IFF10001-WO-PCT METHODS FOR REDUCING SYRUP VISCOSITY CROSS-REFERENCE TO RELATED APPLICATION [001] This application claims priority from U.S. Provisional Application No. 63/609,436, filed December 13, 2023, the contents of which are hereby incorporated by reference in their entirety. INCORPORATION BY REFERENCE OF THE SEQUENCE LISTING [002] The present application is being filed with a Sequence Listing in electronic format. The Sequence Listing is provided as a file entitled IFF10001WOPCTSeqList.xml, created on December 12, 2024, which is 12,084 bytes in size. The information in the electronic format of the Sequence Listing is incorporated by reference in its entirety. FIELD OF THE INVENTION [003] Provided herein are methods and compositions for reducing the viscosity of syrup produced as a byproduct of fermentation processes. BACKGROUND [004] Processes for producing fermentation products from biomass yield not only desirable fermentation products, e.g., ethanol, but also liquid and solid byproducts from which valuable co- products may be recovered. For example, whole stillage, the liquids and solids remaining after separation of the desired fermentation product (e.g., ethanol), can be further processed into proteinaceous ingredients for animal feed and/or oil for biodiesel production, feed or food additives or products, or other bio-renewable products. [005] Whole stillage consists of both suspended and dissolved solids at various ratios depending on the biomass feedstock, processing, and the fermentation conditions employed. In a typical dry mill corn ethanol facility, most of the suspended solids are removed as roughly 35% weight (wt) solids wet cake, while the majority of the water containing dissolved solids are split into a recycled liquid stream or backset and thin stillage. Dewatered wet cake can be dried to provide “Distillers Dried Grains” (DDG) used as nutrient in animal feed. Thin stillage is typically evaporated to provide condensate and syrup or may alternatively be recycled directly to the slurry tank as backset. Condensate may either be forwarded to a methanator before being discharged or may be recycled to the slurry tank. The syrup may be blended into DDG or added to the wet cake before drying to produce Distillers Dried Grain with Solubles (DDGS). Oil may also be recovered from the thin stillage and/or syrup/evaporated centrate as a co-product for use in biodiesel production or other biorenewable products.
IFF10001-WO-PCT [006] Depending on fermentation characteristics and the addition of various compounds and enzymes during fermentation and/or upstream processes, the characteristics of the syrup may be drastically affected, specifically (but not limited to) composition and rheology. Syrup viscosity can be a significant issue for biorefineries, for example, by decreasing flow characteristics, coating walls, damaging pumps, plugging piping networks and centrifuges as well as increasing fouling rates. Increases in viscosity can therefore significantly impact plant efficiency and the quality and amount of co-product produced. [007] Thus, there is a need for methods and compositions capable of reducing syrup viscosity. The methods and compositions provided herein address these and other needs in the art. SUMMARY OF THE INVENTION [008] Provided herein are methods for reducing syrup viscosity, including: (a) liquefying a starch-containing material with an alpha-amylase to produce a starch liquefact; (b) saccharifying the starch liquefact with a glucoamylase to produce a saccharification product; (c) fermenting the saccharification product using a fermenting microorganism to produce a fermentation product; (d) recovering the fermentation product to produce a whole stillage; (e) separating the whole stillage into thin stillage and wet cake; and (f) concentrating the thin stillage to produce a syrup; where a viscosity reducing composition including a cellulase, a hemicellulase, a protease, or any combination thereof, is added during step (f). In some embodiments, the viscosity reducing composition is added to the thin stillage, to an intermediate product formed during concentrating the thin stillage to produce a syrup, and/or to the syrup produced in step (f). In some embodiments, step (b) and step (c) are combined as a simultaneous saccharification and fermentation step (SSF). In some embodiments, the cellulase includes a cellobiohydrolase, an endoglucanase, a beta- glucosidase, or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase BGL, or any combination thereof. In some embodiments, the cellulase includes an endoglucanase, where the endoglucanase has an amino acid sequence with at least 60, 70, 80, 90, 95, 98, or 100% sequence identity to the sequence set forth by SEQ ID NOs: 4 or 7. In some embodiments, the cellulase is derived from a fungus. In some embodiments, the cellulase is derived from a strain of Trichoderma. In some embodiments, the cellulase is derived from a strain of Trichoderma reesei. In some embodiments, the hemicellulase includes a xylanase. In some embodiments, the hemicellulase includes a GH10 xylanase. In some embodiments, the hemicellulase includes a sequence having at least 60% sequence identity to the sequence set forth by SEQ ID NO: 1. In some embodiments, the protease includes a sequence having at least 60% sequence identity to the sequence set forth by SEQ ID
IFF10001-WO-PCT NO: 3. In some embodiments, the protease includes an amino acid sequence having at least 60, 70, 80, 90, 95, 98, or 100% sequence identity to the sequence set forth by SEQ ID NOs: 3 or 9. In some embodiments, the viscosity reducing composition further includes an alpha-amylase. In some embodiments, the alpha-amylase includes an amino acid sequence having at least 60, 70, 80, 90, 95, 98, or 100% sequence identity to the sequence set forth by SEQ ID NOs: 5 or 6. In some embodiments, (i) a composition including a cellulase, a hemicellulase, a protease, or any combination thereof is added during step (a), step (b), and/or step (c); or (ii) where step (b) and step (c) are combined as a simultaneous saccharification and fermentation step (SSF) and a composition including a cellulase, a hemicellulase, a protease, or any combination thereof is added during step (a) and/or the SSF. In some embodiments, the composition including a cellulase, a hemicellulase, a protease, or any combination thereof comprises the viscosity reducing composition. In some embodiments, the viscosity of the syrup is decreased compared to a syrup not treated with the viscosity reducing composition. In some embodiments, the starch-containing material is corn. In some embodiments, the starch-containing material is wheat. [009] Each of the aspects and embodiments described herein are capable of being used together, unless excluded either explicitly or clearly from the context of the embodiment or aspect. BRIEF DESCRIPTION OF THE DRAWINGS [0010] FIG. 1 shows the syrup flow rankings (mean ± SD) of Viscous Syrup following either no enzyme treatment (Viscous Syrup – Nil) or treatment with a Trichoderma Whole Cellulase, Engineered Trichoderma Whole Cellulase 1, Engineered Trichoderma Whole Cellulase 2, Thermostable Xylanase, or Thermostable Protease. The Baseline Syrup (Baseline Syrup – Nil) ranking is also shown. DETAILED DESCRIPTION [0011] The use of biomass as feedstock for biorefining has emerged as an environmentally friendly means of producing usable products from sustainable resources. Exemplary beneficial fermentation products produced from biomass include, but are not limited to, alcohols, such as industrial or potable alcohols (e.g., ethanol); biochemicals, such as organic acids (e.g., acetic acid, lactic acid), amino acids (e.g., lysine), and vitamins (e.g., vitamin B, vitamin C); sweeteners, such as sugar, fructose, low calorie sweeteners, syrups (e.g., high fructose corn syrup); and soluble fiber. However, in addition to producing the fermentation product, the processes also produce byproducts (fermentation byproducts) such as liquids and solids that contain valuable components, e.g., protein, oil, that can be sold as fermentation co-products, such as ingredients for animal feed
IFF10001-WO-PCT and/or oil for biodiesel production, feed or food additives or products, or other bio-renewable products. [0012] The conversion of biomass to a fermentation product such as alcohol typically involves multiple processing steps, also referred to herein as unit operations, and reagents, each of which is important for maximizing conversion efficiency, throughput, consistency, concentration, and/or yield. By way of example, a common production process for producing ethanol by fermentation of a biomass feedstock typically involves unit operations including, but not limited to, milling or grinding feedstock, liquefaction, saccharification, fermentation, and distillation, and reagents such as enzymes and additional components, e.g., microorganisms and/or chemicals, may be added during a unit operation to facilitate the reaction. For example, during alcohol production, liquefaction includes thinning a mash via partial hydrolysis to lower its viscosity. Enzymes, e.g., alpha-amylases, may be used to facilitate the thinning process. During saccharification, complex carbohydrates of the thinned mash are converted to monosaccharides. Again, enzymes, e.g., glucoamylases, may be used to facilitate the conversion process. The resulting sugars are then converted by yeasts to ethanol during the fermentation process step. It is also possible to include enzymes, e.g., glucoamylases, fungal alpha amylase, and trehalase, to control the glucose profile during fermentation. Reagents, such as enzymes, may also be used in the steps of milling and distillation to facilitate the process. In some cases, the saccharification and fermentation steps may be combined into a single simultaneous saccharification and fermentation (SSF) step. Other well- known processes include raw starch hydrolysis (RSH), which includes milling the starch- containing material and then performing SSF below the initial gelatinization temperature. Enzymes, e.g., acid fungal amylases, glucoamylases, may also be used in such processes to produce fermentation products. [0013] Fermentation products from alcohol production processes are typically removed as liquid during a distillation step, which leaves behind other liquids and solids that may be referred to herein as fermentation byproducts. A fermentation byproduct as used herein refers to the remaining liquids and solids that can be further processed to recover a fermentation co-product. For example, in some cases, a fermentation byproduct is a whole stillage, which includes liquids and solids that can be separated into, e.g., thin stillage and wet cake, respectively. In some cases, thin stillage and/or wet cake may be further processed to produce fermentation co-products. For example, whole stillage consisting of both suspended and dissolved solids may be separated into wet cake, containing a majority of the suspended solids, and thin stillage containing dissolved solids. Dewatered wet cake can be dried to provide “Distillers Dried Grains” (DDG) used as nutrient in animal feed. Thin stillage is typically evaporated to provide condensate and syrup or
IFF10001-WO-PCT may alternatively be recycled directly to the slurry tank as backset. Condensate may either be forwarded to a methanator before being discharged or may be recycled to the slurry tank. The syrup may be blended into DDG or added to the wet cake before drying to produce Distillers Dried Grain with Solubles (DDGS). Oil from the thin stillage and/or syrup/evaporated centrate may also be recovered as a co-product for use, for example, in biodiesel production or other biorenewable products. [0014] To increase and/or improve fermentation product and/or co-product yield and recovery, various compounds and enzymes may be added during fermentation and/or upstream processes. Such strategies can impact syrup characteristics, such as composition and rheology, dramatically. Indeed, even the characteristics of the fermentation can drastically affect the syrup byproduct. As shown in the Examples (see, Section II), the use of enzymes, such as hemicellulases and cellulases, during unit operations prior to distillation, e.g., during saccharification, fermentation, or SSF, caused increases in syrup viscosity. Increases in syrup viscosity can be a significant issue for biorefineries, for example, by decreasing flow characteristics, coating walls, damaging pumps, plugging piping networks and centrifuges as well as increasing fouling rates. Increases in viscosity can therefore significantly impact plant efficiency and the quality and amount of co-product produced. [0015] However, as further demonstrated in the Examples, it was unexpectedly found that addition of cellulases, hemicellulases, proteases, and combinations thereof, to the syrup decreased the syrup viscosity. Such enzymes and combinations thereof are referred to generally herein as a viscosity reduction composition. In some embodiments, treating the syrup with a viscosity reduction composition improves oil recovery from the syrup. Thus, provided herein are methods including viscosity reduction compositions containing cellulases, hemicellulases, proteases, and combinations thereof that decrease syrup viscosity. In some embodiments, the viscosity reducing composition may include or further include an alpha-amylase. [0016] The headings provided herein are not limitations of the various aspects or embodiments of this disclosure which can be had by reference to the specification as a whole. The section headings used herein are for organizational purposes only and are not to be construed as limiting the subject matter described. The reader will appreciate that statements made in one section may apply to other sections. Any terms defined may be more fully defined by reference to the specification as a whole. [0017] All publications, including patent documents, scientific articles, and databases, referred to in this application are incorporated by reference in their entirety for all purposes to the same extent as if each individual publication were individually incorporated by reference. Nothing herein is to
IFF10001-WO-PCT be construed as an admission that such publications constitute prior art to the claims appended hereto. If a definition set forth herein is contrary to or otherwise inconsistent with a definition set forth in the patents, applications, published applications, and other publications that are herein incorporated by reference, the definition set forth herein prevails over the definition that is incorporated herein by reference. Definitions [0018] Definitions of terms may appear throughout the specification. It is to be understood that this disclosure is not limited to particular embodiments described, as such may, of course, vary. It is also to be understood that the terminology used herein is for the purpose of describing particular embodiments only and is not intended to be limiting. [0019] It must be noted that as used herein and in the appended claims, the singular forms "a," "an," and “the” include plural referents unless the context clearly dictates otherwise. For example, “a” or “an” include “at least one” and “one or more.” [0020] The terms “comprising,” “comprises,” and “comprised of” as used herein are synonymous with “including,” “includes,” “containing,” “contains,” and grammatical variants thereof, and are inclusive or open-ended and do not exclude additional, non-recited members, elements, or method steps. The terms “comprising,” “comprises,” “comprised of,” “including,” includes,” “containing,” “contains,” and grammatical variants thereof also include the term “consisting of.” [0021] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art. [0022] As used herein, the term “ethanol” refers to ethanol produced as a result of a biological fermentation process. [0023] As used herein, the phrase “recovering ethanol” refers to the purification and/or isolation of ethanol. Suitably, the recovery results in ethanol that is substantially free of other components (e.g. contaminants). Therefore, the recovery may result in an alcohol that is at least about 90% pure, at least about 95% pure, at least 99% pure. In some embodiments, the recovery may result in an alcohol that is at least about 99.9% pure. [0024] As used herein, the term “whole stillage” includes the material that remains at the end of the distillation process after recovery of the fermentation product, e.g., ethanol. [0025] As used herein, “thin stillage” refers to the separated liquid fraction of the whole stillage. [0026] As used herein, “wet cake” refers to the separated solid fraction of the whole stillage. [0027] As used herein, the term “oil” refers to a co-product extractable from a byproduct of a biological fermentation process.
