate, or bicarbonate of a metal cation such as lithium, sodium, potassium, calcium, magnesium, or aluminum, or an organic primary, secondary, or tertiary amine. Quaternary amine salts can be prepared, such as those derived from methylamine, dimethylamine, trimethylamine, triethylamine, diethylamine, ethylamine, tributylamine, pyridine, N,N-dimethylaniline, N-methylpiperidine, N-methylmorpholine, dicyclohexylamine, procaine, dibenzylamine, N,N-dibenzylphenethylamine, 1 -ephenamine and N,N'- dibenzylethylenediamine, ethylenediamine, ethanolamine, diethanolamine, piperidine, piperazine, and the like.
[412] In some other aspects, the present disclosure relates to methods of synthesizing the compounds of Formula (I). In some embodiments, the compounds of Formula (I) are readily prepared in high yields by reducing an acridinium-containing compound, e.g., wherein R’ and R” are H, to an acridan intermediate, followed by functionalization of the acridan intermediate at the 2- or 2,7-positions, e.g., by one of Cl, Br, I, NO2, t-butyl, or SO3H. In some embodiments, the compound of Formula (I) is functionalized at the 2- or 2,7-positions with I. In some
embodiments, the functionalized acridinium-containing compounds are further treated with a water soluble thiol, e.g., mercaptopropanesulfonic acid (MPS), in the presence of a catalyst, e.g., a Pd-containing catalyst, resulting in an acridinium sulfide of Formula (I), wherein (i) R’ is -S- L3-SO3H and R” is H or (ii) both R’ and R” are -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[413] In some aspects, a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring is disclosed. In some embodiments, a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring comprises: reducing the acridinium ring to an acridan intermediate using a first solution comprising at least one reducing agent and at least one acid; and reacting the acridan intermediate with an electrophile to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring.
[414] In some embodiments, the at least one acid in the first solution comprises at least one of ammonium chloride, formic acid, acetic acid, benzoic acid, hydrofluoric acid, phosphoric acid, sulfurous acid, hypochlorous acid, citric acid, carbonic acid, nitrous acid, oxalic acid, boric acid, ammonium sulfate, and ammonium oxalate. In some embodiments, the at least one reducing agent comprises zinc or a hydride (e.g., sodium borohydride, lithium aluminum hydride, sodium cyanoborohydride, sodium triacetoxyborohydride). In some embodiments, the at least one reducing agent comprises zinc. In some embodiments, the zinc comprises elemental zinc, a zinc alloy, or a combination of both. In some embodiments, the first solution further comprises a water-miscible alcohol, preferably ethanol. In some embodiments, the acridinium ring is reduced at temperature in a range from about 50°C to about 90°C, preferably about 60°C to about 80°C. In some embodiments, the acridan intermediate is isolated prior to reacting with the electrophile.
[415] In some embodiments, the acridan intermediate is represented by formula (III):
[416] In some embodiments, the electrophile comprises at least one of Cl, Br, I, NO2, SO3H, and t-butyl.
[417] In some embodiments, the electrophile is Cl, the reaction of the acridan intermediate with Cl to form a product comprising the acridinium functionalized at the 2- or the 2,7- position, or both, of the acridinium ring includes a chlorination reaction, and the products are represented by Formulas (IVa) and (IVb):
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I).
[418] In some embodiments, a method of producing the compound of Formula (IVa), (IVb), or both (IVa) and (IVb) comprises: mixing the acridan intermediate of Formula (III) with a second solution in a container, wherein the second solution comprises a chloride salt, N-
chlorosuccinimide, and p-toluenesulfonic acid in a water/acetonitrile mixture and stirring for about 12 h to about 24 h, preferably about 16 h to about 20 h; adding an aqueous solution of at least one oxidant to the container, in portions, over about 30 min to about 90 min, preferably about 45 min to about 75 min; and isolating and optionally purifying the product comprising the compound of Formula (IVa), (IVb), or both (IVa) and (IVb).
[419] In some embodiments, the oxidant comprises FeCh, 2,3 -Dichloro -5,6- -dicyano- 1 ,4- benzoquinone (DDQ), chloranil, or ceric ammonium nitrate (CAN).
[420] In some other embodiments, instead of chloride salt, N-chlorosuccinimide, and p- toluenesulfonic acid, the chlorination reaction comprises the use of sulfuryl chloride or chlorine gas.
[421] In some embodiments, if both are formed, the compound of Formula (IVa) is separated from the compound of Formula (IVb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
[422] In some embodiments, the electrophile is Br, the reaction of the acridan intermediate with Br to form a product comprising the acridinium functionalized at the 2- or 2,7- position, or both, of the acridinium ring includes a bromination reaction, and the products are represented by
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I).
[423] In some embodiments, a method of producing the compound of Formula (Va), (Vb), or both (Va) and (Vb) comprises: dissolving the acridan intermediate of Formula (III) in a third solution in a container; adding bromine to the container and stirring for about 30 min to about 1 h; and isolating and optionally purifying the product comprising the compound of Formula (Va),
(Vb), or both (Va) and (Vb). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
[424] In some other embodiments, the bromination reaction comprises the use of pyridinium tribromide (PyBn), hypobromous acid (HOBr), or N-bromosuccinimide.
[425] In some embodiments, if both are formed, the compound of Formula (Va) is separated from the compound of Formula (Vb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
[426] In some embodiments, the electrophile is I, the reaction of the acridan intermediate with I to form a product comprising the acridinium functionalized at the 2- or the 2,7- position, or both, of the acridinium ring includes a iodination reaction, and the products are represented by Formulas (Via) and (VIb):
[427] In some embodiments, a method of producing the compound of Formula (Via), (VIb), or both (Via) and (VIb) comprises: dissolving the acridan intermediate of Formula (III) in a third solution in a container; adding iodine monochloride to the container and stirring for about 30 min to about 1 h; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
[428] In some embodiments, if both are formed, the compound of Formula (Via) is separated from the compound of Formula (VIb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
[429] In some embodiments, the electrophile is NO2, the reaction of the acridan intermediate with NO2 to form a product comprising the acridinium functionalized at the 2,7- position of the acridinium ring includes a nitration reaction, and the products are represented by Formulas (Vila) and (Vllb):
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I).
[430] In some embodiments, a method of producing the compound of Formula (Vila),
(Vllb), or both (Vila) and (Vllb) comprises: dissolving the acridan intermediate of Formula (III) in a third solution in a container; adding nitric acid to the container; and isolating and optionally purifying the product comprising the compound of Formula (Vila), (Vllb), or both (Vila) and (Vllb). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
[431] In some embodiments, if both are formed, the compound of Formula (Vila) is separated from the compound of Formula (Vllb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC). [432] In some embodiments, the electrophile is SO3H, the reaction of the acridan intermediate with SO3H to form a product comprising the acridinium functionalized at the 2,7- position of the acridinium ring includes a sulfonation reaction, and the products are represented by Formulas (Villa) and (Vlllb):
[433] In some embodiments, a method of producing the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb) comprises: dissolving the acridan intermediate in a fourth solution in a container; adding chlorosulfonic acid to the container and stirring for about 5 min to about 30 min; adding water to the container and stirring for about 10 min to about 30 min; and isolating and optionally purifying the product comprising the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb). In some embodiments, the fourth solution comprises acetonitrile.
[434] In some embodiments, if both are formed, the compound of Formula (Villa) is separated from the compound of Formula (Vlllb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
[435] In some embodiments, the electrophile is t-butyl, the reaction of the acridan intermediate with t-butyl to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring includes a Friedel-Craft alkylation reaction, and the products are represented by Formulas (IXa) and (IXb):
[436] In some embodiments, a method of producing the compound of Formula (IXa), (IXb), or both (IXa) and (IXb) comprises: dissolving the acridan intermediate in a fifth solution in a container; adding a Lewis acid to the container and heating at about 40°C to about 60°C for about 18-24 hours; reducing solvent in vacuo followed by the addition of a water/acetonitrile solution to the container; adding an aqueous solution of an oxidant to the container dropwise over time; and isolating and optionally purifying the product comprising the compound of Formula (IXa), (IXb), or both (IXa) and (IXb).
[437] In some embodiments, the fifth solution comprises at least one of t-butyl chloride, t- butyl bromide, t-butyl iodide, an alcohol (e.g., straight-chained or branched Ci-Ce alcohol), or an ether (e.g., Rx-o-Ry, wherein Rx and Ry are independently selected from alkyl or aryl groups). In some embodiments, the Lewis acid includes, but is not limited to, AICI3. In some embodiments, the oxidant comprises FeCh, ZjS-Dichloro-SjS-dicyano-lAbenzoquinone (DDQ), chloranil, or ceric ammonium nitrate (CAN).
[438] In some embodiments, if both are formed, the compound of Formula (IXa) is separated from the compound of Formula (IXb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
[439] In some embodiments, the nitro group(s) of the compound of Formula (Vila) or (Vllb) is reduced to an amine group in the compound of Formula (Xa) or (Xb), respectively:
[440] In some embodiments, a method of producing the compound of Formula (Xa) or (Xb), comprises: dissolving the compound of Formula (Vila) or (Vllb) in a sixth solution in a container; adding a reducing agent to the container and stirring for about 12-24 hours; and isolating and optionally purifying the product comprising the compound of Formula (Xa) or (Xb).
[441] In some embodiments, the sixth solution comprises at least one alcohol, e.g., ethanol.
[442] In some embodiments, if both are formed, the compound of Formula (Xa) is separated from the compound of Formula (Xb). In some embodiments, the separation is effectuated using chromatography, e.g., high-performance liquid chromatography (HPLC).
[443] In some embodiments, a method of functionalizing an acridinium-containing compound with I at a 2- or a 2,7-position, or both, of an acridinium ring comprises: reducing the acridinium ring to an acridan intermediate of Formula (III) using a first solution comprising at least one reducing agent and an acid; dissolving the acridan intermediate of Formula (III) in a third solution; adding iodine monochloride to the dissolved acridan intermediate and stirring for about 30 min to about 1 h; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid.
[444] In some embodiments, a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring using a Suzuki-Mfyaura cross-coupling
reaction is described (V. Pandaras, et al., Tetrahedron Letters, 2013, 54(35), 4712-4716). The generalized Suzuki-Miyaura cross-coupling reaction is:
wherein X is one of Cl, Br, or I. It should be appreciated that although a phenyl group is shown attached to the B(OH)2 group, the phenyl group can be replaced by any aryl or heteroaryl group, as described herein. It should also be appreciated that the B(OH)2 group can be replaced by a boronate ester. In some embodiments, acridinium-containing compounds with at least one or two aryl/heteroaryl groups substituted at the 2 or 2,7-position can be synthesized. Suzuki coupling conditions can be used wherein aryl/heteroaryl-boronic acids are reacted with lodo- CPSP or Dilodo-CPSP, DiEA and a Pd(Ph3)4 catalyst in MeCN/water to give 2- or 2,7- substiuted acridiniums. Aryl and heteroaryl substituents described herein include, but are not limited to, phenyl, 4-hydroxyphenyl, 4-dimethylaminophenyl, 4-cyano-phenyl, 4- aminomethylphenyl, pyrrolyl, furanyl, 5 -cyanothienyl, 5 -methylthienyl, 5-methoxythienyl, 4- methoxythienyl, 3 -methoxythienyl, 5 -methylthiothienyl, thienothiophenyl, bis(5 -methylthienyl), difuranyl, and 3,4-ethylenedioxythiophene. This series yielded interesting results in both the acridinium absorbance/color as well as triggered chemiluminescence. Overall red-shifted chemiluminescence was observed in each system with varying amounts of light output. Notably, a large difference was observed between 5-MeO and 5-Cyano thiophene-CPSP constructs with chemiluminescence emission wavelength maxima ranging from 625 nm to 475 nm (compare, e.g., Figure 30B and Figure 32B).
[445] In some embodiments, a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring comprises: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a boronic acid comprising A’ or a boronate ester comprising A’, wherein A’ is a substituted or unsubstituted aryl group or a substituted or unsubstituted heteroaryl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and
isolating and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
In some embodiments, the compounds of Formula (Xia), (Xlb), (Xllla), (Xlllb), (XlVa), (XlVb), and (XV) are synthesized using this method, wherein Rg and Rg’ are defined herein.
[446] In some embodiments, a method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring with a substituted or unsubstituted pyrrolyl group comprises: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a tert-butyloxycarbonyl (Boc)-protected boronic acid comprising A’ or a Boc-protected boronate ester comprising A’, wherein A’ is a substituted or unsubstituted pyrrolyl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating the product comprising the acridinium-containing compound comprising the Boc-protected pyrrolyl group; dissolving the acridinium-containing compound comprising the Boc-protected pyrrolyl group in dichloromethane and trifluoroacetic acid for about 2-6 hours; removing the solvent in vacuo; and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
In some embodiments, the compounds of Formula (Xlla) and (Xllb) are synthesized using this method, wherein Rg and Rg’ are defined herein.
[447] In some embodiments, the reactant compound comprises iodo-CPSP or diiodo-CPSP.
[448] In some further aspects, isolated, functionalized acridinium-containing compounds, e.g., any of those of Formulas (IV)-(IX), can be further reacted to produce an acridinium sulfide of Formula (I), wherein (i) R’ -S-L3-SO3H and R” is H or (ii) both R’ and R” are -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[449] In some embodiments, a method of producing an acridinium monosulfide of Formula (XVI) comprises: reacting a product of any one of Formulas (IVa)-(IXa) with a reaction mixture
comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium monosulfide product of Formula (XVI):
wherein Y, Ri, R2, Li, L2, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I). [450] In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di-tert-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'- amino- 1,1 '-biphenyl)] palladium (II) methanesulfonate (tBuXPhos Pd G3). In some embodiments, the method further comprises isolating and optionally purifying the acridinium monosulfide product. In some embodiments, the monosulfide moiety of Formula (XVI) is oxidized to a sulfone using, e.g., meta-Chloroperoxybenzoic acid (mCPBA).
[451] In some embodiments, a method of producing an acridinium monosulfide of Formula
(XVI), wherein Y, Ri, R2, Li, L2, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), comprises: reducing an acridinium ring of an acridinium-containing compound to an acridan intermediate of Formula (III) using a first solution comprising at least one reducing agent and an acid; dissolving the acridan intermediate of Formula (III) in a third solution;
adding iodine monochloride to the dissolved acridan intermediate and stirring for about 30 min to about 1 h to produce acridinium functionalized at the 2-position; isolating and optionally purifying the acridinium functionalized at the 2-position; reacting the acridinium functionalized at the 2-position with a reaction mixture comprising a water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium monosulfide product of Formula (XVI). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid. In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di- terf-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1 , 1 '-biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3). In some embodiments, the method further comprises isolating and optionally purifying the acridinium monosulfide product.
[452] In some embodiments, a method of producing an acridinium disulfide of Formula (XVII) comprises: reacting a product of any one of Formulas (IVb)-(IXb) with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium disulfide product of Formula (XVII):
wherein Y, Ri, R2, Li, L2, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I).
[453] In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected ftom: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di-terf-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'- amino- 1,1 '-biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3). In some embodiments, the method further comprises isolating and optionally purifying the acridinium disulfide product.
[454] In some embodiments, a method of producing an acridinium disulfide of Formula (XVII), wherein Y, Ri, R2, Li, L2, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), comprises: reducing an acridinium ring of an acridinium-containing compound to an acridan intermediate of Formula (III) using a first solution comprising at least one reducing agent and an acid; dissolving the acridan intermediate of Formula (III) in a third solution; adding iodine monochloride to the dissolved acridan intermediate and stirring for about 30 min to about 1 h to produce acridinium functionalized at the 2,7-positions; isolating and optionally purifying the acridinium functionalized at the 2,7-positions; reacting the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium disulfide product of Formula (XVII). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid. In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di- tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1 , 1 '-biphenyl)] palladium(II)
methanesulfonate (tBuXPhos Pd G3). In some embodiments, the method further comprises isolating and optionally purifying the acridinium disulfide product.
[455] In some further aspects, a compound of Formula (XVII), e.g., CPSP3, can be oxidized to produce a sulfoxide of either Formula (XVIII) or (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[456] In some embodiments, a method of producing a sulfoxide of Formula (XVIII) or (XIX) comprises: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating and optionally purifying the sulfoxide compound of Formula (XVIII) or (XIX) , wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[457] In some embodiments, the at least one organic solvent includes, but is not limited to, tetrahydrofiiran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF). In some embodiments, the oxidizing agent comprises mCPBA. In some embodiments, the compound of Formula (XVII) is CPSP3.
[458] In some further aspects, isolated, functionalized acridinium-containing compounds, e.g., any of those of Formulas (IV)-(IX), can be further reacted to produce a compound of Formula (I), wherein R’ is -S-L3-SO3H and R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[459] In some embodiments, a method of producing an acridinium sulfone compound of Formula (XXI) comprises: reacting a product of any one of Formulas (IVb)-(IXb), with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an
acridinium monosulfide product of Formula (I), wherein R’ is -S-L3-SO3H and R” is selected from Cl, Br, I, -NO2, t-butyl, or -SO3H; oxidizing the R’ sulfide to a sulfone group; reacting the R” group selected from Cl, Br, I, -NO2, t-butyl, or -SO3H with additional water soluble thiol, in the presence of a Pd-containing catalyst, to produce the acridinium sulfone compound of Formula (XXI):
wherein Y, Ri, R2, Li, L2, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I). [460] In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di-tert-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'- amino- 1,1 '-biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3). In some embodiments, the method further comprises isolating and optionally purifying the acridinium sulfone product. It should be appreciated by the person skilled in the art that the conversion of the product of any one of Formulas (IVb)-(IXb) to the monosulfide is effectuated by controlling the amount of thiol being introduced to the reaction.
[461] In some embodiments, the remaining sulfide moiety of Formula (XXI) is oxidized to a sulfone using, e.g., mCPBA, to yield an acridinium disulfone.
[462] In some embodiments, a method of producing an acridinium sulfone of Formula (XXI), wherein Y, Ri, R2, Li, L2, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), comprises: reducing an acridinium ring of an acridinium-containing compound to an acridan intermediate of Formula (III) using a first solution comprising at least one reducing agent and an acid; dissolving the acridan intermediate of Formula (III) in a third solution; adding iodine monochloride to the dissolved acridan intermediate and stirring for about 30 min to about 1 h to produce acridinium functionalized at the 2,7-positions; isolating and optionally purifying the acridinium functionalized at the 2,7-positions; reacting the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridinium monosulfide product of Formula (I), wherein R’ is -S-L3-SO3H and R” is I; oxidizing the R’ sulfide to a sulfone group; reacting the R” I with additional water soluble thiol, in the presence of a Pd-containing catalyst, to produce the acridinium sulfone compound of Figure (XXI). In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid. In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di-tert-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'- amino- 1,1 '-biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3). In some embodiments, the method further comprises isolating and optionally purifying the acridinium sulfone product. In some embodiments, the remaining sulfide moiety of Formula (XXI) is oxidized to a sulfone using, e.g., mCPBA, to yield an acridinium disulfone.
