WO2025132928A1 - Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid - Google Patents
Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid Download PDFInfo
- Publication number
- WO2025132928A1 WO2025132928A1 PCT/EP2024/087612 EP2024087612W WO2025132928A1 WO 2025132928 A1 WO2025132928 A1 WO 2025132928A1 EP 2024087612 W EP2024087612 W EP 2024087612W WO 2025132928 A1 WO2025132928 A1 WO 2025132928A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- composition
- acid
- chosen
- compounds
- mixtures
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/08—Preparations for bleaching the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/19—Cosmetics or similar toiletry preparations characterised by the composition containing inorganic ingredients
- A61K8/22—Peroxides; Oxygen; Ozone
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/365—Hydroxycarboxylic acids; Ketocarboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/40—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing nitrogen
- A61K8/44—Aminocarboxylic acids or derivatives thereof, e.g. aminocarboxylic acids containing sulfur; Salts; Esters or N-acylated derivatives thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/42—Colour properties
- A61K2800/43—Pigments; Dyes
- A61K2800/432—Direct dyes
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/80—Process related aspects concerning the preparation of the cosmetic composition or the storage or application thereof
- A61K2800/88—Two- or multipart kits
- A61K2800/884—Sequential application
Definitions
- TITLE Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid
- the present invention relates to a process for lightening keratin fibers, in particular human keratin fibers such as the hair, using one or more peroxygenated salts, one or more osmolytes and one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof.
- tone height is generally used to characterize the degree or level of lightening.
- tone is based on the classification of natural shades, one tone separating each shade from the shade immediately following or preceding it. This definition and the classification of natural shades are well known to hairstyling professionals and are published in the book Sciences des.s capillaires [The Science of Hair Care] by Charles Zviak, 1988, published by Masson, pages 215 and 278.
- the tone heights range from 1 (black) to 10 (very light blond), one unit corresponding to one tone; the higher the figure, the lighter the shade.
- Lightening thus allows the head of hair to be given a lighter tone height than its original natural tone.
- the processes for lightening the hair generally consist in using an aqueous composition comprising at least one oxidizing agent, under alkaline pH conditions in the vast majority of cases.
- This oxidizing agent has the role of degrading the melanin of the hair, which, depending on the nature of the oxidizing agent present, leads to more or less pronounced lightening of the fibers.
- the oxidizing agent is generally hydrogen peroxide.
- peroxygenated salts for instance persulfates
- these peroxygenated salts are contained in compositions which, at the time of use, are mixed with an aqueous composition comprising hydrogen peroxide.
- an alkaline agent is used. This alkaline agent also causes swelling of the keratin fibers, with opening of the scales, which favors penetration of the oxidizing agent into the fiber, and thus increases the efficacy of the reaction.
- alkaline agents and peroxygenated salts may impair the quality of the hair.
- the essential causes of this deterioration in hair quality are a reduction in its cosmetic properties, such as sheen, and a deterioration in its mechanical properties, more particularly a deterioration in its mechanical strength, which may also be reflected by an increase in its porosity.
- the hair is weakened and may become brittle during subsequent treatments such as blow-drying.
- the lightening of dark hair is thus particularly delicate, as it requires the use of a large amount of peroxygenated salts if it is desired to significantly lighten the hair, which can weaken or even damage it, notably leading to hair manageability problems.
- one of the objects of the present invention is to propose a process for lightening keratin fibers, preferably human keratin fibers such as the hair, which does not have the drawbacks mentioned above, i.e. which is capable of reducing hair damage while at the same time leading to very good lightening performance.
- the invention also relates to a composition
- a composition comprising:
- the invention also relates to a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition comprising (a) one or more peroxygenated salts, (b) one or more osmolytes and (c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and at least a second compartment containing an oxidizing composition comprising one or more additional oxidizing agents, different from the peroxygenated salts.
- the invention also relates to a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition comprising (a) one or more peroxygenated salts, a second compartment comprising (b) one or more osmolytes and (c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and optionally at least a third compartment containing an oxidizing composition comprising one or more additional oxidizing agents, different from the peroxygenated salts.
- the process according to the invention allows hair damage to be reduced without impacting on the lightening performance. It notably allows a high level of lightening to be obtained, of up to nine tones, while at the same time reducing hair manageability problems.
- FIG .1 depicts the structure of certain examples of osmolytes that may be used in the process according to the invention.
- the process for lightening keratin fibers according to the invention uses compounds (a), (b), (c) and optionally (d) described hereinbelow.
- the process according to the invention uses one or more peroxygenated salts.
- the peroxygenated salts are chosen from persulfates; perborates; alkali metal, alkaline-earth metal or ammonium percarbonates; magnesium peroxide; and mixtures thereof.
- the process according to the invention comprises the use of at least one persulfate.
- persulfates also known as peroxysulfates
- SO 5 2- anions peroxomonosulfate anion
- S 2 O 8 2 ' anions peroxodisulfate anion
- the persulfates according to the invention are chosen from peroxodisulfates.
- the process according to the invention comprises the use of at least one peroxygenated salt chosen from persulfates; preferably from alkali metal persulfates, alkaline-earth metal persulfates, ammonium persulfates, and mixtures thereof; more preferentially from (bis)tetrabutylammonium persulfate, barium persulfate, magnesium persulfate, calcium persulfate, sodium persulfate, potassium persulfate, ammonium persulfate, and mixtures thereof; even more preferentially from sodium persulfate, potassium persulfate, ammonium persulfate, and mixtures thereof; better still from potassium persulfate, ammonium persulfate, and mixtures thereof.
- persulfates preferably from alkali metal persulfates, alkaline-earth metal persulfates, ammonium persulfates, and mixtures thereof; more preferentially from (bis)te
- the peroxygenated salt(s) used as compound (a) in one or more compositions that are useful in the process according to the invention preferably represent from 1% to 70% by weight, more preferentially from 5% to 65% by weight, even more preferentially from 10% to 60% by weight, and better still from 20% to 55%, or even from 30% to 55% by weight, relative to the total weight of the composition by weight, relative to the total weight of the composition(s) containing them.
- the persulfate(s) used as compound(s) (a) in one or more compositions that are useful in the process according to the invention preferably represent from 1% to 70% by weight, more preferentially from 5% to 65% by weight, even more preferentially from 10% to 60% by weight, and better still from 20% to 55%, or even from 30% to 55% by weight, relative to the total weight of the composition by weight, relative to the total weight of the composition(s) containing them.
- Osmolytes are organic molecules, generally of low molecular weight.
- osmolyte does not include carboxylic amino acids, sulfonic amino acids or salts thereof.
- Osmolytes may be chosen from:
- R 2 and R 3 which may be identical or different, represent a C1-C4 alkyl group, preferably a C1-C2 alkyl group, preferably methyl;
- - A is N or S
- - m is 0 or 1 ;
- - n is 0 or 1 ;
- - X is a divalent, linear or branched, saturated or unsaturated alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH 2 ); and
- - Y- is -COO- or -OSO 3 _ ; it being understood that:
- R 4 represents a hydrogen atom or a saturated, linear or branched C1-C8 alkyl group, preferably a C1-C4 alkyl group; and R 5 forms, with the nitrogen atom, a saturated 5- to 8-membered, preferably 5- to 6-membered, heterocycle optionally substituted with one or more groups chosen from hydroxyl or C1-C4 alkyl; and
- the polyols may be chosen from C2-C16 polyols, such as C2-C12 or C2-C8 polyols.
- Useful polyols may optionally be chosen from diols and triols, and may optionally be linear or branched, saturated or unsaturated, and substituted or unsubstituted. Any stereoisomer of the polyols may be used.
- the polyols are chosen from glycerol, mannitol, xylitol, ribitol, pinitol, myoinositol, sorbitol and mixtures thereof.
- the sugars may be chosen from C3-C6 monosaccharides, for instance pentoses and/or derivatives thereof.
- the sugars may optionally be chosen from xylose, arabinose, ribose, 2-deoxyribose, ribulose, deoxyribulose, arabinose, xylulose, allose, altrose, glucose (including dextrose), glucosamine, mannose, gulose, idose, galactose, talose, sorbose, psicose, fructose, tagatose or combinations of two or more of these sugars.
- the sugars may be chosen from disaccharides, for example sucrose (also known as saccharose), maltose, lactose, cellobiose, trehalose, dextran, or from polysaccharides, for example maltotriose, starch, dextrins, cellulose, glycogen or combinations of two or more thereof.
- disaccharides for example sucrose (also known as saccharose), maltose, lactose, cellobiose, trehalose, dextran, or from polysaccharides, for example maltotriose, starch, dextrins, cellulose, glycogen or combinations of two or more thereof.
- the polyamines that may be used in the process according to the invention may constitute functional groups of primary and/or secondary and/or tertiary and/or quaternary amines.
- the polyamines may be chosen from diamines, triamines, tetramines, pentamines and polymeric polyamines or polyimines, for instance hexamethylenediamine, diethylenetriamine, diethylenetetramine, polyethylenimine (PEI), polyvinylamine, polyetheramine, polylysine, ethylenediamine, 1 ,3-diaminopropane, cadaverine, spermidine, spermine, putrescine, tetraethylmethylenediamine, triethylenetetramine, or combinations of two or more of these elements.
- the polyamines are chosen from putrescine, spermidine, spermine and mixtures thereof.
- the process according to the invention uses at least one osmolyte chosen from the compounds of formula (I) which are derivatives of zwitterionic amino acids bearing a quaternary ammonium group and comprising a total of from 1 to 12 carbon atoms, better still from 2 to 10 carbon atoms, even better still from 3 to 8 carbon atoms.
- the process according to the invention uses at least one compound of formula (I) in which Y- is -COO-.
- the process according to the invention uses at least one compound of formula (I) in which Ri and R 2 are identical and represent methyl, A is N and Y is -COO-.
- X is a divalent, linear or branched, saturated or unsaturated alkyl group containing from 1 to 6 carbon atoms, preferably 1 to 4 carbon atoms, preferably 1 to 2 carbon atoms.
- the process according to the invention uses at least one compound of formula (I) in which X is a divalent, linear or branched, preferably saturated, alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, optionally substituted with a group chosen from hydroxyl or amino.
- the process according to the invention uses at least one compound of formula (I) in which X is a divalent, linear or branched, preferably saturated, alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, preferably from 1 to 2 carbon atoms (unsubstituted), such as methylene or ethylene.
- X is a divalent, linear or branched, preferably saturated, alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, preferably from 1 to 2 carbon atoms (unsubstituted), such as methylene or ethylene.
- R 4 and R 5 which may be identical or different, may
- the process according to the invention uses at least one compound of formula (I) in which R 4 represents a hydrogen atom.
- the process according to the invention uses at least one compound of formula (I) in which R 5 represents a hydrogen atom or a saturated, linear or branched C1-C6, preferably C1-C4, hydrocarbon-based chain, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH 2 ), -SH, -COOH or -CONH 2 .
- R 5 represents a hydrogen atom or a saturated, linear or branched C1-C6, preferably C1-C4, hydrocarbon-based chain, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH 2 ), -SH, -COOH or -CONH 2 .
- the compounds of formula (I) are chosen from one of the compounds indicated in FIG.1.
- the compounds of formula (I) are chosen from valine betaine, glutamic acid betaine, glutamine betaine, trimethyllysine, glycine betaine (trimethylglycine), histidine betaine, alanine betaine, choline sulfate, pipecolic acid betaine, proline betaine, hydroxyproline betaine, tyrosine betaine, phenylalanine betaine, tryptophan betaine, leucine betaine, isoleucine betaine, dimethylsulfoniopropionate and mixtures thereof.
- the process according to the invention uses trimethylglycine (also referred to herein as glycine betaine or “betaine”), optionally in combination with at least one additional osmolyte, for example at least one additional compound of formula (I), preferably chosen from the compounds indicated in Figure 1.
- trimethylglycine also referred to herein as glycine betaine or “betaine”
- additional osmolyte for example at least one additional compound of formula (I), preferably chosen from the compounds indicated in Figure 1.
- the osmolyte(s) used as compound (b) in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
- the trimethylglycine used as compound (b) in one or more compositions that are useful in the process according to the invention preferably represents from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
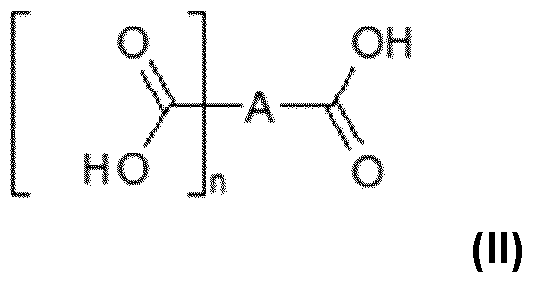
- the process according to the invention uses one or more hydroxylated polycarboxylic acids.
- the hydroxylated polycarboxylic acid(s) are chosen from the hydroxylated polycarboxylic acid(s) of formula (II) below: in which formula (II):
- - A is a multivalent, saturated or unsaturated, linear, branched, cyclic or even aromatic hydrocarbon-based group comprising from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, substituted with one or more hydroxyl (OH) groups.
- A is a multivalent (C1-C6)alkylene and better still (C2-C4)alkylene or phenylene group, substituted with one or more hydroxyl groups.
- hydroxylated polycarboxylic acids may be chosen from:
- the hydroxylated polycarboxylic acid, a salt thereof or mixtures thereof is chosen from citric acid, a salt thereof and mixtures thereof.
- the hydroxylated polycarboxylic acid(s), a salt thereof or mixtures thereof used as compound (c) in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
- citric acid, a salt thereof or mixtures thereof used as compound (c) in one or more compositions that are useful in the process according to the invention represents from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
- the citric acid used as compound (c) in one or more compositions that are useful in the process according to the invention represents from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
- the process may use one or more chemical oxidizing agents other than the peroxygenated salts.
- the process according to the invention uses one or more chemical oxidizing agents other than the peroxygenated salts.
- the chemical oxidizing agent(s) are chosen from hydrogen peroxide, hydrogen peroxide-generating systems other than peroxygenated salts, and mixtures thereof.
- the hydrogen peroxide-generating systems other than peroxygenated salts may be chosen from urea peroxide, polymeric complexes that can release hydrogen peroxide, oxidases, and mixtures thereof.
- Oxidases can produce hydrogen peroxide in the presence of a suitable substrate, for instance glucose in the case of glucose oxidase or uric acid with uricase.
- the process uses hydrogen peroxide as chemical oxidizing agent.
- composition(s) comprising hydrogen peroxide or a hydrogen peroxidegenerating system may also include various adjuvants conventionally used in compositions for dyeing keratin fibers as defined below.
- the chemical oxidizing agent(s) other than the peroxygenated salts preferably represent from 0.01% to 30% by weight, preferentially from 0.1% to 20% by weight, better still from 1% to 15% by weight relative to the total weight of the composition(s) containing them.
- hydrogen peroxide preferably represents from 0.01% to 30% by weight, preferentially from 0.1% to 20% by weight, better still from 1% to 15% by weight relative to the total weight of the composition(s) containing them.
- the process according to the invention may also use one or more of the following compounds.
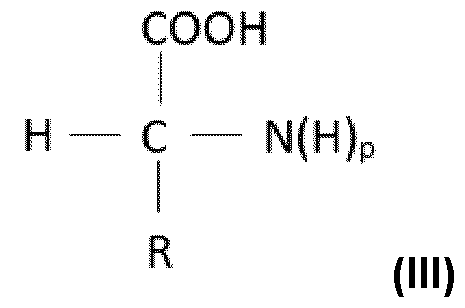
- the process according to the invention may optionally use one or more compounds of amino acid type.
- the term “compound of amino acid type” means an organic compound comprising one or more carboxylic acid and/or sulfonic acid functions and one or more amine functions, it being possible for the amine function(s) to be endocyclic, optionally in salt form.
- the compounds of amino acid type are different from the osmolytes described previously.
- the compound(s) of amino acid type are chosen from compounds of amino acid type comprising only one or more carboxylic acid functions (thus not comprising any sulfonic acid functions) and/or salts thereof.
- Said compounds are also called compounds of aminocarboxylic acid type and are particularly preferred.
- R forms, with the nitrogen atom, a saturated 5- to 8-membered heterocycle, preferably a 5-membered heterocycle, it being possible for this ring to be substituted with one or more groups chosen from hydroxyl or (C1-C4)alkyl;
- R represents a hydrogen atom or a saturated, linear or branched, (C1- C12)alkyl, preferably (C1-C4)alkyl, group, optionally interrupted with one or more heteroatoms or groups chosen from -S-, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH 2 ), -SH, -
- R forms, with the nitrogen atom, a saturated 5-membered heterocycle, this ring not being substituted.
- p 2.
- R represents a hydrogen atom or a saturated, linear or branched (Ci-C 4 )alkyl group, optionally interrupted with an -S- heteroatom and/or optionally substituted with one or two groups chosen from hydroxyl, amino or -NH-C(NH)-NH 2 .
- p 2 and R represents a hydrogen atom.
- the compounds of amino acid type may also be a salt of a compound of formula (III).
- These salts comprise the salts with organic or mineral bases, for example the salts of alkali metals, for instance the lithium, sodium or potassium salts; the salts of alkaline-earth metals, for instance the magnesium or calcium salts, and the zinc salts.
- organic or mineral bases for example the salts of alkali metals, for instance the lithium, sodium or potassium salts; the salts of alkaline-earth metals, for instance the magnesium or calcium salts, and the zinc salts.
- the compounds of amino acid type may be in the form of an optical isomer of L, D or DL configuration, preferably of L configuration.
- the compound(s) of amino acid type according to the invention are chosen from glycine, proline, methionine, serine, arginine, lysine, salts thereof (notably alkali metal, alkaline-earth metal or zinc salts) and mixtures thereof.
- the compound(s) of amino acid type according to the invention are chosen from glycine, proline, methionine, serine, arginine, salts thereof and mixtures thereof.
- the compound of amino acid type is chosen from glycine, arginine, salts thereof (notably alkali metal, alkaline-earth metal or zinc salts) and mixtures thereof.
- the compound of amino acid type is arginine.
- the compound(s) of amino acid type, preferably of aminocarboxylic acid type, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 20% by weight, preferably from 0.05% to 10% by weight, more preferably from 0.1% to 5% by weight, better still from 0.2% to 2% by weight, relative to the total weight of the composition(s) containing them.
- the compound(s) of amino acid type chosen from glycine, proline, methionine, serine, arginine, lysine, salts thereof and mixtures thereof, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 20% by weight, preferably from 0.05% to 10% by weight, more preferably from 0.1% to 5% by weight and better still from 0.2% to 2% by weight, relative to the total weight of the composition(s) containing them.
- the process according to the invention may also use one or more mineral, organic or hybrid alkaline agents.
- the alkaline agents according to the invention are different from the osmolytes and amino acids described previously.
- the process according to the invention uses one or more alkaline agents.
- alkaline agent and “basifying agent” are used interchangeably.
- the mineral basifying agent(s) are preferably chosen from aqueous ammonia, alkali metal carbonates or bicarbonates such as sodium (hydrogen) carbonate and potassium (hydrogen) carbonate, ammonium (hydrogen) carbonate, alkali metal or alkaline-earth metal phosphates such as sodium phosphates or potassium phosphates, sodium or potassium hydroxides, alkali metal or alkaline-earth metal silicates or metasilicates such as sodium silicate, sodium metasilicate, and mixtures thereof.
- alkali metal carbonates or bicarbonates such as sodium (hydrogen) carbonate and potassium (hydrogen) carbonate, ammonium (hydrogen) carbonate
- alkali metal or alkaline-earth metal phosphates such as sodium phosphates or potassium phosphates, sodium or potassium hydroxides
- alkali metal or alkaline-earth metal silicates or metasilicates such as sodium silicate, sodium metasilicate, and mixtures thereof.
- the organic basifying agent(s) are preferably chosen from alkanolamines, organic amines other than alkanolamines, oxyethylenated and/or oxypropylenated ethylenediamines, 1 ,3- diaminopropane, spermine, spermidine and mixtures thereof.
- alkanolamine means an organic amine comprising a primary, secondary or tertiary amine function, and one or more linear or branched C1-C8 alkyl groups bearing one or more hydroxyl radicals.
- Organic amines chosen from alkanolamines such as monoalkanolamines, dialkanolamines or trialkanolamines comprising one to three identical or different C1-C4 hydroxyalkyl radicals are in particular suitable for performing the invention.
- the alkanolamine(s) are chosen from monoethanolamine (MEA), diethanolamine, triethanolamine, monoisopropanolamine, diisopropanolamine, N,N- dimethylethanolamine, 2-amino-2-methyl-1-propanol, triisopropanolamine, 2-amino-2- methyl-1 ,3-propanediol, 3-amino-1 ,2-propanediol, 3-dimethylamino-1 ,2-propanediol, tris(hydroxymethyl)aminomethane and mixtures thereof.
- MEA monoethanolamine
- diethanolamine triethanolamine
- monoisopropanolamine diisopropanolamine
- N,N- dimethylethanolamine 2-amino-2-methyl-1-propanol
- 2-amino-2-methyl-1-propanol triisopropanolamine
- 2-amino-2- methyl-1 ,3-propanediol 3-amino
- the organic amine may also be chosen from organic amines of heterocyclic type. Besides histidine that has already been mentioned in the amino acids, mention may in particular be made of pyridine, piperidine, imidazole, triazole, tetrazole and benzimidazole.