IFF10001-WO-PCT [0028] As used herein, the phrase “recovering oil” refers to the purification, extraction, and/or isolation of oil. Suitably, the recovery results in oil that is substantially free of other components (e.g. contaminants). Therefore, the recovery may result in an oil that is at least about 90% pure, at least about 95% pure, at least 99% pure. In some embodiments, the recovery may result in an oil that is at least about 99.9% pure. [0029] As used herein, the term “Enzyme Commission” Number, abbreviated “EC", refers to enzyme nomenclature recommendations of the Nomenclature Committee of the International Union of Biochemistry and Molecular Biology (NC-IUBMB), as generally known to one skilled in the art (e.g., see enzyme nomenclature from NC-IUBMB, 1992 (Academic Press, San Diego, California), including supplements 1-5 published in 1994 (Eur. I. Biochem., 223: 1-5), 1995 (Eur. I. Biochem. 232: 1-6); 1996 (Eur. J. Biochem, 237: 1-5), 1997 (Eur. J. Biochem. 250: 1-6) and 1999 (Eur. J. Biochem. 264: 610-650), respectively. Likewise, the nomenclature is regularly supplemented and updated (see, e.g., chem.qmul.ac.uk/iubmb/enzyme/mdex.html). [0030] As used herein, the term “cellulase” means enzymes that hydrolyze cellulosic material. Such enzymes include endoglucanase(s), cellobiohydrolase(s), beta-glucosidase(s), or combinations thereof. The two basic approaches for measuring cellulolytic activity include: (1) measuring the total cellulolytic activity, and (2) measuring the individual cellulolytic activities (endoglucanases, cellobiohydrolases, and beta-glucosidases) as reviewed in Zhang et al., Outlook for cellulase improvement: Screening and selection strategies, 2006, Biotechnology Advances 24: 452-481. Total cellulolytic activity is usually measured using insoluble substrates, including Whatman No1 filter paper, microcrystalline cellulose, bacterial cellulose, algal cellulose, cotton, pretreated lignocellulose, etc. The most common total cellulolytic activity assay is the filter paper assay using Whatman No1 filter paper as the substrate. The assay was established by the International Union of Pure and Applied Chemistry (IUPAC) (chose, 1987, Measurement of cellulase activities, Pure Appl. Chem.59: 257-68). [0031] As used herein, the term “beta-glucosidase” means a beta-D-glucoside glucohydrolase (E.C. 3.2.1.21) that catalyzes the hydrolysis of terminal non-reducing beta-D-glucose residues with the release of beta-D-glucose. [0032] As used herein, the term “cellobiohydrolase” includes 1,4-p-D-glucan glucohydrolases (EC 3.2.1.74) as well as 1,4-p-D-glucan cellobiohydrolase (EC 3.2.1.91). Enzymes having activity classified according to EC 3.2.1.176, e.g., cellulose 1,4-beta-cellobiosidase (reducing end), may also be considered cellobiohydrolases herein. Cellobiohydrolases typically cleave cellulose strands to produce cellobiose. In some embodiments, the cellobiohydrolase is a cellobiohydrolase I (CBHI). In some embodiments, the cellobiohydrolase is a cellobiohydrolase II (CBHII).
IFF10001-WO-PCT [0033] As used herein, the term “endoglucanase” means an endo-1,4-(1,3;1,4)-beta-D-glucan 4- glucanohydrolase (EC 3.2.1.4) that catalyzes endohydrolysis of 1,4-beta-D-glycosidic linkages in cellulose, cellulose derivatives (such as carboxymethyl cellulose and hydroxyethyl cellulose), lichenin, beta-1,4 bonds in mixed beta-1,3 glucans such as cereal beta-D-glucans or xyloglucans, and other plant material containing cellulosic components. An endoglucanase may also refer to an enzyme classified according to EC 3.2.1.6. In some embodiments, the endoglucanase is an endoglucanase I (EGI). In some embodiments, the endoglucanase is an endoglucanase II (EGII). [0034] The term “hemicellulase” means enzymes that hydrolyze a hemicellulosic material. See, for example, Shallom, D. and Shoham, Y. Microbial hemicellulases. Current Opinion In Microbiology, 2003, 6(3): 219-228. Hemicellulases are key components in the degradation of plant biomass. Examples of hemicellulases include, but are not limited to, an acetylmannan esterase, an acetylxylan esterase, an arabinanase, an arabinofuranosidase, a coumaric acid esterase, a feruloyl esterase, a galactosidase, a glucuronidase, a glucuronoyl esterase, a mannanase, a mannosidase, a pectate lyase, a xylanase, and a xylosidase. The substrates of these enzymes, the hemicelluloses, are a heterogeneous group of branched and linear polysaccharides that are bound via hydrogen bonds to the cellulose microfibrils in the plant cell wall, crosslinking them into a robust network. Hemicelluloses are also covalently attached to lignin, forming together with cellulose a highly complex structure. The variable structure and organization of hemicelluloses require the concerted action of many enzymes for its complete degradation. The catalytic modules of hemicellulases are either glycoside hydrolases (GHs) that hydrolyze glycosidic bonds, or carbohydrate esterases (CEs), which hydrolyze ester linkages of acetate or ferulic acid side groups. These catalytic modules, based on homology of their primary sequence, can be assigned into GH and CE families. Some families, with an overall similar fold, can be further grouped into clans, marked alphabetically (e.g., GH-A). A most informative and updated classification of these and other carbohydrate active enzymes is available in the Carbohydrate- Active Enzymes (CAZy) database. Hemicellulolytic enzyme activities can be measured according to Ghose and Bisaria, 1987, Pure & Appl. Chem. 59: 1739-1752, at a suitable temperature, e.g., 50°C, 55°C, or 60°C, and pH, e.g., 5.0 or 5.5. [0035] As used herein, the term “protease” includes any enzyme belonging to the EC 3.4 enzyme group (including each of the eighteen subclasses thereof. As described herein, proteins (polypeptides) having protease activity (i.e., proteases), are also known in the art as peptidases, proteinases, peptide hydrolases, and proteolytic enzymes. [0036] As used herein, the term “protease activity” means proteolytic activity (EC 3.4). Protease activity can generally be measured using any assay, in which a substrate is employed, that includes
IFF10001-WO-PCT peptide bonds relevant for the specificity of the protease in question. Assays for pH and assays for temperature are likewise to be adapted to the protease in question. Examples of assay pH-values are pH 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 and examples of assay temperatures are 15°C, 20°C, 25°C, 30°C, 35°C, 37°C, 40°C, 45°C, 50°C, 55°C, 60°C, 65°C, 70°C, 80°C, 90°C, or 95°C. Examples of general protease substrates are casein, bovine serum albumin (BSA) and hemoglobin. [0037] As used herein, proteases may be of the “exo-type” (i.e., exopeptidases) that hydrolyze proteins (peptide bonds) starting at either N-terminal or C-terminal end of the protein chain, or of the “endo-type” (i.e., endopeptidases) that hydrolyze peptide bonds of the non-terminal ends of the protein chain (i.e., internal peptide bonds). [0038] As used herein, “fermentable sugar(s)” refers to saccharides that are capable of being metabolized under fermentation conditions. These fermentable sugars typically refer to glucose, maltose, and maltotriose. In some embodiments, sucrose, galactose, xylose, arabinose etc. may also be a fermentable sugar. Suitably, fermentable sugars may be obtained by the hydrolysis of starch and other polysaccharide compositions (e.g., feedstocks). [0039] As used herein, the term “feedstock” refers to a composition including at least one of the following: starch, cellulose, hemicellulose, lignocellulose, fermentable sugars, or a combination thereof. The feedstock may be a grain (e.g., a cereal, wheat, barley, rye, rice, triticale, millet, milo, sorghum, or corn), a tuber (e.g., potato or cassava), a root, a sugar (e.g., cane sugar, beet sugar, molasses, or a sugar syrup), stillage, wet cake, Distillers Dried Grain with Solubles (DDGS), cellulosic biomass, hemicellulosic biomass, a whey protein, soy based material, lignocellulosic biomass, or combinations thereof. As used herein, a “fraction of a feedstock” refers to any component of a feedstock that is separated out during the processing of the feedstock. [0040] The term “starch” refers herein to any material comprised of the complex polysaccharide carbohydrates of plants, comprised of amylose and amylopectin with the formula (C
6H
10O
5)
x wherein “x” can be any number. In particular, “starch” refers to any plant-based material including, but not limited to grains, cereals, grasses, tubers, and roots, and more particularly wheat, barley, corn, rye, rice, sorghum, brans, cassava, millet, potato, sweet potato, and tapioca. [0041] As used herein, a “starch-containing material” includes starch-containing materials derived from barley, beans, cassava, cereals, corn, milo, peas, potatoes, rice, rye, sago, sorghum, sweet potatoes, tapioca, wheat, and whole grains, or any mixture thereof. A starch-containing material may also be derived from cobs, sugar cane, sugar beets, triticale, millet, banana, or potatoes. In certain embodiments, a starch-containing material is a granular starch composition. In other embodiments, the starch-containing material is derived from whole grain. The starting material is generally selected based on the desired fermentation product. The starch-containing
IFF10001-WO-PCT material may also be a waxy or non-waxy type of corn and/or barley. In some embodiments, the starch-containing material is corn or derived from corn. In another embodiment the starch- containing material is wheat or derived from wheat. [0042] The phrase “granular starch” refers to uncooked (raw) starch, which has not been subject to gelatinization, where “starch gelatinization” means solubilization of starch molecules to form a viscous suspension. [0043] As used herein, “hydrolysis of starch” and the like refers to the cleavage of glycosidic bonds with the addition of water molecules. Thus, enzymes having “starch hydrolysis activity” catalyze the cleavage of glycosidic bonds with the addition of water molecules. [0044] In some embodiments, a starch-containing material may be subjected to one or more processing steps either before, during or after fermentation. As used herein, the phrase “one or more processing steps” includes, but is not limited to, milling, cooking, liquefaction, saccharification, fermentation, and simultaneous saccharification and fermentation (SSF). [0045] As used herein, the term “initial gelatinization temperature” means the lowest temperature at which starch gelatinization commences. [0046] As used herein, the term “milling” refers to any milling of a feedstock, including wet milling, dry grinding, or combinations thereof. Milling refers to a process which aids in breaking up the raw material used for the preparation of the feedstock into appropriately sized particles to facilitate downstream processing of the feedstock (e.g., to facilitate the cooking process). [0047] As used herein, “wet milling” is a process of milling that requires wet steeping of the feedstock (e.g., corn kernel) before processing. Wet steeping is then followed by a series of unit operations carried out in order to recover starch. The grain is typically soaked or “steeped” in water with dilute sulfurous acid for 24 to 48 hours prior to being subjected to a series of grinders. The downstream processes may include removal of oil (e.g., corn oil) followed by further stages to separate out fiber, protein (e.g., gluten) and starch components (e.g., such as the endosperm). This may be achieved by centrifugation, use of screens and hydroclonic separators. The starch and water remaining from this process may then be subjected to fermentation. [0048] As used herein, “dry grinding” refers to a process in which a starting material, such as a grain, is ground into a flour (e.g., meal) before further processing. Generally, the flour is then slurried with water to form a mash (slum) prior to being processed in downstream steps (e.g., saccharification). Ammonia is typically added to the mash and serves to both control the pH and provide a nutrient source to the fermenting organism used in fermentation. Thus, in some embodiments, dry grinding is used during processing of a feedstock (or a fraction thereof).
IFF10001-WO-PCT [0049] In some embodiments, a starch-containing material obtained after milling or dry grinding may be subjected to liquefaction and/or saccharification and/or fermentation and/or simultaneous saccharification and fermentation (SSF). This may be with or without a cooking step (e.g., after milling and before either liquefaction or saccharification). [0050] In some embodiments, the starch-containing material is subjected to cooking. Typically, the cooking process may take place post-milling. In some embodiments, the cooking process may take place at 90°C-120°C. In some embodiments, the cooking may be carried out prior to liquefaction and/or saccharification. In some embodiments, the cooking process may reduce bacteria levels prior to fermentation. In some embodiments, one or more enzymes may be added at this stage or thereafter. In some embodiments, alpha-amylase may be added following the cooking process (e.g., in a liquefaction process). [0051] In some embodiments, starch-containing material is not subjected to cooking. In such an embodiment, saccharification and fermentation, or SSF may be carried out on the feedstock (or fraction thereof) containing granular or raw starch (e.g., starch that has been treated at temperatures below gelatinization of the starch). [0052] As used herein, the term “liquefaction” refers to a process in which the starch is liquefied, usually by increasing the temperature. Liquefaction of the starch results in a significant increase in viscosity. For this reason, amylases may be introduced in order to reduce the viscosity. The temperature at which the starch liquefies varies depending upon the source of the starch. [0053] Starch processing can also be carried out at temperatures from about 25°C to just below the liquefaction temperature. These types of processes are often referred to as “granular starch hydrolysis,” “direct starch hydrolysis,” “raw starch hydrolysis,” “low temperature starch hydrolysis,” and the like. In some cases, the starch is pretreated at temperatures below the liquefaction temperatures in order to enhance enzymatic hydrolysis and/or other processes for treatment of starch. [0054] In some embodiments, liquefaction may be carried out at a lower temperature and/or a “cold cook process” that does not involve complete liquefaction of starch. [0055] The starch-containing material may also undergo saccharification. The saccharification may be separate to fermentation, or simultaneously therewith. Separate saccharification and fermentation is a process whereby starch present in a feedstock or a fraction thereof is converted to glucose and subsequently a fermenting microorganism converts the glucose into ethanol. [0056] As used herein, “simultaneous saccharification and fermentation” or “SSF” is a process whereby starch present in a starch-containing material is converted to glucose and, at the same
IFF10001-WO-PCT time and in the same reactor, a fermenting microorganism converts the glucose into ethanol. In some embodiments, saccharification may be carried out at low temperatures. [0057] As used herein, the terms “saccharifying enzyme” or “saccharifying enzymes” include, but are not limited to, alpha-amylases (EC 3.2.1.1), glucoamylases (EC 3.2.1.3), isoamylases (EC 3.2.1.68), beta-amylases (EC 3.2.1.2), pullulanases (EC 3.2.1.41), endoglucanases (EC 3.2.1.4), cellobiohydrolases (EC 3.2.1.91), beta-glucosidases (E.C. 3.2.1.21), cellulases (EC 3.2.1.74), lichenases (EC 3.1.1.73), lipases (EC 3.1.1.3), lipid acyltransferases (generally classified as EC 2.3.1.x), phospholipases (EC 3.1.1.4, E.C.3.1.1.32, or EC 3.1.1.5), phytases (e.g., 6-phytase (EC 3.1.3.26) or a 3-phytase (EC 3.1.3.8), xylanases (e.g., endo-l,4-β-d-xylanase (EC 3.2.1.8) or 1,4 beta-xylosidase (EC 3.2.1.37) or EC 3.2.1.32, EC 3.1.1.72, EC 3.1.1.73), glucoamylases (EC 3.2.1.3), hemicellulases (e.g., xylanases), or proteases (EC 3.4) , debranching enzymes, cutinases, esterases and/or mannanases (e.g., a beta-mannanase (EC 3.2.1.78)) transferases, glucosidases, and arabinofuranosidase. [0058] As used herein, the terms “admixing” or “adding” refer to the mixing of one or more ingredients and/or enzymes, where the one or more ingredients or enzymes are added in any order and in any combination. In some embodiments, admixing may relate to mixing one or more ingredients and/or enzymes simultaneously or sequentially. [0059] As used herein, “fermentation microorganism,” “fermenting organism,” “ethanol production host,” “ethanol producing organism,” “ethanol producing host,” “ethanol producing host cell,” “ethanologen,” and the like may be used interchangeably and include any microorganism that has the ability to ferment a fermentable sugar source to produce ethanol. [0060] In some embodiments, a fermentation microorganism is a yeast. In some embodiments, the yeast may be selected from the group consisting of Saccharomyces, Kluyveromyces, Zygosaccharomyces, Issatchenkia, Kazachstania and Torulaspora. In some embodiments, an ethanol producing organism may be a bacterium (e.g., Zymomonas, Escherichia, etc.). [0061] As used herein, a “lignocellulosic biomass” may contain cellulose, hemicellulose and the aromatic polymer lignin, hemicellulose, and cellulose (including insoluble arabinoxylans) by themselves are also potential energy sources, as they consist of C5- and C6-saccharides. [0062] In some embodiments, the lignocellulosic biomass may be any cellulosic, hemicellulosic, or lignocellulosic material, for example agricultural residues, bioenergy crops, industrial solid waste, municipal solid waste, sludge from paper manufacture, yard waste, wood waste, forestry waste, and combinations thereof. The lignocellulosic biomass may be selected from the group consisting of corn cobs, crop residues such as corn husks, corn gluten meal (CGM), corn stover, corn fiber, grasses, beet pulp, wheat straw, wheat chaff, oat straw, wheat middlings, wheat shorts,
IFF10001-WO-PCT rice bran, rice hulls, wheat bran, oat hulls, wet cake, DDG, DDGS, palm kernel, citrus pulp, cotton, lignin, barley straw, hay, rice straw, rice hulls, switchgrass, miscanthus, cord grass, reed canary grass, waste paper, sugar cane bagasse, sorghum bagasse, forage sorghum, sorghum stover, soybean stover, soy, components obtained from milling of trees, branches, roots, leaves, wood chips, sawdust, shrubs and bushes, vegetables, fruits, and flowers. [0063] Wet-cake, Distillers Dried Grains and Distillers Dried Grains with Solubles are fermentation byproducts obtained after the removal of ethanol by distillation from fermentation of a grain or a grain mixture by methods employed in the grain distilling industry. Stillage coming from the distillation (e.g., comprising water, remainings of the grain, yeast cells, etc.) is separated into a "solid” part and a "liquid” part. The solid part is called “wet-cake” and can be used as animal feed as such. The liquid part is (partially) evaporated into a syrup (solubles). The liquid part is often referred to as the thin stillage. When the wet-cake is dried it is Distillers Dried Grains (DDG). When the wet-cake is dried together with the syrup (solubles) it is Distillers Dried Grains with Solubles (DDGS). [0064] The terms “recovered,” “isolated,” “extracted,” and “separated,” refer to a compound, protein (polypeptide), cell, nucleic acid, amino acid, oil, or other specified material or component that is removed from at least one other material or component. In some embodiments, the at least one other material or component is at least one other material or component with which the compound, protein (polypeptide), cell, nucleic acid, amino acid, oil, or other specified material or component is naturally associated as found in nature. In some embodiments, the at least one other material or component is at least one other material or component with which the compound, protein (polypeptide), cell, nucleic acid, amino acid, oil, or other specified material or component is associated with under experimental or production conditions and/or systems. For example, an “isolated” polypeptide includes, but is not limited to, a polypeptide removed from a culture broth containing a heterologous host cell expressing the polypeptide. Recovery, for example, may refer to removal (extraction) of oil from a fermentation byproduct. [0065] The term “purified” refers to material (e.g., an isolated compound, polypeptide, polynucleotide, or other specified material or component) that is in a relatively pure state, e.g., at least about 90% pure, at least about 95% pure, at least about 98% pure, or at least about 99% pure. [0066] The term “enriched” refers to material (e.g., an isolated compound, polypeptide, polynucleotide, or other specified material or component) that is about 50% pure, at least about 60% pure, at least about 70% pure, or even at least about 80% pure. [0067] As used herein, "derived from" encompasses "originated from," "obtained from," or "isolated from."
