WO2025128283A1 - Microporous membranes demonstrating improved durability and methods of preparing same - Google Patents
Microporous membranes demonstrating improved durability and methods of preparing same Download PDFInfo
- Publication number
- WO2025128283A1 WO2025128283A1 PCT/US2024/056327 US2024056327W WO2025128283A1 WO 2025128283 A1 WO2025128283 A1 WO 2025128283A1 US 2024056327 W US2024056327 W US 2024056327W WO 2025128283 A1 WO2025128283 A1 WO 2025128283A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- microporous membrane
- weight
- percent
- organic polymer
- polymer matrix
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/26—Polyalkenes
- B01D71/261—Polyethylene
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D69/00—Semi-permeable membranes for separation processes or apparatus characterised by their form, structure or properties; Manufacturing processes specially adapted therefor
- B01D69/14—Dynamic membranes
- B01D69/141—Heterogeneous membranes, e.g. containing dispersed material; Mixed matrix membranes
- B01D69/148—Organic/inorganic mixed matrix membranes
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D71/00—Semi-permeable membranes for separation processes or apparatus characterised by the material; Manufacturing processes specially adapted therefor
- B01D71/06—Organic material
- B01D71/38—Polyalkenylalcohols; Polyalkenylesters; Polyalkenylethers; Polyalkenylaldehydes; Polyalkenylketones; Polyalkenylacetals; Polyalkenylketals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01D—SEPARATION
- B01D2323/00—Details relating to membrane preparation
- B01D2323/15—Use of additives
- B01D2323/21—Fillers
Definitions
- Filled microporous membranes are known to be low-cost, efficient, and environmentally friendly materials. However, they are generally not UV stable and tend to disintegrate after prolonged exposure to ultraviolet radiation, such as during outdoor use. They also have end-use limitations related to high temperature instability, due to their low melting temperatures.
- microporous membranes that demonstrate durability with respect to UV stability and high temperature stability, while maintaining the advantageous properties for which microporous polyolefinic membranes are typically known, such as flexibility, high porosity, and excellent printability. It would be further desirable to prepare such membranes using a process that employs current manufacturing steps and equipment without modification thereof.
- the present disclosure is directed to a microporous membrane comprising 1 ) an organic polymer matrix and 2) finely divided, particulate, substantially waterinsoluble filler distributed throughout the organic polymer matrix.
- the organic polymer matrix 1 comprises (a) a polyolefin component comprising ultrahigh molecular weight (UHMW) polyethylene, present in the organic polymer matrix in an amount of 20 to 99 percent by weight, based on the total weight of the organic polymer matrix; and (b) an aliphatic polyketone polymer, present in the organic polymer matrix in an amount of 1 to 80 percent by weight, based on the total weight of the organic polymer matrix.
- the filler 2) is present in the microporous membrane in an amount of 10 percent to 90 percent by weight, based on the total weight of the microporous membrane.
- any numerical range recited herein is intended to include all sub-ranges subsumed therein.
- a range of “1 to 10” is intended to include all sub-ranges between (and including) the recited minimum value of 1 and the recited maximum value of 10, that is, having a minimum value equal to or greater than 1 and a maximum value of equal to or less than 10.
- microporous material or “microporous membrane” or “microporous sheet” means a material having a network of interconnecting pores, wherein, on a treatment- free, coating-free, printing ink-free, impregnant-free, and pre-bonding basis, the pores typically have a volume average diameter ranging from 0.001 to 1.0 micrometer, and may constitute at least 5 percent by volume of the microporous material as discussed below.
- the volume average diameter of the pores of the microporous material can be determined by mercury porosimetry using an Autopore III porosimeter (Micromeritics, Inc. (Norcross, GA)) in accordance with the accompanying operating manual.
- the volume average pore radius for a single scan is automatically determined by the porosimeter.
- a scan is made in the high pressure range (from 138 kilopascals absolute to 227 megapascals absolute). If approximately 2 percent or less of the total intruded volume occurs at the low end (from 138 to 250 kilopascals absolute) of the high pressure range, the volume average pore diameter is taken as twice the volume average pore radius determined by the porosimeter.
- d 2[v ⁇ r ⁇ /wi +V2 r2 /u’2]/[vi /w ⁇ + 1'2 / W2]
- d is the volume average pore diameter
- v1 is the total volume of mercury intruded in the high pressure range
- v2 is the total volume of mercury intruded in the low pressure range
- r1 is the volume average pore radius determined from the high pressure scan
- r2 is the volume average pore radius determined from the low pressure scan
- w1 is the weight of the sample subjected to the high pressure scan
- w2 is the weight of the sample subjected to the low pressure scan.
- the maximum pore radius detected is sometimes noted. This is taken from the low-pressure range scan, if run; otherwise, it is taken from the high pressure range scan.
- the maximum pore diameter is twice the maximum pore radius.
- some production or treatment steps e.g., coating processes, printing processes, impregnation processes and/or bonding processes, can result in the filling of at least some of the pores of the microporous material, and since some of these processes irreversibly compress the microporous material, the parameters in respect of porosity, volume average diameter of the pores, and maximum pore diameter are determined for the microporous material prior to the application of one or more of such production or treatment steps.
- the organic polymer matrix comprises (a) a polyolefin component, which in turn comprises ultrahigh molecular weight (UHMW) polyethylene.
- UHMW ultrahigh molecular weight
- Non-limiting examples of ultrahigh molecular weight (UHMW) polyethylene can include essentially linear UHMW polyethylene (PE).
- PE UHMW polyethylene
- the intrinsic viscosity of the UHMW polyethylene can, for example, range from at least 6 deciliters/gram, or at least 7 deciliters/gram, or at least 18 deciliters/gram, to at most 50 deciliters/gram, or at most 45 deciliters/gram, or at most 18 deciliters/gram, or at most 16 deciliters/gram.
- the intrinsic viscosity of the UHMW may be, for example, 6 to 50 deciliters/gram, or 6 to 45 deciliters/gram, or 6 to 18 deciliters/gram, or 6 to 16 deciliters/gram, or 7 to 50 deciliters/gram, or 7 to 45 deciliters/gram, or 7 to 18 deciliters/gram, or 7 to 16 deciliters/gram, or 18 to 50 deciliters/gram, or 18 to 45 deciliters/gram.
- intrinsic viscosity may be determined by extrapolating to zero concentration the reduced viscosities or the inherent viscosities of several dilute solutions of the UHMW polyolefin where the solvent is freshly distilled decahydronaphthalene to which 0.2 percent by weight, 3,5-di-tert- butyl-4-hydroxyhydrocinnamic acid, neopentanetetrayl ester [CAS Registry No. 6683- 19-8] has been added.
- the reduced viscosities or the inherent viscosities of the UHMW polyolefin are ascertained from relative viscosities obtained at 135°C using an Ubbelohde No.
- M 5.37x10 4 [n] 1 37 wherein M is the nominal molecular weight and [q] is the intrinsic viscosity of the UHMW polyethylene expressed in deciliters/gram.
- the nominal molecular weight of UHMW polypropylene is empirically related to the intrinsic viscosity of the polymer according to the following equation:
- M 8.88x10 4 [q] 1 25 wherein M is the nominal molecular weight and [q] is the intrinsic viscosity of the UHMW polypropylene expressed in deciliters/gram.
- the UHMW polyethylene may be present in the organic polymer matrix in an amount of at least 18 percent by weight, or at least 25 percent by weight, or at least 40 percent by weight, and at most 99 percent by weight, or at most 97 percent by weight, or at most 95 percent by weight, based on the total weight of the organic polymer matrix.
- the UHMW polyethylene may be present in the organic polymer matrix in an amount of 18 to 99 percent by weight, or 18 to 97 percent by weight, or 18 to 95 percent by weight, or 25 to 99 percent by weight, or 25 to 97 percent by weight, or 25 to 95 percent by weight, or 40 to 99 percent by weight, or 40 to 97 percent by weight, or 40 to 95 percent by weight.
- the polyolefin component (a) further comprises high density polyethylene (HOPE), present in an amount of up to 75 percent by weight, based on the total weight of the polyolefin component (a).
- HOPE high density polyethylene
- the HDPE may be present in the organic polymer matrix 1 ) in an amount of at least 0.1 percent by weight, or at least 1 percent by weight, and at most 74.25 percent by weight, or at most 60 percent by weight, or at most 45 percent by weight, based on the total weight of the organic polymer matrix 1 ).
- the HDPE may be present in the organic polymer matrix in an amount of from 0.1 to 74.25 percent by weight, or 0.1 to 60 percent by weight, or 0.1 to 45 percent by weight, or 1 to 74.25 percent by weight, or 1 to 60 percent by weight, or 1 to 45 percent by weight, based on the total weight of the organic polymer matrix.
- thermoplastic organic polymers also may be present in the polyolefin component (a), provided that their presence does not materially affect the properties of the microporous material substrate in an adverse manner.
- the amount of the other thermoplastic polymer which may be present depends upon the nature of such polymer.
- thermoplastic organic polymers that optionally may be present in the matrix of the microporous material include low density polyethylene, poly(tetrafluoroethylene), polypropylene, copolymers of ethylene and propylene, copolymers of ethylene and butylene, copolymers of ethylene and (meth)acrylic (i.
- the organic polymer matrix 1 further comprises (b) an aliphatic polyketone polymer.
- an aliphatic polyketone polymer Commercially available aliphatic polyketones are typically prepared from carbon monoxide, and one or more alkene monomers, such as ethylene, and propylene monomers.
- a polyketone has only carbon-carbon bonds along its backbone, in contrast to a polyetherketone, which includes ether (-O-) linkages along its backbone.
- Suitable aliphatic polyketone polymers (b) comprise an extrudable semi-crystalline thermoplastic, having suitable melting range for compounding with polyolefin in the process of constructing the microporous substrate.
- Suitable aliphatic polyketone polymers include POKETONETM resins, commercially available from Hyosung Chemical, unreinforced AKROTEK® PK resins, available from AKRO-PLASTIC GmbH, and unreinforced grades of Schulaketon®, available from LyondellBasell.
- the aliphatic polyketone polymers (b) may demonstrate a melt index of 0.5 to 4 g/10 min, typically 2 to 4 g/10 min, or 3 g/10 min, measured according to ASTM D1238-23. Suitable aliphatic polyketone polymers (b) typically demonstrate a melt temperature of 190 to 220 °C, measured according to ASTM D3418-21 .
- the aliphatic polyketone polymer (b) may be present in the organic polymer matrix in an amount of at least 1 percent by weight, or at least 3 percent by weight, or at least 5 percent by weight, and at most 80 percent by weight, or at most 75 percent by weight, or at most 60 percent by weight, based on the total weight of the organic polymer matrix.
- the aliphatic polyketone polymer (b) may be present in the organic polymer matrix in an amount of 1 to 80 percent by weight, or 3 to 80 percent by weight, or 5 to 80 percent by weight, or 1 to 75 percent by weight, or 3 to 75 percent by weight, or 5 to 75 percent by weight, or 1 to 60 percent by weight, or 3 to 60 percent by weight, or 5 to 60 percent by weight, based on the total weight of the organic polymer matrix [0023]
- the microporous membranes of the present disclosure further comprise finely divided, particulate, substantially water-insoluble filler distributed throughout the organic polymer matrix.
- the filler can include any of a number of fillers known in the art.
- the filler should be finely divided and substantially water insoluble to permit uniform distribution throughout the organic polymer matrix during manufacture of the microporous membrane.
- the filler comprises silica, such as precipitated silica, talc, carbon black, charcoal, graphite, titanium oxide, iron oxide, copper oxide, zinc oxide, antimony oxide, zirconia, magnesia, alumina, molybdenum disulfide, zinc sulfide, barium sulfate, strontium sulfate, calcium carbonate, and/or magnesium carbonate.
- the filler comprises silica and/or calcium carbonate.
- the filler may comprise silica and calcium carbonate, wherein the weight ratio of silica to calcium carbonate ranges from 0.2:1 to 1 :0.2.
- the finely divided substantially water-insoluble filler may be in the form of ultimate particles, aggregates of ultimate particles, or a combination of both.
- finely divided is meant that usually at least 90 percent by weight of the filler used in preparing the microporous membrane has gross particle sizes in the range of from 5 to 40 micrometers, as determined by the use of a laser diffraction particle size instrument, LS230 from Beckman Coulton, capable of measuring particle diameters as small as 0.04 micron.
- at least 90 percent by weight of the filler has gross particle sizes in the range of from 10 to 30 micrometers.
- the sizes of the filler agglomerates may be reduced during processing of the ingredients used to prepare the microporous membrane. Accordingly, the distribution of gross particle sizes in the microporous membrane may be smaller than in the raw filler itself.
- the filler particles are substantially water-insoluble, and also can be substantially insoluble in any organic processing liquid used to prepare the microporous material.
- substantially insoluble is meant that less than 3 percent by weight, or less than 1 percent by weight of the filler particles, based on the total weight of the filler particles, dissolves into the liquid phase at 25°C when dispersed therein. This can facilitate retention of the filler in the microporous material.
- the filler typically has a high surface area, allowing the filler to carry much of the processing plasticizer used to form the microporous membrane.
- High surface area fillers are materials of very small particle size, materials that have a high degree of porosity, or materials that exhibit both characteristics.
- the surface area of the filler particles can range from at least 20 square meters per gram, or at least 25 square meters per gram, to at most 900 square meters per gram, or at most 850 square meters per gram; e. g., from 20 to 900 square meters per gram, or from 20 to 850 square meters per gram, or from 25 to 900 square meters per gram, or from 25 to 850 square meters per gram, as determined by the Brunauer, Emmett, Teller (BET) method according to ASTM C 819-77 using nitrogen as the adsorbate but modified by outgassing the system and the sample for one hour at 130°C. Prior to nitrogen sorption, filler samples are dried by heating to 160°C in flowing nitrogen (PS) for 1 hour.