[463] In some further aspects, a compound of Formula (XVII), e.g., CPSP3, can be oxidized to produce a CPSP3-BisSulfone compound of Formula (XXII), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene,
heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[464] In some embodiments, a method of producing a BisSulfone compound of Formula (XXII) comprises: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; quenching an oxidation reaction with dimethyl sulfide and adding an amount of water; and isolating and optionally purifying the BisSulfone compound of Formula (XIX). wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[465] In some embodiments, the at least one organic solvent includes, but is not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF). In some embodiments, the oxidizing agent comprises mCPBA. In some embodiments, the compound of Formula (XVII) is CPSP3.
4. Acridonc-Containing Compounds and Methods of Synthesizing Same
[466] In some aspects, the present disclosure relates to an acridone compound of Formula (XXIII) comprising:
wherein:
Q is -SO3Z or H;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Li is selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalky 1, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl, and wherein at least one of R’ or R” has to be -S(=O)-L3-X, -S-L3-X, or -SO2-L3-X; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents.
[467] In some embodiments, Q is -SO3Z, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Q is -SO3H. In some embodiments, Q is -SO3Z wherein Z is a Ci -Ci6 group.
[468] In some embodiments, Li is selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Li is selected a C1-C10 alkylene. In some embodiments, Li is — CH2CH2CH2 — .
[469] In some embodiments, Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, Ra is hydrogen. In some embodiments, Rb is hydrogen. In some embodiments, Rc is hydrogen. In some embodiments, Rd is hydrogen. In some embodiments, Re is hydrogen. In some embodiments, Rf is hydrogen.
[470] In some embodiments, R’ and R” are each independently selected from H, Cl, Br, I, - NO2, -NH2, t-butyl, -SO3H, -S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, -NH2, -NHR*, - N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)-N(R*)2, -C=N- R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, -CN, -NC, - CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O-ON2, -N3, - N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, -O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O-C(C=O)- NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, - C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)-
N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl, and wherein at least one of R’ or R” has to be -S(=O)-L3-X, -S-L3-X, or -SO2-L3-X. In some embodiments, R’ and/or R” are H. In some embodiments, R’ and/or R” are Cl. In some embodiments, R’ and/or R” are Br. In some embodiments, R’ and/or R” are I. In some embodiments, R’ and/or R” are -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -S-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R’ is -S-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R” is - S-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R" is -S-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R” is - S-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R’ and/or R” are -(S=O)- L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -(S=O)-L3-SC>3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -(S=O)-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R’ is -(S=O)-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R” is -(S=O)-L3-SC>3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R" is -(S=O)-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R” is -(S=O)-L3-SC>3H, wherein L3 is — CH2CH2CH2 — . In some
embodiments, R’ and/or R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R’ is -SO2-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R’ is -SO2-L3-SO3H, wherein L3 is — CH2CH2CH2 — . In some embodiments, R” is -SO2-L3-SO3H, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents. In some embodiments, R" is -SO2-L3-SO3H, wherein L3 is a C1-C10 alkylene. In some embodiments, R” is -SO2-L3-SO3H, wherein L3 is — CH2CH2CH2 — .
[471] In some embodiments, the acridone compound is a compound of Formula (XXIV):
wherein R’ and R” are defined above for Formula (XXIII).
[472] In some embodiments, the acridone compound is any one of the following compounds:
[474] In some embodiments, the compound of Formula (XXIII) is functionalized at the 2- or 2,7 positions and is selected from:
[475] For compounds described herein, groups and substituents thereof may be selected in accordance with permitted valence of the atoms and the substituents, such that the selections and substitutions result in a stable compound, e.g., which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc.
[476] The compounds described herein may exist as stereoisomers wherein asymmetric or chiral centers are present. The stereoisomers are “R” or “S” depending on the configuration of substituents around the chiral carbon atom. The disclosure contemplates various stereoisomers and mixtures thereof, and these are specifically included within the scope of this disclosure. Stereoisomers include enantiomers and diastereomers and mixtures of enantiomers or diastereomers. Individual stereoisomers of the compounds may be prepared synthetically from commercially available starting materials, which contain asymmetric or chiral centers or by preparation of racemic mixtures followed by methods of resolution well-known to those of ordinary skill in the art. These methods of resolution are exemplified by (1) attachment of a mixture of enantiomers to a chiral auxiliary, separation of the resulting mixture of diastereomers
by recrystallization or chromatography, and optional liberation of the optically pure product from the auxiliary as described in Fumiss, Hannaford, Smith, and Tatchell, “Vogel’s Textbook of Practical Organic Chemistry”, 5* edition (1989), Longman Scientific & Technical, Essex CM20 2 JE, England, or (2) direct separation of the mixture of optical enantiomers on chiral chromatographic columns, or (3) fractional recrystallization methods.
[477] It should be understood that the compounds may possess tautomeric forms as well as geometric isomers, and that these also constitute an aspect of the invention,
[478] The present disclosure also includes isotopically-labeled compounds, which are identical to those recited in Formula (XXIII), but for the fact that one or more atoms are replaced by an atom having an atomic mass or mass number different from the atomic mass or mass number usually found in nature. Examples of isotopes suitable for inclusion in the compounds of the invention are hydrogen, carbon, nitrogen, oxygen, phosphorus, sulfur, fluorine, and chlorine, such as, but not limited to, 2H, 3H, l3C, UC, t5N, 1SO, ;70, 3 iP, 32P, 33S, 1SF, and 36C1, respectively. Substitution with heavier isotopes such as deuterium, i.e., 2H, can afford certain advantages resulting from greater metabolic stability, for example increased in vivo half-life, and may therefore be preferred in some circumstances. The compound may incorporate positronemitting isotopes for medical imaging and positron-emitting tomography (PET) studies for determining the distribution of receptors. Suitable positron-emitting isotopes that can be incorporated in compounds of Formula (I) are
13N, ’: 5O, and '8F. Isotopically-labeled compounds of Formula (XXIII) can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described in the accompanying examples using appropriate isotopically-labeled reagent in place of non-isotopicaily-labeied reagent.
[479] A compound disclosed herein may be in the form of a salt. The salts may be prepared during the final isolation and purification of the compounds or separately, for example byreacting a basic group of the compound (e.g., an amino group) with a suitable acid or by reacting an acidic group of the compound (e.g., a carboxylic acid group) with a suitable base. Acid salts may be prepared during the final isolation and purification of the compounds or separately by reacting a suitable group of the compound, such as an amino group, with a suitable acid. For example, a compound may be dissolved in a suitable solvent, such as but not limited to methanol and water, and treated with at least one equivalent of an acid, such hydrochloric acid. The
resulting salt may precipitate cut and be isolated by filtration and dried under reduced pressure. Alternatively, the solvent and excess acid may be removed under reduced pressure to provide a salt. Representative salts include acetate, adipate, alginate, citrate, aspartate, benzoate, benzenesulfonate, bisulfate, butyrate, camphorate, eamphorsulfonate, digluconate, glycerophosphate, hemisuliate, heptanoate, hexanoate, formate, isethionate, fumarate, lactate, maleate, methanesulfonate, naphthylenesulfonate, nicotinate, oxalate, pamoate, pectinate, persulfate, 3 -phenylpropionate, picrate, oxalate, maleate, pivalate, propionate, succinate, tartrate, trichloroacetate, trifluoroacetate, glutamate, para-toluenesulfonate, undecanoate, hydrochloric, hydrobromic, sulfuric, phosphoric and the like. The amino groups of the compounds may also be quatemized with alkyd chlorides, bromides and iodides such as methyl, ethyl, propyl, isopropyl, butyl, lauryl, myristyl, stearyl and the like. Basic addition salts may be prepared during the final isolation and purification of the disclosed compounds by reaction of a carboxyl group with a suitable base such as the hydroxide, carbonate, or bicarbonate of a metal cation such as lithium, sodium, potassium, calcium, magnesium, or aluminum, or an organic primary, secondary, or tertiary amine. Quaternary amine salts can be prepared, such as those derived from methylamine, dimethylamine, trimethylamine, triethylamine, diethylamine, ethylamine, tributylamine, pyridine, N,N-dimethylaniline, N-methylpiperidine, N-methylmorpholine, dicyclohexylamine, procaine, dibenzylamine, N,N-dibenzylphenethylamine, 1-ephenamine and N,N'- dibenzylethylenediamine, ethylenediamine, ethanolamine, diethanolamine, piperidine, piperazine, and the like.
[480] In some other aspects, the present disclosure relates to methods of synthesizing the compounds of Formula (XXIII). In some embodiments, the compounds of Formula (XXIII) are readily prepared by oxidizing an acridinium-containing compound of Formula (I) in an alkaline solution. For example, an acridinium-containing compound of any of Formulas (XVI)-(XXII) or compounds (24)-(32) can be oxidized to produce an acridone of Formulas (XXV)-(XXXII) or compounds (36)-(44), respectively. In some embodiments, the oxidizing agent comprises H2O2.
[481] In some other aspects, a method of functionalizing an acridone-containing compound at a 2,7-position of an acridone ring with iodine is disclosed. In some embodiments, a method of functionalizing an acridone-containing compound at a 2,7-position of an acridone ring with iodine comprises: dissolving an acridone-containing compound in a third solution;
adding iodine monochloride to the third solution and stirring for about 6 h to about 18 h under pressure to produce the acridone-containing compound functionalized at the 2,7-positions in a mixture; dissolving the mixture in DMSO; and isolating and optionally purifying the diiodo-acridone compound.
In some embodiments, the third solution comprises an organic solvent such as dichloromethane and optionally acetic acid. In some embodiments, the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H. In some embodiments, the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H and Li is — CH2CH2CH2 — . In some embodiments, the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H, Li is — CH2CH2CH2— , and Q is -SO3H.
[482] In some other aspects, a method of producing an acridone disulfide of Formula (XXVI) comprises: reacting a dihalo-acridone, e.g., a diiodo-acridone of compound (35), with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridone disulfide product of Formula (XXVI):
wherein Li, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (XXIII). [483] In some embodiments, the water soluble thiol includes, but is not limited to, mercaptopropanesulfonic acid (MSA). In some embodiments, the reaction mixture further comprises at least one species selected from: at least one alkyl amine including, but not limited to, N,N-diisopropylethylamine (DIEA); at least one base; at least one organic solvent including, but not limited to, tetrahydrofiiran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF); and water. In some embodiments, the Pd-containing catalyst includes, but is not limited to, 2-Di-tert-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'- amino-l,l'-biphenyl)] palladium (II) methanesulfonate (tBuXPhos Pd G3). In some
embodiments, the method further comprises isolating and optionally purifying the acridone monosulfide product.
[484] In some further aspects, a compound of Formula (XXVI) can be oxidized to produce a sulfoxide of Formula (XXVII), a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[485] In some embodiments, a method of producing a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI), comprises: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating the bis sulfoxide of Formula (XXVIII), sulfoxide sulfone of Formula (XXIX), and/or bis sulfone of Formula (XXXI) and optionally separating and purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[486] In some embodiments, the at least one organic solvent includes, but is not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF). In some embodiments, the oxidizing agent comprises mCPBA. In some embodiments, the compound of Formula (XXVI) is compound (39). In some embodiments, after addition of the oxidizing agent, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
[487] In some embodiments, a method of producing a sulfoxide of Formula (XXVII) comprises: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; dropwise adding an eighth solution comprising an oxidizing agent to the contents of the container; stirring the contents of the container;
quenching the reaction with dimethyl sulfide; and isolating the sulfoxide of Formula (XXVII) and optionally purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
[488] In some embodiments, the at least one organic solvent includes, but is not limited to, tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF). In some embodiments, the oxidizing agent comprises mCPBA. In some embodiments, the eighth solution further comprises at least one organic solvent, e.g., DMF. In some embodiments, the compound of Formula (XXVI) is compound (39). In some embodiments, after addition of the eighth solution, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes. In some embodiments, the dimethyl sulfide is added dropwise. In some embodiments, immediately after the addition of dimethyl sulfide, a bolus of water is added.
5. Methods of Using the acridinium-containing and acridone-containing compounds
[489] Advantageously, the acridinium sulfides, sulfoxides, and sulfones disclosed herein in Formulas (XVI)-(XXII) and the acridone sulfides, sulfoxides, and sulfones disclosed herein in Formulas (XXV)-(XXXI) have a substantially bright chemiluminescent (CL) or fluorescence emission (depending on the alkyl-sulfide substituent), can be highly water soluble and form stable protein conjugates.
[490] Moreover, the number of sulfide substituents and their oxidation state affect the amount of red-shift in the CL emission, allowing for control over emission wavelength and better pairing with blue-shifted acridinium compounds. Acridinium-containing compounds with red shifted CL continue to be of interest in multicolor CL applications including assay multiplexing, extending dynamic range, and removing hook-effect in assay measurements. Multicolor CL assays can be achieved by combining acridinium molecules with spectrally resolved CL emission profiles and photomultiplier tube (PMT) detectors with color matching filters. Multiplex assays can be used to measure two or more analytes from a patient sample in a single test, thereby increasing testing throughput, reduced time per result, and fewer consumables. Other applications of multicolor CL involve extending the dynamic range of an assay, reducing the
need for retest, and addressing the hook affect. Customers using multiplexed in vitro diagnostic technology could benefit from reduced reagent costs and increase the information content and accuracy of their test result.
[491] In some aspects, compounds of Formula (I) or Formula (XXIII) can be used in assays, such as in, for example, duplex assays, including those described herein in Section 2.
Specifically, in some aspects, the compounds of Formula (I) or Formula (XXIII) can be used in methods for detecting the presence of or determining the concentration or amount one or more analytes of interest in a biological sample obtained from a subject. Detecting the presence of or determining the concentration or amount of an analyte of interest in a biological sample can be used for detecting, identifying and/or monitoring a disease, condition, or illness or monitoring the response of a particular disease, condition, or illness to treatment with a therapeutic (e.g., small or large molecule). For example, the compounds of Formula (I) or Formula (XXIII) can be used to assess the stage and/or monitor the stage or phase of chronic Hepatitis B (HBV) infection or monitoring response to a treatment for chronic HBV. Such a method can involve the steps of: a) performing an assay to detect the presence or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject diagnosed with chronic HBV or receiving a treatment for chronic HBV, where the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody -P-HBcAg-second detection antibody complex is formed; and b) assessing and monitoring stage or phase of chronic HBV infection or monitoring in response to the treatment for chronic HBV based on the presence or level of HBcAg and P- HBcAg in the at least one sample.
[492] By way of another example, the compounds of Formula (I) or Formula (XXIII) can be used to detect the presence of or the level of HBV in a sample obtained from a subject. Such a method can involve the steps of:
a) performing an assay to detect the presence of or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject suspect of having HBV, where the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg- first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg-second detection antibody complex is formed; and b) detecting HBV infection based on the presence or level of HBcAg and P-HBcAg in the at least one sample.
[493] In the above methods, the first detectable label and second detectable label can each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
wherein:
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SOa-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyL heteroarylalkyl, cycloalkylalkyl, and heterocycly lalkyi ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C-N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene,
alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
6. Samples
[494] As used herein, “sample”, “test sample”, “biological sample” refer to fluid sample containing or suspected of containing an analyte of interest. The sample may be derived from any suitable source. In some cases, the sample may comprise a liquid, fluent particulate solid, or fluid suspension of solid particles. In some cases, the sample may be processed prior to the analysis described herein. For example, the sample may be separated or purified from its source prior to analysis; however, in certain embodiments, an unprocessed sample containing an analyte of interest may be assayed directly. In one example, the source containing an analyte of interest is a human bodily substance (e.g., bodily fluid, blood such as whole blood, serum, plasma, urine, saliva, sweat, sputum, semen, mucus, lacrimal fluid, lymph fluid, amniotic fluid, interstitial fluid, lung lavage, cerebrospinal fluid, feces, tissue, organ, or the like). Tissues may include, but are not limited to skeletal muscle tissue, liver tissue, lung tissue, kidney tissue, myocardial tissue, brain tissue, bone marrow, cervix tissue, skin, etc. The sample may be a liquid sample or a liquid extract of a solid sample. In certain cases, the source of the sample may be an organ or tissue, such as a biopsy sample, which may be solubilized by tissue disintegration/cell lysis.
[495] A wide range of volumes of the fluid sample may be analyzed. In a few exemplary embodiments, the sample volume may be about 0.5 nL, about 1 nL, about 3 nL, about 0.01 pL, about 0.1 pL, about 1 pL, about 5 pL, about 10 pL, about 100 pL, about 1 mL, about 5 mL, about 10 mL, or the like. In some cases, the volume of the fluid sample is between about 0.01 pL and about 10 mL, between about 0.01 pL and about 1 mL, between about 0.01 pL and about 100 pL, or between about 0.1 pL and about 10 pL.
[496] In some cases, the fluid sample may be diluted prior to use in an assay. For example, in embodiments where the source containing an analyte of interest is a human body fluid (e.g., blood, serum), the fluid may be diluted with an appropriate solvent (e.g., a buffer such as PBS buffer). A fluid sample may be diluted about 1-fold, about 2-fold, about 3 -fold, about 4-fold,
about 5 -fold, about 6-fold, about 10-fold, about 100-fold, or greater, prior to use. In other cases, the fluid sample is not diluted prior to use in an assay.
[497] In some cases, the sample may undergo pre-analytical processing. Pre-analytical processing may offer additional functionality such as nonspecific protein removal and/or effective yet cheaply implementable mixing functionality. General methods of pre-analytical processing may include the use of electrokinetic trapping, AC electrokinetics, surface acoustic waves, isotachophoresis, dielectrophoresis, electrophoresis, or other pre-concentration techniques known in the art. In some cases, the fluid sample may be concentrated prior to use in an assay. For example, in embodiments where the source containing an analyte of interest is a human body fluid (e.g., blood, serum), the fluid may be concentrated by precipitation, evaporation, filtration, centrifugation, or a combination thereof. A fluid sample may be concentrated about 1-fold, about 2-fold, about 3-fold, about 4-fold, about 5-fold, about 6-fold, about 10-fold, about 100-fold, or greater, prior to use.
[498] It may be desirable to include a control. The control may be analyzed concurrently with the sample from the subject as described above. The results obtained from the subject sample can be compared to the results obtained from the control sample. Standard curves may be provided, with which assay results for the sample may be compared. Such standard curves present levels of marker as a function of assay units, i.e., fluorescent signal intensity, if a fluorescent label is used. Using samples taken from multiple donors, standard curves can be provided for reference levels of an analyte of interest in normal healthy tissue, as well as for “at- risk” levels of the analyte of interest in tissue taken from donors, who may have one or more of the characteristics set forth above.
[499] Thus, in view of the above, a method for determining the presence, amount, or concentration of an analyte of interest in a test sample is provided. The method comprises assaying the test sample for an analyte of interest by an immunoassay, for example, employing at least one capture antibody that binds to an epitope on an analyte of interest and at least one detection antibody that binds to an epitope on an analyte of interest which is different from the epitope for the capture antibody and optionally includes a detectable label, and comprising comparing a signal generated by the detectable label as a direct or indirect indication of the presence, amount or concentration of an analyte of interest in the test sample to a signal generated as a direct or indirect indication of the presence, amount or concentration of an analyte
of interest in a calibrator. The calibrator is optionally, and in some embodiments, is part of a series of calibrators in which each of the calibrators differs from the other calibrators in the series by the concentration of the analyte of interest.
7. Kits and Systems
[500] In some embodiments, the present disclosure further provides kits and systems for detecting the presence, level, or status of an analyte of interest in a sample. In some embodiments, the kits or systems find use in multiplex and/or automated analysis methods. Exemplary reagents include, but are not limited to, antigens, antibodies, colorimetric reagents, enzymes, buffers, etc. Optionally, the kit can also contain at least one calibrator or control. Any calibrator or control can be included in the kit.