- the organic amine may also be chosen from amino acid dipeptides. As amino acid dipeptides that may be used in the present invention, mention may notably be made of carnosine, anserine and balenine.
- the organic amine may also be chosen from compounds including a guanidine function.
- amines of this type other than arginine that may be used in the present invention, mention may notably be made of creatine, creatinine, 1 ,1- dimethylguanidine, 1 , 1 -diethylguanidine, glycocyamine, metformin, agmatine, n- amidoalanine, 3-guanidinopropionic acid, 4-guanidinobutyric acid and 2- ([amino(imino)methyl]amino)ethane-1 -sulfonic acid.
- Use may be made in particular of guanidine carbonate or monoethanolamine hydrochloride as hybrid compounds.
- alkaline agent(s) that are useful according to the invention are preferably chosen from alkanolamines such as monoethanolamine, diethanolamine or triethanolamine; aqueous ammonia, carbonates or bicarbonates such as sodium (hydrogen) carbonate and potassium (hydrogen) carbonate, ammonium (hydrogen) carbonate, alkali metal or alkaline-earth metal silicates or metasilicates such as sodium silicate, sodium metasilicate and mixtures thereof; more preferentially from alkali metal or alkaline-earth metal silicates or metasilicates, such as sodium silicate and metasilicate, and carbonates or bicarbonates, such as ammonium (hydrogen) carbonate, and mixtures thereof, better still from alkali metal or alkaline-earth metal silicates or metasilicates, such as sodium silicate and metasilicate.
- the alkaline agent(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.1% to 50% by weight, more preferentially from 1% to 40% by weight, better still from 5% to 35% by weight and even better still from 10% to 30% by weight, relative to the total weight of the composition(s) containing them.
- the alkaline agent(s) chosen from alkali metal or alkaline-earth metal silicates or metasilicates used in one or more compositions that are useful in the process according to the invention preferably represent from 0.1% to 50% by weight, more preferentially from 1% to 40% by weight, better still from 5% to 35% by weight and even better still from 10% to 30% by weight, relative to the total weight of the composition(s) containing them.
- Direct dyes The process according to the invention may optionally use one or more direct dyes.
- direct dyes means natural and/or synthetic dyes, other than oxidation dyes. These are dyes which will spread superficially over the fiber. They may be ionic, for example cationic or anionic, or nonionic.
- Suitable direct dyes include azo direct dyes; (poly)methine dyes such as cyanines, hemicyanines and styryls; carbonyl dyes; azine dyes; nitro(hetero)aryl dyes; tri(hetero)arylmethane dyes; porphyrin dyes; phthalocyanine- based dyes and natural direct dyes, alone or as mixtures.
- the direct dyes may be chosen from cationic direct dyes.
- cationic direct dyes include: Basic Red 51 , Basic Yellow 87 and Basic Orange 31 or derivatives thereof. Mention may also be made of anthraquinone cationic direct dyes, notably HC Blue 16 and HC Blue 17.
- the direct dyes may be chosen from anionic direct dyes.
- Anionic direct dyes are dyes commonly referred to as “acid” direct dyes owing to their affinity for alkaline substances.
- the term “anionic direct dye” means any direct dye comprising in its structure at least one CO 2 R or SO 3 R substituent with R denoting a hydrogen atom or a cation of a metal or of an amine, or an ammonium ion.
- the anionic dyes may be chosen from direct nitro acid dyes, azo acid dyes, azine acid dyes, triarylmethane acid dyes, indoamine acid dyes, anthraquinone acid dyes, indigoid dyes and natural acid dyes.
- diaryl anionic azo dyes examples include: a) diaryl anionic azo dyes;
- Acid Red 1 Acid Red 4, Acid Red 13, Acid Red 14, Acid Red 18, Acid Red 27, Acid Red 28, Acid Red 32, Acid Red 33, Acid Red 35, Acid Red 37, Acid Red 40, Acid Red 41 , Acid Red 42, Acid Red 44, Pigment Red 57, Acid Red 68, Acid Red 73, Acid Red 135, Acid Red 138, Acid Red 184, Food Red 1 , Food Red 13, Acid Orange 6, Acid Orange 7, Acid Orange 10, Acid Orange 19, Acid Orange 20, Acid Orange 24, Acid Yellow 6, Acid Yellow 9, Acid Yellow 36, Acid Yellow 199, Food Yellow 3; Acid Violet 7, Acid Violet 14, Acid Blue 113, Acid Blue 117, Acid Black 1 , Acid Brown 4, Acid Brown 20, Acid Black 26, Acid Black 52, Food Black 1 , Food Black 2; Food Yellow 3 or Sunset Yellow; or Acid Red 111 , Acid Red 134, Acid Yellow 38; b) pyrazolone anionic azo dyes, including: Acid Red 195, Acid Yellow 23, Acid Yellow 27, Acid Yellow 76 or Acid Yellow 17; c) anthraquinone dyes, including
- nitro dyes including: Acid Brown 13; Acid Orange 3; Acid Yellow 1 , the sodium salt of 2,4-dinitro-1-naphthol-7-sulfonic acid, 2-piperidino-5-nitrobenzenesulfonic acid, 2-(4’-N,N- (2”-hydroxyethyl)amino-2’-nitro)anilineethanesulfonic acid, 4-
- xanthene-based dyes including: Acid Yellow 73; Acid Red 51 ; Acid Red 52; Acid Red 87; Acid Red 92; Acid Red 95; Acid Violet 9;
- indole-based dyes including: Acid Blue 74;
- quinoline-based dyes including: Acid Yellow 2, Acid Yellow 3 and Acid Yellow 5.
- the direct dyes are chosen from anionic direct dyes.
- the direct dye(s) are chosen from blue or violet dyes such as Acid Violet 7, Acid Violet 14, Acid Blue 113, Acid Blue 117, Acid Blue 25, Acid Blue 43, Acid Blue 62, Acid Blue 78 Acid Blue 129, Acid Blue 138, Acid Blue 140, Acid Blue 251 , Acid Violet 42, Acid Violet 43 (also referred to as Violet EXT No. 2), Acid Blue 1 ; Acid Blue 3; Acid Blue 7; Acid Blue 9; Acid Violet 49, Acid Violet 9, Acid Blue 74, Basic Blue 6, Basic Blue 7, Basic Blue 9, Basic Blue 26, Basic Blue 41 , Basic Blue 77 (or HC Blue 15), Basic Blue 99, Basic Violet 1 , Basic Violet 2, Basic Violet 3, Basic Violet 10, Basic Violet 14, HC Blue 16, HC Blue 17.
- blue or violet dyes such as Acid Violet 7, Acid Violet 14, Acid Blue 113, Acid Blue 117, Acid Blue 25, Acid Blue 43, Acid Blue 62, Acid Blue 78 Acid Blue 129, Acid Blue 138, Acid Blue 140, Acid Blue 251 , Acid Violet 42, Acid Violet 43 (also referred to as Violet EXT No
- the preferred direct dyes are Violet EXT No. 2, Acid Blue 62, Acid Blue 9, Basic Violet 2, HC Blue 16, HC Blue17 and Tetrabromophenol Blue, more preferentially Violet EXT No.
- the direct dye(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.001% to 10% by weight, preferably from 0.005% to 5% by weight, more preferably from 0.001% to 3% by weight, relative to the total weight of the composition(s) containing them.
- the process according to the invention may also use one or more surfactants.
- surfactants may preferably be chosen from anionic surfactants, amphoteric surfactants, nonionic surfactants and cationic surfactants and/or mixtures thereof.
- the process according to the invention uses one or more surfactants.
- anionic surfactant means a surfactant including, as ionic or ionizable groups, only anionic groups. These anionic groups are preferably chosen from the following groups: H 2 PO 2 ,
- anionic surfactants that may be used in the process according to the invention, mention may be made of alkyl sulfates, alkyl ether sulfates, alkylamido ether sulfates, alkylaryl polyether sulfates, monoglyceride sulfates, alkyl sulfonates, alkylamide sulfonates, alkylaryl sulfonates, a-olefin sulfonates, paraffin sulfonates, alkyl sulfosuccinates, alkyl ether sulfosuccinates, alkylamide sulfosuccinates, alkyl sulfoacetates, acyl sarcosinates, acyl glutamates, alkyl sulfosuccinamates, acyl isethionates and N-(C1-C4)alkyl-N-acyl taurates, salts of
- anionic surfactants mention may also be made of salts of fatty acids, notably of C8-C24 and preferably C12-C20 fatty acids, other than the (poly)carboxylic acids described previously.
- These compounds may be oxyethylenated and then preferably include from 1 to 50 ethylene oxide units.
- the salts of C 6 -C 24 alkyl monoesters of polyglycoside-polycarboxylic acids may be chosen from C 6 -C 24 alkyl polyglycoside-citrates, C 6 -C 24 alkyl polyglycoside-tartrates and C 6 -C 24 alkyl polyglycoside-sulfosuccinates.
- anionic surfactant(s) When the anionic surfactant(s) are in salt form, they may be chosen from alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcohol salts or alkaline-earth metal salts such as the magnesium salt.
- alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcohol salts or alkaline-earth metal salts such as the magnesium salt.
- amino alcohol salts examples include monoethanolamine, diethanolamine and triethanolamine salts, monoisopropanolamine, diisopropanolamine or triisopropanolamine salts, 2-amino-2-methyl-1-propanol salts, 2- amino-2-methyl-1 ,3-propanediol salts and tris(hydroxymethyl)aminomethane salts.
- Alkali metal or alkaline-earth metal salts and in particular the sodium or magnesium salts are preferably used.
- the anionic surfactants that may be present may be mild anionic surfactants, i.e. anionic surfactants without a sulfate function.
- mild anionic surfactants mention may be made in particular of the following compounds and salts thereof, and also mixtures thereof: polyoxyalkylenated alkyl ether carboxylic acids, polyoxyalkylenated alkylaryl ether carboxylic acids, polyoxyalkylenated alkylamido ether carboxylic acids, in particular those including 2 to 50 ethylene oxide groups, alkyl D-galactoside uronic acids, acyl sarcosinates, acyl glutamates and alkylpolyglycoside carboxylic esters.
- Use may be made most particularly of polyoxyalkylenated alkyl ether carboxylic acids, for instance lauryl ether carboxylic acid (4.5 OE) sold, for example, under the name Akypo RLM 45 CA from Kao.
- lauryl ether carboxylic acid 4.5 OE
- anionic surfactants such as alkyl sulfates or alkyl ether sulfates, and acylglutamates, C12-C20 fatty acid salts, more preferentially alkyl sulfates and C12-C20 fatty acid salts.
- the process of the invention uses one or more surfactants chosen from alkyl sulfates.
- the surfactant(s), preferably anionic surfactants, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 15% by weight, more preferentially from 0.1% to 10% by weight, better still from 0.3% to 5% by weight, and even better still from 0.5% to 3% by weight of the composition(s) containing them.
- the process according to the invention may also use one or more fatty substances.
- the process according to the invention uses one or more fatty substances.
- the term “fatty substance” means an organic compound that is insoluble in water at ordinary room temperature (20-25°C) and at atmospheric pressure (760 mmHg, i.e. 1.013*10 5 Pa), with a solubility in water of less than 5%, preferably less than 1% and even more preferentially less than 0.1%.
- the fatty substances generally have in their structure a hydrocarbon-based chain including at least 6 carbon atoms.
- the fatty substances are generally soluble in organic solvents under the same temperature and pressure conditions, for instance chloroform, ethanol, benzene, liquid petroleum jelly or decamethylcyclopentasiloxane.
- the fatty substances are, moreover, non-(poly)oxyalkylenated and non-(poly)glycerolated.
- the fatty substances do not include in their structure a (poly)ethylene oxide or (poly)glycerol or (polypropylene glycol unit.
- the fatty substance(s) may be chosen from solid fatty substances and/or liquid fatty substances (also called “oil”), and mixtures thereof.
- oil means a “fatty substance” which is liquid, i.e. which is capable of flowing under the action of its own weight at room temperature (25°C) and at atmospheric pressure (760 mmHg, i.e. 1.013*10 5 Pa).
- the viscosity at a temperature of 25°C and at a shear rate of 1 s 1 of the oil is between 10 3 Pa.s and 2 Pa.s. It may be measured using a Thermo Haake RS600 rheometer with cone-plate geometry or an equivalent machine.
- solid fatty substance means a fatty substance that is not liquid at room temperature (20-25°C) and at atmospheric pressure (760 mmHg, i.e. 1.013x10 5 Pa), in particular a solid compound or a compound having a viscosity of greater than 2 Pa.s at a shear rate of 1 s 1 under the conditions mentioned above.
- the fatty substance(s) are chosen from the liquid fatty substances.
- liquid fatty substances are chosen from:
- - fatty alcohols that are liquid at room temperature, bearing a branched and/or unsaturated carbon-based chain containing from 12 to 26 carbon atoms, for instance cetanol, octyldodecanol, linoleyl alcohol, linolenyl alcohol, isostearyl alcohol, oleyl alcohol, 2- hexyldecanol, 2-butyloctanol or 2-undecylpentadecanol;
- the fatty substance(s) are chosen from hydrocarbons comprising more than 16 carbon atoms.
- the fatty substance(s), preferably liquid fatty substances, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 20% by weight, preferably from 0.1% to 15% by weight, and preferentially from 0.2% to 5% by weight of the composition(s) containing them.
- the process according to the present invention may also use one or more associative polymers.
- the process according to the invention uses one or more associative polymers.
- sociative polymers are polymers that are capable, in an aqueous medium, of reversibly associating with each other or with other molecules.
- Their chemical structure more particularly comprises at least one hydrophilic zone or group and at least one hydrophobic zone or group.
- hydrophobic zone or group means a radical or polymer with a saturated or unsaturated, linear or branched hydrocarbon-based chain, comprising at least 10 carbon atoms, preferably from 10 to 30 carbon atoms, in particular from 12 to 30 carbon atoms and more preferentially from 18 to 30 carbon atoms.
- the hydrocarbon-based group originates from a monofunctional compound.
- the hydrophobic group may be derived from a fatty alcohol such as stearyl alcohol, dodecyl alcohol or decyl alcohol. It may also denote a hydrocarbon-based polymer, for instance polybutadiene.
- the associative polymer may be anionic, cationic, amphoteric or nonionic.
- anionic associative polymers those that are particularly preferred according to the invention are polymers formed from 20% to 60% by weight of acrylic acid and/or of methacrylic acid, from 5% to 60% by weight of lower alkyl (meth)acrylates, from 2% to 50% by weight of fatty-chain allyl ether, and from 0 to 1% by weight of a crosslinking agent which is a well-known copolymerizable unsaturated polyethylenic monomer, for instance diallyl phthalate, allyl (meth)acrylate, divinylbenzene, (poly)ethylene glycol dimethacrylate or methylenebisacrylamide.
- a crosslinking agent which is a well-known copolymerizable unsaturated polyethylenic monomer, for instance diallyl phthalate, allyl (meth)acrylate, divinylbenzene, (poly)ethylene glycol dimethacrylate or methylenebisacrylamide.
- crosslinked terpolymers of methacrylic acid, of ethyl acrylate and of polyethylene glycol (10 EO) stearyl alcohol ether (Steareth-10), notably those sold by the company Ciba under the names Salcare SC 80® and Salcare SC 90®, which are aqueous 30% emulsions of a crosslinked terpolymer of methacrylic acid, of ethyl acrylate and of steareth-10 allyl ether (40/50/10).
- (C10-C30)alkyl esters of unsaturated carboxylic acids that are useful in the invention comprise, for example, lauryl acrylate, stearyl acrylate, decyl acrylate, isodecyl acrylate and dodecyl acrylate, and the corresponding methacrylates, lauryl methacrylate, stearyl methacrylate, decyl methacrylate, isodecyl methacrylate and dodecyl methacrylate.
- maleic anhydride/C30-C38 a-olefin/alkyl maleate terpolymers such as the product (maleic anhydride/C30-C38 a-olefin/isopropyl maleate copolymer) sold under the name Performa 1608® by the company Newphase Technologies.
- APMS 2-acrylamido-2-methylpropanesulfonic acid
- the polymers of this family may be chosen notably from random amphiphilic AMPS polymers modified by reaction with a C6-C22 n-monoalkylamine or di-n-alkylamine, and such as those described in patent application WO 00/31154 (which form an integral part of the content of the description). These polymers may also contain other ethylenically unsaturated hydrophilic monomers chosen, for example, from (meth)acrylic acids,
- the preferred polymers of this family are chosen from amphiphilic copolymers of AMPS and of at least one ethylenically unsaturated hydrophobic monomer.
- copolymers may also contain one or more ethylenically unsaturated monomers not including a fatty chain, such as (meth)acrylic acids, ⁇ -substituted alkyl derivatives thereof or esters thereof obtained with monoalcohols or mono- or polyalkylene glycols, (meth)acrylamides, vinylpyrrolidone, maleic anhydride, itaconic acid or maleic acid, or mixtures of these compounds.
- ethylenically unsaturated monomers not including a fatty chain such as (meth)acrylic acids, ⁇ -substituted alkyl derivatives thereof or esters thereof obtained with monoalcohols or mono- or polyalkylene glycols, (meth)acrylamides, vinylpyrrolidone, maleic anhydride, itaconic acid or maleic acid, or mixtures of these compounds.
- - terpolymers including from 10 mol% to 90 mol% of acrylamide units, from 0.1 mol% to 10 mol% of AMPS units and from 5 mol% to 80 mol% of n-(C6-C18)alkylacrylamide units, such as those described in patent US-5 089 578.
- copolymers of totally neutralized AMPS and of dodecyl methacrylate and also crosslinked and noncrosslinked copolymers of AMPS and of n- dodecylmethacrylamide, such as those described in the Morishima articles mentioned above.
- anionic associative polymers preference is given to polymers including i) at least one hydrophilic unit of the olefinic unsaturated carboxylic acid type, and ii) at least one hydrophobic unit of the type such as a (C10-C30) alkyl ester of an unsaturated carboxylic acid (family b)), and copolymers including among their monomers an a,
- cationic associative polymers mention may be made of: (a) cationic associative polyurethanes; (b) the compound sold by the company Noveon under the name Aqua CC and which corresponds to the INCI name Polyacrylate- 1 Crosspolymer.
- Polyacrylate- 1 Crosspolymer is the product of the polymerization of a mixture of monomers comprising: a di(C1-C4 alkyl)amino(C1-C6 alkyl) methacrylate, one or more C1-C30 alkyl esters of (meth)acrylic acid, a polyethoxylated C10-C30 alkyl methacrylate (20-25 mol of ethylene oxide units), a 30/5 polyethylene glycol/polypropylene glycol allyl ether, a hydroxy(C2-C6 alkyl) methacrylate, and an ethylene glycol dimethacrylate.
- the alkyl radicals borne by the above quaternized celluloses or hydroxyethylcelluloses preferably include from 8 to 30 carbon atoms.
- the aryl radicals preferably denote phenyl, benzyl, naphthyl or anthryl groups.
- Examples of quaternized alkylhydroxyethylcelluloses containing C8-C30 fatty chains that may be indicated include the products Quatrisoft LM 200®, Quatrisoft LM-X 529-18-A®, Quatrisoft LM-X 529-18-B® (C12 alkyl) and Quatrisoft LM-X 529-8® (C18 alkyl) sold by the company Aquaion, and the products Crodacel QM®, Crodacel QL® (C12 alkyl) and Crodacel QS® (C18 alkyl) sold by the company Croda and the product Softcat SL 100® sold by the company Aquaion.
- Such polymers are described, for example, in patent application WO-OO/68282.
- cationic poly(vinyllactam) polymers use is notably made of vinylpyrrolidone/dimethylaminopropylmethacrylamide/dodecyl- dimethylmethacrylamidopropylammonium tosylate terpolymers, vinylpyrrolidone/dimethylaminopropylmethacrylamide/cocoyldimethylmethacryl- amidopropylammonium tosylate terpolymers, vinylpyrrolidone/dimethylamino- propylmethacrylamide/lauryldimethylmethacrylamidopropylammonium tosylate or chloride terpolymers.
- amphoteric associative polymers are preferably chosen from those including at least one noncyclic cationic unit. Even more particularly, preference is given to those prepared from or comprising 1 to 20 mol%, preferably 1 .5 to 15 mol% and even more particularly 1.5 to 6 mol% of fatty-chain monomer relative to the total number of moles of monomers.
- Amphoteric associative polymers that may be used in the invention are described and prepared, for example, in patent application WO 98/44012.
- the associative polymers of nonionic type that may be used according to the invention are preferably chosen from:
- copolymers of C1-C6 alkyl methacrylates or acrylates and of amphiphilic monomers including at least one fatty chain for instance the oxyethylenated methyl acrylate/stearyl acrylate copolymer sold by the company Goldschmidt under the name Antil 208, or the copolymer having the INCI name Acrylates/Beheneth-25 Methacrylate Copolymer, such as the product Novethix L-10 Polymer from Lubrizol.