- PS flowing nitrogen
- the inorganic filler often comprises silica, for example, precipitated silica, silica gel, or fumed silica.
- the silica may demonstrate a BET of 125 to 700 m 2 /g, determined as noted above.
- Silica gel is generally produced commercially by acidifying an aqueous solution of a soluble metal silicate, e.g., sodium silicate at low pH with acid.
- the acid employed is generally a strong mineral acid, such as sulfuric acid or hydrochloric acid, although carbon dioxide can be used.
- silica gel may be described as a non-precipitated, coherent, rigid, three- dimensional network of contiguous particles of colloidal amorphous silica. The state of subdivision ranges from large, solid masses to submicroscopic particles, and the degree of hydration from almost anhydrous silica to soft gelatinous masses containing on the order of 100 parts of water per part of silica by weight.
- Precipitated silica generally is produced commercially by combining an aqueous solution of a soluble metal silicate, ordinarily alkali metal silicate such as sodium silicate, and an acid so that colloidal particles of silica will grow in a weakly alkaline solution and be coagulated by the alkali metal ions of the resulting soluble alkali metal salt.
- a soluble metal silicate ordinarily alkali metal silicate such as sodium silicate
- an acid so that colloidal particles of silica will grow in a weakly alkaline solution and be coagulated by the alkali metal ions of the resulting soluble alkali metal salt.
- Various acids may be used, including but not limited to mineral acids. Non-limiting examples of acids that can be used include hydrochloric acid and sulfuric acid, but carbon dioxide can also be used to produce precipitated silica. In the absence of a coagulant, silica is not precipitated from solution at any pH.
- the coagulant used to effect precipitation of silica may be the soluble alkali metal salt produced during formation of the colloidal silica particles, or it may be an added electrolyte, such as a soluble inorganic or organic salt, or it may be a combination of both.
- Precipitated silica can be described as precipitated aggregates of ultimate particles of colloidal amorphous silica that have not at any point existed as macroscopic gel during the preparation. The sizes of the aggregates and the degree of hydration may vary widely.
- Precipitated silica powders differ from silica gels that have been pulverized in that the precipitated silica powders generally have a more open structure, that is, a higher specific pore volume. However, the specific surface area of precipitated silica, as measured by the Brunauer, Emmet, Teller (BET) method using nitrogen as the adsorbate, is often lower than that of silica gel.
- BET Brunauer, Emmet, Teller
- precipitated silicas can be employed as the filler used to prepare the microporous membrane.
- Precipitated silicas are well-known commercial materials, and processes for producing them are described in detail in many United States patents, including United States Patent Numbers 2,940,830 and 4,681 ,750.
- the average ultimate particle size (irrespective of whether or not the ultimate particles are agglomerated) of precipitated silicas used is generally less than 0.1 micrometer, e.g., less than 0.05 micrometer or less than 0.03 micrometer, as determined by transmission electron microscopy.
- suitable precipitated silicas include those sold under the HI-SIL tradename by PPG (Pittsburgh, PA).
- the finely divided, particulate, substantially water-insoluble filler 2) may be present in the microporous membrane in an amount of at least 10 percent by weight, or at least 30 percent by weight, or at least 50 percent by weight, and at most 90 percent by weight, or at most 80 percent by weight, or at most 75 percent by weight, based on the total weight of the microporous membrane.
- the filler 2) may be present in the microporous membrane in an amount of 10 percent to 90 percent by weight, or 10 to 80 percent by weight, or 10 to 75 percent by weight, or 30 percent to 90 percent by weight, or 30 to 80 percent by weight, or 30 to 75 percent by weight, or 50 percent to 90 percent by weight, or 50 to 80 percent by weight, or 50 to 75 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of the present disclosure further comprises a network of interconnecting pores communicating throughout the microporous membrane.
- such pores can make up at least 5 percent by volume, or at least 15 percent by volume, or at least 20 percent by volume, or at least 25 percent by volume, or at least 35 percent by volume, or at least 45 percent by volume, and at most 95 percent by volume, or at most 75 percent by volume.
- the pores can make up 5 to 95 percent by volume, or 15 to 95 percent by volume, or 20 to 95 percent by volume, or 25 to 95 percent by volume, or 35 to 95 percent by volume, or 45 to 95 percent by volume, or 5 to 70 percent by volume, or 15 to 70 percent by volume, or 20 to 70 percent by volume, or 25 to 70 percent by volume, or 35 to 70 percent by volume, or 45 to 70 percent by volume of the microporous material.
- the pores comprise at least 35 percent by volume, or even at least 45 percent by volume of the microporous membrane.
- Porosity can be measured using a Gurley Densometer, model 4340, manufactured by GPI Gurley Precision Instruments (Troy, NY).
- Gurley Densometer model 4340, manufactured by GPI Gurley Precision Instruments (Troy, NY).
- the porosity values reported are a measure of the rate of air flow through a sample or its resistance to an air flow through the sample.
- the unit of measure for this method is a “Gurley second” and represents the time in seconds to pass 100 cc of air through a 1 inch square area using a pressure differential of 4.88 inches of water. Lower values equate to less air flow resistance (more air is allowed to pass freely).
- the measurements are completed using the procedure listed in the manual for MODEL 4340 Automatic Densometer.
- the membrane demonstrates a Gurley porosity of 10 to 5000 seconds, or 250 to 5000 seconds, or 500 to 5000 seconds, or 1000 to 5000 seconds, or 10 to 4500 seconds, or 250 to 4500 seconds, or 500 to 4500 seconds, or 1000 to 4500 seconds.
- the microporous membranes may further comprise a biodegradation promoting material, an antioxidant, a lubricant, a UV absorber and/or a UV light stabilizer distributed throughout the organic polymer matrix.
- Biodegradation promoting materials serve to render the microporous membrane compostable and/or biodegradable. Examples include furanone compounds, glutaric acids, hexadecanoic acid compounds, organoleptic swelling agents such as natural fibers, cultured colloids, cyclo-dextrin, and polylactic acid, catalytic transition metal compounds, metal stearates, and/or metal chelates.
- Suitable lubricants include calcium stearate, calcium-zinc stearate, zinc stearate, magnesium stearate, zinc phosphate, and magnesium phosphate.
- Anti-oxidants such as those sold by BASF under the names IRGANOX® and IRGAFOS®, and those sold by Solvay under the name CYANOX®, are suitable for use.
- suitable UV absorbers include those available under the tradename TINUVIN®, such as TINUVIN ® 1130, available from BASF, and HOSTAVIN® PR-25, available from Clariant.
- UV light stabilizers include benzophenones, benzotriazoles, and hindered amine light stabilizers (HALS).
- HALS hindered amine light stabilizers
- Commercially available examples of UV stabilizers include HOSTAVIN® PR-31 , available from Clariant, TINUVIN® 900, available from BASF, and CYANOX® M528, available from Solvay.
- microporous membranes described above may be prepared by a method comprising:
- step (vii) optionally stretching the continuous sheet in at least one stretching direction above the elastic limit, wherein the stretching occurs during or immediately after step (ii) and/or step (iii), but prior to step (iv), to form a microporous membrane.
- the components of the organic polymer matrix typically in solid form such as powder or pellets
- filler typically in solid form such as powder or pellets
- processing plasticizer typically insoluble inorganic compound
- minor amounts of lubricant and antioxidant are mixed until a substantially uniform mixture is obtained.
- the weight ratio of filler to polymer employed in forming the mixture is essentially the same as that of the microporous membrane to be produced.
- the mixture, together with additional processing plasticizer as needed, is introduced to the heated barrel of a screw extruder. Attached to the extruder is a die, such as a sheeting die, to form the desired end shape.
- a continuous sheet or film formed by a die may be forwarded to a pair of heated calender rolls acting cooperatively to form a continuous sheet of lesser thickness than the continuous sheet exiting from the die.
- the final thickness may depend on the desired end-use application.
- the microporous membrane may have a thickness ranging from 10 to 510 microns, such as 15 to 460 microns, or 15 to 390 microns, or 25 to 260 microns, or 125 to 250 microns.
- the sheet exiting the calendar rolls may then be stretched in at least one stretching direction above the elastic limit.
- Stretching may alternatively take place during or immediately after exiting from the sheeting die or during calendaring, or multiple times during the manufacturing process. Stretching may take place before extraction, after extraction, or both. Additionally, stretching may take place during the application of the pre-treatment composition and/or treatment composition, described in more detail below.
- Stretched microporous material substrate may be produced by stretching the intermediate product in at least one stretching direction above the elastic limit.
- the stretch ratio is at least 1 .2. In many cases, the stretch ratio is at least 1 .5. Often it is at least 2. Frequently, the stretch ratio is in the range of from 1 .2 to 15. Often, the stretch ratio is in the range of from 1 .5 to 10. Usually, the stretch ratio is in the range of from 2 to 6.
- the temperatures at which stretching is accomplished may vary widely. Stretching may be accomplished at ambient room temperature, but usually elevated temperatures are employed. Ambient temperature typically ranges from 60 to 90 °F (15.6 to 32.2 °C), such as a typical room temperature, 72°F (22.2°C).
- the intermediate product may be heated by any of a wide variety of techniques prior to, during, and/or after stretching. Examples of these techniques include radiative heating, such as that provided by electrically heated or gas fired infrared heaters; convective heating, such as that provided by recirculating hot air; and conductive heating, such as that provided by contact with heated rolls.
- the temperatures which are measured for temperature control purposes may vary according to the apparatus used and personal preference.
- temperature-measuring devices may be placed to ascertain the temperatures of the surfaces of infrared heaters, the interiors of infrared heaters, the air temperatures of points between the infrared heaters and the intermediate product, the temperatures of circulating hot air at points within the apparatus, the temperature of hot air entering or leaving the apparatus, the temperatures of the surfaces of rolls used in the stretching process, the temperature of heat transfer fluid entering or leaving such rolls, or film surface temperatures.
- the temperature or temperatures are controlled such that the intermediate product is stretched about evenly so that the variations, if any, in film thickness of the stretched microporous material are within acceptable limits and so that the amount of stretched microporous material outside of those limits is acceptably low.
- the temperatures used for control purposes may or may not be close to those of the intermediate product itself since they depend upon the nature of the apparatus used, the locations of the temperaturemeasuring devices, and the identities of the substances or objects whose temperatures are being measured.
- the film surface temperatures at which stretching is accomplished may vary widely, but in general they are such that the intermediate product is stretched about evenly, as explained above. In most cases, the film surface temperatures during stretching are in the range of from 20°C to 220°C. Often, such temperatures are in the range of from 50°C to 200°C, such as from 75°C to 180°C.
- Stretching may be accomplished in a single step or a plurality of steps as desired.
- the stretching may be accomplished by a single stretching step or a sequence of stretching steps until the desired final stretch ratio is attained.
- the stretching can be conducted by a single biaxial stretching step or a sequence of biaxial stretching steps until the desired final stretch ratios are attained.
- Biaxial stretching may also be accomplished by a sequence of one of more uniaxial stretching steps in one direction and one or more uniaxial stretching steps in another direction. Biaxial stretching steps where the intermediate product is stretched simultaneously in two directions and uniaxial stretching steps may be conducted in sequence in any order.
- Stretching in more than two directions is within contemplation. It may be seen that the various permutations of steps are quite numerous. Other steps, such as cooling, heating, sintering, annealing, reeling, unreeling, and the like, may optionally be included in the overall process as desired.
- Uniaxial stretching is usually accomplished by stretching between two rollers, wherein the second or downstream roller rotates at a greater peripheral speed than the first or upstream roller. Uniaxial stretching can also be accomplished on a standard tentering machine. Biaxial stretching may be accomplished by simultaneously stretching in two different directions on a tentering machine. More commonly, however, biaxial stretching is accomplished by first uniaxially stretching between two differentially rotating rollers as described above, followed by either uniaxially stretching in a different direction using a tenter machine or by biaxially stretching using a tenter machine. The most common type of biaxial stretching is where the two stretching directions are approximately at right angles to each other. In most cases where the continuous sheet is being stretched, one stretching direction is at least approximately parallel to the long axis of the sheet (machine direction) and the other stretching direction is at least approximately perpendicular to the machine direction and is in the plane of the sheet (transverse direction).
- Stretching the sheets prior to extraction of the processing plasticizer allows for thinner films with larger pore sizes than in microporous materials conventionally processed. It is also believed that stretching of the sheets prior to extraction of the processing plasticizer minimizes thermal shrinkage after processing. It also should be noted that stretching of the microporous membrane can be conducted at any point prior to, during, or subsequent to application of the pre-treatment composition (as described herein below), and/or prior to, during, or subsequent to application of the treatment composition. Stretching of the microporous membrane can occur once or multiple times during the treatment process.
- the product passes to a first extraction zone where the processing plasticizer is extracted from the sheet with an organic liquid, which is a good solvent for the processing plasticizer, a poor solvent for the organic polymer, and more volatile than the processing plasticizer.
- organic liquid which is a good solvent for the processing plasticizer, a poor solvent for the organic polymer, and more volatile than the processing plasticizer.
- both the processing plasticizer and the organic extraction liquid are substantially immiscible with water.
- the product then passes to a second extraction zone where the residual organic extraction liquid is extracted from the sheet by steam and/or water.
- the product is then passed through a forced air dryer for substantial removal of residual water and remaining residual organic extraction liquid. From the dryer, the microporous material may be passed to a take-up roll, when it is in the form of a sheet.
- the processing plasticizer has little solvating effect on the organic polymers at 60°C, only a moderate solvating effect at elevated temperatures on the order of 100°C, and a significant solvating effect at elevated temperatures on the order of 200°C. It is a liquid at room temperature and usually it is processing oil, such as paraffinic oil, naphthenic oil, or aromatic oil. Suitable processing oils include those meeting the requirements of ASTM D 2226-82, Types 103 and 104. Those oils which have a pour point of less than 22°C, or less than 10°C, according to ASTM D 97-66 (reapproved 1978) are used most often. Examples of suitable oils include SHELLFLEX 412 and SHELLFLEX 371 oil (Shell Oil Co.