[501] Thus, the present disclosure further provides for diagnostic and quality control kits comprising one or more antibodies or other detection reagents. Optionally the assays, kits and kit components of the disclosure are optimized for use on commercial platforms (e.g., immunoassays on the PRISM®, AxSYM®, ARCHITECT®, Alinity, and EIA (Bead) platforms of Abbott Laboratories, Abbott Park, IL, as well as other commercial and/or in vitro diagnostic assays).
[502] Optionally the kits include quality control reagents (for example, sensitivity panels, calibrators, and positive controls). Preparation of quality control reagents is well known in the art, and is described, e.g., on a variety of immunodiagnostic or nucleic acid product insert sheets.
[503] In another embodiment, the present disclosure provides for a quality control kit comprising one or more antibodies described herein for use as a sensitivity panel to evaluate assay performance characteristics and/or to quantitate and monitor the integrity of the antigen(s) or nucleic acids used in the assay.
[504] The kits can optionally include other reagents required to conduct a diagnostic assay or facilitate quality control evaluations, such as buffers, salts, enzymes, enzyme co-factors, substrates, detection reagents, and the like. Other components, such as buffers and solutions for the isolation and/or treatment of a test sample (e.g., pretreatment reagents), may also be included in the kit. The kit may additionally include one or more other controls. One or more of the components of the kit may be lyophilized and the kit may further comprise reagents suitable for the reconstitution of the lyophilized components.
[505] The various components of the kit optionally are provided in suitable containers. As indicated above, one or more of the containers may be a microtiter plate. The kit further can include containers for holding or storing a sample (e.g., a container or cartridge for a blood or urine sample). Where appropriate, the kit may also optionally contain reaction vessels, mixing vessels and other components that facilitate the preparation of reagents or the test sample. The kit may also include one or more instruments for assisting with obtaining a test sample, such as a syringe, pipette, forceps, measured spoon, or the like.
[506] The kit further can optionally include instructions for use, which may be provided in paper form or in computer-readable form, such as a disc, CD, DVD or the like.
[507] The disclosure as described herein also can be adapted for use in a variety of automated and semi-automated systems (including those wherein the solid phase comprises a microparticle), as described, e.g., in U.S. Patent Nos. 5,089,424 and 5,006,309, and as, e.g., commercially marketed by Abbott Laboratories (Abbott Park, IL) including but not limited to Abbott’s ARCHITECT®, Alinity, AxSYM®, IMX, PRISM®, and Quantum II instruments, as well as other platforms.
[508] All patents and publications mentioned in the specification are indicative of the levels of those skilled in the art to which the disclosure pertains. All patents and publications are herein incorporated by reference to the same extent as if each individual publication was specifically and individually indicated to be incorporated by reference.
[509] The disclosure illustratively described herein suitably may be practiced in the absence of any element or elements, limitation or limitations which is not specifically disclosed herein. Thus, for example, in each instance herein any of the terms “comprising,” “consisting essentially of’ and “consisting of’ may be replaced with either of the other two terms. The terms and expressions which have been employed are used as terms of description and not of limitation, and there is no intention that in the use of such terms and expressions of excluding any equivalents of the features shown and described or portions thereof, but it is recognized that various modifications are possible within the scope of the disclosure claimed. Thus, it should be understood that although the present disclosure includes various embodiments and optional features, modification and variation of the concepts herein disclosed may be resorted to by those skilled in the art, and that such modifications and variations are considered to be within the scope of this disclosure as defined by the appended claims.
EXAMPLES
Example 1: A one step dual channel combo assay run on Dual-Channel Detection System to Extend Assay Dynamic Range
List of Reagents:
[510] Microparticles: from bHCG ARCHITECT® assay kit (Abbott Laboratories, Abbott Park, IL).
[511] Analyte: recombinant bHCG from Scripps Laboratories (San Diego, CA)
[512] Competing analyte: recombinant bHCG labeled with CPSP (blue color).
[513] Conjugate: anti-bHCG antibody (Ab) labeled with CPSP3 (green color)
Instrument:
[514] Dual Channel system: The system is equipped with 2 detectors (PMTs) mounted on the opposite side of the reaction vessel. There is a blue or green filter placed on each detector to allow signal separation.
Methods:
[515] Generating a calibration curve for bHCG: 30jiL of 0.1% bHCG Ab coated microparticles mixed with 60pL bHCG (with known concentration ranging from (0, 10, 250, 1000, 7500, 15000 mIU/mL) and 50pL 0.05ng/mL bHCG-CPSP (competing analyte) and 3nM Ab-CPSP3 (conjugate) mixture, and incubate for 11 minutes, washed, and measure the signal in a dual channel system (See, FIG. 1). For proof of principle, 6 patient samples were tested and calculated the concentration of their bHCG levels using the calibration curve generated with 4PLC curve fit. Table A, below, lists the measured signal on a dual channel detection system for bHCG calibrators and patient samples.
[516] Dose response curve of bHCG with wide concentration range: 25pL of 0.1% bHCG Ab coated microparticles mixed with 15pL bHCG (various concentration) and 50pL 0.05ng/mL bHCG-CPSP (competing analyte) and 3nM Ab-CPSP3 (conjugate) mixture, and incubate for 11 minutes, washed, and measure the signal in a dual channel system (See, FIG. 1)
Table A
[517] As shown in FIG. 2A-2C, the signal generated in the blue channel reflects the bHCG assay in competitive format (FIG. 2 A), which is not sensitive to low concentration of bHCG; Where the signal generated in the green channel reflect the bHCG assay in a one-step sandwich assay format (FIG. 2B), it has great sensitivity at low concentration of bHCG, but prone for hook effect. On the other hand, if the signals from both channels are considered and the signal ratio of the green channel and the blue channel calculated, the high sensitivity from the green channel and wide assay range from the blue channel can be taken advantage of, thereby achieving both high sensitivity, wide dynamic range, and reduction in hook effect (See, FIG. 2C).
[518] As shown in FIG 2D, with decreased sample and microparticle volumes, we can further extend the assay dynamic range using the proposed dual channel assay format to 450,000 mIU/mL, which is 30 times of the current on market assay (15,000m lU/mL). Current on market bHCG assay dynamic range is 0-15,000 mIU/mL. Based on the clinical data, 7% of the patients would test out of range and thus require a retest with a 15X dilution. Using the new method described in the invention, 99.8% of the patient samples would not require any retest (See, Table B).
Example 2: A two-step dual channel combo assay run on Dual-Channel System to Extend Assay Dynamic Range
List of Reagents:
[519] Microparticles: microparticles are coated with Receptor-Binding Domain (RBD) (GenScript, Piscataway, NJ, USA) of the spike protein from SARS-CoV-2.
[520] Analyte: anti-RBD antibody from CO VID- 19 patient sample or vaccinated patient sample.
[521] Competing analyte: a) polyclonal anti-RBD antibody produced in Rabbit (Invitrogen, Waltham, MA, USA), b) goat anti-rabbit antibody (Ab) (Jackson ImmunoResearch, West Grove, PA, USA) labeled with CPSP-Lucifer Yellow (green color)
[522] Conjugate: Goat-anti-human IgG (Jackson ImmunoResearch, West Grove, PA, USA) labeled with CPSP (blue color)
Methods:
[523] Dual Channel: 25 pL of 0.1% RBD coated microparticles + 50pL calibrators or patient sample + 50pL (5 nM) Rabbit anti-RBD polyclonal antibody were incubated for 18 minutes and washed.
[524] 50pL of anti-Hu IgG Ab-CPSP (10 ng/mL) and 50pL anti-Rabbit IgG Ab-CPSP-LY (1.3 nM) were added, incubated for 4 minutes, washed and measured (See, FIG. 3).
Results:
[525] Table C lists the signal response measured in the Competitive and Sandwich (dual) Channels, and the calculated signal ratio for a serial diluted patient sample.
Table C
[526] As shown in FIG. 4A-4C, the signal from the competitive channel (FIG. 4A) is quite flat at low analyte concentration, which limits its sensitivity. In contrast, the signal from the sandwich channel (FIG. 4B) plateaus at high analyte concentration, which limits dynamic range. Therefore, the results confirm that combining the sandwich assay and competitive immunoassay can greatly expand the assay dynamic range without sacrificing assay’s sensitivity (see, FIG. 4C). With a set of well-defined calibrators, the described assay can be a quantitative COVID-19 assay for both disease detection and vaccinated population monitoring without dilution.
Example 3: Preparation of an Acridan Intermediate
[527] In some embodiments, methods of functionalizing chemiluminescent acridiniums at the 2 and 2,7-position of the acridinium ring begin with a H+/2e reduction of an acridinium ring to an acridan intermediate using a metal, e.g., zinc, and weakly acidic solution (e.g., as shown in Scheme 1). Acridans are electron rich and capable of selectively reacting with electrophiles (X+) at the 2 and 7 positions of the acridinium ring. In some embodiments, the electrophiles can include, but are not limited to, Cl, Br, I, NO2, SO3H, and t-butyl. Electrophiles are generated from classical electrophilic aromatic substitution reaction conditions.
Exact Mass: 586.14
CPSP-Acridinium CPSP-Acridan
(45)
[528] 500 mg of carboxypropylsulfopropyl-acridinium (CPSP-acridinium, 9-[N-tosyl-N-(3- carboxypropyl)]-10-(3-sulfopropyl)acridinium carboxamide, also known as CPSP herein) and 1000 mg of NH4CI were dissolved in 40 ml of ethanol and 5 mL of water by heating at 70°C with stirring (~15 mins) (see, Scheme 2). 1000 mg of Zn(0) was added and the mixture stirred for 20 mins until the solution became colorless. The reaction mixture was filtered through a Buchner funnel and the filtrate volume reduced under a Ni(g) stream. The reaction mixture was injected on a 100g Cl 8 Aq flash column and eluted using 100% water to 60% acetonitrile (MeCN or ACN) gradient. Fractions containing the CPSP-acridan product were collected and lyophilized to dryness. Yield - 450 mg white powder.
MS (ESI): m/z 587 (M+H)+
NMR (500 MHz, MeOD) 5 - 7.74 (d, J = 8.35 Hz, 2H), 7.37 (d, J = 8.00, 2H), 7.23 (m, 2H), 7.12 (d, J = 8.60 Hz, 2H), 6.90 (dd, J = 1.05, 2H), 6.77 (dt, J = 0.77, 7.36 Hz, 2H), 5.82 (s, 1H), 4.06 (t, J = 8.58 Hz, 2H) 3.86 (t, J = 7.45 Hz, 2H), 2.97 (t, J = 7.45 Hz, 2H), 2.43 (s, 3H), 2.21- 2.11 (m, 4H), 1.68 (p, J = 7.31 Hz, 2H)
CPSP-Acridan MonoChloro-CPSP DiChloro-CPSP
[45] (1) (2)
[529] In a 20 mL reaction vial equipped with a stir bar, 25 mg of CPSP-acridan was mixed with 30 mg NaCl, 12 mg N-chlorosuccinimide, 18 mg p-toluenesulfonic acid, 1 mL of water, and 1 mL of acetonitrile (see, Scheme 3). The reaction was stirred for 18 hours. Next, 120 mg of FeC13 was dissolved in 3 mL of water and the solution was added portion-wise over the course of 1 hour. To the vial, ~6g of Diaion HP20ss resin was added until all the liquid was absorbed. The solid resin was transferred to a Buchner funnel and washed with 25 mL of water. The aqueous filtrate was discarded, and the resin washed with 25 mL MeCN. The reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% trifluoroacetic acid (TFA) Yield of 2- Monochloro-CPSP was 3.5 mg. Yield of 2,7-Dichloro-CPSP was 8.9 mg.
MonoChloro-CPSP MS (ESI): m/z 619.30 (M+)
H NMR (500 MHz, MeOD and 3% ND4OD) 8 - 7.84 (d, J = 8.20 Hz, 2H), 7.47 (d, J = 8.05, 2H), 7.36 (m, 4H), 6.93 (t, J = 7.48 Hz, 1H), 6.84 (d, J = 7.50 Hz, 1H), 6.54 (s, 1H), 4.22 (t, J = 8.05 Hz, 2H), 3.24 (m, 2H), 3.07 (t, J = 6.63, 2H), 2.50 (s, 3H), 2.28 (p, J = 7.00 Hz, 2H), 1.47 (p, J = 8.78 Hz, 2H), 1.28 (bs, 2H)
DiChloro-CPSP MS (ESI): m/z 653.38 (M+)
JH NMR (500 MHz, MeOD and 3% ND4OD) 5 - 7.84 (d, J = 8.35 Hz, 2H), 7.47 (d, J = 8.20, 2H), 7.34 (m, 4H), 6.68 (d, J = 2.00 Hz, 2H), 4.23 (t, J = 8.40 Hz, 2H), 3.29 (t, J = 7.90 Hz, 2H), 3.05 (t, J = 6.60, 2H), 2.49 (s, 3H), 2.25 (p, J = 8.05 2H), 1.52 (t, J = 7.58 Hz, 2H), 1.23 (p, J = 7.75Hz, 2H)
CPSP-Acridan DiBromo-CPSP
[45] (4)
[530] In a 20 mL reaction vial equipped with a stir bar, 25 mg of CPSP-acridan was dissolved in 2.5 mL of dichloromethane (DCM) and 0.25 mL of acetic acid. To this was added 0.02 mL of bromine and the solution was stirred for 45 minutes (see, Scheme 4). The solvent was removed and the solid was triturated with 5 mL of 1 : 1 DCM-methyl t-butyl ether (MTBE). The solid was collected by vacuum filtration and washed with an additional 5 mL of MTBE. No further purification was necessary. Yield was 30 mg.
MS (ESI): m/z 743.17 (M+)
H NMR (500 MHz, d6-DMSO and 3% ND4OD) 8 - 7.71 (d, J = 8.30 Hz, 2H), 7.51 (d, J = 8.05, 2H), 7.44 (dd, J = 2.40, 9.00 Hz, 2H), 7.24 (d, J = 9.15 Hz, 2H), 6.70 (d, J = 2.30 Hz, 2H), 4.06 (t, J = 8.28 Hz, 2H), 3.16 (t, J = 8.08 Hz, 2H), 2.74 (t, J = 6.73, 2H), 2.46 (s, 3H), 1.99 (m, 2H), 1.27 (t, J = 7.43 Hz, 2H), 0.98 (m, 2H)
CPSP-Acridan Monolodo-CPSP Diiodo-CPSP
[45] (5) (6)
[531] In a 20 mL reaction vial equipped with a stir bar, 25 mg of CPSP-acridan was dissolved in 2.5 mL of DCM and 0.25 mL of acetic acid. To this was added 0.02 mL of iodine monochloride (IC1) and the solution was stirred for 45 minutes (see, Scheme 5). The solvent was removed in vacuo and the crude material was dissolved in 1 mL of MeCN and 1 mL water. The reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Yield of Monoiodo-CPSP was 9 mg. Yield of Diiodo-CPSP was 5.2 mg.
Monolodo-CPSP MS (ESI): m/z 711.35 (M+)
*H NMR (500 MHz, MeOD and 3% ND4OD) 5 - 7.84 (d, J = 8.30 Hz, 2H), 7.65 (dd, J = 2.15, 8.90, 1H), 7.51 (d, J = 8.00 Hz, 4H), 7.40 (m, 1H), 7.31 (d, J = 8.40 Hz, 1H), 7.13 (d, J = 9.00 Hz, 1H), 6.94 (t, 7.35 Hz, 1H), 6.87 (dd, J = 1.43, 7.68 Hz, 1H), 6.81 (d, J = 2.10 Hz, 1H), 4.21
(t, J = 8.43 Hz, 2H), 3.25 (m, 2H), 3.07 (t, J = 6.78, 2H), 2.50 (s, 3H), 2.27 (p, J = 7.91 Hz, 2H),
1.48 (m, 2H), 1.31 (m, 1H), 1.02 (m, 1H)
Dilodo-CPSP MS (ESI): m/z 837.31 (M+)
H NMR (500 MHz, MeOD and 3% ND4OD) 8 - 7.85 (d, J = 8.15 Hz, 2H), 7.66 (d, J = 7.50 Hz, 2H), 7.53 (d, J - 8.34 Hz, 2H) 7.15 (d, 8.90 Hz, 2H), 6.98 (s, 2H), 4.20 (t, 8.15 Hz, 2H), 3.30 (m, 2H), 3.05 (t, J = 6..40 Hz, 2H) 2.50 (s, 3H), 2.25 (m, 2H), 1.52 (t, J = 7.30 Hz, 2H), 1.22 (m, 2H)
CPSP-Acridan DiNitro-CPSP
[45] (8)
[532] In a 20 mL reaction vial equipped with a stir bar, 25 mg of CPSP-acridan was dissolved in 2.5 mL of DCM. To this was added 100 pL of concentrated nitric acid (see, Scheme 6). The solvent was reduced under nitrogen before adding 5 mL of water and 5 mL of acetonitrile. The reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Yield was 8 mg.
CPSP-Acridan Mono-t-butyl-CPSP Di-t-butyl-CPSP
[45] (9) (10)
[533] In a 20 mL reaction vial equipped with a stir bar, 25 mg of CPSP-acridan was mixed with 3 mL of 2-chloro-2-methyl-propane. To this was added 145 mg of AlCh and the reaction was heated to 50°C for 18-24 hours (see, Scheme 7). The solvent was reduced in vacuo and 2 mL of 1 : 1 water/MeCN was added. Next, 30 mg of FeCh was dissolved in 1 mL of water and the solution was added dropwise over 1 minute resulting in a biphasic mixture. To the vial, ~6g of Diaion HP20ss resin was added until all of the liquid was absorbed. The solid resin was transferred to a Buchner funnel and washed with 25 mL of water. The aqueous filtrate was discarded, and the resin washed with 25 mL MeCN. The reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA Yield of Di-t-butyl-CPSP was 6.8 mg. Yield of Mono-t-butyl- CPSP was 3.5 mg.
Mono-t-butyl-CPSP MS (ESI): m/z 641.60 (M+)
NMR (500 MHz, MeOD and 3% ND4OD) 5 - 7.92 (d, J = 8.35 Hz, 2H), 7.47-7.41 (m, 3H), 7.37 (m, 1H), 7.28 (d, J = 8.40 Hz, 1H), 7.25 (d, J= 8.90 Hz, 1H), 6.86 (t, J = 7.38 Hz, 1H), 6.82 (d, 2.30 Hz, 1H), 6.75 (dd, J = 1.40, 7.65 Hz, 1H), 4.24 (t, J = 8.30 Hz, 2H), 3.25 (m, 2H), 3.07 (t, J = 6.83 Hz, 2H), 2.49 (s, 3H), 2.29 (m, 2H), 1.42 (m, 2H), 1.19 (s, 9H), 1.02 (m, 2H)
Di-t-butyl-CPSP MS (ESI): 697.54 (M+)
NMR (500 MHz, MeOD and 3% ND4OD) 6 - 7.97 (d, J = 8.35 Hz, 2H), 7.44-7.40 (m, 4H), 7.22 (d, J = 8.90 Hz, 2H), 6.85 (d, J = 2.30 Hz), 4.22 (t, J = 8.23 Hz, 2H), 3.06 (t, J = 6.83 Hz, 2H), 2.48 (s, 3H), 2.28 (m, 2H), 1.41 (m, 2H) 1.20 (s, 18H), 1.06 (m, 2H)
CPSP-Acridan DiSulfonate-CPSP
[45] (12)
[534] In a 20 mL reaction vial equipped with a stir bar, 58 mg of CPSP-acridan was dissolved in 6 mL of MeCN. To this was added 60pL of chlorosulfonic acid and the reaction was stirred for 15 minutes before adding 3 mL of water (see, Scheme 8). Another 20 minutes of stirring was continued before reducing the solvent by rotovap. The crude disulfonate-acridan was purified over 30g C18Aq flash column using. Fractions containing the product were lyophilized. Next, 10 mg of the purified acridan was then dissolved in 1.5 mL methanol 0.5 mL water before adding 8 mg of 2,3-Dichloro-5,6-dicyano-l,4-benzoquinone (DDQ). The reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Yield was 6.5 mg.