- copolymers of hydrophilic methacrylates or acrylates and of hydrophobic monomers including at least one fatty chain for instance the polyethylene glycol methacrylate/lauryl methacrylate copolymer.
- celluloses or derivatives thereof modified with groups including at least one fatty chain, such as alkyl, arylalkyl or alkylaryl groups or mixtures thereof in which the alkyl groups are of C8, and in particular:
- fatty-chain nonionic polyurethane polyethers are those in which the hydrophilic blocks are linked to the lipophilic blocks via other chemical bonds.
- nonionic fatty-chain polyurethane polyethers that may be used in the invention, use may also be made of Rheolate 205® bearing a urea function, sold by the company Rheox, or Rheolate® 208, 204 or 212, and also Acrysol RM 184®.
- Use may also be made of solutions or dispersions of these polymers, notably in water or in an aqueous/alcoholic medium.
- examples of such polymers that may be mentioned include Rheolate® 255, Rheolate® 278 and Rheolate® 244 sold by the company Rheox.
- Use may also be made of the products DW 1206F and DW 1206J sold by the company Rohm & Haas.
- a polyurethane polyether that may be obtained by polycondensation of at least three compounds comprising (i) at least one polyethylene glycol comprising from 150 to 180 mol of ethylene oxide, (ii) stearyl alcohol or decyl alcohol, and (iii) at least one diisocyanate.
- Aculyn 46® is a polycondensate of polyethylene glycol containing 150 or 180 mol of ethylene oxide, of stearyl alcohol and of methylenebis(4-cyclohexyl isocyanate) (SMDI), at 15% by weight in a matrix of maltodextrin (4%) and water (81%);
- Aculyn 44® is a polycondensate of polyethylene glycol containing 150 or 180 mol of ethylene oxide, of decyl alcohol and of methylenebis(4-cyclohexyl isocyanate) (SMDI), at 35% by weight in a mixture of propylene glycol (39%) and water (26%)].
- the associative polymer(s) are chosen from anionic associative polymers.
- the associative polymer(s) are chosen from acrylic or methacrylic acid homopolymers or copolymers.
- the associative polymer(s) are chosen from polymers including i) at least one hydrophilic unit of the olefinic unsaturated carboxylic acid type, and ii) at least one hydrophobic unit of the type such as a (C10-C30) alkyl ester of unsaturated carboxylic acid, the copolymers including among their monomers an a,
- the associative polymer(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 10% by weight, better still from 0.1% to 8% by weight, even better still from 0.2% to 5% by weight of the composition(s) containing them.
- the process according to the present invention may also use one or more non-associative polysaccharides, which are thus different from the above associative polymers.
- the process according to the invention uses one or more non-associative polysaccharides.
- polysaccharide means a polymer constituted of sugar units.
- sugar unit means an oxygen-bearing hydrocarbon-based compound containing several alcohol functions, with or without aldehyde or ketone functions, and which includes at least 4 carbon atoms.
- the sugar units may be optionally modified by substitution, and/or by oxidation and/or by dehydration.
- sugar units that may be included in the composition of the polysaccharides of the invention are preferably derived from the following sugars: glucose, galactose, arabinose, rhamnose, mannose, xylose, fucose, anhydrogalactose, galacturonic acid, glucuronic acid, mannuronic acid, galactose sulfate, anhydrogalactose sulfate and fructose.
- Non-associative polysaccharides that may notably be mentioned include the following polymers, alone or as a mixture: a) tree or shrub exudates, including:
- acacia gum branched polymer of galactose, arabinose, rhamnose and glucuronic acid
- - ghatti gum polymer derived from arabinose, galactose, mannose, xylose and glucuronic acid
- karaya gum polymer derived from galacturonic acid, galactose, rhamnose and glucuronic acid
- gum tragacanth polymer of galacturonic acid, galactose, fucose, xylose and arabinose
- gums derived from algae including:
- microbial gums including:
- - gellan gum polymer of partially acylated glucose, rhamnose and glucuronic acid
- polymers may be physically or chemically modified.
- physical treatment mention may notably be made of a heat treatment.
- Chemical treatments that may be mentioned include esterification, etherification, amidation or oxidation reactions. These treatments make it possible to produce polymers that may notably be nonionic, anionic or amphoteric.
- guar gums, locust bean gums, starches and celluloses may be modified/treated.
- the guar gums that may be used according to the invention may be modified with C1-C6 (poly)hydroxyalkyl groups.
- C1-C6 (poly)hydroxyalkyl groups examples that may be mentioned include hydroxymethyl, hydroxyethyl, hydroxypropyl and hydroxybutyl groups.
- These guar gums are well known in the prior art and may be prepared, for example, by reacting corresponding alkene oxides, for instance propylene oxides, with the guar gum so as to obtain a guar gum modified with hydroxypropyl groups.
- the degree of hydroxyalkylation preferably ranges from 0.4 to 1 .2 and corresponds to the number of alkylene oxide molecules consumed by the number of free hydroxyl functions present on the guar gum.
- Such guar gums optionally modified with hydroxyalkyl groups are sold, for example, under the trade names Jaguar HP8, Jaguar HP60 and Jaguar HP120 by the company Rhodia Chimie.
- the botanical origin of the starches that may be used in the present invention may be cereals or tubers.
- the starches are chosen, for example, from corn starch, rice oat, starch, cassava starch, barley starch, potato starch, wheat starch, sorghum starch, pea starch and tapioca starch. It is also possible to use hydrolyzates of the starches mentioned above.
- the starch is preferably derived from potato.
- Starch phosphates notably distarch phosphates or compounds rich in distarch phosphate, will preferentially be used, for instance the product sold under the references Prejel VA- 70-T AGGL (gelatinized hydroxypropyl cassava distarch phosphate), Prejel TK1 (gelatinized cassava distarch phosphate) or Prejel 200 (gelatinized acetylated cassava distarch phosphate) by the company Avebe, or Structure Zea from National Starch (gelatinized corn distarch phosphate).
- amphoteric starches may also be used, these amphoteric starches comprising one or more anionic groups and one or more cationic groups.
- the anionic and cationic groups may be bonded to the same reactive site of the starch molecule or to different reactive sites; they are preferably bonded to the same reactive site.
- the anionic groups may be of carboxylic, phosphate or sulfate type, preferably carboxylic type.
- the cationic groups may be of primary, secondary, tertiary or quaternary amine type.
- the polysaccharides that may be used according to the invention may be cellulose-based polymers.
- cellulose-based polymer refers to any polysaccharide compound bearing in its structure sequences of glucose residues linked together via (3-1 ,4 bonds; in addition to unsubstituted celluloses, the cellulose derivatives may be anionic, cationic, amphoteric or nonionic.
- Cellulose-based polymers are also known as celluloses.
- the cellulose-based polymers that may be used according to the invention may be chosen from unsubstituted celluloses, including those in a microcrystalline form, and cellulose ethers.
- cellulose ethers cellulose esters
- cellulose ethers-esters are distinguished.
- cellulose esters are mineral esters of cellulose (cellulose nitrates, sulfates, phosphates, etc.), organic cellulose esters (cellulose monoacetates, triacetates, amidopropionates, acetatebutyrates, acetatepropionates or acetatetrimellitates, etc.), and mixed organic/mineral esters of cellulose, such as cellulose acetatebutyrate sulfates and cellulose acetatepropionate sulfates.
- cellulose ether esters mention may be made of hydroxypropylmethylcellulose phthalates and ethylcellulose sulfates.
- cellulose ethers that may be mentioned are (Ci-C 4 )alkylcelluloses, such as methylcelluloses and ethylcelluloses (for example Ethocel Standard 100 Premium from Dow Chemical); (poly)hydroxy(Ci-C 4 )alkylcelluloses, such as hydroxymethylcelluloses, hydroxyethylcelluloses (for example Natrosol 250 HHR sold by Ashland) and hydroxypropylcelluloses (for example Klucel EF from Aquaion); mixed (poly)hydroxy(Ci- C 4 )alkyl(Ci-C 4 )alkylcelluloses, such as hydroxypropylmethylcelluloses (for example Methocel E4M from Dow Chemical), hydroxyethylmethylcelluloses, hydroxyethylethylcelluloses (for example Bermocoll E 481 FQ from Akzo Nobel) and hydroxybutylmethylcelluloses.
- methylcelluloses and ethylcelluloses for example Ethocel Standard 100 Premium from Dow Chemical
- anionic cellulose ethers mention may be made of (poly)carboxy(CI- C4)alkylcelluloses and salts thereof. Examples that may be mentioned include carboxymethylcelluloses, carboxymethylmethylcelluloses (for example Blanose 7M from the company Aquaion) and carboxymethylhydroxyethylcelluloses, and the sodium salts thereof.
- cationic cellulose derivatives such as cellulose copolymers or cellulose derivatives grafted with a water- soluble quaternary ammonium monomer, and notably described in patent US 4 131 576, such as (poly)hydroxy(C1-C4)alkylcelluloses, for instance hydroxymethyl-, hydroxyethyl- or hydroxypropylcelluloses notably grafted with a methacryloylethyltrimethylammonium, methacrylamidopropyltrimethylammonium or dimethyldiallylammonium salt.
- the commercial products corresponding to this definition are more particularly the products sold under the names Celquat® L 200 and Celquat® H 100 by the company National Starch.
- the non-associative polysaccharide(s) are chosen, alone or as mixtures, from celluloses, guar gums and starches, preferentially from celluloses.
- non-associative polysaccharides are chosen, alone or as a mixture, from cellulose ethers, cellulose esters, cellulose ether esters and guar gums, and preferably from cellulose ethers.
- the non-associative polysaccharide(s) are chosen from (C1-C4)alkylcelluloses such as methylcelluloses and ethylcelluloses; (poly)hydroxy(C1- C4)alkylcelluloses such as hydroxymethylcelluloses, hydroxyethylcelluloses and hydroxypropylcelluloses; mixed (poly)hydroxy(C1-C4)alkyl-(C1-C4)alkylcelluloses such as hydroxypropyl-methylcelluloses, hydroxyethyl-methylcelluloses, hydroxyethylethylcelluloses and hydroxybutyl-methylcelluloses.
- the process according to the invention uses one or more non-associative polysaccharides, preferentially chosen from celluloses, guar gums, starches, and mixtures thereof.
- the non-associative polysaccharide(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 10% by weight, better still from 0.1% to 8% by weight and even better still from 0.2% to 5% by weight of the composition(s) containing them.
- composition(s) of the lightening process according to the invention may also comprise one or more additives, among which mention may be made of mineral thickeners, antidandruff agents, anti-seborrheic agents, agents for preventing hair loss and/or for promoting hair regrowth, vitamins and provitamins including panthenol, sunscreens, mineral or organic pigments, plasticizers, solubilizers, opacifiers or nacreous agents, antioxidants, sequestrants, fragrances, and preserving agents.
- additives among which mention may be made of mineral thickeners, antidandruff agents, anti-seborrheic agents, agents for preventing hair loss and/or for promoting hair regrowth, vitamins and provitamins including panthenol, sunscreens, mineral or organic pigments, plasticizers, solubilizers, opacifiers or nacreous agents, antioxidants, sequestrants, fragrances, and preserving agents.
- the above additives may generally be present in an amount, for each of them, of between 0 and 20% by weight relative to the total weight of the composition(s) containing them.
- keratin fibers may be treated with the compounds described previously in one or more application steps.
- keratin fibers may be treated with one or more compositions which contain, together or separately, the compounds described previously.
- compounds (b) and (c), i.e. the osmolyte(s) (b) and the hydroxylated polycarboxylic acid(s), a salt thereof or mixtures thereof (c) are applied together to the keratin fibers.
- compounds (b) and (c) are contained in the same composition.
- the keratin fibers are treated in one application step, all the compounds (a), (b) and (c) being applied together to the fibers.
- the process comprises a step of applying to the keratin fibers a composition (C) comprising compounds (a), (b), (c), and optionally (d) as defined previously.
- the process according to the invention may comprise, prior to the step of applying composition (C), a step of mixing a composition (C1) comprising compounds (a), (b) and (c), and an oxidizing composition (O) comprising the compound(s) (d).
- This mixing step is preferably performed at the time of use, just before application to the hair of composition (C) resulting from the mixing.
- compositions (C1) and (O) are mixed in a (C1)/(O) weight ratio ranging from 0.1 to 2, preferentially from 0.3 to 1 .5, better still from 0.5 to 1 .
- the process according to the invention may comprise, prior to the step of applying composition (C), a step of mixing compounds (b) and (c) with a composition (C2) comprising the compound(s) (a), and optionally with an oxidizing composition (O) comprising the compound(s) (d).
- composition (C) may be applied to wet or dry keratin fibers.
- the temperature during the lightening process conventionally ranges from room temperature (between 15°C and 25°C) to 80°C and preferably from room temperature to 60°C.
- the keratin fibers are optionally rinsed with water, optionally undergo washing with a shampoo followed by rinsing with water, and are then dried or left to dry.
- the keratin fibers are treated in several application steps, the process using the compounds (a), (b), (c) and optionally (d) described previously from at least two compositions applied separately to the keratin fibers.
- the process of the invention may involve:
- composition (C3) comprising the compounds (a), and optionally (d), and
- composition (C4) comprising compounds (b) and (c).
- step (ii) may be performed before or after step (i).
- step (ii) is performed after step (i).
- Step (ii) of applying composition (C4) may be repeated several times, with or without rinsing between the applications.
- the process involves, prior to step (i), a step of mixing a composition (C2) comprising the compound(s) (a) with an oxidizing composition (O) comprising the compound(s) (d).
- compositions (C3) and (C4) may be applied to wet or dry keratin fibers.
- Composition (C3) is left to stand on the fibers for a period generally from 1 minute to 1 hour, preferably from 5 minutes to 55 minutes.
- Composition (C4) is left to stand on the fibers for a period generally from 1 minute to 30 minutes, better still from 5 minutes to 15 minutes.
- Composition (C4) may be applied before or after composition (C3).
- the process comprises a step of rinsing the keratin fibers between steps (i) and (ii).
- the temperature during the lightening process conventionally ranges from room temperature (between 15°C and 25°C) to 80°C and preferably from room temperature to 60°C.
- the keratin fibers are optionally rinsed with water, optionally undergo washing with a shampoo followed by rinsing with water, and are then dried or left to dry.
- composition (C) comprising the compounds (a), (b), (c) and optionally (d) as defined previously.
- the process may use the compounds (a), (b), (c) and optionally (d) described previously in a single step of applying a composition (C) to keratin fibers.
- composition (C) comprises one or more additional oxidizing agents (d), different from the peroxygenated salts, composition (C) then being a ready-to-use composition.
- composition (C) is aqueous.
- the pH of composition (C), when it is aqueous ranges from 8 to 13, preferentially from 9 to 12.
- composition (C) is derived from the mixing of a composition (C1) comprising (a), (b) and (c) and an oxidizing composition (O) which comprises a chemical oxidizing agent (d) different from the peroxygenated salts (a).
- a composition (C1) comprising (a), (b) and (c) and an oxidizing composition (O) which comprises a chemical oxidizing agent (d) different from the peroxygenated salts (a).
- composition (C1) comprises one or more alkaline agents as described previously.
- composition (C1) comprises one or more surfactants as described previously.
- composition (C1) comprises one or more fatty substances as described previously.
- composition (C1) comprises one or more associative polymers as described previously.
- Composition (C1) may optionally comprise one or more compounds of amino acid type described previously.
- Composition (C1) may optionally comprise one or more direct dyes described previously.
- composition (C1) is preferably anhydrous.
- anhydrous composition refers to a composition which comprises little or no water, notably less than 0.5% water, better still less than 0.1% water, even better still less than 0.05% water, relative to the total weight of the composition.
- composition (C1) does not comprise any water added during its preparation, the water that may be present possibly being provided by the starting materials used during its preparation.
- composition (C1) is a pulverulent composition.
- composition (C1) may be derived from the mixing of compounds (b) and (c) with a composition (C2) comprising the other compounds of composition (C1) described above.
- composition (C2) is preferably anhydrous, and is preferably a pulverulent composition.
- the oxidizing composition (O) which comprises a chemical oxidizing agent (d) different from the peroxygenated salts (a) is preferably an aqueous composition.
- it comprises more than 10% by weight of water, preferably more than 30% by weight of water and even more advantageously more than 50% by weight of water relative to the total weight of the oxidizing composition (O).
- the oxidizing composition (O) comprises water in a content ranging from 10% to 95% by weight, preferably from 30% to 90% by weight, better still from 50% to 80% by weight, relative to the total weight of the oxidizing composition (O).
- the pH of the oxidizing composition (O), when it is aqueous, is preferably less than 7, preferably between 1 and 5, preferentially between 1 .5 and 4.5.
- This pH may be adjusted to the desired value by using one or more acidifying agents, which may notably be chosen from those described previously.
- the process may use the compounds (a), (b), (c) and optionally (d) described previously from two compositions (C3) and (C4) applied separately to keratin fibers.
- composition (C3) comprises one or more compounds (a) and optionally one or more additional oxidizing agents (d), different from peroxygenated salts.
- composition (C3) comprises one or more additional oxidizing agents (d), different from the peroxygenated salts, composition (C3) then being a ready-to-use composition.
- composition (C3) is aqueous.
- the pH of composition (C3), when it is aqueous ranges from 8 to 13, preferentially from 9 to 12.
- composition (C3) is derived from the mixing of a composition (C2) comprising compound(s) (a) and an oxidizing composition (O) as defined previously.
- composition (C2) comprises one or more alkaline agents as described previously.
- composition (C2) comprises one or more surfactants as described previously.
- composition (C2) comprises one or more fatty substances as described previously.
- composition (C2) comprises one or more associative polymers as described previously.
- Composition (C2) may optionally comprise one or more compounds of amino acid type described previously.
- Composition (C2) may optionally comprise one or more direct dyes described previously.
- composition (C2) is anhydrous within the meaning defined previously.
- Composition (C4) comprises compounds (b) and (c) as described previously.
- composition (C4) comprises one or more surfactants as described previously.
- composition (C4) comprises one or more fatty substances as described previously.
- composition (C4) comprises one or more associative polymers as described previously, and notably one or more polysaccharides, preferably one or more cationic polysaccharides.
- Composition (C4) may optionally comprise one or more compounds of amino acid type described previously.
- Composition (C4) may optionally comprise one or more direct dyes described previously.
- composition (C4) is aqueous.
- a subject of the invention is also the use of a composition comprising compounds (a), (b), (c), and optionally (d) as defined previously, for lightening keratin fibers, and in particular the hair.
- Another subject of the invention is a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition (C1) comprising compounds (a), (b) and (c) as described previously, and at least a second compartment containing an oxidizing composition (O) comprising one or more compounds (d) as described previously.
- a composition (C1) comprising compounds (a), (b) and (c) as described previously
- O oxidizing composition
- Another subject of the invention is a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition (C2) comprising the compound(s) (a) as described previously, at least a second compartment containing a composition (C4) comprising compounds (b) and (c) as described previously, and optionally at least a third compartment containing an oxidizing agent composition (O) comprising one or more compounds (d) as described previously.
- the compositions of the device according to the invention are packaged in separate compartments, optionally accompanied by suitable application means, which may be identical or different, such as fine brushes, coarse brushes or sponges.
- the device mentioned above may also be equipped with a means for dispensing the desired mixture onto the hair, for instance the devices described in patent FR 2 586 913.
- compositions A, A1 and A2 were prepared from the compounds whose contents are indicated in the table below:
- composition A Table 1] The following were then added to composition A :
- composition A a composition A - 0.1 g of trimethylglycine (glycine betaine) and 0.1 g of citric acid to 4 g of composition A to obtain a composition A’, and
- compositions A’ and A” were respectively mixed with an oxidizing agent O comprising 9% hydrogen peroxide in the weight ratio 1+1.5.
- Each of the mixtures A’+O and A”+O was then applied to a lock of natural chestnut-brown hair (tone height 4) at a rate of 10 g of mixture per 1 g of lock of hair.
- the lightening of the hair was evaluated in the L*a*b* system, using a Konica Minolta CM- 3600A spectrocolorimeter (illuminant D65, angle 10°, specular component included) in the CIELab system.
- the tensile strength the solidity of the hair which can be measured by the tensile test. This measurement for determining the tensile mechanical properties of the hair is performed using a commercial tool, the MTT600 (mini Tensile Tester) from the company Dia Stron. The modulus of elasticity and the breaking strength of the hair are measured: the higher the values, the stronger the hair;
- cysteic acid and of free proteins at the surface of the hair these compounds are released during oxidation of the hair; the higher the content of cysteic acid and free proteins, the more damaged the hair.
- the cysteic acid present in the hair is assayed by means of a Hitachi L8900 amino acid autoanalyzer; 20 mg of finely cut hair are transferred into a culture tube (Duran). 2.5 ml of 9 N hydrochloric acid are added to the tube with magnetic stirring under nitrogen for 1 min at 2 bar.
- the solution is heated in the tube in an oil bath at 110°C for 16 hours.
- the pH is adjusted to between 1 .6 and 1 .8 and diluted to 50 ml with ultrapure water.
- the solution is then filtered and analyzed in the autoanalyzer.