- suitable organic extraction liquids include, but are not limited to, 1 ,1 ,2-trichloroethylene; perchloroethylene; 1 ,2- dichloroethane; 1 ,1 ,1 -trichloroethane; 1 ,1 ,2-trichloroethane; methylene chloride; chloroform; 1 ,1 ,2-trichloro-1 ,2,2-trifluoroethane; isopropyl alcohol; diethyl ether; acetone; hexane; heptane and toluene.
- One or more azeotropes of halogenated hydrocarbons selected from trans-1 ,2-dichloroethylene, 1 ,1 ,1 ,2,2,3, 4,5, 5,5- decafluoropentane, and/or 1 ,1 ,1 ,3,3-pentafluorobutane also can be employed.
- VERTREL MCA binary azeotrope of 1 ,1 ,1 ,2,2,3,4,5,5,5-dihydrodecafluoropentane and trans-1 ,2-dichloroethylene: 62%/38%)
- VERTREL CCA a ternary azeotrope of 1 ,1 ,1 ,2, 2, 3, 4, 5,5,5- dihydrodecafluorpentane, 1 ,1 ,1 ,3,3-pentafluorbutane, and trans-1 ,2-dichloroethylene: 33%/28%/39%
- VERTREL SDG 80-83% trans-1 ,2-dichloroethylene, 17-20% hydrofluorocarbon mixture
- the filler In the above-described process for producing microporous membrane, extrusion and calendering are facilitated when the filler carries much of the processing plasticizer.
- the capacity of the filler particles to absorb and hold the processing plasticizer is a function of the surface area of the filler. Therefore, the filler typically has a high surface area as discussed above. Inasmuch as it is desirable to essentially retain the filler in the microporous material substrate, the filler should be substantially insoluble in the processing plasticizer and substantially insoluble in the organic extraction liquid when microporous material substrate is produced by the above process.
- the microporous membranes described above may be used to prepare a multilayer membrane comprising two or more adjacent membrane layers.
- the layers may be the same as or different from each other, and at least one layer comprises a microporous membrane described above.
- different embodiments of the microporous membrane described above may be used as separate layers, or the layers may be the same.
- the use of different membrane layers allows for the specific properties afforded by each membrane to be incorporated into the final multilayer product.
- At least one layer of the multilayer membrane may comprise a microporous polyolefinic material, wherein the polyolefinic material comprises linear ultrahigh molecular weight polyethylene, linear ultrahigh molecular weight polypropylene, or a mixture thereof; and finely divided, particulate, substantially water-insoluble silica filler distributed throughout the microporous polyolefinic material.
- polyolefinic materials are available from PPG as TESLIN®.
- the fibers may be natural, such as wood fibers, cotton fibers, wool fibers, silk fibers, and the like; or they may be any of the artificial fibers such as polyester fibers, polyamide fibers, acrylic fibers, modacrylic fibers, rayon fibers, and the like; or they may be combinations of differing kinds of fibers.
- the fibers may be staple and/or they may be continuous. Metal fibers, carbon fibers, and glass fibers are also suitable.
- the multilayer membrane may be constructed by bonding at least one layer of microporous material to at least one layer of porous material.
- the multilayer membrane may comprise one layer or more than one layer of microporous material and one layer or more than one layer of the porous material.
- the multilayer membrane may also optionally be bonded to one or more layers of porous material.
- the microporous material may be bonded to a scrim, which is bonded to a nonwoven fabric. In this case, the microporous material is often also bonded through the open areas of the scrim to the nonwoven fabric.
- the microporous material is bonded to a nonwoven fabric, the other side of which is bonded to a scrim.
- the combinations and permutations of possible layers are quite numerous. Most often, but not always, at least one exterior layer is microporous material.
- Bonding may be made by conventional techniques such as, for example, fusion bonding and adhesive bonding.
- fusion bonding include sealing through use of heated rollers, heated bars, heated plates, heated bands, heated wires, flame bonding, radio frequency (RF) sealing, and ultrasonic sealing. Heat sealing is used most often.
- Solvent bonding may be used where the porous material is soluble in the applied solvent at least to the extent that the surface becomes tacky. After the microporous material has been brought into contact with the tacky surface, the solvent is removed to form a fusion bond.
- thermosetting adhesives thermoplastic adhesive
- adhesives which form the bond by solvent evaporation adhesives which form the bond by evaporation of liquid nonsolvent
- pressure sensitive adhesives are examples of suitable classes of adhesives.
- Foamable compositions may be foamed in contact with the microporous material to form a bond between the resulting porous foam and the microporous material.
- Powder bonding is a technique which is particularly useful for bonding the microporous material to nonwoven webs of staple and/or continuous fibers and to woven or knitted fibers.
- the adhesive employed, and/or the nature of the porous material used the porous material may be readily removable from the microporous material by hand (i. e., peelable), or not (i. e., permanently bonded).
- the microporous material may be continuously bonded to the porous material, or it may be discontinuously bonded to the porous material.
- discontinuous bonds include bonding areas in the form of one or more spots, patches, strips, stripes, open-curved stripes, closed-curved stripes, irregular areas, and the like. When patterns of bonds are involved, they may be random, repetitive, or a combination of both.
- the multilayer membrane may also be formed by a continuous, moisture vapor permeable coating of hydrophobic polymer joined to one side of the microporous membrane described above.
- Such coatings and methods of applying them are described, for example, in United States Patent Number 5,032,450, which is incorporated herein by reference in its entirety.
- the coating is usually an elastomeric solid at ambient temperatures, and often comprises a polysiloxane.
- One surface of the coating may be in interlocking relationship with a surface of the microporous material, but the coating does not grossly penetrate into the interior of the microporous material. The resulting membrane is therefore perceived as a multi-layered article.
- a polymer is “hydrophobic” if the contact angle between a drop of water and a flat surface of the polymer upon which the drop is resting is greater than 90 degrees. Contact angle is discussed more fully in Sears and Zemansky, University Physics, Second Edition, Addison-Wesley Publishing Company, Inc., (1955), pages 231 -235.
- microporous membrane of the present disclosure is particularly useful for bonding to porous polyolefins such as polyethylene and polypropylene materials by heat sealing in the absence of extrinsic intervening adhesive.
- the bond obtained is ordinarily quite strong which is surprising inasmuch as the lamination of materials to polyolefins is usually difficult unless special adhesives are used.
- the multilayer membranes of the present disclosure have many and varied uses including filters, wipers, gaskets, cushion assemblies, signs, printing substrate, substrates for pen and ink drawings, maps (particularly maritime maps), wall coverings, and breathable fabrics for clothing. They are also useful as or as components of bandages, sanitary napkins, incontinence devices, diapers, and the like. They are also useful as the seams, joints, and seals of breathable packages, breathable wearing apparel and the like.
- a microporous membrane comprising
- an organic polymer matrix comprising:
- a polyolefin component present in the organic polymer matrix in an amount of 18 to 99 percent by weight, based on the total weight of the organic polymer matrix, wherein the polyolefin component comprises ultrahigh molecular weight (UHMW) polyethylene; and
- microporous membrane of aspect 1 wherein the polyolefin component is present in the organic polymer matrix in an amount of 18 to 97 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the polyolefin component is present in the organic polymer matrix in an amount of 18 to 95 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the polyolefin component is present in the organic polymer matrix in an amount of 25 to 99 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the polyolefin component is present in the organic polymer matrix in an amount of 25 to 97 percent by weight, based on the total weight of the organic polymer matrix. 6. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 25 to 95 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the polyolefin component is present in the organic polymer matrix in an amount of 40 to 99 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the polyolefin component is present in the organic polymer matrix in an amount of 40 to 97 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 3 to 80 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 5 to 80 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 1 to 75 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 3 to 75 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 5 to 75 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 1 to 60 percent by weight, based on the total weight of the organic polymer matrix.
- the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 3 to 60 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 5 to 60 percent by weight, based on the total weight of the organic polymer matrix.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 10 to 80 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 10 to 75 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 30 to 90 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 30 to 80 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 30 to 75 percent by weight, based on the total weight of the microporous membrane.
- finely divided, particulate, substantially water-insoluble filler distributed throughout said organic polymer matrix is present in the microporous membrane in an amount of 50 to 90 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 50 to 80 percent by weight, based on the total weight of the microporous membrane.
- microporous membrane of any preceding aspect wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 50 to 75 percent by weight, based on the total weight of the microporous membrane.
- HDPE high density polyethylene
- microporous membrane of any preceding aspect wherein the HDPE is present in the organic polymer matrix in an amount of from 0.1 to 45 percent by weight, based on the total weight of the organic polymer matrix.
- the filler comprises silica, talc, carbon black, charcoal, graphite, titanium oxide, iron oxide, copper oxide, zinc oxide, antimony oxide, zirconia, magnesia, alumina, molybdenum disulfide, zinc sulfide, barium sulfate, strontium sulfate, calcium carbonate, and/or magnesium carbonate.
- the filler comprises silica and calcium carbonate, and a weight ratio of silica to calcium carbonate ranges from 0.2:1 to 1 :0.2. 38.
- BET Brunauer, Emmett, Teller
- microporous membrane of any preceding aspect wherein the microporous membrane has a thickness ranging from 10 to 510 microns.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 10 to 5000 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 250 to 5000 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 500 to 5000 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 1000 to 5000 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 10 to 4500 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 250 to 4500 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 500 to 4500 seconds.
- microporous membrane of any previous aspect wherein the membrane demonstrates a Gurley porosity of 1000 to 4500 seconds.
- microporous membrane of any preceding aspect further comprising a biodegradation promoting material distributed throughout the organic polymer matrix, thereby rendering the microporous membrane compostable and/or biodegradable.
- biodegradation promoting material comprises a furanone compound, a glutaric acid, a hexadecanoic acid compound, an organoleptic swelling agent, a catalytic transition metal compound, a metal stearate, and/or a metal chelate.
- microporous membrane of any preceding aspect further comprising an antioxidant, a lubricant, a UV absorber and/or a UV light stabilizer distributed throughout the organic polymer matrix.
- a multilayer membrane comprising two or more adjacent membrane layers, wherein the layers are the same as or different from each other, and wherein at least one layer comprises the microporous membrane of any preceding aspect.
- the multilayer membrane of aspect 51 wherein at least one layer comprises a microporous polyolefinic material, and wherein the polyolefinic material comprises linear ultrahigh molecular weight polyethylene, linear ultrahigh molecular weight polypropylene, or a mixture thereof; and finely divided, particulate, substantially water-insoluble silica filler distributed throughout the microporous polyolefinic material.
- the mixes of the examples were extruded and calendered into final sheet form using an extrusion system including a feeding, extrusion and calendering system described as follows.
- a gravimetric loss in weight feed system K-Tron model K2MLT35D5
- the extruder barrel was comprised of eight temperature zones and a heated adaptor to the sheet die.
- the extrusion mixture feed port was located just prior to the first temperature zone.
- An atmospheric vent was located in the third temperature zone.
- a vacuum vent was located in the seventh temperature zone.
- the mix was fed into the extruder at a rate of 90 g/minute. Additional processing oil also was injected at the first temperature zone, as required, to achieve a total amount of 60-100 parts by weight based on the solid components in Tables 2 and 3.
- Extrudate from the barrel was discharged into a 15-centimeter wide sheet Masterflex® die having a 1.5 millimeter discharge opening.
- the extrusion melt temperature was 203- 210 °C and the throughput was 7.5 kilograms per hour.
- the calendering process was accomplished using a three-roll vertical calender stack with one nip point and one cooling roll. Each of the rolls had a chrome surface. Roll dimensions were approximately 41 cm in length and 14 cm in diameter. The top roll temperature was maintained between 135 °C to 140 °C. The middle roll temperature was maintained between 140 °C. to 145 °C. The bottom roll was a cooling roll wherein the temperature was maintained between 10-21 °C. The extrudate was calendered into sheet form and passed over the bottom water-cooled roll and wound up.
- a sample of sheet cut to a width of approximately 18 cm and an approximate length of 150 cm was rolled up along with a stainless steel wire mesh into a cylinder shape, placed in a canister and exposed to room temperature liquid 1 ,1 ,2- trichloroethylene for approximately 1 hour to extract processing oil from the sheet sample.
- the remaining oil content in the samples was from 3-7 wt. %.
- the extracted sheet was air dried and subjected to test methods described hereinafter.
- HI-SIL 135® precipitated silica obtained commercially from PPG Industries, Inc.
- GUR® 4160 Ultra High Molecular Weight Polyethylene (UHMWPE) commercially available from Ticona Corp, and reported to have a molecular weight of about 9.2 million grams per mole.
- TIPURETM R-103 titanium dioxide commercially available from E.L du Pont de Nemours and Company.
- compositions further containing HDPE in polyolefin component
- CE-2 All compositions except CE-2 yielded smooth continuous microporous sheets. CE-2 did not survive the extrusion conditions and was not subject to testing.
- Gurley porosity was determined using a Gurley densometer, model 4340, manufactured by GPI Gurley Precision Instruments of Troy, N.Y.
- the air flow rate reported was a measure of the rate of air flow through a sample of the example or it' s resistance to an air flow through the sample.
- the unit of measure is a "Gurley second" and represents the time in seconds to pass 100 cc of air through a 1 inch square area using a pressure differential of 4.88 inches of water. Lower values equate to less air flow resistance (more air is allowed to pass freely).
- the measurements were completed using the procedure listed in the manual, MODEL 4340 Automatic Densometer and Smoothness Tester Instruction Manual. As shown in Tables 3 and 4, when using polyketone, highly porous sheets were produced.
- CE-2 with 100% polyketone and no polyolefin in the organic polymer matrix was not processable by the listed method.
- compositions with polyolefin and polyketone produced excellent sheets with good strength.