MS (ESI): m/z 743.51 (M-H)-
NMR (500 MHz, d6-DMSO and 3% ND4OD) 8 - 7.86 (d, J = 8.05, 2H), 7.80 (d, J = 8.00 Hz, 2H), 7.54 (d, J = 7.95 Hz, 2H), 7.43 (d, J = 8.85 Hz, 2H) 7.35 (s, 2H), 4.32 (t, J = 7.85 Hz, 2H), 3.34 (t, J = 7.65 Hz, 2H), 3.10 (t, J = 6.33 Hz, 2H), 2.49 (s, 3H), 2.23 (m, 2H), 1.54 (m, 1H), 1.21 (m, 1H)
Example 10: Summary of the Spectroscopic Properties of Functionalized Acridinium- Containing Compounds
Abs = absorbance; CL = chemiluminescence
Example 11: Comparison of Chemiluminescence Reaction Rates of Acridiniums
[535] FIG. 11. illustrates the chemiluminescence reaction rates of DiChloro-CPSP and Di-t- butyl-CPSP after adding alkaline peroxide.
Example 12: Preparation of CPSP2
[536] 130 mg of MonoIodo-CPSP was dissolved in 7 mL acetonitrile and 3 mL water. To this was added 150 mg of sodium mercaptopropanesulfonic acid, followed by 0.3 mL DiEA where the solution turns from yellow to colorless. Next 25 mg of tBuXPhos Pd G3 catalyst was added and the reaction was stirred for 5 minutes when a light red color appeared. The reaction was quenched by adding 1 mL of 10% TFA in water making the solution darker red. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Fractions containing the product were combined and lyophilized to dryness. Yield - 60 mg red powder. The absorbance spectrum and chemiluminescence spectrum of CPSP2 is shown in FIGs. 13A and 13B, respectively.
MS (ESI): m/z 739.27 (M+) *H NMR (500 MHz, MeOD and 3% ND4OD) 5 - 7.85 (d, J = 8.30 Hz, 2H), 7.51 (d, J = 8.20 Hz, 2H), 7.46 (dd, J = 2.13, 8.78 Hz, 1H), 7.41 (t, J = 8.55 Hz, 1H), 7.30 (m, 2H), 6.93 (t, J = 7.43 Hz, 1H), 6.83 (d, J = 6.70 Hz, 1H), 6.67 (d, J = 1.90 Hz, 1H), 4.22 (t, J = 8.28 Hz, 2H), 3.28 (m, 2H), 3.10 (t, J = 6.88 Hz, 2H), 2.99-2.86 (m, 4H), 2.52 (s, 3H), 2.29 (p, J = 7.50 Hz, 2H), 2.02- 1.94 (m, 2H), 1.54-1.45 (m, 2H), 1.21 (m, 1H), 1.05 (m, 1H)
Example 13: Preparation of CPSP3
[6] (27)
[537] 120 mg of Dilodo-CPSP was dissolved in 2.5 mL acetonitrile and 2.5 mL water. To this was added 120 mg of sodium mercaptopropanesulfonic acid, followed by 0.1 mL DiEA where the solution turns from yellow to colorless. Next 30 mg of tBuXPhos Pd G3 catalyst was added and the reaction was stirred for 5 minutes when a light red color appeared. The reaction was quenched by adding 1 mL of 10% TFA in water making the solution darker red. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. A step gradient method was applied using acetonitrile (Solvent A), water (Solvent B), and 0.5% TFA in water (Solvent C). Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Fractions containing the product were combined and lyophilized to dryness. Yield - 80 mg red powder. The absorbance spectrum and chemiluminescence spectrum of CPSP3 is shown in FIGs. 14A and 14B, respectively.
MS (ESI): m/z 893.39 (M+)
H NMR (500 MHz, d6-DMSO and 3% ND4OD) 5 - 7.74 (d, J = 8.30 Hz, 2H), 7.50 (d, J = 8.15 Hz, 2H), 7.32 (dd, J = 2.10, 8.75 Hz, 2H), 7.21 (d, J = 8.95 Hz, 2H), 6.61 (d, J = 1.90 Hz, 2H), 4.04 (t, J = 7.83 Hz, 2H), 3.10 (t, J = 7.90 Hz, 2H), 2.85 (t, J = 7.05 Hz, 4H), 2.76 (t, J = 6.80 Hz, 2H), 2.60 (m, 4H), 2.45 (s, 3H), 2.00 (m, 2H), 1.80 (p, J = 7.46 Hz, 4H), 1.21 (t, J = 7.53, 2H), 0.92 (m, 2H)
Example 14: Preparation of Iodo-CPSP2
[538] 25 mg of Dilodo-CPSP was dissolved in 1.5 mL THF and 1 mL water. To this was added 5mg of Sodium Mercaptopropanesulfonate, followed by 0.01 mL DiEA and 3 mg of tBuXPhos Pd G3 catalyst and the reaction was stirred for 5 minutes. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Fraction containing the product were collected and lyophilized to dryness. Yield - 15 mg red powder MS (ESI): m/z 865.13 (M+)
Example 15: Preparation of CPSP3-Sulfone
[539] 25 mg of Dilodo-CPSP 15 mg of Iodo-CPSP2 was dissolved in 1 mL of DMF. The solution was cooled to OC in an ice batch and 35 mg was added portion- wise. Stirring continued for 25 minutes, where the product was observed by UPLC-MS. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Isolated fractions contained a mixture of both the acridinium and acridan iodo-sulfone intermediate, which were combined and lyophilized to dryness.
[540] 10 mg of the combined Iodo-Sulfone intermediates were dissolved in 1 mL THF and 1 mL water. To this was added 10 mg of Sodium Mercaptopropanesulfonate, followed by 0.01 mL DiEA and 3 mg of tBuXPhos Pd G3 catalyst and the reaction was stirred for 5 minutes. To the solution was then added 10 mg of FeC13 and stirring continued for 25 minutes. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Fractions containing the product were collected and
lyophilized to dryness. Yield - 4 mg of red/orange powder. The absorbance spectrum and chemiluminescence spectrum of CPSP3-sulfone is shown in FIGs. 15A and 15B, respectively. MS (ESI): m/z 923.38 (M+)
Example 16: Preparation of CPSP3-NHS
[541] 5 mg of CPSP3 was dissolved in 0.5 mL of DMSO. To this was added 2 mg of TSTU and 0.05 mL DiEA. The reaction was stirred for 1 hour before injecting on to a 5g Cl 8 Aq flash column and eluted with a gradient of 100% TFA/water (0.05%) to 100% MeCN. The product was isolated and lyophilized to dryness. Yield - 4.5 mg of red powder.
MS (ESI): m/z 990.47 (M+)
[542] 5 mg of CPSP3 from Example 13 was dissolved in 1 mL of DMF and cooled to 0 °C
(ice bath temperature). Next 3 mg of 77% m-CPBA was added, and the reaction stirred for 15
minutes. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system.
Absorbance @ 254 nm. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. The fraction containing the product was collected and lyophilized to dryness. Yield = 2.2 mg of CPSP3- Sulfoxide. The absorbance spectrum and chemiluminescence spectrum of CPSP3 -sulfoxide is shown in FIGs. 16A and 16B, respectively.
Exact Mass: 957.07 CPSP3 CPSP3-BisSulfone
[27] (32)
[543] 10 mg of the product CPSP3-Acridinium from Example 13 was dissolved in 2 mL of
DMF and cooled to 0 °C (ice bath temperature) and treated with 9.5 mg of 77% m-CPBA in very small portions over 1 hour with LCMS monitoring after each addition. The reaction was quenched with 3 drops of dimethyl sulfide followed by the addition of 2 mL of water. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2545 system. Absorbance @ 254 nm. A step gradient method (Flow rate 40 mL/Minute; mobile phase of ACN/H2O/IO % H2O- 0.5% TFA) was applied using acetonitrile (Solvent A), water (Solvent B), and (Solvent C) 0.5 % TFA in water (held at a constant 10 % throughout the gradient run). Recorder chart speed 5 mm/min. Fractions containing the product were combined and evaporated to dryness. Yield of CPSP3-BisSulfone, 1.7 mg. The absorbance spectrum and chemiluminescence spectrum of CPSP3-BisSulfone is shown in FIGs. 17A and 17B, respectively.
FIRMS (M-H) observed m/z 955.0524, calculated neutral mass 956.0600, found 956.0597 (mass error -0.4 mDa).
Example 19: Preparation of SP3-Acridone
Acridone is the product from the acridinium chemiluminescence reaction. As such, the fluorescence spectrum of acridone should match the spectrum observed from the chemiluminescence reaction. The SP3 -Acridone produced from alkaline peroxide-triggered CPSP3 was isolated. The fluorescence spectrum of SP3 -Acridone was found to match the chemiluminescence spectrum of CPSP3.
[544] 15 mg of CPSP3 from Example 13 was dissolved in 2 mL of water. To this was added
1 mL of 3% H2O2 followed by 1 mL of IM NaOH. A bright flash of green light was observed, and the reaction mixture was immediately purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Fractions containing the product were collected and lyophilized to dryness. Yield = 4 mg red/orange powder. The absorbance spectrum and fluorescence spectrum of SP3 -Acridone is shown in FIGs. 18A and 18B, respectively.
’H NMR (500 MHz, MeOD) 5 - 8.44 (d, J = 2.40 Hz, 2H), 7.95 (d, J = 9.20 Hz, 2H), 7.88 (dd, J = 2.35, 9.10 Hz, 2H), 4.76 (t, J = 8.45 Hz, 2H), 3.20 (t, J = 7.18 Hz, 4H), 3.10 (t, J = 6.65, 2H), 2.99 (t, J = 7.55 Hz, 4H), 2.36 (p, J = 7.48 Hz, 2H), 2.14 (p, J = 7.38 Hz, 4H)
Example 20: Preparation of Dilodo-Acridone from SP-Acridone
Dilodo-Acridone
[33] (35)
[545] 250 mg of 10-(3-sulfopropyl)-Acridone (SP-Acridone) was dissolved in 10 mL of
DCM and 10 mL of acetic acid. To this was added 1 gram of iodine monochloride. The reaction was stirred overnight before evaporating the solvent under reduced pressure. The mixture was dissolved in DMSO and the solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. A step gradient method was applied using acetonitrile (Solvent A), water (Solvent B), and 0.5% TFA in water (Solvent C). Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA.
Example 21: Preparation of SP3-Acridone from Dilodo-Acridone
From Dilodo-Acridone, SP3-Acridone can be obtained using sodium mercaptopropanesulfonic acid and tBuXPhos Pd G3. Spectral properties matched the properties of Example 19.
Exact Mass: 625.02
Di-lodoAcridone SP3-Acridone
[35] (39)
[546] 25 mg of Dilodo-Acridone from Example 20 was dissolved in 2.5 mL acetonitrile and 2.5 mL water. To this was added 50 mg of sodium mercaptopropanesulfonic acid, followed by 0.1 mL DiEA. Next 10 mg of tBuXPhos Pd G3 catalyst was added and the reaction stirred for 60 minutes. The reaction was quenched by adding 1 mL of 10% TFA in water. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. A step gradient method was applied using acetonitrile (Solvent A), water (Solvent B), and 0.5% TFA in water (Solvent C). Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5%TFA. Fractions containing the product were combined and lyophilized to dryness. Yield = 20 mg orange powder. Spectral properties match those of the compound from Example 19.
Example 22: Oxidation of SP3-Acridone
Reaction 1
[547] 10 mg of the product SP3-Acridone (39) from Example 21 was dissolved in 1 mL of DMF and cooled to 0 °C (ice bath temperature) and treated with 0.0045 g of 77% m-CPBA in one portion. This was stirred for 15 minutes at 0 °C and allowed to warm to room temperature over 1 hour. LCMS analysis indicated the presence of SP3-Acridone Bis Sulfoxide (41), SP3- acridone-Bis Sulfoxide Sulfone (42) and SP3-Acridone Bis Sulfone (44) as the major product components (see, FIG. 47). After this time, the reaction was quenched with 3 drops of dimethyl sulfide followed by the addition of 2 mL of water. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2545 system. Absorbance @ 254 nm. A step gradient method (Flow rate 40 mL/Minute; mobile phase of ACN/H2O/IO % H2O-0.5% TFA) was applied using acetonitrile (Solvent A), water (Solvent B), and (Solvent C) 0.5 % TFA in water (held at a constant 10 % throughout the gradient run). Recorder chart speed 5 mm/min. Fractions containing the products were combined and evaporated to dryness. Yield of the individual components: SP3-Acridone Bis Sulfoxide, 5.9 mg, HRMS (M-H) observed m/z 656.0066, calculated neutral mass 657.0137, found 657.0139 (mass error 0.2 mDa); SP3-acridone Sulfoxide Sulfone, 2.2 mg, HRMS (M-H) observed m/z 672.0015, calculated neutral mass 673.0086, found 673.0088 (mass error 0.2
mDa); SP3-Acridone Bis Sulfone, 1.0 mg, HRMS (M-H) observed m/z 687.9972, calculated neutral mass 689.0035, found 689.0045 (mass error 1.0 mDa).
[548] The absorbance spectrum and chemiluminescence spectrum of SP3-Acridone BisSulfoxide is shown in FIGs. 19A and 19B, respectively. The absorbance spectrum and fluorescence spectrum of SP3-Acridone Sulfoxide Sulfone is shown in FIGs. 20A and 20B, respectively. The absorbance spectrum and fluorescence spectrum of SP3-Acridone BisSulfone is shown in FIGs. 21 A and 2 IB, respectively.
Reaction 2
[549] 10 mg of the product SP3-Acridone (39) from Example 21 was dissolved in 4 ml of DMF and cooled to 0 °C (ice bath temperature) and treated with a solution of 0.0015 g of 77% m-CPBA in 2 mL of DMF (dropwise addition over 5 minutes). This was stirred for 15 minutes at 0 °C and allowed to warm to room temperature over 1 hour. LCMS analysis indicated the presence of SP3-Acridone Sulfoxide (40) as the major product component (see, FIG. 47). After this time, the reaction was quenched with 3 drops of dimethyl sulfide followed by the addition of 2 mL of water. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2545 system. Absorbance @ 254 nm. A step gradient method (Flow rate 40 mL/Minute; mobile phase of ACN/H2O/IO % H2O-0.5% TFA) was applied using acetonitrile (Solvent A), water (Solvent B), and (Solvent C) 0.5 % TFA in water (held at a constant 10 % throughout the gradient run). Recorder chart speed 5 mm/min. Fractions containing the product were combined and evaporated to dryness. Yield of SP3-Acridone Sulfoxide, 4.0 mg, HRMS (M-H) observed m/z 640.0110, calculated neutral mass 641.0188, found 641.0183 (mass error -0.4 mDa). The absorbance spectrum and fluorescence spectrum of SP3-Acridone Sulfoxide is shown in FIGs. 22A and 22B, respectively.
Exact Mass: 615.16
2,7-DiNitroCPSP 2,7-DiAminoCPSP
[8] (14)
[550] 5 mg of DiNitro-CPSP from Example 7 was dissolved in 2 mL of ethanol. To this was added 20 mg of SnCh and the reaction was allowed to stir for 18 hours. The reaction mixture was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 50 x 250 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 70 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. Yield was 2.5 mg. The absorbance spectrum and chemiluminescence spectrum of DiAmino-CPSP is shown in FIGs. 23 A and 23B, respectively.
MS (ESI): m/z 615.39 (M+)
Example 24: General preparation of aryl and heteroaryl substituted CPSP compounds
[551] A general scheme was followed for the synthesis of acridinium-containing compounds with at least one or two aryl/heteroaryl groups substituted at the 2 or 2,7-position. In general, 5 mg of lodo-CPSP or 5 mg of Dilodo-CPSP, 0.5 mg of Pd(PPh3)4, and 5 mg of either a boronic acid or a boronate ester was dissolved in 0.5 mL of acetonitrile, 0.5 mL water, and 0.05 mL DiEA. The reaction was stirred for 10-30 minutes at 40°C. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O-0.5% TFA. The fraction containing the product was collected and lyophilized to dryness.
[5] (15, wherein Rg = H)
[552] Based on the general scheme of Example 24, Ph-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of phenylboronic acid pinacol ester. Yield was 4 mg of Ph-CPSP. The absorbance spectrum and chemiluminescence spectrum of Ph-CPSP is shown in FIGs. 24A and
24B, respectively.
[553] Based on the general scheme of Example 24, HPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 4-hydroxyphenylboronic acid pinacol ester. Yield was 2.5 mg of HPh-
CPSP. The absorbance spectrum and chemiluminescence spectrum of HPh-CPSP is shown in FIGs. 25A and 25B, respectively.
MS (ESI): m/z 677.27 (M+)
[5] (15, wherein Rg = -N(Me)2)
[554] Based on the general scheme of Example 24, DMAPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 4-(N,N-Dimethylamino)phenylboronic acid pinacol ester. Yield was
3.5 mg of DMAPh-CPSP. The absorbance spectrum and chemiluminescence spectrum of
(15, wherein Rg = -CN)
[555] Based on the general scheme of Example 24, 4-cyanoPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg Cyanophenylboronic acid. Yield was 3.2 mg of 4-CyanoPh-CPSP.
The absorbance spectrum and chemiluminescence spectrum of 4-CyanoPh-CPSP is shown in FIGs. 27A and 27B, respectively.
MS (ESI): m/z 686.44 (M+)
[556] Based on the general scheme of Example 24, AMPh-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 4-(Aminomethyl)phenylboronic acid pinacol ester hydrochloride. Yield was 4.5 mg of AMPh-CPSP.
MS (ESI): m/z 690.00 (M+)
Example 30: Preparation of Pyrrolyl-CPSP
= H)
[557] Based on the general scheme of Example 24, Pyrrolyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of N-Boc-2-Pyrroleboronic acid the Boc-protected Pyrrolyl-CPSP was obtained. This material was dissolved in 1 mL of DCM and 1 mL of TEA. and allowed to react for 4 hours. The solvent was removed in vacuo. Yield was 2.8 mg of Pyrrolyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of Pyrrolyl-CPSP is shown in FIGs. 28 A and 28B, respectively.
MS (ESI): m/z 650.40 (M+)
[558] Based on the general scheme of Example 24, Furanyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 2-furanylboronic acid. Yield was 3.0 mg of Furanyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of Furanyl-CPSP is shown in FIGs. 29A and 29B, respectively.