IFF10001-WO-PCT [0068] As used herein, the terms “wild-type” and “native” are used interchangeably and refer to genes, proteins, strains, or other components found in nature, or that are not intentionally modified. [0069] The term “amino acid sequence” is synonymous with the terms “polypeptide,” “protein,” and “peptide,” and are used interchangeably. Where such amino acid sequences exhibit activity, they may be referred to as an “enzyme.” The conventional one-letter or three-letter codes for amino acid residues are used, with amino acid sequences being presented in the standard amino- to-carboxy terminal orientation (i.e., N→C). [0070] The term “nucleic acid” encompasses DNA, RNA, heteroduplexes, and synthetic molecules capable of encoding a polypeptide. Nucleic acids may be single stranded or double stranded and may contain chemical modifications. The terms “nucleic acid” and “polynucleotide” are used interchangeably. Because the genetic code is degenerate, more than one codon may be used to encode a particular amino acid, and the present compositions and methods encompass nucleotide sequences that encode a particular amino acid sequence. Unless otherwise indicated, nucleic acid sequences are presented in 5′-to-3′ orientation. [0071] “Hybridization” refers to the process by which one strand of nucleic acid forms a duplex with, i.e., base pairs with, a complementary strand, as occurs during blot hybridization techniques and PCR techniques. Stringent hybridization conditions are exemplified by hybridization under the following conditions: 65°C and 0.1X SSC (where 1X SSC = 0.15 M NaCl, 0.015 M Na3 citrate, pH 7.0). Hybridized, duplex nucleic acids are characterized by a melting temperature (Tm), where one half of the hybridized nucleic acids are unpaired with the complementary strand. Mismatched nucleotides within the duplex lower the Tm. [0072] The terms “transformed,” “stably transformed,” and “transgenic,” used with reference to a cell means that the cell contains a non-native (e.g., heterologous) nucleic acid sequence integrated into its genome or carried as an episome that is maintained through multiple generations. [0073] The term “introduced” in the context of inserting a nucleic acid sequence into a cell, encompasses, but is not limited to, “transfection”, “transformation” and “transduction,” as known in the art. Exemplary methods for introducing polynucleotides or polypeptides by transformation into a host cell, include, but are not limited to, microinjection, electroporation, stable transformation methods, transient transformation methods (such as induced competence using chemical (e.g. divalent cations such as CaCl2), mechanical (electroporation) means, or methods such as those described in published international applications WO 2018/114983 and WO 2010/149721, which are incorporated herein by reference in their entireties), ballistic particle
IFF10001-WO-PCT acceleration (particle bombardment), direct gene transfer, viral-mediated introduction, cell- penetrating peptides, or mesoporous silica nanoparticle (MSN)-mediated direct protein delivery. Introducing a nucleic acid, construct, plasmid, or vector into a host cell may be carried out by conjugation, which is a specific method of natural DNA exchange requiring physical cell-to-cell contact. Introducing a nucleic acid, construct, plasmid, or vector into a host cell may be carried out by transduction, which is the introduction of DNA via a virus (e.g., phage) infection which is also a natural method of DNA exchange. Generally, such methods involve incorporating a polynucleotide within a viral DNA or RNA molecule. [0074] A “host cell” is an organism into which an expression vector, phage, virus, or other nucleic acid sequence including a polynucleotide encoding a polypeptide of interest (e.g., an epimerase) has been introduced. Exemplary host cells are microorganism cells (e.g., bacteria, filamentous fungi, and yeast), mammalian cells, and plant cells capable of expressing the polypeptide of interest. The term “host cell” includes protoplasts created from cells. [0075] The term “heterologous” with reference to a polynucleotide or protein refers to a polynucleotide or protein that does not naturally occur in a host cell. [0076] The term “endogenous” with reference to a polynucleotide or protein refers to a polynucleotide or protein that occurs naturally in the host cell. [0077] Those of skill in the art are well aware of suitable methods for introducing polynucleotides into filamentous fungal cells (e.g., Aspergillus sp., Trichoderma sp., etc.), wherein standard techniques for transformation of filamentous fungi and culturing the fungi (which are well known to one skilled in the art) are used to transform a fungal host cell of the disclosure. Thus, the introduction of a DNA construct or vector into a fungal host cell includes techniques such as transformation, electroporation, nuclear microinjection, transduction, transfection (e.g., lipofection mediated and DEAE-Dextrin mediated transfection), incubation with calcium phosphate DNA precipitate, high velocity bombardment with DNA-coated microprojectiles, gene gun or biolistic transformation, protoplast fusion and the like. General transformation techniques are known in the art (see, e.g., Ausubel et al., 1987, Sambrook et al., 2001 and 2012, and Campbell et al., 1989). Also of use is the Agrobacterium-mediated transfection method such as the one described in U.S. Patent No. 6,255,115. The expression of heterologous proteins in Trichoderma has been described, for example, in U.S. Patent Nos. 6,022,725; 6,268,328; Harkki et al., 1991 and Harkki et al., 1989. Reference is also made to Cao et al. (2000), for transformation of Aspergillus strains. [0078] Transformation of Trichoderma sp. cells generally use protoplasts or cells that have been subjected to a permeability treatment, typically at a density of 10
5 to 10
7/mL, particularly
IFF10001-WO-PCT 2×10
6/mL. A volume of 100 μL of these protoplasts or cells in an appropriate solution (e.g., 1.2 M sorbitol and 50 mM CaCl2) is mixed with the desired DNA. Generally, a high concentration of polyethylene glycol (PEG) is added to the uptake solution. Additives, such as dimethyl sulfoxide, heparin, spermidine, potassium chloride and the like, may also be added to the uptake solution to facilitate transformation. Similar procedures are available for other fungal host cells. [0079] The term “expression” refers to the process by which a polypeptide is produced based on a nucleic acid sequence. The process includes both transcription and translation. [0080] As used herein, the terms “recombinant” or “non-natural” refer to an organism, microorganism, cell, nucleic acid molecule, vector, polypeptide and the like that has at least one engineered genetic alteration, or has been modified by the introduction of a heterologous nucleic acid molecule; or refer to a cell (e.g., a host cell) that has been altered such that the expression of a heterologous nucleic acid molecule or an endogenous nucleic acid molecule or gene can be controlled. Recombinant also refers to a cell that is derived from a non-natural cell or is progeny of a non-natural cell having one or more such modifications. Genetic alterations include, for example, modifications introducing expressible nucleic acid molecules encoding proteins, or other nucleic acid molecule additions, deletions, substitutions, or other functional alteration of a cell’s genetic material. For example, recombinant cells may express genes or other nucleic acid molecules (e.g., polynucleotide constructs) that are not found in identical or homologous form within a native (wild-type) cell or may provide an altered expression pattern of endogenous genes, such as being over-expressed, under-expressed, minimally expressed, or not expressed at all. “Recombination,” “recombining,” or generating a “recombined” nucleic acid is generally the assembly of two or more nucleic acid fragments wherein the assembly gives rise to a chimeric DNA sequence that would not otherwise be found in the genome. [0081] A “selective marker” or “selectable marker” refers to a gene capable of being expressed in a host to facilitate selection of host cells carrying the gene. Examples of selectable markers include but are not limited to antimicrobials (e.g., hygromycin, bleomycin, or chloramphenicol) and/or genes that confer a metabolic advantage, such as a nutritional advantage on the host cell. [0082] A “vector” refers to a polynucleotide sequence designed to introduce nucleic acids into one or more cell types. Vectors include cloning vectors, expression vectors, shuttle vectors, plasmids, phage particles, cassettes and the like. [0083] An “expression vector” refers to a DNA construct comprising a DNA sequence encoding a polypeptide of interest, which coding sequence is operably linked to a suitable control sequence capable of effecting expression of the DNA in a suitable host. Such control sequences may include a promoter to effect transcription, an optional operator sequence to control transcription, a
IFF10001-WO-PCT sequence encoding suitable ribosome binding sites on the mRNA, enhancers and sequences which control termination of transcription and translation. [0084] The term “operably linked” means that specified components are in a relationship (including but not limited to juxtaposition) permitting them to function in an intended manner. For example, a regulatory sequence is operably linked to a coding sequence such that expression of the coding sequence is under control of the regulatory sequences. [0085] A “signal sequence” is a sequence of amino acids attached to the N-terminal portion of a protein, which facilitates the secretion of the protein outside the cell. The mature form of an extracellular protein lacks the signal sequence, which is cleaved off during the secretion process. [0086] The term “specific activity” refers to the number of moles of substrate that can be converted to product by an enzyme or enzyme preparation per unit time under specific conditions. Specific activity is generally expressed as units (U)/mg of protein. [0087] “A cultured cell material” or similar language, refers to a cell lysate or supernatant (including media) that includes a protein of interest as a component. The cell material may be from a cell or host cell that is grown in culture for the purpose of producing the protein. In some embodiments, the viscosity reducing composition is a cultured cell material. For example, the cellulases, hemicellulases, and/or protease may be contained in a cultured cell material. [0088] As used herein, “clarified,” when used in reference to cultured cell material, e.g., a whole broth, means a cultured cell material which has been subjected to at least one clarification process to remove cell debris and/or other insoluble components. Clarification processes, as understood in the art include, but are not limited to, centrifugation techniques, cross-flow membrane filtration techniques, solid/liquid filtration techniques, and the like. [0089] “Percent sequence identity” means that a particular sequence has at least a certain percentage of amino acid residues or nucleotides identical to those in a specified reference sequence, when aligned using e.g., the CLUSTAL W algorithm with default parameters. See Thompson et al. (1994) Nucleic Acids Res.22:4673-4680. Default parameters for the CLUSTAL W algorithm are: Gap opening penalty: 10.0 Gap extension penalty: 0.05 Protein weight matrix: BLOSUM series DNA weight matrix: IUB Delay divergent sequences %: 40 Gap separation distance: 8 DNA transitions weight: 0.50
IFF10001-WO-PCT List hydrophilic residues: GPSNDQEKR Use negative matrix: OFF Toggle Residue specific penalties: ON Toggle hydrophilic penalties: ON Toggle end gap separation penalty OFF. [0090] Deletions are counted as non-identical residues, compared to a reference sequence. Deletions occurring at either terminus are included. [0091] The term “degree of polymerization” (DP) refers to the number (n) of anhydro- glucopyranose units in a given saccharide. Examples of DP1 are the monosaccharides glucose and fructose. Examples of DP2 are the disaccharides maltose and sucrose. The term “DE,” or “dextrose equivalent,” is defined as the percentage of reducing sugar, i.e., D-glucose, as a fraction of total carbohydrate in a syrup. [0092] The term “increased” as used herein can refer to a quantity or activity that is at least about 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11%, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, 20%, 50%, 100%, or 200% more than the quantity or activity for which the increased quantity or activity is being compared. The terms “increased,” “elevated,” “enhanced,” “greater than,” “improved,” and the like are used interchangeably herein. [0093] The term “decreased” as used herein can refer to a quantity or activity that is at least about 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11%, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, 20%, 50%, 100%, or 200% less than the quantity or activity for which the decreased quantity or activity is being compared. The terms “decreased,” “lowered,” “reduced,” “less than,” and the like are used interchangeably herein. [0094] Where a range of values is provided, it is understood that each intervening value, to the tenth of the unit of the lower limit unless the context clearly dictates otherwise, between the upper and lower limits of that range is also specifically disclosed. Each smaller range between any stated value or intervening value in a stated range and any other stated or intervening value in that stated range is encompassed within this disclosure. The upper and lower limits of these smaller ranges may independently be included or excluded in the range, and each range where either, neither or both limits are included in the smaller ranges is also encompassed within this disclosure, subject to any specifically excluded limit in the stated range. Where the stated range includes one or both of the limits, ranges excluding either or both of those included limits are also included in this disclosure. [0095] Numerical values and ranges may be presented herein with the numerical value being preceded by the term “about.” The term “about” is used herein to provide literal support for the
IFF10001-WO-PCT exact number that it precedes, as well as a number that is near to or approximately the number that the term precedes. In determining whether a number is near to or approximately a specifically recited number, the near or approximating unrecited number can be a number which, in the context in which it is presented, provides the substantial equivalent of the specifically recited number. For example, in connection with a numerical value, the term “about” refers to a range of -10% to +10% of the numerical value, unless the term is otherwise specifically defined in context. All values and ranges implicitly include the term “about” unless the context clearly dictates otherwise. I. METHODS AND COMPOSITIONS FOR REDUCING VISCOSITY OF FERMENTATION BYPRODUCTS [0096] Provided herein are methods for reducing the viscosity of syrup produced as a byproduct of a fermentation process using a viscosity reducing composition including one or more enzymes. In some embodiments, the fermentation process is an ethanol production process. Thus, in an aspect is provided a method for reducing syrup viscosity, including: (a) liquefying a starch- containing material with an alpha-amylase to produce a starch liquefact; (b) saccharifying the starch liquefact with a glucoamylase to produce a saccharification product; (c) fermenting the saccharification product using a fermenting organism to produce a fermentation product; (d) recovering the fermentation product to form a whole stillage; (e) separating the whole stillage into thin stillage and wet cake; and (f) concentrating the thin stillage to produce a syrup; where a viscosity reducing composition including a cellulase, a hemicellulase, a protease or any combination thereof, is added during step (f). In another aspect is provided a method for reducing syrup viscosity, including: (a) liquefying a starch-containing material with an alpha-amylase to produce a starch liquefact; (b) saccharifying the starch liquefact with a glucoamylase to produce a saccharification product; (c) fermenting the saccharification product using a fermenting organism to produce a fermentation product; (d) recovering the fermentation product to form a whole stillage; (e) separating the whole stillage into thin stillage and wet cake; and (f) concentrating the thin stillage to produce a syrup; where a viscosity reducing composition including a cellulase and/or a hemicellulase is added during step (f). In another aspect is provided a method for reducing syrup viscosity, including: (a) liquefying a starch-containing material with an alpha-amylase to produce a starch liquefact; (b) saccharifying the starch liquefact with a glucoamylase to produce a saccharification product; (c) fermenting the saccharification product using a fermenting organism to produce a fermentation product; (d) recovering the fermentation product to form a whole stillage; (e) separating the whole stillage into thin stillage and wet cake; and (f) concentrating the thin stillage to produce a syrup; where a viscosity reducing composition including a protease, is added during step (f). In another aspect is provided a method for reducing