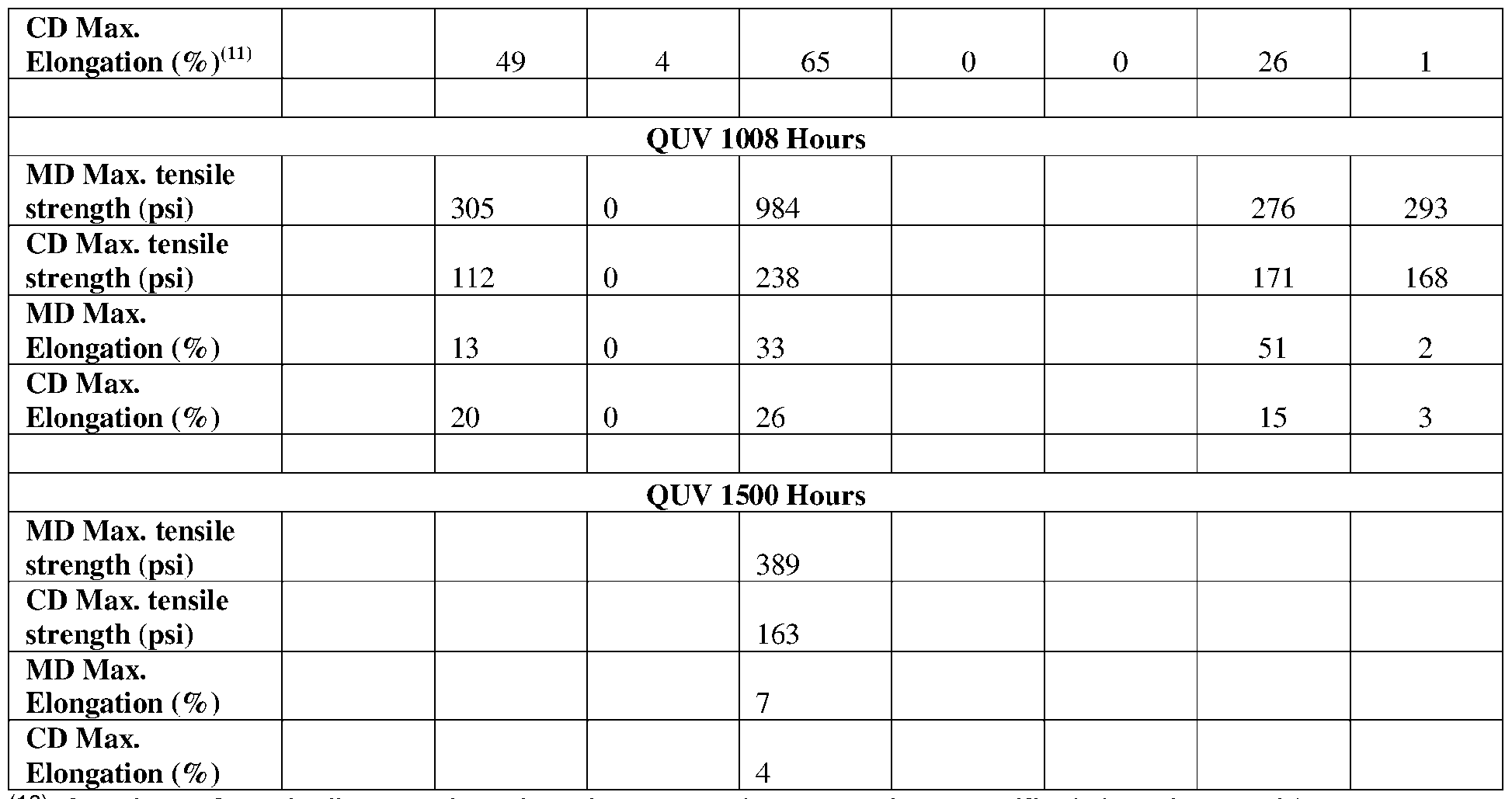
- UVA-340 test methods for QUV accelerated weathering in accordance with SAE J2020 (UVA).
- Samples were mounted on 6 x 3 inch aluminum Q-panels and taped only at the top and bottom, half inch from the edge, of the panel to keep the sheets stable.
- the panels were exposed to ultraviolet light (UV) irradiance of approximately 0.72 W/m2 in a weatherometer equipped with UVA-340 lamps.
- UV ultraviolet light
- the panels were removed from the testing cabinet and the sheets carefully released from the aluminum panels by removing the holding adhesive tape on the top and bottom parts of the panel.
- the 3 x 6 inch size sheets were cut into 3 strips of equal size using a die cutter into approximately 0.5 x 4.5 inch (1.2 x 5.08 cm) strips where the length of the strip represented the MD.
- the 0.5 x 4.5 inch (1 .2 x 5.08 cm) strips were cut in an orientation in corresponding to the CD orientation of the produced microporous sheet.
- Each strip was tested individually to give a total of three tensile readings from the three strips and the arithmetic average calculated from the data in accordance with ASTM D882-18, as previously described, to obtain their corresponding maximum tensile strength and maximum tensile elongation.
- a value of “0” indicates that the sheet was intact at the specified time interval but was unable to undergo testing; e.g. the sheet broke before or at the onset of testing.
- Thermal stability property was measured by tensile modulus of elasticity retention as shown in Table 6.
- the thermoforming properties of the sheets were predictably superior to the comparative.
- High temperature tensile measurements showed a more stable microporous sheet at 155 °C at both low and high strain rates as illustrated by CE-7 vs Example 10.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Inorganic Chemistry (AREA)
- Dispersion Chemistry (AREA)
- Manufacture Of Porous Articles, And Recovery And Treatment Of Waste Products (AREA)
Abstract
A microporous membrane is provided, comprising 1) an organic polymer matrix and 2) particulate filler distributed throughout the matrix. The matrix comprises a polyolefin component comprising ultrahigh molecular weight polyethylene present in the organic polymer matrix in an amount of 1 to 80 percent by weight, and an aliphatic polyketone polymer present in the organic polymer matrix in an amount of 1 to 80 percent by weight. The particulate filler demonstrates gross particle sizes in the range of from 5 to 40 micrometers, as determined by the use of a laser diffraction particle size instrument, is distributed throughout the organic polymer matrix, and present in the microporous membrane in an amount of 10 to 90 percent by weight, based on the total weight of the microporous membrane.
Description
MICROPOROUS MEMBRANES DEMONSTRATING IMPROVED DURABILITY AND METHODS OF PREPARING SAME
FIELD OF THE DISCLOSURE
[0001] The present disclosure relates to microporous membranes that demonstrate outstanding durability, and methods for preparing the microporous membranes.
BACKGROUND
[0002] The manufacture of microporous polyolefinic materials and membranes for various industrial and consumer applications is well known. Filled microporous membranes are known to be low-cost, efficient, and environmentally friendly materials. However, they are generally not UV stable and tend to disintegrate after prolonged exposure to ultraviolet radiation, such as during outdoor use. They also have end-use limitations related to high temperature instability, due to their low melting temperatures.
[0003] Approaches to solving UV instability of these membranes have included addition of UV absorbers and light stabilizers during manufacturing. These additives, however, can only improve UV stability by 5-10%; they are also expensive and may contain substances of concern. Microporous substrates may be coated to modify their surfaces and improve UV properties; however, such coatings tend to have short lifespans, and the vulnerability of the underlying base substrate poses a failure risk if the protective coating is breached, scratched, or degraded.
[0004] It would be desirable to provide microporous membranes that demonstrate durability with respect to UV stability and high temperature stability, while maintaining the advantageous properties for which microporous polyolefinic membranes are typically known, such as flexibility, high porosity, and excellent printability. It would be further desirable to prepare such membranes using a process that employs current manufacturing steps and equipment without modification thereof.
SUMMARY OF THE DISCLOSURE
[0005] The present disclosure is directed to a microporous membrane comprising 1 ) an organic polymer matrix and 2) finely divided, particulate, substantially waterinsoluble filler distributed throughout the organic polymer matrix. The organic polymer matrix 1 ) comprises (a) a polyolefin component comprising ultrahigh molecular weight
(UHMW) polyethylene, present in the organic polymer matrix in an amount of 20 to 99 percent by weight, based on the total weight of the organic polymer matrix; and (b) an aliphatic polyketone polymer, present in the organic polymer matrix in an amount of 1 to 80 percent by weight, based on the total weight of the organic polymer matrix. The filler 2) is present in the microporous membrane in an amount of 10 percent to 90 percent by weight, based on the total weight of the microporous membrane.
DETAILED DESCRIPTION
[0006] Other than in any operating examples, or where otherwise indicated, all numbers expressing, for example, quantities of ingredients used in the specification and claims are to be understood as being modified in all instances by the term “about”. Accordingly, unless indicated to the contrary, the numerical parameters set forth in the following specification and attached claims are approximations that may vary depending upon the desired properties to be obtained by the disclosed membranes. At the very least, and not as an attempt to limit the application of the doctrine of equivalents to the scope of the claims, each numerical parameter should at least be construed in light of the number of reported significant digits and by applying ordinary rounding techniques.
[0007] Notwithstanding that the numerical ranges and parameters setting forth the broad scope of the disclosure are approximations, the numerical values set forth in the specific examples are reported as precisely as possible. Any numerical value, however, inherently contains certain errors necessarily resulting from the standard variation found in their respective testing measurements.
[0008] Also, it should be understood that any numerical range recited herein is intended to include all sub-ranges subsumed therein. For example, a range of “1 to 10” is intended to include all sub-ranges between (and including) the recited minimum value of 1 and the recited maximum value of 10, that is, having a minimum value equal to or greater than 1 and a maximum value of equal to or less than 10.
[0009] In this application, the use of the singular includes the plural and plural encompasses singular, unless specifically stated otherwise. Further, in this application, the use of “a” or “an” means “at least one” unless specifically stated otherwise. For example, “an” additive, “a” silica, and the like refer to one or more of these items. Also, as used herein, the term “polymer” is meant to refer to prepolymers,
oligomers, and both homopolymers and copolymers. The term “resin” is used interchangeably with “polymer.”
[0010] The present disclosure is directed to a microporous membrane. As used herein, “microporous material” or “microporous membrane” or “microporous sheet” means a material having a network of interconnecting pores, wherein, on a treatment- free, coating-free, printing ink-free, impregnant-free, and pre-bonding basis, the pores typically have a volume average diameter ranging from 0.001 to 1.0 micrometer, and may constitute at least 5 percent by volume of the microporous material as discussed below.
[0011] The volume average diameter of the pores of the microporous material can be determined by mercury porosimetry using an Autopore III porosimeter (Micromeritics, Inc. (Norcross, GA)) in accordance with the accompanying operating manual. The volume average pore radius for a single scan is automatically determined by the porosimeter. In operating the porosimeter, a scan is made in the high pressure range (from 138 kilopascals absolute to 227 megapascals absolute). If approximately 2 percent or less of the total intruded volume occurs at the low end (from 138 to 250 kilopascals absolute) of the high pressure range, the volume average pore diameter is taken as twice the volume average pore radius determined by the porosimeter. Otherwise, an additional scan is made in the low pressure range (from 7 to 165 kilopascals absolute) and the volume average pore diameter is calculated according to the equation: d=2[v\ r\ /wi +V2 r2 /u’2]/[vi /w\ + 1'2 / W2] wherein d is the volume average pore diameter, v1 is the total volume of mercury intruded in the high pressure range, v2 is the total volume of mercury intruded in the low pressure range, r1 is the volume average pore radius determined from the high pressure scan, r2 is the volume average pore radius determined from the low pressure scan, w1 is the weight of the sample subjected to the high pressure scan, and w2 is the weight of the sample subjected to the low pressure scan.
[0012] In the course of determining the volume average pore diameter of the above procedure, the maximum pore radius detected is sometimes noted. This is taken from the low-pressure range scan, if run; otherwise, it is taken from the high pressure range scan. The maximum pore diameter is twice the maximum pore radius. Inasmuch as some production or treatment steps, e.g., coating processes, printing processes, impregnation processes and/or bonding processes, can result in the filling of at least
some of the pores of the microporous material, and since some of these processes irreversibly compress the microporous material, the parameters in respect of porosity, volume average diameter of the pores, and maximum pore diameter are determined for the microporous material prior to the application of one or more of such production or treatment steps.
[0013] The organic polymer matrix comprises (a) a polyolefin component, which in turn comprises ultrahigh molecular weight (UHMW) polyethylene. Non-limiting examples of ultrahigh molecular weight (UHMW) polyethylene can include essentially linear UHMW polyethylene (PE). Inasmuch as UHMW polyolefins are not thermoset polymers having an infinite molecular weight, they are technically classified as thermoplastic materials.
[0014] While there is no particular restriction on the upper limit of the intrinsic viscosity of the UHMW polyethylene, the intrinsic viscosity can, for example, range from at least 6 deciliters/gram, or at least 7 deciliters/gram, or at least 18 deciliters/gram, to at most 50 deciliters/gram, or at most 45 deciliters/gram, or at most 18 deciliters/gram, or at most 16 deciliters/gram. Thus, the intrinsic viscosity of the UHMW may be, for example, 6 to 50 deciliters/gram, or 6 to 45 deciliters/gram, or 6 to 18 deciliters/gram, or 6 to 16 deciliters/gram, or 7 to 50 deciliters/gram, or 7 to 45 deciliters/gram, or 7 to 18 deciliters/gram, or 7 to 16 deciliters/gram, or 18 to 50 deciliters/gram, or 18 to 45 deciliters/gram.