- the quantitative analysis of the labile proteins is performed with the same equipment as for the cysteic acid assay.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Emergency Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Inorganic Chemistry (AREA)
- Cosmetics (AREA)
Abstract
The invention relates to a process for lightening keratin fibers, preferably human keratin fibers, notably the hair, in which said fibers are treated, in one or more steps, with one or more compositions containing, together or separately: (a) one or more peroxygenated salts; (b) one or more osmolytes; (c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and (d) optionally, one or more additional oxidizing agents, different from the peroxygenated salts.
Description
DESCRIPTION
TITLE: Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid
The present invention relates to a process for lightening keratin fibers, in particular human keratin fibers such as the hair, using one or more peroxygenated salts, one or more osmolytes and one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof.
In the field of hair lightening, the tone height is generally used to characterize the degree or level of lightening. The notion of “tone” is based on the classification of natural shades, one tone separating each shade from the shade immediately following or preceding it. This definition and the classification of natural shades are well known to hairstyling professionals and are published in the book Sciences des traitements capillaires [The Science of Hair Care] by Charles Zviak, 1988, published by Masson, pages 215 and 278.
The tone heights range from 1 (black) to 10 (very light blond), one unit corresponding to one tone; the higher the figure, the lighter the shade.
Lightening thus allows the head of hair to be given a lighter tone height than its original natural tone.
The processes for lightening the hair generally consist in using an aqueous composition comprising at least one oxidizing agent, under alkaline pH conditions in the vast majority of cases.
This oxidizing agent has the role of degrading the melanin of the hair, which, depending on the nature of the oxidizing agent present, leads to more or less pronounced lightening of the fibers. Thus, for relatively mild lightening, the oxidizing agent is generally hydrogen peroxide. When greater lightening is sought, notably lightening of at least five tones, use is usually made of peroxygenated salts, for instance persulfates, in the presence of hydrogen peroxide. These peroxygenated salts are contained in compositions which, at the time of use, are mixed with an aqueous composition comprising hydrogen peroxide.
In order to adjust the pH of the compositions to an alkaline pH to allow activation of the oxidizing agent, an alkaline agent is used. This alkaline agent also causes swelling of the keratin fibers, with opening of the scales, which favors penetration of the oxidizing agent into the fiber, and thus increases the efficacy of the reaction.
However, the use of alkaline agents and peroxygenated salts may impair the quality of the hair. The essential causes of this deterioration in hair quality are a reduction in its cosmetic properties, such as sheen, and a deterioration in its mechanical properties, more particularly a deterioration in its mechanical strength, which may also be reflected by an increase in its porosity. The hair is weakened and may become brittle during subsequent treatments such as blow-drying.
The lightening of dark hair is thus particularly delicate, as it requires the use of a large amount of peroxygenated salts if it is desired to significantly lighten the hair, which can weaken or even damage it, notably leading to hair manageability problems.
Thus, one of the objects of the present invention is to propose a process for lightening keratin fibers, preferably human keratin fibers such as the hair, which does not have the
drawbacks mentioned above, i.e. which is capable of reducing hair damage while at the same time leading to very good lightening performance.
This and other aims are achieved by the present invention, one subject of which is thus a process for lightening keratin fibers, preferably human keratin fibers, notably the hair, in which said fibers are treated, in one or more steps, with one or more compositions containing, together or separately:
(a) one or more peroxygenated salts;
(b) one or more osmolytes;
(c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and
(d) optionally, one or more additional oxidizing agents, different from the peroxygenated salts.
The invention also relates to a composition comprising:
(a) one or more peroxygenated salts;
(b) one or more osmolytes;
(c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and
(d) optionally, one or more additional oxidizing agents, different from the peroxygenated salts, and also to the use of this composition for lightening keratin fibers, and in particular the hair.
The invention also relates to a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition comprising (a) one or more peroxygenated salts, (b) one or more osmolytes and (c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and at least a second compartment containing an oxidizing composition comprising one or more additional oxidizing agents, different from the peroxygenated salts.
The invention also relates to a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition comprising (a) one or more peroxygenated salts, a second compartment comprising (b) one or more osmolytes and (c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and optionally at least a third compartment containing an oxidizing composition comprising one or more additional oxidizing agents, different from the peroxygenated salts.
The process according to the invention allows hair damage to be reduced without impacting on the lightening performance. It notably allows a high level of lightening to be obtained, of up to nine tones, while at the same time reducing hair manageability problems.
Other subjects, features, aspects and advantages of the invention will emerge even more clearly on reading the following description and examples and on examining the appended drawing, on which:
[Fig .1 ] depicts the structure of certain examples of osmolytes that may be used in the process according to the invention.
In the text hereinbelow, unless otherwise indicated, the limits of a range of values are included in that range, notably in the expressions “between” and “ranging from ... to ...”.
Moreover, the expression “at least one” used in the present description is equivalent to the expression “one or more”.
The terms “lightening” and “bleaching” are synonymous and may be used interchangeably.
1. Compounds
The process for lightening keratin fibers according to the invention uses compounds (a), (b), (c) and optionally (d) described hereinbelow.
(a) Peroxygenated salts
The process according to the invention uses one or more peroxygenated salts.
Preferably, the peroxygenated salts are chosen from persulfates; perborates; alkali metal, alkaline-earth metal or ammonium percarbonates; magnesium peroxide; and mixtures thereof.
More preferentially, the process according to the invention comprises the use of at least one persulfate.
For the purposes of the invention, persulfates, also known as peroxysulfates, correspond to SO5 2- anions (peroxomonosulfate anion) or S2O8 2' anions (peroxodisulfate anion) or to compounds comprising at least one of these anions.
Preferably, the persulfates according to the invention are chosen from peroxodisulfates.
According to a preferred embodiment of the invention, the process according to the invention comprises the use of at least one peroxygenated salt chosen from persulfates; preferably from alkali metal persulfates, alkaline-earth metal persulfates, ammonium persulfates, and mixtures thereof; more preferentially from (bis)tetrabutylammonium persulfate, barium persulfate, magnesium persulfate, calcium persulfate, sodium persulfate, potassium persulfate, ammonium persulfate, and mixtures thereof; even more preferentially from sodium persulfate, potassium persulfate, ammonium persulfate, and mixtures thereof; better still from potassium persulfate, ammonium persulfate, and mixtures thereof.
According to the invention, the peroxygenated salt(s) used as compound (a) in one or more compositions that are useful in the process according to the invention preferably represent from 1% to 70% by weight, more preferentially from 5% to 65% by weight, even more preferentially from 10% to 60% by weight, and better still from 20% to 55%, or even from 30% to 55% by weight, relative to the total weight of the composition by weight, relative to the total weight of the composition(s) containing them.
Preferably, the persulfate(s) used as compound(s) (a) in one or more compositions that are useful in the process according to the invention preferably represent from 1% to 70% by weight, more preferentially from 5% to 65% by weight, even more preferentially from 10% to 60% by weight, and better still from 20% to 55%, or even from 30% to 55% by weight, relative to the total weight of the composition by weight, relative to the total weight of the composition(s) containing them.
(b) Osmolytes
The process according to the invention uses one or more osmolytes. Osmolytes are organic molecules, generally of low molecular weight.
In the context of the present invention, the term “osmolyte” does not include carboxylic amino acids, sulfonic amino acids or salts thereof.
Osmolytes may be chosen from:
(i) carbohydrates,
(ii) polyamines, and/or
(iii) compounds of formula (I):
(Rl)(R2)(R3)m-A+-CR4R5-(X)n-Y- (I) in which:
- Ri, R2 and R3, which may be identical or different, represent a C1-C4 alkyl group, preferably a C1-C2 alkyl group, preferably methyl;
- A is N or S;
- m is 0 or 1 ;
- n is 0 or 1 ;
- X is a divalent, linear or branched, saturated or unsaturated alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2); and
- Y- is -COO- or -OSO3 _; it being understood that:
- when A is S, then m = 0 and R4 and R5, which may be identical or different, represent a hydrogen atom or a saturated, unsaturated, linear, branched and/or cyclic, C1-C10, preferably C1-C6, more preferentially C1-C4, hydrocarbon-based chain; optionally interrupted with one or more heteroatoms or groups chosen from -S-, -N=, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (- OH), amino (-NH2), -SH, -COOH, or-CONH2;
- when A is N and m = 0, then R4 represents a hydrogen atom or a saturated, linear or branched C1-C8 alkyl group, preferably a C1-C4 alkyl group; and R5 forms, with the nitrogen atom, a saturated 5- to 8-membered, preferably 5- to 6-membered, heterocycle optionally substituted with one or more groups chosen from hydroxyl or C1-C4 alkyl; and
- when A is N and m = 1 , then R4 and R5, which may be identical or different, represent a hydrogen atom or a saturated, unsaturated, linear, branched and/or cyclic C1-C10, preferably C1-C6, more preferentially C1-C4, hydrocarbon-based chain; optionally interrupted with one or more heteroatoms or groups chosen from -S-, -N=, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (- OH), amino (-NH2), -SH, -COOH or -CONH2.
The carbohydrates that may be used in the process according to the invention include polyols and sugars.
By way of example, the polyols may be chosen from C2-C16 polyols, such as C2-C12 or C2-C8 polyols. Useful polyols may optionally be chosen from diols and triols, and may optionally be linear or branched, saturated or unsaturated, and substituted or unsubstituted. Any stereoisomer of the polyols may be used. According to certain preferred embodiments, the polyols may be chosen from glycols such as ethylene glycol, propylene glycol, butylene glycol, hexylene glycol, pentylene glycol, 1 ,3-propanediol,
diethylene glycol, triethylene glycol, tetraethylene glycol, pentaethylene glycol, dipropylene glycol, caprylyl glycol, glycerol, diglycerol, ethylhexyglycerol or mixtures of two or more of these elements. According to other preferred embodiments, the polyols may be chosen from propanediol, butanediol, pentanediol, hexanediol, heptanediol, octanediol, 1 ,2,6-hexanetriol, 1 ,2,4-butanetriol, trimethylolpropane, 2-butene-1 ,4-diol, 2- ethyl-1 ,3-hexanediol, 2-methyl-2,4-pentanediol, caprylyl glycol, 1 ,2-hexanediol, 1 ,2- pentanediol, 2-ethyl-2-methyl-1 ,3-propanediol, 3,3-dimethyl-1 ,2-butanediol, 2,2-diethyl- 1 ,3-propanediol, 2-methyl-2-propyl-1 ,3-propanediol, 2,4-dimethyl-2,4-pentanediol, 2,5- dimethyl-2,5-hexanediol, 5-hexene-1 ,2-diol, 2-ethyl-1 ,3-hexanediol, 4-methyl-1 ,2- pentanediol, or mixtures of two or more of these elements.
Preferably, the polyols are chosen from glycerol, mannitol, xylitol, ribitol, pinitol, myoinositol, sorbitol and mixtures thereof.
By way of example, the sugars may be chosen from C3-C6 monosaccharides, for instance pentoses and/or derivatives thereof. For example, the sugars may optionally be chosen from xylose, arabinose, ribose, 2-deoxyribose, ribulose, deoxyribulose, arabinose, xylulose, allose, altrose, glucose (including dextrose), glucosamine, mannose, gulose, idose, galactose, talose, sorbose, psicose, fructose, tagatose or combinations of two or more of these sugars. According to one embodiment, the sugars may be chosen from disaccharides, for example sucrose (also known as saccharose), maltose, lactose, cellobiose, trehalose, dextran, or from polysaccharides, for example maltotriose, starch, dextrins, cellulose, glycogen or combinations of two or more thereof.
Preferably, the sugars are chosen from trehalose, glucose, sucrose, fructose, fructans, and mixtures thereof.
The polyamines that may be used in the process according to the invention may constitute functional groups of primary and/or secondary and/or tertiary and/or quaternary amines. By way of example, the polyamines may be chosen from diamines, triamines, tetramines, pentamines and polymeric polyamines or polyimines, for instance hexamethylenediamine, diethylenetriamine, diethylenetetramine, polyethylenimine (PEI), polyvinylamine, polyetheramine, polylysine, ethylenediamine, 1 ,3-diaminopropane, cadaverine, spermidine, spermine, putrescine, tetraethylmethylenediamine, triethylenetetramine, or combinations of two or more of these elements.
Preferably, the polyamines are chosen from putrescine, spermidine, spermine and mixtures thereof.
Preferably, the process according to the invention uses at least one osmolyte chosen from the compounds of formula (I).
Preferably, the process according to the invention uses at least one osmolyte chosen from the compounds of formula (I) which are derivatives of zwitterionic amino acids bearing a quaternary ammonium group and comprising a total of from 1 to 12 carbon atoms, better still from 2 to 10 carbon atoms, even better still from 3 to 8 carbon atoms.
Preferably, the process of the invention uses at least one compound of formula (I) in which Ri and R2 are identical and represent methyl.
Preferably, the process according to the invention uses at least one compound of formula (I) in which A is N.
Preferably, the process according to the invention uses at least one compound of formula (I) in which Y- is -COO-.
More preferably, the process according to the invention uses at least one compound of formula (I) in which Ri and R2 are identical and represent methyl, A is N and Y is -COO-.
Preferably, the process according to the invention uses at least one compound of formula (I) in which R3 is methyl.
Preferably, X is a divalent, linear or branched, saturated or unsaturated alkyl group containing from 1 to 6 carbon atoms, preferably 1 to 4 carbon atoms, preferably 1 to 2 carbon atoms.
Preferentially, the process according to the invention uses at least one compound of formula (I) in which X is a divalent, linear or branched, preferably saturated, alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, optionally substituted with a group chosen from hydroxyl or amino.
Particularly preferably, the process according to the invention uses at least one compound of formula (I) in which X is a divalent, linear or branched, preferably saturated, alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, preferably from 1 to 2 carbon atoms (unsubstituted), such as methylene or ethylene.
Preferably, the process according to the invention uses at least one compound of formula (I) in which R4 and R5, which may be identical or different, may be chosen from a hydrogen atom, a saturated, linear or branched C1-C10, preferably C1-C6, more preferentially C1-C4, alkyl group; a phenyl group; a benzyl group; an alkyl group substituted with a cyclic group (saturated or unsaturated, mono- or bicyclic); a cyclic group (saturated or unsaturated, mono- or bicyclic) substituted with an alkyl group; all these groups being optionally interrupted with one or more heteroatoms or groups chosen from -S-, -N=, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2), -SH, -COOH or -CONH2.
Preferably, the process according to the invention uses at least one compound of formula (I) in which R4 and R5 may be chosen from a hydrogen atom, a saturated, linear or branched C1-C6, preferably C1-C4, alkyl; optionally substituted with a group chosen from hydroxyl (-OH), amino (-NH2), -COOH or -CONH2.
In a particularly preferred manner, when A is N and m = 0, R5 forms, with the nitrogen atom, a saturated 5- to 8-membered, preferably 5- to 6-membered, heterocycle optionally substituted with a hydroxyl group.
More preferentially, the process according to the invention uses at least one compound of formula (I) in which Ri = R2 = methyl, A is N, Y is COO-, and X, if present, is a divalent alkyl group which may be linear or branched, saturated or unsaturated, containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, more preferentially from 1 to 2 carbon atoms.
Preferably, the process according to the invention uses at least one compound of formula (I) in which R4 represents a hydrogen atom.
Preferably, the process according to the invention uses at least one compound of formula (I) in which R5 represents a hydrogen atom or a saturated, linear or branched C1-C6, preferably C1-C4, hydrocarbon-based chain, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2), -SH, -COOH or -CONH2.
Preferentially, the process according to the invention uses at least one compound of formula (I) in which Ri = R2 = R3 = methyl; A is N; Y- is COO; X is a divalent, linear or branched, preferably saturated, alkyl group containing from 1 to 4 carbon atoms, optionally substituted with a group chosen from hydroxyl or amino, preferably 1 to 2 carbon atoms (unsubstituted), such as methylene or ethylene; R4 is hydrogen; and R5 forms, with the nitrogen atom, a saturated 5- to 8-membered, preferably 5- to 6- membered, heterocycle optionally substituted with a hydroxyl group.
Preferentially, the compounds of formula (I) are chosen from one of the compounds indicated in FIG.1.
Preferably, the compounds of formula (I) are chosen from valine betaine, glutamic acid betaine, glutamine betaine, trimethyllysine, glycine betaine (trimethylglycine), histidine betaine, alanine betaine, choline sulfate, pipecolic acid betaine, proline betaine, hydroxyproline betaine, tyrosine betaine, phenylalanine betaine, tryptophan betaine, leucine betaine, isoleucine betaine, dimethylsulfoniopropionate and mixtures thereof.
In a particularly preferred manner, the process according to the invention uses trimethylglycine (also referred to herein as glycine betaine or “betaine”), optionally in combination with at least one additional osmolyte, for example at least one additional compound of formula (I), preferably chosen from the compounds indicated in Figure 1.
According to the invention, the osmolyte(s) used as compound (b) in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
Preferably, the trimethylglycine used as compound (b) in one or more compositions that are useful in the process according to the invention preferably represents from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
(c) Hydroxylated polycarboxylic acids
The process according to the invention uses one or more hydroxylated polycarboxylic acids.
Preferably, the hydroxylated polycarboxylic acid(s) are chosen from the hydroxylated polycarboxylic acid(s) of formula (II) below:
in which formula (II):
- n is an integer between 1 and 5, better still between 1 and 3, preferably n = 1 or 2, more preferably n = 2;
- A is a multivalent, saturated or unsaturated, linear, branched, cyclic or even aromatic hydrocarbon-based group comprising from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, substituted with one or more hydroxyl (OH) groups.
Preferably, A is a multivalent (C1-C6)alkylene and better still (C2-C4)alkylene or phenylene group, substituted with one or more hydroxyl groups.
Preferably, the hydroxylated polycarboxylic acids of formula (II) are alpha-hydroxy acids, for which A is a (C1-C6)alkylene and better still (C2-C4)alkylene group, or a phenylene group, substituted with 1 or 2 hydroxyl groups, preferably 1 hydroxyl group; and n = 1 to 2.
In particular, mention may be made of the hydroxylated polycarboxylic acids of formula (II) in which n = 1 or 2, and A is a di- or trivalent (C1-C6)alkyl group, better still a (C2- C4)alkyl group, substituted with one or more hydroxyl groups, notably 1 or 2 OH, preferably 1 OH.
Preferentially, the hydroxylated polycarboxylic acids may be chosen from:
- citric acid (n = 2 and A trivalent = -CH2-CHOH-CH2-);
- malic acid (n = 1 and A divalent = -CH2-CH(OH)-); and
- tartaric acid (n = 1 and A divalent = -CH(OH)-CH(OH)-).
Even more preferentially, the hydroxylated polycarboxylic acid, a salt thereof or mixtures thereof is chosen from citric acid, a salt thereof and mixtures thereof.
According to the invention, the hydroxylated polycarboxylic acid(s), a salt thereof or mixtures thereof used as compound (c) in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
Preferably, citric acid, a salt thereof or mixtures thereof used as compound (c) in one or more compositions that are useful in the process according to the invention represents from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
Preferably, the citric acid used as compound (c) in one or more compositions that are useful in the process according to the invention represents from 0.05% to 20% by weight, preferably from 0.1% to 10% by weight, more preferentially from 0.5% to 8% by weight, even more preferentially from 0.8% to 5% by weight, better still from 1 .0% to 3% by weight, relative to the total weight of the composition(s) containing them.
(d) Chemical oxidizing agents
According to the present invention, the process may use one or more chemical oxidizing agents other than the peroxygenated salts.
Preferably, the process according to the invention uses one or more chemical oxidizing agents other than the peroxygenated salts.
Preferably, the chemical oxidizing agent(s) are chosen from hydrogen peroxide, hydrogen peroxide-generating systems other than peroxygenated salts, and mixtures thereof.
The hydrogen peroxide-generating systems other than peroxygenated salts may be chosen from urea peroxide, polymeric complexes that can release hydrogen peroxide, oxidases, and mixtures thereof.
As examples of polymeric complexes that can release hydrogen peroxide, mention may be made of polyvinylpyrrolidone/H202 in particular in powder form, and the other polymeric complexes described in US 5 008 093, US 3 376 110 and US 5 183 901 .
Oxidases can produce hydrogen peroxide in the presence of a suitable substrate, for instance glucose in the case of glucose oxidase or uric acid with uricase.
Preferably, the process uses hydrogen peroxide as chemical oxidizing agent.
Moreover, the composition(s) comprising hydrogen peroxide or a hydrogen peroxidegenerating system may also include various adjuvants conventionally used in compositions for dyeing keratin fibers as defined below.
According to a particular mode of the invention, the chemical oxidizing agent(s) other than the peroxygenated salts preferably represent from 0.01% to 30% by weight, preferentially from 0.1% to 20% by weight, better still from 1% to 15% by weight relative to the total weight of the composition(s) containing them.
According to a particular mode of the invention, hydrogen peroxide preferably represents from 0.01% to 30% by weight, preferentially from 0.1% to 20% by weight, better still from 1% to 15% by weight relative to the total weight of the composition(s) containing them.
The process according to the invention may also use one or more of the following compounds.
Compound(s) of amino acid type
The process according to the invention may optionally use one or more compounds of amino acid type.