IFF10001-WO-PCT syrup viscosity, including: (a) liquefying a starch-containing material with an alpha-amylase to produce a starch liquefact; (b) saccharifying the starch liquefact with a glucoamylase to produce a saccharification product; (c) fermenting the saccharification product using a fermenting organism to produce a fermentation product; (d) recovering the fermentation product to form a whole stillage; (e) separating the whole stillage into thin stillage and wet cake; and (f) concentrating the thin stillage to produce a syrup; where a viscosity reducing composition including an alpha-amylase, is added during step (f). [0097] As described above, processes for producing alcohols, e.g., ethanol, from starch- containing materials are generally well known. The “conventional process” includes liquefying gelatinized starch at high temperature typically using an alpha-amylase, e.g., a bacterial alpha- amylase, followed by simultaneous saccharification and fermentation (SSF) carried out in the presence of a glucoamylase and a fermenting microorganism. The “raw starch hydrolysis” process includes simultaneously saccharifying and fermenting granular starch below the initial gelatinization temperature, typically in the presence of an acid fungal alpha-amylase and a glucoamylase. The fermentation product is then recovered, e.g., by distillation, leaving a collection of liquids and solids commonly referred to as whole stillage. The whole stillage fermentation byproduct may be further processed to produce valuable co-products. In some embodiments, the methods for recovery include separating (dewatering) the whole stillage to form a thin stillage and wet cake. In some embodiments, the thin stillage is processed to produce a syrup. In some embodiments, oil is recovered from the syrup. In some embodiments, the syrup is added to DDG to produce DDGS. As described herein, viscosity reducing compositions may be added to the thin stillage, during the production of syrup from the thin stillage, and/or to the syrup to decrease syrup viscosity. A. Starch-Containing Material [0098] In some embodiments, the starch-containing material is or is derived from corn, cobs, sugar cane, sugar beets, wheat, barley, rye, triticale, milo, sago, millet, cassava, tapioca, sorghum, rice, peas, bean, banana, or potatoes. In some embodiments, the starch-containing material is or is derived from corn. In some embodiments, the starch-containing material is or is derived from wheat. [0099] Starch from a grain may be ground or whole and may include solids, such as corn kernels, bran and/or cobs. The starch may also be highly refined raw starch or feedstock from starch refinery processes. [00100] Native starch consists of microscopic granules, which are insoluble in water at room temperature. When an aqueous starch slurry is heated, the granules swell and eventually burst,
IFF10001-WO-PCT dispersing the starch molecules into the solution. At temperatures up to about 50°C to 75°C, the swelling may be reversible. However, with higher temperatures, an irreversible swelling called “gelatinization” begins. During “gelatinization” there is a dramatic increase in viscosity. [00101] Granular starch to be processed may be a highly refined starch quality (e.g., at least 90%, at least 95%, at least 97%, or at least 99.5% pure), or it may be a crude starch-containing material including (e.g., milled) whole grains including non-starch fractions such as germ residues and fibers. The raw material (e.g., feedstock), such as whole grains, may be reduced in particle size (e.g., by milling, in order to open up the structure and allowing for further processing). In dry milling, whole kernels are milled and used. Wet milling gives a good separation of germ and meal (starch granules and protein) and is often applied at locations where the starch hydrolysate is used in the production of, for example, syrups. Both dry and wet milling are well known in the art of starch processing and may be used in a process of the instant disclosure. Methods for reducing the particle size of the starch-containing material are well known to those skilled in the art. B. Liquefaction [00102] In some embodiments, the starch (e.g., of the starch-containing material) is thinned or “liquefied” so that it can be suitably processed. In some embodiments, liquefying a starch- containing material produces a starch liquefact. This reduction in viscosity is primarily attained by enzymatic degradation in current commercial practice. Liquefaction is generally carried out in the presence of an alpha-amylase (e.g., a bacterial alpha-amylase and/or acid fungal alpha- amylase). In some embodiments, liquefying a starch-containing material with an alpha-amylase produces a starch liquefact. In some embodiments, a phytase is also present during liquefaction. In some embodiments, enzymes such as a xylanase and/or beta-glucanase are also present during liquefaction. [00103] During liquefaction, the long-chained starch is degraded into branched and linear shorter units (maltodextrins) by an alpha-amylase. Liquefaction may be carried out as a three-step hot slurry process. The slurry is heated to between 60-95°C and an alpha-amylase is added to initiate liquefaction (thinning). The slurry may be jet-cooked at between 95-140°C (e.g., 105- 125°C, for about 1-15 minutes, e.g., about 3-10 minutes, especially about 5 minutes). The slurry is then cooled to 60-95 °C and more alpha-amylase is added to obtain final hydrolysis (secondary liquefaction). The jet-cooking process is carried out at pH 4.5-6.5, typically at a pH between 5 and 6. The alpha-amylase may be added as a single dose, for example, before jet cooking. The liquefaction process is carried out at between 70-95°C, such as 80-90°C, such as about 85°C, for about 10 minutes to 5 hours, typically for 1-2 hours. The pH is between 4 and 7, such as between 4.5 and 5.5. In order to ensure optimal enzyme stability under these conditions, calcium may be
IFF10001-WO-PCT added (to provide 1-60 ppm free calcium ions, such as about 40 ppm free calcium ions). After such treatment, the liquefied starch will typically have a “dextrose equivalent” (DE) of 10-15. Generally, such liquefaction and liquefaction conditions are well known in the art. 1. Enzymes Present and/or Added during Liquefaction [00104] Starch can be hydrolyzed into simpler carbohydrates by acids, various enzymes, or a combination thereof. For example, the primary enzymes used to hydrolyze starch into simpler carbohydrates are endo-amylases, exo-amylases, and debranching enzymes, which generally hydrolyze amylose and amylopectin. Amylose is hydrolyzed mainly by amylases, while amylopectin also requires debranching enzymes such as pullulanases (EC 3.2.1.41) for complete hydrolysis. [00105] An exemplary endo-amylase is an alpha-amylase (EC 3.2.1.1), specific for α-1,4- linkages of amylose and amylopectin. Exo-amylases have the ability to hydrolyze both α-1,4- linkages and α-1,6-linkages of amylose and amylopectin. An exemplary exo-amylase is amyloglucosidase (often referred to as glucoamylase; EC 3.2.1.20). Beta-amylase is an enzyme that has the ability to hydrolyze the α-1,4-linkages of amylose. Debranching enzymes such as pullulanases (EC 3.2.1.41) hydrolyze α-1,6-linkages in amylopectin, where hydrolysis products of debranching enzymes are mainly maltotriose and maltose. [00106] Alpha-amylases for use in liquefaction can be bacterial acid stable alpha-amylases, for example an alpha-amylase is from a Bacillus sp. such as Bacillus stearothermophilus or Bacillus lichenformis. In some embodiments, the alpha-amylase is Bacillus stearothermophilus alpha- amylase, or a variant thereof. [00107] In some embodiments, addition of an enzyme during liquefaction may facilitate an increase in co-product production. In some cases, adding an enzyme during liquefaction increases the amount of oil in a fermentation byproduct. For example, certain enzymes added during liquefaction may increase the amount and/or accessibility of oil in a fermentation byproduct. In some embodiments, adding an enzyme during liquefaction increases the amount of protein in a fermentation byproduct. For example, certain enzymes added during liquefaction may increase the amount and/or accessibility of protein in a fermentation byproduct. Thus, in some embodiments, the methods for reducing syrup viscosity include adding enzymes during liquefaction. In some embodiments, a cellulase, a hemicellulase, a protease, or any combination thereof may be added during liquefaction. In some embodiments, a cellulase is added during liquefaction. In some embodiments, the cellulase includes a cellobiohydrolase (EC 3.2.1.91 and/or EC 3.2.1.74), an endoglucanase (EC 3.2.1.4), beta-glucosidase (EC 3.2.1.21), or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase (EC 3.2.1.91, EC
IFF10001-WO-PCT 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), beta-glucosidase (EC 3.2.1.21), or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), or any combination thereof. In some embodiments, the cellulase is derived from Trichoderma reesei. In some embodiments, a hemicellulase is added during liquefaction. In some embodiments, the hemicellulase includes a xylanase. In some embodiments, the hemicellulase includes a GH10 xylanase. In some embodiments, the hemicellulase includes a xylanase having a sequence set forth by SEQ ID NO: 1, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 1. [00108] SEQ ID NO: 1: QAADSIDKLIKNKGKLYYGTITDPPLLGVAKDVAIIKADFGAVTPENSGKWDATEPSQG KFNFTSFDQVVNFAQQNGLYVRGHTLVWHGQLPQWVKNINDKATLTKVIENHVTQVV GRYKGKIYAWDVVNEIFEWDGTLRKDSHFNNVFGNDDYVGIAFRAARKADPNAKLYI NDYSLDSGSASKVTKGMVPSVKKWLSQGVPVDGIGSQTHLDPGQAGQIQGALTALANS GVKEVAITELDIRTAPANDYATVTKACLNVPKCIGITVWGVSDKNSWRKEHDSLLFDA NYNPKPAYYAVVNALR [00109] In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 1. [00110] In some embodiments, a protease is added during liquefaction. In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 2, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 2. [00111] SEQ ID NO: 2: MNRPSTRRTVRALLTAALAATALTAPAAPSLAQEASQEAALTRDLDLTGTEVALLRAA ESEAMDREEELSAVLGSDFGGVYLAPETGEVTVAVTDPAAVPVVEQSGATAQVVTFGE
IFF10001-WO-PCT TALNDFVDSLNAVADRADEQITGWYTDLAADTVVITAFPGGSAAAEELAALAGVDER AVRVTESAARPQLFADVIGGNPYYFGGYRCSIGFSVRKGSDTGFATAGHCGETGTLTRS PEGVVAGSYFPGRDMGWVRLTGADTVTPLVNRYDGGTVTVTGSQEAVTGSSVCRSGS TTGWRCGIIQSKNQTVRYAEGTVTGLTRTTACAEAGDSGGPWLTGSQAQGVTSGGSGN CRTGGITYFQPINPLLSYFGLELVTG [00112] In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 2. [00113] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 3, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 3. SEQ ID NO: 3 is the mature sequence of SEQ ID NO: 2. [00114] SEQ ID NO: 3 ADVIGGNPYYFGGYRCSIGFSVRKGSDTGFATAGHCGETGTLTRSPEGVVAGSYFPGRD MGWVRLTGADTVTPLVNRYDGGTVTVTGSQEAVTGSSVCRSGSTTGWRCGIIQSKNQT VRYAEGTVTGLTRTTACAEAGDSGGPWLTGSQAQGVTSGGSGNCRTGGITYFQPINPLL SYFGLELVTG [00115] In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 3. [00116] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 8, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 8. [00117] SEQ ID NO: 8
IFF10001-WO-PCT MQTFGAFLVSFLAASGLAAALPTEGQKTASVEVQYNKNYVPHGPTALFKAKRKYGAPI SDNLKSLVAARQAKQALAKRQTGSAPNHPSDSADSEYITSVSIGTPAQVLPLDFDTGSS DLWVFSSETPKSSATGHAIYTPSKSSTSKKVSGASWSISYGDGSSSSGDVYTDKVTIGGF SVNTQGVESATRVSTEFVQDTVISGLVGLAFDSGNQVRPHPQKTWFSNAASSLAEPLFT ADLRHGQNGSYNFGYIDTSVAKGPVAYTPVDNSQGFWEFTASGYSVGGGKLNRNSIDG IADTGTTLLLLDDNVVDAYYANVQSAQYDNQQEGVVFDCDEDLPSFSFGVGSSTITIPG DLLNLTPLEEGSSTCFGGLQSSSGIGINIFGDVALKAALVVFDLGNERLGWAQK [00118] In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 8. [00119] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 9, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 9. SEQ ID NO: 9 is the mature sequence of SEQ ID NO: 8. [00120] SEQ ID NO: 9 LPTEGQKTASVEVQYNKNYVPHGPTALFKAKRKYGAPISDNLKSLVAARQAKQALAK RQTGSAPNHPSDSADSEYITSVSIGTPAQVLPLDFDTGSSDLWVFSSETPKSSATGHAIYT PSKSSTSKKVSGASWSISYGDGSSSSGDVYTDKVTIGGFSVNTQGVESATRVSTEFVQDT VISGLVGLAFDSGNQVRPHPQKTWFSNAASSLAEPLFTADLRHGQNGSYNFGYIDTSVA KGPVAYTPVDNSQGFWEFTASGYSVGGGKLNRNSIDGIADTGTTLLLLDDNVVDAYYA NVQSAQYDNQQEGVVFDCDEDLPSFSFGVGSSTITIPGDLLNLTPLEEGSSTCFGGLQSSS GIGINIFGDVALKAALVVFDLGNERLGWAQK [00121] In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least
IFF10001-WO-PCT 95% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 9. [00122] In some embodiments, any combination of cellulase, hemicellulase, or protease described herein is added during liquefaction. C. Saccharification and Fermentation [00123] Saccharification allows the complex carbohydrates of the starch liquefact (thinned mash) to be converted to monosaccharides. The resulting sugars are then converted by yeasts to ethanol during the fermentation process step. Saccharification may be carried out using conditions well-known in the art with a saccharifying enzyme (e.g., a glucoamylase, a beta-amylase). For example, a complete saccharification step may last from about 24 hours to about 72 hours. In some embodiments, a pre-saccharification step of approximately 40-90 minutes at a temperature between 30-65°C (typically about 60°C) may be used, followed by complete saccharification during fermentation in a simultaneous saccharification and fermentation (SSF) process. Saccharification is generally performed at a temperature in the range of 20-75°C (e.g., 25-65°C and 40-70°C, typically about 60°C), and at a pH between about 4 and 5 (e.g., at about pH 4.5). [00124] The saccharification and fermentation steps may be carried out either sequentially or simultaneously. In some embodiments, saccharification and fermentation are performed simultaneously (SSF). However, it is also common to perform a pre-saccharification step for about 30 minutes to 2 hours (e.g., 30 to 90 minutes) at a temperature of 30 to 65°C, typically about 60°C which is followed by a complete saccharification during fermentation (i.e., simultaneous saccharification and fermentation; SSF). In a simultaneous saccharification and fermentation (SSF) process, there is no holding stage for saccharification, rather, the ethanologen and enzymes are added together, and the process is then carried out at a temperature of 25-40°C (such as between 28°C and 35°C, such as between 30°C and 34°C, such as about 32°C). The SSF process may be carried out at a pH from about 3 and 7, generally from pH 4.0 to 6.5, or pH 4.5 to 5.5. [00125] In some embodiments, ethanol is produced from starch-containing material without gelatinization (i.e., without cooking) of the starch-containing material (e.g., a "raw starch hydrolysis" process). For example, the ethanol fermentation product can be produced without liquefying the aqueous slurry containing the starch-containing material and water. Thus, in some embodiments, the process includes saccharifying a (e.g., milled) starch-containing material (e.g., granular starch) below the initial gelatinization temperature, in the presence of saccharifying enzymes (e.g., an alpha-amylase) to produce sugars that can be fermented into ethanol by a suitable fermenting organism.