MS (ESI): m/z 651.29 (M+)
Example 32: Preparation of 5-Cyano-Thienyl-CPSP
[5] (21, wherein Rg = -CN)
[559] Based on the general scheme of Example 24, 5-cyano-thienyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 5-cyanothiophene-2-boronic acid. Yield was 2.6 mg of 5-
Cyano-Thienyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of 5-Cyano-
Thienyl-CPSP is shown in FIGs. 30A and 30B, respectively.
MS (ESI): m/z 692.29 (M+)
[560] Based on the general scheme of Example 24, 5-Me-thienyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 5-Methylthiophene-2-boronic acidpinacol ester. Yield was 2.8 mg of 5-Me-Thienyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of 5- Me-Thienyl-CPSP is shown in FIGs. 31 A and 3 IB, respectively.
MS (ESI): m/z 681.26 (M+)
[561] Based on the general scheme of Example 24, 5-MeO-thienyl-CPSP was prepared from
5 mg of lodo-CPSP and 5 mg of 5 -Methoxythiophene-2 -boronic acid pinacol ester. Yield was
2.7 mg of 5-MeO-Thienyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of
5-MeO-Thienyl-CPSP is shown in FIGs. 32A and 32B, respectively.
MS (ESI): m/z 697.10 (M+)
[5] (21, wherein Rg = -OMe)
[562] Based on the general scheme of Example 24, 4-MeO-thienyl-CPSP was prepared ftom
5 mg of lodo-CPSP and 5 mg of 5 -Methoxythiophene-2 -boronic acid pinacol ester. Yield was
2.5 mg of 4-MeO-Thienyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of 4-MeO-Thienyl-CPSP is shown in FIGs. 33A and 33B, respectively.
MS (ESI): m/z 697.32 (M+)
[5] (21, wherein Rg = -OMe)
[563] Based on the general scheme of Example 24, 3-MeO-thienyl-CPSP was prepared from
5 mg of lodo-CPSP and 5 mg of 3 -Methoxythiophene-2 -boronic acid. Yield was 1.7 mg of 3-
MeO-Thienyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of 3-MeO-
Thienyl-CPSP is shown in FIGs. 34A and 34B, respectively.
MS (ESI): m/z 697.03 (M+)
[5] (21, wherein Rg = -SMe)
[564] Based on the general scheme of Example 24, 5-MeS-thienyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of 5-Methylthiothiophene-2-boronic acid pinacol ester. Yield was 1.1 mg of 5-MeS-Thienyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of 5-MeS-Thienyl-CPSP is shown in FIGs. 35A and 35B, respectively.
MS (ESI): m/z 713.31 (M+)
Exact Mass: 723.10 lodo-CPSP Dithienyl-CPSP
[5] (23)
[565] Based on the general scheme of Example 24, Thienothiophenyl-CPSP was prepared from 5 mg of lodo-CPSP and 5 mg of Thienothiophene-2-boronic acid. Yield was 4.5 mg of Thienothiophenyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of Thienothiophenyl-CPSP is shown in FIGs. 36A and 36B, respectively.
MS (ESI): m/z 723.30 (M+)
Example 39: Preparation of Bis(5-Me-Thienyl)-CPSP
[566] Based on the general scheme of Example 24, Bis(5-Me-Thienyl)-CPSP was prepared from 5 mg of Dilodo-CPSP and 5 mg of 5-Methylthiophene-2-boronic acid pinacol ester. Yield was 2 mg of Bis(5-Me-Thienyl)-CPSP. The absorbance spectrum and chemiluminescence spectrum of Bis(5-Me-Thienyl)-CPSP is shown in FIGs. 37A and 37B, respectively.
MS (ESI): m/z 777.26 (M+)
[567] Based on the general scheme of Example 24, Difuranyl-CPSP was prepared from 5 mg of Dilodo-CPSP and 5 mg of 2-Furanylboronic acid. Yield was 3.9 mg of DiFuranyl-CPSP. The absorbance spectrum and chemiluminescence spectrum of DiFuranyl-CPSP is shown in FIGs. 38A and 38B, respectively.
(15, wherein Rg = -CH2NH2) (15, wherein Rg = -CH2NH-TAMRA)
[568] 5 mg of AMPh-CPSP from Example 29 and 5 mg of tetramethylrhodamine (TAMRA)
NHS Ester were dissolved in 0.5 mL of DMSO. To this was added 0.05 mL of DiEA and the reaction mixture was stirred for 18 hours. The solution was purified by reverse phase HPLC by elution of a single injection on a YMC ODS AQ 30 x 150 mm I.D. steel column with a Waters Separations 2000 system. Absorbance @ 254 nm. Recorder chart speed 5 mm/min. A manual step gradient method (flow rate 40 mL/min) was used with a mobile phase of ACN/H2O/H2O- 0.5% TFA. Fraction containing the product were collected and lyophilized to dryness. Yield - 2 mg. The 7absorbance spectrum and chemiluminescence spectrum of TAMRA-Ph-CPSP is shown in FIGs. 39A and 39B, respectively.
MS (ESI): m/z 551.59 (M++)
Example 42: Summary of Chemiluminescence of Acridinium Compounds Having Sulfide Substitutions at 2- and 2,7 Positions
[569] As described herein, acridinium-containing compounds with at least one sulfide, or with one sulfide and one sulfone, substituted at the 2 or 2,7-position were synthesized.
Advantageously, the resulting sulfide-containing acridinium compounds are highly water-soluble species. Treatment of the mono- or di-substituted acridinium-sulfides with standard alkaline peroxide conditions resulted in a bright blue-green or green CL color, with a CL Xmax = 490 nm and 505 nm, respectively. The CL emission wavelength is significantly red-shifted from the CPSP molecule with CL A,max = 445 nm. Interestingly, further spectral shifts were observed when the oxidation state of the sulfur atom is increased. The non-symmetric molecule CPSP3-Sulfone,
with a sulfone-sulfide substitution, was observed to be further red-shifted with CL Xmax = 520 nm (compare, e.g., FIGs. 12B (CPSP), 13B (CPSP2), 14B (CPSP3), and 15B(CPSP3 -Sulfone)).
[570] A summary of the characteristics of the modified acridinium-containing compounds described herein is provided in Table E.
[571] Previous atempts in using red-shifted Acr systems for IVD applications suffered from increased hydrophobicity, poor protein conjugate stability, lower CL emission intensity, and in some cases higher levels of emission wavelength cross-talk. The acridinium-sulfides described herein have brighter CL emission and depending on the alkyl-sulfide substituent, can be highly water soluble and form stable protein conjugates. Moreover, the number of sulfide substituents and their oxidation state affect the amount of red-shift in the CL emission, allowing for control over emission wavelength and beter pairing with blue-shifted acridinium-containing compounds. Accordingly, the molecules described herein can find use in multicolor-CL experiments including multiplexed assays, expanding dynamic range, and internal calibration methods.
Example 43: Resolving CPSP and CPSP3 signals in the CPSP and CPSP3 mixture with high precision
[572] Various amounts of CPSP were mixed with CPSP3 at signal range from 1 :500 to 500: 1. As shown in FIG. 41, with the chosen optical filters (shaded blue and green on the figure), signals from CPSP or CPSP3 can be resolved at high precision, with the coefficient of variation (CV) of the resolved signals from the lower signal species less than 5% if the signal ratio between the two
compounds is within 100-fold difference (see, FIG. 42). This suggests the selected acridinium pair can be used to generate signals at two independent optical channels with minimal interference.
Example 44: Multiplexing: Duplex SARS-Cov2 IgG and IgM assay with CPSP and CPSP3 and a Dual Channel Detection System
[573] A SARS-Cov2 IgG and IgM assay was prepared on ARCHITECT® using Abbott on- market ARCHITECT® kit using an on-market kit with prototype (adapted) assay files, and the Chemi luminescent Microparticle Immuno Assay (CMIA) reads were performed on a stand-alone dual-channel test stand. The anti-IgM antibody conjugate was labeled with CPSP3-NHS, and mixed with anti-IgG antibody labeled with CPSP (from the original kit). FIG. 43 shows the IgG and IgM antibody levels from a single patient post vaccination using simultaneous dual channel measurements.
Example 45: Expanding Assay Dynamic Range with a Flag for Hook Sample using a Dual Channel Detection System
[574] Analytes from a patient sample have a wide range of concentration. Often, the assays could not cover the required dynamic range, thus sample dilution is required. More than 10% of current immunoassays have a sample dilution protocol implemented in the assay file. This is not cost or time effective. With the implement of dual color acridinium-containing compounds and a dual channel instrument, the assay dynamic range can be easily extended such that sample dilution is not needed. The following example shows the bHCG assay range was expanded from 0-15,000 mIU/mL to 0-450,000 mIU/mL with improved sensitivity.
[575] The Anti-HCG conjugates were labeled with CPSP or CPSP3-NHS. O.lnM anti-HCG- CPSP was placed in the assay specific diluent (ASD) bottle. 2nM anti-HCG-CPSP3 was placed in the conjugate bottle. The microparticles are used from ARCHITECT® bHCG assay kit. 60 pL bHCG sample was mixed with 25 pL microparticles and 50 pL ASD, after 18 minutes incubation, unbound sample and anti-HCG-CPSP were washed away; Then 50 pL of anti-HCG- CPSP3 were introduced to the reaction vessel (RV). A schematic of the reaction process is shown in FIG. 44A. After 4 minutes incubation and wash, the sample was triggered and read with a dual channel system.
[576] The assay curves were ploted based on signal measured in blue channel and green channel (see, FIGs. 45A-45C), the green channel also serves as the flag channel to indicate whether the measured signal corresponds to the rising section or the descending section of the Alternative Cal Curve. When combined, the assay dynamic range can cover all bHCG levels from a patient sample, thus eliminating the need for retest or auto-dilution. As seen in FIGS. 46A-46C, when comparing to standard ARCHITECT® bHCG assay (e.g., Alternative Cal Curve), the method using the acridinium-containing compounds described herein has improved sensitivity and wider dynamic range.
Example 46: Duplex assay Hepatitis B core Antigen with CPSP and CPSP3 and a Dual Channel Detection System
[577] Described herein is the development and use of a fully automated duplex assay to specifically and simultaneously analyze phosphorylated HBcAg (P-HBcAg) and nonphosphorylated HBcAg (HBcAg).
[578] The duplex assay developed is a chemiluminescent microparticle immunoassays (CMIA) on a fully automated platform which use specific monoclonal antibodies to capture and detect either phosphorylated or non-phosphorylated HBcAg.
[579] METHODS:
[580] P-HBcAg/HBcAg detection: Recombinant phosphorylated HBcAg (P-rHBcAg) and non-phosphorylated HBcAg (rHBcAg) were fully automated analyzed on the Alinity i dual channel instrument. P-HBcAg/HBcAg was captured and detected by monoclonal antibodies directed to phosphorylated or non-phosphorylated HBcAg. Detection was achieved by antibodies labeled with two different luminescent molecules, which emit photons of light at different wavelengths (blue and green). Using a dual detection system and specific filters enables the simultaneously detection of P-HBcAg and HBcAg.
[581] RESULTS:
[582] Table F shows the P-HBcAg/HBcAg duplex assay with simultaneous detection of P- HBcAg/HBcAg, which were determined in triplicate using recombinant P-HBcAg/HBcAg. RLUs for CPSP were corrected by subtraction of residual RLUs obtained from CPSP3 that passed the filter and vice versa. Abl binds to P-rHBcAg and Ab2 binds to rHBcAg.
Table F
Example 47: Resolving IgG and IgM mixture with Dichloro-CPSP and Ditbutyl-CPSP and a Single Channel Detection System
[583] In previous examples, it was shown that signals can be separated with filters in the spectrum domain. It is also possible to separate signals in the time domain. Dichloro-CPSP (fast reacting) and ditbutyl-CPSP (slow reacting) have very distinctive reaction kinetics (see, Figure
11). For simple analysis, the 3 second read interval was split into two-time windows, from 0- 0.75 seconds (Time Window 1) for dichloro-CPSP and 0.75-3 seconds (Time Window 2) for ditbutyl-CPSP. There is only 2.6% bleed through of dichloro-CPSP into Time Window 2. Conversely, there is 22.7% of bleed through of ditbutyl-CPSP into Time Window 1). It is critical to use these correction factors when combining the signals on a single detector.
[584] For proof of principle: Samples of human IgG and IgM were labeled with biotin and used as analyte. Anti-human IgG antibody was labeled with dichloro-CPSP and anti-human IgM antibody was labeled with ditbutyl-CPSP. Various IgG-bt and IgM-bt mixture were first captured by Streptavidin-coated microparticles (MPs) (0.1%, Invitrogen) and detected with anti-IgG- dichloro-CPSP and anti-IgM-ditbutyl-CPSP secondary antibodies. FIG. 48 shows the resolved signal of IgG and IgM from its mixture. The advantage of using the time domain for signal separation is simplicity of hardware requirement. It can be directly measured on a single channel system instead of dual channel system.
Example 48: Duplex SARS-Cov2 IgG and IgM assay performed with Dichloro-CPSP and Ditbutyl-CPSP and a single channel detection system
[585] A SARS-Cov2 IgG and IgM assay was performed on COVTD-vaccinated patient sample using Abbott on-market ARCHITECT® kit. The conjugates were replaced with dichloro- CPSP labeled anti-human IgG antibody and /or ditbutyl-CPSP labeled anti-human IgM antibody.
[586] FIG. 49A-49C show the separate measurement of IgG or IgM antibody levels from a single patient pre- and post-vaccination (Day 0, 15, 29, 57, 82) using either dichloro-CPSP labeled anti-human IgG antibody or ditbutyl-CPSP labeled anti-human IgM antibody. As expected, IgM levels initially spike 1 -2 weeks after exposure and then drop. IgG levels rise more slowly and then remain in high abundance over the course of months. FIG 49C shows simultaneous measurement of IgG and IgM antibody levels from a single patient pre and post vaccination (Day 0, 15, 29, 57, 82) using dichloro-CPSP labeled anti-human IgG antibody and Di-t-butyl-CPSP labeled anti-human IgM antibody. The dose response profile of the individual IgG and IgM assay agrees well with the duplex IgG and IgM assay.
Example 49: A twostep dual channel combo assay run on Dual-Channel Detection System to Extend Assay Dynamic Range
[587] Reagents and instruments are previously described in Example 1. The assay kit comprises a microparticle bottle (off shelf from Abbott bHCG ARCHITECT® assay kit), a Conjugate bottle (1 nM Fab-CPSP3 Conjugate diluted in phosphate buffer), A Tracer bottle (6.25 IU/L bHCG-CPSP tracer in MES buffer).
Methods
[588] Generating a calibration curve for bHCG: 25 pL of 0.1% bHCG Ab coated microparticles mixed with 25 pL bHCG (with known concentration ranging from (0 to 450,000 mIU/mL) and 50pL bHCG-CPSP tracer were allowed to react for 18 minutes and washed to remove unbound sample and tracer. To the reaction vessel, 50pL Fab-CPSP3 conjugate was added and incubated for 4 minutes. Then, after a wash, a dual channel system was used to measure the signal with pre-trigger and trigger solution (See, FIG. 44B). The signals were processed in the similar manner to Example 1. With the established calibration curve (FIGs. 5OA-5OB), 92 patient samples were measured on the dual channel system and the concentration of each sample was calculated using the established calibration curve. Patient sample concentrations range from 1 mIU/mL to 200,000 mIU/mL. The concentration of each sample
was also pre-determined on a commercial instrument. FIGs. 50C-50E shows the correlation of pregnant patients’ bHCG value measured on the two systems. For any sample above 15000 mIU/mL, they were first flagged on the Abbott commercial instrument, then the instrument performed a 15X sample dilution before re-running the test, whereas the dual channel system does not require the extra step of dilution and re-testing.
[589] For reasons of completeness, various aspects of the disclosure are set out in the following numbered clauses:
Clause 1. A method for determining the amount of an analyte of interest in a biological sample, the method comprising the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. performing, in the same reaction vessel, a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
Clause 2. The method of clause 1, wherein the calibration curve is obtained by the steps of: a. performing, in a reaction vessel, a sandwich immunoassay on the analyte of interest having a known concentration, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; b. performing, in the same reaction vessel, a competitive immunoassay on analyte of interest having a known concentration, wherein the competitive immunoassay comprises a
capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and c. calculating the signal ratio of first detectable signal and the second detectable signal; and d. varying concentrations of the analyte of interest and repeating steps a-c to generate a calibration curve using the signal ratios generated in step c.
Clause 3. The method of clause 1 or clause 2, wherein the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
Clause 4. The method of any of clauses 1 to 3, wherein the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a chromophore, a chemiluminescent label, or any combinations thereof.
Clause 5. The method of any of clauses 1 to 4, wherein the first detectable signal comprises a first color and the second detectable signal comprises a second color.
Clause 6. The method of any of clauses 1 to 4, wherein the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
Clause 7. The method of any of clauses 1 to 6, wherein the method further comprises using a first detection component to detect the first detectable signal generated by the first detectable label and a second detection component to detect the second detectable signal generated by the second detectable label.
Clause 8. The method of clause 7, wherein the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
Clause 9. The method of any of clauses 1 to 8, wherein the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”), that competes with the analyte of interest wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
Clause 10. The method of any of clauses 1 to 8, wherein the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
Clause 11. The method of any of clauses 1 to 10, wherein the first detection reagent or the second detection reagent is an antigen or an antibody.
Clause 12. The method of clauses 1 to 11, wherein the analyte of interest and the competing analyte are each captured on a solid support.
Clause 13. The method of any of clauses 1 to 12, wherein the capture reagent comprises an antibody or an antigen.
Clause 14. The method of any of clauses 1 to 13, wherein the sandwich assay is performed in one step or two steps.
Clause 15. The method of any of clauses 1 to 14, wherein the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained from the subject.
Clause 16. A system for determining the amount of an analyte of interest in a biological sample, the system comprising: a. a calibration curve for the analyte of interest; b. a sandwich immunoassay wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. a competitive immunoassay wherein the competitive immunoassay comprises a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. a device comprising: i. a reaction vessel for performing the sandwich immunoassay and the competition assay; ii. a first detection component to determine the amount of the first detectable signal generated by the first detectable label; and iii. a second detection component to determine the amount of the second detectable signal generated by the second label,
wherein: (i) the amount of the first detectable signal determined by the first detection component and the amount of the second detectable signal determined by the second detection component is used to calculate the signal ratio of first detectable signal and second detectable signal; and (ii) the signal ratio is used to determine the amount of the analyte of interest in the biological sample.
Clause 17. The system of clause 16, wherein the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
Clause 18. The system of clause 16 or clause 17, wherein the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a biotin/avidin label, a chromophore, a chemiluminescent label, or any combinations thereof.
Clause 19. The system of any of clauses 16 to 18, wherein the first detectable signal comprises a first color and the second detectable signal comprises a second color.
Clause 20. The system of any of clauses 16 to 18, wherein the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
Clause 21. The system of any of clauses 16 to 20, wherein the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
Clause 22. The system of any of clauses 16 to 21, wherein the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
Clause 23. The system of any of clauses 16 to 21, wherein the first detection reagent, the second detection reagent or the first detection agent and the second detection reagent is an antigen or an antibody.
Clause 24. The system of any of clause 22 or clause 23, wherein the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
Clause 25. The system of any of clauses 16 to 24, wherein the capture reagent comprises an antibody or an antigen.
Clause 26. The system of any of clauses 16 to 25, wherein the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained from the subject.