[0015] For purposes of the present disclosure, intrinsic viscosity may be determined by extrapolating to zero concentration the reduced viscosities or the inherent viscosities of several dilute solutions of the UHMW polyolefin where the solvent is freshly distilled decahydronaphthalene to which 0.2 percent by weight, 3,5-di-tert- butyl-4-hydroxyhydrocinnamic acid, neopentanetetrayl ester [CAS Registry No. 6683- 19-8] has been added. The reduced viscosities or the inherent viscosities of the UHMW polyolefin are ascertained from relative viscosities obtained at 135°C using an Ubbelohde No. 1 viscometer in accordance with the general procedures of ASTM D 4020-81 , except that several dilute solutions of differing concentration are employed. [0016] The nominal molecular weight of UHMW polyethylene is empirically related to the intrinsic viscosity of the polymer in accordance with the following equation:
M=5.37x104[n]1 37 wherein M is the nominal molecular weight and [q] is the intrinsic viscosity of the UHMW polyethylene expressed in deciliters/gram. Similarly, the nominal molecular
weight of UHMW polypropylene is empirically related to the intrinsic viscosity of the polymer according to the following equation:
M=8.88x104[q]1 25 wherein M is the nominal molecular weight and [q] is the intrinsic viscosity of the UHMW polypropylene expressed in deciliters/gram.
[0017] The UHMW polyethylene may be present in the organic polymer matrix in an amount of at least 18 percent by weight, or at least 25 percent by weight, or at least 40 percent by weight, and at most 99 percent by weight, or at most 97 percent by weight, or at most 95 percent by weight, based on the total weight of the organic polymer matrix. For example, the UHMW polyethylene may be present in the organic polymer matrix in an amount of 18 to 99 percent by weight, or 18 to 97 percent by weight, or 18 to 95 percent by weight, or 25 to 99 percent by weight, or 25 to 97 percent by weight, or 25 to 95 percent by weight, or 40 to 99 percent by weight, or 40 to 97 percent by weight, or 40 to 95 percent by weight.
[0018] Often, the polyolefin component (a) further comprises high density polyethylene (HOPE), present in an amount of up to 75 percent by weight, based on the total weight of the polyolefin component (a). When used, the HDPE may be present in the organic polymer matrix 1 ) in an amount of at least 0.1 percent by weight, or at least 1 percent by weight, and at most 74.25 percent by weight, or at most 60 percent by weight, or at most 45 percent by weight, based on the total weight of the organic polymer matrix 1 ). For example, the HDPE may be present in the organic polymer matrix in an amount of from 0.1 to 74.25 percent by weight, or 0.1 to 60 percent by weight, or 0.1 to 45 percent by weight, or 1 to 74.25 percent by weight, or 1 to 60 percent by weight, or 1 to 45 percent by weight, based on the total weight of the organic polymer matrix.
[0019] In certain examples, other thermoplastic organic polymers also may be present in the polyolefin component (a), provided that their presence does not materially affect the properties of the microporous material substrate in an adverse manner. The amount of the other thermoplastic polymer which may be present depends upon the nature of such polymer. Non-limiting examples of thermoplastic organic polymers that optionally may be present in the matrix of the microporous material include low density polyethylene, poly(tetrafluoroethylene), polypropylene, copolymers of ethylene and propylene, copolymers of ethylene and butylene, copolymers of ethylene and (meth)acrylic (i. e., acrylic and/or methacrylic) acid,
polyetherketone, polyvinylidene fluoride (PVDF), polysulfone, and/or polyethersulfone. If desired, all or a portion of the carboxyl groups of carboxyl-containing copolymers can be neutralized with sodium, zinc, or the like. Note that the phrase “and/or” when used in a list is meant to encompass alternative examples including each individual component in the list as well as any combination of components. For example, the list “A, B, and/or C” is meant to encompass seven separate examples that include A, or B, or C, or A + B, or A + C, or B + C, or A + B + C.
[0020] The organic polymer matrix 1 ) further comprises (b) an aliphatic polyketone polymer. Commercially available aliphatic polyketones are typically prepared from carbon monoxide, and one or more alkene monomers, such as ethylene, and propylene monomers. For the purposes of the present disclosure, a polyketone has only carbon-carbon bonds along its backbone, in contrast to a polyetherketone, which includes ether (-O-) linkages along its backbone. Suitable aliphatic polyketone polymers (b) comprise an extrudable semi-crystalline thermoplastic, having suitable melting range for compounding with polyolefin in the process of constructing the microporous substrate. Examples of suitable aliphatic polyketone polymers (b) include POKETONE™ resins, commercially available from Hyosung Chemical, unreinforced AKROTEK® PK resins, available from AKRO-PLASTIC GmbH, and unreinforced grades of Schulaketon®, available from LyondellBasell.
[0021] The aliphatic polyketone polymers (b) may demonstrate a melt index of 0.5 to 4 g/10 min, typically 2 to 4 g/10 min, or 3 g/10 min, measured according to ASTM D1238-23. Suitable aliphatic polyketone polymers (b) typically demonstrate a melt temperature of 190 to 220 °C, measured according to ASTM D3418-21 .
[0022] The aliphatic polyketone polymer (b) may be present in the organic polymer matrix in an amount of at least 1 percent by weight, or at least 3 percent by weight, or at least 5 percent by weight, and at most 80 percent by weight, or at most 75 percent by weight, or at most 60 percent by weight, based on the total weight of the organic polymer matrix. For example, the aliphatic polyketone polymer (b) may be present in the organic polymer matrix in an amount of 1 to 80 percent by weight, or 3 to 80 percent by weight, or 5 to 80 percent by weight, or 1 to 75 percent by weight, or 3 to 75 percent by weight, or 5 to 75 percent by weight, or 1 to 60 percent by weight, or 3 to 60 percent by weight, or 5 to 60 percent by weight, based on the total weight of the organic polymer matrix
[0023] The microporous membranes of the present disclosure further comprise finely divided, particulate, substantially water-insoluble filler distributed throughout the organic polymer matrix.
[0024] The filler can include any of a number of fillers known in the art. The filler should be finely divided and substantially water insoluble to permit uniform distribution throughout the organic polymer matrix during manufacture of the microporous membrane. Generally, the filler comprises silica, such as precipitated silica, talc, carbon black, charcoal, graphite, titanium oxide, iron oxide, copper oxide, zinc oxide, antimony oxide, zirconia, magnesia, alumina, molybdenum disulfide, zinc sulfide, barium sulfate, strontium sulfate, calcium carbonate, and/or magnesium carbonate. Often the filler comprises silica and/or calcium carbonate. For example, the filler may comprise silica and calcium carbonate, wherein the weight ratio of silica to calcium carbonate ranges from 0.2:1 to 1 :0.2.
[0025] The finely divided substantially water-insoluble filler may be in the form of ultimate particles, aggregates of ultimate particles, or a combination of both. By “finely divided” is meant that usually at least 90 percent by weight of the filler used in preparing the microporous membrane has gross particle sizes in the range of from 5 to 40 micrometers, as determined by the use of a laser diffraction particle size instrument, LS230 from Beckman Coulton, capable of measuring particle diameters as small as 0.04 micron. Typically, at least 90 percent by weight of the filler has gross particle sizes in the range of from 10 to 30 micrometers. The sizes of the filler agglomerates may be reduced during processing of the ingredients used to prepare the microporous membrane. Accordingly, the distribution of gross particle sizes in the microporous membrane may be smaller than in the raw filler itself.
[0026] As mentioned previously, the filler particles are substantially water-insoluble, and also can be substantially insoluble in any organic processing liquid used to prepare the microporous material. By “substantially insoluble” is meant that less than 3 percent by weight, or less than 1 percent by weight of the filler particles, based on the total weight of the filler particles, dissolves into the liquid phase at 25°C when dispersed therein. This can facilitate retention of the filler in the microporous material. [0027] The filler typically has a high surface area, allowing the filler to carry much of the processing plasticizer used to form the microporous membrane. High surface area fillers are materials of very small particle size, materials that have a high degree of porosity, or materials that exhibit both characteristics. The surface area of the filler
particles can range from at least 20 square meters per gram, or at least 25 square meters per gram, to at most 900 square meters per gram, or at most 850 square meters per gram; e. g., from 20 to 900 square meters per gram, or from 20 to 850 square meters per gram, or from 25 to 900 square meters per gram, or from 25 to 850 square meters per gram, as determined by the Brunauer, Emmett, Teller (BET) method according to ASTM C 819-77 using nitrogen as the adsorbate but modified by outgassing the system and the sample for one hour at 130°C. Prior to nitrogen sorption, filler samples are dried by heating to 160°C in flowing nitrogen (PS) for 1 hour.
[0028] The inorganic filler often comprises silica, for example, precipitated silica, silica gel, or fumed silica. The silica may demonstrate a BET of 125 to 700 m2/g, determined as noted above.
[0029] Silica gel is generally produced commercially by acidifying an aqueous solution of a soluble metal silicate, e.g., sodium silicate at low pH with acid. The acid employed is generally a strong mineral acid, such as sulfuric acid or hydrochloric acid, although carbon dioxide can be used. Inasmuch as there is essentially no difference in density between the gel phase and the surrounding liquid phase while the viscosity is low, the gel phase does not settle out, that is to say, it does not precipitate. Consequently, silica gel may be described as a non-precipitated, coherent, rigid, three- dimensional network of contiguous particles of colloidal amorphous silica. The state of subdivision ranges from large, solid masses to submicroscopic particles, and the degree of hydration from almost anhydrous silica to soft gelatinous masses containing on the order of 100 parts of water per part of silica by weight.
[0030] Precipitated silica generally is produced commercially by combining an aqueous solution of a soluble metal silicate, ordinarily alkali metal silicate such as sodium silicate, and an acid so that colloidal particles of silica will grow in a weakly alkaline solution and be coagulated by the alkali metal ions of the resulting soluble alkali metal salt. Various acids may be used, including but not limited to mineral acids. Non-limiting examples of acids that can be used include hydrochloric acid and sulfuric acid, but carbon dioxide can also be used to produce precipitated silica. In the absence of a coagulant, silica is not precipitated from solution at any pH. The coagulant used to effect precipitation of silica may be the soluble alkali metal salt produced during formation of the colloidal silica particles, or it may be an added electrolyte, such as a soluble inorganic or organic salt, or it may be a combination of both.
[0031] Precipitated silica can be described as precipitated aggregates of ultimate particles of colloidal amorphous silica that have not at any point existed as macroscopic gel during the preparation. The sizes of the aggregates and the degree of hydration may vary widely. Precipitated silica powders differ from silica gels that have been pulverized in that the precipitated silica powders generally have a more open structure, that is, a higher specific pore volume. However, the specific surface area of precipitated silica, as measured by the Brunauer, Emmet, Teller (BET) method using nitrogen as the adsorbate, is often lower than that of silica gel.
[0032] Many different precipitated silicas can be employed as the filler used to prepare the microporous membrane. Precipitated silicas are well-known commercial materials, and processes for producing them are described in detail in many United States patents, including United States Patent Numbers 2,940,830 and 4,681 ,750. The average ultimate particle size (irrespective of whether or not the ultimate particles are agglomerated) of precipitated silicas used is generally less than 0.1 micrometer, e.g., less than 0.05 micrometer or less than 0.03 micrometer, as determined by transmission electron microscopy. Non-limiting examples of suitable precipitated silicas include those sold under the HI-SIL tradename by PPG (Pittsburgh, PA).
[0033] The finely divided, particulate, substantially water-insoluble filler 2) may be present in the microporous membrane in an amount of at least 10 percent by weight, or at least 30 percent by weight, or at least 50 percent by weight, and at most 90 percent by weight, or at most 80 percent by weight, or at most 75 percent by weight, based on the total weight of the microporous membrane. For example, the filler 2) may be present in the microporous membrane in an amount of 10 percent to 90 percent by weight, or 10 to 80 percent by weight, or 10 to 75 percent by weight, or 30 percent to 90 percent by weight, or 30 to 80 percent by weight, or 30 to 75 percent by weight, or 50 percent to 90 percent by weight, or 50 to 80 percent by weight, or 50 to 75 percent by weight, based on the total weight of the microporous membrane.
[0034] The microporous membrane of the present disclosure further comprises a network of interconnecting pores communicating throughout the microporous membrane.
[0035] On a treatment-free, coating free, or impregnant-free basis, such pores can make up at least 5 percent by volume, or at least 15 percent by volume, or at least 20 percent by volume, or at least 25 percent by volume, or at least 35 percent by volume, or at least 45 percent by volume, and at most 95 percent by volume, or at most 75
percent by volume. Thus, the pores can make up 5 to 95 percent by volume, or 15 to 95 percent by volume, or 20 to 95 percent by volume, or 25 to 95 percent by volume, or 35 to 95 percent by volume, or 45 to 95 percent by volume, or 5 to 70 percent by volume, or 15 to 70 percent by volume, or 20 to 70 percent by volume, or 25 to 70 percent by volume, or 35 to 70 percent by volume, or 45 to 70 percent by volume of the microporous material. Often, the pores comprise at least 35 percent by volume, or even at least 45 percent by volume of the microporous membrane.
[0036] Porosity can be measured using a Gurley Densometer, model 4340, manufactured by GPI Gurley Precision Instruments (Troy, NY). The porosity values reported are a measure of the rate of air flow through a sample or its resistance to an air flow through the sample. The unit of measure for this method is a “Gurley second” and represents the time in seconds to pass 100 cc of air through a 1 inch square area using a pressure differential of 4.88 inches of water. Lower values equate to less air flow resistance (more air is allowed to pass freely). For purposes of the present disclosure, the measurements are completed using the procedure listed in the manual for MODEL 4340 Automatic Densometer. Typically, the membrane demonstrates a Gurley porosity of 10 to 5000 seconds, or 250 to 5000 seconds, or 500 to 5000 seconds, or 1000 to 5000 seconds, or 10 to 4500 seconds, or 250 to 4500 seconds, or 500 to 4500 seconds, or 1000 to 4500 seconds.