IFF10001-WO-PCT [00126] In some embodiments, the saccharification and fermentation steps are performed simultaneously, where the saccharifying enzymes and fermenting organisms (e.g., a yeast strain) are added together and fermentation is carried out at a temperature of 25-40°C. The SSF process may be carried out at a pH from about 3 to 7 (e.g., pH 4.0 to 6.5, or pH 4.5 to 5.5). In some embodiments, fermentation is performed for about 6 to 120 hours. [00127] As generally defined above, the initial gelatinization temperature means the lowest temperature at which starch gelatinization commences. In general, starch heated in water begins to gelatinize between about 50°C and 75°C; the exact temperature of gelatinization depends on the specific starch and can readily be determined by the skilled artisan. Thus, the initial gelatinization temperature may vary according to the feedstock, to the particular variety of the feedstock as well as with the growth conditions. In some embodiments, a temperature below the initial gelatinization temperature means that the temperature typically lies in the range between 30-75°C, preferably between 45-60°C. For example, in some embodiments, the process is carried at a temperature from 25°C to 40°C, 28°C to 35°C, 30°C to 34°C, or about 32°C. [00128] Fermentation conditions are generally determined based on the type of starch- containing material used, the available fermentable sugars and/or the fermenting organism(s) used in the fermentation process. One skilled in the art can readily determine suitable fermentation conditions. The fermentation may be carried out at conventionally used conditions. In some embodiments, fermentation processes are anaerobic processes. [00129] For example, fermentations may be carried out at temperatures as high as 75°C (e.g., between 40-70°C or 50-60°C). However, bacteria with a significantly lower temperature optimum down to about room temperature (about 20°C) are also known. Examples of suitable fermenting organisms can be found in Section I-C-3 below. [00130] For ethanol production using yeast, the fermentation may go on for 24 to 96 hours. In some embodiments, the fermentation may go on for 35 to 60 hours. In some embodiments, the fermentation is carried out at a temperature between 20°C to 40°C. In some embodiments, the fermentation is carried out at a temperature between 26°C to 34°C. In some embodiments, the fermentation is carried out at a temperature of about 32°C. The fermentation may include, in addition to a fermenting microorganism (e.g., yeast), nutrients, and additional enzymes, including phytases. The use of yeast in fermentation is well known in the art. [00131] In some embodiments, fermentation is carried out at a pH in the range between 3.0 and 7.0 (e.g., pH 3.5 to 6, or pH 4 to 5). In some embodiments, fermentations are ongoing for 6-96 hours. The fermentation processes of the disclosure may be performed as a batch process or as a continuous process. Fermentations may be conducted in an ultrafiltration system wherein the
IFF10001-WO-PCT retentate is held under recirculation in the presence of solids, water, and the fermenting microorganism, and where the permeate is the desired fermentation product containing liquid. Likewise, contemplated are methods/processes conducted in continuous membrane reactors with ultrafiltration membranes and where the retentate is held under recirculation in presence of solids, water, and the fermenting organism(s) and where the permeate is the fermentation product containing liquid. After fermentation, the fermenting organism may be separated from the fermented slurry and recycled. [00132] Subsequent to fermentation or SSF, the fermentation product may be separated from the fermentation medium. The fermentation product, e.g., ethanol, can be recovered from the fermentation medium using any method known in the art including, but not limited to, chromatography, electrophoretic procedures, differential solubility, distillation, or extraction. For example, alcohol is separated from the fermented starch-containing material and purified by conventional methods of distillation. Thus, in some embodiments, the method includes distillation to obtain the fermentation product, e.g., ethanol. The fermentation and the distillation may be carried out simultaneously and/or separately/sequentially. In some embodiments, the distillation is followed by one or more process steps for further refinement of the fermentation product. [00133] In some embodiments, the fermentation product may be extracted from the fermentation medium by micro or membrane filtration techniques. Ethanol with a purity of up to about 96 vol. % can be obtained, which can be used as, for example, fuel ethanol, drinking ethanol, i.e., potable neutral spirits, or industrial ethanol. [00134] Following the completion of the distillation process, the remaining material (whole stillage) may be further processed to produce fermentation co-products. 1. Glucoamylases for Use in Saccharification and/or Fermentation Processes [00135] In some embodiments, a carbohydrate source generating enzyme present during saccharification is a glucoamylase. Thus, in some embodiments, a glucoamylase is present and/or added in a saccharification process and/or a fermentation process. In some embodiments, a glucoamylase is present and/or added in a simultaneous saccharification and fermentation (SSF) process. [00136] For example, in some embodiments, a glucoamylase present and/or added in a saccharification process and/or a fermentation process and/or in an SSF process is of fungal origin. In some embodiments, the glucoamylase is derived from a strain of Aspergillus (e.g., A. niger, A. awamori, A. oryzae). In some embodiments, the glucoamylase is derived from a strain of Trichoderma, e.g., T. reesei. In some embodiments, the glucoamylase is derived from
IFF10001-WO-PCT Talaromyces (e.g., T. emersonii), Trametes (e.g., T. cingulate), and the like. In some embodiments, the glucoamylase is a variant glucoamylase derived from native glucoamylase. [00137] Glucoamylases can be added to the saccharification and/or fermentation in an amount of 0.0001-20 AGU/g DS, such as, for example, 0.001-10 AGU/g DS, or between 0.01-5 AGU/g DS (e.g., 0.1-2 AGU/g DS or 0.1-0.5 AGU/g DS). [00138] Commercially available compositions comprising glucoamylase are generally known to one skilled in the art. Thus, in certain other embodiments, a glucoamylase is present and/or added in saccharification and/or fermentation in combination with an alpha-amylase, described below. 2. Alpha-Amylase Present and/or Added in Saccharification, Fermentation, or SSF [00139] In some embodiments, an alpha-amylase is present and/or added in a saccharification process and/or a fermentation process described herein. In some embodiments, the alpha-amylase is of a fungal origin or a bacterial origin. For example, in some embodiments, the alpha-amylase is a fungal acid stable alpha-amylase. A fungal acid stable alpha-amylase is an alpha-amylase that has activity in the pH range of 3.0 to 7.0 and preferably in the pH range from 3.5 to 6.5, including activity at a pH of about 4.0, 4.5, 5.0, 5.5, and 6.0. [00140] In some embodiments, an alpha-amylase of the disclosure is derived from the genus Aspergillus, such as A. terreus, A. niger, A. oryzae, A. awamori, and A. kawachi. In some embodiments, the alpha-amylase is derived from the genus Rhizomucor (e.g., R. pusillus), or the genus Meripilus (e.g., M. giganteus). [00141] In some embodiments, the ratio between glucoamylase and alpha-amylase present and/or added during saccharification and/or fermentation may be in the range from 500:1 to 1:1, 250:1 to 1:1, 100:1 to 1:1, 100:2 to 100:50, or 100:3 to 100:70. [00142] In some embodiments, a saccharifying enzyme may be used. In some embodiments, an enzyme preparation for use in saccharification may be used. In some embodiments, the enzyme preparation includes one or more saccharifying enzymes. In some embodiments, the enzyme preparation includes alpha-amylase (EC 3.2.1.1), glucoamylase (E.C. 3.2.1.3), isoamylase (EC 3.2.1.68), beta-amylase (EC 3.2.1.2), pullulanase (EC 3.2.1.41) and Aspergillopepsin 1 (EC 3.4.23.18). In some embodiments, the enzyme preparation includes one or more of an alpha- amylase (EC 3.2.1.1), a glucoamylase (EC 3.2.1.3), an isoamylase (EC 3.2.1.68), a beta-amylase (EC 3.2.1.2), a pullulanase (EC 3.2.1.41) and an Aspergillopepsin 1 (EC 3.4.23.18). 3. Fermenting Organisms [00143] In some embodiments, a fermenting organism may be a bacterium from one or more genus selected from the group consisting of Zymomonas, Arthrobacter, Bacillus, Clostridium,
IFF10001-WO-PCT Erwinia, Escherichia, Klebsiella, Lactobacillus, Pseudomonas, Streptomyces, and Thermoanaerobacter. In some embodiments, the bacterium is Zymomonas mobilis or Escherichia coli. [00144] In some embodiments, the fermenting organism may be a fungus. The fungus for use in accordance with the present invention may be any ascomycetous fungus (e.g., an Ascomycete). [00145] In some embodiments, the fermenting organism is a yeast. In some embodiments, the yeast may be selected from the group consisting of Saccharomyces, Kluyveromyces, Zygosaccharomyces, Issatchenkia, Kazachstania, and Torulaspora. In some embodiments, the yeast may be one or more selected from the group consisting of Saccharomyces cerevisiae, Saccharomyces bayanus, Saccharomyces carlsbergensis, Saccharomyces kudriavtsevii, Saccharomyces kudriavzevii, and Saccharomyces pasforianus. In some embodiments, the fermenting organism is a Saccharomyces cerevisiae. In some embodiments, the fermenting organism is a Saccharomyces cerevisiae var. diastaticus yeast. [00146] In some embodiments, the fermenting organism may be a non-naturally occurring microorganism. For example, in some embodiments, the fermenting organism is a yeast including one or more genetic modifications, where the one or more genetic modifications increase the amount and/or rate of ethanol production and/or reduce the amount of an undesirable fermentation product, such as glycerol and/or acetate. 4. Other Enzymes for use in Saccharification, Fermentation, or SSF [00147] In some embodiments, addition of an enzyme during saccharification, fermentation, or SSF may facilitate an increase in co-product production. In some cases, adding an enzyme during saccharification, fermentation, or SSF increases the amount of oil in a fermentation byproduct. For example, certain enzymes added during saccharification, fermentation, or SSF may increase the amount and/or accessibility of oil (co-product) in a fermentation byproduct. In some cases, adding an enzyme during saccharification, fermentation, or SSF increases the amount of protein in a fermentation byproduct. For example, certain enzymes added during saccharification, fermentation, or SSF may increase the amount and/or accessibility of protein in a fermentation byproduct. Thus, in some embodiments, the method for reducing syrup viscosity includes adding enzymes during saccharification, fermentation, or SSF. In some embodiments, the method for reducing syrup viscosity includes adding enzymes during saccharification. In some embodiments, the method for reducing syrup viscosity includes adding enzymes during fermentation. In some embodiments, the method for reducing syrup viscosity includes adding enzymes during saccharification and fermentation. In some embodiments, the method for reducing syrup viscosity includes adding enzymes during SSF.