V is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, Rl is -SO2-A, wherein A is selected from alkyd, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, or heterocyclylalkyl; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalky 1, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(-O)2-OR*, -O-S(-O)2-OR*, -NR*-S(-O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents.
Clause 28. The compound of clause 27, wherein Y is nitrogen.
Clause 29. The compound of clause 28, wherein A is aryl that is unsubstituted or substituted with 1, 2, 3, 4, or 5 substituents selected from C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, and selenyl.
Clause 30. The compound of any of clauses 27-29, wherein R2 is -COOZ.
Clause 31. The compound of clause 30, wherein Z is selected from hydrogen, C1-C4 alkyl, or N-hydroxysuccinimide.
Clause 32. The compound of any of clauses 27-31, wherein Li and L2 are each independently C1-C4 alkylene.
Clause 33. The compound of any of clauses 27-32, wherein Ra, Rb, Rc, Rd, Re, and Rf are each hydrogen.
Clause 34. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are hydrogen.
Clause 35. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are -S-L3-SO3H.
Clause 36. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are -S(=O)-L3-SO3H.
Clause 37. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are -SO2-L3-SO3H.
Clause 38. The compound of any of clauses 27-33, wherein R’ is -SO2-L3-SO3H and R” is -S-L3-SO3H.
Clause 39. The compound of any of clauses 27-33, wherein R’ is -S-L3-SO3H and R” is one of H, Cl, Br, I, -NO2, t-butyl, or -SO3H.
Clause 40. The compound of any of clauses 27-33, wherein R’ is -S-L3-SO3H and R” is H.
Clause 41. The compound of any of clauses 27-33, wherein R’ is -SO2-L3-SO3H and R” is one of Cl, Br, I, -NO2, t-butyl, or -SO3H.
Clause 42. The compound of any of clauses 27-33, wherein R’ is -(S=O)-L3-SC>3H and R” is -S-L3-SO3H.
Clause 43. The compound of any of clauses 27-33, wherein R’ is -(S=O)-L3-SC>3H and R” is one of Cl, Br, I, -NO2, t-butyl, or -SO3H.
Clause 44. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are a substituted or unsubstituted phenyl group.
Clause 45. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are a phenyl group substituted with at least one of hydroxy, cyano, alkylamino, dialkylamino, or aminoalkyl.
Clause 46. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are a substituted or unsubstituted heteroaryl group.
Clause 47. The compound of any of clauses 27-33, wherein R’, R”, or both R’ and R” are a substituted or unsubstituted heteroaryl group selected from pyrrolyl, furanyl, thienyl, or thienothiophenyl.
Clause 48. The compound of any of clauses 27-33, wherein the compound has the Formula (Ila):
wherein R’ and R” are defined above for Formula (I).
Clause 49. The compound of any of clauses 27-33, wherein the compound is:
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), and wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano,
nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
Clause 54. A method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring, said method comprising: reducing the acridinium ring to an acridan intermediate using a first solution comprising at least one reducing agent and at least one acid; and reacting the acridan intermediate with an electrophile to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring.
Clause 55. The method of clause 54, wherein the at least one acid comprises at least one of ammonium chloride, formic acid, acetic acid, benzoic acid, hydrofluoric acid, phosphoric acid, sulfurous acid, hypochlorous acid, citric acid, carbonic acid, nitrous acid, oxalic acid, boric acid, ammonium sulfate, or ammonium oxalate.
Clause 56. The method of clauses 54 or 55, wherein the at least one reducing agent comprises zinc or a hydride.
Clause 57. The method of clause 56, wherein the zinc comprises elemental zinc, a zinc alloy, or a combination of both.
Clause 58. The method of clause 56, wherein the hydride comprises sodium borohydride, lithium aluminum hydride, sodium cyanoborohydride, or sodium triacetoxyborohydride.
Clause 59. The method of any of clauses 54-58, wherein the first solution further comprises a water-miscible alcohol, preferably ethanol.
Clause 60. The method of any of clauses 54-59, wherein the acridinium ring is reduced at temperature in a range from about 50°C to about 90°C, preferably about 60°C to about 80°C.
Clause 61. The method of any of clauses 54-60, wherein the acridinium-containing compound is represented by the compound of Formula (I) of any of claims 27-46.
Clause 62. The method of clause 61, wherein R’ = R” = H.
Clause 63. The method of any of clauses 54-62, wherein the acridan intermediate is represented by Formula (III):
wherein Y, Ri, R2, Li, L2, Q, Rs, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I).
Clause 64. The method of any of clauses 54-63, wherein the electrophile comprises at least one of Cl, Br, I, NO2, SO3H, or t-butyl.
Clause 65. The method of clause 64, wherein the electrophile is Cl and the products are Formula (IVa), (IVb), or both (IVa) and (IVb).
Clause 66. The method of clause 65, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium fimctionalized with Cl at the 2- or the 2,7- position, or both, of the acridinium ring includes a chlorination reaction.
Clause 67. The method of clauses 65 or 66, further comprising: mixing the acridan intermediate of Formula (III) with a second solution in a container, and stirring for about 12 hours to about 24 hours, preferably about 16 hours to about 20 hours; adding an aqueous solution of at least one oxidant to the container, in portions, over about 30 minutes to about 90 minutes, preferably about 45 minutes to about 75 minutes; and isolating and optionally purifying the product comprising the compound of Formula (IVa), (IVb), or both (IVa) and (IVb).
Clause 68. The method of clause 67, wherein the second solution comprises a chloride salt, N-chlorosuccinimide, and p-toluenesulfonic acid in a water/acetonitrile mixture.
Clause 69. The method of clauses 67 or 68, wherein the oxidant comprises FeCh, 2,3- Dichloro-5,6-dicyano-l,4-benzoquinone (DDQ), chloranil, or ceric ammonium nitrate (CAN).
Clause 70. The method of clause 64, wherein the electrophile is Br and the products are Formula (Va), (Vb), or both (Va) and (Vb).
Clause 71. The method of clause 70, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with Br at the 2- or the 2,7- position, or both, of the acridinium ring includes a bromination reaction.
Clause 72. The method of clauses 70 or 71, further comprising: dissolving the acridan intermediate in a third solution in a container; adding bromine to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Va), (Vb), or both (Va) and (Vb).
Clause 73. The method of clause 64, wherein the electrophile is I and the products are Formula (Via), (VIb), or both (Via) and (Vlb).
Clause 74. The method of clause 73, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with I at the 2- or the 2,7- position, or both, of the acridinium ring includes an iodination reaction.
Clause 75. The method of clauses 73 or 74, further comprising: dissolving the acridan intermediate in a third solution in a container; adding iodine monochloride to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb).
Clause 76. The method of clause 64, wherein the electrophile is NO2 and the products are Formula (Vila), (Vllb), or both (Vila) and (Vllb).
Clause 77. The method of clause 76, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with NO2 at the 2- or the 2,7- position, or both, of the acridinium ring includes a nitration reaction.
Clause 78. The method of clauses 76 or 77, further comprising: dissolving the acridan intermediate in a third solution in a container; adding nitric acid to the container; and isolating and optionally purifying the product comprising the compound of Formula (Vila), (Vllb), or both (Vila) and (Vllb).
Clause 79. The method of clause 64, wherein the electrophile is SO3H and the products are Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
Clause 80. The method of clause 79, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with SO3H at the 2- or the 2,7- position, or both, of the acridinium ring includes a sulfonation reaction.
Clause 81. The method of clauses 79 or 80, further comprising: dissolving the acridan intermediate in a fourth solution in a container; adding chlorosulfonic acid to the container and stirring for about 5 minutes to about 30 minutes; adding water to the container and stirring for about 10 minutes to about 30 minutes; and isolating and optionally purifying the product comprising the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
Clause 82. The method of clause 64, wherein the electrophile is t-butyl and the products are Formula (IXa), (IXb), or both (IXa) and (IXb).
Clause 83. The method of clause 82, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with t-butyl at the 2- or the 2,7- position, or both, of the acridinium ring includes a Friedel-Craft alkylation reaction.
Clause 84. The method of clauses 82 or 83, further comprising: dissolving the acridan intermediate in a fifth solution in a container; adding a Lewis acid to the container and heating at about 40°C to about 60°C for about 18-24 hours; reducing solvent in vacuo followed by the addition of a water/acetonitrile solution to the container; adding an aqueous solution of an oxidant to the container dropwise over time; and isolating and optionally purifying the product comprising the compound of Formula (IXa), (IXb), or both (IXa) and (IXb).
Clause 85. The method of any of clauses 54-84, further comprising reacting the product comprising the acridinium functionalized at the 2 -position with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (XVI).
Clause 86. The method of clause 85, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
Clause 87. The method of clauses 85 or 86, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
Clause 88. The method of any of clauses 85-87, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
Clause 89. The method of any of clauses 85-88, wherein the Pd-containing catalyst comprises 2-Di-ter/-butylphosphino-2',4',6'-triisopropyl-l,l'-biphenyl)-2-(2'-amino-l,l'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
Clause 90. The method of any of clauses 85-89, further comprising isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
Clause 91. The method of any of clauses 54-84, further comprising reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium disulfide compound of Formula (XVII).
Clause 92. The method of clause 91, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
Clause 93. The method of clauses 91 or 92, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
Clause 94. The method of any of clauses 91-93, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
Clause 95. The method of any of clauses 91-94, wherein the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
Clause 96. The method of any of clauses 92-95, further comprising isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
Clause 97. The method of any of clauses 54-84, further comprising: reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (I), wherein R’ is -S-L3-SO3H and R” is selected from Cl, Br, I, -NO2, t-butyl, or -SO3H; oxidizing the sulfide moiety to a sulfone; reacting the R” group selected from Cl, Br, I, -NO2, t-butyl, or -SO3H with the reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium sulfone compound of Formula (XXI).
Clause 98. The method of clause 97, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
Clause 99. The method of clauses 97 or 98, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
Clause 100. The method of any of clauses 97-99, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
Clause 101. The method of any of clauses 97-100, wherein the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
Clause 102. The method of any of clauses 97-101, wherein the sulfide moiety is oxidized using meta-Chloroperoxybenzoic acid (mCPBA).
Clause 103. The method of any of clauses 97-102, further comprising isolating and optionally purifying the acrinidium sulfone compound of Formula (XXI).
Clause 104. The method of any of clauses 76-78, wherein the NOi group(s) are reduced to amine group(s) using a reducing agent to obtain the compound of Formula (Xa) or (Xb).
Clause 105. The method of clause 104, wherein the reducing agent comprising SnCh.
Clause 106. A method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring comprising: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a boronic acid comprising A’ or a boronate ester comprising A’, wherein A’ is a substituted or unsubstituted aryl group or a substituted or unsubstituted heteroaryl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container; stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
Clause 107. The method of clause 106, wherein the acridinium compound is functionalized by:
(i) at least one phenyl group to produce a compound of Formula (Xia) or (Xlb);
(ii) at least one furanyl group to produce a compound of Formula (Xllla) or (Xlllb);
(iii) at least one thienyl group to produce a compound of Formula (XlVa) or (XlVb); or
(iv) at least one thienothiophenyl group to produce a compound of Formula (XV),
wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
Clause 108. The method of clause 106, wherein the acridinium compound is functionalized by at least one pyrrolyl group to produce a compound of Formula (Xlla) or (Xllb), wherein the boronic acid comprising A’ or the boronate ester comprising A’ is tertbutyloxycarbonyl (Boc)-protected, and wherein the method comprises after the stirring step: isolating the product comprising the acridinium-containing compound comprising a Boc-protected pyrrolyl group; dissolving the acridinium-containing compound comprising the Boc-protected pyrrolyl group in dichloromethane and trifluoroacetic acid for about 2-6 hours; removing the solvent in vacuo,' and optionally purifying the product comprising the compounds of Formula Xlla or Xllb, wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
Clause 109. The method of any of clauses 106-108, wherein the reactant compound comprises iodo-CPSP or diiodo-CPSP.
Clause 110. A method of producing a sulfoxide of Formula (XVIII) or (XIX), said method comprising: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and
isolating and optionally purifying the sulfoxide compound of Formula (XVIII) or (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
Clause 111. The method of clause 110, wherein the at least one organic solvent comprises tetrahydrofiiran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
Clause 112. The method of clauses 110 or 111, wherein the oxidizing agent comprises mCPBA.
Clause 113. The method of any of clauses 110-112, wherein the compound of Formula (XVII) is CPSP3.
Clause 114. A method of producing a BisSulfone compound of Formula (XXII) comprises: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; quenching an oxidation reaction with dimethyl sulfide and adding an amount of water; and isolating and optionally purifying the BisSulfone compound of Formula (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
Clause 115. The method of clause 114, wherein the at least one organic solvent comprises tetrahydrofiiran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
Clause 116. The method of clauses 114 or 115, wherein the oxidizing agent comprises mCPBA.
Clause 117. The method of any of clauses 114-116, wherein the compound of Formula (XVII) is CPSP3.
Q is -SO3Z or H;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Li is selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(^O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, -
S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2 wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein at least one of R’ or R” has to be -S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
Clause 119. The compound of clause 118, wherein Q is -SO3Z, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents, preferably -SO3H.
Clause 120. The compound of clauses 118 or 119, wherein Li is a C1-C10 alkylene, preferably — CH2CH2CH2— .
Clause 121. The compound of any of clauses 118-120, wherein Ra, Rb, Rc, Rd, Re, and Rf are each hydrogen.
Clause 122. The compound of any of clauses 118-121, wherein R’, R”, or both R’ and R” are -S-L3-SO3H.
Clause 123. The compound of any of clauses 118-121, wherein R’, R”, or both R’ and R” are -S(=O)-L3-SO3H.
Clause 124. The compound of any of clauses 118-121, wherein R’, R”, or both R’ and R” are -SCh-Ls-SChH.
Clause 125. The compound of any of clauses 118-121, wherein R’ is -SCh-Ls-SChH and R” is -S-L3-SO3H.
Clause 126. The compound of any of clauses 118-121, wherein R’ is -SO2-L3-SO3H and R” is -S(=O)-L3-SO3H.
Clause 127. The compound of any of clauses 118-121, wherein R’, R”, or both R’ and R’ are a halogen, preferably I.
Clause 128. The compound of any of clauses 118-121, having Formula (XXIV):
Clause 129. The compound of any of clauses 118-121, wherein the acridone compound is any one of the following compounds:
Clause 130. The compound of any of clauses 118-121, wherein the acridone compound is a compound of any one of Formulas:
OK
Clause 132. A method of functionalizing an acridone-containing compound at a 2,7- position of an acridone ring with iodine, comprising: dissolving an acridone-containing compound in a third solution; adding iodine monochloride to the third solution and stirring for about 6 h to about 18 h under pressure to produce the acridone-containing compound functionalized at the 2,7-positions in a mixture; dissolving the mixture in DMSO; and isolating and optionally purifying the diiodo-acridone compound.
Clause 133. The method of clause 132, wherein the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H.
Clause 134. The method of clause 132, wherein the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H and Li is — CH2CH2CH2 — .
Clause 135. The method of clause 132, wherein the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H, Li is CH2CH2CH2 — , and Q is -SO3H.
Clause 136. A method of producing an acridone disulfide of Formula (XXVI) comprising: reacting a dihalo-acridone with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridone disulfide product of Formula (XXVI), wherein Li, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (XXIII). Clause 137. The method of clause 136, wherein the dihalo-acridone is compound (35). Clause 138. The method of clauses 136 or 137, wherein the water-soluble thiol comprises mercaptopropanesulfonic acid (MSA).
Clause 139. The method of any of clauses 136-138, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
Clause 140. The method of any of clauses 136-139, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
Clause 141. The method of any of clauses 136-139, wherein the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino-l , 1 '- biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3).
Clause 142. The method of any of clauses 136-141, further comprising isolating and optionally purifying the acridone disulfide compound of Formula (XXVI).
Clause 143. A method of producing a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI), comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating the bis sulfoxide of Formula (XXVIII), sulfoxide sulfone of Formula (XXIX), and/or bis sulfone of Formula (XXXI) and optionally separating and purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
Clause 144. The method of clause 143, wherein the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
Clause 145. The method of clauses 143 or 144, wherein the oxidizing agent comprises mCPBA.
Clause 146. The method of any of clauses 143-145, wherein the compound of Formula (XXVI) is compound (39).
Clause 147. The method of any of clauses 143-146, wherein after addition of the oxidizing agent, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
Clause 148. A method of producing a sulfoxide of Formula (XXVII) comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; dropwise adding an eighth solution comprising an oxidizing agent to the contents of the container; stirring the contents of the container; quenching the reaction with dimethyl sulfide; and isolating the sulfoxide of Formula (XXVII) and optionally purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
Clause 149. The method of clause 148, wherein the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
Clause 150. The method of clauses 148 or 149, wherein the oxidizing agent comprises mCPBA.
Clause 151. The method of any of clauses 148-150, wherein the compound of Formula (XXVI) is compound (39).
Clause 152. The method of any of clauses 148-151, wherein the eighth solution further comprises at least one organic solvent.
Clause 153. The method of any of clauses 148-152, wherein after addition of the eighth solution, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
Clause 154. The method of any of clauses 148-153, wherein the dimethyl sulfide is added dropwise.
Clause 155. The method of clauses 154, wherein immediately after the addition of dimethyl sulfide, water is added.
Clause 156. A method of assessing and monitoring stage or phase of chronic Hepatitis B (HBV) infection or monitoring response to a treatment for chronic HBV in a subject, the method comprising the steps of: a) performing an assay to detect the presence or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject diagnosed with chronic HBV or receiving a treatment for chronic HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg-second detection antibody complex is formed; and b) assessing and monitoring stage or phase of chronic HBV infection or monitoring in response to the treatment for chronic HBV based on the presence or level of HBcAg and P- HBcAg in the at least one sample, wherein the first detectable label and second detectable label each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyh heterearylalkyh cycloalkylalkyl, or heterocycly lalkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or
unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCh, CF2C1, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=0)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2 wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, and heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
Clause 157. A method of detecting the presence of or level of Hepatitis B (HBV) infection, the method comprising the steps of: a) performing an assay to detect the presence of or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject suspected of having HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection
antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg-second detection antibody complex is formed; and b) detecting HBV infection based on the presence or level of HBcAg and P-HBcAg in the at least one sample, wherein the first detectable label and second detectable label each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
wherein:
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SOs-A, wherein A is selected from alkyd, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, or heterocyclylalkyl; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=0)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2 wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, and heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
Clause 158. The method of clause 1, wherein the analyte of interest is bHCG, SARS- CoV-2, hepatitis B core antigen (HBcAg), phosphorylated hepatitis B core antigen, or combinations thereof.
Clause 159. A method for determining the amount of an analyte of interest in a biological sample, the method comprising the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a first reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. performing, in the second reaction vessel, a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein: (i) the sandwich immunoassay and competitive immunoassay are performed simultaneously, or sequentially, in any order: (ii) the second detectable label is identical to the first detectable label; (iii) the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and (iv) the first detectable signal and the second detectable signal are detected by an instrument comprising a single detection component; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
Claims
1. A method for determining the amount of an analyte of interest in a biological sample, the method comprising the steps of: a. obtaining a calibration curve for the analyte of interest; b. performing, in a reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. performing, in the same reaction vessel, a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
2. The method of claim 1, wherein the calibration curve is obtained by the steps of: a. performing, in a reaction vessel, a sandwich immunoassay on the analyte of interest having a known concentration, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; b. performing, in the same reaction vessel, a competitive immunoassay on analyte of interest having a known concentration, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and c. calculating the signal ratio of first detectable signal and the second detectable signal; and
d. varying concentrations of the analyte of interest and repeating steps a-c to generate a calibration curve using the signal ratios generated in step c.