[0037] The microporous membranes may further comprise a biodegradation promoting material, an antioxidant, a lubricant, a UV absorber and/or a UV light stabilizer distributed throughout the organic polymer matrix. Biodegradation promoting materials serve to render the microporous membrane compostable and/or biodegradable. Examples include furanone compounds, glutaric acids, hexadecanoic acid compounds, organoleptic swelling agents such as natural fibers, cultured colloids, cyclo-dextrin, and polylactic acid, catalytic transition metal compounds, metal stearates, and/or metal chelates. Examples of suitable lubricants include calcium stearate, calcium-zinc stearate, zinc stearate, magnesium stearate, zinc phosphate, and magnesium phosphate. Anti-oxidants such as those sold by BASF under the names IRGANOX® and IRGAFOS®, and those sold by Solvay under the name CYANOX®, are suitable for use. Non-limiting examples of suitable UV absorbers include those available under the tradename TINUVIN®, such as TINUVIN ® 1130, available from BASF, and HOSTAVIN® PR-25, available from Clariant. Examples of UV light stabilizers include benzophenones, benzotriazoles, and hindered amine light
stabilizers (HALS). Commercially available examples of UV stabilizers include HOSTAVIN® PR-31 , available from Clariant, TINUVIN® 900, available from BASF, and CYANOX® M528, available from Solvay.
[0038] The microporous membranes described above may be prepared by a method comprising:
(i) mixing the organic polymer matrix, filler, and a processing plasticizer until a substantially uniform mixture is obtained, wherein the processing plasticizer is present in an amount of 30 to 80 percent by weight based on the total weight of the mixture;
(ii) introducing the mixture, optionally with additional processing plasticizer, into a heated barrel of a screw extruder and extruding the mixture through a sheeting die to form a continuous sheet;
(iii) forwarding the continuous sheet formed by the die to a pair of heated calender rolls acting cooperatively to form a continuous sheet of lesser thickness than the continuous sheet exiting from the die;
(iv) passing the sheet to a first extraction zone where the processing plasticizer is extracted from the sheet with an organic liquid;
(v) passing the continuous sheet to a second extraction zone where residual organic extraction liquid is extracted from the sheet by steam and/or water;
(vi) passing the continuous sheet through a dryer for substantial removal of residual water and remaining residual organic extraction liquid; and
(vii) optionally stretching the continuous sheet in at least one stretching direction above the elastic limit, wherein the stretching occurs during or immediately after step (ii) and/or step (iii), but prior to step (iv), to form a microporous membrane. [0039] In an exemplary process, the components of the organic polymer matrix (typically in solid form such as powder or pellets), filler, processing plasticizer, and minor amounts of lubricant and antioxidant are mixed until a substantially uniform mixture is obtained. The weight ratio of filler to polymer employed in forming the mixture is essentially the same as that of the microporous membrane to be produced. The mixture, together with additional processing plasticizer as needed, is introduced to the heated barrel of a screw extruder. Attached to the extruder is a die, such as a sheeting die, to form the desired end shape.
[0040] When the material is formed into a sheet or film, a continuous sheet or film formed by a die may be forwarded to a pair of heated calender rolls acting
cooperatively to form a continuous sheet of lesser thickness than the continuous sheet exiting from the die. The final thickness may depend on the desired end-use application. The microporous membrane may have a thickness ranging from 10 to 510 microns, such as 15 to 460 microns, or 15 to 390 microns, or 25 to 260 microns, or 125 to 250 microns.
[0041] Optionally, the sheet exiting the calendar rolls may then be stretched in at least one stretching direction above the elastic limit. Stretching may alternatively take place during or immediately after exiting from the sheeting die or during calendaring, or multiple times during the manufacturing process. Stretching may take place before extraction, after extraction, or both. Additionally, stretching may take place during the application of the pre-treatment composition and/or treatment composition, described in more detail below. Stretched microporous material substrate may be produced by stretching the intermediate product in at least one stretching direction above the elastic limit. Usually, the stretch ratio is at least 1 .2. In many cases, the stretch ratio is at least 1 .5. Often it is at least 2. Frequently, the stretch ratio is in the range of from 1 .2 to 15. Often, the stretch ratio is in the range of from 1 .5 to 10. Usually, the stretch ratio is in the range of from 2 to 6.
[0042] The temperatures at which stretching is accomplished may vary widely. Stretching may be accomplished at ambient room temperature, but usually elevated temperatures are employed. Ambient temperature typically ranges from 60 to 90 °F (15.6 to 32.2 °C), such as a typical room temperature, 72°F (22.2°C). The intermediate product may be heated by any of a wide variety of techniques prior to, during, and/or after stretching. Examples of these techniques include radiative heating, such as that provided by electrically heated or gas fired infrared heaters; convective heating, such as that provided by recirculating hot air; and conductive heating, such as that provided by contact with heated rolls. The temperatures which are measured for temperature control purposes may vary according to the apparatus used and personal preference. For example, temperature-measuring devices may be placed to ascertain the temperatures of the surfaces of infrared heaters, the interiors of infrared heaters, the air temperatures of points between the infrared heaters and the intermediate product, the temperatures of circulating hot air at points within the apparatus, the temperature of hot air entering or leaving the apparatus, the temperatures of the surfaces of rolls used in the stretching process, the temperature of heat transfer fluid entering or leaving such rolls, or film surface temperatures. In general, the temperature or temperatures
are controlled such that the intermediate product is stretched about evenly so that the variations, if any, in film thickness of the stretched microporous material are within acceptable limits and so that the amount of stretched microporous material outside of those limits is acceptably low. It will be apparent that the temperatures used for control purposes may or may not be close to those of the intermediate product itself since they depend upon the nature of the apparatus used, the locations of the temperaturemeasuring devices, and the identities of the substances or objects whose temperatures are being measured.
[0043] In view of the locations of the heating devices and the line speeds usually employed during stretching, gradients of varying temperatures may or may not be present through the thickness of the intermediate product. Also, because of such line speeds, it is impracticable to measure these temperature gradients. The presence of gradients of varying temperatures, when they occur, makes it unreasonable to refer to a singular film temperature. Accordingly, film surface temperatures, which can be measured, are best used for characterizing the thermal condition of the intermediate product.
[0044] The film surface temperatures at which stretching is accomplished may vary widely, but in general they are such that the intermediate product is stretched about evenly, as explained above. In most cases, the film surface temperatures during stretching are in the range of from 20°C to 220°C. Often, such temperatures are in the range of from 50°C to 200°C, such as from 75°C to 180°C.
[0045] Stretching may be accomplished in a single step or a plurality of steps as desired. For example, when the intermediate product is to be stretched in a single direction (uniaxial stretching), the stretching may be accomplished by a single stretching step or a sequence of stretching steps until the desired final stretch ratio is attained. Similarly, when the intermediate product is to be stretched in two directions (biaxial stretching), the stretching can be conducted by a single biaxial stretching step or a sequence of biaxial stretching steps until the desired final stretch ratios are attained. Biaxial stretching may also be accomplished by a sequence of one of more uniaxial stretching steps in one direction and one or more uniaxial stretching steps in another direction. Biaxial stretching steps where the intermediate product is stretched simultaneously in two directions and uniaxial stretching steps may be conducted in sequence in any order. Stretching in more than two directions is within contemplation. It may be seen that the various permutations of steps are quite numerous. Other steps,
such as cooling, heating, sintering, annealing, reeling, unreeling, and the like, may optionally be included in the overall process as desired.
[0046] Various types of stretching apparatus are well known and may be used to accomplish stretching of the intermediate product. Uniaxial stretching is usually accomplished by stretching between two rollers, wherein the second or downstream roller rotates at a greater peripheral speed than the first or upstream roller. Uniaxial stretching can also be accomplished on a standard tentering machine. Biaxial stretching may be accomplished by simultaneously stretching in two different directions on a tentering machine. More commonly, however, biaxial stretching is accomplished by first uniaxially stretching between two differentially rotating rollers as described above, followed by either uniaxially stretching in a different direction using a tenter machine or by biaxially stretching using a tenter machine. The most common type of biaxial stretching is where the two stretching directions are approximately at right angles to each other. In most cases where the continuous sheet is being stretched, one stretching direction is at least approximately parallel to the long axis of the sheet (machine direction) and the other stretching direction is at least approximately perpendicular to the machine direction and is in the plane of the sheet (transverse direction).
[0047] Stretching the sheets prior to extraction of the processing plasticizer allows for thinner films with larger pore sizes than in microporous materials conventionally processed. It is also believed that stretching of the sheets prior to extraction of the processing plasticizer minimizes thermal shrinkage after processing. It also should be noted that stretching of the microporous membrane can be conducted at any point prior to, during, or subsequent to application of the pre-treatment composition (as described herein below), and/or prior to, during, or subsequent to application of the treatment composition. Stretching of the microporous membrane can occur once or multiple times during the treatment process.
[0048] The product passes to a first extraction zone where the processing plasticizer is extracted from the sheet with an organic liquid, which is a good solvent for the processing plasticizer, a poor solvent for the organic polymer, and more volatile than the processing plasticizer. Usually, but not necessarily, both the processing plasticizer and the organic extraction liquid are substantially immiscible with water. The product then passes to a second extraction zone where the residual organic extraction liquid is extracted from the sheet by steam and/or water. The product is then passed through
a forced air dryer for substantial removal of residual water and remaining residual organic extraction liquid. From the dryer, the microporous material may be passed to a take-up roll, when it is in the form of a sheet.
[0049] The processing plasticizer has little solvating effect on the organic polymers at 60°C, only a moderate solvating effect at elevated temperatures on the order of 100°C, and a significant solvating effect at elevated temperatures on the order of 200°C. It is a liquid at room temperature and usually it is processing oil, such as paraffinic oil, naphthenic oil, or aromatic oil. Suitable processing oils include those meeting the requirements of ASTM D 2226-82, Types 103 and 104. Those oils which have a pour point of less than 22°C, or less than 10°C, according to ASTM D 97-66 (reapproved 1978) are used most often. Examples of suitable oils include SHELLFLEX 412 and SHELLFLEX 371 oil (Shell Oil Co. (Houston, TX)), which are solvent refined and hydrotreated oils derived from naphthenic crude. It is expected that other materials, including the phthalate ester plasticizers such as dibutyl phthalate, bis(2- ethylhexyl) phthalate, diisodecyl phthalate, dicyclohexyl phthalate, butyl benzyl phthalate, and ditridecyl phthalate will function satisfactorily as processing plasticizers. [0050] There are many organic extraction liquids that can be used in the process of manufacturing the microporous membrane. Examples of suitable organic extraction liquids include, but are not limited to, 1 ,1 ,2-trichloroethylene; perchloroethylene; 1 ,2- dichloroethane; 1 ,1 ,1 -trichloroethane; 1 ,1 ,2-trichloroethane; methylene chloride; chloroform; 1 ,1 ,2-trichloro-1 ,2,2-trifluoroethane; isopropyl alcohol; diethyl ether; acetone; hexane; heptane and toluene. One or more azeotropes of halogenated hydrocarbons selected from trans-1 ,2-dichloroethylene, 1 ,1 ,1 ,2,2,3, 4,5, 5,5- decafluoropentane, and/or 1 ,1 ,1 ,3,3-pentafluorobutane also can be employed. Such materials are available commercially as VERTREL MCA (a binary azeotrope of 1 ,1 ,1 ,2,2,3,4,5,5,5-dihydrodecafluoropentane and trans-1 ,2-dichloroethylene: 62%/38%) and VERTREL CCA (a ternary azeotrope of 1 ,1 ,1 ,2, 2, 3, 4, 5,5,5- dihydrodecafluorpentane, 1 ,1 ,1 ,3,3-pentafluorbutane, and trans-1 ,2-dichloroethylene: 33%/28%/39%); VERTREL SDG (80-83% trans-1 ,2-dichloroethylene, 17-20% hydrofluorocarbon mixture), all available from MicroCare Corporation (New Britain, CT).
[0051] In the above-described process for producing microporous membrane, extrusion and calendering are facilitated when the filler carries much of the processing plasticizer. The capacity of the filler particles to absorb and hold the processing
plasticizer is a function of the surface area of the filler. Therefore, the filler typically has a high surface area as discussed above. Inasmuch as it is desirable to essentially retain the filler in the microporous material substrate, the filler should be substantially insoluble in the processing plasticizer and substantially insoluble in the organic extraction liquid when microporous material substrate is produced by the above process. The residual processing plasticizer content is usually less than 15 percent by weight of the resulting microporous material and this may be reduced even further to levels, such as less than 5 percent by weight, by additional extractions using the same or a different organic extraction liquid. The resulting microporous membranes may be further processed depending on the desired application.
[0052] The microporous membranes described above may be used to prepare a multilayer membrane comprising two or more adjacent membrane layers. The layers may be the same as or different from each other, and at least one layer comprises a microporous membrane described above. For example, different embodiments of the microporous membrane described above may be used as separate layers, or the layers may be the same. The use of different membrane layers allows for the specific properties afforded by each membrane to be incorporated into the final multilayer product.
[0053] In certain examples, at least one layer of the multilayer membrane may comprise a microporous polyolefinic material, wherein the polyolefinic material comprises linear ultrahigh molecular weight polyethylene, linear ultrahigh molecular weight polypropylene, or a mixture thereof; and finely divided, particulate, substantially water-insoluble silica filler distributed throughout the microporous polyolefinic material. Such polyolefinic materials are available from PPG as TESLIN®.
[0054] Examples of other suitable materials that may be used as an additional layer include porous thermoplastic polymeric sheet or film, porous thermoset polymeric sheet or film, porous elastomeric sheet or film, and open celled foams. Other examples include fabrics, such as woven fabrics, knitted fabrics, nonwoven fabrics, and scrims. Still other examples include fiber mats, paper, synthetic paper, felt, and the like. Further examples of suitable porous materials include materials which are microporous such as the stretched microporous materials and precursor microporous materials described above, as well as other materials which are microporous. Regarding porous materials which may be based on fibers, the fibers may be natural, such as wood fibers, cotton fibers, wool fibers, silk fibers, and the like; or they may be
any of the artificial fibers such as polyester fibers, polyamide fibers, acrylic fibers, modacrylic fibers, rayon fibers, and the like; or they may be combinations of differing kinds of fibers. The fibers may be staple and/or they may be continuous. Metal fibers, carbon fibers, and glass fibers are also suitable.