For the purposes of the present invention, the term “compound of amino acid type” means an organic compound comprising one or more carboxylic acid and/or sulfonic acid functions and one or more amine functions, it being possible for the amine function(s) to be endocyclic, optionally in salt form.
The compounds of amino acid type are different from the osmolytes described previously.
Preferably, the compound(s) of amino acid type are chosen from compounds of amino acid type comprising only one or more carboxylic acid functions (thus not comprising any sulfonic acid functions) and/or salts thereof. Said compounds are also called compounds of aminocarboxylic acid type and are particularly preferred.
When the process of the invention uses one or more compounds of amino acid type, the process according to the invention preferably uses one or more compounds of amino acid type chosen from the compounds corresponding to formula (III) below and/or salts thereof.
The compounds of amino acid type may thus correspond to formula (III):
in which p is an integer equal to 1 or 2, it being understood that:
- when p = 1 , R forms, with the nitrogen atom, a saturated 5- to 8-membered heterocycle, preferably a 5-membered heterocycle, it being possible for this ring to be substituted with one or more groups chosen from hydroxyl or (C1-C4)alkyl;
- when p = 2, R represents a hydrogen atom or a saturated, linear or branched, (C1- C12)alkyl, preferably (C1-C4)alkyl, group, optionally interrupted with one or more heteroatoms or groups chosen from -S-, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2), -SH, -
COOH, -CONH2 or -NH-C(NH)-NH2;
Preferably, when p = 1 , R forms, with the nitrogen atom, a saturated 5-membered heterocycle, this ring not being substituted.
Preferably, p = 2.
Preferably, when p = 2, R represents a hydrogen atom or a saturated, linear or branched (Ci-C4)alkyl group, optionally interrupted with an -S- heteroatom and/or optionally substituted with one or two groups chosen from hydroxyl, amino or -NH-C(NH)-NH2.
Preferentially, p = 2 and R represents a hydrogen atom.
The compounds of amino acid type may also be a salt of a compound of formula (III).
These salts comprise the salts with organic or mineral bases, for example the salts of alkali metals, for instance the lithium, sodium or potassium salts; the salts of alkaline-earth metals, for instance the magnesium or calcium salts, and the zinc salts.
The compounds of amino acid type may be in the form of an optical isomer of L, D or DL configuration, preferably of L configuration.
As examples according to the present invention of compounds in the form of an optical isomer of L configuration, mention may be made of L-proline, L-methionine, L-serine, L- arginine and L-lysine.
Preferably, the compound(s) of amino acid type according to the invention are chosen from glycine, proline, methionine, serine, arginine, lysine, salts thereof (notably alkali metal, alkaline-earth metal or zinc salts) and mixtures thereof.
Preferentially, the compound(s) of amino acid type according to the invention are chosen from glycine, proline, methionine, serine, arginine, salts thereof and mixtures thereof.
Better still, the compound of amino acid type is chosen from glycine, arginine, salts thereof (notably alkali metal, alkaline-earth metal or zinc salts) and mixtures thereof.
Preferably, the compound of amino acid type is arginine.
When they are present, the compound(s) of amino acid type, preferably of aminocarboxylic acid type, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 20% by weight, preferably from 0.05% to 10% by weight, more preferably from 0.1% to 5% by weight, better still from 0.2% to 2% by weight, relative to the total weight of the composition(s) containing them.
In particular, when they are present, the compound(s) of amino acid type chosen from glycine, proline, methionine, serine, arginine, lysine, salts thereof and mixtures thereof, used in one or more compositions that are useful in the process according to the invention, preferably represent from 0.01% to 20% by weight, preferably from 0.05% to 10% by weight, more preferably from 0.1% to 5% by weight and better still from 0.2% to 2% by weight, relative to the total weight of the composition(s) containing them.
Alkaline agent
The process according to the invention may also use one or more mineral, organic or hybrid alkaline agents.
The alkaline agents according to the invention are different from the osmolytes and amino acids described previously.
Preferably, the process according to the invention uses one or more alkaline agents.
For the purposes of the present invention, the terms “alkaline agent” and “basifying agent” are used interchangeably.
The mineral basifying agent(s) are preferably chosen from aqueous ammonia, alkali metal carbonates or bicarbonates such as sodium (hydrogen) carbonate and potassium (hydrogen) carbonate, ammonium (hydrogen) carbonate, alkali metal or alkaline-earth metal phosphates such as sodium phosphates or potassium phosphates, sodium or potassium hydroxides, alkali metal or alkaline-earth metal silicates or metasilicates such as sodium silicate, sodium metasilicate, and mixtures thereof.
The organic basifying agent(s) are preferably chosen from alkanolamines, organic amines other than alkanolamines, oxyethylenated and/or oxypropylenated ethylenediamines, 1 ,3- diaminopropane, spermine, spermidine and mixtures thereof.
The term “alkanolamine” means an organic amine comprising a primary, secondary or tertiary amine function, and one or more linear or branched C1-C8 alkyl groups bearing one or more hydroxyl radicals.
Organic amines chosen from alkanolamines such as monoalkanolamines, dialkanolamines or trialkanolamines comprising one to three identical or different C1-C4 hydroxyalkyl radicals are in particular suitable for performing the invention.
In particular, the alkanolamine(s) are chosen from monoethanolamine (MEA), diethanolamine, triethanolamine, monoisopropanolamine, diisopropanolamine, N,N- dimethylethanolamine, 2-amino-2-methyl-1-propanol, triisopropanolamine, 2-amino-2- methyl-1 ,3-propanediol, 3-amino-1 ,2-propanediol, 3-dimethylamino-1 ,2-propanediol, tris(hydroxymethyl)aminomethane and mixtures thereof.
The organic amine may also be chosen from organic amines of heterocyclic type. Besides histidine that has already been mentioned in the amino acids, mention may in particular be made of pyridine, piperidine, imidazole, triazole, tetrazole and benzimidazole. The organic amine may also be chosen from amino acid dipeptides. As amino acid dipeptides that may be used in the present invention, mention may notably be made of carnosine, anserine and balenine. The organic amine may also be chosen from compounds including a guanidine function. As amines of this type other than arginine that may be used in the present invention, mention may notably be made of creatine, creatinine, 1 ,1- dimethylguanidine, 1 , 1 -diethylguanidine, glycocyamine, metformin, agmatine, n- amidoalanine, 3-guanidinopropionic acid, 4-guanidinobutyric acid and 2- ([amino(imino)methyl]amino)ethane-1 -sulfonic acid.
Use may be made in particular of guanidine carbonate or monoethanolamine hydrochloride as hybrid compounds.
The alkaline agent(s) that are useful according to the invention are preferably chosen from alkanolamines such as monoethanolamine, diethanolamine or triethanolamine; aqueous ammonia, carbonates or bicarbonates such as sodium (hydrogen) carbonate and potassium (hydrogen) carbonate, ammonium (hydrogen) carbonate, alkali metal or alkaline-earth metal silicates or metasilicates such as sodium silicate, sodium metasilicate and mixtures thereof; more preferentially from alkali metal or alkaline-earth metal silicates or metasilicates, such as sodium silicate and metasilicate, and carbonates or bicarbonates, such as ammonium (hydrogen) carbonate, and mixtures thereof, better still from alkali metal or alkaline-earth metal silicates or metasilicates, such as sodium silicate and metasilicate.
When they are present, the alkaline agent(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.1% to 50% by weight, more preferentially from 1% to 40% by weight, better still from 5% to 35% by weight and even better still from 10% to 30% by weight, relative to the total weight of the composition(s) containing them.
When they are present, the alkaline agent(s) chosen from alkali metal or alkaline-earth metal silicates or metasilicates used in one or more compositions that are useful in the process according to the invention preferably represent from 0.1% to 50% by weight, more preferentially from 1% to 40% by weight, better still from 5% to 35% by weight and even better still from 10% to 30% by weight, relative to the total weight of the composition(s) containing them.
Direct dyes
The process according to the invention may optionally use one or more direct dyes.
The term “direct dyes” means natural and/or synthetic dyes, other than oxidation dyes. These are dyes which will spread superficially over the fiber. They may be ionic, for example cationic or anionic, or nonionic.
Examples of suitable direct dyes that may be mentioned include azo direct dyes; (poly)methine dyes such as cyanines, hemicyanines and styryls; carbonyl dyes; azine dyes; nitro(hetero)aryl dyes; tri(hetero)arylmethane dyes; porphyrin dyes; phthalocyanine- based dyes and natural direct dyes, alone or as mixtures.
The direct dyes may be chosen from cationic direct dyes.
Among the cationic direct dyes, mention may be made of dyes bearing endocyclic azo and hydrazono cationic charges, including: Basic Red 51 , Basic Yellow 87 and Basic Orange 31 or derivatives thereof. Mention may also be made of anthraquinone cationic direct dyes, notably HC Blue 16 and HC Blue 17.
The direct dyes may be chosen from anionic direct dyes. Anionic direct dyes are dyes commonly referred to as “acid” direct dyes owing to their affinity for alkaline substances. The term “anionic direct dye” means any direct dye comprising in its structure at least one CO2R or SO3R substituent with R denoting a hydrogen atom or a cation of a metal or of an amine, or an ammonium ion. The anionic dyes may be chosen from direct nitro acid dyes, azo acid dyes, azine acid dyes, triarylmethane acid dyes, indoamine acid dyes, anthraquinone acid dyes, indigoid dyes and natural acid dyes.
Examples that may be mentioned include: a) diaryl anionic azo dyes;
Examples that may be mentioned include: Acid Red 1 , Acid Red 4, Acid Red 13, Acid Red 14, Acid Red 18, Acid Red 27, Acid Red 28, Acid Red 32, Acid Red 33, Acid Red 35, Acid Red 37, Acid Red 40, Acid Red 41 , Acid Red 42, Acid Red 44, Pigment Red 57, Acid Red 68, Acid Red 73, Acid Red 135, Acid Red 138, Acid Red 184, Food Red 1 , Food Red 13, Acid Orange 6, Acid Orange 7, Acid Orange 10, Acid Orange 19, Acid Orange 20, Acid Orange 24, Acid Yellow 6, Acid Yellow 9, Acid Yellow 36, Acid Yellow 199, Food Yellow 3; Acid Violet 7, Acid Violet 14, Acid Blue 113, Acid Blue 117, Acid Black 1 , Acid Brown 4, Acid Brown 20, Acid Black 26, Acid Black 52, Food Black 1 , Food Black 2; Food Yellow 3 or Sunset Yellow; or Acid Red 111 , Acid Red 134, Acid Yellow 38; b) pyrazolone anionic azo dyes, including: Acid Red 195, Acid Yellow 23, Acid Yellow 27, Acid Yellow 76 or Acid Yellow 17; c) anthraquinone dyes, including: Acid Blue 25, Acid Blue 43, Acid Blue 62, Acid Blue 78, Acid Blue 129, Acid Blue 138, Acid Blue 140, Acid Blue 251 , Acid Green 25, Acid Green 41 , Acid Violet 42, Acid Violet 43, Mordant Red 3; EXT Violet No. 2; and Acid Black 48; d) nitro dyes, including: Acid Brown 13; Acid Orange 3; Acid Yellow 1 , the sodium salt of 2,4-dinitro-1-naphthol-7-sulfonic acid, 2-piperidino-5-nitrobenzenesulfonic acid, 2-(4’-N,N- (2”-hydroxyethyl)amino-2’-nitro)anilineethanesulfonic acid, 4-|3-hydroxyethylamino-3- nitrobenzenesulfonic acid; EXT D&C Yellow 7;
e) triarylmethane dyes, including: Acid Blue 1 ; Acid Blue 3; Acid Blue 7, Acid Blue 9; Acid Violet 49; Acid Green 3; Acid Green 5; Acid Green 50; Tetrabromophenol Blue. f) xanthene-based dyes, including: Acid Yellow 73; Acid Red 51 ; Acid Red 52; Acid Red 87; Acid Red 92; Acid Red 95; Acid Violet 9; g) indole-based dyes, including: Acid Blue 74; h) quinoline-based dyes, including: Acid Yellow 2, Acid Yellow 3 and Acid Yellow 5.
Among the natural direct dyes that may be used according to the invention, mention may be made of lawsone, juglone, alizarin, purpurin, carminic acid, kermesic acid, purpurogallin, protocatechaldehyde, indigo, isatin, curcumin, spinulosin, apigenin and orceins. Extracts or decoctions containing these natural dyes may also be used, including henna-based poultices or extracts.
Preferably, the direct dyes are chosen from anionic direct dyes.
Even more preferably, the direct dye(s) are chosen from blue or violet dyes such as Acid Violet 7, Acid Violet 14, Acid Blue 113, Acid Blue 117, Acid Blue 25, Acid Blue 43, Acid Blue 62, Acid Blue 78 Acid Blue 129, Acid Blue 138, Acid Blue 140, Acid Blue 251 , Acid Violet 42, Acid Violet 43 (also referred to as Violet EXT No. 2), Acid Blue 1 ; Acid Blue 3; Acid Blue 7; Acid Blue 9; Acid Violet 49, Acid Violet 9, Acid Blue 74, Basic Blue 6, Basic Blue 7, Basic Blue 9, Basic Blue 26, Basic Blue 41 , Basic Blue 77 (or HC Blue 15), Basic Blue 99, Basic Violet 1 , Basic Violet 2, Basic Violet 3, Basic Violet 10, Basic Violet 14, HC Blue 16, HC Blue 17.
The preferred direct dyes are Violet EXT No. 2, Acid Blue 62, Acid Blue 9, Basic Violet 2, HC Blue 16, HC Blue17 and Tetrabromophenol Blue, more preferentially Violet EXT No.
2, Acid Blue 62, Acid Blue 9, HC Blue 16, HC Blue17 and Tetrabromophenol Blue.
When they are present, the direct dye(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.001% to 10% by weight, preferably from 0.005% to 5% by weight, more preferably from 0.001% to 3% by weight, relative to the total weight of the composition(s) containing them.
Surfactants
The process according to the invention may also use one or more surfactants.
These surfactants may preferably be chosen from anionic surfactants, amphoteric surfactants, nonionic surfactants and cationic surfactants and/or mixtures thereof.
Preferably, the process according to the invention uses one or more surfactants.
Preferably, the process according to the invention uses one or more anionic surfactants.
The term “anionic surfactant” means a surfactant including, as ionic or ionizable groups, only anionic groups. These anionic groups are preferably chosen from the following groups: H2PO2,
As examples of anionic surfactants that may be used in the process according to the invention, mention may be made of alkyl sulfates, alkyl ether sulfates, alkylamido ether sulfates, alkylaryl polyether sulfates, monoglyceride sulfates, alkyl sulfonates, alkylamide
sulfonates, alkylaryl sulfonates, a-olefin sulfonates, paraffin sulfonates, alkyl sulfosuccinates, alkyl ether sulfosuccinates, alkylamide sulfosuccinates, alkyl sulfoacetates, acyl sarcosinates, acyl glutamates, alkyl sulfosuccinamates, acyl isethionates and N-(C1-C4)alkyl-N-acyl taurates, salts of alkyl monoesters of polyglycoside-polycarboxylic acids, acyl lactylates, salts of D-galactoside uronic acids, salts of alkyl ether carboxylic acids, salts of alkylaryl ether carboxylic acids, salts of alkylamido ether carboxylic acids; and the corresponding non-salified forms of all these compounds; the alkyl and acyl groups of all these compounds (unless otherwise mentioned) generally including from 6 to 24 carbon atoms and the aryl group generally denoting a phenyl group.
Among the anionic surfactants, mention may also be made of salts of fatty acids, notably of C8-C24 and preferably C12-C20 fatty acids, other than the (poly)carboxylic acids described previously.
These compounds may be oxyethylenated and then preferably include from 1 to 50 ethylene oxide units.
The salts of C6-C24 alkyl monoesters of polyglycoside-polycarboxylic acids may be chosen from C6-C24 alkyl polyglycoside-citrates, C6-C24 alkyl polyglycoside-tartrates and C6-C24 alkyl polyglycoside-sulfosuccinates.
When the anionic surfactant(s) are in salt form, they may be chosen from alkali metal salts such as the sodium or potassium salt and preferably the sodium salt, ammonium salts, amine salts and in particular amino alcohol salts or alkaline-earth metal salts such as the magnesium salt.
Examples of amino alcohol salts that may notably be mentioned include monoethanolamine, diethanolamine and triethanolamine salts, monoisopropanolamine, diisopropanolamine or triisopropanolamine salts, 2-amino-2-methyl-1-propanol salts, 2- amino-2-methyl-1 ,3-propanediol salts and tris(hydroxymethyl)aminomethane salts.
Alkali metal or alkaline-earth metal salts and in particular the sodium or magnesium salts are preferably used.
The anionic surfactants that may be present may be mild anionic surfactants, i.e. anionic surfactants without a sulfate function.
As regards the mild anionic surfactants, mention may be made in particular of the following compounds and salts thereof, and also mixtures thereof: polyoxyalkylenated alkyl ether carboxylic acids, polyoxyalkylenated alkylaryl ether carboxylic acids, polyoxyalkylenated alkylamido ether carboxylic acids, in particular those including 2 to 50 ethylene oxide groups, alkyl D-galactoside uronic acids, acyl sarcosinates, acyl glutamates and alkylpolyglycoside carboxylic esters.
Use may be made most particularly of polyoxyalkylenated alkyl ether carboxylic acids, for instance lauryl ether carboxylic acid (4.5 OE) sold, for example, under the name Akypo RLM 45 CA from Kao.
Among the anionic surfactants mentioned above, use is preferably made of sulfated surfactants such as alkyl sulfates or alkyl ether sulfates, and acylglutamates, C12-C20 fatty acid salts, more preferentially alkyl sulfates and C12-C20 fatty acid salts.
According to a preferred embodiment, the process of the invention uses one or more surfactants chosen from alkyl sulfates.
When they are present, the surfactant(s), preferably anionic surfactants, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 15% by weight, more preferentially from 0.1% to 10% by weight, better still from 0.3% to 5% by weight, and even better still from 0.5% to 3% by weight of the composition(s) containing them.
Fatty substances
The process according to the invention may also use one or more fatty substances.
Preferably, the process according to the invention uses one or more fatty substances.
For the purposes of the present invention, the term “fatty substance” means an organic compound that is insoluble in water at ordinary room temperature (20-25°C) and at atmospheric pressure (760 mmHg, i.e. 1.013*105 Pa), with a solubility in water of less than 5%, preferably less than 1% and even more preferentially less than 0.1%. The fatty substances generally have in their structure a hydrocarbon-based chain including at least 6 carbon atoms. In addition, the fatty substances are generally soluble in organic solvents under the same temperature and pressure conditions, for instance chloroform, ethanol, benzene, liquid petroleum jelly or decamethylcyclopentasiloxane.
The fatty substances are, moreover, non-(poly)oxyalkylenated and non-(poly)glycerolated. In other words, the fatty substances do not include in their structure a (poly)ethylene oxide or (poly)glycerol or (polypropylene glycol unit.
The fatty substance(s) may be chosen from solid fatty substances and/or liquid fatty substances (also called “oil”), and mixtures thereof.
The term “oil” means a “fatty substance” which is liquid, i.e. which is capable of flowing under the action of its own weight at room temperature (25°C) and at atmospheric pressure (760 mmHg, i.e. 1.013*105 Pa). Preferably, the viscosity at a temperature of 25°C and at a shear rate of 1 s 1 of the oil is between 103 Pa.s and 2 Pa.s. It may be measured using a Thermo Haake RS600 rheometer with cone-plate geometry or an equivalent machine.
For the purposes of the present invention, the term “solid fatty substance” means a fatty substance that is not liquid at room temperature (20-25°C) and at atmospheric pressure (760 mmHg, i.e. 1.013x105 Pa), in particular a solid compound or a compound having a viscosity of greater than 2 Pa.s at a shear rate of 1 s 1 under the conditions mentioned above.
Preferably, the fatty substance(s) are chosen from the liquid fatty substances.
Advantageously, the liquid fatty substances are chosen from:
- fatty alcohols that are liquid at room temperature, bearing a branched and/or unsaturated carbon-based chain containing from 12 to 26 carbon atoms, for instance cetanol, octyldodecanol, linoleyl alcohol, linolenyl alcohol, isostearyl alcohol, oleyl alcohol, 2- hexyldecanol, 2-butyloctanol or 2-undecylpentadecanol;
- C6-Ci6 hydrocarbons;
- hydrocarbons comprising more than 16 carbon atoms;
- silicones and mixtures thereof.
Preferably, the fatty substance(s) are chosen from hydrocarbons comprising more than 16 carbon atoms.
Preferably, the process according to the invention uses one or more fatty substances, preferentially chosen from liquid fatty substances, better still from hydrocarbons comprising more than 16 carbon atoms.
When they are present, the fatty substance(s), preferably liquid fatty substances, used in one or more compositions that are useful in the process according to the invention preferably represent from 0.01% to 20% by weight, preferably from 0.1% to 15% by weight, and preferentially from 0.2% to 5% by weight of the composition(s) containing them.
Associative polymers
The process according to the present invention may also use one or more associative polymers.
Preferably, the process according to the invention uses one or more associative polymers.