3. The method of claim 1 or claim 2, wherein the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
4. The method of any of claims 1 to 3, wherein the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a chromophore, a chemiluminescent label, or any combinations thereof.
5. The method of any of claims 1 to 4, wherein the first detectable signal comprises a first color and the second detectable signal comprises a second color.
6. The method of any of claims 1 to 4, wherein the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
7. The method of any of claims 1 to 6, wherein the method further comprises using a first detection component to detect the first detectable signal generated by the first detectable label and a second detection component to detect the second detectable signal generated by the second detectable label.
8. The method of claim 7, wherein the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
9. The method of any of claims 1 to 8, wherein the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”), that competes with the analyte of interest wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
10. The method of any of claims 1 to 8, wherein the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
11. The method of any of claims 1 to 10, wherein the first detection reagent or the second detection reagent is an antigen or an antibody.
12. The method of claims 1 to 11, wherein the analyte of interest and the competing analyte are each captured on a solid support.
13. The method of any of claims 1 to 12, wherein the capture reagent comprises an antibody or an antigen.
14. The method of any of claims 1 to 13, wherein the sandwich assay is performed in one step or two steps.
15. The method of any of claims 1 to 14, wherein the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained from the subject.
16. A system for determining the amount of an analyte of interest in a biological sample, the system comprising: a. a calibration curve for the analyte of interest; b. a sandwich immunoassay wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. a competitive immunoassay wherein the competitive immunoassay comprises a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein the second detectable label is not identical to the first detectable label and the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; d. a device comprising: i. a reaction vessel for performing the sandwich immunoassay and the competition assay; ii. a first detection component to determine the amount of the first detectable signal generated by the first detectable label; and iii. a second detection component to determine the amount of the second detectable signal generated by the second label, wherein: (i) the amount of the first detectable signal determined by the first detection component and the amount of the second detectable signal determined by the second detection component is used to calculate the signal ratio of first detectable signal and second detectable signal; and (ii) the signal ratio is used to determine the amount of the analyte of interest in the biological sample.
17. The system of claim 16, wherein the sandwich immunoassay and the competitive immunoassay are each performed simultaneously or sequentially.
18. The system of claim 16 or claim 17, wherein the first detectable label and second detectable label are each a fluorophore, a radioactive moiety, an enzyme, a biotin/avidin label, a chromophore, a chemiluminescent label, or any combinations thereof.
19. The system of any of claims 16 to 18, wherein the first detectable signal comprises a first color and the second detectable signal comprises a second color.
20. The system of any of claims 16 to 18, wherein the first detectable signal and second detectable signal are distinguishable based on reaction kinetics.
21. The system of any of claims 16 to 20, wherein the first detection component and second detection component each comprise a filter, wherein the filter on the first detection component and the filter on the second detection component are not identical.
22. The system of any of claims 16 to 21, wherein the competitive immunoassay further comprises an analyte labeled with a detectable label (“competing analyte”) that competes with the analyte of interest, wherein the competing analyte is the same analyte as the analyte of interest or an analog thereof.
23. The system of any of claims 16 to 21, wherein the first detection reagent, the second detection reagent or the first detection agent and the second detection reagent is an antigen or an antibody.
24. The system of any of claim 22 or claim 23, wherein the competing analyte comprises an analog of the analyte of interest and further wherein, the second detection reagent does not bind to the competing analyte.
25. The system of any of claims 16 to 24, wherein the capture reagent comprises an antibody or an antigen.
26. The system of any of claims 16 to 25, wherein the biological sample used in the sandwich immunoassay and the competitive immunoassay are the same biological sample obtained from the subject or are different biological samples obtained from the subject.
27. A compound of Formula (I) comprising
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyh heteroarylalkyh cycloalkylalkyl, or heterocycly lalkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or
unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCh, CF2C1, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=0)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2, wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents.
28. The compound of claim 27, wherein Y is nitrogen.
29. The compound of claim 28, wherein A is aryl that is unsubstituted or substituted with 1, 2, 3, 4, or 5 substituents selected from C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, and selenyl.
30. The compound of any of claims 27-29, wherein R2 is -COOZ.
31. The compound of claim 30, wherein Z is selected from hydrogen, C1-C4 alkyl, or N- hydroxysuccinimide.
32. The compound of any of claims 27-31, wherein Li and L2 are each independently C1-C4 alkylene.
33. The compound of any of claims 27-32, wherein Ra, Rb, Rc, Rd, Re, and Rf are each hydrogen.
34. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are hydrogen.
35. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are -S-L3- SO3H.
36. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are -S(=O)-L3- SO3H.
37. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are -SO2-L3- SO3H.
38. The compound of any of claims 27-33, wherein R’ is -SO2-L3-SO3H and R” is -S-L3- SO3H.
39. The compound of any of claims 27-33, wherein R’ is -S-L3-SO3H and R” is one of H, Cl, Br, I, -NO2, t-butyl, or -SO3H.
40. The compound of any of claims 27-33, wherein R’ is -S-L3-SO3H and R” is H.
41. The compound of any of claims 27-33, wherein R’ is -SO2-L3-SO3H and R” is one of Cl, Br, I, -NO2, t-butyl, or -SO3H.
42. The compound of any of claims 27-33, wherein R’ is -(S=O)-L3-SC>3H and R” is -S-L3- SO3H.
43. The compound of any of claims 27-33, wherein R’ is -(S=O)-L3-SO3H and R” is one of Cl, Br, I, -NO2, t-butyl, or -SO3H.
44. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are a substituted or unsubstituted phenyl group.
45. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are a phenyl group substituted with at least one of hydroxy, cyano, alkylamino, dialkylamino, or aminoalkyl.
46. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are a substituted or unsubstituted heteroaryl group.
47. The compound of any of claims 27-33, wherein R’, R”, or both R’ and R” are a substituted or unsubstituted heteroaryl group selected from pyrrolyl, furanyl, thienyl, or thienothiophenyl.
48. The compound of any of claims 27-33, wherein the compound has the Formula (Ila):
wherein Y, Ri, R2, Li, L2, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (I), and wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
53. The compound of any of claims 27-33, selected from:
54. A method of functionalizing an acridinium-containing compound at a 2- or a 2,7-position of an acridinium ring, said method comprising: reducing the acridinium ring to an acridan intermediate using a first solution comprising at least one reducing agent and at least one acid; and reacting the acridan intermediate with an electrophile to form a product comprising the acridinium functionalized at the 2- or the 2,7- position of the acridinium ring.
55. The method of claim 54, wherein the at least one acid comprises at least one of ammonium chloride, formic acid, acetic acid, benzoic acid, hydrofluoric acid, phosphoric acid,
sulfurous acid, hypochlorous acid, citric acid, carbonic acid, nitrous acid, oxalic acid, boric acid, ammonium sulfate, or ammonium oxalate.
56. The method of claims 54 or 55, wherein the at least one reducing agent comprises zinc or a hydride.
57. The method of claim 56, wherein the zinc comprises elemental zinc, a zinc alloy, or a combination of both.
58. The method of claim 56, wherein the hydride comprises sodium borohydride, lithium aluminum hydride, sodium cyanoborohydride, or sodium triacetoxyborohydride.
59. The method of any of claims 54-58, wherein the first solution further comprises a water- miscible alcohol, preferably ethanol.
60. The method of any of claims 54-59, wherein the acridinium ring is reduced at temperature in a range from about 50°C to about 90°C, preferably about 60°C to about 80°C.
61. The method of any of claims 54-60, wherein the acridinium-containing compound is represented by the compound of Formula (I) of any of claims 27-46.
62. The method of claim 61, wherein R’ = R” = H.
63. The method of any of claims 54-62, wherein the acridan intermediate is represented by
64. The method of any of claims 54-63, wherein the electrophile comprises at least one of Cl, Br, I, NO2, SO3H, or t-butyl.
65. The method of claim 64, wherein the electrophile is Cl and the products are Formula (IVa), (IVb), or both (IVa) and (IVb).
66. The method of claim 65, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with Cl at the 2- or the 2,7- position, or both, of the acridinium ring includes a chlorination reaction.
67. The method of claims 65 or 66, further comprising: mixing the acridan intermediate of Formula (III) with a second solution in a container, and stirring for about 12 hours to about 24 hours, preferably about 16 hours to about 20 hours; adding an aqueous solution of at least one oxidant to the container, in portions, over about 30 minutes to about 90 minutes, preferably about 45 minutes to about 75 minutes; and isolating and optionally purifying the product comprising the compound of Formula (IVa), (IVb), or both (IVa) and (IVb).
68. The method of claim 67, wherein the second solution comprises a chloride salt, N- chlorosuccinimide, and p-toluenesulfonic acid in a water/acetonitrile mixture.
69. The method of claims 67 or 68, wherein the oxidant comprises FeCh, 2,3-Dichloro-5,6- dicyano-l,4-benzoquinone (DDQ), chloranil, or ceric ammonium nitrate (CAN).
70. The method of claim 64, wherein the electrophile is Br and the products are Formula (Va), (Vb), or both (Va) and (Vb).
71. The method of claim 70, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with Br at the 2- or the 2,7- position, or both, of the acridinium ring includes a bromination reaction.
72. The method of claims 70 or 71, further comprising: dissolving the acridan intermediate in a third solution in a container; adding bromine to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Va), (Vb), or both (Va) and (Vb).
73. The method of claim 64, wherein the electrophile is I and the products are Formula (Via), (VIb), or both (Via) and (VIb).
74. The method of claim 73, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with I at the 2- or the 2,7- position, or both, of the acridinium ring includes an iodination reaction.
75. The method of claims 73 or 74, further comprising: dissolving the acridan intermediate in a third solution in a container; adding iodine monochloride to the container and stirring for about 30 minutes to about 1 hour; and isolating and optionally purifying the product comprising the compound of Formula (Via), (VIb), or both (Via) and (VIb).
76. The method of claim 64, wherein the electrophile is NO2 and the products are Formula (Vila), (Vllb), or both (Vila) and (Vllb).
77. The method of claim 76, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with NO2 at the 2- or the 2,7- position, or both, of the acridinium ring includes a nitration reaction.
78. The method of claims 76 or 77, further comprising: dissolving the acridan intermediate in a third solution in a container; adding nitric acid to the container; and isolating and optionally purifying the product comprising the compound of Formula (Vila), (Vllb), or both (Vila) and (Vllb).
79. The method of claim 64, wherein the electrophile is SO3H and the products are Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
80. The method of claim 79, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with SO3H at the 2- or the 2,7- position, or both, of the acridinium ring includes a sulfonation reaction.
81. The method of claims 79 or 80, further comprising: dissolving the acridan intermediate in a fourth solution in a container; adding chlorosulfonic acid to the container and stirring for about 5 minutes to about 30 minutes; adding water to the container and stirring for about 10 minutes to about 30 minutes; and isolating and optionally purifying the product comprising the compound of Formula (Villa), (Vlllb), or both (Villa) and (Vlllb).
82. The method of claim 64, wherein the electrophile is t-butyl and the products are Formula (IXa), (IXb), or both (IXa) and (IXb).
83. The method of claim 82, wherein a reaction of the acridan intermediate of Formula (III) to form a product comprising the acridinium functionalized with t-butyl at the 2- or the 2,7- position, or both, of the acridinium ring includes a Friedel-Craft alkylation reaction.
84. The method of claims 82 or 83, further comprising: dissolving the acridan intermediate in a fifth solution in a container; adding a Lewis acid to the container and heating at about 40°C to about 60°C for about 18-24 hours; reducing solvent in vacuo followed by the addition of a water/acetonitrile solution to the container; adding an aqueous solution of an oxidant to the container dropwise over time; and isolating and optionally purifying the product comprising the compound of Formula (IXa), (IXb), or both (IXa) and (IXb).
85. The method of any of claims 54-84, further comprising reacting the product comprising the acridinium functionalized at the 2-position with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (XVI).
86. The method of claim 85, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
87. The method of claims 85 or 86, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
88. The method of any of claims 85-87, wherein the reaction mixture further comprises N,N- diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
89. The method of any of claims 85-88, wherein the Pd-containing catalyst comprises 2-Di- tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1 , 1 '-biphenyl)] palladium(II) methanesulfonate (ZBuXPhos Pd G3).
90. The method of any of claims 85-89, further comprising isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
91. The method of any of claims 54-84, further comprising reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium disulfide compound of Formula (XVII).
92. The method of claim 91, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
93. The method of claims 91 or 92, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
94. The method of any of claims 91-93, wherein the reaction mixture further comprises N,N- diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
95. The method of any of claims 91-94, wherein the Pd-containing catalyst comprises 2-Di- terZ-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1 , 1 '-biphenyl)] palladium(II) methanesulfonate (tBuXPhos Pd G3).
96. The method of any of claims 92-95, further comprising isolating and optionally purifying the acrinidium monosulfide compound of Formula (XVI).
97. The method of any of claims 54-84, further comprising: reacting the product comprising the acridinium functionalized at the 2,7-positions with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium monosulfide compound of Formula (I), wherein R’ is -S-L3-SO3H and R” is selected from Cl, Br, I, -NO2, t- butyl, or -SO3H; oxidizing the sulfide moiety to a sulfone; reacting the R” group selected from Cl, Br, I, -NO2, t-butyl, or -SO3H with the reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acrinidium sulfone compound of Formula (XXI).
98. The method of claim 97, wherein the water soluble thiol comprises mercaptopropanesulfonic acid (MSA).
99. The method of claims 97 or 98, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
100. The method of any of claims 97-99, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
101. The method of any of claims 97-100, wherein the Pd-containing catalyst comprises 2- Di-terZ-butylphosphino-2',4',6'-triisopropyl-l , 1 '-biphenyl)-2-(2'-amino-l ,l'-biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
102. The method of any of claims 97-101, wherein the sulfide moiety is oxidized using meta-Chloroperoxybenzoic acid (mCPBA).
103. The method of any of claims 97-102, further comprising isolating and optionally purifying the acrinidium sulfone compound of Formula (XXI).
104. The method of any of claims 76-78, wherein the NO2 group(s) are reduced to amine group(s) using a reducing agent to obtain the compound of Formula (Xa) or (Xb).
105. The method of claim 104, wherein the reducing agent comprising SnCh.
106. A method of functionalizing an acridinium-containing compound at a 2- or a 2,7- position of an acridinium ring comprising: dissolving a reactant compound of one of Formulas (IVa), (IVb), (Va), (Vb), (Via) or (VIb), Pd(PPh3)4, and a boronic acid comprising A’ or a boronate ester comprising A’, wherein A’ is a substituted or unsubstituted aryl group or a substituted or unsubstituted heteroaryl group, in a seventh solution comprising acetonitrile, water, and DiEA, in a container;
stirring the contents of the container at about 30°C to about 50°C for about 10-30 minutes; and isolating and optionally purifying the product comprising the acridinium compound functionalized by A’ at the 2- or a 2,7-position of the acridinium ring.
107. The method of claim 106, wherein the acridinium compound is functionalized by:
(i) at least one phenyl group to produce a compound of Formula (Xia) or (Xlb);
(ii) at least one furanyl group to produce a compound of Formula (Xllla) or (Xlllb);
(iii) at least one thienyl group to produce a compound of Formula (XlVa) or (XlVb); or
(iv) at least one thienothiophenyl group to produce a compound of Formula (XV), wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl, arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
108. The method of claim 106, wherein the acridinium compound is functionalized by at least one pyrrolyl group to produce a compound of Formula (Xlla) or (Xllb), wherein the boronic acid comprising A’ or the boronate ester comprising A’ is tert-butyloxycarbonyl (Boc)- protected, and wherein the method comprises after the stirring step: isolating the product comprising the acridinium-containing compound comprising a Boc-protected pyrrolyl group; dissolving the acridinium-containing compound comprising the Boc-protected pyrrolyl group in dichloromethane and trifluoroacetic acid for about 2-6 hours; removing the solvent in vacuo,- and optionally purifying the product comprising the compounds of Formula Xlla or Xllb, wherein Rg and Rg’ are each independently selected from hydrogen, halogen, =O, =S, cyano, nitro, alkyl, alkenyl, alkynyl, haloalkyl, haloalkoxy, heteroalkyl, cycloalkyl, cycloalkenyl, aryl, heteroaryl, heterocycle, cycloalkylalkyl, heteroarylalkyl, arylalkyl, hydroxy, hydroxyalkyl, alkoxy, alkoxyalkyl, alkylene, aryloxy, arylalkyloxy, amino, alkylamino, dialkylamino, acylamino, aminoalkyl, arylamino, sulfonylamino, sulfinylamino, sulfonyl, alkylsulfonyl,
arylsulfonyl, aminosulfonyl, sulfinyl, alkylthio, carboxy ( — COOH), ketone, amide, carbamate, phosphoryl, selenyl, or acyl.
109. The method of any of claims 106-108, wherein the reactant compound comprises iodo- CPSP or diiodo-CPSP.
110. A method of producing a sulfoxide of Formula (XVIII) or (XIX), said method comprising: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating and optionally purifying the sulfoxide compound of Formula (XVIII) or (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
111. The method of claim 110, wherein the at least one organic solvent comprises tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
112. The method of claims 110 or 111, wherein the oxidizing agent comprises mCPBA.
113. The method of any of claims 110-112, wherein the compound of Formula (XVII) is CPSP3.
114. A method of producing a BisSulfone compound of Formula (XXII) comprises: dissolving a compound of Formula (XVII) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; quenching an oxidation reaction with dimethyl sulfide and adding an amount of water; and isolating and optionally purifying the BisSulfone compound of Formula (XIX), wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
115. The method of claim 114, wherein the at least one organic solvent comprises tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
116. The method of claims 114 or 115, wherein the oxidizing agent comprises mCPBA.
117. The method of any of claims 114-116, wherein the compound of Formula (XVII) is CPSP3.
Q is -SO3Z or H;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Li is selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalky 1, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -0-CF3, -0-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*,
-C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2 wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, or heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein at least one of R’ or R” has to be -S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
119. The compound of claim 118, wherein Q is -SO3Z, wherein Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl, optionally substituted with 1, 2, 3, 4, or 5 substituents, preferably -SO3H.
120. The compound of claim 118 or 119, wherein Li is a C1-C10 alkylene, preferably — CH2CH2CH2— .
121. The compound of any of claims 118-120, wherein Ra, Rb, Rc, Rd, Re, and Rf are each hydrogen.
122. The compound of any of claims 118-121, wherein R’, R”, or both R’ and R” are -S-L3- SO3H.
123. The compound of any of claims 118-121, wherein R’, R”, or both R’ and R” are - S(=O)-L3-SO3H.
124. The compound of any of claims 118-121, wherein R’, R”, or both R’ and R” are - SO2-L3-SO3H.
125. The compound of any of claims 118-121, wherein R’ is -SO2-L3-SO3H and R” is - S-L3-SO3H.
126. The compound of any of claims 118-121, wherein R’ is -SCh-Ls-SChH and R” is - S(-O)-L3-SO3H.
127. The compound of any of claims 118-121, wherein R’, R”, or both R’ and R’ are a halogen, preferably I.
130. The compound of any of claims 118-121, wherein the acridone compound is a compound of any one of Formulas:
132. A method of functionalizing an acridone-containing compound at a 2,7-position of an acridone ring with iodine, comprising: dissolving an acridone-containing compound in a third solution; adding iodine monochloride to the third solution and stirring for about 6 h to about 18 h under pressure to produce the acridone-containing compound functionalized at the 2,7-positions in a mixture; dissolving the mixture in DMSO; and isolating and optionally purifying the diiodo-acridone compound.