[0055] The multilayer membrane may be constructed by bonding at least one layer of microporous material to at least one layer of porous material. The multilayer membrane may comprise one layer or more than one layer of microporous material and one layer or more than one layer of the porous material. The multilayer membrane may also optionally be bonded to one or more layers of porous material. For example, the microporous material may be bonded to a scrim, which is bonded to a nonwoven fabric. In this case, the microporous material is often also bonded through the open areas of the scrim to the nonwoven fabric. As another example, the microporous material is bonded to a nonwoven fabric, the other side of which is bonded to a scrim. The combinations and permutations of possible layers are quite numerous. Most often, but not always, at least one exterior layer is microporous material.
[0056] Bonding may be made by conventional techniques such as, for example, fusion bonding and adhesive bonding. Examples of fusion bonding include sealing through use of heated rollers, heated bars, heated plates, heated bands, heated wires, flame bonding, radio frequency (RF) sealing, and ultrasonic sealing. Heat sealing is used most often. Solvent bonding may be used where the porous material is soluble in the applied solvent at least to the extent that the surface becomes tacky. After the microporous material has been brought into contact with the tacky surface, the solvent is removed to form a fusion bond.
[0057] Many adhesives which are well known may be used to accomplish bonding. Examples of suitable classes of adhesives include thermosetting adhesives, thermoplastic adhesive, adhesives which form the bond by solvent evaporation, adhesives which form the bond by evaporation of liquid nonsolvent, and pressure sensitive adhesives.
[0058] Foamable compositions may be foamed in contact with the microporous material to form a bond between the resulting porous foam and the microporous material.
[0059] Powder bonding is a technique which is particularly useful for bonding the microporous material to nonwoven webs of staple and/or continuous fibers and to woven or knitted fibers.
[0060] Depending upon the bonding technique employed, the adhesive employed, and/or the nature of the porous material used, the porous material may be readily removable from the microporous material by hand (i. e., peelable), or not (i. e., permanently bonded).
[0061] The microporous material may be continuously bonded to the porous material, or it may be discontinuously bonded to the porous material. Examples of discontinuous bonds include bonding areas in the form of one or more spots, patches, strips, stripes, open-curved stripes, closed-curved stripes, irregular areas, and the like. When patterns of bonds are involved, they may be random, repetitive, or a combination of both.
[0062] The multilayer membrane may also be formed by a continuous, moisture vapor permeable coating of hydrophobic polymer joined to one side of the microporous membrane described above. Such coatings and methods of applying them are described, for example, in United States Patent Number 5,032,450, which is incorporated herein by reference in its entirety. The coating is usually an elastomeric solid at ambient temperatures, and often comprises a polysiloxane. One surface of the coating may be in interlocking relationship with a surface of the microporous material, but the coating does not grossly penetrate into the interior of the microporous material. The resulting membrane is therefore perceived as a multi-layered article.
[0063] As used herein, a polymer is “hydrophobic” if the contact angle between a drop of water and a flat surface of the polymer upon which the drop is resting is greater than 90 degrees. Contact angle is discussed more fully in Sears and Zemansky, University Physics, Second Edition, Addison-Wesley Publishing Company, Inc., (1955), pages 231 -235.
[0064] The microporous membrane of the present disclosure is particularly useful for bonding to porous polyolefins such as polyethylene and polypropylene materials by heat sealing in the absence of extrinsic intervening adhesive. The bond obtained is ordinarily quite strong which is surprising inasmuch as the lamination of materials to polyolefins is usually difficult unless special adhesives are used.
[0065] The multilayer membranes of the present disclosure have many and varied uses including filters, wipers, gaskets, cushion assemblies, signs, printing substrate, substrates for pen and ink drawings, maps (particularly maritime maps), wall coverings, and breathable fabrics for clothing. They are also useful as or as components of bandages, sanitary napkins, incontinence devices, diapers, and the
like. They are also useful as the seams, joints, and seals of breathable packages, breathable wearing apparel and the like.
[0066] The present disclosure is further drawn to the following aspects:
1 . A microporous membrane comprising
1 ) an organic polymer matrix comprising:
(a) a polyolefin component, present in the organic polymer matrix in an amount of 18 to 99 percent by weight, based on the total weight of the organic polymer matrix, wherein the polyolefin component comprises ultrahigh molecular weight (UHMW) polyethylene; and
(b) an aliphatic polyketone polymer, present in the organic polymer matrix in an amount of 1 to 80 percent by weight, based on the total weight of the organic polymer matrix; and
2) finely divided, particulate, substantially water-insoluble filler distributed throughout said organic polymer matrix, present in the microporous membrane in an amount of 10 to 90 percent by weight, based on the total weight of the microporous membrane.
2. The microporous membrane of aspect 1 , wherein the polyolefin component is present in the organic polymer matrix in an amount of 18 to 97 percent by weight, based on the total weight of the organic polymer matrix.
3. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 18 to 95 percent by weight, based on the total weight of the organic polymer matrix.
4. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 25 to 99 percent by weight, based on the total weight of the organic polymer matrix.
5. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 25 to 97 percent by weight, based on the total weight of the organic polymer matrix.
6. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 25 to 95 percent by weight, based on the total weight of the organic polymer matrix.
7. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 40 to 99 percent by weight, based on the total weight of the organic polymer matrix.
8. The microporous membrane of any preceding aspect, wherein the polyolefin component is present in the organic polymer matrix in an amount of 40 to 97 percent by weight, based on the total weight of the organic polymer matrix.
9. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 3 to 80 percent by weight, based on the total weight of the organic polymer matrix.
10. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 5 to 80 percent by weight, based on the total weight of the organic polymer matrix.
11 . The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 1 to 75 percent by weight, based on the total weight of the organic polymer matrix.
12. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 3 to 75 percent by weight, based on the total weight of the organic polymer matrix.
13. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 5 to 75 percent by weight, based on the total weight of the organic polymer matrix.
14. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 1 to 60 percent by weight, based on the total weight of the organic polymer matrix.
15. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 3 to 60 percent by weight, based on the total weight of the organic polymer matrix.
16. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer is present in the organic polymer matrix in an amount of 5 to 60 percent by weight, based on the total weight of the organic polymer matrix.
17. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 10 to 80 percent by weight, based on the total weight of the microporous membrane.
18. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 10 to 75 percent by weight, based on the total weight of the microporous membrane.
19. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 30 to 90 percent by weight, based on the total weight of the microporous membrane.
20. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 30 to 80 percent by weight, based on the total weight of the microporous membrane.
21 . The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 30 to 75 percent by weight, based on the total weight of the microporous membrane.
22. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler distributed throughout said organic polymer matrix is present in the microporous membrane in an amount of 50 to 90 percent by weight, based on the total weight of the microporous membrane.
23. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 50 to 80 percent by weight, based on the total weight of the microporous membrane.
24. The microporous membrane of any preceding aspect, wherein the finely divided, particulate, substantially water-insoluble filler is present in the microporous membrane in an amount of 50 to 75 percent by weight, based on the total weight of the microporous membrane.
25. The microporous membrane of any preceding aspect, wherein the polyolefin component further comprises high density polyethylene (HDPE).
26. The microporous membrane of any preceding aspect, wherein the HDPE is present in the organic polymer matrix in an amount of from 0.1 to 74.25 percent by weight, based on the total weight of the organic polymer matrix.
27. The microporous membrane of any preceding aspect, wherein the HDPE is present in the organic polymer matrix in an amount of from 0.1 to 60 percent by weight, based on the total weight of the organic polymer matrix.
28. The microporous membrane of any preceding aspect, wherein the HDPE is present in the organic polymer matrix in an amount of from 0.1 to 45 percent by weight, based on the total weight of the organic polymer matrix.
29. The microporous membrane of any preceding aspect, wherein the HDPE is present in the organic polymer matrix in an amount of from 1 to 74.25 percent by weight, based on the total weight of the organic polymer matrix.
30. The microporous membrane of any preceding aspect, wherein the HDPE is present in the organic polymer matrix in an amount of from 1 to 60 percent by weight, based on the total weight of the organic polymer matrix.
31 . The microporous membrane of any preceding aspect, wherein the HDPE is present in the organic polymer matrix in an amount of from 1 to 45 percent by weight, based on the total weight of the organic polymer matrix.
32. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer demonstrates a melt index of 0.5 to 4 g/10 min, measured according to ASTM D1238-23.
33. The microporous membrane of any preceding aspect, wherein the aliphatic polyketone polymer demonstrates a melt temperature of 190 to 220 °C, measured according to ASTM D3418-21 .
34. The microporous membrane of any preceding aspect, wherein the filler comprises silica, talc, carbon black, charcoal, graphite, titanium oxide, iron oxide, copper oxide, zinc oxide, antimony oxide, zirconia, magnesia, alumina, molybdenum disulfide, zinc sulfide, barium sulfate, strontium sulfate, calcium carbonate, and/or magnesium carbonate.
35. The microporous membrane of any preceding aspect, wherein the filler comprises silica and/or calcium carbonate.
36. The microporous membrane of any preceding aspect, wherein the filler comprises precipitated silica.
37. The microporous membrane of any preceding aspect, wherein the filler comprises silica and calcium carbonate, and a weight ratio of silica to calcium carbonate ranges from 0.2:1 to 1 :0.2.
38. The microporous membrane of any preceding aspect, wherein the filler comprises silica and wherein the silica demonstrates a BET of 125 to 700 m2/g as determined by Brunauer, Emmett, Teller (BET) method according to ASTM C 819-77.
39. The microporous membrane of any preceding aspect, wherein the microporous membrane has a thickness ranging from 10 to 510 microns.
40 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 10 to 5000 seconds.
41 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 250 to 5000 seconds.
42 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 500 to 5000 seconds.
43 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 1000 to 5000 seconds.
44 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 10 to 4500 seconds.
45 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 250 to 4500 seconds.
46 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 500 to 4500 seconds.
47 . The microporous membrane of any previous aspect, wherein the membrane demonstrates a Gurley porosity of 1000 to 4500 seconds.
48. The microporous membrane of any preceding aspect, further comprising a biodegradation promoting material distributed throughout the organic polymer
matrix, thereby rendering the microporous membrane compostable and/or biodegradable.
49. The microporous membrane of aspect 48, wherein the biodegradation promoting material comprises a furanone compound, a glutaric acid, a hexadecanoic acid compound, an organoleptic swelling agent, a catalytic transition metal compound, a metal stearate, and/or a metal chelate.
50. The microporous membrane of any preceding aspect, further comprising an antioxidant, a lubricant, a UV absorber and/or a UV light stabilizer distributed throughout the organic polymer matrix.
51 . A multilayer membrane comprising two or more adjacent membrane layers, wherein the layers are the same as or different from each other, and wherein at least one layer comprises the microporous membrane of any preceding aspect.
52. The multilayer membrane of aspect 51 , wherein at least one layer comprises a microporous polyolefinic material, and wherein the polyolefinic material comprises linear ultrahigh molecular weight polyethylene, linear ultrahigh molecular weight polypropylene, or a mixture thereof; and finely divided, particulate, substantially water-insoluble silica filler distributed throughout the microporous polyolefinic material.
[0067] The following examples are intended to illustrate various aspects of the disclosure, and should not be construed as limiting the disclosure in any way. Components that are mentioned elsewhere in the specification as suitable alternative materials for use, but which are not demonstrated in the working Examples below, are expected to provide results comparable to their demonstrated counterparts. Unless otherwise indicated, all parts are by weight.
EXAMPLES
Mix preparation
[0068] The components listed for each example and comparative example in Tables 1 and 2, scaled to a total of 2230 g, were weighed into a Littleford FT-130 Henschel horizontal mixer with one high intensity chopper style mixing blade. The mixture was premixed for approximately 15 seconds using the plough blades only. Heated process oil, (TUFFLO® 6066 process oil, commercially available from PPC Lubricants) was pumped into mixer over 40-60 seconds, with only the plough blades running. The high intensity chopper blade was turned on, along with the plough blades, and the components were mixed for 30 seconds. The mixer was shut off and the internal sides of the mixer were scraped down to ensure all ingredients were evenly mixed. The mixer was restarted with both high intensity chopper and plough blades turned on, and the mix was mixed for an additional 30 seconds. The mixer was turned off and the mix transferred into a storage container.
Extrusion, Calendering and Extraction
[0069] The mixes of the examples were extruded and calendered into final sheet form using an extrusion system including a feeding, extrusion and calendering system described as follows. A gravimetric loss in weight feed system (K-Tron model K2MLT35D5) was used to feed each of the respective mixes into a 27 mm twin screw extruder Leistritz Micro-27gg. The extruder barrel was comprised of eight temperature zones and a heated adaptor to the sheet die. The extrusion mixture feed port was located just prior to the first temperature zone. An atmospheric vent was located in the third temperature zone. A vacuum vent was located in the seventh temperature zone. [0070] The mix was fed into the extruder at a rate of 90 g/minute. Additional processing oil also was injected at the first temperature zone, as required, to achieve a total amount of 60-100 parts by weight based on the solid components in Tables 2 and 3.
[0071] Extrudate from the barrel was discharged into a 15-centimeter wide sheet Masterflex® die having a 1.5 millimeter discharge opening. The extrusion melt temperature was 203- 210 °C and the throughput was 7.5 kilograms per hour.
[0072] The calendering process was accomplished using a three-roll vertical calender stack with one nip point and one cooling roll. Each of the rolls had a chrome surface. Roll dimensions were approximately 41 cm in length and 14 cm in diameter. The top roll temperature was maintained between 135 °C to 140 °C. The middle roll
temperature was maintained between 140 °C. to 145 °C. The bottom roll was a cooling roll wherein the temperature was maintained between 10-21 °C. The extrudate was calendered into sheet form and passed over the bottom water-cooled roll and wound up.