It is recalled that “associative polymers” are polymers that are capable, in an aqueous medium, of reversibly associating with each other or with other molecules.
Their chemical structure more particularly comprises at least one hydrophilic zone or group and at least one hydrophobic zone or group.
The term “hydrophobic zone or group” means a radical or polymer with a saturated or unsaturated, linear or branched hydrocarbon-based chain, comprising at least 10 carbon atoms, preferably from 10 to 30 carbon atoms, in particular from 12 to 30 carbon atoms and more preferentially from 18 to 30 carbon atoms.
Preferentially, the hydrocarbon-based group originates from a monofunctional compound. By way of example, the hydrophobic group may be derived from a fatty alcohol such as stearyl alcohol, dodecyl alcohol or decyl alcohol. It may also denote a hydrocarbon-based polymer, for instance polybutadiene.
The associative polymer may be anionic, cationic, amphoteric or nonionic.
Among the associative polymers of anionic type, mention may be made of:
- (a) those including at least one hydrophilic unit and at least one fatty-chain allyl ether unit, more particularly those whose hydrophilic unit is constituted by an ethylenically unsaturated anionic monomer, even more particularly by a vinylcarboxylic acid and most particularly by an acrylic acid or a methacrylic acid or mixtures thereof.
Among these anionic associative polymers, those that are particularly preferred according to the invention are polymers formed from 20% to 60% by weight of acrylic acid and/or of methacrylic acid, from 5% to 60% by weight of lower alkyl (meth)acrylates, from 2% to 50% by weight of fatty-chain allyl ether, and from 0 to 1% by weight of a crosslinking agent which is a well-known copolymerizable unsaturated polyethylenic monomer, for
instance diallyl phthalate, allyl (meth)acrylate, divinylbenzene, (poly)ethylene glycol dimethacrylate or methylenebisacrylamide.
Among the latter polymers, those most particularly preferred are crosslinked terpolymers of methacrylic acid, of ethyl acrylate and of polyethylene glycol (10 EO) stearyl alcohol ether (Steareth-10), notably those sold by the company Ciba under the names Salcare SC 80® and Salcare SC 90®, which are aqueous 30% emulsions of a crosslinked terpolymer of methacrylic acid, of ethyl acrylate and of steareth-10 allyl ether (40/50/10).
- (b) those including i) at least one hydrophilic unit of unsaturated olefinic carboxylic acid type, and ii) at least one hydrophobic unit of the type such as a (C10-C30) alkyl ester of an unsaturated carboxylic acid.
(C10-C30)alkyl esters of unsaturated carboxylic acids that are useful in the invention comprise, for example, lauryl acrylate, stearyl acrylate, decyl acrylate, isodecyl acrylate and dodecyl acrylate, and the corresponding methacrylates, lauryl methacrylate, stearyl methacrylate, decyl methacrylate, isodecyl methacrylate and dodecyl methacrylate.
Anionic polymers of this type are described and prepared, for example, according to patents US 3 915 921 and US 4 509 949.
Among the anionic associative polymers of this type, use will be made more particularly of those formed from 95% to 60% by weight of acrylic acid (hydrophilic unit), 4% to 40% by weight of C10-C30 alkyl acrylate (hydrophobic unit) and 0 to 6% by weight of crosslinking polymerizable monomer, or alternatively those formed from 98% to 96% by weight of acrylic acid (hydrophilic unit), 1% to 4% by weight of C10-C30 alkyl acrylate (hydrophobic unit) and 0.1% to 0.6% by weight of crosslinking polymerizable monomer such as those described previously.
Among said polymers above, the ones most particularly preferred according to the present invention are the products sold by the company Goodrich under the trade names Pemulen TR1®, Pemulen TR2®, Carbopol 1382®, the product sold by the company Lubrizol under the trade name Carbopol ETD 2020 Polymer® (INCI name: Acrylates/C 10-30 Alkyl Acrylate Crosspolymer), the product sold by the company SEPPIC under the name Coatex SX®, and even more preferentially Carbopol ETD 2020 Polymer®.
Mention may also be made of the acrylic acid/lauryl methacrylate/vinylpyrrolidone terpolymer sold under the name Acrylidone LM by the company ISP.
- (c) maleic anhydride/C30-C38 a-olefin/alkyl maleate terpolymers, such as the product (maleic anhydride/C30-C38 a-olefin/isopropyl maleate copolymer) sold under the name Performa 1608® by the company Newphase Technologies.
- (d) acrylic terpolymers comprising: i) approximately 20% to 70% by weight of an a,|3-monoethylenically unsaturated carboxylic acid [A], ii) approximately 20% to 80% by weight of an a,|3-monoethylenically unsaturated nonsurfactant monomer other than [A], iii) approximately 0.5% to 60% by weight of a nonionic monourethane which is the reaction product of a monohydric surfactant with a monoethylenically unsaturated monoisocyanate,
such as those described in patent application EP-A-0 173 109 and more particularly the terpolymer described in Example 3, namely a methacrylic acid/methyl acrylate/behenyl alcohol dimethyl-meta-isopropenylbenzylisocyanate ethoxylated (40 OE) terpolymer, as an aqueous 25% dispersion.
- (e) copolymers including, among their monomers, an a,|3-monoethylenically unsaturated carboxylic acid and an ester of an a,|3-monoethylenically unsaturated carboxylic acid and of an oxyalkylenated fatty alcohol.
Preferentially, these compounds also comprise as monomer an ester of an a, [3- monoethylenically unsaturated carboxylic acid and of a C1-C4 alcohol.
By way of example of compounds of this type, mention may be made of Aculyn 22® sold by the company Rohm and Haas, which is an oxyalkylenated methacrylic acid/ethyl acrylate/stearyl methacrylate terpolymer, and also Aculyn 88®, also sold by the company Rohm and Haas, or Aculyn 28® sold by the company Rohm and Haas, which is an oxyalkylenated methacrylic acid/ethyl acrylate/behenyl methacrylate terpolymer (INCI name: Acrylates/Beheneth-25 Methacrylate Copolymer), and also Novethix L-10 Polymer® sold by Lubrizol.
- (f) amphiphilic polymers including at least one ethylenically unsaturated monomer bearing a sulfonic group, in free or partially or totally neutralized form and comprising at least one hydrophobic part. These polymers may be crosslinked or non-crosslinked. They are preferably crosslinked.
The ethylenically unsaturated monomers bearing a sulfonic group are notably chosen from vinylsulfonic acid, styrenesulfonic acid, (meth)acrylamido(C1-C22)alkylsulfonic acids, N-(C1-C22)alkyl(meth)acrylamido(C1-C22)alkylsulfonic acids such as undecylacrylamidomethanesulfonic acid, and also partially or totally neutralized forms thereof.
More preferentially, use will be made of (meth)acrylamido(C1-C22)alkylsulfonic acids, for instance acrylamidomethanesulfonic acid, acrylamidoethanesulfonic acid, acrylamidopropanesulfonic acid, 2-acrylamido-2-methylpropanesulfonic acid, methacrylamido-2-methylpropanesulfonic acid, 2-acrylamido-n-butanesulfonic acid, 2- acrylamido-2,4,4-trimethylpentanesulfonic acid, 2-methacrylamidododecylsulfonic acid or 2-acrylamido-2,6-dimethyl-3-heptanesulfonic acid, and also partially or totally neutralized forms thereof.
Use will more particularly be made of 2-acrylamido-2-methylpropanesulfonic acid (AMPS), and also partially or totally neutralized forms thereof.
The polymers of this family may be chosen notably from random amphiphilic AMPS polymers modified by reaction with a C6-C22 n-monoalkylamine or di-n-alkylamine, and such as those described in patent application WO 00/31154 (which form an integral part of the content of the description). These polymers may also contain other ethylenically unsaturated hydrophilic monomers chosen, for example, from (meth)acrylic acids, |3- substituted alkyl derivatives thereof or esters thereof obtained with monoalcohols or mono- or polyalkylene glycols, (meth)acrylamides, vinylpyrrolidone, maleic anhydride, itaconic acid or maleic acid, or mixtures of these compounds.
The preferred polymers of this family are chosen from amphiphilic copolymers of AMPS and of at least one ethylenically unsaturated hydrophobic monomer.
These same copolymers may also contain one or more ethylenically unsaturated monomers not including a fatty chain, such as (meth)acrylic acids, ^-substituted alkyl derivatives thereof or esters thereof obtained with monoalcohols or mono- or polyalkylene glycols, (meth)acrylamides, vinylpyrrolidone, maleic anhydride, itaconic acid or maleic acid, or mixtures of these compounds.
These copolymers are notably described in patent application EP-A 750 899, patent US 5 089 578 and in the following publications from Yotaro Morishima:
- Self-assembling amphiphilic polyelectrolytes and their nanostructures, Chinese Journal of Polymer Science, Vol. 18, No. 40, (2000), 323-336;
- Micelle formation of random copolymers of sodium 2-(acrylamido)-2- methylpropanesulfonate and a nonionic surfactant macromonomer in water as studied by fluorescence and dynamic light scattering, Macromolecules, Vol. 33, No. 10 (2000), 3694- 3704;
- Solution properties of micelle networks formed by nonionic moieties covalently bound to an polyelectrolyte: salt effects on rheological behavior - Langmuir, Vol. 16, No. 12, (2000) 5324-5332;
- Stimuli responsive amphiphilic copolymers of sodium 2-(acrylamido)-2- methylpropanesulfonate and associative macromonomers, Polym. Preprint, Div. Polym. Chem., 40(2), (1999), 220-221.
Among these polymers, mention may be made of:
- crosslinked or noncrosslinked, neutralized or non-neutralized copolymers including from 15% to 60% by weight of AMPS units and from 40% to 85% by weight of (C8- C16)alkyl(meth)acrylamide units or of (C8-C16)alkyl (meth)acrylate units relative to the polymer, such as those described in patent application EP-A 750 899;
- terpolymers including from 10 mol% to 90 mol% of acrylamide units, from 0.1 mol% to 10 mol% of AMPS units and from 5 mol% to 80 mol% of n-(C6-C18)alkylacrylamide units, such as those described in patent US-5 089 578.
Mention may also be made of copolymers of totally neutralized AMPS and of dodecyl methacrylate, and also crosslinked and noncrosslinked copolymers of AMPS and of n- dodecylmethacrylamide, such as those described in the Morishima articles mentioned above.
Among the anionic associative polymers according to the invention, preference is given to polymers including i) at least one hydrophilic unit of the olefinic unsaturated carboxylic acid type, and ii) at least one hydrophobic unit of the type such as a (C10-C30) alkyl ester of an unsaturated carboxylic acid (family b)), and copolymers including among their monomers an a,|3-monoethylenically unsaturated carboxylic acid and an ester of an a,|3- monoethylenically unsaturated carboxylic acid and of an oxyalkylenated fatty alcohol (family e)).
Among the cationic associative polymers, mention may be made of: (a) cationic associative polyurethanes;
(b) the compound sold by the company Noveon under the name Aqua CC and which corresponds to the INCI name Polyacrylate- 1 Crosspolymer.
Polyacrylate- 1 Crosspolymer is the product of the polymerization of a mixture of monomers comprising: a di(C1-C4 alkyl)amino(C1-C6 alkyl) methacrylate, one or more C1-C30 alkyl esters of (meth)acrylic acid, a polyethoxylated C10-C30 alkyl methacrylate (20-25 mol of ethylene oxide units), a 30/5 polyethylene glycol/polypropylene glycol allyl ether, a hydroxy(C2-C6 alkyl) methacrylate, and an ethylene glycol dimethacrylate.
(c) quaternized (poly)hydroxyethylcelluloses modified with groups including at least one fatty chain, such as alkyl, arylalkyl or alkylaryl groups including at least 8 carbon atoms, or mixtures thereof. The alkyl radicals borne by the above quaternized celluloses or hydroxyethylcelluloses preferably include from 8 to 30 carbon atoms. The aryl radicals preferably denote phenyl, benzyl, naphthyl or anthryl groups. Examples of quaternized alkylhydroxyethylcelluloses containing C8-C30 fatty chains that may be indicated include the products Quatrisoft LM 200®, Quatrisoft LM-X 529-18-A®, Quatrisoft LM-X 529-18-B® (C12 alkyl) and Quatrisoft LM-X 529-8® (C18 alkyl) sold by the company Aquaion, and the products Crodacel QM®, Crodacel QL® (C12 alkyl) and Crodacel QS® (C18 alkyl) sold by the company Croda and the product Softcat SL 100® sold by the company Aquaion.
(d) cationic polyvinyllactam polymers.
Such polymers are described, for example, in patent application WO-OO/68282.
As cationic poly(vinyllactam) polymers according to the invention, use is notably made of vinylpyrrolidone/dimethylaminopropylmethacrylamide/dodecyl- dimethylmethacrylamidopropylammonium tosylate terpolymers, vinylpyrrolidone/dimethylaminopropylmethacrylamide/cocoyldimethylmethacryl- amidopropylammonium tosylate terpolymers, vinylpyrrolidone/dimethylamino- propylmethacrylamide/lauryldimethylmethacrylamidopropylammonium tosylate or chloride terpolymers.
The amphoteric associative polymers are preferably chosen from those including at least one noncyclic cationic unit. Even more particularly, preference is given to those prepared from or comprising 1 to 20 mol%, preferably 1 .5 to 15 mol% and even more particularly 1.5 to 6 mol% of fatty-chain monomer relative to the total number of moles of monomers.
Amphoteric associative polymers that may be used in the invention are described and prepared, for example, in patent application WO 98/44012.
Among the amphoteric associative polymers, the ones that are preferred are acrylic acid/(meth)acrylamidopropyltrimethylammonium chloride/stearyl methacrylate terpolymers.
The associative polymers of nonionic type that may be used according to the invention are preferably chosen from:
(a) copolymers of vinylpyrrolidone and of fatty-chain hydrophobic monomers, of which examples that may be mentioned include:
- the products Antaron V216® or Ganex V216® (vinylpyrrolidone/hexadecene copolymer), sold by the company ISP;
- the products Antaron V220® or Ganex V220® (vinylpyrrolidone/eicosene copolymer), sold by the company ISP;
(b) copolymers of C1-C6 alkyl methacrylates or acrylates and of amphiphilic monomers including at least one fatty chain, for instance the oxyethylenated methyl acrylate/stearyl acrylate copolymer sold by the company Goldschmidt under the name Antil 208, or the copolymer having the INCI name Acrylates/Beheneth-25 Methacrylate Copolymer, such as the product Novethix L-10 Polymer from Lubrizol.
(c) copolymers of hydrophilic methacrylates or acrylates and of hydrophobic monomers including at least one fatty chain, for instance the polyethylene glycol methacrylate/lauryl methacrylate copolymer.
(d) polyurethane polyethers including in their chain both hydrophilic blocks usually of polyoxyethylenated nature and hydrophobic blocks, which may be aliphatic sequences alone and/or cycloaliphatic and/or aromatic sequences;
(e) polymers with an aminoplast ether backbone containing at least one fatty chain, such as the Pure Thix® compounds sold by the company Sud-Chemie;
(f) celluloses or derivatives thereof, modified with groups including at least one fatty chain, such as alkyl, arylalkyl or alkylaryl groups or mixtures thereof in which the alkyl groups are of C8, and in particular:
* nonionic alkylhydroxyethylcelluloses such as the products Natrosol Plus Grade 330 CS and Polysurf 67 (C16 alkyl) sold by the company Aquaion;
* nonionic nonoxynylhydroxyethylcelluloses such as the product Amercell HM-1500 sold by the company Amerchol;
* nonionic alkylcelluloses, such as the product Bermocoll EHM 100 sold by the company Berol Nobel;
(g) associative guar derivatives, for instance hydroxypropyl guars modified with a fatty chain, such as the product Esaflor HM 22 (modified with a C22 alkyl chain) sold by the company Lamberti; the product Miracare XC 95-3 (modified with a C14 alkyl chain) and the product RE 205-146 (modified with a C20 alkyl chain) sold by Rhodia Chimie.
Preferably, the polyurethane polyethers include at least two hydrocarbon-based lipophilic chains containing from 6 to 30 carbon atoms, separated by a hydrophilic block, the hydrocarbon-based chains possibly being side chains or chains at the end of the hydrophilic block. In particular, it is possible for one or more side chains to be envisaged. In addition, the polymer may include a hydrocarbon-based chain at one end or at both ends of a hydrophilic block.
The polyurethane polyethers may be multiblock, in particular in triblock form. The hydrophobic blocks may be at each end of the chain (for example: triblock copolymer bearing a hydrophilic central block) or distributed both at the ends and in the chain (for example, multiblock copolymer). These same polymers may also be graft polymers or starburst polymers.
The fatty-chain nonionic polyurethane polyethers may be triblock copolymers, the hydrophilic block of which is a polyoxyethylenated chain including from 50 to 1000 oxyethylene groups. The nonionic polyurethane polyethers include a urethane bond between the hydrophilic blocks, whence arises the name.
By extension, also included among the fatty-chain nonionic polyurethane polyethers are those in which the hydrophilic blocks are linked to the lipophilic blocks via other chemical bonds.
As examples of nonionic fatty-chain polyurethane polyethers that may be used in the invention, use may also be made of Rheolate 205® bearing a urea function, sold by the company Rheox, or Rheolate® 208, 204 or 212, and also Acrysol RM 184®.
Mention may also be made of the product Elfacos T210® containing a C12-C14 alkyl chain, and the product Elfacos T212® containing a Ci8 alkyl chain, from Akzo.
The product DW 1206B® from Rohm & Haas containing a C20 alkyl chain and a urethane bond, sold at a solids content of 20% in water, may also be used.
Use may also be made of solutions or dispersions of these polymers, notably in water or in an aqueous/alcoholic medium. Examples of such polymers that may be mentioned include Rheolate® 255, Rheolate® 278 and Rheolate® 244 sold by the company Rheox. Use may also be made of the products DW 1206F and DW 1206J sold by the company Rohm & Haas.
The polyurethane polyethers that may be used according to the invention are in particular those described in the article by G. Fonnum, J. Bakke and Fk. Hansen - Colloid Polym. Sci., 271 , 380-389 (1993).
It is even more particularly preferred to use a polyurethane polyether that may be obtained by polycondensation of at least three compounds comprising (i) at least one polyethylene glycol comprising from 150 to 180 mol of ethylene oxide, (ii) stearyl alcohol or decyl alcohol, and (iii) at least one diisocyanate.
Such polyurethane polyethers are sold notably by the company Rohm & Haas under the names Aculyn 46® and Aculyn 44® [Aculyn 46® is a polycondensate of polyethylene glycol containing 150 or 180 mol of ethylene oxide, of stearyl alcohol and of methylenebis(4-cyclohexyl isocyanate) (SMDI), at 15% by weight in a matrix of maltodextrin (4%) and water (81%); Aculyn 44® is a polycondensate of polyethylene glycol containing 150 or 180 mol of ethylene oxide, of decyl alcohol and of methylenebis(4-cyclohexyl isocyanate) (SMDI), at 35% by weight in a mixture of propylene glycol (39%) and water (26%)].
Preferably, the associative polymer(s) are chosen from anionic associative polymers.
Preferably, the associative polymer(s) are chosen from acrylic or methacrylic acid homopolymers or copolymers.
Preferably also, the associative polymer(s) are chosen from polymers including i) at least one hydrophilic unit of the olefinic unsaturated carboxylic acid type, and ii) at least one hydrophobic unit of the type such as a (C10-C30) alkyl ester of unsaturated carboxylic acid, the copolymers including among their monomers an a,|3-monoethylenically
unsaturated carboxylic acid and an ester of an a,|3-monoethylenically unsaturated carboxylic acid and of an oxyalkylenated fatty alcohol, and mixtures thereof.
Preferably, the process according to the invention uses one or more associative polymers, preferentially chosen from anionic associative polymers, preferably from homopolymers or copolymers of acrylic or methacrylic acid.
When they are present, the associative polymer(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 10% by weight, better still from 0.1% to 8% by weight, even better still from 0.2% to 5% by weight of the composition(s) containing them.
Non-associative polysaccharides
The process according to the present invention may also use one or more non-associative polysaccharides, which are thus different from the above associative polymers.
Preferably, the process according to the invention uses one or more non-associative polysaccharides.
In the present invention, the term “polysaccharide” means a polymer constituted of sugar units. The term “sugar unit” means an oxygen-bearing hydrocarbon-based compound containing several alcohol functions, with or without aldehyde or ketone functions, and which includes at least 4 carbon atoms. The sugar units may be optionally modified by substitution, and/or by oxidation and/or by dehydration.
The sugar units that may be included in the composition of the polysaccharides of the invention are preferably derived from the following sugars: glucose, galactose, arabinose, rhamnose, mannose, xylose, fucose, anhydrogalactose, galacturonic acid, glucuronic acid, mannuronic acid, galactose sulfate, anhydrogalactose sulfate and fructose.