133. The method of claim 132, wherein the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H.
134. The method of claim 132, wherein the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H and Li is — CH2CH2CH2— .
135. The method of claim 132, wherein the acridone-containing compound comprises Formula (XXIII), wherein R’ = R” = H, Li is CH2CH2CH2 , and Q is -SO3H.
136. A method of producing an acridone disulfide of Formula (XXVI) comprising: reacting a dihalo-acridone with a reaction mixture comprising water soluble thiol, in the presence of a Pd-containing catalyst, to produce an acridone disulfide product of Formula (XXVI), wherein Li, L3, Q, Ra, Rb, Rc, Rd, Re, and Rf are defined above for Formula (XXIII).
137. The method of claim 136, wherein the dihalo-acridone is compound (35).
138. The method of claims 136 or 137, wherein the water-soluble thiol comprises mercaptopropanesulfonic acid (MSA).
139. The method of any of claims 136-138, wherein the reaction mixture further comprises at least one alkyl amine, at least one organic solvent, or water.
140. The method of any of claims 136-139, wherein the reaction mixture further comprises N,N-diisopropylethylamine (DIEA), tetrahydrofuran (THF) or water.
141. The method of any of claims 136-139, wherein the Pd-containing catalyst comprises 2-Di-tert-butylphosphino-2',4',6'-triisopropyl- 1 , 1 '-biphenyl)-2-(2'-amino- 1,1'- biphenyl)] palladium(II) methanesulfonate (fBuXPhos Pd G3).
142. The method of any of claims 136-141, further comprising isolating and optionally purifying the acridone disulfide compound of Formula (XXVI).
143. A method of producing a bis sulfoxide of Formula (XXVIII), a sulfoxide sulfone of Formula (XXIX), and/or a bis sulfone of Formula (XXXI), comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; adding an oxidizing agent to the contents of the container with stirring; and isolating the bis sulfoxide of Formula (XXVIII), sulfoxide sulfone of Formula (XXIX), and/or bis sulfone of Formula (XXXI) and optionally separating and purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
144. The method of claim 143, wherein the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
145. The method of claims 143 or 144, wherein the oxidizing agent comprises mCPBA.
146. The method of any of claims 143-145, wherein the compound of Formula (XXVI) is compound (39).
147. The method of any of claims 143-146, wherein after addition of the oxidizing agent, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
148. A method of producing a sulfoxide of Formula (XXVII) comprising: dissolving a compound of Formula (XXVI) in at least one organic solvent in a container; cooling the contents of the container to about 5°C to about -5°C; dropwise adding an eighth solution comprising an oxidizing agent to the contents of the container; stirring the contents of the container; quenching the reaction with dimethyl sulfide; and isolating the sulfoxide of Formula (XXVII) and optionally purifying same, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, optionally substituted with 1, 2, 3, 4, or 5 substituents.
149. The method of claim 148, wherein the at least one organic solvent comprises at least one of tetrahydrofuran (THF), acetonitrile, dioxane, dimethylsulfoxide, and dimethylformamide (DMF).
150. The method of claims 148 or 149, wherein the oxidizing agent comprises mCPBA.
151. The method of any of claims 148-150, wherein the compound of Formula (XXVI) is compound (39).
152. The method of any of claims 148-151, wherein the eighth solution further comprises at least one organic solvent.
153. The method of any of claims 148-152, wherein after addition of the eighth solution, the contents are stirred for about 10-30 minutes at about 5°C to about -5°C and thereafter allowed to warm to room temperature over the course of about 40-80 minutes.
154. The method of any of claims 148-153, wherein the dimethyl sulfide is added dropwise.
155. The method of claim 154, wherein immediately after the addition of dimethyl sulfide, water is added.
156. A method of assessing and monitoring stage or phase of chronic Hepatitis B (HBV) infection or monitoring response to a treatment for chronic HBV in a subject, the method comprising the steps of: a) performing an assay to detect the presence or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained ftom a subject diagnosed with chronic HBV or receiving a treatment for chronic HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg-second detection antibody complex is formed; and b) assessing and monitoring stage or phase of chronic HBV infection or monitoring in response to the treatment for chronic HBV based on the presence or level of HBcAg and P- HBcAg in the at least one sample, wherein the first detectable label and second detectable label each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
wherein:
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycioalkyl, heterocyclyl, arylaikyi, he tern aryl alky k cycloalkylalkyl, or heterocycly ialkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalky 1, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCI3, CF2CI, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, -
S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2J -O-P(=O)(OR*)2J -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2 wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, and heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
157. A method of detecting the presence of or level of Hepatitis B (HBV) infection, the method comprising the steps of: a) performing an assay to detect the presence of or level of Hepatitis B core antigen (HBcAg) and phosphorylated Hepatitis B core antigen (P-HBcAg) in at least one sample obtained from a subject suspected of having HBV, wherein the assay comprises contacting the at least one sample with (a) at least one first capture antibody that specifically binds to HBcAg and at least one first detection antibody comprising a first detectable label that binds to an epitope not bound by the first capture antibody such that a first capture antibody-HBcAg-first detection antibody complex is formed; and (b) at least one least one second antibody that specifically binds to P-HBcAg, and at least one second detection antibody comprising a second detectable label that binds to an epitope not bound by the second capture antibody such that a second capture antibody-P-HBcAg-second detection antibody complex is formed; and b) detecting HBV infection based on the presence or level of HBcAg and P-HBcAg in the at least one sample, wherein the first detectable label and second detectable label each comprise, independently, a compound of Formula (I) or a compound of Formula (XXIII):
Y is selected from nitrogen, oxygen, and sulfur; when Y is nitrogen, R1 is -SO2-A, wherein A is selected from alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyh heteroarylalkyh cycloalkylalkyl, or heterocycly lalkyl ; when Y is oxygen or sulfur, R1 is absent;
Q is -SO3Z or H;
Li and L2 are each independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene;
R2 is selected from -SO3Z and -COOZ;
Z is selected from hydrogen, alkyl, alkenyl, aikynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, or heteroalkyl;
Ra, Rb, Rc, Rd, Re, and Rf are each independently selected from hydrogen, C1-C4 alkyl, C1-C4 alkoxy, Ci-C4haloalkyl, C1-C4 haloalkoxy, halo, hydroxy, cyano, nitro, amino, carboxy, sulfonyl, phosphoryl, or selenyl;
R’ and R” are each independently selected from H, Cl, Br, I, -NO2, -NH2, t-butyl, -SO3H, - S(=O)-L3-X, -S-L3-X, -SO2-L3-X, substituted or unsubstituted aryl groups, and substituted or
unsubstituted heteroaryl groups, wherein L3 is independently selected from alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene, wherein X is selected from -F, -Cl, -Br, -I, -OH, -OR*, - NH2, -NHR*, -N(R*)2, -N(R*)3, -N(R*)-OH, -N(->O)(R*)2, -O-N(R*)2, -N(R*)-O-R*, -N(R*)- N(R*)2, -C=N-R*, -N=C(R*)2, -C=N-(R*)2, -C(=NR*)(-N(R*)2), -C(H)(=N-OH), -SH, -SR*, - CN, -NC, -CHF2, -CCh, CF2C1, -CF3, -O-CF3, -O-CHF2, -O-SO3, -NO, -NO2, -NO3, -O-NO, -O- ON2, -N3, -N2-R*, -Si(R*)3, -C(=O)-R*, -CHO, -CO2H, -CO2R*, -C(=O)-S-R*, -O-(C=O)-H, - O(C=O)-R*, -S-C(=O)-R*, -C(=O)-NH2, -C(=O)-NHR*, -C(=O)-N(R*)2, -C(=O)-NHNH2, -O- C(C=O)-NHNH2, -C(=S)-NH2, -C(=S)-NHR*, -C(=S)-N(R*)2, -N(R*)-CHO, -N(R*)-C(=O)-R*, -C(=NR*)-OR*, -SCN, -NCS, -NSO, -SSR*, -N(R*)-C(=O)-NH2, -N(R*)-C(=O)-NHR*, - N(R*)-C(=O)-N(R*)2, -N(R*)-C(=S)-NH2, -N(R*)-C(=S)-NHR*, -N(R*)-C(=S)-N(R*)2, - S(=O)-R*, -S(=O)2-R*, -O-S(=O)2-R*, -S(=O)2-OR*, -O-S(=O)2-OR*, -NR*-S(=O)2-R*, - S(=O)2-N(R*)2, -P(R*)2, -O-P(=O)(OR*)2, -P(=O)(OR*)2, -PO3H, -O-PO3H, or -P(=O)(OH)- N(R*)2 wherein each R* is independently selected from H, alkyl, heteroalkyl, cycloalkyl, heterocycloalkyl, alkenyl, alkynyl, aryl, and heteroaryl; wherein each alkyl, alkenyl, alkynyl, aryl, heteroaryl, cycloalkyl, heterocyclyl, arylalkyl, heteroarylalkyl, cycloalkylalkyl, heterocyclylalkyl, aryloxy, heteroalkyl, alkylene, alkenylene, alkynylene, heteroalkylene, heteroalkenylene, heteroalkynylene, arylene, heteroarylene, cycloalkylene, or heterocycloalkylene is independently optionally substituted with 1, 2, 3, 4, or 5 substituents, and wherein when the compound is that of Formula (XXIII), at least one of R’ or R” has to be - S(=O)-L3-X, -S-L3-X, or -SO2-L3-X.
158. The method of claim 1, wherein the analyte of interest is bHCG, SARS-CoV-2, hepatitis B core antigen (HBcAg), phosphorylated hepatitis B core antigen, or combinations thereof.
159. A method for determining the amount of an analyte of interest in a biological sample, the method comprising the steps of: a. obtaining a calibration curve for the analyte of interest;
b. performing, in a first reaction vessel, a sandwich immunoassay on analyte of interest, wherein the sandwich immunoassay comprises a capture reagent and a first detection reagent comprising a first detectable label capable of generating a first detectable signal; c. performing, in the second reaction vessel, a competitive immunoassay on analyte of interest, wherein the competitive immunoassay comprises a capture reagent and a second detection reagent comprising a second detectable label capable of generating a second detectable signal and further wherein: (i) the sandwich immunoassay and competitive immunoassay are performed simultaneously, or sequentially, in any order: (ii) the second detectable label is identical to the first detectable label; (iii) the capture reagent used in the sandwich immunoassay and the competitive immunoassay are identical; and (iv) the first detectable signal and the second detectable signal are detected by an instrument comprising a single detection component; d. calculating the signal ratio of first detectable signal and the second detectable signal; and e. determining the amount of the analyte in the biological sample from a subject based on the signal ratio determined in step d and the calibration curve.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363615305P | 2023-12-28 | 2023-12-28 | |
| US63/615,305 | 2023-12-28 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2025145020A2 true WO2025145020A2 (en) | 2025-07-03 |
| WO2025145020A3 WO2025145020A3 (en) | 2025-08-07 |
Family
ID=94478705
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/062078 Pending WO2025145020A2 (en) | 2023-12-28 | 2024-12-27 | Methods, systems, compounds, and kits for determining an analyte of interest in a test sample |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025145020A2 (en) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4554101A (en) | 1981-01-09 | 1985-11-19 | New York Blood Center, Inc. | Identification and preparation of epitopes on antigens and allergens on the basis of hydrophilicity |
| US5006309A (en) | 1988-04-22 | 1991-04-09 | Abbott Laboratories | Immunoassay device with liquid transfer between wells by washing |
| US5089424A (en) | 1988-06-14 | 1992-02-18 | Abbott Laboratories | Method and apparatus for heterogeneous chemiluminescence assay |
| WO2001058956A2 (en) | 2000-02-10 | 2001-08-16 | Abbott Laboratories | Antibodies that bind human interleukin-18 and methods of making and using |
| US20060134713A1 (en) | 2002-03-21 | 2006-06-22 | Lifescan, Inc. | Biosensor apparatus and methods of use |
| US9239284B2 (en) | 2007-12-19 | 2016-01-19 | Singulex, Inc. | Scanning analyzer for single molecule detection and methods of use |
| WO2016161400A1 (en) | 2015-04-03 | 2016-10-06 | Abbott Laboratories | Devices and methods for sample analysis |
| WO2016161402A1 (en) | 2015-04-03 | 2016-10-06 | Abbott Laboratories | Devices and methods for sample analysis |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ATE423101T1 (en) * | 1998-08-11 | 2009-03-15 | Siemens Healthcare Diagnostics | NEAR INFRARED CHEMILUMINESCENT ACRIDINIUM DERIVATIVES AND THEIR USE |
| WO2003023360A2 (en) * | 2001-09-10 | 2003-03-20 | Meso Scale Technologies, Llc | Methods and apparatus for conducting multiple measurements on a sample |
| WO2016090244A2 (en) * | 2014-12-05 | 2016-06-09 | Mbio Diagnostics, Inc. | Methods and devices for performing high dynamic range immunoassays |
-
2024
- 2024-12-27 WO PCT/US2024/062078 patent/WO2025145020A2/en active Pending
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4554101A (en) | 1981-01-09 | 1985-11-19 | New York Blood Center, Inc. | Identification and preparation of epitopes on antigens and allergens on the basis of hydrophilicity |
| US5006309A (en) | 1988-04-22 | 1991-04-09 | Abbott Laboratories | Immunoassay device with liquid transfer between wells by washing |
| US5089424A (en) | 1988-06-14 | 1992-02-18 | Abbott Laboratories | Method and apparatus for heterogeneous chemiluminescence assay |
| WO2001058956A2 (en) | 2000-02-10 | 2001-08-16 | Abbott Laboratories | Antibodies that bind human interleukin-18 and methods of making and using |
| US20060134713A1 (en) | 2002-03-21 | 2006-06-22 | Lifescan, Inc. | Biosensor apparatus and methods of use |
| US9239284B2 (en) | 2007-12-19 | 2016-01-19 | Singulex, Inc. | Scanning analyzer for single molecule detection and methods of use |
| WO2016161400A1 (en) | 2015-04-03 | 2016-10-06 | Abbott Laboratories | Devices and methods for sample analysis |
| WO2016161402A1 (en) | 2015-04-03 | 2016-10-06 | Abbott Laboratories | Devices and methods for sample analysis |
Non-Patent Citations (20)
| Title |
|---|
| BREIMAN, L., MACHINE LEARNING, vol. 45, 2001, pages 5 - 32 |
| BREIMAN, L.FRIEDMAN, J. H.OLSHEN, R. A.STONE, C. J.: "Classification and regression trees", 1984, WADSWORTH |
| CHOTHIA ET AL., NATURE, vol. 342, 1989, pages 877 - 883 |
| CHOTHIALESK, J. MOL. BIOL., vol. 196, 1987, pages 901 - 917 |
| DUDA, R. O.HART, P. E.STORK, D. G.: "Pattern Classification", 2001, WILEY INTERSCIENCE |
| FRIEDMAN, J. H., J. OF THE AMERICAN STATISTICAL ASSOCIATION, vol. 84, 1989, pages 165 - 75 |
| FURNISSHANNAFORDSMITHTATCHELL: "Vogel's Textbook of Practical Organic Chemistry", 1989, LONGMAN SCIENTIFIC & TECHNICAL |
| HAUGLAND: "Handbook of Fluorescent Probes and Research Chemicals", 1996, MOLECULAR PROBES, INC. |
| HOLLIGER ET AL., PROC. NATL. ACAD. SCI. USA, vol. 90, no. 14, 1993, pages 6444 - 6448 |
| KYTE ET AL., J. MOL. BIOL., vol. 157, 1982, pages 105 - 132 |
| MACCALLUM, J. MOL. BIOL., vol. 262, no. 5, 1996, pages 732 - 745 |
| MILSTEIN ET AL., NATURE, vol. 305, no. 5934, 1983, pages 537 - 540 |
| PADLAN, FASEB J., vol. 9, 1995, pages 133 - 139 |
| PEPE, M. S.: "The Statistical Evaluation of Medical Tests for Classification and Prediction", OXFORD STATISTICAL SCIENCE SERIES, vol. 28, 2003 |
| POLAKVAN NOORDEN: "Introduction to Immunocytochemistry", 1997, SPRINGER VERLAG |
| PURE APPL. CHEM., vol. 45, 1976, pages 13 - 30 |
| RUCZINSKI, J. OF COMPUTATIONAL AND GRAPHICAL STATISTICS, vol. 12, 2003, pages 475 - 511 |
| STAERZ ET AL., NATURE, vol. 314, no. 6012, 1985, pages 628 - 631 |
| V. PANDARAS ET AL., TETRAHEDRON LETTERS, vol. 54, no. 35, 2013, pages 4712 - 4716 |
| WU, C. ET AL., NATURE BIOTECHNOLOGY, vol. 25, no. 11, 2007, pages 1290 - 1297 |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2025145020A3 (en) | 2025-08-07 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP6236519B2 (en) | Assays with a wide dynamic range | |
| EP3349011B1 (en) | Liver cancer test method | |
| JP7675063B2 (en) | Chemiluminescent Compounds for Multiplexing | |
| Ye et al. | Colloidal gold-based immunochromatographic biosensor for quantitative detection of S100B in serum samples | |
| US11047854B2 (en) | Methods for reducing noise in signal-generating digital assays | |
| RU2636822C2 (en) | Method for multispecific binding agent detection | |
| US11079395B2 (en) | Methods for predicting major adverse cardiovascular events in subjects with coronary artery disease | |
| EP3919509B1 (en) | Method for immunological analysis of free aim in biological sample | |
| Gholami et al. | A paper-based optical sensor for the screening of viruses through the cysteine residues of their surface proteins: A proof of concept on the detection of coronavirus infection | |
| WO2025145020A2 (en) | Methods, systems, compounds, and kits for determining an analyte of interest in a test sample | |
| EP3129785B1 (en) | Diagnosis of cancer by detecting dimeric il-18 | |
| JP7485755B2 (en) | Methods for detecting assay interferents and increasing dynamic range - Patents.com | |
| KR20260003222A (en) | Diagnosis of terminal hepatocellular carcinoma | |
| Dong et al. | Colloidal gold immunochromatographic strips for the detection of kynurenine in clinical samples | |
| JP7315965B2 (en) | Method for detecting viral liver cancer | |
| Darwish et al. | A highly sensitive automated flow immunosensor based on kinetic exclusion analysis for determination of the cancer marker 8-hydroxy-2′-deoxyguanosine in urine | |
| Cho et al. | Minimum-step immuno-analysis based on continuous recycling of the capture antibody | |
| JPWO2014057528A1 (en) | Method for detecting lung squamous cell carcinoma | |
| WO2025198859A1 (en) | Synthesis of thyroid hormone derivatives and their role as calibrators for thyroid hormone immunoassays | |
| CN121511407A (en) | Diagnosis of advanced hepatocellular carcinoma | |
| WO2019175254A1 (en) | Reduction of interferences in immunoassays | |
| HK1208731B (en) | Method for the detection of a multispecific binder |