IFF10001-WO-PCT [00148] In some embodiments, a cellulase, a hemicellulase, a protease, or any combination thereof may be added during saccharification, fermentation, or SSF. In some embodiments, a cellulase, a hemicellulase, a protease, or any combination thereof may be added during saccharification. In some embodiments, a cellulase, a hemicellulase, a protease, or any combination thereof may be added during fermentation. In some embodiments, a cellulase, a hemicellulase, a protease, or any combination thereof may be added during SSF. [00149] In some embodiments, a cellulase is added during saccharification, fermentation, or SSF. In some embodiments, a cellulase is added during saccharification and/or fermentation. In some embodiments, a cellulase is added during SSF. In some embodiments, a cellulase is present during saccharification, fermentation, or SSF. In some embodiments, a cellulase is present during saccharification and/or fermentation. In some embodiments, a cellulase is present during SSF. In some embodiments, the cellulase includes a cellobiohydrolase (EC 3.2.1.91 and/or EC 3.2.1.74), an endoglucanase (EC 3.2.1.4), beta-glucosidase (EC 3.2.1.21), or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), beta-glucosidase (EC 3.2.1.21), or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), or any combination thereof. In some embodiments, the cellulase is derived from Trichoderma reesei. [00150] In some embodiments, a hemicellulase is added during saccharification, fermentation, or SSF. In some embodiments, a hemicellulase is added during saccharification and/or fermentation. In some embodiments, a hemicellulase is added during SSF. In some embodiments, a hemicellulase is present during saccharification, fermentation, or SSF. In some embodiments, a hemicellulase is present during saccharification and/or fermentation. In some embodiments, a hemicellulase is present during SSF. In some embodiments, the hemicellulase includes a xylanase. In some embodiments, the hemicellulase includes a GH10 xylanase. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence set forth by SEQ ID NO: 1, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at
IFF10001-WO-PCT least 90% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 1. [00151] In some embodiments, a protease is added during saccharification, fermentation, or SSF. In some embodiments, a protease is added during saccharification and/or fermentation. In some embodiments, a protease is added during SSF. In some embodiments, a protease is present during saccharification, fermentation, or SSF. In some embodiments, a protease is present during saccharification and/or fermentation. In some embodiments, a protease is present during SSF. In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 2, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 3, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 3. [00152] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 8, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid
IFF10001-WO-PCT sequence with at least 60% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 9, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 9. [00153] In some embodiments, any combination of cellulase, hemicellulase, or protease described herein is added during saccharification and/or fermentation or SSF. In some embodiments, any combination of cellulase, hemicellulase, or protease described herein is present during saccharification and/or fermentation or SSF. [00154] Other enzymes for potential use during the saccharification and/or fermentation, or SSF include, but are not limited to, trehalase and pullulanase. In some embodiments, the use of enzymes during saccharification and/or fermentation, or SSF determines the enzymes of the viscosity reducing composition. For example, in some embodiments, when a composition including a cellulase, a hemicellulase, a protease, or any combination thereof that is added or present during saccharification and/or fermentation, or SSF, the same enzymes or enzyme classes may be contained in the viscosity reducing composition. D. Fermentation Byproducts [00155] The whole stillage fermentation byproduct remaining after removal of the fermentation product may be separated or partitioned into a solid and liquid phase by one or more methods for separating the thin stillage from the wet cake. Separating whole stillage into thin stillage and wet
IFF10001-WO-PCT cake to remove a significant portion of the liquid/water may be accomplished using any suitable separation technique, including, but not limited to, centrifugation, pressing, and filtration. In some embodiments, the separation (dewatering) is carried out by centrifugation. In some cases, industrial centrifuges may be used, such as, for example, decanter type centrifuges or high-speed decanter type centrifuges. In some embodiments, the separation (dewatering) is carried out using other conventional separation equipment such as a plate/frame filter presses, belt filter presses, screw presses, gravity thickeners and deckers, or similar equipment. Methods of separation are known in the art and may be incorporated in the methods described herein. [00156] Thin stillage typically contains 4-8 percent dry solids (DS) (mainly proteins, soluble fiber, fine fibers, and cell wall components) and has a temperature of about 60-90 degrees centigrade. The thin stillage stream may be condensed by evaporation to provide two process streams including: (i) an evaporator condensate stream including condensed water removed from the thin stillage during evaporation, and (ii) a syrup stream, including a more concentrated stream of the non-volatile dissolved and non-dissolved solids, such as non-fermentable sugars and oil, remaining in the thin stillage as the result of removing the evaporated water. [00157] The methods provided herein contemplate the addition of enzymes to the thin stillage, during the step of concentrating the thin stillage into a syrup, and/or into the syrup produced. In some embodiments, the enzymes added are a viscosity reducing composition including a cellulase, a hemicellulase, a protease, or any combination thereof. In some embodiments, the enzymes added are a viscosity reducing composition including a cellulase. In some embodiments, the enzymes added are a viscosity reducing composition including a hemicellulase. In some embodiments, the enzymes added are a viscosity reducing composition including a protease. In some embodiments, the enzymes added are a viscosity reducing composition including a cellulase and a hemicellulase. In some embodiments, the enzymes added are a viscosity reducing composition including a cellulase and a protease. In some embodiments, the enzymes added are a viscosity reducing composition including a hemicellulase and a protease. In some embodiments, the enzymes added are a viscosity reducing composition including an alpha-amylase. In some embodiments, the enzymes added are a viscosity reducing composition including one or more of a cellulase, a hemicellulase, a protease, or an alpha-amylase described herein. [00158] In some embodiments, the viscosity reducing composition includes a cellulase. In some embodiments, the cellulase includes a cellobiohydrolase (EC 3.2.1.91 and/or EC 3.2.1.74), an endoglucanase (EC 3.2.1.4), a beta-glucosidase (EC 3.2.1.21), or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), beta-glucosidase (EC 3.2.1.21),
IFF10001-WO-PCT or any combination thereof. In some embodiments, the cellulase includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), or any combination thereof. [00159] In some embodiments, the viscosity reducing composition includes an endoglucanase. In some embodiments, the endoglucanase has the amino acid sequence set forth by SEQ ID NO: 4, or an amino acid sequence having at least 50%, 60%, 70%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or more sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has an amino acid sequence having at least 60% sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has an amino acid sequence having at least 70% sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has an amino acid sequence having at least 80% sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has an amino acid sequence having at least 90% sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has an amino acid sequence having at least 95% sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has an amino acid sequence having at least 99% sequence identity to SEQ ID NO: 4. In some embodiments, the endoglucanase has the amino acid sequence set forth by SEQ ID NO: 4. [00160] SEQ ID NO: 4: MRSTTLLSGLAALAQLGTASYVLSDDYGNGMDFFSKWTFFTGSDPTNGYVTYVDQAT AQSAGLISADANNSVYIGVDHTNVASGSGRQSVRLTSINTYTHGLIILDLAHMPGSVCGS WPAFWTVGANWPNGGEIDIIEGVNQQTTDAMTLHTSDGCTINDSGFTGTLSTSNCYVD APGQSNNAGCGIDSTSSQSYGDGFNAIGGGVYATEWTSDYIKIWFFPRGSIPSDITSGSPD PSGWGTPAAAFQGNCDIDSHFTAQNIVFDITFCGDWAGNVWSSGSCASLASTCNAYVQ NNPSAFTETYWTINSLKVYEDNGSSAHNATLPATTSSRLRRNRRAVRSHQ [00161] In some embodiments, the endoglucanase has the amino acid sequence set forth by SEQ ID NO: 7, or an amino acid sequence having at least 50%, 60%, 70%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or more sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase has an amino acid sequence having at least 60% sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase has an amino acid sequence having at least 70% sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase has an amino acid sequence having at least 80% sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase has an amino acid sequence having at least 90% sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase has an amino acid sequence having at least 95% sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase
IFF10001-WO-PCT has an amino acid sequence having at least 99% sequence identity to SEQ ID NO: 7. In some embodiments, the endoglucanase has the amino acid sequence set forth by SEQ ID NO: 7. [00162] SEQ ID NO: 7: QQTVWGQCGGIGWSGPTNCAPGSACSTLNPYYAQCIPGATTITTSTRPPSGPTTTTRATS TSSSTPPTSSGVRFAGVNIAGFDFGCTTDGTCVTSKVYPPLKNFTGSNNYPDGIGQMQHF VNDDGMTIFRLPVGWQYLVNNNLGGNLDSTSISKYDQLVQGCLSLGAYCIVDIHNYAR WNGGIIGQGGPTNAQFTSLWSQLASKYASQSRVWFGIMNEPHDVNINTWAATVQEVV TAIRNAGATSQFISLPGNDWQSAGAFISDGSAAALSQVTNPDGSTTNLIFDVHKYLDSDN SGTHAECTTNNIDGAFSPLATWLRQNNRQAILTETGGGNVQSCIQDMCQQIQYLNQNS DVYLGYVGWGAGSFDSTYVLTETPTGSGNSWTDTSLVSSCLARK [00163] In some embodiments, the cellulase is derived from a fungus. In some embodiments, the cellulase is derived from a strain of Aspergillus. In some embodiments, the strain of Aspergillus is a strain of Aspergillus aurantiacus, Aspergillus niger, or Aspergillus oryzae. In some embodiments, the cellulase is derived from a strain of Chrysosporium. In some embodiments, the strain of Chrysosporium is a strain of Chrysosporium iucknowense. In an embodiment, the cellulase is derived from a strain of Humicola. In some embodiments, the strain of Humicola is a strain of Humicola insolens. In some embodiments, the cellulase is derived from a strain of Penicillium. In some embodiments, the strain of Penicillium is a strain of Penicillium emersonii or Penicillium oxalicum. In some embodiments, the cellulase is derived from a strain of Talaromyces. In some embodiments, the strain Talaromyces is a strain of Talaromyces aurantiacus or Talaromyces emersonii. In some embodiments, the cellulase is derived from a strain of Trichoderma. In some embodiments, the strain of Trichoderma is a strain of Trichoderma reesei. [00164] In some embodiments, the cellulase is derived from a strain of Trichoderma reesei. In some embodiments, the cellulase is a cultured cell material, e.g., a cultured cell material from Trichoderma reesei. [00165] In some embodiments, the fungus from which the cellulase is derived is a host cell. In some embodiments, the fungus is a recombinant fungus (e.g., host cell) that expresses, over- expresses, or does not express one or more cellulases. In some embodiments, the expressed or over-expressed cellulase is a recombinant cellulase. In some embodiments, the expressed or over- expressed cellulase is a heterologous cellulase. In some embodiments, the over-expressed cellulase is a native cellulase. In some embodiments, native, heterologous, and/or recombinant cellulase are derived from the fungus. [00166] In some embodiments, the cellulase is contained in a cultured cell material, e.g., a cultured cell material from a fungus. In some embodiments, the cellulase is contained in a cultured
IFF10001-WO-PCT cell material, e.g., a cultured cell material from a fungus where the fungus is a host cell (e.g., a recombinant fungus). In some embodiments, the cultured cell material is a whole broth. In some embodiments, the cultured cell material is clarified. [00167] In some embodiments, the fungus from which the cellulase is derived also expresses a hemicellulase. In some embodiments, the hemicellulase is a xylanase, an acetylmannan esterase, an acetylxylan esterase, an arabinanase, an arabinofuranosidase, a coumaric acid esterase, a feruloyl esterase, a galactosidase, a glucuronidase, a glucuronoyl esterase, a mannanase, a mannosidase, a pectate lyase, a xylosidase, or any combination thereof. In some embodiments, the hemicellulase includes a xylanase. In some embodiments, the hemicellulase includes an esterase. In some embodiments, the esterase is a feruloyl esterase (EC 3.1.1.73). In some embodiments, the esterase is an acetylxylan esterase (EC 3.1.1.72). In some embodiments, the hemicellulase includes an arabinofuranosidase (EC 3.2.1.55). In some embodiments, the hemicellulase is a native hemicellulase, e.g., a native xylanase, esterase, arabinofuranosidase. In some embodiments, the hemicellulase is a recombinant hemicellulase, e.g., a recombinant xylanase, esterase, arabinofuranosidase. In some embodiments, the fungus from which the cellulase is derived is a host cell that expresses, over-expresses, or does not express a hemicellulase. In some embodiments, the expressed or over-expressed hemicellulase is a recombinant hemicellulase. In some embodiments, the over-expressed hemicellulase is a native hemicellulase. [00168] In some cases, for example if the cellulase is a cultured cell material, the cellulase includes one or more hemicellulases. In some embodiments, the one or more hemicellulases include native and/or recombinant hemicellulases. [00169] In some embodiments, the viscosity reducing composition includes a hemicellulase. In some cases, the enzyme composition includes a hemicellulase that is not or is in addition to a hemicellulase that may be part of a cultured cell material including cellulases. [00170] In some embodiments, the hemicellulase includes a xylanase, an acetylmannan esterase, an acetylxylan esterase, an arabinanase, an arabinofuranosidase, a coumaric acid esterase, a feruloyl esterase, a galactosidase, a glucuronidase, a glucuronoyl esterase, a mannanase, a mannosidase, a pectate lyase, and a xylosidase or any combination thereof. In some embodiments, the hemicellulase includes a xylanase. In some embodiments, the hemicellulase includes an arabinofuranosidase (EC 3.2.1.55). In some embodiments, the hemicellulase includes an esterase. In some embodiments, the esterase is a feruloyl esterase (EC 3.1.1.73). In some embodiments, the esterase is an acetylxylan esterase (EC 3.1.1.72). In some embodiments, the hemicellulase is a native hemicellulase. In some embodiments, the hemicellulase is a recombinant hemicellulase, e.g., a recombinant xylanase, esterase, arabinofuranosidase.