Non-associative polysaccharides that may notably be mentioned include the following polymers, alone or as a mixture: a) tree or shrub exudates, including:
- acacia gum (branched polymer of galactose, arabinose, rhamnose and glucuronic acid);
- ghatti gum (polymer derived from arabinose, galactose, mannose, xylose and glucuronic acid);
- karaya gum (polymer derived from galacturonic acid, galactose, rhamnose and glucuronic acid);
- gum tragacanth (polymer of galacturonic acid, galactose, fucose, xylose and arabinose); b) gums derived from algae, including:
- agar (polymer derived from galactose and anhydrogalactose);
- alginates (polymers of mannuronic acid and of glucuronic acid);
- carrageenans and furcellerans (polymers of galactose sulfate and of anhydrogalactose sulfate); c) gums derived from seeds or tubers, including:
- guar gum (polymer of mannose and galactose);
- locust bean gum (polymer of mannose and galactose);
- fenugreek gum (polymer of mannose and galactose);
- tamarind gum (polymer of galactose, xylose and glucose);
- konjac gum (polymer of glucose and mannose); d) microbial gums, including:
- xanthan gum (polymer of glucose, mannose acetate, mannose/pyruvic acid and glucuronic acid);
- gellan gum (polymer of partially acylated glucose, rhamnose and glucuronic acid);
- scleroglucan gum (glucose polymer); e) polymers extracted from plants, including:
- celluloses (glucose polymers);
- starches (glucose polymers) and
- inulin.
These polymers may be physically or chemically modified. As physical treatment, mention may notably be made of a heat treatment. Chemical treatments that may be mentioned include esterification, etherification, amidation or oxidation reactions. These treatments make it possible to produce polymers that may notably be nonionic, anionic or amphoteric.
In particular, the guar gums, locust bean gums, starches and celluloses may be modified/treated.
The guar gums that may be used according to the invention may be modified with C1-C6 (poly)hydroxyalkyl groups. Among the C1-C6 (poly)hydroxyalkyl groups, examples that may be mentioned include hydroxymethyl, hydroxyethyl, hydroxypropyl and hydroxybutyl groups. These guar gums are well known in the prior art and may be prepared, for example, by reacting corresponding alkene oxides, for instance propylene oxides, with the guar gum so as to obtain a guar gum modified with hydroxypropyl groups. The degree of hydroxyalkylation preferably ranges from 0.4 to 1 .2 and corresponds to the number of alkylene oxide molecules consumed by the number of free hydroxyl functions present on the guar gum.
Such guar gums optionally modified with hydroxyalkyl groups are sold, for example, under the trade names Jaguar HP8, Jaguar HP60 and Jaguar HP120 by the company Rhodia Chimie.
The botanical origin of the starches that may be used in the present invention may be cereals or tubers. Thus, the starches are chosen, for example, from corn starch, rice oat, starch, cassava starch, barley starch, potato starch, wheat starch, sorghum starch, pea starch and tapioca starch. It is also possible to use hydrolyzates of the starches mentioned above. The starch is preferably derived from potato.
Starch phosphates, notably distarch phosphates or compounds rich in distarch phosphate, will preferentially be used, for instance the product sold under the references Prejel VA- 70-T AGGL (gelatinized hydroxypropyl cassava distarch phosphate), Prejel TK1 (gelatinized cassava distarch phosphate) or Prejel 200 (gelatinized acetylated cassava distarch phosphate) by the company Avebe, or Structure Zea from National Starch (gelatinized corn distarch phosphate).
According to the invention, amphoteric starches may also be used, these amphoteric starches comprising one or more anionic groups and one or more cationic groups. The anionic and cationic groups may be bonded to the same reactive site of the starch molecule or to different reactive sites; they are preferably bonded to the same reactive
site. The anionic groups may be of carboxylic, phosphate or sulfate type, preferably carboxylic type. The cationic groups may be of primary, secondary, tertiary or quaternary amine type.
The polysaccharides that may be used according to the invention may be cellulose-based polymers.
According to the invention, the term “cellulose-based polymer” refers to any polysaccharide compound bearing in its structure sequences of glucose residues linked together via (3-1 ,4 bonds; in addition to unsubstituted celluloses, the cellulose derivatives may be anionic, cationic, amphoteric or nonionic.
Cellulose-based polymers are also known as celluloses.
Thus, the cellulose-based polymers that may be used according to the invention may be chosen from unsubstituted celluloses, including those in a microcrystalline form, and cellulose ethers.
Among these cellulose-based polymers, cellulose ethers, cellulose esters and cellulose ethers-esters are distinguished.
Among the cellulose esters are mineral esters of cellulose (cellulose nitrates, sulfates, phosphates, etc.), organic cellulose esters (cellulose monoacetates, triacetates, amidopropionates, acetatebutyrates, acetatepropionates or acetatetrimellitates, etc.), and mixed organic/mineral esters of cellulose, such as cellulose acetatebutyrate sulfates and cellulose acetatepropionate sulfates. Among the cellulose ether esters, mention may be made of hydroxypropylmethylcellulose phthalates and ethylcellulose sulfates.
Among the cellulose ethers that may be mentioned are (Ci-C4)alkylcelluloses, such as methylcelluloses and ethylcelluloses (for example Ethocel Standard 100 Premium from Dow Chemical); (poly)hydroxy(Ci-C4)alkylcelluloses, such as hydroxymethylcelluloses, hydroxyethylcelluloses (for example Natrosol 250 HHR sold by Ashland) and hydroxypropylcelluloses (for example Klucel EF from Aquaion); mixed (poly)hydroxy(Ci- C4)alkyl(Ci-C4)alkylcelluloses, such as hydroxypropylmethylcelluloses (for example Methocel E4M from Dow Chemical), hydroxyethylmethylcelluloses, hydroxyethylethylcelluloses (for example Bermocoll E 481 FQ from Akzo Nobel) and hydroxybutylmethylcelluloses.
Among the anionic cellulose ethers, mention may be made of (poly)carboxy(CI- C4)alkylcelluloses and salts thereof. Examples that may be mentioned include carboxymethylcelluloses, carboxymethylmethylcelluloses (for example Blanose 7M from the company Aquaion) and carboxymethylhydroxyethylcelluloses, and the sodium salts thereof.
Among the cationic cellulose ethers, mention may be made of cationic cellulose derivatives such as cellulose copolymers or cellulose derivatives grafted with a water- soluble quaternary ammonium monomer, and notably described in patent US 4 131 576, such as (poly)hydroxy(C1-C4)alkylcelluloses, for instance hydroxymethyl-, hydroxyethyl- or hydroxypropylcelluloses notably grafted with a methacryloylethyltrimethylammonium, methacrylamidopropyltrimethylammonium or dimethyldiallylammonium salt. The commercial products corresponding to this definition are more particularly the products
sold under the names Celquat® L 200 and Celquat® H 100 by the company National Starch.
Preferably, the non-associative polysaccharide(s) are chosen, alone or as mixtures, from celluloses, guar gums and starches, preferentially from celluloses.
Better still, the non-associative polysaccharides are chosen, alone or as a mixture, from cellulose ethers, cellulose esters, cellulose ether esters and guar gums, and preferably from cellulose ethers.
In a particularly preferred manner, the non-associative polysaccharide(s) are chosen from (C1-C4)alkylcelluloses such as methylcelluloses and ethylcelluloses; (poly)hydroxy(C1- C4)alkylcelluloses such as hydroxymethylcelluloses, hydroxyethylcelluloses and hydroxypropylcelluloses; mixed (poly)hydroxy(C1-C4)alkyl-(C1-C4)alkylcelluloses such as hydroxypropyl-methylcelluloses, hydroxyethyl-methylcelluloses, hydroxyethylethylcelluloses and hydroxybutyl-methylcelluloses.
Preferably, the process according to the invention uses one or more non-associative polysaccharides, preferentially chosen from celluloses, guar gums, starches, and mixtures thereof.
When they are present, the non-associative polysaccharide(s) used in one or more compositions that are useful in the process according to the invention preferably represent from 0.05% to 10% by weight, better still from 0.1% to 8% by weight and even better still from 0.2% to 5% by weight of the composition(s) containing them.
Additives
The composition(s) of the lightening process according to the invention may also comprise one or more additives, among which mention may be made of mineral thickeners, antidandruff agents, anti-seborrheic agents, agents for preventing hair loss and/or for promoting hair regrowth, vitamins and provitamins including panthenol, sunscreens, mineral or organic pigments, plasticizers, solubilizers, opacifiers or nacreous agents, antioxidants, sequestrants, fragrances, and preserving agents.
Needless to say, a person skilled in the art will take care to select this or these optional additional compound(s) such that the advantageous properties intrinsically associated with the composition(s) of the process according to the invention are not, or are not substantially, adversely affected by the envisaged addition(s).
The above additives may generally be present in an amount, for each of them, of between 0 and 20% by weight relative to the total weight of the composition(s) containing them.
2. Process
According to the lightening process of the invention, keratin fibers may be treated with the compounds described previously in one or more application steps.
According to the process of the invention, keratin fibers may be treated with one or more compositions which contain, together or separately, the compounds described previously.
According to a preferred embodiment, compounds (b) and (c), i.e. the osmolyte(s) (b) and the hydroxylated polycarboxylic acid(s), a salt thereof or mixtures thereof (c) are applied
together to the keratin fibers. Preferably, compounds (b) and (c) are contained in the same composition.
According to a preferred embodiment, the keratin fibers are treated in one application step, all the compounds (a), (b) and (c) being applied together to the fibers.
According to this embodiment, the process comprises a step of applying to the keratin fibers a composition (C) comprising compounds (a), (b), (c), and optionally (d) as defined previously.
The process according to the invention may comprise, prior to the step of applying composition (C), a step of mixing a composition (C1) comprising compounds (a), (b) and (c), and an oxidizing composition (O) comprising the compound(s) (d).
This mixing step is preferably performed at the time of use, just before application to the hair of composition (C) resulting from the mixing.
Preferably, compositions (C1) and (O) are mixed in a (C1)/(O) weight ratio ranging from 0.1 to 2, preferentially from 0.3 to 1 .5, better still from 0.5 to 1 .
As a variant, the process according to the invention may comprise, prior to the step of applying composition (C), a step of mixing compounds (b) and (c) with a composition (C2) comprising the compound(s) (a), and optionally with an oxidizing composition (O) comprising the compound(s) (d).
Preferably, composition (C) may be applied to wet or dry keratin fibers.
It is left in place on the fibers for a time generally of from 1 minute to 1 hour and preferably from 5 minutes to 55 minutes.
The temperature during the lightening process conventionally ranges from room temperature (between 15°C and 25°C) to 80°C and preferably from room temperature to 60°C.
After the treatment, the keratin fibers are optionally rinsed with water, optionally undergo washing with a shampoo followed by rinsing with water, and are then dried or left to dry.
According to another preferred embodiment, the keratin fibers are treated in several application steps, the process using the compounds (a), (b), (c) and optionally (d) described previously from at least two compositions applied separately to the keratin fibers.
According to this embodiment, the process of the invention may involve:
(i) applying to the keratin fibers a composition (C3) comprising the compounds (a), and optionally (d), and
(ii) applying to the keratin fibers a composition (C4) comprising compounds (b) and (c).
According to this embodiment, step (ii) may be performed before or after step (i). Preferably, step (ii) is performed after step (i).
Step (ii) of applying composition (C4) may be repeated several times, with or without rinsing between the applications.
Preferably, the process involves, prior to step (i), a step of mixing a composition (C2) comprising the compound(s) (a) with an oxidizing composition (O) comprising the compound(s) (d).
Preferably, compositions (C3) and (C4) may be applied to wet or dry keratin fibers.
Composition (C3) is left to stand on the fibers for a period generally from 1 minute to 1 hour, preferably from 5 minutes to 55 minutes.
Composition (C4) is left to stand on the fibers for a period generally from 1 minute to 30 minutes, better still from 5 minutes to 15 minutes.
Composition (C4) may be applied before or after composition (C3).
Preferably, the process comprises a step of rinsing the keratin fibers between steps (i) and (ii).
The temperature during the lightening process conventionally ranges from room temperature (between 15°C and 25°C) to 80°C and preferably from room temperature to 60°C.
After the treatment, the keratin fibers are optionally rinsed with water, optionally undergo washing with a shampoo followed by rinsing with water, and are then dried or left to dry.
3. Compositions
Another subject of the invention is a composition (C) comprising the compounds (a), (b), (c) and optionally (d) as defined previously.
As indicated previously, according to a first preferred embodiment of the invention, the process may use the compounds (a), (b), (c) and optionally (d) described previously in a single step of applying a composition (C) to keratin fibers.
Preferably, composition (C) comprises one or more additional oxidizing agents (d), different from the peroxygenated salts, composition (C) then being a ready-to-use composition.
Preferably, composition (C) is aqueous. Preferably, the pH of composition (C), when it is aqueous, ranges from 8 to 13, preferentially from 9 to 12.
Preferably, composition (C) is derived from the mixing of a composition (C1) comprising (a), (b) and (c) and an oxidizing composition (O) which comprises a chemical oxidizing agent (d) different from the peroxygenated salts (a).
Preferably, composition (C1) comprises one or more alkaline agents as described previously.
Preferably, composition (C1) comprises one or more surfactants as described previously.
Preferably, composition (C1) comprises one or more fatty substances as described previously.
Preferably, composition (C1) comprises one or more associative polymers as described previously.
Composition (C1) may optionally comprise one or more compounds of amino acid type described previously.
Composition (C1) may optionally comprise one or more direct dyes described previously.
Composition (C1) is preferably anhydrous. The term “anhydrous composition” refers to a composition which comprises little or no water, notably less than 0.5% water, better still less than 0.1% water, even better still less than 0.05% water, relative to the total weight of the composition. In particular, composition (C1) does not comprise any water added during its preparation, the water that may be present possibly being provided by the starting materials used during its preparation.
Preferably, composition (C1) is a pulverulent composition.
According to a particular embodiment, composition (C1) may be derived from the mixing of compounds (b) and (c) with a composition (C2) comprising the other compounds of composition (C1) described above.
According to this embodiment, composition (C2) is preferably anhydrous, and is preferably a pulverulent composition.
The oxidizing composition (O) which comprises a chemical oxidizing agent (d) different from the peroxygenated salts (a) is preferably an aqueous composition. In particular, it comprises more than 10% by weight of water, preferably more than 30% by weight of water and even more advantageously more than 50% by weight of water relative to the total weight of the oxidizing composition (O). Preferably, the oxidizing composition (O) comprises water in a content ranging from 10% to 95% by weight, preferably from 30% to 90% by weight, better still from 50% to 80% by weight, relative to the total weight of the oxidizing composition (O).
The pH of the oxidizing composition (O), when it is aqueous, is preferably less than 7, preferably between 1 and 5, preferentially between 1 .5 and 4.5. This pH may be adjusted to the desired value by using one or more acidifying agents, which may notably be chosen from those described previously.
As indicated previously, according to a second preferred embodiment of the invention, the process may use the compounds (a), (b), (c) and optionally (d) described previously from two compositions (C3) and (C4) applied separately to keratin fibers.
According to this embodiment, composition (C3) comprises one or more compounds (a) and optionally one or more additional oxidizing agents (d), different from peroxygenated salts.
Preferably, composition (C3) comprises one or more additional oxidizing agents (d), different from the peroxygenated salts, composition (C3) then being a ready-to-use composition.
Preferably, composition (C3) is aqueous. Preferably, the pH of composition (C3), when it is aqueous, ranges from 8 to 13, preferentially from 9 to 12.
Preferably, composition (C3) is derived from the mixing of a composition (C2) comprising compound(s) (a) and an oxidizing composition (O) as defined previously.
Preferably, composition (C2) comprises one or more alkaline agents as described previously.
Preferably, composition (C2) comprises one or more surfactants as described previously.
Preferably, composition (C2) comprises one or more fatty substances as described previously.
Preferably, composition (C2) comprises one or more associative polymers as described previously.
Composition (C2) may optionally comprise one or more compounds of amino acid type described previously.
Composition (C2) may optionally comprise one or more direct dyes described previously.
Preferably, composition (C2) is anhydrous within the meaning defined previously.
Composition (C4) comprises compounds (b) and (c) as described previously.
Preferably, composition (C4) comprises one or more surfactants as described previously.
Preferably, composition (C4) comprises one or more fatty substances as described previously.
Preferably, composition (C4) comprises one or more associative polymers as described previously, and notably one or more polysaccharides, preferably one or more cationic polysaccharides.
Composition (C4) may optionally comprise one or more compounds of amino acid type described previously.
Composition (C4) may optionally comprise one or more direct dyes described previously.
Preferably, composition (C4) is aqueous.
A subject of the invention is also the use of a composition comprising compounds (a), (b), (c), and optionally (d) as defined previously, for lightening keratin fibers, and in particular the hair.
4. Multi-compartment device/kit
Another subject of the invention is a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition (C1) comprising compounds (a), (b) and (c) as described previously, and at least a second compartment containing an oxidizing composition (O) comprising one or more compounds (d) as described previously.
Another subject of the invention is a device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition (C2) comprising the compound(s) (a) as described previously, at least a second compartment containing a composition (C4) comprising compounds (b) and (c) as described previously, and optionally at least a third compartment containing an oxidizing agent composition (O) comprising one or more compounds (d) as described previously.
The compositions of the device according to the invention are packaged in separate compartments, optionally accompanied by suitable application means, which may be identical or different, such as fine brushes, coarse brushes or sponges.
The device mentioned above may also be equipped with a means for dispensing the desired mixture onto the hair, for instance the devices described in patent FR 2 586 913.
The examples that follow serve to illustrate the invention without, however, being limiting in nature.
Examples
In the examples that follow, all the amounts are given as mass percentages of active material (AM) relative to the total weight of the composition (unless otherwise mentioned).
Compositions
Compositions A, A1 and A2 were prepared from the compounds whose contents are indicated in the table below:
- 0.1 g of trimethylglycine (glycine betaine) and 0.1 g of citric acid to 4 g of composition A to obtain a composition A’, and
- 0.1 g of trimethylglycine (glycine betaine) to 4 g of composition A to obtain a composition A”.
At the time of use, compositions A’ and A” were respectively mixed with an oxidizing agent O comprising 9% hydrogen peroxide in the weight ratio 1+1.5.
Each of the mixtures A’+O and A”+O was then applied to a lock of natural chestnut-brown hair (tone height 4) at a rate of 10 g of mixture per 1 g of lock of hair.
After a leave-on time of 50 min at 33°C under an occlusive aluminum foil-type system, the locks were rinsed, and then washed and dried at 60°C in an oven.
Colorimetric evaluation
The lightening of the hair was evaluated in the L*a*b* system, using a Konica Minolta CM- 3600A spectrocolorimeter (illuminant D65, angle 10°, specular component included) in the CIELab system.
In this system, L* represents the intensity of the color, a* indicates the green/red axis and b* the yellow/blue axis. The higher the value of L*, the lighter the color.
Evaluation of the condition of the hair
The integrity of the hair was evaluated with regard to several parameters:
- the tensile strength: the solidity of the hair which can be measured by the tensile test. This measurement for determining the tensile mechanical properties of the hair is performed using a commercial tool, the MTT600 (mini Tensile Tester) from the company Dia Stron. The modulus of elasticity and the breaking strength of the hair are measured: the higher the values, the stronger the hair;
- quantitative evaluation of cysteic acid and of free proteins at the surface of the hair: these compounds are released during oxidation of the hair; the higher the content of cysteic acid and free proteins, the more damaged the hair.
The cysteic acid present in the hair is assayed by means of a Hitachi L8900 amino acid autoanalyzer; 20 mg of finely cut hair are transferred into a culture tube (Duran). 2.5 ml of 9 N hydrochloric acid are added to the tube with magnetic stirring under nitrogen for 1 min at 2 bar.
The solution is heated in the tube in an oil bath at 110°C for 16 hours. The pH is adjusted to between 1 .6 and 1 .8 and diluted to 50 ml with ultrapure water. The solution is then filtered and analyzed in the autoanalyzer.
The quantitative analysis of the labile proteins (i.e. the free proteins) is performed with the same equipment as for the cysteic acid assay.
20 mg of finely cut hair are transferred into a 1.5 ml Eppendorf tube. 400 pl of 0.2 M tris(hydroxymethyl)aminomethane base (TRIZMA base) and 400 pl of 0.2 M 2- mercaptoethanol are added to the tube and stirred with a thermomixer set at 37°C for 16 hours at 700 rpm. After 16 h of extraction, the residual contents are transferred into a Duran tube using a liquipette, and concentrated under compressed air for 4 hours until the solvent has completely evaporated off.
1 ml of 9N hydrochloric acid is added to each tube, which are then heated for 16 hours at 110°C. The hydrolyzed solution is concentrated under compressed air for 12 hours until
evaporation is complete, 1 ml of pH 2 buffer is added, followed by vortexing, filtration and analysis on the Hitachi autoanalyzer.
Results
Table 2]
The results show that a composition according to the invention, with equivalent lightening performance, maintains better hair integrity (significant increase in the modulus of elasticity and the breaking strength, and lower amounts of cysteic acid and of labile proteins) relative to the comparative composition.
Claims
[Claim 1] A process for lightening keratin fibers, preferably human keratin fibers, notably the hair, in which said fibers are treated, in one or more steps, with one or more compositions containing, together or separately:
(a) one or more peroxygenated salts;
(b) one or more osmolytes;
(c) one or more hydroxylated polycarboxylic acids, a salt thereof or mixtures thereof, and
(d) optionally, one or more additional oxidizing agents, different from the peroxygenated salts.