[0073] A sample of sheet cut to a width of approximately 18 cm and an approximate length of 150 cm was rolled up along with a stainless steel wire mesh into a cylinder shape, placed in a canister and exposed to room temperature liquid 1 ,1 ,2- trichloroethylene for approximately 1 hour to extract processing oil from the sheet sample. The remaining oil content in the samples was from 3-7 wt. %. The extracted sheet was air dried and subjected to test methods described hereinafter.
1 . HI-SIL 135® precipitated silica obtained commercially from PPG Industries, Inc.
2. GUR® 4160 Ultra High Molecular Weight Polyethylene (UHMWPE) commercially available from Ticona Corp, and reported to have a molecular weight of about 9.2 million grams per mole.
3.An aliphatic polyketone with a reported melt flow rate 220 °C/2.16 Kg of 3 g/10 min and reported melt temperature of approximately 200°C, commercially available from Hyosung Chemical with trade name Poketone.
4. TIPURE™ R-103 titanium dioxide commercially available from E.L du Pont de Nemours and Company.
5. An antioxidant commercially available from Cytec Industries, Inc.
6. technical grade.
7. Cyasorb Synergy Solutions® M528 light stabilizing additive commercially available from Solvay.
8. FINA® 1288 High Density Polyethylene (HDPE) commercially available from Total Petrochemicals.
[0074] All compositions except CE-2 yielded smooth continuous microporous sheets. CE-2 did not survive the extrusion conditions and was not subject to testing.
Testing and Results
[0075] Physical properties measured on the extracted and dried films and the results obtained are listed in Table 3. Thickness was determined using an Ono Sokki thickness gauge EG-225. Two 4.5 x 5 inch (11 .43 cm x 12.7 cm) specimens were cut from each sample and the thickness for each specimen was measured in nine places (at least % of an inch (1 .91 cm) from any edge). The arithmetic average of the readings was recorded in mils.
[0076] The Maximum Elongation or tensile modulus of elasticity and the Maximum Tensile Strength or tensile energy to break the samples was tested in accordance with ASTM D882-18 modified by using a sample crosshead speed of 5.08 cm/minute until 0.508 cm of linear travel speed was completed, at which time the crosshead speed was accelerated to 50.8 cm/second, and, where the sample width was approximately 1 .2 cm and the sample gauge length is 5.08 cm. Measurements were taken in both machine direction orientation (MD) from samples whose major axis was oriented along the length of the sheet, and cross direction orientation (CD) from samples whose major axis was oriented across the sheet.
[0077] Gurley porosity was determined using a Gurley densometer, model 4340, manufactured by GPI Gurley Precision Instruments of Troy, N.Y. The air flow rate reported was a measure of the rate of air flow through a sample of the example or it' s resistance to an air flow through the sample. The unit of measure is a "Gurley second" and represents the time in seconds to pass 100 cc of air through a 1 inch square area using a pressure differential of 4.88 inches of water. Lower values equate to less air flow resistance (more air is allowed to pass freely). The measurements were completed using the procedure listed in the manual, MODEL 4340 Automatic Densometer and Smoothness Tester Instruction Manual. As shown in Tables 3 and 4, when using polyketone, highly porous sheets were produced.
9. CE-2, with 100% polyketone and no polyolefin in the organic polymer matrix was not processable by the listed method.
[0078] As demonstrated in Tables 3 and 4, compositions with polyolefin and polyketone produced excellent sheets with good strength.
UV Stability testing
[0079] The samples were tested following common UVA-340 test methods for QUV accelerated weathering in accordance with SAE J2020 (UVA). Samples were mounted on 6 x 3 inch aluminum Q-panels and taped only at the top and bottom, half inch from the edge, of the panel to keep the sheets stable. The panels were exposed to ultraviolet light (UV) irradiance of approximately 0.72 W/m2 in a weatherometer equipped with UVA-340 lamps. A cycle of 8 hours at 70 °C UV exposure with a conditioning of 4 hours at 50 °C was used.
[0080] After the determined duration of exposure, e.g. 336 hours, the panels were removed from the testing cabinet and the sheets carefully released from the aluminum panels by removing the holding adhesive tape on the top and bottom parts of the panel. The 3 x 6 inch size sheets were cut into 3 strips of equal size using a die cutter into approximately 0.5 x 4.5 inch (1.2 x 5.08 cm) strips where the length of the strip
represented the MD. To test the properties of the CD the 0.5 x 4.5 inch (1 .2 x 5.08 cm) strips were cut in an orientation in corresponding to the CD orientation of the produced microporous sheet. Each strip was tested individually to give a total of three tensile readings from the three strips and the arithmetic average calculated from the data in accordance with ASTM D882-18, as previously described, to obtain their corresponding maximum tensile strength and maximum tensile elongation.
[0081] As illustrated in Table 5 formulations comprising the aliphatic polyketone exhibited significantly enhanced UV stability. Microporous substrates according to the disclosure demonstrated at least a two-fold increase in UV stability, some surviving up to 1500 hours of UV exposure time while the comparative examples disintegrated at or shortly after 336 hours under the same UV exposure conditions. All examples which contain polyketone according to the disclosure maintain stability through UV exposure far beyond those without polyketone regardless of the tensile strength prior to testing. Additionally, Example 11 demonstrates that a light stabilizer was not required to significantly improve UV stability compared to Example 10.
(10) A value of “0” indicates that the sheet was intact at the specified time interval but was unable to undergo testing; e.g. the sheet broke before or at the onset of testing.
(11) An empty cell indicates (unless otherwise indicated) that the sheet was disintegrated at the specified time interval and therefore could not be prepared for testing.
[0082] Thermal stability property was measured by tensile modulus of elasticity retention as shown in Table 6. The thermoforming properties of the sheets were predictably superior to the comparative. High temperature tensile measurements showed a more stable microporous sheet at 155 °C at both low and high strain rates as illustrated by CE-7 vs Example 10.
[0083] The results in Table 6 demonstrate that the membranes according to the disclosure retain elongation at elevated temperatures, wherein the comparative example CE-7 actually lost flexibil ity/elongation at elevated temperatures. At a higher strain rate (10 in/min compared to 2 in/min) Example 10 exhibits yet higher elongation.
[0084] Whereas particular examples of this disclosure have been described above for purposes of illustration, it will be evident to those skilled in the art that numerous variations of the details of the present disclosure may be made without departing from the disclosure as defined in the appended claims.
Claims
1 . A microporous membrane comprising
1 ) an organic polymer matrix comprising:
(a) a polyolefin component, present in the organic polymer matrix in an amount of 18 to 99 percent by weight, based on the total weight of the organic polymer matrix, wherein the polyolefin component comprises ultrahigh molecular weight (UHMW) polyethylene; and
(b) an aliphatic polyketone polymer, present in the organic polymer matrix in an amount of 1 to 80 percent by weight, based on the total weight of the organic polymer matrix; and
2) finely divided, particulate, substantially water-insoluble filler having gross particle sizes in the range of from 5 to 40 micrometers, as determined by the use of a laser diffraction particle size instrument, wherein said filler is distributed throughout said organic polymer matrix, and is present in the microporous membrane in an amount of 10 to 90 percent by weight, based on the total weight of the microporous membrane.
2. The microporous membrane of claim 1 , wherein the polyolefin component further comprises high density polyethylene (HDPE).
3. The microporous membrane of claim 1 or 2, wherein the HDPE is present in the organic polymer matrix in an amount of from 0.1 to 74.25 percent by weight, based on the total weight of the organic polymer matrix.
4. The microporous membrane of any of claims 1 to 3, wherein the aliphatic polyketone polymer demonstrates a melt index of 0.5 to 4 g/10 min, measured according to ASTM D1238-23.
5. The microporous membrane of claim 4, wherein the aliphatic polyketone polymer demonstrates a melt temperature of 190 to 220 °C, measured according to ASTM D3418-21 .
6. The microporous membrane of any of claims 1 to 5, wherein the filler comprises silica, talc, carbon black, charcoal, graphite, titanium oxide, iron oxide, copper oxide, zinc oxide, antimony oxide, zirconia, magnesia, alumina, molybdenum disulfide, zinc sulfide, barium sulfate, strontium sulfate, calcium carbonate, and/or magnesium carbonate.
7. The microporous membrane of claim 6, wherein the filler comprises silica and/or calcium carbonate.
8. The microporous membrane of claim 6 or 7, wherein the filler comprises precipitated silica.
9. The microporous membrane of claim 7, wherein the filler comprises silica and calcium carbonate, and a weight ratio of silica to calcium carbonate ranges from 0.2:1 to 1 :0.2.
10. The microporous membrane of any of claims 6 to 9, wherein the filler comprises silica and wherein the silica demonstrates a BET of 125 to 700 m2/g as determined by Brunauer, Emmett, Teller (BET) method according to ASTM C 819-77.
11. The microporous membrane of any of claims 1 to 10, wherein the microporous membrane has a thickness ranging from 10 to 510 microns .
12 . The microporous membrane of any of claims 1 to 11 , wherein the membrane demonstrates a Gurley porosity of 10 to 5000 seconds.
13. The microporous membrane of any of claims 1 to 12, further comprising a biodegradation promoting material distributed throughout the organic polymer matrix, thereby rendering the microporous membrane compostable and/or biodegradable.
14. The microporous membrane of claim 13 wherein the biodegradation promoting material comprises a furanone compound, a glutaric acid, a hexadecanoic
acid compound, an organoleptic swelling agent, a catalytic transition metal compound, a metal stearate, and/or a metal chelate.
15. The microporous membrane of any of claims 1 to 14, further comprising an antioxidant, a lubricant, a UV absorber and/or a UV light stabilizer distributed throughout the organic polymer matrix.
16. A multilayer membrane comprising two or more adjacent membrane layers, wherein the layers are the same as or different from each other, and wherein at least one layer comprises the microporous membrane of any of claims 1 to 15.
17. The multilayer membrane of claim 16, wherein at least one layer comprises a microporous polyolefinic material, and wherein the polyolefinic material comprises linear ultrahigh molecular weight polyethylene, linear ultrahigh molecular weight polypropylene, or a mixture thereof; and finely divided, particulate, substantially water-insoluble silica filler distributed throughout the microporous polyolefinic material.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363609382P | 2023-12-13 | 2023-12-13 | |
| US63/609,382 | 2023-12-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025128283A1 true WO2025128283A1 (en) | 2025-06-19 |
Family
ID=93799535
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/056327 Pending WO2025128283A1 (en) | 2023-12-13 | 2024-11-18 | Microporous membranes demonstrating improved durability and methods of preparing same |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025128283A1 (en) |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2940830A (en) | 1955-08-23 | 1960-06-14 | Columbia Southern Chem Corp | Method of preparing silica pigments |
| US4681750A (en) | 1985-07-29 | 1987-07-21 | Ppg Industries, Inc. | Preparation of amorphous, precipitated silica and siliceous filler-reinforced microporous polymeric separator |
| US5032450A (en) | 1990-01-31 | 1991-07-16 | Ppg Industries, Inc. | Microporous material having a coating of hydrophobic polymer |
| US20110256364A1 (en) * | 2010-04-15 | 2011-10-20 | Ppg Industries Ohio, Inc. | Microporous material |
-
2024
- 2024-11-18 WO PCT/US2024/056327 patent/WO2025128283A1/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2940830A (en) | 1955-08-23 | 1960-06-14 | Columbia Southern Chem Corp | Method of preparing silica pigments |
| US4681750A (en) | 1985-07-29 | 1987-07-21 | Ppg Industries, Inc. | Preparation of amorphous, precipitated silica and siliceous filler-reinforced microporous polymeric separator |
| US5032450A (en) | 1990-01-31 | 1991-07-16 | Ppg Industries, Inc. | Microporous material having a coating of hydrophobic polymer |
| US20110256364A1 (en) * | 2010-04-15 | 2011-10-20 | Ppg Industries Ohio, Inc. | Microporous material |
Non-Patent Citations (1)
| Title |
|---|
| SEARSZEMANSKY: "University Physics", 1955, ADDISON-WESLEY PUBLISHING COMPANY, INC., pages: 231 - 235 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP2558520B1 (en) | Microporous material | |
| AU717212B2 (en) | Very thin microporous material | |
| US5032450A (en) | Microporous material having a coating of hydrophobic polymer | |
| JP5202816B2 (en) | Polyolefin microporous membrane and method for producing the same | |
| JP5567535B2 (en) | Porous polypropylene film | |
| CA1316314C (en) | Stretched microporous material | |
| US20080286593A1 (en) | Multi-layer articles prepared from microporous materials | |
| US10857252B2 (en) | Microporous material | |
| US20070138682A1 (en) | Microporous film of semicrystalline polymers and method for preparing the same | |
| CA2925895A1 (en) | Microporous material | |
| US20090311504A1 (en) | Microporous materials and multi-layer articles prepared therefrom | |
| JP2016089009A (en) | Moisture permeable film and moisture permeable film laminate | |
| WO2025128283A1 (en) | Microporous membranes demonstrating improved durability and methods of preparing same | |
| JP5705605B2 (en) | Polypropylene resin porous sheet and method for producing polypropylene resin porous sheet | |
| JP2024144193A (en) | Stretched porous film, roll, sanitary product, and method for producing stretched porous film | |
| JP7135709B2 (en) | porous film | |
| JP2024144192A (en) | Stretched porous film, roll, sanitary product, and method for producing stretched porous film | |
| JP2024142061A (en) | Stretched porous film, roll, disposable warmer, and method for producing stretched porous film | |
| JP7172498B2 (en) | porous film | |
| KR20080064251A (en) | Breathable film | |
| JP2023113225A (en) | Polyethylene-based porous stretched film | |
| JP2025135508A (en) | Stretched film and method for producing the same | |
| JP2024133997A (en) | Stretched porous film and method for producing same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24818129 Country of ref document: EP Kind code of ref document: A1 |