IFF10001-WO-PCT [00171] In some embodiments, the hemicellulase is not natively or recombinantly expressed by a fungus from which the cellulases are derived. Thus, in some cases, the enzyme composition includes a hemicellulase that is not contained in a cultured cell material. In some embodiments, the enzyme composition includes a hemicellulase, as described herein, that is native to a fungus from which the cellulase is derived and a hemicellulase that is heterologous to the fungus from which the cellulases are derived. In some embodiments, the hemicellulase is a heterologous xylanase. In some embodiments, the hemicellulase is a recombinant xylanase. [00172] In some embodiments, the hemicellulase includes a GH10 xylanase. In some embodiments, the hemicellulase includes a xylanase having the amino acid sequence set forth by SEQ ID NO: 1, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulases includes a xylanase having an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 1. In some embodiments, the hemicellulase includes a xylanase having the amino acid sequence of SEQ ID NO: 1. [00173] In some embodiments, the viscosity reducing composition includes a cellulase and/or a GH10 xylanase. In some embodiments, the viscosity reducing composition includes a cellulase derived from a strain of Trichoderma reesei and a GH10 xylanase. In some embodiments, the viscosity reducing composition includes a cellulase and a xylanase having at least 60, 70, 80, 85, 90, 95, or 99% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellulase and a xylanase having at least 80, 85, 90, 95, or 99% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellulase and a xylanase having at least 90% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellulase and a xylanase having at least 95% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellulase and a xylanase having at least 99% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellulase and a
IFF10001-WO-PCT xylanase having the sequence set forth by SEQ ID NO: 1. In any of the foregoing, the cellulase of the viscosity reducing composition is derived from Trichoderma reesei. [00174] In some embodiments, the viscosity reducing composition includes one or more of a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), or a GH10 xylanase. In some embodiments, the viscosity reducing composition includes one or more of a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta- glucosidase (BGL), or a xylanase having at least 60, 70, 80, 85, 90, 95, or 99% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having at least 60% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having at least 70% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having at least 80% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having at least 85% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having at least 90% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having at least 95% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase (BGL), and a xylanase having the sequence set forth by SEQ ID NO: 1. In some embodiments, any of the viscosity reducing compositions described herein further include a protease. [00175] In some embodiments, the viscosity reducing composition includes one or more of a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), or a GH10 xylanase. In some embodiments,
IFF10001-WO-PCT the viscosity reducing composition includes one or more of a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta- glucosidase (EC 3.2.1.21), or a xylanase having at least 60, 70, 80, 85, 90, 95, or 99% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having at least 60% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having at least 70% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having at least 80% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having at least 85% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having at least 90% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having at least 95% sequence identity to SEQ ID NO: 1. In some embodiments, the viscosity reducing composition includes a cellobiohydrolase (EC 3.2.1.91, EC 3.2.1.74, and/or EC 3.2.1.176), an endoglucanase (EC 3.2.1.4 and/or EC 3.2.1.6), a beta-glucosidase (EC 3.2.1.21), and a xylanase having the sequence set forth by SEQ ID NO: 1. In some embodiments, any of the viscosity reducing compositions described herein further include a protease. [00176] In some embodiments, the viscosity reducing composition includes or further includes a protease. In some cases, any of the foregoing cellulase and hemicellulase combinations may further include a protease. In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 2, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 80%
IFF10001-WO-PCT sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 2. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 2. [00177] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 3, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 3. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 3. [00178] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 8, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 8. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 8. [00179] In some embodiments, the protease has an amino acid sequence set forth by SEQ ID NO: 9, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 80% sequence identity
IFF10001-WO-PCT to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 9. In some embodiments, the protease has the amino acid sequence of SEQ ID NO: 9. [00180] In some embodiments, the viscosity reducing composition includes or further includes an alpha-amylase. In some cases, any of the foregoing cellulase, hemicellulase, or protease combinations may further include an alpha-amylase. In some embodiments, the alpha-amylase has an amino acid sequence set forth by SEQ ID NO: 5, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 5. [00181] SEQ ID NO: 5 AAPFNGTMMQYFEWYLPDDGTLWTKVANEANNLSSLGITALWLPPAYKGTSRSDVGY GVYDLYDLGEFNQKGTVRTKYGTKAQYLQAIQAAHAAGMQVYADVVFDHKGGADG TEWVDAVEVNPSDRNQEISGTYQIQAWTKFDFPGRGNTYSSFKWRWYHFDGVDWDES RKLSRIYKFRGIGKAWDWEVDTENGNYDYLMYADLDMDHPEVVTELKNWGKWYVN TTNIDGFRLDAVKHIKFQFFPDWLSYVRSQTGKPLFTVGEYWSYDINKLHNYITKTNGT MSLFDAPLHNKFYTASKSGGAFDMRTLMTNTLMKDQPTLAVTFVDNHDTEPGQALQS WVDPWFKPLAYAFILTRQEGYPCVFYGDYYGIPQYNIPSLKSKIDPLLIARRDYAYGTQ HDYLDHSDIIGWTREGVTEKPGSGLAALITDGPGGSKWMYVGKQHAGKVFYDLTGNR SDTVTINSDGWGEFKVNGGSVSVWVPRKTT [00182] In some embodiments, the alpha-amylase has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 5. In some embodiments, the alpha-amylase has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 5. In some embodiments, the alpha-amylase has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 5. In some embodiments, the alpha-amylase has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 5. In some embodiments, the alpha-amylase has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 5. In some embodiments, the alpha-amylase has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 5. In some embodiments, the alpha-amylase has the amino acid sequence of SEQ ID NO: 5. [00183] In some embodiments, the alpha-amylase has an amino acid sequence set forth by SEQ ID NO: 6, or a sequence having at least 50, 60, 70, 80, 85, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99% or more sequence identity to SEQ ID NO: 6. [00184] SEQ ID NO: 6
IFF10001-WO-PCT AATNGTMMQYFEWYVPNDGQQWNRLRTDAPYLSSVGITAVWTPPAYKGTSQADVGY GPYDLYDLGEFNQKGTVRTKYGTKGELKSAVNTLHSNGIQVYGDVVMNHKAGADYT ENVTAVEVNPSNRYQETSGEYNIQAWTGFNFPGRGTTYSNWKWQWFHFDGTDWDQS RSLSRIFKFHGKAWDWPVSSENGNYDYLMYADYDYDHPDVVNEMKKWGVWYANEV GLDGYRLDAVKHIKFSFLKDWVDNARAATGKEMFTVGEYWQNDLGALNNYLAKVNY NQSLFDAPLHYNFYAASTGGGAYDMRNILNNTLVASNPTKAVTLVENHDTQPGQSLES TVQPWFKPLAYAFILTRSGGYPAVFYGDMYGTKGTTTYEIPALKSKIEPLLKARKDYAY GTQRDYIDNPDVIGWTREGDSTKAKSGLATVITDGPGGSKRMYVGTSNAGEIWYDLTG NRTDKITIGSDGYATFPVNGGSVSVWVQQ [00185] In some embodiments, the alpha-amylase has an amino acid sequence with at least 60% sequence identity to SEQ ID NO: 6. In some embodiments, the alpha-amylase has an amino acid sequence with at least 70% sequence identity to SEQ ID NO: 6. In some embodiments, the alpha-amylase has an amino acid sequence with at least 80% sequence identity to SEQ ID NO: 6. In some embodiments, the alpha-amylase has an amino acid sequence with at least 90% sequence identity to SEQ ID NO: 6. In some embodiments, the alpha-amylase has an amino acid sequence with at least 95% sequence identity to SEQ ID NO: 6. In some embodiments, the alpha-amylase has an amino acid sequence with at least 99% sequence identity to SEQ ID NO: 6. In some embodiments, the alpha-amylase has the amino acid sequence of SEQ ID NO: 6. [00186] In any of the foregoing, the cellulases of the viscosity reducing composition are derived from Trichoderma reesei. [00187] In some embodiments, the viscosity reducing composition and the compositions including a cellulase, a hemicellulase, a protease, or any combination thereof, include the same cellulases, hemicellulases, and/or proteases. In some embodiments, the viscosity reducing composition and the compositions including a cellulase, a hemicellulase, a protease, or any combination thereof, include different cellulases, hemicellulases, and/or proteases. In some embodiments, the viscosity reducing composition and the compositions including a cellulase, a hemicellulase, a protease, or any combination thereof, include the same and different cellulases, hemicellulases, and/or proteases. [00188] In some embodiments, the viscosity reducing composition is added to any one or more of the thin stillage, an intermediate product formed during concentrating the thin stillage to a produce a syrup, or the syrup produced. In some embodiments, the viscosity reducing composition is added to the thin stillage. In some embodiments, the viscosity reducing composition is added to an intermediate product formed during concentrating the thin stillage to a produce a syrup. In some embodiments, the viscosity reducing composition is added to the syrup.
IFF10001-WO-PCT [00189] The viscosity reducing composition as described in any of these embodiments may be incubated with the thin stillage, intermediate product, and/or syrup for a period of time sufficient to decrease the viscosity of the syrup. In some embodiments, the incubation period with the viscosity reducing composition is at least 5 minutes, at least 10 minutes, at least 15 minutes, at least 20 minutes at least 30 minutes, at least 45 minutes, at least 1 hour, at least 2 hours, at least 3 hours, at least 4 hours, at least 5 hours, at least 6 hours, at least 8 hours, at least 10 hours, at least 12 hours, at least 14 hours, at least 15 hours, at least 16 hours, at least 18 hours, at least 20 hours, at least 24 hours, at least 30 hours, at least 36 hours, at least 40 hours, at least 41 hours, at least 42 hours, at least 43 hours, at least 44 hours, at least 45 hours, at least 46 hours, at least 47 hours, at least 48 hours, at least 50 hours, at least 55 hours, at least 60 hours, at least 62 hours, at least 64 hours, at least 68 hours, at least 70 hours, or at least 72 hours. In some embodiments, the incubation period is 48 hours. In some embodiments, the incubation period is at least or about 30 minutes. In some embodiments, the incubation period is in the range of 30 minutes to 6 hours. In some embodiments, the incubation period is in the range of 1 to 6 hours. In some embodiments, the incubation period is in the range of 2 to 6 hours. In some embodiments, the incubation period is in the range of 3 to 6 hours. In some embodiments, the incubation period is in the range of 4 to 6 hours. In some embodiments, the incubation period is in the range of 5 to 6 hours. Those skilled in the art will appreciate that to achieve longer incubation periods, containers, such as holding tanks, may be used for the desired time period. [00190] In some embodiments, the conditions (e.g., temperature, pH, viscosity reducing composition dosing, etc.) may be adjusted to further optimize the reduction in syrup viscosity. For example, the temperature may be adjusted in a holding tank to a set point or within a range that is optimal for the enzymes of the viscosity reducing composition. In some embodiments, the temperature range may be from 20°C to 90°C. In some embodiments, the temperature range may be from 30°C to 85°C. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.00001% weight (wt) enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.0001% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.001% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.005% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.01% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.05% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of at least about 0.1% wt enzyme/wt. In some embodiments, the viscosity
IFF10001-WO-PCT reducing composition is dosed at a concentration of at least about 0.5% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.00001% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.0001% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.001% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.005% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.01% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.05% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.1% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.25% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.5% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the viscosity reducing composition is dosed at a concentration of about 0.75% wt enzyme/wt to about 1% wt enzyme/wt. In some embodiments, the enzyme wt/wt is enzyme wt/wt syrup. In some embodiments, the enzyme wt/wt is enzyme wt/wt thin stillage. In some embodiments, the enzyme wt/wt is enzyme wt/wt intermediate product formed during concentrating the thin stillage to a produce a syrup. In some embodiments, the enzyme wt is the weight of the viscosity reducing composition. [00191] The pH of the incubation may range from about 3.0 to about 7. In some embodiments, the pH is in a range of about 3.0 to 6.0. In some embodiments, the pH is in a range of about 3.5 to 6.0. In some embodiments, the pH is in a range of about 3.5 to 5. In an embodiment, the pH is about 3.0. In some embodiments, the pH is about 3.1. In some embodiments, the pH is about 3.2. In some embodiments, the pH is about 3.3. In some embodiments, the pH is about 3.4. In some embodiments, the pH is about 3.5. In some embodiments, the pH is about 3.6. In some embodiments, the pH is about 3.7. In some embodiments, the pH is about 3.8. In some embodiments, the pH is about 3.9. In some embodiments, the pH is about 4.0. In some embodiments, the pH is about 4.1. In some embodiments, the pH is about 4.2. In some embodiments, the pH is about 4.3. In some embodiments, the pH is about 4.4. In some embodiments, the pH is about 4.5. In some embodiments, the pH is about 4.6. In some embodiments, the pH is about 4.7. In some embodiments, the pH is about 4.8. In some embodiments, the pH is about 4.9. In some embodiments, the pH is about 5.0. In some
IFF10001-WO-PCT embodiments, the pH is about 5.1. In some embodiments, the pH is about 5.2. In some embodiments, the pH is about 5.3. In some embodiments, the pH is about 5.4. In some embodiments, the pH is about 5.5. In some embodiments, the pH is about 5.6. In some embodiments, the pH is about 5.7. In some embodiments, the pH is about 5.8. In some embodiments, the pH is about 5.9. In some embodiments, the pH is about 6.0. In some embodiments, the pH is about 6.1. In some embodiments, the pH is about 6.2. In some embodiments, the pH is about 6.3. In some embodiments, the pH is about 6.4. In some embodiments, the pH is about 6.5. In some embodiments, the pH is about 6.6. In some embodiments, the pH is about 6.7. In some embodiments, the pH is about 6.8. In some embodiments, the pH is about 6.9. In some embodiments, the pH is about 7.0. [00192] In some embodiments, the viscosity of the syrup is less than the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the decrease in syrup viscosity is at least 1, 2, 3, 4, 5, 10, 20, 30, 40, 50% or more compared to the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the decrease in syrup viscosity is at least 2% or more compared to the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the decrease in syrup viscosity is at least 5% or more compared to the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the decrease in syrup viscosity is at least 10% or more compared to the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the decrease in syrup viscosity is at least 15% or more compared to the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the decrease in syrup viscosity is at least 20% or more compared to the viscosity of a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, oil is recovered from the syrup. In some embodiments, an increased amount of oil is recovered from the syrup compared to a syrup not treated with a viscosity reducing composition as described herein. In some embodiments, the increase in oil recovered is at least 1, 2, 3, 4, 5, 10, 20% or more compared to the oil recovered from a syrup not treated with a viscosity reducing composition as described herein. It will be appreciated that all comparisons are made between syrups produced under identical conditions with the only difference being the presence or absence of the viscosity reducing composition. [00193] Between 5-90 vol-%, such as between 10-80%, such as between 15-70%, such as between 20-60% of thin stillage (e.g., optionally hydrolyzed) may be recycled (as backset) to step
IFF10001-WO-PCT (a). The recycled thin stillage (backset) may constitute from about 1-70 vol.%, preferably 15-60% vol.%, especially from about 30 to 50 vol.% of the slurry formed in step (a). [00194] In some embodiments, the process further comprises recycling at least a portion of the thin stillage stream to the slurry, optionally after oil has been extracted from the thin stillage stream. EXEMPLARY EMBODIMENTS [00195] Among the provided embodiments are: 1. A method for reducing syrup viscosity, comprising: (a) liquefying a starch-containing material with an alpha-amylase to produce a starch liquefact; (b) saccharifying the starch liquefact with a glucoamylase to produce a saccharification product; (c) fermenting the saccharification product using a fermenting organism to produce a fermentation product; (d) recovering the fermentation product to produce a whole stillage; (e) separating the whole stillage into thin stillage and wet cake; and (f) concentrating the thin stillage to produce a syrup; wherein a viscosity reducing composition comprising a cellulase, a hemicellulase, a protease, or any combination thereof, is added during step (f). 2. The method of embodiment 1, wherein the viscosity reducing composition is added to the thin stillage, to an intermediate product formed during concentrating the thin stillage to a produce a syrup, and/or to the syrup produced in step (f). 3. The method of embodiment 1 or embodiment 2, wherein step (b) and step (c) are combined as a simultaneous saccharification and fermentation step (SSF). 4. The method of any one of embodiments 1-3, wherein the cellulase comprises a cellobiohydrolase, an endoglucanase, a beta-glucosidase, or any combination thereof. 5. The method of any one of embodiments 1-4, wherein the cellulase comprises a cellobiohydrolase I (CBHI), a cellobiohydrolase II (CBHII), an endoglucanase I (EGI), an endoglucanase II (EGII), a beta-glucosidase BGL, or any combination thereof. 6. The method of any one of embodiments 1-5, wherein the cellulase is derived from a fungus. 7. The method of any one of embodiments 1-6, wherein the cellulase is derived from a strain of Trichoderma, optionally Trichoderma reesei. 8. The method of any one of embodiments 1-7, wherein the hemicellulase comprises a xylanase. 9. The method of any one of embodiments 1-8, wherein the hemicellulase comprises a GH10 xylanase. 10. The method of any one of embodiments 1-9, wherein the hemicellulase comprises a sequence having at least 60% sequence identity to the sequence set forth by SEQ ID NO: 1.