[Claim 2] The process as claimed in the preceding claim, in which the peroxygenated salt(s) (a) are chosen from alkali metal, alkaline-earth metal or ammonium persulfates, perborates and percarbonates, magnesium peroxide and mixtures thereof, preferably from alkali metal persulfates, alkaline-earth metal persulfates, ammonium persulfates, and mixtures thereof; more preferentially from (bis)tetrabutylammonium persulfate, barium persulfate, magnesium persulfate, calcium persulfate, sodium persulfate, potassium persulfate, ammonium persulfate, and mixtures thereof; even more preferentially from sodium persulfate, potassium persulfate, ammonium persulfate, and mixtures thereof; better still from potassium persulfate, ammonium persulfate, and mixtures thereof.
[Claim 3] The process as claimed in either of the preceding claims, in which the osmolyte(s) (b) are chosen from carbohydrates, polyamines and the compounds of formula (I):
(Ri)(R2)(R3)m-A+-CR4R5-(X)n-Y- (I) in which:
- Ri, R2 and R3, which may be identical or different, represent a C1-C4 alkyl group, preferably a C1-C2 alkyl group, preferably methyl;
- A is N or S;
- m is 0 or 1 ;
- n is 0 or 1 ;
- X is a divalent, linear or branched, saturated or unsaturated alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2); and
- Y’ is -COO- or -OSO3 _; it being understood that:
- when A is S, then m = 0 and R4 and R5, which may be identical or different, represent a hydrogen atom or a saturated, unsaturated, linear, branched and/or cyclic, C1-C10, preferably C1-C6, more preferentially C1-C4, hydrocarbon-based chain; optionally interrupted with one or more heteroatoms or groups chosen from -S-, -N=, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2), -SH, -COOH, or-CONH2;
- when A is N and m = 0, then R4 represents a hydrogen atom or a saturated, linear or branched C1-C8 alkyl group, preferably a C1-C4 alkyl group; and R5 forms, with
the nitrogen atom, a saturated 5- to 8-membered, preferably 5- to 6-membered, heterocycle optionally substituted with one or more groups chosen from hydroxyl or C1-C4 alkyl; and
- when A is N and m = 1 , then R4 and R5, which may be identical or different, represent a hydrogen atom or a saturated, unsaturated, linear, branched and/or cyclic C1-C10, preferably C1-C6, more preferentially C1-C4, hydrocarbon-based chain; optionally interrupted with one or more heteroatoms or groups chosen from -S-, -N=, -NH- or -C(NH)- and/or optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (-NH2), -SH, -COOH or -CONH2.
[Claim 4] The process as claimed in the preceding claim, in which the osmolyte(s) (b) are chosen from the compounds of formula (I), preferably from the compounds of formula (I) in which Ri and R2 are identical and represent methyl, A is N, Y is -COO-, and X, when it is present, is a divalent, linear or branched, saturated or unsaturated alkyl group containing from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, preferably 1 to 2 carbon atoms.
[Claim 5] The process as claimed in the preceding claim, in which R4 represents a hydrogen atom and R5 represents a hydrogen atom or a saturated, linear or branched C1-C6, preferably C1-C4, hydrocarbon-based chain, optionally substituted with one or more groups chosen from hydroxyl (-OH), amino (- NH2), -SH, -COOH or -CONH2.
[Claim 6] The process as claimed in any one of the preceding claims, in which the osmolyte(s) (b) are chosen from valine betaine, glutamic acid betaine, glutamine betaine, trimethyllysine, glycine betaine (trimethylglycine), histidine betaine, alanine betaine, choline sulfate, pipecolic acid betaine, proline betaine, hydroxyproline betaine, tyrosine betaine, phenylalanine betaine, tryptophan betaine, leucine betaine, isoleucine betaine, dimethylsulfoniopropionate and mixtures thereof, preferably glycine betaine (trimethylglycine).
[Claim 7] The process as claimed in any one of the preceding claims, in which the hydroxylated polycarboxylic acid(s), a salt thereof or mixtures thereof (c) are chosen from the compounds corresponding to formula (II) below:
in which:
- n is an integer between 1 and 5, better still between 1 and 3, preferably n = 1 or 2, more preferably n = 2;
- A is a multivalent, saturated or unsaturated, linear, branched, cyclic or even
aromatic hydrocarbon-based group comprising from 1 to 6 carbon atoms, preferably from 1 to 4 carbon atoms, substituted with one or more hydroxyl (OH) groups; preferably, A being a multivalent (C1-C6)alkylene and better still (C2- C4)alkylene group, or a phenylene group, substituted with one or more hydroxyl groups.
[Claim 8] The process as claimed in any one of the preceding claims, in which the hydroxylated polycarboxylic acid(s), a salt thereof or mixtures thereof (c) are chosen from the compounds corresponding to formula (II) in which A is a (C1- C6)alkylene and better still (C2-C4)alkylene group, or a phenylene group, substituted with 1 or 2 hydroxyl groups, preferably 1 hydroxyl group; and n = 1 or 2; preferably chosen from the compounds corresponding to formula (II) in which n = 1 or 2, and A is a di- or trivalent (C1-C6)alkyl and better still (C2-C4)alkyl group, substituted with one or more hydroxyl groups, notably 1 or 2 OH, preferably 1 OH; better still chosen from citric acid, malic acid, tartaric acid, salts thereof and mixtures thereof; more preferentially, chosen from citric acid, salts thereof and mixtures thereof, better still citric acid.
[Claim 9] The process as claimed in any one of the preceding claims, in which the lightening process uses one or more additional oxidizing agents (d), different from peroxygenated salts, preferably chosen from hydrogen peroxide, hydrogen peroxide-generating systems different from peroxygenated salts and mixtures thereof, preferentially hydrogen peroxide.
[Claim 10] The process as claimed in any one of the preceding claims, in which the lightening process uses one or more direct dyes, preferably chosen from nonionic direct dyes, anionic direct dyes, cationic direct dyes and mixtures thereof, preferably from azo direct dyes; (poly)methine dyes such as cyanines, hemicyanines and styryls; carbonyl dyes; azine dyes; nitro(hetero)aryl dyes; tri(hetero)arylmethane dyes; porphyrin dyes; phthalocyanine dyes and natural direct dyes, alone or as mixtures.
[Claim 11] The process as claimed in any one of the preceding claims, in which the lightening process uses one or more alkaline agents, preferably chosen from aqueous ammonia, alkaline carbonates or bicarbonates such as sodium (hydrogen) carbonate and potassium (hydrogen) carbonate, ammonium (hydrogen) carbonate, alkali metal or alkaline-earth metal phosphates such as sodium phosphate or potassium phosphate, sodium or potassium hydroxides, alkali metal or alkaline- earth metal silicates or metasilicates such as sodium metasilicate and sodium silicate, and mixtures thereof, preferentially from alkali metal or alkaline-earth metal silicates or metasilicates, such as sodium silicate and metasilicate, and carbonates or bicarbonates and mixtures thereof, better still from alkali metal or alkaline-earth metal silicates or metasilicates, such as sodium silicate and metasilicate, and mixtures thereof.
[Claim 12] The process as claimed in any one of the preceding claims, which comprises a step of applying to the keratin fibers a composition (C) comprising compounds (a), (b) and (c), and optionally (d) as defined in any one of the preceding claims, preferably a composition (C) comprising compounds (a), (b), (c) and (d).
[Claim 13] The process as claimed in claim 12, which comprises, prior to the step of applying composition (C), a step of mixing
- a composition (C1) comprising compounds (a), (b) and (c), and
- an oxidizing agent (O) comprising the compound(s) (d).
[Claim 14] The process as claimed in claim 12, which comprises, prior to the step of applying composition (C), a step of mixing compounds (b) and (c) with a composition (C2) comprising the compound(s) (a), and optionally with an oxidizing composition (O) comprising the compound(s) (d).
[Claim 15] The process as claimed in any one of claims 1 to 11 , which involves:
(i) applying to keratin fibers a composition (C3) comprising the compounds (a), and optionally (d), preferably a composition (C3) comprising compounds (a) and (d),
(ii) applying to the keratin fibers a composition (C4) comprising compounds (b) and (c), step (ii) being performed before or after step (i), preferably after step (i).
[Claim 16] The process as claimed in the preceding claim, which involves, prior to step (i), a step of mixing a composition (C2) comprising the compound(s) (a) with an oxidizing composition (O) comprising the compound(s) (d).
[Claim 17] A composition comprising compounds (a), (b), (c), and optionally (d) as defined in any one of claims 1 to 11 , preferably compounds (a), (b), (c) and (d).
[Claim 18] The use of a composition (C) as defined in the preceding claim, for lightening keratin fibers, and in particular the hair.
[Claim 19] A device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition (C1) comprising compounds (a), (b) and (c) as defined in any one of claims 1 to 11 , and at least a second compartment containing an oxidizing composition (O) comprising one or more compounds (d) as defined in claim 9.
[Claim 20] A device containing at least two compartments, for lightening keratin fibers, comprising at least a first compartment containing a composition (C2) comprising the compound(s) (a) as defined in any one of claims 1 to 11 , at least a second compartment containing a composition (C4) comprising compounds (b) and (c) as defined in any one of claims 1 to 11 , and optionally at least a third compartment
containing an oxidizing composition (O) comprising one or more compounds (d) as defined in claim 9.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR2314614A FR3157123A1 (en) | 2023-12-20 | 2023-12-20 | Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid. |
| FRFR2314614 | 2023-12-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025132928A1 true WO2025132928A1 (en) | 2025-06-26 |
Family
ID=89661543
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/087612 Pending WO2025132928A1 (en) | 2023-12-20 | 2024-12-19 | Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid |
Country Status (2)
| Country | Link |
|---|---|
| FR (1) | FR3157123A1 (en) |
| WO (1) | WO2025132928A1 (en) |
Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3376110A (en) | 1967-01-30 | 1968-04-02 | Gen Aniline & Film Corp | Solid stabilized hydrogen peroxide compositions |
| US3915921A (en) | 1974-07-02 | 1975-10-28 | Goodrich Co B F | Unsaturated carboxylic acid-long chain alkyl ester copolymers and tri-polymers water thickening agents and emulsifiers |
| US4131576A (en) | 1977-12-15 | 1978-12-26 | National Starch And Chemical Corporation | Process for the preparation of graft copolymers of a water soluble monomer and polysaccharide employing a two-phase reaction system |
| US4509949A (en) | 1983-06-13 | 1985-04-09 | The B. F. Goodrich Company | Water thickening agents consisting of copolymers of crosslinked acrylic acids and esters |
| EP0173109A2 (en) | 1984-08-23 | 1986-03-05 | DeSOTO, INC. | Alkali soluble latex thickeners |
| FR2586913A1 (en) | 1985-09-10 | 1987-03-13 | Oreal | PROCESS FOR FORMING IN IN SITU A COMPOSITION CONSISTING OF TWO PARTS CONDITIONED SEPARATELY AND DISPENSING ASSEMBLY FOR IMPLEMENTING SAID METHOD |
| US5008093A (en) | 1989-11-08 | 1991-04-16 | Gaf Chemicals Corporation | Anhydrous complexes of PVP and hydrogen peroxide |
| US5089578A (en) | 1986-03-28 | 1992-02-18 | Exxon Research And Engineering Company | Hydrophobically associating terpolymers containing sulfonate functionality |
| US5183901A (en) | 1992-01-24 | 1993-02-02 | Isp Investments Inc. | Urea-hydrogen peroxide-polyvinylpyrrolidone |
| EP0750899A2 (en) | 1995-06-30 | 1997-01-02 | Shiseido Company Limited | An emulsifier or solubilizer which consists of a water soluble amphiphilic polyelectrolyte, and an emulsified composition or a solubilized composition and an emulsified cosmetic or a solubilized cosmetic containing it |
| WO1998044012A1 (en) | 1997-03-31 | 1998-10-08 | Calgon Corporation | Ampholyte polymers for use in personal care products |
| WO2000031154A1 (en) | 1998-11-23 | 2000-06-02 | Sofitech N.V. | Invertible emulsions stabilised by amphiphilic polymers and application to bore fluids |
| WO2000068282A1 (en) | 1999-05-07 | 2000-11-16 | Isp Investments Inc. | Conditioning/styling terpolymers |
| EP1390000B1 (en) * | 2001-05-02 | 2006-11-02 | Finnfeeds Finland OY | Use of betaine in hair care |
| EP1905421A1 (en) * | 2006-09-29 | 2008-04-02 | Kao Corporation | Hair dye composition |
| US20180116930A1 (en) * | 2016-11-02 | 2018-05-03 | L'oreal | Hair lightening compositions and methods of use |
| US20220062142A1 (en) * | 2020-08-26 | 2022-03-03 | L'oreal | Hair lightening compositions providing damage protection and sensorial benefits and methods of use |
| FR3115204A1 (en) * | 2020-10-15 | 2022-04-22 | L'oreal | Hair Lightening Compositions WITH DAMAGE PROTECTION AND SENSORY BENEFITS AND METHODS OF USE |
| WO2023247610A1 (en) * | 2022-06-22 | 2023-12-28 | L'oreal | Composition for lightening keratin fibres and process for lightening keratin fibres using said composition |
-
2023
- 2023-12-20 FR FR2314614A patent/FR3157123A1/en active Pending
-
2024
- 2024-12-19 WO PCT/EP2024/087612 patent/WO2025132928A1/en active Pending
Patent Citations (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3376110A (en) | 1967-01-30 | 1968-04-02 | Gen Aniline & Film Corp | Solid stabilized hydrogen peroxide compositions |
| US3915921A (en) | 1974-07-02 | 1975-10-28 | Goodrich Co B F | Unsaturated carboxylic acid-long chain alkyl ester copolymers and tri-polymers water thickening agents and emulsifiers |
| US4131576A (en) | 1977-12-15 | 1978-12-26 | National Starch And Chemical Corporation | Process for the preparation of graft copolymers of a water soluble monomer and polysaccharide employing a two-phase reaction system |
| US4509949A (en) | 1983-06-13 | 1985-04-09 | The B. F. Goodrich Company | Water thickening agents consisting of copolymers of crosslinked acrylic acids and esters |
| EP0173109A2 (en) | 1984-08-23 | 1986-03-05 | DeSOTO, INC. | Alkali soluble latex thickeners |
| FR2586913A1 (en) | 1985-09-10 | 1987-03-13 | Oreal | PROCESS FOR FORMING IN IN SITU A COMPOSITION CONSISTING OF TWO PARTS CONDITIONED SEPARATELY AND DISPENSING ASSEMBLY FOR IMPLEMENTING SAID METHOD |
| US5089578A (en) | 1986-03-28 | 1992-02-18 | Exxon Research And Engineering Company | Hydrophobically associating terpolymers containing sulfonate functionality |
| US5008093A (en) | 1989-11-08 | 1991-04-16 | Gaf Chemicals Corporation | Anhydrous complexes of PVP and hydrogen peroxide |
| US5183901A (en) | 1992-01-24 | 1993-02-02 | Isp Investments Inc. | Urea-hydrogen peroxide-polyvinylpyrrolidone |
| EP0750899A2 (en) | 1995-06-30 | 1997-01-02 | Shiseido Company Limited | An emulsifier or solubilizer which consists of a water soluble amphiphilic polyelectrolyte, and an emulsified composition or a solubilized composition and an emulsified cosmetic or a solubilized cosmetic containing it |
| WO1998044012A1 (en) | 1997-03-31 | 1998-10-08 | Calgon Corporation | Ampholyte polymers for use in personal care products |
| WO2000031154A1 (en) | 1998-11-23 | 2000-06-02 | Sofitech N.V. | Invertible emulsions stabilised by amphiphilic polymers and application to bore fluids |
| WO2000068282A1 (en) | 1999-05-07 | 2000-11-16 | Isp Investments Inc. | Conditioning/styling terpolymers |
| EP1390000B1 (en) * | 2001-05-02 | 2006-11-02 | Finnfeeds Finland OY | Use of betaine in hair care |
| EP1905421A1 (en) * | 2006-09-29 | 2008-04-02 | Kao Corporation | Hair dye composition |
| US20180116930A1 (en) * | 2016-11-02 | 2018-05-03 | L'oreal | Hair lightening compositions and methods of use |
| US20220062142A1 (en) * | 2020-08-26 | 2022-03-03 | L'oreal | Hair lightening compositions providing damage protection and sensorial benefits and methods of use |
| FR3115204A1 (en) * | 2020-10-15 | 2022-04-22 | L'oreal | Hair Lightening Compositions WITH DAMAGE PROTECTION AND SENSORY BENEFITS AND METHODS OF USE |
| WO2023247610A1 (en) * | 2022-06-22 | 2023-12-28 | L'oreal | Composition for lightening keratin fibres and process for lightening keratin fibres using said composition |
Non-Patent Citations (6)
Also Published As
| Publication number | Publication date |
|---|---|
| FR3157123A1 (en) | 2025-06-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3389618B1 (en) | Process for treating keratin fibres using an aqueous composition comprising a combination of particular alkoxysilanes | |
| FR3082735A1 (en) | HAIR COLORING COMPOSITION COMPRISING AN OXIDATION DYE, A SCLEROGLUCAN GUM AND AN ASSOCIATIVE POLYMER. | |
| JP2004534790A (en) | Use of dicationic compounds for dyeing human keratin fibers and compositions containing the same | |
| FR3082742A1 (en) | HAIR COLORING COMPOSITION COMPRISING AN OXIDATION DYE, A SCLEROGLUCAN GUM, AND AN ALKYLPOLYGLYCOSIDE. | |
| FR3082740A1 (en) | HAIR COLORING COMPOSITION COMPRISING AN OXIDATION DYE, A SCLEROGLUCAN GUM, AN ALKALNOLAMINE AND A MINERAL ALKALINE AGENT. | |
| FR3082736A1 (en) | HAIR COLORING COMPOSITION COMPRISING AN OXIDATION DYE, A SCLEROGLUCANE GUM AND AN ALKALINE AGENT OF AMINO ACID TYPE. | |
| ES3012798T3 (en) | Hair dyeing process comprising a step of dyeing with henna and/or indigo and a treatment step comprising the application of a composition comprising a buffer system | |
| EP1462092A1 (en) | Dyeing composition for keratin fibers comprising a hydroxycarboxylic acid or a salt thereof, ready to use composition comprising the preceding, dyeing process and kit | |
| EP2677996B1 (en) | Composition for dyeing keratinous fibres comprising a direct dye having a disulphide/thiol functional group, a thickening polymer, an ethoxylated fatty alcohol and/or a nonionic surfactant, an alkaline agent and a reducing agent | |
| FR3044902A1 (en) | PROCESS FOR TREATING KERATIN FIBERS USING AN AQUEOUS COMPOSITION COMPRISING AN ASSOCIATION OF PARTICULAR ALCOXYSILANES | |
| WO2025132928A1 (en) | Process for lightening keratin fibers using a peroxygenated salt, an osmolyte and a hydroxylated polycarboxylic acid | |
| WO2019121631A1 (en) | Process for treating keratin fibers using a halochromic fluorescein direct dye, an alkaline revealer and then an acidic eraser | |
| FR3082741A1 (en) | HAIR COLORING COMPOSITION COMPRISING AN OXIDATION DYE, A SCLEROGLUCAN GUM AND A PARTICULAR CARBOXYLIC ACID. | |
| FR3082737A1 (en) | HAIR COLORING COMPOSITION COMPRISING AN OXIDATION DYE, A SCLEROGLUCAN GUM, AND A FIXING POLYMER. | |
| US11273112B2 (en) | Process for treating keratin fibers using a halochromic triarylmethane direct dye, an alkaline revealer and then an acidic eraser | |
| US11813349B2 (en) | Hair colouring compositions in gel or emulsion-gel form | |
| FR3102362A1 (en) | ALKALINE COMPOSITION COMPRISING AT LEAST THREE DIFFERENT ALKALINE AGENTS FOR THE TREATMENT OF KERATINIC FIBERS, METHODS AND USES | |
| EP1810662B1 (en) | Non-detergent hair fixing cosmetic composition comprising an ionic fixing polymer and an ester of polyethylene glycol and fatty acid, process of fixing hair and uses | |
| FR3083098A1 (en) | KERATIN FIBER LOOPING PROCESS COMPRISING THE APPLICATION TO THE FIBERS OF A COMPOSITION CONTAINING THIOLACTIC ACID | |
| FR3113377A1 (en) | COMPOSITIONS AND METHODS FOR MODIFYING HAIR COLOR | |
| FR3157162A3 (en) | COMPOSITIONS AND METHODS FOR TREATING HAIR | |
| FR3157165A3 (en) | COMPOSITIONS AND METHODS FOR TREATING CURLY HAIR | |
| FR3157170A3 (en) | HAIR TREATMENT PROCESSES | |
| WO2025129301A1 (en) | Methods for treating keratin fibers with osmolytes, carboxylic acid and amino acids | |
| WO2025129289A1 (en) | Methods for treating keratin fibers in a lightening process |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24834731 Country of ref document: EP Kind code of ref document: A1 |