IFF10001-WO-PCT 11. The method of any one of embodiments 1-10, wherein the protease comprises a sequence having at least 60% sequence identity to the sequence set forth by SEQ ID NO: 3. 12. The method of any one of embodiments 1-11, wherein: (i) a composition comprising a cellulase, a hemicellulase, a protease, or any combination thereof is added during step (a), step (b), and/or step (c); or (ii) wherein step (b) and step (c) are combined as a simultaneous saccharification and fermentation step (SSF) and a composition comprising a cellulase, a hemicellulase, a protease, or any combination thereof is added during step (a) and/or the SSF. 13. The method of embodiment 12, wherein the composition comprising a cellulase, a hemicellulase, a protease, or any combination thereof comprises the viscosity reducing composition. 14. The method of any one of embodiments 1-13, wherein the viscosity of the syrup is decreased compared to a syrup not treated with the viscosity reducing composition. 15. The method of any one of embodiments 1-14, wherein the starch-containing material is corn. II. EXAMPLES [00196] The following examples are included for illustrative purposes only and are not intended to limit the scope of the invention. Example 1: Screening enzyme classes on syrup for flowability improvement [00197] High and low viscosity syrups were collected from a commercial dry grind ethanol plant and frozen, prior to testing in a laboratory-scale experiment. The high viscosity syrup (termed “Viscous Syrup”) was collected from the plant during a cellulase and xylanase SSF trial, during which an engineered Trichoderma whole cellulase and a thermostable xylanase were added into SSF. The low viscosity syrup (termed “Baseline Syrup”) was collected prior to the cellulase and xylanase SSF trial. [00198] The frozen syrups were thawed and incubated at 70°C for about 3 hours to pasteurize the syrups and allow for the heated syrups to be transferred to reaction vessels. Four grams of heated Viscous Syrup were weighed into 20 mL glass GC-MS vials. To each vial, enzyme was added at a dose of 0.5% volume of enzyme per weight of syrup. Table E1 shows the 15 enzyme compositions tested. The enzymes tested included cellulases, xylanase, proteases as well as amylases, as also described in Table E1. The Engineered Trichoderma Whole Cellulase 1, Trichoderma Whole Cellulase, Engineered Trichoderma Whole Cellulase 2, Engineered Trichoderma Whole Cellulase 3, Whole Cellulase, and Engineered Trichoderma Whole Cellulase 4 all included cellobiohydrolase (CBHI and CBHII), endoglucanase (EGI and EGII), and beta- glucosidase. No-enzyme control vials were also prepared for the Viscous and Baseline Syrups,
IFF10001-WO-PCT with only water added to the syrups, instead of enzyme. Vials were placed into a forced air incubator set to 65°C, shaking at 200 rpm, for 2 hours. [00199] Following the incubation, vials were inverted and placed upon a slanted tray that was at a 50-degree angle from the tabletop surface. The flowability of each syrup was evaluated by the speed at which it slid down the vial. The viscosity of the resulting samples was described by five different categories, including Low, Low-Moderate, Moderate, Moderate-High, or High. Samples defined by the “Low” viscosity category flowed fastest, whereas samples defined by the “High” viscosity category did not flow at all. It is assumed that flowability is inversely related to viscosity, such that enzymes that rank high in flowability correspond to low viscosity. Tregger et al., 2008. [00200] Table E1 shows the resulting viscosity for the 15 enzymes tested, as well as the Viscous and Baseline syrups without enzyme added. The enzymes that improved syrup viscosity the most (Low-Moderate category) were Trichoderma Whole Cellulase, Engineered Trichoderma Whole Cellulase 1, Engineered Trichoderma Whole Cellulase 2, and Thermostable Xylanase. Following this category, three additional enzymes provided some benefit and resulted in syrups in the Moderate category, including Engineered Whole Cellulase 3, Xylanase-Amylase-Protease Blend, and Thermostable Protease. All other enzymes tested had either no or very little impact on syrup viscosity. Table E1. Samples tested for syrup flowability. Shown below is a description of each sample, as well as the observed syrup flowability following incubation. Conditions were ranked into

Example 2: Enzyme treatment to improve syrup flowability [00201] High and low viscosity syrups were collected and frozen as described in Example 1.
IFF10001-WO-PCT [00202] The frozen syrups were thawed and incubated at 70°C for around 3 hours to pasteurize the syrups and allow for the heated syrups to be transferred to reaction vessels.4.0 grams of heated Viscous Syrup were weighed into 15 mL polypropylene tubes. To each tube, enzyme was added at a dose of 0.1% volume of enzyme per weight of syrup. Six enzyme products were tested, including Trichoderma Whole Cellulase, Engineered Trichoderma Whole Cellulase 1, Engineered Trichoderma Whole Cellulase 2, Thermostable Xylanase (SEQ ID NO: 1), all of which contain cellulase and hemicellulase activities, and a Thermostable Protease (SEQ ID NO: 3). No-enzyme control tubes were also prepared for the Viscous and Baseline Syrups, with only water added to the syrups, instead of enzyme. Tubes were placed into a forced air incubator set to 80°C, shaking at 200 rpm, for 2 hours. [00203] Following the incubation, blinded tubes were inverted and placed upon a slanted tray that was at a 50-degree angle from the tabletop surface. The flowability of each syrup was evaluated by ranking the speed at which it slid down the tube. The conditions were ranked from 1 to 8, with 1 being the fastest and 8 being the slowest. After completing the ranking, the tubes were turned back to the proper orientation and mildly centrifuged at approximately 400 x g for 30 seconds to allow the syrup to re-collect at the bottom of each tube. Following this, the tubes were inverted and ranked again. This process was repeated an additional two times, for a total of four rankings. [00204] FIG.1 shows the average speed rankings and corresponding standard deviations of the four observations. It is assumed that flowability is inversely related to viscosity, such that enzymes that rank high in flowability correspond to low viscosity. Tregger et al., 2008. All enzymes tested resulted in an improvement to the syrup flow speed, with Trichoderma Whole Cellulase resulting in the best overall improvement to the Viscous Syrup, followed by Engineered Trichoderma Whole Cellulase 1, Engineered Trichoderma Whole Cellulase 2, Thermostable Xylanase, and finally Thermostable Protease. Example 3: Enzyme treatment to reduce syrup viscosity [00205] High and low viscosity syrups were collected and frozen as described in Example 1. [00206] Frozen syrups were thawed and incubated at 70°C for approximately 3 hours to pasteurize the syrups and allow for the heated syrups to be transferred to reaction vessels. Thirty- five grams of pasteurized Viscous or Baseline Syrup were weighed into 50 mL polypropylene conical screw-cap tubes. To each tube, Thermostable Protease (SEQ ID NO: 3), Trichoderma Whole Cellulase, or a blend of both products were added to each of the syrups, as listed in Table E2. The enzymes tested include a protease and/or cellulase composition, as described in Table E1. No-enzyme controls were also prepared for the Viscous and Baseline Syrups. Tubes were
IFF10001-WO-PCT incubated in a water bath at either 80°C for 2 hours (for Viscous Syrup) or 88°C for 0.7 hours (for Baseline Syrup) with orbital shaking at 100 rpm. Following incubation, viscosity was measured on a Brookfield LVDVE Viscometer. Percent viscosity reduction over the no-enzyme control, by syrup type, was calculated for each sample, and listed in Table E2. Table E2. Enzyme treatments to reduce syrup viscosities. Enzymes were added to Viscous or Baseline Syrups and incubated prior to viscosity measurement and calculation of r nt r d ti n v r th n nz m ntr l p ase, and a

blend of both products reduce syrup viscosity. Example 4: Viscosity reduction in syrup from corn [00208] Syrups from two commercial corn ethanol plants were obtained. The syrups were evaporated thin stillage at the dry solid (DS) content that was obtained at the plant. Syrup 1 had a DS of 42% and Syrup 2 had a DS of 27%. [00209] The effect of different enzyme activities on the viscosity of the syrups was measured using a Rapid Visco Analyzer (RVA 4500) (PerkinElmer, Waltham, MA). A sample of 25 grams of the syrup was placed in an aluminum RVA cup. Enzymes, single or in combination, were added to the RVA cup in the amount indicated in Table E3. Enzymes were added after 4 minutes to ensure they were added when the temperature of the syrup had reached 60
oC. The RVA cup was placed in the RVA 4500 and a temperature program was started. Temperature in RVA was kept at 60
oC for 30 minutes before temperature was dropped to 30
oC. Temperature was kept at 30
oC for 10 minutes. RVA rotation (using standard spindle) was set at 80 rpm. The average viscosity of the last 5 minutes was recorded. A blank run, without enzyme addition, was used as control. Viscosities of the enzyme treated runs were compared to the blank and the relative viscosity (%) was calculated as viscosity of the enzyme treated run divided by the viscosity of the blank run. Viscosity reduction was calculated as 100 – relative viscosity in percentages. [00210] The enzymes tested are shown in Table E3 below. Enzymes were dosed as kg enzyme product per MT (metric ton) of dry solids in the syrup (kg/MT DS). [00211] The results shown in Table E3 demonstrate the substantial impact of enzymes on syrup viscosity. Although the effects varied by syrup, enzymes that consistently showed viscosity reduction included cellulase (e.g., endoglucanase), xylanase, protease, and alpha-amylase.
IFF10001-WO-PCT Enzyme blends, for example, xylanase, endoglucanase, and protease, also provided viscosity reduction. Table E3: Viscosity reduction by syrup and enzyme treatment Syrup Enzyme Treatment Enzyme dose Viscosity

IFF10001-WO-PCT Thermostable Protease (SEQ ID NO: 3); Endoglucanase (EC 3.2.1.4; SEQ ID NO: 7)
[00212] Syrups from commercial wheat ethanol plants were obtained. The syrup was evaporated thin stillage at the DS that was obtained at the plant. Syrup 1 had a DS of 37.6%, Syrup 2 had a DS of 41%, and Syrup 3 had a DS of 32.1%. Syrup 3 was obtained from a blend of mainly wheat with barley, rye, and triticale included. Viscosity reduction by enzymes was tested as described in Example 4 above. [00213] The results shown in Table E4 demonstrate that the enzymes had a substantial impact on syrups from wheat. Although the effects varied by syrup, enzymes that consistently showed good viscosity reduction on syrups from wheat included cellulase (e.g., endoglucanase), xylanase, protease, and alpha-amylase. Blends of enzymes also performed well in reducing syrup viscosity. Table E4: Viscosity reduction by syrup and enzyme treatment Syrup Enzyme Treatment Enzyme dose Viscosity
IFF10001-WO-PCT Trichoderma Whole Cellulase (cellobiohydrolase, 0.3;0.4 15% endoglucanase, and beta-glucosidase);
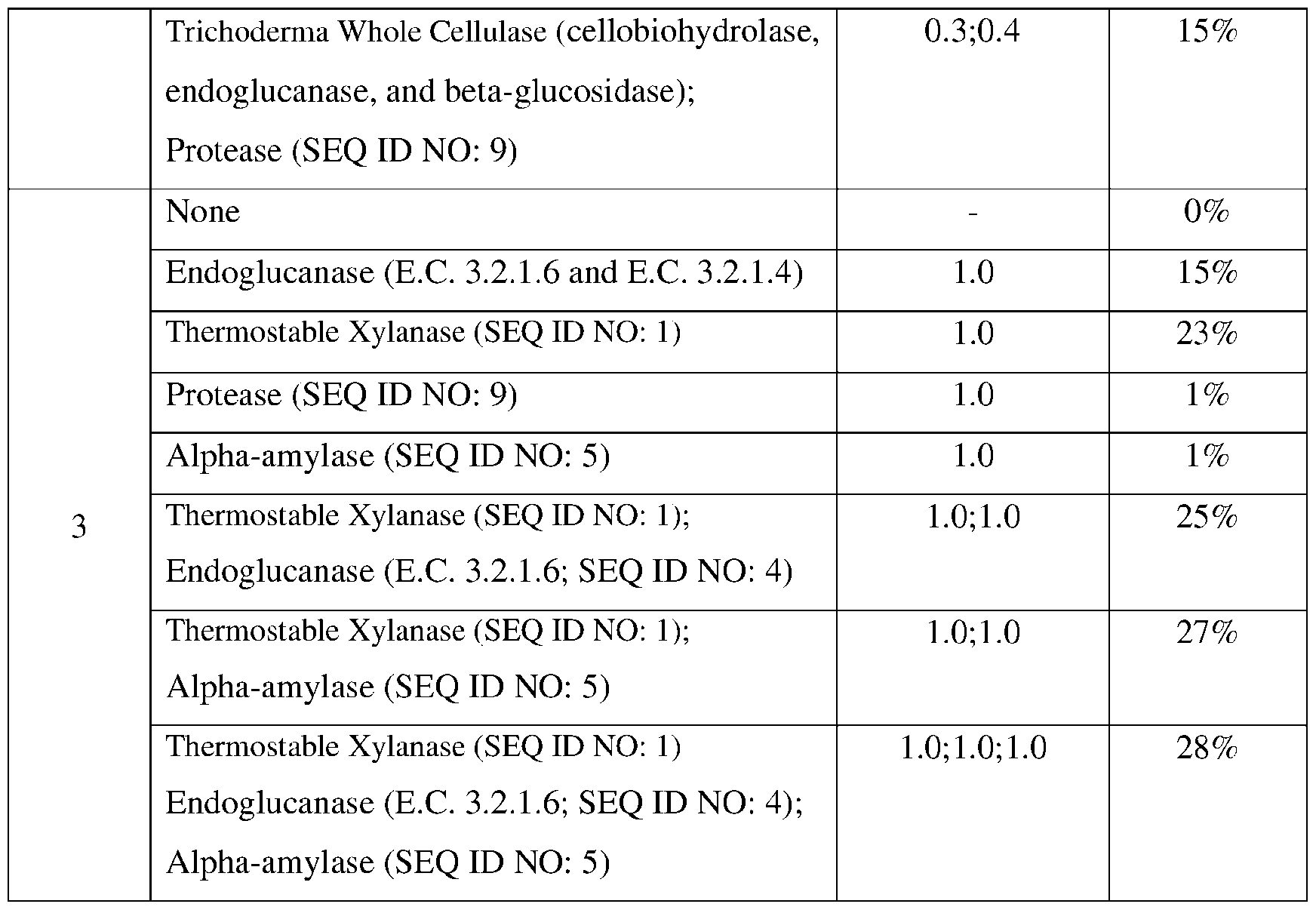
[00 ] e presen nven on s no n ended o be m ed n scope o e par cu ar d sclosed embodiments, which are provided, for example, to illustrate various aspects of the invention. Various modifications to the compositions and methods described will become apparent from the description and teachings herein. Such variations may be practiced without departing from the true scope and spirit of the disclosure and are intended to fall within the scope of the present disclosure. Although the invention may be described in connection with specific preferred embodiments, it should be understood that the invention as claimed should not be unduly limited to such specific embodiments. Indeed, various modifications of the described modes for carrying out the invention which are obvious to those skilled in molecular biology or related fields are intended to be within the scope of the following claims.





